User login
Delaying RT for higher-risk prostate cancer found safe
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
FROM JAMA ONCOLOGY
Fecal transplant shows promise in reducing alcohol craving
Fecal microbiota transplantation results in a short-term reduction in alcohol craving in patients with alcohol-induced cirrhosis who can’t stop drinking, results from a new study show.
And that reduction could lead to a better psychosocial quality of life for patients with cirrhosis and alcohol use disorder, said investigator Jasmohan Bajaj, MD, from Virginia Commonwealth University, Richmond.
“This is the most common addiction disorder worldwide, but we have nothing to treat these patients with,” he said.
Cirrhosis is associated with an altered gut-brain axis. It leads to organ damage in several parts of the body, including the brain, gut, pancreas, and liver. This makes changing the gut microbes “an attractive target,” Dr. Bajaj said at the Digital International Liver Congress 2020.
For their phase 1, double-blind study, he and his colleagues assessed 20 men from a Virginia veteran’s hospital with untreatable alcohol use disorder who were not eligible for liver transplantation.
All had failed behavioral or pharmacologic therapy and were unwilling to try again. “That’s what made them good candidates to try something new,” Dr. Bajaj said during a press briefing.
Mean age in the study cohort was 65 years, mean Model for End-Stage Liver disease score was 8.9, and demographic characteristics were similar between the 10 men randomly assigned to fecal transplantation and the 10 assigned to placebo. One man in each group dropped out of the study.
The investigators evaluated cravings, microbiota, and quality of life during the 30-day study period.
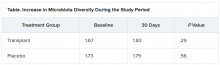
At day 15, significantly more men in the transplant group than in the placebo group experienced a reduction in alcohol cravings (90% vs. 30%).
At 30 days, levels of creatinine, serum interleukin-6, and lipopolysaccharide-binding protein were lower in the transplant group than in the placebo group. In addition, levels of butyrate and isobutyrate increased, as did cognition and quality of life scores.
There was also a decrease in urinary ethyl glucuronide in the transplant group, which “is the objective criteria for alcohol intake,” Dr. Bajaj reported, noting that there was no change in ethyl glucuronide in the placebo group.
The increase in microbiota diversity was significant in the transplant group but not in the placebo group. Alistipes, Odoribacter, and Roseburia were more abundant in the transplant group than in the placebo group.
During the 30-day study period, two men in the placebo group required medical attention, one for hyponatremia and the other for atrial fibrillation. However, no adverse events were seen in any men in the transplant group. “This was the No. 1 result,” Dr. Bajaj said.
Liver disease and the microbiome
“Understanding of interactions between the human and microbiome genome [metagenome] in health and disease has represented one of the major areas of progress in the last few years,” said Luca Valenti, MD, from the University of Milan, who is a member of the scientific committee of the European Association the Study of the Liver, which organized the congress.
“These studies lay the groundwork for the exploitation of this new knowledge for the treatment of liver disease,” he said.
“We are [now] diagnosing liver disease and the stages of liver disease based on microbiome changes,” said Jonel Trebicka, MD, PhD, from University Hospital Frankfurt (Germany), who chaired a session at the congress on the role of the microbiome in liver disease.
“This and other studies have shown us that the microbiome itself may influence liver disease,” he added.
Dr. Bajaj is considered one of the world’s experts on cirrhosis and the microbiome, Dr. Trebicka explained. Last year, Dr. Bajaj and his team demonstrated that fecal microbiota transplantation can reduce the incidence of recurrent hepatic encephalopathy, as reported by Medscape Medical News.
The current study also “shows clearly that the microbiome plays a role in craving. FMT reduces the desire for alcohol,” said Dr. Trebicka.
“The way to the brain is through the gut,” Dr. Bajaj said.
Dr. Bajaj, Dr. Trebicka, and Dr. Valenti disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Fecal microbiota transplantation results in a short-term reduction in alcohol craving in patients with alcohol-induced cirrhosis who can’t stop drinking, results from a new study show.
And that reduction could lead to a better psychosocial quality of life for patients with cirrhosis and alcohol use disorder, said investigator Jasmohan Bajaj, MD, from Virginia Commonwealth University, Richmond.
“This is the most common addiction disorder worldwide, but we have nothing to treat these patients with,” he said.
Cirrhosis is associated with an altered gut-brain axis. It leads to organ damage in several parts of the body, including the brain, gut, pancreas, and liver. This makes changing the gut microbes “an attractive target,” Dr. Bajaj said at the Digital International Liver Congress 2020.
For their phase 1, double-blind study, he and his colleagues assessed 20 men from a Virginia veteran’s hospital with untreatable alcohol use disorder who were not eligible for liver transplantation.
All had failed behavioral or pharmacologic therapy and were unwilling to try again. “That’s what made them good candidates to try something new,” Dr. Bajaj said during a press briefing.
Mean age in the study cohort was 65 years, mean Model for End-Stage Liver disease score was 8.9, and demographic characteristics were similar between the 10 men randomly assigned to fecal transplantation and the 10 assigned to placebo. One man in each group dropped out of the study.
The investigators evaluated cravings, microbiota, and quality of life during the 30-day study period.
At day 15, significantly more men in the transplant group than in the placebo group experienced a reduction in alcohol cravings (90% vs. 30%).
At 30 days, levels of creatinine, serum interleukin-6, and lipopolysaccharide-binding protein were lower in the transplant group than in the placebo group. In addition, levels of butyrate and isobutyrate increased, as did cognition and quality of life scores.
There was also a decrease in urinary ethyl glucuronide in the transplant group, which “is the objective criteria for alcohol intake,” Dr. Bajaj reported, noting that there was no change in ethyl glucuronide in the placebo group.
The increase in microbiota diversity was significant in the transplant group but not in the placebo group. Alistipes, Odoribacter, and Roseburia were more abundant in the transplant group than in the placebo group.
During the 30-day study period, two men in the placebo group required medical attention, one for hyponatremia and the other for atrial fibrillation. However, no adverse events were seen in any men in the transplant group. “This was the No. 1 result,” Dr. Bajaj said.
Liver disease and the microbiome
“Understanding of interactions between the human and microbiome genome [metagenome] in health and disease has represented one of the major areas of progress in the last few years,” said Luca Valenti, MD, from the University of Milan, who is a member of the scientific committee of the European Association the Study of the Liver, which organized the congress.
“These studies lay the groundwork for the exploitation of this new knowledge for the treatment of liver disease,” he said.
“We are [now] diagnosing liver disease and the stages of liver disease based on microbiome changes,” said Jonel Trebicka, MD, PhD, from University Hospital Frankfurt (Germany), who chaired a session at the congress on the role of the microbiome in liver disease.
“This and other studies have shown us that the microbiome itself may influence liver disease,” he added.
Dr. Bajaj is considered one of the world’s experts on cirrhosis and the microbiome, Dr. Trebicka explained. Last year, Dr. Bajaj and his team demonstrated that fecal microbiota transplantation can reduce the incidence of recurrent hepatic encephalopathy, as reported by Medscape Medical News.
The current study also “shows clearly that the microbiome plays a role in craving. FMT reduces the desire for alcohol,” said Dr. Trebicka.
“The way to the brain is through the gut,” Dr. Bajaj said.
Dr. Bajaj, Dr. Trebicka, and Dr. Valenti disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Fecal microbiota transplantation results in a short-term reduction in alcohol craving in patients with alcohol-induced cirrhosis who can’t stop drinking, results from a new study show.
And that reduction could lead to a better psychosocial quality of life for patients with cirrhosis and alcohol use disorder, said investigator Jasmohan Bajaj, MD, from Virginia Commonwealth University, Richmond.
“This is the most common addiction disorder worldwide, but we have nothing to treat these patients with,” he said.
Cirrhosis is associated with an altered gut-brain axis. It leads to organ damage in several parts of the body, including the brain, gut, pancreas, and liver. This makes changing the gut microbes “an attractive target,” Dr. Bajaj said at the Digital International Liver Congress 2020.
For their phase 1, double-blind study, he and his colleagues assessed 20 men from a Virginia veteran’s hospital with untreatable alcohol use disorder who were not eligible for liver transplantation.
All had failed behavioral or pharmacologic therapy and were unwilling to try again. “That’s what made them good candidates to try something new,” Dr. Bajaj said during a press briefing.
Mean age in the study cohort was 65 years, mean Model for End-Stage Liver disease score was 8.9, and demographic characteristics were similar between the 10 men randomly assigned to fecal transplantation and the 10 assigned to placebo. One man in each group dropped out of the study.
The investigators evaluated cravings, microbiota, and quality of life during the 30-day study period.
At day 15, significantly more men in the transplant group than in the placebo group experienced a reduction in alcohol cravings (90% vs. 30%).
At 30 days, levels of creatinine, serum interleukin-6, and lipopolysaccharide-binding protein were lower in the transplant group than in the placebo group. In addition, levels of butyrate and isobutyrate increased, as did cognition and quality of life scores.
There was also a decrease in urinary ethyl glucuronide in the transplant group, which “is the objective criteria for alcohol intake,” Dr. Bajaj reported, noting that there was no change in ethyl glucuronide in the placebo group.
The increase in microbiota diversity was significant in the transplant group but not in the placebo group. Alistipes, Odoribacter, and Roseburia were more abundant in the transplant group than in the placebo group.
During the 30-day study period, two men in the placebo group required medical attention, one for hyponatremia and the other for atrial fibrillation. However, no adverse events were seen in any men in the transplant group. “This was the No. 1 result,” Dr. Bajaj said.
Liver disease and the microbiome
“Understanding of interactions between the human and microbiome genome [metagenome] in health and disease has represented one of the major areas of progress in the last few years,” said Luca Valenti, MD, from the University of Milan, who is a member of the scientific committee of the European Association the Study of the Liver, which organized the congress.
