User login
Osteoporosis management: Use a goal-oriented, individualized approach
Recommendations for care are evolving, with increasingly sophisticated screening and diagnostic tools and a broadening array of treatment options.
As the population of older adults rises, primary osteoporosis has become a problem of public health significance, resulting in more than 2 million fractures and $19 billion in related costs annually in the United States.1 Despite the availability of effective primary and secondary preventive measures, many older adults do not receive adequate information on bone health from their primary care provider.2 Initiation of osteoporosis treatment is low even among patients who have had an osteoporotic fracture: Fewer than one-quarter of older adults with hip fracture have begun taking osteoporosis medication within 12 months of hospital discharge.3
In this overview of osteoporosis care, we provide information on how to evaluate and manage older adults in primary care settings who are at risk of, or have been given a diagnosis of, primary osteoporosis. The guidance that we offer reflects the most recent updates and recommendations by relevant professional societies.1,4-7
The nature and scope of an urgent problem
Osteoporosis is a skeletal disorder characterized by low bone mass and deterioration of bone structure that causes bone fragility and increases the risk of fracture.8 Operationally, it is defined by the World Health Organization as a bone mineral density (BMD) score below 2.5 SD from the mean value for a young White woman (ie, T-score ≤ –2.5).9 Primary osteoporosis is age related and occurs mostly in postmenopausal women and older men, affecting 25% of women and 5% of men ≥ 65 years.10
An osteoporotic fracture is particularly devastating in an older adult because it can cause pain, reduced mobility, depression, and social isolation and can increase the risk of related mortality.1 The National Osteoporosis Foundation estimates that 20% of older adults who sustain a hip fracture die within 1 year due to complications of the fracture itself or surgical repair.1 Therefore, it is of paramount importance to identify patients who are at increased risk of fracture and intervene early.
Clinical manifestations
Osteoporosis does not have a primary presentation; rather, disease manifests clinically when a patient develops complications. Often, a fragility fracture is the first sign in an older person.11
A fracture is the most important complication of osteoporosis and can result from low-trauma injury or a fall from standing height—thus, the term “fragility fracture.” Osteoporotic fractures commonly involve the vertebra, hip, and wrist. Hip and extremity fractures can result in limited or lost mobility and depression. Vertebral fractures can be asymptomatic or result in kyphosis and loss of height. Fractures can give rise to pain.
Age and female sexare risk factors
TABLE 11,6,10 lists risk factors associated with osteoporosis. Age is the most important; prevalence of osteoporosis increases with age. Other nonmodifiable risk factors include female sex (the disease appears earlier in women who enter menopause prematurely), family history of osteoporosis, and race and ethnicity. Twenty percent of Asian and non-Hispanic White women > 50 years have osteoporosis.1 A study showed that Mexican Americans are at higher risk of osteoporosis than non-Hispanic Whites; non-Hispanic Blacks are least affected.10
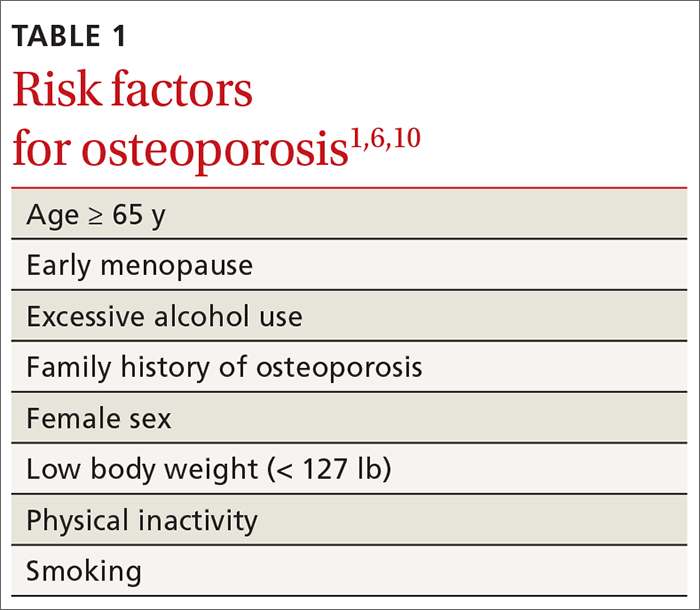
Other risk factors include low body weight (< 127 lb) and a history of fractures after age 50. Behavioral risk factors include smoking, excessive alcohol intake (> 3 drinks/d), poor nutrition, and a sedentary lifestyle.1,6
Continue to: Who should be screened?...
Who should be screened?
Screening is generally performed with a clinical evaluation and a dual-energy x-ray absorptiometry (DXA) scan of BMD. Measurement of BMD is generally recommended for screening all women ≥ 65 years and those < 65 years whose 10-year risk of fracture is equivalent to that of a 65-year-old White woman (see “Assessment of fracture risk” later in the article). For men, the US Preventive Services Task Force recommends screening those with a prior fracture or a secondary risk factor for disease.5 However, the National Osteoporosis Foundation recommends screening all men ≥ 70 years and those 50 to 69 years whose risk profile shows heightened risk.1,4
DXA of the spine and hip is preferred; the distal one-third of the radius (termed “33% radius”) of the nondominant arm can be used when spine and hip BMD cannot be interpreted because of bone changes from the disease process or artifacts, or in certain diseases in which the wrist region shows the earliest change (eg, primary hyperparathyroidism).6,7
Clinical evaluation includes a detailed history, physical examination, laboratory screening, and assessment for risk of fracture.
❚ History. Explore the presence of risk factors, including fractures in adulthood, falls, medication use, alcohol and tobacco use, family history of osteoporosis, and chronic disease.6,7
❚ Physical exam. Assess height, including any loss (> 1.5 in) since the patient’s second or third decade of life; kyphosis; frailty; and balance and mobility problems.4,6,7
❚ Laboratory and imaging studies. Perform basic laboratory testing when DXA is abnormal, including thyroid function, serum calcium, and renal function.6,12 Radiography of the lateral spine might be necessary, especially when there is kyphosis or loss of height. Assess for vertebral fracture, using lateral spine radiography, when vertebral involvement is suspected.6,7
❚ Assessment of fracture risk. Fracture risk can be assessed with any of a number of tools, including:
- Simplified Calculated Osteoporosis Risk Estimation (SCORE): www.medicalalgorithms.com/simplified-calculated-osteoporosis-risk-estimation-tool
- Osteoporosis Risk Assessment Instrument (ORAI): www.physio-pedia.com/The_Osteoporosis_Risk_Assessment_Instrument_(ORAI)
- Osteoporosis Index of Risk (OSIRIS): https://www.tandfonline.com/doi/abs/10.1080/gye.16.3.245.250?journalCode=igye20
- Osteoporosis Self-Assessment Tool (OST): www.ncbi.nlm.nih.gov/books/NBK45516/figure/ch10.f2/
- FRAX tool5: www.sheffield.ac.uk/FRAX.
The FRAX tool is widely used. It assesses a patient’s 10-year risk of fracture.
Diagnosis is based on these criteria
Diagnosis of osteoporosis is based on any 1 or more of the following criteria6:
- a history of fragility fracture not explained by metabolic bone disease
- T-score ≤ –2.5 (lumbar, hip, femoral neck, or 33% radius)
- a nation-specific FRAX score (in the absence of access to DXA).
❚ Secondary disease. Patients in whom secondary osteoporosis is suspected should undergo laboratory investigation to ascertain the cause; treatment of the underlying pathology might then be required. Evaluation for a secondary cause might include a complete blood count, comprehensive metabolic panel, protein electrophoresis and urinary protein electrophoresis (to rule out myeloproliferative and hematologic diseases), and tests of serum 25-hydroxyvitamin D, parathyroid hormone, serum calcium, alkaline phosphatase, 24-hour urinary calcium, sodium, and creatinine.6,7 Specialized testing for biochemical markers of bone turnover—so-called bone-turnover markers—can be considered as part of the initial evaluation and follow-up, although the tests are not recommended by the US Preventive Services Task Force (see “Monitoring the efficacy of treatment,” later in the article, for more information about these markers).6
Although BMD by DXA remains the gold standard in screening for and diagnosing osteoporosis, a high rate of fracture is seen in patients with certain diseases, such as type 2 diabetes and ankylosing spondylitis, who have a nonosteoporotic low T-score. This raises concerns about the usefulness of BMD for diagnosing osteoporosis in patients who have one of these diseases.13-16
❚
❚ Trabecular bone score (TBS), a surrogate bone-quality measure that is calculated based on the spine DXA image, has recently been introduced in clinical practice, and can be used to predict fracture risk in conjunction with BMD assessment by DXA and the FRAX score.17 TBS provides an indirect index of the trabecular microarchitecture using pixel gray-level variation in lumbar spine DXA images.18 Three categories of TBS (≤ 1.200, degraded microarchitecture; 1.200-1.350, partially degraded microarchitecture; and > 1.350, normal microarchitecture) have been reported to correspond with a T-score of, respectively, ≤ −2.5; −2.5 to −1.0; and > −1.0.18 TBS can be used only in patients with a body mass index of 15 to 37.5.19,20
There is no recommendation for monitoring bone quality using TBS after osteoporosis treatment. Such monitoring is at the clinician’s discretion for appropriate patients who might not show a risk of fracture, based on BMD measurement.
Continue to: Putting preventive measures into practice...
Putting preventive measures into practice
Measures to prevent osteoporosis and preserve bone health (TABLE 21,6) are best started in childhood but can be initiated at any age and maintained through the lifespan. Encourage older adults to adopt dietary and behavioral strategies to improve their bone health and prevent fracture. We recommend the following strategies; take each patient’s individual situation into consideration when electing to adopt any of these measures.

❚ Vitamin D. Consider checking the serum 25-hydroxyvitamin D level and providing supplementation (800-1000 IU daily, the National Osteoporosis Foundation recommends1) as necessary to maintain the level at 30-50 ng/mL.6
❚ Calcium. Encourage a daily dietary calcium intake of 1000-1200 mg. Supplement calcium if you determine that diet does not provide an adequate amount.
❚ Alcohol. Advise patients to limit consumption to < 3 drinks a day.
❚ Tobacco. Advise smoking cessation.
❚ Activity. Encourage an active lifestyle, including regular weight-bearing and balance exercises and resistance exercises such as Pilates, weightlifting, and tai chi. The regimen should be tailored to the patient’s individual situation.
❚ Medical therapy for concomitant illness. When possible, prescribe medications for chronic comorbidities that can also benefit bone health. For example, long-term use of angiotensin-converting enzyme (ACE) inhibitors and thiazide diuretics for hypertension are associated with a slower decline in BMD in some populations.21-23
Tailor treatment to patient’s circumstances
TABLE 34,6,24 describes indications for pharmacotherapy in osteoporosis. Pharmacotherapy is recommended in all cases of osteoporosis and osteopenia when fracture risk is high.24

Generally, you should undertake a discussion with the patient of the relative risks and benefits of treatment, taking into account their values and preferences, to come to a shared decision. Tailoring treatment, based on the patient’s distinctive circumstances, through shared decision-making is key to compliance.25
Pharmacotherapy is not indicated in patients whose risk of fracture is low; however, you should reassess such patients every 2 to 4 years.26 Women with a very high BMD might not need to be retested with DXA any sooner than every 10 to 15 years.
There are 3 main classes of first-line pharmacotherapeutic agents for osteoporosis in older adults (TABLE 44,6,7,26-41): antiresorptives (bisphosphonates and denosumab), anabolics (teriparatide and abaloparatide), and a monoclonal sclerostin antibody (romosozumab). (TABLE 44,6,7,26-41 and the discussion in this section also remark on the selective estrogen-receptor modulator raloxifene, which is used in special clinical circumstances but has been removed from the first line of osteoporosis pharmacotherapy.)
❚ Bisphosphonates. Oral bisphosphonates (alendronate, ibandronate, risedronate) can be used as initial treatment in patients with a high risk of fracture.35 Bisphosphonates have been shown to reduce fracture risk and improve BMD. When an oral bisphosphonate cannot be tolerated, intravenous zoledronate or ibandronate can be used.41
Patients treated with a bisphosphonate should be assessed for their fracture risk after 3 to 5 years of treatment26; when intravenous zoledronate is given as initial therapy, patients should be assessed after 3 years. After assessment, patients who remain at high risk should continue treatment; those whose fracture risk has decreased to low or moderate should have treatment temporarily suspended (bisphosphonate holiday) for as long as 5 years.26 Patients on bisphosphonate holiday should have their fracture risk assessed at 2- to 4-year intervals.26 Restart treatment if there is an increase in fracture risk (eg, a decrease in BMD) or if a fracture occurs. Bisphosphonates have a prolonged effect on BMD—for many years after treatment is discontinued.27,28
Oral bisphosphonates are associated with gastroesophageal reflux disease, difficulty swallowing, and gastritis. Rare adverse effects include osteonecrosis of the jaw and atypical femur fracture.29
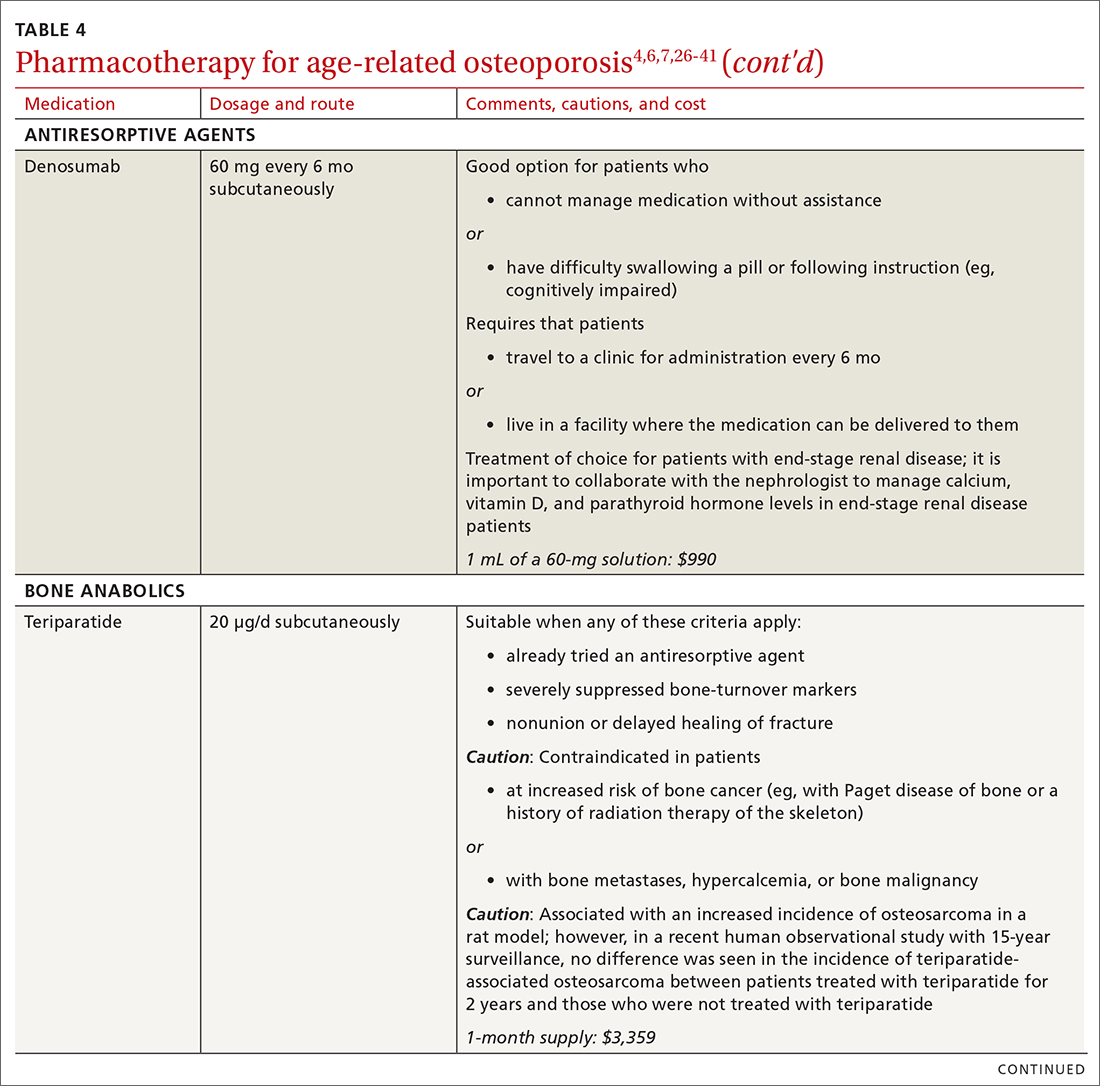
❚ Denosumab, a recombinant human antibody, is a relatively newer antiresorptive for initial treatment. Denosumab, 60 mg, is given subcutaneously every 6 months. The drug can be used when bisphosphonates are contraindicated, the patient finds the bisphosphonate dosing regimen difficult to follow, or the patient is unresponsive to bisphosphonates.
Patients taking denosumab are reassessed every 5 to 10 years to determine whether to continue therapy or change to a new drug. Abrupt discontinuation of therapy can lead to rebound bone loss and increased risk of fracture.30-32 As with bisphosphonates, long-term use can be associated with osteonecrosis of the jaw and atypical femur fracture.33
There is no recommendation for a drug holiday for denosumab. An increase in, or no loss of, bone density and no new fractures while being treated are signs of effective treatment. There is no guideline for stopping denosumab, unless the patient develops adverse effects.
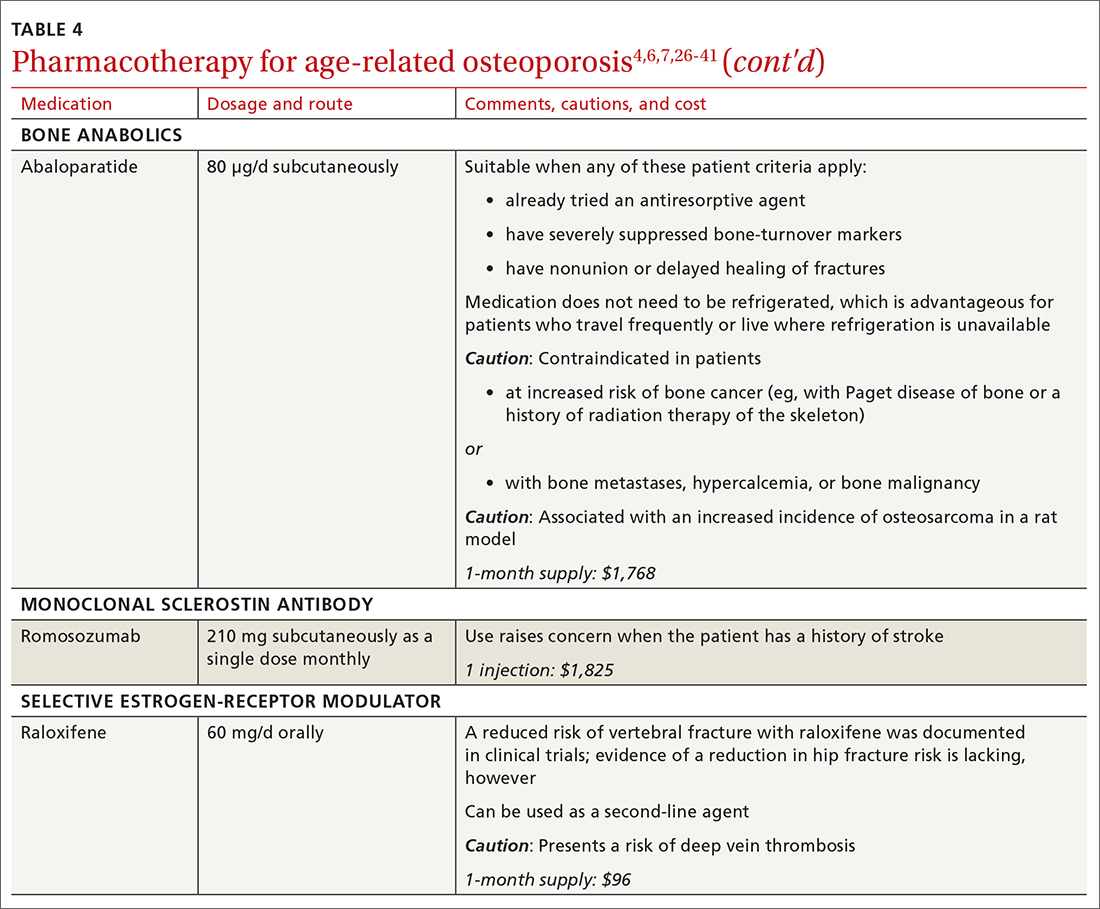
❚ Bone anabolics. Patients with a very high risk of fracture (eg, who have sustained multiple vertebral fractures), can begin treatment with teriparatide (20 μg/d subcutaneously) or abaloparatide (80 μg/d subcutaneously) for as long as 2 years, followed by treatment with an antiresorptive, such as a bisphosphonate.4,6 Teriparatide can be used in patients who have not responded to an antiresorptive as first-line treatment.
