User login
Cutis is a peer-reviewed clinical journal for the dermatologist, allergist, and general practitioner published monthly since 1965. Concise clinical articles present the practical side of dermatology, helping physicians to improve patient care. Cutis is referenced in Index Medicus/MEDLINE and is written and edited by industry leaders.
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')
A peer-reviewed, indexed journal for dermatologists with original research, image quizzes, cases and reviews, and columns.
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
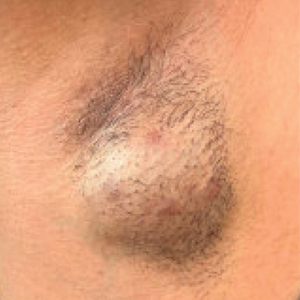
Hidradenitis suppurativa (HS) is a chronic relapsing inflammatory skin disorder affecting apocrine gland–bearing areas such as the axillae, inguinal regions, and anogenital area.1 It manifests with painful nodules, abscesses, sinus tract formation, and scarring.2 The disease strongly impacts patients’ quality of life due to pain, malodor, and psychosocial burden.3
The exact etiology of HS is multifactorial, involving genetic predisposition, mechanical stress, hormonal influences, dysbiosis, and immune dysregulation.4 Obesity and metabolic syndrome are highly prevalent among patients with HS and are considered exacerbating factors.5 Adipose tissue contributes to systemic inflammation through the secretion of proinflammatory cytokines such as tumor necrosis factor (TNF) α and interleukins (ILs).6
Management of HS includes lifestyle modifications, medical therapy, and surgical interventions. Medical treatments encompass antibiotics, retinoids, hormonal therapy, immunosuppressants, and immunomodulators such as anti-TNF and anti–IL-17 agents.7 Despite available therapies, many patients have suboptimal responses or experience adverse effects and dramatic reductions in their quality of life.3
Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) are incretin-based therapies used in type 2 diabetes and obesity management.8 They enhance insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety.9 Beyond glycemic control, GLP-1 RAs exhibit anti-inflammatory properties and cardiovascular benefits.10
Given the high prevalence of obesity and metabolic syndrome in patients with HS as well as the anti-inflammatory effects of GLP-1 RAs, these agents may offer therapeutic benefits in HS.11 We conducted a systematic review to evaluate the existing evidence on the efficacy and safety of GLP-1 RAs in the treatment of HS.
Methods
A systematic review was conducted via a PubMed search of articles indexed for MEDLINE in October 2024, following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines12 using the terms hidradenitis suppurativa OR acne inversa AND GLP-1 receptor agonist OR glucagon-like peptide-1 receptor agonist OR liraglutide OR semaglutide OR exenatide OR dulaglutide. No filters were applied to limit the search by language or publication date.
Inclusion criteria were clinical trials, observational studies (cohort, case control, cross-sectional), and case reports/series involving patients diagnosed with HS treated with GLP-1 RAs. Outcomes of interest included clinical improvement in HS severity (eg, lesion count, pain assessment, HS-specific scores), safety, and adverse events. Exclusion criteria included animal studies or in vitro experiments, reviews, editorials, and opinion pieces without original patient data; studies not in English; and studies not reporting clinical outcomes related to HS.
Two independent reviewers (N.R.K. and S.K.C.) screened the titles and abstracts for relevance. Full-text articles of potentially eligible studies were retrieved for detailed evaluation. Data extracted included study design, patient demographics, intervention details, outcomes, and adverse events. Discrepancies were resolved through discussion.
Results
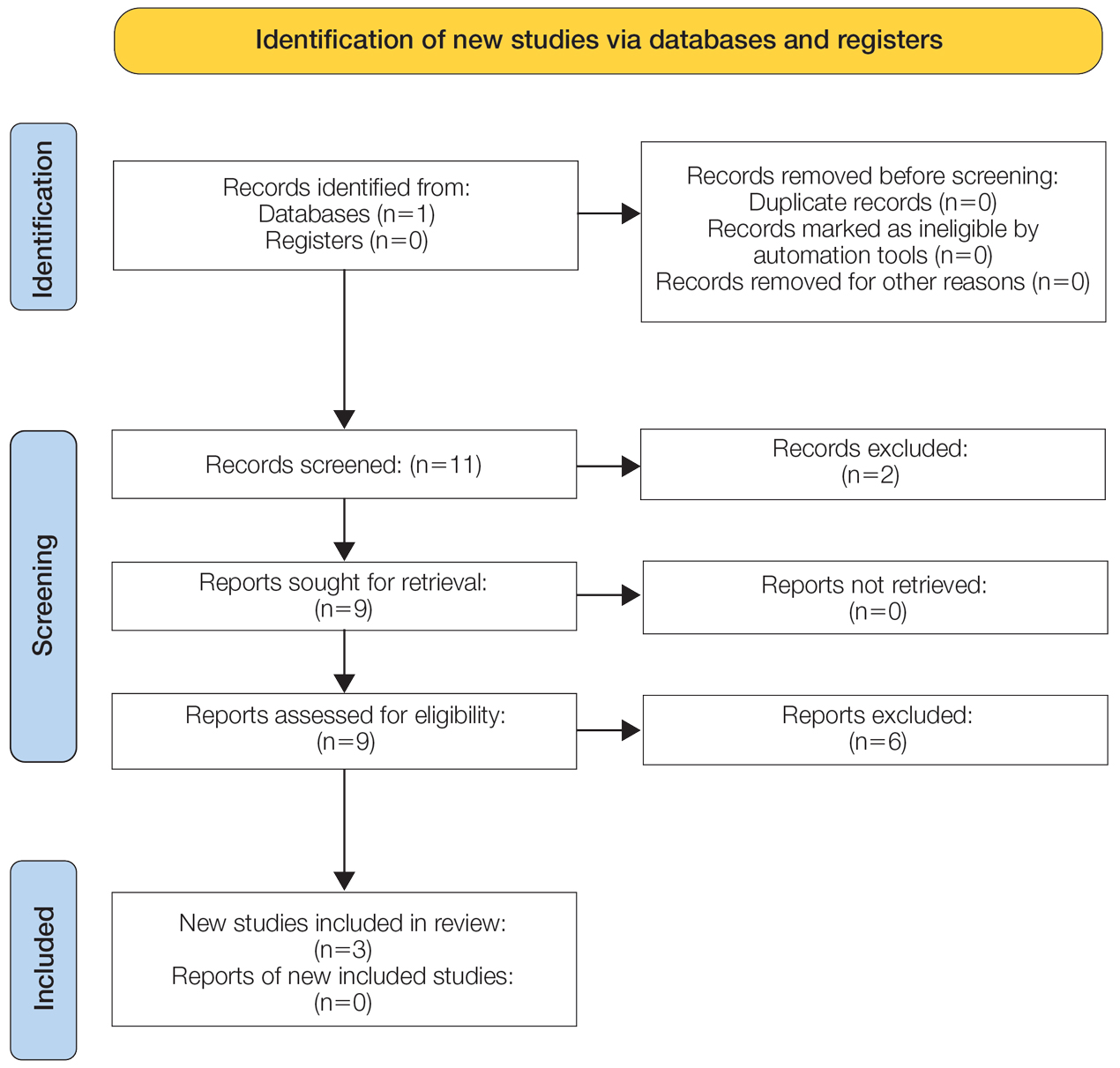
The initial search yielded 11 articles (Figure). After screening titles and abstracts, 9 articles were selected for full-text review. Of these, 3 articles met the inclusion criteria. These studies included 3 case reports. Interventions involved liraglutide (2 reports)13,14 and semaglutide15 (1 report)(Table). The patient population consisted of adult patients with HS with comorbid diabetes, obesity, and/or metabolic syndrome.
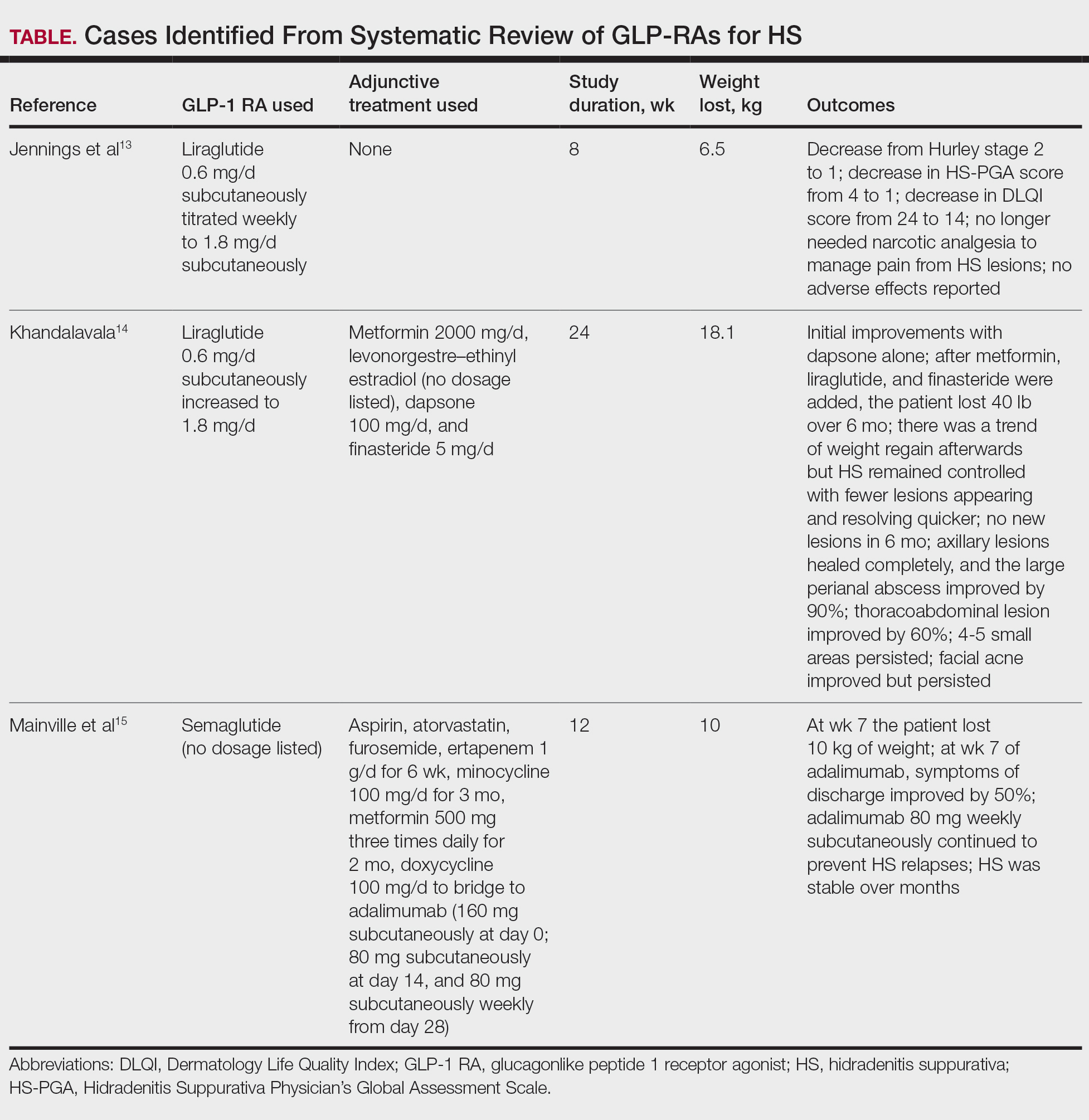
Jennings et al13 reported a 31-year-old obese woman with a history of smoking and Hurley stage 2 HS, a Hidradenitis Suppurativa Physician’s Global Assessment score of 4, a Dermatology Life Quality Index score of 24, and a body mass index of 45.3. She was treated with liraglutide monotherapy, starting with 0.6 mg subcutaneously once daily then titrating weekly to 1.8 mg subcutaneously. After 4 weeks, outcomes showed a reduction in Hidradenitis Suppurativa Physician’s Global Assessment (score=1) and Dermatology Life Quality Index (score=14) scores, and the patient lost 4.5 kg from baseline. The patient’s Hurley stage decreased from 2 to 1. After another 4 weeks, the patient’s weight decreased by a further 2 kg and HS remained controlled. No adverse events were recorded.
Khandalavala14 reported a single case of a 19-year-old woman with severe HS, obesity, and metabolic syndrome of 8 years’ duration treated with liraglutide. The patient had a weight of 215 lb with a body mass index of 37. With a combination of metformin 2000 mg/d, liraglutide 0.6 mg/d subcutaneously increased to 1.8 mg/d over 2 months, levonorgestrel-ethinyl estradiol (no dosage listed), dapsone 100 mg/d, and finasteride 5 mg/d, there was a marked reduction in nodules and abscesses after 6 months, with a weight loss of 40 lb (19% body weight). No adverse events were reported.
Mainville et al15 described a 59-year-old woman with refractory HS who showed improvement with a combination of intravenous ertapenem 1 g/d for 6 weeks, minocycline 100 mg/d for 3 months, metformin 500 mg three times daily for 2 months, doxycycline 100 mg/d to bridge to adalimumab (160 mg subcutaneously starting dose then 80 mg subcutaneously), and semaglutide (no dosage listed). After semaglutide was introduced, the patient lost 10 kg. The only adverse event was diarrhea.
Comment
The limited but growing body of evidence suggests that GLP-1 RAs may be beneficial in managing HS, particularly in patients with comorbid obesity. Treatment with liraglutide or semaglutide was associated with marked improvements in clinical severity scores, lesion counts, pain reduction, and quality of life.
As adjunct therapy, GLP-1 RAs could serve alongside standard HS treatments such as antibiotics and biologics. Addressing obesity, a known risk factor and disease modifier in HS, may lead to better disease control. The therapeutic benefits of GLP-1 RAs in HS are attributed to weight loss, which reduces adipose tissue and systemic inflammation.16 The anti-inflammatory effects of GLP-1 RAs involve the reduction of proinflammatory cytokines such as IL-6 and TNF-α.17 Metabolic improvements, including enhanced insulin sensitivity and lipid profile, also may contribute to disease modulation.17
Limitations—Because our analysis was limited to 3 case reports, the strength of the evidence is limited. These case reports also lack the standardized use of the Hidradenitis Suppurativa Clinical Response scoring system that generally is found in randomized controlled trials (RCTs). The lack of RCTs precludes definitive conclusions about efficacy. Future directions include the need for well-designed RCTs with large sample sizes to confirm findings, assessment of long-term safety and tolerability in patients with HS, and further research into the molecular mechanisms by which GLP-1 RAs affect HS pathophysiology. Of note, it is imperative to be aware of the medication shortage for all GLP-1 RAs when prescribing these medications for patients with HS.
Conclusion
Glucagonlike peptide 1 RAs show promise as a therapeutic option for HS, especially in patients with obesity and metabolic disturbances. The observed benefits likely result from weight loss and anti-inflammatory effects. Other drugs targeting glucose-dependent insulinotropic polypeptide and glucagon also are being studied thoroughly as options for managing HS. Although preliminary results are encouraging, robust clinical trials are needed to establish efficacy, optimal dosing, and safety in this patient population.
- Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
- Napolitano M, Megna M, Timoshchuk EA, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105-115. doi:10.2147/CCID.S111019
- Chernyshov PV, Finlay AY, Tomas-Aragones L, et al. Quality of life in hidradenitis suppurativa: an update. Int J Environ Res Public Health. 2021;18:6131. doi:10.3390/ijerph18116131
- Seyed Jafari SM, Hunger RE, Schlapbach C. Hidradenitis suppurativa: current understanding of pathogenic mechanisms and suggestion for treatment algorithm. Front Med (Lausanne). 2020;7:68. doi:10.3389/fmed.2020.00068
- Alotaibi HM. Incidence, risk factors, and prognosis of hidradenitis suppurativa across the globe: insights from the literature. Clin Cosmet Investig Dermatol. 2023;16:545-552. doi:10.2147/CCID.S402453
- Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi:10.3389/fimmu.2018.02965
- Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi:10.1159/000507323
- Brown E, Cuthbertson DJ, Wilding JP. Newer GLP-1 receptor agonists and obesity-diabetes. Peptides. 2018;100:61-67. doi:10.1016/j.peptides.2017.12.009
- Cornell S. A review of GLP‐1 receptor agonists in type 2 diabetes: a focus on the mechanism of action of once‐weekly agents. J Clin Pharm Ther. 2020;45(suppl 1):17-27. doi:10.1111/jcpt.13230
- Lee YS, Jun HS. Anti-inflammatory effects of GLP-1-based therapies beyond glucose control. Mediators Inflamm. 2016;2016:3094642. doi:10.1155/2016/3094642
- Mintoff D, Benhadou F, Pace NP, et al. Metabolic syndrome and hidradenitis suppurativa: epidemiological, molecular, and therapeutic aspects. Int J Dermatol. 2022;61:1175-1186. doi:10.1111/ijd.15910
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi:10.1159/000473873
- Mainville L, MacHaalany J, Veillette H. Hidradenitis suppurativa patient requiring cardiac procedure with inguinal access: case management with ertapenem. SAGE Open Med Case Rep. 2024;12:2050313X241274819. doi:10.1177/2050313X241274819
- Hamed K, Alosaimi MN, Ali BA, et al. Glucagon-like peptide-1 (GLP-1) receptor agonists: exploring their impact on diabetes, obesity, and cardiovascular health through a comprehensive literature review. Cureus. 2024;16:E68390. doi:10.7759/cureus.68390
- Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367. doi:10.1177/20420188231222367
Hidradenitis suppurativa (HS) is a chronic relapsing inflammatory skin disorder affecting apocrine gland–bearing areas such as the axillae, inguinal regions, and anogenital area.1 It manifests with painful nodules, abscesses, sinus tract formation, and scarring.2 The disease strongly impacts patients’ quality of life due to pain, malodor, and psychosocial burden.3
The exact etiology of HS is multifactorial, involving genetic predisposition, mechanical stress, hormonal influences, dysbiosis, and immune dysregulation.4 Obesity and metabolic syndrome are highly prevalent among patients with HS and are considered exacerbating factors.5 Adipose tissue contributes to systemic inflammation through the secretion of proinflammatory cytokines such as tumor necrosis factor (TNF) α and interleukins (ILs).6
Management of HS includes lifestyle modifications, medical therapy, and surgical interventions. Medical treatments encompass antibiotics, retinoids, hormonal therapy, immunosuppressants, and immunomodulators such as anti-TNF and anti–IL-17 agents.7 Despite available therapies, many patients have suboptimal responses or experience adverse effects and dramatic reductions in their quality of life.3
Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) are incretin-based therapies used in type 2 diabetes and obesity management.8 They enhance insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety.9 Beyond glycemic control, GLP-1 RAs exhibit anti-inflammatory properties and cardiovascular benefits.10
Given the high prevalence of obesity and metabolic syndrome in patients with HS as well as the anti-inflammatory effects of GLP-1 RAs, these agents may offer therapeutic benefits in HS.11 We conducted a systematic review to evaluate the existing evidence on the efficacy and safety of GLP-1 RAs in the treatment of HS.
Methods
A systematic review was conducted via a PubMed search of articles indexed for MEDLINE in October 2024, following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines12 using the terms hidradenitis suppurativa OR acne inversa AND GLP-1 receptor agonist OR glucagon-like peptide-1 receptor agonist OR liraglutide OR semaglutide OR exenatide OR dulaglutide. No filters were applied to limit the search by language or publication date.
Inclusion criteria were clinical trials, observational studies (cohort, case control, cross-sectional), and case reports/series involving patients diagnosed with HS treated with GLP-1 RAs. Outcomes of interest included clinical improvement in HS severity (eg, lesion count, pain assessment, HS-specific scores), safety, and adverse events. Exclusion criteria included animal studies or in vitro experiments, reviews, editorials, and opinion pieces without original patient data; studies not in English; and studies not reporting clinical outcomes related to HS.
Two independent reviewers (N.R.K. and S.K.C.) screened the titles and abstracts for relevance. Full-text articles of potentially eligible studies were retrieved for detailed evaluation. Data extracted included study design, patient demographics, intervention details, outcomes, and adverse events. Discrepancies were resolved through discussion.
Results
The initial search yielded 11 articles (Figure). After screening titles and abstracts, 9 articles were selected for full-text review. Of these, 3 articles met the inclusion criteria. These studies included 3 case reports. Interventions involved liraglutide (2 reports)13,14 and semaglutide15 (1 report)(Table). The patient population consisted of adult patients with HS with comorbid diabetes, obesity, and/or metabolic syndrome.


Jennings et al13 reported a 31-year-old obese woman with a history of smoking and Hurley stage 2 HS, a Hidradenitis Suppurativa Physician’s Global Assessment score of 4, a Dermatology Life Quality Index score of 24, and a body mass index of 45.3. She was treated with liraglutide monotherapy, starting with 0.6 mg subcutaneously once daily then titrating weekly to 1.8 mg subcutaneously. After 4 weeks, outcomes showed a reduction in Hidradenitis Suppurativa Physician’s Global Assessment (score=1) and Dermatology Life Quality Index (score=14) scores, and the patient lost 4.5 kg from baseline. The patient’s Hurley stage decreased from 2 to 1. After another 4 weeks, the patient’s weight decreased by a further 2 kg and HS remained controlled. No adverse events were recorded.
Khandalavala14 reported a single case of a 19-year-old woman with severe HS, obesity, and metabolic syndrome of 8 years’ duration treated with liraglutide. The patient had a weight of 215 lb with a body mass index of 37. With a combination of metformin 2000 mg/d, liraglutide 0.6 mg/d subcutaneously increased to 1.8 mg/d over 2 months, levonorgestrel-ethinyl estradiol (no dosage listed), dapsone 100 mg/d, and finasteride 5 mg/d, there was a marked reduction in nodules and abscesses after 6 months, with a weight loss of 40 lb (19% body weight). No adverse events were reported.
Mainville et al15 described a 59-year-old woman with refractory HS who showed improvement with a combination of intravenous ertapenem 1 g/d for 6 weeks, minocycline 100 mg/d for 3 months, metformin 500 mg three times daily for 2 months, doxycycline 100 mg/d to bridge to adalimumab (160 mg subcutaneously starting dose then 80 mg subcutaneously), and semaglutide (no dosage listed). After semaglutide was introduced, the patient lost 10 kg. The only adverse event was diarrhea.
Comment
The limited but growing body of evidence suggests that GLP-1 RAs may be beneficial in managing HS, particularly in patients with comorbid obesity. Treatment with liraglutide or semaglutide was associated with marked improvements in clinical severity scores, lesion counts, pain reduction, and quality of life.
As adjunct therapy, GLP-1 RAs could serve alongside standard HS treatments such as antibiotics and biologics. Addressing obesity, a known risk factor and disease modifier in HS, may lead to better disease control. The therapeutic benefits of GLP-1 RAs in HS are attributed to weight loss, which reduces adipose tissue and systemic inflammation.16 The anti-inflammatory effects of GLP-1 RAs involve the reduction of proinflammatory cytokines such as IL-6 and TNF-α.17 Metabolic improvements, including enhanced insulin sensitivity and lipid profile, also may contribute to disease modulation.17
Limitations—Because our analysis was limited to 3 case reports, the strength of the evidence is limited. These case reports also lack the standardized use of the Hidradenitis Suppurativa Clinical Response scoring system that generally is found in randomized controlled trials (RCTs). The lack of RCTs precludes definitive conclusions about efficacy. Future directions include the need for well-designed RCTs with large sample sizes to confirm findings, assessment of long-term safety and tolerability in patients with HS, and further research into the molecular mechanisms by which GLP-1 RAs affect HS pathophysiology. Of note, it is imperative to be aware of the medication shortage for all GLP-1 RAs when prescribing these medications for patients with HS.
Conclusion
Glucagonlike peptide 1 RAs show promise as a therapeutic option for HS, especially in patients with obesity and metabolic disturbances. The observed benefits likely result from weight loss and anti-inflammatory effects. Other drugs targeting glucose-dependent insulinotropic polypeptide and glucagon also are being studied thoroughly as options for managing HS. Although preliminary results are encouraging, robust clinical trials are needed to establish efficacy, optimal dosing, and safety in this patient population.
Hidradenitis suppurativa (HS) is a chronic relapsing inflammatory skin disorder affecting apocrine gland–bearing areas such as the axillae, inguinal regions, and anogenital area.1 It manifests with painful nodules, abscesses, sinus tract formation, and scarring.2 The disease strongly impacts patients’ quality of life due to pain, malodor, and psychosocial burden.3
The exact etiology of HS is multifactorial, involving genetic predisposition, mechanical stress, hormonal influences, dysbiosis, and immune dysregulation.4 Obesity and metabolic syndrome are highly prevalent among patients with HS and are considered exacerbating factors.5 Adipose tissue contributes to systemic inflammation through the secretion of proinflammatory cytokines such as tumor necrosis factor (TNF) α and interleukins (ILs).6
Management of HS includes lifestyle modifications, medical therapy, and surgical interventions. Medical treatments encompass antibiotics, retinoids, hormonal therapy, immunosuppressants, and immunomodulators such as anti-TNF and anti–IL-17 agents.7 Despite available therapies, many patients have suboptimal responses or experience adverse effects and dramatic reductions in their quality of life.3
Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) are incretin-based therapies used in type 2 diabetes and obesity management.8 They enhance insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety.9 Beyond glycemic control, GLP-1 RAs exhibit anti-inflammatory properties and cardiovascular benefits.10
Given the high prevalence of obesity and metabolic syndrome in patients with HS as well as the anti-inflammatory effects of GLP-1 RAs, these agents may offer therapeutic benefits in HS.11 We conducted a systematic review to evaluate the existing evidence on the efficacy and safety of GLP-1 RAs in the treatment of HS.
Methods
A systematic review was conducted via a PubMed search of articles indexed for MEDLINE in October 2024, following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines12 using the terms hidradenitis suppurativa OR acne inversa AND GLP-1 receptor agonist OR glucagon-like peptide-1 receptor agonist OR liraglutide OR semaglutide OR exenatide OR dulaglutide. No filters were applied to limit the search by language or publication date.
Inclusion criteria were clinical trials, observational studies (cohort, case control, cross-sectional), and case reports/series involving patients diagnosed with HS treated with GLP-1 RAs. Outcomes of interest included clinical improvement in HS severity (eg, lesion count, pain assessment, HS-specific scores), safety, and adverse events. Exclusion criteria included animal studies or in vitro experiments, reviews, editorials, and opinion pieces without original patient data; studies not in English; and studies not reporting clinical outcomes related to HS.
Two independent reviewers (N.R.K. and S.K.C.) screened the titles and abstracts for relevance. Full-text articles of potentially eligible studies were retrieved for detailed evaluation. Data extracted included study design, patient demographics, intervention details, outcomes, and adverse events. Discrepancies were resolved through discussion.
Results
The initial search yielded 11 articles (Figure). After screening titles and abstracts, 9 articles were selected for full-text review. Of these, 3 articles met the inclusion criteria. These studies included 3 case reports. Interventions involved liraglutide (2 reports)13,14 and semaglutide15 (1 report)(Table). The patient population consisted of adult patients with HS with comorbid diabetes, obesity, and/or metabolic syndrome.


