User login
Hospitalist Sedation Service
There is growing demand for safe and effective procedural sedation in pediatric facilities around the country. Multiple articles published in the last 10 years have addressed the shortage of pediatric anesthesiologists to meet this rising need.14 In 2005, Lalwani and Michel published results of a survey of North American children's hospitals that showed 87% of institutions reporting barriers to development of a pediatric sedation service, and that the most common barrier was shortage of anesthesiologists.5 In our hospital, the wait time for an outpatient sedated pediatric magnetic resonance imaging (MRI) grew to as long as 6 weeks. Many institutions have had to look for unique ways to solve this problem. Pediatric sedation programs have been developed which utilize intensivists, emergency medicine physicians, nurse anesthetists, or trained sedation nurses to provide safe pediatric sedation.611 Each of these programs has grown from the particular strengths and needs at each institution. In many institutions, hospitalists are the best candidates to meet this need because of their knowledge of patient needs and safety. They are accessible and receptive to obtaining additional training and, therefore, are a natural fit to provide this service.
As more non‐anesthesiologists are called upon to meet this growing need, the principles and practice of safe sedation must be followed. The Pediatric Sedation Research Consortium, a collaborative group of 37 locations that provide data on pediatric sedations, has published their findings on the safety of pediatric sedation/anesthesia outside of the operating room (OR) performed by anesthesiologists and non‐anesthesiologists.1213 This data has been very valuable, given that studies from single institutions will often lack the power to investigate the rare, but potentially devastating, adverse events which can occur in pediatric sedation/anesthesia.
Despite the widespread use of propofol by non‐anesthesiologists, the lack of substantial data regarding safety by these providers makes its use controversial. A search of the literature finds only 1 prior article that describes the use of propofol by general pediatricians. This was sedation for endoscopy, and the sedation was performed by specially trained pediatric residents in Italy.14 A recently published study from the Consortium regarding the use of propofol showed that the majority of propofol sedations were being performed by intensivists (49%), emergency medicine physicians (36%), or anesthesiologists (10%). General pediatricians or hospitalists performed just 2% of the cases in this series.13
In 2003, a large group of experienced pediatric hospitalists were already providing sedation in the Emergency Unit (EU) and Center for After‐hours Referrals for Emergency Services (CARES) at St Louis Children's Hospital. The Division of Hospital Medicine was approached to meet the demand for increased sedation services at our institution. The Division of Pediatric Anesthesia agreed to provide our physicians with the appropriate training to provide safe, effective, and efficient sedations for painful and non‐painful procedures outside of the OR. One of our sedation units, the Ambulatory Procedure Center (APC), has been described in detail in a prior publication by Strauser Sterni et al.15 Here, we will describe the operations of our sedation services, and specifically describe the training required for our hospitalists to provide sedation services.
METHODS
St Louis Children's Hospital is a 250‐bed tertiary‐care teaching hospital affiliated with Washington University School of Medicine. The Division of Hospitalist Medicine is today comprised of 43 physicians who provide care in the EU, CARES, inpatient units, Transport, and Sedation Services at St Louis Children's Hospital. Our division also provides pediatric care in the EU, inpatient units, newborn nursery, and labor and delivery at 3 affiliated hospitals. In 2003, we developed a dedicated program in our division to meet our institutional need for sedation, with training and oversight by the Division of Pediatric Anesthesia. We developed a structured 3‐tiered program of sedation providers to manage all of our sedation needs. We then designed a training program for these 3 tiers of sedation providers. The 3‐tired program is based on the level of sedation training of each member.
Current American Academy of Pediatrics (AAP) guidelines state:
The practitioner responsible for the treatment of the patient and/or the administration of drugs for sedation must be competent to use such techniques, provide the level of monitoring provided in these guidelines, and manage complications of these techniques (ie, to be able to rescue the patient). Because the level of intended sedation may be exceeded, the practitioner must be sufficiently skilled to provide rescue should the child progress to a level of deep sedation. The practitioner must be trained in, and capable of providing, at the minimum, bag‐valve‐mask ventilation to be able to oxygenate a child who develops airway obstruction or apnea. Training in, and maintenance of, advanced pediatric airway skills is required; regular skills reinforcement is strongly encouraged.16
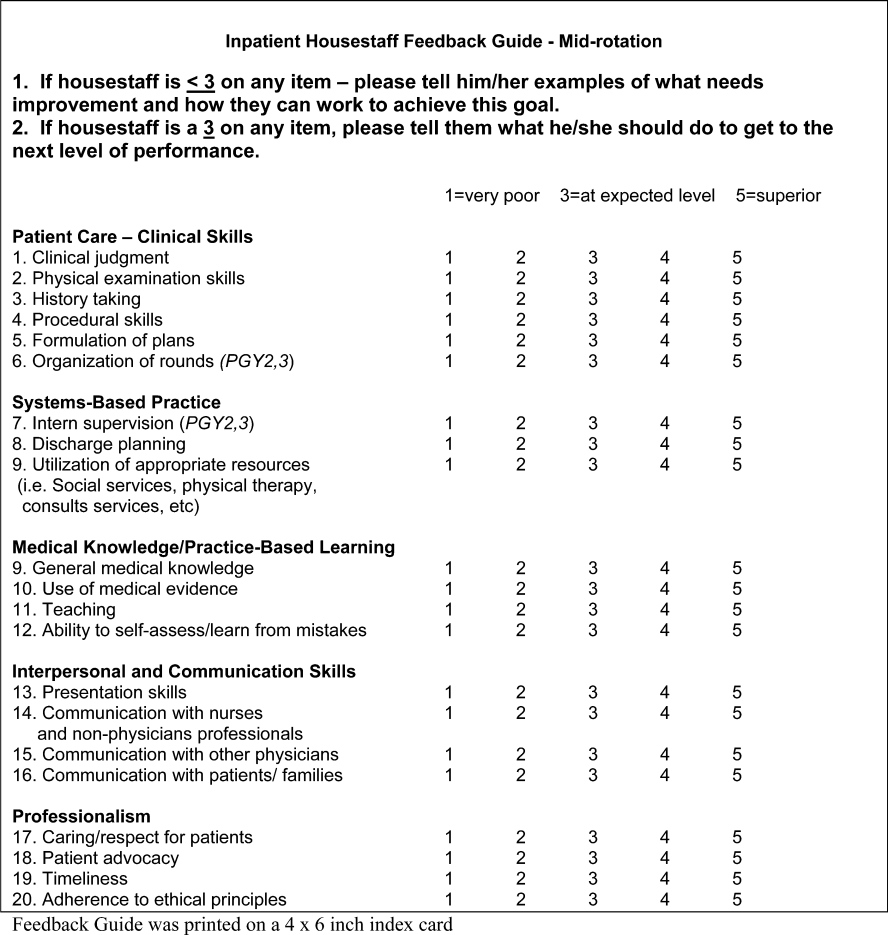
Our first‐tier sedation providers are junior faculty who provide sedation in the EU and CARES, and in the EU at our community hospitals. The first tier completes sedation training as part of overall hospitalist orientation in order to provide this service. The second tier goes through an advanced sedation provider program to provide sedation in the APC, Pediatric Acute Wound Service (PAWS), inpatient units, and After Hours sedation call, as well as the locations from the first tier. The third tier completes a more complex advanced sedation training program, specifically using propofol, and provides propofol sedation in the APC only, as well as providing sedation in all of the units from the first and second tiers. The responsibilities of the hospitalist providing sedation are described in detail by tier below, including the specific training requirements necessary for each tier (Table 1).
|
| Tier One |
| Provides sedation services in the EU |
| Drugs: ketamine, nitrous oxide |
| Training consists of: |
| 1‐hr didactic hospitalist orientation |
| 4 days of shadowing a hospitalist on the sedation service |
| Continuing on‐the‐job training |
| Tier Two |
| Provides sedation throughout the hospital: EU, APC, PAWS, and night/weekend call for urgent needs |
| Drugs: ketamine, nitrous oxide, plus pentobarbital or dexmedetomidine for radiologic procedures for both inpatients and outpatients |
| Training consists of: |
| 1 yr of first tier experience |
| 2‐hr didactic session with anesthesia |
| 1‐hr advanced hospitalist sedation orientation |
| 5 days of operating room training with an anesthesiologist |
| Tier Three |
| Provides sedation throughout the hospital: EU, APC, PAWS, and night/weekend call |
| Drugs: ketamine, nitrous oxide, pentobarbital, dexmedetomidine, and propofol |
| Training consists of: |
| 1 yr of second tier experience |
| 3‐hr didactic lecture with anesthesia |
| 10 days of operating room training |
| 25 supervised propofol sedations |
| Maintenance of certification requires >75 propofol sedations every 2 yr |
Tier One
First‐tier hospitalist sedation providers perform sedation services in the EU and CARES. The staffing model in St Louis Children's Hospital EU is comprised of a pediatric emergency medicine‐trained attending or fellow and a pediatric hospitalist who both help to oversee care within the unit. The unit is also staffed by pediatric residents, emergency medicine residents, medical students, and nurse practitioners. One of the main responsibilities of the pediatric hospitalist, however, is sedation within the unit. In CARES, the pediatric hospitalist is the attending providing direct care to patients without trainees. Any procedure requiring sedation in CARES would be performed by the hospitalist. The hospitalist providing care in the EU at both of our community hospitals would also be the physician to perform procedural sedation within the unit. Procedural sedation in all of these units are primarily for fracture reduction, laceration repair, abscess incision and drainage, foreign body removal, lumbar puncture, joint aspiration, burn debridement, and radiology imaging. Sedations performed within these units are classified as moderate or deep sedation.16 Common medications used by Tier‐One sedation providers are intravenous ketamine, inhaled nitrous oxide in combination with oral oxycodone or oral/ intravenous midazolam, intravenous pentobarbital, and occasional intravenous fentanyl in combination with intravenous midazolam.
For a hospitalist to perform any pediatric sedation within the 4 hospitals in our program, the physician must be credentialed in accordance with the specific criteria of each institution. There are varied institutional policies across all hospitals nationally. At St Louis Children's Hospital, sedation credentialing criteria states that a sedation provider must review the specific institutional policies governing sedation and perform 25 supervised sedations, before any independent sedation is attempted. The type of procedure requiring sedation and the medications used for the sedation may vary among these 25 supervised sedations. Given the structure of our program, the majority of supervised sedations are for painful procedures utilizing ketamine or a combination of oxycodone and nitrous oxide.
Our division also requires the Tier‐One group to shadow at least 4 shifts with senior hospitalists in Tiers Two or Three providing sedation, in a unit where there is an average of 6 sedations performed per day. The Tier‐One sedation hospitalist must also attend a 1‐hour didactic orientation session where the principles and practice of sedation are taught. This didactic session provides the principles of pediatric sedation and defines the important skills necessary to provide safe sedation and recovery. In addition, hospitalists are trained to recognize which children can be safely sedated by a hospitalist, and manage common side effects and adverse events during and after sedation.
The Tier‐One hospitalist performing sedation in the EU/CARES is responsible for performing pre‐procedure sedation evaluation, developing a sedation plan, and delivering procedural sedation. A dedicated sedation‐trained nurse is available throughout the procedure to record vital signs, leaving the hospitalist free to monitor the patient directly, titrate sedation medications, and manage airway or adverse events as they arise. A separate provider is responsible for performing the actual procedure. The patient continues to be monitored by the sedation nurse during the recovery period, while the hospitalist remains immediately available in the unit to address any problems. At St Louis Children's Hospital, specific monitoring and documentation criteria, using standard forms and sedation scores, are strictly adhered to for every sedation, both during the sedation and throughout the recovery phase. These criteria are based upon the AAP guidelines for monitoring and management of pediatric sedation as described by Cot and Wilson.16 Hospital Medicine provides services in the EU 17 hours per day, 7 days a week. On average, 3 sedations are provided by the hospitalist per 8‐hour shift in the EU.
Tier Two
Second‐tier providers perform all services provided by Tier‐One providers, as well as expanded sedation services on the inpatient units, APC, and PAWS. Tier‐Two providers also provide on‐call services for urgent night, weekend, and holiday sedation needs.
APC and PAWS provide more specialized sedation care than that provided in the EU. PAWS is a separate and dedicated wound care unit housed on the surgical/post‐op floor where children are sedated for painful wound care, primarily burn debridement, abscess incision and drainage (I&D), and dressing changes. Sedation services are occasionally provided for other wound care issues or procedures. Both inpatients and outpatients are seen in this unit. The unit operates 10 hours per day, 7 days a week. The PAWS unit has 2 rooms specifically equipped for sedation, monitoring, and rescue, as well as 2 additional rooms for recovery or for patients not requiring sedation. Sedations in PAWS generally utilize intravenous ketamine or inhaled nitrous oxide coupled with premedication of oxycodone. Responsibilities of the hospitalist in the unit include completing and documenting the pre‐sedation evaluation, developing an appropriate sedation plan, delivering sedation medications, ongoing monitoring and documenting of vital signs throughout the case, and recovery of the patient. All nursing staff in this unit are sedation‐trained and are responsible for continued patient monitoring during the recovery period, until the patient has returned to baseline and is safe for discharge or transfer.
The APC has been described in a prior publication.15 Hospitalist sedations in the APC are performed primarily for radiology procedures, the majority of which are MRI but also include computed tomographic (CT) and nuclear medicine scans. Sedation is also provided for automated brainstem response (ABR), electromyogram (EMG), and peripherally inserted central catheter (PICC) placements. Like PAWS, APC serves both inpatients and outpatients. The APC is staffed 10 hours per day, 5 days per week. The primary sedation medications used in this unit are ketamine, dexmedetomidine, and occasionally fentanyl and midazolam. There are 11 patient beds, all equipped for patient monitoring and recovery. Hospitalists in the APC may provide direct patient care or supervise sedation‐trained nurses delivering sedation services after having a pre‐sedation evaluation performed by the hospitalist. If a sedation‐trained nurse is delivering sedation, the hospitalist may be doing other interruptible tasks, but is immediately available on the unit to respond to any concerns from the sedation nurse. All units are fully equipped with resuscitation equipment/crash carts, and an anesthesiologist is readily available to come to the unit from the OR in the event of an issue. A rapid response team that consists of a pediatric intensive care unit (ICU) fellow, respiratory therapist, and pediatric ICU charge nurse is also always on call.
Urgent sedations on the inpatient wards are common and can usually be accommodated in PAWS or APC. Rarely, however, an MRI, CT, joint aspiration, abscess drainage, or lumbar puncture must be completed urgently in the evenings, weekends, or holidays. In this situation, the hospitalist is responsible for performing the pre‐sedation evaluation, developing an appropriate sedation technique, delivering the sedation medications, monitoring and documenting during the procedure as well as throughout the recovery period, until the patient has returned to baseline. Sedation‐trained nursing staff are available to provide assistance. The sedation medications commonly used in these after‐hours sedations include ketamine for short or painful procedures and dexmedetomidine for longer radiology studies.
Training for second‐tier services consists of a minimum of 1 year of first‐tier sedation experience, a 2‐hour didactic lecture with Pediatric Anesthesia, a 1‐hour hospitalist orientation for advanced sedation providers, and 5 days of OR training with an anesthesiologist. Operating room training focuses on building skills in bag‐mask ventilation, intravenous (IV) placement, endotracheal intubation, and laryngeal mask airway (LMA) placement.
Tier Three
Third‐tier sedation providers have completed all of the training of a Tier‐Two provider and have had additional training to prepare them to deliver propofol for non‐painful procedures. Hospitalist‐delivered propofol sedation is provided exclusively in the APC for non‐painful procedures. The hospitalist is responsible for the pre‐sedation evaluation, induction and maintenance of sedation, and patient monitoring and documentation of vital signs. Monitoring for propofol sedation includes end‐tidal carbon dioxide monitoring in addition to electrocardiogram (EKG), respiratory rate (RR), pulse oximetry, and non‐invasive blood pressure (NIBP). A sedation‐trained nurse is present during induction and assists with patient positioning within the scanner. The nurse will then assume care of the patient at the completion of the procedure to continue patient monitoring during recovery.
Training for Tier‐Three providers consists of a 3‐hour didactic session with Anesthesia, 10 days of OR training, use of simulation scenarios, and a written exam. The hospitalist must then perform 25 supervised propofol sedations before being credentialed to provide propofol sedation independently. To maintain certification for Tier‐Three services, hospitalists must perform at least 75 propofol sedations every 2 years.
RESULTS
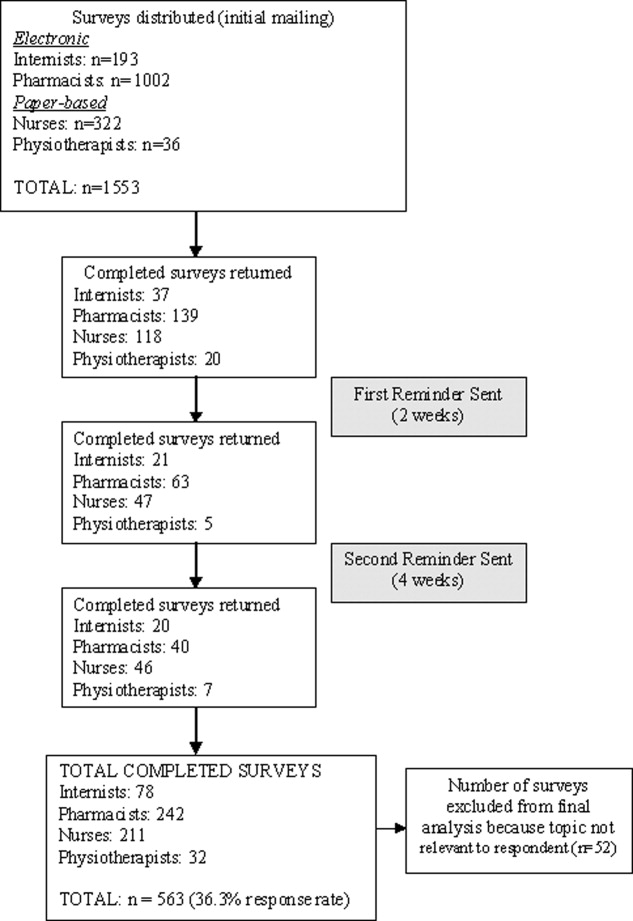
Utilizing this design and training method, we have developed a successful pediatric hospitalist sedation program. Based on fiscal year 2009 billing data, the Division of Hospital Medicine performed 2471 sedations. There were 2069 sedations performed in APC or PAWS; of those, 1017 were performed on inpatients and 1052 were performed on outpatients. Hospitalists performed 402 sedations on patients in the EU. The EU numbers are likely much larger given that, for over half the year, billing data was not collected from the EU. Unfortunately, we did not have billing data regarding night and weekend sedations, but our best estimate is 1 to 2 per week. The wait time for an outpatient sedated pediatric MRI has gone from 6 weeks to 2 days or less. As of July 2010, we have trained 90 providers at Tier One, 32 at Tier Two, and 11 at Tier Three. We currently have 43 hospitalists providing Tier‐One sedation, 18 providing Tier‐Two, and 6 providing Tier‐Three. Average cumulative hospitalist experience is 1 year for Tier One, 5 years for Tier Two, and 10 years for Tier Three.
DISCUSSION
We believe this is the first description of a pediatric hospitalist training program for a sedation service. However, it is clear that many other pediatric hospitalists are performing sedation and developing similar training programs. When starting a program such as this, there are many things to consider. First, patient volume/demand must allow for each hospitalist to perform sedations on a regular basis, both for training and Maintenance of Certification. Second, Anesthesia must be willing to provide training and oversight. Third, the hospital or university must be willing to support the cost associated with the training period. Finally, negotiating with third party payers for reimbursement is critical to financial sustainability.
The success of our program hinged upon the ability to develop a strong and collaborative relationship with Anesthesiology. Many factors played into making this relationship work. Initially, Anesthesia approached us to help them meet an unmet clinical need. Because of this, we were viewed as helpful and as problem solvers, rather than as a threat. Additionally, each division had a sedation service champion that pushed for the development of a hospitalist sedation service. Lastly, regular meetings with Anesthesia, and the intense training program itself, helped to develop a sense of collegiality between the divisions.
We have faced many challenges and learned many lessons while developing this program. There is a significant cost to training sedation providers; 47 hospitalists trained to provide Tier‐One sedation have left the program. Of those, 16 hospitalists completed training for Tier‐Two sedation, and 5 completed Tier Three. The Tier‐Two training described earlier requires approximately 50 hours of dedicated time away from other hospitalist duties, while Tier Three requires an additional 125 hours. The majority of our turnover occurred in the first few years of the program. From a financial perspective, we have had to reserve sedation training beyond Tier One to hospitalists who are able to demonstrate evidence of a long‐term commitment to our division. Every person providing Tier‐Three sedation has been with the division over 6 years. From a broader perspective, we are providing hospitalists with an important and useful skill that may enhance their careerssafe and effective sedation.
Balancing the volume of cases is another issue to consider. Our goal is to provide safe and timely sedation, therefore we need to have enough scheduled cases to maintain competency and financial viability, but we must also leave adequate flexibility in the schedule for urgent cases.
In addition to the operating room training, we are beginning to incorporate pediatric simulation as an adjunct to our training. We have designed simulation scenarios which address issues of obstruction, apnea, hypotension, bronchospasm, and aspiration. However, OR training remains a mandatory requirement for sedation training and, at times, can be challenging to schedule.
We complete a post‐sedation assessment on all patients; we are currently performing a chart review of over 1600 patients sedated with propofol, to look at the rate of planned and unplanned interventions. We believe this data will show that our training has been successful, and that with analysis of our Quality Improvement data, we can improve the safety and efficacy of our sedation program even further.
CONCLUSIONS
A pediatric hospitalist sedation service, with proper training and oversight, can successfully augment sedation services provided by anesthesiologists. As has been stated in prior publications, a defined system, and the use of a dedicated well‐trained team makes a sedation service a success.1719 A collegial and mutually respectful relationship between Anesthesia and non‐Anesthesia sedation providers is critical in developing and maintaining a successful sedation program.
- ,,,,.Anesthesia services outside of the operating room.Pediatr Nurs.2007;33(3):232,234,236–237.
- ,.Pediatric sedation/anesthesia outside the operating room.Curr Opin Anaesthesiol.2008;21(4):494–498.
- ,.Procedural sedation in the pediatric patient.Anesthesiol Clin North Am.2005;23(4):635–654, viii.
- .Pediatric sedation: can it be safely performed by non‐anesthesiologists?Curr Opin Anaesthesiol.2002;15(4):455–459.
- ,.Pediatric sedation in North American children's hospitals: a survey of anesthesia providers.Paediatr Anaesth.2005;15(3):209–213.
- ,,, et al.Safety of propofol sedation for pediatric outpatient procedures.Clin Pediatr (Phila).2009;48(8):819–823.
- ,,, et al.High dose dexmedetomidine as the sole sedative for pediatric MRI.Paediatr Anaesth.2008;18(5):403–411.
- ,.Successful implementation of a radiology sedation service staffed exclusively by pediatric emergency physicians.Pediatrics.2006;117(3):e413–e422.
- ,.Management of children undergoing painful procedures in the emergency department by non‐anesthesiologists.Isr Med Assoc J.2004;6(6):350–355.
- ,,,,.Development of a nurse‐led sedation service for paediatric magnetic resonance imaging.Lancet.1999;353(9165):1667–1671.
- ,,.Propofol sedation: intensivists' experience with 7304 cases in a children's hospital.Pediatrics.2007;120(6):e1411–e1417.
- .Risk and safety of pediatric sedation/anesthesia for procedures outside the operating room.Curr Opin Anaesthesiol.2009;22(4):509–513.
- ,,,,.The incidence and nature of adverse events during pediatric sedation/anesthesia with propofol for procedures outside the operating room: a report from the Pediatric Sedation Research Consortium.Anesth Analg.2009;108(3):795–804.
- ,,, et al.Deep sedation with propofol for upper gastrointestinal endoscopy in children, administered by specially trained pediatricians: a prospective case series with emphasis on side effects.Endoscopy.2006;38(4):368–375.
- ,,,,.A model for pediatric sedation centers using pharmacologic sedation for successful completion of radiologic and procedural studies.J Radiol Nurs2008;27(2):46–60.
- ,.Guidelines for monitoring and management of pediatric patients during and after sedation for diagnostic and therapeutic procedures: an update.Pediatrics.2006;118(6):2587–2602.
- ,.Non‐anesthesiologist‐provided pediatric procedural sedation: an update.Curr Opin Anaesthesiol.2007;20(4):365–372.
- ,,.Professional skills and competence for safe and effective procedural sedation in children: recommendations based on a systematic review of the literature.Int J Pediatr.2010; doi://10.1155/2010/934298.
- ,.Pediatric sedation outside the operating room: the year in review.Curr Opin Anaesthesiol.2005;18(4):442–446.
There is growing demand for safe and effective procedural sedation in pediatric facilities around the country. Multiple articles published in the last 10 years have addressed the shortage of pediatric anesthesiologists to meet this rising need.14 In 2005, Lalwani and Michel published results of a survey of North American children's hospitals that showed 87% of institutions reporting barriers to development of a pediatric sedation service, and that the most common barrier was shortage of anesthesiologists.5 In our hospital, the wait time for an outpatient sedated pediatric magnetic resonance imaging (MRI) grew to as long as 6 weeks. Many institutions have had to look for unique ways to solve this problem. Pediatric sedation programs have been developed which utilize intensivists, emergency medicine physicians, nurse anesthetists, or trained sedation nurses to provide safe pediatric sedation.611 Each of these programs has grown from the particular strengths and needs at each institution. In many institutions, hospitalists are the best candidates to meet this need because of their knowledge of patient needs and safety. They are accessible and receptive to obtaining additional training and, therefore, are a natural fit to provide this service.
As more non‐anesthesiologists are called upon to meet this growing need, the principles and practice of safe sedation must be followed. The Pediatric Sedation Research Consortium, a collaborative group of 37 locations that provide data on pediatric sedations, has published their findings on the safety of pediatric sedation/anesthesia outside of the operating room (OR) performed by anesthesiologists and non‐anesthesiologists.1213 This data has been very valuable, given that studies from single institutions will often lack the power to investigate the rare, but potentially devastating, adverse events which can occur in pediatric sedation/anesthesia.
Despite the widespread use of propofol by non‐anesthesiologists, the lack of substantial data regarding safety by these providers makes its use controversial. A search of the literature finds only 1 prior article that describes the use of propofol by general pediatricians. This was sedation for endoscopy, and the sedation was performed by specially trained pediatric residents in Italy.14 A recently published study from the Consortium regarding the use of propofol showed that the majority of propofol sedations were being performed by intensivists (49%), emergency medicine physicians (36%), or anesthesiologists (10%). General pediatricians or hospitalists performed just 2% of the cases in this series.13
In 2003, a large group of experienced pediatric hospitalists were already providing sedation in the Emergency Unit (EU) and Center for After‐hours Referrals for Emergency Services (CARES) at St Louis Children's Hospital. The Division of Hospital Medicine was approached to meet the demand for increased sedation services at our institution. The Division of Pediatric Anesthesia agreed to provide our physicians with the appropriate training to provide safe, effective, and efficient sedations for painful and non‐painful procedures outside of the OR. One of our sedation units, the Ambulatory Procedure Center (APC), has been described in detail in a prior publication by Strauser Sterni et al.15 Here, we will describe the operations of our sedation services, and specifically describe the training required for our hospitalists to provide sedation services.
METHODS
St Louis Children's Hospital is a 250‐bed tertiary‐care teaching hospital affiliated with Washington University School of Medicine. The Division of Hospitalist Medicine is today comprised of 43 physicians who provide care in the EU, CARES, inpatient units, Transport, and Sedation Services at St Louis Children's Hospital. Our division also provides pediatric care in the EU, inpatient units, newborn nursery, and labor and delivery at 3 affiliated hospitals. In 2003, we developed a dedicated program in our division to meet our institutional need for sedation, with training and oversight by the Division of Pediatric Anesthesia. We developed a structured 3‐tiered program of sedation providers to manage all of our sedation needs. We then designed a training program for these 3 tiers of sedation providers. The 3‐tired program is based on the level of sedation training of each member.
Current American Academy of Pediatrics (AAP) guidelines state:
The practitioner responsible for the treatment of the patient and/or the administration of drugs for sedation must be competent to use such techniques, provide the level of monitoring provided in these guidelines, and manage complications of these techniques (ie, to be able to rescue the patient). Because the level of intended sedation may be exceeded, the practitioner must be sufficiently skilled to provide rescue should the child progress to a level of deep sedation. The practitioner must be trained in, and capable of providing, at the minimum, bag‐valve‐mask ventilation to be able to oxygenate a child who develops airway obstruction or apnea. Training in, and maintenance of, advanced pediatric airway skills is required; regular skills reinforcement is strongly encouraged.16
Our first‐tier sedation providers are junior faculty who provide sedation in the EU and CARES, and in the EU at our community hospitals. The first tier completes sedation training as part of overall hospitalist orientation in order to provide this service. The second tier goes through an advanced sedation provider program to provide sedation in the APC, Pediatric Acute Wound Service (PAWS), inpatient units, and After Hours sedation call, as well as the locations from the first tier. The third tier completes a more complex advanced sedation training program, specifically using propofol, and provides propofol sedation in the APC only, as well as providing sedation in all of the units from the first and second tiers. The responsibilities of the hospitalist providing sedation are described in detail by tier below, including the specific training requirements necessary for each tier (Table 1).
|
| Tier One |
| Provides sedation services in the EU |
| Drugs: ketamine, nitrous oxide |
| Training consists of: |
| 1‐hr didactic hospitalist orientation |
| 4 days of shadowing a hospitalist on the sedation service |
| Continuing on‐the‐job training |
| Tier Two |
| Provides sedation throughout the hospital: EU, APC, PAWS, and night/weekend call for urgent needs |
| Drugs: ketamine, nitrous oxide, plus pentobarbital or dexmedetomidine for radiologic procedures for both inpatients and outpatients |
| Training consists of: |
| 1 yr of first tier experience |
| 2‐hr didactic session with anesthesia |
| 1‐hr advanced hospitalist sedation orientation |
| 5 days of operating room training with an anesthesiologist |
| Tier Three |
| Provides sedation throughout the hospital: EU, APC, PAWS, and night/weekend call |
| Drugs: ketamine, nitrous oxide, pentobarbital, dexmedetomidine, and propofol |
| Training consists of: |
| 1 yr of second tier experience |
| 3‐hr didactic lecture with anesthesia |
| 10 days of operating room training |
| 25 supervised propofol sedations |
| Maintenance of certification requires >75 propofol sedations every 2 yr |
Tier One
First‐tier hospitalist sedation providers perform sedation services in the EU and CARES. The staffing model in St Louis Children's Hospital EU is comprised of a pediatric emergency medicine‐trained attending or fellow and a pediatric hospitalist who both help to oversee care within the unit. The unit is also staffed by pediatric residents, emergency medicine residents, medical students, and nurse practitioners. One of the main responsibilities of the pediatric hospitalist, however, is sedation within the unit. In CARES, the pediatric hospitalist is the attending providing direct care to patients without trainees. Any procedure requiring sedation in CARES would be performed by the hospitalist. The hospitalist providing care in the EU at both of our community hospitals would also be the physician to perform procedural sedation within the unit. Procedural sedation in all of these units are primarily for fracture reduction, laceration repair, abscess incision and drainage, foreign body removal, lumbar puncture, joint aspiration, burn debridement, and radiology imaging. Sedations performed within these units are classified as moderate or deep sedation.16 Common medications used by Tier‐One sedation providers are intravenous ketamine, inhaled nitrous oxide in combination with oral oxycodone or oral/ intravenous midazolam, intravenous pentobarbital, and occasional intravenous fentanyl in combination with intravenous midazolam.
For a hospitalist to perform any pediatric sedation within the 4 hospitals in our program, the physician must be credentialed in accordance with the specific criteria of each institution. There are varied institutional policies across all hospitals nationally. At St Louis Children's Hospital, sedation credentialing criteria states that a sedation provider must review the specific institutional policies governing sedation and perform 25 supervised sedations, before any independent sedation is attempted. The type of procedure requiring sedation and the medications used for the sedation may vary among these 25 supervised sedations. Given the structure of our program, the majority of supervised sedations are for painful procedures utilizing ketamine or a combination of oxycodone and nitrous oxide.
Our division also requires the Tier‐One group to shadow at least 4 shifts with senior hospitalists in Tiers Two or Three providing sedation, in a unit where there is an average of 6 sedations performed per day. The Tier‐One sedation hospitalist must also attend a 1‐hour didactic orientation session where the principles and practice of sedation are taught. This didactic session provides the principles of pediatric sedation and defines the important skills necessary to provide safe sedation and recovery. In addition, hospitalists are trained to recognize which children can be safely sedated by a hospitalist, and manage common side effects and adverse events during and after sedation.
The Tier‐One hospitalist performing sedation in the EU/CARES is responsible for performing pre‐procedure sedation evaluation, developing a sedation plan, and delivering procedural sedation. A dedicated sedation‐trained nurse is available throughout the procedure to record vital signs, leaving the hospitalist free to monitor the patient directly, titrate sedation medications, and manage airway or adverse events as they arise. A separate provider is responsible for performing the actual procedure. The patient continues to be monitored by the sedation nurse during the recovery period, while the hospitalist remains immediately available in the unit to address any problems. At St Louis Children's Hospital, specific monitoring and documentation criteria, using standard forms and sedation scores, are strictly adhered to for every sedation, both during the sedation and throughout the recovery phase. These criteria are based upon the AAP guidelines for monitoring and management of pediatric sedation as described by Cot and Wilson.16 Hospital Medicine provides services in the EU 17 hours per day, 7 days a week. On average, 3 sedations are provided by the hospitalist per 8‐hour shift in the EU.
Tier Two
Second‐tier providers perform all services provided by Tier‐One providers, as well as expanded sedation services on the inpatient units, APC, and PAWS. Tier‐Two providers also provide on‐call services for urgent night, weekend, and holiday sedation needs.
APC and PAWS provide more specialized sedation care than that provided in the EU. PAWS is a separate and dedicated wound care unit housed on the surgical/post‐op floor where children are sedated for painful wound care, primarily burn debridement, abscess incision and drainage (I&D), and dressing changes. Sedation services are occasionally provided for other wound care issues or procedures. Both inpatients and outpatients are seen in this unit. The unit operates 10 hours per day, 7 days a week. The PAWS unit has 2 rooms specifically equipped for sedation, monitoring, and rescue, as well as 2 additional rooms for recovery or for patients not requiring sedation. Sedations in PAWS generally utilize intravenous ketamine or inhaled nitrous oxide coupled with premedication of oxycodone. Responsibilities of the hospitalist in the unit include completing and documenting the pre‐sedation evaluation, developing an appropriate sedation plan, delivering sedation medications, ongoing monitoring and documenting of vital signs throughout the case, and recovery of the patient. All nursing staff in this unit are sedation‐trained and are responsible for continued patient monitoring during the recovery period, until the patient has returned to baseline and is safe for discharge or transfer.
The APC has been described in a prior publication.15 Hospitalist sedations in the APC are performed primarily for radiology procedures, the majority of which are MRI but also include computed tomographic (CT) and nuclear medicine scans. Sedation is also provided for automated brainstem response (ABR), electromyogram (EMG), and peripherally inserted central catheter (PICC) placements. Like PAWS, APC serves both inpatients and outpatients. The APC is staffed 10 hours per day, 5 days per week. The primary sedation medications used in this unit are ketamine, dexmedetomidine, and occasionally fentanyl and midazolam. There are 11 patient beds, all equipped for patient monitoring and recovery. Hospitalists in the APC may provide direct patient care or supervise sedation‐trained nurses delivering sedation services after having a pre‐sedation evaluation performed by the hospitalist. If a sedation‐trained nurse is delivering sedation, the hospitalist may be doing other interruptible tasks, but is immediately available on the unit to respond to any concerns from the sedation nurse. All units are fully equipped with resuscitation equipment/crash carts, and an anesthesiologist is readily available to come to the unit from the OR in the event of an issue. A rapid response team that consists of a pediatric intensive care unit (ICU) fellow, respiratory therapist, and pediatric ICU charge nurse is also always on call.
Urgent sedations on the inpatient wards are common and can usually be accommodated in PAWS or APC. Rarely, however, an MRI, CT, joint aspiration, abscess drainage, or lumbar puncture must be completed urgently in the evenings, weekends, or holidays. In this situation, the hospitalist is responsible for performing the pre‐sedation evaluation, developing an appropriate sedation technique, delivering the sedation medications, monitoring and documenting during the procedure as well as throughout the recovery period, until the patient has returned to baseline. Sedation‐trained nursing staff are available to provide assistance. The sedation medications commonly used in these after‐hours sedations include ketamine for short or painful procedures and dexmedetomidine for longer radiology studies.
Training for second‐tier services consists of a minimum of 1 year of first‐tier sedation experience, a 2‐hour didactic lecture with Pediatric Anesthesia, a 1‐hour hospitalist orientation for advanced sedation providers, and 5 days of OR training with an anesthesiologist. Operating room training focuses on building skills in bag‐mask ventilation, intravenous (IV) placement, endotracheal intubation, and laryngeal mask airway (LMA) placement.
Tier Three
Third‐tier sedation providers have completed all of the training of a Tier‐Two provider and have had additional training to prepare them to deliver propofol for non‐painful procedures. Hospitalist‐delivered propofol sedation is provided exclusively in the APC for non‐painful procedures. The hospitalist is responsible for the pre‐sedation evaluation, induction and maintenance of sedation, and patient monitoring and documentation of vital signs. Monitoring for propofol sedation includes end‐tidal carbon dioxide monitoring in addition to electrocardiogram (EKG), respiratory rate (RR), pulse oximetry, and non‐invasive blood pressure (NIBP). A sedation‐trained nurse is present during induction and assists with patient positioning within the scanner. The nurse will then assume care of the patient at the completion of the procedure to continue patient monitoring during recovery.
Training for Tier‐Three providers consists of a 3‐hour didactic session with Anesthesia, 10 days of OR training, use of simulation scenarios, and a written exam. The hospitalist must then perform 25 supervised propofol sedations before being credentialed to provide propofol sedation independently. To maintain certification for Tier‐Three services, hospitalists must perform at least 75 propofol sedations every 2 years.
RESULTS
Utilizing this design and training method, we have developed a successful pediatric hospitalist sedation program. Based on fiscal year 2009 billing data, the Division of Hospital Medicine performed 2471 sedations. There were 2069 sedations performed in APC or PAWS; of those, 1017 were performed on inpatients and 1052 were performed on outpatients. Hospitalists performed 402 sedations on patients in the EU. The EU numbers are likely much larger given that, for over half the year, billing data was not collected from the EU. Unfortunately, we did not have billing data regarding night and weekend sedations, but our best estimate is 1 to 2 per week. The wait time for an outpatient sedated pediatric MRI has gone from 6 weeks to 2 days or less. As of July 2010, we have trained 90 providers at Tier One, 32 at Tier Two, and 11 at Tier Three. We currently have 43 hospitalists providing Tier‐One sedation, 18 providing Tier‐Two, and 6 providing Tier‐Three. Average cumulative hospitalist experience is 1 year for Tier One, 5 years for Tier Two, and 10 years for Tier Three.
DISCUSSION
We believe this is the first description of a pediatric hospitalist training program for a sedation service. However, it is clear that many other pediatric hospitalists are performing sedation and developing similar training programs. When starting a program such as this, there are many things to consider. First, patient volume/demand must allow for each hospitalist to perform sedations on a regular basis, both for training and Maintenance of Certification. Second, Anesthesia must be willing to provide training and oversight. Third, the hospital or university must be willing to support the cost associated with the training period. Finally, negotiating with third party payers for reimbursement is critical to financial sustainability.
The success of our program hinged upon the ability to develop a strong and collaborative relationship with Anesthesiology. Many factors played into making this relationship work. Initially, Anesthesia approached us to help them meet an unmet clinical need. Because of this, we were viewed as helpful and as problem solvers, rather than as a threat. Additionally, each division had a sedation service champion that pushed for the development of a hospitalist sedation service. Lastly, regular meetings with Anesthesia, and the intense training program itself, helped to develop a sense of collegiality between the divisions.
We have faced many challenges and learned many lessons while developing this program. There is a significant cost to training sedation providers; 47 hospitalists trained to provide Tier‐One sedation have left the program. Of those, 16 hospitalists completed training for Tier‐Two sedation, and 5 completed Tier Three. The Tier‐Two training described earlier requires approximately 50 hours of dedicated time away from other hospitalist duties, while Tier Three requires an additional 125 hours. The majority of our turnover occurred in the first few years of the program. From a financial perspective, we have had to reserve sedation training beyond Tier One to hospitalists who are able to demonstrate evidence of a long‐term commitment to our division. Every person providing Tier‐Three sedation has been with the division over 6 years. From a broader perspective, we are providing hospitalists with an important and useful skill that may enhance their careerssafe and effective sedation.
Balancing the volume of cases is another issue to consider. Our goal is to provide safe and timely sedation, therefore we need to have enough scheduled cases to maintain competency and financial viability, but we must also leave adequate flexibility in the schedule for urgent cases.
In addition to the operating room training, we are beginning to incorporate pediatric simulation as an adjunct to our training. We have designed simulation scenarios which address issues of obstruction, apnea, hypotension, bronchospasm, and aspiration. However, OR training remains a mandatory requirement for sedation training and, at times, can be challenging to schedule.
We complete a post‐sedation assessment on all patients; we are currently performing a chart review of over 1600 patients sedated with propofol, to look at the rate of planned and unplanned interventions. We believe this data will show that our training has been successful, and that with analysis of our Quality Improvement data, we can improve the safety and efficacy of our sedation program even further.
CONCLUSIONS
A pediatric hospitalist sedation service, with proper training and oversight, can successfully augment sedation services provided by anesthesiologists. As has been stated in prior publications, a defined system, and the use of a dedicated well‐trained team makes a sedation service a success.1719 A collegial and mutually respectful relationship between Anesthesia and non‐Anesthesia sedation providers is critical in developing and maintaining a successful sedation program.
There is growing demand for safe and effective procedural sedation in pediatric facilities around the country. Multiple articles published in the last 10 years have addressed the shortage of pediatric anesthesiologists to meet this rising need.14 In 2005, Lalwani and Michel published results of a survey of North American children's hospitals that showed 87% of institutions reporting barriers to development of a pediatric sedation service, and that the most common barrier was shortage of anesthesiologists.5 In our hospital, the wait time for an outpatient sedated pediatric magnetic resonance imaging (MRI) grew to as long as 6 weeks. Many institutions have had to look for unique ways to solve this problem. Pediatric sedation programs have been developed which utilize intensivists, emergency medicine physicians, nurse anesthetists, or trained sedation nurses to provide safe pediatric sedation.611 Each of these programs has grown from the particular strengths and needs at each institution. In many institutions, hospitalists are the best candidates to meet this need because of their knowledge of patient needs and safety. They are accessible and receptive to obtaining additional training and, therefore, are a natural fit to provide this service.
As more non‐anesthesiologists are called upon to meet this growing need, the principles and practice of safe sedation must be followed. The Pediatric Sedation Research Consortium, a collaborative group of 37 locations that provide data on pediatric sedations, has published their findings on the safety of pediatric sedation/anesthesia outside of the operating room (OR) performed by anesthesiologists and non‐anesthesiologists.1213 This data has been very valuable, given that studies from single institutions will often lack the power to investigate the rare, but potentially devastating, adverse events which can occur in pediatric sedation/anesthesia.
Despite the widespread use of propofol by non‐anesthesiologists, the lack of substantial data regarding safety by these providers makes its use controversial. A search of the literature finds only 1 prior article that describes the use of propofol by general pediatricians. This was sedation for endoscopy, and the sedation was performed by specially trained pediatric residents in Italy.14 A recently published study from the Consortium regarding the use of propofol showed that the majority of propofol sedations were being performed by intensivists (49%), emergency medicine physicians (36%), or anesthesiologists (10%). General pediatricians or hospitalists performed just 2% of the cases in this series.13
In 2003, a large group of experienced pediatric hospitalists were already providing sedation in the Emergency Unit (EU) and Center for After‐hours Referrals for Emergency Services (CARES) at St Louis Children's Hospital. The Division of Hospital Medicine was approached to meet the demand for increased sedation services at our institution. The Division of Pediatric Anesthesia agreed to provide our physicians with the appropriate training to provide safe, effective, and efficient sedations for painful and non‐painful procedures outside of the OR. One of our sedation units, the Ambulatory Procedure Center (APC), has been described in detail in a prior publication by Strauser Sterni et al.15 Here, we will describe the operations of our sedation services, and specifically describe the training required for our hospitalists to provide sedation services.
METHODS
St Louis Children's Hospital is a 250‐bed tertiary‐care teaching hospital affiliated with Washington University School of Medicine. The Division of Hospitalist Medicine is today comprised of 43 physicians who provide care in the EU, CARES, inpatient units, Transport, and Sedation Services at St Louis Children's Hospital. Our division also provides pediatric care in the EU, inpatient units, newborn nursery, and labor and delivery at 3 affiliated hospitals. In 2003, we developed a dedicated program in our division to meet our institutional need for sedation, with training and oversight by the Division of Pediatric Anesthesia. We developed a structured 3‐tiered program of sedation providers to manage all of our sedation needs. We then designed a training program for these 3 tiers of sedation providers. The 3‐tired program is based on the level of sedation training of each member.
Current American Academy of Pediatrics (AAP) guidelines state:
The practitioner responsible for the treatment of the patient and/or the administration of drugs for sedation must be competent to use such techniques, provide the level of monitoring provided in these guidelines, and manage complications of these techniques (ie, to be able to rescue the patient). Because the level of intended sedation may be exceeded, the practitioner must be sufficiently skilled to provide rescue should the child progress to a level of deep sedation. The practitioner must be trained in, and capable of providing, at the minimum, bag‐valve‐mask ventilation to be able to oxygenate a child who develops airway obstruction or apnea. Training in, and maintenance of, advanced pediatric airway skills is required; regular skills reinforcement is strongly encouraged.16
Our first‐tier sedation providers are junior faculty who provide sedation in the EU and CARES, and in the EU at our community hospitals. The first tier completes sedation training as part of overall hospitalist orientation in order to provide this service. The second tier goes through an advanced sedation provider program to provide sedation in the APC, Pediatric Acute Wound Service (PAWS), inpatient units, and After Hours sedation call, as well as the locations from the first tier. The third tier completes a more complex advanced sedation training program, specifically using propofol, and provides propofol sedation in the APC only, as well as providing sedation in all of the units from the first and second tiers. The responsibilities of the hospitalist providing sedation are described in detail by tier below, including the specific training requirements necessary for each tier (Table 1).
|
| Tier One |
| Provides sedation services in the EU |
| Drugs: ketamine, nitrous oxide |
| Training consists of: |
| 1‐hr didactic hospitalist orientation |
| 4 days of shadowing a hospitalist on the sedation service |
| Continuing on‐the‐job training |
| Tier Two |
| Provides sedation throughout the hospital: EU, APC, PAWS, and night/weekend call for urgent needs |
| Drugs: ketamine, nitrous oxide, plus pentobarbital or dexmedetomidine for radiologic procedures for both inpatients and outpatients |
| Training consists of: |
| 1 yr of first tier experience |
| 2‐hr didactic session with anesthesia |
| 1‐hr advanced hospitalist sedation orientation |
| 5 days of operating room training with an anesthesiologist |
| Tier Three |
| Provides sedation throughout the hospital: EU, APC, PAWS, and night/weekend call |
| Drugs: ketamine, nitrous oxide, pentobarbital, dexmedetomidine, and propofol |
| Training consists of: |
| 1 yr of second tier experience |
| 3‐hr didactic lecture with anesthesia |
| 10 days of operating room training |
| 25 supervised propofol sedations |
| Maintenance of certification requires >75 propofol sedations every 2 yr |
Tier One
First‐tier hospitalist sedation providers perform sedation services in the EU and CARES. The staffing model in St Louis Children's Hospital EU is comprised of a pediatric emergency medicine‐trained attending or fellow and a pediatric hospitalist who both help to oversee care within the unit. The unit is also staffed by pediatric residents, emergency medicine residents, medical students, and nurse practitioners. One of the main responsibilities of the pediatric hospitalist, however, is sedation within the unit. In CARES, the pediatric hospitalist is the attending providing direct care to patients without trainees. Any procedure requiring sedation in CARES would be performed by the hospitalist. The hospitalist providing care in the EU at both of our community hospitals would also be the physician to perform procedural sedation within the unit. Procedural sedation in all of these units are primarily for fracture reduction, laceration repair, abscess incision and drainage, foreign body removal, lumbar puncture, joint aspiration, burn debridement, and radiology imaging. Sedations performed within these units are classified as moderate or deep sedation.16 Common medications used by Tier‐One sedation providers are intravenous ketamine, inhaled nitrous oxide in combination with oral oxycodone or oral/ intravenous midazolam, intravenous pentobarbital, and occasional intravenous fentanyl in combination with intravenous midazolam.
For a hospitalist to perform any pediatric sedation within the 4 hospitals in our program, the physician must be credentialed in accordance with the specific criteria of each institution. There are varied institutional policies across all hospitals nationally. At St Louis Children's Hospital, sedation credentialing criteria states that a sedation provider must review the specific institutional policies governing sedation and perform 25 supervised sedations, before any independent sedation is attempted. The type of procedure requiring sedation and the medications used for the sedation may vary among these 25 supervised sedations. Given the structure of our program, the majority of supervised sedations are for painful procedures utilizing ketamine or a combination of oxycodone and nitrous oxide.
Our division also requires the Tier‐One group to shadow at least 4 shifts with senior hospitalists in Tiers Two or Three providing sedation, in a unit where there is an average of 6 sedations performed per day. The Tier‐One sedation hospitalist must also attend a 1‐hour didactic orientation session where the principles and practice of sedation are taught. This didactic session provides the principles of pediatric sedation and defines the important skills necessary to provide safe sedation and recovery. In addition, hospitalists are trained to recognize which children can be safely sedated by a hospitalist, and manage common side effects and adverse events during and after sedation.
The Tier‐One hospitalist performing sedation in the EU/CARES is responsible for performing pre‐procedure sedation evaluation, developing a sedation plan, and delivering procedural sedation. A dedicated sedation‐trained nurse is available throughout the procedure to record vital signs, leaving the hospitalist free to monitor the patient directly, titrate sedation medications, and manage airway or adverse events as they arise. A separate provider is responsible for performing the actual procedure. The patient continues to be monitored by the sedation nurse during the recovery period, while the hospitalist remains immediately available in the unit to address any problems. At St Louis Children's Hospital, specific monitoring and documentation criteria, using standard forms and sedation scores, are strictly adhered to for every sedation, both during the sedation and throughout the recovery phase. These criteria are based upon the AAP guidelines for monitoring and management of pediatric sedation as described by Cot and Wilson.16 Hospital Medicine provides services in the EU 17 hours per day, 7 days a week. On average, 3 sedations are provided by the hospitalist per 8‐hour shift in the EU.
Tier Two
Second‐tier providers perform all services provided by Tier‐One providers, as well as expanded sedation services on the inpatient units, APC, and PAWS. Tier‐Two providers also provide on‐call services for urgent night, weekend, and holiday sedation needs.
APC and PAWS provide more specialized sedation care than that provided in the EU. PAWS is a separate and dedicated wound care unit housed on the surgical/post‐op floor where children are sedated for painful wound care, primarily burn debridement, abscess incision and drainage (I&D), and dressing changes. Sedation services are occasionally provided for other wound care issues or procedures. Both inpatients and outpatients are seen in this unit. The unit operates 10 hours per day, 7 days a week. The PAWS unit has 2 rooms specifically equipped for sedation, monitoring, and rescue, as well as 2 additional rooms for recovery or for patients not requiring sedation. Sedations in PAWS generally utilize intravenous ketamine or inhaled nitrous oxide coupled with premedication of oxycodone. Responsibilities of the hospitalist in the unit include completing and documenting the pre‐sedation evaluation, developing an appropriate sedation plan, delivering sedation medications, ongoing monitoring and documenting of vital signs throughout the case, and recovery of the patient. All nursing staff in this unit are sedation‐trained and are responsible for continued patient monitoring during the recovery period, until the patient has returned to baseline and is safe for discharge or transfer.
The APC has been described in a prior publication.15 Hospitalist sedations in the APC are performed primarily for radiology procedures, the majority of which are MRI but also include computed tomographic (CT) and nuclear medicine scans. Sedation is also provided for automated brainstem response (ABR), electromyogram (EMG), and peripherally inserted central catheter (PICC) placements. Like PAWS, APC serves both inpatients and outpatients. The APC is staffed 10 hours per day, 5 days per week. The primary sedation medications used in this unit are ketamine, dexmedetomidine, and occasionally fentanyl and midazolam. There are 11 patient beds, all equipped for patient monitoring and recovery. Hospitalists in the APC may provide direct patient care or supervise sedation‐trained nurses delivering sedation services after having a pre‐sedation evaluation performed by the hospitalist. If a sedation‐trained nurse is delivering sedation, the hospitalist may be doing other interruptible tasks, but is immediately available on the unit to respond to any concerns from the sedation nurse. All units are fully equipped with resuscitation equipment/crash carts, and an anesthesiologist is readily available to come to the unit from the OR in the event of an issue. A rapid response team that consists of a pediatric intensive care unit (ICU) fellow, respiratory therapist, and pediatric ICU charge nurse is also always on call.
Urgent sedations on the inpatient wards are common and can usually be accommodated in PAWS or APC. Rarely, however, an MRI, CT, joint aspiration, abscess drainage, or lumbar puncture must be completed urgently in the evenings, weekends, or holidays. In this situation, the hospitalist is responsible for performing the pre‐sedation evaluation, developing an appropriate sedation technique, delivering the sedation medications, monitoring and documenting during the procedure as well as throughout the recovery period, until the patient has returned to baseline. Sedation‐trained nursing staff are available to provide assistance. The sedation medications commonly used in these after‐hours sedations include ketamine for short or painful procedures and dexmedetomidine for longer radiology studies.
Training for second‐tier services consists of a minimum of 1 year of first‐tier sedation experience, a 2‐hour didactic lecture with Pediatric Anesthesia, a 1‐hour hospitalist orientation for advanced sedation providers, and 5 days of OR training with an anesthesiologist. Operating room training focuses on building skills in bag‐mask ventilation, intravenous (IV) placement, endotracheal intubation, and laryngeal mask airway (LMA) placement.
Tier Three
Third‐tier sedation providers have completed all of the training of a Tier‐Two provider and have had additional training to prepare them to deliver propofol for non‐painful procedures. Hospitalist‐delivered propofol sedation is provided exclusively in the APC for non‐painful procedures. The hospitalist is responsible for the pre‐sedation evaluation, induction and maintenance of sedation, and patient monitoring and documentation of vital signs. Monitoring for propofol sedation includes end‐tidal carbon dioxide monitoring in addition to electrocardiogram (EKG), respiratory rate (RR), pulse oximetry, and non‐invasive blood pressure (NIBP). A sedation‐trained nurse is present during induction and assists with patient positioning within the scanner. The nurse will then assume care of the patient at the completion of the procedure to continue patient monitoring during recovery.
Training for Tier‐Three providers consists of a 3‐hour didactic session with Anesthesia, 10 days of OR training, use of simulation scenarios, and a written exam. The hospitalist must then perform 25 supervised propofol sedations before being credentialed to provide propofol sedation independently. To maintain certification for Tier‐Three services, hospitalists must perform at least 75 propofol sedations every 2 years.
RESULTS
Utilizing this design and training method, we have developed a successful pediatric hospitalist sedation program. Based on fiscal year 2009 billing data, the Division of Hospital Medicine performed 2471 sedations. There were 2069 sedations performed in APC or PAWS; of those, 1017 were performed on inpatients and 1052 were performed on outpatients. Hospitalists performed 402 sedations on patients in the EU. The EU numbers are likely much larger given that, for over half the year, billing data was not collected from the EU. Unfortunately, we did not have billing data regarding night and weekend sedations, but our best estimate is 1 to 2 per week. The wait time for an outpatient sedated pediatric MRI has gone from 6 weeks to 2 days or less. As of July 2010, we have trained 90 providers at Tier One, 32 at Tier Two, and 11 at Tier Three. We currently have 43 hospitalists providing Tier‐One sedation, 18 providing Tier‐Two, and 6 providing Tier‐Three. Average cumulative hospitalist experience is 1 year for Tier One, 5 years for Tier Two, and 10 years for Tier Three.
DISCUSSION
We believe this is the first description of a pediatric hospitalist training program for a sedation service. However, it is clear that many other pediatric hospitalists are performing sedation and developing similar training programs. When starting a program such as this, there are many things to consider. First, patient volume/demand must allow for each hospitalist to perform sedations on a regular basis, both for training and Maintenance of Certification. Second, Anesthesia must be willing to provide training and oversight. Third, the hospital or university must be willing to support the cost associated with the training period. Finally, negotiating with third party payers for reimbursement is critical to financial sustainability.
The success of our program hinged upon the ability to develop a strong and collaborative relationship with Anesthesiology. Many factors played into making this relationship work. Initially, Anesthesia approached us to help them meet an unmet clinical need. Because of this, we were viewed as helpful and as problem solvers, rather than as a threat. Additionally, each division had a sedation service champion that pushed for the development of a hospitalist sedation service. Lastly, regular meetings with Anesthesia, and the intense training program itself, helped to develop a sense of collegiality between the divisions.
We have faced many challenges and learned many lessons while developing this program. There is a significant cost to training sedation providers; 47 hospitalists trained to provide Tier‐One sedation have left the program. Of those, 16 hospitalists completed training for Tier‐Two sedation, and 5 completed Tier Three. The Tier‐Two training described earlier requires approximately 50 hours of dedicated time away from other hospitalist duties, while Tier Three requires an additional 125 hours. The majority of our turnover occurred in the first few years of the program. From a financial perspective, we have had to reserve sedation training beyond Tier One to hospitalists who are able to demonstrate evidence of a long‐term commitment to our division. Every person providing Tier‐Three sedation has been with the division over 6 years. From a broader perspective, we are providing hospitalists with an important and useful skill that may enhance their careerssafe and effective sedation.
Balancing the volume of cases is another issue to consider. Our goal is to provide safe and timely sedation, therefore we need to have enough scheduled cases to maintain competency and financial viability, but we must also leave adequate flexibility in the schedule for urgent cases.
In addition to the operating room training, we are beginning to incorporate pediatric simulation as an adjunct to our training. We have designed simulation scenarios which address issues of obstruction, apnea, hypotension, bronchospasm, and aspiration. However, OR training remains a mandatory requirement for sedation training and, at times, can be challenging to schedule.
We complete a post‐sedation assessment on all patients; we are currently performing a chart review of over 1600 patients sedated with propofol, to look at the rate of planned and unplanned interventions. We believe this data will show that our training has been successful, and that with analysis of our Quality Improvement data, we can improve the safety and efficacy of our sedation program even further.
CONCLUSIONS
A pediatric hospitalist sedation service, with proper training and oversight, can successfully augment sedation services provided by anesthesiologists. As has been stated in prior publications, a defined system, and the use of a dedicated well‐trained team makes a sedation service a success.1719 A collegial and mutually respectful relationship between Anesthesia and non‐Anesthesia sedation providers is critical in developing and maintaining a successful sedation program.
- ,,,,.Anesthesia services outside of the operating room.Pediatr Nurs.2007;33(3):232,234,236–237.
- ,.Pediatric sedation/anesthesia outside the operating room.Curr Opin Anaesthesiol.2008;21(4):494–498.
- ,.Procedural sedation in the pediatric patient.Anesthesiol Clin North Am.2005;23(4):635–654, viii.
- .Pediatric sedation: can it be safely performed by non‐anesthesiologists?Curr Opin Anaesthesiol.2002;15(4):455–459.
- ,.Pediatric sedation in North American children's hospitals: a survey of anesthesia providers.Paediatr Anaesth.2005;15(3):209–213.
- ,,, et al.Safety of propofol sedation for pediatric outpatient procedures.Clin Pediatr (Phila).2009;48(8):819–823.
- ,,, et al.High dose dexmedetomidine as the sole sedative for pediatric MRI.Paediatr Anaesth.2008;18(5):403–411.
- ,.Successful implementation of a radiology sedation service staffed exclusively by pediatric emergency physicians.Pediatrics.2006;117(3):e413–e422.
- ,.Management of children undergoing painful procedures in the emergency department by non‐anesthesiologists.Isr Med Assoc J.2004;6(6):350–355.
- ,,,,.Development of a nurse‐led sedation service for paediatric magnetic resonance imaging.Lancet.1999;353(9165):1667–1671.
- ,,.Propofol sedation: intensivists' experience with 7304 cases in a children's hospital.Pediatrics.2007;120(6):e1411–e1417.
- .Risk and safety of pediatric sedation/anesthesia for procedures outside the operating room.Curr Opin Anaesthesiol.2009;22(4):509–513.
- ,,,,.The incidence and nature of adverse events during pediatric sedation/anesthesia with propofol for procedures outside the operating room: a report from the Pediatric Sedation Research Consortium.Anesth Analg.2009;108(3):795–804.
- ,,, et al.Deep sedation with propofol for upper gastrointestinal endoscopy in children, administered by specially trained pediatricians: a prospective case series with emphasis on side effects.Endoscopy.2006;38(4):368–375.
- ,,,,.A model for pediatric sedation centers using pharmacologic sedation for successful completion of radiologic and procedural studies.J Radiol Nurs2008;27(2):46–60.
- ,.Guidelines for monitoring and management of pediatric patients during and after sedation for diagnostic and therapeutic procedures: an update.Pediatrics.2006;118(6):2587–2602.
- ,.Non‐anesthesiologist‐provided pediatric procedural sedation: an update.Curr Opin Anaesthesiol.2007;20(4):365–372.
- ,,.Professional skills and competence for safe and effective procedural sedation in children: recommendations based on a systematic review of the literature.Int J Pediatr.2010; doi://10.1155/2010/934298.
- ,.Pediatric sedation outside the operating room: the year in review.Curr Opin Anaesthesiol.2005;18(4):442–446.
- ,,,,.Anesthesia services outside of the operating room.Pediatr Nurs.2007;33(3):232,234,236–237.
- ,.Pediatric sedation/anesthesia outside the operating room.Curr Opin Anaesthesiol.2008;21(4):494–498.
- ,.Procedural sedation in the pediatric patient.Anesthesiol Clin North Am.2005;23(4):635–654, viii.
- .Pediatric sedation: can it be safely performed by non‐anesthesiologists?Curr Opin Anaesthesiol.2002;15(4):455–459.
- ,.Pediatric sedation in North American children's hospitals: a survey of anesthesia providers.Paediatr Anaesth.2005;15(3):209–213.
- ,,, et al.Safety of propofol sedation for pediatric outpatient procedures.Clin Pediatr (Phila).2009;48(8):819–823.
- ,,, et al.High dose dexmedetomidine as the sole sedative for pediatric MRI.Paediatr Anaesth.2008;18(5):403–411.
- ,.Successful implementation of a radiology sedation service staffed exclusively by pediatric emergency physicians.Pediatrics.2006;117(3):e413–e422.
- ,.Management of children undergoing painful procedures in the emergency department by non‐anesthesiologists.Isr Med Assoc J.2004;6(6):350–355.
- ,,,,.Development of a nurse‐led sedation service for paediatric magnetic resonance imaging.Lancet.1999;353(9165):1667–1671.
- ,,.Propofol sedation: intensivists' experience with 7304 cases in a children's hospital.Pediatrics.2007;120(6):e1411–e1417.
- .Risk and safety of pediatric sedation/anesthesia for procedures outside the operating room.Curr Opin Anaesthesiol.2009;22(4):509–513.
- ,,,,.The incidence and nature of adverse events during pediatric sedation/anesthesia with propofol for procedures outside the operating room: a report from the Pediatric Sedation Research Consortium.Anesth Analg.2009;108(3):795–804.
- ,,, et al.Deep sedation with propofol for upper gastrointestinal endoscopy in children, administered by specially trained pediatricians: a prospective case series with emphasis on side effects.Endoscopy.2006;38(4):368–375.
- ,,,,.A model for pediatric sedation centers using pharmacologic sedation for successful completion of radiologic and procedural studies.J Radiol Nurs2008;27(2):46–60.
- ,.Guidelines for monitoring and management of pediatric patients during and after sedation for diagnostic and therapeutic procedures: an update.Pediatrics.2006;118(6):2587–2602.
- ,.Non‐anesthesiologist‐provided pediatric procedural sedation: an update.Curr Opin Anaesthesiol.2007;20(4):365–372.
- ,,.Professional skills and competence for safe and effective procedural sedation in children: recommendations based on a systematic review of the literature.Int J Pediatr.2010; doi://10.1155/2010/934298.
- ,.Pediatric sedation outside the operating room: the year in review.Curr Opin Anaesthesiol.2005;18(4):442–446.
Teamwork in Hospitals
Teamwork is important in providing high‐quality hospital care. Despite tremendous efforts in the 10 years since publication of the Institute of Medicine's To Err is Human report,1 hospitalized patients remain at risk for adverse events (AEs).2 Although many AEs are not preventable, a large portion of those which are identified as preventable can be attributed to communication and teamwork failures.35 A Joint Commission study indicated that communication failures were the root cause for two‐thirds of the 3548 sentinel events reported from 1995 to 2005.6 Another study, involving interviews of resident physicians about recent medical mishaps, found that communication failures contributed to 91% of the AEs they reported.5
Teamwork also plays an important role in other aspects of hospital care delivery. Patients' ratings of nurse‐physician coordination correlate with their overall perception of the quality of care received.7, 8 A study of Veterans Health Administration (VHA) hospitals found that teamwork culture was significantly and positively associated with overall patient satisfaction.9 Another VHA study found that hospitals with higher teamwork culture ratings had lower nurse resignations rates.10 Furthermore, poor teamwork within hospitals may have an adverse effect on financial performance, as a result of inefficiencies in physician and nurse workflow.11
Some organizations are capable of operating in complex, hazardous environments while maintaining exceptional performance over long periods of time. These high reliability organizations (HRO) include aircraft carriers, air traffic control systems, and nuclear power plants, and are characterized by their preoccupation with failure, reluctance to simplify interpretations, sensitivity to operations, commitment to resilience, and deference to expertise.12, 13 Preoccupation with failure is manifested by an organization's efforts to avoid complacency and persist in the search for additional risks. Reluctance to simplify interpretations is exemplified by an interest in pursuing a deep understanding of the issues that arise. Sensitivity to operations is the close attention paid to input from front‐line personnel and processes. Commitment to resilience relates to an organization's ability to contain errors once they occur and mitigate harm. Deference to expertise describes the practice of having authority migrate to the people with the most expertise, regardless of rank. Collectively, these qualities produce a state of mindfulness, allowing teams to anticipate and become aware of unexpected events, yet also quickly contain and learn from them. Recent publications have highlighted the need for hospitals to learn from HROs and the teams within them.14, 15
Recognizing the importance of teamwork in hospitals, senior leadership from the American College of Physician Executives (ACPE), the American Hospital Association (AHA), the American Organization of Nurse Executives (AONE), and the Society of Hospital Medicine (SHM) established the High Performance Teams and the Hospital of the Future project. This collaborative learning effort aims to redesign care delivery to provide optimal value to hospitalized patients. As an initial step, the High Performance Teams and the Hospital of the Future project team completed a literature review related to teamwork in hospitals. The purpose of this report is to summarize the current understanding of teamwork, describe interventions designed to improve teamwork, and make practical recommendations for hospitals to assess and improve teamwork‐related performance. We approach teamwork from the hospitalized patient's perspective, and restrict our discussion to interactions occurring among healthcare professionals within the hospital. We recognize the importance of teamwork at all points in the continuum of patient care. Highly functional inpatient teams should be integrated into an overall system of coordinated and collaborative care.
TEAMWORK: DEFINITION AND CONSTRUCTS
Physicians, nurses, and other healthcare professionals spend a great deal of their time on communication and coordination of care activities.1618 In spite of this and the patient safety concerns previously noted, interpersonal communication skills and teamwork have been historically underemphasized in professional training.1922 A team is defined as 2 or more individuals with specified roles interacting adaptively, interdependently, and dynamically toward a shared and common goal.23 Elements of effective teamwork have been identified through research conducted in aviation, the military, and more recently, healthcare. Salas and colleagues have synthesized this research into 5 core components: team leadership, mutual performance monitoring, backup behavior, adaptability, and team orientation (see Table 1).23 Additionally, 3 supporting and coordinating mechanisms are essential for effective teamwork: shared mental model, closed‐loop communication, and mutual trust (see Table 1).23 High‐performing teams use these elements to develop a culture for speaking up, and situational awareness among team members. Situational awareness refers to a person's perception and understanding of their dynamic environment, and human errors often result from a lack of such awareness.24 These teamwork constructs provide the foundational basis for understanding how hospitals can identify teamwork challenges, assess team performance, and design effective interventions.
| Teamwork | Definition | Behavioral Examples |
|---|---|---|
| ||
| Component | ||
| Team leadership | The leader directs and coordinates team members activities | Facilitate team problem solving; |
| Provide performance expectations; | ||
| Clarify team member roles; | ||
| Assist in conflict resolution | ||
| Mutual performance monitoring | Team members are able to monitor one another's performance | Identify mistakes and lapses in other team member actions; |
| Provide feedback to fellow team members to facilitate self‐correction | ||
| Backup behavior | Team members anticipate and respond to one another's needs | Recognize workload distribution problem; |
| Shift work responsibilities to underutilized members | ||
| Adaptability | The team adjusts strategies based on new information | Identify cues that change has occurred and develop plan to deal with changes; |
| Remain vigilant to change in internal and external environment | ||
| Team orientation | Team members prioritize team goals above individual goals | Take into account alternate solutions by teammates; |
| Increased task involvement, information sharing, and participatory goal setting | ||
| Coordinating mechanism | ||
| Shared mental model | An organizing knowledge of the task of the team and how members will interact to achieve their goal | Anticipate and predict each other's needs; |
| Identify changes in team, task, or teammates | ||
| Closed‐loop communication | Acknowledgement and confirmation of information received | Follow up with team members to ensure message received; |
| Acknowledge that message was received; | ||
| Clarify information received | ||
| Mutual trust | Shared belief that team members will perform their roles | Share information; |
| Willingly admit mistakes and accept feedback | ||
CHALLENGES TO EFFECTIVE TEAMWORK
Several important and unique barriers to teamwork exist in hospitals. Teams are large and formed in an ad hoc fashion. On a given day, a patient's hospital team might include a hospitalist, a nurse, a case manager, a pharmacist, and 1 or more consulting physicians and therapists. Team members in each respective discipline care for multiple patients at the same time, yet few hospitals align team membership (ie, patient assignment). Therefore, a nurse caring for 4 patients may interact with 4 different hospitalists. Similarly, a hospitalist caring for 14 patients may interact with multiple nurses in a given day. Team membership is ever changing because hospital professionals work in shifts and rotations. Finally, team members are seldom in the same place at the same time because physicians often care for patients on multiple units and floors, while nurses and other team members are often unit‐based. Salas and others have noted that team size, instability, and geographic dispersion of membership serve as important barriers to improving teamwork.25, 26 As a result of these barriers, nurses and physicians do not communicate consistently, and often disagree on the daily plan of care for their patients.27, 28 When communication does occur, clinicians may overestimate how well their messages are understood by other team members, reflecting a phenomenon well known in communication psychology related to egocentric thought processes.29, 30
The traditionally steep hierarchy within medicine may also serve as a barrier to teamwork. Studies in intensive care units (ICUs), operating rooms, and general medical units reveal widely discrepant views on the quality of collaboration and communication between healthcare professionals.3133 Although physicians generally give high ratings to the quality of collaboration with nurses, nurses consistently rate the quality of collaboration with physicians as poor. Similarly, specialist physicians rate collaboration with hospitalists higher than hospitalists rate collaboration with specialists.33 Effective teams in other high‐risk industries, like aviation, strive to flatten hierarchy so that team members feel comfortable raising concerns and engaging in open and respectful communications.34
The effect of technology on communication practices and teamwork is complex and incompletely understood. The implementation of electronic heath records and computerized provider order entry systems fundamentally changes work‐flow, and may result in less synchronization and feedback during nurse‐physician collaboration.35 Similarly, the expanded use of text messages delivered via alphanumeric paging or mobile phone results in a transition toward asynchronous modes of communication. These asynchronous modes allow healthcare professionals to review and respond to messages at their convenience, and may reduce unnecessary interruptions. Research shows that these systems are popular among clinicians.3638 However, receipt and understanding of the intended message may not be confirmed with the use of asynchronous modes of communication. Moreover, important face‐to‐face communication elements (tone of voice, expression, gesture, eye contract)39, 40 are lacking. One promising approach is a system which sends low‐priority messages to a Web‐based task list for periodic review, while allowing higher priority messages to pass through to an alphanumeric pager and interrupt the intended recipient.41 Another common frustration in hospitals, despite advancing technology, is difficulty identifying the correct physician(s) and nurse(s) caring for a particular patient at a given point in time.33 Wong and colleagues found that 14% of pages in their hospital were initially sent to the wrong physician.42
ASSESSMENT OF TEAMWORK
One of the challenges in improving teamwork is the difficulty in measuring it. Teamwork assessment entails measuring the performance of teams composed of multiple individuals. Methods of teamwork assessment can be broadly categorized as self assessment, peer assessment, direct observation, survey of team climate or culture, and measurement of the outcome of effective teamwork. While self‐report tools are easy to administer and can capture affective components influencing team performance, they may not reflect actual skills on the part of individuals or teams. Peer assessment includes the use of 360‐degree evaluations or multisource feedback, and provides an evaluation of individual performance.4347
Direct observation provides a more accurate assessment of team‐related behaviors using trained observers. Observers use checklists and/or behaviorally anchored rating scales (BARS) to evaluate individual and team performance. A number of BARS have been developed and validated for the evaluation of team performance.4852 Of note, direct observation may be difficult in settings in which team members are not in the same place at the same time. An alternative method, which may be better suited for general medical units, is the use of survey instruments designed to assess attitudes and teamwork climate.5355 Importantly, higher survey ratings of collaboration and teamwork have been associated with better patient outcomes in observational studies.5658
The ultimate goal of teamwork efforts is to improve patient outcomes. Because patient outcomes are affected by a number of factors and because hospitals frequently engage in multiple, simultaneous efforts to improve care, it is often difficult to clearly link improved outcomes with teamwork interventions. Continued efforts to rigorously evaluate teamwork interventions should remain a priority, particularly as the cost of these interventions must be weighed against other interventions and investments.
EXAMPLES OF SUCCESSFUL INTERVENTIONS
A number of interventions have been used to improve teamwork in hospitals (see Table 2).
| Intervention | Advantages | Disadvantages |
|---|---|---|
| Localization of physicians | Increases frequency of nurse‐physician communication; provides foundation for additional interventions | Insufficient in creating a shared mental model; does not specifically enhance communication skills |
| Daily goals‐of‐care forms and checklists | Provides structure to interdisciplinary discussions and ensures input from all team members | May be completed in a perfunctory manner and may not be updated as plans of care evolve |
| Teamwork training | Emphasizes improved communication behaviors relevant across a range of team member interactions | Requires time and deliberate practice of new skills; effect may be attenuated if members are dispersed. |
| Interdisciplinary rounds | Provides a forum for regular interdisciplinary communication | Requires leadership to organize discussion and does not address need for updates as plans of care evolve |
Geographic Localization of Physicians
As mentioned earlier, physicians in large hospitals may care for patients on multiple units or floors. Designating certain physicians to care for patients admitted to specific units may improve efficiency and communication among healthcare professionals. One study recently reported on the effect of localization of hospital physicians to specific patient care units. Localization resulted in an increase in the rate of nurse‐physician communication, but did not improve providers' shared understanding of the plan of care.56 Notably, localizing physicians may improve the feasibility of additional interventions, like teamwork training and interdisciplinary rounds.
Daily Goals of Care and Surgery Safety Checklists
In ICU and operating room settings, physicians and nurses work in proximity, allowing interdisciplinary discussions to occur at the bedside. The finding that professionals in ICUs and operating rooms have widely discrepant views on the quality of collaboration31, 32 indicates that proximity, alone, is not sufficient for effective communication. Pronovost et al. used a daily goals form for bedside ICU rounds in an effort to standardize communication about the daily plan of care.57 The form defined essential goals of care for patients, and its use resulted in a significant improvement in the team's understanding of the daily goals. Narasimhan et al. performed a similar study using a daily goals worksheet during ICU rounds,58 and also found a significant improvement in physicians' and nurses' ratings of their understanding of the goals of care. The forms used in these studies provided structure to the interdisciplinary conversations during rounds to create a shared understanding of patients' plans of care.
Haynes and colleagues recently reported on the use of a surgical safety checklist in a large, multicenter pre‐post study.59 The checklist consisted of verbal confirmation of the completion of basic steps essential to safe care in the operating room, and provided structure to communication among surgical team members to ensure a shared understanding of the operative plan. The intervention resulted in a significant reduction in inpatient complications and mortality.
Team Training
Formalized team training, based on crew resource management, has been studied as a potential method to improve teamwork in a variety of medical settings.6062 Training emphasizes the core components of successful teamwork and essential coordinating mechanisms previously mentioned.23 Team training appears to positively influence culture, as assessed by teamwork and patient safety climate survey instruments.60 Based on these findings and extensive research demonstrating the success of teamwork training in aviation,63 the Agency for Healthcare Research and Quality (AHRQ) and the Department of Defense (DoD) have partnered in offering the Team Strategies and Tools to Enhance Performance and Patient Safety (TeamSTEPPS) program, designed to improve teamwork skills for healthcare professionals.64, 65
Only a handful of studies have evaluated the effectiveness of teamwork training programs on patient outcomes, and the results are mixed.66 Morey et al. found a reduction in the rate of observed errors as a result of teamwork training in emergency departments, but observers in the study were not blinded with regard to whether teams had undergone training.61 A research group in the United Kingdom evaluated the benefit of simulation‐based team training on outcomes in an obstetrical setting.67, 68 Training included management of specific complications, including shoulder dystocia and uterine cord prolapse. Using retrospective chart review, the investigators found a significant reduction in the proportion of babies born with an obstetric brachial palsy injury and a reduction in the time from diagnosis of uterine cord prolapse to infant delivery. Nielsen and colleagues also evaluated the use of teamwork training in an obstetric setting.62 In a cluster randomized controlled trial, the investigators found no reduction in the rate of adverse outcomes. Differences in the duration of teamwork training and the degree of emphasis on deliberate practice of new skills (eg, with the use of simulation‐based training) likely explains the lack of consistent results.
Very little research has evaluated teamwork training in the general medical environment.69, 70 Sehgal and colleagues recently published an evaluation of the effect of teamwork training delivered to internal medicine residents, hospitalists, nurses, pharmacists, case managers, and social workers on medical services in 3 Northern California hospitals.69 The 4‐hour training sessions covered topical areas of safety culture, teamwork, and communication through didactics, videos, facilitated discussions, and small group role plays to practice new skills and behaviors. The intervention was rated highly among participants,69 and the training along with subsequent follow‐up interventions resulted in improved patient perceptions of teamwork and communication but had no impact on key patient outcomes.71
Interdisciplinary Rounds
Interdisciplinary rounds (IDR) have been used for many years as a means to assemble team members in a single location,7275 and the use of IDR has been associated with lower mortality among ICU patients.76 Interdisciplinary rounds may be particularly useful for clinical settings in which team members are traditionally dispersed in time and place, such as medical‐surgical units. Recent studies have evaluated the effect of structured inter‐disciplinary rounds (SIDR),77, 78 which combine a structured format for communication, similar to a daily goals‐of‐care form, with a forum for daily interdisciplinary meetings. Though no effect was seen on length of stay or cost, SIDR resulted in significantly higher ratings of the quality of collaboration and teamwork climate, and a reduction in the rate of AEs.79 Importantly, the majority of clinicians in the studies agreed that SIDR improved the efficiency of their work day, and expressed a desire that SIDR continue indefinitely. Many investigators have emphasized the importance of leadership during IDR, often by a medical director, nurse manager, or both.74, 77, 78
Summary of Interventions to Improve Teamwork
Localization of physicians increases the frequency of nurse‐physician communication, but is insufficient in creating a shared understanding of patients' plans of care. Providing structure for the discussion among team members (eg, daily goals of care forms and checklists) ensures that critical elements of the plan of care are communicated. Teamwork training is based upon a strong foundation of research both inside and outside of healthcare, and has demonstrated improved knowledge of teamwork principles, attitudes about the importance of teamwork, and overall safety climate. Creating a forum for team members to assemble and discuss their patients (eg, IDR) can overcome some of the unique barriers to collaboration in settings where members are dispersed in time and space. Leaders wishing to improve interdisciplinary teamwork should consider implementing a combination of complementary interventions. For example, localization may increase the frequency of team member interactions, the quality of which may be enhanced with teamwork training and reinforced with the use of structured communication tools and IDR. Future research should evaluate the effect of these combined interventions.
CONCLUSIONS
In summary, teamwork is critically important to provide safe and effective care. Important and unique barriers to teamwork exist in hospitals. We recommend the use of survey instruments, such as those mentioned earlier, as the most feasible method to assess teamwork in the general medical setting. Because each intervention addresses only a portion of the barriers to optimal teamwork, we encourage leaders to use a multifaceted approach. We recommend the implementation of a combination of interventions with adaptations to fit unique clinical settings and local culture.
Acknowledgements
This manuscript was prepared as part of the High Performance Teams and the Hospital of the Future project, a collaborative effort including senior leadership from the American College of Physician Executives, the American Hospital Association, the American Organization of Nurse Executives, and the Society of Hospital Medicine. The authors thank Taylor Marsh for her administrative support and help in coordinating project meetings.
- To Err Is Human: Building a Safer Health System.Washington, DC:Institute of Medicine;1999.
- ,,,,,.Temporal trends in rates of patient harm resulting from medical care.N Engl J Med.2010;363(22):2124–2134.
- ,,.Exploring the causes of adverse events in NHS hospital practice.J R Soc Med.2001;94(7):322–330.
- ,,,,,.The Quality in Australian Health Care Study.Med J Aust.1995;163(9):458–471.
- ,,.Communication failures: an insidious contributor to medical mishaps.Acad Med.2004;79(2):186–194.
- Improving America's Hospitals: The Joint Commission's Annual Report on Quality and Safety 2007. Available at: http://www.jointcommissionreport.org. Accessed November2007.
- ,,.Patient perceptions of coordinated care: the importance of organized communication in hospitals.J Healthc Qual.1999;21(5):18–23.
- ,,.Am I safe here? Improving patients' perceptions of safety in hospitals.J Nurs Care Qual.2006;21(1):30–40.
- ,,.Teamwork culture and patient satisfaction in hospitals.Med Care.2004;42(5):492–498.
- ,,.The influence of teamwork culture on physician and nurse resignation rates in hospitals.Health Serv Manage Res.2008;21(1):23–31.
- ,,.Quantifying the economic impact of communication inefficiencies in U.S. hospitals.J Healthc Manag.2010;55(4):265–282.
- ,.Managing the Unexpected: Assuring High Performance in an Age of Complexity.San Francisco, CA:Jossey‐Bass;2001.
- .Some characteristics of high reliability organizations.Organization Science.1990;1(2):160–177.
- ,,.Teamwork as an essential component of high‐reliability organizations.Health Serv Res.2006;41(4 pt 2):1576–1598.
- ,,,.Promoting health care safety through training high reliability teams.Qual Saf Health Care.2005;14(4):303–309.
- ,,,,,.Analyzing the time and value of housestaff inpatient work.J Gen Intern Med.1998;13(8):534–540.
- ,,, et al.Quantifying nursing workflow in medication administration.J Nurs Adm.2008;38(1):19–26.
- ,,.How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- ,,,,.Redesigning residency education in internal medicine: a position paper from the Association of Program Directors in Internal Medicine.Ann Intern Med.2006;144(12):920–926.
- ,,,.Hospitalists' perceptions of their residency training needs: results of a national survey.Am J Med.2001;111(3):247–254.
- ,,.Redesigning training for internal medicine.Ann Intern Med.2006;144(12):927–932.
- ,,,,.The role of teamwork in the professional education of physicians: current status and assessment recommendations.Jt Comm J Qual Patient Saf.2005;31(4):185–202.
- ,,.Is there a “big five” in teamwork?Small Group Research.2005;36:555–599.
- ,,.Objective measures of situation awareness in a simulated medical environment.Qual Saf Health Care.2004;13(suppl 1):i65–i71.
- ,.What do we know about health care team effectiveness? A review of the literature.Med Care Res Rev.2006;63(3):263–300.
- ,,, et al.Does team training improve team performance? A meta‐analysis.Hum Factors.2008;50(6):903–933.
- ,,,,,.Can we talk? Priorities for patient care differed among health care providers. AHRQ Publication No. 05–0021‐1.Rockville, MD:Agency for Healthcare Research and Quality;2005.
- ,,, et al.Patterns of nurse–physicians communication and agreement on the plan of care.Qual Saf Health Care.2010;19:195–199.
- ,,,,.Interns overestimate the effectiveness of their hand‐off communication.Pediatrics.2010;125(3):491–496.
- ,.Speakers' overestimation of their effectiveness.Psychol Sci.2002;13(3):207–212.
- ,,, et al.Operating room teamwork among physicians and nurses: teamwork in the eye of the beholder.J Am Coll Surg.2006;202(5):746–752.
- ,,.Discrepant attitudes about teamwork among critical care nurses and physicians.Crit Care Med.2003;31(3):956–959.
- ,,,,,.Teamwork on inpatient medical units: assessing attitudes and barriers.Qual Saf Health Care.2010;19(2):117–121.
- ,,.Error, stress, and teamwork in medicine and aviation: cross sectional surveys.BMJ.2000;320(7237):745–749.
- ,,,,.Impact of a computerized physician order entry system on nurse‐physician collaboration in the medication process.Int J Med Inform.2008;77(11):735–744.
- ,,,,.Alphanumeric paging in an academic hospital setting.Am J Surg.2006;191(4):561–565.
- ,,,.Implementation and evaluation of an alpha‐numeric paging system on a resident inpatient teaching service.J Hosp Med.2009;4(8):E34–E40.
- ,,, et al.The use of smartphones for clinical communication on internal medicine wards.J Hosp Med.2010;5(9):553–559.
- ,.Organizational information requirements, media richness, and structural design.Management Science.1986;32(5):554–571.
- ,.Decoding of inconsistent communications of personality and social psychology.J Pers Soc Psychol.1967;6(1):109–114.
- ,,,,.Beyond paging: building a Web‐based communication tool for nurses and physicians.J Gen Intern Med.2009;24(1):105–110.
- ,,, et al.Frequency and clinical importance of pages sent to the wrong physician.Arch Intern Med.2009;169(11):1072–1073.
- ,,, et al.Evaluation of resident communication skills and professionalism: a matter of perspective?Pediatrics.2006;118(4):1371–1379.
- ,,, et al.Effect of multisource feedback on resident communication skills and professionalism: a randomized controlled trial.Arch Pediatr Adolesc Med.2007;161(1):44–49.
- .Multisource feedback in the assessment of physician competencies.J Contin Educ Health Prof.2003;23(1):4–12.
- ,.Reliability of a 360‐degree evaluation to assess resident competence.Am J Phys Med Rehabil.2007;86(10):845–852.
- ,,,.Pilot study of a 360‐degree assessment instrument for physical medicine 82(5):394–402.
- ,,,,,.Anaesthetists' Non‐Technical Skills (ANTS): evaluation of a behavioural marker system.Br J Anaesth.2003;90(5):580–588.
- ,,,.Using the Communication and Teamwork Skills (CATS) Assessment to measure health care team performance.Jt Comm J Qual Patient Saf.2007;33(9):549–558.
- ,,, et al.The Mayo High Performance Teamwork Scale: reliability and validity for evaluating key crew resource management skills.Simul Healthc.2007;2(1):4–10.
- ,,,,,.Reliability of a revised NOTECHS scale for use in surgical teams.Am J Surg.2008;196(2):184–190.
- ,,,,,.Observational teamwork assessment for surgery: construct validation with expert versus novice raters.Ann Surg.2009;249(6):1047–1051.
- ,,, et al.The Safety Attitudes Questionnaire: psychometric properties, benchmarking data, and emerging research.BMC Health Serv Res.2006;6:44.
- .Development of an instrument to measure collaboration and satisfaction about care decisions.J Adv Nurs.1994;20(1):176–182.
- ,,,,,.Psychometric properties of an attitude scale measuring physician‐nurse collaboration.Eval Health Prof.1999;22(2):208–220.
- ,,, et al.Impact of localizing physicians to hospital units on nurse‐physician communication and agreement on the plan of care.J Gen Intern Med.2009;24(11):1223–1227.
- ,,,,,.Improving communication in the ICU using daily goals.J Crit Care.2003;18(2):71–75.
- ,,,,.Improving nurse‐physician communication and satisfaction in the intensive care unit with a daily goals worksheet.Am J Crit Care.2006;15(2):217–222.
- ,,, et al.A surgical safety checklist to reduce morbidity and mortality in a global population.N Engl J Med.2009;360(5):491–499.
- ,,, et al.Effect of crew resource management training in a multidisciplinary obstetrical setting.Int J Qual Health Care.2008;20(4):254–263.
- ,,, et al.Error reduction and performance improvement in the emergency department through formal teamwork training: evaluation results of the MedTeams project.Health Serv Res.2002;37(6):1553–1581.
- ,,, et al.Effects of teamwork training on adverse outcomes and process of care in labor and delivery: a randomized controlled trial.Obstet Gynecol.2007;109(1):48–55.
- ,,,,.Medical Teamwork and Patient Safety: The Evidence‐Based Relation.Rockville, MD:Agency for Healthcare Research and Quality;2005.
- Agency for Healthcare Research and Quality. TeamSTEPPS Home. Available at: http://teamstepps.ahrq.gov/index.htm. Accessed January 18,2010.
- ,.TeamSTEPPS: assuring optimal teamwork in clinical settings.Am J Med Qual.2007;22(3):214–217.
- ,,,.Does crew resource management training work? An update, an extension, and some critical needs.Hum Factors.2006;48(2):392–412.
- ,,, et al.Improving neonatal outcome through practical shoulder dystocia training.Obstet Gynecol.2008;112(1):14–20.
- ,,, et al.Retrospective cohort study of diagnosis‐delivery interval with umbilical cord prolapse: the effect of team training.Br J Obstet Gynaecol.2009;116(8):1089–1096.
- ,,, et al.A multidisciplinary teamwork training program: the Triad for Optimal Patient Safety (TOPS) experience.J Gen Intern Med.2008;23(12):2053–2057.
- ,,,,.Teambuilding and leadership training in an internal medicine residency training program.J Gen Intern Med.2004;19(6):692–697.
- ,,, et al. Effects of a multicenter teamwork and communication program on patient outcomes: results from the Triad for Optimal Patient Safety (TOPS) project. In press.
- ,,, et al.The effect of a multidisciplinary hospitalist/physician and advanced practice nurse collaboration on hospital costs.J Nurs Adm.2006;36(2):79–85.
- ,,.A firm trial of interdisciplinary rounds on the inpatient medical wards: an intervention designed using continuous quality improvement.Med Care.1998;36(8 suppl):AS4–A12.
- ,,,,.Use of multidisciplinary rounds to simultaneously improve quality outcomes, enhance resident education, and shorten length of stay.J Gen Intern Med.2007;22(8):1073–1079.
- ,,,.Effect of a multidisciplinary intervention on communication and collaboration among physicians and nurses.Am J Crit Care.2005;14(1):71–77.
- ,,,,.The effect of multidisciplinary care teams on intensive care unit mortality.Arch Intern Med.2010;170(4):369–376.
- ,,,,,.Improving teamwork: impact of structured interdisciplinary rounds on a hospitalist unit.J Hosp Med.2011;6(2):88–93.
- ,,,,,.Improving teamwork: impact of structured interdisciplinary rounds on a medical teaching unit.J Gen Intern Med.2010;25(8):826–832.
- ,,, et al.Structured interdisciplinary rounds in a medical teaching unit: improving patient safety.Arch Intern Med.2011;171(7):678–684.
Teamwork is important in providing high‐quality hospital care. Despite tremendous efforts in the 10 years since publication of the Institute of Medicine's To Err is Human report,1 hospitalized patients remain at risk for adverse events (AEs).2 Although many AEs are not preventable, a large portion of those which are identified as preventable can be attributed to communication and teamwork failures.35 A Joint Commission study indicated that communication failures were the root cause for two‐thirds of the 3548 sentinel events reported from 1995 to 2005.6 Another study, involving interviews of resident physicians about recent medical mishaps, found that communication failures contributed to 91% of the AEs they reported.5
Teamwork also plays an important role in other aspects of hospital care delivery. Patients' ratings of nurse‐physician coordination correlate with their overall perception of the quality of care received.7, 8 A study of Veterans Health Administration (VHA) hospitals found that teamwork culture was significantly and positively associated with overall patient satisfaction.9 Another VHA study found that hospitals with higher teamwork culture ratings had lower nurse resignations rates.10 Furthermore, poor teamwork within hospitals may have an adverse effect on financial performance, as a result of inefficiencies in physician and nurse workflow.11
Some organizations are capable of operating in complex, hazardous environments while maintaining exceptional performance over long periods of time. These high reliability organizations (HRO) include aircraft carriers, air traffic control systems, and nuclear power plants, and are characterized by their preoccupation with failure, reluctance to simplify interpretations, sensitivity to operations, commitment to resilience, and deference to expertise.12, 13 Preoccupation with failure is manifested by an organization's efforts to avoid complacency and persist in the search for additional risks. Reluctance to simplify interpretations is exemplified by an interest in pursuing a deep understanding of the issues that arise. Sensitivity to operations is the close attention paid to input from front‐line personnel and processes. Commitment to resilience relates to an organization's ability to contain errors once they occur and mitigate harm. Deference to expertise describes the practice of having authority migrate to the people with the most expertise, regardless of rank. Collectively, these qualities produce a state of mindfulness, allowing teams to anticipate and become aware of unexpected events, yet also quickly contain and learn from them. Recent publications have highlighted the need for hospitals to learn from HROs and the teams within them.14, 15
Recognizing the importance of teamwork in hospitals, senior leadership from the American College of Physician Executives (ACPE), the American Hospital Association (AHA), the American Organization of Nurse Executives (AONE), and the Society of Hospital Medicine (SHM) established the High Performance Teams and the Hospital of the Future project. This collaborative learning effort aims to redesign care delivery to provide optimal value to hospitalized patients. As an initial step, the High Performance Teams and the Hospital of the Future project team completed a literature review related to teamwork in hospitals. The purpose of this report is to summarize the current understanding of teamwork, describe interventions designed to improve teamwork, and make practical recommendations for hospitals to assess and improve teamwork‐related performance. We approach teamwork from the hospitalized patient's perspective, and restrict our discussion to interactions occurring among healthcare professionals within the hospital. We recognize the importance of teamwork at all points in the continuum of patient care. Highly functional inpatient teams should be integrated into an overall system of coordinated and collaborative care.
TEAMWORK: DEFINITION AND CONSTRUCTS
Physicians, nurses, and other healthcare professionals spend a great deal of their time on communication and coordination of care activities.1618 In spite of this and the patient safety concerns previously noted, interpersonal communication skills and teamwork have been historically underemphasized in professional training.1922 A team is defined as 2 or more individuals with specified roles interacting adaptively, interdependently, and dynamically toward a shared and common goal.23 Elements of effective teamwork have been identified through research conducted in aviation, the military, and more recently, healthcare. Salas and colleagues have synthesized this research into 5 core components: team leadership, mutual performance monitoring, backup behavior, adaptability, and team orientation (see Table 1).23 Additionally, 3 supporting and coordinating mechanisms are essential for effective teamwork: shared mental model, closed‐loop communication, and mutual trust (see Table 1).23 High‐performing teams use these elements to develop a culture for speaking up, and situational awareness among team members. Situational awareness refers to a person's perception and understanding of their dynamic environment, and human errors often result from a lack of such awareness.24 These teamwork constructs provide the foundational basis for understanding how hospitals can identify teamwork challenges, assess team performance, and design effective interventions.
| Teamwork | Definition | Behavioral Examples |
|---|---|---|
| ||
| Component | ||
| Team leadership | The leader directs and coordinates team members activities | Facilitate team problem solving; |
| Provide performance expectations; | ||
| Clarify team member roles; | ||
| Assist in conflict resolution | ||
| Mutual performance monitoring | Team members are able to monitor one another's performance | Identify mistakes and lapses in other team member actions; |
| Provide feedback to fellow team members to facilitate self‐correction | ||
| Backup behavior | Team members anticipate and respond to one another's needs | Recognize workload distribution problem; |
| Shift work responsibilities to underutilized members | ||
| Adaptability | The team adjusts strategies based on new information | Identify cues that change has occurred and develop plan to deal with changes; |
| Remain vigilant to change in internal and external environment | ||
| Team orientation | Team members prioritize team goals above individual goals | Take into account alternate solutions by teammates; |
| Increased task involvement, information sharing, and participatory goal setting | ||
| Coordinating mechanism | ||
| Shared mental model | An organizing knowledge of the task of the team and how members will interact to achieve their goal | Anticipate and predict each other's needs; |
| Identify changes in team, task, or teammates | ||
| Closed‐loop communication | Acknowledgement and confirmation of information received | Follow up with team members to ensure message received; |
| Acknowledge that message was received; | ||
| Clarify information received | ||
| Mutual trust | Shared belief that team members will perform their roles | Share information; |
| Willingly admit mistakes and accept feedback | ||
CHALLENGES TO EFFECTIVE TEAMWORK
Several important and unique barriers to teamwork exist in hospitals. Teams are large and formed in an ad hoc fashion. On a given day, a patient's hospital team might include a hospitalist, a nurse, a case manager, a pharmacist, and 1 or more consulting physicians and therapists. Team members in each respective discipline care for multiple patients at the same time, yet few hospitals align team membership (ie, patient assignment). Therefore, a nurse caring for 4 patients may interact with 4 different hospitalists. Similarly, a hospitalist caring for 14 patients may interact with multiple nurses in a given day. Team membership is ever changing because hospital professionals work in shifts and rotations. Finally, team members are seldom in the same place at the same time because physicians often care for patients on multiple units and floors, while nurses and other team members are often unit‐based. Salas and others have noted that team size, instability, and geographic dispersion of membership serve as important barriers to improving teamwork.25, 26 As a result of these barriers, nurses and physicians do not communicate consistently, and often disagree on the daily plan of care for their patients.27, 28 When communication does occur, clinicians may overestimate how well their messages are understood by other team members, reflecting a phenomenon well known in communication psychology related to egocentric thought processes.29, 30
The traditionally steep hierarchy within medicine may also serve as a barrier to teamwork. Studies in intensive care units (ICUs), operating rooms, and general medical units reveal widely discrepant views on the quality of collaboration and communication between healthcare professionals.3133 Although physicians generally give high ratings to the quality of collaboration with nurses, nurses consistently rate the quality of collaboration with physicians as poor. Similarly, specialist physicians rate collaboration with hospitalists higher than hospitalists rate collaboration with specialists.33 Effective teams in other high‐risk industries, like aviation, strive to flatten hierarchy so that team members feel comfortable raising concerns and engaging in open and respectful communications.34
The effect of technology on communication practices and teamwork is complex and incompletely understood. The implementation of electronic heath records and computerized provider order entry systems fundamentally changes work‐flow, and may result in less synchronization and feedback during nurse‐physician collaboration.35 Similarly, the expanded use of text messages delivered via alphanumeric paging or mobile phone results in a transition toward asynchronous modes of communication. These asynchronous modes allow healthcare professionals to review and respond to messages at their convenience, and may reduce unnecessary interruptions. Research shows that these systems are popular among clinicians.3638 However, receipt and understanding of the intended message may not be confirmed with the use of asynchronous modes of communication. Moreover, important face‐to‐face communication elements (tone of voice, expression, gesture, eye contract)39, 40 are lacking. One promising approach is a system which sends low‐priority messages to a Web‐based task list for periodic review, while allowing higher priority messages to pass through to an alphanumeric pager and interrupt the intended recipient.41 Another common frustration in hospitals, despite advancing technology, is difficulty identifying the correct physician(s) and nurse(s) caring for a particular patient at a given point in time.33 Wong and colleagues found that 14% of pages in their hospital were initially sent to the wrong physician.42
ASSESSMENT OF TEAMWORK
One of the challenges in improving teamwork is the difficulty in measuring it. Teamwork assessment entails measuring the performance of teams composed of multiple individuals. Methods of teamwork assessment can be broadly categorized as self assessment, peer assessment, direct observation, survey of team climate or culture, and measurement of the outcome of effective teamwork. While self‐report tools are easy to administer and can capture affective components influencing team performance, they may not reflect actual skills on the part of individuals or teams. Peer assessment includes the use of 360‐degree evaluations or multisource feedback, and provides an evaluation of individual performance.4347
Direct observation provides a more accurate assessment of team‐related behaviors using trained observers. Observers use checklists and/or behaviorally anchored rating scales (BARS) to evaluate individual and team performance. A number of BARS have been developed and validated for the evaluation of team performance.4852 Of note, direct observation may be difficult in settings in which team members are not in the same place at the same time. An alternative method, which may be better suited for general medical units, is the use of survey instruments designed to assess attitudes and teamwork climate.5355 Importantly, higher survey ratings of collaboration and teamwork have been associated with better patient outcomes in observational studies.5658
The ultimate goal of teamwork efforts is to improve patient outcomes. Because patient outcomes are affected by a number of factors and because hospitals frequently engage in multiple, simultaneous efforts to improve care, it is often difficult to clearly link improved outcomes with teamwork interventions. Continued efforts to rigorously evaluate teamwork interventions should remain a priority, particularly as the cost of these interventions must be weighed against other interventions and investments.
EXAMPLES OF SUCCESSFUL INTERVENTIONS
A number of interventions have been used to improve teamwork in hospitals (see Table 2).
| Intervention | Advantages | Disadvantages |
|---|---|---|
| Localization of physicians | Increases frequency of nurse‐physician communication; provides foundation for additional interventions | Insufficient in creating a shared mental model; does not specifically enhance communication skills |
| Daily goals‐of‐care forms and checklists | Provides structure to interdisciplinary discussions and ensures input from all team members | May be completed in a perfunctory manner and may not be updated as plans of care evolve |
| Teamwork training | Emphasizes improved communication behaviors relevant across a range of team member interactions | Requires time and deliberate practice of new skills; effect may be attenuated if members are dispersed. |
| Interdisciplinary rounds | Provides a forum for regular interdisciplinary communication | Requires leadership to organize discussion and does not address need for updates as plans of care evolve |
Geographic Localization of Physicians
As mentioned earlier, physicians in large hospitals may care for patients on multiple units or floors. Designating certain physicians to care for patients admitted to specific units may improve efficiency and communication among healthcare professionals. One study recently reported on the effect of localization of hospital physicians to specific patient care units. Localization resulted in an increase in the rate of nurse‐physician communication, but did not improve providers' shared understanding of the plan of care.56 Notably, localizing physicians may improve the feasibility of additional interventions, like teamwork training and interdisciplinary rounds.
Daily Goals of Care and Surgery Safety Checklists
In ICU and operating room settings, physicians and nurses work in proximity, allowing interdisciplinary discussions to occur at the bedside. The finding that professionals in ICUs and operating rooms have widely discrepant views on the quality of collaboration31, 32 indicates that proximity, alone, is not sufficient for effective communication. Pronovost et al. used a daily goals form for bedside ICU rounds in an effort to standardize communication about the daily plan of care.57 The form defined essential goals of care for patients, and its use resulted in a significant improvement in the team's understanding of the daily goals. Narasimhan et al. performed a similar study using a daily goals worksheet during ICU rounds,58 and also found a significant improvement in physicians' and nurses' ratings of their understanding of the goals of care. The forms used in these studies provided structure to the interdisciplinary conversations during rounds to create a shared understanding of patients' plans of care.
Haynes and colleagues recently reported on the use of a surgical safety checklist in a large, multicenter pre‐post study.59 The checklist consisted of verbal confirmation of the completion of basic steps essential to safe care in the operating room, and provided structure to communication among surgical team members to ensure a shared understanding of the operative plan. The intervention resulted in a significant reduction in inpatient complications and mortality.
Team Training
Formalized team training, based on crew resource management, has been studied as a potential method to improve teamwork in a variety of medical settings.6062 Training emphasizes the core components of successful teamwork and essential coordinating mechanisms previously mentioned.23 Team training appears to positively influence culture, as assessed by teamwork and patient safety climate survey instruments.60 Based on these findings and extensive research demonstrating the success of teamwork training in aviation,63 the Agency for Healthcare Research and Quality (AHRQ) and the Department of Defense (DoD) have partnered in offering the Team Strategies and Tools to Enhance Performance and Patient Safety (TeamSTEPPS) program, designed to improve teamwork skills for healthcare professionals.64, 65
Only a handful of studies have evaluated the effectiveness of teamwork training programs on patient outcomes, and the results are mixed.66 Morey et al. found a reduction in the rate of observed errors as a result of teamwork training in emergency departments, but observers in the study were not blinded with regard to whether teams had undergone training.61 A research group in the United Kingdom evaluated the benefit of simulation‐based team training on outcomes in an obstetrical setting.67, 68 Training included management of specific complications, including shoulder dystocia and uterine cord prolapse. Using retrospective chart review, the investigators found a significant reduction in the proportion of babies born with an obstetric brachial palsy injury and a reduction in the time from diagnosis of uterine cord prolapse to infant delivery. Nielsen and colleagues also evaluated the use of teamwork training in an obstetric setting.62 In a cluster randomized controlled trial, the investigators found no reduction in the rate of adverse outcomes. Differences in the duration of teamwork training and the degree of emphasis on deliberate practice of new skills (eg, with the use of simulation‐based training) likely explains the lack of consistent results.
Very little research has evaluated teamwork training in the general medical environment.69, 70 Sehgal and colleagues recently published an evaluation of the effect of teamwork training delivered to internal medicine residents, hospitalists, nurses, pharmacists, case managers, and social workers on medical services in 3 Northern California hospitals.69 The 4‐hour training sessions covered topical areas of safety culture, teamwork, and communication through didactics, videos, facilitated discussions, and small group role plays to practice new skills and behaviors. The intervention was rated highly among participants,69 and the training along with subsequent follow‐up interventions resulted in improved patient perceptions of teamwork and communication but had no impact on key patient outcomes.71
Interdisciplinary Rounds
Interdisciplinary rounds (IDR) have been used for many years as a means to assemble team members in a single location,7275 and the use of IDR has been associated with lower mortality among ICU patients.76 Interdisciplinary rounds may be particularly useful for clinical settings in which team members are traditionally dispersed in time and place, such as medical‐surgical units. Recent studies have evaluated the effect of structured inter‐disciplinary rounds (SIDR),77, 78 which combine a structured format for communication, similar to a daily goals‐of‐care form, with a forum for daily interdisciplinary meetings. Though no effect was seen on length of stay or cost, SIDR resulted in significantly higher ratings of the quality of collaboration and teamwork climate, and a reduction in the rate of AEs.79 Importantly, the majority of clinicians in the studies agreed that SIDR improved the efficiency of their work day, and expressed a desire that SIDR continue indefinitely. Many investigators have emphasized the importance of leadership during IDR, often by a medical director, nurse manager, or both.74, 77, 78
Summary of Interventions to Improve Teamwork
Localization of physicians increases the frequency of nurse‐physician communication, but is insufficient in creating a shared understanding of patients' plans of care. Providing structure for the discussion among team members (eg, daily goals of care forms and checklists) ensures that critical elements of the plan of care are communicated. Teamwork training is based upon a strong foundation of research both inside and outside of healthcare, and has demonstrated improved knowledge of teamwork principles, attitudes about the importance of teamwork, and overall safety climate. Creating a forum for team members to assemble and discuss their patients (eg, IDR) can overcome some of the unique barriers to collaboration in settings where members are dispersed in time and space. Leaders wishing to improve interdisciplinary teamwork should consider implementing a combination of complementary interventions. For example, localization may increase the frequency of team member interactions, the quality of which may be enhanced with teamwork training and reinforced with the use of structured communication tools and IDR. Future research should evaluate the effect of these combined interventions.
CONCLUSIONS
In summary, teamwork is critically important to provide safe and effective care. Important and unique barriers to teamwork exist in hospitals. We recommend the use of survey instruments, such as those mentioned earlier, as the most feasible method to assess teamwork in the general medical setting. Because each intervention addresses only a portion of the barriers to optimal teamwork, we encourage leaders to use a multifaceted approach. We recommend the implementation of a combination of interventions with adaptations to fit unique clinical settings and local culture.
Acknowledgements
This manuscript was prepared as part of the High Performance Teams and the Hospital of the Future project, a collaborative effort including senior leadership from the American College of Physician Executives, the American Hospital Association, the American Organization of Nurse Executives, and the Society of Hospital Medicine. The authors thank Taylor Marsh for her administrative support and help in coordinating project meetings.
Teamwork is important in providing high‐quality hospital care. Despite tremendous efforts in the 10 years since publication of the Institute of Medicine's To Err is Human report,1 hospitalized patients remain at risk for adverse events (AEs).2 Although many AEs are not preventable, a large portion of those which are identified as preventable can be attributed to communication and teamwork failures.35 A Joint Commission study indicated that communication failures were the root cause for two‐thirds of the 3548 sentinel events reported from 1995 to 2005.6 Another study, involving interviews of resident physicians about recent medical mishaps, found that communication failures contributed to 91% of the AEs they reported.5
Teamwork also plays an important role in other aspects of hospital care delivery. Patients' ratings of nurse‐physician coordination correlate with their overall perception of the quality of care received.7, 8 A study of Veterans Health Administration (VHA) hospitals found that teamwork culture was significantly and positively associated with overall patient satisfaction.9 Another VHA study found that hospitals with higher teamwork culture ratings had lower nurse resignations rates.10 Furthermore, poor teamwork within hospitals may have an adverse effect on financial performance, as a result of inefficiencies in physician and nurse workflow.11
Some organizations are capable of operating in complex, hazardous environments while maintaining exceptional performance over long periods of time. These high reliability organizations (HRO) include aircraft carriers, air traffic control systems, and nuclear power plants, and are characterized by their preoccupation with failure, reluctance to simplify interpretations, sensitivity to operations, commitment to resilience, and deference to expertise.12, 13 Preoccupation with failure is manifested by an organization's efforts to avoid complacency and persist in the search for additional risks. Reluctance to simplify interpretations is exemplified by an interest in pursuing a deep understanding of the issues that arise. Sensitivity to operations is the close attention paid to input from front‐line personnel and processes. Commitment to resilience relates to an organization's ability to contain errors once they occur and mitigate harm. Deference to expertise describes the practice of having authority migrate to the people with the most expertise, regardless of rank. Collectively, these qualities produce a state of mindfulness, allowing teams to anticipate and become aware of unexpected events, yet also quickly contain and learn from them. Recent publications have highlighted the need for hospitals to learn from HROs and the teams within them.14, 15
Recognizing the importance of teamwork in hospitals, senior leadership from the American College of Physician Executives (ACPE), the American Hospital Association (AHA), the American Organization of Nurse Executives (AONE), and the Society of Hospital Medicine (SHM) established the High Performance Teams and the Hospital of the Future project. This collaborative learning effort aims to redesign care delivery to provide optimal value to hospitalized patients. As an initial step, the High Performance Teams and the Hospital of the Future project team completed a literature review related to teamwork in hospitals. The purpose of this report is to summarize the current understanding of teamwork, describe interventions designed to improve teamwork, and make practical recommendations for hospitals to assess and improve teamwork‐related performance. We approach teamwork from the hospitalized patient's perspective, and restrict our discussion to interactions occurring among healthcare professionals within the hospital. We recognize the importance of teamwork at all points in the continuum of patient care. Highly functional inpatient teams should be integrated into an overall system of coordinated and collaborative care.
TEAMWORK: DEFINITION AND CONSTRUCTS
Physicians, nurses, and other healthcare professionals spend a great deal of their time on communication and coordination of care activities.1618 In spite of this and the patient safety concerns previously noted, interpersonal communication skills and teamwork have been historically underemphasized in professional training.1922 A team is defined as 2 or more individuals with specified roles interacting adaptively, interdependently, and dynamically toward a shared and common goal.23 Elements of effective teamwork have been identified through research conducted in aviation, the military, and more recently, healthcare. Salas and colleagues have synthesized this research into 5 core components: team leadership, mutual performance monitoring, backup behavior, adaptability, and team orientation (see Table 1).23 Additionally, 3 supporting and coordinating mechanisms are essential for effective teamwork: shared mental model, closed‐loop communication, and mutual trust (see Table 1).23 High‐performing teams use these elements to develop a culture for speaking up, and situational awareness among team members. Situational awareness refers to a person's perception and understanding of their dynamic environment, and human errors often result from a lack of such awareness.24 These teamwork constructs provide the foundational basis for understanding how hospitals can identify teamwork challenges, assess team performance, and design effective interventions.
| Teamwork | Definition | Behavioral Examples |
|---|---|---|
| ||
| Component | ||
| Team leadership | The leader directs and coordinates team members activities | Facilitate team problem solving; |
| Provide performance expectations; | ||
| Clarify team member roles; | ||
| Assist in conflict resolution | ||
| Mutual performance monitoring | Team members are able to monitor one another's performance | Identify mistakes and lapses in other team member actions; |
| Provide feedback to fellow team members to facilitate self‐correction | ||
| Backup behavior | Team members anticipate and respond to one another's needs | Recognize workload distribution problem; |
| Shift work responsibilities to underutilized members | ||
| Adaptability | The team adjusts strategies based on new information | Identify cues that change has occurred and develop plan to deal with changes; |
| Remain vigilant to change in internal and external environment | ||
| Team orientation | Team members prioritize team goals above individual goals | Take into account alternate solutions by teammates; |
| Increased task involvement, information sharing, and participatory goal setting | ||
| Coordinating mechanism | ||
| Shared mental model | An organizing knowledge of the task of the team and how members will interact to achieve their goal | Anticipate and predict each other's needs; |
| Identify changes in team, task, or teammates | ||
| Closed‐loop communication | Acknowledgement and confirmation of information received | Follow up with team members to ensure message received; |
| Acknowledge that message was received; | ||
| Clarify information received | ||
| Mutual trust | Shared belief that team members will perform their roles | Share information; |
| Willingly admit mistakes and accept feedback | ||
CHALLENGES TO EFFECTIVE TEAMWORK
Several important and unique barriers to teamwork exist in hospitals. Teams are large and formed in an ad hoc fashion. On a given day, a patient's hospital team might include a hospitalist, a nurse, a case manager, a pharmacist, and 1 or more consulting physicians and therapists. Team members in each respective discipline care for multiple patients at the same time, yet few hospitals align team membership (ie, patient assignment). Therefore, a nurse caring for 4 patients may interact with 4 different hospitalists. Similarly, a hospitalist caring for 14 patients may interact with multiple nurses in a given day. Team membership is ever changing because hospital professionals work in shifts and rotations. Finally, team members are seldom in the same place at the same time because physicians often care for patients on multiple units and floors, while nurses and other team members are often unit‐based. Salas and others have noted that team size, instability, and geographic dispersion of membership serve as important barriers to improving teamwork.25, 26 As a result of these barriers, nurses and physicians do not communicate consistently, and often disagree on the daily plan of care for their patients.27, 28 When communication does occur, clinicians may overestimate how well their messages are understood by other team members, reflecting a phenomenon well known in communication psychology related to egocentric thought processes.29, 30
The traditionally steep hierarchy within medicine may also serve as a barrier to teamwork. Studies in intensive care units (ICUs), operating rooms, and general medical units reveal widely discrepant views on the quality of collaboration and communication between healthcare professionals.3133 Although physicians generally give high ratings to the quality of collaboration with nurses, nurses consistently rate the quality of collaboration with physicians as poor. Similarly, specialist physicians rate collaboration with hospitalists higher than hospitalists rate collaboration with specialists.33 Effective teams in other high‐risk industries, like aviation, strive to flatten hierarchy so that team members feel comfortable raising concerns and engaging in open and respectful communications.34
The effect of technology on communication practices and teamwork is complex and incompletely understood. The implementation of electronic heath records and computerized provider order entry systems fundamentally changes work‐flow, and may result in less synchronization and feedback during nurse‐physician collaboration.35 Similarly, the expanded use of text messages delivered via alphanumeric paging or mobile phone results in a transition toward asynchronous modes of communication. These asynchronous modes allow healthcare professionals to review and respond to messages at their convenience, and may reduce unnecessary interruptions. Research shows that these systems are popular among clinicians.3638 However, receipt and understanding of the intended message may not be confirmed with the use of asynchronous modes of communication. Moreover, important face‐to‐face communication elements (tone of voice, expression, gesture, eye contract)39, 40 are lacking. One promising approach is a system which sends low‐priority messages to a Web‐based task list for periodic review, while allowing higher priority messages to pass through to an alphanumeric pager and interrupt the intended recipient.41 Another common frustration in hospitals, despite advancing technology, is difficulty identifying the correct physician(s) and nurse(s) caring for a particular patient at a given point in time.33 Wong and colleagues found that 14% of pages in their hospital were initially sent to the wrong physician.42
ASSESSMENT OF TEAMWORK
One of the challenges in improving teamwork is the difficulty in measuring it. Teamwork assessment entails measuring the performance of teams composed of multiple individuals. Methods of teamwork assessment can be broadly categorized as self assessment, peer assessment, direct observation, survey of team climate or culture, and measurement of the outcome of effective teamwork. While self‐report tools are easy to administer and can capture affective components influencing team performance, they may not reflect actual skills on the part of individuals or teams. Peer assessment includes the use of 360‐degree evaluations or multisource feedback, and provides an evaluation of individual performance.4347
Direct observation provides a more accurate assessment of team‐related behaviors using trained observers. Observers use checklists and/or behaviorally anchored rating scales (BARS) to evaluate individual and team performance. A number of BARS have been developed and validated for the evaluation of team performance.4852 Of note, direct observation may be difficult in settings in which team members are not in the same place at the same time. An alternative method, which may be better suited for general medical units, is the use of survey instruments designed to assess attitudes and teamwork climate.5355 Importantly, higher survey ratings of collaboration and teamwork have been associated with better patient outcomes in observational studies.5658
The ultimate goal of teamwork efforts is to improve patient outcomes. Because patient outcomes are affected by a number of factors and because hospitals frequently engage in multiple, simultaneous efforts to improve care, it is often difficult to clearly link improved outcomes with teamwork interventions. Continued efforts to rigorously evaluate teamwork interventions should remain a priority, particularly as the cost of these interventions must be weighed against other interventions and investments.
EXAMPLES OF SUCCESSFUL INTERVENTIONS
A number of interventions have been used to improve teamwork in hospitals (see Table 2).
| Intervention | Advantages | Disadvantages |
|---|---|---|
| Localization of physicians | Increases frequency of nurse‐physician communication; provides foundation for additional interventions | Insufficient in creating a shared mental model; does not specifically enhance communication skills |
| Daily goals‐of‐care forms and checklists | Provides structure to interdisciplinary discussions and ensures input from all team members | May be completed in a perfunctory manner and may not be updated as plans of care evolve |
| Teamwork training | Emphasizes improved communication behaviors relevant across a range of team member interactions | Requires time and deliberate practice of new skills; effect may be attenuated if members are dispersed. |
| Interdisciplinary rounds | Provides a forum for regular interdisciplinary communication | Requires leadership to organize discussion and does not address need for updates as plans of care evolve |
Geographic Localization of Physicians
As mentioned earlier, physicians in large hospitals may care for patients on multiple units or floors. Designating certain physicians to care for patients admitted to specific units may improve efficiency and communication among healthcare professionals. One study recently reported on the effect of localization of hospital physicians to specific patient care units. Localization resulted in an increase in the rate of nurse‐physician communication, but did not improve providers' shared understanding of the plan of care.56 Notably, localizing physicians may improve the feasibility of additional interventions, like teamwork training and interdisciplinary rounds.
Daily Goals of Care and Surgery Safety Checklists
In ICU and operating room settings, physicians and nurses work in proximity, allowing interdisciplinary discussions to occur at the bedside. The finding that professionals in ICUs and operating rooms have widely discrepant views on the quality of collaboration31, 32 indicates that proximity, alone, is not sufficient for effective communication. Pronovost et al. used a daily goals form for bedside ICU rounds in an effort to standardize communication about the daily plan of care.57 The form defined essential goals of care for patients, and its use resulted in a significant improvement in the team's understanding of the daily goals. Narasimhan et al. performed a similar study using a daily goals worksheet during ICU rounds,58 and also found a significant improvement in physicians' and nurses' ratings of their understanding of the goals of care. The forms used in these studies provided structure to the interdisciplinary conversations during rounds to create a shared understanding of patients' plans of care.
Haynes and colleagues recently reported on the use of a surgical safety checklist in a large, multicenter pre‐post study.59 The checklist consisted of verbal confirmation of the completion of basic steps essential to safe care in the operating room, and provided structure to communication among surgical team members to ensure a shared understanding of the operative plan. The intervention resulted in a significant reduction in inpatient complications and mortality.
Team Training
Formalized team training, based on crew resource management, has been studied as a potential method to improve teamwork in a variety of medical settings.6062 Training emphasizes the core components of successful teamwork and essential coordinating mechanisms previously mentioned.23 Team training appears to positively influence culture, as assessed by teamwork and patient safety climate survey instruments.60 Based on these findings and extensive research demonstrating the success of teamwork training in aviation,63 the Agency for Healthcare Research and Quality (AHRQ) and the Department of Defense (DoD) have partnered in offering the Team Strategies and Tools to Enhance Performance and Patient Safety (TeamSTEPPS) program, designed to improve teamwork skills for healthcare professionals.64, 65
Only a handful of studies have evaluated the effectiveness of teamwork training programs on patient outcomes, and the results are mixed.66 Morey et al. found a reduction in the rate of observed errors as a result of teamwork training in emergency departments, but observers in the study were not blinded with regard to whether teams had undergone training.61 A research group in the United Kingdom evaluated the benefit of simulation‐based team training on outcomes in an obstetrical setting.67, 68 Training included management of specific complications, including shoulder dystocia and uterine cord prolapse. Using retrospective chart review, the investigators found a significant reduction in the proportion of babies born with an obstetric brachial palsy injury and a reduction in the time from diagnosis of uterine cord prolapse to infant delivery. Nielsen and colleagues also evaluated the use of teamwork training in an obstetric setting.62 In a cluster randomized controlled trial, the investigators found no reduction in the rate of adverse outcomes. Differences in the duration of teamwork training and the degree of emphasis on deliberate practice of new skills (eg, with the use of simulation‐based training) likely explains the lack of consistent results.
Very little research has evaluated teamwork training in the general medical environment.69, 70 Sehgal and colleagues recently published an evaluation of the effect of teamwork training delivered to internal medicine residents, hospitalists, nurses, pharmacists, case managers, and social workers on medical services in 3 Northern California hospitals.69 The 4‐hour training sessions covered topical areas of safety culture, teamwork, and communication through didactics, videos, facilitated discussions, and small group role plays to practice new skills and behaviors. The intervention was rated highly among participants,69 and the training along with subsequent follow‐up interventions resulted in improved patient perceptions of teamwork and communication but had no impact on key patient outcomes.71
Interdisciplinary Rounds
Interdisciplinary rounds (IDR) have been used for many years as a means to assemble team members in a single location,7275 and the use of IDR has been associated with lower mortality among ICU patients.76 Interdisciplinary rounds may be particularly useful for clinical settings in which team members are traditionally dispersed in time and place, such as medical‐surgical units. Recent studies have evaluated the effect of structured inter‐disciplinary rounds (SIDR),77, 78 which combine a structured format for communication, similar to a daily goals‐of‐care form, with a forum for daily interdisciplinary meetings. Though no effect was seen on length of stay or cost, SIDR resulted in significantly higher ratings of the quality of collaboration and teamwork climate, and a reduction in the rate of AEs.79 Importantly, the majority of clinicians in the studies agreed that SIDR improved the efficiency of their work day, and expressed a desire that SIDR continue indefinitely. Many investigators have emphasized the importance of leadership during IDR, often by a medical director, nurse manager, or both.74, 77, 78
Summary of Interventions to Improve Teamwork
Localization of physicians increases the frequency of nurse‐physician communication, but is insufficient in creating a shared understanding of patients' plans of care. Providing structure for the discussion among team members (eg, daily goals of care forms and checklists) ensures that critical elements of the plan of care are communicated. Teamwork training is based upon a strong foundation of research both inside and outside of healthcare, and has demonstrated improved knowledge of teamwork principles, attitudes about the importance of teamwork, and overall safety climate. Creating a forum for team members to assemble and discuss their patients (eg, IDR) can overcome some of the unique barriers to collaboration in settings where members are dispersed in time and space. Leaders wishing to improve interdisciplinary teamwork should consider implementing a combination of complementary interventions. For example, localization may increase the frequency of team member interactions, the quality of which may be enhanced with teamwork training and reinforced with the use of structured communication tools and IDR. Future research should evaluate the effect of these combined interventions.
CONCLUSIONS
In summary, teamwork is critically important to provide safe and effective care. Important and unique barriers to teamwork exist in hospitals. We recommend the use of survey instruments, such as those mentioned earlier, as the most feasible method to assess teamwork in the general medical setting. Because each intervention addresses only a portion of the barriers to optimal teamwork, we encourage leaders to use a multifaceted approach. We recommend the implementation of a combination of interventions with adaptations to fit unique clinical settings and local culture.
Acknowledgements
This manuscript was prepared as part of the High Performance Teams and the Hospital of the Future project, a collaborative effort including senior leadership from the American College of Physician Executives, the American Hospital Association, the American Organization of Nurse Executives, and the Society of Hospital Medicine. The authors thank Taylor Marsh for her administrative support and help in coordinating project meetings.
- To Err Is Human: Building a Safer Health System.Washington, DC:Institute of Medicine;1999.
- ,,,,,.Temporal trends in rates of patient harm resulting from medical care.N Engl J Med.2010;363(22):2124–2134.
- ,,.Exploring the causes of adverse events in NHS hospital practice.J R Soc Med.2001;94(7):322–330.
- ,,,,,.The Quality in Australian Health Care Study.Med J Aust.1995;163(9):458–471.
- ,,.Communication failures: an insidious contributor to medical mishaps.Acad Med.2004;79(2):186–194.
- Improving America's Hospitals: The Joint Commission's Annual Report on Quality and Safety 2007. Available at: http://www.jointcommissionreport.org. Accessed November2007.
- ,,.Patient perceptions of coordinated care: the importance of organized communication in hospitals.J Healthc Qual.1999;21(5):18–23.
- ,,.Am I safe here? Improving patients' perceptions of safety in hospitals.J Nurs Care Qual.2006;21(1):30–40.
- ,,.Teamwork culture and patient satisfaction in hospitals.Med Care.2004;42(5):492–498.
- ,,.The influence of teamwork culture on physician and nurse resignation rates in hospitals.Health Serv Manage Res.2008;21(1):23–31.
- ,,.Quantifying the economic impact of communication inefficiencies in U.S. hospitals.J Healthc Manag.2010;55(4):265–282.
- ,.Managing the Unexpected: Assuring High Performance in an Age of Complexity.San Francisco, CA:Jossey‐Bass;2001.
- .Some characteristics of high reliability organizations.Organization Science.1990;1(2):160–177.
- ,,.Teamwork as an essential component of high‐reliability organizations.Health Serv Res.2006;41(4 pt 2):1576–1598.
- ,,,.Promoting health care safety through training high reliability teams.Qual Saf Health Care.2005;14(4):303–309.
- ,,,,,.Analyzing the time and value of housestaff inpatient work.J Gen Intern Med.1998;13(8):534–540.
- ,,, et al.Quantifying nursing workflow in medication administration.J Nurs Adm.2008;38(1):19–26.
- ,,.How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- ,,,,.Redesigning residency education in internal medicine: a position paper from the Association of Program Directors in Internal Medicine.Ann Intern Med.2006;144(12):920–926.
- ,,,.Hospitalists' perceptions of their residency training needs: results of a national survey.Am J Med.2001;111(3):247–254.
- ,,.Redesigning training for internal medicine.Ann Intern Med.2006;144(12):927–932.
- ,,,,.The role of teamwork in the professional education of physicians: current status and assessment recommendations.Jt Comm J Qual Patient Saf.2005;31(4):185–202.
- ,,.Is there a “big five” in teamwork?Small Group Research.2005;36:555–599.
- ,,.Objective measures of situation awareness in a simulated medical environment.Qual Saf Health Care.2004;13(suppl 1):i65–i71.
- ,.What do we know about health care team effectiveness? A review of the literature.Med Care Res Rev.2006;63(3):263–300.
- ,,, et al.Does team training improve team performance? A meta‐analysis.Hum Factors.2008;50(6):903–933.
- ,,,,,.Can we talk? Priorities for patient care differed among health care providers. AHRQ Publication No. 05–0021‐1.Rockville, MD:Agency for Healthcare Research and Quality;2005.
- ,,, et al.Patterns of nurse–physicians communication and agreement on the plan of care.Qual Saf Health Care.2010;19:195–199.
- ,,,,.Interns overestimate the effectiveness of their hand‐off communication.Pediatrics.2010;125(3):491–496.
- ,.Speakers' overestimation of their effectiveness.Psychol Sci.2002;13(3):207–212.
- ,,, et al.Operating room teamwork among physicians and nurses: teamwork in the eye of the beholder.J Am Coll Surg.2006;202(5):746–752.
- ,,.Discrepant attitudes about teamwork among critical care nurses and physicians.Crit Care Med.2003;31(3):956–959.
- ,,,,,.Teamwork on inpatient medical units: assessing attitudes and barriers.Qual Saf Health Care.2010;19(2):117–121.
- ,,.Error, stress, and teamwork in medicine and aviation: cross sectional surveys.BMJ.2000;320(7237):745–749.
- ,,,,.Impact of a computerized physician order entry system on nurse‐physician collaboration in the medication process.Int J Med Inform.2008;77(11):735–744.
- ,,,,.Alphanumeric paging in an academic hospital setting.Am J Surg.2006;191(4):561–565.
- ,,,.Implementation and evaluation of an alpha‐numeric paging system on a resident inpatient teaching service.J Hosp Med.2009;4(8):E34–E40.
- ,,, et al.The use of smartphones for clinical communication on internal medicine wards.J Hosp Med.2010;5(9):553–559.
- ,.Organizational information requirements, media richness, and structural design.Management Science.1986;32(5):554–571.
- ,.Decoding of inconsistent communications of personality and social psychology.J Pers Soc Psychol.1967;6(1):109–114.
- ,,,,.Beyond paging: building a Web‐based communication tool for nurses and physicians.J Gen Intern Med.2009;24(1):105–110.
- ,,, et al.Frequency and clinical importance of pages sent to the wrong physician.Arch Intern Med.2009;169(11):1072–1073.
- ,,, et al.Evaluation of resident communication skills and professionalism: a matter of perspective?Pediatrics.2006;118(4):1371–1379.
- ,,, et al.Effect of multisource feedback on resident communication skills and professionalism: a randomized controlled trial.Arch Pediatr Adolesc Med.2007;161(1):44–49.
- .Multisource feedback in the assessment of physician competencies.J Contin Educ Health Prof.2003;23(1):4–12.
- ,.Reliability of a 360‐degree evaluation to assess resident competence.Am J Phys Med Rehabil.2007;86(10):845–852.
- ,,,.Pilot study of a 360‐degree assessment instrument for physical medicine 82(5):394–402.
- ,,,,,.Anaesthetists' Non‐Technical Skills (ANTS): evaluation of a behavioural marker system.Br J Anaesth.2003;90(5):580–588.
- ,,,.Using the Communication and Teamwork Skills (CATS) Assessment to measure health care team performance.Jt Comm J Qual Patient Saf.2007;33(9):549–558.
- ,,, et al.The Mayo High Performance Teamwork Scale: reliability and validity for evaluating key crew resource management skills.Simul Healthc.2007;2(1):4–10.
- ,,,,,.Reliability of a revised NOTECHS scale for use in surgical teams.Am J Surg.2008;196(2):184–190.
- ,,,,,.Observational teamwork assessment for surgery: construct validation with expert versus novice raters.Ann Surg.2009;249(6):1047–1051.
- ,,, et al.The Safety Attitudes Questionnaire: psychometric properties, benchmarking data, and emerging research.BMC Health Serv Res.2006;6:44.
- .Development of an instrument to measure collaboration and satisfaction about care decisions.J Adv Nurs.1994;20(1):176–182.
- ,,,,,.Psychometric properties of an attitude scale measuring physician‐nurse collaboration.Eval Health Prof.1999;22(2):208–220.
- ,,, et al.Impact of localizing physicians to hospital units on nurse‐physician communication and agreement on the plan of care.J Gen Intern Med.2009;24(11):1223–1227.
- ,,,,,.Improving communication in the ICU using daily goals.J Crit Care.2003;18(2):71–75.
- ,,,,.Improving nurse‐physician communication and satisfaction in the intensive care unit with a daily goals worksheet.Am J Crit Care.2006;15(2):217–222.
- ,,, et al.A surgical safety checklist to reduce morbidity and mortality in a global population.N Engl J Med.2009;360(5):491–499.
- ,,, et al.Effect of crew resource management training in a multidisciplinary obstetrical setting.Int J Qual Health Care.2008;20(4):254–263.
- ,,, et al.Error reduction and performance improvement in the emergency department through formal teamwork training: evaluation results of the MedTeams project.Health Serv Res.2002;37(6):1553–1581.
- ,,, et al.Effects of teamwork training on adverse outcomes and process of care in labor and delivery: a randomized controlled trial.Obstet Gynecol.2007;109(1):48–55.
- ,,,,.Medical Teamwork and Patient Safety: The Evidence‐Based Relation.Rockville, MD:Agency for Healthcare Research and Quality;2005.
- Agency for Healthcare Research and Quality. TeamSTEPPS Home. Available at: http://teamstepps.ahrq.gov/index.htm. Accessed January 18,2010.
- ,.TeamSTEPPS: assuring optimal teamwork in clinical settings.Am J Med Qual.2007;22(3):214–217.
- ,,,.Does crew resource management training work? An update, an extension, and some critical needs.Hum Factors.2006;48(2):392–412.
- ,,, et al.Improving neonatal outcome through practical shoulder dystocia training.Obstet Gynecol.2008;112(1):14–20.
- ,,, et al.Retrospective cohort study of diagnosis‐delivery interval with umbilical cord prolapse: the effect of team training.Br J Obstet Gynaecol.2009;116(8):1089–1096.
- ,,, et al.A multidisciplinary teamwork training program: the Triad for Optimal Patient Safety (TOPS) experience.J Gen Intern Med.2008;23(12):2053–2057.
- ,,,,.Teambuilding and leadership training in an internal medicine residency training program.J Gen Intern Med.2004;19(6):692–697.
- ,,, et al. Effects of a multicenter teamwork and communication program on patient outcomes: results from the Triad for Optimal Patient Safety (TOPS) project. In press.
- ,,, et al.The effect of a multidisciplinary hospitalist/physician and advanced practice nurse collaboration on hospital costs.J Nurs Adm.2006;36(2):79–85.
- ,,.A firm trial of interdisciplinary rounds on the inpatient medical wards: an intervention designed using continuous quality improvement.Med Care.1998;36(8 suppl):AS4–A12.
- ,,,,.Use of multidisciplinary rounds to simultaneously improve quality outcomes, enhance resident education, and shorten length of stay.J Gen Intern Med.2007;22(8):1073–1079.
- ,,,.Effect of a multidisciplinary intervention on communication and collaboration among physicians and nurses.Am J Crit Care.2005;14(1):71–77.
- ,,,,.The effect of multidisciplinary care teams on intensive care unit mortality.Arch Intern Med.2010;170(4):369–376.
- ,,,,,.Improving teamwork: impact of structured interdisciplinary rounds on a hospitalist unit.J Hosp Med.2011;6(2):88–93.
- ,,,,,.Improving teamwork: impact of structured interdisciplinary rounds on a medical teaching unit.J Gen Intern Med.2010;25(8):826–832.
- ,,, et al.Structured interdisciplinary rounds in a medical teaching unit: improving patient safety.Arch Intern Med.2011;171(7):678–684.
- To Err Is Human: Building a Safer Health System.Washington, DC:Institute of Medicine;1999.
- ,,,,,.Temporal trends in rates of patient harm resulting from medical care.N Engl J Med.2010;363(22):2124–2134.
- ,,.Exploring the causes of adverse events in NHS hospital practice.J R Soc Med.2001;94(7):322–330.
- ,,,,,.The Quality in Australian Health Care Study.Med J Aust.1995;163(9):458–471.
- ,,.Communication failures: an insidious contributor to medical mishaps.Acad Med.2004;79(2):186–194.
- Improving America's Hospitals: The Joint Commission's Annual Report on Quality and Safety 2007. Available at: http://www.jointcommissionreport.org. Accessed November2007.
- ,,.Patient perceptions of coordinated care: the importance of organized communication in hospitals.J Healthc Qual.1999;21(5):18–23.
- ,,.Am I safe here? Improving patients' perceptions of safety in hospitals.J Nurs Care Qual.2006;21(1):30–40.
- ,,.Teamwork culture and patient satisfaction in hospitals.Med Care.2004;42(5):492–498.
- ,,.The influence of teamwork culture on physician and nurse resignation rates in hospitals.Health Serv Manage Res.2008;21(1):23–31.
- ,,.Quantifying the economic impact of communication inefficiencies in U.S. hospitals.J Healthc Manag.2010;55(4):265–282.
- ,.Managing the Unexpected: Assuring High Performance in an Age of Complexity.San Francisco, CA:Jossey‐Bass;2001.
- .Some characteristics of high reliability organizations.Organization Science.1990;1(2):160–177.
- ,,.Teamwork as an essential component of high‐reliability organizations.Health Serv Res.2006;41(4 pt 2):1576–1598.
- ,,,.Promoting health care safety through training high reliability teams.Qual Saf Health Care.2005;14(4):303–309.
- ,,,,,.Analyzing the time and value of housestaff inpatient work.J Gen Intern Med.1998;13(8):534–540.
- ,,, et al.Quantifying nursing workflow in medication administration.J Nurs Adm.2008;38(1):19–26.
- ,,.How hospitalists spend their time: insights on efficiency and safety.J Hosp Med.2006;1(2):88–93.
- ,,,,.Redesigning residency education in internal medicine: a position paper from the Association of Program Directors in Internal Medicine.Ann Intern Med.2006;144(12):920–926.
- ,,,.Hospitalists' perceptions of their residency training needs: results of a national survey.Am J Med.2001;111(3):247–254.
- ,,.Redesigning training for internal medicine.Ann Intern Med.2006;144(12):927–932.
- ,,,,.The role of teamwork in the professional education of physicians: current status and assessment recommendations.Jt Comm J Qual Patient Saf.2005;31(4):185–202.
- ,,.Is there a “big five” in teamwork?Small Group Research.2005;36:555–599.
- ,,.Objective measures of situation awareness in a simulated medical environment.Qual Saf Health Care.2004;13(suppl 1):i65–i71.
- ,.What do we know about health care team effectiveness? A review of the literature.Med Care Res Rev.2006;63(3):263–300.
- ,,, et al.Does team training improve team performance? A meta‐analysis.Hum Factors.2008;50(6):903–933.
- ,,,,,.Can we talk? Priorities for patient care differed among health care providers. AHRQ Publication No. 05–0021‐1.Rockville, MD:Agency for Healthcare Research and Quality;2005.
- ,,, et al.Patterns of nurse–physicians communication and agreement on the plan of care.Qual Saf Health Care.2010;19:195–199.
- ,,,,.Interns overestimate the effectiveness of their hand‐off communication.Pediatrics.2010;125(3):491–496.
- ,.Speakers' overestimation of their effectiveness.Psychol Sci.2002;13(3):207–212.
- ,,, et al.Operating room teamwork among physicians and nurses: teamwork in the eye of the beholder.J Am Coll Surg.2006;202(5):746–752.
- ,,.Discrepant attitudes about teamwork among critical care nurses and physicians.Crit Care Med.2003;31(3):956–959.
- ,,,,,.Teamwork on inpatient medical units: assessing attitudes and barriers.Qual Saf Health Care.2010;19(2):117–121.
- ,,.Error, stress, and teamwork in medicine and aviation: cross sectional surveys.BMJ.2000;320(7237):745–749.
- ,,,,.Impact of a computerized physician order entry system on nurse‐physician collaboration in the medication process.Int J Med Inform.2008;77(11):735–744.
- ,,,,.Alphanumeric paging in an academic hospital setting.Am J Surg.2006;191(4):561–565.
- ,,,.Implementation and evaluation of an alpha‐numeric paging system on a resident inpatient teaching service.J Hosp Med.2009;4(8):E34–E40.
- ,,, et al.The use of smartphones for clinical communication on internal medicine wards.J Hosp Med.2010;5(9):553–559.
- ,.Organizational information requirements, media richness, and structural design.Management Science.1986;32(5):554–571.
- ,.Decoding of inconsistent communications of personality and social psychology.J Pers Soc Psychol.1967;6(1):109–114.
- ,,,,.Beyond paging: building a Web‐based communication tool for nurses and physicians.J Gen Intern Med.2009;24(1):105–110.
- ,,, et al.Frequency and clinical importance of pages sent to the wrong physician.Arch Intern Med.2009;169(11):1072–1073.
- ,,, et al.Evaluation of resident communication skills and professionalism: a matter of perspective?Pediatrics.2006;118(4):1371–1379.
- ,,, et al.Effect of multisource feedback on resident communication skills and professionalism: a randomized controlled trial.Arch Pediatr Adolesc Med.2007;161(1):44–49.
- .Multisource feedback in the assessment of physician competencies.J Contin Educ Health Prof.2003;23(1):4–12.
- ,.Reliability of a 360‐degree evaluation to assess resident competence.Am J Phys Med Rehabil.2007;86(10):845–852.
- ,,,.Pilot study of a 360‐degree assessment instrument for physical medicine 82(5):394–402.
- ,,,,,.Anaesthetists' Non‐Technical Skills (ANTS): evaluation of a behavioural marker system.Br J Anaesth.2003;90(5):580–588.
- ,,,.Using the Communication and Teamwork Skills (CATS) Assessment to measure health care team performance.Jt Comm J Qual Patient Saf.2007;33(9):549–558.
- ,,, et al.The Mayo High Performance Teamwork Scale: reliability and validity for evaluating key crew resource management skills.Simul Healthc.2007;2(1):4–10.
- ,,,,,.Reliability of a revised NOTECHS scale for use in surgical teams.Am J Surg.2008;196(2):184–190.
- ,,,,,.Observational teamwork assessment for surgery: construct validation with expert versus novice raters.Ann Surg.2009;249(6):1047–1051.
- ,,, et al.The Safety Attitudes Questionnaire: psychometric properties, benchmarking data, and emerging research.BMC Health Serv Res.2006;6:44.
- .Development of an instrument to measure collaboration and satisfaction about care decisions.J Adv Nurs.1994;20(1):176–182.
- ,,,,,.Psychometric properties of an attitude scale measuring physician‐nurse collaboration.Eval Health Prof.1999;22(2):208–220.
- ,,, et al.Impact of localizing physicians to hospital units on nurse‐physician communication and agreement on the plan of care.J Gen Intern Med.2009;24(11):1223–1227.
- ,,,,,.Improving communication in the ICU using daily goals.J Crit Care.2003;18(2):71–75.
- ,,,,.Improving nurse‐physician communication and satisfaction in the intensive care unit with a daily goals worksheet.Am J Crit Care.2006;15(2):217–222.
- ,,, et al.A surgical safety checklist to reduce morbidity and mortality in a global population.N Engl J Med.2009;360(5):491–499.
- ,,, et al.Effect of crew resource management training in a multidisciplinary obstetrical setting.Int J Qual Health Care.2008;20(4):254–263.
- ,,, et al.Error reduction and performance improvement in the emergency department through formal teamwork training: evaluation results of the MedTeams project.Health Serv Res.2002;37(6):1553–1581.
- ,,, et al.Effects of teamwork training on adverse outcomes and process of care in labor and delivery: a randomized controlled trial.Obstet Gynecol.2007;109(1):48–55.
- ,,,,.Medical Teamwork and Patient Safety: The Evidence‐Based Relation.Rockville, MD:Agency for Healthcare Research and Quality;2005.
- Agency for Healthcare Research and Quality. TeamSTEPPS Home. Available at: http://teamstepps.ahrq.gov/index.htm. Accessed January 18,2010.
- ,.TeamSTEPPS: assuring optimal teamwork in clinical settings.Am J Med Qual.2007;22(3):214–217.
- ,,,.Does crew resource management training work? An update, an extension, and some critical needs.Hum Factors.2006;48(2):392–412.
- ,,, et al.Improving neonatal outcome through practical shoulder dystocia training.Obstet Gynecol.2008;112(1):14–20.
- ,,, et al.Retrospective cohort study of diagnosis‐delivery interval with umbilical cord prolapse: the effect of team training.Br J Obstet Gynaecol.2009;116(8):1089–1096.
- ,,, et al.A multidisciplinary teamwork training program: the Triad for Optimal Patient Safety (TOPS) experience.J Gen Intern Med.2008;23(12):2053–2057.
- ,,,,.Teambuilding and leadership training in an internal medicine residency training program.J Gen Intern Med.2004;19(6):692–697.
- ,,, et al. Effects of a multicenter teamwork and communication program on patient outcomes: results from the Triad for Optimal Patient Safety (TOPS) project. In press.
- ,,, et al.The effect of a multidisciplinary hospitalist/physician and advanced practice nurse collaboration on hospital costs.J Nurs Adm.2006;36(2):79–85.
- ,,.A firm trial of interdisciplinary rounds on the inpatient medical wards: an intervention designed using continuous quality improvement.Med Care.1998;36(8 suppl):AS4–A12.
- ,,,,.Use of multidisciplinary rounds to simultaneously improve quality outcomes, enhance resident education, and shorten length of stay.J Gen Intern Med.2007;22(8):1073–1079.
- ,,,.Effect of a multidisciplinary intervention on communication and collaboration among physicians and nurses.Am J Crit Care.2005;14(1):71–77.
- ,,,,.The effect of multidisciplinary care teams on intensive care unit mortality.Arch Intern Med.2010;170(4):369–376.
- ,,,,,.Improving teamwork: impact of structured interdisciplinary rounds on a hospitalist unit.J Hosp Med.2011;6(2):88–93.
- ,,,,,.Improving teamwork: impact of structured interdisciplinary rounds on a medical teaching unit.J Gen Intern Med.2010;25(8):826–832.
- ,,, et al.Structured interdisciplinary rounds in a medical teaching unit: improving patient safety.Arch Intern Med.2011;171(7):678–684.
PESI vs PREP
Acute pulmonary embolism (PE) is associated with significant morbidity and mortality.1 While expeditious diagnosis and management results in reduced mortality, the ability to rapidly and accurately identify those at increased risk for death remains elusive. Multiple studies have utilized various biomarkers as risk stratification tools, however, these approaches have proven to have many limitations. For example, both serum brain natriuretic peptide (BNP) and troponin levels have been studied as possible risk stratification tools. Those with elevated levels of these following a PE may have concomitant right ventricular (RV) dysfunction and/or hemodynamic instability. Thus, they may face a greater risk for cardiovascular collapse and death. The low positive predictive value of these biomarkers (14%‐44%) has limited their clinical utility.24 Furthermore, imaging modalities, such as echocardiography, which is considered the clinical gold standard for determining the presence of acute RV dysfunction in PE, may not be readily available and may require special expertise for interpretation.5
Conversely, the need to identify acute PE patients at low risk for death is just as important. Recent studies suggest that carefully selected patients can successfully be managed as outpatients which can subsequently lead to significant cost savings and patient satisfaction. Movement towards enhanced outpatient resources and the advent of subcutaneous anticoagulants have made outpatient management of acute PE an appealing possibility. However, proper education, close follow‐up, and a rigorous selection process to recognize those at minimal risk for a fatal complication must all be available before clinicians prematurely discharge these patients to home.
Recently, clinical scoring tools have been developed to aid in risk stratifying patients with acute PE to accurately determine patient outcome. The pulmonary embolism severity index (PESI) is a reproducible scoring system that accurately predicts 30‐day and 90‐day mortality.6, 7 It consists of 11 clinical variables that can be quickly assessed at the time of diagnosis (Table 1A). The fact that biomarkers and imaging technology, such as echocardiography, are unnecessary to compute a PESI score demonstrates the appeal of this system. Similar to the PESI, Sanchez et al.8 have proposed the prognosis in pulmonary embolism (PREP) score as an alternate clinical risk tool in PE (Table 1B). Contrary to PESI, the PREP only uses 3 clinical variables to accurately predict vital outcome with an area under the receiver operating characteristic (AUROC) curve of 0.73 (95% confidence interval [CI], 0.65‐0.82). While both scoring systems have been developed to predict 30‐day mortality in acute PE, the comparative validity of these prognostic tools has not been assessed.
| Predictors | Points Assigned |
|---|---|
| |
| Demographic characteristics | |
| Age (yr) | Age (yr) |
| Male sex | +10 |
| Comorbid conditions | |
| Cancer | +30 |
| Heart failure | +10 |
| Chronic lung disease | +10 |
| Clinical findings | |
| Pulse 110 beats/min | +20 |
| Systolic blood pressure <100 mm Hg | +30 |
| Respiratory rate 30 breaths/min | +20 |
| Temperature <36C | +20 |
| Altered mental status* | +60 |
| Arterial oxygen saturation <90% | +20 |
| Prognostic Factor | Points Assigned |
|---|---|
| |
| Altered mental status* | +10 |
| Cardiogenic shock (systolic blood pressure <90 mm Hg) | +6 |
| Cancer | +6 |
We hypothesized that the PESI more precisely risk stratifies the risk for death in acute PE compared to the PREP. Furthermore, we theorized that the PESI more reliably predicts not only 30‐day but also 90‐day mortality. To test our hypothesis, we performed a retrospective analysis, of all consecutive patients diagnosed with acute PE at our hospital, to compare the prognostic accuracy of these 2 scoring systems.
METHODS
Subjects and Definitions
Between October 2007 and February 2009, adults (age 18 years) diagnosed the day prior with acute PE were identified on a daily basis. This study cohort has been described elsewhere.7 Patients with newly diagnosed PE were eligible for enrollment. Those expected to die within 30 days of their acute PE, such as individuals suffering from a terminal condition (metastatic cancer) or critical illness being transitioned to comfort care, were excluded (n = 32). Patients with multiple admissions for acute PE were included only during the first episode. PE was diagnosed using objective criteria through 1 of the following modalities: high probability ventilation‐perfusion (V/Q) scintigraphy, computed tomography (CT) of the chest with PE protocol, or magnetic resonance imaging (MRI) of the chest. A list of patients who had the above imaging studies to evaluate for PE was provided to study personnel daily by the radiology department; this list was generated every morning and consisted of the day prior's studies. Patient management was not influenced by the research team and was the responsibility of the primary team. This study was approved by our local institutional review board and consent was not required.
We calculated the PESI as described by Aujesky and colleagues.6 For outpatients admitted with acute PE, clinical findings available just prior to, and after, diagnosis were used for scoring. For inpatients diagnosed with PE, clinical findings available during the 24 hours just prior to diagnosis were included. Raw PESI scores were converted to risk class (I‐V), and then further dichotomized into low‐risk (class I‐II) and high‐risk (class III‐V) groups (Table 2). The PREP score was computed based on the presence of altered mental status (AMS), cancer, and cardiogenic shock defined as a systolic blood pressure <90 mm Hg (Table 1B). A raw PREP score of <7 was then characterized as low risk for mortality, while scores 7 were considered high risk.
| PESI Score | Class | n | 30‐Day Mortality by Class (%) | 90‐Day Mortality by Class (%) | Low vs High Risk |
|---|---|---|---|---|---|
| |||||
| 65 | I | 49 | 0 (0.0) | 0 (0.0) | Low |
| 66‐85 | II | 59 | 0 (0.0) | 0 (0.0) | |
| 86‐105 | III | 60 | 0 (0.0) | 0 (0.0) | High |
| 106‐125 | IV | 56 | 2 (3.4) | 4 (6.9) | |
| >125 | V | 69 | 7 (9.2) | 8 (10.5) | |
Finally, the PESI and PREP scores were compared based on their ability to predict all‐cause 30‐day and 90‐day mortality. To determine vital status and date of death, we reviewed the Social Security Death Index 90 days after enrollment of all subjects was completed.
Statistical Analysis
To assess the predictive ability of the 2 scoring tools for death, we determined the negative predictive value and computed the AUROC curves for both scoring systems. AUROC curves were constructed for raw scores and when scores were further segregated by class and risk groups. Additionally, 95% CIs were estimated to determine the accuracy of the discriminatory power of the PESI score versus the PREP score.
Post hoc, we calculated the power of our study to assess whether the difference noted in AUROC curves between the PESI and PREP was adequate to truly determine statistical significance. We used methodology described by Hanley and McNeil to compare continuous values.9 Assuming an alpha of 0.05 and a 20% difference in the AUROC curves, as described in our results, the power in our study was 0.35. Therefore, an approximate sample size of 1000 would be necessary to determine statistical significance. This analysis was performed using Power Analysis and Sample Size (PASS) 11.
RESULTS
The final cohort included 302 subjects (mean age: 59.7 17.2 years; 44.0% males). As Table 3 reveals, the majority of PEs was diagnosed via CT scan (76%). On presentation, 6.6% had cardiogenic shock, while 5.0% had altered censorium. In terms of comorbid conditions, 25.2% had congestive heart failure, 25.2% had cancer, and 22.2% had a prior venous thromboembolic event. Overall, 3.0% and 4.0% met our primary outcomes of death within 30‐days and 90‐days of their acute PEs, respectively.
| |
| Demographics | |
| Age (yr), mean SD | 59.7 17.2 |
| Male sex, % | 44% |
| Diagnostic methodology | |
| CT chest, n (%) | 230 (76.2) |
| V/Q scan, n (%) | 71 (23.5) |
| MRA chest, n (%) | 1 (0.3) |
| Comorbidities | |
| Malignancy, n (%) | 76 (25.2) |
| Congestive heart failure, n (%) | 76 (25.2) |
| Chronic lung disease, n (%) | 72 (23.8) |
| Recent orthopedic surgery, n (%) | 22 (7.3) |
| Prior cerebrovascular accident, n (%) | 31 (10.3) |
| Prior venous thromboembolic disease, n (%) | 67 (22.2) |
The rates of 30‐day and 90‐day mortality, respectively, increased with increasing score for both the PESI and the PREP. No patients in PESI class I died by either time point, while 9.2% of PESI class V subjects expired by 30 days (P < 0.0001) and 10.5% died by 90 days (P = 0.003) (Table 2). Based on PESI, 30‐day death rates were 4.6% in the high‐risk cohort versus 0% in the low‐risk group (P = 0.023). Conversely, 7.1% of high‐risk PREP subjects died by day 30 versus 1% of low‐risk subjects (P = 0.004) (Figure 1A). Those stratified into the PESI high‐risk group had a 90‐day mortality of 6.2% versus 0% for the low‐risk group (P = 0.008) versus 9.1% in those deemed high risk by PREP, as compared to 1.5% of those scored as low risk by PREP (P = 0.001) (Figure 1B).
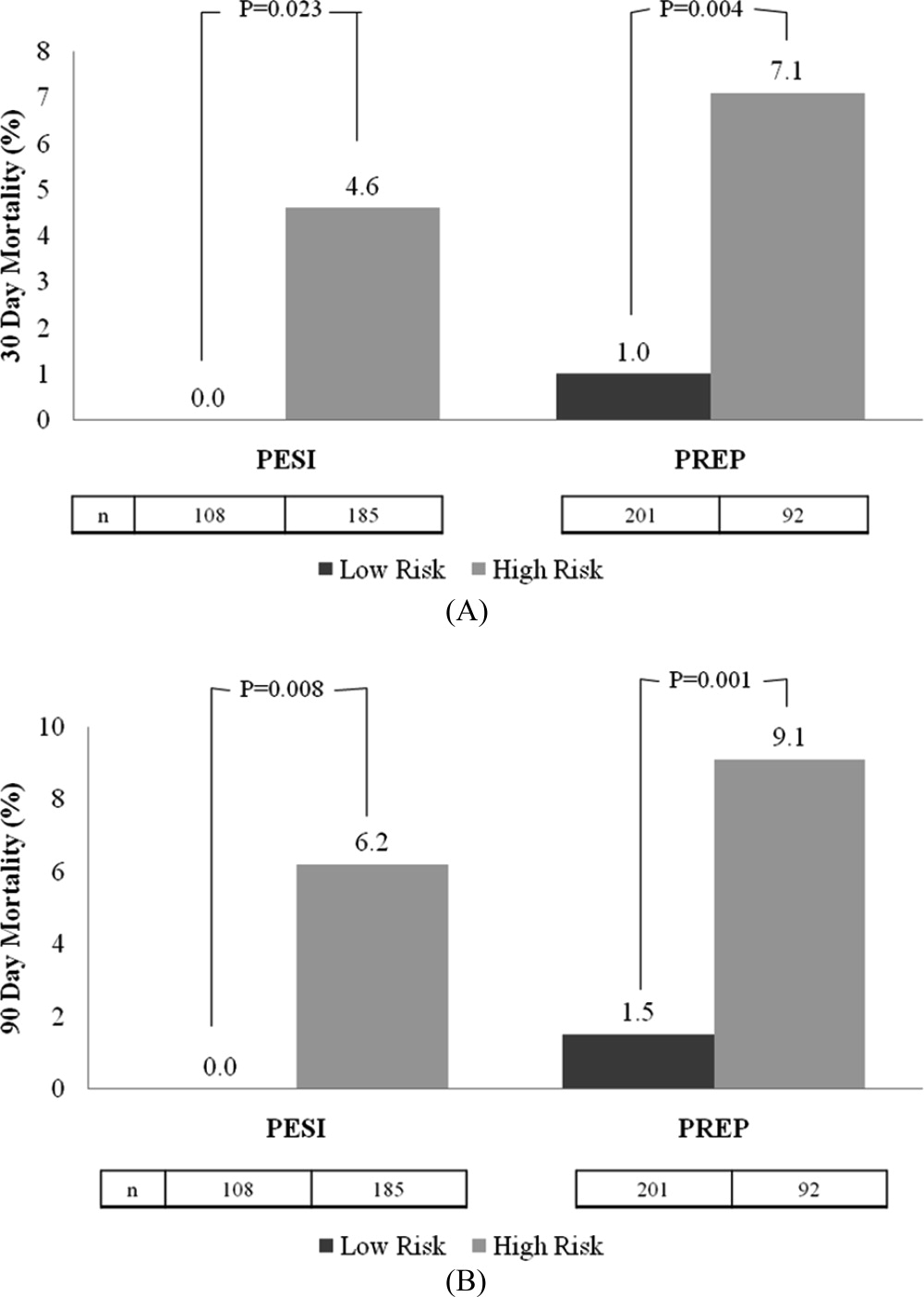
Regarding the 30‐day mortality, the negative predictive value of the PESI was 100% (95% CI, 98.6%‐100%) while that for PREP was 99.0% (95% CI, 97.6%‐99.7%); the ability of the PREP to predict 30‐day mortality was similar to the PESI (Table 4). The AUROCs for PESI and PREP for predicting 30‐day death were also equivalent; for the raw PESI score, this measured 0.858 (95% CI, 0.773‐0.943), compared to 0.719 (95% CI, 0.563‐0.875) for PREP. When these scores were dichotomized to high‐risk versus low‐risk groups, the AUROC for the PESI was 0.684 (95% CI, 0.559‐0.810) and 0.732 (95% CI, 0.571‐0.893) for PREP.
| 30‐Day Mortality | 90‐Day Mortality | |||
|---|---|---|---|---|
| Scoring System | AUROC | 95% CI | AUROC | 95% CI |
| ||||
| Raw PESI | 0.858 | 0.773‐0.943 | 0.835 | 0.762‐0.907 |
| PESI class | 0.835 | 0.756‐0.914 | 0.813 | 0.738‐0.888 |
| PESI high vs low risk | 0.684 | 0.559‐0.810 | 0.686 | 0.576‐0.796 |
| Raw PREP | 0.719 | 0.563‐0.875 | 0.704 | 0.564‐0.844 |
| PREP high vs low risk | 0.732 | 0.571‐0.893 | 0.720 | 0.574‐0.865 |
In terms of 90‐day mortality, the negative predictive values of PESI and PREP did not change: 100% (95% CI, 97.4%‐100%) and 98.5% (95% CI, 96.9%‐99.5%), respectively. The ability of PESI and PREP as predictors of 90‐day mortality was equivalent (Table 4). Here, the AUROC for the raw PESI score remained excellent at 0.835 (97% CI, 0.762‐0.907). The AUROC for PREP was akin to that of PESI at 0.704 (95% CI, 0.564‐0.844). Segregating scores into high‐risk versus low‐risk groups demonstrated that the AUROC for PESI was 0.686 (95% CI, 0.576‐0.796) compared to 0.720 (95% CI, 0.574‐0.865) for PREP.
DISCUSSION
This retrospective analysis of patients with acute PE confirms that both the PESI and the PREP are accurate scoring tools for identifying patients at low risk of death. Under both rubrics, as the score increases, the likelihood of death also increases. More importantly, we demonstrate that the negative predictive value for both the PREP and PESI are excellent. Thus, these scoring tools can distinguish those at higher risk for death versus those at low risk in a simple‐to‐apply manner. In comparing these 2 scoring systems, the PREP comparably identifies acute PE patients at risk for death when contrasted with the PESI. Given the fewer required scoring points to calculate PREP and its ability to accurately predict clinically relevant outcomes, this simpler scoring system may have greater clinical utility.
Prior studies have validated the PESI as a risk stratification tool to predict 30‐day and 90‐day mortalities. In their original derivation of the PESI, Aujesky et al. demonstrated that higher PESI scores correlated with death at 30 days.6 Acute PE patients classified into risk class I had a short‐term mortality rate of 1.1% compared to nearly 25% of patients risk stratified into risk class V. The same authors subsequently verified that there is a linear relationship between PESI score and risk of death at 90 days.10 We have also confirmed the accuracy of the PESI for identifying persons at high risk for death and documented the limited interobserver variability in this tool.7 In combination, there is evidence that the PESI can accurately predict vital outcome. Despite the effectiveness of the PESI, it is a somewhat cumbersome scoring system. It requires gathering information on 11 clinical variables, each with a different score allocation to ultimately compute the PESI score. In contrast, the PREP only requires knowing 3 clinical variables: presence of cancer, mental status, and the presence of cardiogenic shock. Akin to the PESI, the PREP and mortality are linearly related, where higher PREP scores result in higher 30‐day and 90‐day mortalities.
Our analysis helps expand the evidence regarding clinical risk stratification in PE in several ways. First, we verify that both the PESI and PREP are accurate predictors of short‐term mortality. While this has been accomplished for the PESI in prior studies, to our knowledge, this is the first confirmatory study for PREP's utility as a risk stratification tool. Second, we demonstrate that PREP is also an accurate predictor of intermediate‐term mortality. If the eventual goal is to develop tools that allow for the initial outpatient management of acute PE, clinicians require data on longer‐term outcomes to ensure that later harms do not arise based on a decision to defer hospitalization. Prior observational studies and randomized controlled clinical trials have proven that appropriately selected individuals face similar rates of complications following acute PE, whether they are managed in or outside of a hospital setting.1116 The key limitation of these earlier efforts, though, was that there was no clear standardized approach to determining whom could be safely managed solely as an outpatient. Finally, our study is unique in that we compare the discriminatory power of these 2 risk‐scoring schemes and illustrate their equivalence. As a scoring system that only requires 3 variables, the PREP is easier and simpler, and may therefore have more clinical utility than the PESI. The high negative predictive value of the PREP suggests that it has potential in identifying patients with acute PE who can safely be managed on an outpatient basis. However, given the complexity of factors associated with the decision for early discharge, these scores should be used in conjunction with, and not supplant, clinical judgment for outpatient management. Of course, formal prospective management trials incorporating both the PREP and PESI are needed to validate this concept.
Why does PREP perform so well despite the fact that it focuses on so few clinical variables? Essentially, the PREP is an effective scoring tool for acute PE because of its ability to identify individuals at risk for progressing to shock. The presence of AMS in acute PE has been associated with a greater likelihood of death, as it likely arises as a consequence of severe shock or RV strain resulting in decreased cerebral blood flow. Alternatively, altered censorium could represent a manifestation of hypoxemia from significant V/Q mismatching and/or pulmonary shunting due to the obstructive clot. This, too, portends a poorer prognosis secondary to impending respiratory failure from hypoxemia. Thus, individuals with an acute PE presenting with altered mentation merit very close observation. Similarly, pending hemodynamic instability is a concerning manifestation that warrants inpatient monitoring.5, 17, 18 At the very minimum, these individuals have RV strain and should therefore be admitted to the hospital to potentially administer more aggressive treatment modalities (ie, thrombolytics or thrombectomy). The last clinical criteria involves the presence of malignancy. The presence of a cancer may serve as a surrogate marker for those at increased risk for early recurrent thromboembolic phenomena, since malignancy is associated with a hypercoagulable state.17, 19 Perhaps there is a threshold whereby accumulating clot resulting in RV strain ensues with subsequent poorer outcomes. Thus, it clinically and physiologically seems logical that, in the absence of any of these findings, patients with acute PE will have lower mortality rates.
Thus far, other methods used for risk stratification may either be expensive, not really obtainable, or not routinely available at the time of presentation. For example, confirmation of RV strain with an echocardiogram requires a skilled technician and interpreter. In contrast, both the PESI and PREP are scored based on multiple clinical findings. Hence, they are not dependent upon a single test to determine outcome, but on various clinical variables making these scoring tools comprehensive, simple, and reliable approaches of recognizing low‐risk patients.
Our analysis has several limitations. First, the retrospective nature of this analysis subjects it to multiple forms of bias. We attempted to eliminate these biases by defining, a priori, the time frame from which vital signs can be used during scoring. We also used all‐cause mortality as our primary endpoint to minimize the possibility of ascertainment bias. However, this type of bias could not be completely eliminated since data collected was not specifically for the purpose of this study. Second, this single‐center study may limit the generalizability of these findings; yet, the diversity of patients admitted to this 900‐bed, tertiary care facility, as well as the inclusion of both inpatients and outpatients, helps to mitigate this concern. Third, the exclusion of individuals with expectant deaths within <30 days limits the applicability of these findings to this group. We chose to exclude persons with anticipated short‐term mortality to reduce the tally of patients who did not receive therapeutic treatment (ie, those transitioned to comfort care). Fourth, the use of the Social Security Death Index objectively determines death status for all‐cause mortality but cannot delineate cause‐specific death. Consequently, death strictly due to PE could not be assessed. Fifth, the original investigators for PREP assessed the PREP score with and without BNP and left‐to‐right ventricular diameter ratios. Although their results demonstrated similar AUROCs for the PREP score with and without BNP to predict 30‐day outcomes, this was a finding we could not confirm due to inconsistencies in measuring BNP and echocardiograms in our cohort. Also, our post hoc power analysis demonstrates that our findings may be limited by sample size. The lack of statistically significant differences between the PESI and the PREP may, in fact, be due to the small sample size versus true effect. Finally, tolerance for medical therapy and compliance with treatment were not documented and, therefore, were immeasurable. Poor compliance to anticoagulants or intolerability increases risk for recurrent PE, while excessive anticoagulation increases likelihood of bleeding.
In summary, the PREP and PESI can both safely predict 30‐day and 90‐day outcomes. However, the simplicity of the PREP renders it more clinician friendly. The fact that only 3 clinical noninvasive variables are required would ultimately make it the preferred bedside tool to risk stratify patients for acute PE. The high negative predictive value and comparable AUROCs establishes the effectiveness of these 2 scoring systems in recognizing low‐risk patients. Irrespective of the clinician's choice to use 1 tool over the other, both have potential for clinical application at the bedside and in clinical trials. Nevertheless, further evidence is required before they are utilized to triage patients for outpatient therapy.
- .Pulmonary embolism: what have we learned since Virchow? Natural history, pathophysiology, and diagnosis.Chest.2002;122:1440–1456.
- ,,, et al.The incidence and prognostic significance of elevated cardiac troponins in patients with submassive pulmonary embolism.J Thromb Haemost.2005;3:508–513.
- ,,, et al.Biomarker‐based risk assessment model in acute pulmonary embolism.Eur Heart J.2005;26:2166–2172.
- ,.Cardiac biomarkers for risk stratification of patients with acute pulmonary embolism.Circulation.2003;108:2191–2194.
- ,,, et al.Prognostic role of echocardiography among patients with acute pulmonary embolism and a systolic arterial pressure of 90 mm Hg or higher.Arch Intern Med.2005;165:1777–1781.
- ,,, et al.Derivation and validation of a prognostic model for pulmonary embolism.Am J Respir Crit Care Med.2005;172:1041–1046.
- ,,.The validation and reproducibility of the pulmonary embolism severity index.J Thromb Haemost.2010;8:1509–1514.
- ,,, et al.Prognostic factors for pulmonary embolism: the prep study, a prospective multicenter cohort study.Am J Respir Crit Care Med.2010;181:168–173.
- ,.A method of comparing the areas under receiver operating characteristic curves derived from the same cases.Radiology.1983;148:839–843.
- ,,, et al.Prospective validation of the pulmonary embolism severity index. A clinical prognostic model for pulmonary embolism.Thromb Haemost.2008;100:943–948.
- ,,, et al.Expanding eligibility for outpatient treatment of deep venous thrombosis and pulmonary embolism with low‐molecular‐weight heparin: a comparison of patient self‐injection with homecare injection.Arch Intern Med.1998;158:1809–1812.
- ,,, et al.Outpatient treatment of pulmonary embolism with dalteparin.Thromb Haemost.2000;83:209–211.
- ,,, et al.Outpatient treatment of pulmonary embolism is feasible and safe in a substantial proportion of patients.J Thromb Haemost.2003;1:186–187.
- ,,, et al.A randomized trial comparing 2 low‐molecular‐weight heparins for the outpatient treatment of deep vein thrombosis and pulmonary embolism.Arch Intern Med.2005;165:733–738.
- ,,, et al.Early discharge of patients with pulmonary embolism: a two‐phase observational study.Eur Respir J.2007;30:708–714.
- ,,, et al.Home treatment in pulmonary embolism.Thromb Res.2010;126:e1–e5.
- ,,.Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER).Lancet.1999;353:1386–1389.
- ,,, et al.Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta‐analysis of the randomized controlled trials.Circulation.2004;110:744–749.
- ,,, et al.Predictors of survival after deep vein thrombosis and pulmonary embolism: a population‐based, cohort study.Arch Intern Med.1999;159:445–453.
Acute pulmonary embolism (PE) is associated with significant morbidity and mortality.1 While expeditious diagnosis and management results in reduced mortality, the ability to rapidly and accurately identify those at increased risk for death remains elusive. Multiple studies have utilized various biomarkers as risk stratification tools, however, these approaches have proven to have many limitations. For example, both serum brain natriuretic peptide (BNP) and troponin levels have been studied as possible risk stratification tools. Those with elevated levels of these following a PE may have concomitant right ventricular (RV) dysfunction and/or hemodynamic instability. Thus, they may face a greater risk for cardiovascular collapse and death. The low positive predictive value of these biomarkers (14%‐44%) has limited their clinical utility.24 Furthermore, imaging modalities, such as echocardiography, which is considered the clinical gold standard for determining the presence of acute RV dysfunction in PE, may not be readily available and may require special expertise for interpretation.5
Conversely, the need to identify acute PE patients at low risk for death is just as important. Recent studies suggest that carefully selected patients can successfully be managed as outpatients which can subsequently lead to significant cost savings and patient satisfaction. Movement towards enhanced outpatient resources and the advent of subcutaneous anticoagulants have made outpatient management of acute PE an appealing possibility. However, proper education, close follow‐up, and a rigorous selection process to recognize those at minimal risk for a fatal complication must all be available before clinicians prematurely discharge these patients to home.
Recently, clinical scoring tools have been developed to aid in risk stratifying patients with acute PE to accurately determine patient outcome. The pulmonary embolism severity index (PESI) is a reproducible scoring system that accurately predicts 30‐day and 90‐day mortality.6, 7 It consists of 11 clinical variables that can be quickly assessed at the time of diagnosis (Table 1A). The fact that biomarkers and imaging technology, such as echocardiography, are unnecessary to compute a PESI score demonstrates the appeal of this system. Similar to the PESI, Sanchez et al.8 have proposed the prognosis in pulmonary embolism (PREP) score as an alternate clinical risk tool in PE (Table 1B). Contrary to PESI, the PREP only uses 3 clinical variables to accurately predict vital outcome with an area under the receiver operating characteristic (AUROC) curve of 0.73 (95% confidence interval [CI], 0.65‐0.82). While both scoring systems have been developed to predict 30‐day mortality in acute PE, the comparative validity of these prognostic tools has not been assessed.
| Predictors | Points Assigned |
|---|---|
| |
| Demographic characteristics | |
| Age (yr) | Age (yr) |
| Male sex | +10 |
| Comorbid conditions | |
| Cancer | +30 |
| Heart failure | +10 |
| Chronic lung disease | +10 |
| Clinical findings | |
| Pulse 110 beats/min | +20 |
| Systolic blood pressure <100 mm Hg | +30 |
| Respiratory rate 30 breaths/min | +20 |
| Temperature <36C | +20 |
| Altered mental status* | +60 |
| Arterial oxygen saturation <90% | +20 |
| Prognostic Factor | Points Assigned |
|---|---|
| |
| Altered mental status* | +10 |
| Cardiogenic shock (systolic blood pressure <90 mm Hg) | +6 |
| Cancer | +6 |
We hypothesized that the PESI more precisely risk stratifies the risk for death in acute PE compared to the PREP. Furthermore, we theorized that the PESI more reliably predicts not only 30‐day but also 90‐day mortality. To test our hypothesis, we performed a retrospective analysis, of all consecutive patients diagnosed with acute PE at our hospital, to compare the prognostic accuracy of these 2 scoring systems.
METHODS
Subjects and Definitions
Between October 2007 and February 2009, adults (age 18 years) diagnosed the day prior with acute PE were identified on a daily basis. This study cohort has been described elsewhere.7 Patients with newly diagnosed PE were eligible for enrollment. Those expected to die within 30 days of their acute PE, such as individuals suffering from a terminal condition (metastatic cancer) or critical illness being transitioned to comfort care, were excluded (n = 32). Patients with multiple admissions for acute PE were included only during the first episode. PE was diagnosed using objective criteria through 1 of the following modalities: high probability ventilation‐perfusion (V/Q) scintigraphy, computed tomography (CT) of the chest with PE protocol, or magnetic resonance imaging (MRI) of the chest. A list of patients who had the above imaging studies to evaluate for PE was provided to study personnel daily by the radiology department; this list was generated every morning and consisted of the day prior's studies. Patient management was not influenced by the research team and was the responsibility of the primary team. This study was approved by our local institutional review board and consent was not required.
We calculated the PESI as described by Aujesky and colleagues.6 For outpatients admitted with acute PE, clinical findings available just prior to, and after, diagnosis were used for scoring. For inpatients diagnosed with PE, clinical findings available during the 24 hours just prior to diagnosis were included. Raw PESI scores were converted to risk class (I‐V), and then further dichotomized into low‐risk (class I‐II) and high‐risk (class III‐V) groups (Table 2). The PREP score was computed based on the presence of altered mental status (AMS), cancer, and cardiogenic shock defined as a systolic blood pressure <90 mm Hg (Table 1B). A raw PREP score of <7 was then characterized as low risk for mortality, while scores 7 were considered high risk.
| PESI Score | Class | n | 30‐Day Mortality by Class (%) | 90‐Day Mortality by Class (%) | Low vs High Risk |
|---|---|---|---|---|---|
| |||||
| 65 | I | 49 | 0 (0.0) | 0 (0.0) | Low |
| 66‐85 | II | 59 | 0 (0.0) | 0 (0.0) | |
| 86‐105 | III | 60 | 0 (0.0) | 0 (0.0) | High |
| 106‐125 | IV | 56 | 2 (3.4) | 4 (6.9) | |
| >125 | V | 69 | 7 (9.2) | 8 (10.5) | |
Finally, the PESI and PREP scores were compared based on their ability to predict all‐cause 30‐day and 90‐day mortality. To determine vital status and date of death, we reviewed the Social Security Death Index 90 days after enrollment of all subjects was completed.
Statistical Analysis
To assess the predictive ability of the 2 scoring tools for death, we determined the negative predictive value and computed the AUROC curves for both scoring systems. AUROC curves were constructed for raw scores and when scores were further segregated by class and risk groups. Additionally, 95% CIs were estimated to determine the accuracy of the discriminatory power of the PESI score versus the PREP score.
Post hoc, we calculated the power of our study to assess whether the difference noted in AUROC curves between the PESI and PREP was adequate to truly determine statistical significance. We used methodology described by Hanley and McNeil to compare continuous values.9 Assuming an alpha of 0.05 and a 20% difference in the AUROC curves, as described in our results, the power in our study was 0.35. Therefore, an approximate sample size of 1000 would be necessary to determine statistical significance. This analysis was performed using Power Analysis and Sample Size (PASS) 11.
RESULTS
The final cohort included 302 subjects (mean age: 59.7 17.2 years; 44.0% males). As Table 3 reveals, the majority of PEs was diagnosed via CT scan (76%). On presentation, 6.6% had cardiogenic shock, while 5.0% had altered censorium. In terms of comorbid conditions, 25.2% had congestive heart failure, 25.2% had cancer, and 22.2% had a prior venous thromboembolic event. Overall, 3.0% and 4.0% met our primary outcomes of death within 30‐days and 90‐days of their acute PEs, respectively.
| |
| Demographics | |
| Age (yr), mean SD | 59.7 17.2 |
| Male sex, % | 44% |
| Diagnostic methodology | |
| CT chest, n (%) | 230 (76.2) |
| V/Q scan, n (%) | 71 (23.5) |
| MRA chest, n (%) | 1 (0.3) |
| Comorbidities | |
| Malignancy, n (%) | 76 (25.2) |
| Congestive heart failure, n (%) | 76 (25.2) |
| Chronic lung disease, n (%) | 72 (23.8) |
| Recent orthopedic surgery, n (%) | 22 (7.3) |
| Prior cerebrovascular accident, n (%) | 31 (10.3) |
| Prior venous thromboembolic disease, n (%) | 67 (22.2) |
The rates of 30‐day and 90‐day mortality, respectively, increased with increasing score for both the PESI and the PREP. No patients in PESI class I died by either time point, while 9.2% of PESI class V subjects expired by 30 days (P < 0.0001) and 10.5% died by 90 days (P = 0.003) (Table 2). Based on PESI, 30‐day death rates were 4.6% in the high‐risk cohort versus 0% in the low‐risk group (P = 0.023). Conversely, 7.1% of high‐risk PREP subjects died by day 30 versus 1% of low‐risk subjects (P = 0.004) (Figure 1A). Those stratified into the PESI high‐risk group had a 90‐day mortality of 6.2% versus 0% for the low‐risk group (P = 0.008) versus 9.1% in those deemed high risk by PREP, as compared to 1.5% of those scored as low risk by PREP (P = 0.001) (Figure 1B).

