User login
Americans’ sun protection practices fall short of intentions
commissioned by the American Academy of Dermatology.
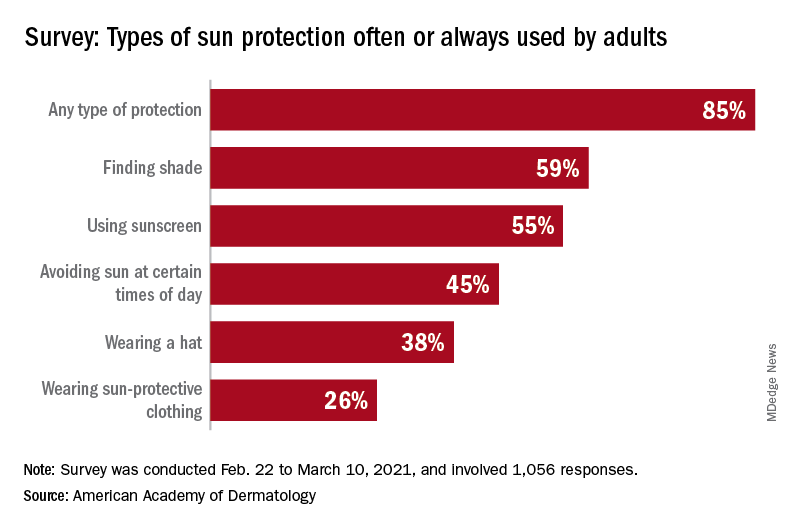
With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
commissioned by the American Academy of Dermatology.

With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
commissioned by the American Academy of Dermatology.

With the pandemic seemingly behind it, the United States enters the summer months facing the paradox of sun protection. Four out of five adults know that sunscreen should be reapplied every 2 hours when they’re outdoors, but only one in three make the actual effort, and 77% are likely to use sunscreen at the beach or a pool, compared with 41% when they’re gardening or working outside on their homes, the AAD reported.
“These findings are surprising and seem to suggest that many people do not take skin cancer seriously or perhaps believe skin cancer won’t happen to them,” Robert T. Brodell, MD, professor of dermatology at the University of Mississippi Medical Center, Jackson, said in a written statement from the AAD, adding that “unprotected exposure to ultraviolet rays is the most preventable risk factor for skin cancer, including melanoma.”
A quarter of all survey respondents reported getting sunburned in 2020, with the youngest adults most likely to feel the wrath of the sun. Sunburn was reported by 43% of those aged 18-23 years, 37% of those aged 24-39, 25% of the 40- to 55-year-olds, 12% of the 56- to 74-year-olds, and 7% of those aged 75 and older. More than a quarter of those who got sunburned said that it was bad enough to make their clothes feel uncomfortable, the academy said.
“Americans see the damaging effects of the sun on their skin as they get older, and two out of three look back and wish they had been more careful. But when it comes to cancer, specifically, most feel unconcerned in spite of their own risk,” according to a statement from Versta Research, which conducted the poll on behalf of the AAD. The survey was conducted from Feb. 22 to March 10, 2021, and involved 1,056 respondents, with a ±3% margin of error.
The lack of concern for skin cancer looks like this: More than two-thirds of the respondents (69%) have at least one possible risk factor – lighter skin tone, blue or green eyes, more than 50 moles, family history – but only 36% expressed concern about developing it. “Indeed, half of survey respondents (49%) say they are more worried about avoiding sunburn than they are about preventing skin cancer, and a third (32%) are more worried about avoiding premature wrinkles than they are about preventing cancer,” the AAD said.
The AAD is considering the creation of a social media quiz or interactive tool, and if the results of this survey were recast as a potential “Knowledge and Awareness Quiz” and graded with a traditional scheme (A = 90%-100%, B = 80%-89%, etc.), then 34% of the respondents would have failed, 15% would have gotten a D, and only 5% would have earned an A, the academy noted.
Common Comorbidities of Chronic Rhinosinusitis With Nasal Polyps
The majority of patients with chronic rhinosinusitis with nasal polyps (CRSwNP) have both upper and lower airway comorbidities such as asthma, allergic rhinosinusitis, and GERD. Recent data show that more than 50% of patients with CRSwNP have concurrent asthma.
Comorbidities in CRSwNP increase the risk for refractory disease, lead to repeated surgical procedures, and adversely affect patients' quality of life.
Dr Anju Peters, from Northwestern University Feinberg School of Medicine, discusses the importance of identifying comorbidities in patients with CRSwNP in order to create a more effective and personalized treatment plan.
---
Anju T. Peters, MD, MSci, Professor, Department of Medicine, Division of Allergy-Immunology, Northwestern University Feinberg School of Medicine; Director of Clinical Research, Associate Chief of Research, Education, and Clinical Affairs, Division of Allergy-Immunology, Northwestern Medicine, Chicago, Illinois.
Anju T. Peters, MD, MSci, has disclosed the following relevant financial relationships:
Serve(d) on the advisory board for: Sanofi Regeneron; Optinose; AstraZeneca. Received research grant from: AstraZeneca; Optinose.
The majority of patients with chronic rhinosinusitis with nasal polyps (CRSwNP) have both upper and lower airway comorbidities such as asthma, allergic rhinosinusitis, and GERD. Recent data show that more than 50% of patients with CRSwNP have concurrent asthma.
Comorbidities in CRSwNP increase the risk for refractory disease, lead to repeated surgical procedures, and adversely affect patients' quality of life.
Dr Anju Peters, from Northwestern University Feinberg School of Medicine, discusses the importance of identifying comorbidities in patients with CRSwNP in order to create a more effective and personalized treatment plan.
---
Anju T. Peters, MD, MSci, Professor, Department of Medicine, Division of Allergy-Immunology, Northwestern University Feinberg School of Medicine; Director of Clinical Research, Associate Chief of Research, Education, and Clinical Affairs, Division of Allergy-Immunology, Northwestern Medicine, Chicago, Illinois.
Anju T. Peters, MD, MSci, has disclosed the following relevant financial relationships:
Serve(d) on the advisory board for: Sanofi Regeneron; Optinose; AstraZeneca. Received research grant from: AstraZeneca; Optinose.
The majority of patients with chronic rhinosinusitis with nasal polyps (CRSwNP) have both upper and lower airway comorbidities such as asthma, allergic rhinosinusitis, and GERD. Recent data show that more than 50% of patients with CRSwNP have concurrent asthma.
Comorbidities in CRSwNP increase the risk for refractory disease, lead to repeated surgical procedures, and adversely affect patients' quality of life.
Dr Anju Peters, from Northwestern University Feinberg School of Medicine, discusses the importance of identifying comorbidities in patients with CRSwNP in order to create a more effective and personalized treatment plan.
---
Anju T. Peters, MD, MSci, Professor, Department of Medicine, Division of Allergy-Immunology, Northwestern University Feinberg School of Medicine; Director of Clinical Research, Associate Chief of Research, Education, and Clinical Affairs, Division of Allergy-Immunology, Northwestern Medicine, Chicago, Illinois.
Anju T. Peters, MD, MSci, has disclosed the following relevant financial relationships:
Serve(d) on the advisory board for: Sanofi Regeneron; Optinose; AstraZeneca. Received research grant from: AstraZeneca; Optinose.

AGA, GI societies support lowering CRC screening age
American Gastroenterological Association, American College of Gastroenterology, and American Society for Gastrointestinal Endoscopy issued a statement of support that also notes our Multi-Society Task Force on Colorectal Cancer is finalizing our own recommendation to start screening at 45 years of age as well.
Incoming AGA President John M. Inadomi, MD, AGAF, notes that, “We expect this important change to save lives and improve the health of the U.S. population.”
AGA fully supports the decision of the U.S. Preventive Services Task Force to reduce the age at which to initiate screening among individuals at average risk for development of colorectal cancer to 45 years. This decision harmonizes the recommendations between the major U.S. screening guidelines including the American Cancer Society and American College of Physicians.
“The analysis by the USPSTF is timely and incredibly helpful to population health and to gastroenterologists and other providers,” says Bishr Omary, MD, PhD, AGAF, president of AGA. “We now have clear guidance to start colorectal cancer screening at age 45 for those with average risk and discontinue screening after age 85.”
American Gastroenterological Association, American College of Gastroenterology, and American Society for Gastrointestinal Endoscopy issued a statement of support that also notes our Multi-Society Task Force on Colorectal Cancer is finalizing our own recommendation to start screening at 45 years of age as well.
Incoming AGA President John M. Inadomi, MD, AGAF, notes that, “We expect this important change to save lives and improve the health of the U.S. population.”
AGA fully supports the decision of the U.S. Preventive Services Task Force to reduce the age at which to initiate screening among individuals at average risk for development of colorectal cancer to 45 years. This decision harmonizes the recommendations between the major U.S. screening guidelines including the American Cancer Society and American College of Physicians.
“The analysis by the USPSTF is timely and incredibly helpful to population health and to gastroenterologists and other providers,” says Bishr Omary, MD, PhD, AGAF, president of AGA. “We now have clear guidance to start colorectal cancer screening at age 45 for those with average risk and discontinue screening after age 85.”
American Gastroenterological Association, American College of Gastroenterology, and American Society for Gastrointestinal Endoscopy issued a statement of support that also notes our Multi-Society Task Force on Colorectal Cancer is finalizing our own recommendation to start screening at 45 years of age as well.
Incoming AGA President John M. Inadomi, MD, AGAF, notes that, “We expect this important change to save lives and improve the health of the U.S. population.”
AGA fully supports the decision of the U.S. Preventive Services Task Force to reduce the age at which to initiate screening among individuals at average risk for development of colorectal cancer to 45 years. This decision harmonizes the recommendations between the major U.S. screening guidelines including the American Cancer Society and American College of Physicians.
“The analysis by the USPSTF is timely and incredibly helpful to population health and to gastroenterologists and other providers,” says Bishr Omary, MD, PhD, AGAF, president of AGA. “We now have clear guidance to start colorectal cancer screening at age 45 for those with average risk and discontinue screening after age 85.”
The 2021-2022 research awards cycle is now open
We are pleased to announce that the AGA Research Foundation’s research awards cycle is now open.
The cycle begins with our two specialty awards focused on digestive and gastric cancers – applications are due on July 21.
AGA–Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer: One $40,000 award supports an early career investigator who holds a career development award devoted to digestive cancer research.
AGA–R. Robert & Sally Funderburg Research Award in Gastric Cancer One $100,000 award supports an established investigator working on novel approaches in gastric cancer research.
In addition to our usual awards portfolio focused on a broad range of digestive diseases, we have established several new awards that will fund research focused on health and health care disparities. Click on the links below to learn more about each award and application requirements.
- Pilot Research Awards: Currently accepting applications
- Research Scholar Awards: Open Aug. 12
We are pleased to announce that the AGA Research Foundation’s research awards cycle is now open.
The cycle begins with our two specialty awards focused on digestive and gastric cancers – applications are due on July 21.
AGA–Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer: One $40,000 award supports an early career investigator who holds a career development award devoted to digestive cancer research.
AGA–R. Robert & Sally Funderburg Research Award in Gastric Cancer One $100,000 award supports an established investigator working on novel approaches in gastric cancer research.
In addition to our usual awards portfolio focused on a broad range of digestive diseases, we have established several new awards that will fund research focused on health and health care disparities. Click on the links below to learn more about each award and application requirements.
- Pilot Research Awards: Currently accepting applications
- Research Scholar Awards: Open Aug. 12
We are pleased to announce that the AGA Research Foundation’s research awards cycle is now open.
The cycle begins with our two specialty awards focused on digestive and gastric cancers – applications are due on July 21.
