User login
For MD-IQ use only
More eczema in children exposed to toxic metals in utero
published Oct. 27, 2021, in JAMA Network Open.
In this multicenter cohort study, led by epidemiologist Shu-Li Wang, PhD, of the National Institute of Environmental Health Sciences, in Taiwan, each twofold increase in prenatal arsenic level correlated with a 2.4-fold higher rate of atopic dermatitis in 4-year-olds.
Atopic diseases have been on the rise. Eczema (atopic dermatitis) is the first stage of the so-called atopic march, followed by food allergies, allergic rhinitis, and asthma later in childhood. Previous research has linked heavy metal exposure to allergic diseases in adults. In another study by Dr. Wang and colleagues that was published in 2021, prenatal and early-life arsenic exposure was found to correlate with higher rates of allergic rhinitis and asthma in children. In that study, the participants were followed every 2-3 years through the age of 14 as part of the Taiwan Maternal and Infant Cohort Study.
The new study included 370 mother and child pairs who were enrolled in that birth cohort study between October 2012 and May 2015. During their third trimester of pregnancy, women completed questionnaires about their lifestyle, diet, and living environment. In addition, their height, weight, and blood pressure were recorded, and urine samples were taken. In follow-up interviews 3-4 years later, the mothers were asked whether their child had ever been diagnosed with atopic dermatitis.
The researchers used an inductively coupled plasma mass spectrometer to analyze the participants’ urine samples. They assessed for exposures in utero to eight metals: arsenic, cadmium, lead, cobalt, copper, nickel, thallium, and zinc.
Each unit increase of an index that estimates the combined exposure to these metals during pregnancy was associated with 63% higher odds of atopic dermatitis in the children by age 4. The researchers adjusted for parental allergies (yes or no), mother’s educational level (<12 years, 13-16 years, or >16 years), geographic area (central or eastern Taiwan), exposure to tobacco smoke during pregnancy, and the child’s gender. Arsenic (40.1%) and cadmium (20.5%) accounted for most of the metal coexposure index.
A wealth of previous research links arsenic exposure during adulthood to skin disease and immune dysfunction. Early-life arsenic exposure has been linked with elevated risk for various adult disorders, including cancer, diabetes, and heart disease, years later. In light of such research, “the findings in this paper are not surprising,” J. Christopher States, PhD, director of the Center for Integrative Environmental Health Science at the University of Louisville (Ky.), told this news organization. “Low-level arsenic exposure does not cause disease immediately, but it does appear to have long-lasting effects, making individuals susceptible to ‘second hits’ with another environmental agent.”
Research into the molecular mechanisms for these links has shown that arsenic and cadmium exposure can promote allergic phenotypes in immune cells. “We think the toxic metals activate the alarmin pathway, thus inducing innate lymphoid cells, then activating T-helper 2 cells, which drive immunoglobulin E production and breakdown of the epithelium and promotion of allergies,” said Kari Nadeau, MD, PhD, director of the Sean N. Parker Center for Allergy and Asthma Research at Stanford University. Dr. Nadeau led that study, published in 2017 in PLOS One, along with epidemiologist Margaret Karagas, PhD, of Geisel School of Medicine at Dartmouth, Hanover, N.H.
As for what pregnant women can do to minimize their exposure to heavy metals, “that is a difficult problem and primarily a function of where one lives,” said Dr. States.
Drinking water and food are major sources of arsenic exposure. Groundwater is naturally contaminated with arsenic deposits that seep in from bedrock, said Dr. States. The U.S. Environmental Protection Agency regulates arsenic levels in public drinking water that is supplied to more than a few thousand people. However, small water supplies and private wells are unregulated, he said, and having these water sources tested for arsenic or fitted with systems to reduce arsenic can be very expensive.
Among foods, rice can have high concentrations of arsenic, Dr. Karagas told this news organization. To minimize arsenic exposure through the diet, women can limit rice-based foods, according to a web-based tool developed by her and coworkers.
In addition, tobacco smoke is a major source of cadmium exposure and a moderate source of arsenic exposure, Dr. States noted. Women can reduce their exposure to these metals by avoiding tobacco and secondhand smoke.
The study was supported by grants from the National Health Research Institutes, Chung Shan Medical University Hospital, Taiwan Ministry of Science and Technology, and the Taiwan Environmental Protection Administration. The authors and quoted experts report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published Oct. 27, 2021, in JAMA Network Open.
In this multicenter cohort study, led by epidemiologist Shu-Li Wang, PhD, of the National Institute of Environmental Health Sciences, in Taiwan, each twofold increase in prenatal arsenic level correlated with a 2.4-fold higher rate of atopic dermatitis in 4-year-olds.
Atopic diseases have been on the rise. Eczema (atopic dermatitis) is the first stage of the so-called atopic march, followed by food allergies, allergic rhinitis, and asthma later in childhood. Previous research has linked heavy metal exposure to allergic diseases in adults. In another study by Dr. Wang and colleagues that was published in 2021, prenatal and early-life arsenic exposure was found to correlate with higher rates of allergic rhinitis and asthma in children. In that study, the participants were followed every 2-3 years through the age of 14 as part of the Taiwan Maternal and Infant Cohort Study.
The new study included 370 mother and child pairs who were enrolled in that birth cohort study between October 2012 and May 2015. During their third trimester of pregnancy, women completed questionnaires about their lifestyle, diet, and living environment. In addition, their height, weight, and blood pressure were recorded, and urine samples were taken. In follow-up interviews 3-4 years later, the mothers were asked whether their child had ever been diagnosed with atopic dermatitis.
The researchers used an inductively coupled plasma mass spectrometer to analyze the participants’ urine samples. They assessed for exposures in utero to eight metals: arsenic, cadmium, lead, cobalt, copper, nickel, thallium, and zinc.
Each unit increase of an index that estimates the combined exposure to these metals during pregnancy was associated with 63% higher odds of atopic dermatitis in the children by age 4. The researchers adjusted for parental allergies (yes or no), mother’s educational level (<12 years, 13-16 years, or >16 years), geographic area (central or eastern Taiwan), exposure to tobacco smoke during pregnancy, and the child’s gender. Arsenic (40.1%) and cadmium (20.5%) accounted for most of the metal coexposure index.
A wealth of previous research links arsenic exposure during adulthood to skin disease and immune dysfunction. Early-life arsenic exposure has been linked with elevated risk for various adult disorders, including cancer, diabetes, and heart disease, years later. In light of such research, “the findings in this paper are not surprising,” J. Christopher States, PhD, director of the Center for Integrative Environmental Health Science at the University of Louisville (Ky.), told this news organization. “Low-level arsenic exposure does not cause disease immediately, but it does appear to have long-lasting effects, making individuals susceptible to ‘second hits’ with another environmental agent.”
Research into the molecular mechanisms for these links has shown that arsenic and cadmium exposure can promote allergic phenotypes in immune cells. “We think the toxic metals activate the alarmin pathway, thus inducing innate lymphoid cells, then activating T-helper 2 cells, which drive immunoglobulin E production and breakdown of the epithelium and promotion of allergies,” said Kari Nadeau, MD, PhD, director of the Sean N. Parker Center for Allergy and Asthma Research at Stanford University. Dr. Nadeau led that study, published in 2017 in PLOS One, along with epidemiologist Margaret Karagas, PhD, of Geisel School of Medicine at Dartmouth, Hanover, N.H.
As for what pregnant women can do to minimize their exposure to heavy metals, “that is a difficult problem and primarily a function of where one lives,” said Dr. States.
Drinking water and food are major sources of arsenic exposure. Groundwater is naturally contaminated with arsenic deposits that seep in from bedrock, said Dr. States. The U.S. Environmental Protection Agency regulates arsenic levels in public drinking water that is supplied to more than a few thousand people. However, small water supplies and private wells are unregulated, he said, and having these water sources tested for arsenic or fitted with systems to reduce arsenic can be very expensive.
Among foods, rice can have high concentrations of arsenic, Dr. Karagas told this news organization. To minimize arsenic exposure through the diet, women can limit rice-based foods, according to a web-based tool developed by her and coworkers.
In addition, tobacco smoke is a major source of cadmium exposure and a moderate source of arsenic exposure, Dr. States noted. Women can reduce their exposure to these metals by avoiding tobacco and secondhand smoke.
The study was supported by grants from the National Health Research Institutes, Chung Shan Medical University Hospital, Taiwan Ministry of Science and Technology, and the Taiwan Environmental Protection Administration. The authors and quoted experts report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
published Oct. 27, 2021, in JAMA Network Open.
In this multicenter cohort study, led by epidemiologist Shu-Li Wang, PhD, of the National Institute of Environmental Health Sciences, in Taiwan, each twofold increase in prenatal arsenic level correlated with a 2.4-fold higher rate of atopic dermatitis in 4-year-olds.
Atopic diseases have been on the rise. Eczema (atopic dermatitis) is the first stage of the so-called atopic march, followed by food allergies, allergic rhinitis, and asthma later in childhood. Previous research has linked heavy metal exposure to allergic diseases in adults. In another study by Dr. Wang and colleagues that was published in 2021, prenatal and early-life arsenic exposure was found to correlate with higher rates of allergic rhinitis and asthma in children. In that study, the participants were followed every 2-3 years through the age of 14 as part of the Taiwan Maternal and Infant Cohort Study.
The new study included 370 mother and child pairs who were enrolled in that birth cohort study between October 2012 and May 2015. During their third trimester of pregnancy, women completed questionnaires about their lifestyle, diet, and living environment. In addition, their height, weight, and blood pressure were recorded, and urine samples were taken. In follow-up interviews 3-4 years later, the mothers were asked whether their child had ever been diagnosed with atopic dermatitis.
The researchers used an inductively coupled plasma mass spectrometer to analyze the participants’ urine samples. They assessed for exposures in utero to eight metals: arsenic, cadmium, lead, cobalt, copper, nickel, thallium, and zinc.
Each unit increase of an index that estimates the combined exposure to these metals during pregnancy was associated with 63% higher odds of atopic dermatitis in the children by age 4. The researchers adjusted for parental allergies (yes or no), mother’s educational level (<12 years, 13-16 years, or >16 years), geographic area (central or eastern Taiwan), exposure to tobacco smoke during pregnancy, and the child’s gender. Arsenic (40.1%) and cadmium (20.5%) accounted for most of the metal coexposure index.
A wealth of previous research links arsenic exposure during adulthood to skin disease and immune dysfunction. Early-life arsenic exposure has been linked with elevated risk for various adult disorders, including cancer, diabetes, and heart disease, years later. In light of such research, “the findings in this paper are not surprising,” J. Christopher States, PhD, director of the Center for Integrative Environmental Health Science at the University of Louisville (Ky.), told this news organization. “Low-level arsenic exposure does not cause disease immediately, but it does appear to have long-lasting effects, making individuals susceptible to ‘second hits’ with another environmental agent.”
Research into the molecular mechanisms for these links has shown that arsenic and cadmium exposure can promote allergic phenotypes in immune cells. “We think the toxic metals activate the alarmin pathway, thus inducing innate lymphoid cells, then activating T-helper 2 cells, which drive immunoglobulin E production and breakdown of the epithelium and promotion of allergies,” said Kari Nadeau, MD, PhD, director of the Sean N. Parker Center for Allergy and Asthma Research at Stanford University. Dr. Nadeau led that study, published in 2017 in PLOS One, along with epidemiologist Margaret Karagas, PhD, of Geisel School of Medicine at Dartmouth, Hanover, N.H.
As for what pregnant women can do to minimize their exposure to heavy metals, “that is a difficult problem and primarily a function of where one lives,” said Dr. States.
Drinking water and food are major sources of arsenic exposure. Groundwater is naturally contaminated with arsenic deposits that seep in from bedrock, said Dr. States. The U.S. Environmental Protection Agency regulates arsenic levels in public drinking water that is supplied to more than a few thousand people. However, small water supplies and private wells are unregulated, he said, and having these water sources tested for arsenic or fitted with systems to reduce arsenic can be very expensive.
Among foods, rice can have high concentrations of arsenic, Dr. Karagas told this news organization. To minimize arsenic exposure through the diet, women can limit rice-based foods, according to a web-based tool developed by her and coworkers.
In addition, tobacco smoke is a major source of cadmium exposure and a moderate source of arsenic exposure, Dr. States noted. Women can reduce their exposure to these metals by avoiding tobacco and secondhand smoke.
The study was supported by grants from the National Health Research Institutes, Chung Shan Medical University Hospital, Taiwan Ministry of Science and Technology, and the Taiwan Environmental Protection Administration. The authors and quoted experts report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Improving Unadjusted and Adjusted Mortality With an Early Warning Sepsis System in the Emergency Department and Inpatient Wards
In 1997, Elizabeth McGlynn wrote, “Measuring quality is no simple task.”1 We are reminded of this seminal Health Affairs article at a very pertinent point—as health care practice progresses, measuring the impact of performance improvement initiatives on clinical care delivery remains integral to monitoring overall effectiveness of quality. Mortality outcomes are a major focus of quality.
Inpatient mortality within the Veterans Health Administration (VHA) was measured as actual number of deaths (unadjusted mortality), and adjusted mortality was calculated using the standardized mortality ratio (SMR). SMR included actual number of deaths during hospitalization or within 1 day of hospital discharge divided by predicted number of deaths using a risk-adjusted formula and was calculated separately for acute level of care (LOC) and the intensive care unit (ICU). Using risk-adjusted SMR, if an observed/expected ratio was > 1.0, there were more inpatient deaths than expected; if < 1.0, fewer inpatient deaths occurred than predicted; and if 1.0, observed number of inpatient deaths was equivalent to expected number of deaths.2
Mortality reduction is a complex area of performance improvement. Health care facilities often focus their efforts on the biggest mortality contributors. According to Dantes and Epstein, sepsis results in about 265,000 deaths annually in the United States.3 Reinhart and colleagues demonstrated that sepsis is a worldwide issue resulting in approximately 30 million cases and 6 million deaths annually.4 Furthermore, Kumar and colleagues have noted that when sepsis progresses to septic shock, survival decreases by almost 8% for each hour delay in sepsis identification and treatment.5
Improvements in sepsis management have been multifaceted. The Surviving Sepsis Campaign guidelines created sepsis treatment bundles to guide early diagnosis/treatment of sepsis.6 In addition to awareness and sepsis care bundles, a plethora of informatics solutions within electronic health record (EHR) systems have demonstrated improved sepsis care.7-16 Various approaches to early diagnosis and management of sepsis have been collectively referred to as an early warning sepsis system (EWSS).
An EWSS typically contains automated decision support tools that are integrated in the EHR and meant to assist health care professionals with clinical workflow decision-making. Automated decision support tools within the EHR have a variety of functions, such as clinical care reminders and alerts.17
Sepsis screening tools function as a form of automated decision support and may be incorporated into the EHR to support the EWSS. Although sepsis screening tools vary, they frequently include a combination of data involving vital signs, laboratory values and/or physical examination findings, such as mental status evaluation.The Modified Early Warning Signs (MEWS) + Sepsis Recognition Score (SRS) is one example of a sepsis screening tool.7,16
At Malcom Randall Veterans Affairs Medical Center (MRVAMC) in Gainesville, Florida, we identified a quality improvement project opportunity to improve sepsis care in the emergency department (ED) and inpatient wards using the VHA EHR system, the Computerized Patient Record System (CPRS), which is supported by the Veterans Information Systems and Technology Architecture (VistA).18 A VistA/CPRS EWSS was developed using Lean Six Sigma DMAIC (define, measure, analyze, improve, and control) methodology.19 During the improve stage, informatics solutions were applied and included a combination of EHR interventions, such as template design, an order set, and clinical reminders. Clinical reminders have a wide variety of use, such as reminders for clinical tasks and as automated decision support within clinical workflows using Boolean logic.
To the best of our knowledge, there has been no published application of an EWSS within VistA/CPRS. In this study, we outline the strategic development of an EWSS in VistA/CPRS that assisted clinical staff with identification and treatment of sepsis; improved documentation of sepsis when present; and associated with improvement in unadjusted and adjusted inpatient mortality.
Methods
According to policy activities that constitute research at MRVAMC, no institutional review board approval was required as this work met criteria for operational improvement activities exempt from ethics review.
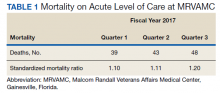
The North Florida/South Georgia Veterans Health System (NF/SGVHS) includes MRVAMC, a large academic hospital with rotating residents/fellows and multiple specialty care services. MRVAMC comprised 144 beds on the medicine/surgery wards; 48 beds in the psychiatry unit; 18 intermediate LOC beds; and 27 ICU beds. The MRVAMC SMR was identified as an improvement opportunity during fiscal year (FY) 2017 (Table 1). Its adjusted mortality for acute LOC demonstrated an observed/expected ratio of > 1.0 suggesting more inpatient deaths were observed than expected. The number of deaths (unadjusted mortality) on acute LOC at MRVAMC was noted to be rising during the first 3 quarters of FY 2017. A deeper examination of data by Pyramid Analytics (www.pyramidanalytics.com) discovered that sepsis was the primary driver for inpatient mortality on acute LOC at MRVAMC. Our goal was to reduce inpatient sepsis-related mortality via development of an EWSS that leveraged VistA/CPRS to improve early identification and treatment of sepsis in the ED and inpatient wards.
Emergency Department
Given the importance of recognizing sepsis early, the sepsis team focused on improvement opportunities at the initial point of patient contact: ED triage. The goal was to incorporate automated VistA/CPRS decision support to assist clinicians with identifying sepsis in triage using MEWS, which was chosen to optimize immediate hospital-wide buy-in. Clinical staff were already familiar with MEWS, which was in use on the inpatient wards.
Flow through the ED and availability of resources differed from the wards. Hence, modification to MEWS on the wards was necessary to fit clinical workflow in the ED. Temperature, heart rate (HR), respiratory rate (RR), systolic blood pressure (SBP), mental status, and white blood cell count (WBC) factored into a MEWS + SRS score on the wards (Table 2). For the ED, MEWS included temperature, HR, RR and SBP, but excluded mental status and WBC. Mental status assessment was excluded due to technical infeasibility (while vital signs could be automatically calculated in real time for a MEWS score, that was not possible for mental status changes). WBC was excluded from the ED as laboratory test results would not be available in triage.
MEWS + SRS scores were calculated in VistA by using clinical reminders. Clinical reminder logic included a series of conditional statements based on various combinations of MEWS + SRS clinical data entered in the EHR. When ED triage vital signs data were entered in CPRS, clinical data were stored and processed according to clinical reminder logic in VistA and displayed to the user in CPRS. While MEWS of ≥ 5 triggered a sepsis alert on the wards, the ≥ 4 threshold was used in the ED given mental status and WBC were excluded from calculations in triage (eAppendix 1 available at doi:10.12788/fp.0194).
Once a sepsis alert was triggered in triage for MEWS ≥ 4, ED nursing staff prioritized bed location and expedited staffing with an ED attending physician for early assessment. The ED attending then performed an assessment to confirm whether sepsis was present and direct early treatment. Although every patient who triggered a sepsis alert in triage did not meet clinical findings of sepsis, patients with MEWS ≥ 4 were frequently ill and required timely intervention.
If an ED attending physician agreed with a sepsis diagnosis, the physician had access to a sepsis workup and treatment order set in CPRS (eAppendix 2 available at doi:10.12788/fp.0194). The sepsis order set incorporated recommendations from the Surviving Sepsis Campaign guidelines and included orders for 2 large-bore peripheral IV lines; aggressive fluid resuscitation (30 mL/kg) for patients with clinical findings of hypoperfusion; broad-spectrum antibiotics; and frequent ordering of laboratory tests and imaging during initial sepsis workup.6 Vancomycin and cefepime were selected as routine broad-spectrum antibiotics in the order set when sepsis was suspected based on local antimicrobial stewardship and safety-efficacy profiles. For example, Luther and colleagues demonstrated that cefepime has lower rates of acute kidney injury when combined with vancomycin vs vancomycin + piperacillin-tazobactam.20 If a β-lactam antibiotic could not be used due to a patient’s drug allergy history, aztreonam was available as an alternative option.
The design of the order set also functioned as a communication interface with clinical pharmacists. Given the large volume of antibiotics ordered in the ED, it was difficult for pharmacists to prioritize antibiotic order verification. While stat orders convey high priority, they often lack specificity. When antibiotic orders were selected from the sepsis order set, comments were already included that stated: “STAT. First dose for sepsis protocol” (eAppendix 3 available at doi:10.12788/fp.0194). This standardized communication conveyed a sense of urgency and a collective understanding that patients with suspected sepsis required timely order verification and administration of antibiotics.
Hospital Ward
Mental status and WBC were included on the wards to monitor for possible signs of sepsis, using MEWS + SRS, which was routinely monitored by nursing every 4 to 8 hours. When MEWS + SRS was ≥ 5 points, ward nursing staff called a sepsis alert.7,16 Early response team (ERT) members received telephone notifications of the alert. ERT staff proceeded with immediate evaluation and treatment at the bedside along with determination for most appropriate LOC. The ERT members included an ICU physician and nurse; respiratory therapist; and nursing supervisor/bed flow coordinator. During bedside evaluation, if the ERT or primary team agreed with a sepsis diagnosis, the ERT or primary team used the sepsis order set to ensure standardized procedures. Stat orders generated through the sepsis order set pathway conveyed a sense of urgency and need for immediate order verification and administration of antibiotics.
In addition to clinical care process improvement, accurate documentation also was emphasized in the EWSS. When a sepsis alert was called, a clinician from the primary team was expected to complete a standardized progress note, which communicated clinical findings, a treatment plan, and captured severity of illness (eAppendix 4 available at doi:10.12788/fp.0194). It included sections for subjective, objective, assessment, and plan. In addition, data objects were created for vital signs and common laboratory findings that retrieved important clinical data from VistA and inserted it into the CPRS note.21
Nursing staff on the wards were expected to communicate results with the primary team for clinical decision making when a patient had a MEWS + SRS of 3 to 4. A sepsis alert may have been called at the discretion of clinical team members but was not required if the score was < 5. Additionally, vital signs were expected to be checked by the nursing staff on the wards at least every 4 hours for closer monitoring.
Sepsis Review Meetings
Weekly meetings were scheduled to review sepsis cases to assess diagnosis, treatment, and documentation entered in the patient record. The team conducting sepsis reviews comprised the chief of staff, chief of quality management, director of patient safety, physician utilization management advisor, chief resident in quality and patient safety (CRQS), and inpatient pharmacy supervisor. In addition, ad hoc physicians and nurses from different specialty areas, such as infectious diseases, hospitalist section, ICU, and the ED participated on request for subject matter expertise when needed. At the conclusion of weekly sepsis meetings, sepsis team members provided feedback to the clinical staff for continuous improvement purposes.
Results
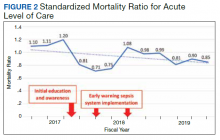
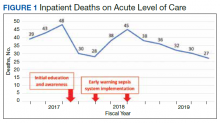
Before implementation of an EWSS at NF/SGVHS, a plan was devised to increase awareness and educate staff on sepsis-related mortality in late FY 2017. Awareness and education about sepsis-related mortality was organized at physician, nursing, and pharmacy leadership clinical staff meetings. Posters about early warning signs of sepsis also were displayed on the nursing units for educational purposes and to convey the importance of early recognition/treatment of sepsis. In addition, the CRQS was the quality leader for house staff and led sepsis campaign change efforts for residents/fellows. An immediate improvement in unadjusted mortality at MRVAMC was noted with initial sepsis awareness and education. From FY 2017, quarter 3 to FY 2018, quarter 1, the number of acute LOC inpatient deaths decreased from 48 to 28, a 42% reduction in unadjusted mortality at MRVAMC (Figure 1). Additionally, the acute LOC SMR improved from 1.20 during FY 2017, quarter 3 down to as low as 0.71 during FY 2018, quarter 1 (Figure 2).
The number of MRVAMC inpatient deaths increased from 28 in FY 2018, quarter 1 to 45 in FY 2018, quarter 3. While acute LOC showed improvement in unadjusted mortality after sepsis education/awareness, it was felt continuous improvement could not be sustained with education alone. An EWSS was designed and implemented within the EHR system in FY 2018. Following implementation of EWSS and reeducating staff on early recognition and treatment of sepsis, acute LOC inpatient deaths decreased from 45 in FY 2018, quarter 3 through FY 2019 where unadjusted mortality was as low as 27 during FY 2019, quarter 4. The MRVAMC acute LOC SMR was consistently < 1.0 from FY 2018, quarter 4 through FY 2019, quarter 4.
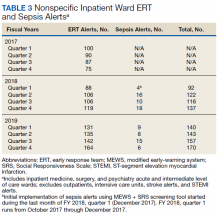
In addition to the observed decrease in acute LOC inpatient deaths and improved SMR, the number of ERT alerts and sepsis alerts on the inpatient wards were monitored from FY 2017 through FY 2019. ERT alerts listed in Table 3 were nonspecific and initiated by nursing staff on the wards where a patient’s clinical status was identified as worsening while sepsis alerts were specific ERT alerts called by the ward nursing staff due to concerns for sepsis. The inpatient wards included inpatient medicine, surgery, and psychiatry acute care and the intermediate level of care unit while outpatient clinical areas of treatment, intensive care units, stroke alerts, and STEMI alerts were excluded.
From FY 2017 to FY 2018, quarter 1, the number of nonspecific ERT alerts varied between 75 to 100. Sepsis alerts were not available until December 2017 while the EWSS was in development. Afterward, nonspecific ERT alerts and sepsis alerts were monitored each quarter. Sepsis alerts ranged from 4 to 18. Nonspecific ERT alerts + sepsis alerts continued to increase from FY 2018, quarter 3 through FY 2019, quarter 4.
Discussion
Implementation of the EWSS was associated with improved unadjusted mortality and adjusted mortality for acute LOC at MRVAMC. Although variation exists with application of EWSS at other medical centers, there was similarity with improved sepsis outcomes reported at other health care systems after EWSS implementation.7-16
Improved unadjusted mortality and adjusted mortality for acute LOC at MRVAMC was likely due to multiple contributing factors. First, during design and implementation of the EWSS, project work was interdisciplinary with input from physicians, nurses, and pharmacists from multiple specialties (ie, ED, ICU, and the medicine service); quality management and data analysis specialists; and clinical informatics. Second, facility commitment to improving early recognition and treatment of sepsis from leadership level down to front-line staff was evident. Weekly sepsis meetings with the NF/SGVHS chief of staff helped to sustain EWSS efforts and to identify additional improvement opportunities. Third, integrated informatics solutions within the EHR helped identify early sepsis and minimized human error as well as assisted with coordination of sepsis care across services. Fourth, the focus was on both early identification and treatment of sepsis in the ED and hospital wards. Although it cannot be deduced whether there was causation between reduced inpatient mortality and an increased number of nonspecific ERT alerts+ sepsis alerts on the inpatient wards after EWSS implementation, inpatient deaths decreased and SMR improved. Finally, the EWSS emphasized both the importance of evidence-based clinical care of sepsis and standardized documentation to appropriately capture clinical severity of illness.
Limitations
This program has limitations. The EWSS was studied at a single VHA facility. Veteran demographics and local epidemiology may limit conclusion of outcomes to an individual VHA facility located in a specific geographical region. Additional research is necessary to demonstrate reproducibility and determine whether applicable to other VHA facilities and community care settings.
SMR is a risk-adjusted formula developed by the VHA Inpatient Evaluation Center, which included numerous factors such as diagnosis, comorbid conditions, age, marital status, procedures, source of admission, specific laboratory values, medical or surgical diagnosis-related group, ICU stays, immunosuppressive status, and a COVID-19 positive indicator (added after this study). Further research is needed to evaluate sepsis-related outcomes using the EWSS during the COVID-19 pandemic.
EWSS in the literature have demonstrated various approaches to early identification and treatment of sepsis and have used different sepsis screening tools.22 Evidence suggests that the MEWS + SRS sepsis screening tool may result in false-positive screenings.23-27 Additional research into the specificity of this sepsis screening tool is needed. Ward nursing staff were encouraged to initiate automatic sepsis alerts when MEWS + SRS was ≥ 5; however, this still depended on human factors. Because sepsis alerts are software-specific and others were incompatible with the VHA EHR, it was necessary to design our own EWSS.
Despite improvement with MRVAMC acute LOC unadjusted and adjusted mortality with our EWSS, we did not identify any actual improvement in earlier antibiotic administration times once sepsis was recognized. While accurate documentation regarding degree of sepsis improved, a MRVAMC clinical documentation improvement program was expanded in FY 2018. Therefore, it is difficult to demonstrate causation related to improved sepsis documentation with template changes alone. While sepsis alerts on the inpatient wards were variable since EWSS implementation, nonspecific ERT alerts increased. It is unclear whether some sepsis alerts were called as nonspecific ERT alerts, making it impossible to know the true number of sepsis alerts.
MRVAMC experienced an increase in nurse turnover during FY 2018 and as a teaching hospital had frequent rotating residents and fellows new to processes/protocols. These factors may have contributed to variations in unadjusted mortality. Also the decrease in inpatient mortality and improvement in SMR on acute LOC could have been the result of factors other than the EWSS and the effect of education alone may have been at least as good as that of the EWSS intervention.
Conclusions
Education along with the possible implementation of an EWSS at NF/SGVHS was associated with a decrease in the number of inpatient deaths on MRVAMC’s acute LOC wards from as high as 48 in FY 2017, quarter 3 to as low as 27 in FY 2019, quarter 4 resulting in as large of an improvement as a 44% reduction in unadjusted mortality from FY 2017 to FY 2019. In addition, MRVAMC’s acute LOC SMR improved from > 1.0 to < 1.0, demonstrating fewer inpatient mortalities than predicted from FY 2017 to FY 2019.
This multifaceted interventional strategy may be effectively applied at other VHA health care facilities that use the same EHR system. Next steps may include determining the specificity of MEWS + SRS as a sepsis screening tool; studying outcomes of MRVAMC’s EWSS during the COVID-19 era; and conducting a multicentered study on this EWSS across multiple VHA facilities.
1. McGlynn EA. Six challenges in measuring the quality of health care. Health Aff (Millwood). 1997;16(3):7-21. doi:10.1377/hlthaff.16.3.7
2. US Department of Veterans Affairs, Veterans Health Administration. Strategic Analytics for Improvement and Learning (SAIL) value model measure definitions. Updated May 15, 2019. Accessed October 11, 2021. https://www.va.gov/QUALITYOFCARE/measure-up/SAIL_definitions.asp
3. Dantes RB, Epstein L. Combatting sepsis: a public health perspective. Clin Infect Dis. 2018;67(8):1300-1302. doi:10.1093/cid/ciy342
4. Reinhart K, Daniels R, Kissoon N, Machado FR, Schachter RD, Finfer S. Recognizing sepsis as a global health priority - a WHO resolution. N Engl J Med. 2017;377(5):414-417. doi:10.1056/NEJMp1707170
5. Kumar A, Roberts D, Wood KE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34(6):1589-1596. doi:10.1097/01.CCM.0000217961.75225.E9
6. Rhodes A, Evans LE, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45(3):486-552. doi:10.1097/CCM.0000000000002255
7. Guirgis FW, Jones L, Esma R, et al. Managing sepsis: electronic recognition, rapid response teams, and standardized care save lives. J Crit Care. 2017;40:296-302. doi:10.1016/j.jcrc.2017.04.005
8. Whippy A, Skeath M, Crawford B, et al. Kaiser Permanente’s performance improvement system, part 3: multisite improvements in care for patients with sepsis. Jt Comm J Qual Patient Saf. 2011;37(11):483-493. doi:10.1016/s1553-7250(11)37061-4
9. Harrison AM, Thongprayoon C, Kashyap R, et al. Developing the surveillance algorithm for detection of failure to recognize and treat severe sepsis. Mayo Clin Proc. 2015;90(2):166-175. doi:10.1016/j.mayocp.2014.11.014
10. Rothman M, Levy M, Dellinger RP, et al. Sepsis as 2 problems: identifying sepsis at admission and predicting onset in the hospital using an electronic medical record-based acuity score. J Crit Care. 2017;38:237-244. doi:10.1016/j.jcrc.2016.11.037
11. Back JS, Jin Y, Jin T, Lee SM. Development and validation of an automated sepsis risk assessment system. Res Nurs Health. 2016;39(5):317-327. doi:10.1002/nur.21734
12. Khurana HS, Groves RH Jr, Simons MP, et al. Real-time automated sampling of electronic medical records predicts hospital mortality. Am J Med. 2016;129(7):688-698.e2. doi:10.1016/j.amjmed.2016.02.037
13. Umscheid CA, Betesh J, VanZandbergen C, et al. Development, implementation, and impact of an automated early warning and response system for sepsis. J Hosp Med. 2015;10(1):26-31. doi:10.1002/jhm.2259
14. Vogel L. EMR alert cuts sepsis deaths. CMAJ. 2014;186(2):E80. doi:10.1503/cmaj.109-4686
15. Jones SL, Ashton CM, Kiehne L, et al. Reductions in sepsis mortality and costs after design and implementation of a nurse-based early recognition and response program. Jt Comm J Qual Patient Saf. 2015;41(11):483-491. doi:10.1016/s1553-7250(15)41063-3
16. Croft CA, Moore FA, Efron PA, et al. Computer versus paper system for recognition and management of sepsis in surgical intensive care. J Trauma Acute Care Surg. 2014;76(2):311-319. doi:10.1097/TA.0000000000000121
17. Tcheng JE, Bakken S, Bates DW, et al, eds. Optimizing Strategies for Clinical Decision Support: Summary of a Meeting Series. National Academy of Medicine; 2017. Accessed October 11, 2021. https://nam.edu/wp-content/uploads/2017/11/Optimizing-Strategies-for-Clinical-Decision-Support.pdf
18. US Department of Veterans Affairs. History of IT at VA. Updated January 1, 2020. Accessed October 11, 2021. https://www.oit.va.gov/about/history.cfm
19. GoLeanSixSigma. DMAIC: The 5 Phases of Lean Six Sigma. Published 2012. Accessed October 11, 2021. https://goleansixsigma.com/wp-content/uploads/2012/02/DMAIC-The-5-Phases-of-Lean-Six-Sigma-www.GoLeanSixSigma.com_.pdf
20. Luther MK, Timbrook TT, Caffrey AR, Dosa D, Lodise TP, LaPlante KL. Vancomycin plus piperacillin-tazobactam and acute kidney injury in adults: a systematic review and meta-analysis. Crit Care Med. 2018;46(1):12-20. doi:10.1097/CCM.0000000000002769
21. International Business Machines Corp. Overview of data objects. Accessed October 11, 2021. https://www.ibm.com/support/knowledgecenter/en/SSLTBW_2.3.0/com.ibm.zos.v2r3.cbclx01/data_objects.htm
22. Churpek MM, Snyder A, Han X, et al. Quick sepsis-related organ failure assessment, systemic inflammatory response syndrome, and early warning scores for detecting clinical deterioration in infected patients outside the intensive care unit. Am J Respir Crit Care Med. 2017;195(7):906-911. doi:10.1164/rccm.201604-0854OC
23. Ghanem-Zoubi NO, Vardi M, Laor A, Weber G, Bitterman H. Assessment of disease-severity scoring systems for patients with sepsis in general internal medicine departments. Crit Care. 2011;15(2):R95. doi:10.1186/cc10102
24. Hamilton F, Arnold D, Baird A, Albur M, Whiting P. Early Warning scores do not accurately predict mortality in sepsis: a meta-analysis and systematic review of the literature. J Infect. 2018;76(3):241-248. doi:10.1016/j.jinf.2018.01.002
25. Martino IF, Figgiaconi V, Seminari E, Muzzi A, Corbella M, Perlini S. The role of qSOFA compared to other prognostic scores in septic patients upon admission to the emergency department. Eur J Intern Med. 2018;53:e11-e13. doi:10.1016/j.ejim.2018.05.022
26. Nannan Panday RS, Minderhoud TC, Alam N, Nanayakkara PWB. Prognostic value of early warning scores in the emergency department (ED) and acute medical unit (AMU): A narrative review. Eur J Intern Med. 2017;45:20-31. doi:10.1016/j.ejim.2017.09.027
27. Jayasundera R, Neilly M, Smith TO, Myint PK. Are early warning scores useful predictors for mortality and morbidity in hospitalised acutely unwell older patients? A systematic review. J Clin Med. 2018;7(10):309. Published 2018 Sep 28. doi:10.3390/jcm7100309
In 1997, Elizabeth McGlynn wrote, “Measuring quality is no simple task.”1 We are reminded of this seminal Health Affairs article at a very pertinent point—as health care practice progresses, measuring the impact of performance improvement initiatives on clinical care delivery remains integral to monitoring overall effectiveness of quality. Mortality outcomes are a major focus of quality.
Inpatient mortality within the Veterans Health Administration (VHA) was measured as actual number of deaths (unadjusted mortality), and adjusted mortality was calculated using the standardized mortality ratio (SMR). SMR included actual number of deaths during hospitalization or within 1 day of hospital discharge divided by predicted number of deaths using a risk-adjusted formula and was calculated separately for acute level of care (LOC) and the intensive care unit (ICU). Using risk-adjusted SMR, if an observed/expected ratio was > 1.0, there were more inpatient deaths than expected; if < 1.0, fewer inpatient deaths occurred than predicted; and if 1.0, observed number of inpatient deaths was equivalent to expected number of deaths.2
Mortality reduction is a complex area of performance improvement. Health care facilities often focus their efforts on the biggest mortality contributors. According to Dantes and Epstein, sepsis results in about 265,000 deaths annually in the United States.3 Reinhart and colleagues demonstrated that sepsis is a worldwide issue resulting in approximately 30 million cases and 6 million deaths annually.4 Furthermore, Kumar and colleagues have noted that when sepsis progresses to septic shock, survival decreases by almost 8% for each hour delay in sepsis identification and treatment.5
Improvements in sepsis management have been multifaceted. The Surviving Sepsis Campaign guidelines created sepsis treatment bundles to guide early diagnosis/treatment of sepsis.6 In addition to awareness and sepsis care bundles, a plethora of informatics solutions within electronic health record (EHR) systems have demonstrated improved sepsis care.7-16 Various approaches to early diagnosis and management of sepsis have been collectively referred to as an early warning sepsis system (EWSS).
An EWSS typically contains automated decision support tools that are integrated in the EHR and meant to assist health care professionals with clinical workflow decision-making. Automated decision support tools within the EHR have a variety of functions, such as clinical care reminders and alerts.17
Sepsis screening tools function as a form of automated decision support and may be incorporated into the EHR to support the EWSS. Although sepsis screening tools vary, they frequently include a combination of data involving vital signs, laboratory values and/or physical examination findings, such as mental status evaluation.The Modified Early Warning Signs (MEWS) + Sepsis Recognition Score (SRS) is one example of a sepsis screening tool.7,16
At Malcom Randall Veterans Affairs Medical Center (MRVAMC) in Gainesville, Florida, we identified a quality improvement project opportunity to improve sepsis care in the emergency department (ED) and inpatient wards using the VHA EHR system, the Computerized Patient Record System (CPRS), which is supported by the Veterans Information Systems and Technology Architecture (VistA).18 A VistA/CPRS EWSS was developed using Lean Six Sigma DMAIC (define, measure, analyze, improve, and control) methodology.19 During the improve stage, informatics solutions were applied and included a combination of EHR interventions, such as template design, an order set, and clinical reminders. Clinical reminders have a wide variety of use, such as reminders for clinical tasks and as automated decision support within clinical workflows using Boolean logic.
To the best of our knowledge, there has been no published application of an EWSS within VistA/CPRS. In this study, we outline the strategic development of an EWSS in VistA/CPRS that assisted clinical staff with identification and treatment of sepsis; improved documentation of sepsis when present; and associated with improvement in unadjusted and adjusted inpatient mortality.
Methods
According to policy activities that constitute research at MRVAMC, no institutional review board approval was required as this work met criteria for operational improvement activities exempt from ethics review.
The North Florida/South Georgia Veterans Health System (NF/SGVHS) includes MRVAMC, a large academic hospital with rotating residents/fellows and multiple specialty care services. MRVAMC comprised 144 beds on the medicine/surgery wards; 48 beds in the psychiatry unit; 18 intermediate LOC beds; and 27 ICU beds. The MRVAMC SMR was identified as an improvement opportunity during fiscal year (FY) 2017 (Table 1). Its adjusted mortality for acute LOC demonstrated an observed/expected ratio of > 1.0 suggesting more inpatient deaths were observed than expected. The number of deaths (unadjusted mortality) on acute LOC at MRVAMC was noted to be rising during the first 3 quarters of FY 2017. A deeper examination of data by Pyramid Analytics (www.pyramidanalytics.com) discovered that sepsis was the primary driver for inpatient mortality on acute LOC at MRVAMC. Our goal was to reduce inpatient sepsis-related mortality via development of an EWSS that leveraged VistA/CPRS to improve early identification and treatment of sepsis in the ED and inpatient wards.
Emergency Department
Given the importance of recognizing sepsis early, the sepsis team focused on improvement opportunities at the initial point of patient contact: ED triage. The goal was to incorporate automated VistA/CPRS decision support to assist clinicians with identifying sepsis in triage using MEWS, which was chosen to optimize immediate hospital-wide buy-in. Clinical staff were already familiar with MEWS, which was in use on the inpatient wards.
Flow through the ED and availability of resources differed from the wards. Hence, modification to MEWS on the wards was necessary to fit clinical workflow in the ED. Temperature, heart rate (HR), respiratory rate (RR), systolic blood pressure (SBP), mental status, and white blood cell count (WBC) factored into a MEWS + SRS score on the wards (Table 2). For the ED, MEWS included temperature, HR, RR and SBP, but excluded mental status and WBC. Mental status assessment was excluded due to technical infeasibility (while vital signs could be automatically calculated in real time for a MEWS score, that was not possible for mental status changes). WBC was excluded from the ED as laboratory test results would not be available in triage.
MEWS + SRS scores were calculated in VistA by using clinical reminders. Clinical reminder logic included a series of conditional statements based on various combinations of MEWS + SRS clinical data entered in the EHR. When ED triage vital signs data were entered in CPRS, clinical data were stored and processed according to clinical reminder logic in VistA and displayed to the user in CPRS. While MEWS of ≥ 5 triggered a sepsis alert on the wards, the ≥ 4 threshold was used in the ED given mental status and WBC were excluded from calculations in triage (eAppendix 1 available at doi:10.12788/fp.0194).
