User login
Stridor in the Pediatric Patient
The distinct features of the pediatric airway make respiratory failure an important concern independent of the underlying cause.
Cases
Case 1
It’s a busy shift on an unusually chilly and rainy July night. Emergency medical services (EMS) brings in a 9-month-old boy who woke up with a “squeaking” noise. His parents reported that he has had a fever, cough, rhinorrhea, and difficulty breathing for the past 2 days; however, they did not hear the noisy breathing until the night of presentation. When the patient is examined, it is noted that he has inspiratory stridor at rest, moderate subcostal retractions, and an occasional deep cough. Upper airway transmitted noises were present, but otherwise the patient had clear lungs.
The patient’s vital signs at presentation were: blood pressure (BP), 85/55 mm Hg; heart rate (HR), 163 beats/min; respiratory rate (RR), 55 breaths/min; and temperature (T), 101.8°F. Oxygen saturation was 90% on room air. The patient’s mother wants to know how the respiratory distress will be fixed and is inquiring if they will have to stay in the hospital overnight.
Case 2
As work begins on the child described above, EMS brings in a 3-year-old girl who appears to be in moderate-severe respiratory distress. Her parents report that she started to drool earlier in the day followed by coughing and occasional gagging. Her parents relay that they thought the symptoms were because of post-nasal drip due to her cold, but the respiratory distress seems to be getting worse, and she now has very noisy breathing and is reluctant to lay down. Upon examination, both inspiratory and expiratory stridor is heard, and it is noted that moderate subcostal retractions are present when the patient is supine.
The patient’s vital signs at presentation were: BP, 89/58 mm Hg; HR, 144 beats/min; RR, 52 breaths/min; and T, 99.5°F. Oxygen saturation was 88% on room air. The nursing staff asked what to do next and why the 2 stridor cases are being managed so differently.
Stridor
Stridor is a high-pitched, harsh sound heard during respiration, predominantly during inspiration, as a result of turbulent air passage.1-3 Stridor is not a diagnosis in itself, but rather a sign of underlying acute or chronic etiology, which needs to be classified based on elicited history and examination.1,4 While the etiology of obstruction may be infectious, congenital, mechanical, or traumatic, the distinct features of the pediatric airway make respiratory failure an important concern independent of the underlying cause.
Anatomy
There are several anatomical differences unique to the pediatric airway that make children, especially infants under 1 year of age, more susceptible to airway obstruction.5,6The pediatric larynx is more anterior and superior and less fibrous than adults, thereby more compliant.6,7 The pediatric epiglottis is longer, omega-shaped, and softer, and the tongue is larger in comparison to the size of the oral cavity, resulting in obstructed airflow.6,7 Children also have a larger and more prominent occiput, causing mechanical obstruction by flexion of the neck when supine.6 While the cricoid cartilage was previously believed to be the narrowest portion of the airway, more recent measurement techniques have challenged this and shown that the glottic and subglottic areas may be narrower in children.7,8
Worsening obstruction resulting in a decreased airway radius leads to increased turbulence to air flow, which is explained by Poiseuille’s law.1,2,7 The resistance to airflow becomes inversely related to the fourth power of the radius, so even a small change in an already narrow pediatric airway can make a huge difference.5 In practice, this means 1 mm of mucosal edema will decrease the cross-sectional area of the airway by 75% and increase the resistance of airflow by a magnitude of 16 to 32 times depending on the level of turbulence.7The airway can be divided into extrathoracic and intrathoracic regions, separated by the vocal cords.13 Inspiratory stridor is typically due to extrathoracic obstruction and expiratory stridor is due to intrathoracic obstruction below the level of the cords. Biphasic stridor, however, may indicate a fixed obstruction at the level of the cords.2,3,5,9
Acute Differentials
The pediatric patient is at high risk for respiratory decompensation when the upper airway is acutely compromised. The history, physical examination, and phases of stridor can help determine the underlying diagnosis and definitive treatment plan.
Acute Croup
Acute croup (laryngotracheitis) is a clinical diagnosis based on acute onset of barky cough and inspiratory stridor.1 It is usually secondary to an infection, most commonly viral (parainfluenza virus), resulting in edema and increased secretions of the subglottic mucosa.7 The onset is typically preceded by upper respiratory illness (URI) symptoms, and is often worse at night or after waking from a nap.1 The peak incidence is between ages 6 months to 3 years. While croup is a clinical diagnosis, an anteroposterior X-ray will often show a steeple sign.7,10
Spasmodic Croup
Spasmodic croup is an atypical presentation usually seen in children 8 years or older without a preceding URI. Patients will wake up overnight with a harsh brass-like cough, stridor, and hoarse voice. The etiology is unclear, but often these patients have a history of atopy and respond in part to treatment with antihistamines.
Foreign Body Aspiration
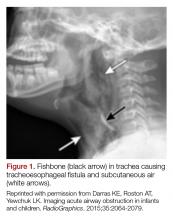
All that is acutely stridulous is not croup. Stridor from foreign body aspiration is sudden in onset and children do not always present with a history suggestive of foreign body aspiration. Diagnosis requires a high index of suspicion because the event is often unwitnessed and typical patients are pre-verbal. The physical examination may reveal diminished air entry along with stridor. Diagnosis is made by obtaining an X-ray (lateral neck [Figure 1], lateral decubitus, or inspiratory/expiratory chest X-ray) or by bronchoscopy which is both diagnostic and therapeutic.7,10 Remember to always think of foreign body aspiration in children with acute stridor who have neither fever nor antecedent URI symptoms.
Bacterial Tracheitis
Bacterial tracheitis should be suspected in toxic appearing children who present with respiratory distress and stridor but who have a poor response to nebulized epinephrine. It is typically seen in toddlers and school-aged children, and like a viral tracheitis, presentation can be preceded by either URI or fever.10 Stridor is caused by subglottic edema and mucopurulent secretions in the airway.7,10 Infection is most commonly by S. aureus but initial antibiotic choice should be broad spectrum, and include a third-generation cephalosporin or beta-lactamase resistant penicillin.
Epiglottitis
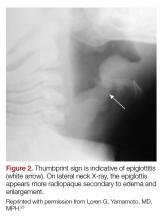
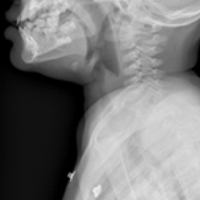
Epiglottitis is edema of the epiglottis, most commonly secondary to bacterial infection. The epidemiology of epiglottitis has changed dramatically since widespread immunization for H. influenza with a significantly decreased incidence and change in the average age of presentation to 14.6 years (previously 5.6 years).7 The clinical course begins with sore throat, dysphonia, refusal to eat and progressive difficulty handling secretions with eventual drooling, stridor, tripoding, and toxic appearance. Epiglottitis can be differentiated from croup and bacterial tracheitis because presentation typically lacks a cough.7,10,11 Diagnosis is made either by direct visualization of the epiglottis or a lateral neck X-ray showing a ‘thumb print’ sign (Figure 2).12 Emergency department treatment is similar to the management of the child with a partial foreign body occlusion and focuses on maintaining the airway and minimizing anything that agitates the patient. Intravenous (IV) antibiotic coverage is similar to bacterial tracheitis (third-generation cephalosporin or a beta-lactamase resistant penicillin).
Retropharyngeal Abscess
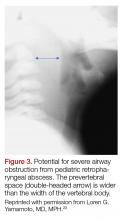
The most common chief complaint of retropharyngeal abscess (RPA) is neck pain (38%) with fever. As such, it can clinically be mistaken for meningitis on initial presentation. Retropharyngeal abscess will present rarely with either stridor or associated respiratory distress, and it can also mimic croup on initial presentation. Physical examination findings which differentiate this entity include limited or painful neck extension (45%), torticollis (36.5%), and to a lesser extent limitation of neck flexion (12.5%).13 The median age at diagnosis is 36 months with 75% of patients less than 5 years. Typical presentation is insidious with fever and URI symptoms preceding onset. Diagnosis can be made with a lateral neck X-ray showing widening of the prevertebral space (Figure 3), but the gold standard diagnostic study is a computed tomography with contrast.14 Management is IV antibiotics covering aerobic and anaerobic bacteria (eg, ampicillin-sulbactam) ± surgical intervention.
Caustic Ingestion
Caustic ingestion is most commonly accidental and seen in children aged 12 months to 2 years. However, with recent fads, such as the “Tide Pod challenge” teenagers are also at risk. Airway compromise and stridor are secondary to mucosal injury and edema. Oral injury is not always a useful marker for significant distal injury. A complete evaluation of the upper airway and digestive tract within 48 hours after known/suspected caustic ingestion is recommended to assess full extent of damage.15
Chronic Differentials
Laryngomalacia
Laryngomalacia is a congenital weakness of laryngeal tissues, and it is the most common cause of both chronic stridor and neonatal stridor. It is characterized by progressive worsening of symptoms with crying/feeding and supine positioning. Diagnosis is made by bronchoscopy and management is conservative unless there are life threatening apneic or cyanotic events.7
Rings/Slings
There are many anatomic structures with the potential to cause extrinsic airway compression which present with stridor. This type of stridor is often biphasic. Examples include innominate artery compression, double aortic arch, aberrant subclavian artery, and pulmonary artery sling.7
Stridor presenting in children with a history of prematurity or prolonged intubation should raise concern for subglottic and tracheal stenosis.16
Evaluation
Regardless of the etiology of stridor, efforts should be made to keep the patient calm (ie, allow the parent to keep holding a young child, limit any examination not absolutely necessary). Much of the examination can be completed from a distance without disturbing the child.17 Observation of the inspiratory:expiratory (I:E) ratio can localize the level of airway obstruction. For example, an I:E ratio weighted toward a longer inspiration indicates an extrathoracic airway obstruction. Whereas an I:E with a prolonged expiratory phase is consistent with intrathoracic obstruction (eg, terminal bronchial obstruction).17
Another way to localize the level of obstruction is to look for changes in the voice; patients who present with a change in their voice have a subglottic partial obstruction such as croup. However, patients with a muffled voice or drooling have a supraglottic obstruction such as epiglottitis or RPA.17
Management
Management of stridor focuses on reducing airway obstruction, which is usually secondary to edema in the acute setting.
Viral Laryngotracheitis
Oral steroids are the mainstay of treatment. Research has shown dexamethasone is preferred over prednisolone.18-20 Steroids are not only useful in moderate to severe laryngotracheitis but also have a therapeutic role in children with mild laryngotracheitis.18 In hospital settings the parenteral formulation of dexamethasone can be safely given orally with good effect. There is no therapeutic advantage in acute laryngotracheitis to giving dexamethasone via either the IV or intramuscular route vs oral.21 In the outpatient setting, decadron tablets can be crushed and mixed in with a young child’s favorite soft food (eg, mashed potatoes or apple sauce). The authors recommend this strategy in lieu of prescribing dexamethasone suspension as its dilute concentration (1 mg/10 ml) results in a need for a child to receive a relatively large volume of a distasteful liquid. There is a wide therapeutic range of dexamethasone with studies documenting efficacy for laryngotracheitis in doses ranging from 0.15 mg/kg to 0.6 mg/kg. To date there are no large studies which demonstrate routine therapeutic utility of subsequent doses of dexamethasone. Nebulized budesonide (2.5 mg) can be given if oral steroids are not tolerated, however it is significantly more expensive.
Racemic epinephrine is the agent of choice for rapid onset of action in children who demonstrate stridor at rest. It causes vasoconstriction in the laryngeal mucosa, promotes bronchial smooth muscle relaxation, and thinning of bronchial secretions. It offers short-term relief of symptoms until steroids start to work. There is no rebound effect or worsening of symptoms once the epinephrine wears off, but children who receive this drug should be observed in the ED for a period of time (2-3 hours is standard of care in many hospitals) for return of symptoms.10,22 Patients who are persistently symptomatic 4 hours after administration of steroids or who require repeat doses of racemic epinephrine should be admitted for observation.10There are no contraindications to adjuvant treatments, such as antipyretics and non-sedating analgesics. Clinicians should maintain a high index of suspicion for anatomic airway anomalies that may need further evaluation/direct visualization in pediatric patients who present with repeated episodes of croup.10
Case Conclusions
Case 1
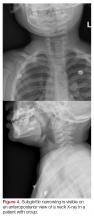
The 9-month-old boy with stridor was noted to have increased stridor while fussy, but even at rest some inspiratory stridor was present. A barky cough was noted in the examination room. The patient was placed on a monitor and a nebulized racemic epinephrine treatment was started. A single dose of oral dexamethasone was given shortly after presentation to the ED. Since the patient had inspiratory stridor at rest with associated tachypnea and hypoxia on initial presentation, a neck X-ray was obtained (Figure 4). Subglottic narrowing was identified on the imaging, but both the epiglottis and the prevertebral space were normal in appearance and no foreign bodies were visualized. The inspiratory stridor at rest, tachypnea, and mild hypoxia all improved after treatment, the patient was observed for 2 hours in the ED without recurrence of respiratory distress and was able to be discharged home with a diagnosis of acute croup.
Case 2
The 3-year-old girl was noted to be in significant positional respiratory distress, so the physician asked her parents to keep her calm in her position of comfort. She was calmly and quickly placed on a monitor with age-appropriate distraction techniques in place and advanced airway equipment at the bedside. A portable chest X-ray was obtained and revealed a coin was partially obstructing the trachea. Care was taken in the ED to avoid all interventions such as IV access that might upset the child so as not to inadvertently convert this partial airway obstruction to a complete obstruction. The otolaryngology team was called urgently, and the patient was transported to the operating room for foreign body removal in a controlled environment.
1. Bjornson CL, Johnson DW. Croup. Lancet. 2008;371(9609):329-339. doi:10.1016/S0140-6736(08)60170-1.
2. Escobar ML, Needleman J. Stridor. Pediatr Rev. 2015;36(3):135-137. doi:10.1542/pir.36-3-135.
3. Pfleger A, Eber E. Assessment and causes of stridor. Paediatr Respir Rev. 2016;18:64-72. doi:10.1016/j.prrv.2015.10.003.
4. Boudewyns A, Claes J, Van de Heyning P. Clinical practice: an approach to stridor in infants and children. Eur J Pediatr. 2010;169(2):135-141. doi:10.1007/s00431-009-1044-7.
5. Kelley PB, Simon JE. Racemic epinephrine use in croup and disposition. Am J Emerg Med. 1992;10(3):181-183.
6. Mandal A, Kabra SK, Lodha R. Upper airway obstruction in children. Indian J Pediatr. 2015;82(8):737-744.
7. Marchese A, Langhan ML. Management of airway obstruction and stridor in pediatric patients. Pediatr Emerg Med Pract. 2017;14(11):1-24.
8. Wani TM, Bissonnette B, Rafiq Malik M, et al. Age-based analysis of pediatric upper airway dimensions using computed tomography imaging. Pediatr Pulmonol. 2016;51(3):267-271. doi:10.1002/ppul.23232.
9. Donaldson D, Poleski D, Knipple E, et al. Intramuscular versus oral dexamethasone for the treatment of moderate-to-severe croup: a randomized, double-blind trial. Acad Emerg Med. 2003;10(1):16-21.
10. Boudewyns A, Claes J, Van de Heyning P. Clinical practice: an approach to stridor in infants and children. Eur J Pediatr. 2010;169(2):135-141. doi:10.1007/s00431-009-1044-7.
11. Tibballs J, Watson T. Symptoms and signs differentiating croup and epiglottitis. J Paediatr Child Health. 2011;47(3):77-82. doi:10.1111/j.1440-1754.2010.01892.x.
12. Sobol SE, Zapata S. Epiglottitis and croup. Otolaryngol Clin North Am. 2008;41(3):551-566. doi:10.1016/j.otc.2008.01.012.
13. Craig FW, Schunk JE. Retropharyngeal abscess in children: clinical presentation, utility of imaging, and current management. Pediatrics. 2003;111(6 Pt 1):1394-1398.
14. Roberson DW. Pediatric retropharyngeal abscesses. Clin Pediatr Emerg Med. 2004;5(1):37-40.
15. Riffat F, Cheng A. Pediatric caustic ingestion: 50 consecutive cases and a review of the literature. Dis Esophagus. 2009;22(1):89-94. doi:10.1111/j.1442-2050.2008.00867.x.
16. Daniel SJ. The upper airway: congenital malformations. Paediatr Respir Revi. 2006;7 Suppl 1:S260-S263.
17. Arutyunyan H, Spangler M. Pediatric upper airway obstruction. Peds RAP Web site. https://www.hippoed.com/peds/rap/episode/pedsrapfebruary/pediatricupper. Published February 2018. Accessed August 31, 2018.
18. Bjornson CL, Klassen TP, Williamson J, et al; Pediatric Emergency Research Canada Network. A randomized trial of a single dose of oral dexamethasone for mild croup. N Engl J Med. 2004;351(13):1306-1313.
19. Fifoot AA, Ting JY. Comparison between single-dose oral prednisolone and oral dexamethasone in the treatment of croup: a randomized, double-blinded clinical trial. Emerg Med Australas. 2007;19(1):51-58.
20. Sparrow A, Geelhoed G. Prednisolone versus dexamethasone in croup: a randomised equivalence trial. Arch Dis Child. 2005;91(7):580-583.
