User login
For MD-IQ use only
Medical technology should keep patient in mind
Indeed, science and technology provide opportunities to improve outcomes in ways not even imagined 100 years ago, yet we must acknowledge that technology also threatens to erect barriers between us and our patients. We can be easily tempted to confuse new care delivery tools with the actual care itself.
Threats to the physician-patient relationship
Medical history provides many examples of how our zeal to innovate can have untoward consequences to the physician-patient relationship.
In the late 1800s, for example, to convey a sense of science, purity of intent, and trust, the medical community began wearing white coats. Those white coats have been discussed as creating emotional distance between physicians and their patients.1
Even when we in the medical community are slow and reluctant to change, the external forces propelling us forward often seem unstoppable; kinetic aspirations to innovate electronic information systems and new applications seem suddenly to revolutionize care delivery when we least expect it. The rapidity of change in technology can sometimes be dizzying but can at the same time can occur so swiftly we don’t even notice it.
After René Laennec invented the stethoscope in the early 1800s, clinicians no longer needed to physically lean in and place an ear directly onto patients to hear their hearts beating. This created a distance from patients that was still lamented 50 years later, when a professor of medicine is reported to have said, “he that hath ears to hear, let him use his ears and not a stethoscope.” Still, while the stethoscope has literally distanced us from patients, it is such an important tool that we no longer think about this distancing. We have adapted over time to remain close to our patients, to sincerely listen to their thoughts and reassure them that we hear them without the need to feel our ears on their chests.
Francis Peabody, the eminent Harvard physician, wrote an essay in 1927 titled, “The Care of the Patient.” At the end of the first paragraph, he states: “The most common criticism made at present by older practitioners is that young graduates ... are too “scientific” and do not know how to take care of patients.” He goes on to say that “one of the essential qualities of the clinician is interest in humanity, for the secret of the care of the patient is in caring for the patient.”2
We agree with Dr. Peabody. As we embrace science and technology that can change health outcomes, our patients’ needs to feel understood and cared for will not diminish. Instead, that need will continue to be an important aspect of our struggle and joy in providing holistic, humane, competent care into the future.
Twenty-first century physicians have access to an ever-growing trove of data, yet our ability to truly know our patients seems somehow less accessible. Home health devices have begun to provide a flow of information about parameters, ranging from continuous glucose readings to home blood pressures, weights, and inspiratory flow readings. These data can provide much more accurate insight into patients than what we can glean from one point in time during an office visit. Yet we need to remember that behind the data are people with dreams and desires, not just table entries in an electronic health record.
In 1923, the German philosopher Martin Buber published the book for which he is best known, “I and Thou.” In that book, Mr. Buber says that there are two ways we can approach relationships: “I-Thou” or “I-It.” In I-It relationships, we view the other person as an “it” to be used to accomplish a purpose, or to be experienced without his or her full involvement. In an I-Thou relationship, we appreciate the other people for all their complexity, in their full humanness. We must consciously remind ourselves amid the rush of technology that there are real people behind those data. We must acknowledge and approach each person as a unique individual who has dreams, goals, fears, and wishes that may be different from ours but to which we can still relate.
‘From the Beating End of the Stethoscope’
John Ciardi, an American poet, said the following in a poem titled, “Lines From the Beating End of the Stethoscope”:
I speak, as I say, the patient’s point of view.
But, given time, doctors are patients, too.
And there’s our bond: beyond anatomy,
Or in it, through it, to the mystery
Medicine takes the pulse of and lets go
Forever unexplained. It’s art, we know,
Not science at the heart. Doctor be whole,
I won’t insist the patient is a soul,
But he’s a something, possibly laughable,
Or possibly sublime, but not quite graphable.
Not quite containable on a bed chart.
Where science touches man it turns to art.3
This poem is a reminder of the subtle needs of patients during their encounters with doctors, especially around many of the most important decisions and events in their lives. Patients’ needs are varied, complex, difficult to discern, and not able to be fully explained or understood through math and science.
Einstein warned us that the modern age would be characterized by a perfection of means and a confusion of goals.4 As clinicians, we should strive to clarify and align our goals with those of our patients, providing care that is real, compassionate, and personal, not just an optimized means to achieve standardized metrics. While technology can assist us in this pursuit, we’ll need be careful that our enchantment with innovation does not cloud our actual goal: truly caring for our patients.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
References
1. Jones VA. The white coat: Why not follow suit? JAMA. 1999;281(5):478. doi: 10.1001/jama.281.5.478-JMS0203-5-1
2. Peabody, Francis (1927). “The care of the patient.” JAMA. 88(12):877-82. doi: 10.1001/jama.1927.02680380001001.
3. Ciardi, John. Lines from the Beating End of the Stethoscope. Saturday Review, Nov. 18, 1968.
4. Albert Einstein, Out of My Later Years, 1950.
Indeed, science and technology provide opportunities to improve outcomes in ways not even imagined 100 years ago, yet we must acknowledge that technology also threatens to erect barriers between us and our patients. We can be easily tempted to confuse new care delivery tools with the actual care itself.
Threats to the physician-patient relationship
Medical history provides many examples of how our zeal to innovate can have untoward consequences to the physician-patient relationship.
In the late 1800s, for example, to convey a sense of science, purity of intent, and trust, the medical community began wearing white coats. Those white coats have been discussed as creating emotional distance between physicians and their patients.1
Even when we in the medical community are slow and reluctant to change, the external forces propelling us forward often seem unstoppable; kinetic aspirations to innovate electronic information systems and new applications seem suddenly to revolutionize care delivery when we least expect it. The rapidity of change in technology can sometimes be dizzying but can at the same time can occur so swiftly we don’t even notice it.
After René Laennec invented the stethoscope in the early 1800s, clinicians no longer needed to physically lean in and place an ear directly onto patients to hear their hearts beating. This created a distance from patients that was still lamented 50 years later, when a professor of medicine is reported to have said, “he that hath ears to hear, let him use his ears and not a stethoscope.” Still, while the stethoscope has literally distanced us from patients, it is such an important tool that we no longer think about this distancing. We have adapted over time to remain close to our patients, to sincerely listen to their thoughts and reassure them that we hear them without the need to feel our ears on their chests.
Francis Peabody, the eminent Harvard physician, wrote an essay in 1927 titled, “The Care of the Patient.” At the end of the first paragraph, he states: “The most common criticism made at present by older practitioners is that young graduates ... are too “scientific” and do not know how to take care of patients.” He goes on to say that “one of the essential qualities of the clinician is interest in humanity, for the secret of the care of the patient is in caring for the patient.”2
We agree with Dr. Peabody. As we embrace science and technology that can change health outcomes, our patients’ needs to feel understood and cared for will not diminish. Instead, that need will continue to be an important aspect of our struggle and joy in providing holistic, humane, competent care into the future.
Twenty-first century physicians have access to an ever-growing trove of data, yet our ability to truly know our patients seems somehow less accessible. Home health devices have begun to provide a flow of information about parameters, ranging from continuous glucose readings to home blood pressures, weights, and inspiratory flow readings. These data can provide much more accurate insight into patients than what we can glean from one point in time during an office visit. Yet we need to remember that behind the data are people with dreams and desires, not just table entries in an electronic health record.
In 1923, the German philosopher Martin Buber published the book for which he is best known, “I and Thou.” In that book, Mr. Buber says that there are two ways we can approach relationships: “I-Thou” or “I-It.” In I-It relationships, we view the other person as an “it” to be used to accomplish a purpose, or to be experienced without his or her full involvement. In an I-Thou relationship, we appreciate the other people for all their complexity, in their full humanness. We must consciously remind ourselves amid the rush of technology that there are real people behind those data. We must acknowledge and approach each person as a unique individual who has dreams, goals, fears, and wishes that may be different from ours but to which we can still relate.
‘From the Beating End of the Stethoscope’
John Ciardi, an American poet, said the following in a poem titled, “Lines From the Beating End of the Stethoscope”:
I speak, as I say, the patient’s point of view.
But, given time, doctors are patients, too.
And there’s our bond: beyond anatomy,
Or in it, through it, to the mystery
Medicine takes the pulse of and lets go
Forever unexplained. It’s art, we know,
Not science at the heart. Doctor be whole,
I won’t insist the patient is a soul,
But he’s a something, possibly laughable,
Or possibly sublime, but not quite graphable.
Not quite containable on a bed chart.
Where science touches man it turns to art.3
This poem is a reminder of the subtle needs of patients during their encounters with doctors, especially around many of the most important decisions and events in their lives. Patients’ needs are varied, complex, difficult to discern, and not able to be fully explained or understood through math and science.
Einstein warned us that the modern age would be characterized by a perfection of means and a confusion of goals.4 As clinicians, we should strive to clarify and align our goals with those of our patients, providing care that is real, compassionate, and personal, not just an optimized means to achieve standardized metrics. While technology can assist us in this pursuit, we’ll need be careful that our enchantment with innovation does not cloud our actual goal: truly caring for our patients.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
References
1. Jones VA. The white coat: Why not follow suit? JAMA. 1999;281(5):478. doi: 10.1001/jama.281.5.478-JMS0203-5-1
2. Peabody, Francis (1927). “The care of the patient.” JAMA. 88(12):877-82. doi: 10.1001/jama.1927.02680380001001.
3. Ciardi, John. Lines from the Beating End of the Stethoscope. Saturday Review, Nov. 18, 1968.
4. Albert Einstein, Out of My Later Years, 1950.
Indeed, science and technology provide opportunities to improve outcomes in ways not even imagined 100 years ago, yet we must acknowledge that technology also threatens to erect barriers between us and our patients. We can be easily tempted to confuse new care delivery tools with the actual care itself.
Threats to the physician-patient relationship
Medical history provides many examples of how our zeal to innovate can have untoward consequences to the physician-patient relationship.
In the late 1800s, for example, to convey a sense of science, purity of intent, and trust, the medical community began wearing white coats. Those white coats have been discussed as creating emotional distance between physicians and their patients.1
Even when we in the medical community are slow and reluctant to change, the external forces propelling us forward often seem unstoppable; kinetic aspirations to innovate electronic information systems and new applications seem suddenly to revolutionize care delivery when we least expect it. The rapidity of change in technology can sometimes be dizzying but can at the same time can occur so swiftly we don’t even notice it.
After René Laennec invented the stethoscope in the early 1800s, clinicians no longer needed to physically lean in and place an ear directly onto patients to hear their hearts beating. This created a distance from patients that was still lamented 50 years later, when a professor of medicine is reported to have said, “he that hath ears to hear, let him use his ears and not a stethoscope.” Still, while the stethoscope has literally distanced us from patients, it is such an important tool that we no longer think about this distancing. We have adapted over time to remain close to our patients, to sincerely listen to their thoughts and reassure them that we hear them without the need to feel our ears on their chests.
Francis Peabody, the eminent Harvard physician, wrote an essay in 1927 titled, “The Care of the Patient.” At the end of the first paragraph, he states: “The most common criticism made at present by older practitioners is that young graduates ... are too “scientific” and do not know how to take care of patients.” He goes on to say that “one of the essential qualities of the clinician is interest in humanity, for the secret of the care of the patient is in caring for the patient.”2
We agree with Dr. Peabody. As we embrace science and technology that can change health outcomes, our patients’ needs to feel understood and cared for will not diminish. Instead, that need will continue to be an important aspect of our struggle and joy in providing holistic, humane, competent care into the future.
Twenty-first century physicians have access to an ever-growing trove of data, yet our ability to truly know our patients seems somehow less accessible. Home health devices have begun to provide a flow of information about parameters, ranging from continuous glucose readings to home blood pressures, weights, and inspiratory flow readings. These data can provide much more accurate insight into patients than what we can glean from one point in time during an office visit. Yet we need to remember that behind the data are people with dreams and desires, not just table entries in an electronic health record.
In 1923, the German philosopher Martin Buber published the book for which he is best known, “I and Thou.” In that book, Mr. Buber says that there are two ways we can approach relationships: “I-Thou” or “I-It.” In I-It relationships, we view the other person as an “it” to be used to accomplish a purpose, or to be experienced without his or her full involvement. In an I-Thou relationship, we appreciate the other people for all their complexity, in their full humanness. We must consciously remind ourselves amid the rush of technology that there are real people behind those data. We must acknowledge and approach each person as a unique individual who has dreams, goals, fears, and wishes that may be different from ours but to which we can still relate.
‘From the Beating End of the Stethoscope’
John Ciardi, an American poet, said the following in a poem titled, “Lines From the Beating End of the Stethoscope”:
I speak, as I say, the patient’s point of view.
But, given time, doctors are patients, too.
And there’s our bond: beyond anatomy,
Or in it, through it, to the mystery
Medicine takes the pulse of and lets go
Forever unexplained. It’s art, we know,
Not science at the heart. Doctor be whole,
I won’t insist the patient is a soul,
But he’s a something, possibly laughable,
Or possibly sublime, but not quite graphable.
Not quite containable on a bed chart.
Where science touches man it turns to art.3
This poem is a reminder of the subtle needs of patients during their encounters with doctors, especially around many of the most important decisions and events in their lives. Patients’ needs are varied, complex, difficult to discern, and not able to be fully explained or understood through math and science.
Einstein warned us that the modern age would be characterized by a perfection of means and a confusion of goals.4 As clinicians, we should strive to clarify and align our goals with those of our patients, providing care that is real, compassionate, and personal, not just an optimized means to achieve standardized metrics. While technology can assist us in this pursuit, we’ll need be careful that our enchantment with innovation does not cloud our actual goal: truly caring for our patients.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
References
1. Jones VA. The white coat: Why not follow suit? JAMA. 1999;281(5):478. doi: 10.1001/jama.281.5.478-JMS0203-5-1
2. Peabody, Francis (1927). “The care of the patient.” JAMA. 88(12):877-82. doi: 10.1001/jama.1927.02680380001001.
3. Ciardi, John. Lines from the Beating End of the Stethoscope. Saturday Review, Nov. 18, 1968.
4. Albert Einstein, Out of My Later Years, 1950.
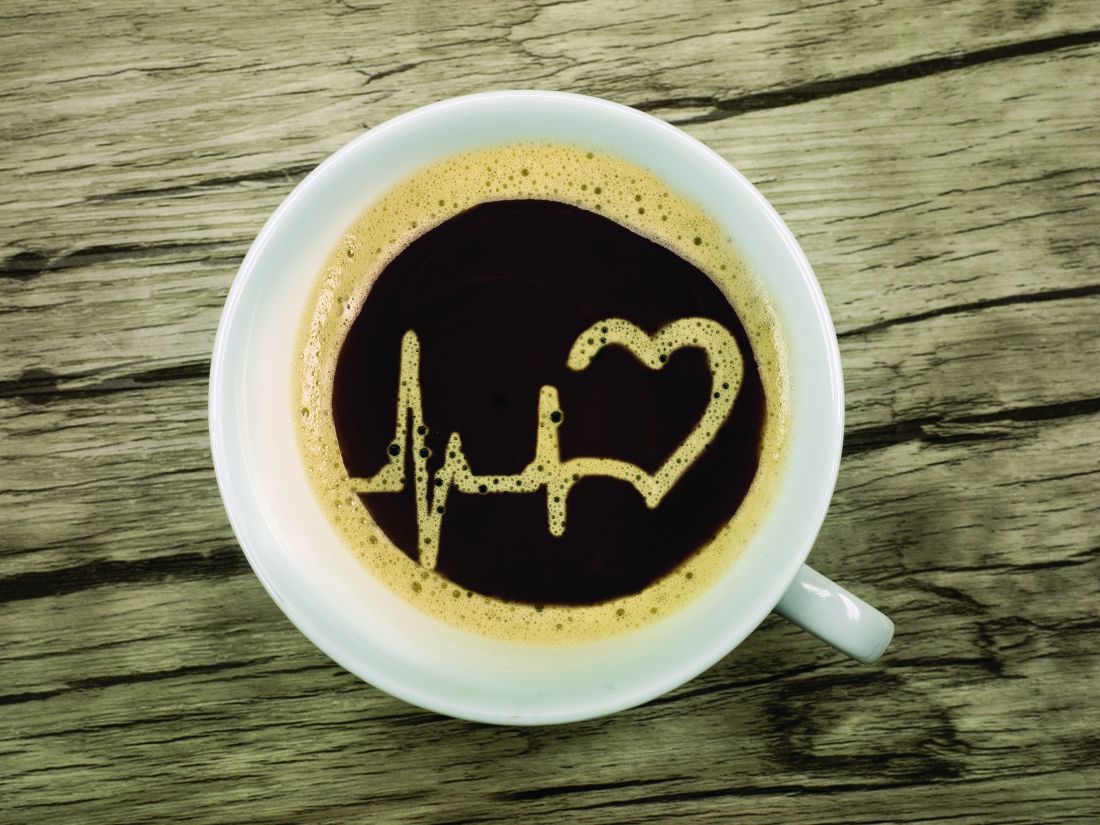
Coffee or tea? Drinking both tied to lower stroke, dementia risk
Drinking coffee or tea is associated with reduced risk for stroke and dementia, with the biggest benefit associated with consuming both beverages, new research suggests.
Investigators found that individuals who drank two to three cups of coffee and two to three cups of tea per day had a 30% decrease in incidence of stroke and a 28% lower risk for dementia compared with those who did not.
“From a public health perspective, because regular tea and coffee drinkers comprise such a large proportion of the population and because these beverages tend to be consumed habitually throughout adult life, even small potential health benefits or risks associated with tea and coffee intake may have important public health implications,” the investigators wrote.