Once a sepsis alert was triggered in triage for MEWS ≥ 4, ED nursing staff prioritized bed location and expedited staffing with an ED attending physician for early assessment. The ED attending then performed an assessment to confirm whether sepsis was present and direct early treatment. Although every patient who triggered a sepsis alert in triage did not meet clinical findings of sepsis, patients with MEWS ≥ 4 were frequently ill and required timely intervention.
If an ED attending physician agreed with a sepsis diagnosis, the physician had access to a sepsis workup and treatment order set in CPRS (eAppendix 2 available at doi:10.12788/fp.0194). The sepsis order set incorporated recommendations from the Surviving Sepsis Campaign guidelines and included orders for 2 large-bore peripheral IV lines; aggressive fluid resuscitation (30 mL/kg) for patients with clinical findings of hypoperfusion; broad-spectrum antibiotics; and frequent ordering of laboratory tests and imaging during initial sepsis workup.6 Vancomycin and cefepime were selected as routine broad-spectrum antibiotics in the order set when sepsis was suspected based on local antimicrobial stewardship and safety-efficacy profiles. For example, Luther and colleagues demonstrated that cefepime has lower rates of acute kidney injury when combined with vancomycin vs vancomycin + piperacillin-tazobactam.20 If a β-lactam antibiotic could not be used due to a patient’s drug allergy history, aztreonam was available as an alternative option.
The design of the order set also functioned as a communication interface with clinical pharmacists. Given the large volume of antibiotics ordered in the ED, it was difficult for pharmacists to prioritize antibiotic order verification. While stat orders convey high priority, they often lack specificity. When antibiotic orders were selected from the sepsis order set, comments were already included that stated: “STAT. First dose for sepsis protocol” (eAppendix 3 available at doi:10.12788/fp.0194). This standardized communication conveyed a sense of urgency and a collective understanding that patients with suspected sepsis required timely order verification and administration of antibiotics.
Hospital Ward
Mental status and WBC were included on the wards to monitor for possible signs of sepsis, using MEWS + SRS, which was routinely monitored by nursing every 4 to 8 hours. When MEWS + SRS was ≥ 5 points, ward nursing staff called a sepsis alert.7,16 Early response team (ERT) members received telephone notifications of the alert. ERT staff proceeded with immediate evaluation and treatment at the bedside along with determination for most appropriate LOC. The ERT members included an ICU physician and nurse; respiratory therapist; and nursing supervisor/bed flow coordinator. During bedside evaluation, if the ERT or primary team agreed with a sepsis diagnosis, the ERT or primary team used the sepsis order set to ensure standardized procedures. Stat orders generated through the sepsis order set pathway conveyed a sense of urgency and need for immediate order verification and administration of antibiotics.
In addition to clinical care process improvement, accurate documentation also was emphasized in the EWSS. When a sepsis alert was called, a clinician from the primary team was expected to complete a standardized progress note, which communicated clinical findings, a treatment plan, and captured severity of illness (eAppendix 4 available at doi:10.12788/fp.0194). It included sections for subjective, objective, assessment, and plan. In addition, data objects were created for vital signs and common laboratory findings that retrieved important clinical data from VistA and inserted it into the CPRS note.21
Nursing staff on the wards were expected to communicate results with the primary team for clinical decision making when a patient had a MEWS + SRS of 3 to 4. A sepsis alert may have been called at the discretion of clinical team members but was not required if the score was < 5. Additionally, vital signs were expected to be checked by the nursing staff on the wards at least every 4 hours for closer monitoring.
Sepsis Review Meetings
Weekly meetings were scheduled to review sepsis cases to assess diagnosis, treatment, and documentation entered in the patient record. The team conducting sepsis reviews comprised the chief of staff, chief of quality management, director of patient safety, physician utilization management advisor, chief resident in quality and patient safety (CRQS), and inpatient pharmacy supervisor. In addition, ad hoc physicians and nurses from different specialty areas, such as infectious diseases, hospitalist section, ICU, and the ED participated on request for subject matter expertise when needed. At the conclusion of weekly sepsis meetings, sepsis team members provided feedback to the clinical staff for continuous improvement purposes.
Results
Before implementation of an EWSS at NF/SGVHS, a plan was devised to increase awareness and educate staff on sepsis-related mortality in late FY 2017. Awareness and education about sepsis-related mortality was organized at physician, nursing, and pharmacy leadership clinical staff meetings. Posters about early warning signs of sepsis also were displayed on the nursing units for educational purposes and to convey the importance of early recognition/treatment of sepsis. In addition, the CRQS was the quality leader for house staff and led sepsis campaign change efforts for residents/fellows. An immediate improvement in unadjusted mortality at MRVAMC was noted with initial sepsis awareness and education. From FY 2017, quarter 3 to FY 2018, quarter 1, the number of acute LOC inpatient deaths decreased from 48 to 28, a 42% reduction in unadjusted mortality at MRVAMC (Figure 1). Additionally, the acute LOC SMR improved from 1.20 during FY 2017, quarter 3 down to as low as 0.71 during FY 2018, quarter 1 (Figure 2).
The number of MRVAMC inpatient deaths increased from 28 in FY 2018, quarter 1 to 45 in FY 2018, quarter 3. While acute LOC showed improvement in unadjusted mortality after sepsis education/awareness, it was felt continuous improvement could not be sustained with education alone. An EWSS was designed and implemented within the EHR system in FY 2018. Following implementation of EWSS and reeducating staff on early recognition and treatment of sepsis, acute LOC inpatient deaths decreased from 45 in FY 2018, quarter 3 through FY 2019 where unadjusted mortality was as low as 27 during FY 2019, quarter 4. The MRVAMC acute LOC SMR was consistently < 1.0 from FY 2018, quarter 4 through FY 2019, quarter 4.
In addition to the observed decrease in acute LOC inpatient deaths and improved SMR, the number of ERT alerts and sepsis alerts on the inpatient wards were monitored from FY 2017 through FY 2019. ERT alerts listed in Table 3 were nonspecific and initiated by nursing staff on the wards where a patient’s clinical status was identified as worsening while sepsis alerts were specific ERT alerts called by the ward nursing staff due to concerns for sepsis. The inpatient wards included inpatient medicine, surgery, and psychiatry acute care and the intermediate level of care unit while outpatient clinical areas of treatment, intensive care units, stroke alerts, and STEMI alerts were excluded.
From FY 2017 to FY 2018, quarter 1, the number of nonspecific ERT alerts varied between 75 to 100. Sepsis alerts were not available until December 2017 while the EWSS was in development. Afterward, nonspecific ERT alerts and sepsis alerts were monitored each quarter. Sepsis alerts ranged from 4 to 18. Nonspecific ERT alerts + sepsis alerts continued to increase from FY 2018, quarter 3 through FY 2019, quarter 4.
Discussion
Implementation of the EWSS was associated with improved unadjusted mortality and adjusted mortality for acute LOC at MRVAMC. Although variation exists with application of EWSS at other medical centers, there was similarity with improved sepsis outcomes reported at other health care systems after EWSS implementation.7-16
Improved unadjusted mortality and adjusted mortality for acute LOC at MRVAMC was likely due to multiple contributing factors. First, during design and implementation of the EWSS, project work was interdisciplinary with input from physicians, nurses, and pharmacists from multiple specialties (ie, ED, ICU, and the medicine service); quality management and data analysis specialists; and clinical informatics. Second, facility commitment to improving early recognition and treatment of sepsis from leadership level down to front-line staff was evident. Weekly sepsis meetings with the NF/SGVHS chief of staff helped to sustain EWSS efforts and to identify additional improvement opportunities. Third, integrated informatics solutions within the EHR helped identify early sepsis and minimized human error as well as assisted with coordination of sepsis care across services. Fourth, the focus was on both early identification and treatment of sepsis in the ED and hospital wards. Although it cannot be deduced whether there was causation between reduced inpatient mortality and an increased number of nonspecific ERT alerts+ sepsis alerts on the inpatient wards after EWSS implementation, inpatient deaths decreased and SMR improved. Finally, the EWSS emphasized both the importance of evidence-based clinical care of sepsis and standardized documentation to appropriately capture clinical severity of illness.
Limitations
This program has limitations. The EWSS was studied at a single VHA facility. Veteran demographics and local epidemiology may limit conclusion of outcomes to an individual VHA facility located in a specific geographical region. Additional research is necessary to demonstrate reproducibility and determine whether applicable to other VHA facilities and community care settings.
SMR is a risk-adjusted formula developed by the VHA Inpatient Evaluation Center, which included numerous factors such as diagnosis, comorbid conditions, age, marital status, procedures, source of admission, specific laboratory values, medical or surgical diagnosis-related group, ICU stays, immunosuppressive status, and a COVID-19 positive indicator (added after this study). Further research is needed to evaluate sepsis-related outcomes using the EWSS during the COVID-19 pandemic.
EWSS in the literature have demonstrated various approaches to early identification and treatment of sepsis and have used different sepsis screening tools.22 Evidence suggests that the MEWS + SRS sepsis screening tool may result in false-positive screenings.23-27 Additional research into the specificity of this sepsis screening tool is needed. Ward nursing staff were encouraged to initiate automatic sepsis alerts when MEWS + SRS was ≥ 5; however, this still depended on human factors. Because sepsis alerts are software-specific and others were incompatible with the VHA EHR, it was necessary to design our own EWSS.
Despite improvement with MRVAMC acute LOC unadjusted and adjusted mortality with our EWSS, we did not identify any actual improvement in earlier antibiotic administration times once sepsis was recognized. While accurate documentation regarding degree of sepsis improved, a MRVAMC clinical documentation improvement program was expanded in FY 2018. Therefore, it is difficult to demonstrate causation related to improved sepsis documentation with template changes alone. While sepsis alerts on the inpatient wards were variable since EWSS implementation, nonspecific ERT alerts increased. It is unclear whether some sepsis alerts were called as nonspecific ERT alerts, making it impossible to know the true number of sepsis alerts.
MRVAMC experienced an increase in nurse turnover during FY 2018 and as a teaching hospital had frequent rotating residents and fellows new to processes/protocols. These factors may have contributed to variations in unadjusted mortality. Also the decrease in inpatient mortality and improvement in SMR on acute LOC could have been the result of factors other than the EWSS and the effect of education alone may have been at least as good as that of the EWSS intervention.
Conclusions
Education along with the possible implementation of an EWSS at NF/SGVHS was associated with a decrease in the number of inpatient deaths on MRVAMC’s acute LOC wards from as high as 48 in FY 2017, quarter 3 to as low as 27 in FY 2019, quarter 4 resulting in as large of an improvement as a 44% reduction in unadjusted mortality from FY 2017 to FY 2019. In addition, MRVAMC’s acute LOC SMR improved from > 1.0 to < 1.0, demonstrating fewer inpatient mortalities than predicted from FY 2017 to FY 2019.
This multifaceted interventional strategy may be effectively applied at other VHA health care facilities that use the same EHR system. Next steps may include determining the specificity of MEWS + SRS as a sepsis screening tool; studying outcomes of MRVAMC’s EWSS during the COVID-19 era; and conducting a multicentered study on this EWSS across multiple VHA facilities.
In 1997, Elizabeth McGlynn wrote, “Measuring quality is no simple task.”1 We are reminded of this seminal Health Affairs article at a very pertinent point—as health care practice progresses, measuring the impact of performance improvement initiatives on clinical care delivery remains integral to monitoring overall effectiveness of quality. Mortality outcomes are a major focus of quality.
Inpatient mortality within the Veterans Health Administration (VHA) was measured as actual number of deaths (unadjusted mortality), and adjusted mortality was calculated using the standardized mortality ratio (SMR). SMR included actual number of deaths during hospitalization or within 1 day of hospital discharge divided by predicted number of deaths using a risk-adjusted formula and was calculated separately for acute level of care (LOC) and the intensive care unit (ICU). Using risk-adjusted SMR, if an observed/expected ratio was > 1.0, there were more inpatient deaths than expected; if < 1.0, fewer inpatient deaths occurred than predicted; and if 1.0, observed number of inpatient deaths was equivalent to expected number of deaths.2
Mortality reduction is a complex area of performance improvement. Health care facilities often focus their efforts on the biggest mortality contributors. According to Dantes and Epstein, sepsis results in about 265,000 deaths annually in the United States.3 Reinhart and colleagues demonstrated that sepsis is a worldwide issue resulting in approximately 30 million cases and 6 million deaths annually.4 Furthermore, Kumar and colleagues have noted that when sepsis progresses to septic shock, survival decreases by almost 8% for each hour delay in sepsis identification and treatment.5
Improvements in sepsis management have been multifaceted. The Surviving Sepsis Campaign guidelines created sepsis treatment bundles to guide early diagnosis/treatment of sepsis.6 In addition to awareness and sepsis care bundles, a plethora of informatics solutions within electronic health record (EHR) systems have demonstrated improved sepsis care.7-16 Various approaches to early diagnosis and management of sepsis have been collectively referred to as an early warning sepsis system (EWSS).
An EWSS typically contains automated decision support tools that are integrated in the EHR and meant to assist health care professionals with clinical workflow decision-making. Automated decision support tools within the EHR have a variety of functions, such as clinical care reminders and alerts.17
Sepsis screening tools function as a form of automated decision support and may be incorporated into the EHR to support the EWSS. Although sepsis screening tools vary, they frequently include a combination of data involving vital signs, laboratory values and/or physical examination findings, such as mental status evaluation.The Modified Early Warning Signs (MEWS) + Sepsis Recognition Score (SRS) is one example of a sepsis screening tool.7,16
At Malcom Randall Veterans Affairs Medical Center (MRVAMC) in Gainesville, Florida, we identified a quality improvement project opportunity to improve sepsis care in the emergency department (ED) and inpatient wards using the VHA EHR system, the Computerized Patient Record System (CPRS), which is supported by the Veterans Information Systems and Technology Architecture (VistA).18 A VistA/CPRS EWSS was developed using Lean Six Sigma DMAIC (define, measure, analyze, improve, and control) methodology.19 During the improve stage, informatics solutions were applied and included a combination of EHR interventions, such as template design, an order set, and clinical reminders. Clinical reminders have a wide variety of use, such as reminders for clinical tasks and as automated decision support within clinical workflows using Boolean logic.
To the best of our knowledge, there has been no published application of an EWSS within VistA/CPRS. In this study, we outline the strategic development of an EWSS in VistA/CPRS that assisted clinical staff with identification and treatment of sepsis; improved documentation of sepsis when present; and associated with improvement in unadjusted and adjusted inpatient mortality.
Methods
According to policy activities that constitute research at MRVAMC, no institutional review board approval was required as this work met criteria for operational improvement activities exempt from ethics review.
The North Florida/South Georgia Veterans Health System (NF/SGVHS) includes MRVAMC, a large academic hospital with rotating residents/fellows and multiple specialty care services. MRVAMC comprised 144 beds on the medicine/surgery wards; 48 beds in the psychiatry unit; 18 intermediate LOC beds; and 27 ICU beds. The MRVAMC SMR was identified as an improvement opportunity during fiscal year (FY) 2017 (Table 1). Its adjusted mortality for acute LOC demonstrated an observed/expected ratio of > 1.0 suggesting more inpatient deaths were observed than expected. The number of deaths (unadjusted mortality) on acute LOC at MRVAMC was noted to be rising during the first 3 quarters of FY 2017. A deeper examination of data by Pyramid Analytics (www.pyramidanalytics.com) discovered that sepsis was the primary driver for inpatient mortality on acute LOC at MRVAMC. Our goal was to reduce inpatient sepsis-related mortality via development of an EWSS that leveraged VistA/CPRS to improve early identification and treatment of sepsis in the ED and inpatient wards.
Emergency Department
Given the importance of recognizing sepsis early, the sepsis team focused on improvement opportunities at the initial point of patient contact: ED triage. The goal was to incorporate automated VistA/CPRS decision support to assist clinicians with identifying sepsis in triage using MEWS, which was chosen to optimize immediate hospital-wide buy-in. Clinical staff were already familiar with MEWS, which was in use on the inpatient wards.
Flow through the ED and availability of resources differed from the wards. Hence, modification to MEWS on the wards was necessary to fit clinical workflow in the ED. Temperature, heart rate (HR), respiratory rate (RR), systolic blood pressure (SBP), mental status, and white blood cell count (WBC) factored into a MEWS + SRS score on the wards (Table 2). For the ED, MEWS included temperature, HR, RR and SBP, but excluded mental status and WBC. Mental status assessment was excluded due to technical infeasibility (while vital signs could be automatically calculated in real time for a MEWS score, that was not possible for mental status changes). WBC was excluded from the ED as laboratory test results would not be available in triage.
MEWS + SRS scores were calculated in VistA by using clinical reminders. Clinical reminder logic included a series of conditional statements based on various combinations of MEWS + SRS clinical data entered in the EHR. When ED triage vital signs data were entered in CPRS, clinical data were stored and processed according to clinical reminder logic in VistA and displayed to the user in CPRS. While MEWS of ≥ 5 triggered a sepsis alert on the wards, the ≥ 4 threshold was used in the ED given mental status and WBC were excluded from calculations in triage (eAppendix 1 available at doi:10.12788/fp.0194).
Once a sepsis alert was triggered in triage for MEWS ≥ 4, ED nursing staff prioritized bed location and expedited staffing with an ED attending physician for early assessment. The ED attending then performed an assessment to confirm whether sepsis was present and direct early treatment. Although every patient who triggered a sepsis alert in triage did not meet clinical findings of sepsis, patients with MEWS ≥ 4 were frequently ill and required timely intervention.
If an ED attending physician agreed with a sepsis diagnosis, the physician had access to a sepsis workup and treatment order set in CPRS (eAppendix 2 available at doi:10.12788/fp.0194). The sepsis order set incorporated recommendations from the Surviving Sepsis Campaign guidelines and included orders for 2 large-bore peripheral IV lines; aggressive fluid resuscitation (30 mL/kg) for patients with clinical findings of hypoperfusion; broad-spectrum antibiotics; and frequent ordering of laboratory tests and imaging during initial sepsis workup.6 Vancomycin and cefepime were selected as routine broad-spectrum antibiotics in the order set when sepsis was suspected based on local antimicrobial stewardship and safety-efficacy profiles. For example, Luther and colleagues demonstrated that cefepime has lower rates of acute kidney injury when combined with vancomycin vs vancomycin + piperacillin-tazobactam.20 If a β-lactam antibiotic could not be used due to a patient’s drug allergy history, aztreonam was available as an alternative option.
The design of the order set also functioned as a communication interface with clinical pharmacists. Given the large volume of antibiotics ordered in the ED, it was difficult for pharmacists to prioritize antibiotic order verification. While stat orders convey high priority, they often lack specificity. When antibiotic orders were selected from the sepsis order set, comments were already included that stated: “STAT. First dose for sepsis protocol” (eAppendix 3 available at doi:10.12788/fp.0194). This standardized communication conveyed a sense of urgency and a collective understanding that patients with suspected sepsis required timely order verification and administration of antibiotics.
Hospital Ward
Mental status and WBC were included on the wards to monitor for possible signs of sepsis, using MEWS + SRS, which was routinely monitored by nursing every 4 to 8 hours. When MEWS + SRS was ≥ 5 points, ward nursing staff called a sepsis alert.7,16 Early response team (ERT) members received telephone notifications of the alert. ERT staff proceeded with immediate evaluation and treatment at the bedside along with determination for most appropriate LOC. The ERT members included an ICU physician and nurse; respiratory therapist; and nursing supervisor/bed flow coordinator. During bedside evaluation, if the ERT or primary team agreed with a sepsis diagnosis, the ERT or primary team used the sepsis order set to ensure standardized procedures. Stat orders generated through the sepsis order set pathway conveyed a sense of urgency and need for immediate order verification and administration of antibiotics.
In addition to clinical care process improvement, accurate documentation also was emphasized in the EWSS. When a sepsis alert was called, a clinician from the primary team was expected to complete a standardized progress note, which communicated clinical findings, a treatment plan, and captured severity of illness (eAppendix 4 available at doi:10.12788/fp.0194). It included sections for subjective, objective, assessment, and plan. In addition, data objects were created for vital signs and common laboratory findings that retrieved important clinical data from VistA and inserted it into the CPRS note.21
Nursing staff on the wards were expected to communicate results with the primary team for clinical decision making when a patient had a MEWS + SRS of 3 to 4. A sepsis alert may have been called at the discretion of clinical team members but was not required if the score was < 5. Additionally, vital signs were expected to be checked by the nursing staff on the wards at least every 4 hours for closer monitoring.
Sepsis Review Meetings
Weekly meetings were scheduled to review sepsis cases to assess diagnosis, treatment, and documentation entered in the patient record. The team conducting sepsis reviews comprised the chief of staff, chief of quality management, director of patient safety, physician utilization management advisor, chief resident in quality and patient safety (CRQS), and inpatient pharmacy supervisor. In addition, ad hoc physicians and nurses from different specialty areas, such as infectious diseases, hospitalist section, ICU, and the ED participated on request for subject matter expertise when needed. At the conclusion of weekly sepsis meetings, sepsis team members provided feedback to the clinical staff for continuous improvement purposes.
Results
Before implementation of an EWSS at NF/SGVHS, a plan was devised to increase awareness and educate staff on sepsis-related mortality in late FY 2017. Awareness and education about sepsis-related mortality was organized at physician, nursing, and pharmacy leadership clinical staff meetings. Posters about early warning signs of sepsis also were displayed on the nursing units for educational purposes and to convey the importance of early recognition/treatment of sepsis. In addition, the CRQS was the quality leader for house staff and led sepsis campaign change efforts for residents/fellows. An immediate improvement in unadjusted mortality at MRVAMC was noted with initial sepsis awareness and education. From FY 2017, quarter 3 to FY 2018, quarter 1, the number of acute LOC inpatient deaths decreased from 48 to 28, a 42% reduction in unadjusted mortality at MRVAMC (Figure 1). Additionally, the acute LOC SMR improved from 1.20 during FY 2017, quarter 3 down to as low as 0.71 during FY 2018, quarter 1 (Figure 2).
The number of MRVAMC inpatient deaths increased from 28 in FY 2018, quarter 1 to 45 in FY 2018, quarter 3. While acute LOC showed improvement in unadjusted mortality after sepsis education/awareness, it was felt continuous improvement could not be sustained with education alone. An EWSS was designed and implemented within the EHR system in FY 2018. Following implementation of EWSS and reeducating staff on early recognition and treatment of sepsis, acute LOC inpatient deaths decreased from 45 in FY 2018, quarter 3 through FY 2019 where unadjusted mortality was as low as 27 during FY 2019, quarter 4. The MRVAMC acute LOC SMR was consistently < 1.0 from FY 2018, quarter 4 through FY 2019, quarter 4.
In addition to the observed decrease in acute LOC inpatient deaths and improved SMR, the number of ERT alerts and sepsis alerts on the inpatient wards were monitored from FY 2017 through FY 2019. ERT alerts listed in Table 3 were nonspecific and initiated by nursing staff on the wards where a patient’s clinical status was identified as worsening while sepsis alerts were specific ERT alerts called by the ward nursing staff due to concerns for sepsis. The inpatient wards included inpatient medicine, surgery, and psychiatry acute care and the intermediate level of care unit while outpatient clinical areas of treatment, intensive care units, stroke alerts, and STEMI alerts were excluded.
From FY 2017 to FY 2018, quarter 1, the number of nonspecific ERT alerts varied between 75 to 100. Sepsis alerts were not available until December 2017 while the EWSS was in development. Afterward, nonspecific ERT alerts and sepsis alerts were monitored each quarter. Sepsis alerts ranged from 4 to 18. Nonspecific ERT alerts + sepsis alerts continued to increase from FY 2018, quarter 3 through FY 2019, quarter 4.
Discussion
Implementation of the EWSS was associated with improved unadjusted mortality and adjusted mortality for acute LOC at MRVAMC. Although variation exists with application of EWSS at other medical centers, there was similarity with improved sepsis outcomes reported at other health care systems after EWSS implementation.7-16
Improved unadjusted mortality and adjusted mortality for acute LOC at MRVAMC was likely due to multiple contributing factors. First, during design and implementation of the EWSS, project work was interdisciplinary with input from physicians, nurses, and pharmacists from multiple specialties (ie, ED, ICU, and the medicine service); quality management and data analysis specialists; and clinical informatics. Second, facility commitment to improving early recognition and treatment of sepsis from leadership level down to front-line staff was evident. Weekly sepsis meetings with the NF/SGVHS chief of staff helped to sustain EWSS efforts and to identify additional improvement opportunities. Third, integrated informatics solutions within the EHR helped identify early sepsis and minimized human error as well as assisted with coordination of sepsis care across services. Fourth, the focus was on both early identification and treatment of sepsis in the ED and hospital wards. Although it cannot be deduced whether there was causation between reduced inpatient mortality and an increased number of nonspecific ERT alerts+ sepsis alerts on the inpatient wards after EWSS implementation, inpatient deaths decreased and SMR improved. Finally, the EWSS emphasized both the importance of evidence-based clinical care of sepsis and standardized documentation to appropriately capture clinical severity of illness.
Limitations
This program has limitations. The EWSS was studied at a single VHA facility. Veteran demographics and local epidemiology may limit conclusion of outcomes to an individual VHA facility located in a specific geographical region. Additional research is necessary to demonstrate reproducibility and determine whether applicable to other VHA facilities and community care settings.
SMR is a risk-adjusted formula developed by the VHA Inpatient Evaluation Center, which included numerous factors such as diagnosis, comorbid conditions, age, marital status, procedures, source of admission, specific laboratory values, medical or surgical diagnosis-related group, ICU stays, immunosuppressive status, and a COVID-19 positive indicator (added after this study). Further research is needed to evaluate sepsis-related outcomes using the EWSS during the COVID-19 pandemic.
EWSS in the literature have demonstrated various approaches to early identification and treatment of sepsis and have used different sepsis screening tools.22 Evidence suggests that the MEWS + SRS sepsis screening tool may result in false-positive screenings.23-27 Additional research into the specificity of this sepsis screening tool is needed. Ward nursing staff were encouraged to initiate automatic sepsis alerts when MEWS + SRS was ≥ 5; however, this still depended on human factors. Because sepsis alerts are software-specific and others were incompatible with the VHA EHR, it was necessary to design our own EWSS.
Despite improvement with MRVAMC acute LOC unadjusted and adjusted mortality with our EWSS, we did not identify any actual improvement in earlier antibiotic administration times once sepsis was recognized. While accurate documentation regarding degree of sepsis improved, a MRVAMC clinical documentation improvement program was expanded in FY 2018. Therefore, it is difficult to demonstrate causation related to improved sepsis documentation with template changes alone. While sepsis alerts on the inpatient wards were variable since EWSS implementation, nonspecific ERT alerts increased. It is unclear whether some sepsis alerts were called as nonspecific ERT alerts, making it impossible to know the true number of sepsis alerts.
MRVAMC experienced an increase in nurse turnover during FY 2018 and as a teaching hospital had frequent rotating residents and fellows new to processes/protocols. These factors may have contributed to variations in unadjusted mortality. Also the decrease in inpatient mortality and improvement in SMR on acute LOC could have been the result of factors other than the EWSS and the effect of education alone may have been at least as good as that of the EWSS intervention.
Conclusions
Education along with the possible implementation of an EWSS at NF/SGVHS was associated with a decrease in the number of inpatient deaths on MRVAMC’s acute LOC wards from as high as 48 in FY 2017, quarter 3 to as low as 27 in FY 2019, quarter 4 resulting in as large of an improvement as a 44% reduction in unadjusted mortality from FY 2017 to FY 2019. In addition, MRVAMC’s acute LOC SMR improved from > 1.0 to < 1.0, demonstrating fewer inpatient mortalities than predicted from FY 2017 to FY 2019.
This multifaceted interventional strategy may be effectively applied at other VHA health care facilities that use the same EHR system. Next steps may include determining the specificity of MEWS + SRS as a sepsis screening tool; studying outcomes of MRVAMC’s EWSS during the COVID-19 era; and conducting a multicentered study on this EWSS across multiple VHA facilities.
1. McGlynn EA. Six challenges in measuring the quality of health care. Health Aff (Millwood). 1997;16(3):7-21. doi:10.1377/hlthaff.16.3.7
2. US Department of Veterans Affairs, Veterans Health Administration. Strategic Analytics for Improvement and Learning (SAIL) value model measure definitions. Updated May 15, 2019. Accessed October 11, 2021. https://www.va.gov/QUALITYOFCARE/measure-up/SAIL_definitions.asp
3. Dantes RB, Epstein L. Combatting sepsis: a public health perspective. Clin Infect Dis. 2018;67(8):1300-1302. doi:10.1093/cid/ciy342
4. Reinhart K, Daniels R, Kissoon N, Machado FR, Schachter RD, Finfer S. Recognizing sepsis as a global health priority - a WHO resolution. N Engl J Med. 2017;377(5):414-417. doi:10.1056/NEJMp1707170
5. Kumar A, Roberts D, Wood KE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34(6):1589-1596. doi:10.1097/01.CCM.0000217961.75225.E9
6. Rhodes A, Evans LE, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45(3):486-552. doi:10.1097/CCM.0000000000002255
7. Guirgis FW, Jones L, Esma R, et al. Managing sepsis: electronic recognition, rapid response teams, and standardized care save lives. J Crit Care. 2017;40:296-302. doi:10.1016/j.jcrc.2017.04.005
8. Whippy A, Skeath M, Crawford B, et al. Kaiser Permanente’s performance improvement system, part 3: multisite improvements in care for patients with sepsis. Jt Comm J Qual Patient Saf. 2011;37(11):483-493. doi:10.1016/s1553-7250(11)37061-4
9. Harrison AM, Thongprayoon C, Kashyap R, et al. Developing the surveillance algorithm for detection of failure to recognize and treat severe sepsis. Mayo Clin Proc. 2015;90(2):166-175. doi:10.1016/j.mayocp.2014.11.014
10. Rothman M, Levy M, Dellinger RP, et al. Sepsis as 2 problems: identifying sepsis at admission and predicting onset in the hospital using an electronic medical record-based acuity score. J Crit Care. 2017;38:237-244. doi:10.1016/j.jcrc.2016.11.037
11. Back JS, Jin Y, Jin T, Lee SM. Development and validation of an automated sepsis risk assessment system. Res Nurs Health. 2016;39(5):317-327. doi:10.1002/nur.21734
12. Khurana HS, Groves RH Jr, Simons MP, et al. Real-time automated sampling of electronic medical records predicts hospital mortality. Am J Med. 2016;129(7):688-698.e2. doi:10.1016/j.amjmed.2016.02.037
13. Umscheid CA, Betesh J, VanZandbergen C, et al. Development, implementation, and impact of an automated early warning and response system for sepsis. J Hosp Med. 2015;10(1):26-31. doi:10.1002/jhm.2259
14. Vogel L. EMR alert cuts sepsis deaths. CMAJ. 2014;186(2):E80. doi:10.1503/cmaj.109-4686
15. Jones SL, Ashton CM, Kiehne L, et al. Reductions in sepsis mortality and costs after design and implementation of a nurse-based early recognition and response program. Jt Comm J Qual Patient Saf. 2015;41(11):483-491. doi:10.1016/s1553-7250(15)41063-3
16. Croft CA, Moore FA, Efron PA, et al. Computer versus paper system for recognition and management of sepsis in surgical intensive care. J Trauma Acute Care Surg. 2014;76(2):311-319. doi:10.1097/TA.0000000000000121
17. Tcheng JE, Bakken S, Bates DW, et al, eds. Optimizing Strategies for Clinical Decision Support: Summary of a Meeting Series. National Academy of Medicine; 2017. Accessed October 11, 2021. https://nam.edu/wp-content/uploads/2017/11/Optimizing-Strategies-for-Clinical-Decision-Support.pdf
18. US Department of Veterans Affairs. History of IT at VA. Updated January 1, 2020. Accessed October 11, 2021. https://www.oit.va.gov/about/history.cfm
19. GoLeanSixSigma. DMAIC: The 5 Phases of Lean Six Sigma. Published 2012. Accessed October 11, 2021. https://goleansixsigma.com/wp-content/uploads/2012/02/DMAIC-The-5-Phases-of-Lean-Six-Sigma-www.GoLeanSixSigma.com_.pdf
20. Luther MK, Timbrook TT, Caffrey AR, Dosa D, Lodise TP, LaPlante KL. Vancomycin plus piperacillin-tazobactam and acute kidney injury in adults: a systematic review and meta-analysis. Crit Care Med. 2018;46(1):12-20. doi:10.1097/CCM.0000000000002769
21. International Business Machines Corp. Overview of data objects. Accessed October 11, 2021. https://www.ibm.com/support/knowledgecenter/en/SSLTBW_2.3.0/com.ibm.zos.v2r3.cbclx01/data_objects.htm
22. Churpek MM, Snyder A, Han X, et al. Quick sepsis-related organ failure assessment, systemic inflammatory response syndrome, and early warning scores for detecting clinical deterioration in infected patients outside the intensive care unit. Am J Respir Crit Care Med. 2017;195(7):906-911. doi:10.1164/rccm.201604-0854OC
23. Ghanem-Zoubi NO, Vardi M, Laor A, Weber G, Bitterman H. Assessment of disease-severity scoring systems for patients with sepsis in general internal medicine departments. Crit Care. 2011;15(2):R95. doi:10.1186/cc10102
24. Hamilton F, Arnold D, Baird A, Albur M, Whiting P. Early Warning scores do not accurately predict mortality in sepsis: a meta-analysis and systematic review of the literature. J Infect. 2018;76(3):241-248. doi:10.1016/j.jinf.2018.01.002
25. Martino IF, Figgiaconi V, Seminari E, Muzzi A, Corbella M, Perlini S. The role of qSOFA compared to other prognostic scores in septic patients upon admission to the emergency department. Eur J Intern Med. 2018;53:e11-e13. doi:10.1016/j.ejim.2018.05.022
26. Nannan Panday RS, Minderhoud TC, Alam N, Nanayakkara PWB. Prognostic value of early warning scores in the emergency department (ED) and acute medical unit (AMU): A narrative review. Eur J Intern Med. 2017;45:20-31. doi:10.1016/j.ejim.2017.09.027
27. Jayasundera R, Neilly M, Smith TO, Myint PK. Are early warning scores useful predictors for mortality and morbidity in hospitalised acutely unwell older patients? A systematic review. J Clin Med. 2018;7(10):309. Published 2018 Sep 28. doi:10.3390/jcm7100309
1. McGlynn EA. Six challenges in measuring the quality of health care. Health Aff (Millwood). 1997;16(3):7-21. doi:10.1377/hlthaff.16.3.7
2. US Department of Veterans Affairs, Veterans Health Administration. Strategic Analytics for Improvement and Learning (SAIL) value model measure definitions. Updated May 15, 2019. Accessed October 11, 2021. https://www.va.gov/QUALITYOFCARE/measure-up/SAIL_definitions.asp
3. Dantes RB, Epstein L. Combatting sepsis: a public health perspective. Clin Infect Dis. 2018;67(8):1300-1302. doi:10.1093/cid/ciy342
4. Reinhart K, Daniels R, Kissoon N, Machado FR, Schachter RD, Finfer S. Recognizing sepsis as a global health priority - a WHO resolution. N Engl J Med. 2017;377(5):414-417. doi:10.1056/NEJMp1707170
5. Kumar A, Roberts D, Wood KE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34(6):1589-1596. doi:10.1097/01.CCM.0000217961.75225.E9
6. Rhodes A, Evans LE, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45(3):486-552. doi:10.1097/CCM.0000000000002255
7. Guirgis FW, Jones L, Esma R, et al. Managing sepsis: electronic recognition, rapid response teams, and standardized care save lives. J Crit Care. 2017;40:296-302. doi:10.1016/j.jcrc.2017.04.005
8. Whippy A, Skeath M, Crawford B, et al. Kaiser Permanente’s performance improvement system, part 3: multisite improvements in care for patients with sepsis. Jt Comm J Qual Patient Saf. 2011;37(11):483-493. doi:10.1016/s1553-7250(11)37061-4
9. Harrison AM, Thongprayoon C, Kashyap R, et al. Developing the surveillance algorithm for detection of failure to recognize and treat severe sepsis. Mayo Clin Proc. 2015;90(2):166-175. doi:10.1016/j.mayocp.2014.11.014
10. Rothman M, Levy M, Dellinger RP, et al. Sepsis as 2 problems: identifying sepsis at admission and predicting onset in the hospital using an electronic medical record-based acuity score. J Crit Care. 2017;38:237-244. doi:10.1016/j.jcrc.2016.11.037
11. Back JS, Jin Y, Jin T, Lee SM. Development and validation of an automated sepsis risk assessment system. Res Nurs Health. 2016;39(5):317-327. doi:10.1002/nur.21734
12. Khurana HS, Groves RH Jr, Simons MP, et al. Real-time automated sampling of electronic medical records predicts hospital mortality. Am J Med. 2016;129(7):688-698.e2. doi:10.1016/j.amjmed.2016.02.037
13. Umscheid CA, Betesh J, VanZandbergen C, et al. Development, implementation, and impact of an automated early warning and response system for sepsis. J Hosp Med. 2015;10(1):26-31. doi:10.1002/jhm.2259
14. Vogel L. EMR alert cuts sepsis deaths. CMAJ. 2014;186(2):E80. doi:10.1503/cmaj.109-4686
15. Jones SL, Ashton CM, Kiehne L, et al. Reductions in sepsis mortality and costs after design and implementation of a nurse-based early recognition and response program. Jt Comm J Qual Patient Saf. 2015;41(11):483-491. doi:10.1016/s1553-7250(15)41063-3
16. Croft CA, Moore FA, Efron PA, et al. Computer versus paper system for recognition and management of sepsis in surgical intensive care. J Trauma Acute Care Surg. 2014;76(2):311-319. doi:10.1097/TA.0000000000000121
17. Tcheng JE, Bakken S, Bates DW, et al, eds. Optimizing Strategies for Clinical Decision Support: Summary of a Meeting Series. National Academy of Medicine; 2017. Accessed October 11, 2021. https://nam.edu/wp-content/uploads/2017/11/Optimizing-Strategies-for-Clinical-Decision-Support.pdf
18. US Department of Veterans Affairs. History of IT at VA. Updated January 1, 2020. Accessed October 11, 2021. https://www.oit.va.gov/about/history.cfm
19. GoLeanSixSigma. DMAIC: The 5 Phases of Lean Six Sigma. Published 2012. Accessed October 11, 2021. https://goleansixsigma.com/wp-content/uploads/2012/02/DMAIC-The-5-Phases-of-Lean-Six-Sigma-www.GoLeanSixSigma.com_.pdf
20. Luther MK, Timbrook TT, Caffrey AR, Dosa D, Lodise TP, LaPlante KL. Vancomycin plus piperacillin-tazobactam and acute kidney injury in adults: a systematic review and meta-analysis. Crit Care Med. 2018;46(1):12-20. doi:10.1097/CCM.0000000000002769
21. International Business Machines Corp. Overview of data objects. Accessed October 11, 2021. https://www.ibm.com/support/knowledgecenter/en/SSLTBW_2.3.0/com.ibm.zos.v2r3.cbclx01/data_objects.htm
22. Churpek MM, Snyder A, Han X, et al. Quick sepsis-related organ failure assessment, systemic inflammatory response syndrome, and early warning scores for detecting clinical deterioration in infected patients outside the intensive care unit. Am J Respir Crit Care Med. 2017;195(7):906-911. doi:10.1164/rccm.201604-0854OC
23. Ghanem-Zoubi NO, Vardi M, Laor A, Weber G, Bitterman H. Assessment of disease-severity scoring systems for patients with sepsis in general internal medicine departments. Crit Care. 2011;15(2):R95. doi:10.1186/cc10102
24. Hamilton F, Arnold D, Baird A, Albur M, Whiting P. Early Warning scores do not accurately predict mortality in sepsis: a meta-analysis and systematic review of the literature. J Infect. 2018;76(3):241-248. doi:10.1016/j.jinf.2018.01.002
25. Martino IF, Figgiaconi V, Seminari E, Muzzi A, Corbella M, Perlini S. The role of qSOFA compared to other prognostic scores in septic patients upon admission to the emergency department. Eur J Intern Med. 2018;53:e11-e13. doi:10.1016/j.ejim.2018.05.022
26. Nannan Panday RS, Minderhoud TC, Alam N, Nanayakkara PWB. Prognostic value of early warning scores in the emergency department (ED) and acute medical unit (AMU): A narrative review. Eur J Intern Med. 2017;45:20-31. doi:10.1016/j.ejim.2017.09.027
27. Jayasundera R, Neilly M, Smith TO, Myint PK. Are early warning scores useful predictors for mortality and morbidity in hospitalised acutely unwell older patients? A systematic review. J Clin Med. 2018;7(10):309. Published 2018 Sep 28. doi:10.3390/jcm7100309
Vitamin D and omega-3 supplements reduce autoimmune disease risk
For those of us who cannot sit in the sun and fish all day, the next best thing for preventing autoimmune diseases may be supplementation with vitamin D and fish oil-derived omega-3 fatty acids, results of a large prospective randomized trial suggest.
Among nearly 26,000 adults enrolled in a randomized trial designed primarily to study the effects of vitamin D and omega-3 supplementation on incident cancer and cardiovascular disease, 5, and 5 years of omega-3 fatty acid supplementation was associated with an 18% reduction in confirmed and probable incident autoimmune diseases, reported Karen H. Costenbader, MD, MPH, of Brigham & Women’s Hospital in Boston.
“The clinical importance of these results is very high, given that these are nontoxic, well-tolerated supplements, and that there are no other known effective therapies to reduce the incidence of autoimmune diseases,” she said during the virtual annual meeting of the American College of Rheumatology.
“People do have to take the supplements a long time to start to see the reduction in risk, especially for vitamin D, but they make biological sense, and autoimmune diseases develop slowly over time, so taking it today isn’t going to reduce risk of developing something tomorrow,” Dr. Costenbader said in an interview.
“These supplements have other health benefits. Obviously, fish oil is anti-inflammatory, and vitamin D is good for osteoporosis prevention, especially in our patients who take glucocorticoids. People who are otherwise healthy and have a family history of autoimmune disease might also consider starting to take these supplements,” she said.
After watching her presentation, session co-moderator Gregg Silverman, MD, from the NYU Langone School of Medicine in New York, who was not involved in the study, commented “I’m going to [nutrition store] GNC to get some vitamins.”
When asked for comment, the other session moderator, Tracy Frech, MD, of Vanderbilt University, Nashville, said, “I think Dr. Costenbader’s work is very important and her presentation excellent. My current practice is replacement of vitamin D in all autoimmune disease patients with low levels and per bone health guidelines. Additionally, I discuss omega-3 supplementation with Sjögren’s [syndrome] patients as a consideration.”