Jennings et al13 reported a 31-year-old obese woman with a history of smoking and Hurley stage 2 HS, a Hidradenitis Suppurativa Physician’s Global Assessment score of 4, a Dermatology Life Quality Index score of 24, and a body mass index of 45.3. She was treated with liraglutide monotherapy, starting with 0.6 mg subcutaneously once daily then titrating weekly to 1.8 mg subcutaneously. After 4 weeks, outcomes showed a reduction in Hidradenitis Suppurativa Physician’s Global Assessment (score=1) and Dermatology Life Quality Index (score=14) scores, and the patient lost 4.5 kg from baseline. The patient’s Hurley stage decreased from 2 to 1. After another 4 weeks, the patient’s weight decreased by a further 2 kg and HS remained controlled. No adverse events were recorded.
Khandalavala14 reported a single case of a 19-year-old woman with severe HS, obesity, and metabolic syndrome of 8 years’ duration treated with liraglutide. The patient had a weight of 215 lb with a body mass index of 37. With a combination of metformin 2000 mg/d, liraglutide 0.6 mg/d subcutaneously increased to 1.8 mg/d over 2 months, levonorgestrel-ethinyl estradiol (no dosage listed), dapsone 100 mg/d, and finasteride 5 mg/d, there was a marked reduction in nodules and abscesses after 6 months, with a weight loss of 40 lb (19% body weight). No adverse events were reported.
Mainville et al15 described a 59-year-old woman with refractory HS who showed improvement with a combination of intravenous ertapenem 1 g/d for 6 weeks, minocycline 100 mg/d for 3 months, metformin 500 mg three times daily for 2 months, doxycycline 100 mg/d to bridge to adalimumab (160 mg subcutaneously starting dose then 80 mg subcutaneously), and semaglutide (no dosage listed). After semaglutide was introduced, the patient lost 10 kg. The only adverse event was diarrhea.
Comment
The limited but growing body of evidence suggests that GLP-1 RAs may be beneficial in managing HS, particularly in patients with comorbid obesity. Treatment with liraglutide or semaglutide was associated with marked improvements in clinical severity scores, lesion counts, pain reduction, and quality of life.
As adjunct therapy, GLP-1 RAs could serve alongside standard HS treatments such as antibiotics and biologics. Addressing obesity, a known risk factor and disease modifier in HS, may lead to better disease control. The therapeutic benefits of GLP-1 RAs in HS are attributed to weight loss, which reduces adipose tissue and systemic inflammation.16 The anti-inflammatory effects of GLP-1 RAs involve the reduction of proinflammatory cytokines such as IL-6 and TNF-α.17 Metabolic improvements, including enhanced insulin sensitivity and lipid profile, also may contribute to disease modulation.17
Limitations—Because our analysis was limited to 3 case reports, the strength of the evidence is limited. These case reports also lack the standardized use of the Hidradenitis Suppurativa Clinical Response scoring system that generally is found in randomized controlled trials (RCTs). The lack of RCTs precludes definitive conclusions about efficacy. Future directions include the need for well-designed RCTs with large sample sizes to confirm findings, assessment of long-term safety and tolerability in patients with HS, and further research into the molecular mechanisms by which GLP-1 RAs affect HS pathophysiology. Of note, it is imperative to be aware of the medication shortage for all GLP-1 RAs when prescribing these medications for patients with HS.
Conclusion
Glucagonlike peptide 1 RAs show promise as a therapeutic option for HS, especially in patients with obesity and metabolic disturbances. The observed benefits likely result from weight loss and anti-inflammatory effects. Other drugs targeting glucose-dependent insulinotropic polypeptide and glucagon also are being studied thoroughly as options for managing HS. Although preliminary results are encouraging, robust clinical trials are needed to establish efficacy, optimal dosing, and safety in this patient population.
- Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
- Napolitano M, Megna M, Timoshchuk EA, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105-115. doi:10.2147/CCID.S111019
- Chernyshov PV, Finlay AY, Tomas-Aragones L, et al. Quality of life in hidradenitis suppurativa: an update. Int J Environ Res Public Health. 2021;18:6131. doi:10.3390/ijerph18116131
- Seyed Jafari SM, Hunger RE, Schlapbach C. Hidradenitis suppurativa: current understanding of pathogenic mechanisms and suggestion for treatment algorithm. Front Med (Lausanne). 2020;7:68. doi:10.3389/fmed.2020.00068
- Alotaibi HM. Incidence, risk factors, and prognosis of hidradenitis suppurativa across the globe: insights from the literature. Clin Cosmet Investig Dermatol. 2023;16:545-552. doi:10.2147/CCID.S402453
- Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi:10.3389/fimmu.2018.02965
- Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi:10.1159/000507323
- Brown E, Cuthbertson DJ, Wilding JP. Newer GLP-1 receptor agonists and obesity-diabetes. Peptides. 2018;100:61-67. doi:10.1016/j.peptides.2017.12.009
- Cornell S. A review of GLP‐1 receptor agonists in type 2 diabetes: a focus on the mechanism of action of once‐weekly agents. J Clin Pharm Ther. 2020;45(suppl 1):17-27. doi:10.1111/jcpt.13230
- Lee YS, Jun HS. Anti-inflammatory effects of GLP-1-based therapies beyond glucose control. Mediators Inflamm. 2016;2016:3094642. doi:10.1155/2016/3094642
- Mintoff D, Benhadou F, Pace NP, et al. Metabolic syndrome and hidradenitis suppurativa: epidemiological, molecular, and therapeutic aspects. Int J Dermatol. 2022;61:1175-1186. doi:10.1111/ijd.15910
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi:10.1159/000473873
- Mainville L, MacHaalany J, Veillette H. Hidradenitis suppurativa patient requiring cardiac procedure with inguinal access: case management with ertapenem. SAGE Open Med Case Rep. 2024;12:2050313X241274819. doi:10.1177/2050313X241274819
- Hamed K, Alosaimi MN, Ali BA, et al. Glucagon-like peptide-1 (GLP-1) receptor agonists: exploring their impact on diabetes, obesity, and cardiovascular health through a comprehensive literature review. Cureus. 2024;16:E68390. doi:10.7759/cureus.68390
- Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367. doi:10.1177/20420188231222367
- Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
- Napolitano M, Megna M, Timoshchuk EA, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105-115. doi:10.2147/CCID.S111019
- Chernyshov PV, Finlay AY, Tomas-Aragones L, et al. Quality of life in hidradenitis suppurativa: an update. Int J Environ Res Public Health. 2021;18:6131. doi:10.3390/ijerph18116131
- Seyed Jafari SM, Hunger RE, Schlapbach C. Hidradenitis suppurativa: current understanding of pathogenic mechanisms and suggestion for treatment algorithm. Front Med (Lausanne). 2020;7:68. doi:10.3389/fmed.2020.00068
- Alotaibi HM. Incidence, risk factors, and prognosis of hidradenitis suppurativa across the globe: insights from the literature. Clin Cosmet Investig Dermatol. 2023;16:545-552. doi:10.2147/CCID.S402453
- Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi:10.3389/fimmu.2018.02965
- Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi:10.1159/000507323
- Brown E, Cuthbertson DJ, Wilding JP. Newer GLP-1 receptor agonists and obesity-diabetes. Peptides. 2018;100:61-67. doi:10.1016/j.peptides.2017.12.009
- Cornell S. A review of GLP‐1 receptor agonists in type 2 diabetes: a focus on the mechanism of action of once‐weekly agents. J Clin Pharm Ther. 2020;45(suppl 1):17-27. doi:10.1111/jcpt.13230
- Lee YS, Jun HS. Anti-inflammatory effects of GLP-1-based therapies beyond glucose control. Mediators Inflamm. 2016;2016:3094642. doi:10.1155/2016/3094642
- Mintoff D, Benhadou F, Pace NP, et al. Metabolic syndrome and hidradenitis suppurativa: epidemiological, molecular, and therapeutic aspects. Int J Dermatol. 2022;61:1175-1186. doi:10.1111/ijd.15910
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi:10.1159/000473873
- Mainville L, MacHaalany J, Veillette H. Hidradenitis suppurativa patient requiring cardiac procedure with inguinal access: case management with ertapenem. SAGE Open Med Case Rep. 2024;12:2050313X241274819. doi:10.1177/2050313X241274819
- Hamed K, Alosaimi MN, Ali BA, et al. Glucagon-like peptide-1 (GLP-1) receptor agonists: exploring their impact on diabetes, obesity, and cardiovascular health through a comprehensive literature review. Cureus. 2024;16:E68390. doi:10.7759/cureus.68390
- Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367. doi:10.1177/20420188231222367
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
Practice Points
- Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) can be used adjunctively to manage hidradenitis suppurativa (HS) symptoms.
- The anti-inflammatory properties of GLP-1 RAs as well as their tendency to cause weight loss and manage metabolic syndrome improve the outcome of HS.
- Although current evidence is limited to case reports, these agents can be successfully integrated with existing protocols (biologics, antibiotics, or metformin); however, clinicians should monitor for gastrointestinal adverse effects.
Hypergammaglobulinemic Purpura of Waldenström With Primary and Autoimmune Associations
Hypergammaglobulinemic Purpura of Waldenström With Primary and Autoimmune Associations
Hypergammaglobulinemic purpura of Waldenström (HGPW) is a rare chronic skin condition characterized by recurrent petechiae and purpura on the lower legs, elevated erythrocyte sedimentation rate (ESR), polyclonal hypergammaglobulinemia, and elevated titers of IgG and IgA rheumatoid factor (RF).1,2 This condition can be a primary (idiopathic) syndrome or secondary to an autoimmune connective tissue disease. We report 2 cases of patients with episodic skin eruptions that were consistent with HGPW.
Patient 1
A 41-year-old woman presented to our clinic with a rash on the legs of 20 years’ duration. She had first been evaluated at an outside dermatology clinic 5 years prior, and a biopsy performed at the time led to a diagnosis of leukocytoclastic vasculitis (LCV). The rash affected her ability to work, as her job involved standing for prolonged periods of time. If she stood for more than 2 hours, she experienced leg pain and worsening of the rash. The rash also was exacerbated by nonsteroidal anti-inflammatory drugs but improved with multiple days of rest. She had been on dapsone 75 mg daily, but the dose was reduced to 50 mg daily after elevated liver enzymes were noted. This regimen had improved her rash for 4 years until she experienced breakthrough symptoms, leading to her re-evaluation. Prior outside therapies included systemic steroids with limited response, then oral dapsone.
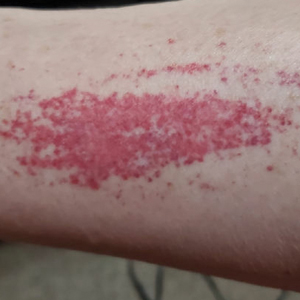
Upon our initial evaluation, laboratory tests were notable for an elevated ESR of 43 mm/h. Results of antinuclear antibody (ANA), anti–double-stranded DNA, extractable nuclear antigen, RF, HIV, cryoglobulin, hepatitis panel, serum protein electrophoresis, complete blood count, basic metabolic panel, urinalysis, and thyroid-stimulating hormone testing were within reference range. Physical examination revealed scattered pinpoint violaceous papules on the lower extremities. Photographs on the patient’s phone from 2 months prior showed a more robust manifestation with diffuse palpable purpura on the lower extremities.
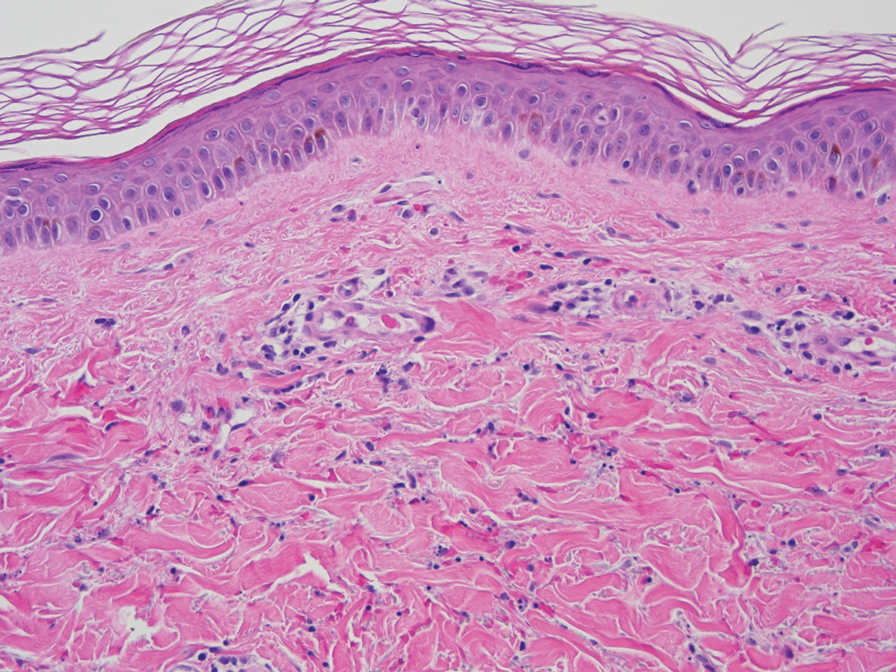
At 3-year follow-up, laboratory evaluation including ESR, IgA, IgG, IgM, serum protein electrophoresis with reflex immunofixation, and Mycoplasma pneumoniae IgM/IgG showed elevated ESR (29 mm/h) and IgG (1654 mg), with otherwise unremarkable results. Because of the extended period of time since the previous biopsy, a repeat biopsy with hematoxylin and eosin staining and direct immunofluorescence was performed. Biopsy from the left calf demonstrated a perivascular and interstitial infiltrate with lymphocytes and neutrophils with nuclear debris and hemorrhage (Figure 1). Direct immunofluorescence was positive for IgA, C3, and fibrin within vessel walls (Figure 2).
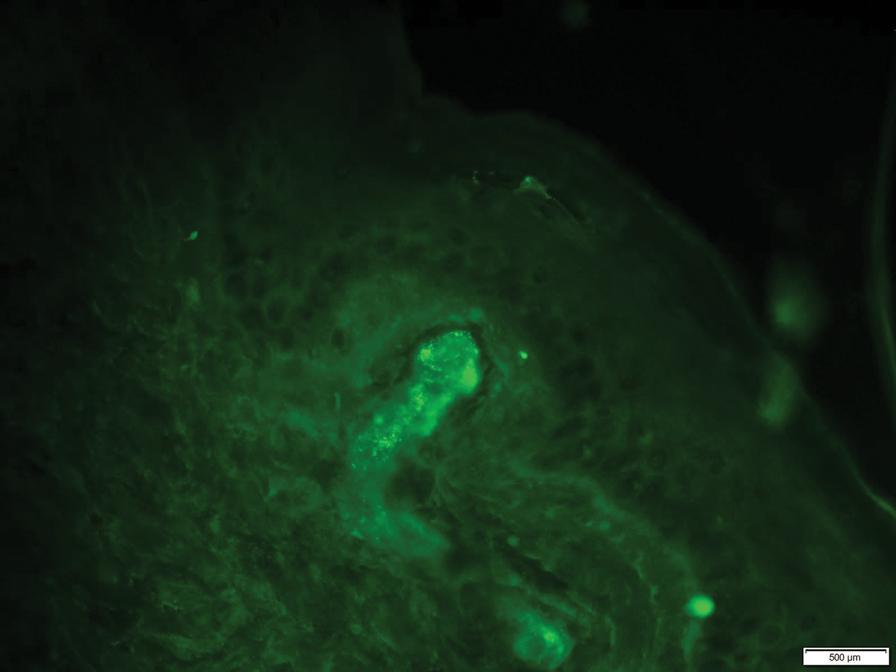
Overall the features of recurrent dependent palpable purpura and the pathology findings were consistent with evolving LCV. Given the chronic nature of her symptoms; flares with prolonged standing; presence of polyclonol hypergammaglobulinemia; and negative evaluation for underling autoimmune disease, infection, and malignancy, the clinicopathologic correlation was most consistent with primary HGPW. The patient was treated with colchicine 0.6 mg twice daily and continued on dapsone 50 mg daily. The colchicine was reduced to once daily due to diarrhea. Nonetheless, the patient had less frequent and less intense flares. On follow-up examination 4 months later, she was satisfied with her current level of control and did not wish to escalate her treatment.
Patient 2
A 53-year-old woman with a 1-year history of sicca symptoms presented for evaluation of a transient rash on the legs and feet of 2 months’ duration. At that time, the heels began to feel swollen. The rash was painful on the feet and caused calf myalgias. She did not endorse pruritus or pain elsewhere. The rash was not associated with prolonged standing, walking, or wearing tight socks. She had no fevers, chills, or joint pain. Flares would come and go within a week.
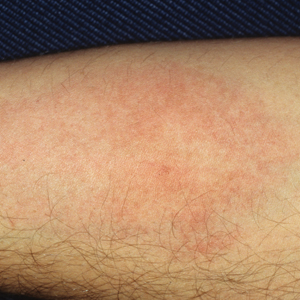
Laboratory evaluation was notable for an ANA of 1:1280 (reference range, 1:80) with positive anti-Ro/SS-A and anti-La/SS-B. Rheumatology evaluation confirmed the diagnosis of Sjögren syndrome. Physical examination revealed minimal petechiae on the heel of the left foot. Photographs from the previous month provided by the patient revealed linear petechiae of the lower extremities with postinflammatory hyperpigmentation (Figure 3). An additional photograph from the prior week revealed more diffuse erythematous plaques without secondary changes on the feet up to the ankles (Figure 4).
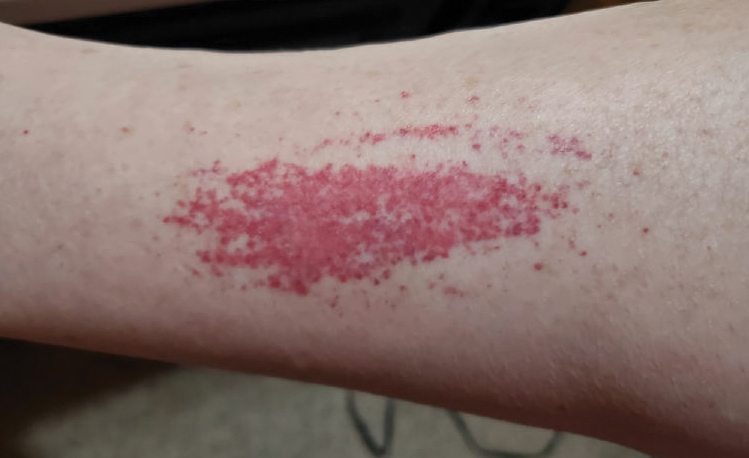
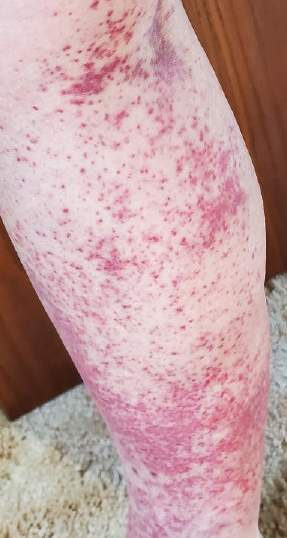
The patient experienced a recurrence of the rash within a month and had an expedited visit for biopsies, which demonstrated mixed inflammation with neutrophils, nuclear debris, hemorrhage, and C3 and fibrin immunoreactants within vessel walls. As with patient 1, the features were consistent with LCV.
In the context of Sjögren syndrome and elevated IgG and RF, the patient’s symptoms were consistent with secondary HGPW. Rheumatology prescribed hydroxychloroquine 400 mg daily alternating every other day with 300 mg and 0.6 mg of colchicine. The rash cleared within approximately 1 month.
Comment
Also known as benign hypergammaglobulinemic purpura, HGPW is a rare purpuric eruption that is exacerbated with prolonged standing and increased hydrostatic pressure.3 First described in 1943, HGPW is characterized by recurrent petechiae, purpuric macules, or palpable purpura, depending on the degree of inflammation.1,4,5 It typically is distributed on the bilateral lower extremities or trunk. Chronic postinflammatory hyperpigmentation with hemosiderin deposition also can be observed. The lesions last for up to 1 week at a time and are frequently asymmetrically distributed.2
Patient 1 demonstrated the typical clinical manifestations and laboratory findings of HGPW. The eruption often is asymptomatic, and patients report that the skin worsens with prolonged immobilization, walking, and wearing of tight clothing.2,6-8 Increased hydrostatic pressure is thought to cause the erythrocyte extravasation, resulting in the purpuric lesions. However, patient 2 was less typical, presenting with prominent skin pain and myalgias. Some patients experience discomfort, burning dysesthesia, pruritus, and swelling of the affected area.1 Hypergammaglobulinemic purpura of Waldenström is a chronic condition. Recurrent episodes can occur yearly or as frequently as multiple times per week.8
Women are most commonly diagnosed with HGPW, but many cases have been reported in children.9,10 In spite of the “condition being considered largely benign,” women with a diagnosis of HGPW require preconception counseling due to risks for congenital heart block, neonatal lupus, intrauterine growth restriction, intrauterine demise, and preterm birth.7,9,11,12
The etiology of the rash remains undefined. It is hypothesized that it develops due to underlying immune dysregulation with associated immune complex formation and deposition in the blood vessel wall.1 Small circulating immune complexes containing IgG or IgA RF are a specific finding in patients with HGPW. These highly soluble autoantibodies are hypothesized to influence the rapid appearance and disappearance of lesions.1
The role of hypergammaglobulinemia in the pathogenesis of HGPW is unknown.13 Serum IgG levels do not correlate with the appearance and regression of lesions.13 Additionally, hypergammaglobulinemia can be found in autoimmune connective tissue diseases such as Sjögren syndrome without resulting cutaneous vasculitis.13
Characteristic laboratory abnormalities include polyclonal hypergammaglobulinemia, elevated ESR, and elevated IgA and IgG RF. Positive ANA and anti-Ro/SS-A and anti-La/SS-B indicate a potential to develop autoimmune connective tissue diseases, including Sjögren syndrome, systemic lupus erythematosus, and rheumatoid arthritis.1,14 Additional recommended workup includes complete blood counts, metabolic panel, complement levels, urinalysis, and urine protein/creatinine ratio.9 Repeat monitoring for antibodies, inflammatory markers, immunoglobulins, and RF should be completed 3 months after initial evaluation. Patients with symptoms of systemic disease should have laboratory evaluation repeated.
Erythrocyte sedimentation rate abnormalities are a defining feature of HGPW. Erythrocyte sedimentation rate is an inexpensive and commonly ordered inflammatory marker that measures settling of erythrocytes within 1 hour and can be elevated by plasma proteins such as gamma globulins. Erythrocyte sedimentation rate is nonspecific and is not sensitive as a general screening test. It can be elevated by autoimmune connective tissue disease, infection, and malignancy.15 Notably, ESR is not specific to inflammation. Confounding factors include red blood cell abnormalities, physiologic factors, and the quantity of plasma proteins such as fibrinogen.16 These positively charged plasma proteins neutralize the negative surface charge of erythrocytes, resulting in erythrocytes that are prone to rouleaux formation.17
The utility of the ESR is to expedite the diagnostic process and indicate the need for further workup.16 Patients with mild to moderate elevation in ESR without an identified etiology should have repeat testing to confirm the validity of the laboratory value. Patients with an ESR higher than 100 mm/h are more likely have an infectious cause, collagen vascular disease, or underlying malignancy.15 Elevation of ESR in HGPW is likely a result of increased immunoglobulins and acute phase proteins.17
The histopathology of HGPW is nonspecific and may show LCV or erythrocyte extravasation with mild perivascular lymphocytic infiltrates.1,9 Direct immunofluorescence testing may show immune-complex deposition.5 For patients with evidence of LCV, the biopsy of a fresh but well-developed lesion is important in confirming the presence of vasculitis.1 Incorrect sampling may lead to underreporting of LCV with HGPW.3
Associated underlying conditions include Sjögren syndrome, systemic lupus erythematosus, rheumatoid arthritis, hepatitis C, and hematologic malignancies.1,3 Our patients demonstrated primary and secondary causes of HGPW. Patient 1’s case was not associated with any autoimmune disease but demonstrated chronic recurrence. Patient 2’s case was secondary to Sjögren syndrome.
In patients with suspected HGPW, differential diagnoses to consider include IgA vasculitis, cutaneous small vessel vasculitis, pigmented purpuric dermatoses, idiopathic thrombocytopenic purpura, thrombotic thrombocytopenic purpura, and scurvy.1,4
For patients with primary disease, treatment is focused on symptom management with compression stockings and avoidance of triggers. Compression stockings may exacerbate purpura but can provide symptom relief in some individuals.14 Patients with frequent or painful episodes can benefit from systemic treatment. In patients with an underlying disease, systemic therapies include prednisone, hydroxychloroquine, indomethacin, colchicine, chlorambucil, mycophenolate mofetil, rituximab, and plasmapheresis. Dapsone, a treatment for LCV, has been reported to be beneficial in patients with a neutrophilic infiltrate.18
Hypergammaglobulinemic purpura of Waldenström requires a thorough evaluation due to its association with underlying systemic disease. Patients without evidence of systemic disease should receive long-term monitoring and coordination of care with rheumatology, as systemic manifestations can develop years after the initial cutaneous manifestation. Dermatologists should consider HGPW in the differential diagnosis for cutaneous vasculitides.
- Piette WW. Purpura: mechanisms and differential diagnosis.In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. Elsevier Health Sciences; 2018:376-389.
- Finder KA, McCollough ML, Dixon SL, et al. Hypergammaglobulinemic purpura of Waldenström. J Am Acad Dermatol. 1990;23(4 Pt 1):669-676. doi:10.1016/0190-9622(90)70271-i
- Mathis J, Zirwas M, Elkins CT, et al. Persistent and progressive purpura in a patient with an elevated rheumatoid factor and polyclonal gammopathy (hypergammaglobulinemic purpura of Waldenström). J Am Acad Dermatol. 2015;72:374-376. doi:10.1016/j.jaad.2013.02.020
- 4. Alexandrescu DT, Levi M. The vascular purpuras. In: Kaushansky K, Prchal JT, Burns LJ, et al, eds. Williams Hematology. 10th ed. McGraw Hill; 2021:1-34.
- Lewin JM, Hunt R, Fischer M, et al. Hypergammaglobulinemic purpura of Waldenström. Dermatol Online J. 2012;18:2.
- Habib GS, Stimmer MM, Quismorio FP. Hypergammaglobulinemic purpura of Waldenstrom associated with systemic lupus erythematosus: report of a case and review of the literature. Lupus. 1995;4:19-22. doi:10.1177/096120339500400105
- Maeda-Tanaka M, Haruta S, Sado T, et al. Juvenile-onset hypergammaglobulinemic purpura and fetal congenital heart block.J Dermatol. 2006;33:714-718. doi:10.1111/j.1346-8138.2006.00166.x
- Malaviya AN, Kaushik P, Budhiraja S, et al. Hypergammaglobulinemic purpura of Waldenström: report of 3 cases with a short review. Clin Exp Rheumatol. 2000;18:518-522.
- Theisen E, Lee DE, Pei S, et al. Hypergammaglobulinemic purpura of Waldenström in children. Pediatr Dermatol. 2020;37:467-475. doi:10.1111/pde.14120
- Martini A, Ravelli A, Viola S, et al. Hypergammaglobulinemic purpura in childhood. Report of two cases and review of the literature. Helv Paediatr Acta. 1988;43:225-231.
- Jolly EC, Hunt BJ, Ellis S, et al. “Benign” hypergammaglobulinemic purpura is not benign in pregnancy. Clin Rheumatol. 2009;28(Suppl 1):S11-S15. doi:10.1007/s10067-008-1038-2
- Cheung VY, Bocking AD, Hollomby D, et al. Waldenström hypergammaglobulinemic purpura and pregnancy. Obstet Gynecol. 1993;82(4 Pt 2 Suppl):685-687.
- Kimura K, Miyabe C, Miyata R, et al. Hypergammaglobulinemic purpura: does hypergammaglobulinemia cause purpura? J Dermatol. 2021;48:e556-e557. doi:10.1111/1346-8138.16122
- Frankel A, Ingraffea A, Massé M, et al. Hypergammaglobulinemic purpura of Waldenström. Cutis. 2010;86:23-24.
- Brigden ML. Clinical utility of the erythrocyte sedimentation rate. Am Fam Physician. 1999;60:1443-1450.
- Solberg BL, Olson RJ. Clinical utility of the erythrocyte sedimentation rate: a case study. Clin Lab Sci. 2014;27:72-77.
- Tishkowski K, Gupta V. Erythrocyte sedimentation rate. In: StatPearls. StatPearls Publishing; May 9, 2021.
- Cheah J, Fields T. Hypergammaglobulinemic purpura of Waldenström. October 2018. Accessed November 14, 2021. https://www.hss.edu/files/HSS-Grand-Rounds-Complex-Cases-Vol7-Issue3.pdf
Hypergammaglobulinemic purpura of Waldenström (HGPW) is a rare chronic skin condition characterized by recurrent petechiae and purpura on the lower legs, elevated erythrocyte sedimentation rate (ESR), polyclonal hypergammaglobulinemia, and elevated titers of IgG and IgA rheumatoid factor (RF).1,2 This condition can be a primary (idiopathic) syndrome or secondary to an autoimmune connective tissue disease. We report 2 cases of patients with episodic skin eruptions that were consistent with HGPW.
Patient 1
A 41-year-old woman presented to our clinic with a rash on the legs of 20 years’ duration. She had first been evaluated at an outside dermatology clinic 5 years prior, and a biopsy performed at the time led to a diagnosis of leukocytoclastic vasculitis (LCV). The rash affected her ability to work, as her job involved standing for prolonged periods of time. If she stood for more than 2 hours, she experienced leg pain and worsening of the rash. The rash also was exacerbated by nonsteroidal anti-inflammatory drugs but improved with multiple days of rest. She had been on dapsone 75 mg daily, but the dose was reduced to 50 mg daily after elevated liver enzymes were noted. This regimen had improved her rash for 4 years until she experienced breakthrough symptoms, leading to her re-evaluation. Prior outside therapies included systemic steroids with limited response, then oral dapsone.
Upon our initial evaluation, laboratory tests were notable for an elevated ESR of 43 mm/h. Results of antinuclear antibody (ANA), anti–double-stranded DNA, extractable nuclear antigen, RF, HIV, cryoglobulin, hepatitis panel, serum protein electrophoresis, complete blood count, basic metabolic panel, urinalysis, and thyroid-stimulating hormone testing were within reference range. Physical examination revealed scattered pinpoint violaceous papules on the lower extremities. Photographs on the patient’s phone from 2 months prior showed a more robust manifestation with diffuse palpable purpura on the lower extremities.
At 3-year follow-up, laboratory evaluation including ESR, IgA, IgG, IgM, serum protein electrophoresis with reflex immunofixation, and Mycoplasma pneumoniae IgM/IgG showed elevated ESR (29 mm/h) and IgG (1654 mg), with otherwise unremarkable results. Because of the extended period of time since the previous biopsy, a repeat biopsy with hematoxylin and eosin staining and direct immunofluorescence was performed. Biopsy from the left calf demonstrated a perivascular and interstitial infiltrate with lymphocytes and neutrophils with nuclear debris and hemorrhage (Figure 1). Direct immunofluorescence was positive for IgA, C3, and fibrin within vessel walls (Figure 2).


