User login
Glitter Effects of Nail Art on Optical Coherence Tomography
Practice Gap
Nail art can skew the results of optical coherence tomography (OCT), a noninvasive imaging technology that is used to visualize nail morphology in diseases such as psoriatic arthritis and onychomycosis, with a penetration depth of 2 mm and high-resolution images.1 Few studies have evaluated the effects of nail art on OCT. Saleah and colleagues1 found that clear, semitransparent, and red nail polishes do not interfere with visualization of the nail plate, whereas nontransparent gel polish and art stones obscure the image. They did not comment on the effect of glitter nail art in their study, though they did test 1 nail that contained glitter.1 Monpeurt et al2 compared matte and glossy nail polishes. They found that matte polish was readily identifiable from the nail plate, whereas glossy polish presented a greater number of artifacts.2
The Solution
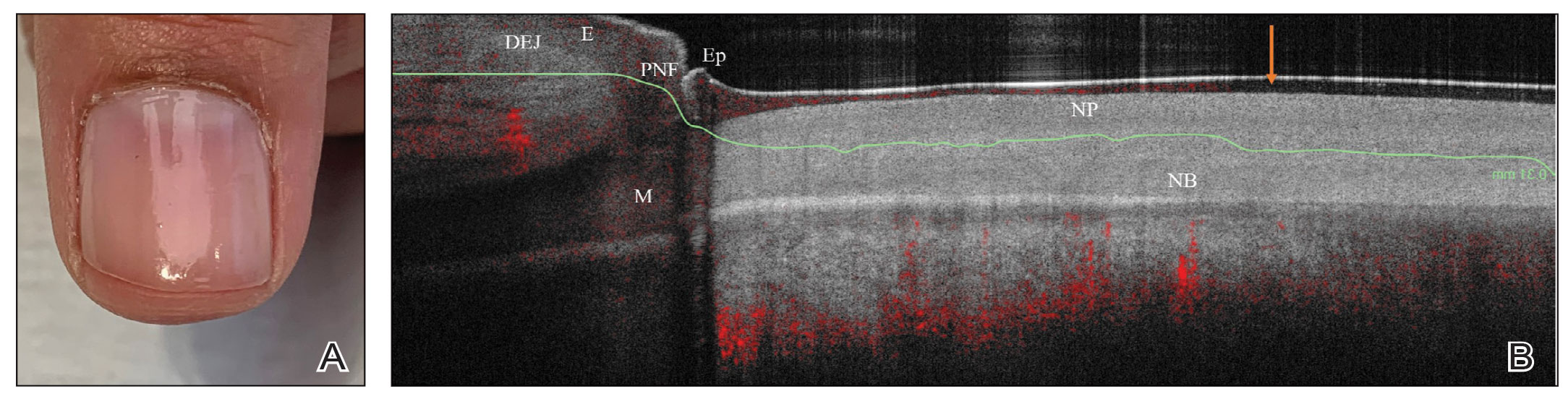
We looked at 3 glitter nail polishes—gold, pink, and silver—that were scanned by OCT to assess the effect of the polish on the resulting image. We determined that glitter particles completely obscured the nail bed and nail plate, regardless of color (Figure 1). Glossy clear polish imparted a distinct film on the top of the nail plate that did not obscure the nail plate or the nail bed (Figure 2).
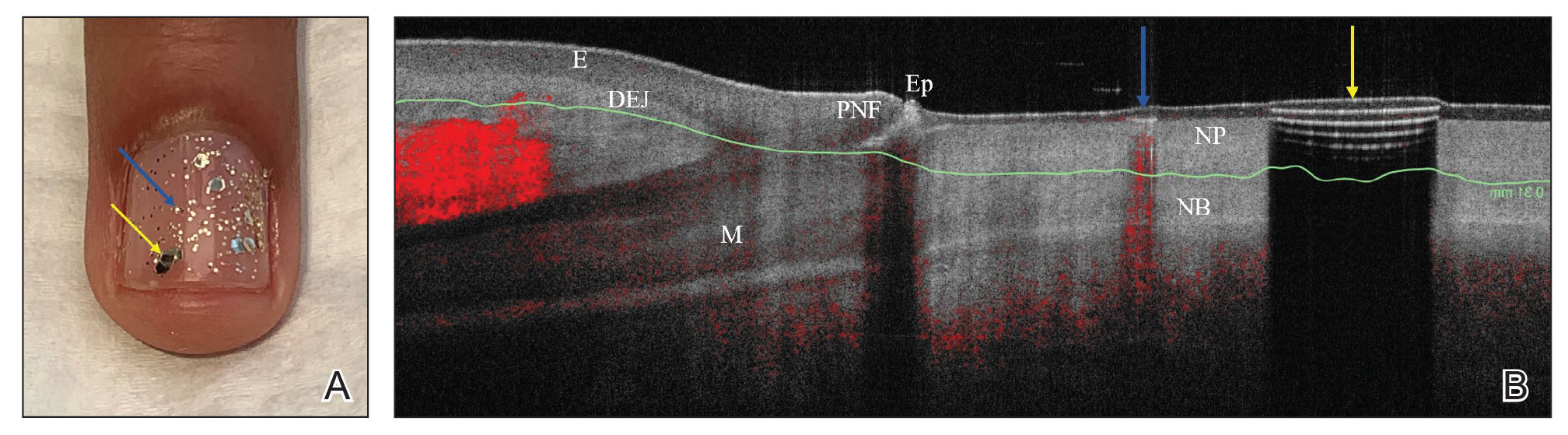
We conclude that glitter nail polish contains numerous reflective solid particles that interfere with OCT imaging of the nail plate and nail bed. As a result, we recommend removal of nail art to properly assess nail pathology. Because removal may need to be conducted by a nail technician, the treating clinician should inform the patient ahead of time to come to the appointment with bare (ie, unpolished) nails.
Practice Implications
Bringing awareness to the necessity of removing nail art prior to OCT imaging is crucial because many patients partake in its application, and removal may require the involvement of a professional nail technician. If a patient can be made aware that they should remove all nail art in advance, they will be better prepared for an OCT imaging session. Such a protocol increases efficiency, decreases diagnostic delay, and reduces cost associated with multiple office visits.
- Saleah S, Kim P, Seong D, et al. A preliminary study of post-progressive nail-art effects on in vivo nail plate using optical coherence tomography-based intensity profiling assessment. Sci Rep. 2021;11:666. doi:10.1038/s41598-020-79497-3
- Monpeurt C, Cinotti E, Hebert M, et al. Thickness and morphology assessment of nail polishes applied on nails by high-definition optical coherence tomography. Skin Res Technol. 2018;24:156-157. doi:10.1111/srt.12406
Practice Gap
Nail art can skew the results of optical coherence tomography (OCT), a noninvasive imaging technology that is used to visualize nail morphology in diseases such as psoriatic arthritis and onychomycosis, with a penetration depth of 2 mm and high-resolution images.1 Few studies have evaluated the effects of nail art on OCT. Saleah and colleagues1 found that clear, semitransparent, and red nail polishes do not interfere with visualization of the nail plate, whereas nontransparent gel polish and art stones obscure the image. They did not comment on the effect of glitter nail art in their study, though they did test 1 nail that contained glitter.1 Monpeurt et al2 compared matte and glossy nail polishes. They found that matte polish was readily identifiable from the nail plate, whereas glossy polish presented a greater number of artifacts.2
The Solution
We looked at 3 glitter nail polishes—gold, pink, and silver—that were scanned by OCT to assess the effect of the polish on the resulting image. We determined that glitter particles completely obscured the nail bed and nail plate, regardless of color (Figure 1). Glossy clear polish imparted a distinct film on the top of the nail plate that did not obscure the nail plate or the nail bed (Figure 2).

We conclude that glitter nail polish contains numerous reflective solid particles that interfere with OCT imaging of the nail plate and nail bed. As a result, we recommend removal of nail art to properly assess nail pathology. Because removal may need to be conducted by a nail technician, the treating clinician should inform the patient ahead of time to come to the appointment with bare (ie, unpolished) nails.

Practice Implications
Bringing awareness to the necessity of removing nail art prior to OCT imaging is crucial because many patients partake in its application, and removal may require the involvement of a professional nail technician. If a patient can be made aware that they should remove all nail art in advance, they will be better prepared for an OCT imaging session. Such a protocol increases efficiency, decreases diagnostic delay, and reduces cost associated with multiple office visits.
Practice Gap
Nail art can skew the results of optical coherence tomography (OCT), a noninvasive imaging technology that is used to visualize nail morphology in diseases such as psoriatic arthritis and onychomycosis, with a penetration depth of 2 mm and high-resolution images.1 Few studies have evaluated the effects of nail art on OCT. Saleah and colleagues1 found that clear, semitransparent, and red nail polishes do not interfere with visualization of the nail plate, whereas nontransparent gel polish and art stones obscure the image. They did not comment on the effect of glitter nail art in their study, though they did test 1 nail that contained glitter.1 Monpeurt et al2 compared matte and glossy nail polishes. They found that matte polish was readily identifiable from the nail plate, whereas glossy polish presented a greater number of artifacts.2
The Solution
We looked at 3 glitter nail polishes—gold, pink, and silver—that were scanned by OCT to assess the effect of the polish on the resulting image. We determined that glitter particles completely obscured the nail bed and nail plate, regardless of color (Figure 1). Glossy clear polish imparted a distinct film on the top of the nail plate that did not obscure the nail plate or the nail bed (Figure 2).

We conclude that glitter nail polish contains numerous reflective solid particles that interfere with OCT imaging of the nail plate and nail bed. As a result, we recommend removal of nail art to properly assess nail pathology. Because removal may need to be conducted by a nail technician, the treating clinician should inform the patient ahead of time to come to the appointment with bare (ie, unpolished) nails.

Practice Implications
Bringing awareness to the necessity of removing nail art prior to OCT imaging is crucial because many patients partake in its application, and removal may require the involvement of a professional nail technician. If a patient can be made aware that they should remove all nail art in advance, they will be better prepared for an OCT imaging session. Such a protocol increases efficiency, decreases diagnostic delay, and reduces cost associated with multiple office visits.
- Saleah S, Kim P, Seong D, et al. A preliminary study of post-progressive nail-art effects on in vivo nail plate using optical coherence tomography-based intensity profiling assessment. Sci Rep. 2021;11:666. doi:10.1038/s41598-020-79497-3
- Monpeurt C, Cinotti E, Hebert M, et al. Thickness and morphology assessment of nail polishes applied on nails by high-definition optical coherence tomography. Skin Res Technol. 2018;24:156-157. doi:10.1111/srt.12406
- Saleah S, Kim P, Seong D, et al. A preliminary study of post-progressive nail-art effects on in vivo nail plate using optical coherence tomography-based intensity profiling assessment. Sci Rep. 2021;11:666. doi:10.1038/s41598-020-79497-3
- Monpeurt C, Cinotti E, Hebert M, et al. Thickness and morphology assessment of nail polishes applied on nails by high-definition optical coherence tomography. Skin Res Technol. 2018;24:156-157. doi:10.1111/srt.12406
Lower racial disparity in melanoma diagnoses in vets than U.S. men overall, study finds
a new analysis shows.
“The trend of a lower racial disparity in the VA in the proportion of melanomas with local disease and in the proportion of distant metastasis at presentation was observed across age groups,” wrote Martin A. Weinstock MD, PhD, and Rachel K. Lim, of the department of dermatology at Brown University, Providence, R.I., and the Center for Dermatoepidemiology at the VA Providence Healthcare System. The study was published online in the Journal of the American Academy of Dermatology.
“Melanoma was the fourth-most common cancer [diagnosed] in male VA patients in 2010,” wrote the authors, who also pointed out that “prior surveys found that 11%-13% of U.S. active-duty personnel routinely use sunscreen despite significant occupational sun exposure. Racial disparities are important concerns in the VA and elsewhere.”
To compare the stage of melanoma at presentation among White and non-Whites patients in the VA and in the general U.S. population, the researchers identified invasive cutaneous melanoma cases from 2000 to 2019 in the VA Corporate Data Warehouse and the Surveillance, Epidemiology and End Results Program (SEER).
They restricted the analysis to men because of the small proportion of women in the at-risk veteran population and excluded cases with an age younger than 20, those with unknown histology, and melanoma in situ. The researchers performed two-tailed z-tests to evaluate the difference in proportions of melanoma stages between the veteran population and the general population.
The analysis included 44,077 cases of invasive melanoma in the VA and 217,030 in SEER. Racial disparities in melanoma staging were substantially less pronounced in the VA than in SEER.
In the VA, localized disease represented 77.9% of melanomas among Whites versus 71.0% among non-Whites. But in SEER, localized disease represented 80.7% of melanomas among Whites versus 61.5% in non-Whites – over double the VA disparity (P < .0001).
Likewise, the disparity between Whites and nonwhites observed for regional or distant metastatic disease at presentation in the VA was lower than the disparity observed in SEER. For example, in the VA, distant metastatic disease at presentation represented 6.1% of melanomas among Whites versus 8.6% among non-Whites, while in SEER it represented 4.8% of melanomas among Whites versus 11.3% in non-Whites – again, more than double the VA disparity (P < .0001).
“These differences between the VA and SEER were less marked” among those older than 65 years, the researchers wrote. “Notably, the differences between VA and SEER in racial disparities among those greater than 65 in age were still significant for localized disease and for distant metastasis.”
The findings suggest that the VA “may be more effective in reducing racial disparities in melanoma stage at diagnosis, potentially due to all patients in the VA dataset having insured access to health care, regardless of socioeconomic status,” the researchers concluded. Similarly, the decreased difference in racial disparities observed in patients older than 65 across systems “may be related to the availability of Medicare to the older general populations. The authors acknowledged several study limitations, such as the predominantly elderly and male VA population, potentially underreported utilization of non-VA dermatologic care, and variation in geographic regions covered by each database.
Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, who was asked to comment on the work, said in an interview he would have liked to see a more detailed breakdown of the younger patients, “for those in their 30s and 40s, to see if this trend held up.”
He would have also liked to see how the data trended over time, adding, “while this, broadly, may be good news for our veterans, attributing this finding to a reduction in access disparity or some other organizational intervention seems a little premature. Regardless, Dr. Weinstock has given us, once again, information from our veterans to probe for the betterment of all patients.”
The researchers reported having no relevant disclosures and the study had no funding. Dr. Blalock disclosed that he has served as a principal investigator for Castle Biosciences.
a new analysis shows.
“The trend of a lower racial disparity in the VA in the proportion of melanomas with local disease and in the proportion of distant metastasis at presentation was observed across age groups,” wrote Martin A. Weinstock MD, PhD, and Rachel K. Lim, of the department of dermatology at Brown University, Providence, R.I., and the Center for Dermatoepidemiology at the VA Providence Healthcare System. The study was published online in the Journal of the American Academy of Dermatology.
“Melanoma was the fourth-most common cancer [diagnosed] in male VA patients in 2010,” wrote the authors, who also pointed out that “prior surveys found that 11%-13% of U.S. active-duty personnel routinely use sunscreen despite significant occupational sun exposure. Racial disparities are important concerns in the VA and elsewhere.”
To compare the stage of melanoma at presentation among White and non-Whites patients in the VA and in the general U.S. population, the researchers identified invasive cutaneous melanoma cases from 2000 to 2019 in the VA Corporate Data Warehouse and the Surveillance, Epidemiology and End Results Program (SEER).
They restricted the analysis to men because of the small proportion of women in the at-risk veteran population and excluded cases with an age younger than 20, those with unknown histology, and melanoma in situ. The researchers performed two-tailed z-tests to evaluate the difference in proportions of melanoma stages between the veteran population and the general population.
The analysis included 44,077 cases of invasive melanoma in the VA and 217,030 in SEER. Racial disparities in melanoma staging were substantially less pronounced in the VA than in SEER.
In the VA, localized disease represented 77.9% of melanomas among Whites versus 71.0% among non-Whites. But in SEER, localized disease represented 80.7% of melanomas among Whites versus 61.5% in non-Whites – over double the VA disparity (P < .0001).
Likewise, the disparity between Whites and nonwhites observed for regional or distant metastatic disease at presentation in the VA was lower than the disparity observed in SEER. For example, in the VA, distant metastatic disease at presentation represented 6.1% of melanomas among Whites versus 8.6% among non-Whites, while in SEER it represented 4.8% of melanomas among Whites versus 11.3% in non-Whites – again, more than double the VA disparity (P < .0001).
“These differences between the VA and SEER were less marked” among those older than 65 years, the researchers wrote. “Notably, the differences between VA and SEER in racial disparities among those greater than 65 in age were still significant for localized disease and for distant metastasis.”
The findings suggest that the VA “may be more effective in reducing racial disparities in melanoma stage at diagnosis, potentially due to all patients in the VA dataset having insured access to health care, regardless of socioeconomic status,” the researchers concluded. Similarly, the decreased difference in racial disparities observed in patients older than 65 across systems “may be related to the availability of Medicare to the older general populations. The authors acknowledged several study limitations, such as the predominantly elderly and male VA population, potentially underreported utilization of non-VA dermatologic care, and variation in geographic regions covered by each database.
Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, who was asked to comment on the work, said in an interview he would have liked to see a more detailed breakdown of the younger patients, “for those in their 30s and 40s, to see if this trend held up.”
He would have also liked to see how the data trended over time, adding, “while this, broadly, may be good news for our veterans, attributing this finding to a reduction in access disparity or some other organizational intervention seems a little premature. Regardless, Dr. Weinstock has given us, once again, information from our veterans to probe for the betterment of all patients.”
The researchers reported having no relevant disclosures and the study had no funding. Dr. Blalock disclosed that he has served as a principal investigator for Castle Biosciences.
a new analysis shows.
“The trend of a lower racial disparity in the VA in the proportion of melanomas with local disease and in the proportion of distant metastasis at presentation was observed across age groups,” wrote Martin A. Weinstock MD, PhD, and Rachel K. Lim, of the department of dermatology at Brown University, Providence, R.I., and the Center for Dermatoepidemiology at the VA Providence Healthcare System. The study was published online in the Journal of the American Academy of Dermatology.
“Melanoma was the fourth-most common cancer [diagnosed] in male VA patients in 2010,” wrote the authors, who also pointed out that “prior surveys found that 11%-13% of U.S. active-duty personnel routinely use sunscreen despite significant occupational sun exposure. Racial disparities are important concerns in the VA and elsewhere.”
To compare the stage of melanoma at presentation among White and non-Whites patients in the VA and in the general U.S. population, the researchers identified invasive cutaneous melanoma cases from 2000 to 2019 in the VA Corporate Data Warehouse and the Surveillance, Epidemiology and End Results Program (SEER).
They restricted the analysis to men because of the small proportion of women in the at-risk veteran population and excluded cases with an age younger than 20, those with unknown histology, and melanoma in situ. The researchers performed two-tailed z-tests to evaluate the difference in proportions of melanoma stages between the veteran population and the general population.
The analysis included 44,077 cases of invasive melanoma in the VA and 217,030 in SEER. Racial disparities in melanoma staging were substantially less pronounced in the VA than in SEER.
In the VA, localized disease represented 77.9% of melanomas among Whites versus 71.0% among non-Whites. But in SEER, localized disease represented 80.7% of melanomas among Whites versus 61.5% in non-Whites – over double the VA disparity (P < .0001).
Likewise, the disparity between Whites and nonwhites observed for regional or distant metastatic disease at presentation in the VA was lower than the disparity observed in SEER. For example, in the VA, distant metastatic disease at presentation represented 6.1% of melanomas among Whites versus 8.6% among non-Whites, while in SEER it represented 4.8% of melanomas among Whites versus 11.3% in non-Whites – again, more than double the VA disparity (P < .0001).
