User login
Appropriateness of performing in-office uterine aspiration
In their article, "Uterine aspiration: From OR to office" (February 2019), Lauren Thaxton, MD, MBA, and Bri Tristan, MD, made the case for why, in appropriate clinical situations, office-based uterine aspiration, compared with uterine aspiration in the OR, should be the standard surgical management of early pregnancy failure. Their reasons included an equivalent safety profile, reduced costs, and patient-centered characteristics.
OBG Management posed this query to readers in a website poll: "Should the standard location for uterine apiration be in the office?" See how readers responded, below.
Poll results
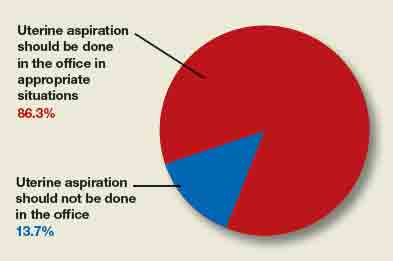
A total of 73 readers cast their vote:
- 86.3% (63 readers) said yes, in appropriate clinical situations
- 13.7% (10 readers) said no
Reader comments
"Yes, in appropriate clinical situations."
-Yardlie Toussaint-Foster, DO, Downingtown, Pennsylvania
"I have been doing it this way (in the office) for years, up to 11 to 12 weeks without complication."
-John Lane, MD, Raleigh, North Carolina
In their article, "Uterine aspiration: From OR to office" (February 2019), Lauren Thaxton, MD, MBA, and Bri Tristan, MD, made the case for why, in appropriate clinical situations, office-based uterine aspiration, compared with uterine aspiration in the OR, should be the standard surgical management of early pregnancy failure. Their reasons included an equivalent safety profile, reduced costs, and patient-centered characteristics.
OBG Management posed this query to readers in a website poll: "Should the standard location for uterine apiration be in the office?" See how readers responded, below.
Poll results
A total of 73 readers cast their vote:
- 86.3% (63 readers) said yes, in appropriate clinical situations
- 13.7% (10 readers) said no
Reader comments
"Yes, in appropriate clinical situations."
-Yardlie Toussaint-Foster, DO, Downingtown, Pennsylvania
"I have been doing it this way (in the office) for years, up to 11 to 12 weeks without complication."
-John Lane, MD, Raleigh, North Carolina

In their article, "Uterine aspiration: From OR to office" (February 2019), Lauren Thaxton, MD, MBA, and Bri Tristan, MD, made the case for why, in appropriate clinical situations, office-based uterine aspiration, compared with uterine aspiration in the OR, should be the standard surgical management of early pregnancy failure. Their reasons included an equivalent safety profile, reduced costs, and patient-centered characteristics.
OBG Management posed this query to readers in a website poll: "Should the standard location for uterine apiration be in the office?" See how readers responded, below.
Poll results
A total of 73 readers cast their vote:
- 86.3% (63 readers) said yes, in appropriate clinical situations
- 13.7% (10 readers) said no
Reader comments
"Yes, in appropriate clinical situations."
-Yardlie Toussaint-Foster, DO, Downingtown, Pennsylvania
"I have been doing it this way (in the office) for years, up to 11 to 12 weeks without complication."
-John Lane, MD, Raleigh, North Carolina

Split-dose oxycodone protocol reduces opioid use after cesarean
according to a study published in Obstetrics & Gynecology.
A retrospective study reviewed medical records of 1,050 women undergoing cesarean delivery, 508 of whom were treated after a change in protocol for postdelivery oxycodone orders. Instead of a 5-mg oral dose given for a verbal pain score of 4/10 or below and 10 mg for a pain score of 5-10/10, patients were given 2.5-mg or 5-mg dose respectively, with a nurse check after 1 hour to see if more of the same dosage was needed.
The split-dose approach was associated with a 56% reduction in median opioid consumption in the first 48 hours after cesarean delivery; 10 mg before the change in practice to 4.4 mg after it. There was also a 6.9-percentage-point decrease in the number of patients needing any postoperative opioids.
While the study did show a slight increase in average verbal pain scores in the first 58 hours after surgery – from a mean of 1.8 before the split-dose protocol was introduced to 2 after it was introduced – there was no increase in the use of nonsteroidal anti-inflammatory drugs, acetaminophen, or gabapentin, and no difference in peak verbal pain scores.
“Our goal with the introduction of this new order set was to use a patient-centered, response-feedback approach to postcesarean delivery analgesia in the form of split doses of oxycodone rather than the traditional standard dose model,” wrote Jalal A. Nanji, MD, of the department of anesthesiology and pain medicine at the University of Alberta, Edmonton, and coauthors. “Involving patients in the decision for how much postcesarean delivery analgesia they will receive has been found to reduce opioid use and improve maternal satisfaction.”
The number of patients reporting postoperative nausea or vomiting was halved in those treated with the split-dose regimen, with no difference in mean overall patient satisfaction score.
Dr. Nanji and associates wrote that women viewed avoiding nausea or vomiting after a cesarean as a high priority, and targeting the root cause – excessive opioid use – was preferable to treating nausea and vomiting with antiemetics.
They also noted that input from nursing staff was vital in developing the new split-order set, not only because it directly affected nursing work flow but also to optimize the process.
“With the opioid epidemic on the rise and the increase in efforts by physicians to decrease outpatient opioid prescriptions, this study is extremely relevant and timely,” commented Marissa Platner, MD, an assistant professor in maternal-fetal medicine at Emory University, Atlanta.
“Although this study is retrospective and, therefore, there are inherent biases and an inability to control all contributing factors, it clearly demonstrates that, overall, there seem to be improved outcomes with split-dose protocol of opioid administration during the postoperative period in terms of overall patient satisfaction, opioid consumption, and postoperative nausea and vomiting. The patient-centered nature and response-feedback design of this study also contributes to its strength and improves its generalizability. In order to encourage others to considering adapting protocol in other institutions, it should be evaluated via a randomized controlled trial," Dr. Platner said in an interview.*
"The premise and execution of this study were novel and interesting," commented Katrina Mark, MD, associate professor of obstetrics, gynecology & reproductive sciences at the University of Maryland School of Medicine. "The authors found that by decreasing the standard doses of oxycodone ordered after a cesarean section and asking women if they desired better pain control, rather than reacting only to a pain score, patients’ overall postoperative usage of opiates also decreased. In decreasing the amount of opiates used, the authors also observed a decrease some of the side effects associated with opiate use, which is promising.
"This study, among other recent studies, highlights the fact that postoperative prescribing standards are not evidence-based and may lead to overprescribing of opiates. Improving prescribing practices is a noble and important goal. In this study, a change in clinical practice among both nurses and prescribers is likely what caused the greatest change. The use of a protocol which prescribed oxycodone based on asking if a woman desired improved pain control, rather than prescribing only based on her pain score response, makes a lot of intuitive sense. Decreasing opioid consumption requires education of healthcare providers and patients, and protocols like this one will help to encourage that conversation," she noted in an interview.
"Before the findings of this study can be widely adopted, however, there are two major points that will need to be addressed," Dr. Mark emphasized. "The first is patient satisfaction. The peak pain scores were not different between the groups, but the mean pain scores were. The authors deemed this clinically insignificant, which it may be. However, without the patients’ perspective on this new protocol, it is difficult to tell if the opioid usage decreased because women actually needed less opiates or if it decreased because the system discouraged opioid use and made it more challenging for them to obtain the medicine they needed to achieve adequate pain control. The desire to decrease opioid prescribing is warranted, and likely completely appropriate, but there is certainly a role for opioids in pain management. We should not be so motivated to decrease use that we cause unnecessary suffering. The second point that will need to be addressed is the effect on nursing practice. There was no standardized evaluation of the impact that this protocol had on the nursing staff, and it is unclear if this protocol would require greater resources than may be readily available at all hospitals."**
The study was supported by the department of anesthesiology, perioperative, and pain medicine at Stanford (Calif.) University. One author declared travel funding from a university. No other conflicts of interest were declared. Dr. Platner and Dr. Mark also had no relevant financial disclosures.*
SOURCE: Nanji J et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003305.
*This article was updated on 7/15/2019.
**It was updated again on 7/17/2019.
according to a study published in Obstetrics & Gynecology.
A retrospective study reviewed medical records of 1,050 women undergoing cesarean delivery, 508 of whom were treated after a change in protocol for postdelivery oxycodone orders. Instead of a 5-mg oral dose given for a verbal pain score of 4/10 or below and 10 mg for a pain score of 5-10/10, patients were given 2.5-mg or 5-mg dose respectively, with a nurse check after 1 hour to see if more of the same dosage was needed.
The split-dose approach was associated with a 56% reduction in median opioid consumption in the first 48 hours after cesarean delivery; 10 mg before the change in practice to 4.4 mg after it. There was also a 6.9-percentage-point decrease in the number of patients needing any postoperative opioids.
While the study did show a slight increase in average verbal pain scores in the first 58 hours after surgery – from a mean of 1.8 before the split-dose protocol was introduced to 2 after it was introduced – there was no increase in the use of nonsteroidal anti-inflammatory drugs, acetaminophen, or gabapentin, and no difference in peak verbal pain scores.
“Our goal with the introduction of this new order set was to use a patient-centered, response-feedback approach to postcesarean delivery analgesia in the form of split doses of oxycodone rather than the traditional standard dose model,” wrote Jalal A. Nanji, MD, of the department of anesthesiology and pain medicine at the University of Alberta, Edmonton, and coauthors. “Involving patients in the decision for how much postcesarean delivery analgesia they will receive has been found to reduce opioid use and improve maternal satisfaction.”
