User login
Is the United States addressing maternal mortality rates from preeclampsia/eclampsia and chronic hypertension?
Ananth CV, Brandt JS, Hill J, et al. Historical and recent changes in maternal mortality due to hypertensive disorders in the United States, 1979 to 2018. Hypertension. 2021;78:1414–1422. doi: 10.1161/HYPERTENSIONAHA.121.17661.
EXPERT COMMENTARY
Maternal mortality is a pressing public health issue and is largely preventable. Up to 10% of all US pregnancies are complicated by a hypertensive disorder, and rates of chronic hypertension and severe preeclampsia have steadily increased over the last 4 decades. However, maternal mortality is an outcome in a population with advancing maternal age, increasing obesity, and undermanaged chronic disease. The MMR due to hypertension is substantially higher among Black women compared with White women. Countless studies attribute systemic racism to these disparities.
Details of the study
Spanning 40 years, a recent study by Ananth and colleagues included live births across all 50 United States and Washington, DC. Of the 1.5 million live births examined, there were 3,287 hypertension-related maternal deaths.
Data were deidentified and available in the public domain. The researchers compiled mortality data and live births among women aged 15 to 49. The MMR was considered the death of a woman during pregnancy or within the 42 days following a live birth.
Key points of the study included:
- An estimated two-thirds of maternal deaths are preventable.
- The hypertension-related MMR was 2.1 per 100,000 live births.
- Preeclampsia-related MMR decreased, while hypertension-related MMR increased.
- The MMR from chronic hypertension has increased annually by 9.2%.
- Pregnancies among women with advanced maternal age have grown, especially among those over age 40.
- The MMR due to hypertension increases with age and is highest among women age 45 to 49.
Study strengths and limitations
A major strength of this study is the sheer size of the sample. This is one of the largest studies that examined changes in the MMR in the United States.
As with any study that spans a long period, a primary limitation is inconsistencies in the data collected. In 2003, the US death certificate was revised to include a set of “pregnancy checkboxes” indicating pregnancy at the time of death.
There also have been shifts in diagnostic coding and criteria for preeclampsia.
Classification of race and ethnicity has improved and broadened over time. Despite these limitations, the overarching trends are compelling. ●
This study’s authors note that maternal mortality is largely preventable. Patients need to be aware of their health and how to adopt healthy behaviors long before pregnancy is even a consideration. Primary and secondary prevention are essential for reducing the MMR.
Clinicians who care for women have an opportunity to emphasize cardiac health at every visit. This includes strict blood pressure control through modifiable behaviors like diet and exercise. The busy clinician could consider a 1- to 2-minute pitch to emphasize that heart disease is the leading cause of death in women both during pregnancy and later in life. A tool from the American Heart Association, Life’s Simple 7 (https://www.heart .org/en/healthy-living/healthy-lifestyle/my-life-check--lifes-sim ple-7), can help guide this language.
In office and clinical settings, consider strategies to raise awareness among staff and colleagues about cultural sensitivities to improve the health of all patients. Addressing systemic racism in the US health care system is critical to mitigate racial inequities in the rates of MMR. An editorial in The New England Journal of Medicine urges clinicians to observe patient color rather than be “color blind.”1 The editorialists note that “physician-citizens must recognize the harm inflicted by discrimination and racism and consider this environmental agent of disease as a vital sign— alongside blood pressure, pulse, weight, and temperature—that provides important information about a patient’s condition.”1
LAUREN B. GOLFER, WHNP-BC, AND MARY L. ROSSER, MD, PHD
- Evans MK, Rosenbaum L, Malina D, et al. Diagnosing and treating systemic racism. N Engl J Med. 2020;383:274-276. https://www.nejm.org/doi/full/10.1056/NEJMe2021693. Accessed February 24, 2022.
Ananth CV, Brandt JS, Hill J, et al. Historical and recent changes in maternal mortality due to hypertensive disorders in the United States, 1979 to 2018. Hypertension. 2021;78:1414–1422. doi: 10.1161/HYPERTENSIONAHA.121.17661.
EXPERT COMMENTARY
Maternal mortality is a pressing public health issue and is largely preventable. Up to 10% of all US pregnancies are complicated by a hypertensive disorder, and rates of chronic hypertension and severe preeclampsia have steadily increased over the last 4 decades. However, maternal mortality is an outcome in a population with advancing maternal age, increasing obesity, and undermanaged chronic disease. The MMR due to hypertension is substantially higher among Black women compared with White women. Countless studies attribute systemic racism to these disparities.
Details of the study
Spanning 40 years, a recent study by Ananth and colleagues included live births across all 50 United States and Washington, DC. Of the 1.5 million live births examined, there were 3,287 hypertension-related maternal deaths.
Data were deidentified and available in the public domain. The researchers compiled mortality data and live births among women aged 15 to 49. The MMR was considered the death of a woman during pregnancy or within the 42 days following a live birth.
Key points of the study included:
- An estimated two-thirds of maternal deaths are preventable.
- The hypertension-related MMR was 2.1 per 100,000 live births.
- Preeclampsia-related MMR decreased, while hypertension-related MMR increased.
- The MMR from chronic hypertension has increased annually by 9.2%.
- Pregnancies among women with advanced maternal age have grown, especially among those over age 40.
- The MMR due to hypertension increases with age and is highest among women age 45 to 49.
Study strengths and limitations
A major strength of this study is the sheer size of the sample. This is one of the largest studies that examined changes in the MMR in the United States.
As with any study that spans a long period, a primary limitation is inconsistencies in the data collected. In 2003, the US death certificate was revised to include a set of “pregnancy checkboxes” indicating pregnancy at the time of death.
There also have been shifts in diagnostic coding and criteria for preeclampsia.
Classification of race and ethnicity has improved and broadened over time. Despite these limitations, the overarching trends are compelling. ●
This study’s authors note that maternal mortality is largely preventable. Patients need to be aware of their health and how to adopt healthy behaviors long before pregnancy is even a consideration. Primary and secondary prevention are essential for reducing the MMR.
Clinicians who care for women have an opportunity to emphasize cardiac health at every visit. This includes strict blood pressure control through modifiable behaviors like diet and exercise. The busy clinician could consider a 1- to 2-minute pitch to emphasize that heart disease is the leading cause of death in women both during pregnancy and later in life. A tool from the American Heart Association, Life’s Simple 7 (https://www.heart .org/en/healthy-living/healthy-lifestyle/my-life-check--lifes-sim ple-7), can help guide this language.
In office and clinical settings, consider strategies to raise awareness among staff and colleagues about cultural sensitivities to improve the health of all patients. Addressing systemic racism in the US health care system is critical to mitigate racial inequities in the rates of MMR. An editorial in The New England Journal of Medicine urges clinicians to observe patient color rather than be “color blind.”1 The editorialists note that “physician-citizens must recognize the harm inflicted by discrimination and racism and consider this environmental agent of disease as a vital sign— alongside blood pressure, pulse, weight, and temperature—that provides important information about a patient’s condition.”1
LAUREN B. GOLFER, WHNP-BC, AND MARY L. ROSSER, MD, PHD
Ananth CV, Brandt JS, Hill J, et al. Historical and recent changes in maternal mortality due to hypertensive disorders in the United States, 1979 to 2018. Hypertension. 2021;78:1414–1422. doi: 10.1161/HYPERTENSIONAHA.121.17661.
EXPERT COMMENTARY
Maternal mortality is a pressing public health issue and is largely preventable. Up to 10% of all US pregnancies are complicated by a hypertensive disorder, and rates of chronic hypertension and severe preeclampsia have steadily increased over the last 4 decades. However, maternal mortality is an outcome in a population with advancing maternal age, increasing obesity, and undermanaged chronic disease. The MMR due to hypertension is substantially higher among Black women compared with White women. Countless studies attribute systemic racism to these disparities.
Details of the study
Spanning 40 years, a recent study by Ananth and colleagues included live births across all 50 United States and Washington, DC. Of the 1.5 million live births examined, there were 3,287 hypertension-related maternal deaths.
Data were deidentified and available in the public domain. The researchers compiled mortality data and live births among women aged 15 to 49. The MMR was considered the death of a woman during pregnancy or within the 42 days following a live birth.
Key points of the study included:
- An estimated two-thirds of maternal deaths are preventable.
- The hypertension-related MMR was 2.1 per 100,000 live births.
- Preeclampsia-related MMR decreased, while hypertension-related MMR increased.
- The MMR from chronic hypertension has increased annually by 9.2%.
- Pregnancies among women with advanced maternal age have grown, especially among those over age 40.
- The MMR due to hypertension increases with age and is highest among women age 45 to 49.
Study strengths and limitations
A major strength of this study is the sheer size of the sample. This is one of the largest studies that examined changes in the MMR in the United States.
As with any study that spans a long period, a primary limitation is inconsistencies in the data collected. In 2003, the US death certificate was revised to include a set of “pregnancy checkboxes” indicating pregnancy at the time of death.
There also have been shifts in diagnostic coding and criteria for preeclampsia.
Classification of race and ethnicity has improved and broadened over time. Despite these limitations, the overarching trends are compelling. ●
This study’s authors note that maternal mortality is largely preventable. Patients need to be aware of their health and how to adopt healthy behaviors long before pregnancy is even a consideration. Primary and secondary prevention are essential for reducing the MMR.
Clinicians who care for women have an opportunity to emphasize cardiac health at every visit. This includes strict blood pressure control through modifiable behaviors like diet and exercise. The busy clinician could consider a 1- to 2-minute pitch to emphasize that heart disease is the leading cause of death in women both during pregnancy and later in life. A tool from the American Heart Association, Life’s Simple 7 (https://www.heart .org/en/healthy-living/healthy-lifestyle/my-life-check--lifes-sim ple-7), can help guide this language.
In office and clinical settings, consider strategies to raise awareness among staff and colleagues about cultural sensitivities to improve the health of all patients. Addressing systemic racism in the US health care system is critical to mitigate racial inequities in the rates of MMR. An editorial in The New England Journal of Medicine urges clinicians to observe patient color rather than be “color blind.”1 The editorialists note that “physician-citizens must recognize the harm inflicted by discrimination and racism and consider this environmental agent of disease as a vital sign— alongside blood pressure, pulse, weight, and temperature—that provides important information about a patient’s condition.”1
LAUREN B. GOLFER, WHNP-BC, AND MARY L. ROSSER, MD, PHD
- Evans MK, Rosenbaum L, Malina D, et al. Diagnosing and treating systemic racism. N Engl J Med. 2020;383:274-276. https://www.nejm.org/doi/full/10.1056/NEJMe2021693. Accessed February 24, 2022.
- Evans MK, Rosenbaum L, Malina D, et al. Diagnosing and treating systemic racism. N Engl J Med. 2020;383:274-276. https://www.nejm.org/doi/full/10.1056/NEJMe2021693. Accessed February 24, 2022.
Will NAAT replace microscopy for the identification of organisms causing vaginitis?
Over the past 200 years, identification of the specific organism causing an infection has evolved from a reliance on patient history and physical examination to the use of microscopic examination of relevant biological samples to the rise of microbial culture and immunological testing as the gold standards for diagnosis. More recently, advances in nucleic acid testing have made nucleic acid amplification testing (NAAT) a primary method for identifying the specific organism causing an infection.
The evolution of the diagnosis of gonorrhea in clinical practice is a good example of the inexorable evolution of diagnostic techniques from physical examination to microscopic analysis to culture and finally to NAAT. Neiseer discovered Neisseria gonorrhea in 1879.1 In 19th century general medical practice gonorrhea was often diagnosed based on history and physical examination and sometimes microscopy was also utilized.2 In the mid-20th century, it was realized that culture was a superior approach to diagnosis of gonorrhea, and it became the gold standard for diagnosis in general practice.3 NAAT has now replaced culture as the gold standard for the diagnosis of gonorrhea because of its superior performance in clinical practice.4 It may now be time to consider using NAAT rather than microscopy and culture in general practice for the identification of specific microorganisms causing vaginitis.
Trichomoniasis
Vaginitis caused by Trichomonas vaginalis is characterized by a discharge that is foamy and green-yellow in color, with a vaginal pH that is >4.5. Microscopy of a vaginal specimen has low sensitivity, in the range of 50%, for detecting T vaginalis.5-7 There are many factors that make microscopy a poor approach to the diagnosis of T vaginalis, including the rapid decrease in protozoan motility once a vaginal specimen is placed on a glass slide and the similar size of non-motile T vaginalis and other cells in the vagina.
Given the low sensitivity of microscopy for the diagnosis of trichomoniasis, the American College of Obstetricians and Gynecologists (ACOG) recommends NAAT as a primary approach to test for T vaginalis, with culture or NAAT testing as alternative approaches.8 The Centers for Disease Control and Prevention (CDC) recommends that if a wet mount is negative for T vaginalis that NAAT should be utilized.9
In this 2-step testing process, the first step is to test the vaginal pH and perform a microscopic examination of a vaginal specimen for T vaginalis. If T vaginalis organisms are detected, the diagnosis of trichomoniasis is confirmed. If organisms are not detected the second step would be to send a vaginal or urine specimen for NAAT for T vaginalis or for culture. An advantage of NAAT over culture is that urine specimens can be used for diagnosis of T vaginalis while urine specimens are not suitable for culture because of low sensitivity. For patients diagnosed with trichomoniasis, the CDC recommends that testing be repeated in 3 months because of high rates of reinfection. NAAT would be an optimal test to use in this situation.
Continue to: Bacterial vaginosis and candidiasis...
Bacterial vaginosis and candidiasis
ACOG recommends using Amsel criteria or Nugent scoring of a specimen colorized with a Gram stain for the diagnosis of bacterial vaginosis and microscopy or culture for the diagnosis of candidiasis.8 Recent research reports that NAAT testing for bacterial vaginosis and candidiasis may be more sensitive than standard office-based approaches for detecting these two causes of vaginitis. In a study of approximately 1,740 patients with symptoms of vaginitis, vaginal specimens were analyzed using NAAT or standard office approaches to diagnosis.10 In this study the diagnostic gold standards were Nugent scoring with Amsel criteria to resolve intermediate Nugent scores for bacterial vaginosis and culture for Candida. The study demonstrated the superiority of NAAT testing over standard office approaches for the identification of the cause of the vaginitis. NAAT testing was reported to have superior sensitivity for diagnosing bacterial vaginosis compared with the original Amsel criteria (93% vs 76%, respectively (P <.0001), with similar respective specificities of 92% and 94% .10 NAAT testing also had superior sensitivity for diagnosing Candidiasis compared with microscopy after potassium hydroxide treatment of a vaginal specimen (91% vs 58%, respectively (P <.0001).10 NAAT testing also had superior specificity compared with microscopy after potassium hydroxide treatment of a vaginal specimen (94% vs 89%, respectively (P < .0005).10
In another study comparing NAAT with clinical diagnosis for 466 patients with symptoms of vaginitis, standard office approaches to the diagnosis of vaginitis resulted in the failure to identify the correct infection in a large number of cases. For the diagnosis of bacterial vaginosis, clinicians missed 42% of the cases identified by NAAT. For the diagnosis of Candida, clinicians missed 46% of the cases identified by NAAT. For T vaginalis diagnosis, clinicians missed 72% of the cases identified by NAAT. Clearly, this resulted in clinicians not treating many infections detected by NAAT.11
Continue to: One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis...
One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis
In a recent study, 1,471 patients with a symptom of vaginitis (abnormal vaginal discharge, itching or irritation, or odor) self-collected a vaginal swab and had a vaginal swab collected by a clinician.12 The swabs were placed in buffer and the samples were tested by NAAT using the BD Max system (Franklin Lakes, New Jersey) for the presence of nucleic acid sequences of the microorganisms responsible for the most common causes of vaginitis. In this cohort, using the clinician collected vaginal swabs for NAAT, the investigators reported the following pattern of detection of nucleic acid sequences: 36.1%, bacterial vaginosis pattern; 16.2%, Candida spp.; 1.6%, T vaginalis; 0.7%, Candida glabrata; and 0.1%, Candida krusei. Nucleic acid sequences of multiple organisms were detected in 21.7% of patients, including 13.9% with bacterial vaginosis pattern plus Candida spp., 4.9% with bacterial vaginosis pattern plus T vaginalis, 0.3% with Candida spp. plus T vaginalis, 0.2% with Candida spp. plus Candida glabrata, 0.2% with bacterial vaginosis pattern plus Candida glabrata, and 2.2% with all 3 organisms. A total of 23.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.
In another study of 1,491 patients with a symptom of vaginitis, clinician-collected vaginal swabs were tested by NAAT using the Aptima BV and Aptima Candida/Trichomonas systems (Hologic, Marlborough, Massachusetts) for the presence of nucleic acid sequences of microorganisms responsible for most cases of vaginitis.13 The investigators reported the following pattern of detection of nucleic acid sequences: 28.6%, bacterial vaginosis pattern; 14.2%, Candida spp.; 3%, T vaginalis; 1.9%, Candida glabrata.13 Nucleic acid sequences from multiple organisms were detected in 23.3% of patients. Nucleic acid sequences suggesting the presence of two different causes of vaginitis were detected among 20.8% of patients, including bacterial vaginosis plus Candida spp., 11.1%; bacterial vaginosis plus T vaginalis, 7.2%; Candida spp. plus T vaginalis, 1.0%; Candida spp. plus Candida glabrata, 0.9%; bacterial vaginosis plus Candida spp., 0.5%; Candida glabrata plus T vaginalis, 0.1%. Nucleic acid sequences suggesting the presence of 3 different causes of vaginitis were detected in 2.4% of patients, the most common being the combination of bacterial vaginosis plus Candida spp. plus T vaginalis, 1.7% and bacterial vaginosis plus Candida spp. plus Candida glabrata, 0.5%. Nucleic acid sequences suggesting the presence of 4 different causes of vaginitis were detected in 0.1% of patients. A total of 28.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.13
In clinical practice it is uncommon to see the diagnosis of multiple causes of vaginitis recorded in the medical record of a patient. This suggests that we are not effectively identifying the 20% of patients with multiple causes of vaginitis.
When multiple organisms that cause vaginitis are present, NAAT is superior to clinical evaluation for diagnosis
In a study of 1,264 patients with symptoms of vaginitis who had an identified microbial cause, more than 20% had multiple organisms detected by NAAT
Patient collection of a vaginal swab for NAAT
Multiple studies have reported that collection of a vaginal swab for NAAT by the patient or a clinician results in similar excellent test performance.4,12,13 This observation might catalyze the development of clinical protocols where patients with vaginitis could collect the swab for NAAT analysis, without needing to have a speculum examination by a clinician.
When collecting a vaginal specimen for NAAT it is important that no vaginal lubricants or creams contaminate the collection swab. Vaginal lubricants and creams may inhibit the polymerase chain reaction enzymes resulting in a false negative. The swab may be directly inserted into the vagina to collect the specimen or a speculum without a lubricant, except water can be used to facilitate specimen collection. To collect a specimen without a speculum the swab is inserted 2 inches into the vagina and rotated for 10 to 15 seconds.
What should clinicians do while waiting for a NAAT result?
A major problem with NAAT testing for vaginitis is that the results are not available at the initial patient visit, impacting the ability to make an immediate diagnosis and provide targeted antibiotic treatment. Given that bacterial vaginosis and Candida species are the most common causes of infectious vaginitis in many populations of gynecology patients, one approach is to initiate treatment with one dose of an oral antifungal agent and a multiday course of vaginal metronidazole. Once the NAAT test results are available, the treatment can be refined to specific infectious agents identified by the test, or the antibiotics can be discontinued if no relevant microorganisms are detected. Another approach would be to wait until the NAAT test is completed and then prescribe the appropriate antibiotic. My sense is that most patients would not favor this wait and see approach.
Barriers to the use of NAAT for vaginitis
A barrier to the use of NAAT for the diagnosis of vaginitis is that leading organizations do not currently recommend NAAT as a primary approach to diagnosis, favoring microscopy and measurement of vaginal pH.9 In addition, clinicians and patients may be rightfully concerned about the cost of NAAT, which can be substantial.
Vaginitis, especially when it is recurrent, can be stressful14 and have an impact on a patient’s quality of life15,16 and sexual health.17 Arguably, our current practice algorithms for diagnosing the cause of vaginitis are not optimized.18 Our failure to accurately diagnose the cause of vaginitis contributes to inappropriate antibiotic treatment and return visits because of inadequate initial treatment.18 We can improve and simplify our approach to the diagnosis of vaginitis by prioritizing the use of NAAT.19 In turn, reliably making the right diagnosis will result in the optimization of treatment. ●
- Jose PP, Vivekanandan V, Sobhanakumari K. Gonorrhea: Historical outlook. J Skin Sex Transm Dis. 2020;2:110-114.
- Bayly HW. The diagnosis and treatment of chronic gonorrhoea and its local complications. Br Med J. 1914;14:584-587.
- Stuart RD. The diagnosis and control of gonorrhoea by bacteriological cultures: with a preliminary report on a new method for transporting clinical material. Glasgow Med J. 1946;27:131-142.
- Wilson JD, Wallace HE, Loftus-Keeling M, et al. Swab-yourself trial with economic monitoring and testing for infections collectively (SYSTEMATIC): Part 2. A diagnostic accuracy and cost-effectiveness study comparing rectal, pharyngeal and urogenital samples analyzed individually, versus as a pooled specimen, for the diagnosis of gonorrhea and chlamydia. Clin Infect Dis. 2021;73:e3183-3193.
- Hollman D, Coupey SM, Fox AS, et al. Screening for Trichomonas vaginalis in high-risk adolescent females with a new NAAT: association with ethnicity, symptoms and prior and current STIs. J Pediatr Adolesc Gynecol. 2010;23:312-316.
- Roth AM, Williams JA, Ly R. et al. Changing sexually transmitted infection screening protocol will result in improved case finding for Trichomonas vaginalis among high-risk female populations. Sex Transm Dis. 2011;38:398-400.
- Hobbs MM, Sena AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infection. 2013;89:434-438.
- Vaginitis in nonpregnant patients. ACOG Practice Bulletin No 215. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2020;135:e1-e17.
- Workowksi KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines 2021. MMWR. 2021;70:1-187.
- Schwebke JR, Gaydos CA, Hyirjesy P, et al. Diagnostic performance of a molecular test versus clinician assessment of vaginitis. J Clin Microbiol. 2018;56:e00252-18.
- Broache M, Cammarata CL, Stonebraker E, et al. Performance of vaginal panel assay compared with clinical diagnosis of vaginitis. Obstet Gynecol. 2021;138:853-859.
- Gaydos CA, Beqaj S, Schwebke JR, et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet Gynecol. 2017;130:181-189.
- Schwebke JR, Taylor SN, Ackerman N, et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginalis assays: results from a prospective multi-center study. J Clin Microbiol. 2020;58:e01643-19.
- Ehrstrom S, Kornfeld D, Rylander E. Perceived stress in women with recurrent vulvovaginal candidiasis. J Psychosomatic Obstet Gynecol. 2007;28:169-176.
- Abellea S, Guelfucci F, Wagner J, et al. Subjective health status and health-related quality of life among women with recurrent vulvovaginal candidosis in Europe and the USA. Health Quality Life Outcomes. 2013;11:169.
- Fukazawa EI, Witkin SS, Robial R, et al. Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet. 2019;300:647-650.
- Giraldo PC, Polpeta NC, Juliato CT, et al. Evaluation of sexual function in Brazilian women with recurrent vulvovaginal candidiasis and localized provoked vulvodynia. J Sex Med. 2012;9:805-811.
- Hillier SL, Austin M, Macio I, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021;72:1538-1543.
- . Sobel JD. Syndromic treatment of women with vulvovaginal symptoms in the United States: a call to action. Clin Infect Dis. 2021;72:1544-1545.
Over the past 200 years, identification of the specific organism causing an infection has evolved from a reliance on patient history and physical examination to the use of microscopic examination of relevant biological samples to the rise of microbial culture and immunological testing as the gold standards for diagnosis. More recently, advances in nucleic acid testing have made nucleic acid amplification testing (NAAT) a primary method for identifying the specific organism causing an infection.
The evolution of the diagnosis of gonorrhea in clinical practice is a good example of the inexorable evolution of diagnostic techniques from physical examination to microscopic analysis to culture and finally to NAAT. Neiseer discovered Neisseria gonorrhea in 1879.1 In 19th century general medical practice gonorrhea was often diagnosed based on history and physical examination and sometimes microscopy was also utilized.2 In the mid-20th century, it was realized that culture was a superior approach to diagnosis of gonorrhea, and it became the gold standard for diagnosis in general practice.3 NAAT has now replaced culture as the gold standard for the diagnosis of gonorrhea because of its superior performance in clinical practice.4 It may now be time to consider using NAAT rather than microscopy and culture in general practice for the identification of specific microorganisms causing vaginitis.
