User login
In Bladder Cancer, New Systemic Treatments Arise
MINNEAPOLIS -- A new era of systemic treatment for bladder cancer has arrived, a US Department of Veterans Affairs (VA) hematologist/oncologist told colleagues, and more changes await on the horizon.
“After a historically long dry spell, you're seeing novel drugs and combinations under investigation,” said Elizabeth Henry, MD, of Edward Hines, Jr. VA Hospital and Loyola University Medical Center in Chicago, Illinois, in a presentation at the annual meeting of the Association of VA Hematology/Oncology. “Our treatment paradigm will almost certainly continue to change.”
There’s a major need for new approaches in bladder cancer, Dr. Henry said. While the median survival for patients with metastatic disease treated has risen, it remains low at about 15 months. And, she said, the 5-year survival rate is about 15% with modern treatments.
Platinum-based chemotherapy is still the first-line treatment, she said, and cisplatin-based combos remain the standard. However, many patients are not eligible to take cisplatin because of factors such as reduced performance status, impaired renal function, peripheral neuropathy, hearing loss and heart failure. “Many patients have renal insufficiency and are platinum ineligible from the get-go,” Dr. Henry said.
In these patients, immune checkpoint inhibitors are an option, but research suggests they may lead to poorer survival in those with PDL1-low tumors. In 2018, the US Food and Drug Administration (FDA) advised their use as first-line treatment only in PDL1-high, cisplatin-ineligible patients or those who can’t undergo chemotherapy, she said.
As second-line therapy after platinum treatment, Dr. Henry said, several drugs targeting the PD1/PDL1 pathway are now FDA-approved with response rates at 15% to 25%; only 1 (pembrolizumab) has level 1 evidence from a phase 3 randomized clinical trial.
Single-agent chemotherapy is an option for patients who have worsened or cannot undergo treatment with immune checkpoint inhibitors. However, according to Dr. Henry, response rates are low (about 10%-15%) and there's no prospective or randomized control trial data showing a survival benefit.
What now? Targeted approaches are entering the picture. For example, fibroblast growth factor receptor inhibitors, which target a pathway that often mutates in bladder cancer. One drug, erdafitinib (Balversa), received FDA approval earlier this year based on a phase 2 trial data that showed an objective response rate (ORR) of 40%. Dr. Henry cautioned that unusual adverse effects can occur, including hyperphosphatemia (a disorder that boosts phosphate levels), ocular toxicity (which can lead to retinal detachment), and toxicity of the skin and hair.
“Patients need to be closely followed if they're starting this as a targeted drug,” Dr. Henry said.
Anti-Nectin-4 antibody drug conjugate, which targets urothelial carcinomas with uniformly high expression of the Nectin-4 cell surface marker, also is showing promise. Recent research suggests a “remarkable” ORR of 42% and nearly 8 months duration of response, she said.
Adverse effects include rash, peripheral neuropathy, and hyperglycemia. “Overall, this is thought to be a relatively well-tolerated therapy,” she said.
In terms of other advances, “we are moving closer to an era of molecular subtype-specific therapeutic strategies,” Dr. Henry said, and the National Comprehensive Cancer Network recommends early molecular testing in stage IIIB/IV urothelial cancer. “It can help facilitate treatment decisions and prevent delays in later lines of therapy, although we're still limited by development of individualized biomarker assays for specific drugs.”
Moving forward, she said, “continued research is needed to learn how to incorporate predictive molecular profiles to optimize treatment selection.”
Dr. Henry reported no relevant disclosures.
MINNEAPOLIS -- A new era of systemic treatment for bladder cancer has arrived, a US Department of Veterans Affairs (VA) hematologist/oncologist told colleagues, and more changes await on the horizon.
“After a historically long dry spell, you're seeing novel drugs and combinations under investigation,” said Elizabeth Henry, MD, of Edward Hines, Jr. VA Hospital and Loyola University Medical Center in Chicago, Illinois, in a presentation at the annual meeting of the Association of VA Hematology/Oncology. “Our treatment paradigm will almost certainly continue to change.”
There’s a major need for new approaches in bladder cancer, Dr. Henry said. While the median survival for patients with metastatic disease treated has risen, it remains low at about 15 months. And, she said, the 5-year survival rate is about 15% with modern treatments.
Platinum-based chemotherapy is still the first-line treatment, she said, and cisplatin-based combos remain the standard. However, many patients are not eligible to take cisplatin because of factors such as reduced performance status, impaired renal function, peripheral neuropathy, hearing loss and heart failure. “Many patients have renal insufficiency and are platinum ineligible from the get-go,” Dr. Henry said.
In these patients, immune checkpoint inhibitors are an option, but research suggests they may lead to poorer survival in those with PDL1-low tumors. In 2018, the US Food and Drug Administration (FDA) advised their use as first-line treatment only in PDL1-high, cisplatin-ineligible patients or those who can’t undergo chemotherapy, she said.
As second-line therapy after platinum treatment, Dr. Henry said, several drugs targeting the PD1/PDL1 pathway are now FDA-approved with response rates at 15% to 25%; only 1 (pembrolizumab) has level 1 evidence from a phase 3 randomized clinical trial.
Single-agent chemotherapy is an option for patients who have worsened or cannot undergo treatment with immune checkpoint inhibitors. However, according to Dr. Henry, response rates are low (about 10%-15%) and there's no prospective or randomized control trial data showing a survival benefit.
What now? Targeted approaches are entering the picture. For example, fibroblast growth factor receptor inhibitors, which target a pathway that often mutates in bladder cancer. One drug, erdafitinib (Balversa), received FDA approval earlier this year based on a phase 2 trial data that showed an objective response rate (ORR) of 40%. Dr. Henry cautioned that unusual adverse effects can occur, including hyperphosphatemia (a disorder that boosts phosphate levels), ocular toxicity (which can lead to retinal detachment), and toxicity of the skin and hair.
“Patients need to be closely followed if they're starting this as a targeted drug,” Dr. Henry said.
Anti-Nectin-4 antibody drug conjugate, which targets urothelial carcinomas with uniformly high expression of the Nectin-4 cell surface marker, also is showing promise. Recent research suggests a “remarkable” ORR of 42% and nearly 8 months duration of response, she said.
Adverse effects include rash, peripheral neuropathy, and hyperglycemia. “Overall, this is thought to be a relatively well-tolerated therapy,” she said.
In terms of other advances, “we are moving closer to an era of molecular subtype-specific therapeutic strategies,” Dr. Henry said, and the National Comprehensive Cancer Network recommends early molecular testing in stage IIIB/IV urothelial cancer. “It can help facilitate treatment decisions and prevent delays in later lines of therapy, although we're still limited by development of individualized biomarker assays for specific drugs.”
Moving forward, she said, “continued research is needed to learn how to incorporate predictive molecular profiles to optimize treatment selection.”
Dr. Henry reported no relevant disclosures.
MINNEAPOLIS -- A new era of systemic treatment for bladder cancer has arrived, a US Department of Veterans Affairs (VA) hematologist/oncologist told colleagues, and more changes await on the horizon.
“After a historically long dry spell, you're seeing novel drugs and combinations under investigation,” said Elizabeth Henry, MD, of Edward Hines, Jr. VA Hospital and Loyola University Medical Center in Chicago, Illinois, in a presentation at the annual meeting of the Association of VA Hematology/Oncology. “Our treatment paradigm will almost certainly continue to change.”
There’s a major need for new approaches in bladder cancer, Dr. Henry said. While the median survival for patients with metastatic disease treated has risen, it remains low at about 15 months. And, she said, the 5-year survival rate is about 15% with modern treatments.
Platinum-based chemotherapy is still the first-line treatment, she said, and cisplatin-based combos remain the standard. However, many patients are not eligible to take cisplatin because of factors such as reduced performance status, impaired renal function, peripheral neuropathy, hearing loss and heart failure. “Many patients have renal insufficiency and are platinum ineligible from the get-go,” Dr. Henry said.
In these patients, immune checkpoint inhibitors are an option, but research suggests they may lead to poorer survival in those with PDL1-low tumors. In 2018, the US Food and Drug Administration (FDA) advised their use as first-line treatment only in PDL1-high, cisplatin-ineligible patients or those who can’t undergo chemotherapy, she said.
As second-line therapy after platinum treatment, Dr. Henry said, several drugs targeting the PD1/PDL1 pathway are now FDA-approved with response rates at 15% to 25%; only 1 (pembrolizumab) has level 1 evidence from a phase 3 randomized clinical trial.
Single-agent chemotherapy is an option for patients who have worsened or cannot undergo treatment with immune checkpoint inhibitors. However, according to Dr. Henry, response rates are low (about 10%-15%) and there's no prospective or randomized control trial data showing a survival benefit.
What now? Targeted approaches are entering the picture. For example, fibroblast growth factor receptor inhibitors, which target a pathway that often mutates in bladder cancer. One drug, erdafitinib (Balversa), received FDA approval earlier this year based on a phase 2 trial data that showed an objective response rate (ORR) of 40%. Dr. Henry cautioned that unusual adverse effects can occur, including hyperphosphatemia (a disorder that boosts phosphate levels), ocular toxicity (which can lead to retinal detachment), and toxicity of the skin and hair.
“Patients need to be closely followed if they're starting this as a targeted drug,” Dr. Henry said.
Anti-Nectin-4 antibody drug conjugate, which targets urothelial carcinomas with uniformly high expression of the Nectin-4 cell surface marker, also is showing promise. Recent research suggests a “remarkable” ORR of 42% and nearly 8 months duration of response, she said.
Adverse effects include rash, peripheral neuropathy, and hyperglycemia. “Overall, this is thought to be a relatively well-tolerated therapy,” she said.
In terms of other advances, “we are moving closer to an era of molecular subtype-specific therapeutic strategies,” Dr. Henry said, and the National Comprehensive Cancer Network recommends early molecular testing in stage IIIB/IV urothelial cancer. “It can help facilitate treatment decisions and prevent delays in later lines of therapy, although we're still limited by development of individualized biomarker assays for specific drugs.”
Moving forward, she said, “continued research is needed to learn how to incorporate predictive molecular profiles to optimize treatment selection.”
Dr. Henry reported no relevant disclosures.
Nerves, Neuropeptides, and the Nervous System in the Pathogenesis of Psoriasis
Background
Psoriasis is a complex, multifactorial, systemic disease that is associated with numerous neurologic comorbidities, including stroke, multiple sclerosis, epilepsy, migraine, restless leg syndrome, Parkinson disease, and less frequently Guillain-Barré syndrome and myasthenia gravis. Anxiety and depression also are frequently seen in patients with psoriasis.1 In recent years, heightened understanding of the pathogenesis and disease mechanisms involved in psoriasis has led to the development of therapies designed to help to control the chronic inflammation associated with the disease, such as immunobiologics and small molecules.2
Although tremendous effort has gone into elucidating the immunologic underpinnings of psoriasis (certainly a worthwhile endeavor), less attention has been given to the role the nervous system plays in its pathogenesis.3,4 Nonetheless, clinical evidence suggests that the nervous system plays an important role in the pathophysiology of psoriasis and is deserving of further investigation.3
Nerves and Neuropeptides
Psychological stress is known to exacerbate psoriasis, which points to the involvement of the nervous system in psoriasis.3,5,6 In addition to provoking the sympathetic response, psychological stressors have been shown to affect the peripheral nervous system in psoriasis by modulating the skin’s network of nerves and neuropeptides.6-11 A small study divided patients with psoriasis into low-stress and high-stress groups based on their clinical examinations and answers to questionnaires. Immunohistochemical analysis showed patients in the high-stress group had elevated levels of calcitonin gene-related peptide and vasoactive intestinal polypeptide as well as reduced levels of the neuropeptide-degrading enzyme chymase compared to the low-stress group.12 Two later studies showed calcitonin gene-related peptide stimulates keratinocyte proliferation3,13 and is found at increased levels in psoriatic skin.3,14 Similarly, higher quantities of vasoactive intestinal peptide-positive nerve fibers in the epidermis and dermis are found in psoriatic plaques compared to nonlesional and normal skin.3,15
Early research suggested that substance P (SP) released from cutaneous nerve fibers causes a local neurogenic response that elicits psoriasis in predisposed individuals.16 However, there have been conflicting reports of both higher and lower levels of SP in involved and noninvolved skin in patients with psoriasis compared with healthy individuals, making the role of SP in psoriasis ambiguous.3,15,17
Nerve growth factor (NGF), a principal mediator of neurogenic inflammation, also is suspected of playing a role in the pathogenesis of psoriasis.3,6 Studies have shown NGF prevents apoptosis of keratinocytes, activates T cells, and is found in higher levels in psoriatic skin compared to controls.3,18,19
Neuropeptides also may play a contributory role in the itching and Köbner phenomenon that are seen with psoriasis.3 The Köbner phenomenon refers to the formation of psoriatic lesions in uninvolved skin of patients with psoriasis following cutaneous trauma.20 Increased levels of NGF in nonlesional skin of patients with psoriasis are believed to contribute to the development of psoriatic plaques following trauma by triggering an inflammatory response that upregulates other neuropeptides, such as SP and calcitonin gene-related peptide.3 These neuropeptides generate keratinocyte proliferation, which in turn further increase NGF expression; as such, a cycle of inflammation and formation of psoriatic lesions is engendered.3,5 A noteworthy correlation also has been shown between the severity of pruritus and density of NGF-immunoreactive keratinocytes, high-affinity NGF receptors, protein gene product 9.5–immunoreactive intraepidermal fibers, and immunoreactive vessels for E-selectin.3,21
Spontaneous Clearing of Psoriasis
Spontaneous remission of psoriasis after cerebrovascular accident was first described in a case report published in 1998.22 Other cases have reported protective effects from psoriasis and psoriatic arthritis in limbs affected by poliomyelitis.23,24 Conversely, recurrence of skin lesions in areas corresponding to nervous system injury also have been reported in cases in which patients regained neurologic function; when permanent nerve damage was sustained, psoriasis did not recur,4 which confirms that peripheral nerves play a role in the pathogenesis of psoriasis.3 It is believed that peripheral nerve damage leads to reduced secretion of neuropeptides, and central nervous system injury can propagate similar downstream effects.3,25

Reports of psoriasis remission in the wake of peripheral and central nervous system injury from surgical nerve resection as well as cerebrovascular accident, as documented in the case presented here, provide clinical evidence in support of the neurocutaneous pathway’s role in psoriasis.3,4 Several reports have described clinical improvement of psoriasis following sensory cutaneous nerve damage, suggesting inflammation of the cutaneous nerves may be involved in the pathogenesis of psoriasis.3,6 Clearance of psoriatic plaques at the site of injury occurred following nerve resection; after reinnervation of the affected areas, disease recurrence occurred.6,26-28 More recently, cutaneous denervation was shown to improve acanthosis and IL-23 expression in mice with psoriasiform skin.3,25 Intradermal injections of calcitonin gene-related peptide and/or a SP agonist into the denervated areas reversed this denervation-mediated improvement.3,25
Bottom Line
This case report describes spontaneous clearing of psoriasis following a cerebrovascular accident. Improvement in psoriasis in the absence of neural inputs suggest the nervous system plays a crucial role in the development of psoriatic disease.4 A better understanding of the neuropeptides involved in the neurologic-mediated clearance of psoriasis may contribute to the development of improved targeted therapies, specifically designed to target the neurologic aspects of psoriasis.3 Neuropeptides such as nerve growth factor, calcitonin gene-related peptide, and vasoactive intestinal peptide, and possibly SP may play an important role in the pathogenesis of psoriasis and may one day be ideal targets for novel therapies.
