User login
Minimally invasive cesarean: Improving an innovative technique
- A simplified abdominal incision makes the traditional extensive dissection associated with the Pfannenstiel incision unnecessary.
- A soft, self-retaining abdominal retractor offers increased exposure, atraumatic retraction, incision protection, and adjustable height while facilitating delivery of the fetal head by creating a rigid border around the abdominal incision.
- Bladder-flap omission has been associated with reduced operative time and incision-delivery interval, decreased blood loss, and less need for postoperative analgesics.
Is the extensive dissection of the Pfannenstiel incision necessary in cesarean delivery? Is bladder dissection essential? Must the visceral and parietal peritoneum be closed?
The success of our minimally invasive cesarean technique suggests the answer is “no.”
The approach described here features a short operative time; minimal instrumentation; reduced surgical dissection; decreased postoperative pain; and reduced risk of blood loss, infection, and wound complications. It is easily learned and cost-effective, with a brief postoperative recovery period.
Among the updates made from the technique’s initial publication in the mid-1990s1,2:
- addition of routine perioperative oxygen (80%), to reduce the risk of surgical wound infection (see “Perioperative considerations”)
- regular use of forced warm air covers applied to the anterior skin surface, to help patients maintain normothermia
- addition of a soft, self-retaining abdominal retractor—which creates an atraumatic circle of exposure up to a calculated 177 cm2 for a 15-cm incision (versus 113 cm2 calculated for traditional retraction)
- vertical, rather than lateral, digital extension of the initial transverse uterine incision
- identification of a subgroup in whom peritoneal closure is strongly recommended
- new data on the procedure’s effectiveness.3
Routinely use prophylactic antibiotics. Several recent studies have concluded that perioperative antibiotics reduce the incidence of endometritis and wound infection following elective and nonelective cesarean section.37 Administer ampicillin or a first-generation cephalosporin at umbilical cord clamping. For patients allergic to penicillin and cephalosporins, choose an alternative, such as clindamycin.
Give antacids 30 minutes before anesthesia (general or regional) to prevent pneumonitis from inhalation of gastric contents.
Clip pubic hair, rather than shave, to reduce the risk of wound infection.
Insert a Foley catheter, empty the bladder, and keep the catheter in place.
Position the patient in a 10°left lateral tilt, to avoid hypotension associated with aortocaval occlusion.
Routinely administer supplemental perioperative oxygen (80%), with either general or regional anesthesia; this activates alveolar immune defenses and halves the risk of surgical wound infections.
Neutrophil oxidative killing and phagocytosis—the most important defenses against surgical pathogens—depend on the partial pressure of oxygen in contaminated tissue. Giving supplemental oxygen during and for the first 2 hours after the procedure (by mask) is a practical, inexpensive way to reduce the incidence of surgical wound infection.38,39
Using 80% oxygen during and, for a short period, after surgery does not cause pulmonary toxicity such as atelectasis or impaired pulmonary function.40
Ensure normal body heat during and after cesarean to reduce the risk of postoperative surgical infection.40,41 Forced warm air covers applied to the anterior skin surface are the most effective way for warming surgical patients.9 IV fluid warming, though appropriate when large volumes are to be administered, is unnecessary for smaller operations.41
Modified abdominal incision reduces dissection
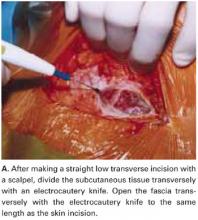
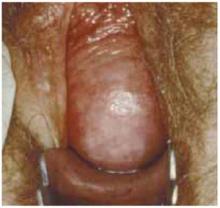
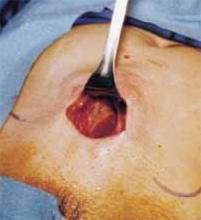
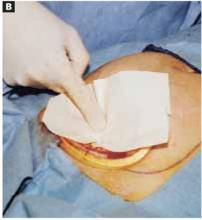
Make a straight low transverse incision with a scalpel, at a point approximately 3 to 4 cm above the symphysis pubis (FIGURE 1A).
The length of the incision is individualized (13 to 15 cm), though difficult fetal extraction is more likely if the abdominal incision is less than 15 cm.4,5
Divide the subcutaneous tissue transversely with an electrocautery knife. In a cutting and coagulation blend mode, the knife divides the fat while achieving hemostasis. To improve hemostasis, coagulate the blood vessels that cross the subcutaneous fat layer in a brushing manner before dividing them. To prevent unnecessary dead space, avoid filleting the fat and separating adherent subcutaneous fat from the anterior rectus fascia beyond what is needed to expose the fascia.
Open the fascia transversely with the electrocautery knife to the same length as the skin incision. Coagulate the blood vessels that cross the fascia before dividing them. Identify the median raphe by pulling up the superior edge of the abdominal incision.
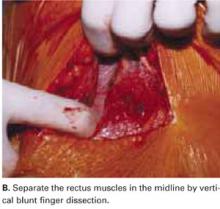
Separate the rectus muscles in the midline by vertical blunt finger dissection (FIGURE 1B). If digital dissection is inefficient due to a dense, thick, or scarred median raphe, use an electrocautery knife, a scalpel, or scissors.
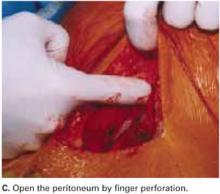
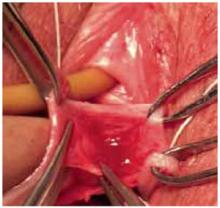
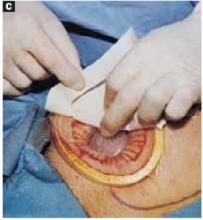
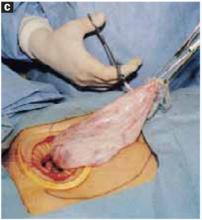
Open the peritoneum. This is facilitated by upward traction and elevation of the superior edge of the abdominal incision that lifts the peritoneum, allowing easy digital perforation using the index or middle finger (FIGURE 1C).
If this maneuver is not feasible, open the peritoneum in the traditional fashion.
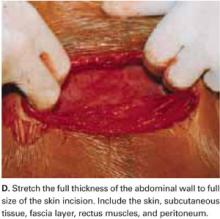
Stretch the full thickness of the abdominal wall to full size of the skin incision, using 1 or 2 fingers of each hand (FIGURE 1D). Incorporate the skin, subcutaneous tissue, fascia layer, rectus muscles, and peritoneum. An assistant’s hands may be required. When needed, extend the peritoneal opening transversely on either side, to the midline and away from the bladder.
Digitally stretching the full-thickness abdominal incision is easily achieved due to the mechanical stretching of the anterior abdominal wall, edema, and increased vascularization that occur during pregnancy.
FIGURE 1A Create a modified abdominal incision
FIGURE 1B Create a modified abdominal incision
FIGURE 1C Create a modified abdominal incision
FIGURE 1D Create a modified abdominal incision
Retractor facilitates exposure
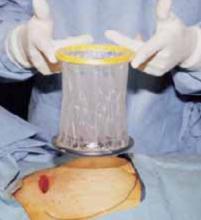
Our positive experience with the soft, self-retaining abdominal retractor for minilaparotomy and laparotomy6-8 compelled us to incorporate its use for cesarean 3 years ago.
The device consists of a flexible plastic inner ring and a firmer outer ring connected by a soft plastic sleeve. We use the large size of either of the 2 models currently available: the Mobius (Apple Medical Corporation, Marlboro, Miss) and the Protractor (Weck Closure Systems, Research Triangle Park, NC).
Introduce a hand through the laparotomy incision and evaluate the pelvis to ensure that no significant adhesions are present that may interfere with swift placement of the inner ring. If significant adhesions are found, use traditional metal retractors instead.
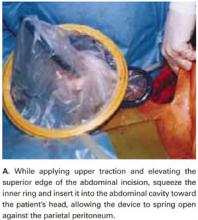
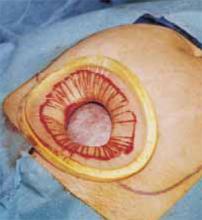
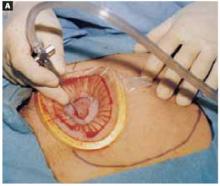
Squeeze the inner ring and insert it into the abdominal cavity toward the patient’s head, allowing the device to spring open against the parietal peritoneum (FIGURE 2A). Apply upper traction and elevate the superior edge of the abdominal incision to facilitate placement. Perform a digital check to ensure no tissue is trapped between the inner ring and the abdominal wall.
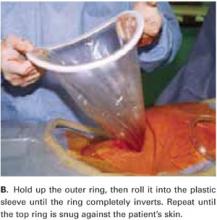
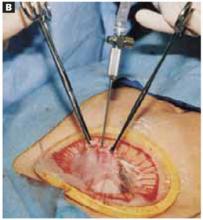
Hold up the outer ring, then roll the plastic sleeve until the ring completely inverts (FIGURE 2B). Repeat the process until the top ring is snug against the patient’s skin.
Advantages of the soft, self-retaining abdominal retractor include atraumatic retraction; incision protection; and adjustable height, making it ideal for obese patients.