Evidence base
Dr. Costenbader noted that in a 2013 observational study from France, vitamin D derived through ultraviolet (UV) light exposure was associated with a lower risk for incident Crohn’s disease but not ulcerative colitis, and in two analyses of data in 2014 from the Nurses’ Health Study, both high plasma levels of 25-OH vitamin D and geographic residence in areas of high UV exposure were associated with a decreased incidence of rheumatoid arthritis (RA).
Other observational studies have supported omega-3 fatty acids for their anti-inflammatory properties, including a 2005 Danish prospective cohort study showing a lower risk for RA in participants who reported higher levels of fatty fish intake. In a separate study conducted in 2017, healthy volunteers with higher omega-3 fatty acid/total lipid proportions in red blood cell membranes had a lower prevalence of anti-cyclic citrullinated peptide (anti-CCP) antibodies and rheumatoid factor and a lower incidence of progression to inflammatory arthritis, she said.
Ancillary study
Despite the evidence, however, there have been no prospective randomized trials to test the effects of either vitamin D or omega-3 fatty acid supplementation on the incidence of autoimmune disease over time.
To rectify this, Dr. Costenbader and colleagues piggybacked an ancillary study onto the Vitamin D and Omega-3 Trial (VITAL), which had primary outcomes of cancer and cardiovascular disease incidence.
A total of 25,871 participants were enrolled, including 12,786 men aged 50 and older, and 13,085 women aged 55 and older.
The study had a 2 x 2 factorial design, with patients randomly assigned to vitamin D 2,000 IU/day or placebo, and then further randomized to either 1 g/day omega-3 fatty acids or placebo in both the vitamin D and placebo primary randomization arms.
At baseline 16,956 participants were assayed for 25-OH vitamin D and plasma omega 3 index, the ratio of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) to total fatty acids. Participants self-reported baseline and all incident autoimmune diseases annually, with the reports confirmed by medical record review and disease criteria whenever possible.
Results
At 5 years of follow-up, confirmed incident autoimmune diseases had occurred in 123 patients in the active vitamin D group, compared with 155 in the placebo vitamin D group, translating into a hazard ratio (HR) for vitamin D of 0.78 (P = .045).
In the active omega-3 arm, 130 participants developed an autoimmune disease, compared with 148 in the placebo omega-3 arm, which translated into a nonsignificant HR of 0.85.
There was no statistical interaction between the two supplements. The investigators did observe an interaction between vitamin D and body mass index, with the effect stronger among participants with low BMI (P = .02). There also was an interaction between omega-3 fatty acids with a family history of autoimmune disease (P = .03).
In multivariate analysis adjusted for age, sex, race, and other supplement arm, vitamin D alone was associated with an HR for incident autoimmune disease of 0.68 (P = .02), omega-3 alone was associated with a nonsignificant HR of 0.74, and the combination was associated with an HR of 0.69 (P = .03).
Dr. Costenbader and colleagues acknowledged that the study was limited by the lack of a high-risk or nutritionally-deficient population, where the effects of supplementation might be larger; the restriction of the sample to older adults; and to the difficulty of confirming incident autoimmune thyroid disease from patient reports.
Cheryl Koehn, an arthritis patient advocate from Vancouver, Canada, who was not involved in the study, commented in the “chat” section of the presentation that her rheumatologist “has recommended vitamin D for years now. Says basically everyone north of Boston is vitamin D deficient. I take 1,000 IU per day. Been taking it for years.” Ms. Koehn is the founder and president of Arthritis Consumer Experts, a website that provides education to those with arthritis.
“Agreed. I tell every patient to take vitamin D supplement,” commented Fatma Dedeoglu, MD, a rheumatologist at Boston Children’s Hospital.
A version of this article first appeared on Medscape.com.
For those of us who cannot sit in the sun and fish all day, the next best thing for preventing autoimmune diseases may be supplementation with vitamin D and fish oil-derived omega-3 fatty acids, results of a large prospective randomized trial suggest.
Among nearly 26,000 adults enrolled in a randomized trial designed primarily to study the effects of vitamin D and omega-3 supplementation on incident cancer and cardiovascular disease, 5, and 5 years of omega-3 fatty acid supplementation was associated with an 18% reduction in confirmed and probable incident autoimmune diseases, reported Karen H. Costenbader, MD, MPH, of Brigham & Women’s Hospital in Boston.
“The clinical importance of these results is very high, given that these are nontoxic, well-tolerated supplements, and that there are no other known effective therapies to reduce the incidence of autoimmune diseases,” she said during the virtual annual meeting of the American College of Rheumatology.
“People do have to take the supplements a long time to start to see the reduction in risk, especially for vitamin D, but they make biological sense, and autoimmune diseases develop slowly over time, so taking it today isn’t going to reduce risk of developing something tomorrow,” Dr. Costenbader said in an interview.
“These supplements have other health benefits. Obviously, fish oil is anti-inflammatory, and vitamin D is good for osteoporosis prevention, especially in our patients who take glucocorticoids. People who are otherwise healthy and have a family history of autoimmune disease might also consider starting to take these supplements,” she said.
After watching her presentation, session co-moderator Gregg Silverman, MD, from the NYU Langone School of Medicine in New York, who was not involved in the study, commented “I’m going to [nutrition store] GNC to get some vitamins.”
When asked for comment, the other session moderator, Tracy Frech, MD, of Vanderbilt University, Nashville, said, “I think Dr. Costenbader’s work is very important and her presentation excellent. My current practice is replacement of vitamin D in all autoimmune disease patients with low levels and per bone health guidelines. Additionally, I discuss omega-3 supplementation with Sjögren’s [syndrome] patients as a consideration.”
Evidence base
Dr. Costenbader noted that in a 2013 observational study from France, vitamin D derived through ultraviolet (UV) light exposure was associated with a lower risk for incident Crohn’s disease but not ulcerative colitis, and in two analyses of data in 2014 from the Nurses’ Health Study, both high plasma levels of 25-OH vitamin D and geographic residence in areas of high UV exposure were associated with a decreased incidence of rheumatoid arthritis (RA).
Other observational studies have supported omega-3 fatty acids for their anti-inflammatory properties, including a 2005 Danish prospective cohort study showing a lower risk for RA in participants who reported higher levels of fatty fish intake. In a separate study conducted in 2017, healthy volunteers with higher omega-3 fatty acid/total lipid proportions in red blood cell membranes had a lower prevalence of anti-cyclic citrullinated peptide (anti-CCP) antibodies and rheumatoid factor and a lower incidence of progression to inflammatory arthritis, she said.
Ancillary study
Despite the evidence, however, there have been no prospective randomized trials to test the effects of either vitamin D or omega-3 fatty acid supplementation on the incidence of autoimmune disease over time.
To rectify this, Dr. Costenbader and colleagues piggybacked an ancillary study onto the Vitamin D and Omega-3 Trial (VITAL), which had primary outcomes of cancer and cardiovascular disease incidence.
A total of 25,871 participants were enrolled, including 12,786 men aged 50 and older, and 13,085 women aged 55 and older.
The study had a 2 x 2 factorial design, with patients randomly assigned to vitamin D 2,000 IU/day or placebo, and then further randomized to either 1 g/day omega-3 fatty acids or placebo in both the vitamin D and placebo primary randomization arms.
At baseline 16,956 participants were assayed for 25-OH vitamin D and plasma omega 3 index, the ratio of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) to total fatty acids. Participants self-reported baseline and all incident autoimmune diseases annually, with the reports confirmed by medical record review and disease criteria whenever possible.
Results
At 5 years of follow-up, confirmed incident autoimmune diseases had occurred in 123 patients in the active vitamin D group, compared with 155 in the placebo vitamin D group, translating into a hazard ratio (HR) for vitamin D of 0.78 (P = .045).
In the active omega-3 arm, 130 participants developed an autoimmune disease, compared with 148 in the placebo omega-3 arm, which translated into a nonsignificant HR of 0.85.
There was no statistical interaction between the two supplements. The investigators did observe an interaction between vitamin D and body mass index, with the effect stronger among participants with low BMI (P = .02). There also was an interaction between omega-3 fatty acids with a family history of autoimmune disease (P = .03).
In multivariate analysis adjusted for age, sex, race, and other supplement arm, vitamin D alone was associated with an HR for incident autoimmune disease of 0.68 (P = .02), omega-3 alone was associated with a nonsignificant HR of 0.74, and the combination was associated with an HR of 0.69 (P = .03).
Dr. Costenbader and colleagues acknowledged that the study was limited by the lack of a high-risk or nutritionally-deficient population, where the effects of supplementation might be larger; the restriction of the sample to older adults; and to the difficulty of confirming incident autoimmune thyroid disease from patient reports.
Cheryl Koehn, an arthritis patient advocate from Vancouver, Canada, who was not involved in the study, commented in the “chat” section of the presentation that her rheumatologist “has recommended vitamin D for years now. Says basically everyone north of Boston is vitamin D deficient. I take 1,000 IU per day. Been taking it for years.” Ms. Koehn is the founder and president of Arthritis Consumer Experts, a website that provides education to those with arthritis.
“Agreed. I tell every patient to take vitamin D supplement,” commented Fatma Dedeoglu, MD, a rheumatologist at Boston Children’s Hospital.
A version of this article first appeared on Medscape.com.
For those of us who cannot sit in the sun and fish all day, the next best thing for preventing autoimmune diseases may be supplementation with vitamin D and fish oil-derived omega-3 fatty acids, results of a large prospective randomized trial suggest.
Among nearly 26,000 adults enrolled in a randomized trial designed primarily to study the effects of vitamin D and omega-3 supplementation on incident cancer and cardiovascular disease, 5, and 5 years of omega-3 fatty acid supplementation was associated with an 18% reduction in confirmed and probable incident autoimmune diseases, reported Karen H. Costenbader, MD, MPH, of Brigham & Women’s Hospital in Boston.
“The clinical importance of these results is very high, given that these are nontoxic, well-tolerated supplements, and that there are no other known effective therapies to reduce the incidence of autoimmune diseases,” she said during the virtual annual meeting of the American College of Rheumatology.
“People do have to take the supplements a long time to start to see the reduction in risk, especially for vitamin D, but they make biological sense, and autoimmune diseases develop slowly over time, so taking it today isn’t going to reduce risk of developing something tomorrow,” Dr. Costenbader said in an interview.
“These supplements have other health benefits. Obviously, fish oil is anti-inflammatory, and vitamin D is good for osteoporosis prevention, especially in our patients who take glucocorticoids. People who are otherwise healthy and have a family history of autoimmune disease might also consider starting to take these supplements,” she said.
After watching her presentation, session co-moderator Gregg Silverman, MD, from the NYU Langone School of Medicine in New York, who was not involved in the study, commented “I’m going to [nutrition store] GNC to get some vitamins.”
When asked for comment, the other session moderator, Tracy Frech, MD, of Vanderbilt University, Nashville, said, “I think Dr. Costenbader’s work is very important and her presentation excellent. My current practice is replacement of vitamin D in all autoimmune disease patients with low levels and per bone health guidelines. Additionally, I discuss omega-3 supplementation with Sjögren’s [syndrome] patients as a consideration.”
Evidence base
Dr. Costenbader noted that in a 2013 observational study from France, vitamin D derived through ultraviolet (UV) light exposure was associated with a lower risk for incident Crohn’s disease but not ulcerative colitis, and in two analyses of data in 2014 from the Nurses’ Health Study, both high plasma levels of 25-OH vitamin D and geographic residence in areas of high UV exposure were associated with a decreased incidence of rheumatoid arthritis (RA).
Other observational studies have supported omega-3 fatty acids for their anti-inflammatory properties, including a 2005 Danish prospective cohort study showing a lower risk for RA in participants who reported higher levels of fatty fish intake. In a separate study conducted in 2017, healthy volunteers with higher omega-3 fatty acid/total lipid proportions in red blood cell membranes had a lower prevalence of anti-cyclic citrullinated peptide (anti-CCP) antibodies and rheumatoid factor and a lower incidence of progression to inflammatory arthritis, she said.
Ancillary study
Despite the evidence, however, there have been no prospective randomized trials to test the effects of either vitamin D or omega-3 fatty acid supplementation on the incidence of autoimmune disease over time.
To rectify this, Dr. Costenbader and colleagues piggybacked an ancillary study onto the Vitamin D and Omega-3 Trial (VITAL), which had primary outcomes of cancer and cardiovascular disease incidence.
A total of 25,871 participants were enrolled, including 12,786 men aged 50 and older, and 13,085 women aged 55 and older.
The study had a 2 x 2 factorial design, with patients randomly assigned to vitamin D 2,000 IU/day or placebo, and then further randomized to either 1 g/day omega-3 fatty acids or placebo in both the vitamin D and placebo primary randomization arms.
At baseline 16,956 participants were assayed for 25-OH vitamin D and plasma omega 3 index, the ratio of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) to total fatty acids. Participants self-reported baseline and all incident autoimmune diseases annually, with the reports confirmed by medical record review and disease criteria whenever possible.
Results
At 5 years of follow-up, confirmed incident autoimmune diseases had occurred in 123 patients in the active vitamin D group, compared with 155 in the placebo vitamin D group, translating into a hazard ratio (HR) for vitamin D of 0.78 (P = .045).
In the active omega-3 arm, 130 participants developed an autoimmune disease, compared with 148 in the placebo omega-3 arm, which translated into a nonsignificant HR of 0.85.
There was no statistical interaction between the two supplements. The investigators did observe an interaction between vitamin D and body mass index, with the effect stronger among participants with low BMI (P = .02). There also was an interaction between omega-3 fatty acids with a family history of autoimmune disease (P = .03).
In multivariate analysis adjusted for age, sex, race, and other supplement arm, vitamin D alone was associated with an HR for incident autoimmune disease of 0.68 (P = .02), omega-3 alone was associated with a nonsignificant HR of 0.74, and the combination was associated with an HR of 0.69 (P = .03).
Dr. Costenbader and colleagues acknowledged that the study was limited by the lack of a high-risk or nutritionally-deficient population, where the effects of supplementation might be larger; the restriction of the sample to older adults; and to the difficulty of confirming incident autoimmune thyroid disease from patient reports.
Cheryl Koehn, an arthritis patient advocate from Vancouver, Canada, who was not involved in the study, commented in the “chat” section of the presentation that her rheumatologist “has recommended vitamin D for years now. Says basically everyone north of Boston is vitamin D deficient. I take 1,000 IU per day. Been taking it for years.” Ms. Koehn is the founder and president of Arthritis Consumer Experts, a website that provides education to those with arthritis.
“Agreed. I tell every patient to take vitamin D supplement,” commented Fatma Dedeoglu, MD, a rheumatologist at Boston Children’s Hospital.
A version of this article first appeared on Medscape.com.
FROM ACR 2021
Artificial Intelligence: Review of Current and Future Applications in Medicine
Artificial Intelligence (AI) was first described in 1956 and refers to machines having the ability to learn as they receive and process information, resulting in the ability to “think” like humans.1 AI’s impact in medicine is increasing; currently, at least 29 AI medical devices and algorithms are approved by the US Food and Drug Administration (FDA) in a variety of areas, including radiograph interpretation, managing glucose levels in patients with diabetes mellitus, analyzing electrocardiograms (ECGs), and diagnosing sleep disorders among others.2 Significantly, in 2020, the Centers for Medicare and Medicaid Services (CMS) announced the first reimbursement to hospitals for an AI platform, a model for early detection of strokes.3 AI is rapidly becoming an integral part of health care, and its role will only increase in the future (Table).
As knowledge in medicine is expanding exponentially, AI has great potential to assist with handling complex patient care data. The concept of exponential growth is not a natural one. As Bini described, with exponential growth the volume of knowledge amassed over the past 10 years will now occur in perhaps only 1 year.1 Likewise, equivalent advances over the past year may take just a few months. This phenomenon is partly due to the law of accelerating returns, which states that advances feed on themselves, continually increasing the rate of further advances.4 The volume of medical data doubles every 2 to 5 years.5 Fortunately, the field of AI is growing exponentially as well and can help health care practitioners (HCPs) keep pace, allowing the continued delivery of effective health care.
In this report, we review common terminology, principles, and general applications of AI, followed by current and potential applications of AI for selected medical specialties. Finally, we discuss AI’s future in health care, along with potential risks and pitfalls.
AI Overview
AI refers to machine programs that can “learn” or think based on past experiences. This functionality contrasts with simple rules-based programming available to health care for years. An example of rules-based programming is the warfarindosing.org website developed by Barnes-Jewish Hospital at Washington University Medical Center, which guides initial warfarin dosing.6,7 The prescriber inputs detailed patient information, including age, sex, height, weight, tobacco history, medications, laboratory results, and genotype if available. The application then calculates recommended warfarin dosing regimens to avoid over- or underanticoagulation. While the dosing algorithm may be complex, it depends entirely on preprogrammed rules. The program does not learn to reach its conclusions and recommendations from patient data.
In contrast, one of the most common subsets of AI is machine learning (ML). ML describes a program that “learns from experience and improves its performance as it learns.”1 With ML, the computer is initially provided with a training data set—data with known outcomes or labels. Because the initial data are input from known samples, this type of AI is known as supervised learning.8-10 As an example, we recently reported using ML to diagnose various types of cancer from pathology slides.11 In one experiment, we captured images of colon adenocarcinoma and normal colon (these 2 groups represent the training data set). Unlike traditional programming, we did not define characteristics that would differentiate colon cancer from normal; rather, the machine learned these characteristics independently by assessing the labeled images provided. A second data set (the validation data set) was used to evaluate the program and fine-tune the ML training model’s parameters. Finally, the program was presented with new images of cancer and normal cases for final assessment of accuracy (test data set). Our program learned to recognize differences from the images provided and was able to differentiate normal and cancer images with > 95% accuracy.
Advances in computer processing have allowed for the development of artificial neural networks (ANNs). While there are several types of ANNs, the most common types used for image classification and segmentation are known as convolutional neural networks (CNNs).9,12-14 The programs are designed to work similar to the human brain, specifically the visual cortex.15,16 As data are acquired, they are processed by various layers in the program. Much like neurons in the brain, one layer decides whether to advance information to the next.13,14 CNNs can be many layers deep, leading to the term deep learning: “computational models that are composed of multiple processing layers to learn representations of data with multiple levels of abstraction.”1,13,17
ANNs can process larger volumes of data. This advance has led to the development of unstructured or unsupervised learning. With this type of learning, imputing defined features (ie, predetermined answers) of the training data set described above is no longer required.1,8,10,14 The advantage of unsupervised learning is that the program can be presented raw data and extract meaningful interpretation without human input, often with less bias than may exist with supervised learning.1,18 If shown enough data, the program can extract relevant features to make conclusions independently without predefined definitions, potentially uncovering markers not previously known. For example, several studies have used unsupervised learning to search patient data to assess readmission risks of patients with congestive heart failure.10,19,20 AI compiled features independently and not previously defined, predicting patients at greater risk for readmission superior to traditional methods.
A more detailed description of the various terminologies and techniques of AI is beyond the scope of this review.9,10,17,21 However, in this basic overview, we describe 4 general areas that AI impacts health care (Figure).
Health Care Applications
Image analysis has seen the most AI health care applications.8,15 AI has shown potential in interpreting many types of medical images, including pathology slides, radiographs of various types, retina and other eye scans, and photographs of skin lesions. Many studies have demonstrated that AI can interpret these images as accurately as or even better than experienced clinicians.9,13,22-29 Studies have suggested AI interpretation of radiographs may better distinguish patients infected with COVID-19 from other causes of pneumonia, and AI interpretation of pathology slides may detect specific genetic mutations not previously identified without additional molecular tests.11,14,23,24,30-32
The second area in which AI can impact health care is improving workflow and efficiency. AI has improved surgery scheduling, saving significant revenue, and decreased patient wait times for appointments.1 AI can screen and triage radiographs, allowing attention to be directed to critical patients. This use would be valuable in many busy clinical settings, such as the recent COVID-19 pandemic.8,23 Similarly, AI can screen retina images to prioritize urgent conditions.25 AI has improved pathologists’ efficiency when used to detect breast metastases.33 Finally, AI may reduce medical errors, thereby ensuring patient safety.8,9,34
A third health care benefit of AI is in public health and epidemiology. AI can assist with clinical decision-making and diagnoses in low-income countries and areas with limited health care resources and personnel.25,29 AI can improve identification of infectious outbreaks, such as tuberculosis, malaria, dengue fever, and influenza.29,35-40 AI has been used to predict transmission patterns of the Zika virus and the current COVID-19 pandemic.41,42 Applications can stratify the risk of outbreaks based on multiple factors, including age, income, race, atypical geographic clusters, and seasonal factors like rainfall and temperature.35,36,38,43 AI has been used to assess morbidity and mortality, such as predicting disease severity with malaria and identifying treatment failures in tuberculosis.29
Finally, AI can dramatically impact health care due to processing large data sets or disconnected volumes of patient information—so-called big data.44-46 An example is the widespread use of electronic health records (EHRs) such as the Computerized Patient Record System used in Veteran Affairs medical centers (VAMCs). Much of patient information exists in written text: HCP notes, laboratory and radiology reports, medication records, etc. Natural language processing (NLP) allows platforms to sort through extensive volumes of data on complex patients at rates much faster than human capability, which has great potential to assist with diagnosis and treatment decisions.9
Medical literature is being produced at rates that exceed our ability to digest. More than 200,000 cancer-related articles were published in 2019 alone.14 NLP capabilities of AI have the potential to rapidly sort through this extensive medical literature and relate specific verbiage in patient records guiding therapy.46 IBM Watson, a supercomputer based on ML and NLP, demonstrates this concept with many potential applications, only some of which relate to health care.1,9 Watson has an oncology component to assimilate multiple aspects of patient care, including clinical notes, pathology results, radiograph findings, staging, and a tumor’s genetic profile. It coordinates these inputs from the EHR and mines medical literature and research databases to recommend treatment options.1,46 AI can assess and compile far greater patient data and therapeutic options than would be feasible by individual clinicians, thus providing customized patient care.47 Watson has partnered with numerous medical centers, including MD Anderson Cancer Center and Memorial Sloan Kettering Cancer Center, with variable success.44,47-49 While the full potential of Watson appears not yet realized, these AI-driven approaches will likely play an important role in leveraging the hidden value in the expanding volume of health care information.
Medical Specialty Applications
Radiology
Currently > 70% of FDA-approved AI medical devices are in the field of radiology.2 Most radiology departments have used AI-friendly digital imaging for years, such as the picture archiving and communication systems used by numerous health care systems, including VAMCs.2,15 Gray-scale images common in radiology lend themselves to standardization, although AI is not limited to black-and- white image interpretation.15
An abundance of literature describes plain radiograph interpretation using AI. One FDA-approved platform improved X-ray diagnosis of wrist fractures when used by emergency medicine clinicians.2,50 AI has been applied to chest X-ray (CXR) interpretation of many conditions, including pneumonia, tuberculosis, malignant lung lesions, and COVID-19.23,25,28,44,51-53 For example, Nam and colleagues suggested AI is better at diagnosing malignant pulmonary nodules from CXRs than are trained radiologists.28
In addition to plain radiographs, AI has been applied to many other imaging technologies, including ultrasounds, positron emission tomography, mammograms, computed tomography (CT), and magnetic resonance imaging (MRI).15,26,44,48,54-56 A large study demonstrated that ML platforms significantly reduced the time to diagnose intracranial hemorrhages on CT and identified subtle hemorrhages missed by radiologists.55 Other studies have claimed that AI programs may be better than radiologists in detecting cancer in screening mammograms, and 3 FDA-approved devices focus on mammogram interpretation.2,15,54,57 There is also great interest in MRI applications to detect and predict prognosis for breast cancer based on imaging findings.21,56
Aside from providing accurate diagnoses, other studies focus on AI radiograph interpretation to assist with patient screening, triage, improving time to final diagnosis, providing a rapid “second opinion,” and even monitoring disease progression and offering insights into prognosis.8,21,23,52,55,56,58 These features help in busy urban centers but may play an even greater role in areas with limited access to health care or trained specialists such as radiologists.52
Cardiology
Cardiology has the second highest number of FDA-approved AI applications.2 Many cardiology AI platforms involve image analysis, as described in several recent reviews.45,59,60 AI has been applied to echocardiography to measure ejection fractions, detect valvular disease, and assess heart failure from hypertrophic and restrictive cardiomyopathy and amyloidosis.45,48,59 Applications for cardiac CT scans and CT angiography have successfully quantified both calcified and noncalcified coronary artery plaques and lumen assessments, assessed myocardial perfusion, and performed coronary artery calcium scoring.45,59,60 Likewise, AI applications for cardiac MRI have been used to quantitate ejection fraction, large vessel flow assessment, and cardiac scar burden.45,59
For years ECG devices have provided interpretation with limited accuracy using preprogrammed parameters.48 However, the application of AI allows ECG interpretation on par with trained cardiologists. Numerous such AI applications exist, and 2 FDA-approved devices perform ECG interpretation.2,61-64 One of these devices incorporates an AI-powered stethoscope to detect atrial fibrillation and heart murmurs.65
Pathology
The advancement of whole slide imaging, wherein entire slides can be scanned and digitized at high speed and resolution, creates great potential for AI applications in pathology.12,24,32,33,66 A landmark study demonstrating the potential of AI for assessing whole slide imaging examined sentinel lymph node metastases in patients with breast cancer.22 Multiple algorithms in the study demonstrated that AI was equivalent or better than pathologists in detecting metastases, especially when the pathologists were time-constrained consistent with a normal working environment. Significantly, the most accurate and efficient diagnoses were achieved when the pathologist and AI interpretations were used together.22,33
AI has shown promise in diagnosing many other entities, including cancers of the prostate (including Gleason scoring), lung, colon, breast, and skin.11,12,24,27,32,67 In addition, AI has shown great potential in scoring biomarkers important for prognosis and treatment, such as immunohistochemistry (IHC) labeling of Ki-67 and PD-L1.32 Pathologists can have difficulty classifying certain tumors or determining the site of origin for metastases, often having to rely on IHC with limited success. The unique features of image analysis with AI have the potential to assist in classifying difficult tumors and identifying sites of origin for metastatic disease based on morphology alone.11
Oncology depends heavily on molecular pathology testing to dictate treatment options and determine prognosis. Preliminary studies suggest that AI interpretation alone has the potential to delineate whether certain molecular mutations are present in tumors from various sites.11,14,24,32 One study combined histology and genomic results for AI interpretation that improved prognostic predictions.68 In addition, AI analysis may have potential in predicting tumor recurrence or prognosis based on cellular features, as demonstrated for lung cancer and melanoma.67,69,70
Ophthalmology
AI applications for ophthalmology have focused on diabetic retinopathy, age-related macular degeneration, glaucoma, retinopathy of prematurity, age-related and congenital cataracts, and retinal vein occlusion.71-73 Diabetic retinopathy is a leading cause of blindness and has been studied by numerous platforms with good success, most having used color fundus photography.71,72 One study showed AI could diagnose diabetic retinopathy and diabetic macular edema with specificities similar to ophthalmologists.74 In 2018, the FDA approved the AI platform IDx-DR. This diagnostic system classifies retinal images and recommends referral for patients determined to have “more than mild diabetic retinopathy” and reexamination within a year for other patients.8,75 Significantly, the platform recommendations do not require confirmation by a clinician.8
AI has been applied to other modalities in ophthalmology such as optical coherence tomography (OCT) to diagnose retinal disease and to predict appropriate management of congenital cataracts.25,73,76 For example, an AI application using OCT has been demonstrated to match or exceed the accuracy of retinal experts in diagnosing and triaging patients with a variety of retinal pathologies, including patients needing urgent referrals.77
Dermatology
Multiple studies demonstrate AI performs at least equal to experienced dermatologists in differentiating selected skin lesions.78-81 For example, Esteva and colleagues demonstrated AI could differentiate keratinocyte carcinomas from benign seborrheic keratoses and malignant melanomas from benign nevi with accuracy equal to 21 board-certified dermatologists.78
AI is applicable to various imaging procedures common to dermatology, such as dermoscopy, very high-frequency ultrasound, and reflectance confocal microscopy.82 Several studies have demonstrated that AI interpretation compared favorably to dermatologists evaluating dermoscopy to assess melanocytic lesions.78-81,83
A limitation in these studies is that they differentiate only a few diagnoses.82 Furthermore, dermatologists have sensory input such as touch and visual examination under various conditions, something AI has yet to replicate.15,34,84 Also, most AI devices use no or limited clinical information.81 Dermatologists can recognize rarer conditions for which AI models may have had limited or no training.34 Nevertheless, a recent study assessed AI for the diagnosis of 134 separate skin disorders with promising results, including providing diagnoses with accuracy comparable to that of dermatologists and providing accurate treatment strategies.84 As Topol points out, most skin lesions are diagnosed in the primary care setting where AI can have a greater impact when used in conjunction with the clinical impression, especially where specialists are in limited supply.48,78
Finally, dermatology lends itself to using portable or smartphone applications (apps) wherein the user can photograph a lesion for analysis by AI algorithms to assess the need for further evaluation or make treatment recommendations.34,84,85 Although results from currently available apps are not encouraging, they may play a greater role as the technology advances.34,85
Oncology
Applications of AI in oncology include predicting prognosis for patients with cancer based on histologic and/or genetic information.14,68,86 Programs can predict the risk of complications before and recurrence risks after surgery for malignancies.44,87-89 AI can also assist in treatment planning and predict treatment failure with radiation therapy.90,91
AI has great potential in processing the large volumes of patient data in cancer genomics. Next-generation sequencing has allowed for the identification of millions of DNA sequences in a single tumor to detect genetic anomalies.92 Thousands of mutations can be found in individual tumor samples, and processing this information and determining its significance can be beyond human capability.14 We know little about the effects of various mutation combinations, and most tumors have a heterogeneous molecular profile among different cell populations.14,93 The presence or absence of various mutations can have diagnostic, prognostic, and therapeutic implications.93 AI has great potential to sort through these complex data and identify actionable findings.
More than 200,000 cancer-related articles were published in 2019, and publications in the field of cancer genomics are increasing exponentially.14,92,93 Patel and colleagues assessed the utility of IBM Watson for Genomics against results from a molecular tumor board.93 Watson for Genomics identified potentially significant mutations not identified by the tumor board in 32% of patients. Most mutations were related to new clinical trials not yet added to the tumor board watch list, demonstrating the role AI will have in processing the large volume of genetic data required to deliver personalized medicine moving forward.
Gastroenterology
AI has shown promise in predicting risk or outcomes based on clinical parameters in various common gastroenterology problems, including gastric reflux, acute pancreatitis, gastrointestinal bleeding, celiac disease, and inflammatory bowel disease.94,95 AI endoscopic analysis has demonstrated potential in assessing Barrett’s esophagus, gastric Helicobacter pylori infections, gastric atrophy, and gastric intestinal metaplasia.95 Applications have been used to assess esophageal, gastric, and colonic malignancies, including depth of invasion based on endoscopic images.95 Finally, studies have evaluated AI to assess small colon polyps during colonoscopy, including differentiating benign and premalignant polyps with success comparable to gastroenterologists.94,95 AI has been shown to increase the speed and accuracy of gastroenterologists in detecting small polyps during colonoscopy.48 In a prospective randomized study, colonoscopies performed using an AI device identified significantly more small adenomatous polyps than colonoscopies without AI.96
Neurology
It has been suggested that AI technologies are well suited for application in neurology due to the subtle presentation of many neurologic diseases.16 Viz LVO, the first CMS-approved AI reimbursement for the diagnosis of strokes, analyzes CTs to detect early ischemic strokes and alerts the medical team, thus shortening time to treatment.3,97 Many other AI platforms are in use or development that use CT and MRI for the early detection of strokes as well as for treatment and prognosis.9,97
AI technologies have been applied to neurodegenerative diseases, such as Alzheimer and Parkinson diseases.16,98 For example, several studies have evaluated patient movements in Parkinson disease for both early diagnosis and to assess response to treatment.98 These evaluations included assessment with both external cameras as well as wearable devices and smartphone apps.
AI has also been applied to seizure disorders, attempting to determine seizure type, localize the area of seizure onset, and address the challenges of identifying seizures in neonates.99,100 Other potential applications range from early detection and prognosis predictions for cases of multiple sclerosis to restoring movement in paralysis from a variety of conditions such as spinal cord injury.9,101,102
Mental Health
Due to the interactive nature of mental health care, the field has been slower to develop AI applications.18 With heavy reliance on textual information (eg, clinic notes, mood rating scales, and documentation of conversations), successful AI applications in this field will likely rely heavily on NLP.18 However, studies investigating the application of AI to mental health have also incorporated data such as brain imaging, smartphone monitoring, and social media platforms, such as Facebook and Twitter.18,103,104
The risk of suicide is higher in veteran patients, and ML algorithms have had limited success in predicting suicide risk in both veteran and nonveteran populations.104-106 While early models have low positive predictive values and low sensitivities, they still promise to be a useful tool in conjunction with traditional risk assessments.106 Kessler and colleagues suggest that combining multiple rather than single ML algorithms might lead to greater success.105,106
AI may assist in diagnosing other mental health disorders, including major depressive disorder, attention deficit hyperactivity disorder (ADHD), schizophrenia, posttraumatic stress disorder, and Alzheimer disease.103,104,107 These investigations are in the early stages with limited clinical applicability. However, 2 AI applications awaiting FDA approval relate to ADHD and opioid use.2 Furthermore, potential exists for AI to not only assist with prevention and diagnosis of ADHD, but also to identify optimal treatment options.2,103
General and Personalized Medicine
Additional AI applications include diagnosing patients with suspected sepsis, measuring liver iron concentrations, predicting hospital mortality at the time of admission, and more.2,108,109 AI can guide end-of-life decisions such as resuscitation status or whether to initiate mechanical ventilation.48
AI-driven smartphone apps can be beneficial to both patients and clinicians. Examples include predicting nonadherence to anticoagulation therapy, monitoring heart rhythms for atrial fibrillation or signs of hyperkalemia in patients with renal failure, and improving outcomes for patients with diabetes mellitus by decreasing glycemic variability and reducing hypoglycemia.8,48,110,111 The potential for AI applications to health care and personalized medicine are almost limitless.
Discussion
With ever-increasing expectations for all health care sectors to deliver timely, fiscally-responsible, high-quality health care, AI has the potential to have numerous impacts. AI can improve diagnostic accuracy while limiting errors and impact patient safety such as assisting with prescription delivery.8,9,34 It can screen and triage patients, alerting clinicians to those needing more urgent evaluation.8,23,77,97 AI also may increase a clinician’s efficiency and speed to render a diagnosis.12,13,55,97 AI can provide a rapid second opinion, an ability especially beneficial in underserved areas with shortages of specialists.23,25,26,29,34 Similarly, AI may decrease the inter- and intraobserver variability common in many medical specialties.12,27,45 AI applications can also monitor disease progression, identifying patients at greatest risk, and provide information for prognosis.21,23,56,58 Finally, as described with applications using IBM Watson, AI can allow for an integrated approach to health care that is currently lacking.
We have described many reports suggesting AI can render diagnoses as well as or better than experienced clinicians, and speculation exists that AI will replace many roles currently performed by health care practitioners.9,26 However, most studies demonstrate that AI’s diagnostic benefits are best realized when used to supplement a clinician’s impression.8,22,30,33,52,54,56,69,84 AI is not likely to replace humans in health care in the foreseeable future. The technology can be likened to the impact of CT scans developed in the 1970s in neurology. Prior to such detailed imaging, neurologists spent extensive time performing detailed physicals to render diagnoses and locate lesions before surgery. There was mistrust of this new technology and concern that CT scans would eliminate the need for neurologists.112 On the contrary, neurology is alive and well, frequently being augmented by the technologies once speculated to replace it.
Commercial AI health care platforms represented a $2 billion industry in 2018 and are growing rapidly each year.13,32 Many AI products are offered ready for implementation for various tasks, including diagnostics, patient management, and improved efficiency. Others will likely be provided as templates suitable for modification to meet the specific needs of the facility, practice, or specialty for its patient population.
AI Risks and Limitations
AI has several risks and limitations. Although there is progress in explainable AI, at times we still struggle to understand how the output provided by machine learning algorithms was created.44,48 The many layers associated with deep learning self-determine the criteria to reach its conclusion, and these criteria can continually evolve. The parameters of deep learning are not preprogrammed, and there are too many individual data points to be extrapolated or deconvoluted for evaluation at our current level of knowledge.26,51 These apparent lack of constraints cause concern for patient safety and suggest that greater validation and continued scrutiny of validity is required.8,48 Efforts are underway to create explainable AI programs to make their processes more transparent, but such clarification is limited presently.14,26,48,77
Another challenge of AI is determining the amount of training data required to function optimally. Also, if the output describes multiple variables or diagnoses, are each equally valid?113 Furthermore, many AI applications look for a specific process, such as cancer diagnoses on CXRs. However, how coexisting conditions like cardiomegaly, emphysema, pneumonia, etc, seen on CXRs will affect the diagnosis needs to be considered.51,52 Zech and colleagues provide the example that diagnoses for pneumothorax are frequently rendered on CXRs with chest tubes in place.51 They suggest that CNNs may develop a bias toward diagnosing pneumothorax when chest tubes are present. Many current studies approach an issue in isolation, a situation not realistic in real-world clinical practice.26
Most studies on AI have been retrospective, and frequently data used to train the program are preselected.13,26 The data are typically validated on available databases rather than actual patients in the clinical setting, limiting confidence in the validity of the AI output when applied to real-world situations. Currently, fewer than 12 prospective trials had been published comparing AI with traditional clinical care.13,114 Randomized prospective clinical trials are even fewer, with none currently reported from the United States.13,114 The results from several studies have been shown to diminish when repeated prospectively.114
The FDA has created a new category known as Software as a Medical Device and has a Digital Health Innovation Action Plan to regulate AI platforms. Still, the process of AI regulation is of necessity different from traditional approval processes and is continually evolving.8 The FDA approval process cannot account for the fact that the program’s parameters may continually evolve or adapt.2
Guidelines for investigating and reporting AI research with its unique attributes are being developed. Examples include the TRIPOD-ML statement and others.49,115 In September 2020, 2 publications addressed the paucity of gold-standard randomized clinical trials in clinical AI applications.116,117 The SPIRIT-AI statement expands on the original SPIRIT statement published in 2013 to guide minimal reporting standards for AI clinical trial protocols to promote transparency of design and methodology.116 Similarly, the CONSORT-AI extension, stemming from the original CONSORT statement in 1996, aims to ensure quality reporting of randomized controlled trials in AI.117
Another risk with AI is that while an individual physician making a mistake may adversely affect 1 patient, a single mistake in an AI algorithm could potentially affect thousands of patients.48 Also, AI programs developed for patient populations at a facility may not translate to another. Referred to as overfitting, this phenomenon relates to selection bias in training data sets.15,34,49,51,52 Studies have shown that programs that underrepresent certain group characteristics such as age, sex, or race may be less effective when applied to a population in which these characteristics have differing representations.8,48,49 This problem of underrepresentation has been demonstrated in programs interpreting pathology slides, radiographs, and skin lesions.15,32,51
Admittedly, most of these challenges are not specific to AI and existed in health care previously. Physicians make mistakes, treatments are sometimes used without adequate prospective studies, and medications are given without understanding their mechanism of action, much like AI-facilitated processes reach a conclusion that cannot be fully explained.48
Conclusions
The view that AI will dramatically impact health care in the coming years will likely prove true. However, much work is needed, especially because of the paucity of prospective clinical trials as has been historically required in medical research. Any concern that AI will replace HCPs seems unwarranted. Early studies suggest that even AI programs that appear to exceed human interpretation perform best when working in cooperation with and oversight from clinicians. AI’s greatest potential appears to be its ability to augment care from health professionals, improving efficiency and accuracy, and should be anticipated with enthusiasm as the field moves forward at an exponential rate.
Acknowledgments
The authors thank Makenna G. Thomas for proofreading and review of the manuscript. This material is the result of work supported with resources and the use of facilities at the James A. Haley Veterans’ Hospital. This research has been approved by the James A. Haley Veteran’s Hospital Office of Communications and Media.
1. Bini SA. Artificial intelligence, machine learning, deep learning, and cognitive computing: what do these terms mean and how will they impact health care? J Arthroplasty. 2018;33(8):2358-2361. doi:10.1016/j.arth.2018.02.067
2. Benjamens S, Dhunnoo P, Meskó B. The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database. NPJ Digit Med. 2020;3:118. doi:10.1038/s41746-020-00324-0
3. Viz. AI powered synchronized stroke care. Accessed September 15, 2021. https://www.viz.ai/ischemic-stroke
4. Buchanan M. The law of accelerating returns. Nat Phys. 2008;4(7):507. doi:10.1038/nphys1010
5. IBM Watson Health computes a pair of new solutions to improve healthcare data and security. Published September 10, 2015. Accessed October 21, 2020. https://www.techrepublic.com/article/ibm-watson-health-computes-a-pair-of-new-solutions-to-improve-healthcare-data-and-security
6. Borkowski AA, Kardani A, Mastorides SM, Thomas LB. Warfarin pharmacogenomics: recommendations with available patented clinical technologies. Recent Pat Biotechnol. 2014;8(2):110-115. doi:10.2174/1872208309666140904112003
7. Washington University in St. Louis. Warfarin dosing. Accessed September 15, 2021. http://www.warfarindosing.org/Source/Home.aspx
8. He J, Baxter SL, Xu J, Xu J, Zhou X, Zhang K. The practical implementation of artificial intelligence technologies in medicine. Nat Med. 2019;25(1):30-36. doi:10.1038/s41591-018-0307-0
9. Jiang F, Jiang Y, Zhi H, et al. Artificial intelligence in healthcare: past, present and future. Stroke Vasc Neurol. 2017;2(4):230-243. Published 2017 Jun 21. doi:10.1136/svn-2017-000101
10. Johnson KW, Torres Soto J, Glicksberg BS, et al. Artificial intelligence in cardiology. J Am Coll Cardiol. 2018;71(23):2668-2679. doi:10.1016/j.jacc.2018.03.521
11. Borkowski AA, Wilson CP, Borkowski SA, et al. Comparing artificial intelligence platforms for histopathologic cancer diagnosis. Fed Pract. 2019;36(10):456-463.