- Amanat M, Salehi M, Rezaei N. Neurological and psychiatric disorders in psoriasis. Rev Neurosci. 2018;29:805-813.
- Eberle FC, Brück J, Holstein J, et al. Recent advances in understanding psoriasis [published April 28, 2016]. F1000Res. doi:10.12688/f1000research.7927.1.
- Lee EB, Reynolds KA, Pithadia DJ, et al. Clearance of psoriasis after ischemic stroke. Cutis. 2019;103:74-76.
- Zhu TH, Nakamura M, Farahnik B, et al. The role of the nervous system in the pathophysiology of psoriasis: a review of cases of psoriasis remission or improvement following denervation injury. Am J Clin Dermatol. 2016;17:257-263.
- Raychaudhuri SP, Farber EM. Neuroimmunologic aspects of psoriasis. Cutis. 2000;66:357-362.
- Kwon CW, Fried RG, Nousari Y, et al. Psoriasis: psychosomatic, somatopsychic, or both? Clin Dermatol. 2018;36:698-703.
- Lotti T, D’Erme AM, Hercogová J. The role of neuropeptides in the control of regional immunity. Clin Dermatol. 2014;32:633-645.
- Hall JM, Cruser D, Podawiltz A, et al. Psychological stress and the cutaneous immune response: roles of the HPA axis and the sympathetic nervous system in atopic dermatitis and psoriasis [published online August 30, 2012]. Dermatol Res Pract. 2012;2012:403908.
- Raychaudhuri SK, Raychaudhuri SP. NGF and its receptor system: a new dimension in the pathogenesis of psoriasis and psoriatic arthritis. Ann N Y Acad Sci. 2009;1173:470-477.
- Glaser R, Kiecolt-Glaser JK. Stress-induced immune dysfunction: implications for health. Nat Rev Immunol. 2005;5:243-251.
- Levi-Montalcini R, Skaper SD, Dal Toso R, et al. Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci. 1996;19:514-520.
- Harvima IT, Viinamäki H, Naukkarinen A, et al. Association of cutaneous mast cells and sensory nerves with psychic stress in psoriasis. Psychother Psychosom. 1993;60:168-176.
- He Y, Ding G, Wang X, et al. Calcitonin gene‐related peptide in Langerhans cells in psoriatic plaque lesions. Chin Med J (Engl). 2000;113:747-751.
- Chu DQ, Choy M, Foster P, et al. A comparative study of the ability of calcitonin gene‐related peptide and adrenomedullin13–52 to modulate microvascular but not thermal hyperalgesia responses. Br J Pharmacol. 2000;130:1589-1596.
- Al’Abadie MS, Senior HJ, Bleehen SS, et al. Neuropeptides and general neuronal marker in psoriasis—an immunohistochemical study. Clin Exp Dermatol. 1995;20:384-389.
- Farber EM, Nickoloff BJ, Recht B, et al. Stress, symmetry, and psoriasis: possible role of neuropeptides. J Am Acad Dermatol. 1986;14(2, pt 1):305-311.
- Pincelli C, Fantini F, Romualdi P, et al. Substance P is diminished and vasoactive intestinal peptide is augmented in psoriatic lesions and these peptides exert disparate effects on the proliferation of cultured human keratinocytes. J Invest Dermatol. 1992;98:421-427.
- Raychaudhuri SP, Jiang WY, Farber EM. Psoriatic keratinocytes express high levels of nerve growth factor. Acta Derm Venereol. 1998;78:84-86.
- Pincelli C. Nerve growth factor and keratinocytes: a role in psoriasis. Eur J Dermatol. 2000;10:85-90.
- Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
- Nakamura M, Toyoda M, Morohashi M. Pruritogenic mediators in psoriasis vulgaris: comparative evaluation of itch-associated cutaneous factors. Br J Dermatol. 2003;149:718-730.
- Stratigos AJ, Katoulis AK, Stavrianeas NG. Spontaneous clearing of psoriasis after stroke. J Am Acad Dermatol. 1998;38(5, pt 1):768-770.
- Wang TS, Tsai TF. Psoriasis sparing the lower limb with postpoliomyelitis residual paralysis. Br J Dermatol. 2014;171:429-431.
- Weiner SR, Bassett LW, Reichman RP. Protective effect of poliomyelitis on psoriatic arthritis. Arthritis Rheum. 1985;28:703-706.
- Ostrowski SM, Belkai A, Loyd CM, et al. Cutaneous denervation of psoriasiform mouse skin improves acanthosis and inflammation in a sensory neuropeptide-dependent manner. J Invest Dermatol. 2011;131:1530-1538.
- Farber EM, Lanigan SW, Boer J. The role of cutaneous sensory nerves in the maintenance of psoriasis. Int J Dermatol. 1990;29:418-420.
- Dewing SB. Remission of psoriasis associated with cutaneous nerve section. Arch Dermatol. 1971;104:220-221.
- Perlman HH. Remission of psoriasis vulgaris from the use of nerve-blocking agents. Arch Dermatol. 1972;105:128-129.
Background
Psoriasis is a complex, multifactorial, systemic disease that is associated with numerous neurologic comorbidities, including stroke, multiple sclerosis, epilepsy, migraine, restless leg syndrome, Parkinson disease, and less frequently Guillain-Barré syndrome and myasthenia gravis. Anxiety and depression also are frequently seen in patients with psoriasis.1 In recent years, heightened understanding of the pathogenesis and disease mechanisms involved in psoriasis has led to the development of therapies designed to help to control the chronic inflammation associated with the disease, such as immunobiologics and small molecules.2

Although tremendous effort has gone into elucidating the immunologic underpinnings of psoriasis (certainly a worthwhile endeavor), less attention has been given to the role the nervous system plays in its pathogenesis.3,4 Nonetheless, clinical evidence suggests that the nervous system plays an important role in the pathophysiology of psoriasis and is deserving of further investigation.3
Nerves and Neuropeptides
Psychological stress is known to exacerbate psoriasis, which points to the involvement of the nervous system in psoriasis.3,5,6 In addition to provoking the sympathetic response, psychological stressors have been shown to affect the peripheral nervous system in psoriasis by modulating the skin’s network of nerves and neuropeptides.6-11 A small study divided patients with psoriasis into low-stress and high-stress groups based on their clinical examinations and answers to questionnaires. Immunohistochemical analysis showed patients in the high-stress group had elevated levels of calcitonin gene-related peptide and vasoactive intestinal polypeptide as well as reduced levels of the neuropeptide-degrading enzyme chymase compared to the low-stress group.12 Two later studies showed calcitonin gene-related peptide stimulates keratinocyte proliferation3,13 and is found at increased levels in psoriatic skin.3,14 Similarly, higher quantities of vasoactive intestinal peptide-positive nerve fibers in the epidermis and dermis are found in psoriatic plaques compared to nonlesional and normal skin.3,15

Early research suggested that substance P (SP) released from cutaneous nerve fibers causes a local neurogenic response that elicits psoriasis in predisposed individuals.16 However, there have been conflicting reports of both higher and lower levels of SP in involved and noninvolved skin in patients with psoriasis compared with healthy individuals, making the role of SP in psoriasis ambiguous.3,15,17
Nerve growth factor (NGF), a principal mediator of neurogenic inflammation, also is suspected of playing a role in the pathogenesis of psoriasis.3,6 Studies have shown NGF prevents apoptosis of keratinocytes, activates T cells, and is found in higher levels in psoriatic skin compared to controls.3,18,19

Neuropeptides also may play a contributory role in the itching and Köbner phenomenon that are seen with psoriasis.3 The Köbner phenomenon refers to the formation of psoriatic lesions in uninvolved skin of patients with psoriasis following cutaneous trauma.20 Increased levels of NGF in nonlesional skin of patients with psoriasis are believed to contribute to the development of psoriatic plaques following trauma by triggering an inflammatory response that upregulates other neuropeptides, such as SP and calcitonin gene-related peptide.3 These neuropeptides generate keratinocyte proliferation, which in turn further increase NGF expression; as such, a cycle of inflammation and formation of psoriatic lesions is engendered.3,5 A noteworthy correlation also has been shown between the severity of pruritus and density of NGF-immunoreactive keratinocytes, high-affinity NGF receptors, protein gene product 9.5–immunoreactive intraepidermal fibers, and immunoreactive vessels for E-selectin.3,21
Spontaneous Clearing of Psoriasis
Spontaneous remission of psoriasis after cerebrovascular accident was first described in a case report published in 1998.22 Other cases have reported protective effects from psoriasis and psoriatic arthritis in limbs affected by poliomyelitis.23,24 Conversely, recurrence of skin lesions in areas corresponding to nervous system injury also have been reported in cases in which patients regained neurologic function; when permanent nerve damage was sustained, psoriasis did not recur,4 which confirms that peripheral nerves play a role in the pathogenesis of psoriasis.3 It is believed that peripheral nerve damage leads to reduced secretion of neuropeptides, and central nervous system injury can propagate similar downstream effects.3,25

Reports of psoriasis remission in the wake of peripheral and central nervous system injury from surgical nerve resection as well as cerebrovascular accident, as documented in the case presented here, provide clinical evidence in support of the neurocutaneous pathway’s role in psoriasis.3,4 Several reports have described clinical improvement of psoriasis following sensory cutaneous nerve damage, suggesting inflammation of the cutaneous nerves may be involved in the pathogenesis of psoriasis.3,6 Clearance of psoriatic plaques at the site of injury occurred following nerve resection; after reinnervation of the affected areas, disease recurrence occurred.6,26-28 More recently, cutaneous denervation was shown to improve acanthosis and IL-23 expression in mice with psoriasiform skin.3,25 Intradermal injections of calcitonin gene-related peptide and/or a SP agonist into the denervated areas reversed this denervation-mediated improvement.3,25
Bottom Line
This case report describes spontaneous clearing of psoriasis following a cerebrovascular accident. Improvement in psoriasis in the absence of neural inputs suggest the nervous system plays a crucial role in the development of psoriatic disease.4 A better understanding of the neuropeptides involved in the neurologic-mediated clearance of psoriasis may contribute to the development of improved targeted therapies, specifically designed to target the neurologic aspects of psoriasis.3 Neuropeptides such as nerve growth factor, calcitonin gene-related peptide, and vasoactive intestinal peptide, and possibly SP may play an important role in the pathogenesis of psoriasis and may one day be ideal targets for novel therapies.
Background
Psoriasis is a complex, multifactorial, systemic disease that is associated with numerous neurologic comorbidities, including stroke, multiple sclerosis, epilepsy, migraine, restless leg syndrome, Parkinson disease, and less frequently Guillain-Barré syndrome and myasthenia gravis. Anxiety and depression also are frequently seen in patients with psoriasis.1 In recent years, heightened understanding of the pathogenesis and disease mechanisms involved in psoriasis has led to the development of therapies designed to help to control the chronic inflammation associated with the disease, such as immunobiologics and small molecules.2

Although tremendous effort has gone into elucidating the immunologic underpinnings of psoriasis (certainly a worthwhile endeavor), less attention has been given to the role the nervous system plays in its pathogenesis.3,4 Nonetheless, clinical evidence suggests that the nervous system plays an important role in the pathophysiology of psoriasis and is deserving of further investigation.3
Nerves and Neuropeptides
Psychological stress is known to exacerbate psoriasis, which points to the involvement of the nervous system in psoriasis.3,5,6 In addition to provoking the sympathetic response, psychological stressors have been shown to affect the peripheral nervous system in psoriasis by modulating the skin’s network of nerves and neuropeptides.6-11 A small study divided patients with psoriasis into low-stress and high-stress groups based on their clinical examinations and answers to questionnaires. Immunohistochemical analysis showed patients in the high-stress group had elevated levels of calcitonin gene-related peptide and vasoactive intestinal polypeptide as well as reduced levels of the neuropeptide-degrading enzyme chymase compared to the low-stress group.12 Two later studies showed calcitonin gene-related peptide stimulates keratinocyte proliferation3,13 and is found at increased levels in psoriatic skin.3,14 Similarly, higher quantities of vasoactive intestinal peptide-positive nerve fibers in the epidermis and dermis are found in psoriatic plaques compared to nonlesional and normal skin.3,15

Early research suggested that substance P (SP) released from cutaneous nerve fibers causes a local neurogenic response that elicits psoriasis in predisposed individuals.16 However, there have been conflicting reports of both higher and lower levels of SP in involved and noninvolved skin in patients with psoriasis compared with healthy individuals, making the role of SP in psoriasis ambiguous.3,15,17
Nerve growth factor (NGF), a principal mediator of neurogenic inflammation, also is suspected of playing a role in the pathogenesis of psoriasis.3,6 Studies have shown NGF prevents apoptosis of keratinocytes, activates T cells, and is found in higher levels in psoriatic skin compared to controls.3,18,19

Neuropeptides also may play a contributory role in the itching and Köbner phenomenon that are seen with psoriasis.3 The Köbner phenomenon refers to the formation of psoriatic lesions in uninvolved skin of patients with psoriasis following cutaneous trauma.20 Increased levels of NGF in nonlesional skin of patients with psoriasis are believed to contribute to the development of psoriatic plaques following trauma by triggering an inflammatory response that upregulates other neuropeptides, such as SP and calcitonin gene-related peptide.3 These neuropeptides generate keratinocyte proliferation, which in turn further increase NGF expression; as such, a cycle of inflammation and formation of psoriatic lesions is engendered.3,5 A noteworthy correlation also has been shown between the severity of pruritus and density of NGF-immunoreactive keratinocytes, high-affinity NGF receptors, protein gene product 9.5–immunoreactive intraepidermal fibers, and immunoreactive vessels for E-selectin.3,21
Spontaneous Clearing of Psoriasis
Spontaneous remission of psoriasis after cerebrovascular accident was first described in a case report published in 1998.22 Other cases have reported protective effects from psoriasis and psoriatic arthritis in limbs affected by poliomyelitis.23,24 Conversely, recurrence of skin lesions in areas corresponding to nervous system injury also have been reported in cases in which patients regained neurologic function; when permanent nerve damage was sustained, psoriasis did not recur,4 which confirms that peripheral nerves play a role in the pathogenesis of psoriasis.3 It is believed that peripheral nerve damage leads to reduced secretion of neuropeptides, and central nervous system injury can propagate similar downstream effects.3,25

Reports of psoriasis remission in the wake of peripheral and central nervous system injury from surgical nerve resection as well as cerebrovascular accident, as documented in the case presented here, provide clinical evidence in support of the neurocutaneous pathway’s role in psoriasis.3,4 Several reports have described clinical improvement of psoriasis following sensory cutaneous nerve damage, suggesting inflammation of the cutaneous nerves may be involved in the pathogenesis of psoriasis.3,6 Clearance of psoriatic plaques at the site of injury occurred following nerve resection; after reinnervation of the affected areas, disease recurrence occurred.6,26-28 More recently, cutaneous denervation was shown to improve acanthosis and IL-23 expression in mice with psoriasiform skin.3,25 Intradermal injections of calcitonin gene-related peptide and/or a SP agonist into the denervated areas reversed this denervation-mediated improvement.3,25
Bottom Line
This case report describes spontaneous clearing of psoriasis following a cerebrovascular accident. Improvement in psoriasis in the absence of neural inputs suggest the nervous system plays a crucial role in the development of psoriatic disease.4 A better understanding of the neuropeptides involved in the neurologic-mediated clearance of psoriasis may contribute to the development of improved targeted therapies, specifically designed to target the neurologic aspects of psoriasis.3 Neuropeptides such as nerve growth factor, calcitonin gene-related peptide, and vasoactive intestinal peptide, and possibly SP may play an important role in the pathogenesis of psoriasis and may one day be ideal targets for novel therapies.