FIGURE 2A Place the abdominal retractor
FIGURE 2B Place the abdominal retractor
Transverse hysterotomy in lower uterine segment
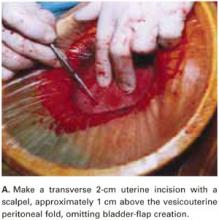
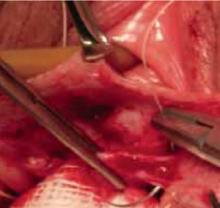
Make a transverse 2-cm uterine incision with a scalpel, approximately 1 cm above the vesicouterine peritoneal fold (identify this using gentle digital pressure to elevate the uterus) (FIGURE 3A).
Traditional bladder dissection is eliminated. We have found that when an adequate transverse hysterotomy is performed, a bladder flap is not required.1-3 In a recent randomized trial, Hohlagschwandtner et al9 confirmed our findings that bladder-flap omission was associated with reduced operative time and incision-delivery interval, decreased blood loss, and less need for postoperative analgesics.
An additional advantage of this omission: We can avoid making the uterine incision too low—especially when the cervix is fully dilated. Further, making the hysterotomy (uterine serosa and myometrium together) slightly above the vesicouterine peritoneal fold without bladder flap dissection frees the loose connective tissue between the uterus and the urinary bladder, allowing the spontaneous descent of the bladder.
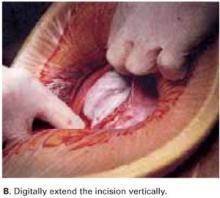
Digitally extend the transverse uterine incision. We extend the initial incision not laterally but, instead, vertically (FIGURE 3B). The vertical digital traction on the initial transverse uterine incision creates a transverse dissociation of the horizontal myometrium fibers; this results in a transverse extension of the original incision.
This modification prevents unintended and uncontrolled lateral extensions of the incision that may lacerate the uterine vessels.10 It also prevents the uterine incision from becoming an inverted “U” and the undesirable accumulation of myometrium fibers at the ends of the incision. Such incisions usually do not reapproximate well during hysterotomy closure and may lead to sacculation-type defects.11
FIGURE 3A Use a standard hysterotomy in the lower uterine segment
FIGURE 3B Use a standard hysterotomy in the lower uterine segment
Delivering the fetus
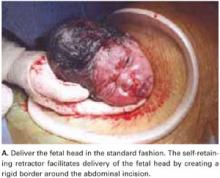
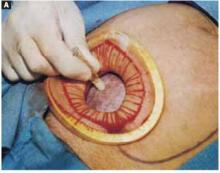
For vertex presentations, place your hand into the uterine cavity between the lower edge of the hysterotomy and fetal head. While applying transabdominal fundal pressure, lift the head with your fingers and deliver it through the incision (FIGURE 4A).
The self-retaining retractor facilitates delivery of the fetal head by creating a rigid border around the abdominal incision. The back of the surgeon’s hand that is in the uterine cavity achieves better leverage, as it now rests on a rigid plane on the inferior part of the incision rather than on the back hand and the softer and pliable wound edge of the standard abdominal incision.
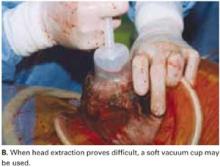
Vacuum-assistance. When delivery of the fetal head proves difficult, we use the soft vacuum cup, which avoids unnecessary intrauterine manipulation that may result in fetal or maternal trauma (FIGURE 4B).12
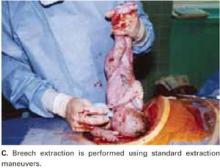
Breech extraction or transverse lie delivery is performed using standard extraction maneuvers (FIGURE 4C).13 We have found no need to perform T or J vertical extensions of the low transverse uterine incision, even in cases of a poorly developed lower uterine segment, abnormal presentation, or prematurity.
FIGURE 4A Deliver the fetal head
FIGURE 4B Deliver the fetal head
FIGURE 4C Deliver the fetal head
Uterine incision: 1-layer, in situ close
Remove the placenta only after it separates spontaneously—this greatly reduces blood loss compared with immediate manual placental delivery or umbilical-cord traction. Manually deliver the placenta only if it has not separated spontaneously after 5 minutes. Avoid routine manual clean-out of the uterine cavity if the placenta is completely delivered.1-3,14
Dilate the cervix to facilitate lochial discharge, when necessary.
Decrease uterine bleeding by massaging the uterus and administering diluted oxytocin.
Close the uterine incision in situ. Disadvantages of routine uterine exteriorization include discomfort and vomiting resulting from traction, exposure of the fallopian tubes to unnecessary trauma, increased risk of infection, possible rupture of the uteroovarian veins, and pulmonary embolism.
Exteriorization may also increase the risk of adhesion formation by exposing the uterine serosa to the abrasive effects of drying or microscopic abrasion if sponges are used to hold the uterus in place.1,14-17
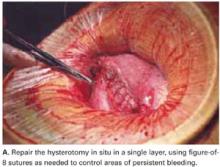
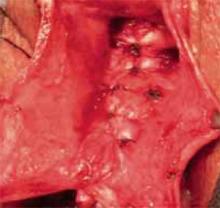
Use a single-layer closure with a running suture of polyglycolic acid to close the uterine incision (FIGURE 5A). If needed, use atraumatic clamps to grasp the edges of the hysterotomy to facilitate visualization and closure.
Begin suturing at a point just beyond one end of the incision, continuing to the opposite side. Be sure to penetrate the full thickness of the myometrium and avoid incorporating the decidua in the suture line. After the uterine closure, use individual figure-of-8 sutures to control areas of persistent bleeding.
Compared with 2-layer closures, single-layer closure of a low transverse incision is associated with reduced operating time, improved hemostasis, and less tissue disruption; introduces less foreign material into the surgical site; reduces the need for postoperative analgesia; and potentially reduces infectious morbidity. A trial of labor after a cesarean section with 1-layer closure appears safe.18-22
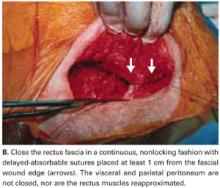
We do not close the vesicouterine fold and parietal peritoneum (FIGURE 5B) or reapproximate the rectus muscles. It has been shown that a new peritoneal layer is formed within days of the original incision closure.
Leaving the peritoneum open leads to no increased postoperative complications, nor obvious differences in wound healing, wound dehiscence, or incidence of postoperative adhesions. Compared to women without peritoneal closure, those with peritoneal closure require a longer operative time, greater amounts of post-operative narcotics for postoperative pain, a greater need for bowel stimulants, and a longer postoperative hospitalization.23
In 1995, we performed laparoscopy for gynecologic conditions in 23 patients and repeat cesarean in 10 patients who had undergone our technique (with visceral and parietal peritoneum left open).2 We found no adhesions and a normal peritoneal lining in all cases.
In 1 subgroup, peritoneal closure is strongly recommended: cases in which 1 or both rectus muscles have been transected to increase surgical exposure. Extremely thick fibromuscular adhesions between the low anterior surface of the uterus and the under-surface of the rectus musculature may develop if the parietal peritoneum is not closed.
FIGURE 5A Close the incisions
FIGURE 5B Close the incisions
Abdominal closure
Close the rectus fascia with a continuous nonlocking 0 suture after the rectus muscles fall into place. Place entry and exit sites at 1-cm intervals, 1.5 cm beyond the wound edges—this minimizes the risk of sutures pulling through fascia. Interrupted sutures offer no greater tensile strength than continuous suturing.24 Tight, continuous suturing can lead to tissue ischemia, which may weaken the sutured fascia, and thus should be avoided.
The subcutaneous layer is not closed separately. One exception is obese patients with at least 2 cm of thick subcutaneous tissue. Thickness of subcutaneous tissue appears to be a significant risk factor for wound infection after cesarean section.25
In patients with thick subcutaneous tissue, you can reduce tension on the skin edges and the risk of subcutaneous infection, seroma formation, and wound disruption by placing interrupted 3-0 absorbable synthetic sutures to eliminate dead space.26-28 We have found no need to place Penrose drains or closed drainage systems in the subcutaneous layer.29-31
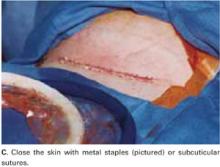
Close the skin using a subcuticular continuous absorbable 4-0 suture or, alternatively, metal staples, which are removed 4 days after surgery (FIGURE 5C).
FIGURE 5C Close the incisions
Prompt postoperative recovery
The patient may drink fluids in the recovery room, can resume a regular solid food diet within 4 hours after surgery, and may resume mobility as soon as anesthesia wears off.
Early breast-feeding is also allowed in the recovery room. In our experience, this has not been associated with complications, has been highly appreciated by the patients, and has led to earlier hospital discharge.
Control pain with meperidine (50–75 mg) or morphine (10 mg) parenterally every 3 to 4 hours. Other options include oxycodone and acetaminophen, oxycodone and aspirin, and ibuprofen.
Patients are usually discharged 48 to 72 hours following surgery.
Our experience: The numbers
Using this updated system, we have successfully completed 300 consecutive cesarean sections (210 primary, 90 repeat).