The number of patients reporting postoperative nausea or vomiting was halved in those treated with the split-dose regimen, with no difference in mean overall patient satisfaction score.
Dr. Nanji and associates wrote that women viewed avoiding nausea or vomiting after a cesarean as a high priority, and targeting the root cause – excessive opioid use – was preferable to treating nausea and vomiting with antiemetics.
They also noted that input from nursing staff was vital in developing the new split-order set, not only because it directly affected nursing work flow but also to optimize the process.
“With the opioid epidemic on the rise and the increase in efforts by physicians to decrease outpatient opioid prescriptions, this study is extremely relevant and timely,” commented Marissa Platner, MD, an assistant professor in maternal-fetal medicine at Emory University, Atlanta.
“Although this study is retrospective and, therefore, there are inherent biases and an inability to control all contributing factors, it clearly demonstrates that, overall, there seem to be improved outcomes with split-dose protocol of opioid administration during the postoperative period in terms of overall patient satisfaction, opioid consumption, and postoperative nausea and vomiting. The patient-centered nature and response-feedback design of this study also contributes to its strength and improves its generalizability. In order to encourage others to considering adapting protocol in other institutions, it should be evaluated via a randomized controlled trial," Dr. Platner said in an interview.*
"The premise and execution of this study were novel and interesting," commented Katrina Mark, MD, associate professor of obstetrics, gynecology & reproductive sciences at the University of Maryland School of Medicine. "The authors found that by decreasing the standard doses of oxycodone ordered after a cesarean section and asking women if they desired better pain control, rather than reacting only to a pain score, patients’ overall postoperative usage of opiates also decreased. In decreasing the amount of opiates used, the authors also observed a decrease some of the side effects associated with opiate use, which is promising.
"This study, among other recent studies, highlights the fact that postoperative prescribing standards are not evidence-based and may lead to overprescribing of opiates. Improving prescribing practices is a noble and important goal. In this study, a change in clinical practice among both nurses and prescribers is likely what caused the greatest change. The use of a protocol which prescribed oxycodone based on asking if a woman desired improved pain control, rather than prescribing only based on her pain score response, makes a lot of intuitive sense. Decreasing opioid consumption requires education of healthcare providers and patients, and protocols like this one will help to encourage that conversation," she noted in an interview.
"Before the findings of this study can be widely adopted, however, there are two major points that will need to be addressed," Dr. Mark emphasized. "The first is patient satisfaction. The peak pain scores were not different between the groups, but the mean pain scores were. The authors deemed this clinically insignificant, which it may be. However, without the patients’ perspective on this new protocol, it is difficult to tell if the opioid usage decreased because women actually needed less opiates or if it decreased because the system discouraged opioid use and made it more challenging for them to obtain the medicine they needed to achieve adequate pain control. The desire to decrease opioid prescribing is warranted, and likely completely appropriate, but there is certainly a role for opioids in pain management. We should not be so motivated to decrease use that we cause unnecessary suffering. The second point that will need to be addressed is the effect on nursing practice. There was no standardized evaluation of the impact that this protocol had on the nursing staff, and it is unclear if this protocol would require greater resources than may be readily available at all hospitals."**
The study was supported by the department of anesthesiology, perioperative, and pain medicine at Stanford (Calif.) University. One author declared travel funding from a university. No other conflicts of interest were declared. Dr. Platner and Dr. Mark also had no relevant financial disclosures.*
SOURCE: Nanji J et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003305.
*This article was updated on 7/15/2019.
**It was updated again on 7/17/2019.
according to a study published in Obstetrics & Gynecology.
A retrospective study reviewed medical records of 1,050 women undergoing cesarean delivery, 508 of whom were treated after a change in protocol for postdelivery oxycodone orders. Instead of a 5-mg oral dose given for a verbal pain score of 4/10 or below and 10 mg for a pain score of 5-10/10, patients were given 2.5-mg or 5-mg dose respectively, with a nurse check after 1 hour to see if more of the same dosage was needed.
The split-dose approach was associated with a 56% reduction in median opioid consumption in the first 48 hours after cesarean delivery; 10 mg before the change in practice to 4.4 mg after it. There was also a 6.9-percentage-point decrease in the number of patients needing any postoperative opioids.
While the study did show a slight increase in average verbal pain scores in the first 58 hours after surgery – from a mean of 1.8 before the split-dose protocol was introduced to 2 after it was introduced – there was no increase in the use of nonsteroidal anti-inflammatory drugs, acetaminophen, or gabapentin, and no difference in peak verbal pain scores.
“Our goal with the introduction of this new order set was to use a patient-centered, response-feedback approach to postcesarean delivery analgesia in the form of split doses of oxycodone rather than the traditional standard dose model,” wrote Jalal A. Nanji, MD, of the department of anesthesiology and pain medicine at the University of Alberta, Edmonton, and coauthors. “Involving patients in the decision for how much postcesarean delivery analgesia they will receive has been found to reduce opioid use and improve maternal satisfaction.”
The number of patients reporting postoperative nausea or vomiting was halved in those treated with the split-dose regimen, with no difference in mean overall patient satisfaction score.
Dr. Nanji and associates wrote that women viewed avoiding nausea or vomiting after a cesarean as a high priority, and targeting the root cause – excessive opioid use – was preferable to treating nausea and vomiting with antiemetics.
They also noted that input from nursing staff was vital in developing the new split-order set, not only because it directly affected nursing work flow but also to optimize the process.
“With the opioid epidemic on the rise and the increase in efforts by physicians to decrease outpatient opioid prescriptions, this study is extremely relevant and timely,” commented Marissa Platner, MD, an assistant professor in maternal-fetal medicine at Emory University, Atlanta.
“Although this study is retrospective and, therefore, there are inherent biases and an inability to control all contributing factors, it clearly demonstrates that, overall, there seem to be improved outcomes with split-dose protocol of opioid administration during the postoperative period in terms of overall patient satisfaction, opioid consumption, and postoperative nausea and vomiting. The patient-centered nature and response-feedback design of this study also contributes to its strength and improves its generalizability. In order to encourage others to considering adapting protocol in other institutions, it should be evaluated via a randomized controlled trial," Dr. Platner said in an interview.*
"The premise and execution of this study were novel and interesting," commented Katrina Mark, MD, associate professor of obstetrics, gynecology & reproductive sciences at the University of Maryland School of Medicine. "The authors found that by decreasing the standard doses of oxycodone ordered after a cesarean section and asking women if they desired better pain control, rather than reacting only to a pain score, patients’ overall postoperative usage of opiates also decreased. In decreasing the amount of opiates used, the authors also observed a decrease some of the side effects associated with opiate use, which is promising.
"This study, among other recent studies, highlights the fact that postoperative prescribing standards are not evidence-based and may lead to overprescribing of opiates. Improving prescribing practices is a noble and important goal. In this study, a change in clinical practice among both nurses and prescribers is likely what caused the greatest change. The use of a protocol which prescribed oxycodone based on asking if a woman desired improved pain control, rather than prescribing only based on her pain score response, makes a lot of intuitive sense. Decreasing opioid consumption requires education of healthcare providers and patients, and protocols like this one will help to encourage that conversation," she noted in an interview.
"Before the findings of this study can be widely adopted, however, there are two major points that will need to be addressed," Dr. Mark emphasized. "The first is patient satisfaction. The peak pain scores were not different between the groups, but the mean pain scores were. The authors deemed this clinically insignificant, which it may be. However, without the patients’ perspective on this new protocol, it is difficult to tell if the opioid usage decreased because women actually needed less opiates or if it decreased because the system discouraged opioid use and made it more challenging for them to obtain the medicine they needed to achieve adequate pain control. The desire to decrease opioid prescribing is warranted, and likely completely appropriate, but there is certainly a role for opioids in pain management. We should not be so motivated to decrease use that we cause unnecessary suffering. The second point that will need to be addressed is the effect on nursing practice. There was no standardized evaluation of the impact that this protocol had on the nursing staff, and it is unclear if this protocol would require greater resources than may be readily available at all hospitals."**
The study was supported by the department of anesthesiology, perioperative, and pain medicine at Stanford (Calif.) University. One author declared travel funding from a university. No other conflicts of interest were declared. Dr. Platner and Dr. Mark also had no relevant financial disclosures.*
SOURCE: Nanji J et al. Obstet Gynecol. 2019. doi: 10.1097/AOG.0000000000003305.
*This article was updated on 7/15/2019.
**It was updated again on 7/17/2019.
FROM OBSTETRICS & GYNECOLOGY
Updated Patient Fliers Available Now
The SVS Foundation’s patient fliers project is completed, and fliers are available free to members on the SVS website. Nine vascular topics are addressed in updated fliers, including Carotid Artery Disease, Diabetes, Peripheral Arterial Disease, and more. These were redesigned to be useful in a patient waiting room, or to hand to a patient during an office visit. They are available in both English and Spanish, and can be found on the SVS website here.
The SVS Foundation’s patient fliers project is completed, and fliers are available free to members on the SVS website. Nine vascular topics are addressed in updated fliers, including Carotid Artery Disease, Diabetes, Peripheral Arterial Disease, and more. These were redesigned to be useful in a patient waiting room, or to hand to a patient during an office visit. They are available in both English and Spanish, and can be found on the SVS website here.
The SVS Foundation’s patient fliers project is completed, and fliers are available free to members on the SVS website. Nine vascular topics are addressed in updated fliers, including Carotid Artery Disease, Diabetes, Peripheral Arterial Disease, and more. These were redesigned to be useful in a patient waiting room, or to hand to a patient during an office visit. They are available in both English and Spanish, and can be found on the SVS website here.