The study was published online Nov. 16 in PLOS Medicine.
Synergistic effect?
Whereas earlier studies have shown significant health benefits from moderate coffee and tea intake separately, few have examined the effect of drinking both.
Researchers enrolled 365,682 participants from the UK Biobank for the analysis of coffee and tea consumption and stroke and dementia risk and 13,352 participants for the analysis of poststroke dementia.
During a median follow-up of 11.4 years, 2.8% of participants experienced a stroke and 1.4% developed dementia.
After adjustment for confounders, stroke risk was 10% lower in those who drank a half-cup to a cup of coffee per day (P < .001) and 8% lower in those who had more than two cups a day (P = .009). Tea drinkers who had more than two cups a day saw a 16% reduction in stroke (P < .001).
Those who drank both coffee and tea during the day saw the greatest benefit. Drinking two to three cups of coffee and two to three cups of tea lowered stroke risk by 32% (P < .001) and dementia risk by 28% (P = .002).
Drinking both beverages offered significantly greater benefits than drinking just coffee or tea alone, with an 11% lower risk for stroke (P < .001), an 8% lower risk for dementia (P = .001), and 18% lower risk for vascular dementia (P = .001).
Among those participants who experienced a stroke during the follow-up period, drinking two to three cups of coffee was associated with 20% lower risk for poststroke dementia (P = .044), and for those who drank both coffee and tea (half to one cup of coffee and two to three cups of tea per day) the risk for poststroke dementia was lowered by 50% (P =.006).
There was no significant association between coffee and tea consumption and risk for hemorrhagic stroke or Alzheimer’s disease.
The study was funded by the National Natural Science Foundation of China. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Drinking coffee or tea is associated with reduced risk for stroke and dementia, with the biggest benefit associated with consuming both beverages, new research suggests.
Investigators found that individuals who drank two to three cups of coffee and two to three cups of tea per day had a 30% decrease in incidence of stroke and a 28% lower risk for dementia compared with those who did not.
“From a public health perspective, because regular tea and coffee drinkers comprise such a large proportion of the population and because these beverages tend to be consumed habitually throughout adult life, even small potential health benefits or risks associated with tea and coffee intake may have important public health implications,” the investigators wrote.
The study was published online Nov. 16 in PLOS Medicine.
Synergistic effect?
Whereas earlier studies have shown significant health benefits from moderate coffee and tea intake separately, few have examined the effect of drinking both.
Researchers enrolled 365,682 participants from the UK Biobank for the analysis of coffee and tea consumption and stroke and dementia risk and 13,352 participants for the analysis of poststroke dementia.
During a median follow-up of 11.4 years, 2.8% of participants experienced a stroke and 1.4% developed dementia.
After adjustment for confounders, stroke risk was 10% lower in those who drank a half-cup to a cup of coffee per day (P < .001) and 8% lower in those who had more than two cups a day (P = .009). Tea drinkers who had more than two cups a day saw a 16% reduction in stroke (P < .001).
Those who drank both coffee and tea during the day saw the greatest benefit. Drinking two to three cups of coffee and two to three cups of tea lowered stroke risk by 32% (P < .001) and dementia risk by 28% (P = .002).
Drinking both beverages offered significantly greater benefits than drinking just coffee or tea alone, with an 11% lower risk for stroke (P < .001), an 8% lower risk for dementia (P = .001), and 18% lower risk for vascular dementia (P = .001).
Among those participants who experienced a stroke during the follow-up period, drinking two to three cups of coffee was associated with 20% lower risk for poststroke dementia (P = .044), and for those who drank both coffee and tea (half to one cup of coffee and two to three cups of tea per day) the risk for poststroke dementia was lowered by 50% (P =.006).
There was no significant association between coffee and tea consumption and risk for hemorrhagic stroke or Alzheimer’s disease.
The study was funded by the National Natural Science Foundation of China. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Drinking coffee or tea is associated with reduced risk for stroke and dementia, with the biggest benefit associated with consuming both beverages, new research suggests.
Investigators found that individuals who drank two to three cups of coffee and two to three cups of tea per day had a 30% decrease in incidence of stroke and a 28% lower risk for dementia compared with those who did not.
“From a public health perspective, because regular tea and coffee drinkers comprise such a large proportion of the population and because these beverages tend to be consumed habitually throughout adult life, even small potential health benefits or risks associated with tea and coffee intake may have important public health implications,” the investigators wrote.
The study was published online Nov. 16 in PLOS Medicine.
Synergistic effect?
Whereas earlier studies have shown significant health benefits from moderate coffee and tea intake separately, few have examined the effect of drinking both.
Researchers enrolled 365,682 participants from the UK Biobank for the analysis of coffee and tea consumption and stroke and dementia risk and 13,352 participants for the analysis of poststroke dementia.
During a median follow-up of 11.4 years, 2.8% of participants experienced a stroke and 1.4% developed dementia.
After adjustment for confounders, stroke risk was 10% lower in those who drank a half-cup to a cup of coffee per day (P < .001) and 8% lower in those who had more than two cups a day (P = .009). Tea drinkers who had more than two cups a day saw a 16% reduction in stroke (P < .001).
Those who drank both coffee and tea during the day saw the greatest benefit. Drinking two to three cups of coffee and two to three cups of tea lowered stroke risk by 32% (P < .001) and dementia risk by 28% (P = .002).
Drinking both beverages offered significantly greater benefits than drinking just coffee or tea alone, with an 11% lower risk for stroke (P < .001), an 8% lower risk for dementia (P = .001), and 18% lower risk for vascular dementia (P = .001).
Among those participants who experienced a stroke during the follow-up period, drinking two to three cups of coffee was associated with 20% lower risk for poststroke dementia (P = .044), and for those who drank both coffee and tea (half to one cup of coffee and two to three cups of tea per day) the risk for poststroke dementia was lowered by 50% (P =.006).
There was no significant association between coffee and tea consumption and risk for hemorrhagic stroke or Alzheimer’s disease.
The study was funded by the National Natural Science Foundation of China. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CRAVE: Drinking coffee not linked to atrial arrhythmias
A novel trial using real-time monitoring found that drinking coffee did not increase atrial arrhythmias but was associated with more premature ventricular contractions.
There was no increase in premature atrial contractions (PACs) or supraventricular tachycardia (SVT) with coffee consumption, and, in fact, there was less SVT in per protocol analyses.
Coffee consumption was also linked to a “clinically meaningful increase in physical activity as well as a clinically meaningful reduction in sleep,” coprincipal investigator Gregory M. Marcus, MD, of the University of California, San Francisco, reported at the American Heart Association scientific sessions.
Although some professional society guidelines warn against caffeine consumption to avoid arrhythmias, he noted that the data have been mixed and that growing evidence suggests coffee consumption may actually lower the risk for arrhythmias, diabetes, and even mortality. The exact relationship has been hard to prove, however, as most coffee studies are observational and rely on self-report.
The Coffee and Real-time Atrial and Ventricular Ectopy (CRAVE) trial took advantage of digital health tools to examine the effect of caffeine consumption on cardiac ectopy burden in 100 healthy volunteers using an N-of-1 design. The primary outcomes were daily PAC and premature ventricular contraction (PVC) counts.
Participants consumed as much coffee as they wanted for 1 day and avoided all caffeine the next, alternating the assignment in 2-day blocks over 2 weeks. They used a smartphone app to receive daily coffee assignments and reminders and wore a continuous recording electrocardiography monitor (ZioPatch, iRhythm Technologies); a continuous glucose monitor (Dexcom); and Fitbit Flex 2, which recorded step counts and sleep duration.
At baseline, 21% of participants drank six to seven cups of coffee per month, 29% drank one cup per day, 21% drank two to three cups per day, and 3% drank four to five cups per day. The U.S. Food and Drug Administration has cited 400 mg per day, or about four or five cups of coffee, as generally safe for healthy adults.
To assess adherence, participants were asked to press the button on the ZioPatch for every coffee drink and were queried daily regarding actual coffee consumption the previous day. Date-stamped receipts for coffee purchases were reimbursed, and smartphone geolocation was used to track coffee shop visits. The great majority of times, participants followed their assignment by all measures, Dr. Marcus said.
ITT and per protocol analyses
ZioPatch data collected over a median of 13.3 days showed a daily median of 12.8 PACs, 7.5 PVCs, 1 nonsustained SVT, and 1 nonsustained ventricular tachycardia.
In intention-to-treat (ITT) analyses, there was no evidence of a relationship between coffee consumption and daily PAC counts (RR, 1.09; 95% confidence interval, 0.98-1.20; P = .10).
In contrast, participants had an average of 54% more PVCs on days randomized to coffee by ITT (RR, 1.54; 95% CI, 1.19-2.00; P = .001), and, per protocol, those consuming more than two cups of coffee per day had a doubling of PVCs (RR, 2.20; 95% CI, 1.24-3.92; P = .007).
No relationship was observed with coffee consumption and SVT episodes in ITT analyses (RR, 0.84; 95% CI, 0.69-1.03; P = .10), but, per protocol, every additional coffee drink consumed in real time was associated with a 12% lower risk for an SVT episode (RR, 0.88; 95% CI, 0.79-0.99; P = .028).
No significant relationships were observed with VT episodes, which were admittedly rare, Dr. Marcus said.
In ITT analyses that adjusted for day of the week, participants took an average of 1,058 more steps on days they drank coffee (95% CI, 441-1,675 steps; P = .001) but slept 36 fewer minutes (95% CI, 22-50 minutes; P < .001).
Per protocol, every additional coffee drink was associated with 587 more steps per day (95% CI, 355-820 steps; P < .001) and 18 fewer minutes of sleep (95% CI, 13-23 minutes; P < .001).
No significant differences in glucose levels were observed. Genetic analyses revealed two significant interactions: fast coffee metabolizers had a heightened risk for PVCs and slow metabolizers experienced more sleep deprivation, Dr. Marcus said.
Typical patients?
Dedicated discussant Sana Al-Khatib, MD, MHS, Duke University Medical Center, Durham, N.C., said CRAVE is a “well-conducted and informative trial” that very nicely and effectively used a digital health platform.
She pointed out, however, that the trial enrolled healthy volunteers who not only owned a smartphone but were able to interact with the study team using it. They also had an average age of 38 years, median body mass index of 24 kg/m2, and no prior arrhythmias or cardiovascular issues. “These are not representative of the average patient that we see in clinical practice.”
“The other thing to keep in mind is that the primary outcome that they looked at, while relevant, is not adequate in my view to help us derive definitive conclusions about how coffee consumption affects clinically meaningful arrhythmias,” Dr. Al-Khatib said. “Yes, PACs trigger atrial fibrillation, but they don’t do so in every patient. And PVCs have been shown to be associated with increased mortality as well as worsened cardiovascular outcomes, but that’s mostly in patients with structural heart disease.”
She praised the investigators for including genetic data in their analysis. “Whether the results related to physical activity and sleep translate into any major effect on clinical outcomes deserves a study.”
The overall findings need to be replicated by other groups, in other populations, and examine hard outcomes over longer follow-up, concluded Dr. Al-Khatib.
Speaking to this news organization, Dr. Marcus countered that the participants were “pretty run of the mill” coffee drinkers of all ages and that the study highlights the complexity of coffee consumption as well as providing unique data inferring causality regarding increasing physical activity.
“Because coffee is so commonly consumed, highlighting the actual effects is important, and the hope is that understanding those true causal effects and minimizing confounding will help tailor recommendations regarding coffee consumption,” he said. “For those concerned about atrial fibrillation, for example, these data suggest that avoiding coffee does not necessarily make sense to reduce the risk of atrial fibrillation. For those with ventricular arrhythmias, abstinence or minimizing coffee may be a worthwhile experiment.”
Kalyanam Shivkumar, MD, PhD, director of the cardiac arrhythmia center at the University of California, Los Angeles, told this news organization that CRAVE is an important and much-needed study that provides reassuring and objective data for a common clinical question.
“It fits in with the emerging consensus that, in itself, coffee is not problematic,” he said. “And it provides a nice framework for what we’ll be seeing in the future – more studies that use these types of long ECG recordings and interlinking that data with biological readouts.”
Although it is too early to draw any conclusions regarding the genetic analyses, “future studies could use this as a baseline to further explore what happens between fast and slow metabolizers. This is a very useful stepping stone to putting data in context for an individual patient.”
Unless coffee consumption is excessive, such as over five cups per day in young people, all of the evidence points to coffee and caffeine being safe, Chip Lavie, MD, a frequent coffee researcher and medical director of cardiac rehabilitation and prevention at John Ochsner Heart and Vascular Institute, New Orleans, told this news organization.
“The benefits of coffee on physical activity/sleep seem to outweigh the risks as this current study suggests,” he said. “This study also supports the safety with regards to atrial arrhythmias, and suggests that those with symptomatic PVCs could try reducing coffee to see if they feel better. In total, however, the benefits of one or several cups of coffee per day on cardiovascular disease outweigh the risks.”
The study was funded by the University of California, San Francisco. Dr. Marcus reports research with the National Institutes of Health, the Patient-Centered Outcomes Research Institute, Tobacco-Related Disease Research Program, Medtronic, Eight Sleep, and Baylis; consulting for InCarda Therapeutics and Johnson & Johnson; and equity in InCarda Therapeutics as cofounder.
A version of this article first appeared on Medscape.com.
A novel trial using real-time monitoring found that drinking coffee did not increase atrial arrhythmias but was associated with more premature ventricular contractions.
There was no increase in premature atrial contractions (PACs) or supraventricular tachycardia (SVT) with coffee consumption, and, in fact, there was less SVT in per protocol analyses.
Coffee consumption was also linked to a “clinically meaningful increase in physical activity as well as a clinically meaningful reduction in sleep,” coprincipal investigator Gregory M. Marcus, MD, of the University of California, San Francisco, reported at the American Heart Association scientific sessions.
Although some professional society guidelines warn against caffeine consumption to avoid arrhythmias, he noted that the data have been mixed and that growing evidence suggests coffee consumption may actually lower the risk for arrhythmias, diabetes, and even mortality. The exact relationship has been hard to prove, however, as most coffee studies are observational and rely on self-report.
The Coffee and Real-time Atrial and Ventricular Ectopy (CRAVE) trial took advantage of digital health tools to examine the effect of caffeine consumption on cardiac ectopy burden in 100 healthy volunteers using an N-of-1 design. The primary outcomes were daily PAC and premature ventricular contraction (PVC) counts.
Participants consumed as much coffee as they wanted for 1 day and avoided all caffeine the next, alternating the assignment in 2-day blocks over 2 weeks. They used a smartphone app to receive daily coffee assignments and reminders and wore a continuous recording electrocardiography monitor (ZioPatch, iRhythm Technologies); a continuous glucose monitor (Dexcom); and Fitbit Flex 2, which recorded step counts and sleep duration.
At baseline, 21% of participants drank six to seven cups of coffee per month, 29% drank one cup per day, 21% drank two to three cups per day, and 3% drank four to five cups per day. The U.S. Food and Drug Administration has cited 400 mg per day, or about four or five cups of coffee, as generally safe for healthy adults.
To assess adherence, participants were asked to press the button on the ZioPatch for every coffee drink and were queried daily regarding actual coffee consumption the previous day. Date-stamped receipts for coffee purchases were reimbursed, and smartphone geolocation was used to track coffee shop visits. The great majority of times, participants followed their assignment by all measures, Dr. Marcus said.
ITT and per protocol analyses
ZioPatch data collected over a median of 13.3 days showed a daily median of 12.8 PACs, 7.5 PVCs, 1 nonsustained SVT, and 1 nonsustained ventricular tachycardia.
In intention-to-treat (ITT) analyses, there was no evidence of a relationship between coffee consumption and daily PAC counts (RR, 1.09; 95% confidence interval, 0.98-1.20; P = .10).
In contrast, participants had an average of 54% more PVCs on days randomized to coffee by ITT (RR, 1.54; 95% CI, 1.19-2.00; P = .001), and, per protocol, those consuming more than two cups of coffee per day had a doubling of PVCs (RR, 2.20; 95% CI, 1.24-3.92; P = .007).
No relationship was observed with coffee consumption and SVT episodes in ITT analyses (RR, 0.84; 95% CI, 0.69-1.03; P = .10), but, per protocol, every additional coffee drink consumed in real time was associated with a 12% lower risk for an SVT episode (RR, 0.88; 95% CI, 0.79-0.99; P = .028).
No significant relationships were observed with VT episodes, which were admittedly rare, Dr. Marcus said.
In ITT analyses that adjusted for day of the week, participants took an average of 1,058 more steps on days they drank coffee (95% CI, 441-1,675 steps; P = .001) but slept 36 fewer minutes (95% CI, 22-50 minutes; P < .001).
Per protocol, every additional coffee drink was associated with 587 more steps per day (95% CI, 355-820 steps; P < .001) and 18 fewer minutes of sleep (95% CI, 13-23 minutes; P < .001).
No significant differences in glucose levels were observed. Genetic analyses revealed two significant interactions: fast coffee metabolizers had a heightened risk for PVCs and slow metabolizers experienced more sleep deprivation, Dr. Marcus said.
Typical patients?
Dedicated discussant Sana Al-Khatib, MD, MHS, Duke University Medical Center, Durham, N.C., said CRAVE is a “well-conducted and informative trial” that very nicely and effectively used a digital health platform.
