User login
Updated Guidelines for COPD Management: 2023 GOLD Strategy Report
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2023 Report). Published 2023. Accessed June 6, 2023. https://goldcopd.org/2023-gold-report-2/
- Celli B et al. Am J Respir Crit Care Med. 2022;206(11):1317. doi:10.1164/rccm.202204-0671PP
- Han M et al. Lancet Respir Med. 2013;1(1):43-50. doi:10.1016/S2213-2600(12)70044-9
- Klijn SL et al. NPJ Prim Care Respir Med. 2017;27(1):24. doi:10.1038/s41533-017-0022-1
- Chan AH et al. J Allergy Clin Immunol Pract. 2015;3(3):335-349.e1-e5. doi:10.1016/j.jaip.2015.01.024
- Brusselle G et al. Int J Chron Obstruct Pulmon Dis. 2015;10:2207-2217. doi:10.2147/COPD.S91694
- Salvi SS, Barnes PJ. Lancet. 2009;374(9691):733-743. doi:10.1016/S0140-6736(09)61303-9
- Trupin L et al. Eur Respir J. 2003;22(3):462-469. doi:10.1183/09031936.03.00094203
- Celli BR et al. Am J Respir Crit Care Med. 2021;204(11):1251-1258. doi:10.1164/rccm.202108-1819PP
- Barnes PJ, Celli BR. Eur Respir J. 2009;33(5):1165-1185. doi:10.1183/09031936.00128008
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2023 Report). Published 2023. Accessed June 6, 2023. https://goldcopd.org/2023-gold-report-2/
- Celli B et al. Am J Respir Crit Care Med. 2022;206(11):1317. doi:10.1164/rccm.202204-0671PP
- Han M et al. Lancet Respir Med. 2013;1(1):43-50. doi:10.1016/S2213-2600(12)70044-9
- Klijn SL et al. NPJ Prim Care Respir Med. 2017;27(1):24. doi:10.1038/s41533-017-0022-1
- Chan AH et al. J Allergy Clin Immunol Pract. 2015;3(3):335-349.e1-e5. doi:10.1016/j.jaip.2015.01.024
- Brusselle G et al. Int J Chron Obstruct Pulmon Dis. 2015;10:2207-2217. doi:10.2147/COPD.S91694
- Salvi SS, Barnes PJ. Lancet. 2009;374(9691):733-743. doi:10.1016/S0140-6736(09)61303-9
- Trupin L et al. Eur Respir J. 2003;22(3):462-469. doi:10.1183/09031936.03.00094203
- Celli BR et al. Am J Respir Crit Care Med. 2021;204(11):1251-1258. doi:10.1164/rccm.202108-1819PP
- Barnes PJ, Celli BR. Eur Respir J. 2009;33(5):1165-1185. doi:10.1183/09031936.00128008
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2023 Report). Published 2023. Accessed June 6, 2023. https://goldcopd.org/2023-gold-report-2/
- Celli B et al. Am J Respir Crit Care Med. 2022;206(11):1317. doi:10.1164/rccm.202204-0671PP
- Han M et al. Lancet Respir Med. 2013;1(1):43-50. doi:10.1016/S2213-2600(12)70044-9
- Klijn SL et al. NPJ Prim Care Respir Med. 2017;27(1):24. doi:10.1038/s41533-017-0022-1
- Chan AH et al. J Allergy Clin Immunol Pract. 2015;3(3):335-349.e1-e5. doi:10.1016/j.jaip.2015.01.024
- Brusselle G et al. Int J Chron Obstruct Pulmon Dis. 2015;10:2207-2217. doi:10.2147/COPD.S91694
- Salvi SS, Barnes PJ. Lancet. 2009;374(9691):733-743. doi:10.1016/S0140-6736(09)61303-9
- Trupin L et al. Eur Respir J. 2003;22(3):462-469. doi:10.1183/09031936.03.00094203
- Celli BR et al. Am J Respir Crit Care Med. 2021;204(11):1251-1258. doi:10.1164/rccm.202108-1819PP
- Barnes PJ, Celli BR. Eur Respir J. 2009;33(5):1165-1185. doi:10.1183/09031936.00128008
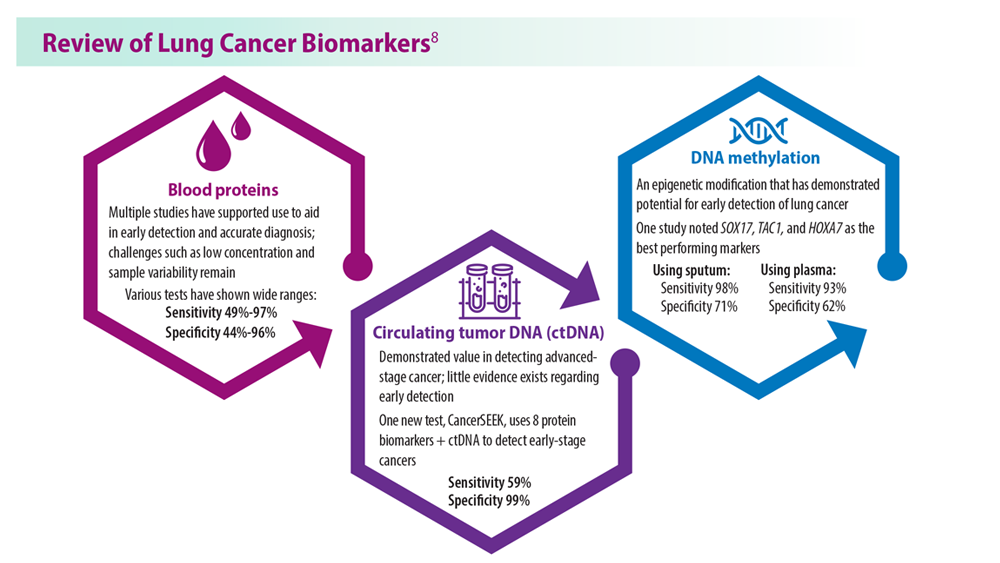
Lung Cancer Screening: A Need for Adjunctive Testing
- Naidch DP et al. Radiology. 1990;175(3):729-731. doi:10.1148/radiology.175.3.2343122
- Kaneko M et al. Radiology. 1996;201(3):798-802. doi:10.1148/radiology.201.3.8939234
- National Lung Screening Trial Research Team. Radiology. 2011;258(1):243-253. doi:10.1148/radiol.10091808
- National Lung Screening Trial Research Team. J Thorac Oncol. 2019;14(10):1732-1742. doi:10.1016/j.jtho.2019.05.044
- Mazzone PJ et al. Chest. 2021;160(5):e427-e494. doi:10.1016/j.chest.2021.06.063
- Tanner NT et al. Chest. 2023;S0012-3692(23)00175-7. doi:10.1016/j.chest.2023.02.003
- National Lung Screening Trial Research Team. N Engl J Med. 2011;365(5):395- 409. doi:10.1056/NEJMoa1102873
- Marmor HN et al. Curr Chall Thorac Surg. 2023;5:5. doi:10.21037/ccts-20-171
- Naidch DP et al. Radiology. 1990;175(3):729-731. doi:10.1148/radiology.175.3.2343122
- Kaneko M et al. Radiology. 1996;201(3):798-802. doi:10.1148/radiology.201.3.8939234
- National Lung Screening Trial Research Team. Radiology. 2011;258(1):243-253. doi:10.1148/radiol.10091808
- National Lung Screening Trial Research Team. J Thorac Oncol. 2019;14(10):1732-1742. doi:10.1016/j.jtho.2019.05.044
- Mazzone PJ et al. Chest. 2021;160(5):e427-e494. doi:10.1016/j.chest.2021.06.063
- Tanner NT et al. Chest. 2023;S0012-3692(23)00175-7. doi:10.1016/j.chest.2023.02.003
- National Lung Screening Trial Research Team. N Engl J Med. 2011;365(5):395- 409. doi:10.1056/NEJMoa1102873
- Marmor HN et al. Curr Chall Thorac Surg. 2023;5:5. doi:10.21037/ccts-20-171
- Naidch DP et al. Radiology. 1990;175(3):729-731. doi:10.1148/radiology.175.3.2343122
- Kaneko M et al. Radiology. 1996;201(3):798-802. doi:10.1148/radiology.201.3.8939234
- National Lung Screening Trial Research Team. Radiology. 2011;258(1):243-253. doi:10.1148/radiol.10091808
- National Lung Screening Trial Research Team. J Thorac Oncol. 2019;14(10):1732-1742. doi:10.1016/j.jtho.2019.05.044
- Mazzone PJ et al. Chest. 2021;160(5):e427-e494. doi:10.1016/j.chest.2021.06.063
- Tanner NT et al. Chest. 2023;S0012-3692(23)00175-7. doi:10.1016/j.chest.2023.02.003
- National Lung Screening Trial Research Team. N Engl J Med. 2011;365(5):395- 409. doi:10.1056/NEJMoa1102873
- Marmor HN et al. Curr Chall Thorac Surg. 2023;5:5. doi:10.21037/ccts-20-171
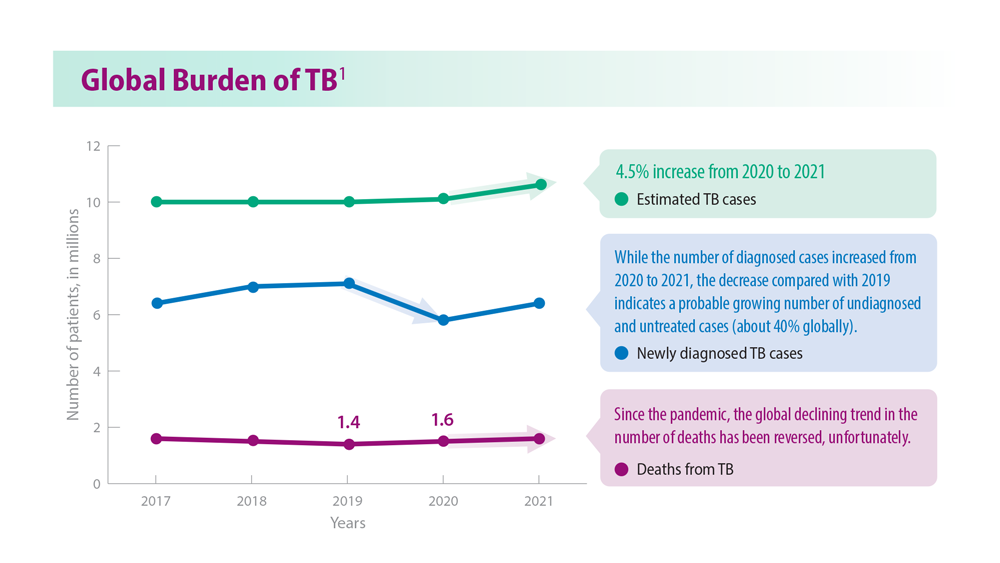
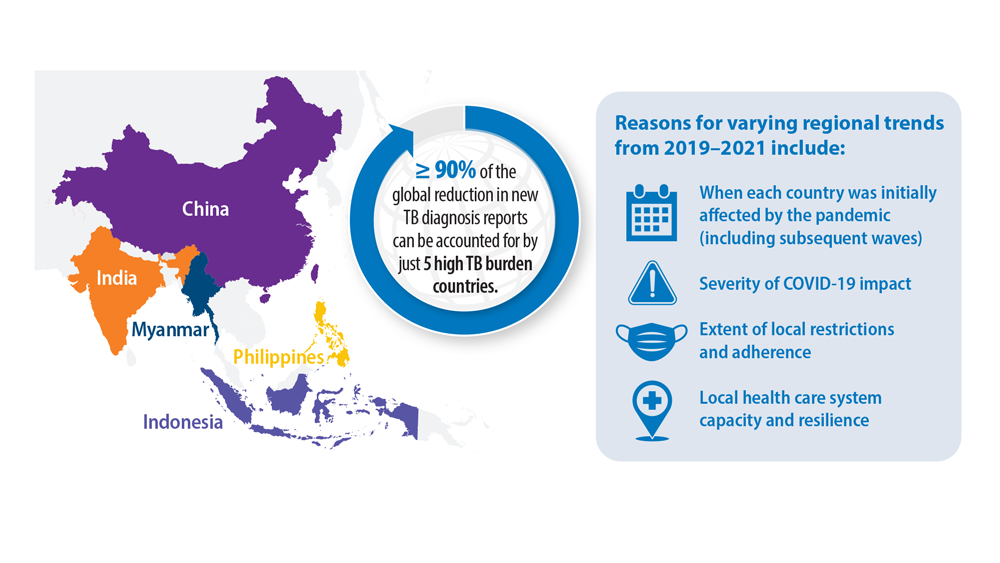
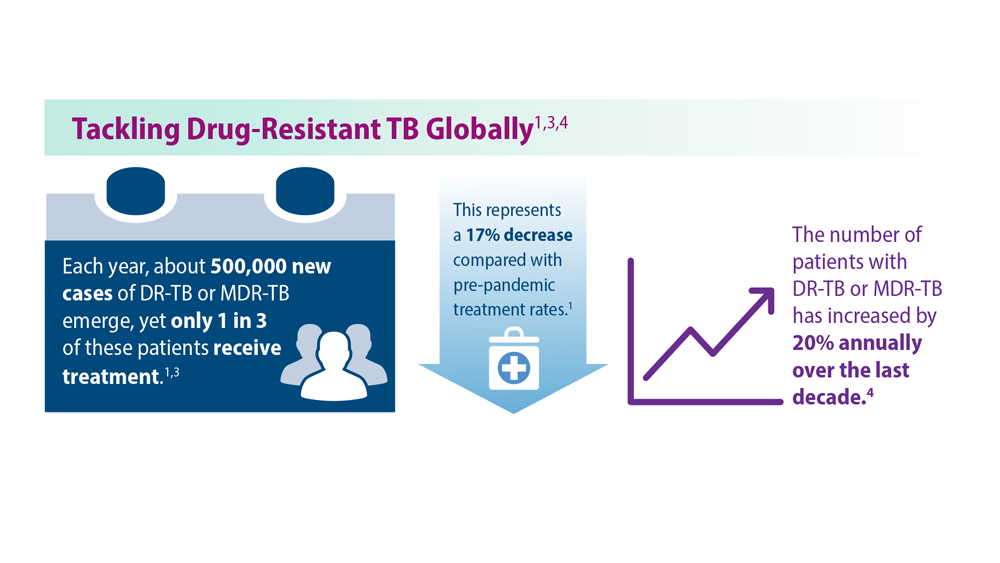
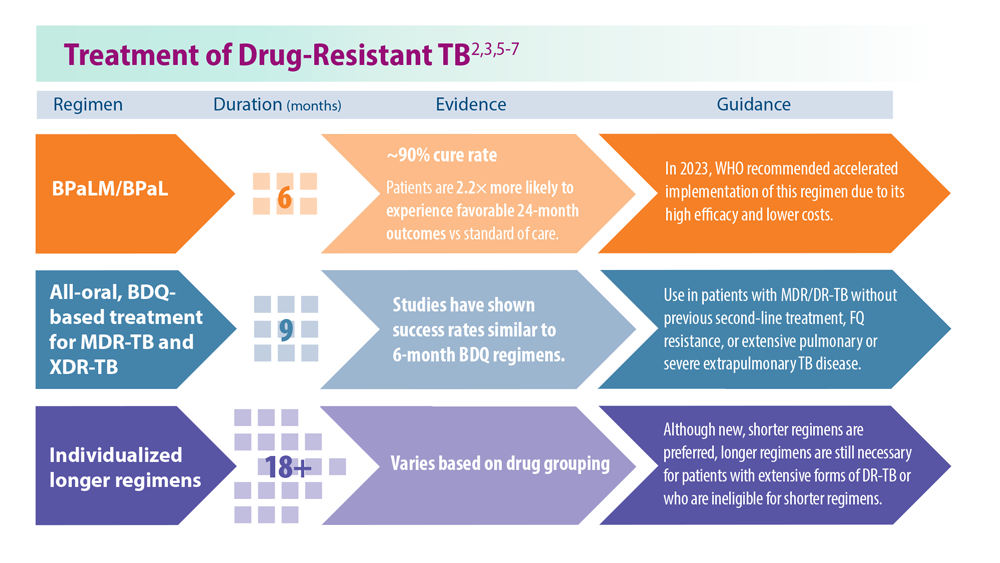
Tuberculosis Management: Returning to Pre-Pandemic Priorities
- Global tuberculosis report 2022. World Health Organization. Published October 27, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240061729
- WHO consolidated guidelines on tuberculosis. Module 4: treatment – drug-resistant tuberculosis treatment, 2022 update. World Health Organization. Published December 15, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240063129
- Migliori GB, Tiberi S. Int J Tuberc Lung Dis. 2022 ;26(7):590-591. doi:10.5588/ijtld.22.0263.