Regarding the 30‐day mortality, the negative predictive value of the PESI was 100% (95% CI, 98.6%‐100%) while that for PREP was 99.0% (95% CI, 97.6%‐99.7%); the ability of the PREP to predict 30‐day mortality was similar to the PESI (Table 4). The AUROCs for PESI and PREP for predicting 30‐day death were also equivalent; for the raw PESI score, this measured 0.858 (95% CI, 0.773‐0.943), compared to 0.719 (95% CI, 0.563‐0.875) for PREP. When these scores were dichotomized to high‐risk versus low‐risk groups, the AUROC for the PESI was 0.684 (95% CI, 0.559‐0.810) and 0.732 (95% CI, 0.571‐0.893) for PREP.
| 30‐Day Mortality | 90‐Day Mortality | |||
|---|---|---|---|---|
| Scoring System | AUROC | 95% CI | AUROC | 95% CI |
| ||||
| Raw PESI | 0.858 | 0.773‐0.943 | 0.835 | 0.762‐0.907 |
| PESI class | 0.835 | 0.756‐0.914 | 0.813 | 0.738‐0.888 |
| PESI high vs low risk | 0.684 | 0.559‐0.810 | 0.686 | 0.576‐0.796 |
| Raw PREP | 0.719 | 0.563‐0.875 | 0.704 | 0.564‐0.844 |
| PREP high vs low risk | 0.732 | 0.571‐0.893 | 0.720 | 0.574‐0.865 |
In terms of 90‐day mortality, the negative predictive values of PESI and PREP did not change: 100% (95% CI, 97.4%‐100%) and 98.5% (95% CI, 96.9%‐99.5%), respectively. The ability of PESI and PREP as predictors of 90‐day mortality was equivalent (Table 4). Here, the AUROC for the raw PESI score remained excellent at 0.835 (97% CI, 0.762‐0.907). The AUROC for PREP was akin to that of PESI at 0.704 (95% CI, 0.564‐0.844). Segregating scores into high‐risk versus low‐risk groups demonstrated that the AUROC for PESI was 0.686 (95% CI, 0.576‐0.796) compared to 0.720 (95% CI, 0.574‐0.865) for PREP.
DISCUSSION
This retrospective analysis of patients with acute PE confirms that both the PESI and the PREP are accurate scoring tools for identifying patients at low risk of death. Under both rubrics, as the score increases, the likelihood of death also increases. More importantly, we demonstrate that the negative predictive value for both the PREP and PESI are excellent. Thus, these scoring tools can distinguish those at higher risk for death versus those at low risk in a simple‐to‐apply manner. In comparing these 2 scoring systems, the PREP comparably identifies acute PE patients at risk for death when contrasted with the PESI. Given the fewer required scoring points to calculate PREP and its ability to accurately predict clinically relevant outcomes, this simpler scoring system may have greater clinical utility.
Prior studies have validated the PESI as a risk stratification tool to predict 30‐day and 90‐day mortalities. In their original derivation of the PESI, Aujesky et al. demonstrated that higher PESI scores correlated with death at 30 days.6 Acute PE patients classified into risk class I had a short‐term mortality rate of 1.1% compared to nearly 25% of patients risk stratified into risk class V. The same authors subsequently verified that there is a linear relationship between PESI score and risk of death at 90 days.10 We have also confirmed the accuracy of the PESI for identifying persons at high risk for death and documented the limited interobserver variability in this tool.7 In combination, there is evidence that the PESI can accurately predict vital outcome. Despite the effectiveness of the PESI, it is a somewhat cumbersome scoring system. It requires gathering information on 11 clinical variables, each with a different score allocation to ultimately compute the PESI score. In contrast, the PREP only requires knowing 3 clinical variables: presence of cancer, mental status, and the presence of cardiogenic shock. Akin to the PESI, the PREP and mortality are linearly related, where higher PREP scores result in higher 30‐day and 90‐day mortalities.
Our analysis helps expand the evidence regarding clinical risk stratification in PE in several ways. First, we verify that both the PESI and PREP are accurate predictors of short‐term mortality. While this has been accomplished for the PESI in prior studies, to our knowledge, this is the first confirmatory study for PREP's utility as a risk stratification tool. Second, we demonstrate that PREP is also an accurate predictor of intermediate‐term mortality. If the eventual goal is to develop tools that allow for the initial outpatient management of acute PE, clinicians require data on longer‐term outcomes to ensure that later harms do not arise based on a decision to defer hospitalization. Prior observational studies and randomized controlled clinical trials have proven that appropriately selected individuals face similar rates of complications following acute PE, whether they are managed in or outside of a hospital setting.1116 The key limitation of these earlier efforts, though, was that there was no clear standardized approach to determining whom could be safely managed solely as an outpatient. Finally, our study is unique in that we compare the discriminatory power of these 2 risk‐scoring schemes and illustrate their equivalence. As a scoring system that only requires 3 variables, the PREP is easier and simpler, and may therefore have more clinical utility than the PESI. The high negative predictive value of the PREP suggests that it has potential in identifying patients with acute PE who can safely be managed on an outpatient basis. However, given the complexity of factors associated with the decision for early discharge, these scores should be used in conjunction with, and not supplant, clinical judgment for outpatient management. Of course, formal prospective management trials incorporating both the PREP and PESI are needed to validate this concept.
Why does PREP perform so well despite the fact that it focuses on so few clinical variables? Essentially, the PREP is an effective scoring tool for acute PE because of its ability to identify individuals at risk for progressing to shock. The presence of AMS in acute PE has been associated with a greater likelihood of death, as it likely arises as a consequence of severe shock or RV strain resulting in decreased cerebral blood flow. Alternatively, altered censorium could represent a manifestation of hypoxemia from significant V/Q mismatching and/or pulmonary shunting due to the obstructive clot. This, too, portends a poorer prognosis secondary to impending respiratory failure from hypoxemia. Thus, individuals with an acute PE presenting with altered mentation merit very close observation. Similarly, pending hemodynamic instability is a concerning manifestation that warrants inpatient monitoring.5, 17, 18 At the very minimum, these individuals have RV strain and should therefore be admitted to the hospital to potentially administer more aggressive treatment modalities (ie, thrombolytics or thrombectomy). The last clinical criteria involves the presence of malignancy. The presence of a cancer may serve as a surrogate marker for those at increased risk for early recurrent thromboembolic phenomena, since malignancy is associated with a hypercoagulable state.17, 19 Perhaps there is a threshold whereby accumulating clot resulting in RV strain ensues with subsequent poorer outcomes. Thus, it clinically and physiologically seems logical that, in the absence of any of these findings, patients with acute PE will have lower mortality rates.
Thus far, other methods used for risk stratification may either be expensive, not really obtainable, or not routinely available at the time of presentation. For example, confirmation of RV strain with an echocardiogram requires a skilled technician and interpreter. In contrast, both the PESI and PREP are scored based on multiple clinical findings. Hence, they are not dependent upon a single test to determine outcome, but on various clinical variables making these scoring tools comprehensive, simple, and reliable approaches of recognizing low‐risk patients.
Our analysis has several limitations. First, the retrospective nature of this analysis subjects it to multiple forms of bias. We attempted to eliminate these biases by defining, a priori, the time frame from which vital signs can be used during scoring. We also used all‐cause mortality as our primary endpoint to minimize the possibility of ascertainment bias. However, this type of bias could not be completely eliminated since data collected was not specifically for the purpose of this study. Second, this single‐center study may limit the generalizability of these findings; yet, the diversity of patients admitted to this 900‐bed, tertiary care facility, as well as the inclusion of both inpatients and outpatients, helps to mitigate this concern. Third, the exclusion of individuals with expectant deaths within <30 days limits the applicability of these findings to this group. We chose to exclude persons with anticipated short‐term mortality to reduce the tally of patients who did not receive therapeutic treatment (ie, those transitioned to comfort care). Fourth, the use of the Social Security Death Index objectively determines death status for all‐cause mortality but cannot delineate cause‐specific death. Consequently, death strictly due to PE could not be assessed. Fifth, the original investigators for PREP assessed the PREP score with and without BNP and left‐to‐right ventricular diameter ratios. Although their results demonstrated similar AUROCs for the PREP score with and without BNP to predict 30‐day outcomes, this was a finding we could not confirm due to inconsistencies in measuring BNP and echocardiograms in our cohort. Also, our post hoc power analysis demonstrates that our findings may be limited by sample size. The lack of statistically significant differences between the PESI and the PREP may, in fact, be due to the small sample size versus true effect. Finally, tolerance for medical therapy and compliance with treatment were not documented and, therefore, were immeasurable. Poor compliance to anticoagulants or intolerability increases risk for recurrent PE, while excessive anticoagulation increases likelihood of bleeding.
In summary, the PREP and PESI can both safely predict 30‐day and 90‐day outcomes. However, the simplicity of the PREP renders it more clinician friendly. The fact that only 3 clinical noninvasive variables are required would ultimately make it the preferred bedside tool to risk stratify patients for acute PE. The high negative predictive value and comparable AUROCs establishes the effectiveness of these 2 scoring systems in recognizing low‐risk patients. Irrespective of the clinician's choice to use 1 tool over the other, both have potential for clinical application at the bedside and in clinical trials. Nevertheless, further evidence is required before they are utilized to triage patients for outpatient therapy.
Acute pulmonary embolism (PE) is associated with significant morbidity and mortality.1 While expeditious diagnosis and management results in reduced mortality, the ability to rapidly and accurately identify those at increased risk for death remains elusive. Multiple studies have utilized various biomarkers as risk stratification tools, however, these approaches have proven to have many limitations. For example, both serum brain natriuretic peptide (BNP) and troponin levels have been studied as possible risk stratification tools. Those with elevated levels of these following a PE may have concomitant right ventricular (RV) dysfunction and/or hemodynamic instability. Thus, they may face a greater risk for cardiovascular collapse and death. The low positive predictive value of these biomarkers (14%‐44%) has limited their clinical utility.24 Furthermore, imaging modalities, such as echocardiography, which is considered the clinical gold standard for determining the presence of acute RV dysfunction in PE, may not be readily available and may require special expertise for interpretation.5
Conversely, the need to identify acute PE patients at low risk for death is just as important. Recent studies suggest that carefully selected patients can successfully be managed as outpatients which can subsequently lead to significant cost savings and patient satisfaction. Movement towards enhanced outpatient resources and the advent of subcutaneous anticoagulants have made outpatient management of acute PE an appealing possibility. However, proper education, close follow‐up, and a rigorous selection process to recognize those at minimal risk for a fatal complication must all be available before clinicians prematurely discharge these patients to home.
Recently, clinical scoring tools have been developed to aid in risk stratifying patients with acute PE to accurately determine patient outcome. The pulmonary embolism severity index (PESI) is a reproducible scoring system that accurately predicts 30‐day and 90‐day mortality.6, 7 It consists of 11 clinical variables that can be quickly assessed at the time of diagnosis (Table 1A). The fact that biomarkers and imaging technology, such as echocardiography, are unnecessary to compute a PESI score demonstrates the appeal of this system. Similar to the PESI, Sanchez et al.8 have proposed the prognosis in pulmonary embolism (PREP) score as an alternate clinical risk tool in PE (Table 1B). Contrary to PESI, the PREP only uses 3 clinical variables to accurately predict vital outcome with an area under the receiver operating characteristic (AUROC) curve of 0.73 (95% confidence interval [CI], 0.65‐0.82). While both scoring systems have been developed to predict 30‐day mortality in acute PE, the comparative validity of these prognostic tools has not been assessed.
| Predictors | Points Assigned |
|---|---|
| |
| Demographic characteristics | |
| Age (yr) | Age (yr) |
| Male sex | +10 |
| Comorbid conditions | |
| Cancer | +30 |
| Heart failure | +10 |
| Chronic lung disease | +10 |
| Clinical findings | |
| Pulse 110 beats/min | +20 |
| Systolic blood pressure <100 mm Hg | +30 |
| Respiratory rate 30 breaths/min | +20 |
| Temperature <36C | +20 |
| Altered mental status* | +60 |
| Arterial oxygen saturation <90% | +20 |
| Prognostic Factor | Points Assigned |
|---|---|
| |
| Altered mental status* | +10 |
| Cardiogenic shock (systolic blood pressure <90 mm Hg) | +6 |
| Cancer | +6 |
We hypothesized that the PESI more precisely risk stratifies the risk for death in acute PE compared to the PREP. Furthermore, we theorized that the PESI more reliably predicts not only 30‐day but also 90‐day mortality. To test our hypothesis, we performed a retrospective analysis, of all consecutive patients diagnosed with acute PE at our hospital, to compare the prognostic accuracy of these 2 scoring systems.
METHODS
Subjects and Definitions
Between October 2007 and February 2009, adults (age 18 years) diagnosed the day prior with acute PE were identified on a daily basis. This study cohort has been described elsewhere.7 Patients with newly diagnosed PE were eligible for enrollment. Those expected to die within 30 days of their acute PE, such as individuals suffering from a terminal condition (metastatic cancer) or critical illness being transitioned to comfort care, were excluded (n = 32). Patients with multiple admissions for acute PE were included only during the first episode. PE was diagnosed using objective criteria through 1 of the following modalities: high probability ventilation‐perfusion (V/Q) scintigraphy, computed tomography (CT) of the chest with PE protocol, or magnetic resonance imaging (MRI) of the chest. A list of patients who had the above imaging studies to evaluate for PE was provided to study personnel daily by the radiology department; this list was generated every morning and consisted of the day prior's studies. Patient management was not influenced by the research team and was the responsibility of the primary team. This study was approved by our local institutional review board and consent was not required.
We calculated the PESI as described by Aujesky and colleagues.6 For outpatients admitted with acute PE, clinical findings available just prior to, and after, diagnosis were used for scoring. For inpatients diagnosed with PE, clinical findings available during the 24 hours just prior to diagnosis were included. Raw PESI scores were converted to risk class (I‐V), and then further dichotomized into low‐risk (class I‐II) and high‐risk (class III‐V) groups (Table 2). The PREP score was computed based on the presence of altered mental status (AMS), cancer, and cardiogenic shock defined as a systolic blood pressure <90 mm Hg (Table 1B). A raw PREP score of <7 was then characterized as low risk for mortality, while scores 7 were considered high risk.
| PESI Score | Class | n | 30‐Day Mortality by Class (%) | 90‐Day Mortality by Class (%) | Low vs High Risk |
|---|---|---|---|---|---|
| |||||
| 65 | I | 49 | 0 (0.0) | 0 (0.0) | Low |
| 66‐85 | II | 59 | 0 (0.0) | 0 (0.0) | |
| 86‐105 | III | 60 | 0 (0.0) | 0 (0.0) | High |
| 106‐125 | IV | 56 | 2 (3.4) | 4 (6.9) | |
| >125 | V | 69 | 7 (9.2) | 8 (10.5) | |
Finally, the PESI and PREP scores were compared based on their ability to predict all‐cause 30‐day and 90‐day mortality. To determine vital status and date of death, we reviewed the Social Security Death Index 90 days after enrollment of all subjects was completed.
Statistical Analysis
To assess the predictive ability of the 2 scoring tools for death, we determined the negative predictive value and computed the AUROC curves for both scoring systems. AUROC curves were constructed for raw scores and when scores were further segregated by class and risk groups. Additionally, 95% CIs were estimated to determine the accuracy of the discriminatory power of the PESI score versus the PREP score.
Post hoc, we calculated the power of our study to assess whether the difference noted in AUROC curves between the PESI and PREP was adequate to truly determine statistical significance. We used methodology described by Hanley and McNeil to compare continuous values.9 Assuming an alpha of 0.05 and a 20% difference in the AUROC curves, as described in our results, the power in our study was 0.35. Therefore, an approximate sample size of 1000 would be necessary to determine statistical significance. This analysis was performed using Power Analysis and Sample Size (PASS) 11.
RESULTS
The final cohort included 302 subjects (mean age: 59.7 17.2 years; 44.0% males). As Table 3 reveals, the majority of PEs was diagnosed via CT scan (76%). On presentation, 6.6% had cardiogenic shock, while 5.0% had altered censorium. In terms of comorbid conditions, 25.2% had congestive heart failure, 25.2% had cancer, and 22.2% had a prior venous thromboembolic event. Overall, 3.0% and 4.0% met our primary outcomes of death within 30‐days and 90‐days of their acute PEs, respectively.
| |
| Demographics | |
| Age (yr), mean SD | 59.7 17.2 |
| Male sex, % | 44% |
| Diagnostic methodology | |
| CT chest, n (%) | 230 (76.2) |
| V/Q scan, n (%) | 71 (23.5) |
| MRA chest, n (%) | 1 (0.3) |
| Comorbidities | |
| Malignancy, n (%) | 76 (25.2) |
| Congestive heart failure, n (%) | 76 (25.2) |
| Chronic lung disease, n (%) | 72 (23.8) |
| Recent orthopedic surgery, n (%) | 22 (7.3) |
| Prior cerebrovascular accident, n (%) | 31 (10.3) |
| Prior venous thromboembolic disease, n (%) | 67 (22.2) |
The rates of 30‐day and 90‐day mortality, respectively, increased with increasing score for both the PESI and the PREP. No patients in PESI class I died by either time point, while 9.2% of PESI class V subjects expired by 30 days (P < 0.0001) and 10.5% died by 90 days (P = 0.003) (Table 2). Based on PESI, 30‐day death rates were 4.6% in the high‐risk cohort versus 0% in the low‐risk group (P = 0.023). Conversely, 7.1% of high‐risk PREP subjects died by day 30 versus 1% of low‐risk subjects (P = 0.004) (Figure 1A). Those stratified into the PESI high‐risk group had a 90‐day mortality of 6.2% versus 0% for the low‐risk group (P = 0.008) versus 9.1% in those deemed high risk by PREP, as compared to 1.5% of those scored as low risk by PREP (P = 0.001) (Figure 1B).