AGA–Caroline Craig Augustyn & Damian Augustyn Award in Digestive Cancer: One $40,000 award supports an early career investigator who holds a career development award devoted to digestive cancer research.
AGA–R. Robert & Sally Funderburg Research Award in Gastric Cancer One $100,000 award supports an established investigator working on novel approaches in gastric cancer research.
In addition to our usual awards portfolio focused on a broad range of digestive diseases, we have established several new awards that will fund research focused on health and health care disparities. Click on the links below to learn more about each award and application requirements.
- Pilot Research Awards: Currently accepting applications
- Research Scholar Awards: Open Aug. 12
The gift you should be talking about
If you want to make a lasting impact at the AGA Research Foundation, one of the easiest ways is to name us as a beneficiary of one of your assets, such as your retirement plan, life insurance policy, bank account, or donor-advised fund.
When you do, don’t forget to notify us of your decisions. Many charities and individuals aren’t aware that they have been named to receive a gift. Informing them helps preserve your intentions and ensures that your beneficiaries are able to follow your wishes.
Steps to protect the people and charities you love
- Review your beneficiary designations periodically because circumstances change throughout your lifetime.
- Alert your beneficiaries that you have a life insurance policy or have named them as beneficiaries of a retirement plan.
- Share the location and details of the policy or plan with your beneficiaries.
As you update your beneficiary designations, consider making a gift of a life insurance policy or retirement plan to the AGA Research Foundation so that we can continue to progress with our mission. Then let us know about your decision so that we can carry out your wishes as intended and thank you for your gift.
We want to hear from you
If you have already named the AGA Research Foundation as a beneficiary of a life insurance policy or retirement plan assets, please contact us at [email protected] today. If you are still creating your estate plan, we would be happy to answer any questions you may have about making this type of gift.
If you want to make a lasting impact at the AGA Research Foundation, one of the easiest ways is to name us as a beneficiary of one of your assets, such as your retirement plan, life insurance policy, bank account, or donor-advised fund.
When you do, don’t forget to notify us of your decisions. Many charities and individuals aren’t aware that they have been named to receive a gift. Informing them helps preserve your intentions and ensures that your beneficiaries are able to follow your wishes.
Steps to protect the people and charities you love
- Review your beneficiary designations periodically because circumstances change throughout your lifetime.
- Alert your beneficiaries that you have a life insurance policy or have named them as beneficiaries of a retirement plan.
- Share the location and details of the policy or plan with your beneficiaries.
As you update your beneficiary designations, consider making a gift of a life insurance policy or retirement plan to the AGA Research Foundation so that we can continue to progress with our mission. Then let us know about your decision so that we can carry out your wishes as intended and thank you for your gift.
We want to hear from you
If you have already named the AGA Research Foundation as a beneficiary of a life insurance policy or retirement plan assets, please contact us at [email protected] today. If you are still creating your estate plan, we would be happy to answer any questions you may have about making this type of gift.
If you want to make a lasting impact at the AGA Research Foundation, one of the easiest ways is to name us as a beneficiary of one of your assets, such as your retirement plan, life insurance policy, bank account, or donor-advised fund.
When you do, don’t forget to notify us of your decisions. Many charities and individuals aren’t aware that they have been named to receive a gift. Informing them helps preserve your intentions and ensures that your beneficiaries are able to follow your wishes.
Steps to protect the people and charities you love
- Review your beneficiary designations periodically because circumstances change throughout your lifetime.
- Alert your beneficiaries that you have a life insurance policy or have named them as beneficiaries of a retirement plan.
- Share the location and details of the policy or plan with your beneficiaries.
As you update your beneficiary designations, consider making a gift of a life insurance policy or retirement plan to the AGA Research Foundation so that we can continue to progress with our mission. Then let us know about your decision so that we can carry out your wishes as intended and thank you for your gift.
We want to hear from you
If you have already named the AGA Research Foundation as a beneficiary of a life insurance policy or retirement plan assets, please contact us at [email protected] today. If you are still creating your estate plan, we would be happy to answer any questions you may have about making this type of gift.
Top cases
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community (https://community.gastro.org) to seek advice from colleagues about therapy and disease management options, best practices, and diagnoses. Here’s a preview of a recent popular clinical discussion:
From Rafael Ching Companioni, MD: Malnutrition, elevated liver enzymes, anemia, and malabsorption
“Early 30 year-old female who was initially referred to GI in December 2020 for abnormal liver enzymes ALT 263, AST 114, alk phosp 212, albumin 3.2, bili [within normal limits]. At that time, she reports some diarrhea, few episodes of diarrhea per day, diffuse abdominal pain, ~20 LBs weight loss. She denied herbal medications, OTC medications or other medications. Last travel was 2 years ago to England. No history of anorexia nervosa or bulimia. On examination, cachexia and extremity edema. She has iron deficiency anemia and reactive thrombocytosis. Her initial lipid panel in November 2020, the lipid panel shows total cholesterol 208, LDL 113, triglycerides 227.
“She is still losing weight: 20 lbs from Feb 2021. The liver enzymes elevation resolved. She has anemia, malnutrition and malabsorption. I recommended gluten free diet, MVI, iron pills, protein bars. I had ordered scleroderma workup and SIBO tests today. I am planning to do MRE.”
See how AGA members responded and join the discussion: https://community.gastro.org/posts/24416.
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community (https://community.gastro.org) to seek advice from colleagues about therapy and disease management options, best practices, and diagnoses. Here’s a preview of a recent popular clinical discussion:
From Rafael Ching Companioni, MD: Malnutrition, elevated liver enzymes, anemia, and malabsorption
“Early 30 year-old female who was initially referred to GI in December 2020 for abnormal liver enzymes ALT 263, AST 114, alk phosp 212, albumin 3.2, bili [within normal limits]. At that time, she reports some diarrhea, few episodes of diarrhea per day, diffuse abdominal pain, ~20 LBs weight loss. She denied herbal medications, OTC medications or other medications. Last travel was 2 years ago to England. No history of anorexia nervosa or bulimia. On examination, cachexia and extremity edema. She has iron deficiency anemia and reactive thrombocytosis. Her initial lipid panel in November 2020, the lipid panel shows total cholesterol 208, LDL 113, triglycerides 227.
“She is still losing weight: 20 lbs from Feb 2021. The liver enzymes elevation resolved. She has anemia, malnutrition and malabsorption. I recommended gluten free diet, MVI, iron pills, protein bars. I had ordered scleroderma workup and SIBO tests today. I am planning to do MRE.”
See how AGA members responded and join the discussion: https://community.gastro.org/posts/24416.
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community (https://community.gastro.org) to seek advice from colleagues about therapy and disease management options, best practices, and diagnoses. Here’s a preview of a recent popular clinical discussion:
From Rafael Ching Companioni, MD: Malnutrition, elevated liver enzymes, anemia, and malabsorption
“Early 30 year-old female who was initially referred to GI in December 2020 for abnormal liver enzymes ALT 263, AST 114, alk phosp 212, albumin 3.2, bili [within normal limits]. At that time, she reports some diarrhea, few episodes of diarrhea per day, diffuse abdominal pain, ~20 LBs weight loss. She denied herbal medications, OTC medications or other medications. Last travel was 2 years ago to England. No history of anorexia nervosa or bulimia. On examination, cachexia and extremity edema. She has iron deficiency anemia and reactive thrombocytosis. Her initial lipid panel in November 2020, the lipid panel shows total cholesterol 208, LDL 113, triglycerides 227.
“She is still losing weight: 20 lbs from Feb 2021. The liver enzymes elevation resolved. She has anemia, malnutrition and malabsorption. I recommended gluten free diet, MVI, iron pills, protein bars. I had ordered scleroderma workup and SIBO tests today. I am planning to do MRE.”
See how AGA members responded and join the discussion: https://community.gastro.org/posts/24416.
Lessons from COVID-19 and planning for a postpandemic screening surge
It is not an exaggeration to say that everything in my gastroenterology practice changed in response to COVID-19.
Due to the overwhelming surge that Massachusetts saw in the early days of the pandemic, the Department of Public Health issued a moratorium on elective procedures in mid-March of 2020, for both hospitals and ambulatory surgery centers. The moratorium included colorectal cancer (CRC) screenings and other procedures that make up a significant portion of the services we provide to our community. Greater Boston Gastroenterology treats patients in and around the area of Framingham, Mass. – not too far outside of Boston. In our practice, we have seven physicians and three nurse practitioners, with one main office and two satellite offices. By national standards, our practice would be considered small, but it is on the larger side of independent GI physician practices in the commonwealth.
Nationally, moratoria on elective procedures led to one of the steepest drop-offs in screenings for cancers, including colorectal cancer. In late summer of 2020, it was estimated that CRC screenings dropped by 86 percent. Two-thirds of independent GI practices saw a significant decline in patient volume, and many believe that they may not get it back.
However, I’m an optimist in this situation, and I believe that as life gets more normal, people will get back to screenings. With the recommendation by the U.S. Preventative Services Task Force that CRC screening should begin at age 45, I expect that there will be an additional increase in screening soon.
Pivoting and developing a reopening plan
Almost immediately after the Department of Public Health issued the moratorium, Greater Boston Gastroenterology began putting together a reopening plan that would allow us to continue treating some patients and prepare for a surge once restrictions were lifted.
Part of our plan was to stay informed by talking with other practices about what they were doing and to stay abreast of policy changes at the local, state, and federal levels.
We also needed to keep our patients informed to alleviate safety concerns. Just prior to our reopening, we developed videos of the precautions that we were taking in all our facilities to assure our patients that we were doing everything possible to keep them safe. We also put information on our website through every stage of reopening so patients could know what to expect at their visits.
Helping our staff feel safe as they returned to work was also an important focus of our reopening plan. We prepared for our eventual reopening by installing safety measures such as plexiglass barriers and HEPA filter machines for our common areas and exam rooms. We also procured access to rapid turnaround polymerase chain reaction (PCR) testing that allowed us to regularly test all patients seeking elective procedures. Additionally, we invested in point-of-care antigen tests for the office, and we regularly test all our patient-facing staff.
We had corralled enough personal protective equipment to keep our office infusion services operating with our nurses and patients feeling safe. The preparation allowed us to resume in-person visits almost immediately after the Department of Public Health allowed us to reopen.
Once we reopened, we concentrated on in-office visits for patients who were under 65 and at lower risk for COVID-19, while focusing our telemedicine efforts on patients who were older and at higher risk. We’re now back to seeing all patients who want to have in-office visits and are actually above par for our visits. The number of procedures we have performed in the last 3 months is similar to the 3 months before the pandemic.
During the pandemic, Massachusetts had the best conversion to telehealth in the nation, and it worked well for patients and providers. The key was to use several telehealth apps, as using only one may not work for everyone. Having several options made it likely that we would be able to do complete visits and connect with patients. When we needed to, we switched to telephone visits.
All the physicians and staff in our practice are telemedicine enthusiasts, and it will remain a significant part of our practices as long as Medicare, the state health plans, and commercial payers remain supportive.
Planning for a surge in screenings
There may be a surge in screenings once more people are vaccinated and comfortable getting back into the office, and we’re planning for this as well. We’ve recruited new physicians and have expanded our available hours for procedures at our ambulatory surgery centers (ASCs). Surprisingly, we have found that there is a lot of interest from physicians for weekend shifts at the ASC, and we now have a physician waiting list for Saturday procedure time.
With the new lower age for recommended screening, there will be a lag with primary care physicians referring their younger patients. This may provide some time to prepare for an increase in screenings resulting from this new policy.
Another strategy that has worked well for us is to train and develop our advanced practitioners into nonphysician experts in GI and liver disease. Greater Boston Gastroenterology has used this strategy since its founding, and we think our most experienced nurse practitioners could rival any office-based gastroenterologist in their acumen and capabilities.