Overall the features of recurrent dependent palpable purpura and the pathology findings were consistent with evolving LCV. Given the chronic nature of her symptoms; flares with prolonged standing; presence of polyclonol hypergammaglobulinemia; and negative evaluation for underling autoimmune disease, infection, and malignancy, the clinicopathologic correlation was most consistent with primary HGPW. The patient was treated with colchicine 0.6 mg twice daily and continued on dapsone 50 mg daily. The colchicine was reduced to once daily due to diarrhea. Nonetheless, the patient had less frequent and less intense flares. On follow-up examination 4 months later, she was satisfied with her current level of control and did not wish to escalate her treatment.
Patient 2
A 53-year-old woman with a 1-year history of sicca symptoms presented for evaluation of a transient rash on the legs and feet of 2 months’ duration. At that time, the heels began to feel swollen. The rash was painful on the feet and caused calf myalgias. She did not endorse pruritus or pain elsewhere. The rash was not associated with prolonged standing, walking, or wearing tight socks. She had no fevers, chills, or joint pain. Flares would come and go within a week.
Laboratory evaluation was notable for an ANA of 1:1280 (reference range, 1:80) with positive anti-Ro/SS-A and anti-La/SS-B. Rheumatology evaluation confirmed the diagnosis of Sjögren syndrome. Physical examination revealed minimal petechiae on the heel of the left foot. Photographs from the previous month provided by the patient revealed linear petechiae of the lower extremities with postinflammatory hyperpigmentation (Figure 3). An additional photograph from the prior week revealed more diffuse erythematous plaques without secondary changes on the feet up to the ankles (Figure 4).


The patient experienced a recurrence of the rash within a month and had an expedited visit for biopsies, which demonstrated mixed inflammation with neutrophils, nuclear debris, hemorrhage, and C3 and fibrin immunoreactants within vessel walls. As with patient 1, the features were consistent with LCV.
In the context of Sjögren syndrome and elevated IgG and RF, the patient’s symptoms were consistent with secondary HGPW. Rheumatology prescribed hydroxychloroquine 400 mg daily alternating every other day with 300 mg and 0.6 mg of colchicine. The rash cleared within approximately 1 month.
Comment
Also known as benign hypergammaglobulinemic purpura, HGPW is a rare purpuric eruption that is exacerbated with prolonged standing and increased hydrostatic pressure.3 First described in 1943, HGPW is characterized by recurrent petechiae, purpuric macules, or palpable purpura, depending on the degree of inflammation.1,4,5 It typically is distributed on the bilateral lower extremities or trunk. Chronic postinflammatory hyperpigmentation with hemosiderin deposition also can be observed. The lesions last for up to 1 week at a time and are frequently asymmetrically distributed.2
Patient 1 demonstrated the typical clinical manifestations and laboratory findings of HGPW. The eruption often is asymptomatic, and patients report that the skin worsens with prolonged immobilization, walking, and wearing of tight clothing.2,6-8 Increased hydrostatic pressure is thought to cause the erythrocyte extravasation, resulting in the purpuric lesions. However, patient 2 was less typical, presenting with prominent skin pain and myalgias. Some patients experience discomfort, burning dysesthesia, pruritus, and swelling of the affected area.1 Hypergammaglobulinemic purpura of Waldenström is a chronic condition. Recurrent episodes can occur yearly or as frequently as multiple times per week.8
Women are most commonly diagnosed with HGPW, but many cases have been reported in children.9,10 In spite of the “condition being considered largely benign,” women with a diagnosis of HGPW require preconception counseling due to risks for congenital heart block, neonatal lupus, intrauterine growth restriction, intrauterine demise, and preterm birth.7,9,11,12
The etiology of the rash remains undefined. It is hypothesized that it develops due to underlying immune dysregulation with associated immune complex formation and deposition in the blood vessel wall.1 Small circulating immune complexes containing IgG or IgA RF are a specific finding in patients with HGPW. These highly soluble autoantibodies are hypothesized to influence the rapid appearance and disappearance of lesions.1
The role of hypergammaglobulinemia in the pathogenesis of HGPW is unknown.13 Serum IgG levels do not correlate with the appearance and regression of lesions.13 Additionally, hypergammaglobulinemia can be found in autoimmune connective tissue diseases such as Sjögren syndrome without resulting cutaneous vasculitis.13
Characteristic laboratory abnormalities include polyclonal hypergammaglobulinemia, elevated ESR, and elevated IgA and IgG RF. Positive ANA and anti-Ro/SS-A and anti-La/SS-B indicate a potential to develop autoimmune connective tissue diseases, including Sjögren syndrome, systemic lupus erythematosus, and rheumatoid arthritis.1,14 Additional recommended workup includes complete blood counts, metabolic panel, complement levels, urinalysis, and urine protein/creatinine ratio.9 Repeat monitoring for antibodies, inflammatory markers, immunoglobulins, and RF should be completed 3 months after initial evaluation. Patients with symptoms of systemic disease should have laboratory evaluation repeated.
Erythrocyte sedimentation rate abnormalities are a defining feature of HGPW. Erythrocyte sedimentation rate is an inexpensive and commonly ordered inflammatory marker that measures settling of erythrocytes within 1 hour and can be elevated by plasma proteins such as gamma globulins. Erythrocyte sedimentation rate is nonspecific and is not sensitive as a general screening test. It can be elevated by autoimmune connective tissue disease, infection, and malignancy.15 Notably, ESR is not specific to inflammation. Confounding factors include red blood cell abnormalities, physiologic factors, and the quantity of plasma proteins such as fibrinogen.16 These positively charged plasma proteins neutralize the negative surface charge of erythrocytes, resulting in erythrocytes that are prone to rouleaux formation.17
The utility of the ESR is to expedite the diagnostic process and indicate the need for further workup.16 Patients with mild to moderate elevation in ESR without an identified etiology should have repeat testing to confirm the validity of the laboratory value. Patients with an ESR higher than 100 mm/h are more likely have an infectious cause, collagen vascular disease, or underlying malignancy.15 Elevation of ESR in HGPW is likely a result of increased immunoglobulins and acute phase proteins.17
The histopathology of HGPW is nonspecific and may show LCV or erythrocyte extravasation with mild perivascular lymphocytic infiltrates.1,9 Direct immunofluorescence testing may show immune-complex deposition.5 For patients with evidence of LCV, the biopsy of a fresh but well-developed lesion is important in confirming the presence of vasculitis.1 Incorrect sampling may lead to underreporting of LCV with HGPW.3
Associated underlying conditions include Sjögren syndrome, systemic lupus erythematosus, rheumatoid arthritis, hepatitis C, and hematologic malignancies.1,3 Our patients demonstrated primary and secondary causes of HGPW. Patient 1’s case was not associated with any autoimmune disease but demonstrated chronic recurrence. Patient 2’s case was secondary to Sjögren syndrome.
In patients with suspected HGPW, differential diagnoses to consider include IgA vasculitis, cutaneous small vessel vasculitis, pigmented purpuric dermatoses, idiopathic thrombocytopenic purpura, thrombotic thrombocytopenic purpura, and scurvy.1,4
For patients with primary disease, treatment is focused on symptom management with compression stockings and avoidance of triggers. Compression stockings may exacerbate purpura but can provide symptom relief in some individuals.14 Patients with frequent or painful episodes can benefit from systemic treatment. In patients with an underlying disease, systemic therapies include prednisone, hydroxychloroquine, indomethacin, colchicine, chlorambucil, mycophenolate mofetil, rituximab, and plasmapheresis. Dapsone, a treatment for LCV, has been reported to be beneficial in patients with a neutrophilic infiltrate.18
Hypergammaglobulinemic purpura of Waldenström requires a thorough evaluation due to its association with underlying systemic disease. Patients without evidence of systemic disease should receive long-term monitoring and coordination of care with rheumatology, as systemic manifestations can develop years after the initial cutaneous manifestation. Dermatologists should consider HGPW in the differential diagnosis for cutaneous vasculitides.
Hypergammaglobulinemic purpura of Waldenström (HGPW) is a rare chronic skin condition characterized by recurrent petechiae and purpura on the lower legs, elevated erythrocyte sedimentation rate (ESR), polyclonal hypergammaglobulinemia, and elevated titers of IgG and IgA rheumatoid factor (RF).1,2 This condition can be a primary (idiopathic) syndrome or secondary to an autoimmune connective tissue disease. We report 2 cases of patients with episodic skin eruptions that were consistent with HGPW.
Patient 1
A 41-year-old woman presented to our clinic with a rash on the legs of 20 years’ duration. She had first been evaluated at an outside dermatology clinic 5 years prior, and a biopsy performed at the time led to a diagnosis of leukocytoclastic vasculitis (LCV). The rash affected her ability to work, as her job involved standing for prolonged periods of time. If she stood for more than 2 hours, she experienced leg pain and worsening of the rash. The rash also was exacerbated by nonsteroidal anti-inflammatory drugs but improved with multiple days of rest. She had been on dapsone 75 mg daily, but the dose was reduced to 50 mg daily after elevated liver enzymes were noted. This regimen had improved her rash for 4 years until she experienced breakthrough symptoms, leading to her re-evaluation. Prior outside therapies included systemic steroids with limited response, then oral dapsone.
Upon our initial evaluation, laboratory tests were notable for an elevated ESR of 43 mm/h. Results of antinuclear antibody (ANA), anti–double-stranded DNA, extractable nuclear antigen, RF, HIV, cryoglobulin, hepatitis panel, serum protein electrophoresis, complete blood count, basic metabolic panel, urinalysis, and thyroid-stimulating hormone testing were within reference range. Physical examination revealed scattered pinpoint violaceous papules on the lower extremities. Photographs on the patient’s phone from 2 months prior showed a more robust manifestation with diffuse palpable purpura on the lower extremities.
At 3-year follow-up, laboratory evaluation including ESR, IgA, IgG, IgM, serum protein electrophoresis with reflex immunofixation, and Mycoplasma pneumoniae IgM/IgG showed elevated ESR (29 mm/h) and IgG (1654 mg), with otherwise unremarkable results. Because of the extended period of time since the previous biopsy, a repeat biopsy with hematoxylin and eosin staining and direct immunofluorescence was performed. Biopsy from the left calf demonstrated a perivascular and interstitial infiltrate with lymphocytes and neutrophils with nuclear debris and hemorrhage (Figure 1). Direct immunofluorescence was positive for IgA, C3, and fibrin within vessel walls (Figure 2).


Overall the features of recurrent dependent palpable purpura and the pathology findings were consistent with evolving LCV. Given the chronic nature of her symptoms; flares with prolonged standing; presence of polyclonol hypergammaglobulinemia; and negative evaluation for underling autoimmune disease, infection, and malignancy, the clinicopathologic correlation was most consistent with primary HGPW. The patient was treated with colchicine 0.6 mg twice daily and continued on dapsone 50 mg daily. The colchicine was reduced to once daily due to diarrhea. Nonetheless, the patient had less frequent and less intense flares. On follow-up examination 4 months later, she was satisfied with her current level of control and did not wish to escalate her treatment.
Patient 2
A 53-year-old woman with a 1-year history of sicca symptoms presented for evaluation of a transient rash on the legs and feet of 2 months’ duration. At that time, the heels began to feel swollen. The rash was painful on the feet and caused calf myalgias. She did not endorse pruritus or pain elsewhere. The rash was not associated with prolonged standing, walking, or wearing tight socks. She had no fevers, chills, or joint pain. Flares would come and go within a week.
Laboratory evaluation was notable for an ANA of 1:1280 (reference range, 1:80) with positive anti-Ro/SS-A and anti-La/SS-B. Rheumatology evaluation confirmed the diagnosis of Sjögren syndrome. Physical examination revealed minimal petechiae on the heel of the left foot. Photographs from the previous month provided by the patient revealed linear petechiae of the lower extremities with postinflammatory hyperpigmentation (Figure 3). An additional photograph from the prior week revealed more diffuse erythematous plaques without secondary changes on the feet up to the ankles (Figure 4).