- Lange C et al. Am J Respir Crit Care Med. 2022;205(10):1142-1144. doi:10.1164/rccm.202202-0393ED
- Esmail A et al. Am J Respir Crit Care Med. 2022;205(10):1214-1227. doi:10.1164/rccm.202107-1779OC
- WHO BPaLM Accelerator Platform: to support the call to action for implementation of the shorter and more effective treatment for all people suffering from drug-resistant TB. World Health Organization. Published May 9, 2023. Accessed June 26, 2023. https://www.who.int/news-room/events/detail/2023/05/09/default-calendar/who-bpalm-accelerator-platform–to-support-the-call-to-action-for-implementation-of-the-shorter-and-moreeffective-
- Trevisi L et al. Am J Respir Crit Care Med. 2023;207(11):1525-1532. doi:10.1164/rccm.202211-2125OC
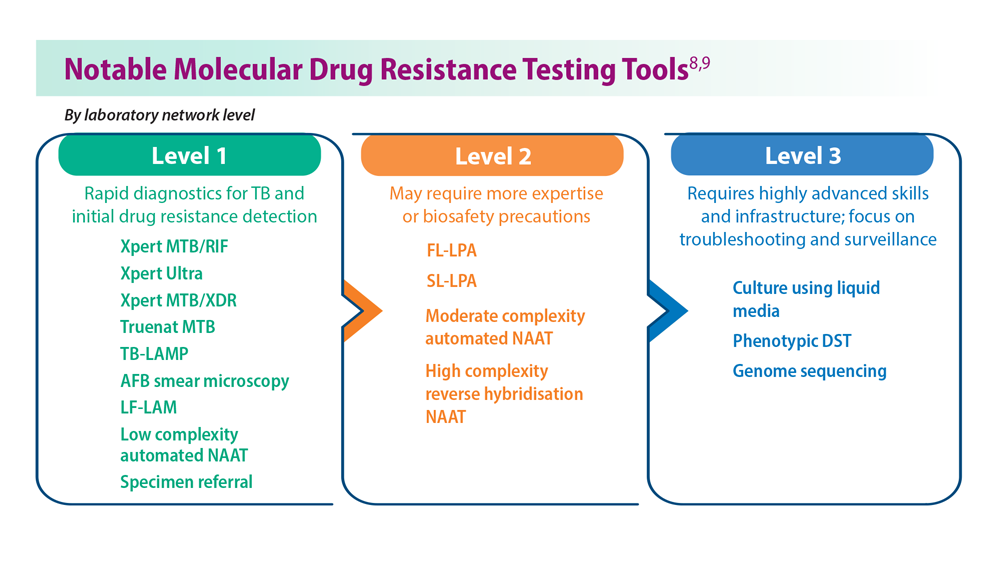
- Domínguez J et al; TBnet and RESIST-TB networks. Lancet Infect Dis. 2023;23(4):e122-e137. doi:10.1016/S1473-3099(22)00875-1
- WHO operational handbook on tuberculosis: module 3: diagnosis: rapid diagnostics for tuberculosis detection, 2021 update. World Health Organization. Published July 7, 2021. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240030589treatment-for-all-people-suffering-from-drug-resistant-tb
- Global tuberculosis report 2022. World Health Organization. Published October 27, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240061729
- WHO consolidated guidelines on tuberculosis. Module 4: treatment – drug-resistant tuberculosis treatment, 2022 update. World Health Organization. Published December 15, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240063129
- Migliori GB, Tiberi S. Int J Tuberc Lung Dis. 2022 ;26(7):590-591. doi:10.5588/ijtld.22.0263.
- Lange C et al. Am J Respir Crit Care Med. 2022;205(10):1142-1144. doi:10.1164/rccm.202202-0393ED
- Esmail A et al. Am J Respir Crit Care Med. 2022;205(10):1214-1227. doi:10.1164/rccm.202107-1779OC
- WHO BPaLM Accelerator Platform: to support the call to action for implementation of the shorter and more effective treatment for all people suffering from drug-resistant TB. World Health Organization. Published May 9, 2023. Accessed June 26, 2023. https://www.who.int/news-room/events/detail/2023/05/09/default-calendar/who-bpalm-accelerator-platform–to-support-the-call-to-action-for-implementation-of-the-shorter-and-moreeffective-
- Trevisi L et al. Am J Respir Crit Care Med. 2023;207(11):1525-1532. doi:10.1164/rccm.202211-2125OC
- Domínguez J et al; TBnet and RESIST-TB networks. Lancet Infect Dis. 2023;23(4):e122-e137. doi:10.1016/S1473-3099(22)00875-1
- WHO operational handbook on tuberculosis: module 3: diagnosis: rapid diagnostics for tuberculosis detection, 2021 update. World Health Organization. Published July 7, 2021. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240030589treatment-for-all-people-suffering-from-drug-resistant-tb
- Global tuberculosis report 2022. World Health Organization. Published October 27, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240061729
- WHO consolidated guidelines on tuberculosis. Module 4: treatment – drug-resistant tuberculosis treatment, 2022 update. World Health Organization. Published December 15, 2022. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240063129
- Migliori GB, Tiberi S. Int J Tuberc Lung Dis. 2022 ;26(7):590-591. doi:10.5588/ijtld.22.0263.
- Lange C et al. Am J Respir Crit Care Med. 2022;205(10):1142-1144. doi:10.1164/rccm.202202-0393ED
- Esmail A et al. Am J Respir Crit Care Med. 2022;205(10):1214-1227. doi:10.1164/rccm.202107-1779OC
- WHO BPaLM Accelerator Platform: to support the call to action for implementation of the shorter and more effective treatment for all people suffering from drug-resistant TB. World Health Organization. Published May 9, 2023. Accessed June 26, 2023. https://www.who.int/news-room/events/detail/2023/05/09/default-calendar/who-bpalm-accelerator-platform–to-support-the-call-to-action-for-implementation-of-the-shorter-and-moreeffective-
- Trevisi L et al. Am J Respir Crit Care Med. 2023;207(11):1525-1532. doi:10.1164/rccm.202211-2125OC
- Domínguez J et al; TBnet and RESIST-TB networks. Lancet Infect Dis. 2023;23(4):e122-e137. doi:10.1016/S1473-3099(22)00875-1
- WHO operational handbook on tuberculosis: module 3: diagnosis: rapid diagnostics for tuberculosis detection, 2021 update. World Health Organization. Published July 7, 2021. Accessed June 26, 2023. https://www.who.int/publications/i/item/9789240030589treatment-for-all-people-suffering-from-drug-resistant-tb
CPAP adherence curbs severe cardiovascular disease outcomes
, based on data from more than 4,000 individuals.
Obstructive sleep apnea (OSA) is associated with an increased risk of cardiovascular diseases, but the association between management of OSA with a continuous positive-airway pressure device (CPAP) and major adverse cardiac or cerebrovascular events (MACCEs) remains unclear, wrote Manuel Sánchez-de-la-Torre, PhD, of the University of Lleida, Spain, and colleagues.
In a meta-analysis published in JAMA, the researchers reviewed data from 4,186 individuals with a mean age of 61.2 years; 82.1% were men. The study population included 2,097 patients who used CPAP and 2,089 who did not. The mean apnea-hypopnea index (AHI) was 31.2 events per hour, and OSA was defined as an oxygen desaturation index of 12 events or more per hour or an AHI of 15 events or more per hour. The composite primary outcome included the first MACCE, or death from cardiovascular causes, myocardial infarction, stroke, revascularization procedure, hospital admission for heart failure, hospital admission for unstable angina, or hospital admission for transient ischemic attack. Each of these components was a secondary endpoint.
Overall, the primary outcome of MACCE was similar for CPAP and non-CPAP using patients (hazard ratio, 1.01) with a total of 349 MACCE events in the CPAP group and 342 in the non-CPAP group. The mean adherence to CPAP was 3.1 hours per day. A total of 38.5% of patients in the CPAP group met the criteria for good adherence, defined as a mean of 4 or more hours per day.
However, as defined, good adherence to CPAP significantly reduced the risk of MACCE, compared with no CPAP use (HR, 0.69), and a sensitivity analysis showed a significant risk reduction, compared with patients who did not meet the criteria for good adherence (HR, 0.55; P = .005).
“Adherence to treatment is complex to determine and there are other potential factors that could affect patient adherence, such as health education, motivation, attitude, self-efficacy, psychosocial factors, and other health care system–related features,” the researchers wrote in their discussion.
The findings were limited by several factors including the evaluation only of CPAP as a treatment for OSA, and the inability to assess separate components of the composite endpoint, the researchers noted. Other limitations included the relatively small number of female patients, reliance mainly on at-home sleep apnea tests, and the potential for selection bias, they said.
However, the results suggest that CPAP adherence is important to prevention of secondary cardiovascular outcomes in OSA patients, and that implementation of specific and personalized strategies to improve adherence to treatment should be a clinical priority, they concluded.
The study was funded by the Instituto de Salud Carlos III, the European Union and FEDER, IRBLleida–Fundació Dr Pifarré, SEPAR, ResMed Ltd. (Australia), Associació Lleidatana de Respiratori, and CIBERES. Dr Sánchez-de-la-Torre also disclosed financial support from a Ramón y Cajal grant.
, based on data from more than 4,000 individuals.
Obstructive sleep apnea (OSA) is associated with an increased risk of cardiovascular diseases, but the association between management of OSA with a continuous positive-airway pressure device (CPAP) and major adverse cardiac or cerebrovascular events (MACCEs) remains unclear, wrote Manuel Sánchez-de-la-Torre, PhD, of the University of Lleida, Spain, and colleagues.
In a meta-analysis published in JAMA, the researchers reviewed data from 4,186 individuals with a mean age of 61.2 years; 82.1% were men. The study population included 2,097 patients who used CPAP and 2,089 who did not. The mean apnea-hypopnea index (AHI) was 31.2 events per hour, and OSA was defined as an oxygen desaturation index of 12 events or more per hour or an AHI of 15 events or more per hour. The composite primary outcome included the first MACCE, or death from cardiovascular causes, myocardial infarction, stroke, revascularization procedure, hospital admission for heart failure, hospital admission for unstable angina, or hospital admission for transient ischemic attack. Each of these components was a secondary endpoint.