Both abaloparatide and teriparatide might be associated with a risk of osteosarcoma and are contraindicated in patients who are at increased risk of osteosarcoma.36,39,40
❚ Romosozumab, a monoclonal sclerostin antibody, can be used in patients with very high risk of fracture or with multiple vertebral fractures. Romosozumab increases bone formation and reduces bone resorption. It is given monthly, 210 mg subcutaneously, for 1 year. The recommendation is that patients who have completed a course of romosozumab continue with antiresorptive treatment.26
Romosozumab is associated with an increase in the risk of cardiovascular disease, including stroke and myocardial infarction.26
❚ Raloxifene, a selective estrogen-receptor modulator, is no longer a first-line agent for osteoporosis in older adults34 because of its association with an increased risk of deep-vein thrombosis, pulmonary embolism, and lethal stroke. However, raloxifene can be used, at 60 mg/d, when bisphosphonates or denosumab are unsuitable. In addition, raloxifene is particularly useful in women with a high risk of breast cancer and in men who are taking a long-acting gonadotropin-releasing hormone agonist for prostate cancer.37,38
Continue to: Influence of chronic...
Influence of chronic diseaseon bone health
Chronic diseases—hypertension, type 2 diabetes, hyperthyroidism, rheumatoid arthritis, ankylosing spondylitis, and gastroenterologic disorders such as celiac disease and ulcerative colitis—are known to affect bone loss that can hasten osteoporosis.16,18,21 Furthermore, medications used to treat chronic diseases are known to affect bone health: Some, such as statins, ACE inhibitors, and hydrochlorothiazide, are bone protective; others, such as steroids, pioglitazone, and selective serotonin reuptake inhibitors, accelerate bone loss.1,14,42,43 It is important to be aware of the effect of a patient’s chronic diseases, and treatments for those diseases, on bone health, to help develop an individualized osteoporosis prevention plan.
Monitoring the efficacy of treatment
Treatment of osteoporosis should not be initiated without baseline measurement of BMD of the spine and hip. Subsequent to establishing that baseline, serial measurement of BMD can be used to (1) determine when treatment needs to be initiated for an untreated patient and (2) assess response in a treated patient. There is no consensus on the interval at which DXA should be repeated for the purpose of monitoring treatment response; frequency depends on the individual’s circumstances and the medication used. Notably, many physicians repeat DXA after 2 years of treatment8; however, the American College of Physicians recommends against repeating DXA within the first 5 years of pharmacotherapy in women.24
Patients with suspected vertebral fracture or those with loss of height > 1.5 inches require lateral radiographs of the thoracic and lumbar spine to assess the status of fractures.4,6
❚ Bone-turnover markers measured in serum can be used to assess treatment efficacy and patient adherence. The formation marker procollagen type I N-terminal propeptide (P1NP) and the resorption marker beta C-terminal cross-linking telopeptide of type 1 collagen (bCTX) are preferred for evaluating bone turnover in the clinical setting. Assessing P1NP and bCTX at baseline and after 3 months of treatment might be effective in monitoring adherence, particularly in patients taking a bisphosphonate.44
Be sure to address fall prevention
It is important to address falls, and how to prevent them, in patients with osteoporosis. Falls can precipitate fracture in older adults with reduced BMD, and fractures are the most common and debilitating manifestation of osteoporosis. Your discussion of falls with patients should include45:
- consequences of falls
- cautions about medications that can cloud mental alertness
- use of appropriate footwear
- home safety, such as adequate lighting, removal of floor clutter, and installation of handrails in the bathroom and stairwells and on outside steps.
- having an annual comprehensive eye exam.
Osteoporosis is avoidable and treatable
Earlier research reported various expressions of number needed to treat for medical management of osteoporosis—making it difficult to follow a single number as a reference for gauging the effectiveness of pharmacotherapy.46,47 However, for older adults of different ethnic and racial backgrounds with multiple comorbidities and polypharmacy, it might be more pragmatic in primary care to establish a model of goal-oriented, individualized care. By focusing on prevention of bone loss, and being mindful that the risk of fracture almost doubles with a decrease of 1 SD in BMD, you can translate numbers to goals of care.48
In the United States, approximately one-half of osteoporosis cases in adults ≥ 50 years are managed by primary care providers. As a chronic disease, osteoporosis requires that you, first, provide regular monitoring and assessment, because risk can vary with comorbidities,49 and, second, discuss and initiate screening and treatment as appropriate, which can be done annually during a well-care visit.
CORRESPONDENCE
Nahid Rianon, MD, DrPH, Department of Family and Community Medicine, UTHealth McGovern Medical School, 6431 Fannin Street #JJL 324C, Houston, TX, 77030; [email protected]
- What is osteoporosis and what causes it? National Osteoporosis Foundation Website. 2020. Accessed April 28, 2021. www.nof.org/patients/what-is-osteoporosis/
- des Bordes J, Prasad S, Pratt G, et al. Knowledge, beliefs, and concerns about bone health from a systematic review and metasynthesis of qualitative studies. PLoS One. 2020;15:e0227765. doi: 10.1371/journal.pone.0227765
- Solomon DH, Johnston SS, Boytsov NN, et al. Osteoporosis medication use after hip fracture in U.S. patients between 2002 and 2011. J Bone Miner Res. 2014;29:1929-1937. doi: 10.1002/jbmr.2202
- Cosman F, de Beur SJ, LeBoff MS, et al; National Osteoporosis Foundation. Clinician's guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359-2381. doi: 10.1007/s00198-014-2794-2
- US Preventive Services Task Force; Curry SJ, Krist AH, Owens DK, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:2521-2531. doi: 10.1001/jama.2018.7498
- Camacho PM, Petak SM, Binkley N, et al. American Association of Clinical Endocrinologists and American College of Endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis - 2016. Endocr Pract. 2016;22(suppl 4):1-42. doi: 10.4158/EP161435.GL
- Watts NB, Adler RA, Bilezikian JP, et al; Endocrine Society. Osteoporosis in men: an Endocrine Society clinical practice guideline.J Clin Endocrinol Metab. 2012;97:1802-1822. doi: 10.1210/jc.2011-3045
- US Department of Health and Human Services. Bone Health and Osteoporosis: A Report of the Surgeon General. US Department of Health and Human Services, Public Health Service, Office of the Surgeon General; 2004. Accessed April 28, 2021. www.ncbi.nlm.nih.gov/books/NBK45513/pdf/Bookshelf_NBK45513.pdf
- Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser. 1994;843:1-129.
- Looker AC, Frenk SM. Percentage of adults aged 65 and over with osteoporosis or low bone mass at the femur neck or lumbar spine: United States, 2005--2010. Centers for Disease Control and Prevention, National Center for Health Statistics, Division of Health and Nutrition Examination Surveys. August 2015. Accessed April 28, 2021. www.cdc.gov/nchs/data/hestat/osteoporsis/osteoporosis2005_2010.pdf
- Kerschan-Schindl K. Prevention and rehabilitation of osteoporosis. Wien Med Wochenschr. 2016;166:22-27. doi: 10.1007/s10354-015-0417-y
- Tarantino U, Iolascon G, Cianferotti L, et al. Clinical guidelines for the prevention and treatment of osteoporosis: summary statements and recommendations from the Italian Society for Orthopaedics and Traumatology. J Orthop Traumatol. 2017;18(suppl 1):3-36. doi: 10.1007/s10195-017-0474-7
- Martineau P, Leslie WD, Johansson H, et al. In which patients does lumbar spine trabecular bone score (TBS) have the largest effect? Bone. 2018;113:161-168. doi: 10.1016/j.bone.2018.05.026
- Rianon NJ, Smith SM, Lee M, et al. Glycemic control and bone turnover in older Mexican Americans with type 2 diabetes. J Osteoporos. 2018;2018:7153021. doi: 10.1155/2018/7153021
- Richards C, Hans D, Leslie WD. Trabecular bone score (TBS) predicts fracture in ankylosing spondylitis: The Manitoba BMD Registry. J Clin Densitom. 2020;23:543-548. doi: 10.1016/j.jocd.2020.01.003
- Xue Y, Baker AL, Nader S, et al. Lumbar spine trabecular bone score (TBS) reflects diminished bone quality in patients with diabetes mellitus and oral glucocorticoid therapy. J Clin Densitom. 2018;21:185-192. doi: 10.1016/j.jocd.2017.09.003
- Silva BC, Broy SB, Boutroy S, et al. Fracture risk prediction by non-BMD DXA measures: the 2015 ISCD Official Positions Part 2: trabecular bone score. J Clin Densitom. 2015;18:309-330. doi: 10.1016/j.jocd.2015.06.008
- Silva BC, Leslie WD, Resch H, et al. Trabecular bone score: a noninvasive analytical method based upon the DXA image. J Bone Miner Res. 2014;29:518-530. doi: 10.1002/jbmr.2176
- Leslie WD, Aubry-Rozier B, Lamy O, et al; Manitoba Bone Density Program. TBS (trabecular bone score) and diabetes-related fracture risk. J Clin Endocrinol Metab. 2013;98:602-609.
- Looker AC, Sarafrazi Isfahani N, Fan B, et al. Trabecular bone scores and lumbar spine bone mineral density of US adults: comparison of relationships with demographic and body size variables. Osteoporos Int. 2016;27:2467-2475. doi: 10.1007/s00198-016-3550-6
- Rianon N, Ambrose CG, Pervin H, et al. Long-term use of angiotensin-converting enzyme inhibitors protects against bone loss in African-American elderly men. Arch Osteoporos. 2017;12:94. doi: 10.1007/s11657-017-0387-3
- Morton DJ, Barrett-Connor EL, Edelstein SL. Thiazides and bone mineral density in elderly men and women. Am J Epidemiol. 1994;139:1107-1115. doi: 10.1093/oxfordjournals.aje.a116954
- Sigurdsson G, Franzson L. Increased bone mineral density in a population-based group of 70-year-old women on thiazide diuretics, independent of parathyroid hormone levels. J Intern Med. 2001;250:51-56. doi: 10.1046/j.1365-2796.2001.00850.x
- Qaseem A, Forciea MA, McLean RM, et al; Clinical Guidelines Committee of the American College of Physicians. Treatment of low bone density or osteoporosis to prevent fractures in men and women: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017;166:818-839. doi: 10.7326/M15-1361
- des Bordes JKA, Suarez-Almazor ME, Volk RJ, et al. Online educational tool to promote bone health in cancer survivors. J Health Commun. 2017;22:808-817. doi: 10.1080/10810730.2017.1360415
- Shoback D, Rosen CJ, Black DM, et al. Pharmacological management of osteoporosis in postmenopausal women: an Endocrine Society guideline update. J Clin Endocrinol Metab. 2020;105:587-594. doi: 10.1210/clinem/dgaa048
- Black DM, Schwartz AV, Ensrud KE, et al; FLEX Research Group. Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA. 2006;296:2927-2938. doi: 10.1001/jama.296.24.2927
- Bone HG, Hosking D, Devogelaer J-P, et al. Ten years' experience with alendronate for osteoporosis in postmenopausal women. N Engl J Med. 2004;350:1189-1199. doi: 10.1056/NEJMoa030897
- Khosla S, Burr D, Cauley J, et al; American Society for Bone and Mineral Research. Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2007;22:1479-1491. doi: 10.1359/jbmr.0707onj
- Bone HG, Bolognese MA, Yuen CK, et al. Effects of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with low bone mass. J Clin Endocrinol Metab. 2011;96:972-980. doi: 10.1210/jc.2010-1502
- Cummings SR, Ferrari S, Eastell R, et al. Vertebral fractures after discontinuation of denosumab: a post hoc analysis of the randomized placebo-controlled FREEDOM Trial and its extension. J Bone Miner Res. 2018;33:190-198. doi: 10.1002/jbmr.3337
- Symonds C, Kline G. Warning of an increased risk of vertebral fracture after stopping denosumab. CMAJ. 2018;190:E485-E486. doi: 10.1503/cmaj.180115
- Aljohani S, Gaudin R, Weiser J, et al. Osteonecrosis of the jaw in patients treated with denosumab: a multicenter case series. J Craniomaxillofac Surg. 2018;46:1515-1525. doi: 10.1016/j.jcms.2018.05.046
- Barrett-Connor E, Mosca L, Collins P, et al; Raloxifene Use for The Heart (RUTH) Trial Investigators. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355:125-137. doi: 10.1056/NEJMoa062462
- Chesnut CH 3rd, Skag A, Christiansen C, et al; Oral Ibandronate Osteoporosis Vertebral Fracture Trial in North America and Europe (BONE). Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis. J Bone Miner Res. 2004;19:1241-1249. doi: 10.1359/JBMR.040325
- Gilsenban A, Midkiff K, Kellier-Steele N, et al. Teriparatide did not increase adult osteosarcoma incidence in a 15-year US postmarketing surveillance study. J Bone Miner Res. 2021;36:244-252. doi: 10.1002/jbmr.4188
- Cuzick J, Sestak I, Bonanni B, et al; SERM Chemoprevention of Breast Cancer Overview Group. Selective oestrogen receptor modulators in prevention of breast cancer: an updated meta-analysis of individual participant data. Lancet. 2013;381:1827-1834. doi: 10.1016/S0140-6736(13)60140-3
- Smith MR, Fallon MA, Lee H, et al. Raloxifene to prevent gonadotropin-releasing hormone agonist-induced bone loss in men with prostate cancer: a randomized controlled trial. J Clin Endocrinol Metab. 2004;89:3841-3846. doi: 10.1210/jc.2003-032058
- TYMLOS. Prescribing information. Radius Health, Inc.; April 2017. Accessed May 20, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2017/208743lbl.pdf
- FORTEO. Prescribing information. Eli Lilly and Co.; April 2020. Accessed May 20, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2020/021318s053lbl.pdf
- Wooltorton E. Patients receiving intravenous bisphosphonates should avoid invasive dental procedures. Can Med Assoc J. 2003;172:1684. doi: https://doi.org/10.1503/cmaj.050640
- Chiadika SM, Shobayo FO, Naqvi SH, et al. Lower femoral neck bone mineral density (BMD) in elderly women not on statins. Women Health. 2019;59:845-853. doi: 10.1080/03630242.2019.1567646
- Saraykar S, John V, Cao B, et al. Association of selective serotonin reuptake inhibitors and bone mineral density in elderly women. J Clin Densitom. 2018;21:193-199. doi: 10.1016/j.jocd.2017.05.016
- Lorentzon M, Branco J, Brandi ML, et al. Algorithm for the use of biochemical markers of bone turnover in the diagnosis, assessment and follow-up of treatment for osteoporosis. Adv Ther. 2019;36:2811-2824. doi: 10.1007/s12325-019-01063-9
- STEADI--older adult fall prevention. Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. 2019. Accessed April 28, 2021. www.cdc.gov/steadi/patient.html
- Cummings SR, San Martin J, McClung MR, et al; FREEDOM Trial. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361:756-765. doi: 10.1056/NEJMoa0809493
- Zhou Z, Chen C, Zhang J, et al. Safety of denosumab in postmenopausal women with osteoporosis or low bone mineral density: a meta-analysis. Int J Clin Exp Pathol. 2014;7:2113-2122.
- Faulkner KG. Bone matters: are density increases necessary to reduce fracture risk? J Bone Miner Res. 2000;15:183-187. doi: 10.1359/jbmr.2000.15.2.183
- Rianon N, Anand D, Rasu R. Changing trends in osteoporosis care from specialty to primary care physicians. Curr Med Res Opin. 2013;29:881-888. doi: 10.1185/03007995.2013.809335
Recommendations for care are evolving, with increasingly sophisticated screening and diagnostic tools and a broadening array of treatment options.
Recommendations for care are evolving, with increasingly sophisticated screening and diagnostic tools and a broadening array of treatment options.
As the population of older adults rises, primary osteoporosis has become a problem of public health significance, resulting in more than 2 million fractures and $19 billion in related costs annually in the United States.1 Despite the availability of effective primary and secondary preventive measures, many older adults do not receive adequate information on bone health from their primary care provider.2 Initiation of osteoporosis treatment is low even among patients who have had an osteoporotic fracture: Fewer than one-quarter of older adults with hip fracture have begun taking osteoporosis medication within 12 months of hospital discharge.3
In this overview of osteoporosis care, we provide information on how to evaluate and manage older adults in primary care settings who are at risk of, or have been given a diagnosis of, primary osteoporosis. The guidance that we offer reflects the most recent updates and recommendations by relevant professional societies.1,4-7
The nature and scope of an urgent problem
Osteoporosis is a skeletal disorder characterized by low bone mass and deterioration of bone structure that causes bone fragility and increases the risk of fracture.8 Operationally, it is defined by the World Health Organization as a bone mineral density (BMD) score below 2.5 SD from the mean value for a young White woman (ie, T-score ≤ –2.5).9 Primary osteoporosis is age related and occurs mostly in postmenopausal women and older men, affecting 25% of women and 5% of men ≥ 65 years.10
An osteoporotic fracture is particularly devastating in an older adult because it can cause pain, reduced mobility, depression, and social isolation and can increase the risk of related mortality.1 The National Osteoporosis Foundation estimates that 20% of older adults who sustain a hip fracture die within 1 year due to complications of the fracture itself or surgical repair.1 Therefore, it is of paramount importance to identify patients who are at increased risk of fracture and intervene early.
Clinical manifestations
Osteoporosis does not have a primary presentation; rather, disease manifests clinically when a patient develops complications. Often, a fragility fracture is the first sign in an older person.11
A fracture is the most important complication of osteoporosis and can result from low-trauma injury or a fall from standing height—thus, the term “fragility fracture.” Osteoporotic fractures commonly involve the vertebra, hip, and wrist. Hip and extremity fractures can result in limited or lost mobility and depression. Vertebral fractures can be asymptomatic or result in kyphosis and loss of height. Fractures can give rise to pain.
Age and female sexare risk factors
TABLE 11,6,10 lists risk factors associated with osteoporosis. Age is the most important; prevalence of osteoporosis increases with age. Other nonmodifiable risk factors include female sex (the disease appears earlier in women who enter menopause prematurely), family history of osteoporosis, and race and ethnicity. Twenty percent of Asian and non-Hispanic White women > 50 years have osteoporosis.1 A study showed that Mexican Americans are at higher risk of osteoporosis than non-Hispanic Whites; non-Hispanic Blacks are least affected.10

Other risk factors include low body weight (< 127 lb) and a history of fractures after age 50. Behavioral risk factors include smoking, excessive alcohol intake (> 3 drinks/d), poor nutrition, and a sedentary lifestyle.1,6
Continue to: Who should be screened?...
Who should be screened?
Screening is generally performed with a clinical evaluation and a dual-energy x-ray absorptiometry (DXA) scan of BMD. Measurement of BMD is generally recommended for screening all women ≥ 65 years and those < 65 years whose 10-year risk of fracture is equivalent to that of a 65-year-old White woman (see “Assessment of fracture risk” later in the article). For men, the US Preventive Services Task Force recommends screening those with a prior fracture or a secondary risk factor for disease.5 However, the National Osteoporosis Foundation recommends screening all men ≥ 70 years and those 50 to 69 years whose risk profile shows heightened risk.1,4
DXA of the spine and hip is preferred; the distal one-third of the radius (termed “33% radius”) of the nondominant arm can be used when spine and hip BMD cannot be interpreted because of bone changes from the disease process or artifacts, or in certain diseases in which the wrist region shows the earliest change (eg, primary hyperparathyroidism).6,7
Clinical evaluation includes a detailed history, physical examination, laboratory screening, and assessment for risk of fracture.