“These studies lay the groundwork for the exploitation of this new knowledge for the treatment of liver disease,” he said.
“We are [now] diagnosing liver disease and the stages of liver disease based on microbiome changes,” said Jonel Trebicka, MD, PhD, from University Hospital Frankfurt (Germany), who chaired a session at the congress on the role of the microbiome in liver disease.
“This and other studies have shown us that the microbiome itself may influence liver disease,” he added.
Dr. Bajaj is considered one of the world’s experts on cirrhosis and the microbiome, Dr. Trebicka explained. Last year, Dr. Bajaj and his team demonstrated that fecal microbiota transplantation can reduce the incidence of recurrent hepatic encephalopathy, as reported by Medscape Medical News.
The current study also “shows clearly that the microbiome plays a role in craving. FMT reduces the desire for alcohol,” said Dr. Trebicka.
“The way to the brain is through the gut,” Dr. Bajaj said.
Dr. Bajaj, Dr. Trebicka, and Dr. Valenti disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Psoriasis, PsA, and pregnancy: Tailoring treatment with increasing data
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
Interstitial lung abnormalities linked to COPD exacerbations
Patients with chronic obstructive pulmonary disease who also had certain interstitial lung abnormalities experienced more exacerbations and reduced lung function than those without such abnormalities, findings from a retrospective study has shown.
Interstitial lung abnormalities (ILA) are considered precursor lesions of interstitial lung disease and previous studies suggested an association with poor outcomes among chronic obstructive pulmonary disease (COPD) patients, but data on long-term clinical relevance are limited, wrote Tae Seung Lee, MD, of Seoul (South Korea) National University Hospital, and colleagues.
In a study published in Chest, the researchers reviewed data from 363 COPD patients including 44 with equivocal ILA and 103 with definite ILA. Overall, the ILA patients were older and had poorer lung function than non-ILA patients. Patients received chest CT scan and longitudinal pulmonary function tests between January 2013 and December 2018.
Over an average follow-up period of 5.4 years, patients with ILA experienced significantly more acute COPD exacerbations than did those without ILA (adjusted odds ratio, 2.03). The percentages of frequent exacerbators among patients with no ILA, equivocal ILA, and definite ILA were 8.3%, 15.9%, and 20.4%, respectively.
“Acute exacerbation is an important event during the clinical course of COPD, because it is associated with temporary or persistent reductions in lung function, lower quality of life, hospitalization, and mortality,” the researchers noted.
In a multivariate analysis, the annual decline in lung function (FEV1) was –35.7 in patients with equivocal ILA, compared with –28.0 in patients with no ILA and –15.9 in those with definite ILA.
“This may be due to the distribution of the spirometric stages in each group, and to the resulting changes in lung function,” the researchers wrote. In this study, “the equivocal ILA group had a significantly lower baseline FEV1 than the other groups. In our study population, the lower the spirometric stage, the faster the annual decline in FEV1, consistent with the results of a prior prospective study of a COPD cohort.”
The findings were limited by several factors including the retrospective design and relatively small number of ILA patients, as well as the limited evaluation of ILA and potential for selection bias, the researchers noted. However, the result support the impact of ILA on exacerbations and accelerated decline in lung function in COPD patients.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Lee TS et al. Chest. 2020 Aug 13. doi: 10.1016/j.chest.2020.08.017.
Patients with chronic obstructive pulmonary disease who also had certain interstitial lung abnormalities experienced more exacerbations and reduced lung function than those without such abnormalities, findings from a retrospective study has shown.
Interstitial lung abnormalities (ILA) are considered precursor lesions of interstitial lung disease and previous studies suggested an association with poor outcomes among chronic obstructive pulmonary disease (COPD) patients, but data on long-term clinical relevance are limited, wrote Tae Seung Lee, MD, of Seoul (South Korea) National University Hospital, and colleagues.
In a study published in Chest, the researchers reviewed data from 363 COPD patients including 44 with equivocal ILA and 103 with definite ILA. Overall, the ILA patients were older and had poorer lung function than non-ILA patients. Patients received chest CT scan and longitudinal pulmonary function tests between January 2013 and December 2018.
Over an average follow-up period of 5.4 years, patients with ILA experienced significantly more acute COPD exacerbations than did those without ILA (adjusted odds ratio, 2.03). The percentages of frequent exacerbators among patients with no ILA, equivocal ILA, and definite ILA were 8.3%, 15.9%, and 20.4%, respectively.
“Acute exacerbation is an important event during the clinical course of COPD, because it is associated with temporary or persistent reductions in lung function, lower quality of life, hospitalization, and mortality,” the researchers noted.
In a multivariate analysis, the annual decline in lung function (FEV1) was –35.7 in patients with equivocal ILA, compared with –28.0 in patients with no ILA and –15.9 in those with definite ILA.
“This may be due to the distribution of the spirometric stages in each group, and to the resulting changes in lung function,” the researchers wrote. In this study, “the equivocal ILA group had a significantly lower baseline FEV1 than the other groups. In our study population, the lower the spirometric stage, the faster the annual decline in FEV1, consistent with the results of a prior prospective study of a COPD cohort.”
The findings were limited by several factors including the retrospective design and relatively small number of ILA patients, as well as the limited evaluation of ILA and potential for selection bias, the researchers noted. However, the result support the impact of ILA on exacerbations and accelerated decline in lung function in COPD patients.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Lee TS et al. Chest. 2020 Aug 13. doi: 10.1016/j.chest.2020.08.017.
Patients with chronic obstructive pulmonary disease who also had certain interstitial lung abnormalities experienced more exacerbations and reduced lung function than those without such abnormalities, findings from a retrospective study has shown.
Interstitial lung abnormalities (ILA) are considered precursor lesions of interstitial lung disease and previous studies suggested an association with poor outcomes among chronic obstructive pulmonary disease (COPD) patients, but data on long-term clinical relevance are limited, wrote Tae Seung Lee, MD, of Seoul (South Korea) National University Hospital, and colleagues.
In a study published in Chest, the researchers reviewed data from 363 COPD patients including 44 with equivocal ILA and 103 with definite ILA. Overall, the ILA patients were older and had poorer lung function than non-ILA patients. Patients received chest CT scan and longitudinal pulmonary function tests between January 2013 and December 2018.
Over an average follow-up period of 5.4 years, patients with ILA experienced significantly more acute COPD exacerbations than did those without ILA (adjusted odds ratio, 2.03). The percentages of frequent exacerbators among patients with no ILA, equivocal ILA, and definite ILA were 8.3%, 15.9%, and 20.4%, respectively.
“Acute exacerbation is an important event during the clinical course of COPD, because it is associated with temporary or persistent reductions in lung function, lower quality of life, hospitalization, and mortality,” the researchers noted.
In a multivariate analysis, the annual decline in lung function (FEV1) was –35.7 in patients with equivocal ILA, compared with –28.0 in patients with no ILA and –15.9 in those with definite ILA.
“This may be due to the distribution of the spirometric stages in each group, and to the resulting changes in lung function,” the researchers wrote. In this study, “the equivocal ILA group had a significantly lower baseline FEV1 than the other groups. In our study population, the lower the spirometric stage, the faster the annual decline in FEV1, consistent with the results of a prior prospective study of a COPD cohort.”
The findings were limited by several factors including the retrospective design and relatively small number of ILA patients, as well as the limited evaluation of ILA and potential for selection bias, the researchers noted. However, the result support the impact of ILA on exacerbations and accelerated decline in lung function in COPD patients.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Lee TS et al. Chest. 2020 Aug 13. doi: 10.1016/j.chest.2020.08.017.
FROM CHEST
Napabucasin suppressed tumor growth in DLBCL cell lines
The STAT3 pathway is important to the development of many cancers, but so far, no Food and Drug Administration–approved drugs are ready for clinical use. Napabucasin, a novel oral small-molecule inhibitor of signal transducer and activator of transcription 3 (STAT3), blocks tumor growth and resists metastasis in a broad spectrum of solid tumors. Napabucasin was now found to be effective in inhibiting diffuse large beta cell lymphoma (DLBCL) in cell lines and in in vitro testing, as reported by Xue Li of the West China Hospital, Sichuan (China) University, and colleagues.
In addition, the effects of napabucasin were found to be synergistic with the use of doxorubicin, a standard DLBCL therapy agent, according to the report, published online in Cancer Letters.
‘Dramatic’ results
The researchers found that 34% (23/69) of DLBCL patients expressed STAT3 in tumor tissues. When they tested napabucasin in a variety of DLBCL cell lines they found that the drug exhibited potent cytotoxicity in a dose-dependent manner. In addition, they found that napabucasin induced intrinsic and extrinsic cell apoptosis, downregulated the expression of STAT3 target genes, including the antiapoptotic protein Mcl-1, and regulated the mitogen-activated protein kinase (MAPK) pathway, all important indicators of antitumor effectiveness in vitro.
In cells treated with napabucasin and doxorubicin alone and in combination, napabucasin alone significantly suppressed tumor growth, compared with that of the control (P < .01), achieving tumor growth inhibition (TGI) of 78.8%. The combination treatment, “with a dramatic TGI of 98.2%,” was more effective than doxorubicin monotherapy (TGI = 63.2%; P < .05), according to the researchers.
“Our study provided evidence that napabucasin is an attractive candidate drug either as a monotherapy or in combination therapies for DLBCL treatment. Further work studying the clinical efficacy and combination treatment schedule should be performed for personalized therapy,” the researchers concluded.
The work was supported by grants from the Chinese government. The authors stated that they had no conflicts of interest.
SOURCE: Li X et al. Cancer Lett. 2020 Aug 14. doi: 10.1016/j.canlet.2020.07.032.
