User login
FDA has the resources to answer questions about medicines and pregnancy
There are about 6 million pregnancies in the United States each year, and it’s estimated that 50% of pregnant women take at least one medicine. Physicians play an important role in helping their pregnant patients make informed health choices, especially when it comes to the safe use of medications.
- Emphasize that patients should always talk to their health care provider before taking any medicines, herbs, or vitamins. The website provides tips on how to start the conversation with patients about what medicines, herbs, or vitamins to avoid when pregnant.
- Encourage patients to check the drug label and other information that comes with their medicine to learn about possible risks for women who are pregnant or breastfeeding.
- Assist pregnant patients with changing medications as needed.
- Advise pregnant patients if they need to take more or less of their medicines.
- Advise patients about medicines that can and cannot be used when they start breastfeeding.
- Encourage patients to talk about any problems they have with their medicine.
- Report any serious problems your pregnant patients have had after taking a medicine to the FDA. It falls to physicians to report to the FDA any cases of serious side effects, problems with product quality, and product-use errors or with any of the following products: human drugs, medical devices, blood products and other biologics (except vaccines), and/or medical foods.
- Encourage patients to enroll in a Pregnancy Exposure Registry, if applicable at the FDA website, which collects information on pregnancy outcomes in women who already are taking medications. Observational studies of the patients that physicians help enroll in a pregnancy exposure registry can improve drug safety information for medicines used during pregnancy and can be used to update drug labeling. The observational studies included with the registries can also help physicians make medicine recommendations for use during pregnancy. The list includes contact information for each registry. Physicians can check online to see if there is a registry for their patients’ medicine.
The FDA website also provides information about the Pregnancy and Lactation Labeling Final Rule, which requires changes to the content and format for information presented in prescription drug labeling. The changes are implemented to help health care providers assess risk versus benefit and in subsequent counseling of pregnant women and nursing mothers.
The FDA offers free medicine safety and pregnancy resources for pregnant women, including downloadable infographics; a Medicines Record Keeper brochure in English, Spanish
Pregnancy is an exciting time for women, but they may have questions and concerns about how medicines they take will affect their babies. Our pregnancy website can help make a woman’s pregnancy happier and healthier.
Dr. Yao is the director of the division of pediatrics and maternal health, Office of Drug Evaluation IV, Center for Drug Evaluation and Research at the FDA, in Silver Spring, Md.
There are about 6 million pregnancies in the United States each year, and it’s estimated that 50% of pregnant women take at least one medicine. Physicians play an important role in helping their pregnant patients make informed health choices, especially when it comes to the safe use of medications.
- Emphasize that patients should always talk to their health care provider before taking any medicines, herbs, or vitamins. The website provides tips on how to start the conversation with patients about what medicines, herbs, or vitamins to avoid when pregnant.
- Encourage patients to check the drug label and other information that comes with their medicine to learn about possible risks for women who are pregnant or breastfeeding.
- Assist pregnant patients with changing medications as needed.
- Advise pregnant patients if they need to take more or less of their medicines.
- Advise patients about medicines that can and cannot be used when they start breastfeeding.
- Encourage patients to talk about any problems they have with their medicine.
- Report any serious problems your pregnant patients have had after taking a medicine to the FDA. It falls to physicians to report to the FDA any cases of serious side effects, problems with product quality, and product-use errors or with any of the following products: human drugs, medical devices, blood products and other biologics (except vaccines), and/or medical foods.
- Encourage patients to enroll in a Pregnancy Exposure Registry, if applicable at the FDA website, which collects information on pregnancy outcomes in women who already are taking medications. Observational studies of the patients that physicians help enroll in a pregnancy exposure registry can improve drug safety information for medicines used during pregnancy and can be used to update drug labeling. The observational studies included with the registries can also help physicians make medicine recommendations for use during pregnancy. The list includes contact information for each registry. Physicians can check online to see if there is a registry for their patients’ medicine.
The FDA website also provides information about the Pregnancy and Lactation Labeling Final Rule, which requires changes to the content and format for information presented in prescription drug labeling. The changes are implemented to help health care providers assess risk versus benefit and in subsequent counseling of pregnant women and nursing mothers.
The FDA offers free medicine safety and pregnancy resources for pregnant women, including downloadable infographics; a Medicines Record Keeper brochure in English, Spanish
Pregnancy is an exciting time for women, but they may have questions and concerns about how medicines they take will affect their babies. Our pregnancy website can help make a woman’s pregnancy happier and healthier.
Dr. Yao is the director of the division of pediatrics and maternal health, Office of Drug Evaluation IV, Center for Drug Evaluation and Research at the FDA, in Silver Spring, Md.
There are about 6 million pregnancies in the United States each year, and it’s estimated that 50% of pregnant women take at least one medicine. Physicians play an important role in helping their pregnant patients make informed health choices, especially when it comes to the safe use of medications.
- Emphasize that patients should always talk to their health care provider before taking any medicines, herbs, or vitamins. The website provides tips on how to start the conversation with patients about what medicines, herbs, or vitamins to avoid when pregnant.
- Encourage patients to check the drug label and other information that comes with their medicine to learn about possible risks for women who are pregnant or breastfeeding.
- Assist pregnant patients with changing medications as needed.
- Advise pregnant patients if they need to take more or less of their medicines.
- Advise patients about medicines that can and cannot be used when they start breastfeeding.
- Encourage patients to talk about any problems they have with their medicine.
- Report any serious problems your pregnant patients have had after taking a medicine to the FDA. It falls to physicians to report to the FDA any cases of serious side effects, problems with product quality, and product-use errors or with any of the following products: human drugs, medical devices, blood products and other biologics (except vaccines), and/or medical foods.
- Encourage patients to enroll in a Pregnancy Exposure Registry, if applicable at the FDA website, which collects information on pregnancy outcomes in women who already are taking medications. Observational studies of the patients that physicians help enroll in a pregnancy exposure registry can improve drug safety information for medicines used during pregnancy and can be used to update drug labeling. The observational studies included with the registries can also help physicians make medicine recommendations for use during pregnancy. The list includes contact information for each registry. Physicians can check online to see if there is a registry for their patients’ medicine.
The FDA website also provides information about the Pregnancy and Lactation Labeling Final Rule, which requires changes to the content and format for information presented in prescription drug labeling. The changes are implemented to help health care providers assess risk versus benefit and in subsequent counseling of pregnant women and nursing mothers.
The FDA offers free medicine safety and pregnancy resources for pregnant women, including downloadable infographics; a Medicines Record Keeper brochure in English, Spanish
Pregnancy is an exciting time for women, but they may have questions and concerns about how medicines they take will affect their babies. Our pregnancy website can help make a woman’s pregnancy happier and healthier.
Dr. Yao is the director of the division of pediatrics and maternal health, Office of Drug Evaluation IV, Center for Drug Evaluation and Research at the FDA, in Silver Spring, Md.
Septicemia admissions almost tripled from 2005 to 2014
Admissions for septicemia nearly tripled from 2005 to 2014, as it became the third most common diagnosis for hospital stays, according to the Agency for Healthcare Research and Quality.
There were over 1.5 million hospital stays with a principal diagnosis of septicemia in 2014, an increase of 192% over the 518,000 stays in 2005. The only diagnoses with more admissions in 2014 were pregnancy/childbirth with 4.1 million stays and newborns/neonates at almost 4 million, although both were down from 2005. That year, septicemia did not even rank among the top 10 diagnoses, the AHRQ reported.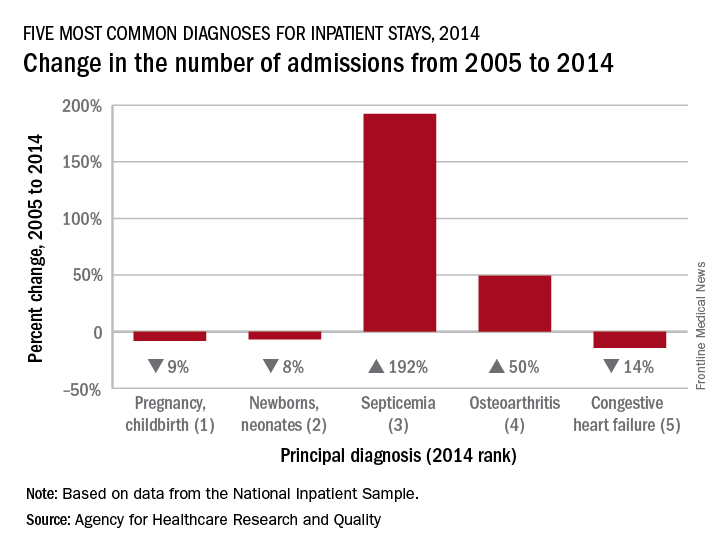
Pneumonia, which was the third most common diagnosis in 2005, dropped by 32% and ended up in sixth place in 2014, while admissions for coronary atherosclerosis, which was fourth in 2005, decreased by 63%, dropping out of the top 10, by 2014, the AHRQ said.
Septicemia was the most common diagnosis for inpatient stays among those aged 75 years and older and the second most common for those aged 65-74 and 45-64. The leading nonmaternal, nonneonatal diagnosis in the two youngest age groups, 0-17 and 18-44 years, was mood disorders, and the most common cause of admissions for those aged 45-64 and 65-74 was osteoarthritis, the AHRQ reported.
Admissions for septicemia nearly tripled from 2005 to 2014, as it became the third most common diagnosis for hospital stays, according to the Agency for Healthcare Research and Quality.
There were over 1.5 million hospital stays with a principal diagnosis of septicemia in 2014, an increase of 192% over the 518,000 stays in 2005. The only diagnoses with more admissions in 2014 were pregnancy/childbirth with 4.1 million stays and newborns/neonates at almost 4 million, although both were down from 2005. That year, septicemia did not even rank among the top 10 diagnoses, the AHRQ reported.
Pneumonia, which was the third most common diagnosis in 2005, dropped by 32% and ended up in sixth place in 2014, while admissions for coronary atherosclerosis, which was fourth in 2005, decreased by 63%, dropping out of the top 10, by 2014, the AHRQ said.
Septicemia was the most common diagnosis for inpatient stays among those aged 75 years and older and the second most common for those aged 65-74 and 45-64. The leading nonmaternal, nonneonatal diagnosis in the two youngest age groups, 0-17 and 18-44 years, was mood disorders, and the most common cause of admissions for those aged 45-64 and 65-74 was osteoarthritis, the AHRQ reported.
Admissions for septicemia nearly tripled from 2005 to 2014, as it became the third most common diagnosis for hospital stays, according to the Agency for Healthcare Research and Quality.
There were over 1.5 million hospital stays with a principal diagnosis of septicemia in 2014, an increase of 192% over the 518,000 stays in 2005. The only diagnoses with more admissions in 2014 were pregnancy/childbirth with 4.1 million stays and newborns/neonates at almost 4 million, although both were down from 2005. That year, septicemia did not even rank among the top 10 diagnoses, the AHRQ reported.
Pneumonia, which was the third most common diagnosis in 2005, dropped by 32% and ended up in sixth place in 2014, while admissions for coronary atherosclerosis, which was fourth in 2005, decreased by 63%, dropping out of the top 10, by 2014, the AHRQ said.
Septicemia was the most common diagnosis for inpatient stays among those aged 75 years and older and the second most common for those aged 65-74 and 45-64. The leading nonmaternal, nonneonatal diagnosis in the two youngest age groups, 0-17 and 18-44 years, was mood disorders, and the most common cause of admissions for those aged 45-64 and 65-74 was osteoarthritis, the AHRQ reported.
Bad news keeps piling up for Absorb coronary scaffold
PARIS – Device thrombosis occurred nearly four times more frequently in recipients of the Absorb everolimus-eluting bioresorbable vascular scaffold than with the Xience everolimus-eluting metallic stent during 2 years of prospective follow-up in the randomized AIDA trial.
AIDA (the Amsterdam Investigator-Initiated Absorb Strategy All-Comers Trial) was the first randomized trial designed to compare the Absorb scaffold to a drug-eluting metallic stent in a broad patient population reflecting routine real-world clinical practice. The disturbing AIDA finding follows upon earlier serious concerns raised regarding an increased risk of scaffold thrombosis – and the particularly worrisome complication of late thrombosis – in the ABSORB Japan and ABSORB II trials, Joanna J. Wykrzykowska, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The device was approved by the Food and Drug Administration in July 2016. In March 2017 the agency issued a safety alert regarding the Absorb scaffold after release of the 2-year data from the 2,008-patient ABSORB III trial showing a significantly higher rate of target-lesion failure than with the Xience stent. Both devices are marketed by Abbott Vascular.