The average operating time was 19 minutes; average blood loss, 405 mL.
There was no postoperative febrile morbidity, wound infection, wound disruption, or wound hematoma.Only 3 patients developed superficial wound seromas, which were easily resolved. There were no intraoperative or postoperative complications.
We attribute the absence of wound infection to routine prophylactic antibiotics; supplemental perioperative oxygen; maintenance of normothermia; use of an electrocautery knife to create rapid hemostatic division of subcutaneous fat and anterior rectus fascia; a simplified abdominal wall opening; and placement of a self-retaining atraumatic retractor.10,32-36
Additional surgical procedures (tubal sterilization, myomectomy, appendectomy, and adhesiolysis) did not alter the recovery period, and all patients were able to get out of bed, shower, breast-feed, and care for their infants within 8 hours of surgery.
No infant complications related to the cesarean procedure occurred.
Patients were discharged from the hospital within 72 hours.
A prospective comparison study demonstrated that the original Pelosi-type of cesarean delivery was faster to perform, more cost-effective, and resulted in less maternal morbidity than the traditional cesarean technique.3
Dr. Pelosi II is a consultant for Apple Medical Corporation. Dr. Pelosi III reports no financial relationships relevant to this article.
1. Pelosi MA, Ortega I. Cesarean section: Pelosi simplified technique. Rev Chil Obstet Gynecol. 1994;59:372-377.
2. Pelosi MA, II, Pelosi MA, III. Simplified cesarean section. Contemp OB/GYN. 1995;40:89-100.
3. Wood RM, Simon H, Oz AU. Pelosi-type vs traditional cesarean delivery: a prospective comparison. J Reprod Med. 1999;44:788-795.
4. Finan MA, Mastrogiannis DS, Spellacy WN. The “Allis” test for easy cesarean delivery. Am J Obstet Gynecol. 1991;164:772-775.
5. Ayers IW, Morley GW. Surgical incision for cesarean section. Obstet Gynecol. 1987;70:706-712.
6. Pelosi MA, II, Pelosi MA, III. Self-retaining abdominal retractor for minilaparotomy. Obstet Gynecol. 2000;96:775-778.
7. Pelosi MA, II, Pelosi MA, III. Pelosi minilaparotomy hysterectomy: Effective alternative to laparoscopy and laparotomy. OBG Manag. April 2003;16-33.
8. Pelosi MA, II, Pelosi MA, III. A novel minilaparotomy approach for large ovarian cysts. OBG Manag. February 2004:;17-30.
9. Hohlagschwandtner M, Ruecklinger E, Husslein P, et al. Is the formation of a bladder flap at cesarean necessary? A randomized trial. Obstet Gynecol. 2001;98:1089-1092.
10. Ferrari A, Frigerio L, Origoni M, et al. Modified Stark procedure for cesarean section. J Pelv Surg. 1996;2:239-244.
11. Field CS. Surgical techniques for cesarean section. Obstet Gynecol Clin North Amer. 1988;15:657-672.
12. Pelosi MA, Apuzzio J. Use of the soft, silicone obstetric vacuum cup for delivery of the fetal head at cesarean section. J Reprod Med. 1984;29:289-292.
13. Pelosi MA, Apuzzio J, Fricchione D, et al. The intra-abdominal version technique for delivery of transverse lie by low segment cesarean section. Am J Obstet Gynecol. 1979;135:1009-1012.
14. Magann EF, Dodson MK, Albert JR, et al. Blood loss at the time of cesarean section by method of placental removal and exteriorization versus in situ repair of the uterine incision. Surg Gynecol Obstet. 1993;177:389-392.
15. Stock RJ, Shelton H. Fatal pulmonary embolism occurring 2 hours after exteriorization of the uterus for repair following cesareans section. Milit Med. 1985;150:549-550.
16. Hershey DW, Quilligan EJ. Extra-abdominal uterine exteriorization at cesarean section. Obstet Genecol. 1978;52:189-191.
17. Wahab MA, Karantzis P, Eccersley PS, et al. A randomized, controlled study of uterine exteriorization and repair at cesarean section. Br J Obstet Gynecol. 1999;106:913-916.
18. Hauth JC, Owen J, Davis RO. Transverse uterine incision closure: one versus two layers. Am J Obstet Gynecol. 1992;167:1108-1111.
19. Ohel G, Younis JS, Lang N, et al. Double-layer closure of uterine incision with visceral and parietal peritoneal closure: Are they obligatory steps of routine cesarean sections? J Matern Fetal Med. 1996;5:366-369.
20. Tucker JM, Hauth JC, Hodgkins P, et al. Trial of labor after a one- or two-layer closure of a low transverse uterine incision. Am J Obstet Gynecol. 1993;168:545-546.
21. Chapman SJ, Owen J, Hauth JC. One- versus two-layer closure of a low transverse cesarean: the next pregnancy. Obstet Gynecol. 1997;89:16-18.
22. Jelsema RD, Wittinger JA, Vander Kolk KJ. Continuous, nonlocking, single-layer repair of the low transverse uterine incision. J Reprod Med. 1993;38:393-396.
23. Tulandi T, Al-Jaroudi D. Nonclosure of peritoneum: a reappraisal. Am J Obstet Gynecol. 2003;189:609-612.
24. Fagniez PL, Hay JM, Lacaine F. Abdominal midline incision closure. Arch Surg. 1985;120:1351-1355.
25. Vermillion ST, Lamoutte C, Soper DE, et al. Wound infection after cesarean: effect of subcutaneous tissue thickness. Obstet Gynecol. 2000;95:923-926.
26. Naumann RW, Hauth JC, Owen J, et al. Subcutaneous tissue approximation in relation to wound disruption after cesarean delivery in obese women. Obstet Gynecol. 1995;85:412-416.
27. Cetin A, Cetin M. Superficial wound disruption after cesarean delivery: effect of the depth and closure of subcutaneous tissue. Int J Gynaecol Obstet. 1997;57:17-21.
28. Del Valle GO, Combs P, Qualls C, et al. Does closure of Camper fascia reduce the incidence of post-cesarean superficial wound disruption? Obstet Gynecol. 1992;80:1013-1016.
29. Loong RL, Rogers MS, Chang AM. A controlled trial of wound drainage at cesarean section. Aust NZ J Obstet Gynaecol. 1988;28:266-269.
30. Saunders NJ, Barclay C. Closed suction wound drainage and lower-segment cesarean section. Br J Obstet Gynaecol. 1988;95:1060-1062.
31. Magann EF, Chauhan SP, Rodts-Palenik S, Bufkin L, Martin JN, Jr, Morrison JC. Subcutaneous stitch closure versus subcutaneous drain to prevent wound disruption after cesarean delivery: a randomized clinical trial. Am J Obstet Gynecol. 2002;186:1119-1123.
32. Stark M, Finkel AR. Comparison between the Joel-Cohen and Pfannenstiel incisions in cesarean section. Eur J Obstet Gynecol Reprod Biol. 1994;53:121-122.
33. Joel-Cohen S. Abdominal and vaginal hysterectomy. New techniques based on time and motion studies. London, England: William Heinemann Medical Books; 1972.
34. Hemsell DL, Hemsell PG, Nobles B, et al. Abdominal wound problems after hysterectomy with electrocautery versus scalpel subcutaneous incision. Infect Dis Obstet Gynecol. 1993;1:27-30.
35. Johnson CD, Serpell JW. Wound infection after abdominal incision with scalpel or diathermy. Br J Surg. 1990;77:626-629.
36. Hussain SA, Hussain S. Incisions with knife or diathermy and postoperative pain. Br J Surg. 1988;75:1179-1182.
37. American College of Obstetricians and Gynecologist. ACOG Practice Bulletin. Clinical management guidelines for obstetricians-gynecologists. No. 43, May, 2003. Management of preterm labor. Obstet Gynecol. 2003;101:1039-1047.
38. Greif R, Akca O, Hurn E-P, et al. Supplemental perioperative oxygen to reduce the incidence of surgical wound infection. N Engl J Med. 2000;342:161-167.
39. Sorensen LT, Karlsmark T, Gottrup F. Abstinence from smoking reduces incisional wound infection: a randomized controlled trial. Ann Surg. 2003;238:1-5.
40. Sessler DI, Akca O. Preventing surgical site infections without drugs. Contemp OB/GYN. 2004;49:78-87.
41. Kurz A, Sessler DI, Lenhardt R. Perioperative normothermia to reduce the incidence of surgical wound infection and shorten hospitalization. N Engl J Med. 1996;334:1209-1215.
- A simplified abdominal incision makes the traditional extensive dissection associated with the Pfannenstiel incision unnecessary.
- A soft, self-retaining abdominal retractor offers increased exposure, atraumatic retraction, incision protection, and adjustable height while facilitating delivery of the fetal head by creating a rigid border around the abdominal incision.
- Bladder-flap omission has been associated with reduced operative time and incision-delivery interval, decreased blood loss, and less need for postoperative analgesics.
Is the extensive dissection of the Pfannenstiel incision necessary in cesarean delivery? Is bladder dissection essential? Must the visceral and parietal peritoneum be closed?