Overall, the primary outcome of MACCE was similar for CPAP and non-CPAP using patients (hazard ratio, 1.01) with a total of 349 MACCE events in the CPAP group and 342 in the non-CPAP group. The mean adherence to CPAP was 3.1 hours per day. A total of 38.5% of patients in the CPAP group met the criteria for good adherence, defined as a mean of 4 or more hours per day.
However, as defined, good adherence to CPAP significantly reduced the risk of MACCE, compared with no CPAP use (HR, 0.69), and a sensitivity analysis showed a significant risk reduction, compared with patients who did not meet the criteria for good adherence (HR, 0.55; P = .005).
“Adherence to treatment is complex to determine and there are other potential factors that could affect patient adherence, such as health education, motivation, attitude, self-efficacy, psychosocial factors, and other health care system–related features,” the researchers wrote in their discussion.
The findings were limited by several factors including the evaluation only of CPAP as a treatment for OSA, and the inability to assess separate components of the composite endpoint, the researchers noted. Other limitations included the relatively small number of female patients, reliance mainly on at-home sleep apnea tests, and the potential for selection bias, they said.
However, the results suggest that CPAP adherence is important to prevention of secondary cardiovascular outcomes in OSA patients, and that implementation of specific and personalized strategies to improve adherence to treatment should be a clinical priority, they concluded.
The study was funded by the Instituto de Salud Carlos III, the European Union and FEDER, IRBLleida–Fundació Dr Pifarré, SEPAR, ResMed Ltd. (Australia), Associació Lleidatana de Respiratori, and CIBERES. Dr Sánchez-de-la-Torre also disclosed financial support from a Ramón y Cajal grant.
, based on data from more than 4,000 individuals.
Obstructive sleep apnea (OSA) is associated with an increased risk of cardiovascular diseases, but the association between management of OSA with a continuous positive-airway pressure device (CPAP) and major adverse cardiac or cerebrovascular events (MACCEs) remains unclear, wrote Manuel Sánchez-de-la-Torre, PhD, of the University of Lleida, Spain, and colleagues.
In a meta-analysis published in JAMA, the researchers reviewed data from 4,186 individuals with a mean age of 61.2 years; 82.1% were men. The study population included 2,097 patients who used CPAP and 2,089 who did not. The mean apnea-hypopnea index (AHI) was 31.2 events per hour, and OSA was defined as an oxygen desaturation index of 12 events or more per hour or an AHI of 15 events or more per hour. The composite primary outcome included the first MACCE, or death from cardiovascular causes, myocardial infarction, stroke, revascularization procedure, hospital admission for heart failure, hospital admission for unstable angina, or hospital admission for transient ischemic attack. Each of these components was a secondary endpoint.
Overall, the primary outcome of MACCE was similar for CPAP and non-CPAP using patients (hazard ratio, 1.01) with a total of 349 MACCE events in the CPAP group and 342 in the non-CPAP group. The mean adherence to CPAP was 3.1 hours per day. A total of 38.5% of patients in the CPAP group met the criteria for good adherence, defined as a mean of 4 or more hours per day.
However, as defined, good adherence to CPAP significantly reduced the risk of MACCE, compared with no CPAP use (HR, 0.69), and a sensitivity analysis showed a significant risk reduction, compared with patients who did not meet the criteria for good adherence (HR, 0.55; P = .005).
“Adherence to treatment is complex to determine and there are other potential factors that could affect patient adherence, such as health education, motivation, attitude, self-efficacy, psychosocial factors, and other health care system–related features,” the researchers wrote in their discussion.
The findings were limited by several factors including the evaluation only of CPAP as a treatment for OSA, and the inability to assess separate components of the composite endpoint, the researchers noted. Other limitations included the relatively small number of female patients, reliance mainly on at-home sleep apnea tests, and the potential for selection bias, they said.
However, the results suggest that CPAP adherence is important to prevention of secondary cardiovascular outcomes in OSA patients, and that implementation of specific and personalized strategies to improve adherence to treatment should be a clinical priority, they concluded.
The study was funded by the Instituto de Salud Carlos III, the European Union and FEDER, IRBLleida–Fundació Dr Pifarré, SEPAR, ResMed Ltd. (Australia), Associació Lleidatana de Respiratori, and CIBERES. Dr Sánchez-de-la-Torre also disclosed financial support from a Ramón y Cajal grant.
FROM JAMA
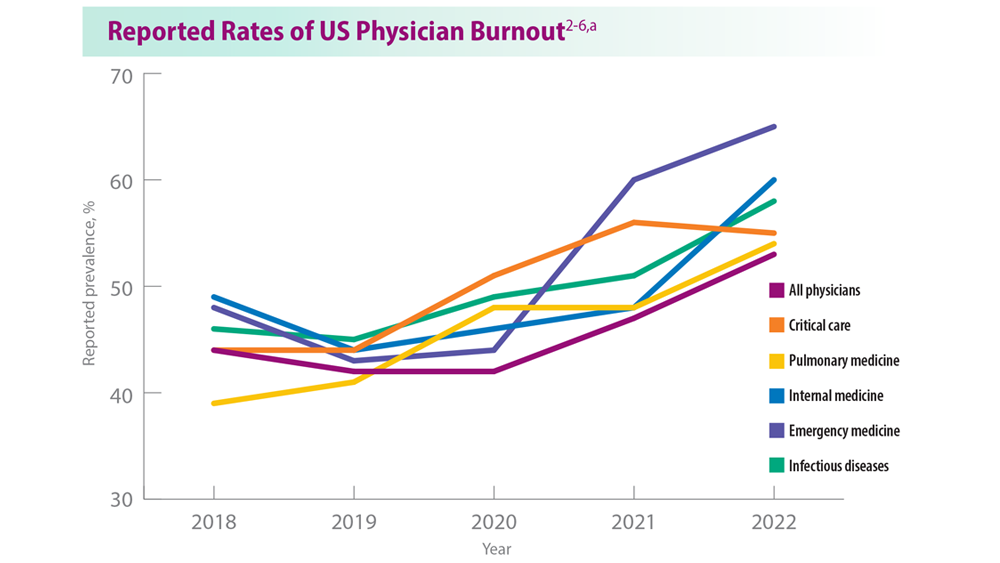
Addressing Physician Burnout in Pulmonology and Critical Care
- Moss M et al. Crit Care Med. 2016;44(7):1414-1421. doi:10.1097/CCM.000000000000188
- Medscape National Physician Burnout, Depression & Suicide Report 2019. Medscape. January 16, 2019. Accessed June 22, 2023. https://www.medscape.com/slideshow/2019-lifestyle-burnout-depression-6011056#1
- Medscape National Physician Burnout & Suicide Report 2020: The Generational Divide. Medscape. January 15, 2020. Accessed June 22, 2023. https://www.medscape.com/slideshow/2020-lifestyle-burnout-6012460#1
- ‘Death by 1000 Cuts’: Medscape National Physician Burnout & Suicide Report 2021. Medscape. January 22, 2021. Accessed June 22, 2023. https://www.medscape.com/slideshow/2021-lifestyle-burnout-6013456#2
- Physician Burnout Report 2022: Stress, Anxiety, and Anger. Medscape. January 21, 2022. Accessed June 22, 2023. https://www.medscape.com/slideshow/2022-lifestyle-burnout-6014664#1
- ‘I Cry but No One Cares’: Physician Burnout & Depression Report 2023. Medscape. January 27, 2023. Accessed June 22, 2023. https://www.medscape.com/slideshow/2023-lifestyle-burnout-6016058#1
- Murthy VH. N Engl J Med. 2022;387(7):577-579. doi:10.1056/NEJMp2207252
- Vranas KC et al. Chest. 2021;160(5):1714-1728. doi:10.1016/j.chest.2021.05.041
- Kerlin MP et al. Ann Am Thorac Soc. 2022;19(2):329-331. doi:10.1513/AnnalsATS.202105-567RL
- Dean W et al. Fed Pract. 2019;36(9):400-402. PMID: 31571807
- Association of American Medical Colleges. The Complexities of Physician Supply and Demand: Projections from 2019 to 2034. June 2021. https://www.aamc.org/media/54681/download?attachment
- Medscape Pulmonologist Lifestyle, Happiness & Burnout Report 2023: Contentment Amid Stress. February 24, 2023. Accessed June 28, 2023. https://www.medscape.com/slideshow/2023-lifestyle-pulmonologist-6016092#1
- Medscape Intensivist Lifestyle, Happiness & Burnout Report 2023: Contentment Amid Stress. February 24, 2023. Accessed June 28, 2023. https://www.medscape.com/slideshow/2023-lifestyle-intensivist-6016072#1
- Moss M et al. Crit Care Med. 2016;44(7):1414-1421. doi:10.1097/CCM.000000000000188
- Medscape National Physician Burnout, Depression & Suicide Report 2019. Medscape. January 16, 2019. Accessed June 22, 2023. https://www.medscape.com/slideshow/2019-lifestyle-burnout-depression-6011056#1
- Medscape National Physician Burnout & Suicide Report 2020: The Generational Divide. Medscape. January 15, 2020. Accessed June 22, 2023. https://www.medscape.com/slideshow/2020-lifestyle-burnout-6012460#1
- ‘Death by 1000 Cuts’: Medscape National Physician Burnout & Suicide Report 2021. Medscape. January 22, 2021. Accessed June 22, 2023. https://www.medscape.com/slideshow/2021-lifestyle-burnout-6013456#2
- Physician Burnout Report 2022: Stress, Anxiety, and Anger. Medscape. January 21, 2022. Accessed June 22, 2023. https://www.medscape.com/slideshow/2022-lifestyle-burnout-6014664#1
- ‘I Cry but No One Cares’: Physician Burnout & Depression Report 2023. Medscape. January 27, 2023. Accessed June 22, 2023. https://www.medscape.com/slideshow/2023-lifestyle-burnout-6016058#1
- Murthy VH. N Engl J Med. 2022;387(7):577-579. doi:10.1056/NEJMp2207252
- Vranas KC et al. Chest. 2021;160(5):1714-1728. doi:10.1016/j.chest.2021.05.041
- Kerlin MP et al. Ann Am Thorac Soc. 2022;19(2):329-331. doi:10.1513/AnnalsATS.202105-567RL
- Dean W et al. Fed Pract. 2019;36(9):400-402. PMID: 31571807
- Association of American Medical Colleges. The Complexities of Physician Supply and Demand: Projections from 2019 to 2034. June 2021. https://www.aamc.org/media/54681/download?attachment
- Medscape Pulmonologist Lifestyle, Happiness & Burnout Report 2023: Contentment Amid Stress. February 24, 2023. Accessed June 28, 2023. https://www.medscape.com/slideshow/2023-lifestyle-pulmonologist-6016092#1
- Medscape Intensivist Lifestyle, Happiness & Burnout Report 2023: Contentment Amid Stress. February 24, 2023. Accessed June 28, 2023. https://www.medscape.com/slideshow/2023-lifestyle-intensivist-6016072#1
- Moss M et al. Crit Care Med. 2016;44(7):1414-1421. doi:10.1097/CCM.000000000000188
- Medscape National Physician Burnout, Depression & Suicide Report 2019. Medscape. January 16, 2019. Accessed June 22, 2023. https://www.medscape.com/slideshow/2019-lifestyle-burnout-depression-6011056#1
- Medscape National Physician Burnout & Suicide Report 2020: The Generational Divide. Medscape. January 15, 2020. Accessed June 22, 2023. https://www.medscape.com/slideshow/2020-lifestyle-burnout-6012460#1
- ‘Death by 1000 Cuts’: Medscape National Physician Burnout & Suicide Report 2021. Medscape. January 22, 2021. Accessed June 22, 2023. https://www.medscape.com/slideshow/2021-lifestyle-burnout-6013456#2
- Physician Burnout Report 2022: Stress, Anxiety, and Anger. Medscape. January 21, 2022. Accessed June 22, 2023. https://www.medscape.com/slideshow/2022-lifestyle-burnout-6014664#1
- ‘I Cry but No One Cares’: Physician Burnout & Depression Report 2023. Medscape. January 27, 2023. Accessed June 22, 2023. https://www.medscape.com/slideshow/2023-lifestyle-burnout-6016058#1
- Murthy VH. N Engl J Med. 2022;387(7):577-579. doi:10.1056/NEJMp2207252
- Vranas KC et al. Chest. 2021;160(5):1714-1728. doi:10.1016/j.chest.2021.05.041
- Kerlin MP et al. Ann Am Thorac Soc. 2022;19(2):329-331. doi:10.1513/AnnalsATS.202105-567RL
- Dean W et al. Fed Pract. 2019;36(9):400-402. PMID: 31571807
- Association of American Medical Colleges. The Complexities of Physician Supply and Demand: Projections from 2019 to 2034. June 2021. https://www.aamc.org/media/54681/download?attachment
- Medscape Pulmonologist Lifestyle, Happiness & Burnout Report 2023: Contentment Amid Stress. February 24, 2023. Accessed June 28, 2023. https://www.medscape.com/slideshow/2023-lifestyle-pulmonologist-6016092#1
- Medscape Intensivist Lifestyle, Happiness & Burnout Report 2023: Contentment Amid Stress. February 24, 2023. Accessed June 28, 2023. https://www.medscape.com/slideshow/2023-lifestyle-intensivist-6016072#1
Reducing cognitive impairment from SCLC brain metastases
For patients with up to 10 brain metastases from small cell lung cancer (SCLC), stereotactic radiosurgery was associated with less cognitive impairment than whole-brain radiation therapy (WBRT) without compromising overall survival, results of the randomized ENCEPHALON (ARO 2018-9) trial suggest.