Trichomoniasis
Vaginitis caused by Trichomonas vaginalis is characterized by a discharge that is foamy and green-yellow in color, with a vaginal pH that is >4.5. Microscopy of a vaginal specimen has low sensitivity, in the range of 50%, for detecting T vaginalis.5-7 There are many factors that make microscopy a poor approach to the diagnosis of T vaginalis, including the rapid decrease in protozoan motility once a vaginal specimen is placed on a glass slide and the similar size of non-motile T vaginalis and other cells in the vagina.
Given the low sensitivity of microscopy for the diagnosis of trichomoniasis, the American College of Obstetricians and Gynecologists (ACOG) recommends NAAT as a primary approach to test for T vaginalis, with culture or NAAT testing as alternative approaches.8 The Centers for Disease Control and Prevention (CDC) recommends that if a wet mount is negative for T vaginalis that NAAT should be utilized.9
In this 2-step testing process, the first step is to test the vaginal pH and perform a microscopic examination of a vaginal specimen for T vaginalis. If T vaginalis organisms are detected, the diagnosis of trichomoniasis is confirmed. If organisms are not detected the second step would be to send a vaginal or urine specimen for NAAT for T vaginalis or for culture. An advantage of NAAT over culture is that urine specimens can be used for diagnosis of T vaginalis while urine specimens are not suitable for culture because of low sensitivity. For patients diagnosed with trichomoniasis, the CDC recommends that testing be repeated in 3 months because of high rates of reinfection. NAAT would be an optimal test to use in this situation.
Continue to: Bacterial vaginosis and candidiasis...
Bacterial vaginosis and candidiasis
ACOG recommends using Amsel criteria or Nugent scoring of a specimen colorized with a Gram stain for the diagnosis of bacterial vaginosis and microscopy or culture for the diagnosis of candidiasis.8 Recent research reports that NAAT testing for bacterial vaginosis and candidiasis may be more sensitive than standard office-based approaches for detecting these two causes of vaginitis. In a study of approximately 1,740 patients with symptoms of vaginitis, vaginal specimens were analyzed using NAAT or standard office approaches to diagnosis.10 In this study the diagnostic gold standards were Nugent scoring with Amsel criteria to resolve intermediate Nugent scores for bacterial vaginosis and culture for Candida. The study demonstrated the superiority of NAAT testing over standard office approaches for the identification of the cause of the vaginitis. NAAT testing was reported to have superior sensitivity for diagnosing bacterial vaginosis compared with the original Amsel criteria (93% vs 76%, respectively (P <.0001), with similar respective specificities of 92% and 94% .10 NAAT testing also had superior sensitivity for diagnosing Candidiasis compared with microscopy after potassium hydroxide treatment of a vaginal specimen (91% vs 58%, respectively (P <.0001).10 NAAT testing also had superior specificity compared with microscopy after potassium hydroxide treatment of a vaginal specimen (94% vs 89%, respectively (P < .0005).10
In another study comparing NAAT with clinical diagnosis for 466 patients with symptoms of vaginitis, standard office approaches to the diagnosis of vaginitis resulted in the failure to identify the correct infection in a large number of cases. For the diagnosis of bacterial vaginosis, clinicians missed 42% of the cases identified by NAAT. For the diagnosis of Candida, clinicians missed 46% of the cases identified by NAAT. For T vaginalis diagnosis, clinicians missed 72% of the cases identified by NAAT. Clearly, this resulted in clinicians not treating many infections detected by NAAT.11
Continue to: One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis...
One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis
In a recent study, 1,471 patients with a symptom of vaginitis (abnormal vaginal discharge, itching or irritation, or odor) self-collected a vaginal swab and had a vaginal swab collected by a clinician.12 The swabs were placed in buffer and the samples were tested by NAAT using the BD Max system (Franklin Lakes, New Jersey) for the presence of nucleic acid sequences of the microorganisms responsible for the most common causes of vaginitis. In this cohort, using the clinician collected vaginal swabs for NAAT, the investigators reported the following pattern of detection of nucleic acid sequences: 36.1%, bacterial vaginosis pattern; 16.2%, Candida spp.; 1.6%, T vaginalis; 0.7%, Candida glabrata; and 0.1%, Candida krusei. Nucleic acid sequences of multiple organisms were detected in 21.7% of patients, including 13.9% with bacterial vaginosis pattern plus Candida spp., 4.9% with bacterial vaginosis pattern plus T vaginalis, 0.3% with Candida spp. plus T vaginalis, 0.2% with Candida spp. plus Candida glabrata, 0.2% with bacterial vaginosis pattern plus Candida glabrata, and 2.2% with all 3 organisms. A total of 23.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.
In another study of 1,491 patients with a symptom of vaginitis, clinician-collected vaginal swabs were tested by NAAT using the Aptima BV and Aptima Candida/Trichomonas systems (Hologic, Marlborough, Massachusetts) for the presence of nucleic acid sequences of microorganisms responsible for most cases of vaginitis.13 The investigators reported the following pattern of detection of nucleic acid sequences: 28.6%, bacterial vaginosis pattern; 14.2%, Candida spp.; 3%, T vaginalis; 1.9%, Candida glabrata.13 Nucleic acid sequences from multiple organisms were detected in 23.3% of patients. Nucleic acid sequences suggesting the presence of two different causes of vaginitis were detected among 20.8% of patients, including bacterial vaginosis plus Candida spp., 11.1%; bacterial vaginosis plus T vaginalis, 7.2%; Candida spp. plus T vaginalis, 1.0%; Candida spp. plus Candida glabrata, 0.9%; bacterial vaginosis plus Candida spp., 0.5%; Candida glabrata plus T vaginalis, 0.1%. Nucleic acid sequences suggesting the presence of 3 different causes of vaginitis were detected in 2.4% of patients, the most common being the combination of bacterial vaginosis plus Candida spp. plus T vaginalis, 1.7% and bacterial vaginosis plus Candida spp. plus Candida glabrata, 0.5%. Nucleic acid sequences suggesting the presence of 4 different causes of vaginitis were detected in 0.1% of patients. A total of 28.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.13
In clinical practice it is uncommon to see the diagnosis of multiple causes of vaginitis recorded in the medical record of a patient. This suggests that we are not effectively identifying the 20% of patients with multiple causes of vaginitis.
When multiple organisms that cause vaginitis are present, NAAT is superior to clinical evaluation for diagnosis
In a study of 1,264 patients with symptoms of vaginitis who had an identified microbial cause, more than 20% had multiple organisms detected by NAAT
Patient collection of a vaginal swab for NAAT
Multiple studies have reported that collection of a vaginal swab for NAAT by the patient or a clinician results in similar excellent test performance.4,12,13 This observation might catalyze the development of clinical protocols where patients with vaginitis could collect the swab for NAAT analysis, without needing to have a speculum examination by a clinician.
When collecting a vaginal specimen for NAAT it is important that no vaginal lubricants or creams contaminate the collection swab. Vaginal lubricants and creams may inhibit the polymerase chain reaction enzymes resulting in a false negative. The swab may be directly inserted into the vagina to collect the specimen or a speculum without a lubricant, except water can be used to facilitate specimen collection. To collect a specimen without a speculum the swab is inserted 2 inches into the vagina and rotated for 10 to 15 seconds.
What should clinicians do while waiting for a NAAT result?
A major problem with NAAT testing for vaginitis is that the results are not available at the initial patient visit, impacting the ability to make an immediate diagnosis and provide targeted antibiotic treatment. Given that bacterial vaginosis and Candida species are the most common causes of infectious vaginitis in many populations of gynecology patients, one approach is to initiate treatment with one dose of an oral antifungal agent and a multiday course of vaginal metronidazole. Once the NAAT test results are available, the treatment can be refined to specific infectious agents identified by the test, or the antibiotics can be discontinued if no relevant microorganisms are detected. Another approach would be to wait until the NAAT test is completed and then prescribe the appropriate antibiotic. My sense is that most patients would not favor this wait and see approach.
Barriers to the use of NAAT for vaginitis
A barrier to the use of NAAT for the diagnosis of vaginitis is that leading organizations do not currently recommend NAAT as a primary approach to diagnosis, favoring microscopy and measurement of vaginal pH.9 In addition, clinicians and patients may be rightfully concerned about the cost of NAAT, which can be substantial.
Vaginitis, especially when it is recurrent, can be stressful14 and have an impact on a patient’s quality of life15,16 and sexual health.17 Arguably, our current practice algorithms for diagnosing the cause of vaginitis are not optimized.18 Our failure to accurately diagnose the cause of vaginitis contributes to inappropriate antibiotic treatment and return visits because of inadequate initial treatment.18 We can improve and simplify our approach to the diagnosis of vaginitis by prioritizing the use of NAAT.19 In turn, reliably making the right diagnosis will result in the optimization of treatment. ●
Over the past 200 years, identification of the specific organism causing an infection has evolved from a reliance on patient history and physical examination to the use of microscopic examination of relevant biological samples to the rise of microbial culture and immunological testing as the gold standards for diagnosis. More recently, advances in nucleic acid testing have made nucleic acid amplification testing (NAAT) a primary method for identifying the specific organism causing an infection.
The evolution of the diagnosis of gonorrhea in clinical practice is a good example of the inexorable evolution of diagnostic techniques from physical examination to microscopic analysis to culture and finally to NAAT. Neiseer discovered Neisseria gonorrhea in 1879.1 In 19th century general medical practice gonorrhea was often diagnosed based on history and physical examination and sometimes microscopy was also utilized.2 In the mid-20th century, it was realized that culture was a superior approach to diagnosis of gonorrhea, and it became the gold standard for diagnosis in general practice.3 NAAT has now replaced culture as the gold standard for the diagnosis of gonorrhea because of its superior performance in clinical practice.4 It may now be time to consider using NAAT rather than microscopy and culture in general practice for the identification of specific microorganisms causing vaginitis.
Trichomoniasis
Vaginitis caused by Trichomonas vaginalis is characterized by a discharge that is foamy and green-yellow in color, with a vaginal pH that is >4.5. Microscopy of a vaginal specimen has low sensitivity, in the range of 50%, for detecting T vaginalis.5-7 There are many factors that make microscopy a poor approach to the diagnosis of T vaginalis, including the rapid decrease in protozoan motility once a vaginal specimen is placed on a glass slide and the similar size of non-motile T vaginalis and other cells in the vagina.
Given the low sensitivity of microscopy for the diagnosis of trichomoniasis, the American College of Obstetricians and Gynecologists (ACOG) recommends NAAT as a primary approach to test for T vaginalis, with culture or NAAT testing as alternative approaches.8 The Centers for Disease Control and Prevention (CDC) recommends that if a wet mount is negative for T vaginalis that NAAT should be utilized.9
In this 2-step testing process, the first step is to test the vaginal pH and perform a microscopic examination of a vaginal specimen for T vaginalis. If T vaginalis organisms are detected, the diagnosis of trichomoniasis is confirmed. If organisms are not detected the second step would be to send a vaginal or urine specimen for NAAT for T vaginalis or for culture. An advantage of NAAT over culture is that urine specimens can be used for diagnosis of T vaginalis while urine specimens are not suitable for culture because of low sensitivity. For patients diagnosed with trichomoniasis, the CDC recommends that testing be repeated in 3 months because of high rates of reinfection. NAAT would be an optimal test to use in this situation.
Continue to: Bacterial vaginosis and candidiasis...
Bacterial vaginosis and candidiasis
ACOG recommends using Amsel criteria or Nugent scoring of a specimen colorized with a Gram stain for the diagnosis of bacterial vaginosis and microscopy or culture for the diagnosis of candidiasis.8 Recent research reports that NAAT testing for bacterial vaginosis and candidiasis may be more sensitive than standard office-based approaches for detecting these two causes of vaginitis. In a study of approximately 1,740 patients with symptoms of vaginitis, vaginal specimens were analyzed using NAAT or standard office approaches to diagnosis.10 In this study the diagnostic gold standards were Nugent scoring with Amsel criteria to resolve intermediate Nugent scores for bacterial vaginosis and culture for Candida. The study demonstrated the superiority of NAAT testing over standard office approaches for the identification of the cause of the vaginitis. NAAT testing was reported to have superior sensitivity for diagnosing bacterial vaginosis compared with the original Amsel criteria (93% vs 76%, respectively (P <.0001), with similar respective specificities of 92% and 94% .10 NAAT testing also had superior sensitivity for diagnosing Candidiasis compared with microscopy after potassium hydroxide treatment of a vaginal specimen (91% vs 58%, respectively (P <.0001).10 NAAT testing also had superior specificity compared with microscopy after potassium hydroxide treatment of a vaginal specimen (94% vs 89%, respectively (P < .0005).10
In another study comparing NAAT with clinical diagnosis for 466 patients with symptoms of vaginitis, standard office approaches to the diagnosis of vaginitis resulted in the failure to identify the correct infection in a large number of cases. For the diagnosis of bacterial vaginosis, clinicians missed 42% of the cases identified by NAAT. For the diagnosis of Candida, clinicians missed 46% of the cases identified by NAAT. For T vaginalis diagnosis, clinicians missed 72% of the cases identified by NAAT. Clearly, this resulted in clinicians not treating many infections detected by NAAT.11
Continue to: One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis...
One in 5 patients with symptoms of vaginitis have 2 causes of vaginitis
In a recent study, 1,471 patients with a symptom of vaginitis (abnormal vaginal discharge, itching or irritation, or odor) self-collected a vaginal swab and had a vaginal swab collected by a clinician.12 The swabs were placed in buffer and the samples were tested by NAAT using the BD Max system (Franklin Lakes, New Jersey) for the presence of nucleic acid sequences of the microorganisms responsible for the most common causes of vaginitis. In this cohort, using the clinician collected vaginal swabs for NAAT, the investigators reported the following pattern of detection of nucleic acid sequences: 36.1%, bacterial vaginosis pattern; 16.2%, Candida spp.; 1.6%, T vaginalis; 0.7%, Candida glabrata; and 0.1%, Candida krusei. Nucleic acid sequences of multiple organisms were detected in 21.7% of patients, including 13.9% with bacterial vaginosis pattern plus Candida spp., 4.9% with bacterial vaginosis pattern plus T vaginalis, 0.3% with Candida spp. plus T vaginalis, 0.2% with Candida spp. plus Candida glabrata, 0.2% with bacterial vaginosis pattern plus Candida glabrata, and 2.2% with all 3 organisms. A total of 23.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.
In another study of 1,491 patients with a symptom of vaginitis, clinician-collected vaginal swabs were tested by NAAT using the Aptima BV and Aptima Candida/Trichomonas systems (Hologic, Marlborough, Massachusetts) for the presence of nucleic acid sequences of microorganisms responsible for most cases of vaginitis.13 The investigators reported the following pattern of detection of nucleic acid sequences: 28.6%, bacterial vaginosis pattern; 14.2%, Candida spp.; 3%, T vaginalis; 1.9%, Candida glabrata.13 Nucleic acid sequences from multiple organisms were detected in 23.3% of patients. Nucleic acid sequences suggesting the presence of two different causes of vaginitis were detected among 20.8% of patients, including bacterial vaginosis plus Candida spp., 11.1%; bacterial vaginosis plus T vaginalis, 7.2%; Candida spp. plus T vaginalis, 1.0%; Candida spp. plus Candida glabrata, 0.9%; bacterial vaginosis plus Candida spp., 0.5%; Candida glabrata plus T vaginalis, 0.1%. Nucleic acid sequences suggesting the presence of 3 different causes of vaginitis were detected in 2.4% of patients, the most common being the combination of bacterial vaginosis plus Candida spp. plus T vaginalis, 1.7% and bacterial vaginosis plus Candida spp. plus Candida glabrata, 0.5%. Nucleic acid sequences suggesting the presence of 4 different causes of vaginitis were detected in 0.1% of patients. A total of 28.8% of the women had no detectable nucleic acid sequences associated with organisms known to cause vaginitis.13
In clinical practice it is uncommon to see the diagnosis of multiple causes of vaginitis recorded in the medical record of a patient. This suggests that we are not effectively identifying the 20% of patients with multiple causes of vaginitis.
When multiple organisms that cause vaginitis are present, NAAT is superior to clinical evaluation for diagnosis
In a study of 1,264 patients with symptoms of vaginitis who had an identified microbial cause, more than 20% had multiple organisms detected by NAAT
Patient collection of a vaginal swab for NAAT
Multiple studies have reported that collection of a vaginal swab for NAAT by the patient or a clinician results in similar excellent test performance.4,12,13 This observation might catalyze the development of clinical protocols where patients with vaginitis could collect the swab for NAAT analysis, without needing to have a speculum examination by a clinician.
When collecting a vaginal specimen for NAAT it is important that no vaginal lubricants or creams contaminate the collection swab. Vaginal lubricants and creams may inhibit the polymerase chain reaction enzymes resulting in a false negative. The swab may be directly inserted into the vagina to collect the specimen or a speculum without a lubricant, except water can be used to facilitate specimen collection. To collect a specimen without a speculum the swab is inserted 2 inches into the vagina and rotated for 10 to 15 seconds.
What should clinicians do while waiting for a NAAT result?
A major problem with NAAT testing for vaginitis is that the results are not available at the initial patient visit, impacting the ability to make an immediate diagnosis and provide targeted antibiotic treatment. Given that bacterial vaginosis and Candida species are the most common causes of infectious vaginitis in many populations of gynecology patients, one approach is to initiate treatment with one dose of an oral antifungal agent and a multiday course of vaginal metronidazole. Once the NAAT test results are available, the treatment can be refined to specific infectious agents identified by the test, or the antibiotics can be discontinued if no relevant microorganisms are detected. Another approach would be to wait until the NAAT test is completed and then prescribe the appropriate antibiotic. My sense is that most patients would not favor this wait and see approach.
Barriers to the use of NAAT for vaginitis
A barrier to the use of NAAT for the diagnosis of vaginitis is that leading organizations do not currently recommend NAAT as a primary approach to diagnosis, favoring microscopy and measurement of vaginal pH.9 In addition, clinicians and patients may be rightfully concerned about the cost of NAAT, which can be substantial.
Vaginitis, especially when it is recurrent, can be stressful14 and have an impact on a patient’s quality of life15,16 and sexual health.17 Arguably, our current practice algorithms for diagnosing the cause of vaginitis are not optimized.18 Our failure to accurately diagnose the cause of vaginitis contributes to inappropriate antibiotic treatment and return visits because of inadequate initial treatment.18 We can improve and simplify our approach to the diagnosis of vaginitis by prioritizing the use of NAAT.19 In turn, reliably making the right diagnosis will result in the optimization of treatment. ●
- Jose PP, Vivekanandan V, Sobhanakumari K. Gonorrhea: Historical outlook. J Skin Sex Transm Dis. 2020;2:110-114.
- Bayly HW. The diagnosis and treatment of chronic gonorrhoea and its local complications. Br Med J. 1914;14:584-587.
- Stuart RD. The diagnosis and control of gonorrhoea by bacteriological cultures: with a preliminary report on a new method for transporting clinical material. Glasgow Med J. 1946;27:131-142.
- Wilson JD, Wallace HE, Loftus-Keeling M, et al. Swab-yourself trial with economic monitoring and testing for infections collectively (SYSTEMATIC): Part 2. A diagnostic accuracy and cost-effectiveness study comparing rectal, pharyngeal and urogenital samples analyzed individually, versus as a pooled specimen, for the diagnosis of gonorrhea and chlamydia. Clin Infect Dis. 2021;73:e3183-3193.
- Hollman D, Coupey SM, Fox AS, et al. Screening for Trichomonas vaginalis in high-risk adolescent females with a new NAAT: association with ethnicity, symptoms and prior and current STIs. J Pediatr Adolesc Gynecol. 2010;23:312-316.
- Roth AM, Williams JA, Ly R. et al. Changing sexually transmitted infection screening protocol will result in improved case finding for Trichomonas vaginalis among high-risk female populations. Sex Transm Dis. 2011;38:398-400.
- Hobbs MM, Sena AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infection. 2013;89:434-438.
- Vaginitis in nonpregnant patients. ACOG Practice Bulletin No 215. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2020;135:e1-e17.
- Workowksi KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines 2021. MMWR. 2021;70:1-187.
- Schwebke JR, Gaydos CA, Hyirjesy P, et al. Diagnostic performance of a molecular test versus clinician assessment of vaginitis. J Clin Microbiol. 2018;56:e00252-18.
- Broache M, Cammarata CL, Stonebraker E, et al. Performance of vaginal panel assay compared with clinical diagnosis of vaginitis. Obstet Gynecol. 2021;138:853-859.
- Gaydos CA, Beqaj S, Schwebke JR, et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet Gynecol. 2017;130:181-189.
- Schwebke JR, Taylor SN, Ackerman N, et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginalis assays: results from a prospective multi-center study. J Clin Microbiol. 2020;58:e01643-19.
- Ehrstrom S, Kornfeld D, Rylander E. Perceived stress in women with recurrent vulvovaginal candidiasis. J Psychosomatic Obstet Gynecol. 2007;28:169-176.
- Abellea S, Guelfucci F, Wagner J, et al. Subjective health status and health-related quality of life among women with recurrent vulvovaginal candidosis in Europe and the USA. Health Quality Life Outcomes. 2013;11:169.
- Fukazawa EI, Witkin SS, Robial R, et al. Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet. 2019;300:647-650.
- Giraldo PC, Polpeta NC, Juliato CT, et al. Evaluation of sexual function in Brazilian women with recurrent vulvovaginal candidiasis and localized provoked vulvodynia. J Sex Med. 2012;9:805-811.
- Hillier SL, Austin M, Macio I, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021;72:1538-1543.
- . Sobel JD. Syndromic treatment of women with vulvovaginal symptoms in the United States: a call to action. Clin Infect Dis. 2021;72:1544-1545.
- Jose PP, Vivekanandan V, Sobhanakumari K. Gonorrhea: Historical outlook. J Skin Sex Transm Dis. 2020;2:110-114.
- Bayly HW. The diagnosis and treatment of chronic gonorrhoea and its local complications. Br Med J. 1914;14:584-587.
- Stuart RD. The diagnosis and control of gonorrhoea by bacteriological cultures: with a preliminary report on a new method for transporting clinical material. Glasgow Med J. 1946;27:131-142.
- Wilson JD, Wallace HE, Loftus-Keeling M, et al. Swab-yourself trial with economic monitoring and testing for infections collectively (SYSTEMATIC): Part 2. A diagnostic accuracy and cost-effectiveness study comparing rectal, pharyngeal and urogenital samples analyzed individually, versus as a pooled specimen, for the diagnosis of gonorrhea and chlamydia. Clin Infect Dis. 2021;73:e3183-3193.
- Hollman D, Coupey SM, Fox AS, et al. Screening for Trichomonas vaginalis in high-risk adolescent females with a new NAAT: association with ethnicity, symptoms and prior and current STIs. J Pediatr Adolesc Gynecol. 2010;23:312-316.
- Roth AM, Williams JA, Ly R. et al. Changing sexually transmitted infection screening protocol will result in improved case finding for Trichomonas vaginalis among high-risk female populations. Sex Transm Dis. 2011;38:398-400.
- Hobbs MM, Sena AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infection. 2013;89:434-438.
- Vaginitis in nonpregnant patients. ACOG Practice Bulletin No 215. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2020;135:e1-e17.
- Workowksi KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines 2021. MMWR. 2021;70:1-187.
- Schwebke JR, Gaydos CA, Hyirjesy P, et al. Diagnostic performance of a molecular test versus clinician assessment of vaginitis. J Clin Microbiol. 2018;56:e00252-18.
- Broache M, Cammarata CL, Stonebraker E, et al. Performance of vaginal panel assay compared with clinical diagnosis of vaginitis. Obstet Gynecol. 2021;138:853-859.
- Gaydos CA, Beqaj S, Schwebke JR, et al. Clinical validation of a test for the diagnosis of vaginitis. Obstet Gynecol. 2017;130:181-189.
- Schwebke JR, Taylor SN, Ackerman N, et al. Clinical validation of the Aptima bacterial vaginosis and Aptima Candida/Trichomonas vaginalis assays: results from a prospective multi-center study. J Clin Microbiol. 2020;58:e01643-19.
- Ehrstrom S, Kornfeld D, Rylander E. Perceived stress in women with recurrent vulvovaginal candidiasis. J Psychosomatic Obstet Gynecol. 2007;28:169-176.
- Abellea S, Guelfucci F, Wagner J, et al. Subjective health status and health-related quality of life among women with recurrent vulvovaginal candidosis in Europe and the USA. Health Quality Life Outcomes. 2013;11:169.