She pointed out, however, that the trial enrolled healthy volunteers who not only owned a smartphone but were able to interact with the study team using it. They also had an average age of 38 years, median body mass index of 24 kg/m2, and no prior arrhythmias or cardiovascular issues. “These are not representative of the average patient that we see in clinical practice.”
“The other thing to keep in mind is that the primary outcome that they looked at, while relevant, is not adequate in my view to help us derive definitive conclusions about how coffee consumption affects clinically meaningful arrhythmias,” Dr. Al-Khatib said. “Yes, PACs trigger atrial fibrillation, but they don’t do so in every patient. And PVCs have been shown to be associated with increased mortality as well as worsened cardiovascular outcomes, but that’s mostly in patients with structural heart disease.”
She praised the investigators for including genetic data in their analysis. “Whether the results related to physical activity and sleep translate into any major effect on clinical outcomes deserves a study.”
The overall findings need to be replicated by other groups, in other populations, and examine hard outcomes over longer follow-up, concluded Dr. Al-Khatib.
Speaking to this news organization, Dr. Marcus countered that the participants were “pretty run of the mill” coffee drinkers of all ages and that the study highlights the complexity of coffee consumption as well as providing unique data inferring causality regarding increasing physical activity.
“Because coffee is so commonly consumed, highlighting the actual effects is important, and the hope is that understanding those true causal effects and minimizing confounding will help tailor recommendations regarding coffee consumption,” he said. “For those concerned about atrial fibrillation, for example, these data suggest that avoiding coffee does not necessarily make sense to reduce the risk of atrial fibrillation. For those with ventricular arrhythmias, abstinence or minimizing coffee may be a worthwhile experiment.”
Kalyanam Shivkumar, MD, PhD, director of the cardiac arrhythmia center at the University of California, Los Angeles, told this news organization that CRAVE is an important and much-needed study that provides reassuring and objective data for a common clinical question.
“It fits in with the emerging consensus that, in itself, coffee is not problematic,” he said. “And it provides a nice framework for what we’ll be seeing in the future – more studies that use these types of long ECG recordings and interlinking that data with biological readouts.”
Although it is too early to draw any conclusions regarding the genetic analyses, “future studies could use this as a baseline to further explore what happens between fast and slow metabolizers. This is a very useful stepping stone to putting data in context for an individual patient.”
Unless coffee consumption is excessive, such as over five cups per day in young people, all of the evidence points to coffee and caffeine being safe, Chip Lavie, MD, a frequent coffee researcher and medical director of cardiac rehabilitation and prevention at John Ochsner Heart and Vascular Institute, New Orleans, told this news organization.
“The benefits of coffee on physical activity/sleep seem to outweigh the risks as this current study suggests,” he said. “This study also supports the safety with regards to atrial arrhythmias, and suggests that those with symptomatic PVCs could try reducing coffee to see if they feel better. In total, however, the benefits of one or several cups of coffee per day on cardiovascular disease outweigh the risks.”
The study was funded by the University of California, San Francisco. Dr. Marcus reports research with the National Institutes of Health, the Patient-Centered Outcomes Research Institute, Tobacco-Related Disease Research Program, Medtronic, Eight Sleep, and Baylis; consulting for InCarda Therapeutics and Johnson & Johnson; and equity in InCarda Therapeutics as cofounder.
A version of this article first appeared on Medscape.com.
A novel trial using real-time monitoring found that drinking coffee did not increase atrial arrhythmias but was associated with more premature ventricular contractions.
There was no increase in premature atrial contractions (PACs) or supraventricular tachycardia (SVT) with coffee consumption, and, in fact, there was less SVT in per protocol analyses.
Coffee consumption was also linked to a “clinically meaningful increase in physical activity as well as a clinically meaningful reduction in sleep,” coprincipal investigator Gregory M. Marcus, MD, of the University of California, San Francisco, reported at the American Heart Association scientific sessions.
Although some professional society guidelines warn against caffeine consumption to avoid arrhythmias, he noted that the data have been mixed and that growing evidence suggests coffee consumption may actually lower the risk for arrhythmias, diabetes, and even mortality. The exact relationship has been hard to prove, however, as most coffee studies are observational and rely on self-report.
The Coffee and Real-time Atrial and Ventricular Ectopy (CRAVE) trial took advantage of digital health tools to examine the effect of caffeine consumption on cardiac ectopy burden in 100 healthy volunteers using an N-of-1 design. The primary outcomes were daily PAC and premature ventricular contraction (PVC) counts.
Participants consumed as much coffee as they wanted for 1 day and avoided all caffeine the next, alternating the assignment in 2-day blocks over 2 weeks. They used a smartphone app to receive daily coffee assignments and reminders and wore a continuous recording electrocardiography monitor (ZioPatch, iRhythm Technologies); a continuous glucose monitor (Dexcom); and Fitbit Flex 2, which recorded step counts and sleep duration.
At baseline, 21% of participants drank six to seven cups of coffee per month, 29% drank one cup per day, 21% drank two to three cups per day, and 3% drank four to five cups per day. The U.S. Food and Drug Administration has cited 400 mg per day, or about four or five cups of coffee, as generally safe for healthy adults.
To assess adherence, participants were asked to press the button on the ZioPatch for every coffee drink and were queried daily regarding actual coffee consumption the previous day. Date-stamped receipts for coffee purchases were reimbursed, and smartphone geolocation was used to track coffee shop visits. The great majority of times, participants followed their assignment by all measures, Dr. Marcus said.
ITT and per protocol analyses
ZioPatch data collected over a median of 13.3 days showed a daily median of 12.8 PACs, 7.5 PVCs, 1 nonsustained SVT, and 1 nonsustained ventricular tachycardia.
In intention-to-treat (ITT) analyses, there was no evidence of a relationship between coffee consumption and daily PAC counts (RR, 1.09; 95% confidence interval, 0.98-1.20; P = .10).
In contrast, participants had an average of 54% more PVCs on days randomized to coffee by ITT (RR, 1.54; 95% CI, 1.19-2.00; P = .001), and, per protocol, those consuming more than two cups of coffee per day had a doubling of PVCs (RR, 2.20; 95% CI, 1.24-3.92; P = .007).
No relationship was observed with coffee consumption and SVT episodes in ITT analyses (RR, 0.84; 95% CI, 0.69-1.03; P = .10), but, per protocol, every additional coffee drink consumed in real time was associated with a 12% lower risk for an SVT episode (RR, 0.88; 95% CI, 0.79-0.99; P = .028).
No significant relationships were observed with VT episodes, which were admittedly rare, Dr. Marcus said.
In ITT analyses that adjusted for day of the week, participants took an average of 1,058 more steps on days they drank coffee (95% CI, 441-1,675 steps; P = .001) but slept 36 fewer minutes (95% CI, 22-50 minutes; P < .001).
Per protocol, every additional coffee drink was associated with 587 more steps per day (95% CI, 355-820 steps; P < .001) and 18 fewer minutes of sleep (95% CI, 13-23 minutes; P < .001).
No significant differences in glucose levels were observed. Genetic analyses revealed two significant interactions: fast coffee metabolizers had a heightened risk for PVCs and slow metabolizers experienced more sleep deprivation, Dr. Marcus said.
Typical patients?
Dedicated discussant Sana Al-Khatib, MD, MHS, Duke University Medical Center, Durham, N.C., said CRAVE is a “well-conducted and informative trial” that very nicely and effectively used a digital health platform.
She pointed out, however, that the trial enrolled healthy volunteers who not only owned a smartphone but were able to interact with the study team using it. They also had an average age of 38 years, median body mass index of 24 kg/m2, and no prior arrhythmias or cardiovascular issues. “These are not representative of the average patient that we see in clinical practice.”
“The other thing to keep in mind is that the primary outcome that they looked at, while relevant, is not adequate in my view to help us derive definitive conclusions about how coffee consumption affects clinically meaningful arrhythmias,” Dr. Al-Khatib said. “Yes, PACs trigger atrial fibrillation, but they don’t do so in every patient. And PVCs have been shown to be associated with increased mortality as well as worsened cardiovascular outcomes, but that’s mostly in patients with structural heart disease.”
She praised the investigators for including genetic data in their analysis. “Whether the results related to physical activity and sleep translate into any major effect on clinical outcomes deserves a study.”
The overall findings need to be replicated by other groups, in other populations, and examine hard outcomes over longer follow-up, concluded Dr. Al-Khatib.
Speaking to this news organization, Dr. Marcus countered that the participants were “pretty run of the mill” coffee drinkers of all ages and that the study highlights the complexity of coffee consumption as well as providing unique data inferring causality regarding increasing physical activity.
“Because coffee is so commonly consumed, highlighting the actual effects is important, and the hope is that understanding those true causal effects and minimizing confounding will help tailor recommendations regarding coffee consumption,” he said. “For those concerned about atrial fibrillation, for example, these data suggest that avoiding coffee does not necessarily make sense to reduce the risk of atrial fibrillation. For those with ventricular arrhythmias, abstinence or minimizing coffee may be a worthwhile experiment.”
Kalyanam Shivkumar, MD, PhD, director of the cardiac arrhythmia center at the University of California, Los Angeles, told this news organization that CRAVE is an important and much-needed study that provides reassuring and objective data for a common clinical question.
“It fits in with the emerging consensus that, in itself, coffee is not problematic,” he said. “And it provides a nice framework for what we’ll be seeing in the future – more studies that use these types of long ECG recordings and interlinking that data with biological readouts.”
Although it is too early to draw any conclusions regarding the genetic analyses, “future studies could use this as a baseline to further explore what happens between fast and slow metabolizers. This is a very useful stepping stone to putting data in context for an individual patient.”
Unless coffee consumption is excessive, such as over five cups per day in young people, all of the evidence points to coffee and caffeine being safe, Chip Lavie, MD, a frequent coffee researcher and medical director of cardiac rehabilitation and prevention at John Ochsner Heart and Vascular Institute, New Orleans, told this news organization.
“The benefits of coffee on physical activity/sleep seem to outweigh the risks as this current study suggests,” he said. “This study also supports the safety with regards to atrial arrhythmias, and suggests that those with symptomatic PVCs could try reducing coffee to see if they feel better. In total, however, the benefits of one or several cups of coffee per day on cardiovascular disease outweigh the risks.”
The study was funded by the University of California, San Francisco. Dr. Marcus reports research with the National Institutes of Health, the Patient-Centered Outcomes Research Institute, Tobacco-Related Disease Research Program, Medtronic, Eight Sleep, and Baylis; consulting for InCarda Therapeutics and Johnson & Johnson; and equity in InCarda Therapeutics as cofounder.
A version of this article first appeared on Medscape.com.
FROM AHA 2021
Striae gravidarum: More than a ‘nuisance,’ say researchers
In the study of healthy pregnant women, “we found that SG can be associated with a host of negative reactions reflecting increased psychological and emotional distress,” reported Kaveri Karhade, MD, from the Berman Skin Institute, Los Altos, Calif., and coauthors from the University of Michigan, Ann Arbor. Dr. Karhade was with the department of dermatology at the University of Michigan at the time the study was conducted.
“We suggest that health care providers should avoid thinking of SG as merely a cosmetic ‘nuisance,’ ” they wrote in an article published in the International Journal of Women’s Dermatology. “Instead, it would be reasonable for providers to approach SG like other dermatologic concerns, and to consider asking patients whether SG cause emotional distress and whether prevention or treatment strategies should be attempted, even if not completely effective and potentially costly.”
The investigators did not evaluate treatments, but Frank Wang, MD, senior author of the study and professor of clinical dermatology at the University of Michigan Medicine, said in an interview that, “while they aren’t completely effective, some treatments can still help.” In addition, “recommending something also shows that you are listening to patients’ concerns – taking their concerns and skin lesions seriously,” he said.
Patient survey
The authors conducted a cross-sectional survey of 116 healthy pregnant women with SG. Participants were asked about the emotional and psychological effects of the lesions and how SG affects quality of life. The survey was modeled on questions from the Dermatology Life Quality Index, which asks about the impact of skin disease on embarrassment/self-consciousness, clothing choice, leisure activities, and interpersonal problems. “Content of questions was also devised from direct discussion with pregnant women attending clinic appointments or participating in other research studies on SG at our institution, and discussion with expert colleagues in obstetrics and dermatology,” the authors explained.
The survey consisted of 35 questions concerning demographics, pregnancy characteristics, personal and family history of SG, specific physical concerns about SG, impact of SG on attitude toward pregnancy, willingness to prevent SG or seek treatment, severity of SG (self-evaluated), the impact of SG on specific life-quality facets, and the location of lesions.
About two-thirds of respondents were aged 25-36 years and were White; the remainder self-identified as Asian, Black, Native American, or “other.” Most women reported “average” weight gain during the current pregnancy. Almost half of participants (45%) reporting a history of SG from prior pregnancies, and 65% reported a family history of SG.
The abdomen was identified most frequently as the location of SG (75%), followed by the breasts (43%), hips (43%), thighs (36%), buttocks (19%), and other areas (6%).
For most women (75%), permanency of the lesions was their top concern. About half (51%) reported that they had attempted to prevent SG, mostly with topical creams or oils. Three-quarters (75%) expressed interest in seeking treatment for SG, but this percentage dropped significantly to 33% (P =.008) if that treatment would not be covered by insurance.
Regarding the psychological impact of SG, embarrassment/self-consciousness correlated most strongly with lesion severity, followed by general quality of life, impact on choice of attire, impact on self-image/self-esteem, feelings of anxiety/depression related to SG, alteration of social/leisure activities related to SG (all P < .0001), and creation of interpersonal problems related to SG (P = .02).
The investigators also found that an increase in the effect of SG on self-image/self-esteem was “moderately associated” with younger age (P < .001) and that increased embarrassment related to SG was “moderately associated” with weight gain during pregnancy (P < .001).
“For years, stretch marks have been a topic to avoid and something many women try to hide,” Timothy Johnson, MD, professor of obstetrics and gynecology at the University of Michigan and coauthor of the study, said in a press release from the university. “Pregnant women talk about stretch marks with me every single week at clinic, and it’s time we break the stigma and start talking about them openly with all patients. ... By doing this study, we have an opportunity to normalize stretch marks in the context of all other dermatological conditions.”
Asked to comment on the findings, Tina Alster, MD, director of the Washington Institute of Dermatologic Laser Surgery and clinical professor of dermatology at Georgetown University, Washington, said her 3 decades of clinical experience support the authors’ findings. “Most patients who have striae are very self-conscious about them and report that their presence has negatively impacted their quality of life and self-confidence,” she said in an interview. “Of course, patients who come to my office are interested in having them treated, so my patient subset is skewed.”
She said treatment strategies that she discusses with patients include topical retinol/retinoids, which she said provide “low clinical response”; microneedling, which provides “marked” clinical response; and nonablative laser treatment, which provides “good” clinical response.
Considering particular patient characteristics, including budget, Dr. Alster said, “For those on a limited budget, I would propose daily use of a topical retinol, despite the low clinical effect. Many retinol-containing products are available over the counter. Prescription-strength retinoic acid tends to be pricey, often costing as much as in-office treatments.” Medical microneedling (not the cosmetic “roller” microneedling performed by aestheticians), she added, “gives the best results for the money and produces clinical results that mirror those achieved with lasers.”
Dr. Wang agreed that even recommending less expensive and less efficacious options such as over-the-counter creams can help alleviate patients’ concerns. “It shows that you are being holistic – not just caring for medical issues around pregnancy, but that you also take the emotional/psychological concerns of pregnant individuals and new parents seriously and that you recognize the impact of skin problems on quality of life. In the end, recommending something – in other words, providing some options, like creams or other therapies, for instance – is still, in my opinion, better than not recommending anything.”
Dr. Wang is involved with a study that is currently enrolling patients and that is evaluating the formation of early SG, which includes performing skin biopsies as soon as lesions appear.
The study had no funding. The study authors and Dr. Alster disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the study of healthy pregnant women, “we found that SG can be associated with a host of negative reactions reflecting increased psychological and emotional distress,” reported Kaveri Karhade, MD, from the Berman Skin Institute, Los Altos, Calif., and coauthors from the University of Michigan, Ann Arbor. Dr. Karhade was with the department of dermatology at the University of Michigan at the time the study was conducted.
“We suggest that health care providers should avoid thinking of SG as merely a cosmetic ‘nuisance,’ ” they wrote in an article published in the International Journal of Women’s Dermatology. “Instead, it would be reasonable for providers to approach SG like other dermatologic concerns, and to consider asking patients whether SG cause emotional distress and whether prevention or treatment strategies should be attempted, even if not completely effective and potentially costly.”
The investigators did not evaluate treatments, but Frank Wang, MD, senior author of the study and professor of clinical dermatology at the University of Michigan Medicine, said in an interview that, “while they aren’t completely effective, some treatments can still help.” In addition, “recommending something also shows that you are listening to patients’ concerns – taking their concerns and skin lesions seriously,” he said.
Patient survey
The authors conducted a cross-sectional survey of 116 healthy pregnant women with SG. Participants were asked about the emotional and psychological effects of the lesions and how SG affects quality of life. The survey was modeled on questions from the Dermatology Life Quality Index, which asks about the impact of skin disease on embarrassment/self-consciousness, clothing choice, leisure activities, and interpersonal problems. “Content of questions was also devised from direct discussion with pregnant women attending clinic appointments or participating in other research studies on SG at our institution, and discussion with expert colleagues in obstetrics and dermatology,” the authors explained.