The STAT3 pathway is important to the development of many cancers, but so far, no Food and Drug Administration–approved drugs are ready for clinical use. Napabucasin, a novel oral small-molecule inhibitor of signal transducer and activator of transcription 3 (STAT3), blocks tumor growth and resists metastasis in a broad spectrum of solid tumors. Napabucasin was now found to be effective in inhibiting diffuse large beta cell lymphoma (DLBCL) in cell lines and in in vitro testing, as reported by Xue Li of the West China Hospital, Sichuan (China) University, and colleagues.
In addition, the effects of napabucasin were found to be synergistic with the use of doxorubicin, a standard DLBCL therapy agent, according to the report, published online in Cancer Letters.
‘Dramatic’ results
The researchers found that 34% (23/69) of DLBCL patients expressed STAT3 in tumor tissues. When they tested napabucasin in a variety of DLBCL cell lines they found that the drug exhibited potent cytotoxicity in a dose-dependent manner. In addition, they found that napabucasin induced intrinsic and extrinsic cell apoptosis, downregulated the expression of STAT3 target genes, including the antiapoptotic protein Mcl-1, and regulated the mitogen-activated protein kinase (MAPK) pathway, all important indicators of antitumor effectiveness in vitro.
In cells treated with napabucasin and doxorubicin alone and in combination, napabucasin alone significantly suppressed tumor growth, compared with that of the control (P < .01), achieving tumor growth inhibition (TGI) of 78.8%. The combination treatment, “with a dramatic TGI of 98.2%,” was more effective than doxorubicin monotherapy (TGI = 63.2%; P < .05), according to the researchers.
“Our study provided evidence that napabucasin is an attractive candidate drug either as a monotherapy or in combination therapies for DLBCL treatment. Further work studying the clinical efficacy and combination treatment schedule should be performed for personalized therapy,” the researchers concluded.
The work was supported by grants from the Chinese government. The authors stated that they had no conflicts of interest.
SOURCE: Li X et al. Cancer Lett. 2020 Aug 14. doi: 10.1016/j.canlet.2020.07.032.
The STAT3 pathway is important to the development of many cancers, but so far, no Food and Drug Administration–approved drugs are ready for clinical use. Napabucasin, a novel oral small-molecule inhibitor of signal transducer and activator of transcription 3 (STAT3), blocks tumor growth and resists metastasis in a broad spectrum of solid tumors. Napabucasin was now found to be effective in inhibiting diffuse large beta cell lymphoma (DLBCL) in cell lines and in in vitro testing, as reported by Xue Li of the West China Hospital, Sichuan (China) University, and colleagues.
In addition, the effects of napabucasin were found to be synergistic with the use of doxorubicin, a standard DLBCL therapy agent, according to the report, published online in Cancer Letters.
‘Dramatic’ results
The researchers found that 34% (23/69) of DLBCL patients expressed STAT3 in tumor tissues. When they tested napabucasin in a variety of DLBCL cell lines they found that the drug exhibited potent cytotoxicity in a dose-dependent manner. In addition, they found that napabucasin induced intrinsic and extrinsic cell apoptosis, downregulated the expression of STAT3 target genes, including the antiapoptotic protein Mcl-1, and regulated the mitogen-activated protein kinase (MAPK) pathway, all important indicators of antitumor effectiveness in vitro.
In cells treated with napabucasin and doxorubicin alone and in combination, napabucasin alone significantly suppressed tumor growth, compared with that of the control (P < .01), achieving tumor growth inhibition (TGI) of 78.8%. The combination treatment, “with a dramatic TGI of 98.2%,” was more effective than doxorubicin monotherapy (TGI = 63.2%; P < .05), according to the researchers.
“Our study provided evidence that napabucasin is an attractive candidate drug either as a monotherapy or in combination therapies for DLBCL treatment. Further work studying the clinical efficacy and combination treatment schedule should be performed for personalized therapy,” the researchers concluded.
The work was supported by grants from the Chinese government. The authors stated that they had no conflicts of interest.
SOURCE: Li X et al. Cancer Lett. 2020 Aug 14. doi: 10.1016/j.canlet.2020.07.032.
FROM CANCER LETTERS
IMPACT-AFib: Single mailing fails to budge oral anticoagulant uptake for AFib
A single educational mailing sent by several U.S. health plans to their patients with atrial fibrillation who were candidates for oral anticoagulation, but had not yet started a regimen, failed to boost them over their prescription hurdle and facilitate starting an antithrombotic regimen.
By 1 year following the intervention, a mere 10% of patients in both the intervention and a control arm of the randomized trial had begun treatment, with no signal of incremental uptake because of the mailing, Sean D. Pokorney, MD, said at the virtual annual congress of the European Society of Cardiology. Included in the mailing was an educational letter citing the patient’s atrial fibrillation (AFib) diagnosis, a statement regarding their suitability for oral anticoagulation, some information about the treatment, and a suggestion that recipients discuss this with their personal physician.
Dr. Pokorney acknowledged that the single mailing to patients may not have been adequate to capture patients’ attention and trigger an action, and that repeated messaging via multiple platforms and in coordination with interventions aimed at their health care providers may be what’s needed.
“It will take repeated interventions and engagements. We will need different methods to move the needle,” said Dr. Pokorney, a cardiac electrophysiologist at Duke University in Durham, N.C. The goal is to “empower patients to talk with their health care providers, and to become agents of change” in their care, he explained, but the single, mailed prod wasn’t enough.
An earlier study run by Dr. Pokorney and several of his colleagues used a broader panel of interventions aimed at both patients and clinicians to encourage increased prescribing of oral anticoagulants in five middle income countries, and documented successfully increasing the uptake rate by threefold compared with control patients (Lancet. 2017 Oct 14;390[10104]:1737-46). The current study tested the efficacy of a “much lower-impact intervention,” he admitted.
“The data are “sobering and eye-opening,” said Kalyanam Shivkumar, MD, a cardiac electrophysiologist and professor of medicine at the University of California, Los Angeles. “We’re stuck with this big challenge,” the gap between “what medicine can do and what it actually does” when evidence-based interventions fail to gain traction in everyday practice, he said in an interview.
The numbers collected during the new study highlighted the treatment gap. The IMPACT-AFib study randomized 23,546 patients with AFib and a CHA2DS2-VASc score of at least 2, denoting a stroke risk that warrants oral anticoagulation, to the intervention group, and 23,787 patients to the control arm. The patient selection process began with nearly 200,000 patients who met these criteria, but the researchers excluded 67% because they were already on an oral anticoagulant regimen, an uptake level that roughly matched the 50%-60% level usually seen among U.S. patients, Dr. Pokorney noted. That number coupled with the incremental uptake rate of only 10% of the enrolled patients during the trial, despite their uniform suitability for treatment, underscored how low uptake rates tend to remain stuck over time.
Enrolled patients averaged 78 years of age, with nearly two-thirds at least 75 years old, and with an average CHA2DS2-VASc score of 4.5.
The trial featured a novel design as the first clinical trial to take advantage of the Sentinel program for phase 4 data collection and study devised by the Food and Drug Administration, said Dr. Pokorney. The Sentinel program relies on data partners to provide information; for the IMPACT-AFib study, data came from five large U.S. health systems: Aetna, HealthCore, Humana, Harvard Pilgrim Healthcare, and Optum. Each of these systems sent the mailing to their targeted member patients.
In addition to sending just a single, mailed intervention, the study may have also been limited by the mailing’s content. The educational text, presented by Dr. Pokorney during his talk, focused largely on the potential risks of oral anticoagulation, the limited availability of antidote agents, potential drug and food interactions, and a brief entry about the risk for stroke associated with AFib along with a chart that a patient could use to hand calculate their CHA2DS2-VASc score. What the mailing lacked was discussion of the benefits of oral anticoagulation, noted study discussant Christophe LeClercq, MD, a cardiac electrophysiologist and professor of cardiology at the University of Rennes, France.
IMPACT-AFib received no commercial funding, and Dr. Pokorney and Dr. Shivkumar had no disclosures. Dr. Leclercq has received honoraria from Abbott, Biotronik, Boston Scientific, Livanova, and Medtronic.
A single educational mailing sent by several U.S. health plans to their patients with atrial fibrillation who were candidates for oral anticoagulation, but had not yet started a regimen, failed to boost them over their prescription hurdle and facilitate starting an antithrombotic regimen.
By 1 year following the intervention, a mere 10% of patients in both the intervention and a control arm of the randomized trial had begun treatment, with no signal of incremental uptake because of the mailing, Sean D. Pokorney, MD, said at the virtual annual congress of the European Society of Cardiology. Included in the mailing was an educational letter citing the patient’s atrial fibrillation (AFib) diagnosis, a statement regarding their suitability for oral anticoagulation, some information about the treatment, and a suggestion that recipients discuss this with their personal physician.
Dr. Pokorney acknowledged that the single mailing to patients may not have been adequate to capture patients’ attention and trigger an action, and that repeated messaging via multiple platforms and in coordination with interventions aimed at their health care providers may be what’s needed.
“It will take repeated interventions and engagements. We will need different methods to move the needle,” said Dr. Pokorney, a cardiac electrophysiologist at Duke University in Durham, N.C. The goal is to “empower patients to talk with their health care providers, and to become agents of change” in their care, he explained, but the single, mailed prod wasn’t enough.
An earlier study run by Dr. Pokorney and several of his colleagues used a broader panel of interventions aimed at both patients and clinicians to encourage increased prescribing of oral anticoagulants in five middle income countries, and documented successfully increasing the uptake rate by threefold compared with control patients (Lancet. 2017 Oct 14;390[10104]:1737-46). The current study tested the efficacy of a “much lower-impact intervention,” he admitted.
