User login
Zeroing in on the cause of your patient's facial pain
› Advise patients who have a temporomandibular disorder that in addition to taking their medication as prescribed, they should limit activities that require moving their jaw, modify their diet, and minimize stress; they may require physical therapy and therapeutic exercises. C
› Consider prescribing a tricyclic antidepressant for patients with persistent idiopathic facial pain. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Facial pain is a common complaint: Up to 22% of adults in the United States experience orofacial pain during any 6-month period.1 Yet this type of pain can be difficult to diagnose due to the many structures of the face and mouth, pain referral patterns, and insufficient diagnostic tools.
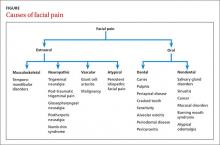
Specifically, extraoral facial pain can be the result of temporomandibular disorders, neuropathic disorders, vascular disorders, or atypical causes, whereas facial pain stemming from inside the mouth can have a dental or nondental cause (FIGURE). Overlapping characteristics can make it difficult to distinguish these disorders. To help you to better diagnose and manage facial pain, we describe the most common causes and underlying pathological processes.
Extraoral facial pain
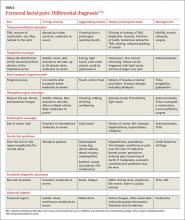
Extraoral pain refers to the pain that occurs on the face outside of the oral cavity. The TABLE2-15 summarizes the site, timing and severity, aggravating factors, history and exam findings, and management of several common causes of extraoral facial pain.
Musculoskeletal pain
Temporomandibular disorders (TMD) are a broad group of problems that affect the temporomandibular joint (TMJ), muscles of mastication, and/or associated bony and soft tissue structures.6 They may occur secondary to malocclusion, traumatic injuries, oral parafunctional habits (eg, bruxism), hormonal influences, or psychogenic factors.6 TMD is more prevalent in women, with a peak occurrence between ages 20 and 40 years.6,8
TMD can be articular (intracapsular) or nonarticular (extracapsular). Nonarticular disorders (>50% of TMD) usually affect the muscles of mastication and include chronic conditions such as fibromyalgia, muscle strain, and myopathies.8 Muscle-related pain and dysfunction are believed to arise from parafunctional habits such as bruxism or clenching. Articular disorders include synovitis/capsulitis, joint effusion, trauma/fracture, internal derangement (disturbance in the normal anatomic relationship between the disc and condyle), arthritis, and neoplasm.16
What you’ll see. Orofacial pain (usually dull and located in the preauricular region), joint noise, and restricted jaw function are key signs and symptoms of TMD. Exacerbation of pain with mandibular functions (eg, chewing, yawning, or swallowing) is a pathognomonic sign. Joint sounds such as clicking or crepitus are common. In most cases, crepitus correlates with osteoarthritis.6 Nonspecific TMD symptoms include headache, earache, insomnia, tinnitus, and neck and shoulder pain.6
The gold standard of diagnosis of TMD consists of taking a detailed history, evaluating the patient’s head and neck, and conducting a general physical examination and behavioral/psychological assessment.17 Imaging of the TMJ and associated structures is essential.17
Treatment. Nonsteroidal anti-inflammatory drugs, opioids, muscle relaxants, antidepressants, anticonvulsants, anxiolytics, and corticosteroids are options for treating TMD.6,8 Isometric jaw exercises, maxillomandibular appliances, and physical therapy are valuable adjuncts for pain relief. Advise patients to establish a self-care routine to reduce TMJ pain that might include changing their head posture or sleeping position, and limiting activities that require using their jaw, such as clenching, bruxism, and excessive gum chewing. Some patients may need to adopt a non-chewing diet that consists of liquid or pureed food. Massage and moist heat can help relax muscles of mastication and improve range of motion.
Approximately 5% of patients with TMD undergo surgery, typically simple arthrocentesis, arthroscopy, arthrotomy, or modified condylotomy.6 Total joint replacement is indicated only for patients with severely damaged joints with end-stage disease when all other conservative treatments have failed. Joint replacement primarily restores form and function; pain relief is a secondary benefit.8
Neuropathic pain
Trigeminal neuralgia (TN) is sudden, usually unilateral, severe, brief, stabbing, recurrent episodes of pain in the distribution of one or more branches of the trigeminal nerve.9 It most commonly presents in the lower 2 branches of the trigeminal nerve and usually is caused by compression of the trigeminal nerve root by vascular or nonvascular causes.4 The pain is severe and can profoundly impact a patient’s quality of life.
TN attacks typically last from a few seconds to up to 2 minutes. As many as 30 attacks can occur daily, with refractory periods between attacks. After the initial attack, individuals are left with a residual dull or burning pain. TN can be triggered by face washing, teeth brushing, speaking, eating, shaving, or cold wind.4
Diagnosis can be tricky because more than half of patients with TN experience less severe pain after the main sharp attack; this presentation is called TN type II.7 A detailed patient history and careful evaluation can help identify patients with TN type II. TN can be misdiagnosed as TMD, especially if it presents unilaterally.15
Treatment. Anticonvulsants are the primary medications used to treat TN.
Post-traumatic trigeminal pain is usually the result of an injury or dental procedure, such as facial trauma, tooth extraction, root canal, or dental implants.12,18,19 Nerve injury is assumed to be the cause. This type of pain can start within 3 to 6 months of a trauma. It is located in the trigeminal area and patients describe it as burning, tingling and, at times, sharp.15 Patients who have sustained injury to the lingual or inferior alveolar nerves have reported feeling “pins and needles.”12
Common triggers include temperature changes or simple touch. Not all injuries result in pain; some patients may have only sensory impairment15 or sensory deficits such as allodynia or hypoesthesia.
Treatment. The first line of treatment for post-traumatic trigeminal pain is tricyclic antidepressants (TCAs) followed by pregabalin or gabapentin.14
Glossopharyngeal neuralgia (GN) is similar in presentation to TN but is much rarer.15 GN pain occurs deep in the throat, ear, or posterior tongue.15 When the pain occurs in the inner ear, GN can be misdiagnosed as TMD. In most cases, no cause of GN can be determined.
Patients describe GN pain as shooting, sharp, and electrical shock-like, lasting from seconds to minutes, with recurrent attacks throughout the day. Like TN, GN can present as episodes of attacks that last weeks to months. Triggers include chewing, drinking, swallowing, and talking, as well as light touch.13,15 Some patients with GN experience syncope due to the anatomical proximity of the vagus nerve.14
Treatment. Anticonvulsants are the first-line treatment for GN. Local anesthetics or surgery can be considered for patients who don’t improve after medical therapy.15
Postherpetic neuralgia (PHN) can cause facial pain when the characteristic vesicular rash of the varicella zoster virus (shingles) occurs on the face. PHN usually affects the first division of trigeminal nerve, but the second and third divisions can be affected as well.13
What you’ll see. The acute phase of PHN begins a few days before the initial rash has resolved and can last up to a month after. A new pain may begin one to 6 months after the initial rash has healed.20 This pain, which patients often describe as sharp, stabbing, or burning, can be constant or intermittent. Dysesthesia, hypoesthesia, and allodynia may also occur within the affected dermatome.
PHN is usually diagnosed based on the patient’s history and clinical presentation. However, direct fluorescent antibody stain, viral culture, or polymerase chain reaction performed on vesicular fluid from a herpetic lesion during the initial rash are the laboratory tests of choice if confirmation is needed.
Treatment. PHN is managed with anticonvulsants and TCAs.
Numb chin syndrome (NCS) is characterized by hypoesthesia, paresthesia, thermalgesic anesthesia, or pain over the chin in the region supplied by the mental nerve, a terminal branch of the mandibular division of the trigeminal nerve.5,21,22
NCS can be caused by odontogenic conditions, such as dental abscess, dental anesthesia, dental trauma, or osteomyelitis; systemic conditions such as amyloidosis, sickle cell disease, sarcoidosis, multiple sclerosis, human immunodeficiency virus, or diabetes; or malignancies such as lymphoma, leukemia, breast cancer, lung cancer, prostate cancer, or head and neck cancers.21 In one study of patients with NCS, cancer was the cause of the condition in 89% of patients.22
What you’ll see. NCS is characterized by numbness of the skin in the lower lip, chin and mucous membrane inside the lip that extends to the midline.5 Depending upon the etiology, patients may present with percussion-induced pain, loosening of teeth, sequestra, and mobility of fractured segments. Patients with metastatic malignancy may develop constitutional symptoms.
Making the diagnosis. Panoramic radiography is a useful starting point. If possible, a computerized tomography scan of the head and neck should also be done. Nuclear bone scintigraphy (bone scanning) may help identify bone disease such as osteomyelitis. A biopsy may be needed if a mass lesion is present.
Treatment. In NCS that is the result of a dental etiology, the prognosis usually is good. For example, NCS that is the result of an abscess usually resolves after the abscess is drained. However, if NCS is caused by metastasis, the prognosis is grim; the average length of survival after diagnosis is approximately 5 months if NCS is caused by mandibular metastasis and 12 months if leptomeningeal metastasis is present. Treatment does little to affect the outcome in these cases.21,22
Atypical pain
Persistent idiopathic facial pain (PIFP), previously known as atypical facial pain, is a persistent facial pain that does not have the classical characteristics of cranial neuralgias and for which there is no obvious cause.2,10,23 PIFP is not triggered by any of the factors that typically precipitate neuralgias.2 The onset may be spontaneous or associated with dental intervention or facial injury, but it usually does not have a demonstrable local cause.24,25
Neuropathic mechanisms that might be at work in PIFP include nociceptor sensitization, phenotypic changes and ectopic activity from the nociceptors, central sensitization possibly maintained by ongoing activity from initially damaged peripheral tissues, sympathetic abnormal activity, alteration of segmental inhibitory control, or hyperactivity or hypoactivity of descending controls.2
PIFP is most frequently reported in women in their 40s and 50s.25 The history of a patient with PIFP often include mood disorders, chronic pain, or poor coping skills.14 Patients complain of a steady, unilateral, poorly localized pain that is deep, constant, aching, pulling, or crushing. It is usually present all day, every day. The constancy of the pain is its distinguishing feature. In the beginning, this pain may be in a limited area on one side of the face, usually the nasolabial folds or the angle of the mandible. Later, it may affect both sides of the face and extend to the neck and upper limbs.23,24 Most patients with PIFP report other symptoms, including headache, neck and backache, dermatitis, pruritus, irritable bowel, and dysfunctional uterine bleeding.26
Making the diagnosis. A targeted history and accurate clinical examination are essential.2,10 Although there are no formal diagnostic criteria, a patient can be assumed to have PIFP if:2,10
• There is pain in the face for most of the day or all day, every day.
• Initially, the pain may be confined to a portion of the face, but it is poorly localized and deep.
• The pain is not associated with other physical signs or loss of sensation.
• Imaging does not reveal an obvious anatomic or structural cause.
Treatment. Treatment of PIFP can be difficult and unsatisfactory.23 Counseling to educate patients about the chronic and nonmalignant nature of the illness is the mainstay of treatment, followed by pharmacotherapy.23 TCAs have shown a moderate effect in several trials. Gabapentin, topiramate, carbamazepine, and pregabalin also have shown limited to modest benefit in some patients. Surgical therapies appear to be of little or no use.23 Experimental treatments such as pulsed radiofrequency, low-energy level diode laser have shown success in small studies.10,23
Vascular pain
Giant cell arteritis (GCA) is a systemic, chronic vasculitis involving the large and medium-sized vessels, mainly the extracranial branches of the carotid artery.6,11 It predominantly affects people older than age 50 and is more common among women and those of Scandinavian ethnicity.27
The cause of GCA is unclear. Genetic predisposition linked to humoral and cellmediated immunity is believed to play a role.28 Familial aggregation and predominance of the HLA-DR4 allele has been reported in patients with GCA.6
What you’ll see. The most common signs and symptoms of GCA are temporal headache (seen in two-thirds of patients), jaw claudication and tenderness, and swelling of the temporal artery.6,11 The headache of GCA usually is unilateral, severe, boring or lancinating, and localized to the temporal or occipital regions of the scalp.6 Other orofacial manifestations include trismus, throat pain that develops while chewing, changes in tongue sensation and tongue claudication, tooth pain, dysphagia, dysarthria, submandibular mass, lip and chin numbness, macroglossia, glossitis, lip and tongue necrosis, and facial swelling.11
Visual symptoms include diplopia, ptosis, and possibly blindness if treatment is not instituted at first suspicion. Ocular symptoms result from anterior ischemic optic neuropathy, posterior ischemic optic neuropathy, or central retinal or cilioretinal artery occlusion.6,28 Patients have also reported low-grade fever, asthenia, anorexia, weight loss, and generalized aches.11,28
Making the diagnosis. Arterial biopsy is the gold standard for diagnosis of GCA. It is usually performed on the temporal artery and is positive in 80% to 95% of people with the condition.28 Other useful lab tests include erythrocyte sedimentation rate (ESR; elevated), white blood cell count (mildly elevated), and C-reactive protein (elevated).
Treatment. Prednisone is used to treat GCA, in initial doses ranging from 30 to 80 mg. A maintenance dose may be required for up to 2 years, with close follow-up and periodic ESR measurements.28
Malignancy is a rare cause of facial pain. The pain may be due to metastasis of extracranial bony or soft tissue as it compresses cervical and cranial nerves.3 Lung cancer can cause referred pain in the periauricular region by compressing the vagus nerve, and this pain can be misdiagnosed as dental pain, atypical facial pain, TMD, or TN.3,29 The facial pain of lung cancer is unilateral and on the same side as the lung neoplasm, and commonly is referred to the jaw, ear, or temporal region. While many patients have continuous pain, some report intermittent pain or pain that lasts for hours.3 Facial pain caused by a malignancy is differentiated from other sources of facial pain by the presence of associated symptoms such as weight loss, cough, and hemoptysis.
Treatment. Treatment can include radiation or chemotherapy.29
The mouth is often the source of lower facial pain
Pain in the oral cavity is the most common cause of pain in the lower face.15 Intraoral pain usually is caused by disease in the following structures:
1. Dentition (eg, caries, dentin sensitivity, pulpal disease)
2. Periodontium (eg, gingivitis, acute or chronic periodontal disease, sensitivity related to gum recession, alveolar bone pathology)
3. Other soft and hard tissues, such as the palate, floor of mouth, buccal mucosa, non-tooth supporting bone, and tongue (eg, mucosal diseases, neoplasms, pain related to parafunction or trauma).
Rarely, intraoral pain may be referred. For example, myofascial pain might cause diffuse tooth pain.30
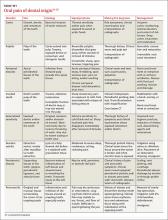
See TABLE W131-35 at the end of this article for a summary of the etiology, signs/symptoms, diagnosis, and management of these and other dental causes of oral facial pain.
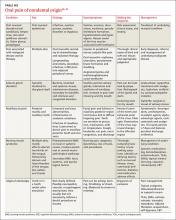
Nondental causes of oral facial pain can be associated with oral mucosal disorders, malignant disease and its therapy, salivary gland disorders, maxillary sinusitis, burning mouth syndrome, or atypical odontalgia. See TABLE W236-41 for a more detailed description of these conditions.
CORRESPONDENCE
Tamer H. Said, MD, MetroHealth Medical Center, 2500 MetroHealth Drive, Cleveland, Ohio 44109; [email protected]
1. Lipton JA, Ship JA, Larach-Robinson D. Estimated prevalence and distribution of reported orofacial pain in the United States. J Am Dent Assoc. 1993;124:115-1121.
2. Agostoni E, Frigerio R, Santoro P. Atypical facial pain: clinical considerations and differential diagnosis. Neurol Sci. 2005;26:S71-S74.
3. Bajwa Z, Ho C, Khan S, et al. Overview of craniofacial pain. UpTo-Date Web site. Available at: http://www.uptodate.com/contents/overview-of-craniofacial-pain. Accessed January 28, 2015.