Regarding the 30‐day mortality, the negative predictive value of the PESI was 100% (95% CI, 98.6%‐100%) while that for PREP was 99.0% (95% CI, 97.6%‐99.7%); the ability of the PREP to predict 30‐day mortality was similar to the PESI (Table 4). The AUROCs for PESI and PREP for predicting 30‐day death were also equivalent; for the raw PESI score, this measured 0.858 (95% CI, 0.773‐0.943), compared to 0.719 (95% CI, 0.563‐0.875) for PREP. When these scores were dichotomized to high‐risk versus low‐risk groups, the AUROC for the PESI was 0.684 (95% CI, 0.559‐0.810) and 0.732 (95% CI, 0.571‐0.893) for PREP.
| 30‐Day Mortality | 90‐Day Mortality | |||
|---|---|---|---|---|
| Scoring System | AUROC | 95% CI | AUROC | 95% CI |
| ||||
| Raw PESI | 0.858 | 0.773‐0.943 | 0.835 | 0.762‐0.907 |
| PESI class | 0.835 | 0.756‐0.914 | 0.813 | 0.738‐0.888 |
| PESI high vs low risk | 0.684 | 0.559‐0.810 | 0.686 | 0.576‐0.796 |
| Raw PREP | 0.719 | 0.563‐0.875 | 0.704 | 0.564‐0.844 |
| PREP high vs low risk | 0.732 | 0.571‐0.893 | 0.720 | 0.574‐0.865 |
In terms of 90‐day mortality, the negative predictive values of PESI and PREP did not change: 100% (95% CI, 97.4%‐100%) and 98.5% (95% CI, 96.9%‐99.5%), respectively. The ability of PESI and PREP as predictors of 90‐day mortality was equivalent (Table 4). Here, the AUROC for the raw PESI score remained excellent at 0.835 (97% CI, 0.762‐0.907). The AUROC for PREP was akin to that of PESI at 0.704 (95% CI, 0.564‐0.844). Segregating scores into high‐risk versus low‐risk groups demonstrated that the AUROC for PESI was 0.686 (95% CI, 0.576‐0.796) compared to 0.720 (95% CI, 0.574‐0.865) for PREP.
DISCUSSION
This retrospective analysis of patients with acute PE confirms that both the PESI and the PREP are accurate scoring tools for identifying patients at low risk of death. Under both rubrics, as the score increases, the likelihood of death also increases. More importantly, we demonstrate that the negative predictive value for both the PREP and PESI are excellent. Thus, these scoring tools can distinguish those at higher risk for death versus those at low risk in a simple‐to‐apply manner. In comparing these 2 scoring systems, the PREP comparably identifies acute PE patients at risk for death when contrasted with the PESI. Given the fewer required scoring points to calculate PREP and its ability to accurately predict clinically relevant outcomes, this simpler scoring system may have greater clinical utility.
Prior studies have validated the PESI as a risk stratification tool to predict 30‐day and 90‐day mortalities. In their original derivation of the PESI, Aujesky et al. demonstrated that higher PESI scores correlated with death at 30 days.6 Acute PE patients classified into risk class I had a short‐term mortality rate of 1.1% compared to nearly 25% of patients risk stratified into risk class V. The same authors subsequently verified that there is a linear relationship between PESI score and risk of death at 90 days.10 We have also confirmed the accuracy of the PESI for identifying persons at high risk for death and documented the limited interobserver variability in this tool.7 In combination, there is evidence that the PESI can accurately predict vital outcome. Despite the effectiveness of the PESI, it is a somewhat cumbersome scoring system. It requires gathering information on 11 clinical variables, each with a different score allocation to ultimately compute the PESI score. In contrast, the PREP only requires knowing 3 clinical variables: presence of cancer, mental status, and the presence of cardiogenic shock. Akin to the PESI, the PREP and mortality are linearly related, where higher PREP scores result in higher 30‐day and 90‐day mortalities.
Our analysis helps expand the evidence regarding clinical risk stratification in PE in several ways. First, we verify that both the PESI and PREP are accurate predictors of short‐term mortality. While this has been accomplished for the PESI in prior studies, to our knowledge, this is the first confirmatory study for PREP's utility as a risk stratification tool. Second, we demonstrate that PREP is also an accurate predictor of intermediate‐term mortality. If the eventual goal is to develop tools that allow for the initial outpatient management of acute PE, clinicians require data on longer‐term outcomes to ensure that later harms do not arise based on a decision to defer hospitalization. Prior observational studies and randomized controlled clinical trials have proven that appropriately selected individuals face similar rates of complications following acute PE, whether they are managed in or outside of a hospital setting.1116 The key limitation of these earlier efforts, though, was that there was no clear standardized approach to determining whom could be safely managed solely as an outpatient. Finally, our study is unique in that we compare the discriminatory power of these 2 risk‐scoring schemes and illustrate their equivalence. As a scoring system that only requires 3 variables, the PREP is easier and simpler, and may therefore have more clinical utility than the PESI. The high negative predictive value of the PREP suggests that it has potential in identifying patients with acute PE who can safely be managed on an outpatient basis. However, given the complexity of factors associated with the decision for early discharge, these scores should be used in conjunction with, and not supplant, clinical judgment for outpatient management. Of course, formal prospective management trials incorporating both the PREP and PESI are needed to validate this concept.
Why does PREP perform so well despite the fact that it focuses on so few clinical variables? Essentially, the PREP is an effective scoring tool for acute PE because of its ability to identify individuals at risk for progressing to shock. The presence of AMS in acute PE has been associated with a greater likelihood of death, as it likely arises as a consequence of severe shock or RV strain resulting in decreased cerebral blood flow. Alternatively, altered censorium could represent a manifestation of hypoxemia from significant V/Q mismatching and/or pulmonary shunting due to the obstructive clot. This, too, portends a poorer prognosis secondary to impending respiratory failure from hypoxemia. Thus, individuals with an acute PE presenting with altered mentation merit very close observation. Similarly, pending hemodynamic instability is a concerning manifestation that warrants inpatient monitoring.5, 17, 18 At the very minimum, these individuals have RV strain and should therefore be admitted to the hospital to potentially administer more aggressive treatment modalities (ie, thrombolytics or thrombectomy). The last clinical criteria involves the presence of malignancy. The presence of a cancer may serve as a surrogate marker for those at increased risk for early recurrent thromboembolic phenomena, since malignancy is associated with a hypercoagulable state.17, 19 Perhaps there is a threshold whereby accumulating clot resulting in RV strain ensues with subsequent poorer outcomes. Thus, it clinically and physiologically seems logical that, in the absence of any of these findings, patients with acute PE will have lower mortality rates.
Thus far, other methods used for risk stratification may either be expensive, not really obtainable, or not routinely available at the time of presentation. For example, confirmation of RV strain with an echocardiogram requires a skilled technician and interpreter. In contrast, both the PESI and PREP are scored based on multiple clinical findings. Hence, they are not dependent upon a single test to determine outcome, but on various clinical variables making these scoring tools comprehensive, simple, and reliable approaches of recognizing low‐risk patients.
Our analysis has several limitations. First, the retrospective nature of this analysis subjects it to multiple forms of bias. We attempted to eliminate these biases by defining, a priori, the time frame from which vital signs can be used during scoring. We also used all‐cause mortality as our primary endpoint to minimize the possibility of ascertainment bias. However, this type of bias could not be completely eliminated since data collected was not specifically for the purpose of this study. Second, this single‐center study may limit the generalizability of these findings; yet, the diversity of patients admitted to this 900‐bed, tertiary care facility, as well as the inclusion of both inpatients and outpatients, helps to mitigate this concern. Third, the exclusion of individuals with expectant deaths within <30 days limits the applicability of these findings to this group. We chose to exclude persons with anticipated short‐term mortality to reduce the tally of patients who did not receive therapeutic treatment (ie, those transitioned to comfort care). Fourth, the use of the Social Security Death Index objectively determines death status for all‐cause mortality but cannot delineate cause‐specific death. Consequently, death strictly due to PE could not be assessed. Fifth, the original investigators for PREP assessed the PREP score with and without BNP and left‐to‐right ventricular diameter ratios. Although their results demonstrated similar AUROCs for the PREP score with and without BNP to predict 30‐day outcomes, this was a finding we could not confirm due to inconsistencies in measuring BNP and echocardiograms in our cohort. Also, our post hoc power analysis demonstrates that our findings may be limited by sample size. The lack of statistically significant differences between the PESI and the PREP may, in fact, be due to the small sample size versus true effect. Finally, tolerance for medical therapy and compliance with treatment were not documented and, therefore, were immeasurable. Poor compliance to anticoagulants or intolerability increases risk for recurrent PE, while excessive anticoagulation increases likelihood of bleeding.
In summary, the PREP and PESI can both safely predict 30‐day and 90‐day outcomes. However, the simplicity of the PREP renders it more clinician friendly. The fact that only 3 clinical noninvasive variables are required would ultimately make it the preferred bedside tool to risk stratify patients for acute PE. The high negative predictive value and comparable AUROCs establishes the effectiveness of these 2 scoring systems in recognizing low‐risk patients. Irrespective of the clinician's choice to use 1 tool over the other, both have potential for clinical application at the bedside and in clinical trials. Nevertheless, further evidence is required before they are utilized to triage patients for outpatient therapy.
- .Pulmonary embolism: what have we learned since Virchow? Natural history, pathophysiology, and diagnosis.Chest.2002;122:1440–1456.
- ,,, et al.The incidence and prognostic significance of elevated cardiac troponins in patients with submassive pulmonary embolism.J Thromb Haemost.2005;3:508–513.
- ,,, et al.Biomarker‐based risk assessment model in acute pulmonary embolism.Eur Heart J.2005;26:2166–2172.
- ,.Cardiac biomarkers for risk stratification of patients with acute pulmonary embolism.Circulation.2003;108:2191–2194.
- ,,, et al.Prognostic role of echocardiography among patients with acute pulmonary embolism and a systolic arterial pressure of 90 mm Hg or higher.Arch Intern Med.2005;165:1777–1781.
- ,,, et al.Derivation and validation of a prognostic model for pulmonary embolism.Am J Respir Crit Care Med.2005;172:1041–1046.
- ,,.The validation and reproducibility of the pulmonary embolism severity index.J Thromb Haemost.2010;8:1509–1514.
- ,,, et al.Prognostic factors for pulmonary embolism: the prep study, a prospective multicenter cohort study.Am J Respir Crit Care Med.2010;181:168–173.
- ,.A method of comparing the areas under receiver operating characteristic curves derived from the same cases.Radiology.1983;148:839–843.
- ,,, et al.Prospective validation of the pulmonary embolism severity index. A clinical prognostic model for pulmonary embolism.Thromb Haemost.2008;100:943–948.
- ,,, et al.Expanding eligibility for outpatient treatment of deep venous thrombosis and pulmonary embolism with low‐molecular‐weight heparin: a comparison of patient self‐injection with homecare injection.Arch Intern Med.1998;158:1809–1812.
- ,,, et al.Outpatient treatment of pulmonary embolism with dalteparin.Thromb Haemost.2000;83:209–211.
- ,,, et al.Outpatient treatment of pulmonary embolism is feasible and safe in a substantial proportion of patients.J Thromb Haemost.2003;1:186–187.
- ,,, et al.A randomized trial comparing 2 low‐molecular‐weight heparins for the outpatient treatment of deep vein thrombosis and pulmonary embolism.Arch Intern Med.2005;165:733–738.
- ,,, et al.Early discharge of patients with pulmonary embolism: a two‐phase observational study.Eur Respir J.2007;30:708–714.
- ,,, et al.Home treatment in pulmonary embolism.Thromb Res.2010;126:e1–e5.
- ,,.Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER).Lancet.1999;353:1386–1389.
- ,,, et al.Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta‐analysis of the randomized controlled trials.Circulation.2004;110:744–749.
- ,,, et al.Predictors of survival after deep vein thrombosis and pulmonary embolism: a population‐based, cohort study.Arch Intern Med.1999;159:445–453.
- .Pulmonary embolism: what have we learned since Virchow? Natural history, pathophysiology, and diagnosis.Chest.2002;122:1440–1456.
- ,,, et al.The incidence and prognostic significance of elevated cardiac troponins in patients with submassive pulmonary embolism.J Thromb Haemost.2005;3:508–513.
- ,,, et al.Biomarker‐based risk assessment model in acute pulmonary embolism.Eur Heart J.2005;26:2166–2172.
- ,.Cardiac biomarkers for risk stratification of patients with acute pulmonary embolism.Circulation.2003;108:2191–2194.
- ,,, et al.Prognostic role of echocardiography among patients with acute pulmonary embolism and a systolic arterial pressure of 90 mm Hg or higher.Arch Intern Med.2005;165:1777–1781.
- ,,, et al.Derivation and validation of a prognostic model for pulmonary embolism.Am J Respir Crit Care Med.2005;172:1041–1046.
- ,,.The validation and reproducibility of the pulmonary embolism severity index.J Thromb Haemost.2010;8:1509–1514.
- ,,, et al.Prognostic factors for pulmonary embolism: the prep study, a prospective multicenter cohort study.Am J Respir Crit Care Med.2010;181:168–173.
- ,.A method of comparing the areas under receiver operating characteristic curves derived from the same cases.Radiology.1983;148:839–843.
- ,,, et al.Prospective validation of the pulmonary embolism severity index. A clinical prognostic model for pulmonary embolism.Thromb Haemost.2008;100:943–948.
- ,,, et al.Expanding eligibility for outpatient treatment of deep venous thrombosis and pulmonary embolism with low‐molecular‐weight heparin: a comparison of patient self‐injection with homecare injection.Arch Intern Med.1998;158:1809–1812.
- ,,, et al.Outpatient treatment of pulmonary embolism with dalteparin.Thromb Haemost.2000;83:209–211.
- ,,, et al.Outpatient treatment of pulmonary embolism is feasible and safe in a substantial proportion of patients.J Thromb Haemost.2003;1:186–187.
- ,,, et al.A randomized trial comparing 2 low‐molecular‐weight heparins for the outpatient treatment of deep vein thrombosis and pulmonary embolism.Arch Intern Med.2005;165:733–738.
- ,,, et al.Early discharge of patients with pulmonary embolism: a two‐phase observational study.Eur Respir J.2007;30:708–714.
- ,,, et al.Home treatment in pulmonary embolism.Thromb Res.2010;126:e1–e5.
- ,,.Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER).Lancet.1999;353:1386–1389.
- ,,, et al.Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta‐analysis of the randomized controlled trials.Circulation.2004;110:744–749.
- ,,, et al.Predictors of survival after deep vein thrombosis and pulmonary embolism: a population‐based, cohort study.Arch Intern Med.1999;159:445–453.
Copyright © 2011 Society of Hospital Medicine
Predicting Antibiotic Resistance in HCAP
Healthcare associated pneumonia (HCAP) is defined as pneumonia that is present upon admission, and occurs in patients that have recently been hospitalized, reside in a nursing home, or have had other recent healthcare exposures. Practice guidelines developed by the American Thoracic Society (ATS) and the Infectious Diseases Society of America (IDSA), recommend strategies for the diagnosis and treatment of patients with HCAP.1 A premise of the guidelines is that recent healthcare exposure places patients at risk for infection due to multi‐drug resistant (MDR) pathogens such as methicillin‐resistant Staphylococcus aureus (MRSA) or Pseudomonas aeruginosa. In addition to criteria utilized to define HCAP, the guidelines state that recent immunosuppression and antibiotic exposure are risk factors for pneumonia due to MDR pathogens. In contrast to the treatment of community‐acquired pneumonia (CAP), the guidelines recommend empirical administration of antibiotics with activity against MRSA and Pseudomonas aeruginosa for all patients with HCAP.
We recently reported that antimicrobial resistance to CAP antibiotics (CAP‐resistance) was identified in one‐third of culture‐positive patients with HCAP.2 Data regarding the predictive ability of the guideline‐defined criteria specific to HCAP are limited.3 Evaluation and potential refinement of the criteria to identify patients at risk for MDR pathogens can aid in making antibiotic‐related treatment decisions.
The purposes of this study are to: 1) develop and validate a model to predict CAP‐resistance among patients with HCAP, and to compare the model's predictive performance to a model that includes traditional guideline‐defined risk factors; and 2) develop models to predict recovery of pathogen‐specific etiology (MRSA and Pseudomonas aeruginosa), and to compare the predictive performance of the pathogen‐specific and CAP‐resistance models.
METHODS
Patients with HCAP who were admitted to 6 Veterans Affairs Medical Centers (VAMC) in the northwestern United States between January 1, 2003 and December 31, 2008 were included in the retrospective cohort study. The cohort was identified utilizing medical records data extracted from the Veterans Integrated Service Network (VISN20) Data Warehouse. The Data Warehouse is a centralized open architecture relational database that houses medical and administrative records data for VISN20 patients. This research complies with all federal guidelines and VAMC policies relative to human subjects and clinical research.
Subjects were identified by the following pneumonia‐related discharge International Classification of Diseases (ICD‐9 CM) codes: 1) a primary diagnosis of 480‐483; 485‐487.0 (pneumonia); or 2) a primary diagnosis of 507.0 (pneumonitis), 518.8 (respiratory failure), or 0.38 (septicemia), and a secondary diagnosis of 480‐483; 485‐487.0.4 Eligibility required that patients received antibiotic therapy for pneumonia within 24 hours of admission, continue inpatient treatment for >24 hours, and meet any of the following guideline‐defined criteria: 1) hospitalization during the preceding 90 days; 2) admission from a nursing home; 3) outpatient or home wound care, outpatient or home infusion therapy, or chronic hemodialysis.1 In addition, patients not meeting guideline‐defined criteria, who had frequent healthcare system exposure, defined as 12 Emergency Department, Medicine, or Surgery clinic visits within 90 days of admission, were also included. Patients were excluded if they were directly transferred from another hospital, or had pneumonia‐related ICD‐9 codes but received inpatient care for pneumonia in a non‐VA hospital.
Study data included medical records for the year prior to admission for HCAP through 30 days afterwards. Data included: demographics; domicile preceding admission; healthcare utilization including diagnosis and procedure codes; inpatient medications administered, and outpatient prescription fills; vital signs; and laboratory test results, including cultures and susceptibilities.
Guideline‐defined criteria for predicting CAP‐resistance were similar to those used to identify the study cohort. Nursing home admission included patients who were directly admitted from a nursing home, skilled nursing facility, or domiciliary. Prior hospitalization 2 days within 90 days was calculated by summing the length of stay for all admissions during the preceding 90 days. Outpatient intravenous therapy, chronic hemodialysis, and wound care therapy was determined from medication administration records and relevant Current Procedural Terminology (CPT) or ICD‐9 procedure codes for care administered within 30 days. Antibiotic exposure was defined as administration of 1 dose of antibiotic during inpatient care, or fill of an outpatient prescription for 1 antibiotic dose within 90 days preceding admission. Immunosuppression was defined as: human immunodeficiency virus (HIV) diagnosis; white blood cell (WBC) count of 2500 cells/mm3 within 30 days of admission; corticosteroid ingestion during prior admission, or outpatient prescription fills for a corticosteroid with quantity sufficient to last 14 days preceding admission; or inpatient ingestion of, or outpatient prescription fills for, transplant or rheumatologic‐related immunosuppressants within 90 days preceding admission.
Additional variables assessed to predict CAP‐resistance were obtained as follows. First, modifications of guideline‐defined criteria were constructed. These included: direct nursing home admission, or recent nursing home stay preceding admission; total days of hospitalization within 90 days preceding admission; specific antibiotic exposures, including dates since last exposure preceding admission; and individual components of the immunosuppression criterion. Other cohort‐developed variables included: demographics; substance use history; chronic comorbidity determined by individual and composite measures of Charlson score; pulmonary disease history (eg, bronchiectasis); type and frequency of outpatient visits; consecutive (2) prescription fills for chronic medications of interest; clinical and surveillance culture results preceding admission; admitting ward; vital signs; and relevant hematology and chemistry labs.5
Sputum, blood, and bronchoscopy‐collected cultures obtained within 48 hours after admission were assessed to determine specimen acceptability. Poor sputum specimens were defined by Gram stain quantitative results indicating >10 epithelial cells (EPI) per low power field (LPF), or in the absence of quantitative results, semi‐quantitative results indicating 2‐4+EPI. Single positive blood cultures with results indicating likely contaminants were considered poor specimens. All bronchoscopy‐obtained specimens were considered acceptable. All cultures classified as poor specimens were excluded, and microbiology results were evaluated for the remaining specimens.2, 6 Organisms thought to represent colonization or contamination were excluded: coagulase‐negative (CN) Staphylococcus, Enterococcus sp, Bacillus sp, Proprionibacterium sp, and Candida sp. Recovery of a potential pneumonia pathogen from 1 acceptable culture constituted a culture‐positive admission.
CAP‐resistance was determined for each isolate. CAP‐resistance was defined as non‐susceptibility to non‐pseudomonal third generation cephalosporins (ceftriaxone or cefotaxime) or non‐pseudomonal 8‐methoxy fluoroquinolones (moxifloxacin, gatifloxacin), the VA preferred agents for treatment of CAP.7 There were differences between facilities in susceptibility reporting criteria; therefore, the following approach was used to determine CAP‐resistance. First, MRSA and Pseudomonas aeruginosa isolates were classified as CAP‐resistant. Second, susceptibility results were directly utilized to determine CAP‐resistance if both antibiotic results were available. Third, if only a surrogate antibiotic from a class was reported, a representative antibiotic consistent with Clinical Laboratory Standards Institute reporting criteria was utilized.8 Finally, expert rules determined CAP‐resistance for select potential pneumonia pathogens (eg, Haemophilus sp) if antibiotic susceptibility results for both cephalosporin and fluoroquinolone classes were not reported.815 Presence of 1 CAP‐resistant isolate resulted in a CAP‐resistant classification for an admission. MRSA and Pseudomonas aeruginosa endpoints were defined in a similar manner. Only the first admission for each patient was utilized in the analysis.
The probability of CAP‐resistance was predicted from guideline‐defined criteria (guideline‐defined model) with logistic regression. Next, non‐guideline variables were classified as high, medium, or low interest for association with CAP‐resistance. Variables were assessed for collinearity. A model of CAP‐resistance was developed from variables of high interest. Guideline‐defined criteria were omitted to allow consideration of more specific measures (eg, specific antibiotic exposures as opposed to receipt of antibiotics within the preceding 90 days) during this stage. Next, guideline‐defined criteria, and subsequently variables of lesser interest, were added in an attempt to improve the model. Annual trends and plausible interactions were considered. Model selection was by Akaike's Information Criterion (AIC).16 To promote model reliability, the final model was required to lack evidence of over‐fitting in bootstrapped internal validation.17 The guideline‐defined and cohort‐developed models were compared by difference in area under receiver operating characteristic (aROC) curves. The model development process was repeated for MRSA and Pseudomonas aeruginosa endpoints. Finally, to determine if the CAP‐resistance model sufficiently predicted pathogen‐specific MDR, the CAP‐resistance model was re‐estimated for MRSA and Pseudomonas aeruginosa endpoints. Statistical analysis was performed with R version 2.10.0 (The R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
The cohort was comprised of 1300 patients with HCAP. Of these, 375 (28.8% [26.4‐31.4]) met culture‐positive criteria for potential pneumonia pathogens. CAP‐resistant organisms were identified in 118 (31.5% [26.8‐36.4]) patients within 48 hours of admission. CAP‐resistant organisms included: MRSA (49.2% [40.4‐58.1]), Pseudomonas aeruginosa (29.5% [21.9‐38.1]), Enterobacteriaceae (11.4% [6.5‐18.0]), Gram‐negative non‐enterics (8.3% [4.2‐14.4]), Streptococcus pneumoniae (1.5% [0.2‐5.4]), and opportunistic organisms (eg, Mycobacterium spp) (8.3% [4.2‐14.4]). Differences in select characteristics and exposures between culture‐positive and culture‐negative admissions, as well as CAP‐resistant and CAP‐sensitive admissions, were evident (Table 1).
| Characteristic | Culture‐Negative Admissions (n = 925) | Culture‐Positive Admissions (n = 375) | P Value | CAP‐Sensitive Admissions (n = 257) | CAP‐Resistant Admissions (n = 118) | P Value |
|---|---|---|---|---|---|---|
| ||||||
| Demographics | ||||||
| Age (mean/SD) | 71.9 (12.1) | 71.4 (12.4) | 0.44 | 70.4 (12.4) | 72.9 (12.3) | 0.07 |
| Gender (% male) | 97.1 | 98.8 | 0.07 | 98.4 | 99.2 | 1.00 |
| Primary inclusion diagnosis (%) | ||||||
| Pneumonia | 93.1 | 85.9 | <0.01 | 87.2 | 83.1 | 0.87 |
| Aspiration pneumonitis with pneumonia pneumonia witpneumonia | 1.5 | 4.3 | 0.02 | 4.6 | 3.3 | 0.48 |
| Septicemia with pneumonia | 2.6 | 6.2 | <0.01 | 5.1 | 8.5 | 0.25 |
| Respiratory failure with pneumonia | 2.8 | 3.5 | 0.50 | 3.1 | 5.1 | 0.38 |
| HCAP inclusion criteria (%) | ||||||
| Nursing home residence | 31.2 | 35.9 | 0.08 | 30.4 | 46.6 | <0.01 |
| Hospitalization of >2 days in last 90 days | 58.7 | 57.6 | 0.73 | 52.1 | 62.7 | 0.06 |
| Intravenous therapy in last 30 days | 19.5 | 20.7 | 0.61 | 19.5 | 21.2 | 0.68 |
| Outpatient wound care in last 30 days | 2.7 | 2.7 | 1.00 | 3.1 | 1.7 | 0.73 |
| Chronic dialysis in last 30 days | 2.5 | 1.7 | 0.45 | 1.2 | 2.5 | 0.38 |
| Hospitalization duration 0‐2 days in last 90 days | 10.2 | 11.2 | 0.57 | 12.5 | 5.9 | 0.22 |
| >12 ED or clinic visits in last 90 days | 44.1 | 44.6 | 0.86 | 44.0 | 41.5 | 0.74 |
| Other guideline‐defined MDR criteria (%) | ||||||
| Antibiotics in last 90 days | 63.8 | 61.6 | 0.47 | 57.2 | 66.1 | 0.11 |
| Recent immunosuppression | 19.3 | 23.9 | 0.53 | 24.1 | 22.0 | 0.70 |
| Severity of illness (%) | ||||||
| Admitted to the ICU | 21.8 | 41.6 | <0.01 | 26.3 | 38.6* | <0.01 |
| Mechanical ventilation | 5.6 | 12.7 | <0.01 | 12.1 | 12.7 | 0.87 |
| Comorbidity (%) | ||||||
| Charlson comorbidity score (mean/SD) | 4.3 (3.0) | 4.3 (3.0) | 0.85 | 4.1 (3.1) | 4.5 (2.8) | 0.20 |
| Diabetes | 33.8 | 29.2 | 0.10 | 27.2 | 39.0 | 0.07 |
| Prior antibiotic use (%) | ||||||
| Any cephalosporin | 42.0 | 39.9 | 0.48 | 32.3 | 51.7 | <0.01 |
| Third generation cephalosporin | 24.5 | 23.7 | 0.78 | 18.3 | 30.5 | 0.01 |
| Anti‐pseudomonal fluoroquinolone | 28.5 | 28.4 | 1.0 | 23.3 | 37.3 | 0.02 |
| 8‐Methoxy fluoroquinolone | 20.1 | 23.9 | 0.10 | 24.1 | 24.5 | 1.00 |
| Prior corticosteroid use (%) | ||||||
| Systemic steroids (>10 mg/day prednisone) | 11.1 | 13.2 | 0.28 | 11.3 | 16.1 | 0.24 |
| Inhaled steroids | 7.5 | 10.0 | 0.11 | 8.9 | 10.2 | 0.71 |
| Prior MDR cultured (%) | ||||||
| MRSA within <90 days | 4.2 | 7.7 | <0.01 | 2.7 | 15.3 | <0.01 |
| MRSA >90 days but <365 days | 5.6 | 6.5 | 0.54 | 3.9 | 10.2 | 0.03 |
| P. aeruginosa within 365 days | 5.7 | 11.5 | <0.01 | 5.8 | 19.5 | <0.01 |
Of the guideline‐defined criteria, direct admission from a nursing home, prior hospitalization, and recent antibiotic exposure were associated with CAP‐resistance (Table 2). The cohort‐derived CAP‐resistance model included 6 variables. Prior MRSA colonization or infection within 90 days preceding admission was strongly predictive of CAP‐resistance. A composite variable consisting of direct admission from a nursing home or admission from the community after recent discharge from a nursing home was more predictive than direct admission from a nursing home alone. Exposure to cephalosporin antibiotics within the prior year was also predictive of CAP‐resistance. Subcategorizing cephalosporins by class or by most recent exposure in 90‐day increments did not improve the model. The remaining predictors in the model were guideline‐defined infusion therapy criterion, diabetes, and intensive care unit (ICU) admission.
| Guidelinedefined model of CAPResistant HCAP | AIC 461.1 | CohortDeveloped Model of CAPresistant HCAP | AIC 431.1 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P Value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Nursing home residence at time of admission | 2.6 | 1.64.4 | <0.001 | Nursing home residence or discharge 180 days prior to admission | 2.3 | 1.43.8 | 0.002 |
| Antibiotic exposure 90 days prior to admission | 1.7 | 1.02.8 | 0.054 | Positive MRSA status: 90 days prior to admission | 6.4 | 2.617.8 | <0.001 |
| Hospitalization 2 days, 90 days prior to admission | 1.6 | 1.02.6 | 0.066 | >90 days but 365 days prior to admission | 2.3 | 0.95.9 | 0.074 |
| Infusion therapy 30 days prior to admission | 1.5 | 0.82.8 | 0.173 | Cephalosporin exposure 365 days prior to admission | 1.8 | 1.12.9 | 0.019 |
| Wound care therapy 30 days prior to admission | 0.5 | 0.12.1 | 0.370 | Infusion therapy 30 days prior to admission | 1.9 | 1.03.5 | 0.044 |
| Hemodialysis therapy 30 days prior to admission | 1.8 | 0.311.2 | 0.497 | Diabetes | 1.7 | 1.02.8 | 0.044 |
| Recent immunosuppression | 0.9 | 0.51.6 | 0.670 | Direct ICU admission upon hospitalization | 1.6 | 1.02.6 | 0.053 |
Of the guideline‐defined criteria, direct admission from a nursing home was most predictive of MRSA HCAP (n = 57), followed by prior hospitalization and recent antibiotic exposure (Table 3). The cohort‐developed model of MRSA HCAP included predictors common to the CAP‐resistance model: direct admission from a nursing home or patients who were recently discharged from a nursing home, history of prior MRSA, and diabetes. Positive MRSA status within 90 days preceding admission exhibited the strongest prediction of MRSA HCAP. Exposure to anti‐pseudomonal fluoroquinolones (ciprofloxacin and levofloxacin) within the prior year was also predictive of MRSA HCAP, however, exposure to 8‐methoxy fluoroquinolone was not (crude odds ratio (OR) = 0.7 [0.3‐1.4]; final model adjusted OR = 0.6 [0.2‐1.2]). Exposure to third generation cephalosporins within the previous year was more predictive than other cephalosporin exposures, and more predictive than exposure times categorized in 90‐day increments.
| Guideline‐Defined Model of MRSA HCAP | AIC 316.3 | Cohort‐Developed Model of MRSA HCAP | AIC 279.2 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P Value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Nursing home residence at time of admission | 2.6 | 1.44.8 | 0.003 | Nursing home residence or discharge 180 days prior to admission | 2.8 | 1.55.3 | 0.002 |
| Hospitalization 2 days, 90 days prior to admission | 1.8 | 1.03.5 | 0.075 | Positive MRSA status: 90 days prior to admission | 7.7 | 3.119.6 | <0.001 |
| Antibiotic exposure 90 days prior to admission | 1.6 | 0.93.3 | 0.143 | >90 days but 365 days prior to admission | 1.4 | 0.54.1 | 0.507 |
| Recent immunosuppression | 0.6 | 0.31.3 | 0.244 | Anti‐pseudomonal fluoroquinolone exposure 365 days prior to admission | 2.4 | 1.24.6 | 0.009 |
| Wound care therapy 30 days prior to admission | 0.5 | 0.03.3 | 0.582 | Diabetes | 2.2 | 1.24.3 | 0.012 |
| Infusion therapy 30 days prior to admission | 0.9 | 0.42.0 | 0.793 | Chronic inhaled corticosteroids | 2.8 | 1.17.1 | 0.031 |
| Chronic hemodialysis 30 days prior to admission* | Third generation cephalosporin exposure 365 days prior to admission | 2.1 | 1.04.1 | 0.040 | |||
Of the guideline‐defined criteria, only prior hospitalization within 90 days and admission from a nursing home were predictive of Pseudomonas aeruginosa HCAP (n = 36) (Table 4). In the cohort‐developed model of Pseudomonas aeruginosa HCAP, Pseudomonas aeruginosa was predicted by prior cephalosporin exposure within the preceding year, prior culture of Pseudomonas aeruginosa from any anatomical source within the preceding year, and chronic steroid use of 10 mg/day prednisone equivalents. Again, the model was not improved by subcategorizing cephalosporin by class or by most recent exposure time. Finally, a negative annual trend in Pseudomonas aeruginosa HCAP was evident.
| Guideline‐defined model of Pseudomonas aeruginosa HCAP | AIC 234.8 | Cohort‐developed model of Pseudomonas aeruginosa HCAP | AIC 211.1 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Hospitalization 2 days, 90 days prior to admission | 2.5 | 1.16.0 | 0.034 | Cephalosporin exposure 365 days prior to admission | 3.8 | 1.88.8 | <0.001 |
| Nursing home residence at time of admission | 2.1 | 1.04.6 | 0.059 | Positive Pseudomonas aeruginosa culture 365 days prior to admission | 3.3 | 1.47.8 | 0.006 |
| Chronic hemodialysis 30 days prior to admission | 5.0 | 0.631.2 | 0.093 | Chronic steroid dose of 10 mg/day prednisone equivalents prior to admission | 3.0 | 1.36.9 | 0.010 |
| Antibiotic exposure 90 days prior to admission | 1.9 | 0.84.7 | 0.150 | Year of study | 0.8 | 0.71.0 | 0.069 |
| Infusion therapy 30 days prior to admission | 1.8 | 0.74.2 | 0.172 | ||||
| Recent immunosuppression | 1.1 | 0.52.5 | 0.764 | ||||
| Wound care therapy 30 days prior to admission* | |||||||
The cohort‐developed model of CAP‐resistance was re‐estimated for MRSA and Pseudomonas aeruginosa endpoints. Only positive MRSA status within 90 days preceding admission was associated with both endpoints (OR = 8.7 [3.5‐22.1] for MRSA; OR = 4.3 [1.4‐12.2] for Pseudomonas aeruginosa). Direct or recent nursing home residence (OR = 2.4 [1.3‐4.6]) and diabetes (OR = 2.4 [1.3‐4.5]) were highly predictive of MRSA, but not Pseudomonas aeruginosa (OR = 1.8 [0.8‐3.9] for nursing home residence; OR = 1.3 [0.6‐2.7] for diabetes), respectively. Cephalosporin exposure preceding admission was highly predictive of Pseudomonas aeruginosa (OR = 4.0 [1.9‐9.3]), but not with MRSA (OR = 1.1 [0.6‐2.1]). In these models, all estimated odds ratios were >1.0, consistent with the cohort‐developed model of CAP‐resistance.
For each endpoint, the cohort‐developed model was more predictive than the guideline‐defined model (Table 5) (to view ROC curves see Supporting Figures 1 to 3 in the online version of the article.). The cohort‐developed model of CAP‐resistance re‐estimated for pathogen‐specific endpoints resulted in similar predictive performance. To assess performance of the cohort developed models by facility, aROC was calculated for each of the 3 larger sites separately and for the 3 smaller facilities combined due to limited counts. Site specific aROC ranged from 0.652 to 0.762 for CAP‐resistance, 0.725 to 0.815 for MRSA, and 0.719 to 0.801 for Pseudomonas aeruginosa. The cohort‐developed model of CAP‐resistance re‐estimated for pathogen‐specific endpoints resulted in similar predictive performance.
| Model | Outcome Variable | Predictive Variables | aROC | (95% CI) | Model Comparison | aROC Difference | (95% CI) | P Value |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| 1 | CAP‐resistance | Guideline‐defined | 0.630 | (0.570, 0.691) | 2‐1 | 0.079 | (0.018, 0.139) | 0.011 |
| 2 | CAP‐resistance | Cohort‐developed | 0.709 | (0.650, 0.768) | ||||
| 3 | MRSA | Guideline‐defined | 0.638 | (0.560, 0.712) | 4‐3 | 0.135 | (0.057, 0.213) | <0.001 |
| 4 | MRSA | Cohort‐developed | 0.773 | (0.703, 0.844) | ||||
| 5 | Pseudomonas aeruginosa | Guideline‐defined | 0.680 | (0.593, 0.768) | 6‐5 | 0.090 | (0.193, 0.193) | 0.090 |
| 6 | Pseudomonas aeruginosa | Cohort‐developed | 0.770 | (0.683, 0.857) | ||||
| 7 | MRSA | Cohort‐developed from CAP‐resistance model | 0.755 | (0.682, 0.828) | 7‐4 | 0.018 | (0.067, 0.031) | 0.467 |
| 8 | Pseudomonas aeruginosa | Cohort‐developed from CAP‐resistance model | 0.755 | (0.665, 0.845) | 8‐6 | 0.015 | (0.079, 0.049) | 0.650 |
A nomogram for the cohort‐developed model of CAP‐resistance can provide the predicted probability of culturing a CAP‐resistant organism for an individual patient (Table 6). Point scores assigned to levels of variables, are summed to obtain a total score, and the total score corresponds to a predicted probability of CAP‐resistance. The prevalence of CAP‐resistance (%) from highest to lowest quartile of predicted probability was 92.9, 58.8, 32.9, and 18.5, respectively.
| A. Scoring | |
|---|---|
| Variable | Score |
| B. Predicted Probability of CAP‐Resistance* | |
| Total Score | % Chance of CAP‐Resistance |
| |
| Positive MRSA status prior to admission | |
| 90 days | +100 |
| >90 days but 365 days | +45 |
| Nursing home residence or discharge 180 days prior to admission | +45 |
| Infusion therapy 30 days prior to admission | +35 |
| Cephalosporin exposure 365 days prior to admission | +30 |
| Diabetes | +30 |
| Direct ICU admission upon hospitalization | +25 |
| <35 | <20 |
| 3565 | 2030 |
| 6590 | 3040 |
| 90110 | 4050 |
| 110130 | 5060 |
| 130155 | 6070 |
| 155185 | 7080 |
| 185230 | 8090 |
| >230 | >90 |
DISCUSSION
In this study, select ATS/IDSA guideline‐defined criteria predicted identification of CAP‐resistant organisms in patients with HCAP. Admission from a nursing home was most predictive of CAP‐resistant organisms, whereas recent hospitalization and antibiotic exposure were predictive to a lesser extent. There was weak evidence of associations between recent infusion and chronic hemodialysis criteria with MDR endpoints. Recent wound care and a composite definition of immunosuppression were not predictive of these endpoints.
The cohort‐developed model resulted in improved prediction of CAP‐resistance endpoints. Culture history, particularly history of MRSA within 90 days preceding admission, was a strong predictor of MDR endpoints. The MRSA history variable definition included cultures from all anatomical sources and nares polymerase chain reaction surveillance results, the latter increasing in 2007‐2008 due to the implementation of the VA MRSA initiative.18 This finding suggests that prior culture results should be considered when selecting empirical antimicrobial therapy, and the rapid proliferation of electronic medical records increases potential to utilize this information routinely. While the guideline‐defined nursing home admission criterion was a strong predictor of CAP‐resistance, admission from the community after recent discharge from a nursing home, in addition to direct admission from a nursing home, was also important.
Similarities in variables included in the pathogen‐specific and CAP‐resistance models reflect the importance of MRSA in defining the CAP‐resistance endpoint. Both CAP‐resistance and MRSA models included prior MRSA status, diabetes, and ICU admission, whereas cephalosporin exposure was common to the Pseudomonas aeruginosa and CAP‐resistance models. Annual trends in CAP‐resistance and MRSA recovery were not identified. The negative annual trend in Pseudomonas aeruginosa HCAP is unexplained and beyond the scope of this study. The percentage of culture‐positive admissions with Pseudomonas aeruginosa HCAP averaged 12% in 2003‐2006, but dropped to <5% in 2007‐2008. A potential explanation is that identification and isolation of patients with MRSA, as a result of the VA‐wide MRSA initiative, may have impacted Pseudomonas aeruginosa colonization by isolating patients co‐colonized with these pathogens during prior healthcare exposures. This is consistent with the observation that when the cohort‐derived CAP‐resistance model was refit with the Pseudomonas aeruginosa endpoint, recent MRSA colonization was strongly predictive of Pseudomonas aeruginosa. Despite differences between variables in pathogen‐specific and CAP‐resistant models, the CAP‐resistance model provided a similar degree of MRSA and Pseudomonas aeruginosa prediction. Finally, as a study purpose included developing best predictive models for each endpoint, and not merely identifying associations, there were other plausible models not reported.
Study strengths included use of the VISN20 Data Warehouse, which provided an integrated outpatient and inpatient medical record. This facilitated analysis of prior healthcare exposures and inpatient study endpoints. In addition, poor blood and sputum specimens and unlikely pneumonia pathogens were not included in establishing MDR endpoints. The variable set explored in regression modeling was extensive and detailed, and analysis included time and intensity‐based components of the variables. Importantly, a standardized approach to regression modeling was specified in advance, which included identification of variables with high potential for association with MDR endpoints, model selection by AIC, re‐evaluation of guideline‐defined criteria and variables of lower interest, and bootstrapped internal model validation.19
Study limitations included the use of ICD‐9 codes to establish a pneumonia diagnosis, which may lack sensitivity and specificity. However, an enhanced ICD‐9based algorithm superior to other claims‐based definitions of pneumonia was utilized.4, 20 Veterans may have received care at non‐VA facilities impacting identification of all healthcare system exposures preceding admission. Data for microbial endpoints were obtained from sterile and non‐sterile site cultures, and it was not possible to determine if the cultured organisms were truly pathogenic. While pathogen‐specific endpoints were not affected, the use of expert rules in select cases to establish CAP‐resistance may have impacted precision for this endpoint. It is also possible that refitting the cohort‐developed CAP‐resistance model for pathogen‐specific endpoints resulted in optimistic aROC due to model over‐fitting. Finally, the cohort was comprised of elderly males, and caution is warranted in extrapolating the results to other populations.
The predictive ability of the guideline‐defined criteria to identify patients with MDR pathogens has been studied. A prospective observational cohort study of 625 consecutive ICU admissions determined that the guideline‐defined criteriaprior antimicrobial treatment, nursing home residence, and prior hospitalizationwere associated with recovery of MDR colonization.21 Shorr et al., investigating a retrospective cohort of 619 patients with HCAP, reported that recent hospitalization, nursing home residence, hemodialysis, and ICU admission were associated with infections caused by CAP‐resistant organisms.22 This study did not report antimicrobial exposures. Our study complements these studies by evaluating existing HCAP guideline criteria, and identifying specific antibiotic exposure, prior culture data, comorbid illness, and immunosuppressive medications that are predictive of MDR infection.
Studies comparing the bacterial etiology of patients with pneumonia in nursing homes relative to CAP, have demonstrated mixed results in recovery of Gram‐negative MDR pathogens, but generally increased MRSA pneumonia.3 Our study suggests that a nursing home stay in the last 6 months is associated with an increased risk for MRSA, but not Pseudomonas aeruginosa, although this was limited by small sample size. Recent infusion therapy has not been previously reported to be associated with MDR pathogens in an HCAP population. In our study, this criterion was predictive of CAP‐resistance in the cohort‐developed model, but not in conjunction with other variables in the guideline‐defined model. Predictors of pathogen‐specific HCAP are limited to an aforementioned single prior study, which identified recent hospitalization, nursing home residence, and ICU admission as risk factors for MRSA HCAP.22
Many studies have investigated risks for infection with MRSA and Pseudomonas aeruginosa outside of the context of HCAP. Predictor variables in cohort‐developed pathogen‐specific models in our study are known risk factors for colonization or infection with these pathogens. For example, antecedent MRSA colonization has been noted as a strong risk factor for MRSA infection, particularly pneumonia.23, 24 Further, patients with diabetes and inhaled corticosteroid exposure are immunosuppressed and at increased risk for colonization with MRSA.25, 26 Likewise, bronchiolar colonization and corticosteroid exposures are known risk factors for pneumonia due to Pseudomonas aeruginosa.27
Many studies have identified prior antibiotic use as a risk factor for infections caused by MRSA and Pseudomonas aeruginosa. However, this criterion is excessively broad and specific antimicrobial exposures carry different magnitudes of risk. Third generation cephalosporins and anti‐pseudomonal fluoroquinolones are commonly reported antibiotics associated with risk for MRSA infection, whereas 8‐methoxy fluoroquinolones appear not to possess the same effect.2831 Likewise, cephalosporins have been reported as risk factors for MDR Pseudomonas aeruginosa infections.32
Several areas of research involving HCAP MDR risk should be investigated. First, the predictive models developed in our and other studies should be evaluated in larger, more diverse populations to establish generalizability. Second, empirical broad‐spectrum antibiotic therapy in all patients with HCAP results in overtreatment of many patients. To date, no reported models provided optimal performance for selecting empirical therapy for unstable ICU patients with HCAP, and many patients do not receive de‐escalation therapy. Thus, models to identify patients with low probability of MDR pathogens upon admission and to aid in de‐escalation are warranted. Finally, the negative trend in Pseudomonas aeruginosa HCAP requires confirmation and further study.
In conclusion, of the ATS/IDSA guideline‐defined criteria for MDR, nursing home admission, recent hospitalization, and antibiotic exposure were predictive of the recovery of CAP‐resistant organisms. Alternative models primarily based on prior culture data, specific antibiotic exposures, and immunosuppression‐related variables improved predictive performance of HCAP associated with MDR.
- American Thoracic Society; Infectious Diseases Society of America.Guidelines for the management of adults with hospital‐acquired, ventilator‐associated, and healthcare‐associated pneumonia.Am J Respir Crit Care Med.2005;171(4):388–416.
- ,,,.The etiology of health care associated pneumonia (HCAP) [abstract K‐282]. Presented at the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy; September2009; San Francisco, CA.
- ,.What are the important risk factors for healthcare‐associated pneumonia?Semin Respir Crit Care Med.2009;30(1):26–35.
- ,,,.Accuracy of administrative data for identifying patients with pneumonia.Am J Med Qual.2005;20(6):319–328.
- ,,.Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases.J Clin Epidemiol.1992;45:613–619.
- ,,,.How often is a microbial etiology identified in health care associated pneumonia (HCAP)? [abstract K‐289]. Presented at the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy; September2009; San Francisco, CA.
- Fluoroquinolone use criteria. Washington D.C. Guidelines Developed by the Pharmacy Benefits Management Strategic Health Care Group and Medical Advisory Panel, Veterans Health Administration, Department of Veterans Affairs. Last update, November2006. http://www.pbm.va.gov/Clinical%20Guidance/Criteria%20For%20Use/Fluoroquinolone,%20Criteria%20for%20Use.pdf. Last accessed August 20th, 2011.
- Performance Standards for Antimicrobial Susceptibility Testing; 18th Informational Supplement. M100‐S18.Wayne, PA:Clinical Laboratory Standards Institute;2009.
- ,,.Antimicrobial activity of DC‐159a, a new fluoroquinolone, against 1,149 recently collected clinical isolates.Antimicrob Agents Chemother.2008;52(10):3763–3775.
- ,,, et al.Mortality from invasive pneumococcal pneumonia in the era of antibiotic resistance, 1995–1997.Am J Public Health.2000;90(2):223–229.
- ,.Comparative activity of moxifloxacin against Gram‐positive clinical isolates.J Antimicrob Chemother.2000;45(1):31–39.
- ,,.Antimicrobial susceptibility of extended‐spectrum‐beta‐lactamase producers and multi‐drug resistant Acinetobacter baumannii throughout the United States and comparative in vitro activity of tigecycline, a new glycylcycline antimicrobial.Diagn Microbiol Infect Dis.2007;57(4):423–428.
- ,,.Antimicrobial susceptibility profile of contemporary clinical strains of Stenotropomonas maltophila isolates: can moxifloxacin activity be predicted by levofloxacin MIC results?J Chemother.2008;20(1):38–42.
- ,,, et al.Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults.Clin Infect Dis.2007;44(suppl 2):S27–S72.
- Gilbert DN, Moellering RC, Eliopoulos GM, Sande, MA, eds.The Sanford Guide to Antimicrobial Therapy.38th ed.Speryville, VA:Antimicrobial Therapy;2008.
- .A new look at the statistical model identification.IEEE Trans Automat Contr.1974;19(6):716–723.
- .Estimating the error rate of a prediction rule: improvement on cross‐validation.J Am Stat Assoc.1987;78:316–331.
- ,,, et. Al.Beyond beta: lessons learned from implementation of the Department of Veterans Affairs Methicillin‐Resistant Staphylococcus aureus Prevention Initiative.Infect Control Hosp Epidemiol.2010;31(7):763–765.
- ,,.An appraisal of multivariable logistic models in the pulmonary and critical care literature.Chest.2003;123(3):923–928.
- ,,,,,.Improved clinical outcomes with utilization of a community‐acquired pneumonia guideline.Chest.2006;130(3):794–799.
- ,,,,,.Accuracy of American Thoracic Society/Infectious Diseases Society of America criteria in predicting infection or colonization with multidrug‐resistant bacteria at intensive‐care unit admission.Clin Microbiol Infect.2009;16(7):902–908.
- ,,,.Prediction of infections due to antibiotic resistant bacteria by select risk factors for healthcare associated pneumonia.Arch Intern Med.2008;168(20):2205–2210.
- ,,,,.Methicillin‐resistant Staphylococcus aureus (MRSA) nares colonization at hospital admission and its effect on subsequent MRSA infection.Clin Infect Dis.2004;39(6):776–782.
- ,.Risk of infection and death due to methicillin‐resistant Staphylococcus aureus in long‐term carriers.Clin Infect Dis.2008;47(2):176–181.
- ,,,.Epidemiology of methicillin‐resistant Staphylococcus aureus in a university medical center day care facility.Infect Control Hosp Epidemiol.2009;30(10):985–992.
- ,,,,,.Colonization and infection with antibiotic‐resistant bacteria in a long‐term care facility.J Am Geriatr Soc.1994;42(10):1062–1069.
- ,,, et al.American Thoracic Society. Guidelines for the management of adults with community‐acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy, and prevention.Am J Respir Crit Care Med.2001;163(7):1730–1754.
- ,.Modification in prescribing practices for third‐generation cephalosporins and ciprofloxacin is associated with a reduction in methicillin‐resistant Staphylococcus aureus bacteraemia rate.J Hosp Infect.2008;69(4):328–336.
- ,,,.Evaluation of an intervention designed to decrease the rate of nosocomial methicillin‐resistant Staphylococcus aureus infection by encouraging decreased fluoroquinolone use.Infect Control Hosp Epidemiol.2006;27(2):155–169.
- ,.Using interrupted time series analysis to assess associations of fluoroquinolone formulary changes with susceptibility of gram‐negative pathogens and isolation rates of methicillin‐resistant Staphylococcus aureus.Antimicrob Agents Chemother.2006;50(6):2106–2112.
- ,.Dichotomous selection of high‐level oxacillin resistance in Staphylococcus aureus by fluoroquinolones.Int J Antimicrob Agents.2010;36(3):216–221.
- ,,,,.Multidrug‐resistant Pseudomonas aeruginosa: risk factors and clinical impact.Antimicrob Agents Chemother.2006;50(1):43–48.
Healthcare associated pneumonia (HCAP) is defined as pneumonia that is present upon admission, and occurs in patients that have recently been hospitalized, reside in a nursing home, or have had other recent healthcare exposures. Practice guidelines developed by the American Thoracic Society (ATS) and the Infectious Diseases Society of America (IDSA), recommend strategies for the diagnosis and treatment of patients with HCAP.1 A premise of the guidelines is that recent healthcare exposure places patients at risk for infection due to multi‐drug resistant (MDR) pathogens such as methicillin‐resistant Staphylococcus aureus (MRSA) or Pseudomonas aeruginosa. In addition to criteria utilized to define HCAP, the guidelines state that recent immunosuppression and antibiotic exposure are risk factors for pneumonia due to MDR pathogens. In contrast to the treatment of community‐acquired pneumonia (CAP), the guidelines recommend empirical administration of antibiotics with activity against MRSA and Pseudomonas aeruginosa for all patients with HCAP.
We recently reported that antimicrobial resistance to CAP antibiotics (CAP‐resistance) was identified in one‐third of culture‐positive patients with HCAP.2 Data regarding the predictive ability of the guideline‐defined criteria specific to HCAP are limited.3 Evaluation and potential refinement of the criteria to identify patients at risk for MDR pathogens can aid in making antibiotic‐related treatment decisions.
The purposes of this study are to: 1) develop and validate a model to predict CAP‐resistance among patients with HCAP, and to compare the model's predictive performance to a model that includes traditional guideline‐defined risk factors; and 2) develop models to predict recovery of pathogen‐specific etiology (MRSA and Pseudomonas aeruginosa), and to compare the predictive performance of the pathogen‐specific and CAP‐resistance models.
METHODS
Patients with HCAP who were admitted to 6 Veterans Affairs Medical Centers (VAMC) in the northwestern United States between January 1, 2003 and December 31, 2008 were included in the retrospective cohort study. The cohort was identified utilizing medical records data extracted from the Veterans Integrated Service Network (VISN20) Data Warehouse. The Data Warehouse is a centralized open architecture relational database that houses medical and administrative records data for VISN20 patients. This research complies with all federal guidelines and VAMC policies relative to human subjects and clinical research.
Subjects were identified by the following pneumonia‐related discharge International Classification of Diseases (ICD‐9 CM) codes: 1) a primary diagnosis of 480‐483; 485‐487.0 (pneumonia); or 2) a primary diagnosis of 507.0 (pneumonitis), 518.8 (respiratory failure), or 0.38 (septicemia), and a secondary diagnosis of 480‐483; 485‐487.0.4 Eligibility required that patients received antibiotic therapy for pneumonia within 24 hours of admission, continue inpatient treatment for >24 hours, and meet any of the following guideline‐defined criteria: 1) hospitalization during the preceding 90 days; 2) admission from a nursing home; 3) outpatient or home wound care, outpatient or home infusion therapy, or chronic hemodialysis.1 In addition, patients not meeting guideline‐defined criteria, who had frequent healthcare system exposure, defined as 12 Emergency Department, Medicine, or Surgery clinic visits within 90 days of admission, were also included. Patients were excluded if they were directly transferred from another hospital, or had pneumonia‐related ICD‐9 codes but received inpatient care for pneumonia in a non‐VA hospital.
Study data included medical records for the year prior to admission for HCAP through 30 days afterwards. Data included: demographics; domicile preceding admission; healthcare utilization including diagnosis and procedure codes; inpatient medications administered, and outpatient prescription fills; vital signs; and laboratory test results, including cultures and susceptibilities.
Guideline‐defined criteria for predicting CAP‐resistance were similar to those used to identify the study cohort. Nursing home admission included patients who were directly admitted from a nursing home, skilled nursing facility, or domiciliary. Prior hospitalization 2 days within 90 days was calculated by summing the length of stay for all admissions during the preceding 90 days. Outpatient intravenous therapy, chronic hemodialysis, and wound care therapy was determined from medication administration records and relevant Current Procedural Terminology (CPT) or ICD‐9 procedure codes for care administered within 30 days. Antibiotic exposure was defined as administration of 1 dose of antibiotic during inpatient care, or fill of an outpatient prescription for 1 antibiotic dose within 90 days preceding admission. Immunosuppression was defined as: human immunodeficiency virus (HIV) diagnosis; white blood cell (WBC) count of 2500 cells/mm3 within 30 days of admission; corticosteroid ingestion during prior admission, or outpatient prescription fills for a corticosteroid with quantity sufficient to last 14 days preceding admission; or inpatient ingestion of, or outpatient prescription fills for, transplant or rheumatologic‐related immunosuppressants within 90 days preceding admission.
Additional variables assessed to predict CAP‐resistance were obtained as follows. First, modifications of guideline‐defined criteria were constructed. These included: direct nursing home admission, or recent nursing home stay preceding admission; total days of hospitalization within 90 days preceding admission; specific antibiotic exposures, including dates since last exposure preceding admission; and individual components of the immunosuppression criterion. Other cohort‐developed variables included: demographics; substance use history; chronic comorbidity determined by individual and composite measures of Charlson score; pulmonary disease history (eg, bronchiectasis); type and frequency of outpatient visits; consecutive (2) prescription fills for chronic medications of interest; clinical and surveillance culture results preceding admission; admitting ward; vital signs; and relevant hematology and chemistry labs.5
Sputum, blood, and bronchoscopy‐collected cultures obtained within 48 hours after admission were assessed to determine specimen acceptability. Poor sputum specimens were defined by Gram stain quantitative results indicating >10 epithelial cells (EPI) per low power field (LPF), or in the absence of quantitative results, semi‐quantitative results indicating 2‐4+EPI. Single positive blood cultures with results indicating likely contaminants were considered poor specimens. All bronchoscopy‐obtained specimens were considered acceptable. All cultures classified as poor specimens were excluded, and microbiology results were evaluated for the remaining specimens.2, 6 Organisms thought to represent colonization or contamination were excluded: coagulase‐negative (CN) Staphylococcus, Enterococcus sp, Bacillus sp, Proprionibacterium sp, and Candida sp. Recovery of a potential pneumonia pathogen from 1 acceptable culture constituted a culture‐positive admission.
CAP‐resistance was determined for each isolate. CAP‐resistance was defined as non‐susceptibility to non‐pseudomonal third generation cephalosporins (ceftriaxone or cefotaxime) or non‐pseudomonal 8‐methoxy fluoroquinolones (moxifloxacin, gatifloxacin), the VA preferred agents for treatment of CAP.7 There were differences between facilities in susceptibility reporting criteria; therefore, the following approach was used to determine CAP‐resistance. First, MRSA and Pseudomonas aeruginosa isolates were classified as CAP‐resistant. Second, susceptibility results were directly utilized to determine CAP‐resistance if both antibiotic results were available. Third, if only a surrogate antibiotic from a class was reported, a representative antibiotic consistent with Clinical Laboratory Standards Institute reporting criteria was utilized.8 Finally, expert rules determined CAP‐resistance for select potential pneumonia pathogens (eg, Haemophilus sp) if antibiotic susceptibility results for both cephalosporin and fluoroquinolone classes were not reported.815 Presence of 1 CAP‐resistant isolate resulted in a CAP‐resistant classification for an admission. MRSA and Pseudomonas aeruginosa endpoints were defined in a similar manner. Only the first admission for each patient was utilized in the analysis.
The probability of CAP‐resistance was predicted from guideline‐defined criteria (guideline‐defined model) with logistic regression. Next, non‐guideline variables were classified as high, medium, or low interest for association with CAP‐resistance. Variables were assessed for collinearity. A model of CAP‐resistance was developed from variables of high interest. Guideline‐defined criteria were omitted to allow consideration of more specific measures (eg, specific antibiotic exposures as opposed to receipt of antibiotics within the preceding 90 days) during this stage. Next, guideline‐defined criteria, and subsequently variables of lesser interest, were added in an attempt to improve the model. Annual trends and plausible interactions were considered. Model selection was by Akaike's Information Criterion (AIC).16 To promote model reliability, the final model was required to lack evidence of over‐fitting in bootstrapped internal validation.17 The guideline‐defined and cohort‐developed models were compared by difference in area under receiver operating characteristic (aROC) curves. The model development process was repeated for MRSA and Pseudomonas aeruginosa endpoints. Finally, to determine if the CAP‐resistance model sufficiently predicted pathogen‐specific MDR, the CAP‐resistance model was re‐estimated for MRSA and Pseudomonas aeruginosa endpoints. Statistical analysis was performed with R version 2.10.0 (The R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
The cohort was comprised of 1300 patients with HCAP. Of these, 375 (28.8% [26.4‐31.4]) met culture‐positive criteria for potential pneumonia pathogens. CAP‐resistant organisms were identified in 118 (31.5% [26.8‐36.4]) patients within 48 hours of admission. CAP‐resistant organisms included: MRSA (49.2% [40.4‐58.1]), Pseudomonas aeruginosa (29.5% [21.9‐38.1]), Enterobacteriaceae (11.4% [6.5‐18.0]), Gram‐negative non‐enterics (8.3% [4.2‐14.4]), Streptococcus pneumoniae (1.5% [0.2‐5.4]), and opportunistic organisms (eg, Mycobacterium spp) (8.3% [4.2‐14.4]). Differences in select characteristics and exposures between culture‐positive and culture‐negative admissions, as well as CAP‐resistant and CAP‐sensitive admissions, were evident (Table 1).
| Characteristic | Culture‐Negative Admissions (n = 925) | Culture‐Positive Admissions (n = 375) | P Value | CAP‐Sensitive Admissions (n = 257) | CAP‐Resistant Admissions (n = 118) | P Value |
|---|---|---|---|---|---|---|
| ||||||
| Demographics | ||||||
| Age (mean/SD) | 71.9 (12.1) | 71.4 (12.4) | 0.44 | 70.4 (12.4) | 72.9 (12.3) | 0.07 |
| Gender (% male) | 97.1 | 98.8 | 0.07 | 98.4 | 99.2 | 1.00 |
| Primary inclusion diagnosis (%) | ||||||
| Pneumonia | 93.1 | 85.9 | <0.01 | 87.2 | 83.1 | 0.87 |
| Aspiration pneumonitis with pneumonia pneumonia witpneumonia | 1.5 | 4.3 | 0.02 | 4.6 | 3.3 | 0.48 |
| Septicemia with pneumonia | 2.6 | 6.2 | <0.01 | 5.1 | 8.5 | 0.25 |
| Respiratory failure with pneumonia | 2.8 | 3.5 | 0.50 | 3.1 | 5.1 | 0.38 |
| HCAP inclusion criteria (%) | ||||||
| Nursing home residence | 31.2 | 35.9 | 0.08 | 30.4 | 46.6 | <0.01 |
| Hospitalization of >2 days in last 90 days | 58.7 | 57.6 | 0.73 | 52.1 | 62.7 | 0.06 |
| Intravenous therapy in last 30 days | 19.5 | 20.7 | 0.61 | 19.5 | 21.2 | 0.68 |
| Outpatient wound care in last 30 days | 2.7 | 2.7 | 1.00 | 3.1 | 1.7 | 0.73 |
| Chronic dialysis in last 30 days | 2.5 | 1.7 | 0.45 | 1.2 | 2.5 | 0.38 |
| Hospitalization duration 0‐2 days in last 90 days | 10.2 | 11.2 | 0.57 | 12.5 | 5.9 | 0.22 |
| >12 ED or clinic visits in last 90 days | 44.1 | 44.6 | 0.86 | 44.0 | 41.5 | 0.74 |
| Other guideline‐defined MDR criteria (%) | ||||||
| Antibiotics in last 90 days | 63.8 | 61.6 | 0.47 | 57.2 | 66.1 | 0.11 |
| Recent immunosuppression | 19.3 | 23.9 | 0.53 | 24.1 | 22.0 | 0.70 |
| Severity of illness (%) | ||||||
| Admitted to the ICU | 21.8 | 41.6 | <0.01 | 26.3 | 38.6* | <0.01 |
| Mechanical ventilation | 5.6 | 12.7 | <0.01 | 12.1 | 12.7 | 0.87 |
| Comorbidity (%) | ||||||
| Charlson comorbidity score (mean/SD) | 4.3 (3.0) | 4.3 (3.0) | 0.85 | 4.1 (3.1) | 4.5 (2.8) | 0.20 |
| Diabetes | 33.8 | 29.2 | 0.10 | 27.2 | 39.0 | 0.07 |
| Prior antibiotic use (%) | ||||||
| Any cephalosporin | 42.0 | 39.9 | 0.48 | 32.3 | 51.7 | <0.01 |
| Third generation cephalosporin | 24.5 | 23.7 | 0.78 | 18.3 | 30.5 | 0.01 |
| Anti‐pseudomonal fluoroquinolone | 28.5 | 28.4 | 1.0 | 23.3 | 37.3 | 0.02 |
| 8‐Methoxy fluoroquinolone | 20.1 | 23.9 | 0.10 | 24.1 | 24.5 | 1.00 |
| Prior corticosteroid use (%) | ||||||
| Systemic steroids (>10 mg/day prednisone) | 11.1 | 13.2 | 0.28 | 11.3 | 16.1 | 0.24 |
| Inhaled steroids | 7.5 | 10.0 | 0.11 | 8.9 | 10.2 | 0.71 |
| Prior MDR cultured (%) | ||||||
| MRSA within <90 days | 4.2 | 7.7 | <0.01 | 2.7 | 15.3 | <0.01 |
| MRSA >90 days but <365 days | 5.6 | 6.5 | 0.54 | 3.9 | 10.2 | 0.03 |
| P. aeruginosa within 365 days | 5.7 | 11.5 | <0.01 | 5.8 | 19.5 | <0.01 |
Of the guideline‐defined criteria, direct admission from a nursing home, prior hospitalization, and recent antibiotic exposure were associated with CAP‐resistance (Table 2). The cohort‐derived CAP‐resistance model included 6 variables. Prior MRSA colonization or infection within 90 days preceding admission was strongly predictive of CAP‐resistance. A composite variable consisting of direct admission from a nursing home or admission from the community after recent discharge from a nursing home was more predictive than direct admission from a nursing home alone. Exposure to cephalosporin antibiotics within the prior year was also predictive of CAP‐resistance. Subcategorizing cephalosporins by class or by most recent exposure in 90‐day increments did not improve the model. The remaining predictors in the model were guideline‐defined infusion therapy criterion, diabetes, and intensive care unit (ICU) admission.
| Guidelinedefined model of CAPResistant HCAP | AIC 461.1 | CohortDeveloped Model of CAPresistant HCAP | AIC 431.1 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P Value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Nursing home residence at time of admission | 2.6 | 1.64.4 | <0.001 | Nursing home residence or discharge 180 days prior to admission | 2.3 | 1.43.8 | 0.002 |
| Antibiotic exposure 90 days prior to admission | 1.7 | 1.02.8 | 0.054 | Positive MRSA status: 90 days prior to admission | 6.4 | 2.617.8 | <0.001 |
| Hospitalization 2 days, 90 days prior to admission | 1.6 | 1.02.6 | 0.066 | >90 days but 365 days prior to admission | 2.3 | 0.95.9 | 0.074 |
| Infusion therapy 30 days prior to admission | 1.5 | 0.82.8 | 0.173 | Cephalosporin exposure 365 days prior to admission | 1.8 | 1.12.9 | 0.019 |
| Wound care therapy 30 days prior to admission | 0.5 | 0.12.1 | 0.370 | Infusion therapy 30 days prior to admission | 1.9 | 1.03.5 | 0.044 |
| Hemodialysis therapy 30 days prior to admission | 1.8 | 0.311.2 | 0.497 | Diabetes | 1.7 | 1.02.8 | 0.044 |
| Recent immunosuppression | 0.9 | 0.51.6 | 0.670 | Direct ICU admission upon hospitalization | 1.6 | 1.02.6 | 0.053 |
Of the guideline‐defined criteria, direct admission from a nursing home was most predictive of MRSA HCAP (n = 57), followed by prior hospitalization and recent antibiotic exposure (Table 3). The cohort‐developed model of MRSA HCAP included predictors common to the CAP‐resistance model: direct admission from a nursing home or patients who were recently discharged from a nursing home, history of prior MRSA, and diabetes. Positive MRSA status within 90 days preceding admission exhibited the strongest prediction of MRSA HCAP. Exposure to anti‐pseudomonal fluoroquinolones (ciprofloxacin and levofloxacin) within the prior year was also predictive of MRSA HCAP, however, exposure to 8‐methoxy fluoroquinolone was not (crude odds ratio (OR) = 0.7 [0.3‐1.4]; final model adjusted OR = 0.6 [0.2‐1.2]). Exposure to third generation cephalosporins within the previous year was more predictive than other cephalosporin exposures, and more predictive than exposure times categorized in 90‐day increments.
| Guideline‐Defined Model of MRSA HCAP | AIC 316.3 | Cohort‐Developed Model of MRSA HCAP | AIC 279.2 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P Value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Nursing home residence at time of admission | 2.6 | 1.44.8 | 0.003 | Nursing home residence or discharge 180 days prior to admission | 2.8 | 1.55.3 | 0.002 |
| Hospitalization 2 days, 90 days prior to admission | 1.8 | 1.03.5 | 0.075 | Positive MRSA status: 90 days prior to admission | 7.7 | 3.119.6 | <0.001 |
| Antibiotic exposure 90 days prior to admission | 1.6 | 0.93.3 | 0.143 | >90 days but 365 days prior to admission | 1.4 | 0.54.1 | 0.507 |
| Recent immunosuppression | 0.6 | 0.31.3 | 0.244 | Anti‐pseudomonal fluoroquinolone exposure 365 days prior to admission | 2.4 | 1.24.6 | 0.009 |
| Wound care therapy 30 days prior to admission | 0.5 | 0.03.3 | 0.582 | Diabetes | 2.2 | 1.24.3 | 0.012 |
| Infusion therapy 30 days prior to admission | 0.9 | 0.42.0 | 0.793 | Chronic inhaled corticosteroids | 2.8 | 1.17.1 | 0.031 |
| Chronic hemodialysis 30 days prior to admission* | Third generation cephalosporin exposure 365 days prior to admission | 2.1 | 1.04.1 | 0.040 | |||
Of the guideline‐defined criteria, only prior hospitalization within 90 days and admission from a nursing home were predictive of Pseudomonas aeruginosa HCAP (n = 36) (Table 4). In the cohort‐developed model of Pseudomonas aeruginosa HCAP, Pseudomonas aeruginosa was predicted by prior cephalosporin exposure within the preceding year, prior culture of Pseudomonas aeruginosa from any anatomical source within the preceding year, and chronic steroid use of 10 mg/day prednisone equivalents. Again, the model was not improved by subcategorizing cephalosporin by class or by most recent exposure time. Finally, a negative annual trend in Pseudomonas aeruginosa HCAP was evident.
| Guideline‐defined model of Pseudomonas aeruginosa HCAP | AIC 234.8 | Cohort‐developed model of Pseudomonas aeruginosa HCAP | AIC 211.1 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Hospitalization 2 days, 90 days prior to admission | 2.5 | 1.16.0 | 0.034 | Cephalosporin exposure 365 days prior to admission | 3.8 | 1.88.8 | <0.001 |
| Nursing home residence at time of admission | 2.1 | 1.04.6 | 0.059 | Positive Pseudomonas aeruginosa culture 365 days prior to admission | 3.3 | 1.47.8 | 0.006 |
| Chronic hemodialysis 30 days prior to admission | 5.0 | 0.631.2 | 0.093 | Chronic steroid dose of 10 mg/day prednisone equivalents prior to admission | 3.0 | 1.36.9 | 0.010 |
| Antibiotic exposure 90 days prior to admission | 1.9 | 0.84.7 | 0.150 | Year of study | 0.8 | 0.71.0 | 0.069 |
| Infusion therapy 30 days prior to admission | 1.8 | 0.74.2 | 0.172 | ||||
| Recent immunosuppression | 1.1 | 0.52.5 | 0.764 | ||||
| Wound care therapy 30 days prior to admission* | |||||||
The cohort‐developed model of CAP‐resistance was re‐estimated for MRSA and Pseudomonas aeruginosa endpoints. Only positive MRSA status within 90 days preceding admission was associated with both endpoints (OR = 8.7 [3.5‐22.1] for MRSA; OR = 4.3 [1.4‐12.2] for Pseudomonas aeruginosa). Direct or recent nursing home residence (OR = 2.4 [1.3‐4.6]) and diabetes (OR = 2.4 [1.3‐4.5]) were highly predictive of MRSA, but not Pseudomonas aeruginosa (OR = 1.8 [0.8‐3.9] for nursing home residence; OR = 1.3 [0.6‐2.7] for diabetes), respectively. Cephalosporin exposure preceding admission was highly predictive of Pseudomonas aeruginosa (OR = 4.0 [1.9‐9.3]), but not with MRSA (OR = 1.1 [0.6‐2.1]). In these models, all estimated odds ratios were >1.0, consistent with the cohort‐developed model of CAP‐resistance.
For each endpoint, the cohort‐developed model was more predictive than the guideline‐defined model (Table 5) (to view ROC curves see Supporting Figures 1 to 3 in the online version of the article.). The cohort‐developed model of CAP‐resistance re‐estimated for pathogen‐specific endpoints resulted in similar predictive performance. To assess performance of the cohort developed models by facility, aROC was calculated for each of the 3 larger sites separately and for the 3 smaller facilities combined due to limited counts. Site specific aROC ranged from 0.652 to 0.762 for CAP‐resistance, 0.725 to 0.815 for MRSA, and 0.719 to 0.801 for Pseudomonas aeruginosa. The cohort‐developed model of CAP‐resistance re‐estimated for pathogen‐specific endpoints resulted in similar predictive performance.
| Model | Outcome Variable | Predictive Variables | aROC | (95% CI) | Model Comparison | aROC Difference | (95% CI) | P Value |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| 1 | CAP‐resistance | Guideline‐defined | 0.630 | (0.570, 0.691) | 2‐1 | 0.079 | (0.018, 0.139) | 0.011 |
| 2 | CAP‐resistance | Cohort‐developed | 0.709 | (0.650, 0.768) | ||||
| 3 | MRSA | Guideline‐defined | 0.638 | (0.560, 0.712) | 4‐3 | 0.135 | (0.057, 0.213) | <0.001 |
| 4 | MRSA | Cohort‐developed | 0.773 | (0.703, 0.844) | ||||
| 5 | Pseudomonas aeruginosa | Guideline‐defined | 0.680 | (0.593, 0.768) | 6‐5 | 0.090 | (0.193, 0.193) | 0.090 |
| 6 | Pseudomonas aeruginosa | Cohort‐developed | 0.770 | (0.683, 0.857) | ||||
| 7 | MRSA | Cohort‐developed from CAP‐resistance model | 0.755 | (0.682, 0.828) | 7‐4 | 0.018 | (0.067, 0.031) | 0.467 |
| 8 | Pseudomonas aeruginosa | Cohort‐developed from CAP‐resistance model | 0.755 | (0.665, 0.845) | 8‐6 | 0.015 | (0.079, 0.049) | 0.650 |
A nomogram for the cohort‐developed model of CAP‐resistance can provide the predicted probability of culturing a CAP‐resistant organism for an individual patient (Table 6). Point scores assigned to levels of variables, are summed to obtain a total score, and the total score corresponds to a predicted probability of CAP‐resistance. The prevalence of CAP‐resistance (%) from highest to lowest quartile of predicted probability was 92.9, 58.8, 32.9, and 18.5, respectively.
| A. Scoring | |
|---|---|
| Variable | Score |
| B. Predicted Probability of CAP‐Resistance* | |
| Total Score | % Chance of CAP‐Resistance |
| |
| Positive MRSA status prior to admission | |
| 90 days | +100 |
| >90 days but 365 days | +45 |
| Nursing home residence or discharge 180 days prior to admission | +45 |
| Infusion therapy 30 days prior to admission | +35 |
| Cephalosporin exposure 365 days prior to admission | +30 |
| Diabetes | +30 |
| Direct ICU admission upon hospitalization | +25 |
| <35 | <20 |
| 3565 | 2030 |
| 6590 | 3040 |
| 90110 | 4050 |
| 110130 | 5060 |
| 130155 | 6070 |
| 155185 | 7080 |
| 185230 | 8090 |
| >230 | >90 |
DISCUSSION
In this study, select ATS/IDSA guideline‐defined criteria predicted identification of CAP‐resistant organisms in patients with HCAP. Admission from a nursing home was most predictive of CAP‐resistant organisms, whereas recent hospitalization and antibiotic exposure were predictive to a lesser extent. There was weak evidence of associations between recent infusion and chronic hemodialysis criteria with MDR endpoints. Recent wound care and a composite definition of immunosuppression were not predictive of these endpoints.
The cohort‐developed model resulted in improved prediction of CAP‐resistance endpoints. Culture history, particularly history of MRSA within 90 days preceding admission, was a strong predictor of MDR endpoints. The MRSA history variable definition included cultures from all anatomical sources and nares polymerase chain reaction surveillance results, the latter increasing in 2007‐2008 due to the implementation of the VA MRSA initiative.18 This finding suggests that prior culture results should be considered when selecting empirical antimicrobial therapy, and the rapid proliferation of electronic medical records increases potential to utilize this information routinely. While the guideline‐defined nursing home admission criterion was a strong predictor of CAP‐resistance, admission from the community after recent discharge from a nursing home, in addition to direct admission from a nursing home, was also important.
Similarities in variables included in the pathogen‐specific and CAP‐resistance models reflect the importance of MRSA in defining the CAP‐resistance endpoint. Both CAP‐resistance and MRSA models included prior MRSA status, diabetes, and ICU admission, whereas cephalosporin exposure was common to the Pseudomonas aeruginosa and CAP‐resistance models. Annual trends in CAP‐resistance and MRSA recovery were not identified. The negative annual trend in Pseudomonas aeruginosa HCAP is unexplained and beyond the scope of this study. The percentage of culture‐positive admissions with Pseudomonas aeruginosa HCAP averaged 12% in 2003‐2006, but dropped to <5% in 2007‐2008. A potential explanation is that identification and isolation of patients with MRSA, as a result of the VA‐wide MRSA initiative, may have impacted Pseudomonas aeruginosa colonization by isolating patients co‐colonized with these pathogens during prior healthcare exposures. This is consistent with the observation that when the cohort‐derived CAP‐resistance model was refit with the Pseudomonas aeruginosa endpoint, recent MRSA colonization was strongly predictive of Pseudomonas aeruginosa. Despite differences between variables in pathogen‐specific and CAP‐resistant models, the CAP‐resistance model provided a similar degree of MRSA and Pseudomonas aeruginosa prediction. Finally, as a study purpose included developing best predictive models for each endpoint, and not merely identifying associations, there were other plausible models not reported.
Study strengths included use of the VISN20 Data Warehouse, which provided an integrated outpatient and inpatient medical record. This facilitated analysis of prior healthcare exposures and inpatient study endpoints. In addition, poor blood and sputum specimens and unlikely pneumonia pathogens were not included in establishing MDR endpoints. The variable set explored in regression modeling was extensive and detailed, and analysis included time and intensity‐based components of the variables. Importantly, a standardized approach to regression modeling was specified in advance, which included identification of variables with high potential for association with MDR endpoints, model selection by AIC, re‐evaluation of guideline‐defined criteria and variables of lower interest, and bootstrapped internal model validation.19
Study limitations included the use of ICD‐9 codes to establish a pneumonia diagnosis, which may lack sensitivity and specificity. However, an enhanced ICD‐9based algorithm superior to other claims‐based definitions of pneumonia was utilized.4, 20 Veterans may have received care at non‐VA facilities impacting identification of all healthcare system exposures preceding admission. Data for microbial endpoints were obtained from sterile and non‐sterile site cultures, and it was not possible to determine if the cultured organisms were truly pathogenic. While pathogen‐specific endpoints were not affected, the use of expert rules in select cases to establish CAP‐resistance may have impacted precision for this endpoint. It is also possible that refitting the cohort‐developed CAP‐resistance model for pathogen‐specific endpoints resulted in optimistic aROC due to model over‐fitting. Finally, the cohort was comprised of elderly males, and caution is warranted in extrapolating the results to other populations.
The predictive ability of the guideline‐defined criteria to identify patients with MDR pathogens has been studied. A prospective observational cohort study of 625 consecutive ICU admissions determined that the guideline‐defined criteriaprior antimicrobial treatment, nursing home residence, and prior hospitalizationwere associated with recovery of MDR colonization.21 Shorr et al., investigating a retrospective cohort of 619 patients with HCAP, reported that recent hospitalization, nursing home residence, hemodialysis, and ICU admission were associated with infections caused by CAP‐resistant organisms.22 This study did not report antimicrobial exposures. Our study complements these studies by evaluating existing HCAP guideline criteria, and identifying specific antibiotic exposure, prior culture data, comorbid illness, and immunosuppressive medications that are predictive of MDR infection.
Studies comparing the bacterial etiology of patients with pneumonia in nursing homes relative to CAP, have demonstrated mixed results in recovery of Gram‐negative MDR pathogens, but generally increased MRSA pneumonia.3 Our study suggests that a nursing home stay in the last 6 months is associated with an increased risk for MRSA, but not Pseudomonas aeruginosa, although this was limited by small sample size. Recent infusion therapy has not been previously reported to be associated with MDR pathogens in an HCAP population. In our study, this criterion was predictive of CAP‐resistance in the cohort‐developed model, but not in conjunction with other variables in the guideline‐defined model. Predictors of pathogen‐specific HCAP are limited to an aforementioned single prior study, which identified recent hospitalization, nursing home residence, and ICU admission as risk factors for MRSA HCAP.22
Many studies have investigated risks for infection with MRSA and Pseudomonas aeruginosa outside of the context of HCAP. Predictor variables in cohort‐developed pathogen‐specific models in our study are known risk factors for colonization or infection with these pathogens. For example, antecedent MRSA colonization has been noted as a strong risk factor for MRSA infection, particularly pneumonia.23, 24 Further, patients with diabetes and inhaled corticosteroid exposure are immunosuppressed and at increased risk for colonization with MRSA.25, 26 Likewise, bronchiolar colonization and corticosteroid exposures are known risk factors for pneumonia due to Pseudomonas aeruginosa.27
Many studies have identified prior antibiotic use as a risk factor for infections caused by MRSA and Pseudomonas aeruginosa. However, this criterion is excessively broad and specific antimicrobial exposures carry different magnitudes of risk. Third generation cephalosporins and anti‐pseudomonal fluoroquinolones are commonly reported antibiotics associated with risk for MRSA infection, whereas 8‐methoxy fluoroquinolones appear not to possess the same effect.2831 Likewise, cephalosporins have been reported as risk factors for MDR Pseudomonas aeruginosa infections.32
Several areas of research involving HCAP MDR risk should be investigated. First, the predictive models developed in our and other studies should be evaluated in larger, more diverse populations to establish generalizability. Second, empirical broad‐spectrum antibiotic therapy in all patients with HCAP results in overtreatment of many patients. To date, no reported models provided optimal performance for selecting empirical therapy for unstable ICU patients with HCAP, and many patients do not receive de‐escalation therapy. Thus, models to identify patients with low probability of MDR pathogens upon admission and to aid in de‐escalation are warranted. Finally, the negative trend in Pseudomonas aeruginosa HCAP requires confirmation and further study.
In conclusion, of the ATS/IDSA guideline‐defined criteria for MDR, nursing home admission, recent hospitalization, and antibiotic exposure were predictive of the recovery of CAP‐resistant organisms. Alternative models primarily based on prior culture data, specific antibiotic exposures, and immunosuppression‐related variables improved predictive performance of HCAP associated with MDR.
Healthcare associated pneumonia (HCAP) is defined as pneumonia that is present upon admission, and occurs in patients that have recently been hospitalized, reside in a nursing home, or have had other recent healthcare exposures. Practice guidelines developed by the American Thoracic Society (ATS) and the Infectious Diseases Society of America (IDSA), recommend strategies for the diagnosis and treatment of patients with HCAP.1 A premise of the guidelines is that recent healthcare exposure places patients at risk for infection due to multi‐drug resistant (MDR) pathogens such as methicillin‐resistant Staphylococcus aureus (MRSA) or Pseudomonas aeruginosa. In addition to criteria utilized to define HCAP, the guidelines state that recent immunosuppression and antibiotic exposure are risk factors for pneumonia due to MDR pathogens. In contrast to the treatment of community‐acquired pneumonia (CAP), the guidelines recommend empirical administration of antibiotics with activity against MRSA and Pseudomonas aeruginosa for all patients with HCAP.
We recently reported that antimicrobial resistance to CAP antibiotics (CAP‐resistance) was identified in one‐third of culture‐positive patients with HCAP.2 Data regarding the predictive ability of the guideline‐defined criteria specific to HCAP are limited.3 Evaluation and potential refinement of the criteria to identify patients at risk for MDR pathogens can aid in making antibiotic‐related treatment decisions.
The purposes of this study are to: 1) develop and validate a model to predict CAP‐resistance among patients with HCAP, and to compare the model's predictive performance to a model that includes traditional guideline‐defined risk factors; and 2) develop models to predict recovery of pathogen‐specific etiology (MRSA and Pseudomonas aeruginosa), and to compare the predictive performance of the pathogen‐specific and CAP‐resistance models.
METHODS
Patients with HCAP who were admitted to 6 Veterans Affairs Medical Centers (VAMC) in the northwestern United States between January 1, 2003 and December 31, 2008 were included in the retrospective cohort study. The cohort was identified utilizing medical records data extracted from the Veterans Integrated Service Network (VISN20) Data Warehouse. The Data Warehouse is a centralized open architecture relational database that houses medical and administrative records data for VISN20 patients. This research complies with all federal guidelines and VAMC policies relative to human subjects and clinical research.
Subjects were identified by the following pneumonia‐related discharge International Classification of Diseases (ICD‐9 CM) codes: 1) a primary diagnosis of 480‐483; 485‐487.0 (pneumonia); or 2) a primary diagnosis of 507.0 (pneumonitis), 518.8 (respiratory failure), or 0.38 (septicemia), and a secondary diagnosis of 480‐483; 485‐487.0.4 Eligibility required that patients received antibiotic therapy for pneumonia within 24 hours of admission, continue inpatient treatment for >24 hours, and meet any of the following guideline‐defined criteria: 1) hospitalization during the preceding 90 days; 2) admission from a nursing home; 3) outpatient or home wound care, outpatient or home infusion therapy, or chronic hemodialysis.1 In addition, patients not meeting guideline‐defined criteria, who had frequent healthcare system exposure, defined as 12 Emergency Department, Medicine, or Surgery clinic visits within 90 days of admission, were also included. Patients were excluded if they were directly transferred from another hospital, or had pneumonia‐related ICD‐9 codes but received inpatient care for pneumonia in a non‐VA hospital.
Study data included medical records for the year prior to admission for HCAP through 30 days afterwards. Data included: demographics; domicile preceding admission; healthcare utilization including diagnosis and procedure codes; inpatient medications administered, and outpatient prescription fills; vital signs; and laboratory test results, including cultures and susceptibilities.
Guideline‐defined criteria for predicting CAP‐resistance were similar to those used to identify the study cohort. Nursing home admission included patients who were directly admitted from a nursing home, skilled nursing facility, or domiciliary. Prior hospitalization 2 days within 90 days was calculated by summing the length of stay for all admissions during the preceding 90 days. Outpatient intravenous therapy, chronic hemodialysis, and wound care therapy was determined from medication administration records and relevant Current Procedural Terminology (CPT) or ICD‐9 procedure codes for care administered within 30 days. Antibiotic exposure was defined as administration of 1 dose of antibiotic during inpatient care, or fill of an outpatient prescription for 1 antibiotic dose within 90 days preceding admission. Immunosuppression was defined as: human immunodeficiency virus (HIV) diagnosis; white blood cell (WBC) count of 2500 cells/mm3 within 30 days of admission; corticosteroid ingestion during prior admission, or outpatient prescription fills for a corticosteroid with quantity sufficient to last 14 days preceding admission; or inpatient ingestion of, or outpatient prescription fills for, transplant or rheumatologic‐related immunosuppressants within 90 days preceding admission.
Additional variables assessed to predict CAP‐resistance were obtained as follows. First, modifications of guideline‐defined criteria were constructed. These included: direct nursing home admission, or recent nursing home stay preceding admission; total days of hospitalization within 90 days preceding admission; specific antibiotic exposures, including dates since last exposure preceding admission; and individual components of the immunosuppression criterion. Other cohort‐developed variables included: demographics; substance use history; chronic comorbidity determined by individual and composite measures of Charlson score; pulmonary disease history (eg, bronchiectasis); type and frequency of outpatient visits; consecutive (2) prescription fills for chronic medications of interest; clinical and surveillance culture results preceding admission; admitting ward; vital signs; and relevant hematology and chemistry labs.5
Sputum, blood, and bronchoscopy‐collected cultures obtained within 48 hours after admission were assessed to determine specimen acceptability. Poor sputum specimens were defined by Gram stain quantitative results indicating >10 epithelial cells (EPI) per low power field (LPF), or in the absence of quantitative results, semi‐quantitative results indicating 2‐4+EPI. Single positive blood cultures with results indicating likely contaminants were considered poor specimens. All bronchoscopy‐obtained specimens were considered acceptable. All cultures classified as poor specimens were excluded, and microbiology results were evaluated for the remaining specimens.2, 6 Organisms thought to represent colonization or contamination were excluded: coagulase‐negative (CN) Staphylococcus, Enterococcus sp, Bacillus sp, Proprionibacterium sp, and Candida sp. Recovery of a potential pneumonia pathogen from 1 acceptable culture constituted a culture‐positive admission.
CAP‐resistance was determined for each isolate. CAP‐resistance was defined as non‐susceptibility to non‐pseudomonal third generation cephalosporins (ceftriaxone or cefotaxime) or non‐pseudomonal 8‐methoxy fluoroquinolones (moxifloxacin, gatifloxacin), the VA preferred agents for treatment of CAP.7 There were differences between facilities in susceptibility reporting criteria; therefore, the following approach was used to determine CAP‐resistance. First, MRSA and Pseudomonas aeruginosa isolates were classified as CAP‐resistant. Second, susceptibility results were directly utilized to determine CAP‐resistance if both antibiotic results were available. Third, if only a surrogate antibiotic from a class was reported, a representative antibiotic consistent with Clinical Laboratory Standards Institute reporting criteria was utilized.8 Finally, expert rules determined CAP‐resistance for select potential pneumonia pathogens (eg, Haemophilus sp) if antibiotic susceptibility results for both cephalosporin and fluoroquinolone classes were not reported.815 Presence of 1 CAP‐resistant isolate resulted in a CAP‐resistant classification for an admission. MRSA and Pseudomonas aeruginosa endpoints were defined in a similar manner. Only the first admission for each patient was utilized in the analysis.
The probability of CAP‐resistance was predicted from guideline‐defined criteria (guideline‐defined model) with logistic regression. Next, non‐guideline variables were classified as high, medium, or low interest for association with CAP‐resistance. Variables were assessed for collinearity. A model of CAP‐resistance was developed from variables of high interest. Guideline‐defined criteria were omitted to allow consideration of more specific measures (eg, specific antibiotic exposures as opposed to receipt of antibiotics within the preceding 90 days) during this stage. Next, guideline‐defined criteria, and subsequently variables of lesser interest, were added in an attempt to improve the model. Annual trends and plausible interactions were considered. Model selection was by Akaike's Information Criterion (AIC).16 To promote model reliability, the final model was required to lack evidence of over‐fitting in bootstrapped internal validation.17 The guideline‐defined and cohort‐developed models were compared by difference in area under receiver operating characteristic (aROC) curves. The model development process was repeated for MRSA and Pseudomonas aeruginosa endpoints. Finally, to determine if the CAP‐resistance model sufficiently predicted pathogen‐specific MDR, the CAP‐resistance model was re‐estimated for MRSA and Pseudomonas aeruginosa endpoints. Statistical analysis was performed with R version 2.10.0 (The R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
The cohort was comprised of 1300 patients with HCAP. Of these, 375 (28.8% [26.4‐31.4]) met culture‐positive criteria for potential pneumonia pathogens. CAP‐resistant organisms were identified in 118 (31.5% [26.8‐36.4]) patients within 48 hours of admission. CAP‐resistant organisms included: MRSA (49.2% [40.4‐58.1]), Pseudomonas aeruginosa (29.5% [21.9‐38.1]), Enterobacteriaceae (11.4% [6.5‐18.0]), Gram‐negative non‐enterics (8.3% [4.2‐14.4]), Streptococcus pneumoniae (1.5% [0.2‐5.4]), and opportunistic organisms (eg, Mycobacterium spp) (8.3% [4.2‐14.4]). Differences in select characteristics and exposures between culture‐positive and culture‐negative admissions, as well as CAP‐resistant and CAP‐sensitive admissions, were evident (Table 1).
| Characteristic | Culture‐Negative Admissions (n = 925) | Culture‐Positive Admissions (n = 375) | P Value | CAP‐Sensitive Admissions (n = 257) | CAP‐Resistant Admissions (n = 118) | P Value |
|---|---|---|---|---|---|---|
| ||||||
| Demographics | ||||||
| Age (mean/SD) | 71.9 (12.1) | 71.4 (12.4) | 0.44 | 70.4 (12.4) | 72.9 (12.3) | 0.07 |
| Gender (% male) | 97.1 | 98.8 | 0.07 | 98.4 | 99.2 | 1.00 |
| Primary inclusion diagnosis (%) | ||||||
| Pneumonia | 93.1 | 85.9 | <0.01 | 87.2 | 83.1 | 0.87 |
| Aspiration pneumonitis with pneumonia pneumonia witpneumonia | 1.5 | 4.3 | 0.02 | 4.6 | 3.3 | 0.48 |
| Septicemia with pneumonia | 2.6 | 6.2 | <0.01 | 5.1 | 8.5 | 0.25 |
| Respiratory failure with pneumonia | 2.8 | 3.5 | 0.50 | 3.1 | 5.1 | 0.38 |
| HCAP inclusion criteria (%) | ||||||
| Nursing home residence | 31.2 | 35.9 | 0.08 | 30.4 | 46.6 | <0.01 |
| Hospitalization of >2 days in last 90 days | 58.7 | 57.6 | 0.73 | 52.1 | 62.7 | 0.06 |
| Intravenous therapy in last 30 days | 19.5 | 20.7 | 0.61 | 19.5 | 21.2 | 0.68 |
| Outpatient wound care in last 30 days | 2.7 | 2.7 | 1.00 | 3.1 | 1.7 | 0.73 |
| Chronic dialysis in last 30 days | 2.5 | 1.7 | 0.45 | 1.2 | 2.5 | 0.38 |
| Hospitalization duration 0‐2 days in last 90 days | 10.2 | 11.2 | 0.57 | 12.5 | 5.9 | 0.22 |
| >12 ED or clinic visits in last 90 days | 44.1 | 44.6 | 0.86 | 44.0 | 41.5 | 0.74 |
| Other guideline‐defined MDR criteria (%) | ||||||
| Antibiotics in last 90 days | 63.8 | 61.6 | 0.47 | 57.2 | 66.1 | 0.11 |
| Recent immunosuppression | 19.3 | 23.9 | 0.53 | 24.1 | 22.0 | 0.70 |
| Severity of illness (%) | ||||||
| Admitted to the ICU | 21.8 | 41.6 | <0.01 | 26.3 | 38.6* | <0.01 |
| Mechanical ventilation | 5.6 | 12.7 | <0.01 | 12.1 | 12.7 | 0.87 |
| Comorbidity (%) | ||||||
| Charlson comorbidity score (mean/SD) | 4.3 (3.0) | 4.3 (3.0) | 0.85 | 4.1 (3.1) | 4.5 (2.8) | 0.20 |
| Diabetes | 33.8 | 29.2 | 0.10 | 27.2 | 39.0 | 0.07 |
| Prior antibiotic use (%) | ||||||
| Any cephalosporin | 42.0 | 39.9 | 0.48 | 32.3 | 51.7 | <0.01 |
| Third generation cephalosporin | 24.5 | 23.7 | 0.78 | 18.3 | 30.5 | 0.01 |
| Anti‐pseudomonal fluoroquinolone | 28.5 | 28.4 | 1.0 | 23.3 | 37.3 | 0.02 |
| 8‐Methoxy fluoroquinolone | 20.1 | 23.9 | 0.10 | 24.1 | 24.5 | 1.00 |
| Prior corticosteroid use (%) | ||||||
| Systemic steroids (>10 mg/day prednisone) | 11.1 | 13.2 | 0.28 | 11.3 | 16.1 | 0.24 |
| Inhaled steroids | 7.5 | 10.0 | 0.11 | 8.9 | 10.2 | 0.71 |
| Prior MDR cultured (%) | ||||||
| MRSA within <90 days | 4.2 | 7.7 | <0.01 | 2.7 | 15.3 | <0.01 |
| MRSA >90 days but <365 days | 5.6 | 6.5 | 0.54 | 3.9 | 10.2 | 0.03 |
| P. aeruginosa within 365 days | 5.7 | 11.5 | <0.01 | 5.8 | 19.5 | <0.01 |
Of the guideline‐defined criteria, direct admission from a nursing home, prior hospitalization, and recent antibiotic exposure were associated with CAP‐resistance (Table 2). The cohort‐derived CAP‐resistance model included 6 variables. Prior MRSA colonization or infection within 90 days preceding admission was strongly predictive of CAP‐resistance. A composite variable consisting of direct admission from a nursing home or admission from the community after recent discharge from a nursing home was more predictive than direct admission from a nursing home alone. Exposure to cephalosporin antibiotics within the prior year was also predictive of CAP‐resistance. Subcategorizing cephalosporins by class or by most recent exposure in 90‐day increments did not improve the model. The remaining predictors in the model were guideline‐defined infusion therapy criterion, diabetes, and intensive care unit (ICU) admission.
| Guidelinedefined model of CAPResistant HCAP | AIC 461.1 | CohortDeveloped Model of CAPresistant HCAP | AIC 431.1 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P Value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Nursing home residence at time of admission | 2.6 | 1.64.4 | <0.001 | Nursing home residence or discharge 180 days prior to admission | 2.3 | 1.43.8 | 0.002 |
| Antibiotic exposure 90 days prior to admission | 1.7 | 1.02.8 | 0.054 | Positive MRSA status: 90 days prior to admission | 6.4 | 2.617.8 | <0.001 |
| Hospitalization 2 days, 90 days prior to admission | 1.6 | 1.02.6 | 0.066 | >90 days but 365 days prior to admission | 2.3 | 0.95.9 | 0.074 |
| Infusion therapy 30 days prior to admission | 1.5 | 0.82.8 | 0.173 | Cephalosporin exposure 365 days prior to admission | 1.8 | 1.12.9 | 0.019 |
| Wound care therapy 30 days prior to admission | 0.5 | 0.12.1 | 0.370 | Infusion therapy 30 days prior to admission | 1.9 | 1.03.5 | 0.044 |
| Hemodialysis therapy 30 days prior to admission | 1.8 | 0.311.2 | 0.497 | Diabetes | 1.7 | 1.02.8 | 0.044 |
| Recent immunosuppression | 0.9 | 0.51.6 | 0.670 | Direct ICU admission upon hospitalization | 1.6 | 1.02.6 | 0.053 |
Of the guideline‐defined criteria, direct admission from a nursing home was most predictive of MRSA HCAP (n = 57), followed by prior hospitalization and recent antibiotic exposure (Table 3). The cohort‐developed model of MRSA HCAP included predictors common to the CAP‐resistance model: direct admission from a nursing home or patients who were recently discharged from a nursing home, history of prior MRSA, and diabetes. Positive MRSA status within 90 days preceding admission exhibited the strongest prediction of MRSA HCAP. Exposure to anti‐pseudomonal fluoroquinolones (ciprofloxacin and levofloxacin) within the prior year was also predictive of MRSA HCAP, however, exposure to 8‐methoxy fluoroquinolone was not (crude odds ratio (OR) = 0.7 [0.3‐1.4]; final model adjusted OR = 0.6 [0.2‐1.2]). Exposure to third generation cephalosporins within the previous year was more predictive than other cephalosporin exposures, and more predictive than exposure times categorized in 90‐day increments.
| Guideline‐Defined Model of MRSA HCAP | AIC 316.3 | Cohort‐Developed Model of MRSA HCAP | AIC 279.2 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P Value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Nursing home residence at time of admission | 2.6 | 1.44.8 | 0.003 | Nursing home residence or discharge 180 days prior to admission | 2.8 | 1.55.3 | 0.002 |
| Hospitalization 2 days, 90 days prior to admission | 1.8 | 1.03.5 | 0.075 | Positive MRSA status: 90 days prior to admission | 7.7 | 3.119.6 | <0.001 |
| Antibiotic exposure 90 days prior to admission | 1.6 | 0.93.3 | 0.143 | >90 days but 365 days prior to admission | 1.4 | 0.54.1 | 0.507 |
| Recent immunosuppression | 0.6 | 0.31.3 | 0.244 | Anti‐pseudomonal fluoroquinolone exposure 365 days prior to admission | 2.4 | 1.24.6 | 0.009 |
| Wound care therapy 30 days prior to admission | 0.5 | 0.03.3 | 0.582 | Diabetes | 2.2 | 1.24.3 | 0.012 |
| Infusion therapy 30 days prior to admission | 0.9 | 0.42.0 | 0.793 | Chronic inhaled corticosteroids | 2.8 | 1.17.1 | 0.031 |
| Chronic hemodialysis 30 days prior to admission* | Third generation cephalosporin exposure 365 days prior to admission | 2.1 | 1.04.1 | 0.040 | |||
Of the guideline‐defined criteria, only prior hospitalization within 90 days and admission from a nursing home were predictive of Pseudomonas aeruginosa HCAP (n = 36) (Table 4). In the cohort‐developed model of Pseudomonas aeruginosa HCAP, Pseudomonas aeruginosa was predicted by prior cephalosporin exposure within the preceding year, prior culture of Pseudomonas aeruginosa from any anatomical source within the preceding year, and chronic steroid use of 10 mg/day prednisone equivalents. Again, the model was not improved by subcategorizing cephalosporin by class or by most recent exposure time. Finally, a negative annual trend in Pseudomonas aeruginosa HCAP was evident.
| Guideline‐defined model of Pseudomonas aeruginosa HCAP | AIC 234.8 | Cohort‐developed model of Pseudomonas aeruginosa HCAP | AIC 211.1 | ||||
|---|---|---|---|---|---|---|---|
| Variable | OR | 95% CI | P Value | Variable | OR | 95% CI | P value |
| |||||||
| (Intercept) | NA | NA | NA | (Intercept) | NA | NA | NA |
| Hospitalization 2 days, 90 days prior to admission | 2.5 | 1.16.0 | 0.034 | Cephalosporin exposure 365 days prior to admission | 3.8 | 1.88.8 | <0.001 |
| Nursing home residence at time of admission | 2.1 | 1.04.6 | 0.059 | Positive Pseudomonas aeruginosa culture 365 days prior to admission | 3.3 | 1.47.8 | 0.006 |
| Chronic hemodialysis 30 days prior to admission | 5.0 | 0.631.2 | 0.093 | Chronic steroid dose of 10 mg/day prednisone equivalents prior to admission | 3.0 | 1.36.9 | 0.010 |
| Antibiotic exposure 90 days prior to admission | 1.9 | 0.84.7 | 0.150 | Year of study | 0.8 | 0.71.0 | 0.069 |
| Infusion therapy 30 days prior to admission | 1.8 | 0.74.2 | 0.172 | ||||
| Recent immunosuppression | 1.1 | 0.52.5 | 0.764 | ||||
| Wound care therapy 30 days prior to admission* | |||||||
The cohort‐developed model of CAP‐resistance was re‐estimated for MRSA and Pseudomonas aeruginosa endpoints. Only positive MRSA status within 90 days preceding admission was associated with both endpoints (OR = 8.7 [3.5‐22.1] for MRSA; OR = 4.3 [1.4‐12.2] for Pseudomonas aeruginosa). Direct or recent nursing home residence (OR = 2.4 [1.3‐4.6]) and diabetes (OR = 2.4 [1.3‐4.5]) were highly predictive of MRSA, but not Pseudomonas aeruginosa (OR = 1.8 [0.8‐3.9] for nursing home residence; OR = 1.3 [0.6‐2.7] for diabetes), respectively. Cephalosporin exposure preceding admission was highly predictive of Pseudomonas aeruginosa (OR = 4.0 [1.9‐9.3]), but not with MRSA (OR = 1.1 [0.6‐2.1]). In these models, all estimated odds ratios were >1.0, consistent with the cohort‐developed model of CAP‐resistance.
For each endpoint, the cohort‐developed model was more predictive than the guideline‐defined model (Table 5) (to view ROC curves see Supporting Figures 1 to 3 in the online version of the article.). The cohort‐developed model of CAP‐resistance re‐estimated for pathogen‐specific endpoints resulted in similar predictive performance. To assess performance of the cohort developed models by facility, aROC was calculated for each of the 3 larger sites separately and for the 3 smaller facilities combined due to limited counts. Site specific aROC ranged from 0.652 to 0.762 for CAP‐resistance, 0.725 to 0.815 for MRSA, and 0.719 to 0.801 for Pseudomonas aeruginosa. The cohort‐developed model of CAP‐resistance re‐estimated for pathogen‐specific endpoints resulted in similar predictive performance.
| Model | Outcome Variable | Predictive Variables | aROC | (95% CI) | Model Comparison | aROC Difference | (95% CI) | P Value |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| 1 | CAP‐resistance | Guideline‐defined | 0.630 | (0.570, 0.691) | 2‐1 | 0.079 | (0.018, 0.139) | 0.011 |
| 2 | CAP‐resistance | Cohort‐developed | 0.709 | (0.650, 0.768) | ||||
| 3 | MRSA | Guideline‐defined | 0.638 | (0.560, 0.712) | 4‐3 | 0.135 | (0.057, 0.213) | <0.001 |
| 4 | MRSA | Cohort‐developed | 0.773 | (0.703, 0.844) | ||||
| 5 | Pseudomonas aeruginosa | Guideline‐defined | 0.680 | (0.593, 0.768) | 6‐5 | 0.090 | (0.193, 0.193) | 0.090 |
| 6 | Pseudomonas aeruginosa | Cohort‐developed | 0.770 | (0.683, 0.857) | ||||
| 7 | MRSA | Cohort‐developed from CAP‐resistance model | 0.755 | (0.682, 0.828) | 7‐4 | 0.018 | (0.067, 0.031) | 0.467 |
| 8 | Pseudomonas aeruginosa | Cohort‐developed from CAP‐resistance model | 0.755 | (0.665, 0.845) | 8‐6 | 0.015 | (0.079, 0.049) | 0.650 |
A nomogram for the cohort‐developed model of CAP‐resistance can provide the predicted probability of culturing a CAP‐resistant organism for an individual patient (Table 6). Point scores assigned to levels of variables, are summed to obtain a total score, and the total score corresponds to a predicted probability of CAP‐resistance. The prevalence of CAP‐resistance (%) from highest to lowest quartile of predicted probability was 92.9, 58.8, 32.9, and 18.5, respectively.
| A. Scoring | |
|---|---|
| Variable | Score |
| B. Predicted Probability of CAP‐Resistance* | |
| Total Score | % Chance of CAP‐Resistance |
| |
| Positive MRSA status prior to admission | |
| 90 days | +100 |
| >90 days but 365 days | +45 |
| Nursing home residence or discharge 180 days prior to admission | +45 |
| Infusion therapy 30 days prior to admission | +35 |
| Cephalosporin exposure 365 days prior to admission | +30 |
| Diabetes | +30 |
| Direct ICU admission upon hospitalization | +25 |
| <35 | <20 |
| 3565 | 2030 |
| 6590 | 3040 |
| 90110 | 4050 |
| 110130 | 5060 |
| 130155 | 6070 |
| 155185 | 7080 |
| 185230 | 8090 |
| >230 | >90 |
DISCUSSION
In this study, select ATS/IDSA guideline‐defined criteria predicted identification of CAP‐resistant organisms in patients with HCAP. Admission from a nursing home was most predictive of CAP‐resistant organisms, whereas recent hospitalization and antibiotic exposure were predictive to a lesser extent. There was weak evidence of associations between recent infusion and chronic hemodialysis criteria with MDR endpoints. Recent wound care and a composite definition of immunosuppression were not predictive of these endpoints.
The cohort‐developed model resulted in improved prediction of CAP‐resistance endpoints. Culture history, particularly history of MRSA within 90 days preceding admission, was a strong predictor of MDR endpoints. The MRSA history variable definition included cultures from all anatomical sources and nares polymerase chain reaction surveillance results, the latter increasing in 2007‐2008 due to the implementation of the VA MRSA initiative.18 This finding suggests that prior culture results should be considered when selecting empirical antimicrobial therapy, and the rapid proliferation of electronic medical records increases potential to utilize this information routinely. While the guideline‐defined nursing home admission criterion was a strong predictor of CAP‐resistance, admission from the community after recent discharge from a nursing home, in addition to direct admission from a nursing home, was also important.
Similarities in variables included in the pathogen‐specific and CAP‐resistance models reflect the importance of MRSA in defining the CAP‐resistance endpoint. Both CAP‐resistance and MRSA models included prior MRSA status, diabetes, and ICU admission, whereas cephalosporin exposure was common to the Pseudomonas aeruginosa and CAP‐resistance models. Annual trends in CAP‐resistance and MRSA recovery were not identified. The negative annual trend in Pseudomonas aeruginosa HCAP is unexplained and beyond the scope of this study. The percentage of culture‐positive admissions with Pseudomonas aeruginosa HCAP averaged 12% in 2003‐2006, but dropped to <5% in 2007‐2008. A potential explanation is that identification and isolation of patients with MRSA, as a result of the VA‐wide MRSA initiative, may have impacted Pseudomonas aeruginosa colonization by isolating patients co‐colonized with these pathogens during prior healthcare exposures. This is consistent with the observation that when the cohort‐derived CAP‐resistance model was refit with the Pseudomonas aeruginosa endpoint, recent MRSA colonization was strongly predictive of Pseudomonas aeruginosa. Despite differences between variables in pathogen‐specific and CAP‐resistant models, the CAP‐resistance model provided a similar degree of MRSA and Pseudomonas aeruginosa prediction. Finally, as a study purpose included developing best predictive models for each endpoint, and not merely identifying associations, there were other plausible models not reported.
Study strengths included use of the VISN20 Data Warehouse, which provided an integrated outpatient and inpatient medical record. This facilitated analysis of prior healthcare exposures and inpatient study endpoints. In addition, poor blood and sputum specimens and unlikely pneumonia pathogens were not included in establishing MDR endpoints. The variable set explored in regression modeling was extensive and detailed, and analysis included time and intensity‐based components of the variables. Importantly, a standardized approach to regression modeling was specified in advance, which included identification of variables with high potential for association with MDR endpoints, model selection by AIC, re‐evaluation of guideline‐defined criteria and variables of lower interest, and bootstrapped internal model validation.19
Study limitations included the use of ICD‐9 codes to establish a pneumonia diagnosis, which may lack sensitivity and specificity. However, an enhanced ICD‐9based algorithm superior to other claims‐based definitions of pneumonia was utilized.4, 20 Veterans may have received care at non‐VA facilities impacting identification of all healthcare system exposures preceding admission. Data for microbial endpoints were obtained from sterile and non‐sterile site cultures, and it was not possible to determine if the cultured organisms were truly pathogenic. While pathogen‐specific endpoints were not affected, the use of expert rules in select cases to establish CAP‐resistance may have impacted precision for this endpoint. It is also possible that refitting the cohort‐developed CAP‐resistance model for pathogen‐specific endpoints resulted in optimistic aROC due to model over‐fitting. Finally, the cohort was comprised of elderly males, and caution is warranted in extrapolating the results to other populations.
The predictive ability of the guideline‐defined criteria to identify patients with MDR pathogens has been studied. A prospective observational cohort study of 625 consecutive ICU admissions determined that the guideline‐defined criteriaprior antimicrobial treatment, nursing home residence, and prior hospitalizationwere associated with recovery of MDR colonization.21 Shorr et al., investigating a retrospective cohort of 619 patients with HCAP, reported that recent hospitalization, nursing home residence, hemodialysis, and ICU admission were associated with infections caused by CAP‐resistant organisms.22 This study did not report antimicrobial exposures. Our study complements these studies by evaluating existing HCAP guideline criteria, and identifying specific antibiotic exposure, prior culture data, comorbid illness, and immunosuppressive medications that are predictive of MDR infection.
Studies comparing the bacterial etiology of patients with pneumonia in nursing homes relative to CAP, have demonstrated mixed results in recovery of Gram‐negative MDR pathogens, but generally increased MRSA pneumonia.3 Our study suggests that a nursing home stay in the last 6 months is associated with an increased risk for MRSA, but not Pseudomonas aeruginosa, although this was limited by small sample size. Recent infusion therapy has not been previously reported to be associated with MDR pathogens in an HCAP population. In our study, this criterion was predictive of CAP‐resistance in the cohort‐developed model, but not in conjunction with other variables in the guideline‐defined model. Predictors of pathogen‐specific HCAP are limited to an aforementioned single prior study, which identified recent hospitalization, nursing home residence, and ICU admission as risk factors for MRSA HCAP.22
Many studies have investigated risks for infection with MRSA and Pseudomonas aeruginosa outside of the context of HCAP. Predictor variables in cohort‐developed pathogen‐specific models in our study are known risk factors for colonization or infection with these pathogens. For example, antecedent MRSA colonization has been noted as a strong risk factor for MRSA infection, particularly pneumonia.23, 24 Further, patients with diabetes and inhaled corticosteroid exposure are immunosuppressed and at increased risk for colonization with MRSA.25, 26 Likewise, bronchiolar colonization and corticosteroid exposures are known risk factors for pneumonia due to Pseudomonas aeruginosa.27
Many studies have identified prior antibiotic use as a risk factor for infections caused by MRSA and Pseudomonas aeruginosa. However, this criterion is excessively broad and specific antimicrobial exposures carry different magnitudes of risk. Third generation cephalosporins and anti‐pseudomonal fluoroquinolones are commonly reported antibiotics associated with risk for MRSA infection, whereas 8‐methoxy fluoroquinolones appear not to possess the same effect.2831 Likewise, cephalosporins have been reported as risk factors for MDR Pseudomonas aeruginosa infections.32
Several areas of research involving HCAP MDR risk should be investigated. First, the predictive models developed in our and other studies should be evaluated in larger, more diverse populations to establish generalizability. Second, empirical broad‐spectrum antibiotic therapy in all patients with HCAP results in overtreatment of many patients. To date, no reported models provided optimal performance for selecting empirical therapy for unstable ICU patients with HCAP, and many patients do not receive de‐escalation therapy. Thus, models to identify patients with low probability of MDR pathogens upon admission and to aid in de‐escalation are warranted. Finally, the negative trend in Pseudomonas aeruginosa HCAP requires confirmation and further study.
In conclusion, of the ATS/IDSA guideline‐defined criteria for MDR, nursing home admission, recent hospitalization, and antibiotic exposure were predictive of the recovery of CAP‐resistant organisms. Alternative models primarily based on prior culture data, specific antibiotic exposures, and immunosuppression‐related variables improved predictive performance of HCAP associated with MDR.
- American Thoracic Society; Infectious Diseases Society of America.Guidelines for the management of adults with hospital‐acquired, ventilator‐associated, and healthcare‐associated pneumonia.Am J Respir Crit Care Med.2005;171(4):388–416.
- ,,,.The etiology of health care associated pneumonia (HCAP) [abstract K‐282]. Presented at the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy; September2009; San Francisco, CA.
- ,.What are the important risk factors for healthcare‐associated pneumonia?Semin Respir Crit Care Med.2009;30(1):26–35.
- ,,,.Accuracy of administrative data for identifying patients with pneumonia.Am J Med Qual.2005;20(6):319–328.
- ,,.Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases.J Clin Epidemiol.1992;45:613–619.
- ,,,.How often is a microbial etiology identified in health care associated pneumonia (HCAP)? [abstract K‐289]. Presented at the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy; September2009; San Francisco, CA.
- Fluoroquinolone use criteria. Washington D.C. Guidelines Developed by the Pharmacy Benefits Management Strategic Health Care Group and Medical Advisory Panel, Veterans Health Administration, Department of Veterans Affairs. Last update, November2006. http://www.pbm.va.gov/Clinical%20Guidance/Criteria%20For%20Use/Fluoroquinolone,%20Criteria%20for%20Use.pdf. Last accessed August 20th, 2011.
- Performance Standards for Antimicrobial Susceptibility Testing; 18th Informational Supplement. M100‐S18.Wayne, PA:Clinical Laboratory Standards Institute;2009.
- ,,.Antimicrobial activity of DC‐159a, a new fluoroquinolone, against 1,149 recently collected clinical isolates.Antimicrob Agents Chemother.2008;52(10):3763–3775.
- ,,, et al.Mortality from invasive pneumococcal pneumonia in the era of antibiotic resistance, 1995–1997.Am J Public Health.2000;90(2):223–229.
- ,.Comparative activity of moxifloxacin against Gram‐positive clinical isolates.J Antimicrob Chemother.2000;45(1):31–39.
- ,,.Antimicrobial susceptibility of extended‐spectrum‐beta‐lactamase producers and multi‐drug resistant Acinetobacter baumannii throughout the United States and comparative in vitro activity of tigecycline, a new glycylcycline antimicrobial.Diagn Microbiol Infect Dis.2007;57(4):423–428.
- ,,.Antimicrobial susceptibility profile of contemporary clinical strains of Stenotropomonas maltophila isolates: can moxifloxacin activity be predicted by levofloxacin MIC results?J Chemother.2008;20(1):38–42.
- ,,, et al.Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults.Clin Infect Dis.2007;44(suppl 2):S27–S72.
- Gilbert DN, Moellering RC, Eliopoulos GM, Sande, MA, eds.The Sanford Guide to Antimicrobial Therapy.38th ed.Speryville, VA:Antimicrobial Therapy;2008.
- .A new look at the statistical model identification.IEEE Trans Automat Contr.1974;19(6):716–723.
- .Estimating the error rate of a prediction rule: improvement on cross‐validation.J Am Stat Assoc.1987;78:316–331.
- ,,, et. Al.Beyond beta: lessons learned from implementation of the Department of Veterans Affairs Methicillin‐Resistant Staphylococcus aureus Prevention Initiative.Infect Control Hosp Epidemiol.2010;31(7):763–765.
- ,,.An appraisal of multivariable logistic models in the pulmonary and critical care literature.Chest.2003;123(3):923–928.
- ,,,,,.Improved clinical outcomes with utilization of a community‐acquired pneumonia guideline.Chest.2006;130(3):794–799.
- ,,,,,.Accuracy of American Thoracic Society/Infectious Diseases Society of America criteria in predicting infection or colonization with multidrug‐resistant bacteria at intensive‐care unit admission.Clin Microbiol Infect.2009;16(7):902–908.
- ,,,.Prediction of infections due to antibiotic resistant bacteria by select risk factors for healthcare associated pneumonia.Arch Intern Med.2008;168(20):2205–2210.
- ,,,,.Methicillin‐resistant Staphylococcus aureus (MRSA) nares colonization at hospital admission and its effect on subsequent MRSA infection.Clin Infect Dis.2004;39(6):776–782.
- ,.Risk of infection and death due to methicillin‐resistant Staphylococcus aureus in long‐term carriers.Clin Infect Dis.2008;47(2):176–181.
- ,,,.Epidemiology of methicillin‐resistant Staphylococcus aureus in a university medical center day care facility.Infect Control Hosp Epidemiol.2009;30(10):985–992.
- ,,,,,.Colonization and infection with antibiotic‐resistant bacteria in a long‐term care facility.J Am Geriatr Soc.1994;42(10):1062–1069.
- ,,, et al.American Thoracic Society. Guidelines for the management of adults with community‐acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy, and prevention.Am J Respir Crit Care Med.2001;163(7):1730–1754.
- ,.Modification in prescribing practices for third‐generation cephalosporins and ciprofloxacin is associated with a reduction in methicillin‐resistant Staphylococcus aureus bacteraemia rate.J Hosp Infect.2008;69(4):328–336.
- ,,,.Evaluation of an intervention designed to decrease the rate of nosocomial methicillin‐resistant Staphylococcus aureus infection by encouraging decreased fluoroquinolone use.Infect Control Hosp Epidemiol.2006;27(2):155–169.
- ,.Using interrupted time series analysis to assess associations of fluoroquinolone formulary changes with susceptibility of gram‐negative pathogens and isolation rates of methicillin‐resistant Staphylococcus aureus.Antimicrob Agents Chemother.2006;50(6):2106–2112.
- ,.Dichotomous selection of high‐level oxacillin resistance in Staphylococcus aureus by fluoroquinolones.Int J Antimicrob Agents.2010;36(3):216–221.
- ,,,,.Multidrug‐resistant Pseudomonas aeruginosa: risk factors and clinical impact.Antimicrob Agents Chemother.2006;50(1):43–48.
- American Thoracic Society; Infectious Diseases Society of America.Guidelines for the management of adults with hospital‐acquired, ventilator‐associated, and healthcare‐associated pneumonia.Am J Respir Crit Care Med.2005;171(4):388–416.
- ,,,.The etiology of health care associated pneumonia (HCAP) [abstract K‐282]. Presented at the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy; September2009; San Francisco, CA.
- ,.What are the important risk factors for healthcare‐associated pneumonia?Semin Respir Crit Care Med.2009;30(1):26–35.
- ,,,.Accuracy of administrative data for identifying patients with pneumonia.Am J Med Qual.2005;20(6):319–328.
- ,,.Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases.J Clin Epidemiol.1992;45:613–619.
- ,,,.How often is a microbial etiology identified in health care associated pneumonia (HCAP)? [abstract K‐289]. Presented at the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy; September2009; San Francisco, CA.
- Fluoroquinolone use criteria. Washington D.C. Guidelines Developed by the Pharmacy Benefits Management Strategic Health Care Group and Medical Advisory Panel, Veterans Health Administration, Department of Veterans Affairs. Last update, November2006. http://www.pbm.va.gov/Clinical%20Guidance/Criteria%20For%20Use/Fluoroquinolone,%20Criteria%20for%20Use.pdf. Last accessed August 20th, 2011.
- Performance Standards for Antimicrobial Susceptibility Testing; 18th Informational Supplement. M100‐S18.Wayne, PA:Clinical Laboratory Standards Institute;2009.
- ,,.Antimicrobial activity of DC‐159a, a new fluoroquinolone, against 1,149 recently collected clinical isolates.Antimicrob Agents Chemother.2008;52(10):3763–3775.
- ,,, et al.Mortality from invasive pneumococcal pneumonia in the era of antibiotic resistance, 1995–1997.Am J Public Health.2000;90(2):223–229.
- ,.Comparative activity of moxifloxacin against Gram‐positive clinical isolates.J Antimicrob Chemother.2000;45(1):31–39.
- ,,.Antimicrobial susceptibility of extended‐spectrum‐beta‐lactamase producers and multi‐drug resistant Acinetobacter baumannii throughout the United States and comparative in vitro activity of tigecycline, a new glycylcycline antimicrobial.Diagn Microbiol Infect Dis.2007;57(4):423–428.
- ,,.Antimicrobial susceptibility profile of contemporary clinical strains of Stenotropomonas maltophila isolates: can moxifloxacin activity be predicted by levofloxacin MIC results?J Chemother.2008;20(1):38–42.
- ,,, et al.Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community‐acquired pneumonia in adults.Clin Infect Dis.2007;44(suppl 2):S27–S72.
- Gilbert DN, Moellering RC, Eliopoulos GM, Sande, MA, eds.The Sanford Guide to Antimicrobial Therapy.38th ed.Speryville, VA:Antimicrobial Therapy;2008.
- .A new look at the statistical model identification.IEEE Trans Automat Contr.1974;19(6):716–723.
- .Estimating the error rate of a prediction rule: improvement on cross‐validation.J Am Stat Assoc.1987;78:316–331.
- ,,, et. Al.Beyond beta: lessons learned from implementation of the Department of Veterans Affairs Methicillin‐Resistant Staphylococcus aureus Prevention Initiative.Infect Control Hosp Epidemiol.2010;31(7):763–765.
- ,,.An appraisal of multivariable logistic models in the pulmonary and critical care literature.Chest.2003;123(3):923–928.
- ,,,,,.Improved clinical outcomes with utilization of a community‐acquired pneumonia guideline.Chest.2006;130(3):794–799.
- ,,,,,.Accuracy of American Thoracic Society/Infectious Diseases Society of America criteria in predicting infection or colonization with multidrug‐resistant bacteria at intensive‐care unit admission.Clin Microbiol Infect.2009;16(7):902–908.
- ,,,.Prediction of infections due to antibiotic resistant bacteria by select risk factors for healthcare associated pneumonia.Arch Intern Med.2008;168(20):2205–2210.
- ,,,,.Methicillin‐resistant Staphylococcus aureus (MRSA) nares colonization at hospital admission and its effect on subsequent MRSA infection.Clin Infect Dis.2004;39(6):776–782.
- ,.Risk of infection and death due to methicillin‐resistant Staphylococcus aureus in long‐term carriers.Clin Infect Dis.2008;47(2):176–181.
- ,,,.Epidemiology of methicillin‐resistant Staphylococcus aureus in a university medical center day care facility.Infect Control Hosp Epidemiol.2009;30(10):985–992.
- ,,,,,.Colonization and infection with antibiotic‐resistant bacteria in a long‐term care facility.J Am Geriatr Soc.1994;42(10):1062–1069.
- ,,, et al.American Thoracic Society. Guidelines for the management of adults with community‐acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy, and prevention.Am J Respir Crit Care Med.2001;163(7):1730–1754.
- ,.Modification in prescribing practices for third‐generation cephalosporins and ciprofloxacin is associated with a reduction in methicillin‐resistant Staphylococcus aureus bacteraemia rate.J Hosp Infect.2008;69(4):328–336.
- ,,,.Evaluation of an intervention designed to decrease the rate of nosocomial methicillin‐resistant Staphylococcus aureus infection by encouraging decreased fluoroquinolone use.Infect Control Hosp Epidemiol.2006;27(2):155–169.
- ,.Using interrupted time series analysis to assess associations of fluoroquinolone formulary changes with susceptibility of gram‐negative pathogens and isolation rates of methicillin‐resistant Staphylococcus aureus.Antimicrob Agents Chemother.2006;50(6):2106–2112.
- ,.Dichotomous selection of high‐level oxacillin resistance in Staphylococcus aureus by fluoroquinolones.Int J Antimicrob Agents.2010;36(3):216–221.
- ,,,,.Multidrug‐resistant Pseudomonas aeruginosa: risk factors and clinical impact.Antimicrob Agents Chemother.2006;50(1):43–48.
Copyright © 2011 Society of Hospital Medicine
Outcomes of Delayed ICU Transfer
Hospitalized patients who require transfer from medical wards to the intensive care unit (ICU) have high in‐hospital mortality, in some reports exceeding 55%.14 In a previous report in this journal, we found that while these unplanned ICU transfers occurred in only 4% of hospitalizations, they were present in nearly one‐quarter of fatal hospitalizations and were associated with substantial increases in resource utilization.4 For these reasons, interventions aimed at identifying and treating this high‐risk group have received considerable attention and have been proposed as measures of inpatient safety.2, 49
Notably, mortality among patients with unplanned ICU transfers exceeds mortality among patients admitted to the ICU directly from the emergency department (ED)a group traditionally considered to have the highest risk of death.13, 10 Previous single‐center studies suggest that increased mortality rates are present even among patients transferred within 24 hours of hospital admission, and reinforce the notion that earlier recognition of critical illness may result in improved outcomes.1113 However, these studies have been performed primarily in small cohorts of heterogeneous patients, and may obscure the independent effect of unplanned transfers on mortality and hamper efforts to use unplanned transfer rates as a metric of healthcare quality.1, 2, 4, 9
In this study, we evaluated early unplanned ICU transfers drawn from a cohort of 499,995 hospitalizations in an integrated healthcare delivery system. Using patient data, extracted from the automated electronic medical record, we matched unplanned transfer cases to patients directly admitted to the ICU and described the association between delayed ICU transfers and adverse outcomes.
METHODS
Setting and Participants
We performed a retrospective analysis of adult patient (age 18 years) hospitalizations at 21 Northern California Kaiser Permanente (KP) Medical Care Program hospitals between January 2007 and December 2009. This work expanded on our previous report of hospital stays from November 2006 to January 2008.4 The 21 study hospitals used the same electronic health information systems; databases captured admission, discharge, and bed history data. The use of these databases for research has been described in our previous study and other reports; hospital characteristics, unit staffing, and resource levels have also been detailed previously.4, 1417 This study was approved by the KP Institutional Review Board.
Identifying Unplanned Transfers
We evaluated patients with medical hospitalizationsdefined as those whose first hospital location was not in a surgical setting such as the operating room or post‐anesthesia recovery areawhose admission originated in the ED; patients admitted for surgery were removed because of significant differences in observed mortality (see Supporting Information Appendix Figure 1 and Appendix Table 1 in the online version of this article). Patients whose admission did not originate in the ED were excluded to eliminate confounding resulting from differences in preadmission care. We also excluded patients admitted for gynecological and pregnancy‐related care because of low hospital mortality.
Initial patient locations included the medical wards (wards); the transitional care unit (TCU); and the intensive care unit (ICU). Bed history data, based on time stamps and available for all patients, were used to track patient locations from the time of admission, defined as the first non‐ED hospital location, until discharge. Patient length of stay (LOS) was calculated at each location and for the entire hospitalization.
Transfers to the ICU after a patient's initial admission to the ward or TCU were termed unplanned (or delayed) ICU transfers; patients admitted from the ED to the ICU were termed direct ICU admit patients. Direct ICU admit patients were excluded from the unplanned transfer group even if they required a readmission to the ICU later in their hospital course. We focused on patients with unplanned ICU transfers early after hospitalization to identify those in whom prompt recognition and intervention could be effective; thus, our primary analyses were on patients with transfers within 24 hours of admission. In secondary analysis, we also evaluated patients with unplanned ICU transfers occurring within 48 hours after hospital admission.
Admission Severity of Illness
To account for severity of illness at admission, we used a predicted mortality measure developed at KP.14 This method strictly utilizes information available prior to hospital admissionincluding that from the ED; variables included age, gender, admitting diagnosis, and measures of laboratory test and comorbid disease burden. The method, derived using 259,669 KP hospitalizations, produced a c‐statistic of 0.88 for inpatient mortality; external validation, based on 188,724 hospitalizations in Ottawa, produced a c‐statistic of 0.92.14, 18
Admitting diagnoses were based on admission International Classification of Diseases, 9th revision (ICD‐9) codes, and grouped into 44 broad Primary Conditions based on pathophysiologic plausibility and mortality rates.14 The method also quantified each patient's physiologic derangement and preexisting disease burden based on automated laboratory and comorbidity measuresthe Laboratory Acute Physiology Score (LAPS) and the Comorbidity Point Score (COPS).14
In brief, the LAPS was derived from 14 possible test results obtained in the 24‐hour time period preceding hospitalization, including: anion gap; arterial pH, PaCO2, and PaO2; bicarbonate; serum levels of albumin, total bilirubin, creatinine, glucose, sodium, and troponin I; blood urea nitrogen; creatinine; hematocrit; and total white blood cell count.14 The COPS was calculated from each subject's inpatient and outpatient diagnoses, based on Diagnostic Cost Groups software,19 during the 12‐month period preceding hospitalization.14 Increasing LAPS and COPS values were associated with increases in hospital mortality; detailed information about the development, application, and validation are available in previous work.14, 18
Statistical Analysis
Evaluating excess adverse outcomes associated with unplanned transfers requires adequate control of confounding variables. Our approach to reduce confounding was multivariable case matchinga technique used for assessing treatment effects in observational data.20, 21 Patients with unplanned transfersidentified as caseswere matched with similar controls based on observed variables at the time of hospital admission.
We first matched patients with unplanned ICU transfers within 24 hours of hospital admission to direct ICU admit controls based on predicted in‐hospital mortality (to within 1%); age (by decade); gender; and admitting diagnosis. If a case was matched to multiple controls, we selected 1 control with the most similar admission characteristics (weekday or weekend admission and nursing shift). The risk of death associated with unplanned transfers was estimated using multivariable conditional logistic regression. In secondary analysis, we repeated this analysis only among case‐control pairs within the same hospital facilities.
To cross‐validate the results from multivariable matching techniques, we also performed mixed‐effects multivariable logistic regression including all early unplanned transfer patients and direct ICU admit patients, while adjusting for predicted hospital mortality, age, gender, admitting diagnosis, LAPS, COPS, weekend versus weekday admission, nursing shift, and hospital facility random effects. We repeated these same analyses where cases were defined as patients transferred to the ICU within 48 hours of hospitalization.
Unplanned Transfer Timing
Using bed history data, we identified the elapsed time from admission to unplanned transfer, and categorized patients in increments of elapsed time from admission to unplanned transfer. Time‐to‐unplanned transfer was summarized using Kaplan‐Meier curve.
All analyses were performed in Stata/IC 11.0 for Mac (StataCorp LP, College Station, TX). Continuous variables were reported as mean standard deviation (SD). Cohort comparisons were performed with analysis of variance (ANOVA). Categorical variables were summarized using frequencies and compared with chi‐squared testing. A P value <0.05 was considered statistically significant.
RESULTS
During the study period, 313,797 medical hospitalizations originated in the ED (Table 1). Overall, patients' mean age was 67 18 years; 53.7% were female. Patient characteristics differed significantly based on the need for ICU admission. For example, average LAPS was highest among patients admitted directly to the ICU and lowest among patients who never required ICU care (P < 0.01). Patients with unplanned ICU transfers during hospitalization had longer length of stay and higher hospital mortality than direct ICU admit patients (P < 0.01). Overall, more than 1 in 15 patients experienced an unplanned transfer to the ICU.
| Early Delayed ICU Transfer (by Elapsed Time Since Hospital Admission) | ||||
|---|---|---|---|---|
| Variable | Overall | Within 24 hr | Within 48 hr | Direct ICU Admit |
| ||||
| No. (%) | 313,797 | 6,369 (2.0) | 9,816 (3.1) | 29,929 (9.5) |
| Age* | 67 18 | 67 16 | 68 16 | 64 17 |
| Female* | 169,358 (53.7) | 3,125 (49.1) | 4,882 (49.7) | 14,488 (48.4) |
| Weekend admission* | 83,327 (26.6) | 1,783 (28.0) | 2,733 (27.8) | 8,152 (27.2) |
| Nursing shift at admission* | ||||
| Day (7 AM‐3 PM) | 65,303 (20.8) | 1,335 (21.0) | 2,112 (21.5) | 7,065 (23.6) |
| Evening (3 PM‐11 PM) | 155,037 (49.4) | 2,990 (47.0) | 4,691 (47.8) | 13,158 (44.0) |
| Night (11 PM‐7 AM) | 93,457 (29.8) | 2,044 (32.1) | 3,013 (30.7) | 9,706 (32.4) |
| Initial hospital location* | ||||
| Ward | 234,915 (82.8) | 5,177 (81.3) | 7,987 (81.4) | |
| Transitional care unit | 48,953 (17.2) | 1,192 (18.7) | 1,829 (18.6) | |
| LAPS* | 24 19 | 28 20 | 28 20 | 35 25 |
| COPS* | 98 67 | 105 70 | 106 70 | 99 71 |
| Length of stay (days) | 4.6 7.5 | 8.4 12.2 | 9.1 13.4 | 6.4 9.5 |
| In‐hospital mortality | 12,686 (4.0) | 800 (12.6) | 1,388 (14.1) | 3,602 (12.0) |
The majority of unplanned transfers occurred within the first 48 hours of hospitalization (57.6%, Figure 1); nearly 80% occurred within the first 4 days. The rate of unplanned transfer peaked within 24 hours of hospital admission and decreased gradually as elapsed hospital LOS increased (Figure 1). While most patients experienced a single unplanned ICU transfer, 12.7% required multiple transfers to the ICU throughout their hospitalization.
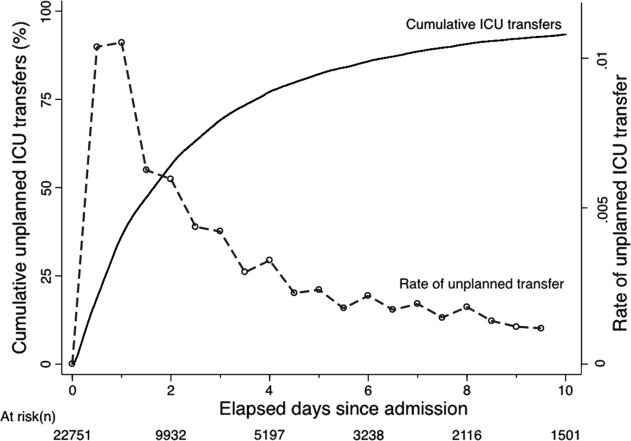
Multivariable case matching between unplanned transfer cases within 24 hours of admission and direct ICU admit controls resulted in 5839 (92%) case‐control pairs (Table 2). Matched pairs were most frequently admitted with diagnoses in Primary Condition groups that included respiratory infections and pneumonia (15.6%); angina, acute myocardial infarction (AMI), and heart failure (15.6%); or gastrointestinal bleeding (13.8%).
| ICU Cohorts (by Elapsed Time to Transfer Since Hospital Admission) | ||||
|---|---|---|---|---|
| Within 24 hr (n = 5,839) | Within 48 hr (n = 8,976) | |||
| Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | |
| ||||
| Age | 67 16 | 66 16 | 67 16 | 67 16 |
| Female | 2,868 (49.1) | 2,868 (49.1) | 4,477 (49.9) | 4,477 (49.9) |
| Admitting diagnosis | ||||
| Pneumonia | 911 (15.6) | 911 (15.6) | 1,526 (17.0) | 1,526 (17.0) |
| Heart failure or MI | 909 (15.6) | 909 (15.6) | 1,331 (14.8) | 1,331 (14.8) |
| Gastrointestinal bleeding | 806 (13.8) | 806 (13.8) | 1,191 (13.3) | 1,191 (13.3) |
| Infections (including sepsis) | 295 (5.1) | 295 (5.1) | 474 (5.3) | 474 (5.3) |
| Outcomes | ||||
| Length of stay (days)* | 8 12 | 6 9 | 9 13 | 6 9 |
| In‐hospital mortality* | 678 (11.6) | 498 (8.5) | 1,181 (13.2) | 814 (9.1) |
In‐hospital mortality was significantly higher among cases (11.6%) than among ICU controls (8.5%, P < 0.001); mean LOS was also longer among cases (8 12 days) than among controls (6 9 days, P < 0.001). Unplanned transfer cases were at an increased odds of death when compared with ICU controls (adjusted odds ratio [OR], 1.44; 95% confidence interval [CI], 1.26‐1.64; P < 0.001); they also had a significantly higher observed‐to‐expected mortality ratio. When cases and controls were matched by hospital facility, the number of case‐control pairs decreased (2949 pairs; 42% matching frequency) but the odds of death was of similar magnitude (OR, 1.43; 95% CI, 1.21‐1.68; P < 0.001). Multivariable mixed‐effects logistic regression including all early unplanned transfer and direct ICU admit patients produced an effect size of similar magnitude (OR, 1.37; 95% CI, 1.24‐1.50; P < 0.001).
Results were similar when cases were limited to patients with transfers within 12 hours of admission; mortality was 10.9% among cases and 9.1% among controls (P = 0.02). When including patients with unplanned transfers within 48 hours of hospital admission, the difference in mortality between cases and controls increased (13.2% vs 9.1%, P < 0.001). The odds of death among patients with unplanned transfers increased as the elapsed time between admission and ICU transfer lengthened (Figure 2); the adjusted OR was statistically significant at each point between 8 and 48 hours.
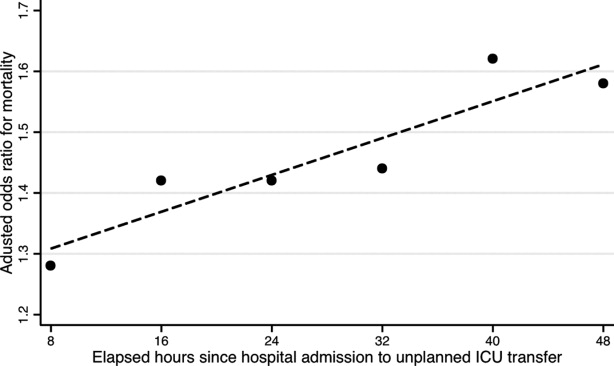
When stratified by admitting diagnosis groups, cases with unplanned transfers within the first 48 hours had increased mortality compared with matched controls in some categories (Table 3). For example, for patients in the respiratory infection and pneumonia group, mortality was 16.8% among unplanned transfer cases and 13.0% among early matched ICU controls (P < 0.01). A similar pattern was present in groups including: gastrointestinal bleeding, chronic obstructive pulmonary disease (COPD) exacerbation, and seizure groups (Table 3). However, for patients with AMI alone, mortality was 5.0% among cases and 3.7% among matched controls (P = 0.12). Patients with sepsis had a mortality rate of 15.2% among cases and 20.8% among matched controls (P = 0.07). Similarly, patients with stroke had a mortality rate of 12.4% among unplanned transfer cases and 11.4% in the matched controls (P = 0.54).
| Primary Condition Group | Mortality in ICU Case‐Control Cohorts, No. (%) | |||
|---|---|---|---|---|
| Within 24 hr | Within 48 hr | |||
| Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | |
| ||||
| Respiratory infections | 143 (15.7) | 126 (13.8) | 493 (16.8) | 380 (13.0) |
| Angina, heart failure, or MI | 60 (6.6) | 41 (4.5) | 324 (7.7) | 152 (3.6) |
| Acute MI alone | 16 (5.7) | 17 (6.1) | 82 (5.0) | 61 (3.7) |
| Gastrointestinal bleeding | 96 (11.9) | 55 (6.8) | 549 (19.3) | 188 (6.6) |
| Infections including sepsis | 20 (9.8) | 52 (11.2) | 228 (14.8) | 220 (14.2) |
| Sepsis alone | 32 (18.9) | 31 (18.3) | 123 (15.2) | 168 (20.8) |
| COPD exacerbation | 20 (9.8) | 12 (5.9) | 74 (10.8) | 43 (6.3) |
| Stroke | 18 (10.2) | 19 (10.8) | 77 (12.4) | 71 (11.4) |
| Seizure | 21 (8.6) | 9 (3.7) | 68 (7.1) | 34 (3.6) |
DISCUSSION
This study found that unplanned ICU transfers were common among medical patients, occurring in 5% of all hospitalizations originating in the ED. The majority of unplanned transfers occurred within 48 hours of admission; the rate of ICU transfers peaked within 24 hours after hospitalization. Compared with patients admitted directly from the ED to the ICU, those transferred early after admission had significantly increased mortality; for example, patients transferred within 24 hours were at a 44% increased odds of hospital death. The adverse outcomes associated with unplanned transfers varied considerably by admission diagnosis subgroups.
Our findings confirm previous reports of increased mortality among patients with unplanned ICU transfers. Escarce and Kelley reported that patients admitted to the ICU from non‐ED locationsincluding wards, intermediate care units, and other hospitalswere at an increased risk of hospital death.1 Multiple subsequent studies have confirmed the increased mortality among patients with unplanned transfers.24, 10, 13, 22, 23 We previously evaluated patients who required a transfer to any higher level of care and reported an observed‐to‐expected mortality ratio of 2.93.4
Fewer studies, however, have evaluated the association between the timing of unplanned transfers and inpatient outcomes; previous small reports suggest that delays in ICU transfer adversely affect mortality and length of stay.12, 13, 24 Parkhe et al. compared 99 direct ICU admit patients with 23 who experienced early unplanned transfers; mortality at 30 days was significantly higher among patients with unplanned transfers.13 The current multifacility study included considerably more patients and confirmed an in‐hospital mortality gapalbeit a smaller onebetween patients with early transfers and those directly admitted to the ICU.
We focused on unplanned transfers during the earliest phase of hospitalization to identify patients who might benefit from improved recognition of, and intervention for, impending critical illness. We found that even patients requiring transfers within 8 hours of hospital admission were at an increased risk of death. Bapoje et al. recently reported that as many as 80% of early unplanned transfers were preventable and that most resulted from inappropriate admission triage.11 Together, these findings suggest that heightened attention to identifying such patients at admission or within the first day of hospitalizationwhen the rates of unplanned transfers peakis critical.
Several important limitations should be recognized in interpreting these results. First, this study was not designed to specifically identify the reasons for unplanned transfers, limiting our ability to characterize episodes in which timely care could have prevented excess mortality. Notably, while previous work suggests that many early unplanned transfers might be prevented with appropriate triage, it is likely that some excess deaths are not preventable even if every patient could be admitted to the ICU directly.
We were able to characterize patient outcomes by admitting diagnoses. Patients admitted for pneumonia and respiratory infection, gastrointestinal bleeding, COPD exacerbation, or seizures demonstrated excess mortality compared with matched ICU controls, while those with AMI, sepsis, and stroke did not. It is possible that differences in diagnosis‐specific excess mortality resulted from increasing adherence to well‐defined practice guidelines for specific high‐risk conditions.2527 For example, international awareness campaigns for the treatment of sepsis, AMI, and strokeSurviving Sepsis, Door‐to‐Balloon, and F.A.S.T.emphasize early interventions to minimize morbidity and mortality.
Second, the data utilized in this study were based on automated variables extracted from the electronic medical record. Mortality prediction models based on automated variables have demonstrated excellent performance among ICU and non‐ICU populations14, 18, 28; however, the inclusion of additional data (eg, vital signs or neurological status) would likely improve baseline risk adjustment.5, 10, 2931 Multiple studies have demonstrated that vital signs and clinician judgment can predict patients at an increased risk of deterioration.5, 10, 2931 Such data might also provide insight into residual factors that influenced clinicians' decisions to triage patients to an ICU versus non‐ICU admissiona focus area of our ongoing research efforts. Utilizing electronically available data, however, facilitated the identification of a cohort of patients far larger than that in prior studies. Where previous work has also been limited by substantial variability in baseline characteristics among study subjects,1, 2, 12, 13 our large sample produced a high percentage of multivariable case matches.
Third, we chose to match patients with a severity of illness index based on variables available at the time of hospital admission. While this mortality prediction model has demonstrated excellent performance in internal and external populations,14, 18 it is calibrated for general inpatient, rather than critically ill, populations. It remains possible that case matching with ICU‐specific severity of illness scores might alter matching characteristics, however, previous studies suggest that severity of illness, as measured by these scores, is comparable between direct ICU admits and early ICU transfers.13 Importantly, our matching procedure avoided the potential confounding known to exist with the use of prediction models based on discharge or intra‐hospitalization data.32, 33
Finally, while we were able to evaluate unplanned transfer timing in a multifacility sample, all patient care occurred within a large integrated healthcare delivery system. The overall observed mortality in our study was lower than that reported in prior studies which considered more limited patient cohorts.1, 2, 12, 13, 22 Thus, differences in patient case‐mix or ICU structure must be considered when applying our results to other healthcare delivery systems.
This hypothesis‐generating study, based on a large, multifacility sample of hospitalizations, suggests several areas of future investigation. Future work should detail specific aspects of care among patients with unplanned transfer, including: evaluating the structures and processes involved in triage decisions, measuring the effects on mortality through implementation of interventions (eg, rapid response teams or diagnosis‐specific treatment protocols), and defining the causes and risk factors for unplanned transfers by elapsed time.
In conclusion, the risk of an unplanned ICU transfera common event among hospitalized patientsis highest within 24 hours of hospitalization. Patients with early unplanned transfers have increased mortality and length of stay compared to those admitted directly to the ICU. Even patients transferred to the ICU within 8 hours of hospital admission are at an increased risk of death when compared with those admitted directly. Substantial variability in unplanned transfer outcomes exists based on admitting diagnoses. Future research should characterize unplanned transfers in greater detail with the goal of identifying patients that would benefit from improved triage and early ICU transfer.
- ,.Admission source to the medical intensive care unit predicts hospital death independent of APACHE II score.JAMA.1990;264(18):2389–2394.
- ,,,,,.Unplanned admission to intensive care after emergency hospitalisation: risk factors and development of a nomogram for individualising risk.Resuscitation.2009;80(2):224–230.
- ,.Outcome of intensive care patients in a group of British intensive care units.Crit Care Med.1998;26(8):1337–1345.
- ,,,,,.Intra‐hospital transfers to a higher level of care: contribution to total hospital and intensive care unit (ICU) mortality and length of stay (LOS).J Hosp Med.2010;6(2):74–80.
- ,.Medical patients at high risk for catastrophic deterioration.Crit Care Med.1987;15(5):510–515.
- ,,, et al.Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial.Lancet.2005;365(9477):2091–2097.
- ,,, et al.Effect of a rapid response team on hospital‐wide mortality and code rates outside the ICU in a children's hospital.JAMA.2007;298(19):2267–2274.
- ,,,,,.Validity of unplanned admission to an intensive care unit as a measure of patient safety in surgical patients.Anesthesiology.2005;103(6):1121–1129.
- ,,,.The 100,000 lives campaign: setting a goal and a deadline for improving health care quality.JAMA.2006;295(3):324–327.
- ,,, et al.Duration of life‐threatening antecedents prior to intensive care admission.Intensive Care Med.2002;28(11):1629–1634.
- ,,,.Unplanned transfers to a medical intensive care unit: causes and relationship to preventable errors in care.J Hosp Med.2011;6(2):68–72.
- ,,,,.Inpatient transfers to the intensive care unit: delays are associated with increased mortality and morbidity.J Gen Intern Med.2003;18(2):77–83.
- ,,,.Outcome of emergency department patients with delayed admission to an intensive care unit.Emerg Med (Fremantle).2002;14(1):50–57.
- ,,,,,.Risk‐adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases.Med Care.2008;46(3):232–239.
- ,,, et al.Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases.Am J Manag Care.2008;14(3):158–166.
- .Linking automated databases for research in managed care settings.Ann Intern Med.1997;127(8 pt 2):719–724.
- ,,, et al.Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice?JAMA.2003;290(20):2685–2692.
- ,,,.The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population.J Clin Epidemiol.2009;63(7):798–803.
- ,.Refinements to the diagnostic cost group (DCG) model.Inquiry.1995;32(4):418–429.
- ,.Excess length of stay, charges, and mortality attributable to medical injuries during hospitalization.JAMA.2003;290(14):1868–1874.
- .Optimal matching in observational studies.J Am Stat Assoc.1989;84:1024–1032.
- ,,,.Admissions to intensive care units from emergency departments: a descriptive study.Emerg Med J.2005;22(6):423–428.
- ,,,.Using administrative data to develop a nomogram for individualising risk of unplanned admission to intensive care.Resuscitation.2008;79(2):241–248.
- ,,,.Unplanned intensive care unit transfers: a useful tool to improve quality of care [abstract]. In: Hospital Medicine 2010 abstract booklet. Society of Hospital Medicine 2010 Annual Meeting, April 9–11, 2010, Washington, DC;2010:10–11.
- ,,, et al.Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008.Crit Care Med.2008;36(1):296–327.
- ,,, et al.2009 Focused Updates: ACC/AHA Guidelines for the Management of Patients With ST‐Elevation Myocardial Infarction (updating the 2004 Guideline and 2007 Focused Update) and ACC/AHA/SCAI Guidelines on Percutaneous Coronary Intervention (updating the 2005 Guideline and 2007 Focused Update): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.Circulation.2009;120(22):2271–2306.
- ,,, et al.Translating evidence into practice: a decade of efforts by the American Heart Association/American Stroke Association to reduce death and disability due to stroke: a presidential advisory from the American Heart Association/American Stroke Association.Stroke.2010;41(5):1051–1065.
- ,,, et al.Veterans Affairs intensive care unit risk adjustment model: validation, updating, recalibration.Crit Care Med.2008;36(4):1031–1042.
- ,,, et al.Recommended guidelines for monitoring, reporting, and conducting research on medical emergency team, outreach, and rapid response systems: an Utstein‐style scientific statement: a scientific statement from the International Liaison Committee on Resuscitation (American Heart Association, Australian Resuscitation Council, European Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Council of Southern Africa, and the New Zealand Resuscitation Council); the American Heart Association Emergency Cardiovascular Care Committee; the Council on Cardiopulmonary, Perioperative, and Critical Care; and the Interdisciplinary Working Group on Quality of Care and Outcomes Research.Circulation.2007;116(21):2481–2500.
- ,,,,,.Realizing the potential of clinical judgment: a real‐time strategy for predicting outcomes and cost for medical inpatients.Am J Med.2000;109(3):189–195.
- ,,.Physiological values and procedures in the 24 h before ICU admission from the ward.Anaesthesia.1999;54(6):529–534.
- ,,,,,.Predicting who dies depends on how severity is measured: implications for evaluating patient outcomes.Ann Intern Med.1995;123(10):763–770.
- ,,, et al.Enhancement of claims data to improve risk adjustment of hospital mortality.JAMA.2007;297(1):71–76.
Hospitalized patients who require transfer from medical wards to the intensive care unit (ICU) have high in‐hospital mortality, in some reports exceeding 55%.14 In a previous report in this journal, we found that while these unplanned ICU transfers occurred in only 4% of hospitalizations, they were present in nearly one‐quarter of fatal hospitalizations and were associated with substantial increases in resource utilization.4 For these reasons, interventions aimed at identifying and treating this high‐risk group have received considerable attention and have been proposed as measures of inpatient safety.2, 49
Notably, mortality among patients with unplanned ICU transfers exceeds mortality among patients admitted to the ICU directly from the emergency department (ED)a group traditionally considered to have the highest risk of death.13, 10 Previous single‐center studies suggest that increased mortality rates are present even among patients transferred within 24 hours of hospital admission, and reinforce the notion that earlier recognition of critical illness may result in improved outcomes.1113 However, these studies have been performed primarily in small cohorts of heterogeneous patients, and may obscure the independent effect of unplanned transfers on mortality and hamper efforts to use unplanned transfer rates as a metric of healthcare quality.1, 2, 4, 9
In this study, we evaluated early unplanned ICU transfers drawn from a cohort of 499,995 hospitalizations in an integrated healthcare delivery system. Using patient data, extracted from the automated electronic medical record, we matched unplanned transfer cases to patients directly admitted to the ICU and described the association between delayed ICU transfers and adverse outcomes.
METHODS
Setting and Participants
We performed a retrospective analysis of adult patient (age 18 years) hospitalizations at 21 Northern California Kaiser Permanente (KP) Medical Care Program hospitals between January 2007 and December 2009. This work expanded on our previous report of hospital stays from November 2006 to January 2008.4 The 21 study hospitals used the same electronic health information systems; databases captured admission, discharge, and bed history data. The use of these databases for research has been described in our previous study and other reports; hospital characteristics, unit staffing, and resource levels have also been detailed previously.4, 1417 This study was approved by the KP Institutional Review Board.
Identifying Unplanned Transfers
We evaluated patients with medical hospitalizationsdefined as those whose first hospital location was not in a surgical setting such as the operating room or post‐anesthesia recovery areawhose admission originated in the ED; patients admitted for surgery were removed because of significant differences in observed mortality (see Supporting Information Appendix Figure 1 and Appendix Table 1 in the online version of this article). Patients whose admission did not originate in the ED were excluded to eliminate confounding resulting from differences in preadmission care. We also excluded patients admitted for gynecological and pregnancy‐related care because of low hospital mortality.
Initial patient locations included the medical wards (wards); the transitional care unit (TCU); and the intensive care unit (ICU). Bed history data, based on time stamps and available for all patients, were used to track patient locations from the time of admission, defined as the first non‐ED hospital location, until discharge. Patient length of stay (LOS) was calculated at each location and for the entire hospitalization.
Transfers to the ICU after a patient's initial admission to the ward or TCU were termed unplanned (or delayed) ICU transfers; patients admitted from the ED to the ICU were termed direct ICU admit patients. Direct ICU admit patients were excluded from the unplanned transfer group even if they required a readmission to the ICU later in their hospital course. We focused on patients with unplanned ICU transfers early after hospitalization to identify those in whom prompt recognition and intervention could be effective; thus, our primary analyses were on patients with transfers within 24 hours of admission. In secondary analysis, we also evaluated patients with unplanned ICU transfers occurring within 48 hours after hospital admission.
Admission Severity of Illness
To account for severity of illness at admission, we used a predicted mortality measure developed at KP.14 This method strictly utilizes information available prior to hospital admissionincluding that from the ED; variables included age, gender, admitting diagnosis, and measures of laboratory test and comorbid disease burden. The method, derived using 259,669 KP hospitalizations, produced a c‐statistic of 0.88 for inpatient mortality; external validation, based on 188,724 hospitalizations in Ottawa, produced a c‐statistic of 0.92.14, 18
Admitting diagnoses were based on admission International Classification of Diseases, 9th revision (ICD‐9) codes, and grouped into 44 broad Primary Conditions based on pathophysiologic plausibility and mortality rates.14 The method also quantified each patient's physiologic derangement and preexisting disease burden based on automated laboratory and comorbidity measuresthe Laboratory Acute Physiology Score (LAPS) and the Comorbidity Point Score (COPS).14
In brief, the LAPS was derived from 14 possible test results obtained in the 24‐hour time period preceding hospitalization, including: anion gap; arterial pH, PaCO2, and PaO2; bicarbonate; serum levels of albumin, total bilirubin, creatinine, glucose, sodium, and troponin I; blood urea nitrogen; creatinine; hematocrit; and total white blood cell count.14 The COPS was calculated from each subject's inpatient and outpatient diagnoses, based on Diagnostic Cost Groups software,19 during the 12‐month period preceding hospitalization.14 Increasing LAPS and COPS values were associated with increases in hospital mortality; detailed information about the development, application, and validation are available in previous work.14, 18
Statistical Analysis
Evaluating excess adverse outcomes associated with unplanned transfers requires adequate control of confounding variables. Our approach to reduce confounding was multivariable case matchinga technique used for assessing treatment effects in observational data.20, 21 Patients with unplanned transfersidentified as caseswere matched with similar controls based on observed variables at the time of hospital admission.
We first matched patients with unplanned ICU transfers within 24 hours of hospital admission to direct ICU admit controls based on predicted in‐hospital mortality (to within 1%); age (by decade); gender; and admitting diagnosis. If a case was matched to multiple controls, we selected 1 control with the most similar admission characteristics (weekday or weekend admission and nursing shift). The risk of death associated with unplanned transfers was estimated using multivariable conditional logistic regression. In secondary analysis, we repeated this analysis only among case‐control pairs within the same hospital facilities.
To cross‐validate the results from multivariable matching techniques, we also performed mixed‐effects multivariable logistic regression including all early unplanned transfer patients and direct ICU admit patients, while adjusting for predicted hospital mortality, age, gender, admitting diagnosis, LAPS, COPS, weekend versus weekday admission, nursing shift, and hospital facility random effects. We repeated these same analyses where cases were defined as patients transferred to the ICU within 48 hours of hospitalization.
Unplanned Transfer Timing
Using bed history data, we identified the elapsed time from admission to unplanned transfer, and categorized patients in increments of elapsed time from admission to unplanned transfer. Time‐to‐unplanned transfer was summarized using Kaplan‐Meier curve.
All analyses were performed in Stata/IC 11.0 for Mac (StataCorp LP, College Station, TX). Continuous variables were reported as mean standard deviation (SD). Cohort comparisons were performed with analysis of variance (ANOVA). Categorical variables were summarized using frequencies and compared with chi‐squared testing. A P value <0.05 was considered statistically significant.
RESULTS
During the study period, 313,797 medical hospitalizations originated in the ED (Table 1). Overall, patients' mean age was 67 18 years; 53.7% were female. Patient characteristics differed significantly based on the need for ICU admission. For example, average LAPS was highest among patients admitted directly to the ICU and lowest among patients who never required ICU care (P < 0.01). Patients with unplanned ICU transfers during hospitalization had longer length of stay and higher hospital mortality than direct ICU admit patients (P < 0.01). Overall, more than 1 in 15 patients experienced an unplanned transfer to the ICU.
| Early Delayed ICU Transfer (by Elapsed Time Since Hospital Admission) | ||||
|---|---|---|---|---|
| Variable | Overall | Within 24 hr | Within 48 hr | Direct ICU Admit |
| ||||
| No. (%) | 313,797 | 6,369 (2.0) | 9,816 (3.1) | 29,929 (9.5) |
| Age* | 67 18 | 67 16 | 68 16 | 64 17 |
| Female* | 169,358 (53.7) | 3,125 (49.1) | 4,882 (49.7) | 14,488 (48.4) |
| Weekend admission* | 83,327 (26.6) | 1,783 (28.0) | 2,733 (27.8) | 8,152 (27.2) |
| Nursing shift at admission* | ||||
| Day (7 AM‐3 PM) | 65,303 (20.8) | 1,335 (21.0) | 2,112 (21.5) | 7,065 (23.6) |
| Evening (3 PM‐11 PM) | 155,037 (49.4) | 2,990 (47.0) | 4,691 (47.8) | 13,158 (44.0) |
| Night (11 PM‐7 AM) | 93,457 (29.8) | 2,044 (32.1) | 3,013 (30.7) | 9,706 (32.4) |
| Initial hospital location* | ||||
| Ward | 234,915 (82.8) | 5,177 (81.3) | 7,987 (81.4) | |
| Transitional care unit | 48,953 (17.2) | 1,192 (18.7) | 1,829 (18.6) | |
| LAPS* | 24 19 | 28 20 | 28 20 | 35 25 |
| COPS* | 98 67 | 105 70 | 106 70 | 99 71 |
| Length of stay (days) | 4.6 7.5 | 8.4 12.2 | 9.1 13.4 | 6.4 9.5 |
| In‐hospital mortality | 12,686 (4.0) | 800 (12.6) | 1,388 (14.1) | 3,602 (12.0) |
The majority of unplanned transfers occurred within the first 48 hours of hospitalization (57.6%, Figure 1); nearly 80% occurred within the first 4 days. The rate of unplanned transfer peaked within 24 hours of hospital admission and decreased gradually as elapsed hospital LOS increased (Figure 1). While most patients experienced a single unplanned ICU transfer, 12.7% required multiple transfers to the ICU throughout their hospitalization.