Over the last 3 years we have transitioned our nonphysician practitioners into the inpatient setting. As a result, consults are completed earlier in the day, and we are better able to help coordinate inpatient procedure scheduling, discharge planning, and outpatient follow-up.
The time we spend on training is worth it. It improves customer service, allows us to book appointments with shorter notice, and overall has a positive effect on our bottom line. Utilizing our advanced providers in this capacity will help us manage any volume increases we see in the near future. In addition, most patients in our community are used to seeing advanced providers in their physician’s office, so the acceptance among our patients is high.
Being flexible and favoring strategic planning
Overall, I think the greatest thing we learned during the pandemic is that we need to be flexible. It was a helpful reminder that, in medicine, things are constantly changing. I remember when passing the GI boards seemed like my final step, but everyone comes to realize it is just the first step in the journey.
As an early-career physician, you should remember the hard work that helped you get to medical school, land a good residency, stand out to get a fellowship, and master your specialty. Harness that personal drive and energy and keep moving forward. Remember that your first job is unlikely to be your last. Try not to see your choices as either/or – either academic or private practice, hospital-employed or self-employed. The boundaries are blurring. We have long careers and face myriad opportunities for professional advancement.
Be patient. Some goals take time to achieve. At each stage be prepared to work hard, use your time wisely, and try not to lose sight of maximizing your professional happiness.
Dr. Dickstein is a practicing gastroenterologist at Greater Boston Gastroenterology in Framingham, Mass., and serves on the executive committee of the Digestive Health Physicians Association. He has no conflicts to declare.
It is not an exaggeration to say that everything in my gastroenterology practice changed in response to COVID-19.
Due to the overwhelming surge that Massachusetts saw in the early days of the pandemic, the Department of Public Health issued a moratorium on elective procedures in mid-March of 2020, for both hospitals and ambulatory surgery centers. The moratorium included colorectal cancer (CRC) screenings and other procedures that make up a significant portion of the services we provide to our community. Greater Boston Gastroenterology treats patients in and around the area of Framingham, Mass. – not too far outside of Boston. In our practice, we have seven physicians and three nurse practitioners, with one main office and two satellite offices. By national standards, our practice would be considered small, but it is on the larger side of independent GI physician practices in the commonwealth.
Nationally, moratoria on elective procedures led to one of the steepest drop-offs in screenings for cancers, including colorectal cancer. In late summer of 2020, it was estimated that CRC screenings dropped by 86 percent. Two-thirds of independent GI practices saw a significant decline in patient volume, and many believe that they may not get it back.
However, I’m an optimist in this situation, and I believe that as life gets more normal, people will get back to screenings. With the recommendation by the U.S. Preventative Services Task Force that CRC screening should begin at age 45, I expect that there will be an additional increase in screening soon.
Pivoting and developing a reopening plan
Almost immediately after the Department of Public Health issued the moratorium, Greater Boston Gastroenterology began putting together a reopening plan that would allow us to continue treating some patients and prepare for a surge once restrictions were lifted.
Part of our plan was to stay informed by talking with other practices about what they were doing and to stay abreast of policy changes at the local, state, and federal levels.
We also needed to keep our patients informed to alleviate safety concerns. Just prior to our reopening, we developed videos of the precautions that we were taking in all our facilities to assure our patients that we were doing everything possible to keep them safe. We also put information on our website through every stage of reopening so patients could know what to expect at their visits.
Helping our staff feel safe as they returned to work was also an important focus of our reopening plan. We prepared for our eventual reopening by installing safety measures such as plexiglass barriers and HEPA filter machines for our common areas and exam rooms. We also procured access to rapid turnaround polymerase chain reaction (PCR) testing that allowed us to regularly test all patients seeking elective procedures. Additionally, we invested in point-of-care antigen tests for the office, and we regularly test all our patient-facing staff.
We had corralled enough personal protective equipment to keep our office infusion services operating with our nurses and patients feeling safe. The preparation allowed us to resume in-person visits almost immediately after the Department of Public Health allowed us to reopen.
Once we reopened, we concentrated on in-office visits for patients who were under 65 and at lower risk for COVID-19, while focusing our telemedicine efforts on patients who were older and at higher risk. We’re now back to seeing all patients who want to have in-office visits and are actually above par for our visits. The number of procedures we have performed in the last 3 months is similar to the 3 months before the pandemic.
During the pandemic, Massachusetts had the best conversion to telehealth in the nation, and it worked well for patients and providers. The key was to use several telehealth apps, as using only one may not work for everyone. Having several options made it likely that we would be able to do complete visits and connect with patients. When we needed to, we switched to telephone visits.
All the physicians and staff in our practice are telemedicine enthusiasts, and it will remain a significant part of our practices as long as Medicare, the state health plans, and commercial payers remain supportive.
Planning for a surge in screenings
There may be a surge in screenings once more people are vaccinated and comfortable getting back into the office, and we’re planning for this as well. We’ve recruited new physicians and have expanded our available hours for procedures at our ambulatory surgery centers (ASCs). Surprisingly, we have found that there is a lot of interest from physicians for weekend shifts at the ASC, and we now have a physician waiting list for Saturday procedure time.
With the new lower age for recommended screening, there will be a lag with primary care physicians referring their younger patients. This may provide some time to prepare for an increase in screenings resulting from this new policy.
Another strategy that has worked well for us is to train and develop our advanced practitioners into nonphysician experts in GI and liver disease. Greater Boston Gastroenterology has used this strategy since its founding, and we think our most experienced nurse practitioners could rival any office-based gastroenterologist in their acumen and capabilities.
Over the last 3 years we have transitioned our nonphysician practitioners into the inpatient setting. As a result, consults are completed earlier in the day, and we are better able to help coordinate inpatient procedure scheduling, discharge planning, and outpatient follow-up.
The time we spend on training is worth it. It improves customer service, allows us to book appointments with shorter notice, and overall has a positive effect on our bottom line. Utilizing our advanced providers in this capacity will help us manage any volume increases we see in the near future. In addition, most patients in our community are used to seeing advanced providers in their physician’s office, so the acceptance among our patients is high.
Being flexible and favoring strategic planning
Overall, I think the greatest thing we learned during the pandemic is that we need to be flexible. It was a helpful reminder that, in medicine, things are constantly changing. I remember when passing the GI boards seemed like my final step, but everyone comes to realize it is just the first step in the journey.
As an early-career physician, you should remember the hard work that helped you get to medical school, land a good residency, stand out to get a fellowship, and master your specialty. Harness that personal drive and energy and keep moving forward. Remember that your first job is unlikely to be your last. Try not to see your choices as either/or – either academic or private practice, hospital-employed or self-employed. The boundaries are blurring. We have long careers and face myriad opportunities for professional advancement.
Be patient. Some goals take time to achieve. At each stage be prepared to work hard, use your time wisely, and try not to lose sight of maximizing your professional happiness.
Dr. Dickstein is a practicing gastroenterologist at Greater Boston Gastroenterology in Framingham, Mass., and serves on the executive committee of the Digestive Health Physicians Association. He has no conflicts to declare.
It is not an exaggeration to say that everything in my gastroenterology practice changed in response to COVID-19.
Due to the overwhelming surge that Massachusetts saw in the early days of the pandemic, the Department of Public Health issued a moratorium on elective procedures in mid-March of 2020, for both hospitals and ambulatory surgery centers. The moratorium included colorectal cancer (CRC) screenings and other procedures that make up a significant portion of the services we provide to our community. Greater Boston Gastroenterology treats patients in and around the area of Framingham, Mass. – not too far outside of Boston. In our practice, we have seven physicians and three nurse practitioners, with one main office and two satellite offices. By national standards, our practice would be considered small, but it is on the larger side of independent GI physician practices in the commonwealth.
Nationally, moratoria on elective procedures led to one of the steepest drop-offs in screenings for cancers, including colorectal cancer. In late summer of 2020, it was estimated that CRC screenings dropped by 86 percent. Two-thirds of independent GI practices saw a significant decline in patient volume, and many believe that they may not get it back.
However, I’m an optimist in this situation, and I believe that as life gets more normal, people will get back to screenings. With the recommendation by the U.S. Preventative Services Task Force that CRC screening should begin at age 45, I expect that there will be an additional increase in screening soon.
Pivoting and developing a reopening plan
Almost immediately after the Department of Public Health issued the moratorium, Greater Boston Gastroenterology began putting together a reopening plan that would allow us to continue treating some patients and prepare for a surge once restrictions were lifted.
Part of our plan was to stay informed by talking with other practices about what they were doing and to stay abreast of policy changes at the local, state, and federal levels.
We also needed to keep our patients informed to alleviate safety concerns. Just prior to our reopening, we developed videos of the precautions that we were taking in all our facilities to assure our patients that we were doing everything possible to keep them safe. We also put information on our website through every stage of reopening so patients could know what to expect at their visits.
Helping our staff feel safe as they returned to work was also an important focus of our reopening plan. We prepared for our eventual reopening by installing safety measures such as plexiglass barriers and HEPA filter machines for our common areas and exam rooms. We also procured access to rapid turnaround polymerase chain reaction (PCR) testing that allowed us to regularly test all patients seeking elective procedures. Additionally, we invested in point-of-care antigen tests for the office, and we regularly test all our patient-facing staff.
We had corralled enough personal protective equipment to keep our office infusion services operating with our nurses and patients feeling safe. The preparation allowed us to resume in-person visits almost immediately after the Department of Public Health allowed us to reopen.
Once we reopened, we concentrated on in-office visits for patients who were under 65 and at lower risk for COVID-19, while focusing our telemedicine efforts on patients who were older and at higher risk. We’re now back to seeing all patients who want to have in-office visits and are actually above par for our visits. The number of procedures we have performed in the last 3 months is similar to the 3 months before the pandemic.
During the pandemic, Massachusetts had the best conversion to telehealth in the nation, and it worked well for patients and providers. The key was to use several telehealth apps, as using only one may not work for everyone. Having several options made it likely that we would be able to do complete visits and connect with patients. When we needed to, we switched to telephone visits.
All the physicians and staff in our practice are telemedicine enthusiasts, and it will remain a significant part of our practices as long as Medicare, the state health plans, and commercial payers remain supportive.
Planning for a surge in screenings
There may be a surge in screenings once more people are vaccinated and comfortable getting back into the office, and we’re planning for this as well. We’ve recruited new physicians and have expanded our available hours for procedures at our ambulatory surgery centers (ASCs). Surprisingly, we have found that there is a lot of interest from physicians for weekend shifts at the ASC, and we now have a physician waiting list for Saturday procedure time.
With the new lower age for recommended screening, there will be a lag with primary care physicians referring their younger patients. This may provide some time to prepare for an increase in screenings resulting from this new policy.
Another strategy that has worked well for us is to train and develop our advanced practitioners into nonphysician experts in GI and liver disease. Greater Boston Gastroenterology has used this strategy since its founding, and we think our most experienced nurse practitioners could rival any office-based gastroenterologist in their acumen and capabilities.
Over the last 3 years we have transitioned our nonphysician practitioners into the inpatient setting. As a result, consults are completed earlier in the day, and we are better able to help coordinate inpatient procedure scheduling, discharge planning, and outpatient follow-up.
The time we spend on training is worth it. It improves customer service, allows us to book appointments with shorter notice, and overall has a positive effect on our bottom line. Utilizing our advanced providers in this capacity will help us manage any volume increases we see in the near future. In addition, most patients in our community are used to seeing advanced providers in their physician’s office, so the acceptance among our patients is high.