4. Bendtsen L, Birk S, Kasch H, et al. Reference programme: Diagnosis and treatment of headache disorders and facial pain. Danish Headache Society, 2nd Edition, 2012. J Headache Pain. 2012;13:S1-S29.
5. Divya KS, Moran NA, Atkin PA. Numb chin syndrome: a case series and discussion. Br Dent J. 2010;208:157-160.
6. Kapur N, Kamel IR, Herlich A. Oral and craniofacial pain: diagnosis, pathophysiology, and treatment. Int Anesthesiol Clin. 2003;41:115-150.
7. Limonadi FM, McCartney S, Burchiel KJ. Design of an artificial neural network for diagnosis of facial pain syndromes. Stereotact Funct Neurosurg. 2006;84:212-220.
8. Liu F, Steinkeler A. Epidemiology, diagnosis, and treatment of temporomandibular disorders. Dent Clin North Am. 2013;57:465-479.
9. Merskey H, Bogduk N (eds). Classification of Chronic Pain. Descriptors of Chronic Pain Syndromes and Definition of Pain Terms, 2nd ed. Seattle, WA: International Association for the Study of Pain Press; 1994.
10. Nguyen CT, Wang MB. Complementary and integrative treatments: atypical facial pain. Otolaryngol Clin North Am. 2013;46:367-382.
11. Reiter S, Winocur E, Goldsmith C, et al. Giant cell arteritis misdiagnosed as temporomandibular disorder: a case report and review of the literature. J Orofac Pain. 2009;23:360-365.
12. Renton T, Adey-Viscuso D, Meechan JG, et al. Trigeminal nerve injuries in relation to local anaesthesia in mandibular injections. Br Dent J. 2010;209:E15.
13. Shephard MK, Macgregor EA, Zakrzewska JM. Orofacial pain: a guide for the headache physician. Headache. 2014;54:22-39.
14. Zakrzewska JM. Differential diagnosis of facial pain and guidelines for management. Br J Anaesth. 2013;111:95-104.
15. Zakrzewska JM. Multi-dimensionality of chronic pain of the oral cavity and face. J Headache Pain. 2013;14:37.
16. Herb K, Cho S, Stiles MA. Temporomandibular joint pain and dysfunction. Curr Pain Headache Rep. 2006;10:408-414.
17. American Society of Temporomandibular Joint Surgeons. Guidelines for diagnosis and management of disorders involving the temporomandibular joint and related musculoskeletal structures. Cranio. 2003;21:68-76.
18. Benoliel R, Zadik Y, Eliav E, et al. Peripheral painful traumatic trigeminal neuropathy: clinical features in 91 cases and proposal of novel diagnostic criteria. J Orofac Pain. 2012;26:49-58.
19. Brooke RI. Atypical odontalgia. A report of twenty-two cases. Oral Surg Oral Med Oral Pathol. 1980;49:196-199.
20. Bouhassira D, Chassany O, Gaillat J, et al. Patient perspective on herpes zoster and its complications: an observational prospective study in patients aged over 50 years in general practice. Pain. 2012;153:342-349.
21. Baskaran RK, Krishnamoorthy, Smith M. Numb chin syndrome—a reflection of systemic malignancy. World J Surg Oncol. 2006;4:52.
22. Lata J, Kumar P. Numb chin syndrome: a case report and review of the literature. Indian J Dent Res. 2010;21:135-137.
23. Cornelissen P, van Kleef M, Mekhail N, et al. Evidence-based interventional pain medicine according to clinical diagnoses. 3. Persistent idiopathic facial pain. Pain Pract. 2009;9:443-448.
24. Didier H, Marchetti C, Borromeo G, et al. Persistent idiopathic facial pain: multidisciplinary approach and assumption of comorbidity. Neurol Sci. 2010;31:S189-S195.
25. Klasser G. Management of persistent idiopathic facial pain. J Can Dent Assoc. 2013;79:d71.
26. Abiko Y, Matsuoka H, Chiba I, et al. Current evidence on atypical odontalgia: diagnosis and clinical management. Int J Dent. 2012;2012:518548.
27. Sheldon CA, White VA, Holland SP. Giant cell arteritis presenting with bilateral loss of vision and jaw pain: reminder of a potentially devastating condition. J Can Dent Assoc. 2011;77:b55.
28. Rockey JG, Anand R. Tongue necrosis secondary to temporal arteritis: a case report and literature review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;94:471-473.
29. Sarlani E, Schwartz AH, Greenspan JD, et al. Facial pain as first manifestation of lung cancer: a case of lung cancer-related cluster headache and a review of the literature. J Orofac Pain. 2003;17:262-267.
30. Kumar A, Brennan MT. Differential diagnosis of orofacial pain and temporomandibular disorder. Dent Clin North Am. 2013;57:419-428.
31. Laudenbach JM, Simon Z. Common dental and periodontal diseases: evaluation and management. Med Clin North Am. 2014;98:1239-1260.
32. Napeñas JJ. Intraoral pain disorders. Dent Clin North Am. 2013;57:429-447.
33. Vickers ER, Zakrzewska JM. Dental causes of orofacial pain. In: Orofacial Pain. Zakrzewska JM, ed. Oxford, UK: Oxford University Press; 2009:69-81.
34. Pierse JE, Dym H, Clarkson E. Diagnosis and management of common postextraction complications. Dent Clin North Am. 2012;56:75-93.
35. Renton T. Dental (odontogenic) pain. Br J Pain. 2011;5:2-7.
36. Yatani H, Komiyama O, Matsuka Y, et al. Systematic review and recommendations for nonodontogenic toothache. J Oral Rehabil. 2014;41:843-852.
37. Klasser GD, Fischer DJ, Epstein JB. Burning mouth syndrome: recognition, understanding, and management. Oral Maxillofac Surg Clin North Am. 2008;20:255-271.
38. Balasubramaniam R, Turner LN, Fischer D, et al. Non-odontogenic toothache revisited. Open Journal of Stomatology. 2011;1:92-102.
39. Patton LL, Siegel MA, Benoliel R, et al. Management of burning mouth syndrome: systematic review and management recommendations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:S39.e1-e13.
40. Cascarini L, McGurk M. Epidemiology of salivary gland infections. Oral Maxillofac Surg Clin North Am. 2009;21:353-357.
41. Hegarty AM, Zakrzewska JM. Differential diagnosis for orofacial pain, including sinusitis, TMD, trigeminal neuralgia. Dent Update. 2011;38:396-400,402-403,405-406.
› Advise patients who have a temporomandibular disorder that in addition to taking their medication as prescribed, they should limit activities that require moving their jaw, modify their diet, and minimize stress; they may require physical therapy and therapeutic exercises. C
› Consider prescribing a tricyclic antidepressant for patients with persistent idiopathic facial pain. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Facial pain is a common complaint: Up to 22% of adults in the United States experience orofacial pain during any 6-month period.1 Yet this type of pain can be difficult to diagnose due to the many structures of the face and mouth, pain referral patterns, and insufficient diagnostic tools.
Specifically, extraoral facial pain can be the result of temporomandibular disorders, neuropathic disorders, vascular disorders, or atypical causes, whereas facial pain stemming from inside the mouth can have a dental or nondental cause (FIGURE). Overlapping characteristics can make it difficult to distinguish these disorders. To help you to better diagnose and manage facial pain, we describe the most common causes and underlying pathological processes.
Extraoral facial pain
Extraoral pain refers to the pain that occurs on the face outside of the oral cavity. The TABLE2-15 summarizes the site, timing and severity, aggravating factors, history and exam findings, and management of several common causes of extraoral facial pain.
Musculoskeletal pain
Temporomandibular disorders (TMD) are a broad group of problems that affect the temporomandibular joint (TMJ), muscles of mastication, and/or associated bony and soft tissue structures.6 They may occur secondary to malocclusion, traumatic injuries, oral parafunctional habits (eg, bruxism), hormonal influences, or psychogenic factors.6 TMD is more prevalent in women, with a peak occurrence between ages 20 and 40 years.6,8
TMD can be articular (intracapsular) or nonarticular (extracapsular). Nonarticular disorders (>50% of TMD) usually affect the muscles of mastication and include chronic conditions such as fibromyalgia, muscle strain, and myopathies.8 Muscle-related pain and dysfunction are believed to arise from parafunctional habits such as bruxism or clenching. Articular disorders include synovitis/capsulitis, joint effusion, trauma/fracture, internal derangement (disturbance in the normal anatomic relationship between the disc and condyle), arthritis, and neoplasm.16
What you’ll see. Orofacial pain (usually dull and located in the preauricular region), joint noise, and restricted jaw function are key signs and symptoms of TMD. Exacerbation of pain with mandibular functions (eg, chewing, yawning, or swallowing) is a pathognomonic sign. Joint sounds such as clicking or crepitus are common. In most cases, crepitus correlates with osteoarthritis.6 Nonspecific TMD symptoms include headache, earache, insomnia, tinnitus, and neck and shoulder pain.6
The gold standard of diagnosis of TMD consists of taking a detailed history, evaluating the patient’s head and neck, and conducting a general physical examination and behavioral/psychological assessment.17 Imaging of the TMJ and associated structures is essential.17
Treatment. Nonsteroidal anti-inflammatory drugs, opioids, muscle relaxants, antidepressants, anticonvulsants, anxiolytics, and corticosteroids are options for treating TMD.6,8 Isometric jaw exercises, maxillomandibular appliances, and physical therapy are valuable adjuncts for pain relief. Advise patients to establish a self-care routine to reduce TMJ pain that might include changing their head posture or sleeping position, and limiting activities that require using their jaw, such as clenching, bruxism, and excessive gum chewing. Some patients may need to adopt a non-chewing diet that consists of liquid or pureed food. Massage and moist heat can help relax muscles of mastication and improve range of motion.
Approximately 5% of patients with TMD undergo surgery, typically simple arthrocentesis, arthroscopy, arthrotomy, or modified condylotomy.6 Total joint replacement is indicated only for patients with severely damaged joints with end-stage disease when all other conservative treatments have failed. Joint replacement primarily restores form and function; pain relief is a secondary benefit.8
Neuropathic pain
Trigeminal neuralgia (TN) is sudden, usually unilateral, severe, brief, stabbing, recurrent episodes of pain in the distribution of one or more branches of the trigeminal nerve.9 It most commonly presents in the lower 2 branches of the trigeminal nerve and usually is caused by compression of the trigeminal nerve root by vascular or nonvascular causes.4 The pain is severe and can profoundly impact a patient’s quality of life.
TN attacks typically last from a few seconds to up to 2 minutes. As many as 30 attacks can occur daily, with refractory periods between attacks. After the initial attack, individuals are left with a residual dull or burning pain. TN can be triggered by face washing, teeth brushing, speaking, eating, shaving, or cold wind.4
Diagnosis can be tricky because more than half of patients with TN experience less severe pain after the main sharp attack; this presentation is called TN type II.7 A detailed patient history and careful evaluation can help identify patients with TN type II. TN can be misdiagnosed as TMD, especially if it presents unilaterally.15
Treatment. Anticonvulsants are the primary medications used to treat TN.
Post-traumatic trigeminal pain is usually the result of an injury or dental procedure, such as facial trauma, tooth extraction, root canal, or dental implants.12,18,19 Nerve injury is assumed to be the cause. This type of pain can start within 3 to 6 months of a trauma. It is located in the trigeminal area and patients describe it as burning, tingling and, at times, sharp.15 Patients who have sustained injury to the lingual or inferior alveolar nerves have reported feeling “pins and needles.”12
Common triggers include temperature changes or simple touch. Not all injuries result in pain; some patients may have only sensory impairment15 or sensory deficits such as allodynia or hypoesthesia.
Treatment. The first line of treatment for post-traumatic trigeminal pain is tricyclic antidepressants (TCAs) followed by pregabalin or gabapentin.14
Glossopharyngeal neuralgia (GN) is similar in presentation to TN but is much rarer.15 GN pain occurs deep in the throat, ear, or posterior tongue.15 When the pain occurs in the inner ear, GN can be misdiagnosed as TMD. In most cases, no cause of GN can be determined.
Patients describe GN pain as shooting, sharp, and electrical shock-like, lasting from seconds to minutes, with recurrent attacks throughout the day. Like TN, GN can present as episodes of attacks that last weeks to months. Triggers include chewing, drinking, swallowing, and talking, as well as light touch.13,15 Some patients with GN experience syncope due to the anatomical proximity of the vagus nerve.14
Treatment. Anticonvulsants are the first-line treatment for GN. Local anesthetics or surgery can be considered for patients who don’t improve after medical therapy.15
Postherpetic neuralgia (PHN) can cause facial pain when the characteristic vesicular rash of the varicella zoster virus (shingles) occurs on the face. PHN usually affects the first division of trigeminal nerve, but the second and third divisions can be affected as well.13
What you’ll see. The acute phase of PHN begins a few days before the initial rash has resolved and can last up to a month after. A new pain may begin one to 6 months after the initial rash has healed.20 This pain, which patients often describe as sharp, stabbing, or burning, can be constant or intermittent. Dysesthesia, hypoesthesia, and allodynia may also occur within the affected dermatome.
PHN is usually diagnosed based on the patient’s history and clinical presentation. However, direct fluorescent antibody stain, viral culture, or polymerase chain reaction performed on vesicular fluid from a herpetic lesion during the initial rash are the laboratory tests of choice if confirmation is needed.
Treatment. PHN is managed with anticonvulsants and TCAs.
Numb chin syndrome (NCS) is characterized by hypoesthesia, paresthesia, thermalgesic anesthesia, or pain over the chin in the region supplied by the mental nerve, a terminal branch of the mandibular division of the trigeminal nerve.5,21,22
NCS can be caused by odontogenic conditions, such as dental abscess, dental anesthesia, dental trauma, or osteomyelitis; systemic conditions such as amyloidosis, sickle cell disease, sarcoidosis, multiple sclerosis, human immunodeficiency virus, or diabetes; or malignancies such as lymphoma, leukemia, breast cancer, lung cancer, prostate cancer, or head and neck cancers.21 In one study of patients with NCS, cancer was the cause of the condition in 89% of patients.22
What you’ll see. NCS is characterized by numbness of the skin in the lower lip, chin and mucous membrane inside the lip that extends to the midline.5 Depending upon the etiology, patients may present with percussion-induced pain, loosening of teeth, sequestra, and mobility of fractured segments. Patients with metastatic malignancy may develop constitutional symptoms.
Making the diagnosis. Panoramic radiography is a useful starting point. If possible, a computerized tomography scan of the head and neck should also be done. Nuclear bone scintigraphy (bone scanning) may help identify bone disease such as osteomyelitis. A biopsy may be needed if a mass lesion is present.
Treatment. In NCS that is the result of a dental etiology, the prognosis usually is good. For example, NCS that is the result of an abscess usually resolves after the abscess is drained. However, if NCS is caused by metastasis, the prognosis is grim; the average length of survival after diagnosis is approximately 5 months if NCS is caused by mandibular metastasis and 12 months if leptomeningeal metastasis is present. Treatment does little to affect the outcome in these cases.21,22
Atypical pain
Persistent idiopathic facial pain (PIFP), previously known as atypical facial pain, is a persistent facial pain that does not have the classical characteristics of cranial neuralgias and for which there is no obvious cause.2,10,23 PIFP is not triggered by any of the factors that typically precipitate neuralgias.2 The onset may be spontaneous or associated with dental intervention or facial injury, but it usually does not have a demonstrable local cause.24,25
Neuropathic mechanisms that might be at work in PIFP include nociceptor sensitization, phenotypic changes and ectopic activity from the nociceptors, central sensitization possibly maintained by ongoing activity from initially damaged peripheral tissues, sympathetic abnormal activity, alteration of segmental inhibitory control, or hyperactivity or hypoactivity of descending controls.2
PIFP is most frequently reported in women in their 40s and 50s.25 The history of a patient with PIFP often include mood disorders, chronic pain, or poor coping skills.14 Patients complain of a steady, unilateral, poorly localized pain that is deep, constant, aching, pulling, or crushing. It is usually present all day, every day. The constancy of the pain is its distinguishing feature. In the beginning, this pain may be in a limited area on one side of the face, usually the nasolabial folds or the angle of the mandible. Later, it may affect both sides of the face and extend to the neck and upper limbs.23,24 Most patients with PIFP report other symptoms, including headache, neck and backache, dermatitis, pruritus, irritable bowel, and dysfunctional uterine bleeding.26
Making the diagnosis. A targeted history and accurate clinical examination are essential.2,10 Although there are no formal diagnostic criteria, a patient can be assumed to have PIFP if:2,10
• There is pain in the face for most of the day or all day, every day.