Multivariable case matching between unplanned transfer cases within 24 hours of admission and direct ICU admit controls resulted in 5839 (92%) case‐control pairs (Table 2). Matched pairs were most frequently admitted with diagnoses in Primary Condition groups that included respiratory infections and pneumonia (15.6%); angina, acute myocardial infarction (AMI), and heart failure (15.6%); or gastrointestinal bleeding (13.8%).
| ICU Cohorts (by Elapsed Time to Transfer Since Hospital Admission) | ||||
|---|---|---|---|---|
| Within 24 hr (n = 5,839) | Within 48 hr (n = 8,976) | |||
| Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | |
| ||||
| Age | 67 16 | 66 16 | 67 16 | 67 16 |
| Female | 2,868 (49.1) | 2,868 (49.1) | 4,477 (49.9) | 4,477 (49.9) |
| Admitting diagnosis | ||||
| Pneumonia | 911 (15.6) | 911 (15.6) | 1,526 (17.0) | 1,526 (17.0) |
| Heart failure or MI | 909 (15.6) | 909 (15.6) | 1,331 (14.8) | 1,331 (14.8) |
| Gastrointestinal bleeding | 806 (13.8) | 806 (13.8) | 1,191 (13.3) | 1,191 (13.3) |
| Infections (including sepsis) | 295 (5.1) | 295 (5.1) | 474 (5.3) | 474 (5.3) |
| Outcomes | ||||
| Length of stay (days)* | 8 12 | 6 9 | 9 13 | 6 9 |
| In‐hospital mortality* | 678 (11.6) | 498 (8.5) | 1,181 (13.2) | 814 (9.1) |
In‐hospital mortality was significantly higher among cases (11.6%) than among ICU controls (8.5%, P < 0.001); mean LOS was also longer among cases (8 12 days) than among controls (6 9 days, P < 0.001). Unplanned transfer cases were at an increased odds of death when compared with ICU controls (adjusted odds ratio [OR], 1.44; 95% confidence interval [CI], 1.26‐1.64; P < 0.001); they also had a significantly higher observed‐to‐expected mortality ratio. When cases and controls were matched by hospital facility, the number of case‐control pairs decreased (2949 pairs; 42% matching frequency) but the odds of death was of similar magnitude (OR, 1.43; 95% CI, 1.21‐1.68; P < 0.001). Multivariable mixed‐effects logistic regression including all early unplanned transfer and direct ICU admit patients produced an effect size of similar magnitude (OR, 1.37; 95% CI, 1.24‐1.50; P < 0.001).
Results were similar when cases were limited to patients with transfers within 12 hours of admission; mortality was 10.9% among cases and 9.1% among controls (P = 0.02). When including patients with unplanned transfers within 48 hours of hospital admission, the difference in mortality between cases and controls increased (13.2% vs 9.1%, P < 0.001). The odds of death among patients with unplanned transfers increased as the elapsed time between admission and ICU transfer lengthened (Figure 2); the adjusted OR was statistically significant at each point between 8 and 48 hours.