Among 56 patients with one to 10 SCLC brain metastases, 24% of those who received WBRT demonstrated significant declines in memory function 3 months after treatment, compared with 7% of patients whose metastases were treated with stereotactic radiosurgery alone. Preliminary data showed no significant differences in overall survival between the treatment groups at 6 months of follow-up, Denise Bernhardt, MD, from the Technical University of Munich, reported at the American Society of Radiation Oncology (ASTRO) annual meeting.
“We propose stereotactic radiosurgery should be an option for patients with up to 10 brain metastases in small cell lung cancer,” Dr. Bernhardt said during her presentation.
Vinai Gondi, MD, who was not involved in the study, said that the primary results from the trial – while limited by the study’s small size and missing data – are notable.
Patients with brain metastases from most cancer types typically receive stereotactic radiosurgery but WBRT has remained the standard of care to control brain metastases among patients with SCLC.
“This is the first prospective trial of radiosurgery versus whole-brain radiotherapy for small cell lung cancer brain metastases, and it’s important to recognize how important this is,” said Dr. Gondi, director of Radiation Oncology and codirector of the Brain Tumor Center at Northwestern Medicine Cancer Center, Warrenville, Ill.
Prior trials that have asked the same question did not include SCLC because many of those patients received prophylactic cranial irradiation, Dr. Gondi explained. Prophylactic cranial irradiation, however, has been on the decline among patients with brain metastases from SCLC, following a study from Japan showing no difference in survival among those who received the therapy and those followed with observation as well as evidence demonstrating significant toxicities associated with the technique.
Now “with the declining use of prophylactic cranial irradiation, the emergence of brain metastases is increasing significantly in volume in the small cell lung cancer population,” said Dr. Gondi, who is principal investigator on a phase 3 trial exploring stereotactic radiosurgery versus WBRT in a similar patient population.
In a previous retrospective trial), Dr. Bernhardt and colleagues found that first-line stereotactic radiosurgery did not compromise survival, compared with WBRT, but patients receiving stereotactic radiosurgery did have a higher risk for intracranial failure.
In the current study, the investigators compared the neurocognitive responses in patients with brain metastases from SCLC treated with stereotactic radiosurgery or WBRT.
Enrolled patients had histologically confirmed extensive disease with up to 10 metastatic brain lesions and had not previously received either therapeutic or prophylactic brain irradiation. After stratifying patients by synchronous versus metachronous disease, 56 patients were randomly assigned to either WBRT, at a total dose of 30 Gy delivered in 10 fractions, or to stereotactic radiosurgery with 20 Gy, 18 Gy, or fractionated stereotactic radiosurgery with 30 Gy in 5 Gy fractions for lesions larger than 3 cm.
The primary endpoint was neurocognition after radiation therapy as defined by a decline from baseline of at least five points on the Hopkins Verbal Learning Test-Revised (HVLT-R) total recall subscale at 3 months. Secondary endpoints included survival outcomes, additional neurocognitive assessments of motor skills, executive function, attention, memory, and processing as well as quality-of-life measures.
The investigators expected a high rate of study dropout and planned their statistical analysis accordingly, using a method for estimating the likely values of missing data based on observed data.
Among 26 patients who eventually underwent stereotactic radiosurgery, 18 did not meet the primary endpoint and 2 (7%) demonstrated declines on the HVLT-R subscale of 5 or more points. Data for the remaining 6 patients were missing.
Among the 25 who underwent WBRT, 13 did not meet the primary endpoint and 6 (24%) demonstrated declines of at least 5 points. Data for 6 of the remaining patients were missing.
Although more patients in the WBRT arm had significant declines in neurocognitive function, the difference between the groups was not significant, due to the high proportion of study dropouts – approximately one-fourth of patients in each arm. But the analysis suggested that the neuroprotective effect of stereotactic radiosurgery was notable, Dr. Bernhardt said.
At 6 months, the team also found no significant difference in the survival probability between the treatment groups (P = .36). The median time to death was 124 days among patients who received stereotactic radiosurgery and 131 days among patients who received WBRT.
Dr. Gondi said the data from ENCEPHALON, while promising, need to be carefully scrutinized because of the small sample sizes and the possibility for unintended bias.
ARO 2018-9 is an investigator-initiated trial funded by Accuray. Dr. Bernhardt disclosed consulting actives, fees, travel expenses, and research funding from Accuray and others. Dr. Gondi disclosed honoraria from UpToDate.
A version of this article appeared on Medscape.com.
For patients with up to 10 brain metastases from small cell lung cancer (SCLC), stereotactic radiosurgery was associated with less cognitive impairment than whole-brain radiation therapy (WBRT) without compromising overall survival, results of the randomized ENCEPHALON (ARO 2018-9) trial suggest.
Among 56 patients with one to 10 SCLC brain metastases, 24% of those who received WBRT demonstrated significant declines in memory function 3 months after treatment, compared with 7% of patients whose metastases were treated with stereotactic radiosurgery alone. Preliminary data showed no significant differences in overall survival between the treatment groups at 6 months of follow-up, Denise Bernhardt, MD, from the Technical University of Munich, reported at the American Society of Radiation Oncology (ASTRO) annual meeting.
“We propose stereotactic radiosurgery should be an option for patients with up to 10 brain metastases in small cell lung cancer,” Dr. Bernhardt said during her presentation.
Vinai Gondi, MD, who was not involved in the study, said that the primary results from the trial – while limited by the study’s small size and missing data – are notable.
Patients with brain metastases from most cancer types typically receive stereotactic radiosurgery but WBRT has remained the standard of care to control brain metastases among patients with SCLC.
“This is the first prospective trial of radiosurgery versus whole-brain radiotherapy for small cell lung cancer brain metastases, and it’s important to recognize how important this is,” said Dr. Gondi, director of Radiation Oncology and codirector of the Brain Tumor Center at Northwestern Medicine Cancer Center, Warrenville, Ill.
Prior trials that have asked the same question did not include SCLC because many of those patients received prophylactic cranial irradiation, Dr. Gondi explained. Prophylactic cranial irradiation, however, has been on the decline among patients with brain metastases from SCLC, following a study from Japan showing no difference in survival among those who received the therapy and those followed with observation as well as evidence demonstrating significant toxicities associated with the technique.
Now “with the declining use of prophylactic cranial irradiation, the emergence of brain metastases is increasing significantly in volume in the small cell lung cancer population,” said Dr. Gondi, who is principal investigator on a phase 3 trial exploring stereotactic radiosurgery versus WBRT in a similar patient population.
In a previous retrospective trial), Dr. Bernhardt and colleagues found that first-line stereotactic radiosurgery did not compromise survival, compared with WBRT, but patients receiving stereotactic radiosurgery did have a higher risk for intracranial failure.
In the current study, the investigators compared the neurocognitive responses in patients with brain metastases from SCLC treated with stereotactic radiosurgery or WBRT.
Enrolled patients had histologically confirmed extensive disease with up to 10 metastatic brain lesions and had not previously received either therapeutic or prophylactic brain irradiation. After stratifying patients by synchronous versus metachronous disease, 56 patients were randomly assigned to either WBRT, at a total dose of 30 Gy delivered in 10 fractions, or to stereotactic radiosurgery with 20 Gy, 18 Gy, or fractionated stereotactic radiosurgery with 30 Gy in 5 Gy fractions for lesions larger than 3 cm.
The primary endpoint was neurocognition after radiation therapy as defined by a decline from baseline of at least five points on the Hopkins Verbal Learning Test-Revised (HVLT-R) total recall subscale at 3 months. Secondary endpoints included survival outcomes, additional neurocognitive assessments of motor skills, executive function, attention, memory, and processing as well as quality-of-life measures.
The investigators expected a high rate of study dropout and planned their statistical analysis accordingly, using a method for estimating the likely values of missing data based on observed data.
Among 26 patients who eventually underwent stereotactic radiosurgery, 18 did not meet the primary endpoint and 2 (7%) demonstrated declines on the HVLT-R subscale of 5 or more points. Data for the remaining 6 patients were missing.
Among the 25 who underwent WBRT, 13 did not meet the primary endpoint and 6 (24%) demonstrated declines of at least 5 points. Data for 6 of the remaining patients were missing.
Although more patients in the WBRT arm had significant declines in neurocognitive function, the difference between the groups was not significant, due to the high proportion of study dropouts – approximately one-fourth of patients in each arm. But the analysis suggested that the neuroprotective effect of stereotactic radiosurgery was notable, Dr. Bernhardt said.
At 6 months, the team also found no significant difference in the survival probability between the treatment groups (P = .36). The median time to death was 124 days among patients who received stereotactic radiosurgery and 131 days among patients who received WBRT.
Dr. Gondi said the data from ENCEPHALON, while promising, need to be carefully scrutinized because of the small sample sizes and the possibility for unintended bias.
ARO 2018-9 is an investigator-initiated trial funded by Accuray. Dr. Bernhardt disclosed consulting actives, fees, travel expenses, and research funding from Accuray and others. Dr. Gondi disclosed honoraria from UpToDate.
A version of this article appeared on Medscape.com.
For patients with up to 10 brain metastases from small cell lung cancer (SCLC), stereotactic radiosurgery was associated with less cognitive impairment than whole-brain radiation therapy (WBRT) without compromising overall survival, results of the randomized ENCEPHALON (ARO 2018-9) trial suggest.
Among 56 patients with one to 10 SCLC brain metastases, 24% of those who received WBRT demonstrated significant declines in memory function 3 months after treatment, compared with 7% of patients whose metastases were treated with stereotactic radiosurgery alone. Preliminary data showed no significant differences in overall survival between the treatment groups at 6 months of follow-up, Denise Bernhardt, MD, from the Technical University of Munich, reported at the American Society of Radiation Oncology (ASTRO) annual meeting.
“We propose stereotactic radiosurgery should be an option for patients with up to 10 brain metastases in small cell lung cancer,” Dr. Bernhardt said during her presentation.
Vinai Gondi, MD, who was not involved in the study, said that the primary results from the trial – while limited by the study’s small size and missing data – are notable.
Patients with brain metastases from most cancer types typically receive stereotactic radiosurgery but WBRT has remained the standard of care to control brain metastases among patients with SCLC.
“This is the first prospective trial of radiosurgery versus whole-brain radiotherapy for small cell lung cancer brain metastases, and it’s important to recognize how important this is,” said Dr. Gondi, director of Radiation Oncology and codirector of the Brain Tumor Center at Northwestern Medicine Cancer Center, Warrenville, Ill.
Prior trials that have asked the same question did not include SCLC because many of those patients received prophylactic cranial irradiation, Dr. Gondi explained. Prophylactic cranial irradiation, however, has been on the decline among patients with brain metastases from SCLC, following a study from Japan showing no difference in survival among those who received the therapy and those followed with observation as well as evidence demonstrating significant toxicities associated with the technique.
Now “with the declining use of prophylactic cranial irradiation, the emergence of brain metastases is increasing significantly in volume in the small cell lung cancer population,” said Dr. Gondi, who is principal investigator on a phase 3 trial exploring stereotactic radiosurgery versus WBRT in a similar patient population.
In a previous retrospective trial), Dr. Bernhardt and colleagues found that first-line stereotactic radiosurgery did not compromise survival, compared with WBRT, but patients receiving stereotactic radiosurgery did have a higher risk for intracranial failure.
In the current study, the investigators compared the neurocognitive responses in patients with brain metastases from SCLC treated with stereotactic radiosurgery or WBRT.
Enrolled patients had histologically confirmed extensive disease with up to 10 metastatic brain lesions and had not previously received either therapeutic or prophylactic brain irradiation. After stratifying patients by synchronous versus metachronous disease, 56 patients were randomly assigned to either WBRT, at a total dose of 30 Gy delivered in 10 fractions, or to stereotactic radiosurgery with 20 Gy, 18 Gy, or fractionated stereotactic radiosurgery with 30 Gy in 5 Gy fractions for lesions larger than 3 cm.
The primary endpoint was neurocognition after radiation therapy as defined by a decline from baseline of at least five points on the Hopkins Verbal Learning Test-Revised (HVLT-R) total recall subscale at 3 months. Secondary endpoints included survival outcomes, additional neurocognitive assessments of motor skills, executive function, attention, memory, and processing as well as quality-of-life measures.
The investigators expected a high rate of study dropout and planned their statistical analysis accordingly, using a method for estimating the likely values of missing data based on observed data.
Among 26 patients who eventually underwent stereotactic radiosurgery, 18 did not meet the primary endpoint and 2 (7%) demonstrated declines on the HVLT-R subscale of 5 or more points. Data for the remaining 6 patients were missing.
Among the 25 who underwent WBRT, 13 did not meet the primary endpoint and 6 (24%) demonstrated declines of at least 5 points. Data for 6 of the remaining patients were missing.
Although more patients in the WBRT arm had significant declines in neurocognitive function, the difference between the groups was not significant, due to the high proportion of study dropouts – approximately one-fourth of patients in each arm. But the analysis suggested that the neuroprotective effect of stereotactic radiosurgery was notable, Dr. Bernhardt said.