❚ History. Explore the presence of risk factors, including fractures in adulthood, falls, medication use, alcohol and tobacco use, family history of osteoporosis, and chronic disease.6,7
❚ Physical exam. Assess height, including any loss (> 1.5 in) since the patient’s second or third decade of life; kyphosis; frailty; and balance and mobility problems.4,6,7
❚ Laboratory and imaging studies. Perform basic laboratory testing when DXA is abnormal, including thyroid function, serum calcium, and renal function.6,12 Radiography of the lateral spine might be necessary, especially when there is kyphosis or loss of height. Assess for vertebral fracture, using lateral spine radiography, when vertebral involvement is suspected.6,7
❚ Assessment of fracture risk. Fracture risk can be assessed with any of a number of tools, including:
- Simplified Calculated Osteoporosis Risk Estimation (SCORE): www.medicalalgorithms.com/simplified-calculated-osteoporosis-risk-estimation-tool
- Osteoporosis Risk Assessment Instrument (ORAI): www.physio-pedia.com/The_Osteoporosis_Risk_Assessment_Instrument_(ORAI)
- Osteoporosis Index of Risk (OSIRIS): https://www.tandfonline.com/doi/abs/10.1080/gye.16.3.245.250?journalCode=igye20
- Osteoporosis Self-Assessment Tool (OST): www.ncbi.nlm.nih.gov/books/NBK45516/figure/ch10.f2/
- FRAX tool5: www.sheffield.ac.uk/FRAX.
The FRAX tool is widely used. It assesses a patient’s 10-year risk of fracture.
Diagnosis is based on these criteria
Diagnosis of osteoporosis is based on any 1 or more of the following criteria6:
- a history of fragility fracture not explained by metabolic bone disease
- T-score ≤ –2.5 (lumbar, hip, femoral neck, or 33% radius)
- a nation-specific FRAX score (in the absence of access to DXA).
❚ Secondary disease. Patients in whom secondary osteoporosis is suspected should undergo laboratory investigation to ascertain the cause; treatment of the underlying pathology might then be required. Evaluation for a secondary cause might include a complete blood count, comprehensive metabolic panel, protein electrophoresis and urinary protein electrophoresis (to rule out myeloproliferative and hematologic diseases), and tests of serum 25-hydroxyvitamin D, parathyroid hormone, serum calcium, alkaline phosphatase, 24-hour urinary calcium, sodium, and creatinine.6,7 Specialized testing for biochemical markers of bone turnover—so-called bone-turnover markers—can be considered as part of the initial evaluation and follow-up, although the tests are not recommended by the US Preventive Services Task Force (see “Monitoring the efficacy of treatment,” later in the article, for more information about these markers).6
Although BMD by DXA remains the gold standard in screening for and diagnosing osteoporosis, a high rate of fracture is seen in patients with certain diseases, such as type 2 diabetes and ankylosing spondylitis, who have a nonosteoporotic low T-score. This raises concerns about the usefulness of BMD for diagnosing osteoporosis in patients who have one of these diseases.13-16
❚
❚ Trabecular bone score (TBS), a surrogate bone-quality measure that is calculated based on the spine DXA image, has recently been introduced in clinical practice, and can be used to predict fracture risk in conjunction with BMD assessment by DXA and the FRAX score.17 TBS provides an indirect index of the trabecular microarchitecture using pixel gray-level variation in lumbar spine DXA images.18 Three categories of TBS (≤ 1.200, degraded microarchitecture; 1.200-1.350, partially degraded microarchitecture; and > 1.350, normal microarchitecture) have been reported to correspond with a T-score of, respectively, ≤ −2.5; −2.5 to −1.0; and > −1.0.18 TBS can be used only in patients with a body mass index of 15 to 37.5.19,20
There is no recommendation for monitoring bone quality using TBS after osteoporosis treatment. Such monitoring is at the clinician’s discretion for appropriate patients who might not show a risk of fracture, based on BMD measurement.
Continue to: Putting preventive measures into practice...
Putting preventive measures into practice
Measures to prevent osteoporosis and preserve bone health (TABLE 21,6) are best started in childhood but can be initiated at any age and maintained through the lifespan. Encourage older adults to adopt dietary and behavioral strategies to improve their bone health and prevent fracture. We recommend the following strategies; take each patient’s individual situation into consideration when electing to adopt any of these measures.

❚ Vitamin D. Consider checking the serum 25-hydroxyvitamin D level and providing supplementation (800-1000 IU daily, the National Osteoporosis Foundation recommends1) as necessary to maintain the level at 30-50 ng/mL.6
❚ Calcium. Encourage a daily dietary calcium intake of 1000-1200 mg. Supplement calcium if you determine that diet does not provide an adequate amount.
❚ Alcohol. Advise patients to limit consumption to < 3 drinks a day.
❚ Tobacco. Advise smoking cessation.
❚ Activity. Encourage an active lifestyle, including regular weight-bearing and balance exercises and resistance exercises such as Pilates, weightlifting, and tai chi. The regimen should be tailored to the patient’s individual situation.
❚ Medical therapy for concomitant illness. When possible, prescribe medications for chronic comorbidities that can also benefit bone health. For example, long-term use of angiotensin-converting enzyme (ACE) inhibitors and thiazide diuretics for hypertension are associated with a slower decline in BMD in some populations.21-23
Tailor treatment to patient’s circumstances
TABLE 34,6,24 describes indications for pharmacotherapy in osteoporosis. Pharmacotherapy is recommended in all cases of osteoporosis and osteopenia when fracture risk is high.24

Generally, you should undertake a discussion with the patient of the relative risks and benefits of treatment, taking into account their values and preferences, to come to a shared decision. Tailoring treatment, based on the patient’s distinctive circumstances, through shared decision-making is key to compliance.25
Pharmacotherapy is not indicated in patients whose risk of fracture is low; however, you should reassess such patients every 2 to 4 years.26 Women with a very high BMD might not need to be retested with DXA any sooner than every 10 to 15 years.
There are 3 main classes of first-line pharmacotherapeutic agents for osteoporosis in older adults (TABLE 44,6,7,26-41): antiresorptives (bisphosphonates and denosumab), anabolics (teriparatide and abaloparatide), and a monoclonal sclerostin antibody (romosozumab). (TABLE 44,6,7,26-41 and the discussion in this section also remark on the selective estrogen-receptor modulator raloxifene, which is used in special clinical circumstances but has been removed from the first line of osteoporosis pharmacotherapy.)



❚ Bisphosphonates. Oral bisphosphonates (alendronate, ibandronate, risedronate) can be used as initial treatment in patients with a high risk of fracture.35 Bisphosphonates have been shown to reduce fracture risk and improve BMD. When an oral bisphosphonate cannot be tolerated, intravenous zoledronate or ibandronate can be used.41
Patients treated with a bisphosphonate should be assessed for their fracture risk after 3 to 5 years of treatment26; when intravenous zoledronate is given as initial therapy, patients should be assessed after 3 years. After assessment, patients who remain at high risk should continue treatment; those whose fracture risk has decreased to low or moderate should have treatment temporarily suspended (bisphosphonate holiday) for as long as 5 years.26 Patients on bisphosphonate holiday should have their fracture risk assessed at 2- to 4-year intervals.26 Restart treatment if there is an increase in fracture risk (eg, a decrease in BMD) or if a fracture occurs. Bisphosphonates have a prolonged effect on BMD—for many years after treatment is discontinued.27,28
Oral bisphosphonates are associated with gastroesophageal reflux disease, difficulty swallowing, and gastritis. Rare adverse effects include osteonecrosis of the jaw and atypical femur fracture.29
❚ Denosumab, a recombinant human antibody, is a relatively newer antiresorptive for initial treatment. Denosumab, 60 mg, is given subcutaneously every 6 months. The drug can be used when bisphosphonates are contraindicated, the patient finds the bisphosphonate dosing regimen difficult to follow, or the patient is unresponsive to bisphosphonates.
Patients taking denosumab are reassessed every 5 to 10 years to determine whether to continue therapy or change to a new drug. Abrupt discontinuation of therapy can lead to rebound bone loss and increased risk of fracture.30-32 As with bisphosphonates, long-term use can be associated with osteonecrosis of the jaw and atypical femur fracture.33
There is no recommendation for a drug holiday for denosumab. An increase in, or no loss of, bone density and no new fractures while being treated are signs of effective treatment. There is no guideline for stopping denosumab, unless the patient develops adverse effects.
❚ Bone anabolics. Patients with a very high risk of fracture (eg, who have sustained multiple vertebral fractures), can begin treatment with teriparatide (20 μg/d subcutaneously) or abaloparatide (80 μg/d subcutaneously) for as long as 2 years, followed by treatment with an antiresorptive, such as a bisphosphonate.4,6 Teriparatide can be used in patients who have not responded to an antiresorptive as first-line treatment.
Both abaloparatide and teriparatide might be associated with a risk of osteosarcoma and are contraindicated in patients who are at increased risk of osteosarcoma.36,39,40
❚ Romosozumab, a monoclonal sclerostin antibody, can be used in patients with very high risk of fracture or with multiple vertebral fractures. Romosozumab increases bone formation and reduces bone resorption. It is given monthly, 210 mg subcutaneously, for 1 year. The recommendation is that patients who have completed a course of romosozumab continue with antiresorptive treatment.26
Romosozumab is associated with an increase in the risk of cardiovascular disease, including stroke and myocardial infarction.26
❚ Raloxifene, a selective estrogen-receptor modulator, is no longer a first-line agent for osteoporosis in older adults34 because of its association with an increased risk of deep-vein thrombosis, pulmonary embolism, and lethal stroke. However, raloxifene can be used, at 60 mg/d, when bisphosphonates or denosumab are unsuitable. In addition, raloxifene is particularly useful in women with a high risk of breast cancer and in men who are taking a long-acting gonadotropin-releasing hormone agonist for prostate cancer.37,38
Continue to: Influence of chronic...
Influence of chronic diseaseon bone health
Chronic diseases—hypertension, type 2 diabetes, hyperthyroidism, rheumatoid arthritis, ankylosing spondylitis, and gastroenterologic disorders such as celiac disease and ulcerative colitis—are known to affect bone loss that can hasten osteoporosis.16,18,21 Furthermore, medications used to treat chronic diseases are known to affect bone health: Some, such as statins, ACE inhibitors, and hydrochlorothiazide, are bone protective; others, such as steroids, pioglitazone, and selective serotonin reuptake inhibitors, accelerate bone loss.1,14,42,43 It is important to be aware of the effect of a patient’s chronic diseases, and treatments for those diseases, on bone health, to help develop an individualized osteoporosis prevention plan.
Monitoring the efficacy of treatment
Treatment of osteoporosis should not be initiated without baseline measurement of BMD of the spine and hip. Subsequent to establishing that baseline, serial measurement of BMD can be used to (1) determine when treatment needs to be initiated for an untreated patient and (2) assess response in a treated patient. There is no consensus on the interval at which DXA should be repeated for the purpose of monitoring treatment response; frequency depends on the individual’s circumstances and the medication used. Notably, many physicians repeat DXA after 2 years of treatment8; however, the American College of Physicians recommends against repeating DXA within the first 5 years of pharmacotherapy in women.24
Patients with suspected vertebral fracture or those with loss of height > 1.5 inches require lateral radiographs of the thoracic and lumbar spine to assess the status of fractures.4,6
❚ Bone-turnover markers measured in serum can be used to assess treatment efficacy and patient adherence. The formation marker procollagen type I N-terminal propeptide (P1NP) and the resorption marker beta C-terminal cross-linking telopeptide of type 1 collagen (bCTX) are preferred for evaluating bone turnover in the clinical setting. Assessing P1NP and bCTX at baseline and after 3 months of treatment might be effective in monitoring adherence, particularly in patients taking a bisphosphonate.44
Be sure to address fall prevention
It is important to address falls, and how to prevent them, in patients with osteoporosis. Falls can precipitate fracture in older adults with reduced BMD, and fractures are the most common and debilitating manifestation of osteoporosis. Your discussion of falls with patients should include45:
- consequences of falls
- cautions about medications that can cloud mental alertness
- use of appropriate footwear
- home safety, such as adequate lighting, removal of floor clutter, and installation of handrails in the bathroom and stairwells and on outside steps.
- having an annual comprehensive eye exam.
Osteoporosis is avoidable and treatable
Earlier research reported various expressions of number needed to treat for medical management of osteoporosis—making it difficult to follow a single number as a reference for gauging the effectiveness of pharmacotherapy.46,47 However, for older adults of different ethnic and racial backgrounds with multiple comorbidities and polypharmacy, it might be more pragmatic in primary care to establish a model of goal-oriented, individualized care. By focusing on prevention of bone loss, and being mindful that the risk of fracture almost doubles with a decrease of 1 SD in BMD, you can translate numbers to goals of care.48
In the United States, approximately one-half of osteoporosis cases in adults ≥ 50 years are managed by primary care providers. As a chronic disease, osteoporosis requires that you, first, provide regular monitoring and assessment, because risk can vary with comorbidities,49 and, second, discuss and initiate screening and treatment as appropriate, which can be done annually during a well-care visit.
CORRESPONDENCE
Nahid Rianon, MD, DrPH, Department of Family and Community Medicine, UTHealth McGovern Medical School, 6431 Fannin Street #JJL 324C, Houston, TX, 77030; [email protected]
As the population of older adults rises, primary osteoporosis has become a problem of public health significance, resulting in more than 2 million fractures and $19 billion in related costs annually in the United States.1 Despite the availability of effective primary and secondary preventive measures, many older adults do not receive adequate information on bone health from their primary care provider.2 Initiation of osteoporosis treatment is low even among patients who have had an osteoporotic fracture: Fewer than one-quarter of older adults with hip fracture have begun taking osteoporosis medication within 12 months of hospital discharge.3
In this overview of osteoporosis care, we provide information on how to evaluate and manage older adults in primary care settings who are at risk of, or have been given a diagnosis of, primary osteoporosis. The guidance that we offer reflects the most recent updates and recommendations by relevant professional societies.1,4-7
The nature and scope of an urgent problem
Osteoporosis is a skeletal disorder characterized by low bone mass and deterioration of bone structure that causes bone fragility and increases the risk of fracture.8 Operationally, it is defined by the World Health Organization as a bone mineral density (BMD) score below 2.5 SD from the mean value for a young White woman (ie, T-score ≤ –2.5).9 Primary osteoporosis is age related and occurs mostly in postmenopausal women and older men, affecting 25% of women and 5% of men ≥ 65 years.10
An osteoporotic fracture is particularly devastating in an older adult because it can cause pain, reduced mobility, depression, and social isolation and can increase the risk of related mortality.1 The National Osteoporosis Foundation estimates that 20% of older adults who sustain a hip fracture die within 1 year due to complications of the fracture itself or surgical repair.1 Therefore, it is of paramount importance to identify patients who are at increased risk of fracture and intervene early.
Clinical manifestations
Osteoporosis does not have a primary presentation; rather, disease manifests clinically when a patient develops complications. Often, a fragility fracture is the first sign in an older person.11
A fracture is the most important complication of osteoporosis and can result from low-trauma injury or a fall from standing height—thus, the term “fragility fracture.” Osteoporotic fractures commonly involve the vertebra, hip, and wrist. Hip and extremity fractures can result in limited or lost mobility and depression. Vertebral fractures can be asymptomatic or result in kyphosis and loss of height. Fractures can give rise to pain.
Age and female sexare risk factors
TABLE 11,6,10 lists risk factors associated with osteoporosis. Age is the most important; prevalence of osteoporosis increases with age. Other nonmodifiable risk factors include female sex (the disease appears earlier in women who enter menopause prematurely), family history of osteoporosis, and race and ethnicity. Twenty percent of Asian and non-Hispanic White women > 50 years have osteoporosis.1 A study showed that Mexican Americans are at higher risk of osteoporosis than non-Hispanic Whites; non-Hispanic Blacks are least affected.10

Other risk factors include low body weight (< 127 lb) and a history of fractures after age 50. Behavioral risk factors include smoking, excessive alcohol intake (> 3 drinks/d), poor nutrition, and a sedentary lifestyle.1,6
Continue to: Who should be screened?...
Who should be screened?
Screening is generally performed with a clinical evaluation and a dual-energy x-ray absorptiometry (DXA) scan of BMD. Measurement of BMD is generally recommended for screening all women ≥ 65 years and those < 65 years whose 10-year risk of fracture is equivalent to that of a 65-year-old White woman (see “Assessment of fracture risk” later in the article). For men, the US Preventive Services Task Force recommends screening those with a prior fracture or a secondary risk factor for disease.5 However, the National Osteoporosis Foundation recommends screening all men ≥ 70 years and those 50 to 69 years whose risk profile shows heightened risk.1,4
DXA of the spine and hip is preferred; the distal one-third of the radius (termed “33% radius”) of the nondominant arm can be used when spine and hip BMD cannot be interpreted because of bone changes from the disease process or artifacts, or in certain diseases in which the wrist region shows the earliest change (eg, primary hyperparathyroidism).6,7
Clinical evaluation includes a detailed history, physical examination, laboratory screening, and assessment for risk of fracture.
❚ History. Explore the presence of risk factors, including fractures in adulthood, falls, medication use, alcohol and tobacco use, family history of osteoporosis, and chronic disease.6,7
❚ Physical exam. Assess height, including any loss (> 1.5 in) since the patient’s second or third decade of life; kyphosis; frailty; and balance and mobility problems.4,6,7
❚ Laboratory and imaging studies. Perform basic laboratory testing when DXA is abnormal, including thyroid function, serum calcium, and renal function.6,12 Radiography of the lateral spine might be necessary, especially when there is kyphosis or loss of height. Assess for vertebral fracture, using lateral spine radiography, when vertebral involvement is suspected.6,7
❚ Assessment of fracture risk. Fracture risk can be assessed with any of a number of tools, including:
- Simplified Calculated Osteoporosis Risk Estimation (SCORE): www.medicalalgorithms.com/simplified-calculated-osteoporosis-risk-estimation-tool
- Osteoporosis Risk Assessment Instrument (ORAI): www.physio-pedia.com/The_Osteoporosis_Risk_Assessment_Instrument_(ORAI)
- Osteoporosis Index of Risk (OSIRIS): https://www.tandfonline.com/doi/abs/10.1080/gye.16.3.245.250?journalCode=igye20
- Osteoporosis Self-Assessment Tool (OST): www.ncbi.nlm.nih.gov/books/NBK45516/figure/ch10.f2/
- FRAX tool5: www.sheffield.ac.uk/FRAX.
The FRAX tool is widely used. It assesses a patient’s 10-year risk of fracture.
Diagnosis is based on these criteria
Diagnosis of osteoporosis is based on any 1 or more of the following criteria6:
- a history of fragility fracture not explained by metabolic bone disease
- T-score ≤ –2.5 (lumbar, hip, femoral neck, or 33% radius)
- a nation-specific FRAX score (in the absence of access to DXA).
❚ Secondary disease. Patients in whom secondary osteoporosis is suspected should undergo laboratory investigation to ascertain the cause; treatment of the underlying pathology might then be required. Evaluation for a secondary cause might include a complete blood count, comprehensive metabolic panel, protein electrophoresis and urinary protein electrophoresis (to rule out myeloproliferative and hematologic diseases), and tests of serum 25-hydroxyvitamin D, parathyroid hormone, serum calcium, alkaline phosphatase, 24-hour urinary calcium, sodium, and creatinine.6,7 Specialized testing for biochemical markers of bone turnover—so-called bone-turnover markers—can be considered as part of the initial evaluation and follow-up, although the tests are not recommended by the US Preventive Services Task Force (see “Monitoring the efficacy of treatment,” later in the article, for more information about these markers).6
Although BMD by DXA remains the gold standard in screening for and diagnosing osteoporosis, a high rate of fracture is seen in patients with certain diseases, such as type 2 diabetes and ankylosing spondylitis, who have a nonosteoporotic low T-score. This raises concerns about the usefulness of BMD for diagnosing osteoporosis in patients who have one of these diseases.13-16
❚
❚ Trabecular bone score (TBS), a surrogate bone-quality measure that is calculated based on the spine DXA image, has recently been introduced in clinical practice, and can be used to predict fracture risk in conjunction with BMD assessment by DXA and the FRAX score.17 TBS provides an indirect index of the trabecular microarchitecture using pixel gray-level variation in lumbar spine DXA images.18 Three categories of TBS (≤ 1.200, degraded microarchitecture; 1.200-1.350, partially degraded microarchitecture; and > 1.350, normal microarchitecture) have been reported to correspond with a T-score of, respectively, ≤ −2.5; −2.5 to −1.0; and > −1.0.18 TBS can be used only in patients with a body mass index of 15 to 37.5.19,20
There is no recommendation for monitoring bone quality using TBS after osteoporosis treatment. Such monitoring is at the clinician’s discretion for appropriate patients who might not show a risk of fracture, based on BMD measurement.
Continue to: Putting preventive measures into practice...
Putting preventive measures into practice
Measures to prevent osteoporosis and preserve bone health (TABLE 21,6) are best started in childhood but can be initiated at any age and maintained through the lifespan. Encourage older adults to adopt dietary and behavioral strategies to improve their bone health and prevent fracture. We recommend the following strategies; take each patient’s individual situation into consideration when electing to adopt any of these measures.