21. Donaldson D, Poleski D, Knipple E, et al. Intramuscular versus oral dexamethasone for the treatment of moderate-to-severe croup: a randomized, double-blind trial. Acad Emerg Med. 2003;10(1):16-21.
22. Kelley PB, Simon JE. Racemic epinephrine use in croup and disposition. Am J Emerg Med. 1992;10(3):181-183.
23. Yamamoto LG. Test your skill in reading pediatric lateral necks. University of Hawaii Web site. https://www.hawaii.edu/medicine/pediatrics/pemxray/v2c20.html. Accessed September 13, 2018.
The distinct features of the pediatric airway make respiratory failure an important concern independent of the underlying cause.
The distinct features of the pediatric airway make respiratory failure an important concern independent of the underlying cause.
Cases
Case 1
It’s a busy shift on an unusually chilly and rainy July night. Emergency medical services (EMS) brings in a 9-month-old boy who woke up with a “squeaking” noise. His parents reported that he has had a fever, cough, rhinorrhea, and difficulty breathing for the past 2 days; however, they did not hear the noisy breathing until the night of presentation. When the patient is examined, it is noted that he has inspiratory stridor at rest, moderate subcostal retractions, and an occasional deep cough. Upper airway transmitted noises were present, but otherwise the patient had clear lungs.
The patient’s vital signs at presentation were: blood pressure (BP), 85/55 mm Hg; heart rate (HR), 163 beats/min; respiratory rate (RR), 55 breaths/min; and temperature (T), 101.8°F. Oxygen saturation was 90% on room air. The patient’s mother wants to know how the respiratory distress will be fixed and is inquiring if they will have to stay in the hospital overnight.
Case 2
As work begins on the child described above, EMS brings in a 3-year-old girl who appears to be in moderate-severe respiratory distress. Her parents report that she started to drool earlier in the day followed by coughing and occasional gagging. Her parents relay that they thought the symptoms were because of post-nasal drip due to her cold, but the respiratory distress seems to be getting worse, and she now has very noisy breathing and is reluctant to lay down. Upon examination, both inspiratory and expiratory stridor is heard, and it is noted that moderate subcostal retractions are present when the patient is supine.
The patient’s vital signs at presentation were: BP, 89/58 mm Hg; HR, 144 beats/min; RR, 52 breaths/min; and T, 99.5°F. Oxygen saturation was 88% on room air. The nursing staff asked what to do next and why the 2 stridor cases are being managed so differently.
Stridor
Stridor is a high-pitched, harsh sound heard during respiration, predominantly during inspiration, as a result of turbulent air passage.1-3 Stridor is not a diagnosis in itself, but rather a sign of underlying acute or chronic etiology, which needs to be classified based on elicited history and examination.1,4 While the etiology of obstruction may be infectious, congenital, mechanical, or traumatic, the distinct features of the pediatric airway make respiratory failure an important concern independent of the underlying cause.
Anatomy
There are several anatomical differences unique to the pediatric airway that make children, especially infants under 1 year of age, more susceptible to airway obstruction.5,6The pediatric larynx is more anterior and superior and less fibrous than adults, thereby more compliant.6,7 The pediatric epiglottis is longer, omega-shaped, and softer, and the tongue is larger in comparison to the size of the oral cavity, resulting in obstructed airflow.6,7 Children also have a larger and more prominent occiput, causing mechanical obstruction by flexion of the neck when supine.6 While the cricoid cartilage was previously believed to be the narrowest portion of the airway, more recent measurement techniques have challenged this and shown that the glottic and subglottic areas may be narrower in children.7,8
Worsening obstruction resulting in a decreased airway radius leads to increased turbulence to air flow, which is explained by Poiseuille’s law.1,2,7 The resistance to airflow becomes inversely related to the fourth power of the radius, so even a small change in an already narrow pediatric airway can make a huge difference.5 In practice, this means 1 mm of mucosal edema will decrease the cross-sectional area of the airway by 75% and increase the resistance of airflow by a magnitude of 16 to 32 times depending on the level of turbulence.7The airway can be divided into extrathoracic and intrathoracic regions, separated by the vocal cords.13 Inspiratory stridor is typically due to extrathoracic obstruction and expiratory stridor is due to intrathoracic obstruction below the level of the cords. Biphasic stridor, however, may indicate a fixed obstruction at the level of the cords.2,3,5,9
Acute Differentials
The pediatric patient is at high risk for respiratory decompensation when the upper airway is acutely compromised. The history, physical examination, and phases of stridor can help determine the underlying diagnosis and definitive treatment plan.
Acute Croup
Acute croup (laryngotracheitis) is a clinical diagnosis based on acute onset of barky cough and inspiratory stridor.1 It is usually secondary to an infection, most commonly viral (parainfluenza virus), resulting in edema and increased secretions of the subglottic mucosa.7 The onset is typically preceded by upper respiratory illness (URI) symptoms, and is often worse at night or after waking from a nap.1 The peak incidence is between ages 6 months to 3 years. While croup is a clinical diagnosis, an anteroposterior X-ray will often show a steeple sign.7,10
Spasmodic Croup
Spasmodic croup is an atypical presentation usually seen in children 8 years or older without a preceding URI. Patients will wake up overnight with a harsh brass-like cough, stridor, and hoarse voice. The etiology is unclear, but often these patients have a history of atopy and respond in part to treatment with antihistamines.
Foreign Body Aspiration
All that is acutely stridulous is not croup. Stridor from foreign body aspiration is sudden in onset and children do not always present with a history suggestive of foreign body aspiration. Diagnosis requires a high index of suspicion because the event is often unwitnessed and typical patients are pre-verbal. The physical examination may reveal diminished air entry along with stridor. Diagnosis is made by obtaining an X-ray (lateral neck [Figure 1], lateral decubitus, or inspiratory/expiratory chest X-ray) or by bronchoscopy which is both diagnostic and therapeutic.7,10 Remember to always think of foreign body aspiration in children with acute stridor who have neither fever nor antecedent URI symptoms.
Bacterial Tracheitis
Bacterial tracheitis should be suspected in toxic appearing children who present with respiratory distress and stridor but who have a poor response to nebulized epinephrine. It is typically seen in toddlers and school-aged children, and like a viral tracheitis, presentation can be preceded by either URI or fever.10 Stridor is caused by subglottic edema and mucopurulent secretions in the airway.7,10 Infection is most commonly by S. aureus but initial antibiotic choice should be broad spectrum, and include a third-generation cephalosporin or beta-lactamase resistant penicillin.
Epiglottitis
Epiglottitis is edema of the epiglottis, most commonly secondary to bacterial infection. The epidemiology of epiglottitis has changed dramatically since widespread immunization for H. influenza with a significantly decreased incidence and change in the average age of presentation to 14.6 years (previously 5.6 years).7 The clinical course begins with sore throat, dysphonia, refusal to eat and progressive difficulty handling secretions with eventual drooling, stridor, tripoding, and toxic appearance. Epiglottitis can be differentiated from croup and bacterial tracheitis because presentation typically lacks a cough.7,10,11 Diagnosis is made either by direct visualization of the epiglottis or a lateral neck X-ray showing a ‘thumb print’ sign (Figure 2).12 Emergency department treatment is similar to the management of the child with a partial foreign body occlusion and focuses on maintaining the airway and minimizing anything that agitates the patient. Intravenous (IV) antibiotic coverage is similar to bacterial tracheitis (third-generation cephalosporin or a beta-lactamase resistant penicillin).
Retropharyngeal Abscess
The most common chief complaint of retropharyngeal abscess (RPA) is neck pain (38%) with fever. As such, it can clinically be mistaken for meningitis on initial presentation. Retropharyngeal abscess will present rarely with either stridor or associated respiratory distress, and it can also mimic croup on initial presentation. Physical examination findings which differentiate this entity include limited or painful neck extension (45%), torticollis (36.5%), and to a lesser extent limitation of neck flexion (12.5%).13 The median age at diagnosis is 36 months with 75% of patients less than 5 years. Typical presentation is insidious with fever and URI symptoms preceding onset. Diagnosis can be made with a lateral neck X-ray showing widening of the prevertebral space (Figure 3), but the gold standard diagnostic study is a computed tomography with contrast.14 Management is IV antibiotics covering aerobic and anaerobic bacteria (eg, ampicillin-sulbactam) ± surgical intervention.
Caustic Ingestion
Caustic ingestion is most commonly accidental and seen in children aged 12 months to 2 years. However, with recent fads, such as the “Tide Pod challenge” teenagers are also at risk. Airway compromise and stridor are secondary to mucosal injury and edema. Oral injury is not always a useful marker for significant distal injury. A complete evaluation of the upper airway and digestive tract within 48 hours after known/suspected caustic ingestion is recommended to assess full extent of damage.15
Chronic Differentials
Laryngomalacia
Laryngomalacia is a congenital weakness of laryngeal tissues, and it is the most common cause of both chronic stridor and neonatal stridor. It is characterized by progressive worsening of symptoms with crying/feeding and supine positioning. Diagnosis is made by bronchoscopy and management is conservative unless there are life threatening apneic or cyanotic events.7
Rings/Slings
There are many anatomic structures with the potential to cause extrinsic airway compression which present with stridor. This type of stridor is often biphasic. Examples include innominate artery compression, double aortic arch, aberrant subclavian artery, and pulmonary artery sling.7
Stridor presenting in children with a history of prematurity or prolonged intubation should raise concern for subglottic and tracheal stenosis.16
Evaluation
Regardless of the etiology of stridor, efforts should be made to keep the patient calm (ie, allow the parent to keep holding a young child, limit any examination not absolutely necessary). Much of the examination can be completed from a distance without disturbing the child.17 Observation of the inspiratory:expiratory (I:E) ratio can localize the level of airway obstruction. For example, an I:E ratio weighted toward a longer inspiration indicates an extrathoracic airway obstruction. Whereas an I:E with a prolonged expiratory phase is consistent with intrathoracic obstruction (eg, terminal bronchial obstruction).17
Another way to localize the level of obstruction is to look for changes in the voice; patients who present with a change in their voice have a subglottic partial obstruction such as croup. However, patients with a muffled voice or drooling have a supraglottic obstruction such as epiglottitis or RPA.17
Management
Management of stridor focuses on reducing airway obstruction, which is usually secondary to edema in the acute setting.
Viral Laryngotracheitis
Oral steroids are the mainstay of treatment. Research has shown dexamethasone is preferred over prednisolone.18-20 Steroids are not only useful in moderate to severe laryngotracheitis but also have a therapeutic role in children with mild laryngotracheitis.18 In hospital settings the parenteral formulation of dexamethasone can be safely given orally with good effect. There is no therapeutic advantage in acute laryngotracheitis to giving dexamethasone via either the IV or intramuscular route vs oral.21 In the outpatient setting, decadron tablets can be crushed and mixed in with a young child’s favorite soft food (eg, mashed potatoes or apple sauce). The authors recommend this strategy in lieu of prescribing dexamethasone suspension as its dilute concentration (1 mg/10 ml) results in a need for a child to receive a relatively large volume of a distasteful liquid. There is a wide therapeutic range of dexamethasone with studies documenting efficacy for laryngotracheitis in doses ranging from 0.15 mg/kg to 0.6 mg/kg. To date there are no large studies which demonstrate routine therapeutic utility of subsequent doses of dexamethasone. Nebulized budesonide (2.5 mg) can be given if oral steroids are not tolerated, however it is significantly more expensive.
Racemic epinephrine is the agent of choice for rapid onset of action in children who demonstrate stridor at rest. It causes vasoconstriction in the laryngeal mucosa, promotes bronchial smooth muscle relaxation, and thinning of bronchial secretions. It offers short-term relief of symptoms until steroids start to work. There is no rebound effect or worsening of symptoms once the epinephrine wears off, but children who receive this drug should be observed in the ED for a period of time (2-3 hours is standard of care in many hospitals) for return of symptoms.10,22 Patients who are persistently symptomatic 4 hours after administration of steroids or who require repeat doses of racemic epinephrine should be admitted for observation.10There are no contraindications to adjuvant treatments, such as antipyretics and non-sedating analgesics. Clinicians should maintain a high index of suspicion for anatomic airway anomalies that may need further evaluation/direct visualization in pediatric patients who present with repeated episodes of croup.10
Case Conclusions
Case 1
The 9-month-old boy with stridor was noted to have increased stridor while fussy, but even at rest some inspiratory stridor was present. A barky cough was noted in the examination room. The patient was placed on a monitor and a nebulized racemic epinephrine treatment was started. A single dose of oral dexamethasone was given shortly after presentation to the ED. Since the patient had inspiratory stridor at rest with associated tachypnea and hypoxia on initial presentation, a neck X-ray was obtained (Figure 4). Subglottic narrowing was identified on the imaging, but both the epiglottis and the prevertebral space were normal in appearance and no foreign bodies were visualized. The inspiratory stridor at rest, tachypnea, and mild hypoxia all improved after treatment, the patient was observed for 2 hours in the ED without recurrence of respiratory distress and was able to be discharged home with a diagnosis of acute croup.
Case 2
The 3-year-old girl was noted to be in significant positional respiratory distress, so the physician asked her parents to keep her calm in her position of comfort. She was calmly and quickly placed on a monitor with age-appropriate distraction techniques in place and advanced airway equipment at the bedside. A portable chest X-ray was obtained and revealed a coin was partially obstructing the trachea. Care was taken in the ED to avoid all interventions such as IV access that might upset the child so as not to inadvertently convert this partial airway obstruction to a complete obstruction. The otolaryngology team was called urgently, and the patient was transported to the operating room for foreign body removal in a controlled environment.
Cases
Case 1
It’s a busy shift on an unusually chilly and rainy July night. Emergency medical services (EMS) brings in a 9-month-old boy who woke up with a “squeaking” noise. His parents reported that he has had a fever, cough, rhinorrhea, and difficulty breathing for the past 2 days; however, they did not hear the noisy breathing until the night of presentation. When the patient is examined, it is noted that he has inspiratory stridor at rest, moderate subcostal retractions, and an occasional deep cough. Upper airway transmitted noises were present, but otherwise the patient had clear lungs.
The patient’s vital signs at presentation were: blood pressure (BP), 85/55 mm Hg; heart rate (HR), 163 beats/min; respiratory rate (RR), 55 breaths/min; and temperature (T), 101.8°F. Oxygen saturation was 90% on room air. The patient’s mother wants to know how the respiratory distress will be fixed and is inquiring if they will have to stay in the hospital overnight.
Case 2
As work begins on the child described above, EMS brings in a 3-year-old girl who appears to be in moderate-severe respiratory distress. Her parents report that she started to drool earlier in the day followed by coughing and occasional gagging. Her parents relay that they thought the symptoms were because of post-nasal drip due to her cold, but the respiratory distress seems to be getting worse, and she now has very noisy breathing and is reluctant to lay down. Upon examination, both inspiratory and expiratory stridor is heard, and it is noted that moderate subcostal retractions are present when the patient is supine.
The patient’s vital signs at presentation were: BP, 89/58 mm Hg; HR, 144 beats/min; RR, 52 breaths/min; and T, 99.5°F. Oxygen saturation was 88% on room air. The nursing staff asked what to do next and why the 2 stridor cases are being managed so differently.
Stridor
Stridor is a high-pitched, harsh sound heard during respiration, predominantly during inspiration, as a result of turbulent air passage.1-3 Stridor is not a diagnosis in itself, but rather a sign of underlying acute or chronic etiology, which needs to be classified based on elicited history and examination.1,4 While the etiology of obstruction may be infectious, congenital, mechanical, or traumatic, the distinct features of the pediatric airway make respiratory failure an important concern independent of the underlying cause.
Anatomy
There are several anatomical differences unique to the pediatric airway that make children, especially infants under 1 year of age, more susceptible to airway obstruction.5,6The pediatric larynx is more anterior and superior and less fibrous than adults, thereby more compliant.6,7 The pediatric epiglottis is longer, omega-shaped, and softer, and the tongue is larger in comparison to the size of the oral cavity, resulting in obstructed airflow.6,7 Children also have a larger and more prominent occiput, causing mechanical obstruction by flexion of the neck when supine.6 While the cricoid cartilage was previously believed to be the narrowest portion of the airway, more recent measurement techniques have challenged this and shown that the glottic and subglottic areas may be narrower in children.7,8
Worsening obstruction resulting in a decreased airway radius leads to increased turbulence to air flow, which is explained by Poiseuille’s law.1,2,7 The resistance to airflow becomes inversely related to the fourth power of the radius, so even a small change in an already narrow pediatric airway can make a huge difference.5 In practice, this means 1 mm of mucosal edema will decrease the cross-sectional area of the airway by 75% and increase the resistance of airflow by a magnitude of 16 to 32 times depending on the level of turbulence.7The airway can be divided into extrathoracic and intrathoracic regions, separated by the vocal cords.13 Inspiratory stridor is typically due to extrathoracic obstruction and expiratory stridor is due to intrathoracic obstruction below the level of the cords. Biphasic stridor, however, may indicate a fixed obstruction at the level of the cords.2,3,5,9
Acute Differentials
The pediatric patient is at high risk for respiratory decompensation when the upper airway is acutely compromised. The history, physical examination, and phases of stridor can help determine the underlying diagnosis and definitive treatment plan.