At 6 months, the team also found no significant difference in the survival probability between the treatment groups (P = .36). The median time to death was 124 days among patients who received stereotactic radiosurgery and 131 days among patients who received WBRT.
Dr. Gondi said the data from ENCEPHALON, while promising, need to be carefully scrutinized because of the small sample sizes and the possibility for unintended bias.
ARO 2018-9 is an investigator-initiated trial funded by Accuray. Dr. Bernhardt disclosed consulting actives, fees, travel expenses, and research funding from Accuray and others. Dr. Gondi disclosed honoraria from UpToDate.
A version of this article appeared on Medscape.com.
Hormone therapy less effective in menopausal women with obesity
PHILADELPHIA – , according to a small, retrospective study presented at the annual meeting of the Menopause Society (formerly the North American Menopause Society).
More than 40% of women over age 40 in the United States have obesity, presenter Anita Pershad, MD, an ob.gyn. medical resident at Eastern Virginia Medical School, Norfolk, told attendees. Yet most of the large-scale studies investigating perimenopausal and postmenopausal hormone therapy included participants without major medical comorbidities, so little data exist on how effectively HT works in women with these comorbidities, she said
“The main takeaway of our study is that obesity may worsen a woman’s menopausal symptoms and limit the amount of relief she gets from hormone therapy,” Dr. Pershad said in an interview. “It remains unclear if hormone therapy is less effective in women with obesity overall, or if the expected efficacy can be achieved with alternative design and administration routes. A potential mechanism of action for the observed decreased effect could be due to adipose tissue acting as a heat insulator, promoting the effects of vasomotor symptoms.”
Dr. Pershad and her colleagues conducted a retrospective review of the medical records of 119 patients who presented to a menopause clinic at a Midsouth urban academic medical center between July 2018 and December 2022. Obesity was defined as having a body mass index (BMI) of 30 kg/m2 or greater.
The patients with and without obesity were similar in terms of age, duration of menopause, use of hormone therapy, and therapy acceptance, but patients with obesity were more likely to identify themselves as Black (71% vs. 40%). Women with obesity were also significantly more likely than women without obesity to report vasomotor symptoms (74% vs. 45%, P = .002), genitourinary/vulvovaginal symptoms (60% vs. 21%, P < .001), mood disturbances (11% vs. 0%, P = .18), and decreased libido (29% vs. 11%, P = .017).
There were no significant differences in comorbidities between women with and without obesity, and among women who received systemic or localized HT, the same standard dosing was used for both groups.
Women with obesity were much less likely to see a satisfying reduction in their menopausal symptoms than women without obesity (odds ratio 0.07, 95% confidence interval, 0.01-0.64; P = .006), though the subgroups for each category of HT were small. Among the 20 women receiving systemic hormone therapy, only 1 of the 12 with obesity (8.3%) reported improvement in symptoms, compared with 7 of the 8 women without obesity (88%; P = .0004). Among 33 women using localized hormone therapy, 46% of the 24 women with obesity vs. 89% of the 9 women without obesity experienced symptom improvement (P = .026).
The proportions of women reporting relief from only lifestyle modifications or from nonhormonal medications, such as SSRIs/SNRIs, trazodone, and clonidine, were not statistically different. There were 33 women who relied only on lifestyle modifications, with 31% of the 16 women with obesity and 59% of the 17 women without obesity reporting improvement in their symptoms (P = .112). Similarly, among the 33 women using nonhormonal medications, 75% of the 20 women with obesity and 77% of the 13 women without obesity experienced relief (P = .9).
Women with obesity are undertreated
Dr. Pershad emphasized the need to improve care and counseling for diverse patients seeking treatment for menopausal symptoms.
“More research is needed to examine how women with medical comorbidities are uniquely impacted by menopause and respond to therapies,” Dr. Pershad said in an interview. “This can be achieved by actively including more diverse patient populations in women’s health studies, burdened by the social determinants of health and medical comorbidities such as obesity.”
Stephanie S. Faubion, MD, MBA, director for Mayo Clinic’s Center for Women’s Health, Rochester, Minn., and medical director for The Menopause Society, was not surprised by the findings, particularly given that women with obesity tend to have more hot flashes and night sweats as a result of their extra weight. However, dosage data was not adjusted for BMI in the study and data on hormone levels was unavailable, she said, so it’s difficult to determine from the data whether HT was less effective for women with obesity or whether they were underdosed.
“I think women with obesity are undertreated,” Dr. Faubion said in an interview. “My guess is people are afraid. Women with obesity also may have other comorbidities,” such as hypertension and diabetes, she said, and “the greater the number of cardiovascular risk factors, the higher risk hormone therapy is.” Providers may therefore be leery of prescribing HT or prescribing it at an appropriately high enough dose to treat menopausal symptoms.
Common practice is to start patients at the lowest dose and titrate up according to symptoms, but “if people are afraid of it, they’re going to start the lowest dose” and may not increase it, Dr. Faubion said. She noted that other nonhormonal options are available, though providers should be conscientious about selecting ones whose adverse events do not include weight gain.
Although the study focused on an understudied population within hormone therapy research, the study was limited by its small size, low overall use of hormone therapy, recall bias, and the researchers’ inability to control for other medications the participants may have been taking.
Dr. Pershad said she is continuing research to try to identify the mechanisms underlying the reduced efficacy in women with obesity.
The research did not use any external funding. Dr. Pershad had no industry disclosures, but her colleagues reported honoraria from or speaking for TherapeuticsMD, Astella Pharma, Scynexis, Pharmavite, and Pfizer. Dr. Faubion had no disclosures.
PHILADELPHIA – , according to a small, retrospective study presented at the annual meeting of the Menopause Society (formerly the North American Menopause Society).
More than 40% of women over age 40 in the United States have obesity, presenter Anita Pershad, MD, an ob.gyn. medical resident at Eastern Virginia Medical School, Norfolk, told attendees. Yet most of the large-scale studies investigating perimenopausal and postmenopausal hormone therapy included participants without major medical comorbidities, so little data exist on how effectively HT works in women with these comorbidities, she said
“The main takeaway of our study is that obesity may worsen a woman’s menopausal symptoms and limit the amount of relief she gets from hormone therapy,” Dr. Pershad said in an interview. “It remains unclear if hormone therapy is less effective in women with obesity overall, or if the expected efficacy can be achieved with alternative design and administration routes. A potential mechanism of action for the observed decreased effect could be due to adipose tissue acting as a heat insulator, promoting the effects of vasomotor symptoms.”
Dr. Pershad and her colleagues conducted a retrospective review of the medical records of 119 patients who presented to a menopause clinic at a Midsouth urban academic medical center between July 2018 and December 2022. Obesity was defined as having a body mass index (BMI) of 30 kg/m2 or greater.
The patients with and without obesity were similar in terms of age, duration of menopause, use of hormone therapy, and therapy acceptance, but patients with obesity were more likely to identify themselves as Black (71% vs. 40%). Women with obesity were also significantly more likely than women without obesity to report vasomotor symptoms (74% vs. 45%, P = .002), genitourinary/vulvovaginal symptoms (60% vs. 21%, P < .001), mood disturbances (11% vs. 0%, P = .18), and decreased libido (29% vs. 11%, P = .017).
There were no significant differences in comorbidities between women with and without obesity, and among women who received systemic or localized HT, the same standard dosing was used for both groups.
Women with obesity were much less likely to see a satisfying reduction in their menopausal symptoms than women without obesity (odds ratio 0.07, 95% confidence interval, 0.01-0.64; P = .006), though the subgroups for each category of HT were small. Among the 20 women receiving systemic hormone therapy, only 1 of the 12 with obesity (8.3%) reported improvement in symptoms, compared with 7 of the 8 women without obesity (88%; P = .0004). Among 33 women using localized hormone therapy, 46% of the 24 women with obesity vs. 89% of the 9 women without obesity experienced symptom improvement (P = .026).
The proportions of women reporting relief from only lifestyle modifications or from nonhormonal medications, such as SSRIs/SNRIs, trazodone, and clonidine, were not statistically different. There were 33 women who relied only on lifestyle modifications, with 31% of the 16 women with obesity and 59% of the 17 women without obesity reporting improvement in their symptoms (P = .112). Similarly, among the 33 women using nonhormonal medications, 75% of the 20 women with obesity and 77% of the 13 women without obesity experienced relief (P = .9).
Women with obesity are undertreated
Dr. Pershad emphasized the need to improve care and counseling for diverse patients seeking treatment for menopausal symptoms.
“More research is needed to examine how women with medical comorbidities are uniquely impacted by menopause and respond to therapies,” Dr. Pershad said in an interview. “This can be achieved by actively including more diverse patient populations in women’s health studies, burdened by the social determinants of health and medical comorbidities such as obesity.”
Stephanie S. Faubion, MD, MBA, director for Mayo Clinic’s Center for Women’s Health, Rochester, Minn., and medical director for The Menopause Society, was not surprised by the findings, particularly given that women with obesity tend to have more hot flashes and night sweats as a result of their extra weight. However, dosage data was not adjusted for BMI in the study and data on hormone levels was unavailable, she said, so it’s difficult to determine from the data whether HT was less effective for women with obesity or whether they were underdosed.
“I think women with obesity are undertreated,” Dr. Faubion said in an interview. “My guess is people are afraid. Women with obesity also may have other comorbidities,” such as hypertension and diabetes, she said, and “the greater the number of cardiovascular risk factors, the higher risk hormone therapy is.” Providers may therefore be leery of prescribing HT or prescribing it at an appropriately high enough dose to treat menopausal symptoms.
Common practice is to start patients at the lowest dose and titrate up according to symptoms, but “if people are afraid of it, they’re going to start the lowest dose” and may not increase it, Dr. Faubion said. She noted that other nonhormonal options are available, though providers should be conscientious about selecting ones whose adverse events do not include weight gain.
Although the study focused on an understudied population within hormone therapy research, the study was limited by its small size, low overall use of hormone therapy, recall bias, and the researchers’ inability to control for other medications the participants may have been taking.
Dr. Pershad said she is continuing research to try to identify the mechanisms underlying the reduced efficacy in women with obesity.
The research did not use any external funding. Dr. Pershad had no industry disclosures, but her colleagues reported honoraria from or speaking for TherapeuticsMD, Astella Pharma, Scynexis, Pharmavite, and Pfizer. Dr. Faubion had no disclosures.
PHILADELPHIA – , according to a small, retrospective study presented at the annual meeting of the Menopause Society (formerly the North American Menopause Society).
More than 40% of women over age 40 in the United States have obesity, presenter Anita Pershad, MD, an ob.gyn. medical resident at Eastern Virginia Medical School, Norfolk, told attendees. Yet most of the large-scale studies investigating perimenopausal and postmenopausal hormone therapy included participants without major medical comorbidities, so little data exist on how effectively HT works in women with these comorbidities, she said
“The main takeaway of our study is that obesity may worsen a woman’s menopausal symptoms and limit the amount of relief she gets from hormone therapy,” Dr. Pershad said in an interview. “It remains unclear if hormone therapy is less effective in women with obesity overall, or if the expected efficacy can be achieved with alternative design and administration routes. A potential mechanism of action for the observed decreased effect could be due to adipose tissue acting as a heat insulator, promoting the effects of vasomotor symptoms.”
Dr. Pershad and her colleagues conducted a retrospective review of the medical records of 119 patients who presented to a menopause clinic at a Midsouth urban academic medical center between July 2018 and December 2022. Obesity was defined as having a body mass index (BMI) of 30 kg/m2 or greater.
The patients with and without obesity were similar in terms of age, duration of menopause, use of hormone therapy, and therapy acceptance, but patients with obesity were more likely to identify themselves as Black (71% vs. 40%). Women with obesity were also significantly more likely than women without obesity to report vasomotor symptoms (74% vs. 45%, P = .002), genitourinary/vulvovaginal symptoms (60% vs. 21%, P < .001), mood disturbances (11% vs. 0%, P = .18), and decreased libido (29% vs. 11%, P = .017).
There were no significant differences in comorbidities between women with and without obesity, and among women who received systemic or localized HT, the same standard dosing was used for both groups.
Women with obesity were much less likely to see a satisfying reduction in their menopausal symptoms than women without obesity (odds ratio 0.07, 95% confidence interval, 0.01-0.64; P = .006), though the subgroups for each category of HT were small. Among the 20 women receiving systemic hormone therapy, only 1 of the 12 with obesity (8.3%) reported improvement in symptoms, compared with 7 of the 8 women without obesity (88%; P = .0004). Among 33 women using localized hormone therapy, 46% of the 24 women with obesity vs. 89% of the 9 women without obesity experienced symptom improvement (P = .026).
The proportions of women reporting relief from only lifestyle modifications or from nonhormonal medications, such as SSRIs/SNRIs, trazodone, and clonidine, were not statistically different. There were 33 women who relied only on lifestyle modifications, with 31% of the 16 women with obesity and 59% of the 17 women without obesity reporting improvement in their symptoms (P = .112). Similarly, among the 33 women using nonhormonal medications, 75% of the 20 women with obesity and 77% of the 13 women without obesity experienced relief (P = .9).
Women with obesity are undertreated
Dr. Pershad emphasized the need to improve care and counseling for diverse patients seeking treatment for menopausal symptoms.
“More research is needed to examine how women with medical comorbidities are uniquely impacted by menopause and respond to therapies,” Dr. Pershad said in an interview. “This can be achieved by actively including more diverse patient populations in women’s health studies, burdened by the social determinants of health and medical comorbidities such as obesity.”