• Initially, the pain may be confined to a portion of the face, but it is poorly localized and deep.
• The pain is not associated with other physical signs or loss of sensation.
• Imaging does not reveal an obvious anatomic or structural cause.
Treatment. Treatment of PIFP can be difficult and unsatisfactory.23 Counseling to educate patients about the chronic and nonmalignant nature of the illness is the mainstay of treatment, followed by pharmacotherapy.23 TCAs have shown a moderate effect in several trials. Gabapentin, topiramate, carbamazepine, and pregabalin also have shown limited to modest benefit in some patients. Surgical therapies appear to be of little or no use.23 Experimental treatments such as pulsed radiofrequency, low-energy level diode laser have shown success in small studies.10,23
Vascular pain
Giant cell arteritis (GCA) is a systemic, chronic vasculitis involving the large and medium-sized vessels, mainly the extracranial branches of the carotid artery.6,11 It predominantly affects people older than age 50 and is more common among women and those of Scandinavian ethnicity.27
The cause of GCA is unclear. Genetic predisposition linked to humoral and cellmediated immunity is believed to play a role.28 Familial aggregation and predominance of the HLA-DR4 allele has been reported in patients with GCA.6
What you’ll see. The most common signs and symptoms of GCA are temporal headache (seen in two-thirds of patients), jaw claudication and tenderness, and swelling of the temporal artery.6,11 The headache of GCA usually is unilateral, severe, boring or lancinating, and localized to the temporal or occipital regions of the scalp.6 Other orofacial manifestations include trismus, throat pain that develops while chewing, changes in tongue sensation and tongue claudication, tooth pain, dysphagia, dysarthria, submandibular mass, lip and chin numbness, macroglossia, glossitis, lip and tongue necrosis, and facial swelling.11
Visual symptoms include diplopia, ptosis, and possibly blindness if treatment is not instituted at first suspicion. Ocular symptoms result from anterior ischemic optic neuropathy, posterior ischemic optic neuropathy, or central retinal or cilioretinal artery occlusion.6,28 Patients have also reported low-grade fever, asthenia, anorexia, weight loss, and generalized aches.11,28
Making the diagnosis. Arterial biopsy is the gold standard for diagnosis of GCA. It is usually performed on the temporal artery and is positive in 80% to 95% of people with the condition.28 Other useful lab tests include erythrocyte sedimentation rate (ESR; elevated), white blood cell count (mildly elevated), and C-reactive protein (elevated).
Treatment. Prednisone is used to treat GCA, in initial doses ranging from 30 to 80 mg. A maintenance dose may be required for up to 2 years, with close follow-up and periodic ESR measurements.28
Malignancy is a rare cause of facial pain. The pain may be due to metastasis of extracranial bony or soft tissue as it compresses cervical and cranial nerves.3 Lung cancer can cause referred pain in the periauricular region by compressing the vagus nerve, and this pain can be misdiagnosed as dental pain, atypical facial pain, TMD, or TN.3,29 The facial pain of lung cancer is unilateral and on the same side as the lung neoplasm, and commonly is referred to the jaw, ear, or temporal region. While many patients have continuous pain, some report intermittent pain or pain that lasts for hours.3 Facial pain caused by a malignancy is differentiated from other sources of facial pain by the presence of associated symptoms such as weight loss, cough, and hemoptysis.
Treatment. Treatment can include radiation or chemotherapy.29
The mouth is often the source of lower facial pain
Pain in the oral cavity is the most common cause of pain in the lower face.15 Intraoral pain usually is caused by disease in the following structures:
1. Dentition (eg, caries, dentin sensitivity, pulpal disease)
2. Periodontium (eg, gingivitis, acute or chronic periodontal disease, sensitivity related to gum recession, alveolar bone pathology)
3. Other soft and hard tissues, such as the palate, floor of mouth, buccal mucosa, non-tooth supporting bone, and tongue (eg, mucosal diseases, neoplasms, pain related to parafunction or trauma).
Rarely, intraoral pain may be referred. For example, myofascial pain might cause diffuse tooth pain.30
See TABLE W131-35 at the end of this article for a summary of the etiology, signs/symptoms, diagnosis, and management of these and other dental causes of oral facial pain.
Nondental causes of oral facial pain can be associated with oral mucosal disorders, malignant disease and its therapy, salivary gland disorders, maxillary sinusitis, burning mouth syndrome, or atypical odontalgia. See TABLE W236-41 for a more detailed description of these conditions.
CORRESPONDENCE
Tamer H. Said, MD, MetroHealth Medical Center, 2500 MetroHealth Drive, Cleveland, Ohio 44109; [email protected]
› Advise patients who have a temporomandibular disorder that in addition to taking their medication as prescribed, they should limit activities that require moving their jaw, modify their diet, and minimize stress; they may require physical therapy and therapeutic exercises. C
› Consider prescribing a tricyclic antidepressant for patients with persistent idiopathic facial pain. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Facial pain is a common complaint: Up to 22% of adults in the United States experience orofacial pain during any 6-month period.1 Yet this type of pain can be difficult to diagnose due to the many structures of the face and mouth, pain referral patterns, and insufficient diagnostic tools.
Specifically, extraoral facial pain can be the result of temporomandibular disorders, neuropathic disorders, vascular disorders, or atypical causes, whereas facial pain stemming from inside the mouth can have a dental or nondental cause (FIGURE). Overlapping characteristics can make it difficult to distinguish these disorders. To help you to better diagnose and manage facial pain, we describe the most common causes and underlying pathological processes.
Extraoral facial pain
Extraoral pain refers to the pain that occurs on the face outside of the oral cavity. The TABLE2-15 summarizes the site, timing and severity, aggravating factors, history and exam findings, and management of several common causes of extraoral facial pain.
Musculoskeletal pain
Temporomandibular disorders (TMD) are a broad group of problems that affect the temporomandibular joint (TMJ), muscles of mastication, and/or associated bony and soft tissue structures.6 They may occur secondary to malocclusion, traumatic injuries, oral parafunctional habits (eg, bruxism), hormonal influences, or psychogenic factors.6 TMD is more prevalent in women, with a peak occurrence between ages 20 and 40 years.6,8
TMD can be articular (intracapsular) or nonarticular (extracapsular). Nonarticular disorders (>50% of TMD) usually affect the muscles of mastication and include chronic conditions such as fibromyalgia, muscle strain, and myopathies.8 Muscle-related pain and dysfunction are believed to arise from parafunctional habits such as bruxism or clenching. Articular disorders include synovitis/capsulitis, joint effusion, trauma/fracture, internal derangement (disturbance in the normal anatomic relationship between the disc and condyle), arthritis, and neoplasm.16
What you’ll see. Orofacial pain (usually dull and located in the preauricular region), joint noise, and restricted jaw function are key signs and symptoms of TMD. Exacerbation of pain with mandibular functions (eg, chewing, yawning, or swallowing) is a pathognomonic sign. Joint sounds such as clicking or crepitus are common. In most cases, crepitus correlates with osteoarthritis.6 Nonspecific TMD symptoms include headache, earache, insomnia, tinnitus, and neck and shoulder pain.6
The gold standard of diagnosis of TMD consists of taking a detailed history, evaluating the patient’s head and neck, and conducting a general physical examination and behavioral/psychological assessment.17 Imaging of the TMJ and associated structures is essential.17
Treatment. Nonsteroidal anti-inflammatory drugs, opioids, muscle relaxants, antidepressants, anticonvulsants, anxiolytics, and corticosteroids are options for treating TMD.6,8 Isometric jaw exercises, maxillomandibular appliances, and physical therapy are valuable adjuncts for pain relief. Advise patients to establish a self-care routine to reduce TMJ pain that might include changing their head posture or sleeping position, and limiting activities that require using their jaw, such as clenching, bruxism, and excessive gum chewing. Some patients may need to adopt a non-chewing diet that consists of liquid or pureed food. Massage and moist heat can help relax muscles of mastication and improve range of motion.
Approximately 5% of patients with TMD undergo surgery, typically simple arthrocentesis, arthroscopy, arthrotomy, or modified condylotomy.6 Total joint replacement is indicated only for patients with severely damaged joints with end-stage disease when all other conservative treatments have failed. Joint replacement primarily restores form and function; pain relief is a secondary benefit.8
Neuropathic pain
Trigeminal neuralgia (TN) is sudden, usually unilateral, severe, brief, stabbing, recurrent episodes of pain in the distribution of one or more branches of the trigeminal nerve.9 It most commonly presents in the lower 2 branches of the trigeminal nerve and usually is caused by compression of the trigeminal nerve root by vascular or nonvascular causes.4 The pain is severe and can profoundly impact a patient’s quality of life.
TN attacks typically last from a few seconds to up to 2 minutes. As many as 30 attacks can occur daily, with refractory periods between attacks. After the initial attack, individuals are left with a residual dull or burning pain. TN can be triggered by face washing, teeth brushing, speaking, eating, shaving, or cold wind.4
Diagnosis can be tricky because more than half of patients with TN experience less severe pain after the main sharp attack; this presentation is called TN type II.7 A detailed patient history and careful evaluation can help identify patients with TN type II. TN can be misdiagnosed as TMD, especially if it presents unilaterally.15
Treatment. Anticonvulsants are the primary medications used to treat TN.
Post-traumatic trigeminal pain is usually the result of an injury or dental procedure, such as facial trauma, tooth extraction, root canal, or dental implants.12,18,19 Nerve injury is assumed to be the cause. This type of pain can start within 3 to 6 months of a trauma. It is located in the trigeminal area and patients describe it as burning, tingling and, at times, sharp.15 Patients who have sustained injury to the lingual or inferior alveolar nerves have reported feeling “pins and needles.”12
Common triggers include temperature changes or simple touch. Not all injuries result in pain; some patients may have only sensory impairment15 or sensory deficits such as allodynia or hypoesthesia.
Treatment. The first line of treatment for post-traumatic trigeminal pain is tricyclic antidepressants (TCAs) followed by pregabalin or gabapentin.14
Glossopharyngeal neuralgia (GN) is similar in presentation to TN but is much rarer.15 GN pain occurs deep in the throat, ear, or posterior tongue.15 When the pain occurs in the inner ear, GN can be misdiagnosed as TMD. In most cases, no cause of GN can be determined.
Patients describe GN pain as shooting, sharp, and electrical shock-like, lasting from seconds to minutes, with recurrent attacks throughout the day. Like TN, GN can present as episodes of attacks that last weeks to months. Triggers include chewing, drinking, swallowing, and talking, as well as light touch.13,15 Some patients with GN experience syncope due to the anatomical proximity of the vagus nerve.14
Treatment. Anticonvulsants are the first-line treatment for GN. Local anesthetics or surgery can be considered for patients who don’t improve after medical therapy.15
Postherpetic neuralgia (PHN) can cause facial pain when the characteristic vesicular rash of the varicella zoster virus (shingles) occurs on the face. PHN usually affects the first division of trigeminal nerve, but the second and third divisions can be affected as well.13
What you’ll see. The acute phase of PHN begins a few days before the initial rash has resolved and can last up to a month after. A new pain may begin one to 6 months after the initial rash has healed.20 This pain, which patients often describe as sharp, stabbing, or burning, can be constant or intermittent. Dysesthesia, hypoesthesia, and allodynia may also occur within the affected dermatome.
PHN is usually diagnosed based on the patient’s history and clinical presentation. However, direct fluorescent antibody stain, viral culture, or polymerase chain reaction performed on vesicular fluid from a herpetic lesion during the initial rash are the laboratory tests of choice if confirmation is needed.
Treatment. PHN is managed with anticonvulsants and TCAs.
Numb chin syndrome (NCS) is characterized by hypoesthesia, paresthesia, thermalgesic anesthesia, or pain over the chin in the region supplied by the mental nerve, a terminal branch of the mandibular division of the trigeminal nerve.5,21,22
NCS can be caused by odontogenic conditions, such as dental abscess, dental anesthesia, dental trauma, or osteomyelitis; systemic conditions such as amyloidosis, sickle cell disease, sarcoidosis, multiple sclerosis, human immunodeficiency virus, or diabetes; or malignancies such as lymphoma, leukemia, breast cancer, lung cancer, prostate cancer, or head and neck cancers.21 In one study of patients with NCS, cancer was the cause of the condition in 89% of patients.22
What you’ll see. NCS is characterized by numbness of the skin in the lower lip, chin and mucous membrane inside the lip that extends to the midline.5 Depending upon the etiology, patients may present with percussion-induced pain, loosening of teeth, sequestra, and mobility of fractured segments. Patients with metastatic malignancy may develop constitutional symptoms.
Making the diagnosis. Panoramic radiography is a useful starting point. If possible, a computerized tomography scan of the head and neck should also be done. Nuclear bone scintigraphy (bone scanning) may help identify bone disease such as osteomyelitis. A biopsy may be needed if a mass lesion is present.
Treatment. In NCS that is the result of a dental etiology, the prognosis usually is good. For example, NCS that is the result of an abscess usually resolves after the abscess is drained. However, if NCS is caused by metastasis, the prognosis is grim; the average length of survival after diagnosis is approximately 5 months if NCS is caused by mandibular metastasis and 12 months if leptomeningeal metastasis is present. Treatment does little to affect the outcome in these cases.21,22
Atypical pain
Persistent idiopathic facial pain (PIFP), previously known as atypical facial pain, is a persistent facial pain that does not have the classical characteristics of cranial neuralgias and for which there is no obvious cause.2,10,23 PIFP is not triggered by any of the factors that typically precipitate neuralgias.2 The onset may be spontaneous or associated with dental intervention or facial injury, but it usually does not have a demonstrable local cause.24,25
Neuropathic mechanisms that might be at work in PIFP include nociceptor sensitization, phenotypic changes and ectopic activity from the nociceptors, central sensitization possibly maintained by ongoing activity from initially damaged peripheral tissues, sympathetic abnormal activity, alteration of segmental inhibitory control, or hyperactivity or hypoactivity of descending controls.2
PIFP is most frequently reported in women in their 40s and 50s.25 The history of a patient with PIFP often include mood disorders, chronic pain, or poor coping skills.14 Patients complain of a steady, unilateral, poorly localized pain that is deep, constant, aching, pulling, or crushing. It is usually present all day, every day. The constancy of the pain is its distinguishing feature. In the beginning, this pain may be in a limited area on one side of the face, usually the nasolabial folds or the angle of the mandible. Later, it may affect both sides of the face and extend to the neck and upper limbs.23,24 Most patients with PIFP report other symptoms, including headache, neck and backache, dermatitis, pruritus, irritable bowel, and dysfunctional uterine bleeding.26
Making the diagnosis. A targeted history and accurate clinical examination are essential.2,10 Although there are no formal diagnostic criteria, a patient can be assumed to have PIFP if:2,10
• There is pain in the face for most of the day or all day, every day.
• Initially, the pain may be confined to a portion of the face, but it is poorly localized and deep.
• The pain is not associated with other physical signs or loss of sensation.
• Imaging does not reveal an obvious anatomic or structural cause.