“These differences between the VA and SEER were less marked” among those older than 65 years, the researchers wrote. “Notably, the differences between VA and SEER in racial disparities among those greater than 65 in age were still significant for localized disease and for distant metastasis.”
The findings suggest that the VA “may be more effective in reducing racial disparities in melanoma stage at diagnosis, potentially due to all patients in the VA dataset having insured access to health care, regardless of socioeconomic status,” the researchers concluded. Similarly, the decreased difference in racial disparities observed in patients older than 65 across systems “may be related to the availability of Medicare to the older general populations. The authors acknowledged several study limitations, such as the predominantly elderly and male VA population, potentially underreported utilization of non-VA dermatologic care, and variation in geographic regions covered by each database.
Travis W. Blalock, MD, director of dermatologic surgery, Mohs micrographic surgery, and cutaneous oncology at Emory University, Atlanta, who was asked to comment on the work, said in an interview he would have liked to see a more detailed breakdown of the younger patients, “for those in their 30s and 40s, to see if this trend held up.”
He would have also liked to see how the data trended over time, adding, “while this, broadly, may be good news for our veterans, attributing this finding to a reduction in access disparity or some other organizational intervention seems a little premature. Regardless, Dr. Weinstock has given us, once again, information from our veterans to probe for the betterment of all patients.”
The researchers reported having no relevant disclosures and the study had no funding. Dr. Blalock disclosed that he has served as a principal investigator for Castle Biosciences.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Continuous glucose monitors come to hospitals
But that technological future will require ensuring that the monitoring devices are as accurate as the conventional method, experts told this news organization.
In 2020, the U.S. Food and Drug Administration enabled in-hospital use of CGMs to reduce contact between patients and health care providers during the COVID-19 pandemic. Diabetes is a risk factor for more severe COVID, meaning that many patients with the infection also required ongoing care for their blood sugar problems.
Prior to the pandemic, in-person finger-stick tests were the primary means of measuring glucose for hospitalized patients with diabetes.
The trouble is that finger-stick measurements quickly become inaccurate.
“Glucose is a measurement that changes pretty rapidly,” said Eileen Faulds, RN, PhD, an endocrinology nurse and health services researcher at the Ohio State University, Columbus. Finger sticks might occur only four or five times per day, Dr. Faulds noted, or as often as every hour for people who receive insulin intravenously. But even that more frequent pace is far from continuous.
“With CGM we can get the glucose level in real time,” Dr. Faulds said.
Dr. Faulds is lead author of a new study in the Journal of Diabetes Science and Technology, which shows that nurses in the ICU believe that using continuous monitors, subcutaneous filaments connected to sensors that regularly report glucose levels, enables better patient care than does relying on periodic glucose tests alone. Nurses still used traditional finger sticks, which Dr. Faulds notes are highly accurate at the time of the reading.
In a 2022 study, glucose levels generated by CGM and those measured by finger sticks varied by up to 14%. A hybrid care model combining CGMs and finger stick tests may emerge, Dr. Faulds said.
A gusher of glucose data
People with diabetes have long been able to use CGMs in their daily lives, which typically report the glucose value to a smartphone or watch. The devices are now part of hospital care as well. In 2022, the Food and Drug Administration granted a breakthrough therapy designation to the company Dexcom for use of its CGMs to manage care of people with diabetes in hospitals.
One open question is how often CGMs should report glucose readings for optimum patient health. Dexcom’s G6 CGM reports glucose levels every five minutes, for example, whereas Abbott’s FreeStyle Libre 2 delivers glucose values every minute.
“We wouldn’t look at each value, we would look at the big picture,” to determine if a patient is at risk of becoming hyper- or hypoglycemic, said Lizda Guerrero-Arroyo, MD, a postdoctoral fellow in endocrinology at the Emory University School of Medicine, Atlanta. Dr. Guerrero-Arroyo recently reported that clinicians in multiple ICUs began to use CGMs in conjunction with finger sticks during the pandemic and felt the devices could reduce patient discomfort.
“A finger stick is very painful,” Dr. Guerrero-Arroyo said, and a bottleneck for nursing staff who administer these tests. In contrast, Dr. Faulds said, CGM placement is essentially painless and requires less labor on the ward to manage.
Beyond use in the ICU, clinicians are also experimenting with use of CGMs to monitor blood sugar levels in people with diabetes who are undergoing general surgery. And other researchers are describing how to integrate data from CGMs into patient care tools such as the electronic health record, although a standard way to do this does not yet exist.
Assuming CGMs remain part of the mix for in-hospital care of people with diabetes, clinicians may mainly need trend summaries of how glucose levels rise and fall over time, said data scientist Samantha Spierling Bagsic, PhD, of the Scripps Whittier Diabetes Institute, San Diego. Dr. Guerrero-Arroyo said that she shares that vision. But a minute-by-minute analysis of glucose levels also may be necessary to get a granular sense of how changing a patient’s insulin level affects their blood sugar, Dr. Spierling Bagsic said.
“We need to figure out what data different audiences need, how often we need to measure glucose, and how to present that information to different audiences in different ways,” said Dr. Spierling Bagsic, a co-author of the study about integrating CGM data into patient care tools.
The wider use of CGMs in hospitals may be one silver lining of the COVID-19 pandemic. As an inpatient endocrinology nurse, Dr. Faulds said that she wanted to use CGMs prior to the outbreak, but at that point, a critical mass of studies about their benefits was missing.
“We all know the terrible things that happened during the pandemic,” Dr. Faulds said. “But it gave us the allowance to use CGMs, and we saw that nurses loved them.”
Dr. Faulds reports relationships with Dexcom and Insulet and has received an honorarium from Medscape. Dr. Guerrero-Arroyo and Dr. Spierling Bagsic reported no financial conflicts of interest.
A version of this article originally appeared on Medscape.com.
But that technological future will require ensuring that the monitoring devices are as accurate as the conventional method, experts told this news organization.
In 2020, the U.S. Food and Drug Administration enabled in-hospital use of CGMs to reduce contact between patients and health care providers during the COVID-19 pandemic. Diabetes is a risk factor for more severe COVID, meaning that many patients with the infection also required ongoing care for their blood sugar problems.
Prior to the pandemic, in-person finger-stick tests were the primary means of measuring glucose for hospitalized patients with diabetes.
The trouble is that finger-stick measurements quickly become inaccurate.
“Glucose is a measurement that changes pretty rapidly,” said Eileen Faulds, RN, PhD, an endocrinology nurse and health services researcher at the Ohio State University, Columbus. Finger sticks might occur only four or five times per day, Dr. Faulds noted, or as often as every hour for people who receive insulin intravenously. But even that more frequent pace is far from continuous.
“With CGM we can get the glucose level in real time,” Dr. Faulds said.
Dr. Faulds is lead author of a new study in the Journal of Diabetes Science and Technology, which shows that nurses in the ICU believe that using continuous monitors, subcutaneous filaments connected to sensors that regularly report glucose levels, enables better patient care than does relying on periodic glucose tests alone. Nurses still used traditional finger sticks, which Dr. Faulds notes are highly accurate at the time of the reading.
In a 2022 study, glucose levels generated by CGM and those measured by finger sticks varied by up to 14%. A hybrid care model combining CGMs and finger stick tests may emerge, Dr. Faulds said.
A gusher of glucose data
People with diabetes have long been able to use CGMs in their daily lives, which typically report the glucose value to a smartphone or watch. The devices are now part of hospital care as well. In 2022, the Food and Drug Administration granted a breakthrough therapy designation to the company Dexcom for use of its CGMs to manage care of people with diabetes in hospitals.
One open question is how often CGMs should report glucose readings for optimum patient health. Dexcom’s G6 CGM reports glucose levels every five minutes, for example, whereas Abbott’s FreeStyle Libre 2 delivers glucose values every minute.
“We wouldn’t look at each value, we would look at the big picture,” to determine if a patient is at risk of becoming hyper- or hypoglycemic, said Lizda Guerrero-Arroyo, MD, a postdoctoral fellow in endocrinology at the Emory University School of Medicine, Atlanta. Dr. Guerrero-Arroyo recently reported that clinicians in multiple ICUs began to use CGMs in conjunction with finger sticks during the pandemic and felt the devices could reduce patient discomfort.
“A finger stick is very painful,” Dr. Guerrero-Arroyo said, and a bottleneck for nursing staff who administer these tests. In contrast, Dr. Faulds said, CGM placement is essentially painless and requires less labor on the ward to manage.
Beyond use in the ICU, clinicians are also experimenting with use of CGMs to monitor blood sugar levels in people with diabetes who are undergoing general surgery. And other researchers are describing how to integrate data from CGMs into patient care tools such as the electronic health record, although a standard way to do this does not yet exist.
Assuming CGMs remain part of the mix for in-hospital care of people with diabetes, clinicians may mainly need trend summaries of how glucose levels rise and fall over time, said data scientist Samantha Spierling Bagsic, PhD, of the Scripps Whittier Diabetes Institute, San Diego. Dr. Guerrero-Arroyo said that she shares that vision. But a minute-by-minute analysis of glucose levels also may be necessary to get a granular sense of how changing a patient’s insulin level affects their blood sugar, Dr. Spierling Bagsic said.
“We need to figure out what data different audiences need, how often we need to measure glucose, and how to present that information to different audiences in different ways,” said Dr. Spierling Bagsic, a co-author of the study about integrating CGM data into patient care tools.
The wider use of CGMs in hospitals may be one silver lining of the COVID-19 pandemic. As an inpatient endocrinology nurse, Dr. Faulds said that she wanted to use CGMs prior to the outbreak, but at that point, a critical mass of studies about their benefits was missing.
“We all know the terrible things that happened during the pandemic,” Dr. Faulds said. “But it gave us the allowance to use CGMs, and we saw that nurses loved them.”
Dr. Faulds reports relationships with Dexcom and Insulet and has received an honorarium from Medscape. Dr. Guerrero-Arroyo and Dr. Spierling Bagsic reported no financial conflicts of interest.
A version of this article originally appeared on Medscape.com.
But that technological future will require ensuring that the monitoring devices are as accurate as the conventional method, experts told this news organization.
In 2020, the U.S. Food and Drug Administration enabled in-hospital use of CGMs to reduce contact between patients and health care providers during the COVID-19 pandemic. Diabetes is a risk factor for more severe COVID, meaning that many patients with the infection also required ongoing care for their blood sugar problems.
Prior to the pandemic, in-person finger-stick tests were the primary means of measuring glucose for hospitalized patients with diabetes.
The trouble is that finger-stick measurements quickly become inaccurate.
“Glucose is a measurement that changes pretty rapidly,” said Eileen Faulds, RN, PhD, an endocrinology nurse and health services researcher at the Ohio State University, Columbus. Finger sticks might occur only four or five times per day, Dr. Faulds noted, or as often as every hour for people who receive insulin intravenously. But even that more frequent pace is far from continuous.
“With CGM we can get the glucose level in real time,” Dr. Faulds said.
Dr. Faulds is lead author of a new study in the Journal of Diabetes Science and Technology, which shows that nurses in the ICU believe that using continuous monitors, subcutaneous filaments connected to sensors that regularly report glucose levels, enables better patient care than does relying on periodic glucose tests alone. Nurses still used traditional finger sticks, which Dr. Faulds notes are highly accurate at the time of the reading.
In a 2022 study, glucose levels generated by CGM and those measured by finger sticks varied by up to 14%. A hybrid care model combining CGMs and finger stick tests may emerge, Dr. Faulds said.
A gusher of glucose data
People with diabetes have long been able to use CGMs in their daily lives, which typically report the glucose value to a smartphone or watch. The devices are now part of hospital care as well. In 2022, the Food and Drug Administration granted a breakthrough therapy designation to the company Dexcom for use of its CGMs to manage care of people with diabetes in hospitals.
One open question is how often CGMs should report glucose readings for optimum patient health. Dexcom’s G6 CGM reports glucose levels every five minutes, for example, whereas Abbott’s FreeStyle Libre 2 delivers glucose values every minute.
“We wouldn’t look at each value, we would look at the big picture,” to determine if a patient is at risk of becoming hyper- or hypoglycemic, said Lizda Guerrero-Arroyo, MD, a postdoctoral fellow in endocrinology at the Emory University School of Medicine, Atlanta. Dr. Guerrero-Arroyo recently reported that clinicians in multiple ICUs began to use CGMs in conjunction with finger sticks during the pandemic and felt the devices could reduce patient discomfort.
“A finger stick is very painful,” Dr. Guerrero-Arroyo said, and a bottleneck for nursing staff who administer these tests. In contrast, Dr. Faulds said, CGM placement is essentially painless and requires less labor on the ward to manage.
Beyond use in the ICU, clinicians are also experimenting with use of CGMs to monitor blood sugar levels in people with diabetes who are undergoing general surgery. And other researchers are describing how to integrate data from CGMs into patient care tools such as the electronic health record, although a standard way to do this does not yet exist.
Assuming CGMs remain part of the mix for in-hospital care of people with diabetes, clinicians may mainly need trend summaries of how glucose levels rise and fall over time, said data scientist Samantha Spierling Bagsic, PhD, of the Scripps Whittier Diabetes Institute, San Diego. Dr. Guerrero-Arroyo said that she shares that vision. But a minute-by-minute analysis of glucose levels also may be necessary to get a granular sense of how changing a patient’s insulin level affects their blood sugar, Dr. Spierling Bagsic said.
“We need to figure out what data different audiences need, how often we need to measure glucose, and how to present that information to different audiences in different ways,” said Dr. Spierling Bagsic, a co-author of the study about integrating CGM data into patient care tools.
The wider use of CGMs in hospitals may be one silver lining of the COVID-19 pandemic. As an inpatient endocrinology nurse, Dr. Faulds said that she wanted to use CGMs prior to the outbreak, but at that point, a critical mass of studies about their benefits was missing.
“We all know the terrible things that happened during the pandemic,” Dr. Faulds said. “But it gave us the allowance to use CGMs, and we saw that nurses loved them.”
Dr. Faulds reports relationships with Dexcom and Insulet and has received an honorarium from Medscape. Dr. Guerrero-Arroyo and Dr. Spierling Bagsic reported no financial conflicts of interest.
A version of this article originally appeared on Medscape.com.
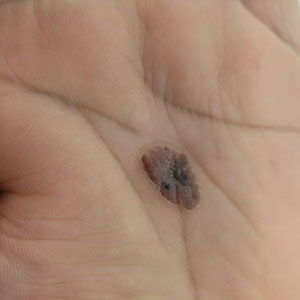
Oval Brown Plaque on the Palm
The Diagnosis: Poroma
Histopathology showed an endophytic expansion of the epidermis by bland, uniform, basaloid epithelial cells with focal ductal differentiation and an abrupt transition with surrounding epidermal keratinocytes (Figure), consistent with a diagnosis of poroma. The patient elected to monitor the lesion rather than to have it excised.
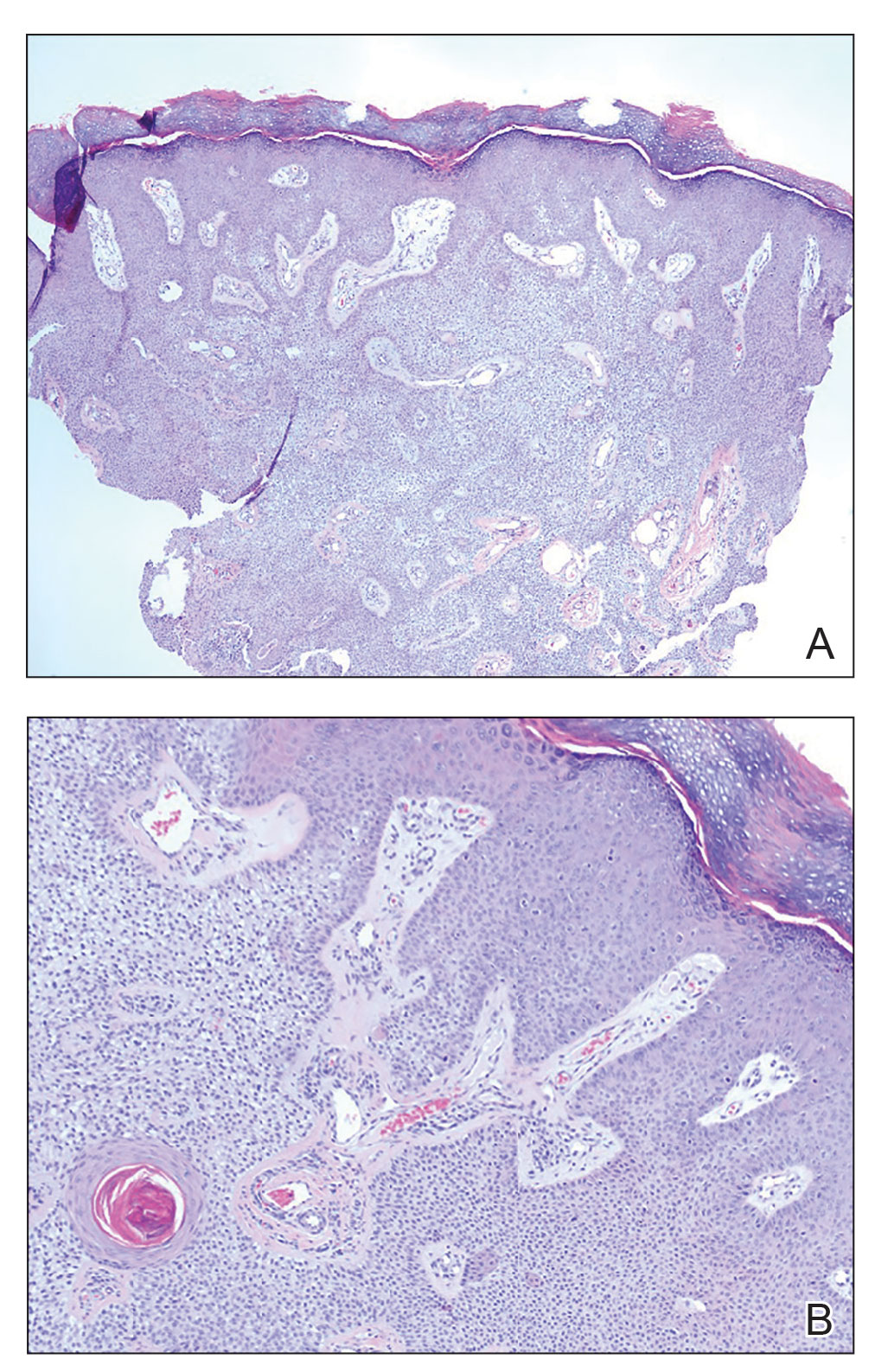
Eccrine poroma, used interchangeably with the term poroma, is a rare benign adnexal tumor of the eccrine sweat glands resulting from proliferation of the acrosyringium.1,2 It often occurs on the palms or soles, though it also can arise anywhere sweat glands are present.1 Eccrine poromas often appear in middle-aged individuals as singular, well-circumscribed, red-brown papules or nodules.3 A characteristic feature is a shallow, cup-shaped depression within the larger papule or nodule.1
Because the condition is benign and often asymptomatic, it can be safely monitored for progression.1 However, if the lesion is symptomatic or located in a sensitive area, complete excision is curative.4 Eccrine poromas can recur, making close monitoring following excision important.5 The development of bleeding, itching, or pain in a previously asymptomatic lesion may indicate possible malignant transformation, which occurs in only 18% of cases.6
The differential diagnosis includes basal cell carcinoma, circumscribed acral hypokeratosis, Kaposi sarcoma, and pyogenic granuloma. Basal cell carcinoma is the most common type of skin cancer.7 In rare cases it has been shown to present on the palms or soles as a slowgrowing, reddish-pink papule or plaque with central ulceration. It typically is asymptomatic. Histopathology shows dermal nests of basaloid cells with peripheral palisading, stromal mucin, and peritumoral clefts. Treatment is surgical excision.7
Circumscribed acral hypokeratosis presents on the palms or soles as a solitary, shallow, well-defined lesion with a flat base and raised border.8 It often is red-pink in color and most frequently occurs in middle-aged women. Although the cause of the condition is unknown, it is thought to be the result of trauma or human papillomavirus infection.8 Biopsy results characteristically show hypokeratosis demarcated by a sharp and frayed cutoff from uninvolved acral skin with discrete hypogranulosis, dilated blood vessels in the papillary dermis, and slightly thickened collagen fibers in the reticular dermis.9 Surgical excision is a potential treatment option, as topical corticosteroids, retinoids, and calcipotriene have not been shown to be effective; spontaneous resolution has been reported.8
Kaposi sarcoma is a vascular neoplasm that is associated with human herpesvirus 8 infection.10 It typically presents on mucocutaneous sites and the lower extremities. Palmar involvement has been reported in rare cases, occurring as a solitary, well-demarcated, violaceous macule or patch that may be painful.10-12 Characteristic histopathologic features include a proliferation in the dermis of slitlike vascular spaces and spindle cell proliferation.13 Treatment options include cryosurgery; pulsed dye laser; and topical, intralesional, or systemic chemotherapy agents, depending on the stage of the patient’s disease. Antiretroviral therapy is indicated for patients with Kaposi sarcoma secondary to AIDS.14
Pyogenic granuloma presents as a solitary red-brown or bluish-black papule or nodule that bleeds easily when manipulated.15 It commonly occurs following trauma, typically on the fingers, feet, and lips.6 Although benign, potential complications include ulceration and blood loss. Pyogenic granulomas can be treated via curettage and cautery, excision, cryosurgery, or pulsed dye laser.15
- Wankhade V, Singh R, Sadhwani V, et al. Eccrine poroma. Indian Dermatol Online J. 2015;6:304-305.