The success of our minimally invasive cesarean technique suggests the answer is “no.”
The approach described here features a short operative time; minimal instrumentation; reduced surgical dissection; decreased postoperative pain; and reduced risk of blood loss, infection, and wound complications. It is easily learned and cost-effective, with a brief postoperative recovery period.
Among the updates made from the technique’s initial publication in the mid-1990s1,2:
- addition of routine perioperative oxygen (80%), to reduce the risk of surgical wound infection (see “Perioperative considerations”)
- regular use of forced warm air covers applied to the anterior skin surface, to help patients maintain normothermia
- addition of a soft, self-retaining abdominal retractor—which creates an atraumatic circle of exposure up to a calculated 177 cm2 for a 15-cm incision (versus 113 cm2 calculated for traditional retraction)
- vertical, rather than lateral, digital extension of the initial transverse uterine incision
- identification of a subgroup in whom peritoneal closure is strongly recommended
- new data on the procedure’s effectiveness.3
Routinely use prophylactic antibiotics. Several recent studies have concluded that perioperative antibiotics reduce the incidence of endometritis and wound infection following elective and nonelective cesarean section.37 Administer ampicillin or a first-generation cephalosporin at umbilical cord clamping. For patients allergic to penicillin and cephalosporins, choose an alternative, such as clindamycin.
Give antacids 30 minutes before anesthesia (general or regional) to prevent pneumonitis from inhalation of gastric contents.
Clip pubic hair, rather than shave, to reduce the risk of wound infection.
Insert a Foley catheter, empty the bladder, and keep the catheter in place.
Position the patient in a 10°left lateral tilt, to avoid hypotension associated with aortocaval occlusion.
Routinely administer supplemental perioperative oxygen (80%), with either general or regional anesthesia; this activates alveolar immune defenses and halves the risk of surgical wound infections.
Neutrophil oxidative killing and phagocytosis—the most important defenses against surgical pathogens—depend on the partial pressure of oxygen in contaminated tissue. Giving supplemental oxygen during and for the first 2 hours after the procedure (by mask) is a practical, inexpensive way to reduce the incidence of surgical wound infection.38,39
Using 80% oxygen during and, for a short period, after surgery does not cause pulmonary toxicity such as atelectasis or impaired pulmonary function.40
Ensure normal body heat during and after cesarean to reduce the risk of postoperative surgical infection.40,41 Forced warm air covers applied to the anterior skin surface are the most effective way for warming surgical patients.9 IV fluid warming, though appropriate when large volumes are to be administered, is unnecessary for smaller operations.41
Modified abdominal incision reduces dissection
Make a straight low transverse incision with a scalpel, at a point approximately 3 to 4 cm above the symphysis pubis (FIGURE 1A).
The length of the incision is individualized (13 to 15 cm), though difficult fetal extraction is more likely if the abdominal incision is less than 15 cm.4,5
Divide the subcutaneous tissue transversely with an electrocautery knife. In a cutting and coagulation blend mode, the knife divides the fat while achieving hemostasis. To improve hemostasis, coagulate the blood vessels that cross the subcutaneous fat layer in a brushing manner before dividing them. To prevent unnecessary dead space, avoid filleting the fat and separating adherent subcutaneous fat from the anterior rectus fascia beyond what is needed to expose the fascia.
Open the fascia transversely with the electrocautery knife to the same length as the skin incision. Coagulate the blood vessels that cross the fascia before dividing them. Identify the median raphe by pulling up the superior edge of the abdominal incision.
Separate the rectus muscles in the midline by vertical blunt finger dissection (FIGURE 1B). If digital dissection is inefficient due to a dense, thick, or scarred median raphe, use an electrocautery knife, a scalpel, or scissors.
Open the peritoneum. This is facilitated by upward traction and elevation of the superior edge of the abdominal incision that lifts the peritoneum, allowing easy digital perforation using the index or middle finger (FIGURE 1C).
If this maneuver is not feasible, open the peritoneum in the traditional fashion.
Stretch the full thickness of the abdominal wall to full size of the skin incision, using 1 or 2 fingers of each hand (FIGURE 1D). Incorporate the skin, subcutaneous tissue, fascia layer, rectus muscles, and peritoneum. An assistant’s hands may be required. When needed, extend the peritoneal opening transversely on either side, to the midline and away from the bladder.
Digitally stretching the full-thickness abdominal incision is easily achieved due to the mechanical stretching of the anterior abdominal wall, edema, and increased vascularization that occur during pregnancy.
FIGURE 1A Create a modified abdominal incision
FIGURE 1B Create a modified abdominal incision
FIGURE 1C Create a modified abdominal incision
FIGURE 1D Create a modified abdominal incision
Retractor facilitates exposure
Our positive experience with the soft, self-retaining abdominal retractor for minilaparotomy and laparotomy6-8 compelled us to incorporate its use for cesarean 3 years ago.
The device consists of a flexible plastic inner ring and a firmer outer ring connected by a soft plastic sleeve. We use the large size of either of the 2 models currently available: the Mobius (Apple Medical Corporation, Marlboro, Miss) and the Protractor (Weck Closure Systems, Research Triangle Park, NC).
Introduce a hand through the laparotomy incision and evaluate the pelvis to ensure that no significant adhesions are present that may interfere with swift placement of the inner ring. If significant adhesions are found, use traditional metal retractors instead.
Squeeze the inner ring and insert it into the abdominal cavity toward the patient’s head, allowing the device to spring open against the parietal peritoneum (FIGURE 2A). Apply upper traction and elevate the superior edge of the abdominal incision to facilitate placement. Perform a digital check to ensure no tissue is trapped between the inner ring and the abdominal wall.
Hold up the outer ring, then roll the plastic sleeve until the ring completely inverts (FIGURE 2B). Repeat the process until the top ring is snug against the patient’s skin.
Advantages of the soft, self-retaining abdominal retractor include atraumatic retraction; incision protection; and adjustable height, making it ideal for obese patients.
FIGURE 2A Place the abdominal retractor
FIGURE 2B Place the abdominal retractor
Transverse hysterotomy in lower uterine segment
Make a transverse 2-cm uterine incision with a scalpel, approximately 1 cm above the vesicouterine peritoneal fold (identify this using gentle digital pressure to elevate the uterus) (FIGURE 3A).
Traditional bladder dissection is eliminated. We have found that when an adequate transverse hysterotomy is performed, a bladder flap is not required.1-3 In a recent randomized trial, Hohlagschwandtner et al9 confirmed our findings that bladder-flap omission was associated with reduced operative time and incision-delivery interval, decreased blood loss, and less need for postoperative analgesics.
An additional advantage of this omission: We can avoid making the uterine incision too low—especially when the cervix is fully dilated. Further, making the hysterotomy (uterine serosa and myometrium together) slightly above the vesicouterine peritoneal fold without bladder flap dissection frees the loose connective tissue between the uterus and the urinary bladder, allowing the spontaneous descent of the bladder.
Digitally extend the transverse uterine incision. We extend the initial incision not laterally but, instead, vertically (FIGURE 3B). The vertical digital traction on the initial transverse uterine incision creates a transverse dissociation of the horizontal myometrium fibers; this results in a transverse extension of the original incision.
This modification prevents unintended and uncontrolled lateral extensions of the incision that may lacerate the uterine vessels.10 It also prevents the uterine incision from becoming an inverted “U” and the undesirable accumulation of myometrium fibers at the ends of the incision. Such incisions usually do not reapproximate well during hysterotomy closure and may lead to sacculation-type defects.11
FIGURE 3A Use a standard hysterotomy in the lower uterine segment
FIGURE 3B Use a standard hysterotomy in the lower uterine segment
Delivering the fetus
For vertex presentations, place your hand into the uterine cavity between the lower edge of the hysterotomy and fetal head. While applying transabdominal fundal pressure, lift the head with your fingers and deliver it through the incision (FIGURE 4A).
The self-retaining retractor facilitates delivery of the fetal head by creating a rigid border around the abdominal incision. The back of the surgeon’s hand that is in the uterine cavity achieves better leverage, as it now rests on a rigid plane on the inferior part of the incision rather than on the back hand and the softer and pliable wound edge of the standard abdominal incision.
Vacuum-assistance. When delivery of the fetal head proves difficult, we use the soft vacuum cup, which avoids unnecessary intrauterine manipulation that may result in fetal or maternal trauma (FIGURE 4B).12
Breech extraction or transverse lie delivery is performed using standard extraction maneuvers (FIGURE 4C).13 We have found no need to perform T or J vertical extensions of the low transverse uterine incision, even in cases of a poorly developed lower uterine segment, abnormal presentation, or prematurity.
FIGURE 4A Deliver the fetal head
FIGURE 4B Deliver the fetal head
FIGURE 4C Deliver the fetal head
Uterine incision: 1-layer, in situ close
Remove the placenta only after it separates spontaneously—this greatly reduces blood loss compared with immediate manual placental delivery or umbilical-cord traction. Manually deliver the placenta only if it has not separated spontaneously after 5 minutes. Avoid routine manual clean-out of the uterine cavity if the placenta is completely delivered.1-3,14
Dilate the cervix to facilitate lochial discharge, when necessary.