❚ Vitamin D. Consider checking the serum 25-hydroxyvitamin D level and providing supplementation (800-1000 IU daily, the National Osteoporosis Foundation recommends1) as necessary to maintain the level at 30-50 ng/mL.6
❚ Calcium. Encourage a daily dietary calcium intake of 1000-1200 mg. Supplement calcium if you determine that diet does not provide an adequate amount.
❚ Alcohol. Advise patients to limit consumption to < 3 drinks a day.
❚ Tobacco. Advise smoking cessation.
❚ Activity. Encourage an active lifestyle, including regular weight-bearing and balance exercises and resistance exercises such as Pilates, weightlifting, and tai chi. The regimen should be tailored to the patient’s individual situation.
❚ Medical therapy for concomitant illness. When possible, prescribe medications for chronic comorbidities that can also benefit bone health. For example, long-term use of angiotensin-converting enzyme (ACE) inhibitors and thiazide diuretics for hypertension are associated with a slower decline in BMD in some populations.21-23
Tailor treatment to patient’s circumstances
TABLE 34,6,24 describes indications for pharmacotherapy in osteoporosis. Pharmacotherapy is recommended in all cases of osteoporosis and osteopenia when fracture risk is high.24

Generally, you should undertake a discussion with the patient of the relative risks and benefits of treatment, taking into account their values and preferences, to come to a shared decision. Tailoring treatment, based on the patient’s distinctive circumstances, through shared decision-making is key to compliance.25
Pharmacotherapy is not indicated in patients whose risk of fracture is low; however, you should reassess such patients every 2 to 4 years.26 Women with a very high BMD might not need to be retested with DXA any sooner than every 10 to 15 years.
There are 3 main classes of first-line pharmacotherapeutic agents for osteoporosis in older adults (TABLE 44,6,7,26-41): antiresorptives (bisphosphonates and denosumab), anabolics (teriparatide and abaloparatide), and a monoclonal sclerostin antibody (romosozumab). (TABLE 44,6,7,26-41 and the discussion in this section also remark on the selective estrogen-receptor modulator raloxifene, which is used in special clinical circumstances but has been removed from the first line of osteoporosis pharmacotherapy.)



❚ Bisphosphonates. Oral bisphosphonates (alendronate, ibandronate, risedronate) can be used as initial treatment in patients with a high risk of fracture.35 Bisphosphonates have been shown to reduce fracture risk and improve BMD. When an oral bisphosphonate cannot be tolerated, intravenous zoledronate or ibandronate can be used.41
Patients treated with a bisphosphonate should be assessed for their fracture risk after 3 to 5 years of treatment26; when intravenous zoledronate is given as initial therapy, patients should be assessed after 3 years. After assessment, patients who remain at high risk should continue treatment; those whose fracture risk has decreased to low or moderate should have treatment temporarily suspended (bisphosphonate holiday) for as long as 5 years.26 Patients on bisphosphonate holiday should have their fracture risk assessed at 2- to 4-year intervals.26 Restart treatment if there is an increase in fracture risk (eg, a decrease in BMD) or if a fracture occurs. Bisphosphonates have a prolonged effect on BMD—for many years after treatment is discontinued.27,28
Oral bisphosphonates are associated with gastroesophageal reflux disease, difficulty swallowing, and gastritis. Rare adverse effects include osteonecrosis of the jaw and atypical femur fracture.29
❚ Denosumab, a recombinant human antibody, is a relatively newer antiresorptive for initial treatment. Denosumab, 60 mg, is given subcutaneously every 6 months. The drug can be used when bisphosphonates are contraindicated, the patient finds the bisphosphonate dosing regimen difficult to follow, or the patient is unresponsive to bisphosphonates.
Patients taking denosumab are reassessed every 5 to 10 years to determine whether to continue therapy or change to a new drug. Abrupt discontinuation of therapy can lead to rebound bone loss and increased risk of fracture.30-32 As with bisphosphonates, long-term use can be associated with osteonecrosis of the jaw and atypical femur fracture.33
There is no recommendation for a drug holiday for denosumab. An increase in, or no loss of, bone density and no new fractures while being treated are signs of effective treatment. There is no guideline for stopping denosumab, unless the patient develops adverse effects.
❚ Bone anabolics. Patients with a very high risk of fracture (eg, who have sustained multiple vertebral fractures), can begin treatment with teriparatide (20 μg/d subcutaneously) or abaloparatide (80 μg/d subcutaneously) for as long as 2 years, followed by treatment with an antiresorptive, such as a bisphosphonate.4,6 Teriparatide can be used in patients who have not responded to an antiresorptive as first-line treatment.
Both abaloparatide and teriparatide might be associated with a risk of osteosarcoma and are contraindicated in patients who are at increased risk of osteosarcoma.36,39,40
❚ Romosozumab, a monoclonal sclerostin antibody, can be used in patients with very high risk of fracture or with multiple vertebral fractures. Romosozumab increases bone formation and reduces bone resorption. It is given monthly, 210 mg subcutaneously, for 1 year. The recommendation is that patients who have completed a course of romosozumab continue with antiresorptive treatment.26
Romosozumab is associated with an increase in the risk of cardiovascular disease, including stroke and myocardial infarction.26
❚ Raloxifene, a selective estrogen-receptor modulator, is no longer a first-line agent for osteoporosis in older adults34 because of its association with an increased risk of deep-vein thrombosis, pulmonary embolism, and lethal stroke. However, raloxifene can be used, at 60 mg/d, when bisphosphonates or denosumab are unsuitable. In addition, raloxifene is particularly useful in women with a high risk of breast cancer and in men who are taking a long-acting gonadotropin-releasing hormone agonist for prostate cancer.37,38
Continue to: Influence of chronic...
Influence of chronic diseaseon bone health
Chronic diseases—hypertension, type 2 diabetes, hyperthyroidism, rheumatoid arthritis, ankylosing spondylitis, and gastroenterologic disorders such as celiac disease and ulcerative colitis—are known to affect bone loss that can hasten osteoporosis.16,18,21 Furthermore, medications used to treat chronic diseases are known to affect bone health: Some, such as statins, ACE inhibitors, and hydrochlorothiazide, are bone protective; others, such as steroids, pioglitazone, and selective serotonin reuptake inhibitors, accelerate bone loss.1,14,42,43 It is important to be aware of the effect of a patient’s chronic diseases, and treatments for those diseases, on bone health, to help develop an individualized osteoporosis prevention plan.
Monitoring the efficacy of treatment
Treatment of osteoporosis should not be initiated without baseline measurement of BMD of the spine and hip. Subsequent to establishing that baseline, serial measurement of BMD can be used to (1) determine when treatment needs to be initiated for an untreated patient and (2) assess response in a treated patient. There is no consensus on the interval at which DXA should be repeated for the purpose of monitoring treatment response; frequency depends on the individual’s circumstances and the medication used. Notably, many physicians repeat DXA after 2 years of treatment8; however, the American College of Physicians recommends against repeating DXA within the first 5 years of pharmacotherapy in women.24
Patients with suspected vertebral fracture or those with loss of height > 1.5 inches require lateral radiographs of the thoracic and lumbar spine to assess the status of fractures.4,6
❚ Bone-turnover markers measured in serum can be used to assess treatment efficacy and patient adherence. The formation marker procollagen type I N-terminal propeptide (P1NP) and the resorption marker beta C-terminal cross-linking telopeptide of type 1 collagen (bCTX) are preferred for evaluating bone turnover in the clinical setting. Assessing P1NP and bCTX at baseline and after 3 months of treatment might be effective in monitoring adherence, particularly in patients taking a bisphosphonate.44
Be sure to address fall prevention
It is important to address falls, and how to prevent them, in patients with osteoporosis. Falls can precipitate fracture in older adults with reduced BMD, and fractures are the most common and debilitating manifestation of osteoporosis. Your discussion of falls with patients should include45:
- consequences of falls
- cautions about medications that can cloud mental alertness
- use of appropriate footwear
- home safety, such as adequate lighting, removal of floor clutter, and installation of handrails in the bathroom and stairwells and on outside steps.
- having an annual comprehensive eye exam.
Osteoporosis is avoidable and treatable
Earlier research reported various expressions of number needed to treat for medical management of osteoporosis—making it difficult to follow a single number as a reference for gauging the effectiveness of pharmacotherapy.46,47 However, for older adults of different ethnic and racial backgrounds with multiple comorbidities and polypharmacy, it might be more pragmatic in primary care to establish a model of goal-oriented, individualized care. By focusing on prevention of bone loss, and being mindful that the risk of fracture almost doubles with a decrease of 1 SD in BMD, you can translate numbers to goals of care.48
In the United States, approximately one-half of osteoporosis cases in adults ≥ 50 years are managed by primary care providers. As a chronic disease, osteoporosis requires that you, first, provide regular monitoring and assessment, because risk can vary with comorbidities,49 and, second, discuss and initiate screening and treatment as appropriate, which can be done annually during a well-care visit.
CORRESPONDENCE
Nahid Rianon, MD, DrPH, Department of Family and Community Medicine, UTHealth McGovern Medical School, 6431 Fannin Street #JJL 324C, Houston, TX, 77030; [email protected]
- What is osteoporosis and what causes it? National Osteoporosis Foundation Website. 2020. Accessed April 28, 2021. www.nof.org/patients/what-is-osteoporosis/
- des Bordes J, Prasad S, Pratt G, et al. Knowledge, beliefs, and concerns about bone health from a systematic review and metasynthesis of qualitative studies. PLoS One. 2020;15:e0227765. doi: 10.1371/journal.pone.0227765
- Solomon DH, Johnston SS, Boytsov NN, et al. Osteoporosis medication use after hip fracture in U.S. patients between 2002 and 2011. J Bone Miner Res. 2014;29:1929-1937. doi: 10.1002/jbmr.2202
- Cosman F, de Beur SJ, LeBoff MS, et al; National Osteoporosis Foundation. Clinician's guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359-2381. doi: 10.1007/s00198-014-2794-2
- US Preventive Services Task Force; Curry SJ, Krist AH, Owens DK, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:2521-2531. doi: 10.1001/jama.2018.7498
- Camacho PM, Petak SM, Binkley N, et al. American Association of Clinical Endocrinologists and American College of Endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis - 2016. Endocr Pract. 2016;22(suppl 4):1-42. doi: 10.4158/EP161435.GL
- Watts NB, Adler RA, Bilezikian JP, et al; Endocrine Society. Osteoporosis in men: an Endocrine Society clinical practice guideline.J Clin Endocrinol Metab. 2012;97:1802-1822. doi: 10.1210/jc.2011-3045
- US Department of Health and Human Services. Bone Health and Osteoporosis: A Report of the Surgeon General. US Department of Health and Human Services, Public Health Service, Office of the Surgeon General; 2004. Accessed April 28, 2021. www.ncbi.nlm.nih.gov/books/NBK45513/pdf/Bookshelf_NBK45513.pdf
- Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser. 1994;843:1-129.
- Looker AC, Frenk SM. Percentage of adults aged 65 and over with osteoporosis or low bone mass at the femur neck or lumbar spine: United States, 2005--2010. Centers for Disease Control and Prevention, National Center for Health Statistics, Division of Health and Nutrition Examination Surveys. August 2015. Accessed April 28, 2021. www.cdc.gov/nchs/data/hestat/osteoporsis/osteoporosis2005_2010.pdf
- Kerschan-Schindl K. Prevention and rehabilitation of osteoporosis. Wien Med Wochenschr. 2016;166:22-27. doi: 10.1007/s10354-015-0417-y
- Tarantino U, Iolascon G, Cianferotti L, et al. Clinical guidelines for the prevention and treatment of osteoporosis: summary statements and recommendations from the Italian Society for Orthopaedics and Traumatology. J Orthop Traumatol. 2017;18(suppl 1):3-36. doi: 10.1007/s10195-017-0474-7
- Martineau P, Leslie WD, Johansson H, et al. In which patients does lumbar spine trabecular bone score (TBS) have the largest effect? Bone. 2018;113:161-168. doi: 10.1016/j.bone.2018.05.026
- Rianon NJ, Smith SM, Lee M, et al. Glycemic control and bone turnover in older Mexican Americans with type 2 diabetes. J Osteoporos. 2018;2018:7153021. doi: 10.1155/2018/7153021
- Richards C, Hans D, Leslie WD. Trabecular bone score (TBS) predicts fracture in ankylosing spondylitis: The Manitoba BMD Registry. J Clin Densitom. 2020;23:543-548. doi: 10.1016/j.jocd.2020.01.003
- Xue Y, Baker AL, Nader S, et al. Lumbar spine trabecular bone score (TBS) reflects diminished bone quality in patients with diabetes mellitus and oral glucocorticoid therapy. J Clin Densitom. 2018;21:185-192. doi: 10.1016/j.jocd.2017.09.003
- Silva BC, Broy SB, Boutroy S, et al. Fracture risk prediction by non-BMD DXA measures: the 2015 ISCD Official Positions Part 2: trabecular bone score. J Clin Densitom. 2015;18:309-330. doi: 10.1016/j.jocd.2015.06.008
- Silva BC, Leslie WD, Resch H, et al. Trabecular bone score: a noninvasive analytical method based upon the DXA image. J Bone Miner Res. 2014;29:518-530. doi: 10.1002/jbmr.2176
- Leslie WD, Aubry-Rozier B, Lamy O, et al; Manitoba Bone Density Program. TBS (trabecular bone score) and diabetes-related fracture risk. J Clin Endocrinol Metab. 2013;98:602-609.
- Looker AC, Sarafrazi Isfahani N, Fan B, et al. Trabecular bone scores and lumbar spine bone mineral density of US adults: comparison of relationships with demographic and body size variables. Osteoporos Int. 2016;27:2467-2475. doi: 10.1007/s00198-016-3550-6
- Rianon N, Ambrose CG, Pervin H, et al. Long-term use of angiotensin-converting enzyme inhibitors protects against bone loss in African-American elderly men. Arch Osteoporos. 2017;12:94. doi: 10.1007/s11657-017-0387-3
- Morton DJ, Barrett-Connor EL, Edelstein SL. Thiazides and bone mineral density in elderly men and women. Am J Epidemiol. 1994;139:1107-1115. doi: 10.1093/oxfordjournals.aje.a116954
- Sigurdsson G, Franzson L. Increased bone mineral density in a population-based group of 70-year-old women on thiazide diuretics, independent of parathyroid hormone levels. J Intern Med. 2001;250:51-56. doi: 10.1046/j.1365-2796.2001.00850.x
- Qaseem A, Forciea MA, McLean RM, et al; Clinical Guidelines Committee of the American College of Physicians. Treatment of low bone density or osteoporosis to prevent fractures in men and women: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017;166:818-839. doi: 10.7326/M15-1361
- des Bordes JKA, Suarez-Almazor ME, Volk RJ, et al. Online educational tool to promote bone health in cancer survivors. J Health Commun. 2017;22:808-817. doi: 10.1080/10810730.2017.1360415
- Shoback D, Rosen CJ, Black DM, et al. Pharmacological management of osteoporosis in postmenopausal women: an Endocrine Society guideline update. J Clin Endocrinol Metab. 2020;105:587-594. doi: 10.1210/clinem/dgaa048
- Black DM, Schwartz AV, Ensrud KE, et al; FLEX Research Group. Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA. 2006;296:2927-2938. doi: 10.1001/jama.296.24.2927
- Bone HG, Hosking D, Devogelaer J-P, et al. Ten years' experience with alendronate for osteoporosis in postmenopausal women. N Engl J Med. 2004;350:1189-1199. doi: 10.1056/NEJMoa030897
- Khosla S, Burr D, Cauley J, et al; American Society for Bone and Mineral Research. Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2007;22:1479-1491. doi: 10.1359/jbmr.0707onj
- Bone HG, Bolognese MA, Yuen CK, et al. Effects of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with low bone mass. J Clin Endocrinol Metab. 2011;96:972-980. doi: 10.1210/jc.2010-1502
- Cummings SR, Ferrari S, Eastell R, et al. Vertebral fractures after discontinuation of denosumab: a post hoc analysis of the randomized placebo-controlled FREEDOM Trial and its extension. J Bone Miner Res. 2018;33:190-198. doi: 10.1002/jbmr.3337
- Symonds C, Kline G. Warning of an increased risk of vertebral fracture after stopping denosumab. CMAJ. 2018;190:E485-E486. doi: 10.1503/cmaj.180115
- Aljohani S, Gaudin R, Weiser J, et al. Osteonecrosis of the jaw in patients treated with denosumab: a multicenter case series. J Craniomaxillofac Surg. 2018;46:1515-1525. doi: 10.1016/j.jcms.2018.05.046
- Barrett-Connor E, Mosca L, Collins P, et al; Raloxifene Use for The Heart (RUTH) Trial Investigators. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355:125-137. doi: 10.1056/NEJMoa062462
- Chesnut CH 3rd, Skag A, Christiansen C, et al; Oral Ibandronate Osteoporosis Vertebral Fracture Trial in North America and Europe (BONE). Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis. J Bone Miner Res. 2004;19:1241-1249. doi: 10.1359/JBMR.040325
- Gilsenban A, Midkiff K, Kellier-Steele N, et al. Teriparatide did not increase adult osteosarcoma incidence in a 15-year US postmarketing surveillance study. J Bone Miner Res. 2021;36:244-252. doi: 10.1002/jbmr.4188
- Cuzick J, Sestak I, Bonanni B, et al; SERM Chemoprevention of Breast Cancer Overview Group. Selective oestrogen receptor modulators in prevention of breast cancer: an updated meta-analysis of individual participant data. Lancet. 2013;381:1827-1834. doi: 10.1016/S0140-6736(13)60140-3
- Smith MR, Fallon MA, Lee H, et al. Raloxifene to prevent gonadotropin-releasing hormone agonist-induced bone loss in men with prostate cancer: a randomized controlled trial. J Clin Endocrinol Metab. 2004;89:3841-3846. doi: 10.1210/jc.2003-032058
- TYMLOS. Prescribing information. Radius Health, Inc.; April 2017. Accessed May 20, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2017/208743lbl.pdf
- FORTEO. Prescribing information. Eli Lilly and Co.; April 2020. Accessed May 20, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2020/021318s053lbl.pdf
- Wooltorton E. Patients receiving intravenous bisphosphonates should avoid invasive dental procedures. Can Med Assoc J. 2003;172:1684. doi: https://doi.org/10.1503/cmaj.050640
- Chiadika SM, Shobayo FO, Naqvi SH, et al. Lower femoral neck bone mineral density (BMD) in elderly women not on statins. Women Health. 2019;59:845-853. doi: 10.1080/03630242.2019.1567646
- Saraykar S, John V, Cao B, et al. Association of selective serotonin reuptake inhibitors and bone mineral density in elderly women. J Clin Densitom. 2018;21:193-199. doi: 10.1016/j.jocd.2017.05.016
- Lorentzon M, Branco J, Brandi ML, et al. Algorithm for the use of biochemical markers of bone turnover in the diagnosis, assessment and follow-up of treatment for osteoporosis. Adv Ther. 2019;36:2811-2824. doi: 10.1007/s12325-019-01063-9
- STEADI--older adult fall prevention. Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. 2019. Accessed April 28, 2021. www.cdc.gov/steadi/patient.html
- Cummings SR, San Martin J, McClung MR, et al; FREEDOM Trial. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361:756-765. doi: 10.1056/NEJMoa0809493
- Zhou Z, Chen C, Zhang J, et al. Safety of denosumab in postmenopausal women with osteoporosis or low bone mineral density: a meta-analysis. Int J Clin Exp Pathol. 2014;7:2113-2122.
- Faulkner KG. Bone matters: are density increases necessary to reduce fracture risk? J Bone Miner Res. 2000;15:183-187. doi: 10.1359/jbmr.2000.15.2.183
- Rianon N, Anand D, Rasu R. Changing trends in osteoporosis care from specialty to primary care physicians. Curr Med Res Opin. 2013;29:881-888. doi: 10.1185/03007995.2013.809335
- What is osteoporosis and what causes it? National Osteoporosis Foundation Website. 2020. Accessed April 28, 2021. www.nof.org/patients/what-is-osteoporosis/
- des Bordes J, Prasad S, Pratt G, et al. Knowledge, beliefs, and concerns about bone health from a systematic review and metasynthesis of qualitative studies. PLoS One. 2020;15:e0227765. doi: 10.1371/journal.pone.0227765
- Solomon DH, Johnston SS, Boytsov NN, et al. Osteoporosis medication use after hip fracture in U.S. patients between 2002 and 2011. J Bone Miner Res. 2014;29:1929-1937. doi: 10.1002/jbmr.2202
- Cosman F, de Beur SJ, LeBoff MS, et al; National Osteoporosis Foundation. Clinician's guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359-2381. doi: 10.1007/s00198-014-2794-2
- US Preventive Services Task Force; Curry SJ, Krist AH, Owens DK, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:2521-2531. doi: 10.1001/jama.2018.7498
- Camacho PM, Petak SM, Binkley N, et al. American Association of Clinical Endocrinologists and American College of Endocrinology clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis - 2016. Endocr Pract. 2016;22(suppl 4):1-42. doi: 10.4158/EP161435.GL
- Watts NB, Adler RA, Bilezikian JP, et al; Endocrine Society. Osteoporosis in men: an Endocrine Society clinical practice guideline.J Clin Endocrinol Metab. 2012;97:1802-1822. doi: 10.1210/jc.2011-3045
- US Department of Health and Human Services. Bone Health and Osteoporosis: A Report of the Surgeon General. US Department of Health and Human Services, Public Health Service, Office of the Surgeon General; 2004. Accessed April 28, 2021. www.ncbi.nlm.nih.gov/books/NBK45513/pdf/Bookshelf_NBK45513.pdf
- Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser. 1994;843:1-129.