- Yorulmaz A, Aksoy GG, Ozhamam EU. A growing mass under the nail: subungual eccrine poroma. Skin Appendage Disord. 2020;6:254-257.
- Wang Y, Liu M, Zheng Y, et al. Eccrine poroma presented as spindleshaped plaque: a case report. Medicine (Baltimore). 2021;100:E25971. doi:10.1097/MD.0000000000025971
- Sharma M, Singh M, Gupta K, et al. Eccrine poroma of the eyelid. Indian J Ophthalmol. 2020;68:2522.
- Rasool MN, Hawary MB. Benign eccrine poroma in the palm of the hand. Ann Saudi Med. 2004;24:46-47.
- Sawaya JL, Khachemoune A. Poroma: a review of eccrine, apocrine, and malignant forms [published online April 2, 2014]. Int J Dermatol. 2014;53:1053-1061. doi:10.1111/ijd.12448
- López-Sánchez C, Ferguson P, Collgros H. Basal cell carcinoma of the palm: an unusual presentation of a common tumour [published online August 6, 2019]. Australas J Dermatol. 2020;61:69-70. doi:10.1111/ajd.13129
- Berk DR, Böer A, Bauschard FD, et al. Circumscribed acral hypokeratosis [published online April 6, 2007]. J Am Acad Dermatol. 2007;57:292-296. doi:10.1016/j.jaad.2007.02.022
- Majluf-Cáceres P, Vera-Kellet C, González-Bombardiere S. New dermoscopic keys for circumscribed acral hypokeratosis: report of four cases. Dermatol Pract Concept. 2021;11:E2021010. doi:10.5826/dpc.1102a10
- Simonart T, De Dobbeleer G, Stallenberg B. Classic Kaposi’s sarcoma of the palm in a metallurgist: role of iron filings in its development? Br J Dermatol. 2003;148:1061-1063. doi:10.1046/j.1365-2133.2003.05331.x
- Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294. doi:10.5858/arpa.2012-0101-RS
- Al Zolibani AA, Al Robaee AA. Primary palmoplantar Kaposi’s sarcoma: an unusual presentation. Skinmed. 2006;5:248-249. doi:10.1111/j.1540-9740.2006.04662.x
- Cesarman E, Damania B, Krown SE, et al. Kaposi sarcoma. Nat Rev Dis Primers. 2019;5:9. doi:10.1038/s41572-019-0060-9
- Etemad SA, Dewan AK. Kaposi sarcoma updates [published online July 10, 2019]. Dermatol Clin. 2019;37:505-517. doi:10.1016/j. det.2019.05.008
- Murthy SC, Nagaraj A. Pyogenic granuloma. Indian Pediatr. 2012;49:855. doi:10.1007/s13312-012-0184-4
The Diagnosis: Poroma
Histopathology showed an endophytic expansion of the epidermis by bland, uniform, basaloid epithelial cells with focal ductal differentiation and an abrupt transition with surrounding epidermal keratinocytes (Figure), consistent with a diagnosis of poroma. The patient elected to monitor the lesion rather than to have it excised.

Eccrine poroma, used interchangeably with the term poroma, is a rare benign adnexal tumor of the eccrine sweat glands resulting from proliferation of the acrosyringium.1,2 It often occurs on the palms or soles, though it also can arise anywhere sweat glands are present.1 Eccrine poromas often appear in middle-aged individuals as singular, well-circumscribed, red-brown papules or nodules.3 A characteristic feature is a shallow, cup-shaped depression within the larger papule or nodule.1
Because the condition is benign and often asymptomatic, it can be safely monitored for progression.1 However, if the lesion is symptomatic or located in a sensitive area, complete excision is curative.4 Eccrine poromas can recur, making close monitoring following excision important.5 The development of bleeding, itching, or pain in a previously asymptomatic lesion may indicate possible malignant transformation, which occurs in only 18% of cases.6
The differential diagnosis includes basal cell carcinoma, circumscribed acral hypokeratosis, Kaposi sarcoma, and pyogenic granuloma. Basal cell carcinoma is the most common type of skin cancer.7 In rare cases it has been shown to present on the palms or soles as a slowgrowing, reddish-pink papule or plaque with central ulceration. It typically is asymptomatic. Histopathology shows dermal nests of basaloid cells with peripheral palisading, stromal mucin, and peritumoral clefts. Treatment is surgical excision.7
Circumscribed acral hypokeratosis presents on the palms or soles as a solitary, shallow, well-defined lesion with a flat base and raised border.8 It often is red-pink in color and most frequently occurs in middle-aged women. Although the cause of the condition is unknown, it is thought to be the result of trauma or human papillomavirus infection.8 Biopsy results characteristically show hypokeratosis demarcated by a sharp and frayed cutoff from uninvolved acral skin with discrete hypogranulosis, dilated blood vessels in the papillary dermis, and slightly thickened collagen fibers in the reticular dermis.9 Surgical excision is a potential treatment option, as topical corticosteroids, retinoids, and calcipotriene have not been shown to be effective; spontaneous resolution has been reported.8
Kaposi sarcoma is a vascular neoplasm that is associated with human herpesvirus 8 infection.10 It typically presents on mucocutaneous sites and the lower extremities. Palmar involvement has been reported in rare cases, occurring as a solitary, well-demarcated, violaceous macule or patch that may be painful.10-12 Characteristic histopathologic features include a proliferation in the dermis of slitlike vascular spaces and spindle cell proliferation.13 Treatment options include cryosurgery; pulsed dye laser; and topical, intralesional, or systemic chemotherapy agents, depending on the stage of the patient’s disease. Antiretroviral therapy is indicated for patients with Kaposi sarcoma secondary to AIDS.14
Pyogenic granuloma presents as a solitary red-brown or bluish-black papule or nodule that bleeds easily when manipulated.15 It commonly occurs following trauma, typically on the fingers, feet, and lips.6 Although benign, potential complications include ulceration and blood loss. Pyogenic granulomas can be treated via curettage and cautery, excision, cryosurgery, or pulsed dye laser.15
The Diagnosis: Poroma
Histopathology showed an endophytic expansion of the epidermis by bland, uniform, basaloid epithelial cells with focal ductal differentiation and an abrupt transition with surrounding epidermal keratinocytes (Figure), consistent with a diagnosis of poroma. The patient elected to monitor the lesion rather than to have it excised.

Eccrine poroma, used interchangeably with the term poroma, is a rare benign adnexal tumor of the eccrine sweat glands resulting from proliferation of the acrosyringium.1,2 It often occurs on the palms or soles, though it also can arise anywhere sweat glands are present.1 Eccrine poromas often appear in middle-aged individuals as singular, well-circumscribed, red-brown papules or nodules.3 A characteristic feature is a shallow, cup-shaped depression within the larger papule or nodule.1
Because the condition is benign and often asymptomatic, it can be safely monitored for progression.1 However, if the lesion is symptomatic or located in a sensitive area, complete excision is curative.4 Eccrine poromas can recur, making close monitoring following excision important.5 The development of bleeding, itching, or pain in a previously asymptomatic lesion may indicate possible malignant transformation, which occurs in only 18% of cases.6
The differential diagnosis includes basal cell carcinoma, circumscribed acral hypokeratosis, Kaposi sarcoma, and pyogenic granuloma. Basal cell carcinoma is the most common type of skin cancer.7 In rare cases it has been shown to present on the palms or soles as a slowgrowing, reddish-pink papule or plaque with central ulceration. It typically is asymptomatic. Histopathology shows dermal nests of basaloid cells with peripheral palisading, stromal mucin, and peritumoral clefts. Treatment is surgical excision.7
Circumscribed acral hypokeratosis presents on the palms or soles as a solitary, shallow, well-defined lesion with a flat base and raised border.8 It often is red-pink in color and most frequently occurs in middle-aged women. Although the cause of the condition is unknown, it is thought to be the result of trauma or human papillomavirus infection.8 Biopsy results characteristically show hypokeratosis demarcated by a sharp and frayed cutoff from uninvolved acral skin with discrete hypogranulosis, dilated blood vessels in the papillary dermis, and slightly thickened collagen fibers in the reticular dermis.9 Surgical excision is a potential treatment option, as topical corticosteroids, retinoids, and calcipotriene have not been shown to be effective; spontaneous resolution has been reported.8
Kaposi sarcoma is a vascular neoplasm that is associated with human herpesvirus 8 infection.10 It typically presents on mucocutaneous sites and the lower extremities. Palmar involvement has been reported in rare cases, occurring as a solitary, well-demarcated, violaceous macule or patch that may be painful.10-12 Characteristic histopathologic features include a proliferation in the dermis of slitlike vascular spaces and spindle cell proliferation.13 Treatment options include cryosurgery; pulsed dye laser; and topical, intralesional, or systemic chemotherapy agents, depending on the stage of the patient’s disease. Antiretroviral therapy is indicated for patients with Kaposi sarcoma secondary to AIDS.14
Pyogenic granuloma presents as a solitary red-brown or bluish-black papule or nodule that bleeds easily when manipulated.15 It commonly occurs following trauma, typically on the fingers, feet, and lips.6 Although benign, potential complications include ulceration and blood loss. Pyogenic granulomas can be treated via curettage and cautery, excision, cryosurgery, or pulsed dye laser.15
- Wankhade V, Singh R, Sadhwani V, et al. Eccrine poroma. Indian Dermatol Online J. 2015;6:304-305.
- Yorulmaz A, Aksoy GG, Ozhamam EU. A growing mass under the nail: subungual eccrine poroma. Skin Appendage Disord. 2020;6:254-257.
- Wang Y, Liu M, Zheng Y, et al. Eccrine poroma presented as spindleshaped plaque: a case report. Medicine (Baltimore). 2021;100:E25971. doi:10.1097/MD.0000000000025971
- Sharma M, Singh M, Gupta K, et al. Eccrine poroma of the eyelid. Indian J Ophthalmol. 2020;68:2522.
- Rasool MN, Hawary MB. Benign eccrine poroma in the palm of the hand. Ann Saudi Med. 2004;24:46-47.
- Sawaya JL, Khachemoune A. Poroma: a review of eccrine, apocrine, and malignant forms [published online April 2, 2014]. Int J Dermatol. 2014;53:1053-1061. doi:10.1111/ijd.12448
- López-Sánchez C, Ferguson P, Collgros H. Basal cell carcinoma of the palm: an unusual presentation of a common tumour [published online August 6, 2019]. Australas J Dermatol. 2020;61:69-70. doi:10.1111/ajd.13129
- Berk DR, Böer A, Bauschard FD, et al. Circumscribed acral hypokeratosis [published online April 6, 2007]. J Am Acad Dermatol. 2007;57:292-296. doi:10.1016/j.jaad.2007.02.022
- Majluf-Cáceres P, Vera-Kellet C, González-Bombardiere S. New dermoscopic keys for circumscribed acral hypokeratosis: report of four cases. Dermatol Pract Concept. 2021;11:E2021010. doi:10.5826/dpc.1102a10
- Simonart T, De Dobbeleer G, Stallenberg B. Classic Kaposi’s sarcoma of the palm in a metallurgist: role of iron filings in its development? Br J Dermatol. 2003;148:1061-1063. doi:10.1046/j.1365-2133.2003.05331.x
- Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294. doi:10.5858/arpa.2012-0101-RS
- Al Zolibani AA, Al Robaee AA. Primary palmoplantar Kaposi’s sarcoma: an unusual presentation. Skinmed. 2006;5:248-249. doi:10.1111/j.1540-9740.2006.04662.x
- Cesarman E, Damania B, Krown SE, et al. Kaposi sarcoma. Nat Rev Dis Primers. 2019;5:9. doi:10.1038/s41572-019-0060-9
- Etemad SA, Dewan AK. Kaposi sarcoma updates [published online July 10, 2019]. Dermatol Clin. 2019;37:505-517. doi:10.1016/j. det.2019.05.008
- Murthy SC, Nagaraj A. Pyogenic granuloma. Indian Pediatr. 2012;49:855. doi:10.1007/s13312-012-0184-4
- Wankhade V, Singh R, Sadhwani V, et al. Eccrine poroma. Indian Dermatol Online J. 2015;6:304-305.
- Yorulmaz A, Aksoy GG, Ozhamam EU. A growing mass under the nail: subungual eccrine poroma. Skin Appendage Disord. 2020;6:254-257.
- Wang Y, Liu M, Zheng Y, et al. Eccrine poroma presented as spindleshaped plaque: a case report. Medicine (Baltimore). 2021;100:E25971. doi:10.1097/MD.0000000000025971
- Sharma M, Singh M, Gupta K, et al. Eccrine poroma of the eyelid. Indian J Ophthalmol. 2020;68:2522.
- Rasool MN, Hawary MB. Benign eccrine poroma in the palm of the hand. Ann Saudi Med. 2004;24:46-47.
- Sawaya JL, Khachemoune A. Poroma: a review of eccrine, apocrine, and malignant forms [published online April 2, 2014]. Int J Dermatol. 2014;53:1053-1061. doi:10.1111/ijd.12448
- López-Sánchez C, Ferguson P, Collgros H. Basal cell carcinoma of the palm: an unusual presentation of a common tumour [published online August 6, 2019]. Australas J Dermatol. 2020;61:69-70. doi:10.1111/ajd.13129
- Berk DR, Böer A, Bauschard FD, et al. Circumscribed acral hypokeratosis [published online April 6, 2007]. J Am Acad Dermatol. 2007;57:292-296. doi:10.1016/j.jaad.2007.02.022
- Majluf-Cáceres P, Vera-Kellet C, González-Bombardiere S. New dermoscopic keys for circumscribed acral hypokeratosis: report of four cases. Dermatol Pract Concept. 2021;11:E2021010. doi:10.5826/dpc.1102a10
- Simonart T, De Dobbeleer G, Stallenberg B. Classic Kaposi’s sarcoma of the palm in a metallurgist: role of iron filings in its development? Br J Dermatol. 2003;148:1061-1063. doi:10.1046/j.1365-2133.2003.05331.x
- Radu O, Pantanowitz L. Kaposi sarcoma. Arch Pathol Lab Med. 2013;137:289-294. doi:10.5858/arpa.2012-0101-RS
- Al Zolibani AA, Al Robaee AA. Primary palmoplantar Kaposi’s sarcoma: an unusual presentation. Skinmed. 2006;5:248-249. doi:10.1111/j.1540-9740.2006.04662.x
- Cesarman E, Damania B, Krown SE, et al. Kaposi sarcoma. Nat Rev Dis Primers. 2019;5:9. doi:10.1038/s41572-019-0060-9
- Etemad SA, Dewan AK. Kaposi sarcoma updates [published online July 10, 2019]. Dermatol Clin. 2019;37:505-517. doi:10.1016/j. det.2019.05.008
- Murthy SC, Nagaraj A. Pyogenic granuloma. Indian Pediatr. 2012;49:855. doi:10.1007/s13312-012-0184-4
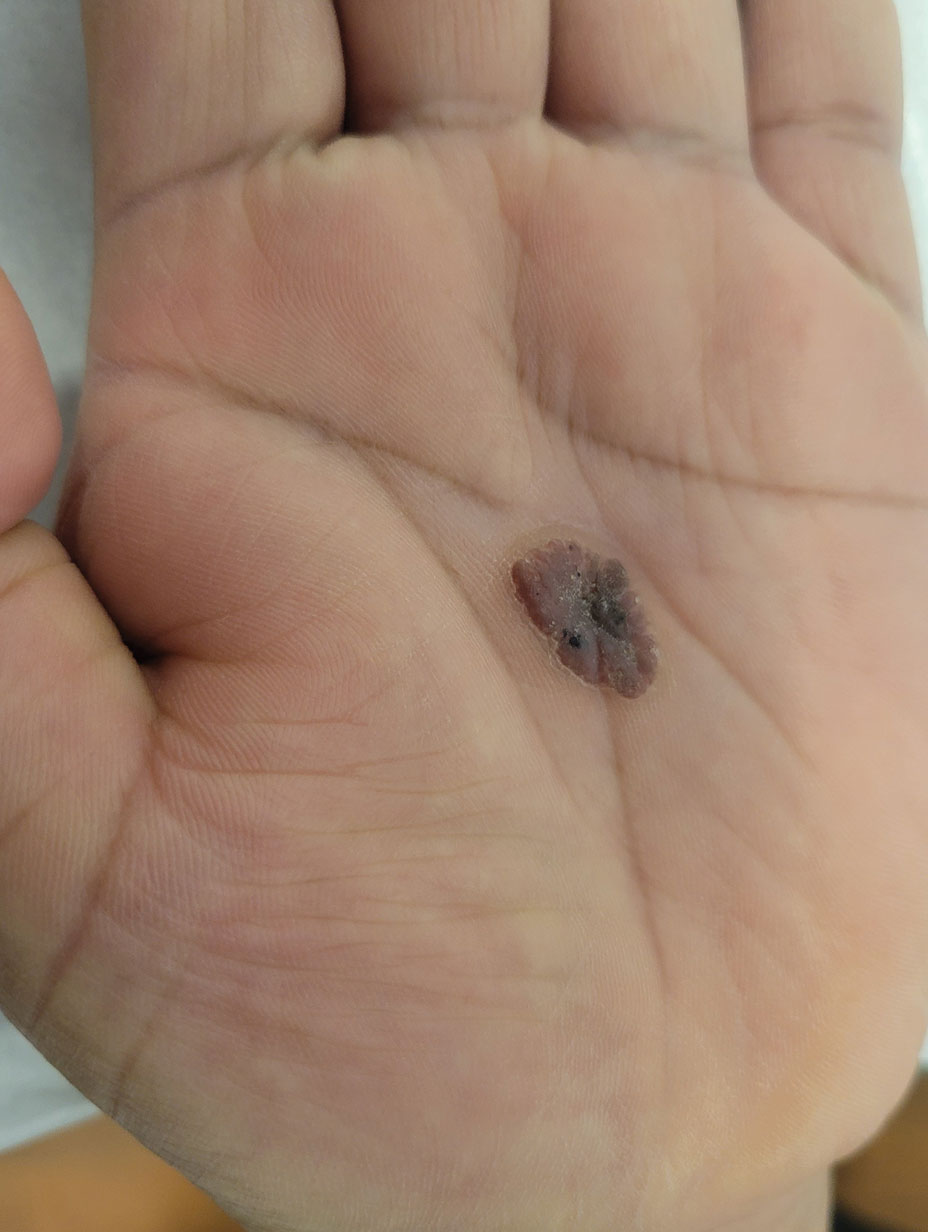
A 43-year-old woman presented with a painful lesion on the palm of 30 years’ duration that had grown in size. Physical examination revealed an oval, brown, lobulated plaque with a hyperkeratotic rim on the left palm. She reported bleeding and pain. A shallow cup-shaped depression was noted within the plaque. A 4-mm punch biopsy was performed.