Treatment. Treatment of PIFP can be difficult and unsatisfactory.23 Counseling to educate patients about the chronic and nonmalignant nature of the illness is the mainstay of treatment, followed by pharmacotherapy.23 TCAs have shown a moderate effect in several trials. Gabapentin, topiramate, carbamazepine, and pregabalin also have shown limited to modest benefit in some patients. Surgical therapies appear to be of little or no use.23 Experimental treatments such as pulsed radiofrequency, low-energy level diode laser have shown success in small studies.10,23
Vascular pain
Giant cell arteritis (GCA) is a systemic, chronic vasculitis involving the large and medium-sized vessels, mainly the extracranial branches of the carotid artery.6,11 It predominantly affects people older than age 50 and is more common among women and those of Scandinavian ethnicity.27
The cause of GCA is unclear. Genetic predisposition linked to humoral and cellmediated immunity is believed to play a role.28 Familial aggregation and predominance of the HLA-DR4 allele has been reported in patients with GCA.6
What you’ll see. The most common signs and symptoms of GCA are temporal headache (seen in two-thirds of patients), jaw claudication and tenderness, and swelling of the temporal artery.6,11 The headache of GCA usually is unilateral, severe, boring or lancinating, and localized to the temporal or occipital regions of the scalp.6 Other orofacial manifestations include trismus, throat pain that develops while chewing, changes in tongue sensation and tongue claudication, tooth pain, dysphagia, dysarthria, submandibular mass, lip and chin numbness, macroglossia, glossitis, lip and tongue necrosis, and facial swelling.11
Visual symptoms include diplopia, ptosis, and possibly blindness if treatment is not instituted at first suspicion. Ocular symptoms result from anterior ischemic optic neuropathy, posterior ischemic optic neuropathy, or central retinal or cilioretinal artery occlusion.6,28 Patients have also reported low-grade fever, asthenia, anorexia, weight loss, and generalized aches.11,28
Making the diagnosis. Arterial biopsy is the gold standard for diagnosis of GCA. It is usually performed on the temporal artery and is positive in 80% to 95% of people with the condition.28 Other useful lab tests include erythrocyte sedimentation rate (ESR; elevated), white blood cell count (mildly elevated), and C-reactive protein (elevated).
Treatment. Prednisone is used to treat GCA, in initial doses ranging from 30 to 80 mg. A maintenance dose may be required for up to 2 years, with close follow-up and periodic ESR measurements.28
Malignancy is a rare cause of facial pain. The pain may be due to metastasis of extracranial bony or soft tissue as it compresses cervical and cranial nerves.3 Lung cancer can cause referred pain in the periauricular region by compressing the vagus nerve, and this pain can be misdiagnosed as dental pain, atypical facial pain, TMD, or TN.3,29 The facial pain of lung cancer is unilateral and on the same side as the lung neoplasm, and commonly is referred to the jaw, ear, or temporal region. While many patients have continuous pain, some report intermittent pain or pain that lasts for hours.3 Facial pain caused by a malignancy is differentiated from other sources of facial pain by the presence of associated symptoms such as weight loss, cough, and hemoptysis.
Treatment. Treatment can include radiation or chemotherapy.29
The mouth is often the source of lower facial pain
Pain in the oral cavity is the most common cause of pain in the lower face.15 Intraoral pain usually is caused by disease in the following structures:
1. Dentition (eg, caries, dentin sensitivity, pulpal disease)
2. Periodontium (eg, gingivitis, acute or chronic periodontal disease, sensitivity related to gum recession, alveolar bone pathology)
3. Other soft and hard tissues, such as the palate, floor of mouth, buccal mucosa, non-tooth supporting bone, and tongue (eg, mucosal diseases, neoplasms, pain related to parafunction or trauma).
Rarely, intraoral pain may be referred. For example, myofascial pain might cause diffuse tooth pain.30
See TABLE W131-35 at the end of this article for a summary of the etiology, signs/symptoms, diagnosis, and management of these and other dental causes of oral facial pain.
Nondental causes of oral facial pain can be associated with oral mucosal disorders, malignant disease and its therapy, salivary gland disorders, maxillary sinusitis, burning mouth syndrome, or atypical odontalgia. See TABLE W236-41 for a more detailed description of these conditions.
CORRESPONDENCE
Tamer H. Said, MD, MetroHealth Medical Center, 2500 MetroHealth Drive, Cleveland, Ohio 44109; [email protected]
1. Lipton JA, Ship JA, Larach-Robinson D. Estimated prevalence and distribution of reported orofacial pain in the United States. J Am Dent Assoc. 1993;124:115-1121.
2. Agostoni E, Frigerio R, Santoro P. Atypical facial pain: clinical considerations and differential diagnosis. Neurol Sci. 2005;26:S71-S74.
3. Bajwa Z, Ho C, Khan S, et al. Overview of craniofacial pain. UpTo-Date Web site. Available at: http://www.uptodate.com/contents/overview-of-craniofacial-pain. Accessed January 28, 2015.
4. Bendtsen L, Birk S, Kasch H, et al. Reference programme: Diagnosis and treatment of headache disorders and facial pain. Danish Headache Society, 2nd Edition, 2012. J Headache Pain. 2012;13:S1-S29.
5. Divya KS, Moran NA, Atkin PA. Numb chin syndrome: a case series and discussion. Br Dent J. 2010;208:157-160.
6. Kapur N, Kamel IR, Herlich A. Oral and craniofacial pain: diagnosis, pathophysiology, and treatment. Int Anesthesiol Clin. 2003;41:115-150.
7. Limonadi FM, McCartney S, Burchiel KJ. Design of an artificial neural network for diagnosis of facial pain syndromes. Stereotact Funct Neurosurg. 2006;84:212-220.
8. Liu F, Steinkeler A. Epidemiology, diagnosis, and treatment of temporomandibular disorders. Dent Clin North Am. 2013;57:465-479.
9. Merskey H, Bogduk N (eds). Classification of Chronic Pain. Descriptors of Chronic Pain Syndromes and Definition of Pain Terms, 2nd ed. Seattle, WA: International Association for the Study of Pain Press; 1994.
10. Nguyen CT, Wang MB. Complementary and integrative treatments: atypical facial pain. Otolaryngol Clin North Am. 2013;46:367-382.
11. Reiter S, Winocur E, Goldsmith C, et al. Giant cell arteritis misdiagnosed as temporomandibular disorder: a case report and review of the literature. J Orofac Pain. 2009;23:360-365.
12. Renton T, Adey-Viscuso D, Meechan JG, et al. Trigeminal nerve injuries in relation to local anaesthesia in mandibular injections. Br Dent J. 2010;209:E15.
13. Shephard MK, Macgregor EA, Zakrzewska JM. Orofacial pain: a guide for the headache physician. Headache. 2014;54:22-39.
14. Zakrzewska JM. Differential diagnosis of facial pain and guidelines for management. Br J Anaesth. 2013;111:95-104.
15. Zakrzewska JM. Multi-dimensionality of chronic pain of the oral cavity and face. J Headache Pain. 2013;14:37.
16. Herb K, Cho S, Stiles MA. Temporomandibular joint pain and dysfunction. Curr Pain Headache Rep. 2006;10:408-414.
17. American Society of Temporomandibular Joint Surgeons. Guidelines for diagnosis and management of disorders involving the temporomandibular joint and related musculoskeletal structures. Cranio. 2003;21:68-76.
18. Benoliel R, Zadik Y, Eliav E, et al. Peripheral painful traumatic trigeminal neuropathy: clinical features in 91 cases and proposal of novel diagnostic criteria. J Orofac Pain. 2012;26:49-58.
19. Brooke RI. Atypical odontalgia. A report of twenty-two cases. Oral Surg Oral Med Oral Pathol. 1980;49:196-199.
20. Bouhassira D, Chassany O, Gaillat J, et al. Patient perspective on herpes zoster and its complications: an observational prospective study in patients aged over 50 years in general practice. Pain. 2012;153:342-349.
21. Baskaran RK, Krishnamoorthy, Smith M. Numb chin syndrome—a reflection of systemic malignancy. World J Surg Oncol. 2006;4:52.
22. Lata J, Kumar P. Numb chin syndrome: a case report and review of the literature. Indian J Dent Res. 2010;21:135-137.
23. Cornelissen P, van Kleef M, Mekhail N, et al. Evidence-based interventional pain medicine according to clinical diagnoses. 3. Persistent idiopathic facial pain. Pain Pract. 2009;9:443-448.
24. Didier H, Marchetti C, Borromeo G, et al. Persistent idiopathic facial pain: multidisciplinary approach and assumption of comorbidity. Neurol Sci. 2010;31:S189-S195.
25. Klasser G. Management of persistent idiopathic facial pain. J Can Dent Assoc. 2013;79:d71.
26. Abiko Y, Matsuoka H, Chiba I, et al. Current evidence on atypical odontalgia: diagnosis and clinical management. Int J Dent. 2012;2012:518548.
27. Sheldon CA, White VA, Holland SP. Giant cell arteritis presenting with bilateral loss of vision and jaw pain: reminder of a potentially devastating condition. J Can Dent Assoc. 2011;77:b55.
28. Rockey JG, Anand R. Tongue necrosis secondary to temporal arteritis: a case report and literature review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;94:471-473.
29. Sarlani E, Schwartz AH, Greenspan JD, et al. Facial pain as first manifestation of lung cancer: a case of lung cancer-related cluster headache and a review of the literature. J Orofac Pain. 2003;17:262-267.
30. Kumar A, Brennan MT. Differential diagnosis of orofacial pain and temporomandibular disorder. Dent Clin North Am. 2013;57:419-428.
31. Laudenbach JM, Simon Z. Common dental and periodontal diseases: evaluation and management. Med Clin North Am. 2014;98:1239-1260.
32. Napeñas JJ. Intraoral pain disorders. Dent Clin North Am. 2013;57:429-447.
33. Vickers ER, Zakrzewska JM. Dental causes of orofacial pain. In: Orofacial Pain. Zakrzewska JM, ed. Oxford, UK: Oxford University Press; 2009:69-81.
34. Pierse JE, Dym H, Clarkson E. Diagnosis and management of common postextraction complications. Dent Clin North Am. 2012;56:75-93.
35. Renton T. Dental (odontogenic) pain. Br J Pain. 2011;5:2-7.
36. Yatani H, Komiyama O, Matsuka Y, et al. Systematic review and recommendations for nonodontogenic toothache. J Oral Rehabil. 2014;41:843-852.
37. Klasser GD, Fischer DJ, Epstein JB. Burning mouth syndrome: recognition, understanding, and management. Oral Maxillofac Surg Clin North Am. 2008;20:255-271.
38. Balasubramaniam R, Turner LN, Fischer D, et al. Non-odontogenic toothache revisited. Open Journal of Stomatology. 2011;1:92-102.
39. Patton LL, Siegel MA, Benoliel R, et al. Management of burning mouth syndrome: systematic review and management recommendations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:S39.e1-e13.
40. Cascarini L, McGurk M. Epidemiology of salivary gland infections. Oral Maxillofac Surg Clin North Am. 2009;21:353-357.
41. Hegarty AM, Zakrzewska JM. Differential diagnosis for orofacial pain, including sinusitis, TMD, trigeminal neuralgia. Dent Update. 2011;38:396-400,402-403,405-406.
1. Lipton JA, Ship JA, Larach-Robinson D. Estimated prevalence and distribution of reported orofacial pain in the United States. J Am Dent Assoc. 1993;124:115-1121.
2. Agostoni E, Frigerio R, Santoro P. Atypical facial pain: clinical considerations and differential diagnosis. Neurol Sci. 2005;26:S71-S74.
3. Bajwa Z, Ho C, Khan S, et al. Overview of craniofacial pain. UpTo-Date Web site. Available at: http://www.uptodate.com/contents/overview-of-craniofacial-pain. Accessed January 28, 2015.
4. Bendtsen L, Birk S, Kasch H, et al. Reference programme: Diagnosis and treatment of headache disorders and facial pain. Danish Headache Society, 2nd Edition, 2012. J Headache Pain. 2012;13:S1-S29.
5. Divya KS, Moran NA, Atkin PA. Numb chin syndrome: a case series and discussion. Br Dent J. 2010;208:157-160.
6. Kapur N, Kamel IR, Herlich A. Oral and craniofacial pain: diagnosis, pathophysiology, and treatment. Int Anesthesiol Clin. 2003;41:115-150.
7. Limonadi FM, McCartney S, Burchiel KJ. Design of an artificial neural network for diagnosis of facial pain syndromes. Stereotact Funct Neurosurg. 2006;84:212-220.
8. Liu F, Steinkeler A. Epidemiology, diagnosis, and treatment of temporomandibular disorders. Dent Clin North Am. 2013;57:465-479.
9. Merskey H, Bogduk N (eds). Classification of Chronic Pain. Descriptors of Chronic Pain Syndromes and Definition of Pain Terms, 2nd ed. Seattle, WA: International Association for the Study of Pain Press; 1994.
10. Nguyen CT, Wang MB. Complementary and integrative treatments: atypical facial pain. Otolaryngol Clin North Am. 2013;46:367-382.
11. Reiter S, Winocur E, Goldsmith C, et al. Giant cell arteritis misdiagnosed as temporomandibular disorder: a case report and review of the literature. J Orofac Pain. 2009;23:360-365.
12. Renton T, Adey-Viscuso D, Meechan JG, et al. Trigeminal nerve injuries in relation to local anaesthesia in mandibular injections. Br Dent J. 2010;209:E15.
13. Shephard MK, Macgregor EA, Zakrzewska JM. Orofacial pain: a guide for the headache physician. Headache. 2014;54:22-39.
14. Zakrzewska JM. Differential diagnosis of facial pain and guidelines for management. Br J Anaesth. 2013;111:95-104.
15. Zakrzewska JM. Multi-dimensionality of chronic pain of the oral cavity and face. J Headache Pain. 2013;14:37.
16. Herb K, Cho S, Stiles MA. Temporomandibular joint pain and dysfunction. Curr Pain Headache Rep. 2006;10:408-414.
17. American Society of Temporomandibular Joint Surgeons. Guidelines for diagnosis and management of disorders involving the temporomandibular joint and related musculoskeletal structures. Cranio. 2003;21:68-76.
18. Benoliel R, Zadik Y, Eliav E, et al. Peripheral painful traumatic trigeminal neuropathy: clinical features in 91 cases and proposal of novel diagnostic criteria. J Orofac Pain. 2012;26:49-58.
19. Brooke RI. Atypical odontalgia. A report of twenty-two cases. Oral Surg Oral Med Oral Pathol. 1980;49:196-199.
20. Bouhassira D, Chassany O, Gaillat J, et al. Patient perspective on herpes zoster and its complications: an observational prospective study in patients aged over 50 years in general practice. Pain. 2012;153:342-349.
21. Baskaran RK, Krishnamoorthy, Smith M. Numb chin syndrome—a reflection of systemic malignancy. World J Surg Oncol. 2006;4:52.
22. Lata J, Kumar P. Numb chin syndrome: a case report and review of the literature. Indian J Dent Res. 2010;21:135-137.
23. Cornelissen P, van Kleef M, Mekhail N, et al. Evidence-based interventional pain medicine according to clinical diagnoses. 3. Persistent idiopathic facial pain. Pain Pract. 2009;9:443-448.
24. Didier H, Marchetti C, Borromeo G, et al. Persistent idiopathic facial pain: multidisciplinary approach and assumption of comorbidity. Neurol Sci. 2010;31:S189-S195.
25. Klasser G. Management of persistent idiopathic facial pain. J Can Dent Assoc. 2013;79:d71.
26. Abiko Y, Matsuoka H, Chiba I, et al. Current evidence on atypical odontalgia: diagnosis and clinical management. Int J Dent. 2012;2012:518548.
27. Sheldon CA, White VA, Holland SP. Giant cell arteritis presenting with bilateral loss of vision and jaw pain: reminder of a potentially devastating condition. J Can Dent Assoc. 2011;77:b55.
28. Rockey JG, Anand R. Tongue necrosis secondary to temporal arteritis: a case report and literature review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002;94:471-473.
29. Sarlani E, Schwartz AH, Greenspan JD, et al. Facial pain as first manifestation of lung cancer: a case of lung cancer-related cluster headache and a review of the literature. J Orofac Pain. 2003;17:262-267.
30. Kumar A, Brennan MT. Differential diagnosis of orofacial pain and temporomandibular disorder. Dent Clin North Am. 2013;57:419-428.