The patient experienced a recurrence of the rash within a month and had an expedited visit for biopsies, which demonstrated mixed inflammation with neutrophils, nuclear debris, hemorrhage, and C3 and fibrin immunoreactants within vessel walls. As with patient 1, the features were consistent with LCV.
In the context of Sjögren syndrome and elevated IgG and RF, the patient’s symptoms were consistent with secondary HGPW. Rheumatology prescribed hydroxychloroquine 400 mg daily alternating every other day with 300 mg and 0.6 mg of colchicine. The rash cleared within approximately 1 month.
Comment
Also known as benign hypergammaglobulinemic purpura, HGPW is a rare purpuric eruption that is exacerbated with prolonged standing and increased hydrostatic pressure.3 First described in 1943, HGPW is characterized by recurrent petechiae, purpuric macules, or palpable purpura, depending on the degree of inflammation.1,4,5 It typically is distributed on the bilateral lower extremities or trunk. Chronic postinflammatory hyperpigmentation with hemosiderin deposition also can be observed. The lesions last for up to 1 week at a time and are frequently asymmetrically distributed.2
Patient 1 demonstrated the typical clinical manifestations and laboratory findings of HGPW. The eruption often is asymptomatic, and patients report that the skin worsens with prolonged immobilization, walking, and wearing of tight clothing.2,6-8 Increased hydrostatic pressure is thought to cause the erythrocyte extravasation, resulting in the purpuric lesions. However, patient 2 was less typical, presenting with prominent skin pain and myalgias. Some patients experience discomfort, burning dysesthesia, pruritus, and swelling of the affected area.1 Hypergammaglobulinemic purpura of Waldenström is a chronic condition. Recurrent episodes can occur yearly or as frequently as multiple times per week.8
Women are most commonly diagnosed with HGPW, but many cases have been reported in children.9,10 In spite of the “condition being considered largely benign,” women with a diagnosis of HGPW require preconception counseling due to risks for congenital heart block, neonatal lupus, intrauterine growth restriction, intrauterine demise, and preterm birth.7,9,11,12
The etiology of the rash remains undefined. It is hypothesized that it develops due to underlying immune dysregulation with associated immune complex formation and deposition in the blood vessel wall.1 Small circulating immune complexes containing IgG or IgA RF are a specific finding in patients with HGPW. These highly soluble autoantibodies are hypothesized to influence the rapid appearance and disappearance of lesions.1
The role of hypergammaglobulinemia in the pathogenesis of HGPW is unknown.13 Serum IgG levels do not correlate with the appearance and regression of lesions.13 Additionally, hypergammaglobulinemia can be found in autoimmune connective tissue diseases such as Sjögren syndrome without resulting cutaneous vasculitis.13
Characteristic laboratory abnormalities include polyclonal hypergammaglobulinemia, elevated ESR, and elevated IgA and IgG RF. Positive ANA and anti-Ro/SS-A and anti-La/SS-B indicate a potential to develop autoimmune connective tissue diseases, including Sjögren syndrome, systemic lupus erythematosus, and rheumatoid arthritis.1,14 Additional recommended workup includes complete blood counts, metabolic panel, complement levels, urinalysis, and urine protein/creatinine ratio.9 Repeat monitoring for antibodies, inflammatory markers, immunoglobulins, and RF should be completed 3 months after initial evaluation. Patients with symptoms of systemic disease should have laboratory evaluation repeated.
Erythrocyte sedimentation rate abnormalities are a defining feature of HGPW. Erythrocyte sedimentation rate is an inexpensive and commonly ordered inflammatory marker that measures settling of erythrocytes within 1 hour and can be elevated by plasma proteins such as gamma globulins. Erythrocyte sedimentation rate is nonspecific and is not sensitive as a general screening test. It can be elevated by autoimmune connective tissue disease, infection, and malignancy.15 Notably, ESR is not specific to inflammation. Confounding factors include red blood cell abnormalities, physiologic factors, and the quantity of plasma proteins such as fibrinogen.16 These positively charged plasma proteins neutralize the negative surface charge of erythrocytes, resulting in erythrocytes that are prone to rouleaux formation.17
The utility of the ESR is to expedite the diagnostic process and indicate the need for further workup.16 Patients with mild to moderate elevation in ESR without an identified etiology should have repeat testing to confirm the validity of the laboratory value. Patients with an ESR higher than 100 mm/h are more likely have an infectious cause, collagen vascular disease, or underlying malignancy.15 Elevation of ESR in HGPW is likely a result of increased immunoglobulins and acute phase proteins.17
The histopathology of HGPW is nonspecific and may show LCV or erythrocyte extravasation with mild perivascular lymphocytic infiltrates.1,9 Direct immunofluorescence testing may show immune-complex deposition.5 For patients with evidence of LCV, the biopsy of a fresh but well-developed lesion is important in confirming the presence of vasculitis.1 Incorrect sampling may lead to underreporting of LCV with HGPW.3
Associated underlying conditions include Sjögren syndrome, systemic lupus erythematosus, rheumatoid arthritis, hepatitis C, and hematologic malignancies.1,3 Our patients demonstrated primary and secondary causes of HGPW. Patient 1’s case was not associated with any autoimmune disease but demonstrated chronic recurrence. Patient 2’s case was secondary to Sjögren syndrome.
In patients with suspected HGPW, differential diagnoses to consider include IgA vasculitis, cutaneous small vessel vasculitis, pigmented purpuric dermatoses, idiopathic thrombocytopenic purpura, thrombotic thrombocytopenic purpura, and scurvy.1,4
For patients with primary disease, treatment is focused on symptom management with compression stockings and avoidance of triggers. Compression stockings may exacerbate purpura but can provide symptom relief in some individuals.14 Patients with frequent or painful episodes can benefit from systemic treatment. In patients with an underlying disease, systemic therapies include prednisone, hydroxychloroquine, indomethacin, colchicine, chlorambucil, mycophenolate mofetil, rituximab, and plasmapheresis. Dapsone, a treatment for LCV, has been reported to be beneficial in patients with a neutrophilic infiltrate.18
Hypergammaglobulinemic purpura of Waldenström requires a thorough evaluation due to its association with underlying systemic disease. Patients without evidence of systemic disease should receive long-term monitoring and coordination of care with rheumatology, as systemic manifestations can develop years after the initial cutaneous manifestation. Dermatologists should consider HGPW in the differential diagnosis for cutaneous vasculitides.
- Piette WW. Purpura: mechanisms and differential diagnosis.In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. Elsevier Health Sciences; 2018:376-389.
- Finder KA, McCollough ML, Dixon SL, et al. Hypergammaglobulinemic purpura of Waldenström. J Am Acad Dermatol. 1990;23(4 Pt 1):669-676. doi:10.1016/0190-9622(90)70271-i
- Mathis J, Zirwas M, Elkins CT, et al. Persistent and progressive purpura in a patient with an elevated rheumatoid factor and polyclonal gammopathy (hypergammaglobulinemic purpura of Waldenström). J Am Acad Dermatol. 2015;72:374-376. doi:10.1016/j.jaad.2013.02.020
- 4. Alexandrescu DT, Levi M. The vascular purpuras. In: Kaushansky K, Prchal JT, Burns LJ, et al, eds. Williams Hematology. 10th ed. McGraw Hill; 2021:1-34.
- Lewin JM, Hunt R, Fischer M, et al. Hypergammaglobulinemic purpura of Waldenström. Dermatol Online J. 2012;18:2.
- Habib GS, Stimmer MM, Quismorio FP. Hypergammaglobulinemic purpura of Waldenstrom associated with systemic lupus erythematosus: report of a case and review of the literature. Lupus. 1995;4:19-22. doi:10.1177/096120339500400105
- Maeda-Tanaka M, Haruta S, Sado T, et al. Juvenile-onset hypergammaglobulinemic purpura and fetal congenital heart block.J Dermatol. 2006;33:714-718. doi:10.1111/j.1346-8138.2006.00166.x
- Malaviya AN, Kaushik P, Budhiraja S, et al. Hypergammaglobulinemic purpura of Waldenström: report of 3 cases with a short review. Clin Exp Rheumatol. 2000;18:518-522.
- Theisen E, Lee DE, Pei S, et al. Hypergammaglobulinemic purpura of Waldenström in children. Pediatr Dermatol. 2020;37:467-475. doi:10.1111/pde.14120
- Martini A, Ravelli A, Viola S, et al. Hypergammaglobulinemic purpura in childhood. Report of two cases and review of the literature. Helv Paediatr Acta. 1988;43:225-231.
- Jolly EC, Hunt BJ, Ellis S, et al. “Benign” hypergammaglobulinemic purpura is not benign in pregnancy. Clin Rheumatol. 2009;28(Suppl 1):S11-S15. doi:10.1007/s10067-008-1038-2
- Cheung VY, Bocking AD, Hollomby D, et al. Waldenström hypergammaglobulinemic purpura and pregnancy. Obstet Gynecol. 1993;82(4 Pt 2 Suppl):685-687.
- Kimura K, Miyabe C, Miyata R, et al. Hypergammaglobulinemic purpura: does hypergammaglobulinemia cause purpura? J Dermatol. 2021;48:e556-e557. doi:10.1111/1346-8138.16122
- Frankel A, Ingraffea A, Massé M, et al. Hypergammaglobulinemic purpura of Waldenström. Cutis. 2010;86:23-24.
- Brigden ML. Clinical utility of the erythrocyte sedimentation rate. Am Fam Physician. 1999;60:1443-1450.
- Solberg BL, Olson RJ. Clinical utility of the erythrocyte sedimentation rate: a case study. Clin Lab Sci. 2014;27:72-77.
- Tishkowski K, Gupta V. Erythrocyte sedimentation rate. In: StatPearls. StatPearls Publishing; May 9, 2021.
- Cheah J, Fields T. Hypergammaglobulinemic purpura of Waldenström. October 2018. Accessed November 14, 2021. https://www.hss.edu/files/HSS-Grand-Rounds-Complex-Cases-Vol7-Issue3.pdf
- Piette WW. Purpura: mechanisms and differential diagnosis.In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. Elsevier Health Sciences; 2018:376-389.
- Finder KA, McCollough ML, Dixon SL, et al. Hypergammaglobulinemic purpura of Waldenström. J Am Acad Dermatol. 1990;23(4 Pt 1):669-676. doi:10.1016/0190-9622(90)70271-i
- Mathis J, Zirwas M, Elkins CT, et al. Persistent and progressive purpura in a patient with an elevated rheumatoid factor and polyclonal gammopathy (hypergammaglobulinemic purpura of Waldenström). J Am Acad Dermatol. 2015;72:374-376. doi:10.1016/j.jaad.2013.02.020
- 4. Alexandrescu DT, Levi M. The vascular purpuras. In: Kaushansky K, Prchal JT, Burns LJ, et al, eds. Williams Hematology. 10th ed. McGraw Hill; 2021:1-34.
- Lewin JM, Hunt R, Fischer M, et al. Hypergammaglobulinemic purpura of Waldenström. Dermatol Online J. 2012;18:2.
- Habib GS, Stimmer MM, Quismorio FP. Hypergammaglobulinemic purpura of Waldenstrom associated with systemic lupus erythematosus: report of a case and review of the literature. Lupus. 1995;4:19-22. doi:10.1177/096120339500400105
- Maeda-Tanaka M, Haruta S, Sado T, et al. Juvenile-onset hypergammaglobulinemic purpura and fetal congenital heart block.J Dermatol. 2006;33:714-718. doi:10.1111/j.1346-8138.2006.00166.x
- Malaviya AN, Kaushik P, Budhiraja S, et al. Hypergammaglobulinemic purpura of Waldenström: report of 3 cases with a short review. Clin Exp Rheumatol. 2000;18:518-522.
- Theisen E, Lee DE, Pei S, et al. Hypergammaglobulinemic purpura of Waldenström in children. Pediatr Dermatol. 2020;37:467-475. doi:10.1111/pde.14120
- Martini A, Ravelli A, Viola S, et al. Hypergammaglobulinemic purpura in childhood. Report of two cases and review of the literature. Helv Paediatr Acta. 1988;43:225-231.
- Jolly EC, Hunt BJ, Ellis S, et al. “Benign” hypergammaglobulinemic purpura is not benign in pregnancy. Clin Rheumatol. 2009;28(Suppl 1):S11-S15. doi:10.1007/s10067-008-1038-2
- Cheung VY, Bocking AD, Hollomby D, et al. Waldenström hypergammaglobulinemic purpura and pregnancy. Obstet Gynecol. 1993;82(4 Pt 2 Suppl):685-687.
- Kimura K, Miyabe C, Miyata R, et al. Hypergammaglobulinemic purpura: does hypergammaglobulinemia cause purpura? J Dermatol. 2021;48:e556-e557. doi:10.1111/1346-8138.16122
- Frankel A, Ingraffea A, Massé M, et al. Hypergammaglobulinemic purpura of Waldenström. Cutis. 2010;86:23-24.
- Brigden ML. Clinical utility of the erythrocyte sedimentation rate. Am Fam Physician. 1999;60:1443-1450.
- Solberg BL, Olson RJ. Clinical utility of the erythrocyte sedimentation rate: a case study. Clin Lab Sci. 2014;27:72-77.
- Tishkowski K, Gupta V. Erythrocyte sedimentation rate. In: StatPearls. StatPearls Publishing; May 9, 2021.
- Cheah J, Fields T. Hypergammaglobulinemic purpura of Waldenström. October 2018. Accessed November 14, 2021. https://www.hss.edu/files/HSS-Grand-Rounds-Complex-Cases-Vol7-Issue3.pdf
Hypergammaglobulinemic Purpura of Waldenström With Primary and Autoimmune Associations
Hypergammaglobulinemic Purpura of Waldenström With Primary and Autoimmune Associations
Practice Points
- Elevation of the erythrocyte sedimentation rate (ESR) is nonspecific for inflammation and may be observed in the setting of increased immunoglobulin levels.
- Patients with elevated ESR and clinical evidence of recurrent petechiae and purpura should be screened for monoclonal and polyclonal gammopathies.
A Solitary Axillary Subcutaneous Mass
A Solitary Axillary Subcutaneous Mass
THE DIAGNOSIS: Cutaneous Rosai-Dorfman Disease
The clinical differential diagnosis in our patient included a broad array of soft-tissue neoplasms ranging from benign entities to sarcomas. Histology was notable for a dense, dermal-based, lymphohistiocytic infiltrate with alternating hypocellular and hypercellular areas imparting a marbled appearance on low-power view (Figure, A). Further immunohistochemical staining revealed large, S100-positive histiocytes containing intact inflammatory cells (emperipolesis), which confirmed a diagnosis of cutaneous Rosai-Dorfman disease (RDD)(Figure, B). Our patient elected to undergo surgical removal of the mass, and he will be monitored for recurrence.
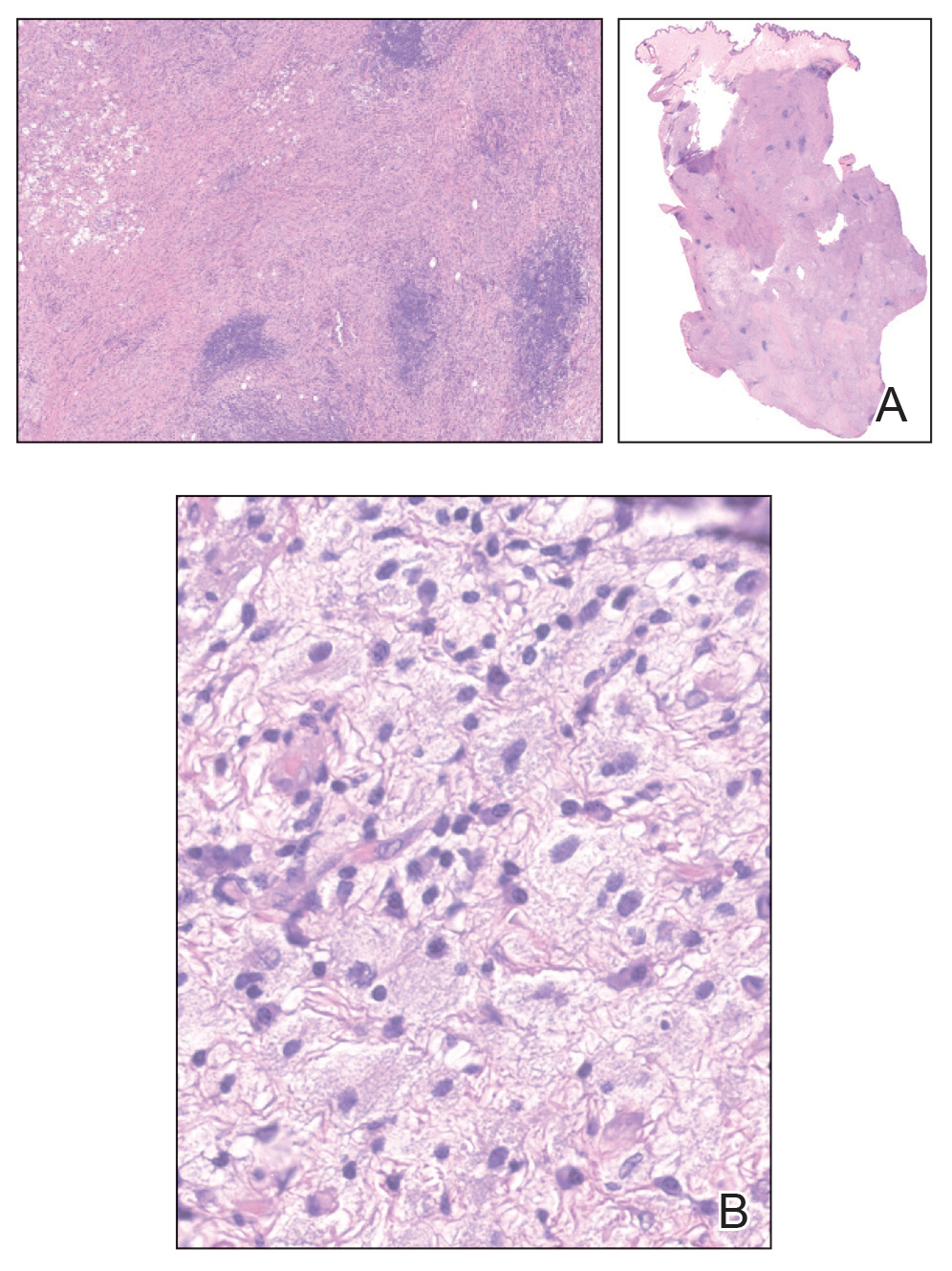
Rosai-Dorfman disease is a non–Langerhans cell histiocytosis that most commonly affects the lymph nodes but can affect other organs including the skin. Rosai-Dorfman disease initially was documented in the medical literature in 1969 by Rosai and Dorfman1 as benign sinus histiocytosis with massive lymphadenopathy. Classic RDD usually manifests with painless cervical lymphadenopathy in children or young adults along with fever, leukocytosis, anemia, polyclonal hypergammaglobulinemia, and elevated inflammatory markers.2,3 Extranodal involvement has been reported in up to 43% of cases, with common sites including the skin, central nervous system, and gastrointestinal tract.3,4
Cutaneous RDD is a distinct, less common clinical entity that is limited to the skin and shows no nodal involvement or systemic symptoms such as fever, night sweats, or weight loss.5 Cutaneous RDD classically manifests with localized indurated papules and plaques, but it can manifest with tumorlike lesions in the subcutaneous tissues.6 Cutaneous RDD is very rare, with fewer than 200 known case reports in the literature as of 2014; in comparison to classic forms of RDD, cutaneous RDD has a female predominance.7,8 There are few reports of isolated cutaneous disease manifesting as soft-tissue masses, and our case represents a rare case of cutaneous RDD manifesting as a solitary soft-tissue mass in the axilla.9-11 Diagnosis of cutaneous RDD is challenging due to its variable clinical manifestations and nonspecific imaging findings, requiring clinicopathologic correlation.
Imaging of subcutaneous RDD lesions typically shows well-defined, irregularly shaped masses with homogenous enhancement on computed tomography/ magnetic resonance imaging. Additional imaging with positron emission tomography/computed tomography is recommended to examine for organ involvement, as RDD lesions have avid uptake.12,13 Imaging may help differentiate RDD lesions from malignant neoplasms prior to biopsy. Additional workup includes baseline laboratory testing with inflammatory markers and a complete blood count for evaluation of laboratory abnormalities seen in classic RDD, including leukocytosis, anemia, or systemic inflammation.12 Following imaging and laboratory testing, definitive diagnosis of RDD necessitates histopathologic examination.
Although cutaneous RDD is clinically distinct from its classic RDD counterpart, the conditions share the same characteristic histologic features.5 Histology is notable for a dense mixed inflammatory infiltrate comprised of large pale histiocytes exhibiting emperipolesis, lymphocytes, plasma cells, and occasional eosinophils and neutrophils. Histiocytes stain positive for CD68, CD163, and S100 and are negative for Langerhans cell markers CD1a and CD207.6
The etiology of RDD remains poorly understood. Classic RDD has been associated with both sporadic and familial forms, with somatic mutations identified in the mitogen-activated protein kinase/KRAS pathway in up to one-third of cases, and less frequently in the BRAF gene.14,15 Germline mutations in familial cases of RDD have been identified in the SLC29A3 gene; mutations in this gene are associated with a spectrum of syndromes with histiocytosis and lymphadenopathy.14,15 In contrast, molecular drivers have yet to be identified in cutaneous RDD lesions, and the current predominant hypothesis is that cutaneous RDD has a reactive or immunologic pathophysiology. Autoimmune diseases, infections, and lymphomas have been reported to co-occur with both classic and cutaneous RDD.15 While subclinical viral infections such as Epstein-Barr virus and human herpesvirus 6 have been identified in RDD cases, studies have failed to prove their role as pathogenic drivers of the disease.14,16,17 Commonly reported comorbidities include systemic lupus erythematous, diabetes, hemolytic anemia, acute/chronic uveitis (though it is controversial whether these cases represent orbital involvement in systemic RDD), and Crohn disease.7,8,18,19 Immunohistochemical findings have supported that cells within RDD are activated monocytes responding to T-cell cytokine signaling following an infectious or immunologic insult.20,21
Consensus guidelines on treatment for cutaneous RDD recommend either observation for asymptomatic disease or surgical excision for unifocal lesions with consideration of systemic therapy for refractory cutaneous disease.22,23 Most patients with cutaneous RDD have self-limited disease, but long-term follow-up is recommended following surgical excision to monitor for recurrence, especially if there is a residual positive margin.24 Radiation therapy also may have to be utilized for residual or recurrent disease that becomes symptomatic; however, further studies are needed to determine its efficacy in limiting recurrence.4,12,25 Systemic treatment options include immunosuppressive or immunomodulatory agents such as corticosteroids, methotrexate, and rituximab.5 There currently are no guidelines on length of follow-up, but surveillance is recommended initially at 4 months, followed by 6- to 12-month intervals.22
- Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy. a newly recognized benign clinicopathological entity. Arch Pathol. 1969;87:63-70.
- Foucar E, Rosai J, Dorfman R. Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease): review of the entity. Semin Diagn Pathol. 1990;7:19-73.
- Stefanato CM, Ellerin PS, Bhawan J. Cutaneous sinus histiocytosis (Rosai-Dorfman disease) presenting clinically as vasculitis. J Am Acad Dermatol. 2002;46:775-778.
- Dalia S, Sagatys E, Sokol L, et al. Rosai-Dorfman Disease: tumor biology, clinical features, pathology, and treatment. Cancer Control. 2014;21:322-327.
- Bruce-Brand C, Schneider JW, Schubert P. Rosai-Dorfman disease: an overview. J Clin Pathol. 2020;73:697.
- Bolognia J, Jorizzo J, Schaffer J. Dermatology. 3rd ed. ed. Elsevier Saunders 2012.
- Salva KA, Stenstrom M, Breadon JY, et al. Possible association of cutaneous rosai-dorfman disease and chronic crohn disease: a case series report. JAMA Dermatol. 2014;150:177-181.
- Brenn T, Calonje E, Granter SR, et al. Cutaneous Rosai-Dorfman disease is a distinct clinical entity. Am J Dermatopathol. 2002; 24:385-391.
- Betini N, Munger AM, Rottmann D, et al. Rare presentation of Rosai- Dorfman disease in soft tissue: diagnostic findings and surgical treatment. Case Rep Surg. 2022;2022:8440836.
- Cravero JC, Ibrahim S. Recurrent soft tissue rosai dorfman disease of right medial thigh lipoma with lymph node involvement. Fed Pract. 2024;41(suppl 2):S20-S23
- Tenny SO, McGinness M, Zhang D, et al. Rosai-Dorfman disease presenting as a breast mass and enlarged axillary lymph node mimicking malignancy: a case report and review of the literature. Breast J. 2011;17:516-520.
- Goyal G, Ravindran A, Young JR, et al. Clinicopathological features, treatment approaches, and outcomes in Rosai-Dorfman disease. Haematologica. 2020;105:348-357.
- Li H, Li D, Xia J, et al. Radiological features of Rosai-Dorfman disease: case series and review of the literature. Clin Radiol. 2022;77:E799-E805.
- Elbaz Younes I, Sokol L, Zhang L. Rosai-Dorfman disease between proliferation and neoplasia. Cancers. 2022;14:5271.
- Ravindran A, Rech KL. How I diagnose Rosai-Dorfman disease. Am J Clin Pathol. 2023;160:1-10.
- Kutlubay Z, Bairamov O, Sevim A, et al. Rosai-Dorfman disease: a case report with nodal and cutaneous involvement and review of the literature. Am J Dermatopathol. 2014;36:353-357.
- Luppi M, Barozzi P, Garber R, et al. Expression of human herpesvirus 6 antigens in benign and malignant lymphoproliferative diseases. Am J Pathol. 1998;153:815-823.
- Wang KH, Chen WY, Liu HN, et al. Cutaneous Rosai-Dorfman disease: clinicopathological profiles, spectrum and evolution of 21 lesions in six patients. Br J Dermatol. 2006;154:277-286.
- Vaiselbuh SR, Bryceson YT, Allen CE, et al. Updates on histiocytic disorders. Pediatr Blood Cancer. 2014;61:1329-1335.
- Ravindran A, Goyal G, Go RS, et al. Rosai-Dorfman disease displays a unique monocyte-macrophage phenotype characterized by expression of OCT2. Am J Surg Pathol. 2021;45:35-44.
- Hoogewerf CJ, van Baar ME, Middelkoop E, et al. Impact of facial burns: relationship between depressive symptoms, self-esteem and scar severity. Gen Hosp Psychiatry. 2014;36:271-276.
- Abla O, Jacobsen E, Picarsic J, et al. Consensus recommendations for the diagnosis and clinical management of Rosai-Dorfman-Destombes disease. Blood. 2018;131:2877-2890.
- Al-Khateeb THH. Cutaneous Rosai-Dorfman disease of the face: a comprehensive literature review and case report. J Oral Maxillofacial Surg. 2016;74:528-540.
- Cheng SP, Jeng KS, Liu CL. Subcutaneous Rosai–Dorfman disease: is surgical excision justified? J Eur Acad Dermatol Venereol. 2005; 19:747-750.
- Garcia RA, DiCarlo EF. Rosai-Dorfman disease of bone and soft tissue. Arch Pathol Lab Med. 2021;146:40-46.
THE DIAGNOSIS: Cutaneous Rosai-Dorfman Disease
The clinical differential diagnosis in our patient included a broad array of soft-tissue neoplasms ranging from benign entities to sarcomas. Histology was notable for a dense, dermal-based, lymphohistiocytic infiltrate with alternating hypocellular and hypercellular areas imparting a marbled appearance on low-power view (Figure, A). Further immunohistochemical staining revealed large, S100-positive histiocytes containing intact inflammatory cells (emperipolesis), which confirmed a diagnosis of cutaneous Rosai-Dorfman disease (RDD)(Figure, B). Our patient elected to undergo surgical removal of the mass, and he will be monitored for recurrence.