- Amanat M, Salehi M, Rezaei N. Neurological and psychiatric disorders in psoriasis. Rev Neurosci. 2018;29:805-813.
- Eberle FC, Brück J, Holstein J, et al. Recent advances in understanding psoriasis [published April 28, 2016]. F1000Res. doi:10.12688/f1000research.7927.1.
- Lee EB, Reynolds KA, Pithadia DJ, et al. Clearance of psoriasis after ischemic stroke. Cutis. 2019;103:74-76.
- Zhu TH, Nakamura M, Farahnik B, et al. The role of the nervous system in the pathophysiology of psoriasis: a review of cases of psoriasis remission or improvement following denervation injury. Am J Clin Dermatol. 2016;17:257-263.
- Raychaudhuri SP, Farber EM. Neuroimmunologic aspects of psoriasis. Cutis. 2000;66:357-362.
- Kwon CW, Fried RG, Nousari Y, et al. Psoriasis: psychosomatic, somatopsychic, or both? Clin Dermatol. 2018;36:698-703.
- Lotti T, D’Erme AM, Hercogová J. The role of neuropeptides in the control of regional immunity. Clin Dermatol. 2014;32:633-645.
- Hall JM, Cruser D, Podawiltz A, et al. Psychological stress and the cutaneous immune response: roles of the HPA axis and the sympathetic nervous system in atopic dermatitis and psoriasis [published online August 30, 2012]. Dermatol Res Pract. 2012;2012:403908.
- Raychaudhuri SK, Raychaudhuri SP. NGF and its receptor system: a new dimension in the pathogenesis of psoriasis and psoriatic arthritis. Ann N Y Acad Sci. 2009;1173:470-477.
- Glaser R, Kiecolt-Glaser JK. Stress-induced immune dysfunction: implications for health. Nat Rev Immunol. 2005;5:243-251.
- Levi-Montalcini R, Skaper SD, Dal Toso R, et al. Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci. 1996;19:514-520.
- Harvima IT, Viinamäki H, Naukkarinen A, et al. Association of cutaneous mast cells and sensory nerves with psychic stress in psoriasis. Psychother Psychosom. 1993;60:168-176.
- He Y, Ding G, Wang X, et al. Calcitonin gene‐related peptide in Langerhans cells in psoriatic plaque lesions. Chin Med J (Engl). 2000;113:747-751.
- Chu DQ, Choy M, Foster P, et al. A comparative study of the ability of calcitonin gene‐related peptide and adrenomedullin13–52 to modulate microvascular but not thermal hyperalgesia responses. Br J Pharmacol. 2000;130:1589-1596.
- Al’Abadie MS, Senior HJ, Bleehen SS, et al. Neuropeptides and general neuronal marker in psoriasis—an immunohistochemical study. Clin Exp Dermatol. 1995;20:384-389.
- Farber EM, Nickoloff BJ, Recht B, et al. Stress, symmetry, and psoriasis: possible role of neuropeptides. J Am Acad Dermatol. 1986;14(2, pt 1):305-311.
- Pincelli C, Fantini F, Romualdi P, et al. Substance P is diminished and vasoactive intestinal peptide is augmented in psoriatic lesions and these peptides exert disparate effects on the proliferation of cultured human keratinocytes. J Invest Dermatol. 1992;98:421-427.
- Raychaudhuri SP, Jiang WY, Farber EM. Psoriatic keratinocytes express high levels of nerve growth factor. Acta Derm Venereol. 1998;78:84-86.
- Pincelli C. Nerve growth factor and keratinocytes: a role in psoriasis. Eur J Dermatol. 2000;10:85-90.
- Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
- Nakamura M, Toyoda M, Morohashi M. Pruritogenic mediators in psoriasis vulgaris: comparative evaluation of itch-associated cutaneous factors. Br J Dermatol. 2003;149:718-730.
- Stratigos AJ, Katoulis AK, Stavrianeas NG. Spontaneous clearing of psoriasis after stroke. J Am Acad Dermatol. 1998;38(5, pt 1):768-770.
- Wang TS, Tsai TF. Psoriasis sparing the lower limb with postpoliomyelitis residual paralysis. Br J Dermatol. 2014;171:429-431.
- Weiner SR, Bassett LW, Reichman RP. Protective effect of poliomyelitis on psoriatic arthritis. Arthritis Rheum. 1985;28:703-706.
- Ostrowski SM, Belkai A, Loyd CM, et al. Cutaneous denervation of psoriasiform mouse skin improves acanthosis and inflammation in a sensory neuropeptide-dependent manner. J Invest Dermatol. 2011;131:1530-1538.
- Farber EM, Lanigan SW, Boer J. The role of cutaneous sensory nerves in the maintenance of psoriasis. Int J Dermatol. 1990;29:418-420.
- Dewing SB. Remission of psoriasis associated with cutaneous nerve section. Arch Dermatol. 1971;104:220-221.
- Perlman HH. Remission of psoriasis vulgaris from the use of nerve-blocking agents. Arch Dermatol. 1972;105:128-129.
- Amanat M, Salehi M, Rezaei N. Neurological and psychiatric disorders in psoriasis. Rev Neurosci. 2018;29:805-813.
- Eberle FC, Brück J, Holstein J, et al. Recent advances in understanding psoriasis [published April 28, 2016]. F1000Res. doi:10.12688/f1000research.7927.1.
- Lee EB, Reynolds KA, Pithadia DJ, et al. Clearance of psoriasis after ischemic stroke. Cutis. 2019;103:74-76.
- Zhu TH, Nakamura M, Farahnik B, et al. The role of the nervous system in the pathophysiology of psoriasis: a review of cases of psoriasis remission or improvement following denervation injury. Am J Clin Dermatol. 2016;17:257-263.
- Raychaudhuri SP, Farber EM. Neuroimmunologic aspects of psoriasis. Cutis. 2000;66:357-362.
- Kwon CW, Fried RG, Nousari Y, et al. Psoriasis: psychosomatic, somatopsychic, or both? Clin Dermatol. 2018;36:698-703.
- Lotti T, D’Erme AM, Hercogová J. The role of neuropeptides in the control of regional immunity. Clin Dermatol. 2014;32:633-645.
- Hall JM, Cruser D, Podawiltz A, et al. Psychological stress and the cutaneous immune response: roles of the HPA axis and the sympathetic nervous system in atopic dermatitis and psoriasis [published online August 30, 2012]. Dermatol Res Pract. 2012;2012:403908.
- Raychaudhuri SK, Raychaudhuri SP. NGF and its receptor system: a new dimension in the pathogenesis of psoriasis and psoriatic arthritis. Ann N Y Acad Sci. 2009;1173:470-477.
- Glaser R, Kiecolt-Glaser JK. Stress-induced immune dysfunction: implications for health. Nat Rev Immunol. 2005;5:243-251.
- Levi-Montalcini R, Skaper SD, Dal Toso R, et al. Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci. 1996;19:514-520.
- Harvima IT, Viinamäki H, Naukkarinen A, et al. Association of cutaneous mast cells and sensory nerves with psychic stress in psoriasis. Psychother Psychosom. 1993;60:168-176.
- He Y, Ding G, Wang X, et al. Calcitonin gene‐related peptide in Langerhans cells in psoriatic plaque lesions. Chin Med J (Engl). 2000;113:747-751.
- Chu DQ, Choy M, Foster P, et al. A comparative study of the ability of calcitonin gene‐related peptide and adrenomedullin13–52 to modulate microvascular but not thermal hyperalgesia responses. Br J Pharmacol. 2000;130:1589-1596.
- Al’Abadie MS, Senior HJ, Bleehen SS, et al. Neuropeptides and general neuronal marker in psoriasis—an immunohistochemical study. Clin Exp Dermatol. 1995;20:384-389.
- Farber EM, Nickoloff BJ, Recht B, et al. Stress, symmetry, and psoriasis: possible role of neuropeptides. J Am Acad Dermatol. 1986;14(2, pt 1):305-311.
- Pincelli C, Fantini F, Romualdi P, et al. Substance P is diminished and vasoactive intestinal peptide is augmented in psoriatic lesions and these peptides exert disparate effects on the proliferation of cultured human keratinocytes. J Invest Dermatol. 1992;98:421-427.
- Raychaudhuri SP, Jiang WY, Farber EM. Psoriatic keratinocytes express high levels of nerve growth factor. Acta Derm Venereol. 1998;78:84-86.
- Pincelli C. Nerve growth factor and keratinocytes: a role in psoriasis. Eur J Dermatol. 2000;10:85-90.
- Sagi L, Trau H. The Koebner phenomenon. Clin Dermatol. 2011;29:231-236.
- Nakamura M, Toyoda M, Morohashi M. Pruritogenic mediators in psoriasis vulgaris: comparative evaluation of itch-associated cutaneous factors. Br J Dermatol. 2003;149:718-730.
- Stratigos AJ, Katoulis AK, Stavrianeas NG. Spontaneous clearing of psoriasis after stroke. J Am Acad Dermatol. 1998;38(5, pt 1):768-770.
- Wang TS, Tsai TF. Psoriasis sparing the lower limb with postpoliomyelitis residual paralysis. Br J Dermatol. 2014;171:429-431.
- Weiner SR, Bassett LW, Reichman RP. Protective effect of poliomyelitis on psoriatic arthritis. Arthritis Rheum. 1985;28:703-706.
- Ostrowski SM, Belkai A, Loyd CM, et al. Cutaneous denervation of psoriasiform mouse skin improves acanthosis and inflammation in a sensory neuropeptide-dependent manner. J Invest Dermatol. 2011;131:1530-1538.
- Farber EM, Lanigan SW, Boer J. The role of cutaneous sensory nerves in the maintenance of psoriasis. Int J Dermatol. 1990;29:418-420.
- Dewing SB. Remission of psoriasis associated with cutaneous nerve section. Arch Dermatol. 1971;104:220-221.
- Perlman HH. Remission of psoriasis vulgaris from the use of nerve-blocking agents. Arch Dermatol. 1972;105:128-129.
The Case
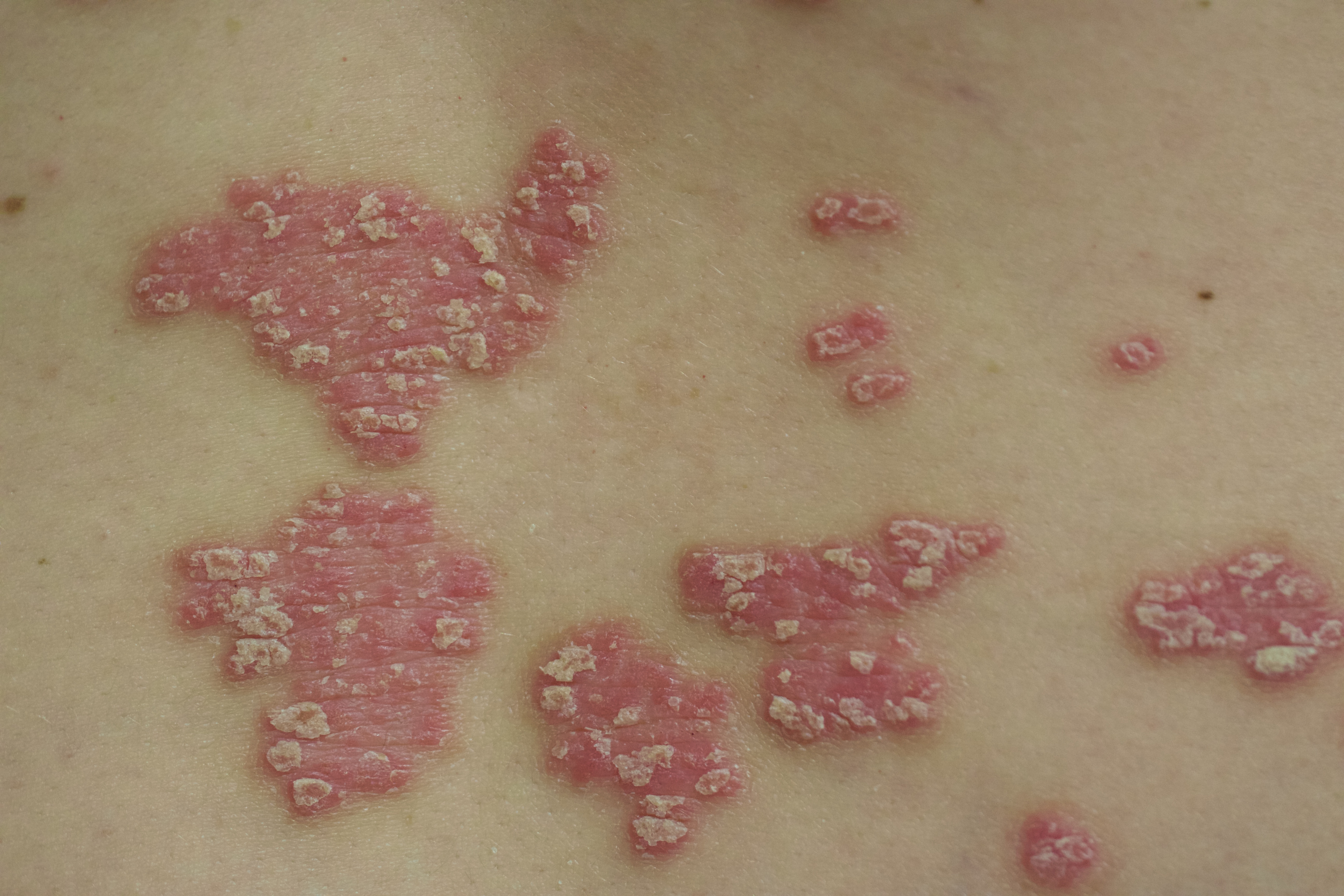
A 52-year-old man with psoriasis presented to the dermatology clinic for follow-up. The patient had been using topical clobetasol and apremilast with limited success; however, he had not yet tried biologic therapy. Physical examination revealed erythematous, scaly, indurated papules and plaques on the chest, abdomen, back, arms, and legs, consistent with psoriasis. Affected body surface area was approximately 10%.