Apply for the Research Career Development Travel Award
The SVS Foundation developed the Research Career Development Travel Awards program to develop strong leaders in vascular surgery research. Recipients of the award will be assigned SVS research mentors who will provide guidance and discuss academic career advancement. They’ll also receive financial support to be used for travel, hotel accommodations and registration expenses for a research course. Applicants must be an SVS Candidate or Active Member who’s completed postgraduate clinical training in vascular surgery and has been in practice no more than seven years. Apply before August 15 to be considered.
The SVS Foundation developed the Research Career Development Travel Awards program to develop strong leaders in vascular surgery research. Recipients of the award will be assigned SVS research mentors who will provide guidance and discuss academic career advancement. They’ll also receive financial support to be used for travel, hotel accommodations and registration expenses for a research course. Applicants must be an SVS Candidate or Active Member who’s completed postgraduate clinical training in vascular surgery and has been in practice no more than seven years. Apply before August 15 to be considered.
The SVS Foundation developed the Research Career Development Travel Awards program to develop strong leaders in vascular surgery research. Recipients of the award will be assigned SVS research mentors who will provide guidance and discuss academic career advancement. They’ll also receive financial support to be used for travel, hotel accommodations and registration expenses for a research course. Applicants must be an SVS Candidate or Active Member who’s completed postgraduate clinical training in vascular surgery and has been in practice no more than seven years. Apply before August 15 to be considered.
VAM Photos Now Available
If you attended the 2019 Vascular Annual Meeting in National Harbor, Md., be sure to check out the many photos now available on the SVS Flickr account. Relive the education sessions, the exhibit hall, the alumni receptions and the marvelous “Vascular Spectacular” gala. View, download and share these as much as you’d like. If you had your headshot taken at the SVS Booth, you can find that photo here.
If you attended the 2019 Vascular Annual Meeting in National Harbor, Md., be sure to check out the many photos now available on the SVS Flickr account. Relive the education sessions, the exhibit hall, the alumni receptions and the marvelous “Vascular Spectacular” gala. View, download and share these as much as you’d like. If you had your headshot taken at the SVS Booth, you can find that photo here.
If you attended the 2019 Vascular Annual Meeting in National Harbor, Md., be sure to check out the many photos now available on the SVS Flickr account. Relive the education sessions, the exhibit hall, the alumni receptions and the marvelous “Vascular Spectacular” gala. View, download and share these as much as you’d like. If you had your headshot taken at the SVS Booth, you can find that photo here.
Bakuchiol
Bakuchiol [(1E,3S)-3-ethenyl-3,7-dimethyl-1,6-octadien-1-yl]phenol, a prenylated phenolic monoterpene found in the seeds and leaves of various plants, particularly Psoralea corylifolia, has been used to treat a broad array of disorders, including skin conditions, in the traditional medical practices of China, Japan, and Korea, as well as Ayurvedic medicine in India.1-6 Specifically, the seeds of as well as cardiovascular diseases, nephritis, osteoporosis, and cancer.7-9
This primary active ingredient is reputed to exert antioxidant, antibacterial, anti-inflammatory, antiaging, and estrogen-like functions, and recent data suggest anticancer activity, including activity against skin cancer. Its antiaging properties manifest via preservation of cutaneous collagen.4 The plant itself has displayed a wide range of biological functions, such as antibacterial, anticancer, cytotoxic, cardiac, diaphoretic, diuretic, stimulant, aphrodisiac, and tonifying activities.8,9 A 2016 quantitative analysis of Psoralea corylifolia and seven of its standard constituents (psoralen, angelicin, neobavaisoflavone, psoralidin, isobavachalcone, bavachinin, and bakuchiol) using high-performance liquid chromatography revealed that bakuchiol is the strongest phytochemical ingredient in the plant, which the investigators found also confers neuroprotective and antineuroinflammatory benefits.3
Other species contain bakuchiol, and its biological activities have been harnessed in other folk medical traditions. The monoterpene is an important constituent found in Ulmus davidiana var. japonica, which is used for its anti-inflammatory properties in traditional Korean medicine.10 Further, bakuchiol and 3-hydroxy-bakuchiol have been identified as key components isolated from Psoralea glandulosa, which is a shrub used in Chilean folk medicine to treat cutaneous disorders engendered by bacteria and fungus.11 Topical applications of bakuchiol have been demonstrated to confer antiaging benefits.12 This column briefly identifies some of the various uses emerging for this compelling botanical agent.
Antiaging activities
In 2014, Yu et al. found that bakuchiol may impart antiaging benefits by supporting the cellular activity of the expression level of human skin fibroblasts (ESF-1), as well as production of collagen types I and III, while reducing the matrix metalloproteinase-1 mRNA expression.13
The same year, Chaudhuri et al. compared the skin care–related activities of retinol and bakuchiol, finding their gene expression profiles very similar. In addition, they observed that bakuchiol up-regulated collagen types I and IV in a DNA microarray study and stimulated type III collagen production in a model of mature fibroblasts. Further, the investigators formulated bakuchiol into a skin care product and tested it clinically, with twice daily applications over 12 weeks yielding significant amelioration in lines and wrinkles, pigmentation, elasticity, and firmness, as well as overall diminished photodamage without provoking redness. They concluded that bakuchiol can act as an antiaging agent through regulation of gene expression comparable to retinol.1
Retinoids without reactions?
In 2017, Ma et al. set out to synthesize and test in psoriatic cytokine–treated cultures of keratinocytes and organotypic skin substitutes a new substance created by combining two skin-active compounds (bakuchiol and salicylic acid) into bakuchiol salicylate (bakusylan), with the intention of rendering a novel functional retinoid. The researchers reported that the gene expression profile showed elimination of various retinoid-like proinflammatory responses, without a loss of normalizing activity. They concluded that their work may result in a new class of functional retinoids.14
Early this year, Dhaliwal et al. reported on a randomized, double-blind, 12-week study of 44 patients who applied either bakuchiol 0.5% cream twice daily or retinol 0.5% cream daily. Facial photographs were evaluated at baseline, 4, 8, and 12 weeks, and a blinded dermatologist rated pigmentation and erythema. Side effects were also noted by subjects in tolerability assessment questionnaires. Both compounds significantly reduced wrinkles and hyperpigmentation, with no statistical variance found between the two. More facial skin scaling and stinging was experienced by the retinol group. The investigators concluded that bakuchiol exhibits photoaging activity comparable with retinol and appears to be an emerging alternative to retinol because it is better tolerated.12 Notably, there is one report to date of an allergic reaction to topical bakuchiol.15
Topical combination therapies for hyperpigmentation, photodamage, and acne
Bakuchiol was a key ingredient incorporated into a 0.5% retinol treatment evaluated in a 12-week, open-label, single-center clinical-usage trial of 44 women with mild to moderate hyperpigmentation and photodamaged facial skin who took a dual product regimen. This 2016 study showed that the retinol and vitamin C facial regimen yielded a statistically significant amelioration in clinical grading of all parameters.16
A 2015 randomized controlled clinical trial in 111 subjects evaluated the use of adapalene 0.1% gel and a formulation containing bakuchiol, Ginkgo biloba extract, and mannitol in patients with acne. Patients were randomized to the adapalene and botanical formulation or adapalene and vehicle cream for 2 months. Both treatment groups experienced improvements according to all measured outcomes. The botanical formulation was associated with a statistically significant edge over the vehicle combination in reducing inflammatory lesions, investigator global assessment, and intensity of seborrhea. Quality of life was also perceived to be better with the combination of adapalene and the bakuchiol-containing product, which was deemed to be safe with good local tolerability.17
A subsequent evaluation by a different team also considered the antibacterial, anti-inflammatory, and antioxidative potential of this combination product via in vitro, ex vivo, and clinical studies. The work by Trompezinski et al. revealed that bakuchiol displays nearly twice the antioxidative potential asthat of vitamin E. The bakuchiol-containing cream was shown in acne patients to successfully regulate sebum composition by raising linolenic and sapienic acid levels while lowering oleic acid levels. Its efficacy against Propionibacterium acnes was also suggested by a decrease in the number of skin surface porphyrins. The investigators concluded that the formulation serves as an effective adjuvant acne treatment by attacking inflammation, dysseborrhea, and proliferation of Propionibacterium acnes.18
Anticancer activity
In 2016, Kim et al. demonstrated that bakuchiol exhibits chemopreventive activity by hindering epidermal growth factor (EGF)–induced neoplastic cell transformation. In what was the first mechanistic study to reveal molecular targets for the anticancer activity of this substance, the investigators found that bakuchiol also reduced the viability and suppressed anchorage-independent growth of A431 human epithelial carcinoma cells. They identified Hck, Blk, and p38 MAPK as the molecular targets of what they identified as a potent anticancer compound.2
Skin-whitening potential
In 2010, Ohno et al. found that bakuchiol, along with other ingredients, isolated from Piper longum demonstrated strong suppressive activity against melanin production in B16 mouse melanoma cells and may have potential to affect melanin synthesis in human skin.19 Further, with use of a new method for screening tyrosinase, Cheng et al. found in 2017 that four substances used in traditional Chinese medicine (quercetin, kaempferol, bavachinin, and bakuchiol) displayed the potential for inhibiting tyrosinase.20
Conclusion
A compound that acts like a retinoid – yielding antiacne and antiaging effects – without provoking irritation? Most dermatologists and their patients would say, sign me up. Bakuchiol, an active ingredient in various plants, especially Psoralea corylifolia, seems to present that kind of profile. While more research is necessary, experience with this herbal ingredient in traditional medicine and an increasing body of research, including clinical results, provides reasons for optimism that this ingredient may have a versatile role to play in topical skin care, particularly in its retinoid-like functions.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014), as well as a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems. Write to her at [email protected].