Stephanie S. Faubion, MD, MBA, director for Mayo Clinic’s Center for Women’s Health, Rochester, Minn., and medical director for The Menopause Society, was not surprised by the findings, particularly given that women with obesity tend to have more hot flashes and night sweats as a result of their extra weight. However, dosage data was not adjusted for BMI in the study and data on hormone levels was unavailable, she said, so it’s difficult to determine from the data whether HT was less effective for women with obesity or whether they were underdosed.
“I think women with obesity are undertreated,” Dr. Faubion said in an interview. “My guess is people are afraid. Women with obesity also may have other comorbidities,” such as hypertension and diabetes, she said, and “the greater the number of cardiovascular risk factors, the higher risk hormone therapy is.” Providers may therefore be leery of prescribing HT or prescribing it at an appropriately high enough dose to treat menopausal symptoms.
Common practice is to start patients at the lowest dose and titrate up according to symptoms, but “if people are afraid of it, they’re going to start the lowest dose” and may not increase it, Dr. Faubion said. She noted that other nonhormonal options are available, though providers should be conscientious about selecting ones whose adverse events do not include weight gain.
Although the study focused on an understudied population within hormone therapy research, the study was limited by its small size, low overall use of hormone therapy, recall bias, and the researchers’ inability to control for other medications the participants may have been taking.
Dr. Pershad said she is continuing research to try to identify the mechanisms underlying the reduced efficacy in women with obesity.
The research did not use any external funding. Dr. Pershad had no industry disclosures, but her colleagues reported honoraria from or speaking for TherapeuticsMD, Astella Pharma, Scynexis, Pharmavite, and Pfizer. Dr. Faubion had no disclosures.
AT THE MENOPAUSE SOCIETY ANNUAL MEETING
Short-course SBRT: Standard of care in prostate cancer?
SAN DIEGO – delivered at higher doses in 20-39 fractions, according to new data from the phase 3 randomized PACE-B trial.
Overall, the 5-year event-free survival rates were 95.8% among patients who received SBRT and 94.6% among those who had conventional radiation. The incidence of adverse events was also low in both groups, with no significant differences observed between the trial arms.
The similar event-free survival and toxicity profiles in both groups provide more support for SBRT, which treats prostate cancer with larger radiation fractions over a shorter time period.
“I think we can also say now with a high level of confidence that SBRT can be considered a new standard of care for low and favorable intermediate-risk prostate cancer,” said Nicholas van As, MD, MB, from the Royal Marsden NHS Foundation Trust and Institute of Cancer Research in London, who presented efficacy and safety results from the noninferiority trial at the American Society for Radiation Oncology (ASTRO) annual meeting. SBRT is more convenient for patients and more cost-effective for health care providers, Dr. Van As added.
Invited discussant Alejandro Berlin, MD, MSc, from Princess Margaret Cancer Centre and the University of Toronto, agreed “that this should be a standard of care for low and favorable intermediate-risk prostate cancer,” an option already endorsed by relevant guidelines.
But, Dr. Berlin noted, SBRT requires careful attention to technique to achieve the desired results. Further research will be needed to identify and potentially reduce variability among radiation oncology practice regarding margins, dosimetry goals, dose heterogeneity, treatment schedules, and other factors, he said.
An international trial
PACE-B is one of three branches of a multi-center collaboration among 37 radiation therapy centers in the United Kingdom, Ireland, and Canada.
In the trial, investigators enrolled 874 patients with T1c or T2c prostate cancer, Gleason score of 3+4 or less, prostate-specific antigen (PSA) level no higher than 20 ng/mL, MRI staging, and no prior androgen deprivation therapy. Investigators then randomly assigned them on a 1:1 basis to receive either conventional radiation (n = 441) or SBRT (n = 433).
At the start of the trial, patients who were assigned to the conventional radiation group received 78 Gy in 39 fractions over 4-8 weeks. However, after results from the CHHiP trial, which showed that a 60-Gy, 20-fraction regimen was not inferior to a 74-Gy, 37-fraction regimen, the PACE-B investigators modified the protocol to 62 Gy delivered in 20 fractions.
Patients assigned to SBRT received 36.25 Gy divided into give fractions delivered over 1-2 weeks, with 40 Gy to the clinical target volume.
The primary outcome was noninferiority of SBRT, measured as whether patients remained free of biochemical clinical failure. Biochemical clinical failure was defined as evidence that the cancer was returning, such as an increase in PSA levels or distant metastases or death from prostate cancer.
At a median follow-up of 73.1 months, 5-year event-free survival rates were 94.6% for patients who received conventional radiation therapy and 95.8% for patients who received SBRT, meeting the prespecified criteria for noninferiority of SBRT (P = .007).
Freedom from biochemical and clinical failure, the trial’s primary endpoint, “was significantly better on both arms than our original power calculation, where we expected control rates of approximately 85%,” Dr. Van As said in an ASTRO plenary session.
Toxicity rates were also low in both study arms. The rate of grade 2 or greater urogenital side effects at 5 years was 5.5% in the SBRT arm and 3.2% in the conventional therapy arm. Grade 2 or greater gastrointestinal side effects occurred in only two patients, one in each study arm.
Given the findings, “I think it’s now imperative that our surgeons discuss this data with their patients before they perform prostatectomies,” Dr. Van As said.
Neha Vapiwala, MD, president-elect of ASTRO who moderated a media briefing where Dr. Van As summarized the PACE-B data, commented that “this study was conducted very rigorously, with excellent quality assurance.”
The study also highlights that clinicians in the United States have considerable catching up to do, said Dr. Vapiwala, from the Hospital of the University of Pennsylvania, Philadelphia.
In the United States, “we are way behind our colleagues on the other side of the pond,” she said. “We are way behind in our uptake of ultra-hypofractionated radiation [such as SBRT], and I do believe that some of that comes from the lack of feeling comfortable with the techniques that are needed and the expertise that is needed.”
PACE-B was funded by Accuray. Dr. Van As disclosed research grants from the company and consulting fees from Varian. Dr. Berlin reported no conflict of interest relevant to the study. Dr. Vapiwala has disclosed a consulting or advisory role with Bayer.
A version of this article first appeared on Medscape.com.
SAN DIEGO – delivered at higher doses in 20-39 fractions, according to new data from the phase 3 randomized PACE-B trial.
Overall, the 5-year event-free survival rates were 95.8% among patients who received SBRT and 94.6% among those who had conventional radiation. The incidence of adverse events was also low in both groups, with no significant differences observed between the trial arms.
The similar event-free survival and toxicity profiles in both groups provide more support for SBRT, which treats prostate cancer with larger radiation fractions over a shorter time period.
“I think we can also say now with a high level of confidence that SBRT can be considered a new standard of care for low and favorable intermediate-risk prostate cancer,” said Nicholas van As, MD, MB, from the Royal Marsden NHS Foundation Trust and Institute of Cancer Research in London, who presented efficacy and safety results from the noninferiority trial at the American Society for Radiation Oncology (ASTRO) annual meeting. SBRT is more convenient for patients and more cost-effective for health care providers, Dr. Van As added.
Invited discussant Alejandro Berlin, MD, MSc, from Princess Margaret Cancer Centre and the University of Toronto, agreed “that this should be a standard of care for low and favorable intermediate-risk prostate cancer,” an option already endorsed by relevant guidelines.
But, Dr. Berlin noted, SBRT requires careful attention to technique to achieve the desired results. Further research will be needed to identify and potentially reduce variability among radiation oncology practice regarding margins, dosimetry goals, dose heterogeneity, treatment schedules, and other factors, he said.
An international trial
PACE-B is one of three branches of a multi-center collaboration among 37 radiation therapy centers in the United Kingdom, Ireland, and Canada.
In the trial, investigators enrolled 874 patients with T1c or T2c prostate cancer, Gleason score of 3+4 or less, prostate-specific antigen (PSA) level no higher than 20 ng/mL, MRI staging, and no prior androgen deprivation therapy. Investigators then randomly assigned them on a 1:1 basis to receive either conventional radiation (n = 441) or SBRT (n = 433).
At the start of the trial, patients who were assigned to the conventional radiation group received 78 Gy in 39 fractions over 4-8 weeks. However, after results from the CHHiP trial, which showed that a 60-Gy, 20-fraction regimen was not inferior to a 74-Gy, 37-fraction regimen, the PACE-B investigators modified the protocol to 62 Gy delivered in 20 fractions.
Patients assigned to SBRT received 36.25 Gy divided into give fractions delivered over 1-2 weeks, with 40 Gy to the clinical target volume.
The primary outcome was noninferiority of SBRT, measured as whether patients remained free of biochemical clinical failure. Biochemical clinical failure was defined as evidence that the cancer was returning, such as an increase in PSA levels or distant metastases or death from prostate cancer.
At a median follow-up of 73.1 months, 5-year event-free survival rates were 94.6% for patients who received conventional radiation therapy and 95.8% for patients who received SBRT, meeting the prespecified criteria for noninferiority of SBRT (P = .007).
Freedom from biochemical and clinical failure, the trial’s primary endpoint, “was significantly better on both arms than our original power calculation, where we expected control rates of approximately 85%,” Dr. Van As said in an ASTRO plenary session.
Toxicity rates were also low in both study arms. The rate of grade 2 or greater urogenital side effects at 5 years was 5.5% in the SBRT arm and 3.2% in the conventional therapy arm. Grade 2 or greater gastrointestinal side effects occurred in only two patients, one in each study arm.
Given the findings, “I think it’s now imperative that our surgeons discuss this data with their patients before they perform prostatectomies,” Dr. Van As said.
Neha Vapiwala, MD, president-elect of ASTRO who moderated a media briefing where Dr. Van As summarized the PACE-B data, commented that “this study was conducted very rigorously, with excellent quality assurance.”
The study also highlights that clinicians in the United States have considerable catching up to do, said Dr. Vapiwala, from the Hospital of the University of Pennsylvania, Philadelphia.
In the United States, “we are way behind our colleagues on the other side of the pond,” she said. “We are way behind in our uptake of ultra-hypofractionated radiation [such as SBRT], and I do believe that some of that comes from the lack of feeling comfortable with the techniques that are needed and the expertise that is needed.”
PACE-B was funded by Accuray. Dr. Van As disclosed research grants from the company and consulting fees from Varian. Dr. Berlin reported no conflict of interest relevant to the study. Dr. Vapiwala has disclosed a consulting or advisory role with Bayer.
A version of this article first appeared on Medscape.com.
SAN DIEGO – delivered at higher doses in 20-39 fractions, according to new data from the phase 3 randomized PACE-B trial.
Overall, the 5-year event-free survival rates were 95.8% among patients who received SBRT and 94.6% among those who had conventional radiation. The incidence of adverse events was also low in both groups, with no significant differences observed between the trial arms.
The similar event-free survival and toxicity profiles in both groups provide more support for SBRT, which treats prostate cancer with larger radiation fractions over a shorter time period.
“I think we can also say now with a high level of confidence that SBRT can be considered a new standard of care for low and favorable intermediate-risk prostate cancer,” said Nicholas van As, MD, MB, from the Royal Marsden NHS Foundation Trust and Institute of Cancer Research in London, who presented efficacy and safety results from the noninferiority trial at the American Society for Radiation Oncology (ASTRO) annual meeting. SBRT is more convenient for patients and more cost-effective for health care providers, Dr. Van As added.
Invited discussant Alejandro Berlin, MD, MSc, from Princess Margaret Cancer Centre and the University of Toronto, agreed “that this should be a standard of care for low and favorable intermediate-risk prostate cancer,” an option already endorsed by relevant guidelines.
But, Dr. Berlin noted, SBRT requires careful attention to technique to achieve the desired results. Further research will be needed to identify and potentially reduce variability among radiation oncology practice regarding margins, dosimetry goals, dose heterogeneity, treatment schedules, and other factors, he said.
An international trial
PACE-B is one of three branches of a multi-center collaboration among 37 radiation therapy centers in the United Kingdom, Ireland, and Canada.
In the trial, investigators enrolled 874 patients with T1c or T2c prostate cancer, Gleason score of 3+4 or less, prostate-specific antigen (PSA) level no higher than 20 ng/mL, MRI staging, and no prior androgen deprivation therapy. Investigators then randomly assigned them on a 1:1 basis to receive either conventional radiation (n = 441) or SBRT (n = 433).
At the start of the trial, patients who were assigned to the conventional radiation group received 78 Gy in 39 fractions over 4-8 weeks. However, after results from the CHHiP trial, which showed that a 60-Gy, 20-fraction regimen was not inferior to a 74-Gy, 37-fraction regimen, the PACE-B investigators modified the protocol to 62 Gy delivered in 20 fractions.
Patients assigned to SBRT received 36.25 Gy divided into give fractions delivered over 1-2 weeks, with 40 Gy to the clinical target volume.
The primary outcome was noninferiority of SBRT, measured as whether patients remained free of biochemical clinical failure. Biochemical clinical failure was defined as evidence that the cancer was returning, such as an increase in PSA levels or distant metastases or death from prostate cancer.
At a median follow-up of 73.1 months, 5-year event-free survival rates were 94.6% for patients who received conventional radiation therapy and 95.8% for patients who received SBRT, meeting the prespecified criteria for noninferiority of SBRT (P = .007).