Treatment
Ustekinumab was prescribed, but the patient did not pick it up from the pharmacy.
Approximately 1 month later, the patient presented to the emergency department with left-sided weakness and numbness. He was subsequently hospitalized for treatment of stroke. During hospitalization, the patient was started on lisinopril, aspirin, and atorvastatin. He also was given subcu-taneous enoxaparin with plans to initiate warfarin as an outpatient. No therapies for the treatment of psoriasis were given during his admission. Three days after being admitted, he was discharged to a skilled nursing facility.
Patient Outcome
Three months following discharge, the patient returned to the dermatology clinic for follow-up. After his stroke, he reported that his psoriasis had cleared and had not returned. Physical examination revealed his skin was clear of psoriatic lesions.
This case was adapted from Lee EB, Reynolds KA, Pithadia DJ, et al. Clearance of psoriasis after ischemic stroke. Cutis. 2019;103:74-76.
Clinical Trials Should Be Standard of Care
MINNEAPOLIS -- Clinical trials should be considered standard of care in oncology, the president of the Association of VA Hematology/Oncology (AVAHO) declared, and he urged colleagues to bypass obstacles and embrace them in patient care.
Clinical trials help many people, not just those in the studies, as they are indicators of robust oncology programs. “There’s a significant correlation between clinical trial activity [at sites] and improvement in survival in cancer,” said oncologist Mark Klein, MD, of the Minneapolis VA Health Care System and University of Minnesota, in a presentation at the 2019 annual meeting of AVAHO.
But the numbers suggest that the VA has lagged behind on the clinical trial front, Dr. Klein said. He pointed to internal statistics from 2001 to 2003, reported in 2010, which found clinical trial participation rates among men in the VA were lower (0.37%) than those of the national rates (0.74%).
Specifically, the VA and national participation rates were 1.16% and 0.30%, respectively, for colorectal cancer, 0.67% and 0.30% for lung cancer, 0.70% and 0.47% for prostate cancer, 0.52% and 0.74% for myeloma. According to Dr. Klein, lung and prostate cancer account for about half of all cancer diagnoses in the VA.
But the participation rates were much higher among the VA hospitals that took part in clinical trials, he said, at 2% for colorectal cancer, 1.4% for lung cancer, 2.5% for prostate cancer, 18.2% for myeloma and 2.07% overall. These numbers make it clear, he said, that it’s possible to improve: “We can do better.”
Dr. Klein pointed to other numbers that suggest the VA facilities that do participate in clinical trials typically do not take part in more than 1 or 2. In 2016, he said, an analysis found that open interventional trials were in progress at 82 VA facilities. About 35 facilities had little activity with only 1 trial in progress. “It has changed since then, but not dramatically.”
Meanwhile, he said, 2016 numbers also showed that about two-thirds of open interventional trials in the VA only included a single VA site. “We weren’t playing together, having consortia and working as a team.”
There are obstacles to improvement, he said, and fixes will take time. “It probably takes 5, 10-plus years,” he said.
These barriers include:
- Lack of adequate trial offering. Changing regulations is key here;
- Strict eligibility criteria. Easing the criteria and more effective screening are helpful;
- Long distances to travel to cancer clinics. Telemedicine could make a difference on this front;
- Patient concerns about being a “guinea pig.” Patient education can help;
- Provider concerns such as worry about extra work. The VA can work to ease burdens and provide incentives for trial enrollment; and
- High costs. Study sponsors may pay for trial drugs, helping facilities to lower expenses or even reach cost neutrality.
There are more solutions to boost participation, Dr. Klein said, including education, engagement and partnerships.
On the partnership front, he highlighted NAVIGATE, an interagency consortium of the VA and the National Cancer Institute. This collaboration aims to boost enrollment of VA patients in clinical trials, he said, and there are designated NAVIGATE sites across the country from coast to coast.
A partnership between AVAHO and the National Association of Veterans’ Research and Education Foundations is also boosting clinical trials in VA oncology. Thanks to this partnership, 1 trial has completed accrual, 6 multisite trials are active, and 5 are starting up, according to Dr. Klein.
In the big picture, Dr. Klein said, it’s clear that “we’ve got a lot of barriers in the VA. Anybody who works in the VA knows that. But I’ve seen this meeting grow and grow, and I’ve seen teamwork and partnerships form.”
MINNEAPOLIS -- Clinical trials should be considered standard of care in oncology, the president of the Association of VA Hematology/Oncology (AVAHO) declared, and he urged colleagues to bypass obstacles and embrace them in patient care.
Clinical trials help many people, not just those in the studies, as they are indicators of robust oncology programs. “There’s a significant correlation between clinical trial activity [at sites] and improvement in survival in cancer,” said oncologist Mark Klein, MD, of the Minneapolis VA Health Care System and University of Minnesota, in a presentation at the 2019 annual meeting of AVAHO.
But the numbers suggest that the VA has lagged behind on the clinical trial front, Dr. Klein said. He pointed to internal statistics from 2001 to 2003, reported in 2010, which found clinical trial participation rates among men in the VA were lower (0.37%) than those of the national rates (0.74%).
Specifically, the VA and national participation rates were 1.16% and 0.30%, respectively, for colorectal cancer, 0.67% and 0.30% for lung cancer, 0.70% and 0.47% for prostate cancer, 0.52% and 0.74% for myeloma. According to Dr. Klein, lung and prostate cancer account for about half of all cancer diagnoses in the VA.
But the participation rates were much higher among the VA hospitals that took part in clinical trials, he said, at 2% for colorectal cancer, 1.4% for lung cancer, 2.5% for prostate cancer, 18.2% for myeloma and 2.07% overall. These numbers make it clear, he said, that it’s possible to improve: “We can do better.”
Dr. Klein pointed to other numbers that suggest the VA facilities that do participate in clinical trials typically do not take part in more than 1 or 2. In 2016, he said, an analysis found that open interventional trials were in progress at 82 VA facilities. About 35 facilities had little activity with only 1 trial in progress. “It has changed since then, but not dramatically.”
Meanwhile, he said, 2016 numbers also showed that about two-thirds of open interventional trials in the VA only included a single VA site. “We weren’t playing together, having consortia and working as a team.”
There are obstacles to improvement, he said, and fixes will take time. “It probably takes 5, 10-plus years,” he said.
These barriers include:
- Lack of adequate trial offering. Changing regulations is key here;
- Strict eligibility criteria. Easing the criteria and more effective screening are helpful;
- Long distances to travel to cancer clinics. Telemedicine could make a difference on this front;
- Patient concerns about being a “guinea pig.” Patient education can help;
- Provider concerns such as worry about extra work. The VA can work to ease burdens and provide incentives for trial enrollment; and
- High costs. Study sponsors may pay for trial drugs, helping facilities to lower expenses or even reach cost neutrality.
There are more solutions to boost participation, Dr. Klein said, including education, engagement and partnerships.
On the partnership front, he highlighted NAVIGATE, an interagency consortium of the VA and the National Cancer Institute. This collaboration aims to boost enrollment of VA patients in clinical trials, he said, and there are designated NAVIGATE sites across the country from coast to coast.
A partnership between AVAHO and the National Association of Veterans’ Research and Education Foundations is also boosting clinical trials in VA oncology. Thanks to this partnership, 1 trial has completed accrual, 6 multisite trials are active, and 5 are starting up, according to Dr. Klein.
In the big picture, Dr. Klein said, it’s clear that “we’ve got a lot of barriers in the VA. Anybody who works in the VA knows that. But I’ve seen this meeting grow and grow, and I’ve seen teamwork and partnerships form.”
MINNEAPOLIS -- Clinical trials should be considered standard of care in oncology, the president of the Association of VA Hematology/Oncology (AVAHO) declared, and he urged colleagues to bypass obstacles and embrace them in patient care.
Clinical trials help many people, not just those in the studies, as they are indicators of robust oncology programs. “There’s a significant correlation between clinical trial activity [at sites] and improvement in survival in cancer,” said oncologist Mark Klein, MD, of the Minneapolis VA Health Care System and University of Minnesota, in a presentation at the 2019 annual meeting of AVAHO.
But the numbers suggest that the VA has lagged behind on the clinical trial front, Dr. Klein said. He pointed to internal statistics from 2001 to 2003, reported in 2010, which found clinical trial participation rates among men in the VA were lower (0.37%) than those of the national rates (0.74%).
Specifically, the VA and national participation rates were 1.16% and 0.30%, respectively, for colorectal cancer, 0.67% and 0.30% for lung cancer, 0.70% and 0.47% for prostate cancer, 0.52% and 0.74% for myeloma. According to Dr. Klein, lung and prostate cancer account for about half of all cancer diagnoses in the VA.
But the participation rates were much higher among the VA hospitals that took part in clinical trials, he said, at 2% for colorectal cancer, 1.4% for lung cancer, 2.5% for prostate cancer, 18.2% for myeloma and 2.07% overall. These numbers make it clear, he said, that it’s possible to improve: “We can do better.”
Dr. Klein pointed to other numbers that suggest the VA facilities that do participate in clinical trials typically do not take part in more than 1 or 2. In 2016, he said, an analysis found that open interventional trials were in progress at 82 VA facilities. About 35 facilities had little activity with only 1 trial in progress. “It has changed since then, but not dramatically.”
Meanwhile, he said, 2016 numbers also showed that about two-thirds of open interventional trials in the VA only included a single VA site. “We weren’t playing together, having consortia and working as a team.”
There are obstacles to improvement, he said, and fixes will take time. “It probably takes 5, 10-plus years,” he said.
These barriers include:
- Lack of adequate trial offering. Changing regulations is key here;
- Strict eligibility criteria. Easing the criteria and more effective screening are helpful;
- Long distances to travel to cancer clinics. Telemedicine could make a difference on this front;
- Patient concerns about being a “guinea pig.” Patient education can help;
- Provider concerns such as worry about extra work. The VA can work to ease burdens and provide incentives for trial enrollment; and
- High costs. Study sponsors may pay for trial drugs, helping facilities to lower expenses or even reach cost neutrality.
There are more solutions to boost participation, Dr. Klein said, including education, engagement and partnerships.
On the partnership front, he highlighted NAVIGATE, an interagency consortium of the VA and the National Cancer Institute. This collaboration aims to boost enrollment of VA patients in clinical trials, he said, and there are designated NAVIGATE sites across the country from coast to coast.
A partnership between AVAHO and the National Association of Veterans’ Research and Education Foundations is also boosting clinical trials in VA oncology. Thanks to this partnership, 1 trial has completed accrual, 6 multisite trials are active, and 5 are starting up, according to Dr. Klein.
In the big picture, Dr. Klein said, it’s clear that “we’ve got a lot of barriers in the VA. Anybody who works in the VA knows that. But I’ve seen this meeting grow and grow, and I’ve seen teamwork and partnerships form.”
Closure of women’s health clinics may negatively impact cervical cancer outcomes
.
States with a decreased number of women’s clinics per capita between 2010 and 2013 were found to have less screening for cervical cancer, more advanced stage of cervical cancer at presentation, and higher mortality from cervical cancer than states with no decrease in clinics, reported lead author Amar J. Srivastava, MD, of Washington University in St. Louis, who also noted that these changes occurred within a relatively short time frame.
“We know that women are generally diagnosed through the utilization of Pap smears,” Dr. Srivastava said during a presentation at the annual meeting of the American Society for Radiation Oncology. “These are low-cost tests that are available at multiple low-cost women’s health clinics. Unfortunately ... over the course of the past decade, we’ve seen a significant reduction of these clinics throughout the United States.”
“Between 2010 and 2013, which is the period of interest in this study, we know that about 100 of these women’s health clinics closed,” Dr. Srivastava said. “This was due to a combination of several factors; some of it was due to funding, some of it was due to restructuring of the clinics, and there were also laws passed throughout many states that ultimately led to the closure of many clinics.”
To determine the impact of these closures, the investigators first divided states into those that had women’s clinic closures between 2010 and 2013 and those that did not. Comparisons between these two cohorts involved the use of two databases. The first was the Behavioral Risk Factors Surveillance Study (BRFSS), which provided data from 197,143 cases, enabling assessment of differences between screening availability. The second database was the Surveillance, Epidemiology, and End Results (SEER) registry, which provided data from 10,652 patients, facilitating comparisons of stage at time of diagnosis and mortality rate.
Results were described in terms of relative differences between the two cohorts. For instance, screening rate among women with cervical cancer in states that had a decreased number of clinics was 1.63% lower than in states that did not lose clinics. This disparity was more pronounced in specific demographic subgroups, including Hispanic women (–5.82%), women aged between 21 and 34 years (–5.19%), unmarried women (–4.10%), and uninsured women (–6.88%).
“Historically, these are marginalized, underserved groups, and unfortunately, it comes as no surprise that these were the groups of women who were most dramatically hit by these changes,” Dr. Srivastava said.
Early-stage diagnosis was also significantly less common in states that had a decreased number of clinics, by a margin of 13.2%. Finally, the overall mortality rate among women with cervical cancer was 36% higher in states with clinic closures, a difference that climbed to 40% when comparing only metro residents.
Connecting the dots, Dr. Srivastava suggested that the decreased availability of screening may have led to fewer diagnoses at an early stage, which is more curable than late-stage disease, ultimately translating to a higher mortality rate. After noting that this chain of causality cannot be confirmed, owing to the retrospective nature of the study, Dr. Srivastava finished his presentation with a call to action.
“These findings should really give us some pause,” he said, “as physicians, as people who care about other people, to spend some time, try to figure out what’s going on, and try to address this disparity.”
After the presentation, Geraldine M. Jacobsen, MD, chair of radiation oncology at West Virginia University Cancer Institute, in Morgantown, W.V., echoed Dr. Srivastava’s concern.
“This study really raises broader questions,” Dr. Jacobsen said. “In the United States we’re always engaged in an ongoing dialogue about health care, health care policy, [and] health care costs. But a study like this brings to us the human face of what these dialogues mean. Policy affects people, and if we make changes in health care policy or health care legislation, we’re impacting people’s health and people’s lives.”
The investigators disclosed relationships with Phelps County Regional Medical Center, the Elsa U. Pardee Foundation, the American Society of Clinical Oncology, and ASTRO.
SOURCE: Srivastava AJ et al. ASTRO 2019, Abstract 202.
.