12. Cruz-Roa A, Gilmore H, Basavanhally A, et al. High-throughput adaptive sampling for whole-slide histopathology image analysis (HASHI) via convolutional neural networks: application to invasive breast cancer detection. PLoS One. 2018;13(5):e0196828. Published 2018 May 24. doi:10.1371/journal.pone.0196828
13. Nagendran M, Chen Y, Lovejoy CA, et al. Artificial intelligence versus clinicians: systematic review of design, reporting standards, and claims of deep learning studies. BMJ. 2020;368:m689. Published 2020 Mar 25. doi:10.1136/bmj.m689
14. Shimizu H, Nakayama KI. Artificial intelligence in oncology. Cancer Sci. 2020;111(5):1452-1460. doi:10.1111/cas.14377
15. Talebi-Liasi F, Markowitz O. Is artificial intelligence going to replace dermatologists?. Cutis. 2020;105(1):28-31.
16. Valliani AA, Ranti D, Oermann EK. Deep learning and neurology: a systematic review. Neurol Ther. 2019;8(2):351-365. doi:10.1007/s40120-019-00153-8
17. LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521(7553):436-444. doi:10.1038/nature14539
18. Graham S, Depp C, Lee EE, et al. Artificial intelligence for mental health and mental illnesses: an overview. Curr Psychiatry Rep. 2019;21(11):116. Published 2019 Nov 7. doi:10.1007/s11920-019-1094-0
19. Golas SB, Shibahara T, Agboola S, et al. A machine learning model to predict the risk of 30-day readmissions in patients with heart failure: a retrospective analysis of electronic medical records data. BMC Med Inform Decis Mak. 2018;18(1):44. Published 2018 Jun 22. doi:10.1186/s12911-018-0620-z
20. Mortazavi BJ, Downing NS, Bucholz EM, et al. Analysis of machine learning techniques for heart failure readmissions. Circ Cardiovasc Qual Outcomes. 2016;9(6):629-640. doi:10.1161/CIRCOUTCOMES.116.003039
21. Meyer-Bäse A, Morra L, Meyer-Bäse U, Pinker K. Current status and future perspectives of artificial intelligence in magnetic resonance breast imaging. Contrast Media Mol Imaging. 2020;2020:6805710. Published 2020 Aug 28. doi:10.1155/2020/6805710
22. Ehteshami Bejnordi B, Veta M, Johannes van Diest P, et al. Diagnostic assessment of deep learning algorithms for detection of lymph node metastases in women with breast cancer. JAMA. 2017;318(22):2199-2210. doi:10.1001/jama.2017.14585
23. Borkowski AA, Viswanadhan NA, Thomas LB, Guzman RD, Deland LA, Mastorides SM. Using artificial intelligence for COVID-19 chest X-ray diagnosis. Fed Pract. 2020;37(9):398-404. doi:10.12788/fp.0045
24. Coudray N, Ocampo PS, Sakellaropoulos T, et al. Classification and mutation prediction from non-small cell lung cancer histopathology images using deep learning. Nat Med. 2018;24(10):1559-1567. doi:10.1038/s41591-018-0177-5
25. Kermany DS, Goldbaum M, Cai W, et al. Identifying medical diagnoses and treatable diseases by image-based deep learning. Cell. 2018;172(5):1122-1131.e9. doi:10.1016/j.cell.2018.02.010
26. Liu X, Faes L, Kale AU, et al. A comparison of deep learning performance against health-care professionals in detecting diseases from medical imaging: a systematic review and meta-analysis. Lancet Digit Health. 2019;1(6):e271-e297. doi:10.1016/S2589-7500(19)30123-2
27. Nagpal K, Foote D, Liu Y, et al. Development and validation of a deep learning algorithm for improving Gleason scoring of prostate cancer [published correction appears in NPJ Digit Med. 2019 Nov 19;2:113]. NPJ Digit Med. 2019;2:48. Published 2019 Jun 7. doi:10.1038/s41746-019-0112-2
28. Nam JG, Park S, Hwang EJ, et al. Development and validation of deep learning-based automatic detection algorithm for malignant pulmonary nodules on chest radiographs. Radiology. 2019;290(1):218-228. doi:10.1148/radiol.2018180237
29. Schwalbe N, Wahl B. Artificial intelligence and the future of global health. Lancet. 2020;395(10236):1579-1586. doi:10.1016/S0140-6736(20)30226-9
30. Bai HX, Wang R, Xiong Z, et al. Artificial intelligence augmentation of radiologist performance in distinguishing COVID-19 from pneumonia of other origin at chest CT [published correction appears in Radiology. 2021 Apr;299(1):E225]. Radiology. 2020;296(3):E156-E165. doi:10.1148/radiol.2020201491
31. Li L, Qin L, Xu Z, et al. Using artificial intelligence to detect COVID-19 and community-acquired pneumonia based on pulmonary CT: evaluation of the diagnostic accuracy. Radiology. 2020;296(2):E65-E71. doi:10.1148/radiol.2020200905
32. Serag A, Ion-Margineanu A, Qureshi H, et al. Translational AI and deep learning in diagnostic pathology. Front Med (Lausanne). 2019;6:185. Published 2019 Oct 1. doi:10.3389/fmed.2019.00185

33. Wang D, Khosla A, Gargeya R, Irshad H, Beck AH. Deep learning for identifying metastatic breast cancer. ArXiv. 2016 June 18:arXiv:1606.05718v1. Published online June 18, 2016. Accessed September 15, 2021. http://arxiv.org/abs/1606.05718
34. Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatology Dermatol Surg. 2019;23(2):57-60. doi:10.4103/jdds.jdds_19_19
35. Mollalo A, Mao L, Rashidi P, Glass GE. A GIS-based artificial neural network model for spatial distribution of tuberculosis across the continental United States. Int J Environ Res Public Health. 2019;16(1):157. Published 2019 Jan 8. doi:10.3390/ijerph16010157
36. Haddawy P, Hasan AHMI, Kasantikul R, et al. Spatiotemporal Bayesian networks for malaria prediction. Artif Intell Med. 2018;84:127-138. doi:10.1016/j.artmed.2017.12.002
37. Laureano-Rosario AE, Duncan AP, Mendez-Lazaro PA, et al. Application of artificial neural networks for dengue fever outbreak predictions in the northwest coast of Yucatan, Mexico and San Juan, Puerto Rico. Trop Med Infect Dis. 2018;3(1):5. Published 2018 Jan 5. doi:10.3390/tropicalmed3010005
38. Buczak AL, Koshute PT, Babin SM, Feighner BH, Lewis SH. A data-driven epidemiological prediction method for dengue outbreaks using local and remote sensing data. BMC Med Inform Decis Mak. 2012;12:124. Published 2012 Nov 5. doi:10.1186/1472-6947-12-124
39. Scavuzzo JM, Trucco F, Espinosa M, et al. Modeling dengue vector population using remotely sensed data and machine learning. Acta Trop. 2018;185:167-175. doi:10.1016/j.actatropica.2018.05.003
40. Xue H, Bai Y, Hu H, Liang H. Influenza activity surveillance based on multiple regression model and artificial neural network. IEEE Access. 2018;6:563-575. doi:10.1109/ACCESS.2017.2771798
41. Jiang D, Hao M, Ding F, Fu J, Li M. Mapping the transmission risk of Zika virus using machine learning models. Acta Trop. 2018;185:391-399. doi:10.1016/j.actatropica.2018.06.021
42. Bragazzi NL, Dai H, Damiani G, Behzadifar M, Martini M, Wu J. How big data and artificial intelligence can help better manage the COVID-19 pandemic. Int J Environ Res Public Health. 2020;17(9):3176. Published 2020 May 2. doi:10.3390/ijerph17093176
43. Lake IR, Colón-González FJ, Barker GC, Morbey RA, Smith GE, Elliot AJ. Machine learning to refine decision making within a syndromic surveillance service. BMC Public Health. 2019;19(1):559. Published 2019 May 14. doi:10.1186/s12889-019-6916-9
44. Khan OF, Bebb G, Alimohamed NA. Artificial intelligence in medicine: what oncologists need to know about its potential-and its limitations. Oncol Exch. 2017;16(4):8-13. Accessed September 1, 2021. http://www.oncologyex.com/pdf/vol16_no4/feature_khan-ai.pdf
45. Badano LP, Keller DM, Muraru D, Torlasco C, Parati G. Artificial intelligence and cardiovascular imaging: A win-win combination. Anatol J Cardiol. 2020;24(4):214-223. doi:10.14744/AnatolJCardiol.2020.94491
46. Murdoch TB, Detsky AS. The inevitable application of big data to health care. JAMA. 2013;309(13):1351-1352. doi:10.1001/jama.2013.393
47. Greatbatch O, Garrett A, Snape K. The impact of artificial intelligence on the current and future practice of clinical cancer genomics. Genet Res (Camb). 2019;101:e9. Published 2019 Oct 31. doi:10.1017/S0016672319000089
48. Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44-56. doi:10.1038/s41591-018-0300-7
49. Vollmer S, Mateen BA, Bohner G, et al. Machine learning and artificial intelligence research for patient benefit: 20 critical questions on transparency, replicability, ethics, and effectiveness [published correction appears in BMJ. 2020 Apr 1;369:m1312]. BMJ. 2020;368:l6927. Published 2020 Mar 20. doi:10.1136/bmj.l6927
50. Lindsey R, Daluiski A, Chopra S, et al. Deep neural network improves fracture detection by clinicians. Proc Natl Acad Sci U S A. 2018;115(45):11591-11596. doi:10.1073/pnas.1806905115
51. Zech JR, Badgeley MA, Liu M, Costa AB, Titano JJ, Oermann EK. Variable generalization performance of a deep learning model to detect pneumonia in chest radiographs: a cross-sectional study. PLoS Med. 2018;15(11):e1002683. doi:10.1371/journal.pmed.1002683
52. Lakhani P, Sundaram B. Deep learning at chest radiography: automated classification of pulmonary tuberculosis by using convolutional neural networks. Radiology. 2017;284(2):574-582. doi:10.1148/radiol.2017162326
53. Rajpurkar P, Joshi A, Pareek A, et al. CheXpedition: investigating generalization challenges for translation of chest x-ray algorithms to the clinical setting. ArXiv. 2020 Feb 26:arXiv:2002.11379v2. Revised March 11, 2020. Accessed September 15, 2021. http://arxiv.org/abs/2002.11379
54. Salim M, Wåhlin E, Dembrower K, et al. External evaluation of 3 commercial artificial intelligence algorithms for independent assessment of screening mammograms. JAMA Oncol. 2020;6(10):1581-1588. doi:10.1001/jamaoncol.2020.3321
55. Arbabshirani MR, Fornwalt BK, Mongelluzzo GJ, et al. Advanced machine learning in action: identification of intracranial hemorrhage on computed tomography scans of the head with clinical workflow integration. NPJ Digit Med. 2018;1:9. doi:10.1038/s41746-017-0015-z
56. Sheth D, Giger ML. Artificial intelligence in the interpretation of breast cancer on MRI. J Magn Reson Imaging. 2020;51(5):1310-1324. doi:10.1002/jmri.26878
57. McKinney SM, Sieniek M, Godbole V, et al. International evaluation of an AI system for breast cancer screening. Nature. 2020;577(7788):89-94. doi:10.1038/s41586-019-1799-6
58. Booth AL, Abels E, McCaffrey P. Development of a prognostic model for mortality in COVID-19 infection using machine learning. Mod Pathol. 2021;34(3):522-531. doi:10.1038/s41379-020-00700-x
59. Xu B, Kocyigit D, Grimm R, Griffin BP, Cheng F. Applications of artificial intelligence in multimodality cardiovascular imaging: a state-of-the-art review. Prog Cardiovasc Dis. 2020;63(3):367-376. doi:10.1016/j.pcad.2020.03.003
60. Dey D, Slomka PJ, Leeson P, et al. Artificial intelligence in cardiovascular imaging: JACC state-of-the-art review. J Am Coll Cardiol. 2019;73(11):1317-1335. doi:10.1016/j.jacc.2018.12.054
61. Carewell Health. AI powered ECG diagnosis solutions. Accessed November 2, 2020. https://www.carewellhealth.com/products_aiecg.html
62. Strodthoff N, Strodthoff C. Detecting and interpreting myocardial infarction using fully convolutional neural networks. Physiol Meas. 2019;40(1):015001. doi:10.1088/1361-6579/aaf34d
63. Hannun AY, Rajpurkar P, Haghpanahi M, et al. Cardiologist-level arrhythmia detection and classification in ambulatory electrocardiograms using a deep neural network. Nat Med. 2019;25(1):65-69. doi:10.1038/s41591-018-0268-3
64. Kwon JM, Jeon KH, Kim HM, et al. Comparing the performance of artificial intelligence and conventional diagnosis criteria for detecting left ventricular hypertrophy using electrocardiography. Europace. 2020;22(3):412-419. doi:10.1093/europace/euz324

65. Eko. FDA clears Eko’s AFib and heart murmur detection algorithms, making it the first AI-powered stethoscope to screen for serious heart conditions [press release]. Published January 28, 2020. Accessed September 15, 2021. https://www.businesswire.com/news/home/20200128005232/en/FDA-Clears-Eko’s-AFib-and-Heart-Murmur-Detection-Algorithms-Making-It-the-First-AI-Powered-Stethoscope-to-Screen-for-Serious-Heart-Conditions
66. Cruz-Roa A, Gilmore H, Basavanhally A, et al. Accurate and reproducible invasive breast cancer detection in whole-slide images: a deep learning approach for quantifying tumor extent. Sci Rep. 2017;7:46450. doi:10.1038/srep46450
67. Acs B, Rantalainen M, Hartman J. Artificial intelligence as the next step towards precision pathology. J Intern Med. 2020;288(1):62-81. doi:10.1111/joim.13030
68. Mobadersany P, Yousefi S, Amgad M, et al. Predicting cancer outcomes from histology and genomics using convolutional networks. Proc Natl Acad Sci U S A. 2018;115(13):E2970-E2979. doi:10.1073/pnas.1717139115
69. Wang X, Janowczyk A, Zhou Y, et al. Prediction of recurrence in early stage non-small cell lung cancer using computer extracted nuclear features from digital H&E images. Sci Rep. 2017;7:13543. doi:10.1038/s41598-017-13773-7
70. Kulkarni PM, Robinson EJ, Pradhan JS, et al. Deep learning based on standard H&E images of primary melanoma tumors identifies patients at risk for visceral recurrence and death. Clin Cancer Res. 2020;26(5):1126-1134. doi:10.1158/1078-0432.CCR-19-1495
71. Du XL, Li WB, Hu BJ. Application of artificial intelligence in ophthalmology. Int J Ophthalmol. 2018;11(9):1555-1561. doi:10.18240/ijo.2018.09.21
72. Gunasekeran DV, Wong TY. Artificial intelligence in ophthalmology in 2020: a technology on the cusp for translation and implementation. Asia Pac J Ophthalmol (Phila). 2020;9(2):61-66. doi:10.1097/01.APO.0000656984.56467.2c
73. Ting DSW, Pasquale LR, Peng L, et al. Artificial intelligence and deep learning in ophthalmology. Br J Ophthalmol. 2019;103(2):167-175. doi:10.1136/bjophthalmol-2018-313173
74. Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-2410. doi:10.1001/jama.2016.17216
75. US Food and Drug Administration. FDA permits marketing of artificial intelligence-based device to detect certain diabetes-related eye problems [press release]. Published April 11, 2018. Accessed September 15, 2021. https://www.fda.gov/news-events/press-announcements/fda-permits-marketing-artificial-intelligence-based-device-detect-certain-diabetes-related-eye
76. Long E, Chen J, Wu X, et al. Artificial intelligence manages congenital cataract with individualized prediction and telehealth computing. NPJ Digit Med. 2020;3:112. doi:10.1038/s41746-020-00319-x
77. De Fauw J, Ledsam JR, Romera-Paredes B, et al. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat Med. 2018;24(9):1342-1350. doi:10.1038/s41591-018-0107-6
78. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118. doi:10.1038/nature21056
79. Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
80. Brinker TJ, Hekler A, Enk AH, et al. A convolutional neural network trained with dermoscopic images performed on par with 145 dermatologists in a clinical melanoma image classification task. Eur J Cancer. 2019;111:148-154. doi:10.1016/j.ejca.2019.02.005
81. Haenssle HA, Fink C, Schneiderbauer R, et al. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists. Ann Oncol. 2018;29(8):1836-1842. doi:10.1093/annonc/mdy166
82. Li CX, Shen CB, Xue K, et al. Artificial intelligence in dermatology: past, present, and future. Chin Med J (Engl). 2019;132(17):2017-2020. doi:10.1097/CM9.0000000000000372
83. Tschandl P, Codella N, Akay BN, et al. Comparison of the accuracy of human readers versus machine-learning algorithms for pigmented skin lesion classification: an open, web-based, international, diagnostic study. Lancet Oncol. 2019;20(7):938-947. doi:10.1016/S1470-2045(19)30333-X
84. Han SS, Park I, Eun Chang SE, et al. Augmented intelligence dermatology: deep neural networks empower medical professionals in diagnosing skin cancer and predicting treatment options for 134 skin disorders. J Invest Dermatol. 2020;140(9):1753-1761. doi:10.1016/j.jid.2020.01.019
85. Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies [published correction appears in BMJ. 2020 Feb 25;368:m645]. BMJ. 2020;368:m127. Published 2020 Feb 10. doi:10.1136/bmj.m127
86. Chen YC, Ke WC, Chiu HW. Risk classification of cancer survival using ANN with gene expression data from multiple laboratories. Comput Biol Med. 2014;48:1-7. doi:10.1016/j.compbiomed.2014.02.006
87. Kim W, Kim KS, Lee JE, et al. Development of novel breast cancer recurrence prediction model using support vector machine. J Breast Cancer. 2012;15(2):230-238. doi:10.4048/jbc.2012.15.2.230
88. Merath K, Hyer JM, Mehta R, et al. Use of machine learning for prediction of patient risk of postoperative complications after liver, pancreatic, and colorectal surgery. J Gastrointest Surg. 2020;24(8):1843-1851. doi:10.1007/s11605-019-04338-2
89. Santos-García G, Varela G, Novoa N, Jiménez MF. Prediction of postoperative morbidity after lung resection using an artificial neural network ensemble. Artif Intell Med. 2004;30(1):61-69. doi:10.1016/S0933-3657(03)00059-9
90. Ibragimov B, Xing L. Segmentation of organs-at-risks in head and neck CT images using convolutional neural networks. Med Phys. 2017;44(2):547-557. doi:10.1002/mp.12045
91. Lou B, Doken S, Zhuang T, et al. An image-based deep learning framework for individualizing radiotherapy dose. Lancet Digit Health. 2019;1(3):e136-e147. doi:10.1016/S2589-7500(19)30058-5
92. Xu J, Yang P, Xue S, et al. Translating cancer genomics into precision medicine with artificial intelligence: applications, challenges and future perspectives. Hum Genet. 2019;138(2):109-124. doi:10.1007/s00439-019-01970-5
93. Patel NM, Michelini VV, Snell JM, et al. Enhancing next‐generation sequencing‐guided cancer care through cognitive computing. Oncologist. 2018;23(2):179-185. doi:10.1634/theoncologist.2017-0170
94. Le Berre C, Sandborn WJ, Aridhi S, et al. Application of artificial intelligence to gastroenterology and hepatology. Gastroenterology. 2020;158(1):76-94.e2. doi:10.1053/j.gastro.2019.08.058
95. Yang YJ, Bang CS. Application of artificial intelligence in gastroenterology. World J Gastroenterol. 2019;25(14):1666-1683. doi:10.3748/wjg.v25.i14.1666
96. Wang P, Berzin TM, Glissen Brown JR, et al. Real-time automatic detection system increases colonoscopic polyp and adenoma detection rates: a prospective randomised controlled study. Gut. 2019;68(10):1813-1819. doi:10.1136/gutjnl-2018-317500

97. Gupta R, Krishnam SP, Schaefer PW, Lev MH, Gonzalez RG. An East Coast perspective on artificial intelligence and machine learning: part 2: ischemic stroke imaging and triage. Neuroimaging Clin N Am. 2020;30(4):467-478. doi:10.1016/j.nic.2020.08.002
98. Beli M, Bobi V, Badža M, Šolaja N, Duri-Jovii M, Kosti VS. Artificial intelligence for assisting diagnostics and assessment of Parkinson’s disease—a review. Clin Neurol Neurosurg. 2019;184:105442. doi:10.1016/j.clineuro.2019.105442
99. An S, Kang C, Lee HW. Artificial intelligence and computational approaches for epilepsy. J Epilepsy Res. 2020;10(1):8-17. doi:10.14581/jer.20003
100. Pavel AM, Rennie JM, de Vries LS, et al. A machine-learning algorithm for neonatal seizure recognition: a multicentre, randomised, controlled trial. Lancet Child Adolesc Health. 2020;4(10):740-749. doi:10.1016/S2352-4642(20)30239-X
101. Afzal HMR, Luo S, Ramadan S, Lechner-Scott J. The emerging role of artificial intelligence in multiple sclerosis imaging [published online ahead of print, 2020 Oct 28]. Mult Scler. 2020;1352458520966298. doi:10.1177/1352458520966298
102. Bouton CE. Restoring movement in paralysis with a bioelectronic neural bypass approach: current state and future directions. Cold Spring Harb Perspect Med. 2019;9(11):a034306. doi:10.1101/cshperspect.a034306
103. Durstewitz D, Koppe G, Meyer-Lindenberg A. Deep neural networks in psychiatry. Mol Psychiatry. 2019;24(11):1583-1598. doi:10.1038/s41380-019-0365-9
104. Fonseka TM, Bhat V, Kennedy SH. The utility of artificial intelligence in suicide risk prediction and the management of suicidal behaviors. Aust N Z J Psychiatry. 2019;53(10):954-964. doi:10.1177/0004867419864428
105. Kessler RC, Hwang I, Hoffmire CA, et al. Developing a practical suicide risk prediction model for targeting high-risk patients in the Veterans Health Administration. Int J Methods Psychiatr Res. 2017;26(3):e1575. doi:10.1002/mpr.1575
106. Kessler RC, Bauer MS, Bishop TM, et al. Using administrative data to predict suicide after psychiatric hospitalization in the Veterans Health Administration System. Front Psychiatry. 2020;11:390. doi:10.3389/fpsyt.2020.00390
107. Kessler RC, van Loo HM, Wardenaar KJ, et al. Testing a machine-learning algorithm to predict the persistence and severity of major depressive disorder from baseline self-reports. Mol Psychiatry. 2016;21(10):1366-1371. doi:10.1038/mp.2015.198
108. Horng S, Sontag DA, Halpern Y, Jernite Y, Shapiro NI, Nathanson LA. Creating an automated trigger for sepsis clinical decision support at emergency department triage using machine learning. PLoS One. 2017;12(4):e0174708. doi:10.1371/journal.pone.0174708
109. Soffer S, Klang E, Barash Y, Grossman E, Zimlichman E. Predicting in-hospital mortality at admission to the medical ward: a big-data machine learning model. Am J Med. 2021;134(2):227-234.e4. doi:10.1016/j.amjmed.2020.07.014
110. Labovitz DL, Shafner L, Reyes Gil M, Virmani D, Hanina A. Using artificial intelligence to reduce the risk of nonadherence in patients on anticoagulation therapy. Stroke. 2017;48(5):1416-1419. doi:10.1161/STROKEAHA.116.016281
111. Forlenza GP. Use of artificial intelligence to improve diabetes outcomes in patients using multiple daily injections therapy. Diabetes Technol Ther. 2019;21(S2):S24-S28. doi:10.1089/dia.2019.0077
112. Poser CM. CT scan and the practice of neurology. Arch Neurol. 1977;34(2):132. doi:10.1001/archneur.1977.00500140086023
113. Angus DC. Randomized clinical trials of artificial intelligence. JAMA. 2020;323(11):1043-1045. doi:10.1001/jama.2020.1039
114. Topol EJ. Welcoming new guidelines for AI clinical research. Nat Med. 2020;26(9):1318-1320. doi:10.1038/s41591-020-1042-x
115. Collins GS, Moons KGM. Reporting of artificial intelligence prediction models. Lancet. 2019;393(10181):1577-1579. doi:10.1016/S0140-6736(19)30037-6
116. Cruz Rivera S, Liu X, Chan AW, et al. Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension. Nat Med. 2020;26(9):1351-1363. doi:10.1038/s41591-020-1037-7
117. Liu X, Cruz Rivera S, Moher D, Calvert MJ, Denniston AK; SPIRIT-AI and CONSORT-AI Working Group. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension. Nat Med. 2020;26(9):1364-1374. doi:10.1038/s41591-020-1034-x
118. McCulloch WS, Pitts W. A logical calculus of the ideas immanent in nervous activity. Bull Math Biophys. 1943;5(4):115-133. doi:10.1007/BF02478259
119. Samuel AL. Some studies in machine learning using the game of Checkers. IBM J Res Dev. 1959;3(3):535-554. Accessed September 15, 2021. https://citeseerx.ist.psu.edu/viewdoc/summary?doi=10.1.1.368.2254
120. Sonoda M, Takano M, Miyahara J, Kato H. Computed radiography utilizing scanning laser stimulated luminescence. Radiology. 1983;148(3):833-838. doi:10.1148/radiology.148.3.6878707
121. Dechter R. Learning while searching in constraint-satisfaction-problems. AAAI’86: proceedings of the fifth AAAI national conference on artificial intelligence. Published 1986. Accessed September 15, 2021. https://www.aaai.org/Papers/AAAI/1986/AAAI86-029.pdf
122. Le Cun Y, Jackel LD, Boser B, et al. Handwritten digit recognition: applications of neural network chips and automatic learning. IEEE Commun Mag. 1989;27(11):41-46. doi:10.1109/35.41400
123. US Food and Drug Administration. FDA allows marketing of first whole slide imaging system for digital pathology [press release]. Published April 12, 2017. Accessed September 15, 2021. https://www.fda.gov/news-events/press-announcements/fda-allows-marketing-first-whole-slide-imaging-system-digital-pathology
Artificial Intelligence (AI) was first described in 1956 and refers to machines having the ability to learn as they receive and process information, resulting in the ability to “think” like humans.1 AI’s impact in medicine is increasing; currently, at least 29 AI medical devices and algorithms are approved by the US Food and Drug Administration (FDA) in a variety of areas, including radiograph interpretation, managing glucose levels in patients with diabetes mellitus, analyzing electrocardiograms (ECGs), and diagnosing sleep disorders among others.2 Significantly, in 2020, the Centers for Medicare and Medicaid Services (CMS) announced the first reimbursement to hospitals for an AI platform, a model for early detection of strokes.3 AI is rapidly becoming an integral part of health care, and its role will only increase in the future (Table).
As knowledge in medicine is expanding exponentially, AI has great potential to assist with handling complex patient care data. The concept of exponential growth is not a natural one. As Bini described, with exponential growth the volume of knowledge amassed over the past 10 years will now occur in perhaps only 1 year.1 Likewise, equivalent advances over the past year may take just a few months. This phenomenon is partly due to the law of accelerating returns, which states that advances feed on themselves, continually increasing the rate of further advances.4 The volume of medical data doubles every 2 to 5 years.5 Fortunately, the field of AI is growing exponentially as well and can help health care practitioners (HCPs) keep pace, allowing the continued delivery of effective health care.
In this report, we review common terminology, principles, and general applications of AI, followed by current and potential applications of AI for selected medical specialties. Finally, we discuss AI’s future in health care, along with potential risks and pitfalls.
AI Overview
AI refers to machine programs that can “learn” or think based on past experiences. This functionality contrasts with simple rules-based programming available to health care for years. An example of rules-based programming is the warfarindosing.org website developed by Barnes-Jewish Hospital at Washington University Medical Center, which guides initial warfarin dosing.6,7 The prescriber inputs detailed patient information, including age, sex, height, weight, tobacco history, medications, laboratory results, and genotype if available. The application then calculates recommended warfarin dosing regimens to avoid over- or underanticoagulation. While the dosing algorithm may be complex, it depends entirely on preprogrammed rules. The program does not learn to reach its conclusions and recommendations from patient data.
In contrast, one of the most common subsets of AI is machine learning (ML). ML describes a program that “learns from experience and improves its performance as it learns.”1 With ML, the computer is initially provided with a training data set—data with known outcomes or labels. Because the initial data are input from known samples, this type of AI is known as supervised learning.8-10 As an example, we recently reported using ML to diagnose various types of cancer from pathology slides.11 In one experiment, we captured images of colon adenocarcinoma and normal colon (these 2 groups represent the training data set). Unlike traditional programming, we did not define characteristics that would differentiate colon cancer from normal; rather, the machine learned these characteristics independently by assessing the labeled images provided. A second data set (the validation data set) was used to evaluate the program and fine-tune the ML training model’s parameters. Finally, the program was presented with new images of cancer and normal cases for final assessment of accuracy (test data set). Our program learned to recognize differences from the images provided and was able to differentiate normal and cancer images with > 95% accuracy.
Advances in computer processing have allowed for the development of artificial neural networks (ANNs). While there are several types of ANNs, the most common types used for image classification and segmentation are known as convolutional neural networks (CNNs).9,12-14 The programs are designed to work similar to the human brain, specifically the visual cortex.15,16 As data are acquired, they are processed by various layers in the program. Much like neurons in the brain, one layer decides whether to advance information to the next.13,14 CNNs can be many layers deep, leading to the term deep learning: “computational models that are composed of multiple processing layers to learn representations of data with multiple levels of abstraction.”1,13,17
ANNs can process larger volumes of data. This advance has led to the development of unstructured or unsupervised learning. With this type of learning, imputing defined features (ie, predetermined answers) of the training data set described above is no longer required.1,8,10,14 The advantage of unsupervised learning is that the program can be presented raw data and extract meaningful interpretation without human input, often with less bias than may exist with supervised learning.1,18 If shown enough data, the program can extract relevant features to make conclusions independently without predefined definitions, potentially uncovering markers not previously known. For example, several studies have used unsupervised learning to search patient data to assess readmission risks of patients with congestive heart failure.10,19,20 AI compiled features independently and not previously defined, predicting patients at greater risk for readmission superior to traditional methods.
A more detailed description of the various terminologies and techniques of AI is beyond the scope of this review.9,10,17,21 However, in this basic overview, we describe 4 general areas that AI impacts health care (Figure).
Health Care Applications
Image analysis has seen the most AI health care applications.8,15 AI has shown potential in interpreting many types of medical images, including pathology slides, radiographs of various types, retina and other eye scans, and photographs of skin lesions. Many studies have demonstrated that AI can interpret these images as accurately as or even better than experienced clinicians.9,13,22-29 Studies have suggested AI interpretation of radiographs may better distinguish patients infected with COVID-19 from other causes of pneumonia, and AI interpretation of pathology slides may detect specific genetic mutations not previously identified without additional molecular tests.11,14,23,24,30-32
The second area in which AI can impact health care is improving workflow and efficiency. AI has improved surgery scheduling, saving significant revenue, and decreased patient wait times for appointments.1 AI can screen and triage radiographs, allowing attention to be directed to critical patients. This use would be valuable in many busy clinical settings, such as the recent COVID-19 pandemic.8,23 Similarly, AI can screen retina images to prioritize urgent conditions.25 AI has improved pathologists’ efficiency when used to detect breast metastases.33 Finally, AI may reduce medical errors, thereby ensuring patient safety.8,9,34
A third health care benefit of AI is in public health and epidemiology. AI can assist with clinical decision-making and diagnoses in low-income countries and areas with limited health care resources and personnel.25,29 AI can improve identification of infectious outbreaks, such as tuberculosis, malaria, dengue fever, and influenza.29,35-40 AI has been used to predict transmission patterns of the Zika virus and the current COVID-19 pandemic.41,42 Applications can stratify the risk of outbreaks based on multiple factors, including age, income, race, atypical geographic clusters, and seasonal factors like rainfall and temperature.35,36,38,43 AI has been used to assess morbidity and mortality, such as predicting disease severity with malaria and identifying treatment failures in tuberculosis.29
Finally, AI can dramatically impact health care due to processing large data sets or disconnected volumes of patient information—so-called big data.44-46 An example is the widespread use of electronic health records (EHRs) such as the Computerized Patient Record System used in Veteran Affairs medical centers (VAMCs). Much of patient information exists in written text: HCP notes, laboratory and radiology reports, medication records, etc. Natural language processing (NLP) allows platforms to sort through extensive volumes of data on complex patients at rates much faster than human capability, which has great potential to assist with diagnosis and treatment decisions.9
Medical literature is being produced at rates that exceed our ability to digest. More than 200,000 cancer-related articles were published in 2019 alone.14 NLP capabilities of AI have the potential to rapidly sort through this extensive medical literature and relate specific verbiage in patient records guiding therapy.46 IBM Watson, a supercomputer based on ML and NLP, demonstrates this concept with many potential applications, only some of which relate to health care.1,9 Watson has an oncology component to assimilate multiple aspects of patient care, including clinical notes, pathology results, radiograph findings, staging, and a tumor’s genetic profile. It coordinates these inputs from the EHR and mines medical literature and research databases to recommend treatment options.1,46 AI can assess and compile far greater patient data and therapeutic options than would be feasible by individual clinicians, thus providing customized patient care.47 Watson has partnered with numerous medical centers, including MD Anderson Cancer Center and Memorial Sloan Kettering Cancer Center, with variable success.44,47-49 While the full potential of Watson appears not yet realized, these AI-driven approaches will likely play an important role in leveraging the hidden value in the expanding volume of health care information.
Medical Specialty Applications
Radiology
Currently > 70% of FDA-approved AI medical devices are in the field of radiology.2 Most radiology departments have used AI-friendly digital imaging for years, such as the picture archiving and communication systems used by numerous health care systems, including VAMCs.2,15 Gray-scale images common in radiology lend themselves to standardization, although AI is not limited to black-and- white image interpretation.15
An abundance of literature describes plain radiograph interpretation using AI. One FDA-approved platform improved X-ray diagnosis of wrist fractures when used by emergency medicine clinicians.2,50 AI has been applied to chest X-ray (CXR) interpretation of many conditions, including pneumonia, tuberculosis, malignant lung lesions, and COVID-19.23,25,28,44,51-53 For example, Nam and colleagues suggested AI is better at diagnosing malignant pulmonary nodules from CXRs than are trained radiologists.28
In addition to plain radiographs, AI has been applied to many other imaging technologies, including ultrasounds, positron emission tomography, mammograms, computed tomography (CT), and magnetic resonance imaging (MRI).15,26,44,48,54-56 A large study demonstrated that ML platforms significantly reduced the time to diagnose intracranial hemorrhages on CT and identified subtle hemorrhages missed by radiologists.55 Other studies have claimed that AI programs may be better than radiologists in detecting cancer in screening mammograms, and 3 FDA-approved devices focus on mammogram interpretation.2,15,54,57 There is also great interest in MRI applications to detect and predict prognosis for breast cancer based on imaging findings.21,56
Aside from providing accurate diagnoses, other studies focus on AI radiograph interpretation to assist with patient screening, triage, improving time to final diagnosis, providing a rapid “second opinion,” and even monitoring disease progression and offering insights into prognosis.8,21,23,52,55,56,58 These features help in busy urban centers but may play an even greater role in areas with limited access to health care or trained specialists such as radiologists.52
Cardiology
Cardiology has the second highest number of FDA-approved AI applications.2 Many cardiology AI platforms involve image analysis, as described in several recent reviews.45,59,60 AI has been applied to echocardiography to measure ejection fractions, detect valvular disease, and assess heart failure from hypertrophic and restrictive cardiomyopathy and amyloidosis.45,48,59 Applications for cardiac CT scans and CT angiography have successfully quantified both calcified and noncalcified coronary artery plaques and lumen assessments, assessed myocardial perfusion, and performed coronary artery calcium scoring.45,59,60 Likewise, AI applications for cardiac MRI have been used to quantitate ejection fraction, large vessel flow assessment, and cardiac scar burden.45,59
For years ECG devices have provided interpretation with limited accuracy using preprogrammed parameters.48 However, the application of AI allows ECG interpretation on par with trained cardiologists. Numerous such AI applications exist, and 2 FDA-approved devices perform ECG interpretation.2,61-64 One of these devices incorporates an AI-powered stethoscope to detect atrial fibrillation and heart murmurs.65
Pathology
The advancement of whole slide imaging, wherein entire slides can be scanned and digitized at high speed and resolution, creates great potential for AI applications in pathology.12,24,32,33,66 A landmark study demonstrating the potential of AI for assessing whole slide imaging examined sentinel lymph node metastases in patients with breast cancer.22 Multiple algorithms in the study demonstrated that AI was equivalent or better than pathologists in detecting metastases, especially when the pathologists were time-constrained consistent with a normal working environment. Significantly, the most accurate and efficient diagnoses were achieved when the pathologist and AI interpretations were used together.22,33
AI has shown promise in diagnosing many other entities, including cancers of the prostate (including Gleason scoring), lung, colon, breast, and skin.11,12,24,27,32,67 In addition, AI has shown great potential in scoring biomarkers important for prognosis and treatment, such as immunohistochemistry (IHC) labeling of Ki-67 and PD-L1.32 Pathologists can have difficulty classifying certain tumors or determining the site of origin for metastases, often having to rely on IHC with limited success. The unique features of image analysis with AI have the potential to assist in classifying difficult tumors and identifying sites of origin for metastatic disease based on morphology alone.11
Oncology depends heavily on molecular pathology testing to dictate treatment options and determine prognosis. Preliminary studies suggest that AI interpretation alone has the potential to delineate whether certain molecular mutations are present in tumors from various sites.11,14,24,32 One study combined histology and genomic results for AI interpretation that improved prognostic predictions.68 In addition, AI analysis may have potential in predicting tumor recurrence or prognosis based on cellular features, as demonstrated for lung cancer and melanoma.67,69,70
Ophthalmology
AI applications for ophthalmology have focused on diabetic retinopathy, age-related macular degeneration, glaucoma, retinopathy of prematurity, age-related and congenital cataracts, and retinal vein occlusion.71-73 Diabetic retinopathy is a leading cause of blindness and has been studied by numerous platforms with good success, most having used color fundus photography.71,72 One study showed AI could diagnose diabetic retinopathy and diabetic macular edema with specificities similar to ophthalmologists.74 In 2018, the FDA approved the AI platform IDx-DR. This diagnostic system classifies retinal images and recommends referral for patients determined to have “more than mild diabetic retinopathy” and reexamination within a year for other patients.8,75 Significantly, the platform recommendations do not require confirmation by a clinician.8
AI has been applied to other modalities in ophthalmology such as optical coherence tomography (OCT) to diagnose retinal disease and to predict appropriate management of congenital cataracts.25,73,76 For example, an AI application using OCT has been demonstrated to match or exceed the accuracy of retinal experts in diagnosing and triaging patients with a variety of retinal pathologies, including patients needing urgent referrals.77
Dermatology
Multiple studies demonstrate AI performs at least equal to experienced dermatologists in differentiating selected skin lesions.78-81 For example, Esteva and colleagues demonstrated AI could differentiate keratinocyte carcinomas from benign seborrheic keratoses and malignant melanomas from benign nevi with accuracy equal to 21 board-certified dermatologists.78
AI is applicable to various imaging procedures common to dermatology, such as dermoscopy, very high-frequency ultrasound, and reflectance confocal microscopy.82 Several studies have demonstrated that AI interpretation compared favorably to dermatologists evaluating dermoscopy to assess melanocytic lesions.78-81,83
A limitation in these studies is that they differentiate only a few diagnoses.82 Furthermore, dermatologists have sensory input such as touch and visual examination under various conditions, something AI has yet to replicate.15,34,84 Also, most AI devices use no or limited clinical information.81 Dermatologists can recognize rarer conditions for which AI models may have had limited or no training.34 Nevertheless, a recent study assessed AI for the diagnosis of 134 separate skin disorders with promising results, including providing diagnoses with accuracy comparable to that of dermatologists and providing accurate treatment strategies.84 As Topol points out, most skin lesions are diagnosed in the primary care setting where AI can have a greater impact when used in conjunction with the clinical impression, especially where specialists are in limited supply.48,78
Finally, dermatology lends itself to using portable or smartphone applications (apps) wherein the user can photograph a lesion for analysis by AI algorithms to assess the need for further evaluation or make treatment recommendations.34,84,85 Although results from currently available apps are not encouraging, they may play a greater role as the technology advances.34,85
Oncology
Applications of AI in oncology include predicting prognosis for patients with cancer based on histologic and/or genetic information.14,68,86 Programs can predict the risk of complications before and recurrence risks after surgery for malignancies.44,87-89 AI can also assist in treatment planning and predict treatment failure with radiation therapy.90,91
AI has great potential in processing the large volumes of patient data in cancer genomics. Next-generation sequencing has allowed for the identification of millions of DNA sequences in a single tumor to detect genetic anomalies.92 Thousands of mutations can be found in individual tumor samples, and processing this information and determining its significance can be beyond human capability.14 We know little about the effects of various mutation combinations, and most tumors have a heterogeneous molecular profile among different cell populations.14,93 The presence or absence of various mutations can have diagnostic, prognostic, and therapeutic implications.93 AI has great potential to sort through these complex data and identify actionable findings.
More than 200,000 cancer-related articles were published in 2019, and publications in the field of cancer genomics are increasing exponentially.14,92,93 Patel and colleagues assessed the utility of IBM Watson for Genomics against results from a molecular tumor board.93 Watson for Genomics identified potentially significant mutations not identified by the tumor board in 32% of patients. Most mutations were related to new clinical trials not yet added to the tumor board watch list, demonstrating the role AI will have in processing the large volume of genetic data required to deliver personalized medicine moving forward.
Gastroenterology
AI has shown promise in predicting risk or outcomes based on clinical parameters in various common gastroenterology problems, including gastric reflux, acute pancreatitis, gastrointestinal bleeding, celiac disease, and inflammatory bowel disease.94,95 AI endoscopic analysis has demonstrated potential in assessing Barrett’s esophagus, gastric Helicobacter pylori infections, gastric atrophy, and gastric intestinal metaplasia.95 Applications have been used to assess esophageal, gastric, and colonic malignancies, including depth of invasion based on endoscopic images.95 Finally, studies have evaluated AI to assess small colon polyps during colonoscopy, including differentiating benign and premalignant polyps with success comparable to gastroenterologists.94,95 AI has been shown to increase the speed and accuracy of gastroenterologists in detecting small polyps during colonoscopy.48 In a prospective randomized study, colonoscopies performed using an AI device identified significantly more small adenomatous polyps than colonoscopies without AI.96
Neurology
It has been suggested that AI technologies are well suited for application in neurology due to the subtle presentation of many neurologic diseases.16 Viz LVO, the first CMS-approved AI reimbursement for the diagnosis of strokes, analyzes CTs to detect early ischemic strokes and alerts the medical team, thus shortening time to treatment.3,97 Many other AI platforms are in use or development that use CT and MRI for the early detection of strokes as well as for treatment and prognosis.9,97
AI technologies have been applied to neurodegenerative diseases, such as Alzheimer and Parkinson diseases.16,98 For example, several studies have evaluated patient movements in Parkinson disease for both early diagnosis and to assess response to treatment.98 These evaluations included assessment with both external cameras as well as wearable devices and smartphone apps.