AIDA was a single-blind multicenter Dutch trial that randomized 1,845 patients undergoing PCI, 55% of whom presented with acute coronary syndrome and 26% of whom had ST-elevation MI. The primary endpoint was target vessel failure, a composite of cardiac death, target vessel MI, or target vessel revascularization. The 2-year cumulative rate did not differ significantly between the two study arms: 11.7% in the scaffold group and 10.7% in the metallic stent recipients.
However, definite or probable device thrombosis occurred in 3.5% of the scaffold group compared with 0.9% of metallic stent recipients, for a highly significant 3.9-fold increased risk. This was associated with a significantly increased 2-year cumulative risk of MI: 5.5% versus 3.2%.
On the basis of this unsettling finding, coupled with the fact that ABSORB II investigators did not find any instance of very late scaffold thrombosis among 63 patients who remained on dual-antiplatelet therapy (DAPT) continuously for up to 3 years, Dr. Wykrzykowska and her coinvestigators have informed AIDA participants of their treatment assignment. They have also recommended that the Absorb recipients go on extended DAPT, even though there is no high-grade evidence as yet that this will prevent late scaffold thrombosis or that the drug-induced increased bleeding risk of prolonged DAPT might cancel or perhaps even outweigh the potential protection against device thrombosis.
On top of all this, implantation of the scaffold entails a longer procedure time and a greater volume of contrast material.
Discussant Mahmoud Hashemian, MD, observed that while bioresorbable vascular scaffolds are “physiologically ideal” because, unlike metallic stents, theoretically they leave no permanent implant to impede vasomotion and serve as a nidus for neoatherosclerosis, to date they have shown no real-world benefits over current-generation drug-eluting metallic stents, but only disadvantages.
“This doesn’t mean we have to feel hopeless. I’m not hopeless at all,” said Dr. Hashemian, an interventional cardiologist at Day General Hospital in Tehran. “I’m sure this [bioresorbable scaffolds] will be the future of our stents. But it needs more work. The company tells me they are going to launch a newer one, maybe next year, with thinner struts and more expandability.”
Asked about the likely mechanism of prolonged thrombosis risk with Absorb, Dr. Wykrzykowska was quick to say no one really knows at this point.
“Technique [predilation at a 1:1 balloon-to-artery ratio with an appropriately sized balloon] can obviously improve things in the short term for early events, but I don’t think we understand the biology of late events. We don’t understand the interaction between the device and the vessel. It’s extremely complex,” she said.
AIDA was funded by an unrestricted educational grant from Abbott Vascular. Dr. Wykrzykowska reported receiving consulting and lecture fees from the company.
PARIS – Device thrombosis occurred nearly four times more frequently in recipients of the Absorb everolimus-eluting bioresorbable vascular scaffold than with the Xience everolimus-eluting metallic stent during 2 years of prospective follow-up in the randomized AIDA trial.
AIDA (the Amsterdam Investigator-Initiated Absorb Strategy All-Comers Trial) was the first randomized trial designed to compare the Absorb scaffold to a drug-eluting metallic stent in a broad patient population reflecting routine real-world clinical practice. The disturbing AIDA finding follows upon earlier serious concerns raised regarding an increased risk of scaffold thrombosis – and the particularly worrisome complication of late thrombosis – in the ABSORB Japan and ABSORB II trials, Joanna J. Wykrzykowska, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The device was approved by the Food and Drug Administration in July 2016. In March 2017 the agency issued a safety alert regarding the Absorb scaffold after release of the 2-year data from the 2,008-patient ABSORB III trial showing a significantly higher rate of target-lesion failure than with the Xience stent. Both devices are marketed by Abbott Vascular.
AIDA was a single-blind multicenter Dutch trial that randomized 1,845 patients undergoing PCI, 55% of whom presented with acute coronary syndrome and 26% of whom had ST-elevation MI. The primary endpoint was target vessel failure, a composite of cardiac death, target vessel MI, or target vessel revascularization. The 2-year cumulative rate did not differ significantly between the two study arms: 11.7% in the scaffold group and 10.7% in the metallic stent recipients.
However, definite or probable device thrombosis occurred in 3.5% of the scaffold group compared with 0.9% of metallic stent recipients, for a highly significant 3.9-fold increased risk. This was associated with a significantly increased 2-year cumulative risk of MI: 5.5% versus 3.2%.
On the basis of this unsettling finding, coupled with the fact that ABSORB II investigators did not find any instance of very late scaffold thrombosis among 63 patients who remained on dual-antiplatelet therapy (DAPT) continuously for up to 3 years, Dr. Wykrzykowska and her coinvestigators have informed AIDA participants of their treatment assignment. They have also recommended that the Absorb recipients go on extended DAPT, even though there is no high-grade evidence as yet that this will prevent late scaffold thrombosis or that the drug-induced increased bleeding risk of prolonged DAPT might cancel or perhaps even outweigh the potential protection against device thrombosis.
On top of all this, implantation of the scaffold entails a longer procedure time and a greater volume of contrast material.
Discussant Mahmoud Hashemian, MD, observed that while bioresorbable vascular scaffolds are “physiologically ideal” because, unlike metallic stents, theoretically they leave no permanent implant to impede vasomotion and serve as a nidus for neoatherosclerosis, to date they have shown no real-world benefits over current-generation drug-eluting metallic stents, but only disadvantages.
“This doesn’t mean we have to feel hopeless. I’m not hopeless at all,” said Dr. Hashemian, an interventional cardiologist at Day General Hospital in Tehran. “I’m sure this [bioresorbable scaffolds] will be the future of our stents. But it needs more work. The company tells me they are going to launch a newer one, maybe next year, with thinner struts and more expandability.”
Asked about the likely mechanism of prolonged thrombosis risk with Absorb, Dr. Wykrzykowska was quick to say no one really knows at this point.
“Technique [predilation at a 1:1 balloon-to-artery ratio with an appropriately sized balloon] can obviously improve things in the short term for early events, but I don’t think we understand the biology of late events. We don’t understand the interaction between the device and the vessel. It’s extremely complex,” she said.
AIDA was funded by an unrestricted educational grant from Abbott Vascular. Dr. Wykrzykowska reported receiving consulting and lecture fees from the company.
PARIS – Device thrombosis occurred nearly four times more frequently in recipients of the Absorb everolimus-eluting bioresorbable vascular scaffold than with the Xience everolimus-eluting metallic stent during 2 years of prospective follow-up in the randomized AIDA trial.
AIDA (the Amsterdam Investigator-Initiated Absorb Strategy All-Comers Trial) was the first randomized trial designed to compare the Absorb scaffold to a drug-eluting metallic stent in a broad patient population reflecting routine real-world clinical practice. The disturbing AIDA finding follows upon earlier serious concerns raised regarding an increased risk of scaffold thrombosis – and the particularly worrisome complication of late thrombosis – in the ABSORB Japan and ABSORB II trials, Joanna J. Wykrzykowska, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
The device was approved by the Food and Drug Administration in July 2016. In March 2017 the agency issued a safety alert regarding the Absorb scaffold after release of the 2-year data from the 2,008-patient ABSORB III trial showing a significantly higher rate of target-lesion failure than with the Xience stent. Both devices are marketed by Abbott Vascular.
AIDA was a single-blind multicenter Dutch trial that randomized 1,845 patients undergoing PCI, 55% of whom presented with acute coronary syndrome and 26% of whom had ST-elevation MI. The primary endpoint was target vessel failure, a composite of cardiac death, target vessel MI, or target vessel revascularization. The 2-year cumulative rate did not differ significantly between the two study arms: 11.7% in the scaffold group and 10.7% in the metallic stent recipients.
However, definite or probable device thrombosis occurred in 3.5% of the scaffold group compared with 0.9% of metallic stent recipients, for a highly significant 3.9-fold increased risk. This was associated with a significantly increased 2-year cumulative risk of MI: 5.5% versus 3.2%.
On the basis of this unsettling finding, coupled with the fact that ABSORB II investigators did not find any instance of very late scaffold thrombosis among 63 patients who remained on dual-antiplatelet therapy (DAPT) continuously for up to 3 years, Dr. Wykrzykowska and her coinvestigators have informed AIDA participants of their treatment assignment. They have also recommended that the Absorb recipients go on extended DAPT, even though there is no high-grade evidence as yet that this will prevent late scaffold thrombosis or that the drug-induced increased bleeding risk of prolonged DAPT might cancel or perhaps even outweigh the potential protection against device thrombosis.
On top of all this, implantation of the scaffold entails a longer procedure time and a greater volume of contrast material.
Discussant Mahmoud Hashemian, MD, observed that while bioresorbable vascular scaffolds are “physiologically ideal” because, unlike metallic stents, theoretically they leave no permanent implant to impede vasomotion and serve as a nidus for neoatherosclerosis, to date they have shown no real-world benefits over current-generation drug-eluting metallic stents, but only disadvantages.
“This doesn’t mean we have to feel hopeless. I’m not hopeless at all,” said Dr. Hashemian, an interventional cardiologist at Day General Hospital in Tehran. “I’m sure this [bioresorbable scaffolds] will be the future of our stents. But it needs more work. The company tells me they are going to launch a newer one, maybe next year, with thinner struts and more expandability.”
Asked about the likely mechanism of prolonged thrombosis risk with Absorb, Dr. Wykrzykowska was quick to say no one really knows at this point.
“Technique [predilation at a 1:1 balloon-to-artery ratio with an appropriately sized balloon] can obviously improve things in the short term for early events, but I don’t think we understand the biology of late events. We don’t understand the interaction between the device and the vessel. It’s extremely complex,” she said.
AIDA was funded by an unrestricted educational grant from Abbott Vascular. Dr. Wykrzykowska reported receiving consulting and lecture fees from the company.
AT EUROPCR
Key clinical point:
Major finding: During 2 years of prospective follow-up, definite or probable device thrombosis occurred in 3.5% of recipients of a bioresorbable vascular scaffold, compared with 0.9% of metallic stent recipients, for a highly significant 3.9-fold increased risk.
Data source: AIDA, a single-blind multicenter Dutch trial that randomized a broadly representative group of 1,845 patients undergoing PCI to the Absorb bioresorbable vascular scaffold or the Xience everolimus-eluting metallic stent.
Disclosures: The AIDA study was funded by an unrestricted educational grant from Abbott Vascular. The presenter reported receiving consulting and lecture fees from the company.
Reframe view of borderline personality disorder patients as survivors
WASHINGTON – A view of borderline personality disorder as neurobiological in nature can help clinicians extend their patience and empathy to these notoriously difficult-to-treat patients, according to an expert.
“Unfortunately, there are those who ... are just too frustrated by” [patients with borderline personality disorder], said Carmen V. Pinto, MD, an Elizabethtown, Ky.–based psychiatrist who specializes in treating patients with the disorder.
The diagnosis occurs in up to 2% of the population and is twice as common in women. About three-quarters of borderline personality disorder patients will attempt suicide at least once, and up to 1 in 10 complete suicide, said Dr. Pinto, assistant clinical professor of psychiatry at the University of Louisville (Ky.).
“There is an art to dealing with patients who are impulsive, and sometimes dangerous and scary,” Dr. Pinto said at Summit in Neurology & Psychiatry, held by Global Academy for Medical Education.
He emphasized the effect of unstable interpersonal relationships combined with frontolimbic brain abnormalities on the lives of patients with borderline personality disorder. In some cases, borderline personality disorder patients experienced traumatic events such as sexual abuse that was personal and ongoing – and occurred while the personality was being formed.
“These are normal people who have been exposed to abnormal stress,” Dr. Pinto said. “So you have to be careful, and see them not as victims but as survivors still suffering.”
Citing several lines of study into the neurobiochemical mechanisms of action in the disorder, Dr. Pinto discussed the role of disruption to the brain’s cortical-limbic circuit that serves as the brain’s basic stress response. In this population, an overactive amygdala and hypofunctioning frontal cortex continually signal threats even when none exist – exciting the sympathetic nervous system – as well as the endocrine and immune systems.
“The circuit gets overwhelmed, either because there really is too much to deal with, or there was poor functioning to start with, so the brain is going off like a pinball machine,” Dr. Pinto said. “If [the patients perceive] even the slightest offense, their limbic system lights up, and they have trouble turning it off, which is why it can be so difficult to deal with these folks in therapy.”
He explained the borderline personality patient’s typical negative valence as the result of this hyper-threat detection, often rooted in experiences in which people who professed to care for them acted contrary to their words, as in cases of incest. “They learn they can’t trust what is said, so they rely on nonverbal cues,” Dr. Pinto said. “They aren’t listening to what is said to them.”