Being flexible and favoring strategic planning
Overall, I think the greatest thing we learned during the pandemic is that we need to be flexible. It was a helpful reminder that, in medicine, things are constantly changing. I remember when passing the GI boards seemed like my final step, but everyone comes to realize it is just the first step in the journey.
As an early-career physician, you should remember the hard work that helped you get to medical school, land a good residency, stand out to get a fellowship, and master your specialty. Harness that personal drive and energy and keep moving forward. Remember that your first job is unlikely to be your last. Try not to see your choices as either/or – either academic or private practice, hospital-employed or self-employed. The boundaries are blurring. We have long careers and face myriad opportunities for professional advancement.
Be patient. Some goals take time to achieve. At each stage be prepared to work hard, use your time wisely, and try not to lose sight of maximizing your professional happiness.
Dr. Dickstein is a practicing gastroenterologist at Greater Boston Gastroenterology in Framingham, Mass., and serves on the executive committee of the Digestive Health Physicians Association. He has no conflicts to declare.
Telemedicine is poised to drive new models of care
Telemedicine has been proposed as a solution for an array of health care access problems over decades of gradual growth. The vast ramping up of , according to an update at the annual health policy and advocacy conference sponsored by the American College of Chest Physicians.
“The cat is out of the bag,” said Jaspal Singh, MD, professor of medicine, Atrium Health, Charlotte, N.C. Due to changes in access and reimbursement to telemedicine driven by the pandemic, he said, “we now have permission to explore new models of care.”
Prior to February 2020, telemedicine was crawling forward at a leisurely pace, according to Dr. Singh. After March 2020, it broke into a run due to enormous demand and was met by a rapid response from the U.S. Congress. The first of four legislative bills that directly or indirectly supported telemedicine was passed on March 6, 2020.
The Centers for Medicare and Medicaid Services responded in kind, making modifications in a number of rules that removed obstacles to telehealth. One modification on April 6, 2020, for example, removed the requirement for a preexisting relationship between the clinician and patient, Dr. Singh said. The CMS also subsequently modified reimbursement policies in order to make telemedicine more tenable for physicians.
Given the risk of contagion from face-to-face encounters, telemedicine in the early days of the pandemic was not just attractive but the only practical and safe approach to medical care in many circumstances. Physicians and patients were anxious for health care that did not require in-office visits even though many critical issues for telemedicine, including its relative effectiveness, had not yet been fully evaluated.
Much has been learned regarding the feasibility and acceptability of telemedicine during the pandemic, but Dr. Singh noted that quality of care relative to in-person visits remains weakly supported for most indications. Indeed, he outlined sizable list of incompletely resolved issues, including optimal payment models, management of privacy concerns, and how to balance advantages to disadvantages.
For patients and physicians, the strengths of telemedicine include greater convenience made possible by the elimination of travel and waiting rooms. For the health care system, it can include less infrastructure and overhead. For many physicians, telemedicine might be perceived as more efficient.
On the other hand, some patients might feel that a clinical encounter is incomplete without a physical examination even when the physician does not feel the physical examination is needed, according to Dr. Singh. He cited a survey suggesting nearly half of patients expressed concern about a lack of connection to health care providers following a virtual visit.
In the same 2020 National Poll on Healthy Aging 2020 survey conducted by the University of Michigan 67% of respondents reported that the quality of care was not as good as that provided by in-patient visits, and 24% expressed concern about privacy. However, at the time the poll was taken in May 2020, experience with telemedicine among many of the respondents may have been limited. As telemedicine is integrated into routine care, perceptions might change as experience increases.
A distinction between telemedicine in routine care and telemedicine as a strategy to respond to a pandemic is important, Dr. Singh indicated. Dr. Singh was the lead author for a position paper on telemedicine for the diagnosis and treatment of sleep disorders from the American Academy of Sleep Medicine 5 years ago, but he acknowledged that models of care might differ when responding to abnormal surges in health care demand.
The surge in demand for COVID-19–related care engendered numerous innovative solutions. As examples, Dr. Singh recounted how a virtual hospital was created at his own institution. In a published study, 1,477 patients diagnosed with COVID-19 over a 6-week period remained at home and received care in a virtual observation unit (VCU) or a virtual acute care unit (VACU) . Only a small percentage required eventual hospital admission. In the VACU, patients were able to receive advanced care including IV fluids and some form of respiratory support .
It is unclear how the COVID-19 pandemic will change telemedicine. Now, with declining cases of the infection, telemedicine is back to a walk after the sprint required during the height of the pandemic, according to Dr. Singh. However, Dr. Singh thinks many physicians and patients will have a different perception of telemedicine after the widespread exposure to this type of care.
In terms of the relative role of in-patient and virtual visits across indications, “we do not know how this will play out, but we will probably end up toggling between the two,” Dr. Singh said.
This is an area that is being followed closely by the CHEST Health Policy and Advocacy Committee, according to Kathleen Sarmiento, MD, director, VISN 21 Sleep Clinical Resource Hub for the San Francisco VA Health Care System. A member of that committee and moderator of the session in which Dr. Singh spoke,
Dr. Sarmiento called the effort to bring permanent coverage of telehealth services “the shared responsibility of every medical society engaged in advocacy.”
However, she cautioned that there might be intended and unintended consequences from telehealth that require analysis to develop policies that are in the best interests of effective care. She said, the “ACCP, along with its sister societies, does have a role in supporting the evaluation of the impact of these changes on both patients and providers in the fields of pulmonary medicine, critical care, and sleep medicine.”
Dr. Singh reports a financial relationship with AstraZeneca. Dr. Sarmiento reports no relevant financial relationship with AstraZeneca.
Telemedicine has been proposed as a solution for an array of health care access problems over decades of gradual growth. The vast ramping up of , according to an update at the annual health policy and advocacy conference sponsored by the American College of Chest Physicians.
“The cat is out of the bag,” said Jaspal Singh, MD, professor of medicine, Atrium Health, Charlotte, N.C. Due to changes in access and reimbursement to telemedicine driven by the pandemic, he said, “we now have permission to explore new models of care.”
Prior to February 2020, telemedicine was crawling forward at a leisurely pace, according to Dr. Singh. After March 2020, it broke into a run due to enormous demand and was met by a rapid response from the U.S. Congress. The first of four legislative bills that directly or indirectly supported telemedicine was passed on March 6, 2020.
The Centers for Medicare and Medicaid Services responded in kind, making modifications in a number of rules that removed obstacles to telehealth. One modification on April 6, 2020, for example, removed the requirement for a preexisting relationship between the clinician and patient, Dr. Singh said. The CMS also subsequently modified reimbursement policies in order to make telemedicine more tenable for physicians.
Given the risk of contagion from face-to-face encounters, telemedicine in the early days of the pandemic was not just attractive but the only practical and safe approach to medical care in many circumstances. Physicians and patients were anxious for health care that did not require in-office visits even though many critical issues for telemedicine, including its relative effectiveness, had not yet been fully evaluated.
Much has been learned regarding the feasibility and acceptability of telemedicine during the pandemic, but Dr. Singh noted that quality of care relative to in-person visits remains weakly supported for most indications. Indeed, he outlined sizable list of incompletely resolved issues, including optimal payment models, management of privacy concerns, and how to balance advantages to disadvantages.
For patients and physicians, the strengths of telemedicine include greater convenience made possible by the elimination of travel and waiting rooms. For the health care system, it can include less infrastructure and overhead. For many physicians, telemedicine might be perceived as more efficient.
On the other hand, some patients might feel that a clinical encounter is incomplete without a physical examination even when the physician does not feel the physical examination is needed, according to Dr. Singh. He cited a survey suggesting nearly half of patients expressed concern about a lack of connection to health care providers following a virtual visit.
In the same 2020 National Poll on Healthy Aging 2020 survey conducted by the University of Michigan 67% of respondents reported that the quality of care was not as good as that provided by in-patient visits, and 24% expressed concern about privacy. However, at the time the poll was taken in May 2020, experience with telemedicine among many of the respondents may have been limited. As telemedicine is integrated into routine care, perceptions might change as experience increases.
A distinction between telemedicine in routine care and telemedicine as a strategy to respond to a pandemic is important, Dr. Singh indicated. Dr. Singh was the lead author for a position paper on telemedicine for the diagnosis and treatment of sleep disorders from the American Academy of Sleep Medicine 5 years ago, but he acknowledged that models of care might differ when responding to abnormal surges in health care demand.
The surge in demand for COVID-19–related care engendered numerous innovative solutions. As examples, Dr. Singh recounted how a virtual hospital was created at his own institution. In a published study, 1,477 patients diagnosed with COVID-19 over a 6-week period remained at home and received care in a virtual observation unit (VCU) or a virtual acute care unit (VACU) . Only a small percentage required eventual hospital admission. In the VACU, patients were able to receive advanced care including IV fluids and some form of respiratory support .
It is unclear how the COVID-19 pandemic will change telemedicine. Now, with declining cases of the infection, telemedicine is back to a walk after the sprint required during the height of the pandemic, according to Dr. Singh. However, Dr. Singh thinks many physicians and patients will have a different perception of telemedicine after the widespread exposure to this type of care.
In terms of the relative role of in-patient and virtual visits across indications, “we do not know how this will play out, but we will probably end up toggling between the two,” Dr. Singh said.
This is an area that is being followed closely by the CHEST Health Policy and Advocacy Committee, according to Kathleen Sarmiento, MD, director, VISN 21 Sleep Clinical Resource Hub for the San Francisco VA Health Care System. A member of that committee and moderator of the session in which Dr. Singh spoke,
Dr. Sarmiento called the effort to bring permanent coverage of telehealth services “the shared responsibility of every medical society engaged in advocacy.”
However, she cautioned that there might be intended and unintended consequences from telehealth that require analysis to develop policies that are in the best interests of effective care. She said, the “ACCP, along with its sister societies, does have a role in supporting the evaluation of the impact of these changes on both patients and providers in the fields of pulmonary medicine, critical care, and sleep medicine.”
Dr. Singh reports a financial relationship with AstraZeneca. Dr. Sarmiento reports no relevant financial relationship with AstraZeneca.
Telemedicine has been proposed as a solution for an array of health care access problems over decades of gradual growth. The vast ramping up of , according to an update at the annual health policy and advocacy conference sponsored by the American College of Chest Physicians.
“The cat is out of the bag,” said Jaspal Singh, MD, professor of medicine, Atrium Health, Charlotte, N.C. Due to changes in access and reimbursement to telemedicine driven by the pandemic, he said, “we now have permission to explore new models of care.”
Prior to February 2020, telemedicine was crawling forward at a leisurely pace, according to Dr. Singh. After March 2020, it broke into a run due to enormous demand and was met by a rapid response from the U.S. Congress. The first of four legislative bills that directly or indirectly supported telemedicine was passed on March 6, 2020.
The Centers for Medicare and Medicaid Services responded in kind, making modifications in a number of rules that removed obstacles to telehealth. One modification on April 6, 2020, for example, removed the requirement for a preexisting relationship between the clinician and patient, Dr. Singh said. The CMS also subsequently modified reimbursement policies in order to make telemedicine more tenable for physicians.
Given the risk of contagion from face-to-face encounters, telemedicine in the early days of the pandemic was not just attractive but the only practical and safe approach to medical care in many circumstances. Physicians and patients were anxious for health care that did not require in-office visits even though many critical issues for telemedicine, including its relative effectiveness, had not yet been fully evaluated.
Much has been learned regarding the feasibility and acceptability of telemedicine during the pandemic, but Dr. Singh noted that quality of care relative to in-person visits remains weakly supported for most indications. Indeed, he outlined sizable list of incompletely resolved issues, including optimal payment models, management of privacy concerns, and how to balance advantages to disadvantages.