31. Laudenbach JM, Simon Z. Common dental and periodontal diseases: evaluation and management. Med Clin North Am. 2014;98:1239-1260.
32. Napeñas JJ. Intraoral pain disorders. Dent Clin North Am. 2013;57:429-447.
33. Vickers ER, Zakrzewska JM. Dental causes of orofacial pain. In: Orofacial Pain. Zakrzewska JM, ed. Oxford, UK: Oxford University Press; 2009:69-81.
34. Pierse JE, Dym H, Clarkson E. Diagnosis and management of common postextraction complications. Dent Clin North Am. 2012;56:75-93.
35. Renton T. Dental (odontogenic) pain. Br J Pain. 2011;5:2-7.
36. Yatani H, Komiyama O, Matsuka Y, et al. Systematic review and recommendations for nonodontogenic toothache. J Oral Rehabil. 2014;41:843-852.
37. Klasser GD, Fischer DJ, Epstein JB. Burning mouth syndrome: recognition, understanding, and management. Oral Maxillofac Surg Clin North Am. 2008;20:255-271.
38. Balasubramaniam R, Turner LN, Fischer D, et al. Non-odontogenic toothache revisited. Open Journal of Stomatology. 2011;1:92-102.
39. Patton LL, Siegel MA, Benoliel R, et al. Management of burning mouth syndrome: systematic review and management recommendations. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;103:S39.e1-e13.
40. Cascarini L, McGurk M. Epidemiology of salivary gland infections. Oral Maxillofac Surg Clin North Am. 2009;21:353-357.
41. Hegarty AM, Zakrzewska JM. Differential diagnosis for orofacial pain, including sinusitis, TMD, trigeminal neuralgia. Dent Update. 2011;38:396-400,402-403,405-406.
Treatment outcomes in stage IIIA non–small-cell lung cancer in a community cancer center
Objective To analyze demographics and treatment outcomes in patients with stage IIIA NSCLC at a community cancer center.
Methods We reviewed charts of 226 patients diagnosed with stage IIIA NSCLC from January 2003 to December 2008 treated at our community cancer center. Results Median overall survival for all patients and sequentially and concurrently treated chemoradiation patients were 18 months, and 18 months, and 20 months, respectively. Median overall survival for women and men was 24 months and 16 months, respectively.
Limitations Study design was retrospective and some medical records were not available. However, this population is likely representative of patients treated in similar settings.
Conclusions In our population, advanced age and male gender were associated with lower median survival. Responses to concurrent and sequential chemoradiation seemed to differ based on age group, which may be useful as a prognostic guideline for similar populations.
Funding Helen F Graham Cancer Center and Research Institute
Click on the PDF icon at the top of this introduction to read the full article.
Objective To analyze demographics and treatment outcomes in patients with stage IIIA NSCLC at a community cancer center.
Methods We reviewed charts of 226 patients diagnosed with stage IIIA NSCLC from January 2003 to December 2008 treated at our community cancer center. Results Median overall survival for all patients and sequentially and concurrently treated chemoradiation patients were 18 months, and 18 months, and 20 months, respectively. Median overall survival for women and men was 24 months and 16 months, respectively.
Limitations Study design was retrospective and some medical records were not available. However, this population is likely representative of patients treated in similar settings.
Conclusions In our population, advanced age and male gender were associated with lower median survival. Responses to concurrent and sequential chemoradiation seemed to differ based on age group, which may be useful as a prognostic guideline for similar populations.
Funding Helen F Graham Cancer Center and Research Institute
Click on the PDF icon at the top of this introduction to read the full article.
Objective To analyze demographics and treatment outcomes in patients with stage IIIA NSCLC at a community cancer center.
Methods We reviewed charts of 226 patients diagnosed with stage IIIA NSCLC from January 2003 to December 2008 treated at our community cancer center. Results Median overall survival for all patients and sequentially and concurrently treated chemoradiation patients were 18 months, and 18 months, and 20 months, respectively. Median overall survival for women and men was 24 months and 16 months, respectively.
Limitations Study design was retrospective and some medical records were not available. However, this population is likely representative of patients treated in similar settings.
Conclusions In our population, advanced age and male gender were associated with lower median survival. Responses to concurrent and sequential chemoradiation seemed to differ based on age group, which may be useful as a prognostic guideline for similar populations.
Funding Helen F Graham Cancer Center and Research Institute
Click on the PDF icon at the top of this introduction to read the full article.
Migraine May Increase Smokers’ Risk of Stroke
Among current smokers, migraine may increase the risk of stroke and combined vascular events, according to research published online ahead of print July 22 in Neurology. Migraine may not be associated with these outcomes among nonsmokers, however.
Teshamae S. Monteith, MD, Assistant Professor of Clinical Neurology at University of Miami School of Medicine, and colleagues found that study participants with migraine had twice the risk of silent brain infarctions, but they considered the findings to be consistent with previous data that suggest that migraine is not a significant risk factor for stroke among older subjects. “We thought that factors associated with a greater migraine burden, such as obesity, might put migraineurs more at risk of vascular events, but this was not the case,” said Dr. Monteith.
Cohort Was Ethnically Diverse
Data have suggested that migraine with aura is an independent risk factor for ischemic stroke in women younger than 45. Migraine also has been associated with an unfavorable cardiovascular risk profile. Dr. Monteith and colleagues initiated their study to assess the association between migraine with and without aura and stroke. They examined data from the prospective Northern Manhattan Study, which enrolled an ethnically diverse, older, community-based cohort.
Eligible participants were stroke-free, older than 40, and had lived in northern Manhattan for three months or longer. Dr. Monteith’s group excluded participants with a history of meningitis, head trauma, or radiation to rule out individuals with the potential for secondary headache. They also excluded people with a myocardial infarction before baseline.
Bilingual research assistants collected data through interviews in English or Spanish. Baseline data included demographics, socioeconomic factors, medical history and medication use, vascular risk factors, family history, and migraine history. The investigators adapted standard questions regarding hypertension, diabetes, cigarette smoking, and cardiac conditions using the Centers for Disease Control and Prevention’s Behavioral Risk Factor Surveillance System. Self-reported migraine was assessed with a questionnaire, and additional questions closely adhered to the International Classification of Headache Disorders, second edition criteria for migraine.
Study participants were screened annually by phone for changes in clinical status, and patients who screened positive were invited for an interview and examination by a neurologist. The primary outcome was adjudicated stroke. The secondary outcomes were confirmed combined vascular events (ie, stroke, myocardial infarction, or vascular death), myocardial infarction, and vascular death.
Migraine Tripled Smokers’ Stroke Risk
The researchers had information on migraine status for 1,292 participants. Among this population, 262 participants had migraine (75 with aura, 187 without aura). People with migraine were younger and more likely to be women and have Medicaid or no insurance and several vascular risk factors. Over a mean follow-up of 11 years, the researchers observed 294 combined vascular events, including 114 strokes, 94 myocardial infarctions, and 178 vascular deaths.
Migraine was not associated with risk of combined vascular events including stroke or stroke-only outcomes. When they examined migraine with aura and migraine without aura separately, the investigators found no associations in relation to combined vascular events including stroke or to stroke alone. Age at baseline, sex, race or ethnicity, smoking, moderate alcohol use, moderate to heavy physical activity, BMI, hypertension, hypercholesterolemia, or diabetes did not modify the effect.
The researchers did, however, observe an interaction between current smoking and stroke. They also found an interaction between current smoking and combined vascular events. A stratified analysis yielded a hazard ratio of stroke for migraine versus no migraine among current smokers of 3.17. Among former smokers, the hazard ratio was 0.87, and among participants who had never smoked, the hazard ratio was 0.49 when controlling for socioeconomic and vascular risk factors.
Mechanism of Increased Risk Is Unclear
Previous research has indicated that migraine with aura is an independent risk factor of recurrent ischemic stroke and other vascular events in young patients with ischemic stroke. In addition, stroke risk associated with migraine with aura was greater in younger than in older women in the Women’s Health Study. “Perhaps our participants were too old to display such a relationship between migraine with aura and stroke in both men and women of postmenopausal age,” said Dr. Monteith.
Oxidative stress may be the mechanism by which migraine increases stroke risk among smokers. Oxidative stress may have a role in migraine and may increase susceptibility to vascular events among active smokers. Furthermore, prothrombotic states, decreased platelet hemostasis time, and endothelial dysfunction, which are associated with migraine, are plausible mechanisms that may enhance stroke risk in active smokers. “We suspect that a synergic action may occur between vascular changes of migraine and smoking as an effect modifier, although further work is necessary to elucidate this association,” said Dr. Monteith.
The data appear to suggest that vascular changes in migraine are an important subclinical vascular marker for stroke and combined vascular events among active smokers. Because smoking may be common among migraineurs, the authors recommended that smoking cessation counseling be encouraged as a part of routine migraine care throughout the patient’s lifetime. “The identification of modifiable vascular risk factors and treatments may have beneficial outcomes for stroke reduction in the elderly population with migraine,” they concluded.
—Erik Greb
Suggested Reading
Monteith TS, Gardener H, Rundek T, et al. Migraine and risk of stroke in older adults: Northern Manhattan Study. Neurology. 2015 Jul 22 [Epub ahead of print].
Among current smokers, migraine may increase the risk of stroke and combined vascular events, according to research published online ahead of print July 22 in Neurology. Migraine may not be associated with these outcomes among nonsmokers, however.
Teshamae S. Monteith, MD, Assistant Professor of Clinical Neurology at University of Miami School of Medicine, and colleagues found that study participants with migraine had twice the risk of silent brain infarctions, but they considered the findings to be consistent with previous data that suggest that migraine is not a significant risk factor for stroke among older subjects. “We thought that factors associated with a greater migraine burden, such as obesity, might put migraineurs more at risk of vascular events, but this was not the case,” said Dr. Monteith.
Cohort Was Ethnically Diverse
Data have suggested that migraine with aura is an independent risk factor for ischemic stroke in women younger than 45. Migraine also has been associated with an unfavorable cardiovascular risk profile. Dr. Monteith and colleagues initiated their study to assess the association between migraine with and without aura and stroke. They examined data from the prospective Northern Manhattan Study, which enrolled an ethnically diverse, older, community-based cohort.
Eligible participants were stroke-free, older than 40, and had lived in northern Manhattan for three months or longer. Dr. Monteith’s group excluded participants with a history of meningitis, head trauma, or radiation to rule out individuals with the potential for secondary headache. They also excluded people with a myocardial infarction before baseline.
Bilingual research assistants collected data through interviews in English or Spanish. Baseline data included demographics, socioeconomic factors, medical history and medication use, vascular risk factors, family history, and migraine history. The investigators adapted standard questions regarding hypertension, diabetes, cigarette smoking, and cardiac conditions using the Centers for Disease Control and Prevention’s Behavioral Risk Factor Surveillance System. Self-reported migraine was assessed with a questionnaire, and additional questions closely adhered to the International Classification of Headache Disorders, second edition criteria for migraine.
Study participants were screened annually by phone for changes in clinical status, and patients who screened positive were invited for an interview and examination by a neurologist. The primary outcome was adjudicated stroke. The secondary outcomes were confirmed combined vascular events (ie, stroke, myocardial infarction, or vascular death), myocardial infarction, and vascular death.
Migraine Tripled Smokers’ Stroke Risk
The researchers had information on migraine status for 1,292 participants. Among this population, 262 participants had migraine (75 with aura, 187 without aura). People with migraine were younger and more likely to be women and have Medicaid or no insurance and several vascular risk factors. Over a mean follow-up of 11 years, the researchers observed 294 combined vascular events, including 114 strokes, 94 myocardial infarctions, and 178 vascular deaths.
Migraine was not associated with risk of combined vascular events including stroke or stroke-only outcomes. When they examined migraine with aura and migraine without aura separately, the investigators found no associations in relation to combined vascular events including stroke or to stroke alone. Age at baseline, sex, race or ethnicity, smoking, moderate alcohol use, moderate to heavy physical activity, BMI, hypertension, hypercholesterolemia, or diabetes did not modify the effect.
The researchers did, however, observe an interaction between current smoking and stroke. They also found an interaction between current smoking and combined vascular events. A stratified analysis yielded a hazard ratio of stroke for migraine versus no migraine among current smokers of 3.17. Among former smokers, the hazard ratio was 0.87, and among participants who had never smoked, the hazard ratio was 0.49 when controlling for socioeconomic and vascular risk factors.
Mechanism of Increased Risk Is Unclear
Previous research has indicated that migraine with aura is an independent risk factor of recurrent ischemic stroke and other vascular events in young patients with ischemic stroke. In addition, stroke risk associated with migraine with aura was greater in younger than in older women in the Women’s Health Study. “Perhaps our participants were too old to display such a relationship between migraine with aura and stroke in both men and women of postmenopausal age,” said Dr. Monteith.
Oxidative stress may be the mechanism by which migraine increases stroke risk among smokers. Oxidative stress may have a role in migraine and may increase susceptibility to vascular events among active smokers. Furthermore, prothrombotic states, decreased platelet hemostasis time, and endothelial dysfunction, which are associated with migraine, are plausible mechanisms that may enhance stroke risk in active smokers. “We suspect that a synergic action may occur between vascular changes of migraine and smoking as an effect modifier, although further work is necessary to elucidate this association,” said Dr. Monteith.
The data appear to suggest that vascular changes in migraine are an important subclinical vascular marker for stroke and combined vascular events among active smokers. Because smoking may be common among migraineurs, the authors recommended that smoking cessation counseling be encouraged as a part of routine migraine care throughout the patient’s lifetime. “The identification of modifiable vascular risk factors and treatments may have beneficial outcomes for stroke reduction in the elderly population with migraine,” they concluded.
—Erik Greb
Among current smokers, migraine may increase the risk of stroke and combined vascular events, according to research published online ahead of print July 22 in Neurology. Migraine may not be associated with these outcomes among nonsmokers, however.
Teshamae S. Monteith, MD, Assistant Professor of Clinical Neurology at University of Miami School of Medicine, and colleagues found that study participants with migraine had twice the risk of silent brain infarctions, but they considered the findings to be consistent with previous data that suggest that migraine is not a significant risk factor for stroke among older subjects. “We thought that factors associated with a greater migraine burden, such as obesity, might put migraineurs more at risk of vascular events, but this was not the case,” said Dr. Monteith.
Cohort Was Ethnically Diverse
Data have suggested that migraine with aura is an independent risk factor for ischemic stroke in women younger than 45. Migraine also has been associated with an unfavorable cardiovascular risk profile. Dr. Monteith and colleagues initiated their study to assess the association between migraine with and without aura and stroke. They examined data from the prospective Northern Manhattan Study, which enrolled an ethnically diverse, older, community-based cohort.
Eligible participants were stroke-free, older than 40, and had lived in northern Manhattan for three months or longer. Dr. Monteith’s group excluded participants with a history of meningitis, head trauma, or radiation to rule out individuals with the potential for secondary headache. They also excluded people with a myocardial infarction before baseline.
Bilingual research assistants collected data through interviews in English or Spanish. Baseline data included demographics, socioeconomic factors, medical history and medication use, vascular risk factors, family history, and migraine history. The investigators adapted standard questions regarding hypertension, diabetes, cigarette smoking, and cardiac conditions using the Centers for Disease Control and Prevention’s Behavioral Risk Factor Surveillance System. Self-reported migraine was assessed with a questionnaire, and additional questions closely adhered to the International Classification of Headache Disorders, second edition criteria for migraine.
Study participants were screened annually by phone for changes in clinical status, and patients who screened positive were invited for an interview and examination by a neurologist. The primary outcome was adjudicated stroke. The secondary outcomes were confirmed combined vascular events (ie, stroke, myocardial infarction, or vascular death), myocardial infarction, and vascular death.
Migraine Tripled Smokers’ Stroke Risk
The researchers had information on migraine status for 1,292 participants. Among this population, 262 participants had migraine (75 with aura, 187 without aura). People with migraine were younger and more likely to be women and have Medicaid or no insurance and several vascular risk factors. Over a mean follow-up of 11 years, the researchers observed 294 combined vascular events, including 114 strokes, 94 myocardial infarctions, and 178 vascular deaths.