Decrease uterine bleeding by massaging the uterus and administering diluted oxytocin.
Close the uterine incision in situ. Disadvantages of routine uterine exteriorization include discomfort and vomiting resulting from traction, exposure of the fallopian tubes to unnecessary trauma, increased risk of infection, possible rupture of the uteroovarian veins, and pulmonary embolism.
Exteriorization may also increase the risk of adhesion formation by exposing the uterine serosa to the abrasive effects of drying or microscopic abrasion if sponges are used to hold the uterus in place.1,14-17
Use a single-layer closure with a running suture of polyglycolic acid to close the uterine incision (FIGURE 5A). If needed, use atraumatic clamps to grasp the edges of the hysterotomy to facilitate visualization and closure.
Begin suturing at a point just beyond one end of the incision, continuing to the opposite side. Be sure to penetrate the full thickness of the myometrium and avoid incorporating the decidua in the suture line. After the uterine closure, use individual figure-of-8 sutures to control areas of persistent bleeding.
Compared with 2-layer closures, single-layer closure of a low transverse incision is associated with reduced operating time, improved hemostasis, and less tissue disruption; introduces less foreign material into the surgical site; reduces the need for postoperative analgesia; and potentially reduces infectious morbidity. A trial of labor after a cesarean section with 1-layer closure appears safe.18-22
We do not close the vesicouterine fold and parietal peritoneum (FIGURE 5B) or reapproximate the rectus muscles. It has been shown that a new peritoneal layer is formed within days of the original incision closure.
Leaving the peritoneum open leads to no increased postoperative complications, nor obvious differences in wound healing, wound dehiscence, or incidence of postoperative adhesions. Compared to women without peritoneal closure, those with peritoneal closure require a longer operative time, greater amounts of post-operative narcotics for postoperative pain, a greater need for bowel stimulants, and a longer postoperative hospitalization.23
In 1995, we performed laparoscopy for gynecologic conditions in 23 patients and repeat cesarean in 10 patients who had undergone our technique (with visceral and parietal peritoneum left open).2 We found no adhesions and a normal peritoneal lining in all cases.
In 1 subgroup, peritoneal closure is strongly recommended: cases in which 1 or both rectus muscles have been transected to increase surgical exposure. Extremely thick fibromuscular adhesions between the low anterior surface of the uterus and the under-surface of the rectus musculature may develop if the parietal peritoneum is not closed.
FIGURE 5A Close the incisions
FIGURE 5B Close the incisions
Abdominal closure
Close the rectus fascia with a continuous nonlocking 0 suture after the rectus muscles fall into place. Place entry and exit sites at 1-cm intervals, 1.5 cm beyond the wound edges—this minimizes the risk of sutures pulling through fascia. Interrupted sutures offer no greater tensile strength than continuous suturing.24 Tight, continuous suturing can lead to tissue ischemia, which may weaken the sutured fascia, and thus should be avoided.
The subcutaneous layer is not closed separately. One exception is obese patients with at least 2 cm of thick subcutaneous tissue. Thickness of subcutaneous tissue appears to be a significant risk factor for wound infection after cesarean section.25
In patients with thick subcutaneous tissue, you can reduce tension on the skin edges and the risk of subcutaneous infection, seroma formation, and wound disruption by placing interrupted 3-0 absorbable synthetic sutures to eliminate dead space.26-28 We have found no need to place Penrose drains or closed drainage systems in the subcutaneous layer.29-31
Close the skin using a subcuticular continuous absorbable 4-0 suture or, alternatively, metal staples, which are removed 4 days after surgery (FIGURE 5C).
FIGURE 5C Close the incisions
Prompt postoperative recovery
The patient may drink fluids in the recovery room, can resume a regular solid food diet within 4 hours after surgery, and may resume mobility as soon as anesthesia wears off.
Early breast-feeding is also allowed in the recovery room. In our experience, this has not been associated with complications, has been highly appreciated by the patients, and has led to earlier hospital discharge.
Control pain with meperidine (50–75 mg) or morphine (10 mg) parenterally every 3 to 4 hours. Other options include oxycodone and acetaminophen, oxycodone and aspirin, and ibuprofen.
Patients are usually discharged 48 to 72 hours following surgery.
Our experience: The numbers
Using this updated system, we have successfully completed 300 consecutive cesarean sections (210 primary, 90 repeat).
The average operating time was 19 minutes; average blood loss, 405 mL.
There was no postoperative febrile morbidity, wound infection, wound disruption, or wound hematoma.Only 3 patients developed superficial wound seromas, which were easily resolved. There were no intraoperative or postoperative complications.
We attribute the absence of wound infection to routine prophylactic antibiotics; supplemental perioperative oxygen; maintenance of normothermia; use of an electrocautery knife to create rapid hemostatic division of subcutaneous fat and anterior rectus fascia; a simplified abdominal wall opening; and placement of a self-retaining atraumatic retractor.10,32-36
Additional surgical procedures (tubal sterilization, myomectomy, appendectomy, and adhesiolysis) did not alter the recovery period, and all patients were able to get out of bed, shower, breast-feed, and care for their infants within 8 hours of surgery.
No infant complications related to the cesarean procedure occurred.
Patients were discharged from the hospital within 72 hours.
A prospective comparison study demonstrated that the original Pelosi-type of cesarean delivery was faster to perform, more cost-effective, and resulted in less maternal morbidity than the traditional cesarean technique.3
Dr. Pelosi II is a consultant for Apple Medical Corporation. Dr. Pelosi III reports no financial relationships relevant to this article.
- A simplified abdominal incision makes the traditional extensive dissection associated with the Pfannenstiel incision unnecessary.
- A soft, self-retaining abdominal retractor offers increased exposure, atraumatic retraction, incision protection, and adjustable height while facilitating delivery of the fetal head by creating a rigid border around the abdominal incision.
- Bladder-flap omission has been associated with reduced operative time and incision-delivery interval, decreased blood loss, and less need for postoperative analgesics.
Is the extensive dissection of the Pfannenstiel incision necessary in cesarean delivery? Is bladder dissection essential? Must the visceral and parietal peritoneum be closed?
The success of our minimally invasive cesarean technique suggests the answer is “no.”
The approach described here features a short operative time; minimal instrumentation; reduced surgical dissection; decreased postoperative pain; and reduced risk of blood loss, infection, and wound complications. It is easily learned and cost-effective, with a brief postoperative recovery period.
Among the updates made from the technique’s initial publication in the mid-1990s1,2:
- addition of routine perioperative oxygen (80%), to reduce the risk of surgical wound infection (see “Perioperative considerations”)
- regular use of forced warm air covers applied to the anterior skin surface, to help patients maintain normothermia
- addition of a soft, self-retaining abdominal retractor—which creates an atraumatic circle of exposure up to a calculated 177 cm2 for a 15-cm incision (versus 113 cm2 calculated for traditional retraction)
- vertical, rather than lateral, digital extension of the initial transverse uterine incision
- identification of a subgroup in whom peritoneal closure is strongly recommended
- new data on the procedure’s effectiveness.3
Routinely use prophylactic antibiotics. Several recent studies have concluded that perioperative antibiotics reduce the incidence of endometritis and wound infection following elective and nonelective cesarean section.37 Administer ampicillin or a first-generation cephalosporin at umbilical cord clamping. For patients allergic to penicillin and cephalosporins, choose an alternative, such as clindamycin.
Give antacids 30 minutes before anesthesia (general or regional) to prevent pneumonitis from inhalation of gastric contents.
Clip pubic hair, rather than shave, to reduce the risk of wound infection.
Insert a Foley catheter, empty the bladder, and keep the catheter in place.
Position the patient in a 10°left lateral tilt, to avoid hypotension associated with aortocaval occlusion.
Routinely administer supplemental perioperative oxygen (80%), with either general or regional anesthesia; this activates alveolar immune defenses and halves the risk of surgical wound infections.
Neutrophil oxidative killing and phagocytosis—the most important defenses against surgical pathogens—depend on the partial pressure of oxygen in contaminated tissue. Giving supplemental oxygen during and for the first 2 hours after the procedure (by mask) is a practical, inexpensive way to reduce the incidence of surgical wound infection.38,39
Using 80% oxygen during and, for a short period, after surgery does not cause pulmonary toxicity such as atelectasis or impaired pulmonary function.40
Ensure normal body heat during and after cesarean to reduce the risk of postoperative surgical infection.40,41 Forced warm air covers applied to the anterior skin surface are the most effective way for warming surgical patients.9 IV fluid warming, though appropriate when large volumes are to be administered, is unnecessary for smaller operations.41
Modified abdominal incision reduces dissection
Make a straight low transverse incision with a scalpel, at a point approximately 3 to 4 cm above the symphysis pubis (FIGURE 1A).
The length of the incision is individualized (13 to 15 cm), though difficult fetal extraction is more likely if the abdominal incision is less than 15 cm.4,5
Divide the subcutaneous tissue transversely with an electrocautery knife. In a cutting and coagulation blend mode, the knife divides the fat while achieving hemostasis. To improve hemostasis, coagulate the blood vessels that cross the subcutaneous fat layer in a brushing manner before dividing them. To prevent unnecessary dead space, avoid filleting the fat and separating adherent subcutaneous fat from the anterior rectus fascia beyond what is needed to expose the fascia.