References
1. Chaudhuri RK et al. Int J Cosmet Sci. 2014 Jun;36(3):221-30.
2. Kim JE et al. Oncotarget. 2016 Mar 22;7(12):14616-27.
3. Kim YJ et al. Molecules. 2016 Aug 17. doi: 10.3390/molecules21081076.
4. Xin Z et al. Pharmacol Res. 2019 Mar;141:208-13.
5. Lev-Tov H. Br J Dermatol. 2019 Feb;180(2):253-4.
6. Shrestha S et al. J Ayurveda Integr Med. 2018 Jul - Sep; 9(3):209-12.
7. Li CC et al. Evid Based Complement Alternat Med. 2016. doi: 10.1155/2016/8108643.
8. Hu C et al. Fitoterapia. 2015 Oct;106:129-34.
9. Yan DM et al. J Ethnopharmacol. 2010 Apr 21;128(3):697-702.
10. Choi SY et al. J Med Food. 2010 Aug;13(4):1019-23.
11. Madrid A et al. J Ethnopharmacol. 2012 Dec 18;144(3):809-11.
12. Dhaliwal S et al. Br J Dermatol. 2019 Feb;180(2):289-96.
13. Yu Q et al. Zhong Yao Cai. 2014 Apr;37(4):632-5.
14. Ma S et al. Clin Exp Dermatol. 2017 Apr;42(3):251-60.
15. Malinauskiene L et al. Contact Dermatitis. 2019 Jun;80(6):398-9.
16. Herndon JH Jr, et al. J Drugs Dermatol. 2016 Apr;15(4):476-82.
17. Poláková K et al. Clin Cosmet Investig Dermatol. 2015 Apr 10;8:187-91.
18. Trompezinski S et al. Clin Cosmet Investig Dermatol. 2016 Aug 31;9:233-9.
19. Ohno O et al. Biosci Biotechnol Biochem. 2010;74(7):1504-6.
20. Cheng M et al. Electrophoresis. 2017 Feb;38(3-4):486-93.
Bakuchiol [(1E,3S)-3-ethenyl-3,7-dimethyl-1,6-octadien-1-yl]phenol, a prenylated phenolic monoterpene found in the seeds and leaves of various plants, particularly Psoralea corylifolia, has been used to treat a broad array of disorders, including skin conditions, in the traditional medical practices of China, Japan, and Korea, as well as Ayurvedic medicine in India.1-6 Specifically, the seeds of as well as cardiovascular diseases, nephritis, osteoporosis, and cancer.7-9
This primary active ingredient is reputed to exert antioxidant, antibacterial, anti-inflammatory, antiaging, and estrogen-like functions, and recent data suggest anticancer activity, including activity against skin cancer. Its antiaging properties manifest via preservation of cutaneous collagen.4 The plant itself has displayed a wide range of biological functions, such as antibacterial, anticancer, cytotoxic, cardiac, diaphoretic, diuretic, stimulant, aphrodisiac, and tonifying activities.8,9 A 2016 quantitative analysis of Psoralea corylifolia and seven of its standard constituents (psoralen, angelicin, neobavaisoflavone, psoralidin, isobavachalcone, bavachinin, and bakuchiol) using high-performance liquid chromatography revealed that bakuchiol is the strongest phytochemical ingredient in the plant, which the investigators found also confers neuroprotective and antineuroinflammatory benefits.3
Other species contain bakuchiol, and its biological activities have been harnessed in other folk medical traditions. The monoterpene is an important constituent found in Ulmus davidiana var. japonica, which is used for its anti-inflammatory properties in traditional Korean medicine.10 Further, bakuchiol and 3-hydroxy-bakuchiol have been identified as key components isolated from Psoralea glandulosa, which is a shrub used in Chilean folk medicine to treat cutaneous disorders engendered by bacteria and fungus.11 Topical applications of bakuchiol have been demonstrated to confer antiaging benefits.12 This column briefly identifies some of the various uses emerging for this compelling botanical agent.
Antiaging activities
In 2014, Yu et al. found that bakuchiol may impart antiaging benefits by supporting the cellular activity of the expression level of human skin fibroblasts (ESF-1), as well as production of collagen types I and III, while reducing the matrix metalloproteinase-1 mRNA expression.13
The same year, Chaudhuri et al. compared the skin care–related activities of retinol and bakuchiol, finding their gene expression profiles very similar. In addition, they observed that bakuchiol up-regulated collagen types I and IV in a DNA microarray study and stimulated type III collagen production in a model of mature fibroblasts. Further, the investigators formulated bakuchiol into a skin care product and tested it clinically, with twice daily applications over 12 weeks yielding significant amelioration in lines and wrinkles, pigmentation, elasticity, and firmness, as well as overall diminished photodamage without provoking redness. They concluded that bakuchiol can act as an antiaging agent through regulation of gene expression comparable to retinol.1
Retinoids without reactions?
In 2017, Ma et al. set out to synthesize and test in psoriatic cytokine–treated cultures of keratinocytes and organotypic skin substitutes a new substance created by combining two skin-active compounds (bakuchiol and salicylic acid) into bakuchiol salicylate (bakusylan), with the intention of rendering a novel functional retinoid. The researchers reported that the gene expression profile showed elimination of various retinoid-like proinflammatory responses, without a loss of normalizing activity. They concluded that their work may result in a new class of functional retinoids.14
Early this year, Dhaliwal et al. reported on a randomized, double-blind, 12-week study of 44 patients who applied either bakuchiol 0.5% cream twice daily or retinol 0.5% cream daily. Facial photographs were evaluated at baseline, 4, 8, and 12 weeks, and a blinded dermatologist rated pigmentation and erythema. Side effects were also noted by subjects in tolerability assessment questionnaires. Both compounds significantly reduced wrinkles and hyperpigmentation, with no statistical variance found between the two. More facial skin scaling and stinging was experienced by the retinol group. The investigators concluded that bakuchiol exhibits photoaging activity comparable with retinol and appears to be an emerging alternative to retinol because it is better tolerated.12 Notably, there is one report to date of an allergic reaction to topical bakuchiol.15
Topical combination therapies for hyperpigmentation, photodamage, and acne
Bakuchiol was a key ingredient incorporated into a 0.5% retinol treatment evaluated in a 12-week, open-label, single-center clinical-usage trial of 44 women with mild to moderate hyperpigmentation and photodamaged facial skin who took a dual product regimen. This 2016 study showed that the retinol and vitamin C facial regimen yielded a statistically significant amelioration in clinical grading of all parameters.16
A 2015 randomized controlled clinical trial in 111 subjects evaluated the use of adapalene 0.1% gel and a formulation containing bakuchiol, Ginkgo biloba extract, and mannitol in patients with acne. Patients were randomized to the adapalene and botanical formulation or adapalene and vehicle cream for 2 months. Both treatment groups experienced improvements according to all measured outcomes. The botanical formulation was associated with a statistically significant edge over the vehicle combination in reducing inflammatory lesions, investigator global assessment, and intensity of seborrhea. Quality of life was also perceived to be better with the combination of adapalene and the bakuchiol-containing product, which was deemed to be safe with good local tolerability.17
A subsequent evaluation by a different team also considered the antibacterial, anti-inflammatory, and antioxidative potential of this combination product via in vitro, ex vivo, and clinical studies. The work by Trompezinski et al. revealed that bakuchiol displays nearly twice the antioxidative potential asthat of vitamin E. The bakuchiol-containing cream was shown in acne patients to successfully regulate sebum composition by raising linolenic and sapienic acid levels while lowering oleic acid levels. Its efficacy against Propionibacterium acnes was also suggested by a decrease in the number of skin surface porphyrins. The investigators concluded that the formulation serves as an effective adjuvant acne treatment by attacking inflammation, dysseborrhea, and proliferation of Propionibacterium acnes.18
Anticancer activity
In 2016, Kim et al. demonstrated that bakuchiol exhibits chemopreventive activity by hindering epidermal growth factor (EGF)–induced neoplastic cell transformation. In what was the first mechanistic study to reveal molecular targets for the anticancer activity of this substance, the investigators found that bakuchiol also reduced the viability and suppressed anchorage-independent growth of A431 human epithelial carcinoma cells. They identified Hck, Blk, and p38 MAPK as the molecular targets of what they identified as a potent anticancer compound.2
Skin-whitening potential
In 2010, Ohno et al. found that bakuchiol, along with other ingredients, isolated from Piper longum demonstrated strong suppressive activity against melanin production in B16 mouse melanoma cells and may have potential to affect melanin synthesis in human skin.19 Further, with use of a new method for screening tyrosinase, Cheng et al. found in 2017 that four substances used in traditional Chinese medicine (quercetin, kaempferol, bavachinin, and bakuchiol) displayed the potential for inhibiting tyrosinase.20
Conclusion
A compound that acts like a retinoid – yielding antiacne and antiaging effects – without provoking irritation? Most dermatologists and their patients would say, sign me up. Bakuchiol, an active ingredient in various plants, especially Psoralea corylifolia, seems to present that kind of profile. While more research is necessary, experience with this herbal ingredient in traditional medicine and an increasing body of research, including clinical results, provides reasons for optimism that this ingredient may have a versatile role to play in topical skin care, particularly in its retinoid-like functions.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014), as well as a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems. Write to her at [email protected].