Migraine was not associated with risk of combined vascular events including stroke or stroke-only outcomes. When they examined migraine with aura and migraine without aura separately, the investigators found no associations in relation to combined vascular events including stroke or to stroke alone. Age at baseline, sex, race or ethnicity, smoking, moderate alcohol use, moderate to heavy physical activity, BMI, hypertension, hypercholesterolemia, or diabetes did not modify the effect.
The researchers did, however, observe an interaction between current smoking and stroke. They also found an interaction between current smoking and combined vascular events. A stratified analysis yielded a hazard ratio of stroke for migraine versus no migraine among current smokers of 3.17. Among former smokers, the hazard ratio was 0.87, and among participants who had never smoked, the hazard ratio was 0.49 when controlling for socioeconomic and vascular risk factors.
Mechanism of Increased Risk Is Unclear
Previous research has indicated that migraine with aura is an independent risk factor of recurrent ischemic stroke and other vascular events in young patients with ischemic stroke. In addition, stroke risk associated with migraine with aura was greater in younger than in older women in the Women’s Health Study. “Perhaps our participants were too old to display such a relationship between migraine with aura and stroke in both men and women of postmenopausal age,” said Dr. Monteith.
Oxidative stress may be the mechanism by which migraine increases stroke risk among smokers. Oxidative stress may have a role in migraine and may increase susceptibility to vascular events among active smokers. Furthermore, prothrombotic states, decreased platelet hemostasis time, and endothelial dysfunction, which are associated with migraine, are plausible mechanisms that may enhance stroke risk in active smokers. “We suspect that a synergic action may occur between vascular changes of migraine and smoking as an effect modifier, although further work is necessary to elucidate this association,” said Dr. Monteith.
The data appear to suggest that vascular changes in migraine are an important subclinical vascular marker for stroke and combined vascular events among active smokers. Because smoking may be common among migraineurs, the authors recommended that smoking cessation counseling be encouraged as a part of routine migraine care throughout the patient’s lifetime. “The identification of modifiable vascular risk factors and treatments may have beneficial outcomes for stroke reduction in the elderly population with migraine,” they concluded.
—Erik Greb
Suggested Reading
Monteith TS, Gardener H, Rundek T, et al. Migraine and risk of stroke in older adults: Northern Manhattan Study. Neurology. 2015 Jul 22 [Epub ahead of print].
Suggested Reading
Monteith TS, Gardener H, Rundek T, et al. Migraine and risk of stroke in older adults: Northern Manhattan Study. Neurology. 2015 Jul 22 [Epub ahead of print].
Impact of bladder volume on radiation dose to the rectum in the definitive treatment of prostate cancer
Background and objective Our group created and routinely reviewed a dedicated prostate intensity-modulated radiation therapy (IMRT) delivery program. Previously, a retrospective review of our experience demonstrated that a larger bladder volume reduced radiation dose to the rectum. We conducted an observational study to confirm this relationship.
Methods Men receiving definitive radiation for prostate cancer were eligible for the study. Eligible patients received 2 computed axial tomography (CT) scans on the day of their planning CT scan: 1 with a full bladder and 1 with an empty bladder. On each CT data set, the prostate, rectum, bladder, penile bulb, and femoral heads were contoured. 2 IMRT plans were completed on each dataset: 1 by a medical dosimetrist and 1 by a medical physicist. The study plans targeted the prostate to 79.2 Gray (Gy) while respecting predefined dose tolerances to the other contoured structures. Rectal doses were compared on empty and full bladder CT data sets.
Results From June 29, 2010 to December 14, 2011, 17 full bladder data sets and 15 empty bladder data sets were available for analysis. Median change in bladder volume was 63 ml. Full vs empty bladder set-up was associated with a statistically significant reduction in the mean rectal dose of 25.41 Gy vs 27.6 Gy (P = .031).
Limitations Small sample size and small variations in bladder volumes.
Conclusions A greater bladder volume resulted in a reduced mean dose to the rectum irrespective of planning method.
Funding/sponsorship None
Click on the PDF icon at the top of this introduction to read the full article.
Background and objective Our group created and routinely reviewed a dedicated prostate intensity-modulated radiation therapy (IMRT) delivery program. Previously, a retrospective review of our experience demonstrated that a larger bladder volume reduced radiation dose to the rectum. We conducted an observational study to confirm this relationship.
Methods Men receiving definitive radiation for prostate cancer were eligible for the study. Eligible patients received 2 computed axial tomography (CT) scans on the day of their planning CT scan: 1 with a full bladder and 1 with an empty bladder. On each CT data set, the prostate, rectum, bladder, penile bulb, and femoral heads were contoured. 2 IMRT plans were completed on each dataset: 1 by a medical dosimetrist and 1 by a medical physicist. The study plans targeted the prostate to 79.2 Gray (Gy) while respecting predefined dose tolerances to the other contoured structures. Rectal doses were compared on empty and full bladder CT data sets.
Results From June 29, 2010 to December 14, 2011, 17 full bladder data sets and 15 empty bladder data sets were available for analysis. Median change in bladder volume was 63 ml. Full vs empty bladder set-up was associated with a statistically significant reduction in the mean rectal dose of 25.41 Gy vs 27.6 Gy (P = .031).
Limitations Small sample size and small variations in bladder volumes.
Conclusions A greater bladder volume resulted in a reduced mean dose to the rectum irrespective of planning method.
Funding/sponsorship None
Click on the PDF icon at the top of this introduction to read the full article.
Background and objective Our group created and routinely reviewed a dedicated prostate intensity-modulated radiation therapy (IMRT) delivery program. Previously, a retrospective review of our experience demonstrated that a larger bladder volume reduced radiation dose to the rectum. We conducted an observational study to confirm this relationship.
Methods Men receiving definitive radiation for prostate cancer were eligible for the study. Eligible patients received 2 computed axial tomography (CT) scans on the day of their planning CT scan: 1 with a full bladder and 1 with an empty bladder. On each CT data set, the prostate, rectum, bladder, penile bulb, and femoral heads were contoured. 2 IMRT plans were completed on each dataset: 1 by a medical dosimetrist and 1 by a medical physicist. The study plans targeted the prostate to 79.2 Gray (Gy) while respecting predefined dose tolerances to the other contoured structures. Rectal doses were compared on empty and full bladder CT data sets.
Results From June 29, 2010 to December 14, 2011, 17 full bladder data sets and 15 empty bladder data sets were available for analysis. Median change in bladder volume was 63 ml. Full vs empty bladder set-up was associated with a statistically significant reduction in the mean rectal dose of 25.41 Gy vs 27.6 Gy (P = .031).
Limitations Small sample size and small variations in bladder volumes.
Conclusions A greater bladder volume resulted in a reduced mean dose to the rectum irrespective of planning method.
Funding/sponsorship None
Click on the PDF icon at the top of this introduction to read the full article.
Lung cancer in HIV-infected patients and the role of targeted therapy
Lung cancer is one of the most common malignancies in HIV-infected patients. Prevalence and mortality outcomes are higher in HIV-infected populations than in noninfected patients. There are several oral agents available for patients who harbor specific mutations, but little is known about mutations and affected pathways in HIV-infected patients with lung cancer. Recent trials have facilitated the inclusion of HIV-infected patients in clinical trials, but the population is remains underrepresented in oncology trials. Here, we review the literature on lung cancer in HIV-infected patients, and discuss common mutations in lung cancer and HIV-infected patients, the role of mutational analysis, and the potential role of targeted therapy in the treatment of lung cancer in HIV-infected populations.
Click on the PDF icon at the top of this introduction to read the full article.
Lung cancer is one of the most common malignancies in HIV-infected patients. Prevalence and mortality outcomes are higher in HIV-infected populations than in noninfected patients. There are several oral agents available for patients who harbor specific mutations, but little is known about mutations and affected pathways in HIV-infected patients with lung cancer. Recent trials have facilitated the inclusion of HIV-infected patients in clinical trials, but the population is remains underrepresented in oncology trials. Here, we review the literature on lung cancer in HIV-infected patients, and discuss common mutations in lung cancer and HIV-infected patients, the role of mutational analysis, and the potential role of targeted therapy in the treatment of lung cancer in HIV-infected populations.
Click on the PDF icon at the top of this introduction to read the full article.
Lung cancer is one of the most common malignancies in HIV-infected patients. Prevalence and mortality outcomes are higher in HIV-infected populations than in noninfected patients. There are several oral agents available for patients who harbor specific mutations, but little is known about mutations and affected pathways in HIV-infected patients with lung cancer. Recent trials have facilitated the inclusion of HIV-infected patients in clinical trials, but the population is remains underrepresented in oncology trials. Here, we review the literature on lung cancer in HIV-infected patients, and discuss common mutations in lung cancer and HIV-infected patients, the role of mutational analysis, and the potential role of targeted therapy in the treatment of lung cancer in HIV-infected populations.
Click on the PDF icon at the top of this introduction to read the full article.
FDA expands use of eltrombopag
Photo courtesy of GSK
The US Food and Drug Administration (FDA) has approved an expanded use for eltrombopag (Promacta) to include children 1 year of age and older with chronic immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy.
The updated label also includes a new oral suspension formulation of eltrombopag designed for younger children who may not be able to swallow tablets.
Eltrombopag was previously approved by the FDA in a tablet formulation in June 2015 for ITP patients ages 6 and older and in 2008 for use in adults with ITP.
The label expansion of eltrombopag was based on data from 2 double-blind, placebo-controlled trials—the phase 2 PETIT trial and the phase 3 PETIT2 trial.
PETIT trials: Efficacy
The PETIT trial included 67 ITP patients stratified by age cohort (12-17 years, 6-11 years, and 1-5 years). They were randomized (2:1) to receive eltrombopag or placebo for 7 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects achieving platelet counts of 50 x 109/L or higher at least once between days 8 and 43 of the randomized period of the study.
Significantly more patients in the eltrombopag arm met this endpoint—62.2%—compared to 31.8% in the placebo arm (P=0.011).
The PETIT2 trial enrolled 92 patients with chronic ITP who were randomized (2:1) to receive eltrombopag or placebo for 13 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects who achieved platelet counts of 50 x 109/L or higher for at least 6 out of 8 weeks, between weeks 5 and 12 of the randomized period.
Significantly more patients in the eltrombopag arm met this endpoint—41.3%—compared to 3.4% of patients in the placebo arm (P<0.001).
PETIT trials: Safety
For both trials, there were 107 eltrombopag-treated patients evaluable for safety.
The most common adverse events that occurred more frequently in the eltrombopag arms than the placebo arms were upper respiratory tract infection, nasopharyngitis, cough, diarrhea, pyrexia, rhinitis, abdominal pain, oropharyngeal pain, toothache, increased ALT or AST, rash, and rhinorrhea.
Serious adverse events were reported in 8% of patients during the randomized part of both trials, although no serious adverse event occurred in more than 1 patient (1%).
An ALT elevation of at least 3 times the upper limit of normal occurred in 5% of eltrombopag-treated patients. Of those patients, 2% had ALT increases of at least 5 times the upper limit of normal.
There were no deaths or thromboembolic events during either study.
Prescribing information
The recommended dose and schedule of eltrombopag for pediatric patients age 6 and older is 50 mg daily or 25 mg daily of the tablet formulation for patients with East Asian ancestry. The recommended dose for all patients age 1 to 5 years is 25 mg daily of the powder for oral suspension formulation.
Eltrombopag is marketed as Promacta in the US and Revolade in most other countries. For more information on the drug, see the full prescribing information. ![]()

Photo courtesy of GSK
The US Food and Drug Administration (FDA) has approved an expanded use for eltrombopag (Promacta) to include children 1 year of age and older with chronic immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy.
The updated label also includes a new oral suspension formulation of eltrombopag designed for younger children who may not be able to swallow tablets.
Eltrombopag was previously approved by the FDA in a tablet formulation in June 2015 for ITP patients ages 6 and older and in 2008 for use in adults with ITP.
The label expansion of eltrombopag was based on data from 2 double-blind, placebo-controlled trials—the phase 2 PETIT trial and the phase 3 PETIT2 trial.
PETIT trials: Efficacy
The PETIT trial included 67 ITP patients stratified by age cohort (12-17 years, 6-11 years, and 1-5 years). They were randomized (2:1) to receive eltrombopag or placebo for 7 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects achieving platelet counts of 50 x 109/L or higher at least once between days 8 and 43 of the randomized period of the study.
Significantly more patients in the eltrombopag arm met this endpoint—62.2%—compared to 31.8% in the placebo arm (P=0.011).
The PETIT2 trial enrolled 92 patients with chronic ITP who were randomized (2:1) to receive eltrombopag or placebo for 13 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects who achieved platelet counts of 50 x 109/L or higher for at least 6 out of 8 weeks, between weeks 5 and 12 of the randomized period.
Significantly more patients in the eltrombopag arm met this endpoint—41.3%—compared to 3.4% of patients in the placebo arm (P<0.001).
PETIT trials: Safety
For both trials, there were 107 eltrombopag-treated patients evaluable for safety.
The most common adverse events that occurred more frequently in the eltrombopag arms than the placebo arms were upper respiratory tract infection, nasopharyngitis, cough, diarrhea, pyrexia, rhinitis, abdominal pain, oropharyngeal pain, toothache, increased ALT or AST, rash, and rhinorrhea.
Serious adverse events were reported in 8% of patients during the randomized part of both trials, although no serious adverse event occurred in more than 1 patient (1%).
An ALT elevation of at least 3 times the upper limit of normal occurred in 5% of eltrombopag-treated patients. Of those patients, 2% had ALT increases of at least 5 times the upper limit of normal.
There were no deaths or thromboembolic events during either study.
Prescribing information
The recommended dose and schedule of eltrombopag for pediatric patients age 6 and older is 50 mg daily or 25 mg daily of the tablet formulation for patients with East Asian ancestry. The recommended dose for all patients age 1 to 5 years is 25 mg daily of the powder for oral suspension formulation.
Eltrombopag is marketed as Promacta in the US and Revolade in most other countries. For more information on the drug, see the full prescribing information. ![]()

Photo courtesy of GSK
The US Food and Drug Administration (FDA) has approved an expanded use for eltrombopag (Promacta) to include children 1 year of age and older with chronic immune thrombocytopenia (ITP) who have had an insufficient response to corticosteroids, immunoglobulins, or splenectomy.
The updated label also includes a new oral suspension formulation of eltrombopag designed for younger children who may not be able to swallow tablets.
Eltrombopag was previously approved by the FDA in a tablet formulation in June 2015 for ITP patients ages 6 and older and in 2008 for use in adults with ITP.
The label expansion of eltrombopag was based on data from 2 double-blind, placebo-controlled trials—the phase 2 PETIT trial and the phase 3 PETIT2 trial.
PETIT trials: Efficacy
The PETIT trial included 67 ITP patients stratified by age cohort (12-17 years, 6-11 years, and 1-5 years). They were randomized (2:1) to receive eltrombopag or placebo for 7 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects achieving platelet counts of 50 x 109/L or higher at least once between days 8 and 43 of the randomized period of the study.
Significantly more patients in the eltrombopag arm met this endpoint—62.2%—compared to 31.8% in the placebo arm (P=0.011).
The PETIT2 trial enrolled 92 patients with chronic ITP who were randomized (2:1) to receive eltrombopag or placebo for 13 weeks. The eltrombopag dose was titrated to a target platelet count of 50-200 x 109/L.
The primary efficacy endpoint was the proportion of subjects who achieved platelet counts of 50 x 109/L or higher for at least 6 out of 8 weeks, between weeks 5 and 12 of the randomized period.
Significantly more patients in the eltrombopag arm met this endpoint—41.3%—compared to 3.4% of patients in the placebo arm (P<0.001).
PETIT trials: Safety
For both trials, there were 107 eltrombopag-treated patients evaluable for safety.