- Looker AC, Frenk SM. Percentage of adults aged 65 and over with osteoporosis or low bone mass at the femur neck or lumbar spine: United States, 2005--2010. Centers for Disease Control and Prevention, National Center for Health Statistics, Division of Health and Nutrition Examination Surveys. August 2015. Accessed April 28, 2021. www.cdc.gov/nchs/data/hestat/osteoporsis/osteoporosis2005_2010.pdf
- Kerschan-Schindl K. Prevention and rehabilitation of osteoporosis. Wien Med Wochenschr. 2016;166:22-27. doi: 10.1007/s10354-015-0417-y
- Tarantino U, Iolascon G, Cianferotti L, et al. Clinical guidelines for the prevention and treatment of osteoporosis: summary statements and recommendations from the Italian Society for Orthopaedics and Traumatology. J Orthop Traumatol. 2017;18(suppl 1):3-36. doi: 10.1007/s10195-017-0474-7
- Martineau P, Leslie WD, Johansson H, et al. In which patients does lumbar spine trabecular bone score (TBS) have the largest effect? Bone. 2018;113:161-168. doi: 10.1016/j.bone.2018.05.026
- Rianon NJ, Smith SM, Lee M, et al. Glycemic control and bone turnover in older Mexican Americans with type 2 diabetes. J Osteoporos. 2018;2018:7153021. doi: 10.1155/2018/7153021
- Richards C, Hans D, Leslie WD. Trabecular bone score (TBS) predicts fracture in ankylosing spondylitis: The Manitoba BMD Registry. J Clin Densitom. 2020;23:543-548. doi: 10.1016/j.jocd.2020.01.003
- Xue Y, Baker AL, Nader S, et al. Lumbar spine trabecular bone score (TBS) reflects diminished bone quality in patients with diabetes mellitus and oral glucocorticoid therapy. J Clin Densitom. 2018;21:185-192. doi: 10.1016/j.jocd.2017.09.003
- Silva BC, Broy SB, Boutroy S, et al. Fracture risk prediction by non-BMD DXA measures: the 2015 ISCD Official Positions Part 2: trabecular bone score. J Clin Densitom. 2015;18:309-330. doi: 10.1016/j.jocd.2015.06.008
- Silva BC, Leslie WD, Resch H, et al. Trabecular bone score: a noninvasive analytical method based upon the DXA image. J Bone Miner Res. 2014;29:518-530. doi: 10.1002/jbmr.2176
- Leslie WD, Aubry-Rozier B, Lamy O, et al; Manitoba Bone Density Program. TBS (trabecular bone score) and diabetes-related fracture risk. J Clin Endocrinol Metab. 2013;98:602-609.
- Looker AC, Sarafrazi Isfahani N, Fan B, et al. Trabecular bone scores and lumbar spine bone mineral density of US adults: comparison of relationships with demographic and body size variables. Osteoporos Int. 2016;27:2467-2475. doi: 10.1007/s00198-016-3550-6
- Rianon N, Ambrose CG, Pervin H, et al. Long-term use of angiotensin-converting enzyme inhibitors protects against bone loss in African-American elderly men. Arch Osteoporos. 2017;12:94. doi: 10.1007/s11657-017-0387-3
- Morton DJ, Barrett-Connor EL, Edelstein SL. Thiazides and bone mineral density in elderly men and women. Am J Epidemiol. 1994;139:1107-1115. doi: 10.1093/oxfordjournals.aje.a116954
- Sigurdsson G, Franzson L. Increased bone mineral density in a population-based group of 70-year-old women on thiazide diuretics, independent of parathyroid hormone levels. J Intern Med. 2001;250:51-56. doi: 10.1046/j.1365-2796.2001.00850.x
- Qaseem A, Forciea MA, McLean RM, et al; Clinical Guidelines Committee of the American College of Physicians. Treatment of low bone density or osteoporosis to prevent fractures in men and women: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017;166:818-839. doi: 10.7326/M15-1361
- des Bordes JKA, Suarez-Almazor ME, Volk RJ, et al. Online educational tool to promote bone health in cancer survivors. J Health Commun. 2017;22:808-817. doi: 10.1080/10810730.2017.1360415
- Shoback D, Rosen CJ, Black DM, et al. Pharmacological management of osteoporosis in postmenopausal women: an Endocrine Society guideline update. J Clin Endocrinol Metab. 2020;105:587-594. doi: 10.1210/clinem/dgaa048
- Black DM, Schwartz AV, Ensrud KE, et al; FLEX Research Group. Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA. 2006;296:2927-2938. doi: 10.1001/jama.296.24.2927
- Bone HG, Hosking D, Devogelaer J-P, et al. Ten years' experience with alendronate for osteoporosis in postmenopausal women. N Engl J Med. 2004;350:1189-1199. doi: 10.1056/NEJMoa030897
- Khosla S, Burr D, Cauley J, et al; American Society for Bone and Mineral Research. Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2007;22:1479-1491. doi: 10.1359/jbmr.0707onj
- Bone HG, Bolognese MA, Yuen CK, et al. Effects of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with low bone mass. J Clin Endocrinol Metab. 2011;96:972-980. doi: 10.1210/jc.2010-1502
- Cummings SR, Ferrari S, Eastell R, et al. Vertebral fractures after discontinuation of denosumab: a post hoc analysis of the randomized placebo-controlled FREEDOM Trial and its extension. J Bone Miner Res. 2018;33:190-198. doi: 10.1002/jbmr.3337
- Symonds C, Kline G. Warning of an increased risk of vertebral fracture after stopping denosumab. CMAJ. 2018;190:E485-E486. doi: 10.1503/cmaj.180115
- Aljohani S, Gaudin R, Weiser J, et al. Osteonecrosis of the jaw in patients treated with denosumab: a multicenter case series. J Craniomaxillofac Surg. 2018;46:1515-1525. doi: 10.1016/j.jcms.2018.05.046
- Barrett-Connor E, Mosca L, Collins P, et al; Raloxifene Use for The Heart (RUTH) Trial Investigators. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355:125-137. doi: 10.1056/NEJMoa062462
- Chesnut CH 3rd, Skag A, Christiansen C, et al; Oral Ibandronate Osteoporosis Vertebral Fracture Trial in North America and Europe (BONE). Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis. J Bone Miner Res. 2004;19:1241-1249. doi: 10.1359/JBMR.040325
- Gilsenban A, Midkiff K, Kellier-Steele N, et al. Teriparatide did not increase adult osteosarcoma incidence in a 15-year US postmarketing surveillance study. J Bone Miner Res. 2021;36:244-252. doi: 10.1002/jbmr.4188
- Cuzick J, Sestak I, Bonanni B, et al; SERM Chemoprevention of Breast Cancer Overview Group. Selective oestrogen receptor modulators in prevention of breast cancer: an updated meta-analysis of individual participant data. Lancet. 2013;381:1827-1834. doi: 10.1016/S0140-6736(13)60140-3
- Smith MR, Fallon MA, Lee H, et al. Raloxifene to prevent gonadotropin-releasing hormone agonist-induced bone loss in men with prostate cancer: a randomized controlled trial. J Clin Endocrinol Metab. 2004;89:3841-3846. doi: 10.1210/jc.2003-032058
- TYMLOS. Prescribing information. Radius Health, Inc.; April 2017. Accessed May 20, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2017/208743lbl.pdf
- FORTEO. Prescribing information. Eli Lilly and Co.; April 2020. Accessed May 20, 2021. www.accessdata.fda.gov/drugsatfda_docs/label/2020/021318s053lbl.pdf
- Wooltorton E. Patients receiving intravenous bisphosphonates should avoid invasive dental procedures. Can Med Assoc J. 2003;172:1684. doi: https://doi.org/10.1503/cmaj.050640
- Chiadika SM, Shobayo FO, Naqvi SH, et al. Lower femoral neck bone mineral density (BMD) in elderly women not on statins. Women Health. 2019;59:845-853. doi: 10.1080/03630242.2019.1567646
- Saraykar S, John V, Cao B, et al. Association of selective serotonin reuptake inhibitors and bone mineral density in elderly women. J Clin Densitom. 2018;21:193-199. doi: 10.1016/j.jocd.2017.05.016
- Lorentzon M, Branco J, Brandi ML, et al. Algorithm for the use of biochemical markers of bone turnover in the diagnosis, assessment and follow-up of treatment for osteoporosis. Adv Ther. 2019;36:2811-2824. doi: 10.1007/s12325-019-01063-9
- STEADI--older adult fall prevention. Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. 2019. Accessed April 28, 2021. www.cdc.gov/steadi/patient.html
- Cummings SR, San Martin J, McClung MR, et al; FREEDOM Trial. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361:756-765. doi: 10.1056/NEJMoa0809493
- Zhou Z, Chen C, Zhang J, et al. Safety of denosumab in postmenopausal women with osteoporosis or low bone mineral density: a meta-analysis. Int J Clin Exp Pathol. 2014;7:2113-2122.
- Faulkner KG. Bone matters: are density increases necessary to reduce fracture risk? J Bone Miner Res. 2000;15:183-187. doi: 10.1359/jbmr.2000.15.2.183
- Rianon N, Anand D, Rasu R. Changing trends in osteoporosis care from specialty to primary care physicians. Curr Med Res Opin. 2013;29:881-888. doi: 10.1185/03007995.2013.809335
PRACTICE RECOMMENDATIONS
❯ Consider screening for osteoporosis, using bone mineral density (BMD) by dual-energy X-ray absorptiometry (DXA), in all postmenopausal women ≥ 65 years and in women < 65 years at high risk of osteoporosis.
❯ Consider screening in men ≥ 70 years and in younger men at high risk of fracture.
❯ Use the trabecular bone score with DXA BMD to screen patients at high risk of fracture who have a normal BMD—eg, patients with type 2 diabetes or ankylosing spondylitis.
❯ Offer individualized pharmacotherapy to older patients with a diagnosis of osteoporosis and to those at high risk of fracture.
Neurodegeneration complicates psychiatric care for Parkinson’s patients
Managing depression and anxiety in Parkinson’s disease should start with a review of medications and involve multidisciplinary care, according to a recent summary of evidence.
“Depression and anxiety have a complex relationship with the disease and while the exact mechanism for this association is unknown, both disturbances occur with increased prevalence across the disease course and when present earlier in life, increase the risk of PD by about twofold,” wrote Gregory M. Pontone, MD, of Johns Hopkins University, Baltimore, and colleagues.
Randomized trials to guide treatment of anxiety and depression in patients with Parkinson’s disease (PD) are limited, the researchers noted. However, data from a longitudinal study showed that PD patients whose depression remitted spontaneously or responded to treatment were able to attain a level of function similar to that of never-depressed PD patients, Dr. Pontone and colleagues said.
The researchers offered a pair of treatment algorithms to help guide clinicians in managing depression and anxiety in PD. However, a caveat to keep in mind is that “the benefit of antidepressant medications, used for depression or anxiety, can be confounded when motor symptoms are not optimally treated,” the researchers emphasized.
For depression, the researchers advised starting with some lab work; “at a minimum we suggest checking a complete blood count, metabolic panel, TSH, B12, and folate,” they noted. They recommended an antidepressant, cognitive-behavioral therapy, or both, as a first-line treatment, such as monotherapy with selective norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors. They advised titrating the chosen monotherapy to a minimum effective dose over a 2- to 3-week period to assess response.
“We recommend continuing antidepressant therapy for at least 1 year based on literature in non-PD populations and anecdotal clinical experience. At 1 year, if not in remission, consider continuing treatment or augmenting to improve response,” the researchers said.
, and they recommended using anxiety rating scales to diagnose anxiety in PD. “Given the high prevalence of atypical anxiety syndromes in PD and their potential association with both motor and nonmotor symptoms of the disease, working with the neurologist to achieve optimal control of PD is an essential first step to alleviating anxiety,” they emphasized.
The researchers also advised addressing comorbidities, including cardiovascular disease, chronic pain, diabetes, gastrointestinal issues, hyperthyroidism, and lung disease, all of which can be associated with anxiety. Once comorbidities are addressed, they advised caution given the lack of evidence for efficacy of both pharmacologic and nonpharmacologic anxiety treatments for PD patients. However, first-tier treatment for anxiety could include monotherapy with serotonin-norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors, they said.
PD patients with depression and anxiety also may benefit from nonpharmacologic interventions, including exercise, mindfulness, relaxation therapy, and cognitive behavioral therapy the researchers said.
Although the algorithm may not differ significantly from current treatment protocols, it highlights aspects unique to PD patients, the researchers said. In particular, the algorithm shows “that interventions used for motor symptoms, for example, dopamine agonists, may be especially potent for mood in the PD population and that augmentation strategies, such as antipsychotics and lithium, may not be well tolerated given their outsized risk of adverse events in PD,” they said.
“While an article of this kind cannot hope to address the gap in knowledge on comparative efficacy between interventions, it can guide readers on the best strategies for implementation and risk mitigation in PD – essentially focusing more on effectiveness,” they concluded.
The study received no outside funding. Dr. Pontone disclosed serving as a consultant for Acadia Pharmaceuticals and Concert Pharmaceuticals.
Managing depression and anxiety in Parkinson’s disease should start with a review of medications and involve multidisciplinary care, according to a recent summary of evidence.
“Depression and anxiety have a complex relationship with the disease and while the exact mechanism for this association is unknown, both disturbances occur with increased prevalence across the disease course and when present earlier in life, increase the risk of PD by about twofold,” wrote Gregory M. Pontone, MD, of Johns Hopkins University, Baltimore, and colleagues.
Randomized trials to guide treatment of anxiety and depression in patients with Parkinson’s disease (PD) are limited, the researchers noted. However, data from a longitudinal study showed that PD patients whose depression remitted spontaneously or responded to treatment were able to attain a level of function similar to that of never-depressed PD patients, Dr. Pontone and colleagues said.
The researchers offered a pair of treatment algorithms to help guide clinicians in managing depression and anxiety in PD. However, a caveat to keep in mind is that “the benefit of antidepressant medications, used for depression or anxiety, can be confounded when motor symptoms are not optimally treated,” the researchers emphasized.
For depression, the researchers advised starting with some lab work; “at a minimum we suggest checking a complete blood count, metabolic panel, TSH, B12, and folate,” they noted. They recommended an antidepressant, cognitive-behavioral therapy, or both, as a first-line treatment, such as monotherapy with selective norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors. They advised titrating the chosen monotherapy to a minimum effective dose over a 2- to 3-week period to assess response.
“We recommend continuing antidepressant therapy for at least 1 year based on literature in non-PD populations and anecdotal clinical experience. At 1 year, if not in remission, consider continuing treatment or augmenting to improve response,” the researchers said.
, and they recommended using anxiety rating scales to diagnose anxiety in PD. “Given the high prevalence of atypical anxiety syndromes in PD and their potential association with both motor and nonmotor symptoms of the disease, working with the neurologist to achieve optimal control of PD is an essential first step to alleviating anxiety,” they emphasized.
The researchers also advised addressing comorbidities, including cardiovascular disease, chronic pain, diabetes, gastrointestinal issues, hyperthyroidism, and lung disease, all of which can be associated with anxiety. Once comorbidities are addressed, they advised caution given the lack of evidence for efficacy of both pharmacologic and nonpharmacologic anxiety treatments for PD patients. However, first-tier treatment for anxiety could include monotherapy with serotonin-norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors, they said.
PD patients with depression and anxiety also may benefit from nonpharmacologic interventions, including exercise, mindfulness, relaxation therapy, and cognitive behavioral therapy the researchers said.
Although the algorithm may not differ significantly from current treatment protocols, it highlights aspects unique to PD patients, the researchers said. In particular, the algorithm shows “that interventions used for motor symptoms, for example, dopamine agonists, may be especially potent for mood in the PD population and that augmentation strategies, such as antipsychotics and lithium, may not be well tolerated given their outsized risk of adverse events in PD,” they said.
“While an article of this kind cannot hope to address the gap in knowledge on comparative efficacy between interventions, it can guide readers on the best strategies for implementation and risk mitigation in PD – essentially focusing more on effectiveness,” they concluded.
The study received no outside funding. Dr. Pontone disclosed serving as a consultant for Acadia Pharmaceuticals and Concert Pharmaceuticals.
Managing depression and anxiety in Parkinson’s disease should start with a review of medications and involve multidisciplinary care, according to a recent summary of evidence.
“Depression and anxiety have a complex relationship with the disease and while the exact mechanism for this association is unknown, both disturbances occur with increased prevalence across the disease course and when present earlier in life, increase the risk of PD by about twofold,” wrote Gregory M. Pontone, MD, of Johns Hopkins University, Baltimore, and colleagues.
Randomized trials to guide treatment of anxiety and depression in patients with Parkinson’s disease (PD) are limited, the researchers noted. However, data from a longitudinal study showed that PD patients whose depression remitted spontaneously or responded to treatment were able to attain a level of function similar to that of never-depressed PD patients, Dr. Pontone and colleagues said.
The researchers offered a pair of treatment algorithms to help guide clinicians in managing depression and anxiety in PD. However, a caveat to keep in mind is that “the benefit of antidepressant medications, used for depression or anxiety, can be confounded when motor symptoms are not optimally treated,” the researchers emphasized.
For depression, the researchers advised starting with some lab work; “at a minimum we suggest checking a complete blood count, metabolic panel, TSH, B12, and folate,” they noted. They recommended an antidepressant, cognitive-behavioral therapy, or both, as a first-line treatment, such as monotherapy with selective norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors. They advised titrating the chosen monotherapy to a minimum effective dose over a 2- to 3-week period to assess response.
“We recommend continuing antidepressant therapy for at least 1 year based on literature in non-PD populations and anecdotal clinical experience. At 1 year, if not in remission, consider continuing treatment or augmenting to improve response,” the researchers said.
, and they recommended using anxiety rating scales to diagnose anxiety in PD. “Given the high prevalence of atypical anxiety syndromes in PD and their potential association with both motor and nonmotor symptoms of the disease, working with the neurologist to achieve optimal control of PD is an essential first step to alleviating anxiety,” they emphasized.
The researchers also advised addressing comorbidities, including cardiovascular disease, chronic pain, diabetes, gastrointestinal issues, hyperthyroidism, and lung disease, all of which can be associated with anxiety. Once comorbidities are addressed, they advised caution given the lack of evidence for efficacy of both pharmacologic and nonpharmacologic anxiety treatments for PD patients. However, first-tier treatment for anxiety could include monotherapy with serotonin-norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors, they said.
PD patients with depression and anxiety also may benefit from nonpharmacologic interventions, including exercise, mindfulness, relaxation therapy, and cognitive behavioral therapy the researchers said.
Although the algorithm may not differ significantly from current treatment protocols, it highlights aspects unique to PD patients, the researchers said. In particular, the algorithm shows “that interventions used for motor symptoms, for example, dopamine agonists, may be especially potent for mood in the PD population and that augmentation strategies, such as antipsychotics and lithium, may not be well tolerated given their outsized risk of adverse events in PD,” they said.
“While an article of this kind cannot hope to address the gap in knowledge on comparative efficacy between interventions, it can guide readers on the best strategies for implementation and risk mitigation in PD – essentially focusing more on effectiveness,” they concluded.
The study received no outside funding. Dr. Pontone disclosed serving as a consultant for Acadia Pharmaceuticals and Concert Pharmaceuticals.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY
Safety-net burden linked with poorer inpatient cirrhosis outcomes
Patients with cirrhosis treated at hospitals with the highest safety-net burden, defined by their proportion of Medicaid or uninsured patients, had a 5% higher mortality rate than patients who were treated at hospitals with the lowest burden, according to a study of over 300,000 patients.