References
1. Chaudhuri RK et al. Int J Cosmet Sci. 2014 Jun;36(3):221-30.
2. Kim JE et al. Oncotarget. 2016 Mar 22;7(12):14616-27.
3. Kim YJ et al. Molecules. 2016 Aug 17. doi: 10.3390/molecules21081076.
4. Xin Z et al. Pharmacol Res. 2019 Mar;141:208-13.
5. Lev-Tov H. Br J Dermatol. 2019 Feb;180(2):253-4.
6. Shrestha S et al. J Ayurveda Integr Med. 2018 Jul - Sep; 9(3):209-12.
7. Li CC et al. Evid Based Complement Alternat Med. 2016. doi: 10.1155/2016/8108643.
8. Hu C et al. Fitoterapia. 2015 Oct;106:129-34.
9. Yan DM et al. J Ethnopharmacol. 2010 Apr 21;128(3):697-702.
10. Choi SY et al. J Med Food. 2010 Aug;13(4):1019-23.
11. Madrid A et al. J Ethnopharmacol. 2012 Dec 18;144(3):809-11.
12. Dhaliwal S et al. Br J Dermatol. 2019 Feb;180(2):289-96.
13. Yu Q et al. Zhong Yao Cai. 2014 Apr;37(4):632-5.
14. Ma S et al. Clin Exp Dermatol. 2017 Apr;42(3):251-60.
15. Malinauskiene L et al. Contact Dermatitis. 2019 Jun;80(6):398-9.
16. Herndon JH Jr, et al. J Drugs Dermatol. 2016 Apr;15(4):476-82.
17. Poláková K et al. Clin Cosmet Investig Dermatol. 2015 Apr 10;8:187-91.
18. Trompezinski S et al. Clin Cosmet Investig Dermatol. 2016 Aug 31;9:233-9.
19. Ohno O et al. Biosci Biotechnol Biochem. 2010;74(7):1504-6.
20. Cheng M et al. Electrophoresis. 2017 Feb;38(3-4):486-93.
Bakuchiol [(1E,3S)-3-ethenyl-3,7-dimethyl-1,6-octadien-1-yl]phenol, a prenylated phenolic monoterpene found in the seeds and leaves of various plants, particularly Psoralea corylifolia, has been used to treat a broad array of disorders, including skin conditions, in the traditional medical practices of China, Japan, and Korea, as well as Ayurvedic medicine in India.1-6 Specifically, the seeds of as well as cardiovascular diseases, nephritis, osteoporosis, and cancer.7-9
This primary active ingredient is reputed to exert antioxidant, antibacterial, anti-inflammatory, antiaging, and estrogen-like functions, and recent data suggest anticancer activity, including activity against skin cancer. Its antiaging properties manifest via preservation of cutaneous collagen.4 The plant itself has displayed a wide range of biological functions, such as antibacterial, anticancer, cytotoxic, cardiac, diaphoretic, diuretic, stimulant, aphrodisiac, and tonifying activities.8,9 A 2016 quantitative analysis of Psoralea corylifolia and seven of its standard constituents (psoralen, angelicin, neobavaisoflavone, psoralidin, isobavachalcone, bavachinin, and bakuchiol) using high-performance liquid chromatography revealed that bakuchiol is the strongest phytochemical ingredient in the plant, which the investigators found also confers neuroprotective and antineuroinflammatory benefits.3
Other species contain bakuchiol, and its biological activities have been harnessed in other folk medical traditions. The monoterpene is an important constituent found in Ulmus davidiana var. japonica, which is used for its anti-inflammatory properties in traditional Korean medicine.10 Further, bakuchiol and 3-hydroxy-bakuchiol have been identified as key components isolated from Psoralea glandulosa, which is a shrub used in Chilean folk medicine to treat cutaneous disorders engendered by bacteria and fungus.11 Topical applications of bakuchiol have been demonstrated to confer antiaging benefits.12 This column briefly identifies some of the various uses emerging for this compelling botanical agent.
Antiaging activities
In 2014, Yu et al. found that bakuchiol may impart antiaging benefits by supporting the cellular activity of the expression level of human skin fibroblasts (ESF-1), as well as production of collagen types I and III, while reducing the matrix metalloproteinase-1 mRNA expression.13
The same year, Chaudhuri et al. compared the skin care–related activities of retinol and bakuchiol, finding their gene expression profiles very similar. In addition, they observed that bakuchiol up-regulated collagen types I and IV in a DNA microarray study and stimulated type III collagen production in a model of mature fibroblasts. Further, the investigators formulated bakuchiol into a skin care product and tested it clinically, with twice daily applications over 12 weeks yielding significant amelioration in lines and wrinkles, pigmentation, elasticity, and firmness, as well as overall diminished photodamage without provoking redness. They concluded that bakuchiol can act as an antiaging agent through regulation of gene expression comparable to retinol.1
Retinoids without reactions?
In 2017, Ma et al. set out to synthesize and test in psoriatic cytokine–treated cultures of keratinocytes and organotypic skin substitutes a new substance created by combining two skin-active compounds (bakuchiol and salicylic acid) into bakuchiol salicylate (bakusylan), with the intention of rendering a novel functional retinoid. The researchers reported that the gene expression profile showed elimination of various retinoid-like proinflammatory responses, without a loss of normalizing activity. They concluded that their work may result in a new class of functional retinoids.14
Early this year, Dhaliwal et al. reported on a randomized, double-blind, 12-week study of 44 patients who applied either bakuchiol 0.5% cream twice daily or retinol 0.5% cream daily. Facial photographs were evaluated at baseline, 4, 8, and 12 weeks, and a blinded dermatologist rated pigmentation and erythema. Side effects were also noted by subjects in tolerability assessment questionnaires. Both compounds significantly reduced wrinkles and hyperpigmentation, with no statistical variance found between the two. More facial skin scaling and stinging was experienced by the retinol group. The investigators concluded that bakuchiol exhibits photoaging activity comparable with retinol and appears to be an emerging alternative to retinol because it is better tolerated.12 Notably, there is one report to date of an allergic reaction to topical bakuchiol.15
Topical combination therapies for hyperpigmentation, photodamage, and acne
Bakuchiol was a key ingredient incorporated into a 0.5% retinol treatment evaluated in a 12-week, open-label, single-center clinical-usage trial of 44 women with mild to moderate hyperpigmentation and photodamaged facial skin who took a dual product regimen. This 2016 study showed that the retinol and vitamin C facial regimen yielded a statistically significant amelioration in clinical grading of all parameters.16
A 2015 randomized controlled clinical trial in 111 subjects evaluated the use of adapalene 0.1% gel and a formulation containing bakuchiol, Ginkgo biloba extract, and mannitol in patients with acne. Patients were randomized to the adapalene and botanical formulation or adapalene and vehicle cream for 2 months. Both treatment groups experienced improvements according to all measured outcomes. The botanical formulation was associated with a statistically significant edge over the vehicle combination in reducing inflammatory lesions, investigator global assessment, and intensity of seborrhea. Quality of life was also perceived to be better with the combination of adapalene and the bakuchiol-containing product, which was deemed to be safe with good local tolerability.17
A subsequent evaluation by a different team also considered the antibacterial, anti-inflammatory, and antioxidative potential of this combination product via in vitro, ex vivo, and clinical studies. The work by Trompezinski et al. revealed that bakuchiol displays nearly twice the antioxidative potential asthat of vitamin E. The bakuchiol-containing cream was shown in acne patients to successfully regulate sebum composition by raising linolenic and sapienic acid levels while lowering oleic acid levels. Its efficacy against Propionibacterium acnes was also suggested by a decrease in the number of skin surface porphyrins. The investigators concluded that the formulation serves as an effective adjuvant acne treatment by attacking inflammation, dysseborrhea, and proliferation of Propionibacterium acnes.18
Anticancer activity
In 2016, Kim et al. demonstrated that bakuchiol exhibits chemopreventive activity by hindering epidermal growth factor (EGF)–induced neoplastic cell transformation. In what was the first mechanistic study to reveal molecular targets for the anticancer activity of this substance, the investigators found that bakuchiol also reduced the viability and suppressed anchorage-independent growth of A431 human epithelial carcinoma cells. They identified Hck, Blk, and p38 MAPK as the molecular targets of what they identified as a potent anticancer compound.2
Skin-whitening potential
In 2010, Ohno et al. found that bakuchiol, along with other ingredients, isolated from Piper longum demonstrated strong suppressive activity against melanin production in B16 mouse melanoma cells and may have potential to affect melanin synthesis in human skin.19 Further, with use of a new method for screening tyrosinase, Cheng et al. found in 2017 that four substances used in traditional Chinese medicine (quercetin, kaempferol, bavachinin, and bakuchiol) displayed the potential for inhibiting tyrosinase.20
Conclusion
A compound that acts like a retinoid – yielding antiacne and antiaging effects – without provoking irritation? Most dermatologists and their patients would say, sign me up. Bakuchiol, an active ingredient in various plants, especially Psoralea corylifolia, seems to present that kind of profile. While more research is necessary, experience with this herbal ingredient in traditional medicine and an increasing body of research, including clinical results, provides reasons for optimism that this ingredient may have a versatile role to play in topical skin care, particularly in its retinoid-like functions.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote two textbooks: “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and “Cosmeceuticals and Cosmetic Ingredients” (New York: McGraw-Hill, 2014), as well as a New York Times Best Sellers book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Evolus, Galderma, and Revance. She is the founder and CEO of Skin Type Solutions Franchise Systems. Write to her at [email protected].