The survey consisted of 35 questions concerning demographics, pregnancy characteristics, personal and family history of SG, specific physical concerns about SG, impact of SG on attitude toward pregnancy, willingness to prevent SG or seek treatment, severity of SG (self-evaluated), the impact of SG on specific life-quality facets, and the location of lesions.
About two-thirds of respondents were aged 25-36 years and were White; the remainder self-identified as Asian, Black, Native American, or “other.” Most women reported “average” weight gain during the current pregnancy. Almost half of participants (45%) reporting a history of SG from prior pregnancies, and 65% reported a family history of SG.
The abdomen was identified most frequently as the location of SG (75%), followed by the breasts (43%), hips (43%), thighs (36%), buttocks (19%), and other areas (6%).
For most women (75%), permanency of the lesions was their top concern. About half (51%) reported that they had attempted to prevent SG, mostly with topical creams or oils. Three-quarters (75%) expressed interest in seeking treatment for SG, but this percentage dropped significantly to 33% (P =.008) if that treatment would not be covered by insurance.
Regarding the psychological impact of SG, embarrassment/self-consciousness correlated most strongly with lesion severity, followed by general quality of life, impact on choice of attire, impact on self-image/self-esteem, feelings of anxiety/depression related to SG, alteration of social/leisure activities related to SG (all P < .0001), and creation of interpersonal problems related to SG (P = .02).
The investigators also found that an increase in the effect of SG on self-image/self-esteem was “moderately associated” with younger age (P < .001) and that increased embarrassment related to SG was “moderately associated” with weight gain during pregnancy (P < .001).
“For years, stretch marks have been a topic to avoid and something many women try to hide,” Timothy Johnson, MD, professor of obstetrics and gynecology at the University of Michigan and coauthor of the study, said in a press release from the university. “Pregnant women talk about stretch marks with me every single week at clinic, and it’s time we break the stigma and start talking about them openly with all patients. ... By doing this study, we have an opportunity to normalize stretch marks in the context of all other dermatological conditions.”
Asked to comment on the findings, Tina Alster, MD, director of the Washington Institute of Dermatologic Laser Surgery and clinical professor of dermatology at Georgetown University, Washington, said her 3 decades of clinical experience support the authors’ findings. “Most patients who have striae are very self-conscious about them and report that their presence has negatively impacted their quality of life and self-confidence,” she said in an interview. “Of course, patients who come to my office are interested in having them treated, so my patient subset is skewed.”
She said treatment strategies that she discusses with patients include topical retinol/retinoids, which she said provide “low clinical response”; microneedling, which provides “marked” clinical response; and nonablative laser treatment, which provides “good” clinical response.
Considering particular patient characteristics, including budget, Dr. Alster said, “For those on a limited budget, I would propose daily use of a topical retinol, despite the low clinical effect. Many retinol-containing products are available over the counter. Prescription-strength retinoic acid tends to be pricey, often costing as much as in-office treatments.” Medical microneedling (not the cosmetic “roller” microneedling performed by aestheticians), she added, “gives the best results for the money and produces clinical results that mirror those achieved with lasers.”
Dr. Wang agreed that even recommending less expensive and less efficacious options such as over-the-counter creams can help alleviate patients’ concerns. “It shows that you are being holistic – not just caring for medical issues around pregnancy, but that you also take the emotional/psychological concerns of pregnant individuals and new parents seriously and that you recognize the impact of skin problems on quality of life. In the end, recommending something – in other words, providing some options, like creams or other therapies, for instance – is still, in my opinion, better than not recommending anything.”
Dr. Wang is involved with a study that is currently enrolling patients and that is evaluating the formation of early SG, which includes performing skin biopsies as soon as lesions appear.
The study had no funding. The study authors and Dr. Alster disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the study of healthy pregnant women, “we found that SG can be associated with a host of negative reactions reflecting increased psychological and emotional distress,” reported Kaveri Karhade, MD, from the Berman Skin Institute, Los Altos, Calif., and coauthors from the University of Michigan, Ann Arbor. Dr. Karhade was with the department of dermatology at the University of Michigan at the time the study was conducted.
“We suggest that health care providers should avoid thinking of SG as merely a cosmetic ‘nuisance,’ ” they wrote in an article published in the International Journal of Women’s Dermatology. “Instead, it would be reasonable for providers to approach SG like other dermatologic concerns, and to consider asking patients whether SG cause emotional distress and whether prevention or treatment strategies should be attempted, even if not completely effective and potentially costly.”
The investigators did not evaluate treatments, but Frank Wang, MD, senior author of the study and professor of clinical dermatology at the University of Michigan Medicine, said in an interview that, “while they aren’t completely effective, some treatments can still help.” In addition, “recommending something also shows that you are listening to patients’ concerns – taking their concerns and skin lesions seriously,” he said.
Patient survey
The authors conducted a cross-sectional survey of 116 healthy pregnant women with SG. Participants were asked about the emotional and psychological effects of the lesions and how SG affects quality of life. The survey was modeled on questions from the Dermatology Life Quality Index, which asks about the impact of skin disease on embarrassment/self-consciousness, clothing choice, leisure activities, and interpersonal problems. “Content of questions was also devised from direct discussion with pregnant women attending clinic appointments or participating in other research studies on SG at our institution, and discussion with expert colleagues in obstetrics and dermatology,” the authors explained.
The survey consisted of 35 questions concerning demographics, pregnancy characteristics, personal and family history of SG, specific physical concerns about SG, impact of SG on attitude toward pregnancy, willingness to prevent SG or seek treatment, severity of SG (self-evaluated), the impact of SG on specific life-quality facets, and the location of lesions.
About two-thirds of respondents were aged 25-36 years and were White; the remainder self-identified as Asian, Black, Native American, or “other.” Most women reported “average” weight gain during the current pregnancy. Almost half of participants (45%) reporting a history of SG from prior pregnancies, and 65% reported a family history of SG.
The abdomen was identified most frequently as the location of SG (75%), followed by the breasts (43%), hips (43%), thighs (36%), buttocks (19%), and other areas (6%).
For most women (75%), permanency of the lesions was their top concern. About half (51%) reported that they had attempted to prevent SG, mostly with topical creams or oils. Three-quarters (75%) expressed interest in seeking treatment for SG, but this percentage dropped significantly to 33% (P =.008) if that treatment would not be covered by insurance.
Regarding the psychological impact of SG, embarrassment/self-consciousness correlated most strongly with lesion severity, followed by general quality of life, impact on choice of attire, impact on self-image/self-esteem, feelings of anxiety/depression related to SG, alteration of social/leisure activities related to SG (all P < .0001), and creation of interpersonal problems related to SG (P = .02).
The investigators also found that an increase in the effect of SG on self-image/self-esteem was “moderately associated” with younger age (P < .001) and that increased embarrassment related to SG was “moderately associated” with weight gain during pregnancy (P < .001).
“For years, stretch marks have been a topic to avoid and something many women try to hide,” Timothy Johnson, MD, professor of obstetrics and gynecology at the University of Michigan and coauthor of the study, said in a press release from the university. “Pregnant women talk about stretch marks with me every single week at clinic, and it’s time we break the stigma and start talking about them openly with all patients. ... By doing this study, we have an opportunity to normalize stretch marks in the context of all other dermatological conditions.”
Asked to comment on the findings, Tina Alster, MD, director of the Washington Institute of Dermatologic Laser Surgery and clinical professor of dermatology at Georgetown University, Washington, said her 3 decades of clinical experience support the authors’ findings. “Most patients who have striae are very self-conscious about them and report that their presence has negatively impacted their quality of life and self-confidence,” she said in an interview. “Of course, patients who come to my office are interested in having them treated, so my patient subset is skewed.”
She said treatment strategies that she discusses with patients include topical retinol/retinoids, which she said provide “low clinical response”; microneedling, which provides “marked” clinical response; and nonablative laser treatment, which provides “good” clinical response.
Considering particular patient characteristics, including budget, Dr. Alster said, “For those on a limited budget, I would propose daily use of a topical retinol, despite the low clinical effect. Many retinol-containing products are available over the counter. Prescription-strength retinoic acid tends to be pricey, often costing as much as in-office treatments.” Medical microneedling (not the cosmetic “roller” microneedling performed by aestheticians), she added, “gives the best results for the money and produces clinical results that mirror those achieved with lasers.”
Dr. Wang agreed that even recommending less expensive and less efficacious options such as over-the-counter creams can help alleviate patients’ concerns. “It shows that you are being holistic – not just caring for medical issues around pregnancy, but that you also take the emotional/psychological concerns of pregnant individuals and new parents seriously and that you recognize the impact of skin problems on quality of life. In the end, recommending something – in other words, providing some options, like creams or other therapies, for instance – is still, in my opinion, better than not recommending anything.”
Dr. Wang is involved with a study that is currently enrolling patients and that is evaluating the formation of early SG, which includes performing skin biopsies as soon as lesions appear.
The study had no funding. The study authors and Dr. Alster disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Substantial declines in mortality for most cancers
according to a new analysis.
Researchers found that rates for all cancers combined declined by 27% overall between 1971 and 2019 and decreased significantly for 12 of the 15 top cancer sites analyzed.
The data revealed even greater mortality declines for certain cancers in particular years. For example, mortality from lung cancer was 44% lower in 2019, compared with its peak rate in 1993, whereas it was only 13% lower, compared with morality rates in 1971.
“The cancer mortality rate has reduced considerably since 1971 overall and for most cancer sites because of improvements in prevention, early detection, and treatment,” lead author Ahmedin Jemal, DVM, PhD, American Cancer Society, Kennesaw, Ga., and colleagues wrote.
Advances in surgery, radiotherapy, chemotherapy, precision medicine, and combinations therapies over the past 5 decades have contributed to these significant declines in mortality, Dr. Jemal and colleagues explained. The researchers also credit the “expanded investment” in the National Cancer Institute’s annual budget following the 1971 National Cancer Act, which increased the budget 25-fold from $227 million in 1971 to $6 billion in 2019.
The report, published online Nov. 11, 2021, in JAMA Oncology, analyzed mortality rates for all cancers as well as the top 15 sites using the National Center for Health Statistics.
The researchers found that, overall, deaths declined significantly for all cancers over the study period. Some of the biggest headway since 1971 occurred for stomach and cervical cancers – with 72% and 69% lower mortality rates, respectively – as well as colorectal cancer (56%), oral cavity and pharynx cancer (43%), and ovarian cancer (41%). Mortality rates of female breast cancer and prostate cancer also dropped considerably – both by 39%.
“The decline in mortality for female breast, cervical, colorectal, and prostate cancer in part reflects increased detection (and removal) of premalignant lesions and early-stage cancers,” Dr. Jemal and colleagues noted.
Data suggest that screening likely explains about half of the observed decline in mortality from colorectal cancer between 1975 and 2002. A 2018 study also found that the use of adjuvant chemotherapy was responsible for 63% of the decline in mortality from female breast cancer between 2000 and 2012.
In addition, the authors noted, “the decline in lung, oral cavity and bladder cancers largely reflects reductions in smoking because of enhanced public awareness of the health consequences, implementation of increased cigarette excise taxes, and comprehensive smoke-free laws.”
However, mortality did increase in a few categories. For instance, the mortality rate from pancreatic cancer increased by 3% between 1971 and 2019, and by 8% for both esophageal and brain cancers. Mortality rates from cancer were also greater for 29% of the U.S. counties included in the analysis, mostly those in the South.
The increase in mortality from pancreatic cancer likely reflects the growing rates of obesity in the United States, along with no real advances in pancreatic cancer prevention, early detection, or treatment, the authors suggested. In addition, lack of progress in regions of the south may be related to unequal access to improvements in treatment compared with other parts of the country.
“Improving equity through investment in the social determinants of health and implementation research is critical to furthering the national cancer-control agenda,” the authors concluded.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a new analysis.
Researchers found that rates for all cancers combined declined by 27% overall between 1971 and 2019 and decreased significantly for 12 of the 15 top cancer sites analyzed.
The data revealed even greater mortality declines for certain cancers in particular years. For example, mortality from lung cancer was 44% lower in 2019, compared with its peak rate in 1993, whereas it was only 13% lower, compared with morality rates in 1971.
“The cancer mortality rate has reduced considerably since 1971 overall and for most cancer sites because of improvements in prevention, early detection, and treatment,” lead author Ahmedin Jemal, DVM, PhD, American Cancer Society, Kennesaw, Ga., and colleagues wrote.
Advances in surgery, radiotherapy, chemotherapy, precision medicine, and combinations therapies over the past 5 decades have contributed to these significant declines in mortality, Dr. Jemal and colleagues explained. The researchers also credit the “expanded investment” in the National Cancer Institute’s annual budget following the 1971 National Cancer Act, which increased the budget 25-fold from $227 million in 1971 to $6 billion in 2019.
The report, published online Nov. 11, 2021, in JAMA Oncology, analyzed mortality rates for all cancers as well as the top 15 sites using the National Center for Health Statistics.
The researchers found that, overall, deaths declined significantly for all cancers over the study period. Some of the biggest headway since 1971 occurred for stomach and cervical cancers – with 72% and 69% lower mortality rates, respectively – as well as colorectal cancer (56%), oral cavity and pharynx cancer (43%), and ovarian cancer (41%). Mortality rates of female breast cancer and prostate cancer also dropped considerably – both by 39%.
“The decline in mortality for female breast, cervical, colorectal, and prostate cancer in part reflects increased detection (and removal) of premalignant lesions and early-stage cancers,” Dr. Jemal and colleagues noted.
Data suggest that screening likely explains about half of the observed decline in mortality from colorectal cancer between 1975 and 2002. A 2018 study also found that the use of adjuvant chemotherapy was responsible for 63% of the decline in mortality from female breast cancer between 2000 and 2012.
In addition, the authors noted, “the decline in lung, oral cavity and bladder cancers largely reflects reductions in smoking because of enhanced public awareness of the health consequences, implementation of increased cigarette excise taxes, and comprehensive smoke-free laws.”
However, mortality did increase in a few categories. For instance, the mortality rate from pancreatic cancer increased by 3% between 1971 and 2019, and by 8% for both esophageal and brain cancers. Mortality rates from cancer were also greater for 29% of the U.S. counties included in the analysis, mostly those in the South.
The increase in mortality from pancreatic cancer likely reflects the growing rates of obesity in the United States, along with no real advances in pancreatic cancer prevention, early detection, or treatment, the authors suggested. In addition, lack of progress in regions of the south may be related to unequal access to improvements in treatment compared with other parts of the country.
“Improving equity through investment in the social determinants of health and implementation research is critical to furthering the national cancer-control agenda,” the authors concluded.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a new analysis.
Researchers found that rates for all cancers combined declined by 27% overall between 1971 and 2019 and decreased significantly for 12 of the 15 top cancer sites analyzed.
The data revealed even greater mortality declines for certain cancers in particular years. For example, mortality from lung cancer was 44% lower in 2019, compared with its peak rate in 1993, whereas it was only 13% lower, compared with morality rates in 1971.
“The cancer mortality rate has reduced considerably since 1971 overall and for most cancer sites because of improvements in prevention, early detection, and treatment,” lead author Ahmedin Jemal, DVM, PhD, American Cancer Society, Kennesaw, Ga., and colleagues wrote.
Advances in surgery, radiotherapy, chemotherapy, precision medicine, and combinations therapies over the past 5 decades have contributed to these significant declines in mortality, Dr. Jemal and colleagues explained. The researchers also credit the “expanded investment” in the National Cancer Institute’s annual budget following the 1971 National Cancer Act, which increased the budget 25-fold from $227 million in 1971 to $6 billion in 2019.
The report, published online Nov. 11, 2021, in JAMA Oncology, analyzed mortality rates for all cancers as well as the top 15 sites using the National Center for Health Statistics.
The researchers found that, overall, deaths declined significantly for all cancers over the study period. Some of the biggest headway since 1971 occurred for stomach and cervical cancers – with 72% and 69% lower mortality rates, respectively – as well as colorectal cancer (56%), oral cavity and pharynx cancer (43%), and ovarian cancer (41%). Mortality rates of female breast cancer and prostate cancer also dropped considerably – both by 39%.
“The decline in mortality for female breast, cervical, colorectal, and prostate cancer in part reflects increased detection (and removal) of premalignant lesions and early-stage cancers,” Dr. Jemal and colleagues noted.
Data suggest that screening likely explains about half of the observed decline in mortality from colorectal cancer between 1975 and 2002. A 2018 study also found that the use of adjuvant chemotherapy was responsible for 63% of the decline in mortality from female breast cancer between 2000 and 2012.
In addition, the authors noted, “the decline in lung, oral cavity and bladder cancers largely reflects reductions in smoking because of enhanced public awareness of the health consequences, implementation of increased cigarette excise taxes, and comprehensive smoke-free laws.”
However, mortality did increase in a few categories. For instance, the mortality rate from pancreatic cancer increased by 3% between 1971 and 2019, and by 8% for both esophageal and brain cancers. Mortality rates from cancer were also greater for 29% of the U.S. counties included in the analysis, mostly those in the South.
The increase in mortality from pancreatic cancer likely reflects the growing rates of obesity in the United States, along with no real advances in pancreatic cancer prevention, early detection, or treatment, the authors suggested. In addition, lack of progress in regions of the south may be related to unequal access to improvements in treatment compared with other parts of the country.