Rosai-Dorfman disease is a non–Langerhans cell histiocytosis that most commonly affects the lymph nodes but can affect other organs including the skin. Rosai-Dorfman disease initially was documented in the medical literature in 1969 by Rosai and Dorfman1 as benign sinus histiocytosis with massive lymphadenopathy. Classic RDD usually manifests with painless cervical lymphadenopathy in children or young adults along with fever, leukocytosis, anemia, polyclonal hypergammaglobulinemia, and elevated inflammatory markers.2,3 Extranodal involvement has been reported in up to 43% of cases, with common sites including the skin, central nervous system, and gastrointestinal tract.3,4
Cutaneous RDD is a distinct, less common clinical entity that is limited to the skin and shows no nodal involvement or systemic symptoms such as fever, night sweats, or weight loss.5 Cutaneous RDD classically manifests with localized indurated papules and plaques, but it can manifest with tumorlike lesions in the subcutaneous tissues.6 Cutaneous RDD is very rare, with fewer than 200 known case reports in the literature as of 2014; in comparison to classic forms of RDD, cutaneous RDD has a female predominance.7,8 There are few reports of isolated cutaneous disease manifesting as soft-tissue masses, and our case represents a rare case of cutaneous RDD manifesting as a solitary soft-tissue mass in the axilla.9-11 Diagnosis of cutaneous RDD is challenging due to its variable clinical manifestations and nonspecific imaging findings, requiring clinicopathologic correlation.
Imaging of subcutaneous RDD lesions typically shows well-defined, irregularly shaped masses with homogenous enhancement on computed tomography/ magnetic resonance imaging. Additional imaging with positron emission tomography/computed tomography is recommended to examine for organ involvement, as RDD lesions have avid uptake.12,13 Imaging may help differentiate RDD lesions from malignant neoplasms prior to biopsy. Additional workup includes baseline laboratory testing with inflammatory markers and a complete blood count for evaluation of laboratory abnormalities seen in classic RDD, including leukocytosis, anemia, or systemic inflammation.12 Following imaging and laboratory testing, definitive diagnosis of RDD necessitates histopathologic examination.
Although cutaneous RDD is clinically distinct from its classic RDD counterpart, the conditions share the same characteristic histologic features.5 Histology is notable for a dense mixed inflammatory infiltrate comprised of large pale histiocytes exhibiting emperipolesis, lymphocytes, plasma cells, and occasional eosinophils and neutrophils. Histiocytes stain positive for CD68, CD163, and S100 and are negative for Langerhans cell markers CD1a and CD207.6
The etiology of RDD remains poorly understood. Classic RDD has been associated with both sporadic and familial forms, with somatic mutations identified in the mitogen-activated protein kinase/KRAS pathway in up to one-third of cases, and less frequently in the BRAF gene.14,15 Germline mutations in familial cases of RDD have been identified in the SLC29A3 gene; mutations in this gene are associated with a spectrum of syndromes with histiocytosis and lymphadenopathy.14,15 In contrast, molecular drivers have yet to be identified in cutaneous RDD lesions, and the current predominant hypothesis is that cutaneous RDD has a reactive or immunologic pathophysiology. Autoimmune diseases, infections, and lymphomas have been reported to co-occur with both classic and cutaneous RDD.15 While subclinical viral infections such as Epstein-Barr virus and human herpesvirus 6 have been identified in RDD cases, studies have failed to prove their role as pathogenic drivers of the disease.14,16,17 Commonly reported comorbidities include systemic lupus erythematous, diabetes, hemolytic anemia, acute/chronic uveitis (though it is controversial whether these cases represent orbital involvement in systemic RDD), and Crohn disease.7,8,18,19 Immunohistochemical findings have supported that cells within RDD are activated monocytes responding to T-cell cytokine signaling following an infectious or immunologic insult.20,21
Consensus guidelines on treatment for cutaneous RDD recommend either observation for asymptomatic disease or surgical excision for unifocal lesions with consideration of systemic therapy for refractory cutaneous disease.22,23 Most patients with cutaneous RDD have self-limited disease, but long-term follow-up is recommended following surgical excision to monitor for recurrence, especially if there is a residual positive margin.24 Radiation therapy also may have to be utilized for residual or recurrent disease that becomes symptomatic; however, further studies are needed to determine its efficacy in limiting recurrence.4,12,25 Systemic treatment options include immunosuppressive or immunomodulatory agents such as corticosteroids, methotrexate, and rituximab.5 There currently are no guidelines on length of follow-up, but surveillance is recommended initially at 4 months, followed by 6- to 12-month intervals.22
THE DIAGNOSIS: Cutaneous Rosai-Dorfman Disease
The clinical differential diagnosis in our patient included a broad array of soft-tissue neoplasms ranging from benign entities to sarcomas. Histology was notable for a dense, dermal-based, lymphohistiocytic infiltrate with alternating hypocellular and hypercellular areas imparting a marbled appearance on low-power view (Figure, A). Further immunohistochemical staining revealed large, S100-positive histiocytes containing intact inflammatory cells (emperipolesis), which confirmed a diagnosis of cutaneous Rosai-Dorfman disease (RDD)(Figure, B). Our patient elected to undergo surgical removal of the mass, and he will be monitored for recurrence.

Rosai-Dorfman disease is a non–Langerhans cell histiocytosis that most commonly affects the lymph nodes but can affect other organs including the skin. Rosai-Dorfman disease initially was documented in the medical literature in 1969 by Rosai and Dorfman1 as benign sinus histiocytosis with massive lymphadenopathy. Classic RDD usually manifests with painless cervical lymphadenopathy in children or young adults along with fever, leukocytosis, anemia, polyclonal hypergammaglobulinemia, and elevated inflammatory markers.2,3 Extranodal involvement has been reported in up to 43% of cases, with common sites including the skin, central nervous system, and gastrointestinal tract.3,4
Cutaneous RDD is a distinct, less common clinical entity that is limited to the skin and shows no nodal involvement or systemic symptoms such as fever, night sweats, or weight loss.5 Cutaneous RDD classically manifests with localized indurated papules and plaques, but it can manifest with tumorlike lesions in the subcutaneous tissues.6 Cutaneous RDD is very rare, with fewer than 200 known case reports in the literature as of 2014; in comparison to classic forms of RDD, cutaneous RDD has a female predominance.7,8 There are few reports of isolated cutaneous disease manifesting as soft-tissue masses, and our case represents a rare case of cutaneous RDD manifesting as a solitary soft-tissue mass in the axilla.9-11 Diagnosis of cutaneous RDD is challenging due to its variable clinical manifestations and nonspecific imaging findings, requiring clinicopathologic correlation.
Imaging of subcutaneous RDD lesions typically shows well-defined, irregularly shaped masses with homogenous enhancement on computed tomography/ magnetic resonance imaging. Additional imaging with positron emission tomography/computed tomography is recommended to examine for organ involvement, as RDD lesions have avid uptake.12,13 Imaging may help differentiate RDD lesions from malignant neoplasms prior to biopsy. Additional workup includes baseline laboratory testing with inflammatory markers and a complete blood count for evaluation of laboratory abnormalities seen in classic RDD, including leukocytosis, anemia, or systemic inflammation.12 Following imaging and laboratory testing, definitive diagnosis of RDD necessitates histopathologic examination.
Although cutaneous RDD is clinically distinct from its classic RDD counterpart, the conditions share the same characteristic histologic features.5 Histology is notable for a dense mixed inflammatory infiltrate comprised of large pale histiocytes exhibiting emperipolesis, lymphocytes, plasma cells, and occasional eosinophils and neutrophils. Histiocytes stain positive for CD68, CD163, and S100 and are negative for Langerhans cell markers CD1a and CD207.6
The etiology of RDD remains poorly understood. Classic RDD has been associated with both sporadic and familial forms, with somatic mutations identified in the mitogen-activated protein kinase/KRAS pathway in up to one-third of cases, and less frequently in the BRAF gene.14,15 Germline mutations in familial cases of RDD have been identified in the SLC29A3 gene; mutations in this gene are associated with a spectrum of syndromes with histiocytosis and lymphadenopathy.14,15 In contrast, molecular drivers have yet to be identified in cutaneous RDD lesions, and the current predominant hypothesis is that cutaneous RDD has a reactive or immunologic pathophysiology. Autoimmune diseases, infections, and lymphomas have been reported to co-occur with both classic and cutaneous RDD.15 While subclinical viral infections such as Epstein-Barr virus and human herpesvirus 6 have been identified in RDD cases, studies have failed to prove their role as pathogenic drivers of the disease.14,16,17 Commonly reported comorbidities include systemic lupus erythematous, diabetes, hemolytic anemia, acute/chronic uveitis (though it is controversial whether these cases represent orbital involvement in systemic RDD), and Crohn disease.7,8,18,19 Immunohistochemical findings have supported that cells within RDD are activated monocytes responding to T-cell cytokine signaling following an infectious or immunologic insult.20,21
Consensus guidelines on treatment for cutaneous RDD recommend either observation for asymptomatic disease or surgical excision for unifocal lesions with consideration of systemic therapy for refractory cutaneous disease.22,23 Most patients with cutaneous RDD have self-limited disease, but long-term follow-up is recommended following surgical excision to monitor for recurrence, especially if there is a residual positive margin.24 Radiation therapy also may have to be utilized for residual or recurrent disease that becomes symptomatic; however, further studies are needed to determine its efficacy in limiting recurrence.4,12,25 Systemic treatment options include immunosuppressive or immunomodulatory agents such as corticosteroids, methotrexate, and rituximab.5 There currently are no guidelines on length of follow-up, but surveillance is recommended initially at 4 months, followed by 6- to 12-month intervals.22
- Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy. a newly recognized benign clinicopathological entity. Arch Pathol. 1969;87:63-70.
- Foucar E, Rosai J, Dorfman R. Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease): review of the entity. Semin Diagn Pathol. 1990;7:19-73.
- Stefanato CM, Ellerin PS, Bhawan J. Cutaneous sinus histiocytosis (Rosai-Dorfman disease) presenting clinically as vasculitis. J Am Acad Dermatol. 2002;46:775-778.
- Dalia S, Sagatys E, Sokol L, et al. Rosai-Dorfman Disease: tumor biology, clinical features, pathology, and treatment. Cancer Control. 2014;21:322-327.
- Bruce-Brand C, Schneider JW, Schubert P. Rosai-Dorfman disease: an overview. J Clin Pathol. 2020;73:697.
- Bolognia J, Jorizzo J, Schaffer J. Dermatology. 3rd ed. ed. Elsevier Saunders 2012.
- Salva KA, Stenstrom M, Breadon JY, et al. Possible association of cutaneous rosai-dorfman disease and chronic crohn disease: a case series report. JAMA Dermatol. 2014;150:177-181.
- Brenn T, Calonje E, Granter SR, et al. Cutaneous Rosai-Dorfman disease is a distinct clinical entity. Am J Dermatopathol. 2002; 24:385-391.
- Betini N, Munger AM, Rottmann D, et al. Rare presentation of Rosai- Dorfman disease in soft tissue: diagnostic findings and surgical treatment. Case Rep Surg. 2022;2022:8440836.
- Cravero JC, Ibrahim S. Recurrent soft tissue rosai dorfman disease of right medial thigh lipoma with lymph node involvement. Fed Pract. 2024;41(suppl 2):S20-S23
- Tenny SO, McGinness M, Zhang D, et al. Rosai-Dorfman disease presenting as a breast mass and enlarged axillary lymph node mimicking malignancy: a case report and review of the literature. Breast J. 2011;17:516-520.
- Goyal G, Ravindran A, Young JR, et al. Clinicopathological features, treatment approaches, and outcomes in Rosai-Dorfman disease. Haematologica. 2020;105:348-357.
- Li H, Li D, Xia J, et al. Radiological features of Rosai-Dorfman disease: case series and review of the literature. Clin Radiol. 2022;77:E799-E805.
- Elbaz Younes I, Sokol L, Zhang L. Rosai-Dorfman disease between proliferation and neoplasia. Cancers. 2022;14:5271.
- Ravindran A, Rech KL. How I diagnose Rosai-Dorfman disease. Am J Clin Pathol. 2023;160:1-10.
- Kutlubay Z, Bairamov O, Sevim A, et al. Rosai-Dorfman disease: a case report with nodal and cutaneous involvement and review of the literature. Am J Dermatopathol. 2014;36:353-357.
- Luppi M, Barozzi P, Garber R, et al. Expression of human herpesvirus 6 antigens in benign and malignant lymphoproliferative diseases. Am J Pathol. 1998;153:815-823.
- Wang KH, Chen WY, Liu HN, et al. Cutaneous Rosai-Dorfman disease: clinicopathological profiles, spectrum and evolution of 21 lesions in six patients. Br J Dermatol. 2006;154:277-286.
- Vaiselbuh SR, Bryceson YT, Allen CE, et al. Updates on histiocytic disorders. Pediatr Blood Cancer. 2014;61:1329-1335.
- Ravindran A, Goyal G, Go RS, et al. Rosai-Dorfman disease displays a unique monocyte-macrophage phenotype characterized by expression of OCT2. Am J Surg Pathol. 2021;45:35-44.
- Hoogewerf CJ, van Baar ME, Middelkoop E, et al. Impact of facial burns: relationship between depressive symptoms, self-esteem and scar severity. Gen Hosp Psychiatry. 2014;36:271-276.
- Abla O, Jacobsen E, Picarsic J, et al. Consensus recommendations for the diagnosis and clinical management of Rosai-Dorfman-Destombes disease. Blood. 2018;131:2877-2890.
- Al-Khateeb THH. Cutaneous Rosai-Dorfman disease of the face: a comprehensive literature review and case report. J Oral Maxillofacial Surg. 2016;74:528-540.
- Cheng SP, Jeng KS, Liu CL. Subcutaneous Rosai–Dorfman disease: is surgical excision justified? J Eur Acad Dermatol Venereol. 2005; 19:747-750.
- Garcia RA, DiCarlo EF. Rosai-Dorfman disease of bone and soft tissue. Arch Pathol Lab Med. 2021;146:40-46.
- Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy. a newly recognized benign clinicopathological entity. Arch Pathol. 1969;87:63-70.
- Foucar E, Rosai J, Dorfman R. Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease): review of the entity. Semin Diagn Pathol. 1990;7:19-73.
- Stefanato CM, Ellerin PS, Bhawan J. Cutaneous sinus histiocytosis (Rosai-Dorfman disease) presenting clinically as vasculitis. J Am Acad Dermatol. 2002;46:775-778.
- Dalia S, Sagatys E, Sokol L, et al. Rosai-Dorfman Disease: tumor biology, clinical features, pathology, and treatment. Cancer Control. 2014;21:322-327.
- Bruce-Brand C, Schneider JW, Schubert P. Rosai-Dorfman disease: an overview. J Clin Pathol. 2020;73:697.
- Bolognia J, Jorizzo J, Schaffer J. Dermatology. 3rd ed. ed. Elsevier Saunders 2012.
- Salva KA, Stenstrom M, Breadon JY, et al. Possible association of cutaneous rosai-dorfman disease and chronic crohn disease: a case series report. JAMA Dermatol. 2014;150:177-181.
- Brenn T, Calonje E, Granter SR, et al. Cutaneous Rosai-Dorfman disease is a distinct clinical entity. Am J Dermatopathol. 2002; 24:385-391.
- Betini N, Munger AM, Rottmann D, et al. Rare presentation of Rosai- Dorfman disease in soft tissue: diagnostic findings and surgical treatment. Case Rep Surg. 2022;2022:8440836.
- Cravero JC, Ibrahim S. Recurrent soft tissue rosai dorfman disease of right medial thigh lipoma with lymph node involvement. Fed Pract. 2024;41(suppl 2):S20-S23
- Tenny SO, McGinness M, Zhang D, et al. Rosai-Dorfman disease presenting as a breast mass and enlarged axillary lymph node mimicking malignancy: a case report and review of the literature. Breast J. 2011;17:516-520.
- Goyal G, Ravindran A, Young JR, et al. Clinicopathological features, treatment approaches, and outcomes in Rosai-Dorfman disease. Haematologica. 2020;105:348-357.
- Li H, Li D, Xia J, et al. Radiological features of Rosai-Dorfman disease: case series and review of the literature. Clin Radiol. 2022;77:E799-E805.
- Elbaz Younes I, Sokol L, Zhang L. Rosai-Dorfman disease between proliferation and neoplasia. Cancers. 2022;14:5271.
- Ravindran A, Rech KL. How I diagnose Rosai-Dorfman disease. Am J Clin Pathol. 2023;160:1-10.
- Kutlubay Z, Bairamov O, Sevim A, et al. Rosai-Dorfman disease: a case report with nodal and cutaneous involvement and review of the literature. Am J Dermatopathol. 2014;36:353-357.
- Luppi M, Barozzi P, Garber R, et al. Expression of human herpesvirus 6 antigens in benign and malignant lymphoproliferative diseases. Am J Pathol. 1998;153:815-823.
- Wang KH, Chen WY, Liu HN, et al. Cutaneous Rosai-Dorfman disease: clinicopathological profiles, spectrum and evolution of 21 lesions in six patients. Br J Dermatol. 2006;154:277-286.
- Vaiselbuh SR, Bryceson YT, Allen CE, et al. Updates on histiocytic disorders. Pediatr Blood Cancer. 2014;61:1329-1335.
- Ravindran A, Goyal G, Go RS, et al. Rosai-Dorfman disease displays a unique monocyte-macrophage phenotype characterized by expression of OCT2. Am J Surg Pathol. 2021;45:35-44.
- Hoogewerf CJ, van Baar ME, Middelkoop E, et al. Impact of facial burns: relationship between depressive symptoms, self-esteem and scar severity. Gen Hosp Psychiatry. 2014;36:271-276.
- Abla O, Jacobsen E, Picarsic J, et al. Consensus recommendations for the diagnosis and clinical management of Rosai-Dorfman-Destombes disease. Blood. 2018;131:2877-2890.
- Al-Khateeb THH. Cutaneous Rosai-Dorfman disease of the face: a comprehensive literature review and case report. J Oral Maxillofacial Surg. 2016;74:528-540.
- Cheng SP, Jeng KS, Liu CL. Subcutaneous Rosai–Dorfman disease: is surgical excision justified? J Eur Acad Dermatol Venereol. 2005; 19:747-750.
- Garcia RA, DiCarlo EF. Rosai-Dorfman disease of bone and soft tissue. Arch Pathol Lab Med. 2021;146:40-46.
A Solitary Axillary Subcutaneous Mass
A Solitary Axillary Subcutaneous Mass
A 34-year-old man presented to our dermatology clinic for evaluation of a lesion in the right axilla of 1 year’s duration that had recently increased in size. The lesion was nontender and intermittently pruritic and was associated with focal hypohidrosis. The patient denied any fevers, chills, or recent weight change. His medical history was otherwise unremarkable. His only medications were daily ashwagandha and vitamin B and C supplements. On physical examination, a firm, 6-cm, subcutaneous nodule was noted in the right axilla with central alopecia and without a clear punctum. He had no palpable cervical, postauricular, or inguinal lymphadenopathy. The left axilla was clear, and there were no other relevant skin findings. Laboratory testing including a complete blood count, comprehensive metabolic panel, and sexually transmitted infections panel was unremarkable. Ultrasonography and subsequent magnetic resonance imaging of the right axilla showed a 4.9-cm nodule located in the subcutaneous fat with minimal deep infiltration and relatively smooth margins. An incisional biopsy of the lesion was performed.
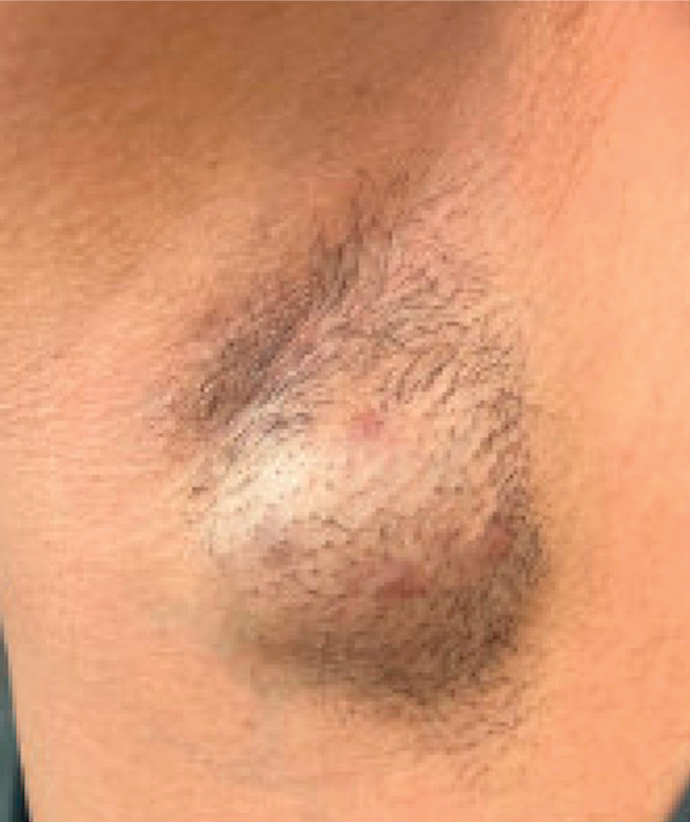
Xylazine-Induced Skin Necrosis
Xylazine-Induced Skin Necrosis
To the Editor:
Xylazine, commonly referred to by its street name tranq, is a veterinary tranquilizer that has recently gained attention due to its increasing misuse in human populations. It often is combined with recreational drugs like fentanyl to extend the duration of drug effects. As a partial α2 receptor agonist, xylazine acts by reducing dopamine and norepinephrine release, resulting in sedative effects. This case report highlights xylazine skin necrosis manifesting as wrist drop and chronic wounds in a patient with a history of intravenous (IV) drug use.
A 35-year-old man with a history of IV drug use presented to the emergency department with a nonprogressive right wrist drop that had persisted for 2 weeks, along with new-onset left wrist drop of 1 day’s duration. The patient did not report any sensory symptoms or pain. Physical examination revealed an ulcerated necrotic plaque with hemorrhagic crust and focal areas of scarring on the right posterior forearm (Figure 1). The left hand exhibited a well-healed pink scar symmetric to the ulcer on the right forearm. The patient reported a history of a similar ulcer on the left hand that had resolved after discontinuation of IV drug use in that arm. He denied any history of trauma to the area.
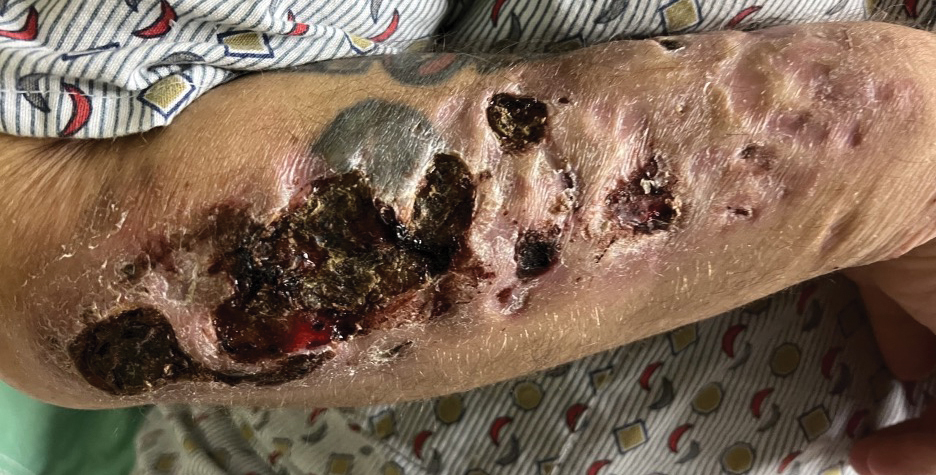
The patient’s laboratory results demonstrated elevated inflammatory markers, including an erythrocyte sedimentation rate of 105 mm/h (reference range, <15 mm/h in men younger than 50 years) and a C-reactive protein level of 7.7 mg/dL (reference range, <0.9 mg/dL). Additionally, antinuclear antibody and antineutrophil cytoplasmic antibody tests were positive. A urine drug screen returned positive results for various substances, including cocaine, cocaine metabolites, fentanyl, norfentanyl, β-hydroxyfentanyl or fentanyl metabolite, caffeine, caffeine metabolite or theophylline, nicotine metabolite, and xylazine. Magnetic resonance imaging of the right upper extremity excluded osteomyelitis but revealed multiple subepidermal abscesses.
A punch biopsy from the right forearm demonstrated an ulcer with a mixed infiltrate, dermal necrosis, and clusters of Gram-positive cocci, indicating a bacterial infection. There was no evidence of leukocytoclastic vasculitis (Figures 2 and 3). Electromyography confirmed mononeuritis multiplex as the cause of the right wrist drop. The patient was found to have cytoplasmic antineutrophil cytoplasmic antibody–positive vasculitis in the setting of levamisole-adulterated cocaine use. Since no vasculitis was identified on histopathology of the ulcer and xylazine was detected on drug screening, a diagnosis of xylazine-induced skin necrosis was made. In our case, the patient did not show evidence of active osteomyelitis or sepsis and left the hospital against medical advice without adequate wound debridement.
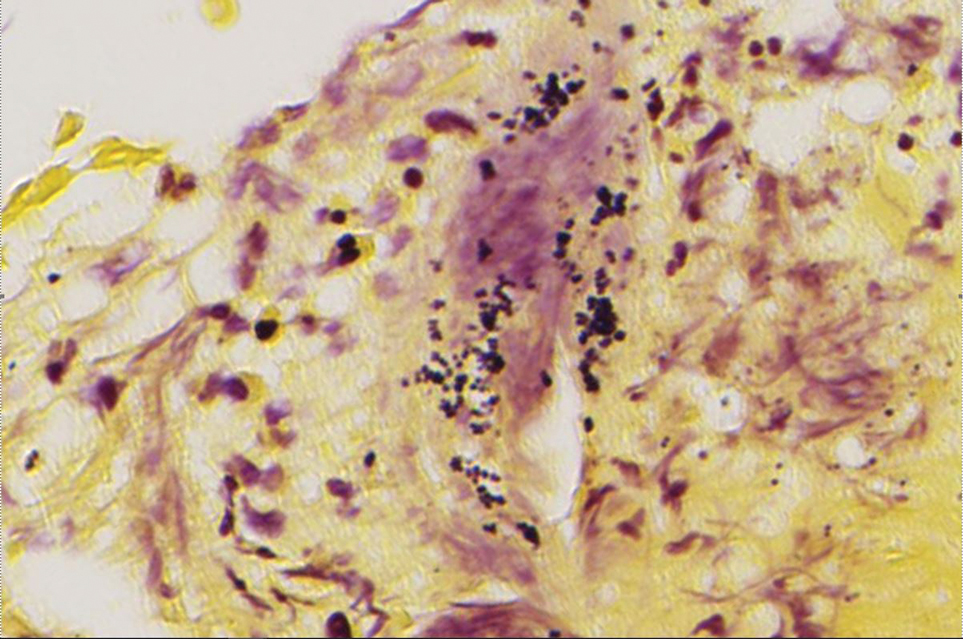
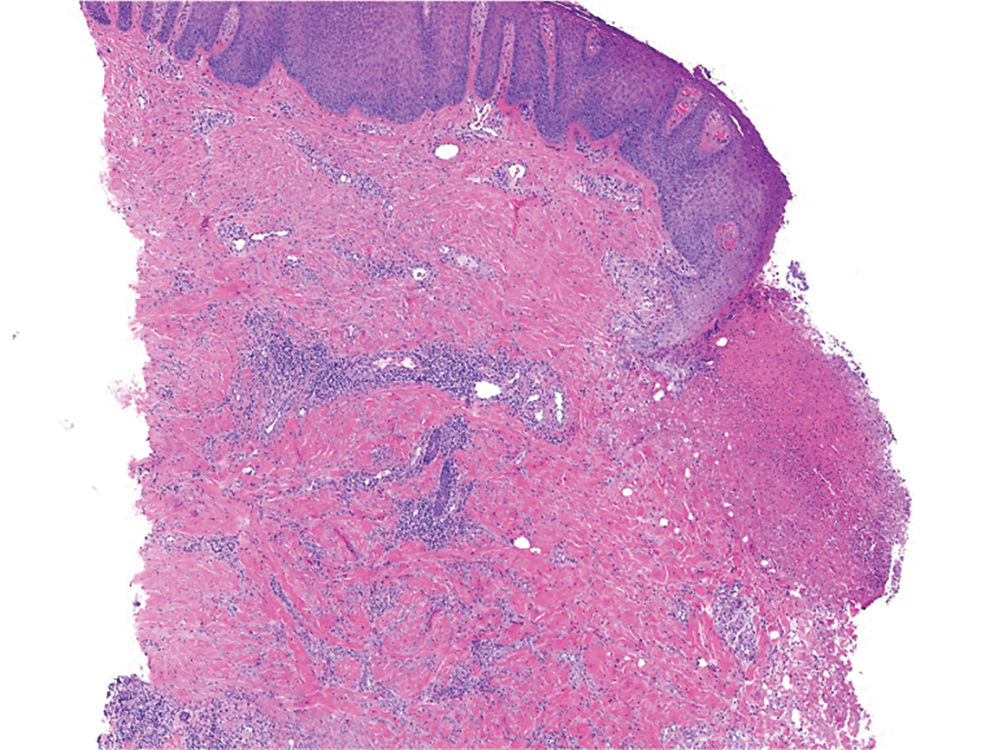
Our case highlights xylazine-induced skin necrosis that can occur in individuals who use IV drugs. The combination of xylazine with other recreational drugs such as fentanyl poses unique challenges for clinicians. Xylazine has been increasingly found in cases of overdose-related mortality1 and recently has been reported to induce skin ulcers.2 Xylazine intoxication, though uncommon, can result in distinct clinical presentations, including recalcitrant skin ulcers and deep necrotizing wounds.
The precise mechanism behind these wounds remains unclear. Xylazine is a partial α2 receptor agonist, and it is postulated that the necrotic wounds develop secondary to local vasoconstriction, leading to decreased skin perfusion.3 A recent study found that xylazine used in combination with cocaine or an active metabolite in heroin can cause cytotoxicity to vascular endothelial cells, which can lead to dysregulation of vascular tone.4 Decreased perfusion and impaired wound healing put patients at risk for secondary infections, infected ulcers, osteomyelitis, and sepsis.
In patients with known fentanyl use in conjunction with skin necrosis, a high degree of suspicion for xylazine intoxication should be employed. Ruling out vasculitis (via serologic markers and skin biopsy) as well as atypical skin infections is important in these patients to identify potential cases of xylazine-induced skin necrosis. Other IV drugs such as krokodil (desomorphine) can cause severe skin necrosis and therefore should be considered in these patients. Early detection of these skin ulcers is imperative, as delayed diagnosis increases the risk for osteomyelitis and/or the need for amputation.
This case emphasizes the importance of health care providers remaining vigilant about emerging trends in drug misuse. Early recognition of xylazine intoxication and its potential complications is crucial for timely intervention and appropriate management, which may include wound debridement and antibiotic therapy. In addition, proper counseling regarding discontinuation of drug use is important in wound healing, though this poses a challenging conversation with the patient. Increased awareness among health care professionals and continued research in illicit drug–induced skin necrosis will aid in better understanding and addressing the growing issue of xylazine misuse.
- Friedman J, Montero F, Bourgois P, et al. Xylazine spreads across the US: a growing component of the increasingly synthetic and polysubstance overdose crisis. Drug Alcohol Depend. 2022;233:109380. doi:10.1016/j.drugalcdep.2022.109380
- Malayala SV, Papudesi BN, Bobb R, et al. Xylazine-induced skin ulcers in a person who injects drugs in Philadelphia, Pennsylvania, USA. Cureus. 2022;14:E28160. doi:10.7759/cureus.28160
- McNinch J, Maguire M, Wallace L, et al. A case of skin necrosis caused by intravenous xylazine abuse. Abstract presented at: SHM Converge; May 3-7, 2021.
- Silva-Torres LA, Vélez C, Lyvia Alvarez J, et al. Toxic effects of xylazine on endothelial cells in combination with cocaine and 6-monoacetylmorphine. Toxicol In Vitro. 2014;28:1312-1319. doi:10.1016/j.tiv.2014.06.013
To the Editor:
Xylazine, commonly referred to by its street name tranq, is a veterinary tranquilizer that has recently gained attention due to its increasing misuse in human populations. It often is combined with recreational drugs like fentanyl to extend the duration of drug effects. As a partial α2 receptor agonist, xylazine acts by reducing dopamine and norepinephrine release, resulting in sedative effects. This case report highlights xylazine skin necrosis manifesting as wrist drop and chronic wounds in a patient with a history of intravenous (IV) drug use.
A 35-year-old man with a history of IV drug use presented to the emergency department with a nonprogressive right wrist drop that had persisted for 2 weeks, along with new-onset left wrist drop of 1 day’s duration. The patient did not report any sensory symptoms or pain. Physical examination revealed an ulcerated necrotic plaque with hemorrhagic crust and focal areas of scarring on the right posterior forearm (Figure 1). The left hand exhibited a well-healed pink scar symmetric to the ulcer on the right forearm. The patient reported a history of a similar ulcer on the left hand that had resolved after discontinuation of IV drug use in that arm. He denied any history of trauma to the area.