- Fukazawa EI, Witkin SS, Robial R, et al. Influence of recurrent vulvovaginal candidiasis on quality of life issues. Arch Gynecol Obstet. 2019;300:647-650.
- Giraldo PC, Polpeta NC, Juliato CT, et al. Evaluation of sexual function in Brazilian women with recurrent vulvovaginal candidiasis and localized provoked vulvodynia. J Sex Med. 2012;9:805-811.
- Hillier SL, Austin M, Macio I, et al. Diagnosis and treatment of vaginal discharge syndromes in community practice settings. Clin Infect Dis. 2021;72:1538-1543.
- . Sobel JD. Syndromic treatment of women with vulvovaginal symptoms in the United States: a call to action. Clin Infect Dis. 2021;72:1544-1545.
Appropriate cancer screening for women with dense breasts
We have been interested in the quiz series focused on breast cancer screening for women with dense breasts presented in
The concerns with breast cancer in particular
Breast cancer is not cervical cancer. It isn’t one disease. It is a multitude of diseases that happen to show up in the breast. Some are relatively slow-growing—the kinds of cancers that lend themselves to screening and to early intervention. But other cancers are rapidly-growing; they show up no matter how often or what modality we use for screening. Our goal should be to find an approach to screening that can diagnose breast cancer at a stage where we can intervene and positively impact breast cancer specific and overall mortality.
Screening guidelines vary
The variety of screening guidelines published by different professional organizations reflect differing assumptions and sets of values related to the early diagnosis and treatment of breast cancer. (For a comprehensive table of current screening guidelines, see https://www.cdc.gov/cancer/breast/pdf/breast-cancer-screening-guidelines-508.pdf.)
ACOG’s approach—to offer screening at age 40 but to begin by at least age 50 and, through shared decision making with the patient, screen every 1 or 2 years—is focused on capturing as many cases as we can identify, while minimizing the harms of false-positives.1 The perspective of the US Preventive Services Task Force (USPSTF) recommendations (to screen every 2 years beginning at age 50) is at the population level, a cost-effective approach that will have the greatest benefit while minimizing harms in the population at large.2 The American Society of Breast Surgeons recommends screening to begin by age 40.3 Like the breast surgeons, radiologists dedicated to breast imaging are focused on an individual rather than a population level. They strive to identify each and every instance of possible cancer, and therefore recommend annual screening beginning at age 40.4 However, with more aggressive screening in average-risk women many cases of ductal carcinoma in situ (DCIS) are identified—a lesion that, if not detected, may not impact the woman’s health during her lifetime—representing what some might call “overdiagnosis.” Yet there may be some instances in which the DCIS might affect an individual woman’s health. Unfortunately, we can’t prospectively distinguish between the first and the second types of cases.
We follow American College of Obstetricians and Gynecologists and US Preventive Services Task Force guidelines in discussing screening (both its hazards and benefits) with our average-risk patients beginning at age 40. We talk about risk factors for breast cancer, including breast density, but let patients know that no specific additional imaging is advised, and that density is more common in younger women (one consideration in earlier screening) and is quite common in general. Although we do not send follow-up letters to patients with dense breasts, we do educate our staff so that they can respond appropriately should patients call with questions.
Of course, we all bring to the table values that will impact the decisions that we make for ourselves and for our patients. What an ObGyn might suggest may differ from what a radiologist might suggest. Although we follow recommendations made by the radiologist at screening, an ObGyn wants to take care of the whole human being. We are concerned with bones, heart, everything about the patient, so we approach a patient in a different way. These priorities are reflected in the current varying breast cancer screening guidelines.
Continue to: Research on breast cancer screening varies by design...
Research on breast cancer screening varies by design
There has not been a randomized clinical trial conducted on screening mammography since the days of the analog mammogram. The research that has been conducted is difficult to compare due to variations in screening ages and intervals, technology sensitivity, and patient adherence with recommended screening. Treatments for breast cancer also have changed dramatically over time, so the findings of older studies may no longer be relevant to current breast cancer screening. The kind of analysis that needs to be done is an interrupted time series, where you can look at the trajectory of breast cancer survival and whether screening mammography shifts that survival in any way.
One specific study from Australia measured the impact of newer available breast cancer treatments, including tamoxifen for women with receptor-positive tumors and newer chemotherapy strategies.5 The authors analyzed screening mammography trends in one large province where women aged 50 to 69 were offered biennial screening. Trends from the 1990s showed that more women were being screened over time. Simultaneously, however, advances in therapy were entering clinical practice. The researchers pointed to a substantial decline in mortality from breast cancer from the early 1980s until 2013. But their conclusion was that none of the decline in mortality for breast cancer could be attributed to screening mammography when they looked at time trends; from their perspective all of the important decline in breast cancer mortality resulted from better treatment. They concluded that government programs should not support screening mammography.5
That is a recommendation that we do not support. However, we do recognize the conundrum that mammography is less sensitive among those who have dense breasts. In order to have congruent professional guidelines, we support research funding to determine which types, starting ages, and intervals of screening would be best in various patient populations. The USPSTF cites data from studies performed in the 1980s based on outdated technology; more recent (and relevant) randomized clinical trials have not been performed, and yet this information is critical to provide sufficient evidence to develop appropriate guidelines.
Our recommendations for gathering new data
The kind of data we would find most valuable would assess how different screening strategies impact overall mortality and breast cancer-specific mortality. It would require decades of follow-up—which of course means that screening technology will change over that time. A surrogate for evaluating overall survival is to look at interval cancers, which are all breast cancers diagnosed following negative mammograms and prior to the next screening. These cancers may or may not be biologically active, again focusing us on the need to look at overall survival of the patient. In addition, reducing breast cancer mortality may not reduce overall mortality, because the treatment for breast cancer may cause heart disease, or osteoporosis, or something else that impacts overall survival. These are important considerations for women and physicians who are making choices on treatment. What matters to a patient are 2 overlapping questions:
- Do I have a life-threatening condition or do I not?
- Has screening identified a condition that might lead to treatment that’s unnecessary?
The problem is that with breast cancer we can’t tell the difference. We do not understand the biological potential of a lesion when we evaluate an image on MRI, or computed tomography (CT), or mammography.
A re-look at presented data
A trial conducted by Bakker and colleagues6 was discussed by the authors of the DenseBreast-info.org quiz in which they recommended breast MRI for all women with extremely dense breasts (but no other risk factors for breast cancer) detected on screening mammograms.7 The Bakker study was large and conducted in the Netherlands. The primary outcome of the trial was to compare the incidence of interval breast cancers of women aged 50 to 75 randomly assigned to MRI versus those assigned to continued screening mammography every 2 years. Importantly, among the more than 8,000 women who were assigned to MRI, 59%, or fewer than two-thirds, chose to actually undergo MRI.
Among women randomized to MRI, 20 interval cancers were found—4 were diagnosed in those who actually had MRIs, and 16 were diagnosed among women who were randomized to MRI but didn’t undergo the study. Among women assigned to screening mammography only, 161 interval cancers were diagnosed among more than 32,000 women screened. The primary outcome findings were 2.5 interval cancers per 1,000 screenings among women randomly assigned to MRI, and 5 interval cancers per 1,000 screenings among those randomly assigned to mammography only.6
Because the trial included women aged 50 and older, we can’t apply these results to younger women, who often undergo screening mammography in the United States. In addition, the majority of the population in the Netherlands are of Western European ethnicity, a less-diverse population of women than in the United States. Furthermore, among the tumors that were detected in the MRI group, a larger proportion were DCIS, early-stage tumors, well differentiated, and hormone receptor-positive. This observation supports that many of the MRI-detected tumors were cases of overdiagnosis, or the detection of tumors destined not to cause clinical problems for the patient during her lifetime, or for which earlier diagnosis would impact survival.
We also know that treatment of these small ER-positive tumors carries risks for patients, as we may treat them by depriving a patient of estrogen for the rest of her life, with potential consequences of sexual dysfunction, osteoporosis, and perhaps cardiovascular disease depending on her age at the time of that diagnosis. Weighing the risks and benefits of not only treatment but also use of more sensitive screening techniques such as MRI is extremely important. Although Bakker and colleagues’ study results are interesting, we do not feel they support routinely recommending MRI for women found to have extremely dense breasts with mammography.
Overdiagnosis: A difficult concept
One reason overdiagnosis is so challenging to understand is that it can’t be directly measured, which makes comprehending it that much more problematic for clinicians and our patients.
One way to help grasp the overall issue is to compare screening mammography with cervical and colon cancer screening.
We are well aware that cervical cancer screening has reduced the incidence of mortality from invasive cervical cancer.8 We can argue very validly that the biggest success in any cancer screening program in history and globally has been cervical cancer screening. Our specialty, in particular, should feel proud about this. Screening colonoscopy also has repeatedly been found to reduce colon cancer mortality.9 For breast cancer, decades of media messaging have emphasized the benefits of screening mammograms; however, in contrast with cervical cancer screening and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic or advanced disease. Danish authors pointed out in 2017 that screening mammography has not achieved the hoped for or the promised reduction in breast cancer mortality.10
A report published in the March 2022, issue of Annals of Internal Medicine used modeling techniques to estimate the incidence of overdiagnosis and concluded that, among women aged 50-74 years receiving biennial screening mammograms (consistent with USPSTF recommendations), more than 15% of screen-detected breast cancers would represent cases of overdiagnosis. Of note, the study authors found that, among screen-detected cancers, the proportion representing overdiagnosis among women in their 60s (16.7%) and early 70s (23.6%) was higher than among women in their 50s-60s (11.5%-11.6%).11
The former Chief Medical and Scientific Officer for the American Cancer Society Otis Brawley, MD, has stated that, at the same time that breast cancer screening should not be abandoned, “We must acknowledge that overdiagnosis is common. The benefits of screening have been overstated, and some patients considered as ‘cured’ from breast cancer have, in fact, been harmed by unneeded treatment.”12
“Everybody loves early detection,” said Donald Berry, PhD, from MD Anderson Cancer Center, “but it comes with harms.” He points out that mortality rates have improved for breast cancer, but he attributes it to improved treatment. “The harms [of screening] we know, but the benefits of screening are very uncertain.”13
The importance of health equity is receiving more attention. When examining equity according to breast cancer mortality, ethnic minority populations have worse cancer survival outcomes than White women; the mortality rate is 40% higher among Black women than among White women.1 Lower survival rates are also noted among lower socioeconomic groups and among women who live in rural areas. Lower survival rates among ethnic minority women are also noted for cervical and colorectal cancers.2
In the past, these disparities in mortality were attributed to the historically lower breast cancer screening rates among Black women compared with White women. However, decades of efforts to increase mammography rates have effectively addressed much of the racial/ethnic gap in screening rates.1 In fact, a 2021 study showed Black and Hispanic women to have 6% to 10% higher rates of breast, cervical, and colorectal cancer screening than White women according to US Preventive Services Task Force guidelines.2 The study authors point out that other national data have demonstrated similar results and conclude that “higher cancer mortality among racial/ethnic minority groups will not be reduced solely by increasing rates of cancer screening. Although preventive screenings and timely diagnosis are important elements of prognosis, they are just 2 elements of many along the cancer care continuum that need to be addressed to eliminate disparities in cancer mortality.”
Unfortunately, the randomized trials that have been conducted on mammography have been conducted overwhelmingly in White populations. National registry studies from the Netherlands and Sweden are not representative patient populations for the United States. Recently, the US government proposed an ambitious plan to cut cancer mortality rates and has promised vast amounts of research funding to achieve that goal.3 Hopefully, this funding will support studies which enroll diverse patient populations. We hope to gain knowledge on what elements along the cancer care continuum can be addressed to better reduce or eliminate cancer mortality inequities.
References
1. National Cancer Institute. SEER Explorer. https://seer.cancer.gov/explorer/. Accessed February 9, 2022.
2. Benavidez GA, Zgodic A, Zahnd WE, Eberth JM. Disparities in Meeting USPSTF Breast, Cervical, and Colorectal Cancer Screening Guidelines Among Women in the United States. Prev Chronic Dis. 2021;18:200315. doi: http://dx.doi.org/10.5888/pcd18.200315.
3. Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
Continue to: Limitations of breast MRI...
Limitations of breast MRI
Overall, MRI is a diagnostic and monitoring test. It is costlier than mammography, and because it is not recommended in guidelines as a screening modality for most women, it is not typically covered by insurance. Abbreviated (rapid) MRI is a non-standardized imaging strategy being used at a few health centers. It has a shorter protocol overall than MRI, so it takes less time than current MRI and is less expensive, but there are few data on sensitivity and specificity. It is yet to be determined which populations could benefit from this newer technology.
As mentioned, 41% of women in the Bakker et al trial who were randomly assigned to breast MRI chose not to proceed with that exam even though it would have been at no cost to them.6 Anecdotally, some patients who have undergone MRI say they would forgo it a second time as a screening modality because it was a very unpleasant, stressful experience. It’s not a perfect test, although it is more sensitive than mammography.
Other options for following up dense-breast screening. Besides MRI and abbreviated MRI, the following modalities can be used to evaluate women found to have dense breasts with screening mammograms: CT mammography with contrast, molecular breast imaging, and ultrasonography.
Screening and treatment advances
3D mammography. In the US, the great majority of screening mammography now is performed with tomosynthesis, or what our patients sometimes call 3D mammography. In fact, it is approaching standard of care. Women whose screening mammography includes tomosynthesis are less likely to experience a so-called callback for additional imaging with diagnostic mammography or breast ultrasonography.14
Liquid biopsy. A potential major advancement for making decisions about when to treat cancers in general involves determining the biological behavior of a tumor, based on analysis of either circulating tumor DNA or proteins in the blood. As more experience with this new technology accumulates, the role of liquid biopsies for breast cancer will expand.15 Liquid biopsies for screening remain investigational for now, but they hold tremendous potential.
Noninvasive proteomics. With the development of noninvasive proteomic biomarkers obtained from blood, saliva, or nipple aspiration fluid, there exists the possibility of not just evaluating an image of a tumor seen on a mammogram, but actually studying the biological characteristics of that lesion.16 The cost of this technology is far less in terms of resources than MRI or molecular-based imaging, and actually reveals the flaws with using image-based screening. With proteomics, we can tell whether or not a lump is generating proteins that are going to make that disease biologically meaningful, and treatment decisions can be based on that information. This idea has the potential to disrupt our current breast cancer screening paradigm.
Advocacy’s role in mandating legislation
Many advocacy groups lobby on Capitol Hill for legislation related to health care, but we don’t feel that is the best way to make scientific decisions, and it’s not the way to do medicine. Passionate people, who truly believe that their outcome would have been different had something else been done, have every right to advocate, and should. However, without longer-term data focusing on breast cancer and overall mortality, rather than surrogate outcomes like interval cancers, it is not clear that routinely recommending supplemental MRI will improve survival for women with extremely dense breasts. Unfortunately, overall, earlier diagnosis of highly aggressive breast cancer tumors does not result in better outcomes for patients. ●
- American College of Obstetricians and Gynecologists. Practice Bulletin number 179: breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130: e1-e16. doi: 10.1097/AOG.0000000000002158.
- Sui AL, U.S. Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296. doi: 10.7326/M15-2886.
- The American Society of Breast Surgeons. Position statement on screening mammography. https://www.breastsurgeons.org/docs /statements/Position-Statement-on-ScreeningMammography.pdf. Accessed February 15, 2022.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average-risk: update from the ACR and Society of Breast Imaging. J Am College Radiol. 2021;18:1280-1288.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Seitzman R, Berg W. Average-risk women with dense breasts—what breast screening is appropriate? OBG Manag. 2021;33:18-19. doi: 10.12788/obgm.0155.
- Gopalani SV, Janitz AE, Campbell JE. Cervical cancer incidence and mortality among non-hispanic African American and White women, United States, 1999-2015. J Natl Med Assoc. 2020;112:632-638. doi: 10.1016 /j.jnma.2020.06.007.
- Niikura R, Hirata Y, Suzuki N, et al. Colonoscopy reduces colorectal cancer mortality: a multicenter, long-term, colonoscopy-based cohort study. PLoS One. 2017;12:e0185294.
- Jørgensen KJ, Gøtzsche PC, Kalager M, et al. Breast cancer screening in Denmark. Ann Intern Med. 2017;167:524. doi: 10.7326/L17-0270.
- Ryser MD, Lange J, Inoue IL, et al. Estimation of breast cancer overdiagnosis in a U.S. breast screening cohort. Ann Intern Med. 2022 March 1. doi: 10.7326/M21-3577.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis. Ann Intern Med. 2017;166:364-365. doi:10.7326/M16-2850.
- Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
- Conant EF, Barlow WE, Herschorn SD, et al. Association of digital breast tomosynthesis vs digital mammography with cancer detection and recall rates by age and breast density. JAMA Oncol. 2019;5:635-642. doi: 10.1001 /jamaoncol.2018.7078.
- Tay TK, Tan PH. Liquid biopsy in breast cancer: a focused review. Arch Pathol Lab Med. 2021;145: 678-686. doi: 10.5858/arpa.2019-0559-RA.
- Debald M, Wolgarten M, Walgenbach-Brunagel G, et al. Non-invasive proteomics—thinking about personalized breast cancer screening and treatment. EPMA J. 2010;1:413-420. doi: 10.1007 /s13167-010-0039-9.
We have been interested in the quiz series focused on breast cancer screening for women with dense breasts presented in
The concerns with breast cancer in particular
Breast cancer is not cervical cancer. It isn’t one disease. It is a multitude of diseases that happen to show up in the breast. Some are relatively slow-growing—the kinds of cancers that lend themselves to screening and to early intervention. But other cancers are rapidly-growing; they show up no matter how often or what modality we use for screening. Our goal should be to find an approach to screening that can diagnose breast cancer at a stage where we can intervene and positively impact breast cancer specific and overall mortality.
Screening guidelines vary
The variety of screening guidelines published by different professional organizations reflect differing assumptions and sets of values related to the early diagnosis and treatment of breast cancer. (For a comprehensive table of current screening guidelines, see https://www.cdc.gov/cancer/breast/pdf/breast-cancer-screening-guidelines-508.pdf.)
ACOG’s approach—to offer screening at age 40 but to begin by at least age 50 and, through shared decision making with the patient, screen every 1 or 2 years—is focused on capturing as many cases as we can identify, while minimizing the harms of false-positives.1 The perspective of the US Preventive Services Task Force (USPSTF) recommendations (to screen every 2 years beginning at age 50) is at the population level, a cost-effective approach that will have the greatest benefit while minimizing harms in the population at large.2 The American Society of Breast Surgeons recommends screening to begin by age 40.3 Like the breast surgeons, radiologists dedicated to breast imaging are focused on an individual rather than a population level. They strive to identify each and every instance of possible cancer, and therefore recommend annual screening beginning at age 40.4 However, with more aggressive screening in average-risk women many cases of ductal carcinoma in situ (DCIS) are identified—a lesion that, if not detected, may not impact the woman’s health during her lifetime—representing what some might call “overdiagnosis.” Yet there may be some instances in which the DCIS might affect an individual woman’s health. Unfortunately, we can’t prospectively distinguish between the first and the second types of cases.
We follow American College of Obstetricians and Gynecologists and US Preventive Services Task Force guidelines in discussing screening (both its hazards and benefits) with our average-risk patients beginning at age 40. We talk about risk factors for breast cancer, including breast density, but let patients know that no specific additional imaging is advised, and that density is more common in younger women (one consideration in earlier screening) and is quite common in general. Although we do not send follow-up letters to patients with dense breasts, we do educate our staff so that they can respond appropriately should patients call with questions.
Of course, we all bring to the table values that will impact the decisions that we make for ourselves and for our patients. What an ObGyn might suggest may differ from what a radiologist might suggest. Although we follow recommendations made by the radiologist at screening, an ObGyn wants to take care of the whole human being. We are concerned with bones, heart, everything about the patient, so we approach a patient in a different way. These priorities are reflected in the current varying breast cancer screening guidelines.
Continue to: Research on breast cancer screening varies by design...
Research on breast cancer screening varies by design
There has not been a randomized clinical trial conducted on screening mammography since the days of the analog mammogram. The research that has been conducted is difficult to compare due to variations in screening ages and intervals, technology sensitivity, and patient adherence with recommended screening. Treatments for breast cancer also have changed dramatically over time, so the findings of older studies may no longer be relevant to current breast cancer screening. The kind of analysis that needs to be done is an interrupted time series, where you can look at the trajectory of breast cancer survival and whether screening mammography shifts that survival in any way.
One specific study from Australia measured the impact of newer available breast cancer treatments, including tamoxifen for women with receptor-positive tumors and newer chemotherapy strategies.5 The authors analyzed screening mammography trends in one large province where women aged 50 to 69 were offered biennial screening. Trends from the 1990s showed that more women were being screened over time. Simultaneously, however, advances in therapy were entering clinical practice. The researchers pointed to a substantial decline in mortality from breast cancer from the early 1980s until 2013. But their conclusion was that none of the decline in mortality for breast cancer could be attributed to screening mammography when they looked at time trends; from their perspective all of the important decline in breast cancer mortality resulted from better treatment. They concluded that government programs should not support screening mammography.5
That is a recommendation that we do not support. However, we do recognize the conundrum that mammography is less sensitive among those who have dense breasts. In order to have congruent professional guidelines, we support research funding to determine which types, starting ages, and intervals of screening would be best in various patient populations. The USPSTF cites data from studies performed in the 1980s based on outdated technology; more recent (and relevant) randomized clinical trials have not been performed, and yet this information is critical to provide sufficient evidence to develop appropriate guidelines.
Our recommendations for gathering new data
The kind of data we would find most valuable would assess how different screening strategies impact overall mortality and breast cancer-specific mortality. It would require decades of follow-up—which of course means that screening technology will change over that time. A surrogate for evaluating overall survival is to look at interval cancers, which are all breast cancers diagnosed following negative mammograms and prior to the next screening. These cancers may or may not be biologically active, again focusing us on the need to look at overall survival of the patient. In addition, reducing breast cancer mortality may not reduce overall mortality, because the treatment for breast cancer may cause heart disease, or osteoporosis, or something else that impacts overall survival. These are important considerations for women and physicians who are making choices on treatment. What matters to a patient are 2 overlapping questions:
- Do I have a life-threatening condition or do I not?
- Has screening identified a condition that might lead to treatment that’s unnecessary?
The problem is that with breast cancer we can’t tell the difference. We do not understand the biological potential of a lesion when we evaluate an image on MRI, or computed tomography (CT), or mammography.
A re-look at presented data
A trial conducted by Bakker and colleagues6 was discussed by the authors of the DenseBreast-info.org quiz in which they recommended breast MRI for all women with extremely dense breasts (but no other risk factors for breast cancer) detected on screening mammograms.7 The Bakker study was large and conducted in the Netherlands. The primary outcome of the trial was to compare the incidence of interval breast cancers of women aged 50 to 75 randomly assigned to MRI versus those assigned to continued screening mammography every 2 years. Importantly, among the more than 8,000 women who were assigned to MRI, 59%, or fewer than two-thirds, chose to actually undergo MRI.
Among women randomized to MRI, 20 interval cancers were found—4 were diagnosed in those who actually had MRIs, and 16 were diagnosed among women who were randomized to MRI but didn’t undergo the study. Among women assigned to screening mammography only, 161 interval cancers were diagnosed among more than 32,000 women screened. The primary outcome findings were 2.5 interval cancers per 1,000 screenings among women randomly assigned to MRI, and 5 interval cancers per 1,000 screenings among those randomly assigned to mammography only.6
Because the trial included women aged 50 and older, we can’t apply these results to younger women, who often undergo screening mammography in the United States. In addition, the majority of the population in the Netherlands are of Western European ethnicity, a less-diverse population of women than in the United States. Furthermore, among the tumors that were detected in the MRI group, a larger proportion were DCIS, early-stage tumors, well differentiated, and hormone receptor-positive. This observation supports that many of the MRI-detected tumors were cases of overdiagnosis, or the detection of tumors destined not to cause clinical problems for the patient during her lifetime, or for which earlier diagnosis would impact survival.
We also know that treatment of these small ER-positive tumors carries risks for patients, as we may treat them by depriving a patient of estrogen for the rest of her life, with potential consequences of sexual dysfunction, osteoporosis, and perhaps cardiovascular disease depending on her age at the time of that diagnosis. Weighing the risks and benefits of not only treatment but also use of more sensitive screening techniques such as MRI is extremely important. Although Bakker and colleagues’ study results are interesting, we do not feel they support routinely recommending MRI for women found to have extremely dense breasts with mammography.
Overdiagnosis: A difficult concept
One reason overdiagnosis is so challenging to understand is that it can’t be directly measured, which makes comprehending it that much more problematic for clinicians and our patients.