“Improving equity through investment in the social determinants of health and implementation research is critical to furthering the national cancer-control agenda,” the authors concluded.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA ONCOLOGY
Most community physicians say clinical pathways improve care
Dr. Wong presented findings from the journal’s annual Oncology Clinical Pathways Benchmarking Survey at the Oncology Clinical Pathways Congress, which was held in October. As fee for service gives way to performance-based and risk-bearing reimbursements, he said, “we are observing renewed interest in pathways implementation among a more diverse group of [health care providers].”
More survey respondents said they expected to implement pathways within the next 2 years than in past surveys. “I think it’s partly because payors are starting to delegate more care decisions back to oncology practices, making them more accountable for the care they provide,” Dr. Wong said.
The 2021 survey included 871 respondents, most of them direct care providers based in community practices. At 94%, most said they believed clinical pathways increased quality of care, 87% said they improved clinical outcomes, and 84% said they controlled costs.
Also presented at the meeting were preliminary findings of the JCP 2021 Care Pathways Working Group, which identified barriers to wider clinical pathways use. These include a fragmented health care system, minimal interoperability between systems, lack of integration into practice work flows, lack of reduction in administrative burden and lack of understanding by payers of the impact of social determinants of health.
Oncology clinical pathways are protocols and drug regimens for cancer care. They are used by oncology practices, academic medical centers, health systems, payors, and third-party vendors to address efficacy, safety, tolerability, and cost, but physicians have raised concerns about the administrative burden of working with pathways or pathways that emphasize cost-cutting at the expense of treatment choices or the flexibility to respond to unique patient circumstances.
The American Society of Clinical Oncology responded to member concerns about pathways in a 2016 Policy Statement on Clinical Pathways in Oncology. The following year, ASCO issued Criteria for High-Quality Clinical Pathways, offering a mechanism for evaluating the quality of a pathway, which according to ASCO’s criteria should be expert driven, evidence based, patient focused, up to date, and comprehensive, with multiple stakeholder input.
“There’s uncertainty among providers as the health care system continues to evolve toward value-based care models,” said Stephen Grubbs, MD, ASCO’s vice president of care delivery. “There are a lot of challenges. Practices are at different points in their journey toward value-based care and how to reconcile their care delivery models with the alternate payment models.”
Robin T. Zon, MD, FACP, FASCO, a medical oncologist at Michiana Hematology Oncology in Mishawaka, Ind., and chair of ASCO’s Pathways Task Force, which has since disbanded, was asked if she thought integration of pathways into practice has improved in the 4 years since the task force completed its work. “We think so, but we don’t have the data to support that conclusion,” she replied. “We were concerned about how we could make life easier for clinicians having to deal with preauthorization and helping them with the administrative burdens. Our society is trying to point to the path forward.”
Social determinants come to the fore
Also widely discussed at the congress was the need for greater equity in health care and greater responsiveness to social determinants of health, Dr. Wong said. Disparities in care are common throughout chronic disease care, and social determinants are getting more attention with the growing emphasis on patient-centered care.
Patient preferences and circumstances come into play, for example, when the patient can’t afford a prescribed treatment, or if a recommended protocol of infusions for 4 or 5 days in a row conflicts with the patient’s need to keep working. “If you don’t have a caregiver readily able to take you to the doctor’s office, that impacts your choice of treatment,” Dr. Wong said. Other social factors include geography, life experience, tolerance for side effects, and racial or ethnic diversity.
“I think the personalized approach is growing – compared to 4 or 5 years ago, when social determinants and patient preferences weren’t really talked about,” he said. How payors incorporate these considerations varies widely, but larger practices are starting to talk to payors about taking on financial risk, and clinical pathways can help them control risk and cost. “It has to be a collaborative process with whomever you’re talking to. The movement will be successful to the degree we collaborate in a common direction.”
Complicated treatments
Ray Page, DO, PhD, FASCO, a medical oncologist and hematologist at the Center for Cancer and Blood Disorders in Fort Worth, Tex., said his group has used clinical pathways, offered by Elsevier and originally developed at the University of Pittsburgh Medical Center, since 2007. “It’s part of the culture of our practice, and a requirement to work here. Cancer medicine is becoming so complicated, no oncologist can humanly keep track of it all. You’ve got to have good tools,” he said.
“Part of the nuance of dealing with insurers is that we’ve tried to negotiate using our compliance with evidence-based clinical pathways.” Collaboration is the ultimate goal, Dr. Page said. “But that gets harder as health care becomes more corporatized and vertically integrated.”
Alan Balch, PhD, CEO of the National Patient Advocate Foundation in Washington, D.C., said that well-designed pathways offer a way to ensure that evidence-based cancer care is practiced, and that providers are presented with a short list of treatment options based on evidence-based guidelines like those from the National Comprehensive Cancer Network.
“But if you want to be consumer centric, reflecting the circumstances of the individual patient, you either have to make your pathway more sophisticated and nuanced in the choices it offers – or see it as just one tool in treatment decision-making, while allowing other, patient-centered processes by which the patient’s circumstances and preferences are considered.” Is there a name for that process? “It’s called shared decision-making,” Dr. Balch replied.
“I’m optimistic that oncology care is becoming more person centered, whether by pathways or other means,” he said. “How long that will take, and in what form, is another conversation. But there is greater awareness of the need.”
People are talking to each other more about pathways implementation, Dr. Zon added. Pathways uptake will probably never be 100%, and the large academic medical centers will continue to have their own ways of caring for the sickest of the sick outside of the pathways.
Dr. Zon wondered if there could be a more comprehensive or universal oncology pathway incorporating symptom control, triaging, preventive screening, supportive and palliative care, and the end of life, all of which have fairly standardized approaches. “At the congress, I proposed thinking about a different approach for the pathways model – one that is not only more patient centric, but incorporates social determinants of health and patient experience, reflecting different cultures and communities, combining these other approaches to be more comprehensive and supporting best approaches to cancer care while reducing total costs of care.”
Dr. Wong presented findings from the journal’s annual Oncology Clinical Pathways Benchmarking Survey at the Oncology Clinical Pathways Congress, which was held in October. As fee for service gives way to performance-based and risk-bearing reimbursements, he said, “we are observing renewed interest in pathways implementation among a more diverse group of [health care providers].”
More survey respondents said they expected to implement pathways within the next 2 years than in past surveys. “I think it’s partly because payors are starting to delegate more care decisions back to oncology practices, making them more accountable for the care they provide,” Dr. Wong said.
The 2021 survey included 871 respondents, most of them direct care providers based in community practices. At 94%, most said they believed clinical pathways increased quality of care, 87% said they improved clinical outcomes, and 84% said they controlled costs.
Also presented at the meeting were preliminary findings of the JCP 2021 Care Pathways Working Group, which identified barriers to wider clinical pathways use. These include a fragmented health care system, minimal interoperability between systems, lack of integration into practice work flows, lack of reduction in administrative burden and lack of understanding by payers of the impact of social determinants of health.
Oncology clinical pathways are protocols and drug regimens for cancer care. They are used by oncology practices, academic medical centers, health systems, payors, and third-party vendors to address efficacy, safety, tolerability, and cost, but physicians have raised concerns about the administrative burden of working with pathways or pathways that emphasize cost-cutting at the expense of treatment choices or the flexibility to respond to unique patient circumstances.
The American Society of Clinical Oncology responded to member concerns about pathways in a 2016 Policy Statement on Clinical Pathways in Oncology. The following year, ASCO issued Criteria for High-Quality Clinical Pathways, offering a mechanism for evaluating the quality of a pathway, which according to ASCO’s criteria should be expert driven, evidence based, patient focused, up to date, and comprehensive, with multiple stakeholder input.
“There’s uncertainty among providers as the health care system continues to evolve toward value-based care models,” said Stephen Grubbs, MD, ASCO’s vice president of care delivery. “There are a lot of challenges. Practices are at different points in their journey toward value-based care and how to reconcile their care delivery models with the alternate payment models.”
Robin T. Zon, MD, FACP, FASCO, a medical oncologist at Michiana Hematology Oncology in Mishawaka, Ind., and chair of ASCO’s Pathways Task Force, which has since disbanded, was asked if she thought integration of pathways into practice has improved in the 4 years since the task force completed its work. “We think so, but we don’t have the data to support that conclusion,” she replied. “We were concerned about how we could make life easier for clinicians having to deal with preauthorization and helping them with the administrative burdens. Our society is trying to point to the path forward.”
Social determinants come to the fore
Also widely discussed at the congress was the need for greater equity in health care and greater responsiveness to social determinants of health, Dr. Wong said. Disparities in care are common throughout chronic disease care, and social determinants are getting more attention with the growing emphasis on patient-centered care.
Patient preferences and circumstances come into play, for example, when the patient can’t afford a prescribed treatment, or if a recommended protocol of infusions for 4 or 5 days in a row conflicts with the patient’s need to keep working. “If you don’t have a caregiver readily able to take you to the doctor’s office, that impacts your choice of treatment,” Dr. Wong said. Other social factors include geography, life experience, tolerance for side effects, and racial or ethnic diversity.
“I think the personalized approach is growing – compared to 4 or 5 years ago, when social determinants and patient preferences weren’t really talked about,” he said. How payors incorporate these considerations varies widely, but larger practices are starting to talk to payors about taking on financial risk, and clinical pathways can help them control risk and cost. “It has to be a collaborative process with whomever you’re talking to. The movement will be successful to the degree we collaborate in a common direction.”
Complicated treatments
Ray Page, DO, PhD, FASCO, a medical oncologist and hematologist at the Center for Cancer and Blood Disorders in Fort Worth, Tex., said his group has used clinical pathways, offered by Elsevier and originally developed at the University of Pittsburgh Medical Center, since 2007. “It’s part of the culture of our practice, and a requirement to work here. Cancer medicine is becoming so complicated, no oncologist can humanly keep track of it all. You’ve got to have good tools,” he said.
“Part of the nuance of dealing with insurers is that we’ve tried to negotiate using our compliance with evidence-based clinical pathways.” Collaboration is the ultimate goal, Dr. Page said. “But that gets harder as health care becomes more corporatized and vertically integrated.”
Alan Balch, PhD, CEO of the National Patient Advocate Foundation in Washington, D.C., said that well-designed pathways offer a way to ensure that evidence-based cancer care is practiced, and that providers are presented with a short list of treatment options based on evidence-based guidelines like those from the National Comprehensive Cancer Network.
“But if you want to be consumer centric, reflecting the circumstances of the individual patient, you either have to make your pathway more sophisticated and nuanced in the choices it offers – or see it as just one tool in treatment decision-making, while allowing other, patient-centered processes by which the patient’s circumstances and preferences are considered.” Is there a name for that process? “It’s called shared decision-making,” Dr. Balch replied.
“I’m optimistic that oncology care is becoming more person centered, whether by pathways or other means,” he said. “How long that will take, and in what form, is another conversation. But there is greater awareness of the need.”
People are talking to each other more about pathways implementation, Dr. Zon added. Pathways uptake will probably never be 100%, and the large academic medical centers will continue to have their own ways of caring for the sickest of the sick outside of the pathways.
Dr. Zon wondered if there could be a more comprehensive or universal oncology pathway incorporating symptom control, triaging, preventive screening, supportive and palliative care, and the end of life, all of which have fairly standardized approaches. “At the congress, I proposed thinking about a different approach for the pathways model – one that is not only more patient centric, but incorporates social determinants of health and patient experience, reflecting different cultures and communities, combining these other approaches to be more comprehensive and supporting best approaches to cancer care while reducing total costs of care.”
Dr. Wong presented findings from the journal’s annual Oncology Clinical Pathways Benchmarking Survey at the Oncology Clinical Pathways Congress, which was held in October. As fee for service gives way to performance-based and risk-bearing reimbursements, he said, “we are observing renewed interest in pathways implementation among a more diverse group of [health care providers].”
More survey respondents said they expected to implement pathways within the next 2 years than in past surveys. “I think it’s partly because payors are starting to delegate more care decisions back to oncology practices, making them more accountable for the care they provide,” Dr. Wong said.
The 2021 survey included 871 respondents, most of them direct care providers based in community practices. At 94%, most said they believed clinical pathways increased quality of care, 87% said they improved clinical outcomes, and 84% said they controlled costs.
Also presented at the meeting were preliminary findings of the JCP 2021 Care Pathways Working Group, which identified barriers to wider clinical pathways use. These include a fragmented health care system, minimal interoperability between systems, lack of integration into practice work flows, lack of reduction in administrative burden and lack of understanding by payers of the impact of social determinants of health.
Oncology clinical pathways are protocols and drug regimens for cancer care. They are used by oncology practices, academic medical centers, health systems, payors, and third-party vendors to address efficacy, safety, tolerability, and cost, but physicians have raised concerns about the administrative burden of working with pathways or pathways that emphasize cost-cutting at the expense of treatment choices or the flexibility to respond to unique patient circumstances.
The American Society of Clinical Oncology responded to member concerns about pathways in a 2016 Policy Statement on Clinical Pathways in Oncology. The following year, ASCO issued Criteria for High-Quality Clinical Pathways, offering a mechanism for evaluating the quality of a pathway, which according to ASCO’s criteria should be expert driven, evidence based, patient focused, up to date, and comprehensive, with multiple stakeholder input.
“There’s uncertainty among providers as the health care system continues to evolve toward value-based care models,” said Stephen Grubbs, MD, ASCO’s vice president of care delivery. “There are a lot of challenges. Practices are at different points in their journey toward value-based care and how to reconcile their care delivery models with the alternate payment models.”
Robin T. Zon, MD, FACP, FASCO, a medical oncologist at Michiana Hematology Oncology in Mishawaka, Ind., and chair of ASCO’s Pathways Task Force, which has since disbanded, was asked if she thought integration of pathways into practice has improved in the 4 years since the task force completed its work. “We think so, but we don’t have the data to support that conclusion,” she replied. “We were concerned about how we could make life easier for clinicians having to deal with preauthorization and helping them with the administrative burdens. Our society is trying to point to the path forward.”
Social determinants come to the fore
Also widely discussed at the congress was the need for greater equity in health care and greater responsiveness to social determinants of health, Dr. Wong said. Disparities in care are common throughout chronic disease care, and social determinants are getting more attention with the growing emphasis on patient-centered care.
Patient preferences and circumstances come into play, for example, when the patient can’t afford a prescribed treatment, or if a recommended protocol of infusions for 4 or 5 days in a row conflicts with the patient’s need to keep working. “If you don’t have a caregiver readily able to take you to the doctor’s office, that impacts your choice of treatment,” Dr. Wong said. Other social factors include geography, life experience, tolerance for side effects, and racial or ethnic diversity.
“I think the personalized approach is growing – compared to 4 or 5 years ago, when social determinants and patient preferences weren’t really talked about,” he said. How payors incorporate these considerations varies widely, but larger practices are starting to talk to payors about taking on financial risk, and clinical pathways can help them control risk and cost. “It has to be a collaborative process with whomever you’re talking to. The movement will be successful to the degree we collaborate in a common direction.”
Complicated treatments
Ray Page, DO, PhD, FASCO, a medical oncologist and hematologist at the Center for Cancer and Blood Disorders in Fort Worth, Tex., said his group has used clinical pathways, offered by Elsevier and originally developed at the University of Pittsburgh Medical Center, since 2007. “It’s part of the culture of our practice, and a requirement to work here. Cancer medicine is becoming so complicated, no oncologist can humanly keep track of it all. You’ve got to have good tools,” he said.
“Part of the nuance of dealing with insurers is that we’ve tried to negotiate using our compliance with evidence-based clinical pathways.” Collaboration is the ultimate goal, Dr. Page said. “But that gets harder as health care becomes more corporatized and vertically integrated.”
Alan Balch, PhD, CEO of the National Patient Advocate Foundation in Washington, D.C., said that well-designed pathways offer a way to ensure that evidence-based cancer care is practiced, and that providers are presented with a short list of treatment options based on evidence-based guidelines like those from the National Comprehensive Cancer Network.
“But if you want to be consumer centric, reflecting the circumstances of the individual patient, you either have to make your pathway more sophisticated and nuanced in the choices it offers – or see it as just one tool in treatment decision-making, while allowing other, patient-centered processes by which the patient’s circumstances and preferences are considered.” Is there a name for that process? “It’s called shared decision-making,” Dr. Balch replied.
“I’m optimistic that oncology care is becoming more person centered, whether by pathways or other means,” he said. “How long that will take, and in what form, is another conversation. But there is greater awareness of the need.”
People are talking to each other more about pathways implementation, Dr. Zon added. Pathways uptake will probably never be 100%, and the large academic medical centers will continue to have their own ways of caring for the sickest of the sick outside of the pathways.
Dr. Zon wondered if there could be a more comprehensive or universal oncology pathway incorporating symptom control, triaging, preventive screening, supportive and palliative care, and the end of life, all of which have fairly standardized approaches. “At the congress, I proposed thinking about a different approach for the pathways model – one that is not only more patient centric, but incorporates social determinants of health and patient experience, reflecting different cultures and communities, combining these other approaches to be more comprehensive and supporting best approaches to cancer care while reducing total costs of care.”
Cannabinoids being studied for a variety of dermatologic conditions
.
“When you walk into places like CVS or Walgreens, you see lots of displays for CBD creams and oils,” Todd S. Anhalt, MD, said during the annual meeting of the Pacific Dermatologic Association. “The problem is, we don’t know what’s in them or who made them or how good they are. That’s going to be a problem for a while.”