“The data are “sobering and eye-opening,” said Kalyanam Shivkumar, MD, a cardiac electrophysiologist and professor of medicine at the University of California, Los Angeles. “We’re stuck with this big challenge,” the gap between “what medicine can do and what it actually does” when evidence-based interventions fail to gain traction in everyday practice, he said in an interview.
The numbers collected during the new study highlighted the treatment gap. The IMPACT-AFib study randomized 23,546 patients with AFib and a CHA2DS2-VASc score of at least 2, denoting a stroke risk that warrants oral anticoagulation, to the intervention group, and 23,787 patients to the control arm. The patient selection process began with nearly 200,000 patients who met these criteria, but the researchers excluded 67% because they were already on an oral anticoagulant regimen, an uptake level that roughly matched the 50%-60% level usually seen among U.S. patients, Dr. Pokorney noted. That number coupled with the incremental uptake rate of only 10% of the enrolled patients during the trial, despite their uniform suitability for treatment, underscored how low uptake rates tend to remain stuck over time.
Enrolled patients averaged 78 years of age, with nearly two-thirds at least 75 years old, and with an average CHA2DS2-VASc score of 4.5.
The trial featured a novel design as the first clinical trial to take advantage of the Sentinel program for phase 4 data collection and study devised by the Food and Drug Administration, said Dr. Pokorney. The Sentinel program relies on data partners to provide information; for the IMPACT-AFib study, data came from five large U.S. health systems: Aetna, HealthCore, Humana, Harvard Pilgrim Healthcare, and Optum. Each of these systems sent the mailing to their targeted member patients.
In addition to sending just a single, mailed intervention, the study may have also been limited by the mailing’s content. The educational text, presented by Dr. Pokorney during his talk, focused largely on the potential risks of oral anticoagulation, the limited availability of antidote agents, potential drug and food interactions, and a brief entry about the risk for stroke associated with AFib along with a chart that a patient could use to hand calculate their CHA2DS2-VASc score. What the mailing lacked was discussion of the benefits of oral anticoagulation, noted study discussant Christophe LeClercq, MD, a cardiac electrophysiologist and professor of cardiology at the University of Rennes, France.
IMPACT-AFib received no commercial funding, and Dr. Pokorney and Dr. Shivkumar had no disclosures. Dr. Leclercq has received honoraria from Abbott, Biotronik, Boston Scientific, Livanova, and Medtronic.
A single educational mailing sent by several U.S. health plans to their patients with atrial fibrillation who were candidates for oral anticoagulation, but had not yet started a regimen, failed to boost them over their prescription hurdle and facilitate starting an antithrombotic regimen.
By 1 year following the intervention, a mere 10% of patients in both the intervention and a control arm of the randomized trial had begun treatment, with no signal of incremental uptake because of the mailing, Sean D. Pokorney, MD, said at the virtual annual congress of the European Society of Cardiology. Included in the mailing was an educational letter citing the patient’s atrial fibrillation (AFib) diagnosis, a statement regarding their suitability for oral anticoagulation, some information about the treatment, and a suggestion that recipients discuss this with their personal physician.
Dr. Pokorney acknowledged that the single mailing to patients may not have been adequate to capture patients’ attention and trigger an action, and that repeated messaging via multiple platforms and in coordination with interventions aimed at their health care providers may be what’s needed.
“It will take repeated interventions and engagements. We will need different methods to move the needle,” said Dr. Pokorney, a cardiac electrophysiologist at Duke University in Durham, N.C. The goal is to “empower patients to talk with their health care providers, and to become agents of change” in their care, he explained, but the single, mailed prod wasn’t enough.
An earlier study run by Dr. Pokorney and several of his colleagues used a broader panel of interventions aimed at both patients and clinicians to encourage increased prescribing of oral anticoagulants in five middle income countries, and documented successfully increasing the uptake rate by threefold compared with control patients (Lancet. 2017 Oct 14;390[10104]:1737-46). The current study tested the efficacy of a “much lower-impact intervention,” he admitted.
“The data are “sobering and eye-opening,” said Kalyanam Shivkumar, MD, a cardiac electrophysiologist and professor of medicine at the University of California, Los Angeles. “We’re stuck with this big challenge,” the gap between “what medicine can do and what it actually does” when evidence-based interventions fail to gain traction in everyday practice, he said in an interview.
The numbers collected during the new study highlighted the treatment gap. The IMPACT-AFib study randomized 23,546 patients with AFib and a CHA2DS2-VASc score of at least 2, denoting a stroke risk that warrants oral anticoagulation, to the intervention group, and 23,787 patients to the control arm. The patient selection process began with nearly 200,000 patients who met these criteria, but the researchers excluded 67% because they were already on an oral anticoagulant regimen, an uptake level that roughly matched the 50%-60% level usually seen among U.S. patients, Dr. Pokorney noted. That number coupled with the incremental uptake rate of only 10% of the enrolled patients during the trial, despite their uniform suitability for treatment, underscored how low uptake rates tend to remain stuck over time.
Enrolled patients averaged 78 years of age, with nearly two-thirds at least 75 years old, and with an average CHA2DS2-VASc score of 4.5.
The trial featured a novel design as the first clinical trial to take advantage of the Sentinel program for phase 4 data collection and study devised by the Food and Drug Administration, said Dr. Pokorney. The Sentinel program relies on data partners to provide information; for the IMPACT-AFib study, data came from five large U.S. health systems: Aetna, HealthCore, Humana, Harvard Pilgrim Healthcare, and Optum. Each of these systems sent the mailing to their targeted member patients.
In addition to sending just a single, mailed intervention, the study may have also been limited by the mailing’s content. The educational text, presented by Dr. Pokorney during his talk, focused largely on the potential risks of oral anticoagulation, the limited availability of antidote agents, potential drug and food interactions, and a brief entry about the risk for stroke associated with AFib along with a chart that a patient could use to hand calculate their CHA2DS2-VASc score. What the mailing lacked was discussion of the benefits of oral anticoagulation, noted study discussant Christophe LeClercq, MD, a cardiac electrophysiologist and professor of cardiology at the University of Rennes, France.
IMPACT-AFib received no commercial funding, and Dr. Pokorney and Dr. Shivkumar had no disclosures. Dr. Leclercq has received honoraria from Abbott, Biotronik, Boston Scientific, Livanova, and Medtronic.
FROM ESC CONGRESS 2020
Colorism can lead to intrafamily conflict
The color hue of a person’s skin is the most obvious criteria for society to judge a person and has always been deeply rooted in racism. Discrimination based on skin color is called colorism and is usually meted out by members of the same race and in the same family. The general belief is that someone with a lighter complexion is more beautiful, intelligent, or valuable than someone with a darker complexion. The term colorism can be widely applied in our assessment of conflict within families and society. The following case example gives guidance for psychiatrists faced with a family where colorism fuels family conflict.
Meeting the family
The Jaspers, a Black family, arrive at the psychiatrist’s office. They come in and look around before they choose their seats. Dr. Sally watches who sits next to whom and how they organize themselves in the office. After brief introductions, Mr. Jaspers begins, explaining why they are there.
“We are always fighting. We need this to stop. She, my wife, contradicts me all the time. Our kids are getting frustrated, and Bruce is acting out in school. He got in a fight again last week.”
Everyone looks at Bruce. He is darker skinned than the other siblings and carries all the African features in a family that favors the lighter end of the color spectrum. He sits next to his mother who leans into him. Mrs. Jaspers speaks next.
“Bruce gets picked on in school.”
Mr. Jaspers responds, “Well, if you didn’t run in there all the time and take him out, maybe he would learn how to deal with it better!”
“But they are mistreating him,” Mrs. Jaspers says.
The other children look away and play with their phones. Dr. Sally wonders whether this is a pattern: The parents fighting about how to deal with Bruce and his difficulties in the world – and the other siblings getting ignored and not included.
Dr. Sally asks Mrs. Jaspers for more details. She tells a narrative that is a strong thread in this family’s story.
“As you can see, Bruce is darker than the rest of our children. When we see the rest of our family, they all comment on what good skin and light coloring and good hair the other children have. Bruce just sits there. He is always being left out. He doesn’t speak up for himself. Maybe they think he can’t hear them, but I know he does and it affects him. They say the others are more intelligent, but I don’t think that is true. Bruce just gets picked on in school and he doesn’t feel like he matters. He doesn’t say anything, so maybe people think he doesn’t care, but I know he does.”
Dr. Sally turns to Bruce, who is still sitting silently next to his mother, his head down.
“Bruce, what do you have to say?”
Bruce shrugs his shoulders. His siblings still do not want to be drawn in and are otherwise occupied.
At this point, Dr. Sally might be thinking that she could see Bruce alone to assess his depression/self-esteem and maybe find ways to try to build him up. She does not want this to be an opportunity wasted. The goal is to work with the family to get Bruce where he needs to be faster and help the whole family.
Dr. Sally presses on. “Mr. Jaspers, what is your opinion?”
“She babies him. She treats him differently from the other kids. She is driving a wedge between him and his siblings. We fight about it all the time. She is driving a wedge between us, too.”
Mrs. Jaspers jumps in: “But you don’t know what it is like! When I was the only Black person in math class, I got picked on all the time! It made me self-conscious, and I couldn’t do my work. “
The other siblings look up briefly then back down at their devices. Dr. Sally asks them as a group:
“Can I ask you a question as a family? Can I ask the children a question?” They look up again. “Is this what goes on at home?” They all nod but offer no details.
Dr. Sally asks the oldest: “How does this affect your relationship with Bruce?”
They all look back and forth at each other. There is another long silence.