Dr. Pinto cited a study of how this patient population had trouble distinguishing neutral faces from faces expressing anger or boredom, resulting in their believing that most people they encounter are unhappy with them in some way, causing them to retreat or have stressful relationships with others.
“Therapeutic neutrality is not the best way to approach this person, because they read it as ‘you are mad at me’ or ‘you’re bored with me’ or ‘you don’t like me,’ ” Dr. Pinto said, noting that he likes to reassure patients that they are doing the best they can with what they have. However, he warned against how often, in these patients’ quest to feel loved and accepted, they might ask for hugs or other contact. Rather than panic, use the opportunity to help the patient practice having healthy boundaries by explaining that not hugging them is designed to protect them, Dr. Pinto said.
Among the DSM-5 criteria for diagnosing this disorder is that the patient has an “enduring pattern of inner experience and behavior that deviates markedly from the expectations of the individual’s culture.” Because there are no imaging or laboratory tests to confirm a deviant inner experience, the clinician must turn to the patient for assessment while quelling the impulse to assign meanings to action and insight.
“You really have to be quiet and listen. They are the expert with their inner experience, and you have to let them tell you what that is,” he said, noting that the distortion of how they view themselves in relation to others is painful and pervasive, crossing all areas of their lives.
Because relationships are so difficult for these patients to develop, Dr. Pinto urged keeping in mind that it is possible they have no other therapeutic relationship in their lives. “That is a scary responsibility, to think they have no one else to talk to but you,” he said. This underscores the need for clinicians to protect themselves, too. “If I don’t feel safe, I can’t do a good job for them – and I tell them that,” Dr. Pinto said.
Global Academy and this news organization are owned by the same company. Dr. Pinto had no relevant disclosures, although he said he is a paid speaker for Otsuka, Lundbeck, Janssen, and other pharmaceutical companies.
[email protected]
On Twitter @whitneymcknight
WASHINGTON – A view of borderline personality disorder as neurobiological in nature can help clinicians extend their patience and empathy to these notoriously difficult-to-treat patients, according to an expert.
“Unfortunately, there are those who ... are just too frustrated by” [patients with borderline personality disorder], said Carmen V. Pinto, MD, an Elizabethtown, Ky.–based psychiatrist who specializes in treating patients with the disorder.
The diagnosis occurs in up to 2% of the population and is twice as common in women. About three-quarters of borderline personality disorder patients will attempt suicide at least once, and up to 1 in 10 complete suicide, said Dr. Pinto, assistant clinical professor of psychiatry at the University of Louisville (Ky.).
“There is an art to dealing with patients who are impulsive, and sometimes dangerous and scary,” Dr. Pinto said at Summit in Neurology & Psychiatry, held by Global Academy for Medical Education.
He emphasized the effect of unstable interpersonal relationships combined with frontolimbic brain abnormalities on the lives of patients with borderline personality disorder. In some cases, borderline personality disorder patients experienced traumatic events such as sexual abuse that was personal and ongoing – and occurred while the personality was being formed.
“These are normal people who have been exposed to abnormal stress,” Dr. Pinto said. “So you have to be careful, and see them not as victims but as survivors still suffering.”
Citing several lines of study into the neurobiochemical mechanisms of action in the disorder, Dr. Pinto discussed the role of disruption to the brain’s cortical-limbic circuit that serves as the brain’s basic stress response. In this population, an overactive amygdala and hypofunctioning frontal cortex continually signal threats even when none exist – exciting the sympathetic nervous system – as well as the endocrine and immune systems.
“The circuit gets overwhelmed, either because there really is too much to deal with, or there was poor functioning to start with, so the brain is going off like a pinball machine,” Dr. Pinto said. “If [the patients perceive] even the slightest offense, their limbic system lights up, and they have trouble turning it off, which is why it can be so difficult to deal with these folks in therapy.”
He explained the borderline personality patient’s typical negative valence as the result of this hyper-threat detection, often rooted in experiences in which people who professed to care for them acted contrary to their words, as in cases of incest. “They learn they can’t trust what is said, so they rely on nonverbal cues,” Dr. Pinto said. “They aren’t listening to what is said to them.”
Dr. Pinto cited a study of how this patient population had trouble distinguishing neutral faces from faces expressing anger or boredom, resulting in their believing that most people they encounter are unhappy with them in some way, causing them to retreat or have stressful relationships with others.
“Therapeutic neutrality is not the best way to approach this person, because they read it as ‘you are mad at me’ or ‘you’re bored with me’ or ‘you don’t like me,’ ” Dr. Pinto said, noting that he likes to reassure patients that they are doing the best they can with what they have. However, he warned against how often, in these patients’ quest to feel loved and accepted, they might ask for hugs or other contact. Rather than panic, use the opportunity to help the patient practice having healthy boundaries by explaining that not hugging them is designed to protect them, Dr. Pinto said.
Among the DSM-5 criteria for diagnosing this disorder is that the patient has an “enduring pattern of inner experience and behavior that deviates markedly from the expectations of the individual’s culture.” Because there are no imaging or laboratory tests to confirm a deviant inner experience, the clinician must turn to the patient for assessment while quelling the impulse to assign meanings to action and insight.
“You really have to be quiet and listen. They are the expert with their inner experience, and you have to let them tell you what that is,” he said, noting that the distortion of how they view themselves in relation to others is painful and pervasive, crossing all areas of their lives.
Because relationships are so difficult for these patients to develop, Dr. Pinto urged keeping in mind that it is possible they have no other therapeutic relationship in their lives. “That is a scary responsibility, to think they have no one else to talk to but you,” he said. This underscores the need for clinicians to protect themselves, too. “If I don’t feel safe, I can’t do a good job for them – and I tell them that,” Dr. Pinto said.
Global Academy and this news organization are owned by the same company. Dr. Pinto had no relevant disclosures, although he said he is a paid speaker for Otsuka, Lundbeck, Janssen, and other pharmaceutical companies.
[email protected]
On Twitter @whitneymcknight
WASHINGTON – A view of borderline personality disorder as neurobiological in nature can help clinicians extend their patience and empathy to these notoriously difficult-to-treat patients, according to an expert.
“Unfortunately, there are those who ... are just too frustrated by” [patients with borderline personality disorder], said Carmen V. Pinto, MD, an Elizabethtown, Ky.–based psychiatrist who specializes in treating patients with the disorder.
The diagnosis occurs in up to 2% of the population and is twice as common in women. About three-quarters of borderline personality disorder patients will attempt suicide at least once, and up to 1 in 10 complete suicide, said Dr. Pinto, assistant clinical professor of psychiatry at the University of Louisville (Ky.).
“There is an art to dealing with patients who are impulsive, and sometimes dangerous and scary,” Dr. Pinto said at Summit in Neurology & Psychiatry, held by Global Academy for Medical Education.
He emphasized the effect of unstable interpersonal relationships combined with frontolimbic brain abnormalities on the lives of patients with borderline personality disorder. In some cases, borderline personality disorder patients experienced traumatic events such as sexual abuse that was personal and ongoing – and occurred while the personality was being formed.
“These are normal people who have been exposed to abnormal stress,” Dr. Pinto said. “So you have to be careful, and see them not as victims but as survivors still suffering.”
Citing several lines of study into the neurobiochemical mechanisms of action in the disorder, Dr. Pinto discussed the role of disruption to the brain’s cortical-limbic circuit that serves as the brain’s basic stress response. In this population, an overactive amygdala and hypofunctioning frontal cortex continually signal threats even when none exist – exciting the sympathetic nervous system – as well as the endocrine and immune systems.
“The circuit gets overwhelmed, either because there really is too much to deal with, or there was poor functioning to start with, so the brain is going off like a pinball machine,” Dr. Pinto said. “If [the patients perceive] even the slightest offense, their limbic system lights up, and they have trouble turning it off, which is why it can be so difficult to deal with these folks in therapy.”
He explained the borderline personality patient’s typical negative valence as the result of this hyper-threat detection, often rooted in experiences in which people who professed to care for them acted contrary to their words, as in cases of incest. “They learn they can’t trust what is said, so they rely on nonverbal cues,” Dr. Pinto said. “They aren’t listening to what is said to them.”
Dr. Pinto cited a study of how this patient population had trouble distinguishing neutral faces from faces expressing anger or boredom, resulting in their believing that most people they encounter are unhappy with them in some way, causing them to retreat or have stressful relationships with others.
“Therapeutic neutrality is not the best way to approach this person, because they read it as ‘you are mad at me’ or ‘you’re bored with me’ or ‘you don’t like me,’ ” Dr. Pinto said, noting that he likes to reassure patients that they are doing the best they can with what they have. However, he warned against how often, in these patients’ quest to feel loved and accepted, they might ask for hugs or other contact. Rather than panic, use the opportunity to help the patient practice having healthy boundaries by explaining that not hugging them is designed to protect them, Dr. Pinto said.
Among the DSM-5 criteria for diagnosing this disorder is that the patient has an “enduring pattern of inner experience and behavior that deviates markedly from the expectations of the individual’s culture.” Because there are no imaging or laboratory tests to confirm a deviant inner experience, the clinician must turn to the patient for assessment while quelling the impulse to assign meanings to action and insight.
“You really have to be quiet and listen. They are the expert with their inner experience, and you have to let them tell you what that is,” he said, noting that the distortion of how they view themselves in relation to others is painful and pervasive, crossing all areas of their lives.
Because relationships are so difficult for these patients to develop, Dr. Pinto urged keeping in mind that it is possible they have no other therapeutic relationship in their lives. “That is a scary responsibility, to think they have no one else to talk to but you,” he said. This underscores the need for clinicians to protect themselves, too. “If I don’t feel safe, I can’t do a good job for them – and I tell them that,” Dr. Pinto said.
Global Academy and this news organization are owned by the same company. Dr. Pinto had no relevant disclosures, although he said he is a paid speaker for Otsuka, Lundbeck, Janssen, and other pharmaceutical companies.
[email protected]
On Twitter @whitneymcknight
EXPERT ANALYSIS FROM SUMMIT IN NEUROLOGY & PSYCHIATRY
FDA approves new panel to identify mCRC patients for panitumumab treatment
The Food and Drug Administration has approved the Praxis Extended RAS Panel for the identification of metastatic colorectal cancer patients who can be treated with panitumumab.
The Praxis Extended RAS Panel is able to detect 56 specific mutations in the RAS genes of mCRC patients, and is the first next generation sequencing test approved by the FDA capable of testing more than one RAS gene mutation. If RAS mutations are not detected, then panitumumab is indicated, and if a mutation is detected, panitumumab is not indicated, according to the FDA statement.
“Panitumumab’s product labeling has been modified to align the indication for panitumumab and intended use for the Praxis Extended RAS Panel,” the FDA noted.
The Food and Drug Administration has approved the Praxis Extended RAS Panel for the identification of metastatic colorectal cancer patients who can be treated with panitumumab.
The Praxis Extended RAS Panel is able to detect 56 specific mutations in the RAS genes of mCRC patients, and is the first next generation sequencing test approved by the FDA capable of testing more than one RAS gene mutation. If RAS mutations are not detected, then panitumumab is indicated, and if a mutation is detected, panitumumab is not indicated, according to the FDA statement.
“Panitumumab’s product labeling has been modified to align the indication for panitumumab and intended use for the Praxis Extended RAS Panel,” the FDA noted.
The Food and Drug Administration has approved the Praxis Extended RAS Panel for the identification of metastatic colorectal cancer patients who can be treated with panitumumab.
The Praxis Extended RAS Panel is able to detect 56 specific mutations in the RAS genes of mCRC patients, and is the first next generation sequencing test approved by the FDA capable of testing more than one RAS gene mutation. If RAS mutations are not detected, then panitumumab is indicated, and if a mutation is detected, panitumumab is not indicated, according to the FDA statement.
“Panitumumab’s product labeling has been modified to align the indication for panitumumab and intended use for the Praxis Extended RAS Panel,” the FDA noted.
New on the streets: Drug for nerve pain boosts high for opioid abusers
ATHENS, OHIO – On April 5, Ciera Smith sat in a car parked on the gravel driveway of the Rural Women’s Recovery Program here with a choice to make: Go to jail or enter treatment for her addiction.
Ms. Smith, 22, started abusing drugs when she was 18 years old, enticed by the “good time” she and her friends found in smoking marijuana. She later turned to addictive painkillers, then antianxiety medications such as Xanax and eventually Suboxone, a narcotic often used to replace opioids when treating addiction.