Acute Croup
Acute croup (laryngotracheitis) is a clinical diagnosis based on acute onset of barky cough and inspiratory stridor.1 It is usually secondary to an infection, most commonly viral (parainfluenza virus), resulting in edema and increased secretions of the subglottic mucosa.7 The onset is typically preceded by upper respiratory illness (URI) symptoms, and is often worse at night or after waking from a nap.1 The peak incidence is between ages 6 months to 3 years. While croup is a clinical diagnosis, an anteroposterior X-ray will often show a steeple sign.7,10
Spasmodic Croup
Spasmodic croup is an atypical presentation usually seen in children 8 years or older without a preceding URI. Patients will wake up overnight with a harsh brass-like cough, stridor, and hoarse voice. The etiology is unclear, but often these patients have a history of atopy and respond in part to treatment with antihistamines.
Foreign Body Aspiration
All that is acutely stridulous is not croup. Stridor from foreign body aspiration is sudden in onset and children do not always present with a history suggestive of foreign body aspiration. Diagnosis requires a high index of suspicion because the event is often unwitnessed and typical patients are pre-verbal. The physical examination may reveal diminished air entry along with stridor. Diagnosis is made by obtaining an X-ray (lateral neck [Figure 1], lateral decubitus, or inspiratory/expiratory chest X-ray) or by bronchoscopy which is both diagnostic and therapeutic.7,10 Remember to always think of foreign body aspiration in children with acute stridor who have neither fever nor antecedent URI symptoms.
Bacterial Tracheitis
Bacterial tracheitis should be suspected in toxic appearing children who present with respiratory distress and stridor but who have a poor response to nebulized epinephrine. It is typically seen in toddlers and school-aged children, and like a viral tracheitis, presentation can be preceded by either URI or fever.10 Stridor is caused by subglottic edema and mucopurulent secretions in the airway.7,10 Infection is most commonly by S. aureus but initial antibiotic choice should be broad spectrum, and include a third-generation cephalosporin or beta-lactamase resistant penicillin.
Epiglottitis
Epiglottitis is edema of the epiglottis, most commonly secondary to bacterial infection. The epidemiology of epiglottitis has changed dramatically since widespread immunization for H. influenza with a significantly decreased incidence and change in the average age of presentation to 14.6 years (previously 5.6 years).7 The clinical course begins with sore throat, dysphonia, refusal to eat and progressive difficulty handling secretions with eventual drooling, stridor, tripoding, and toxic appearance. Epiglottitis can be differentiated from croup and bacterial tracheitis because presentation typically lacks a cough.7,10,11 Diagnosis is made either by direct visualization of the epiglottis or a lateral neck X-ray showing a ‘thumb print’ sign (Figure 2).12 Emergency department treatment is similar to the management of the child with a partial foreign body occlusion and focuses on maintaining the airway and minimizing anything that agitates the patient. Intravenous (IV) antibiotic coverage is similar to bacterial tracheitis (third-generation cephalosporin or a beta-lactamase resistant penicillin).
Retropharyngeal Abscess
The most common chief complaint of retropharyngeal abscess (RPA) is neck pain (38%) with fever. As such, it can clinically be mistaken for meningitis on initial presentation. Retropharyngeal abscess will present rarely with either stridor or associated respiratory distress, and it can also mimic croup on initial presentation. Physical examination findings which differentiate this entity include limited or painful neck extension (45%), torticollis (36.5%), and to a lesser extent limitation of neck flexion (12.5%).13 The median age at diagnosis is 36 months with 75% of patients less than 5 years. Typical presentation is insidious with fever and URI symptoms preceding onset. Diagnosis can be made with a lateral neck X-ray showing widening of the prevertebral space (Figure 3), but the gold standard diagnostic study is a computed tomography with contrast.14 Management is IV antibiotics covering aerobic and anaerobic bacteria (eg, ampicillin-sulbactam) ± surgical intervention.
Caustic Ingestion
Caustic ingestion is most commonly accidental and seen in children aged 12 months to 2 years. However, with recent fads, such as the “Tide Pod challenge” teenagers are also at risk. Airway compromise and stridor are secondary to mucosal injury and edema. Oral injury is not always a useful marker for significant distal injury. A complete evaluation of the upper airway and digestive tract within 48 hours after known/suspected caustic ingestion is recommended to assess full extent of damage.15
Chronic Differentials
Laryngomalacia
Laryngomalacia is a congenital weakness of laryngeal tissues, and it is the most common cause of both chronic stridor and neonatal stridor. It is characterized by progressive worsening of symptoms with crying/feeding and supine positioning. Diagnosis is made by bronchoscopy and management is conservative unless there are life threatening apneic or cyanotic events.7
Rings/Slings
There are many anatomic structures with the potential to cause extrinsic airway compression which present with stridor. This type of stridor is often biphasic. Examples include innominate artery compression, double aortic arch, aberrant subclavian artery, and pulmonary artery sling.7
Stridor presenting in children with a history of prematurity or prolonged intubation should raise concern for subglottic and tracheal stenosis.16
Evaluation
Regardless of the etiology of stridor, efforts should be made to keep the patient calm (ie, allow the parent to keep holding a young child, limit any examination not absolutely necessary). Much of the examination can be completed from a distance without disturbing the child.17 Observation of the inspiratory:expiratory (I:E) ratio can localize the level of airway obstruction. For example, an I:E ratio weighted toward a longer inspiration indicates an extrathoracic airway obstruction. Whereas an I:E with a prolonged expiratory phase is consistent with intrathoracic obstruction (eg, terminal bronchial obstruction).17
Another way to localize the level of obstruction is to look for changes in the voice; patients who present with a change in their voice have a subglottic partial obstruction such as croup. However, patients with a muffled voice or drooling have a supraglottic obstruction such as epiglottitis or RPA.17
Management
Management of stridor focuses on reducing airway obstruction, which is usually secondary to edema in the acute setting.
Viral Laryngotracheitis
Oral steroids are the mainstay of treatment. Research has shown dexamethasone is preferred over prednisolone.18-20 Steroids are not only useful in moderate to severe laryngotracheitis but also have a therapeutic role in children with mild laryngotracheitis.18 In hospital settings the parenteral formulation of dexamethasone can be safely given orally with good effect. There is no therapeutic advantage in acute laryngotracheitis to giving dexamethasone via either the IV or intramuscular route vs oral.21 In the outpatient setting, decadron tablets can be crushed and mixed in with a young child’s favorite soft food (eg, mashed potatoes or apple sauce). The authors recommend this strategy in lieu of prescribing dexamethasone suspension as its dilute concentration (1 mg/10 ml) results in a need for a child to receive a relatively large volume of a distasteful liquid. There is a wide therapeutic range of dexamethasone with studies documenting efficacy for laryngotracheitis in doses ranging from 0.15 mg/kg to 0.6 mg/kg. To date there are no large studies which demonstrate routine therapeutic utility of subsequent doses of dexamethasone. Nebulized budesonide (2.5 mg) can be given if oral steroids are not tolerated, however it is significantly more expensive.
Racemic epinephrine is the agent of choice for rapid onset of action in children who demonstrate stridor at rest. It causes vasoconstriction in the laryngeal mucosa, promotes bronchial smooth muscle relaxation, and thinning of bronchial secretions. It offers short-term relief of symptoms until steroids start to work. There is no rebound effect or worsening of symptoms once the epinephrine wears off, but children who receive this drug should be observed in the ED for a period of time (2-3 hours is standard of care in many hospitals) for return of symptoms.10,22 Patients who are persistently symptomatic 4 hours after administration of steroids or who require repeat doses of racemic epinephrine should be admitted for observation.10There are no contraindications to adjuvant treatments, such as antipyretics and non-sedating analgesics. Clinicians should maintain a high index of suspicion for anatomic airway anomalies that may need further evaluation/direct visualization in pediatric patients who present with repeated episodes of croup.10
Case Conclusions
Case 1
The 9-month-old boy with stridor was noted to have increased stridor while fussy, but even at rest some inspiratory stridor was present. A barky cough was noted in the examination room. The patient was placed on a monitor and a nebulized racemic epinephrine treatment was started. A single dose of oral dexamethasone was given shortly after presentation to the ED. Since the patient had inspiratory stridor at rest with associated tachypnea and hypoxia on initial presentation, a neck X-ray was obtained (Figure 4). Subglottic narrowing was identified on the imaging, but both the epiglottis and the prevertebral space were normal in appearance and no foreign bodies were visualized. The inspiratory stridor at rest, tachypnea, and mild hypoxia all improved after treatment, the patient was observed for 2 hours in the ED without recurrence of respiratory distress and was able to be discharged home with a diagnosis of acute croup.
Case 2
The 3-year-old girl was noted to be in significant positional respiratory distress, so the physician asked her parents to keep her calm in her position of comfort. She was calmly and quickly placed on a monitor with age-appropriate distraction techniques in place and advanced airway equipment at the bedside. A portable chest X-ray was obtained and revealed a coin was partially obstructing the trachea. Care was taken in the ED to avoid all interventions such as IV access that might upset the child so as not to inadvertently convert this partial airway obstruction to a complete obstruction. The otolaryngology team was called urgently, and the patient was transported to the operating room for foreign body removal in a controlled environment.
1. Bjornson CL, Johnson DW. Croup. Lancet. 2008;371(9609):329-339. doi:10.1016/S0140-6736(08)60170-1.
2. Escobar ML, Needleman J. Stridor. Pediatr Rev. 2015;36(3):135-137. doi:10.1542/pir.36-3-135.
3. Pfleger A, Eber E. Assessment and causes of stridor. Paediatr Respir Rev. 2016;18:64-72. doi:10.1016/j.prrv.2015.10.003.
4. Boudewyns A, Claes J, Van de Heyning P. Clinical practice: an approach to stridor in infants and children. Eur J Pediatr. 2010;169(2):135-141. doi:10.1007/s00431-009-1044-7.
5. Kelley PB, Simon JE. Racemic epinephrine use in croup and disposition. Am J Emerg Med. 1992;10(3):181-183.
6. Mandal A, Kabra SK, Lodha R. Upper airway obstruction in children. Indian J Pediatr. 2015;82(8):737-744.
7. Marchese A, Langhan ML. Management of airway obstruction and stridor in pediatric patients. Pediatr Emerg Med Pract. 2017;14(11):1-24.
8. Wani TM, Bissonnette B, Rafiq Malik M, et al. Age-based analysis of pediatric upper airway dimensions using computed tomography imaging. Pediatr Pulmonol. 2016;51(3):267-271. doi:10.1002/ppul.23232.
9. Donaldson D, Poleski D, Knipple E, et al. Intramuscular versus oral dexamethasone for the treatment of moderate-to-severe croup: a randomized, double-blind trial. Acad Emerg Med. 2003;10(1):16-21.
10. Boudewyns A, Claes J, Van de Heyning P. Clinical practice: an approach to stridor in infants and children. Eur J Pediatr. 2010;169(2):135-141. doi:10.1007/s00431-009-1044-7.
11. Tibballs J, Watson T. Symptoms and signs differentiating croup and epiglottitis. J Paediatr Child Health. 2011;47(3):77-82. doi:10.1111/j.1440-1754.2010.01892.x.
12. Sobol SE, Zapata S. Epiglottitis and croup. Otolaryngol Clin North Am. 2008;41(3):551-566. doi:10.1016/j.otc.2008.01.012.
13. Craig FW, Schunk JE. Retropharyngeal abscess in children: clinical presentation, utility of imaging, and current management. Pediatrics. 2003;111(6 Pt 1):1394-1398.
14. Roberson DW. Pediatric retropharyngeal abscesses. Clin Pediatr Emerg Med. 2004;5(1):37-40.
15. Riffat F, Cheng A. Pediatric caustic ingestion: 50 consecutive cases and a review of the literature. Dis Esophagus. 2009;22(1):89-94. doi:10.1111/j.1442-2050.2008.00867.x.
16. Daniel SJ. The upper airway: congenital malformations. Paediatr Respir Revi. 2006;7 Suppl 1:S260-S263.
17. Arutyunyan H, Spangler M. Pediatric upper airway obstruction. Peds RAP Web site. https://www.hippoed.com/peds/rap/episode/pedsrapfebruary/pediatricupper. Published February 2018. Accessed August 31, 2018.
18. Bjornson CL, Klassen TP, Williamson J, et al; Pediatric Emergency Research Canada Network. A randomized trial of a single dose of oral dexamethasone for mild croup. N Engl J Med. 2004;351(13):1306-1313.
19. Fifoot AA, Ting JY. Comparison between single-dose oral prednisolone and oral dexamethasone in the treatment of croup: a randomized, double-blinded clinical trial. Emerg Med Australas. 2007;19(1):51-58.
20. Sparrow A, Geelhoed G. Prednisolone versus dexamethasone in croup: a randomised equivalence trial. Arch Dis Child. 2005;91(7):580-583.
21. Donaldson D, Poleski D, Knipple E, et al. Intramuscular versus oral dexamethasone for the treatment of moderate-to-severe croup: a randomized, double-blind trial. Acad Emerg Med. 2003;10(1):16-21.
22. Kelley PB, Simon JE. Racemic epinephrine use in croup and disposition. Am J Emerg Med. 1992;10(3):181-183.
23. Yamamoto LG. Test your skill in reading pediatric lateral necks. University of Hawaii Web site. https://www.hawaii.edu/medicine/pediatrics/pemxray/v2c20.html. Accessed September 13, 2018.
1. Bjornson CL, Johnson DW. Croup. Lancet. 2008;371(9609):329-339. doi:10.1016/S0140-6736(08)60170-1.
2. Escobar ML, Needleman J. Stridor. Pediatr Rev. 2015;36(3):135-137. doi:10.1542/pir.36-3-135.
3. Pfleger A, Eber E. Assessment and causes of stridor. Paediatr Respir Rev. 2016;18:64-72. doi:10.1016/j.prrv.2015.10.003.
4. Boudewyns A, Claes J, Van de Heyning P. Clinical practice: an approach to stridor in infants and children. Eur J Pediatr. 2010;169(2):135-141. doi:10.1007/s00431-009-1044-7.
5. Kelley PB, Simon JE. Racemic epinephrine use in croup and disposition. Am J Emerg Med. 1992;10(3):181-183.
6. Mandal A, Kabra SK, Lodha R. Upper airway obstruction in children. Indian J Pediatr. 2015;82(8):737-744.
7. Marchese A, Langhan ML. Management of airway obstruction and stridor in pediatric patients. Pediatr Emerg Med Pract. 2017;14(11):1-24.
8. Wani TM, Bissonnette B, Rafiq Malik M, et al. Age-based analysis of pediatric upper airway dimensions using computed tomography imaging. Pediatr Pulmonol. 2016;51(3):267-271. doi:10.1002/ppul.23232.
9. Donaldson D, Poleski D, Knipple E, et al. Intramuscular versus oral dexamethasone for the treatment of moderate-to-severe croup: a randomized, double-blind trial. Acad Emerg Med. 2003;10(1):16-21.
10. Boudewyns A, Claes J, Van de Heyning P. Clinical practice: an approach to stridor in infants and children. Eur J Pediatr. 2010;169(2):135-141. doi:10.1007/s00431-009-1044-7.
11. Tibballs J, Watson T. Symptoms and signs differentiating croup and epiglottitis. J Paediatr Child Health. 2011;47(3):77-82. doi:10.1111/j.1440-1754.2010.01892.x.
12. Sobol SE, Zapata S. Epiglottitis and croup. Otolaryngol Clin North Am. 2008;41(3):551-566. doi:10.1016/j.otc.2008.01.012.
13. Craig FW, Schunk JE. Retropharyngeal abscess in children: clinical presentation, utility of imaging, and current management. Pediatrics. 2003;111(6 Pt 1):1394-1398.
14. Roberson DW. Pediatric retropharyngeal abscesses. Clin Pediatr Emerg Med. 2004;5(1):37-40.
15. Riffat F, Cheng A. Pediatric caustic ingestion: 50 consecutive cases and a review of the literature. Dis Esophagus. 2009;22(1):89-94. doi:10.1111/j.1442-2050.2008.00867.x.
16. Daniel SJ. The upper airway: congenital malformations. Paediatr Respir Revi. 2006;7 Suppl 1:S260-S263.
17. Arutyunyan H, Spangler M. Pediatric upper airway obstruction. Peds RAP Web site. https://www.hippoed.com/peds/rap/episode/pedsrapfebruary/pediatricupper. Published February 2018. Accessed August 31, 2018.
18. Bjornson CL, Klassen TP, Williamson J, et al; Pediatric Emergency Research Canada Network. A randomized trial of a single dose of oral dexamethasone for mild croup. N Engl J Med. 2004;351(13):1306-1313.
19. Fifoot AA, Ting JY. Comparison between single-dose oral prednisolone and oral dexamethasone in the treatment of croup: a randomized, double-blinded clinical trial. Emerg Med Australas. 2007;19(1):51-58.
20. Sparrow A, Geelhoed G. Prednisolone versus dexamethasone in croup: a randomised equivalence trial. Arch Dis Child. 2005;91(7):580-583.