States with a decreased number of women’s clinics per capita between 2010 and 2013 were found to have less screening for cervical cancer, more advanced stage of cervical cancer at presentation, and higher mortality from cervical cancer than states with no decrease in clinics, reported lead author Amar J. Srivastava, MD, of Washington University in St. Louis, who also noted that these changes occurred within a relatively short time frame.
“We know that women are generally diagnosed through the utilization of Pap smears,” Dr. Srivastava said during a presentation at the annual meeting of the American Society for Radiation Oncology. “These are low-cost tests that are available at multiple low-cost women’s health clinics. Unfortunately ... over the course of the past decade, we’ve seen a significant reduction of these clinics throughout the United States.”
“Between 2010 and 2013, which is the period of interest in this study, we know that about 100 of these women’s health clinics closed,” Dr. Srivastava said. “This was due to a combination of several factors; some of it was due to funding, some of it was due to restructuring of the clinics, and there were also laws passed throughout many states that ultimately led to the closure of many clinics.”
To determine the impact of these closures, the investigators first divided states into those that had women’s clinic closures between 2010 and 2013 and those that did not. Comparisons between these two cohorts involved the use of two databases. The first was the Behavioral Risk Factors Surveillance Study (BRFSS), which provided data from 197,143 cases, enabling assessment of differences between screening availability. The second database was the Surveillance, Epidemiology, and End Results (SEER) registry, which provided data from 10,652 patients, facilitating comparisons of stage at time of diagnosis and mortality rate.
Results were described in terms of relative differences between the two cohorts. For instance, screening rate among women with cervical cancer in states that had a decreased number of clinics was 1.63% lower than in states that did not lose clinics. This disparity was more pronounced in specific demographic subgroups, including Hispanic women (–5.82%), women aged between 21 and 34 years (–5.19%), unmarried women (–4.10%), and uninsured women (–6.88%).
“Historically, these are marginalized, underserved groups, and unfortunately, it comes as no surprise that these were the groups of women who were most dramatically hit by these changes,” Dr. Srivastava said.
Early-stage diagnosis was also significantly less common in states that had a decreased number of clinics, by a margin of 13.2%. Finally, the overall mortality rate among women with cervical cancer was 36% higher in states with clinic closures, a difference that climbed to 40% when comparing only metro residents.
Connecting the dots, Dr. Srivastava suggested that the decreased availability of screening may have led to fewer diagnoses at an early stage, which is more curable than late-stage disease, ultimately translating to a higher mortality rate. After noting that this chain of causality cannot be confirmed, owing to the retrospective nature of the study, Dr. Srivastava finished his presentation with a call to action.
“These findings should really give us some pause,” he said, “as physicians, as people who care about other people, to spend some time, try to figure out what’s going on, and try to address this disparity.”
After the presentation, Geraldine M. Jacobsen, MD, chair of radiation oncology at West Virginia University Cancer Institute, in Morgantown, W.V., echoed Dr. Srivastava’s concern.
“This study really raises broader questions,” Dr. Jacobsen said. “In the United States we’re always engaged in an ongoing dialogue about health care, health care policy, [and] health care costs. But a study like this brings to us the human face of what these dialogues mean. Policy affects people, and if we make changes in health care policy or health care legislation, we’re impacting people’s health and people’s lives.”
The investigators disclosed relationships with Phelps County Regional Medical Center, the Elsa U. Pardee Foundation, the American Society of Clinical Oncology, and ASTRO.
SOURCE: Srivastava AJ et al. ASTRO 2019, Abstract 202.
.
States with a decreased number of women’s clinics per capita between 2010 and 2013 were found to have less screening for cervical cancer, more advanced stage of cervical cancer at presentation, and higher mortality from cervical cancer than states with no decrease in clinics, reported lead author Amar J. Srivastava, MD, of Washington University in St. Louis, who also noted that these changes occurred within a relatively short time frame.
“We know that women are generally diagnosed through the utilization of Pap smears,” Dr. Srivastava said during a presentation at the annual meeting of the American Society for Radiation Oncology. “These are low-cost tests that are available at multiple low-cost women’s health clinics. Unfortunately ... over the course of the past decade, we’ve seen a significant reduction of these clinics throughout the United States.”
“Between 2010 and 2013, which is the period of interest in this study, we know that about 100 of these women’s health clinics closed,” Dr. Srivastava said. “This was due to a combination of several factors; some of it was due to funding, some of it was due to restructuring of the clinics, and there were also laws passed throughout many states that ultimately led to the closure of many clinics.”
To determine the impact of these closures, the investigators first divided states into those that had women’s clinic closures between 2010 and 2013 and those that did not. Comparisons between these two cohorts involved the use of two databases. The first was the Behavioral Risk Factors Surveillance Study (BRFSS), which provided data from 197,143 cases, enabling assessment of differences between screening availability. The second database was the Surveillance, Epidemiology, and End Results (SEER) registry, which provided data from 10,652 patients, facilitating comparisons of stage at time of diagnosis and mortality rate.
Results were described in terms of relative differences between the two cohorts. For instance, screening rate among women with cervical cancer in states that had a decreased number of clinics was 1.63% lower than in states that did not lose clinics. This disparity was more pronounced in specific demographic subgroups, including Hispanic women (–5.82%), women aged between 21 and 34 years (–5.19%), unmarried women (–4.10%), and uninsured women (–6.88%).
“Historically, these are marginalized, underserved groups, and unfortunately, it comes as no surprise that these were the groups of women who were most dramatically hit by these changes,” Dr. Srivastava said.
Early-stage diagnosis was also significantly less common in states that had a decreased number of clinics, by a margin of 13.2%. Finally, the overall mortality rate among women with cervical cancer was 36% higher in states with clinic closures, a difference that climbed to 40% when comparing only metro residents.
Connecting the dots, Dr. Srivastava suggested that the decreased availability of screening may have led to fewer diagnoses at an early stage, which is more curable than late-stage disease, ultimately translating to a higher mortality rate. After noting that this chain of causality cannot be confirmed, owing to the retrospective nature of the study, Dr. Srivastava finished his presentation with a call to action.
“These findings should really give us some pause,” he said, “as physicians, as people who care about other people, to spend some time, try to figure out what’s going on, and try to address this disparity.”
After the presentation, Geraldine M. Jacobsen, MD, chair of radiation oncology at West Virginia University Cancer Institute, in Morgantown, W.V., echoed Dr. Srivastava’s concern.
“This study really raises broader questions,” Dr. Jacobsen said. “In the United States we’re always engaged in an ongoing dialogue about health care, health care policy, [and] health care costs. But a study like this brings to us the human face of what these dialogues mean. Policy affects people, and if we make changes in health care policy or health care legislation, we’re impacting people’s health and people’s lives.”
The investigators disclosed relationships with Phelps County Regional Medical Center, the Elsa U. Pardee Foundation, the American Society of Clinical Oncology, and ASTRO.
SOURCE: Srivastava AJ et al. ASTRO 2019, Abstract 202.
REPORTING FROM ASTRO 2019
Business case for interoperability remains elusive
In a recent survey issued by the Center for Connected Medicine that asked, “What is needed most to push interoperability forward in health care?” 53% of the 100 IT and business leader participants at hospitals and health systems answered “senior leadership commitment to interoperability as a top strategic priority.”
That interoperability continues to be an elusive target despite being a regulatory emphasis for more than a decade comes as no surprise.
“You have to put interoperability into the context of the challenges of being a leader in health care delivery organization or a hospital,” Robert Bart, MD, chief medical information officer of the Health Services Division at the University of Pittsburgh Medical Center, said in an interview. UPMC operates the Center for Connected Medicine in partnership with GE Healthcare and Nokia.
“For many health care delivery systems, the operational margin is extremely small,” he continued. “From a strategic perspective, they may be prioritizing things or opportunities that directly contribute to the bottom line and the financial success or even financial viability of the organization. “Interoperability certainly helps health care overall in the U.S., but whether it contributes directly to the financial bottom line of any given specific organization might vary significantly.”
But Dr. Bart said it really is not the financial incentives, a staple in the early days of the meaningful use program to spur adoption, that will get electronic health records and other health IT into a more interoperable space.
“I am not sure that financial incentives – unless they are significantly different in amounts than they currently are – are going to be the sole single reason why organizations prioritize interoperability higher,” he said.
Rather, he sees two key components that will drive interoperability.
First, he cited efforts by the Centers for Medicare & Medicaid Services to bring ownership of health data to the individual as an important driver.
“I think that is a better direction to push as opposed to financial incentives because I think that is really where we philosophically and then therefore operationally need to get to,” Dr. Bart said.
The second would take a lot more involvement from government that likely is more difficult to achieve: getting a more clear definition of health IT standards.
“We have accomplished some forward movement in interoperability, but we probably have far more road in front of us than we have left behind us as it relates to interoperability,” he said. “A fair amount of that is related to standards which, unfortunately, oftentimes are left up to interpretation. So when it comes to operationalizing these standards between different vendors or different health care systems, they sometimes don’t match up and add even more challenge to the exchange of information.”
And with industry not taking an active lead in solving some of the problems related to the standardization of data needed to drive interoperability, he suggested government should play a bigger role.
“I would like the government to take a stronger hand in helping health care define the standards so the standards are actually much more executable and there is a lot less interpretation, which I think would ease the ability for interoperability to flourish a lot more,” he said.
The move to value-based care could also provide added incentive to drive interoperability, Dr. Bart said.
Value-based care is “certainly not going to raise more barriers toward interoperability and potentially could lower some as organizations recognize that it may be a path to decreasing the costs of repeating tests that will not improve the quality of care delivered,” he said.
But overall, it would appear that getting to an interoperable point is going to be a challenge.
The report notes that “beyond tasks required by regulation or related to basic functioning (such as sharing data within their own system), interoperability still presents a challenge to organizations. Fewer than 4 in 10 report success with sharing data with other health systems, effectiveness in tapping into unstructured data, and effectiveness in reducing the cost of care.”
SOURCE: A report issued by the Center for Connected Medicine called “Improving Health Care Interoperability: Are We Making Progress?”
In a recent survey issued by the Center for Connected Medicine that asked, “What is needed most to push interoperability forward in health care?” 53% of the 100 IT and business leader participants at hospitals and health systems answered “senior leadership commitment to interoperability as a top strategic priority.”
That interoperability continues to be an elusive target despite being a regulatory emphasis for more than a decade comes as no surprise.
“You have to put interoperability into the context of the challenges of being a leader in health care delivery organization or a hospital,” Robert Bart, MD, chief medical information officer of the Health Services Division at the University of Pittsburgh Medical Center, said in an interview. UPMC operates the Center for Connected Medicine in partnership with GE Healthcare and Nokia.
“For many health care delivery systems, the operational margin is extremely small,” he continued. “From a strategic perspective, they may be prioritizing things or opportunities that directly contribute to the bottom line and the financial success or even financial viability of the organization. “Interoperability certainly helps health care overall in the U.S., but whether it contributes directly to the financial bottom line of any given specific organization might vary significantly.”
But Dr. Bart said it really is not the financial incentives, a staple in the early days of the meaningful use program to spur adoption, that will get electronic health records and other health IT into a more interoperable space.
“I am not sure that financial incentives – unless they are significantly different in amounts than they currently are – are going to be the sole single reason why organizations prioritize interoperability higher,” he said.
Rather, he sees two key components that will drive interoperability.
First, he cited efforts by the Centers for Medicare & Medicaid Services to bring ownership of health data to the individual as an important driver.
“I think that is a better direction to push as opposed to financial incentives because I think that is really where we philosophically and then therefore operationally need to get to,” Dr. Bart said.
The second would take a lot more involvement from government that likely is more difficult to achieve: getting a more clear definition of health IT standards.
“We have accomplished some forward movement in interoperability, but we probably have far more road in front of us than we have left behind us as it relates to interoperability,” he said. “A fair amount of that is related to standards which, unfortunately, oftentimes are left up to interpretation. So when it comes to operationalizing these standards between different vendors or different health care systems, they sometimes don’t match up and add even more challenge to the exchange of information.”
And with industry not taking an active lead in solving some of the problems related to the standardization of data needed to drive interoperability, he suggested government should play a bigger role.
“I would like the government to take a stronger hand in helping health care define the standards so the standards are actually much more executable and there is a lot less interpretation, which I think would ease the ability for interoperability to flourish a lot more,” he said.
The move to value-based care could also provide added incentive to drive interoperability, Dr. Bart said.
Value-based care is “certainly not going to raise more barriers toward interoperability and potentially could lower some as organizations recognize that it may be a path to decreasing the costs of repeating tests that will not improve the quality of care delivered,” he said.
But overall, it would appear that getting to an interoperable point is going to be a challenge.
The report notes that “beyond tasks required by regulation or related to basic functioning (such as sharing data within their own system), interoperability still presents a challenge to organizations. Fewer than 4 in 10 report success with sharing data with other health systems, effectiveness in tapping into unstructured data, and effectiveness in reducing the cost of care.”
SOURCE: A report issued by the Center for Connected Medicine called “Improving Health Care Interoperability: Are We Making Progress?”
In a recent survey issued by the Center for Connected Medicine that asked, “What is needed most to push interoperability forward in health care?” 53% of the 100 IT and business leader participants at hospitals and health systems answered “senior leadership commitment to interoperability as a top strategic priority.”
That interoperability continues to be an elusive target despite being a regulatory emphasis for more than a decade comes as no surprise.
“You have to put interoperability into the context of the challenges of being a leader in health care delivery organization or a hospital,” Robert Bart, MD, chief medical information officer of the Health Services Division at the University of Pittsburgh Medical Center, said in an interview. UPMC operates the Center for Connected Medicine in partnership with GE Healthcare and Nokia.
“For many health care delivery systems, the operational margin is extremely small,” he continued. “From a strategic perspective, they may be prioritizing things or opportunities that directly contribute to the bottom line and the financial success or even financial viability of the organization. “Interoperability certainly helps health care overall in the U.S., but whether it contributes directly to the financial bottom line of any given specific organization might vary significantly.”
But Dr. Bart said it really is not the financial incentives, a staple in the early days of the meaningful use program to spur adoption, that will get electronic health records and other health IT into a more interoperable space.
“I am not sure that financial incentives – unless they are significantly different in amounts than they currently are – are going to be the sole single reason why organizations prioritize interoperability higher,” he said.
Rather, he sees two key components that will drive interoperability.
First, he cited efforts by the Centers for Medicare & Medicaid Services to bring ownership of health data to the individual as an important driver.
“I think that is a better direction to push as opposed to financial incentives because I think that is really where we philosophically and then therefore operationally need to get to,” Dr. Bart said.
The second would take a lot more involvement from government that likely is more difficult to achieve: getting a more clear definition of health IT standards.
“We have accomplished some forward movement in interoperability, but we probably have far more road in front of us than we have left behind us as it relates to interoperability,” he said. “A fair amount of that is related to standards which, unfortunately, oftentimes are left up to interpretation. So when it comes to operationalizing these standards between different vendors or different health care systems, they sometimes don’t match up and add even more challenge to the exchange of information.”