Before stepping out of the car, she decided she needed one more high before treatment. She reached into her purse and then swallowed a handful of gabapentin pills.
Last December, Ohio’s Board of Pharmacy began reporting sales of gabapentin prescriptions in its regular monitoring of controlled substances. The drug, which is neither an opioid nor designated a controlled substance by federal authorities, is used to treat nerve pain. But the board found that it was the most prescribed medication on its list that month, surpassing oxycodone by more than 9 million doses. In February, the Ohio Substance Abuse Monitoring Network issued an alert regarding increasing misuse across the state.
And it’s not just in Ohio. Gabapentin’s ability to tackle multiple ailments has helped make it one of the most popular medications in the United States. In May, it was the fifth-most prescribed drug in the nation, according to GoodRx.
Gabapentin is approved by the Food and Drug Administration to treat epilepsy and pain related to nerve damage, called neuropathy. Also known by its brand name, Neurontin, the drug acts as a sedative. It is widely considered nonaddictive and touted by the federal Centers for Disease Control and Prevention as an alternative intervention to opiates for chronic pain. Generally, doctors prescribe no more than 1,800-2,400 mg of gabapentin per day, according to information on the Mayo Clinic’s website.
Gabapentin does not carry the same risk of lethal overdoses as opioids, but drug experts say the effects of using gabapentin for long periods of time or in very high quantities, particularly among such sensitive populations as pregnant women, are not well known.
As providers dole out the drug in mass quantities for conditions such as restless legs syndrome and alcoholism, it is being subverted to a drug of abuse. Gabapentin can enhance the euphoria caused by an opioid and stave off drug withdrawals. In addition, it can bypass the blocking effects of medications used for addiction treatment, enabling patients to get high while in recovery.
Athens, home to Ohio University, lies in the southeastern corner of the state, which has been ravaged by the opioid epidemic. Despite experience in combating illicit drug use, law enforcement officials and drug counselors say the addition of gabapentin adds a new obstacle.
“I don’t know if we have a clear picture of the risk,” said Joe Gay, executive director of Health Recovery Services, a network of substance abuse recovery centers headquartered in Athens.
‘Available to be abused’
A literature review published in 2016 in the journal Addiction found about a fifth of those who abuse opiates misuse gabapentin. A separate 2015 study of adults in Appalachian Kentucky who abused opiates found 15% of participants also misused gabapentin in the past 6 months “to get high.”
In the same year, the drug was involved in 109 overdose deaths in West Virginia, the Charleston Gazette-Mail reported.
Rachel Quivey, an Athens pharmacist, said she noticed signs of gabapentin misuse half a decade ago when patients began picking up the drug several days before their prescription ran out.
“Gabapentin is so readily available,” she said. “That, in my opinion, is where a lot of that danger is. It’s available to be abused.”
In May, Ms. Quivey’s pharmacy filled roughly 33 prescriptions of gabapentin per week, dispensing 90-120 pills for each client.
For customers who arrive with scripts demanding a high dosage of the drug, Ms. Quivey sometimes calls the doctor to discuss her concerns. But many of them aren’t aware of gabapentin misuse, she said.
Even as gabapentin gets restocked regularly on Ms. Quivey’s shelves, the drug’s presence is increasing on the streets of Athens. A 300-milligram pill sells for as little as 75 cents.
Yet, according to Chuck Haegele, field supervisor for the major crimes unit at the Athens City Police Department, law enforcement can do little to stop its spread. That’s because gabapentin is not categorized as a controlled substance. That designation places restrictions on who can possess and dispense the drug.
“There’s really not much we can do at this point,” he said. “If it’s not controlled … it’s not illegal for somebody that’s not prescribed it to possess it.”
Mr. Haegele said he heard about the drug less than 3 months ago when an officer accidentally received a text message from someone offering to sell it. The police force, he said, is still trying to assess the threat of gabapentin.
Little testing
Nearly anyone arrested and found to struggle with addiction in Athens is given the option to go through a drug-court program to get treatment. But officials said that some exploit the absence of routine exams for gabapentin to get high while testing clean.
Brice Johnson, a probation officer at Athens County Municipal Court, said participants in the municipal court’s substance abuse mentally ill program undergo gabapentin testing only when abuse is suspected. Screenings are not regularly done on every client because gabapentin abuse has not been a concern and the testing adds expense, he said.
The rehab program run through the county prosecutor’s office, called Fresh Start, does test for gabapentin. Its latest round of screenings detected the drug in 5 of its roughly 238 active participants, prosecutor Keller Blackburn said.
Linda Holley, a clinical supervisor at an Athens outpatient program run by the Health Recovery Services, said she suspects at least half of her clients on Suboxone treatment abuse gabapentin. But the center can’t afford to regularly test every participant.
Ms. Holley said she sees clients who are prescribed gabapentin but, because of health privacy laws, she can’t share their status as a person in recovery to an outside provider without written consent. The restrictions give clients in recovery an opportunity to get high using drugs they obtained legally and still pass a drug test.
“With the gabapentin, I wish there were more we could do, but our hands are tied,” she said. “We can’t do anything but educate the client and discourage” them from using such medications.
Ms. Smith visited two separate doctors to secure a prescription. As she rotated through drug court, Narcotics Anonymous meetings, jail for relapsing on cocaine, and house arrest enforced with an ankle bracelet, she said her gabapentin abuse wasn’t detected until she arrived at the residential recovery center.
Today, Ms. Smith sticks to the recovery process. Expecting a baby in early July, her successful completion of the program not only means sobriety but also allows her the opportunity to restore custody of her eldest daughter and raise her children.
She intends to relocate her family away from the friends and routines that helped lead her to addiction and said she will help guide her daughter away from making similar mistakes.
“All I can do is be there and give her the knowledge that I can about addiction,” Ms. Smith said, “and hope that she chooses to go on the right path.”
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
ATHENS, OHIO – On April 5, Ciera Smith sat in a car parked on the gravel driveway of the Rural Women’s Recovery Program here with a choice to make: Go to jail or enter treatment for her addiction.
Ms. Smith, 22, started abusing drugs when she was 18 years old, enticed by the “good time” she and her friends found in smoking marijuana. She later turned to addictive painkillers, then antianxiety medications such as Xanax and eventually Suboxone, a narcotic often used to replace opioids when treating addiction.
Before stepping out of the car, she decided she needed one more high before treatment. She reached into her purse and then swallowed a handful of gabapentin pills.
Last December, Ohio’s Board of Pharmacy began reporting sales of gabapentin prescriptions in its regular monitoring of controlled substances. The drug, which is neither an opioid nor designated a controlled substance by federal authorities, is used to treat nerve pain. But the board found that it was the most prescribed medication on its list that month, surpassing oxycodone by more than 9 million doses. In February, the Ohio Substance Abuse Monitoring Network issued an alert regarding increasing misuse across the state.
And it’s not just in Ohio. Gabapentin’s ability to tackle multiple ailments has helped make it one of the most popular medications in the United States. In May, it was the fifth-most prescribed drug in the nation, according to GoodRx.
Gabapentin is approved by the Food and Drug Administration to treat epilepsy and pain related to nerve damage, called neuropathy. Also known by its brand name, Neurontin, the drug acts as a sedative. It is widely considered nonaddictive and touted by the federal Centers for Disease Control and Prevention as an alternative intervention to opiates for chronic pain. Generally, doctors prescribe no more than 1,800-2,400 mg of gabapentin per day, according to information on the Mayo Clinic’s website.
Gabapentin does not carry the same risk of lethal overdoses as opioids, but drug experts say the effects of using gabapentin for long periods of time or in very high quantities, particularly among such sensitive populations as pregnant women, are not well known.
As providers dole out the drug in mass quantities for conditions such as restless legs syndrome and alcoholism, it is being subverted to a drug of abuse. Gabapentin can enhance the euphoria caused by an opioid and stave off drug withdrawals. In addition, it can bypass the blocking effects of medications used for addiction treatment, enabling patients to get high while in recovery.
Athens, home to Ohio University, lies in the southeastern corner of the state, which has been ravaged by the opioid epidemic. Despite experience in combating illicit drug use, law enforcement officials and drug counselors say the addition of gabapentin adds a new obstacle.
“I don’t know if we have a clear picture of the risk,” said Joe Gay, executive director of Health Recovery Services, a network of substance abuse recovery centers headquartered in Athens.
‘Available to be abused’
A literature review published in 2016 in the journal Addiction found about a fifth of those who abuse opiates misuse gabapentin. A separate 2015 study of adults in Appalachian Kentucky who abused opiates found 15% of participants also misused gabapentin in the past 6 months “to get high.”
In the same year, the drug was involved in 109 overdose deaths in West Virginia, the Charleston Gazette-Mail reported.
Rachel Quivey, an Athens pharmacist, said she noticed signs of gabapentin misuse half a decade ago when patients began picking up the drug several days before their prescription ran out.
“Gabapentin is so readily available,” she said. “That, in my opinion, is where a lot of that danger is. It’s available to be abused.”
In May, Ms. Quivey’s pharmacy filled roughly 33 prescriptions of gabapentin per week, dispensing 90-120 pills for each client.
For customers who arrive with scripts demanding a high dosage of the drug, Ms. Quivey sometimes calls the doctor to discuss her concerns. But many of them aren’t aware of gabapentin misuse, she said.
Even as gabapentin gets restocked regularly on Ms. Quivey’s shelves, the drug’s presence is increasing on the streets of Athens. A 300-milligram pill sells for as little as 75 cents.
Yet, according to Chuck Haegele, field supervisor for the major crimes unit at the Athens City Police Department, law enforcement can do little to stop its spread. That’s because gabapentin is not categorized as a controlled substance. That designation places restrictions on who can possess and dispense the drug.
“There’s really not much we can do at this point,” he said. “If it’s not controlled … it’s not illegal for somebody that’s not prescribed it to possess it.”
Mr. Haegele said he heard about the drug less than 3 months ago when an officer accidentally received a text message from someone offering to sell it. The police force, he said, is still trying to assess the threat of gabapentin.
Little testing
Nearly anyone arrested and found to struggle with addiction in Athens is given the option to go through a drug-court program to get treatment. But officials said that some exploit the absence of routine exams for gabapentin to get high while testing clean.
Brice Johnson, a probation officer at Athens County Municipal Court, said participants in the municipal court’s substance abuse mentally ill program undergo gabapentin testing only when abuse is suspected. Screenings are not regularly done on every client because gabapentin abuse has not been a concern and the testing adds expense, he said.
The rehab program run through the county prosecutor’s office, called Fresh Start, does test for gabapentin. Its latest round of screenings detected the drug in 5 of its roughly 238 active participants, prosecutor Keller Blackburn said.
Linda Holley, a clinical supervisor at an Athens outpatient program run by the Health Recovery Services, said she suspects at least half of her clients on Suboxone treatment abuse gabapentin. But the center can’t afford to regularly test every participant.
Ms. Holley said she sees clients who are prescribed gabapentin but, because of health privacy laws, she can’t share their status as a person in recovery to an outside provider without written consent. The restrictions give clients in recovery an opportunity to get high using drugs they obtained legally and still pass a drug test.
“With the gabapentin, I wish there were more we could do, but our hands are tied,” she said. “We can’t do anything but educate the client and discourage” them from using such medications.
Ms. Smith visited two separate doctors to secure a prescription. As she rotated through drug court, Narcotics Anonymous meetings, jail for relapsing on cocaine, and house arrest enforced with an ankle bracelet, she said her gabapentin abuse wasn’t detected until she arrived at the residential recovery center.
Today, Ms. Smith sticks to the recovery process. Expecting a baby in early July, her successful completion of the program not only means sobriety but also allows her the opportunity to restore custody of her eldest daughter and raise her children.
She intends to relocate her family away from the friends and routines that helped lead her to addiction and said she will help guide her daughter away from making similar mistakes.
“All I can do is be there and give her the knowledge that I can about addiction,” Ms. Smith said, “and hope that she chooses to go on the right path.”
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
ATHENS, OHIO – On April 5, Ciera Smith sat in a car parked on the gravel driveway of the Rural Women’s Recovery Program here with a choice to make: Go to jail or enter treatment for her addiction.
Ms. Smith, 22, started abusing drugs when she was 18 years old, enticed by the “good time” she and her friends found in smoking marijuana. She later turned to addictive painkillers, then antianxiety medications such as Xanax and eventually Suboxone, a narcotic often used to replace opioids when treating addiction.
Before stepping out of the car, she decided she needed one more high before treatment. She reached into her purse and then swallowed a handful of gabapentin pills.