Open the fascia transversely with the electrocautery knife to the same length as the skin incision. Coagulate the blood vessels that cross the fascia before dividing them. Identify the median raphe by pulling up the superior edge of the abdominal incision.
Separate the rectus muscles in the midline by vertical blunt finger dissection (FIGURE 1B). If digital dissection is inefficient due to a dense, thick, or scarred median raphe, use an electrocautery knife, a scalpel, or scissors.
Open the peritoneum. This is facilitated by upward traction and elevation of the superior edge of the abdominal incision that lifts the peritoneum, allowing easy digital perforation using the index or middle finger (FIGURE 1C).
If this maneuver is not feasible, open the peritoneum in the traditional fashion.
Stretch the full thickness of the abdominal wall to full size of the skin incision, using 1 or 2 fingers of each hand (FIGURE 1D). Incorporate the skin, subcutaneous tissue, fascia layer, rectus muscles, and peritoneum. An assistant’s hands may be required. When needed, extend the peritoneal opening transversely on either side, to the midline and away from the bladder.
Digitally stretching the full-thickness abdominal incision is easily achieved due to the mechanical stretching of the anterior abdominal wall, edema, and increased vascularization that occur during pregnancy.
FIGURE 1A Create a modified abdominal incision
FIGURE 1B Create a modified abdominal incision
FIGURE 1C Create a modified abdominal incision
FIGURE 1D Create a modified abdominal incision
Retractor facilitates exposure
Our positive experience with the soft, self-retaining abdominal retractor for minilaparotomy and laparotomy6-8 compelled us to incorporate its use for cesarean 3 years ago.
The device consists of a flexible plastic inner ring and a firmer outer ring connected by a soft plastic sleeve. We use the large size of either of the 2 models currently available: the Mobius (Apple Medical Corporation, Marlboro, Miss) and the Protractor (Weck Closure Systems, Research Triangle Park, NC).
Introduce a hand through the laparotomy incision and evaluate the pelvis to ensure that no significant adhesions are present that may interfere with swift placement of the inner ring. If significant adhesions are found, use traditional metal retractors instead.
Squeeze the inner ring and insert it into the abdominal cavity toward the patient’s head, allowing the device to spring open against the parietal peritoneum (FIGURE 2A). Apply upper traction and elevate the superior edge of the abdominal incision to facilitate placement. Perform a digital check to ensure no tissue is trapped between the inner ring and the abdominal wall.
Hold up the outer ring, then roll the plastic sleeve until the ring completely inverts (FIGURE 2B). Repeat the process until the top ring is snug against the patient’s skin.
Advantages of the soft, self-retaining abdominal retractor include atraumatic retraction; incision protection; and adjustable height, making it ideal for obese patients.
FIGURE 2A Place the abdominal retractor
FIGURE 2B Place the abdominal retractor
Transverse hysterotomy in lower uterine segment
Make a transverse 2-cm uterine incision with a scalpel, approximately 1 cm above the vesicouterine peritoneal fold (identify this using gentle digital pressure to elevate the uterus) (FIGURE 3A).
Traditional bladder dissection is eliminated. We have found that when an adequate transverse hysterotomy is performed, a bladder flap is not required.1-3 In a recent randomized trial, Hohlagschwandtner et al9 confirmed our findings that bladder-flap omission was associated with reduced operative time and incision-delivery interval, decreased blood loss, and less need for postoperative analgesics.
An additional advantage of this omission: We can avoid making the uterine incision too low—especially when the cervix is fully dilated. Further, making the hysterotomy (uterine serosa and myometrium together) slightly above the vesicouterine peritoneal fold without bladder flap dissection frees the loose connective tissue between the uterus and the urinary bladder, allowing the spontaneous descent of the bladder.
Digitally extend the transverse uterine incision. We extend the initial incision not laterally but, instead, vertically (FIGURE 3B). The vertical digital traction on the initial transverse uterine incision creates a transverse dissociation of the horizontal myometrium fibers; this results in a transverse extension of the original incision.
This modification prevents unintended and uncontrolled lateral extensions of the incision that may lacerate the uterine vessels.10 It also prevents the uterine incision from becoming an inverted “U” and the undesirable accumulation of myometrium fibers at the ends of the incision. Such incisions usually do not reapproximate well during hysterotomy closure and may lead to sacculation-type defects.11
FIGURE 3A Use a standard hysterotomy in the lower uterine segment
FIGURE 3B Use a standard hysterotomy in the lower uterine segment
Delivering the fetus
For vertex presentations, place your hand into the uterine cavity between the lower edge of the hysterotomy and fetal head. While applying transabdominal fundal pressure, lift the head with your fingers and deliver it through the incision (FIGURE 4A).
The self-retaining retractor facilitates delivery of the fetal head by creating a rigid border around the abdominal incision. The back of the surgeon’s hand that is in the uterine cavity achieves better leverage, as it now rests on a rigid plane on the inferior part of the incision rather than on the back hand and the softer and pliable wound edge of the standard abdominal incision.
Vacuum-assistance. When delivery of the fetal head proves difficult, we use the soft vacuum cup, which avoids unnecessary intrauterine manipulation that may result in fetal or maternal trauma (FIGURE 4B).12
Breech extraction or transverse lie delivery is performed using standard extraction maneuvers (FIGURE 4C).13 We have found no need to perform T or J vertical extensions of the low transverse uterine incision, even in cases of a poorly developed lower uterine segment, abnormal presentation, or prematurity.
FIGURE 4A Deliver the fetal head
FIGURE 4B Deliver the fetal head
FIGURE 4C Deliver the fetal head
Uterine incision: 1-layer, in situ close
Remove the placenta only after it separates spontaneously—this greatly reduces blood loss compared with immediate manual placental delivery or umbilical-cord traction. Manually deliver the placenta only if it has not separated spontaneously after 5 minutes. Avoid routine manual clean-out of the uterine cavity if the placenta is completely delivered.1-3,14
Dilate the cervix to facilitate lochial discharge, when necessary.
Decrease uterine bleeding by massaging the uterus and administering diluted oxytocin.
Close the uterine incision in situ. Disadvantages of routine uterine exteriorization include discomfort and vomiting resulting from traction, exposure of the fallopian tubes to unnecessary trauma, increased risk of infection, possible rupture of the uteroovarian veins, and pulmonary embolism.
Exteriorization may also increase the risk of adhesion formation by exposing the uterine serosa to the abrasive effects of drying or microscopic abrasion if sponges are used to hold the uterus in place.1,14-17
Use a single-layer closure with a running suture of polyglycolic acid to close the uterine incision (FIGURE 5A). If needed, use atraumatic clamps to grasp the edges of the hysterotomy to facilitate visualization and closure.
Begin suturing at a point just beyond one end of the incision, continuing to the opposite side. Be sure to penetrate the full thickness of the myometrium and avoid incorporating the decidua in the suture line. After the uterine closure, use individual figure-of-8 sutures to control areas of persistent bleeding.
Compared with 2-layer closures, single-layer closure of a low transverse incision is associated with reduced operating time, improved hemostasis, and less tissue disruption; introduces less foreign material into the surgical site; reduces the need for postoperative analgesia; and potentially reduces infectious morbidity. A trial of labor after a cesarean section with 1-layer closure appears safe.18-22
We do not close the vesicouterine fold and parietal peritoneum (FIGURE 5B) or reapproximate the rectus muscles. It has been shown that a new peritoneal layer is formed within days of the original incision closure.
Leaving the peritoneum open leads to no increased postoperative complications, nor obvious differences in wound healing, wound dehiscence, or incidence of postoperative adhesions. Compared to women without peritoneal closure, those with peritoneal closure require a longer operative time, greater amounts of post-operative narcotics for postoperative pain, a greater need for bowel stimulants, and a longer postoperative hospitalization.23
In 1995, we performed laparoscopy for gynecologic conditions in 23 patients and repeat cesarean in 10 patients who had undergone our technique (with visceral and parietal peritoneum left open).2 We found no adhesions and a normal peritoneal lining in all cases.
In 1 subgroup, peritoneal closure is strongly recommended: cases in which 1 or both rectus muscles have been transected to increase surgical exposure. Extremely thick fibromuscular adhesions between the low anterior surface of the uterus and the under-surface of the rectus musculature may develop if the parietal peritoneum is not closed.
FIGURE 5A Close the incisions
FIGURE 5B Close the incisions
Abdominal closure
Close the rectus fascia with a continuous nonlocking 0 suture after the rectus muscles fall into place. Place entry and exit sites at 1-cm intervals, 1.5 cm beyond the wound edges—this minimizes the risk of sutures pulling through fascia. Interrupted sutures offer no greater tensile strength than continuous suturing.24 Tight, continuous suturing can lead to tissue ischemia, which may weaken the sutured fascia, and thus should be avoided.