According to Dr. Anhalt, clinical professor emeritus of dermatology at Stanford (Calif.) University, there are about 140 active cannabinoid compounds in cannabis, but the most important ones are THC and cannabidiol (CBD). There are three types of cannabinoids, based on where the cannabidiol is produced: endocannabinoids, which are produced in the human body; phytocannabinoids, which are derived from plants such as marijuana and hemp; and synthetic cannabinoids, which are derived in labs.
Dr. Anhalt described the endocannabinoid system as a conserved network of molecular signaling made of several components: signaling molecules (endocannabinoids), endocannabinoid receptors (CB-1 and CB-2), enzymes, and transporters. There is also overlap between cannabinoids and terpenes, which are responsible for flavor and aroma in plants and marijuana and can enhance the effects of CBD.
“For the most part, CB-1 receptors are in the central nervous system and CB-2 [receptors] are mostly in the periphery,” including the skin and digestive system, said Dr. Anhalt, who practices at the California Skin Institute in Los Altos, Calif. “This is interesting because one of the main conditions I recommend cannabidiol for is in patients with peripheral neuropathy, despite the fact they may be on all sorts of medications such as Neurontin and Lyrica or tricyclic antidepressants. Sometimes they don’t get much relief from those. I have had many patients tell me that they have had reduction of pain and increased functionality using the CBD creams.” CB-2 receptors, he noted, are located in keratinocytes, sensory receptors, sweat glands, fibroblasts, Langerhans cells, melanocytes, and sebaceous glands.
Recent research shows that the endocannabinoid system is involved in modulation of the CNS and in immune function, particularly skin homeostasis and barrier function. “We know that barrier function can be affected by the generation of oxidative species,” he said. “The stress that it causes can decrease barrier function and lead to cytokine release and itch. CBDs have been shown to enter cells, target and upregulate genes with decreased oxidation and inflammation, and protect membrane integrity in skin cells. Therefore, this might be helpful in atopic dermatitis.” Other potential uses in dermatology include wound healing, acne, hair growth modulation, skin and hair pigmentation, skin infections, psoriasis, and cutaneous malignancies, as well as neuropathic pain.
Evidence is strongest for neuropathic pain, he said, which is mediated by CB-1 receptors peripherally, followed by itch and atopic dermatitis. The authors of a 2017 systematic review concluded that “low-strength” evidence exists to suggest that cannabis alleviates neuropathic pain, with insufficient evidence for other types of pain.
Topical CBD comes in various forms: oils (usually hemp oil), creams, and lotions, Dr. Anhalt said. “I advise patients to apply it 2-4 times per day depending on how anxious or uncomfortable they are. It takes my patients 10 days to 2 weeks before they notice anything at all.”
For atopic dermatitis, it could be useful “not to use it instead of a moisturizer, but as a moisturizer,” Dr. Anhalt advised. “You can have a patient get big jars of CBD creams and lotions. They may have to try a few before they find one that they really like, but you can replace all of the other moisturizers that you’re using right now in patients who have a lot of itch.”
As for CBD’s effect on peripheral neuropathy, the medical literature is lacking, but some studies show low to moderate evidence of efficacy. For example, a Cochrane Review found that a 30% or greater pain reduction was achieved by 39% of patients who used cannabis-based treatments, vs. 33% of those on placebo.
“I would not suggest CBD as a first-line drug unless it’s very mild peripheral neuropathy, but for patients who are on gabapentin who are better but not better enough, this is an excellent adjunct,” Dr. Anhalt said. “It’s worth trying. It’s not too expensive and it’s really safe.”
The application of topical CBD to treat cutaneous malignancies has not yet shown evidence of significant efficacy, while using CBDs for acne holds promise. “The endogenous cannabinoid system is involved in the production of lipids,” he said. “Cannabinoids have an antilipogenic activity, so they decrease sebum production. CBD could help patients with mild acne who are reluctant to use other types of medications. For this and other potential dermatologic applications, lots more studies need to be done.”
Dr. Anhalt reported having no financial disclosures.
.
“When you walk into places like CVS or Walgreens, you see lots of displays for CBD creams and oils,” Todd S. Anhalt, MD, said during the annual meeting of the Pacific Dermatologic Association. “The problem is, we don’t know what’s in them or who made them or how good they are. That’s going to be a problem for a while.”
According to Dr. Anhalt, clinical professor emeritus of dermatology at Stanford (Calif.) University, there are about 140 active cannabinoid compounds in cannabis, but the most important ones are THC and cannabidiol (CBD). There are three types of cannabinoids, based on where the cannabidiol is produced: endocannabinoids, which are produced in the human body; phytocannabinoids, which are derived from plants such as marijuana and hemp; and synthetic cannabinoids, which are derived in labs.
Dr. Anhalt described the endocannabinoid system as a conserved network of molecular signaling made of several components: signaling molecules (endocannabinoids), endocannabinoid receptors (CB-1 and CB-2), enzymes, and transporters. There is also overlap between cannabinoids and terpenes, which are responsible for flavor and aroma in plants and marijuana and can enhance the effects of CBD.
“For the most part, CB-1 receptors are in the central nervous system and CB-2 [receptors] are mostly in the periphery,” including the skin and digestive system, said Dr. Anhalt, who practices at the California Skin Institute in Los Altos, Calif. “This is interesting because one of the main conditions I recommend cannabidiol for is in patients with peripheral neuropathy, despite the fact they may be on all sorts of medications such as Neurontin and Lyrica or tricyclic antidepressants. Sometimes they don’t get much relief from those. I have had many patients tell me that they have had reduction of pain and increased functionality using the CBD creams.” CB-2 receptors, he noted, are located in keratinocytes, sensory receptors, sweat glands, fibroblasts, Langerhans cells, melanocytes, and sebaceous glands.
Recent research shows that the endocannabinoid system is involved in modulation of the CNS and in immune function, particularly skin homeostasis and barrier function. “We know that barrier function can be affected by the generation of oxidative species,” he said. “The stress that it causes can decrease barrier function and lead to cytokine release and itch. CBDs have been shown to enter cells, target and upregulate genes with decreased oxidation and inflammation, and protect membrane integrity in skin cells. Therefore, this might be helpful in atopic dermatitis.” Other potential uses in dermatology include wound healing, acne, hair growth modulation, skin and hair pigmentation, skin infections, psoriasis, and cutaneous malignancies, as well as neuropathic pain.
Evidence is strongest for neuropathic pain, he said, which is mediated by CB-1 receptors peripherally, followed by itch and atopic dermatitis. The authors of a 2017 systematic review concluded that “low-strength” evidence exists to suggest that cannabis alleviates neuropathic pain, with insufficient evidence for other types of pain.
Topical CBD comes in various forms: oils (usually hemp oil), creams, and lotions, Dr. Anhalt said. “I advise patients to apply it 2-4 times per day depending on how anxious or uncomfortable they are. It takes my patients 10 days to 2 weeks before they notice anything at all.”
For atopic dermatitis, it could be useful “not to use it instead of a moisturizer, but as a moisturizer,” Dr. Anhalt advised. “You can have a patient get big jars of CBD creams and lotions. They may have to try a few before they find one that they really like, but you can replace all of the other moisturizers that you’re using right now in patients who have a lot of itch.”
As for CBD’s effect on peripheral neuropathy, the medical literature is lacking, but some studies show low to moderate evidence of efficacy. For example, a Cochrane Review found that a 30% or greater pain reduction was achieved by 39% of patients who used cannabis-based treatments, vs. 33% of those on placebo.
“I would not suggest CBD as a first-line drug unless it’s very mild peripheral neuropathy, but for patients who are on gabapentin who are better but not better enough, this is an excellent adjunct,” Dr. Anhalt said. “It’s worth trying. It’s not too expensive and it’s really safe.”
The application of topical CBD to treat cutaneous malignancies has not yet shown evidence of significant efficacy, while using CBDs for acne holds promise. “The endogenous cannabinoid system is involved in the production of lipids,” he said. “Cannabinoids have an antilipogenic activity, so they decrease sebum production. CBD could help patients with mild acne who are reluctant to use other types of medications. For this and other potential dermatologic applications, lots more studies need to be done.”
Dr. Anhalt reported having no financial disclosures.
.
“When you walk into places like CVS or Walgreens, you see lots of displays for CBD creams and oils,” Todd S. Anhalt, MD, said during the annual meeting of the Pacific Dermatologic Association. “The problem is, we don’t know what’s in them or who made them or how good they are. That’s going to be a problem for a while.”
According to Dr. Anhalt, clinical professor emeritus of dermatology at Stanford (Calif.) University, there are about 140 active cannabinoid compounds in cannabis, but the most important ones are THC and cannabidiol (CBD). There are three types of cannabinoids, based on where the cannabidiol is produced: endocannabinoids, which are produced in the human body; phytocannabinoids, which are derived from plants such as marijuana and hemp; and synthetic cannabinoids, which are derived in labs.
Dr. Anhalt described the endocannabinoid system as a conserved network of molecular signaling made of several components: signaling molecules (endocannabinoids), endocannabinoid receptors (CB-1 and CB-2), enzymes, and transporters. There is also overlap between cannabinoids and terpenes, which are responsible for flavor and aroma in plants and marijuana and can enhance the effects of CBD.
“For the most part, CB-1 receptors are in the central nervous system and CB-2 [receptors] are mostly in the periphery,” including the skin and digestive system, said Dr. Anhalt, who practices at the California Skin Institute in Los Altos, Calif. “This is interesting because one of the main conditions I recommend cannabidiol for is in patients with peripheral neuropathy, despite the fact they may be on all sorts of medications such as Neurontin and Lyrica or tricyclic antidepressants. Sometimes they don’t get much relief from those. I have had many patients tell me that they have had reduction of pain and increased functionality using the CBD creams.” CB-2 receptors, he noted, are located in keratinocytes, sensory receptors, sweat glands, fibroblasts, Langerhans cells, melanocytes, and sebaceous glands.
Recent research shows that the endocannabinoid system is involved in modulation of the CNS and in immune function, particularly skin homeostasis and barrier function. “We know that barrier function can be affected by the generation of oxidative species,” he said. “The stress that it causes can decrease barrier function and lead to cytokine release and itch. CBDs have been shown to enter cells, target and upregulate genes with decreased oxidation and inflammation, and protect membrane integrity in skin cells. Therefore, this might be helpful in atopic dermatitis.” Other potential uses in dermatology include wound healing, acne, hair growth modulation, skin and hair pigmentation, skin infections, psoriasis, and cutaneous malignancies, as well as neuropathic pain.
Evidence is strongest for neuropathic pain, he said, which is mediated by CB-1 receptors peripherally, followed by itch and atopic dermatitis. The authors of a 2017 systematic review concluded that “low-strength” evidence exists to suggest that cannabis alleviates neuropathic pain, with insufficient evidence for other types of pain.
Topical CBD comes in various forms: oils (usually hemp oil), creams, and lotions, Dr. Anhalt said. “I advise patients to apply it 2-4 times per day depending on how anxious or uncomfortable they are. It takes my patients 10 days to 2 weeks before they notice anything at all.”
For atopic dermatitis, it could be useful “not to use it instead of a moisturizer, but as a moisturizer,” Dr. Anhalt advised. “You can have a patient get big jars of CBD creams and lotions. They may have to try a few before they find one that they really like, but you can replace all of the other moisturizers that you’re using right now in patients who have a lot of itch.”
As for CBD’s effect on peripheral neuropathy, the medical literature is lacking, but some studies show low to moderate evidence of efficacy. For example, a Cochrane Review found that a 30% or greater pain reduction was achieved by 39% of patients who used cannabis-based treatments, vs. 33% of those on placebo.
“I would not suggest CBD as a first-line drug unless it’s very mild peripheral neuropathy, but for patients who are on gabapentin who are better but not better enough, this is an excellent adjunct,” Dr. Anhalt said. “It’s worth trying. It’s not too expensive and it’s really safe.”
The application of topical CBD to treat cutaneous malignancies has not yet shown evidence of significant efficacy, while using CBDs for acne holds promise. “The endogenous cannabinoid system is involved in the production of lipids,” he said. “Cannabinoids have an antilipogenic activity, so they decrease sebum production. CBD could help patients with mild acne who are reluctant to use other types of medications. For this and other potential dermatologic applications, lots more studies need to be done.”
Dr. Anhalt reported having no financial disclosures.
FROM PDA 2021
Should you tell your doctor that you’re a doctor?
The question drew spirited debate when urologist Ashley Winter, MD, made a simple, straightforward request on Twitter: “If you are a doctor & you come to an appointment please tell me you are a doctor, not because I will treat you differently but because it’s easier to speak in jargon.”
She later added, “This doesn’t’ mean I would be less patient-focused or emotional with a physician or other [healthcare worker]. Just means that, instead of saying ‘you will have a catheter draining your urine to a bag,’ I can say, ‘you will have a Foley.’ ”
The Tweet followed an encounter with a patient who told Dr. Winter that he was a doctor only after she had gone to some length explaining a surgical procedure in lay terms.
“I explained the surgery, obviously assuming he was an intelligent adult, but using fully layman’s terms,” she said in an interview. The patient then told her that he was a doctor. “I guess I felt this embarrassment — I wouldn’t have treated him differently, but I just could have discussed the procedure with him in more professional terms.”
“To some extent, it was my own fault,” she commented in an interview. “I didn’t take the time to ask [about his work] at the beginning of the consultation, but that’s a fine line, also,” added Dr. Winter, a urologist and sexual medicine physician in Portland, Ore.
“You know that patient is there because they want care from you and it’s not necessarily always at the forefront of importance to be asking them what they do for their work, but alternatively, if you don’t ask then you put them in this position where they have to find a way to go ahead and tell you.”
Several people chimed in on the thread to voice their thoughts on the matter. Some commiserated with Dr. Winter’s experience:
“I took care of a retired cardiologist in the hospital as a second-year resident and honest to god he let me ramble on ‘explaining’ his echo result and never told me. I found out a couple days later and wanted to die,” posted @MaddyAndrewsMD.
Another recalled a similarly embarrassing experience when she “went on and on” discussing headaches with a patient whose husband “was in the corner smirking.”
“They told my attending later [that the] husband was a retired FM doc who practiced medicine longer than I’ve been alive. I wanted to die,” posted @JSinghDO.
Many on the thread, though, were doctors and other healthcare professionals speaking as patients. Some said they didn’t want to disclose their status as a healthcare provider because they felt it affected the care they received.
For example, @drhelenrainford commented: “In my experience my care is less ‘caring’ when they know I am a [doctor]. I get spoken to like they are discussing a patient with me — no empathy just facts and difficult results just blurted out without consideration. Awful awful time as an inpatient …but that’s another story!”
@Dr_B_Ring said: “Nope – You and I speak different jargon – I would want you to speak to me like a human that doesn’t know your jargon. My ego would get in the way of asking about the acronyms I don’t know if you knew I was a fellow physician.”
Conversely, @lozzlemcfozzle said: “Honestly I prefer not to tell my Doctors — I’ve found people skip explanations assuming I ‘know,’ or seem a little nervous when I tell them!”
Others said they felt uncomfortable — pretentious, even — in announcing their status, or worried that they might come across as expecting special care.
“It’s such a tough needle to thread. Want to tell people early but not come off as demanding special treatment, but don’t want to wait too long and it seems like a trap,” said @MDaware.
Twitter user @MsBabyCatcher wrote: “I have a hard time doing this because I don’t want people to think I’m being pretentious or going to micromanage/dictate care.”
Replying to @MsBabyCatcher, @RedStethoscope wrote: “I used to think this too until I got [very poor] care a few times, and was advised by other doctor moms to ‘play the doctor card.’ I have gotten better/more compassionate care by making sure it’s clear that I’m a physician (which is junk, but here we are).”
Several of those responding used the words “tricky” and “awkward,” suggesting a common theme for doctors presenting as patients.
“I struggle with this. My 5-year-old broke her arm this weekend, we spent hours in the ED, of my own hospital, I never mentioned it because I didn’t want to get preferential care. But as they were explaining her type of fracture, it felt awkward and inefficient,” said @lindsay_petty.
To avoid the awkwardness, a number of respondents said they purposefully use medical jargon to open up a conversation rather than just offering up the information that they are a doctor.
Still others offered suggestions on how to broach the subject more directly when presenting as a patient:
‘”Just FYI I’m a X doc but I’m here because I really want your help and advice!” That’s what I usually do,” wrote @drcakefm.
@BeeSting14618 Tweeted: “I usually say ‘I know some of this but I’m here because I want YOUR guidance. Also I may ask dumb questions, and I’ll tell you if a question is asking your opinion or making a request.’”
A few others injected a bit of humor: “I just do the 14-part handshake that only doctors know. Is that not customary?” quipped @Branmiz25.
“Ah yes, that transmits the entire [history of present illness],” replied Dr. Winter.
Jokes aside, the topic is obviously one that touched on a shared experience among healthcare providers, Dr. Winter commented. The Twitter thread she started just “blew up.”
That’s typically a sign that the Tweet is relatable for a lot of people, she said.
“It’s definitely something that all of us as care providers and as patients understand. It’s a funny, awkward thing that can really change an interaction, so we probably all feel pretty strongly about our experiences related to that,” she added.
The debate begs the question: Is there a duty or ethical reason to disclose?
“I definitely think it is very reasonable to disclose that one is a medical professional to another doctor,” medical ethicist Charlotte Blease, PhD, said in an interview. “There are good reasons to believe doing so might make a difference to the quality of communication and transparency.”