The patient’s laboratory results demonstrated elevated inflammatory markers, including an erythrocyte sedimentation rate of 105 mm/h (reference range, <15 mm/h in men younger than 50 years) and a C-reactive protein level of 7.7 mg/dL (reference range, <0.9 mg/dL). Additionally, antinuclear antibody and antineutrophil cytoplasmic antibody tests were positive. A urine drug screen returned positive results for various substances, including cocaine, cocaine metabolites, fentanyl, norfentanyl, β-hydroxyfentanyl or fentanyl metabolite, caffeine, caffeine metabolite or theophylline, nicotine metabolite, and xylazine. Magnetic resonance imaging of the right upper extremity excluded osteomyelitis but revealed multiple subepidermal abscesses.
A punch biopsy from the right forearm demonstrated an ulcer with a mixed infiltrate, dermal necrosis, and clusters of Gram-positive cocci, indicating a bacterial infection. There was no evidence of leukocytoclastic vasculitis (Figures 2 and 3). Electromyography confirmed mononeuritis multiplex as the cause of the right wrist drop. The patient was found to have cytoplasmic antineutrophil cytoplasmic antibody–positive vasculitis in the setting of levamisole-adulterated cocaine use. Since no vasculitis was identified on histopathology of the ulcer and xylazine was detected on drug screening, a diagnosis of xylazine-induced skin necrosis was made. In our case, the patient did not show evidence of active osteomyelitis or sepsis and left the hospital against medical advice without adequate wound debridement.


Our case highlights xylazine-induced skin necrosis that can occur in individuals who use IV drugs. The combination of xylazine with other recreational drugs such as fentanyl poses unique challenges for clinicians. Xylazine has been increasingly found in cases of overdose-related mortality1 and recently has been reported to induce skin ulcers.2 Xylazine intoxication, though uncommon, can result in distinct clinical presentations, including recalcitrant skin ulcers and deep necrotizing wounds.
The precise mechanism behind these wounds remains unclear. Xylazine is a partial α2 receptor agonist, and it is postulated that the necrotic wounds develop secondary to local vasoconstriction, leading to decreased skin perfusion.3 A recent study found that xylazine used in combination with cocaine or an active metabolite in heroin can cause cytotoxicity to vascular endothelial cells, which can lead to dysregulation of vascular tone.4 Decreased perfusion and impaired wound healing put patients at risk for secondary infections, infected ulcers, osteomyelitis, and sepsis.
In patients with known fentanyl use in conjunction with skin necrosis, a high degree of suspicion for xylazine intoxication should be employed. Ruling out vasculitis (via serologic markers and skin biopsy) as well as atypical skin infections is important in these patients to identify potential cases of xylazine-induced skin necrosis. Other IV drugs such as krokodil (desomorphine) can cause severe skin necrosis and therefore should be considered in these patients. Early detection of these skin ulcers is imperative, as delayed diagnosis increases the risk for osteomyelitis and/or the need for amputation.
This case emphasizes the importance of health care providers remaining vigilant about emerging trends in drug misuse. Early recognition of xylazine intoxication and its potential complications is crucial for timely intervention and appropriate management, which may include wound debridement and antibiotic therapy. In addition, proper counseling regarding discontinuation of drug use is important in wound healing, though this poses a challenging conversation with the patient. Increased awareness among health care professionals and continued research in illicit drug–induced skin necrosis will aid in better understanding and addressing the growing issue of xylazine misuse.
To the Editor:
Xylazine, commonly referred to by its street name tranq, is a veterinary tranquilizer that has recently gained attention due to its increasing misuse in human populations. It often is combined with recreational drugs like fentanyl to extend the duration of drug effects. As a partial α2 receptor agonist, xylazine acts by reducing dopamine and norepinephrine release, resulting in sedative effects. This case report highlights xylazine skin necrosis manifesting as wrist drop and chronic wounds in a patient with a history of intravenous (IV) drug use.
A 35-year-old man with a history of IV drug use presented to the emergency department with a nonprogressive right wrist drop that had persisted for 2 weeks, along with new-onset left wrist drop of 1 day’s duration. The patient did not report any sensory symptoms or pain. Physical examination revealed an ulcerated necrotic plaque with hemorrhagic crust and focal areas of scarring on the right posterior forearm (Figure 1). The left hand exhibited a well-healed pink scar symmetric to the ulcer on the right forearm. The patient reported a history of a similar ulcer on the left hand that had resolved after discontinuation of IV drug use in that arm. He denied any history of trauma to the area.

The patient’s laboratory results demonstrated elevated inflammatory markers, including an erythrocyte sedimentation rate of 105 mm/h (reference range, <15 mm/h in men younger than 50 years) and a C-reactive protein level of 7.7 mg/dL (reference range, <0.9 mg/dL). Additionally, antinuclear antibody and antineutrophil cytoplasmic antibody tests were positive. A urine drug screen returned positive results for various substances, including cocaine, cocaine metabolites, fentanyl, norfentanyl, β-hydroxyfentanyl or fentanyl metabolite, caffeine, caffeine metabolite or theophylline, nicotine metabolite, and xylazine. Magnetic resonance imaging of the right upper extremity excluded osteomyelitis but revealed multiple subepidermal abscesses.
A punch biopsy from the right forearm demonstrated an ulcer with a mixed infiltrate, dermal necrosis, and clusters of Gram-positive cocci, indicating a bacterial infection. There was no evidence of leukocytoclastic vasculitis (Figures 2 and 3). Electromyography confirmed mononeuritis multiplex as the cause of the right wrist drop. The patient was found to have cytoplasmic antineutrophil cytoplasmic antibody–positive vasculitis in the setting of levamisole-adulterated cocaine use. Since no vasculitis was identified on histopathology of the ulcer and xylazine was detected on drug screening, a diagnosis of xylazine-induced skin necrosis was made. In our case, the patient did not show evidence of active osteomyelitis or sepsis and left the hospital against medical advice without adequate wound debridement.


Our case highlights xylazine-induced skin necrosis that can occur in individuals who use IV drugs. The combination of xylazine with other recreational drugs such as fentanyl poses unique challenges for clinicians. Xylazine has been increasingly found in cases of overdose-related mortality1 and recently has been reported to induce skin ulcers.2 Xylazine intoxication, though uncommon, can result in distinct clinical presentations, including recalcitrant skin ulcers and deep necrotizing wounds.
The precise mechanism behind these wounds remains unclear. Xylazine is a partial α2 receptor agonist, and it is postulated that the necrotic wounds develop secondary to local vasoconstriction, leading to decreased skin perfusion.3 A recent study found that xylazine used in combination with cocaine or an active metabolite in heroin can cause cytotoxicity to vascular endothelial cells, which can lead to dysregulation of vascular tone.4 Decreased perfusion and impaired wound healing put patients at risk for secondary infections, infected ulcers, osteomyelitis, and sepsis.
In patients with known fentanyl use in conjunction with skin necrosis, a high degree of suspicion for xylazine intoxication should be employed. Ruling out vasculitis (via serologic markers and skin biopsy) as well as atypical skin infections is important in these patients to identify potential cases of xylazine-induced skin necrosis. Other IV drugs such as krokodil (desomorphine) can cause severe skin necrosis and therefore should be considered in these patients. Early detection of these skin ulcers is imperative, as delayed diagnosis increases the risk for osteomyelitis and/or the need for amputation.
This case emphasizes the importance of health care providers remaining vigilant about emerging trends in drug misuse. Early recognition of xylazine intoxication and its potential complications is crucial for timely intervention and appropriate management, which may include wound debridement and antibiotic therapy. In addition, proper counseling regarding discontinuation of drug use is important in wound healing, though this poses a challenging conversation with the patient. Increased awareness among health care professionals and continued research in illicit drug–induced skin necrosis will aid in better understanding and addressing the growing issue of xylazine misuse.
- Friedman J, Montero F, Bourgois P, et al. Xylazine spreads across the US: a growing component of the increasingly synthetic and polysubstance overdose crisis. Drug Alcohol Depend. 2022;233:109380. doi:10.1016/j.drugalcdep.2022.109380
- Malayala SV, Papudesi BN, Bobb R, et al. Xylazine-induced skin ulcers in a person who injects drugs in Philadelphia, Pennsylvania, USA. Cureus. 2022;14:E28160. doi:10.7759/cureus.28160
- McNinch J, Maguire M, Wallace L, et al. A case of skin necrosis caused by intravenous xylazine abuse. Abstract presented at: SHM Converge; May 3-7, 2021.
- Silva-Torres LA, Vélez C, Lyvia Alvarez J, et al. Toxic effects of xylazine on endothelial cells in combination with cocaine and 6-monoacetylmorphine. Toxicol In Vitro. 2014;28:1312-1319. doi:10.1016/j.tiv.2014.06.013
- Friedman J, Montero F, Bourgois P, et al. Xylazine spreads across the US: a growing component of the increasingly synthetic and polysubstance overdose crisis. Drug Alcohol Depend. 2022;233:109380. doi:10.1016/j.drugalcdep.2022.109380
- Malayala SV, Papudesi BN, Bobb R, et al. Xylazine-induced skin ulcers in a person who injects drugs in Philadelphia, Pennsylvania, USA. Cureus. 2022;14:E28160. doi:10.7759/cureus.28160
- McNinch J, Maguire M, Wallace L, et al. A case of skin necrosis caused by intravenous xylazine abuse. Abstract presented at: SHM Converge; May 3-7, 2021.
- Silva-Torres LA, Vélez C, Lyvia Alvarez J, et al. Toxic effects of xylazine on endothelial cells in combination with cocaine and 6-monoacetylmorphine. Toxicol In Vitro. 2014;28:1312-1319. doi:10.1016/j.tiv.2014.06.013
Xylazine-Induced Skin Necrosis
Xylazine-Induced Skin Necrosis
Practice Points
- Dermatologists should be aware of the potential for xylazine to cause ulcers in patients with a history of intravenous drug use.
- Early recognition of xylazine skin ulcers is imperative, as delayed diagnosis increases morbidity such as soft-tissue and bone infection, sepsis, and death.
Multiple Papules and Pustules on the Face and Neck
Multiple Papules and Pustules on the Face and Neck
THE DIAGNOSIS: Demodicosis
Direct microscopic examination of the purulent fluid revealed a considerable number of actively motile Demodex mites (Figure). Based on the microscopy results and the patient’s history of prolonged topical immunosuppressive therapy, a known risk factor for Demodex overgrowth, a diagnosis of demodicosis was made. The patient was prescribed a single dose of oral metronidazole 2 g as well as metronidazole solution 0.5% to be applied 3 times daily. The folliculitis gradually improved and eventually resolved completely.
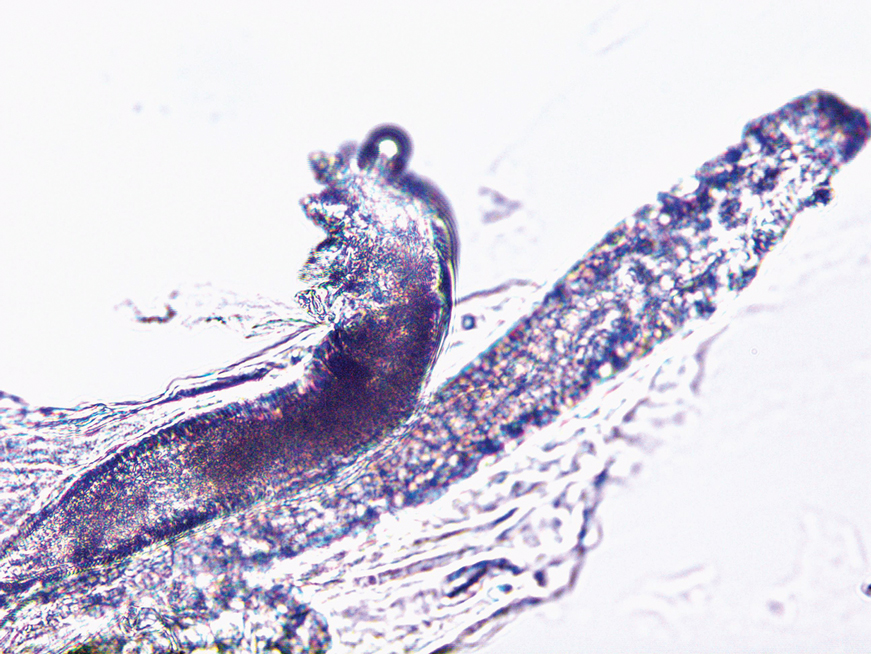
Demodex is a parasitic mite inhabiting the pilosebaceous units of human skin. Evidence suggests the vast majority of adults carry these mites. Demodex mites maintain a balance with the human immune system in appropriate microenvironments, with the immune system controlling their numbers without eliciting an inflammatory response; however, immunosuppression, as induced by topical corticosteroids and other immunomodulators, can lead to an increase in Demodex mite populations on facial skin. Clinical manifestations and severity of demodicosis are highly variable, ranging from nonspecific dry, sensitive skin and papules to nodules or granulomas, depending on mite density, the cutaneous microenvironment, and the host immune response.1 Consequently, demodicosis often is mistaken for other dermatologic conditions with similar skin lesions.
High Demodex mite density is considered a pathogenic factor in demodicosis; therefore, determining Demodex mite density is essential to the diagnosis of demodicosis. Standard skin surface biopsy and direct microscopic examination commonly are used methods for measuring Demodex mite density; however, the accuracy of these methods is subject to the technical proficiency of the investigator. Noninvasive examination tools like dermoscopy and confocal laser scanning also offer advantages in diagnosing demodicosis. Dermoscopy, by direct contact with skin lesions, typically reveals gelatinous filaments extending from the follicular openings.
Importantly, Demodex mite density alone does not determine the severity of clinical symptoms. In addition, mites may migrate to the skin surface or reside deep within follicles, rendering them difficult to detect with standard examination methods.1 Therefore, diagnostic criteria should extend beyond mite proliferation to include characteristic clinical lesions, response to acaricidal therapy, and normalization of mite density.
Rosacea was included in the differential diagnosis for our patient, but it typically manifests in the central facial area (eg, forehead, nose, chin). Patients may have a history of facial flushing associated with alcohol consumption, heat exposure, or emotional stress.2 Additionally, rosacea typically has an insidious onset and does not erupt suddenly within a short period of time; however, our patient presented with a sudden onset of widespread papules and pustules on the face without facial flushing, and there was no exacerbation upon exposure to heat or emotional stress. Furthermore, rosacea tends to be recurrent and challenging to cure, whereas our patient responded rapidly to treatment without recurrence. Therefore, the likelihood of rosacea was minimal. Histopathologic examination also can differentiate between rosacea and demodicosis. Histologically, the features of rosacea include dilated blood and lymphatic vessels and infiltration of T lymphocytes, macrophages, and mast cells around blood vessels, often with increased solar elastosis and dermal edema.3 Demodicosis can reveal Demodex mites within the infundibulum of hair follicles, with dense neutrophil and monocyte infiltration around and between the infundibula.4
Bacterial folliculitis is primarily characterized by perifollicular erythema, papules, and pustules, often accompanied by pain. Positive bacterial culture of purulent fluid is indicative.5 Our patient’s lesions shared certain similarities with bacterial folliculitis but lacked the characteristic pain, instead exhibiting pronounced pruritus. Remarkable therapeutic efficacy was observed following topical acaricidal treatment, thus rendering the diagnosis of bacterial folliculitis less probable.
Acne vulgaris is a noninfectious folliculitis caused by follicular occlusion. Abnormal keratinization leads to the obstruction of follicles by keratin, hindering the outflow of sebum from the follicles. Sebum accumulation within the follicles provides a rich substrate for Propionibacterium acnes, which metabolizes sebum into proinflammatory free fatty acids, resulting in the formation of comedones, papules, and pustules.5 Our patient did not exhibit comedonal lesions on the face and lacked a seborrheic complexion, hence diminishing the likelihood of acne vulgaris.
Tinea corporis is another intensely pruritic condition, especially in areas subjected to prolonged use of topical immunosuppressants. It is caused by dermatophyte fungi and typically manifests as erythematous pruritic patches, often presenting as ring-shaped lesions with active margins and sometimes accompanied by scaling.6 While long-term use of immunosuppressants may be a risk factor for fungal infections and increase the probability of tinea corporis, our patient’s presentation of papules and pustules without a ring-shaped configuration or scaling diminished the likelihood of tinea corporis.
Our patient represents an intriguing case of an eruptive form of demodicosis induced by long-term intermittent and inconsistent application of topical immunosuppressive agents. Demodicosis encompasses a spectrum of clinical presentations, including pityriasis folliculorum, rosacealike, folliculitislike, and perioral dermatitis–like forms.1 It is prone to misdiagnosis, as it is clinically similar to other conditions, such as acne, rosacea, or bacterial folliculitis, and it also is susceptible to missed diagnosis. Demodicosis tends to erupt in immunocompromised individuals, and the use of topical immunosuppressive and corticosteroid medications can exacerbate Demodex activity. Dermatologists should be aware that demodicosis is not a rare skin disorder, and timely identification and diagnosis can reduce the incidence of disease and improve quality of life for affected patients. Conversely, the consequences of misdiagnosis can be severe, with inappropriate treatment potentially exacerbating the condition.
- Paichitrojjana A. Demodex: the worst enemies are the ones that used to be friends. Dermatol Reports. 2022;14:9339. doi:10.4081 /dr.2022.9339
- Del RJ, Baldwin H, Bhatia N, et al. A review of the diagnostic and therapeutic gaps in rosacea management: consensus opinion. Dermatol Ther (Heidelb). 2024;14:271-284. doi:10.1007/s13555-023-01087-8
- Powell FC. The histopathology of rosacea: ‘where’s the beef?’ Dermatology. 2004;209:173-174. doi:10.1159/000079884
- Helou W, Avitan-Hersh E, Bergman R. Demodex folliculitis of the scalp: clinicopathological study of an uncommon entity. Am J Dermatopathol. 2016;38:658-663. doi:10.1097/DAD.0000000000000512
- Laureano AC, Schwartz RA, Cohen PJ. Facial bacterial infections: folliculitis. Clin Dermatol. 2014;32:711-714. doi:10.1016 /j.clindermatol.2014.02.009
- Leung AK, Lam JM, Leong KF, et al. Tinea corporis: an updated review. Drugs Context. 2020;9. doi:10.7573/dic.2020-5-6
THE DIAGNOSIS: Demodicosis
Direct microscopic examination of the purulent fluid revealed a considerable number of actively motile Demodex mites (Figure). Based on the microscopy results and the patient’s history of prolonged topical immunosuppressive therapy, a known risk factor for Demodex overgrowth, a diagnosis of demodicosis was made. The patient was prescribed a single dose of oral metronidazole 2 g as well as metronidazole solution 0.5% to be applied 3 times daily. The folliculitis gradually improved and eventually resolved completely.