Last December, Ohio’s Board of Pharmacy began reporting sales of gabapentin prescriptions in its regular monitoring of controlled substances. The drug, which is neither an opioid nor designated a controlled substance by federal authorities, is used to treat nerve pain. But the board found that it was the most prescribed medication on its list that month, surpassing oxycodone by more than 9 million doses. In February, the Ohio Substance Abuse Monitoring Network issued an alert regarding increasing misuse across the state.
And it’s not just in Ohio. Gabapentin’s ability to tackle multiple ailments has helped make it one of the most popular medications in the United States. In May, it was the fifth-most prescribed drug in the nation, according to GoodRx.
Gabapentin is approved by the Food and Drug Administration to treat epilepsy and pain related to nerve damage, called neuropathy. Also known by its brand name, Neurontin, the drug acts as a sedative. It is widely considered nonaddictive and touted by the federal Centers for Disease Control and Prevention as an alternative intervention to opiates for chronic pain. Generally, doctors prescribe no more than 1,800-2,400 mg of gabapentin per day, according to information on the Mayo Clinic’s website.
Gabapentin does not carry the same risk of lethal overdoses as opioids, but drug experts say the effects of using gabapentin for long periods of time or in very high quantities, particularly among such sensitive populations as pregnant women, are not well known.
As providers dole out the drug in mass quantities for conditions such as restless legs syndrome and alcoholism, it is being subverted to a drug of abuse. Gabapentin can enhance the euphoria caused by an opioid and stave off drug withdrawals. In addition, it can bypass the blocking effects of medications used for addiction treatment, enabling patients to get high while in recovery.
Athens, home to Ohio University, lies in the southeastern corner of the state, which has been ravaged by the opioid epidemic. Despite experience in combating illicit drug use, law enforcement officials and drug counselors say the addition of gabapentin adds a new obstacle.
“I don’t know if we have a clear picture of the risk,” said Joe Gay, executive director of Health Recovery Services, a network of substance abuse recovery centers headquartered in Athens.
‘Available to be abused’
A literature review published in 2016 in the journal Addiction found about a fifth of those who abuse opiates misuse gabapentin. A separate 2015 study of adults in Appalachian Kentucky who abused opiates found 15% of participants also misused gabapentin in the past 6 months “to get high.”
In the same year, the drug was involved in 109 overdose deaths in West Virginia, the Charleston Gazette-Mail reported.
Rachel Quivey, an Athens pharmacist, said she noticed signs of gabapentin misuse half a decade ago when patients began picking up the drug several days before their prescription ran out.
“Gabapentin is so readily available,” she said. “That, in my opinion, is where a lot of that danger is. It’s available to be abused.”
In May, Ms. Quivey’s pharmacy filled roughly 33 prescriptions of gabapentin per week, dispensing 90-120 pills for each client.
For customers who arrive with scripts demanding a high dosage of the drug, Ms. Quivey sometimes calls the doctor to discuss her concerns. But many of them aren’t aware of gabapentin misuse, she said.
Even as gabapentin gets restocked regularly on Ms. Quivey’s shelves, the drug’s presence is increasing on the streets of Athens. A 300-milligram pill sells for as little as 75 cents.
Yet, according to Chuck Haegele, field supervisor for the major crimes unit at the Athens City Police Department, law enforcement can do little to stop its spread. That’s because gabapentin is not categorized as a controlled substance. That designation places restrictions on who can possess and dispense the drug.
“There’s really not much we can do at this point,” he said. “If it’s not controlled … it’s not illegal for somebody that’s not prescribed it to possess it.”
Mr. Haegele said he heard about the drug less than 3 months ago when an officer accidentally received a text message from someone offering to sell it. The police force, he said, is still trying to assess the threat of gabapentin.
Little testing
Nearly anyone arrested and found to struggle with addiction in Athens is given the option to go through a drug-court program to get treatment. But officials said that some exploit the absence of routine exams for gabapentin to get high while testing clean.
Brice Johnson, a probation officer at Athens County Municipal Court, said participants in the municipal court’s substance abuse mentally ill program undergo gabapentin testing only when abuse is suspected. Screenings are not regularly done on every client because gabapentin abuse has not been a concern and the testing adds expense, he said.
The rehab program run through the county prosecutor’s office, called Fresh Start, does test for gabapentin. Its latest round of screenings detected the drug in 5 of its roughly 238 active participants, prosecutor Keller Blackburn said.
Linda Holley, a clinical supervisor at an Athens outpatient program run by the Health Recovery Services, said she suspects at least half of her clients on Suboxone treatment abuse gabapentin. But the center can’t afford to regularly test every participant.
Ms. Holley said she sees clients who are prescribed gabapentin but, because of health privacy laws, she can’t share their status as a person in recovery to an outside provider without written consent. The restrictions give clients in recovery an opportunity to get high using drugs they obtained legally and still pass a drug test.
“With the gabapentin, I wish there were more we could do, but our hands are tied,” she said. “We can’t do anything but educate the client and discourage” them from using such medications.
Ms. Smith visited two separate doctors to secure a prescription. As she rotated through drug court, Narcotics Anonymous meetings, jail for relapsing on cocaine, and house arrest enforced with an ankle bracelet, she said her gabapentin abuse wasn’t detected until she arrived at the residential recovery center.
Today, Ms. Smith sticks to the recovery process. Expecting a baby in early July, her successful completion of the program not only means sobriety but also allows her the opportunity to restore custody of her eldest daughter and raise her children.
She intends to relocate her family away from the friends and routines that helped lead her to addiction and said she will help guide her daughter away from making similar mistakes.
“All I can do is be there and give her the knowledge that I can about addiction,” Ms. Smith said, “and hope that she chooses to go on the right path.”
Kaiser Health News is a national health policy news service that is part of the nonpartisan Henry J. Kaiser Family Foundation.
First-episode psychosis is a ‘brain attack,’ and LAIs can prevent recurrence, expert says
washington – As data mount confirming the neurodegenerative effects of psychotic episodes in schizophrenia, one expert urges psychiatrists to think of psychosis as a “brain attack” which, like heart attacks, must be prevented from recurring.
“Schizophrenia doesn’t have to be progressive neurodegenerative unless patients relapse again and again, but that happens all the time because we give our patients pills they don’t take as prescribed. There are many reasons for poor adherence,” Henry A. Nasrallah, MD, said at the meeting held by Global Academy for Medical Education.
In a presentation dedicated to the emerging science reshaping views on how schizophrenia occurs, how it can be prevented, and why it is a syndrome with genetic etiologies, Dr. Nasrallah emphasized that there now exist enough data to show that timely intervention with LAIs reliably prevent relapse in most patients, thereby averting progressive neurodegeneration and subsequent disability in people who develop schizophrenia.
“Researchers now stage schizophrenia. Just like cancer, the more advanced the stage, the worse the outcome,” said Dr. Nasrallah, the Sydney W. Souers Endowed Chair and professor of psychiatry and behavioral neuroscience at Saint Louis University, told his audience. “The additional damaging effects of the second episode is what leads to clinical deterioration and can start the process of treatment resistance. But if no psychotic episodes are allowed to recur after the first episode, many patients can return to their baseline functioning, such as school or work.”
The field still is clarifying the neurodevelopmental aspects of schizophrenia, including genetic and in utero adverse events that disrupt brain development, as well as the appropriate types and timing of intervention in the prodromal phase. However, Dr. Nasrallah explained, science already has demonstrated how the neurotoxic effects of psychosis in the brain of a person with schizophrenia lead to brain tissue degradation with every psychotic episode. The result is a progressive decline in social and vocational functioning.
Psychosis is associated with activation of microglia, which are monocytic cells that cross the blood-brain barrier during fetal life, settling in the brain and ultimately comprising 10%-15% of all brain cells. Once activated, they trigger an immune response, leading to neuroinflammation and oxidative stress (free radicals). However, Dr. Nasrallah said, rather than protect the brain, these processes destroy gray and white matter – particularly in the cortical region – degrading the brain and leaving it more compromised, especially if another episode of psychosis occurs.
Another factor in the vulnerability of the brain in schizophrenia is mitochondrial dysfunction. As mitochondria are the primary source of antioxidants – such as glutathione – the deficit in antioxidants increases oxidative stress, furthering the brain’s vulnerability to tissue loss.
Among other biological processes thought to be implicated in neurodegeneration with schizophrenia, Dr. Nasrallah said, are impairment of antiapoptotic signaling, glutamate excitotoxicity, hypercortisolemia, and gamma-aminobutyric acid hypofunction.
The overall effect of these neurotoxic blows is a brain that experiences white and gray matter pathologies, leading to impaired neuroplasticity with increasing levels of white matter disconnectivity, Dr. Nasrallah said.
He pointed out that studies have shown a loss of 1% of total brain volume and 3% of gray matter volume with the first psychotic episode. Cerebral ventricles expand by about 7%. “The different parts of the brain no longer communicate properly with each other across myelinated fibers, which is postulated as an explanation for cognitive impairment, thought disorder, negative symptoms, and lack of insight – all of which can contribute to nonadherence to treatment.”
Prompt treatment of the first episode of psychosis and starting the patient on an LAI can protect the brain from another destructive round of neuroinflammation and oxidative stress. He recommends that his patients take omega-3 fatty acids to expedite the anti-inflammatory response of the antipsychotic drug. First-generation antipsychotics are not neuroprotective, according to Dr. Nasrallah, whose own research shows that haloperidol is itself neurotoxic and kills neurons – although it is an efficacious antipsychotic.
Updated American Psychiatric Association practice guidelines for treating first-episode psychosis, published in 2010, do not recommend LAIs as a first-line treatment. Neither do the National Institute of Mental Health’s Schizophrenia Patient Outcomes Research Team treatment recommendations and summary statements, published in 2009. Similarly, neither document stresses atypicals over first-generation antipsychotics.
However, Dr. Nasrallah cited a randomized, controlled study from the University of California, Los Angeles, showing that in 86 patients with first-episode psychosis who were given the same antipsychotic in either oral or LAI form, at the end of 1-year follow-up, the researchers reported a 650% higher relapse rate in the oral group (33%), compared with the LAI group (5%) (JAMA Psychiatry. 2015 Aug;72[8]:822-9).
“I tell my residents to behave like cardiologists,” Dr. Nasrallah said. “When cardiologists have a patient who experienced the first heart attack, they make it an absolute goal never to let the patient have another myocardial infarction because the first one permanently killed part of the myocardium, and a second heart attack will either kill the person or make him need a heart transplant. They implement a multifaceted intervention to achieve that goal. We psychiatrists should regard [the first episode of psychosis] as a ‘brain attack,’ and we should never let patients have another one again. The best intervention we have for this is starting an atypical LAI in first-episode patients.”
Global Academy and this news organization are owned by the same company. Dr. Nasrallah has conducted Food and Drug Administration clinical trials with LAIs and has several industry ties, including with Alkermes, Janssen, and Otsuka.
[email protected]
On Twitter @whitneymcknight
washington – As data mount confirming the neurodegenerative effects of psychotic episodes in schizophrenia, one expert urges psychiatrists to think of psychosis as a “brain attack” which, like heart attacks, must be prevented from recurring.
“Schizophrenia doesn’t have to be progressive neurodegenerative unless patients relapse again and again, but that happens all the time because we give our patients pills they don’t take as prescribed. There are many reasons for poor adherence,” Henry A. Nasrallah, MD, said at the meeting held by Global Academy for Medical Education.
In a presentation dedicated to the emerging science reshaping views on how schizophrenia occurs, how it can be prevented, and why it is a syndrome with genetic etiologies, Dr. Nasrallah emphasized that there now exist enough data to show that timely intervention with LAIs reliably prevent relapse in most patients, thereby averting progressive neurodegeneration and subsequent disability in people who develop schizophrenia.
“Researchers now stage schizophrenia. Just like cancer, the more advanced the stage, the worse the outcome,” said Dr. Nasrallah, the Sydney W. Souers Endowed Chair and professor of psychiatry and behavioral neuroscience at Saint Louis University, told his audience. “The additional damaging effects of the second episode is what leads to clinical deterioration and can start the process of treatment resistance. But if no psychotic episodes are allowed to recur after the first episode, many patients can return to their baseline functioning, such as school or work.”
The field still is clarifying the neurodevelopmental aspects of schizophrenia, including genetic and in utero adverse events that disrupt brain development, as well as the appropriate types and timing of intervention in the prodromal phase. However, Dr. Nasrallah explained, science already has demonstrated how the neurotoxic effects of psychosis in the brain of a person with schizophrenia lead to brain tissue degradation with every psychotic episode. The result is a progressive decline in social and vocational functioning.