The subcutaneous layer is not closed separately. One exception is obese patients with at least 2 cm of thick subcutaneous tissue. Thickness of subcutaneous tissue appears to be a significant risk factor for wound infection after cesarean section.25
In patients with thick subcutaneous tissue, you can reduce tension on the skin edges and the risk of subcutaneous infection, seroma formation, and wound disruption by placing interrupted 3-0 absorbable synthetic sutures to eliminate dead space.26-28 We have found no need to place Penrose drains or closed drainage systems in the subcutaneous layer.29-31
Close the skin using a subcuticular continuous absorbable 4-0 suture or, alternatively, metal staples, which are removed 4 days after surgery (FIGURE 5C).
FIGURE 5C Close the incisions
Prompt postoperative recovery
The patient may drink fluids in the recovery room, can resume a regular solid food diet within 4 hours after surgery, and may resume mobility as soon as anesthesia wears off.
Early breast-feeding is also allowed in the recovery room. In our experience, this has not been associated with complications, has been highly appreciated by the patients, and has led to earlier hospital discharge.
Control pain with meperidine (50–75 mg) or morphine (10 mg) parenterally every 3 to 4 hours. Other options include oxycodone and acetaminophen, oxycodone and aspirin, and ibuprofen.
Patients are usually discharged 48 to 72 hours following surgery.
Our experience: The numbers
Using this updated system, we have successfully completed 300 consecutive cesarean sections (210 primary, 90 repeat).
The average operating time was 19 minutes; average blood loss, 405 mL.
There was no postoperative febrile morbidity, wound infection, wound disruption, or wound hematoma.Only 3 patients developed superficial wound seromas, which were easily resolved. There were no intraoperative or postoperative complications.
We attribute the absence of wound infection to routine prophylactic antibiotics; supplemental perioperative oxygen; maintenance of normothermia; use of an electrocautery knife to create rapid hemostatic division of subcutaneous fat and anterior rectus fascia; a simplified abdominal wall opening; and placement of a self-retaining atraumatic retractor.10,32-36
Additional surgical procedures (tubal sterilization, myomectomy, appendectomy, and adhesiolysis) did not alter the recovery period, and all patients were able to get out of bed, shower, breast-feed, and care for their infants within 8 hours of surgery.
No infant complications related to the cesarean procedure occurred.
Patients were discharged from the hospital within 72 hours.
A prospective comparison study demonstrated that the original Pelosi-type of cesarean delivery was faster to perform, more cost-effective, and resulted in less maternal morbidity than the traditional cesarean technique.3
Dr. Pelosi II is a consultant for Apple Medical Corporation. Dr. Pelosi III reports no financial relationships relevant to this article.
1. Pelosi MA, Ortega I. Cesarean section: Pelosi simplified technique. Rev Chil Obstet Gynecol. 1994;59:372-377.
2. Pelosi MA, II, Pelosi MA, III. Simplified cesarean section. Contemp OB/GYN. 1995;40:89-100.
3. Wood RM, Simon H, Oz AU. Pelosi-type vs traditional cesarean delivery: a prospective comparison. J Reprod Med. 1999;44:788-795.
4. Finan MA, Mastrogiannis DS, Spellacy WN. The “Allis” test for easy cesarean delivery. Am J Obstet Gynecol. 1991;164:772-775.
5. Ayers IW, Morley GW. Surgical incision for cesarean section. Obstet Gynecol. 1987;70:706-712.
6. Pelosi MA, II, Pelosi MA, III. Self-retaining abdominal retractor for minilaparotomy. Obstet Gynecol. 2000;96:775-778.
7. Pelosi MA, II, Pelosi MA, III. Pelosi minilaparotomy hysterectomy: Effective alternative to laparoscopy and laparotomy. OBG Manag. April 2003;16-33.
8. Pelosi MA, II, Pelosi MA, III. A novel minilaparotomy approach for large ovarian cysts. OBG Manag. February 2004:;17-30.
9. Hohlagschwandtner M, Ruecklinger E, Husslein P, et al. Is the formation of a bladder flap at cesarean necessary? A randomized trial. Obstet Gynecol. 2001;98:1089-1092.
10. Ferrari A, Frigerio L, Origoni M, et al. Modified Stark procedure for cesarean section. J Pelv Surg. 1996;2:239-244.
11. Field CS. Surgical techniques for cesarean section. Obstet Gynecol Clin North Amer. 1988;15:657-672.
12. Pelosi MA, Apuzzio J. Use of the soft, silicone obstetric vacuum cup for delivery of the fetal head at cesarean section. J Reprod Med. 1984;29:289-292.
13. Pelosi MA, Apuzzio J, Fricchione D, et al. The intra-abdominal version technique for delivery of transverse lie by low segment cesarean section. Am J Obstet Gynecol. 1979;135:1009-1012.
14. Magann EF, Dodson MK, Albert JR, et al. Blood loss at the time of cesarean section by method of placental removal and exteriorization versus in situ repair of the uterine incision. Surg Gynecol Obstet. 1993;177:389-392.
15. Stock RJ, Shelton H. Fatal pulmonary embolism occurring 2 hours after exteriorization of the uterus for repair following cesareans section. Milit Med. 1985;150:549-550.
16. Hershey DW, Quilligan EJ. Extra-abdominal uterine exteriorization at cesarean section. Obstet Genecol. 1978;52:189-191.
17. Wahab MA, Karantzis P, Eccersley PS, et al. A randomized, controlled study of uterine exteriorization and repair at cesarean section. Br J Obstet Gynecol. 1999;106:913-916.
18. Hauth JC, Owen J, Davis RO. Transverse uterine incision closure: one versus two layers. Am J Obstet Gynecol. 1992;167:1108-1111.
19. Ohel G, Younis JS, Lang N, et al. Double-layer closure of uterine incision with visceral and parietal peritoneal closure: Are they obligatory steps of routine cesarean sections? J Matern Fetal Med. 1996;5:366-369.
20. Tucker JM, Hauth JC, Hodgkins P, et al. Trial of labor after a one- or two-layer closure of a low transverse uterine incision. Am J Obstet Gynecol. 1993;168:545-546.
21. Chapman SJ, Owen J, Hauth JC. One- versus two-layer closure of a low transverse cesarean: the next pregnancy. Obstet Gynecol. 1997;89:16-18.
22. Jelsema RD, Wittinger JA, Vander Kolk KJ. Continuous, nonlocking, single-layer repair of the low transverse uterine incision. J Reprod Med. 1993;38:393-396.
23. Tulandi T, Al-Jaroudi D. Nonclosure of peritoneum: a reappraisal. Am J Obstet Gynecol. 2003;189:609-612.
24. Fagniez PL, Hay JM, Lacaine F. Abdominal midline incision closure. Arch Surg. 1985;120:1351-1355.
25. Vermillion ST, Lamoutte C, Soper DE, et al. Wound infection after cesarean: effect of subcutaneous tissue thickness. Obstet Gynecol. 2000;95:923-926.
26. Naumann RW, Hauth JC, Owen J, et al. Subcutaneous tissue approximation in relation to wound disruption after cesarean delivery in obese women. Obstet Gynecol. 1995;85:412-416.
27. Cetin A, Cetin M. Superficial wound disruption after cesarean delivery: effect of the depth and closure of subcutaneous tissue. Int J Gynaecol Obstet. 1997;57:17-21.
28. Del Valle GO, Combs P, Qualls C, et al. Does closure of Camper fascia reduce the incidence of post-cesarean superficial wound disruption? Obstet Gynecol. 1992;80:1013-1016.
29. Loong RL, Rogers MS, Chang AM. A controlled trial of wound drainage at cesarean section. Aust NZ J Obstet Gynaecol. 1988;28:266-269.
30. Saunders NJ, Barclay C. Closed suction wound drainage and lower-segment cesarean section. Br J Obstet Gynaecol. 1988;95:1060-1062.
31. Magann EF, Chauhan SP, Rodts-Palenik S, Bufkin L, Martin JN, Jr, Morrison JC. Subcutaneous stitch closure versus subcutaneous drain to prevent wound disruption after cesarean delivery: a randomized clinical trial. Am J Obstet Gynecol. 2002;186:1119-1123.
32. Stark M, Finkel AR. Comparison between the Joel-Cohen and Pfannenstiel incisions in cesarean section. Eur J Obstet Gynecol Reprod Biol. 1994;53:121-122.
33. Joel-Cohen S. Abdominal and vaginal hysterectomy. New techniques based on time and motion studies. London, England: William Heinemann Medical Books; 1972.
34. Hemsell DL, Hemsell PG, Nobles B, et al. Abdominal wound problems after hysterectomy with electrocautery versus scalpel subcutaneous incision. Infect Dis Obstet Gynecol. 1993;1:27-30.
35. Johnson CD, Serpell JW. Wound infection after abdominal incision with scalpel or diathermy. Br J Surg. 1990;77:626-629.
36. Hussain SA, Hussain S. Incisions with knife or diathermy and postoperative pain. Br J Surg. 1988;75:1179-1182.
37. American College of Obstetricians and Gynecologist. ACOG Practice Bulletin. Clinical management guidelines for obstetricians-gynecologists. No. 43, May, 2003. Management of preterm labor. Obstet Gynecol. 2003;101:1039-1047.