One way to help grasp the overall issue is to compare screening mammography with cervical and colon cancer screening.
We are well aware that cervical cancer screening has reduced the incidence of mortality from invasive cervical cancer.8 We can argue very validly that the biggest success in any cancer screening program in history and globally has been cervical cancer screening. Our specialty, in particular, should feel proud about this. Screening colonoscopy also has repeatedly been found to reduce colon cancer mortality.9 For breast cancer, decades of media messaging have emphasized the benefits of screening mammograms; however, in contrast with cervical cancer screening and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic or advanced disease. Danish authors pointed out in 2017 that screening mammography has not achieved the hoped for or the promised reduction in breast cancer mortality.10
A report published in the March 2022, issue of Annals of Internal Medicine used modeling techniques to estimate the incidence of overdiagnosis and concluded that, among women aged 50-74 years receiving biennial screening mammograms (consistent with USPSTF recommendations), more than 15% of screen-detected breast cancers would represent cases of overdiagnosis. Of note, the study authors found that, among screen-detected cancers, the proportion representing overdiagnosis among women in their 60s (16.7%) and early 70s (23.6%) was higher than among women in their 50s-60s (11.5%-11.6%).11
The former Chief Medical and Scientific Officer for the American Cancer Society Otis Brawley, MD, has stated that, at the same time that breast cancer screening should not be abandoned, “We must acknowledge that overdiagnosis is common. The benefits of screening have been overstated, and some patients considered as ‘cured’ from breast cancer have, in fact, been harmed by unneeded treatment.”12
“Everybody loves early detection,” said Donald Berry, PhD, from MD Anderson Cancer Center, “but it comes with harms.” He points out that mortality rates have improved for breast cancer, but he attributes it to improved treatment. “The harms [of screening] we know, but the benefits of screening are very uncertain.”13
The importance of health equity is receiving more attention. When examining equity according to breast cancer mortality, ethnic minority populations have worse cancer survival outcomes than White women; the mortality rate is 40% higher among Black women than among White women.1 Lower survival rates are also noted among lower socioeconomic groups and among women who live in rural areas. Lower survival rates among ethnic minority women are also noted for cervical and colorectal cancers.2
In the past, these disparities in mortality were attributed to the historically lower breast cancer screening rates among Black women compared with White women. However, decades of efforts to increase mammography rates have effectively addressed much of the racial/ethnic gap in screening rates.1 In fact, a 2021 study showed Black and Hispanic women to have 6% to 10% higher rates of breast, cervical, and colorectal cancer screening than White women according to US Preventive Services Task Force guidelines.2 The study authors point out that other national data have demonstrated similar results and conclude that “higher cancer mortality among racial/ethnic minority groups will not be reduced solely by increasing rates of cancer screening. Although preventive screenings and timely diagnosis are important elements of prognosis, they are just 2 elements of many along the cancer care continuum that need to be addressed to eliminate disparities in cancer mortality.”
Unfortunately, the randomized trials that have been conducted on mammography have been conducted overwhelmingly in White populations. National registry studies from the Netherlands and Sweden are not representative patient populations for the United States. Recently, the US government proposed an ambitious plan to cut cancer mortality rates and has promised vast amounts of research funding to achieve that goal.3 Hopefully, this funding will support studies which enroll diverse patient populations. We hope to gain knowledge on what elements along the cancer care continuum can be addressed to better reduce or eliminate cancer mortality inequities.
References
1. National Cancer Institute. SEER Explorer. https://seer.cancer.gov/explorer/. Accessed February 9, 2022.
2. Benavidez GA, Zgodic A, Zahnd WE, Eberth JM. Disparities in Meeting USPSTF Breast, Cervical, and Colorectal Cancer Screening Guidelines Among Women in the United States. Prev Chronic Dis. 2021;18:200315. doi: http://dx.doi.org/10.5888/pcd18.200315.
3. Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
Continue to: Limitations of breast MRI...
Limitations of breast MRI
Overall, MRI is a diagnostic and monitoring test. It is costlier than mammography, and because it is not recommended in guidelines as a screening modality for most women, it is not typically covered by insurance. Abbreviated (rapid) MRI is a non-standardized imaging strategy being used at a few health centers. It has a shorter protocol overall than MRI, so it takes less time than current MRI and is less expensive, but there are few data on sensitivity and specificity. It is yet to be determined which populations could benefit from this newer technology.
As mentioned, 41% of women in the Bakker et al trial who were randomly assigned to breast MRI chose not to proceed with that exam even though it would have been at no cost to them.6 Anecdotally, some patients who have undergone MRI say they would forgo it a second time as a screening modality because it was a very unpleasant, stressful experience. It’s not a perfect test, although it is more sensitive than mammography.
Other options for following up dense-breast screening. Besides MRI and abbreviated MRI, the following modalities can be used to evaluate women found to have dense breasts with screening mammograms: CT mammography with contrast, molecular breast imaging, and ultrasonography.
Screening and treatment advances
3D mammography. In the US, the great majority of screening mammography now is performed with tomosynthesis, or what our patients sometimes call 3D mammography. In fact, it is approaching standard of care. Women whose screening mammography includes tomosynthesis are less likely to experience a so-called callback for additional imaging with diagnostic mammography or breast ultrasonography.14
Liquid biopsy. A potential major advancement for making decisions about when to treat cancers in general involves determining the biological behavior of a tumor, based on analysis of either circulating tumor DNA or proteins in the blood. As more experience with this new technology accumulates, the role of liquid biopsies for breast cancer will expand.15 Liquid biopsies for screening remain investigational for now, but they hold tremendous potential.
Noninvasive proteomics. With the development of noninvasive proteomic biomarkers obtained from blood, saliva, or nipple aspiration fluid, there exists the possibility of not just evaluating an image of a tumor seen on a mammogram, but actually studying the biological characteristics of that lesion.16 The cost of this technology is far less in terms of resources than MRI or molecular-based imaging, and actually reveals the flaws with using image-based screening. With proteomics, we can tell whether or not a lump is generating proteins that are going to make that disease biologically meaningful, and treatment decisions can be based on that information. This idea has the potential to disrupt our current breast cancer screening paradigm.
Advocacy’s role in mandating legislation
Many advocacy groups lobby on Capitol Hill for legislation related to health care, but we don’t feel that is the best way to make scientific decisions, and it’s not the way to do medicine. Passionate people, who truly believe that their outcome would have been different had something else been done, have every right to advocate, and should. However, without longer-term data focusing on breast cancer and overall mortality, rather than surrogate outcomes like interval cancers, it is not clear that routinely recommending supplemental MRI will improve survival for women with extremely dense breasts. Unfortunately, overall, earlier diagnosis of highly aggressive breast cancer tumors does not result in better outcomes for patients. ●
We have been interested in the quiz series focused on breast cancer screening for women with dense breasts presented in
The concerns with breast cancer in particular
Breast cancer is not cervical cancer. It isn’t one disease. It is a multitude of diseases that happen to show up in the breast. Some are relatively slow-growing—the kinds of cancers that lend themselves to screening and to early intervention. But other cancers are rapidly-growing; they show up no matter how often or what modality we use for screening. Our goal should be to find an approach to screening that can diagnose breast cancer at a stage where we can intervene and positively impact breast cancer specific and overall mortality.
Screening guidelines vary
The variety of screening guidelines published by different professional organizations reflect differing assumptions and sets of values related to the early diagnosis and treatment of breast cancer. (For a comprehensive table of current screening guidelines, see https://www.cdc.gov/cancer/breast/pdf/breast-cancer-screening-guidelines-508.pdf.)
ACOG’s approach—to offer screening at age 40 but to begin by at least age 50 and, through shared decision making with the patient, screen every 1 or 2 years—is focused on capturing as many cases as we can identify, while minimizing the harms of false-positives.1 The perspective of the US Preventive Services Task Force (USPSTF) recommendations (to screen every 2 years beginning at age 50) is at the population level, a cost-effective approach that will have the greatest benefit while minimizing harms in the population at large.2 The American Society of Breast Surgeons recommends screening to begin by age 40.3 Like the breast surgeons, radiologists dedicated to breast imaging are focused on an individual rather than a population level. They strive to identify each and every instance of possible cancer, and therefore recommend annual screening beginning at age 40.4 However, with more aggressive screening in average-risk women many cases of ductal carcinoma in situ (DCIS) are identified—a lesion that, if not detected, may not impact the woman’s health during her lifetime—representing what some might call “overdiagnosis.” Yet there may be some instances in which the DCIS might affect an individual woman’s health. Unfortunately, we can’t prospectively distinguish between the first and the second types of cases.
We follow American College of Obstetricians and Gynecologists and US Preventive Services Task Force guidelines in discussing screening (both its hazards and benefits) with our average-risk patients beginning at age 40. We talk about risk factors for breast cancer, including breast density, but let patients know that no specific additional imaging is advised, and that density is more common in younger women (one consideration in earlier screening) and is quite common in general. Although we do not send follow-up letters to patients with dense breasts, we do educate our staff so that they can respond appropriately should patients call with questions.
Of course, we all bring to the table values that will impact the decisions that we make for ourselves and for our patients. What an ObGyn might suggest may differ from what a radiologist might suggest. Although we follow recommendations made by the radiologist at screening, an ObGyn wants to take care of the whole human being. We are concerned with bones, heart, everything about the patient, so we approach a patient in a different way. These priorities are reflected in the current varying breast cancer screening guidelines.
Continue to: Research on breast cancer screening varies by design...
Research on breast cancer screening varies by design
There has not been a randomized clinical trial conducted on screening mammography since the days of the analog mammogram. The research that has been conducted is difficult to compare due to variations in screening ages and intervals, technology sensitivity, and patient adherence with recommended screening. Treatments for breast cancer also have changed dramatically over time, so the findings of older studies may no longer be relevant to current breast cancer screening. The kind of analysis that needs to be done is an interrupted time series, where you can look at the trajectory of breast cancer survival and whether screening mammography shifts that survival in any way.
One specific study from Australia measured the impact of newer available breast cancer treatments, including tamoxifen for women with receptor-positive tumors and newer chemotherapy strategies.5 The authors analyzed screening mammography trends in one large province where women aged 50 to 69 were offered biennial screening. Trends from the 1990s showed that more women were being screened over time. Simultaneously, however, advances in therapy were entering clinical practice. The researchers pointed to a substantial decline in mortality from breast cancer from the early 1980s until 2013. But their conclusion was that none of the decline in mortality for breast cancer could be attributed to screening mammography when they looked at time trends; from their perspective all of the important decline in breast cancer mortality resulted from better treatment. They concluded that government programs should not support screening mammography.5
That is a recommendation that we do not support. However, we do recognize the conundrum that mammography is less sensitive among those who have dense breasts. In order to have congruent professional guidelines, we support research funding to determine which types, starting ages, and intervals of screening would be best in various patient populations. The USPSTF cites data from studies performed in the 1980s based on outdated technology; more recent (and relevant) randomized clinical trials have not been performed, and yet this information is critical to provide sufficient evidence to develop appropriate guidelines.
Our recommendations for gathering new data
The kind of data we would find most valuable would assess how different screening strategies impact overall mortality and breast cancer-specific mortality. It would require decades of follow-up—which of course means that screening technology will change over that time. A surrogate for evaluating overall survival is to look at interval cancers, which are all breast cancers diagnosed following negative mammograms and prior to the next screening. These cancers may or may not be biologically active, again focusing us on the need to look at overall survival of the patient. In addition, reducing breast cancer mortality may not reduce overall mortality, because the treatment for breast cancer may cause heart disease, or osteoporosis, or something else that impacts overall survival. These are important considerations for women and physicians who are making choices on treatment. What matters to a patient are 2 overlapping questions:
- Do I have a life-threatening condition or do I not?
- Has screening identified a condition that might lead to treatment that’s unnecessary?
The problem is that with breast cancer we can’t tell the difference. We do not understand the biological potential of a lesion when we evaluate an image on MRI, or computed tomography (CT), or mammography.
A re-look at presented data
A trial conducted by Bakker and colleagues6 was discussed by the authors of the DenseBreast-info.org quiz in which they recommended breast MRI for all women with extremely dense breasts (but no other risk factors for breast cancer) detected on screening mammograms.7 The Bakker study was large and conducted in the Netherlands. The primary outcome of the trial was to compare the incidence of interval breast cancers of women aged 50 to 75 randomly assigned to MRI versus those assigned to continued screening mammography every 2 years. Importantly, among the more than 8,000 women who were assigned to MRI, 59%, or fewer than two-thirds, chose to actually undergo MRI.
Among women randomized to MRI, 20 interval cancers were found—4 were diagnosed in those who actually had MRIs, and 16 were diagnosed among women who were randomized to MRI but didn’t undergo the study. Among women assigned to screening mammography only, 161 interval cancers were diagnosed among more than 32,000 women screened. The primary outcome findings were 2.5 interval cancers per 1,000 screenings among women randomly assigned to MRI, and 5 interval cancers per 1,000 screenings among those randomly assigned to mammography only.6
Because the trial included women aged 50 and older, we can’t apply these results to younger women, who often undergo screening mammography in the United States. In addition, the majority of the population in the Netherlands are of Western European ethnicity, a less-diverse population of women than in the United States. Furthermore, among the tumors that were detected in the MRI group, a larger proportion were DCIS, early-stage tumors, well differentiated, and hormone receptor-positive. This observation supports that many of the MRI-detected tumors were cases of overdiagnosis, or the detection of tumors destined not to cause clinical problems for the patient during her lifetime, or for which earlier diagnosis would impact survival.
We also know that treatment of these small ER-positive tumors carries risks for patients, as we may treat them by depriving a patient of estrogen for the rest of her life, with potential consequences of sexual dysfunction, osteoporosis, and perhaps cardiovascular disease depending on her age at the time of that diagnosis. Weighing the risks and benefits of not only treatment but also use of more sensitive screening techniques such as MRI is extremely important. Although Bakker and colleagues’ study results are interesting, we do not feel they support routinely recommending MRI for women found to have extremely dense breasts with mammography.
Overdiagnosis: A difficult concept
One reason overdiagnosis is so challenging to understand is that it can’t be directly measured, which makes comprehending it that much more problematic for clinicians and our patients.
One way to help grasp the overall issue is to compare screening mammography with cervical and colon cancer screening.
We are well aware that cervical cancer screening has reduced the incidence of mortality from invasive cervical cancer.8 We can argue very validly that the biggest success in any cancer screening program in history and globally has been cervical cancer screening. Our specialty, in particular, should feel proud about this. Screening colonoscopy also has repeatedly been found to reduce colon cancer mortality.9 For breast cancer, decades of media messaging have emphasized the benefits of screening mammograms; however, in contrast with cervical cancer screening and colonoscopy, screening mammography has not reduced the incidence of breast cancer presenting with metastatic or advanced disease. Danish authors pointed out in 2017 that screening mammography has not achieved the hoped for or the promised reduction in breast cancer mortality.10
A report published in the March 2022, issue of Annals of Internal Medicine used modeling techniques to estimate the incidence of overdiagnosis and concluded that, among women aged 50-74 years receiving biennial screening mammograms (consistent with USPSTF recommendations), more than 15% of screen-detected breast cancers would represent cases of overdiagnosis. Of note, the study authors found that, among screen-detected cancers, the proportion representing overdiagnosis among women in their 60s (16.7%) and early 70s (23.6%) was higher than among women in their 50s-60s (11.5%-11.6%).11
The former Chief Medical and Scientific Officer for the American Cancer Society Otis Brawley, MD, has stated that, at the same time that breast cancer screening should not be abandoned, “We must acknowledge that overdiagnosis is common. The benefits of screening have been overstated, and some patients considered as ‘cured’ from breast cancer have, in fact, been harmed by unneeded treatment.”12
“Everybody loves early detection,” said Donald Berry, PhD, from MD Anderson Cancer Center, “but it comes with harms.” He points out that mortality rates have improved for breast cancer, but he attributes it to improved treatment. “The harms [of screening] we know, but the benefits of screening are very uncertain.”13
The importance of health equity is receiving more attention. When examining equity according to breast cancer mortality, ethnic minority populations have worse cancer survival outcomes than White women; the mortality rate is 40% higher among Black women than among White women.1 Lower survival rates are also noted among lower socioeconomic groups and among women who live in rural areas. Lower survival rates among ethnic minority women are also noted for cervical and colorectal cancers.2
In the past, these disparities in mortality were attributed to the historically lower breast cancer screening rates among Black women compared with White women. However, decades of efforts to increase mammography rates have effectively addressed much of the racial/ethnic gap in screening rates.1 In fact, a 2021 study showed Black and Hispanic women to have 6% to 10% higher rates of breast, cervical, and colorectal cancer screening than White women according to US Preventive Services Task Force guidelines.2 The study authors point out that other national data have demonstrated similar results and conclude that “higher cancer mortality among racial/ethnic minority groups will not be reduced solely by increasing rates of cancer screening. Although preventive screenings and timely diagnosis are important elements of prognosis, they are just 2 elements of many along the cancer care continuum that need to be addressed to eliminate disparities in cancer mortality.”
Unfortunately, the randomized trials that have been conducted on mammography have been conducted overwhelmingly in White populations. National registry studies from the Netherlands and Sweden are not representative patient populations for the United States. Recently, the US government proposed an ambitious plan to cut cancer mortality rates and has promised vast amounts of research funding to achieve that goal.3 Hopefully, this funding will support studies which enroll diverse patient populations. We hope to gain knowledge on what elements along the cancer care continuum can be addressed to better reduce or eliminate cancer mortality inequities.
References
1. National Cancer Institute. SEER Explorer. https://seer.cancer.gov/explorer/. Accessed February 9, 2022.
2. Benavidez GA, Zgodic A, Zahnd WE, Eberth JM. Disparities in Meeting USPSTF Breast, Cervical, and Colorectal Cancer Screening Guidelines Among Women in the United States. Prev Chronic Dis. 2021;18:200315. doi: http://dx.doi.org/10.5888/pcd18.200315.
3. Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
Continue to: Limitations of breast MRI...
Limitations of breast MRI
Overall, MRI is a diagnostic and monitoring test. It is costlier than mammography, and because it is not recommended in guidelines as a screening modality for most women, it is not typically covered by insurance. Abbreviated (rapid) MRI is a non-standardized imaging strategy being used at a few health centers. It has a shorter protocol overall than MRI, so it takes less time than current MRI and is less expensive, but there are few data on sensitivity and specificity. It is yet to be determined which populations could benefit from this newer technology.
As mentioned, 41% of women in the Bakker et al trial who were randomly assigned to breast MRI chose not to proceed with that exam even though it would have been at no cost to them.6 Anecdotally, some patients who have undergone MRI say they would forgo it a second time as a screening modality because it was a very unpleasant, stressful experience. It’s not a perfect test, although it is more sensitive than mammography.
Other options for following up dense-breast screening. Besides MRI and abbreviated MRI, the following modalities can be used to evaluate women found to have dense breasts with screening mammograms: CT mammography with contrast, molecular breast imaging, and ultrasonography.
Screening and treatment advances
3D mammography. In the US, the great majority of screening mammography now is performed with tomosynthesis, or what our patients sometimes call 3D mammography. In fact, it is approaching standard of care. Women whose screening mammography includes tomosynthesis are less likely to experience a so-called callback for additional imaging with diagnostic mammography or breast ultrasonography.14
Liquid biopsy. A potential major advancement for making decisions about when to treat cancers in general involves determining the biological behavior of a tumor, based on analysis of either circulating tumor DNA or proteins in the blood. As more experience with this new technology accumulates, the role of liquid biopsies for breast cancer will expand.15 Liquid biopsies for screening remain investigational for now, but they hold tremendous potential.
Noninvasive proteomics. With the development of noninvasive proteomic biomarkers obtained from blood, saliva, or nipple aspiration fluid, there exists the possibility of not just evaluating an image of a tumor seen on a mammogram, but actually studying the biological characteristics of that lesion.16 The cost of this technology is far less in terms of resources than MRI or molecular-based imaging, and actually reveals the flaws with using image-based screening. With proteomics, we can tell whether or not a lump is generating proteins that are going to make that disease biologically meaningful, and treatment decisions can be based on that information. This idea has the potential to disrupt our current breast cancer screening paradigm.
Advocacy’s role in mandating legislation
Many advocacy groups lobby on Capitol Hill for legislation related to health care, but we don’t feel that is the best way to make scientific decisions, and it’s not the way to do medicine. Passionate people, who truly believe that their outcome would have been different had something else been done, have every right to advocate, and should. However, without longer-term data focusing on breast cancer and overall mortality, rather than surrogate outcomes like interval cancers, it is not clear that routinely recommending supplemental MRI will improve survival for women with extremely dense breasts. Unfortunately, overall, earlier diagnosis of highly aggressive breast cancer tumors does not result in better outcomes for patients. ●
- American College of Obstetricians and Gynecologists. Practice Bulletin number 179: breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130: e1-e16. doi: 10.1097/AOG.0000000000002158.
- Sui AL, U.S. Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296. doi: 10.7326/M15-2886.
- The American Society of Breast Surgeons. Position statement on screening mammography. https://www.breastsurgeons.org/docs /statements/Position-Statement-on-ScreeningMammography.pdf. Accessed February 15, 2022.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average-risk: update from the ACR and Society of Breast Imaging. J Am College Radiol. 2021;18:1280-1288.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Seitzman R, Berg W. Average-risk women with dense breasts—what breast screening is appropriate? OBG Manag. 2021;33:18-19. doi: 10.12788/obgm.0155.
- Gopalani SV, Janitz AE, Campbell JE. Cervical cancer incidence and mortality among non-hispanic African American and White women, United States, 1999-2015. J Natl Med Assoc. 2020;112:632-638. doi: 10.1016 /j.jnma.2020.06.007.
- Niikura R, Hirata Y, Suzuki N, et al. Colonoscopy reduces colorectal cancer mortality: a multicenter, long-term, colonoscopy-based cohort study. PLoS One. 2017;12:e0185294.
- Jørgensen KJ, Gøtzsche PC, Kalager M, et al. Breast cancer screening in Denmark. Ann Intern Med. 2017;167:524. doi: 10.7326/L17-0270.
- Ryser MD, Lange J, Inoue IL, et al. Estimation of breast cancer overdiagnosis in a U.S. breast screening cohort. Ann Intern Med. 2022 March 1. doi: 10.7326/M21-3577.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis. Ann Intern Med. 2017;166:364-365. doi:10.7326/M16-2850.
- Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
- Conant EF, Barlow WE, Herschorn SD, et al. Association of digital breast tomosynthesis vs digital mammography with cancer detection and recall rates by age and breast density. JAMA Oncol. 2019;5:635-642. doi: 10.1001 /jamaoncol.2018.7078.
- Tay TK, Tan PH. Liquid biopsy in breast cancer: a focused review. Arch Pathol Lab Med. 2021;145: 678-686. doi: 10.5858/arpa.2019-0559-RA.
- Debald M, Wolgarten M, Walgenbach-Brunagel G, et al. Non-invasive proteomics—thinking about personalized breast cancer screening and treatment. EPMA J. 2010;1:413-420. doi: 10.1007 /s13167-010-0039-9.
- American College of Obstetricians and Gynecologists. Practice Bulletin number 179: breast cancer risk assessment and screening in average-risk women. Obstet Gynecol. 2017;130: e1-e16. doi: 10.1097/AOG.0000000000002158.
- Sui AL, U.S. Preventive Services Task Force. Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2016;164:279-296. doi: 10.7326/M15-2886.
- The American Society of Breast Surgeons. Position statement on screening mammography. https://www.breastsurgeons.org/docs /statements/Position-Statement-on-ScreeningMammography.pdf. Accessed February 15, 2022.
- Monticciolo DL, Malak SF, Friedewald SM, et al. Breast cancer screening recommendations inclusive of all women at average-risk: update from the ACR and Society of Breast Imaging. J Am College Radiol. 2021;18:1280-1288.
- Burton R, Stevenson C. Assessment of breast cancer mortality trends associated with mammographic screening and adjuvant therapy from 1986 to 2013 in the state of Victoria, Australia. JAMA Netw Open. 2020;3:e208249.
- Bakker MF, de Lange SV, Pijnappel RM, et al. Supplemental MRI screening for women with extremely dense breast tissue. N Engl J Med. 2019;381:2091-2102. doi: 10.1056/NEJMoa1903986.
- Seitzman R, Berg W. Average-risk women with dense breasts—what breast screening is appropriate? OBG Manag. 2021;33:18-19. doi: 10.12788/obgm.0155.
- Gopalani SV, Janitz AE, Campbell JE. Cervical cancer incidence and mortality among non-hispanic African American and White women, United States, 1999-2015. J Natl Med Assoc. 2020;112:632-638. doi: 10.1016 /j.jnma.2020.06.007.