21. Donaldson D, Poleski D, Knipple E, et al. Intramuscular versus oral dexamethasone for the treatment of moderate-to-severe croup: a randomized, double-blind trial. Acad Emerg Med. 2003;10(1):16-21.
22. Kelley PB, Simon JE. Racemic epinephrine use in croup and disposition. Am J Emerg Med. 1992;10(3):181-183.
23. Yamamoto LG. Test your skill in reading pediatric lateral necks. University of Hawaii Web site. https://www.hawaii.edu/medicine/pediatrics/pemxray/v2c20.html. Accessed September 13, 2018.
IL inhibitor options move psoriasis treatment forward
Psoriasis patients have many options, and more are on the way, according to J. Mark Jackson, MD, of the University of Louisville, Ky.
“Know the information regarding each [treatment] to best care for your patients,” Dr. Jackson said in a presentation at the annual Coastal Dermatology Symposium.
Dr. Jackson particularly addressed the interleukin (IL)-17 inhibitors (brodalumab, ixekizumab, and secukinumab) and the IL-23 inhibitors (guselkumab, risankizumab, and tildrakizumab).
Complete clearance rates can reach 50% and higher over the long term when treating patients with IL-17 inhibitors, but patients must maintain regular dosing to maintain a response, he said.
Overall, comparisons of IL-17 inhibitors with etanercept, adalimumab, and ustekinumab “demonstrate better efficacy with no evidence of compromising safety,” he noted.
For example, secukinumab demonstrated significantly superior results when compared with ustekinumab in a randomized trial (J Am Acad Dermatol. 2015;73: 400-9). After 16 weeks of treatment, 79% of secukinumab patients achieved a 90% reduction in Psoriasis Area and Severity Index score (PASI 90) versus 58% of ustekinumab patients, he said, and the drug safety profile was consistent with the pivotal phase 3 studies of secukinumab.
Concerns persist about increased risk of inflammatory bowel disease, Crohn’s disease, and ulcerative colitis in patients taking secukinumab and other IL-17 inhibitors, but data indicate that rates are low. The risk is low “and may be related to psoriasis and not the therapy,” he explained.
Ixekizumab has been associated with more injection site reactions than secukinumab, but these tend to be mild, Dr. Jackson said. Advantages of ixekizumab are that it works quickly and has demonstrated effectiveness against genital, palmoplantar, scalp, and nail psoriasis, he added.
Brodalumab also works quickly, but it has the unique inclusion of a Risk Evaluation and Mitigation Strategies (REMS) program because of suicidal ideation and behavior in clinical trials, he noted, adding that there are more data showing rates are low and the REMS program is easier to deal with than the isotretinoin REMS. The increased risk of superficial Staphylococcus and Candida infections are noted on IL-17 inhibitor labels, but this has not been a significant issue in trials or clinical practice, he said.
What is also exciting about the IL-17 inhibitors are the approvals of ixekizumab and secukinumab for patients with psoriatic arthritis (PsA), with both agents demonstrating the ability to inhibit the structural progression of joint damage over time, Dr. Jackson commented. These data seem to be on par with that of the TNF-inhibitors, although time will tell how this bears out clinically, he noted.
IL-23 inhibitors guselkumab, tildrakizumab, and risankizumab (not yet approved) have shown similar effectiveness and are well tolerated by patients, with few injection site reactions or adverse events reported, Dr. Jackson said. The dosing regimens of each of these drugs, administered subcutaneously, are easy to follow: Treatment starts with an initial dose of either 100 mg (guselkumab and tildrakizumab) or 150 mg (risankizumab), which is followed by doses at 4 weeks and then doses every 8 weeks (guselkumab) or 12 weeks (tildrakizumab and risankizumab).
For example, in a comparison study of risankizumab with a dosage of 150 mg subcutaneously at week 0, 4, then every 12 weeks, 75% of risankizumab patients achieved PASI 90 at 16 weeks and 82% at 52 weeks, compared with 42% and 44%, respectively, for adalimumab patients.
In addition, the IL-23 inhibitors have demonstrated some benefits for PsA patients in clinical trials, but they are not currently indicated for PsA, he said.
Dr. Jackson disclosed having received research, honoraria, consulting, and/or other support from AbbVie, Accuitis, Aclaris, Celgene, Dr. Reddy’s, Galderma, Janssen, Lilly, Medimetriks, Novartis, Pfizer, Promius, Ralexar, Sienna, and TopMD.
The meeting is jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are both owned by Frontline Medical Communications.
Psoriasis patients have many options, and more are on the way, according to J. Mark Jackson, MD, of the University of Louisville, Ky.
“Know the information regarding each [treatment] to best care for your patients,” Dr. Jackson said in a presentation at the annual Coastal Dermatology Symposium.
Dr. Jackson particularly addressed the interleukin (IL)-17 inhibitors (brodalumab, ixekizumab, and secukinumab) and the IL-23 inhibitors (guselkumab, risankizumab, and tildrakizumab).
Complete clearance rates can reach 50% and higher over the long term when treating patients with IL-17 inhibitors, but patients must maintain regular dosing to maintain a response, he said.
Overall, comparisons of IL-17 inhibitors with etanercept, adalimumab, and ustekinumab “demonstrate better efficacy with no evidence of compromising safety,” he noted.
For example, secukinumab demonstrated significantly superior results when compared with ustekinumab in a randomized trial (J Am Acad Dermatol. 2015;73: 400-9). After 16 weeks of treatment, 79% of secukinumab patients achieved a 90% reduction in Psoriasis Area and Severity Index score (PASI 90) versus 58% of ustekinumab patients, he said, and the drug safety profile was consistent with the pivotal phase 3 studies of secukinumab.
Concerns persist about increased risk of inflammatory bowel disease, Crohn’s disease, and ulcerative colitis in patients taking secukinumab and other IL-17 inhibitors, but data indicate that rates are low. The risk is low “and may be related to psoriasis and not the therapy,” he explained.
Ixekizumab has been associated with more injection site reactions than secukinumab, but these tend to be mild, Dr. Jackson said. Advantages of ixekizumab are that it works quickly and has demonstrated effectiveness against genital, palmoplantar, scalp, and nail psoriasis, he added.
Brodalumab also works quickly, but it has the unique inclusion of a Risk Evaluation and Mitigation Strategies (REMS) program because of suicidal ideation and behavior in clinical trials, he noted, adding that there are more data showing rates are low and the REMS program is easier to deal with than the isotretinoin REMS. The increased risk of superficial Staphylococcus and Candida infections are noted on IL-17 inhibitor labels, but this has not been a significant issue in trials or clinical practice, he said.
What is also exciting about the IL-17 inhibitors are the approvals of ixekizumab and secukinumab for patients with psoriatic arthritis (PsA), with both agents demonstrating the ability to inhibit the structural progression of joint damage over time, Dr. Jackson commented. These data seem to be on par with that of the TNF-inhibitors, although time will tell how this bears out clinically, he noted.
IL-23 inhibitors guselkumab, tildrakizumab, and risankizumab (not yet approved) have shown similar effectiveness and are well tolerated by patients, with few injection site reactions or adverse events reported, Dr. Jackson said. The dosing regimens of each of these drugs, administered subcutaneously, are easy to follow: Treatment starts with an initial dose of either 100 mg (guselkumab and tildrakizumab) or 150 mg (risankizumab), which is followed by doses at 4 weeks and then doses every 8 weeks (guselkumab) or 12 weeks (tildrakizumab and risankizumab).
For example, in a comparison study of risankizumab with a dosage of 150 mg subcutaneously at week 0, 4, then every 12 weeks, 75% of risankizumab patients achieved PASI 90 at 16 weeks and 82% at 52 weeks, compared with 42% and 44%, respectively, for adalimumab patients.
In addition, the IL-23 inhibitors have demonstrated some benefits for PsA patients in clinical trials, but they are not currently indicated for PsA, he said.
Dr. Jackson disclosed having received research, honoraria, consulting, and/or other support from AbbVie, Accuitis, Aclaris, Celgene, Dr. Reddy’s, Galderma, Janssen, Lilly, Medimetriks, Novartis, Pfizer, Promius, Ralexar, Sienna, and TopMD.
The meeting is jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are both owned by Frontline Medical Communications.
Psoriasis patients have many options, and more are on the way, according to J. Mark Jackson, MD, of the University of Louisville, Ky.
“Know the information regarding each [treatment] to best care for your patients,” Dr. Jackson said in a presentation at the annual Coastal Dermatology Symposium.
Dr. Jackson particularly addressed the interleukin (IL)-17 inhibitors (brodalumab, ixekizumab, and secukinumab) and the IL-23 inhibitors (guselkumab, risankizumab, and tildrakizumab).
Complete clearance rates can reach 50% and higher over the long term when treating patients with IL-17 inhibitors, but patients must maintain regular dosing to maintain a response, he said.
Overall, comparisons of IL-17 inhibitors with etanercept, adalimumab, and ustekinumab “demonstrate better efficacy with no evidence of compromising safety,” he noted.
For example, secukinumab demonstrated significantly superior results when compared with ustekinumab in a randomized trial (J Am Acad Dermatol. 2015;73: 400-9). After 16 weeks of treatment, 79% of secukinumab patients achieved a 90% reduction in Psoriasis Area and Severity Index score (PASI 90) versus 58% of ustekinumab patients, he said, and the drug safety profile was consistent with the pivotal phase 3 studies of secukinumab.
Concerns persist about increased risk of inflammatory bowel disease, Crohn’s disease, and ulcerative colitis in patients taking secukinumab and other IL-17 inhibitors, but data indicate that rates are low. The risk is low “and may be related to psoriasis and not the therapy,” he explained.
Ixekizumab has been associated with more injection site reactions than secukinumab, but these tend to be mild, Dr. Jackson said. Advantages of ixekizumab are that it works quickly and has demonstrated effectiveness against genital, palmoplantar, scalp, and nail psoriasis, he added.
Brodalumab also works quickly, but it has the unique inclusion of a Risk Evaluation and Mitigation Strategies (REMS) program because of suicidal ideation and behavior in clinical trials, he noted, adding that there are more data showing rates are low and the REMS program is easier to deal with than the isotretinoin REMS. The increased risk of superficial Staphylococcus and Candida infections are noted on IL-17 inhibitor labels, but this has not been a significant issue in trials or clinical practice, he said.
What is also exciting about the IL-17 inhibitors are the approvals of ixekizumab and secukinumab for patients with psoriatic arthritis (PsA), with both agents demonstrating the ability to inhibit the structural progression of joint damage over time, Dr. Jackson commented. These data seem to be on par with that of the TNF-inhibitors, although time will tell how this bears out clinically, he noted.
IL-23 inhibitors guselkumab, tildrakizumab, and risankizumab (not yet approved) have shown similar effectiveness and are well tolerated by patients, with few injection site reactions or adverse events reported, Dr. Jackson said. The dosing regimens of each of these drugs, administered subcutaneously, are easy to follow: Treatment starts with an initial dose of either 100 mg (guselkumab and tildrakizumab) or 150 mg (risankizumab), which is followed by doses at 4 weeks and then doses every 8 weeks (guselkumab) or 12 weeks (tildrakizumab and risankizumab).
For example, in a comparison study of risankizumab with a dosage of 150 mg subcutaneously at week 0, 4, then every 12 weeks, 75% of risankizumab patients achieved PASI 90 at 16 weeks and 82% at 52 weeks, compared with 42% and 44%, respectively, for adalimumab patients.
In addition, the IL-23 inhibitors have demonstrated some benefits for PsA patients in clinical trials, but they are not currently indicated for PsA, he said.
Dr. Jackson disclosed having received research, honoraria, consulting, and/or other support from AbbVie, Accuitis, Aclaris, Celgene, Dr. Reddy’s, Galderma, Janssen, Lilly, Medimetriks, Novartis, Pfizer, Promius, Ralexar, Sienna, and TopMD.
The meeting is jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are both owned by Frontline Medical Communications.
FROM THE COASTAL DERMATOLOGY SYMPOSIUM
The patient who doesn’t like you
About a year ago, I had a patient come in who didn’t like me.
It seemed like a normal visit. My secretary had him fill out the usual forms and copied his insurance cards, and I took him back to my office. We do this many times, every day.
He came back to my office, and I asked him what brought him to my care.
Instead of starting his medical history, though, he immediately gave me a long list of complaints. He didn’t like my appearance. Or my secretary. Or my forms. Or us asking if he’d had any previous tests. Or the parking at my office. Or the phone system. Or a coffee stain in my building’s elevator carpeting.
A whole list of stuff, none actually related to his reason for coming in. I let him rant for a minute, thinking maybe he’d get to the point, but he just kept getting angrier and bringing up more grievances.
I finally interrupted him and said, “Sir, if you’re unhappy with me, you are welcome to end the appointment and leave now.” He told me he wasn’t going to pay for the visit (not that I would have charged him for it) and stomped out. My secretary shredded his info. There’s always other stuff that needs my attention, so I busied myself with that until the next appointment arrived.
Twenty years ago this probably would have really upset me. But today? Not at all.
Like most other doctors, I want to help people. I enjoy doing that. It’s why I’m here. But I’ve also learned that there are some people I’ll never be able to work with under any circumstances. Some will just never like me as a physician, my casual appearance, or small practice.
People like this guy happen a few times a year. Experience teaches that you can’t be everyone’s doctor, can’t make everyone happy, and can’t have them all like you. If they don’t, that’s part of life. You can’t predict interpersonal chemistry and worrying about such things isn’t good for your blood pressure. You can’t change others.
Ironically, the same gentleman called recently, saying he needed to get in with me now. My secretary called him back, reminded him of what happened last year and suggested he go elsewhere.
His response? “I didn’t like your office then and still don’t.”
I’m okay with that. You can’t please everyone. Sometimes it’s not even worth trying.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
About a year ago, I had a patient come in who didn’t like me.
It seemed like a normal visit. My secretary had him fill out the usual forms and copied his insurance cards, and I took him back to my office. We do this many times, every day.
He came back to my office, and I asked him what brought him to my care.
Instead of starting his medical history, though, he immediately gave me a long list of complaints. He didn’t like my appearance. Or my secretary. Or my forms. Or us asking if he’d had any previous tests. Or the parking at my office. Or the phone system. Or a coffee stain in my building’s elevator carpeting.
A whole list of stuff, none actually related to his reason for coming in. I let him rant for a minute, thinking maybe he’d get to the point, but he just kept getting angrier and bringing up more grievances.
I finally interrupted him and said, “Sir, if you’re unhappy with me, you are welcome to end the appointment and leave now.” He told me he wasn’t going to pay for the visit (not that I would have charged him for it) and stomped out. My secretary shredded his info. There’s always other stuff that needs my attention, so I busied myself with that until the next appointment arrived.
Twenty years ago this probably would have really upset me. But today? Not at all.
Like most other doctors, I want to help people. I enjoy doing that. It’s why I’m here. But I’ve also learned that there are some people I’ll never be able to work with under any circumstances. Some will just never like me as a physician, my casual appearance, or small practice.
People like this guy happen a few times a year. Experience teaches that you can’t be everyone’s doctor, can’t make everyone happy, and can’t have them all like you. If they don’t, that’s part of life. You can’t predict interpersonal chemistry and worrying about such things isn’t good for your blood pressure. You can’t change others.
Ironically, the same gentleman called recently, saying he needed to get in with me now. My secretary called him back, reminded him of what happened last year and suggested he go elsewhere.
His response? “I didn’t like your office then and still don’t.”
I’m okay with that. You can’t please everyone. Sometimes it’s not even worth trying.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
About a year ago, I had a patient come in who didn’t like me.
It seemed like a normal visit. My secretary had him fill out the usual forms and copied his insurance cards, and I took him back to my office. We do this many times, every day.
He came back to my office, and I asked him what brought him to my care.
Instead of starting his medical history, though, he immediately gave me a long list of complaints. He didn’t like my appearance. Or my secretary. Or my forms. Or us asking if he’d had any previous tests. Or the parking at my office. Or the phone system. Or a coffee stain in my building’s elevator carpeting.
A whole list of stuff, none actually related to his reason for coming in. I let him rant for a minute, thinking maybe he’d get to the point, but he just kept getting angrier and bringing up more grievances.
I finally interrupted him and said, “Sir, if you’re unhappy with me, you are welcome to end the appointment and leave now.” He told me he wasn’t going to pay for the visit (not that I would have charged him for it) and stomped out. My secretary shredded his info. There’s always other stuff that needs my attention, so I busied myself with that until the next appointment arrived.
Twenty years ago this probably would have really upset me. But today? Not at all.
Like most other doctors, I want to help people. I enjoy doing that. It’s why I’m here. But I’ve also learned that there are some people I’ll never be able to work with under any circumstances. Some will just never like me as a physician, my casual appearance, or small practice.
People like this guy happen a few times a year. Experience teaches that you can’t be everyone’s doctor, can’t make everyone happy, and can’t have them all like you. If they don’t, that’s part of life. You can’t predict interpersonal chemistry and worrying about such things isn’t good for your blood pressure. You can’t change others.
Ironically, the same gentleman called recently, saying he needed to get in with me now. My secretary called him back, reminded him of what happened last year and suggested he go elsewhere.