AI has also been applied to seizure disorders, attempting to determine seizure type, localize the area of seizure onset, and address the challenges of identifying seizures in neonates.99,100 Other potential applications range from early detection and prognosis predictions for cases of multiple sclerosis to restoring movement in paralysis from a variety of conditions such as spinal cord injury.9,101,102
Mental Health
Due to the interactive nature of mental health care, the field has been slower to develop AI applications.18 With heavy reliance on textual information (eg, clinic notes, mood rating scales, and documentation of conversations), successful AI applications in this field will likely rely heavily on NLP.18 However, studies investigating the application of AI to mental health have also incorporated data such as brain imaging, smartphone monitoring, and social media platforms, such as Facebook and Twitter.18,103,104
The risk of suicide is higher in veteran patients, and ML algorithms have had limited success in predicting suicide risk in both veteran and nonveteran populations.104-106 While early models have low positive predictive values and low sensitivities, they still promise to be a useful tool in conjunction with traditional risk assessments.106 Kessler and colleagues suggest that combining multiple rather than single ML algorithms might lead to greater success.105,106
AI may assist in diagnosing other mental health disorders, including major depressive disorder, attention deficit hyperactivity disorder (ADHD), schizophrenia, posttraumatic stress disorder, and Alzheimer disease.103,104,107 These investigations are in the early stages with limited clinical applicability. However, 2 AI applications awaiting FDA approval relate to ADHD and opioid use.2 Furthermore, potential exists for AI to not only assist with prevention and diagnosis of ADHD, but also to identify optimal treatment options.2,103
General and Personalized Medicine
Additional AI applications include diagnosing patients with suspected sepsis, measuring liver iron concentrations, predicting hospital mortality at the time of admission, and more.2,108,109 AI can guide end-of-life decisions such as resuscitation status or whether to initiate mechanical ventilation.48
AI-driven smartphone apps can be beneficial to both patients and clinicians. Examples include predicting nonadherence to anticoagulation therapy, monitoring heart rhythms for atrial fibrillation or signs of hyperkalemia in patients with renal failure, and improving outcomes for patients with diabetes mellitus by decreasing glycemic variability and reducing hypoglycemia.8,48,110,111 The potential for AI applications to health care and personalized medicine are almost limitless.
Discussion
With ever-increasing expectations for all health care sectors to deliver timely, fiscally-responsible, high-quality health care, AI has the potential to have numerous impacts. AI can improve diagnostic accuracy while limiting errors and impact patient safety such as assisting with prescription delivery.8,9,34 It can screen and triage patients, alerting clinicians to those needing more urgent evaluation.8,23,77,97 AI also may increase a clinician’s efficiency and speed to render a diagnosis.12,13,55,97 AI can provide a rapid second opinion, an ability especially beneficial in underserved areas with shortages of specialists.23,25,26,29,34 Similarly, AI may decrease the inter- and intraobserver variability common in many medical specialties.12,27,45 AI applications can also monitor disease progression, identifying patients at greatest risk, and provide information for prognosis.21,23,56,58 Finally, as described with applications using IBM Watson, AI can allow for an integrated approach to health care that is currently lacking.
We have described many reports suggesting AI can render diagnoses as well as or better than experienced clinicians, and speculation exists that AI will replace many roles currently performed by health care practitioners.9,26 However, most studies demonstrate that AI’s diagnostic benefits are best realized when used to supplement a clinician’s impression.8,22,30,33,52,54,56,69,84 AI is not likely to replace humans in health care in the foreseeable future. The technology can be likened to the impact of CT scans developed in the 1970s in neurology. Prior to such detailed imaging, neurologists spent extensive time performing detailed physicals to render diagnoses and locate lesions before surgery. There was mistrust of this new technology and concern that CT scans would eliminate the need for neurologists.112 On the contrary, neurology is alive and well, frequently being augmented by the technologies once speculated to replace it.
Commercial AI health care platforms represented a $2 billion industry in 2018 and are growing rapidly each year.13,32 Many AI products are offered ready for implementation for various tasks, including diagnostics, patient management, and improved efficiency. Others will likely be provided as templates suitable for modification to meet the specific needs of the facility, practice, or specialty for its patient population.
AI Risks and Limitations
AI has several risks and limitations. Although there is progress in explainable AI, at times we still struggle to understand how the output provided by machine learning algorithms was created.44,48 The many layers associated with deep learning self-determine the criteria to reach its conclusion, and these criteria can continually evolve. The parameters of deep learning are not preprogrammed, and there are too many individual data points to be extrapolated or deconvoluted for evaluation at our current level of knowledge.26,51 These apparent lack of constraints cause concern for patient safety and suggest that greater validation and continued scrutiny of validity is required.8,48 Efforts are underway to create explainable AI programs to make their processes more transparent, but such clarification is limited presently.14,26,48,77
Another challenge of AI is determining the amount of training data required to function optimally. Also, if the output describes multiple variables or diagnoses, are each equally valid?113 Furthermore, many AI applications look for a specific process, such as cancer diagnoses on CXRs. However, how coexisting conditions like cardiomegaly, emphysema, pneumonia, etc, seen on CXRs will affect the diagnosis needs to be considered.51,52 Zech and colleagues provide the example that diagnoses for pneumothorax are frequently rendered on CXRs with chest tubes in place.51 They suggest that CNNs may develop a bias toward diagnosing pneumothorax when chest tubes are present. Many current studies approach an issue in isolation, a situation not realistic in real-world clinical practice.26
Most studies on AI have been retrospective, and frequently data used to train the program are preselected.13,26 The data are typically validated on available databases rather than actual patients in the clinical setting, limiting confidence in the validity of the AI output when applied to real-world situations. Currently, fewer than 12 prospective trials had been published comparing AI with traditional clinical care.13,114 Randomized prospective clinical trials are even fewer, with none currently reported from the United States.13,114 The results from several studies have been shown to diminish when repeated prospectively.114
The FDA has created a new category known as Software as a Medical Device and has a Digital Health Innovation Action Plan to regulate AI platforms. Still, the process of AI regulation is of necessity different from traditional approval processes and is continually evolving.8 The FDA approval process cannot account for the fact that the program’s parameters may continually evolve or adapt.2
Guidelines for investigating and reporting AI research with its unique attributes are being developed. Examples include the TRIPOD-ML statement and others.49,115 In September 2020, 2 publications addressed the paucity of gold-standard randomized clinical trials in clinical AI applications.116,117 The SPIRIT-AI statement expands on the original SPIRIT statement published in 2013 to guide minimal reporting standards for AI clinical trial protocols to promote transparency of design and methodology.116 Similarly, the CONSORT-AI extension, stemming from the original CONSORT statement in 1996, aims to ensure quality reporting of randomized controlled trials in AI.117
Another risk with AI is that while an individual physician making a mistake may adversely affect 1 patient, a single mistake in an AI algorithm could potentially affect thousands of patients.48 Also, AI programs developed for patient populations at a facility may not translate to another. Referred to as overfitting, this phenomenon relates to selection bias in training data sets.15,34,49,51,52 Studies have shown that programs that underrepresent certain group characteristics such as age, sex, or race may be less effective when applied to a population in which these characteristics have differing representations.8,48,49 This problem of underrepresentation has been demonstrated in programs interpreting pathology slides, radiographs, and skin lesions.15,32,51
Admittedly, most of these challenges are not specific to AI and existed in health care previously. Physicians make mistakes, treatments are sometimes used without adequate prospective studies, and medications are given without understanding their mechanism of action, much like AI-facilitated processes reach a conclusion that cannot be fully explained.48
Conclusions
The view that AI will dramatically impact health care in the coming years will likely prove true. However, much work is needed, especially because of the paucity of prospective clinical trials as has been historically required in medical research. Any concern that AI will replace HCPs seems unwarranted. Early studies suggest that even AI programs that appear to exceed human interpretation perform best when working in cooperation with and oversight from clinicians. AI’s greatest potential appears to be its ability to augment care from health professionals, improving efficiency and accuracy, and should be anticipated with enthusiasm as the field moves forward at an exponential rate.
Acknowledgments
The authors thank Makenna G. Thomas for proofreading and review of the manuscript. This material is the result of work supported with resources and the use of facilities at the James A. Haley Veterans’ Hospital. This research has been approved by the James A. Haley Veteran’s Hospital Office of Communications and Media.
Artificial Intelligence (AI) was first described in 1956 and refers to machines having the ability to learn as they receive and process information, resulting in the ability to “think” like humans.1 AI’s impact in medicine is increasing; currently, at least 29 AI medical devices and algorithms are approved by the US Food and Drug Administration (FDA) in a variety of areas, including radiograph interpretation, managing glucose levels in patients with diabetes mellitus, analyzing electrocardiograms (ECGs), and diagnosing sleep disorders among others.2 Significantly, in 2020, the Centers for Medicare and Medicaid Services (CMS) announced the first reimbursement to hospitals for an AI platform, a model for early detection of strokes.3 AI is rapidly becoming an integral part of health care, and its role will only increase in the future (Table).
As knowledge in medicine is expanding exponentially, AI has great potential to assist with handling complex patient care data. The concept of exponential growth is not a natural one. As Bini described, with exponential growth the volume of knowledge amassed over the past 10 years will now occur in perhaps only 1 year.1 Likewise, equivalent advances over the past year may take just a few months. This phenomenon is partly due to the law of accelerating returns, which states that advances feed on themselves, continually increasing the rate of further advances.4 The volume of medical data doubles every 2 to 5 years.5 Fortunately, the field of AI is growing exponentially as well and can help health care practitioners (HCPs) keep pace, allowing the continued delivery of effective health care.
In this report, we review common terminology, principles, and general applications of AI, followed by current and potential applications of AI for selected medical specialties. Finally, we discuss AI’s future in health care, along with potential risks and pitfalls.
AI Overview
AI refers to machine programs that can “learn” or think based on past experiences. This functionality contrasts with simple rules-based programming available to health care for years. An example of rules-based programming is the warfarindosing.org website developed by Barnes-Jewish Hospital at Washington University Medical Center, which guides initial warfarin dosing.6,7 The prescriber inputs detailed patient information, including age, sex, height, weight, tobacco history, medications, laboratory results, and genotype if available. The application then calculates recommended warfarin dosing regimens to avoid over- or underanticoagulation. While the dosing algorithm may be complex, it depends entirely on preprogrammed rules. The program does not learn to reach its conclusions and recommendations from patient data.
In contrast, one of the most common subsets of AI is machine learning (ML). ML describes a program that “learns from experience and improves its performance as it learns.”1 With ML, the computer is initially provided with a training data set—data with known outcomes or labels. Because the initial data are input from known samples, this type of AI is known as supervised learning.8-10 As an example, we recently reported using ML to diagnose various types of cancer from pathology slides.11 In one experiment, we captured images of colon adenocarcinoma and normal colon (these 2 groups represent the training data set). Unlike traditional programming, we did not define characteristics that would differentiate colon cancer from normal; rather, the machine learned these characteristics independently by assessing the labeled images provided. A second data set (the validation data set) was used to evaluate the program and fine-tune the ML training model’s parameters. Finally, the program was presented with new images of cancer and normal cases for final assessment of accuracy (test data set). Our program learned to recognize differences from the images provided and was able to differentiate normal and cancer images with > 95% accuracy.
Advances in computer processing have allowed for the development of artificial neural networks (ANNs). While there are several types of ANNs, the most common types used for image classification and segmentation are known as convolutional neural networks (CNNs).9,12-14 The programs are designed to work similar to the human brain, specifically the visual cortex.15,16 As data are acquired, they are processed by various layers in the program. Much like neurons in the brain, one layer decides whether to advance information to the next.13,14 CNNs can be many layers deep, leading to the term deep learning: “computational models that are composed of multiple processing layers to learn representations of data with multiple levels of abstraction.”1,13,17
ANNs can process larger volumes of data. This advance has led to the development of unstructured or unsupervised learning. With this type of learning, imputing defined features (ie, predetermined answers) of the training data set described above is no longer required.1,8,10,14 The advantage of unsupervised learning is that the program can be presented raw data and extract meaningful interpretation without human input, often with less bias than may exist with supervised learning.1,18 If shown enough data, the program can extract relevant features to make conclusions independently without predefined definitions, potentially uncovering markers not previously known. For example, several studies have used unsupervised learning to search patient data to assess readmission risks of patients with congestive heart failure.10,19,20 AI compiled features independently and not previously defined, predicting patients at greater risk for readmission superior to traditional methods.
A more detailed description of the various terminologies and techniques of AI is beyond the scope of this review.9,10,17,21 However, in this basic overview, we describe 4 general areas that AI impacts health care (Figure).
Health Care Applications
Image analysis has seen the most AI health care applications.8,15 AI has shown potential in interpreting many types of medical images, including pathology slides, radiographs of various types, retina and other eye scans, and photographs of skin lesions. Many studies have demonstrated that AI can interpret these images as accurately as or even better than experienced clinicians.9,13,22-29 Studies have suggested AI interpretation of radiographs may better distinguish patients infected with COVID-19 from other causes of pneumonia, and AI interpretation of pathology slides may detect specific genetic mutations not previously identified without additional molecular tests.11,14,23,24,30-32
The second area in which AI can impact health care is improving workflow and efficiency. AI has improved surgery scheduling, saving significant revenue, and decreased patient wait times for appointments.1 AI can screen and triage radiographs, allowing attention to be directed to critical patients. This use would be valuable in many busy clinical settings, such as the recent COVID-19 pandemic.8,23 Similarly, AI can screen retina images to prioritize urgent conditions.25 AI has improved pathologists’ efficiency when used to detect breast metastases.33 Finally, AI may reduce medical errors, thereby ensuring patient safety.8,9,34
A third health care benefit of AI is in public health and epidemiology. AI can assist with clinical decision-making and diagnoses in low-income countries and areas with limited health care resources and personnel.25,29 AI can improve identification of infectious outbreaks, such as tuberculosis, malaria, dengue fever, and influenza.29,35-40 AI has been used to predict transmission patterns of the Zika virus and the current COVID-19 pandemic.41,42 Applications can stratify the risk of outbreaks based on multiple factors, including age, income, race, atypical geographic clusters, and seasonal factors like rainfall and temperature.35,36,38,43 AI has been used to assess morbidity and mortality, such as predicting disease severity with malaria and identifying treatment failures in tuberculosis.29
Finally, AI can dramatically impact health care due to processing large data sets or disconnected volumes of patient information—so-called big data.44-46 An example is the widespread use of electronic health records (EHRs) such as the Computerized Patient Record System used in Veteran Affairs medical centers (VAMCs). Much of patient information exists in written text: HCP notes, laboratory and radiology reports, medication records, etc. Natural language processing (NLP) allows platforms to sort through extensive volumes of data on complex patients at rates much faster than human capability, which has great potential to assist with diagnosis and treatment decisions.9
Medical literature is being produced at rates that exceed our ability to digest. More than 200,000 cancer-related articles were published in 2019 alone.14 NLP capabilities of AI have the potential to rapidly sort through this extensive medical literature and relate specific verbiage in patient records guiding therapy.46 IBM Watson, a supercomputer based on ML and NLP, demonstrates this concept with many potential applications, only some of which relate to health care.1,9 Watson has an oncology component to assimilate multiple aspects of patient care, including clinical notes, pathology results, radiograph findings, staging, and a tumor’s genetic profile. It coordinates these inputs from the EHR and mines medical literature and research databases to recommend treatment options.1,46 AI can assess and compile far greater patient data and therapeutic options than would be feasible by individual clinicians, thus providing customized patient care.47 Watson has partnered with numerous medical centers, including MD Anderson Cancer Center and Memorial Sloan Kettering Cancer Center, with variable success.44,47-49 While the full potential of Watson appears not yet realized, these AI-driven approaches will likely play an important role in leveraging the hidden value in the expanding volume of health care information.
Medical Specialty Applications
Radiology
Currently > 70% of FDA-approved AI medical devices are in the field of radiology.2 Most radiology departments have used AI-friendly digital imaging for years, such as the picture archiving and communication systems used by numerous health care systems, including VAMCs.2,15 Gray-scale images common in radiology lend themselves to standardization, although AI is not limited to black-and- white image interpretation.15
An abundance of literature describes plain radiograph interpretation using AI. One FDA-approved platform improved X-ray diagnosis of wrist fractures when used by emergency medicine clinicians.2,50 AI has been applied to chest X-ray (CXR) interpretation of many conditions, including pneumonia, tuberculosis, malignant lung lesions, and COVID-19.23,25,28,44,51-53 For example, Nam and colleagues suggested AI is better at diagnosing malignant pulmonary nodules from CXRs than are trained radiologists.28
In addition to plain radiographs, AI has been applied to many other imaging technologies, including ultrasounds, positron emission tomography, mammograms, computed tomography (CT), and magnetic resonance imaging (MRI).15,26,44,48,54-56 A large study demonstrated that ML platforms significantly reduced the time to diagnose intracranial hemorrhages on CT and identified subtle hemorrhages missed by radiologists.55 Other studies have claimed that AI programs may be better than radiologists in detecting cancer in screening mammograms, and 3 FDA-approved devices focus on mammogram interpretation.2,15,54,57 There is also great interest in MRI applications to detect and predict prognosis for breast cancer based on imaging findings.21,56
Aside from providing accurate diagnoses, other studies focus on AI radiograph interpretation to assist with patient screening, triage, improving time to final diagnosis, providing a rapid “second opinion,” and even monitoring disease progression and offering insights into prognosis.8,21,23,52,55,56,58 These features help in busy urban centers but may play an even greater role in areas with limited access to health care or trained specialists such as radiologists.52
Cardiology
Cardiology has the second highest number of FDA-approved AI applications.2 Many cardiology AI platforms involve image analysis, as described in several recent reviews.45,59,60 AI has been applied to echocardiography to measure ejection fractions, detect valvular disease, and assess heart failure from hypertrophic and restrictive cardiomyopathy and amyloidosis.45,48,59 Applications for cardiac CT scans and CT angiography have successfully quantified both calcified and noncalcified coronary artery plaques and lumen assessments, assessed myocardial perfusion, and performed coronary artery calcium scoring.45,59,60 Likewise, AI applications for cardiac MRI have been used to quantitate ejection fraction, large vessel flow assessment, and cardiac scar burden.45,59
For years ECG devices have provided interpretation with limited accuracy using preprogrammed parameters.48 However, the application of AI allows ECG interpretation on par with trained cardiologists. Numerous such AI applications exist, and 2 FDA-approved devices perform ECG interpretation.2,61-64 One of these devices incorporates an AI-powered stethoscope to detect atrial fibrillation and heart murmurs.65
Pathology
The advancement of whole slide imaging, wherein entire slides can be scanned and digitized at high speed and resolution, creates great potential for AI applications in pathology.12,24,32,33,66 A landmark study demonstrating the potential of AI for assessing whole slide imaging examined sentinel lymph node metastases in patients with breast cancer.22 Multiple algorithms in the study demonstrated that AI was equivalent or better than pathologists in detecting metastases, especially when the pathologists were time-constrained consistent with a normal working environment. Significantly, the most accurate and efficient diagnoses were achieved when the pathologist and AI interpretations were used together.22,33
AI has shown promise in diagnosing many other entities, including cancers of the prostate (including Gleason scoring), lung, colon, breast, and skin.11,12,24,27,32,67 In addition, AI has shown great potential in scoring biomarkers important for prognosis and treatment, such as immunohistochemistry (IHC) labeling of Ki-67 and PD-L1.32 Pathologists can have difficulty classifying certain tumors or determining the site of origin for metastases, often having to rely on IHC with limited success. The unique features of image analysis with AI have the potential to assist in classifying difficult tumors and identifying sites of origin for metastatic disease based on morphology alone.11
Oncology depends heavily on molecular pathology testing to dictate treatment options and determine prognosis. Preliminary studies suggest that AI interpretation alone has the potential to delineate whether certain molecular mutations are present in tumors from various sites.11,14,24,32 One study combined histology and genomic results for AI interpretation that improved prognostic predictions.68 In addition, AI analysis may have potential in predicting tumor recurrence or prognosis based on cellular features, as demonstrated for lung cancer and melanoma.67,69,70
Ophthalmology
AI applications for ophthalmology have focused on diabetic retinopathy, age-related macular degeneration, glaucoma, retinopathy of prematurity, age-related and congenital cataracts, and retinal vein occlusion.71-73 Diabetic retinopathy is a leading cause of blindness and has been studied by numerous platforms with good success, most having used color fundus photography.71,72 One study showed AI could diagnose diabetic retinopathy and diabetic macular edema with specificities similar to ophthalmologists.74 In 2018, the FDA approved the AI platform IDx-DR. This diagnostic system classifies retinal images and recommends referral for patients determined to have “more than mild diabetic retinopathy” and reexamination within a year for other patients.8,75 Significantly, the platform recommendations do not require confirmation by a clinician.8
AI has been applied to other modalities in ophthalmology such as optical coherence tomography (OCT) to diagnose retinal disease and to predict appropriate management of congenital cataracts.25,73,76 For example, an AI application using OCT has been demonstrated to match or exceed the accuracy of retinal experts in diagnosing and triaging patients with a variety of retinal pathologies, including patients needing urgent referrals.77
Dermatology
Multiple studies demonstrate AI performs at least equal to experienced dermatologists in differentiating selected skin lesions.78-81 For example, Esteva and colleagues demonstrated AI could differentiate keratinocyte carcinomas from benign seborrheic keratoses and malignant melanomas from benign nevi with accuracy equal to 21 board-certified dermatologists.78
AI is applicable to various imaging procedures common to dermatology, such as dermoscopy, very high-frequency ultrasound, and reflectance confocal microscopy.82 Several studies have demonstrated that AI interpretation compared favorably to dermatologists evaluating dermoscopy to assess melanocytic lesions.78-81,83
A limitation in these studies is that they differentiate only a few diagnoses.82 Furthermore, dermatologists have sensory input such as touch and visual examination under various conditions, something AI has yet to replicate.15,34,84 Also, most AI devices use no or limited clinical information.81 Dermatologists can recognize rarer conditions for which AI models may have had limited or no training.34 Nevertheless, a recent study assessed AI for the diagnosis of 134 separate skin disorders with promising results, including providing diagnoses with accuracy comparable to that of dermatologists and providing accurate treatment strategies.84 As Topol points out, most skin lesions are diagnosed in the primary care setting where AI can have a greater impact when used in conjunction with the clinical impression, especially where specialists are in limited supply.48,78
Finally, dermatology lends itself to using portable or smartphone applications (apps) wherein the user can photograph a lesion for analysis by AI algorithms to assess the need for further evaluation or make treatment recommendations.34,84,85 Although results from currently available apps are not encouraging, they may play a greater role as the technology advances.34,85
Oncology
Applications of AI in oncology include predicting prognosis for patients with cancer based on histologic and/or genetic information.14,68,86 Programs can predict the risk of complications before and recurrence risks after surgery for malignancies.44,87-89 AI can also assist in treatment planning and predict treatment failure with radiation therapy.90,91
AI has great potential in processing the large volumes of patient data in cancer genomics. Next-generation sequencing has allowed for the identification of millions of DNA sequences in a single tumor to detect genetic anomalies.92 Thousands of mutations can be found in individual tumor samples, and processing this information and determining its significance can be beyond human capability.14 We know little about the effects of various mutation combinations, and most tumors have a heterogeneous molecular profile among different cell populations.14,93 The presence or absence of various mutations can have diagnostic, prognostic, and therapeutic implications.93 AI has great potential to sort through these complex data and identify actionable findings.
More than 200,000 cancer-related articles were published in 2019, and publications in the field of cancer genomics are increasing exponentially.14,92,93 Patel and colleagues assessed the utility of IBM Watson for Genomics against results from a molecular tumor board.93 Watson for Genomics identified potentially significant mutations not identified by the tumor board in 32% of patients. Most mutations were related to new clinical trials not yet added to the tumor board watch list, demonstrating the role AI will have in processing the large volume of genetic data required to deliver personalized medicine moving forward.
Gastroenterology
AI has shown promise in predicting risk or outcomes based on clinical parameters in various common gastroenterology problems, including gastric reflux, acute pancreatitis, gastrointestinal bleeding, celiac disease, and inflammatory bowel disease.94,95 AI endoscopic analysis has demonstrated potential in assessing Barrett’s esophagus, gastric Helicobacter pylori infections, gastric atrophy, and gastric intestinal metaplasia.95 Applications have been used to assess esophageal, gastric, and colonic malignancies, including depth of invasion based on endoscopic images.95 Finally, studies have evaluated AI to assess small colon polyps during colonoscopy, including differentiating benign and premalignant polyps with success comparable to gastroenterologists.94,95 AI has been shown to increase the speed and accuracy of gastroenterologists in detecting small polyps during colonoscopy.48 In a prospective randomized study, colonoscopies performed using an AI device identified significantly more small adenomatous polyps than colonoscopies without AI.96
Neurology
It has been suggested that AI technologies are well suited for application in neurology due to the subtle presentation of many neurologic diseases.16 Viz LVO, the first CMS-approved AI reimbursement for the diagnosis of strokes, analyzes CTs to detect early ischemic strokes and alerts the medical team, thus shortening time to treatment.3,97 Many other AI platforms are in use or development that use CT and MRI for the early detection of strokes as well as for treatment and prognosis.9,97
AI technologies have been applied to neurodegenerative diseases, such as Alzheimer and Parkinson diseases.16,98 For example, several studies have evaluated patient movements in Parkinson disease for both early diagnosis and to assess response to treatment.98 These evaluations included assessment with both external cameras as well as wearable devices and smartphone apps.
AI has also been applied to seizure disorders, attempting to determine seizure type, localize the area of seizure onset, and address the challenges of identifying seizures in neonates.99,100 Other potential applications range from early detection and prognosis predictions for cases of multiple sclerosis to restoring movement in paralysis from a variety of conditions such as spinal cord injury.9,101,102
Mental Health
Due to the interactive nature of mental health care, the field has been slower to develop AI applications.18 With heavy reliance on textual information (eg, clinic notes, mood rating scales, and documentation of conversations), successful AI applications in this field will likely rely heavily on NLP.18 However, studies investigating the application of AI to mental health have also incorporated data such as brain imaging, smartphone monitoring, and social media platforms, such as Facebook and Twitter.18,103,104
The risk of suicide is higher in veteran patients, and ML algorithms have had limited success in predicting suicide risk in both veteran and nonveteran populations.104-106 While early models have low positive predictive values and low sensitivities, they still promise to be a useful tool in conjunction with traditional risk assessments.106 Kessler and colleagues suggest that combining multiple rather than single ML algorithms might lead to greater success.105,106
AI may assist in diagnosing other mental health disorders, including major depressive disorder, attention deficit hyperactivity disorder (ADHD), schizophrenia, posttraumatic stress disorder, and Alzheimer disease.103,104,107 These investigations are in the early stages with limited clinical applicability. However, 2 AI applications awaiting FDA approval relate to ADHD and opioid use.2 Furthermore, potential exists for AI to not only assist with prevention and diagnosis of ADHD, but also to identify optimal treatment options.2,103
General and Personalized Medicine
Additional AI applications include diagnosing patients with suspected sepsis, measuring liver iron concentrations, predicting hospital mortality at the time of admission, and more.2,108,109 AI can guide end-of-life decisions such as resuscitation status or whether to initiate mechanical ventilation.48
AI-driven smartphone apps can be beneficial to both patients and clinicians. Examples include predicting nonadherence to anticoagulation therapy, monitoring heart rhythms for atrial fibrillation or signs of hyperkalemia in patients with renal failure, and improving outcomes for patients with diabetes mellitus by decreasing glycemic variability and reducing hypoglycemia.8,48,110,111 The potential for AI applications to health care and personalized medicine are almost limitless.
Discussion
With ever-increasing expectations for all health care sectors to deliver timely, fiscally-responsible, high-quality health care, AI has the potential to have numerous impacts. AI can improve diagnostic accuracy while limiting errors and impact patient safety such as assisting with prescription delivery.8,9,34 It can screen and triage patients, alerting clinicians to those needing more urgent evaluation.8,23,77,97 AI also may increase a clinician’s efficiency and speed to render a diagnosis.12,13,55,97 AI can provide a rapid second opinion, an ability especially beneficial in underserved areas with shortages of specialists.23,25,26,29,34 Similarly, AI may decrease the inter- and intraobserver variability common in many medical specialties.12,27,45 AI applications can also monitor disease progression, identifying patients at greatest risk, and provide information for prognosis.21,23,56,58 Finally, as described with applications using IBM Watson, AI can allow for an integrated approach to health care that is currently lacking.
We have described many reports suggesting AI can render diagnoses as well as or better than experienced clinicians, and speculation exists that AI will replace many roles currently performed by health care practitioners.9,26 However, most studies demonstrate that AI’s diagnostic benefits are best realized when used to supplement a clinician’s impression.8,22,30,33,52,54,56,69,84 AI is not likely to replace humans in health care in the foreseeable future. The technology can be likened to the impact of CT scans developed in the 1970s in neurology. Prior to such detailed imaging, neurologists spent extensive time performing detailed physicals to render diagnoses and locate lesions before surgery. There was mistrust of this new technology and concern that CT scans would eliminate the need for neurologists.112 On the contrary, neurology is alive and well, frequently being augmented by the technologies once speculated to replace it.
Commercial AI health care platforms represented a $2 billion industry in 2018 and are growing rapidly each year.13,32 Many AI products are offered ready for implementation for various tasks, including diagnostics, patient management, and improved efficiency. Others will likely be provided as templates suitable for modification to meet the specific needs of the facility, practice, or specialty for its patient population.
AI Risks and Limitations
AI has several risks and limitations. Although there is progress in explainable AI, at times we still struggle to understand how the output provided by machine learning algorithms was created.44,48 The many layers associated with deep learning self-determine the criteria to reach its conclusion, and these criteria can continually evolve. The parameters of deep learning are not preprogrammed, and there are too many individual data points to be extrapolated or deconvoluted for evaluation at our current level of knowledge.26,51 These apparent lack of constraints cause concern for patient safety and suggest that greater validation and continued scrutiny of validity is required.8,48 Efforts are underway to create explainable AI programs to make their processes more transparent, but such clarification is limited presently.14,26,48,77
Another challenge of AI is determining the amount of training data required to function optimally. Also, if the output describes multiple variables or diagnoses, are each equally valid?113 Furthermore, many AI applications look for a specific process, such as cancer diagnoses on CXRs. However, how coexisting conditions like cardiomegaly, emphysema, pneumonia, etc, seen on CXRs will affect the diagnosis needs to be considered.51,52 Zech and colleagues provide the example that diagnoses for pneumothorax are frequently rendered on CXRs with chest tubes in place.51 They suggest that CNNs may develop a bias toward diagnosing pneumothorax when chest tubes are present. Many current studies approach an issue in isolation, a situation not realistic in real-world clinical practice.26
Most studies on AI have been retrospective, and frequently data used to train the program are preselected.13,26 The data are typically validated on available databases rather than actual patients in the clinical setting, limiting confidence in the validity of the AI output when applied to real-world situations. Currently, fewer than 12 prospective trials had been published comparing AI with traditional clinical care.13,114 Randomized prospective clinical trials are even fewer, with none currently reported from the United States.13,114 The results from several studies have been shown to diminish when repeated prospectively.114
The FDA has created a new category known as Software as a Medical Device and has a Digital Health Innovation Action Plan to regulate AI platforms. Still, the process of AI regulation is of necessity different from traditional approval processes and is continually evolving.8 The FDA approval process cannot account for the fact that the program’s parameters may continually evolve or adapt.2
Guidelines for investigating and reporting AI research with its unique attributes are being developed. Examples include the TRIPOD-ML statement and others.49,115 In September 2020, 2 publications addressed the paucity of gold-standard randomized clinical trials in clinical AI applications.116,117 The SPIRIT-AI statement expands on the original SPIRIT statement published in 2013 to guide minimal reporting standards for AI clinical trial protocols to promote transparency of design and methodology.116 Similarly, the CONSORT-AI extension, stemming from the original CONSORT statement in 1996, aims to ensure quality reporting of randomized controlled trials in AI.117
Another risk with AI is that while an individual physician making a mistake may adversely affect 1 patient, a single mistake in an AI algorithm could potentially affect thousands of patients.48 Also, AI programs developed for patient populations at a facility may not translate to another. Referred to as overfitting, this phenomenon relates to selection bias in training data sets.15,34,49,51,52 Studies have shown that programs that underrepresent certain group characteristics such as age, sex, or race may be less effective when applied to a population in which these characteristics have differing representations.8,48,49 This problem of underrepresentation has been demonstrated in programs interpreting pathology slides, radiographs, and skin lesions.15,32,51
Admittedly, most of these challenges are not specific to AI and existed in health care previously. Physicians make mistakes, treatments are sometimes used without adequate prospective studies, and medications are given without understanding their mechanism of action, much like AI-facilitated processes reach a conclusion that cannot be fully explained.48
Conclusions
The view that AI will dramatically impact health care in the coming years will likely prove true. However, much work is needed, especially because of the paucity of prospective clinical trials as has been historically required in medical research. Any concern that AI will replace HCPs seems unwarranted. Early studies suggest that even AI programs that appear to exceed human interpretation perform best when working in cooperation with and oversight from clinicians. AI’s greatest potential appears to be its ability to augment care from health professionals, improving efficiency and accuracy, and should be anticipated with enthusiasm as the field moves forward at an exponential rate.
Acknowledgments
The authors thank Makenna G. Thomas for proofreading and review of the manuscript. This material is the result of work supported with resources and the use of facilities at the James A. Haley Veterans’ Hospital. This research has been approved by the James A. Haley Veteran’s Hospital Office of Communications and Media.
1. Bini SA. Artificial intelligence, machine learning, deep learning, and cognitive computing: what do these terms mean and how will they impact health care? J Arthroplasty. 2018;33(8):2358-2361. doi:10.1016/j.arth.2018.02.067
2. Benjamens S, Dhunnoo P, Meskó B. The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database. NPJ Digit Med. 2020;3:118. doi:10.1038/s41746-020-00324-0
3. Viz. AI powered synchronized stroke care. Accessed September 15, 2021. https://www.viz.ai/ischemic-stroke
4. Buchanan M. The law of accelerating returns. Nat Phys. 2008;4(7):507. doi:10.1038/nphys1010
5. IBM Watson Health computes a pair of new solutions to improve healthcare data and security. Published September 10, 2015. Accessed October 21, 2020. https://www.techrepublic.com/article/ibm-watson-health-computes-a-pair-of-new-solutions-to-improve-healthcare-data-and-security
6. Borkowski AA, Kardani A, Mastorides SM, Thomas LB. Warfarin pharmacogenomics: recommendations with available patented clinical technologies. Recent Pat Biotechnol. 2014;8(2):110-115. doi:10.2174/1872208309666140904112003
7. Washington University in St. Louis. Warfarin dosing. Accessed September 15, 2021. http://www.warfarindosing.org/Source/Home.aspx
8. He J, Baxter SL, Xu J, Xu J, Zhou X, Zhang K. The practical implementation of artificial intelligence technologies in medicine. Nat Med. 2019;25(1):30-36. doi:10.1038/s41591-018-0307-0
9. Jiang F, Jiang Y, Zhi H, et al. Artificial intelligence in healthcare: past, present and future. Stroke Vasc Neurol. 2017;2(4):230-243. Published 2017 Jun 21. doi:10.1136/svn-2017-000101
10. Johnson KW, Torres Soto J, Glicksberg BS, et al. Artificial intelligence in cardiology. J Am Coll Cardiol. 2018;71(23):2668-2679. doi:10.1016/j.jacc.2018.03.521
11. Borkowski AA, Wilson CP, Borkowski SA, et al. Comparing artificial intelligence platforms for histopathologic cancer diagnosis. Fed Pract. 2019;36(10):456-463.
12. Cruz-Roa A, Gilmore H, Basavanhally A, et al. High-throughput adaptive sampling for whole-slide histopathology image analysis (HASHI) via convolutional neural networks: application to invasive breast cancer detection. PLoS One. 2018;13(5):e0196828. Published 2018 May 24. doi:10.1371/journal.pone.0196828
13. Nagendran M, Chen Y, Lovejoy CA, et al. Artificial intelligence versus clinicians: systematic review of design, reporting standards, and claims of deep learning studies. BMJ. 2020;368:m689. Published 2020 Mar 25. doi:10.1136/bmj.m689
14. Shimizu H, Nakayama KI. Artificial intelligence in oncology. Cancer Sci. 2020;111(5):1452-1460. doi:10.1111/cas.14377