References
1. Chaudhuri RK et al. Int J Cosmet Sci. 2014 Jun;36(3):221-30.
2. Kim JE et al. Oncotarget. 2016 Mar 22;7(12):14616-27.
3. Kim YJ et al. Molecules. 2016 Aug 17. doi: 10.3390/molecules21081076.
4. Xin Z et al. Pharmacol Res. 2019 Mar;141:208-13.
5. Lev-Tov H. Br J Dermatol. 2019 Feb;180(2):253-4.
6. Shrestha S et al. J Ayurveda Integr Med. 2018 Jul - Sep; 9(3):209-12.
7. Li CC et al. Evid Based Complement Alternat Med. 2016. doi: 10.1155/2016/8108643.
8. Hu C et al. Fitoterapia. 2015 Oct;106:129-34.
9. Yan DM et al. J Ethnopharmacol. 2010 Apr 21;128(3):697-702.
10. Choi SY et al. J Med Food. 2010 Aug;13(4):1019-23.
11. Madrid A et al. J Ethnopharmacol. 2012 Dec 18;144(3):809-11.
12. Dhaliwal S et al. Br J Dermatol. 2019 Feb;180(2):289-96.
13. Yu Q et al. Zhong Yao Cai. 2014 Apr;37(4):632-5.
14. Ma S et al. Clin Exp Dermatol. 2017 Apr;42(3):251-60.
15. Malinauskiene L et al. Contact Dermatitis. 2019 Jun;80(6):398-9.
16. Herndon JH Jr, et al. J Drugs Dermatol. 2016 Apr;15(4):476-82.
17. Poláková K et al. Clin Cosmet Investig Dermatol. 2015 Apr 10;8:187-91.
18. Trompezinski S et al. Clin Cosmet Investig Dermatol. 2016 Aug 31;9:233-9.
19. Ohno O et al. Biosci Biotechnol Biochem. 2010;74(7):1504-6.
20. Cheng M et al. Electrophoresis. 2017 Feb;38(3-4):486-93.
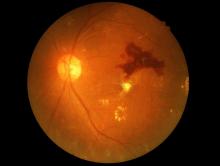
Microvascular disease: An independent and exacerbating risk factor for amputation
Individuals with microvascular disease (MVD) showed a significantly increased risk of lower limb amputation in the absence of peripheral artery disease (PAD), according to the results of a large database analysis published online in Circulation.
Furthermore, those who had both MVD and PAD had a greater than 20-fold increased risk of amputation than if they had either PAD or MVD alone, according to Joshua A. Beckman, MD, of Vanderbilt University, Nashville, Tenn., and colleagues.
“The novelty of these findings becomes clear when put into the current framework of critical limb ischemia,” they wrote.
“In a recent state of the art review of [critical limb ischemia], MVD as a whole or its components did not receive a single mention. Our work shows that MVD helps identify a population not previously considered at particularly high risk for amputation and, when added to PAD, identify a group of patients at very high risk for amputation,” they continued.
Dr. Beckman and colleagues assessed individuals in the Veterans Aging Cohort Study (VACS), a prospective longitudinal cohort of veterans. They included all VACS participants who were alive as of April 1, 2003 with the baseline as a participant’s first clinic visit on or after this date. Participants were followed from baseline to the minimum of: date of lower extremity amputation, death, or Dec. 31, 2014.
They assessed four levels of vascular involvement: neither MVD nor PAD, MVD alone, PAD alone, and MVD plus PAD, with the primary outcome being lower limb amputation, all based on a variety of measures including appropriate ICD-9 or CPT codes.
The rate of incident amputation over a median of 9.3 years of follow-up was 1.16 per 1000 person-years. At the time of amputation, retinopathy was present in 69%, nephropathy in 67%, and neuropathy in 78% of participants.
After multivariable adjustment for 216 demographic characteristics, cardiovascular disease risk factors, and other potential confounders, they found that, compared with participants without either vascular disease, the presence of MVD alone was associated with a 3.7-fold increased risk of amputation, PAD alone conferred a 13.9-fold elevated risk of amputation, and the combination of PAD and MVD was associated with a 22.7-fold increased risk of amputation.
They also found that the location of amputation also varied depending on the type of vascular disease at the time of amputation.
Participants with MVD alone accounted for 18% of all amputations, 21% of below ankle amputations, 15% of below knee amputations, and 6% of all above knee amputations. Participants with PAD alone accounted for 22% of all amputations, 17% of below ankle, 25% of below knee, and 39% of above knee amputations. The combination of MVD and PAD accounted for 45% of all amputation and caused the most amputation at all limb levels. In addition, they found a statistically significant variation in vascular involvement and level of amputation, with MVD more likely to cause a below-ankle amputation and PAD more likely to cause below- and above-knee amputations (P less than .001)
“MVD likely participates importantly in the development of adverse limb events in PAD and suggests additional patient populations who may benefit from greater foot surveillance to minimize amputation,” the researchers concluded.
The study was supported by grants from the American Heart Association. Dr. Beckman reported consulting for multiple pharmaceutical companies and serving on the DSMC for Bayer and Novartis.
SOURCE: Beckman JA et al. Circulation. 2019. doi: 10.1161/CIRCULATIONAHA.119.040672.
Individuals with microvascular disease (MVD) showed a significantly increased risk of lower limb amputation in the absence of peripheral artery disease (PAD), according to the results of a large database analysis published online in Circulation.
Furthermore, those who had both MVD and PAD had a greater than 20-fold increased risk of amputation than if they had either PAD or MVD alone, according to Joshua A. Beckman, MD, of Vanderbilt University, Nashville, Tenn., and colleagues.
“The novelty of these findings becomes clear when put into the current framework of critical limb ischemia,” they wrote.
“In a recent state of the art review of [critical limb ischemia], MVD as a whole or its components did not receive a single mention. Our work shows that MVD helps identify a population not previously considered at particularly high risk for amputation and, when added to PAD, identify a group of patients at very high risk for amputation,” they continued.
Dr. Beckman and colleagues assessed individuals in the Veterans Aging Cohort Study (VACS), a prospective longitudinal cohort of veterans. They included all VACS participants who were alive as of April 1, 2003 with the baseline as a participant’s first clinic visit on or after this date. Participants were followed from baseline to the minimum of: date of lower extremity amputation, death, or Dec. 31, 2014.
They assessed four levels of vascular involvement: neither MVD nor PAD, MVD alone, PAD alone, and MVD plus PAD, with the primary outcome being lower limb amputation, all based on a variety of measures including appropriate ICD-9 or CPT codes.
The rate of incident amputation over a median of 9.3 years of follow-up was 1.16 per 1000 person-years. At the time of amputation, retinopathy was present in 69%, nephropathy in 67%, and neuropathy in 78% of participants.
After multivariable adjustment for 216 demographic characteristics, cardiovascular disease risk factors, and other potential confounders, they found that, compared with participants without either vascular disease, the presence of MVD alone was associated with a 3.7-fold increased risk of amputation, PAD alone conferred a 13.9-fold elevated risk of amputation, and the combination of PAD and MVD was associated with a 22.7-fold increased risk of amputation.
They also found that the location of amputation also varied depending on the type of vascular disease at the time of amputation.
Participants with MVD alone accounted for 18% of all amputations, 21% of below ankle amputations, 15% of below knee amputations, and 6% of all above knee amputations. Participants with PAD alone accounted for 22% of all amputations, 17% of below ankle, 25% of below knee, and 39% of above knee amputations. The combination of MVD and PAD accounted for 45% of all amputation and caused the most amputation at all limb levels. In addition, they found a statistically significant variation in vascular involvement and level of amputation, with MVD more likely to cause a below-ankle amputation and PAD more likely to cause below- and above-knee amputations (P less than .001)
“MVD likely participates importantly in the development of adverse limb events in PAD and suggests additional patient populations who may benefit from greater foot surveillance to minimize amputation,” the researchers concluded.
The study was supported by grants from the American Heart Association. Dr. Beckman reported consulting for multiple pharmaceutical companies and serving on the DSMC for Bayer and Novartis.
SOURCE: Beckman JA et al. Circulation. 2019. doi: 10.1161/CIRCULATIONAHA.119.040672.
Individuals with microvascular disease (MVD) showed a significantly increased risk of lower limb amputation in the absence of peripheral artery disease (PAD), according to the results of a large database analysis published online in Circulation.
Furthermore, those who had both MVD and PAD had a greater than 20-fold increased risk of amputation than if they had either PAD or MVD alone, according to Joshua A. Beckman, MD, of Vanderbilt University, Nashville, Tenn., and colleagues.
“The novelty of these findings becomes clear when put into the current framework of critical limb ischemia,” they wrote.
“In a recent state of the art review of [critical limb ischemia], MVD as a whole or its components did not receive a single mention. Our work shows that MVD helps identify a population not previously considered at particularly high risk for amputation and, when added to PAD, identify a group of patients at very high risk for amputation,” they continued.
Dr. Beckman and colleagues assessed individuals in the Veterans Aging Cohort Study (VACS), a prospective longitudinal cohort of veterans. They included all VACS participants who were alive as of April 1, 2003 with the baseline as a participant’s first clinic visit on or after this date. Participants were followed from baseline to the minimum of: date of lower extremity amputation, death, or Dec. 31, 2014.
They assessed four levels of vascular involvement: neither MVD nor PAD, MVD alone, PAD alone, and MVD plus PAD, with the primary outcome being lower limb amputation, all based on a variety of measures including appropriate ICD-9 or CPT codes.
The rate of incident amputation over a median of 9.3 years of follow-up was 1.16 per 1000 person-years. At the time of amputation, retinopathy was present in 69%, nephropathy in 67%, and neuropathy in 78% of participants.
After multivariable adjustment for 216 demographic characteristics, cardiovascular disease risk factors, and other potential confounders, they found that, compared with participants without either vascular disease, the presence of MVD alone was associated with a 3.7-fold increased risk of amputation, PAD alone conferred a 13.9-fold elevated risk of amputation, and the combination of PAD and MVD was associated with a 22.7-fold increased risk of amputation.
They also found that the location of amputation also varied depending on the type of vascular disease at the time of amputation.