Long-term freedom from NMOSD relapse with satralizumab
DENVER – , new research shows.
“In long-term observations, we are seeing a nice, sustained suppression of relapses early, as well as late, in treatment,” said study investigator Anthony Traboulsee, MD, University of British Columbia, Vancouver, in presenting the findings at the annual meeting of the Consortium of Multiple Sclerosis Centers.
“It remains very tolerable with no participants discontinuing because of side effects,” he said. “And importantly, [there are] no signs of a delayed risk of infections for both monotherapy and combination therapy.”
Satralizumab, a monoclonal recycling antibody, targets the interleukin (IL)–6 receptor, which is elevated in the serum and cerebrospinal fluid of patients in NMOSD.
The drug was approved by the Food and Drug Administration in 2020 for the treatment of AQP4 antibody–positive NMOSD after favorable results from two key trials: SAkuraSky and SAkuraStar.
The FDA approval marked satralizumab as the third therapy for NMOSD, following eculizumab (Soliris) and inebilizumab (Uplizna).
Satralizumab is administered in subcutaneous injections every 4 weeks after a run-in period of injections at weeks 0, 2, and 4.
Longest trial to date
To evaluate the drug’s long-term efficacy in the treatment of AQP4 IgG–positive NMOSD, patients from the two previous phase 3 trials were entered into the single arm, open-label SAkuraMoon study and continued treatment with the satralizumab 120 mg injections once monthly, with or without immunosuppressive therapy.
The study included 106 patients (mean age 44 years, 89.6% women), all of whom had received one or more doses of satralizumab by the data cutoff of January 2022.
With a median duration of satralizumab exposure of 5 years, the overall adjusted annualized rate of investigator protocol-defined relapse (ARR) was 0.09.
Longitudinal assessment further showed no significant increase in the relapse rate over the course of the study, with an ARR rate of 0.16 at year 1; 0.10 at year 2; 0.05 at year 3; and 0.07 at year 4.
At week 240 (4.6 years), 72% of satralizumab-treated patients were relapse-free, with 91% free from severe relapse.
In addition, 85% of patients had no sustained disability, as measured by Expanded Disability Status Scale (EDSS) worsening, over the study period.
Asked if there are potential subgroups of patients who may be more susceptible to the worsening of disability, Dr. Traboulsee responded “not that we can tell as of yet.”
“I would like to explore this further as this is a relatively new observation, and, as far as I know, this is the longest follow-up for an NMO treatment trial cohort,” he said.
Favorable safety profile
The safety profile was also favorable, consistent with results in the earlier trials. The longer exposure to satralizumab was not associated with a higher risk of severe (grade 3 or higher) laboratory changes versus the double-blind studies. “Rates of adverse events and serious adverse events with overall satralizumab treatment were comparable with the double-blind periods,” said Dr. Traboulsee.
“With satralizumab combined with immunosuppressant therapy, we’re not seeing an increased rate of infections, because it’s not an immune suppressant – it doesn’t suppress lymphocytes or lower immunoglobulin,” he added.
While the use of combination therapy has been an important clinical concern, Dr. Traboulsee noted that “this does not appear to be the case with satralizumab when combined with daily prednisone or daily azathioprine.”
“There is no increased risk of infections, compared with placebo, and it interestingly appears lower than patients on prednisone or azathioprine alone,” he said.
While the median follow-up was 5 years, some in the clinical trial population have been on treatment for up to 7.9 years.
“Based on the current safety and efficacy data, they could stay on this therapy indefinitely, in my opinion,” Dr. Traboulsee said.
In addition to its long-term safety and efficacy, satralizumab “is easy for patients to take and does not require access to an infusion center. It’s easy for physicians to monitor safety, especially since no additional vaccinations or precautions are required beyond what is done in routine care.”
“What I conclude from that clinically is that this is a highly effective and safe therapy by itself or in combination with another agent,” Dr. Traboulsee said.
He noted that the lack of a bump in infections is “really encouraging and very important with a chronic disease that affects elderly patients. So far, so good,” he added.
‘A good first-line therapy’
Commenting on the study, Shailee Shah, MD, an assistant professor in the neuroimmunology division at Vanderbilt University Medical Center, Nashville, Tenn., agreed that the findings bode well for satralizumab’s long-term benefits.
“These are promising results and suggest that satralizumab is very effective in the long term, and even when patients relapse, those relapses are less severe than they would likely be if the patient were off therapy,” she said.
She noted that, while the ability to self-administer injections with satralizumab is convenient, preferences vary.
“This is patient dependent,” Dr. Shah said. “For some patients an injectable medication is ideal but for others an infusion medication [such as eculizumab] is preferred.”
Overall, however, Dr. Shah described satralizumab as “a good first-line therapy for patients with NMOSD in addition to eculizumab/ravulizumab and inebilizumab.”
“It is reasonable to consider this medication in isolation or with concomitant immunosuppressive therapy,” she said.
Dr. Traboulsee’s disclosures include relationships with Novartis, Roche, Sanofi (Genzyme), Ingo Kleiter, Alexion, Almirall, Bayer, Biogen, Celgene, Genentech, Hexal, Horizon, Merck, and Sanofi. Dr. Shah reports that she has served on advisory boards for Horizon, Alexion, and Genentech.
DENVER – , new research shows.
“In long-term observations, we are seeing a nice, sustained suppression of relapses early, as well as late, in treatment,” said study investigator Anthony Traboulsee, MD, University of British Columbia, Vancouver, in presenting the findings at the annual meeting of the Consortium of Multiple Sclerosis Centers.
“It remains very tolerable with no participants discontinuing because of side effects,” he said. “And importantly, [there are] no signs of a delayed risk of infections for both monotherapy and combination therapy.”
Satralizumab, a monoclonal recycling antibody, targets the interleukin (IL)–6 receptor, which is elevated in the serum and cerebrospinal fluid of patients in NMOSD.
The drug was approved by the Food and Drug Administration in 2020 for the treatment of AQP4 antibody–positive NMOSD after favorable results from two key trials: SAkuraSky and SAkuraStar.
The FDA approval marked satralizumab as the third therapy for NMOSD, following eculizumab (Soliris) and inebilizumab (Uplizna).
Satralizumab is administered in subcutaneous injections every 4 weeks after a run-in period of injections at weeks 0, 2, and 4.
Longest trial to date
To evaluate the drug’s long-term efficacy in the treatment of AQP4 IgG–positive NMOSD, patients from the two previous phase 3 trials were entered into the single arm, open-label SAkuraMoon study and continued treatment with the satralizumab 120 mg injections once monthly, with or without immunosuppressive therapy.
The study included 106 patients (mean age 44 years, 89.6% women), all of whom had received one or more doses of satralizumab by the data cutoff of January 2022.
With a median duration of satralizumab exposure of 5 years, the overall adjusted annualized rate of investigator protocol-defined relapse (ARR) was 0.09.
Longitudinal assessment further showed no significant increase in the relapse rate over the course of the study, with an ARR rate of 0.16 at year 1; 0.10 at year 2; 0.05 at year 3; and 0.07 at year 4.
At week 240 (4.6 years), 72% of satralizumab-treated patients were relapse-free, with 91% free from severe relapse.
In addition, 85% of patients had no sustained disability, as measured by Expanded Disability Status Scale (EDSS) worsening, over the study period.
Asked if there are potential subgroups of patients who may be more susceptible to the worsening of disability, Dr. Traboulsee responded “not that we can tell as of yet.”
“I would like to explore this further as this is a relatively new observation, and, as far as I know, this is the longest follow-up for an NMO treatment trial cohort,” he said.
Favorable safety profile
The safety profile was also favorable, consistent with results in the earlier trials. The longer exposure to satralizumab was not associated with a higher risk of severe (grade 3 or higher) laboratory changes versus the double-blind studies. “Rates of adverse events and serious adverse events with overall satralizumab treatment were comparable with the double-blind periods,” said Dr. Traboulsee.
“With satralizumab combined with immunosuppressant therapy, we’re not seeing an increased rate of infections, because it’s not an immune suppressant – it doesn’t suppress lymphocytes or lower immunoglobulin,” he added.
While the use of combination therapy has been an important clinical concern, Dr. Traboulsee noted that “this does not appear to be the case with satralizumab when combined with daily prednisone or daily azathioprine.”
“There is no increased risk of infections, compared with placebo, and it interestingly appears lower than patients on prednisone or azathioprine alone,” he said.
While the median follow-up was 5 years, some in the clinical trial population have been on treatment for up to 7.9 years.
“Based on the current safety and efficacy data, they could stay on this therapy indefinitely, in my opinion,” Dr. Traboulsee said.
In addition to its long-term safety and efficacy, satralizumab “is easy for patients to take and does not require access to an infusion center. It’s easy for physicians to monitor safety, especially since no additional vaccinations or precautions are required beyond what is done in routine care.”
“What I conclude from that clinically is that this is a highly effective and safe therapy by itself or in combination with another agent,” Dr. Traboulsee said.
He noted that the lack of a bump in infections is “really encouraging and very important with a chronic disease that affects elderly patients. So far, so good,” he added.
‘A good first-line therapy’
Commenting on the study, Shailee Shah, MD, an assistant professor in the neuroimmunology division at Vanderbilt University Medical Center, Nashville, Tenn., agreed that the findings bode well for satralizumab’s long-term benefits.
“These are promising results and suggest that satralizumab is very effective in the long term, and even when patients relapse, those relapses are less severe than they would likely be if the patient were off therapy,” she said.
She noted that, while the ability to self-administer injections with satralizumab is convenient, preferences vary.
“This is patient dependent,” Dr. Shah said. “For some patients an injectable medication is ideal but for others an infusion medication [such as eculizumab] is preferred.”
Overall, however, Dr. Shah described satralizumab as “a good first-line therapy for patients with NMOSD in addition to eculizumab/ravulizumab and inebilizumab.”
“It is reasonable to consider this medication in isolation or with concomitant immunosuppressive therapy,” she said.
Dr. Traboulsee’s disclosures include relationships with Novartis, Roche, Sanofi (Genzyme), Ingo Kleiter, Alexion, Almirall, Bayer, Biogen, Celgene, Genentech, Hexal, Horizon, Merck, and Sanofi. Dr. Shah reports that she has served on advisory boards for Horizon, Alexion, and Genentech.
DENVER – , new research shows.
“In long-term observations, we are seeing a nice, sustained suppression of relapses early, as well as late, in treatment,” said study investigator Anthony Traboulsee, MD, University of British Columbia, Vancouver, in presenting the findings at the annual meeting of the Consortium of Multiple Sclerosis Centers.
“It remains very tolerable with no participants discontinuing because of side effects,” he said. “And importantly, [there are] no signs of a delayed risk of infections for both monotherapy and combination therapy.”
Satralizumab, a monoclonal recycling antibody, targets the interleukin (IL)–6 receptor, which is elevated in the serum and cerebrospinal fluid of patients in NMOSD.
The drug was approved by the Food and Drug Administration in 2020 for the treatment of AQP4 antibody–positive NMOSD after favorable results from two key trials: SAkuraSky and SAkuraStar.
The FDA approval marked satralizumab as the third therapy for NMOSD, following eculizumab (Soliris) and inebilizumab (Uplizna).
Satralizumab is administered in subcutaneous injections every 4 weeks after a run-in period of injections at weeks 0, 2, and 4.
Longest trial to date
To evaluate the drug’s long-term efficacy in the treatment of AQP4 IgG–positive NMOSD, patients from the two previous phase 3 trials were entered into the single arm, open-label SAkuraMoon study and continued treatment with the satralizumab 120 mg injections once monthly, with or without immunosuppressive therapy.
The study included 106 patients (mean age 44 years, 89.6% women), all of whom had received one or more doses of satralizumab by the data cutoff of January 2022.
With a median duration of satralizumab exposure of 5 years, the overall adjusted annualized rate of investigator protocol-defined relapse (ARR) was 0.09.
Longitudinal assessment further showed no significant increase in the relapse rate over the course of the study, with an ARR rate of 0.16 at year 1; 0.10 at year 2; 0.05 at year 3; and 0.07 at year 4.
At week 240 (4.6 years), 72% of satralizumab-treated patients were relapse-free, with 91% free from severe relapse.
In addition, 85% of patients had no sustained disability, as measured by Expanded Disability Status Scale (EDSS) worsening, over the study period.
Asked if there are potential subgroups of patients who may be more susceptible to the worsening of disability, Dr. Traboulsee responded “not that we can tell as of yet.”
“I would like to explore this further as this is a relatively new observation, and, as far as I know, this is the longest follow-up for an NMO treatment trial cohort,” he said.
Favorable safety profile
The safety profile was also favorable, consistent with results in the earlier trials. The longer exposure to satralizumab was not associated with a higher risk of severe (grade 3 or higher) laboratory changes versus the double-blind studies. “Rates of adverse events and serious adverse events with overall satralizumab treatment were comparable with the double-blind periods,” said Dr. Traboulsee.
“With satralizumab combined with immunosuppressant therapy, we’re not seeing an increased rate of infections, because it’s not an immune suppressant – it doesn’t suppress lymphocytes or lower immunoglobulin,” he added.
While the use of combination therapy has been an important clinical concern, Dr. Traboulsee noted that “this does not appear to be the case with satralizumab when combined with daily prednisone or daily azathioprine.”
“There is no increased risk of infections, compared with placebo, and it interestingly appears lower than patients on prednisone or azathioprine alone,” he said.
While the median follow-up was 5 years, some in the clinical trial population have been on treatment for up to 7.9 years.
“Based on the current safety and efficacy data, they could stay on this therapy indefinitely, in my opinion,” Dr. Traboulsee said.
In addition to its long-term safety and efficacy, satralizumab “is easy for patients to take and does not require access to an infusion center. It’s easy for physicians to monitor safety, especially since no additional vaccinations or precautions are required beyond what is done in routine care.”
“What I conclude from that clinically is that this is a highly effective and safe therapy by itself or in combination with another agent,” Dr. Traboulsee said.
He noted that the lack of a bump in infections is “really encouraging and very important with a chronic disease that affects elderly patients. So far, so good,” he added.
‘A good first-line therapy’
Commenting on the study, Shailee Shah, MD, an assistant professor in the neuroimmunology division at Vanderbilt University Medical Center, Nashville, Tenn., agreed that the findings bode well for satralizumab’s long-term benefits.
“These are promising results and suggest that satralizumab is very effective in the long term, and even when patients relapse, those relapses are less severe than they would likely be if the patient were off therapy,” she said.
She noted that, while the ability to self-administer injections with satralizumab is convenient, preferences vary.
“This is patient dependent,” Dr. Shah said. “For some patients an injectable medication is ideal but for others an infusion medication [such as eculizumab] is preferred.”
Overall, however, Dr. Shah described satralizumab as “a good first-line therapy for patients with NMOSD in addition to eculizumab/ravulizumab and inebilizumab.”
“It is reasonable to consider this medication in isolation or with concomitant immunosuppressive therapy,” she said.
Dr. Traboulsee’s disclosures include relationships with Novartis, Roche, Sanofi (Genzyme), Ingo Kleiter, Alexion, Almirall, Bayer, Biogen, Celgene, Genentech, Hexal, Horizon, Merck, and Sanofi. Dr. Shah reports that she has served on advisory boards for Horizon, Alexion, and Genentech.
AT CMSC 2023
MDs with chronic illness live in a different medical world
Linda Bluestein remembers all the doctors who missed, ignored, or incompletely diagnosed her chronic illness.
There was the orthopedic surgeon who noted her hyperextended elbows but failed to check any of her other joints. The gastroenterologist who insisted on performing multiple scoping procedures but wouldn’t discuss how to manage her symptoms. The other surgeon who, after performing arthroscopy on her injured knee, yelled at her: “There is nothing wrong with your knee! You’re fine!” in a room full of people.
And then there was the rheumatologist who said: “Oh, you want something to be wrong with you?”
“No,” she replied, “I want an explanation. I want to keep working. I just want to know why these things keep happening to me.”
The medical frustration she experienced was especially difficult because, like her health care providers, Linda Bluestein has an MD after her name. She is a board-certified anesthesiologist and integrative medicine physician.
Along with the physically demanding schedule of medical practice, they must cope with what many call a “culture of invincibility” within medicine. Doctors are not supposed to get sick. In fact, the unwritten rule is presenteeism – to function without adequate food or sleep and to never prioritize their own self-care over their dedication to their patients.
Whether their conditions are visible, such as muscular dystrophy and multiple sclerosis, or invisible, such as fibromyalgia and mental illnesses – and now, long COVID – these doctors often meet significant stigma. They fight the assumption that they are less capable than their colleagues.
But they also experience an invaluable benefit: They gain firsthand knowledge of the patient experience, a profound understanding which, they say, enhances how they care for their own patients.
What it takes to become a doctor when you have a chronic condition
In short, it’s not easy.
Data from the 2018 National Health Interview Survey show that more than half of U.S. adults had at least one of several chronic conditions, including rheumatoid arthritis, asthma, diabetes, hypertension, and kidney problems. Nearly a third of respondents had more than one condition. But fewer than 5% of medical students and 3% of practicing physicians report having a chronic illness or disability, according to studies from 2019 and 2021.
While that could mean that fewer people with chronic illness enter medicine, cases also exist in which aspiring physicians with conditions were dissuaded from pursuing a career in medicine at all.
Amy Stenehjem, MD, a physical medicine and rehabilitation physician, is one of the exceptions. Diagnosed with several autoimmune-related conditions as a teenager and young adult, Dr. Stenehjem was determined to become a doctor. In her 20s, her health was relatively stable, and she was able to manage medical school and residency. Her training institutions agreed to provide some accommodations that helped her succeed.
“They let me build some flexibility into the training,” Dr. Stenehjem said. “In medical school, when I knew I wasn’t going to be able to do a particular specialty as a career, they let me work with an attending doctor that did not require a lot of on-call time during that particular rotation.”
Dr. Stenehjem specialized in chronic neck and back disorders, fibromyalgia, chronic fatigue syndrome (myalgic encephalomyelitis), and autoimmune-related diseases. She practiced for more than a decade. But in 2011, her condition spiraled. She couldn’t walk a few steps or even sit upright without experiencing dizziness and shortness of breath. She had debilitating fatigue and episodes of fever, rash, headaches, and joint pain.
It would take 7 years and more than 20 doctors to determine Dr. Stenehjem’s multiple diagnoses. In addition to her autoimmune diseases, she was diagnosed with postural orthostatic tachycardia syndrome, autoinflammatory periodic fever syndrome, Lyme disease, and reactivated Epstein-Barr infection.
While she suspects that her providers gave her more “leeway” because she was a physician, many did not show a deep understanding of the severity of her symptoms and the impact those symptoms had.
“When I was practicing, I really didn’t fully understand the impact chronic illness had on my patients,” Dr. Stenehjem said. “Things like chronic dizziness, headaches, fatigue, pain, or brain fog can be really hard to understand unless you’ve experienced these symptoms. When I got sick, I finally realized, ‘Oh my goodness, when a patient says they’re dealing with fatigue, this is not your normal, I’m-super-tired-from-being-on-call fatigue. This is I-can’t-get-out-of-bed fatigue.’ That’s what people with chronic illness often deal with on a daily basis.”