The study, which was published in the Journal of Clinical Gastroenterology, analyzed inpatient data from the National Inpatient Sample (NIS) database focusing on a 4-year time span between 2012 and 2016. The hospitals were categorized by safety-net burden, which was defined as having either a high, medium, or low number of uninsured patients or patients with Medicaid.
This is the first-known study to evaluate the impact of a hospital’s safety-net burden on hospitalization outcomes in cirrhosis patients, wrote authors Robert J. Wong, MD, MS, of Stanford (Calif.) University and Grishma Hirode, MAS, of the University of Toronto. Previous studies have shown that safety-net hospitals, especially those with a high safety-net burden, have poorer patient outcomes. These hospitals also serve a patient population that is at high risk for chronic liver disease and cirrhosis.
The new analysis included 322,944 individual hospitalizations of patients with cirrhosis. Of these, 57.8% were male, 63.7% were White, 9.9% were Black, and 15.6% were Hispanic. In terms of safety-net burden, 107,446 hospitalizations were at high-burden hospitals, 103,508 were at medium-burden hospitals, and 111,990 hospitalizations were at low-burden hospitals.
Overall, cirrhosis-related hospitalizations in hospitals with the highest burden were found to have significantly greater odds of in-hospital mortality than the lowest tertile hospitals (odds ratio, 1.05, P = .044). The patients were also younger (mean age, 56.7 years vs. 59.8 years in low-burden hospitals). They also had a higher proportion of male patients, minority patients, Hispanic patients, and patients with Medicaid or no insurance.
The odds of hospitalization in the highest tertile hospitals were found to be significantly higher, compared with the middle and lowest tertiles for Blacks and Hispanics, compared with Whites (OR 1.26 and OR 1.63, respectively). Black patients (OR, 1.26; 95%CI, 1.17-1.35; P < .001) and Hispanic patients (OR, 1.63; 95% CI, 1.50-1.78; P< .001) were more likely to be admitted for care at high-burden hospitals (26% to 54%). In-hospital mortality rates among all hospitalizations were 5.95% and the rate did not significantly differ by hospital burden status.
“Despite adjusting for safety-net burden, our study continued to demonstrate ethnic disparities in in-hospital mortality among cirrhosis-related hospitalizations,” the researchers wrote. Overall, the odds of in-hospital mortality were 27% higher in Black patients as compared with White patients.
However, significantly lower mortality was observed in Hispanic patients as compared with White patients (4.9% vs. 6.0%, P < .001), but why this occurred was not entirely clear. “Hispanic patients may be more likely to have NASH [nonalcoholic steatohepatitis]-related cirrhosis, which generally has a slower disease progression, compared with [hepatitis C virus] or alcoholic cirrhosis. As such, it is likely that NASH-cirrhosis Hispanic patients had less severe disease at presentation,” the researchers wrote.
Study design has limitations, but shows concerning trends
The study findings were limited by several factors including the inability to show causality based on the observational study design and cross-sectional nature of the database, the researchers said. The NIS database records individual hospitalizations, not individual patient data which means that it may include repeat hospitalizations from the same patient. In addition, the study was limited by a lack of data on outpatient cirrhosis outcomes and non–liver-related comorbidities.
However, the finding that ethnic minorities with cirrhosis were significantly more likely to be hospitalized in high safety-net hospitals than White patients is concerning, and more research is needed, they said.
“These observations highlight that, while disparities in resources and health care delivery inherent to safety-net health systems may partly explain and provide opportunities to improve cirrhosis hospitalization care, they alone do not explain all of the ethnic disparities in cirrhosis outcomes observed,” they concluded.
The current study was important to conduct at this time because rates of cirrhosis are on the rise, Michael Volk, MD, of Loma Linda (Calif.) University Health, said in an interview. “Millions of patients receive care in safety-net hospitals across the country.”
Dr. Volk said that he was not surprised by the overall outcomes. “Unfortunately, I expected that patient outcomes would be worse at safety-net hospitals than wealthier hospitals. However, I was surprised that Blacks had higher in-hospital mortality than Whites, even after adjusting for the hospital.”
Dr. Volk echoed the study’s stated limitation of the lack of data to address disparities.
“Additional research is needed to determine whether the higher in-hospital mortality among Blacks is related to biological differences such as differential rates of disease progression, or social differences such as access to outpatient care,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Volk had no relevant financial conflicts to disclose.
Patients with cirrhosis treated at hospitals with the highest safety-net burden, defined by their proportion of Medicaid or uninsured patients, had a 5% higher mortality rate than patients who were treated at hospitals with the lowest burden, according to a study of over 300,000 patients.
The study, which was published in the Journal of Clinical Gastroenterology, analyzed inpatient data from the National Inpatient Sample (NIS) database focusing on a 4-year time span between 2012 and 2016. The hospitals were categorized by safety-net burden, which was defined as having either a high, medium, or low number of uninsured patients or patients with Medicaid.
This is the first-known study to evaluate the impact of a hospital’s safety-net burden on hospitalization outcomes in cirrhosis patients, wrote authors Robert J. Wong, MD, MS, of Stanford (Calif.) University and Grishma Hirode, MAS, of the University of Toronto. Previous studies have shown that safety-net hospitals, especially those with a high safety-net burden, have poorer patient outcomes. These hospitals also serve a patient population that is at high risk for chronic liver disease and cirrhosis.
The new analysis included 322,944 individual hospitalizations of patients with cirrhosis. Of these, 57.8% were male, 63.7% were White, 9.9% were Black, and 15.6% were Hispanic. In terms of safety-net burden, 107,446 hospitalizations were at high-burden hospitals, 103,508 were at medium-burden hospitals, and 111,990 hospitalizations were at low-burden hospitals.
Overall, cirrhosis-related hospitalizations in hospitals with the highest burden were found to have significantly greater odds of in-hospital mortality than the lowest tertile hospitals (odds ratio, 1.05, P = .044). The patients were also younger (mean age, 56.7 years vs. 59.8 years in low-burden hospitals). They also had a higher proportion of male patients, minority patients, Hispanic patients, and patients with Medicaid or no insurance.
The odds of hospitalization in the highest tertile hospitals were found to be significantly higher, compared with the middle and lowest tertiles for Blacks and Hispanics, compared with Whites (OR 1.26 and OR 1.63, respectively). Black patients (OR, 1.26; 95%CI, 1.17-1.35; P < .001) and Hispanic patients (OR, 1.63; 95% CI, 1.50-1.78; P< .001) were more likely to be admitted for care at high-burden hospitals (26% to 54%). In-hospital mortality rates among all hospitalizations were 5.95% and the rate did not significantly differ by hospital burden status.
“Despite adjusting for safety-net burden, our study continued to demonstrate ethnic disparities in in-hospital mortality among cirrhosis-related hospitalizations,” the researchers wrote. Overall, the odds of in-hospital mortality were 27% higher in Black patients as compared with White patients.
However, significantly lower mortality was observed in Hispanic patients as compared with White patients (4.9% vs. 6.0%, P < .001), but why this occurred was not entirely clear. “Hispanic patients may be more likely to have NASH [nonalcoholic steatohepatitis]-related cirrhosis, which generally has a slower disease progression, compared with [hepatitis C virus] or alcoholic cirrhosis. As such, it is likely that NASH-cirrhosis Hispanic patients had less severe disease at presentation,” the researchers wrote.
Study design has limitations, but shows concerning trends
The study findings were limited by several factors including the inability to show causality based on the observational study design and cross-sectional nature of the database, the researchers said. The NIS database records individual hospitalizations, not individual patient data which means that it may include repeat hospitalizations from the same patient. In addition, the study was limited by a lack of data on outpatient cirrhosis outcomes and non–liver-related comorbidities.
However, the finding that ethnic minorities with cirrhosis were significantly more likely to be hospitalized in high safety-net hospitals than White patients is concerning, and more research is needed, they said.
“These observations highlight that, while disparities in resources and health care delivery inherent to safety-net health systems may partly explain and provide opportunities to improve cirrhosis hospitalization care, they alone do not explain all of the ethnic disparities in cirrhosis outcomes observed,” they concluded.
The current study was important to conduct at this time because rates of cirrhosis are on the rise, Michael Volk, MD, of Loma Linda (Calif.) University Health, said in an interview. “Millions of patients receive care in safety-net hospitals across the country.”
Dr. Volk said that he was not surprised by the overall outcomes. “Unfortunately, I expected that patient outcomes would be worse at safety-net hospitals than wealthier hospitals. However, I was surprised that Blacks had higher in-hospital mortality than Whites, even after adjusting for the hospital.”
Dr. Volk echoed the study’s stated limitation of the lack of data to address disparities.
“Additional research is needed to determine whether the higher in-hospital mortality among Blacks is related to biological differences such as differential rates of disease progression, or social differences such as access to outpatient care,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Volk had no relevant financial conflicts to disclose.
Patients with cirrhosis treated at hospitals with the highest safety-net burden, defined by their proportion of Medicaid or uninsured patients, had a 5% higher mortality rate than patients who were treated at hospitals with the lowest burden, according to a study of over 300,000 patients.
The study, which was published in the Journal of Clinical Gastroenterology, analyzed inpatient data from the National Inpatient Sample (NIS) database focusing on a 4-year time span between 2012 and 2016. The hospitals were categorized by safety-net burden, which was defined as having either a high, medium, or low number of uninsured patients or patients with Medicaid.
This is the first-known study to evaluate the impact of a hospital’s safety-net burden on hospitalization outcomes in cirrhosis patients, wrote authors Robert J. Wong, MD, MS, of Stanford (Calif.) University and Grishma Hirode, MAS, of the University of Toronto. Previous studies have shown that safety-net hospitals, especially those with a high safety-net burden, have poorer patient outcomes. These hospitals also serve a patient population that is at high risk for chronic liver disease and cirrhosis.
The new analysis included 322,944 individual hospitalizations of patients with cirrhosis. Of these, 57.8% were male, 63.7% were White, 9.9% were Black, and 15.6% were Hispanic. In terms of safety-net burden, 107,446 hospitalizations were at high-burden hospitals, 103,508 were at medium-burden hospitals, and 111,990 hospitalizations were at low-burden hospitals.
Overall, cirrhosis-related hospitalizations in hospitals with the highest burden were found to have significantly greater odds of in-hospital mortality than the lowest tertile hospitals (odds ratio, 1.05, P = .044). The patients were also younger (mean age, 56.7 years vs. 59.8 years in low-burden hospitals). They also had a higher proportion of male patients, minority patients, Hispanic patients, and patients with Medicaid or no insurance.
The odds of hospitalization in the highest tertile hospitals were found to be significantly higher, compared with the middle and lowest tertiles for Blacks and Hispanics, compared with Whites (OR 1.26 and OR 1.63, respectively). Black patients (OR, 1.26; 95%CI, 1.17-1.35; P < .001) and Hispanic patients (OR, 1.63; 95% CI, 1.50-1.78; P< .001) were more likely to be admitted for care at high-burden hospitals (26% to 54%). In-hospital mortality rates among all hospitalizations were 5.95% and the rate did not significantly differ by hospital burden status.
“Despite adjusting for safety-net burden, our study continued to demonstrate ethnic disparities in in-hospital mortality among cirrhosis-related hospitalizations,” the researchers wrote. Overall, the odds of in-hospital mortality were 27% higher in Black patients as compared with White patients.
However, significantly lower mortality was observed in Hispanic patients as compared with White patients (4.9% vs. 6.0%, P < .001), but why this occurred was not entirely clear. “Hispanic patients may be more likely to have NASH [nonalcoholic steatohepatitis]-related cirrhosis, which generally has a slower disease progression, compared with [hepatitis C virus] or alcoholic cirrhosis. As such, it is likely that NASH-cirrhosis Hispanic patients had less severe disease at presentation,” the researchers wrote.
Study design has limitations, but shows concerning trends
The study findings were limited by several factors including the inability to show causality based on the observational study design and cross-sectional nature of the database, the researchers said. The NIS database records individual hospitalizations, not individual patient data which means that it may include repeat hospitalizations from the same patient. In addition, the study was limited by a lack of data on outpatient cirrhosis outcomes and non–liver-related comorbidities.
However, the finding that ethnic minorities with cirrhosis were significantly more likely to be hospitalized in high safety-net hospitals than White patients is concerning, and more research is needed, they said.
“These observations highlight that, while disparities in resources and health care delivery inherent to safety-net health systems may partly explain and provide opportunities to improve cirrhosis hospitalization care, they alone do not explain all of the ethnic disparities in cirrhosis outcomes observed,” they concluded.
The current study was important to conduct at this time because rates of cirrhosis are on the rise, Michael Volk, MD, of Loma Linda (Calif.) University Health, said in an interview. “Millions of patients receive care in safety-net hospitals across the country.”
Dr. Volk said that he was not surprised by the overall outcomes. “Unfortunately, I expected that patient outcomes would be worse at safety-net hospitals than wealthier hospitals. However, I was surprised that Blacks had higher in-hospital mortality than Whites, even after adjusting for the hospital.”
Dr. Volk echoed the study’s stated limitation of the lack of data to address disparities.
“Additional research is needed to determine whether the higher in-hospital mortality among Blacks is related to biological differences such as differential rates of disease progression, or social differences such as access to outpatient care,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Volk had no relevant financial conflicts to disclose.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Long-acting injectable antipsychotics deserve first-line consideration
Long-acting injectable (LAI) antipsychotic therapies continue to be largely reserved for patients who already have adherence issues. But most patients, including first-episode patients, should be considering drugs with this mode of delivery for their broad array of advantages, according to an overview presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
“Due to the perception that long-acting injectables are for coercive applications, patients are not hearing about the many reasons this option might be preferable,” reported Diana Perkins, MD, MPH, professor, department of psychiatry, University of North Carolina, Chapel Hill.
According to Dr. Perkins, patients starting treatment for first-episode schizophrenia are particularly unlikely to even hear about injectable therapies even though “these might be the best candidates.”
The advantage of LAI in first-episode patients is that it takes adherence off the table as a risk for treatment failure. Ultimately, many patients educated about LAI in the context of other options, prefer LAI, according to Dr. Perkins, and she provided data to support these assertions.
Of several published articles reporting that patients fail to discuss LAI with patients, one was published last year (J Clin Psychopharmacol. 2020 Jul/Aug;40:346-9). For evidence that patients prefer LAI over pills for antipsychotic treatment, Dr. Perkins cited a study with data from more than 1,400 patients (Patient Prefer Adherence. 2020;14:1093-102).
In this latter analysis, 77% of patients preferred LAI over pills, Dr. Perkins reported at the virtual meeting, presented by MedscapeLive. Moreover, 50% of patients preferred every-3-month injections over monthly injections. For those who preferred LAI, the reasons included the convenience of avoiding daily therapy but also the empowerment derived from feeling healthier from not having to depend on a daily schedule of treatment.
LAI has benefits independent of adherence
“Some patients prefer injectables because the daily pills remind them of their illness even when they are feeling well,” Dr. Perkins said.
By eliminating pills and pill bottles, injections also provide a certain degree of confidentiality, said Dr. Perkins. She said that LAI, which avoids the peaks and troughs inherent in pill taking, provides more consistent drug levels even to the degree that doses can sometimes be lowered and adverse events reduced.
Yet, of all the reasons that patients might want to consider LAI, the most compelling is a lower risk of failure from inadequate adherence to daily pills. The evidence that patients do not take daily pills for chronic diseases, whether schizophrenia or any other illness that requires maintenance regimens for persistent disease control, is extensive and overwhelming, according to Dr. Perkins, who cited multiple studies.
For schizophrenia, with a relapsing and remitting pattern of symptoms, poor adherence is a particular risk. When symptoms abate, the cue and motivation for adherence dissipates.
“Most patients with schizophrenia already have some ambivalence about taking their medication.
When patients forget their daily pills or when the they decide to skip doses when they feel well, they do not necessarily tell their physicians.
Poor adherence often goes unrecognized
“Clinicians are not very good at estimating adherence in their patients,” Dr. Perkins said. While she listed several strategies to help patients adhere to daily pills, she also said that LAI should not be discounted as a means to circumvent this problem altogether.
Importantly, suboptimal adherence by itself places the patient at a high risk for relapse. In one study of partial adherence, delayed doses were associated with increased risk of hospitalization overall, but each increment in further delay led to still more risk (Psychiatr Serv. 2004;55:886-91).
Imperfect adherence to daily pills is very common. In one study evaluating 34,000 veterans with schizophrenia, the proportion with consistently good adherence over the 12 months of study was not quite 40% (J Clin Psychiatry. 2006 Oct;67[10]:1542-50). While most of the remaining were intermittently adherent, more than 15% of the populations was consistently poorly adherent.
Simple regimens and adherence closely linked
In general, the simpler the regimen, the greater the adherence, according to Dr. Perkins. For oral therapies, one pill per day is associated with greater adherence than is more than one. LAI, which is even simpler, is consistently associated with a large relative advantage. In one comparative study, 77% were adherent to LAI versus about 30% (P < .001) over a period of 12 months (Ment Health Clin. 2018 Mar 26;8[2]:56-62).
In a large recently published meta-analysis, LAI was associated with objective advantages over oral therapy (Lancet Psychiatry. 2021 May 1;8:[5]387-404). This included a statistically significant lower risk of hospitalizations or relapses for LAI over oral therapy whether the two modes of delivery were compared in randomized controls studies (RR 0.88; P = .033) or cohort studies (RR 0.92; P = .0044).
Overall, clinicians should discard several false assumptions about LAI, including the fact they are not appropriate in first-episode disease and that patients will not elect to take LAI if informed of the choices, according to Dr. Perkins. Rather than just listing LAI among options, Dr. Perkins suggested that clinicians should specifically explain the many advantages.
“When people say I do not like shots, this is not necessarily a no,” she said. If patients understand the potential for a reduced risk of relapse without having to take a daily pill, injections might be preferable whether or not they like shots in particular.
This is not an isolated view among experienced physicians. John M. Kane, MD, chairman of psychiatry, Zucker Hillside Hospital, Northwell Health, Glen Oaks, N.Y., also sees LAI as method of drug delivery that should be considered widely in schizophrenia.
“I am in complete agreement with Dr. Perkins that long-acting injectable formulations of antipsychotic medications should be presented routinely to patients and families as an option for illness management and relapse prevention,” said Dr. Kane, who was a coauthor of the meta-analysis published in Lancet Psychiatry.
“This would include patients early in the course of illness who currently have an unnecessarily high risk of relapse and hospitalization,” he added.
MedscapeLive and this news organization are owned by the same parent company. Dr. Perkins has a financial relationship with Alkermes. Dr. Kane has financial relationships with Abbott, AstraZeneca, Bristol-Myers Squibb, Janssen, Lilly, Otsuka, Pfizer, and Wyeth.
Long-acting injectable (LAI) antipsychotic therapies continue to be largely reserved for patients who already have adherence issues. But most patients, including first-episode patients, should be considering drugs with this mode of delivery for their broad array of advantages, according to an overview presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
“Due to the perception that long-acting injectables are for coercive applications, patients are not hearing about the many reasons this option might be preferable,” reported Diana Perkins, MD, MPH, professor, department of psychiatry, University of North Carolina, Chapel Hill.
According to Dr. Perkins, patients starting treatment for first-episode schizophrenia are particularly unlikely to even hear about injectable therapies even though “these might be the best candidates.”
The advantage of LAI in first-episode patients is that it takes adherence off the table as a risk for treatment failure. Ultimately, many patients educated about LAI in the context of other options, prefer LAI, according to Dr. Perkins, and she provided data to support these assertions.
Of several published articles reporting that patients fail to discuss LAI with patients, one was published last year (J Clin Psychopharmacol. 2020 Jul/Aug;40:346-9). For evidence that patients prefer LAI over pills for antipsychotic treatment, Dr. Perkins cited a study with data from more than 1,400 patients (Patient Prefer Adherence. 2020;14:1093-102).
In this latter analysis, 77% of patients preferred LAI over pills, Dr. Perkins reported at the virtual meeting, presented by MedscapeLive. Moreover, 50% of patients preferred every-3-month injections over monthly injections. For those who preferred LAI, the reasons included the convenience of avoiding daily therapy but also the empowerment derived from feeling healthier from not having to depend on a daily schedule of treatment.
LAI has benefits independent of adherence
“Some patients prefer injectables because the daily pills remind them of their illness even when they are feeling well,” Dr. Perkins said.
By eliminating pills and pill bottles, injections also provide a certain degree of confidentiality, said Dr. Perkins. She said that LAI, which avoids the peaks and troughs inherent in pill taking, provides more consistent drug levels even to the degree that doses can sometimes be lowered and adverse events reduced.