For patients and physicians, the strengths of telemedicine include greater convenience made possible by the elimination of travel and waiting rooms. For the health care system, it can include less infrastructure and overhead. For many physicians, telemedicine might be perceived as more efficient.
On the other hand, some patients might feel that a clinical encounter is incomplete without a physical examination even when the physician does not feel the physical examination is needed, according to Dr. Singh. He cited a survey suggesting nearly half of patients expressed concern about a lack of connection to health care providers following a virtual visit.
In the same 2020 National Poll on Healthy Aging 2020 survey conducted by the University of Michigan 67% of respondents reported that the quality of care was not as good as that provided by in-patient visits, and 24% expressed concern about privacy. However, at the time the poll was taken in May 2020, experience with telemedicine among many of the respondents may have been limited. As telemedicine is integrated into routine care, perceptions might change as experience increases.
A distinction between telemedicine in routine care and telemedicine as a strategy to respond to a pandemic is important, Dr. Singh indicated. Dr. Singh was the lead author for a position paper on telemedicine for the diagnosis and treatment of sleep disorders from the American Academy of Sleep Medicine 5 years ago, but he acknowledged that models of care might differ when responding to abnormal surges in health care demand.
The surge in demand for COVID-19–related care engendered numerous innovative solutions. As examples, Dr. Singh recounted how a virtual hospital was created at his own institution. In a published study, 1,477 patients diagnosed with COVID-19 over a 6-week period remained at home and received care in a virtual observation unit (VCU) or a virtual acute care unit (VACU) . Only a small percentage required eventual hospital admission. In the VACU, patients were able to receive advanced care including IV fluids and some form of respiratory support .
It is unclear how the COVID-19 pandemic will change telemedicine. Now, with declining cases of the infection, telemedicine is back to a walk after the sprint required during the height of the pandemic, according to Dr. Singh. However, Dr. Singh thinks many physicians and patients will have a different perception of telemedicine after the widespread exposure to this type of care.
In terms of the relative role of in-patient and virtual visits across indications, “we do not know how this will play out, but we will probably end up toggling between the two,” Dr. Singh said.
This is an area that is being followed closely by the CHEST Health Policy and Advocacy Committee, according to Kathleen Sarmiento, MD, director, VISN 21 Sleep Clinical Resource Hub for the San Francisco VA Health Care System. A member of that committee and moderator of the session in which Dr. Singh spoke,
Dr. Sarmiento called the effort to bring permanent coverage of telehealth services “the shared responsibility of every medical society engaged in advocacy.”
However, she cautioned that there might be intended and unintended consequences from telehealth that require analysis to develop policies that are in the best interests of effective care. She said, the “ACCP, along with its sister societies, does have a role in supporting the evaluation of the impact of these changes on both patients and providers in the fields of pulmonary medicine, critical care, and sleep medicine.”
Dr. Singh reports a financial relationship with AstraZeneca. Dr. Sarmiento reports no relevant financial relationship with AstraZeneca.
FROM A HEALTH POLICY AND ADVOCACY CONFERENCE
Low-dose nitrous oxide shows benefit for resistant depression
A 1-hour treatment with a low concentration of nitrous oxide, commonly known as “laughing gas,” appears to relieve symptoms of treatment-resistant major depression (TRMD), with effects lasting as long as several weeks, new research suggests.
In a trial with a crossover design, investigators randomly assigned 28 patients with severe TRMD to receive a single 1-hour inhalation of placebo or nitrous oxide once a month over a 3-month period. Participants received an inhalation of placebo; a 25% concentration of nitrous oxide; and a 50% concentration of nitrous oxide. Sessions were conducted 4 weeks apart.
Both doses of nitrous oxide were associated with substantial improvement in depressive symptoms for roughly 85% of participants. However, the 25% concentration had a lower risk for adverse effects, which included sedation, nausea, and mild dissociation, compared to the 50% concentration.
“Twenty-five percent nitrous oxide has similar efficacy, compared to 50% nitrous oxide, and reduced side effects fourfold,” lead author Peter Nagele, MD, MSc, chair and professor of anesthesia and critical care, University of Chicago, said in an interview.
“We also observed that many patients had a 2-week improvement of depressive symptoms after a nitrous oxide treatment,” said Dr. Nagele, who also is professor of psychiatry and behavioral neuroscience.
The study was published online June 9 in Science Translational Medicine.
Further refinement
A previous proof-of-principle study conducted by the same researchers demonstrated that a 1-hour inhalation of 50% nitrous oxide had rapid antidepressant effects for patients with TRMD.
The current phase 2 trial “is a follow-up study to our earlier 2015 pilot trial and was designed to further refine the dose of nitrous oxide needed for antidepressant efficacy,” Dr. Nagele said.
“An important secondary aim [of the current study] was to determine whether a lower dose – 25% – would reduce side effects, and a third aim was to determine how long the antidepressants effects last,” he explained.
To investigate, the researchers enrolled 28 patients (median [interquartile range (IQR)] age 39 years [26-68 years]; 71% women; 96% White) to have three inhalation sessions (placebo, 25%, and 50% nitrous oxide) at 4-week intervals. Twenty patients completed all three inhalation sessions, and four completed ≥1 treatment.
Participants had “sustained and refractory depressive illness,” with a mean illness lifetime duration of 17.5 years and an extensive history of antidepressant drug failure (median, 4.5 [2-10] adequate-dose/duration antidepressants).
Some patients had undergone vagus nerve stimulation, electroconvulsive therapy, or repetitive transcranial magnetic stimulation, or had received ketamine (4%, 8%, 13%, and 8%, respectively).
The primary outcome was improvement on the 21-item Hamilton Depression rating Scale (HDRS-21) score over a 2-week observation period.
‘Stronger evidence’
Compared to placebo, nitrous oxide significantly improved depressive symptoms (P = .01). There was no significant difference between the 25% and the 50% concentrations (P = .58).
The estimated difference in HDRS-21 scores between the placebo and various treatment groups are shown in the following table.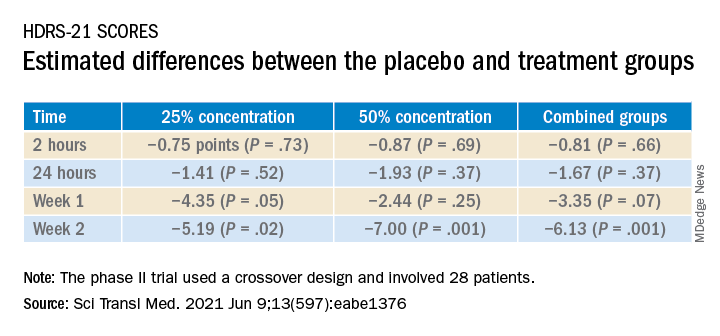
To ensure there where were carryover effects between the two doses, the researchers performed an analysis to ascertain whether order of receipt of the higher dose was related to the 2-week HDRS-21 score; they found no significant effect of trial order (P = .22).
The 20 patients who completed the entire course of treatment “experienced a clinically significant improvement in depressive symptoms from a median baseline HDRS-21 score of 20.5 (IQR, 19.0 to 25.5) to 8.5 (IQR, 2.0 to 16.0) at study completion, corresponding to a median change of −11.0 points (IQR, −3.3 to −14.0 points; P < .0001) after the 3-month study period,” the investigators noted.
The types of treatment response and improvement in depressive symptoms from baseline to study completion are listed in the table below.
There were statistically significant differences in adverse events between the two treatment doses; 47 events occurred following inhalation of the 50% concentration, compared to 11 after inhalation of the 25% concentration. There were six adverse events after inhalation of placebo (P < .0001).
“None of the adverse events were serious, and nearly all occurred either during or immediately after the treatment session and resolved within several hours,” the authors reported.
“We need to be remindful that – despite the exciting results of the study – the study was small and cannot be considered definitive evidence; as such, it is too early to advocate for the use of nitrous oxide in everyday clinical practice,” Dr. Nagele said.
Nevertheless, on the basis of the current findings, he stated.
Rapid-acting antidepressants
Commenting on the study in an interview, Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the Mood Disorders Psychopharmacology Unit at Toronto Western Hospital, noted that the research into nitrous oxide is “part of an interest in rapid-acting antidepressants.”
Dr. McIntyre, also the chairman and executive director of the Brain and Cognition Discovery Foundation, Toronto, who was not involved with the study, found it “interesting” that “almost 20% of the sample had previously had suboptimal outcomes to ketamine and/or neurostimulation, meaning these patients had serious refractory illness, but the benefit [of nitrous oxide] was sustained at 2 weeks.”
Studies of the use of nitrous oxide for patients with bipolar depression “would be warranted, since it appears generally safe and well tolerated,” said Dr. McIntyre, director of the Depression and Bipolar Support Alliance.
The study was sponsored by an award to Dr. Nagele from the NARSAD Independent Investigator Award from the Brain and Behavior Research Foundation and an award to Dr. Nagele and other coauthors from the Taylor Family Institute for Innovative Psychiatric Research at Washington University in St. Louis. Dr. Nagele receives funding from the National Institute of Mental Health the American Foundation for Prevention of Suicide, and the Brain and Behavior Research Foundation; has received research funding and honoraria from Roche Diagnostics and Abbott Diagnostics; and has previously filed for intellectual property protection related to the use of nitrous oxide in major depression. The other authors’ disclosures are listed on the original article. Dr. McIntyre has received research grant support from CIHR/GACD/Chinese National Natural Research Foundation; speaker/consultation fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Eisai, Minerva, Intra-Cellular, and Abbvie. Dr. McIntyre is also CEO of AltMed.
A version of this article first appeared on Medscape.com.
A 1-hour treatment with a low concentration of nitrous oxide, commonly known as “laughing gas,” appears to relieve symptoms of treatment-resistant major depression (TRMD), with effects lasting as long as several weeks, new research suggests.
In a trial with a crossover design, investigators randomly assigned 28 patients with severe TRMD to receive a single 1-hour inhalation of placebo or nitrous oxide once a month over a 3-month period. Participants received an inhalation of placebo; a 25% concentration of nitrous oxide; and a 50% concentration of nitrous oxide. Sessions were conducted 4 weeks apart.
Both doses of nitrous oxide were associated with substantial improvement in depressive symptoms for roughly 85% of participants. However, the 25% concentration had a lower risk for adverse effects, which included sedation, nausea, and mild dissociation, compared to the 50% concentration.
“Twenty-five percent nitrous oxide has similar efficacy, compared to 50% nitrous oxide, and reduced side effects fourfold,” lead author Peter Nagele, MD, MSc, chair and professor of anesthesia and critical care, University of Chicago, said in an interview.
“We also observed that many patients had a 2-week improvement of depressive symptoms after a nitrous oxide treatment,” said Dr. Nagele, who also is professor of psychiatry and behavioral neuroscience.
The study was published online June 9 in Science Translational Medicine.
Further refinement
A previous proof-of-principle study conducted by the same researchers demonstrated that a 1-hour inhalation of 50% nitrous oxide had rapid antidepressant effects for patients with TRMD.
The current phase 2 trial “is a follow-up study to our earlier 2015 pilot trial and was designed to further refine the dose of nitrous oxide needed for antidepressant efficacy,” Dr. Nagele said.
“An important secondary aim [of the current study] was to determine whether a lower dose – 25% – would reduce side effects, and a third aim was to determine how long the antidepressants effects last,” he explained.
To investigate, the researchers enrolled 28 patients (median [interquartile range (IQR)] age 39 years [26-68 years]; 71% women; 96% White) to have three inhalation sessions (placebo, 25%, and 50% nitrous oxide) at 4-week intervals. Twenty patients completed all three inhalation sessions, and four completed ≥1 treatment.