15. Talebi-Liasi F, Markowitz O. Is artificial intelligence going to replace dermatologists?. Cutis. 2020;105(1):28-31.
16. Valliani AA, Ranti D, Oermann EK. Deep learning and neurology: a systematic review. Neurol Ther. 2019;8(2):351-365. doi:10.1007/s40120-019-00153-8
17. LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521(7553):436-444. doi:10.1038/nature14539
18. Graham S, Depp C, Lee EE, et al. Artificial intelligence for mental health and mental illnesses: an overview. Curr Psychiatry Rep. 2019;21(11):116. Published 2019 Nov 7. doi:10.1007/s11920-019-1094-0
19. Golas SB, Shibahara T, Agboola S, et al. A machine learning model to predict the risk of 30-day readmissions in patients with heart failure: a retrospective analysis of electronic medical records data. BMC Med Inform Decis Mak. 2018;18(1):44. Published 2018 Jun 22. doi:10.1186/s12911-018-0620-z
20. Mortazavi BJ, Downing NS, Bucholz EM, et al. Analysis of machine learning techniques for heart failure readmissions. Circ Cardiovasc Qual Outcomes. 2016;9(6):629-640. doi:10.1161/CIRCOUTCOMES.116.003039
21. Meyer-Bäse A, Morra L, Meyer-Bäse U, Pinker K. Current status and future perspectives of artificial intelligence in magnetic resonance breast imaging. Contrast Media Mol Imaging. 2020;2020:6805710. Published 2020 Aug 28. doi:10.1155/2020/6805710
22. Ehteshami Bejnordi B, Veta M, Johannes van Diest P, et al. Diagnostic assessment of deep learning algorithms for detection of lymph node metastases in women with breast cancer. JAMA. 2017;318(22):2199-2210. doi:10.1001/jama.2017.14585
23. Borkowski AA, Viswanadhan NA, Thomas LB, Guzman RD, Deland LA, Mastorides SM. Using artificial intelligence for COVID-19 chest X-ray diagnosis. Fed Pract. 2020;37(9):398-404. doi:10.12788/fp.0045
24. Coudray N, Ocampo PS, Sakellaropoulos T, et al. Classification and mutation prediction from non-small cell lung cancer histopathology images using deep learning. Nat Med. 2018;24(10):1559-1567. doi:10.1038/s41591-018-0177-5
25. Kermany DS, Goldbaum M, Cai W, et al. Identifying medical diagnoses and treatable diseases by image-based deep learning. Cell. 2018;172(5):1122-1131.e9. doi:10.1016/j.cell.2018.02.010
26. Liu X, Faes L, Kale AU, et al. A comparison of deep learning performance against health-care professionals in detecting diseases from medical imaging: a systematic review and meta-analysis. Lancet Digit Health. 2019;1(6):e271-e297. doi:10.1016/S2589-7500(19)30123-2
27. Nagpal K, Foote D, Liu Y, et al. Development and validation of a deep learning algorithm for improving Gleason scoring of prostate cancer [published correction appears in NPJ Digit Med. 2019 Nov 19;2:113]. NPJ Digit Med. 2019;2:48. Published 2019 Jun 7. doi:10.1038/s41746-019-0112-2
28. Nam JG, Park S, Hwang EJ, et al. Development and validation of deep learning-based automatic detection algorithm for malignant pulmonary nodules on chest radiographs. Radiology. 2019;290(1):218-228. doi:10.1148/radiol.2018180237
29. Schwalbe N, Wahl B. Artificial intelligence and the future of global health. Lancet. 2020;395(10236):1579-1586. doi:10.1016/S0140-6736(20)30226-9
30. Bai HX, Wang R, Xiong Z, et al. Artificial intelligence augmentation of radiologist performance in distinguishing COVID-19 from pneumonia of other origin at chest CT [published correction appears in Radiology. 2021 Apr;299(1):E225]. Radiology. 2020;296(3):E156-E165. doi:10.1148/radiol.2020201491
31. Li L, Qin L, Xu Z, et al. Using artificial intelligence to detect COVID-19 and community-acquired pneumonia based on pulmonary CT: evaluation of the diagnostic accuracy. Radiology. 2020;296(2):E65-E71. doi:10.1148/radiol.2020200905
32. Serag A, Ion-Margineanu A, Qureshi H, et al. Translational AI and deep learning in diagnostic pathology. Front Med (Lausanne). 2019;6:185. Published 2019 Oct 1. doi:10.3389/fmed.2019.00185

33. Wang D, Khosla A, Gargeya R, Irshad H, Beck AH. Deep learning for identifying metastatic breast cancer. ArXiv. 2016 June 18:arXiv:1606.05718v1. Published online June 18, 2016. Accessed September 15, 2021. http://arxiv.org/abs/1606.05718
34. Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatology Dermatol Surg. 2019;23(2):57-60. doi:10.4103/jdds.jdds_19_19
35. Mollalo A, Mao L, Rashidi P, Glass GE. A GIS-based artificial neural network model for spatial distribution of tuberculosis across the continental United States. Int J Environ Res Public Health. 2019;16(1):157. Published 2019 Jan 8. doi:10.3390/ijerph16010157
36. Haddawy P, Hasan AHMI, Kasantikul R, et al. Spatiotemporal Bayesian networks for malaria prediction. Artif Intell Med. 2018;84:127-138. doi:10.1016/j.artmed.2017.12.002
37. Laureano-Rosario AE, Duncan AP, Mendez-Lazaro PA, et al. Application of artificial neural networks for dengue fever outbreak predictions in the northwest coast of Yucatan, Mexico and San Juan, Puerto Rico. Trop Med Infect Dis. 2018;3(1):5. Published 2018 Jan 5. doi:10.3390/tropicalmed3010005
38. Buczak AL, Koshute PT, Babin SM, Feighner BH, Lewis SH. A data-driven epidemiological prediction method for dengue outbreaks using local and remote sensing data. BMC Med Inform Decis Mak. 2012;12:124. Published 2012 Nov 5. doi:10.1186/1472-6947-12-124
39. Scavuzzo JM, Trucco F, Espinosa M, et al. Modeling dengue vector population using remotely sensed data and machine learning. Acta Trop. 2018;185:167-175. doi:10.1016/j.actatropica.2018.05.003
40. Xue H, Bai Y, Hu H, Liang H. Influenza activity surveillance based on multiple regression model and artificial neural network. IEEE Access. 2018;6:563-575. doi:10.1109/ACCESS.2017.2771798
41. Jiang D, Hao M, Ding F, Fu J, Li M. Mapping the transmission risk of Zika virus using machine learning models. Acta Trop. 2018;185:391-399. doi:10.1016/j.actatropica.2018.06.021
42. Bragazzi NL, Dai H, Damiani G, Behzadifar M, Martini M, Wu J. How big data and artificial intelligence can help better manage the COVID-19 pandemic. Int J Environ Res Public Health. 2020;17(9):3176. Published 2020 May 2. doi:10.3390/ijerph17093176
43. Lake IR, Colón-González FJ, Barker GC, Morbey RA, Smith GE, Elliot AJ. Machine learning to refine decision making within a syndromic surveillance service. BMC Public Health. 2019;19(1):559. Published 2019 May 14. doi:10.1186/s12889-019-6916-9
44. Khan OF, Bebb G, Alimohamed NA. Artificial intelligence in medicine: what oncologists need to know about its potential-and its limitations. Oncol Exch. 2017;16(4):8-13. Accessed September 1, 2021. http://www.oncologyex.com/pdf/vol16_no4/feature_khan-ai.pdf
45. Badano LP, Keller DM, Muraru D, Torlasco C, Parati G. Artificial intelligence and cardiovascular imaging: A win-win combination. Anatol J Cardiol. 2020;24(4):214-223. doi:10.14744/AnatolJCardiol.2020.94491
46. Murdoch TB, Detsky AS. The inevitable application of big data to health care. JAMA. 2013;309(13):1351-1352. doi:10.1001/jama.2013.393
47. Greatbatch O, Garrett A, Snape K. The impact of artificial intelligence on the current and future practice of clinical cancer genomics. Genet Res (Camb). 2019;101:e9. Published 2019 Oct 31. doi:10.1017/S0016672319000089
48. Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44-56. doi:10.1038/s41591-018-0300-7
49. Vollmer S, Mateen BA, Bohner G, et al. Machine learning and artificial intelligence research for patient benefit: 20 critical questions on transparency, replicability, ethics, and effectiveness [published correction appears in BMJ. 2020 Apr 1;369:m1312]. BMJ. 2020;368:l6927. Published 2020 Mar 20. doi:10.1136/bmj.l6927
50. Lindsey R, Daluiski A, Chopra S, et al. Deep neural network improves fracture detection by clinicians. Proc Natl Acad Sci U S A. 2018;115(45):11591-11596. doi:10.1073/pnas.1806905115
51. Zech JR, Badgeley MA, Liu M, Costa AB, Titano JJ, Oermann EK. Variable generalization performance of a deep learning model to detect pneumonia in chest radiographs: a cross-sectional study. PLoS Med. 2018;15(11):e1002683. doi:10.1371/journal.pmed.1002683
52. Lakhani P, Sundaram B. Deep learning at chest radiography: automated classification of pulmonary tuberculosis by using convolutional neural networks. Radiology. 2017;284(2):574-582. doi:10.1148/radiol.2017162326
53. Rajpurkar P, Joshi A, Pareek A, et al. CheXpedition: investigating generalization challenges for translation of chest x-ray algorithms to the clinical setting. ArXiv. 2020 Feb 26:arXiv:2002.11379v2. Revised March 11, 2020. Accessed September 15, 2021. http://arxiv.org/abs/2002.11379
54. Salim M, Wåhlin E, Dembrower K, et al. External evaluation of 3 commercial artificial intelligence algorithms for independent assessment of screening mammograms. JAMA Oncol. 2020;6(10):1581-1588. doi:10.1001/jamaoncol.2020.3321
55. Arbabshirani MR, Fornwalt BK, Mongelluzzo GJ, et al. Advanced machine learning in action: identification of intracranial hemorrhage on computed tomography scans of the head with clinical workflow integration. NPJ Digit Med. 2018;1:9. doi:10.1038/s41746-017-0015-z
56. Sheth D, Giger ML. Artificial intelligence in the interpretation of breast cancer on MRI. J Magn Reson Imaging. 2020;51(5):1310-1324. doi:10.1002/jmri.26878
57. McKinney SM, Sieniek M, Godbole V, et al. International evaluation of an AI system for breast cancer screening. Nature. 2020;577(7788):89-94. doi:10.1038/s41586-019-1799-6
58. Booth AL, Abels E, McCaffrey P. Development of a prognostic model for mortality in COVID-19 infection using machine learning. Mod Pathol. 2021;34(3):522-531. doi:10.1038/s41379-020-00700-x
59. Xu B, Kocyigit D, Grimm R, Griffin BP, Cheng F. Applications of artificial intelligence in multimodality cardiovascular imaging: a state-of-the-art review. Prog Cardiovasc Dis. 2020;63(3):367-376. doi:10.1016/j.pcad.2020.03.003
60. Dey D, Slomka PJ, Leeson P, et al. Artificial intelligence in cardiovascular imaging: JACC state-of-the-art review. J Am Coll Cardiol. 2019;73(11):1317-1335. doi:10.1016/j.jacc.2018.12.054
61. Carewell Health. AI powered ECG diagnosis solutions. Accessed November 2, 2020. https://www.carewellhealth.com/products_aiecg.html
62. Strodthoff N, Strodthoff C. Detecting and interpreting myocardial infarction using fully convolutional neural networks. Physiol Meas. 2019;40(1):015001. doi:10.1088/1361-6579/aaf34d
63. Hannun AY, Rajpurkar P, Haghpanahi M, et al. Cardiologist-level arrhythmia detection and classification in ambulatory electrocardiograms using a deep neural network. Nat Med. 2019;25(1):65-69. doi:10.1038/s41591-018-0268-3
64. Kwon JM, Jeon KH, Kim HM, et al. Comparing the performance of artificial intelligence and conventional diagnosis criteria for detecting left ventricular hypertrophy using electrocardiography. Europace. 2020;22(3):412-419. doi:10.1093/europace/euz324

65. Eko. FDA clears Eko’s AFib and heart murmur detection algorithms, making it the first AI-powered stethoscope to screen for serious heart conditions [press release]. Published January 28, 2020. Accessed September 15, 2021. https://www.businesswire.com/news/home/20200128005232/en/FDA-Clears-Eko’s-AFib-and-Heart-Murmur-Detection-Algorithms-Making-It-the-First-AI-Powered-Stethoscope-to-Screen-for-Serious-Heart-Conditions
66. Cruz-Roa A, Gilmore H, Basavanhally A, et al. Accurate and reproducible invasive breast cancer detection in whole-slide images: a deep learning approach for quantifying tumor extent. Sci Rep. 2017;7:46450. doi:10.1038/srep46450
67. Acs B, Rantalainen M, Hartman J. Artificial intelligence as the next step towards precision pathology. J Intern Med. 2020;288(1):62-81. doi:10.1111/joim.13030
68. Mobadersany P, Yousefi S, Amgad M, et al. Predicting cancer outcomes from histology and genomics using convolutional networks. Proc Natl Acad Sci U S A. 2018;115(13):E2970-E2979. doi:10.1073/pnas.1717139115
69. Wang X, Janowczyk A, Zhou Y, et al. Prediction of recurrence in early stage non-small cell lung cancer using computer extracted nuclear features from digital H&E images. Sci Rep. 2017;7:13543. doi:10.1038/s41598-017-13773-7
70. Kulkarni PM, Robinson EJ, Pradhan JS, et al. Deep learning based on standard H&E images of primary melanoma tumors identifies patients at risk for visceral recurrence and death. Clin Cancer Res. 2020;26(5):1126-1134. doi:10.1158/1078-0432.CCR-19-1495
71. Du XL, Li WB, Hu BJ. Application of artificial intelligence in ophthalmology. Int J Ophthalmol. 2018;11(9):1555-1561. doi:10.18240/ijo.2018.09.21
72. Gunasekeran DV, Wong TY. Artificial intelligence in ophthalmology in 2020: a technology on the cusp for translation and implementation. Asia Pac J Ophthalmol (Phila). 2020;9(2):61-66. doi:10.1097/01.APO.0000656984.56467.2c
73. Ting DSW, Pasquale LR, Peng L, et al. Artificial intelligence and deep learning in ophthalmology. Br J Ophthalmol. 2019;103(2):167-175. doi:10.1136/bjophthalmol-2018-313173
74. Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-2410. doi:10.1001/jama.2016.17216
75. US Food and Drug Administration. FDA permits marketing of artificial intelligence-based device to detect certain diabetes-related eye problems [press release]. Published April 11, 2018. Accessed September 15, 2021. https://www.fda.gov/news-events/press-announcements/fda-permits-marketing-artificial-intelligence-based-device-detect-certain-diabetes-related-eye
76. Long E, Chen J, Wu X, et al. Artificial intelligence manages congenital cataract with individualized prediction and telehealth computing. NPJ Digit Med. 2020;3:112. doi:10.1038/s41746-020-00319-x
77. De Fauw J, Ledsam JR, Romera-Paredes B, et al. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat Med. 2018;24(9):1342-1350. doi:10.1038/s41591-018-0107-6
78. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118. doi:10.1038/nature21056
79. Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
80. Brinker TJ, Hekler A, Enk AH, et al. A convolutional neural network trained with dermoscopic images performed on par with 145 dermatologists in a clinical melanoma image classification task. Eur J Cancer. 2019;111:148-154. doi:10.1016/j.ejca.2019.02.005
81. Haenssle HA, Fink C, Schneiderbauer R, et al. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists. Ann Oncol. 2018;29(8):1836-1842. doi:10.1093/annonc/mdy166
82. Li CX, Shen CB, Xue K, et al. Artificial intelligence in dermatology: past, present, and future. Chin Med J (Engl). 2019;132(17):2017-2020. doi:10.1097/CM9.0000000000000372
83. Tschandl P, Codella N, Akay BN, et al. Comparison of the accuracy of human readers versus machine-learning algorithms for pigmented skin lesion classification: an open, web-based, international, diagnostic study. Lancet Oncol. 2019;20(7):938-947. doi:10.1016/S1470-2045(19)30333-X
84. Han SS, Park I, Eun Chang SE, et al. Augmented intelligence dermatology: deep neural networks empower medical professionals in diagnosing skin cancer and predicting treatment options for 134 skin disorders. J Invest Dermatol. 2020;140(9):1753-1761. doi:10.1016/j.jid.2020.01.019
85. Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies [published correction appears in BMJ. 2020 Feb 25;368:m645]. BMJ. 2020;368:m127. Published 2020 Feb 10. doi:10.1136/bmj.m127
86. Chen YC, Ke WC, Chiu HW. Risk classification of cancer survival using ANN with gene expression data from multiple laboratories. Comput Biol Med. 2014;48:1-7. doi:10.1016/j.compbiomed.2014.02.006
87. Kim W, Kim KS, Lee JE, et al. Development of novel breast cancer recurrence prediction model using support vector machine. J Breast Cancer. 2012;15(2):230-238. doi:10.4048/jbc.2012.15.2.230
88. Merath K, Hyer JM, Mehta R, et al. Use of machine learning for prediction of patient risk of postoperative complications after liver, pancreatic, and colorectal surgery. J Gastrointest Surg. 2020;24(8):1843-1851. doi:10.1007/s11605-019-04338-2
89. Santos-García G, Varela G, Novoa N, Jiménez MF. Prediction of postoperative morbidity after lung resection using an artificial neural network ensemble. Artif Intell Med. 2004;30(1):61-69. doi:10.1016/S0933-3657(03)00059-9
90. Ibragimov B, Xing L. Segmentation of organs-at-risks in head and neck CT images using convolutional neural networks. Med Phys. 2017;44(2):547-557. doi:10.1002/mp.12045
91. Lou B, Doken S, Zhuang T, et al. An image-based deep learning framework for individualizing radiotherapy dose. Lancet Digit Health. 2019;1(3):e136-e147. doi:10.1016/S2589-7500(19)30058-5
92. Xu J, Yang P, Xue S, et al. Translating cancer genomics into precision medicine with artificial intelligence: applications, challenges and future perspectives. Hum Genet. 2019;138(2):109-124. doi:10.1007/s00439-019-01970-5
93. Patel NM, Michelini VV, Snell JM, et al. Enhancing next‐generation sequencing‐guided cancer care through cognitive computing. Oncologist. 2018;23(2):179-185. doi:10.1634/theoncologist.2017-0170
94. Le Berre C, Sandborn WJ, Aridhi S, et al. Application of artificial intelligence to gastroenterology and hepatology. Gastroenterology. 2020;158(1):76-94.e2. doi:10.1053/j.gastro.2019.08.058
95. Yang YJ, Bang CS. Application of artificial intelligence in gastroenterology. World J Gastroenterol. 2019;25(14):1666-1683. doi:10.3748/wjg.v25.i14.1666
96. Wang P, Berzin TM, Glissen Brown JR, et al. Real-time automatic detection system increases colonoscopic polyp and adenoma detection rates: a prospective randomised controlled study. Gut. 2019;68(10):1813-1819. doi:10.1136/gutjnl-2018-317500

97. Gupta R, Krishnam SP, Schaefer PW, Lev MH, Gonzalez RG. An East Coast perspective on artificial intelligence and machine learning: part 2: ischemic stroke imaging and triage. Neuroimaging Clin N Am. 2020;30(4):467-478. doi:10.1016/j.nic.2020.08.002
98. Beli M, Bobi V, Badža M, Šolaja N, Duri-Jovii M, Kosti VS. Artificial intelligence for assisting diagnostics and assessment of Parkinson’s disease—a review. Clin Neurol Neurosurg. 2019;184:105442. doi:10.1016/j.clineuro.2019.105442
99. An S, Kang C, Lee HW. Artificial intelligence and computational approaches for epilepsy. J Epilepsy Res. 2020;10(1):8-17. doi:10.14581/jer.20003
100. Pavel AM, Rennie JM, de Vries LS, et al. A machine-learning algorithm for neonatal seizure recognition: a multicentre, randomised, controlled trial. Lancet Child Adolesc Health. 2020;4(10):740-749. doi:10.1016/S2352-4642(20)30239-X
101. Afzal HMR, Luo S, Ramadan S, Lechner-Scott J. The emerging role of artificial intelligence in multiple sclerosis imaging [published online ahead of print, 2020 Oct 28]. Mult Scler. 2020;1352458520966298. doi:10.1177/1352458520966298
102. Bouton CE. Restoring movement in paralysis with a bioelectronic neural bypass approach: current state and future directions. Cold Spring Harb Perspect Med. 2019;9(11):a034306. doi:10.1101/cshperspect.a034306
103. Durstewitz D, Koppe G, Meyer-Lindenberg A. Deep neural networks in psychiatry. Mol Psychiatry. 2019;24(11):1583-1598. doi:10.1038/s41380-019-0365-9
104. Fonseka TM, Bhat V, Kennedy SH. The utility of artificial intelligence in suicide risk prediction and the management of suicidal behaviors. Aust N Z J Psychiatry. 2019;53(10):954-964. doi:10.1177/0004867419864428
105. Kessler RC, Hwang I, Hoffmire CA, et al. Developing a practical suicide risk prediction model for targeting high-risk patients in the Veterans Health Administration. Int J Methods Psychiatr Res. 2017;26(3):e1575. doi:10.1002/mpr.1575
106. Kessler RC, Bauer MS, Bishop TM, et al. Using administrative data to predict suicide after psychiatric hospitalization in the Veterans Health Administration System. Front Psychiatry. 2020;11:390. doi:10.3389/fpsyt.2020.00390
107. Kessler RC, van Loo HM, Wardenaar KJ, et al. Testing a machine-learning algorithm to predict the persistence and severity of major depressive disorder from baseline self-reports. Mol Psychiatry. 2016;21(10):1366-1371. doi:10.1038/mp.2015.198
108. Horng S, Sontag DA, Halpern Y, Jernite Y, Shapiro NI, Nathanson LA. Creating an automated trigger for sepsis clinical decision support at emergency department triage using machine learning. PLoS One. 2017;12(4):e0174708. doi:10.1371/journal.pone.0174708
109. Soffer S, Klang E, Barash Y, Grossman E, Zimlichman E. Predicting in-hospital mortality at admission to the medical ward: a big-data machine learning model. Am J Med. 2021;134(2):227-234.e4. doi:10.1016/j.amjmed.2020.07.014
110. Labovitz DL, Shafner L, Reyes Gil M, Virmani D, Hanina A. Using artificial intelligence to reduce the risk of nonadherence in patients on anticoagulation therapy. Stroke. 2017;48(5):1416-1419. doi:10.1161/STROKEAHA.116.016281
111. Forlenza GP. Use of artificial intelligence to improve diabetes outcomes in patients using multiple daily injections therapy. Diabetes Technol Ther. 2019;21(S2):S24-S28. doi:10.1089/dia.2019.0077
112. Poser CM. CT scan and the practice of neurology. Arch Neurol. 1977;34(2):132. doi:10.1001/archneur.1977.00500140086023
113. Angus DC. Randomized clinical trials of artificial intelligence. JAMA. 2020;323(11):1043-1045. doi:10.1001/jama.2020.1039
114. Topol EJ. Welcoming new guidelines for AI clinical research. Nat Med. 2020;26(9):1318-1320. doi:10.1038/s41591-020-1042-x
115. Collins GS, Moons KGM. Reporting of artificial intelligence prediction models. Lancet. 2019;393(10181):1577-1579. doi:10.1016/S0140-6736(19)30037-6
116. Cruz Rivera S, Liu X, Chan AW, et al. Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension. Nat Med. 2020;26(9):1351-1363. doi:10.1038/s41591-020-1037-7
117. Liu X, Cruz Rivera S, Moher D, Calvert MJ, Denniston AK; SPIRIT-AI and CONSORT-AI Working Group. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension. Nat Med. 2020;26(9):1364-1374. doi:10.1038/s41591-020-1034-x
118. McCulloch WS, Pitts W. A logical calculus of the ideas immanent in nervous activity. Bull Math Biophys. 1943;5(4):115-133. doi:10.1007/BF02478259
119. Samuel AL. Some studies in machine learning using the game of Checkers. IBM J Res Dev. 1959;3(3):535-554. Accessed September 15, 2021. https://citeseerx.ist.psu.edu/viewdoc/summary?doi=10.1.1.368.2254
120. Sonoda M, Takano M, Miyahara J, Kato H. Computed radiography utilizing scanning laser stimulated luminescence. Radiology. 1983;148(3):833-838. doi:10.1148/radiology.148.3.6878707
121. Dechter R. Learning while searching in constraint-satisfaction-problems. AAAI’86: proceedings of the fifth AAAI national conference on artificial intelligence. Published 1986. Accessed September 15, 2021. https://www.aaai.org/Papers/AAAI/1986/AAAI86-029.pdf
122. Le Cun Y, Jackel LD, Boser B, et al. Handwritten digit recognition: applications of neural network chips and automatic learning. IEEE Commun Mag. 1989;27(11):41-46. doi:10.1109/35.41400
123. US Food and Drug Administration. FDA allows marketing of first whole slide imaging system for digital pathology [press release]. Published April 12, 2017. Accessed September 15, 2021. https://www.fda.gov/news-events/press-announcements/fda-allows-marketing-first-whole-slide-imaging-system-digital-pathology
1. Bini SA. Artificial intelligence, machine learning, deep learning, and cognitive computing: what do these terms mean and how will they impact health care? J Arthroplasty. 2018;33(8):2358-2361. doi:10.1016/j.arth.2018.02.067
2. Benjamens S, Dhunnoo P, Meskó B. The state of artificial intelligence-based FDA-approved medical devices and algorithms: an online database. NPJ Digit Med. 2020;3:118. doi:10.1038/s41746-020-00324-0
3. Viz. AI powered synchronized stroke care. Accessed September 15, 2021. https://www.viz.ai/ischemic-stroke
4. Buchanan M. The law of accelerating returns. Nat Phys. 2008;4(7):507. doi:10.1038/nphys1010
5. IBM Watson Health computes a pair of new solutions to improve healthcare data and security. Published September 10, 2015. Accessed October 21, 2020. https://www.techrepublic.com/article/ibm-watson-health-computes-a-pair-of-new-solutions-to-improve-healthcare-data-and-security
6. Borkowski AA, Kardani A, Mastorides SM, Thomas LB. Warfarin pharmacogenomics: recommendations with available patented clinical technologies. Recent Pat Biotechnol. 2014;8(2):110-115. doi:10.2174/1872208309666140904112003
7. Washington University in St. Louis. Warfarin dosing. Accessed September 15, 2021. http://www.warfarindosing.org/Source/Home.aspx
8. He J, Baxter SL, Xu J, Xu J, Zhou X, Zhang K. The practical implementation of artificial intelligence technologies in medicine. Nat Med. 2019;25(1):30-36. doi:10.1038/s41591-018-0307-0
9. Jiang F, Jiang Y, Zhi H, et al. Artificial intelligence in healthcare: past, present and future. Stroke Vasc Neurol. 2017;2(4):230-243. Published 2017 Jun 21. doi:10.1136/svn-2017-000101
10. Johnson KW, Torres Soto J, Glicksberg BS, et al. Artificial intelligence in cardiology. J Am Coll Cardiol. 2018;71(23):2668-2679. doi:10.1016/j.jacc.2018.03.521
11. Borkowski AA, Wilson CP, Borkowski SA, et al. Comparing artificial intelligence platforms for histopathologic cancer diagnosis. Fed Pract. 2019;36(10):456-463.
12. Cruz-Roa A, Gilmore H, Basavanhally A, et al. High-throughput adaptive sampling for whole-slide histopathology image analysis (HASHI) via convolutional neural networks: application to invasive breast cancer detection. PLoS One. 2018;13(5):e0196828. Published 2018 May 24. doi:10.1371/journal.pone.0196828
13. Nagendran M, Chen Y, Lovejoy CA, et al. Artificial intelligence versus clinicians: systematic review of design, reporting standards, and claims of deep learning studies. BMJ. 2020;368:m689. Published 2020 Mar 25. doi:10.1136/bmj.m689
14. Shimizu H, Nakayama KI. Artificial intelligence in oncology. Cancer Sci. 2020;111(5):1452-1460. doi:10.1111/cas.14377
15. Talebi-Liasi F, Markowitz O. Is artificial intelligence going to replace dermatologists?. Cutis. 2020;105(1):28-31.
16. Valliani AA, Ranti D, Oermann EK. Deep learning and neurology: a systematic review. Neurol Ther. 2019;8(2):351-365. doi:10.1007/s40120-019-00153-8
17. LeCun Y, Bengio Y, Hinton G. Deep learning. Nature. 2015;521(7553):436-444. doi:10.1038/nature14539
18. Graham S, Depp C, Lee EE, et al. Artificial intelligence for mental health and mental illnesses: an overview. Curr Psychiatry Rep. 2019;21(11):116. Published 2019 Nov 7. doi:10.1007/s11920-019-1094-0
19. Golas SB, Shibahara T, Agboola S, et al. A machine learning model to predict the risk of 30-day readmissions in patients with heart failure: a retrospective analysis of electronic medical records data. BMC Med Inform Decis Mak. 2018;18(1):44. Published 2018 Jun 22. doi:10.1186/s12911-018-0620-z
20. Mortazavi BJ, Downing NS, Bucholz EM, et al. Analysis of machine learning techniques for heart failure readmissions. Circ Cardiovasc Qual Outcomes. 2016;9(6):629-640. doi:10.1161/CIRCOUTCOMES.116.003039
21. Meyer-Bäse A, Morra L, Meyer-Bäse U, Pinker K. Current status and future perspectives of artificial intelligence in magnetic resonance breast imaging. Contrast Media Mol Imaging. 2020;2020:6805710. Published 2020 Aug 28. doi:10.1155/2020/6805710
22. Ehteshami Bejnordi B, Veta M, Johannes van Diest P, et al. Diagnostic assessment of deep learning algorithms for detection of lymph node metastases in women with breast cancer. JAMA. 2017;318(22):2199-2210. doi:10.1001/jama.2017.14585
23. Borkowski AA, Viswanadhan NA, Thomas LB, Guzman RD, Deland LA, Mastorides SM. Using artificial intelligence for COVID-19 chest X-ray diagnosis. Fed Pract. 2020;37(9):398-404. doi:10.12788/fp.0045
24. Coudray N, Ocampo PS, Sakellaropoulos T, et al. Classification and mutation prediction from non-small cell lung cancer histopathology images using deep learning. Nat Med. 2018;24(10):1559-1567. doi:10.1038/s41591-018-0177-5
25. Kermany DS, Goldbaum M, Cai W, et al. Identifying medical diagnoses and treatable diseases by image-based deep learning. Cell. 2018;172(5):1122-1131.e9. doi:10.1016/j.cell.2018.02.010
26. Liu X, Faes L, Kale AU, et al. A comparison of deep learning performance against health-care professionals in detecting diseases from medical imaging: a systematic review and meta-analysis. Lancet Digit Health. 2019;1(6):e271-e297. doi:10.1016/S2589-7500(19)30123-2
27. Nagpal K, Foote D, Liu Y, et al. Development and validation of a deep learning algorithm for improving Gleason scoring of prostate cancer [published correction appears in NPJ Digit Med. 2019 Nov 19;2:113]. NPJ Digit Med. 2019;2:48. Published 2019 Jun 7. doi:10.1038/s41746-019-0112-2
28. Nam JG, Park S, Hwang EJ, et al. Development and validation of deep learning-based automatic detection algorithm for malignant pulmonary nodules on chest radiographs. Radiology. 2019;290(1):218-228. doi:10.1148/radiol.2018180237
29. Schwalbe N, Wahl B. Artificial intelligence and the future of global health. Lancet. 2020;395(10236):1579-1586. doi:10.1016/S0140-6736(20)30226-9
30. Bai HX, Wang R, Xiong Z, et al. Artificial intelligence augmentation of radiologist performance in distinguishing COVID-19 from pneumonia of other origin at chest CT [published correction appears in Radiology. 2021 Apr;299(1):E225]. Radiology. 2020;296(3):E156-E165. doi:10.1148/radiol.2020201491
31. Li L, Qin L, Xu Z, et al. Using artificial intelligence to detect COVID-19 and community-acquired pneumonia based on pulmonary CT: evaluation of the diagnostic accuracy. Radiology. 2020;296(2):E65-E71. doi:10.1148/radiol.2020200905
32. Serag A, Ion-Margineanu A, Qureshi H, et al. Translational AI and deep learning in diagnostic pathology. Front Med (Lausanne). 2019;6:185. Published 2019 Oct 1. doi:10.3389/fmed.2019.00185

33. Wang D, Khosla A, Gargeya R, Irshad H, Beck AH. Deep learning for identifying metastatic breast cancer. ArXiv. 2016 June 18:arXiv:1606.05718v1. Published online June 18, 2016. Accessed September 15, 2021. http://arxiv.org/abs/1606.05718
34. Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatology Dermatol Surg. 2019;23(2):57-60. doi:10.4103/jdds.jdds_19_19
35. Mollalo A, Mao L, Rashidi P, Glass GE. A GIS-based artificial neural network model for spatial distribution of tuberculosis across the continental United States. Int J Environ Res Public Health. 2019;16(1):157. Published 2019 Jan 8. doi:10.3390/ijerph16010157
36. Haddawy P, Hasan AHMI, Kasantikul R, et al. Spatiotemporal Bayesian networks for malaria prediction. Artif Intell Med. 2018;84:127-138. doi:10.1016/j.artmed.2017.12.002
37. Laureano-Rosario AE, Duncan AP, Mendez-Lazaro PA, et al. Application of artificial neural networks for dengue fever outbreak predictions in the northwest coast of Yucatan, Mexico and San Juan, Puerto Rico. Trop Med Infect Dis. 2018;3(1):5. Published 2018 Jan 5. doi:10.3390/tropicalmed3010005
38. Buczak AL, Koshute PT, Babin SM, Feighner BH, Lewis SH. A data-driven epidemiological prediction method for dengue outbreaks using local and remote sensing data. BMC Med Inform Decis Mak. 2012;12:124. Published 2012 Nov 5. doi:10.1186/1472-6947-12-124
39. Scavuzzo JM, Trucco F, Espinosa M, et al. Modeling dengue vector population using remotely sensed data and machine learning. Acta Trop. 2018;185:167-175. doi:10.1016/j.actatropica.2018.05.003
40. Xue H, Bai Y, Hu H, Liang H. Influenza activity surveillance based on multiple regression model and artificial neural network. IEEE Access. 2018;6:563-575. doi:10.1109/ACCESS.2017.2771798
41. Jiang D, Hao M, Ding F, Fu J, Li M. Mapping the transmission risk of Zika virus using machine learning models. Acta Trop. 2018;185:391-399. doi:10.1016/j.actatropica.2018.06.021
42. Bragazzi NL, Dai H, Damiani G, Behzadifar M, Martini M, Wu J. How big data and artificial intelligence can help better manage the COVID-19 pandemic. Int J Environ Res Public Health. 2020;17(9):3176. Published 2020 May 2. doi:10.3390/ijerph17093176
43. Lake IR, Colón-González FJ, Barker GC, Morbey RA, Smith GE, Elliot AJ. Machine learning to refine decision making within a syndromic surveillance service. BMC Public Health. 2019;19(1):559. Published 2019 May 14. doi:10.1186/s12889-019-6916-9
44. Khan OF, Bebb G, Alimohamed NA. Artificial intelligence in medicine: what oncologists need to know about its potential-and its limitations. Oncol Exch. 2017;16(4):8-13. Accessed September 1, 2021. http://www.oncologyex.com/pdf/vol16_no4/feature_khan-ai.pdf
45. Badano LP, Keller DM, Muraru D, Torlasco C, Parati G. Artificial intelligence and cardiovascular imaging: A win-win combination. Anatol J Cardiol. 2020;24(4):214-223. doi:10.14744/AnatolJCardiol.2020.94491
46. Murdoch TB, Detsky AS. The inevitable application of big data to health care. JAMA. 2013;309(13):1351-1352. doi:10.1001/jama.2013.393
47. Greatbatch O, Garrett A, Snape K. The impact of artificial intelligence on the current and future practice of clinical cancer genomics. Genet Res (Camb). 2019;101:e9. Published 2019 Oct 31. doi:10.1017/S0016672319000089
48. Topol EJ. High-performance medicine: the convergence of human and artificial intelligence. Nat Med. 2019;25(1):44-56. doi:10.1038/s41591-018-0300-7
49. Vollmer S, Mateen BA, Bohner G, et al. Machine learning and artificial intelligence research for patient benefit: 20 critical questions on transparency, replicability, ethics, and effectiveness [published correction appears in BMJ. 2020 Apr 1;369:m1312]. BMJ. 2020;368:l6927. Published 2020 Mar 20. doi:10.1136/bmj.l6927
50. Lindsey R, Daluiski A, Chopra S, et al. Deep neural network improves fracture detection by clinicians. Proc Natl Acad Sci U S A. 2018;115(45):11591-11596. doi:10.1073/pnas.1806905115
51. Zech JR, Badgeley MA, Liu M, Costa AB, Titano JJ, Oermann EK. Variable generalization performance of a deep learning model to detect pneumonia in chest radiographs: a cross-sectional study. PLoS Med. 2018;15(11):e1002683. doi:10.1371/journal.pmed.1002683
52. Lakhani P, Sundaram B. Deep learning at chest radiography: automated classification of pulmonary tuberculosis by using convolutional neural networks. Radiology. 2017;284(2):574-582. doi:10.1148/radiol.2017162326
53. Rajpurkar P, Joshi A, Pareek A, et al. CheXpedition: investigating generalization challenges for translation of chest x-ray algorithms to the clinical setting. ArXiv. 2020 Feb 26:arXiv:2002.11379v2. Revised March 11, 2020. Accessed September 15, 2021. http://arxiv.org/abs/2002.11379
54. Salim M, Wåhlin E, Dembrower K, et al. External evaluation of 3 commercial artificial intelligence algorithms for independent assessment of screening mammograms. JAMA Oncol. 2020;6(10):1581-1588. doi:10.1001/jamaoncol.2020.3321
55. Arbabshirani MR, Fornwalt BK, Mongelluzzo GJ, et al. Advanced machine learning in action: identification of intracranial hemorrhage on computed tomography scans of the head with clinical workflow integration. NPJ Digit Med. 2018;1:9. doi:10.1038/s41746-017-0015-z
56. Sheth D, Giger ML. Artificial intelligence in the interpretation of breast cancer on MRI. J Magn Reson Imaging. 2020;51(5):1310-1324. doi:10.1002/jmri.26878
57. McKinney SM, Sieniek M, Godbole V, et al. International evaluation of an AI system for breast cancer screening. Nature. 2020;577(7788):89-94. doi:10.1038/s41586-019-1799-6
58. Booth AL, Abels E, McCaffrey P. Development of a prognostic model for mortality in COVID-19 infection using machine learning. Mod Pathol. 2021;34(3):522-531. doi:10.1038/s41379-020-00700-x
59. Xu B, Kocyigit D, Grimm R, Griffin BP, Cheng F. Applications of artificial intelligence in multimodality cardiovascular imaging: a state-of-the-art review. Prog Cardiovasc Dis. 2020;63(3):367-376. doi:10.1016/j.pcad.2020.03.003
60. Dey D, Slomka PJ, Leeson P, et al. Artificial intelligence in cardiovascular imaging: JACC state-of-the-art review. J Am Coll Cardiol. 2019;73(11):1317-1335. doi:10.1016/j.jacc.2018.12.054
61. Carewell Health. AI powered ECG diagnosis solutions. Accessed November 2, 2020. https://www.carewellhealth.com/products_aiecg.html
62. Strodthoff N, Strodthoff C. Detecting and interpreting myocardial infarction using fully convolutional neural networks. Physiol Meas. 2019;40(1):015001. doi:10.1088/1361-6579/aaf34d
63. Hannun AY, Rajpurkar P, Haghpanahi M, et al. Cardiologist-level arrhythmia detection and classification in ambulatory electrocardiograms using a deep neural network. Nat Med. 2019;25(1):65-69. doi:10.1038/s41591-018-0268-3
64. Kwon JM, Jeon KH, Kim HM, et al. Comparing the performance of artificial intelligence and conventional diagnosis criteria for detecting left ventricular hypertrophy using electrocardiography. Europace. 2020;22(3):412-419. doi:10.1093/europace/euz324

65. Eko. FDA clears Eko’s AFib and heart murmur detection algorithms, making it the first AI-powered stethoscope to screen for serious heart conditions [press release]. Published January 28, 2020. Accessed September 15, 2021. https://www.businesswire.com/news/home/20200128005232/en/FDA-Clears-Eko’s-AFib-and-Heart-Murmur-Detection-Algorithms-Making-It-the-First-AI-Powered-Stethoscope-to-Screen-for-Serious-Heart-Conditions
66. Cruz-Roa A, Gilmore H, Basavanhally A, et al. Accurate and reproducible invasive breast cancer detection in whole-slide images: a deep learning approach for quantifying tumor extent. Sci Rep. 2017;7:46450. doi:10.1038/srep46450
67. Acs B, Rantalainen M, Hartman J. Artificial intelligence as the next step towards precision pathology. J Intern Med. 2020;288(1):62-81. doi:10.1111/joim.13030
68. Mobadersany P, Yousefi S, Amgad M, et al. Predicting cancer outcomes from histology and genomics using convolutional networks. Proc Natl Acad Sci U S A. 2018;115(13):E2970-E2979. doi:10.1073/pnas.1717139115
69. Wang X, Janowczyk A, Zhou Y, et al. Prediction of recurrence in early stage non-small cell lung cancer using computer extracted nuclear features from digital H&E images. Sci Rep. 2017;7:13543. doi:10.1038/s41598-017-13773-7
70. Kulkarni PM, Robinson EJ, Pradhan JS, et al. Deep learning based on standard H&E images of primary melanoma tumors identifies patients at risk for visceral recurrence and death. Clin Cancer Res. 2020;26(5):1126-1134. doi:10.1158/1078-0432.CCR-19-1495
71. Du XL, Li WB, Hu BJ. Application of artificial intelligence in ophthalmology. Int J Ophthalmol. 2018;11(9):1555-1561. doi:10.18240/ijo.2018.09.21
72. Gunasekeran DV, Wong TY. Artificial intelligence in ophthalmology in 2020: a technology on the cusp for translation and implementation. Asia Pac J Ophthalmol (Phila). 2020;9(2):61-66. doi:10.1097/01.APO.0000656984.56467.2c
73. Ting DSW, Pasquale LR, Peng L, et al. Artificial intelligence and deep learning in ophthalmology. Br J Ophthalmol. 2019;103(2):167-175. doi:10.1136/bjophthalmol-2018-313173
74. Gulshan V, Peng L, Coram M, et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA. 2016;316(22):2402-2410. doi:10.1001/jama.2016.17216
75. US Food and Drug Administration. FDA permits marketing of artificial intelligence-based device to detect certain diabetes-related eye problems [press release]. Published April 11, 2018. Accessed September 15, 2021. https://www.fda.gov/news-events/press-announcements/fda-permits-marketing-artificial-intelligence-based-device-detect-certain-diabetes-related-eye
76. Long E, Chen J, Wu X, et al. Artificial intelligence manages congenital cataract with individualized prediction and telehealth computing. NPJ Digit Med. 2020;3:112. doi:10.1038/s41746-020-00319-x
77. De Fauw J, Ledsam JR, Romera-Paredes B, et al. Clinically applicable deep learning for diagnosis and referral in retinal disease. Nat Med. 2018;24(9):1342-1350. doi:10.1038/s41591-018-0107-6
78. Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115-118. doi:10.1038/nature21056
79. Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
80. Brinker TJ, Hekler A, Enk AH, et al. A convolutional neural network trained with dermoscopic images performed on par with 145 dermatologists in a clinical melanoma image classification task. Eur J Cancer. 2019;111:148-154. doi:10.1016/j.ejca.2019.02.005
81. Haenssle HA, Fink C, Schneiderbauer R, et al. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists. Ann Oncol. 2018;29(8):1836-1842. doi:10.1093/annonc/mdy166
82. Li CX, Shen CB, Xue K, et al. Artificial intelligence in dermatology: past, present, and future. Chin Med J (Engl). 2019;132(17):2017-2020. doi:10.1097/CM9.0000000000000372
83. Tschandl P, Codella N, Akay BN, et al. Comparison of the accuracy of human readers versus machine-learning algorithms for pigmented skin lesion classification: an open, web-based, international, diagnostic study. Lancet Oncol. 2019;20(7):938-947. doi:10.1016/S1470-2045(19)30333-X
84. Han SS, Park I, Eun Chang SE, et al. Augmented intelligence dermatology: deep neural networks empower medical professionals in diagnosing skin cancer and predicting treatment options for 134 skin disorders. J Invest Dermatol. 2020;140(9):1753-1761. doi:10.1016/j.jid.2020.01.019
85. Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies [published correction appears in BMJ. 2020 Feb 25;368:m645]. BMJ. 2020;368:m127. Published 2020 Feb 10. doi:10.1136/bmj.m127
86. Chen YC, Ke WC, Chiu HW. Risk classification of cancer survival using ANN with gene expression data from multiple laboratories. Comput Biol Med. 2014;48:1-7. doi:10.1016/j.compbiomed.2014.02.006
87. Kim W, Kim KS, Lee JE, et al. Development of novel breast cancer recurrence prediction model using support vector machine. J Breast Cancer. 2012;15(2):230-238. doi:10.4048/jbc.2012.15.2.230
88. Merath K, Hyer JM, Mehta R, et al. Use of machine learning for prediction of patient risk of postoperative complications after liver, pancreatic, and colorectal surgery. J Gastrointest Surg. 2020;24(8):1843-1851. doi:10.1007/s11605-019-04338-2
89. Santos-García G, Varela G, Novoa N, Jiménez MF. Prediction of postoperative morbidity after lung resection using an artificial neural network ensemble. Artif Intell Med. 2004;30(1):61-69. doi:10.1016/S0933-3657(03)00059-9
90. Ibragimov B, Xing L. Segmentation of organs-at-risks in head and neck CT images using convolutional neural networks. Med Phys. 2017;44(2):547-557. doi:10.1002/mp.12045
91. Lou B, Doken S, Zhuang T, et al. An image-based deep learning framework for individualizing radiotherapy dose. Lancet Digit Health. 2019;1(3):e136-e147. doi:10.1016/S2589-7500(19)30058-5
92. Xu J, Yang P, Xue S, et al. Translating cancer genomics into precision medicine with artificial intelligence: applications, challenges and future perspectives. Hum Genet. 2019;138(2):109-124. doi:10.1007/s00439-019-01970-5
93. Patel NM, Michelini VV, Snell JM, et al. Enhancing next‐generation sequencing‐guided cancer care through cognitive computing. Oncologist. 2018;23(2):179-185. doi:10.1634/theoncologist.2017-0170
94. Le Berre C, Sandborn WJ, Aridhi S, et al. Application of artificial intelligence to gastroenterology and hepatology. Gastroenterology. 2020;158(1):76-94.e2. doi:10.1053/j.gastro.2019.08.058
95. Yang YJ, Bang CS. Application of artificial intelligence in gastroenterology. World J Gastroenterol. 2019;25(14):1666-1683. doi:10.3748/wjg.v25.i14.1666
96. Wang P, Berzin TM, Glissen Brown JR, et al. Real-time automatic detection system increases colonoscopic polyp and adenoma detection rates: a prospective randomised controlled study. Gut. 2019;68(10):1813-1819. doi:10.1136/gutjnl-2018-317500

97. Gupta R, Krishnam SP, Schaefer PW, Lev MH, Gonzalez RG. An East Coast perspective on artificial intelligence and machine learning: part 2: ischemic stroke imaging and triage. Neuroimaging Clin N Am. 2020;30(4):467-478. doi:10.1016/j.nic.2020.08.002
98. Beli M, Bobi V, Badža M, Šolaja N, Duri-Jovii M, Kosti VS. Artificial intelligence for assisting diagnostics and assessment of Parkinson’s disease—a review. Clin Neurol Neurosurg. 2019;184:105442. doi:10.1016/j.clineuro.2019.105442
99. An S, Kang C, Lee HW. Artificial intelligence and computational approaches for epilepsy. J Epilepsy Res. 2020;10(1):8-17. doi:10.14581/jer.20003
100. Pavel AM, Rennie JM, de Vries LS, et al. A machine-learning algorithm for neonatal seizure recognition: a multicentre, randomised, controlled trial. Lancet Child Adolesc Health. 2020;4(10):740-749. doi:10.1016/S2352-4642(20)30239-X
101. Afzal HMR, Luo S, Ramadan S, Lechner-Scott J. The emerging role of artificial intelligence in multiple sclerosis imaging [published online ahead of print, 2020 Oct 28]. Mult Scler. 2020;1352458520966298. doi:10.1177/1352458520966298
102. Bouton CE. Restoring movement in paralysis with a bioelectronic neural bypass approach: current state and future directions. Cold Spring Harb Perspect Med. 2019;9(11):a034306. doi:10.1101/cshperspect.a034306
103. Durstewitz D, Koppe G, Meyer-Lindenberg A. Deep neural networks in psychiatry. Mol Psychiatry. 2019;24(11):1583-1598. doi:10.1038/s41380-019-0365-9
104. Fonseka TM, Bhat V, Kennedy SH. The utility of artificial intelligence in suicide risk prediction and the management of suicidal behaviors. Aust N Z J Psychiatry. 2019;53(10):954-964. doi:10.1177/0004867419864428
105. Kessler RC, Hwang I, Hoffmire CA, et al. Developing a practical suicide risk prediction model for targeting high-risk patients in the Veterans Health Administration. Int J Methods Psychiatr Res. 2017;26(3):e1575. doi:10.1002/mpr.1575
106. Kessler RC, Bauer MS, Bishop TM, et al. Using administrative data to predict suicide after psychiatric hospitalization in the Veterans Health Administration System. Front Psychiatry. 2020;11:390. doi:10.3389/fpsyt.2020.00390
107. Kessler RC, van Loo HM, Wardenaar KJ, et al. Testing a machine-learning algorithm to predict the persistence and severity of major depressive disorder from baseline self-reports. Mol Psychiatry. 2016;21(10):1366-1371. doi:10.1038/mp.2015.198
108. Horng S, Sontag DA, Halpern Y, Jernite Y, Shapiro NI, Nathanson LA. Creating an automated trigger for sepsis clinical decision support at emergency department triage using machine learning. PLoS One. 2017;12(4):e0174708. doi:10.1371/journal.pone.0174708
109. Soffer S, Klang E, Barash Y, Grossman E, Zimlichman E. Predicting in-hospital mortality at admission to the medical ward: a big-data machine learning model. Am J Med. 2021;134(2):227-234.e4. doi:10.1016/j.amjmed.2020.07.014
110. Labovitz DL, Shafner L, Reyes Gil M, Virmani D, Hanina A. Using artificial intelligence to reduce the risk of nonadherence in patients on anticoagulation therapy. Stroke. 2017;48(5):1416-1419. doi:10.1161/STROKEAHA.116.016281
111. Forlenza GP. Use of artificial intelligence to improve diabetes outcomes in patients using multiple daily injections therapy. Diabetes Technol Ther. 2019;21(S2):S24-S28. doi:10.1089/dia.2019.0077
112. Poser CM. CT scan and the practice of neurology. Arch Neurol. 1977;34(2):132. doi:10.1001/archneur.1977.00500140086023
113. Angus DC. Randomized clinical trials of artificial intelligence. JAMA. 2020;323(11):1043-1045. doi:10.1001/jama.2020.1039
114. Topol EJ. Welcoming new guidelines for AI clinical research. Nat Med. 2020;26(9):1318-1320. doi:10.1038/s41591-020-1042-x
115. Collins GS, Moons KGM. Reporting of artificial intelligence prediction models. Lancet. 2019;393(10181):1577-1579. doi:10.1016/S0140-6736(19)30037-6
116. Cruz Rivera S, Liu X, Chan AW, et al. Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension. Nat Med. 2020;26(9):1351-1363. doi:10.1038/s41591-020-1037-7
117. Liu X, Cruz Rivera S, Moher D, Calvert MJ, Denniston AK; SPIRIT-AI and CONSORT-AI Working Group. Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension. Nat Med. 2020;26(9):1364-1374. doi:10.1038/s41591-020-1034-x
118. McCulloch WS, Pitts W. A logical calculus of the ideas immanent in nervous activity. Bull Math Biophys. 1943;5(4):115-133. doi:10.1007/BF02478259
119. Samuel AL. Some studies in machine learning using the game of Checkers. IBM J Res Dev. 1959;3(3):535-554. Accessed September 15, 2021. https://citeseerx.ist.psu.edu/viewdoc/summary?doi=10.1.1.368.2254
120. Sonoda M, Takano M, Miyahara J, Kato H. Computed radiography utilizing scanning laser stimulated luminescence. Radiology. 1983;148(3):833-838. doi:10.1148/radiology.148.3.6878707
121. Dechter R. Learning while searching in constraint-satisfaction-problems. AAAI’86: proceedings of the fifth AAAI national conference on artificial intelligence. Published 1986. Accessed September 15, 2021. https://www.aaai.org/Papers/AAAI/1986/AAAI86-029.pdf
122. Le Cun Y, Jackel LD, Boser B, et al. Handwritten digit recognition: applications of neural network chips and automatic learning. IEEE Commun Mag. 1989;27(11):41-46. doi:10.1109/35.41400
123. US Food and Drug Administration. FDA allows marketing of first whole slide imaging system for digital pathology [press release]. Published April 12, 2017. Accessed September 15, 2021. https://www.fda.gov/news-events/press-announcements/fda-allows-marketing-first-whole-slide-imaging-system-digital-pathology
COVID-19 has brought more complex, longer office visits
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
‘Residents’ Viewpoint’ revisited
We are currently republishing an installment of this column as part of our continuing celebration of Family Practice News’s 50th anniversary.