And with industry not taking an active lead in solving some of the problems related to the standardization of data needed to drive interoperability, he suggested government should play a bigger role.
“I would like the government to take a stronger hand in helping health care define the standards so the standards are actually much more executable and there is a lot less interpretation, which I think would ease the ability for interoperability to flourish a lot more,” he said.
The move to value-based care could also provide added incentive to drive interoperability, Dr. Bart said.
Value-based care is “certainly not going to raise more barriers toward interoperability and potentially could lower some as organizations recognize that it may be a path to decreasing the costs of repeating tests that will not improve the quality of care delivered,” he said.
But overall, it would appear that getting to an interoperable point is going to be a challenge.
The report notes that “beyond tasks required by regulation or related to basic functioning (such as sharing data within their own system), interoperability still presents a challenge to organizations. Fewer than 4 in 10 report success with sharing data with other health systems, effectiveness in tapping into unstructured data, and effectiveness in reducing the cost of care.”
SOURCE: A report issued by the Center for Connected Medicine called “Improving Health Care Interoperability: Are We Making Progress?”
Judicious EEG use identifies pseudosyncope during tilt-table testing
NEW ORLEANS – , according to an investigation from Vanderbilt University, Nashville.
Pseudosyncope, also known as nonsyncopal fainting (NSF), is a conversion disorder where people appear to faint, but don’t lose consciousness. It’s generally thought to be a physical manifestation of traumatic stress. Patients “aren’t faking it; they believe they are fainting,” said senior investigator Italo Biaggioni, MD, a professor of medicine and pharmacology at the university and director of the Vanderbilt Autonomic Dysfunction Center.
It’s “not benign. These patients stop working, stop driving, and need to depend on other people. They can have very dramatic episodes and hurt themselves” when they fall. “They are very disabled,” he said.
NSF is often misdiagnosed and mistreated, and sometimes unrecognized for years. Almost a third of the 39 NSF cases in Dr. Biaggioni’s series, for instance, were on anticonvulsants, and several had undergone cardiac catheterization. Often, NSF is treated as vasovagal syncope, but patients don’t respond to medications. A better way to identify it is needed. “By the time we get them, they’ve been through a lot.” Dr. Biaggioni said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team had a hunch that judicious use of EEG would help, so they limited EEG to suspected NSF cases in a series of 107 refractory syncope patients referred to Vanderbilt for head-up tilt-table testing; 39 (36%) had normal EEGs during an apparent loss of consciousness, as opposed to the slow-wave pattern of true loss of consciousness, and were diagnosed with NSF.
The 36% identified was a marked increase in incidence over more common approaches. Among 64 patients who had tilt-table testing without EEG at Vanderbilt, for instance, three (5%) were diagnosed with NSF. Historically, tilt table plus EEG in all comers has a diagnostic yield of around 18% for the condition. In short, “elective EEG monitoring during tilt-table testing” better “distinguishes between syncope” and NSF, the team concluded.
NSF is suggested by a history of more than 20 episodes of apparent fainting; episodes once a week or more; or losing consciousness for more than 5 minutes. Fainting with eyes closed, or while supine, is also suggestive.
The elective approach prevents inappropriate treatment but is also therapeutic in itself. “When we document with EEG that patients are not really fainting, and explain that to them, it automatically reduces the number of episodes,” Dr. Biaggioni said.
It also saves NSF patients from a nitroglycerin challenge and repeat tilt testing, which is the default in many places when the first round of testing doesn’t trigger an episode. Challenge testing provokes vasovagal syncope in around 10% of even healthy people, so it puts NSF patients at risk for a false positive. As a rule, “we don’t use provocative agents when we [suspect NSF],” he said.
In addition to the 39 NSF cases, 11 patients in the series were diagnosed with vasovagal syncope, and testing didn’t provoke an event in 57 (53%), which isn’t uncommon.
Baseline blood pressure and heart rates were similar across the three groups, and there were more women than men in each. Subjects were in their early 40s, on average.
NSF patients were more likely to be taking anxiety and depression medications. One NSF patient had posttraumatic stress disorder, and two had sexual abuse histories, compared with none in the nondiagnostic and vasovagal groups. The NSF group had a shorter time to an event: 9 minutes versus 19 minutes among vasovagal patients.
Tilt-table testing was done after 6 hours of fasting, and the team used standard 22-channel EEG. Cognitive-behavioral therapy is the go-to treatment for NSF, Dr. Biaggioni said.
There was no industry funding, and the authors didn’t have any disclosures.
SOURCE: Muldowney JA et al. Joint Hypertension 2019, Abstract P3061.
NEW ORLEANS – , according to an investigation from Vanderbilt University, Nashville.
Pseudosyncope, also known as nonsyncopal fainting (NSF), is a conversion disorder where people appear to faint, but don’t lose consciousness. It’s generally thought to be a physical manifestation of traumatic stress. Patients “aren’t faking it; they believe they are fainting,” said senior investigator Italo Biaggioni, MD, a professor of medicine and pharmacology at the university and director of the Vanderbilt Autonomic Dysfunction Center.
It’s “not benign. These patients stop working, stop driving, and need to depend on other people. They can have very dramatic episodes and hurt themselves” when they fall. “They are very disabled,” he said.
NSF is often misdiagnosed and mistreated, and sometimes unrecognized for years. Almost a third of the 39 NSF cases in Dr. Biaggioni’s series, for instance, were on anticonvulsants, and several had undergone cardiac catheterization. Often, NSF is treated as vasovagal syncope, but patients don’t respond to medications. A better way to identify it is needed. “By the time we get them, they’ve been through a lot.” Dr. Biaggioni said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team had a hunch that judicious use of EEG would help, so they limited EEG to suspected NSF cases in a series of 107 refractory syncope patients referred to Vanderbilt for head-up tilt-table testing; 39 (36%) had normal EEGs during an apparent loss of consciousness, as opposed to the slow-wave pattern of true loss of consciousness, and were diagnosed with NSF.
The 36% identified was a marked increase in incidence over more common approaches. Among 64 patients who had tilt-table testing without EEG at Vanderbilt, for instance, three (5%) were diagnosed with NSF. Historically, tilt table plus EEG in all comers has a diagnostic yield of around 18% for the condition. In short, “elective EEG monitoring during tilt-table testing” better “distinguishes between syncope” and NSF, the team concluded.
NSF is suggested by a history of more than 20 episodes of apparent fainting; episodes once a week or more; or losing consciousness for more than 5 minutes. Fainting with eyes closed, or while supine, is also suggestive.
The elective approach prevents inappropriate treatment but is also therapeutic in itself. “When we document with EEG that patients are not really fainting, and explain that to them, it automatically reduces the number of episodes,” Dr. Biaggioni said.
It also saves NSF patients from a nitroglycerin challenge and repeat tilt testing, which is the default in many places when the first round of testing doesn’t trigger an episode. Challenge testing provokes vasovagal syncope in around 10% of even healthy people, so it puts NSF patients at risk for a false positive. As a rule, “we don’t use provocative agents when we [suspect NSF],” he said.
In addition to the 39 NSF cases, 11 patients in the series were diagnosed with vasovagal syncope, and testing didn’t provoke an event in 57 (53%), which isn’t uncommon.
Baseline blood pressure and heart rates were similar across the three groups, and there were more women than men in each. Subjects were in their early 40s, on average.
NSF patients were more likely to be taking anxiety and depression medications. One NSF patient had posttraumatic stress disorder, and two had sexual abuse histories, compared with none in the nondiagnostic and vasovagal groups. The NSF group had a shorter time to an event: 9 minutes versus 19 minutes among vasovagal patients.
Tilt-table testing was done after 6 hours of fasting, and the team used standard 22-channel EEG. Cognitive-behavioral therapy is the go-to treatment for NSF, Dr. Biaggioni said.
There was no industry funding, and the authors didn’t have any disclosures.
SOURCE: Muldowney JA et al. Joint Hypertension 2019, Abstract P3061.
NEW ORLEANS – , according to an investigation from Vanderbilt University, Nashville.
Pseudosyncope, also known as nonsyncopal fainting (NSF), is a conversion disorder where people appear to faint, but don’t lose consciousness. It’s generally thought to be a physical manifestation of traumatic stress. Patients “aren’t faking it; they believe they are fainting,” said senior investigator Italo Biaggioni, MD, a professor of medicine and pharmacology at the university and director of the Vanderbilt Autonomic Dysfunction Center.
It’s “not benign. These patients stop working, stop driving, and need to depend on other people. They can have very dramatic episodes and hurt themselves” when they fall. “They are very disabled,” he said.
NSF is often misdiagnosed and mistreated, and sometimes unrecognized for years. Almost a third of the 39 NSF cases in Dr. Biaggioni’s series, for instance, were on anticonvulsants, and several had undergone cardiac catheterization. Often, NSF is treated as vasovagal syncope, but patients don’t respond to medications. A better way to identify it is needed. “By the time we get them, they’ve been through a lot.” Dr. Biaggioni said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team had a hunch that judicious use of EEG would help, so they limited EEG to suspected NSF cases in a series of 107 refractory syncope patients referred to Vanderbilt for head-up tilt-table testing; 39 (36%) had normal EEGs during an apparent loss of consciousness, as opposed to the slow-wave pattern of true loss of consciousness, and were diagnosed with NSF.
The 36% identified was a marked increase in incidence over more common approaches. Among 64 patients who had tilt-table testing without EEG at Vanderbilt, for instance, three (5%) were diagnosed with NSF. Historically, tilt table plus EEG in all comers has a diagnostic yield of around 18% for the condition. In short, “elective EEG monitoring during tilt-table testing” better “distinguishes between syncope” and NSF, the team concluded.
NSF is suggested by a history of more than 20 episodes of apparent fainting; episodes once a week or more; or losing consciousness for more than 5 minutes. Fainting with eyes closed, or while supine, is also suggestive.
The elective approach prevents inappropriate treatment but is also therapeutic in itself. “When we document with EEG that patients are not really fainting, and explain that to them, it automatically reduces the number of episodes,” Dr. Biaggioni said.
It also saves NSF patients from a nitroglycerin challenge and repeat tilt testing, which is the default in many places when the first round of testing doesn’t trigger an episode. Challenge testing provokes vasovagal syncope in around 10% of even healthy people, so it puts NSF patients at risk for a false positive. As a rule, “we don’t use provocative agents when we [suspect NSF],” he said.
In addition to the 39 NSF cases, 11 patients in the series were diagnosed with vasovagal syncope, and testing didn’t provoke an event in 57 (53%), which isn’t uncommon.
Baseline blood pressure and heart rates were similar across the three groups, and there were more women than men in each. Subjects were in their early 40s, on average.
NSF patients were more likely to be taking anxiety and depression medications. One NSF patient had posttraumatic stress disorder, and two had sexual abuse histories, compared with none in the nondiagnostic and vasovagal groups. The NSF group had a shorter time to an event: 9 minutes versus 19 minutes among vasovagal patients.
Tilt-table testing was done after 6 hours of fasting, and the team used standard 22-channel EEG. Cognitive-behavioral therapy is the go-to treatment for NSF, Dr. Biaggioni said.
There was no industry funding, and the authors didn’t have any disclosures.
SOURCE: Muldowney JA et al. Joint Hypertension 2019, Abstract P3061.
REPORTING FROM JOINT HYPERTENSION 2019
Machine learning–derived risk score predicts heart failure risk in diabetes patients
PHILADELPHIA – For patients with high-risk diabetes, a novel, machine learning–derived risk score based on 10 common clinical variables can identify those facing a heart failure risk of up to nearly 20% over the ensuing 5 years, an investigator said at the annual meeting of the Heart Failure Society of America.
The risk score, dubbed WATCH-DM, has greater accuracy in predicting incident heart failure than traditional risk-based models, and requires no specific cardiovascular biomarkers or imaging, according to Muthiah Vaduganathan, MD, MPH, a cardiologist at Brigham and Women’s Hospital and faculty at Harvard Medical School in Boston.
The tool may help inform risk-based monitoring and introduction of sodium-glucose transporter 2 (SGLT2) inhibitors, which have been shown in multiple clinical trials to prevent heart failure in at-risk patients with type 2 diabetes mellitus (T2DM), Dr. Vaduganathan said.
“Patients identified at high risk based on WATCH-DM should be strongly considered for initiation of SGLT2 inhibitors in clinical practice,” Dr. Vaduganathan said in an interview.
WATCH-DM is available online at cvriskscores.com. Work is underway to integrate the tool into electronic health record systems at Brigham and Women’s Hospital and at the University of Texas Southwestern Medical Center in Dallas. “I expect that to be launched in the next year,” he said.
The WATCH-DM score was developed based on data from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial, including 8,756 T2DM patients with inadequate glycemic control at high cardiovascular risk and no heart failure at baseline.
Starting with 147 variables, the investigators used a decision-tree machine learning approach to identify predictors of heart failure.
“What machine learning does is automate the variable selection process, as a form of artificial intelligence,” Dr. Vaduganathan said.
The WATCH-DM risk score was based on the 10 best-performing predictors as selected by machine learning, including body mass index, age, systolic blood pressure, diastolic blood pressure, fasting plasma glucose, serum creatinine, high-density lipoprotein cholesterol, QRS duration, prior myocardial infarction, and prior coronary artery bypass grafting.
The 5-year risk of heart failure was just 1.1% for patients with WATCH-DM scores in the lowest quintile, increasing in a graded fashion to nearly 20% (17.4%) in the highest quintile, study results show.
Findings of the study were simultaneously published in the journal Diabetes Care.
Dr. Vaduganathan said he is supported by an award from Harvard Catalyst. He provided disclosures related to Amgen, AstraZeneca, Baxter Healthcare, Bayer AG, Boehringer Ingelheim (advisory boards), and with Novartis and the National Institutes of Health (participation on clinical endpoint committees).
SOURCE: HFSA 2019; Segar MW, Vaduganathan M et al. Diabetes Care. doi: 10.2337/dc19-0587.
PHILADELPHIA – For patients with high-risk diabetes, a novel, machine learning–derived risk score based on 10 common clinical variables can identify those facing a heart failure risk of up to nearly 20% over the ensuing 5 years, an investigator said at the annual meeting of the Heart Failure Society of America.
The risk score, dubbed WATCH-DM, has greater accuracy in predicting incident heart failure than traditional risk-based models, and requires no specific cardiovascular biomarkers or imaging, according to Muthiah Vaduganathan, MD, MPH, a cardiologist at Brigham and Women’s Hospital and faculty at Harvard Medical School in Boston.
The tool may help inform risk-based monitoring and introduction of sodium-glucose transporter 2 (SGLT2) inhibitors, which have been shown in multiple clinical trials to prevent heart failure in at-risk patients with type 2 diabetes mellitus (T2DM), Dr. Vaduganathan said.
“Patients identified at high risk based on WATCH-DM should be strongly considered for initiation of SGLT2 inhibitors in clinical practice,” Dr. Vaduganathan said in an interview.