Yet, of all the reasons that patients might want to consider LAI, the most compelling is a lower risk of failure from inadequate adherence to daily pills. The evidence that patients do not take daily pills for chronic diseases, whether schizophrenia or any other illness that requires maintenance regimens for persistent disease control, is extensive and overwhelming, according to Dr. Perkins, who cited multiple studies.
For schizophrenia, with a relapsing and remitting pattern of symptoms, poor adherence is a particular risk. When symptoms abate, the cue and motivation for adherence dissipates.
“Most patients with schizophrenia already have some ambivalence about taking their medication.
When patients forget their daily pills or when the they decide to skip doses when they feel well, they do not necessarily tell their physicians.
Poor adherence often goes unrecognized
“Clinicians are not very good at estimating adherence in their patients,” Dr. Perkins said. While she listed several strategies to help patients adhere to daily pills, she also said that LAI should not be discounted as a means to circumvent this problem altogether.
Importantly, suboptimal adherence by itself places the patient at a high risk for relapse. In one study of partial adherence, delayed doses were associated with increased risk of hospitalization overall, but each increment in further delay led to still more risk (Psychiatr Serv. 2004;55:886-91).
Imperfect adherence to daily pills is very common. In one study evaluating 34,000 veterans with schizophrenia, the proportion with consistently good adherence over the 12 months of study was not quite 40% (J Clin Psychiatry. 2006 Oct;67[10]:1542-50). While most of the remaining were intermittently adherent, more than 15% of the populations was consistently poorly adherent.
Simple regimens and adherence closely linked
In general, the simpler the regimen, the greater the adherence, according to Dr. Perkins. For oral therapies, one pill per day is associated with greater adherence than is more than one. LAI, which is even simpler, is consistently associated with a large relative advantage. In one comparative study, 77% were adherent to LAI versus about 30% (P < .001) over a period of 12 months (Ment Health Clin. 2018 Mar 26;8[2]:56-62).
In a large recently published meta-analysis, LAI was associated with objective advantages over oral therapy (Lancet Psychiatry. 2021 May 1;8:[5]387-404). This included a statistically significant lower risk of hospitalizations or relapses for LAI over oral therapy whether the two modes of delivery were compared in randomized controls studies (RR 0.88; P = .033) or cohort studies (RR 0.92; P = .0044).
Overall, clinicians should discard several false assumptions about LAI, including the fact they are not appropriate in first-episode disease and that patients will not elect to take LAI if informed of the choices, according to Dr. Perkins. Rather than just listing LAI among options, Dr. Perkins suggested that clinicians should specifically explain the many advantages.
“When people say I do not like shots, this is not necessarily a no,” she said. If patients understand the potential for a reduced risk of relapse without having to take a daily pill, injections might be preferable whether or not they like shots in particular.
This is not an isolated view among experienced physicians. John M. Kane, MD, chairman of psychiatry, Zucker Hillside Hospital, Northwell Health, Glen Oaks, N.Y., also sees LAI as method of drug delivery that should be considered widely in schizophrenia.
“I am in complete agreement with Dr. Perkins that long-acting injectable formulations of antipsychotic medications should be presented routinely to patients and families as an option for illness management and relapse prevention,” said Dr. Kane, who was a coauthor of the meta-analysis published in Lancet Psychiatry.
“This would include patients early in the course of illness who currently have an unnecessarily high risk of relapse and hospitalization,” he added.
MedscapeLive and this news organization are owned by the same parent company. Dr. Perkins has a financial relationship with Alkermes. Dr. Kane has financial relationships with Abbott, AstraZeneca, Bristol-Myers Squibb, Janssen, Lilly, Otsuka, Pfizer, and Wyeth.
Long-acting injectable (LAI) antipsychotic therapies continue to be largely reserved for patients who already have adherence issues. But most patients, including first-episode patients, should be considering drugs with this mode of delivery for their broad array of advantages, according to an overview presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
“Due to the perception that long-acting injectables are for coercive applications, patients are not hearing about the many reasons this option might be preferable,” reported Diana Perkins, MD, MPH, professor, department of psychiatry, University of North Carolina, Chapel Hill.
According to Dr. Perkins, patients starting treatment for first-episode schizophrenia are particularly unlikely to even hear about injectable therapies even though “these might be the best candidates.”
The advantage of LAI in first-episode patients is that it takes adherence off the table as a risk for treatment failure. Ultimately, many patients educated about LAI in the context of other options, prefer LAI, according to Dr. Perkins, and she provided data to support these assertions.
Of several published articles reporting that patients fail to discuss LAI with patients, one was published last year (J Clin Psychopharmacol. 2020 Jul/Aug;40:346-9). For evidence that patients prefer LAI over pills for antipsychotic treatment, Dr. Perkins cited a study with data from more than 1,400 patients (Patient Prefer Adherence. 2020;14:1093-102).
In this latter analysis, 77% of patients preferred LAI over pills, Dr. Perkins reported at the virtual meeting, presented by MedscapeLive. Moreover, 50% of patients preferred every-3-month injections over monthly injections. For those who preferred LAI, the reasons included the convenience of avoiding daily therapy but also the empowerment derived from feeling healthier from not having to depend on a daily schedule of treatment.
LAI has benefits independent of adherence
“Some patients prefer injectables because the daily pills remind them of their illness even when they are feeling well,” Dr. Perkins said.
By eliminating pills and pill bottles, injections also provide a certain degree of confidentiality, said Dr. Perkins. She said that LAI, which avoids the peaks and troughs inherent in pill taking, provides more consistent drug levels even to the degree that doses can sometimes be lowered and adverse events reduced.
Yet, of all the reasons that patients might want to consider LAI, the most compelling is a lower risk of failure from inadequate adherence to daily pills. The evidence that patients do not take daily pills for chronic diseases, whether schizophrenia or any other illness that requires maintenance regimens for persistent disease control, is extensive and overwhelming, according to Dr. Perkins, who cited multiple studies.
For schizophrenia, with a relapsing and remitting pattern of symptoms, poor adherence is a particular risk. When symptoms abate, the cue and motivation for adherence dissipates.
“Most patients with schizophrenia already have some ambivalence about taking their medication.
When patients forget their daily pills or when the they decide to skip doses when they feel well, they do not necessarily tell their physicians.
Poor adherence often goes unrecognized
“Clinicians are not very good at estimating adherence in their patients,” Dr. Perkins said. While she listed several strategies to help patients adhere to daily pills, she also said that LAI should not be discounted as a means to circumvent this problem altogether.
Importantly, suboptimal adherence by itself places the patient at a high risk for relapse. In one study of partial adherence, delayed doses were associated with increased risk of hospitalization overall, but each increment in further delay led to still more risk (Psychiatr Serv. 2004;55:886-91).
Imperfect adherence to daily pills is very common. In one study evaluating 34,000 veterans with schizophrenia, the proportion with consistently good adherence over the 12 months of study was not quite 40% (J Clin Psychiatry. 2006 Oct;67[10]:1542-50). While most of the remaining were intermittently adherent, more than 15% of the populations was consistently poorly adherent.
Simple regimens and adherence closely linked
In general, the simpler the regimen, the greater the adherence, according to Dr. Perkins. For oral therapies, one pill per day is associated with greater adherence than is more than one. LAI, which is even simpler, is consistently associated with a large relative advantage. In one comparative study, 77% were adherent to LAI versus about 30% (P < .001) over a period of 12 months (Ment Health Clin. 2018 Mar 26;8[2]:56-62).
In a large recently published meta-analysis, LAI was associated with objective advantages over oral therapy (Lancet Psychiatry. 2021 May 1;8:[5]387-404). This included a statistically significant lower risk of hospitalizations or relapses for LAI over oral therapy whether the two modes of delivery were compared in randomized controls studies (RR 0.88; P = .033) or cohort studies (RR 0.92; P = .0044).
Overall, clinicians should discard several false assumptions about LAI, including the fact they are not appropriate in first-episode disease and that patients will not elect to take LAI if informed of the choices, according to Dr. Perkins. Rather than just listing LAI among options, Dr. Perkins suggested that clinicians should specifically explain the many advantages.
“When people say I do not like shots, this is not necessarily a no,” she said. If patients understand the potential for a reduced risk of relapse without having to take a daily pill, injections might be preferable whether or not they like shots in particular.
This is not an isolated view among experienced physicians. John M. Kane, MD, chairman of psychiatry, Zucker Hillside Hospital, Northwell Health, Glen Oaks, N.Y., also sees LAI as method of drug delivery that should be considered widely in schizophrenia.
“I am in complete agreement with Dr. Perkins that long-acting injectable formulations of antipsychotic medications should be presented routinely to patients and families as an option for illness management and relapse prevention,” said Dr. Kane, who was a coauthor of the meta-analysis published in Lancet Psychiatry.
“This would include patients early in the course of illness who currently have an unnecessarily high risk of relapse and hospitalization,” he added.
MedscapeLive and this news organization are owned by the same parent company. Dr. Perkins has a financial relationship with Alkermes. Dr. Kane has financial relationships with Abbott, AstraZeneca, Bristol-Myers Squibb, Janssen, Lilly, Otsuka, Pfizer, and Wyeth.
FROM CP/AACP PSYCHIATRY UPDATE
Diaphragmatic endometriosis diagnosed many years after symptom onset
Diaphragmatic endometriosis is often diagnosed several years after the start of symptoms – mainly moderate to severe pain – and this is potentially because of general lack of awareness of diaphragmatic endometriosis among the general population and medical professionals.
Findings of the international survey that explored the diagnosis and treatment of diaphragmatic endometriosis were presented at this year’s Royal College of Obstetricians and Gynecologists 2021 Virtual World Congress by medical student Rachel Piccus, MSc, based at the University of Birmingham (England). Robert Sutcliffe, MD, consultant in hepatobiliary and pancreatic surgery, at Queen Elizabeth Hospital Birmingham was senior author. Results were also published in the May 2021 issue of the European Journal of Obstetrics and Gynaecology and Reproductive Biology.
The study found that it took an average of five visits to a primary physician before referral to a gynecologist.
“Late diagnosis could also be due to the idea that diaphragmatic endometriosis symptoms often present before pelvic symptoms and therefore the site of pain is considered atypical for pelvic endometriosis,” Ms. Piccus said, adding that “clinicians are screening for cyclical pain, which is typical of endometriosis, but our study has shown that pain can in fact be more frequent – daily and weekly.”
These significant diagnostic delays, seen from the time of the initial primary care and gynecology consultation has the potential to significantly affect quality of life as seen in pelvic endometriosis, said Ms. Piccus. “These delays are partly due to a lack of awareness among gynecologists, but could also be due to pelvic laparoscopy being insufficient to examine the diaphragm behind the liver.”
Justin Clark, MD, consultant gynaecologist, Birmingham (England) Women’s and Children Hospital, moderated the session and agreed that the study highlights the need for greater awareness of this variant of endometriosis. “Whilst endometriosis affecting the diaphragm, subdiaphragm, and thorax is rare, the condition causes substantial morbidity.”
“Greater knowledge of thoracic endometriosis amongst clinicians in both primary and secondary care is needed to ensure accurate and timely diagnosis,” he added.
Diaphragmatic endometriosis is estimated to affect 1%-1.5% of all endometriosis patients and presents as cyclical pain in the chest, abdomen, and shoulder tip, as well as other respiratory symptoms such as catamenial pneumothorax and difficulty breathing.
“Cross-sectional imaging has shown low sensitivity so upper abdominal laparoscopy is the gold standard; however, this has implications for diagnostic delay because a strong clinical suspicion is required to refer for this invasive procedure,” explained Ms. Piccus referring to one of the reasons underpinning the need for the study.
When successfully diagnosed, treatment requires excision or ablation surgery and studies show symptomatic relief in 75%-100% of cases.
To gauge the extent of delayed diagnosis as well as treatment outcomes from a patient perspective, Ms. Piccus circulated an anonymous online survey among women with a previous history of surgery for diaphragmatic endometriosis.
Diaphragmatic endometriosis pain – daily and weekly as well as cyclical
A total of 137 participants responded to the survey, with a median age of 34 years (range, 19-53). Median age of diaphragmatic endometriosis onset was 27 years (range, 11-50), and importantly, diaphragmatic endometriosis symptoms started before pelvic symptoms in 90 respondents (66%).
The dominant symptom was pain. A total of 38% reported cyclical pain (related to endometrial shedding during menstruation), 15% weekly pain, and 47% daily pain, both of which were worse during the menstrual cycle. Furthermore, 14% reported other symptoms including catamenial pneumothorax, difficulty breathing, and hemoptysis.
“Whilst this cyclical pain is typical of endometriosis, we see that diagnostic delays may be due to misdiagnosis because clinicians are screening for this cyclical pain whilst our study has shown that pain can in fact be more frequent, being daily and weekly,” noted Ms. Piccus. Moderate to severe pain was reported in 67% of respondents and moderate in 31%, only 2% reported pain as mild.
Location of pain comprised moderate to severe pain in the upper abdomen (68%), chest (64%) and shoulder (54%). Pain was right-sided in 54%, left-sided in 11% and bilateral in 35%. Upper back and neck were also reported as sites of pain.
Indirectly providing a measure of the lack of awareness of diaphragmatic endometriosis on behalf of primary care, 122 participants reported initially visiting their primary care physician for help and 65 were given a diagnosis – in only 14 cases was that diaphragmatic endometriosis. There were a range of other gynecologic (e.g. ovarian cyst, two), respiratory (spontaneous pneumothorax, seven), gastrointestinal (gastritis/reflux, eight), musculoskeletal (six), and psychological (anxiety/stress, four) diagnoses.
A median of 5 primary care consultations (range, 1-100) were required before referral to a gynecologist, with 30% seeing a primary care physician over 10 times. A further 14 patients self-referred to gynecologist.
“These findings have implications for diagnostic delay, added Ms. Piccus. “While the majority of respondents were diagnosed less than a year from the first GP visit, the median delay was 2 years, with 31% diagnosed after 5 or more years. One took 23 years for an initial diagnosis.”
Most cases were diagnosed at the time of surgery – 93%, with 52% at pelvic laparoscopy, 35% upper abdominal laparoscopy, with 30% requiring two or more laparoscopies before they were diagnosed with diaphragmatic endometriosis. A total of 7% were diagnosed via cross-sectional imaging prior to surgery.
Treatment outcomes for diaphragmatic endometriosis
Reflecting the literature, surgery to remove the endometriosis lesions was mainly laparoscopic with 47% abdominal excisions, and 29% abdominal ablations; 6% received open abdominal procedures, and 18% received open thoracotomy or video-assisted thoracoscopic surgery.
The survey asked about postoperative symptoms 6 months after surgery and at the time of survey. Symptoms at 6 months post surgery had completely resolved in 18%, shown significant improvement in 48%, and no improvement in 20%. Worsening of symptoms was seen in 14%. Long-term pain was reported by 21% as severe, 27% as moderate, 35% as mild, and 17% had no symptoms.
Further findings included that 23% underwent additional procedures to treat their diaphragmatic endometriosis, and that there was no significant difference between excision and ablation, nor between age of onset of symptoms or length of diagnostic delay.
“Surgical treatment to remove these extra pelvic deposits of endometriosis will depend upon the type and distribution of thoracic endometriosis and a variety of surgical specialties may need to be involved including gynecologists, cardiothoracic, and upper gastrointestinal/liver surgeons,” Dr. Clark said.
He added that familiar hormonal medical treatments for more typical pelvic endometriosis should also be considered for primary and maintenance treatment. “These data suggest a high symptomatic recurrence rate after surgical treatment and so medical treatments should be considered to try and minimize the risks of endometriosis symptoms returning.”
Dr. Clark also pointed out that multidisciplinary clinical teams should be established in specialized centers to plan surgical and medical management to enhance clinical outcomes and collect data to better understand this enigmatic condition.
Ms. Piccus and Dr. Clark have no relevant conflicts of interest.
Diaphragmatic endometriosis is often diagnosed several years after the start of symptoms – mainly moderate to severe pain – and this is potentially because of general lack of awareness of diaphragmatic endometriosis among the general population and medical professionals.
Findings of the international survey that explored the diagnosis and treatment of diaphragmatic endometriosis were presented at this year’s Royal College of Obstetricians and Gynecologists 2021 Virtual World Congress by medical student Rachel Piccus, MSc, based at the University of Birmingham (England). Robert Sutcliffe, MD, consultant in hepatobiliary and pancreatic surgery, at Queen Elizabeth Hospital Birmingham was senior author. Results were also published in the May 2021 issue of the European Journal of Obstetrics and Gynaecology and Reproductive Biology.
The study found that it took an average of five visits to a primary physician before referral to a gynecologist.
“Late diagnosis could also be due to the idea that diaphragmatic endometriosis symptoms often present before pelvic symptoms and therefore the site of pain is considered atypical for pelvic endometriosis,” Ms. Piccus said, adding that “clinicians are screening for cyclical pain, which is typical of endometriosis, but our study has shown that pain can in fact be more frequent – daily and weekly.”
These significant diagnostic delays, seen from the time of the initial primary care and gynecology consultation has the potential to significantly affect quality of life as seen in pelvic endometriosis, said Ms. Piccus. “These delays are partly due to a lack of awareness among gynecologists, but could also be due to pelvic laparoscopy being insufficient to examine the diaphragm behind the liver.”
Justin Clark, MD, consultant gynaecologist, Birmingham (England) Women’s and Children Hospital, moderated the session and agreed that the study highlights the need for greater awareness of this variant of endometriosis. “Whilst endometriosis affecting the diaphragm, subdiaphragm, and thorax is rare, the condition causes substantial morbidity.”
“Greater knowledge of thoracic endometriosis amongst clinicians in both primary and secondary care is needed to ensure accurate and timely diagnosis,” he added.
Diaphragmatic endometriosis is estimated to affect 1%-1.5% of all endometriosis patients and presents as cyclical pain in the chest, abdomen, and shoulder tip, as well as other respiratory symptoms such as catamenial pneumothorax and difficulty breathing.
“Cross-sectional imaging has shown low sensitivity so upper abdominal laparoscopy is the gold standard; however, this has implications for diagnostic delay because a strong clinical suspicion is required to refer for this invasive procedure,” explained Ms. Piccus referring to one of the reasons underpinning the need for the study.
When successfully diagnosed, treatment requires excision or ablation surgery and studies show symptomatic relief in 75%-100% of cases.
To gauge the extent of delayed diagnosis as well as treatment outcomes from a patient perspective, Ms. Piccus circulated an anonymous online survey among women with a previous history of surgery for diaphragmatic endometriosis.
Diaphragmatic endometriosis pain – daily and weekly as well as cyclical
A total of 137 participants responded to the survey, with a median age of 34 years (range, 19-53). Median age of diaphragmatic endometriosis onset was 27 years (range, 11-50), and importantly, diaphragmatic endometriosis symptoms started before pelvic symptoms in 90 respondents (66%).
The dominant symptom was pain. A total of 38% reported cyclical pain (related to endometrial shedding during menstruation), 15% weekly pain, and 47% daily pain, both of which were worse during the menstrual cycle. Furthermore, 14% reported other symptoms including catamenial pneumothorax, difficulty breathing, and hemoptysis.
“Whilst this cyclical pain is typical of endometriosis, we see that diagnostic delays may be due to misdiagnosis because clinicians are screening for this cyclical pain whilst our study has shown that pain can in fact be more frequent, being daily and weekly,” noted Ms. Piccus. Moderate to severe pain was reported in 67% of respondents and moderate in 31%, only 2% reported pain as mild.
Location of pain comprised moderate to severe pain in the upper abdomen (68%), chest (64%) and shoulder (54%). Pain was right-sided in 54%, left-sided in 11% and bilateral in 35%. Upper back and neck were also reported as sites of pain.
Indirectly providing a measure of the lack of awareness of diaphragmatic endometriosis on behalf of primary care, 122 participants reported initially visiting their primary care physician for help and 65 were given a diagnosis – in only 14 cases was that diaphragmatic endometriosis. There were a range of other gynecologic (e.g. ovarian cyst, two), respiratory (spontaneous pneumothorax, seven), gastrointestinal (gastritis/reflux, eight), musculoskeletal (six), and psychological (anxiety/stress, four) diagnoses.
A median of 5 primary care consultations (range, 1-100) were required before referral to a gynecologist, with 30% seeing a primary care physician over 10 times. A further 14 patients self-referred to gynecologist.
“These findings have implications for diagnostic delay, added Ms. Piccus. “While the majority of respondents were diagnosed less than a year from the first GP visit, the median delay was 2 years, with 31% diagnosed after 5 or more years. One took 23 years for an initial diagnosis.”