Bruce A. Bagley, MD, wrote the first batch of these columns, when he was chief resident in family medicine at St. Joseph’s Hospital, Syracuse, N.Y. Joseph E. Scherger, MD, was the second writer for Family Practice News’s monthly “Residents’ Viewpoint.” At the time Dr. Scherger became a columnist, he was a 26-year-old, 2nd-year family practice resident at the Family Medical Center, University Hospital, University of Washington, Seattle.
Dr. Scherger’s first column was published on Feb. 5, 1977. We are republishing his “Residents’ Viewpoint” from June 15, 1977 (see below) and a new column by Victoria Persampiere, DO, who is currently a 2nd-year resident in the family medicine program at Abington Jefferson Health. (See “My experience as a family medicine resident in 2021” after Dr. Scherger’s column.).
We hope you will enjoy comparing and contrasting the experiences of a resident practicing family medicine today to those of a resident practicing family medicine nearly 4½ decades ago.To learn about Dr. Scherger’s current practice and long career, you can read his profile on the cover of the September 2021 issue of Family Practice News or on MDedge.com/FamilyMedicine in our “Family Practice News 50th Anniversary” section.
Art of medicine or deception?
Originally published in Family Practice News on June 15, 1977.
In medical school I learned the science of medicine. There I diligently studied the basic sciences and gained a thorough understanding of the pathophysiology of disease. In the clinical years I learned to apply this knowledge to a wide variety of interesting patients who came to the academic center.
Yet, when I started my family practice residency, I lacked the ability to care for patients. Though I could take a thorough history, perform a complete physical examination, and diagnose and treat specific illnesses, I had little idea how to satisfy patients by meeting their needs.
The art of medicine is the nonscientific part of a successful doctor-patient interaction. For a doctor-patient interaction to be successful, not only must the illness be appropriately addressed, but both patient and physician must be satisfied.
In the university environment, the art of medicine often gets inadequate attention. Indeed, most academic physicians think that only scientific medicine exists and that patients should be satisfied with a sophisticated approach to their problems. Some patients are satisfied, but many are disgruntled. It is not unusual for a patient, after a $1,000 work-up, to go to a family physician or chiropractor for satisfaction.
I was eager to discover the art of medicine at its finest during my rotation away from the university in a rural community. During these 2 months I looked for the pearls of wisdom that allowed community physicians to be so successful. I found that a very explicit technique was used by some physicians to achieve not only satisfaction but adoration from their patients. Unfortunately, this technique is dishonest.
Early in my community experience I was impressed by how often patients told me a doctor had saved them. I heard such statements as “Dr. X saved my leg,” or “Dr. X saved my life.” I know that it does occur, but not as often as I was hearing it.
Investigating these statements I found such stories as, “One day l twisted my ankle very badly, and it became quite swollen. My doctor told me 1 could lose my leg from this but that he would take x-rays, put my leg in an Ace bandage, and give me crutches. In 3 days I was well. I am so thankful he saved my leg.”
And, “One day I had a temperature of 104. All of my muscles ached, my head hurt, and I had a terrible sore throat and cough. My doctor told me l could die from this, but he gave me a medicine and made me stay home. I was sick for about 2 weeks, but I got better. He saved my life.”
Is the art of medicine the art of deception? This horrifying thought actually came to me after hearing several such stories, but I learned that most of the physicians involved in such stories were not well respected by their colleagues.
I learned many honest techniques for successfully caring for patients. The several family physicians with whom I worked, all clinical instructors associated with my residency, were impeccably honest and taught me to combine compassion and efficiency.
Despite learning many positive techniques and having good role models, I left the community experience somewhat saddened by the lack of integrity that can exist in the profession. I was naive in believing that all the nonscientific aspects of medicine that made patients happy must be good.
By experiencing deception, I learned why quackery continues to flourish despite the widespread availability of honest medical care. Most significantly, I learned the importance of a sometimes frustrating humility; my patients with sprained ankles and influenza will not believe I saved their lives.
My experience as a family medicine resident in 2021
I did not get a medical school graduation; I was one of the many thousands of newly graduated students who simply left their 4th-year rotation sites one chilly day in March 2020 and just never went back. My medical school education didn’t end with me walking triumphantly across the stage – a first-generation college student finally achieving the greatest dream in her life. Instead, it ended with a Zoom “graduation” and a cross-country move from Georgia to Pennsylvania amidst the greatest pandemic in recent memory. To say my impostor syndrome was bad would be an understatement.
Residency in the COVID-19 era
The joy and the draw to family medicine for me has always been the broad scope of conditions that we see and treat. From day 1, however, much of my residency has been devoted to one very small subset of patients – those with COVID-19. At one point, our hospital was so strained that our family medicine program had to run a second inpatient service alongside our usual five-resident service team just to provide care to everybody. Patients were in the hallways. The ER was packed to the gills. We were sleepless, terrified, unvaccinated, and desperate to help our patients survive a disease that was incompletely understood, with very few tools in our toolbox to combat it.
I distinctly remember sitting in the workroom with a coresident of mine, our faces seemingly permanently lined from wearing N95s all shift, and saying to him, “I worry I will be a bad family medicine physician. I worry I haven’t seen enough, other than COVID.” It was midway through my intern year; the days were short, so I was driving to and from the hospital in chilly darkness. My patients, like many around the country, were doing poorly. Vaccines seemed like a promise too good to be true. Worst of all: Those of us who were interns, who had no triumphant podium moment to end our medical school education, were suffering with an intense sense of impostor syndrome, which was strengthened by every “there is nothing else we can offer your loved one at this time” conversation we had. My apprehension about not having seen a wider breadth of medicine during my training is a sentiment still widely shared by COVID-era residents.
Luckily, my coresident was supportive.
“We’re going to be great family medicine physicians,” he said. “We’re learning the hard stuff – the bread and butter of FM – up-front. You’ll see.”
In some ways, I think he was right. Clinical skills, empathy, humility, and forging strong relationships are at the center of every family medicine physician’s heart; my generation has had to learn these skills early and under pressure. Sometimes, there are no answers. Sometimes, the best thing a family doctor can do for a patient is to hear them, understand them, and hold their hand.
‘We watched Cinderella together’
Shortly after that conversation with my coresident, I had a particular case which moved me. This gentleman with intellectual disability and COVID had been declining steadily since his admission to the hospital. He was isolated from everybody he knew and loved, but it did not dampen his spirits. He was cheerful to every person who entered his room, clad in their shrouds of PPE, which more often than not felt more like mourning garb than protective wear. I remember very little about this patient’s clinical picture – the COVID, the superimposed pneumonia, the repeated intubations. What I do remember is he loved the Disney classic Cinderella. I knew this because I developed a very close relationship with his family during the course of his hospitalization. Amidst the torrential onslaught of patients, I made sure to call families every day – not because I wanted to, but because my mentors and attendings and coresidents had all drilled into me from day 1 that we are family medicine, and a large part of our role is to advocate for our patients, and to communicate with their loved ones. So I called. I learned a lot about him; his likes, his dislikes, his close bond with his siblings, and of course his lifelong love for Cinderella. On the last week of my ICU rotation, my patient passed peacefully. His nurse and I were bedside. We held his hand. We told him his family loved him. We watched Cinderella together on an iPad encased in protective plastic.
My next rotation was an outpatient one and it looked more like the “bread and butter” of family medicine. But as I whisked in and out of patient rooms, attending to patients with diabetes, with depression, with pain, I could not stop thinking about my hospitalized patients who my coresidents had assumed care of. Each exam room I entered, I rather morbidly thought “this patient could be next on our hospital service.” Without realizing it, I made more of an effort to get to know each patient holistically. I learned who they were as people. I found myself writing small, medically low-yield details in the chart: “Margaret loves to sing in her church choir;” “Katherine is a self-published author.”
I learned from my attendings. As I sat at the precepting table with them, observing their conversations about patients, their collective decades of experience were apparent.
“I’ve been seeing this patient every few weeks since I was a resident,” said one of my attendings.
“I don’t even see my parents that often,” I thought.
The depth of her relationship with, understanding of, and compassion for this patient struck me deeply. This was why I went into family medicine. My attending knew her patients; they were not faceless unknowns in a hospital gown to her. She would have known to play Cinderella for them in the end.
This is a unique time for trainees. We have been challenged, terrified, overwhelmed, and heartbroken. But at no point have we been isolated. We’ve had the generations of doctors before us to lead the way, to teach us the “hard stuff.” We’ve had senior residents to lean on, who have taken us aside and told us, “I can do the goals-of-care talk today; you need a break.” While the plague seems to have passed over our hospital for now, it has left behind a class of family medicine residents who are proud to carry on our specialty’s long tradition of compassionate, empathetic, lifelong care. “We care for all life stages, from cradle to grave,” says every family medicine physician.
My class, for better or for worse, has cared more often for patients in the twilight of their lives, and while it has been hard, I believe it has made us all better doctors. Now, when I hold a newborn in my arms for a well-child check, I am exceptionally grateful – for the opportunities I have been given, for new beginnings amidst so much sadness, and for the great privilege of being a family medicine physician. ■
Dr. Persampiere is a second-year resident in the family medicine residency program at Abington (Pa.) Jefferson Health. You can contact her directly at [email protected] or via [email protected].
We are currently republishing an installment of this column as part of our continuing celebration of Family Practice News’s 50th anniversary.
Bruce A. Bagley, MD, wrote the first batch of these columns, when he was chief resident in family medicine at St. Joseph’s Hospital, Syracuse, N.Y. Joseph E. Scherger, MD, was the second writer for Family Practice News’s monthly “Residents’ Viewpoint.” At the time Dr. Scherger became a columnist, he was a 26-year-old, 2nd-year family practice resident at the Family Medical Center, University Hospital, University of Washington, Seattle.
Dr. Scherger’s first column was published on Feb. 5, 1977. We are republishing his “Residents’ Viewpoint” from June 15, 1977 (see below) and a new column by Victoria Persampiere, DO, who is currently a 2nd-year resident in the family medicine program at Abington Jefferson Health. (See “My experience as a family medicine resident in 2021” after Dr. Scherger’s column.).
We hope you will enjoy comparing and contrasting the experiences of a resident practicing family medicine today to those of a resident practicing family medicine nearly 4½ decades ago.To learn about Dr. Scherger’s current practice and long career, you can read his profile on the cover of the September 2021 issue of Family Practice News or on MDedge.com/FamilyMedicine in our “Family Practice News 50th Anniversary” section.
Art of medicine or deception?
Originally published in Family Practice News on June 15, 1977.
In medical school I learned the science of medicine. There I diligently studied the basic sciences and gained a thorough understanding of the pathophysiology of disease. In the clinical years I learned to apply this knowledge to a wide variety of interesting patients who came to the academic center.
Yet, when I started my family practice residency, I lacked the ability to care for patients. Though I could take a thorough history, perform a complete physical examination, and diagnose and treat specific illnesses, I had little idea how to satisfy patients by meeting their needs.
The art of medicine is the nonscientific part of a successful doctor-patient interaction. For a doctor-patient interaction to be successful, not only must the illness be appropriately addressed, but both patient and physician must be satisfied.
In the university environment, the art of medicine often gets inadequate attention. Indeed, most academic physicians think that only scientific medicine exists and that patients should be satisfied with a sophisticated approach to their problems. Some patients are satisfied, but many are disgruntled. It is not unusual for a patient, after a $1,000 work-up, to go to a family physician or chiropractor for satisfaction.
I was eager to discover the art of medicine at its finest during my rotation away from the university in a rural community. During these 2 months I looked for the pearls of wisdom that allowed community physicians to be so successful. I found that a very explicit technique was used by some physicians to achieve not only satisfaction but adoration from their patients. Unfortunately, this technique is dishonest.
Early in my community experience I was impressed by how often patients told me a doctor had saved them. I heard such statements as “Dr. X saved my leg,” or “Dr. X saved my life.” I know that it does occur, but not as often as I was hearing it.
Investigating these statements I found such stories as, “One day l twisted my ankle very badly, and it became quite swollen. My doctor told me 1 could lose my leg from this but that he would take x-rays, put my leg in an Ace bandage, and give me crutches. In 3 days I was well. I am so thankful he saved my leg.”
And, “One day I had a temperature of 104. All of my muscles ached, my head hurt, and I had a terrible sore throat and cough. My doctor told me l could die from this, but he gave me a medicine and made me stay home. I was sick for about 2 weeks, but I got better. He saved my life.”
Is the art of medicine the art of deception? This horrifying thought actually came to me after hearing several such stories, but I learned that most of the physicians involved in such stories were not well respected by their colleagues.
I learned many honest techniques for successfully caring for patients. The several family physicians with whom I worked, all clinical instructors associated with my residency, were impeccably honest and taught me to combine compassion and efficiency.
Despite learning many positive techniques and having good role models, I left the community experience somewhat saddened by the lack of integrity that can exist in the profession. I was naive in believing that all the nonscientific aspects of medicine that made patients happy must be good.
By experiencing deception, I learned why quackery continues to flourish despite the widespread availability of honest medical care. Most significantly, I learned the importance of a sometimes frustrating humility; my patients with sprained ankles and influenza will not believe I saved their lives.
My experience as a family medicine resident in 2021
I did not get a medical school graduation; I was one of the many thousands of newly graduated students who simply left their 4th-year rotation sites one chilly day in March 2020 and just never went back. My medical school education didn’t end with me walking triumphantly across the stage – a first-generation college student finally achieving the greatest dream in her life. Instead, it ended with a Zoom “graduation” and a cross-country move from Georgia to Pennsylvania amidst the greatest pandemic in recent memory. To say my impostor syndrome was bad would be an understatement.
Residency in the COVID-19 era
The joy and the draw to family medicine for me has always been the broad scope of conditions that we see and treat. From day 1, however, much of my residency has been devoted to one very small subset of patients – those with COVID-19. At one point, our hospital was so strained that our family medicine program had to run a second inpatient service alongside our usual five-resident service team just to provide care to everybody. Patients were in the hallways. The ER was packed to the gills. We were sleepless, terrified, unvaccinated, and desperate to help our patients survive a disease that was incompletely understood, with very few tools in our toolbox to combat it.
I distinctly remember sitting in the workroom with a coresident of mine, our faces seemingly permanently lined from wearing N95s all shift, and saying to him, “I worry I will be a bad family medicine physician. I worry I haven’t seen enough, other than COVID.” It was midway through my intern year; the days were short, so I was driving to and from the hospital in chilly darkness. My patients, like many around the country, were doing poorly. Vaccines seemed like a promise too good to be true. Worst of all: Those of us who were interns, who had no triumphant podium moment to end our medical school education, were suffering with an intense sense of impostor syndrome, which was strengthened by every “there is nothing else we can offer your loved one at this time” conversation we had. My apprehension about not having seen a wider breadth of medicine during my training is a sentiment still widely shared by COVID-era residents.
Luckily, my coresident was supportive.
“We’re going to be great family medicine physicians,” he said. “We’re learning the hard stuff – the bread and butter of FM – up-front. You’ll see.”
In some ways, I think he was right. Clinical skills, empathy, humility, and forging strong relationships are at the center of every family medicine physician’s heart; my generation has had to learn these skills early and under pressure. Sometimes, there are no answers. Sometimes, the best thing a family doctor can do for a patient is to hear them, understand them, and hold their hand.
‘We watched Cinderella together’
Shortly after that conversation with my coresident, I had a particular case which moved me. This gentleman with intellectual disability and COVID had been declining steadily since his admission to the hospital. He was isolated from everybody he knew and loved, but it did not dampen his spirits. He was cheerful to every person who entered his room, clad in their shrouds of PPE, which more often than not felt more like mourning garb than protective wear. I remember very little about this patient’s clinical picture – the COVID, the superimposed pneumonia, the repeated intubations. What I do remember is he loved the Disney classic Cinderella. I knew this because I developed a very close relationship with his family during the course of his hospitalization. Amidst the torrential onslaught of patients, I made sure to call families every day – not because I wanted to, but because my mentors and attendings and coresidents had all drilled into me from day 1 that we are family medicine, and a large part of our role is to advocate for our patients, and to communicate with their loved ones. So I called. I learned a lot about him; his likes, his dislikes, his close bond with his siblings, and of course his lifelong love for Cinderella. On the last week of my ICU rotation, my patient passed peacefully. His nurse and I were bedside. We held his hand. We told him his family loved him. We watched Cinderella together on an iPad encased in protective plastic.
My next rotation was an outpatient one and it looked more like the “bread and butter” of family medicine. But as I whisked in and out of patient rooms, attending to patients with diabetes, with depression, with pain, I could not stop thinking about my hospitalized patients who my coresidents had assumed care of. Each exam room I entered, I rather morbidly thought “this patient could be next on our hospital service.” Without realizing it, I made more of an effort to get to know each patient holistically. I learned who they were as people. I found myself writing small, medically low-yield details in the chart: “Margaret loves to sing in her church choir;” “Katherine is a self-published author.”
I learned from my attendings. As I sat at the precepting table with them, observing their conversations about patients, their collective decades of experience were apparent.
“I’ve been seeing this patient every few weeks since I was a resident,” said one of my attendings.
“I don’t even see my parents that often,” I thought.
The depth of her relationship with, understanding of, and compassion for this patient struck me deeply. This was why I went into family medicine. My attending knew her patients; they were not faceless unknowns in a hospital gown to her. She would have known to play Cinderella for them in the end.
This is a unique time for trainees. We have been challenged, terrified, overwhelmed, and heartbroken. But at no point have we been isolated. We’ve had the generations of doctors before us to lead the way, to teach us the “hard stuff.” We’ve had senior residents to lean on, who have taken us aside and told us, “I can do the goals-of-care talk today; you need a break.” While the plague seems to have passed over our hospital for now, it has left behind a class of family medicine residents who are proud to carry on our specialty’s long tradition of compassionate, empathetic, lifelong care. “We care for all life stages, from cradle to grave,” says every family medicine physician.
My class, for better or for worse, has cared more often for patients in the twilight of their lives, and while it has been hard, I believe it has made us all better doctors. Now, when I hold a newborn in my arms for a well-child check, I am exceptionally grateful – for the opportunities I have been given, for new beginnings amidst so much sadness, and for the great privilege of being a family medicine physician. ■
Dr. Persampiere is a second-year resident in the family medicine residency program at Abington (Pa.) Jefferson Health. You can contact her directly at [email protected] or via [email protected].
We are currently republishing an installment of this column as part of our continuing celebration of Family Practice News’s 50th anniversary.
Bruce A. Bagley, MD, wrote the first batch of these columns, when he was chief resident in family medicine at St. Joseph’s Hospital, Syracuse, N.Y. Joseph E. Scherger, MD, was the second writer for Family Practice News’s monthly “Residents’ Viewpoint.” At the time Dr. Scherger became a columnist, he was a 26-year-old, 2nd-year family practice resident at the Family Medical Center, University Hospital, University of Washington, Seattle.
Dr. Scherger’s first column was published on Feb. 5, 1977. We are republishing his “Residents’ Viewpoint” from June 15, 1977 (see below) and a new column by Victoria Persampiere, DO, who is currently a 2nd-year resident in the family medicine program at Abington Jefferson Health. (See “My experience as a family medicine resident in 2021” after Dr. Scherger’s column.).
We hope you will enjoy comparing and contrasting the experiences of a resident practicing family medicine today to those of a resident practicing family medicine nearly 4½ decades ago.To learn about Dr. Scherger’s current practice and long career, you can read his profile on the cover of the September 2021 issue of Family Practice News or on MDedge.com/FamilyMedicine in our “Family Practice News 50th Anniversary” section.
Art of medicine or deception?
Originally published in Family Practice News on June 15, 1977.
In medical school I learned the science of medicine. There I diligently studied the basic sciences and gained a thorough understanding of the pathophysiology of disease. In the clinical years I learned to apply this knowledge to a wide variety of interesting patients who came to the academic center.
Yet, when I started my family practice residency, I lacked the ability to care for patients. Though I could take a thorough history, perform a complete physical examination, and diagnose and treat specific illnesses, I had little idea how to satisfy patients by meeting their needs.
The art of medicine is the nonscientific part of a successful doctor-patient interaction. For a doctor-patient interaction to be successful, not only must the illness be appropriately addressed, but both patient and physician must be satisfied.
In the university environment, the art of medicine often gets inadequate attention. Indeed, most academic physicians think that only scientific medicine exists and that patients should be satisfied with a sophisticated approach to their problems. Some patients are satisfied, but many are disgruntled. It is not unusual for a patient, after a $1,000 work-up, to go to a family physician or chiropractor for satisfaction.
I was eager to discover the art of medicine at its finest during my rotation away from the university in a rural community. During these 2 months I looked for the pearls of wisdom that allowed community physicians to be so successful. I found that a very explicit technique was used by some physicians to achieve not only satisfaction but adoration from their patients. Unfortunately, this technique is dishonest.
Early in my community experience I was impressed by how often patients told me a doctor had saved them. I heard such statements as “Dr. X saved my leg,” or “Dr. X saved my life.” I know that it does occur, but not as often as I was hearing it.
Investigating these statements I found such stories as, “One day l twisted my ankle very badly, and it became quite swollen. My doctor told me 1 could lose my leg from this but that he would take x-rays, put my leg in an Ace bandage, and give me crutches. In 3 days I was well. I am so thankful he saved my leg.”
And, “One day I had a temperature of 104. All of my muscles ached, my head hurt, and I had a terrible sore throat and cough. My doctor told me l could die from this, but he gave me a medicine and made me stay home. I was sick for about 2 weeks, but I got better. He saved my life.”
Is the art of medicine the art of deception? This horrifying thought actually came to me after hearing several such stories, but I learned that most of the physicians involved in such stories were not well respected by their colleagues.
I learned many honest techniques for successfully caring for patients. The several family physicians with whom I worked, all clinical instructors associated with my residency, were impeccably honest and taught me to combine compassion and efficiency.
Despite learning many positive techniques and having good role models, I left the community experience somewhat saddened by the lack of integrity that can exist in the profession. I was naive in believing that all the nonscientific aspects of medicine that made patients happy must be good.
By experiencing deception, I learned why quackery continues to flourish despite the widespread availability of honest medical care. Most significantly, I learned the importance of a sometimes frustrating humility; my patients with sprained ankles and influenza will not believe I saved their lives.
My experience as a family medicine resident in 2021
I did not get a medical school graduation; I was one of the many thousands of newly graduated students who simply left their 4th-year rotation sites one chilly day in March 2020 and just never went back. My medical school education didn’t end with me walking triumphantly across the stage – a first-generation college student finally achieving the greatest dream in her life. Instead, it ended with a Zoom “graduation” and a cross-country move from Georgia to Pennsylvania amidst the greatest pandemic in recent memory. To say my impostor syndrome was bad would be an understatement.
Residency in the COVID-19 era
The joy and the draw to family medicine for me has always been the broad scope of conditions that we see and treat. From day 1, however, much of my residency has been devoted to one very small subset of patients – those with COVID-19. At one point, our hospital was so strained that our family medicine program had to run a second inpatient service alongside our usual five-resident service team just to provide care to everybody. Patients were in the hallways. The ER was packed to the gills. We were sleepless, terrified, unvaccinated, and desperate to help our patients survive a disease that was incompletely understood, with very few tools in our toolbox to combat it.
I distinctly remember sitting in the workroom with a coresident of mine, our faces seemingly permanently lined from wearing N95s all shift, and saying to him, “I worry I will be a bad family medicine physician. I worry I haven’t seen enough, other than COVID.” It was midway through my intern year; the days were short, so I was driving to and from the hospital in chilly darkness. My patients, like many around the country, were doing poorly. Vaccines seemed like a promise too good to be true. Worst of all: Those of us who were interns, who had no triumphant podium moment to end our medical school education, were suffering with an intense sense of impostor syndrome, which was strengthened by every “there is nothing else we can offer your loved one at this time” conversation we had. My apprehension about not having seen a wider breadth of medicine during my training is a sentiment still widely shared by COVID-era residents.
Luckily, my coresident was supportive.
“We’re going to be great family medicine physicians,” he said. “We’re learning the hard stuff – the bread and butter of FM – up-front. You’ll see.”
In some ways, I think he was right. Clinical skills, empathy, humility, and forging strong relationships are at the center of every family medicine physician’s heart; my generation has had to learn these skills early and under pressure. Sometimes, there are no answers. Sometimes, the best thing a family doctor can do for a patient is to hear them, understand them, and hold their hand.
‘We watched Cinderella together’
Shortly after that conversation with my coresident, I had a particular case which moved me. This gentleman with intellectual disability and COVID had been declining steadily since his admission to the hospital. He was isolated from everybody he knew and loved, but it did not dampen his spirits. He was cheerful to every person who entered his room, clad in their shrouds of PPE, which more often than not felt more like mourning garb than protective wear. I remember very little about this patient’s clinical picture – the COVID, the superimposed pneumonia, the repeated intubations. What I do remember is he loved the Disney classic Cinderella. I knew this because I developed a very close relationship with his family during the course of his hospitalization. Amidst the torrential onslaught of patients, I made sure to call families every day – not because I wanted to, but because my mentors and attendings and coresidents had all drilled into me from day 1 that we are family medicine, and a large part of our role is to advocate for our patients, and to communicate with their loved ones. So I called. I learned a lot about him; his likes, his dislikes, his close bond with his siblings, and of course his lifelong love for Cinderella. On the last week of my ICU rotation, my patient passed peacefully. His nurse and I were bedside. We held his hand. We told him his family loved him. We watched Cinderella together on an iPad encased in protective plastic.
My next rotation was an outpatient one and it looked more like the “bread and butter” of family medicine. But as I whisked in and out of patient rooms, attending to patients with diabetes, with depression, with pain, I could not stop thinking about my hospitalized patients who my coresidents had assumed care of. Each exam room I entered, I rather morbidly thought “this patient could be next on our hospital service.” Without realizing it, I made more of an effort to get to know each patient holistically. I learned who they were as people. I found myself writing small, medically low-yield details in the chart: “Margaret loves to sing in her church choir;” “Katherine is a self-published author.”
I learned from my attendings. As I sat at the precepting table with them, observing their conversations about patients, their collective decades of experience were apparent.
“I’ve been seeing this patient every few weeks since I was a resident,” said one of my attendings.
“I don’t even see my parents that often,” I thought.
The depth of her relationship with, understanding of, and compassion for this patient struck me deeply. This was why I went into family medicine. My attending knew her patients; they were not faceless unknowns in a hospital gown to her. She would have known to play Cinderella for them in the end.
This is a unique time for trainees. We have been challenged, terrified, overwhelmed, and heartbroken. But at no point have we been isolated. We’ve had the generations of doctors before us to lead the way, to teach us the “hard stuff.” We’ve had senior residents to lean on, who have taken us aside and told us, “I can do the goals-of-care talk today; you need a break.” While the plague seems to have passed over our hospital for now, it has left behind a class of family medicine residents who are proud to carry on our specialty’s long tradition of compassionate, empathetic, lifelong care. “We care for all life stages, from cradle to grave,” says every family medicine physician.
My class, for better or for worse, has cared more often for patients in the twilight of their lives, and while it has been hard, I believe it has made us all better doctors. Now, when I hold a newborn in my arms for a well-child check, I am exceptionally grateful – for the opportunities I have been given, for new beginnings amidst so much sadness, and for the great privilege of being a family medicine physician. ■
Dr. Persampiere is a second-year resident in the family medicine residency program at Abington (Pa.) Jefferson Health. You can contact her directly at [email protected] or via [email protected].
Infectious disease pop quiz: Clinical challenges for the ObGyn
In this question-and-answer article (the first in a series), our objective is to reinforce for the clinician several practical points of management for common infectious diseases. The principal references for the answers to the questions are 2 textbook chapters written by Dr. Duff.1,2 Other pertinent references are included in the text.
1. What are the best tests for the diagnosis of congenital cytomegalovirus (CMV) infection?
When congenital CMV is suspected, if the patient is at least 15 weeks’ gestation, an amniocentesis should be performed to test for CMV DNA in the amniotic fluid using polymerase chain reaction (PCR) methodology. If the initial test is negative, amniocentesis should be repeated in approximately 4 weeks. Coincident with amniocentesis, a detailed ultrasound examination should be performed to search for findings suggestive of fetal injury, such as growth restriction, microcephaly, periventricular calcifications, hepatosplenomegaly, echogenic bowel, and serous effusions in the pleural space or abdomen.
2. Which major organisms cause urinary tract infections (UTIs) in women?
The most common causative organism is Escherichia coli, which is responsible for approximately 70% of all UTIs. Klebsiella pneumoniae and Proteus species are the 2 other aerobic gram-negative bacilli that are common uropathogens. In addition, 3 gram-positive cocci are important: enterococci, Staphylococcus saprophyticus, and group B streptococcus.
3. What are the major complications of pyelonephritis in pregnancy?
Pyelonephritis is an important cause of preterm labor, sepsis, and adult respiratory distress syndrome. Most cases of pyelonephritis develop as a result of an untreated or inadequately treated lower urinary tract infection.
4. What is the most ominous manifestation of congenital parvovirus infection, and what is the cause of this abnormality?
Hydrops fetalis is the most ominous complication of congenital parvovirus infection. The virus crosses the placenta and attacks red cell progenitor cells, resulting in an aplastic anemia. In addition, the virus may cause myocarditis that, in turn, may result in cardiac failure in the fetus.
5. What are the major manifestations of congenital rubella syndrome?
Rubella is one of the most highly teratogenic of all the viral infections, particularly when maternal infection occurs in the first trimester. Manifestations of congenital rubella include hearing deficits, cataracts, glaucoma, microcephaly, mental retardation, cardiac malformations such as patent ductus arteriosus and pulmonic stenosis, and growth restriction.
6. Which vaccines are contraindicated in pregnancy?
Live virus vaccines should not be used in pregnancy because of the possibility of teratogenic effects. Live agents include the measles, mumps, and rubella (MMR) vaccine; live influenza vaccine (FluMist); oral polio vaccine; BCG (bacille Calmette-Guerin) vaccine; yellow fever vaccine; and smallpox vaccine.
7. What is the most appropriate treatment for trichomonas infection in pregnancy?
Trichomonas infection should be treated with oral metronidazole 500 mg twice daily for 7 days. Metronidazole also can be given as a single oral 2-g dose. This treatment is not quite as effective as the multidose regimen, but it may be appropriate for patients who are not likely to be adherent with the longer course of treatment.
Resistance to metronidazole is rare; in such instances, oral tinidazole 2 g in a single dose may be effective.
8. For uncomplicated gonorrhea in a pregnant woman, what is the most appropriate treatment?
The current recommendation from the Centers for Disease Control and Prevention for treatment of uncomplicated gonorrhea is a single 500-mg intramuscular dose of ceftriaxone. For the patient who is opposed to an intramuscular injection, an alternative treatment is cefixime 800 mg orally. With either of these regimens, if chlamydia infection cannot be excluded, the pregnant patient also should receive azithromycin 1,000 mg orally in a single dose. In a nonpregnant patient, doxycycline 100 mg orally twice daily for 7 days should be used to cover for concurrent chlamydia infection.
In a patient with an allergy to β-lactam antibiotics, an alternative regimen for treatment of uncomplicated gonorrhea is intramuscular gentamicin 240 mg plus a single 2,000-mg dose of oral azithromycin. (St Cyr S, Barbee L, Workowski KA, et al. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1911-1916.) ●
1. Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
2. Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
In this question-and-answer article (the first in a series), our objective is to reinforce for the clinician several practical points of management for common infectious diseases. The principal references for the answers to the questions are 2 textbook chapters written by Dr. Duff.1,2 Other pertinent references are included in the text.
1. What are the best tests for the diagnosis of congenital cytomegalovirus (CMV) infection?
When congenital CMV is suspected, if the patient is at least 15 weeks’ gestation, an amniocentesis should be performed to test for CMV DNA in the amniotic fluid using polymerase chain reaction (PCR) methodology. If the initial test is negative, amniocentesis should be repeated in approximately 4 weeks. Coincident with amniocentesis, a detailed ultrasound examination should be performed to search for findings suggestive of fetal injury, such as growth restriction, microcephaly, periventricular calcifications, hepatosplenomegaly, echogenic bowel, and serous effusions in the pleural space or abdomen.
2. Which major organisms cause urinary tract infections (UTIs) in women?
The most common causative organism is Escherichia coli, which is responsible for approximately 70% of all UTIs. Klebsiella pneumoniae and Proteus species are the 2 other aerobic gram-negative bacilli that are common uropathogens. In addition, 3 gram-positive cocci are important: enterococci, Staphylococcus saprophyticus, and group B streptococcus.
3. What are the major complications of pyelonephritis in pregnancy?
Pyelonephritis is an important cause of preterm labor, sepsis, and adult respiratory distress syndrome. Most cases of pyelonephritis develop as a result of an untreated or inadequately treated lower urinary tract infection.
4. What is the most ominous manifestation of congenital parvovirus infection, and what is the cause of this abnormality?
Hydrops fetalis is the most ominous complication of congenital parvovirus infection. The virus crosses the placenta and attacks red cell progenitor cells, resulting in an aplastic anemia. In addition, the virus may cause myocarditis that, in turn, may result in cardiac failure in the fetus.
5. What are the major manifestations of congenital rubella syndrome?
Rubella is one of the most highly teratogenic of all the viral infections, particularly when maternal infection occurs in the first trimester. Manifestations of congenital rubella include hearing deficits, cataracts, glaucoma, microcephaly, mental retardation, cardiac malformations such as patent ductus arteriosus and pulmonic stenosis, and growth restriction.
6. Which vaccines are contraindicated in pregnancy?
Live virus vaccines should not be used in pregnancy because of the possibility of teratogenic effects. Live agents include the measles, mumps, and rubella (MMR) vaccine; live influenza vaccine (FluMist); oral polio vaccine; BCG (bacille Calmette-Guerin) vaccine; yellow fever vaccine; and smallpox vaccine.
7. What is the most appropriate treatment for trichomonas infection in pregnancy?
Trichomonas infection should be treated with oral metronidazole 500 mg twice daily for 7 days. Metronidazole also can be given as a single oral 2-g dose. This treatment is not quite as effective as the multidose regimen, but it may be appropriate for patients who are not likely to be adherent with the longer course of treatment.
Resistance to metronidazole is rare; in such instances, oral tinidazole 2 g in a single dose may be effective.
8. For uncomplicated gonorrhea in a pregnant woman, what is the most appropriate treatment?
The current recommendation from the Centers for Disease Control and Prevention for treatment of uncomplicated gonorrhea is a single 500-mg intramuscular dose of ceftriaxone. For the patient who is opposed to an intramuscular injection, an alternative treatment is cefixime 800 mg orally. With either of these regimens, if chlamydia infection cannot be excluded, the pregnant patient also should receive azithromycin 1,000 mg orally in a single dose. In a nonpregnant patient, doxycycline 100 mg orally twice daily for 7 days should be used to cover for concurrent chlamydia infection.
In a patient with an allergy to β-lactam antibiotics, an alternative regimen for treatment of uncomplicated gonorrhea is intramuscular gentamicin 240 mg plus a single 2,000-mg dose of oral azithromycin. (St Cyr S, Barbee L, Workowski KA, et al. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1911-1916.) ●
In this question-and-answer article (the first in a series), our objective is to reinforce for the clinician several practical points of management for common infectious diseases. The principal references for the answers to the questions are 2 textbook chapters written by Dr. Duff.1,2 Other pertinent references are included in the text.
1. What are the best tests for the diagnosis of congenital cytomegalovirus (CMV) infection?
When congenital CMV is suspected, if the patient is at least 15 weeks’ gestation, an amniocentesis should be performed to test for CMV DNA in the amniotic fluid using polymerase chain reaction (PCR) methodology. If the initial test is negative, amniocentesis should be repeated in approximately 4 weeks. Coincident with amniocentesis, a detailed ultrasound examination should be performed to search for findings suggestive of fetal injury, such as growth restriction, microcephaly, periventricular calcifications, hepatosplenomegaly, echogenic bowel, and serous effusions in the pleural space or abdomen.
2. Which major organisms cause urinary tract infections (UTIs) in women?
The most common causative organism is Escherichia coli, which is responsible for approximately 70% of all UTIs. Klebsiella pneumoniae and Proteus species are the 2 other aerobic gram-negative bacilli that are common uropathogens. In addition, 3 gram-positive cocci are important: enterococci, Staphylococcus saprophyticus, and group B streptococcus.
3. What are the major complications of pyelonephritis in pregnancy?
Pyelonephritis is an important cause of preterm labor, sepsis, and adult respiratory distress syndrome. Most cases of pyelonephritis develop as a result of an untreated or inadequately treated lower urinary tract infection.
4. What is the most ominous manifestation of congenital parvovirus infection, and what is the cause of this abnormality?
Hydrops fetalis is the most ominous complication of congenital parvovirus infection. The virus crosses the placenta and attacks red cell progenitor cells, resulting in an aplastic anemia. In addition, the virus may cause myocarditis that, in turn, may result in cardiac failure in the fetus.