Participants with MVD alone accounted for 18% of all amputations, 21% of below ankle amputations, 15% of below knee amputations, and 6% of all above knee amputations. Participants with PAD alone accounted for 22% of all amputations, 17% of below ankle, 25% of below knee, and 39% of above knee amputations. The combination of MVD and PAD accounted for 45% of all amputation and caused the most amputation at all limb levels. In addition, they found a statistically significant variation in vascular involvement and level of amputation, with MVD more likely to cause a below-ankle amputation and PAD more likely to cause below- and above-knee amputations (P less than .001)
“MVD likely participates importantly in the development of adverse limb events in PAD and suggests additional patient populations who may benefit from greater foot surveillance to minimize amputation,” the researchers concluded.
The study was supported by grants from the American Heart Association. Dr. Beckman reported consulting for multiple pharmaceutical companies and serving on the DSMC for Bayer and Novartis.
SOURCE: Beckman JA et al. Circulation. 2019. doi: 10.1161/CIRCULATIONAHA.119.040672.
FROM CIRCULATION
Key clinical point: Microvascular disease yielded a 3.7-fold increased risk of lower limb amputation.
Major finding:
Study details: Database analysis of 125,674 participants in the Veterans Aging Cohort Study from April 2003 through December 2014.
Disclosures: The study was supported by grants from the American Heart Association. Dr. Beckman reported consulting for multiple pharmaceutical companies and serving on the DSMC for Bayer and Novartis.
Source: Beckman JA et al. Circulation. 2019. doi: 10.1161/CIRCULATIONAHA.119.040672.
Y 2 the ED? (Why patients go to the emergency department)
Along with terminal care and inflated drug prices, the excessive number of “inappropriate” ED visits often is cited as a major driver of health care costs in the United States. Why do so many patients choose to go to the ED for complaints that might be better or more economically treated in another setting?
A report by two researchers in the division of emergency medicine at the Boston Children’s Hospital that appeared in the June 2019 Pediatrics suggests that, at least for pediatric patients, “increased insurance coverage neither drove nor counteracted” the recent trends in ED visits. (“Trends in Pediatric Emergency Department Use After the Affordable Care Act,” Pediatrics. 2019 Jun 1. doi: 10.1542/peds.2018-3542).
I guess it’s not surprising – and somewhat comforting – to learn that, when parents believe their child has an emergent condition they give little thought to the cost of care. Is the trend of increasing ED use a result of an evolving definition of an “emergency”? Your grandparents, or certainly your great grandparents, might claim that, when they were young most minor injuries were handled at home, or at least in the neighborhood by someone with first aid experience who wasn’t put off by the sight of blood. However, a trend away from self-reliance in everything from food preparation to auto repair, combined with media overexposure to the serious complications of apparently minor illness and injury, has left most parents feeling fearful and helpless in the face of adversity.
We have to accept as a given that many parents are going to interpret their child’s situation as emergent, even though you and I might not. But what are the factors that prompt a concerned parent to take his child to the ED instead of a physician’s office? It may simply be the path of least resistance. The parent’s past experience may include frustrating and time-consuming attempts to navigate a clunky phone system only to be met by a receptionist or triage nurse who seems more committed to deflecting calls and protecting the physician’s schedule than getting the patient seen.
The call may miraculously get through to someone with a caring voice and the patience to listen, but the parent then learns that the office doesn’t do minor wound care or he is told that the physician almost certainly will want to do an x-ray of any injured extremity and that the ED is a better choice. It doesn’t take very many scenarios like this to prompt a parent to make his first and only call to the ED. To some extent, physician behavior has helped mold parents’ definition of an emergency.
We are encouraged to make our offices a “medical home.” However, it appears the medical home model is one that is built around chronic conditions and behavioral problems and gives little attention to the acute complaints. When you came running into the house with a skinned knee, did your mother tell to you go across the street to the neighbor’s house because blood made her squeamish and she didn’t have any bandages?
There are ways to structure an office and a schedule which are more welcoming to patients with minor emergencies, and I know it is a difficult sell to physicians who are handcuffed by their EHRs and already overwhelmed by patients with time-consuming behavioral complaints. However, if your practice is facing competition from pop-up urgent care centers or if you are increasingly troubled that your patients are receiving fragmented care, it may not be too late to make your practice into a true medical home that welcomes minor emergencies.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Along with terminal care and inflated drug prices, the excessive number of “inappropriate” ED visits often is cited as a major driver of health care costs in the United States. Why do so many patients choose to go to the ED for complaints that might be better or more economically treated in another setting?
A report by two researchers in the division of emergency medicine at the Boston Children’s Hospital that appeared in the June 2019 Pediatrics suggests that, at least for pediatric patients, “increased insurance coverage neither drove nor counteracted” the recent trends in ED visits. (“Trends in Pediatric Emergency Department Use After the Affordable Care Act,” Pediatrics. 2019 Jun 1. doi: 10.1542/peds.2018-3542).
I guess it’s not surprising – and somewhat comforting – to learn that, when parents believe their child has an emergent condition they give little thought to the cost of care. Is the trend of increasing ED use a result of an evolving definition of an “emergency”? Your grandparents, or certainly your great grandparents, might claim that, when they were young most minor injuries were handled at home, or at least in the neighborhood by someone with first aid experience who wasn’t put off by the sight of blood. However, a trend away from self-reliance in everything from food preparation to auto repair, combined with media overexposure to the serious complications of apparently minor illness and injury, has left most parents feeling fearful and helpless in the face of adversity.
We have to accept as a given that many parents are going to interpret their child’s situation as emergent, even though you and I might not. But what are the factors that prompt a concerned parent to take his child to the ED instead of a physician’s office? It may simply be the path of least resistance. The parent’s past experience may include frustrating and time-consuming attempts to navigate a clunky phone system only to be met by a receptionist or triage nurse who seems more committed to deflecting calls and protecting the physician’s schedule than getting the patient seen.
The call may miraculously get through to someone with a caring voice and the patience to listen, but the parent then learns that the office doesn’t do minor wound care or he is told that the physician almost certainly will want to do an x-ray of any injured extremity and that the ED is a better choice. It doesn’t take very many scenarios like this to prompt a parent to make his first and only call to the ED. To some extent, physician behavior has helped mold parents’ definition of an emergency.
We are encouraged to make our offices a “medical home.” However, it appears the medical home model is one that is built around chronic conditions and behavioral problems and gives little attention to the acute complaints. When you came running into the house with a skinned knee, did your mother tell to you go across the street to the neighbor’s house because blood made her squeamish and she didn’t have any bandages?
There are ways to structure an office and a schedule which are more welcoming to patients with minor emergencies, and I know it is a difficult sell to physicians who are handcuffed by their EHRs and already overwhelmed by patients with time-consuming behavioral complaints. However, if your practice is facing competition from pop-up urgent care centers or if you are increasingly troubled that your patients are receiving fragmented care, it may not be too late to make your practice into a true medical home that welcomes minor emergencies.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Along with terminal care and inflated drug prices, the excessive number of “inappropriate” ED visits often is cited as a major driver of health care costs in the United States. Why do so many patients choose to go to the ED for complaints that might be better or more economically treated in another setting?
A report by two researchers in the division of emergency medicine at the Boston Children’s Hospital that appeared in the June 2019 Pediatrics suggests that, at least for pediatric patients, “increased insurance coverage neither drove nor counteracted” the recent trends in ED visits. (“Trends in Pediatric Emergency Department Use After the Affordable Care Act,” Pediatrics. 2019 Jun 1. doi: 10.1542/peds.2018-3542).
I guess it’s not surprising – and somewhat comforting – to learn that, when parents believe their child has an emergent condition they give little thought to the cost of care. Is the trend of increasing ED use a result of an evolving definition of an “emergency”? Your grandparents, or certainly your great grandparents, might claim that, when they were young most minor injuries were handled at home, or at least in the neighborhood by someone with first aid experience who wasn’t put off by the sight of blood. However, a trend away from self-reliance in everything from food preparation to auto repair, combined with media overexposure to the serious complications of apparently minor illness and injury, has left most parents feeling fearful and helpless in the face of adversity.
We have to accept as a given that many parents are going to interpret their child’s situation as emergent, even though you and I might not. But what are the factors that prompt a concerned parent to take his child to the ED instead of a physician’s office? It may simply be the path of least resistance. The parent’s past experience may include frustrating and time-consuming attempts to navigate a clunky phone system only to be met by a receptionist or triage nurse who seems more committed to deflecting calls and protecting the physician’s schedule than getting the patient seen.
The call may miraculously get through to someone with a caring voice and the patience to listen, but the parent then learns that the office doesn’t do minor wound care or he is told that the physician almost certainly will want to do an x-ray of any injured extremity and that the ED is a better choice. It doesn’t take very many scenarios like this to prompt a parent to make his first and only call to the ED. To some extent, physician behavior has helped mold parents’ definition of an emergency.
We are encouraged to make our offices a “medical home.” However, it appears the medical home model is one that is built around chronic conditions and behavioral problems and gives little attention to the acute complaints. When you came running into the house with a skinned knee, did your mother tell to you go across the street to the neighbor’s house because blood made her squeamish and she didn’t have any bandages?
There are ways to structure an office and a schedule which are more welcoming to patients with minor emergencies, and I know it is a difficult sell to physicians who are handcuffed by their EHRs and already overwhelmed by patients with time-consuming behavioral complaints. However, if your practice is facing competition from pop-up urgent care centers or if you are increasingly troubled that your patients are receiving fragmented care, it may not be too late to make your practice into a true medical home that welcomes minor emergencies.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Acquired MMR immunity doesn’t last to age 1 year
according to results of a study in Vaccine.