Demodex is a parasitic mite inhabiting the pilosebaceous units of human skin. Evidence suggests the vast majority of adults carry these mites. Demodex mites maintain a balance with the human immune system in appropriate microenvironments, with the immune system controlling their numbers without eliciting an inflammatory response; however, immunosuppression, as induced by topical corticosteroids and other immunomodulators, can lead to an increase in Demodex mite populations on facial skin. Clinical manifestations and severity of demodicosis are highly variable, ranging from nonspecific dry, sensitive skin and papules to nodules or granulomas, depending on mite density, the cutaneous microenvironment, and the host immune response.1 Consequently, demodicosis often is mistaken for other dermatologic conditions with similar skin lesions.
High Demodex mite density is considered a pathogenic factor in demodicosis; therefore, determining Demodex mite density is essential to the diagnosis of demodicosis. Standard skin surface biopsy and direct microscopic examination commonly are used methods for measuring Demodex mite density; however, the accuracy of these methods is subject to the technical proficiency of the investigator. Noninvasive examination tools like dermoscopy and confocal laser scanning also offer advantages in diagnosing demodicosis. Dermoscopy, by direct contact with skin lesions, typically reveals gelatinous filaments extending from the follicular openings.
Importantly, Demodex mite density alone does not determine the severity of clinical symptoms. In addition, mites may migrate to the skin surface or reside deep within follicles, rendering them difficult to detect with standard examination methods.1 Therefore, diagnostic criteria should extend beyond mite proliferation to include characteristic clinical lesions, response to acaricidal therapy, and normalization of mite density.
Rosacea was included in the differential diagnosis for our patient, but it typically manifests in the central facial area (eg, forehead, nose, chin). Patients may have a history of facial flushing associated with alcohol consumption, heat exposure, or emotional stress.2 Additionally, rosacea typically has an insidious onset and does not erupt suddenly within a short period of time; however, our patient presented with a sudden onset of widespread papules and pustules on the face without facial flushing, and there was no exacerbation upon exposure to heat or emotional stress. Furthermore, rosacea tends to be recurrent and challenging to cure, whereas our patient responded rapidly to treatment without recurrence. Therefore, the likelihood of rosacea was minimal. Histopathologic examination also can differentiate between rosacea and demodicosis. Histologically, the features of rosacea include dilated blood and lymphatic vessels and infiltration of T lymphocytes, macrophages, and mast cells around blood vessels, often with increased solar elastosis and dermal edema.3 Demodicosis can reveal Demodex mites within the infundibulum of hair follicles, with dense neutrophil and monocyte infiltration around and between the infundibula.4
Bacterial folliculitis is primarily characterized by perifollicular erythema, papules, and pustules, often accompanied by pain. Positive bacterial culture of purulent fluid is indicative.5 Our patient’s lesions shared certain similarities with bacterial folliculitis but lacked the characteristic pain, instead exhibiting pronounced pruritus. Remarkable therapeutic efficacy was observed following topical acaricidal treatment, thus rendering the diagnosis of bacterial folliculitis less probable.
Acne vulgaris is a noninfectious folliculitis caused by follicular occlusion. Abnormal keratinization leads to the obstruction of follicles by keratin, hindering the outflow of sebum from the follicles. Sebum accumulation within the follicles provides a rich substrate for Propionibacterium acnes, which metabolizes sebum into proinflammatory free fatty acids, resulting in the formation of comedones, papules, and pustules.5 Our patient did not exhibit comedonal lesions on the face and lacked a seborrheic complexion, hence diminishing the likelihood of acne vulgaris.
Tinea corporis is another intensely pruritic condition, especially in areas subjected to prolonged use of topical immunosuppressants. It is caused by dermatophyte fungi and typically manifests as erythematous pruritic patches, often presenting as ring-shaped lesions with active margins and sometimes accompanied by scaling.6 While long-term use of immunosuppressants may be a risk factor for fungal infections and increase the probability of tinea corporis, our patient’s presentation of papules and pustules without a ring-shaped configuration or scaling diminished the likelihood of tinea corporis.
Our patient represents an intriguing case of an eruptive form of demodicosis induced by long-term intermittent and inconsistent application of topical immunosuppressive agents. Demodicosis encompasses a spectrum of clinical presentations, including pityriasis folliculorum, rosacealike, folliculitislike, and perioral dermatitis–like forms.1 It is prone to misdiagnosis, as it is clinically similar to other conditions, such as acne, rosacea, or bacterial folliculitis, and it also is susceptible to missed diagnosis. Demodicosis tends to erupt in immunocompromised individuals, and the use of topical immunosuppressive and corticosteroid medications can exacerbate Demodex activity. Dermatologists should be aware that demodicosis is not a rare skin disorder, and timely identification and diagnosis can reduce the incidence of disease and improve quality of life for affected patients. Conversely, the consequences of misdiagnosis can be severe, with inappropriate treatment potentially exacerbating the condition.
THE DIAGNOSIS: Demodicosis
Direct microscopic examination of the purulent fluid revealed a considerable number of actively motile Demodex mites (Figure). Based on the microscopy results and the patient’s history of prolonged topical immunosuppressive therapy, a known risk factor for Demodex overgrowth, a diagnosis of demodicosis was made. The patient was prescribed a single dose of oral metronidazole 2 g as well as metronidazole solution 0.5% to be applied 3 times daily. The folliculitis gradually improved and eventually resolved completely.

Demodex is a parasitic mite inhabiting the pilosebaceous units of human skin. Evidence suggests the vast majority of adults carry these mites. Demodex mites maintain a balance with the human immune system in appropriate microenvironments, with the immune system controlling their numbers without eliciting an inflammatory response; however, immunosuppression, as induced by topical corticosteroids and other immunomodulators, can lead to an increase in Demodex mite populations on facial skin. Clinical manifestations and severity of demodicosis are highly variable, ranging from nonspecific dry, sensitive skin and papules to nodules or granulomas, depending on mite density, the cutaneous microenvironment, and the host immune response.1 Consequently, demodicosis often is mistaken for other dermatologic conditions with similar skin lesions.
High Demodex mite density is considered a pathogenic factor in demodicosis; therefore, determining Demodex mite density is essential to the diagnosis of demodicosis. Standard skin surface biopsy and direct microscopic examination commonly are used methods for measuring Demodex mite density; however, the accuracy of these methods is subject to the technical proficiency of the investigator. Noninvasive examination tools like dermoscopy and confocal laser scanning also offer advantages in diagnosing demodicosis. Dermoscopy, by direct contact with skin lesions, typically reveals gelatinous filaments extending from the follicular openings.
Importantly, Demodex mite density alone does not determine the severity of clinical symptoms. In addition, mites may migrate to the skin surface or reside deep within follicles, rendering them difficult to detect with standard examination methods.1 Therefore, diagnostic criteria should extend beyond mite proliferation to include characteristic clinical lesions, response to acaricidal therapy, and normalization of mite density.
Rosacea was included in the differential diagnosis for our patient, but it typically manifests in the central facial area (eg, forehead, nose, chin). Patients may have a history of facial flushing associated with alcohol consumption, heat exposure, or emotional stress.2 Additionally, rosacea typically has an insidious onset and does not erupt suddenly within a short period of time; however, our patient presented with a sudden onset of widespread papules and pustules on the face without facial flushing, and there was no exacerbation upon exposure to heat or emotional stress. Furthermore, rosacea tends to be recurrent and challenging to cure, whereas our patient responded rapidly to treatment without recurrence. Therefore, the likelihood of rosacea was minimal. Histopathologic examination also can differentiate between rosacea and demodicosis. Histologically, the features of rosacea include dilated blood and lymphatic vessels and infiltration of T lymphocytes, macrophages, and mast cells around blood vessels, often with increased solar elastosis and dermal edema.3 Demodicosis can reveal Demodex mites within the infundibulum of hair follicles, with dense neutrophil and monocyte infiltration around and between the infundibula.4
Bacterial folliculitis is primarily characterized by perifollicular erythema, papules, and pustules, often accompanied by pain. Positive bacterial culture of purulent fluid is indicative.5 Our patient’s lesions shared certain similarities with bacterial folliculitis but lacked the characteristic pain, instead exhibiting pronounced pruritus. Remarkable therapeutic efficacy was observed following topical acaricidal treatment, thus rendering the diagnosis of bacterial folliculitis less probable.
Acne vulgaris is a noninfectious folliculitis caused by follicular occlusion. Abnormal keratinization leads to the obstruction of follicles by keratin, hindering the outflow of sebum from the follicles. Sebum accumulation within the follicles provides a rich substrate for Propionibacterium acnes, which metabolizes sebum into proinflammatory free fatty acids, resulting in the formation of comedones, papules, and pustules.5 Our patient did not exhibit comedonal lesions on the face and lacked a seborrheic complexion, hence diminishing the likelihood of acne vulgaris.
Tinea corporis is another intensely pruritic condition, especially in areas subjected to prolonged use of topical immunosuppressants. It is caused by dermatophyte fungi and typically manifests as erythematous pruritic patches, often presenting as ring-shaped lesions with active margins and sometimes accompanied by scaling.6 While long-term use of immunosuppressants may be a risk factor for fungal infections and increase the probability of tinea corporis, our patient’s presentation of papules and pustules without a ring-shaped configuration or scaling diminished the likelihood of tinea corporis.
Our patient represents an intriguing case of an eruptive form of demodicosis induced by long-term intermittent and inconsistent application of topical immunosuppressive agents. Demodicosis encompasses a spectrum of clinical presentations, including pityriasis folliculorum, rosacealike, folliculitislike, and perioral dermatitis–like forms.1 It is prone to misdiagnosis, as it is clinically similar to other conditions, such as acne, rosacea, or bacterial folliculitis, and it also is susceptible to missed diagnosis. Demodicosis tends to erupt in immunocompromised individuals, and the use of topical immunosuppressive and corticosteroid medications can exacerbate Demodex activity. Dermatologists should be aware that demodicosis is not a rare skin disorder, and timely identification and diagnosis can reduce the incidence of disease and improve quality of life for affected patients. Conversely, the consequences of misdiagnosis can be severe, with inappropriate treatment potentially exacerbating the condition.
- Paichitrojjana A. Demodex: the worst enemies are the ones that used to be friends. Dermatol Reports. 2022;14:9339. doi:10.4081 /dr.2022.9339
- Del RJ, Baldwin H, Bhatia N, et al. A review of the diagnostic and therapeutic gaps in rosacea management: consensus opinion. Dermatol Ther (Heidelb). 2024;14:271-284. doi:10.1007/s13555-023-01087-8
- Powell FC. The histopathology of rosacea: ‘where’s the beef?’ Dermatology. 2004;209:173-174. doi:10.1159/000079884
- Helou W, Avitan-Hersh E, Bergman R. Demodex folliculitis of the scalp: clinicopathological study of an uncommon entity. Am J Dermatopathol. 2016;38:658-663. doi:10.1097/DAD.0000000000000512
- Laureano AC, Schwartz RA, Cohen PJ. Facial bacterial infections: folliculitis. Clin Dermatol. 2014;32:711-714. doi:10.1016 /j.clindermatol.2014.02.009
- Leung AK, Lam JM, Leong KF, et al. Tinea corporis: an updated review. Drugs Context. 2020;9. doi:10.7573/dic.2020-5-6
- Paichitrojjana A. Demodex: the worst enemies are the ones that used to be friends. Dermatol Reports. 2022;14:9339. doi:10.4081 /dr.2022.9339
- Del RJ, Baldwin H, Bhatia N, et al. A review of the diagnostic and therapeutic gaps in rosacea management: consensus opinion. Dermatol Ther (Heidelb). 2024;14:271-284. doi:10.1007/s13555-023-01087-8
- Powell FC. The histopathology of rosacea: ‘where’s the beef?’ Dermatology. 2004;209:173-174. doi:10.1159/000079884
- Helou W, Avitan-Hersh E, Bergman R. Demodex folliculitis of the scalp: clinicopathological study of an uncommon entity. Am J Dermatopathol. 2016;38:658-663. doi:10.1097/DAD.0000000000000512
- Laureano AC, Schwartz RA, Cohen PJ. Facial bacterial infections: folliculitis. Clin Dermatol. 2014;32:711-714. doi:10.1016 /j.clindermatol.2014.02.009
- Leung AK, Lam JM, Leong KF, et al. Tinea corporis: an updated review. Drugs Context. 2020;9. doi:10.7573/dic.2020-5-6
Multiple Papules and Pustules on the Face and Neck
Multiple Papules and Pustules on the Face and Neck
A 26-year-old woman presented to our clinic with multiple papules and pustules on the face and neck. One year prior, the patient had developed a pruritic rash on the face after using a new over-the-counter skin care product. An outside physician had diagnosed the rash as contact dermatitis and prescribed tacrolimus cream 0.1%. Initially, the patient noted improvement, but the rash recurred intermittently over the next year. She continued using the cream, but 2 months prior to the current presentation, the patient developed more papules and pustules on the face, prompting further evaluation.
Physical examination at the current presentation revealed widespread papules and pustules on the face and neck. Due to the patient’s aesthetic concerns, a more invasive biopsy was avoided, and purulent fluid from the lesions was collected for microscopic examination.
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).
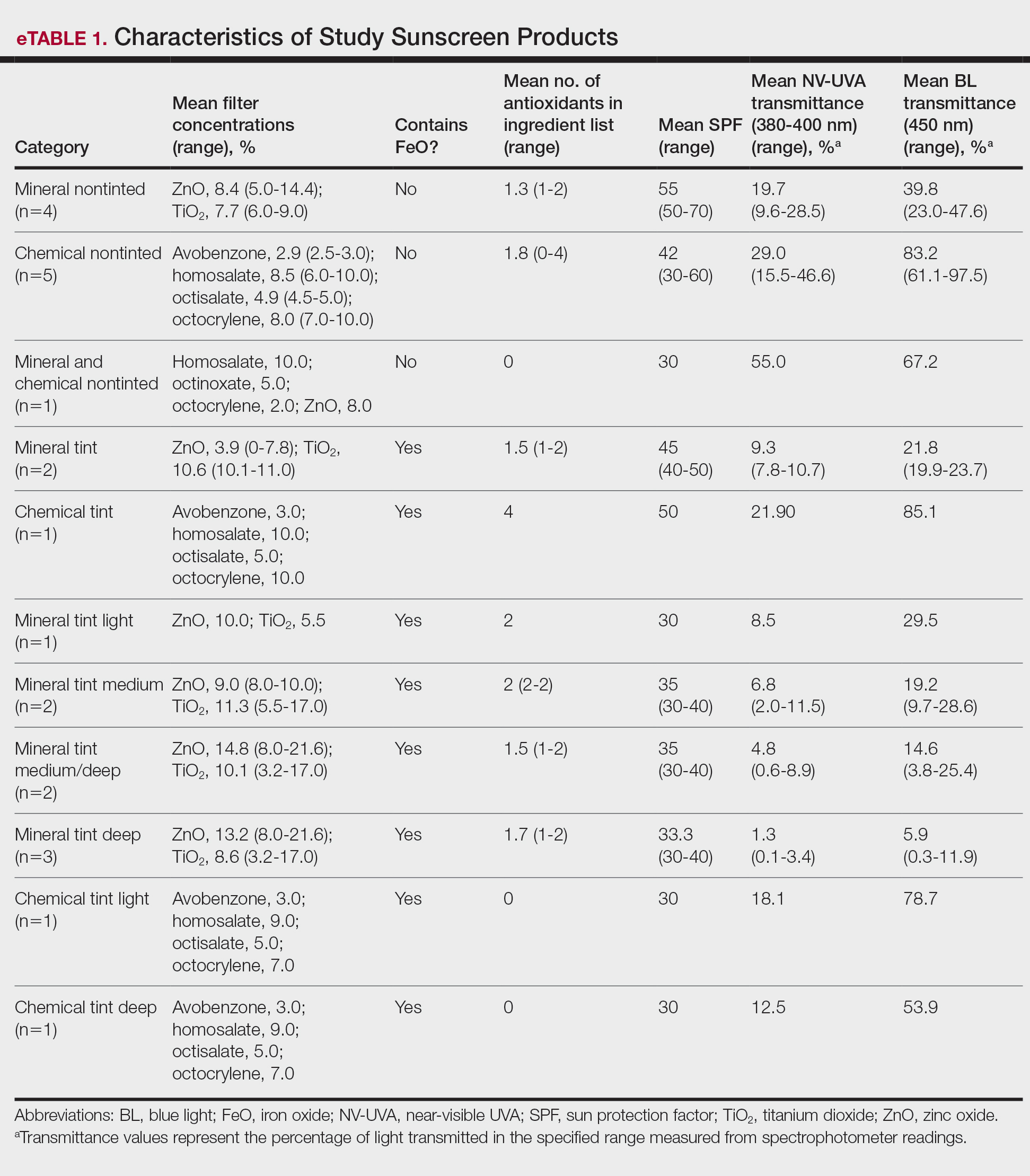
The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).
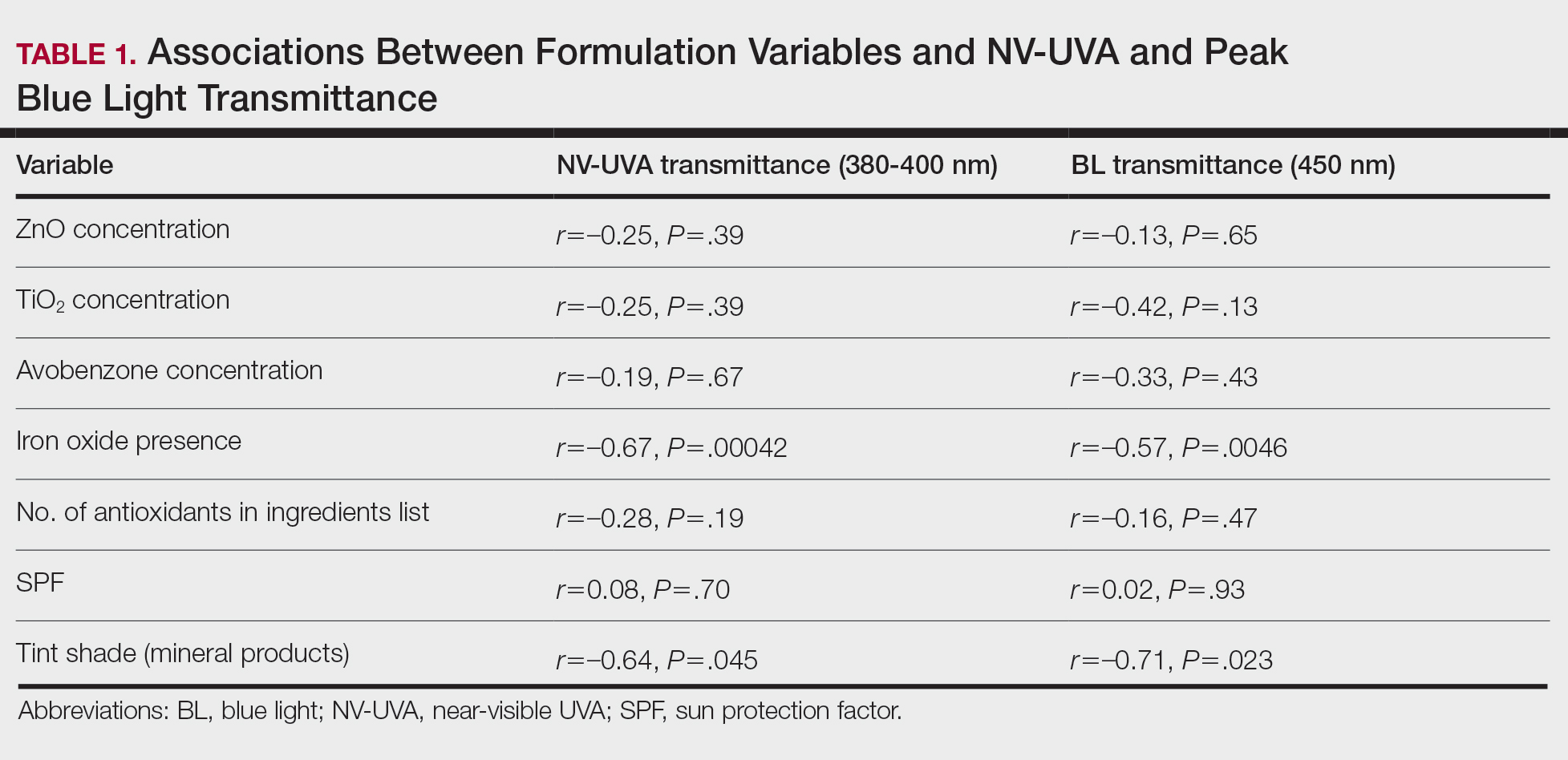
Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).
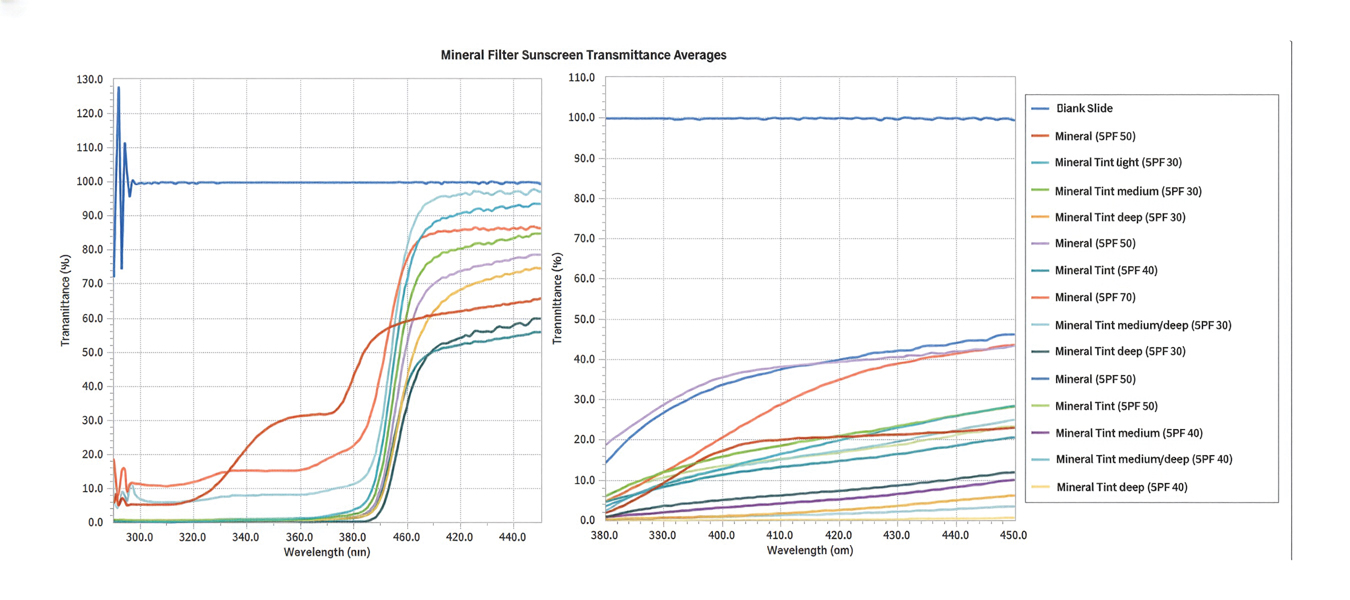
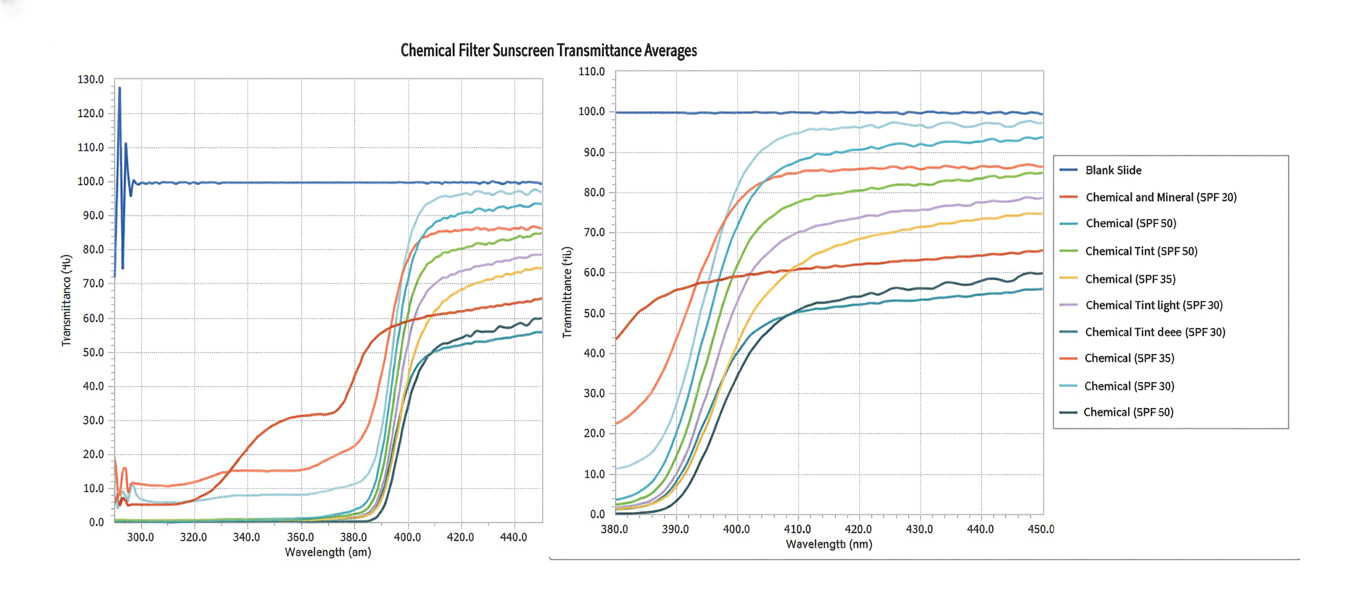
After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).
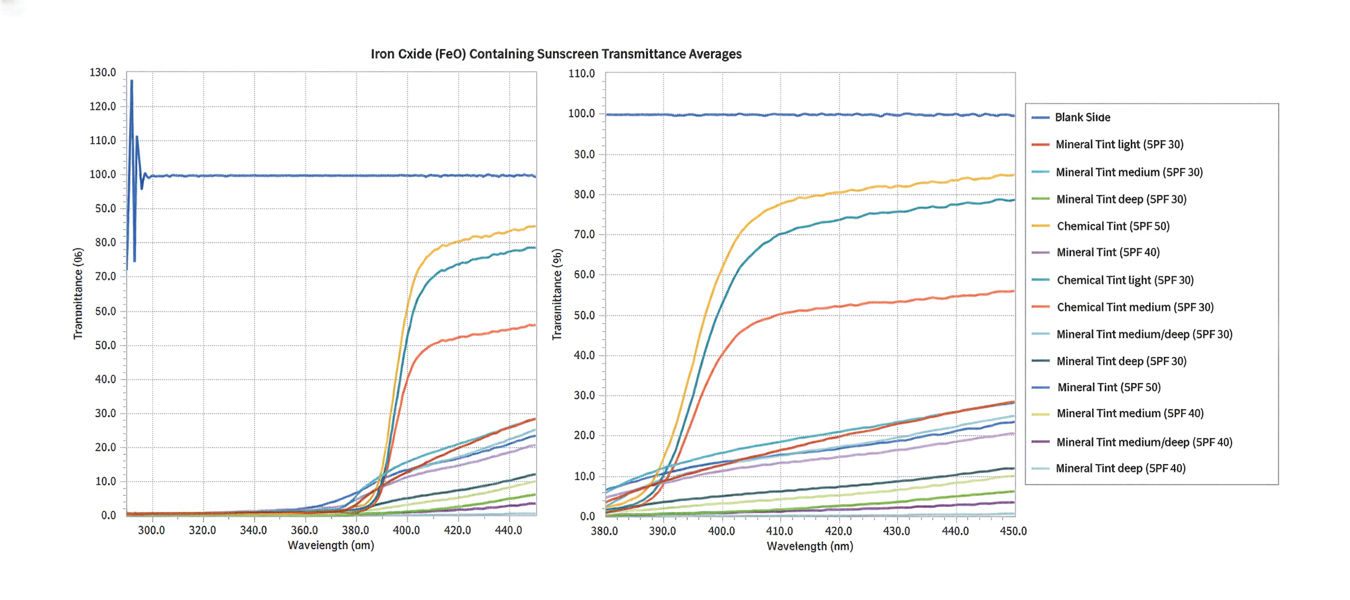
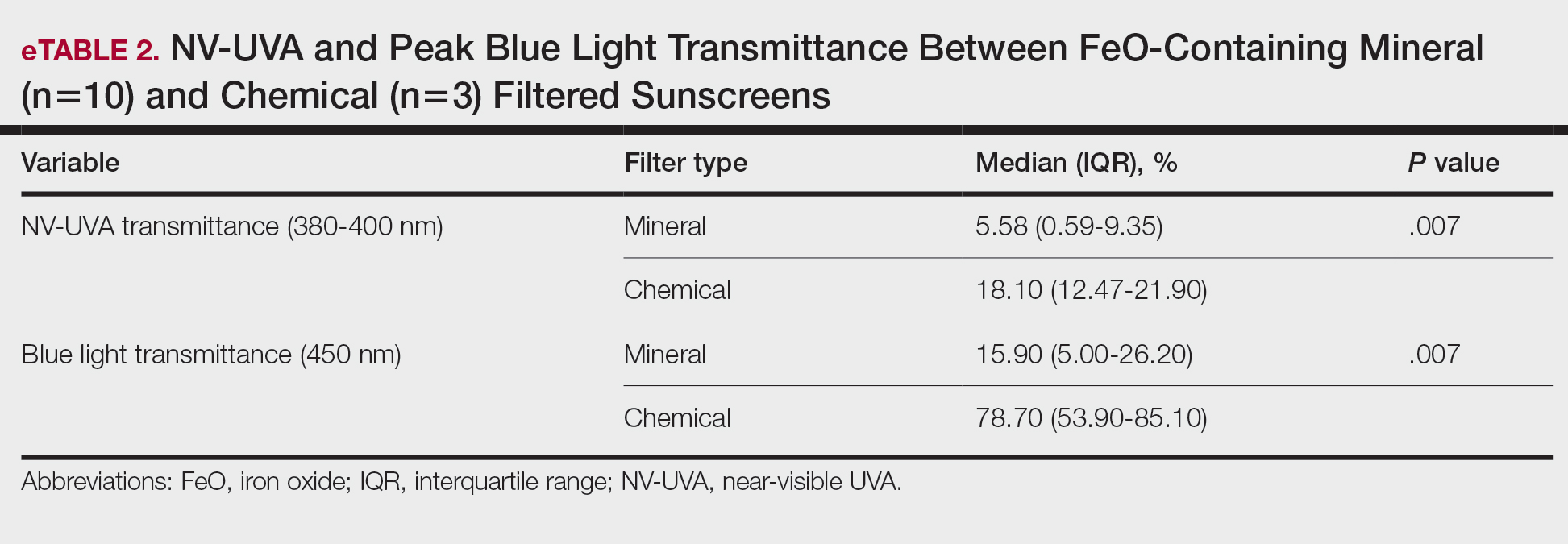
Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).