Freedom from biochemical and clinical failure, the trial’s primary endpoint, “was significantly better on both arms than our original power calculation, where we expected control rates of approximately 85%,” Dr. Van As said in an ASTRO plenary session.
Toxicity rates were also low in both study arms. The rate of grade 2 or greater urogenital side effects at 5 years was 5.5% in the SBRT arm and 3.2% in the conventional therapy arm. Grade 2 or greater gastrointestinal side effects occurred in only two patients, one in each study arm.
Given the findings, “I think it’s now imperative that our surgeons discuss this data with their patients before they perform prostatectomies,” Dr. Van As said.
Neha Vapiwala, MD, president-elect of ASTRO who moderated a media briefing where Dr. Van As summarized the PACE-B data, commented that “this study was conducted very rigorously, with excellent quality assurance.”
The study also highlights that clinicians in the United States have considerable catching up to do, said Dr. Vapiwala, from the Hospital of the University of Pennsylvania, Philadelphia.
In the United States, “we are way behind our colleagues on the other side of the pond,” she said. “We are way behind in our uptake of ultra-hypofractionated radiation [such as SBRT], and I do believe that some of that comes from the lack of feeling comfortable with the techniques that are needed and the expertise that is needed.”
PACE-B was funded by Accuray. Dr. Van As disclosed research grants from the company and consulting fees from Varian. Dr. Berlin reported no conflict of interest relevant to the study. Dr. Vapiwala has disclosed a consulting or advisory role with Bayer.
A version of this article first appeared on Medscape.com.
AT ASTRO 2023
Adopting high-dose radiation vs. conventional after mastectomy could be ‘game changer’
SAN DIEGO – , according to a new prospective, randomized study.
Side effects and physical well-being scores were similar among 400 women who received the two treatment regimens, and outcomes were similar or slightly better in the higher-dose group, reported Rinaa Punglia, MD, MPH, an associate professor of radiation oncology at Dana-Farber Brigham Cancer Center in Boston and colleagues at the annual meeting of the American Society for Radiation Oncology. In a press statement, Dr. Punglia noted that the outcomes weren’t as impressive as researchers had hoped, but it’s positive that higher doses didn’t cause more side effects.
The use of the higher-dose approach, known as hypofractionation, “resulted in fewer treatment breaks and less financial toxicity” vs. conventional fractionation, Dr. Punglia said at a news briefing. The findings of the FABREC study “support the use of hypofractionated postmastectomy radiation for patients with basic reconstruction.”
According to Dr. Punglia, “postmastectomy radiation therapy is indicated for almost one-third of mastectomy patients and improves the lives of patients who are at an elevated risk for recurrence.” However, “the addition of radiation therapy greatly increases the risk of reconstruction complications.”
The typical radiation treatment period is 5-6 weeks in these patients, a major hardship for patients that can take them away from their families for extended periods of time. The researchers sought to understand whether another approach – hypofractionation over 3-4 weeks – is a better option. The strategy is widely used after breast-conserving surgery, she said, and has been linked to similar cancer outcomes, improved quality of life, and improved breast appearance.
From 2018 to 2021, the researchers recruited 400 patients with stage 0-III breast cancer who were treated with mastectomy and immediately underwent implant-based reconstruction (median age = 47.0, 23-79). None had tumors growing into the chest wall or skin.
The patients, spread nationwide across 16 institutions, were randomized to receive conventional fractionation (n = 201, 25 fractions, 5 days a week for 5 weeks of 200 cGy) or hypofractionation (n=199, 16 fractions, 5 days a week, for about 3 weeks of 266 cGy).
The researchers tracked 385 patients over a median follow-up of 40.4 months. There was no statistically significant difference in distant recurrence (12 in conventional fractionation arm, 11 in hypofractionation arm), death (2 in each arm), local recurrence (1 in each arm), or toxicity in the chest wall area (20 in conventional fractionation arm, 19 in hypofractionation arm). Changes in physical well-being scores, the primary endpoint, were similar after controlling for age.
“We found that younger patients randomized to hypofractionation were less bothered by side effects of treatment at 6 months relative to their counterparts who received conventional fractionation,” Dr. Punglia said.
Treatment breaks were more common in the conventional fractionation arm (7.7%, mean = 3.3 days) vs. the hypofractionation arm (2.7%, mean = 2.8 days, P = .03).
Among 51 patients who took unpaid time off work, those who underwent hypofractionation took fewer mean days off (73.7 days vs. 125.8 days for conventional fractionation, P = .046).
The study is the first of its kind to compare conventional fractionation to hypofractionation in this population in a randomized, phase III study, Dr. Punglia said.
At the news briefing, an independent expert – Lori Pierce, MD, a professor of radiation oncology at the University of Michigan – said the new study is a “game changer.”
The findings about the benefits of hypofractionation “will potentially impact thousands of women,” said Dr. Pierce, former president of the American Society of Clinical Oncology. The shorter course of radiation is more convenient for patients, she said, and reduces hardship.
“Without a doubt, these results should be discussed with all patients who have had mastectomy and implant-based reconstruction,” she said.
In an interview, Bruce G. Haffty, MD, MS, professor and chair of Radiation Oncology at Rutgers Cancer Institute of New Jersey, said the study adds to existing data suggesting that shorter courses of therapy “are probably OK.” The new findings “give people a little more confidence that [short courses are] safe in terms of well-being and toxicity.”
However, the follow-up in the trial is relatively short, he said, and longer-term research will be needed to change the standard of care in these patients. “It’ll be an evolving story over the next 5-10 years,” he said.
The study was funded by the Patient-Centered Outcomes Research Institute. Dr. Punglia has no disclosures; disclosures for other authors were not provided. Disclosure information for Dr. Pierce was not provided. Dr. Haffty is an investigator in a similar study called RT CHARM.
SAN DIEGO – , according to a new prospective, randomized study.
Side effects and physical well-being scores were similar among 400 women who received the two treatment regimens, and outcomes were similar or slightly better in the higher-dose group, reported Rinaa Punglia, MD, MPH, an associate professor of radiation oncology at Dana-Farber Brigham Cancer Center in Boston and colleagues at the annual meeting of the American Society for Radiation Oncology. In a press statement, Dr. Punglia noted that the outcomes weren’t as impressive as researchers had hoped, but it’s positive that higher doses didn’t cause more side effects.
The use of the higher-dose approach, known as hypofractionation, “resulted in fewer treatment breaks and less financial toxicity” vs. conventional fractionation, Dr. Punglia said at a news briefing. The findings of the FABREC study “support the use of hypofractionated postmastectomy radiation for patients with basic reconstruction.”
According to Dr. Punglia, “postmastectomy radiation therapy is indicated for almost one-third of mastectomy patients and improves the lives of patients who are at an elevated risk for recurrence.” However, “the addition of radiation therapy greatly increases the risk of reconstruction complications.”
The typical radiation treatment period is 5-6 weeks in these patients, a major hardship for patients that can take them away from their families for extended periods of time. The researchers sought to understand whether another approach – hypofractionation over 3-4 weeks – is a better option. The strategy is widely used after breast-conserving surgery, she said, and has been linked to similar cancer outcomes, improved quality of life, and improved breast appearance.
From 2018 to 2021, the researchers recruited 400 patients with stage 0-III breast cancer who were treated with mastectomy and immediately underwent implant-based reconstruction (median age = 47.0, 23-79). None had tumors growing into the chest wall or skin.
The patients, spread nationwide across 16 institutions, were randomized to receive conventional fractionation (n = 201, 25 fractions, 5 days a week for 5 weeks of 200 cGy) or hypofractionation (n=199, 16 fractions, 5 days a week, for about 3 weeks of 266 cGy).
The researchers tracked 385 patients over a median follow-up of 40.4 months. There was no statistically significant difference in distant recurrence (12 in conventional fractionation arm, 11 in hypofractionation arm), death (2 in each arm), local recurrence (1 in each arm), or toxicity in the chest wall area (20 in conventional fractionation arm, 19 in hypofractionation arm). Changes in physical well-being scores, the primary endpoint, were similar after controlling for age.
“We found that younger patients randomized to hypofractionation were less bothered by side effects of treatment at 6 months relative to their counterparts who received conventional fractionation,” Dr. Punglia said.
Treatment breaks were more common in the conventional fractionation arm (7.7%, mean = 3.3 days) vs. the hypofractionation arm (2.7%, mean = 2.8 days, P = .03).
Among 51 patients who took unpaid time off work, those who underwent hypofractionation took fewer mean days off (73.7 days vs. 125.8 days for conventional fractionation, P = .046).
The study is the first of its kind to compare conventional fractionation to hypofractionation in this population in a randomized, phase III study, Dr. Punglia said.
At the news briefing, an independent expert – Lori Pierce, MD, a professor of radiation oncology at the University of Michigan – said the new study is a “game changer.”
The findings about the benefits of hypofractionation “will potentially impact thousands of women,” said Dr. Pierce, former president of the American Society of Clinical Oncology. The shorter course of radiation is more convenient for patients, she said, and reduces hardship.
“Without a doubt, these results should be discussed with all patients who have had mastectomy and implant-based reconstruction,” she said.
In an interview, Bruce G. Haffty, MD, MS, professor and chair of Radiation Oncology at Rutgers Cancer Institute of New Jersey, said the study adds to existing data suggesting that shorter courses of therapy “are probably OK.” The new findings “give people a little more confidence that [short courses are] safe in terms of well-being and toxicity.”
However, the follow-up in the trial is relatively short, he said, and longer-term research will be needed to change the standard of care in these patients. “It’ll be an evolving story over the next 5-10 years,” he said.
The study was funded by the Patient-Centered Outcomes Research Institute. Dr. Punglia has no disclosures; disclosures for other authors were not provided. Disclosure information for Dr. Pierce was not provided. Dr. Haffty is an investigator in a similar study called RT CHARM.
SAN DIEGO – , according to a new prospective, randomized study.
Side effects and physical well-being scores were similar among 400 women who received the two treatment regimens, and outcomes were similar or slightly better in the higher-dose group, reported Rinaa Punglia, MD, MPH, an associate professor of radiation oncology at Dana-Farber Brigham Cancer Center in Boston and colleagues at the annual meeting of the American Society for Radiation Oncology. In a press statement, Dr. Punglia noted that the outcomes weren’t as impressive as researchers had hoped, but it’s positive that higher doses didn’t cause more side effects.
The use of the higher-dose approach, known as hypofractionation, “resulted in fewer treatment breaks and less financial toxicity” vs. conventional fractionation, Dr. Punglia said at a news briefing. The findings of the FABREC study “support the use of hypofractionated postmastectomy radiation for patients with basic reconstruction.”
According to Dr. Punglia, “postmastectomy radiation therapy is indicated for almost one-third of mastectomy patients and improves the lives of patients who are at an elevated risk for recurrence.” However, “the addition of radiation therapy greatly increases the risk of reconstruction complications.”
The typical radiation treatment period is 5-6 weeks in these patients, a major hardship for patients that can take them away from their families for extended periods of time. The researchers sought to understand whether another approach – hypofractionation over 3-4 weeks – is a better option. The strategy is widely used after breast-conserving surgery, she said, and has been linked to similar cancer outcomes, improved quality of life, and improved breast appearance.
From 2018 to 2021, the researchers recruited 400 patients with stage 0-III breast cancer who were treated with mastectomy and immediately underwent implant-based reconstruction (median age = 47.0, 23-79). None had tumors growing into the chest wall or skin.
The patients, spread nationwide across 16 institutions, were randomized to receive conventional fractionation (n = 201, 25 fractions, 5 days a week for 5 weeks of 200 cGy) or hypofractionation (n=199, 16 fractions, 5 days a week, for about 3 weeks of 266 cGy).
The researchers tracked 385 patients over a median follow-up of 40.4 months. There was no statistically significant difference in distant recurrence (12 in conventional fractionation arm, 11 in hypofractionation arm), death (2 in each arm), local recurrence (1 in each arm), or toxicity in the chest wall area (20 in conventional fractionation arm, 19 in hypofractionation arm). Changes in physical well-being scores, the primary endpoint, were similar after controlling for age.
“We found that younger patients randomized to hypofractionation were less bothered by side effects of treatment at 6 months relative to their counterparts who received conventional fractionation,” Dr. Punglia said.
Treatment breaks were more common in the conventional fractionation arm (7.7%, mean = 3.3 days) vs. the hypofractionation arm (2.7%, mean = 2.8 days, P = .03).
Among 51 patients who took unpaid time off work, those who underwent hypofractionation took fewer mean days off (73.7 days vs. 125.8 days for conventional fractionation, P = .046).
The study is the first of its kind to compare conventional fractionation to hypofractionation in this population in a randomized, phase III study, Dr. Punglia said.
At the news briefing, an independent expert – Lori Pierce, MD, a professor of radiation oncology at the University of Michigan – said the new study is a “game changer.”
The findings about the benefits of hypofractionation “will potentially impact thousands of women,” said Dr. Pierce, former president of the American Society of Clinical Oncology. The shorter course of radiation is more convenient for patients, she said, and reduces hardship.
“Without a doubt, these results should be discussed with all patients who have had mastectomy and implant-based reconstruction,” she said.
In an interview, Bruce G. Haffty, MD, MS, professor and chair of Radiation Oncology at Rutgers Cancer Institute of New Jersey, said the study adds to existing data suggesting that shorter courses of therapy “are probably OK.” The new findings “give people a little more confidence that [short courses are] safe in terms of well-being and toxicity.”