His response? “I didn’t like your office then and still don’t.”
I’m okay with that. You can’t please everyone. Sometimes it’s not even worth trying.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Sweat Regeneration Following CO2 Fractionated Laser Therapy
To the Editor:
It is not uncommon for patients with extensive dermal scarring to overheat due to the inability to regulate body temperature through evaporative heat loss, as lack of perspiration in areas of prior full-thickness skin injury is well known. One of the authors (C.M.H.) previously reported a case of a patient with considerable hypertrophic scarring after surviving an episode of toxic epidermal necrolysis that was likely precipitated by lamotrigine.1 The patient initially presented to our clinic in consultation for laser therapy to improve the pliability and cosmetic appearance of the scars; however, approximately 3 weeks after initiating treatment with a fractional CO2 laser, the patient noticed perspiration in areas where she once lacked the ability to perspire as well as improved functionality.1 It was speculated that scar remodeling stimulated by the CO2 fractional laser allowed new connections to form between eccrine ducts in the dermis and epidermis.2
These findings are even more notable in light of a study by Rittié et al3 that suggested the primary appendages of the skin involved in human wound healing are the eccrine sweat glands. The investigators were able to demonstrate that eccrine sweat glands are major contributors in reepithelialization and wound healing in humans; therefore, it is possible that stimulating these glands with the CO2 laser may promote enhanced reepithelialization in addition to the reestablishment of perspiration and wound healing.3 Considering inadequate wound repair represents a substantial disturbance to the patient and health care system, this finding offers promise as a potential means to decrease morbidity in patients with dermal scarring from burns and traumatic injuries. We have since evaluated and treated 3 patients who demonstrated sweat regeneration following treatment with the fractional CO2 laser (Table).
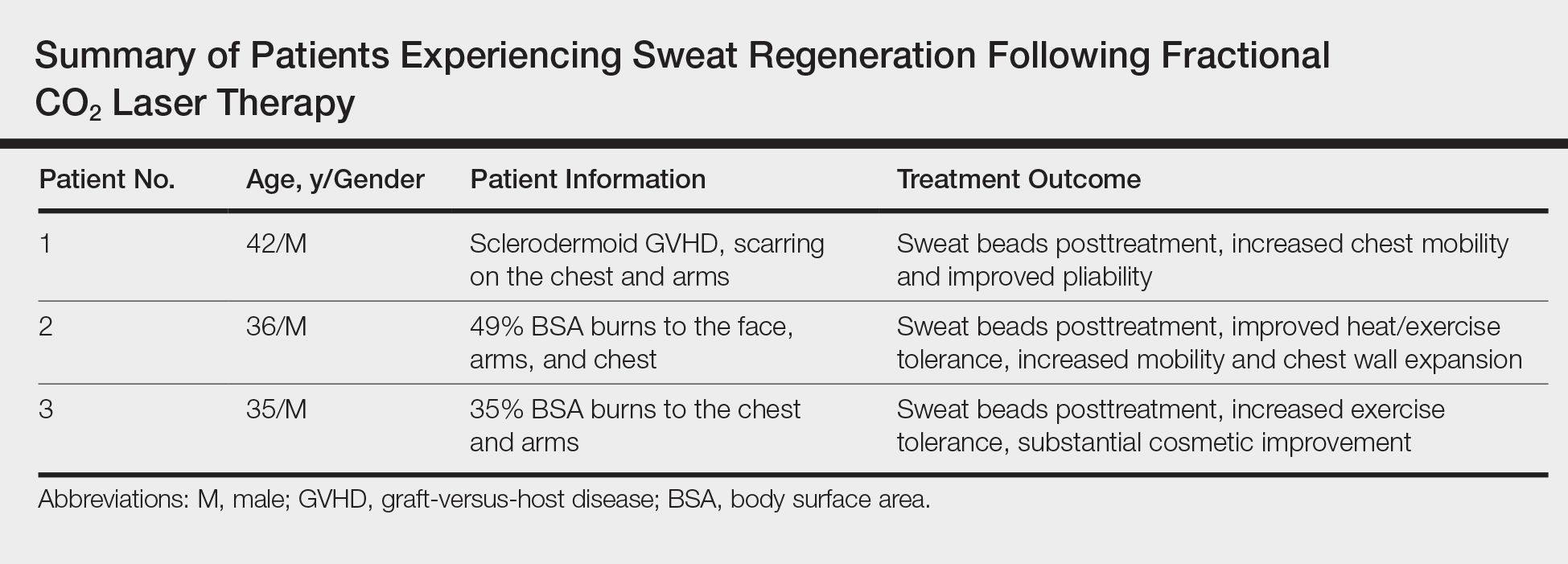
A 42-year-old man was our first patient to demonstrate functional scar improvement following bone marrow transplant for acute lymphoblastic leukemia complicated by chronic sclerodermoid graft-versus-host disease and subsequent extensive scarring on the chest and arms. Approximately 2 weeks after the first treatment with the fractional CO2 laser, the patient began to notice the presence of sweat beads in the treated areas. In addition to the reestablishment of perspiration, the patient had perceived increased mobility with improved pliability and “softness” (as described by family members) in treated areas likely related to scar remodeling.
A 36-year-old wounded army veteran presented with burns to the face, arms, and chest affecting 49% of the body surface area. After only 1 treatment, the patient reported that he could subjectively tolerate 10°F more ambient temperature and work all day outside in south Texas when heat intolerance previously would allow him to work only 2 to 3 hours. Additionally, he noted increased mobility and chest wall expansion, which in combination contributed to overall increased exercise tolerance and enhanced quality of life.
A 35-year-old US Marine and firefighter with burns primarily on the chest and arms involving 35% body surface area experienced increased exercise tolerance and sweat regeneration, particularly on the chest after a single treatment with the fractional CO2 laser but continued to experience improvement after a total of 3 treatments. Additionally, the cosmetic improvement was so substantial that the physician (C.M.H) had to review older photographs to verify the location of the scars.
We have now treated 3 patients with various mechanisms of injury and extensive scarring who noticed improved heat tolerance from sweat regeneration following fractional CO2 laser therapy. At this point, we only have anecdotal evidence of subjective functional improvement, and further research is warranted to elucidate the exact mechanism of action to support our findings.
- Neiner J, Whittemore D, Hivnor C. Buried alive: functional eccrine coils buried under scar tissue? J Am Acad Dermatol. 2011;65:661-663.
- Waibel J, Beer K, Narurkar V, et al. Preliminary observations on fractional ablative resurfacing devices: clinical impressions. J Drugs Dermatol. 2009;8:481-485.
- Rittié L, Sachs D, Orringer J, et al. Eccrine sweat glands are major contributors to reepithelialization of human wounds. Am J Pathol. 2013;1:163-171.
To the Editor:
It is not uncommon for patients with extensive dermal scarring to overheat due to the inability to regulate body temperature through evaporative heat loss, as lack of perspiration in areas of prior full-thickness skin injury is well known. One of the authors (C.M.H.) previously reported a case of a patient with considerable hypertrophic scarring after surviving an episode of toxic epidermal necrolysis that was likely precipitated by lamotrigine.1 The patient initially presented to our clinic in consultation for laser therapy to improve the pliability and cosmetic appearance of the scars; however, approximately 3 weeks after initiating treatment with a fractional CO2 laser, the patient noticed perspiration in areas where she once lacked the ability to perspire as well as improved functionality.1 It was speculated that scar remodeling stimulated by the CO2 fractional laser allowed new connections to form between eccrine ducts in the dermis and epidermis.2
These findings are even more notable in light of a study by Rittié et al3 that suggested the primary appendages of the skin involved in human wound healing are the eccrine sweat glands. The investigators were able to demonstrate that eccrine sweat glands are major contributors in reepithelialization and wound healing in humans; therefore, it is possible that stimulating these glands with the CO2 laser may promote enhanced reepithelialization in addition to the reestablishment of perspiration and wound healing.3 Considering inadequate wound repair represents a substantial disturbance to the patient and health care system, this finding offers promise as a potential means to decrease morbidity in patients with dermal scarring from burns and traumatic injuries. We have since evaluated and treated 3 patients who demonstrated sweat regeneration following treatment with the fractional CO2 laser (Table).

A 42-year-old man was our first patient to demonstrate functional scar improvement following bone marrow transplant for acute lymphoblastic leukemia complicated by chronic sclerodermoid graft-versus-host disease and subsequent extensive scarring on the chest and arms. Approximately 2 weeks after the first treatment with the fractional CO2 laser, the patient began to notice the presence of sweat beads in the treated areas. In addition to the reestablishment of perspiration, the patient had perceived increased mobility with improved pliability and “softness” (as described by family members) in treated areas likely related to scar remodeling.
A 36-year-old wounded army veteran presented with burns to the face, arms, and chest affecting 49% of the body surface area. After only 1 treatment, the patient reported that he could subjectively tolerate 10°F more ambient temperature and work all day outside in south Texas when heat intolerance previously would allow him to work only 2 to 3 hours. Additionally, he noted increased mobility and chest wall expansion, which in combination contributed to overall increased exercise tolerance and enhanced quality of life.
A 35-year-old US Marine and firefighter with burns primarily on the chest and arms involving 35% body surface area experienced increased exercise tolerance and sweat regeneration, particularly on the chest after a single treatment with the fractional CO2 laser but continued to experience improvement after a total of 3 treatments. Additionally, the cosmetic improvement was so substantial that the physician (C.M.H) had to review older photographs to verify the location of the scars.
We have now treated 3 patients with various mechanisms of injury and extensive scarring who noticed improved heat tolerance from sweat regeneration following fractional CO2 laser therapy. At this point, we only have anecdotal evidence of subjective functional improvement, and further research is warranted to elucidate the exact mechanism of action to support our findings.
To the Editor:
It is not uncommon for patients with extensive dermal scarring to overheat due to the inability to regulate body temperature through evaporative heat loss, as lack of perspiration in areas of prior full-thickness skin injury is well known. One of the authors (C.M.H.) previously reported a case of a patient with considerable hypertrophic scarring after surviving an episode of toxic epidermal necrolysis that was likely precipitated by lamotrigine.1 The patient initially presented to our clinic in consultation for laser therapy to improve the pliability and cosmetic appearance of the scars; however, approximately 3 weeks after initiating treatment with a fractional CO2 laser, the patient noticed perspiration in areas where she once lacked the ability to perspire as well as improved functionality.1 It was speculated that scar remodeling stimulated by the CO2 fractional laser allowed new connections to form between eccrine ducts in the dermis and epidermis.2
These findings are even more notable in light of a study by Rittié et al3 that suggested the primary appendages of the skin involved in human wound healing are the eccrine sweat glands. The investigators were able to demonstrate that eccrine sweat glands are major contributors in reepithelialization and wound healing in humans; therefore, it is possible that stimulating these glands with the CO2 laser may promote enhanced reepithelialization in addition to the reestablishment of perspiration and wound healing.3 Considering inadequate wound repair represents a substantial disturbance to the patient and health care system, this finding offers promise as a potential means to decrease morbidity in patients with dermal scarring from burns and traumatic injuries. We have since evaluated and treated 3 patients who demonstrated sweat regeneration following treatment with the fractional CO2 laser (Table).

A 42-year-old man was our first patient to demonstrate functional scar improvement following bone marrow transplant for acute lymphoblastic leukemia complicated by chronic sclerodermoid graft-versus-host disease and subsequent extensive scarring on the chest and arms. Approximately 2 weeks after the first treatment with the fractional CO2 laser, the patient began to notice the presence of sweat beads in the treated areas. In addition to the reestablishment of perspiration, the patient had perceived increased mobility with improved pliability and “softness” (as described by family members) in treated areas likely related to scar remodeling.
A 36-year-old wounded army veteran presented with burns to the face, arms, and chest affecting 49% of the body surface area. After only 1 treatment, the patient reported that he could subjectively tolerate 10°F more ambient temperature and work all day outside in south Texas when heat intolerance previously would allow him to work only 2 to 3 hours. Additionally, he noted increased mobility and chest wall expansion, which in combination contributed to overall increased exercise tolerance and enhanced quality of life.
A 35-year-old US Marine and firefighter with burns primarily on the chest and arms involving 35% body surface area experienced increased exercise tolerance and sweat regeneration, particularly on the chest after a single treatment with the fractional CO2 laser but continued to experience improvement after a total of 3 treatments. Additionally, the cosmetic improvement was so substantial that the physician (C.M.H) had to review older photographs to verify the location of the scars.
We have now treated 3 patients with various mechanisms of injury and extensive scarring who noticed improved heat tolerance from sweat regeneration following fractional CO2 laser therapy. At this point, we only have anecdotal evidence of subjective functional improvement, and further research is warranted to elucidate the exact mechanism of action to support our findings.
- Neiner J, Whittemore D, Hivnor C. Buried alive: functional eccrine coils buried under scar tissue? J Am Acad Dermatol. 2011;65:661-663.
- Waibel J, Beer K, Narurkar V, et al. Preliminary observations on fractional ablative resurfacing devices: clinical impressions. J Drugs Dermatol. 2009;8:481-485.
- Rittié L, Sachs D, Orringer J, et al. Eccrine sweat glands are major contributors to reepithelialization of human wounds. Am J Pathol. 2013;1:163-171.
- Neiner J, Whittemore D, Hivnor C. Buried alive: functional eccrine coils buried under scar tissue? J Am Acad Dermatol. 2011;65:661-663.
- Waibel J, Beer K, Narurkar V, et al. Preliminary observations on fractional ablative resurfacing devices: clinical impressions. J Drugs Dermatol. 2009;8:481-485.
- Rittié L, Sachs D, Orringer J, et al. Eccrine sweat glands are major contributors to reepithelialization of human wounds. Am J Pathol. 2013;1:163-171.
Practice Points
- Treatment of dermal scarring with fractional CO2 laser may contribute to eccrine sweat gland regeneration during the remodeling process in addition to increased skin pliability.
- Sweat regeneration has been demonstrated following treatment with fractional CO2 laser in patients with extensive scarring; this case shows sweat regeneration secondary to burns and chronic sclerodermoid graft-versus-host disease.
New valve to treat emphysema-related hyperinflation demonstrates efficacy
PARIS – A patients with hyperinflation provides acceptable safety and clinically significant improvement in lung function at 12 months, according to results from a multicenter trial presented as a late-breaker at the annual congress of the European Respiratory Society.
Six-month results have been presented previously, but the 12-month results suggest that lung volume reduction associated with placement of the valves provides “durable effectiveness in appropriately selected hyperinflated emphysema patients,” according to Gerard Criner, MD, chair of the thoracic medicine department at Temple University, Philadelphia.
In this randomized controlled trial, called EMPROVE, 172 emphysema patients with severe dyspnea were randomized in a 2:1 fashion to receive a proprietary endobronchial valve or medical therapy alone. The valve, marketed under the brand name Spiration Valve System (SVS), is currently indicated for the treatment of air leaks after lung surgery.
“The valve serves to block airflow from edematous lungs with the objective of blocking hyperinflation and improving lung function,” Dr. Criner explained. The valves are retrievable, if necessary, with bronchoscopy.
High resolution computed tomography (HRCT) was used to identify emphysema obstruction and target valve placement to the most diseased lobes. The average number of valves placed per patient was slightly less than four. Most of the valves (70%) were placed in an upper lobe.
When reported at 6 months, the responder rate, defined as at least 15% improvement in forced expiratory volume in 1 second (FEV1) was 36.8% and 10% in the SVS and control groups, respectively, a difference of 25.7% that Dr. Criner reported as statistically significant although he did not provide P values. At 12 months, the rates were 37.2% and 5.1%, respectively, demonstrating a persistent effect.
Thoracic adverse events were higher at both 6 months (31% vs. 11.9%) and 12 months (21.4% vs. 10.6%) in the treatment group relative to the control group. At 6 months, pneumothorax, which Dr. Criner characterized as “a recognized marker of target lobe volume reduction,” was the only event that occurred significantly more commonly (14.2% vs. 0%) in the SVS group.
Between 6 and 12 months, there were no pneumothorax events in either arm. The higher numerical rates of thoracic adverse events in the treatment arm were acute exacerbations (13.6% vs. 8.5%) and pneumonia in nontreated lobes (7.8% vs. 2.1%), but these rates were not statistically different.
At 6 months, there was a numerically higher rate of all-cause mortality in the treatment group (5.3% vs. 1.7%) but the rate was numerically lower between 6 and 12 months (2.9% vs. 6.4%). The differences were not significantly different at either time point or overall.
A significant reduction in hyperinflation favoring valve placement was accompanied by improvement in objective measures of lung function, such as FEV1, and dyspnea, as measured with the Modified Medical Research Council (mMRC) Dyspnea Scale, at 6 and 12 months. These improvements translated into persistent quality of life benefits as measured with the St. George Respiratory Questionnaire (SGRQ). At 12 months, the SGRQ changes from baseline were a 5.5-point reduction and a four-point gain in the treatment and control groups, respectively. This absolute difference of 9.5 points is slightly more modest than the 13-point difference at 6 months (–8 vs. +5 points), but demonstrates a durable effect, Dr. Criner reported.