Participants had “sustained and refractory depressive illness,” with a mean illness lifetime duration of 17.5 years and an extensive history of antidepressant drug failure (median, 4.5 [2-10] adequate-dose/duration antidepressants).
Some patients had undergone vagus nerve stimulation, electroconvulsive therapy, or repetitive transcranial magnetic stimulation, or had received ketamine (4%, 8%, 13%, and 8%, respectively).
The primary outcome was improvement on the 21-item Hamilton Depression rating Scale (HDRS-21) score over a 2-week observation period.
‘Stronger evidence’
Compared to placebo, nitrous oxide significantly improved depressive symptoms (P = .01). There was no significant difference between the 25% and the 50% concentrations (P = .58).
The estimated difference in HDRS-21 scores between the placebo and various treatment groups are shown in the following table.
To ensure there where were carryover effects between the two doses, the researchers performed an analysis to ascertain whether order of receipt of the higher dose was related to the 2-week HDRS-21 score; they found no significant effect of trial order (P = .22).
The 20 patients who completed the entire course of treatment “experienced a clinically significant improvement in depressive symptoms from a median baseline HDRS-21 score of 20.5 (IQR, 19.0 to 25.5) to 8.5 (IQR, 2.0 to 16.0) at study completion, corresponding to a median change of −11.0 points (IQR, −3.3 to −14.0 points; P < .0001) after the 3-month study period,” the investigators noted.
The types of treatment response and improvement in depressive symptoms from baseline to study completion are listed in the table below.
There were statistically significant differences in adverse events between the two treatment doses; 47 events occurred following inhalation of the 50% concentration, compared to 11 after inhalation of the 25% concentration. There were six adverse events after inhalation of placebo (P < .0001).
“None of the adverse events were serious, and nearly all occurred either during or immediately after the treatment session and resolved within several hours,” the authors reported.
“We need to be remindful that – despite the exciting results of the study – the study was small and cannot be considered definitive evidence; as such, it is too early to advocate for the use of nitrous oxide in everyday clinical practice,” Dr. Nagele said.
Nevertheless, on the basis of the current findings, he stated.
Rapid-acting antidepressants
Commenting on the study in an interview, Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the Mood Disorders Psychopharmacology Unit at Toronto Western Hospital, noted that the research into nitrous oxide is “part of an interest in rapid-acting antidepressants.”
Dr. McIntyre, also the chairman and executive director of the Brain and Cognition Discovery Foundation, Toronto, who was not involved with the study, found it “interesting” that “almost 20% of the sample had previously had suboptimal outcomes to ketamine and/or neurostimulation, meaning these patients had serious refractory illness, but the benefit [of nitrous oxide] was sustained at 2 weeks.”
Studies of the use of nitrous oxide for patients with bipolar depression “would be warranted, since it appears generally safe and well tolerated,” said Dr. McIntyre, director of the Depression and Bipolar Support Alliance.
The study was sponsored by an award to Dr. Nagele from the NARSAD Independent Investigator Award from the Brain and Behavior Research Foundation and an award to Dr. Nagele and other coauthors from the Taylor Family Institute for Innovative Psychiatric Research at Washington University in St. Louis. Dr. Nagele receives funding from the National Institute of Mental Health the American Foundation for Prevention of Suicide, and the Brain and Behavior Research Foundation; has received research funding and honoraria from Roche Diagnostics and Abbott Diagnostics; and has previously filed for intellectual property protection related to the use of nitrous oxide in major depression. The other authors’ disclosures are listed on the original article. Dr. McIntyre has received research grant support from CIHR/GACD/Chinese National Natural Research Foundation; speaker/consultation fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Eisai, Minerva, Intra-Cellular, and Abbvie. Dr. McIntyre is also CEO of AltMed.
A version of this article first appeared on Medscape.com.
A 1-hour treatment with a low concentration of nitrous oxide, commonly known as “laughing gas,” appears to relieve symptoms of treatment-resistant major depression (TRMD), with effects lasting as long as several weeks, new research suggests.
In a trial with a crossover design, investigators randomly assigned 28 patients with severe TRMD to receive a single 1-hour inhalation of placebo or nitrous oxide once a month over a 3-month period. Participants received an inhalation of placebo; a 25% concentration of nitrous oxide; and a 50% concentration of nitrous oxide. Sessions were conducted 4 weeks apart.
Both doses of nitrous oxide were associated with substantial improvement in depressive symptoms for roughly 85% of participants. However, the 25% concentration had a lower risk for adverse effects, which included sedation, nausea, and mild dissociation, compared to the 50% concentration.
“Twenty-five percent nitrous oxide has similar efficacy, compared to 50% nitrous oxide, and reduced side effects fourfold,” lead author Peter Nagele, MD, MSc, chair and professor of anesthesia and critical care, University of Chicago, said in an interview.
“We also observed that many patients had a 2-week improvement of depressive symptoms after a nitrous oxide treatment,” said Dr. Nagele, who also is professor of psychiatry and behavioral neuroscience.
The study was published online June 9 in Science Translational Medicine.
Further refinement
A previous proof-of-principle study conducted by the same researchers demonstrated that a 1-hour inhalation of 50% nitrous oxide had rapid antidepressant effects for patients with TRMD.
The current phase 2 trial “is a follow-up study to our earlier 2015 pilot trial and was designed to further refine the dose of nitrous oxide needed for antidepressant efficacy,” Dr. Nagele said.
“An important secondary aim [of the current study] was to determine whether a lower dose – 25% – would reduce side effects, and a third aim was to determine how long the antidepressants effects last,” he explained.
To investigate, the researchers enrolled 28 patients (median [interquartile range (IQR)] age 39 years [26-68 years]; 71% women; 96% White) to have three inhalation sessions (placebo, 25%, and 50% nitrous oxide) at 4-week intervals. Twenty patients completed all three inhalation sessions, and four completed ≥1 treatment.
Participants had “sustained and refractory depressive illness,” with a mean illness lifetime duration of 17.5 years and an extensive history of antidepressant drug failure (median, 4.5 [2-10] adequate-dose/duration antidepressants).
Some patients had undergone vagus nerve stimulation, electroconvulsive therapy, or repetitive transcranial magnetic stimulation, or had received ketamine (4%, 8%, 13%, and 8%, respectively).
The primary outcome was improvement on the 21-item Hamilton Depression rating Scale (HDRS-21) score over a 2-week observation period.
‘Stronger evidence’
Compared to placebo, nitrous oxide significantly improved depressive symptoms (P = .01). There was no significant difference between the 25% and the 50% concentrations (P = .58).
The estimated difference in HDRS-21 scores between the placebo and various treatment groups are shown in the following table.
To ensure there where were carryover effects between the two doses, the researchers performed an analysis to ascertain whether order of receipt of the higher dose was related to the 2-week HDRS-21 score; they found no significant effect of trial order (P = .22).
The 20 patients who completed the entire course of treatment “experienced a clinically significant improvement in depressive symptoms from a median baseline HDRS-21 score of 20.5 (IQR, 19.0 to 25.5) to 8.5 (IQR, 2.0 to 16.0) at study completion, corresponding to a median change of −11.0 points (IQR, −3.3 to −14.0 points; P < .0001) after the 3-month study period,” the investigators noted.
The types of treatment response and improvement in depressive symptoms from baseline to study completion are listed in the table below.
There were statistically significant differences in adverse events between the two treatment doses; 47 events occurred following inhalation of the 50% concentration, compared to 11 after inhalation of the 25% concentration. There were six adverse events after inhalation of placebo (P < .0001).
“None of the adverse events were serious, and nearly all occurred either during or immediately after the treatment session and resolved within several hours,” the authors reported.
“We need to be remindful that – despite the exciting results of the study – the study was small and cannot be considered definitive evidence; as such, it is too early to advocate for the use of nitrous oxide in everyday clinical practice,” Dr. Nagele said.
Nevertheless, on the basis of the current findings, he stated.
Rapid-acting antidepressants
Commenting on the study in an interview, Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the Mood Disorders Psychopharmacology Unit at Toronto Western Hospital, noted that the research into nitrous oxide is “part of an interest in rapid-acting antidepressants.”
Dr. McIntyre, also the chairman and executive director of the Brain and Cognition Discovery Foundation, Toronto, who was not involved with the study, found it “interesting” that “almost 20% of the sample had previously had suboptimal outcomes to ketamine and/or neurostimulation, meaning these patients had serious refractory illness, but the benefit [of nitrous oxide] was sustained at 2 weeks.”
Studies of the use of nitrous oxide for patients with bipolar depression “would be warranted, since it appears generally safe and well tolerated,” said Dr. McIntyre, director of the Depression and Bipolar Support Alliance.
The study was sponsored by an award to Dr. Nagele from the NARSAD Independent Investigator Award from the Brain and Behavior Research Foundation and an award to Dr. Nagele and other coauthors from the Taylor Family Institute for Innovative Psychiatric Research at Washington University in St. Louis. Dr. Nagele receives funding from the National Institute of Mental Health the American Foundation for Prevention of Suicide, and the Brain and Behavior Research Foundation; has received research funding and honoraria from Roche Diagnostics and Abbott Diagnostics; and has previously filed for intellectual property protection related to the use of nitrous oxide in major depression. The other authors’ disclosures are listed on the original article. Dr. McIntyre has received research grant support from CIHR/GACD/Chinese National Natural Research Foundation; speaker/consultation fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Eisai, Minerva, Intra-Cellular, and Abbvie. Dr. McIntyre is also CEO of AltMed.
A version of this article first appeared on Medscape.com.
Preparing pediatric hospital medicine fellows for leadership
Reflecting on a longitudinal leadership elective experience
The practice of pediatric hospital medicine (PHM) has been evolving and rapidly expanding over the last several decades. Not only has the scope of clinical practice matured and become more defined, but hospitalists now also have the responsibility to advance the performance of hospitals and health care systems. Pediatric hospitalists are increasingly incorporating medical education, research, high-value care, patient quality and safety initiatives, and process improvement into their careers.1 As a result, pediatric hospitalists are occupying a wider range of administrative and leadership positions within the health care system.
The field of PHM has highlighted the importance of leadership in the practice of hospital medicine by dedicating a chapter to “Leadership in Healthcare” in the PHM Core Competencies.1 The competencies define the expertise required of hospitalists and serve as guidance for the development of education, training, and career development series. Hospitalists may seek out opportunities for leadership training at an institutional or national level. Options may include advanced degrees, national conferences, division training seminars, or self-directed learning through reading or observational experiences. Unfortunately, all of these take time and motivation. As a result, hospitalists tend to pursue these opportunities only after they have already been appointed to leadership positions.
PHM fellowship is the optimal time to build a foundation of leadership skills. Over the course of a 2-year fellowship, fellows have a combined 16 weeks dedicated to educational activities beyond direct patient care.2 The Accreditation Council for Graduate Medical Education (ACGME) encourages educational innovation during this time, allowing programs to create unique opportunities for their fellows that will promote progress towards their ultimate career goals.3 This curricular framework provides the flexibility to integrate leadership training into fellowship training.
Many fellows are eager for leadership experiences and mentorship, myself included. As a pediatric chief resident, I was immersed in a diverse range of clinical, educational, research, and administrative responsibilities. I found myself in a leadership position with no prior education on how to manage people or team dynamics, make high-stress decisions on behalf of a group of people, or handle conflict. Although I learned new strategies on a daily basis, the experience showed me how much more I still had to learn in order to be a successful leader. This was one of the reasons I decided to pursue fellowship training. I think many PHM fellowship applicants feel similarly. They may have served in a leadership position in the past but feel underprepared to fulfill leadership positions in the next phase of their careers.