Psychosis is associated with activation of microglia, which are monocytic cells that cross the blood-brain barrier during fetal life, settling in the brain and ultimately comprising 10%-15% of all brain cells. Once activated, they trigger an immune response, leading to neuroinflammation and oxidative stress (free radicals). However, Dr. Nasrallah said, rather than protect the brain, these processes destroy gray and white matter – particularly in the cortical region – degrading the brain and leaving it more compromised, especially if another episode of psychosis occurs.
Another factor in the vulnerability of the brain in schizophrenia is mitochondrial dysfunction. As mitochondria are the primary source of antioxidants – such as glutathione – the deficit in antioxidants increases oxidative stress, furthering the brain’s vulnerability to tissue loss.
Among other biological processes thought to be implicated in neurodegeneration with schizophrenia, Dr. Nasrallah said, are impairment of antiapoptotic signaling, glutamate excitotoxicity, hypercortisolemia, and gamma-aminobutyric acid hypofunction.
The overall effect of these neurotoxic blows is a brain that experiences white and gray matter pathologies, leading to impaired neuroplasticity with increasing levels of white matter disconnectivity, Dr. Nasrallah said.
He pointed out that studies have shown a loss of 1% of total brain volume and 3% of gray matter volume with the first psychotic episode. Cerebral ventricles expand by about 7%. “The different parts of the brain no longer communicate properly with each other across myelinated fibers, which is postulated as an explanation for cognitive impairment, thought disorder, negative symptoms, and lack of insight – all of which can contribute to nonadherence to treatment.”
Prompt treatment of the first episode of psychosis and starting the patient on an LAI can protect the brain from another destructive round of neuroinflammation and oxidative stress. He recommends that his patients take omega-3 fatty acids to expedite the anti-inflammatory response of the antipsychotic drug. First-generation antipsychotics are not neuroprotective, according to Dr. Nasrallah, whose own research shows that haloperidol is itself neurotoxic and kills neurons – although it is an efficacious antipsychotic.
Updated American Psychiatric Association practice guidelines for treating first-episode psychosis, published in 2010, do not recommend LAIs as a first-line treatment. Neither do the National Institute of Mental Health’s Schizophrenia Patient Outcomes Research Team treatment recommendations and summary statements, published in 2009. Similarly, neither document stresses atypicals over first-generation antipsychotics.
However, Dr. Nasrallah cited a randomized, controlled study from the University of California, Los Angeles, showing that in 86 patients with first-episode psychosis who were given the same antipsychotic in either oral or LAI form, at the end of 1-year follow-up, the researchers reported a 650% higher relapse rate in the oral group (33%), compared with the LAI group (5%) (JAMA Psychiatry. 2015 Aug;72[8]:822-9).
“I tell my residents to behave like cardiologists,” Dr. Nasrallah said. “When cardiologists have a patient who experienced the first heart attack, they make it an absolute goal never to let the patient have another myocardial infarction because the first one permanently killed part of the myocardium, and a second heart attack will either kill the person or make him need a heart transplant. They implement a multifaceted intervention to achieve that goal. We psychiatrists should regard [the first episode of psychosis] as a ‘brain attack,’ and we should never let patients have another one again. The best intervention we have for this is starting an atypical LAI in first-episode patients.”
Global Academy and this news organization are owned by the same company. Dr. Nasrallah has conducted Food and Drug Administration clinical trials with LAIs and has several industry ties, including with Alkermes, Janssen, and Otsuka.
[email protected]
On Twitter @whitneymcknight
washington – As data mount confirming the neurodegenerative effects of psychotic episodes in schizophrenia, one expert urges psychiatrists to think of psychosis as a “brain attack” which, like heart attacks, must be prevented from recurring.
“Schizophrenia doesn’t have to be progressive neurodegenerative unless patients relapse again and again, but that happens all the time because we give our patients pills they don’t take as prescribed. There are many reasons for poor adherence,” Henry A. Nasrallah, MD, said at the meeting held by Global Academy for Medical Education.
In a presentation dedicated to the emerging science reshaping views on how schizophrenia occurs, how it can be prevented, and why it is a syndrome with genetic etiologies, Dr. Nasrallah emphasized that there now exist enough data to show that timely intervention with LAIs reliably prevent relapse in most patients, thereby averting progressive neurodegeneration and subsequent disability in people who develop schizophrenia.
“Researchers now stage schizophrenia. Just like cancer, the more advanced the stage, the worse the outcome,” said Dr. Nasrallah, the Sydney W. Souers Endowed Chair and professor of psychiatry and behavioral neuroscience at Saint Louis University, told his audience. “The additional damaging effects of the second episode is what leads to clinical deterioration and can start the process of treatment resistance. But if no psychotic episodes are allowed to recur after the first episode, many patients can return to their baseline functioning, such as school or work.”
The field still is clarifying the neurodevelopmental aspects of schizophrenia, including genetic and in utero adverse events that disrupt brain development, as well as the appropriate types and timing of intervention in the prodromal phase. However, Dr. Nasrallah explained, science already has demonstrated how the neurotoxic effects of psychosis in the brain of a person with schizophrenia lead to brain tissue degradation with every psychotic episode. The result is a progressive decline in social and vocational functioning.
Psychosis is associated with activation of microglia, which are monocytic cells that cross the blood-brain barrier during fetal life, settling in the brain and ultimately comprising 10%-15% of all brain cells. Once activated, they trigger an immune response, leading to neuroinflammation and oxidative stress (free radicals). However, Dr. Nasrallah said, rather than protect the brain, these processes destroy gray and white matter – particularly in the cortical region – degrading the brain and leaving it more compromised, especially if another episode of psychosis occurs.
Another factor in the vulnerability of the brain in schizophrenia is mitochondrial dysfunction. As mitochondria are the primary source of antioxidants – such as glutathione – the deficit in antioxidants increases oxidative stress, furthering the brain’s vulnerability to tissue loss.
Among other biological processes thought to be implicated in neurodegeneration with schizophrenia, Dr. Nasrallah said, are impairment of antiapoptotic signaling, glutamate excitotoxicity, hypercortisolemia, and gamma-aminobutyric acid hypofunction.
The overall effect of these neurotoxic blows is a brain that experiences white and gray matter pathologies, leading to impaired neuroplasticity with increasing levels of white matter disconnectivity, Dr. Nasrallah said.
He pointed out that studies have shown a loss of 1% of total brain volume and 3% of gray matter volume with the first psychotic episode. Cerebral ventricles expand by about 7%. “The different parts of the brain no longer communicate properly with each other across myelinated fibers, which is postulated as an explanation for cognitive impairment, thought disorder, negative symptoms, and lack of insight – all of which can contribute to nonadherence to treatment.”
Prompt treatment of the first episode of psychosis and starting the patient on an LAI can protect the brain from another destructive round of neuroinflammation and oxidative stress. He recommends that his patients take omega-3 fatty acids to expedite the anti-inflammatory response of the antipsychotic drug. First-generation antipsychotics are not neuroprotective, according to Dr. Nasrallah, whose own research shows that haloperidol is itself neurotoxic and kills neurons – although it is an efficacious antipsychotic.
Updated American Psychiatric Association practice guidelines for treating first-episode psychosis, published in 2010, do not recommend LAIs as a first-line treatment. Neither do the National Institute of Mental Health’s Schizophrenia Patient Outcomes Research Team treatment recommendations and summary statements, published in 2009. Similarly, neither document stresses atypicals over first-generation antipsychotics.
However, Dr. Nasrallah cited a randomized, controlled study from the University of California, Los Angeles, showing that in 86 patients with first-episode psychosis who were given the same antipsychotic in either oral or LAI form, at the end of 1-year follow-up, the researchers reported a 650% higher relapse rate in the oral group (33%), compared with the LAI group (5%) (JAMA Psychiatry. 2015 Aug;72[8]:822-9).
“I tell my residents to behave like cardiologists,” Dr. Nasrallah said. “When cardiologists have a patient who experienced the first heart attack, they make it an absolute goal never to let the patient have another myocardial infarction because the first one permanently killed part of the myocardium, and a second heart attack will either kill the person or make him need a heart transplant. They implement a multifaceted intervention to achieve that goal. We psychiatrists should regard [the first episode of psychosis] as a ‘brain attack,’ and we should never let patients have another one again. The best intervention we have for this is starting an atypical LAI in first-episode patients.”
Global Academy and this news organization are owned by the same company. Dr. Nasrallah has conducted Food and Drug Administration clinical trials with LAIs and has several industry ties, including with Alkermes, Janssen, and Otsuka.
[email protected]
On Twitter @whitneymcknight
AT SUMMIT IN NEUROLOGY & PSYCHIATRY
Savolitinib improves PFS in some with MET-driven papillary renal cell carcinoma
Savolitinib, a highly selective inhibitor of c-Met receptor tyrosine kinase, improved progression-free survival in a small portion of patients with MET-driven advanced renal papillary cell cancer.
The molecule, being developed by Hutchison China MediTech Limited and AstraZeneca, extended progression-free survival by about 5 months in these patients, compared with those whose tumors were not MET driven (6.2 vs. 1.4 months, respectively), reported Toni Choueiri, MD, and his colleagues (J Clin Oncol 2017. doi: 10.1200/JCO.2017.72.2967).
Of 109 patients in the phase II study, 44 (40%) had MET-driven disease. The cancer was MET independent in 46 (42%); MET status was unknown in the remainder.
Half of those with MET-driven tumors experienced stable disease, and 61% experienced tumor shrinkage ranging from 0.7% to 66%. Tumor shrinkage occurred in 20% of those with MET-independent tumors. These also responded less vigorously (shrinkage 0.5%-20%).
Of the eight patients exhibiting a partial response, six were still responding to treatment at the study’s end, with response ranging from 2 to 16 months. Two patients experienced progressive disease after 1.8 and 2.8 months. By the end of the study, disease progression had occurred in 75% of the MET-driven cases, 96% of the MET-independent cases, and 74% of cases whose MET status was unknown.
“These results confirm that savolitinib … holds promise as a personalized treatment for patients with metastatic MET-driven papillary renal cell carcinoma,” the investigators wrote. The data also support the June launch of the phase III SAVOIR trial, they noted.
SAVOIR will enroll about 180 patients with MET-driven renal papillary cancer confirmed by a next-generation molecular sequencing developed by the companies for savolitinib response. Patients will be randomized to continuous treatment with savolitinib 600 mg orally, once daily, or intermittent treatment with sunitinib 50 mg orally once daily (4 weeks on/2 weeks off), on a 6-week cycle.
Dr. Choueiri has been a consultant for and received research funding from numerous pharmaceutical companies, including AstraZeneca.
[email protected]
On Twitter @Alz_gal
Savolitinib, a highly selective inhibitor of c-Met receptor tyrosine kinase, improved progression-free survival in a small portion of patients with MET-driven advanced renal papillary cell cancer.
The molecule, being developed by Hutchison China MediTech Limited and AstraZeneca, extended progression-free survival by about 5 months in these patients, compared with those whose tumors were not MET driven (6.2 vs. 1.4 months, respectively), reported Toni Choueiri, MD, and his colleagues (J Clin Oncol 2017. doi: 10.1200/JCO.2017.72.2967).
Of 109 patients in the phase II study, 44 (40%) had MET-driven disease. The cancer was MET independent in 46 (42%); MET status was unknown in the remainder.
Half of those with MET-driven tumors experienced stable disease, and 61% experienced tumor shrinkage ranging from 0.7% to 66%. Tumor shrinkage occurred in 20% of those with MET-independent tumors. These also responded less vigorously (shrinkage 0.5%-20%).
Of the eight patients exhibiting a partial response, six were still responding to treatment at the study’s end, with response ranging from 2 to 16 months. Two patients experienced progressive disease after 1.8 and 2.8 months. By the end of the study, disease progression had occurred in 75% of the MET-driven cases, 96% of the MET-independent cases, and 74% of cases whose MET status was unknown.
“These results confirm that savolitinib … holds promise as a personalized treatment for patients with metastatic MET-driven papillary renal cell carcinoma,” the investigators wrote. The data also support the June launch of the phase III SAVOIR trial, they noted.
SAVOIR will enroll about 180 patients with MET-driven renal papillary cancer confirmed by a next-generation molecular sequencing developed by the companies for savolitinib response. Patients will be randomized to continuous treatment with savolitinib 600 mg orally, once daily, or intermittent treatment with sunitinib 50 mg orally once daily (4 weeks on/2 weeks off), on a 6-week cycle.