Treating the individual
Dr. Stenehjem was aware that her chronic illness would affect her medical career. For Jason Baker, MD, an endocrinologist at Weill Cornell Medicine, New York, it came as a shock. Dr. Baker was a third-year medical student when he experienced increased urination and rapid weight loss. It was only when friends pressed him to visit student health that a blood test revealed type 1 diabetes. Dr. Baker suddenly found himself lying in a hospital bed.
He remembers an attending physician who simply handed him a textbook on diabetic ketoacidosis (DKA) and a resident who informed him that he had kidney damage, which turned out to be untrue. Neither discussed the psychological issues from a frightening diagnosis that would require lifelong, daily management.
“There certainly could have been a bit more empathy from some of the people I dealt with early on,” he said.
Although his training gave him a stark picture of worst-case scenarios, Dr. Baker found that knowledge motivating. “I’d already seen patients come in who had diabetes complications,” Dr. Baker says. “I vowed to never ever get those complications. It was a good balance of fear and motivation.”
Dr. Baker had not planned to specialize in endocrinology, but he quickly realized that his personal diagnosis could help others. Now he often shares his experience with his patients who have diabetes, which he says makes them more comfortable discussing their own problems.
His approach, Dr. Baker explained, is to treat everyone as an individual. Trying to neatly classify patients with chronic illness is a common mistake he notices among physicians.
“There’s a lot of misunderstanding about type 1 versus type 2 [diabetes],” Dr. Baker said, “and trying to categorize people when sometimes people can’t be categorized. That’s really with any chronic condition; there’s no one size fits all.”
Managing his health is still a time-consuming task. At work, he needs breaks to eat, check his blood sugar, or take insulin. “During the workday seeing patients, I have to also remember that I’m a patient,” Dr. Baker said. “I have to be okay with prioritizing my own health. Otherwise I can’t help anybody.”
‘I am not the doctor for you’
Chronic diseases such as diabetes or hypertension are familiar to most doctors, and with good management, patients can usually function normally. When chronic conditions become disabling, however, attitudes in the medical field can change.
According to data from the Centers for Disease Control and Prevention and from studies, people with disabilities experience significant disparities and barriers to care. Some of this can be linked to social determinants of health. People with disabilities are more likely to be poor and to rely on Medicare and Medicaid for insurance coverage. But lack of training, unwillingness to provide accommodations, ignorance of legal requirements, and inaccurate assumptions among physicians also play a role.
These are themes that Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School, Boston, has heard from hundreds of patients with disabilities during the more than 25 years that she has conducted research.
In late 2019, Dr. Iezzoni and coinvestigators fielded a national survey of 714 practicing physicians. Only 40.7% reported they were “very confident” that they could provide the same quality of care to patients with disabilities as they do for other patients. And only 56.5% “strongly agreed” that they welcomed these patients into their practices.
The survey was conducted through a series of small focus groups that Dr. Iezzoni held with physicians in 2018. These yielded views that were startling, and in some cases, overtly discriminatory:
- Doctors complained about the “burden” of caring for a patient with a disability.
- They lacked the time or equipment, such as accessible exam tables or weight scales.
- They admitted to inventing excuses for why appointments were not available or routine diagnostic tests were not performed.
- They described being fearful of lawsuits under the Americans with Disabilities Act.
The overall message was summed up in one doctor’s statement: “I am not the doctor for you.”
“Doctors are people too,” Dr. Iezzoni pointed out. “And so they reflect the same prejudices and stigmatized attitudes of the rest of the population. It might be implicit, so they might not be aware of it. [But] it might be explicit.”
Ableism in the medical field is all too familiar to Dr. Iezzoni. She was diagnosed with multiple sclerosis at age 26 during her first year at Harvard Medical School in the early 1980s. Despite symptom flare-ups, Dr. Iezzoni was able to graduate with her class, but many instructors and administrators had little interest in accommodating her physical limitations. In fact, several physicians discouraged her from continuing to train.
Unable to take call, run up flights of stairs, or stand for hours at a time, Dr. Iezzoni remembers being told by a senior surgeon that she shouldn’t become a doctor since she lacked the “most important quality,” which was “24/7 availability.” A hospital CEO informed her: “There are too many doctors in the country right now for us to worry about training a handicapped physician. If that means that some people get left by the wayside, so be it.”
Ultimately, Harvard Medical School declined to write Dr. Iezzoni a letter of recommendation for an internship. She would never practice medicine. “My career was just truncated from the start,” Dr. Iezzoni said. “It never happened because of discrimination.”
She later learned the legal term for her treatment: constructive dismissal.
“The medical school didn’t outright say, ‘Go away. We’re not allowing you to graduate.’ ” Dr. Iezzoni explained. “But they made my life so difficult that I did so voluntarily.”
Dr. Iezzoni graduated in 1984, before the passage of the ADA in 1990, and she refers to her experience as a “ghost from the past,” a historical reminder of how the legal landscape has changed – even though the tendency toward bias may not have.
The fight for inclusion
Zainub Dhanani, a fifth-year medical student at Stanford (Calif.) University, won’t forget an interview at one of the other schools to which she applied. The interviewer asked how she expected to be in a hospital all day if she had a chronic illness.
“Does it really make sense?” he wanted to know.
The question shocked her in the moment, but now she sees this type of bias as linked to the inequalities that many marginalized groups face in health care and beyond. That’s also why she believes physician-patients are crucial to improve the quality of care for people with chronic illness and other groups that face discrimination.
Who else, she wonders, could provide that “reaffirming” experience for patients or have that “unique edge” other than a provider who has navigated the same world?
Ms. Dhanani is the executive director and founder of Medical Students With Disability and Chronic Illness, an organization dedicated to empowering these students through advocacy, education, accessibility, and community. The group now has 19 chapters at medical schools across the country.
Ms. Dhanani said she has received excellent accommodations from Stanford for her own condition (which she prefers not to disclose), but all medical schools are not as responsive to students with various physical needs. Her organization offers support and resources to inform these future physicians about their options and rights.
“Disability justice is also racial justice,” Ms. Dhanani stressed. “It’s also environmental justice. It’s also gender and sexuality-based justice. Those compounded layers of biases lead to worse and worse levels of care. As a patient, it’s terrifying. And as a future physician, it’s tragic to know that this is something so pervasive and yet so under-addressed in medicine.”
Soldiering on
Unfortunately, for some physicians with chronic illness, there are no practical accommodations that could save their careers in clinical practice.
Dr. Stenehjem now works part-time as a health consultant, helping those with chronic illnesses navigate their health care systems.
Dr. Bluestein offers a similar coaching service to patients with Ehlers-Danlos syndrome (EDS) and other connective tissue and hypermobility disorders. Because of her own EDS, she can no longer practice as an anesthesiologist but instead opened an integrative pain management practice for patients with complex pain conditions..
She believes the idea that doctors are “invincible” needs to change. She recalls the time her former group practice told her in no uncertain terms to “never call in sick.”
The stories she hears from her current clients are similar to her own. She can empathize, knowing firsthand the physical and psychological damage these attitudes can cause.
“When I was at my worst physically, I was also at my worst psychologically,” said Dr. Bluestein. “We tend to think of them as separate, but they go hand in hand. If we can validate people’s experiences rather than disregard them, it has a positive forward cycle, as opposed to the reverse, which is what usually happens.”
A version of this article first appeared on Medscape.com.
Linda Bluestein remembers all the doctors who missed, ignored, or incompletely diagnosed her chronic illness.
There was the orthopedic surgeon who noted her hyperextended elbows but failed to check any of her other joints. The gastroenterologist who insisted on performing multiple scoping procedures but wouldn’t discuss how to manage her symptoms. The other surgeon who, after performing arthroscopy on her injured knee, yelled at her: “There is nothing wrong with your knee! You’re fine!” in a room full of people.
And then there was the rheumatologist who said: “Oh, you want something to be wrong with you?”
“No,” she replied, “I want an explanation. I want to keep working. I just want to know why these things keep happening to me.”
The medical frustration she experienced was especially difficult because, like her health care providers, Linda Bluestein has an MD after her name. She is a board-certified anesthesiologist and integrative medicine physician.
Along with the physically demanding schedule of medical practice, they must cope with what many call a “culture of invincibility” within medicine. Doctors are not supposed to get sick. In fact, the unwritten rule is presenteeism – to function without adequate food or sleep and to never prioritize their own self-care over their dedication to their patients.
Whether their conditions are visible, such as muscular dystrophy and multiple sclerosis, or invisible, such as fibromyalgia and mental illnesses – and now, long COVID – these doctors often meet significant stigma. They fight the assumption that they are less capable than their colleagues.
But they also experience an invaluable benefit: They gain firsthand knowledge of the patient experience, a profound understanding which, they say, enhances how they care for their own patients.
What it takes to become a doctor when you have a chronic condition
In short, it’s not easy.
Data from the 2018 National Health Interview Survey show that more than half of U.S. adults had at least one of several chronic conditions, including rheumatoid arthritis, asthma, diabetes, hypertension, and kidney problems. Nearly a third of respondents had more than one condition. But fewer than 5% of medical students and 3% of practicing physicians report having a chronic illness or disability, according to studies from 2019 and 2021.
While that could mean that fewer people with chronic illness enter medicine, cases also exist in which aspiring physicians with conditions were dissuaded from pursuing a career in medicine at all.
Amy Stenehjem, MD, a physical medicine and rehabilitation physician, is one of the exceptions. Diagnosed with several autoimmune-related conditions as a teenager and young adult, Dr. Stenehjem was determined to become a doctor. In her 20s, her health was relatively stable, and she was able to manage medical school and residency. Her training institutions agreed to provide some accommodations that helped her succeed.
“They let me build some flexibility into the training,” Dr. Stenehjem said. “In medical school, when I knew I wasn’t going to be able to do a particular specialty as a career, they let me work with an attending doctor that did not require a lot of on-call time during that particular rotation.”
Dr. Stenehjem specialized in chronic neck and back disorders, fibromyalgia, chronic fatigue syndrome (myalgic encephalomyelitis), and autoimmune-related diseases. She practiced for more than a decade. But in 2011, her condition spiraled. She couldn’t walk a few steps or even sit upright without experiencing dizziness and shortness of breath. She had debilitating fatigue and episodes of fever, rash, headaches, and joint pain.
It would take 7 years and more than 20 doctors to determine Dr. Stenehjem’s multiple diagnoses. In addition to her autoimmune diseases, she was diagnosed with postural orthostatic tachycardia syndrome, autoinflammatory periodic fever syndrome, Lyme disease, and reactivated Epstein-Barr infection.
While she suspects that her providers gave her more “leeway” because she was a physician, many did not show a deep understanding of the severity of her symptoms and the impact those symptoms had.
“When I was practicing, I really didn’t fully understand the impact chronic illness had on my patients,” Dr. Stenehjem said. “Things like chronic dizziness, headaches, fatigue, pain, or brain fog can be really hard to understand unless you’ve experienced these symptoms. When I got sick, I finally realized, ‘Oh my goodness, when a patient says they’re dealing with fatigue, this is not your normal, I’m-super-tired-from-being-on-call fatigue. This is I-can’t-get-out-of-bed fatigue.’ That’s what people with chronic illness often deal with on a daily basis.”
Treating the individual
Dr. Stenehjem was aware that her chronic illness would affect her medical career. For Jason Baker, MD, an endocrinologist at Weill Cornell Medicine, New York, it came as a shock. Dr. Baker was a third-year medical student when he experienced increased urination and rapid weight loss. It was only when friends pressed him to visit student health that a blood test revealed type 1 diabetes. Dr. Baker suddenly found himself lying in a hospital bed.
He remembers an attending physician who simply handed him a textbook on diabetic ketoacidosis (DKA) and a resident who informed him that he had kidney damage, which turned out to be untrue. Neither discussed the psychological issues from a frightening diagnosis that would require lifelong, daily management.
“There certainly could have been a bit more empathy from some of the people I dealt with early on,” he said.
Although his training gave him a stark picture of worst-case scenarios, Dr. Baker found that knowledge motivating. “I’d already seen patients come in who had diabetes complications,” Dr. Baker says. “I vowed to never ever get those complications. It was a good balance of fear and motivation.”
Dr. Baker had not planned to specialize in endocrinology, but he quickly realized that his personal diagnosis could help others. Now he often shares his experience with his patients who have diabetes, which he says makes them more comfortable discussing their own problems.
His approach, Dr. Baker explained, is to treat everyone as an individual. Trying to neatly classify patients with chronic illness is a common mistake he notices among physicians.
“There’s a lot of misunderstanding about type 1 versus type 2 [diabetes],” Dr. Baker said, “and trying to categorize people when sometimes people can’t be categorized. That’s really with any chronic condition; there’s no one size fits all.”
Managing his health is still a time-consuming task. At work, he needs breaks to eat, check his blood sugar, or take insulin. “During the workday seeing patients, I have to also remember that I’m a patient,” Dr. Baker said. “I have to be okay with prioritizing my own health. Otherwise I can’t help anybody.”
‘I am not the doctor for you’
Chronic diseases such as diabetes or hypertension are familiar to most doctors, and with good management, patients can usually function normally. When chronic conditions become disabling, however, attitudes in the medical field can change.
According to data from the Centers for Disease Control and Prevention and from studies, people with disabilities experience significant disparities and barriers to care. Some of this can be linked to social determinants of health. People with disabilities are more likely to be poor and to rely on Medicare and Medicaid for insurance coverage. But lack of training, unwillingness to provide accommodations, ignorance of legal requirements, and inaccurate assumptions among physicians also play a role.
These are themes that Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School, Boston, has heard from hundreds of patients with disabilities during the more than 25 years that she has conducted research.
In late 2019, Dr. Iezzoni and coinvestigators fielded a national survey of 714 practicing physicians. Only 40.7% reported they were “very confident” that they could provide the same quality of care to patients with disabilities as they do for other patients. And only 56.5% “strongly agreed” that they welcomed these patients into their practices.
The survey was conducted through a series of small focus groups that Dr. Iezzoni held with physicians in 2018. These yielded views that were startling, and in some cases, overtly discriminatory:
- Doctors complained about the “burden” of caring for a patient with a disability.
- They lacked the time or equipment, such as accessible exam tables or weight scales.
- They admitted to inventing excuses for why appointments were not available or routine diagnostic tests were not performed.
- They described being fearful of lawsuits under the Americans with Disabilities Act.
The overall message was summed up in one doctor’s statement: “I am not the doctor for you.”
“Doctors are people too,” Dr. Iezzoni pointed out. “And so they reflect the same prejudices and stigmatized attitudes of the rest of the population. It might be implicit, so they might not be aware of it. [But] it might be explicit.”
Ableism in the medical field is all too familiar to Dr. Iezzoni. She was diagnosed with multiple sclerosis at age 26 during her first year at Harvard Medical School in the early 1980s. Despite symptom flare-ups, Dr. Iezzoni was able to graduate with her class, but many instructors and administrators had little interest in accommodating her physical limitations. In fact, several physicians discouraged her from continuing to train.
Unable to take call, run up flights of stairs, or stand for hours at a time, Dr. Iezzoni remembers being told by a senior surgeon that she shouldn’t become a doctor since she lacked the “most important quality,” which was “24/7 availability.” A hospital CEO informed her: “There are too many doctors in the country right now for us to worry about training a handicapped physician. If that means that some people get left by the wayside, so be it.”
Ultimately, Harvard Medical School declined to write Dr. Iezzoni a letter of recommendation for an internship. She would never practice medicine. “My career was just truncated from the start,” Dr. Iezzoni said. “It never happened because of discrimination.”
She later learned the legal term for her treatment: constructive dismissal.
“The medical school didn’t outright say, ‘Go away. We’re not allowing you to graduate.’ ” Dr. Iezzoni explained. “But they made my life so difficult that I did so voluntarily.”
Dr. Iezzoni graduated in 1984, before the passage of the ADA in 1990, and she refers to her experience as a “ghost from the past,” a historical reminder of how the legal landscape has changed – even though the tendency toward bias may not have.
The fight for inclusion
Zainub Dhanani, a fifth-year medical student at Stanford (Calif.) University, won’t forget an interview at one of the other schools to which she applied. The interviewer asked how she expected to be in a hospital all day if she had a chronic illness.
“Does it really make sense?” he wanted to know.
The question shocked her in the moment, but now she sees this type of bias as linked to the inequalities that many marginalized groups face in health care and beyond. That’s also why she believes physician-patients are crucial to improve the quality of care for people with chronic illness and other groups that face discrimination.
Who else, she wonders, could provide that “reaffirming” experience for patients or have that “unique edge” other than a provider who has navigated the same world?
Ms. Dhanani is the executive director and founder of Medical Students With Disability and Chronic Illness, an organization dedicated to empowering these students through advocacy, education, accessibility, and community. The group now has 19 chapters at medical schools across the country.
Ms. Dhanani said she has received excellent accommodations from Stanford for her own condition (which she prefers not to disclose), but all medical schools are not as responsive to students with various physical needs. Her organization offers support and resources to inform these future physicians about their options and rights.
“Disability justice is also racial justice,” Ms. Dhanani stressed. “It’s also environmental justice. It’s also gender and sexuality-based justice. Those compounded layers of biases lead to worse and worse levels of care. As a patient, it’s terrifying. And as a future physician, it’s tragic to know that this is something so pervasive and yet so under-addressed in medicine.”
Soldiering on
Unfortunately, for some physicians with chronic illness, there are no practical accommodations that could save their careers in clinical practice.
Dr. Stenehjem now works part-time as a health consultant, helping those with chronic illnesses navigate their health care systems.
Dr. Bluestein offers a similar coaching service to patients with Ehlers-Danlos syndrome (EDS) and other connective tissue and hypermobility disorders. Because of her own EDS, she can no longer practice as an anesthesiologist but instead opened an integrative pain management practice for patients with complex pain conditions..
She believes the idea that doctors are “invincible” needs to change. She recalls the time her former group practice told her in no uncertain terms to “never call in sick.”
The stories she hears from her current clients are similar to her own. She can empathize, knowing firsthand the physical and psychological damage these attitudes can cause.
“When I was at my worst physically, I was also at my worst psychologically,” said Dr. Bluestein. “We tend to think of them as separate, but they go hand in hand. If we can validate people’s experiences rather than disregard them, it has a positive forward cycle, as opposed to the reverse, which is what usually happens.”
A version of this article first appeared on Medscape.com.
Linda Bluestein remembers all the doctors who missed, ignored, or incompletely diagnosed her chronic illness.
There was the orthopedic surgeon who noted her hyperextended elbows but failed to check any of her other joints. The gastroenterologist who insisted on performing multiple scoping procedures but wouldn’t discuss how to manage her symptoms. The other surgeon who, after performing arthroscopy on her injured knee, yelled at her: “There is nothing wrong with your knee! You’re fine!” in a room full of people.
And then there was the rheumatologist who said: “Oh, you want something to be wrong with you?”
“No,” she replied, “I want an explanation. I want to keep working. I just want to know why these things keep happening to me.”
The medical frustration she experienced was especially difficult because, like her health care providers, Linda Bluestein has an MD after her name. She is a board-certified anesthesiologist and integrative medicine physician.
Along with the physically demanding schedule of medical practice, they must cope with what many call a “culture of invincibility” within medicine. Doctors are not supposed to get sick. In fact, the unwritten rule is presenteeism – to function without adequate food or sleep and to never prioritize their own self-care over their dedication to their patients.
Whether their conditions are visible, such as muscular dystrophy and multiple sclerosis, or invisible, such as fibromyalgia and mental illnesses – and now, long COVID – these doctors often meet significant stigma. They fight the assumption that they are less capable than their colleagues.