According to Dr. Criner, EMPROVE reinforces the principle that HRCT is effective “for selecting the lobe for therapy and which patients may benefit,” but the most important message from the 12-month results is persistent clinical benefit.
The endobronchial values “provide statistically and clinically meaningful improvements in FEV1, reductions in lobe volume and dyspnea, and improvements in quality of life with an acceptable safety profile,” Dr. Criner reported.
On June 29, 2018, the Food and Drug Administration approved the first endobronchial valve for treatment of emphysema. This valve, marketed under the name Zephyr, was approved on the basis of the pivotal LIBERATE trial, also led by Dr. Criner and published earlier this year (Am J Respir Crit Care Med. 2018 May 22. doi: 10.1164/rccm.201803-0590OC.). The results of EMPROVE are consistent with previous evidence that a one-way valve, by preventing air from entering diseased lobes of patients with emphysema to exacerbate hyperinflation, improves lung function and reduces clinical symptoms.
Dr. Criner reports financial relationships with Olympus, the sponsor of this trial.
PARIS – A patients with hyperinflation provides acceptable safety and clinically significant improvement in lung function at 12 months, according to results from a multicenter trial presented as a late-breaker at the annual congress of the European Respiratory Society.
Six-month results have been presented previously, but the 12-month results suggest that lung volume reduction associated with placement of the valves provides “durable effectiveness in appropriately selected hyperinflated emphysema patients,” according to Gerard Criner, MD, chair of the thoracic medicine department at Temple University, Philadelphia.
In this randomized controlled trial, called EMPROVE, 172 emphysema patients with severe dyspnea were randomized in a 2:1 fashion to receive a proprietary endobronchial valve or medical therapy alone. The valve, marketed under the brand name Spiration Valve System (SVS), is currently indicated for the treatment of air leaks after lung surgery.
“The valve serves to block airflow from edematous lungs with the objective of blocking hyperinflation and improving lung function,” Dr. Criner explained. The valves are retrievable, if necessary, with bronchoscopy.
High resolution computed tomography (HRCT) was used to identify emphysema obstruction and target valve placement to the most diseased lobes. The average number of valves placed per patient was slightly less than four. Most of the valves (70%) were placed in an upper lobe.
When reported at 6 months, the responder rate, defined as at least 15% improvement in forced expiratory volume in 1 second (FEV1) was 36.8% and 10% in the SVS and control groups, respectively, a difference of 25.7% that Dr. Criner reported as statistically significant although he did not provide P values. At 12 months, the rates were 37.2% and 5.1%, respectively, demonstrating a persistent effect.
Thoracic adverse events were higher at both 6 months (31% vs. 11.9%) and 12 months (21.4% vs. 10.6%) in the treatment group relative to the control group. At 6 months, pneumothorax, which Dr. Criner characterized as “a recognized marker of target lobe volume reduction,” was the only event that occurred significantly more commonly (14.2% vs. 0%) in the SVS group.
Between 6 and 12 months, there were no pneumothorax events in either arm. The higher numerical rates of thoracic adverse events in the treatment arm were acute exacerbations (13.6% vs. 8.5%) and pneumonia in nontreated lobes (7.8% vs. 2.1%), but these rates were not statistically different.
At 6 months, there was a numerically higher rate of all-cause mortality in the treatment group (5.3% vs. 1.7%) but the rate was numerically lower between 6 and 12 months (2.9% vs. 6.4%). The differences were not significantly different at either time point or overall.
A significant reduction in hyperinflation favoring valve placement was accompanied by improvement in objective measures of lung function, such as FEV1, and dyspnea, as measured with the Modified Medical Research Council (mMRC) Dyspnea Scale, at 6 and 12 months. These improvements translated into persistent quality of life benefits as measured with the St. George Respiratory Questionnaire (SGRQ). At 12 months, the SGRQ changes from baseline were a 5.5-point reduction and a four-point gain in the treatment and control groups, respectively. This absolute difference of 9.5 points is slightly more modest than the 13-point difference at 6 months (–8 vs. +5 points), but demonstrates a durable effect, Dr. Criner reported.
According to Dr. Criner, EMPROVE reinforces the principle that HRCT is effective “for selecting the lobe for therapy and which patients may benefit,” but the most important message from the 12-month results is persistent clinical benefit.
The endobronchial values “provide statistically and clinically meaningful improvements in FEV1, reductions in lobe volume and dyspnea, and improvements in quality of life with an acceptable safety profile,” Dr. Criner reported.
On June 29, 2018, the Food and Drug Administration approved the first endobronchial valve for treatment of emphysema. This valve, marketed under the name Zephyr, was approved on the basis of the pivotal LIBERATE trial, also led by Dr. Criner and published earlier this year (Am J Respir Crit Care Med. 2018 May 22. doi: 10.1164/rccm.201803-0590OC.). The results of EMPROVE are consistent with previous evidence that a one-way valve, by preventing air from entering diseased lobes of patients with emphysema to exacerbate hyperinflation, improves lung function and reduces clinical symptoms.
Dr. Criner reports financial relationships with Olympus, the sponsor of this trial.
PARIS – A patients with hyperinflation provides acceptable safety and clinically significant improvement in lung function at 12 months, according to results from a multicenter trial presented as a late-breaker at the annual congress of the European Respiratory Society.
Six-month results have been presented previously, but the 12-month results suggest that lung volume reduction associated with placement of the valves provides “durable effectiveness in appropriately selected hyperinflated emphysema patients,” according to Gerard Criner, MD, chair of the thoracic medicine department at Temple University, Philadelphia.
In this randomized controlled trial, called EMPROVE, 172 emphysema patients with severe dyspnea were randomized in a 2:1 fashion to receive a proprietary endobronchial valve or medical therapy alone. The valve, marketed under the brand name Spiration Valve System (SVS), is currently indicated for the treatment of air leaks after lung surgery.
“The valve serves to block airflow from edematous lungs with the objective of blocking hyperinflation and improving lung function,” Dr. Criner explained. The valves are retrievable, if necessary, with bronchoscopy.
High resolution computed tomography (HRCT) was used to identify emphysema obstruction and target valve placement to the most diseased lobes. The average number of valves placed per patient was slightly less than four. Most of the valves (70%) were placed in an upper lobe.
When reported at 6 months, the responder rate, defined as at least 15% improvement in forced expiratory volume in 1 second (FEV1) was 36.8% and 10% in the SVS and control groups, respectively, a difference of 25.7% that Dr. Criner reported as statistically significant although he did not provide P values. At 12 months, the rates were 37.2% and 5.1%, respectively, demonstrating a persistent effect.
Thoracic adverse events were higher at both 6 months (31% vs. 11.9%) and 12 months (21.4% vs. 10.6%) in the treatment group relative to the control group. At 6 months, pneumothorax, which Dr. Criner characterized as “a recognized marker of target lobe volume reduction,” was the only event that occurred significantly more commonly (14.2% vs. 0%) in the SVS group.
Between 6 and 12 months, there were no pneumothorax events in either arm. The higher numerical rates of thoracic adverse events in the treatment arm were acute exacerbations (13.6% vs. 8.5%) and pneumonia in nontreated lobes (7.8% vs. 2.1%), but these rates were not statistically different.
At 6 months, there was a numerically higher rate of all-cause mortality in the treatment group (5.3% vs. 1.7%) but the rate was numerically lower between 6 and 12 months (2.9% vs. 6.4%). The differences were not significantly different at either time point or overall.
A significant reduction in hyperinflation favoring valve placement was accompanied by improvement in objective measures of lung function, such as FEV1, and dyspnea, as measured with the Modified Medical Research Council (mMRC) Dyspnea Scale, at 6 and 12 months. These improvements translated into persistent quality of life benefits as measured with the St. George Respiratory Questionnaire (SGRQ). At 12 months, the SGRQ changes from baseline were a 5.5-point reduction and a four-point gain in the treatment and control groups, respectively. This absolute difference of 9.5 points is slightly more modest than the 13-point difference at 6 months (–8 vs. +5 points), but demonstrates a durable effect, Dr. Criner reported.
According to Dr. Criner, EMPROVE reinforces the principle that HRCT is effective “for selecting the lobe for therapy and which patients may benefit,” but the most important message from the 12-month results is persistent clinical benefit.
The endobronchial values “provide statistically and clinically meaningful improvements in FEV1, reductions in lobe volume and dyspnea, and improvements in quality of life with an acceptable safety profile,” Dr. Criner reported.
On June 29, 2018, the Food and Drug Administration approved the first endobronchial valve for treatment of emphysema. This valve, marketed under the name Zephyr, was approved on the basis of the pivotal LIBERATE trial, also led by Dr. Criner and published earlier this year (Am J Respir Crit Care Med. 2018 May 22. doi: 10.1164/rccm.201803-0590OC.). The results of EMPROVE are consistent with previous evidence that a one-way valve, by preventing air from entering diseased lobes of patients with emphysema to exacerbate hyperinflation, improves lung function and reduces clinical symptoms.
Dr. Criner reports financial relationships with Olympus, the sponsor of this trial.
REPORTING FROM THE ERS CONGRESS 2018
Key clinical point: A one-way valve developed for postoperative air leaks was found effective against emphysema-related hyperinflation.
Major finding: At 12 months, the FEV1 response rate (at least a 15% improvement) was observed in 37.2% of valve-treated patients and 5.1% of controls.
Study details: Randomized, multicenter trial.
Disclosures: Dr. Criner reports financial relationships with Olympus, the sponsor of this trial.
Source: European Respiratory Society 2018 International Congress.
Growth on nose
The FP made the presumptive diagnosis of a nodular basal cell carcinoma.
He explained the importance of performing a biopsy and obtained informed consent. On the same day of the patient’s visit, he injected 1% lidocaine with epinephrine under the lesion with a single stick of a 30 gauge needle. He knew that it was safe to use epinephrine on the nose, and that it would prevent excessive bleeding during the biopsy. Contrary to the myth frequently taught in medical school, the nose is a very vascular area. The physician performed a shave biopsy that removed the top of the lesion flush with the skin around it. (See the Watch & Learn video on “Shave biopsy.”)
The pathology report came back as a nodular basal cell carcinoma. On the following visit, the physician recommended Mohs surgery as a way to preserve the vital anatomy of the nasal ala and achieve the highest cure rate.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP made the presumptive diagnosis of a nodular basal cell carcinoma.
He explained the importance of performing a biopsy and obtained informed consent. On the same day of the patient’s visit, he injected 1% lidocaine with epinephrine under the lesion with a single stick of a 30 gauge needle. He knew that it was safe to use epinephrine on the nose, and that it would prevent excessive bleeding during the biopsy. Contrary to the myth frequently taught in medical school, the nose is a very vascular area. The physician performed a shave biopsy that removed the top of the lesion flush with the skin around it. (See the Watch & Learn video on “Shave biopsy.”)
The pathology report came back as a nodular basal cell carcinoma. On the following visit, the physician recommended Mohs surgery as a way to preserve the vital anatomy of the nasal ala and achieve the highest cure rate.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The FP made the presumptive diagnosis of a nodular basal cell carcinoma.
He explained the importance of performing a biopsy and obtained informed consent. On the same day of the patient’s visit, he injected 1% lidocaine with epinephrine under the lesion with a single stick of a 30 gauge needle. He knew that it was safe to use epinephrine on the nose, and that it would prevent excessive bleeding during the biopsy. Contrary to the myth frequently taught in medical school, the nose is a very vascular area. The physician performed a shave biopsy that removed the top of the lesion flush with the skin around it. (See the Watch & Learn video on “Shave biopsy.”)
The pathology report came back as a nodular basal cell carcinoma. On the following visit, the physician recommended Mohs surgery as a way to preserve the vital anatomy of the nasal ala and achieve the highest cure rate.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Ultrasound can’t rule out pulmonary embolism in the ED
SAN DIEGO – In the ICU, ultrasound has been shown to reduce the need for CT to evaluate potential pulmonary embolism. But in the ED, this strategy hasn’t worked out so far, according to Joseph Brown, MD, of the department of emergency medicine at the University of California, San Francisco.

Based on the data so far, the ED patients were less likely than the ICU patients to have another etiology identified on ultrasound that explained their symptoms. Further, ultrasound alone missed small subsegmental pulmonary emboli that were detected on subsequent CT scans in 2 of 11 patients.
The study is continuing, and Dr. Brown explains in this interview how ultrasound might be combined with other risk stratification measures to safely achieve reductions in CT scans.
SAN DIEGO – In the ICU, ultrasound has been shown to reduce the need for CT to evaluate potential pulmonary embolism. But in the ED, this strategy hasn’t worked out so far, according to Joseph Brown, MD, of the department of emergency medicine at the University of California, San Francisco.

Based on the data so far, the ED patients were less likely than the ICU patients to have another etiology identified on ultrasound that explained their symptoms. Further, ultrasound alone missed small subsegmental pulmonary emboli that were detected on subsequent CT scans in 2 of 11 patients.
The study is continuing, and Dr. Brown explains in this interview how ultrasound might be combined with other risk stratification measures to safely achieve reductions in CT scans.
SAN DIEGO – In the ICU, ultrasound has been shown to reduce the need for CT to evaluate potential pulmonary embolism. But in the ED, this strategy hasn’t worked out so far, according to Joseph Brown, MD, of the department of emergency medicine at the University of California, San Francisco.

Based on the data so far, the ED patients were less likely than the ICU patients to have another etiology identified on ultrasound that explained their symptoms. Further, ultrasound alone missed small subsegmental pulmonary emboli that were detected on subsequent CT scans in 2 of 11 patients.
The study is continuing, and Dr. Brown explains in this interview how ultrasound might be combined with other risk stratification measures to safely achieve reductions in CT scans.
REPORTING FROM ACEP18
This is a drill
She had fallen on a garden implement, lacerating her superficial femoral artery. She used her cell phone to call 911. Thanks to an alert EMS crew and the Stop the Bleed training they recently received, a tourniquet was placed without delay. She got to our trauma bay in about 15 minutes after the tourniquet was applied. Although the patient made it abundantly clear that she was in pain, she was stable with moderate tachycardia and a good blood pressure.
Our trauma team leaped into action. The leader took report from the EMS while two nurses and a second surgeon assessed the patient, got her clothes cut off, and applied monitors. A third nurse got a second IV going. Primary survey was done in less than 90 seconds. The patient then underwent a focused exam including a log roll for back injuries.
The leg wound was still oozing a bit, so a second tourniquet was called for and pressure applied until it could be acquired. The patient was given 5 mg of morphine sulfate, which calmed her down a bit. Labs and x-rays were done quickly. The nursing staff suggested a tetanus booster, and the second surgeon who had gotten a basic past medical history suggested vancomycin since the patient said she was allergic to penicillin. Fifteen minutes after she hit the trauma bay, she was on her way to the OR for exploration, debridement, and vascular repair of her injury.
This was all done by four M2 medical students and five N4 nursing students, none of whom had had previous experience with this type of trauma patient
The students were managing this trauma situation in the simulation center of their medical school with four staff watching. This was their second run through for the afternoon. At debriefing, they compared their work on the first trauma of the day (a stab wound to the right chest) to their second attempt. They were satisfied with their efforts and so were we, the faculty who ran the simulation. Comparing their response to those I’ve seen in real life, I’d say these students understood their roles and responsibilities as well as the sort of thrown-together teams I’ve seen at places where trauma is not the main focus. While these young men and women are in the early stage of training and not ready for a real-world trauma emergency, they have gained knowledge about this kind of situation that I didn’t see until I was in residency and beyond. The times they are a-changing.
A couple of days later I was in Rochester, Minn., attending an American College of Surgeons Advanced Education Institute (ACS/AEI) course on simulations. At the end of that course, we participants were challenged by a manikin in extremis. Everyone there was an expert, had an advanced degree, had some experience in simulation, or were surgeons interested in simulation. I found that, even though this was a simulation and the patient was only a pretend human being, my adrenal cortex performed almost as if I were doing a real resuscitation. Previous training I’d had on teamwork, crew resource management, and ACLS all kicked in, and we got it done. But interestingly, we weren’t perfect. We debriefed and found that, even at our level of experience and training, a simple simulation could be very instructive. Seeing/doing is believing.
High-tech skills in high-risk occupations are well served by simulation training. Much of the airline piloting training is done by simulation. It works well for aviation, nuclear reactors, high voltage line work, and medicine. Most of these disciplines have embraced simulation as an essential part of training. Simulation is part of many surgical training programs, but it has other uses.
When was the last time you practiced a trauma resuscitation, an ultrasound fine-needle biopsy, laparoscopic maneuvers, or an unusual technique that you seldom perform, but when needed, must be pulled off very well? Most of us taking this simulation course agreed that time, money, and ego may get in the way of maintaining those skills for those rare instances when they are needed. Surgeons might want to consider simulation to keep some of our rarely used skills from getting rusty.
If you’re going to make a costly error, I would very much like you to do it on a piece of plastic, not on a patient. There are no consequences for messing up a procedure on a manikin and this kind of practice might teach you something critical. Practicing reduces stress and improves the performance of those placed on the spot by real-life events. Do you think Captain “Sully” Sullenberger could have landed that airliner in the Hudson River safely if he hadn’t practiced with countless mind-numbingly complex simulations? Sure, luck plays a part, and innate ability plays a part. But skill, knowledge, and practice are your best bet when all the eyes in the room swivel to you in a moment of crisis.