“See!” says Mr. Jaspers! “You can't protect him forever, and you are just ostracizing him from the rest of us! “
“But, but, he, he needs to learn different things. He is different. He faces different struggles. The police will stop him more. I am afraid for him.” Mrs. Jaspers starts to cry.
“You can’t protect him forever,” says her husband, gently reaching over to her.
Bruce has psychologically disappeared from the room, hiding behind his mother, who is now the largest and neediest presence in the room. Mr. Jaspers looks at Dr. Sally helplessly.
Dr. Sally asks the important question to the whole family.
“How do you all think this should work? If you don’t think Mrs. Jaspers is right, what do you think should be the way forward?”
This question is the turning point and indicates that Dr. Sally sees that the solution lies in how the family wants to manage things.
“I believe that your whole family has the answers, that you all have thought through this situation much more deeply and for much longer than I have. I am just hearing about it, and I am White and don’t have this experience. I have faith in your family, that with an opportunity to openly discuss this issue, that this knot can be unraveled. It does not mean that there are not more knots to unravel. For today, how to help you all help Bruce, is the work."
Dr. Sally talks to everyone but finishes up by looking at Mr. Jaspers, who has indicated that Bruce is part of the family and should not be treated differently from the other children.
Sean, the oldest sibling, now pipes up: “Bruce gets everything he wants. Mum spoils him; she always takes his side if there are arguments. Bruce knows this, and he steals our stuff because he knows he will get away with it.”
Bruce is quiet and leans in more to his mother. Dr. Sally motions to the mother not to speak.
“Is this true, Bruce?” Silence speaks that the answer is yes. The disparities in the family are aired for a while longer.
“Mrs. Jaspers, it is now your turn to respond.”
“Bruce is darker and faces more challenges than the others; he needs more protection and to know that he is loved.”
“Your family seems to think otherwise. They seem to think that your protection, while admirable, needs to be tempered to allow him to grow into a man who can stand on his own feet.”
Dr. Sally guides the family as a whole to a place where they can agree on the problem. The problem is now framed as a mother who cares too much and is too protective of Bruce but now her love and care need to be tempered. As a mother, she feels that it is her duty to protect her most vulnerable son. The family knows that Bruce will face more social scrutiny than the others, that he will have more internal struggles with self-worth than the others. How can the family help?
This conceptualization causes the family to look searchingly at one another. It is nothing they haven’t thought about privately, but this is the first time they are together thinking about it.
Dr. Sally says that she can help by providing time and space for them to wok through this together. They all agree to come back the following week with some thoughts about moving forward.
Offering perspective on colorism
In her book “Facing the Black Shadow,” couples and family therapist Marlene F. Watson, PhD, discusses colorism.
“African Americans still have a tough time talking about slavery – the origin of colorism. Seriously, what can we really expect to change without acknowledging and challenging the psychological residuals of slavery in our families and communities? What doesn’t get resolved in one generation is passed on to the next so our issues from slavery go from one generation to the next.”
Dr. Watson continues: “Confronting the secret about skin color in our families and communities is necessary for all Black girls to feel lovable, worthy, and deserving of care and for all Black boys to feel their value lies within them, not a dark, light, bright, near-white or White woman. African Americans need to get that preferring light over dark or dark over light is problematic for all of us. Skin color preferences in the African American community follow society’s racial hierarchy. African Americans as a group are at the bottom in the larger society and dark-skinned African Americans are at the bottom in the Black community.”
and patterns about skin color. Her advice is to ask each family member, from oldest to youngest, to identify the spoken and unspoken skin color beliefs he or she experiences in the family. Ask about skin color beliefs from outside that affect family members, and what each person thinks the family could do to stop promoting the “less than/better than” mentality that is often present with skin color assignment.
Thank you to Lynette Ramsingh Barros, who collaborated on creating the case.
Dr. Heru is professor of psychiatry at the University of Colorado Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest.
The color hue of a person’s skin is the most obvious criteria for society to judge a person and has always been deeply rooted in racism. Discrimination based on skin color is called colorism and is usually meted out by members of the same race and in the same family. The general belief is that someone with a lighter complexion is more beautiful, intelligent, or valuable than someone with a darker complexion. The term colorism can be widely applied in our assessment of conflict within families and society. The following case example gives guidance for psychiatrists faced with a family where colorism fuels family conflict.
Meeting the family
The Jaspers, a Black family, arrive at the psychiatrist’s office. They come in and look around before they choose their seats. Dr. Sally watches who sits next to whom and how they organize themselves in the office. After brief introductions, Mr. Jaspers begins, explaining why they are there.
“We are always fighting. We need this to stop. She, my wife, contradicts me all the time. Our kids are getting frustrated, and Bruce is acting out in school. He got in a fight again last week.”
Everyone looks at Bruce. He is darker skinned than the other siblings and carries all the African features in a family that favors the lighter end of the color spectrum. He sits next to his mother who leans into him. Mrs. Jaspers speaks next.
“Bruce gets picked on in school.”
Mr. Jaspers responds, “Well, if you didn’t run in there all the time and take him out, maybe he would learn how to deal with it better!”
“But they are mistreating him,” Mrs. Jaspers says.
The other children look away and play with their phones. Dr. Sally wonders whether this is a pattern: The parents fighting about how to deal with Bruce and his difficulties in the world – and the other siblings getting ignored and not included.
Dr. Sally asks Mrs. Jaspers for more details. She tells a narrative that is a strong thread in this family’s story.
“As you can see, Bruce is darker than the rest of our children. When we see the rest of our family, they all comment on what good skin and light coloring and good hair the other children have. Bruce just sits there. He is always being left out. He doesn’t speak up for himself. Maybe they think he can’t hear them, but I know he does and it affects him. They say the others are more intelligent, but I don’t think that is true. Bruce just gets picked on in school and he doesn’t feel like he matters. He doesn’t say anything, so maybe people think he doesn’t care, but I know he does.”
Dr. Sally turns to Bruce, who is still sitting silently next to his mother, his head down.
“Bruce, what do you have to say?”
Bruce shrugs his shoulders. His siblings still do not want to be drawn in and are otherwise occupied.
At this point, Dr. Sally might be thinking that she could see Bruce alone to assess his depression/self-esteem and maybe find ways to try to build him up. She does not want this to be an opportunity wasted. The goal is to work with the family to get Bruce where he needs to be faster and help the whole family.
Dr. Sally presses on. “Mr. Jaspers, what is your opinion?”
“She babies him. She treats him differently from the other kids. She is driving a wedge between him and his siblings. We fight about it all the time. She is driving a wedge between us, too.”
Mrs. Jaspers jumps in: “But you don’t know what it is like! When I was the only Black person in math class, I got picked on all the time! It made me self-conscious, and I couldn’t do my work. “
The other siblings look up briefly then back down at their devices. Dr. Sally asks them as a group:
“Can I ask you a question as a family? Can I ask the children a question?” They look up again. “Is this what goes on at home?” They all nod but offer no details.
Dr. Sally asks the oldest: “How does this affect your relationship with Bruce?”
They all look back and forth at each other. There is another long silence.
“See!” says Mr. Jaspers! “You can't protect him forever, and you are just ostracizing him from the rest of us! “
“But, but, he, he needs to learn different things. He is different. He faces different struggles. The police will stop him more. I am afraid for him.” Mrs. Jaspers starts to cry.
“You can’t protect him forever,” says her husband, gently reaching over to her.
Bruce has psychologically disappeared from the room, hiding behind his mother, who is now the largest and neediest presence in the room. Mr. Jaspers looks at Dr. Sally helplessly.
Dr. Sally asks the important question to the whole family.
“How do you all think this should work? If you don’t think Mrs. Jaspers is right, what do you think should be the way forward?”
This question is the turning point and indicates that Dr. Sally sees that the solution lies in how the family wants to manage things.
“I believe that your whole family has the answers, that you all have thought through this situation much more deeply and for much longer than I have. I am just hearing about it, and I am White and don’t have this experience. I have faith in your family, that with an opportunity to openly discuss this issue, that this knot can be unraveled. It does not mean that there are not more knots to unravel. For today, how to help you all help Bruce, is the work."
Dr. Sally talks to everyone but finishes up by looking at Mr. Jaspers, who has indicated that Bruce is part of the family and should not be treated differently from the other children.
Sean, the oldest sibling, now pipes up: “Bruce gets everything he wants. Mum spoils him; she always takes his side if there are arguments. Bruce knows this, and he steals our stuff because he knows he will get away with it.”
Bruce is quiet and leans in more to his mother. Dr. Sally motions to the mother not to speak.
“Is this true, Bruce?” Silence speaks that the answer is yes. The disparities in the family are aired for a while longer.
“Mrs. Jaspers, it is now your turn to respond.”
“Bruce is darker and faces more challenges than the others; he needs more protection and to know that he is loved.”
“Your family seems to think otherwise. They seem to think that your protection, while admirable, needs to be tempered to allow him to grow into a man who can stand on his own feet.”
Dr. Sally guides the family as a whole to a place where they can agree on the problem. The problem is now framed as a mother who cares too much and is too protective of Bruce but now her love and care need to be tempered. As a mother, she feels that it is her duty to protect her most vulnerable son. The family knows that Bruce will face more social scrutiny than the others, that he will have more internal struggles with self-worth than the others. How can the family help?
This conceptualization causes the family to look searchingly at one another. It is nothing they haven’t thought about privately, but this is the first time they are together thinking about it.
Dr. Sally says that she can help by providing time and space for them to wok through this together. They all agree to come back the following week with some thoughts about moving forward.
Offering perspective on colorism
In her book “Facing the Black Shadow,” couples and family therapist Marlene F. Watson, PhD, discusses colorism.