WATCH-DM is available online at cvriskscores.com. Work is underway to integrate the tool into electronic health record systems at Brigham and Women’s Hospital and at the University of Texas Southwestern Medical Center in Dallas. “I expect that to be launched in the next year,” he said.
The WATCH-DM score was developed based on data from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial, including 8,756 T2DM patients with inadequate glycemic control at high cardiovascular risk and no heart failure at baseline.
Starting with 147 variables, the investigators used a decision-tree machine learning approach to identify predictors of heart failure.
“What machine learning does is automate the variable selection process, as a form of artificial intelligence,” Dr. Vaduganathan said.
The WATCH-DM risk score was based on the 10 best-performing predictors as selected by machine learning, including body mass index, age, systolic blood pressure, diastolic blood pressure, fasting plasma glucose, serum creatinine, high-density lipoprotein cholesterol, QRS duration, prior myocardial infarction, and prior coronary artery bypass grafting.
The 5-year risk of heart failure was just 1.1% for patients with WATCH-DM scores in the lowest quintile, increasing in a graded fashion to nearly 20% (17.4%) in the highest quintile, study results show.
Findings of the study were simultaneously published in the journal Diabetes Care.
Dr. Vaduganathan said he is supported by an award from Harvard Catalyst. He provided disclosures related to Amgen, AstraZeneca, Baxter Healthcare, Bayer AG, Boehringer Ingelheim (advisory boards), and with Novartis and the National Institutes of Health (participation on clinical endpoint committees).
SOURCE: HFSA 2019; Segar MW, Vaduganathan M et al. Diabetes Care. doi: 10.2337/dc19-0587.
PHILADELPHIA – For patients with high-risk diabetes, a novel, machine learning–derived risk score based on 10 common clinical variables can identify those facing a heart failure risk of up to nearly 20% over the ensuing 5 years, an investigator said at the annual meeting of the Heart Failure Society of America.
The risk score, dubbed WATCH-DM, has greater accuracy in predicting incident heart failure than traditional risk-based models, and requires no specific cardiovascular biomarkers or imaging, according to Muthiah Vaduganathan, MD, MPH, a cardiologist at Brigham and Women’s Hospital and faculty at Harvard Medical School in Boston.
The tool may help inform risk-based monitoring and introduction of sodium-glucose transporter 2 (SGLT2) inhibitors, which have been shown in multiple clinical trials to prevent heart failure in at-risk patients with type 2 diabetes mellitus (T2DM), Dr. Vaduganathan said.
“Patients identified at high risk based on WATCH-DM should be strongly considered for initiation of SGLT2 inhibitors in clinical practice,” Dr. Vaduganathan said in an interview.
WATCH-DM is available online at cvriskscores.com. Work is underway to integrate the tool into electronic health record systems at Brigham and Women’s Hospital and at the University of Texas Southwestern Medical Center in Dallas. “I expect that to be launched in the next year,” he said.
The WATCH-DM score was developed based on data from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial, including 8,756 T2DM patients with inadequate glycemic control at high cardiovascular risk and no heart failure at baseline.
Starting with 147 variables, the investigators used a decision-tree machine learning approach to identify predictors of heart failure.
“What machine learning does is automate the variable selection process, as a form of artificial intelligence,” Dr. Vaduganathan said.
The WATCH-DM risk score was based on the 10 best-performing predictors as selected by machine learning, including body mass index, age, systolic blood pressure, diastolic blood pressure, fasting plasma glucose, serum creatinine, high-density lipoprotein cholesterol, QRS duration, prior myocardial infarction, and prior coronary artery bypass grafting.
The 5-year risk of heart failure was just 1.1% for patients with WATCH-DM scores in the lowest quintile, increasing in a graded fashion to nearly 20% (17.4%) in the highest quintile, study results show.
Findings of the study were simultaneously published in the journal Diabetes Care.
Dr. Vaduganathan said he is supported by an award from Harvard Catalyst. He provided disclosures related to Amgen, AstraZeneca, Baxter Healthcare, Bayer AG, Boehringer Ingelheim (advisory boards), and with Novartis and the National Institutes of Health (participation on clinical endpoint committees).
SOURCE: HFSA 2019; Segar MW, Vaduganathan M et al. Diabetes Care. doi: 10.2337/dc19-0587.
REPORTING FROM HFSA 2019
Hyponatremia almost as common with spironolactone as chlorthalidone
NEW ORLEANS – .
The investigators reviewed hypertension patients whose treatment regimens included one diuretic. Forty on chlorthalidone developed hyponatremia – defined as a serum sodium below 133 mEq/L – across 1,322 prescriptions, for an incidence of 3.03%. There were 31 cases across 1,159 spironolactone prescriptions, an incidence of 2.67%.
Among 14 patients in a substudy who discontinued chlorthalidone after developing hyponatremia at a mean of about 2 weeks, six (43%) subsequently developed hyponatremia on spironolactone, also at an average of about 2 weeks.
The findings suggest that spironolactone is more likely than generally thought to cause hyponatremia, a potentially severe complication of diuretics, and that hyponatremia on chlorthalidone increases the risk, said lead investigator Faris Matanes, MD, a hypertension researcher at the university.
“We used to think” that hyponatremia on spironolactone was “very unlikely, but actually it’s not; the incidence is really close to chlorthalidone,” a well-known cause, and “if a patient develops hyponatremia on chlorthalidone, we should be more careful about giving them spironolactone,” he said.
Almost half the spironolactone cases were on 25 mg/day or less, and over a quarter of the chlorthalidone cases were on 12.5 mg/day. Of the 154 hyponatremia cases across 10,660 hydrochlorothiazide prescriptions (1.44%), over a third were taking 12.5 mg/day or less.
Overall, hyponatremia was diagnosed at a mean of 40.4 days, but sometimes after 2 or more months of treatment.
The findings “mean that even if we start patients on a low dose, we can’t stop checking after one or two normal sodium levels.” Measurements need to be ongoing, Dr. Matanes said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team wanted to get around the limitations of previous diuretic hyponatremia studies, including use of more than one diuretic, markedly poor kidney function, and other confounders. To that end, the study was limited to outpatients on a single diuretic who had normal sodium levels both before and after their hyponatremic episode, and estimated glomerular filtration rates (eGFR) of at least 30 mL/min/1.73 m2. Exclusion criteria included heart failure, cirrhosis, and adrenal insufficiency.
Older white people with lower baseline sodium and eGFR values were most at risk. Contrary to previous reports, hyponatremia wasn’t more likely in men.
The mean sodium level during an episode was 130.2 mEq/L; the majority of patients eventually normalized and continued treatment.
Subjects in the main study were a mean of 66 years old, about two-thirds were white, and about 60% were women. The baseline eGFR was 77.2 mL/min/1.73 m2, and baseline sodium level 135.8 mEq/L.
All but one of the 14 substudy patients were women. Those who became hyponatremic when switched to spironolactone were older (mean 74.2 versus 65.8 years), had lower baseline eGFRs (63.7 versus 69.7 mL/min/1.73 m2), and were more likely to be white, but the differences were not statistically significant.
There was no external funding, and the investigators didn’t have any industry disclosures.
SOURCE: Matanes F et al. Joint Hypertension 2019, Abstracts 187 and 174.
NEW ORLEANS – .
The investigators reviewed hypertension patients whose treatment regimens included one diuretic. Forty on chlorthalidone developed hyponatremia – defined as a serum sodium below 133 mEq/L – across 1,322 prescriptions, for an incidence of 3.03%. There were 31 cases across 1,159 spironolactone prescriptions, an incidence of 2.67%.
Among 14 patients in a substudy who discontinued chlorthalidone after developing hyponatremia at a mean of about 2 weeks, six (43%) subsequently developed hyponatremia on spironolactone, also at an average of about 2 weeks.
The findings suggest that spironolactone is more likely than generally thought to cause hyponatremia, a potentially severe complication of diuretics, and that hyponatremia on chlorthalidone increases the risk, said lead investigator Faris Matanes, MD, a hypertension researcher at the university.
“We used to think” that hyponatremia on spironolactone was “very unlikely, but actually it’s not; the incidence is really close to chlorthalidone,” a well-known cause, and “if a patient develops hyponatremia on chlorthalidone, we should be more careful about giving them spironolactone,” he said.
Almost half the spironolactone cases were on 25 mg/day or less, and over a quarter of the chlorthalidone cases were on 12.5 mg/day. Of the 154 hyponatremia cases across 10,660 hydrochlorothiazide prescriptions (1.44%), over a third were taking 12.5 mg/day or less.
Overall, hyponatremia was diagnosed at a mean of 40.4 days, but sometimes after 2 or more months of treatment.
The findings “mean that even if we start patients on a low dose, we can’t stop checking after one or two normal sodium levels.” Measurements need to be ongoing, Dr. Matanes said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team wanted to get around the limitations of previous diuretic hyponatremia studies, including use of more than one diuretic, markedly poor kidney function, and other confounders. To that end, the study was limited to outpatients on a single diuretic who had normal sodium levels both before and after their hyponatremic episode, and estimated glomerular filtration rates (eGFR) of at least 30 mL/min/1.73 m2. Exclusion criteria included heart failure, cirrhosis, and adrenal insufficiency.
Older white people with lower baseline sodium and eGFR values were most at risk. Contrary to previous reports, hyponatremia wasn’t more likely in men.
The mean sodium level during an episode was 130.2 mEq/L; the majority of patients eventually normalized and continued treatment.
Subjects in the main study were a mean of 66 years old, about two-thirds were white, and about 60% were women. The baseline eGFR was 77.2 mL/min/1.73 m2, and baseline sodium level 135.8 mEq/L.
All but one of the 14 substudy patients were women. Those who became hyponatremic when switched to spironolactone were older (mean 74.2 versus 65.8 years), had lower baseline eGFRs (63.7 versus 69.7 mL/min/1.73 m2), and were more likely to be white, but the differences were not statistically significant.
There was no external funding, and the investigators didn’t have any industry disclosures.
SOURCE: Matanes F et al. Joint Hypertension 2019, Abstracts 187 and 174.
NEW ORLEANS – .
The investigators reviewed hypertension patients whose treatment regimens included one diuretic. Forty on chlorthalidone developed hyponatremia – defined as a serum sodium below 133 mEq/L – across 1,322 prescriptions, for an incidence of 3.03%. There were 31 cases across 1,159 spironolactone prescriptions, an incidence of 2.67%.
Among 14 patients in a substudy who discontinued chlorthalidone after developing hyponatremia at a mean of about 2 weeks, six (43%) subsequently developed hyponatremia on spironolactone, also at an average of about 2 weeks.
The findings suggest that spironolactone is more likely than generally thought to cause hyponatremia, a potentially severe complication of diuretics, and that hyponatremia on chlorthalidone increases the risk, said lead investigator Faris Matanes, MD, a hypertension researcher at the university.
“We used to think” that hyponatremia on spironolactone was “very unlikely, but actually it’s not; the incidence is really close to chlorthalidone,” a well-known cause, and “if a patient develops hyponatremia on chlorthalidone, we should be more careful about giving them spironolactone,” he said.
Almost half the spironolactone cases were on 25 mg/day or less, and over a quarter of the chlorthalidone cases were on 12.5 mg/day. Of the 154 hyponatremia cases across 10,660 hydrochlorothiazide prescriptions (1.44%), over a third were taking 12.5 mg/day or less.
Overall, hyponatremia was diagnosed at a mean of 40.4 days, but sometimes after 2 or more months of treatment.
The findings “mean that even if we start patients on a low dose, we can’t stop checking after one or two normal sodium levels.” Measurements need to be ongoing, Dr. Matanes said at the joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
He and his team wanted to get around the limitations of previous diuretic hyponatremia studies, including use of more than one diuretic, markedly poor kidney function, and other confounders. To that end, the study was limited to outpatients on a single diuretic who had normal sodium levels both before and after their hyponatremic episode, and estimated glomerular filtration rates (eGFR) of at least 30 mL/min/1.73 m2. Exclusion criteria included heart failure, cirrhosis, and adrenal insufficiency.
Older white people with lower baseline sodium and eGFR values were most at risk. Contrary to previous reports, hyponatremia wasn’t more likely in men.
The mean sodium level during an episode was 130.2 mEq/L; the majority of patients eventually normalized and continued treatment.
Subjects in the main study were a mean of 66 years old, about two-thirds were white, and about 60% were women. The baseline eGFR was 77.2 mL/min/1.73 m2, and baseline sodium level 135.8 mEq/L.
All but one of the 14 substudy patients were women. Those who became hyponatremic when switched to spironolactone were older (mean 74.2 versus 65.8 years), had lower baseline eGFRs (63.7 versus 69.7 mL/min/1.73 m2), and were more likely to be white, but the differences were not statistically significant.
There was no external funding, and the investigators didn’t have any industry disclosures.
SOURCE: Matanes F et al. Joint Hypertension 2019, Abstracts 187 and 174.
REPORTING FROM JOINT HYPERTENSION 2019
Oral triplet shows promise for relapsed/refractory myeloma
BOSTON – The all-oral combination of selinexor, lenalidomide, and dexamethasone has demonstrated efficacy in patients with relapsed/refractory multiple myeloma, particularly those who are lenalidomide naive.
In the phase 1/2 STOMP trial (NCT02343042), selinexor plus lenalidomide and dexamethasone produced an overall response rate (ORR) of 60% in all evaluable patients and an ORR of 92% in lenalidomide-naive patients.
Darrell White, MD, of Dalhousie University in Halifax, N.S., presented these results at the International Myeloma Workshop held by the International Myeloma Society.
Dr. White discussed results in myeloma patients who had received at least one prior line of therapy. This arm of the STOMP trial has enrolled 24 patients. Their median age at baseline was 67 years (range, 49-84 years), and their median time from diagnosis was 4.5 years (range, less than 1-22 years).
The patients had received a median of 1 (range, 1-8) prior therapies. Half of patients had received a transplant. All patients had received a proteasome inhibitor (65% refractory), and 38% had received prior lenalidomide (21% refractory).
Dosing
Patients received selinexor at 60 mg once or twice weekly or at 80 mg once weekly on a 28-day cycle. They received dexamethasone at 20 mg twice weekly or 40 mg once weekly and lenalidomide at 25 mg once daily every 21 days.
The recommended phase 2 dosing schedule was selinexor at 60 mg once weekly plus lenalidomide at 25 mg daily and dexamethasone at 40 mg once weekly. Half of patients (n = 12) received this dosing schedule.
There were no dose-limiting toxicities (DLTs) at the recommended phase 2 dose. When selinexor was given at 60 mg twice weekly, DLTs included grade 3 fatigue, grade 3 anorexia and weight loss, and grade 4 thrombocytopenia (n = 2). When selinexor was given at 80 mg once weekly, the DLTs were grade 4 thrombocytopenia (n = 2).