When stratified by admitting diagnosis groups, cases with unplanned transfers within the first 48 hours had increased mortality compared with matched controls in some categories (Table 3). For example, for patients in the respiratory infection and pneumonia group, mortality was 16.8% among unplanned transfer cases and 13.0% among early matched ICU controls (P < 0.01). A similar pattern was present in groups including: gastrointestinal bleeding, chronic obstructive pulmonary disease (COPD) exacerbation, and seizure groups (Table 3). However, for patients with AMI alone, mortality was 5.0% among cases and 3.7% among matched controls (P = 0.12). Patients with sepsis had a mortality rate of 15.2% among cases and 20.8% among matched controls (P = 0.07). Similarly, patients with stroke had a mortality rate of 12.4% among unplanned transfer cases and 11.4% in the matched controls (P = 0.54).
| Primary Condition Group | Mortality in ICU Case‐Control Cohorts, No. (%) | |||
|---|---|---|---|---|
| Within 24 hr | Within 48 hr | |||
| Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | |
| ||||
| Respiratory infections | 143 (15.7) | 126 (13.8) | 493 (16.8) | 380 (13.0) |
| Angina, heart failure, or MI | 60 (6.6) | 41 (4.5) | 324 (7.7) | 152 (3.6) |
| Acute MI alone | 16 (5.7) | 17 (6.1) | 82 (5.0) | 61 (3.7) |
| Gastrointestinal bleeding | 96 (11.9) | 55 (6.8) | 549 (19.3) | 188 (6.6) |
| Infections including sepsis | 20 (9.8) | 52 (11.2) | 228 (14.8) | 220 (14.2) |
| Sepsis alone | 32 (18.9) | 31 (18.3) | 123 (15.2) | 168 (20.8) |
| COPD exacerbation | 20 (9.8) | 12 (5.9) | 74 (10.8) | 43 (6.3) |
| Stroke | 18 (10.2) | 19 (10.8) | 77 (12.4) | 71 (11.4) |
| Seizure | 21 (8.6) | 9 (3.7) | 68 (7.1) | 34 (3.6) |
DISCUSSION
This study found that unplanned ICU transfers were common among medical patients, occurring in 5% of all hospitalizations originating in the ED. The majority of unplanned transfers occurred within 48 hours of admission; the rate of ICU transfers peaked within 24 hours after hospitalization. Compared with patients admitted directly from the ED to the ICU, those transferred early after admission had significantly increased mortality; for example, patients transferred within 24 hours were at a 44% increased odds of hospital death. The adverse outcomes associated with unplanned transfers varied considerably by admission diagnosis subgroups.
Our findings confirm previous reports of increased mortality among patients with unplanned ICU transfers. Escarce and Kelley reported that patients admitted to the ICU from non‐ED locationsincluding wards, intermediate care units, and other hospitalswere at an increased risk of hospital death.1 Multiple subsequent studies have confirmed the increased mortality among patients with unplanned transfers.24, 10, 13, 22, 23 We previously evaluated patients who required a transfer to any higher level of care and reported an observed‐to‐expected mortality ratio of 2.93.4
Fewer studies, however, have evaluated the association between the timing of unplanned transfers and inpatient outcomes; previous small reports suggest that delays in ICU transfer adversely affect mortality and length of stay.12, 13, 24 Parkhe et al. compared 99 direct ICU admit patients with 23 who experienced early unplanned transfers; mortality at 30 days was significantly higher among patients with unplanned transfers.13 The current multifacility study included considerably more patients and confirmed an in‐hospital mortality gapalbeit a smaller onebetween patients with early transfers and those directly admitted to the ICU.
We focused on unplanned transfers during the earliest phase of hospitalization to identify patients who might benefit from improved recognition of, and intervention for, impending critical illness. We found that even patients requiring transfers within 8 hours of hospital admission were at an increased risk of death. Bapoje et al. recently reported that as many as 80% of early unplanned transfers were preventable and that most resulted from inappropriate admission triage.11 Together, these findings suggest that heightened attention to identifying such patients at admission or within the first day of hospitalizationwhen the rates of unplanned transfers peakis critical.
Several important limitations should be recognized in interpreting these results. First, this study was not designed to specifically identify the reasons for unplanned transfers, limiting our ability to characterize episodes in which timely care could have prevented excess mortality. Notably, while previous work suggests that many early unplanned transfers might be prevented with appropriate triage, it is likely that some excess deaths are not preventable even if every patient could be admitted to the ICU directly.
We were able to characterize patient outcomes by admitting diagnoses. Patients admitted for pneumonia and respiratory infection, gastrointestinal bleeding, COPD exacerbation, or seizures demonstrated excess mortality compared with matched ICU controls, while those with AMI, sepsis, and stroke did not. It is possible that differences in diagnosis‐specific excess mortality resulted from increasing adherence to well‐defined practice guidelines for specific high‐risk conditions.2527 For example, international awareness campaigns for the treatment of sepsis, AMI, and strokeSurviving Sepsis, Door‐to‐Balloon, and F.A.S.T.emphasize early interventions to minimize morbidity and mortality.
Second, the data utilized in this study were based on automated variables extracted from the electronic medical record. Mortality prediction models based on automated variables have demonstrated excellent performance among ICU and non‐ICU populations14, 18, 28; however, the inclusion of additional data (eg, vital signs or neurological status) would likely improve baseline risk adjustment.5, 10, 2931 Multiple studies have demonstrated that vital signs and clinician judgment can predict patients at an increased risk of deterioration.5, 10, 2931 Such data might also provide insight into residual factors that influenced clinicians' decisions to triage patients to an ICU versus non‐ICU admissiona focus area of our ongoing research efforts. Utilizing electronically available data, however, facilitated the identification of a cohort of patients far larger than that in prior studies. Where previous work has also been limited by substantial variability in baseline characteristics among study subjects,1, 2, 12, 13 our large sample produced a high percentage of multivariable case matches.
Third, we chose to match patients with a severity of illness index based on variables available at the time of hospital admission. While this mortality prediction model has demonstrated excellent performance in internal and external populations,14, 18 it is calibrated for general inpatient, rather than critically ill, populations. It remains possible that case matching with ICU‐specific severity of illness scores might alter matching characteristics, however, previous studies suggest that severity of illness, as measured by these scores, is comparable between direct ICU admits and early ICU transfers.13 Importantly, our matching procedure avoided the potential confounding known to exist with the use of prediction models based on discharge or intra‐hospitalization data.32, 33
Finally, while we were able to evaluate unplanned transfer timing in a multifacility sample, all patient care occurred within a large integrated healthcare delivery system. The overall observed mortality in our study was lower than that reported in prior studies which considered more limited patient cohorts.1, 2, 12, 13, 22 Thus, differences in patient case‐mix or ICU structure must be considered when applying our results to other healthcare delivery systems.
This hypothesis‐generating study, based on a large, multifacility sample of hospitalizations, suggests several areas of future investigation. Future work should detail specific aspects of care among patients with unplanned transfer, including: evaluating the structures and processes involved in triage decisions, measuring the effects on mortality through implementation of interventions (eg, rapid response teams or diagnosis‐specific treatment protocols), and defining the causes and risk factors for unplanned transfers by elapsed time.
In conclusion, the risk of an unplanned ICU transfera common event among hospitalized patientsis highest within 24 hours of hospitalization. Patients with early unplanned transfers have increased mortality and length of stay compared to those admitted directly to the ICU. Even patients transferred to the ICU within 8 hours of hospital admission are at an increased risk of death when compared with those admitted directly. Substantial variability in unplanned transfer outcomes exists based on admitting diagnoses. Future research should characterize unplanned transfers in greater detail with the goal of identifying patients that would benefit from improved triage and early ICU transfer.
Hospitalized patients who require transfer from medical wards to the intensive care unit (ICU) have high in‐hospital mortality, in some reports exceeding 55%.14 In a previous report in this journal, we found that while these unplanned ICU transfers occurred in only 4% of hospitalizations, they were present in nearly one‐quarter of fatal hospitalizations and were associated with substantial increases in resource utilization.4 For these reasons, interventions aimed at identifying and treating this high‐risk group have received considerable attention and have been proposed as measures of inpatient safety.2, 49
Notably, mortality among patients with unplanned ICU transfers exceeds mortality among patients admitted to the ICU directly from the emergency department (ED)a group traditionally considered to have the highest risk of death.13, 10 Previous single‐center studies suggest that increased mortality rates are present even among patients transferred within 24 hours of hospital admission, and reinforce the notion that earlier recognition of critical illness may result in improved outcomes.1113 However, these studies have been performed primarily in small cohorts of heterogeneous patients, and may obscure the independent effect of unplanned transfers on mortality and hamper efforts to use unplanned transfer rates as a metric of healthcare quality.1, 2, 4, 9
In this study, we evaluated early unplanned ICU transfers drawn from a cohort of 499,995 hospitalizations in an integrated healthcare delivery system. Using patient data, extracted from the automated electronic medical record, we matched unplanned transfer cases to patients directly admitted to the ICU and described the association between delayed ICU transfers and adverse outcomes.
METHODS
Setting and Participants
We performed a retrospective analysis of adult patient (age 18 years) hospitalizations at 21 Northern California Kaiser Permanente (KP) Medical Care Program hospitals between January 2007 and December 2009. This work expanded on our previous report of hospital stays from November 2006 to January 2008.4 The 21 study hospitals used the same electronic health information systems; databases captured admission, discharge, and bed history data. The use of these databases for research has been described in our previous study and other reports; hospital characteristics, unit staffing, and resource levels have also been detailed previously.4, 1417 This study was approved by the KP Institutional Review Board.
Identifying Unplanned Transfers
We evaluated patients with medical hospitalizationsdefined as those whose first hospital location was not in a surgical setting such as the operating room or post‐anesthesia recovery areawhose admission originated in the ED; patients admitted for surgery were removed because of significant differences in observed mortality (see Supporting Information Appendix Figure 1 and Appendix Table 1 in the online version of this article). Patients whose admission did not originate in the ED were excluded to eliminate confounding resulting from differences in preadmission care. We also excluded patients admitted for gynecological and pregnancy‐related care because of low hospital mortality.
Initial patient locations included the medical wards (wards); the transitional care unit (TCU); and the intensive care unit (ICU). Bed history data, based on time stamps and available for all patients, were used to track patient locations from the time of admission, defined as the first non‐ED hospital location, until discharge. Patient length of stay (LOS) was calculated at each location and for the entire hospitalization.
Transfers to the ICU after a patient's initial admission to the ward or TCU were termed unplanned (or delayed) ICU transfers; patients admitted from the ED to the ICU were termed direct ICU admit patients. Direct ICU admit patients were excluded from the unplanned transfer group even if they required a readmission to the ICU later in their hospital course. We focused on patients with unplanned ICU transfers early after hospitalization to identify those in whom prompt recognition and intervention could be effective; thus, our primary analyses were on patients with transfers within 24 hours of admission. In secondary analysis, we also evaluated patients with unplanned ICU transfers occurring within 48 hours after hospital admission.
Admission Severity of Illness
To account for severity of illness at admission, we used a predicted mortality measure developed at KP.14 This method strictly utilizes information available prior to hospital admissionincluding that from the ED; variables included age, gender, admitting diagnosis, and measures of laboratory test and comorbid disease burden. The method, derived using 259,669 KP hospitalizations, produced a c‐statistic of 0.88 for inpatient mortality; external validation, based on 188,724 hospitalizations in Ottawa, produced a c‐statistic of 0.92.14, 18
Admitting diagnoses were based on admission International Classification of Diseases, 9th revision (ICD‐9) codes, and grouped into 44 broad Primary Conditions based on pathophysiologic plausibility and mortality rates.14 The method also quantified each patient's physiologic derangement and preexisting disease burden based on automated laboratory and comorbidity measuresthe Laboratory Acute Physiology Score (LAPS) and the Comorbidity Point Score (COPS).14
In brief, the LAPS was derived from 14 possible test results obtained in the 24‐hour time period preceding hospitalization, including: anion gap; arterial pH, PaCO2, and PaO2; bicarbonate; serum levels of albumin, total bilirubin, creatinine, glucose, sodium, and troponin I; blood urea nitrogen; creatinine; hematocrit; and total white blood cell count.14 The COPS was calculated from each subject's inpatient and outpatient diagnoses, based on Diagnostic Cost Groups software,19 during the 12‐month period preceding hospitalization.14 Increasing LAPS and COPS values were associated with increases in hospital mortality; detailed information about the development, application, and validation are available in previous work.14, 18
Statistical Analysis
Evaluating excess adverse outcomes associated with unplanned transfers requires adequate control of confounding variables. Our approach to reduce confounding was multivariable case matchinga technique used for assessing treatment effects in observational data.20, 21 Patients with unplanned transfersidentified as caseswere matched with similar controls based on observed variables at the time of hospital admission.
We first matched patients with unplanned ICU transfers within 24 hours of hospital admission to direct ICU admit controls based on predicted in‐hospital mortality (to within 1%); age (by decade); gender; and admitting diagnosis. If a case was matched to multiple controls, we selected 1 control with the most similar admission characteristics (weekday or weekend admission and nursing shift). The risk of death associated with unplanned transfers was estimated using multivariable conditional logistic regression. In secondary analysis, we repeated this analysis only among case‐control pairs within the same hospital facilities.
To cross‐validate the results from multivariable matching techniques, we also performed mixed‐effects multivariable logistic regression including all early unplanned transfer patients and direct ICU admit patients, while adjusting for predicted hospital mortality, age, gender, admitting diagnosis, LAPS, COPS, weekend versus weekday admission, nursing shift, and hospital facility random effects. We repeated these same analyses where cases were defined as patients transferred to the ICU within 48 hours of hospitalization.
Unplanned Transfer Timing
Using bed history data, we identified the elapsed time from admission to unplanned transfer, and categorized patients in increments of elapsed time from admission to unplanned transfer. Time‐to‐unplanned transfer was summarized using Kaplan‐Meier curve.
All analyses were performed in Stata/IC 11.0 for Mac (StataCorp LP, College Station, TX). Continuous variables were reported as mean standard deviation (SD). Cohort comparisons were performed with analysis of variance (ANOVA). Categorical variables were summarized using frequencies and compared with chi‐squared testing. A P value <0.05 was considered statistically significant.
RESULTS
During the study period, 313,797 medical hospitalizations originated in the ED (Table 1). Overall, patients' mean age was 67 18 years; 53.7% were female. Patient characteristics differed significantly based on the need for ICU admission. For example, average LAPS was highest among patients admitted directly to the ICU and lowest among patients who never required ICU care (P < 0.01). Patients with unplanned ICU transfers during hospitalization had longer length of stay and higher hospital mortality than direct ICU admit patients (P < 0.01). Overall, more than 1 in 15 patients experienced an unplanned transfer to the ICU.
| Early Delayed ICU Transfer (by Elapsed Time Since Hospital Admission) | ||||
|---|---|---|---|---|
| Variable | Overall | Within 24 hr | Within 48 hr | Direct ICU Admit |
| ||||
| No. (%) | 313,797 | 6,369 (2.0) | 9,816 (3.1) | 29,929 (9.5) |
| Age* | 67 18 | 67 16 | 68 16 | 64 17 |
| Female* | 169,358 (53.7) | 3,125 (49.1) | 4,882 (49.7) | 14,488 (48.4) |
| Weekend admission* | 83,327 (26.6) | 1,783 (28.0) | 2,733 (27.8) | 8,152 (27.2) |
| Nursing shift at admission* | ||||
| Day (7 AM‐3 PM) | 65,303 (20.8) | 1,335 (21.0) | 2,112 (21.5) | 7,065 (23.6) |
| Evening (3 PM‐11 PM) | 155,037 (49.4) | 2,990 (47.0) | 4,691 (47.8) | 13,158 (44.0) |
| Night (11 PM‐7 AM) | 93,457 (29.8) | 2,044 (32.1) | 3,013 (30.7) | 9,706 (32.4) |
| Initial hospital location* | ||||
| Ward | 234,915 (82.8) | 5,177 (81.3) | 7,987 (81.4) | |
| Transitional care unit | 48,953 (17.2) | 1,192 (18.7) | 1,829 (18.6) | |
| LAPS* | 24 19 | 28 20 | 28 20 | 35 25 |
| COPS* | 98 67 | 105 70 | 106 70 | 99 71 |
| Length of stay (days) | 4.6 7.5 | 8.4 12.2 | 9.1 13.4 | 6.4 9.5 |
| In‐hospital mortality | 12,686 (4.0) | 800 (12.6) | 1,388 (14.1) | 3,602 (12.0) |
The majority of unplanned transfers occurred within the first 48 hours of hospitalization (57.6%, Figure 1); nearly 80% occurred within the first 4 days. The rate of unplanned transfer peaked within 24 hours of hospital admission and decreased gradually as elapsed hospital LOS increased (Figure 1). While most patients experienced a single unplanned ICU transfer, 12.7% required multiple transfers to the ICU throughout their hospitalization.

Multivariable case matching between unplanned transfer cases within 24 hours of admission and direct ICU admit controls resulted in 5839 (92%) case‐control pairs (Table 2). Matched pairs were most frequently admitted with diagnoses in Primary Condition groups that included respiratory infections and pneumonia (15.6%); angina, acute myocardial infarction (AMI), and heart failure (15.6%); or gastrointestinal bleeding (13.8%).
| ICU Cohorts (by Elapsed Time to Transfer Since Hospital Admission) | ||||
|---|---|---|---|---|
| Within 24 hr (n = 5,839) | Within 48 hr (n = 8,976) | |||
| Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | |
| ||||
| Age | 67 16 | 66 16 | 67 16 | 67 16 |
| Female | 2,868 (49.1) | 2,868 (49.1) | 4,477 (49.9) | 4,477 (49.9) |
| Admitting diagnosis | ||||
| Pneumonia | 911 (15.6) | 911 (15.6) | 1,526 (17.0) | 1,526 (17.0) |
| Heart failure or MI | 909 (15.6) | 909 (15.6) | 1,331 (14.8) | 1,331 (14.8) |
| Gastrointestinal bleeding | 806 (13.8) | 806 (13.8) | 1,191 (13.3) | 1,191 (13.3) |
| Infections (including sepsis) | 295 (5.1) | 295 (5.1) | 474 (5.3) | 474 (5.3) |
| Outcomes | ||||
| Length of stay (days)* | 8 12 | 6 9 | 9 13 | 6 9 |
| In‐hospital mortality* | 678 (11.6) | 498 (8.5) | 1,181 (13.2) | 814 (9.1) |
In‐hospital mortality was significantly higher among cases (11.6%) than among ICU controls (8.5%, P < 0.001); mean LOS was also longer among cases (8 12 days) than among controls (6 9 days, P < 0.001). Unplanned transfer cases were at an increased odds of death when compared with ICU controls (adjusted odds ratio [OR], 1.44; 95% confidence interval [CI], 1.26‐1.64; P < 0.001); they also had a significantly higher observed‐to‐expected mortality ratio. When cases and controls were matched by hospital facility, the number of case‐control pairs decreased (2949 pairs; 42% matching frequency) but the odds of death was of similar magnitude (OR, 1.43; 95% CI, 1.21‐1.68; P < 0.001). Multivariable mixed‐effects logistic regression including all early unplanned transfer and direct ICU admit patients produced an effect size of similar magnitude (OR, 1.37; 95% CI, 1.24‐1.50; P < 0.001).
Results were similar when cases were limited to patients with transfers within 12 hours of admission; mortality was 10.9% among cases and 9.1% among controls (P = 0.02). When including patients with unplanned transfers within 48 hours of hospital admission, the difference in mortality between cases and controls increased (13.2% vs 9.1%, P < 0.001). The odds of death among patients with unplanned transfers increased as the elapsed time between admission and ICU transfer lengthened (Figure 2); the adjusted OR was statistically significant at each point between 8 and 48 hours.

When stratified by admitting diagnosis groups, cases with unplanned transfers within the first 48 hours had increased mortality compared with matched controls in some categories (Table 3). For example, for patients in the respiratory infection and pneumonia group, mortality was 16.8% among unplanned transfer cases and 13.0% among early matched ICU controls (P < 0.01). A similar pattern was present in groups including: gastrointestinal bleeding, chronic obstructive pulmonary disease (COPD) exacerbation, and seizure groups (Table 3). However, for patients with AMI alone, mortality was 5.0% among cases and 3.7% among matched controls (P = 0.12). Patients with sepsis had a mortality rate of 15.2% among cases and 20.8% among matched controls (P = 0.07). Similarly, patients with stroke had a mortality rate of 12.4% among unplanned transfer cases and 11.4% in the matched controls (P = 0.54).
| Primary Condition Group | Mortality in ICU Case‐Control Cohorts, No. (%) | |||
|---|---|---|---|---|
| Within 24 hr | Within 48 hr | |||
| Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | Delayed ICU Transfer (Case) | Direct ICU Admit (Control) | |
| ||||
| Respiratory infections | 143 (15.7) | 126 (13.8) | 493 (16.8) | 380 (13.0) |
| Angina, heart failure, or MI | 60 (6.6) | 41 (4.5) | 324 (7.7) | 152 (3.6) |
| Acute MI alone | 16 (5.7) | 17 (6.1) | 82 (5.0) | 61 (3.7) |
| Gastrointestinal bleeding | 96 (11.9) | 55 (6.8) | 549 (19.3) | 188 (6.6) |
| Infections including sepsis | 20 (9.8) | 52 (11.2) | 228 (14.8) | 220 (14.2) |
| Sepsis alone | 32 (18.9) | 31 (18.3) | 123 (15.2) | 168 (20.8) |
| COPD exacerbation | 20 (9.8) | 12 (5.9) | 74 (10.8) | 43 (6.3) |
| Stroke | 18 (10.2) | 19 (10.8) | 77 (12.4) | 71 (11.4) |
| Seizure | 21 (8.6) | 9 (3.7) | 68 (7.1) | 34 (3.6) |
DISCUSSION
This study found that unplanned ICU transfers were common among medical patients, occurring in 5% of all hospitalizations originating in the ED. The majority of unplanned transfers occurred within 48 hours of admission; the rate of ICU transfers peaked within 24 hours after hospitalization. Compared with patients admitted directly from the ED to the ICU, those transferred early after admission had significantly increased mortality; for example, patients transferred within 24 hours were at a 44% increased odds of hospital death. The adverse outcomes associated with unplanned transfers varied considerably by admission diagnosis subgroups.
Our findings confirm previous reports of increased mortality among patients with unplanned ICU transfers. Escarce and Kelley reported that patients admitted to the ICU from non‐ED locationsincluding wards, intermediate care units, and other hospitalswere at an increased risk of hospital death.1 Multiple subsequent studies have confirmed the increased mortality among patients with unplanned transfers.24, 10, 13, 22, 23 We previously evaluated patients who required a transfer to any higher level of care and reported an observed‐to‐expected mortality ratio of 2.93.4
Fewer studies, however, have evaluated the association between the timing of unplanned transfers and inpatient outcomes; previous small reports suggest that delays in ICU transfer adversely affect mortality and length of stay.12, 13, 24 Parkhe et al. compared 99 direct ICU admit patients with 23 who experienced early unplanned transfers; mortality at 30 days was significantly higher among patients with unplanned transfers.13 The current multifacility study included considerably more patients and confirmed an in‐hospital mortality gapalbeit a smaller onebetween patients with early transfers and those directly admitted to the ICU.
We focused on unplanned transfers during the earliest phase of hospitalization to identify patients who might benefit from improved recognition of, and intervention for, impending critical illness. We found that even patients requiring transfers within 8 hours of hospital admission were at an increased risk of death. Bapoje et al. recently reported that as many as 80% of early unplanned transfers were preventable and that most resulted from inappropriate admission triage.11 Together, these findings suggest that heightened attention to identifying such patients at admission or within the first day of hospitalizationwhen the rates of unplanned transfers peakis critical.
Several important limitations should be recognized in interpreting these results. First, this study was not designed to specifically identify the reasons for unplanned transfers, limiting our ability to characterize episodes in which timely care could have prevented excess mortality. Notably, while previous work suggests that many early unplanned transfers might be prevented with appropriate triage, it is likely that some excess deaths are not preventable even if every patient could be admitted to the ICU directly.
We were able to characterize patient outcomes by admitting diagnoses. Patients admitted for pneumonia and respiratory infection, gastrointestinal bleeding, COPD exacerbation, or seizures demonstrated excess mortality compared with matched ICU controls, while those with AMI, sepsis, and stroke did not. It is possible that differences in diagnosis‐specific excess mortality resulted from increasing adherence to well‐defined practice guidelines for specific high‐risk conditions.2527 For example, international awareness campaigns for the treatment of sepsis, AMI, and strokeSurviving Sepsis, Door‐to‐Balloon, and F.A.S.T.emphasize early interventions to minimize morbidity and mortality.
Second, the data utilized in this study were based on automated variables extracted from the electronic medical record. Mortality prediction models based on automated variables have demonstrated excellent performance among ICU and non‐ICU populations14, 18, 28; however, the inclusion of additional data (eg, vital signs or neurological status) would likely improve baseline risk adjustment.5, 10, 2931 Multiple studies have demonstrated that vital signs and clinician judgment can predict patients at an increased risk of deterioration.5, 10, 2931 Such data might also provide insight into residual factors that influenced clinicians' decisions to triage patients to an ICU versus non‐ICU admissiona focus area of our ongoing research efforts. Utilizing electronically available data, however, facilitated the identification of a cohort of patients far larger than that in prior studies. Where previous work has also been limited by substantial variability in baseline characteristics among study subjects,1, 2, 12, 13 our large sample produced a high percentage of multivariable case matches.
Third, we chose to match patients with a severity of illness index based on variables available at the time of hospital admission. While this mortality prediction model has demonstrated excellent performance in internal and external populations,14, 18 it is calibrated for general inpatient, rather than critically ill, populations. It remains possible that case matching with ICU‐specific severity of illness scores might alter matching characteristics, however, previous studies suggest that severity of illness, as measured by these scores, is comparable between direct ICU admits and early ICU transfers.13 Importantly, our matching procedure avoided the potential confounding known to exist with the use of prediction models based on discharge or intra‐hospitalization data.32, 33
Finally, while we were able to evaluate unplanned transfer timing in a multifacility sample, all patient care occurred within a large integrated healthcare delivery system. The overall observed mortality in our study was lower than that reported in prior studies which considered more limited patient cohorts.1, 2, 12, 13, 22 Thus, differences in patient case‐mix or ICU structure must be considered when applying our results to other healthcare delivery systems.
This hypothesis‐generating study, based on a large, multifacility sample of hospitalizations, suggests several areas of future investigation. Future work should detail specific aspects of care among patients with unplanned transfer, including: evaluating the structures and processes involved in triage decisions, measuring the effects on mortality through implementation of interventions (eg, rapid response teams or diagnosis‐specific treatment protocols), and defining the causes and risk factors for unplanned transfers by elapsed time.
In conclusion, the risk of an unplanned ICU transfera common event among hospitalized patientsis highest within 24 hours of hospitalization. Patients with early unplanned transfers have increased mortality and length of stay compared to those admitted directly to the ICU. Even patients transferred to the ICU within 8 hours of hospital admission are at an increased risk of death when compared with those admitted directly. Substantial variability in unplanned transfer outcomes exists based on admitting diagnoses. Future research should characterize unplanned transfers in greater detail with the goal of identifying patients that would benefit from improved triage and early ICU transfer.
- ,.Admission source to the medical intensive care unit predicts hospital death independent of APACHE II score.JAMA.1990;264(18):2389–2394.
- ,,,,,.Unplanned admission to intensive care after emergency hospitalisation: risk factors and development of a nomogram for individualising risk.Resuscitation.2009;80(2):224–230.
- ,.Outcome of intensive care patients in a group of British intensive care units.Crit Care Med.1998;26(8):1337–1345.
- ,,,,,.Intra‐hospital transfers to a higher level of care: contribution to total hospital and intensive care unit (ICU) mortality and length of stay (LOS).J Hosp Med.2010;6(2):74–80.
- ,.Medical patients at high risk for catastrophic deterioration.Crit Care Med.1987;15(5):510–515.
- ,,, et al.Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial.Lancet.2005;365(9477):2091–2097.
- ,,, et al.Effect of a rapid response team on hospital‐wide mortality and code rates outside the ICU in a children's hospital.JAMA.2007;298(19):2267–2274.
- ,,,,,.Validity of unplanned admission to an intensive care unit as a measure of patient safety in surgical patients.Anesthesiology.2005;103(6):1121–1129.
- ,,,.The 100,000 lives campaign: setting a goal and a deadline for improving health care quality.JAMA.2006;295(3):324–327.
- ,,, et al.Duration of life‐threatening antecedents prior to intensive care admission.Intensive Care Med.2002;28(11):1629–1634.
- ,,,.Unplanned transfers to a medical intensive care unit: causes and relationship to preventable errors in care.J Hosp Med.2011;6(2):68–72.
- ,,,,.Inpatient transfers to the intensive care unit: delays are associated with increased mortality and morbidity.J Gen Intern Med.2003;18(2):77–83.
- ,,,.Outcome of emergency department patients with delayed admission to an intensive care unit.Emerg Med (Fremantle).2002;14(1):50–57.
- ,,,,,.Risk‐adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases.Med Care.2008;46(3):232–239.
- ,,, et al.Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases.Am J Manag Care.2008;14(3):158–166.
- .Linking automated databases for research in managed care settings.Ann Intern Med.1997;127(8 pt 2):719–724.
- ,,, et al.Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice?JAMA.2003;290(20):2685–2692.
- ,,,.The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population.J Clin Epidemiol.2009;63(7):798–803.
- ,.Refinements to the diagnostic cost group (DCG) model.Inquiry.1995;32(4):418–429.
- ,.Excess length of stay, charges, and mortality attributable to medical injuries during hospitalization.JAMA.2003;290(14):1868–1874.
- .Optimal matching in observational studies.J Am Stat Assoc.1989;84:1024–1032.
- ,,,.Admissions to intensive care units from emergency departments: a descriptive study.Emerg Med J.2005;22(6):423–428.
- ,,,.Using administrative data to develop a nomogram for individualising risk of unplanned admission to intensive care.Resuscitation.2008;79(2):241–248.
- ,,,.Unplanned intensive care unit transfers: a useful tool to improve quality of care [abstract]. In: Hospital Medicine 2010 abstract booklet. Society of Hospital Medicine 2010 Annual Meeting, April 9–11, 2010, Washington, DC;2010:10–11.
- ,,, et al.Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008.Crit Care Med.2008;36(1):296–327.
- ,,, et al.2009 Focused Updates: ACC/AHA Guidelines for the Management of Patients With ST‐Elevation Myocardial Infarction (updating the 2004 Guideline and 2007 Focused Update) and ACC/AHA/SCAI Guidelines on Percutaneous Coronary Intervention (updating the 2005 Guideline and 2007 Focused Update): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.Circulation.2009;120(22):2271–2306.
- ,,, et al.Translating evidence into practice: a decade of efforts by the American Heart Association/American Stroke Association to reduce death and disability due to stroke: a presidential advisory from the American Heart Association/American Stroke Association.Stroke.2010;41(5):1051–1065.
- ,,, et al.Veterans Affairs intensive care unit risk adjustment model: validation, updating, recalibration.Crit Care Med.2008;36(4):1031–1042.
- ,,, et al.Recommended guidelines for monitoring, reporting, and conducting research on medical emergency team, outreach, and rapid response systems: an Utstein‐style scientific statement: a scientific statement from the International Liaison Committee on Resuscitation (American Heart Association, Australian Resuscitation Council, European Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Council of Southern Africa, and the New Zealand Resuscitation Council); the American Heart Association Emergency Cardiovascular Care Committee; the Council on Cardiopulmonary, Perioperative, and Critical Care; and the Interdisciplinary Working Group on Quality of Care and Outcomes Research.Circulation.2007;116(21):2481–2500.
- ,,,,,.Realizing the potential of clinical judgment: a real‐time strategy for predicting outcomes and cost for medical inpatients.Am J Med.2000;109(3):189–195.
- ,,.Physiological values and procedures in the 24 h before ICU admission from the ward.Anaesthesia.1999;54(6):529–534.
- ,,,,,.Predicting who dies depends on how severity is measured: implications for evaluating patient outcomes.Ann Intern Med.1995;123(10):763–770.
- ,,, et al.Enhancement of claims data to improve risk adjustment of hospital mortality.JAMA.2007;297(1):71–76.
- ,.Admission source to the medical intensive care unit predicts hospital death independent of APACHE II score.JAMA.1990;264(18):2389–2394.
- ,,,,,.Unplanned admission to intensive care after emergency hospitalisation: risk factors and development of a nomogram for individualising risk.Resuscitation.2009;80(2):224–230.
- ,.Outcome of intensive care patients in a group of British intensive care units.Crit Care Med.1998;26(8):1337–1345.
- ,,,,,.Intra‐hospital transfers to a higher level of care: contribution to total hospital and intensive care unit (ICU) mortality and length of stay (LOS).J Hosp Med.2010;6(2):74–80.
- ,.Medical patients at high risk for catastrophic deterioration.Crit Care Med.1987;15(5):510–515.
- ,,, et al.Introduction of the medical emergency team (MET) system: a cluster‐randomised controlled trial.Lancet.2005;365(9477):2091–2097.
- ,,, et al.Effect of a rapid response team on hospital‐wide mortality and code rates outside the ICU in a children's hospital.JAMA.2007;298(19):2267–2274.
- ,,,,,.Validity of unplanned admission to an intensive care unit as a measure of patient safety in surgical patients.Anesthesiology.2005;103(6):1121–1129.
- ,,,.The 100,000 lives campaign: setting a goal and a deadline for improving health care quality.JAMA.2006;295(3):324–327.
- ,,, et al.Duration of life‐threatening antecedents prior to intensive care admission.Intensive Care Med.2002;28(11):1629–1634.
- ,,,.Unplanned transfers to a medical intensive care unit: causes and relationship to preventable errors in care.J Hosp Med.2011;6(2):68–72.
- ,,,,.Inpatient transfers to the intensive care unit: delays are associated with increased mortality and morbidity.J Gen Intern Med.2003;18(2):77–83.
- ,,,.Outcome of emergency department patients with delayed admission to an intensive care unit.Emerg Med (Fremantle).2002;14(1):50–57.
- ,,,,,.Risk‐adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases.Med Care.2008;46(3):232–239.
- ,,, et al.Risk adjusting community‐acquired pneumonia hospital outcomes using automated databases.Am J Manag Care.2008;14(3):158–166.
- .Linking automated databases for research in managed care settings.Ann Intern Med.1997;127(8 pt 2):719–724.
- ,,, et al.Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice?JAMA.2003;290(20):2685–2692.
- ,,,.The Kaiser Permanente inpatient risk adjustment methodology was valid in an external patient population.J Clin Epidemiol.2009;63(7):798–803.
- ,.Refinements to the diagnostic cost group (DCG) model.Inquiry.1995;32(4):418–429.
- ,.Excess length of stay, charges, and mortality attributable to medical injuries during hospitalization.JAMA.2003;290(14):1868–1874.
- .Optimal matching in observational studies.J Am Stat Assoc.1989;84:1024–1032.
- ,,,.Admissions to intensive care units from emergency departments: a descriptive study.Emerg Med J.2005;22(6):423–428.
- ,,,.Using administrative data to develop a nomogram for individualising risk of unplanned admission to intensive care.Resuscitation.2008;79(2):241–248.
- ,,,.Unplanned intensive care unit transfers: a useful tool to improve quality of care [abstract]. In: Hospital Medicine 2010 abstract booklet. Society of Hospital Medicine 2010 Annual Meeting, April 9–11, 2010, Washington, DC;2010:10–11.
- ,,, et al.Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008.Crit Care Med.2008;36(1):296–327.
- ,,, et al.2009 Focused Updates: ACC/AHA Guidelines for the Management of Patients With ST‐Elevation Myocardial Infarction (updating the 2004 Guideline and 2007 Focused Update) and ACC/AHA/SCAI Guidelines on Percutaneous Coronary Intervention (updating the 2005 Guideline and 2007 Focused Update): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.Circulation.2009;120(22):2271–2306.
- ,,, et al.Translating evidence into practice: a decade of efforts by the American Heart Association/American Stroke Association to reduce death and disability due to stroke: a presidential advisory from the American Heart Association/American Stroke Association.Stroke.2010;41(5):1051–1065.
- ,,, et al.Veterans Affairs intensive care unit risk adjustment model: validation, updating, recalibration.Crit Care Med.2008;36(4):1031–1042.
- ,,, et al.Recommended guidelines for monitoring, reporting, and conducting research on medical emergency team, outreach, and rapid response systems: an Utstein‐style scientific statement: a scientific statement from the International Liaison Committee on Resuscitation (American Heart Association, Australian Resuscitation Council, European Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Council of Southern Africa, and the New Zealand Resuscitation Council); the American Heart Association Emergency Cardiovascular Care Committee; the Council on Cardiopulmonary, Perioperative, and Critical Care; and the Interdisciplinary Working Group on Quality of Care and Outcomes Research.Circulation.2007;116(21):2481–2500.
- ,,,,,.Realizing the potential of clinical judgment: a real‐time strategy for predicting outcomes and cost for medical inpatients.Am J Med.2000;109(3):189–195.
- ,,.Physiological values and procedures in the 24 h before ICU admission from the ward.Anaesthesia.1999;54(6):529–534.
- ,,,,,.Predicting who dies depends on how severity is measured: implications for evaluating patient outcomes.Ann Intern Med.1995;123(10):763–770.
- ,,, et al.Enhancement of claims data to improve risk adjustment of hospital mortality.JAMA.2007;297(1):71–76.
Copyright © 2011 Society of Hospital Medicine
Severe AH Among Inpatients From the ED
Chronic hypertension affects 32% of adults in the United States.1 Each year, over 20 million emergency department visits involve hypertension.2 While many studies describe the epidemiology and outcomes of chronic hypertension, less is known about severe acute hypertension (AH). Often classified as either hypertensive urgency or emergency, it receives little attention in national treatment guidelines.3 There is a limited understanding of the epidemiology of, and the relationship between, this syndrome and patient outcomes among hospitalized patients. One registry study suggested that severe AH was associated with high rates of new organ damage, need for treatment in the intensive care unit, and a 90‐day readmission rate of 10%.4 This investigation, however, lacked generalizability, because it only enrolled subjects requiring therapy with an intravenous antihypertensive agent and did not provide information on the prevalence of severe AH. Qureshi et al.5 analyzed information in a more representative sample from the national hospital ambulatory care survey, however, the only outcome examined was the correlation between acute blood pressure and stroke. Studies focusing on a selected patient population may be of limited value to hospitalists, because they are commonly required to treat a range of patients presenting to the hospital.
In light of severe syndromes that may be associated with, or complicated by, severe AH presented early during acute care, hospitalists require a better understanding of the prevalence and the relationship between severe AH and attendant mortality and morbidity. In addition, an assessment of the association of severe AH on the need for intensive care unit (ICU) admission and mechanical ventilation (MV) may aid the initial treatment assessments and triage decisions required of hospitalists.
Our objective was to describe the prevalence and implications of severe AH present at the time of evaluation in the ED among patients eventually hospitalized, using clinical data collected on all consecutive admissions across a range of clinical conditions. We sought to determine the independent contribution, if any, of severe AH to hospital mortality, need for MV on admission, as well as hospital length of stay (LOS).
METHODS
Study Design and Setting
This was a retrospective analysis of adults admitted to 114 acute‐care hospitals in the United States from 2005 through 2007. The New England Institutional Review Board/Human Subjects Research Committee (Wellesley, MA) reviewed and approved this study. It was conducted in compliance with the Health Insurance Portability and Accountability Act (HIPAA).
Data
Data were obtained from one of the Clinical Research Databases from CareFusion (formerly Cardinal Health [CareFusion Clinical Research Services, Marlborough, MA]).613 The database contains electronically imported or manually extracted demographic, clinical (eg, comorbidities, vital signs, laboratory values, other clinical findings), and administrative data (eg, diagnosis, procedures, and length of hospitalization). All vital signs were manually extracted, including the highest and the lowest ED systolic blood pressure (SBP) measurements during the ED stay, but before inpatient admission. Patients admitted for childbirth or mental health reasons were not included.
Patients
Our main analysis focused on patients whose highest and lowest SBP were collected in the ED. These patients accounted for approximately 90% of all inpatients admitted through the ED. For the approximately 9% of patients who had only 1 SBP collected, we conducted a sensitivity analysis by both including and excluding them in the main analysis to determine if the absence of a second SBP measurement altered our findings. Patients were grouped into 1 of the following 8 mutually exclusive strata based on the maximum SBP (mmHg) in the ED: <100, 101‐139, 140‐180, 181‐190, 191‐200, 201‐210, 211‐220, and >220.
Measures
The primary exposure of interest was the prevalence of severe AH, defined as at least 1 SBP measurement recorded in the ED in excess of 180 mmHg. Outcome measures included in‐hospital mortality, need for MV on admission day (defined by International Classification of Diseases, 9th revision, Clinical Modification [ICD‐9‐CM] procedure codes of 96.70, 96.71, and 96.72), and LOS. We stratified these outcomes for each inpatient admission according to 1 of 112 mutually exclusive groups of principal diagnoses (see Supporting Appendix A in the online version of this article). To simplify the presentations, we pooled the groups into 9 major disease categories based on organ systems.
Primary Data Analysis
All statistical analyses were performed using Statistical Analysis Software (SAS version 9.01; SAS Institute Inc, Cary, NC). For evaluating trending, we used the Cochran‐Armitage test for dichotomous variables (mortality and MV), and linear regression for continuous variable (LOS). We employed a logistic regression model to estimate risk of mortality and need for MV on admission. We used linear regression models to estimate the LOS associated with severe AH. We modeled each outcome as a function of patient disease severity and SBP strata. Because patients with the most severe AH tended to have higher mortality early in hospitalization, our analysis of LOS was limited to patients who survived index hospitalization.
The original disease‐specific risk‐adjustment models accounting for patient‐level confounding risk factors, including demographics, physiologic presentation on admission (vital signs, altered mental status, and laboratory findings), and chronic conditions, were previously developed and validated.12, 13 We recalibrated each of the 112 models, for the current study cohort, using the logit of predicted probability of death generated from the mortality risk‐adjustment model as a propensity score of disease severity. Using this propensity score as an aggregate severity adjuster, we refit 9 logistic regression models (1 for each major disease category) to estimate the odds ratios for mortality or need for MV for each of the 7 SBP strata in the regression models with 101‐139 mmHg as the reference group. To estimate the attributable LOS (if any) of severe AH among survivors, we fit the 9 LOS models using log transformed LOS (to normalize the potentially skewed distribution of LOS) as the outcome, controlling for disease severity. The attributable LOS and 95% confidence intervals (CI) were estimated from 1000 bootstrap iterations, with the median as the parameter estimate and 2.5th and 97.5th percentile as 95% CI.14, 15
Sensitivity Analysis
To address potential bias of LOS associated with inpatient mortality, we refit 9 LOS models, including both patients who died and those who survived the index hospitalization. The models adjusted for disease severity, mortality, and the interaction of severity and mortality. Because patients with only 1 SBP recorded at ED may be different from those with more than 1 SBP recorded, we conducted analysis by adding these patients in the study cohort to examine the potential change of overall prevalence of AH and associated mortality for the study population.
To address the potential for a center‐specific effect on outcomes, we refit all the models using a mixed model approach.16 The mixed model accounts for both patient‐level risk factors and hospital‐specific effects on the observed outcomes.
RESULTS
Patient Characteristics
The study cohort was comprised of 1,290,804 adults who were admitted through the ED, from 2005 through 2007, and whose highest and lowest SBP measurements were collected in the ED. Median age was 69 years (interquartile range, 53‐81) for the overall population. Median age was 74 (interquartile range, 60‐83) for patients with severe AH (Table 1). Hospital mortality was 3.6% (n = 46,033), with 6119 (13.3%) having severe AH.
| Characteristic | Prevalence, n (Column %) | Severe Acute Hypertension, n (Row %) |
|---|---|---|
| Total number of discharges | 1,290,804 (100.0) | 178,197 (13.8) |
| Mortality | 46,033 (3.6) | 6,119 (13.3) |
| Live discharges | 1,244,771 (96.4) | 172,078 (13.8) |
| Mechanical ventilation on admission | 39,238 (3.0) | 9,508 (24.2) |
| Demographics | ||
| Age, median (1st, 3rd quartiles) | 69 (53, 81) | 74 (60, 83) |
| Male | 587,553 (45.5) | 71,085 (12.1) |
| Female | 703,244 (54.5) | 107,109 (15.2) |
| Race | ||
| White | 949,869 (73.6) | 121,930 (12.8) |
| Black | 220,601 (17.1) | 39,667 (18.0) |
| Other | 120,334 (9.3) | 16,600 (13.8) |
| Insurance | ||
| Medicare | 668,420 (51.8) | 105,078 (15.7) |
| Medicaid | 108,538 (8.4) | 12,259 (11.3) |
| Commercial | 163,858 (12.7) | 18,669 (11.4) |
| Other | 349,988 (27.1) | 42,191 (12.1) |
| Disease system by the principal diagnosis | ||
| Nervous system | 76,744 (5.9) | 22,270 (29.0) |
| Respiratory system | 222,329 (17.2) | 24,678 (11.1) |
| Circulatory system | 416,847 (32.3) | 66,852 (16.0) |
| Digestive system | 186,282 (14.4) | 17,817 (9.6) |
| Hepatobiliary/pancreas system | 52,293 (4.1) | 5,664 (10.8) |
| Endocrine system | 45,050 (3.5) | 6,625 (14.7) |
| Kidney/urinary system | 81,782 (6.3) | 11,050 (13.5) |
| Infectious diseases | 60,353 (4.7) | 4,162 (6.9) |
| Other | 149,124 (11.6) | 19,079 (12.8) |
| Comorbidity by secondary diagnoses | ||
| Hypertension | 729,417 (56.5) | 135,498 (18.6) |
| Fluid and electrolyte disorders | 306,666 (23.8) | 37,836 (12.3) |
| Diabetes without chronic complications | 286,912 (22.2) | 47,979 (16.7) |
| Chronic pulmonary disease | 283,895 (22.0) | 35,977 (12.7) |
| Congestive heart failure | 213,523 (16.5) | 33,956 (15.9) |
| Deficiency anemias | 210,230 (16.3) | 30,266 (14.4) |
| Renal failure | 159,409 (12.3) | 31,984 (20.1) |
| Hypothyroidism | 153,911 (11.9) | 22,441 (14.6) |
| Valvular disease | 140,820 (10.9) | 21,453 (15.2) |
| Depression | 137,259 (10.6) | 16,886 (12.3) |
| Other neurological disorders | 126,954 (9.8) | 19,103 (15.0) |
| Peripheral vascular disease | 88,321 (6.8) | 16,180 (18.3) |
| Obesity | 84,000 (6.5) | 12,351 (14.7) |
| Diabetes with chronic complications | 65,989 (5.1) | 13,093 (19.8) |
| Psychoses | 54,769 (4.2) | 5,555 (10.1) |
| Alcohol abuse | 51,765 (4.0) | 6,014 (11.6) |
| Pulmonary circulation disease | 49,248 (3.8) | 7,128 (14.5) |
| Coagulopathy | 43,584 (3.4) | 4,339 (10.0) |
| Paralysis | 42,128 (3.3) | 8,125 (19.3) |
| Drug abuse | 36,134 (2.8) | 4,779 (13.2) |
| Liver disease | 36,094 (2.8) | 3,218 (8.9) |
| Weight loss | 35,795 (2.8) | 3,726 (10.4) |
| Metastatic cancer | 33,517 (2.6) | 2,498 (7.5) |
| Rheumatoid arthritis | 32,545 (2.5) | 4,300 (13.2) |
| Solid tumor without metastasis | 30,677 (2.4) | 3,035 (9.9) |
| Chronic blood loss anemia | 25,416 (2.0) | 2,268 (8.9) |
| Lymphoma | 9,972 (0.8) | 871 (8.7) |
| Acquired immune deficiency syndrome | 3,048 (0.2) | 307 (10.1) |
| Peptic ulcer disease | 915 (0.1) | 131 (14.3) |
| Discharges by hospital characteristics | ||
| Teaching status | ||
| Teaching hospitals | 899,786 (69.7) | 127,512 (14.2) |
| Nonteaching hospitals | 391,018 (30.3) | 50,685 (13.0) |
| Urban status | ||
| Urban hospitals | 1,164,802 (90.2) | 162,399 (13.9) |
| Rural hospitals | 126,002 (9.8) | 15,798 (12.5) |
| Bed size | ||
| Beds <100 | 36,624 (2.8) | 4,965 (13.6) |
| Beds 100‐300 | 623,327 (48.3) | 80,156 (12.9) |
| Beds >300 | 630,853 (48.9) | 93,076 (14.8) |
Prevalence of Acute Hypertension
A total of 763,634 (59.2%) patients had at least 1 SBP measurement of 140 mmHg during the ED stay, including 178,197 (13.8%) with SBP >180 mmHg. Body systems associated with the highest prevalence of severe AH (SBP >180 mmHg) were nervous (29.0%), circulatory (16.0%), endocrine (14.7%), and kidney/urinary (13.5%) (Figure 1 presents the data in graphic form; Supporting Appendix B, in the online version of this article, presents corresponding data in table form).
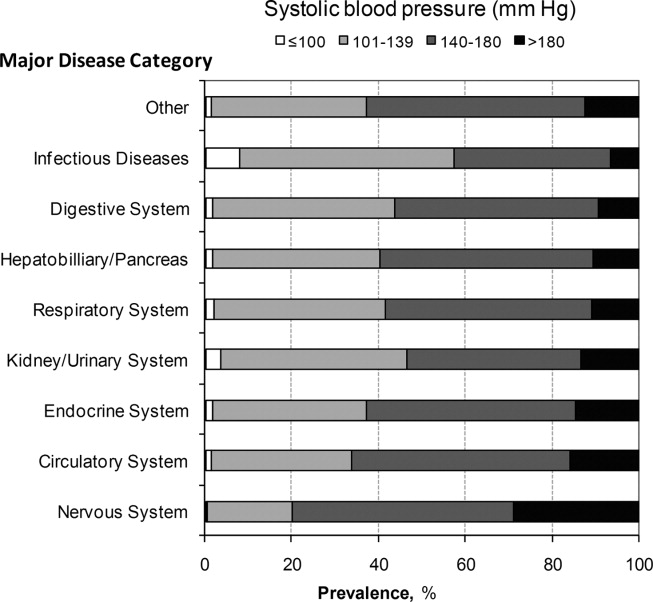
Mortality
Univariable analysis revealed a graded relationship between SBP stratum and mortality risk (Figure 2a; and see Supporting Appendix C in the online version of this article). This relationship was most pronounced for nervous system diseases; mortality rates for each 10 mmHg increase in SBP from 180 to >220 mmHg were 6.5%, 8.1%, 10.0%, 12.0%, and 19.7%, respectively (trending P < 0.0001). The risk‐adjusted increase in mortality odds ratio ranged from 1.04 (95% CI: 0.89, 1.21) to 1.44 (95% CI: 1.25, 1.67) for patients in the severe AH strata compared to patients with SBP of 101 to 139 mmHg (Figure 3). Severe AH was not an independent mortality predictor in other disease categories.
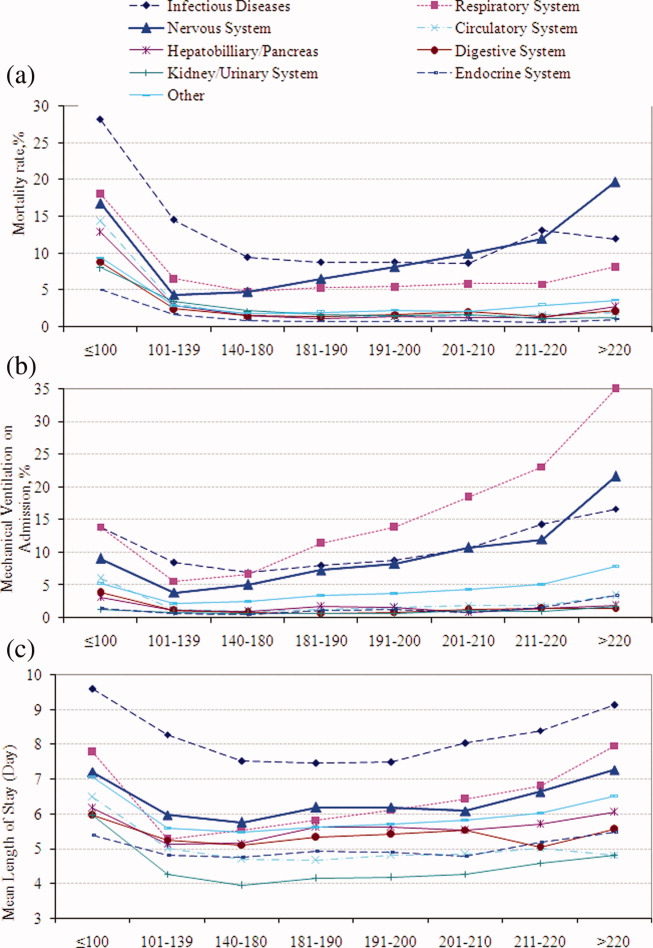
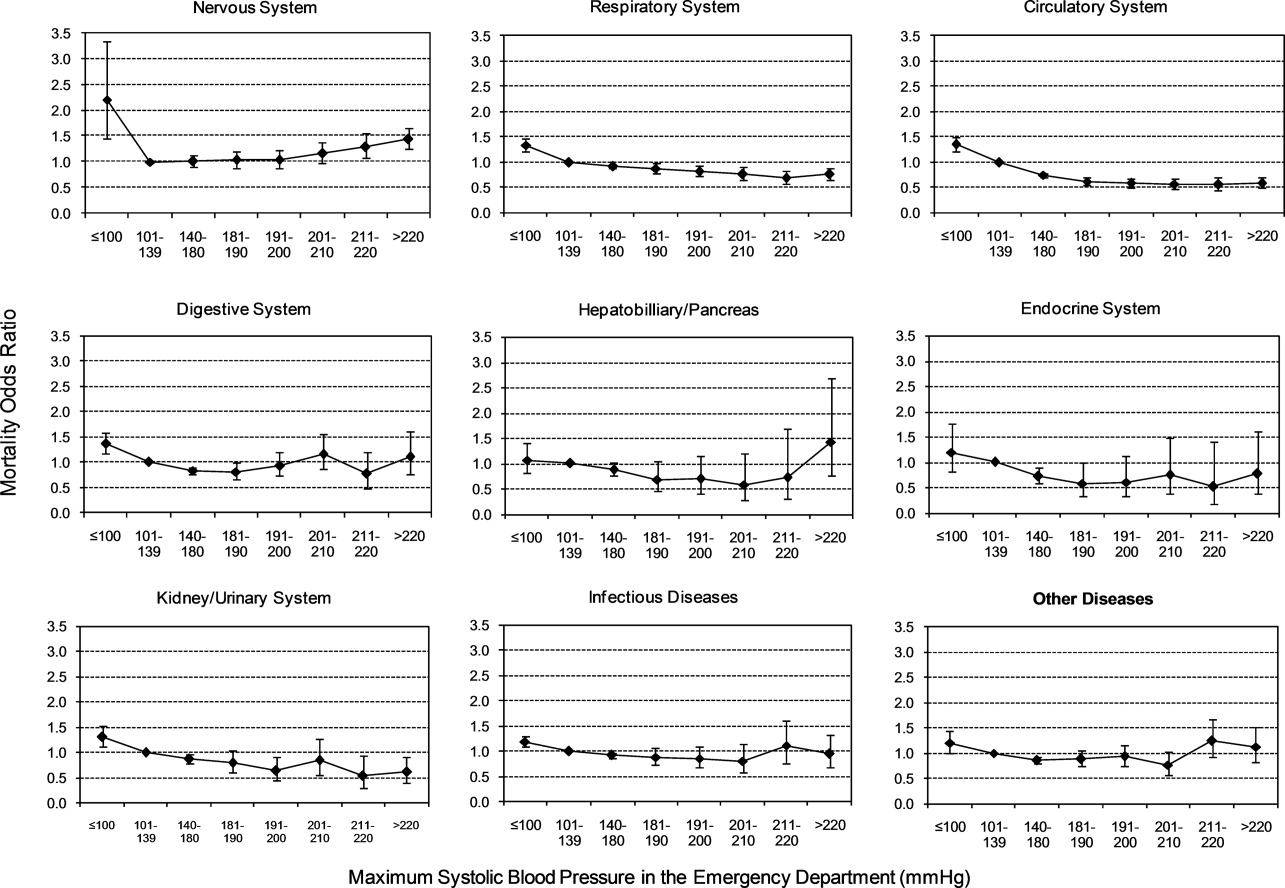
Mechanical Ventilation on Admission
Univariable analysis revealed a graded relationship between severe AH and a need for MV on admission, especially for respiratory, circulatory, and infectious conditions (trending P < 0.0001) (Figure 2b; and see Supporting Appendix C in the online version of this article). In the multivariable analysis, there was a relationship between severe AH stratum and adjusted risk for MV on admission across nearly all disease categories (Figure 4).
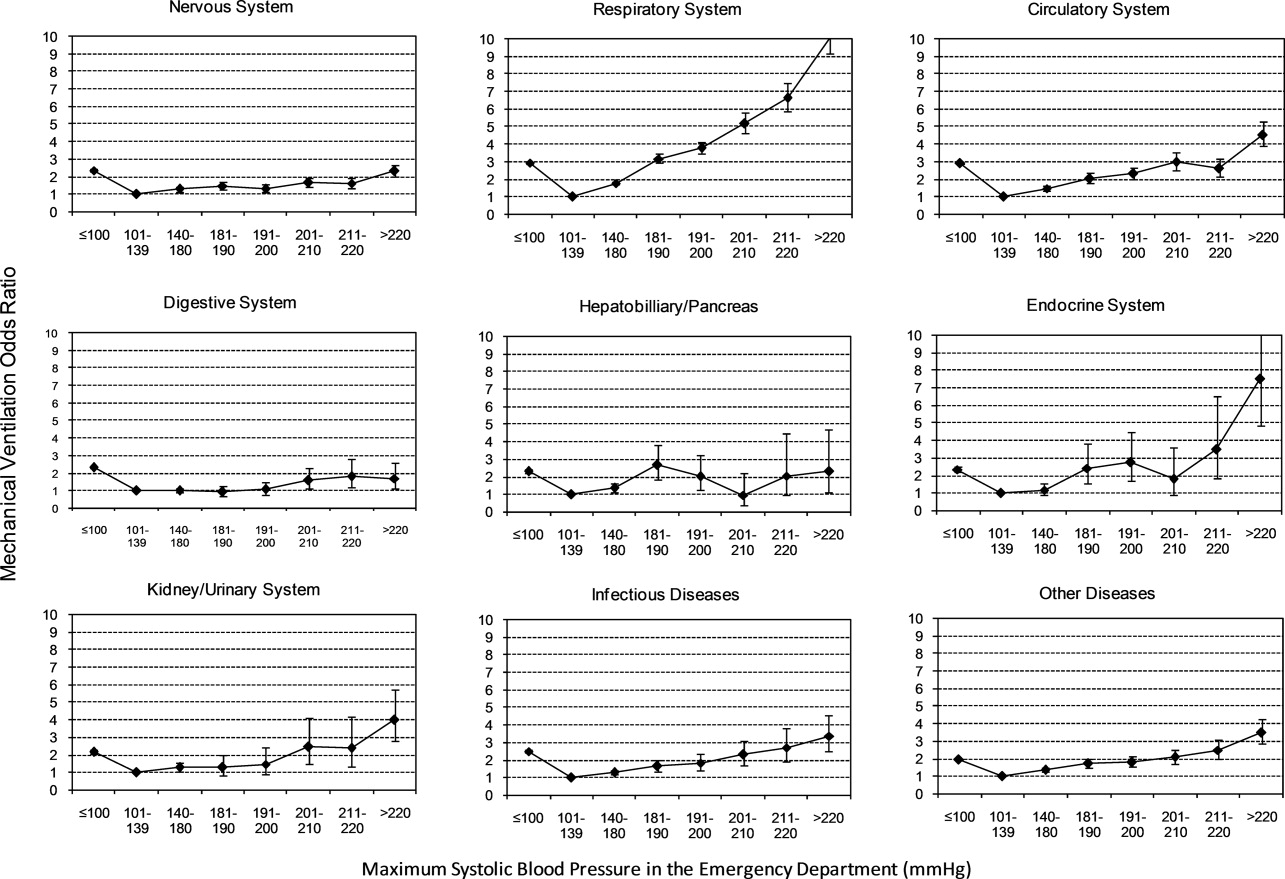
Length of Stay
Univariable analysis revealed a graded relationship between severe AH strata and LOS for nearly all disease categories in survivors (trending P < 0.0001), except for digestive, kidney, and infectious diseases (Figure 2c; and see Supporting Appendix C in the online version of this article). For patients with nervous system diseases, the unadjusted LOS for each 10 mmHg increase in SBP from 180 to >220 was 5.8, 6.1, 6.4, 6.8, and 8.0 days, respectively (trending P < 0.0001). The relationship was similar for other disease categories which showed significant trending.
In the multivariable analysis, the relationship between severe AH strata and adjusted attributable LOS was graded across most disease categories, especially nervous, circulatory, and hepatobiliary diseases (Figure 5). The total adjusted number of hospital days attributable to severe AH was 0.43 days per case for all survivors with severe AH.
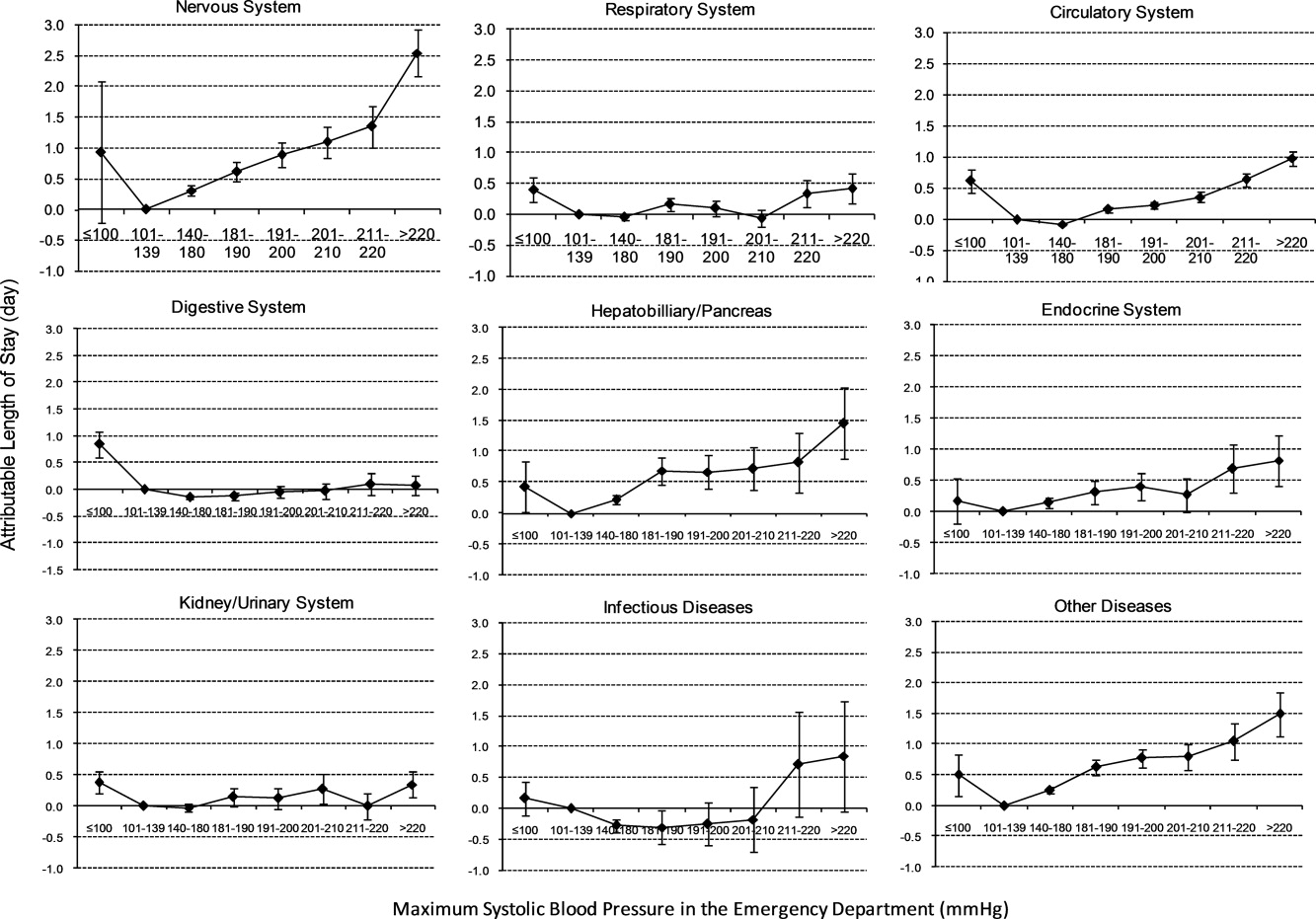
Sensitivity Analysis
Our sensitivity analysis of the LOS estimate, including those patients who died in the hospital, yielded similar findings in attributable LOS due to severe AH. In addition, when we added patients with only 1 SBP documented in the ED to the study cohort, the severe AH prevalence changed negligibly from 13.8% to 13.0%, and the associated mortality remained unchanged at 3.4%. Models using the mix model approach, which take into account hospital‐specific effects, showed similar results.
DISCUSSION
This large‐scale analysis demonstrated that severe AH was present in 13.8% of inpatients admitted through the ED. The prevalence of severe AH varied based on the primary reason for an acute care admission, ranging from 7% in infectious syndromes to nearly 30% in acute neurologic processes. Specific to patients with neurologic disease, initial severe blood pressure elevations independently correlated with mortality. Severe blood pressure elevations at ED were independently associated with an increased need for MV on admission and a prolonged LOS across a range of disease states.
Prior work on hypertension at admission has generally included single‐center analyses or only focused on patients with specific admitting diagnoses. For example, in a single ED analysis, Tilman et al.17 reported that 16% of 10,000 patients presented with elevated blood pressure (140/90 mmHg). In a multicenter review of 7000 persons, Karras et al.18, 19 described 423 patients with severe AH (180/110 mmHg) who comprised 6% of patients seen in the ED during a 1‐week period. Qureshi et al.5 noted severe AH in 13% of patients with acute stroke. While other ED‐based studies examined all ED patients, including those admitted and discharged from the ED, our study focused on those requiring hospitalization. This disparity in illness severity may, in part, explain the higher prevalence of severe AH we noted relative to others.
We further found that the prevalence of severe AH varied based on admitting diagnosis. This difference in prevalence rates by condition seems clinically plausible. Recognition of this pattern may prove valuable to hospitalists who, by the nature of their responsibilities, will encounter a broad range of patients. Because our data were derived from the largest analysis of blood pressure assessments for ED patients who were eventually hospitalized, and encompassed a multiplicity of hospitals, our findings are likely generalizable. Moreover, our large sample size enabled us to examine severe AH at each 10 mmHg increment across a variety of disease states, rather than restricting our analysis to 1 admitting diagnosis.
The independent relationship between severe blood pressure elevation and mortality was detected only in those with neurologic conditions. Incremental increases in SBP beyond 180 mmHg were associated with a stepwise escalation in the risk for death. Although recognition of the importance of blood pressure management in both ischemic and hemorrhagic stroke remains a cornerstone of therapy for these diseases, the stepwise relationship between escalating blood pressure and outcome suggests that further study is needed to determine the optimal management of severe AH among these patients. This relationship, along with the independent association between severe AH at presentation and the need for MV, underscores the importance of severe AH in critical care, representing a major challenge for intensivists and hospitalists, particularly those who practice in neurologic ICUs.
The independent association of severe AH and prolonged LOS represents a novel finding. Few reports have correlated the initial blood pressure with measures of resource use. Katz et al.4 found a median LOS of 6 days among 1000 patients who presented with severe AH and end organ dysfunction in 25 US hospitals. These investigators, however, did not explore the incremental independent contribution of initial blood pressure to LOS. Biologically, severe hypertension may exacerbate both acute and chronic conditions, thus complicating their management and resulting in longer hospitalizations.
Our analysis has limitations. While exposure misclassification is a potential concern, unlike other population‐based studies that use ICD‐9‐CM codes to identify AH cases, we relied on actual measures of blood pressure to identify subjects, thus minimizing this threat to validity. Similarly, since our end pointsmortality, MV on admission, and LOSwere also objective measures, the probability of their misclassification is minimal.
Another concerning contributor to exposure misclassification is the possibility that, in some instances, the initial elevation in BP meeting the inclusion criteria in our study cohort does not reflect the true BP. Indeed, a substantial body of research about BP measurement in the ED suggests that we may have included some persons who likely did not have AH. For example, Pitts and Adams described a regression to the mean phenomenon, with serial BP measurements in the ED, wherein the BP fell by approximately 11 mmHg over 4 hours.20 Baumann and colleagues reported a similar pattern.21 However, the findings of these 2 analyses do not necessarily apply to our study population; we focused on patients admitted to the hospital with an acute condition, while Pitts and Adams20 and Baumann et al.21 examined all ED patients. This distinction is crucial in that patients not admitted are likely less severely ill and systematically different from those who do merit hospitalization. Moreover, both Pitts and Adams20 and Cienki et al.22 observed that the regression to the mean and fall in serial BP measurements were less pronounced in those with the most extensive BP elevations. In our study, we found the strongest relationship between adverse outcomes and BP in patients with the most extreme BP elevations. Thus, misclassification is perhaps less likely to be an issue for these subjects.
In addition, misclassification may result when initially elevated BP simply represents the impact of untreated pain or anxiety in ED patients. However, Backer et al.23 and Tanabe and colleagues24 specifically explored the impact of pain and anxiety on BP in ED subjects, and neither group found a correlation between BP and either acute pain and/or anxiety scores. Our difficulties with case definitions and BP measurements for severe AH demonstrate the need for the creation and adoption of a formal, systematic approach to this syndrome, along with the need for prospective analyses to confirm our findings.
Selection bias represents a second potential threat to validity in our observational study, although this bias is mitigated by including all consecutive acute inpatient admissions to the participating hospitals. Furthermore, inclusion of the 9% of patients who had only 1 ED measurement of SBP collected did not alter the estimate of severe AH prevalence or associated outcomes.
Third, confounding may introduce the potential for false associations derived from observational data. The large sample size of our cohort allowed us to address this concern by adjusting for a large array of confounders. In addition, unlike other large‐scale population‐based studies which typically rely on administrative ICD‐9‐CM codes for risk adjustment, our analysis incorporated actual physiologic and laboratory results measured on admission, as well as a validated severity‐of‐illness scoring system for risk adjustment.12, 13
Although both SBP and diastolic blood pressure (DBP) thresholds are included in traditional definitions of hypertension, selecting SBP as the primary measure is reasonable because SBP >180 mmHg is a more important risk factor for cardiovascular disease than elevated DBP.25 Previous studies reported the relationship between the trend of SBP over time and clinical outcomes,26, 27 but we were not able to investigate the relationship of SBP trend and outcomes because the BP measurements in the our study were not collected in predefined intervals.
It would be ideal if serial blood pressure measures were to be collected at pre‐specified intervals and if more sophisticated schemas were to be used to refine the AH definition. This type of study may be possible in the future when vital signs can be collected automatically with advanced technology. Likewise, electronically captured treatment data could further help researchers to study the impact of process‐of‐care variables, including medications and other management strategies, in relation to outcomes. Finally, outpatient management of chronic hypertension is an integral part of clinical management. Unfortunately, these types of data are not available in our existing database. These limitations notwithstanding, an in‐depth understanding of the association between severe AH and potential adverse clinical and economic outcomes may direct further research in this field.
CONCLUSION
Severe AH appears common and its prevalence varies by underlying clinical condition in patients admitted from the ED. In those with acute neurologic syndromes, the degree of blood pressure elevation correlated with mortality, need for MV, and longer LOS. For many other conditions, elevation of blood pressure appeared to be linked to an increased need for MV and a prolongation in LOS. Future studies are needed to examine the potential impact of both 1) improved long‐term outpatient BP management, and 2) optimal management of severe AH upon admission on improving outcomes of patients hospitalized from the ED with severe AH.
- Health, United States, 2010.Hyattsville, MD:Centers for Disease Control and Prevention, National Center for Health Statistics;2011.
- Agency for Healthcare Research and Quality.Healthcare Cost and Utilization Project (HCUP).Rockville, MD:US Department of Health 289(19):2560–2572.
- ,,, et al.Practice patterns, outcomes, and end‐organ dysfunction for patients with acute severe hypertension: the Studying the Treatment of Acute hyperTension (STAT) registry.Am Heart J.2009;158(4):599–606.e1.
- ,,, et al.Prevalence of elevated blood pressure in 563,704 adult patients with stroke presenting to the ED in the United States.Am J Emerg Med.2007;25(1):32–38.
- ,,, et al.A prediction rule to identify low‐risk patients with community‐acquired pneumonia.N Engl J Med.1997;336(4):243–250.
- ,.A clinical assessment of MedisGroups.JAMA.1988;260(21):3159–3163.
- ,,,,,.Healthcare‐associated bloodstream infection: a distinct entity? Insights from a large U.S. database.Crit Care Med.2006;34(10):2588–2595.
- ,,,,,.Epidemiology and outcomes of health‐care‐associated pneumonia: results from a large US database of culture‐positive pneumonia.Chest.2005;128(6):3854–3862.
- ,,,,,.Morbidity and cost burden of methicillin‐resistant Staphylococcus aureus in early onset ventilator‐associated pneumonia.Crit Care.2006;10(3):R97.
- ,,,,.Evaluation of the complication rate as a measure of quality of care in coronary artery bypass graft surgery.JAMA.1995;274(4):317–323.
- ,,.Using automated clinical data for risk adjustment: development and validation of six disease‐specific mortality predictive models for pay‐for‐performance.Med Care.2007;45(8):789–805.
- ,,,,.Development and validation of a disease‐specific risk adjustment system using automated clinical data.Health Service Research.2010;45:1815–1835.
- ,.Estimating marginal and incremental effects on health outcomes using flexible link and variance function models.Biostatistics.2005;6(1):93–109.
- ,.An Introduction to the Bootstrap.London, England:Chapman 1993.
- ,.Applied Mixed Models in Medicine.2nd ed.Chichester, England:Wiley;2006.
- ,,,,,.Recognizing asymptomatic elevated blood pressure in ED patients: how good (bad) are we?Am J Emerg Med.2007;25(3):313–317.
- ,,, et al.Evaluation and treatment of patients with severely elevated blood pressure in academic emergency departments: a multicenter study.Ann Emerg Med.2006;47(3):230–236.
- ,,, et al.Elevated blood pressure in urban emergency department patients.Acad Emerg Med.2005;12(9):835–843.
- ,.Emergency department hypertension and regression to the mean.Ann Emerg Med.1998;31(2):214–218.
- ,,,.Differing prevalence estimates of elevated blood pressure in ED patients using 4 methods of categorization.Am J Emerg Med.2008;26(5):561–565.
- ,,.The validity of emergency department triage blood pressure measurements.Acad Emerg Med.2004;11(3):237–243.
- ,,.Reproducibility of increased blood pressure during an emergency department or urgent care visit.Ann Emerg Med.2003;41(4):507–512.
- ,,,,,.Increased blood pressure in the emergency department: pain, anxiety, or undiagnosed hypertension?Ann Emerg Med.2008;51(3):221–229.
- ,,.Clinical Advisory Statement. Importance of systolic blood pressure in older Americans.Hypertension.2000;35(5):1021–1024.
- ,,,.High blood pressure in early acute stroke: a sign of a poor outcome?J Hypertens.2006;24(2):381–386.
- ,,,,.Blood pressure as an independent prognostic factor in acute ischemic stroke.Can J Neurol Sci.2006;33(1):34–38.
Chronic hypertension affects 32% of adults in the United States.1 Each year, over 20 million emergency department visits involve hypertension.2 While many studies describe the epidemiology and outcomes of chronic hypertension, less is known about severe acute hypertension (AH). Often classified as either hypertensive urgency or emergency, it receives little attention in national treatment guidelines.3 There is a limited understanding of the epidemiology of, and the relationship between, this syndrome and patient outcomes among hospitalized patients. One registry study suggested that severe AH was associated with high rates of new organ damage, need for treatment in the intensive care unit, and a 90‐day readmission rate of 10%.4 This investigation, however, lacked generalizability, because it only enrolled subjects requiring therapy with an intravenous antihypertensive agent and did not provide information on the prevalence of severe AH. Qureshi et al.5 analyzed information in a more representative sample from the national hospital ambulatory care survey, however, the only outcome examined was the correlation between acute blood pressure and stroke. Studies focusing on a selected patient population may be of limited value to hospitalists, because they are commonly required to treat a range of patients presenting to the hospital.
In light of severe syndromes that may be associated with, or complicated by, severe AH presented early during acute care, hospitalists require a better understanding of the prevalence and the relationship between severe AH and attendant mortality and morbidity. In addition, an assessment of the association of severe AH on the need for intensive care unit (ICU) admission and mechanical ventilation (MV) may aid the initial treatment assessments and triage decisions required of hospitalists.
Our objective was to describe the prevalence and implications of severe AH present at the time of evaluation in the ED among patients eventually hospitalized, using clinical data collected on all consecutive admissions across a range of clinical conditions. We sought to determine the independent contribution, if any, of severe AH to hospital mortality, need for MV on admission, as well as hospital length of stay (LOS).
METHODS
Study Design and Setting
This was a retrospective analysis of adults admitted to 114 acute‐care hospitals in the United States from 2005 through 2007. The New England Institutional Review Board/Human Subjects Research Committee (Wellesley, MA) reviewed and approved this study. It was conducted in compliance with the Health Insurance Portability and Accountability Act (HIPAA).
Data
Data were obtained from one of the Clinical Research Databases from CareFusion (formerly Cardinal Health [CareFusion Clinical Research Services, Marlborough, MA]).613 The database contains electronically imported or manually extracted demographic, clinical (eg, comorbidities, vital signs, laboratory values, other clinical findings), and administrative data (eg, diagnosis, procedures, and length of hospitalization). All vital signs were manually extracted, including the highest and the lowest ED systolic blood pressure (SBP) measurements during the ED stay, but before inpatient admission. Patients admitted for childbirth or mental health reasons were not included.
Patients
Our main analysis focused on patients whose highest and lowest SBP were collected in the ED. These patients accounted for approximately 90% of all inpatients admitted through the ED. For the approximately 9% of patients who had only 1 SBP collected, we conducted a sensitivity analysis by both including and excluding them in the main analysis to determine if the absence of a second SBP measurement altered our findings. Patients were grouped into 1 of the following 8 mutually exclusive strata based on the maximum SBP (mmHg) in the ED: <100, 101‐139, 140‐180, 181‐190, 191‐200, 201‐210, 211‐220, and >220.
Measures
The primary exposure of interest was the prevalence of severe AH, defined as at least 1 SBP measurement recorded in the ED in excess of 180 mmHg. Outcome measures included in‐hospital mortality, need for MV on admission day (defined by International Classification of Diseases, 9th revision, Clinical Modification [ICD‐9‐CM] procedure codes of 96.70, 96.71, and 96.72), and LOS. We stratified these outcomes for each inpatient admission according to 1 of 112 mutually exclusive groups of principal diagnoses (see Supporting Appendix A in the online version of this article). To simplify the presentations, we pooled the groups into 9 major disease categories based on organ systems.
Primary Data Analysis
All statistical analyses were performed using Statistical Analysis Software (SAS version 9.01; SAS Institute Inc, Cary, NC). For evaluating trending, we used the Cochran‐Armitage test for dichotomous variables (mortality and MV), and linear regression for continuous variable (LOS). We employed a logistic regression model to estimate risk of mortality and need for MV on admission. We used linear regression models to estimate the LOS associated with severe AH. We modeled each outcome as a function of patient disease severity and SBP strata. Because patients with the most severe AH tended to have higher mortality early in hospitalization, our analysis of LOS was limited to patients who survived index hospitalization.
The original disease‐specific risk‐adjustment models accounting for patient‐level confounding risk factors, including demographics, physiologic presentation on admission (vital signs, altered mental status, and laboratory findings), and chronic conditions, were previously developed and validated.12, 13 We recalibrated each of the 112 models, for the current study cohort, using the logit of predicted probability of death generated from the mortality risk‐adjustment model as a propensity score of disease severity. Using this propensity score as an aggregate severity adjuster, we refit 9 logistic regression models (1 for each major disease category) to estimate the odds ratios for mortality or need for MV for each of the 7 SBP strata in the regression models with 101‐139 mmHg as the reference group. To estimate the attributable LOS (if any) of severe AH among survivors, we fit the 9 LOS models using log transformed LOS (to normalize the potentially skewed distribution of LOS) as the outcome, controlling for disease severity. The attributable LOS and 95% confidence intervals (CI) were estimated from 1000 bootstrap iterations, with the median as the parameter estimate and 2.5th and 97.5th percentile as 95% CI.14, 15
Sensitivity Analysis
To address potential bias of LOS associated with inpatient mortality, we refit 9 LOS models, including both patients who died and those who survived the index hospitalization. The models adjusted for disease severity, mortality, and the interaction of severity and mortality. Because patients with only 1 SBP recorded at ED may be different from those with more than 1 SBP recorded, we conducted analysis by adding these patients in the study cohort to examine the potential change of overall prevalence of AH and associated mortality for the study population.
To address the potential for a center‐specific effect on outcomes, we refit all the models using a mixed model approach.16 The mixed model accounts for both patient‐level risk factors and hospital‐specific effects on the observed outcomes.
RESULTS
Patient Characteristics
The study cohort was comprised of 1,290,804 adults who were admitted through the ED, from 2005 through 2007, and whose highest and lowest SBP measurements were collected in the ED. Median age was 69 years (interquartile range, 53‐81) for the overall population. Median age was 74 (interquartile range, 60‐83) for patients with severe AH (Table 1). Hospital mortality was 3.6% (n = 46,033), with 6119 (13.3%) having severe AH.
| Characteristic | Prevalence, n (Column %) | Severe Acute Hypertension, n (Row %) |
|---|---|---|
| Total number of discharges | 1,290,804 (100.0) | 178,197 (13.8) |
| Mortality | 46,033 (3.6) | 6,119 (13.3) |
| Live discharges | 1,244,771 (96.4) | 172,078 (13.8) |
| Mechanical ventilation on admission | 39,238 (3.0) | 9,508 (24.2) |
| Demographics | ||
| Age, median (1st, 3rd quartiles) | 69 (53, 81) | 74 (60, 83) |
| Male | 587,553 (45.5) | 71,085 (12.1) |
| Female | 703,244 (54.5) | 107,109 (15.2) |
| Race | ||
| White | 949,869 (73.6) | 121,930 (12.8) |
| Black | 220,601 (17.1) | 39,667 (18.0) |
| Other | 120,334 (9.3) | 16,600 (13.8) |
| Insurance | ||
| Medicare | 668,420 (51.8) | 105,078 (15.7) |
| Medicaid | 108,538 (8.4) | 12,259 (11.3) |
| Commercial | 163,858 (12.7) | 18,669 (11.4) |
| Other | 349,988 (27.1) | 42,191 (12.1) |
| Disease system by the principal diagnosis | ||
| Nervous system | 76,744 (5.9) | 22,270 (29.0) |
| Respiratory system | 222,329 (17.2) | 24,678 (11.1) |
| Circulatory system | 416,847 (32.3) | 66,852 (16.0) |
| Digestive system | 186,282 (14.4) | 17,817 (9.6) |
| Hepatobiliary/pancreas system | 52,293 (4.1) | 5,664 (10.8) |
| Endocrine system | 45,050 (3.5) | 6,625 (14.7) |
| Kidney/urinary system | 81,782 (6.3) | 11,050 (13.5) |
| Infectious diseases | 60,353 (4.7) | 4,162 (6.9) |
| Other | 149,124 (11.6) | 19,079 (12.8) |
| Comorbidity by secondary diagnoses | ||
| Hypertension | 729,417 (56.5) | 135,498 (18.6) |
| Fluid and electrolyte disorders | 306,666 (23.8) | 37,836 (12.3) |
| Diabetes without chronic complications | 286,912 (22.2) | 47,979 (16.7) |
| Chronic pulmonary disease | 283,895 (22.0) | 35,977 (12.7) |
| Congestive heart failure | 213,523 (16.5) | 33,956 (15.9) |
| Deficiency anemias | 210,230 (16.3) | 30,266 (14.4) |
| Renal failure | 159,409 (12.3) | 31,984 (20.1) |
| Hypothyroidism | 153,911 (11.9) | 22,441 (14.6) |
| Valvular disease | 140,820 (10.9) | 21,453 (15.2) |
| Depression | 137,259 (10.6) | 16,886 (12.3) |
| Other neurological disorders | 126,954 (9.8) | 19,103 (15.0) |
| Peripheral vascular disease | 88,321 (6.8) | 16,180 (18.3) |
| Obesity | 84,000 (6.5) | 12,351 (14.7) |
| Diabetes with chronic complications | 65,989 (5.1) | 13,093 (19.8) |
| Psychoses | 54,769 (4.2) | 5,555 (10.1) |
| Alcohol abuse | 51,765 (4.0) | 6,014 (11.6) |
| Pulmonary circulation disease | 49,248 (3.8) | 7,128 (14.5) |
| Coagulopathy | 43,584 (3.4) | 4,339 (10.0) |
| Paralysis | 42,128 (3.3) | 8,125 (19.3) |
| Drug abuse | 36,134 (2.8) | 4,779 (13.2) |
| Liver disease | 36,094 (2.8) | 3,218 (8.9) |
| Weight loss | 35,795 (2.8) | 3,726 (10.4) |
| Metastatic cancer | 33,517 (2.6) | 2,498 (7.5) |
| Rheumatoid arthritis | 32,545 (2.5) | 4,300 (13.2) |
| Solid tumor without metastasis | 30,677 (2.4) | 3,035 (9.9) |
| Chronic blood loss anemia | 25,416 (2.0) | 2,268 (8.9) |
| Lymphoma | 9,972 (0.8) | 871 (8.7) |
| Acquired immune deficiency syndrome | 3,048 (0.2) | 307 (10.1) |
| Peptic ulcer disease | 915 (0.1) | 131 (14.3) |
| Discharges by hospital characteristics | ||
| Teaching status | ||
| Teaching hospitals | 899,786 (69.7) | 127,512 (14.2) |
| Nonteaching hospitals | 391,018 (30.3) | 50,685 (13.0) |
| Urban status | ||
| Urban hospitals | 1,164,802 (90.2) | 162,399 (13.9) |
| Rural hospitals | 126,002 (9.8) | 15,798 (12.5) |
| Bed size | ||
| Beds <100 | 36,624 (2.8) | 4,965 (13.6) |
| Beds 100‐300 | 623,327 (48.3) | 80,156 (12.9) |
| Beds >300 | 630,853 (48.9) | 93,076 (14.8) |
Prevalence of Acute Hypertension
A total of 763,634 (59.2%) patients had at least 1 SBP measurement of 140 mmHg during the ED stay, including 178,197 (13.8%) with SBP >180 mmHg. Body systems associated with the highest prevalence of severe AH (SBP >180 mmHg) were nervous (29.0%), circulatory (16.0%), endocrine (14.7%), and kidney/urinary (13.5%) (Figure 1 presents the data in graphic form; Supporting Appendix B, in the online version of this article, presents corresponding data in table form).

Mortality
Univariable analysis revealed a graded relationship between SBP stratum and mortality risk (Figure 2a; and see Supporting Appendix C in the online version of this article). This relationship was most pronounced for nervous system diseases; mortality rates for each 10 mmHg increase in SBP from 180 to >220 mmHg were 6.5%, 8.1%, 10.0%, 12.0%, and 19.7%, respectively (trending P < 0.0001). The risk‐adjusted increase in mortality odds ratio ranged from 1.04 (95% CI: 0.89, 1.21) to 1.44 (95% CI: 1.25, 1.67) for patients in the severe AH strata compared to patients with SBP of 101 to 139 mmHg (Figure 3). Severe AH was not an independent mortality predictor in other disease categories.


Mechanical Ventilation on Admission
Univariable analysis revealed a graded relationship between severe AH and a need for MV on admission, especially for respiratory, circulatory, and infectious conditions (trending P < 0.0001) (Figure 2b; and see Supporting Appendix C in the online version of this article). In the multivariable analysis, there was a relationship between severe AH stratum and adjusted risk for MV on admission across nearly all disease categories (Figure 4).

Length of Stay
Univariable analysis revealed a graded relationship between severe AH strata and LOS for nearly all disease categories in survivors (trending P < 0.0001), except for digestive, kidney, and infectious diseases (Figure 2c; and see Supporting Appendix C in the online version of this article). For patients with nervous system diseases, the unadjusted LOS for each 10 mmHg increase in SBP from 180 to >220 was 5.8, 6.1, 6.4, 6.8, and 8.0 days, respectively (trending P < 0.0001). The relationship was similar for other disease categories which showed significant trending.
In the multivariable analysis, the relationship between severe AH strata and adjusted attributable LOS was graded across most disease categories, especially nervous, circulatory, and hepatobiliary diseases (Figure 5). The total adjusted number of hospital days attributable to severe AH was 0.43 days per case for all survivors with severe AH.