The most common adverse events that occurred more frequently in the eltrombopag arms than the placebo arms were upper respiratory tract infection, nasopharyngitis, cough, diarrhea, pyrexia, rhinitis, abdominal pain, oropharyngeal pain, toothache, increased ALT or AST, rash, and rhinorrhea.
Serious adverse events were reported in 8% of patients during the randomized part of both trials, although no serious adverse event occurred in more than 1 patient (1%).
An ALT elevation of at least 3 times the upper limit of normal occurred in 5% of eltrombopag-treated patients. Of those patients, 2% had ALT increases of at least 5 times the upper limit of normal.
There were no deaths or thromboembolic events during either study.
Prescribing information
The recommended dose and schedule of eltrombopag for pediatric patients age 6 and older is 50 mg daily or 25 mg daily of the tablet formulation for patients with East Asian ancestry. The recommended dose for all patients age 1 to 5 years is 25 mg daily of the powder for oral suspension formulation.
Eltrombopag is marketed as Promacta in the US and Revolade in most other countries. For more information on the drug, see the full prescribing information. ![]()
Enzyme may be target for malaria, toxoplasmosis
Image by Ke Hu & John Murray
Researchers say they have determined the structure of an enzyme that is vital to the infectious behavior of the parasites that cause toxoplasmosis and malaria.
And this has revealed a potentially druggable target that could prevent the parasites from entering and exiting host cells.
Sebastian Lourido, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts, and his colleagues described this work in PNAS.
The researchers noted that the toxoplasmosis-causing parasite Toxoplasma gondii is closely related to the malaria-causing Plasmodium parasites. So research on T gondii can provide insights into Plasmodium’s inner workings.
For this study, Dr Lourido and his colleagues wanted to learn more about calcium-dependent protein kinases (CDPKs), enzymes that are needed for T gondii and related parasites to invade and exit host cells, move, and reproduce.
To investigate CDPKs, the team used single-domain antibody fragments derived from alpacas. Unlike humans, whose antibodies have a heavy chain and a light chain, alpacas create heavy-chain-only antibodies, which can be engineered into even smaller antibody fragments known as nanobodies.
Alpaca nanobodies have a unique shape that allows them to reach into a protein’s nooks and crannies, which are inaccessible to conventional antibodies.
The researchers identified a nanobody against the T gondii enzyme CDPK1 that binds the kinase’s regulatory domain and revealed a previously unappreciated feature of its activation.
The nanobody, called 1B7, stabilizes CDPK1 in a conformation that allowed the researchers to determine the kinase’s structure and describe the nanobody’s interaction with the molecule.
With the structure in hand, the team created long-timescale molecular dynamics simulations of the enzyme, to model the events leading to kinase inactivation.
Structural homology between CDPKs and the calmodulin-dependent kinases (CaMKs) found in humans led to earlier assumptions that both types of enzymes are activated in a similar fashion. But this new work shows otherwise.
A CaMK is activated when a wedge holding it in an inactive state is knocked away. In contrast, Dr Lourido likened a CDPK’s active conformation to a broken arm that must be splinted in two places to maintain its integrity.
When the rigid splint is removed, the kinase loses its structural ability to function. By blocking CDPK1’s regulatory domain, the 1B7 nanobody inhibits the kinase by preventing the enzyme’s “splint” from attaching.
“This work reveals something interesting about this class of enzymes,” Dr Lourido said. “It’s the first time a calcium-regulated kinase has been shown to be activated in this manner. The principle that we identify is really important. We’ve found a new vulnerability within an enzyme that we know is extremely important to this class of parasites, including Plasmodium . . . , and is absent from humans.”
Because humans lack similar kinases, drugs that target CDPKs would not affect host cells.
“The location where 1B7 binds to CDPK1 is a new drug target that people had not considered before,” said study author Jessica Ingram, PhD, also of the Whitehead Institute for Biomedical Research.
“We’d like to do some drug screens in the presence of the nanobody to see if we can find small molecules that bind in the same way. We could also look at other nanobodies against other kinases to see if this is applicable to other parasites and systems.” ![]()

Image by Ke Hu & John Murray
Researchers say they have determined the structure of an enzyme that is vital to the infectious behavior of the parasites that cause toxoplasmosis and malaria.
And this has revealed a potentially druggable target that could prevent the parasites from entering and exiting host cells.
Sebastian Lourido, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts, and his colleagues described this work in PNAS.
The researchers noted that the toxoplasmosis-causing parasite Toxoplasma gondii is closely related to the malaria-causing Plasmodium parasites. So research on T gondii can provide insights into Plasmodium’s inner workings.
For this study, Dr Lourido and his colleagues wanted to learn more about calcium-dependent protein kinases (CDPKs), enzymes that are needed for T gondii and related parasites to invade and exit host cells, move, and reproduce.
To investigate CDPKs, the team used single-domain antibody fragments derived from alpacas. Unlike humans, whose antibodies have a heavy chain and a light chain, alpacas create heavy-chain-only antibodies, which can be engineered into even smaller antibody fragments known as nanobodies.
Alpaca nanobodies have a unique shape that allows them to reach into a protein’s nooks and crannies, which are inaccessible to conventional antibodies.
The researchers identified a nanobody against the T gondii enzyme CDPK1 that binds the kinase’s regulatory domain and revealed a previously unappreciated feature of its activation.
The nanobody, called 1B7, stabilizes CDPK1 in a conformation that allowed the researchers to determine the kinase’s structure and describe the nanobody’s interaction with the molecule.
With the structure in hand, the team created long-timescale molecular dynamics simulations of the enzyme, to model the events leading to kinase inactivation.
Structural homology between CDPKs and the calmodulin-dependent kinases (CaMKs) found in humans led to earlier assumptions that both types of enzymes are activated in a similar fashion. But this new work shows otherwise.
A CaMK is activated when a wedge holding it in an inactive state is knocked away. In contrast, Dr Lourido likened a CDPK’s active conformation to a broken arm that must be splinted in two places to maintain its integrity.
When the rigid splint is removed, the kinase loses its structural ability to function. By blocking CDPK1’s regulatory domain, the 1B7 nanobody inhibits the kinase by preventing the enzyme’s “splint” from attaching.
“This work reveals something interesting about this class of enzymes,” Dr Lourido said. “It’s the first time a calcium-regulated kinase has been shown to be activated in this manner. The principle that we identify is really important. We’ve found a new vulnerability within an enzyme that we know is extremely important to this class of parasites, including Plasmodium . . . , and is absent from humans.”
Because humans lack similar kinases, drugs that target CDPKs would not affect host cells.
“The location where 1B7 binds to CDPK1 is a new drug target that people had not considered before,” said study author Jessica Ingram, PhD, also of the Whitehead Institute for Biomedical Research.
“We’d like to do some drug screens in the presence of the nanobody to see if we can find small molecules that bind in the same way. We could also look at other nanobodies against other kinases to see if this is applicable to other parasites and systems.” ![]()

Image by Ke Hu & John Murray
Researchers say they have determined the structure of an enzyme that is vital to the infectious behavior of the parasites that cause toxoplasmosis and malaria.
And this has revealed a potentially druggable target that could prevent the parasites from entering and exiting host cells.
Sebastian Lourido, PhD, of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts, and his colleagues described this work in PNAS.
The researchers noted that the toxoplasmosis-causing parasite Toxoplasma gondii is closely related to the malaria-causing Plasmodium parasites. So research on T gondii can provide insights into Plasmodium’s inner workings.
For this study, Dr Lourido and his colleagues wanted to learn more about calcium-dependent protein kinases (CDPKs), enzymes that are needed for T gondii and related parasites to invade and exit host cells, move, and reproduce.
To investigate CDPKs, the team used single-domain antibody fragments derived from alpacas. Unlike humans, whose antibodies have a heavy chain and a light chain, alpacas create heavy-chain-only antibodies, which can be engineered into even smaller antibody fragments known as nanobodies.
Alpaca nanobodies have a unique shape that allows them to reach into a protein’s nooks and crannies, which are inaccessible to conventional antibodies.
The researchers identified a nanobody against the T gondii enzyme CDPK1 that binds the kinase’s regulatory domain and revealed a previously unappreciated feature of its activation.
The nanobody, called 1B7, stabilizes CDPK1 in a conformation that allowed the researchers to determine the kinase’s structure and describe the nanobody’s interaction with the molecule.
With the structure in hand, the team created long-timescale molecular dynamics simulations of the enzyme, to model the events leading to kinase inactivation.
Structural homology between CDPKs and the calmodulin-dependent kinases (CaMKs) found in humans led to earlier assumptions that both types of enzymes are activated in a similar fashion. But this new work shows otherwise.
A CaMK is activated when a wedge holding it in an inactive state is knocked away. In contrast, Dr Lourido likened a CDPK’s active conformation to a broken arm that must be splinted in two places to maintain its integrity.
When the rigid splint is removed, the kinase loses its structural ability to function. By blocking CDPK1’s regulatory domain, the 1B7 nanobody inhibits the kinase by preventing the enzyme’s “splint” from attaching.
“This work reveals something interesting about this class of enzymes,” Dr Lourido said. “It’s the first time a calcium-regulated kinase has been shown to be activated in this manner. The principle that we identify is really important. We’ve found a new vulnerability within an enzyme that we know is extremely important to this class of parasites, including Plasmodium . . . , and is absent from humans.”
Because humans lack similar kinases, drugs that target CDPKs would not affect host cells.
“The location where 1B7 binds to CDPK1 is a new drug target that people had not considered before,” said study author Jessica Ingram, PhD, also of the Whitehead Institute for Biomedical Research.
“We’d like to do some drug screens in the presence of the nanobody to see if we can find small molecules that bind in the same way. We could also look at other nanobodies against other kinases to see if this is applicable to other parasites and systems.” ![]()
Drug gets orphan designation for CDI
The US Food and Drug Administration (FDA) has granted orphan designation to SER-109 for the prevention of recurrent Clostridium difficile infection (CDI) in adults.
SER-109 is a microbiome therapeutic designed to treat recurrent CDI by correcting dysbiosis of the human microbiome.
In a single dose of 4 capsules, SER-109 re-introduces an ecology of purified bacterial spores that should restore the microbiome to a healthy state, allowing it to carry out key biological functions, including resisting Clostridium difficile.
“SER-109 is intended to re-introduce essential bacteria that restore the body’s natural resistance to CDI by re-establishing the ecology of the colonic microbiome,” explained Roger Pomerantz, MD, of Seres Therapeutics, Inc., the company developing SER-109.
“Because we’re focused on treating the underlying cause of the disease, we believe we have the potential to break the cycle of recurrent CDI and have a significant impact for patients.”
SER-109 is currently being investigated in a phase 2 trial. In addition to orphan designation, SER-109 has breakthrough designation from the FDA.
Trials of SER-109
Researchers reported phase 1/2 results with SER-109 at the 2014 Interscience Conference on Antimicrobial Agents and Chemotherapy.
The study had 2 cohorts containing 15 patients each. Patients were between 18 and 90 years old, had 3 or more laboratory-confirmed CDI episodes over 1 year, had a life expectancy greater than 3 months, and were able to give informed consent.
Patients in cohort 1 received a mean SER-109 dose of 1.5 x 109 spores, and those in cohort 2 received a mean dose of 1 x 108 spores. SER-109 was deemed effective if patients did not have a CDI recurrence in the 8-week period after they received SER-109.
In cohort 1, 87% of patients (13/15) achieved the efficacy endpoint. Two patients had transient, self-limited diarrhea with a positive C difficile test, but both reached the week 8 endpoint without needing antibiotic therapy for CDI. Thus, in cohort 1, the clinical cure rate was 100%.
In cohort 2, 93% of patients (14/15) reached the 8-week endpoint CDI-free. One patient failed per protocol.
The researchers said there were no drug-related serious adverse events in this trial.
Seres Therapeutics is currently conducting a multicenter, randomized, placebo-controlled, phase 2 study (ECOSPOR) to assess the efficacy and safety of SER-109 in preventing recurrent CDI. The company expects results from this study to be available mid-2016.
About orphan and breakthrough designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
Orphan designation provides the sponsor of a drug with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US marketing exclusivity if the drug is approved.
The FDA’s breakthrough therapy designation is intended to expedite the development and review of a drug candidate intended to treat a serious or life-threatening condition.
The benefits of breakthrough designation include the same benefits as fast track designation—priority review of a new drug application, rolling review, etc.—plus an organizational commitment involving the FDA’s senior managers with more intensive guidance from the FDA. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation to SER-109 for the prevention of recurrent Clostridium difficile infection (CDI) in adults.
SER-109 is a microbiome therapeutic designed to treat recurrent CDI by correcting dysbiosis of the human microbiome.
In a single dose of 4 capsules, SER-109 re-introduces an ecology of purified bacterial spores that should restore the microbiome to a healthy state, allowing it to carry out key biological functions, including resisting Clostridium difficile.
“SER-109 is intended to re-introduce essential bacteria that restore the body’s natural resistance to CDI by re-establishing the ecology of the colonic microbiome,” explained Roger Pomerantz, MD, of Seres Therapeutics, Inc., the company developing SER-109.
“Because we’re focused on treating the underlying cause of the disease, we believe we have the potential to break the cycle of recurrent CDI and have a significant impact for patients.”
SER-109 is currently being investigated in a phase 2 trial. In addition to orphan designation, SER-109 has breakthrough designation from the FDA.
Trials of SER-109
Researchers reported phase 1/2 results with SER-109 at the 2014 Interscience Conference on Antimicrobial Agents and Chemotherapy.
The study had 2 cohorts containing 15 patients each. Patients were between 18 and 90 years old, had 3 or more laboratory-confirmed CDI episodes over 1 year, had a life expectancy greater than 3 months, and were able to give informed consent.
Patients in cohort 1 received a mean SER-109 dose of 1.5 x 109 spores, and those in cohort 2 received a mean dose of 1 x 108 spores. SER-109 was deemed effective if patients did not have a CDI recurrence in the 8-week period after they received SER-109.
In cohort 1, 87% of patients (13/15) achieved the efficacy endpoint. Two patients had transient, self-limited diarrhea with a positive C difficile test, but both reached the week 8 endpoint without needing antibiotic therapy for CDI. Thus, in cohort 1, the clinical cure rate was 100%.
In cohort 2, 93% of patients (14/15) reached the 8-week endpoint CDI-free. One patient failed per protocol.
The researchers said there were no drug-related serious adverse events in this trial.
Seres Therapeutics is currently conducting a multicenter, randomized, placebo-controlled, phase 2 study (ECOSPOR) to assess the efficacy and safety of SER-109 in preventing recurrent CDI. The company expects results from this study to be available mid-2016.
About orphan and breakthrough designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
Orphan designation provides the sponsor of a drug with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US marketing exclusivity if the drug is approved.
The FDA’s breakthrough therapy designation is intended to expedite the development and review of a drug candidate intended to treat a serious or life-threatening condition.
The benefits of breakthrough designation include the same benefits as fast track designation—priority review of a new drug application, rolling review, etc.—plus an organizational commitment involving the FDA’s senior managers with more intensive guidance from the FDA. ![]()

The US Food and Drug Administration (FDA) has granted orphan designation to SER-109 for the prevention of recurrent Clostridium difficile infection (CDI) in adults.
SER-109 is a microbiome therapeutic designed to treat recurrent CDI by correcting dysbiosis of the human microbiome.
In a single dose of 4 capsules, SER-109 re-introduces an ecology of purified bacterial spores that should restore the microbiome to a healthy state, allowing it to carry out key biological functions, including resisting Clostridium difficile.
“SER-109 is intended to re-introduce essential bacteria that restore the body’s natural resistance to CDI by re-establishing the ecology of the colonic microbiome,” explained Roger Pomerantz, MD, of Seres Therapeutics, Inc., the company developing SER-109.
“Because we’re focused on treating the underlying cause of the disease, we believe we have the potential to break the cycle of recurrent CDI and have a significant impact for patients.”
SER-109 is currently being investigated in a phase 2 trial. In addition to orphan designation, SER-109 has breakthrough designation from the FDA.