You may think that simulators have to cost $100,000 and be completely realistic to do the job. That’s not true. A banana, orange, or stick of butter can be fabulous sims for a med student. Felt and cardboard can make a realistic cricothyroidotomy model.
Surgeons all over the country are using simulation training to learn how to be better without getting real blood on their shoes. If you haven’t participated in a training simulation recently, I double-dog dare you to try it and tell me you found it without merit. The ACS Surgical Simulation Summit is being held in March 2019 in Chicago. You might want to check that out.
Dr. Hughes is clinical professor in the department of surgery and director of medical education at the University of Kansas School of Medicine, Salina, and Coeditor of ACS Surgery News.
She had fallen on a garden implement, lacerating her superficial femoral artery. She used her cell phone to call 911. Thanks to an alert EMS crew and the Stop the Bleed training they recently received, a tourniquet was placed without delay. She got to our trauma bay in about 15 minutes after the tourniquet was applied. Although the patient made it abundantly clear that she was in pain, she was stable with moderate tachycardia and a good blood pressure.
Our trauma team leaped into action. The leader took report from the EMS while two nurses and a second surgeon assessed the patient, got her clothes cut off, and applied monitors. A third nurse got a second IV going. Primary survey was done in less than 90 seconds. The patient then underwent a focused exam including a log roll for back injuries.
The leg wound was still oozing a bit, so a second tourniquet was called for and pressure applied until it could be acquired. The patient was given 5 mg of morphine sulfate, which calmed her down a bit. Labs and x-rays were done quickly. The nursing staff suggested a tetanus booster, and the second surgeon who had gotten a basic past medical history suggested vancomycin since the patient said she was allergic to penicillin. Fifteen minutes after she hit the trauma bay, she was on her way to the OR for exploration, debridement, and vascular repair of her injury.
This was all done by four M2 medical students and five N4 nursing students, none of whom had had previous experience with this type of trauma patient
The students were managing this trauma situation in the simulation center of their medical school with four staff watching. This was their second run through for the afternoon. At debriefing, they compared their work on the first trauma of the day (a stab wound to the right chest) to their second attempt. They were satisfied with their efforts and so were we, the faculty who ran the simulation. Comparing their response to those I’ve seen in real life, I’d say these students understood their roles and responsibilities as well as the sort of thrown-together teams I’ve seen at places where trauma is not the main focus. While these young men and women are in the early stage of training and not ready for a real-world trauma emergency, they have gained knowledge about this kind of situation that I didn’t see until I was in residency and beyond. The times they are a-changing.
A couple of days later I was in Rochester, Minn., attending an American College of Surgeons Advanced Education Institute (ACS/AEI) course on simulations. At the end of that course, we participants were challenged by a manikin in extremis. Everyone there was an expert, had an advanced degree, had some experience in simulation, or were surgeons interested in simulation. I found that, even though this was a simulation and the patient was only a pretend human being, my adrenal cortex performed almost as if I were doing a real resuscitation. Previous training I’d had on teamwork, crew resource management, and ACLS all kicked in, and we got it done. But interestingly, we weren’t perfect. We debriefed and found that, even at our level of experience and training, a simple simulation could be very instructive. Seeing/doing is believing.
High-tech skills in high-risk occupations are well served by simulation training. Much of the airline piloting training is done by simulation. It works well for aviation, nuclear reactors, high voltage line work, and medicine. Most of these disciplines have embraced simulation as an essential part of training. Simulation is part of many surgical training programs, but it has other uses.
When was the last time you practiced a trauma resuscitation, an ultrasound fine-needle biopsy, laparoscopic maneuvers, or an unusual technique that you seldom perform, but when needed, must be pulled off very well? Most of us taking this simulation course agreed that time, money, and ego may get in the way of maintaining those skills for those rare instances when they are needed. Surgeons might want to consider simulation to keep some of our rarely used skills from getting rusty.
If you’re going to make a costly error, I would very much like you to do it on a piece of plastic, not on a patient. There are no consequences for messing up a procedure on a manikin and this kind of practice might teach you something critical. Practicing reduces stress and improves the performance of those placed on the spot by real-life events. Do you think Captain “Sully” Sullenberger could have landed that airliner in the Hudson River safely if he hadn’t practiced with countless mind-numbingly complex simulations? Sure, luck plays a part, and innate ability plays a part. But skill, knowledge, and practice are your best bet when all the eyes in the room swivel to you in a moment of crisis.
You may think that simulators have to cost $100,000 and be completely realistic to do the job. That’s not true. A banana, orange, or stick of butter can be fabulous sims for a med student. Felt and cardboard can make a realistic cricothyroidotomy model.
Surgeons all over the country are using simulation training to learn how to be better without getting real blood on their shoes. If you haven’t participated in a training simulation recently, I double-dog dare you to try it and tell me you found it without merit. The ACS Surgical Simulation Summit is being held in March 2019 in Chicago. You might want to check that out.
Dr. Hughes is clinical professor in the department of surgery and director of medical education at the University of Kansas School of Medicine, Salina, and Coeditor of ACS Surgery News.
She had fallen on a garden implement, lacerating her superficial femoral artery. She used her cell phone to call 911. Thanks to an alert EMS crew and the Stop the Bleed training they recently received, a tourniquet was placed without delay. She got to our trauma bay in about 15 minutes after the tourniquet was applied. Although the patient made it abundantly clear that she was in pain, she was stable with moderate tachycardia and a good blood pressure.
Our trauma team leaped into action. The leader took report from the EMS while two nurses and a second surgeon assessed the patient, got her clothes cut off, and applied monitors. A third nurse got a second IV going. Primary survey was done in less than 90 seconds. The patient then underwent a focused exam including a log roll for back injuries.
The leg wound was still oozing a bit, so a second tourniquet was called for and pressure applied until it could be acquired. The patient was given 5 mg of morphine sulfate, which calmed her down a bit. Labs and x-rays were done quickly. The nursing staff suggested a tetanus booster, and the second surgeon who had gotten a basic past medical history suggested vancomycin since the patient said she was allergic to penicillin. Fifteen minutes after she hit the trauma bay, she was on her way to the OR for exploration, debridement, and vascular repair of her injury.
This was all done by four M2 medical students and five N4 nursing students, none of whom had had previous experience with this type of trauma patient
The students were managing this trauma situation in the simulation center of their medical school with four staff watching. This was their second run through for the afternoon. At debriefing, they compared their work on the first trauma of the day (a stab wound to the right chest) to their second attempt. They were satisfied with their efforts and so were we, the faculty who ran the simulation. Comparing their response to those I’ve seen in real life, I’d say these students understood their roles and responsibilities as well as the sort of thrown-together teams I’ve seen at places where trauma is not the main focus. While these young men and women are in the early stage of training and not ready for a real-world trauma emergency, they have gained knowledge about this kind of situation that I didn’t see until I was in residency and beyond. The times they are a-changing.
A couple of days later I was in Rochester, Minn., attending an American College of Surgeons Advanced Education Institute (ACS/AEI) course on simulations. At the end of that course, we participants were challenged by a manikin in extremis. Everyone there was an expert, had an advanced degree, had some experience in simulation, or were surgeons interested in simulation. I found that, even though this was a simulation and the patient was only a pretend human being, my adrenal cortex performed almost as if I were doing a real resuscitation. Previous training I’d had on teamwork, crew resource management, and ACLS all kicked in, and we got it done. But interestingly, we weren’t perfect. We debriefed and found that, even at our level of experience and training, a simple simulation could be very instructive. Seeing/doing is believing.
High-tech skills in high-risk occupations are well served by simulation training. Much of the airline piloting training is done by simulation. It works well for aviation, nuclear reactors, high voltage line work, and medicine. Most of these disciplines have embraced simulation as an essential part of training. Simulation is part of many surgical training programs, but it has other uses.
When was the last time you practiced a trauma resuscitation, an ultrasound fine-needle biopsy, laparoscopic maneuvers, or an unusual technique that you seldom perform, but when needed, must be pulled off very well? Most of us taking this simulation course agreed that time, money, and ego may get in the way of maintaining those skills for those rare instances when they are needed. Surgeons might want to consider simulation to keep some of our rarely used skills from getting rusty.
If you’re going to make a costly error, I would very much like you to do it on a piece of plastic, not on a patient. There are no consequences for messing up a procedure on a manikin and this kind of practice might teach you something critical. Practicing reduces stress and improves the performance of those placed on the spot by real-life events. Do you think Captain “Sully” Sullenberger could have landed that airliner in the Hudson River safely if he hadn’t practiced with countless mind-numbingly complex simulations? Sure, luck plays a part, and innate ability plays a part. But skill, knowledge, and practice are your best bet when all the eyes in the room swivel to you in a moment of crisis.
You may think that simulators have to cost $100,000 and be completely realistic to do the job. That’s not true. A banana, orange, or stick of butter can be fabulous sims for a med student. Felt and cardboard can make a realistic cricothyroidotomy model.
Surgeons all over the country are using simulation training to learn how to be better without getting real blood on their shoes. If you haven’t participated in a training simulation recently, I double-dog dare you to try it and tell me you found it without merit. The ACS Surgical Simulation Summit is being held in March 2019 in Chicago. You might want to check that out.
Dr. Hughes is clinical professor in the department of surgery and director of medical education at the University of Kansas School of Medicine, Salina, and Coeditor of ACS Surgery News.
New Updates to Afib Guidelines from CHEST
The American College of Chest Physicians® announced updates to the evidence-based guidelines on antithrombotic therapy for atrial fibrillation. The guideline panel submitted the manuscript, Antithrombotic Therapy for Atrial Fibrillation: CHEST Guideline and Expert Panel Report, for publication in the journal CHEST®.
Key recommendations and shifts from previous guidelines include:
• For patients with atrial fibrillation without valvular heart disease, including those with paroxysmal atrial fibrillation who are at low risk of stroke (eg, CHA2DS2VASc score of 0 in males or 1 in females), we suggest no antithrombotic therapy.
• For patients with a single non-sex CHA2DS2VASc stroke risk factor, we suggest oral anticoagulation rather than no therapy, aspirin or combination therapy with aspirin and clopidogrel.
• For those at high risk of stroke, we recommend oral anticoagulation rather than no therapy, aspirin or combination therapy with aspirin and clopidogrel.
• Where we recommend or suggest in favor of oral anticoagulation, we suggest using a novel oral anticoagulant (NOAC) rather than adjusted-dose vitamin K antagonist therapy. With the latter, it is important to aim for good quality anticoagulation control with a time in therapeutic range (TTR) >70%.
• Attention to modifiable bleeding risk factors should be made at each patient contact, and HAS-BLED score should be used to assess the risk of bleeding where high-risk patients (>=3) can be identified for earlier review and follow-up visits.
The complete guideline article is free to view in the Online First section of the journal CHEST.
The American College of Chest Physicians® announced updates to the evidence-based guidelines on antithrombotic therapy for atrial fibrillation. The guideline panel submitted the manuscript, Antithrombotic Therapy for Atrial Fibrillation: CHEST Guideline and Expert Panel Report, for publication in the journal CHEST®.
Key recommendations and shifts from previous guidelines include:
• For patients with atrial fibrillation without valvular heart disease, including those with paroxysmal atrial fibrillation who are at low risk of stroke (eg, CHA2DS2VASc score of 0 in males or 1 in females), we suggest no antithrombotic therapy.
• For patients with a single non-sex CHA2DS2VASc stroke risk factor, we suggest oral anticoagulation rather than no therapy, aspirin or combination therapy with aspirin and clopidogrel.
• For those at high risk of stroke, we recommend oral anticoagulation rather than no therapy, aspirin or combination therapy with aspirin and clopidogrel.
• Where we recommend or suggest in favor of oral anticoagulation, we suggest using a novel oral anticoagulant (NOAC) rather than adjusted-dose vitamin K antagonist therapy. With the latter, it is important to aim for good quality anticoagulation control with a time in therapeutic range (TTR) >70%.
• Attention to modifiable bleeding risk factors should be made at each patient contact, and HAS-BLED score should be used to assess the risk of bleeding where high-risk patients (>=3) can be identified for earlier review and follow-up visits.
The complete guideline article is free to view in the Online First section of the journal CHEST.
The American College of Chest Physicians® announced updates to the evidence-based guidelines on antithrombotic therapy for atrial fibrillation. The guideline panel submitted the manuscript, Antithrombotic Therapy for Atrial Fibrillation: CHEST Guideline and Expert Panel Report, for publication in the journal CHEST®.
Key recommendations and shifts from previous guidelines include:
• For patients with atrial fibrillation without valvular heart disease, including those with paroxysmal atrial fibrillation who are at low risk of stroke (eg, CHA2DS2VASc score of 0 in males or 1 in females), we suggest no antithrombotic therapy.
• For patients with a single non-sex CHA2DS2VASc stroke risk factor, we suggest oral anticoagulation rather than no therapy, aspirin or combination therapy with aspirin and clopidogrel.
• For those at high risk of stroke, we recommend oral anticoagulation rather than no therapy, aspirin or combination therapy with aspirin and clopidogrel.
• Where we recommend or suggest in favor of oral anticoagulation, we suggest using a novel oral anticoagulant (NOAC) rather than adjusted-dose vitamin K antagonist therapy. With the latter, it is important to aim for good quality anticoagulation control with a time in therapeutic range (TTR) >70%.
• Attention to modifiable bleeding risk factors should be made at each patient contact, and HAS-BLED score should be used to assess the risk of bleeding where high-risk patients (>=3) can be identified for earlier review and follow-up visits.
The complete guideline article is free to view in the Online First section of the journal CHEST.
ECMO for ARDS in the modern era
Extracorporeal membrane oxygenation (ECMO) has become increasingly accepted as a rescue therapy for severe respiratory failure from a variety of conditions, though most commonly, the acute respiratory distress syndrome (ARDS) (Thiagarajan R, et al. ASAIO. 2017;63[1]:60). ECMO can provide respiratory or cardiorespiratory support for failing lungs, heart, or both. The most common ECMO configuration used in ARDS is venovenous ECMO, in which blood is withdrawn from a catheter placed in a central vein, pumped through a gas exchange device known as an oxygenator, and returned to the venous system via another catheter. The blood flowing through the oxygenator is separated from a continuous supply of oxygen-rich sweep gas by a semipermeable membrane, across which diffusion-mediated gas exchange occurs, so that the blood exiting it is rich in oxygen and low in carbon dioxide. As venovenous ECMO functions in series with the native circulation, the well-oxygenated blood exiting the ECMO circuit mixes with poorly oxygenated blood flowing through the lungs. Therefore, oxygenation is dependent on native cardiac output to achieve systemic oxygen delivery (Figure 1).
ECMO been used successfully in adults with ARDS since the early 1970s (Hill JD, et al. N Engl J Med. 1972;286[12]:629-34) but, until recently, was limited to small numbers of patients at select global centers and associated with a high-risk profile. In the last decade, however, driven by improvements in ECMO circuit components making the device safer and easier to use, encouraging worldwide experience during the 2009 influenza A (H1N1) pandemic (Davies A, et al. JAMA. 2009;302[17]1888-95), and publication of the Efficacy and Economic Assessment of Conventional Ventilatory Support versus Extracorporeal Membrane Oxygenation for Severe Adult Respiratory Failure (CESAR) trial (Peek GJ, et al. Lancet. 2009;374[9698]:1351-63), ECMO use has markedly increased.
Despite its rapid growth, however, rigorous evidence supporting the use of ECMO has been lacking. The CESAR trial, while impressive in execution, had methodological issues that limited the strength of its conclusions. CESAR was a pragmatic trial that randomized 180 adults with severe respiratory failure from multiple etiologies to conventional management or transfer to an experienced, ECMO-capable center. CESAR met its primary outcome of improved survival without disability in the ECMO-referred group (63% vs 47%, relative risk [RR] 0.69; 95% confidence interval [CI] 0.05 to 0.97, P=.03), but not all patients in that group ultimately received ECMO. In addition, the use of lung protective ventilation was significantly higher in the ECMO-referred group, making it difficult to separate its benefit from that of ECMO. A conservative interpretation is that CESAR showed the clinical benefit of treatment at an ECMO-capable center, experienced in the management of patients with severe respiratory failure.
Not until the release of the Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Distress Syndrome (EOLIA) trial earlier this year (Combes A, et al. N Engl J Med. 2018;378[21]:1965-75), did a modern, randomized controlled trial evaluating the use of ECMO itself exist. The EOLIA trial addressed the limitations of CESAR and randomized adult patients with early, severe ARDS to conventional, standard of care management that included a protocolized lung protective strategy in the control group vs immediate initiation of ECMO combined with an ultra-lung protective strategy (targeting end-inspiratory plateau pressure ≤24 cmH2O) in the intervention group. The primary outcome was all-cause mortality at 60 days. Of note, patients enrolled in EOLIA met entry criteria despite greater than 90% of patients receiving neuromuscular blockade and around 60% treated with prone positioning at the time of randomization (importantly, 90% of control group patients ultimately underwent prone positioning).