Sensitivity Analysis
Our sensitivity analysis of the LOS estimate, including those patients who died in the hospital, yielded similar findings in attributable LOS due to severe AH. In addition, when we added patients with only 1 SBP documented in the ED to the study cohort, the severe AH prevalence changed negligibly from 13.8% to 13.0%, and the associated mortality remained unchanged at 3.4%. Models using the mix model approach, which take into account hospital‐specific effects, showed similar results.
DISCUSSION
This large‐scale analysis demonstrated that severe AH was present in 13.8% of inpatients admitted through the ED. The prevalence of severe AH varied based on the primary reason for an acute care admission, ranging from 7% in infectious syndromes to nearly 30% in acute neurologic processes. Specific to patients with neurologic disease, initial severe blood pressure elevations independently correlated with mortality. Severe blood pressure elevations at ED were independently associated with an increased need for MV on admission and a prolonged LOS across a range of disease states.
Prior work on hypertension at admission has generally included single‐center analyses or only focused on patients with specific admitting diagnoses. For example, in a single ED analysis, Tilman et al.17 reported that 16% of 10,000 patients presented with elevated blood pressure (140/90 mmHg). In a multicenter review of 7000 persons, Karras et al.18, 19 described 423 patients with severe AH (180/110 mmHg) who comprised 6% of patients seen in the ED during a 1‐week period. Qureshi et al.5 noted severe AH in 13% of patients with acute stroke. While other ED‐based studies examined all ED patients, including those admitted and discharged from the ED, our study focused on those requiring hospitalization. This disparity in illness severity may, in part, explain the higher prevalence of severe AH we noted relative to others.
We further found that the prevalence of severe AH varied based on admitting diagnosis. This difference in prevalence rates by condition seems clinically plausible. Recognition of this pattern may prove valuable to hospitalists who, by the nature of their responsibilities, will encounter a broad range of patients. Because our data were derived from the largest analysis of blood pressure assessments for ED patients who were eventually hospitalized, and encompassed a multiplicity of hospitals, our findings are likely generalizable. Moreover, our large sample size enabled us to examine severe AH at each 10 mmHg increment across a variety of disease states, rather than restricting our analysis to 1 admitting diagnosis.
The independent relationship between severe blood pressure elevation and mortality was detected only in those with neurologic conditions. Incremental increases in SBP beyond 180 mmHg were associated with a stepwise escalation in the risk for death. Although recognition of the importance of blood pressure management in both ischemic and hemorrhagic stroke remains a cornerstone of therapy for these diseases, the stepwise relationship between escalating blood pressure and outcome suggests that further study is needed to determine the optimal management of severe AH among these patients. This relationship, along with the independent association between severe AH at presentation and the need for MV, underscores the importance of severe AH in critical care, representing a major challenge for intensivists and hospitalists, particularly those who practice in neurologic ICUs.
The independent association of severe AH and prolonged LOS represents a novel finding. Few reports have correlated the initial blood pressure with measures of resource use. Katz et al.4 found a median LOS of 6 days among 1000 patients who presented with severe AH and end organ dysfunction in 25 US hospitals. These investigators, however, did not explore the incremental independent contribution of initial blood pressure to LOS. Biologically, severe hypertension may exacerbate both acute and chronic conditions, thus complicating their management and resulting in longer hospitalizations.
Our analysis has limitations. While exposure misclassification is a potential concern, unlike other population‐based studies that use ICD‐9‐CM codes to identify AH cases, we relied on actual measures of blood pressure to identify subjects, thus minimizing this threat to validity. Similarly, since our end pointsmortality, MV on admission, and LOSwere also objective measures, the probability of their misclassification is minimal.
Another concerning contributor to exposure misclassification is the possibility that, in some instances, the initial elevation in BP meeting the inclusion criteria in our study cohort does not reflect the true BP. Indeed, a substantial body of research about BP measurement in the ED suggests that we may have included some persons who likely did not have AH. For example, Pitts and Adams described a regression to the mean phenomenon, with serial BP measurements in the ED, wherein the BP fell by approximately 11 mmHg over 4 hours.20 Baumann and colleagues reported a similar pattern.21 However, the findings of these 2 analyses do not necessarily apply to our study population; we focused on patients admitted to the hospital with an acute condition, while Pitts and Adams20 and Baumann et al.21 examined all ED patients. This distinction is crucial in that patients not admitted are likely less severely ill and systematically different from those who do merit hospitalization. Moreover, both Pitts and Adams20 and Cienki et al.22 observed that the regression to the mean and fall in serial BP measurements were less pronounced in those with the most extensive BP elevations. In our study, we found the strongest relationship between adverse outcomes and BP in patients with the most extreme BP elevations. Thus, misclassification is perhaps less likely to be an issue for these subjects.
In addition, misclassification may result when initially elevated BP simply represents the impact of untreated pain or anxiety in ED patients. However, Backer et al.23 and Tanabe and colleagues24 specifically explored the impact of pain and anxiety on BP in ED subjects, and neither group found a correlation between BP and either acute pain and/or anxiety scores. Our difficulties with case definitions and BP measurements for severe AH demonstrate the need for the creation and adoption of a formal, systematic approach to this syndrome, along with the need for prospective analyses to confirm our findings.
Selection bias represents a second potential threat to validity in our observational study, although this bias is mitigated by including all consecutive acute inpatient admissions to the participating hospitals. Furthermore, inclusion of the 9% of patients who had only 1 ED measurement of SBP collected did not alter the estimate of severe AH prevalence or associated outcomes.
Third, confounding may introduce the potential for false associations derived from observational data. The large sample size of our cohort allowed us to address this concern by adjusting for a large array of confounders. In addition, unlike other large‐scale population‐based studies which typically rely on administrative ICD‐9‐CM codes for risk adjustment, our analysis incorporated actual physiologic and laboratory results measured on admission, as well as a validated severity‐of‐illness scoring system for risk adjustment.12, 13
Although both SBP and diastolic blood pressure (DBP) thresholds are included in traditional definitions of hypertension, selecting SBP as the primary measure is reasonable because SBP >180 mmHg is a more important risk factor for cardiovascular disease than elevated DBP.25 Previous studies reported the relationship between the trend of SBP over time and clinical outcomes,26, 27 but we were not able to investigate the relationship of SBP trend and outcomes because the BP measurements in the our study were not collected in predefined intervals.
It would be ideal if serial blood pressure measures were to be collected at pre‐specified intervals and if more sophisticated schemas were to be used to refine the AH definition. This type of study may be possible in the future when vital signs can be collected automatically with advanced technology. Likewise, electronically captured treatment data could further help researchers to study the impact of process‐of‐care variables, including medications and other management strategies, in relation to outcomes. Finally, outpatient management of chronic hypertension is an integral part of clinical management. Unfortunately, these types of data are not available in our existing database. These limitations notwithstanding, an in‐depth understanding of the association between severe AH and potential adverse clinical and economic outcomes may direct further research in this field.
CONCLUSION
Severe AH appears common and its prevalence varies by underlying clinical condition in patients admitted from the ED. In those with acute neurologic syndromes, the degree of blood pressure elevation correlated with mortality, need for MV, and longer LOS. For many other conditions, elevation of blood pressure appeared to be linked to an increased need for MV and a prolongation in LOS. Future studies are needed to examine the potential impact of both 1) improved long‐term outpatient BP management, and 2) optimal management of severe AH upon admission on improving outcomes of patients hospitalized from the ED with severe AH.
Chronic hypertension affects 32% of adults in the United States.1 Each year, over 20 million emergency department visits involve hypertension.2 While many studies describe the epidemiology and outcomes of chronic hypertension, less is known about severe acute hypertension (AH). Often classified as either hypertensive urgency or emergency, it receives little attention in national treatment guidelines.3 There is a limited understanding of the epidemiology of, and the relationship between, this syndrome and patient outcomes among hospitalized patients. One registry study suggested that severe AH was associated with high rates of new organ damage, need for treatment in the intensive care unit, and a 90‐day readmission rate of 10%.4 This investigation, however, lacked generalizability, because it only enrolled subjects requiring therapy with an intravenous antihypertensive agent and did not provide information on the prevalence of severe AH. Qureshi et al.5 analyzed information in a more representative sample from the national hospital ambulatory care survey, however, the only outcome examined was the correlation between acute blood pressure and stroke. Studies focusing on a selected patient population may be of limited value to hospitalists, because they are commonly required to treat a range of patients presenting to the hospital.
In light of severe syndromes that may be associated with, or complicated by, severe AH presented early during acute care, hospitalists require a better understanding of the prevalence and the relationship between severe AH and attendant mortality and morbidity. In addition, an assessment of the association of severe AH on the need for intensive care unit (ICU) admission and mechanical ventilation (MV) may aid the initial treatment assessments and triage decisions required of hospitalists.
Our objective was to describe the prevalence and implications of severe AH present at the time of evaluation in the ED among patients eventually hospitalized, using clinical data collected on all consecutive admissions across a range of clinical conditions. We sought to determine the independent contribution, if any, of severe AH to hospital mortality, need for MV on admission, as well as hospital length of stay (LOS).
METHODS
Study Design and Setting
This was a retrospective analysis of adults admitted to 114 acute‐care hospitals in the United States from 2005 through 2007. The New England Institutional Review Board/Human Subjects Research Committee (Wellesley, MA) reviewed and approved this study. It was conducted in compliance with the Health Insurance Portability and Accountability Act (HIPAA).
Data
Data were obtained from one of the Clinical Research Databases from CareFusion (formerly Cardinal Health [CareFusion Clinical Research Services, Marlborough, MA]).613 The database contains electronically imported or manually extracted demographic, clinical (eg, comorbidities, vital signs, laboratory values, other clinical findings), and administrative data (eg, diagnosis, procedures, and length of hospitalization). All vital signs were manually extracted, including the highest and the lowest ED systolic blood pressure (SBP) measurements during the ED stay, but before inpatient admission. Patients admitted for childbirth or mental health reasons were not included.
Patients
Our main analysis focused on patients whose highest and lowest SBP were collected in the ED. These patients accounted for approximately 90% of all inpatients admitted through the ED. For the approximately 9% of patients who had only 1 SBP collected, we conducted a sensitivity analysis by both including and excluding them in the main analysis to determine if the absence of a second SBP measurement altered our findings. Patients were grouped into 1 of the following 8 mutually exclusive strata based on the maximum SBP (mmHg) in the ED: <100, 101‐139, 140‐180, 181‐190, 191‐200, 201‐210, 211‐220, and >220.
Measures
The primary exposure of interest was the prevalence of severe AH, defined as at least 1 SBP measurement recorded in the ED in excess of 180 mmHg. Outcome measures included in‐hospital mortality, need for MV on admission day (defined by International Classification of Diseases, 9th revision, Clinical Modification [ICD‐9‐CM] procedure codes of 96.70, 96.71, and 96.72), and LOS. We stratified these outcomes for each inpatient admission according to 1 of 112 mutually exclusive groups of principal diagnoses (see Supporting Appendix A in the online version of this article). To simplify the presentations, we pooled the groups into 9 major disease categories based on organ systems.
Primary Data Analysis
All statistical analyses were performed using Statistical Analysis Software (SAS version 9.01; SAS Institute Inc, Cary, NC). For evaluating trending, we used the Cochran‐Armitage test for dichotomous variables (mortality and MV), and linear regression for continuous variable (LOS). We employed a logistic regression model to estimate risk of mortality and need for MV on admission. We used linear regression models to estimate the LOS associated with severe AH. We modeled each outcome as a function of patient disease severity and SBP strata. Because patients with the most severe AH tended to have higher mortality early in hospitalization, our analysis of LOS was limited to patients who survived index hospitalization.
The original disease‐specific risk‐adjustment models accounting for patient‐level confounding risk factors, including demographics, physiologic presentation on admission (vital signs, altered mental status, and laboratory findings), and chronic conditions, were previously developed and validated.12, 13 We recalibrated each of the 112 models, for the current study cohort, using the logit of predicted probability of death generated from the mortality risk‐adjustment model as a propensity score of disease severity. Using this propensity score as an aggregate severity adjuster, we refit 9 logistic regression models (1 for each major disease category) to estimate the odds ratios for mortality or need for MV for each of the 7 SBP strata in the regression models with 101‐139 mmHg as the reference group. To estimate the attributable LOS (if any) of severe AH among survivors, we fit the 9 LOS models using log transformed LOS (to normalize the potentially skewed distribution of LOS) as the outcome, controlling for disease severity. The attributable LOS and 95% confidence intervals (CI) were estimated from 1000 bootstrap iterations, with the median as the parameter estimate and 2.5th and 97.5th percentile as 95% CI.14, 15
Sensitivity Analysis
To address potential bias of LOS associated with inpatient mortality, we refit 9 LOS models, including both patients who died and those who survived the index hospitalization. The models adjusted for disease severity, mortality, and the interaction of severity and mortality. Because patients with only 1 SBP recorded at ED may be different from those with more than 1 SBP recorded, we conducted analysis by adding these patients in the study cohort to examine the potential change of overall prevalence of AH and associated mortality for the study population.
To address the potential for a center‐specific effect on outcomes, we refit all the models using a mixed model approach.16 The mixed model accounts for both patient‐level risk factors and hospital‐specific effects on the observed outcomes.
RESULTS
Patient Characteristics
The study cohort was comprised of 1,290,804 adults who were admitted through the ED, from 2005 through 2007, and whose highest and lowest SBP measurements were collected in the ED. Median age was 69 years (interquartile range, 53‐81) for the overall population. Median age was 74 (interquartile range, 60‐83) for patients with severe AH (Table 1). Hospital mortality was 3.6% (n = 46,033), with 6119 (13.3%) having severe AH.
| Characteristic | Prevalence, n (Column %) | Severe Acute Hypertension, n (Row %) |
|---|---|---|
| Total number of discharges | 1,290,804 (100.0) | 178,197 (13.8) |
| Mortality | 46,033 (3.6) | 6,119 (13.3) |
| Live discharges | 1,244,771 (96.4) | 172,078 (13.8) |
| Mechanical ventilation on admission | 39,238 (3.0) | 9,508 (24.2) |
| Demographics | ||
| Age, median (1st, 3rd quartiles) | 69 (53, 81) | 74 (60, 83) |
| Male | 587,553 (45.5) | 71,085 (12.1) |
| Female | 703,244 (54.5) | 107,109 (15.2) |
| Race | ||
| White | 949,869 (73.6) | 121,930 (12.8) |
| Black | 220,601 (17.1) | 39,667 (18.0) |
| Other | 120,334 (9.3) | 16,600 (13.8) |
| Insurance | ||
| Medicare | 668,420 (51.8) | 105,078 (15.7) |
| Medicaid | 108,538 (8.4) | 12,259 (11.3) |
| Commercial | 163,858 (12.7) | 18,669 (11.4) |
| Other | 349,988 (27.1) | 42,191 (12.1) |
| Disease system by the principal diagnosis | ||
| Nervous system | 76,744 (5.9) | 22,270 (29.0) |
| Respiratory system | 222,329 (17.2) | 24,678 (11.1) |
| Circulatory system | 416,847 (32.3) | 66,852 (16.0) |
| Digestive system | 186,282 (14.4) | 17,817 (9.6) |
| Hepatobiliary/pancreas system | 52,293 (4.1) | 5,664 (10.8) |
| Endocrine system | 45,050 (3.5) | 6,625 (14.7) |
| Kidney/urinary system | 81,782 (6.3) | 11,050 (13.5) |
| Infectious diseases | 60,353 (4.7) | 4,162 (6.9) |
| Other | 149,124 (11.6) | 19,079 (12.8) |
| Comorbidity by secondary diagnoses | ||
| Hypertension | 729,417 (56.5) | 135,498 (18.6) |
| Fluid and electrolyte disorders | 306,666 (23.8) | 37,836 (12.3) |
| Diabetes without chronic complications | 286,912 (22.2) | 47,979 (16.7) |
| Chronic pulmonary disease | 283,895 (22.0) | 35,977 (12.7) |
| Congestive heart failure | 213,523 (16.5) | 33,956 (15.9) |
| Deficiency anemias | 210,230 (16.3) | 30,266 (14.4) |
| Renal failure | 159,409 (12.3) | 31,984 (20.1) |
| Hypothyroidism | 153,911 (11.9) | 22,441 (14.6) |
| Valvular disease | 140,820 (10.9) | 21,453 (15.2) |
| Depression | 137,259 (10.6) | 16,886 (12.3) |
| Other neurological disorders | 126,954 (9.8) | 19,103 (15.0) |
| Peripheral vascular disease | 88,321 (6.8) | 16,180 (18.3) |
| Obesity | 84,000 (6.5) | 12,351 (14.7) |
| Diabetes with chronic complications | 65,989 (5.1) | 13,093 (19.8) |
| Psychoses | 54,769 (4.2) | 5,555 (10.1) |
| Alcohol abuse | 51,765 (4.0) | 6,014 (11.6) |
| Pulmonary circulation disease | 49,248 (3.8) | 7,128 (14.5) |
| Coagulopathy | 43,584 (3.4) | 4,339 (10.0) |
| Paralysis | 42,128 (3.3) | 8,125 (19.3) |
| Drug abuse | 36,134 (2.8) | 4,779 (13.2) |
| Liver disease | 36,094 (2.8) | 3,218 (8.9) |
| Weight loss | 35,795 (2.8) | 3,726 (10.4) |
| Metastatic cancer | 33,517 (2.6) | 2,498 (7.5) |
| Rheumatoid arthritis | 32,545 (2.5) | 4,300 (13.2) |
| Solid tumor without metastasis | 30,677 (2.4) | 3,035 (9.9) |
| Chronic blood loss anemia | 25,416 (2.0) | 2,268 (8.9) |
| Lymphoma | 9,972 (0.8) | 871 (8.7) |
| Acquired immune deficiency syndrome | 3,048 (0.2) | 307 (10.1) |
| Peptic ulcer disease | 915 (0.1) | 131 (14.3) |
| Discharges by hospital characteristics | ||
| Teaching status | ||
| Teaching hospitals | 899,786 (69.7) | 127,512 (14.2) |
| Nonteaching hospitals | 391,018 (30.3) | 50,685 (13.0) |
| Urban status | ||
| Urban hospitals | 1,164,802 (90.2) | 162,399 (13.9) |
| Rural hospitals | 126,002 (9.8) | 15,798 (12.5) |
| Bed size | ||
| Beds <100 | 36,624 (2.8) | 4,965 (13.6) |
| Beds 100‐300 | 623,327 (48.3) | 80,156 (12.9) |
| Beds >300 | 630,853 (48.9) | 93,076 (14.8) |
Prevalence of Acute Hypertension
A total of 763,634 (59.2%) patients had at least 1 SBP measurement of 140 mmHg during the ED stay, including 178,197 (13.8%) with SBP >180 mmHg. Body systems associated with the highest prevalence of severe AH (SBP >180 mmHg) were nervous (29.0%), circulatory (16.0%), endocrine (14.7%), and kidney/urinary (13.5%) (Figure 1 presents the data in graphic form; Supporting Appendix B, in the online version of this article, presents corresponding data in table form).

Mortality
Univariable analysis revealed a graded relationship between SBP stratum and mortality risk (Figure 2a; and see Supporting Appendix C in the online version of this article). This relationship was most pronounced for nervous system diseases; mortality rates for each 10 mmHg increase in SBP from 180 to >220 mmHg were 6.5%, 8.1%, 10.0%, 12.0%, and 19.7%, respectively (trending P < 0.0001). The risk‐adjusted increase in mortality odds ratio ranged from 1.04 (95% CI: 0.89, 1.21) to 1.44 (95% CI: 1.25, 1.67) for patients in the severe AH strata compared to patients with SBP of 101 to 139 mmHg (Figure 3). Severe AH was not an independent mortality predictor in other disease categories.


Mechanical Ventilation on Admission
Univariable analysis revealed a graded relationship between severe AH and a need for MV on admission, especially for respiratory, circulatory, and infectious conditions (trending P < 0.0001) (Figure 2b; and see Supporting Appendix C in the online version of this article). In the multivariable analysis, there was a relationship between severe AH stratum and adjusted risk for MV on admission across nearly all disease categories (Figure 4).

Length of Stay
Univariable analysis revealed a graded relationship between severe AH strata and LOS for nearly all disease categories in survivors (trending P < 0.0001), except for digestive, kidney, and infectious diseases (Figure 2c; and see Supporting Appendix C in the online version of this article). For patients with nervous system diseases, the unadjusted LOS for each 10 mmHg increase in SBP from 180 to >220 was 5.8, 6.1, 6.4, 6.8, and 8.0 days, respectively (trending P < 0.0001). The relationship was similar for other disease categories which showed significant trending.
In the multivariable analysis, the relationship between severe AH strata and adjusted attributable LOS was graded across most disease categories, especially nervous, circulatory, and hepatobiliary diseases (Figure 5). The total adjusted number of hospital days attributable to severe AH was 0.43 days per case for all survivors with severe AH.

Sensitivity Analysis
Our sensitivity analysis of the LOS estimate, including those patients who died in the hospital, yielded similar findings in attributable LOS due to severe AH. In addition, when we added patients with only 1 SBP documented in the ED to the study cohort, the severe AH prevalence changed negligibly from 13.8% to 13.0%, and the associated mortality remained unchanged at 3.4%. Models using the mix model approach, which take into account hospital‐specific effects, showed similar results.
DISCUSSION
This large‐scale analysis demonstrated that severe AH was present in 13.8% of inpatients admitted through the ED. The prevalence of severe AH varied based on the primary reason for an acute care admission, ranging from 7% in infectious syndromes to nearly 30% in acute neurologic processes. Specific to patients with neurologic disease, initial severe blood pressure elevations independently correlated with mortality. Severe blood pressure elevations at ED were independently associated with an increased need for MV on admission and a prolonged LOS across a range of disease states.
Prior work on hypertension at admission has generally included single‐center analyses or only focused on patients with specific admitting diagnoses. For example, in a single ED analysis, Tilman et al.17 reported that 16% of 10,000 patients presented with elevated blood pressure (140/90 mmHg). In a multicenter review of 7000 persons, Karras et al.18, 19 described 423 patients with severe AH (180/110 mmHg) who comprised 6% of patients seen in the ED during a 1‐week period. Qureshi et al.5 noted severe AH in 13% of patients with acute stroke. While other ED‐based studies examined all ED patients, including those admitted and discharged from the ED, our study focused on those requiring hospitalization. This disparity in illness severity may, in part, explain the higher prevalence of severe AH we noted relative to others.
We further found that the prevalence of severe AH varied based on admitting diagnosis. This difference in prevalence rates by condition seems clinically plausible. Recognition of this pattern may prove valuable to hospitalists who, by the nature of their responsibilities, will encounter a broad range of patients. Because our data were derived from the largest analysis of blood pressure assessments for ED patients who were eventually hospitalized, and encompassed a multiplicity of hospitals, our findings are likely generalizable. Moreover, our large sample size enabled us to examine severe AH at each 10 mmHg increment across a variety of disease states, rather than restricting our analysis to 1 admitting diagnosis.
The independent relationship between severe blood pressure elevation and mortality was detected only in those with neurologic conditions. Incremental increases in SBP beyond 180 mmHg were associated with a stepwise escalation in the risk for death. Although recognition of the importance of blood pressure management in both ischemic and hemorrhagic stroke remains a cornerstone of therapy for these diseases, the stepwise relationship between escalating blood pressure and outcome suggests that further study is needed to determine the optimal management of severe AH among these patients. This relationship, along with the independent association between severe AH at presentation and the need for MV, underscores the importance of severe AH in critical care, representing a major challenge for intensivists and hospitalists, particularly those who practice in neurologic ICUs.
The independent association of severe AH and prolonged LOS represents a novel finding. Few reports have correlated the initial blood pressure with measures of resource use. Katz et al.4 found a median LOS of 6 days among 1000 patients who presented with severe AH and end organ dysfunction in 25 US hospitals. These investigators, however, did not explore the incremental independent contribution of initial blood pressure to LOS. Biologically, severe hypertension may exacerbate both acute and chronic conditions, thus complicating their management and resulting in longer hospitalizations.
Our analysis has limitations. While exposure misclassification is a potential concern, unlike other population‐based studies that use ICD‐9‐CM codes to identify AH cases, we relied on actual measures of blood pressure to identify subjects, thus minimizing this threat to validity. Similarly, since our end pointsmortality, MV on admission, and LOSwere also objective measures, the probability of their misclassification is minimal.
Another concerning contributor to exposure misclassification is the possibility that, in some instances, the initial elevation in BP meeting the inclusion criteria in our study cohort does not reflect the true BP. Indeed, a substantial body of research about BP measurement in the ED suggests that we may have included some persons who likely did not have AH. For example, Pitts and Adams described a regression to the mean phenomenon, with serial BP measurements in the ED, wherein the BP fell by approximately 11 mmHg over 4 hours.20 Baumann and colleagues reported a similar pattern.21 However, the findings of these 2 analyses do not necessarily apply to our study population; we focused on patients admitted to the hospital with an acute condition, while Pitts and Adams20 and Baumann et al.21 examined all ED patients. This distinction is crucial in that patients not admitted are likely less severely ill and systematically different from those who do merit hospitalization. Moreover, both Pitts and Adams20 and Cienki et al.22 observed that the regression to the mean and fall in serial BP measurements were less pronounced in those with the most extensive BP elevations. In our study, we found the strongest relationship between adverse outcomes and BP in patients with the most extreme BP elevations. Thus, misclassification is perhaps less likely to be an issue for these subjects.
In addition, misclassification may result when initially elevated BP simply represents the impact of untreated pain or anxiety in ED patients. However, Backer et al.23 and Tanabe and colleagues24 specifically explored the impact of pain and anxiety on BP in ED subjects, and neither group found a correlation between BP and either acute pain and/or anxiety scores. Our difficulties with case definitions and BP measurements for severe AH demonstrate the need for the creation and adoption of a formal, systematic approach to this syndrome, along with the need for prospective analyses to confirm our findings.
Selection bias represents a second potential threat to validity in our observational study, although this bias is mitigated by including all consecutive acute inpatient admissions to the participating hospitals. Furthermore, inclusion of the 9% of patients who had only 1 ED measurement of SBP collected did not alter the estimate of severe AH prevalence or associated outcomes.
Third, confounding may introduce the potential for false associations derived from observational data. The large sample size of our cohort allowed us to address this concern by adjusting for a large array of confounders. In addition, unlike other large‐scale population‐based studies which typically rely on administrative ICD‐9‐CM codes for risk adjustment, our analysis incorporated actual physiologic and laboratory results measured on admission, as well as a validated severity‐of‐illness scoring system for risk adjustment.12, 13
Although both SBP and diastolic blood pressure (DBP) thresholds are included in traditional definitions of hypertension, selecting SBP as the primary measure is reasonable because SBP >180 mmHg is a more important risk factor for cardiovascular disease than elevated DBP.25 Previous studies reported the relationship between the trend of SBP over time and clinical outcomes,26, 27 but we were not able to investigate the relationship of SBP trend and outcomes because the BP measurements in the our study were not collected in predefined intervals.
It would be ideal if serial blood pressure measures were to be collected at pre‐specified intervals and if more sophisticated schemas were to be used to refine the AH definition. This type of study may be possible in the future when vital signs can be collected automatically with advanced technology. Likewise, electronically captured treatment data could further help researchers to study the impact of process‐of‐care variables, including medications and other management strategies, in relation to outcomes. Finally, outpatient management of chronic hypertension is an integral part of clinical management. Unfortunately, these types of data are not available in our existing database. These limitations notwithstanding, an in‐depth understanding of the association between severe AH and potential adverse clinical and economic outcomes may direct further research in this field.
CONCLUSION
Severe AH appears common and its prevalence varies by underlying clinical condition in patients admitted from the ED. In those with acute neurologic syndromes, the degree of blood pressure elevation correlated with mortality, need for MV, and longer LOS. For many other conditions, elevation of blood pressure appeared to be linked to an increased need for MV and a prolongation in LOS. Future studies are needed to examine the potential impact of both 1) improved long‐term outpatient BP management, and 2) optimal management of severe AH upon admission on improving outcomes of patients hospitalized from the ED with severe AH.
- Health, United States, 2010.Hyattsville, MD:Centers for Disease Control and Prevention, National Center for Health Statistics;2011.
- Agency for Healthcare Research and Quality.Healthcare Cost and Utilization Project (HCUP).Rockville, MD:US Department of Health 289(19):2560–2572.
- ,,, et al.Practice patterns, outcomes, and end‐organ dysfunction for patients with acute severe hypertension: the Studying the Treatment of Acute hyperTension (STAT) registry.Am Heart J.2009;158(4):599–606.e1.
- ,,, et al.Prevalence of elevated blood pressure in 563,704 adult patients with stroke presenting to the ED in the United States.Am J Emerg Med.2007;25(1):32–38.
- ,,, et al.A prediction rule to identify low‐risk patients with community‐acquired pneumonia.N Engl J Med.1997;336(4):243–250.
- ,.A clinical assessment of MedisGroups.JAMA.1988;260(21):3159–3163.
- ,,,,,.Healthcare‐associated bloodstream infection: a distinct entity? Insights from a large U.S. database.Crit Care Med.2006;34(10):2588–2595.
- ,,,,,.Epidemiology and outcomes of health‐care‐associated pneumonia: results from a large US database of culture‐positive pneumonia.Chest.2005;128(6):3854–3862.
- ,,,,,.Morbidity and cost burden of methicillin‐resistant Staphylococcus aureus in early onset ventilator‐associated pneumonia.Crit Care.2006;10(3):R97.
- ,,,,.Evaluation of the complication rate as a measure of quality of care in coronary artery bypass graft surgery.JAMA.1995;274(4):317–323.
- ,,.Using automated clinical data for risk adjustment: development and validation of six disease‐specific mortality predictive models for pay‐for‐performance.Med Care.2007;45(8):789–805.
- ,,,,.Development and validation of a disease‐specific risk adjustment system using automated clinical data.Health Service Research.2010;45:1815–1835.
- ,.Estimating marginal and incremental effects on health outcomes using flexible link and variance function models.Biostatistics.2005;6(1):93–109.
- ,.An Introduction to the Bootstrap.London, England:Chapman 1993.
- ,.Applied Mixed Models in Medicine.2nd ed.Chichester, England:Wiley;2006.
- ,,,,,.Recognizing asymptomatic elevated blood pressure in ED patients: how good (bad) are we?Am J Emerg Med.2007;25(3):313–317.
- ,,, et al.Evaluation and treatment of patients with severely elevated blood pressure in academic emergency departments: a multicenter study.Ann Emerg Med.2006;47(3):230–236.
- ,,, et al.Elevated blood pressure in urban emergency department patients.Acad Emerg Med.2005;12(9):835–843.
- ,.Emergency department hypertension and regression to the mean.Ann Emerg Med.1998;31(2):214–218.
- ,,,.Differing prevalence estimates of elevated blood pressure in ED patients using 4 methods of categorization.Am J Emerg Med.2008;26(5):561–565.
- ,,.The validity of emergency department triage blood pressure measurements.Acad Emerg Med.2004;11(3):237–243.
- ,,.Reproducibility of increased blood pressure during an emergency department or urgent care visit.Ann Emerg Med.2003;41(4):507–512.
- ,,,,,.Increased blood pressure in the emergency department: pain, anxiety, or undiagnosed hypertension?Ann Emerg Med.2008;51(3):221–229.
- ,,.Clinical Advisory Statement. Importance of systolic blood pressure in older Americans.Hypertension.2000;35(5):1021–1024.
- ,,,.High blood pressure in early acute stroke: a sign of a poor outcome?J Hypertens.2006;24(2):381–386.
- ,,,,.Blood pressure as an independent prognostic factor in acute ischemic stroke.Can J Neurol Sci.2006;33(1):34–38.
- Health, United States, 2010.Hyattsville, MD:Centers for Disease Control and Prevention, National Center for Health Statistics;2011.
- Agency for Healthcare Research and Quality.Healthcare Cost and Utilization Project (HCUP).Rockville, MD:US Department of Health 289(19):2560–2572.
- ,,, et al.Practice patterns, outcomes, and end‐organ dysfunction for patients with acute severe hypertension: the Studying the Treatment of Acute hyperTension (STAT) registry.Am Heart J.2009;158(4):599–606.e1.
- ,,, et al.Prevalence of elevated blood pressure in 563,704 adult patients with stroke presenting to the ED in the United States.Am J Emerg Med.2007;25(1):32–38.
- ,,, et al.A prediction rule to identify low‐risk patients with community‐acquired pneumonia.N Engl J Med.1997;336(4):243–250.
- ,.A clinical assessment of MedisGroups.JAMA.1988;260(21):3159–3163.
- ,,,,,.Healthcare‐associated bloodstream infection: a distinct entity? Insights from a large U.S. database.Crit Care Med.2006;34(10):2588–2595.
- ,,,,,.Epidemiology and outcomes of health‐care‐associated pneumonia: results from a large US database of culture‐positive pneumonia.Chest.2005;128(6):3854–3862.
- ,,,,,.Morbidity and cost burden of methicillin‐resistant Staphylococcus aureus in early onset ventilator‐associated pneumonia.Crit Care.2006;10(3):R97.
- ,,,,.Evaluation of the complication rate as a measure of quality of care in coronary artery bypass graft surgery.JAMA.1995;274(4):317–323.
- ,,.Using automated clinical data for risk adjustment: development and validation of six disease‐specific mortality predictive models for pay‐for‐performance.Med Care.2007;45(8):789–805.
- ,,,,.Development and validation of a disease‐specific risk adjustment system using automated clinical data.Health Service Research.2010;45:1815–1835.
- ,.Estimating marginal and incremental effects on health outcomes using flexible link and variance function models.Biostatistics.2005;6(1):93–109.
- ,.An Introduction to the Bootstrap.London, England:Chapman 1993.
- ,.Applied Mixed Models in Medicine.2nd ed.Chichester, England:Wiley;2006.
- ,,,,,.Recognizing asymptomatic elevated blood pressure in ED patients: how good (bad) are we?Am J Emerg Med.2007;25(3):313–317.
- ,,, et al.Evaluation and treatment of patients with severely elevated blood pressure in academic emergency departments: a multicenter study.Ann Emerg Med.2006;47(3):230–236.
- ,,, et al.Elevated blood pressure in urban emergency department patients.Acad Emerg Med.2005;12(9):835–843.
- ,.Emergency department hypertension and regression to the mean.Ann Emerg Med.1998;31(2):214–218.
- ,,,.Differing prevalence estimates of elevated blood pressure in ED patients using 4 methods of categorization.Am J Emerg Med.2008;26(5):561–565.
- ,,.The validity of emergency department triage blood pressure measurements.Acad Emerg Med.2004;11(3):237–243.
- ,,.Reproducibility of increased blood pressure during an emergency department or urgent care visit.Ann Emerg Med.2003;41(4):507–512.
- ,,,,,.Increased blood pressure in the emergency department: pain, anxiety, or undiagnosed hypertension?Ann Emerg Med.2008;51(3):221–229.
- ,,.Clinical Advisory Statement. Importance of systolic blood pressure in older Americans.Hypertension.2000;35(5):1021–1024.
- ,,,.High blood pressure in early acute stroke: a sign of a poor outcome?J Hypertens.2006;24(2):381–386.
- ,,,,.Blood pressure as an independent prognostic factor in acute ischemic stroke.Can J Neurol Sci.2006;33(1):34–38.
Copyright © 2011 Society of Hospital Medicine
Inpatient Staffing in Pediatric Programs
Resident duty hour restrictions were initially implemented in New York in 1989 with New York State Code 405 in response to a patient death in a New York City Emergency Department.1 This case initiated an evaluation of potential risks to patient safety when residents were inadequately supervised and overfatigued. In 2003, the Accreditation Council for Graduate Medical Education (ACGME) implemented resident duty hours nationally due to concerns for patient safety and quality of care.2 These restrictions involved the implementation of the 80‐hour work week (averaged over 4 weeks), a maximum duty length of 30 hours, and prescriptive supervision guidelines. In December 2008, the Institute of Medicine (IOM) proposed additional changes to further restrict resident duty hours which also included overnight protected sleep periods and additional days off per month.3 The ACGME responded by mandating new resident duty hour restrictions in October 2010 which will be implemented in July 2011. The ACGME's new changes include a change in the maximum duty hour length for residents in their first year of training (PGY‐1) of 16 hours. Residents in their second year of training (PGY‐2) level and above may work a maximum of 24 hours with an additional 4 hours for transition of care and resident education. The ACGME strongly recommends strategic napping, but do not have a protected overnight sleep period in place4 (Table 1).
| Current Guidelines | IOM Proposed Changes | ACGME Mandated Changes | |
|---|---|---|---|
| December 2008 | October 2010 | ||
| |||
| Maximum hours of work per week | 80 hr averaged over 4 wk | 80 hr averaged over 4 wk | 80 hr averaged over 4 wk |
| Maximum duty length | 30 hr (admitting patients for up to 24 hr, then additional 6 hr for transition of care) | 30 hr with 5 hr protected sleep period (admitting patients for up to 16 hr) | PGY‐1 residents, 16 hr |
| Or | PGY‐2 residents, 24 hr with additional 4 hr for transition of care | ||
| 16 hr with no protected sleep period | |||
| Strategic napping | None | 5 hr protected sleep period for 30 hr shifts | Highly recommended after 16 hr of continuous duty |
| Time off between duty periods | 10 hr after shift | 10 hr after day shift | Recommend 10 hr, but must have at least 8 hr off |
| 12 hr after night shift | In their final years, residents can have less than 8 hr | ||
| 14 hr after 30 hr shifts | |||
| Maximum consecutive nights of night float | None | 4 consecutive nights maximum | 6 consecutive nights maximum |
| Frequency of in‐house call | Every third night, on average | Every third night, no averaging | Every third night, no averaging |
| Days off per month | 4 days off | 5 days off, at least one 48 hr period per month | 4 days off |
| Moonlighting restrictions | Internal moonlighting counts against 80 hr cap | Both internal and external moonlighting count against 80 hr cap | Both internal and external moonlighting count against 80 hr cap |
There is growing concern regarding the impact of these new resident duty hour restrictions on the coverage of inpatient services, particularly during the overnight period. To our knowledge, there is no published national data on how pediatric inpatient teaching services are staffed at night. The objective of this study was to survey the current landscape of pediatric resident coverage of noncritical care inpatient teaching services. In addition, we sought to explore how changes in work hour restrictions might affect the role of pediatric hospitalists in training programs.
METHODS
We developed an institutional review board (IRB)‐approved Web‐based electronic survey. The survey consisted of 17 questions. The survey obtained information regarding the demographics of the program including: number of residents, daily patient census per ward intern, information regarding staff‐only pediatric ward services, overnight coverage, and current attending in‐house overnight coverage (see Appendix). We also examined the prevalence of pediatric hospitalists in training programs, their current role in staffing patients, and how that role may change with the implementation of additional resident duty hour restrictions. Initially, the survey was reviewed and tested by several pediatric hospitalists and program directors. It was then reviewed and approved by the Association of Pediatric Program Director (APPD) research task force. The survey was sent out to 196 US pediatric residency programs via the APPD listserve in January 2010. Program directors were given the option of completing it themselves or specifically designating someone else to complete it. Two reminders were sent. We then sent an additional request for program participation on the pediatric hospitalist listserve. All data was collected by February 2010.
RESULTS
One hundred twenty unique responses were received (61% of total pediatric residency programs). As of 2009, this represented 5201 pediatric residents (58% of total pediatric residents). The average program size was 43 residents (range: 12‐156 residents, median 43). The average daily patient census per ward intern during daytime hours was 6.65 patients (range: 3‐17, median 6). Twenty percent of training programs had staff‐only (no residents) pediatric ward services during daytime hours. In the programs with both staff‐only and resident pediatric ward services, only 19% of patients were covered by the staff‐only teams and 81% of patients were covered by resident teams.
During the overnight period, 86% of resident teams did not have caps on the number of new patient admissions. An average of 3.6 providers per training program were in‐house overnight to accept patient admissions to pediatric wards. Ninety‐four percent of these providers in‐house were residents (399 residents in‐house/425 total providers in‐house each night).
Twenty‐five percent of the training programs that responded to the survey had pediatric hospitalist attendings in‐house at night. This included both overnight and partial nights (ie, until midnight). Other attendings in‐house at night include: neonatal intensive care unit (NICU) attendings (53% of programs), pediatric intensive care unit (PICU) attendings (46% of programs), Pediatric Emergency Medicine attendings (65% of programs), and Pediatric Surgery attendings (6.4% of programs). Twenty‐two percent of programs had no in‐house attendings at night (Figure 1).
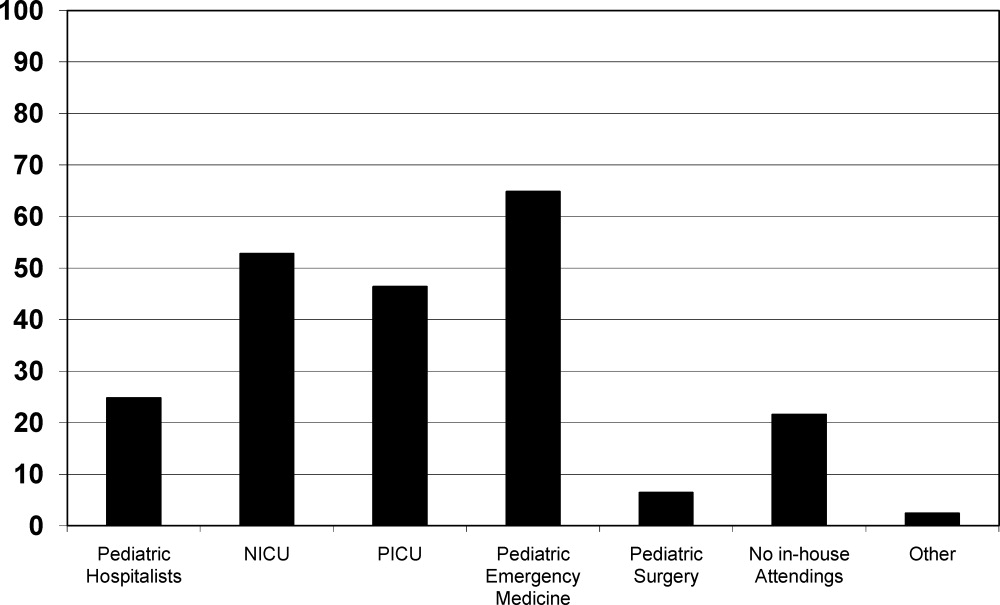
Pediatric hospitalists were involved with 84% (n = 97) of training programs. Sixty percent (n = 58) of the pediatric hospitalist teams were staffed with both teaching attendings and residents. Fourteen percent (n = 14) of the pediatric hospitalist teams did not involve residents (staff‐only) and 25% (n = 25) had both types of teams. Specifically, of the programs that had pediatric hospitalists, 20% (n = 19) of them had hospitalist attendings in‐house 24 hours per day and 13% (n = 12) of teams had hospitalist attendings in‐house into the evening hours for a varying amount of time. Of the programs with hospitalist attendings in‐house 24 hours per day, 52% (n = 11) had started this coverage within the past 3 years.
Looking towards the future, and prior to the enactment of the October 2010 ACGME standards, 31% (n = 35) of the training programs that lacked 24/7 hospitalist in‐house coverage in January 2010 anticipated adding this level of coverage within the next 5 years. Notably, 70% (n = 81) of training programs felt that further resident work hour restrictions, which have since been enacted, would likely require the addition of more hospitalist attendings at night. Our survey allowed program directors to make open‐ended comments on how further work hour restrictions may change inpatient staffing in noncritical care inpatient teaching services.
DISCUSSION
To our knowledge, this was the first national study of pediatric resident coverage in noncritical care inpatient teaching services. While there was significant variation in how inpatient teaching services were covered across these programs, in January 2010, residents were involved in the majority of patient care with only 20% of programs having attending‐only hospitalist teams during the daytime. During the overnight period, the proportion of patient care provided by residents became even more significant with residents representing 94% of the total in‐house providers accepting new admissions. While pediatric hospitalists were prevalent at these training programs, their role in direct patient care overnight was limited. Only 6% of total in‐house providers accepting admissions at night were pediatric hospitalists.
The comments made by program directors are representative of the overall concerns regarding changes to resident work hours (see Table 2). In a position statement by the Association of Pediatric Program Directors in regards to the IOM recommendations, concerns were raised stating that the recommendations of the IOM Committee are intended to enhance patient safety without appropriate consideration for the educational and professional development of trainees.5 While the newly mandated ACGME standards are different than the previous IOM recommendations, it is clear that there will be very significant changes to accommodate these new standards. Our study was done prior to the new ACGME's standards. At the time of the survey, less than a third of programs were anticipating the addition of 24/7 pediatric hospitalist coverage; however, if resident work hours were further restricted, 70% of programs felt that additional hospitalists would be needed. This is a significant increase in the previously anticipated need for overnight attending hospitalist coverage, especially in light of the further restrictions mandated by the ACGME. We know that the response of New York State programs to the 405 regulations varied by program size, but all made significant changes to accommodate the new standards.6 It is clear that many program directors nationally are anticipating significant changes to their residencies when these new restrictions are enacted. The respondents in our survey felt that pediatric hospitalists are going to have to play an even bigger role at night when additional resident work hour restrictions are put into place.
|
| ▪ If the new duty hours are mandated, we would have to go to a night float system to be in compliance. This would require more residents and we do not have the funding to hire more residents. |
| ▪ Restrictions will be costly. It will increase shift work mentality, and increase pt errors due to handovers. If these (work restrictions) are not applied to all doctors (neurosurgeons, ICU doctors), they should not apply to resident doctors. |
| ▪ The additional restrictions may make the hospital consider giving up its residency program in favor of a hospitalist‐only model. |
| ▪ We do not have enough residents to care for the current patient load. |
| ▪ Additional work hour restrictions will lead to more hand‐over care and less ownership of patients by residents who identify themselves as primary patient physicians. Both situations are associated with increased rates of complications and possible sentinel events. |
| ▪ If the hours are reduced, the hospital will be forced to hire physicians for the care of patients. The administration of the hospital is now beginning to ask why they should financially support the training program if the residents are not providing a substantial portion of the hospital care for the patients. |
Pediatric hospital medicine remains a rapidly growing field.7 Eighty‐four percent of pediatric training programs utilize pediatric hospitalists. Over 60% of these pediatric hospitalist teams are involved in teaching teams with residents. While we did not directly study the supply and demand of pediatric hospitalists, there is some concern that even despite its rapid growth, the supply of pediatric hospitalists will not keep up with the demand when further resident work hours restrictions are implemented. At time of submission, a cost‐analysis has not yet been publicly published on the ACGME's new changes. There is data available based on the IOM's 2008 recommendations. A study by Nuckols and Escarce8 suggests that if the IOM's recommendations were implemented, the entire healthcare system nationally would have to develop and fill new full‐time positions equal to 5001 attending physicians, 5984 midlevel providers (nurse practitioners or physician assistants), 320 licensed vocational nurses, 229 nursing aides, and 45 laboratory technicians. This would be equivalent to adding an additional 8247 residency positions across all specialties.810 While the ACGME's new mandated changes are different than the IOM's recommendations, they will also restrict resident duty hours that we believe could lead to gaps in patient care requiring significant personnel changes in the healthcare system.
There are several limitations to our study. We did not study the role of pediatric subspecialty fellows and their involvement in pediatric inpatient services in these training programs. We also did not study the prevalence and use of resident night float systems. While night floats may be used in some programs, it may become more prevalent with the possible restriction in intern work hours down to 16 hours. As with any survey, there remains both volunteer and nonresponse bias with the programs that decide to complete or disregard the survey. Finally, there remains some concern over the data collection after the survey was sent out to the hospitalist listserve. Pediatric hospitalists may have incorrectly filled out the data for their program after their program director had already completed the survey. We attempted to minimize this problem by specifically instructing hospitalists to encourage their program director to fill out the survey if they had not already done so. We also compared computer Internet Protocol (IP) addresses and actual program responses, before and after the hospitalist e‐mail was sent, in an attempt to minimize the chance of including duplicated responses from the same program. Lastly, the January 2010 survey predated the October 2010 ACGME response to the IOM recommendations, and the responses may be different now that the specific restrictions have been mandated with an actual implementation date.
CONCLUSIONS
This study shows that pediatric teaching services varied significantly in how they provided overnight coverage in 2010 prior to new ACGME recommendations. Overall, residents were providing the overwhelming majority of the patient care overnight in pediatric training programs. While hospitalists were prevalent in pediatric training programs, in 2010 they had limited roles in direct patient care at night. The ACGME has now mandated additional residency work hour restrictions to be implemented July 2011. With these restrictions, hospitalists will likely need to expand their services, and additional hospitalists will be needed to provide overnight coverage. It is unclear where those hospitalists will come from and what their role will be. It is also unclear what the impact of increased demand and changed job description will be on the continued evolution of the field of Pediatric Hospital Medicine.
Future work needs to be done to establish benchmarks for inpatient coverage. The benchmarks could include guidelines on balancing patient safety with resident education. This may also involve the implementation of resident night float models. There needs to be monitoring on how changes in resident work hours and staffing affect coverage and, ultimately, how changes affect patient and resident outcomes.
APPENDIX
INPATIENT STAFFING WITHIN PEDIATRIC RESIDENCY PROGRAMS SURVEY
|
| Demographics |
| How many residents are in your residency program? (total, categorical, Med‐Peds, other combined Peds) |
| What is your average daily patient census per ward intern during daytime hours? |
| Does your hospital have a staff‐only (no residents) pediatric ward service during the daytime hours? |
| If your hospital has a staff‐only pediatric ward service, what are the proportion of patients cared for by residents vs staff‐only during daytime hours? |
| Do your residents cap the number of new patient admissions at night? |
| Providers in‐house overnight |
| How many providers do you have in‐house at night until midnight/overnight to accept patient admissions to pediatric wards? (residents, hospitalists, nurse practitioners, other) |
| Do you have attendings in‐house at night? (pediatric hospitalists, NICU, PICU, Peds EM, Peds Surgery, no attendings, other) |
| Pediatric hospitalists |
| Does your hospital have pediatric hospitalists? |
| Are your pediatric hospitalist teams staffed by: (teaching attendings and residents, hospitalist‐staff only, both) |
| If you have a staff‐only hospitalist team (no residents), how long has it been in existence? (less than 1 year, 1‐3 years, 4‐10 years, over 10 years) |
| Are your hospitalist attendings in‐house: (daytime only, 24 hours/day, other) |
| If your hospitalist attendings are in‐house 24/7, how many years has that coverage been available? (less than 1 year, 1‐3 years, 4‐10 years, over 10 years, not available) |
| Future pediatric hospitalist coverage |
| Do you anticipate that your hospital will be adding 24/7 hospitalist attending coverage? (next year, next 2 years, next 5 years, not anticipating adding coverage, 24/7 hospitalist coverage already in place) |
| In your opinion, would further resident work hour restrictions make your hospital more likely to add additional hospitalist attendings at night? (very likely, somewhat likely, neutral, not likely) |
- ,.The Bell Commission: ethical implications for the training of physicians.Mt Sinai J Med.2000;67(2):136–139.
- ,.Restricted duty hours for surgeons and impact on residents quality of life, education, and patient care: a literature review.Patient Saf Surg.2009;3(1):3.
- Institute of Medicine. Resident Duty Hours: Enhancing Sleep, Supervision, and Safety. Released December 02, 2008. Available at: http://www.iom.edu/Reports/2008/Resident‐Duty‐Hours‐Enhancing‐Sleep‐Supervision‐and‐Safety.aspx. Accessed September 20,2009.
- ACGME 2010 Standards “Common Program Requirements.” Available at: http://acgme‐2010standards.org/pdf/Common_Program_ Requirements_07012011.pdf. Accessed January 27,2011.
- Association of Pediatric Program Directors. Association of Pediatric Program Directors (APPD) Position Statement in Response to the IOM Recommendations on Resident Duty Hours.2009. Available at: http://www.appd.org/PDFs/APPD _IOM%20 _Duty _Hours _Report _Position _Paper _4–30‐09.pdf. Accessed March 27, 2010.
- ,,,,.Lessons learned from New York state: fourteen years of experience with work hour limitations.Acad Med.2005;80(5):467–472.
- ,,,.Health care market trends and the evolution of hospitalist use and rolesJ Gen Intern Med.2005;20(2):101–107.
- ,,,,.Cost implications of reduced work hours and workloads for resident physicians.N Engl J Med.2009;360:2202–2215.
- .Revisiting duty‐hour length—IOM recommendations for patient safety and resident education.N Engl J Med.2008;359:2633–2635.
- ,,,.Resident duty hour restrictions: is less really more?J Pediatr.2009;154:631–632.
Resident duty hour restrictions were initially implemented in New York in 1989 with New York State Code 405 in response to a patient death in a New York City Emergency Department.1 This case initiated an evaluation of potential risks to patient safety when residents were inadequately supervised and overfatigued. In 2003, the Accreditation Council for Graduate Medical Education (ACGME) implemented resident duty hours nationally due to concerns for patient safety and quality of care.2 These restrictions involved the implementation of the 80‐hour work week (averaged over 4 weeks), a maximum duty length of 30 hours, and prescriptive supervision guidelines. In December 2008, the Institute of Medicine (IOM) proposed additional changes to further restrict resident duty hours which also included overnight protected sleep periods and additional days off per month.3 The ACGME responded by mandating new resident duty hour restrictions in October 2010 which will be implemented in July 2011. The ACGME's new changes include a change in the maximum duty hour length for residents in their first year of training (PGY‐1) of 16 hours. Residents in their second year of training (PGY‐2) level and above may work a maximum of 24 hours with an additional 4 hours for transition of care and resident education. The ACGME strongly recommends strategic napping, but do not have a protected overnight sleep period in place4 (Table 1).
| Current Guidelines | IOM Proposed Changes | ACGME Mandated Changes | |
|---|---|---|---|
| December 2008 | October 2010 | ||
| |||
| Maximum hours of work per week | 80 hr averaged over 4 wk | 80 hr averaged over 4 wk | 80 hr averaged over 4 wk |
| Maximum duty length | 30 hr (admitting patients for up to 24 hr, then additional 6 hr for transition of care) | 30 hr with 5 hr protected sleep period (admitting patients for up to 16 hr) | PGY‐1 residents, 16 hr |
| Or | PGY‐2 residents, 24 hr with additional 4 hr for transition of care | ||
| 16 hr with no protected sleep period | |||
| Strategic napping | None | 5 hr protected sleep period for 30 hr shifts | Highly recommended after 16 hr of continuous duty |
| Time off between duty periods | 10 hr after shift | 10 hr after day shift | Recommend 10 hr, but must have at least 8 hr off |
| 12 hr after night shift | In their final years, residents can have less than 8 hr | ||
| 14 hr after 30 hr shifts | |||
| Maximum consecutive nights of night float | None | 4 consecutive nights maximum | 6 consecutive nights maximum |
| Frequency of in‐house call | Every third night, on average | Every third night, no averaging | Every third night, no averaging |
| Days off per month | 4 days off | 5 days off, at least one 48 hr period per month | 4 days off |
| Moonlighting restrictions | Internal moonlighting counts against 80 hr cap | Both internal and external moonlighting count against 80 hr cap | Both internal and external moonlighting count against 80 hr cap |
There is growing concern regarding the impact of these new resident duty hour restrictions on the coverage of inpatient services, particularly during the overnight period. To our knowledge, there is no published national data on how pediatric inpatient teaching services are staffed at night. The objective of this study was to survey the current landscape of pediatric resident coverage of noncritical care inpatient teaching services. In addition, we sought to explore how changes in work hour restrictions might affect the role of pediatric hospitalists in training programs.
METHODS
We developed an institutional review board (IRB)‐approved Web‐based electronic survey. The survey consisted of 17 questions. The survey obtained information regarding the demographics of the program including: number of residents, daily patient census per ward intern, information regarding staff‐only pediatric ward services, overnight coverage, and current attending in‐house overnight coverage (see Appendix). We also examined the prevalence of pediatric hospitalists in training programs, their current role in staffing patients, and how that role may change with the implementation of additional resident duty hour restrictions. Initially, the survey was reviewed and tested by several pediatric hospitalists and program directors. It was then reviewed and approved by the Association of Pediatric Program Director (APPD) research task force. The survey was sent out to 196 US pediatric residency programs via the APPD listserve in January 2010. Program directors were given the option of completing it themselves or specifically designating someone else to complete it. Two reminders were sent. We then sent an additional request for program participation on the pediatric hospitalist listserve. All data was collected by February 2010.
RESULTS
One hundred twenty unique responses were received (61% of total pediatric residency programs). As of 2009, this represented 5201 pediatric residents (58% of total pediatric residents). The average program size was 43 residents (range: 12‐156 residents, median 43). The average daily patient census per ward intern during daytime hours was 6.65 patients (range: 3‐17, median 6). Twenty percent of training programs had staff‐only (no residents) pediatric ward services during daytime hours. In the programs with both staff‐only and resident pediatric ward services, only 19% of patients were covered by the staff‐only teams and 81% of patients were covered by resident teams.
During the overnight period, 86% of resident teams did not have caps on the number of new patient admissions. An average of 3.6 providers per training program were in‐house overnight to accept patient admissions to pediatric wards. Ninety‐four percent of these providers in‐house were residents (399 residents in‐house/425 total providers in‐house each night).
Twenty‐five percent of the training programs that responded to the survey had pediatric hospitalist attendings in‐house at night. This included both overnight and partial nights (ie, until midnight). Other attendings in‐house at night include: neonatal intensive care unit (NICU) attendings (53% of programs), pediatric intensive care unit (PICU) attendings (46% of programs), Pediatric Emergency Medicine attendings (65% of programs), and Pediatric Surgery attendings (6.4% of programs). Twenty‐two percent of programs had no in‐house attendings at night (Figure 1).

Pediatric hospitalists were involved with 84% (n = 97) of training programs. Sixty percent (n = 58) of the pediatric hospitalist teams were staffed with both teaching attendings and residents. Fourteen percent (n = 14) of the pediatric hospitalist teams did not involve residents (staff‐only) and 25% (n = 25) had both types of teams. Specifically, of the programs that had pediatric hospitalists, 20% (n = 19) of them had hospitalist attendings in‐house 24 hours per day and 13% (n = 12) of teams had hospitalist attendings in‐house into the evening hours for a varying amount of time. Of the programs with hospitalist attendings in‐house 24 hours per day, 52% (n = 11) had started this coverage within the past 3 years.
Looking towards the future, and prior to the enactment of the October 2010 ACGME standards, 31% (n = 35) of the training programs that lacked 24/7 hospitalist in‐house coverage in January 2010 anticipated adding this level of coverage within the next 5 years. Notably, 70% (n = 81) of training programs felt that further resident work hour restrictions, which have since been enacted, would likely require the addition of more hospitalist attendings at night. Our survey allowed program directors to make open‐ended comments on how further work hour restrictions may change inpatient staffing in noncritical care inpatient teaching services.
DISCUSSION
To our knowledge, this was the first national study of pediatric resident coverage in noncritical care inpatient teaching services. While there was significant variation in how inpatient teaching services were covered across these programs, in January 2010, residents were involved in the majority of patient care with only 20% of programs having attending‐only hospitalist teams during the daytime. During the overnight period, the proportion of patient care provided by residents became even more significant with residents representing 94% of the total in‐house providers accepting new admissions. While pediatric hospitalists were prevalent at these training programs, their role in direct patient care overnight was limited. Only 6% of total in‐house providers accepting admissions at night were pediatric hospitalists.
The comments made by program directors are representative of the overall concerns regarding changes to resident work hours (see Table 2). In a position statement by the Association of Pediatric Program Directors in regards to the IOM recommendations, concerns were raised stating that the recommendations of the IOM Committee are intended to enhance patient safety without appropriate consideration for the educational and professional development of trainees.5 While the newly mandated ACGME standards are different than the previous IOM recommendations, it is clear that there will be very significant changes to accommodate these new standards. Our study was done prior to the new ACGME's standards. At the time of the survey, less than a third of programs were anticipating the addition of 24/7 pediatric hospitalist coverage; however, if resident work hours were further restricted, 70% of programs felt that additional hospitalists would be needed. This is a significant increase in the previously anticipated need for overnight attending hospitalist coverage, especially in light of the further restrictions mandated by the ACGME. We know that the response of New York State programs to the 405 regulations varied by program size, but all made significant changes to accommodate the new standards.6 It is clear that many program directors nationally are anticipating significant changes to their residencies when these new restrictions are enacted. The respondents in our survey felt that pediatric hospitalists are going to have to play an even bigger role at night when additional resident work hour restrictions are put into place.
|
| ▪ If the new duty hours are mandated, we would have to go to a night float system to be in compliance. This would require more residents and we do not have the funding to hire more residents. |
| ▪ Restrictions will be costly. It will increase shift work mentality, and increase pt errors due to handovers. If these (work restrictions) are not applied to all doctors (neurosurgeons, ICU doctors), they should not apply to resident doctors. |
| ▪ The additional restrictions may make the hospital consider giving up its residency program in favor of a hospitalist‐only model. |
| ▪ We do not have enough residents to care for the current patient load. |
| ▪ Additional work hour restrictions will lead to more hand‐over care and less ownership of patients by residents who identify themselves as primary patient physicians. Both situations are associated with increased rates of complications and possible sentinel events. |
| ▪ If the hours are reduced, the hospital will be forced to hire physicians for the care of patients. The administration of the hospital is now beginning to ask why they should financially support the training program if the residents are not providing a substantial portion of the hospital care for the patients. |
Pediatric hospital medicine remains a rapidly growing field.7 Eighty‐four percent of pediatric training programs utilize pediatric hospitalists. Over 60% of these pediatric hospitalist teams are involved in teaching teams with residents. While we did not directly study the supply and demand of pediatric hospitalists, there is some concern that even despite its rapid growth, the supply of pediatric hospitalists will not keep up with the demand when further resident work hours restrictions are implemented. At time of submission, a cost‐analysis has not yet been publicly published on the ACGME's new changes. There is data available based on the IOM's 2008 recommendations. A study by Nuckols and Escarce8 suggests that if the IOM's recommendations were implemented, the entire healthcare system nationally would have to develop and fill new full‐time positions equal to 5001 attending physicians, 5984 midlevel providers (nurse practitioners or physician assistants), 320 licensed vocational nurses, 229 nursing aides, and 45 laboratory technicians. This would be equivalent to adding an additional 8247 residency positions across all specialties.810 While the ACGME's new mandated changes are different than the IOM's recommendations, they will also restrict resident duty hours that we believe could lead to gaps in patient care requiring significant personnel changes in the healthcare system.
There are several limitations to our study. We did not study the role of pediatric subspecialty fellows and their involvement in pediatric inpatient services in these training programs. We also did not study the prevalence and use of resident night float systems. While night floats may be used in some programs, it may become more prevalent with the possible restriction in intern work hours down to 16 hours. As with any survey, there remains both volunteer and nonresponse bias with the programs that decide to complete or disregard the survey. Finally, there remains some concern over the data collection after the survey was sent out to the hospitalist listserve. Pediatric hospitalists may have incorrectly filled out the data for their program after their program director had already completed the survey. We attempted to minimize this problem by specifically instructing hospitalists to encourage their program director to fill out the survey if they had not already done so. We also compared computer Internet Protocol (IP) addresses and actual program responses, before and after the hospitalist e‐mail was sent, in an attempt to minimize the chance of including duplicated responses from the same program. Lastly, the January 2010 survey predated the October 2010 ACGME response to the IOM recommendations, and the responses may be different now that the specific restrictions have been mandated with an actual implementation date.
CONCLUSIONS
This study shows that pediatric teaching services varied significantly in how they provided overnight coverage in 2010 prior to new ACGME recommendations. Overall, residents were providing the overwhelming majority of the patient care overnight in pediatric training programs. While hospitalists were prevalent in pediatric training programs, in 2010 they had limited roles in direct patient care at night. The ACGME has now mandated additional residency work hour restrictions to be implemented July 2011. With these restrictions, hospitalists will likely need to expand their services, and additional hospitalists will be needed to provide overnight coverage. It is unclear where those hospitalists will come from and what their role will be. It is also unclear what the impact of increased demand and changed job description will be on the continued evolution of the field of Pediatric Hospital Medicine.
Future work needs to be done to establish benchmarks for inpatient coverage. The benchmarks could include guidelines on balancing patient safety with resident education. This may also involve the implementation of resident night float models. There needs to be monitoring on how changes in resident work hours and staffing affect coverage and, ultimately, how changes affect patient and resident outcomes.
APPENDIX
INPATIENT STAFFING WITHIN PEDIATRIC RESIDENCY PROGRAMS SURVEY
|
| Demographics |
| How many residents are in your residency program? (total, categorical, Med‐Peds, other combined Peds) |
| What is your average daily patient census per ward intern during daytime hours? |
| Does your hospital have a staff‐only (no residents) pediatric ward service during the daytime hours? |
| If your hospital has a staff‐only pediatric ward service, what are the proportion of patients cared for by residents vs staff‐only during daytime hours? |
| Do your residents cap the number of new patient admissions at night? |
| Providers in‐house overnight |
| How many providers do you have in‐house at night until midnight/overnight to accept patient admissions to pediatric wards? (residents, hospitalists, nurse practitioners, other) |
| Do you have attendings in‐house at night? (pediatric hospitalists, NICU, PICU, Peds EM, Peds Surgery, no attendings, other) |
| Pediatric hospitalists |
| Does your hospital have pediatric hospitalists? |
| Are your pediatric hospitalist teams staffed by: (teaching attendings and residents, hospitalist‐staff only, both) |
| If you have a staff‐only hospitalist team (no residents), how long has it been in existence? (less than 1 year, 1‐3 years, 4‐10 years, over 10 years) |
| Are your hospitalist attendings in‐house: (daytime only, 24 hours/day, other) |
| If your hospitalist attendings are in‐house 24/7, how many years has that coverage been available? (less than 1 year, 1‐3 years, 4‐10 years, over 10 years, not available) |
| Future pediatric hospitalist coverage |
| Do you anticipate that your hospital will be adding 24/7 hospitalist attending coverage? (next year, next 2 years, next 5 years, not anticipating adding coverage, 24/7 hospitalist coverage already in place) |
| In your opinion, would further resident work hour restrictions make your hospital more likely to add additional hospitalist attendings at night? (very likely, somewhat likely, neutral, not likely) |
Resident duty hour restrictions were initially implemented in New York in 1989 with New York State Code 405 in response to a patient death in a New York City Emergency Department.1 This case initiated an evaluation of potential risks to patient safety when residents were inadequately supervised and overfatigued. In 2003, the Accreditation Council for Graduate Medical Education (ACGME) implemented resident duty hours nationally due to concerns for patient safety and quality of care.2 These restrictions involved the implementation of the 80‐hour work week (averaged over 4 weeks), a maximum duty length of 30 hours, and prescriptive supervision guidelines. In December 2008, the Institute of Medicine (IOM) proposed additional changes to further restrict resident duty hours which also included overnight protected sleep periods and additional days off per month.3 The ACGME responded by mandating new resident duty hour restrictions in October 2010 which will be implemented in July 2011. The ACGME's new changes include a change in the maximum duty hour length for residents in their first year of training (PGY‐1) of 16 hours. Residents in their second year of training (PGY‐2) level and above may work a maximum of 24 hours with an additional 4 hours for transition of care and resident education. The ACGME strongly recommends strategic napping, but do not have a protected overnight sleep period in place4 (Table 1).
| Current Guidelines | IOM Proposed Changes | ACGME Mandated Changes | |
|---|---|---|---|
| December 2008 | October 2010 | ||
| |||
| Maximum hours of work per week | 80 hr averaged over 4 wk | 80 hr averaged over 4 wk | 80 hr averaged over 4 wk |
| Maximum duty length | 30 hr (admitting patients for up to 24 hr, then additional 6 hr for transition of care) | 30 hr with 5 hr protected sleep period (admitting patients for up to 16 hr) | PGY‐1 residents, 16 hr |
| Or | PGY‐2 residents, 24 hr with additional 4 hr for transition of care | ||
| 16 hr with no protected sleep period | |||
| Strategic napping | None | 5 hr protected sleep period for 30 hr shifts | Highly recommended after 16 hr of continuous duty |
| Time off between duty periods | 10 hr after shift | 10 hr after day shift | Recommend 10 hr, but must have at least 8 hr off |
| 12 hr after night shift | In their final years, residents can have less than 8 hr | ||
| 14 hr after 30 hr shifts | |||
| Maximum consecutive nights of night float | None | 4 consecutive nights maximum | 6 consecutive nights maximum |
| Frequency of in‐house call | Every third night, on average | Every third night, no averaging | Every third night, no averaging |
| Days off per month | 4 days off | 5 days off, at least one 48 hr period per month | 4 days off |
| Moonlighting restrictions | Internal moonlighting counts against 80 hr cap | Both internal and external moonlighting count against 80 hr cap | Both internal and external moonlighting count against 80 hr cap |
There is growing concern regarding the impact of these new resident duty hour restrictions on the coverage of inpatient services, particularly during the overnight period. To our knowledge, there is no published national data on how pediatric inpatient teaching services are staffed at night. The objective of this study was to survey the current landscape of pediatric resident coverage of noncritical care inpatient teaching services. In addition, we sought to explore how changes in work hour restrictions might affect the role of pediatric hospitalists in training programs.
METHODS
We developed an institutional review board (IRB)‐approved Web‐based electronic survey. The survey consisted of 17 questions. The survey obtained information regarding the demographics of the program including: number of residents, daily patient census per ward intern, information regarding staff‐only pediatric ward services, overnight coverage, and current attending in‐house overnight coverage (see Appendix). We also examined the prevalence of pediatric hospitalists in training programs, their current role in staffing patients, and how that role may change with the implementation of additional resident duty hour restrictions. Initially, the survey was reviewed and tested by several pediatric hospitalists and program directors. It was then reviewed and approved by the Association of Pediatric Program Director (APPD) research task force. The survey was sent out to 196 US pediatric residency programs via the APPD listserve in January 2010. Program directors were given the option of completing it themselves or specifically designating someone else to complete it. Two reminders were sent. We then sent an additional request for program participation on the pediatric hospitalist listserve. All data was collected by February 2010.
RESULTS
One hundred twenty unique responses were received (61% of total pediatric residency programs). As of 2009, this represented 5201 pediatric residents (58% of total pediatric residents). The average program size was 43 residents (range: 12‐156 residents, median 43). The average daily patient census per ward intern during daytime hours was 6.65 patients (range: 3‐17, median 6). Twenty percent of training programs had staff‐only (no residents) pediatric ward services during daytime hours. In the programs with both staff‐only and resident pediatric ward services, only 19% of patients were covered by the staff‐only teams and 81% of patients were covered by resident teams.
During the overnight period, 86% of resident teams did not have caps on the number of new patient admissions. An average of 3.6 providers per training program were in‐house overnight to accept patient admissions to pediatric wards. Ninety‐four percent of these providers in‐house were residents (399 residents in‐house/425 total providers in‐house each night).
Twenty‐five percent of the training programs that responded to the survey had pediatric hospitalist attendings in‐house at night. This included both overnight and partial nights (ie, until midnight). Other attendings in‐house at night include: neonatal intensive care unit (NICU) attendings (53% of programs), pediatric intensive care unit (PICU) attendings (46% of programs), Pediatric Emergency Medicine attendings (65% of programs), and Pediatric Surgery attendings (6.4% of programs). Twenty‐two percent of programs had no in‐house attendings at night (Figure 1).

Pediatric hospitalists were involved with 84% (n = 97) of training programs. Sixty percent (n = 58) of the pediatric hospitalist teams were staffed with both teaching attendings and residents. Fourteen percent (n = 14) of the pediatric hospitalist teams did not involve residents (staff‐only) and 25% (n = 25) had both types of teams. Specifically, of the programs that had pediatric hospitalists, 20% (n = 19) of them had hospitalist attendings in‐house 24 hours per day and 13% (n = 12) of teams had hospitalist attendings in‐house into the evening hours for a varying amount of time. Of the programs with hospitalist attendings in‐house 24 hours per day, 52% (n = 11) had started this coverage within the past 3 years.
Looking towards the future, and prior to the enactment of the October 2010 ACGME standards, 31% (n = 35) of the training programs that lacked 24/7 hospitalist in‐house coverage in January 2010 anticipated adding this level of coverage within the next 5 years. Notably, 70% (n = 81) of training programs felt that further resident work hour restrictions, which have since been enacted, would likely require the addition of more hospitalist attendings at night. Our survey allowed program directors to make open‐ended comments on how further work hour restrictions may change inpatient staffing in noncritical care inpatient teaching services.
DISCUSSION
To our knowledge, this was the first national study of pediatric resident coverage in noncritical care inpatient teaching services. While there was significant variation in how inpatient teaching services were covered across these programs, in January 2010, residents were involved in the majority of patient care with only 20% of programs having attending‐only hospitalist teams during the daytime. During the overnight period, the proportion of patient care provided by residents became even more significant with residents representing 94% of the total in‐house providers accepting new admissions. While pediatric hospitalists were prevalent at these training programs, their role in direct patient care overnight was limited. Only 6% of total in‐house providers accepting admissions at night were pediatric hospitalists.
The comments made by program directors are representative of the overall concerns regarding changes to resident work hours (see Table 2). In a position statement by the Association of Pediatric Program Directors in regards to the IOM recommendations, concerns were raised stating that the recommendations of the IOM Committee are intended to enhance patient safety without appropriate consideration for the educational and professional development of trainees.5 While the newly mandated ACGME standards are different than the previous IOM recommendations, it is clear that there will be very significant changes to accommodate these new standards. Our study was done prior to the new ACGME's standards. At the time of the survey, less than a third of programs were anticipating the addition of 24/7 pediatric hospitalist coverage; however, if resident work hours were further restricted, 70% of programs felt that additional hospitalists would be needed. This is a significant increase in the previously anticipated need for overnight attending hospitalist coverage, especially in light of the further restrictions mandated by the ACGME. We know that the response of New York State programs to the 405 regulations varied by program size, but all made significant changes to accommodate the new standards.6 It is clear that many program directors nationally are anticipating significant changes to their residencies when these new restrictions are enacted. The respondents in our survey felt that pediatric hospitalists are going to have to play an even bigger role at night when additional resident work hour restrictions are put into place.
|
| ▪ If the new duty hours are mandated, we would have to go to a night float system to be in compliance. This would require more residents and we do not have the funding to hire more residents. |
| ▪ Restrictions will be costly. It will increase shift work mentality, and increase pt errors due to handovers. If these (work restrictions) are not applied to all doctors (neurosurgeons, ICU doctors), they should not apply to resident doctors. |
| ▪ The additional restrictions may make the hospital consider giving up its residency program in favor of a hospitalist‐only model. |
| ▪ We do not have enough residents to care for the current patient load. |
| ▪ Additional work hour restrictions will lead to more hand‐over care and less ownership of patients by residents who identify themselves as primary patient physicians. Both situations are associated with increased rates of complications and possible sentinel events. |
| ▪ If the hours are reduced, the hospital will be forced to hire physicians for the care of patients. The administration of the hospital is now beginning to ask why they should financially support the training program if the residents are not providing a substantial portion of the hospital care for the patients. |
Pediatric hospital medicine remains a rapidly growing field.7 Eighty‐four percent of pediatric training programs utilize pediatric hospitalists. Over 60% of these pediatric hospitalist teams are involved in teaching teams with residents. While we did not directly study the supply and demand of pediatric hospitalists, there is some concern that even despite its rapid growth, the supply of pediatric hospitalists will not keep up with the demand when further resident work hours restrictions are implemented. At time of submission, a cost‐analysis has not yet been publicly published on the ACGME's new changes. There is data available based on the IOM's 2008 recommendations. A study by Nuckols and Escarce8 suggests that if the IOM's recommendations were implemented, the entire healthcare system nationally would have to develop and fill new full‐time positions equal to 5001 attending physicians, 5984 midlevel providers (nurse practitioners or physician assistants), 320 licensed vocational nurses, 229 nursing aides, and 45 laboratory technicians. This would be equivalent to adding an additional 8247 residency positions across all specialties.810 While the ACGME's new mandated changes are different than the IOM's recommendations, they will also restrict resident duty hours that we believe could lead to gaps in patient care requiring significant personnel changes in the healthcare system.
There are several limitations to our study. We did not study the role of pediatric subspecialty fellows and their involvement in pediatric inpatient services in these training programs. We also did not study the prevalence and use of resident night float systems. While night floats may be used in some programs, it may become more prevalent with the possible restriction in intern work hours down to 16 hours. As with any survey, there remains both volunteer and nonresponse bias with the programs that decide to complete or disregard the survey. Finally, there remains some concern over the data collection after the survey was sent out to the hospitalist listserve. Pediatric hospitalists may have incorrectly filled out the data for their program after their program director had already completed the survey. We attempted to minimize this problem by specifically instructing hospitalists to encourage their program director to fill out the survey if they had not already done so. We also compared computer Internet Protocol (IP) addresses and actual program responses, before and after the hospitalist e‐mail was sent, in an attempt to minimize the chance of including duplicated responses from the same program. Lastly, the January 2010 survey predated the October 2010 ACGME response to the IOM recommendations, and the responses may be different now that the specific restrictions have been mandated with an actual implementation date.
CONCLUSIONS
This study shows that pediatric teaching services varied significantly in how they provided overnight coverage in 2010 prior to new ACGME recommendations. Overall, residents were providing the overwhelming majority of the patient care overnight in pediatric training programs. While hospitalists were prevalent in pediatric training programs, in 2010 they had limited roles in direct patient care at night. The ACGME has now mandated additional residency work hour restrictions to be implemented July 2011. With these restrictions, hospitalists will likely need to expand their services, and additional hospitalists will be needed to provide overnight coverage. It is unclear where those hospitalists will come from and what their role will be. It is also unclear what the impact of increased demand and changed job description will be on the continued evolution of the field of Pediatric Hospital Medicine.
Future work needs to be done to establish benchmarks for inpatient coverage. The benchmarks could include guidelines on balancing patient safety with resident education. This may also involve the implementation of resident night float models. There needs to be monitoring on how changes in resident work hours and staffing affect coverage and, ultimately, how changes affect patient and resident outcomes.
APPENDIX
INPATIENT STAFFING WITHIN PEDIATRIC RESIDENCY PROGRAMS SURVEY
|
| Demographics |
| How many residents are in your residency program? (total, categorical, Med‐Peds, other combined Peds) |
| What is your average daily patient census per ward intern during daytime hours? |
| Does your hospital have a staff‐only (no residents) pediatric ward service during the daytime hours? |
| If your hospital has a staff‐only pediatric ward service, what are the proportion of patients cared for by residents vs staff‐only during daytime hours? |
| Do your residents cap the number of new patient admissions at night? |
| Providers in‐house overnight |
| How many providers do you have in‐house at night until midnight/overnight to accept patient admissions to pediatric wards? (residents, hospitalists, nurse practitioners, other) |
| Do you have attendings in‐house at night? (pediatric hospitalists, NICU, PICU, Peds EM, Peds Surgery, no attendings, other) |
| Pediatric hospitalists |
| Does your hospital have pediatric hospitalists? |
| Are your pediatric hospitalist teams staffed by: (teaching attendings and residents, hospitalist‐staff only, both) |
| If you have a staff‐only hospitalist team (no residents), how long has it been in existence? (less than 1 year, 1‐3 years, 4‐10 years, over 10 years) |
| Are your hospitalist attendings in‐house: (daytime only, 24 hours/day, other) |
| If your hospitalist attendings are in‐house 24/7, how many years has that coverage been available? (less than 1 year, 1‐3 years, 4‐10 years, over 10 years, not available) |
| Future pediatric hospitalist coverage |
| Do you anticipate that your hospital will be adding 24/7 hospitalist attending coverage? (next year, next 2 years, next 5 years, not anticipating adding coverage, 24/7 hospitalist coverage already in place) |
| In your opinion, would further resident work hour restrictions make your hospital more likely to add additional hospitalist attendings at night? (very likely, somewhat likely, neutral, not likely) |
- ,.The Bell Commission: ethical implications for the training of physicians.Mt Sinai J Med.2000;67(2):136–139.
- ,.Restricted duty hours for surgeons and impact on residents quality of life, education, and patient care: a literature review.Patient Saf Surg.2009;3(1):3.
- Institute of Medicine. Resident Duty Hours: Enhancing Sleep, Supervision, and Safety. Released December 02, 2008. Available at: http://www.iom.edu/Reports/2008/Resident‐Duty‐Hours‐Enhancing‐Sleep‐Supervision‐and‐Safety.aspx. Accessed September 20,2009.
- ACGME 2010 Standards “Common Program Requirements.” Available at: http://acgme‐2010standards.org/pdf/Common_Program_ Requirements_07012011.pdf. Accessed January 27,2011.
- Association of Pediatric Program Directors. Association of Pediatric Program Directors (APPD) Position Statement in Response to the IOM Recommendations on Resident Duty Hours.2009. Available at: http://www.appd.org/PDFs/APPD _IOM%20 _Duty _Hours _Report _Position _Paper _4–30‐09.pdf. Accessed March 27, 2010.
- ,,,,.Lessons learned from New York state: fourteen years of experience with work hour limitations.Acad Med.2005;80(5):467–472.
- ,,,.Health care market trends and the evolution of hospitalist use and rolesJ Gen Intern Med.2005;20(2):101–107.
- ,,,,.Cost implications of reduced work hours and workloads for resident physicians.N Engl J Med.2009;360:2202–2215.
- .Revisiting duty‐hour length—IOM recommendations for patient safety and resident education.N Engl J Med.2008;359:2633–2635.
- ,,,.Resident duty hour restrictions: is less really more?J Pediatr.2009;154:631–632.
- ,.The Bell Commission: ethical implications for the training of physicians.Mt Sinai J Med.2000;67(2):136–139.
- ,.Restricted duty hours for surgeons and impact on residents quality of life, education, and patient care: a literature review.Patient Saf Surg.2009;3(1):3.
- Institute of Medicine. Resident Duty Hours: Enhancing Sleep, Supervision, and Safety. Released December 02, 2008. Available at: http://www.iom.edu/Reports/2008/Resident‐Duty‐Hours‐Enhancing‐Sleep‐Supervision‐and‐Safety.aspx. Accessed September 20,2009.
- ACGME 2010 Standards “Common Program Requirements.” Available at: http://acgme‐2010standards.org/pdf/Common_Program_ Requirements_07012011.pdf. Accessed January 27,2011.
- Association of Pediatric Program Directors. Association of Pediatric Program Directors (APPD) Position Statement in Response to the IOM Recommendations on Resident Duty Hours.2009. Available at: http://www.appd.org/PDFs/APPD _IOM%20 _Duty _Hours _Report _Position _Paper _4–30‐09.pdf. Accessed March 27, 2010.
- ,,,,.Lessons learned from New York state: fourteen years of experience with work hour limitations.Acad Med.2005;80(5):467–472.
- ,,,.Health care market trends and the evolution of hospitalist use and rolesJ Gen Intern Med.2005;20(2):101–107.
- ,,,,.Cost implications of reduced work hours and workloads for resident physicians.N Engl J Med.2009;360:2202–2215.
- .Revisiting duty‐hour length—IOM recommendations for patient safety and resident education.N Engl J Med.2008;359:2633–2635.
- ,,,.Resident duty hour restrictions: is less really more?J Pediatr.2009;154:631–632.
Copyright © 2011 Society of Hospital Medicine
Improving Feedback to Ward Residents
Feedback has long been recognized as pivotal to the attainment of clinical acumen and skills in medical training.1 Formative feedback can give trainees insight into their strengths and weaknesses, and provide them with clear goals and methods to attain those goals.1, 2 In fact, feedback given regularly over time by a respected figure has shown to improve physician performance.3 However, most faculty are not trained to provide effective feedback. As a result, supervisors often believe they are giving more feedback than trainees believe they are receiving, and residents receive little feedback that they perceive as useful.4 Most residents receive little to no feedback on their communications skills4 or professionalism,5 and rarely receive corrective feedback.6, 7
Faculty may fail to give feedback to residents for a number of reasons. Those barriers most commonly cited in the literature are discomfort with criticizing residents,6, 7 lack of time,4 and lack of direct observation of residents in clinical settings.810 Several studies have looked at tools to guide feedback and address the barrier of discomfort with criticism.6, 7, 11 Some showed improvements in overall feedback, though often supervisors gave only positive feedback and avoided giving feedback about weaknesses.6, 7, 11 Despite the recognition of lack of time as a barrier to feedback,4 most studies on feedback interventions thus far have not included setting aside time for the feedback to occur.6, 7, 11, 12 Finally, a number of studies utilized objective structured clinical examinations (OSCEs) coupled with immediate feedback to improve direct observation of residents, with success in improving feedback related to the encounter.9, 10, 13 To address the gaps in the current literature, the goals of our study were to address 2 specific barriers to feedback for residents: lack of time and discomfort with giving feedback.
The aim of this study was to improve Internal Medicine (IM) residents' and attendings' experiences with feedback on the wards using a pocket card and a dedicated feedback session. We developed and evaluated the pocket feedback card and session for faculty to improve the quality and frequency of their feedback to residents in the inpatient setting. We performed a randomized trial to evaluate our intervention. We hypothesized that the intervention would: 1) improve the quality and quantity of attendings' feedback given to IM ward residents; and 2) improve attendings' comfort with feedback delivery on the wards.
PARTICIPANTS AND METHODS
Setting
The study was performed at Mount Sinai Medical Center in New York City, New York, between July 2008 and January 2009.
Participants
Participants in this study were IM residents and ward teaching attendings on inpatient ward teams at Mount Sinai Medical Center from July 2008 to January 2009. There are 12 ward teams on 3 inpatient services (each service has 4 teams) during each block at our hospital. Ward teams are made up of 1 teaching attending, 1 resident, 1 to 3 interns, and 1 to 2 medical students. The majority of attendings are on the ward service for 4‐week blocks, but some are only on for 1 or 2 weeks. Teams included in the randomization were the General Medicine and Gastroenterology/Cardiology service teams. Half of the General Medicine service attendings are hospitalists. Ward teams were excluded from the study randomization if the attending on the team was on the wards for less than 2 weeks, or if the attending had already been assigned to the experimental group in a previous block, given the influence of having used the card and feedback session previously. Since residents were unaware of the intervention and random assignments were based on attendings, residents could be assigned to the intervention group or the control group on any given inpatient rotation. Therefore, a resident could be in the control group in 1 block and the intervention group in his/her next block on the wards or vice versa, or could be assigned to either the intervention or the control group on more than 1 occasion. Because resident participants were blinded to their team's assignment (as intervention or control) and all surveys were anonymous (tracked as intervention or control by the team name only), it was not possible to exclude residents based on their prior participation or to match the surveys completed by the same residents.
Study Design
We performed a prospective randomized study to evaluate our educational innovation. The unit of randomization was the ward team. For each block, approximately half of the 6‐8 teams were randomized to the intervention group and half to the control group. Randomization assignments were completed the day prior to the start of the block using the random allocation software based on the ward team letters (blind to the attending and resident names). Of the 48 possible ward teams (8 teams per block over 6 blocks), 36 teams were randomized to the intervention or control groups, and 12 teams were not based on the above exclusion criteria. Of the 36 teams, 16 (composed of 16 attendings and 48 residents and interns) were randomized to the intervention group, and 20 (composed of 20 attendings and 63 residents and interns) were randomized to the control group.
The study was blinded such that residents and attendings in the control group were unaware of the study. The study was exempt from IRB review by the Mount Sinai Institutional Review Board, and Grants and Contracts Office, as an evaluation of the effectiveness of an instructional technique in medical education.
Intervention Design
We designed a pocket feedback card to guide a feedback session and assist attendings in giving useful feedback to IM residents on the wards (Figure 1).14 The individual items and categories were adapted from the Accreditation Council for Graduate Medical Education (ACGME) Common Program Requirements Core Competencies section and were revised via the expert consensus of the authors.14 We included 20 items related to resident skills, knowledge, attitudes, and behaviors important to the care of hospitalized patients, grouped under the 6 ACGME core competency domains.14 Many of these items correspond to competencies in the Society of Hospital Medicine (SHM) Core Competencies; in particular, the categories of Systems‐Based Practice and Practice‐Based Learning mirror competencies in the SHM Core Competencies Healthcare Systems chapter.15 Each item utilized a 5‐point Likert scale (1 = very poor, 3 = at expected level, 5 = superior) to evaluate resident performance (Figure 1). We created this card to serve as a directive and specific guide for attendings to provide feedback about specific domains and to give more constructive feedback. The card was to be used during a specific dedicated feedback session in order to overcome the commonly cited barrier of lack of time.
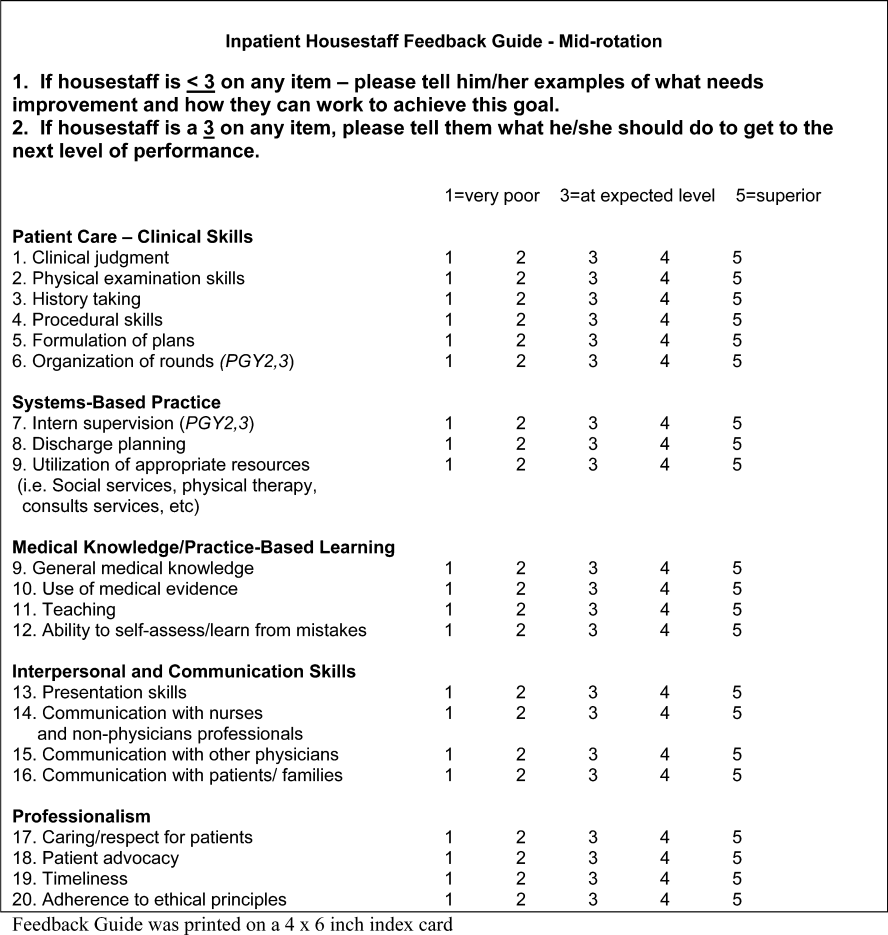
Program Implementation
On the first day of the block, both groups of attendings received the standard inpatient ward orientation given by the program director, including instructions about teaching and administrative responsibilities, and explicit instructions to provide mid‐rotation feedback to residents. Attendings randomized to the intervention group had an additional 5‐minute orientation given by 1 of the investigators. The orientation included a brief discussion on the importance of feedback and an introduction to the items on the card.2 In addition, faculty were instructed to dedicate 1 mid‐rotation attending rounds as a feedback session, to meet individually for 10‐15 minutes with each of the 3‐4 residents on their team, and to use the card to provide feedback on skills in each domain. As noted on the feedback card, if a resident scored less than 3 on a skill set, the attending was instructed to give examples of skills within that domain needing improvement and to offer suggestions for improvement. The intervention group was also asked not to discuss the card or session with others. No other instructions were provided.
Survey Design
At the end of each block, residents and attendings in both groups completed questionnaires to assess satisfaction with, and attitudes toward, feedback (Supporting Information Appendices 1 and 2 in the online version of this article). Survey questions were based on the competency areas included in the feedback card, previously published surveys evaluating feedback interventions,5, 9, 11 and expert opinion. The resident survey was designed to address the impact of feedback on the domains of resident knowledge, clinical and communication skills, and attitudes about feedback from supervisors and peers. We utilized a 5‐point Likert scale including: strongly disagree, disagree, neutral, agree, and strongly agree. The attending survey addressed attendings' satisfaction with feedback encounters and resident performance. At the completion of the study, investigators compared responses in intervention and control groups.
Statistical Analysis
For purposes of analysis, due to the relatively small number of responses for certain answer choices, the Likert scale was converted to a dichotomous variable. The responses of agree and strongly agree were coded as agree; and disagree, strongly disagree, and neutral were coded as disagree. Neutral was coded as disagree in order to avoid overestimating positive attitudes and, in effect, bias our results toward the null hypothesis. Differences between groups were analyzed using chi‐square Fisher's exact test (2‐sided).
Qualitative Interviews
In order to understand the relative contribution of the feedback card versus the feedback session, we performed a qualitative survey of attendings in the intervention group. Following the conclusion of the study period, we selected a convenience sample of 8 attendings from the intervention group for these brief qualitative interviews. We asked 3 basic questions. Was the intervention of the feedback card and dedicated time for feedback useful? Did you find one component, either the card or the dedicated time for feedback, more useful than the other? Were there any negative effects on patient care, education, or other areas, from using an attending rounds as a feedback session? This data was coded and analyzed for common themes.
RESULTS
During the 6‐month study period, 34 teaching attendings (over 36 attending inpatient blocks) and 93 IM residents (over 111 resident inpatient blocks) participated in the study. Thirty‐four of 36 attending surveys and 96 of 111 resident surveys were completed. The overall survey response rates for residents and attendings were 85% and 94%, respectively. Two attendings participated during 2 separate blocks, first in the control group and then in the intervention group, and 18 residents participated during 2 separate blocks. No attendings or residents participated more than twice.
Resident survey response rate was 81.2% in the intervention group and 87.3% in the control group (Table 1). Residents in the intervention group reported receiving more feedback regarding skills they did well (89.7% vs 63.6%, P = 0.004) and skills needing improvement (51.3% vs 25.5%, P = 0.02) than those in the control group. In addition, more intervention residents reported receiving useful information regarding how to improve their skills (53.8% vs 27.3%, P = 0.01), and reported actually improving both their clinical skills (61.5% vs 27.8%, P = 0.001) and their professionalism/communication skills (51.3% vs 29.1%, P = 0.03) based on feedback received from attendings.
| Survey Item | Resident Intervention Agree* % (No.) N = 39 | Resident Control Agree*% (No.) N = 55 | P Value |
|---|---|---|---|
| |||
| I did NOT receive a sufficient amount of feedback from my attending supervisor(s) this block. | 20.5 (8) | 38.2 (21) | 0.08 |
| I received feedback from my attending regarding skills I did well during this block. | 89.7 (35) | 63.6 (35) | 0.004 |
| I received feedback from my attending regarding specific skills that needed improvement during this block. | 51.3 (20) | 25.5 (14) | 0.02 |
| I received useful information from my attending about how to improve my skills during this block. | 53.8 (21) | 27.3 (15) | 0.01 |
| I improved my clinical skills based on feedback I received from my attending this block. | 61.5 (24) | 27.8 (15) | 0.001 |
| I improved my professionalism/communication skills based on feedback I received from my attending this block. | 51.3 (20) | 29.1 (16) | 0.03 |
| I improved my knowledge base because of feedback I received from my attending this block. | 64.1 (25) | 60.0 (33) | 0.83 |
| The feedback I received from my attending this block gave me an overall sense of my performance more than it helped me identify specific areas for improvement. | 64.1 (25) | 65.5 (36) | 1.0 |
| Feedback from colleagues (other interns and residents) is more helpful than feedback from attendings. | 41.0 (16) | 43.6 (24) | 0.84 |
| Independent of feedback received from others, I am able to identify areas in which I need improvement. | 84.6 (33) | 80.0 (44) | 0.60 |
The attending survey response rates for the intervention and control groups were 100% and 90%, respectively. In general, both groups of attendings reported that they were comfortable giving feedback and that they did, in fact, give feedback in each area during their ward block (Table 2). More intervention attendings felt that at least 1 of their residents improved their professionalism/communication skills based on the feedback given (76.9% vs 31.1%, P = 0.02). There were no other significant differences between the groups of attendings.
| Survey Item | Attending Intervention Agree* % (No.) N = 16 | Attending Control Agree* % (No.) N = 18 | P Value |
|---|---|---|---|
| |||
| Giving feedback to housestaff was DIFFICULT this block. | 6.3 (1) | 16.7 (3) | 0.60 |
| I was comfortable giving feedback to my housestaff this block. | 93.8 (15) | 94.4 (17) | 1.00 |
| I did NOT give a sufficient amount of feedback to my housestaff this block. | 18.8 (3) | 38.9 (7) | 0.27 |
| My skills in giving feedback improved during this block. | 50 (8) | 16.7 (3) | 0.07 |
| I gave feedback to housestaff regarding skills they did well during this block. | 100 (16) | 94.4 (17) | 1.00 |
| I gave feedback to housestaff which targeted specific areas for their improvement. | 81.3 (13) | 70.6 (12) | 0.69 |
| At least one of my housestaff improved his/her clinical skills based on feedback I gave this block. | 68.8 (11) | 47.1 (8) | 0.30 |
| At least one of my housestaff improved his/her professionalism/communication skills based on feedback I gave this block. | 76.9 (10) | 31.1 (5) | 0.02 |
| At least one of my housestaff improved his/her fund of knowledge based on feedback I gave this block. | 50.0 (8) | 52.9 (9) | 1.00 |
| Housestaff found the feedback I gave them useful. | 66.7 (10) | 62.5 (10) | 1.00 |
| I find it DIFFICULT to find time during inpatient rotations to give feedback to residents regarding their performance. | 50.0 (8) | 33.3 (6) | 0.49 |
Intervention attendings also shared their attitudes toward the feedback card and session. A majority felt that using 1 attending rounds as a feedback session helped create a dedicated time for giving feedback (68.8%), and that the feedback card helped them to give specific, constructive feedback (62.5%). Most attendings reported they would use the feedback card and session again during future inpatient blocks (81%), and would recommend them to other attendings (75%).
Qualitative data from intervention attending interviews revealed further thoughts about the feedback card and feedback session. Most attendings interviewed (7/8) felt that the card was useful for the structure and topic guidance it provided. Half felt that setting aside time for feedback was the more useful component. The other half reported that, because they usually set aside time for feedback regardless, the card was more useful. None of the attendings felt that the feedback card or session was detrimental for patient care or education, and many said that the intervention had positive effects on these areas. For example, 1 attending said that the session added to patient care because I used particular [patient] cases as examples for giving feedback.
DISCUSSION
In this randomized study, we found that a simple pocket feedback card and dedicated feedback session was acceptable to ward attendings and improved resident satisfaction with feedback. Unlike most prior studies of feedback, we demonstrated more feedback around skills needing improvement, and intervention residents felt the feedback they received helped them improve their skills. Our educational intervention was unique in that it combined a pocket card to structure feedback content and dedicated time to structure the feedback process, to address 2 of the major barriers to giving feedback: lack of time and lack of comfort.
The pocket card itself as a tool for improving feedback is innovative and valuable. As a short but directive guide, the card supports attendings' delivery of relevant and specific feedback about residents' performance, and because it is based on the ACGME competencies, it may help attendings focus feedback on areas in which they will later evaluate residents. The inclusion of a prespecified time for giving feedback was important as well, in that it allowed for face‐to‐face feedback to occur, as opposed to a passing comment after a presentation or brief notes in a written final evaluation. Both the card and the feedback session seemed equally important for the success of this intervention, with attitudes varying based on individual attending preferences. Those who usually set aside time for feedback on their own found the card more useful, whereas those who had more trouble finding time for feedback found the specific session more useful. Most attendings found the intervention as a whole helpful, and without any detrimental effects on patient care or education. The card and session may be particularly valuable for hospital attendings, given their growing presence as teachers and supervisors for residents, and their busy days on the wards.
Our study results have important implications for resident training in the hospital. Improving resident receipt of feedback about strengths and weaknesses is an ACGME training requirement, and specific guidance about how to improve skills is critical for focusing improvement efforts. Previous studies have demonstrated that directive feedback in medical training can lead to a variety of performance improvements, including better evaluations by other professionals,9, 16 and objective improvements in resident communication skills,17 chart documentation,18 and clinical management of patients.11, 15, 19 By improving the quality of feedback across several domains and facilitating the feedback process, our intervention may lead to similar improvements. Future studies should examine the global impact of guided feedback as in our study. Perhaps most importantly, attendings found the intervention acceptable and would recommend its use, implying longer term sustainability of its integration into the hospital routine.
One strength of our study was its prospective randomized design. Despite the importance of rigor in medical education research, there remains a paucity of randomized studies to evaluate educational interventions for residents in inpatient settings. Few studies of feedback interventions in particular have performed randomized trials,5, 6, 11 and only one has examined a feedback intervention in a randomized fashion in the inpatient setting.12 This evaluation of a 20‐minute intervention, and a reminder card for supervising attendings to improve written and verbal feedback to residents, modestly improved the amount of verbal feedback given to residents, but did not impact the number of residents receiving mid‐rotation feedback or feedback overall as our study did by report.12
There were several important limitations to our study. First, because this was a single institution study, we only achieved modest sample sizes, particularly in the attending groups, and were unable to assess all of the differences in attending attitudes related to feedback. Second, control and intervention participants were on service simultaneously, which may have led to contamination of the control group and an underestimation of the true impact of our intervention. Since residents were not exclusive to 1 study group on 1 occasion (18 of the 93 residents participated during 2 separate blocks), our results may be biased. In particular, those residents who had the intervention first, and were subsequently in the control group, may have rated the control experience worse than they would have otherwise, creating a bias in favor of a positive result for our intervention. Nonetheless, we believe this situation was uncommon and the potential associated bias minimal. Further, this study assessed attitudes related to feedback and self‐reported knowledge and skills, but did not directly assess resident knowledge, skills, or patient outcomes. We recognize the importance of these outcomes and hope that future interventions can determine these important downstream effects of feedback. We were also unable to assess the card and session's impact on attendings' comfort with feedback, because most attendings in both groups reported feeling comfortable giving feedback. This result may indicate that attendings actually are comfortable giving feedback, or may suggest some element of social desirability bias. Finally, in this study, we designed an intervention which combined the pocket card and dedicated feedback time. We did not quantitatively examine the effect of either component alone, and it is unclear if offering the feedback card without protected time or offering protected time without a guide would have impacted feedback on the wards. However, qualitative data from our study support the use of both components, and implementing the 2 components together is feasible in any inpatient teaching setting.
Despite these limitations, protected time for feedback guided by a pocket feedback card is a simple intervention that appears to improve feedback quantity and quality for ward residents, and guides them to improve their performance. Our low‐intensity intervention helped attendings give residents the tools to improve their clinical and communication skills. An opportunity to make a positive impact on resident education with such a small intervention is rare. The use of a feedback card with protected feedback time could be easily implemented in any training program, and is a valuable tool for busy hospitalists who are more commonly supervising residents on their inpatient rotations.
- .Feedback in clinical medical education.JAMA.1983;250(6):777–781.
- ,.Giving feedback in medical education: verification of recommended techniques.J Gen Intern Med.1998;13(2):111–116.
- ,,,,.Systematic review of the literature on assessment, feedback and physicians' clinical performance: BEME Guide No. 7.Med Teach.2006;28(2):117–128.
- ,,,.Missed opportunities: a descriptive assessment of teaching and attitudes regarding communication skills in a surgical residency.Curr Surg.2006;63(6):401–409.
- ,,.Impact of a 360‐degree professionalism assessment on faculty comfort and skills in feedback delivery.J Gen Intern Med.2008;23(7):969–972.
- ,.Daily encounter cards facilitate competency‐based feedback while leniency bias persists.CJEM.2008;10(1):44–50.
- ,,,,.Teaching compassion and respect. Attending physicians' responses to problematic behaviors.J Gen Intern Med.1999;14(1):49–55.
- .Faculty and the observation of trainees' clinical skills: problems and opportunities.Acad Med.2004;79(1):16–22.
- ,.Direct observation of residents in the emergency department: a structured educational program.Acad Emerg Med.2009;16(4):343–351.
- ,,, et al.Evaluation of a novel assessment form for observing medical residents: a randomised, controlled trial.Med Educ.2008;42(12):1234–1242.
- ,,, et al.Resident evaluations: the use of daily evaluation forms in rheumatology ambulatory care.J Rheumatol.2009;36(6):1298–1303.
- ,,,.Effectiveness of a focused educational intervention on resident evaluations from faculty a randomized controlled trial.J Gen Intern Med.2001;16(7):427–434.
- ,,.Effects of training in direct observation of medical residents' clinical competence: a randomized trial.Ann Intern Med.2004;140(11):874–881.
- Internal Medicine Program Requirements. ACGME. July 1, 2009. Available at: http://www.acgme.org/acWebsite/downloads/RRC_progReq/140_internal_medicine_07012009.pdf. Accessed November 8,2009.
- ,,,,.How to use the core competencies in hospital medicine: a framework for curriculum development.J Hosp Med. 2006;1(suppl 1):57–67.
- ,,,,.Debriefing in the intensive care unit: a feedback tool to facilitate bedside teaching.Crit Care Med.2007;35(3):738–754.
- ,,, et al.Use of an innovative video feedback technique to enhance communication skills training.Med Educ.2004;38(2):145–157.
- .The impact of feedback to medical housestaff on chart documentation and quality of care in the outpatient setting.J Gen Intern Med.1997;12(6):352–356.
- ,,,.Feedback and the mini clinical evaluation exercise.J Gen Intern Med.2004;19(5 pt 2):558–561.
Feedback has long been recognized as pivotal to the attainment of clinical acumen and skills in medical training.1 Formative feedback can give trainees insight into their strengths and weaknesses, and provide them with clear goals and methods to attain those goals.1, 2 In fact, feedback given regularly over time by a respected figure has shown to improve physician performance.3 However, most faculty are not trained to provide effective feedback. As a result, supervisors often believe they are giving more feedback than trainees believe they are receiving, and residents receive little feedback that they perceive as useful.4 Most residents receive little to no feedback on their communications skills4 or professionalism,5 and rarely receive corrective feedback.6, 7
Faculty may fail to give feedback to residents for a number of reasons. Those barriers most commonly cited in the literature are discomfort with criticizing residents,6, 7 lack of time,4 and lack of direct observation of residents in clinical settings.810 Several studies have looked at tools to guide feedback and address the barrier of discomfort with criticism.6, 7, 11 Some showed improvements in overall feedback, though often supervisors gave only positive feedback and avoided giving feedback about weaknesses.6, 7, 11 Despite the recognition of lack of time as a barrier to feedback,4 most studies on feedback interventions thus far have not included setting aside time for the feedback to occur.6, 7, 11, 12 Finally, a number of studies utilized objective structured clinical examinations (OSCEs) coupled with immediate feedback to improve direct observation of residents, with success in improving feedback related to the encounter.9, 10, 13 To address the gaps in the current literature, the goals of our study were to address 2 specific barriers to feedback for residents: lack of time and discomfort with giving feedback.
The aim of this study was to improve Internal Medicine (IM) residents' and attendings' experiences with feedback on the wards using a pocket card and a dedicated feedback session. We developed and evaluated the pocket feedback card and session for faculty to improve the quality and frequency of their feedback to residents in the inpatient setting. We performed a randomized trial to evaluate our intervention. We hypothesized that the intervention would: 1) improve the quality and quantity of attendings' feedback given to IM ward residents; and 2) improve attendings' comfort with feedback delivery on the wards.
PARTICIPANTS AND METHODS
Setting
The study was performed at Mount Sinai Medical Center in New York City, New York, between July 2008 and January 2009.
Participants
Participants in this study were IM residents and ward teaching attendings on inpatient ward teams at Mount Sinai Medical Center from July 2008 to January 2009. There are 12 ward teams on 3 inpatient services (each service has 4 teams) during each block at our hospital. Ward teams are made up of 1 teaching attending, 1 resident, 1 to 3 interns, and 1 to 2 medical students. The majority of attendings are on the ward service for 4‐week blocks, but some are only on for 1 or 2 weeks. Teams included in the randomization were the General Medicine and Gastroenterology/Cardiology service teams. Half of the General Medicine service attendings are hospitalists. Ward teams were excluded from the study randomization if the attending on the team was on the wards for less than 2 weeks, or if the attending had already been assigned to the experimental group in a previous block, given the influence of having used the card and feedback session previously. Since residents were unaware of the intervention and random assignments were based on attendings, residents could be assigned to the intervention group or the control group on any given inpatient rotation. Therefore, a resident could be in the control group in 1 block and the intervention group in his/her next block on the wards or vice versa, or could be assigned to either the intervention or the control group on more than 1 occasion. Because resident participants were blinded to their team's assignment (as intervention or control) and all surveys were anonymous (tracked as intervention or control by the team name only), it was not possible to exclude residents based on their prior participation or to match the surveys completed by the same residents.
Study Design
We performed a prospective randomized study to evaluate our educational innovation. The unit of randomization was the ward team. For each block, approximately half of the 6‐8 teams were randomized to the intervention group and half to the control group. Randomization assignments were completed the day prior to the start of the block using the random allocation software based on the ward team letters (blind to the attending and resident names). Of the 48 possible ward teams (8 teams per block over 6 blocks), 36 teams were randomized to the intervention or control groups, and 12 teams were not based on the above exclusion criteria. Of the 36 teams, 16 (composed of 16 attendings and 48 residents and interns) were randomized to the intervention group, and 20 (composed of 20 attendings and 63 residents and interns) were randomized to the control group.
The study was blinded such that residents and attendings in the control group were unaware of the study. The study was exempt from IRB review by the Mount Sinai Institutional Review Board, and Grants and Contracts Office, as an evaluation of the effectiveness of an instructional technique in medical education.
Intervention Design
We designed a pocket feedback card to guide a feedback session and assist attendings in giving useful feedback to IM residents on the wards (Figure 1).14 The individual items and categories were adapted from the Accreditation Council for Graduate Medical Education (ACGME) Common Program Requirements Core Competencies section and were revised via the expert consensus of the authors.14 We included 20 items related to resident skills, knowledge, attitudes, and behaviors important to the care of hospitalized patients, grouped under the 6 ACGME core competency domains.14 Many of these items correspond to competencies in the Society of Hospital Medicine (SHM) Core Competencies; in particular, the categories of Systems‐Based Practice and Practice‐Based Learning mirror competencies in the SHM Core Competencies Healthcare Systems chapter.15 Each item utilized a 5‐point Likert scale (1 = very poor, 3 = at expected level, 5 = superior) to evaluate resident performance (Figure 1). We created this card to serve as a directive and specific guide for attendings to provide feedback about specific domains and to give more constructive feedback. The card was to be used during a specific dedicated feedback session in order to overcome the commonly cited barrier of lack of time.