Dr. Choueiri has been a consultant for and received research funding from numerous pharmaceutical companies, including AstraZeneca.
[email protected]
On Twitter @Alz_gal
Savolitinib, a highly selective inhibitor of c-Met receptor tyrosine kinase, improved progression-free survival in a small portion of patients with MET-driven advanced renal papillary cell cancer.
The molecule, being developed by Hutchison China MediTech Limited and AstraZeneca, extended progression-free survival by about 5 months in these patients, compared with those whose tumors were not MET driven (6.2 vs. 1.4 months, respectively), reported Toni Choueiri, MD, and his colleagues (J Clin Oncol 2017. doi: 10.1200/JCO.2017.72.2967).
Of 109 patients in the phase II study, 44 (40%) had MET-driven disease. The cancer was MET independent in 46 (42%); MET status was unknown in the remainder.
Half of those with MET-driven tumors experienced stable disease, and 61% experienced tumor shrinkage ranging from 0.7% to 66%. Tumor shrinkage occurred in 20% of those with MET-independent tumors. These also responded less vigorously (shrinkage 0.5%-20%).
Of the eight patients exhibiting a partial response, six were still responding to treatment at the study’s end, with response ranging from 2 to 16 months. Two patients experienced progressive disease after 1.8 and 2.8 months. By the end of the study, disease progression had occurred in 75% of the MET-driven cases, 96% of the MET-independent cases, and 74% of cases whose MET status was unknown.
“These results confirm that savolitinib … holds promise as a personalized treatment for patients with metastatic MET-driven papillary renal cell carcinoma,” the investigators wrote. The data also support the June launch of the phase III SAVOIR trial, they noted.
SAVOIR will enroll about 180 patients with MET-driven renal papillary cancer confirmed by a next-generation molecular sequencing developed by the companies for savolitinib response. Patients will be randomized to continuous treatment with savolitinib 600 mg orally, once daily, or intermittent treatment with sunitinib 50 mg orally once daily (4 weeks on/2 weeks off), on a 6-week cycle.
Dr. Choueiri has been a consultant for and received research funding from numerous pharmaceutical companies, including AstraZeneca.
[email protected]
On Twitter @Alz_gal
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: A partial response occurred in 18% of these patients, who gained about 5 months of progression-free disease compared with those with MET-independent tumors.
Data source: A phase II trial comprising 109 patients.
Disclosures: Dr. Choueiri has been a consultant for and received research funds from AstraZeneca, which is developing the drug.
Multiply recurrent C. difficile infection is on the rise
A retrospective cohort study of Clostridium difficile infection (CDI), the most common health care–associated infection, found that multiply recurrent CDI (mrCDI) is increasing in incidence, disproportionately to the overall increase in CDI.
Researchers from the University of Pennsylvania, Philadelphia, worked with a database of more than 38 million individuals with private health insurance between January 2001 and December 2012.
Cases of CDI and mrCDI in the study population were determined through ICD-9 diagnosis codes, and prescriptions for treatment. To meet the definition of mrCDI, there had to be at least three courses of treatment lasting at least 14 days each.
In the study population, 45,341 persons developed CDI, of whom 1,669 had mrCDI. The median age was 46 years, and 58.9% were female. Between 2001 and 2012, CDI incidence increased by 42.7% (P = .004), while mrCDI incidence increased by 188.8% (P less than .001).
With increases in CDI and mrCDI incidence, and with the effectiveness of standard antibiotic treatment decreasing with each recurrence, “demand for new antimicrobial therapies and FMT [fecal microbiota transplantation] can be expected to increase considerably in the coming years,” wrote Gene K. Ma, MD, and his coauthors.
As for FMT, the researchers noted that its likely greater demand in the future (as suggested by their study results) highlights the importance of establishing the long-term safety of the procedure (Ann Intern Med. 2017 Jul. doi: 10.7326/M16-2733).
The retrospective cohort study was based on administrative data rather than laboratory data, Sameer D. Saini, MD, MS, and Akbar K. Waljee, MD, noted in an editorial accompanying the study. Further, with Medicare patients excluded from the study (because Medicare data were not available for the full time period studied for private insurance data), the data may not be of relevance to patients older than age 65 years.
But the general conclusion that both CDI and mrCDI are on the rise is a crucial matter. “We must first have a better understanding of mrCDI, its scope and epidemiology, and its associated risk factors. The study by Ma and colleagues begins this important work. A better understanding of the epidemiology of mrCDI is a critical first step toward developing a sound strategy to address this growing public health challenge.”
Dr. Saini and Dr. Waljee are with the VA Ann Arbor (Michigan) Center for Clinical Management. Their editorial accompanied the study in Annals of Internal Medicine (2017 Jul. doi: 10.7326/M17-1565).
The retrospective cohort study was based on administrative data rather than laboratory data, Sameer D. Saini, MD, MS, and Akbar K. Waljee, MD, noted in an editorial accompanying the study. Further, with Medicare patients excluded from the study (because Medicare data were not available for the full time period studied for private insurance data), the data may not be of relevance to patients older than age 65 years.
But the general conclusion that both CDI and mrCDI are on the rise is a crucial matter. “We must first have a better understanding of mrCDI, its scope and epidemiology, and its associated risk factors. The study by Ma and colleagues begins this important work. A better understanding of the epidemiology of mrCDI is a critical first step toward developing a sound strategy to address this growing public health challenge.”
Dr. Saini and Dr. Waljee are with the VA Ann Arbor (Michigan) Center for Clinical Management. Their editorial accompanied the study in Annals of Internal Medicine (2017 Jul. doi: 10.7326/M17-1565).
The retrospective cohort study was based on administrative data rather than laboratory data, Sameer D. Saini, MD, MS, and Akbar K. Waljee, MD, noted in an editorial accompanying the study. Further, with Medicare patients excluded from the study (because Medicare data were not available for the full time period studied for private insurance data), the data may not be of relevance to patients older than age 65 years.
But the general conclusion that both CDI and mrCDI are on the rise is a crucial matter. “We must first have a better understanding of mrCDI, its scope and epidemiology, and its associated risk factors. The study by Ma and colleagues begins this important work. A better understanding of the epidemiology of mrCDI is a critical first step toward developing a sound strategy to address this growing public health challenge.”
Dr. Saini and Dr. Waljee are with the VA Ann Arbor (Michigan) Center for Clinical Management. Their editorial accompanied the study in Annals of Internal Medicine (2017 Jul. doi: 10.7326/M17-1565).
A retrospective cohort study of Clostridium difficile infection (CDI), the most common health care–associated infection, found that multiply recurrent CDI (mrCDI) is increasing in incidence, disproportionately to the overall increase in CDI.
Researchers from the University of Pennsylvania, Philadelphia, worked with a database of more than 38 million individuals with private health insurance between January 2001 and December 2012.
Cases of CDI and mrCDI in the study population were determined through ICD-9 diagnosis codes, and prescriptions for treatment. To meet the definition of mrCDI, there had to be at least three courses of treatment lasting at least 14 days each.
In the study population, 45,341 persons developed CDI, of whom 1,669 had mrCDI. The median age was 46 years, and 58.9% were female. Between 2001 and 2012, CDI incidence increased by 42.7% (P = .004), while mrCDI incidence increased by 188.8% (P less than .001).
With increases in CDI and mrCDI incidence, and with the effectiveness of standard antibiotic treatment decreasing with each recurrence, “demand for new antimicrobial therapies and FMT [fecal microbiota transplantation] can be expected to increase considerably in the coming years,” wrote Gene K. Ma, MD, and his coauthors.
As for FMT, the researchers noted that its likely greater demand in the future (as suggested by their study results) highlights the importance of establishing the long-term safety of the procedure (Ann Intern Med. 2017 Jul. doi: 10.7326/M16-2733).
A retrospective cohort study of Clostridium difficile infection (CDI), the most common health care–associated infection, found that multiply recurrent CDI (mrCDI) is increasing in incidence, disproportionately to the overall increase in CDI.
Researchers from the University of Pennsylvania, Philadelphia, worked with a database of more than 38 million individuals with private health insurance between January 2001 and December 2012.
Cases of CDI and mrCDI in the study population were determined through ICD-9 diagnosis codes, and prescriptions for treatment. To meet the definition of mrCDI, there had to be at least three courses of treatment lasting at least 14 days each.
In the study population, 45,341 persons developed CDI, of whom 1,669 had mrCDI. The median age was 46 years, and 58.9% were female. Between 2001 and 2012, CDI incidence increased by 42.7% (P = .004), while mrCDI incidence increased by 188.8% (P less than .001).
With increases in CDI and mrCDI incidence, and with the effectiveness of standard antibiotic treatment decreasing with each recurrence, “demand for new antimicrobial therapies and FMT [fecal microbiota transplantation] can be expected to increase considerably in the coming years,” wrote Gene K. Ma, MD, and his coauthors.
As for FMT, the researchers noted that its likely greater demand in the future (as suggested by their study results) highlights the importance of establishing the long-term safety of the procedure (Ann Intern Med. 2017 Jul. doi: 10.7326/M16-2733).
FROM ANNALS OF INTERNAL MEDICINE
Hospitalist burnout
Some things I’ve been thinking about:
- Physician well-being, morale, and burnout seem to be getting more attention in both the medical and the lay press.
- Leaders from 10 prestigious health systems and the CEO of the American Medical Association wrote a March 2017 post in the Health Affairs Blog titled “Physician Burnout is a Public Health Crisis: A Message To Our Fellow Health Care CEOs.”
- I’m now regularly hearing and reading mention of the “Quadruple Aim.” The “Triple Aim,” first described in 2008, is the pursuit of excellence in 1) patient experience – both quality of care and patient satisfaction; 2) population health; and 3) cost reduction. The November/December 2014 Annals of Family Medicine included an article recommending “that the Triple Aim be expanded to a Quadruple Aim by adding the goal of improving the work life of health care providers, including clinicians and staff.”1
- The CEO at a community hospital near me chose to make addressing physician burnout one of his top priorities and tied success in the effort to his own compensation bonus.
- In the course of my consulting work with hospitalist groups across the country, I’ve noticed a meaningful increase in the number of our colleagues who seem deeply unhappy with their work and/or burned out. The “Hospitalist Morale Index” may be a worthwhile way for a group to conduct an assessment.2
- I’m concerned that many other hospital care givers, including RNs, social workers, and others, are experiencing levels of distress and/or burnout that might be similar to that of physicians. From where I sit, they seem to be getting less attention, and I can’t tell if that is just because I’m not as immersed in their world or if it reflects reality. It’s pretty disappointing if it’s the latter.
For the most part, I think the causes of hospitalist distress and burnout are very similar to that of doctors in other specialties, and interventions to address the problem can be similar across specialties. Yet, each specialty probably differs in ways that are important to keep in mind.
Hospitalists also bear a huge burden related to observation status. Doctors in most other specialties rarely face complex decisions regarding whether observation is the right choice and are not so often the target of patient/family frustration and anger related to it.
Those seeking to address hospitalist burnout and well-being specifically should keep in mind these uniquely hospitalist issues. I think of them as a chronic disease to manage and mitigate, since “curing” them (making them go away entirely) is probably impossible for the foreseeable future.
What to do?
An Internet search on physician burnout, or other terms related to well-being, will yield more articles with advice to address the problem than you’ll ever have time to read. Trying to read all of them would likely lead to burnout! I think interventions can be divided into two broad categories: organizational efforts and personal efforts.
Like the 10 CEOs mentioned above, health care leaders should acknowledge physician distress and burnout as a meaningful issue that can impede organizational performance and that investments to address it can have a meaningful return on investment. The Health Affairs Blog post listed 11 things the CEOs committed to doing. It’s a list anyone working on this issue should review.
Doctors at The Mayo Clinic have published a great deal of research on physician burnout. In the March 7, 2017, JAMA, (summarized in a YouTube video) they describe several worthwhile organizational changes, as well as some personal strategies.3 They wrote about their experiences with interventions such as a deliberate curriculum to train doctors in self-care (self-reflection, mindfulness, etc.) in a series of one-hour lectures over several months.4 In November 2016, they published a meta-analysis of interventions to address burnout.5
In total, all of the worthwhile recommendations to address burnout leave me feeling like they’re a lot of work, and any individual intervention may not be as helpful as hoped, so that the best way to approach this is with a collection of interventions. In many ways, it is similar to the problem of readmissions: There is a lot of research out there, it’s hard to prove that any single intervention really works, and success lies in implementing a broad set of interventions. And success doesn’t equate to eliminating readmissions, only reducing them.