- Niikura R, Hirata Y, Suzuki N, et al. Colonoscopy reduces colorectal cancer mortality: a multicenter, long-term, colonoscopy-based cohort study. PLoS One. 2017;12:e0185294.
- Jørgensen KJ, Gøtzsche PC, Kalager M, et al. Breast cancer screening in Denmark. Ann Intern Med. 2017;167:524. doi: 10.7326/L17-0270.
- Ryser MD, Lange J, Inoue IL, et al. Estimation of breast cancer overdiagnosis in a U.S. breast screening cohort. Ann Intern Med. 2022 March 1. doi: 10.7326/M21-3577.
- Brawley OW. Accepting the existence of breast cancer overdiagnosis. Ann Intern Med. 2017;166:364-365. doi:10.7326/M16-2850.
- Stohlberg SG, Kolata G. Biden presents ambitious plan to cut cancer death rate in half. The New York Times. February 2, 2022.
- Conant EF, Barlow WE, Herschorn SD, et al. Association of digital breast tomosynthesis vs digital mammography with cancer detection and recall rates by age and breast density. JAMA Oncol. 2019;5:635-642. doi: 10.1001 /jamaoncol.2018.7078.
- Tay TK, Tan PH. Liquid biopsy in breast cancer: a focused review. Arch Pathol Lab Med. 2021;145: 678-686. doi: 10.5858/arpa.2019-0559-RA.
- Debald M, Wolgarten M, Walgenbach-Brunagel G, et al. Non-invasive proteomics—thinking about personalized breast cancer screening and treatment. EPMA J. 2010;1:413-420. doi: 10.1007 /s13167-010-0039-9.
Telehealth apps in ObGyn practice
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.
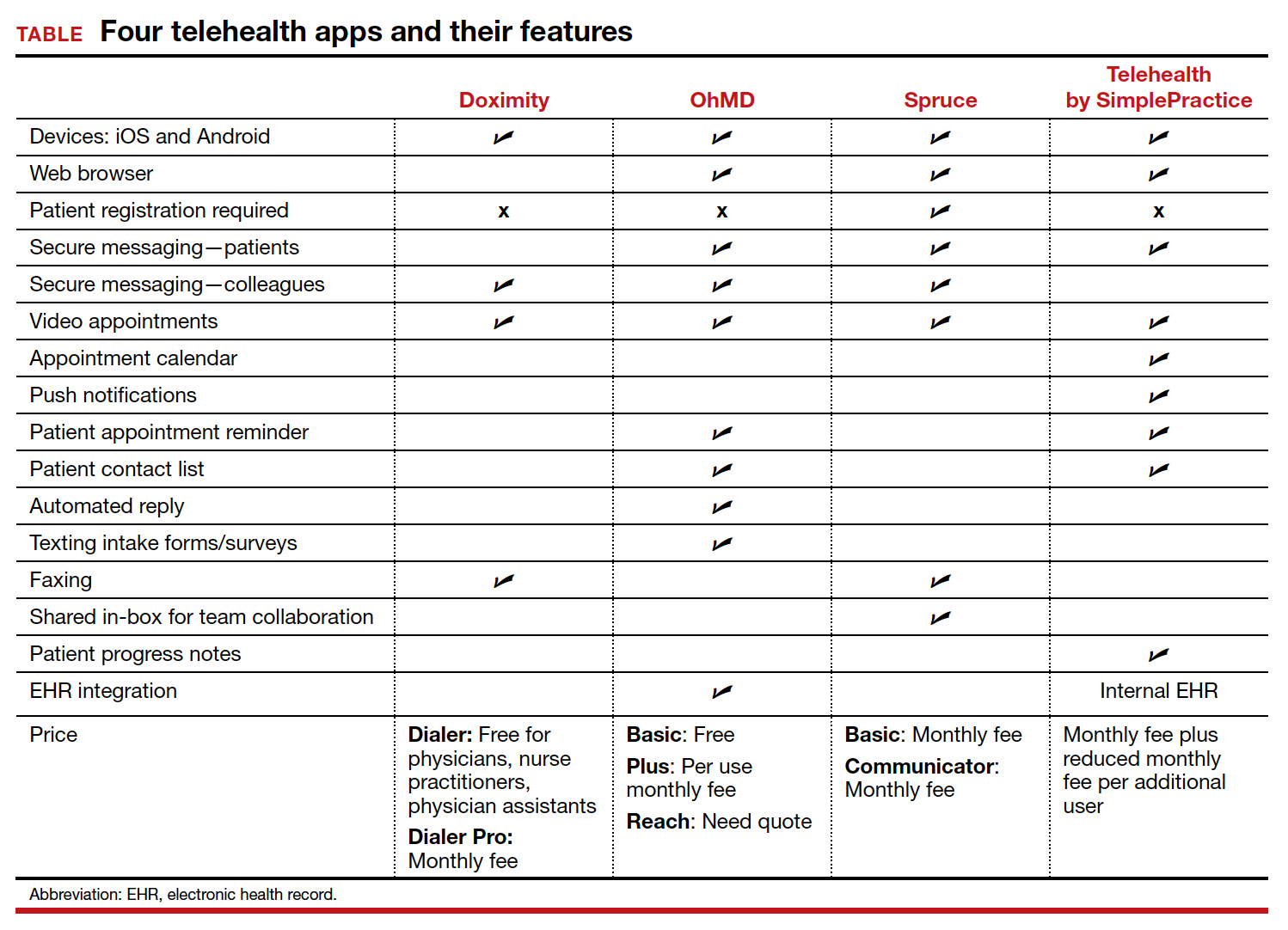
These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.

These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
The COVID-19 pandemic has presented increasing demands on health care systems internationally. In addition to redistribution of inpatient health care resources, outpatient care practices evolved, with health care providers offering streamlined access to care to patients via telehealth.
Due to updated insurance practices, physicians now can receive reimbursement via private insurers, Medicare, and Medicaid (as determined by states) for telehealth visits both related and unrelated to COVID-19 care. Increased telehealth use has advantages, including increased health care access, reduced in-clinic wait times, and reduced patient and physician travel time. Within the field of obstetrics and gynecology, clinicians have used telehealth to maintain access to prenatal maternity care while redirecting resources and minimizing the risk of COVID-19 transmission. Additional advantages include provision of care during expanded hours, including evenings and weekends, to increase patient access without increasing the demand on office support staff and the ability to bill for 5- to 10-minute phone counseling encounters.1 Research shows that patients express satisfaction regarding the quality of telehealth care in the setting of prenatal care.2
In February 2020, the American College of Obstetricians and Gynecologists (ACOG) released a Committee Opinion regarding telehealth use in ObGyn, a sign of telehealth’s likely long-standing role within the field.3 Within the statement, ACOG commented on the increasing application of telemedicine in all aspects of obstetrics and gynecology and recommended that physicians become acquainted with new technologies and consider using them in their practice.
There is a large opportunity for development of mobile applications (apps) to further streamline telehealth-based medical care. During the pandemic, the Centers for Medicare and Medicaid Services instituted waivers for telemedicine use on non-HIPAA (Health Insurance Portability and Accountability Act) compliant video communications products, such as Google+ Hangout and Skype. However, HIPAA-compliant video services are preferred, and many virtual apps have released methods for patient communication that meet HIPAA guidelines.1,4 These apps offer services such as phone- and video-based patient visits, appointment scheduling, secure physician-patient messaging, and electronic health record (EHR) documentation.
App recommendations
To identify current mobile apps with clinical use for the ObGyn, we conducted a search of the Apple App Store using the term “telehealth” between December 1, 2021 and January 1, 2022. We limited search results to apps that had at least 1,000 user ratings and to HIPAA-compliant user communication apps. Based on our review, we selected 4 apps to highlight here: Doximity, OhMD, Spruce, and Telehealth by SimplePractice (TABLE). We excluded apps that were advertised as having internal medical clinicians with first patient encounter on-demand through the app or that were associated with a singular insurance company or hospital system.

These apps are largely enabled for iOS and Android mobile devices and are offered at a range of price points for individual physician and practice-scale clinical implementation. Most apps offer secure messaging services between health care practitioners in addition to HIPAA-compliant patient messaging. Some apps offer additional features with the aim to increase patient attendance; these include push notifications, appointment reminders, and an option for automated replies with clinic information. For an additional fee, several apps offer integration to established EHR systems.
An additional tool
The COVID-19 pandemic caused health care systems and individual clinicians to rapidly evolve their practices to maintain patient access to essential health care. Notably, the pandemic led to accelerated implementation of virtual health care services. Telehealth apps likely will become another tool that ObGyns can use to improve the efficiency of their clinical practice and expand patient access to care. ●
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
- Karram M, Baum N. Telemedicine: a primer for today’s ObGyn. OBG Manag. 2020;32:28-32.
- Marko KI, Ganju N, Krapf JM, et al. A mobile prenatal care app to reduce in-person visits: prospective controlled trial. JMIR Mhealth Uhealth. 2019;7:e10520.
- American College of Obstetricians and Gynecologists. Implementing telehealth in practice: committee opinion no. 798. Obstet Gynecol. 2020;135:e73-e79.
- Karram M, Dooley A, de la Houssaye N, et al. Telemedicine: navigating legal issues. OBG Manag. 2020;32:18-24.
CPAP has only small effect on metabolic syndrome
Continuous positive airway pressure (CPAP) may be only modestly effective for ameliorating metabolic syndrome in patients with moderate to severe obstructive sleep apnea (OSA).
That conclusion comes from investigators in a randomized controlled, trial, who found that, among 100 patients with OSA and a recent diagnosis of metabolic syndrome (MS), 18% of those assigned to use CPAP at night had a reversal of MS at 6 months of follow-up, compared with 4% of controls who were assigned to use nasal strips at night (P = .04).
The majority of patients assigned to CPAP still retained their MS diagnoses at 6 months, and CPAP did not significantly reduce individual components of the syndrome. Use of CPAP was, however, associated with small reductions in visceral fat and improvement in endothelial function, reported Sara Q.C. Giampa, PhD, from the University of São Paulo, and colleagues.
“Despite a significant rate of MS reversibility after CPAP therapy, most of the patients maintained the MS diagnosis. The modest effects of CPAP on MS reversibility underscore the need for combined therapy with CPAP, aiming to maximize metabolic syndrome recovery in parallel with improvements in OSA severity and related symptoms,” according to their study, reported in the journal CHEST®.
Asked whether he still recommends CPAP to patients with OSA and the metabolic syndrome, given the findings, corresponding author Luciano F. Drager, MD, PhD, replied “yes, definitely.”
“Despite the modest rate in reversing metabolic syndrome after CPAP, the rate was 5-fold higher than non-effective treatment (18% vs. 4%),” he said in an interview.
Dr. Drager noted that studies of other single interventions such as physical exercise to reverse MS in patients with OSA also had modest results.
A researcher who studies the relationship between sleep, circadian rhythms, and metabolism commented that, although the patients in the CPAP group were compliant with the assigned equipment and had both reductions in apneic events and improvement in oxygen saturation, the effect of CPAP on the metabolic syndrome was rather small.
“The CPAP was doing what we thought it was supposed to do, but it didn’t have the magnitude of effect on the metabolic syndrome as I expected or I think as the authors expected,” said Deanna Arble, PhD, assistant professor of biological science at Marquette University, Milwaukee.
She noted that the study also failed to detect a significant improvement in the blood pressure component of metabolic syndrome.
“In my experience and my review of the literature, blood pressure tends to be the one that’s improved most dramatically with CPAP,” she said.
Dr. Arble was not involved in the study.
Study details
In the trial, titled TREATOSA-MS, the investigators enrolled 100 patients with a recent diagnosis of metabolic syndrome and moderate to severe OSA, defined as 15 or more apnea-hypopnea index events per hour. The patients were stratified by body mass index and then randomized to undergo therapeutic CPAP or to use nasal strips for 6 months.
At baseline and at the end of each intervention investigators measured anthropometric variables, blood pressure, glucose, and lipid profiles. They also leptin and adiponectin, body composition, food intake, physical activity, subcutaneous and abdominal fat (visceral and hepatic), and endothelial function to control for potential confounders.
As noted previously, they found that after 6 months “most patients with OSA randomized to CPAP retained the MS diagnosis, but the rate of MS reversibility was higher than observed in the placebo group.” The difference in metabolic syndrome reversal, 18% with CPAP versus 4% with nasal strips, translated into a hazard ratio favoring CPAP of 5.27 (P = .04).
Also as noted, in analyses adjusted for baseline values, CPAP did not significantly improve either weight, liver fat, lip profiles, or the adiposity biomarkers leptin and adiponectin, but did have “very modest” influence on reducing visceral fat and improving endothelial function.
Rigorous study
Dr. Arble said that most studies of the association between OSA and metabolic syndrome have focused on only one or two of the parameters that were included in the TREATOSA-MS study, giving the findings additional weight.
“This could potentially be a very good, carefully controlled first insight into how obstructive sleep apnea is related to the metabolic syndrome,” she said.
The study was funded by grants Fundação de Amparo Q22 à Pesquisa do Estado de São Paulo and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior. The authors and Dr. Arble reported having no conflicts of interest to disclose.
Continuous positive airway pressure (CPAP) may be only modestly effective for ameliorating metabolic syndrome in patients with moderate to severe obstructive sleep apnea (OSA).
That conclusion comes from investigators in a randomized controlled, trial, who found that, among 100 patients with OSA and a recent diagnosis of metabolic syndrome (MS), 18% of those assigned to use CPAP at night had a reversal of MS at 6 months of follow-up, compared with 4% of controls who were assigned to use nasal strips at night (P = .04).
The majority of patients assigned to CPAP still retained their MS diagnoses at 6 months, and CPAP did not significantly reduce individual components of the syndrome. Use of CPAP was, however, associated with small reductions in visceral fat and improvement in endothelial function, reported Sara Q.C. Giampa, PhD, from the University of São Paulo, and colleagues.
“Despite a significant rate of MS reversibility after CPAP therapy, most of the patients maintained the MS diagnosis. The modest effects of CPAP on MS reversibility underscore the need for combined therapy with CPAP, aiming to maximize metabolic syndrome recovery in parallel with improvements in OSA severity and related symptoms,” according to their study, reported in the journal CHEST®.
Asked whether he still recommends CPAP to patients with OSA and the metabolic syndrome, given the findings, corresponding author Luciano F. Drager, MD, PhD, replied “yes, definitely.”
“Despite the modest rate in reversing metabolic syndrome after CPAP, the rate was 5-fold higher than non-effective treatment (18% vs. 4%),” he said in an interview.
Dr. Drager noted that studies of other single interventions such as physical exercise to reverse MS in patients with OSA also had modest results.
A researcher who studies the relationship between sleep, circadian rhythms, and metabolism commented that, although the patients in the CPAP group were compliant with the assigned equipment and had both reductions in apneic events and improvement in oxygen saturation, the effect of CPAP on the metabolic syndrome was rather small.
“The CPAP was doing what we thought it was supposed to do, but it didn’t have the magnitude of effect on the metabolic syndrome as I expected or I think as the authors expected,” said Deanna Arble, PhD, assistant professor of biological science at Marquette University, Milwaukee.
She noted that the study also failed to detect a significant improvement in the blood pressure component of metabolic syndrome.
“In my experience and my review of the literature, blood pressure tends to be the one that’s improved most dramatically with CPAP,” she said.
Dr. Arble was not involved in the study.
Study details
In the trial, titled TREATOSA-MS, the investigators enrolled 100 patients with a recent diagnosis of metabolic syndrome and moderate to severe OSA, defined as 15 or more apnea-hypopnea index events per hour. The patients were stratified by body mass index and then randomized to undergo therapeutic CPAP or to use nasal strips for 6 months.
At baseline and at the end of each intervention investigators measured anthropometric variables, blood pressure, glucose, and lipid profiles. They also leptin and adiponectin, body composition, food intake, physical activity, subcutaneous and abdominal fat (visceral and hepatic), and endothelial function to control for potential confounders.
As noted previously, they found that after 6 months “most patients with OSA randomized to CPAP retained the MS diagnosis, but the rate of MS reversibility was higher than observed in the placebo group.” The difference in metabolic syndrome reversal, 18% with CPAP versus 4% with nasal strips, translated into a hazard ratio favoring CPAP of 5.27 (P = .04).
Also as noted, in analyses adjusted for baseline values, CPAP did not significantly improve either weight, liver fat, lip profiles, or the adiposity biomarkers leptin and adiponectin, but did have “very modest” influence on reducing visceral fat and improving endothelial function.
Rigorous study
Dr. Arble said that most studies of the association between OSA and metabolic syndrome have focused on only one or two of the parameters that were included in the TREATOSA-MS study, giving the findings additional weight.
“This could potentially be a very good, carefully controlled first insight into how obstructive sleep apnea is related to the metabolic syndrome,” she said.
The study was funded by grants Fundação de Amparo Q22 à Pesquisa do Estado de São Paulo and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior. The authors and Dr. Arble reported having no conflicts of interest to disclose.
Continuous positive airway pressure (CPAP) may be only modestly effective for ameliorating metabolic syndrome in patients with moderate to severe obstructive sleep apnea (OSA).
That conclusion comes from investigators in a randomized controlled, trial, who found that, among 100 patients with OSA and a recent diagnosis of metabolic syndrome (MS), 18% of those assigned to use CPAP at night had a reversal of MS at 6 months of follow-up, compared with 4% of controls who were assigned to use nasal strips at night (P = .04).
The majority of patients assigned to CPAP still retained their MS diagnoses at 6 months, and CPAP did not significantly reduce individual components of the syndrome. Use of CPAP was, however, associated with small reductions in visceral fat and improvement in endothelial function, reported Sara Q.C. Giampa, PhD, from the University of São Paulo, and colleagues.
“Despite a significant rate of MS reversibility after CPAP therapy, most of the patients maintained the MS diagnosis. The modest effects of CPAP on MS reversibility underscore the need for combined therapy with CPAP, aiming to maximize metabolic syndrome recovery in parallel with improvements in OSA severity and related symptoms,” according to their study, reported in the journal CHEST®.
Asked whether he still recommends CPAP to patients with OSA and the metabolic syndrome, given the findings, corresponding author Luciano F. Drager, MD, PhD, replied “yes, definitely.”
“Despite the modest rate in reversing metabolic syndrome after CPAP, the rate was 5-fold higher than non-effective treatment (18% vs. 4%),” he said in an interview.
Dr. Drager noted that studies of other single interventions such as physical exercise to reverse MS in patients with OSA also had modest results.
A researcher who studies the relationship between sleep, circadian rhythms, and metabolism commented that, although the patients in the CPAP group were compliant with the assigned equipment and had both reductions in apneic events and improvement in oxygen saturation, the effect of CPAP on the metabolic syndrome was rather small.
“The CPAP was doing what we thought it was supposed to do, but it didn’t have the magnitude of effect on the metabolic syndrome as I expected or I think as the authors expected,” said Deanna Arble, PhD, assistant professor of biological science at Marquette University, Milwaukee.
She noted that the study also failed to detect a significant improvement in the blood pressure component of metabolic syndrome.
“In my experience and my review of the literature, blood pressure tends to be the one that’s improved most dramatically with CPAP,” she said.
Dr. Arble was not involved in the study.
Study details
In the trial, titled TREATOSA-MS, the investigators enrolled 100 patients with a recent diagnosis of metabolic syndrome and moderate to severe OSA, defined as 15 or more apnea-hypopnea index events per hour. The patients were stratified by body mass index and then randomized to undergo therapeutic CPAP or to use nasal strips for 6 months.
At baseline and at the end of each intervention investigators measured anthropometric variables, blood pressure, glucose, and lipid profiles. They also leptin and adiponectin, body composition, food intake, physical activity, subcutaneous and abdominal fat (visceral and hepatic), and endothelial function to control for potential confounders.
As noted previously, they found that after 6 months “most patients with OSA randomized to CPAP retained the MS diagnosis, but the rate of MS reversibility was higher than observed in the placebo group.” The difference in metabolic syndrome reversal, 18% with CPAP versus 4% with nasal strips, translated into a hazard ratio favoring CPAP of 5.27 (P = .04).
Also as noted, in analyses adjusted for baseline values, CPAP did not significantly improve either weight, liver fat, lip profiles, or the adiposity biomarkers leptin and adiponectin, but did have “very modest” influence on reducing visceral fat and improving endothelial function.
Rigorous study
Dr. Arble said that most studies of the association between OSA and metabolic syndrome have focused on only one or two of the parameters that were included in the TREATOSA-MS study, giving the findings additional weight.
“This could potentially be a very good, carefully controlled first insight into how obstructive sleep apnea is related to the metabolic syndrome,” she said.
The study was funded by grants Fundação de Amparo Q22 à Pesquisa do Estado de São Paulo and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior. The authors and Dr. Arble reported having no conflicts of interest to disclose.
FROM CHEST
Lights on during sleep can play havoc with metabolism
“The most important finding” is that, compared with one night in a dim light environment, “one night of exposure to a moderate level of room light while sleeping with eyes closed increased heart rate and sympathetic [nervous system] activity during the entire sleep period,” said senior author Phyllis C. Zee, MD, PhD.
And on the morning following the moderate room light condition, a higher amount of insulin secretion was required to normalize glucose levels following ingestion of a bolus of glucose in an oral glucose tolerance test, consistent with higher insulin resistance, Dr. Zee, director of the center for circadian and sleep medicine at Northwestern University, Chicago, told this news organization in an email.
The study by Ivy C. Mason, PhD, also of Northwestern University, and colleagues was published March 14 in the Proceedings of the National Academy of Sciences.
Melatonin levels were similar under the two light conditions, Dr. Zee added, which “suggests that the effect of light during sleep on these cardiometabolic measures were more likely due to activation of the sympathetic [nervous] system and less likely due to changes in sleep or suppression of melatonin by light.”
“Attention to avoiding exposure to light at night during sleep may be beneficial for cardiometabolic health,” the researchers conclude.
That means “turn lights off before sleeping,” Dr. Zee elaborated. If a light is needed for safety reasons, keep it as dim as possible, she advises, and avoid exposure to blue or green light, but instead try red-amber colors.
How light during sleep may affect insulin, melatonin, heart rate
Several studies have investigated the effect of light on sleep and metabolic outcomes, the researchers explain.
In one study, light in the bedroom was associated with obesity in women, and in another study, it was associated with risk of type 2 diabetes in an elderly population.
Research has suggested that nighttime light exposure may alter glucose metabolism by increasing insulin resistance; lowering melatonin levels, which alters insulin secretion; and having an arousing effect on the sympathetic autonomic nervous system (increasing the stress hormone cortisol or heart rate, and decreasing heart rate variability).
However, the effect of a single night of moderate room light exposure across the entire nighttime sleep period has not been fully investigated.
The researchers enrolled and randomized 20 healthy young adults who were 18-40 years old and regularly went to sleep between 9 p.m. and 1 a.m. and slept 6.5-8.5 hours, to sleep 2 nights in the sleep laboratory under two conditions.
Ten participants (eight women, two men) slept in a dim light condition on night 1 and in a moderate light condition on night 2. The other 10 participants (six women, four men) slept 2 nights in the dim light condition.
The moderate light condition consisted of four 60-watt incandescent overhead ceiling light bulbs (a total of 100 lux), which “is bright enough to see, but not to read comfortably,” Dr. Zee explained. “It’s like hallway light in an apartment. But the people were sleeping, so about 90% of the light would be blocked by the eyelids.”
The dim light condition was less than 3 lux, which is dimmer than a night light.
When participants were awake, the room lighting was 240 lux.
Participants in each group were a mean age of 27 years and had a mean body mass index of 23 and 24 kg/m2.
The week before the study, participants went to bed at 11 p.m. and slept for 7 hours (based on actigraphy measures). During the laboratory stay, the participants were allowed to sleep 8 hours, during which polysomnography was performed.
They received standard meals at 2.5, 5, and 11 hours after waking and had 30 minutes to eat them. Snacking and caffeine were not permitted.
Participants were instructed to remain seated or standing in their room, but not exercise, when they were not sleeping. Blood samples to determine melatonin levels were collected hourly during wake and sleep via an intravenous line.
Participants slept for a similar time, around 7 hours, in both conditions.
Although melatonin levels were similar in both conditions, this was a relatively small sample, the researchers caution.
In the room light condition, participants spent proportionately more time in stage N2 sleep and less in slow-wave and rapid eye movement sleep. There was no increase in sleep fragmentation or arousals.