5. What are the major manifestations of congenital rubella syndrome?
Rubella is one of the most highly teratogenic of all the viral infections, particularly when maternal infection occurs in the first trimester. Manifestations of congenital rubella include hearing deficits, cataracts, glaucoma, microcephaly, mental retardation, cardiac malformations such as patent ductus arteriosus and pulmonic stenosis, and growth restriction.
6. Which vaccines are contraindicated in pregnancy?
Live virus vaccines should not be used in pregnancy because of the possibility of teratogenic effects. Live agents include the measles, mumps, and rubella (MMR) vaccine; live influenza vaccine (FluMist); oral polio vaccine; BCG (bacille Calmette-Guerin) vaccine; yellow fever vaccine; and smallpox vaccine.
7. What is the most appropriate treatment for trichomonas infection in pregnancy?
Trichomonas infection should be treated with oral metronidazole 500 mg twice daily for 7 days. Metronidazole also can be given as a single oral 2-g dose. This treatment is not quite as effective as the multidose regimen, but it may be appropriate for patients who are not likely to be adherent with the longer course of treatment.
Resistance to metronidazole is rare; in such instances, oral tinidazole 2 g in a single dose may be effective.
8. For uncomplicated gonorrhea in a pregnant woman, what is the most appropriate treatment?
The current recommendation from the Centers for Disease Control and Prevention for treatment of uncomplicated gonorrhea is a single 500-mg intramuscular dose of ceftriaxone. For the patient who is opposed to an intramuscular injection, an alternative treatment is cefixime 800 mg orally. With either of these regimens, if chlamydia infection cannot be excluded, the pregnant patient also should receive azithromycin 1,000 mg orally in a single dose. In a nonpregnant patient, doxycycline 100 mg orally twice daily for 7 days should be used to cover for concurrent chlamydia infection.
In a patient with an allergy to β-lactam antibiotics, an alternative regimen for treatment of uncomplicated gonorrhea is intramuscular gentamicin 240 mg plus a single 2,000-mg dose of oral azithromycin. (St Cyr S, Barbee L, Workowski KA, et al. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1911-1916.) ●
1. Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
2. Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
1. Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
2. Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
Latest national suicide data released
including pandemic-related job loss, financial strain, and deteriorating mental health, according to new federal statistics.
The number of annual suicides in the United States increased steadily from 2003 through 2018, followed by a 2% decline between 2018 and 2019. There was concern that deaths due to suicide would increase in 2020, but this doesn’t appear to be the case.
The provisional numbers show 45,855 deaths by suicide in the United States in 2020 – 3% lower than in 2019 (47,511), and 5% below the 2018 peak of 48,344 suicides, report Sally Curtin, MA, and colleagues with the National Center for Health Statistics, part of the U.S. Centers for Disease Control and Prevention.
The data were published online Nov. 3 in the National Vital Statistics System (NVSS) Vital Statistics Rapid Release.
On a monthly basis, the number of suicides was lower in 2020 than in 2019 in March through October and December – with the largest drop happening in April 2020 at a time when deaths from COVID-19 were peaking, the authors note. In April 2020, suicide deaths were 14% lower than in April 2019 (3,468 vs. 4,029).
The provisional age-adjusted suicide rate was 3% lower in 2020 (13.5 per 100,000) than in 2019 (13.9 per 100,000). It was 2% lower among men (21.9 compared with 22.4), and 8% lower for women (5.5 compared with 6.0).
Suicide rates among younger adults aged 10 to 34 years rose slightly between 2019 and 2020 but was only significant in those 25 to 34, with a 5% increase between 2019 and 2020.
Individuals aged 35 to 74 years had significant declines in suicide with the largest drop in those aged 45 to 54 years and 55 to 64 years.
Women in all race and Hispanic-origin groups showed declines in suicide rates between 2019 and 2020, but the decline was significant only among non-Hispanic white women (10%).
Suicide rates declined for non-Hispanic white and non-Hispanic Asian men but increased among non-Hispanic black, non-Hispanic American Indian or Alaska Native, and Hispanic men.
This analysis is based on more than 99% of expected death records. Based on previous patterns between provisional and final data, these provisional findings are expected to be consistent with final 2020 data, the authors say.
The study had no commercial funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
including pandemic-related job loss, financial strain, and deteriorating mental health, according to new federal statistics.
The number of annual suicides in the United States increased steadily from 2003 through 2018, followed by a 2% decline between 2018 and 2019. There was concern that deaths due to suicide would increase in 2020, but this doesn’t appear to be the case.
The provisional numbers show 45,855 deaths by suicide in the United States in 2020 – 3% lower than in 2019 (47,511), and 5% below the 2018 peak of 48,344 suicides, report Sally Curtin, MA, and colleagues with the National Center for Health Statistics, part of the U.S. Centers for Disease Control and Prevention.
The data were published online Nov. 3 in the National Vital Statistics System (NVSS) Vital Statistics Rapid Release.
On a monthly basis, the number of suicides was lower in 2020 than in 2019 in March through October and December – with the largest drop happening in April 2020 at a time when deaths from COVID-19 were peaking, the authors note. In April 2020, suicide deaths were 14% lower than in April 2019 (3,468 vs. 4,029).
The provisional age-adjusted suicide rate was 3% lower in 2020 (13.5 per 100,000) than in 2019 (13.9 per 100,000). It was 2% lower among men (21.9 compared with 22.4), and 8% lower for women (5.5 compared with 6.0).
Suicide rates among younger adults aged 10 to 34 years rose slightly between 2019 and 2020 but was only significant in those 25 to 34, with a 5% increase between 2019 and 2020.
Individuals aged 35 to 74 years had significant declines in suicide with the largest drop in those aged 45 to 54 years and 55 to 64 years.
Women in all race and Hispanic-origin groups showed declines in suicide rates between 2019 and 2020, but the decline was significant only among non-Hispanic white women (10%).
Suicide rates declined for non-Hispanic white and non-Hispanic Asian men but increased among non-Hispanic black, non-Hispanic American Indian or Alaska Native, and Hispanic men.
This analysis is based on more than 99% of expected death records. Based on previous patterns between provisional and final data, these provisional findings are expected to be consistent with final 2020 data, the authors say.
The study had no commercial funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
including pandemic-related job loss, financial strain, and deteriorating mental health, according to new federal statistics.
The number of annual suicides in the United States increased steadily from 2003 through 2018, followed by a 2% decline between 2018 and 2019. There was concern that deaths due to suicide would increase in 2020, but this doesn’t appear to be the case.
The provisional numbers show 45,855 deaths by suicide in the United States in 2020 – 3% lower than in 2019 (47,511), and 5% below the 2018 peak of 48,344 suicides, report Sally Curtin, MA, and colleagues with the National Center for Health Statistics, part of the U.S. Centers for Disease Control and Prevention.
The data were published online Nov. 3 in the National Vital Statistics System (NVSS) Vital Statistics Rapid Release.
On a monthly basis, the number of suicides was lower in 2020 than in 2019 in March through October and December – with the largest drop happening in April 2020 at a time when deaths from COVID-19 were peaking, the authors note. In April 2020, suicide deaths were 14% lower than in April 2019 (3,468 vs. 4,029).
The provisional age-adjusted suicide rate was 3% lower in 2020 (13.5 per 100,000) than in 2019 (13.9 per 100,000). It was 2% lower among men (21.9 compared with 22.4), and 8% lower for women (5.5 compared with 6.0).
Suicide rates among younger adults aged 10 to 34 years rose slightly between 2019 and 2020 but was only significant in those 25 to 34, with a 5% increase between 2019 and 2020.
Individuals aged 35 to 74 years had significant declines in suicide with the largest drop in those aged 45 to 54 years and 55 to 64 years.
Women in all race and Hispanic-origin groups showed declines in suicide rates between 2019 and 2020, but the decline was significant only among non-Hispanic white women (10%).
Suicide rates declined for non-Hispanic white and non-Hispanic Asian men but increased among non-Hispanic black, non-Hispanic American Indian or Alaska Native, and Hispanic men.
This analysis is based on more than 99% of expected death records. Based on previous patterns between provisional and final data, these provisional findings are expected to be consistent with final 2020 data, the authors say.
The study had no commercial funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Are oncologists any better at facing their own mortality?
Douglas Flora, MD, an oncologist with St. Elizabeth Healthcare, in Edgewood, Ky., considers himself a deep empath. It’s one reason he became an oncologist.
But when he was diagnosed with kidney cancer in 2017, he was shocked at the places his brain took him. His mind fast-forwarded through treatment options, statistical probabilities, and anguish over his wife and children.
“It’s a very surreal experience,” Dr. Flora said. “In 20 seconds, you go from diagnostics to, ‘What videos will I have to film for my babies?’ “
He could be having a wonderful evening surrounded by friends, music, and beer. Then he would go to the restroom and the realization of what was lurking inside his would body hit him like a brick.
“It’s like the scene in the Harry Potter movies where the Dementors fly over,” he explained. “Everything feels dark. There’s no hope. Everything you thought was good is gone.”
Oncologists counsel patients through life-threatening diagnoses and frightening decisions every day, so one might think they’d be ready to confront their own diagnosis, treatment, and mortality better than anyone. But that’s not always the case.
Does their expertise equip them to navigate their diagnosis and treatment better than their patients? How does the emotional toll of their personal cancer journey change the way they interact with their patients?
Navigating the diagnosis and treatment
In January 2017, Karen Hendershott, MD, a breast surgical oncologist, felt a lump in her armpit while taking a shower. The blunt force of her fate came into view in an instant: It was almost certainly a locally advanced breast cancer that had spread to her lymph nodes and would require surgery, radiotherapy, and chemotherapy.
She said a few unprintable words and headed to work at St. Mary’s Hospital, in Tucson, Ariz., where her assumptions were confirmed.
Taylor Riall, MD, PhD, also suspected cancer.
Last December, Dr. Riall, a general surgeon and surgical oncologist at the University of Arizona Cancer Center, in Tucson, developed a persistent cough. An x-ray revealed a mass in her lung. Initially, she was misdiagnosed with a fungal infection and was given medication that made her skin peel off.
Doctors advised Dr. Riall to monitor her condition for another 6 months. But her knowledge of oncology made her think cancer, so she insisted on more tests. In June 2021, a biopsy confirmed she had lung cancer.
Having oncology expertise helped Dr. Riall and Dr. Hendershott recognize the signs of cancer and push for a diagnosis. But there are also downsides to being hyper-informed, Dr. Hendershott, said.
“I think sometimes knowing everything at once is harder vs. giving yourself time to wrap your mind around this and do it in baby steps,” she explained. “There weren’t any baby steps here.”
Still, oncology practitioners who are diagnosed with cancer are navigating a familiar landscape and are often buoyed by a support network of expert colleagues. That makes a huge difference psychologically, explained Shenitha Edwards, a pharmacy technician at Cancer Specialists of North Florida, in Jacksonville, who was diagnosed with breast cancer in July.
“I felt stronger and a little more ready to fight because I had resources, whereas my patients sometimes do not,” Ms. Edwards said. “I was connected with a lot of people who could help me make informed decisions, so I didn’t have to walk so much in fear.”
It can also prepare practitioners to make bold treatment choices. In Dr. Riall’s case, surgeons were reluctant to excise her tumor because they would have to remove the entire upper lobe of her lung, and she is a marathoner and triathlete. Still, because of her surgical oncology experience, Dr. Riall didn’t flinch at the prospect of a major operation.
“I was, like, ‘Look, just take it out.’ I’m less afraid to have cancer than I am to not know and let it grow,” said Dr. Riall, whose Peloton name is WhoNeeds2Lungs.
Similarly, Dr. Hendershott’s experience gave her the assurance to pursue a more intense strategy. “Because I had a really candid understanding of the risks and what the odds looked like, it helped me be more comfortable with a more aggressive approach,” she said. “There wasn’t a doubt in my mind, particularly [having] a 10-year-old child, that I wanted to do everything I could, and even do a couple of things that were still in clinical trials.”
Almost paradoxically, Mark Lewis’ oncology training gave him the courage to risk watching and waiting after finding benign growths in his parathyroid and malignant tumors in his pancreas. Dr. Lewis monitored the tumors amassing in his pancreas for 8 years. When some grew so large they threatened to metastasize to his liver, he underwent the Whipple procedure to remove the head of the pancreas, part of the small intestine, and the gallbladder.
“It was a bit of a gamble, but one that paid off and allowed me to get my career off the ground and have another child,” said Dr. Lewis, a gastrointestinal oncologist at Intermountain Healthcare, in Salt Lake City. Treating patients for nearly a decade also showed him how fortunate he was to have a slow-growing, operable cancer. That gratitude, he said, gave him mental strength to endure the ordeal.
Whether taking a more aggressive or minimalist approach to their own care, each practitioner’s decision was deeply personal and deeply informed by their oncology expertise.
Although research on this question is scarce, studies show that differences in end-of-life care may occur. According to a 2016 study published in JAMA, physicians choose significantly less intensive end-stage care in three of five categories — undergoing surgery, being admitted to the intensive care unit (ICU), and dying in the hospital — than the general U.S. population. The reason, the researchers posited, is because doctors know these eleventh-hour interventions are typically brutal and futile.
But these differences were fairly small, and a 2019 study published in JAMA Open Network found the opposite: Physicians with cancer were more likely to die in an ICU and receive chemotherapy in the last 6 months of life, suggesting a more aggressive approach to end-of-life care.
When it comes to their own long-term or curative cancer care, oncologists generally don’t seem to approach treatment differently than their patients. In a 2015 study, researchers compared two groups of people with early breast cancer — 46 physicians and 230 well-educated, nonmedically qualified patients — and found no differences in the choices the groups made about whether to undergo mastectomy, chemotherapy, radiotherapy, or breast reconstruction.
Still, no amount of oncology expertise can fully prepare a person for the emotional crucible of cancer.
“A very surreal experience”
Although the fear can become less intense and more manageable over time, it may never truly go away.
At first, despair dragged Flora into an abyss for 6 hours a night, then overcame him 10 times a day, then gripped him briefly at random moments. Four years later and cancer-free, the dread still returns.
Hendershott cried every time she got into her car and contemplated her prognosis. Now 47, she has about a 60% chance of being alive in 15 years, and the fear still hits her.
“I think it’s hard to understand the moments of sheer terror that you have at 2 AM when you’re confronting your own mortality,” she said. “The implications that has not just for you but more importantly for the people that you love and want to protect. That just kind of washes over you in waves that you don’t have much control over.”
Cancer, Riall felt, had smashed her life, but she figured out a way to help herself cope. Severe blood loss, chest tubes, and tests and needles ad nauseum left Riall feeling excruciatingly exhausted after her surgery and delayed her return to work. At the same time, she was passed over for a promotion. Frustrated and dejected, she took comfort in the memory of doing Kintsugi with her surgery residents. The Japanese art form involves shattering pottery with a hammer, fitting the fragments back together, and painting the cracks gold.
“My instinct as a surgeon is to pick up those pieces and put them back together so nobody sees it’s broken,” she reflected. But as a patient, she learned that an important part of recovery is to allow yourself to sit in a broken state and feel angry, miserable, and betrayed by your body. And then examine your shattered priorities and consider how you want to reassemble them.
For Barbara Buttin, MD, a gynecologic oncologist at Cancer Treatment Centers of America, in Chicago, Illinois, it wasn’t cancer that almost took her life. Rather, a near-death experience and life-threatening diagnosis made her a better, more empathetic cancer doctor — a refrain echoed by many oncologist-patients. Confronting her own mortality crystallized what matters in life. She uses that understanding to make sure she understands what matters to her patients ― what they care about most, what their greatest fear is, what is going to keep them up at night.
“We’re part of the same club”
Ultimately, when oncology practitioners become patients, it balances the in-control and vulnerable, the rational and emotional. And their patients respond positively.
In fall 2020, oncology nurse Jenn Adams, RN, turned 40 and underwent her first mammogram. Unexpectedly, it revealed invasive stage I cancer that would require a double mastectomy, chemotherapy, and a year of immunotherapy. A week after her diagnosis, she was scheduled to start a new job at Cancer Clinic, in Bryan, Tex. So, she asked her manager if she could become a patient and an employee.
Ms. Adams worked 5 days a week, but every Thursday at 2 PM, she sat next to her patients while her coworkers became her nurses. Her chemo port was implanted, she lost her hair, and she felt terrible along with her patients. “It just created this incredible bond,” said the mother of three.
Having cancer, Dr. Flora said, “was completely different than I had imagined. When I thought I was walking with [my patients] in the depths of their caves, I wasn’t even visiting their caves.” But, he added, it has also “let me connect with [patients] on a deeper level because we’re part of the same club. You can see their body language change when I share that. They almost relax, like, ‘Oh, this guy gets it. He does understand how terrified I am.’ And I do.”
When Dr. Flora’s patients are scanned, he gives them their results immediately, because he knows what it’s like to wait on tenterhooks. He tells his patients to text him anytime they’re afraid or depressed, which he admits isn’t great for his own mental health but believes is worth it.
Likewise, Dr. Hendershott can hold out her shoulder-length locks to reassure a crying patient that hair does grow back after chemo. She can describe her experience with hormone-blocking pills to allay the fears of a pharmaceutical skeptic.
This role equalizer fosters so much empathy that doctors sometimes find themselves being helped by their patients. When one of Dr. Flora’s patients heard he had cancer, she sent him an email that began. “A wise doctor once told me....” and repeated the advice he’d given her years before.
Dr. Lewis has a special bond with his patients because people who have pancreatic neuroendocrine tumors seek him out for treatment. “I’m getting to take care of people who, on some level, are like my kindred spirits,” he said. “So, I get to see their coping mechanisms and how they do.”
Ms. Edwards told some of her patients about her breast cancer diagnosis, and now they give each other high-fives and share words of encouragement. “I made it a big thing of mine to associate my patients as my family,” she said. “If you’ve learned to embrace love and love people, there’s nothing you wouldn’t do for people. I’ve chosen that to be my practice when I’m dealing with all of my patients.”
Ms. Adams is on a similar mission. She joined a group of moms with cancer so she can receive guidance and then become a guide for others. “I feel like that’s what I want to be at my cancer practice,” she said, “so [my patients] have someone to say, ‘I’m gonna walk alongside you because I’ve been there.’ “
That transformation has made all the heartbreaking moments worth it, Ms. Adams said. “I love the oncology nurse that I get to be now because of my diagnosis. I don’t love the diagnosis. But I love the way it’s changed what I do.”
A version of this article first appeared on Medscape.com.
Douglas Flora, MD, an oncologist with St. Elizabeth Healthcare, in Edgewood, Ky., considers himself a deep empath. It’s one reason he became an oncologist.
But when he was diagnosed with kidney cancer in 2017, he was shocked at the places his brain took him. His mind fast-forwarded through treatment options, statistical probabilities, and anguish over his wife and children.
“It’s a very surreal experience,” Dr. Flora said. “In 20 seconds, you go from diagnostics to, ‘What videos will I have to film for my babies?’ “
He could be having a wonderful evening surrounded by friends, music, and beer. Then he would go to the restroom and the realization of what was lurking inside his would body hit him like a brick.
“It’s like the scene in the Harry Potter movies where the Dementors fly over,” he explained. “Everything feels dark. There’s no hope. Everything you thought was good is gone.”
Oncologists counsel patients through life-threatening diagnoses and frightening decisions every day, so one might think they’d be ready to confront their own diagnosis, treatment, and mortality better than anyone. But that’s not always the case.
Does their expertise equip them to navigate their diagnosis and treatment better than their patients? How does the emotional toll of their personal cancer journey change the way they interact with their patients?
Navigating the diagnosis and treatment
In January 2017, Karen Hendershott, MD, a breast surgical oncologist, felt a lump in her armpit while taking a shower. The blunt force of her fate came into view in an instant: It was almost certainly a locally advanced breast cancer that had spread to her lymph nodes and would require surgery, radiotherapy, and chemotherapy.
She said a few unprintable words and headed to work at St. Mary’s Hospital, in Tucson, Ariz., where her assumptions were confirmed.
Taylor Riall, MD, PhD, also suspected cancer.
Last December, Dr. Riall, a general surgeon and surgical oncologist at the University of Arizona Cancer Center, in Tucson, developed a persistent cough. An x-ray revealed a mass in her lung. Initially, she was misdiagnosed with a fungal infection and was given medication that made her skin peel off.
Doctors advised Dr. Riall to monitor her condition for another 6 months. But her knowledge of oncology made her think cancer, so she insisted on more tests. In June 2021, a biopsy confirmed she had lung cancer.
Having oncology expertise helped Dr. Riall and Dr. Hendershott recognize the signs of cancer and push for a diagnosis. But there are also downsides to being hyper-informed, Dr. Hendershott, said.
“I think sometimes knowing everything at once is harder vs. giving yourself time to wrap your mind around this and do it in baby steps,” she explained. “There weren’t any baby steps here.”
Still, oncology practitioners who are diagnosed with cancer are navigating a familiar landscape and are often buoyed by a support network of expert colleagues. That makes a huge difference psychologically, explained Shenitha Edwards, a pharmacy technician at Cancer Specialists of North Florida, in Jacksonville, who was diagnosed with breast cancer in July.
“I felt stronger and a little more ready to fight because I had resources, whereas my patients sometimes do not,” Ms. Edwards said. “I was connected with a lot of people who could help me make informed decisions, so I didn’t have to walk so much in fear.”
It can also prepare practitioners to make bold treatment choices. In Dr. Riall’s case, surgeons were reluctant to excise her tumor because they would have to remove the entire upper lobe of her lung, and she is a marathoner and triathlete. Still, because of her surgical oncology experience, Dr. Riall didn’t flinch at the prospect of a major operation.
“I was, like, ‘Look, just take it out.’ I’m less afraid to have cancer than I am to not know and let it grow,” said Dr. Riall, whose Peloton name is WhoNeeds2Lungs.
Similarly, Dr. Hendershott’s experience gave her the assurance to pursue a more intense strategy. “Because I had a really candid understanding of the risks and what the odds looked like, it helped me be more comfortable with a more aggressive approach,” she said. “There wasn’t a doubt in my mind, particularly [having] a 10-year-old child, that I wanted to do everything I could, and even do a couple of things that were still in clinical trials.”
Almost paradoxically, Mark Lewis’ oncology training gave him the courage to risk watching and waiting after finding benign growths in his parathyroid and malignant tumors in his pancreas. Dr. Lewis monitored the tumors amassing in his pancreas for 8 years. When some grew so large they threatened to metastasize to his liver, he underwent the Whipple procedure to remove the head of the pancreas, part of the small intestine, and the gallbladder.
“It was a bit of a gamble, but one that paid off and allowed me to get my career off the ground and have another child,” said Dr. Lewis, a gastrointestinal oncologist at Intermountain Healthcare, in Salt Lake City. Treating patients for nearly a decade also showed him how fortunate he was to have a slow-growing, operable cancer. That gratitude, he said, gave him mental strength to endure the ordeal.
Whether taking a more aggressive or minimalist approach to their own care, each practitioner’s decision was deeply personal and deeply informed by their oncology expertise.
Although research on this question is scarce, studies show that differences in end-of-life care may occur. According to a 2016 study published in JAMA, physicians choose significantly less intensive end-stage care in three of five categories — undergoing surgery, being admitted to the intensive care unit (ICU), and dying in the hospital — than the general U.S. population. The reason, the researchers posited, is because doctors know these eleventh-hour interventions are typically brutal and futile.
But these differences were fairly small, and a 2019 study published in JAMA Open Network found the opposite: Physicians with cancer were more likely to die in an ICU and receive chemotherapy in the last 6 months of life, suggesting a more aggressive approach to end-of-life care.
When it comes to their own long-term or curative cancer care, oncologists generally don’t seem to approach treatment differently than their patients. In a 2015 study, researchers compared two groups of people with early breast cancer — 46 physicians and 230 well-educated, nonmedically qualified patients — and found no differences in the choices the groups made about whether to undergo mastectomy, chemotherapy, radiotherapy, or breast reconstruction.
Still, no amount of oncology expertise can fully prepare a person for the emotional crucible of cancer.
“A very surreal experience”
Although the fear can become less intense and more manageable over time, it may never truly go away.
At first, despair dragged Flora into an abyss for 6 hours a night, then overcame him 10 times a day, then gripped him briefly at random moments. Four years later and cancer-free, the dread still returns.
Hendershott cried every time she got into her car and contemplated her prognosis. Now 47, she has about a 60% chance of being alive in 15 years, and the fear still hits her.
“I think it’s hard to understand the moments of sheer terror that you have at 2 AM when you’re confronting your own mortality,” she said. “The implications that has not just for you but more importantly for the people that you love and want to protect. That just kind of washes over you in waves that you don’t have much control over.”
Cancer, Riall felt, had smashed her life, but she figured out a way to help herself cope. Severe blood loss, chest tubes, and tests and needles ad nauseum left Riall feeling excruciatingly exhausted after her surgery and delayed her return to work. At the same time, she was passed over for a promotion. Frustrated and dejected, she took comfort in the memory of doing Kintsugi with her surgery residents. The Japanese art form involves shattering pottery with a hammer, fitting the fragments back together, and painting the cracks gold.
“My instinct as a surgeon is to pick up those pieces and put them back together so nobody sees it’s broken,” she reflected. But as a patient, she learned that an important part of recovery is to allow yourself to sit in a broken state and feel angry, miserable, and betrayed by your body. And then examine your shattered priorities and consider how you want to reassemble them.
For Barbara Buttin, MD, a gynecologic oncologist at Cancer Treatment Centers of America, in Chicago, Illinois, it wasn’t cancer that almost took her life. Rather, a near-death experience and life-threatening diagnosis made her a better, more empathetic cancer doctor — a refrain echoed by many oncologist-patients. Confronting her own mortality crystallized what matters in life. She uses that understanding to make sure she understands what matters to her patients ― what they care about most, what their greatest fear is, what is going to keep them up at night.
“We’re part of the same club”
Ultimately, when oncology practitioners become patients, it balances the in-control and vulnerable, the rational and emotional. And their patients respond positively.
In fall 2020, oncology nurse Jenn Adams, RN, turned 40 and underwent her first mammogram. Unexpectedly, it revealed invasive stage I cancer that would require a double mastectomy, chemotherapy, and a year of immunotherapy. A week after her diagnosis, she was scheduled to start a new job at Cancer Clinic, in Bryan, Tex. So, she asked her manager if she could become a patient and an employee.
Ms. Adams worked 5 days a week, but every Thursday at 2 PM, she sat next to her patients while her coworkers became her nurses. Her chemo port was implanted, she lost her hair, and she felt terrible along with her patients. “It just created this incredible bond,” said the mother of three.
Having cancer, Dr. Flora said, “was completely different than I had imagined. When I thought I was walking with [my patients] in the depths of their caves, I wasn’t even visiting their caves.” But, he added, it has also “let me connect with [patients] on a deeper level because we’re part of the same club. You can see their body language change when I share that. They almost relax, like, ‘Oh, this guy gets it. He does understand how terrified I am.’ And I do.”
When Dr. Flora’s patients are scanned, he gives them their results immediately, because he knows what it’s like to wait on tenterhooks. He tells his patients to text him anytime they’re afraid or depressed, which he admits isn’t great for his own mental health but believes is worth it.
Likewise, Dr. Hendershott can hold out her shoulder-length locks to reassure a crying patient that hair does grow back after chemo. She can describe her experience with hormone-blocking pills to allay the fears of a pharmaceutical skeptic.
This role equalizer fosters so much empathy that doctors sometimes find themselves being helped by their patients. When one of Dr. Flora’s patients heard he had cancer, she sent him an email that began. “A wise doctor once told me....” and repeated the advice he’d given her years before.
Dr. Lewis has a special bond with his patients because people who have pancreatic neuroendocrine tumors seek him out for treatment. “I’m getting to take care of people who, on some level, are like my kindred spirits,” he said. “So, I get to see their coping mechanisms and how they do.”
Ms. Edwards told some of her patients about her breast cancer diagnosis, and now they give each other high-fives and share words of encouragement. “I made it a big thing of mine to associate my patients as my family,” she said. “If you’ve learned to embrace love and love people, there’s nothing you wouldn’t do for people. I’ve chosen that to be my practice when I’m dealing with all of my patients.”
Ms. Adams is on a similar mission. She joined a group of moms with cancer so she can receive guidance and then become a guide for others. “I feel like that’s what I want to be at my cancer practice,” she said, “so [my patients] have someone to say, ‘I’m gonna walk alongside you because I’ve been there.’ “
That transformation has made all the heartbreaking moments worth it, Ms. Adams said. “I love the oncology nurse that I get to be now because of my diagnosis. I don’t love the diagnosis. But I love the way it’s changed what I do.”
A version of this article first appeared on Medscape.com.
Douglas Flora, MD, an oncologist with St. Elizabeth Healthcare, in Edgewood, Ky., considers himself a deep empath. It’s one reason he became an oncologist.
But when he was diagnosed with kidney cancer in 2017, he was shocked at the places his brain took him. His mind fast-forwarded through treatment options, statistical probabilities, and anguish over his wife and children.
“It’s a very surreal experience,” Dr. Flora said. “In 20 seconds, you go from diagnostics to, ‘What videos will I have to film for my babies?’ “
He could be having a wonderful evening surrounded by friends, music, and beer. Then he would go to the restroom and the realization of what was lurking inside his would body hit him like a brick.
“It’s like the scene in the Harry Potter movies where the Dementors fly over,” he explained. “Everything feels dark. There’s no hope. Everything you thought was good is gone.”
Oncologists counsel patients through life-threatening diagnoses and frightening decisions every day, so one might think they’d be ready to confront their own diagnosis, treatment, and mortality better than anyone. But that’s not always the case.
Does their expertise equip them to navigate their diagnosis and treatment better than their patients? How does the emotional toll of their personal cancer journey change the way they interact with their patients?
Navigating the diagnosis and treatment
In January 2017, Karen Hendershott, MD, a breast surgical oncologist, felt a lump in her armpit while taking a shower. The blunt force of her fate came into view in an instant: It was almost certainly a locally advanced breast cancer that had spread to her lymph nodes and would require surgery, radiotherapy, and chemotherapy.
She said a few unprintable words and headed to work at St. Mary’s Hospital, in Tucson, Ariz., where her assumptions were confirmed.
Taylor Riall, MD, PhD, also suspected cancer.
Last December, Dr. Riall, a general surgeon and surgical oncologist at the University of Arizona Cancer Center, in Tucson, developed a persistent cough. An x-ray revealed a mass in her lung. Initially, she was misdiagnosed with a fungal infection and was given medication that made her skin peel off.
Doctors advised Dr. Riall to monitor her condition for another 6 months. But her knowledge of oncology made her think cancer, so she insisted on more tests. In June 2021, a biopsy confirmed she had lung cancer.
Having oncology expertise helped Dr. Riall and Dr. Hendershott recognize the signs of cancer and push for a diagnosis. But there are also downsides to being hyper-informed, Dr. Hendershott, said.
“I think sometimes knowing everything at once is harder vs. giving yourself time to wrap your mind around this and do it in baby steps,” she explained. “There weren’t any baby steps here.”
Still, oncology practitioners who are diagnosed with cancer are navigating a familiar landscape and are often buoyed by a support network of expert colleagues. That makes a huge difference psychologically, explained Shenitha Edwards, a pharmacy technician at Cancer Specialists of North Florida, in Jacksonville, who was diagnosed with breast cancer in July.
“I felt stronger and a little more ready to fight because I had resources, whereas my patients sometimes do not,” Ms. Edwards said. “I was connected with a lot of people who could help me make informed decisions, so I didn’t have to walk so much in fear.”
It can also prepare practitioners to make bold treatment choices. In Dr. Riall’s case, surgeons were reluctant to excise her tumor because they would have to remove the entire upper lobe of her lung, and she is a marathoner and triathlete. Still, because of her surgical oncology experience, Dr. Riall didn’t flinch at the prospect of a major operation.
“I was, like, ‘Look, just take it out.’ I’m less afraid to have cancer than I am to not know and let it grow,” said Dr. Riall, whose Peloton name is WhoNeeds2Lungs.
Similarly, Dr. Hendershott’s experience gave her the assurance to pursue a more intense strategy. “Because I had a really candid understanding of the risks and what the odds looked like, it helped me be more comfortable with a more aggressive approach,” she said. “There wasn’t a doubt in my mind, particularly [having] a 10-year-old child, that I wanted to do everything I could, and even do a couple of things that were still in clinical trials.”
Almost paradoxically, Mark Lewis’ oncology training gave him the courage to risk watching and waiting after finding benign growths in his parathyroid and malignant tumors in his pancreas. Dr. Lewis monitored the tumors amassing in his pancreas for 8 years. When some grew so large they threatened to metastasize to his liver, he underwent the Whipple procedure to remove the head of the pancreas, part of the small intestine, and the gallbladder.
“It was a bit of a gamble, but one that paid off and allowed me to get my career off the ground and have another child,” said Dr. Lewis, a gastrointestinal oncologist at Intermountain Healthcare, in Salt Lake City. Treating patients for nearly a decade also showed him how fortunate he was to have a slow-growing, operable cancer. That gratitude, he said, gave him mental strength to endure the ordeal.
Whether taking a more aggressive or minimalist approach to their own care, each practitioner’s decision was deeply personal and deeply informed by their oncology expertise.
Although research on this question is scarce, studies show that differences in end-of-life care may occur. According to a 2016 study published in JAMA, physicians choose significantly less intensive end-stage care in three of five categories — undergoing surgery, being admitted to the intensive care unit (ICU), and dying in the hospital — than the general U.S. population. The reason, the researchers posited, is because doctors know these eleventh-hour interventions are typically brutal and futile.
But these differences were fairly small, and a 2019 study published in JAMA Open Network found the opposite: Physicians with cancer were more likely to die in an ICU and receive chemotherapy in the last 6 months of life, suggesting a more aggressive approach to end-of-life care.
When it comes to their own long-term or curative cancer care, oncologists generally don’t seem to approach treatment differently than their patients. In a 2015 study, researchers compared two groups of people with early breast cancer — 46 physicians and 230 well-educated, nonmedically qualified patients — and found no differences in the choices the groups made about whether to undergo mastectomy, chemotherapy, radiotherapy, or breast reconstruction.
Still, no amount of oncology expertise can fully prepare a person for the emotional crucible of cancer.
“A very surreal experience”
Although the fear can become less intense and more manageable over time, it may never truly go away.
At first, despair dragged Flora into an abyss for 6 hours a night, then overcame him 10 times a day, then gripped him briefly at random moments. Four years later and cancer-free, the dread still returns.
Hendershott cried every time she got into her car and contemplated her prognosis. Now 47, she has about a 60% chance of being alive in 15 years, and the fear still hits her.
“I think it’s hard to understand the moments of sheer terror that you have at 2 AM when you’re confronting your own mortality,” she said. “The implications that has not just for you but more importantly for the people that you love and want to protect. That just kind of washes over you in waves that you don’t have much control over.”
Cancer, Riall felt, had smashed her life, but she figured out a way to help herself cope. Severe blood loss, chest tubes, and tests and needles ad nauseum left Riall feeling excruciatingly exhausted after her surgery and delayed her return to work. At the same time, she was passed over for a promotion. Frustrated and dejected, she took comfort in the memory of doing Kintsugi with her surgery residents. The Japanese art form involves shattering pottery with a hammer, fitting the fragments back together, and painting the cracks gold.
“My instinct as a surgeon is to pick up those pieces and put them back together so nobody sees it’s broken,” she reflected. But as a patient, she learned that an important part of recovery is to allow yourself to sit in a broken state and feel angry, miserable, and betrayed by your body. And then examine your shattered priorities and consider how you want to reassemble them.
For Barbara Buttin, MD, a gynecologic oncologist at Cancer Treatment Centers of America, in Chicago, Illinois, it wasn’t cancer that almost took her life. Rather, a near-death experience and life-threatening diagnosis made her a better, more empathetic cancer doctor — a refrain echoed by many oncologist-patients. Confronting her own mortality crystallized what matters in life. She uses that understanding to make sure she understands what matters to her patients ― what they care about most, what their greatest fear is, what is going to keep them up at night.
“We’re part of the same club”
Ultimately, when oncology practitioners become patients, it balances the in-control and vulnerable, the rational and emotional. And their patients respond positively.
In fall 2020, oncology nurse Jenn Adams, RN, turned 40 and underwent her first mammogram. Unexpectedly, it revealed invasive stage I cancer that would require a double mastectomy, chemotherapy, and a year of immunotherapy. A week after her diagnosis, she was scheduled to start a new job at Cancer Clinic, in Bryan, Tex. So, she asked her manager if she could become a patient and an employee.
Ms. Adams worked 5 days a week, but every Thursday at 2 PM, she sat next to her patients while her coworkers became her nurses. Her chemo port was implanted, she lost her hair, and she felt terrible along with her patients. “It just created this incredible bond,” said the mother of three.
Having cancer, Dr. Flora said, “was completely different than I had imagined. When I thought I was walking with [my patients] in the depths of their caves, I wasn’t even visiting their caves.” But, he added, it has also “let me connect with [patients] on a deeper level because we’re part of the same club. You can see their body language change when I share that. They almost relax, like, ‘Oh, this guy gets it. He does understand how terrified I am.’ And I do.”
When Dr. Flora’s patients are scanned, he gives them their results immediately, because he knows what it’s like to wait on tenterhooks. He tells his patients to text him anytime they’re afraid or depressed, which he admits isn’t great for his own mental health but believes is worth it.
Likewise, Dr. Hendershott can hold out her shoulder-length locks to reassure a crying patient that hair does grow back after chemo. She can describe her experience with hormone-blocking pills to allay the fears of a pharmaceutical skeptic.
This role equalizer fosters so much empathy that doctors sometimes find themselves being helped by their patients. When one of Dr. Flora’s patients heard he had cancer, she sent him an email that began. “A wise doctor once told me....” and repeated the advice he’d given her years before.
Dr. Lewis has a special bond with his patients because people who have pancreatic neuroendocrine tumors seek him out for treatment. “I’m getting to take care of people who, on some level, are like my kindred spirits,” he said. “So, I get to see their coping mechanisms and how they do.”
Ms. Edwards told some of her patients about her breast cancer diagnosis, and now they give each other high-fives and share words of encouragement. “I made it a big thing of mine to associate my patients as my family,” she said. “If you’ve learned to embrace love and love people, there’s nothing you wouldn’t do for people. I’ve chosen that to be my practice when I’m dealing with all of my patients.”
Ms. Adams is on a similar mission. She joined a group of moms with cancer so she can receive guidance and then become a guide for others. “I feel like that’s what I want to be at my cancer practice,” she said, “so [my patients] have someone to say, ‘I’m gonna walk alongside you because I’ve been there.’ “
That transformation has made all the heartbreaking moments worth it, Ms. Adams said. “I love the oncology nurse that I get to be now because of my diagnosis. I don’t love the diagnosis. But I love the way it’s changed what I do.”
A version of this article first appeared on Medscape.com.
Tiny insects reveal some big secrets in cancer
Uncontrolled growth isn’t the only way cancers wreak havoc on the human body. These aggregations of freely dividing cells also release chemicals that can cause damage from a distance. But pinning down how they harm faraway healthy tissues isn’t straightforward.
Fortunately, biologists can turn to the tiny fruit fly to address some of these questions: This insect’s body is not as complex as ours in many ways, but we share important genes and organ functions.
Fruit flies already are a crucial and inexpensive animal for genetics research. Because their life span is about 7 weeks, investigators can track the effects of mutations across several generations in a short period. The animals also are proving useful for learning how chemicals released by malignant tumors can harm tissues in the body that are not near the cancer.
One recent lesson from the fruit flies involves the blood-brain barrier, which determines which molecules gain access to the brain. Researchers at the University of California, Berkeley, have found that malignant tumors in the tiny insects release interleukin 6 (IL-6), an inflammatory chemical that disrupts this important barrier. The investigators showed that the tumors act similarly in mice.
Even if cancer cells persisted, damage related to IL-6 could be diminished.
Fruit flies and mice are only distant relatives of each other and of humans, and the relevance of this discovery to human cancers has not been established. One hurdle is that IL-6 has many important, normal functions related to health. Researchers need to learn how to target only its unwanted blood-brain barrier effects.
A version of this article first appeared on Medscape.com.
Uncontrolled growth isn’t the only way cancers wreak havoc on the human body. These aggregations of freely dividing cells also release chemicals that can cause damage from a distance. But pinning down how they harm faraway healthy tissues isn’t straightforward.
Fortunately, biologists can turn to the tiny fruit fly to address some of these questions: This insect’s body is not as complex as ours in many ways, but we share important genes and organ functions.
Fruit flies already are a crucial and inexpensive animal for genetics research. Because their life span is about 7 weeks, investigators can track the effects of mutations across several generations in a short period. The animals also are proving useful for learning how chemicals released by malignant tumors can harm tissues in the body that are not near the cancer.
One recent lesson from the fruit flies involves the blood-brain barrier, which determines which molecules gain access to the brain. Researchers at the University of California, Berkeley, have found that malignant tumors in the tiny insects release interleukin 6 (IL-6), an inflammatory chemical that disrupts this important barrier. The investigators showed that the tumors act similarly in mice.
Even if cancer cells persisted, damage related to IL-6 could be diminished.
Fruit flies and mice are only distant relatives of each other and of humans, and the relevance of this discovery to human cancers has not been established. One hurdle is that IL-6 has many important, normal functions related to health. Researchers need to learn how to target only its unwanted blood-brain barrier effects.
A version of this article first appeared on Medscape.com.
Uncontrolled growth isn’t the only way cancers wreak havoc on the human body. These aggregations of freely dividing cells also release chemicals that can cause damage from a distance. But pinning down how they harm faraway healthy tissues isn’t straightforward.
Fortunately, biologists can turn to the tiny fruit fly to address some of these questions: This insect’s body is not as complex as ours in many ways, but we share important genes and organ functions.
Fruit flies already are a crucial and inexpensive animal for genetics research. Because their life span is about 7 weeks, investigators can track the effects of mutations across several generations in a short period. The animals also are proving useful for learning how chemicals released by malignant tumors can harm tissues in the body that are not near the cancer.
One recent lesson from the fruit flies involves the blood-brain barrier, which determines which molecules gain access to the brain. Researchers at the University of California, Berkeley, have found that malignant tumors in the tiny insects release interleukin 6 (IL-6), an inflammatory chemical that disrupts this important barrier. The investigators showed that the tumors act similarly in mice.
Even if cancer cells persisted, damage related to IL-6 could be diminished.
Fruit flies and mice are only distant relatives of each other and of humans, and the relevance of this discovery to human cancers has not been established. One hurdle is that IL-6 has many important, normal functions related to health. Researchers need to learn how to target only its unwanted blood-brain barrier effects.
A version of this article first appeared on Medscape.com.