“African Americans still have a tough time talking about slavery – the origin of colorism. Seriously, what can we really expect to change without acknowledging and challenging the psychological residuals of slavery in our families and communities? What doesn’t get resolved in one generation is passed on to the next so our issues from slavery go from one generation to the next.”
Dr. Watson continues: “Confronting the secret about skin color in our families and communities is necessary for all Black girls to feel lovable, worthy, and deserving of care and for all Black boys to feel their value lies within them, not a dark, light, bright, near-white or White woman. African Americans need to get that preferring light over dark or dark over light is problematic for all of us. Skin color preferences in the African American community follow society’s racial hierarchy. African Americans as a group are at the bottom in the larger society and dark-skinned African Americans are at the bottom in the Black community.”
and patterns about skin color. Her advice is to ask each family member, from oldest to youngest, to identify the spoken and unspoken skin color beliefs he or she experiences in the family. Ask about skin color beliefs from outside that affect family members, and what each person thinks the family could do to stop promoting the “less than/better than” mentality that is often present with skin color assignment.
Thank you to Lynette Ramsingh Barros, who collaborated on creating the case.
Dr. Heru is professor of psychiatry at the University of Colorado Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest.
The color hue of a person’s skin is the most obvious criteria for society to judge a person and has always been deeply rooted in racism. Discrimination based on skin color is called colorism and is usually meted out by members of the same race and in the same family. The general belief is that someone with a lighter complexion is more beautiful, intelligent, or valuable than someone with a darker complexion. The term colorism can be widely applied in our assessment of conflict within families and society. The following case example gives guidance for psychiatrists faced with a family where colorism fuels family conflict.
Meeting the family
The Jaspers, a Black family, arrive at the psychiatrist’s office. They come in and look around before they choose their seats. Dr. Sally watches who sits next to whom and how they organize themselves in the office. After brief introductions, Mr. Jaspers begins, explaining why they are there.
“We are always fighting. We need this to stop. She, my wife, contradicts me all the time. Our kids are getting frustrated, and Bruce is acting out in school. He got in a fight again last week.”
Everyone looks at Bruce. He is darker skinned than the other siblings and carries all the African features in a family that favors the lighter end of the color spectrum. He sits next to his mother who leans into him. Mrs. Jaspers speaks next.
“Bruce gets picked on in school.”
Mr. Jaspers responds, “Well, if you didn’t run in there all the time and take him out, maybe he would learn how to deal with it better!”
“But they are mistreating him,” Mrs. Jaspers says.
The other children look away and play with their phones. Dr. Sally wonders whether this is a pattern: The parents fighting about how to deal with Bruce and his difficulties in the world – and the other siblings getting ignored and not included.
Dr. Sally asks Mrs. Jaspers for more details. She tells a narrative that is a strong thread in this family’s story.
“As you can see, Bruce is darker than the rest of our children. When we see the rest of our family, they all comment on what good skin and light coloring and good hair the other children have. Bruce just sits there. He is always being left out. He doesn’t speak up for himself. Maybe they think he can’t hear them, but I know he does and it affects him. They say the others are more intelligent, but I don’t think that is true. Bruce just gets picked on in school and he doesn’t feel like he matters. He doesn’t say anything, so maybe people think he doesn’t care, but I know he does.”
Dr. Sally turns to Bruce, who is still sitting silently next to his mother, his head down.
“Bruce, what do you have to say?”
Bruce shrugs his shoulders. His siblings still do not want to be drawn in and are otherwise occupied.
At this point, Dr. Sally might be thinking that she could see Bruce alone to assess his depression/self-esteem and maybe find ways to try to build him up. She does not want this to be an opportunity wasted. The goal is to work with the family to get Bruce where he needs to be faster and help the whole family.
Dr. Sally presses on. “Mr. Jaspers, what is your opinion?”
“She babies him. She treats him differently from the other kids. She is driving a wedge between him and his siblings. We fight about it all the time. She is driving a wedge between us, too.”
Mrs. Jaspers jumps in: “But you don’t know what it is like! When I was the only Black person in math class, I got picked on all the time! It made me self-conscious, and I couldn’t do my work. “
The other siblings look up briefly then back down at their devices. Dr. Sally asks them as a group:
“Can I ask you a question as a family? Can I ask the children a question?” They look up again. “Is this what goes on at home?” They all nod but offer no details.
Dr. Sally asks the oldest: “How does this affect your relationship with Bruce?”
They all look back and forth at each other. There is another long silence.
“See!” says Mr. Jaspers! “You can't protect him forever, and you are just ostracizing him from the rest of us! “
“But, but, he, he needs to learn different things. He is different. He faces different struggles. The police will stop him more. I am afraid for him.” Mrs. Jaspers starts to cry.
“You can’t protect him forever,” says her husband, gently reaching over to her.
Bruce has psychologically disappeared from the room, hiding behind his mother, who is now the largest and neediest presence in the room. Mr. Jaspers looks at Dr. Sally helplessly.
Dr. Sally asks the important question to the whole family.
“How do you all think this should work? If you don’t think Mrs. Jaspers is right, what do you think should be the way forward?”
This question is the turning point and indicates that Dr. Sally sees that the solution lies in how the family wants to manage things.
“I believe that your whole family has the answers, that you all have thought through this situation much more deeply and for much longer than I have. I am just hearing about it, and I am White and don’t have this experience. I have faith in your family, that with an opportunity to openly discuss this issue, that this knot can be unraveled. It does not mean that there are not more knots to unravel. For today, how to help you all help Bruce, is the work."
Dr. Sally talks to everyone but finishes up by looking at Mr. Jaspers, who has indicated that Bruce is part of the family and should not be treated differently from the other children.
Sean, the oldest sibling, now pipes up: “Bruce gets everything he wants. Mum spoils him; she always takes his side if there are arguments. Bruce knows this, and he steals our stuff because he knows he will get away with it.”
Bruce is quiet and leans in more to his mother. Dr. Sally motions to the mother not to speak.
“Is this true, Bruce?” Silence speaks that the answer is yes. The disparities in the family are aired for a while longer.
“Mrs. Jaspers, it is now your turn to respond.”
“Bruce is darker and faces more challenges than the others; he needs more protection and to know that he is loved.”
“Your family seems to think otherwise. They seem to think that your protection, while admirable, needs to be tempered to allow him to grow into a man who can stand on his own feet.”
Dr. Sally guides the family as a whole to a place where they can agree on the problem. The problem is now framed as a mother who cares too much and is too protective of Bruce but now her love and care need to be tempered. As a mother, she feels that it is her duty to protect her most vulnerable son. The family knows that Bruce will face more social scrutiny than the others, that he will have more internal struggles with self-worth than the others. How can the family help?
This conceptualization causes the family to look searchingly at one another. It is nothing they haven’t thought about privately, but this is the first time they are together thinking about it.
Dr. Sally says that she can help by providing time and space for them to wok through this together. They all agree to come back the following week with some thoughts about moving forward.
Offering perspective on colorism
In her book “Facing the Black Shadow,” couples and family therapist Marlene F. Watson, PhD, discusses colorism.
“African Americans still have a tough time talking about slavery – the origin of colorism. Seriously, what can we really expect to change without acknowledging and challenging the psychological residuals of slavery in our families and communities? What doesn’t get resolved in one generation is passed on to the next so our issues from slavery go from one generation to the next.”
Dr. Watson continues: “Confronting the secret about skin color in our families and communities is necessary for all Black girls to feel lovable, worthy, and deserving of care and for all Black boys to feel their value lies within them, not a dark, light, bright, near-white or White woman. African Americans need to get that preferring light over dark or dark over light is problematic for all of us. Skin color preferences in the African American community follow society’s racial hierarchy. African Americans as a group are at the bottom in the larger society and dark-skinned African Americans are at the bottom in the Black community.”
and patterns about skin color. Her advice is to ask each family member, from oldest to youngest, to identify the spoken and unspoken skin color beliefs he or she experiences in the family. Ask about skin color beliefs from outside that affect family members, and what each person thinks the family could do to stop promoting the “less than/better than” mentality that is often present with skin color assignment.
Thank you to Lynette Ramsingh Barros, who collaborated on creating the case.
Dr. Heru is professor of psychiatry at the University of Colorado Denver, Aurora. She is editor of “Working With Families in Medical Settings: A Multidisciplinary Guide for Psychiatrists and Other Health Professionals” (New York: Routledge, 2013). She has no conflicts of interest.
REALITY trial supports restrictive transfusion in anemic MI
in the landmark REALITY trial.
Randomized trial data already support a restrictive transfusion strategy in patients undergoing cardiac and noncardiac surgery, as well as in other settings. Those trials deliberately excluded patients with acute myocardial ischemia.
Cardiologists have been loath to adopt a restrictive strategy in the absence of persuasive supporting evidence because of a theoretic concern that low hemoglobin might be particularly harmful to ischemic myocardium. Anemia occurs in 5%-10% patients with MI, and clinicians have been eager for evidence-based guidance on how to best manage it.
“Blood is a precious resource and transfusion is costly, logistically cumbersome, and has side effects,” Philippe Gabriel Steg, MD, chair of the REALITY trial, noted in presenting the study results at the virtual annual congress of the European Society of Cardiology.
REALITY was the first-ever large randomized trial of a restrictive versus liberal transfusion strategy in acute MI. The study, which featured a noninferiority design, included 668 stable patients with acute MI and anemia with a hemoglobin of 7-10 g/dL at 35 hospitals in France and Spain. Participants were randomized to a restrictive strategy in which transfusion was withheld unless the hemoglobin dropped to 8 g/dL or less, or to a conventional liberal strategy triggered by a hemoglobin of 10 g/dL or lower. The transfusion target was a hemoglobin level of 8-10 g/dL in the restrictive strategy group and greater than 11 g/dL in the liberal transfusion group. In the restrictive transfusion group, 36% received at least one RBC transfusion, as did 87% in the liberal transfusion study arm. The restrictive strategy group used 414 fewer units of blood.