Safety
“The side-effect profile was as expected, with mainly grade 3/4 toxicity being hematologic and primarily thrombocytopenia and neutropenia,” Dr. White said. “Frequent gastrointestinal side effects [were] expected. Investigators were able to manage that with appropriate supportive care and antiemetics in particular.”
Among patients who received the recommended phase 2 dose, the grade 4 treatment-related adverse events (AEs) were thrombocytopenia (n = 4) and neutropenia (n = 4). Grade 3 treatment-related AEs were thrombocytopenia (n = 3), neutropenia (n = 4), anemia (n = 1), nausea (n = 1), asthenia (n = 1), fatigue (n = 2), and dehydration (n = 1). Common grade 1/2 treatment-related AEs in patients who received the recommended phase 2 dose were nausea (n = 6), anorexia (n = 5), weight loss (n = 5), vomiting (n = 4), diarrhea (n = 4), and fatigue (n = 4).
Efficacy
In the 20 patients evaluable for response, the ORR was 60% (n = 12). One patient achieved a stringent complete response, three had a very good partial response, and eight had a partial response. The median time to response was 1 month.
The ORR was 92% (11/12) in lenalidomide-naive patients and 13% (1/8) in lenalidomide-exposed patients. The single responder in the lenalidomide-exposed group achieved a partial response.
Among lenalidomide-naive patients who received the recommended phase 2 dose, the median time on treatment was 12 months. The patient who achieved a stringent complete response remained on treatment at last follow-up, as did one partial responder and one patient with a very good partial response.
These results suggest the combination of selinexor, lenalidomide, and dexamethasone “is active, relatively well tolerated, and could warrant further investigation,” Dr. White said.
The STOMP trial is sponsored by Karyopharm Therapeutics. Dr. White disclosed relationships with Karyopharm, Amgen, Celgene, Janssen, Sanofi, and Takeda.
SOURCE: White D et al. IMW 2019, Abstract OAB-083.
BOSTON – The all-oral combination of selinexor, lenalidomide, and dexamethasone has demonstrated efficacy in patients with relapsed/refractory multiple myeloma, particularly those who are lenalidomide naive.
In the phase 1/2 STOMP trial (NCT02343042), selinexor plus lenalidomide and dexamethasone produced an overall response rate (ORR) of 60% in all evaluable patients and an ORR of 92% in lenalidomide-naive patients.
Darrell White, MD, of Dalhousie University in Halifax, N.S., presented these results at the International Myeloma Workshop held by the International Myeloma Society.
Dr. White discussed results in myeloma patients who had received at least one prior line of therapy. This arm of the STOMP trial has enrolled 24 patients. Their median age at baseline was 67 years (range, 49-84 years), and their median time from diagnosis was 4.5 years (range, less than 1-22 years).
The patients had received a median of 1 (range, 1-8) prior therapies. Half of patients had received a transplant. All patients had received a proteasome inhibitor (65% refractory), and 38% had received prior lenalidomide (21% refractory).
Dosing
Patients received selinexor at 60 mg once or twice weekly or at 80 mg once weekly on a 28-day cycle. They received dexamethasone at 20 mg twice weekly or 40 mg once weekly and lenalidomide at 25 mg once daily every 21 days.
The recommended phase 2 dosing schedule was selinexor at 60 mg once weekly plus lenalidomide at 25 mg daily and dexamethasone at 40 mg once weekly. Half of patients (n = 12) received this dosing schedule.
There were no dose-limiting toxicities (DLTs) at the recommended phase 2 dose. When selinexor was given at 60 mg twice weekly, DLTs included grade 3 fatigue, grade 3 anorexia and weight loss, and grade 4 thrombocytopenia (n = 2). When selinexor was given at 80 mg once weekly, the DLTs were grade 4 thrombocytopenia (n = 2).
Safety
“The side-effect profile was as expected, with mainly grade 3/4 toxicity being hematologic and primarily thrombocytopenia and neutropenia,” Dr. White said. “Frequent gastrointestinal side effects [were] expected. Investigators were able to manage that with appropriate supportive care and antiemetics in particular.”
Among patients who received the recommended phase 2 dose, the grade 4 treatment-related adverse events (AEs) were thrombocytopenia (n = 4) and neutropenia (n = 4). Grade 3 treatment-related AEs were thrombocytopenia (n = 3), neutropenia (n = 4), anemia (n = 1), nausea (n = 1), asthenia (n = 1), fatigue (n = 2), and dehydration (n = 1). Common grade 1/2 treatment-related AEs in patients who received the recommended phase 2 dose were nausea (n = 6), anorexia (n = 5), weight loss (n = 5), vomiting (n = 4), diarrhea (n = 4), and fatigue (n = 4).
Efficacy
In the 20 patients evaluable for response, the ORR was 60% (n = 12). One patient achieved a stringent complete response, three had a very good partial response, and eight had a partial response. The median time to response was 1 month.
The ORR was 92% (11/12) in lenalidomide-naive patients and 13% (1/8) in lenalidomide-exposed patients. The single responder in the lenalidomide-exposed group achieved a partial response.
Among lenalidomide-naive patients who received the recommended phase 2 dose, the median time on treatment was 12 months. The patient who achieved a stringent complete response remained on treatment at last follow-up, as did one partial responder and one patient with a very good partial response.
These results suggest the combination of selinexor, lenalidomide, and dexamethasone “is active, relatively well tolerated, and could warrant further investigation,” Dr. White said.
The STOMP trial is sponsored by Karyopharm Therapeutics. Dr. White disclosed relationships with Karyopharm, Amgen, Celgene, Janssen, Sanofi, and Takeda.
SOURCE: White D et al. IMW 2019, Abstract OAB-083.
BOSTON – The all-oral combination of selinexor, lenalidomide, and dexamethasone has demonstrated efficacy in patients with relapsed/refractory multiple myeloma, particularly those who are lenalidomide naive.
In the phase 1/2 STOMP trial (NCT02343042), selinexor plus lenalidomide and dexamethasone produced an overall response rate (ORR) of 60% in all evaluable patients and an ORR of 92% in lenalidomide-naive patients.
Darrell White, MD, of Dalhousie University in Halifax, N.S., presented these results at the International Myeloma Workshop held by the International Myeloma Society.
Dr. White discussed results in myeloma patients who had received at least one prior line of therapy. This arm of the STOMP trial has enrolled 24 patients. Their median age at baseline was 67 years (range, 49-84 years), and their median time from diagnosis was 4.5 years (range, less than 1-22 years).
The patients had received a median of 1 (range, 1-8) prior therapies. Half of patients had received a transplant. All patients had received a proteasome inhibitor (65% refractory), and 38% had received prior lenalidomide (21% refractory).
Dosing
Patients received selinexor at 60 mg once or twice weekly or at 80 mg once weekly on a 28-day cycle. They received dexamethasone at 20 mg twice weekly or 40 mg once weekly and lenalidomide at 25 mg once daily every 21 days.
The recommended phase 2 dosing schedule was selinexor at 60 mg once weekly plus lenalidomide at 25 mg daily and dexamethasone at 40 mg once weekly. Half of patients (n = 12) received this dosing schedule.
There were no dose-limiting toxicities (DLTs) at the recommended phase 2 dose. When selinexor was given at 60 mg twice weekly, DLTs included grade 3 fatigue, grade 3 anorexia and weight loss, and grade 4 thrombocytopenia (n = 2). When selinexor was given at 80 mg once weekly, the DLTs were grade 4 thrombocytopenia (n = 2).
Safety
“The side-effect profile was as expected, with mainly grade 3/4 toxicity being hematologic and primarily thrombocytopenia and neutropenia,” Dr. White said. “Frequent gastrointestinal side effects [were] expected. Investigators were able to manage that with appropriate supportive care and antiemetics in particular.”
Among patients who received the recommended phase 2 dose, the grade 4 treatment-related adverse events (AEs) were thrombocytopenia (n = 4) and neutropenia (n = 4). Grade 3 treatment-related AEs were thrombocytopenia (n = 3), neutropenia (n = 4), anemia (n = 1), nausea (n = 1), asthenia (n = 1), fatigue (n = 2), and dehydration (n = 1). Common grade 1/2 treatment-related AEs in patients who received the recommended phase 2 dose were nausea (n = 6), anorexia (n = 5), weight loss (n = 5), vomiting (n = 4), diarrhea (n = 4), and fatigue (n = 4).
Efficacy
In the 20 patients evaluable for response, the ORR was 60% (n = 12). One patient achieved a stringent complete response, three had a very good partial response, and eight had a partial response. The median time to response was 1 month.
The ORR was 92% (11/12) in lenalidomide-naive patients and 13% (1/8) in lenalidomide-exposed patients. The single responder in the lenalidomide-exposed group achieved a partial response.
Among lenalidomide-naive patients who received the recommended phase 2 dose, the median time on treatment was 12 months. The patient who achieved a stringent complete response remained on treatment at last follow-up, as did one partial responder and one patient with a very good partial response.
These results suggest the combination of selinexor, lenalidomide, and dexamethasone “is active, relatively well tolerated, and could warrant further investigation,” Dr. White said.
The STOMP trial is sponsored by Karyopharm Therapeutics. Dr. White disclosed relationships with Karyopharm, Amgen, Celgene, Janssen, Sanofi, and Takeda.
SOURCE: White D et al. IMW 2019, Abstract OAB-083.
REPORTING FROM IMW 2019
Is EVAR for ruptured AAA worth revisiting?
CHICAGO – Numerous studies have shown conflicting results for endovascular repair in ruptured abdominal aortic aneurysms (AAA), but an analysis of 4,000-plus cases from a national registry has found a 41% reduction in mortality with endovascular repair vs. open repair, according to a presentation at the annual meeting of the Midwestern Vascular Surgery Society.
“EVAR is becoming an increasingly popular strategy for treatment of AAA,” said Samer Alharthi, MD, MPH, of the University of Toledo in Ohio. “As surgeon experience and endovascular technology have improved, a greater percentage of ruptured AAA are being treated by EVAR.”
Dr. Alharthi reported on a retrospective analysis of 4,133 patients who had repair for ruptured AAA in the American College of Surgeons National Surgical Quality Improvement Program database from 2010 to 2016. Notably, the number of EVAR repairs continue to increase and peaked in 2015, with 53% of ruptured AAA treated by EVAR.
Over the term of the study, the overall mortality rate was 22.6% for EVAR and 33.2% for open repair (P less than .001), Dr. Alharthi said. “After adjusting for cofounders, there was a 41% reduction in the mortality rate with the EVAR approach,” he said.
The only appreciable significant difference in demographics between the two groups was a higher percentage of smokers with chronic obstructive pulmonary disease having open repair – 942 (49.2%) vs. 701 (36.2%) – and a higher percentage of patients with end-stage renal disease having EVAR, Dr. Alharthi said. Other comorbidities had no statistically significant difference.
“Complications – pneumonia, reintubation, and acute renal failure – were higher in the open than the EVAR group,” he said. For example, rates of acute renal failure were 15.4% and 8.2% (P less than.001), respectively. Rates of myocardial infarction were similar between the two groups: 6.3% and 6% (P = .74), respectively.
Dr. Alharthi had no financial relationships to disclose.
SOURCE: Alharthi S et al. Midwestern Vascular 2019, Abstract 13.
CHICAGO – Numerous studies have shown conflicting results for endovascular repair in ruptured abdominal aortic aneurysms (AAA), but an analysis of 4,000-plus cases from a national registry has found a 41% reduction in mortality with endovascular repair vs. open repair, according to a presentation at the annual meeting of the Midwestern Vascular Surgery Society.
“EVAR is becoming an increasingly popular strategy for treatment of AAA,” said Samer Alharthi, MD, MPH, of the University of Toledo in Ohio. “As surgeon experience and endovascular technology have improved, a greater percentage of ruptured AAA are being treated by EVAR.”
Dr. Alharthi reported on a retrospective analysis of 4,133 patients who had repair for ruptured AAA in the American College of Surgeons National Surgical Quality Improvement Program database from 2010 to 2016. Notably, the number of EVAR repairs continue to increase and peaked in 2015, with 53% of ruptured AAA treated by EVAR.
Over the term of the study, the overall mortality rate was 22.6% for EVAR and 33.2% for open repair (P less than .001), Dr. Alharthi said. “After adjusting for cofounders, there was a 41% reduction in the mortality rate with the EVAR approach,” he said.
The only appreciable significant difference in demographics between the two groups was a higher percentage of smokers with chronic obstructive pulmonary disease having open repair – 942 (49.2%) vs. 701 (36.2%) – and a higher percentage of patients with end-stage renal disease having EVAR, Dr. Alharthi said. Other comorbidities had no statistically significant difference.
“Complications – pneumonia, reintubation, and acute renal failure – were higher in the open than the EVAR group,” he said. For example, rates of acute renal failure were 15.4% and 8.2% (P less than.001), respectively. Rates of myocardial infarction were similar between the two groups: 6.3% and 6% (P = .74), respectively.
Dr. Alharthi had no financial relationships to disclose.
SOURCE: Alharthi S et al. Midwestern Vascular 2019, Abstract 13.
CHICAGO – Numerous studies have shown conflicting results for endovascular repair in ruptured abdominal aortic aneurysms (AAA), but an analysis of 4,000-plus cases from a national registry has found a 41% reduction in mortality with endovascular repair vs. open repair, according to a presentation at the annual meeting of the Midwestern Vascular Surgery Society.
“EVAR is becoming an increasingly popular strategy for treatment of AAA,” said Samer Alharthi, MD, MPH, of the University of Toledo in Ohio. “As surgeon experience and endovascular technology have improved, a greater percentage of ruptured AAA are being treated by EVAR.”
Dr. Alharthi reported on a retrospective analysis of 4,133 patients who had repair for ruptured AAA in the American College of Surgeons National Surgical Quality Improvement Program database from 2010 to 2016. Notably, the number of EVAR repairs continue to increase and peaked in 2015, with 53% of ruptured AAA treated by EVAR.
Over the term of the study, the overall mortality rate was 22.6% for EVAR and 33.2% for open repair (P less than .001), Dr. Alharthi said. “After adjusting for cofounders, there was a 41% reduction in the mortality rate with the EVAR approach,” he said.
The only appreciable significant difference in demographics between the two groups was a higher percentage of smokers with chronic obstructive pulmonary disease having open repair – 942 (49.2%) vs. 701 (36.2%) – and a higher percentage of patients with end-stage renal disease having EVAR, Dr. Alharthi said. Other comorbidities had no statistically significant difference.
“Complications – pneumonia, reintubation, and acute renal failure – were higher in the open than the EVAR group,” he said. For example, rates of acute renal failure were 15.4% and 8.2% (P less than.001), respectively. Rates of myocardial infarction were similar between the two groups: 6.3% and 6% (P = .74), respectively.
Dr. Alharthi had no financial relationships to disclose.
SOURCE: Alharthi S et al. Midwestern Vascular 2019, Abstract 13.
REPORTING FROM MIDWESTERN VASCULAR 2019