The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).

Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).


After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).


Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).

The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).

Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).


After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).


Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
PRACTICE POINTS
- There is substantial variability in spectral attenuation among tinted sunscreens, highlighting the need for standardized pigment metrics and evidence-based photoprotection guidance for individuals with skin of color (SOC).
- Broad-spectrum labeling, sun protection factor values, UV filter type (mineral vs chemical), and antioxidant inclusion do not reliably predict protection against pigment-inducing UV and visible light wavelengths for individuals with SOC.
- Iron oxide–containing tinted sunscreens demonstrate lower near-visible UVA and blue light transmittance than nontinted formulations, with greater attenuation in mineral vs chemical products. Both pigment and filter type should inform photoprotection recommendations in individuals with SOC.
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3
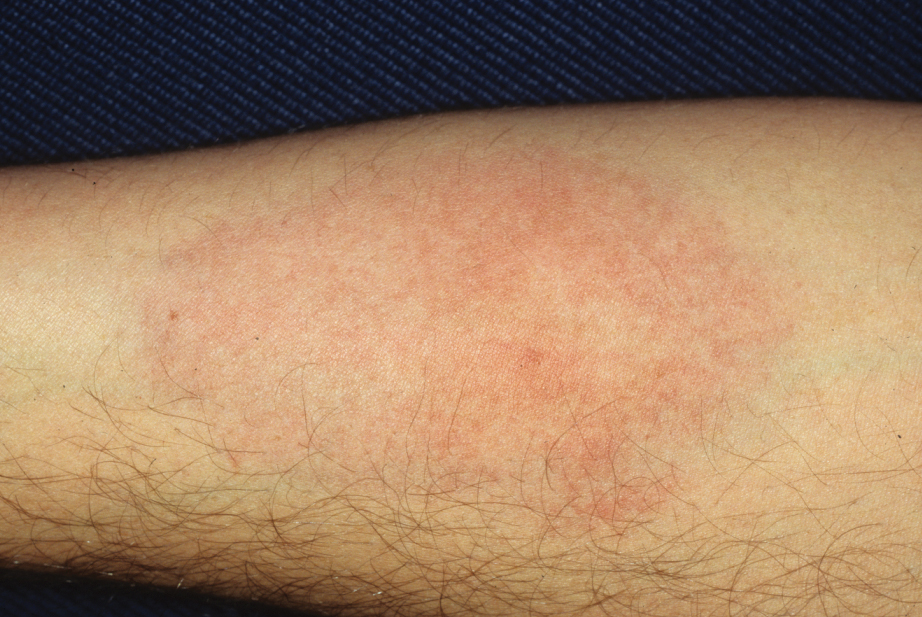
Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3

Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3

Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Practice Points
- Triatomine bugs, commonly known as kissing bugs, are widespread, especially in warmer climates, and their geographic range is expanding.
- The Romaña sign, characterized by unilateral swelling of the eyelid, is common in triatomine bites.
- Triatomine bugs are the primary vector for transmission of the parasite Trypanosoma cruzi, the causative agent of Chagas disease.
- In recent years, T cruzi has been detected in triatomine reduviids in suburban areas of the southwestern United States.
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).
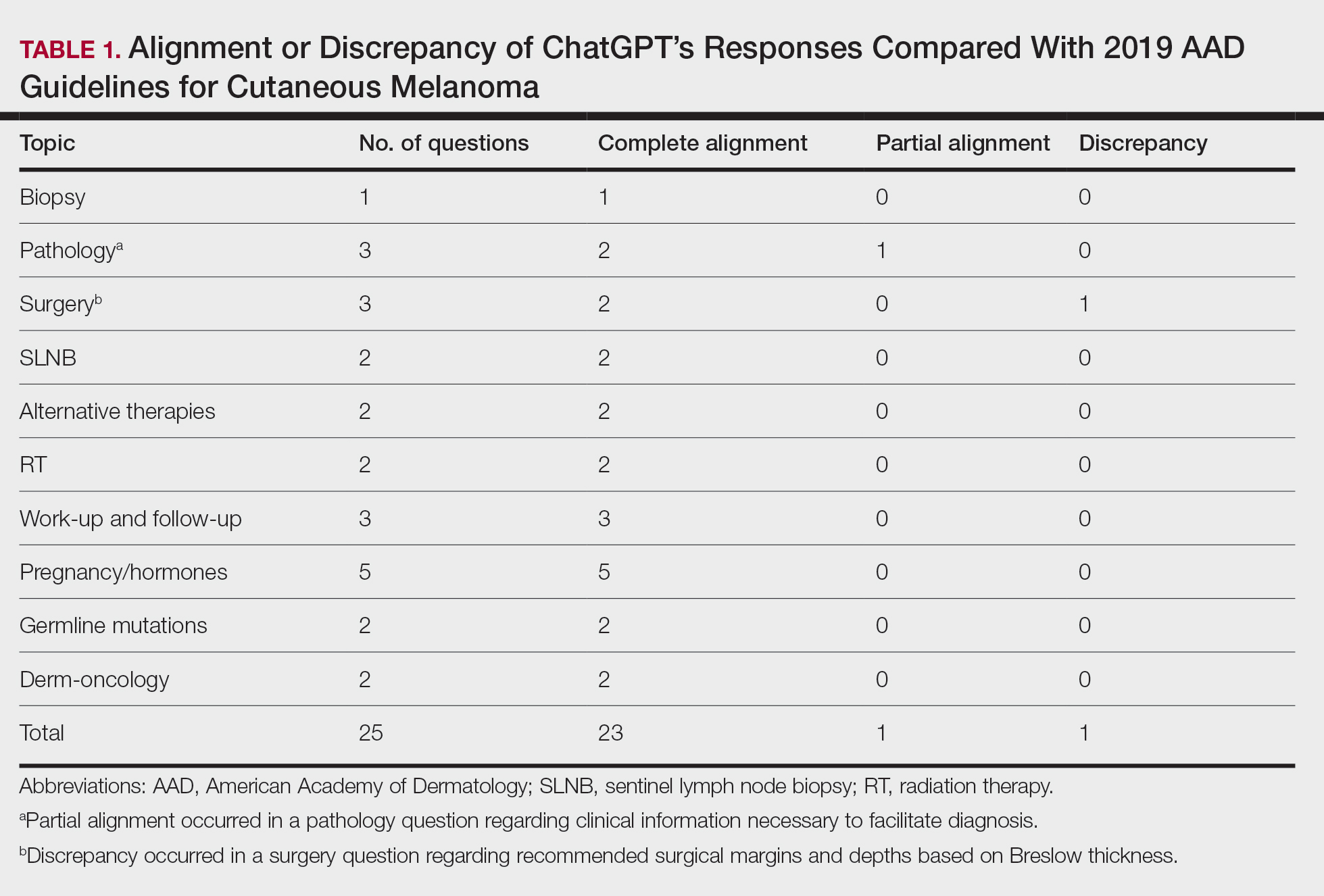

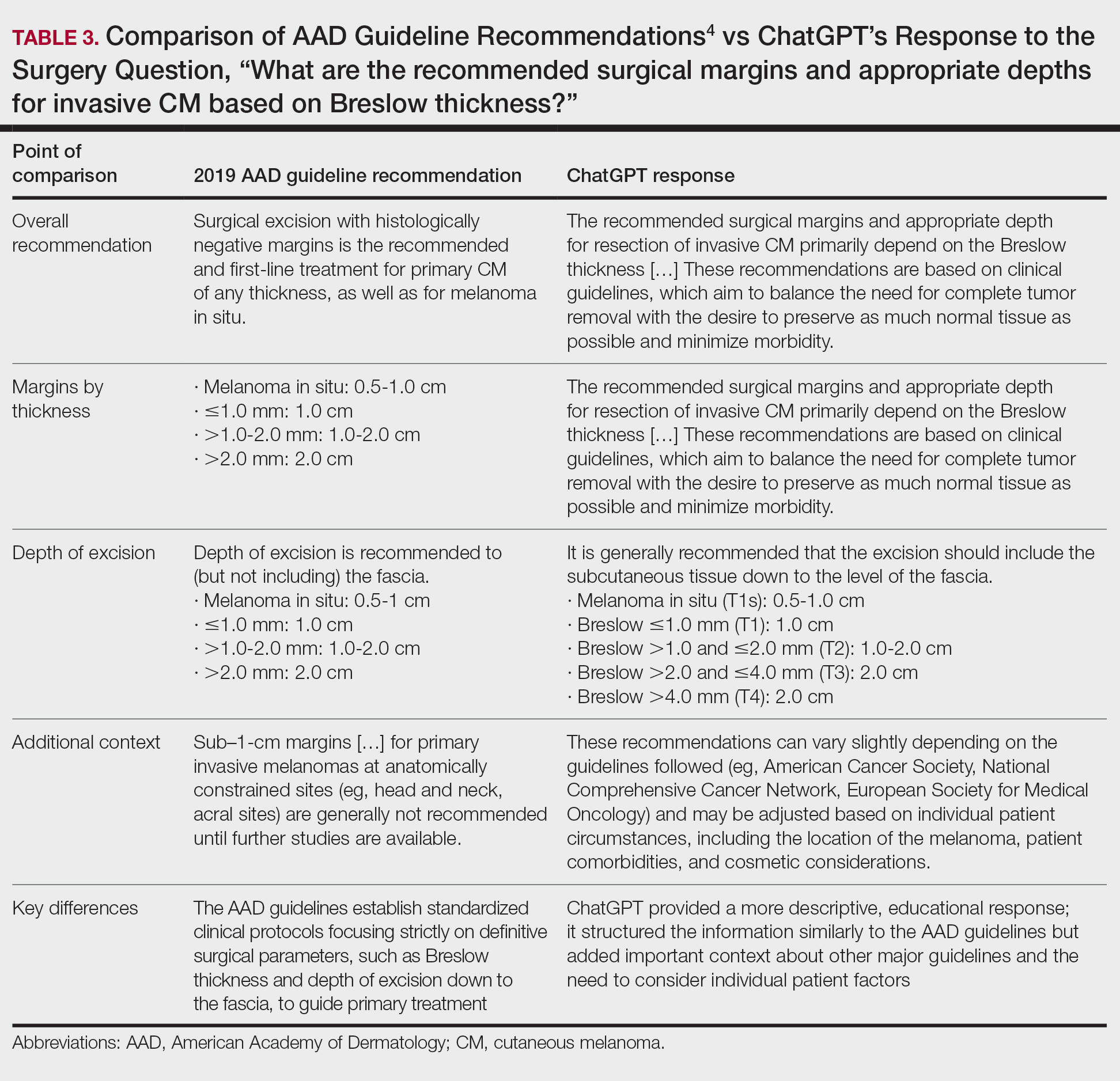
Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).



Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).



Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
PRACTICE POINTS
- ChatGPT provides structured, educational-style responses with broad contextual detail but may omit key clinical nuances such as specific surgical considerations, including staged excision or Mohs micrographic surgery for melanoma in situ.
- Large language models should be viewed as a tool to supplement expert clinical judgment and established guidelines rather than as a standalone replacement for dermatologic decision-making.
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Practice Points
- Military service members face increased UV exposure from occupational and deployment conditions, but sunscreen use is limited by logistical, educational, and behavioral barriers.
- Sunscreen availability in military exchanges partially meets dermatologic guidelines, highlighting opportunities to expand tailored options and integrate sun protection into operational readiness policy.