The research was partly supported by the Center for Circadian and Sleep Medicine at Northwestern University, the National Center for Advancing Translational Sciences, the National Institutes of Health, and the American Heart Association. The researchers have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“The most important finding” is that, compared with one night in a dim light environment, “one night of exposure to a moderate level of room light while sleeping with eyes closed increased heart rate and sympathetic [nervous system] activity during the entire sleep period,” said senior author Phyllis C. Zee, MD, PhD.
And on the morning following the moderate room light condition, a higher amount of insulin secretion was required to normalize glucose levels following ingestion of a bolus of glucose in an oral glucose tolerance test, consistent with higher insulin resistance, Dr. Zee, director of the center for circadian and sleep medicine at Northwestern University, Chicago, told this news organization in an email.
The study by Ivy C. Mason, PhD, also of Northwestern University, and colleagues was published March 14 in the Proceedings of the National Academy of Sciences.
Melatonin levels were similar under the two light conditions, Dr. Zee added, which “suggests that the effect of light during sleep on these cardiometabolic measures were more likely due to activation of the sympathetic [nervous] system and less likely due to changes in sleep or suppression of melatonin by light.”
“Attention to avoiding exposure to light at night during sleep may be beneficial for cardiometabolic health,” the researchers conclude.
That means “turn lights off before sleeping,” Dr. Zee elaborated. If a light is needed for safety reasons, keep it as dim as possible, she advises, and avoid exposure to blue or green light, but instead try red-amber colors.
How light during sleep may affect insulin, melatonin, heart rate
Several studies have investigated the effect of light on sleep and metabolic outcomes, the researchers explain.
In one study, light in the bedroom was associated with obesity in women, and in another study, it was associated with risk of type 2 diabetes in an elderly population.
Research has suggested that nighttime light exposure may alter glucose metabolism by increasing insulin resistance; lowering melatonin levels, which alters insulin secretion; and having an arousing effect on the sympathetic autonomic nervous system (increasing the stress hormone cortisol or heart rate, and decreasing heart rate variability).
However, the effect of a single night of moderate room light exposure across the entire nighttime sleep period has not been fully investigated.
The researchers enrolled and randomized 20 healthy young adults who were 18-40 years old and regularly went to sleep between 9 p.m. and 1 a.m. and slept 6.5-8.5 hours, to sleep 2 nights in the sleep laboratory under two conditions.
Ten participants (eight women, two men) slept in a dim light condition on night 1 and in a moderate light condition on night 2. The other 10 participants (six women, four men) slept 2 nights in the dim light condition.
The moderate light condition consisted of four 60-watt incandescent overhead ceiling light bulbs (a total of 100 lux), which “is bright enough to see, but not to read comfortably,” Dr. Zee explained. “It’s like hallway light in an apartment. But the people were sleeping, so about 90% of the light would be blocked by the eyelids.”
The dim light condition was less than 3 lux, which is dimmer than a night light.
When participants were awake, the room lighting was 240 lux.
Participants in each group were a mean age of 27 years and had a mean body mass index of 23 and 24 kg/m2.
The week before the study, participants went to bed at 11 p.m. and slept for 7 hours (based on actigraphy measures). During the laboratory stay, the participants were allowed to sleep 8 hours, during which polysomnography was performed.
They received standard meals at 2.5, 5, and 11 hours after waking and had 30 minutes to eat them. Snacking and caffeine were not permitted.
Participants were instructed to remain seated or standing in their room, but not exercise, when they were not sleeping. Blood samples to determine melatonin levels were collected hourly during wake and sleep via an intravenous line.
Participants slept for a similar time, around 7 hours, in both conditions.
Although melatonin levels were similar in both conditions, this was a relatively small sample, the researchers caution.
In the room light condition, participants spent proportionately more time in stage N2 sleep and less in slow-wave and rapid eye movement sleep. There was no increase in sleep fragmentation or arousals.
The research was partly supported by the Center for Circadian and Sleep Medicine at Northwestern University, the National Center for Advancing Translational Sciences, the National Institutes of Health, and the American Heart Association. The researchers have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“The most important finding” is that, compared with one night in a dim light environment, “one night of exposure to a moderate level of room light while sleeping with eyes closed increased heart rate and sympathetic [nervous system] activity during the entire sleep period,” said senior author Phyllis C. Zee, MD, PhD.
And on the morning following the moderate room light condition, a higher amount of insulin secretion was required to normalize glucose levels following ingestion of a bolus of glucose in an oral glucose tolerance test, consistent with higher insulin resistance, Dr. Zee, director of the center for circadian and sleep medicine at Northwestern University, Chicago, told this news organization in an email.
The study by Ivy C. Mason, PhD, also of Northwestern University, and colleagues was published March 14 in the Proceedings of the National Academy of Sciences.
Melatonin levels were similar under the two light conditions, Dr. Zee added, which “suggests that the effect of light during sleep on these cardiometabolic measures were more likely due to activation of the sympathetic [nervous] system and less likely due to changes in sleep or suppression of melatonin by light.”
“Attention to avoiding exposure to light at night during sleep may be beneficial for cardiometabolic health,” the researchers conclude.
That means “turn lights off before sleeping,” Dr. Zee elaborated. If a light is needed for safety reasons, keep it as dim as possible, she advises, and avoid exposure to blue or green light, but instead try red-amber colors.
How light during sleep may affect insulin, melatonin, heart rate
Several studies have investigated the effect of light on sleep and metabolic outcomes, the researchers explain.
In one study, light in the bedroom was associated with obesity in women, and in another study, it was associated with risk of type 2 diabetes in an elderly population.
Research has suggested that nighttime light exposure may alter glucose metabolism by increasing insulin resistance; lowering melatonin levels, which alters insulin secretion; and having an arousing effect on the sympathetic autonomic nervous system (increasing the stress hormone cortisol or heart rate, and decreasing heart rate variability).
However, the effect of a single night of moderate room light exposure across the entire nighttime sleep period has not been fully investigated.
The researchers enrolled and randomized 20 healthy young adults who were 18-40 years old and regularly went to sleep between 9 p.m. and 1 a.m. and slept 6.5-8.5 hours, to sleep 2 nights in the sleep laboratory under two conditions.
Ten participants (eight women, two men) slept in a dim light condition on night 1 and in a moderate light condition on night 2. The other 10 participants (six women, four men) slept 2 nights in the dim light condition.
The moderate light condition consisted of four 60-watt incandescent overhead ceiling light bulbs (a total of 100 lux), which “is bright enough to see, but not to read comfortably,” Dr. Zee explained. “It’s like hallway light in an apartment. But the people were sleeping, so about 90% of the light would be blocked by the eyelids.”
The dim light condition was less than 3 lux, which is dimmer than a night light.
When participants were awake, the room lighting was 240 lux.
Participants in each group were a mean age of 27 years and had a mean body mass index of 23 and 24 kg/m2.
The week before the study, participants went to bed at 11 p.m. and slept for 7 hours (based on actigraphy measures). During the laboratory stay, the participants were allowed to sleep 8 hours, during which polysomnography was performed.
They received standard meals at 2.5, 5, and 11 hours after waking and had 30 minutes to eat them. Snacking and caffeine were not permitted.
Participants were instructed to remain seated or standing in their room, but not exercise, when they were not sleeping. Blood samples to determine melatonin levels were collected hourly during wake and sleep via an intravenous line.
Participants slept for a similar time, around 7 hours, in both conditions.
Although melatonin levels were similar in both conditions, this was a relatively small sample, the researchers caution.
In the room light condition, participants spent proportionately more time in stage N2 sleep and less in slow-wave and rapid eye movement sleep. There was no increase in sleep fragmentation or arousals.
The research was partly supported by the Center for Circadian and Sleep Medicine at Northwestern University, the National Center for Advancing Translational Sciences, the National Institutes of Health, and the American Heart Association. The researchers have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
Targeting the endocannabinoid system in migraine
, according to Italian researchers at the University of Pavia, and the C. Mondino National Institute of Neurology Foundation. “The complexity of the endocannabinoid system calls for accurate biochemical and pharmacological characterization of any new compounds undergoing testing and development,” noted Rosaria Greco, PhD. She and her colleagues authored a review on the topic that was published online Feb. 18, 2022, in Headache.
Although cannabis has been investigated for both the treatment and prevention of migraine, evidence for its benefit is weak because of lack of controlled studies, they explained. Archival data from a large database “showed greater improvements in men than in women and suggested that concentrated preparations were more effective than flower consumption.” In addition, a small single-center study linked nabilone, a synthetic cannabinoid, to reductions in pain duration, intensity, and daily intake of analgesics among patients with medication overuse headache. Finally, a pilot study reported a reduction in pain intensity among patients with chronic migraine treated with a combination of tested a combination of delta-9-tetrahydrocannabinol and cannabidiol. “Methodologically sound studies are now needed to investigate the possible effects of cannabis in migraine treatment and to define strains, formulations, and dosage,” they noted.
Not just cannabis
In addition to exogenous cannabis, there are now preclinical studies suggesting other compounds that interact with the endocannabinoid system “are also able to modulate the pathways involved in migraine-related pain,” the study authors wrote. “But the road ahead is still long. Multiple molecules linked to the endocannabinoid system have emerged as potential therapeutic targets.
The complexity of the system demands caution and precise biochemical and pharmacological characterization of the new compounds to be tested and developed.”
Among these compounds are endogenous ligands such as N-arachidonoylethanolamine (anandamide) and 2-arachidonoylglycerol that specifically target CB1 and CB2 receptors. Additionally, there are endocannabinoid-based drugs that also target the CB1/CB2 receptors, as well as other substances, such as lipids (palmitoylethanolamide [PEA]) and enzymes, that do not bind to the CB1/CB2 receptors but are responsible for endocannabinoid biosynthesis.
There is some evidence that the endocannabinoid system may be dysfunctional in patients with migraine, and the authors noted their work has shown that PEA plasma levels are increased during experimentally triggered migraine-like attacks. Thus, some preclinical and preliminary evidence suggests that administration of PEA or anandamide may have analgesic and anti-inflammatory effects in migraine.
Another approach is the inhibition of endocannabinoid catabolic enzymes, which could circumvent the adverse effects associated with direct activation of CB receptors. “Endocannabinoid tone enhancement has been proposed as an alternative modality of activation of CB receptors and is possibly devoid of the psychotropic effects reported with CB receptor agonists,” noted the authors, who have shown in animal and preclinical studies that inhibition of fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase can modulate migraine pain.
Yet another way of indirectly impacting CB receptors is through their allosteric ligands, an approach that “deserves further investigation”, and “might provide interesting leads for clinical development, given that it may have a favorable side-effect profile with limited psychomimetic and depressant effects,” wrote the authors. And finally, inhibition of N-acylethanolamine acid amide hydrolase, the enzyme that preferentially hydrolyzes PEA, might be a promising approach.
“The multiplicity of options and the wealth of data already obtained in animal models underscore the importance of further advancing research in this area,” the authors concluded.
Patients are taking cannabinoids; physicians should learn about them
Commenting on the paper, Alan Rapaport, MD, clinical professor of neurology at the University of California, Los Angeles, said “this well-done paper points out the complexity of the endocannabinoid system and the multiple ways of getting it to work for certain patients. It details some of the studies that show beneficial results in migraine, medication overuse headache, chronic migraine, and pain. Patients with headache, other types of pain, anxiety, nausea, sleep issues, and other symptoms are already taking cannabinoids, usually derived from the marijuana plant, that are not well regulated. A few are prescribed drugs which target CB1 and CB2 receptors. Patients often get relief of some of their symptoms, sometimes getting high and many times not.
“The paper makes the point that previous studies are often small, not carefully controlled, or well documented. We do need to start doing larger, properly designed studies and getting them into the literature. Doctors need to learn more about these treatments. The next step will be to get [Food and Drug Administration]–approved treatments, so physicians and nurses will know exactly what we are giving, the beneficial effects to expect in a certain percentage of patients, and the adverse events to warn our patients about. Cannabinoids have been tried by a large percentage of patients with headache and pain. Now we need to standardize the various treatments that are sure to be suggested in the future.”
The study was funded by the Migraine Research Foundation, and the Italian Ministry of Health. The study authors declared no conflicts of interest.
, according to Italian researchers at the University of Pavia, and the C. Mondino National Institute of Neurology Foundation. “The complexity of the endocannabinoid system calls for accurate biochemical and pharmacological characterization of any new compounds undergoing testing and development,” noted Rosaria Greco, PhD. She and her colleagues authored a review on the topic that was published online Feb. 18, 2022, in Headache.
Although cannabis has been investigated for both the treatment and prevention of migraine, evidence for its benefit is weak because of lack of controlled studies, they explained. Archival data from a large database “showed greater improvements in men than in women and suggested that concentrated preparations were more effective than flower consumption.” In addition, a small single-center study linked nabilone, a synthetic cannabinoid, to reductions in pain duration, intensity, and daily intake of analgesics among patients with medication overuse headache. Finally, a pilot study reported a reduction in pain intensity among patients with chronic migraine treated with a combination of tested a combination of delta-9-tetrahydrocannabinol and cannabidiol. “Methodologically sound studies are now needed to investigate the possible effects of cannabis in migraine treatment and to define strains, formulations, and dosage,” they noted.
Not just cannabis
In addition to exogenous cannabis, there are now preclinical studies suggesting other compounds that interact with the endocannabinoid system “are also able to modulate the pathways involved in migraine-related pain,” the study authors wrote. “But the road ahead is still long. Multiple molecules linked to the endocannabinoid system have emerged as potential therapeutic targets.
The complexity of the system demands caution and precise biochemical and pharmacological characterization of the new compounds to be tested and developed.”
Among these compounds are endogenous ligands such as N-arachidonoylethanolamine (anandamide) and 2-arachidonoylglycerol that specifically target CB1 and CB2 receptors. Additionally, there are endocannabinoid-based drugs that also target the CB1/CB2 receptors, as well as other substances, such as lipids (palmitoylethanolamide [PEA]) and enzymes, that do not bind to the CB1/CB2 receptors but are responsible for endocannabinoid biosynthesis.
There is some evidence that the endocannabinoid system may be dysfunctional in patients with migraine, and the authors noted their work has shown that PEA plasma levels are increased during experimentally triggered migraine-like attacks. Thus, some preclinical and preliminary evidence suggests that administration of PEA or anandamide may have analgesic and anti-inflammatory effects in migraine.
Another approach is the inhibition of endocannabinoid catabolic enzymes, which could circumvent the adverse effects associated with direct activation of CB receptors. “Endocannabinoid tone enhancement has been proposed as an alternative modality of activation of CB receptors and is possibly devoid of the psychotropic effects reported with CB receptor agonists,” noted the authors, who have shown in animal and preclinical studies that inhibition of fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase can modulate migraine pain.
Yet another way of indirectly impacting CB receptors is through their allosteric ligands, an approach that “deserves further investigation”, and “might provide interesting leads for clinical development, given that it may have a favorable side-effect profile with limited psychomimetic and depressant effects,” wrote the authors. And finally, inhibition of N-acylethanolamine acid amide hydrolase, the enzyme that preferentially hydrolyzes PEA, might be a promising approach.
“The multiplicity of options and the wealth of data already obtained in animal models underscore the importance of further advancing research in this area,” the authors concluded.
Patients are taking cannabinoids; physicians should learn about them
Commenting on the paper, Alan Rapaport, MD, clinical professor of neurology at the University of California, Los Angeles, said “this well-done paper points out the complexity of the endocannabinoid system and the multiple ways of getting it to work for certain patients. It details some of the studies that show beneficial results in migraine, medication overuse headache, chronic migraine, and pain. Patients with headache, other types of pain, anxiety, nausea, sleep issues, and other symptoms are already taking cannabinoids, usually derived from the marijuana plant, that are not well regulated. A few are prescribed drugs which target CB1 and CB2 receptors. Patients often get relief of some of their symptoms, sometimes getting high and many times not.
“The paper makes the point that previous studies are often small, not carefully controlled, or well documented. We do need to start doing larger, properly designed studies and getting them into the literature. Doctors need to learn more about these treatments. The next step will be to get [Food and Drug Administration]–approved treatments, so physicians and nurses will know exactly what we are giving, the beneficial effects to expect in a certain percentage of patients, and the adverse events to warn our patients about. Cannabinoids have been tried by a large percentage of patients with headache and pain. Now we need to standardize the various treatments that are sure to be suggested in the future.”
The study was funded by the Migraine Research Foundation, and the Italian Ministry of Health. The study authors declared no conflicts of interest.
, according to Italian researchers at the University of Pavia, and the C. Mondino National Institute of Neurology Foundation. “The complexity of the endocannabinoid system calls for accurate biochemical and pharmacological characterization of any new compounds undergoing testing and development,” noted Rosaria Greco, PhD. She and her colleagues authored a review on the topic that was published online Feb. 18, 2022, in Headache.
Although cannabis has been investigated for both the treatment and prevention of migraine, evidence for its benefit is weak because of lack of controlled studies, they explained. Archival data from a large database “showed greater improvements in men than in women and suggested that concentrated preparations were more effective than flower consumption.” In addition, a small single-center study linked nabilone, a synthetic cannabinoid, to reductions in pain duration, intensity, and daily intake of analgesics among patients with medication overuse headache. Finally, a pilot study reported a reduction in pain intensity among patients with chronic migraine treated with a combination of tested a combination of delta-9-tetrahydrocannabinol and cannabidiol. “Methodologically sound studies are now needed to investigate the possible effects of cannabis in migraine treatment and to define strains, formulations, and dosage,” they noted.
Not just cannabis
In addition to exogenous cannabis, there are now preclinical studies suggesting other compounds that interact with the endocannabinoid system “are also able to modulate the pathways involved in migraine-related pain,” the study authors wrote. “But the road ahead is still long. Multiple molecules linked to the endocannabinoid system have emerged as potential therapeutic targets.
The complexity of the system demands caution and precise biochemical and pharmacological characterization of the new compounds to be tested and developed.”
Among these compounds are endogenous ligands such as N-arachidonoylethanolamine (anandamide) and 2-arachidonoylglycerol that specifically target CB1 and CB2 receptors. Additionally, there are endocannabinoid-based drugs that also target the CB1/CB2 receptors, as well as other substances, such as lipids (palmitoylethanolamide [PEA]) and enzymes, that do not bind to the CB1/CB2 receptors but are responsible for endocannabinoid biosynthesis.
There is some evidence that the endocannabinoid system may be dysfunctional in patients with migraine, and the authors noted their work has shown that PEA plasma levels are increased during experimentally triggered migraine-like attacks. Thus, some preclinical and preliminary evidence suggests that administration of PEA or anandamide may have analgesic and anti-inflammatory effects in migraine.
Another approach is the inhibition of endocannabinoid catabolic enzymes, which could circumvent the adverse effects associated with direct activation of CB receptors. “Endocannabinoid tone enhancement has been proposed as an alternative modality of activation of CB receptors and is possibly devoid of the psychotropic effects reported with CB receptor agonists,” noted the authors, who have shown in animal and preclinical studies that inhibition of fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase can modulate migraine pain.
Yet another way of indirectly impacting CB receptors is through their allosteric ligands, an approach that “deserves further investigation”, and “might provide interesting leads for clinical development, given that it may have a favorable side-effect profile with limited psychomimetic and depressant effects,” wrote the authors. And finally, inhibition of N-acylethanolamine acid amide hydrolase, the enzyme that preferentially hydrolyzes PEA, might be a promising approach.
“The multiplicity of options and the wealth of data already obtained in animal models underscore the importance of further advancing research in this area,” the authors concluded.
Patients are taking cannabinoids; physicians should learn about them
Commenting on the paper, Alan Rapaport, MD, clinical professor of neurology at the University of California, Los Angeles, said “this well-done paper points out the complexity of the endocannabinoid system and the multiple ways of getting it to work for certain patients. It details some of the studies that show beneficial results in migraine, medication overuse headache, chronic migraine, and pain. Patients with headache, other types of pain, anxiety, nausea, sleep issues, and other symptoms are already taking cannabinoids, usually derived from the marijuana plant, that are not well regulated. A few are prescribed drugs which target CB1 and CB2 receptors. Patients often get relief of some of their symptoms, sometimes getting high and many times not.
“The paper makes the point that previous studies are often small, not carefully controlled, or well documented. We do need to start doing larger, properly designed studies and getting them into the literature. Doctors need to learn more about these treatments. The next step will be to get [Food and Drug Administration]–approved treatments, so physicians and nurses will know exactly what we are giving, the beneficial effects to expect in a certain percentage of patients, and the adverse events to warn our patients about. Cannabinoids have been tried by a large percentage of patients with headache and pain. Now we need to standardize the various treatments that are sure to be suggested in the future.”
The study was funded by the Migraine Research Foundation, and the Italian Ministry of Health. The study authors declared no conflicts of interest.
FROM HEADACHE
Real-world data support safety of newer LAA device
More than 18 months after the Watchman FLX device was licensed by the Food and Drug Administration for closure of the left atrial appendage (LAA), a prospective analysis of registry data presented at CRT 2022, sponsored by MedStar Heart & Vascular Institute, supports its safely outside of the clinical trial setting.
The data, drawn from the LAA occlusion registry of the National Cardiovascular Data Registry, showed a mortality rate at 45 days of under 1.0%, which was consistent with the acceptably low rate of other adverse events, according to Samir R. Kapadia, MD, chair of cardiovascular medicine at the Cleveland Clinic.
Only 0.5% had a pericardial effusion within 45 days of LAA closure that required intervention. Of those without effusion, 95% had a leak of less than 3 mm and 82% had no leak at all, according to Dr. Kapadia.
Patients enrolled in this analysis, called SURPASS (Surveillance Post Approval Analysis Plan), had undergone left atrial closure with the device from August 2020 to September 2022. There were no exclusion criteria. Ultimately, 2 years of follow-up is planned.
With more than 16,000 patients enrolled, the data on 14,363 patients in this initial 45-day analysis represents “the largest number of Watchman FLX patients evaluated to date,” Dr. Kapadia reported.
Device implantation success 97.5%
The Watchman FLX, which is delivered to the left atrial appendage by a transcatheter approach, was deployed successfully in 97.5% of all 16,048 patients enrolled in the registry. In the 398 cases without successful deployment, the anatomy was not conducive in nearly 70%. Other reasons included failure to meet device-release criteria and change in patient condition.
The outcomes of interest at 45 days were ischemic strokes, systemic emboli, device-related thrombi, device embolization, and bleeding. The primary endpoints at 2 years will be strokes and thrombotic events.
For stroke, the incidence within 45 days was 0.39%. About 25% of the strokes were hemorrhagic and the remainder were ischemic. There was 1 systemic embolism (0.01%), 5 device embolizations (0.03%), and 30 device-related thrombotic events (0.24%). Major bleeding occurred in 508 patients (3.55%).
For context, Dr. Kapadia compared these results to those observed in the PINNACLE FLX trial, which was a nonrandomized but prospective study of the Watchman FLX published about 1 year ago. In PINNACLE FLX, the enrollment was open to patients indicated for oral anticoagulation but who had an appropriate rationale for seeking a nonpharmacological alternative.
Taken from different studies, the outcomes at 45 days should not be construed as a direct comparison, but the similarity of the results can be considered reassuring, according to Dr. Kapadia.
For the composite safety endpoint of all-cause death, ischemic stroke, systemic embolism, or implantation-related events requiring intervention, the rates in SURPASS (0.4%) and PINNACLE FLX (0.5%) were nearly identical. Device leak rates (82.0% vs. 82.8%), stroke rates (0.4% vs. 0.7%), and all-cause death rates (0.9% vs. 0.5%) were also similar.
The similarity of the SURPASS and PINNACLE FLX data provides another level of reassurance.
“The SURPASS registry confirms the safety of the Watchman Flex in the real-world experience when the device is being used by many different operators in a large patient population,” Dr. Kapadia said in an interview.
In “appropriately selected patients,” the SURPASS data confirm that the Watchman FLX device “provides a safe and effective treatment option,” he added.
Relative to the PINNACLE FLX study, which enrolled 400 patients, it is noteworthy that the median age in SURPASS was older (76 vs. 73.8 years), a potential disadvantage in demonstrating comparable safety. The proportion of non-White patients was similar (6.7% vs. 6.3%). SURPASS had a higher proportion of women (40% vs. 35.5%).
The SURPASS data are credible, according to Vivek Y. Reddy, MD, director of cardiac arrhythmia services, Mount Sinai Health System, New York.
“While there are certainly limitations to registry data, I do feel pretty confident that these procedural complication and success rates [in SURPASS] do indeed reflect reality,” said Dr. Reddy, who was a coauthor of the PINNACLE FLX trial. In general, the SURPASS data “mirror most of our clinical experiences in routine clinical practice.”
With these registry data backing up multiple clinical studies, Dr. Reddy concluded, “I do believe that it is fair to say that Watchman-FLX implantation is a quite safe procedure.”