Most cases were diagnosed at the time of surgery – 93%, with 52% at pelvic laparoscopy, 35% upper abdominal laparoscopy, with 30% requiring two or more laparoscopies before they were diagnosed with diaphragmatic endometriosis. A total of 7% were diagnosed via cross-sectional imaging prior to surgery.
Treatment outcomes for diaphragmatic endometriosis
Reflecting the literature, surgery to remove the endometriosis lesions was mainly laparoscopic with 47% abdominal excisions, and 29% abdominal ablations; 6% received open abdominal procedures, and 18% received open thoracotomy or video-assisted thoracoscopic surgery.
The survey asked about postoperative symptoms 6 months after surgery and at the time of survey. Symptoms at 6 months post surgery had completely resolved in 18%, shown significant improvement in 48%, and no improvement in 20%. Worsening of symptoms was seen in 14%. Long-term pain was reported by 21% as severe, 27% as moderate, 35% as mild, and 17% had no symptoms.
Further findings included that 23% underwent additional procedures to treat their diaphragmatic endometriosis, and that there was no significant difference between excision and ablation, nor between age of onset of symptoms or length of diagnostic delay.
“Surgical treatment to remove these extra pelvic deposits of endometriosis will depend upon the type and distribution of thoracic endometriosis and a variety of surgical specialties may need to be involved including gynecologists, cardiothoracic, and upper gastrointestinal/liver surgeons,” Dr. Clark said.
He added that familiar hormonal medical treatments for more typical pelvic endometriosis should also be considered for primary and maintenance treatment. “These data suggest a high symptomatic recurrence rate after surgical treatment and so medical treatments should be considered to try and minimize the risks of endometriosis symptoms returning.”
Dr. Clark also pointed out that multidisciplinary clinical teams should be established in specialized centers to plan surgical and medical management to enhance clinical outcomes and collect data to better understand this enigmatic condition.
Ms. Piccus and Dr. Clark have no relevant conflicts of interest.
Diaphragmatic endometriosis is often diagnosed several years after the start of symptoms – mainly moderate to severe pain – and this is potentially because of general lack of awareness of diaphragmatic endometriosis among the general population and medical professionals.
Findings of the international survey that explored the diagnosis and treatment of diaphragmatic endometriosis were presented at this year’s Royal College of Obstetricians and Gynecologists 2021 Virtual World Congress by medical student Rachel Piccus, MSc, based at the University of Birmingham (England). Robert Sutcliffe, MD, consultant in hepatobiliary and pancreatic surgery, at Queen Elizabeth Hospital Birmingham was senior author. Results were also published in the May 2021 issue of the European Journal of Obstetrics and Gynaecology and Reproductive Biology.
The study found that it took an average of five visits to a primary physician before referral to a gynecologist.
“Late diagnosis could also be due to the idea that diaphragmatic endometriosis symptoms often present before pelvic symptoms and therefore the site of pain is considered atypical for pelvic endometriosis,” Ms. Piccus said, adding that “clinicians are screening for cyclical pain, which is typical of endometriosis, but our study has shown that pain can in fact be more frequent – daily and weekly.”
These significant diagnostic delays, seen from the time of the initial primary care and gynecology consultation has the potential to significantly affect quality of life as seen in pelvic endometriosis, said Ms. Piccus. “These delays are partly due to a lack of awareness among gynecologists, but could also be due to pelvic laparoscopy being insufficient to examine the diaphragm behind the liver.”
Justin Clark, MD, consultant gynaecologist, Birmingham (England) Women’s and Children Hospital, moderated the session and agreed that the study highlights the need for greater awareness of this variant of endometriosis. “Whilst endometriosis affecting the diaphragm, subdiaphragm, and thorax is rare, the condition causes substantial morbidity.”
“Greater knowledge of thoracic endometriosis amongst clinicians in both primary and secondary care is needed to ensure accurate and timely diagnosis,” he added.
Diaphragmatic endometriosis is estimated to affect 1%-1.5% of all endometriosis patients and presents as cyclical pain in the chest, abdomen, and shoulder tip, as well as other respiratory symptoms such as catamenial pneumothorax and difficulty breathing.
“Cross-sectional imaging has shown low sensitivity so upper abdominal laparoscopy is the gold standard; however, this has implications for diagnostic delay because a strong clinical suspicion is required to refer for this invasive procedure,” explained Ms. Piccus referring to one of the reasons underpinning the need for the study.
When successfully diagnosed, treatment requires excision or ablation surgery and studies show symptomatic relief in 75%-100% of cases.
To gauge the extent of delayed diagnosis as well as treatment outcomes from a patient perspective, Ms. Piccus circulated an anonymous online survey among women with a previous history of surgery for diaphragmatic endometriosis.
Diaphragmatic endometriosis pain – daily and weekly as well as cyclical
A total of 137 participants responded to the survey, with a median age of 34 years (range, 19-53). Median age of diaphragmatic endometriosis onset was 27 years (range, 11-50), and importantly, diaphragmatic endometriosis symptoms started before pelvic symptoms in 90 respondents (66%).
The dominant symptom was pain. A total of 38% reported cyclical pain (related to endometrial shedding during menstruation), 15% weekly pain, and 47% daily pain, both of which were worse during the menstrual cycle. Furthermore, 14% reported other symptoms including catamenial pneumothorax, difficulty breathing, and hemoptysis.
“Whilst this cyclical pain is typical of endometriosis, we see that diagnostic delays may be due to misdiagnosis because clinicians are screening for this cyclical pain whilst our study has shown that pain can in fact be more frequent, being daily and weekly,” noted Ms. Piccus. Moderate to severe pain was reported in 67% of respondents and moderate in 31%, only 2% reported pain as mild.
Location of pain comprised moderate to severe pain in the upper abdomen (68%), chest (64%) and shoulder (54%). Pain was right-sided in 54%, left-sided in 11% and bilateral in 35%. Upper back and neck were also reported as sites of pain.
Indirectly providing a measure of the lack of awareness of diaphragmatic endometriosis on behalf of primary care, 122 participants reported initially visiting their primary care physician for help and 65 were given a diagnosis – in only 14 cases was that diaphragmatic endometriosis. There were a range of other gynecologic (e.g. ovarian cyst, two), respiratory (spontaneous pneumothorax, seven), gastrointestinal (gastritis/reflux, eight), musculoskeletal (six), and psychological (anxiety/stress, four) diagnoses.
A median of 5 primary care consultations (range, 1-100) were required before referral to a gynecologist, with 30% seeing a primary care physician over 10 times. A further 14 patients self-referred to gynecologist.
“These findings have implications for diagnostic delay, added Ms. Piccus. “While the majority of respondents were diagnosed less than a year from the first GP visit, the median delay was 2 years, with 31% diagnosed after 5 or more years. One took 23 years for an initial diagnosis.”
Most cases were diagnosed at the time of surgery – 93%, with 52% at pelvic laparoscopy, 35% upper abdominal laparoscopy, with 30% requiring two or more laparoscopies before they were diagnosed with diaphragmatic endometriosis. A total of 7% were diagnosed via cross-sectional imaging prior to surgery.
Treatment outcomes for diaphragmatic endometriosis
Reflecting the literature, surgery to remove the endometriosis lesions was mainly laparoscopic with 47% abdominal excisions, and 29% abdominal ablations; 6% received open abdominal procedures, and 18% received open thoracotomy or video-assisted thoracoscopic surgery.
The survey asked about postoperative symptoms 6 months after surgery and at the time of survey. Symptoms at 6 months post surgery had completely resolved in 18%, shown significant improvement in 48%, and no improvement in 20%. Worsening of symptoms was seen in 14%. Long-term pain was reported by 21% as severe, 27% as moderate, 35% as mild, and 17% had no symptoms.
Further findings included that 23% underwent additional procedures to treat their diaphragmatic endometriosis, and that there was no significant difference between excision and ablation, nor between age of onset of symptoms or length of diagnostic delay.
“Surgical treatment to remove these extra pelvic deposits of endometriosis will depend upon the type and distribution of thoracic endometriosis and a variety of surgical specialties may need to be involved including gynecologists, cardiothoracic, and upper gastrointestinal/liver surgeons,” Dr. Clark said.
He added that familiar hormonal medical treatments for more typical pelvic endometriosis should also be considered for primary and maintenance treatment. “These data suggest a high symptomatic recurrence rate after surgical treatment and so medical treatments should be considered to try and minimize the risks of endometriosis symptoms returning.”
Dr. Clark also pointed out that multidisciplinary clinical teams should be established in specialized centers to plan surgical and medical management to enhance clinical outcomes and collect data to better understand this enigmatic condition.
Ms. Piccus and Dr. Clark have no relevant conflicts of interest.
Allergic conjunctivitis severely affects children’s quality of life
Allergic conjunctivitis harms quality of life for children and their parents, apparently causing greater day-to-day worries than potentially blinding diseases, researchers report.
Parents worry especially that treatments might not be effective, according to Shi-yao Zhang, MD, and colleagues from Sun Yat-sen University, Guangzhou, China. “This finding suggests that more communication with parents regarding treatment and prognosis is needed,” they write in an article published online June 10 in JAMA Ophthalmology.
One of the most prevalent eye disorders in children, allergic conjunctivitis is often chronic, leading patients to ask repeatedly for help from physicians. It can take an emotional toll and can cause children to miss school.
“With any sign of a slightly pink eye [or a] runny nose, which are very common with allergies, children are being sent home, because everyone’s concerned about COVID,” said Yi Ning J. Strube, MD, an associate professor of ophthalmology and pediatrics at Queen’s University, Kingston, Canada, whose commentary appears in the same issue of JAMA Ophthalmology.
Adolescents are also sometimes accused of smoking cannabis because of their red eyes, she said.
However, little research has examined the effects of allergic conjunctivitis on the quality of life of children and their guardians, Dr. Zhang and colleagues write. To fill that gap, the researchers administered the Pediatric Quality of Life Inventory (PedsQL) to 92 children with allergic conjunctivitis and their parents. The children were aged 5 to 18 years.
The researchers administered the same questionnaire to 96 healthy children of the same ages, along with their parents. These participants served as a control group.
On a scale of 0 to 100, in which a higher score signifies a better quality of life, the median total PedsQL score was 69.6 for children with allergic conjunctivitis versus 96.7 for the control group.
Subscores of physical, emotional, social, and especially school functioning were all significantly lower for the children with allergic conjunctivitis than for the control persons. “Because children generally spend most of their time in the school environment, this outcome raises an issue regarding whether children have a poorer performance in their education,” Dr. Zhang and colleagues write.
Dr. Strube recommends that physicians educate their patients about allergic conjunctivitis using handouts or high-quality websites. She often refers patients and their families to the allergic conjunctivitis webpage of the American Academy of Pediatric Ophthalmology and Strabismus.
She tells parents to have their child “take a shower and wash their hair when they get home before they rub their pollen-filled hair on their pillowcase and make their allergy symptoms worse.”
Parents and schools should try to filter pollen and other allergens from indoor air, she added.
Parents of the children with allergic conjunctivitis in the study also reported lower quality of life; they scored 68.8, versus 96.5 for parents of children in the control group. The differences for both parents and children were statistically significant (P < .001). Overall, the parents’ quality-of-life scores correlated with their children’s (correlation coefficient, r = 0.59; P < .001).
Children with vernal or atopic keratoconjunctivitis scored 3.3 points lower on health-related quality of life than those with seasonal allergic conjunctivitis.
Children with higher corneal fluorescein staining scores also had lower quality-of-life scores. Parents whose children had higher corneal fluorescein staining scores and also those who had multiple consultations with health care practitioners also reported lower quality of life.
The quality-of-life scores of the children with allergic conjunctivitis were lower than scores in previous studies for children with vision-threatening diseases, such as glaucoma and congenital cataract. This may be because glaucoma and cataracts do not typically cause discomfort even if they impair the patient’s vision, said Dr. Strube.
She pointed out one potential flaw in the study: In the cohort with allergic conjunctivitis, 83.7% were boys, compared to 42.7% of the control group. Vernal keratoconjunctivitis affects more boys than girls, and not controlling for this factor could have confounded the data, Dr. Strube said.
It could also be useful to replicate the study in other countries to see whether geographic or cultural factors affected the results, she said. “A lot of these big centers around the world, including in China, have poor air quality, so that may be contributing to patients’ symptoms,” she said. “With regards to reported health quality of life and impact on education, results from different parts of the world may be different, due to parenting styles and education styles,” she said.
The study was supported by the National Natural Science Foundation of China and the Science Foundation of Guangdong Province. Dr. Zhang and colleagues reported no relevant financial relationships. Dr. Strube reported receiving personal fees from Santen Canada Advisory Board Consultant outside the submitted work.
A version of this article first appeared on Medscape.com.
Allergic conjunctivitis harms quality of life for children and their parents, apparently causing greater day-to-day worries than potentially blinding diseases, researchers report.
Parents worry especially that treatments might not be effective, according to Shi-yao Zhang, MD, and colleagues from Sun Yat-sen University, Guangzhou, China. “This finding suggests that more communication with parents regarding treatment and prognosis is needed,” they write in an article published online June 10 in JAMA Ophthalmology.
One of the most prevalent eye disorders in children, allergic conjunctivitis is often chronic, leading patients to ask repeatedly for help from physicians. It can take an emotional toll and can cause children to miss school.
“With any sign of a slightly pink eye [or a] runny nose, which are very common with allergies, children are being sent home, because everyone’s concerned about COVID,” said Yi Ning J. Strube, MD, an associate professor of ophthalmology and pediatrics at Queen’s University, Kingston, Canada, whose commentary appears in the same issue of JAMA Ophthalmology.
Adolescents are also sometimes accused of smoking cannabis because of their red eyes, she said.
However, little research has examined the effects of allergic conjunctivitis on the quality of life of children and their guardians, Dr. Zhang and colleagues write. To fill that gap, the researchers administered the Pediatric Quality of Life Inventory (PedsQL) to 92 children with allergic conjunctivitis and their parents. The children were aged 5 to 18 years.
The researchers administered the same questionnaire to 96 healthy children of the same ages, along with their parents. These participants served as a control group.
On a scale of 0 to 100, in which a higher score signifies a better quality of life, the median total PedsQL score was 69.6 for children with allergic conjunctivitis versus 96.7 for the control group.
Subscores of physical, emotional, social, and especially school functioning were all significantly lower for the children with allergic conjunctivitis than for the control persons. “Because children generally spend most of their time in the school environment, this outcome raises an issue regarding whether children have a poorer performance in their education,” Dr. Zhang and colleagues write.
Dr. Strube recommends that physicians educate their patients about allergic conjunctivitis using handouts or high-quality websites. She often refers patients and their families to the allergic conjunctivitis webpage of the American Academy of Pediatric Ophthalmology and Strabismus.
She tells parents to have their child “take a shower and wash their hair when they get home before they rub their pollen-filled hair on their pillowcase and make their allergy symptoms worse.”
Parents and schools should try to filter pollen and other allergens from indoor air, she added.
Parents of the children with allergic conjunctivitis in the study also reported lower quality of life; they scored 68.8, versus 96.5 for parents of children in the control group. The differences for both parents and children were statistically significant (P < .001). Overall, the parents’ quality-of-life scores correlated with their children’s (correlation coefficient, r = 0.59; P < .001).
Children with vernal or atopic keratoconjunctivitis scored 3.3 points lower on health-related quality of life than those with seasonal allergic conjunctivitis.
Children with higher corneal fluorescein staining scores also had lower quality-of-life scores. Parents whose children had higher corneal fluorescein staining scores and also those who had multiple consultations with health care practitioners also reported lower quality of life.
The quality-of-life scores of the children with allergic conjunctivitis were lower than scores in previous studies for children with vision-threatening diseases, such as glaucoma and congenital cataract. This may be because glaucoma and cataracts do not typically cause discomfort even if they impair the patient’s vision, said Dr. Strube.
She pointed out one potential flaw in the study: In the cohort with allergic conjunctivitis, 83.7% were boys, compared to 42.7% of the control group. Vernal keratoconjunctivitis affects more boys than girls, and not controlling for this factor could have confounded the data, Dr. Strube said.
It could also be useful to replicate the study in other countries to see whether geographic or cultural factors affected the results, she said. “A lot of these big centers around the world, including in China, have poor air quality, so that may be contributing to patients’ symptoms,” she said. “With regards to reported health quality of life and impact on education, results from different parts of the world may be different, due to parenting styles and education styles,” she said.
The study was supported by the National Natural Science Foundation of China and the Science Foundation of Guangdong Province. Dr. Zhang and colleagues reported no relevant financial relationships. Dr. Strube reported receiving personal fees from Santen Canada Advisory Board Consultant outside the submitted work.
A version of this article first appeared on Medscape.com.
Allergic conjunctivitis harms quality of life for children and their parents, apparently causing greater day-to-day worries than potentially blinding diseases, researchers report.
Parents worry especially that treatments might not be effective, according to Shi-yao Zhang, MD, and colleagues from Sun Yat-sen University, Guangzhou, China. “This finding suggests that more communication with parents regarding treatment and prognosis is needed,” they write in an article published online June 10 in JAMA Ophthalmology.
One of the most prevalent eye disorders in children, allergic conjunctivitis is often chronic, leading patients to ask repeatedly for help from physicians. It can take an emotional toll and can cause children to miss school.
“With any sign of a slightly pink eye [or a] runny nose, which are very common with allergies, children are being sent home, because everyone’s concerned about COVID,” said Yi Ning J. Strube, MD, an associate professor of ophthalmology and pediatrics at Queen’s University, Kingston, Canada, whose commentary appears in the same issue of JAMA Ophthalmology.
Adolescents are also sometimes accused of smoking cannabis because of their red eyes, she said.
However, little research has examined the effects of allergic conjunctivitis on the quality of life of children and their guardians, Dr. Zhang and colleagues write. To fill that gap, the researchers administered the Pediatric Quality of Life Inventory (PedsQL) to 92 children with allergic conjunctivitis and their parents. The children were aged 5 to 18 years.
The researchers administered the same questionnaire to 96 healthy children of the same ages, along with their parents. These participants served as a control group.
On a scale of 0 to 100, in which a higher score signifies a better quality of life, the median total PedsQL score was 69.6 for children with allergic conjunctivitis versus 96.7 for the control group.
Subscores of physical, emotional, social, and especially school functioning were all significantly lower for the children with allergic conjunctivitis than for the control persons. “Because children generally spend most of their time in the school environment, this outcome raises an issue regarding whether children have a poorer performance in their education,” Dr. Zhang and colleagues write.
Dr. Strube recommends that physicians educate their patients about allergic conjunctivitis using handouts or high-quality websites. She often refers patients and their families to the allergic conjunctivitis webpage of the American Academy of Pediatric Ophthalmology and Strabismus.
She tells parents to have their child “take a shower and wash their hair when they get home before they rub their pollen-filled hair on their pillowcase and make their allergy symptoms worse.”
Parents and schools should try to filter pollen and other allergens from indoor air, she added.
Parents of the children with allergic conjunctivitis in the study also reported lower quality of life; they scored 68.8, versus 96.5 for parents of children in the control group. The differences for both parents and children were statistically significant (P < .001). Overall, the parents’ quality-of-life scores correlated with their children’s (correlation coefficient, r = 0.59; P < .001).
Children with vernal or atopic keratoconjunctivitis scored 3.3 points lower on health-related quality of life than those with seasonal allergic conjunctivitis.
Children with higher corneal fluorescein staining scores also had lower quality-of-life scores. Parents whose children had higher corneal fluorescein staining scores and also those who had multiple consultations with health care practitioners also reported lower quality of life.
The quality-of-life scores of the children with allergic conjunctivitis were lower than scores in previous studies for children with vision-threatening diseases, such as glaucoma and congenital cataract. This may be because glaucoma and cataracts do not typically cause discomfort even if they impair the patient’s vision, said Dr. Strube.
She pointed out one potential flaw in the study: In the cohort with allergic conjunctivitis, 83.7% were boys, compared to 42.7% of the control group. Vernal keratoconjunctivitis affects more boys than girls, and not controlling for this factor could have confounded the data, Dr. Strube said.
It could also be useful to replicate the study in other countries to see whether geographic or cultural factors affected the results, she said. “A lot of these big centers around the world, including in China, have poor air quality, so that may be contributing to patients’ symptoms,” she said. “With regards to reported health quality of life and impact on education, results from different parts of the world may be different, due to parenting styles and education styles,” she said.
The study was supported by the National Natural Science Foundation of China and the Science Foundation of Guangdong Province. Dr. Zhang and colleagues reported no relevant financial relationships. Dr. Strube reported receiving personal fees from Santen Canada Advisory Board Consultant outside the submitted work.
A version of this article first appeared on Medscape.com.