Program Implementation
On the first day of the block, both groups of attendings received the standard inpatient ward orientation given by the program director, including instructions about teaching and administrative responsibilities, and explicit instructions to provide mid‐rotation feedback to residents. Attendings randomized to the intervention group had an additional 5‐minute orientation given by 1 of the investigators. The orientation included a brief discussion on the importance of feedback and an introduction to the items on the card.2 In addition, faculty were instructed to dedicate 1 mid‐rotation attending rounds as a feedback session, to meet individually for 10‐15 minutes with each of the 3‐4 residents on their team, and to use the card to provide feedback on skills in each domain. As noted on the feedback card, if a resident scored less than 3 on a skill set, the attending was instructed to give examples of skills within that domain needing improvement and to offer suggestions for improvement. The intervention group was also asked not to discuss the card or session with others. No other instructions were provided.
Survey Design
At the end of each block, residents and attendings in both groups completed questionnaires to assess satisfaction with, and attitudes toward, feedback (Supporting Information Appendices 1 and 2 in the online version of this article). Survey questions were based on the competency areas included in the feedback card, previously published surveys evaluating feedback interventions,5, 9, 11 and expert opinion. The resident survey was designed to address the impact of feedback on the domains of resident knowledge, clinical and communication skills, and attitudes about feedback from supervisors and peers. We utilized a 5‐point Likert scale including: strongly disagree, disagree, neutral, agree, and strongly agree. The attending survey addressed attendings' satisfaction with feedback encounters and resident performance. At the completion of the study, investigators compared responses in intervention and control groups.
Statistical Analysis
For purposes of analysis, due to the relatively small number of responses for certain answer choices, the Likert scale was converted to a dichotomous variable. The responses of agree and strongly agree were coded as agree; and disagree, strongly disagree, and neutral were coded as disagree. Neutral was coded as disagree in order to avoid overestimating positive attitudes and, in effect, bias our results toward the null hypothesis. Differences between groups were analyzed using chi‐square Fisher's exact test (2‐sided).
Qualitative Interviews
In order to understand the relative contribution of the feedback card versus the feedback session, we performed a qualitative survey of attendings in the intervention group. Following the conclusion of the study period, we selected a convenience sample of 8 attendings from the intervention group for these brief qualitative interviews. We asked 3 basic questions. Was the intervention of the feedback card and dedicated time for feedback useful? Did you find one component, either the card or the dedicated time for feedback, more useful than the other? Were there any negative effects on patient care, education, or other areas, from using an attending rounds as a feedback session? This data was coded and analyzed for common themes.
RESULTS
During the 6‐month study period, 34 teaching attendings (over 36 attending inpatient blocks) and 93 IM residents (over 111 resident inpatient blocks) participated in the study. Thirty‐four of 36 attending surveys and 96 of 111 resident surveys were completed. The overall survey response rates for residents and attendings were 85% and 94%, respectively. Two attendings participated during 2 separate blocks, first in the control group and then in the intervention group, and 18 residents participated during 2 separate blocks. No attendings or residents participated more than twice.
Resident survey response rate was 81.2% in the intervention group and 87.3% in the control group (Table 1). Residents in the intervention group reported receiving more feedback regarding skills they did well (89.7% vs 63.6%, P = 0.004) and skills needing improvement (51.3% vs 25.5%, P = 0.02) than those in the control group. In addition, more intervention residents reported receiving useful information regarding how to improve their skills (53.8% vs 27.3%, P = 0.01), and reported actually improving both their clinical skills (61.5% vs 27.8%, P = 0.001) and their professionalism/communication skills (51.3% vs 29.1%, P = 0.03) based on feedback received from attendings.
| Survey Item | Resident Intervention Agree* % (No.) N = 39 | Resident Control Agree*% (No.) N = 55 | P Value |
|---|---|---|---|
| |||
| I did NOT receive a sufficient amount of feedback from my attending supervisor(s) this block. | 20.5 (8) | 38.2 (21) | 0.08 |
| I received feedback from my attending regarding skills I did well during this block. | 89.7 (35) | 63.6 (35) | 0.004 |
| I received feedback from my attending regarding specific skills that needed improvement during this block. | 51.3 (20) | 25.5 (14) | 0.02 |
| I received useful information from my attending about how to improve my skills during this block. | 53.8 (21) | 27.3 (15) | 0.01 |
| I improved my clinical skills based on feedback I received from my attending this block. | 61.5 (24) | 27.8 (15) | 0.001 |
| I improved my professionalism/communication skills based on feedback I received from my attending this block. | 51.3 (20) | 29.1 (16) | 0.03 |
| I improved my knowledge base because of feedback I received from my attending this block. | 64.1 (25) | 60.0 (33) | 0.83 |
| The feedback I received from my attending this block gave me an overall sense of my performance more than it helped me identify specific areas for improvement. | 64.1 (25) | 65.5 (36) | 1.0 |
| Feedback from colleagues (other interns and residents) is more helpful than feedback from attendings. | 41.0 (16) | 43.6 (24) | 0.84 |
| Independent of feedback received from others, I am able to identify areas in which I need improvement. | 84.6 (33) | 80.0 (44) | 0.60 |
The attending survey response rates for the intervention and control groups were 100% and 90%, respectively. In general, both groups of attendings reported that they were comfortable giving feedback and that they did, in fact, give feedback in each area during their ward block (Table 2). More intervention attendings felt that at least 1 of their residents improved their professionalism/communication skills based on the feedback given (76.9% vs 31.1%, P = 0.02). There were no other significant differences between the groups of attendings.
| Survey Item | Attending Intervention Agree* % (No.) N = 16 | Attending Control Agree* % (No.) N = 18 | P Value |
|---|---|---|---|
| |||
| Giving feedback to housestaff was DIFFICULT this block. | 6.3 (1) | 16.7 (3) | 0.60 |
| I was comfortable giving feedback to my housestaff this block. | 93.8 (15) | 94.4 (17) | 1.00 |
| I did NOT give a sufficient amount of feedback to my housestaff this block. | 18.8 (3) | 38.9 (7) | 0.27 |
| My skills in giving feedback improved during this block. | 50 (8) | 16.7 (3) | 0.07 |
| I gave feedback to housestaff regarding skills they did well during this block. | 100 (16) | 94.4 (17) | 1.00 |
| I gave feedback to housestaff which targeted specific areas for their improvement. | 81.3 (13) | 70.6 (12) | 0.69 |
| At least one of my housestaff improved his/her clinical skills based on feedback I gave this block. | 68.8 (11) | 47.1 (8) | 0.30 |
| At least one of my housestaff improved his/her professionalism/communication skills based on feedback I gave this block. | 76.9 (10) | 31.1 (5) | 0.02 |
| At least one of my housestaff improved his/her fund of knowledge based on feedback I gave this block. | 50.0 (8) | 52.9 (9) | 1.00 |
| Housestaff found the feedback I gave them useful. | 66.7 (10) | 62.5 (10) | 1.00 |
| I find it DIFFICULT to find time during inpatient rotations to give feedback to residents regarding their performance. | 50.0 (8) | 33.3 (6) | 0.49 |
Intervention attendings also shared their attitudes toward the feedback card and session. A majority felt that using 1 attending rounds as a feedback session helped create a dedicated time for giving feedback (68.8%), and that the feedback card helped them to give specific, constructive feedback (62.5%). Most attendings reported they would use the feedback card and session again during future inpatient blocks (81%), and would recommend them to other attendings (75%).
Qualitative data from intervention attending interviews revealed further thoughts about the feedback card and feedback session. Most attendings interviewed (7/8) felt that the card was useful for the structure and topic guidance it provided. Half felt that setting aside time for feedback was the more useful component. The other half reported that, because they usually set aside time for feedback regardless, the card was more useful. None of the attendings felt that the feedback card or session was detrimental for patient care or education, and many said that the intervention had positive effects on these areas. For example, 1 attending said that the session added to patient care because I used particular [patient] cases as examples for giving feedback.
DISCUSSION
In this randomized study, we found that a simple pocket feedback card and dedicated feedback session was acceptable to ward attendings and improved resident satisfaction with feedback. Unlike most prior studies of feedback, we demonstrated more feedback around skills needing improvement, and intervention residents felt the feedback they received helped them improve their skills. Our educational intervention was unique in that it combined a pocket card to structure feedback content and dedicated time to structure the feedback process, to address 2 of the major barriers to giving feedback: lack of time and lack of comfort.
The pocket card itself as a tool for improving feedback is innovative and valuable. As a short but directive guide, the card supports attendings' delivery of relevant and specific feedback about residents' performance, and because it is based on the ACGME competencies, it may help attendings focus feedback on areas in which they will later evaluate residents. The inclusion of a prespecified time for giving feedback was important as well, in that it allowed for face‐to‐face feedback to occur, as opposed to a passing comment after a presentation or brief notes in a written final evaluation. Both the card and the feedback session seemed equally important for the success of this intervention, with attitudes varying based on individual attending preferences. Those who usually set aside time for feedback on their own found the card more useful, whereas those who had more trouble finding time for feedback found the specific session more useful. Most attendings found the intervention as a whole helpful, and without any detrimental effects on patient care or education. The card and session may be particularly valuable for hospital attendings, given their growing presence as teachers and supervisors for residents, and their busy days on the wards.
Our study results have important implications for resident training in the hospital. Improving resident receipt of feedback about strengths and weaknesses is an ACGME training requirement, and specific guidance about how to improve skills is critical for focusing improvement efforts. Previous studies have demonstrated that directive feedback in medical training can lead to a variety of performance improvements, including better evaluations by other professionals,9, 16 and objective improvements in resident communication skills,17 chart documentation,18 and clinical management of patients.11, 15, 19 By improving the quality of feedback across several domains and facilitating the feedback process, our intervention may lead to similar improvements. Future studies should examine the global impact of guided feedback as in our study. Perhaps most importantly, attendings found the intervention acceptable and would recommend its use, implying longer term sustainability of its integration into the hospital routine.
One strength of our study was its prospective randomized design. Despite the importance of rigor in medical education research, there remains a paucity of randomized studies to evaluate educational interventions for residents in inpatient settings. Few studies of feedback interventions in particular have performed randomized trials,5, 6, 11 and only one has examined a feedback intervention in a randomized fashion in the inpatient setting.12 This evaluation of a 20‐minute intervention, and a reminder card for supervising attendings to improve written and verbal feedback to residents, modestly improved the amount of verbal feedback given to residents, but did not impact the number of residents receiving mid‐rotation feedback or feedback overall as our study did by report.12
There were several important limitations to our study. First, because this was a single institution study, we only achieved modest sample sizes, particularly in the attending groups, and were unable to assess all of the differences in attending attitudes related to feedback. Second, control and intervention participants were on service simultaneously, which may have led to contamination of the control group and an underestimation of the true impact of our intervention. Since residents were not exclusive to 1 study group on 1 occasion (18 of the 93 residents participated during 2 separate blocks), our results may be biased. In particular, those residents who had the intervention first, and were subsequently in the control group, may have rated the control experience worse than they would have otherwise, creating a bias in favor of a positive result for our intervention. Nonetheless, we believe this situation was uncommon and the potential associated bias minimal. Further, this study assessed attitudes related to feedback and self‐reported knowledge and skills, but did not directly assess resident knowledge, skills, or patient outcomes. We recognize the importance of these outcomes and hope that future interventions can determine these important downstream effects of feedback. We were also unable to assess the card and session's impact on attendings' comfort with feedback, because most attendings in both groups reported feeling comfortable giving feedback. This result may indicate that attendings actually are comfortable giving feedback, or may suggest some element of social desirability bias. Finally, in this study, we designed an intervention which combined the pocket card and dedicated feedback time. We did not quantitatively examine the effect of either component alone, and it is unclear if offering the feedback card without protected time or offering protected time without a guide would have impacted feedback on the wards. However, qualitative data from our study support the use of both components, and implementing the 2 components together is feasible in any inpatient teaching setting.
Despite these limitations, protected time for feedback guided by a pocket feedback card is a simple intervention that appears to improve feedback quantity and quality for ward residents, and guides them to improve their performance. Our low‐intensity intervention helped attendings give residents the tools to improve their clinical and communication skills. An opportunity to make a positive impact on resident education with such a small intervention is rare. The use of a feedback card with protected feedback time could be easily implemented in any training program, and is a valuable tool for busy hospitalists who are more commonly supervising residents on their inpatient rotations.
Feedback has long been recognized as pivotal to the attainment of clinical acumen and skills in medical training.1 Formative feedback can give trainees insight into their strengths and weaknesses, and provide them with clear goals and methods to attain those goals.1, 2 In fact, feedback given regularly over time by a respected figure has shown to improve physician performance.3 However, most faculty are not trained to provide effective feedback. As a result, supervisors often believe they are giving more feedback than trainees believe they are receiving, and residents receive little feedback that they perceive as useful.4 Most residents receive little to no feedback on their communications skills4 or professionalism,5 and rarely receive corrective feedback.6, 7
Faculty may fail to give feedback to residents for a number of reasons. Those barriers most commonly cited in the literature are discomfort with criticizing residents,6, 7 lack of time,4 and lack of direct observation of residents in clinical settings.810 Several studies have looked at tools to guide feedback and address the barrier of discomfort with criticism.6, 7, 11 Some showed improvements in overall feedback, though often supervisors gave only positive feedback and avoided giving feedback about weaknesses.6, 7, 11 Despite the recognition of lack of time as a barrier to feedback,4 most studies on feedback interventions thus far have not included setting aside time for the feedback to occur.6, 7, 11, 12 Finally, a number of studies utilized objective structured clinical examinations (OSCEs) coupled with immediate feedback to improve direct observation of residents, with success in improving feedback related to the encounter.9, 10, 13 To address the gaps in the current literature, the goals of our study were to address 2 specific barriers to feedback for residents: lack of time and discomfort with giving feedback.
The aim of this study was to improve Internal Medicine (IM) residents' and attendings' experiences with feedback on the wards using a pocket card and a dedicated feedback session. We developed and evaluated the pocket feedback card and session for faculty to improve the quality and frequency of their feedback to residents in the inpatient setting. We performed a randomized trial to evaluate our intervention. We hypothesized that the intervention would: 1) improve the quality and quantity of attendings' feedback given to IM ward residents; and 2) improve attendings' comfort with feedback delivery on the wards.
PARTICIPANTS AND METHODS
Setting
The study was performed at Mount Sinai Medical Center in New York City, New York, between July 2008 and January 2009.
Participants
Participants in this study were IM residents and ward teaching attendings on inpatient ward teams at Mount Sinai Medical Center from July 2008 to January 2009. There are 12 ward teams on 3 inpatient services (each service has 4 teams) during each block at our hospital. Ward teams are made up of 1 teaching attending, 1 resident, 1 to 3 interns, and 1 to 2 medical students. The majority of attendings are on the ward service for 4‐week blocks, but some are only on for 1 or 2 weeks. Teams included in the randomization were the General Medicine and Gastroenterology/Cardiology service teams. Half of the General Medicine service attendings are hospitalists. Ward teams were excluded from the study randomization if the attending on the team was on the wards for less than 2 weeks, or if the attending had already been assigned to the experimental group in a previous block, given the influence of having used the card and feedback session previously. Since residents were unaware of the intervention and random assignments were based on attendings, residents could be assigned to the intervention group or the control group on any given inpatient rotation. Therefore, a resident could be in the control group in 1 block and the intervention group in his/her next block on the wards or vice versa, or could be assigned to either the intervention or the control group on more than 1 occasion. Because resident participants were blinded to their team's assignment (as intervention or control) and all surveys were anonymous (tracked as intervention or control by the team name only), it was not possible to exclude residents based on their prior participation or to match the surveys completed by the same residents.
Study Design
We performed a prospective randomized study to evaluate our educational innovation. The unit of randomization was the ward team. For each block, approximately half of the 6‐8 teams were randomized to the intervention group and half to the control group. Randomization assignments were completed the day prior to the start of the block using the random allocation software based on the ward team letters (blind to the attending and resident names). Of the 48 possible ward teams (8 teams per block over 6 blocks), 36 teams were randomized to the intervention or control groups, and 12 teams were not based on the above exclusion criteria. Of the 36 teams, 16 (composed of 16 attendings and 48 residents and interns) were randomized to the intervention group, and 20 (composed of 20 attendings and 63 residents and interns) were randomized to the control group.
The study was blinded such that residents and attendings in the control group were unaware of the study. The study was exempt from IRB review by the Mount Sinai Institutional Review Board, and Grants and Contracts Office, as an evaluation of the effectiveness of an instructional technique in medical education.
Intervention Design
We designed a pocket feedback card to guide a feedback session and assist attendings in giving useful feedback to IM residents on the wards (Figure 1).14 The individual items and categories were adapted from the Accreditation Council for Graduate Medical Education (ACGME) Common Program Requirements Core Competencies section and were revised via the expert consensus of the authors.14 We included 20 items related to resident skills, knowledge, attitudes, and behaviors important to the care of hospitalized patients, grouped under the 6 ACGME core competency domains.14 Many of these items correspond to competencies in the Society of Hospital Medicine (SHM) Core Competencies; in particular, the categories of Systems‐Based Practice and Practice‐Based Learning mirror competencies in the SHM Core Competencies Healthcare Systems chapter.15 Each item utilized a 5‐point Likert scale (1 = very poor, 3 = at expected level, 5 = superior) to evaluate resident performance (Figure 1). We created this card to serve as a directive and specific guide for attendings to provide feedback about specific domains and to give more constructive feedback. The card was to be used during a specific dedicated feedback session in order to overcome the commonly cited barrier of lack of time.

Program Implementation
On the first day of the block, both groups of attendings received the standard inpatient ward orientation given by the program director, including instructions about teaching and administrative responsibilities, and explicit instructions to provide mid‐rotation feedback to residents. Attendings randomized to the intervention group had an additional 5‐minute orientation given by 1 of the investigators. The orientation included a brief discussion on the importance of feedback and an introduction to the items on the card.2 In addition, faculty were instructed to dedicate 1 mid‐rotation attending rounds as a feedback session, to meet individually for 10‐15 minutes with each of the 3‐4 residents on their team, and to use the card to provide feedback on skills in each domain. As noted on the feedback card, if a resident scored less than 3 on a skill set, the attending was instructed to give examples of skills within that domain needing improvement and to offer suggestions for improvement. The intervention group was also asked not to discuss the card or session with others. No other instructions were provided.
Survey Design
At the end of each block, residents and attendings in both groups completed questionnaires to assess satisfaction with, and attitudes toward, feedback (Supporting Information Appendices 1 and 2 in the online version of this article). Survey questions were based on the competency areas included in the feedback card, previously published surveys evaluating feedback interventions,5, 9, 11 and expert opinion. The resident survey was designed to address the impact of feedback on the domains of resident knowledge, clinical and communication skills, and attitudes about feedback from supervisors and peers. We utilized a 5‐point Likert scale including: strongly disagree, disagree, neutral, agree, and strongly agree. The attending survey addressed attendings' satisfaction with feedback encounters and resident performance. At the completion of the study, investigators compared responses in intervention and control groups.
Statistical Analysis
For purposes of analysis, due to the relatively small number of responses for certain answer choices, the Likert scale was converted to a dichotomous variable. The responses of agree and strongly agree were coded as agree; and disagree, strongly disagree, and neutral were coded as disagree. Neutral was coded as disagree in order to avoid overestimating positive attitudes and, in effect, bias our results toward the null hypothesis. Differences between groups were analyzed using chi‐square Fisher's exact test (2‐sided).
Qualitative Interviews
In order to understand the relative contribution of the feedback card versus the feedback session, we performed a qualitative survey of attendings in the intervention group. Following the conclusion of the study period, we selected a convenience sample of 8 attendings from the intervention group for these brief qualitative interviews. We asked 3 basic questions. Was the intervention of the feedback card and dedicated time for feedback useful? Did you find one component, either the card or the dedicated time for feedback, more useful than the other? Were there any negative effects on patient care, education, or other areas, from using an attending rounds as a feedback session? This data was coded and analyzed for common themes.
RESULTS
During the 6‐month study period, 34 teaching attendings (over 36 attending inpatient blocks) and 93 IM residents (over 111 resident inpatient blocks) participated in the study. Thirty‐four of 36 attending surveys and 96 of 111 resident surveys were completed. The overall survey response rates for residents and attendings were 85% and 94%, respectively. Two attendings participated during 2 separate blocks, first in the control group and then in the intervention group, and 18 residents participated during 2 separate blocks. No attendings or residents participated more than twice.
Resident survey response rate was 81.2% in the intervention group and 87.3% in the control group (Table 1). Residents in the intervention group reported receiving more feedback regarding skills they did well (89.7% vs 63.6%, P = 0.004) and skills needing improvement (51.3% vs 25.5%, P = 0.02) than those in the control group. In addition, more intervention residents reported receiving useful information regarding how to improve their skills (53.8% vs 27.3%, P = 0.01), and reported actually improving both their clinical skills (61.5% vs 27.8%, P = 0.001) and their professionalism/communication skills (51.3% vs 29.1%, P = 0.03) based on feedback received from attendings.
| Survey Item | Resident Intervention Agree* % (No.) N = 39 | Resident Control Agree*% (No.) N = 55 | P Value |
|---|---|---|---|
| |||
| I did NOT receive a sufficient amount of feedback from my attending supervisor(s) this block. | 20.5 (8) | 38.2 (21) | 0.08 |
| I received feedback from my attending regarding skills I did well during this block. | 89.7 (35) | 63.6 (35) | 0.004 |
| I received feedback from my attending regarding specific skills that needed improvement during this block. | 51.3 (20) | 25.5 (14) | 0.02 |
| I received useful information from my attending about how to improve my skills during this block. | 53.8 (21) | 27.3 (15) | 0.01 |
| I improved my clinical skills based on feedback I received from my attending this block. | 61.5 (24) | 27.8 (15) | 0.001 |
| I improved my professionalism/communication skills based on feedback I received from my attending this block. | 51.3 (20) | 29.1 (16) | 0.03 |
| I improved my knowledge base because of feedback I received from my attending this block. | 64.1 (25) | 60.0 (33) | 0.83 |
| The feedback I received from my attending this block gave me an overall sense of my performance more than it helped me identify specific areas for improvement. | 64.1 (25) | 65.5 (36) | 1.0 |
| Feedback from colleagues (other interns and residents) is more helpful than feedback from attendings. | 41.0 (16) | 43.6 (24) | 0.84 |
| Independent of feedback received from others, I am able to identify areas in which I need improvement. | 84.6 (33) | 80.0 (44) | 0.60 |
The attending survey response rates for the intervention and control groups were 100% and 90%, respectively. In general, both groups of attendings reported that they were comfortable giving feedback and that they did, in fact, give feedback in each area during their ward block (Table 2). More intervention attendings felt that at least 1 of their residents improved their professionalism/communication skills based on the feedback given (76.9% vs 31.1%, P = 0.02). There were no other significant differences between the groups of attendings.
| Survey Item | Attending Intervention Agree* % (No.) N = 16 | Attending Control Agree* % (No.) N = 18 | P Value |
|---|---|---|---|
| |||
| Giving feedback to housestaff was DIFFICULT this block. | 6.3 (1) | 16.7 (3) | 0.60 |
| I was comfortable giving feedback to my housestaff this block. | 93.8 (15) | 94.4 (17) | 1.00 |
| I did NOT give a sufficient amount of feedback to my housestaff this block. | 18.8 (3) | 38.9 (7) | 0.27 |
| My skills in giving feedback improved during this block. | 50 (8) | 16.7 (3) | 0.07 |
| I gave feedback to housestaff regarding skills they did well during this block. | 100 (16) | 94.4 (17) | 1.00 |
| I gave feedback to housestaff which targeted specific areas for their improvement. | 81.3 (13) | 70.6 (12) | 0.69 |
| At least one of my housestaff improved his/her clinical skills based on feedback I gave this block. | 68.8 (11) | 47.1 (8) | 0.30 |
| At least one of my housestaff improved his/her professionalism/communication skills based on feedback I gave this block. | 76.9 (10) | 31.1 (5) | 0.02 |
| At least one of my housestaff improved his/her fund of knowledge based on feedback I gave this block. | 50.0 (8) | 52.9 (9) | 1.00 |
| Housestaff found the feedback I gave them useful. | 66.7 (10) | 62.5 (10) | 1.00 |
| I find it DIFFICULT to find time during inpatient rotations to give feedback to residents regarding their performance. | 50.0 (8) | 33.3 (6) | 0.49 |
Intervention attendings also shared their attitudes toward the feedback card and session. A majority felt that using 1 attending rounds as a feedback session helped create a dedicated time for giving feedback (68.8%), and that the feedback card helped them to give specific, constructive feedback (62.5%). Most attendings reported they would use the feedback card and session again during future inpatient blocks (81%), and would recommend them to other attendings (75%).
Qualitative data from intervention attending interviews revealed further thoughts about the feedback card and feedback session. Most attendings interviewed (7/8) felt that the card was useful for the structure and topic guidance it provided. Half felt that setting aside time for feedback was the more useful component. The other half reported that, because they usually set aside time for feedback regardless, the card was more useful. None of the attendings felt that the feedback card or session was detrimental for patient care or education, and many said that the intervention had positive effects on these areas. For example, 1 attending said that the session added to patient care because I used particular [patient] cases as examples for giving feedback.
DISCUSSION
In this randomized study, we found that a simple pocket feedback card and dedicated feedback session was acceptable to ward attendings and improved resident satisfaction with feedback. Unlike most prior studies of feedback, we demonstrated more feedback around skills needing improvement, and intervention residents felt the feedback they received helped them improve their skills. Our educational intervention was unique in that it combined a pocket card to structure feedback content and dedicated time to structure the feedback process, to address 2 of the major barriers to giving feedback: lack of time and lack of comfort.
The pocket card itself as a tool for improving feedback is innovative and valuable. As a short but directive guide, the card supports attendings' delivery of relevant and specific feedback about residents' performance, and because it is based on the ACGME competencies, it may help attendings focus feedback on areas in which they will later evaluate residents. The inclusion of a prespecified time for giving feedback was important as well, in that it allowed for face‐to‐face feedback to occur, as opposed to a passing comment after a presentation or brief notes in a written final evaluation. Both the card and the feedback session seemed equally important for the success of this intervention, with attitudes varying based on individual attending preferences. Those who usually set aside time for feedback on their own found the card more useful, whereas those who had more trouble finding time for feedback found the specific session more useful. Most attendings found the intervention as a whole helpful, and without any detrimental effects on patient care or education. The card and session may be particularly valuable for hospital attendings, given their growing presence as teachers and supervisors for residents, and their busy days on the wards.
Our study results have important implications for resident training in the hospital. Improving resident receipt of feedback about strengths and weaknesses is an ACGME training requirement, and specific guidance about how to improve skills is critical for focusing improvement efforts. Previous studies have demonstrated that directive feedback in medical training can lead to a variety of performance improvements, including better evaluations by other professionals,9, 16 and objective improvements in resident communication skills,17 chart documentation,18 and clinical management of patients.11, 15, 19 By improving the quality of feedback across several domains and facilitating the feedback process, our intervention may lead to similar improvements. Future studies should examine the global impact of guided feedback as in our study. Perhaps most importantly, attendings found the intervention acceptable and would recommend its use, implying longer term sustainability of its integration into the hospital routine.
One strength of our study was its prospective randomized design. Despite the importance of rigor in medical education research, there remains a paucity of randomized studies to evaluate educational interventions for residents in inpatient settings. Few studies of feedback interventions in particular have performed randomized trials,5, 6, 11 and only one has examined a feedback intervention in a randomized fashion in the inpatient setting.12 This evaluation of a 20‐minute intervention, and a reminder card for supervising attendings to improve written and verbal feedback to residents, modestly improved the amount of verbal feedback given to residents, but did not impact the number of residents receiving mid‐rotation feedback or feedback overall as our study did by report.12
There were several important limitations to our study. First, because this was a single institution study, we only achieved modest sample sizes, particularly in the attending groups, and were unable to assess all of the differences in attending attitudes related to feedback. Second, control and intervention participants were on service simultaneously, which may have led to contamination of the control group and an underestimation of the true impact of our intervention. Since residents were not exclusive to 1 study group on 1 occasion (18 of the 93 residents participated during 2 separate blocks), our results may be biased. In particular, those residents who had the intervention first, and were subsequently in the control group, may have rated the control experience worse than they would have otherwise, creating a bias in favor of a positive result for our intervention. Nonetheless, we believe this situation was uncommon and the potential associated bias minimal. Further, this study assessed attitudes related to feedback and self‐reported knowledge and skills, but did not directly assess resident knowledge, skills, or patient outcomes. We recognize the importance of these outcomes and hope that future interventions can determine these important downstream effects of feedback. We were also unable to assess the card and session's impact on attendings' comfort with feedback, because most attendings in both groups reported feeling comfortable giving feedback. This result may indicate that attendings actually are comfortable giving feedback, or may suggest some element of social desirability bias. Finally, in this study, we designed an intervention which combined the pocket card and dedicated feedback time. We did not quantitatively examine the effect of either component alone, and it is unclear if offering the feedback card without protected time or offering protected time without a guide would have impacted feedback on the wards. However, qualitative data from our study support the use of both components, and implementing the 2 components together is feasible in any inpatient teaching setting.
Despite these limitations, protected time for feedback guided by a pocket feedback card is a simple intervention that appears to improve feedback quantity and quality for ward residents, and guides them to improve their performance. Our low‐intensity intervention helped attendings give residents the tools to improve their clinical and communication skills. An opportunity to make a positive impact on resident education with such a small intervention is rare. The use of a feedback card with protected feedback time could be easily implemented in any training program, and is a valuable tool for busy hospitalists who are more commonly supervising residents on their inpatient rotations.
- .Feedback in clinical medical education.JAMA.1983;250(6):777–781.
- ,.Giving feedback in medical education: verification of recommended techniques.J Gen Intern Med.1998;13(2):111–116.
- ,,,,.Systematic review of the literature on assessment, feedback and physicians' clinical performance: BEME Guide No. 7.Med Teach.2006;28(2):117–128.
- ,,,.Missed opportunities: a descriptive assessment of teaching and attitudes regarding communication skills in a surgical residency.Curr Surg.2006;63(6):401–409.
- ,,.Impact of a 360‐degree professionalism assessment on faculty comfort and skills in feedback delivery.J Gen Intern Med.2008;23(7):969–972.
- ,.Daily encounter cards facilitate competency‐based feedback while leniency bias persists.CJEM.2008;10(1):44–50.
- ,,,,.Teaching compassion and respect. Attending physicians' responses to problematic behaviors.J Gen Intern Med.1999;14(1):49–55.
- .Faculty and the observation of trainees' clinical skills: problems and opportunities.Acad Med.2004;79(1):16–22.
- ,.Direct observation of residents in the emergency department: a structured educational program.Acad Emerg Med.2009;16(4):343–351.
- ,,, et al.Evaluation of a novel assessment form for observing medical residents: a randomised, controlled trial.Med Educ.2008;42(12):1234–1242.
- ,,, et al.Resident evaluations: the use of daily evaluation forms in rheumatology ambulatory care.J Rheumatol.2009;36(6):1298–1303.
- ,,,.Effectiveness of a focused educational intervention on resident evaluations from faculty a randomized controlled trial.J Gen Intern Med.2001;16(7):427–434.
- ,,.Effects of training in direct observation of medical residents' clinical competence: a randomized trial.Ann Intern Med.2004;140(11):874–881.
- Internal Medicine Program Requirements. ACGME. July 1, 2009. Available at: http://www.acgme.org/acWebsite/downloads/RRC_progReq/140_internal_medicine_07012009.pdf. Accessed November 8,2009.
- ,,,,.How to use the core competencies in hospital medicine: a framework for curriculum development.J Hosp Med. 2006;1(suppl 1):57–67.
- ,,,,.Debriefing in the intensive care unit: a feedback tool to facilitate bedside teaching.Crit Care Med.2007;35(3):738–754.
- ,,, et al.Use of an innovative video feedback technique to enhance communication skills training.Med Educ.2004;38(2):145–157.
- .The impact of feedback to medical housestaff on chart documentation and quality of care in the outpatient setting.J Gen Intern Med.1997;12(6):352–356.
- ,,,.Feedback and the mini clinical evaluation exercise.J Gen Intern Med.2004;19(5 pt 2):558–561.
- .Feedback in clinical medical education.JAMA.1983;250(6):777–781.
- ,.Giving feedback in medical education: verification of recommended techniques.J Gen Intern Med.1998;13(2):111–116.
- ,,,,.Systematic review of the literature on assessment, feedback and physicians' clinical performance: BEME Guide No. 7.Med Teach.2006;28(2):117–128.
- ,,,.Missed opportunities: a descriptive assessment of teaching and attitudes regarding communication skills in a surgical residency.Curr Surg.2006;63(6):401–409.
- ,,.Impact of a 360‐degree professionalism assessment on faculty comfort and skills in feedback delivery.J Gen Intern Med.2008;23(7):969–972.
- ,.Daily encounter cards facilitate competency‐based feedback while leniency bias persists.CJEM.2008;10(1):44–50.
- ,,,,.Teaching compassion and respect. Attending physicians' responses to problematic behaviors.J Gen Intern Med.1999;14(1):49–55.
- .Faculty and the observation of trainees' clinical skills: problems and opportunities.Acad Med.2004;79(1):16–22.
- ,.Direct observation of residents in the emergency department: a structured educational program.Acad Emerg Med.2009;16(4):343–351.
- ,,, et al.Evaluation of a novel assessment form for observing medical residents: a randomised, controlled trial.Med Educ.2008;42(12):1234–1242.
- ,,, et al.Resident evaluations: the use of daily evaluation forms in rheumatology ambulatory care.J Rheumatol.2009;36(6):1298–1303.
- ,,,.Effectiveness of a focused educational intervention on resident evaluations from faculty a randomized controlled trial.J Gen Intern Med.2001;16(7):427–434.
- ,,.Effects of training in direct observation of medical residents' clinical competence: a randomized trial.Ann Intern Med.2004;140(11):874–881.
- Internal Medicine Program Requirements. ACGME. July 1, 2009. Available at: http://www.acgme.org/acWebsite/downloads/RRC_progReq/140_internal_medicine_07012009.pdf. Accessed November 8,2009.
- ,,,,.How to use the core competencies in hospital medicine: a framework for curriculum development.J Hosp Med. 2006;1(suppl 1):57–67.
- ,,,,.Debriefing in the intensive care unit: a feedback tool to facilitate bedside teaching.Crit Care Med.2007;35(3):738–754.
- ,,, et al.Use of an innovative video feedback technique to enhance communication skills training.Med Educ.2004;38(2):145–157.
- .The impact of feedback to medical housestaff on chart documentation and quality of care in the outpatient setting.J Gen Intern Med.1997;12(6):352–356.
- ,,,.Feedback and the mini clinical evaluation exercise.J Gen Intern Med.2004;19(5 pt 2):558–561.
Copyright © 2011 Society of Hospital Medicine
Thromboprophylaxis: Survey on Barriers
Each year in North America, over 7 million adults are hospitalized with a medical illness.1 Acute illness and decreased mobility in hospital places patients at increased risk for venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and life‐threatening pulmonary embolism (PE).2 Since VTE remains the most preventable cause of death in hospitalized patients, numerous studies have aimed at reducing the incidence of hospital‐acquired DVT. Aside from cost, the impact of VTE to the healthcare system is felt not only by those who diagnose and treat VTE, but also by those responsible for correcting the severe bleeding that can result from inappropriate use of thromboprophylaxis. Approximately 60% of symptomatic VTE occurs in medical patients, and recent hospitalization for medical illness accounts for 25% of all community‐diagnosed VTE. The Agency for Health Research and Quality ranks DVT prevention as the top priority out of 79 patient safety initiatives, and expert consensus groups provide a strong recommendation that DVT prophylaxis with a low‐dose anticoagulant should be administered to at‐risk hospitalized medical patients.2, 3
Despite the availability, efficacy, and safety of DVT prophylaxis,2 it is discouraging that only 21% to 62% of medical patients receive prophylaxis,49 and only 16% to 40% receive appropriate prophylaxis.46, 1012 However, 70% to 90% of patients in other at‐risk groups, such as surgical patients or critically ill patients, receive prophylaxis.1316 The reason why DVT prophylaxis is so underutilized in medical patients is unclear, as explanations for low rates of clinical practice guideline utilization are multifaceted,17 and few studies have investigated the barriers to optimal thromboprophylaxis.1820
To explore possible reasons for this disparity between evidence and practice, we conducted a cross‐sectional survey of 4 clinician groups involved in the care of hospitalized medical patients. Our objective was to identify barriers and potential solutions to the underutilization of DVT prophylaxis in hospitalized medical patients.
METHODS
Instrument Development
The survey focused on 3 domains: perceived importance, effectiveness, and safety of DVT prophylaxis; perceived barriers to implementation; and perceived potential success and feasibility of interventions to optimize DVT prophylaxis. The survey cover letter outlined background information, study design, and a statement on confidentiality. A prior survey of DVT prophylaxis administered to thrombosis experts was used to generate survey questions.21
Only survey respondents who answered yes to the first question, Are you involved in any aspect of the care of hospitalized general medical patients for whom DVT prophylaxis is considered? were asked to complete the remaining sections. Subsequent questions required respondents to check the box on a 7‐point Likert‐type scale that most accurately reflected their perception (Table 1). A successful intervention was defined as one that, if implemented, would yield the anticipated effect and a feasible intervention as one that was easy to implement without major logistical burden. Respondents were also asked which clinician group was best able to provide a daily assessment of patients' need for DVT prophylaxis, ensure DVT prophylaxis is prescribed, and ensure adherence.
|
| Section 1: Perceptions regarding DVT prophylaxis in hospitalized medical patients* |
| 1. How important an issue is the prevention of DVT in hospitalized general medical patients? |
| 2. To your knowledge, how effective are currently used anticoagulant strategies for the prevention of DVT in hospitalized medical patients? |
| 3. How safe are currently used anticoagulant strategies for the prevention of DVT in hospitalized medical patients? |
| 4. Current anticoagulant prophylaxis strategies are: 1 = underutilized, 4 = appropriately utilized, 7 = overutilized. |
| Section 2: Perceptions regarding barriers to the optimal use of DVT prophylaxis |
| 1. Lack of time to consider DVT prophylaxis in every patient |
| 2. Lack of clear indications for DVT prophylaxis (ie, who should get prophylaxis) |
| 3. Lack of clear contraindications for DVT prophylaxis (ie, who should not get prophylaxis) |
| 4. Lack of awareness about effectiveness of DVT prophylaxis |
| 5. Lack of physician agreement with current DVT prophylaxis guidelines |
| 6. Patient discomfort from subcutaneous injections of anticoagulants |
| 7. Clinician concerns about increased bleeding risk from anticoagulant administration |
| Section 3: Perceptions of interventions relating to DVT prophylaxis |
| 1. Yearly multidisciplinary educational meetings: to engage a wide spectrum of healthcare professionals to review DVT prophylaxis in hospitalized medical patients |
| 2. Posters on the wards: to remind healthcare professionals about DVT prophylaxis and patients who are eligible or ineligible for this treatment |
| 3. Laminated pocket cards: to remind healthcare professionals about DVT prophylaxis and patients who are eligible and ineligible for this treatment |
| 4. Preprinted order sheets: to remind healthcare professionals about DVT prophylaxis and patients who are eligible and ineligible for this treatment |
| 5. Periodic audit and feedback to healthcare providers: E‐mails to physicians containing reports on compliance with DVT prevention practice guidelines over recent years |
| 6. Computerized reminders (to the physicians): to prompt the physician to consider DVT prophylaxis upon opening a patient's electronic medical record |
| 7. Nurse reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 8. Pharmacist reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 9. Physiotherapist reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 10. Use of a local opinion leader (within the hospital) to promote evidence‐based use of DVT prophylaxis guidelines: to educate healthcare professionals on best practices for DVT prophylaxis |
Survey Administration
The survey was distributed between April and July 2007 in both paper‐based and web‐based formats using Survey Monkey software. Ontario members of the Canadian Society of Internal Medicine (n = 193) received a direct electronic invitation (from N.S.L., on behalf of J.D.D.) to participate, while members of the Canadian Society of Hospital Pharmacists (CSHP) (n = 1002) received an electronic invitation from an administrator for the CSHP to participate. The CSHP could not ensure that all members receiving the survey were hospital‐based pharmacists, so it was expected that the response rate from this group would be low. Nurse and physiotherapy managers at a convenience sample of 8 hospitals in Ontario, Canada, distributed paper‐based surveys to their staff using stamped, preaddressed envelopes. Nonresponders in all groups were sent reminders at 2 and 4 weeks.22 Data from all completed surveys were entered into an electronic database by a research coordinator (N.S.L.). A research assistant entered paper‐based survey data in duplicate, with discrepancies resolved by consensus and mediation by a third person (J.C.). The study was conducted with Institutional Ethics Review Board approval, and all respondents provided informed consent to participate. All responses were anonymous and confidential.
Statistical Considerations
Given the exploratory nature of this survey, there was no prespecified hypothesis‐driven respondent sample size. Proportions were used to describe response rates. Survey responses scored on the 7‐point Likert‐type scale were expressed as a mean and 95% confidence interval (CI). Important, highly potentially successful, and highly potentially feasible barriers were defined as those with a mean 5 points. Questions without responses, questions with multiple responses, and questions with illegible responses were treated as missing values. All statistical analyses were done using SAS version 9 (Cary, NC).
RESULTS
Survey Responses
The overall response rate was 36.3% (563/1553), with 65.5% (211/322) of nurses, 40.4% (78/193) of physicians, 24.1% (242/1002) of pharmacists, and 88.8% (32/36) of physiotherapists completing surveys. When pharmacists were removed from the response rate calculation (since it was expected that many of those receiving the survey were not in a primarily hospital‐based practice), the overall response rate rose to 58.3% (321/551). Excluded were 9.2% (52/563) of returned surveys, as respondents indicated the topic was not relevant to their practice. Five hundred eleven surveys were included in the final analysis (Figure 1).
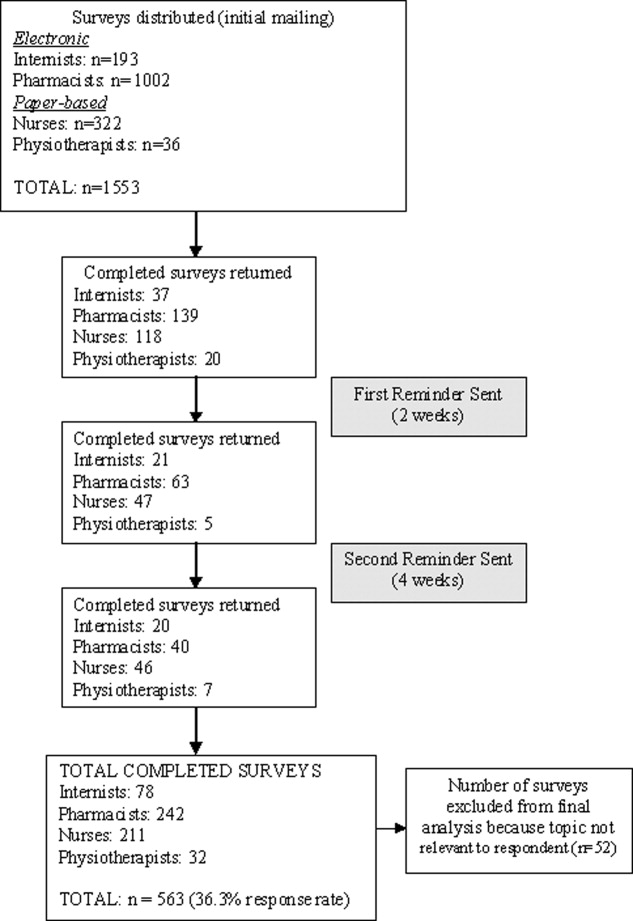
Importance, Effectiveness, Safety, and Appropriateness of DVT Prophylaxis Strategies
DVT prophylaxis was perceived across clinician groups as important (mean score 6.4; 95% CI 6.3 to 6.5), safe (mean 5.5; 95% CI 5.4 to 5.6), and effective (mean 5.5; 95% CI 5.4 to 6.6) (Figure 2). The mean score for the appropriateness of current DVT prophylaxis practices was 3.5 (95% CI 3.4 to 3.7), suggesting an overall perception of underutilization. However, by respondent groups, DVT prophylaxis was considered to be underutilized by physicians (mean 2.5; 95% CI 2.3 to 2.7) and pharmacists (mean 3.1; 95% CI 2.9 to 3.2), while nurses (mean 4.3; 95% CI 4.2 to 4.5) and physiotherapists (mean 3.8; 95% CI, 3.4 to 4.2) tended to consider current strategies as appropriate.
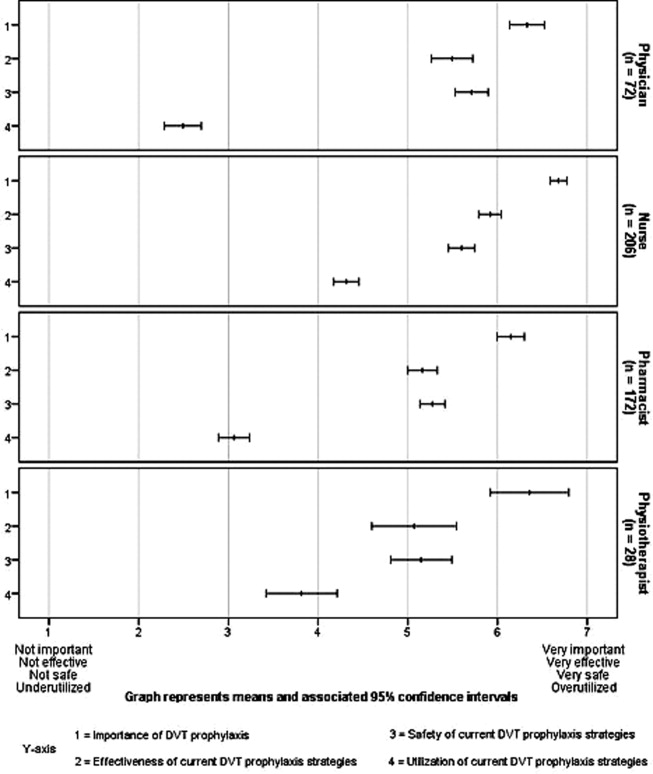
Potential Barriers to DVT Prophylaxis Utilization
Figure 3 demonstrates that no single barrier to DVT prophylaxis utilization was dominant and no barriers were considered very important. Perceived barriers carrying comparable weight were: concerns about bleeding (mean 4.8; 95% CI 4.6 to 4.9); lack of clear indications (mean 4.6; 95% CI 4.5 to 4.8) and contraindications to DVT prophylaxis (mean 4.4; 95% CI 4.3 to 4.6); lack of awareness about effectiveness of DVT prophylaxis (mean 4.5; 95% CI 4.3 to 4.7); and lack of time to consider DVT prophylaxis in every patient (mean 4.4; 95% CI 4.3 to 4.6). Patient discomfort from subcutaneous injections was perceived as the least important barrier (mean 3.8; 95% CI 3.6 to 4.0). Physicians perceived lack of awareness about the effectiveness of DVT prophylaxis as the most important barrier (mean 4.0; 95% CI 3.5 to 4.4), whereas concern about bleeding was dominant among non‐physicians (nurses' mean 5.2; 95% CI 5.0 to 5.5; pharmacists' mean 4.7; 95% CI 4.5 to 4.9; physiotherapists' mean 4.6; 95% CI 3.9 to 5.3).
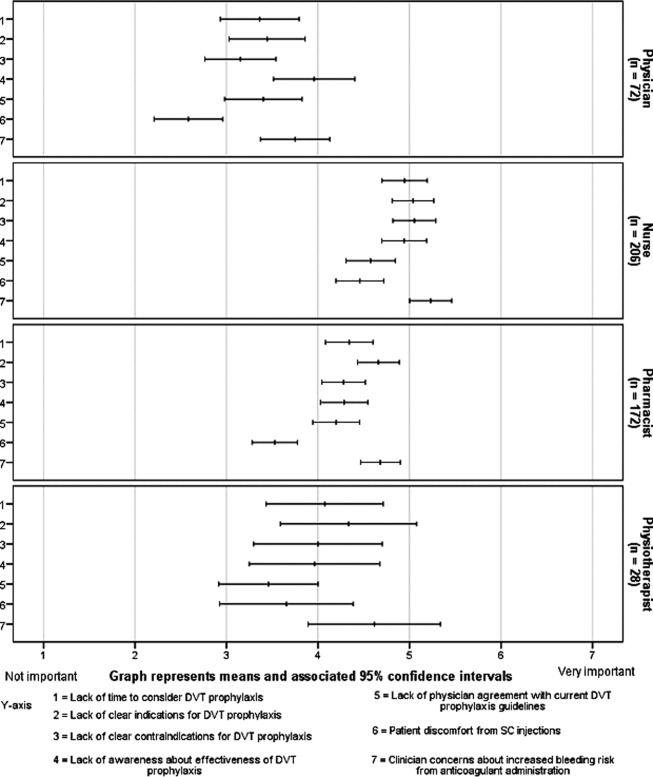
Potential Success and Feasibility of Interventions to Optimize DVT Prophylaxis Utilization
Interventions considered across clinician groups as highly potentially successful were: preprinted order sheets (5.7; 95% CI 5.6 to 5.8); pharmacist reminders to physicians (mean 5.3; 95% CI 5.1 to 5.4); computerized reminders to physicians (mean 5.0; 95% CI 4.9 to 5.2); and use of a local opinion leader (mean 5.0; 95% CI 4.9 to 5.2). Interventions considered highly potentially feasible were: posters (mean 5.7; CI 5.6 to 5.8); preprinted order sheets (mean 5.5; 95% CI 5.4 to 5.7); laminated pocket cards (mean 5.4; 95% CI 5.2 to 5.5); multidisciplinary educational meetings (mean 5.0; 95% CI 4.9 to 5.2); and pharmacist reminders to physicians (mean 5.0; 95% CI 4.9 to 5.1). Preprinted orders and pharmacist reminders were perceived by all clinician groups as having both high potential success and feasibility (Figure 4).
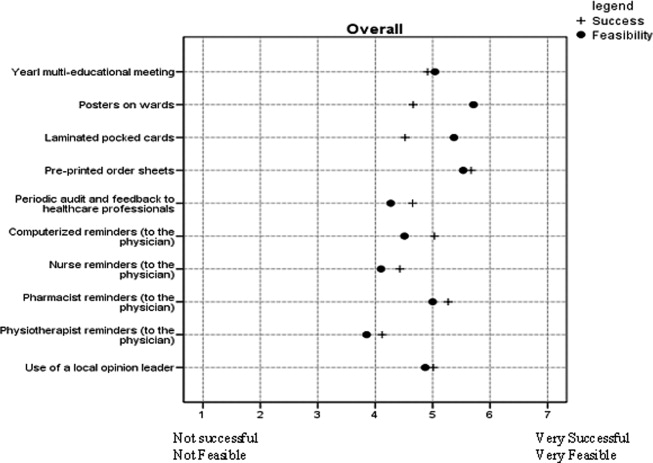
Perceptions on Which Clinician Group Is Best Able to Assess and Implement DVT Prophylaxis
Respondents were divided between considering the attending physician and the bedside nurse as best able to perform a daily assessment of patients' need for DVT prophylaxis (43.4% [204/470] vs 44.0% [207/470], respectively). Respondents from these groups each predominantly thought this responsibility was theirs, with 68.1% (49/72) of physicians and 61.5% (123/200) of nurses perceiving this as their responsibility (Figure 5).
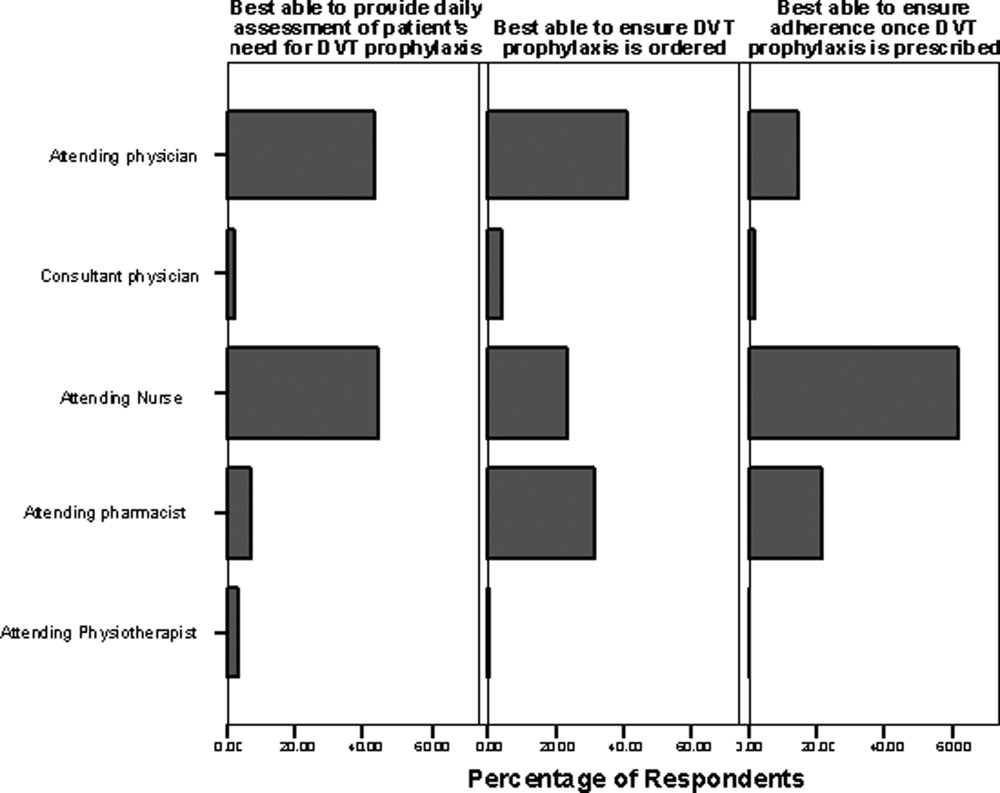
Forty‐one percent (193/471) of respondents perceived the attending physician as best able to ensure that DVT prophylaxis is ordered, while 31.2% (147/471) identified the pharmacist and 23.3% (110/471) identified the bedside nurse as best suited to this role. Among pharmacists, 66.3% (114/172) perceived that the attending pharmacist is best able to perform this task. Among respondents, 61.9% (296/478) felt the bedside nurse is best able to ensure adherence to DVT prophylaxis, with good agreement among all clinician groups.
DISCUSSION
Our survey identified several perceived barriers to optimizing DVT prophylaxis, consistent with those reported in the White Paper sponsored by the American Public Health Association.23 While no single barrier outlined in our survey was dominant, 2 novel barriers were identified: misperception of DVT prophylaxis underutilization, and confusion about roles and responsibilities in the area of DVT prophylaxis. Attention to these barriers may be helpful in developing an intervention aimed at bridging the gap between evidence and practice.
While our survey demonstrates agreement across clinician groups on the importance, efficacy, and safety of DVT prophylaxis, the discordant perceptions that exist about whether DVT prophylaxis is utilized appropriately is an important concern. Physician and pharmacist‐respondents demonstrated awareness that thromboprophylaxis is underutilized in medical patients. However, despite overwhelming published evidence to the contrary, nurses responding to our survey did not tend to recognize the problem of DVT prophylaxis underutilization in hospitalized medical patients. This knowledge deficit may be a significant barrier particularly since the pooled group of respondents indicated that nurses are among those caregivers best able to conduct a daily assessment of patients' need for DVT prophylaxis. A possible explanation for the finding that nurses and physiotherapists demonstrated a relative lack of awareness of the problem of DVT prophylaxis underutilization is ward‐specific healthcare priorities. Nursing and physiotherapy care on surgical wards is aimed at preventing postoperative complications, including DVT. However, its primary focus on medical wards is the management of acute medical problems. Prevention of hospital‐related complications, such as DVT, is often a secondary focus. Therefore, ensuring that all clinician groups are educated about the problem of DVT prophylaxis underutilization is necessary to drive quality improvement. A physician‐based survey on antithrombotic therapies demonstrated a similar need for education on guideline recommendations.20
A second important barrier identified in our survey is that both attending nurses and physicians feel that daily assessment of a patient's need for DVT prophylaxis is their responsibility. Confusion about roles and responsibilities in this area of patient care was reported by Cook et al., who identified that multidisciplinary care was perceived as a barrier to effective VTE prevention.18 Uncertainty as to which group should take ownership of DVT prophylaxis can lead to a diffusion of responsibility, a lack of accountability, and a gap in care. A resolution to whether DVT risk assessment is a nursing or a physician role could be reached through increased interdisciplinary communication and provision of clear definitions of roles to hospital staff.
Survey respondents felt that preprinted orders and pharmacist reminders to physicians were potentially successful and feasible strategies to optimize DVT prophylaxis. These components could be part of a simple tool to initiate prophylaxis. While electronic alerts have been shown to increase prophylaxis rates,24 we suspect that many respondents did not view these as highly important because of limited use of computerized order entry at their facilities. Interestingly, survey respondents did not perceive audit‐and‐feedback systems or local opinion leaders as potentially successful, though previous studies have demonstrated that they can change clinician behavior.25, 26 This may be because respondents may not be aware of the strength of technology‐based interventions (eg, electronic orders) and the role of opinion leaders, and the evidence in support of such interventions.24, 26 A systematic review of studies to improve DVT prophylaxis in hospitals reported that a combination of multiple active strategies is most effective, particularly those that link physician reminders with audit‐and‐feedback.27 For example, in the define study, a multicomponent intervention consisting of interactive educational sessions, verbal and computerized prompts, and individual performance feedback significantly improved adherence to DVT prophylaxis guidelines in critically ill patients.28 Whether a similar intervention could improve adherence to DVT prophylaxis guidelines in hospitalized medical patients merits further study. Any intervention must be paired with better education about which patients should, and should not, receive prophylaxis, as this may address many reported barriers in our survey (including concerns about bleeding). Respondents' uncertainty about these issues is not surprising, as studies of DVT prophylaxis in medical patients are not plentiful.2 However, recent guidelines do identify subgroups of medically ill patients in whom DVT prophylaxis is indicated.2 A clear and simple DVT risk assessment algorithm that identifies medical patients in whom DVT prophylaxis should (or should not) be administered may help to overcome respondents' concerns.
A limitation of our survey is the overall response rate of 36.3%, largely driven by the considerable number of nonresponding pharmacists (n = 760, reflecting 49% of the entire sample). However, the majority of the pharmacists were likely not hospital‐based, were thus not a target of this study, and their low response rate is not surprising. After excluding pharmacists, the response rate was 58.3% (321/551), which is consistent with response rates of other large‐sample surveys.29 The lower response rate for physicians and pharmacists may also reflect web‐based survey dissemination which, despite its feasibility, has lower response rates than paper‐based dissemination.3032 While the sample of physicians was relatively small compared to the other respondent groups surveyed, we aimed to identify barriers to actually implementing VTE prophylaxis, not just ordering prophylaxis, which is a multidisciplinary concern.
Although this survey was based on Canadian healthcare providers' perspectives, we believe the results are generalizable since both US and Canadian‐based studies have found that VTE prophylaxis is underutilized among hospitalized medical patients.4, 6 Furthermore, the American College of Chest Physicians (ACCP) guidelines on VTE prophylaxis, which are well‐recognized in both the United States and Canada, were developed with input from Canadian and American content experts.2 And while the US and Canadian healthcare systems are organized differently, at the patient‐care level, the roles of healthcare professionals are very similar. The generalizability of our findings is, however, limited by the institutional characteristics of respondents. We do not purport that the responses of any of the 4 clinician groups are generalizable to those groups as a whole. Although we surveyed clinicians in teaching and nonteaching, urban and rural practices, perceptions about DVT prophylaxis may be influenced by other factors, including the availability of local preprinted orders, electronic medical records, and quality improvement programs. Another potential limitation is that we did not assess all possible strategies to improve DVT prophylaxis, such as nurse practitioners and computerized decision support systems. These were purposely excluded, as they are not financially feasible in all centers, and thus not generalizable. Finally, like all self‐administered surveys, our findings reflect respondents' perceptions rather than objective observations about practice.
In conclusion, we identified novel and important barriers to optimal DVT prophylaxis utilization and potential interventions to address this important safety concern in hospitalized medical patients. To overcome some of these barriers, we propose an educational intervention prior to delivery of a top‐down, evidence‐based intervention to first increase healthcare providers' knowledge of the safety of DVT prophylaxis, system and team‐based approaches, and which interventions are most likely to be successful so as to encourage greater compliance with the intervention. A top‐down, system‐wide approach, involving the entire healthcare team and hospital administrators, can help drive this communication. As DVT prophylaxis becomes an increasingly important component in hospital accreditation, such solutions become appealing to facilitate change in practices. Results of this survey may inform future knowledge translation interventions by eliminating perceived barriers to DVT prophylaxis and by incorporating strategies that are perceived by healthcare professionals to be successful, feasible, and supported by evidence.
- ,.National hospital discharge survey: annual summary, 1996.Vital Health Stat.1999;13:1–46.
- ,,, et al.Prevention of venous thromboembolism: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th ed).Chest.2008;133:381S–443S.
- ,,, et al. Making health care safer: a critical analysis of patient safety practices. Evidence Report/Technology Assessment: No. 43. AHRQ Publication No. 01‐E058, July 2001. Rockville, MD: Agency for Healthcare Research and Quality. Available at: http://www.ahrq.gov/clinic/ptsafety/. Accessed October 9,2007.
- ,,, et al.Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada.Thromb Res.2007;119:145–155.
- ,,,.Hospitals' compliance with prophylaxis guidelines for venous thromboembolism.Am J Health Syst Pharm.2007;64:69–76.
- ,,,.Thromboprophylaxis rates in US medical centers: success or failure?J Thromb Haemost.2007;5:1610–1616.
- ,,.A retrospective evaluation of adherence to guidelines for prevention of thromboembolic events in general medical inpatients.Can J Hosp Pharm.2006;59:258–263.
- ,,, et al.Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism.Chest.2007;132:936–945.
- ,,,.Venous thromboembolism prophylaxis in medical inpatients: a retrospective chart review.Thromb Res.2003;111:215–219.
- ,,.Missed opportunities for prevention of venous thromboembolism: an evaluation of the use of DVT prophylaxis guidelines.Chest2001;120:1964–1971.
- ,,, et al.Thrombosis prophylaxis in medical patients: a retrospective review of clinical practice patterns.Haematologica.2002;87:746–750.
- ,,, et al.Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross‐sectional study.Lancet.2008;371:387–394.
- ,,,,.The use of low molecular weight heparins for the prevention of postoperative venous thromboembolism in general surgery. A survey of practice in the United States.Int Angiol.2002;1:78–85.
- ,,, et al.Venous thromboembolic disease management patterns in total hip arthroplasty and total knee arthroplasty patients: a survey of the AAHKS membership.J Arthroplasty.2001;6:679–688.
- ,,.Thromboprophylaxis in medical‐surgical intensive care unit patients.J Crit Care.2005;20:320–323.
- ,.Utilization of venous thromboembolism prophylaxis in a medical‐surgical ICU.Chest.1998;113:162–164.
- ,,, et al.Why don't physicians follow clinical practice guidelines? A framework for improvement.JAMA.1999;282:1458–1465.
- ,,, et al.Thromboprophylaxis for hospitalized medical patients: a multicenter qualitative study.J Hosp Med.2009;4;269–275.
- ,,, et al.Definition of immobility in studies of thromboprophylaxis in hospitalized medical patients: a systematic review.J Vasc Nurs.2010;28:54–66.
- ,,, et al.The use of antithrombotic therapies in the prevention and treatment of arterial and venous thrombosis: a survey of current knowledge and practice supporting the need for clinical education.Crit Pathw Cardiol.2010;9:41–48.
- ,,, et al.Antithrombotic and thrombolytic therapy: from evidence to application: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy.Chest.2004;126:688S–696S.
- .Mail and internet surveys: the tailored design method.New York, NY:John Wiley 2000.
- Deep‐vein thrombosis: advancing awareness to protect patient lives. Public Health Leadership Conference on Deep‐Vein Thrombosis. American Public Health Association. Available at: http://www.apha.org/NR/rdonlyres/A209F84A‐7C0E‐4761–9ECF‐61D22E1E11F7/0/DVT_White_Paper.pdf. Accessed May 28,2008.
- ,,, et al.Electronic alerts to prevent venous thromboembolism among hospitalized patients.N Engl J Med.2005;352:969–977.
- ,,, et al.Getting a validated guideline into local practice: implementation and audit of the SIGN guideline on the prevention of deep vein thrombosis in a district general hospital.Scott Med J.1998;43:23–25.
- ,,,.Local opinion leaders: effects on professional practice and health care outcomes.Cochrane Database Syst Rev.2007;24(1):CD000125.
- ,,, et al.A systematic review of strategies to improve prophylaxis for venous thromboembolism in hospitals.Ann Surg.2005;241:397–415.
- ,,, et al.Minimizing errors of omission: behavioural reinforcement of heparin to avert venous emboli: the BEHAVE study.Crit Care Med.2006;34:694–699.
- ,,,.Using the Internet to conduct surveys of health professionals: a valid alternative?Fam Pract.2003;20:545–551.
- ,,,,,.Use of new technology in endourology and laparoscopy by American urologists: Internet and postal survey.Urology.2000;56:760–765.
- ,,,,.E‐mail versus conventional postal mail survey of geriatric chiefs.Gerontologist.2001;41:799–804.
- ,,, et al.Internet versus mailed questionnaires: a randomized comparison.J Med Internet Res.2004;6:e30.
Each year in North America, over 7 million adults are hospitalized with a medical illness.1 Acute illness and decreased mobility in hospital places patients at increased risk for venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and life‐threatening pulmonary embolism (PE).2 Since VTE remains the most preventable cause of death in hospitalized patients, numerous studies have aimed at reducing the incidence of hospital‐acquired DVT. Aside from cost, the impact of VTE to the healthcare system is felt not only by those who diagnose and treat VTE, but also by those responsible for correcting the severe bleeding that can result from inappropriate use of thromboprophylaxis. Approximately 60% of symptomatic VTE occurs in medical patients, and recent hospitalization for medical illness accounts for 25% of all community‐diagnosed VTE. The Agency for Health Research and Quality ranks DVT prevention as the top priority out of 79 patient safety initiatives, and expert consensus groups provide a strong recommendation that DVT prophylaxis with a low‐dose anticoagulant should be administered to at‐risk hospitalized medical patients.2, 3
Despite the availability, efficacy, and safety of DVT prophylaxis,2 it is discouraging that only 21% to 62% of medical patients receive prophylaxis,49 and only 16% to 40% receive appropriate prophylaxis.46, 1012 However, 70% to 90% of patients in other at‐risk groups, such as surgical patients or critically ill patients, receive prophylaxis.1316 The reason why DVT prophylaxis is so underutilized in medical patients is unclear, as explanations for low rates of clinical practice guideline utilization are multifaceted,17 and few studies have investigated the barriers to optimal thromboprophylaxis.1820
To explore possible reasons for this disparity between evidence and practice, we conducted a cross‐sectional survey of 4 clinician groups involved in the care of hospitalized medical patients. Our objective was to identify barriers and potential solutions to the underutilization of DVT prophylaxis in hospitalized medical patients.
METHODS
Instrument Development
The survey focused on 3 domains: perceived importance, effectiveness, and safety of DVT prophylaxis; perceived barriers to implementation; and perceived potential success and feasibility of interventions to optimize DVT prophylaxis. The survey cover letter outlined background information, study design, and a statement on confidentiality. A prior survey of DVT prophylaxis administered to thrombosis experts was used to generate survey questions.21
Only survey respondents who answered yes to the first question, Are you involved in any aspect of the care of hospitalized general medical patients for whom DVT prophylaxis is considered? were asked to complete the remaining sections. Subsequent questions required respondents to check the box on a 7‐point Likert‐type scale that most accurately reflected their perception (Table 1). A successful intervention was defined as one that, if implemented, would yield the anticipated effect and a feasible intervention as one that was easy to implement without major logistical burden. Respondents were also asked which clinician group was best able to provide a daily assessment of patients' need for DVT prophylaxis, ensure DVT prophylaxis is prescribed, and ensure adherence.
|
| Section 1: Perceptions regarding DVT prophylaxis in hospitalized medical patients* |
| 1. How important an issue is the prevention of DVT in hospitalized general medical patients? |
| 2. To your knowledge, how effective are currently used anticoagulant strategies for the prevention of DVT in hospitalized medical patients? |
| 3. How safe are currently used anticoagulant strategies for the prevention of DVT in hospitalized medical patients? |
| 4. Current anticoagulant prophylaxis strategies are: 1 = underutilized, 4 = appropriately utilized, 7 = overutilized. |
| Section 2: Perceptions regarding barriers to the optimal use of DVT prophylaxis |
| 1. Lack of time to consider DVT prophylaxis in every patient |
| 2. Lack of clear indications for DVT prophylaxis (ie, who should get prophylaxis) |
| 3. Lack of clear contraindications for DVT prophylaxis (ie, who should not get prophylaxis) |
| 4. Lack of awareness about effectiveness of DVT prophylaxis |
| 5. Lack of physician agreement with current DVT prophylaxis guidelines |
| 6. Patient discomfort from subcutaneous injections of anticoagulants |
| 7. Clinician concerns about increased bleeding risk from anticoagulant administration |
| Section 3: Perceptions of interventions relating to DVT prophylaxis |
| 1. Yearly multidisciplinary educational meetings: to engage a wide spectrum of healthcare professionals to review DVT prophylaxis in hospitalized medical patients |
| 2. Posters on the wards: to remind healthcare professionals about DVT prophylaxis and patients who are eligible or ineligible for this treatment |
| 3. Laminated pocket cards: to remind healthcare professionals about DVT prophylaxis and patients who are eligible and ineligible for this treatment |
| 4. Preprinted order sheets: to remind healthcare professionals about DVT prophylaxis and patients who are eligible and ineligible for this treatment |
| 5. Periodic audit and feedback to healthcare providers: E‐mails to physicians containing reports on compliance with DVT prevention practice guidelines over recent years |
| 6. Computerized reminders (to the physicians): to prompt the physician to consider DVT prophylaxis upon opening a patient's electronic medical record |
| 7. Nurse reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 8. Pharmacist reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 9. Physiotherapist reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 10. Use of a local opinion leader (within the hospital) to promote evidence‐based use of DVT prophylaxis guidelines: to educate healthcare professionals on best practices for DVT prophylaxis |
Survey Administration
The survey was distributed between April and July 2007 in both paper‐based and web‐based formats using Survey Monkey software. Ontario members of the Canadian Society of Internal Medicine (n = 193) received a direct electronic invitation (from N.S.L., on behalf of J.D.D.) to participate, while members of the Canadian Society of Hospital Pharmacists (CSHP) (n = 1002) received an electronic invitation from an administrator for the CSHP to participate. The CSHP could not ensure that all members receiving the survey were hospital‐based pharmacists, so it was expected that the response rate from this group would be low. Nurse and physiotherapy managers at a convenience sample of 8 hospitals in Ontario, Canada, distributed paper‐based surveys to their staff using stamped, preaddressed envelopes. Nonresponders in all groups were sent reminders at 2 and 4 weeks.22 Data from all completed surveys were entered into an electronic database by a research coordinator (N.S.L.). A research assistant entered paper‐based survey data in duplicate, with discrepancies resolved by consensus and mediation by a third person (J.C.). The study was conducted with Institutional Ethics Review Board approval, and all respondents provided informed consent to participate. All responses were anonymous and confidential.
Statistical Considerations
Given the exploratory nature of this survey, there was no prespecified hypothesis‐driven respondent sample size. Proportions were used to describe response rates. Survey responses scored on the 7‐point Likert‐type scale were expressed as a mean and 95% confidence interval (CI). Important, highly potentially successful, and highly potentially feasible barriers were defined as those with a mean 5 points. Questions without responses, questions with multiple responses, and questions with illegible responses were treated as missing values. All statistical analyses were done using SAS version 9 (Cary, NC).
RESULTS
Survey Responses
The overall response rate was 36.3% (563/1553), with 65.5% (211/322) of nurses, 40.4% (78/193) of physicians, 24.1% (242/1002) of pharmacists, and 88.8% (32/36) of physiotherapists completing surveys. When pharmacists were removed from the response rate calculation (since it was expected that many of those receiving the survey were not in a primarily hospital‐based practice), the overall response rate rose to 58.3% (321/551). Excluded were 9.2% (52/563) of returned surveys, as respondents indicated the topic was not relevant to their practice. Five hundred eleven surveys were included in the final analysis (Figure 1).

Importance, Effectiveness, Safety, and Appropriateness of DVT Prophylaxis Strategies
DVT prophylaxis was perceived across clinician groups as important (mean score 6.4; 95% CI 6.3 to 6.5), safe (mean 5.5; 95% CI 5.4 to 5.6), and effective (mean 5.5; 95% CI 5.4 to 6.6) (Figure 2). The mean score for the appropriateness of current DVT prophylaxis practices was 3.5 (95% CI 3.4 to 3.7), suggesting an overall perception of underutilization. However, by respondent groups, DVT prophylaxis was considered to be underutilized by physicians (mean 2.5; 95% CI 2.3 to 2.7) and pharmacists (mean 3.1; 95% CI 2.9 to 3.2), while nurses (mean 4.3; 95% CI 4.2 to 4.5) and physiotherapists (mean 3.8; 95% CI, 3.4 to 4.2) tended to consider current strategies as appropriate.

Potential Barriers to DVT Prophylaxis Utilization
Figure 3 demonstrates that no single barrier to DVT prophylaxis utilization was dominant and no barriers were considered very important. Perceived barriers carrying comparable weight were: concerns about bleeding (mean 4.8; 95% CI 4.6 to 4.9); lack of clear indications (mean 4.6; 95% CI 4.5 to 4.8) and contraindications to DVT prophylaxis (mean 4.4; 95% CI 4.3 to 4.6); lack of awareness about effectiveness of DVT prophylaxis (mean 4.5; 95% CI 4.3 to 4.7); and lack of time to consider DVT prophylaxis in every patient (mean 4.4; 95% CI 4.3 to 4.6). Patient discomfort from subcutaneous injections was perceived as the least important barrier (mean 3.8; 95% CI 3.6 to 4.0). Physicians perceived lack of awareness about the effectiveness of DVT prophylaxis as the most important barrier (mean 4.0; 95% CI 3.5 to 4.4), whereas concern about bleeding was dominant among non‐physicians (nurses' mean 5.2; 95% CI 5.0 to 5.5; pharmacists' mean 4.7; 95% CI 4.5 to 4.9; physiotherapists' mean 4.6; 95% CI 3.9 to 5.3).

Potential Success and Feasibility of Interventions to Optimize DVT Prophylaxis Utilization
Interventions considered across clinician groups as highly potentially successful were: preprinted order sheets (5.7; 95% CI 5.6 to 5.8); pharmacist reminders to physicians (mean 5.3; 95% CI 5.1 to 5.4); computerized reminders to physicians (mean 5.0; 95% CI 4.9 to 5.2); and use of a local opinion leader (mean 5.0; 95% CI 4.9 to 5.2). Interventions considered highly potentially feasible were: posters (mean 5.7; CI 5.6 to 5.8); preprinted order sheets (mean 5.5; 95% CI 5.4 to 5.7); laminated pocket cards (mean 5.4; 95% CI 5.2 to 5.5); multidisciplinary educational meetings (mean 5.0; 95% CI 4.9 to 5.2); and pharmacist reminders to physicians (mean 5.0; 95% CI 4.9 to 5.1). Preprinted orders and pharmacist reminders were perceived by all clinician groups as having both high potential success and feasibility (Figure 4).

Perceptions on Which Clinician Group Is Best Able to Assess and Implement DVT Prophylaxis
Respondents were divided between considering the attending physician and the bedside nurse as best able to perform a daily assessment of patients' need for DVT prophylaxis (43.4% [204/470] vs 44.0% [207/470], respectively). Respondents from these groups each predominantly thought this responsibility was theirs, with 68.1% (49/72) of physicians and 61.5% (123/200) of nurses perceiving this as their responsibility (Figure 5).