If the ability to use medical terminology or jargon more freely improves patient understanding, autonomy, and shared decision-making, then it may be of benefit, said Dr. Blease, a Keane OpenNotes Scholar at Beth Israel Deaconess Medical Center in Boston.
“Since doctors should strive to communicate effectively with every patient and to respect their unique needs and level of understanding, then I see no reason to deny that one is a medic,” she added.”
Knowing how to share the information is another story.
“This is something that affects all of us as physicians — we’re going to be patients at some point, right?” Dr. Winter commented. “But I don’t think how to disclose that is something that was ever brought up in my medical training.”
“Maybe there should just be a discussion of this one day when people are in medical school — maybe in a professionalism course — to broach this topic or look at if there’s any literature on outcomes related to disclosure of status or what are best practices,” she suggested.
A version of this article first appeared on Medscape.com.
The question drew spirited debate when urologist Ashley Winter, MD, made a simple, straightforward request on Twitter: “If you are a doctor & you come to an appointment please tell me you are a doctor, not because I will treat you differently but because it’s easier to speak in jargon.”
She later added, “This doesn’t’ mean I would be less patient-focused or emotional with a physician or other [healthcare worker]. Just means that, instead of saying ‘you will have a catheter draining your urine to a bag,’ I can say, ‘you will have a Foley.’ ”
The Tweet followed an encounter with a patient who told Dr. Winter that he was a doctor only after she had gone to some length explaining a surgical procedure in lay terms.
“I explained the surgery, obviously assuming he was an intelligent adult, but using fully layman’s terms,” she said in an interview. The patient then told her that he was a doctor. “I guess I felt this embarrassment — I wouldn’t have treated him differently, but I just could have discussed the procedure with him in more professional terms.”
“To some extent, it was my own fault,” she commented in an interview. “I didn’t take the time to ask [about his work] at the beginning of the consultation, but that’s a fine line, also,” added Dr. Winter, a urologist and sexual medicine physician in Portland, Ore.
“You know that patient is there because they want care from you and it’s not necessarily always at the forefront of importance to be asking them what they do for their work, but alternatively, if you don’t ask then you put them in this position where they have to find a way to go ahead and tell you.”
Several people chimed in on the thread to voice their thoughts on the matter. Some commiserated with Dr. Winter’s experience:
“I took care of a retired cardiologist in the hospital as a second-year resident and honest to god he let me ramble on ‘explaining’ his echo result and never told me. I found out a couple days later and wanted to die,” posted @MaddyAndrewsMD.
Another recalled a similarly embarrassing experience when she “went on and on” discussing headaches with a patient whose husband “was in the corner smirking.”
“They told my attending later [that the] husband was a retired FM doc who practiced medicine longer than I’ve been alive. I wanted to die,” posted @JSinghDO.
Many on the thread, though, were doctors and other healthcare professionals speaking as patients. Some said they didn’t want to disclose their status as a healthcare provider because they felt it affected the care they received.
For example, @drhelenrainford commented: “In my experience my care is less ‘caring’ when they know I am a [doctor]. I get spoken to like they are discussing a patient with me — no empathy just facts and difficult results just blurted out without consideration. Awful awful time as an inpatient …but that’s another story!”
@Dr_B_Ring said: “Nope – You and I speak different jargon – I would want you to speak to me like a human that doesn’t know your jargon. My ego would get in the way of asking about the acronyms I don’t know if you knew I was a fellow physician.”
Conversely, @lozzlemcfozzle said: “Honestly I prefer not to tell my Doctors — I’ve found people skip explanations assuming I ‘know,’ or seem a little nervous when I tell them!”
Others said they felt uncomfortable — pretentious, even — in announcing their status, or worried that they might come across as expecting special care.
“It’s such a tough needle to thread. Want to tell people early but not come off as demanding special treatment, but don’t want to wait too long and it seems like a trap,” said @MDaware.
Twitter user @MsBabyCatcher wrote: “I have a hard time doing this because I don’t want people to think I’m being pretentious or going to micromanage/dictate care.”
Replying to @MsBabyCatcher, @RedStethoscope wrote: “I used to think this too until I got [very poor] care a few times, and was advised by other doctor moms to ‘play the doctor card.’ I have gotten better/more compassionate care by making sure it’s clear that I’m a physician (which is junk, but here we are).”
Several of those responding used the words “tricky” and “awkward,” suggesting a common theme for doctors presenting as patients.
“I struggle with this. My 5-year-old broke her arm this weekend, we spent hours in the ED, of my own hospital, I never mentioned it because I didn’t want to get preferential care. But as they were explaining her type of fracture, it felt awkward and inefficient,” said @lindsay_petty.
To avoid the awkwardness, a number of respondents said they purposefully use medical jargon to open up a conversation rather than just offering up the information that they are a doctor.
Still others offered suggestions on how to broach the subject more directly when presenting as a patient:
‘”Just FYI I’m a X doc but I’m here because I really want your help and advice!” That’s what I usually do,” wrote @drcakefm.
@BeeSting14618 Tweeted: “I usually say ‘I know some of this but I’m here because I want YOUR guidance. Also I may ask dumb questions, and I’ll tell you if a question is asking your opinion or making a request.’”
A few others injected a bit of humor: “I just do the 14-part handshake that only doctors know. Is that not customary?” quipped @Branmiz25.
“Ah yes, that transmits the entire [history of present illness],” replied Dr. Winter.
Jokes aside, the topic is obviously one that touched on a shared experience among healthcare providers, Dr. Winter commented. The Twitter thread she started just “blew up.”
That’s typically a sign that the Tweet is relatable for a lot of people, she said.
“It’s definitely something that all of us as care providers and as patients understand. It’s a funny, awkward thing that can really change an interaction, so we probably all feel pretty strongly about our experiences related to that,” she added.
The debate begs the question: Is there a duty or ethical reason to disclose?
“I definitely think it is very reasonable to disclose that one is a medical professional to another doctor,” medical ethicist Charlotte Blease, PhD, said in an interview. “There are good reasons to believe doing so might make a difference to the quality of communication and transparency.”
If the ability to use medical terminology or jargon more freely improves patient understanding, autonomy, and shared decision-making, then it may be of benefit, said Dr. Blease, a Keane OpenNotes Scholar at Beth Israel Deaconess Medical Center in Boston.
“Since doctors should strive to communicate effectively with every patient and to respect their unique needs and level of understanding, then I see no reason to deny that one is a medic,” she added.”
Knowing how to share the information is another story.
“This is something that affects all of us as physicians — we’re going to be patients at some point, right?” Dr. Winter commented. “But I don’t think how to disclose that is something that was ever brought up in my medical training.”
“Maybe there should just be a discussion of this one day when people are in medical school — maybe in a professionalism course — to broach this topic or look at if there’s any literature on outcomes related to disclosure of status or what are best practices,” she suggested.
A version of this article first appeared on Medscape.com.
The question drew spirited debate when urologist Ashley Winter, MD, made a simple, straightforward request on Twitter: “If you are a doctor & you come to an appointment please tell me you are a doctor, not because I will treat you differently but because it’s easier to speak in jargon.”
She later added, “This doesn’t’ mean I would be less patient-focused or emotional with a physician or other [healthcare worker]. Just means that, instead of saying ‘you will have a catheter draining your urine to a bag,’ I can say, ‘you will have a Foley.’ ”
The Tweet followed an encounter with a patient who told Dr. Winter that he was a doctor only after she had gone to some length explaining a surgical procedure in lay terms.
“I explained the surgery, obviously assuming he was an intelligent adult, but using fully layman’s terms,” she said in an interview. The patient then told her that he was a doctor. “I guess I felt this embarrassment — I wouldn’t have treated him differently, but I just could have discussed the procedure with him in more professional terms.”
“To some extent, it was my own fault,” she commented in an interview. “I didn’t take the time to ask [about his work] at the beginning of the consultation, but that’s a fine line, also,” added Dr. Winter, a urologist and sexual medicine physician in Portland, Ore.
“You know that patient is there because they want care from you and it’s not necessarily always at the forefront of importance to be asking them what they do for their work, but alternatively, if you don’t ask then you put them in this position where they have to find a way to go ahead and tell you.”
Several people chimed in on the thread to voice their thoughts on the matter. Some commiserated with Dr. Winter’s experience:
“I took care of a retired cardiologist in the hospital as a second-year resident and honest to god he let me ramble on ‘explaining’ his echo result and never told me. I found out a couple days later and wanted to die,” posted @MaddyAndrewsMD.
Another recalled a similarly embarrassing experience when she “went on and on” discussing headaches with a patient whose husband “was in the corner smirking.”
“They told my attending later [that the] husband was a retired FM doc who practiced medicine longer than I’ve been alive. I wanted to die,” posted @JSinghDO.
Many on the thread, though, were doctors and other healthcare professionals speaking as patients. Some said they didn’t want to disclose their status as a healthcare provider because they felt it affected the care they received.
For example, @drhelenrainford commented: “In my experience my care is less ‘caring’ when they know I am a [doctor]. I get spoken to like they are discussing a patient with me — no empathy just facts and difficult results just blurted out without consideration. Awful awful time as an inpatient …but that’s another story!”
@Dr_B_Ring said: “Nope – You and I speak different jargon – I would want you to speak to me like a human that doesn’t know your jargon. My ego would get in the way of asking about the acronyms I don’t know if you knew I was a fellow physician.”
Conversely, @lozzlemcfozzle said: “Honestly I prefer not to tell my Doctors — I’ve found people skip explanations assuming I ‘know,’ or seem a little nervous when I tell them!”
Others said they felt uncomfortable — pretentious, even — in announcing their status, or worried that they might come across as expecting special care.
“It’s such a tough needle to thread. Want to tell people early but not come off as demanding special treatment, but don’t want to wait too long and it seems like a trap,” said @MDaware.
Twitter user @MsBabyCatcher wrote: “I have a hard time doing this because I don’t want people to think I’m being pretentious or going to micromanage/dictate care.”
Replying to @MsBabyCatcher, @RedStethoscope wrote: “I used to think this too until I got [very poor] care a few times, and was advised by other doctor moms to ‘play the doctor card.’ I have gotten better/more compassionate care by making sure it’s clear that I’m a physician (which is junk, but here we are).”
Several of those responding used the words “tricky” and “awkward,” suggesting a common theme for doctors presenting as patients.
“I struggle with this. My 5-year-old broke her arm this weekend, we spent hours in the ED, of my own hospital, I never mentioned it because I didn’t want to get preferential care. But as they were explaining her type of fracture, it felt awkward and inefficient,” said @lindsay_petty.
To avoid the awkwardness, a number of respondents said they purposefully use medical jargon to open up a conversation rather than just offering up the information that they are a doctor.
Still others offered suggestions on how to broach the subject more directly when presenting as a patient:
‘”Just FYI I’m a X doc but I’m here because I really want your help and advice!” That’s what I usually do,” wrote @drcakefm.
@BeeSting14618 Tweeted: “I usually say ‘I know some of this but I’m here because I want YOUR guidance. Also I may ask dumb questions, and I’ll tell you if a question is asking your opinion or making a request.’”
A few others injected a bit of humor: “I just do the 14-part handshake that only doctors know. Is that not customary?” quipped @Branmiz25.
“Ah yes, that transmits the entire [history of present illness],” replied Dr. Winter.
Jokes aside, the topic is obviously one that touched on a shared experience among healthcare providers, Dr. Winter commented. The Twitter thread she started just “blew up.”
That’s typically a sign that the Tweet is relatable for a lot of people, she said.
“It’s definitely something that all of us as care providers and as patients understand. It’s a funny, awkward thing that can really change an interaction, so we probably all feel pretty strongly about our experiences related to that,” she added.
The debate begs the question: Is there a duty or ethical reason to disclose?
“I definitely think it is very reasonable to disclose that one is a medical professional to another doctor,” medical ethicist Charlotte Blease, PhD, said in an interview. “There are good reasons to believe doing so might make a difference to the quality of communication and transparency.”
If the ability to use medical terminology or jargon more freely improves patient understanding, autonomy, and shared decision-making, then it may be of benefit, said Dr. Blease, a Keane OpenNotes Scholar at Beth Israel Deaconess Medical Center in Boston.
“Since doctors should strive to communicate effectively with every patient and to respect their unique needs and level of understanding, then I see no reason to deny that one is a medic,” she added.”
Knowing how to share the information is another story.
“This is something that affects all of us as physicians — we’re going to be patients at some point, right?” Dr. Winter commented. “But I don’t think how to disclose that is something that was ever brought up in my medical training.”
“Maybe there should just be a discussion of this one day when people are in medical school — maybe in a professionalism course — to broach this topic or look at if there’s any literature on outcomes related to disclosure of status or what are best practices,” she suggested.
A version of this article first appeared on Medscape.com.
Fast foods contain endocrine-disrupting chemicals
, such as chicken nuggets, hamburgers, and cheese pizza, new research suggests.
The first-of-its-kind study, which measured concentrations of chemicals such as phthalates in foods and gloves from U.S. fast food chains, is also the first to detect the plasticizer DEHT in fast foods.
“We knew from prior research that fast food consumption is linked to higher levels of phthalates in people’s bodies, but our study was novel because we actually collected these food items from fast food places and measured them,” said study author Lariah Edwards, PhD, a postdoctoral research scientist at the Milken Institute School of Public Health, George Washington University, Washington.
“Our research added an additional piece of information to the puzzle,” Dr. Edwards said in an interview.
A class of chemicals used in food packaging and food processing equipment, phthalates such as DEHP and DnBP, can leach out of these items and interfere with hormone production, Dr. Edwards said. They are linked with a wide variety of reproductive, developmental, brain, and immune effects, as well as with childhood obesity, asthma, cancer, and cardiovascular problems.
Meanwhile, nonphthalate or replacement plasticizers have been used in place of phthalates, some of which have been banned in certain products. But these plasticizers aren’t well studied, Dr. Edwards said, making the detection of DEHT in fast foods particularly concerning.
“There’s very limited research out there to understand the human health effects” of DEHT in food, she said, “so we’re being exposed before we understand what it’s doing to our health. It’s almost like we’re setting ourselves up for a big experiment.”
The study was recently published in the Journal of Exposure Science & Environmental Epidemiology .
Fast foods containing meat had highest concentrations of chemicals
Dr. Edwards and colleagues obtained 64 food samples, including hamburgers, fries, chicken nuggets, chicken burritos, and cheese pizza, as well as three pairs of unused gloves from six different fast food restaurants in San Antonio.
Using gas chromatography–mass spectrometry, they analyzed the samples for 11 chemicals, including eight phthalates and three replacement plasticizers.
The researchers detected 10 of the 11 chemicals in fast food samples: 81% of foods contained DnBP (di-n-butyl phthalate), and 70% contained DEHP (di(2-ethylhexyl phthalate)). Meanwhile 86% of samples contained replacement plasticizer DEHT (di(2-ethylhexyl terephthalate)).
Overall, fast food samples containing meat — including chicken nuggets, chicken burritos, and hamburgers — contained higher levels of these chemicals, Dr. Edwards noted.
“We know fast food is not the most nutritious, and now we’re seeing these chemicals in it we shouldn’t be exposed to,” she said.
The results also create implications for health equity, Dr. Edwards said, as Black people in the United States report eating more fast foods than other racial and ethnic groups for many reasons, such as longstanding residential segregation.
Many advocacy groups are pushing for stronger regulations on phthalates in foods, she said, and the study can be used to fuel those efforts.
“We’re hoping our findings help people understand what they’re eating and what’s in food,” Dr. Edwards said. “If they want to reduce exposure to phthalates in fast food, they can choose foods without meat in them. But not everyone has the option of reducing fast food consumption — personal choice is important, but policy is what’s going to protect us.”
Dr. Edwards noted that the research was limited by small sample sizes gathered in one U.S. city. Limitations in extraction methods also meant the researchers were able to detect chemicals in gloves only at high concentrations.
“That being said, I do think our results are fairly generalizable,” she added, “because the way fast foods are prepared at these restaurants is fairly consistent.”
The study was funded by the Passport Foundation, Forsythia Foundation, and Marisla Foundation. Dr. Edwards has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, such as chicken nuggets, hamburgers, and cheese pizza, new research suggests.
The first-of-its-kind study, which measured concentrations of chemicals such as phthalates in foods and gloves from U.S. fast food chains, is also the first to detect the plasticizer DEHT in fast foods.
“We knew from prior research that fast food consumption is linked to higher levels of phthalates in people’s bodies, but our study was novel because we actually collected these food items from fast food places and measured them,” said study author Lariah Edwards, PhD, a postdoctoral research scientist at the Milken Institute School of Public Health, George Washington University, Washington.
“Our research added an additional piece of information to the puzzle,” Dr. Edwards said in an interview.
A class of chemicals used in food packaging and food processing equipment, phthalates such as DEHP and DnBP, can leach out of these items and interfere with hormone production, Dr. Edwards said. They are linked with a wide variety of reproductive, developmental, brain, and immune effects, as well as with childhood obesity, asthma, cancer, and cardiovascular problems.
Meanwhile, nonphthalate or replacement plasticizers have been used in place of phthalates, some of which have been banned in certain products. But these plasticizers aren’t well studied, Dr. Edwards said, making the detection of DEHT in fast foods particularly concerning.
“There’s very limited research out there to understand the human health effects” of DEHT in food, she said, “so we’re being exposed before we understand what it’s doing to our health. It’s almost like we’re setting ourselves up for a big experiment.”
The study was recently published in the Journal of Exposure Science & Environmental Epidemiology .