Dr. Kapadia reported no potential conflicts of interest. Dr. Reddy reported a financial relationship with Boston Scientific.
More than 18 months after the Watchman FLX device was licensed by the Food and Drug Administration for closure of the left atrial appendage (LAA), a prospective analysis of registry data presented at CRT 2022, sponsored by MedStar Heart & Vascular Institute, supports its safely outside of the clinical trial setting.
The data, drawn from the LAA occlusion registry of the National Cardiovascular Data Registry, showed a mortality rate at 45 days of under 1.0%, which was consistent with the acceptably low rate of other adverse events, according to Samir R. Kapadia, MD, chair of cardiovascular medicine at the Cleveland Clinic.
Only 0.5% had a pericardial effusion within 45 days of LAA closure that required intervention. Of those without effusion, 95% had a leak of less than 3 mm and 82% had no leak at all, according to Dr. Kapadia.
Patients enrolled in this analysis, called SURPASS (Surveillance Post Approval Analysis Plan), had undergone left atrial closure with the device from August 2020 to September 2022. There were no exclusion criteria. Ultimately, 2 years of follow-up is planned.
With more than 16,000 patients enrolled, the data on 14,363 patients in this initial 45-day analysis represents “the largest number of Watchman FLX patients evaluated to date,” Dr. Kapadia reported.
Device implantation success 97.5%
The Watchman FLX, which is delivered to the left atrial appendage by a transcatheter approach, was deployed successfully in 97.5% of all 16,048 patients enrolled in the registry. In the 398 cases without successful deployment, the anatomy was not conducive in nearly 70%. Other reasons included failure to meet device-release criteria and change in patient condition.
The outcomes of interest at 45 days were ischemic strokes, systemic emboli, device-related thrombi, device embolization, and bleeding. The primary endpoints at 2 years will be strokes and thrombotic events.
For stroke, the incidence within 45 days was 0.39%. About 25% of the strokes were hemorrhagic and the remainder were ischemic. There was 1 systemic embolism (0.01%), 5 device embolizations (0.03%), and 30 device-related thrombotic events (0.24%). Major bleeding occurred in 508 patients (3.55%).
For context, Dr. Kapadia compared these results to those observed in the PINNACLE FLX trial, which was a nonrandomized but prospective study of the Watchman FLX published about 1 year ago. In PINNACLE FLX, the enrollment was open to patients indicated for oral anticoagulation but who had an appropriate rationale for seeking a nonpharmacological alternative.
Taken from different studies, the outcomes at 45 days should not be construed as a direct comparison, but the similarity of the results can be considered reassuring, according to Dr. Kapadia.
For the composite safety endpoint of all-cause death, ischemic stroke, systemic embolism, or implantation-related events requiring intervention, the rates in SURPASS (0.4%) and PINNACLE FLX (0.5%) were nearly identical. Device leak rates (82.0% vs. 82.8%), stroke rates (0.4% vs. 0.7%), and all-cause death rates (0.9% vs. 0.5%) were also similar.
The similarity of the SURPASS and PINNACLE FLX data provides another level of reassurance.
“The SURPASS registry confirms the safety of the Watchman Flex in the real-world experience when the device is being used by many different operators in a large patient population,” Dr. Kapadia said in an interview.
In “appropriately selected patients,” the SURPASS data confirm that the Watchman FLX device “provides a safe and effective treatment option,” he added.
Relative to the PINNACLE FLX study, which enrolled 400 patients, it is noteworthy that the median age in SURPASS was older (76 vs. 73.8 years), a potential disadvantage in demonstrating comparable safety. The proportion of non-White patients was similar (6.7% vs. 6.3%). SURPASS had a higher proportion of women (40% vs. 35.5%).
The SURPASS data are credible, according to Vivek Y. Reddy, MD, director of cardiac arrhythmia services, Mount Sinai Health System, New York.
“While there are certainly limitations to registry data, I do feel pretty confident that these procedural complication and success rates [in SURPASS] do indeed reflect reality,” said Dr. Reddy, who was a coauthor of the PINNACLE FLX trial. In general, the SURPASS data “mirror most of our clinical experiences in routine clinical practice.”
With these registry data backing up multiple clinical studies, Dr. Reddy concluded, “I do believe that it is fair to say that Watchman-FLX implantation is a quite safe procedure.”
Dr. Kapadia reported no potential conflicts of interest. Dr. Reddy reported a financial relationship with Boston Scientific.
More than 18 months after the Watchman FLX device was licensed by the Food and Drug Administration for closure of the left atrial appendage (LAA), a prospective analysis of registry data presented at CRT 2022, sponsored by MedStar Heart & Vascular Institute, supports its safely outside of the clinical trial setting.
The data, drawn from the LAA occlusion registry of the National Cardiovascular Data Registry, showed a mortality rate at 45 days of under 1.0%, which was consistent with the acceptably low rate of other adverse events, according to Samir R. Kapadia, MD, chair of cardiovascular medicine at the Cleveland Clinic.
Only 0.5% had a pericardial effusion within 45 days of LAA closure that required intervention. Of those without effusion, 95% had a leak of less than 3 mm and 82% had no leak at all, according to Dr. Kapadia.
Patients enrolled in this analysis, called SURPASS (Surveillance Post Approval Analysis Plan), had undergone left atrial closure with the device from August 2020 to September 2022. There were no exclusion criteria. Ultimately, 2 years of follow-up is planned.
With more than 16,000 patients enrolled, the data on 14,363 patients in this initial 45-day analysis represents “the largest number of Watchman FLX patients evaluated to date,” Dr. Kapadia reported.
Device implantation success 97.5%
The Watchman FLX, which is delivered to the left atrial appendage by a transcatheter approach, was deployed successfully in 97.5% of all 16,048 patients enrolled in the registry. In the 398 cases without successful deployment, the anatomy was not conducive in nearly 70%. Other reasons included failure to meet device-release criteria and change in patient condition.
The outcomes of interest at 45 days were ischemic strokes, systemic emboli, device-related thrombi, device embolization, and bleeding. The primary endpoints at 2 years will be strokes and thrombotic events.
For stroke, the incidence within 45 days was 0.39%. About 25% of the strokes were hemorrhagic and the remainder were ischemic. There was 1 systemic embolism (0.01%), 5 device embolizations (0.03%), and 30 device-related thrombotic events (0.24%). Major bleeding occurred in 508 patients (3.55%).
For context, Dr. Kapadia compared these results to those observed in the PINNACLE FLX trial, which was a nonrandomized but prospective study of the Watchman FLX published about 1 year ago. In PINNACLE FLX, the enrollment was open to patients indicated for oral anticoagulation but who had an appropriate rationale for seeking a nonpharmacological alternative.
Taken from different studies, the outcomes at 45 days should not be construed as a direct comparison, but the similarity of the results can be considered reassuring, according to Dr. Kapadia.
For the composite safety endpoint of all-cause death, ischemic stroke, systemic embolism, or implantation-related events requiring intervention, the rates in SURPASS (0.4%) and PINNACLE FLX (0.5%) were nearly identical. Device leak rates (82.0% vs. 82.8%), stroke rates (0.4% vs. 0.7%), and all-cause death rates (0.9% vs. 0.5%) were also similar.
The similarity of the SURPASS and PINNACLE FLX data provides another level of reassurance.
“The SURPASS registry confirms the safety of the Watchman Flex in the real-world experience when the device is being used by many different operators in a large patient population,” Dr. Kapadia said in an interview.
In “appropriately selected patients,” the SURPASS data confirm that the Watchman FLX device “provides a safe and effective treatment option,” he added.
Relative to the PINNACLE FLX study, which enrolled 400 patients, it is noteworthy that the median age in SURPASS was older (76 vs. 73.8 years), a potential disadvantage in demonstrating comparable safety. The proportion of non-White patients was similar (6.7% vs. 6.3%). SURPASS had a higher proportion of women (40% vs. 35.5%).
The SURPASS data are credible, according to Vivek Y. Reddy, MD, director of cardiac arrhythmia services, Mount Sinai Health System, New York.
“While there are certainly limitations to registry data, I do feel pretty confident that these procedural complication and success rates [in SURPASS] do indeed reflect reality,” said Dr. Reddy, who was a coauthor of the PINNACLE FLX trial. In general, the SURPASS data “mirror most of our clinical experiences in routine clinical practice.”
With these registry data backing up multiple clinical studies, Dr. Reddy concluded, “I do believe that it is fair to say that Watchman-FLX implantation is a quite safe procedure.”
Dr. Kapadia reported no potential conflicts of interest. Dr. Reddy reported a financial relationship with Boston Scientific.
FROM CRT 2022
Can green tea extract protect against colorectal adenomas?
Green tea extract (GTE) does not appear to protect against colorectal adenoma recurrence, according to a study from Germany.
Preclinical, epidemiologic, and small clinical studies have suggested that GTE and its major active component, epigallocatechin gallate (EGCG), have antineoplastic effects in the colon and rectum.
But the new study found no statistically significant difference in adenoma recurrence in people who took GTE, standardized to 150 mg EGCG, twice daily for 3 years, relative to those who took matching placebo.
However, there was a suggestion of possible benefit in men but not women, which requires further study, Thomas Seufferlein, MD, with Ulm University Hospital, Baden-Württemberg, Germany, and colleagues write.
Their study was published online in The American Journal of Gastroenterology.
Largest trial to date
The MIRACLE trial (Minimizing the Risk of Metachronous Adenomas of the Colorectum With Green Tea Extract) included 879 adults aged 50-80 years. Participants had undergone removal of one or more histologically confirmed colorectal adenomas within 6 months prior to recruitment during colonoscopy, and there were no remaining colorectal adenomas.
There were 432 patients in the GTE group and 447 in the placebo group. Baseline characteristics were well balanced between the groups, and overall adherence to the study protocol was good.
After 3 years, adenomas were detected in 55.7% of participants in the placebo group and in 51.1% of those in the GTE group in the modified intention-to-treat population. This absolute difference of 4.6% in favor of GTE was not statistically significant.
The per protocol analysis also did not show a significant effect of GTE on new adenoma formation in the whole study population.
However, a preplanned subgroup analysis revealed a significant difference in the adenoma recurrence rate in favor of GTE in men but not women.
In men, GTE intake was associated with a significant 12.4% relative and 7.5% absolute reduction of metachronous adenomas, they report.
This potential gender-specific difference in chemoprevention “warrants further investigations,” the study team writes.
The safety profile of GTE as taken in this trial was good, with only grade 1/2 elevations in liver enzymes in the GTE group, compared with the placebo group. However, because the follow-up period was limited to 3 years, the long-term safety of GTE cannot be determined.
The researchers write that, to their knowledge, this study is the largest randomized trial to date of the effect of GTE on adenoma recurrence in a colorectal cancer screening population consisting of White patients.
Caveats and cautionary notes
Reached for comment, David Johnson, MD, professor of medicine and chief of gastroenterology at the Eastern Virginia School of Medicine, Norfolk, noted that “although the study showed no significant differences, the time horizon to show benefit may be longer than the 3-year duration of the study.”
“There are also methodologic issues with the readjustment of the target sample size, which may have led to a type II error, related to underpowering of the sample size,” said Dr. Johnson, who wasn’t involved in the study.
The researchers write that the study initially generated “great interest” and that many centers applied to participate. However, “quite a few” centers did not meet their promised recruitment targets and had to be replaced. Therefore, the statistical analysis plan had to be modified, and the number of participants had to be reduced over the course of the trial, they note.
Dr. Johnson also cautioned that while green tea is a popular drink, “there is strong evidence that green tea extract, found in many herbal and dietary supplements, is among the leading causes listed for drug-induced liver injury, including acute liver failure, urgent liver transplantation, and death.”
The study was fully funded by a grant from German Cancer Aid. The investigators and Dr. Johnson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Green tea extract (GTE) does not appear to protect against colorectal adenoma recurrence, according to a study from Germany.
Preclinical, epidemiologic, and small clinical studies have suggested that GTE and its major active component, epigallocatechin gallate (EGCG), have antineoplastic effects in the colon and rectum.
But the new study found no statistically significant difference in adenoma recurrence in people who took GTE, standardized to 150 mg EGCG, twice daily for 3 years, relative to those who took matching placebo.
However, there was a suggestion of possible benefit in men but not women, which requires further study, Thomas Seufferlein, MD, with Ulm University Hospital, Baden-Württemberg, Germany, and colleagues write.
Their study was published online in The American Journal of Gastroenterology.
Largest trial to date
The MIRACLE trial (Minimizing the Risk of Metachronous Adenomas of the Colorectum With Green Tea Extract) included 879 adults aged 50-80 years. Participants had undergone removal of one or more histologically confirmed colorectal adenomas within 6 months prior to recruitment during colonoscopy, and there were no remaining colorectal adenomas.
There were 432 patients in the GTE group and 447 in the placebo group. Baseline characteristics were well balanced between the groups, and overall adherence to the study protocol was good.
After 3 years, adenomas were detected in 55.7% of participants in the placebo group and in 51.1% of those in the GTE group in the modified intention-to-treat population. This absolute difference of 4.6% in favor of GTE was not statistically significant.
The per protocol analysis also did not show a significant effect of GTE on new adenoma formation in the whole study population.
However, a preplanned subgroup analysis revealed a significant difference in the adenoma recurrence rate in favor of GTE in men but not women.
In men, GTE intake was associated with a significant 12.4% relative and 7.5% absolute reduction of metachronous adenomas, they report.
This potential gender-specific difference in chemoprevention “warrants further investigations,” the study team writes.
The safety profile of GTE as taken in this trial was good, with only grade 1/2 elevations in liver enzymes in the GTE group, compared with the placebo group. However, because the follow-up period was limited to 3 years, the long-term safety of GTE cannot be determined.
The researchers write that, to their knowledge, this study is the largest randomized trial to date of the effect of GTE on adenoma recurrence in a colorectal cancer screening population consisting of White patients.
Caveats and cautionary notes
Reached for comment, David Johnson, MD, professor of medicine and chief of gastroenterology at the Eastern Virginia School of Medicine, Norfolk, noted that “although the study showed no significant differences, the time horizon to show benefit may be longer than the 3-year duration of the study.”
“There are also methodologic issues with the readjustment of the target sample size, which may have led to a type II error, related to underpowering of the sample size,” said Dr. Johnson, who wasn’t involved in the study.
The researchers write that the study initially generated “great interest” and that many centers applied to participate. However, “quite a few” centers did not meet their promised recruitment targets and had to be replaced. Therefore, the statistical analysis plan had to be modified, and the number of participants had to be reduced over the course of the trial, they note.
Dr. Johnson also cautioned that while green tea is a popular drink, “there is strong evidence that green tea extract, found in many herbal and dietary supplements, is among the leading causes listed for drug-induced liver injury, including acute liver failure, urgent liver transplantation, and death.”
The study was fully funded by a grant from German Cancer Aid. The investigators and Dr. Johnson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Green tea extract (GTE) does not appear to protect against colorectal adenoma recurrence, according to a study from Germany.
Preclinical, epidemiologic, and small clinical studies have suggested that GTE and its major active component, epigallocatechin gallate (EGCG), have antineoplastic effects in the colon and rectum.
But the new study found no statistically significant difference in adenoma recurrence in people who took GTE, standardized to 150 mg EGCG, twice daily for 3 years, relative to those who took matching placebo.
However, there was a suggestion of possible benefit in men but not women, which requires further study, Thomas Seufferlein, MD, with Ulm University Hospital, Baden-Württemberg, Germany, and colleagues write.
Their study was published online in The American Journal of Gastroenterology.
Largest trial to date
The MIRACLE trial (Minimizing the Risk of Metachronous Adenomas of the Colorectum With Green Tea Extract) included 879 adults aged 50-80 years. Participants had undergone removal of one or more histologically confirmed colorectal adenomas within 6 months prior to recruitment during colonoscopy, and there were no remaining colorectal adenomas.
There were 432 patients in the GTE group and 447 in the placebo group. Baseline characteristics were well balanced between the groups, and overall adherence to the study protocol was good.
After 3 years, adenomas were detected in 55.7% of participants in the placebo group and in 51.1% of those in the GTE group in the modified intention-to-treat population. This absolute difference of 4.6% in favor of GTE was not statistically significant.
The per protocol analysis also did not show a significant effect of GTE on new adenoma formation in the whole study population.
However, a preplanned subgroup analysis revealed a significant difference in the adenoma recurrence rate in favor of GTE in men but not women.
In men, GTE intake was associated with a significant 12.4% relative and 7.5% absolute reduction of metachronous adenomas, they report.
This potential gender-specific difference in chemoprevention “warrants further investigations,” the study team writes.
The safety profile of GTE as taken in this trial was good, with only grade 1/2 elevations in liver enzymes in the GTE group, compared with the placebo group. However, because the follow-up period was limited to 3 years, the long-term safety of GTE cannot be determined.
The researchers write that, to their knowledge, this study is the largest randomized trial to date of the effect of GTE on adenoma recurrence in a colorectal cancer screening population consisting of White patients.
Caveats and cautionary notes
Reached for comment, David Johnson, MD, professor of medicine and chief of gastroenterology at the Eastern Virginia School of Medicine, Norfolk, noted that “although the study showed no significant differences, the time horizon to show benefit may be longer than the 3-year duration of the study.”
“There are also methodologic issues with the readjustment of the target sample size, which may have led to a type II error, related to underpowering of the sample size,” said Dr. Johnson, who wasn’t involved in the study.
The researchers write that the study initially generated “great interest” and that many centers applied to participate. However, “quite a few” centers did not meet their promised recruitment targets and had to be replaced. Therefore, the statistical analysis plan had to be modified, and the number of participants had to be reduced over the course of the trial, they note.
Dr. Johnson also cautioned that while green tea is a popular drink, “there is strong evidence that green tea extract, found in many herbal and dietary supplements, is among the leading causes listed for drug-induced liver injury, including acute liver failure, urgent liver transplantation, and death.”
The study was fully funded by a grant from German Cancer Aid. The investigators and Dr. Johnson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Digital monitors can relieve asthma burden by boosting medication adherence and inhaler technique
PHOENIX – Before considering oral steroids or biologic therapies, many people with difficult-to-control asthma can reduce symptoms by addressing medication adherence and inhaler technique – and digital monitoring devices can play a key role.
Often physicians “will approach a patient about a biologic if they’re not responding to standard therapy. But we need to sometimes go back to those basic building blocks, like, are you taking the standard therapy?” William C. Anderson, MD, codirector of the multidisciplinary asthma clinic at Children’s Hospital Colorado, Aurora, said in an interview.
At the annual meeting of the American Academy of Allergy, Asthma, and Immunology, he and others presented data highlighting the diagnostic and therapeutic potential of digital monitoring devices for difficult-to-control asthma, the theme of the 2022s meeting.
The Global Initiative for Asthma (GINA) defines asthma as “difficult to control” if it remains uncontrolled despite medium- or high-dose inhaled corticosteroids with a second controller or with maintenance oral steroids, or if the asthma requires high-dose treatment to curb symptoms and exacerbations. About 17% of adult asthma patients have difficult-to-control asthma, according to the 2021 GINA report.
However, correcting for inhaling technique and adherence cuts the 17% down to just 3.7%, Giselle Mosnaim, MD, an allergist at NorthShore University HealthSystem outside Chicago and AAAAI immediate past president, told attendees at a Feb. 25 session on digital technologies for asthma management.
The CRITIKAL study, which reviewed data from more than 5,000 asthma patients, “showed that, if you have critical errors in inhaler technique, this leads to worse asthma outcomes and increased asthma exacerbations,” Dr. Mosnaim said. Sadly, it also shows that, from 1975 to 2014, despite new devices and new technologies, “we still have poor inhaler technique.”
As for ways to measure adherence, physician judgments tend to be inaccurate, patient self-reporting has proved unreliable, and prescription refill data doesn’t indicate whether patients actually used the medications. “The ideal measure of adherence should be objective, accurate and unobtrusive to minimize impact on patient behavior and allow reliable data collection in real-world settings,” Dr. Mosnaim said. “So electronic medication monitors are the gold standard.”
Tracking adherence
A closing afternoon session featured three presentations on research tracking adherence and outcomes in difficult-to-treat asthma patients – two pediatric cohorts and one across all ages. All studies used the Propeller Health sensor, a Food and Drug Administration–cleared device that attaches to the patient’s inhaler and automatically collects information on where, when, and how often they use their medication. The sensor then sends that information to a data cloud accessible to the patient and their health care professional.
Dr. Anderson’s team scoured a nationwide Propeller Health database for 8,000 patients using the digital monitors with controller therapies for asthma or chronic obstructive pulmonary disease (COPD). The study explored whether adherence differed for once-daily versus twice-daily medications, and if adherence differed based on patient age (4-60+ years).
For both asthma and COPD patients, those on once-daily regimens had higher medication adherence, compared with those who were prescribed twice-daily therapies. Plus, a greater proportion of once-daily patients met the prespecified 80% adherence threshold.
Looking across ages, medication use in the youngest group (aged 4-11 years) looked comparable with 30-somethings, “probably because parents are the ones giving the drug,” Dr. Anderson said. Mirroring patterns from other studies, adherence levels dipped in adolescents and young adults, relative to other age subsets.
Since this population-level analysis didn’t include individualized data on exacerbations or asthma control, “we can’t relate this to outcomes,” Dr. Anderson noted. But he said the data correlating medication use with adherence suggest that once-daily formulations may be the better option.
In one of the two pediatric studies, Matt McCulloch, MD, an allergy and immunology fellow working with Dr. Anderson, and colleagues reviewed charts of 40 children who received care at the Colorado Children’s multidisciplinary asthma clinic between 2018 and 2021. Half of these patients used Propeller Health sensors with their daily inhaled controller; the other patients were matched for age, ethnicity, sex, medication level, and disease control and severity – but had no electronic monitoring device.
On the whole, children who used digital monitoring for 12 months did not fare much better than matched controls on lung function (judged by forced expiratory volume) or asthma control (measured by Asthma Control Test scores).
However, within the digital monitoring group, patients who stayed on the Propeller system for 12 months did have better asthma control, fewer exacerbations, and improved asthma severity scores (measured by the Composite Asthma Severity Index), compared with when they first began digital monitoring. These children had all received care at the clinic for a while before their families opted for the electronic sensor, so “the effect wouldn’t have just been from starting in the clinic,” Dr. McCulloch said in an interview.
The gains came despite waning medication adherence. Similar to other digital monitoring studies, use of daily controller therapies in this retrospective analysis began at 50%-80% but dropped considerably during the first 4-5 months before settling into the 20%-30% range by 1 year.
Rachelle Ramsey, PhD, a pediatric research psychologist at Cincinnati Children’s Hospital Medical Center, presented data from 20 children with difficult-to-treat asthma who received 8 weeks of a digital adherence intervention during a 12-month treatment period. They analyzed three subsets – each with interventions based on how well the patients were managing daily controller therapy at baseline.
One patient with high (>80%) baseline adherence just received digital monitoring. The seven patients who began the study with intermediate (50%-80%) adherence received digital monitoring plus prescriptive text messaging. And the 12 children with poorest (<50%) baseline adherence received digital monitoring and a telehealth session in which a behavioral health specialist helped them set goals and create strategies to overcome barriers – for example, keeping the inhaler near their toothbrush in order to pair medication use with a daily habit.
“Overall, we found that matching Propeller with a behavioral intervention really improved adherence,” Dr. Ramsey said in an interview. While patients were receiving the intervention, adherence averaged across all groups increased from 39% to 76%. However, once the intervention period ended, the group’s adherence regressed toward baseline (36%).
Although adherence did not associate with clinical gains in this small study, the use of digital monitoring to improve medication adherence has translated to better outcomes in other recent efforts.
Remote monitoring
In a quality improvement project in the United Kingdom, nurses asked difficult-to-control asthma patients if they understood how to use their corticosteroid/long-acting beta2-agonist (LABA) inhalers and if they were adhering to treatment guidelines.
Those who answered yes to these questions were invited to a 28-day study that involved swapping their steroid/LABA inhalers for a different controller/bronchodilator (fluticasone/salmeterol) with INCA (Inhaler Compliance Assessment), a device that not only tracks adherence but also uses acoustics to gauge inhaler technique.
Among the 23 patients who participated, many had better clinical outcomes after 28 days of INCA monitoring. As Dr. Mosnaim told attendees, “what was amazing is so many of the patients that had been these difficult-to-control asthmatics who would have gone on to oral steroids or perhaps a biologic – lo and behold, you put them on a digital inhaler, and what do you see?” In two-thirds of the patients, “you see FeNo [a test that measures airway inflammation by detecting nitric oxide in exhalations] goes down. You see spirometry improve. You see the asthma control questionnaire improve. You see blood eosinophils go down.”