But they also experience an invaluable benefit: They gain firsthand knowledge of the patient experience, a profound understanding which, they say, enhances how they care for their own patients.
What it takes to become a doctor when you have a chronic condition
In short, it’s not easy.
Data from the 2018 National Health Interview Survey show that more than half of U.S. adults had at least one of several chronic conditions, including rheumatoid arthritis, asthma, diabetes, hypertension, and kidney problems. Nearly a third of respondents had more than one condition. But fewer than 5% of medical students and 3% of practicing physicians report having a chronic illness or disability, according to studies from 2019 and 2021.
While that could mean that fewer people with chronic illness enter medicine, cases also exist in which aspiring physicians with conditions were dissuaded from pursuing a career in medicine at all.
Amy Stenehjem, MD, a physical medicine and rehabilitation physician, is one of the exceptions. Diagnosed with several autoimmune-related conditions as a teenager and young adult, Dr. Stenehjem was determined to become a doctor. In her 20s, her health was relatively stable, and she was able to manage medical school and residency. Her training institutions agreed to provide some accommodations that helped her succeed.
“They let me build some flexibility into the training,” Dr. Stenehjem said. “In medical school, when I knew I wasn’t going to be able to do a particular specialty as a career, they let me work with an attending doctor that did not require a lot of on-call time during that particular rotation.”
Dr. Stenehjem specialized in chronic neck and back disorders, fibromyalgia, chronic fatigue syndrome (myalgic encephalomyelitis), and autoimmune-related diseases. She practiced for more than a decade. But in 2011, her condition spiraled. She couldn’t walk a few steps or even sit upright without experiencing dizziness and shortness of breath. She had debilitating fatigue and episodes of fever, rash, headaches, and joint pain.
It would take 7 years and more than 20 doctors to determine Dr. Stenehjem’s multiple diagnoses. In addition to her autoimmune diseases, she was diagnosed with postural orthostatic tachycardia syndrome, autoinflammatory periodic fever syndrome, Lyme disease, and reactivated Epstein-Barr infection.
While she suspects that her providers gave her more “leeway” because she was a physician, many did not show a deep understanding of the severity of her symptoms and the impact those symptoms had.
“When I was practicing, I really didn’t fully understand the impact chronic illness had on my patients,” Dr. Stenehjem said. “Things like chronic dizziness, headaches, fatigue, pain, or brain fog can be really hard to understand unless you’ve experienced these symptoms. When I got sick, I finally realized, ‘Oh my goodness, when a patient says they’re dealing with fatigue, this is not your normal, I’m-super-tired-from-being-on-call fatigue. This is I-can’t-get-out-of-bed fatigue.’ That’s what people with chronic illness often deal with on a daily basis.”
Treating the individual
Dr. Stenehjem was aware that her chronic illness would affect her medical career. For Jason Baker, MD, an endocrinologist at Weill Cornell Medicine, New York, it came as a shock. Dr. Baker was a third-year medical student when he experienced increased urination and rapid weight loss. It was only when friends pressed him to visit student health that a blood test revealed type 1 diabetes. Dr. Baker suddenly found himself lying in a hospital bed.
He remembers an attending physician who simply handed him a textbook on diabetic ketoacidosis (DKA) and a resident who informed him that he had kidney damage, which turned out to be untrue. Neither discussed the psychological issues from a frightening diagnosis that would require lifelong, daily management.
“There certainly could have been a bit more empathy from some of the people I dealt with early on,” he said.
Although his training gave him a stark picture of worst-case scenarios, Dr. Baker found that knowledge motivating. “I’d already seen patients come in who had diabetes complications,” Dr. Baker says. “I vowed to never ever get those complications. It was a good balance of fear and motivation.”
Dr. Baker had not planned to specialize in endocrinology, but he quickly realized that his personal diagnosis could help others. Now he often shares his experience with his patients who have diabetes, which he says makes them more comfortable discussing their own problems.
His approach, Dr. Baker explained, is to treat everyone as an individual. Trying to neatly classify patients with chronic illness is a common mistake he notices among physicians.
“There’s a lot of misunderstanding about type 1 versus type 2 [diabetes],” Dr. Baker said, “and trying to categorize people when sometimes people can’t be categorized. That’s really with any chronic condition; there’s no one size fits all.”
Managing his health is still a time-consuming task. At work, he needs breaks to eat, check his blood sugar, or take insulin. “During the workday seeing patients, I have to also remember that I’m a patient,” Dr. Baker said. “I have to be okay with prioritizing my own health. Otherwise I can’t help anybody.”
‘I am not the doctor for you’
Chronic diseases such as diabetes or hypertension are familiar to most doctors, and with good management, patients can usually function normally. When chronic conditions become disabling, however, attitudes in the medical field can change.
According to data from the Centers for Disease Control and Prevention and from studies, people with disabilities experience significant disparities and barriers to care. Some of this can be linked to social determinants of health. People with disabilities are more likely to be poor and to rely on Medicare and Medicaid for insurance coverage. But lack of training, unwillingness to provide accommodations, ignorance of legal requirements, and inaccurate assumptions among physicians also play a role.
These are themes that Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School, Boston, has heard from hundreds of patients with disabilities during the more than 25 years that she has conducted research.
In late 2019, Dr. Iezzoni and coinvestigators fielded a national survey of 714 practicing physicians. Only 40.7% reported they were “very confident” that they could provide the same quality of care to patients with disabilities as they do for other patients. And only 56.5% “strongly agreed” that they welcomed these patients into their practices.
The survey was conducted through a series of small focus groups that Dr. Iezzoni held with physicians in 2018. These yielded views that were startling, and in some cases, overtly discriminatory:
- Doctors complained about the “burden” of caring for a patient with a disability.
- They lacked the time or equipment, such as accessible exam tables or weight scales.
- They admitted to inventing excuses for why appointments were not available or routine diagnostic tests were not performed.
- They described being fearful of lawsuits under the Americans with Disabilities Act.
The overall message was summed up in one doctor’s statement: “I am not the doctor for you.”
“Doctors are people too,” Dr. Iezzoni pointed out. “And so they reflect the same prejudices and stigmatized attitudes of the rest of the population. It might be implicit, so they might not be aware of it. [But] it might be explicit.”
Ableism in the medical field is all too familiar to Dr. Iezzoni. She was diagnosed with multiple sclerosis at age 26 during her first year at Harvard Medical School in the early 1980s. Despite symptom flare-ups, Dr. Iezzoni was able to graduate with her class, but many instructors and administrators had little interest in accommodating her physical limitations. In fact, several physicians discouraged her from continuing to train.
Unable to take call, run up flights of stairs, or stand for hours at a time, Dr. Iezzoni remembers being told by a senior surgeon that she shouldn’t become a doctor since she lacked the “most important quality,” which was “24/7 availability.” A hospital CEO informed her: “There are too many doctors in the country right now for us to worry about training a handicapped physician. If that means that some people get left by the wayside, so be it.”
Ultimately, Harvard Medical School declined to write Dr. Iezzoni a letter of recommendation for an internship. She would never practice medicine. “My career was just truncated from the start,” Dr. Iezzoni said. “It never happened because of discrimination.”
She later learned the legal term for her treatment: constructive dismissal.
“The medical school didn’t outright say, ‘Go away. We’re not allowing you to graduate.’ ” Dr. Iezzoni explained. “But they made my life so difficult that I did so voluntarily.”
Dr. Iezzoni graduated in 1984, before the passage of the ADA in 1990, and she refers to her experience as a “ghost from the past,” a historical reminder of how the legal landscape has changed – even though the tendency toward bias may not have.
The fight for inclusion
Zainub Dhanani, a fifth-year medical student at Stanford (Calif.) University, won’t forget an interview at one of the other schools to which she applied. The interviewer asked how she expected to be in a hospital all day if she had a chronic illness.
“Does it really make sense?” he wanted to know.
The question shocked her in the moment, but now she sees this type of bias as linked to the inequalities that many marginalized groups face in health care and beyond. That’s also why she believes physician-patients are crucial to improve the quality of care for people with chronic illness and other groups that face discrimination.
Who else, she wonders, could provide that “reaffirming” experience for patients or have that “unique edge” other than a provider who has navigated the same world?
Ms. Dhanani is the executive director and founder of Medical Students With Disability and Chronic Illness, an organization dedicated to empowering these students through advocacy, education, accessibility, and community. The group now has 19 chapters at medical schools across the country.
Ms. Dhanani said she has received excellent accommodations from Stanford for her own condition (which she prefers not to disclose), but all medical schools are not as responsive to students with various physical needs. Her organization offers support and resources to inform these future physicians about their options and rights.
“Disability justice is also racial justice,” Ms. Dhanani stressed. “It’s also environmental justice. It’s also gender and sexuality-based justice. Those compounded layers of biases lead to worse and worse levels of care. As a patient, it’s terrifying. And as a future physician, it’s tragic to know that this is something so pervasive and yet so under-addressed in medicine.”
Soldiering on
Unfortunately, for some physicians with chronic illness, there are no practical accommodations that could save their careers in clinical practice.
Dr. Stenehjem now works part-time as a health consultant, helping those with chronic illnesses navigate their health care systems.
Dr. Bluestein offers a similar coaching service to patients with Ehlers-Danlos syndrome (EDS) and other connective tissue and hypermobility disorders. Because of her own EDS, she can no longer practice as an anesthesiologist but instead opened an integrative pain management practice for patients with complex pain conditions..
She believes the idea that doctors are “invincible” needs to change. She recalls the time her former group practice told her in no uncertain terms to “never call in sick.”
The stories she hears from her current clients are similar to her own. She can empathize, knowing firsthand the physical and psychological damage these attitudes can cause.
“When I was at my worst physically, I was also at my worst psychologically,” said Dr. Bluestein. “We tend to think of them as separate, but they go hand in hand. If we can validate people’s experiences rather than disregard them, it has a positive forward cycle, as opposed to the reverse, which is what usually happens.”
A version of this article first appeared on Medscape.com.
Community workers may address psychiatrist shortage
SAN FRANCISCO – promises to bring timely, evidence-based health services to those with little to no access to effective care.
The current shortage of mental health clinicians is driven by increased demand from a population more willing to seek psychiatric help and clinicians leaving the workforce. Both factors were exacerbated by the COVID-19 pandemic.
“It would be costly to address the problem through additional specialist training, and doing so would take decades to see any changes,” project director Milton L. Wainberg, MD, professor of clinical psychiatry at Columbia University, New York, and New York State Psychiatric Institute, said in an interview.
A better solution is to train members of the community to be the entry point to the mental health care system, a strategy that has been proven effective.
Details of the project were discussed at the annual meeting of the American Psychiatric Association.
Half of the United States population will be diagnosed with a mental or substance use disorder in their lifetime, but only about half of those will receive proper treatment. That percentage is even greater among lower-income groups and minorities, said Dr. Wainberg.
Despite the availability of multiple evidence-based therapies, there has been no reduction in the global prevalence of psychiatric illness since 1990 – the first time this burden was determined, he said.
Unfeasible model
“The historic paradigm of ongoing long-term care is costly and not a feasible public mental health model. There is no evidence that it works, and there is increasing demand for brief interventions,” said Dr. Wainberg.
The new initiative – called ENGAGE – has its origins in parts of Africa, where nurses had to be trained during the AIDS crisis as there weren’t enough doctors to roll out antiretroviral therapy.
In the United States, the program trains and certifies community workers who are passionate about their community. “Members of the community want to learn how to help their neighbors,” said Dr. Wainberg. “When we give them the opportunity to learn skills that can actually change community members’ symptoms, they are excited.”
The training involves a didactic component and an experiential component, in which trainees work with at least three cases under supervision to demonstrate competency. Technical assistance and other supports, such as refresher training, are offered for a year after training.
Workers ask three initial questions to quickly determine if a person has a mental health disorder. Asking 10 additional questions tells the worker if the person has a common mental health disorder like depression, anxiety, or posttraumatic stress disorder (PTSD), a substance use disorder involving alcohol or drugs, suicide risk, or a severe disorder requiring referral to a mental health specialist.
Those who do not require a referral are offered an intervention personalized to their need.
The training costs $5,000 per person. “We calculated for New York State it would cost only $18 million to train everybody we need,” said Dr. Wainberg.
Cost effective
He stressed the program, which is funded by the New York Office of Mental Health, is cost effective. Just like patients don’t need to see a plastic surgeon to have a small mole removed, they don’t always need to see a psychiatrist for run-of-the-mill mild depression, he said.
To date, 20 workers have been trained and have started to meet with clients in clinics in four New York City neighborhoods/boroughs (Harlem, Brooklyn, the Bronx, and Washington Heights). Additional clinics in West Harlem and Staten Island are expected to begin training soon.
Dr. Wainberg has been inundated with interest in the initiative. “Over the last 3 months I have been having 15 meetings a day” with parties interested in getting more information or wanting to know how to start such a program.
He plans to examine the program’s effectiveness in a number of areas, including patient symptoms, timeliness of services, access, sustainability, and cost. And he aims to expand the project beyond New York.
Mental health specialists shouldn’t worry about becoming irrelevant with the addition of community workers, as the demand is so great, said Dr. Wainberg. “There will always be a need for the kind of care mental health specialists are trained for. This initiative aims to expand capacity for those with less severe symptoms, who might not need an intensive level of intervention.”
Unique program
In a comment, Jonathan E. Alpert, MD, PhD, chair of the department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York, said the project is “unique” and an “excellent” idea.
“This is one of the first pilots that I know of in this country to train lay-members of the community to screen for mental illness and substance use disorders and even to provide evidence-based treatment for people who may have more mild symptoms and might not yet need to see a professional but otherwise would not have access to care.”
Dr. Alpert noted the current challenges of accessing care for a mental health or substance abuse disorder. “Many clinics have wait lists of 3-6 months.”
Another issue is the “stigma and lack of trust” among minority communities when it comes to formal mental health treatments. “Having lay-members who know the community, who look like the community, who understand the community, and who are available for screening and treatment is exceptionally important.”
Although this pilot program will have to be assessed for effectiveness, “the concept behind it is very important,” said Dr. Alpert. “If you’re relying on MDs and PhDs to provide mental health services, there just aren’t enough of us to go around.”
Dr. Wainberg and Dr. Alpert report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO – promises to bring timely, evidence-based health services to those with little to no access to effective care.
The current shortage of mental health clinicians is driven by increased demand from a population more willing to seek psychiatric help and clinicians leaving the workforce. Both factors were exacerbated by the COVID-19 pandemic.
“It would be costly to address the problem through additional specialist training, and doing so would take decades to see any changes,” project director Milton L. Wainberg, MD, professor of clinical psychiatry at Columbia University, New York, and New York State Psychiatric Institute, said in an interview.
A better solution is to train members of the community to be the entry point to the mental health care system, a strategy that has been proven effective.
Details of the project were discussed at the annual meeting of the American Psychiatric Association.
Half of the United States population will be diagnosed with a mental or substance use disorder in their lifetime, but only about half of those will receive proper treatment. That percentage is even greater among lower-income groups and minorities, said Dr. Wainberg.
Despite the availability of multiple evidence-based therapies, there has been no reduction in the global prevalence of psychiatric illness since 1990 – the first time this burden was determined, he said.
Unfeasible model
“The historic paradigm of ongoing long-term care is costly and not a feasible public mental health model. There is no evidence that it works, and there is increasing demand for brief interventions,” said Dr. Wainberg.
The new initiative – called ENGAGE – has its origins in parts of Africa, where nurses had to be trained during the AIDS crisis as there weren’t enough doctors to roll out antiretroviral therapy.
In the United States, the program trains and certifies community workers who are passionate about their community. “Members of the community want to learn how to help their neighbors,” said Dr. Wainberg. “When we give them the opportunity to learn skills that can actually change community members’ symptoms, they are excited.”
The training involves a didactic component and an experiential component, in which trainees work with at least three cases under supervision to demonstrate competency. Technical assistance and other supports, such as refresher training, are offered for a year after training.
Workers ask three initial questions to quickly determine if a person has a mental health disorder. Asking 10 additional questions tells the worker if the person has a common mental health disorder like depression, anxiety, or posttraumatic stress disorder (PTSD), a substance use disorder involving alcohol or drugs, suicide risk, or a severe disorder requiring referral to a mental health specialist.
Those who do not require a referral are offered an intervention personalized to their need.
The training costs $5,000 per person. “We calculated for New York State it would cost only $18 million to train everybody we need,” said Dr. Wainberg.
Cost effective
He stressed the program, which is funded by the New York Office of Mental Health, is cost effective. Just like patients don’t need to see a plastic surgeon to have a small mole removed, they don’t always need to see a psychiatrist for run-of-the-mill mild depression, he said.
To date, 20 workers have been trained and have started to meet with clients in clinics in four New York City neighborhoods/boroughs (Harlem, Brooklyn, the Bronx, and Washington Heights). Additional clinics in West Harlem and Staten Island are expected to begin training soon.
Dr. Wainberg has been inundated with interest in the initiative. “Over the last 3 months I have been having 15 meetings a day” with parties interested in getting more information or wanting to know how to start such a program.
He plans to examine the program’s effectiveness in a number of areas, including patient symptoms, timeliness of services, access, sustainability, and cost. And he aims to expand the project beyond New York.
Mental health specialists shouldn’t worry about becoming irrelevant with the addition of community workers, as the demand is so great, said Dr. Wainberg. “There will always be a need for the kind of care mental health specialists are trained for. This initiative aims to expand capacity for those with less severe symptoms, who might not need an intensive level of intervention.”
Unique program
In a comment, Jonathan E. Alpert, MD, PhD, chair of the department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York, said the project is “unique” and an “excellent” idea.
“This is one of the first pilots that I know of in this country to train lay-members of the community to screen for mental illness and substance use disorders and even to provide evidence-based treatment for people who may have more mild symptoms and might not yet need to see a professional but otherwise would not have access to care.”
Dr. Alpert noted the current challenges of accessing care for a mental health or substance abuse disorder. “Many clinics have wait lists of 3-6 months.”
Another issue is the “stigma and lack of trust” among minority communities when it comes to formal mental health treatments. “Having lay-members who know the community, who look like the community, who understand the community, and who are available for screening and treatment is exceptionally important.”
Although this pilot program will have to be assessed for effectiveness, “the concept behind it is very important,” said Dr. Alpert. “If you’re relying on MDs and PhDs to provide mental health services, there just aren’t enough of us to go around.”
Dr. Wainberg and Dr. Alpert report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO – promises to bring timely, evidence-based health services to those with little to no access to effective care.
The current shortage of mental health clinicians is driven by increased demand from a population more willing to seek psychiatric help and clinicians leaving the workforce. Both factors were exacerbated by the COVID-19 pandemic.
“It would be costly to address the problem through additional specialist training, and doing so would take decades to see any changes,” project director Milton L. Wainberg, MD, professor of clinical psychiatry at Columbia University, New York, and New York State Psychiatric Institute, said in an interview.
A better solution is to train members of the community to be the entry point to the mental health care system, a strategy that has been proven effective.
Details of the project were discussed at the annual meeting of the American Psychiatric Association.
Half of the United States population will be diagnosed with a mental or substance use disorder in their lifetime, but only about half of those will receive proper treatment. That percentage is even greater among lower-income groups and minorities, said Dr. Wainberg.
Despite the availability of multiple evidence-based therapies, there has been no reduction in the global prevalence of psychiatric illness since 1990 – the first time this burden was determined, he said.
Unfeasible model
“The historic paradigm of ongoing long-term care is costly and not a feasible public mental health model. There is no evidence that it works, and there is increasing demand for brief interventions,” said Dr. Wainberg.
The new initiative – called ENGAGE – has its origins in parts of Africa, where nurses had to be trained during the AIDS crisis as there weren’t enough doctors to roll out antiretroviral therapy.