María José Cilleruelo, PhD, of Hospital Universitario Puerta de Hierro in Majadahonda, Spain, and colleagues showed that, although most infants acquire some protective antibodies against MMR from their mothers during gestation, most have lost this protection as early as 3 months of age. This single-center, observational, prospective study was conducted between October 2013 and December 2014, and it began with 146 mother-child pairs, with 99 remaining in follow-up at 3 months, 77 at 6 months, 63 at 9 months, and 30 at 12 months. For measles, 88% of newborns were seropositive, but only 19% were at 3 months; for mumps, 70% of newborns were seropositive, but only 11% were at 3 months; and for rubella, 91% of newborns were seropositive, but only 13% were at 3 months. No infants were seropositive for mumps or rubella at 9 months, and only 2% were for measles. No infants were seropositive for any of these viruses by 12 months of age.
The investigators noted that, given Spain (where the study was conducted) is a country that gives the first MMR vaccine at 12 months of life, these declining titers can leave most infants vulnerable to those viruses before then.
“We suggest that it may be worth considering administering the first dose of MMR vaccine before 12 months of age,” the investigators concluded, although they advised studies be undertaken into the efficacy and safety of administration of that vaccine in infants younger than 12 months. They noted that the biggest limitation of this study was the high percentage of loss to follow-up, which limited the statistical power to make comparisons.
The study was funded by the Fondo de Investigación Sanitaria, and one of the authors was funded by CIBER de Epidemiología y Salud Pública. The authors declared that there are no conflicts of interest.
SOURCE: Cilleruelo MJ et al. Vaccine. 2019;37:4164-71.
according to results of a study in Vaccine.
María José Cilleruelo, PhD, of Hospital Universitario Puerta de Hierro in Majadahonda, Spain, and colleagues showed that, although most infants acquire some protective antibodies against MMR from their mothers during gestation, most have lost this protection as early as 3 months of age. This single-center, observational, prospective study was conducted between October 2013 and December 2014, and it began with 146 mother-child pairs, with 99 remaining in follow-up at 3 months, 77 at 6 months, 63 at 9 months, and 30 at 12 months. For measles, 88% of newborns were seropositive, but only 19% were at 3 months; for mumps, 70% of newborns were seropositive, but only 11% were at 3 months; and for rubella, 91% of newborns were seropositive, but only 13% were at 3 months. No infants were seropositive for mumps or rubella at 9 months, and only 2% were for measles. No infants were seropositive for any of these viruses by 12 months of age.
The investigators noted that, given Spain (where the study was conducted) is a country that gives the first MMR vaccine at 12 months of life, these declining titers can leave most infants vulnerable to those viruses before then.
“We suggest that it may be worth considering administering the first dose of MMR vaccine before 12 months of age,” the investigators concluded, although they advised studies be undertaken into the efficacy and safety of administration of that vaccine in infants younger than 12 months. They noted that the biggest limitation of this study was the high percentage of loss to follow-up, which limited the statistical power to make comparisons.
The study was funded by the Fondo de Investigación Sanitaria, and one of the authors was funded by CIBER de Epidemiología y Salud Pública. The authors declared that there are no conflicts of interest.
SOURCE: Cilleruelo MJ et al. Vaccine. 2019;37:4164-71.
according to results of a study in Vaccine.
María José Cilleruelo, PhD, of Hospital Universitario Puerta de Hierro in Majadahonda, Spain, and colleagues showed that, although most infants acquire some protective antibodies against MMR from their mothers during gestation, most have lost this protection as early as 3 months of age. This single-center, observational, prospective study was conducted between October 2013 and December 2014, and it began with 146 mother-child pairs, with 99 remaining in follow-up at 3 months, 77 at 6 months, 63 at 9 months, and 30 at 12 months. For measles, 88% of newborns were seropositive, but only 19% were at 3 months; for mumps, 70% of newborns were seropositive, but only 11% were at 3 months; and for rubella, 91% of newborns were seropositive, but only 13% were at 3 months. No infants were seropositive for mumps or rubella at 9 months, and only 2% were for measles. No infants were seropositive for any of these viruses by 12 months of age.
The investigators noted that, given Spain (where the study was conducted) is a country that gives the first MMR vaccine at 12 months of life, these declining titers can leave most infants vulnerable to those viruses before then.
“We suggest that it may be worth considering administering the first dose of MMR vaccine before 12 months of age,” the investigators concluded, although they advised studies be undertaken into the efficacy and safety of administration of that vaccine in infants younger than 12 months. They noted that the biggest limitation of this study was the high percentage of loss to follow-up, which limited the statistical power to make comparisons.
The study was funded by the Fondo de Investigación Sanitaria, and one of the authors was funded by CIBER de Epidemiología y Salud Pública. The authors declared that there are no conflicts of interest.
SOURCE: Cilleruelo MJ et al. Vaccine. 2019;37:4164-71.
FROM VACCINE
Consider cutaneous endometriosis in women with umbilical lesions
according to Liza Raffi of the University of Southern California, Los Angeles, and associates.
The report, published in the International Journal of Women’s Dermatology, detailed a case of a woman aged 41 years who presented with a 5-month history of a painful firm subcutaneous nodule in the umbilicus and flares of pain during menstrual periods. Her past history indicated a missed miscarriage (removed by dilation and curettage) and laparoscopic left salpingectomy for a ruptured ectopic pregnancy.
At presentation, the woman reported undergoing fertility treatments including subcutaneous injections of follitropin beta and choriogonadotropin alfa.
Because of the patient’s history of salpingectomy and painful menstrual periods, her physicians suspected cutaneous endometriosis. An ultrasound was performed to rule out fistula, and then a punch biopsy of the nodule was performed. The biopsy showed endometrial glands with encompassing fibrotic stroma, which was consistent with cutaneous endometriosis, likely transplanted during the laparoscopic port site entry during salpingectomy.
The patient chose to undergo surgery for excision of the nodule, declining hormonal therapy because she was undergoing fertility treatment.
“The differential diagnosis of umbilical lesions with similar presentation includes keloid, dermatofibroma, dermatofibrosarcoma protuberans, and cutaneous metastasis of cancer,” the investigators wrote. “Ultimately, patients should be referred to obstetrics & gynecology if they describe classic symptoms including pain with menses, dyspareunia, and infertility and wish to explore diagnostic and therapeutic options.”
Ms. Raffi and associates reported they had no conflicts of interest. There was no external funding.
SOURCE: Raffi L et al. Int J Womens Dermatol. 2019 Jul 2. doi: 10.1016/j.ijwd.2019.06.025.
according to Liza Raffi of the University of Southern California, Los Angeles, and associates.
The report, published in the International Journal of Women’s Dermatology, detailed a case of a woman aged 41 years who presented with a 5-month history of a painful firm subcutaneous nodule in the umbilicus and flares of pain during menstrual periods. Her past history indicated a missed miscarriage (removed by dilation and curettage) and laparoscopic left salpingectomy for a ruptured ectopic pregnancy.
At presentation, the woman reported undergoing fertility treatments including subcutaneous injections of follitropin beta and choriogonadotropin alfa.
Because of the patient’s history of salpingectomy and painful menstrual periods, her physicians suspected cutaneous endometriosis. An ultrasound was performed to rule out fistula, and then a punch biopsy of the nodule was performed. The biopsy showed endometrial glands with encompassing fibrotic stroma, which was consistent with cutaneous endometriosis, likely transplanted during the laparoscopic port site entry during salpingectomy.
The patient chose to undergo surgery for excision of the nodule, declining hormonal therapy because she was undergoing fertility treatment.
“The differential diagnosis of umbilical lesions with similar presentation includes keloid, dermatofibroma, dermatofibrosarcoma protuberans, and cutaneous metastasis of cancer,” the investigators wrote. “Ultimately, patients should be referred to obstetrics & gynecology if they describe classic symptoms including pain with menses, dyspareunia, and infertility and wish to explore diagnostic and therapeutic options.”
Ms. Raffi and associates reported they had no conflicts of interest. There was no external funding.
SOURCE: Raffi L et al. Int J Womens Dermatol. 2019 Jul 2. doi: 10.1016/j.ijwd.2019.06.025.
according to Liza Raffi of the University of Southern California, Los Angeles, and associates.
The report, published in the International Journal of Women’s Dermatology, detailed a case of a woman aged 41 years who presented with a 5-month history of a painful firm subcutaneous nodule in the umbilicus and flares of pain during menstrual periods. Her past history indicated a missed miscarriage (removed by dilation and curettage) and laparoscopic left salpingectomy for a ruptured ectopic pregnancy.
At presentation, the woman reported undergoing fertility treatments including subcutaneous injections of follitropin beta and choriogonadotropin alfa.
Because of the patient’s history of salpingectomy and painful menstrual periods, her physicians suspected cutaneous endometriosis. An ultrasound was performed to rule out fistula, and then a punch biopsy of the nodule was performed. The biopsy showed endometrial glands with encompassing fibrotic stroma, which was consistent with cutaneous endometriosis, likely transplanted during the laparoscopic port site entry during salpingectomy.
The patient chose to undergo surgery for excision of the nodule, declining hormonal therapy because she was undergoing fertility treatment.
“The differential diagnosis of umbilical lesions with similar presentation includes keloid, dermatofibroma, dermatofibrosarcoma protuberans, and cutaneous metastasis of cancer,” the investigators wrote. “Ultimately, patients should be referred to obstetrics & gynecology if they describe classic symptoms including pain with menses, dyspareunia, and infertility and wish to explore diagnostic and therapeutic options.”
Ms. Raffi and associates reported they had no conflicts of interest. There was no external funding.
SOURCE: Raffi L et al. Int J Womens Dermatol. 2019 Jul 2. doi: 10.1016/j.ijwd.2019.06.025.
FROM THE INTERNATIONAL JOURNAL OF WOMEN’S DERMATOLOGY