Trials of SER-109
Researchers reported phase 1/2 results with SER-109 at the 2014 Interscience Conference on Antimicrobial Agents and Chemotherapy.
The study had 2 cohorts containing 15 patients each. Patients were between 18 and 90 years old, had 3 or more laboratory-confirmed CDI episodes over 1 year, had a life expectancy greater than 3 months, and were able to give informed consent.
Patients in cohort 1 received a mean SER-109 dose of 1.5 x 109 spores, and those in cohort 2 received a mean dose of 1 x 108 spores. SER-109 was deemed effective if patients did not have a CDI recurrence in the 8-week period after they received SER-109.
In cohort 1, 87% of patients (13/15) achieved the efficacy endpoint. Two patients had transient, self-limited diarrhea with a positive C difficile test, but both reached the week 8 endpoint without needing antibiotic therapy for CDI. Thus, in cohort 1, the clinical cure rate was 100%.
In cohort 2, 93% of patients (14/15) reached the 8-week endpoint CDI-free. One patient failed per protocol.
The researchers said there were no drug-related serious adverse events in this trial.
Seres Therapeutics is currently conducting a multicenter, randomized, placebo-controlled, phase 2 study (ECOSPOR) to assess the efficacy and safety of SER-109 in preventing recurrent CDI. The company expects results from this study to be available mid-2016.
About orphan and breakthrough designation
The FDA grants orphan designation to drugs that are intended to treat diseases or conditions affecting fewer than 200,000 patients in the US.
Orphan designation provides the sponsor of a drug with various development incentives, including opportunities to apply for research-related tax credits and grant funding, assistance in designing clinical trials, and 7 years of US marketing exclusivity if the drug is approved.
The FDA’s breakthrough therapy designation is intended to expedite the development and review of a drug candidate intended to treat a serious or life-threatening condition.
The benefits of breakthrough designation include the same benefits as fast track designation—priority review of a new drug application, rolling review, etc.—plus an organizational commitment involving the FDA’s senior managers with more intensive guidance from the FDA. ![]()
BET inhibitor appears to cause memory loss in mice

Photo by Aaron Logan
New research suggests the BET inhibitor JQ1 causes molecular changes in mouse neurons and can lead to memory loss in mice.
Investigators believe this discovery, published in Nature Neuroscience, will fuel more research into the neurological effects of BET inhibitors, which are currently under development as potential treatments for a range of hematologic and solid tumor malignancies.
The researchers noted that, although JQ1 has the ability to cross the blood-brain barrier, this may not be the case for other BET inhibitors.
Several companies are testing the inhibitors using unique formulations they’ve optimized in proprietary ways—for example, by adding chemical groups to make a compound more targeted or effective—which might make it more difficult for the drug to cross the blood-brain barrier.
Still, the investigators said their findings suggests more research is needed to determine whether other BET inhibitors can enter the brain, since that could potentially cause unwanted side effects.
“We found that if a drug blocks a BET protein throughout the body, and that drug can get into the brain, you could very well produce neurological side effects,” said study author Erica Korb, PhD, of The Rockefeller University in New York, New York.
Experiments with JQ1
To assess the effects of BET inhibitors on the brain, the researchers used a compound that was designed to thwart the activity of a specific BET protein, Brd4. They used the drug JQ1, which they knew could cross the blood-brain barrier.
The investigators added the drug to mouse neurons grown in the lab, then stimulated the cells in a way that mimicked the process of memory formation. Normally, when neurons receive this type of signal, they begin transcribing genes into proteins, resulting in the formation of new memories—a process that is partly regulated by Brd4.
“To turn a recent experience into a long-term memory, you need to have gene transcription in response to these extracellular signals,” Dr Korb said.
Indeed, when the researchers stimulated mouse neurons with signals that mimicked those they would normally receive in the brain, there were “massive changes” in gene transcription. But when the team performed this experiment after adding JQ1, they saw much less activity.
“After administering a Brd4 inhibitor, we no longer saw those changes in transcription after stimuli,” Dr Korb said.
To test how the drug affected the animals’ memories, the investigators placed the mice in a box with two objects they had never seen before, such as pieces of Lego or tiny figurines. Mice typically explore anything unfamiliar, climbing and sniffing around.
After a few minutes, the researchers took the mice out of the box. One day later, the team put the mice back in, this time with one of the objects from the day before and another, unfamiliar one.
Mice that received a placebo were much more interested in the new object, presumably because the one from the day before was familiar. But mice treated with JQ1 were equally interested in both objects, suggesting they didn’t remember the previous day’s experience.
Next, the investigators took their findings a step further. If JQ1 reduces molecular activity in the brain, they wondered if it could help in conditions marked by too much brain activity, such as epilepsy.
Brd4 regulates a receptor protein present at the synapse, a structure where two neurons connect and transmit signals. When the researchers administered the Brd4 inhibitor, they saw decreased levels of that receptor, and neurons fired much less frequently.
Next, the team gave the drug to mice for a week, then added a chemical that induces seizures. Mice that received JQ1 had a much lower rate of seizures than mice given a placebo.
“In the case of the epileptic brain, when there’s too much activity and neurons talking to each other, this drug could be potentially be beneficial,” Dr Korb concluded. ![]()

Photo by Aaron Logan
New research suggests the BET inhibitor JQ1 causes molecular changes in mouse neurons and can lead to memory loss in mice.
Investigators believe this discovery, published in Nature Neuroscience, will fuel more research into the neurological effects of BET inhibitors, which are currently under development as potential treatments for a range of hematologic and solid tumor malignancies.
The researchers noted that, although JQ1 has the ability to cross the blood-brain barrier, this may not be the case for other BET inhibitors.
Several companies are testing the inhibitors using unique formulations they’ve optimized in proprietary ways—for example, by adding chemical groups to make a compound more targeted or effective—which might make it more difficult for the drug to cross the blood-brain barrier.
Still, the investigators said their findings suggests more research is needed to determine whether other BET inhibitors can enter the brain, since that could potentially cause unwanted side effects.
“We found that if a drug blocks a BET protein throughout the body, and that drug can get into the brain, you could very well produce neurological side effects,” said study author Erica Korb, PhD, of The Rockefeller University in New York, New York.
Experiments with JQ1
To assess the effects of BET inhibitors on the brain, the researchers used a compound that was designed to thwart the activity of a specific BET protein, Brd4. They used the drug JQ1, which they knew could cross the blood-brain barrier.
The investigators added the drug to mouse neurons grown in the lab, then stimulated the cells in a way that mimicked the process of memory formation. Normally, when neurons receive this type of signal, they begin transcribing genes into proteins, resulting in the formation of new memories—a process that is partly regulated by Brd4.
“To turn a recent experience into a long-term memory, you need to have gene transcription in response to these extracellular signals,” Dr Korb said.
Indeed, when the researchers stimulated mouse neurons with signals that mimicked those they would normally receive in the brain, there were “massive changes” in gene transcription. But when the team performed this experiment after adding JQ1, they saw much less activity.
“After administering a Brd4 inhibitor, we no longer saw those changes in transcription after stimuli,” Dr Korb said.
To test how the drug affected the animals’ memories, the investigators placed the mice in a box with two objects they had never seen before, such as pieces of Lego or tiny figurines. Mice typically explore anything unfamiliar, climbing and sniffing around.
After a few minutes, the researchers took the mice out of the box. One day later, the team put the mice back in, this time with one of the objects from the day before and another, unfamiliar one.
Mice that received a placebo were much more interested in the new object, presumably because the one from the day before was familiar. But mice treated with JQ1 were equally interested in both objects, suggesting they didn’t remember the previous day’s experience.
Next, the investigators took their findings a step further. If JQ1 reduces molecular activity in the brain, they wondered if it could help in conditions marked by too much brain activity, such as epilepsy.
Brd4 regulates a receptor protein present at the synapse, a structure where two neurons connect and transmit signals. When the researchers administered the Brd4 inhibitor, they saw decreased levels of that receptor, and neurons fired much less frequently.
Next, the team gave the drug to mice for a week, then added a chemical that induces seizures. Mice that received JQ1 had a much lower rate of seizures than mice given a placebo.
“In the case of the epileptic brain, when there’s too much activity and neurons talking to each other, this drug could be potentially be beneficial,” Dr Korb concluded. ![]()

Photo by Aaron Logan
New research suggests the BET inhibitor JQ1 causes molecular changes in mouse neurons and can lead to memory loss in mice.
Investigators believe this discovery, published in Nature Neuroscience, will fuel more research into the neurological effects of BET inhibitors, which are currently under development as potential treatments for a range of hematologic and solid tumor malignancies.
The researchers noted that, although JQ1 has the ability to cross the blood-brain barrier, this may not be the case for other BET inhibitors.
Several companies are testing the inhibitors using unique formulations they’ve optimized in proprietary ways—for example, by adding chemical groups to make a compound more targeted or effective—which might make it more difficult for the drug to cross the blood-brain barrier.
Still, the investigators said their findings suggests more research is needed to determine whether other BET inhibitors can enter the brain, since that could potentially cause unwanted side effects.
“We found that if a drug blocks a BET protein throughout the body, and that drug can get into the brain, you could very well produce neurological side effects,” said study author Erica Korb, PhD, of The Rockefeller University in New York, New York.
Experiments with JQ1
To assess the effects of BET inhibitors on the brain, the researchers used a compound that was designed to thwart the activity of a specific BET protein, Brd4. They used the drug JQ1, which they knew could cross the blood-brain barrier.
The investigators added the drug to mouse neurons grown in the lab, then stimulated the cells in a way that mimicked the process of memory formation. Normally, when neurons receive this type of signal, they begin transcribing genes into proteins, resulting in the formation of new memories—a process that is partly regulated by Brd4.
“To turn a recent experience into a long-term memory, you need to have gene transcription in response to these extracellular signals,” Dr Korb said.
Indeed, when the researchers stimulated mouse neurons with signals that mimicked those they would normally receive in the brain, there were “massive changes” in gene transcription. But when the team performed this experiment after adding JQ1, they saw much less activity.
“After administering a Brd4 inhibitor, we no longer saw those changes in transcription after stimuli,” Dr Korb said.
To test how the drug affected the animals’ memories, the investigators placed the mice in a box with two objects they had never seen before, such as pieces of Lego or tiny figurines. Mice typically explore anything unfamiliar, climbing and sniffing around.
After a few minutes, the researchers took the mice out of the box. One day later, the team put the mice back in, this time with one of the objects from the day before and another, unfamiliar one.
Mice that received a placebo were much more interested in the new object, presumably because the one from the day before was familiar. But mice treated with JQ1 were equally interested in both objects, suggesting they didn’t remember the previous day’s experience.
Next, the investigators took their findings a step further. If JQ1 reduces molecular activity in the brain, they wondered if it could help in conditions marked by too much brain activity, such as epilepsy.
Brd4 regulates a receptor protein present at the synapse, a structure where two neurons connect and transmit signals. When the researchers administered the Brd4 inhibitor, they saw decreased levels of that receptor, and neurons fired much less frequently.
Next, the team gave the drug to mice for a week, then added a chemical that induces seizures. Mice that received JQ1 had a much lower rate of seizures than mice given a placebo.
“In the case of the epileptic brain, when there’s too much activity and neurons talking to each other, this drug could be potentially be beneficial,” Dr Korb concluded. ![]()
FDA expands Promacta approval to include pediatric patients
The Food and Drug Administration has extended its approval of Promacta (eltrombopag) to include pediatric patients with chronic immune thrombocytopenic purpura, the agency announced Aug. 24.
Approved in 2008 for adults with immune thrombocytopenic purpura (ITP), eltrombopag is now approved for the treatment of the rare blood disorder in patients aged 1 year and older. The drug may be used in children who have not responded to other ITP medications or spleen surgery, the FDA said in a statement.
Eltrombopag’s efficacy and safety in children aged 1-17 years was established in two placebo-controlled trials comprising 159 patients. Findings from the first trial found that over 7 weeks, 62% of patients given eltrombopag had improved platelet counts without rescue therapy between weeks 1 and 6, compared with 32% in the placebo group.
In the second trial, which lasted 13 weeks, 41% of patients taking eltrombopag experienced increased platelet counts for at least 6 out of 8 weeks between weeks 5 and 12, compared with 3% of patients in the placebo group, the FDA reported.
“In both trials, patients taking Promacta also had less need for other treatments to increase their platelet counts, such as corticosteroids or platelet transfusions,” the FDA said. “Among patients taking one or more ITP medications at the start of the trials, about half were able to reduce or discontinue their use of these medications, primarily corticosteroids.”
Eltrombopag may be taken once daily in tablet form, or as a powder mixed with liquid for children aged 1-5 years. It should be used only in ITP patients with an increased risk of bleeding.
The most common side effects in children were infections of the upper respiratory tract or nose and throat, diarrhea, abdominal pain, rash, and increase in liver enzymes.
Promacta is manufactured by Novartis in East Hanover, N.J.
The Food and Drug Administration has extended its approval of Promacta (eltrombopag) to include pediatric patients with chronic immune thrombocytopenic purpura, the agency announced Aug. 24.
Approved in 2008 for adults with immune thrombocytopenic purpura (ITP), eltrombopag is now approved for the treatment of the rare blood disorder in patients aged 1 year and older. The drug may be used in children who have not responded to other ITP medications or spleen surgery, the FDA said in a statement.
Eltrombopag’s efficacy and safety in children aged 1-17 years was established in two placebo-controlled trials comprising 159 patients. Findings from the first trial found that over 7 weeks, 62% of patients given eltrombopag had improved platelet counts without rescue therapy between weeks 1 and 6, compared with 32% in the placebo group.
In the second trial, which lasted 13 weeks, 41% of patients taking eltrombopag experienced increased platelet counts for at least 6 out of 8 weeks between weeks 5 and 12, compared with 3% of patients in the placebo group, the FDA reported.
“In both trials, patients taking Promacta also had less need for other treatments to increase their platelet counts, such as corticosteroids or platelet transfusions,” the FDA said. “Among patients taking one or more ITP medications at the start of the trials, about half were able to reduce or discontinue their use of these medications, primarily corticosteroids.”
Eltrombopag may be taken once daily in tablet form, or as a powder mixed with liquid for children aged 1-5 years. It should be used only in ITP patients with an increased risk of bleeding.
The most common side effects in children were infections of the upper respiratory tract or nose and throat, diarrhea, abdominal pain, rash, and increase in liver enzymes.
Promacta is manufactured by Novartis in East Hanover, N.J.
The Food and Drug Administration has extended its approval of Promacta (eltrombopag) to include pediatric patients with chronic immune thrombocytopenic purpura, the agency announced Aug. 24.
Approved in 2008 for adults with immune thrombocytopenic purpura (ITP), eltrombopag is now approved for the treatment of the rare blood disorder in patients aged 1 year and older. The drug may be used in children who have not responded to other ITP medications or spleen surgery, the FDA said in a statement.
Eltrombopag’s efficacy and safety in children aged 1-17 years was established in two placebo-controlled trials comprising 159 patients. Findings from the first trial found that over 7 weeks, 62% of patients given eltrombopag had improved platelet counts without rescue therapy between weeks 1 and 6, compared with 32% in the placebo group.
In the second trial, which lasted 13 weeks, 41% of patients taking eltrombopag experienced increased platelet counts for at least 6 out of 8 weeks between weeks 5 and 12, compared with 3% of patients in the placebo group, the FDA reported.
“In both trials, patients taking Promacta also had less need for other treatments to increase their platelet counts, such as corticosteroids or platelet transfusions,” the FDA said. “Among patients taking one or more ITP medications at the start of the trials, about half were able to reduce or discontinue their use of these medications, primarily corticosteroids.”
Eltrombopag may be taken once daily in tablet form, or as a powder mixed with liquid for children aged 1-5 years. It should be used only in ITP patients with an increased risk of bleeding.
The most common side effects in children were infections of the upper respiratory tract or nose and throat, diarrhea, abdominal pain, rash, and increase in liver enzymes.
Promacta is manufactured by Novartis in East Hanover, N.J.