The two coprimary endpoints were 30-day major adverse cardiovascular events and cost-effectiveness. The 30-day composite of all-cause mortality, reinfarction, stroke, and emergency percutaneous coronary intervention for myocardial ischemia occurred in 11% of the restrictive transfusion group and 14% of the liberal transfusion group. The resultant 21% relative risk reduction established that the restrictive strategy was noninferior. Of note, all of the individual components of the composite endpoint numerically favored the restrictive approach.
In terms of safety, patients in the restrictive transfusion group were significantly less likely to develop an infection, by a margin of 0% versus 1.5%. The rate of acute lung injury was also significantly lower in the restrictive group: 0.3%, compared with 2.2%. The median hospital length of stay was identical at 7 days in both groups.
The cost-effectiveness analysis concluded that the restrictive transfusion strategy had an 84% probability of being both less expensive and more effective.
Patients were enrolled in REALITY regardless of whether they had active bleeding, as long as the bleeding wasn’t deemed massive and life-threatening. Notably, there was no difference in the results of restrictive versus liberal transfusion regardless of whether active bleeding was present, nor did baseline hemoglobin or the presence or absence of preexisting anemia affect the results.
Dr. Steg noted that a much larger randomized trial of restrictive versus liberal transfusion in the setting of acute MI with anemia is underway in the United States and Canada. The 3,000-patient MINT trial, sponsored by the National Institutes of Health, is testing the superiority of restrictive transfusion, rather than its noninferiority, as in REALITY. Results are a couple of years away.
“I think that will be an important piece of additional evidence,” he said.
Discussant Marco Roffi, MD, didn’t mince words.
“I really love the REALITY trial,” declared Dr. Roffi, professor and vice chairman of the cardiology department and director of the interventional cardiology unit at University Hospital of Geneva.
He ticked off a series of reasons: The trial addressed a common clinical dilemma about which there has been essentially no prior high-quality evidence, it provided convincing results, and it carried important implications for responsible stewardship of the blood supply.
“REALITY allows clinicians to comfortably refrain from transfusing anemic patients presenting with myocardial infarction, and this should lead to a reduction in the consumption of blood products,” Dr. Roffi said.
He applauded the investigators for their success in obtaining public funding for a study lacking a commercial hook. And as a clinical investigator, he was particularly impressed by one of the technical details about the REALITY trial: “I was amazed by the fact that the observed event rates virtually corresponded to the estimated ones used for the power calculations. This is rarely the case in such a trial.”
Dr. Roffi said the REALITY findings should have an immediate impact on clinical practice, as well as on the brand new 2020 ESC guidelines on the management of non–ST-elevation ACS issued during the ESC virtual congress.
The freshly inked guidelines state: “Based on inconsistent study results and the lack of adequately powered randomized, controlled trials, a restrictive policy of transfusion in anemic patients with MI may be considered.” As of today, Dr. Roffi argued, the phrase “may be considered” ought to be replaced by the stronger phrase “should be considered.”
During the discussion period, he was asked if it’s appropriate to extrapolate the REALITY results to patients undergoing transcatheter aortic valve replacement, among whom anemia is highly prevalent.
“I think this is a different patient population. Nevertheless, the concept of being restrictive is one that in my opinion now remains until proven otherwise. So we are being very restrictive in these patients,” he replied.
Asked about possible mechanisms by which liberal transfusion might have detrimental effects in acute MI patients, Dr. Steg cited several, including evidence that transfusion may not improve oxygen delivery to as great an extent as traditionally thought. There is also the risk of volume overload, increased blood viscosity, and enhanced platelet aggregation and activation, which could promote myocardial ischemia.
The REALITY trial was funded by the French Ministry of Health and the Spanish Ministry of Economy and Competitiveness with no commercial support. Outside the scope of the trial, Dr. Steg reported receiving research grants from Bayer, Merck, Servier, and Sanofi as well as serving as a consultant to numerous pharmaceutical companies.
in the landmark REALITY trial.
Randomized trial data already support a restrictive transfusion strategy in patients undergoing cardiac and noncardiac surgery, as well as in other settings. Those trials deliberately excluded patients with acute myocardial ischemia.
Cardiologists have been loath to adopt a restrictive strategy in the absence of persuasive supporting evidence because of a theoretic concern that low hemoglobin might be particularly harmful to ischemic myocardium. Anemia occurs in 5%-10% patients with MI, and clinicians have been eager for evidence-based guidance on how to best manage it.
“Blood is a precious resource and transfusion is costly, logistically cumbersome, and has side effects,” Philippe Gabriel Steg, MD, chair of the REALITY trial, noted in presenting the study results at the virtual annual congress of the European Society of Cardiology.
REALITY was the first-ever large randomized trial of a restrictive versus liberal transfusion strategy in acute MI. The study, which featured a noninferiority design, included 668 stable patients with acute MI and anemia with a hemoglobin of 7-10 g/dL at 35 hospitals in France and Spain. Participants were randomized to a restrictive strategy in which transfusion was withheld unless the hemoglobin dropped to 8 g/dL or less, or to a conventional liberal strategy triggered by a hemoglobin of 10 g/dL or lower. The transfusion target was a hemoglobin level of 8-10 g/dL in the restrictive strategy group and greater than 11 g/dL in the liberal transfusion group. In the restrictive transfusion group, 36% received at least one RBC transfusion, as did 87% in the liberal transfusion study arm. The restrictive strategy group used 414 fewer units of blood.
The two coprimary endpoints were 30-day major adverse cardiovascular events and cost-effectiveness. The 30-day composite of all-cause mortality, reinfarction, stroke, and emergency percutaneous coronary intervention for myocardial ischemia occurred in 11% of the restrictive transfusion group and 14% of the liberal transfusion group. The resultant 21% relative risk reduction established that the restrictive strategy was noninferior. Of note, all of the individual components of the composite endpoint numerically favored the restrictive approach.
In terms of safety, patients in the restrictive transfusion group were significantly less likely to develop an infection, by a margin of 0% versus 1.5%. The rate of acute lung injury was also significantly lower in the restrictive group: 0.3%, compared with 2.2%. The median hospital length of stay was identical at 7 days in both groups.
The cost-effectiveness analysis concluded that the restrictive transfusion strategy had an 84% probability of being both less expensive and more effective.
Patients were enrolled in REALITY regardless of whether they had active bleeding, as long as the bleeding wasn’t deemed massive and life-threatening. Notably, there was no difference in the results of restrictive versus liberal transfusion regardless of whether active bleeding was present, nor did baseline hemoglobin or the presence or absence of preexisting anemia affect the results.
Dr. Steg noted that a much larger randomized trial of restrictive versus liberal transfusion in the setting of acute MI with anemia is underway in the United States and Canada. The 3,000-patient MINT trial, sponsored by the National Institutes of Health, is testing the superiority of restrictive transfusion, rather than its noninferiority, as in REALITY. Results are a couple of years away.
“I think that will be an important piece of additional evidence,” he said.
Discussant Marco Roffi, MD, didn’t mince words.
“I really love the REALITY trial,” declared Dr. Roffi, professor and vice chairman of the cardiology department and director of the interventional cardiology unit at University Hospital of Geneva.
He ticked off a series of reasons: The trial addressed a common clinical dilemma about which there has been essentially no prior high-quality evidence, it provided convincing results, and it carried important implications for responsible stewardship of the blood supply.
“REALITY allows clinicians to comfortably refrain from transfusing anemic patients presenting with myocardial infarction, and this should lead to a reduction in the consumption of blood products,” Dr. Roffi said.
He applauded the investigators for their success in obtaining public funding for a study lacking a commercial hook. And as a clinical investigator, he was particularly impressed by one of the technical details about the REALITY trial: “I was amazed by the fact that the observed event rates virtually corresponded to the estimated ones used for the power calculations. This is rarely the case in such a trial.”
Dr. Roffi said the REALITY findings should have an immediate impact on clinical practice, as well as on the brand new 2020 ESC guidelines on the management of non–ST-elevation ACS issued during the ESC virtual congress.
The freshly inked guidelines state: “Based on inconsistent study results and the lack of adequately powered randomized, controlled trials, a restrictive policy of transfusion in anemic patients with MI may be considered.” As of today, Dr. Roffi argued, the phrase “may be considered” ought to be replaced by the stronger phrase “should be considered.”
During the discussion period, he was asked if it’s appropriate to extrapolate the REALITY results to patients undergoing transcatheter aortic valve replacement, among whom anemia is highly prevalent.
“I think this is a different patient population. Nevertheless, the concept of being restrictive is one that in my opinion now remains until proven otherwise. So we are being very restrictive in these patients,” he replied.
Asked about possible mechanisms by which liberal transfusion might have detrimental effects in acute MI patients, Dr. Steg cited several, including evidence that transfusion may not improve oxygen delivery to as great an extent as traditionally thought. There is also the risk of volume overload, increased blood viscosity, and enhanced platelet aggregation and activation, which could promote myocardial ischemia.
The REALITY trial was funded by the French Ministry of Health and the Spanish Ministry of Economy and Competitiveness with no commercial support. Outside the scope of the trial, Dr. Steg reported receiving research grants from Bayer, Merck, Servier, and Sanofi as well as serving as a consultant to numerous pharmaceutical companies.
in the landmark REALITY trial.
Randomized trial data already support a restrictive transfusion strategy in patients undergoing cardiac and noncardiac surgery, as well as in other settings. Those trials deliberately excluded patients with acute myocardial ischemia.