EOLIA was powered to detect a 20% decrease in mortality in the ECMO group. Based on trial design and the results of the fourth interim analysis, the trial was stopped for futility to reach that endpoint after enrollment of 249 of a maximum 331 patients. Although a 20% mortality reduction was not achieved, 60-day mortality was notably lower in the ECMO-treated group (35% vs 46%, RR 0.76, 95% CI 0.55 to 1.04, P=.09). The key secondary outcome of risk of treatment failure (defined as death in the ECMO group and death or crossover to ECMO in the control group) favored the ECMO group with a RR for mortality of 0.62 (95% CI, 0.47 to 0.82; P<.001), as did other secondary endpoints such as days free of renal and other organ failure.
A major limitation of the trial was that 35 (28%) of control group patients ultimately crossed over to ECMO, which diluted the effect of ECMO observed in the intention-to-treat analysis. Crossover occurred at clinician discretion an average of 6.5 days after randomization and after stringent criteria for crossover was met. These patients were incredibly ill, with a median oxygen saturation of 77%, rapidly worsening inotropic scores, and lactic acidosis; nine individuals had already suffered cardiac arrest, and six had received ECMO as part of extracorporeal cardiopulmonary resuscitation (ECPR), the initiation of venoarterial ECMO during cardiac arrest in attempt to restore spontaneous circulation. Mortality was considerably worse in the crossover group than in conventionally managed cohort overall, and, notably, 33% of patients crossed over to ECMO still survived.
In order to estimate the effect of ECMO on survival times if crossover had not occurred, the authors performed a post-hoc, rank-preserving structural failure time analysis. Though this relies on some assessment regarding the effect of the treatment itself, it showed a hazard ratio for mortality in the ECMO group of 0.51 (95% CI 0.24 to 1.02, P=.055). Although the EOLIA trial was not positive by traditional interpretation, all three major analyses and all secondary endpoints suggest some degree of benefit in patients with severe ARDS managed with ECMO.
Importantly, ECMO was well tolerated (at least when performed at expert centers, as done in this trial). There were significantly more bleeding events and cases of severe thrombocytopenia in the ECMO-treated group, but massive hemorrhage, ischemic and hemorrhagic stroke, arrhythmias, and other complications were similar.
Where do we go from here? Based on the totality of information, it is reasonable to consider ECMO for cases of severe ARDS not responsive to conventional measures, such as a lung protective ventilator strategy, neuromuscular blockade, and prone positioning. Initiation of ECMO may be reasonable prior to implementation of standard of care therapies, in order to permit safe transfer to an experienced center from a center not able to provide them.
Two take-away points: First, it is important to recognize that much of the clinical benefit derived from ECMO may be beyond its ability to normalize gas exchange and be due, at least in part, to the fact that ECMO allows the enhancement of proven lung protective ventilatory strategies. Initiation of ECMO and the “lung rest” it permits reduce the mechanical power applied to the injured alveoli and may attenuate ventilator-induced lung injury, cytokine release, and multiorgan failure that portend poor clinical outcomes in ARDS. Second, ECMO in EOLIA was conducted at expert centers with relatively low rates of complications.
It is too early to know how the critical care community will view ECMO for ARDS in light of EOLIA as well as a growing body of global ECMO experience, or how its wider application may impact the distribution and organization of ECMO centers. Regardless, of paramount importance in using ECMO as a treatment modality is optimizing patient management both prior to and after its initiation.
Dr. Agerstrand is Assistant Professor of Medicine, Director of the Medical ECMO Program, Columbia University College of Physicians and Surgeons, New York-Presbyterian Hospital.
Extracorporeal membrane oxygenation (ECMO) has become increasingly accepted as a rescue therapy for severe respiratory failure from a variety of conditions, though most commonly, the acute respiratory distress syndrome (ARDS) (Thiagarajan R, et al. ASAIO. 2017;63[1]:60). ECMO can provide respiratory or cardiorespiratory support for failing lungs, heart, or both. The most common ECMO configuration used in ARDS is venovenous ECMO, in which blood is withdrawn from a catheter placed in a central vein, pumped through a gas exchange device known as an oxygenator, and returned to the venous system via another catheter. The blood flowing through the oxygenator is separated from a continuous supply of oxygen-rich sweep gas by a semipermeable membrane, across which diffusion-mediated gas exchange occurs, so that the blood exiting it is rich in oxygen and low in carbon dioxide. As venovenous ECMO functions in series with the native circulation, the well-oxygenated blood exiting the ECMO circuit mixes with poorly oxygenated blood flowing through the lungs. Therefore, oxygenation is dependent on native cardiac output to achieve systemic oxygen delivery (Figure 1).
ECMO been used successfully in adults with ARDS since the early 1970s (Hill JD, et al. N Engl J Med. 1972;286[12]:629-34) but, until recently, was limited to small numbers of patients at select global centers and associated with a high-risk profile. In the last decade, however, driven by improvements in ECMO circuit components making the device safer and easier to use, encouraging worldwide experience during the 2009 influenza A (H1N1) pandemic (Davies A, et al. JAMA. 2009;302[17]1888-95), and publication of the Efficacy and Economic Assessment of Conventional Ventilatory Support versus Extracorporeal Membrane Oxygenation for Severe Adult Respiratory Failure (CESAR) trial (Peek GJ, et al. Lancet. 2009;374[9698]:1351-63), ECMO use has markedly increased.
Despite its rapid growth, however, rigorous evidence supporting the use of ECMO has been lacking. The CESAR trial, while impressive in execution, had methodological issues that limited the strength of its conclusions. CESAR was a pragmatic trial that randomized 180 adults with severe respiratory failure from multiple etiologies to conventional management or transfer to an experienced, ECMO-capable center. CESAR met its primary outcome of improved survival without disability in the ECMO-referred group (63% vs 47%, relative risk [RR] 0.69; 95% confidence interval [CI] 0.05 to 0.97, P=.03), but not all patients in that group ultimately received ECMO. In addition, the use of lung protective ventilation was significantly higher in the ECMO-referred group, making it difficult to separate its benefit from that of ECMO. A conservative interpretation is that CESAR showed the clinical benefit of treatment at an ECMO-capable center, experienced in the management of patients with severe respiratory failure.
Not until the release of the Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Distress Syndrome (EOLIA) trial earlier this year (Combes A, et al. N Engl J Med. 2018;378[21]:1965-75), did a modern, randomized controlled trial evaluating the use of ECMO itself exist. The EOLIA trial addressed the limitations of CESAR and randomized adult patients with early, severe ARDS to conventional, standard of care management that included a protocolized lung protective strategy in the control group vs immediate initiation of ECMO combined with an ultra-lung protective strategy (targeting end-inspiratory plateau pressure ≤24 cmH2O) in the intervention group. The primary outcome was all-cause mortality at 60 days. Of note, patients enrolled in EOLIA met entry criteria despite greater than 90% of patients receiving neuromuscular blockade and around 60% treated with prone positioning at the time of randomization (importantly, 90% of control group patients ultimately underwent prone positioning).
EOLIA was powered to detect a 20% decrease in mortality in the ECMO group. Based on trial design and the results of the fourth interim analysis, the trial was stopped for futility to reach that endpoint after enrollment of 249 of a maximum 331 patients. Although a 20% mortality reduction was not achieved, 60-day mortality was notably lower in the ECMO-treated group (35% vs 46%, RR 0.76, 95% CI 0.55 to 1.04, P=.09). The key secondary outcome of risk of treatment failure (defined as death in the ECMO group and death or crossover to ECMO in the control group) favored the ECMO group with a RR for mortality of 0.62 (95% CI, 0.47 to 0.82; P<.001), as did other secondary endpoints such as days free of renal and other organ failure.
A major limitation of the trial was that 35 (28%) of control group patients ultimately crossed over to ECMO, which diluted the effect of ECMO observed in the intention-to-treat analysis. Crossover occurred at clinician discretion an average of 6.5 days after randomization and after stringent criteria for crossover was met. These patients were incredibly ill, with a median oxygen saturation of 77%, rapidly worsening inotropic scores, and lactic acidosis; nine individuals had already suffered cardiac arrest, and six had received ECMO as part of extracorporeal cardiopulmonary resuscitation (ECPR), the initiation of venoarterial ECMO during cardiac arrest in attempt to restore spontaneous circulation. Mortality was considerably worse in the crossover group than in conventionally managed cohort overall, and, notably, 33% of patients crossed over to ECMO still survived.
In order to estimate the effect of ECMO on survival times if crossover had not occurred, the authors performed a post-hoc, rank-preserving structural failure time analysis. Though this relies on some assessment regarding the effect of the treatment itself, it showed a hazard ratio for mortality in the ECMO group of 0.51 (95% CI 0.24 to 1.02, P=.055). Although the EOLIA trial was not positive by traditional interpretation, all three major analyses and all secondary endpoints suggest some degree of benefit in patients with severe ARDS managed with ECMO.
Importantly, ECMO was well tolerated (at least when performed at expert centers, as done in this trial). There were significantly more bleeding events and cases of severe thrombocytopenia in the ECMO-treated group, but massive hemorrhage, ischemic and hemorrhagic stroke, arrhythmias, and other complications were similar.
Where do we go from here? Based on the totality of information, it is reasonable to consider ECMO for cases of severe ARDS not responsive to conventional measures, such as a lung protective ventilator strategy, neuromuscular blockade, and prone positioning. Initiation of ECMO may be reasonable prior to implementation of standard of care therapies, in order to permit safe transfer to an experienced center from a center not able to provide them.
Two take-away points: First, it is important to recognize that much of the clinical benefit derived from ECMO may be beyond its ability to normalize gas exchange and be due, at least in part, to the fact that ECMO allows the enhancement of proven lung protective ventilatory strategies. Initiation of ECMO and the “lung rest” it permits reduce the mechanical power applied to the injured alveoli and may attenuate ventilator-induced lung injury, cytokine release, and multiorgan failure that portend poor clinical outcomes in ARDS. Second, ECMO in EOLIA was conducted at expert centers with relatively low rates of complications.
It is too early to know how the critical care community will view ECMO for ARDS in light of EOLIA as well as a growing body of global ECMO experience, or how its wider application may impact the distribution and organization of ECMO centers. Regardless, of paramount importance in using ECMO as a treatment modality is optimizing patient management both prior to and after its initiation.
Dr. Agerstrand is Assistant Professor of Medicine, Director of the Medical ECMO Program, Columbia University College of Physicians and Surgeons, New York-Presbyterian Hospital.
Extracorporeal membrane oxygenation (ECMO) has become increasingly accepted as a rescue therapy for severe respiratory failure from a variety of conditions, though most commonly, the acute respiratory distress syndrome (ARDS) (Thiagarajan R, et al. ASAIO. 2017;63[1]:60). ECMO can provide respiratory or cardiorespiratory support for failing lungs, heart, or both. The most common ECMO configuration used in ARDS is venovenous ECMO, in which blood is withdrawn from a catheter placed in a central vein, pumped through a gas exchange device known as an oxygenator, and returned to the venous system via another catheter. The blood flowing through the oxygenator is separated from a continuous supply of oxygen-rich sweep gas by a semipermeable membrane, across which diffusion-mediated gas exchange occurs, so that the blood exiting it is rich in oxygen and low in carbon dioxide. As venovenous ECMO functions in series with the native circulation, the well-oxygenated blood exiting the ECMO circuit mixes with poorly oxygenated blood flowing through the lungs. Therefore, oxygenation is dependent on native cardiac output to achieve systemic oxygen delivery (Figure 1).
ECMO been used successfully in adults with ARDS since the early 1970s (Hill JD, et al. N Engl J Med. 1972;286[12]:629-34) but, until recently, was limited to small numbers of patients at select global centers and associated with a high-risk profile. In the last decade, however, driven by improvements in ECMO circuit components making the device safer and easier to use, encouraging worldwide experience during the 2009 influenza A (H1N1) pandemic (Davies A, et al. JAMA. 2009;302[17]1888-95), and publication of the Efficacy and Economic Assessment of Conventional Ventilatory Support versus Extracorporeal Membrane Oxygenation for Severe Adult Respiratory Failure (CESAR) trial (Peek GJ, et al. Lancet. 2009;374[9698]:1351-63), ECMO use has markedly increased.
Despite its rapid growth, however, rigorous evidence supporting the use of ECMO has been lacking. The CESAR trial, while impressive in execution, had methodological issues that limited the strength of its conclusions. CESAR was a pragmatic trial that randomized 180 adults with severe respiratory failure from multiple etiologies to conventional management or transfer to an experienced, ECMO-capable center. CESAR met its primary outcome of improved survival without disability in the ECMO-referred group (63% vs 47%, relative risk [RR] 0.69; 95% confidence interval [CI] 0.05 to 0.97, P=.03), but not all patients in that group ultimately received ECMO. In addition, the use of lung protective ventilation was significantly higher in the ECMO-referred group, making it difficult to separate its benefit from that of ECMO. A conservative interpretation is that CESAR showed the clinical benefit of treatment at an ECMO-capable center, experienced in the management of patients with severe respiratory failure.
Not until the release of the Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Distress Syndrome (EOLIA) trial earlier this year (Combes A, et al. N Engl J Med. 2018;378[21]:1965-75), did a modern, randomized controlled trial evaluating the use of ECMO itself exist. The EOLIA trial addressed the limitations of CESAR and randomized adult patients with early, severe ARDS to conventional, standard of care management that included a protocolized lung protective strategy in the control group vs immediate initiation of ECMO combined with an ultra-lung protective strategy (targeting end-inspiratory plateau pressure ≤24 cmH2O) in the intervention group. The primary outcome was all-cause mortality at 60 days. Of note, patients enrolled in EOLIA met entry criteria despite greater than 90% of patients receiving neuromuscular blockade and around 60% treated with prone positioning at the time of randomization (importantly, 90% of control group patients ultimately underwent prone positioning).
EOLIA was powered to detect a 20% decrease in mortality in the ECMO group. Based on trial design and the results of the fourth interim analysis, the trial was stopped for futility to reach that endpoint after enrollment of 249 of a maximum 331 patients. Although a 20% mortality reduction was not achieved, 60-day mortality was notably lower in the ECMO-treated group (35% vs 46%, RR 0.76, 95% CI 0.55 to 1.04, P=.09). The key secondary outcome of risk of treatment failure (defined as death in the ECMO group and death or crossover to ECMO in the control group) favored the ECMO group with a RR for mortality of 0.62 (95% CI, 0.47 to 0.82; P<.001), as did other secondary endpoints such as days free of renal and other organ failure.
A major limitation of the trial was that 35 (28%) of control group patients ultimately crossed over to ECMO, which diluted the effect of ECMO observed in the intention-to-treat analysis. Crossover occurred at clinician discretion an average of 6.5 days after randomization and after stringent criteria for crossover was met. These patients were incredibly ill, with a median oxygen saturation of 77%, rapidly worsening inotropic scores, and lactic acidosis; nine individuals had already suffered cardiac arrest, and six had received ECMO as part of extracorporeal cardiopulmonary resuscitation (ECPR), the initiation of venoarterial ECMO during cardiac arrest in attempt to restore spontaneous circulation. Mortality was considerably worse in the crossover group than in conventionally managed cohort overall, and, notably, 33% of patients crossed over to ECMO still survived.
In order to estimate the effect of ECMO on survival times if crossover had not occurred, the authors performed a post-hoc, rank-preserving structural failure time analysis. Though this relies on some assessment regarding the effect of the treatment itself, it showed a hazard ratio for mortality in the ECMO group of 0.51 (95% CI 0.24 to 1.02, P=.055). Although the EOLIA trial was not positive by traditional interpretation, all three major analyses and all secondary endpoints suggest some degree of benefit in patients with severe ARDS managed with ECMO.
Importantly, ECMO was well tolerated (at least when performed at expert centers, as done in this trial). There were significantly more bleeding events and cases of severe thrombocytopenia in the ECMO-treated group, but massive hemorrhage, ischemic and hemorrhagic stroke, arrhythmias, and other complications were similar.
Where do we go from here? Based on the totality of information, it is reasonable to consider ECMO for cases of severe ARDS not responsive to conventional measures, such as a lung protective ventilator strategy, neuromuscular blockade, and prone positioning. Initiation of ECMO may be reasonable prior to implementation of standard of care therapies, in order to permit safe transfer to an experienced center from a center not able to provide them.
Two take-away points: First, it is important to recognize that much of the clinical benefit derived from ECMO may be beyond its ability to normalize gas exchange and be due, at least in part, to the fact that ECMO allows the enhancement of proven lung protective ventilatory strategies. Initiation of ECMO and the “lung rest” it permits reduce the mechanical power applied to the injured alveoli and may attenuate ventilator-induced lung injury, cytokine release, and multiorgan failure that portend poor clinical outcomes in ARDS. Second, ECMO in EOLIA was conducted at expert centers with relatively low rates of complications.
It is too early to know how the critical care community will view ECMO for ARDS in light of EOLIA as well as a growing body of global ECMO experience, or how its wider application may impact the distribution and organization of ECMO centers. Regardless, of paramount importance in using ECMO as a treatment modality is optimizing patient management both prior to and after its initiation.
Dr. Agerstrand is Assistant Professor of Medicine, Director of the Medical ECMO Program, Columbia University College of Physicians and Surgeons, New York-Presbyterian Hospital.