Fast foods containing meat had highest concentrations of chemicals
Dr. Edwards and colleagues obtained 64 food samples, including hamburgers, fries, chicken nuggets, chicken burritos, and cheese pizza, as well as three pairs of unused gloves from six different fast food restaurants in San Antonio.
Using gas chromatography–mass spectrometry, they analyzed the samples for 11 chemicals, including eight phthalates and three replacement plasticizers.
The researchers detected 10 of the 11 chemicals in fast food samples: 81% of foods contained DnBP (di-n-butyl phthalate), and 70% contained DEHP (di(2-ethylhexyl phthalate)). Meanwhile 86% of samples contained replacement plasticizer DEHT (di(2-ethylhexyl terephthalate)).
Overall, fast food samples containing meat — including chicken nuggets, chicken burritos, and hamburgers — contained higher levels of these chemicals, Dr. Edwards noted.
“We know fast food is not the most nutritious, and now we’re seeing these chemicals in it we shouldn’t be exposed to,” she said.
The results also create implications for health equity, Dr. Edwards said, as Black people in the United States report eating more fast foods than other racial and ethnic groups for many reasons, such as longstanding residential segregation.
Many advocacy groups are pushing for stronger regulations on phthalates in foods, she said, and the study can be used to fuel those efforts.
“We’re hoping our findings help people understand what they’re eating and what’s in food,” Dr. Edwards said. “If they want to reduce exposure to phthalates in fast food, they can choose foods without meat in them. But not everyone has the option of reducing fast food consumption — personal choice is important, but policy is what’s going to protect us.”
Dr. Edwards noted that the research was limited by small sample sizes gathered in one U.S. city. Limitations in extraction methods also meant the researchers were able to detect chemicals in gloves only at high concentrations.
“That being said, I do think our results are fairly generalizable,” she added, “because the way fast foods are prepared at these restaurants is fairly consistent.”
The study was funded by the Passport Foundation, Forsythia Foundation, and Marisla Foundation. Dr. Edwards has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, such as chicken nuggets, hamburgers, and cheese pizza, new research suggests.
The first-of-its-kind study, which measured concentrations of chemicals such as phthalates in foods and gloves from U.S. fast food chains, is also the first to detect the plasticizer DEHT in fast foods.
“We knew from prior research that fast food consumption is linked to higher levels of phthalates in people’s bodies, but our study was novel because we actually collected these food items from fast food places and measured them,” said study author Lariah Edwards, PhD, a postdoctoral research scientist at the Milken Institute School of Public Health, George Washington University, Washington.
“Our research added an additional piece of information to the puzzle,” Dr. Edwards said in an interview.
A class of chemicals used in food packaging and food processing equipment, phthalates such as DEHP and DnBP, can leach out of these items and interfere with hormone production, Dr. Edwards said. They are linked with a wide variety of reproductive, developmental, brain, and immune effects, as well as with childhood obesity, asthma, cancer, and cardiovascular problems.
Meanwhile, nonphthalate or replacement plasticizers have been used in place of phthalates, some of which have been banned in certain products. But these plasticizers aren’t well studied, Dr. Edwards said, making the detection of DEHT in fast foods particularly concerning.
“There’s very limited research out there to understand the human health effects” of DEHT in food, she said, “so we’re being exposed before we understand what it’s doing to our health. It’s almost like we’re setting ourselves up for a big experiment.”
The study was recently published in the Journal of Exposure Science & Environmental Epidemiology .
Fast foods containing meat had highest concentrations of chemicals
Dr. Edwards and colleagues obtained 64 food samples, including hamburgers, fries, chicken nuggets, chicken burritos, and cheese pizza, as well as three pairs of unused gloves from six different fast food restaurants in San Antonio.
Using gas chromatography–mass spectrometry, they analyzed the samples for 11 chemicals, including eight phthalates and three replacement plasticizers.
The researchers detected 10 of the 11 chemicals in fast food samples: 81% of foods contained DnBP (di-n-butyl phthalate), and 70% contained DEHP (di(2-ethylhexyl phthalate)). Meanwhile 86% of samples contained replacement plasticizer DEHT (di(2-ethylhexyl terephthalate)).
Overall, fast food samples containing meat — including chicken nuggets, chicken burritos, and hamburgers — contained higher levels of these chemicals, Dr. Edwards noted.
“We know fast food is not the most nutritious, and now we’re seeing these chemicals in it we shouldn’t be exposed to,” she said.
The results also create implications for health equity, Dr. Edwards said, as Black people in the United States report eating more fast foods than other racial and ethnic groups for many reasons, such as longstanding residential segregation.
Many advocacy groups are pushing for stronger regulations on phthalates in foods, she said, and the study can be used to fuel those efforts.
“We’re hoping our findings help people understand what they’re eating and what’s in food,” Dr. Edwards said. “If they want to reduce exposure to phthalates in fast food, they can choose foods without meat in them. But not everyone has the option of reducing fast food consumption — personal choice is important, but policy is what’s going to protect us.”
Dr. Edwards noted that the research was limited by small sample sizes gathered in one U.S. city. Limitations in extraction methods also meant the researchers were able to detect chemicals in gloves only at high concentrations.
“That being said, I do think our results are fairly generalizable,” she added, “because the way fast foods are prepared at these restaurants is fairly consistent.”
The study was funded by the Passport Foundation, Forsythia Foundation, and Marisla Foundation. Dr. Edwards has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JOURNAL OF EXPOSURE SCIENCE & ENVIRONMENTAL EPIDEMIOLOGY
Electronic ‘nose’ sniffs out sarcoidosis
An electronic nose (eNose) that measures volatile organic compounds (VOCs) emitted from the lungs successfully distinguished sarcoidosis from interstitial lung disease (ILD) and healthy controls, according to a report in the journal CHEST.
The approach has the potential to generate clinical data that can’t be achieved through other noninvasive means, such as the serum biomarker soluble interleukin-2 receptor (sIL-2R). sIL-2R is often used to track disease activity, but it isn’t specific for diagnosing sarcoidosis, and it isn’t available worldwide.
Sarcoidosis is a granulomatous inflammatory disease with no known cause and can affect most organs, but an estimated 89%-99% of cases affect the lungs. There is no simple noninvasive diagnostic test, leaving physicians to rely on clinical features, biopsies to obtain tissue pathology, and the ruling out of other granulomatous diagnoses.
The challenge is more difficult because sarcoidosis is a heterogeneous disease, with great variation in the number of organs affected, severity, rate of progression, and response to therapy.
Previous researchers have used VOCs in an attempt to diagnose diseases, since the compounds reflect pathophysiological processes. Gas chromatography/mass spectrometry (GCMS) is one method to identify the individual VOCs, but the process is time consuming and complex. Some nevertheless showed potential in sarcoidosis, but failed to reproduce their performance in validation cohorts.
In the new study, a cross-sectional analysis showed that exhaled breath analysis using an eNose had excellent sensitivity and specificity for distinguishing sarcoidosis from ILD and healthy controls, and identified sarcoidosis regardless of pulmonary involvement, pulmonary fibrosis, multiple organ involvement, immunosuppressive treatment, or whether or not pathology supported the diagnosis.
The eNose technology produces a “breath-print” after combining information from a broad range of VOCs. The information originates from an array of metal-oxide semiconductor sensors with partial specificity that artificial intelligence processes to discern patterns. Overall, the system functions similarly to the mammalian olfactory system. The artificial intelligence instead views it as a “breath-print” that it can compare against previously learned patterns.
“It is a quite easy, simple, and quick procedure, which is noninvasive. We can collect a lot of data from the VOCs in the exhaled breath because there are several sensors that cross-react. We can create breath profiles and group patients to see if profiles differ. Ultimately, we can use the profiles to diagnose or detect disease in the earlier stage and more accurately,” said Iris van der Sar, MD. Dr. van der Sar is the lead author on the study and a PhD candidate at Erasmus Medical Center in Rotterdam.
The study requires further prospective validation, but the technology could have important clinical benefits, said senior author and principal investigator Marlies Wijsenbeek, MD, PhD, pulmonologist and head of the Interstitial Lung Disease Center at Erasmus Medical Center. “If we in future can avoid a biopsy, that would be most attractive,” said Dr. Wijsenbeek.
“We hope to come to a point-of-care device that can be used to facilitate early diagnosis at low burden for the patient and health care system,” said Karen Moor, MD, PhD, and post-doc on this project. The researchers also hope to determine if the eNose can help evaluate a patient’s response to therapy.
Studies of eNose technology in other chronic diseases have shown promising results, but not all results have been validated yet in independent or external cohorts.
The current study included 569 outpatients, 252 with sarcoidosis and 317 with ILD, along with 48 healthy controls. The researchers constructed a training set using 168 patients with sarcoidosis and 32 healthy controls, and a validation set using 84 patients with sarcoidosis and 16 healthy controls. The eNose differentiated between patients and controls in both groups, with an area under the curve of 1.00 for each regardless of pulmonary involvement or treatment.
It also distinguished those with sarcoidosis and pulmonary involvement from those with ILD, with an AUC of 0.90 (95% confidence interval, 0.87-0.94) in the training set, and an AUC of 0.87 (95% CI, 0.82-0.93) in the validation set.
It differentiated between pulmonary sarcoidosis and hypersensitivity pneumonitis in the training set (AUC 0.95; 95% CI, 0.90-0.99) and the validation set (AUC, 0.88; 95% CI, 0.75-1.00).
The study received no funding. Dr. Wijsenbeek, Dr. van der Sar, and Dr. Moor have no relevant financial disclosures.
An electronic nose (eNose) that measures volatile organic compounds (VOCs) emitted from the lungs successfully distinguished sarcoidosis from interstitial lung disease (ILD) and healthy controls, according to a report in the journal CHEST.
The approach has the potential to generate clinical data that can’t be achieved through other noninvasive means, such as the serum biomarker soluble interleukin-2 receptor (sIL-2R). sIL-2R is often used to track disease activity, but it isn’t specific for diagnosing sarcoidosis, and it isn’t available worldwide.
Sarcoidosis is a granulomatous inflammatory disease with no known cause and can affect most organs, but an estimated 89%-99% of cases affect the lungs. There is no simple noninvasive diagnostic test, leaving physicians to rely on clinical features, biopsies to obtain tissue pathology, and the ruling out of other granulomatous diagnoses.
The challenge is more difficult because sarcoidosis is a heterogeneous disease, with great variation in the number of organs affected, severity, rate of progression, and response to therapy.
Previous researchers have used VOCs in an attempt to diagnose diseases, since the compounds reflect pathophysiological processes. Gas chromatography/mass spectrometry (GCMS) is one method to identify the individual VOCs, but the process is time consuming and complex. Some nevertheless showed potential in sarcoidosis, but failed to reproduce their performance in validation cohorts.
In the new study, a cross-sectional analysis showed that exhaled breath analysis using an eNose had excellent sensitivity and specificity for distinguishing sarcoidosis from ILD and healthy controls, and identified sarcoidosis regardless of pulmonary involvement, pulmonary fibrosis, multiple organ involvement, immunosuppressive treatment, or whether or not pathology supported the diagnosis.
The eNose technology produces a “breath-print” after combining information from a broad range of VOCs. The information originates from an array of metal-oxide semiconductor sensors with partial specificity that artificial intelligence processes to discern patterns. Overall, the system functions similarly to the mammalian olfactory system. The artificial intelligence instead views it as a “breath-print” that it can compare against previously learned patterns.
“It is a quite easy, simple, and quick procedure, which is noninvasive. We can collect a lot of data from the VOCs in the exhaled breath because there are several sensors that cross-react. We can create breath profiles and group patients to see if profiles differ. Ultimately, we can use the profiles to diagnose or detect disease in the earlier stage and more accurately,” said Iris van der Sar, MD. Dr. van der Sar is the lead author on the study and a PhD candidate at Erasmus Medical Center in Rotterdam.
The study requires further prospective validation, but the technology could have important clinical benefits, said senior author and principal investigator Marlies Wijsenbeek, MD, PhD, pulmonologist and head of the Interstitial Lung Disease Center at Erasmus Medical Center. “If we in future can avoid a biopsy, that would be most attractive,” said Dr. Wijsenbeek.
“We hope to come to a point-of-care device that can be used to facilitate early diagnosis at low burden for the patient and health care system,” said Karen Moor, MD, PhD, and post-doc on this project. The researchers also hope to determine if the eNose can help evaluate a patient’s response to therapy.
Studies of eNose technology in other chronic diseases have shown promising results, but not all results have been validated yet in independent or external cohorts.
The current study included 569 outpatients, 252 with sarcoidosis and 317 with ILD, along with 48 healthy controls. The researchers constructed a training set using 168 patients with sarcoidosis and 32 healthy controls, and a validation set using 84 patients with sarcoidosis and 16 healthy controls. The eNose differentiated between patients and controls in both groups, with an area under the curve of 1.00 for each regardless of pulmonary involvement or treatment.
It also distinguished those with sarcoidosis and pulmonary involvement from those with ILD, with an AUC of 0.90 (95% confidence interval, 0.87-0.94) in the training set, and an AUC of 0.87 (95% CI, 0.82-0.93) in the validation set.
It differentiated between pulmonary sarcoidosis and hypersensitivity pneumonitis in the training set (AUC 0.95; 95% CI, 0.90-0.99) and the validation set (AUC, 0.88; 95% CI, 0.75-1.00).
The study received no funding. Dr. Wijsenbeek, Dr. van der Sar, and Dr. Moor have no relevant financial disclosures.
An electronic nose (eNose) that measures volatile organic compounds (VOCs) emitted from the lungs successfully distinguished sarcoidosis from interstitial lung disease (ILD) and healthy controls, according to a report in the journal CHEST.
The approach has the potential to generate clinical data that can’t be achieved through other noninvasive means, such as the serum biomarker soluble interleukin-2 receptor (sIL-2R). sIL-2R is often used to track disease activity, but it isn’t specific for diagnosing sarcoidosis, and it isn’t available worldwide.
Sarcoidosis is a granulomatous inflammatory disease with no known cause and can affect most organs, but an estimated 89%-99% of cases affect the lungs. There is no simple noninvasive diagnostic test, leaving physicians to rely on clinical features, biopsies to obtain tissue pathology, and the ruling out of other granulomatous diagnoses.
The challenge is more difficult because sarcoidosis is a heterogeneous disease, with great variation in the number of organs affected, severity, rate of progression, and response to therapy.
Previous researchers have used VOCs in an attempt to diagnose diseases, since the compounds reflect pathophysiological processes. Gas chromatography/mass spectrometry (GCMS) is one method to identify the individual VOCs, but the process is time consuming and complex. Some nevertheless showed potential in sarcoidosis, but failed to reproduce their performance in validation cohorts.
In the new study, a cross-sectional analysis showed that exhaled breath analysis using an eNose had excellent sensitivity and specificity for distinguishing sarcoidosis from ILD and healthy controls, and identified sarcoidosis regardless of pulmonary involvement, pulmonary fibrosis, multiple organ involvement, immunosuppressive treatment, or whether or not pathology supported the diagnosis.
The eNose technology produces a “breath-print” after combining information from a broad range of VOCs. The information originates from an array of metal-oxide semiconductor sensors with partial specificity that artificial intelligence processes to discern patterns. Overall, the system functions similarly to the mammalian olfactory system. The artificial intelligence instead views it as a “breath-print” that it can compare against previously learned patterns.
“It is a quite easy, simple, and quick procedure, which is noninvasive. We can collect a lot of data from the VOCs in the exhaled breath because there are several sensors that cross-react. We can create breath profiles and group patients to see if profiles differ. Ultimately, we can use the profiles to diagnose or detect disease in the earlier stage and more accurately,” said Iris van der Sar, MD. Dr. van der Sar is the lead author on the study and a PhD candidate at Erasmus Medical Center in Rotterdam.
The study requires further prospective validation, but the technology could have important clinical benefits, said senior author and principal investigator Marlies Wijsenbeek, MD, PhD, pulmonologist and head of the Interstitial Lung Disease Center at Erasmus Medical Center. “If we in future can avoid a biopsy, that would be most attractive,” said Dr. Wijsenbeek.
“We hope to come to a point-of-care device that can be used to facilitate early diagnosis at low burden for the patient and health care system,” said Karen Moor, MD, PhD, and post-doc on this project. The researchers also hope to determine if the eNose can help evaluate a patient’s response to therapy.
Studies of eNose technology in other chronic diseases have shown promising results, but not all results have been validated yet in independent or external cohorts.
The current study included 569 outpatients, 252 with sarcoidosis and 317 with ILD, along with 48 healthy controls. The researchers constructed a training set using 168 patients with sarcoidosis and 32 healthy controls, and a validation set using 84 patients with sarcoidosis and 16 healthy controls. The eNose differentiated between patients and controls in both groups, with an area under the curve of 1.00 for each regardless of pulmonary involvement or treatment.
It also distinguished those with sarcoidosis and pulmonary involvement from those with ILD, with an AUC of 0.90 (95% confidence interval, 0.87-0.94) in the training set, and an AUC of 0.87 (95% CI, 0.82-0.93) in the validation set.
It differentiated between pulmonary sarcoidosis and hypersensitivity pneumonitis in the training set (AUC 0.95; 95% CI, 0.90-0.99) and the validation set (AUC, 0.88; 95% CI, 0.75-1.00).
The study received no funding. Dr. Wijsenbeek, Dr. van der Sar, and Dr. Moor have no relevant financial disclosures.
FROM CHEST