And in a 2020 randomized trial, Dr. Mosnaim and colleagues recruited 100 adults with uncontrolled asthma who had prescriptions for a daily inhaled corticosteroid and a short-acting beta-agonist (SABA) inhaler. Participants received Propeller sensors for their steroid and SABA inhalers. After a 2-week run-in period to calculate baseline corticosteroid adherence and SABA use for all participants, half the participants were randomly assigned to the control group, which had the app and sensor in silent mode, merely to collect data on medication use – whereas the treatment group received reminders, alerts, and monthly phone calls from providers who gave feedback on adherence and technique.
After 3 months of digital monitoring, patients didn’t use their rescue medication quite as often – as judged by a rise in the percentage of SABA-free days, compared with when they began the study. But the change in SABA-free days relative to baseline was more pronounced in the treatment group (19%) than in the control group (6%).
As seen in the other digital monitoring studies, adherence to daily corticosteroids fell with time, but the drop was milder in treated participants (2%) versus the control group (17%). So in this study, digital monitoring plus mobile app reminders and clinician feedback “prevented against fall in adherence to inhaled steroids over time,” Dr. Mosnaim said.
These results are “very encouraging” and offer “proof of concept that this type of remote monitoring could work,” Thanai Pongdee, MD, an allergist-immunologist with the Mayo Clinic in Rochester, Minn., said in an interview. One limitation was that the study was too short to measure exacerbation rates. A yearlong analysis would be “really fascinating because you’d catch all the seasons of the year – all the pollen seasons, all these things that could exacerbate you. Some people’s asthma can be quite seasonal.”
More important, the clinical utility of digital sensors will depend on how physicians choose to use them. If the doctor puts out a “blanket recommendation for using it but doesn’t ask you about it or doesn’t use the data to inform your care, then I think people just lose engagement and lose excitement over it,” Dr. Ramsey said. But if the health care team “asks you about the data or looks at the data with you or shows you how valuable this can be to your care, then I think that changes things.”
Building these analyses and interactions into the clinic workflow isn’t trivial. “If you have this wealth of data coming in, how are you going to look at it? Are you going to have an individual person assigned to this role? How are you going to respond to alerts?” Dr. Anderson asked.
In addition, because some digital monitors issue alerts when a patient’s asthma is not well controlled, some providers worry about liability if “something bad were to happen if you had that data but didn’t act upon it,” he said. Yet he noted that remote data monitoring is already used routinely in other areas of medicine, such as managing diabetes and heart conditions, “and it’s not like people are getting dinged for that stuff.”
Another issue is cost. Insurance only covers digital monitors in select cases, but it’s a bit of a catch-22. Insurers “don’t want to cover it until they get the data, but you can’t get the data until insurance covers it,” said Dr. Anderson, who added that “this year we finally got CPT reimbursement codes for monitoring devices.”
On the whole, studies of digital medication monitors suggest that better outcomes require “a good partnership between the health care provider and the patient,” Dr. Pongdee said. “It wasn’t like you could just put these things on and expect them to help. You still need that personal relationship to get the optimal results. We can have all this technology, but you still can’t take the people out of it.”
Dr. Mosnaim reported receiving current research grant support from GlaxoSmithKline, Novartis, Sanofi-Regeneron, and Teva; and past research grant support from AstraZeneca, Alk-Abello, and Genentech. She is immediate past president of the AAAAI, and directs the board of directors for the American Board of Allergy and Immunology. Dr. Anderson has served as a consultant for Regeneron, GlaxoSmithKline, and AstraZeneca, and has received research support from Colorado Medicaid. Dr. McCulloch and Dr. Ramsey disclosed no relevant financial relationships. Dr. Pongdee serves as an at-large director on the American Academy of Allergy, Asthma and Immunology board of directors. He receives grant funding from GlaxoSmithKline, and the Mayo Clinic is a trial site for GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
PHOENIX – Before considering oral steroids or biologic therapies, many people with difficult-to-control asthma can reduce symptoms by addressing medication adherence and inhaler technique – and digital monitoring devices can play a key role.
Often physicians “will approach a patient about a biologic if they’re not responding to standard therapy. But we need to sometimes go back to those basic building blocks, like, are you taking the standard therapy?” William C. Anderson, MD, codirector of the multidisciplinary asthma clinic at Children’s Hospital Colorado, Aurora, said in an interview.
At the annual meeting of the American Academy of Allergy, Asthma, and Immunology, he and others presented data highlighting the diagnostic and therapeutic potential of digital monitoring devices for difficult-to-control asthma, the theme of the 2022s meeting.
The Global Initiative for Asthma (GINA) defines asthma as “difficult to control” if it remains uncontrolled despite medium- or high-dose inhaled corticosteroids with a second controller or with maintenance oral steroids, or if the asthma requires high-dose treatment to curb symptoms and exacerbations. About 17% of adult asthma patients have difficult-to-control asthma, according to the 2021 GINA report.
However, correcting for inhaling technique and adherence cuts the 17% down to just 3.7%, Giselle Mosnaim, MD, an allergist at NorthShore University HealthSystem outside Chicago and AAAAI immediate past president, told attendees at a Feb. 25 session on digital technologies for asthma management.
The CRITIKAL study, which reviewed data from more than 5,000 asthma patients, “showed that, if you have critical errors in inhaler technique, this leads to worse asthma outcomes and increased asthma exacerbations,” Dr. Mosnaim said. Sadly, it also shows that, from 1975 to 2014, despite new devices and new technologies, “we still have poor inhaler technique.”
As for ways to measure adherence, physician judgments tend to be inaccurate, patient self-reporting has proved unreliable, and prescription refill data doesn’t indicate whether patients actually used the medications. “The ideal measure of adherence should be objective, accurate and unobtrusive to minimize impact on patient behavior and allow reliable data collection in real-world settings,” Dr. Mosnaim said. “So electronic medication monitors are the gold standard.”
Tracking adherence
A closing afternoon session featured three presentations on research tracking adherence and outcomes in difficult-to-treat asthma patients – two pediatric cohorts and one across all ages. All studies used the Propeller Health sensor, a Food and Drug Administration–cleared device that attaches to the patient’s inhaler and automatically collects information on where, when, and how often they use their medication. The sensor then sends that information to a data cloud accessible to the patient and their health care professional.
Dr. Anderson’s team scoured a nationwide Propeller Health database for 8,000 patients using the digital monitors with controller therapies for asthma or chronic obstructive pulmonary disease (COPD). The study explored whether adherence differed for once-daily versus twice-daily medications, and if adherence differed based on patient age (4-60+ years).
For both asthma and COPD patients, those on once-daily regimens had higher medication adherence, compared with those who were prescribed twice-daily therapies. Plus, a greater proportion of once-daily patients met the prespecified 80% adherence threshold.
Looking across ages, medication use in the youngest group (aged 4-11 years) looked comparable with 30-somethings, “probably because parents are the ones giving the drug,” Dr. Anderson said. Mirroring patterns from other studies, adherence levels dipped in adolescents and young adults, relative to other age subsets.
Since this population-level analysis didn’t include individualized data on exacerbations or asthma control, “we can’t relate this to outcomes,” Dr. Anderson noted. But he said the data correlating medication use with adherence suggest that once-daily formulations may be the better option.
In one of the two pediatric studies, Matt McCulloch, MD, an allergy and immunology fellow working with Dr. Anderson, and colleagues reviewed charts of 40 children who received care at the Colorado Children’s multidisciplinary asthma clinic between 2018 and 2021. Half of these patients used Propeller Health sensors with their daily inhaled controller; the other patients were matched for age, ethnicity, sex, medication level, and disease control and severity – but had no electronic monitoring device.
On the whole, children who used digital monitoring for 12 months did not fare much better than matched controls on lung function (judged by forced expiratory volume) or asthma control (measured by Asthma Control Test scores).
However, within the digital monitoring group, patients who stayed on the Propeller system for 12 months did have better asthma control, fewer exacerbations, and improved asthma severity scores (measured by the Composite Asthma Severity Index), compared with when they first began digital monitoring. These children had all received care at the clinic for a while before their families opted for the electronic sensor, so “the effect wouldn’t have just been from starting in the clinic,” Dr. McCulloch said in an interview.
The gains came despite waning medication adherence. Similar to other digital monitoring studies, use of daily controller therapies in this retrospective analysis began at 50%-80% but dropped considerably during the first 4-5 months before settling into the 20%-30% range by 1 year.
Rachelle Ramsey, PhD, a pediatric research psychologist at Cincinnati Children’s Hospital Medical Center, presented data from 20 children with difficult-to-treat asthma who received 8 weeks of a digital adherence intervention during a 12-month treatment period. They analyzed three subsets – each with interventions based on how well the patients were managing daily controller therapy at baseline.
One patient with high (>80%) baseline adherence just received digital monitoring. The seven patients who began the study with intermediate (50%-80%) adherence received digital monitoring plus prescriptive text messaging. And the 12 children with poorest (<50%) baseline adherence received digital monitoring and a telehealth session in which a behavioral health specialist helped them set goals and create strategies to overcome barriers – for example, keeping the inhaler near their toothbrush in order to pair medication use with a daily habit.
“Overall, we found that matching Propeller with a behavioral intervention really improved adherence,” Dr. Ramsey said in an interview. While patients were receiving the intervention, adherence averaged across all groups increased from 39% to 76%. However, once the intervention period ended, the group’s adherence regressed toward baseline (36%).
Although adherence did not associate with clinical gains in this small study, the use of digital monitoring to improve medication adherence has translated to better outcomes in other recent efforts.
Remote monitoring
In a quality improvement project in the United Kingdom, nurses asked difficult-to-control asthma patients if they understood how to use their corticosteroid/long-acting beta2-agonist (LABA) inhalers and if they were adhering to treatment guidelines.
Those who answered yes to these questions were invited to a 28-day study that involved swapping their steroid/LABA inhalers for a different controller/bronchodilator (fluticasone/salmeterol) with INCA (Inhaler Compliance Assessment), a device that not only tracks adherence but also uses acoustics to gauge inhaler technique.
Among the 23 patients who participated, many had better clinical outcomes after 28 days of INCA monitoring. As Dr. Mosnaim told attendees, “what was amazing is so many of the patients that had been these difficult-to-control asthmatics who would have gone on to oral steroids or perhaps a biologic – lo and behold, you put them on a digital inhaler, and what do you see?” In two-thirds of the patients, “you see FeNo [a test that measures airway inflammation by detecting nitric oxide in exhalations] goes down. You see spirometry improve. You see the asthma control questionnaire improve. You see blood eosinophils go down.”
And in a 2020 randomized trial, Dr. Mosnaim and colleagues recruited 100 adults with uncontrolled asthma who had prescriptions for a daily inhaled corticosteroid and a short-acting beta-agonist (SABA) inhaler. Participants received Propeller sensors for their steroid and SABA inhalers. After a 2-week run-in period to calculate baseline corticosteroid adherence and SABA use for all participants, half the participants were randomly assigned to the control group, which had the app and sensor in silent mode, merely to collect data on medication use – whereas the treatment group received reminders, alerts, and monthly phone calls from providers who gave feedback on adherence and technique.
After 3 months of digital monitoring, patients didn’t use their rescue medication quite as often – as judged by a rise in the percentage of SABA-free days, compared with when they began the study. But the change in SABA-free days relative to baseline was more pronounced in the treatment group (19%) than in the control group (6%).
As seen in the other digital monitoring studies, adherence to daily corticosteroids fell with time, but the drop was milder in treated participants (2%) versus the control group (17%). So in this study, digital monitoring plus mobile app reminders and clinician feedback “prevented against fall in adherence to inhaled steroids over time,” Dr. Mosnaim said.
These results are “very encouraging” and offer “proof of concept that this type of remote monitoring could work,” Thanai Pongdee, MD, an allergist-immunologist with the Mayo Clinic in Rochester, Minn., said in an interview. One limitation was that the study was too short to measure exacerbation rates. A yearlong analysis would be “really fascinating because you’d catch all the seasons of the year – all the pollen seasons, all these things that could exacerbate you. Some people’s asthma can be quite seasonal.”
More important, the clinical utility of digital sensors will depend on how physicians choose to use them. If the doctor puts out a “blanket recommendation for using it but doesn’t ask you about it or doesn’t use the data to inform your care, then I think people just lose engagement and lose excitement over it,” Dr. Ramsey said. But if the health care team “asks you about the data or looks at the data with you or shows you how valuable this can be to your care, then I think that changes things.”
Building these analyses and interactions into the clinic workflow isn’t trivial. “If you have this wealth of data coming in, how are you going to look at it? Are you going to have an individual person assigned to this role? How are you going to respond to alerts?” Dr. Anderson asked.
In addition, because some digital monitors issue alerts when a patient’s asthma is not well controlled, some providers worry about liability if “something bad were to happen if you had that data but didn’t act upon it,” he said. Yet he noted that remote data monitoring is already used routinely in other areas of medicine, such as managing diabetes and heart conditions, “and it’s not like people are getting dinged for that stuff.”
Another issue is cost. Insurance only covers digital monitors in select cases, but it’s a bit of a catch-22. Insurers “don’t want to cover it until they get the data, but you can’t get the data until insurance covers it,” said Dr. Anderson, who added that “this year we finally got CPT reimbursement codes for monitoring devices.”
On the whole, studies of digital medication monitors suggest that better outcomes require “a good partnership between the health care provider and the patient,” Dr. Pongdee said. “It wasn’t like you could just put these things on and expect them to help. You still need that personal relationship to get the optimal results. We can have all this technology, but you still can’t take the people out of it.”
Dr. Mosnaim reported receiving current research grant support from GlaxoSmithKline, Novartis, Sanofi-Regeneron, and Teva; and past research grant support from AstraZeneca, Alk-Abello, and Genentech. She is immediate past president of the AAAAI, and directs the board of directors for the American Board of Allergy and Immunology. Dr. Anderson has served as a consultant for Regeneron, GlaxoSmithKline, and AstraZeneca, and has received research support from Colorado Medicaid. Dr. McCulloch and Dr. Ramsey disclosed no relevant financial relationships. Dr. Pongdee serves as an at-large director on the American Academy of Allergy, Asthma and Immunology board of directors. He receives grant funding from GlaxoSmithKline, and the Mayo Clinic is a trial site for GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
PHOENIX – Before considering oral steroids or biologic therapies, many people with difficult-to-control asthma can reduce symptoms by addressing medication adherence and inhaler technique – and digital monitoring devices can play a key role.
Often physicians “will approach a patient about a biologic if they’re not responding to standard therapy. But we need to sometimes go back to those basic building blocks, like, are you taking the standard therapy?” William C. Anderson, MD, codirector of the multidisciplinary asthma clinic at Children’s Hospital Colorado, Aurora, said in an interview.
At the annual meeting of the American Academy of Allergy, Asthma, and Immunology, he and others presented data highlighting the diagnostic and therapeutic potential of digital monitoring devices for difficult-to-control asthma, the theme of the 2022s meeting.
The Global Initiative for Asthma (GINA) defines asthma as “difficult to control” if it remains uncontrolled despite medium- or high-dose inhaled corticosteroids with a second controller or with maintenance oral steroids, or if the asthma requires high-dose treatment to curb symptoms and exacerbations. About 17% of adult asthma patients have difficult-to-control asthma, according to the 2021 GINA report.
However, correcting for inhaling technique and adherence cuts the 17% down to just 3.7%, Giselle Mosnaim, MD, an allergist at NorthShore University HealthSystem outside Chicago and AAAAI immediate past president, told attendees at a Feb. 25 session on digital technologies for asthma management.
The CRITIKAL study, which reviewed data from more than 5,000 asthma patients, “showed that, if you have critical errors in inhaler technique, this leads to worse asthma outcomes and increased asthma exacerbations,” Dr. Mosnaim said. Sadly, it also shows that, from 1975 to 2014, despite new devices and new technologies, “we still have poor inhaler technique.”
As for ways to measure adherence, physician judgments tend to be inaccurate, patient self-reporting has proved unreliable, and prescription refill data doesn’t indicate whether patients actually used the medications. “The ideal measure of adherence should be objective, accurate and unobtrusive to minimize impact on patient behavior and allow reliable data collection in real-world settings,” Dr. Mosnaim said. “So electronic medication monitors are the gold standard.”
Tracking adherence
A closing afternoon session featured three presentations on research tracking adherence and outcomes in difficult-to-treat asthma patients – two pediatric cohorts and one across all ages. All studies used the Propeller Health sensor, a Food and Drug Administration–cleared device that attaches to the patient’s inhaler and automatically collects information on where, when, and how often they use their medication. The sensor then sends that information to a data cloud accessible to the patient and their health care professional.
Dr. Anderson’s team scoured a nationwide Propeller Health database for 8,000 patients using the digital monitors with controller therapies for asthma or chronic obstructive pulmonary disease (COPD). The study explored whether adherence differed for once-daily versus twice-daily medications, and if adherence differed based on patient age (4-60+ years).
For both asthma and COPD patients, those on once-daily regimens had higher medication adherence, compared with those who were prescribed twice-daily therapies. Plus, a greater proportion of once-daily patients met the prespecified 80% adherence threshold.
Looking across ages, medication use in the youngest group (aged 4-11 years) looked comparable with 30-somethings, “probably because parents are the ones giving the drug,” Dr. Anderson said. Mirroring patterns from other studies, adherence levels dipped in adolescents and young adults, relative to other age subsets.
Since this population-level analysis didn’t include individualized data on exacerbations or asthma control, “we can’t relate this to outcomes,” Dr. Anderson noted. But he said the data correlating medication use with adherence suggest that once-daily formulations may be the better option.
In one of the two pediatric studies, Matt McCulloch, MD, an allergy and immunology fellow working with Dr. Anderson, and colleagues reviewed charts of 40 children who received care at the Colorado Children’s multidisciplinary asthma clinic between 2018 and 2021. Half of these patients used Propeller Health sensors with their daily inhaled controller; the other patients were matched for age, ethnicity, sex, medication level, and disease control and severity – but had no electronic monitoring device.
On the whole, children who used digital monitoring for 12 months did not fare much better than matched controls on lung function (judged by forced expiratory volume) or asthma control (measured by Asthma Control Test scores).
However, within the digital monitoring group, patients who stayed on the Propeller system for 12 months did have better asthma control, fewer exacerbations, and improved asthma severity scores (measured by the Composite Asthma Severity Index), compared with when they first began digital monitoring. These children had all received care at the clinic for a while before their families opted for the electronic sensor, so “the effect wouldn’t have just been from starting in the clinic,” Dr. McCulloch said in an interview.
The gains came despite waning medication adherence. Similar to other digital monitoring studies, use of daily controller therapies in this retrospective analysis began at 50%-80% but dropped considerably during the first 4-5 months before settling into the 20%-30% range by 1 year.
Rachelle Ramsey, PhD, a pediatric research psychologist at Cincinnati Children’s Hospital Medical Center, presented data from 20 children with difficult-to-treat asthma who received 8 weeks of a digital adherence intervention during a 12-month treatment period. They analyzed three subsets – each with interventions based on how well the patients were managing daily controller therapy at baseline.
One patient with high (>80%) baseline adherence just received digital monitoring. The seven patients who began the study with intermediate (50%-80%) adherence received digital monitoring plus prescriptive text messaging. And the 12 children with poorest (<50%) baseline adherence received digital monitoring and a telehealth session in which a behavioral health specialist helped them set goals and create strategies to overcome barriers – for example, keeping the inhaler near their toothbrush in order to pair medication use with a daily habit.
“Overall, we found that matching Propeller with a behavioral intervention really improved adherence,” Dr. Ramsey said in an interview. While patients were receiving the intervention, adherence averaged across all groups increased from 39% to 76%. However, once the intervention period ended, the group’s adherence regressed toward baseline (36%).
Although adherence did not associate with clinical gains in this small study, the use of digital monitoring to improve medication adherence has translated to better outcomes in other recent efforts.
Remote monitoring
In a quality improvement project in the United Kingdom, nurses asked difficult-to-control asthma patients if they understood how to use their corticosteroid/long-acting beta2-agonist (LABA) inhalers and if they were adhering to treatment guidelines.
Those who answered yes to these questions were invited to a 28-day study that involved swapping their steroid/LABA inhalers for a different controller/bronchodilator (fluticasone/salmeterol) with INCA (Inhaler Compliance Assessment), a device that not only tracks adherence but also uses acoustics to gauge inhaler technique.
Among the 23 patients who participated, many had better clinical outcomes after 28 days of INCA monitoring. As Dr. Mosnaim told attendees, “what was amazing is so many of the patients that had been these difficult-to-control asthmatics who would have gone on to oral steroids or perhaps a biologic – lo and behold, you put them on a digital inhaler, and what do you see?” In two-thirds of the patients, “you see FeNo [a test that measures airway inflammation by detecting nitric oxide in exhalations] goes down. You see spirometry improve. You see the asthma control questionnaire improve. You see blood eosinophils go down.”
And in a 2020 randomized trial, Dr. Mosnaim and colleagues recruited 100 adults with uncontrolled asthma who had prescriptions for a daily inhaled corticosteroid and a short-acting beta-agonist (SABA) inhaler. Participants received Propeller sensors for their steroid and SABA inhalers. After a 2-week run-in period to calculate baseline corticosteroid adherence and SABA use for all participants, half the participants were randomly assigned to the control group, which had the app and sensor in silent mode, merely to collect data on medication use – whereas the treatment group received reminders, alerts, and monthly phone calls from providers who gave feedback on adherence and technique.
After 3 months of digital monitoring, patients didn’t use their rescue medication quite as often – as judged by a rise in the percentage of SABA-free days, compared with when they began the study. But the change in SABA-free days relative to baseline was more pronounced in the treatment group (19%) than in the control group (6%).
As seen in the other digital monitoring studies, adherence to daily corticosteroids fell with time, but the drop was milder in treated participants (2%) versus the control group (17%). So in this study, digital monitoring plus mobile app reminders and clinician feedback “prevented against fall in adherence to inhaled steroids over time,” Dr. Mosnaim said.
These results are “very encouraging” and offer “proof of concept that this type of remote monitoring could work,” Thanai Pongdee, MD, an allergist-immunologist with the Mayo Clinic in Rochester, Minn., said in an interview. One limitation was that the study was too short to measure exacerbation rates. A yearlong analysis would be “really fascinating because you’d catch all the seasons of the year – all the pollen seasons, all these things that could exacerbate you. Some people’s asthma can be quite seasonal.”
More important, the clinical utility of digital sensors will depend on how physicians choose to use them. If the doctor puts out a “blanket recommendation for using it but doesn’t ask you about it or doesn’t use the data to inform your care, then I think people just lose engagement and lose excitement over it,” Dr. Ramsey said. But if the health care team “asks you about the data or looks at the data with you or shows you how valuable this can be to your care, then I think that changes things.”
Building these analyses and interactions into the clinic workflow isn’t trivial. “If you have this wealth of data coming in, how are you going to look at it? Are you going to have an individual person assigned to this role? How are you going to respond to alerts?” Dr. Anderson asked.
In addition, because some digital monitors issue alerts when a patient’s asthma is not well controlled, some providers worry about liability if “something bad were to happen if you had that data but didn’t act upon it,” he said. Yet he noted that remote data monitoring is already used routinely in other areas of medicine, such as managing diabetes and heart conditions, “and it’s not like people are getting dinged for that stuff.”
Another issue is cost. Insurance only covers digital monitors in select cases, but it’s a bit of a catch-22. Insurers “don’t want to cover it until they get the data, but you can’t get the data until insurance covers it,” said Dr. Anderson, who added that “this year we finally got CPT reimbursement codes for monitoring devices.”
On the whole, studies of digital medication monitors suggest that better outcomes require “a good partnership between the health care provider and the patient,” Dr. Pongdee said. “It wasn’t like you could just put these things on and expect them to help. You still need that personal relationship to get the optimal results. We can have all this technology, but you still can’t take the people out of it.”
Dr. Mosnaim reported receiving current research grant support from GlaxoSmithKline, Novartis, Sanofi-Regeneron, and Teva; and past research grant support from AstraZeneca, Alk-Abello, and Genentech. She is immediate past president of the AAAAI, and directs the board of directors for the American Board of Allergy and Immunology. Dr. Anderson has served as a consultant for Regeneron, GlaxoSmithKline, and AstraZeneca, and has received research support from Colorado Medicaid. Dr. McCulloch and Dr. Ramsey disclosed no relevant financial relationships. Dr. Pongdee serves as an at-large director on the American Academy of Allergy, Asthma and Immunology board of directors. He receives grant funding from GlaxoSmithKline, and the Mayo Clinic is a trial site for GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
AT AAAAI