Forty‐one percent (193/471) of respondents perceived the attending physician as best able to ensure that DVT prophylaxis is ordered, while 31.2% (147/471) identified the pharmacist and 23.3% (110/471) identified the bedside nurse as best suited to this role. Among pharmacists, 66.3% (114/172) perceived that the attending pharmacist is best able to perform this task. Among respondents, 61.9% (296/478) felt the bedside nurse is best able to ensure adherence to DVT prophylaxis, with good agreement among all clinician groups.
DISCUSSION
Our survey identified several perceived barriers to optimizing DVT prophylaxis, consistent with those reported in the White Paper sponsored by the American Public Health Association.23 While no single barrier outlined in our survey was dominant, 2 novel barriers were identified: misperception of DVT prophylaxis underutilization, and confusion about roles and responsibilities in the area of DVT prophylaxis. Attention to these barriers may be helpful in developing an intervention aimed at bridging the gap between evidence and practice.
While our survey demonstrates agreement across clinician groups on the importance, efficacy, and safety of DVT prophylaxis, the discordant perceptions that exist about whether DVT prophylaxis is utilized appropriately is an important concern. Physician and pharmacist‐respondents demonstrated awareness that thromboprophylaxis is underutilized in medical patients. However, despite overwhelming published evidence to the contrary, nurses responding to our survey did not tend to recognize the problem of DVT prophylaxis underutilization in hospitalized medical patients. This knowledge deficit may be a significant barrier particularly since the pooled group of respondents indicated that nurses are among those caregivers best able to conduct a daily assessment of patients' need for DVT prophylaxis. A possible explanation for the finding that nurses and physiotherapists demonstrated a relative lack of awareness of the problem of DVT prophylaxis underutilization is ward‐specific healthcare priorities. Nursing and physiotherapy care on surgical wards is aimed at preventing postoperative complications, including DVT. However, its primary focus on medical wards is the management of acute medical problems. Prevention of hospital‐related complications, such as DVT, is often a secondary focus. Therefore, ensuring that all clinician groups are educated about the problem of DVT prophylaxis underutilization is necessary to drive quality improvement. A physician‐based survey on antithrombotic therapies demonstrated a similar need for education on guideline recommendations.20
A second important barrier identified in our survey is that both attending nurses and physicians feel that daily assessment of a patient's need for DVT prophylaxis is their responsibility. Confusion about roles and responsibilities in this area of patient care was reported by Cook et al., who identified that multidisciplinary care was perceived as a barrier to effective VTE prevention.18 Uncertainty as to which group should take ownership of DVT prophylaxis can lead to a diffusion of responsibility, a lack of accountability, and a gap in care. A resolution to whether DVT risk assessment is a nursing or a physician role could be reached through increased interdisciplinary communication and provision of clear definitions of roles to hospital staff.
Survey respondents felt that preprinted orders and pharmacist reminders to physicians were potentially successful and feasible strategies to optimize DVT prophylaxis. These components could be part of a simple tool to initiate prophylaxis. While electronic alerts have been shown to increase prophylaxis rates,24 we suspect that many respondents did not view these as highly important because of limited use of computerized order entry at their facilities. Interestingly, survey respondents did not perceive audit‐and‐feedback systems or local opinion leaders as potentially successful, though previous studies have demonstrated that they can change clinician behavior.25, 26 This may be because respondents may not be aware of the strength of technology‐based interventions (eg, electronic orders) and the role of opinion leaders, and the evidence in support of such interventions.24, 26 A systematic review of studies to improve DVT prophylaxis in hospitals reported that a combination of multiple active strategies is most effective, particularly those that link physician reminders with audit‐and‐feedback.27 For example, in the define study, a multicomponent intervention consisting of interactive educational sessions, verbal and computerized prompts, and individual performance feedback significantly improved adherence to DVT prophylaxis guidelines in critically ill patients.28 Whether a similar intervention could improve adherence to DVT prophylaxis guidelines in hospitalized medical patients merits further study. Any intervention must be paired with better education about which patients should, and should not, receive prophylaxis, as this may address many reported barriers in our survey (including concerns about bleeding). Respondents' uncertainty about these issues is not surprising, as studies of DVT prophylaxis in medical patients are not plentiful.2 However, recent guidelines do identify subgroups of medically ill patients in whom DVT prophylaxis is indicated.2 A clear and simple DVT risk assessment algorithm that identifies medical patients in whom DVT prophylaxis should (or should not) be administered may help to overcome respondents' concerns.
A limitation of our survey is the overall response rate of 36.3%, largely driven by the considerable number of nonresponding pharmacists (n = 760, reflecting 49% of the entire sample). However, the majority of the pharmacists were likely not hospital‐based, were thus not a target of this study, and their low response rate is not surprising. After excluding pharmacists, the response rate was 58.3% (321/551), which is consistent with response rates of other large‐sample surveys.29 The lower response rate for physicians and pharmacists may also reflect web‐based survey dissemination which, despite its feasibility, has lower response rates than paper‐based dissemination.3032 While the sample of physicians was relatively small compared to the other respondent groups surveyed, we aimed to identify barriers to actually implementing VTE prophylaxis, not just ordering prophylaxis, which is a multidisciplinary concern.
Although this survey was based on Canadian healthcare providers' perspectives, we believe the results are generalizable since both US and Canadian‐based studies have found that VTE prophylaxis is underutilized among hospitalized medical patients.4, 6 Furthermore, the American College of Chest Physicians (ACCP) guidelines on VTE prophylaxis, which are well‐recognized in both the United States and Canada, were developed with input from Canadian and American content experts.2 And while the US and Canadian healthcare systems are organized differently, at the patient‐care level, the roles of healthcare professionals are very similar. The generalizability of our findings is, however, limited by the institutional characteristics of respondents. We do not purport that the responses of any of the 4 clinician groups are generalizable to those groups as a whole. Although we surveyed clinicians in teaching and nonteaching, urban and rural practices, perceptions about DVT prophylaxis may be influenced by other factors, including the availability of local preprinted orders, electronic medical records, and quality improvement programs. Another potential limitation is that we did not assess all possible strategies to improve DVT prophylaxis, such as nurse practitioners and computerized decision support systems. These were purposely excluded, as they are not financially feasible in all centers, and thus not generalizable. Finally, like all self‐administered surveys, our findings reflect respondents' perceptions rather than objective observations about practice.
In conclusion, we identified novel and important barriers to optimal DVT prophylaxis utilization and potential interventions to address this important safety concern in hospitalized medical patients. To overcome some of these barriers, we propose an educational intervention prior to delivery of a top‐down, evidence‐based intervention to first increase healthcare providers' knowledge of the safety of DVT prophylaxis, system and team‐based approaches, and which interventions are most likely to be successful so as to encourage greater compliance with the intervention. A top‐down, system‐wide approach, involving the entire healthcare team and hospital administrators, can help drive this communication. As DVT prophylaxis becomes an increasingly important component in hospital accreditation, such solutions become appealing to facilitate change in practices. Results of this survey may inform future knowledge translation interventions by eliminating perceived barriers to DVT prophylaxis and by incorporating strategies that are perceived by healthcare professionals to be successful, feasible, and supported by evidence.
Each year in North America, over 7 million adults are hospitalized with a medical illness.1 Acute illness and decreased mobility in hospital places patients at increased risk for venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and life‐threatening pulmonary embolism (PE).2 Since VTE remains the most preventable cause of death in hospitalized patients, numerous studies have aimed at reducing the incidence of hospital‐acquired DVT. Aside from cost, the impact of VTE to the healthcare system is felt not only by those who diagnose and treat VTE, but also by those responsible for correcting the severe bleeding that can result from inappropriate use of thromboprophylaxis. Approximately 60% of symptomatic VTE occurs in medical patients, and recent hospitalization for medical illness accounts for 25% of all community‐diagnosed VTE. The Agency for Health Research and Quality ranks DVT prevention as the top priority out of 79 patient safety initiatives, and expert consensus groups provide a strong recommendation that DVT prophylaxis with a low‐dose anticoagulant should be administered to at‐risk hospitalized medical patients.2, 3
Despite the availability, efficacy, and safety of DVT prophylaxis,2 it is discouraging that only 21% to 62% of medical patients receive prophylaxis,49 and only 16% to 40% receive appropriate prophylaxis.46, 1012 However, 70% to 90% of patients in other at‐risk groups, such as surgical patients or critically ill patients, receive prophylaxis.1316 The reason why DVT prophylaxis is so underutilized in medical patients is unclear, as explanations for low rates of clinical practice guideline utilization are multifaceted,17 and few studies have investigated the barriers to optimal thromboprophylaxis.1820
To explore possible reasons for this disparity between evidence and practice, we conducted a cross‐sectional survey of 4 clinician groups involved in the care of hospitalized medical patients. Our objective was to identify barriers and potential solutions to the underutilization of DVT prophylaxis in hospitalized medical patients.
METHODS
Instrument Development
The survey focused on 3 domains: perceived importance, effectiveness, and safety of DVT prophylaxis; perceived barriers to implementation; and perceived potential success and feasibility of interventions to optimize DVT prophylaxis. The survey cover letter outlined background information, study design, and a statement on confidentiality. A prior survey of DVT prophylaxis administered to thrombosis experts was used to generate survey questions.21
Only survey respondents who answered yes to the first question, Are you involved in any aspect of the care of hospitalized general medical patients for whom DVT prophylaxis is considered? were asked to complete the remaining sections. Subsequent questions required respondents to check the box on a 7‐point Likert‐type scale that most accurately reflected their perception (Table 1). A successful intervention was defined as one that, if implemented, would yield the anticipated effect and a feasible intervention as one that was easy to implement without major logistical burden. Respondents were also asked which clinician group was best able to provide a daily assessment of patients' need for DVT prophylaxis, ensure DVT prophylaxis is prescribed, and ensure adherence.
|
| Section 1: Perceptions regarding DVT prophylaxis in hospitalized medical patients* |
| 1. How important an issue is the prevention of DVT in hospitalized general medical patients? |
| 2. To your knowledge, how effective are currently used anticoagulant strategies for the prevention of DVT in hospitalized medical patients? |
| 3. How safe are currently used anticoagulant strategies for the prevention of DVT in hospitalized medical patients? |
| 4. Current anticoagulant prophylaxis strategies are: 1 = underutilized, 4 = appropriately utilized, 7 = overutilized. |
| Section 2: Perceptions regarding barriers to the optimal use of DVT prophylaxis |
| 1. Lack of time to consider DVT prophylaxis in every patient |
| 2. Lack of clear indications for DVT prophylaxis (ie, who should get prophylaxis) |
| 3. Lack of clear contraindications for DVT prophylaxis (ie, who should not get prophylaxis) |
| 4. Lack of awareness about effectiveness of DVT prophylaxis |
| 5. Lack of physician agreement with current DVT prophylaxis guidelines |
| 6. Patient discomfort from subcutaneous injections of anticoagulants |
| 7. Clinician concerns about increased bleeding risk from anticoagulant administration |
| Section 3: Perceptions of interventions relating to DVT prophylaxis |
| 1. Yearly multidisciplinary educational meetings: to engage a wide spectrum of healthcare professionals to review DVT prophylaxis in hospitalized medical patients |
| 2. Posters on the wards: to remind healthcare professionals about DVT prophylaxis and patients who are eligible or ineligible for this treatment |
| 3. Laminated pocket cards: to remind healthcare professionals about DVT prophylaxis and patients who are eligible and ineligible for this treatment |
| 4. Preprinted order sheets: to remind healthcare professionals about DVT prophylaxis and patients who are eligible and ineligible for this treatment |
| 5. Periodic audit and feedback to healthcare providers: E‐mails to physicians containing reports on compliance with DVT prevention practice guidelines over recent years |
| 6. Computerized reminders (to the physicians): to prompt the physician to consider DVT prophylaxis upon opening a patient's electronic medical record |
| 7. Nurse reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 8. Pharmacist reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 9. Physiotherapist reminders (to the physician): to remind the physician about DVT prophylaxis using written or verbal reminders |
| 10. Use of a local opinion leader (within the hospital) to promote evidence‐based use of DVT prophylaxis guidelines: to educate healthcare professionals on best practices for DVT prophylaxis |
Survey Administration
The survey was distributed between April and July 2007 in both paper‐based and web‐based formats using Survey Monkey software. Ontario members of the Canadian Society of Internal Medicine (n = 193) received a direct electronic invitation (from N.S.L., on behalf of J.D.D.) to participate, while members of the Canadian Society of Hospital Pharmacists (CSHP) (n = 1002) received an electronic invitation from an administrator for the CSHP to participate. The CSHP could not ensure that all members receiving the survey were hospital‐based pharmacists, so it was expected that the response rate from this group would be low. Nurse and physiotherapy managers at a convenience sample of 8 hospitals in Ontario, Canada, distributed paper‐based surveys to their staff using stamped, preaddressed envelopes. Nonresponders in all groups were sent reminders at 2 and 4 weeks.22 Data from all completed surveys were entered into an electronic database by a research coordinator (N.S.L.). A research assistant entered paper‐based survey data in duplicate, with discrepancies resolved by consensus and mediation by a third person (J.C.). The study was conducted with Institutional Ethics Review Board approval, and all respondents provided informed consent to participate. All responses were anonymous and confidential.
Statistical Considerations
Given the exploratory nature of this survey, there was no prespecified hypothesis‐driven respondent sample size. Proportions were used to describe response rates. Survey responses scored on the 7‐point Likert‐type scale were expressed as a mean and 95% confidence interval (CI). Important, highly potentially successful, and highly potentially feasible barriers were defined as those with a mean 5 points. Questions without responses, questions with multiple responses, and questions with illegible responses were treated as missing values. All statistical analyses were done using SAS version 9 (Cary, NC).
RESULTS
Survey Responses
The overall response rate was 36.3% (563/1553), with 65.5% (211/322) of nurses, 40.4% (78/193) of physicians, 24.1% (242/1002) of pharmacists, and 88.8% (32/36) of physiotherapists completing surveys. When pharmacists were removed from the response rate calculation (since it was expected that many of those receiving the survey were not in a primarily hospital‐based practice), the overall response rate rose to 58.3% (321/551). Excluded were 9.2% (52/563) of returned surveys, as respondents indicated the topic was not relevant to their practice. Five hundred eleven surveys were included in the final analysis (Figure 1).

Importance, Effectiveness, Safety, and Appropriateness of DVT Prophylaxis Strategies
DVT prophylaxis was perceived across clinician groups as important (mean score 6.4; 95% CI 6.3 to 6.5), safe (mean 5.5; 95% CI 5.4 to 5.6), and effective (mean 5.5; 95% CI 5.4 to 6.6) (Figure 2). The mean score for the appropriateness of current DVT prophylaxis practices was 3.5 (95% CI 3.4 to 3.7), suggesting an overall perception of underutilization. However, by respondent groups, DVT prophylaxis was considered to be underutilized by physicians (mean 2.5; 95% CI 2.3 to 2.7) and pharmacists (mean 3.1; 95% CI 2.9 to 3.2), while nurses (mean 4.3; 95% CI 4.2 to 4.5) and physiotherapists (mean 3.8; 95% CI, 3.4 to 4.2) tended to consider current strategies as appropriate.

Potential Barriers to DVT Prophylaxis Utilization
Figure 3 demonstrates that no single barrier to DVT prophylaxis utilization was dominant and no barriers were considered very important. Perceived barriers carrying comparable weight were: concerns about bleeding (mean 4.8; 95% CI 4.6 to 4.9); lack of clear indications (mean 4.6; 95% CI 4.5 to 4.8) and contraindications to DVT prophylaxis (mean 4.4; 95% CI 4.3 to 4.6); lack of awareness about effectiveness of DVT prophylaxis (mean 4.5; 95% CI 4.3 to 4.7); and lack of time to consider DVT prophylaxis in every patient (mean 4.4; 95% CI 4.3 to 4.6). Patient discomfort from subcutaneous injections was perceived as the least important barrier (mean 3.8; 95% CI 3.6 to 4.0). Physicians perceived lack of awareness about the effectiveness of DVT prophylaxis as the most important barrier (mean 4.0; 95% CI 3.5 to 4.4), whereas concern about bleeding was dominant among non‐physicians (nurses' mean 5.2; 95% CI 5.0 to 5.5; pharmacists' mean 4.7; 95% CI 4.5 to 4.9; physiotherapists' mean 4.6; 95% CI 3.9 to 5.3).

Potential Success and Feasibility of Interventions to Optimize DVT Prophylaxis Utilization
Interventions considered across clinician groups as highly potentially successful were: preprinted order sheets (5.7; 95% CI 5.6 to 5.8); pharmacist reminders to physicians (mean 5.3; 95% CI 5.1 to 5.4); computerized reminders to physicians (mean 5.0; 95% CI 4.9 to 5.2); and use of a local opinion leader (mean 5.0; 95% CI 4.9 to 5.2). Interventions considered highly potentially feasible were: posters (mean 5.7; CI 5.6 to 5.8); preprinted order sheets (mean 5.5; 95% CI 5.4 to 5.7); laminated pocket cards (mean 5.4; 95% CI 5.2 to 5.5); multidisciplinary educational meetings (mean 5.0; 95% CI 4.9 to 5.2); and pharmacist reminders to physicians (mean 5.0; 95% CI 4.9 to 5.1). Preprinted orders and pharmacist reminders were perceived by all clinician groups as having both high potential success and feasibility (Figure 4).

Perceptions on Which Clinician Group Is Best Able to Assess and Implement DVT Prophylaxis
Respondents were divided between considering the attending physician and the bedside nurse as best able to perform a daily assessment of patients' need for DVT prophylaxis (43.4% [204/470] vs 44.0% [207/470], respectively). Respondents from these groups each predominantly thought this responsibility was theirs, with 68.1% (49/72) of physicians and 61.5% (123/200) of nurses perceiving this as their responsibility (Figure 5).

Forty‐one percent (193/471) of respondents perceived the attending physician as best able to ensure that DVT prophylaxis is ordered, while 31.2% (147/471) identified the pharmacist and 23.3% (110/471) identified the bedside nurse as best suited to this role. Among pharmacists, 66.3% (114/172) perceived that the attending pharmacist is best able to perform this task. Among respondents, 61.9% (296/478) felt the bedside nurse is best able to ensure adherence to DVT prophylaxis, with good agreement among all clinician groups.
DISCUSSION
Our survey identified several perceived barriers to optimizing DVT prophylaxis, consistent with those reported in the White Paper sponsored by the American Public Health Association.23 While no single barrier outlined in our survey was dominant, 2 novel barriers were identified: misperception of DVT prophylaxis underutilization, and confusion about roles and responsibilities in the area of DVT prophylaxis. Attention to these barriers may be helpful in developing an intervention aimed at bridging the gap between evidence and practice.
While our survey demonstrates agreement across clinician groups on the importance, efficacy, and safety of DVT prophylaxis, the discordant perceptions that exist about whether DVT prophylaxis is utilized appropriately is an important concern. Physician and pharmacist‐respondents demonstrated awareness that thromboprophylaxis is underutilized in medical patients. However, despite overwhelming published evidence to the contrary, nurses responding to our survey did not tend to recognize the problem of DVT prophylaxis underutilization in hospitalized medical patients. This knowledge deficit may be a significant barrier particularly since the pooled group of respondents indicated that nurses are among those caregivers best able to conduct a daily assessment of patients' need for DVT prophylaxis. A possible explanation for the finding that nurses and physiotherapists demonstrated a relative lack of awareness of the problem of DVT prophylaxis underutilization is ward‐specific healthcare priorities. Nursing and physiotherapy care on surgical wards is aimed at preventing postoperative complications, including DVT. However, its primary focus on medical wards is the management of acute medical problems. Prevention of hospital‐related complications, such as DVT, is often a secondary focus. Therefore, ensuring that all clinician groups are educated about the problem of DVT prophylaxis underutilization is necessary to drive quality improvement. A physician‐based survey on antithrombotic therapies demonstrated a similar need for education on guideline recommendations.20
A second important barrier identified in our survey is that both attending nurses and physicians feel that daily assessment of a patient's need for DVT prophylaxis is their responsibility. Confusion about roles and responsibilities in this area of patient care was reported by Cook et al., who identified that multidisciplinary care was perceived as a barrier to effective VTE prevention.18 Uncertainty as to which group should take ownership of DVT prophylaxis can lead to a diffusion of responsibility, a lack of accountability, and a gap in care. A resolution to whether DVT risk assessment is a nursing or a physician role could be reached through increased interdisciplinary communication and provision of clear definitions of roles to hospital staff.
Survey respondents felt that preprinted orders and pharmacist reminders to physicians were potentially successful and feasible strategies to optimize DVT prophylaxis. These components could be part of a simple tool to initiate prophylaxis. While electronic alerts have been shown to increase prophylaxis rates,24 we suspect that many respondents did not view these as highly important because of limited use of computerized order entry at their facilities. Interestingly, survey respondents did not perceive audit‐and‐feedback systems or local opinion leaders as potentially successful, though previous studies have demonstrated that they can change clinician behavior.25, 26 This may be because respondents may not be aware of the strength of technology‐based interventions (eg, electronic orders) and the role of opinion leaders, and the evidence in support of such interventions.24, 26 A systematic review of studies to improve DVT prophylaxis in hospitals reported that a combination of multiple active strategies is most effective, particularly those that link physician reminders with audit‐and‐feedback.27 For example, in the define study, a multicomponent intervention consisting of interactive educational sessions, verbal and computerized prompts, and individual performance feedback significantly improved adherence to DVT prophylaxis guidelines in critically ill patients.28 Whether a similar intervention could improve adherence to DVT prophylaxis guidelines in hospitalized medical patients merits further study. Any intervention must be paired with better education about which patients should, and should not, receive prophylaxis, as this may address many reported barriers in our survey (including concerns about bleeding). Respondents' uncertainty about these issues is not surprising, as studies of DVT prophylaxis in medical patients are not plentiful.2 However, recent guidelines do identify subgroups of medically ill patients in whom DVT prophylaxis is indicated.2 A clear and simple DVT risk assessment algorithm that identifies medical patients in whom DVT prophylaxis should (or should not) be administered may help to overcome respondents' concerns.
A limitation of our survey is the overall response rate of 36.3%, largely driven by the considerable number of nonresponding pharmacists (n = 760, reflecting 49% of the entire sample). However, the majority of the pharmacists were likely not hospital‐based, were thus not a target of this study, and their low response rate is not surprising. After excluding pharmacists, the response rate was 58.3% (321/551), which is consistent with response rates of other large‐sample surveys.29 The lower response rate for physicians and pharmacists may also reflect web‐based survey dissemination which, despite its feasibility, has lower response rates than paper‐based dissemination.3032 While the sample of physicians was relatively small compared to the other respondent groups surveyed, we aimed to identify barriers to actually implementing VTE prophylaxis, not just ordering prophylaxis, which is a multidisciplinary concern.
Although this survey was based on Canadian healthcare providers' perspectives, we believe the results are generalizable since both US and Canadian‐based studies have found that VTE prophylaxis is underutilized among hospitalized medical patients.4, 6 Furthermore, the American College of Chest Physicians (ACCP) guidelines on VTE prophylaxis, which are well‐recognized in both the United States and Canada, were developed with input from Canadian and American content experts.2 And while the US and Canadian healthcare systems are organized differently, at the patient‐care level, the roles of healthcare professionals are very similar. The generalizability of our findings is, however, limited by the institutional characteristics of respondents. We do not purport that the responses of any of the 4 clinician groups are generalizable to those groups as a whole. Although we surveyed clinicians in teaching and nonteaching, urban and rural practices, perceptions about DVT prophylaxis may be influenced by other factors, including the availability of local preprinted orders, electronic medical records, and quality improvement programs. Another potential limitation is that we did not assess all possible strategies to improve DVT prophylaxis, such as nurse practitioners and computerized decision support systems. These were purposely excluded, as they are not financially feasible in all centers, and thus not generalizable. Finally, like all self‐administered surveys, our findings reflect respondents' perceptions rather than objective observations about practice.
In conclusion, we identified novel and important barriers to optimal DVT prophylaxis utilization and potential interventions to address this important safety concern in hospitalized medical patients. To overcome some of these barriers, we propose an educational intervention prior to delivery of a top‐down, evidence‐based intervention to first increase healthcare providers' knowledge of the safety of DVT prophylaxis, system and team‐based approaches, and which interventions are most likely to be successful so as to encourage greater compliance with the intervention. A top‐down, system‐wide approach, involving the entire healthcare team and hospital administrators, can help drive this communication. As DVT prophylaxis becomes an increasingly important component in hospital accreditation, such solutions become appealing to facilitate change in practices. Results of this survey may inform future knowledge translation interventions by eliminating perceived barriers to DVT prophylaxis and by incorporating strategies that are perceived by healthcare professionals to be successful, feasible, and supported by evidence.
- ,.National hospital discharge survey: annual summary, 1996.Vital Health Stat.1999;13:1–46.
- ,,, et al.Prevention of venous thromboembolism: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th ed).Chest.2008;133:381S–443S.
- ,,, et al. Making health care safer: a critical analysis of patient safety practices. Evidence Report/Technology Assessment: No. 43. AHRQ Publication No. 01‐E058, July 2001. Rockville, MD: Agency for Healthcare Research and Quality. Available at: http://www.ahrq.gov/clinic/ptsafety/. Accessed October 9,2007.
- ,,, et al.Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada.Thromb Res.2007;119:145–155.
- ,,,.Hospitals' compliance with prophylaxis guidelines for venous thromboembolism.Am J Health Syst Pharm.2007;64:69–76.
- ,,,.Thromboprophylaxis rates in US medical centers: success or failure?J Thromb Haemost.2007;5:1610–1616.
- ,,.A retrospective evaluation of adherence to guidelines for prevention of thromboembolic events in general medical inpatients.Can J Hosp Pharm.2006;59:258–263.
- ,,, et al.Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism.Chest.2007;132:936–945.
- ,,,.Venous thromboembolism prophylaxis in medical inpatients: a retrospective chart review.Thromb Res.2003;111:215–219.
- ,,.Missed opportunities for prevention of venous thromboembolism: an evaluation of the use of DVT prophylaxis guidelines.Chest2001;120:1964–1971.
- ,,, et al.Thrombosis prophylaxis in medical patients: a retrospective review of clinical practice patterns.Haematologica.2002;87:746–750.
- ,,, et al.Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross‐sectional study.Lancet.2008;371:387–394.
- ,,,,.The use of low molecular weight heparins for the prevention of postoperative venous thromboembolism in general surgery. A survey of practice in the United States.Int Angiol.2002;1:78–85.
- ,,, et al.Venous thromboembolic disease management patterns in total hip arthroplasty and total knee arthroplasty patients: a survey of the AAHKS membership.J Arthroplasty.2001;6:679–688.
- ,,.Thromboprophylaxis in medical‐surgical intensive care unit patients.J Crit Care.2005;20:320–323.
- ,.Utilization of venous thromboembolism prophylaxis in a medical‐surgical ICU.Chest.1998;113:162–164.
- ,,, et al.Why don't physicians follow clinical practice guidelines? A framework for improvement.JAMA.1999;282:1458–1465.
- ,,, et al.Thromboprophylaxis for hospitalized medical patients: a multicenter qualitative study.J Hosp Med.2009;4;269–275.
- ,,, et al.Definition of immobility in studies of thromboprophylaxis in hospitalized medical patients: a systematic review.J Vasc Nurs.2010;28:54–66.
- ,,, et al.The use of antithrombotic therapies in the prevention and treatment of arterial and venous thrombosis: a survey of current knowledge and practice supporting the need for clinical education.Crit Pathw Cardiol.2010;9:41–48.
- ,,, et al.Antithrombotic and thrombolytic therapy: from evidence to application: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy.Chest.2004;126:688S–696S.
- .Mail and internet surveys: the tailored design method.New York, NY:John Wiley 2000.
- Deep‐vein thrombosis: advancing awareness to protect patient lives. Public Health Leadership Conference on Deep‐Vein Thrombosis. American Public Health Association. Available at: http://www.apha.org/NR/rdonlyres/A209F84A‐7C0E‐4761–9ECF‐61D22E1E11F7/0/DVT_White_Paper.pdf. Accessed May 28,2008.
- ,,, et al.Electronic alerts to prevent venous thromboembolism among hospitalized patients.N Engl J Med.2005;352:969–977.
- ,,, et al.Getting a validated guideline into local practice: implementation and audit of the SIGN guideline on the prevention of deep vein thrombosis in a district general hospital.Scott Med J.1998;43:23–25.
- ,,,.Local opinion leaders: effects on professional practice and health care outcomes.Cochrane Database Syst Rev.2007;24(1):CD000125.
- ,,, et al.A systematic review of strategies to improve prophylaxis for venous thromboembolism in hospitals.Ann Surg.2005;241:397–415.
- ,,, et al.Minimizing errors of omission: behavioural reinforcement of heparin to avert venous emboli: the BEHAVE study.Crit Care Med.2006;34:694–699.
- ,,,.Using the Internet to conduct surveys of health professionals: a valid alternative?Fam Pract.2003;20:545–551.
- ,,,,,.Use of new technology in endourology and laparoscopy by American urologists: Internet and postal survey.Urology.2000;56:760–765.
- ,,,,.E‐mail versus conventional postal mail survey of geriatric chiefs.Gerontologist.2001;41:799–804.
- ,,, et al.Internet versus mailed questionnaires: a randomized comparison.J Med Internet Res.2004;6:e30.
- ,.National hospital discharge survey: annual summary, 1996.Vital Health Stat.1999;13:1–46.
- ,,, et al.Prevention of venous thromboembolism: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th ed).Chest.2008;133:381S–443S.
- ,,, et al. Making health care safer: a critical analysis of patient safety practices. Evidence Report/Technology Assessment: No. 43. AHRQ Publication No. 01‐E058, July 2001. Rockville, MD: Agency for Healthcare Research and Quality. Available at: http://www.ahrq.gov/clinic/ptsafety/. Accessed October 9,2007.
- ,,, et al.Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada.Thromb Res.2007;119:145–155.
- ,,,.Hospitals' compliance with prophylaxis guidelines for venous thromboembolism.Am J Health Syst Pharm.2007;64:69–76.
- ,,,.Thromboprophylaxis rates in US medical centers: success or failure?J Thromb Haemost.2007;5:1610–1616.
- ,,.A retrospective evaluation of adherence to guidelines for prevention of thromboembolic events in general medical inpatients.Can J Hosp Pharm.2006;59:258–263.
- ,,, et al.Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism.Chest.2007;132:936–945.
- ,,,.Venous thromboembolism prophylaxis in medical inpatients: a retrospective chart review.Thromb Res.2003;111:215–219.
- ,,.Missed opportunities for prevention of venous thromboembolism: an evaluation of the use of DVT prophylaxis guidelines.Chest2001;120:1964–1971.
- ,,, et al.Thrombosis prophylaxis in medical patients: a retrospective review of clinical practice patterns.Haematologica.2002;87:746–750.
- ,,, et al.Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross‐sectional study.Lancet.2008;371:387–394.
- ,,,,.The use of low molecular weight heparins for the prevention of postoperative venous thromboembolism in general surgery. A survey of practice in the United States.Int Angiol.2002;1:78–85.
- ,,, et al.Venous thromboembolic disease management patterns in total hip arthroplasty and total knee arthroplasty patients: a survey of the AAHKS membership.J Arthroplasty.2001;6:679–688.
- ,,.Thromboprophylaxis in medical‐surgical intensive care unit patients.J Crit Care.2005;20:320–323.
- ,.Utilization of venous thromboembolism prophylaxis in a medical‐surgical ICU.Chest.1998;113:162–164.
- ,,, et al.Why don't physicians follow clinical practice guidelines? A framework for improvement.JAMA.1999;282:1458–1465.
- ,,, et al.Thromboprophylaxis for hospitalized medical patients: a multicenter qualitative study.J Hosp Med.2009;4;269–275.
- ,,, et al.Definition of immobility in studies of thromboprophylaxis in hospitalized medical patients: a systematic review.J Vasc Nurs.2010;28:54–66.
- ,,, et al.The use of antithrombotic therapies in the prevention and treatment of arterial and venous thrombosis: a survey of current knowledge and practice supporting the need for clinical education.Crit Pathw Cardiol.2010;9:41–48.
- ,,, et al.Antithrombotic and thrombolytic therapy: from evidence to application: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy.Chest.2004;126:688S–696S.
- .Mail and internet surveys: the tailored design method.New York, NY:John Wiley 2000.
- Deep‐vein thrombosis: advancing awareness to protect patient lives. Public Health Leadership Conference on Deep‐Vein Thrombosis. American Public Health Association. Available at: http://www.apha.org/NR/rdonlyres/A209F84A‐7C0E‐4761–9ECF‐61D22E1E11F7/0/DVT_White_Paper.pdf. Accessed May 28,2008.
- ,,, et al.Electronic alerts to prevent venous thromboembolism among hospitalized patients.N Engl J Med.2005;352:969–977.
- ,,, et al.Getting a validated guideline into local practice: implementation and audit of the SIGN guideline on the prevention of deep vein thrombosis in a district general hospital.Scott Med J.1998;43:23–25.
- ,,,.Local opinion leaders: effects on professional practice and health care outcomes.Cochrane Database Syst Rev.2007;24(1):CD000125.
- ,,, et al.A systematic review of strategies to improve prophylaxis for venous thromboembolism in hospitals.Ann Surg.2005;241:397–415.
- ,,, et al.Minimizing errors of omission: behavioural reinforcement of heparin to avert venous emboli: the BEHAVE study.Crit Care Med.2006;34:694–699.
- ,,,.Using the Internet to conduct surveys of health professionals: a valid alternative?Fam Pract.2003;20:545–551.
- ,,,,,.Use of new technology in endourology and laparoscopy by American urologists: Internet and postal survey.Urology.2000;56:760–765.
- ,,,,.E‐mail versus conventional postal mail survey of geriatric chiefs.Gerontologist.2001;41:799–804.
- ,,, et al.Internet versus mailed questionnaires: a randomized comparison.J Med Internet Res.2004;6:e30.
Copyright © 2011 Society of Hospital Medicine
Accuracy of GoogleTranslate™
The population of patients in the US with limited English proficiency (LEP)those who speak English less than very well1is substantial and continues to grow.1, 2 Patients with LEP are at risk for lower quality health care overall than their English‐speaking counterparts.38 Professional in‐person interpreters greatly improve spoken communication and quality of care for these patients,4, 9 but their assistance is typically based on the clinical encounter. Particularly if interpreting by phone, interpreters are unlikely to be able to help with materials such as discharge instructions or information sheets meant for family members. Professional written translations of patient educational material help to bridge this gap, allowing clinicians to convey detailed written instructions to patients. However, professional translations must be prepared well in advance of any encounter and can only be used for easily anticipated problems.
The need to translate less common, patient‐specific instructions arises spontaneously in clinical practice, and formally prepared written translations are not useful in these situations. Online translation tools such as GoogleTranslate (available at
We conducted a pilot evaluation of an online translation tool as it relates to detailed, complex patient educational material. Our primary goal was to compare the accuracy of a Spanish translation generated by the online tool to that done by a professional agency. Our secondary goals were: 1) to assess whether sentence word length or complexity mediated the accuracy of GT; and 2) to lay the foundation for a more comprehensive study of the accuracy of online translation tools, with respect to patient educational material.
Methods
Translation Tool and Language Choice
We selected Google Translate (GT) since it is one of the more commonly used online translation tools and because Google is the most widely used search engine in the United States.13 GT uses statistical translation methodology to convert text, documents, and websites between languages; statistical translation involves the following three steps. First, the translation program recognizes a sentence to translate. Second, it compares the words and phrases within that sentence to the billions of words in its library (drawn from bilingual professionally translated documents, such as United Nations proceedings). Third, it uses this comparison to generate a translation combining the words and phrases deemed most equivalent between the source sentence and the target language. If there are multiple sentences, the program recognizes and translates each independently. As the body of bilingual work grows, the program learns and refines its rules automatically.14 In contrast, in rule‐based translation, a program would use manually prespecified rules regarding word choice and grammar to generate a translation.15 We assessed GT's accuracy translating from English to Spanish because Spanish is the predominant non‐English language spoken in the US.1
Document Selection and Preparation
We selected the instruction manual regarding warfarin use prepared by the Agency for Healthcare Research and Quality (AHRQ) for this accuracy evaluation. We selected this manual,16 written at a 6th grade reading level, because a professional Spanish translation was available (completed by ASET International Service, LLC, before and independently of this study), and because patient educational material regarding warfarin has been associated with fewer bleeding events.17 We downloaded the English document on October 19, 2009 and used the GT website to translate it en bloc. We then copied the resulting Spanish output into a text file. The English document and the professional Spanish translation (downloaded the same day) were both converted into text files in the same manner.
Grading Methodology
We scored the translation chosen using both manual and automated evaluation techniques. These techniques are widely used in the machine translation literature and are explained below.
Manual Evaluation: Evaluators, Domains, Scoring
We recruited three nonclinician, bilingual, nativeSpanish‐speaking research assistants as evaluators. The evaluators were all college educated with a Bachelor's degree or higher and were of Mexican, Nicaraguan, and Guatemalan ancestry. Each evaluator received a brief orientation regarding the project, as well as an explanation of the scores, and then proceeded to the blinded evaluation independently.
We asked evaluators to score sentences on Likert scales along five primary domains: fluency, adequacy, meaning, severity, and preference. Fluency and adequacy are well accepted components of machine translation evaluation,18 with fluency being an assessment of grammar and readability ranging from 5 (Perfect fluency; like reading a newspaper) to 1 (No fluency; no appreciable grammar, not understandable) and adequacy being an assessment of information preservation ranging from 5 (100% of information conveyed from the original) to 1 (0% of information conveyed from the original). Given that a sentence can be highly adequate but drastically change the connotation and intent of the sentence (eg, a sentence that contains 75% of the correct words but changes a sentence from take this medication twice a day to take this medication once every two days), we asked evaluators to assess meaning, a measure of connotation and intent maintenance, with scores ranging from 5 (Same meaning as original) to 1 (Totally different meaning from the original).19 Evaluators also assessed severity, a new measure of potential harm if a given sentence was assessed as having errors of any kind, ranging from 5 (Error, no effect on patient care) to 1 (Error, dangerous to patient) with an additional option of N/A (Sentence basically accurate). Finally, evaluators rated a blinded preference (also a new measure) for either of two translated sentences, ranging from Strongly prefer translation #1 to Strongly prefer translation #2. The order of the sentences was random (eg, sometimes the professional translation was first and sometimes the GT translation was). We subsequently converted this to preference for the professional translation, ranging from 5 (Strongly prefer the professional translation) to 1 (Strongly prefer the GT translation) in order to standardize the responses (Figures 1 and 2).
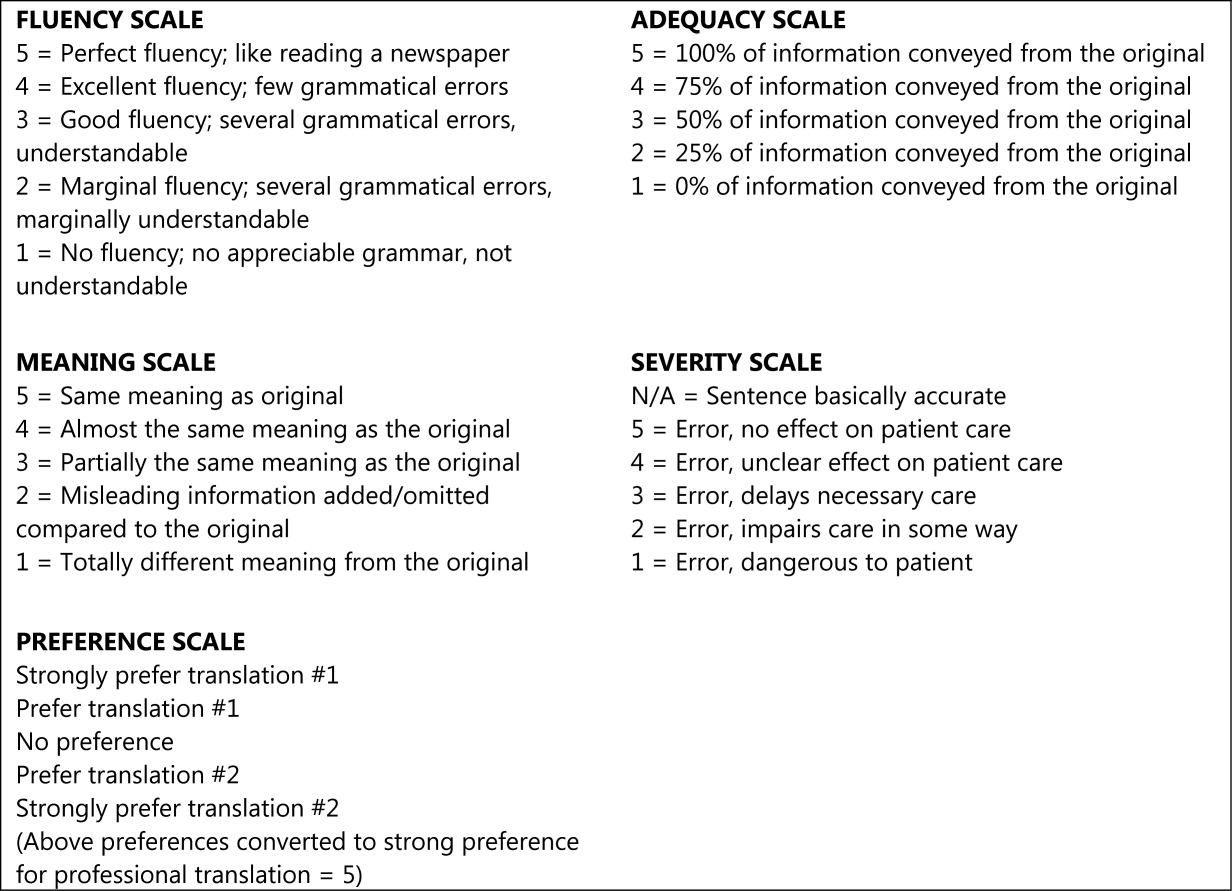
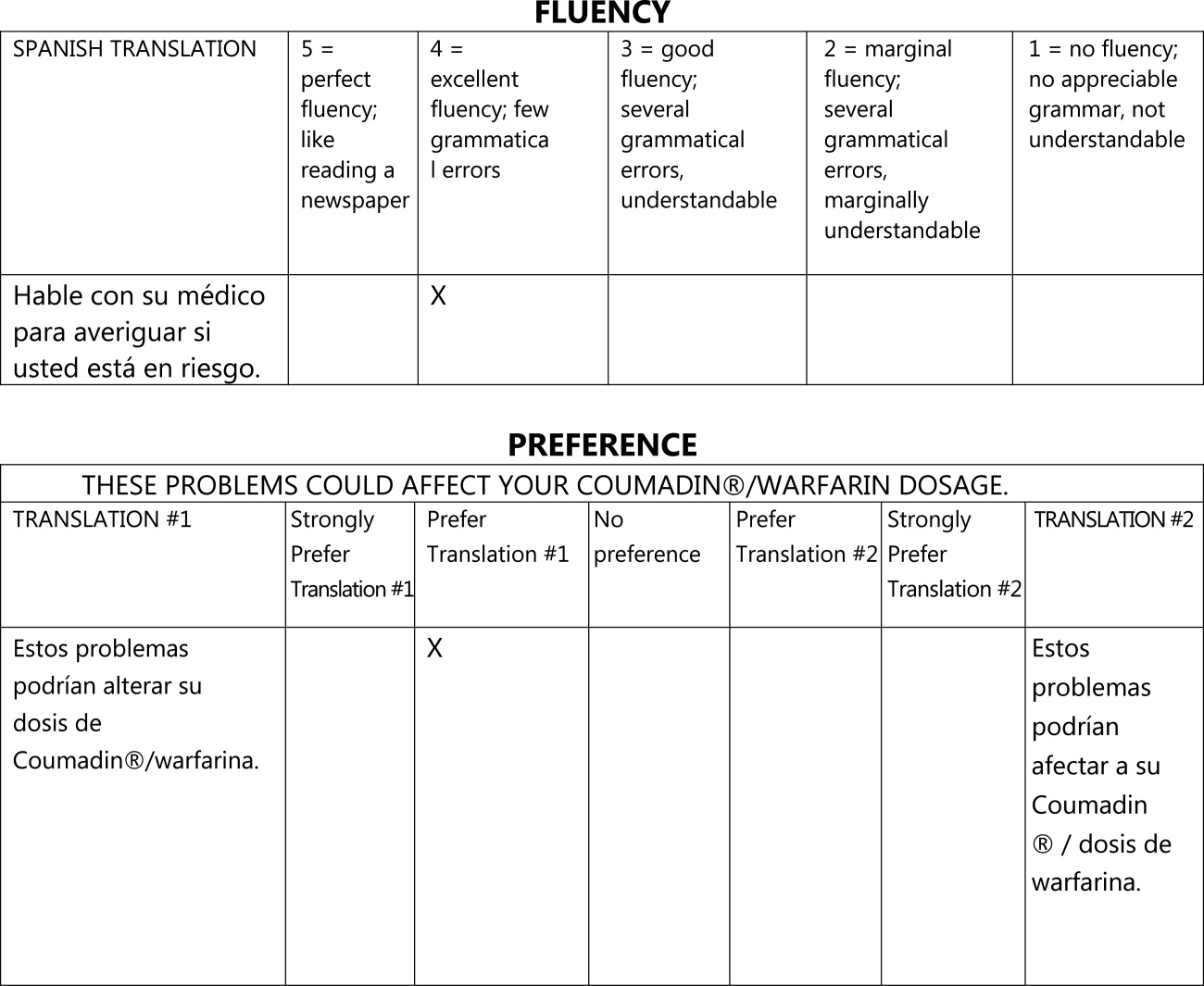
The overall flow of the study is given in Figure 3. Each evaluator initially scored 20 sentences translated by GT and 10 sentences translated professionally along the first four domains. All 30 of these sentences were randomly selected from the original, 263‐sentence pamphlet. For fluency, evaluators had access only to the translated sentence to be scored; for adequacy, meaning, and severity, they had access to both the translated sentence and the original English sentence. Ten of the 30 sentences were further selected randomly for scoring on the preference domain. For these 10 sentences, evaluators compared the GT and professional translations of the same sentence (with the original English sentence available as a reference) and indicated a preference, for any reason, for one translation or the other. Evaluators were blinded to the technique of translation (GT or professional) for all scored sentences and domains. We chose twice as many sentences from the GT preparations for the first four domains to maximize measurements for the translation technology we were evaluating, with the smaller number of professional translations serving as controls.
After scoring the first 30 sentences, evaluators met with one of the authors (R.R.K.) to discuss and consolidate their approach to scoring. They then scored an additional 10 GT‐translated sentences and 5 professionally translated sentences for the first four domains, and 9 of these 15 sentences for preference, to see if the meeting changed their scoring approach. These sentences were selected randomly from the original, 263‐sentence pamphlet, excluding the 30 evaluated in the previous step.
Automated Machine Translation Evaluation
Machine translation researchers have developed automated measures allowing the rapid and inexpensive scoring and rescoring of translations. These automated measures supplement more time‐ and resource‐intensive manual evaluations. The automated measures are based upon how well the translation compares to one or, ideally, multiple professionally prepared reference translations. They correlate well with human judgments on the domains above, especially when multiple reference translations are used (increasing the number of reference translations increases the variability allowed for words and phrases in the machine translation, improving the likelihood that differences in score are related to differences in quality rather than differences in translator preference).20 For this study, we used Metric for Evaluation of Translation with Explicit Ordering (METEOR), a machine translation evaluation system that allows additional flexibility for the machine translation in terms of grading individual sentences and being sensitive to synonyms, word stemming, and word order.21 We obtained a METEOR score for each of the GT‐translated sentences using the professional translation as our reference, and assessed correlation between this automated measure and the manual evaluations for the GT sentences, with the aim of assessing the feasibility of using METEOR in future work on patient educational material translation.
Outcomes and Statistical Analysis
We compared the scores assigned to GT‐translated sentences for each of the five manually scored domains as compared to the scores of the professionally translated sentences, as well as the impact of word count and sentence complexity on the scores achieved specifically by the GT‐translated sentences, using clustered linear regression to account for the fact that each of the 45 sentences were scored by each of the three evaluators. Sentences were classified as simple if they contained one or fewer clauses and complex if they contained more than one clause.22 We also assessed interrater reliability for the manual scoring system using intraclass correlation coefficients and repeatability. Repeatability is an estimate of the maximum difference, with 95% confidence, between scores assigned to the same sentence on the same domain by two different evaluators;23 lower scores indicate greater agreement between evaluators. Since we did not have clinical data or a gold standard, we used repeatability to estimate the value above which a difference between two scores might be clinically significant and not simply due to interrater variability.24 Finally, we assessed the correlation of the manual scores with those calculated by the METEOR automated evaluation tool using Pearson correlation coefficients. All analyses were conducted using Stata 11 (College Station, TX).
Results
Sentence Description
A total of 45 sentences were evaluated by the bilingual research assistants. The initial 30 sentences and the subsequent, post‐consolidation meeting 15 sentences were scored similarly in all outcomes, after adjustment for word length and complexity, so we pooled all 45 sentences (as well as the 19 total sentence pairs scored for preference) for the final analysis. Average sentence lengths were 14.2 words, 15.5 words, and 16.6 words for the English source text, professionally translated sentences, and GT‐translated sentences, respectively. Thirty‐three percent of the English source sentences were simple and 67% were complex.
Manual Evaluation Scores
Sentences translated by GT received worse scores on fluency as compared to the professional translations (3.4 vs 4.7, P < 0.0001). Comparisons for adequacy and meaning were not statistically significantly different. GT‐translated sentences contained more errors of any severity as compared to the professional translations (39% vs 22%, P = 0.05), but a similar number of serious, clinically impactful errors (severity scores of 3, 2, or 1; 4% vs 2%, P = 0.61). However, one GT‐translated sentence was considered erroneous with a severity level of 1 (Error, dangerous to patient). This particular sentence was 25 words long and complex in structure in the original English document; all three evaluators considered the GT translation nonsensical (La hemorragia mayor, llame a su mdico, o ir a la emergencia de un hospital habitacin si usted tiene cualquiera de los siguientes: Red N, oscuro, caf o cola de orina de color.) Evaluators had no overall preference for the professional translation (3.2, 95% confidence interval = 2.7 to 3.7, with 3 indicating no preference; P = 0.36) (Table 1).
| GoogleTranslate Translation | Professional Translation | P Value | |
|---|---|---|---|
| |||
| Fluency* | 3.4 | 4.7 | <0.0001 |
| Adequacy* | 4.5 | 4.8 | 0.19 |
| Meaning* | 4.2 | 4.5 | 0.29 |
| Severity | |||
| Any error | 39% | 22% | 0.05 |
| Serious error | 4% | 2% | 0.61 |
| Preference* | 3.2 | 0.36 | |
Mediation of Scores by Sentence Length or Complexity
We found that sentence length was not associated with scores for fluency, adequacy, meaning, severity, or preference (P > 0.30 in each case). Complexity, however, was significantly associated with preference: evaluators' preferred the professional translation for complex English sentences while being more ambivalent about simple English sentences (3.6 vs 2.6, P = 0.03).
Interrater Reliability and Repeatability
We assessed the interrater reliability for each domain using intraclass correlation coefficients and repeatability. For fluency, the intraclass correlation was best at 0.70; for adequacy, it was 0.58; for meaning, 0.42; for severity, 0.48; and for preference, 0.37. The repeatability scores were 1.4 for fluency, 0.6 for adequacy, 2.2 for meaning, 1.2 for severity, and 3.8 for preference, indicating that two evaluators might give a sentence almost the same score (at most, 1 point apart from one another) for adequacy, but might have opposite preferences regarding which translation of a sentence was superior.
Correlation with METEOR
Correlation between the first four domains and the METEOR scores were less than in prior studies.21 Fluency correlated best with METEOR at 0.53; adequacy correlated least with METEOR at 0.29. The remaining scores were in‐between. All correlations were statistically significant at P < 0.01 (Table 2).
| Correlation with METEOR | P value | |
|---|---|---|
| ||
| Fluency | 0.53 | <0.0001 |
| Adequacy | 0.29 | 0.006 |
| Meaning | 0.33 | 0.002 |
| Severity | 0.39 | 0.002 |
Discussion
In this preliminary study comparing the accuracy of GT to professional translation for patient educational material, we found that GT was inferior to the professional translation in grammatical fluency but generally preserved the content and sense of the original text. Out of 30 GT sentences assessed, there was one substantially erroneous translation that was considered potentially dangerous. Evaluators preferred the professionally translated sentences for complex sentences, but when the English source sentence was simplecontaining a single clausethis preference disappeared.
Like Sharif and Tse,12 we found that for information not arranged in sentences, automated translation sometimes produced nonsensical sentences. In our study, these resulted from an English sentence fragment followed by a bulleted list; in their study, the nonsensical translations resulted from pharmacy labels. The difference in frequency of these errors between our studies may have resulted partly from the translation tool evaluated (GT vs programs used by pharmacies in the Bronx), but may have also been due to our use of machine translation for complete sentencesthe purpose for which it is optimally designed. The hypothesis that machine translations of clinical information are most understandable when used for simple, complete sentences concurs with the methodology used by these tools and requires further study.
GT has the potential to be very useful to clinicians, particularly for those instances when the communication required is both spontaneous and routine or noncritical. For example, in the inpatient setting, patients could communicate diet and other nonclinical requests, as well as ask or answer simple, short questions when the interpreter is not available. In such situations, the low cost and ease of using online translations and machine translation more generally may help to circumvent the tendency of clinicians to get by with inadequate language skills or to avoid communication altogether.25 If used wisely, GT and other online tools could supplement the use of standardized translations and professional interpreters in helping clinicians to overcome language barriers and linguistic inertia, though this will require further assessment.
Ours is a pilot study, and while it suggests a more promising way to use online translation tools, significant further evaluation is required regarding accuracy and applicability prior to widespread use of any machine translation tools for patient care. The document we utilized for evaluation was a professionally translated patient educational brochure provided to individuals starting a complex medication. As online translation tools would most likely not be used in this setting, but rather for spontaneous and less critical patient‐specific instructions, further testing of GT as applied to such scenarios should be considered. Second, we only evaluated GT for English translated into Spanish; its usefulness in other languages will need to be evaluated. It also remains to be seen how easily GT translations will be understood by patients, who may have variable medical understanding and educational attainment as compared to our evaluators. Finally, in this evaluation, we only assessed automated written translation, not automated spoken translation services such as those now available on cellular phones and other mobile devices.11 The latter are based upon translation software with an additional speech recognition interface. These applications may prove to be even more useful than online translation, but the speech recognition component will add an additional layer of potential error and these applications will need to be evaluated on their own merits.
The domains chosen for this study had only moderate interrater reliability as assessed by intraclass correlation and repeatability, with meaning and preference scoring particularly poorly. The latter domains in particular will require more thorough assessment before routine use in online translation assessment. The variability in all domains may have resulted partly from the choice of nonclinicians of different ancestral backgrounds as evaluators. However, this variability is likely better representative of the wide range of patient backgrounds. Because our evaluators were not professional translators, we asked a professional interpreter to grade all sentences to assess the quality of their evaluation. While the interpreter noted slightly fewer errors among the professionally translated sentences (13% vs 22%) and slightly more errors among the GT‐translated sentences (50% vs 39%), and preferred the professional translation slightly more (3.8 vs 3.2), his scores for all of the other measures were almost identical, increasing our confidence in our primary findings (Appendix A). Additionally, since statistical translation is conducted sentence by sentence, in our study evaluators only scored translations at the sentence level. The accuracy of GT for whole paragraphs or entire documents will need to be assessed separately. The correlation between METEOR and the manual evaluation scores was less than in prior studies; while inexpensive to assess, METEOR will have to be recalibrated in optimal circumstanceswith several reference translations available rather than just onebefore it can be used to supplement the assessment of new languages, new materials, other translation technologies, and improvements in a given technology over time for patient educational material.
In summary, GT scored worse in grammar but similarly in content and sense to the professional translation, committing one critical error in translating a complex, fragmented sentence as nonsense. We believe that, with further study and judicious use, GT has the potential to substantially improve clinicians' communication with patients with limited English proficiency in the area of brief spontaneous patient‐specific information, supplementing well the role that professional spoken interpretation and standardized written translations already play.
- ,.Language use and English‐speaking ability: 2000. In:Census 2000 Brief.Washington, DC:US Census Bureau;2003. p. 2. http://www.census.gov/prod/2003pubs/c2kbr‐29.pdf.
- ,,,,.The need for more research on language barriers in health care: a proposed research agenda.Milbank Q.2006;84(1):111–133.
- ,,,.Language proficiency and adverse events in US hospitals: a pilot study.Int J Qual Health Care.2007;19(2):60–67.
- .The impact of medical interpreter services on the quality of health care: a systematic review.Med Care Res Rev.2005;62(3):255–299.
- ,,, et al.Errors in medical interpretation and their potential clinical consequences in pediatric encounters.Pediatrics.2003;111(1):6–14.
- ,,, et al.The effect of English language proficiency on length of stay and in‐hospital mortality.J Gen Intern Med.2004;19(3):221–228.
- ,,,.Influence of language barriers on outcomes of hospital care for general medicine inpatients.J Hosp Med.2010;5(5):276–282.
- ,.Hospitals, language, and culture: a snapshot of the nation. In:Los Angeles, CA:The California Endowment, the Joint Commission;2007. p.51–52. http://www.jointcommission.org/assets/1/6/hlc_paper.pdf.
- ,,,.Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature.Health Serv Res.2007;42(2):727–754.
- .Google's Computing Power Refines Translation Tool.New York Times; March 9,2010. Accessed March 24, 2010. http://www.nytimes.com/2010/03/09/technology/09translate.html?_r=1.
- , Translator. New York Times; March 20,2010. Accessed March 24, 2010. http://www.nytimes.com/2010/03/21/opinion/21bellos.html.
- ,.Accuracy of computer‐generated, Spanish‐language medicine labels.Pediatrics.2010;125(5):960–965.
- .Nielsen NetRatings Search Engine Ratings.SearchEngineWatch; August 22,2006. Accessed March 24, 2010. http://searchenginewatch.com/2156451.
- Google.Google Translate Help;2010. Accessed March 24, 2010. http://translate.google.com/support/?hl=en.
- ,.Chapter 4: Basic strategies. In:An Introduction to Machine Translation;1992. Accessed April 22, 2010. http://www.hutchinsweb.me.uk/IntroMT‐4.pdf
- .Your Guide to Coumadin®/Warfarin Therapy.Agency for Healthcare Research and Quality; August 21,2008. Accessed October 19, 2009. http://www.ahrq.gov/consumer/btpills.htm.
- ,,, et al.Patient reported receipt of medication instructions for warfarin is associated with reduced risk of serious bleeding events.J Gen Intern Med.2008;23(10):1589–1594.
- ,,.The ARPA MT evaluation methodologies: evolution, lessons, and future approaches. In: Proceedings of AMTA, 1994, Columbia, MD; October1994.
- ,.Overview of the IWSLT 2005 evaluation campaign. In: Proceedings of IWSLT 2005, Pittsburgh, PA; October2005.
- ,,,.BLEU: a method for automatic evaluation of machine translation. In: ACL‐2002: 40th Annual Meeting of the Association for Computational Linguistics.2002:311–318.
- ,.METEOR: an automatic metric for MT evaluation with high levels of correlation with human judgments. In: Proceedings of the Second Workshop on Statistical Machine Translation at ACL, Prague, Czech Republic; June2007.
- .The Structure of a Sentence.Ottawa:The Writing Centre, University of Ottawa;2007.
- ,.Statistical methods for assessing agreement between two methods of clinical measurement.Lancet.1986;1(8476):307–310.
- .Measurement, reproducibility, and validity. In:Epidemiologic Methods 203.San Francisco:Department of Biostatistics and Epidemiology, University of California;2009.
- ,,,,.Getting by: underuse of interpreters by resident physicians.J Gen Intern Med.2009;24(2):256–262.
The population of patients in the US with limited English proficiency (LEP)those who speak English less than very well1is substantial and continues to grow.1, 2 Patients with LEP are at risk for lower quality health care overall than their English‐speaking counterparts.38 Professional in‐person interpreters greatly improve spoken communication and quality of care for these patients,4, 9 but their assistance is typically based on the clinical encounter. Particularly if interpreting by phone, interpreters are unlikely to be able to help with materials such as discharge instructions or information sheets meant for family members. Professional written translations of patient educational material help to bridge this gap, allowing clinicians to convey detailed written instructions to patients. However, professional translations must be prepared well in advance of any encounter and can only be used for easily anticipated problems.
The need to translate less common, patient‐specific instructions arises spontaneously in clinical practice, and formally prepared written translations are not useful in these situations. Online translation tools such as GoogleTranslate (available at
We conducted a pilot evaluation of an online translation tool as it relates to detailed, complex patient educational material. Our primary goal was to compare the accuracy of a Spanish translation generated by the online tool to that done by a professional agency. Our secondary goals were: 1) to assess whether sentence word length or complexity mediated the accuracy of GT; and 2) to lay the foundation for a more comprehensive study of the accuracy of online translation tools, with respect to patient educational material.
Methods
Translation Tool and Language Choice
We selected Google Translate (GT) since it is one of the more commonly used online translation tools and because Google is the most widely used search engine in the United States.13 GT uses statistical translation methodology to convert text, documents, and websites between languages; statistical translation involves the following three steps. First, the translation program recognizes a sentence to translate. Second, it compares the words and phrases within that sentence to the billions of words in its library (drawn from bilingual professionally translated documents, such as United Nations proceedings). Third, it uses this comparison to generate a translation combining the words and phrases deemed most equivalent between the source sentence and the target language. If there are multiple sentences, the program recognizes and translates each independently. As the body of bilingual work grows, the program learns and refines its rules automatically.14 In contrast, in rule‐based translation, a program would use manually prespecified rules regarding word choice and grammar to generate a translation.15 We assessed GT's accuracy translating from English to Spanish because Spanish is the predominant non‐English language spoken in the US.1
Document Selection and Preparation
We selected the instruction manual regarding warfarin use prepared by the Agency for Healthcare Research and Quality (AHRQ) for this accuracy evaluation. We selected this manual,16 written at a 6th grade reading level, because a professional Spanish translation was available (completed by ASET International Service, LLC, before and independently of this study), and because patient educational material regarding warfarin has been associated with fewer bleeding events.17 We downloaded the English document on October 19, 2009 and used the GT website to translate it en bloc. We then copied the resulting Spanish output into a text file. The English document and the professional Spanish translation (downloaded the same day) were both converted into text files in the same manner.
Grading Methodology
We scored the translation chosen using both manual and automated evaluation techniques. These techniques are widely used in the machine translation literature and are explained below.
Manual Evaluation: Evaluators, Domains, Scoring
We recruited three nonclinician, bilingual, nativeSpanish‐speaking research assistants as evaluators. The evaluators were all college educated with a Bachelor's degree or higher and were of Mexican, Nicaraguan, and Guatemalan ancestry. Each evaluator received a brief orientation regarding the project, as well as an explanation of the scores, and then proceeded to the blinded evaluation independently.
We asked evaluators to score sentences on Likert scales along five primary domains: fluency, adequacy, meaning, severity, and preference. Fluency and adequacy are well accepted components of machine translation evaluation,18 with fluency being an assessment of grammar and readability ranging from 5 (Perfect fluency; like reading a newspaper) to 1 (No fluency; no appreciable grammar, not understandable) and adequacy being an assessment of information preservation ranging from 5 (100% of information conveyed from the original) to 1 (0% of information conveyed from the original). Given that a sentence can be highly adequate but drastically change the connotation and intent of the sentence (eg, a sentence that contains 75% of the correct words but changes a sentence from take this medication twice a day to take this medication once every two days), we asked evaluators to assess meaning, a measure of connotation and intent maintenance, with scores ranging from 5 (Same meaning as original) to 1 (Totally different meaning from the original).19 Evaluators also assessed severity, a new measure of potential harm if a given sentence was assessed as having errors of any kind, ranging from 5 (Error, no effect on patient care) to 1 (Error, dangerous to patient) with an additional option of N/A (Sentence basically accurate). Finally, evaluators rated a blinded preference (also a new measure) for either of two translated sentences, ranging from Strongly prefer translation #1 to Strongly prefer translation #2. The order of the sentences was random (eg, sometimes the professional translation was first and sometimes the GT translation was). We subsequently converted this to preference for the professional translation, ranging from 5 (Strongly prefer the professional translation) to 1 (Strongly prefer the GT translation) in order to standardize the responses (Figures 1 and 2).


The overall flow of the study is given in Figure 3. Each evaluator initially scored 20 sentences translated by GT and 10 sentences translated professionally along the first four domains. All 30 of these sentences were randomly selected from the original, 263‐sentence pamphlet. For fluency, evaluators had access only to the translated sentence to be scored; for adequacy, meaning, and severity, they had access to both the translated sentence and the original English sentence. Ten of the 30 sentences were further selected randomly for scoring on the preference domain. For these 10 sentences, evaluators compared the GT and professional translations of the same sentence (with the original English sentence available as a reference) and indicated a preference, for any reason, for one translation or the other. Evaluators were blinded to the technique of translation (GT or professional) for all scored sentences and domains. We chose twice as many sentences from the GT preparations for the first four domains to maximize measurements for the translation technology we were evaluating, with the smaller number of professional translations serving as controls.

After scoring the first 30 sentences, evaluators met with one of the authors (R.R.K.) to discuss and consolidate their approach to scoring. They then scored an additional 10 GT‐translated sentences and 5 professionally translated sentences for the first four domains, and 9 of these 15 sentences for preference, to see if the meeting changed their scoring approach. These sentences were selected randomly from the original, 263‐sentence pamphlet, excluding the 30 evaluated in the previous step.
Automated Machine Translation Evaluation
Machine translation researchers have developed automated measures allowing the rapid and inexpensive scoring and rescoring of translations. These automated measures supplement more time‐ and resource‐intensive manual evaluations. The automated measures are based upon how well the translation compares to one or, ideally, multiple professionally prepared reference translations. They correlate well with human judgments on the domains above, especially when multiple reference translations are used (increasing the number of reference translations increases the variability allowed for words and phrases in the machine translation, improving the likelihood that differences in score are related to differences in quality rather than differences in translator preference).20 For this study, we used Metric for Evaluation of Translation with Explicit Ordering (METEOR), a machine translation evaluation system that allows additional flexibility for the machine translation in terms of grading individual sentences and being sensitive to synonyms, word stemming, and word order.21 We obtained a METEOR score for each of the GT‐translated sentences using the professional translation as our reference, and assessed correlation between this automated measure and the manual evaluations for the GT sentences, with the aim of assessing the feasibility of using METEOR in future work on patient educational material translation.
Outcomes and Statistical Analysis
We compared the scores assigned to GT‐translated sentences for each of the five manually scored domains as compared to the scores of the professionally translated sentences, as well as the impact of word count and sentence complexity on the scores achieved specifically by the GT‐translated sentences, using clustered linear regression to account for the fact that each of the 45 sentences were scored by each of the three evaluators. Sentences were classified as simple if they contained one or fewer clauses and complex if they contained more than one clause.22 We also assessed interrater reliability for the manual scoring system using intraclass correlation coefficients and repeatability. Repeatability is an estimate of the maximum difference, with 95% confidence, between scores assigned to the same sentence on the same domain by two different evaluators;23 lower scores indicate greater agreement between evaluators. Since we did not have clinical data or a gold standard, we used repeatability to estimate the value above which a difference between two scores might be clinically significant and not simply due to interrater variability.24 Finally, we assessed the correlation of the manual scores with those calculated by the METEOR automated evaluation tool using Pearson correlation coefficients. All analyses were conducted using Stata 11 (College Station, TX).
Results
Sentence Description
A total of 45 sentences were evaluated by the bilingual research assistants. The initial 30 sentences and the subsequent, post‐consolidation meeting 15 sentences were scored similarly in all outcomes, after adjustment for word length and complexity, so we pooled all 45 sentences (as well as the 19 total sentence pairs scored for preference) for the final analysis. Average sentence lengths were 14.2 words, 15.5 words, and 16.6 words for the English source text, professionally translated sentences, and GT‐translated sentences, respectively. Thirty‐three percent of the English source sentences were simple and 67% were complex.
Manual Evaluation Scores
Sentences translated by GT received worse scores on fluency as compared to the professional translations (3.4 vs 4.7, P < 0.0001). Comparisons for adequacy and meaning were not statistically significantly different. GT‐translated sentences contained more errors of any severity as compared to the professional translations (39% vs 22%, P = 0.05), but a similar number of serious, clinically impactful errors (severity scores of 3, 2, or 1; 4% vs 2%, P = 0.61). However, one GT‐translated sentence was considered erroneous with a severity level of 1 (Error, dangerous to patient). This particular sentence was 25 words long and complex in structure in the original English document; all three evaluators considered the GT translation nonsensical (La hemorragia mayor, llame a su mdico, o ir a la emergencia de un hospital habitacin si usted tiene cualquiera de los siguientes: Red N, oscuro, caf o cola de orina de color.) Evaluators had no overall preference for the professional translation (3.2, 95% confidence interval = 2.7 to 3.7, with 3 indicating no preference; P = 0.36) (Table 1).
| GoogleTranslate Translation | Professional Translation | P Value | |
|---|---|---|---|
| |||
| Fluency* | 3.4 | 4.7 | <0.0001 |
| Adequacy* | 4.5 | 4.8 | 0.19 |
| Meaning* | 4.2 | 4.5 | 0.29 |
| Severity | |||
| Any error | 39% | 22% | 0.05 |
| Serious error | 4% | 2% | 0.61 |
| Preference* | 3.2 | 0.36 | |
Mediation of Scores by Sentence Length or Complexity
We found that sentence length was not associated with scores for fluency, adequacy, meaning, severity, or preference (P > 0.30 in each case). Complexity, however, was significantly associated with preference: evaluators' preferred the professional translation for complex English sentences while being more ambivalent about simple English sentences (3.6 vs 2.6, P = 0.03).
Interrater Reliability and Repeatability
We assessed the interrater reliability for each domain using intraclass correlation coefficients and repeatability. For fluency, the intraclass correlation was best at 0.70; for adequacy, it was 0.58; for meaning, 0.42; for severity, 0.48; and for preference, 0.37. The repeatability scores were 1.4 for fluency, 0.6 for adequacy, 2.2 for meaning, 1.2 for severity, and 3.8 for preference, indicating that two evaluators might give a sentence almost the same score (at most, 1 point apart from one another) for adequacy, but might have opposite preferences regarding which translation of a sentence was superior.
Correlation with METEOR
Correlation between the first four domains and the METEOR scores were less than in prior studies.21 Fluency correlated best with METEOR at 0.53; adequacy correlated least with METEOR at 0.29. The remaining scores were in‐between. All correlations were statistically significant at P < 0.01 (Table 2).
| Correlation with METEOR | P value | |
|---|---|---|
| ||
| Fluency | 0.53 | <0.0001 |
| Adequacy | 0.29 | 0.006 |
| Meaning | 0.33 | 0.002 |
| Severity | 0.39 | 0.002 |
Discussion
In this preliminary study comparing the accuracy of GT to professional translation for patient educational material, we found that GT was inferior to the professional translation in grammatical fluency but generally preserved the content and sense of the original text. Out of 30 GT sentences assessed, there was one substantially erroneous translation that was considered potentially dangerous. Evaluators preferred the professionally translated sentences for complex sentences, but when the English source sentence was simplecontaining a single clausethis preference disappeared.
Like Sharif and Tse,12 we found that for information not arranged in sentences, automated translation sometimes produced nonsensical sentences. In our study, these resulted from an English sentence fragment followed by a bulleted list; in their study, the nonsensical translations resulted from pharmacy labels. The difference in frequency of these errors between our studies may have resulted partly from the translation tool evaluated (GT vs programs used by pharmacies in the Bronx), but may have also been due to our use of machine translation for complete sentencesthe purpose for which it is optimally designed. The hypothesis that machine translations of clinical information are most understandable when used for simple, complete sentences concurs with the methodology used by these tools and requires further study.
GT has the potential to be very useful to clinicians, particularly for those instances when the communication required is both spontaneous and routine or noncritical. For example, in the inpatient setting, patients could communicate diet and other nonclinical requests, as well as ask or answer simple, short questions when the interpreter is not available. In such situations, the low cost and ease of using online translations and machine translation more generally may help to circumvent the tendency of clinicians to get by with inadequate language skills or to avoid communication altogether.25 If used wisely, GT and other online tools could supplement the use of standardized translations and professional interpreters in helping clinicians to overcome language barriers and linguistic inertia, though this will require further assessment.
Ours is a pilot study, and while it suggests a more promising way to use online translation tools, significant further evaluation is required regarding accuracy and applicability prior to widespread use of any machine translation tools for patient care. The document we utilized for evaluation was a professionally translated patient educational brochure provided to individuals starting a complex medication. As online translation tools would most likely not be used in this setting, but rather for spontaneous and less critical patient‐specific instructions, further testing of GT as applied to such scenarios should be considered. Second, we only evaluated GT for English translated into Spanish; its usefulness in other languages will need to be evaluated. It also remains to be seen how easily GT translations will be understood by patients, who may have variable medical understanding and educational attainment as compared to our evaluators. Finally, in this evaluation, we only assessed automated written translation, not automated spoken translation services such as those now available on cellular phones and other mobile devices.11 The latter are based upon translation software with an additional speech recognition interface. These applications may prove to be even more useful than online translation, but the speech recognition component will add an additional layer of potential error and these applications will need to be evaluated on their own merits.
The domains chosen for this study had only moderate interrater reliability as assessed by intraclass correlation and repeatability, with meaning and preference scoring particularly poorly. The latter domains in particular will require more thorough assessment before routine use in online translation assessment. The variability in all domains may have resulted partly from the choice of nonclinicians of different ancestral backgrounds as evaluators. However, this variability is likely better representative of the wide range of patient backgrounds. Because our evaluators were not professional translators, we asked a professional interpreter to grade all sentences to assess the quality of their evaluation. While the interpreter noted slightly fewer errors among the professionally translated sentences (13% vs 22%) and slightly more errors among the GT‐translated sentences (50% vs 39%), and preferred the professional translation slightly more (3.8 vs 3.2), his scores for all of the other measures were almost identical, increasing our confidence in our primary findings (Appendix A). Additionally, since statistical translation is conducted sentence by sentence, in our study evaluators only scored translations at the sentence level. The accuracy of GT for whole paragraphs or entire documents will need to be assessed separately. The correlation between METEOR and the manual evaluation scores was less than in prior studies; while inexpensive to assess, METEOR will have to be recalibrated in optimal circumstanceswith several reference translations available rather than just onebefore it can be used to supplement the assessment of new languages, new materials, other translation technologies, and improvements in a given technology over time for patient educational material.
In summary, GT scored worse in grammar but similarly in content and sense to the professional translation, committing one critical error in translating a complex, fragmented sentence as nonsense. We believe that, with further study and judicious use, GT has the potential to substantially improve clinicians' communication with patients with limited English proficiency in the area of brief spontaneous patient‐specific information, supplementing well the role that professional spoken interpretation and standardized written translations already play.
The population of patients in the US with limited English proficiency (LEP)those who speak English less than very well1is substantial and continues to grow.1, 2 Patients with LEP are at risk for lower quality health care overall than their English‐speaking counterparts.38 Professional in‐person interpreters greatly improve spoken communication and quality of care for these patients,4, 9 but their assistance is typically based on the clinical encounter. Particularly if interpreting by phone, interpreters are unlikely to be able to help with materials such as discharge instructions or information sheets meant for family members. Professional written translations of patient educational material help to bridge this gap, allowing clinicians to convey detailed written instructions to patients. However, professional translations must be prepared well in advance of any encounter and can only be used for easily anticipated problems.
The need to translate less common, patient‐specific instructions arises spontaneously in clinical practice, and formally prepared written translations are not useful in these situations. Online translation tools such as GoogleTranslate (available at
We conducted a pilot evaluation of an online translation tool as it relates to detailed, complex patient educational material. Our primary goal was to compare the accuracy of a Spanish translation generated by the online tool to that done by a professional agency. Our secondary goals were: 1) to assess whether sentence word length or complexity mediated the accuracy of GT; and 2) to lay the foundation for a more comprehensive study of the accuracy of online translation tools, with respect to patient educational material.
Methods
Translation Tool and Language Choice
We selected Google Translate (GT) since it is one of the more commonly used online translation tools and because Google is the most widely used search engine in the United States.13 GT uses statistical translation methodology to convert text, documents, and websites between languages; statistical translation involves the following three steps. First, the translation program recognizes a sentence to translate. Second, it compares the words and phrases within that sentence to the billions of words in its library (drawn from bilingual professionally translated documents, such as United Nations proceedings). Third, it uses this comparison to generate a translation combining the words and phrases deemed most equivalent between the source sentence and the target language. If there are multiple sentences, the program recognizes and translates each independently. As the body of bilingual work grows, the program learns and refines its rules automatically.14 In contrast, in rule‐based translation, a program would use manually prespecified rules regarding word choice and grammar to generate a translation.15 We assessed GT's accuracy translating from English to Spanish because Spanish is the predominant non‐English language spoken in the US.1
Document Selection and Preparation
We selected the instruction manual regarding warfarin use prepared by the Agency for Healthcare Research and Quality (AHRQ) for this accuracy evaluation. We selected this manual,16 written at a 6th grade reading level, because a professional Spanish translation was available (completed by ASET International Service, LLC, before and independently of this study), and because patient educational material regarding warfarin has been associated with fewer bleeding events.17 We downloaded the English document on October 19, 2009 and used the GT website to translate it en bloc. We then copied the resulting Spanish output into a text file. The English document and the professional Spanish translation (downloaded the same day) were both converted into text files in the same manner.
Grading Methodology
We scored the translation chosen using both manual and automated evaluation techniques. These techniques are widely used in the machine translation literature and are explained below.
Manual Evaluation: Evaluators, Domains, Scoring
We recruited three nonclinician, bilingual, nativeSpanish‐speaking research assistants as evaluators. The evaluators were all college educated with a Bachelor's degree or higher and were of Mexican, Nicaraguan, and Guatemalan ancestry. Each evaluator received a brief orientation regarding the project, as well as an explanation of the scores, and then proceeded to the blinded evaluation independently.
We asked evaluators to score sentences on Likert scales along five primary domains: fluency, adequacy, meaning, severity, and preference. Fluency and adequacy are well accepted components of machine translation evaluation,18 with fluency being an assessment of grammar and readability ranging from 5 (Perfect fluency; like reading a newspaper) to 1 (No fluency; no appreciable grammar, not understandable) and adequacy being an assessment of information preservation ranging from 5 (100% of information conveyed from the original) to 1 (0% of information conveyed from the original). Given that a sentence can be highly adequate but drastically change the connotation and intent of the sentence (eg, a sentence that contains 75% of the correct words but changes a sentence from take this medication twice a day to take this medication once every two days), we asked evaluators to assess meaning, a measure of connotation and intent maintenance, with scores ranging from 5 (Same meaning as original) to 1 (Totally different meaning from the original).19 Evaluators also assessed severity, a new measure of potential harm if a given sentence was assessed as having errors of any kind, ranging from 5 (Error, no effect on patient care) to 1 (Error, dangerous to patient) with an additional option of N/A (Sentence basically accurate). Finally, evaluators rated a blinded preference (also a new measure) for either of two translated sentences, ranging from Strongly prefer translation #1 to Strongly prefer translation #2. The order of the sentences was random (eg, sometimes the professional translation was first and sometimes the GT translation was). We subsequently converted this to preference for the professional translation, ranging from 5 (Strongly prefer the professional translation) to 1 (Strongly prefer the GT translation) in order to standardize the responses (Figures 1 and 2).


The overall flow of the study is given in Figure 3. Each evaluator initially scored 20 sentences translated by GT and 10 sentences translated professionally along the first four domains. All 30 of these sentences were randomly selected from the original, 263‐sentence pamphlet. For fluency, evaluators had access only to the translated sentence to be scored; for adequacy, meaning, and severity, they had access to both the translated sentence and the original English sentence. Ten of the 30 sentences were further selected randomly for scoring on the preference domain. For these 10 sentences, evaluators compared the GT and professional translations of the same sentence (with the original English sentence available as a reference) and indicated a preference, for any reason, for one translation or the other. Evaluators were blinded to the technique of translation (GT or professional) for all scored sentences and domains. We chose twice as many sentences from the GT preparations for the first four domains to maximize measurements for the translation technology we were evaluating, with the smaller number of professional translations serving as controls.

After scoring the first 30 sentences, evaluators met with one of the authors (R.R.K.) to discuss and consolidate their approach to scoring. They then scored an additional 10 GT‐translated sentences and 5 professionally translated sentences for the first four domains, and 9 of these 15 sentences for preference, to see if the meeting changed their scoring approach. These sentences were selected randomly from the original, 263‐sentence pamphlet, excluding the 30 evaluated in the previous step.
Automated Machine Translation Evaluation
Machine translation researchers have developed automated measures allowing the rapid and inexpensive scoring and rescoring of translations. These automated measures supplement more time‐ and resource‐intensive manual evaluations. The automated measures are based upon how well the translation compares to one or, ideally, multiple professionally prepared reference translations. They correlate well with human judgments on the domains above, especially when multiple reference translations are used (increasing the number of reference translations increases the variability allowed for words and phrases in the machine translation, improving the likelihood that differences in score are related to differences in quality rather than differences in translator preference).20 For this study, we used Metric for Evaluation of Translation with Explicit Ordering (METEOR), a machine translation evaluation system that allows additional flexibility for the machine translation in terms of grading individual sentences and being sensitive to synonyms, word stemming, and word order.21 We obtained a METEOR score for each of the GT‐translated sentences using the professional translation as our reference, and assessed correlation between this automated measure and the manual evaluations for the GT sentences, with the aim of assessing the feasibility of using METEOR in future work on patient educational material translation.
Outcomes and Statistical Analysis
We compared the scores assigned to GT‐translated sentences for each of the five manually scored domains as compared to the scores of the professionally translated sentences, as well as the impact of word count and sentence complexity on the scores achieved specifically by the GT‐translated sentences, using clustered linear regression to account for the fact that each of the 45 sentences were scored by each of the three evaluators. Sentences were classified as simple if they contained one or fewer clauses and complex if they contained more than one clause.22 We also assessed interrater reliability for the manual scoring system using intraclass correlation coefficients and repeatability. Repeatability is an estimate of the maximum difference, with 95% confidence, between scores assigned to the same sentence on the same domain by two different evaluators;23 lower scores indicate greater agreement between evaluators. Since we did not have clinical data or a gold standard, we used repeatability to estimate the value above which a difference between two scores might be clinically significant and not simply due to interrater variability.24 Finally, we assessed the correlation of the manual scores with those calculated by the METEOR automated evaluation tool using Pearson correlation coefficients. All analyses were conducted using Stata 11 (College Station, TX).
Results
Sentence Description
A total of 45 sentences were evaluated by the bilingual research assistants. The initial 30 sentences and the subsequent, post‐consolidation meeting 15 sentences were scored similarly in all outcomes, after adjustment for word length and complexity, so we pooled all 45 sentences (as well as the 19 total sentence pairs scored for preference) for the final analysis. Average sentence lengths were 14.2 words, 15.5 words, and 16.6 words for the English source text, professionally translated sentences, and GT‐translated sentences, respectively. Thirty‐three percent of the English source sentences were simple and 67% were complex.
Manual Evaluation Scores
Sentences translated by GT received worse scores on fluency as compared to the professional translations (3.4 vs 4.7, P < 0.0001). Comparisons for adequacy and meaning were not statistically significantly different. GT‐translated sentences contained more errors of any severity as compared to the professional translations (39% vs 22%, P = 0.05), but a similar number of serious, clinically impactful errors (severity scores of 3, 2, or 1; 4% vs 2%, P = 0.61). However, one GT‐translated sentence was considered erroneous with a severity level of 1 (Error, dangerous to patient). This particular sentence was 25 words long and complex in structure in the original English document; all three evaluators considered the GT translation nonsensical (La hemorragia mayor, llame a su mdico, o ir a la emergencia de un hospital habitacin si usted tiene cualquiera de los siguientes: Red N, oscuro, caf o cola de orina de color.) Evaluators had no overall preference for the professional translation (3.2, 95% confidence interval = 2.7 to 3.7, with 3 indicating no preference; P = 0.36) (Table 1).
| GoogleTranslate Translation | Professional Translation | P Value | |
|---|---|---|---|
| |||
| Fluency* | 3.4 | 4.7 | <0.0001 |
| Adequacy* | 4.5 | 4.8 | 0.19 |
| Meaning* | 4.2 | 4.5 | 0.29 |
| Severity | |||
| Any error | 39% | 22% | 0.05 |
| Serious error | 4% | 2% | 0.61 |
| Preference* | 3.2 | 0.36 | |
Mediation of Scores by Sentence Length or Complexity
We found that sentence length was not associated with scores for fluency, adequacy, meaning, severity, or preference (P > 0.30 in each case). Complexity, however, was significantly associated with preference: evaluators' preferred the professional translation for complex English sentences while being more ambivalent about simple English sentences (3.6 vs 2.6, P = 0.03).
Interrater Reliability and Repeatability
We assessed the interrater reliability for each domain using intraclass correlation coefficients and repeatability. For fluency, the intraclass correlation was best at 0.70; for adequacy, it was 0.58; for meaning, 0.42; for severity, 0.48; and for preference, 0.37. The repeatability scores were 1.4 for fluency, 0.6 for adequacy, 2.2 for meaning, 1.2 for severity, and 3.8 for preference, indicating that two evaluators might give a sentence almost the same score (at most, 1 point apart from one another) for adequacy, but might have opposite preferences regarding which translation of a sentence was superior.
Correlation with METEOR
Correlation between the first four domains and the METEOR scores were less than in prior studies.21 Fluency correlated best with METEOR at 0.53; adequacy correlated least with METEOR at 0.29. The remaining scores were in‐between. All correlations were statistically significant at P < 0.01 (Table 2).
| Correlation with METEOR | P value | |
|---|---|---|
| ||
| Fluency | 0.53 | <0.0001 |
| Adequacy | 0.29 | 0.006 |
| Meaning | 0.33 | 0.002 |
| Severity | 0.39 | 0.002 |
Discussion
In this preliminary study comparing the accuracy of GT to professional translation for patient educational material, we found that GT was inferior to the professional translation in grammatical fluency but generally preserved the content and sense of the original text. Out of 30 GT sentences assessed, there was one substantially erroneous translation that was considered potentially dangerous. Evaluators preferred the professionally translated sentences for complex sentences, but when the English source sentence was simplecontaining a single clausethis preference disappeared.
Like Sharif and Tse,12 we found that for information not arranged in sentences, automated translation sometimes produced nonsensical sentences. In our study, these resulted from an English sentence fragment followed by a bulleted list; in their study, the nonsensical translations resulted from pharmacy labels. The difference in frequency of these errors between our studies may have resulted partly from the translation tool evaluated (GT vs programs used by pharmacies in the Bronx), but may have also been due to our use of machine translation for complete sentencesthe purpose for which it is optimally designed. The hypothesis that machine translations of clinical information are most understandable when used for simple, complete sentences concurs with the methodology used by these tools and requires further study.
GT has the potential to be very useful to clinicians, particularly for those instances when the communication required is both spontaneous and routine or noncritical. For example, in the inpatient setting, patients could communicate diet and other nonclinical requests, as well as ask or answer simple, short questions when the interpreter is not available. In such situations, the low cost and ease of using online translations and machine translation more generally may help to circumvent the tendency of clinicians to get by with inadequate language skills or to avoid communication altogether.25 If used wisely, GT and other online tools could supplement the use of standardized translations and professional interpreters in helping clinicians to overcome language barriers and linguistic inertia, though this will require further assessment.
Ours is a pilot study, and while it suggests a more promising way to use online translation tools, significant further evaluation is required regarding accuracy and applicability prior to widespread use of any machine translation tools for patient care. The document we utilized for evaluation was a professionally translated patient educational brochure provided to individuals starting a complex medication. As online translation tools would most likely not be used in this setting, but rather for spontaneous and less critical patient‐specific instructions, further testing of GT as applied to such scenarios should be considered. Second, we only evaluated GT for English translated into Spanish; its usefulness in other languages will need to be evaluated. It also remains to be seen how easily GT translations will be understood by patients, who may have variable medical understanding and educational attainment as compared to our evaluators. Finally, in this evaluation, we only assessed automated written translation, not automated spoken translation services such as those now available on cellular phones and other mobile devices.11 The latter are based upon translation software with an additional speech recognition interface. These applications may prove to be even more useful than online translation, but the speech recognition component will add an additional layer of potential error and these applications will need to be evaluated on their own merits.
The domains chosen for this study had only moderate interrater reliability as assessed by intraclass correlation and repeatability, with meaning and preference scoring particularly poorly. The latter domains in particular will require more thorough assessment before routine use in online translation assessment. The variability in all domains may have resulted partly from the choice of nonclinicians of different ancestral backgrounds as evaluators. However, this variability is likely better representative of the wide range of patient backgrounds. Because our evaluators were not professional translators, we asked a professional interpreter to grade all sentences to assess the quality of their evaluation. While the interpreter noted slightly fewer errors among the professionally translated sentences (13% vs 22%) and slightly more errors among the GT‐translated sentences (50% vs 39%), and preferred the professional translation slightly more (3.8 vs 3.2), his scores for all of the other measures were almost identical, increasing our confidence in our primary findings (Appendix A). Additionally, since statistical translation is conducted sentence by sentence, in our study evaluators only scored translations at the sentence level. The accuracy of GT for whole paragraphs or entire documents will need to be assessed separately. The correlation between METEOR and the manual evaluation scores was less than in prior studies; while inexpensive to assess, METEOR will have to be recalibrated in optimal circumstanceswith several reference translations available rather than just onebefore it can be used to supplement the assessment of new languages, new materials, other translation technologies, and improvements in a given technology over time for patient educational material.
In summary, GT scored worse in grammar but similarly in content and sense to the professional translation, committing one critical error in translating a complex, fragmented sentence as nonsense. We believe that, with further study and judicious use, GT has the potential to substantially improve clinicians' communication with patients with limited English proficiency in the area of brief spontaneous patient‐specific information, supplementing well the role that professional spoken interpretation and standardized written translations already play.
- ,.Language use and English‐speaking ability: 2000. In:Census 2000 Brief.Washington, DC:US Census Bureau;2003. p. 2. http://www.census.gov/prod/2003pubs/c2kbr‐29.pdf.
- ,,,,.The need for more research on language barriers in health care: a proposed research agenda.Milbank Q.2006;84(1):111–133.
- ,,,.Language proficiency and adverse events in US hospitals: a pilot study.Int J Qual Health Care.2007;19(2):60–67.
- .The impact of medical interpreter services on the quality of health care: a systematic review.Med Care Res Rev.2005;62(3):255–299.
- ,,, et al.Errors in medical interpretation and their potential clinical consequences in pediatric encounters.Pediatrics.2003;111(1):6–14.
- ,,, et al.The effect of English language proficiency on length of stay and in‐hospital mortality.J Gen Intern Med.2004;19(3):221–228.
- ,,,.Influence of language barriers on outcomes of hospital care for general medicine inpatients.J Hosp Med.2010;5(5):276–282.
- ,.Hospitals, language, and culture: a snapshot of the nation. In:Los Angeles, CA:The California Endowment, the Joint Commission;2007. p.51–52. http://www.jointcommission.org/assets/1/6/hlc_paper.pdf.
- ,,,.Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature.Health Serv Res.2007;42(2):727–754.
- .Google's Computing Power Refines Translation Tool.New York Times; March 9,2010. Accessed March 24, 2010. http://www.nytimes.com/2010/03/09/technology/09translate.html?_r=1.
- , Translator. New York Times; March 20,2010. Accessed March 24, 2010. http://www.nytimes.com/2010/03/21/opinion/21bellos.html.
- ,.Accuracy of computer‐generated, Spanish‐language medicine labels.Pediatrics.2010;125(5):960–965.
- .Nielsen NetRatings Search Engine Ratings.SearchEngineWatch; August 22,2006. Accessed March 24, 2010. http://searchenginewatch.com/2156451.
- Google.Google Translate Help;2010. Accessed March 24, 2010. http://translate.google.com/support/?hl=en.
- ,.Chapter 4: Basic strategies. In:An Introduction to Machine Translation;1992. Accessed April 22, 2010. http://www.hutchinsweb.me.uk/IntroMT‐4.pdf
- .Your Guide to Coumadin®/Warfarin Therapy.Agency for Healthcare Research and Quality; August 21,2008. Accessed October 19, 2009. http://www.ahrq.gov/consumer/btpills.htm.
- ,,, et al.Patient reported receipt of medication instructions for warfarin is associated with reduced risk of serious bleeding events.J Gen Intern Med.2008;23(10):1589–1594.
- ,,.The ARPA MT evaluation methodologies: evolution, lessons, and future approaches. In: Proceedings of AMTA, 1994, Columbia, MD; October1994.
- ,.Overview of the IWSLT 2005 evaluation campaign. In: Proceedings of IWSLT 2005, Pittsburgh, PA; October2005.
- ,,,.BLEU: a method for automatic evaluation of machine translation. In: ACL‐2002: 40th Annual Meeting of the Association for Computational Linguistics.2002:311–318.
- ,.METEOR: an automatic metric for MT evaluation with high levels of correlation with human judgments. In: Proceedings of the Second Workshop on Statistical Machine Translation at ACL, Prague, Czech Republic; June2007.
- .The Structure of a Sentence.Ottawa:The Writing Centre, University of Ottawa;2007.
- ,.Statistical methods for assessing agreement between two methods of clinical measurement.Lancet.1986;1(8476):307–310.
- .Measurement, reproducibility, and validity. In:Epidemiologic Methods 203.San Francisco:Department of Biostatistics and Epidemiology, University of California;2009.
- ,,,,.Getting by: underuse of interpreters by resident physicians.J Gen Intern Med.2009;24(2):256–262.
- ,.Language use and English‐speaking ability: 2000. In:Census 2000 Brief.Washington, DC:US Census Bureau;2003. p. 2. http://www.census.gov/prod/2003pubs/c2kbr‐29.pdf.
- ,,,,.The need for more research on language barriers in health care: a proposed research agenda.Milbank Q.2006;84(1):111–133.
- ,,,.Language proficiency and adverse events in US hospitals: a pilot study.Int J Qual Health Care.2007;19(2):60–67.
- .The impact of medical interpreter services on the quality of health care: a systematic review.Med Care Res Rev.2005;62(3):255–299.
- ,,, et al.Errors in medical interpretation and their potential clinical consequences in pediatric encounters.Pediatrics.2003;111(1):6–14.
- ,,, et al.The effect of English language proficiency on length of stay and in‐hospital mortality.J Gen Intern Med.2004;19(3):221–228.
- ,,,.Influence of language barriers on outcomes of hospital care for general medicine inpatients.J Hosp Med.2010;5(5):276–282.
- ,.Hospitals, language, and culture: a snapshot of the nation. In:Los Angeles, CA:The California Endowment, the Joint Commission;2007. p.51–52. http://www.jointcommission.org/assets/1/6/hlc_paper.pdf.
- ,,,.Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature.Health Serv Res.2007;42(2):727–754.
- .Google's Computing Power Refines Translation Tool.New York Times; March 9,2010. Accessed March 24, 2010. http://www.nytimes.com/2010/03/09/technology/09translate.html?_r=1.
- , Translator. New York Times; March 20,2010. Accessed March 24, 2010. http://www.nytimes.com/2010/03/21/opinion/21bellos.html.
- ,.Accuracy of computer‐generated, Spanish‐language medicine labels.Pediatrics.2010;125(5):960–965.
- .Nielsen NetRatings Search Engine Ratings.SearchEngineWatch; August 22,2006. Accessed March 24, 2010. http://searchenginewatch.com/2156451.
- Google.Google Translate Help;2010. Accessed March 24, 2010. http://translate.google.com/support/?hl=en.
- ,.Chapter 4: Basic strategies. In:An Introduction to Machine Translation;1992. Accessed April 22, 2010. http://www.hutchinsweb.me.uk/IntroMT‐4.pdf
- .Your Guide to Coumadin®/Warfarin Therapy.Agency for Healthcare Research and Quality; August 21,2008. Accessed October 19, 2009. http://www.ahrq.gov/consumer/btpills.htm.
- ,,, et al.Patient reported receipt of medication instructions for warfarin is associated with reduced risk of serious bleeding events.J Gen Intern Med.2008;23(10):1589–1594.
- ,,.The ARPA MT evaluation methodologies: evolution, lessons, and future approaches. In: Proceedings of AMTA, 1994, Columbia, MD; October1994.
- ,.Overview of the IWSLT 2005 evaluation campaign. In: Proceedings of IWSLT 2005, Pittsburgh, PA; October2005.
- ,,,.BLEU: a method for automatic evaluation of machine translation. In: ACL‐2002: 40th Annual Meeting of the Association for Computational Linguistics.2002:311–318.
- ,.METEOR: an automatic metric for MT evaluation with high levels of correlation with human judgments. In: Proceedings of the Second Workshop on Statistical Machine Translation at ACL, Prague, Czech Republic; June2007.
- .The Structure of a Sentence.Ottawa:The Writing Centre, University of Ottawa;2007.
- ,.Statistical methods for assessing agreement between two methods of clinical measurement.Lancet.1986;1(8476):307–310.
- .Measurement, reproducibility, and validity. In:Epidemiologic Methods 203.San Francisco:Department of Biostatistics and Epidemiology, University of California;2009.
- ,,,,.Getting by: underuse of interpreters by resident physicians.J Gen Intern Med.2009;24(2):256–262.
Copyright © 2011 Society of Hospital Medicine