In the United States, the program trains and certifies community workers who are passionate about their community. “Members of the community want to learn how to help their neighbors,” said Dr. Wainberg. “When we give them the opportunity to learn skills that can actually change community members’ symptoms, they are excited.”
The training involves a didactic component and an experiential component, in which trainees work with at least three cases under supervision to demonstrate competency. Technical assistance and other supports, such as refresher training, are offered for a year after training.
Workers ask three initial questions to quickly determine if a person has a mental health disorder. Asking 10 additional questions tells the worker if the person has a common mental health disorder like depression, anxiety, or posttraumatic stress disorder (PTSD), a substance use disorder involving alcohol or drugs, suicide risk, or a severe disorder requiring referral to a mental health specialist.
Those who do not require a referral are offered an intervention personalized to their need.
The training costs $5,000 per person. “We calculated for New York State it would cost only $18 million to train everybody we need,” said Dr. Wainberg.
Cost effective
He stressed the program, which is funded by the New York Office of Mental Health, is cost effective. Just like patients don’t need to see a plastic surgeon to have a small mole removed, they don’t always need to see a psychiatrist for run-of-the-mill mild depression, he said.
To date, 20 workers have been trained and have started to meet with clients in clinics in four New York City neighborhoods/boroughs (Harlem, Brooklyn, the Bronx, and Washington Heights). Additional clinics in West Harlem and Staten Island are expected to begin training soon.
Dr. Wainberg has been inundated with interest in the initiative. “Over the last 3 months I have been having 15 meetings a day” with parties interested in getting more information or wanting to know how to start such a program.
He plans to examine the program’s effectiveness in a number of areas, including patient symptoms, timeliness of services, access, sustainability, and cost. And he aims to expand the project beyond New York.
Mental health specialists shouldn’t worry about becoming irrelevant with the addition of community workers, as the demand is so great, said Dr. Wainberg. “There will always be a need for the kind of care mental health specialists are trained for. This initiative aims to expand capacity for those with less severe symptoms, who might not need an intensive level of intervention.”
Unique program
In a comment, Jonathan E. Alpert, MD, PhD, chair of the department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York, said the project is “unique” and an “excellent” idea.
“This is one of the first pilots that I know of in this country to train lay-members of the community to screen for mental illness and substance use disorders and even to provide evidence-based treatment for people who may have more mild symptoms and might not yet need to see a professional but otherwise would not have access to care.”
Dr. Alpert noted the current challenges of accessing care for a mental health or substance abuse disorder. “Many clinics have wait lists of 3-6 months.”
Another issue is the “stigma and lack of trust” among minority communities when it comes to formal mental health treatments. “Having lay-members who know the community, who look like the community, who understand the community, and who are available for screening and treatment is exceptionally important.”
Although this pilot program will have to be assessed for effectiveness, “the concept behind it is very important,” said Dr. Alpert. “If you’re relying on MDs and PhDs to provide mental health services, there just aren’t enough of us to go around.”
Dr. Wainberg and Dr. Alpert report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM APA 2023
U.S. psychiatrist shortage causing months-long wait times
SAN FRANCISCO –
“Long wait times for mental health care were a huge problem even before the pandemic but especially during the pandemic,” study investigator Erin McDaid, BS, Virginia Tech Carilion School of Medicine, Roanoke, said in an interview.
“It’s not like you have a cold or a virus and maybe you wait a little bit and it goes away. Mental health problems can completely impact your life; you can’t do anything, you can’t go to work, you can’t build relationships, you can’t take care of your kids. It’s a really big issue,” Ms. McDaid said.
The study was presented at the annual meeting of the American Psychiatric Association.
Few psychiatrists taking new patients
To find out just how big an issue wait times are, the researchers examined general psychiatry outpatient availability during the COVID-19 pandemic in five states – New York, California, North Dakota, Virginia, and Wyoming.
Altogether, 948 psychiatrists were sampled. Simulated adult patients made 864 calls seeking an initial psychiatric evaluation for general mental health care. The calls were made late in the pandemic, between May and July 2022.
Only 18.5% of psychiatrists were available to see new patients. The median wait time was 67 days for in-person appointments and 43 days for telepsychiatry appointments (P < .001).
More than half of psychiatrists who were contacted said they were not taking new patients, which was the most common reason given for unavailability.
“This is happening at the worst time, when we are seeing mental health issues spike,” Ms. McDaid said.
Telepsychiatry helpful but no panacea
The fact that wait times were a bit shorter for telepsychiatry is encouraging, Ms. McDaid said.
Telepsychiatry is a potential solution to provider shortages and geographic barriers, but it does not resolve the concerning shortage of psychiatric outpatient care, she noted.
“Psychiatrists adapted very well to telepsychiatry during COVID,” Saul Levin, MD, MPA, chief executive officer and medical director of the APA, noted during a preconference briefing with reporters.
“Before COVID, we always thought that the psychiatrist had to be with the patient in the room,” said Dr. Levin. But now we see that either “sitting inside the room with your psychiatrist or mental health specialist or [being there virtually] has the same effect. The patient is concentrating and working out their problems with you. I think that’s one of the positives – if anything coming out of COVID is positive.”
In an interview, Robert Trestman, MD, chair of the APA Council on Healthcare Systems and Financing, said telepsychiatry “will help, but there is not one simple solution that will fix the problem” regarding access to mental health care.
One promising approach is the collaborative care model, which enlists primary care physicians to provide mental health care in consultation with psychiatry and case management, Dr. Trestman said.
“There’s no question that there aren’t enough providers. There aren’t enough primary care doctors, and there certainly aren’t enough psychiatrists,” Dr. Trestman noted.
Encouragingly, however, the past few years have seen a steady increase in medical students choosing psychiatry.
“Psychiatry is now being thought of as a branch of neuroscience. We are understanding so much more about the field and about the brain. So that’s intriguing and intellectually challenging to many,” Dr. Trestman said.
He also noted that the pandemic has helped to “break down stigma. More people acknowledge and talk about mental health, and when an area is destigmatized, it’s so much easier for people to consider.”
Jack Resneck, Jr., MD, president of the American Medical Association, acknowledged that there is a “severe workforce shortage in health care right now.”
“I’m a physician and the president of the AMA, and it took me way too long to be able to find a primary care physician for myself,” he said.
“I also am a physician who refers patients to rheumatology and endocrinology, psychiatry, and other areas of medicine, and it is, in many geographic areas both rural and urban, a huge struggle right now,” said Dr. Resneck.
The study had no specific funding. Ms. McDaid, Dr. Levin, and Dr. Trestman have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO –
“Long wait times for mental health care were a huge problem even before the pandemic but especially during the pandemic,” study investigator Erin McDaid, BS, Virginia Tech Carilion School of Medicine, Roanoke, said in an interview.
“It’s not like you have a cold or a virus and maybe you wait a little bit and it goes away. Mental health problems can completely impact your life; you can’t do anything, you can’t go to work, you can’t build relationships, you can’t take care of your kids. It’s a really big issue,” Ms. McDaid said.
The study was presented at the annual meeting of the American Psychiatric Association.
Few psychiatrists taking new patients
To find out just how big an issue wait times are, the researchers examined general psychiatry outpatient availability during the COVID-19 pandemic in five states – New York, California, North Dakota, Virginia, and Wyoming.
Altogether, 948 psychiatrists were sampled. Simulated adult patients made 864 calls seeking an initial psychiatric evaluation for general mental health care. The calls were made late in the pandemic, between May and July 2022.
Only 18.5% of psychiatrists were available to see new patients. The median wait time was 67 days for in-person appointments and 43 days for telepsychiatry appointments (P < .001).
More than half of psychiatrists who were contacted said they were not taking new patients, which was the most common reason given for unavailability.
“This is happening at the worst time, when we are seeing mental health issues spike,” Ms. McDaid said.
Telepsychiatry helpful but no panacea
The fact that wait times were a bit shorter for telepsychiatry is encouraging, Ms. McDaid said.
Telepsychiatry is a potential solution to provider shortages and geographic barriers, but it does not resolve the concerning shortage of psychiatric outpatient care, she noted.
“Psychiatrists adapted very well to telepsychiatry during COVID,” Saul Levin, MD, MPA, chief executive officer and medical director of the APA, noted during a preconference briefing with reporters.
“Before COVID, we always thought that the psychiatrist had to be with the patient in the room,” said Dr. Levin. But now we see that either “sitting inside the room with your psychiatrist or mental health specialist or [being there virtually] has the same effect. The patient is concentrating and working out their problems with you. I think that’s one of the positives – if anything coming out of COVID is positive.”
In an interview, Robert Trestman, MD, chair of the APA Council on Healthcare Systems and Financing, said telepsychiatry “will help, but there is not one simple solution that will fix the problem” regarding access to mental health care.
One promising approach is the collaborative care model, which enlists primary care physicians to provide mental health care in consultation with psychiatry and case management, Dr. Trestman said.
“There’s no question that there aren’t enough providers. There aren’t enough primary care doctors, and there certainly aren’t enough psychiatrists,” Dr. Trestman noted.
Encouragingly, however, the past few years have seen a steady increase in medical students choosing psychiatry.
“Psychiatry is now being thought of as a branch of neuroscience. We are understanding so much more about the field and about the brain. So that’s intriguing and intellectually challenging to many,” Dr. Trestman said.
He also noted that the pandemic has helped to “break down stigma. More people acknowledge and talk about mental health, and when an area is destigmatized, it’s so much easier for people to consider.”
Jack Resneck, Jr., MD, president of the American Medical Association, acknowledged that there is a “severe workforce shortage in health care right now.”
“I’m a physician and the president of the AMA, and it took me way too long to be able to find a primary care physician for myself,” he said.
“I also am a physician who refers patients to rheumatology and endocrinology, psychiatry, and other areas of medicine, and it is, in many geographic areas both rural and urban, a huge struggle right now,” said Dr. Resneck.
The study had no specific funding. Ms. McDaid, Dr. Levin, and Dr. Trestman have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SAN FRANCISCO –
“Long wait times for mental health care were a huge problem even before the pandemic but especially during the pandemic,” study investigator Erin McDaid, BS, Virginia Tech Carilion School of Medicine, Roanoke, said in an interview.
“It’s not like you have a cold or a virus and maybe you wait a little bit and it goes away. Mental health problems can completely impact your life; you can’t do anything, you can’t go to work, you can’t build relationships, you can’t take care of your kids. It’s a really big issue,” Ms. McDaid said.
The study was presented at the annual meeting of the American Psychiatric Association.
Few psychiatrists taking new patients
To find out just how big an issue wait times are, the researchers examined general psychiatry outpatient availability during the COVID-19 pandemic in five states – New York, California, North Dakota, Virginia, and Wyoming.
Altogether, 948 psychiatrists were sampled. Simulated adult patients made 864 calls seeking an initial psychiatric evaluation for general mental health care. The calls were made late in the pandemic, between May and July 2022.
Only 18.5% of psychiatrists were available to see new patients. The median wait time was 67 days for in-person appointments and 43 days for telepsychiatry appointments (P < .001).
More than half of psychiatrists who were contacted said they were not taking new patients, which was the most common reason given for unavailability.
“This is happening at the worst time, when we are seeing mental health issues spike,” Ms. McDaid said.
Telepsychiatry helpful but no panacea
The fact that wait times were a bit shorter for telepsychiatry is encouraging, Ms. McDaid said.
Telepsychiatry is a potential solution to provider shortages and geographic barriers, but it does not resolve the concerning shortage of psychiatric outpatient care, she noted.
“Psychiatrists adapted very well to telepsychiatry during COVID,” Saul Levin, MD, MPA, chief executive officer and medical director of the APA, noted during a preconference briefing with reporters.
“Before COVID, we always thought that the psychiatrist had to be with the patient in the room,” said Dr. Levin. But now we see that either “sitting inside the room with your psychiatrist or mental health specialist or [being there virtually] has the same effect. The patient is concentrating and working out their problems with you. I think that’s one of the positives – if anything coming out of COVID is positive.”
In an interview, Robert Trestman, MD, chair of the APA Council on Healthcare Systems and Financing, said telepsychiatry “will help, but there is not one simple solution that will fix the problem” regarding access to mental health care.
One promising approach is the collaborative care model, which enlists primary care physicians to provide mental health care in consultation with psychiatry and case management, Dr. Trestman said.
“There’s no question that there aren’t enough providers. There aren’t enough primary care doctors, and there certainly aren’t enough psychiatrists,” Dr. Trestman noted.
Encouragingly, however, the past few years have seen a steady increase in medical students choosing psychiatry.
“Psychiatry is now being thought of as a branch of neuroscience. We are understanding so much more about the field and about the brain. So that’s intriguing and intellectually challenging to many,” Dr. Trestman said.
He also noted that the pandemic has helped to “break down stigma. More people acknowledge and talk about mental health, and when an area is destigmatized, it’s so much easier for people to consider.”
Jack Resneck, Jr., MD, president of the American Medical Association, acknowledged that there is a “severe workforce shortage in health care right now.”
“I’m a physician and the president of the AMA, and it took me way too long to be able to find a primary care physician for myself,” he said.
“I also am a physician who refers patients to rheumatology and endocrinology, psychiatry, and other areas of medicine, and it is, in many geographic areas both rural and urban, a huge struggle right now,” said Dr. Resneck.
The study had no specific funding. Ms. McDaid, Dr. Levin, and Dr. Trestman have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT APA 2023
Abrocitinib remains effective at 96 weeks, in older as well as younger adults
WASHINGTON – A substantial proportion of , Andrew F. Alexis, MD, MPH, reported in a late-breaker abstract session at the annual Revolutionizing Atopic Dermatitis conference.
The analysis stratified patients by age – 18-50 and over 50 years – and found that the sustained improvement with the JAK-1 selective inhibitor as monotherapy was seen regardless of age. “In practice, patients who are older tend to have had AD for a longer period of time and tend to be more difficult to treat so it’s reassuring to see that even in the over-50 age group, they show substantial responses, even with more stringent endpoints,” said Dr. Alexis, professor of clinical dermatology at Weill Cornell Medical College, New York.
At week 96, for instance, the proportion of patients who achieved at least a 75% improvement from baseline on the Eczema Area and Severity Index (EASI-75) was 73% with the 100-mg dose and 85% with the 200-mg dose in the younger age group, and 86% and 89%, respectively, in the older age group.
An EASI-90 response – one of the more stringent outcomes – was achieved by 45% and 58% in the 18-50 group and 58% and 73% in the over 50 group (for 100-mg and 200-mg doses, respectively), Dr. Alexis reported.
The interim analysis also showed dose-dependent efficacy overall up to 96 weeks in the younger age group but only up to 48 weeks in the older age group. Response to some outcome measures in patients over age 50 years was “less clearly dose dependent after week 48” than earlier, Dr. Alexis said.
The ongoing JADE EXTEND trial enrolled patients who had participated in the phase 3 JADE clinical trials. This analysis covered 1,309 patients who were enrolled by a September 2021 cutoff. The patient population leaned young: Eighty percent (1,046) were aged 18-50, and 20% (263) were over 50.
Patients who were randomly assigned to abrocitinib 200 mg or 100 mg in the parent trials continued to receive the same dose in JADE EXTEND with blinding maintained. Those who received placebo in the qualifying trial were randomly assigned to abrocitinib 200 mg or 100 mg. And patients from JADE DARE continued with their dosing of 200 mg. Grouping by age for the analysis was made based on the age recorded at the screening visit of the qualifying trial.
IGA, PP-NRS, and DLQI results
At week 96, the proportion of patients 18-50 years of age who achieved the Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with at least a 2-grade improvement from baseline was 44% in the 100-mg group and 55% in the 200-mg group. Among patients over 50, these proportions were 51% and 58%, respectively.
The proportion of patients who achieved at least a 4-point improvement from baseline in the Peak Pruritus Numerical Rating Scale (PP-NRS) score was 54% and 66% (on 100 mg and 200 mg, respectively) among those aged 18-50, and 79% and 80%, respectively, among those over 50.
Looking at more stringent outcomes, 26% and 38% in the 18-50 group on 100 mg and 200 mg, respectively, achieved a PP-NRS of 0/1, as did 54% and 44% in the over-50 group.
Lastly, a score of less than 2 on the Dermatology Life Quality Index (DLQI 0/1) was achieved by 32% and 41% of patients aged 18-50 and by 51% and 48% of patients over 50, for the 100-mg and 200-mg doses, respectively.
The decline in dose-dependent efficacy in the older age group after 48 weeks may be due to the smaller sample of older patients and/or the fact that a higher proportion of older patients had moderate baseline disease per their IGA score, versus severe disease, compared with the younger patients, Dr. Alexis said. “We see a skewing toward a bit more severe [disease] in the younger age group compared to the older,” he noted.
Abrocitinib (Cibinqo) is approved for the treatment of moderate to severe AD in adolescents aged 12 and up and adults whose disease is not adequately controlled with other systemic treatments or those for whom the use of these drugs is not advised. It is available in a 50-mg dose for dose adjustments in special populations, but this dose was not studied in the clinical trials, Dr. Alexis noted. The interim analysis did not include safety data.
In a separate presentation in which he reviewed long-term data on AD medications, Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, said that most patients who meet defined endpoints at week 12 of treatment with abrocitinib maintain that response over time. “By and large, there’s a steep initial rise that flattens over the long run, which is what you want to see. People getting that response are generally staying there over the course of treatment,” he said, referring to the JADE EXTEND data up to week 48.
It’s important to also appreciate, however, that the proportion of patients meeting efficacy outcomes in the trials of abrocitinib has grown well beyond 12 weeks, Dr. Chovatiya said.
Pointing to data presented at a 2021 RAD meeting depicting the proportion of 12-week nonresponders achieving a response at weeks 24 and 48 on IGA 0/1, EASI-75, and PP-NRS, Dr. Chovatiya said the level of response grew at both time points. “You’re capturing a chunk of people well beyond the primary endpoint if you keep them on therapy continuously, suggesting that ... we may need to reframe how we’re thinking about oral JAK inhibitors,” he said. “Not only are they rapidly acting, but they are medications that can provide good control and changes in the long run.”
Dr. Alexis and Dr. Chovatiya disclosed ties with Pfizer, which funded the study.
WASHINGTON – A substantial proportion of , Andrew F. Alexis, MD, MPH, reported in a late-breaker abstract session at the annual Revolutionizing Atopic Dermatitis conference.
The analysis stratified patients by age – 18-50 and over 50 years – and found that the sustained improvement with the JAK-1 selective inhibitor as monotherapy was seen regardless of age. “In practice, patients who are older tend to have had AD for a longer period of time and tend to be more difficult to treat so it’s reassuring to see that even in the over-50 age group, they show substantial responses, even with more stringent endpoints,” said Dr. Alexis, professor of clinical dermatology at Weill Cornell Medical College, New York.
At week 96, for instance, the proportion of patients who achieved at least a 75% improvement from baseline on the Eczema Area and Severity Index (EASI-75) was 73% with the 100-mg dose and 85% with the 200-mg dose in the younger age group, and 86% and 89%, respectively, in the older age group.
An EASI-90 response – one of the more stringent outcomes – was achieved by 45% and 58% in the 18-50 group and 58% and 73% in the over 50 group (for 100-mg and 200-mg doses, respectively), Dr. Alexis reported.
The interim analysis also showed dose-dependent efficacy overall up to 96 weeks in the younger age group but only up to 48 weeks in the older age group. Response to some outcome measures in patients over age 50 years was “less clearly dose dependent after week 48” than earlier, Dr. Alexis said.
The ongoing JADE EXTEND trial enrolled patients who had participated in the phase 3 JADE clinical trials. This analysis covered 1,309 patients who were enrolled by a September 2021 cutoff. The patient population leaned young: Eighty percent (1,046) were aged 18-50, and 20% (263) were over 50.