Coda: Is a sabbatical uniquely valuable for hospitalists?
I think a sabbatical might be a good idea for hospitalists. It also seems practical for other doctors, such as radiologists, anesthesiologists, and ED doctors, who don’t have 1:1 continuity relationships with patients. However, it is problematic for primary care doctors and specialists who need to maintain continuity relationships with patients and referring doctors that could be disrupted by a lengthy absence.
I’m not sure a sabbatical would reduce burnout much on its own, but, if properly structured, it seems very likely to reduce staffing turnover, and the sabbatical could be spent in ways that help rejuvenate interest and satisfaction in our work rather than simply taking a long vacation to travel and play golf, etc. It should probably be at least 3 months and better if it lasts a year. A common arrangement is that a doctor becomes eligible for the sabbatical after 10 years and is paid half of her usual compensation while away. I’d like to see more hospitalist groups do this.
Dr. Nelson has had a career in clinical practice as a hospitalist starting in 1988. He is cofounder and past president of SHM and principal in Nelson Flores Hospital Medicine Consultants. He is codirector for SHM’s practice management courses. Write to [email protected].
References
1. Bodenheimer, T and Sninsky, C. From Triple to Quadruple Aim: Care of the Patient Requires Care of the Provider. Ann Fam Med. Nov/Dec 2014.
2. Chandra, S et al. Introducing the Hospitalist Morale Index: A New Tool That May Be Relevant for Improving Provider Retention. JHM. June 2016.
3. Shanafelt, T, Dyrbye, L, West, C. Addressing Physician Burnout: The Way Forward. JAMA. March 7, 2017.
4. West, C et al. Intervention to Promote Physician Well-being, Job Satisfaction, and Professionalism: A Randomized Clinical Trial. JAMA Intern Med. 2014;174(4):527-33.
5. West, C et al. Interventions to prevent and reduce physician burnout: A systematic review and meta-analysis. The Lancet. Nov 5, 2016.
Some things I’ve been thinking about:
- Physician well-being, morale, and burnout seem to be getting more attention in both the medical and the lay press.
- Leaders from 10 prestigious health systems and the CEO of the American Medical Association wrote a March 2017 post in the Health Affairs Blog titled “Physician Burnout is a Public Health Crisis: A Message To Our Fellow Health Care CEOs.”
- I’m now regularly hearing and reading mention of the “Quadruple Aim.” The “Triple Aim,” first described in 2008, is the pursuit of excellence in 1) patient experience – both quality of care and patient satisfaction; 2) population health; and 3) cost reduction. The November/December 2014 Annals of Family Medicine included an article recommending “that the Triple Aim be expanded to a Quadruple Aim by adding the goal of improving the work life of health care providers, including clinicians and staff.”1
- The CEO at a community hospital near me chose to make addressing physician burnout one of his top priorities and tied success in the effort to his own compensation bonus.
- In the course of my consulting work with hospitalist groups across the country, I’ve noticed a meaningful increase in the number of our colleagues who seem deeply unhappy with their work and/or burned out. The “Hospitalist Morale Index” may be a worthwhile way for a group to conduct an assessment.2
- I’m concerned that many other hospital care givers, including RNs, social workers, and others, are experiencing levels of distress and/or burnout that might be similar to that of physicians. From where I sit, they seem to be getting less attention, and I can’t tell if that is just because I’m not as immersed in their world or if it reflects reality. It’s pretty disappointing if it’s the latter.
For the most part, I think the causes of hospitalist distress and burnout are very similar to that of doctors in other specialties, and interventions to address the problem can be similar across specialties. Yet, each specialty probably differs in ways that are important to keep in mind.
Hospitalists also bear a huge burden related to observation status. Doctors in most other specialties rarely face complex decisions regarding whether observation is the right choice and are not so often the target of patient/family frustration and anger related to it.
Those seeking to address hospitalist burnout and well-being specifically should keep in mind these uniquely hospitalist issues. I think of them as a chronic disease to manage and mitigate, since “curing” them (making them go away entirely) is probably impossible for the foreseeable future.
What to do?
An Internet search on physician burnout, or other terms related to well-being, will yield more articles with advice to address the problem than you’ll ever have time to read. Trying to read all of them would likely lead to burnout! I think interventions can be divided into two broad categories: organizational efforts and personal efforts.
Like the 10 CEOs mentioned above, health care leaders should acknowledge physician distress and burnout as a meaningful issue that can impede organizational performance and that investments to address it can have a meaningful return on investment. The Health Affairs Blog post listed 11 things the CEOs committed to doing. It’s a list anyone working on this issue should review.
Doctors at The Mayo Clinic have published a great deal of research on physician burnout. In the March 7, 2017, JAMA, (summarized in a YouTube video) they describe several worthwhile organizational changes, as well as some personal strategies.3 They wrote about their experiences with interventions such as a deliberate curriculum to train doctors in self-care (self-reflection, mindfulness, etc.) in a series of one-hour lectures over several months.4 In November 2016, they published a meta-analysis of interventions to address burnout.5
In total, all of the worthwhile recommendations to address burnout leave me feeling like they’re a lot of work, and any individual intervention may not be as helpful as hoped, so that the best way to approach this is with a collection of interventions. In many ways, it is similar to the problem of readmissions: There is a lot of research out there, it’s hard to prove that any single intervention really works, and success lies in implementing a broad set of interventions. And success doesn’t equate to eliminating readmissions, only reducing them.
Coda: Is a sabbatical uniquely valuable for hospitalists?
I think a sabbatical might be a good idea for hospitalists. It also seems practical for other doctors, such as radiologists, anesthesiologists, and ED doctors, who don’t have 1:1 continuity relationships with patients. However, it is problematic for primary care doctors and specialists who need to maintain continuity relationships with patients and referring doctors that could be disrupted by a lengthy absence.
I’m not sure a sabbatical would reduce burnout much on its own, but, if properly structured, it seems very likely to reduce staffing turnover, and the sabbatical could be spent in ways that help rejuvenate interest and satisfaction in our work rather than simply taking a long vacation to travel and play golf, etc. It should probably be at least 3 months and better if it lasts a year. A common arrangement is that a doctor becomes eligible for the sabbatical after 10 years and is paid half of her usual compensation while away. I’d like to see more hospitalist groups do this.
Dr. Nelson has had a career in clinical practice as a hospitalist starting in 1988. He is cofounder and past president of SHM and principal in Nelson Flores Hospital Medicine Consultants. He is codirector for SHM’s practice management courses. Write to [email protected].
References
1. Bodenheimer, T and Sninsky, C. From Triple to Quadruple Aim: Care of the Patient Requires Care of the Provider. Ann Fam Med. Nov/Dec 2014.
2. Chandra, S et al. Introducing the Hospitalist Morale Index: A New Tool That May Be Relevant for Improving Provider Retention. JHM. June 2016.
3. Shanafelt, T, Dyrbye, L, West, C. Addressing Physician Burnout: The Way Forward. JAMA. March 7, 2017.
4. West, C et al. Intervention to Promote Physician Well-being, Job Satisfaction, and Professionalism: A Randomized Clinical Trial. JAMA Intern Med. 2014;174(4):527-33.
5. West, C et al. Interventions to prevent and reduce physician burnout: A systematic review and meta-analysis. The Lancet. Nov 5, 2016.
Some things I’ve been thinking about:
- Physician well-being, morale, and burnout seem to be getting more attention in both the medical and the lay press.
- Leaders from 10 prestigious health systems and the CEO of the American Medical Association wrote a March 2017 post in the Health Affairs Blog titled “Physician Burnout is a Public Health Crisis: A Message To Our Fellow Health Care CEOs.”
- I’m now regularly hearing and reading mention of the “Quadruple Aim.” The “Triple Aim,” first described in 2008, is the pursuit of excellence in 1) patient experience – both quality of care and patient satisfaction; 2) population health; and 3) cost reduction. The November/December 2014 Annals of Family Medicine included an article recommending “that the Triple Aim be expanded to a Quadruple Aim by adding the goal of improving the work life of health care providers, including clinicians and staff.”1
- The CEO at a community hospital near me chose to make addressing physician burnout one of his top priorities and tied success in the effort to his own compensation bonus.
- In the course of my consulting work with hospitalist groups across the country, I’ve noticed a meaningful increase in the number of our colleagues who seem deeply unhappy with their work and/or burned out. The “Hospitalist Morale Index” may be a worthwhile way for a group to conduct an assessment.2
- I’m concerned that many other hospital care givers, including RNs, social workers, and others, are experiencing levels of distress and/or burnout that might be similar to that of physicians. From where I sit, they seem to be getting less attention, and I can’t tell if that is just because I’m not as immersed in their world or if it reflects reality. It’s pretty disappointing if it’s the latter.
For the most part, I think the causes of hospitalist distress and burnout are very similar to that of doctors in other specialties, and interventions to address the problem can be similar across specialties. Yet, each specialty probably differs in ways that are important to keep in mind.
Hospitalists also bear a huge burden related to observation status. Doctors in most other specialties rarely face complex decisions regarding whether observation is the right choice and are not so often the target of patient/family frustration and anger related to it.
Those seeking to address hospitalist burnout and well-being specifically should keep in mind these uniquely hospitalist issues. I think of them as a chronic disease to manage and mitigate, since “curing” them (making them go away entirely) is probably impossible for the foreseeable future.
What to do?
An Internet search on physician burnout, or other terms related to well-being, will yield more articles with advice to address the problem than you’ll ever have time to read. Trying to read all of them would likely lead to burnout! I think interventions can be divided into two broad categories: organizational efforts and personal efforts.
Like the 10 CEOs mentioned above, health care leaders should acknowledge physician distress and burnout as a meaningful issue that can impede organizational performance and that investments to address it can have a meaningful return on investment. The Health Affairs Blog post listed 11 things the CEOs committed to doing. It’s a list anyone working on this issue should review.
Doctors at The Mayo Clinic have published a great deal of research on physician burnout. In the March 7, 2017, JAMA, (summarized in a YouTube video) they describe several worthwhile organizational changes, as well as some personal strategies.3 They wrote about their experiences with interventions such as a deliberate curriculum to train doctors in self-care (self-reflection, mindfulness, etc.) in a series of one-hour lectures over several months.4 In November 2016, they published a meta-analysis of interventions to address burnout.5
In total, all of the worthwhile recommendations to address burnout leave me feeling like they’re a lot of work, and any individual intervention may not be as helpful as hoped, so that the best way to approach this is with a collection of interventions. In many ways, it is similar to the problem of readmissions: There is a lot of research out there, it’s hard to prove that any single intervention really works, and success lies in implementing a broad set of interventions. And success doesn’t equate to eliminating readmissions, only reducing them.
Coda: Is a sabbatical uniquely valuable for hospitalists?
I think a sabbatical might be a good idea for hospitalists. It also seems practical for other doctors, such as radiologists, anesthesiologists, and ED doctors, who don’t have 1:1 continuity relationships with patients. However, it is problematic for primary care doctors and specialists who need to maintain continuity relationships with patients and referring doctors that could be disrupted by a lengthy absence.
I’m not sure a sabbatical would reduce burnout much on its own, but, if properly structured, it seems very likely to reduce staffing turnover, and the sabbatical could be spent in ways that help rejuvenate interest and satisfaction in our work rather than simply taking a long vacation to travel and play golf, etc. It should probably be at least 3 months and better if it lasts a year. A common arrangement is that a doctor becomes eligible for the sabbatical after 10 years and is paid half of her usual compensation while away. I’d like to see more hospitalist groups do this.
Dr. Nelson has had a career in clinical practice as a hospitalist starting in 1988. He is cofounder and past president of SHM and principal in Nelson Flores Hospital Medicine Consultants. He is codirector for SHM’s practice management courses. Write to [email protected].
References
1. Bodenheimer, T and Sninsky, C. From Triple to Quadruple Aim: Care of the Patient Requires Care of the Provider. Ann Fam Med. Nov/Dec 2014.
2. Chandra, S et al. Introducing the Hospitalist Morale Index: A New Tool That May Be Relevant for Improving Provider Retention. JHM. June 2016.
3. Shanafelt, T, Dyrbye, L, West, C. Addressing Physician Burnout: The Way Forward. JAMA. March 7, 2017.
4. West, C et al. Intervention to Promote Physician Well-being, Job Satisfaction, and Professionalism: A Randomized Clinical Trial. JAMA Intern Med. 2014;174(4):527-33.
5. West, C et al. Interventions to prevent and reduce physician burnout: A systematic review and meta-analysis. The Lancet. Nov 5, 2016.