But despite this eagerness, evidence suggests that fellows do not feel that they receive as much management training as they need to start their careers. In a 2014 survey of PHM fellowship graduates, many held formal leadership positions within their institution (23/51) and within national organizations (6/51), despite having only five years of hospitalist experience on average (including time spent in fellowship). When asked about training needs, respondents identified “hospital program management” as an area where they wished they received more training during fellowship.4
Anyone who has gone through the PHM fellowship interview process can tell you that a common refrain of program directors is, “One of the goals of our program is to create future leaders in PHM.” This led me to wonder: how do fellowship programs prepare their fellows for future leadership positions?
I began my fellowship training at Nationwide Children’s Hospital in the summer of 2020. The program had just designed a longitudinal leadership elective, which the second-year fellow and I decided to pilot together. As I reflected on the first half of this academic year, I realized that it is unique experiences like this elective that make me thankful I pursued fellowship. I want to share with the hospitalist community the structure of the elective and why it has been particularly valuable with the hope that it will inspire similar opportunities for other fellows.
The program is semi-structured but allows the fellow and preceptors the flexibility to decide what activities would benefit that particular fellow. We attend a variety of administrative and committee meetings with each preceptor that expose us to the responsibilities of their positions, their leadership style in action, their approach to crisis management, and differences in divisional operations. On a monthly basis we meet with a preceptor to discuss a topic related to leadership. Examples of topics include how to run a more effective meeting, barriers to organizational change, leading in crisis, and the importance of mission, vision, values, and goals of organizations. The preceptor sends us articles or other learning materials they have found useful on the topic, and these serve as a starting point for our discussions. These discussions provide a point of reflection as we apply the day’s concept to our own prior experiences or to our observations during the elective.
The combination of learning experiences, discussions, and dedicated preceptorship has prepared me far better for future leadership than my past personal and observational experiences. I have summarized my top three reasons why this structure of leadership development is particularly valuable to me as a fellow.
First, the longitudinal structure of the elective allows us to learn from multiple preceptors over the course of the academic year. The preceptors include the current chief of hospital pediatrics at Nationwide Children’s Hospital; the division director of hospital medicine at the Ohio State University Wexner Medical Center; and the physician lead for hospital medicine at one of the satellite hospitals in the region. With faculty from the Department of Pediatrics and the Department of Internal Medicine-Pediatrics in these leadership positions, we have the unique ability to compare and contrast operational systems between the two different hospital systems.
Recently, we also had the opportunity to meet with both the chairman of the department of pediatrics and chief medical officer. All of these physician leaders hold a variety of administrative roles and have differing leadership philosophies, each providing useful insights. For instance, one leader ensures his team holds him accountable as the leader by always asking for honest feedback. He recommends telling those you work with to “never let me fail.” Another leader acknowledges that creating five-year plans can be daunting but encouraged us to still be intentional with our direction on a smaller scale by writing down goals for the year and sharing with a mentor. Ultimately, I came away with a wide variety of perspectives to reference as I go forward.
Second, the learning is contextualized. I can take concepts that I learn through reading and discussions and construct meaning based on observations from meetings or other encounters with different leaders. For example, after reviewing several articles on strategies to make meetings more effective, I started noticing what went well and what didn’t go well in every meeting I attended. I observed preceptors employing many of the strategies successfully with positive feedback. This included not only simple practices, such as setting an agenda to provide a compass for the conversation, but also more nuanced practices like controlling the meeting but not the conversation.
After reading about leadership styles I also found myself analyzing the qualities and strategies of leaders I encountered and reflecting on their approach, noticing what I could possibly interlace in my own practice. Several of the leaders I spoke with during the elective recommended paying attention to the actions of the ineffective bosses or mentors because they can teach you something too: how not to act. I even started applying this strategy to the popular television series The Office – Michael Scott, the regional manager of a fictional paper company, demonstrates some of the best and worst leadership skills in every episode. I am developing a repertoire of strategies to lead and motivate people.
Finally, the design allows for real-time application of new methods to my current practice. One particularly useful tool I have learned is Leader Standard Work, a systematic method to get leaders to maintain stability, problem solve, and drive continuous improvement within their organization.5 I have used elements of Leader Standard Work on a personal level to improve my time management skills and increase my productivity. For example, I reconceptualized my calendar as a standardized checklist and I organized it to allot more time to critical activities, such as my research and scholarly output, and less on administrative tasks. I am also implementing changes to how I prepare and run meetings, collaborate, and communicate with members of my research team.
Mastery requires practice and feedback, so applying concepts even on a small, personal scale shortly after learning them has been very valuable. Over the last several months I have often wished I had this type of structured leadership education during my year as a chief resident. I think I could have been more intentional in my decision-making, possibly being a stronger leader for the program. Now that I am transferring skills into practice right away, I am setting the stage for lasting changes in behavior that will hopefully benefit all those that I work with in the future.
Leadership development through a customizable longitudinal elective may be an effective way to prepare PHM fellow graduates for future leadership positions. Fellows can emerge with the skills and real-world practice to allow them to feel confident in future positions. However, leadership doesn’t end when we get the position. We must remember to continuously ask for feedback and build upon our experiences to evolve as leaders in PHM.
Dr. Westphal is a first-year pediatric hospital medicine fellow at Nationwide Children’s Hospital in Columbus, Ohio with an interest in improving the delivery of quality care for hospitalized infants.
References
1. Maniscalco, J, et al. The Pediatric Hospital Medicine Core Competencies: 2020 Revision. Introduction and Methodology (C). J Hosp Med. 2020;S1;E12-E17. doi: 10.12788/jhm.3391.
2. Jerardi KE, et al; Council of Pediatric Hospital Medicine Fellowship Directors. Development of a Curricular Framework for Pediatric Hospital Medicine Fellowships. Pediatrics. 2017 Jul;140(1):e20170698. doi: 10.1542/peds.2017-0698.
3. ACGME Program Requirements for Graduate Medical Education in Pediatric Hospital Medicine. 2020 Edition. Accessed 2021 Jan 14.
4. Oshimura, JM et al. Current roles and perceived needs of pediatric hospital medicine fellowship graduates. Hosp Pediatr. 2016;6(10):633-7. doi: 10.1542/hpeds.2016-0031.
5. Murli, J. Standard Work for Lean Leaders: One of the Keys to Sustaining Performance Gains. Lean Institute Enterprise, Lean Institute Enterprise Inc. 4 Dec 2013. www.lean.org/common/display/?o=2493
Reflecting on a longitudinal leadership elective experience
Reflecting on a longitudinal leadership elective experience
The practice of pediatric hospital medicine (PHM) has been evolving and rapidly expanding over the last several decades. Not only has the scope of clinical practice matured and become more defined, but hospitalists now also have the responsibility to advance the performance of hospitals and health care systems. Pediatric hospitalists are increasingly incorporating medical education, research, high-value care, patient quality and safety initiatives, and process improvement into their careers.1 As a result, pediatric hospitalists are occupying a wider range of administrative and leadership positions within the health care system.
The field of PHM has highlighted the importance of leadership in the practice of hospital medicine by dedicating a chapter to “Leadership in Healthcare” in the PHM Core Competencies.1 The competencies define the expertise required of hospitalists and serve as guidance for the development of education, training, and career development series. Hospitalists may seek out opportunities for leadership training at an institutional or national level. Options may include advanced degrees, national conferences, division training seminars, or self-directed learning through reading or observational experiences. Unfortunately, all of these take time and motivation. As a result, hospitalists tend to pursue these opportunities only after they have already been appointed to leadership positions.
PHM fellowship is the optimal time to build a foundation of leadership skills. Over the course of a 2-year fellowship, fellows have a combined 16 weeks dedicated to educational activities beyond direct patient care.2 The Accreditation Council for Graduate Medical Education (ACGME) encourages educational innovation during this time, allowing programs to create unique opportunities for their fellows that will promote progress towards their ultimate career goals.3 This curricular framework provides the flexibility to integrate leadership training into fellowship training.
Many fellows are eager for leadership experiences and mentorship, myself included. As a pediatric chief resident, I was immersed in a diverse range of clinical, educational, research, and administrative responsibilities. I found myself in a leadership position with no prior education on how to manage people or team dynamics, make high-stress decisions on behalf of a group of people, or handle conflict. Although I learned new strategies on a daily basis, the experience showed me how much more I still had to learn in order to be a successful leader. This was one of the reasons I decided to pursue fellowship training. I think many PHM fellowship applicants feel similarly. They may have served in a leadership position in the past but feel underprepared to fulfill leadership positions in the next phase of their careers.
But despite this eagerness, evidence suggests that fellows do not feel that they receive as much management training as they need to start their careers. In a 2014 survey of PHM fellowship graduates, many held formal leadership positions within their institution (23/51) and within national organizations (6/51), despite having only five years of hospitalist experience on average (including time spent in fellowship). When asked about training needs, respondents identified “hospital program management” as an area where they wished they received more training during fellowship.4
Anyone who has gone through the PHM fellowship interview process can tell you that a common refrain of program directors is, “One of the goals of our program is to create future leaders in PHM.” This led me to wonder: how do fellowship programs prepare their fellows for future leadership positions?
I began my fellowship training at Nationwide Children’s Hospital in the summer of 2020. The program had just designed a longitudinal leadership elective, which the second-year fellow and I decided to pilot together. As I reflected on the first half of this academic year, I realized that it is unique experiences like this elective that make me thankful I pursued fellowship. I want to share with the hospitalist community the structure of the elective and why it has been particularly valuable with the hope that it will inspire similar opportunities for other fellows.
The program is semi-structured but allows the fellow and preceptors the flexibility to decide what activities would benefit that particular fellow. We attend a variety of administrative and committee meetings with each preceptor that expose us to the responsibilities of their positions, their leadership style in action, their approach to crisis management, and differences in divisional operations. On a monthly basis we meet with a preceptor to discuss a topic related to leadership. Examples of topics include how to run a more effective meeting, barriers to organizational change, leading in crisis, and the importance of mission, vision, values, and goals of organizations. The preceptor sends us articles or other learning materials they have found useful on the topic, and these serve as a starting point for our discussions. These discussions provide a point of reflection as we apply the day’s concept to our own prior experiences or to our observations during the elective.
The combination of learning experiences, discussions, and dedicated preceptorship has prepared me far better for future leadership than my past personal and observational experiences. I have summarized my top three reasons why this structure of leadership development is particularly valuable to me as a fellow.
First, the longitudinal structure of the elective allows us to learn from multiple preceptors over the course of the academic year. The preceptors include the current chief of hospital pediatrics at Nationwide Children’s Hospital; the division director of hospital medicine at the Ohio State University Wexner Medical Center; and the physician lead for hospital medicine at one of the satellite hospitals in the region. With faculty from the Department of Pediatrics and the Department of Internal Medicine-Pediatrics in these leadership positions, we have the unique ability to compare and contrast operational systems between the two different hospital systems.
Recently, we also had the opportunity to meet with both the chairman of the department of pediatrics and chief medical officer. All of these physician leaders hold a variety of administrative roles and have differing leadership philosophies, each providing useful insights. For instance, one leader ensures his team holds him accountable as the leader by always asking for honest feedback. He recommends telling those you work with to “never let me fail.” Another leader acknowledges that creating five-year plans can be daunting but encouraged us to still be intentional with our direction on a smaller scale by writing down goals for the year and sharing with a mentor. Ultimately, I came away with a wide variety of perspectives to reference as I go forward.
Second, the learning is contextualized. I can take concepts that I learn through reading and discussions and construct meaning based on observations from meetings or other encounters with different leaders. For example, after reviewing several articles on strategies to make meetings more effective, I started noticing what went well and what didn’t go well in every meeting I attended. I observed preceptors employing many of the strategies successfully with positive feedback. This included not only simple practices, such as setting an agenda to provide a compass for the conversation, but also more nuanced practices like controlling the meeting but not the conversation.