Patients who were randomly assigned to abrocitinib 200 mg or 100 mg in the parent trials continued to receive the same dose in JADE EXTEND with blinding maintained. Those who received placebo in the qualifying trial were randomly assigned to abrocitinib 200 mg or 100 mg. And patients from JADE DARE continued with their dosing of 200 mg. Grouping by age for the analysis was made based on the age recorded at the screening visit of the qualifying trial.
IGA, PP-NRS, and DLQI results
At week 96, the proportion of patients 18-50 years of age who achieved the Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with at least a 2-grade improvement from baseline was 44% in the 100-mg group and 55% in the 200-mg group. Among patients over 50, these proportions were 51% and 58%, respectively.
The proportion of patients who achieved at least a 4-point improvement from baseline in the Peak Pruritus Numerical Rating Scale (PP-NRS) score was 54% and 66% (on 100 mg and 200 mg, respectively) among those aged 18-50, and 79% and 80%, respectively, among those over 50.
Looking at more stringent outcomes, 26% and 38% in the 18-50 group on 100 mg and 200 mg, respectively, achieved a PP-NRS of 0/1, as did 54% and 44% in the over-50 group.
Lastly, a score of less than 2 on the Dermatology Life Quality Index (DLQI 0/1) was achieved by 32% and 41% of patients aged 18-50 and by 51% and 48% of patients over 50, for the 100-mg and 200-mg doses, respectively.
The decline in dose-dependent efficacy in the older age group after 48 weeks may be due to the smaller sample of older patients and/or the fact that a higher proportion of older patients had moderate baseline disease per their IGA score, versus severe disease, compared with the younger patients, Dr. Alexis said. “We see a skewing toward a bit more severe [disease] in the younger age group compared to the older,” he noted.
Abrocitinib (Cibinqo) is approved for the treatment of moderate to severe AD in adolescents aged 12 and up and adults whose disease is not adequately controlled with other systemic treatments or those for whom the use of these drugs is not advised. It is available in a 50-mg dose for dose adjustments in special populations, but this dose was not studied in the clinical trials, Dr. Alexis noted. The interim analysis did not include safety data.
In a separate presentation in which he reviewed long-term data on AD medications, Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, said that most patients who meet defined endpoints at week 12 of treatment with abrocitinib maintain that response over time. “By and large, there’s a steep initial rise that flattens over the long run, which is what you want to see. People getting that response are generally staying there over the course of treatment,” he said, referring to the JADE EXTEND data up to week 48.
It’s important to also appreciate, however, that the proportion of patients meeting efficacy outcomes in the trials of abrocitinib has grown well beyond 12 weeks, Dr. Chovatiya said.
Pointing to data presented at a 2021 RAD meeting depicting the proportion of 12-week nonresponders achieving a response at weeks 24 and 48 on IGA 0/1, EASI-75, and PP-NRS, Dr. Chovatiya said the level of response grew at both time points. “You’re capturing a chunk of people well beyond the primary endpoint if you keep them on therapy continuously, suggesting that ... we may need to reframe how we’re thinking about oral JAK inhibitors,” he said. “Not only are they rapidly acting, but they are medications that can provide good control and changes in the long run.”
Dr. Alexis and Dr. Chovatiya disclosed ties with Pfizer, which funded the study.
WASHINGTON – A substantial proportion of , Andrew F. Alexis, MD, MPH, reported in a late-breaker abstract session at the annual Revolutionizing Atopic Dermatitis conference.
The analysis stratified patients by age – 18-50 and over 50 years – and found that the sustained improvement with the JAK-1 selective inhibitor as monotherapy was seen regardless of age. “In practice, patients who are older tend to have had AD for a longer period of time and tend to be more difficult to treat so it’s reassuring to see that even in the over-50 age group, they show substantial responses, even with more stringent endpoints,” said Dr. Alexis, professor of clinical dermatology at Weill Cornell Medical College, New York.
At week 96, for instance, the proportion of patients who achieved at least a 75% improvement from baseline on the Eczema Area and Severity Index (EASI-75) was 73% with the 100-mg dose and 85% with the 200-mg dose in the younger age group, and 86% and 89%, respectively, in the older age group.
An EASI-90 response – one of the more stringent outcomes – was achieved by 45% and 58% in the 18-50 group and 58% and 73% in the over 50 group (for 100-mg and 200-mg doses, respectively), Dr. Alexis reported.
The interim analysis also showed dose-dependent efficacy overall up to 96 weeks in the younger age group but only up to 48 weeks in the older age group. Response to some outcome measures in patients over age 50 years was “less clearly dose dependent after week 48” than earlier, Dr. Alexis said.
The ongoing JADE EXTEND trial enrolled patients who had participated in the phase 3 JADE clinical trials. This analysis covered 1,309 patients who were enrolled by a September 2021 cutoff. The patient population leaned young: Eighty percent (1,046) were aged 18-50, and 20% (263) were over 50.
Patients who were randomly assigned to abrocitinib 200 mg or 100 mg in the parent trials continued to receive the same dose in JADE EXTEND with blinding maintained. Those who received placebo in the qualifying trial were randomly assigned to abrocitinib 200 mg or 100 mg. And patients from JADE DARE continued with their dosing of 200 mg. Grouping by age for the analysis was made based on the age recorded at the screening visit of the qualifying trial.
IGA, PP-NRS, and DLQI results
At week 96, the proportion of patients 18-50 years of age who achieved the Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with at least a 2-grade improvement from baseline was 44% in the 100-mg group and 55% in the 200-mg group. Among patients over 50, these proportions were 51% and 58%, respectively.
The proportion of patients who achieved at least a 4-point improvement from baseline in the Peak Pruritus Numerical Rating Scale (PP-NRS) score was 54% and 66% (on 100 mg and 200 mg, respectively) among those aged 18-50, and 79% and 80%, respectively, among those over 50.
Looking at more stringent outcomes, 26% and 38% in the 18-50 group on 100 mg and 200 mg, respectively, achieved a PP-NRS of 0/1, as did 54% and 44% in the over-50 group.
Lastly, a score of less than 2 on the Dermatology Life Quality Index (DLQI 0/1) was achieved by 32% and 41% of patients aged 18-50 and by 51% and 48% of patients over 50, for the 100-mg and 200-mg doses, respectively.
The decline in dose-dependent efficacy in the older age group after 48 weeks may be due to the smaller sample of older patients and/or the fact that a higher proportion of older patients had moderate baseline disease per their IGA score, versus severe disease, compared with the younger patients, Dr. Alexis said. “We see a skewing toward a bit more severe [disease] in the younger age group compared to the older,” he noted.
Abrocitinib (Cibinqo) is approved for the treatment of moderate to severe AD in adolescents aged 12 and up and adults whose disease is not adequately controlled with other systemic treatments or those for whom the use of these drugs is not advised. It is available in a 50-mg dose for dose adjustments in special populations, but this dose was not studied in the clinical trials, Dr. Alexis noted. The interim analysis did not include safety data.
In a separate presentation in which he reviewed long-term data on AD medications, Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, said that most patients who meet defined endpoints at week 12 of treatment with abrocitinib maintain that response over time. “By and large, there’s a steep initial rise that flattens over the long run, which is what you want to see. People getting that response are generally staying there over the course of treatment,” he said, referring to the JADE EXTEND data up to week 48.
It’s important to also appreciate, however, that the proportion of patients meeting efficacy outcomes in the trials of abrocitinib has grown well beyond 12 weeks, Dr. Chovatiya said.
Pointing to data presented at a 2021 RAD meeting depicting the proportion of 12-week nonresponders achieving a response at weeks 24 and 48 on IGA 0/1, EASI-75, and PP-NRS, Dr. Chovatiya said the level of response grew at both time points. “You’re capturing a chunk of people well beyond the primary endpoint if you keep them on therapy continuously, suggesting that ... we may need to reframe how we’re thinking about oral JAK inhibitors,” he said. “Not only are they rapidly acting, but they are medications that can provide good control and changes in the long run.”
Dr. Alexis and Dr. Chovatiya disclosed ties with Pfizer, which funded the study.
AT RAD 2023
Psychiatrists: Don’t fear clozapine in treatment-resistant schizophrenia
SAN FRANCISCO – A trio of psychiatrists urged colleagues at the annual meeting of the American Psychiatric Association to embrace the venerable antipsychotic clozapine in patients with treatment-resistant schizophrenia. They cautioned that clinicians may overestimate the true risk of the adverse effect of neutropenia in minority populations.
“Although clozapine is known to be a life-improving and even potentially lifesaving treatment, it remains underutilized in the U.S.,” said Claire C. Holderness, MD, a psychiatrist at Columbia University Irving Medical Center, New York. “It’s been estimated that between 35% and 40% of all patients with schizophrenia should be considered for a clozapine trial. However, only 4%-5% of patients with schizophrenia in the U.S. have ever received clozapine. This is in sharp contrast to other industrialized countries where approximately 20% or more of patients with schizophrenia are treated with clozapine.”
According to Dr. Holderness, research has shown that clozapine is even less likely to be prescribed to racial and ethnic minorities. A 2022 systematic review, for example, found that Black patients in the United States had between one-third and two-thirds the odds of being treated with the drug, compared with White patients after adjustment for potential confounders such as demographics. Hispanic/Latino patients were also less likely than Whites to be prescribed the drug.
As Dr. Holderness put it, the drug “been shown to be more effective in treatment-resistant schizophrenia than any other antipsychotic medication. Clozapine is also the most cost-effective treatment for treatment-resistant schizophrenia.” So why does this disparity exist despite clozapine’s benefits?
A 2018 systematic review of barriers to the drug’s usage identified several factors: “mandatory blood testing, fear of serious side-effects and lack of adherence by the patients, difficulty in identifying suitable patients, service fragmentation, and inadequate training in or exposure to using clozapine.” A 2016 British study, meanwhile, looked at the reasons that 45% of 316 patients stopped clozapine before 2 years. More than half of these patients stopped because of adverse effects.
Risk of neutropenia
At the APA presentation, psychiatrist Laura Clarke, MD, also of Columbia University Irving Medical Center, noted that there’s concern about one adverse effect in particular: neutropenia, or an abnormally low white blood cell count. Clozapine, she said, has a boxed warning about severe neutropenia that can lead to death.
However, she cautioned that white blood cell counts can be misleading. Some people in non-White ethnic groups have a condition known as benign ethnic neutropenia: their white blood cell counts are abnormal by the standards of people of European heritage, but they’re otherwise healthy. “These individuals do not show an increased risk of infections, and their response to infection is similar to those without them,” she said.
As many as 25%-50% of people of African ancestry may have benign ethnic neutropenia, making their blood levels appear abnormally low. Others with higher levels of the condition include certain Middle Eastern ethnicities and other ethnic groups with darker skin.
In these patents, “clinicians may avoid prescribing clozapine out of the mistaken concern that it can worsen neutropenia,” Dr. Clarke said. In fact, benign ethnic neutropenia “does not increase the risk of clozapine-induced severe neutropenia.”
Dr. Clarke highlighted drug use guidelines from the Clozapine Risk Evaluation and Mitigation Strategy, a Food and Drug Administration–mandated safety program designed to prevent severe neutropenia in patients taking clozapine. The guidelines note that the recommended absolute neutrophil count monitoring algorithm differs when patients are diagnosed with benign ethnic neutropenia.
T. Scott Stroup, MD, MPH, a psychiatrist at Columbia University, New York, urged his colleagues to consider clozapine early on in treatment-resistant schizophrenia. “Don’t go through three, four, or five antipsychotics. Even after trying two, I’d encourage people to [try clozapine].”
However, he acknowledged that “not everyone believes that. Many of my colleagues think that, before you try clozapine, you should have a trial of long-acting injectable medications to rule out pseudo–treatment resistance. I don’t totally agree with that, but I’ve more or less lost that battle,” he added.
In the big picture, Dr. Stroup said, clozapine “is good when other things aren’t working efficacy wise.”
Dr. Holderness and Dr. Clarke have no disclosures. Dr. Stroup discloses grants from the National Institutes of Health and royalties from APA Publishing and UpToDate.
SAN FRANCISCO – A trio of psychiatrists urged colleagues at the annual meeting of the American Psychiatric Association to embrace the venerable antipsychotic clozapine in patients with treatment-resistant schizophrenia. They cautioned that clinicians may overestimate the true risk of the adverse effect of neutropenia in minority populations.
“Although clozapine is known to be a life-improving and even potentially lifesaving treatment, it remains underutilized in the U.S.,” said Claire C. Holderness, MD, a psychiatrist at Columbia University Irving Medical Center, New York. “It’s been estimated that between 35% and 40% of all patients with schizophrenia should be considered for a clozapine trial. However, only 4%-5% of patients with schizophrenia in the U.S. have ever received clozapine. This is in sharp contrast to other industrialized countries where approximately 20% or more of patients with schizophrenia are treated with clozapine.”
According to Dr. Holderness, research has shown that clozapine is even less likely to be prescribed to racial and ethnic minorities. A 2022 systematic review, for example, found that Black patients in the United States had between one-third and two-thirds the odds of being treated with the drug, compared with White patients after adjustment for potential confounders such as demographics. Hispanic/Latino patients were also less likely than Whites to be prescribed the drug.
As Dr. Holderness put it, the drug “been shown to be more effective in treatment-resistant schizophrenia than any other antipsychotic medication. Clozapine is also the most cost-effective treatment for treatment-resistant schizophrenia.” So why does this disparity exist despite clozapine’s benefits?
A 2018 systematic review of barriers to the drug’s usage identified several factors: “mandatory blood testing, fear of serious side-effects and lack of adherence by the patients, difficulty in identifying suitable patients, service fragmentation, and inadequate training in or exposure to using clozapine.” A 2016 British study, meanwhile, looked at the reasons that 45% of 316 patients stopped clozapine before 2 years. More than half of these patients stopped because of adverse effects.
Risk of neutropenia
At the APA presentation, psychiatrist Laura Clarke, MD, also of Columbia University Irving Medical Center, noted that there’s concern about one adverse effect in particular: neutropenia, or an abnormally low white blood cell count. Clozapine, she said, has a boxed warning about severe neutropenia that can lead to death.
However, she cautioned that white blood cell counts can be misleading. Some people in non-White ethnic groups have a condition known as benign ethnic neutropenia: their white blood cell counts are abnormal by the standards of people of European heritage, but they’re otherwise healthy. “These individuals do not show an increased risk of infections, and their response to infection is similar to those without them,” she said.
As many as 25%-50% of people of African ancestry may have benign ethnic neutropenia, making their blood levels appear abnormally low. Others with higher levels of the condition include certain Middle Eastern ethnicities and other ethnic groups with darker skin.
In these patents, “clinicians may avoid prescribing clozapine out of the mistaken concern that it can worsen neutropenia,” Dr. Clarke said. In fact, benign ethnic neutropenia “does not increase the risk of clozapine-induced severe neutropenia.”
Dr. Clarke highlighted drug use guidelines from the Clozapine Risk Evaluation and Mitigation Strategy, a Food and Drug Administration–mandated safety program designed to prevent severe neutropenia in patients taking clozapine. The guidelines note that the recommended absolute neutrophil count monitoring algorithm differs when patients are diagnosed with benign ethnic neutropenia.
T. Scott Stroup, MD, MPH, a psychiatrist at Columbia University, New York, urged his colleagues to consider clozapine early on in treatment-resistant schizophrenia. “Don’t go through three, four, or five antipsychotics. Even after trying two, I’d encourage people to [try clozapine].”
However, he acknowledged that “not everyone believes that. Many of my colleagues think that, before you try clozapine, you should have a trial of long-acting injectable medications to rule out pseudo–treatment resistance. I don’t totally agree with that, but I’ve more or less lost that battle,” he added.
In the big picture, Dr. Stroup said, clozapine “is good when other things aren’t working efficacy wise.”
Dr. Holderness and Dr. Clarke have no disclosures. Dr. Stroup discloses grants from the National Institutes of Health and royalties from APA Publishing and UpToDate.
SAN FRANCISCO – A trio of psychiatrists urged colleagues at the annual meeting of the American Psychiatric Association to embrace the venerable antipsychotic clozapine in patients with treatment-resistant schizophrenia. They cautioned that clinicians may overestimate the true risk of the adverse effect of neutropenia in minority populations.
“Although clozapine is known to be a life-improving and even potentially lifesaving treatment, it remains underutilized in the U.S.,” said Claire C. Holderness, MD, a psychiatrist at Columbia University Irving Medical Center, New York. “It’s been estimated that between 35% and 40% of all patients with schizophrenia should be considered for a clozapine trial. However, only 4%-5% of patients with schizophrenia in the U.S. have ever received clozapine. This is in sharp contrast to other industrialized countries where approximately 20% or more of patients with schizophrenia are treated with clozapine.”
According to Dr. Holderness, research has shown that clozapine is even less likely to be prescribed to racial and ethnic minorities. A 2022 systematic review, for example, found that Black patients in the United States had between one-third and two-thirds the odds of being treated with the drug, compared with White patients after adjustment for potential confounders such as demographics. Hispanic/Latino patients were also less likely than Whites to be prescribed the drug.
As Dr. Holderness put it, the drug “been shown to be more effective in treatment-resistant schizophrenia than any other antipsychotic medication. Clozapine is also the most cost-effective treatment for treatment-resistant schizophrenia.” So why does this disparity exist despite clozapine’s benefits?
A 2018 systematic review of barriers to the drug’s usage identified several factors: “mandatory blood testing, fear of serious side-effects and lack of adherence by the patients, difficulty in identifying suitable patients, service fragmentation, and inadequate training in or exposure to using clozapine.” A 2016 British study, meanwhile, looked at the reasons that 45% of 316 patients stopped clozapine before 2 years. More than half of these patients stopped because of adverse effects.
Risk of neutropenia
At the APA presentation, psychiatrist Laura Clarke, MD, also of Columbia University Irving Medical Center, noted that there’s concern about one adverse effect in particular: neutropenia, or an abnormally low white blood cell count. Clozapine, she said, has a boxed warning about severe neutropenia that can lead to death.
However, she cautioned that white blood cell counts can be misleading. Some people in non-White ethnic groups have a condition known as benign ethnic neutropenia: their white blood cell counts are abnormal by the standards of people of European heritage, but they’re otherwise healthy. “These individuals do not show an increased risk of infections, and their response to infection is similar to those without them,” she said.
As many as 25%-50% of people of African ancestry may have benign ethnic neutropenia, making their blood levels appear abnormally low. Others with higher levels of the condition include certain Middle Eastern ethnicities and other ethnic groups with darker skin.
In these patents, “clinicians may avoid prescribing clozapine out of the mistaken concern that it can worsen neutropenia,” Dr. Clarke said. In fact, benign ethnic neutropenia “does not increase the risk of clozapine-induced severe neutropenia.”
Dr. Clarke highlighted drug use guidelines from the Clozapine Risk Evaluation and Mitigation Strategy, a Food and Drug Administration–mandated safety program designed to prevent severe neutropenia in patients taking clozapine. The guidelines note that the recommended absolute neutrophil count monitoring algorithm differs when patients are diagnosed with benign ethnic neutropenia.
T. Scott Stroup, MD, MPH, a psychiatrist at Columbia University, New York, urged his colleagues to consider clozapine early on in treatment-resistant schizophrenia. “Don’t go through three, four, or five antipsychotics. Even after trying two, I’d encourage people to [try clozapine].”
However, he acknowledged that “not everyone believes that. Many of my colleagues think that, before you try clozapine, you should have a trial of long-acting injectable medications to rule out pseudo–treatment resistance. I don’t totally agree with that, but I’ve more or less lost that battle,” he added.
In the big picture, Dr. Stroup said, clozapine “is good when other things aren’t working efficacy wise.”
Dr. Holderness and Dr. Clarke have no disclosures. Dr. Stroup discloses grants from the National Institutes of Health and royalties from APA Publishing and UpToDate.
AT APA 2023