However, the follow-up in the trial is relatively short, he said, and longer-term research will be needed to change the standard of care in these patients. “It’ll be an evolving story over the next 5-10 years,” he said.
The study was funded by the Patient-Centered Outcomes Research Institute. Dr. Punglia has no disclosures; disclosures for other authors were not provided. Disclosure information for Dr. Pierce was not provided. Dr. Haffty is an investigator in a similar study called RT CHARM.
AT ASTRO 2023
SABR could defer systemic therapy in oligoprogressive breast cancer
SAN DIEGO – Stereotactic ablative body radiotherapy (SABR) appeared to delay the need for changes in systemic therapy in postmenopausal patients with oligoprogressive luminal ER-positive, HER2-negative breast cancer, according to a new phase 2 study.
In the AVATAR trial, patients with one to five metastatic lesions who’d been treated with cyclin-dependent kinase (CDK) 4/6 inhibitors and aromatase inhibitors for at least 6 months underwent SABR. Of those, 47% had event-free survival of more than 6 months, an unexpectedly high figure, reported radiation oncologist Steven David, MBBS, of Peter MacCallum Cancer Center, Melbourne, and colleagues at the annual meeting of the American Society for Radiation Oncology.
“We found surprisingly that SABR delayed a change in therapy by 10 months, which is great for patients. Also, one in three patients had a second round of SABR,” said Dr. David in an interview. “This trial provides the first prospective evidence to delay a change in therapy in this population, and this strategy is ready to go now.”
According to Dr. David, oligoprogressive luminal, ER-positive, HER2-negative, advanced breast cancer cannot be cured. However, patients can live more than 10 years in some cases, and an early treatment – CDK 4/6 inhibitors and aromatase inhibitors – is well tolerated. “Patients can lead a normal life and avoid chemo” as long as those medications keep working.
The goal of the study was to determine if SABR is helpful in these patients. The treatment, which produces highly focused radiation, “has very few side effects and does a great job in eliminating progressing metastases,” Dr. David said.
For the study, researchers recruited 32 subjects at 13 Australian sites. Participants could not have had leptomeningeal disease, previous chemotherapy for metastatic disease, or prior radiotherapy to an oligoprogressing lesion. Most metastases were to bone (n = 44, 71%), node (systemic, n = 8; 13%) and lung (n = 4; 6%).
The patients were treated with SABR, most commonly 24 Gy (n = 25; 43%) and 20 Gy (n = 10; 17%); half had one lesion treated (50%), and 25% had two lesions treated.
The median follow-up was 15.8 months. The median event-free survival was 5.2 months (95% confidence interval, 3.1-9.4 months), with events defined as progression within 6 months or in at least three lesions. Fifteen patients (47%) reached event-free survival of 6 or more months.
Elysia Donovan, MD, MSc, a radiation oncologist at McMaster University, Hamilton, Ont., said in an interview that the new study is thoughtfully designed, although it’s not definitive. “At this point we still do not know the optimal treatment regimen for oligoprogressive breast cancer. The findings of this trial are promising and exciting. However, further randomized trials are required before routine implementation in clinical practice. For now, patients should be considered in a case by case basis with multidisciplinary discussion to determine the optimal systemic therapy regimen at oligoprogression and whether SABR may provide benefit.”
Median modified progression-free survival was 10.4 months, and median progression-free survival was 5.2 months; 31% of patients received SABR for further oligoprogression, and 46% patients remained on CDK4/6 inhibitors and aromatase inhibitors for 12 months. Overall survival was 100%.
A total of 14 patients had grade 1 adverse events, 2 had grade 2 events, and none had grade 3 or higher events; 47% had no treatment-related toxicity.
The strategy “potentially has a place in other cancer types and other breast cancer types,” Dr. David said.
In an interview, Katarzyna Jerzak, MD, MSc, a medical oncologist with Sunnybrook Odette Cancer Center in Toronto, said the findings are promising, although the study is small and the patients are similar. Toxicity was limited, and a 12-month delay in a switch to therapy – reached by 46% – “is very meaningful for patients.” She added that “the positive results should serve as motivation to investigate the strategy further.”
Dr. David said a larger trial called AVATAR 2 is funded and in the works. It will have more patients and more breast cancer subtypes.
The study was funded by the Donald Ratcliffe and Phyllis McLeod Trust. Dr. David disclosed grant/research funding from Roche Genentech, and other authors reported various disclosures including relationships with AstraZeneca, Pfizer, Gilead, and others. Dr. Jerzak disclosed speaker/advisor board/consultant relationships with Amgen, AstraZeneca, Apobiologix, Eli Lilly, Esai, Genomic Health, Gilead, Knight Therapeutics, Merck, Myriad Genetics, Pfizer, Roche, Seagen, and Novartis and research funding from AstraZeneca, Eli Lilly, and Seagen. Dr. Donovan disclosed a Bright Foundation grant for a prospective trial of SABR for oligoprogressive breast cancer.
SAN DIEGO – Stereotactic ablative body radiotherapy (SABR) appeared to delay the need for changes in systemic therapy in postmenopausal patients with oligoprogressive luminal ER-positive, HER2-negative breast cancer, according to a new phase 2 study.
In the AVATAR trial, patients with one to five metastatic lesions who’d been treated with cyclin-dependent kinase (CDK) 4/6 inhibitors and aromatase inhibitors for at least 6 months underwent SABR. Of those, 47% had event-free survival of more than 6 months, an unexpectedly high figure, reported radiation oncologist Steven David, MBBS, of Peter MacCallum Cancer Center, Melbourne, and colleagues at the annual meeting of the American Society for Radiation Oncology.
“We found surprisingly that SABR delayed a change in therapy by 10 months, which is great for patients. Also, one in three patients had a second round of SABR,” said Dr. David in an interview. “This trial provides the first prospective evidence to delay a change in therapy in this population, and this strategy is ready to go now.”
According to Dr. David, oligoprogressive luminal, ER-positive, HER2-negative, advanced breast cancer cannot be cured. However, patients can live more than 10 years in some cases, and an early treatment – CDK 4/6 inhibitors and aromatase inhibitors – is well tolerated. “Patients can lead a normal life and avoid chemo” as long as those medications keep working.
The goal of the study was to determine if SABR is helpful in these patients. The treatment, which produces highly focused radiation, “has very few side effects and does a great job in eliminating progressing metastases,” Dr. David said.
For the study, researchers recruited 32 subjects at 13 Australian sites. Participants could not have had leptomeningeal disease, previous chemotherapy for metastatic disease, or prior radiotherapy to an oligoprogressing lesion. Most metastases were to bone (n = 44, 71%), node (systemic, n = 8; 13%) and lung (n = 4; 6%).
The patients were treated with SABR, most commonly 24 Gy (n = 25; 43%) and 20 Gy (n = 10; 17%); half had one lesion treated (50%), and 25% had two lesions treated.
The median follow-up was 15.8 months. The median event-free survival was 5.2 months (95% confidence interval, 3.1-9.4 months), with events defined as progression within 6 months or in at least three lesions. Fifteen patients (47%) reached event-free survival of 6 or more months.
Elysia Donovan, MD, MSc, a radiation oncologist at McMaster University, Hamilton, Ont., said in an interview that the new study is thoughtfully designed, although it’s not definitive. “At this point we still do not know the optimal treatment regimen for oligoprogressive breast cancer. The findings of this trial are promising and exciting. However, further randomized trials are required before routine implementation in clinical practice. For now, patients should be considered in a case by case basis with multidisciplinary discussion to determine the optimal systemic therapy regimen at oligoprogression and whether SABR may provide benefit.”
Median modified progression-free survival was 10.4 months, and median progression-free survival was 5.2 months; 31% of patients received SABR for further oligoprogression, and 46% patients remained on CDK4/6 inhibitors and aromatase inhibitors for 12 months. Overall survival was 100%.
A total of 14 patients had grade 1 adverse events, 2 had grade 2 events, and none had grade 3 or higher events; 47% had no treatment-related toxicity.
The strategy “potentially has a place in other cancer types and other breast cancer types,” Dr. David said.
In an interview, Katarzyna Jerzak, MD, MSc, a medical oncologist with Sunnybrook Odette Cancer Center in Toronto, said the findings are promising, although the study is small and the patients are similar. Toxicity was limited, and a 12-month delay in a switch to therapy – reached by 46% – “is very meaningful for patients.” She added that “the positive results should serve as motivation to investigate the strategy further.”
Dr. David said a larger trial called AVATAR 2 is funded and in the works. It will have more patients and more breast cancer subtypes.
The study was funded by the Donald Ratcliffe and Phyllis McLeod Trust. Dr. David disclosed grant/research funding from Roche Genentech, and other authors reported various disclosures including relationships with AstraZeneca, Pfizer, Gilead, and others. Dr. Jerzak disclosed speaker/advisor board/consultant relationships with Amgen, AstraZeneca, Apobiologix, Eli Lilly, Esai, Genomic Health, Gilead, Knight Therapeutics, Merck, Myriad Genetics, Pfizer, Roche, Seagen, and Novartis and research funding from AstraZeneca, Eli Lilly, and Seagen. Dr. Donovan disclosed a Bright Foundation grant for a prospective trial of SABR for oligoprogressive breast cancer.
SAN DIEGO – Stereotactic ablative body radiotherapy (SABR) appeared to delay the need for changes in systemic therapy in postmenopausal patients with oligoprogressive luminal ER-positive, HER2-negative breast cancer, according to a new phase 2 study.
In the AVATAR trial, patients with one to five metastatic lesions who’d been treated with cyclin-dependent kinase (CDK) 4/6 inhibitors and aromatase inhibitors for at least 6 months underwent SABR. Of those, 47% had event-free survival of more than 6 months, an unexpectedly high figure, reported radiation oncologist Steven David, MBBS, of Peter MacCallum Cancer Center, Melbourne, and colleagues at the annual meeting of the American Society for Radiation Oncology.
“We found surprisingly that SABR delayed a change in therapy by 10 months, which is great for patients. Also, one in three patients had a second round of SABR,” said Dr. David in an interview. “This trial provides the first prospective evidence to delay a change in therapy in this population, and this strategy is ready to go now.”
According to Dr. David, oligoprogressive luminal, ER-positive, HER2-negative, advanced breast cancer cannot be cured. However, patients can live more than 10 years in some cases, and an early treatment – CDK 4/6 inhibitors and aromatase inhibitors – is well tolerated. “Patients can lead a normal life and avoid chemo” as long as those medications keep working.
The goal of the study was to determine if SABR is helpful in these patients. The treatment, which produces highly focused radiation, “has very few side effects and does a great job in eliminating progressing metastases,” Dr. David said.
For the study, researchers recruited 32 subjects at 13 Australian sites. Participants could not have had leptomeningeal disease, previous chemotherapy for metastatic disease, or prior radiotherapy to an oligoprogressing lesion. Most metastases were to bone (n = 44, 71%), node (systemic, n = 8; 13%) and lung (n = 4; 6%).
The patients were treated with SABR, most commonly 24 Gy (n = 25; 43%) and 20 Gy (n = 10; 17%); half had one lesion treated (50%), and 25% had two lesions treated.
The median follow-up was 15.8 months. The median event-free survival was 5.2 months (95% confidence interval, 3.1-9.4 months), with events defined as progression within 6 months or in at least three lesions. Fifteen patients (47%) reached event-free survival of 6 or more months.
Elysia Donovan, MD, MSc, a radiation oncologist at McMaster University, Hamilton, Ont., said in an interview that the new study is thoughtfully designed, although it’s not definitive. “At this point we still do not know the optimal treatment regimen for oligoprogressive breast cancer. The findings of this trial are promising and exciting. However, further randomized trials are required before routine implementation in clinical practice. For now, patients should be considered in a case by case basis with multidisciplinary discussion to determine the optimal systemic therapy regimen at oligoprogression and whether SABR may provide benefit.”
Median modified progression-free survival was 10.4 months, and median progression-free survival was 5.2 months; 31% of patients received SABR for further oligoprogression, and 46% patients remained on CDK4/6 inhibitors and aromatase inhibitors for 12 months. Overall survival was 100%.
A total of 14 patients had grade 1 adverse events, 2 had grade 2 events, and none had grade 3 or higher events; 47% had no treatment-related toxicity.
The strategy “potentially has a place in other cancer types and other breast cancer types,” Dr. David said.
In an interview, Katarzyna Jerzak, MD, MSc, a medical oncologist with Sunnybrook Odette Cancer Center in Toronto, said the findings are promising, although the study is small and the patients are similar. Toxicity was limited, and a 12-month delay in a switch to therapy – reached by 46% – “is very meaningful for patients.” She added that “the positive results should serve as motivation to investigate the strategy further.”
Dr. David said a larger trial called AVATAR 2 is funded and in the works. It will have more patients and more breast cancer subtypes.
The study was funded by the Donald Ratcliffe and Phyllis McLeod Trust. Dr. David disclosed grant/research funding from Roche Genentech, and other authors reported various disclosures including relationships with AstraZeneca, Pfizer, Gilead, and others. Dr. Jerzak disclosed speaker/advisor board/consultant relationships with Amgen, AstraZeneca, Apobiologix, Eli Lilly, Esai, Genomic Health, Gilead, Knight Therapeutics, Merck, Myriad Genetics, Pfizer, Roche, Seagen, and Novartis and research funding from AstraZeneca, Eli Lilly, and Seagen. Dr. Donovan disclosed a Bright Foundation grant for a prospective trial of SABR for oligoprogressive breast cancer.
AT ASTRO 2023