After reading about leadership styles I also found myself analyzing the qualities and strategies of leaders I encountered and reflecting on their approach, noticing what I could possibly interlace in my own practice. Several of the leaders I spoke with during the elective recommended paying attention to the actions of the ineffective bosses or mentors because they can teach you something too: how not to act. I even started applying this strategy to the popular television series The Office – Michael Scott, the regional manager of a fictional paper company, demonstrates some of the best and worst leadership skills in every episode. I am developing a repertoire of strategies to lead and motivate people.
Finally, the design allows for real-time application of new methods to my current practice. One particularly useful tool I have learned is Leader Standard Work, a systematic method to get leaders to maintain stability, problem solve, and drive continuous improvement within their organization.5 I have used elements of Leader Standard Work on a personal level to improve my time management skills and increase my productivity. For example, I reconceptualized my calendar as a standardized checklist and I organized it to allot more time to critical activities, such as my research and scholarly output, and less on administrative tasks. I am also implementing changes to how I prepare and run meetings, collaborate, and communicate with members of my research team.
Mastery requires practice and feedback, so applying concepts even on a small, personal scale shortly after learning them has been very valuable. Over the last several months I have often wished I had this type of structured leadership education during my year as a chief resident. I think I could have been more intentional in my decision-making, possibly being a stronger leader for the program. Now that I am transferring skills into practice right away, I am setting the stage for lasting changes in behavior that will hopefully benefit all those that I work with in the future.
Leadership development through a customizable longitudinal elective may be an effective way to prepare PHM fellow graduates for future leadership positions. Fellows can emerge with the skills and real-world practice to allow them to feel confident in future positions. However, leadership doesn’t end when we get the position. We must remember to continuously ask for feedback and build upon our experiences to evolve as leaders in PHM.
Dr. Westphal is a first-year pediatric hospital medicine fellow at Nationwide Children’s Hospital in Columbus, Ohio with an interest in improving the delivery of quality care for hospitalized infants.
References
1. Maniscalco, J, et al. The Pediatric Hospital Medicine Core Competencies: 2020 Revision. Introduction and Methodology (C). J Hosp Med. 2020;S1;E12-E17. doi: 10.12788/jhm.3391.
2. Jerardi KE, et al; Council of Pediatric Hospital Medicine Fellowship Directors. Development of a Curricular Framework for Pediatric Hospital Medicine Fellowships. Pediatrics. 2017 Jul;140(1):e20170698. doi: 10.1542/peds.2017-0698.
3. ACGME Program Requirements for Graduate Medical Education in Pediatric Hospital Medicine. 2020 Edition. Accessed 2021 Jan 14.
4. Oshimura, JM et al. Current roles and perceived needs of pediatric hospital medicine fellowship graduates. Hosp Pediatr. 2016;6(10):633-7. doi: 10.1542/hpeds.2016-0031.
5. Murli, J. Standard Work for Lean Leaders: One of the Keys to Sustaining Performance Gains. Lean Institute Enterprise, Lean Institute Enterprise Inc. 4 Dec 2013. www.lean.org/common/display/?o=2493
The practice of pediatric hospital medicine (PHM) has been evolving and rapidly expanding over the last several decades. Not only has the scope of clinical practice matured and become more defined, but hospitalists now also have the responsibility to advance the performance of hospitals and health care systems. Pediatric hospitalists are increasingly incorporating medical education, research, high-value care, patient quality and safety initiatives, and process improvement into their careers.1 As a result, pediatric hospitalists are occupying a wider range of administrative and leadership positions within the health care system.
The field of PHM has highlighted the importance of leadership in the practice of hospital medicine by dedicating a chapter to “Leadership in Healthcare” in the PHM Core Competencies.1 The competencies define the expertise required of hospitalists and serve as guidance for the development of education, training, and career development series. Hospitalists may seek out opportunities for leadership training at an institutional or national level. Options may include advanced degrees, national conferences, division training seminars, or self-directed learning through reading or observational experiences. Unfortunately, all of these take time and motivation. As a result, hospitalists tend to pursue these opportunities only after they have already been appointed to leadership positions.
PHM fellowship is the optimal time to build a foundation of leadership skills. Over the course of a 2-year fellowship, fellows have a combined 16 weeks dedicated to educational activities beyond direct patient care.2 The Accreditation Council for Graduate Medical Education (ACGME) encourages educational innovation during this time, allowing programs to create unique opportunities for their fellows that will promote progress towards their ultimate career goals.3 This curricular framework provides the flexibility to integrate leadership training into fellowship training.
Many fellows are eager for leadership experiences and mentorship, myself included. As a pediatric chief resident, I was immersed in a diverse range of clinical, educational, research, and administrative responsibilities. I found myself in a leadership position with no prior education on how to manage people or team dynamics, make high-stress decisions on behalf of a group of people, or handle conflict. Although I learned new strategies on a daily basis, the experience showed me how much more I still had to learn in order to be a successful leader. This was one of the reasons I decided to pursue fellowship training. I think many PHM fellowship applicants feel similarly. They may have served in a leadership position in the past but feel underprepared to fulfill leadership positions in the next phase of their careers.
But despite this eagerness, evidence suggests that fellows do not feel that they receive as much management training as they need to start their careers. In a 2014 survey of PHM fellowship graduates, many held formal leadership positions within their institution (23/51) and within national organizations (6/51), despite having only five years of hospitalist experience on average (including time spent in fellowship). When asked about training needs, respondents identified “hospital program management” as an area where they wished they received more training during fellowship.4
Anyone who has gone through the PHM fellowship interview process can tell you that a common refrain of program directors is, “One of the goals of our program is to create future leaders in PHM.” This led me to wonder: how do fellowship programs prepare their fellows for future leadership positions?
I began my fellowship training at Nationwide Children’s Hospital in the summer of 2020. The program had just designed a longitudinal leadership elective, which the second-year fellow and I decided to pilot together. As I reflected on the first half of this academic year, I realized that it is unique experiences like this elective that make me thankful I pursued fellowship. I want to share with the hospitalist community the structure of the elective and why it has been particularly valuable with the hope that it will inspire similar opportunities for other fellows.
The program is semi-structured but allows the fellow and preceptors the flexibility to decide what activities would benefit that particular fellow. We attend a variety of administrative and committee meetings with each preceptor that expose us to the responsibilities of their positions, their leadership style in action, their approach to crisis management, and differences in divisional operations. On a monthly basis we meet with a preceptor to discuss a topic related to leadership. Examples of topics include how to run a more effective meeting, barriers to organizational change, leading in crisis, and the importance of mission, vision, values, and goals of organizations. The preceptor sends us articles or other learning materials they have found useful on the topic, and these serve as a starting point for our discussions. These discussions provide a point of reflection as we apply the day’s concept to our own prior experiences or to our observations during the elective.
The combination of learning experiences, discussions, and dedicated preceptorship has prepared me far better for future leadership than my past personal and observational experiences. I have summarized my top three reasons why this structure of leadership development is particularly valuable to me as a fellow.
First, the longitudinal structure of the elective allows us to learn from multiple preceptors over the course of the academic year. The preceptors include the current chief of hospital pediatrics at Nationwide Children’s Hospital; the division director of hospital medicine at the Ohio State University Wexner Medical Center; and the physician lead for hospital medicine at one of the satellite hospitals in the region. With faculty from the Department of Pediatrics and the Department of Internal Medicine-Pediatrics in these leadership positions, we have the unique ability to compare and contrast operational systems between the two different hospital systems.
Recently, we also had the opportunity to meet with both the chairman of the department of pediatrics and chief medical officer. All of these physician leaders hold a variety of administrative roles and have differing leadership philosophies, each providing useful insights. For instance, one leader ensures his team holds him accountable as the leader by always asking for honest feedback. He recommends telling those you work with to “never let me fail.” Another leader acknowledges that creating five-year plans can be daunting but encouraged us to still be intentional with our direction on a smaller scale by writing down goals for the year and sharing with a mentor. Ultimately, I came away with a wide variety of perspectives to reference as I go forward.
Second, the learning is contextualized. I can take concepts that I learn through reading and discussions and construct meaning based on observations from meetings or other encounters with different leaders. For example, after reviewing several articles on strategies to make meetings more effective, I started noticing what went well and what didn’t go well in every meeting I attended. I observed preceptors employing many of the strategies successfully with positive feedback. This included not only simple practices, such as setting an agenda to provide a compass for the conversation, but also more nuanced practices like controlling the meeting but not the conversation.
After reading about leadership styles I also found myself analyzing the qualities and strategies of leaders I encountered and reflecting on their approach, noticing what I could possibly interlace in my own practice. Several of the leaders I spoke with during the elective recommended paying attention to the actions of the ineffective bosses or mentors because they can teach you something too: how not to act. I even started applying this strategy to the popular television series The Office – Michael Scott, the regional manager of a fictional paper company, demonstrates some of the best and worst leadership skills in every episode. I am developing a repertoire of strategies to lead and motivate people.
Finally, the design allows for real-time application of new methods to my current practice. One particularly useful tool I have learned is Leader Standard Work, a systematic method to get leaders to maintain stability, problem solve, and drive continuous improvement within their organization.5 I have used elements of Leader Standard Work on a personal level to improve my time management skills and increase my productivity. For example, I reconceptualized my calendar as a standardized checklist and I organized it to allot more time to critical activities, such as my research and scholarly output, and less on administrative tasks. I am also implementing changes to how I prepare and run meetings, collaborate, and communicate with members of my research team.
Mastery requires practice and feedback, so applying concepts even on a small, personal scale shortly after learning them has been very valuable. Over the last several months I have often wished I had this type of structured leadership education during my year as a chief resident. I think I could have been more intentional in my decision-making, possibly being a stronger leader for the program. Now that I am transferring skills into practice right away, I am setting the stage for lasting changes in behavior that will hopefully benefit all those that I work with in the future.
Leadership development through a customizable longitudinal elective may be an effective way to prepare PHM fellow graduates for future leadership positions. Fellows can emerge with the skills and real-world practice to allow them to feel confident in future positions. However, leadership doesn’t end when we get the position. We must remember to continuously ask for feedback and build upon our experiences to evolve as leaders in PHM.
Dr. Westphal is a first-year pediatric hospital medicine fellow at Nationwide Children’s Hospital in Columbus, Ohio with an interest in improving the delivery of quality care for hospitalized infants.
References
1. Maniscalco, J, et al. The Pediatric Hospital Medicine Core Competencies: 2020 Revision. Introduction and Methodology (C). J Hosp Med. 2020;S1;E12-E17. doi: 10.12788/jhm.3391.
2. Jerardi KE, et al; Council of Pediatric Hospital Medicine Fellowship Directors. Development of a Curricular Framework for Pediatric Hospital Medicine Fellowships. Pediatrics. 2017 Jul;140(1):e20170698. doi: 10.1542/peds.2017-0698.
3. ACGME Program Requirements for Graduate Medical Education in Pediatric Hospital Medicine. 2020 Edition. Accessed 2021 Jan 14.
4. Oshimura, JM et al. Current roles and perceived needs of pediatric hospital medicine fellowship graduates. Hosp Pediatr. 2016;6(10):633-7. doi: 10.1542/hpeds.2016-0031.
5. Murli, J. Standard Work for Lean Leaders: One of the Keys to Sustaining Performance Gains. Lean Institute Enterprise, Lean Institute Enterprise Inc. 4 Dec 2013. www.lean.org/common/display/?o=2493