Cardiologists have been loath to adopt a restrictive strategy in the absence of persuasive supporting evidence because of a theoretic concern that low hemoglobin might be particularly harmful to ischemic myocardium. Anemia occurs in 5%-10% patients with MI, and clinicians have been eager for evidence-based guidance on how to best manage it.
“Blood is a precious resource and transfusion is costly, logistically cumbersome, and has side effects,” Philippe Gabriel Steg, MD, chair of the REALITY trial, noted in presenting the study results at the virtual annual congress of the European Society of Cardiology.
REALITY was the first-ever large randomized trial of a restrictive versus liberal transfusion strategy in acute MI. The study, which featured a noninferiority design, included 668 stable patients with acute MI and anemia with a hemoglobin of 7-10 g/dL at 35 hospitals in France and Spain. Participants were randomized to a restrictive strategy in which transfusion was withheld unless the hemoglobin dropped to 8 g/dL or less, or to a conventional liberal strategy triggered by a hemoglobin of 10 g/dL or lower. The transfusion target was a hemoglobin level of 8-10 g/dL in the restrictive strategy group and greater than 11 g/dL in the liberal transfusion group. In the restrictive transfusion group, 36% received at least one RBC transfusion, as did 87% in the liberal transfusion study arm. The restrictive strategy group used 414 fewer units of blood.
The two coprimary endpoints were 30-day major adverse cardiovascular events and cost-effectiveness. The 30-day composite of all-cause mortality, reinfarction, stroke, and emergency percutaneous coronary intervention for myocardial ischemia occurred in 11% of the restrictive transfusion group and 14% of the liberal transfusion group. The resultant 21% relative risk reduction established that the restrictive strategy was noninferior. Of note, all of the individual components of the composite endpoint numerically favored the restrictive approach.
In terms of safety, patients in the restrictive transfusion group were significantly less likely to develop an infection, by a margin of 0% versus 1.5%. The rate of acute lung injury was also significantly lower in the restrictive group: 0.3%, compared with 2.2%. The median hospital length of stay was identical at 7 days in both groups.
The cost-effectiveness analysis concluded that the restrictive transfusion strategy had an 84% probability of being both less expensive and more effective.
Patients were enrolled in REALITY regardless of whether they had active bleeding, as long as the bleeding wasn’t deemed massive and life-threatening. Notably, there was no difference in the results of restrictive versus liberal transfusion regardless of whether active bleeding was present, nor did baseline hemoglobin or the presence or absence of preexisting anemia affect the results.
Dr. Steg noted that a much larger randomized trial of restrictive versus liberal transfusion in the setting of acute MI with anemia is underway in the United States and Canada. The 3,000-patient MINT trial, sponsored by the National Institutes of Health, is testing the superiority of restrictive transfusion, rather than its noninferiority, as in REALITY. Results are a couple of years away.
“I think that will be an important piece of additional evidence,” he said.
Discussant Marco Roffi, MD, didn’t mince words.
“I really love the REALITY trial,” declared Dr. Roffi, professor and vice chairman of the cardiology department and director of the interventional cardiology unit at University Hospital of Geneva.
He ticked off a series of reasons: The trial addressed a common clinical dilemma about which there has been essentially no prior high-quality evidence, it provided convincing results, and it carried important implications for responsible stewardship of the blood supply.
“REALITY allows clinicians to comfortably refrain from transfusing anemic patients presenting with myocardial infarction, and this should lead to a reduction in the consumption of blood products,” Dr. Roffi said.
He applauded the investigators for their success in obtaining public funding for a study lacking a commercial hook. And as a clinical investigator, he was particularly impressed by one of the technical details about the REALITY trial: “I was amazed by the fact that the observed event rates virtually corresponded to the estimated ones used for the power calculations. This is rarely the case in such a trial.”
Dr. Roffi said the REALITY findings should have an immediate impact on clinical practice, as well as on the brand new 2020 ESC guidelines on the management of non–ST-elevation ACS issued during the ESC virtual congress.
The freshly inked guidelines state: “Based on inconsistent study results and the lack of adequately powered randomized, controlled trials, a restrictive policy of transfusion in anemic patients with MI may be considered.” As of today, Dr. Roffi argued, the phrase “may be considered” ought to be replaced by the stronger phrase “should be considered.”
During the discussion period, he was asked if it’s appropriate to extrapolate the REALITY results to patients undergoing transcatheter aortic valve replacement, among whom anemia is highly prevalent.
“I think this is a different patient population. Nevertheless, the concept of being restrictive is one that in my opinion now remains until proven otherwise. So we are being very restrictive in these patients,” he replied.
Asked about possible mechanisms by which liberal transfusion might have detrimental effects in acute MI patients, Dr. Steg cited several, including evidence that transfusion may not improve oxygen delivery to as great an extent as traditionally thought. There is also the risk of volume overload, increased blood viscosity, and enhanced platelet aggregation and activation, which could promote myocardial ischemia.
The REALITY trial was funded by the French Ministry of Health and the Spanish Ministry of Economy and Competitiveness with no commercial support. Outside the scope of the trial, Dr. Steg reported receiving research grants from Bayer, Merck, Servier, and Sanofi as well as serving as a consultant to numerous pharmaceutical companies.
REPORTING FROM ESC CONGRESS 2020
Latest report adds almost 44,000 child COVID-19 cases in 1 week
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
The new cases bring the cumulative number of infected children to over 476,000, and that figure represents 9.5% of the over 5 million COVID-19 cases reported among all ages, the AAP and the CHA said in their weekly report. The cumulative number of children covers 49 states (New York is not reporting age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
From lowest to highest, the states occupying opposite ends of the cumulative proportion spectrum are New Jersey at 3.4% – New York City was lower with a 3.2% figure but is not a state – and Wyoming at 18.3%, the report showed.
Children represent more than 15% of all reported COVID-19 cases in five other states: Tennessee (17.1%), North Dakota (16.0%), Alaska (15.9%), New Mexico (15.7%), and Minnesota (15.1%). The states just above New Jersey are Florida (5.8%), Connecticut (5.9%), and Massachusetts (6.7%). Texas has a rate of 5.6% but has reported age for only 8% of confirmed cases, the AAP and CHA noted.
Children make up a much lower share of COVID-19 hospitalizations – 1.7% of the cumulative number for all ages – although that figure has been slowly rising over the course of the pandemic: it was 1.2% on July 9 and 0.9% on May 8. Arizona (4.1%) is the highest of the 22 states reporting age for hospitalizations and Hawaii (0.6%) is the lowest, based on the AAP/CHA data.
Mortality figures for children continue to be even lower. Nationwide, 0.07% of all COVID-19 deaths occurred in children, and 19 of the 43 states reporting age distributions have had no deaths yet. Pediatric deaths totaled 101 as of Aug. 27, the two groups reported.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The new cases bring the cumulative number of infected children to over 476,000, and that figure represents 9.5% of the over 5 million COVID-19 cases reported among all ages, the AAP and the CHA said in their weekly report. The cumulative number of children covers 49 states (New York is not reporting age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
From lowest to highest, the states occupying opposite ends of the cumulative proportion spectrum are New Jersey at 3.4% – New York City was lower with a 3.2% figure but is not a state – and Wyoming at 18.3%, the report showed.
Children represent more than 15% of all reported COVID-19 cases in five other states: Tennessee (17.1%), North Dakota (16.0%), Alaska (15.9%), New Mexico (15.7%), and Minnesota (15.1%). The states just above New Jersey are Florida (5.8%), Connecticut (5.9%), and Massachusetts (6.7%). Texas has a rate of 5.6% but has reported age for only 8% of confirmed cases, the AAP and CHA noted.
Children make up a much lower share of COVID-19 hospitalizations – 1.7% of the cumulative number for all ages – although that figure has been slowly rising over the course of the pandemic: it was 1.2% on July 9 and 0.9% on May 8. Arizona (4.1%) is the highest of the 22 states reporting age for hospitalizations and Hawaii (0.6%) is the lowest, based on the AAP/CHA data.
Mortality figures for children continue to be even lower. Nationwide, 0.07% of all COVID-19 deaths occurred in children, and 19 of the 43 states reporting age distributions have had no deaths yet. Pediatric deaths totaled 101 as of Aug. 27, the two groups reported.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The new cases bring the cumulative number of infected children to over 476,000, and that figure represents 9.5% of the over 5 million COVID-19 cases reported among all ages, the AAP and the CHA said in their weekly report. The cumulative number of children covers 49 states (New York is not reporting age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
From lowest to highest, the states occupying opposite ends of the cumulative proportion spectrum are New Jersey at 3.4% – New York City was lower with a 3.2% figure but is not a state – and Wyoming at 18.3%, the report showed.
Children represent more than 15% of all reported COVID-19 cases in five other states: Tennessee (17.1%), North Dakota (16.0%), Alaska (15.9%), New Mexico (15.7%), and Minnesota (15.1%). The states just above New Jersey are Florida (5.8%), Connecticut (5.9%), and Massachusetts (6.7%). Texas has a rate of 5.6% but has reported age for only 8% of confirmed cases, the AAP and CHA noted.
Children make up a much lower share of COVID-19 hospitalizations – 1.7% of the cumulative number for all ages – although that figure has been slowly rising over the course of the pandemic: it was 1.2% on July 9 and 0.9% on May 8. Arizona (4.1%) is the highest of the 22 states reporting age for hospitalizations and Hawaii (0.6%) is the lowest, based on the AAP/CHA data.
Mortality figures for children continue to be even lower. Nationwide, 0.07% of all COVID-19 deaths occurred in children, and 19 of the 43 states reporting age distributions have had no deaths yet. Pediatric deaths totaled 101 as of Aug. 27, the two groups reported.
First randomized trial reassures on ACEIs, ARBs in COVID-19
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.