38. Greif R, Akca O, Hurn E-P, et al. Supplemental perioperative oxygen to reduce the incidence of surgical wound infection. N Engl J Med. 2000;342:161-167.
39. Sorensen LT, Karlsmark T, Gottrup F. Abstinence from smoking reduces incisional wound infection: a randomized controlled trial. Ann Surg. 2003;238:1-5.
40. Sessler DI, Akca O. Preventing surgical site infections without drugs. Contemp OB/GYN. 2004;49:78-87.
41. Kurz A, Sessler DI, Lenhardt R. Perioperative normothermia to reduce the incidence of surgical wound infection and shorten hospitalization. N Engl J Med. 1996;334:1209-1215.
1. Pelosi MA, Ortega I. Cesarean section: Pelosi simplified technique. Rev Chil Obstet Gynecol. 1994;59:372-377.
2. Pelosi MA, II, Pelosi MA, III. Simplified cesarean section. Contemp OB/GYN. 1995;40:89-100.
3. Wood RM, Simon H, Oz AU. Pelosi-type vs traditional cesarean delivery: a prospective comparison. J Reprod Med. 1999;44:788-795.
4. Finan MA, Mastrogiannis DS, Spellacy WN. The “Allis” test for easy cesarean delivery. Am J Obstet Gynecol. 1991;164:772-775.
5. Ayers IW, Morley GW. Surgical incision for cesarean section. Obstet Gynecol. 1987;70:706-712.
6. Pelosi MA, II, Pelosi MA, III. Self-retaining abdominal retractor for minilaparotomy. Obstet Gynecol. 2000;96:775-778.
7. Pelosi MA, II, Pelosi MA, III. Pelosi minilaparotomy hysterectomy: Effective alternative to laparoscopy and laparotomy. OBG Manag. April 2003;16-33.
8. Pelosi MA, II, Pelosi MA, III. A novel minilaparotomy approach for large ovarian cysts. OBG Manag. February 2004:;17-30.
9. Hohlagschwandtner M, Ruecklinger E, Husslein P, et al. Is the formation of a bladder flap at cesarean necessary? A randomized trial. Obstet Gynecol. 2001;98:1089-1092.
10. Ferrari A, Frigerio L, Origoni M, et al. Modified Stark procedure for cesarean section. J Pelv Surg. 1996;2:239-244.
11. Field CS. Surgical techniques for cesarean section. Obstet Gynecol Clin North Amer. 1988;15:657-672.
12. Pelosi MA, Apuzzio J. Use of the soft, silicone obstetric vacuum cup for delivery of the fetal head at cesarean section. J Reprod Med. 1984;29:289-292.
13. Pelosi MA, Apuzzio J, Fricchione D, et al. The intra-abdominal version technique for delivery of transverse lie by low segment cesarean section. Am J Obstet Gynecol. 1979;135:1009-1012.
14. Magann EF, Dodson MK, Albert JR, et al. Blood loss at the time of cesarean section by method of placental removal and exteriorization versus in situ repair of the uterine incision. Surg Gynecol Obstet. 1993;177:389-392.
15. Stock RJ, Shelton H. Fatal pulmonary embolism occurring 2 hours after exteriorization of the uterus for repair following cesareans section. Milit Med. 1985;150:549-550.
16. Hershey DW, Quilligan EJ. Extra-abdominal uterine exteriorization at cesarean section. Obstet Genecol. 1978;52:189-191.
17. Wahab MA, Karantzis P, Eccersley PS, et al. A randomized, controlled study of uterine exteriorization and repair at cesarean section. Br J Obstet Gynecol. 1999;106:913-916.
18. Hauth JC, Owen J, Davis RO. Transverse uterine incision closure: one versus two layers. Am J Obstet Gynecol. 1992;167:1108-1111.
19. Ohel G, Younis JS, Lang N, et al. Double-layer closure of uterine incision with visceral and parietal peritoneal closure: Are they obligatory steps of routine cesarean sections? J Matern Fetal Med. 1996;5:366-369.
20. Tucker JM, Hauth JC, Hodgkins P, et al. Trial of labor after a one- or two-layer closure of a low transverse uterine incision. Am J Obstet Gynecol. 1993;168:545-546.
21. Chapman SJ, Owen J, Hauth JC. One- versus two-layer closure of a low transverse cesarean: the next pregnancy. Obstet Gynecol. 1997;89:16-18.
22. Jelsema RD, Wittinger JA, Vander Kolk KJ. Continuous, nonlocking, single-layer repair of the low transverse uterine incision. J Reprod Med. 1993;38:393-396.
23. Tulandi T, Al-Jaroudi D. Nonclosure of peritoneum: a reappraisal. Am J Obstet Gynecol. 2003;189:609-612.
24. Fagniez PL, Hay JM, Lacaine F. Abdominal midline incision closure. Arch Surg. 1985;120:1351-1355.
25. Vermillion ST, Lamoutte C, Soper DE, et al. Wound infection after cesarean: effect of subcutaneous tissue thickness. Obstet Gynecol. 2000;95:923-926.
26. Naumann RW, Hauth JC, Owen J, et al. Subcutaneous tissue approximation in relation to wound disruption after cesarean delivery in obese women. Obstet Gynecol. 1995;85:412-416.
27. Cetin A, Cetin M. Superficial wound disruption after cesarean delivery: effect of the depth and closure of subcutaneous tissue. Int J Gynaecol Obstet. 1997;57:17-21.
28. Del Valle GO, Combs P, Qualls C, et al. Does closure of Camper fascia reduce the incidence of post-cesarean superficial wound disruption? Obstet Gynecol. 1992;80:1013-1016.
29. Loong RL, Rogers MS, Chang AM. A controlled trial of wound drainage at cesarean section. Aust NZ J Obstet Gynaecol. 1988;28:266-269.
30. Saunders NJ, Barclay C. Closed suction wound drainage and lower-segment cesarean section. Br J Obstet Gynaecol. 1988;95:1060-1062.
31. Magann EF, Chauhan SP, Rodts-Palenik S, Bufkin L, Martin JN, Jr, Morrison JC. Subcutaneous stitch closure versus subcutaneous drain to prevent wound disruption after cesarean delivery: a randomized clinical trial. Am J Obstet Gynecol. 2002;186:1119-1123.
32. Stark M, Finkel AR. Comparison between the Joel-Cohen and Pfannenstiel incisions in cesarean section. Eur J Obstet Gynecol Reprod Biol. 1994;53:121-122.
33. Joel-Cohen S. Abdominal and vaginal hysterectomy. New techniques based on time and motion studies. London, England: William Heinemann Medical Books; 1972.
34. Hemsell DL, Hemsell PG, Nobles B, et al. Abdominal wound problems after hysterectomy with electrocautery versus scalpel subcutaneous incision. Infect Dis Obstet Gynecol. 1993;1:27-30.
35. Johnson CD, Serpell JW. Wound infection after abdominal incision with scalpel or diathermy. Br J Surg. 1990;77:626-629.
36. Hussain SA, Hussain S. Incisions with knife or diathermy and postoperative pain. Br J Surg. 1988;75:1179-1182.
37. American College of Obstetricians and Gynecologist. ACOG Practice Bulletin. Clinical management guidelines for obstetricians-gynecologists. No. 43, May, 2003. Management of preterm labor. Obstet Gynecol. 2003;101:1039-1047.
38. Greif R, Akca O, Hurn E-P, et al. Supplemental perioperative oxygen to reduce the incidence of surgical wound infection. N Engl J Med. 2000;342:161-167.
39. Sorensen LT, Karlsmark T, Gottrup F. Abstinence from smoking reduces incisional wound infection: a randomized controlled trial. Ann Surg. 2003;238:1-5.
40. Sessler DI, Akca O. Preventing surgical site infections without drugs. Contemp OB/GYN. 2004;49:78-87.
41. Kurz A, Sessler DI, Lenhardt R. Perioperative normothermia to reduce the incidence of surgical wound infection and shorten hospitalization. N Engl J Med. 1996;334:1209-1215.
Anterior vaginal wall prolapse: The challenge of cystocele repair
- At this time, the traditional anterior colporrhaphy with attention to apical suspension remains the gold standard.
- If only some defects of the anterior wall are addressed at the time of reconstructive surgery, failure may be more likely.
- Women with grade 3 or 4 cystoceles often have evidence of bladder outlet obstruction on urodynamic testing.
- In 52% of cases, cystoceles coexist with detrusor instability and evidence of impaired detrusor contractility.
- A thorough preoperative evaluation includes assessing the apex, having the patient strain to maximize the defect, looking for paravaginal detachments, and making every effort to “unmask” occult stress urinary incontinence.
Ask a pelvic reconstructive surgeon to name the most difficult challenge, and the answer is likely to be anterior vaginal wall prolapse. The reason: The anterior wall usually is the leading edge of prolapse and the most common site of relaxation or failure following reconstructive surgery. This appears to hold true regardless of surgical route or technique.
Short-term success rates of anterior wall repairs appear promising, but long-term outcomes are not as encouraging. Success usually is claimed as long as the anterior wall is kept above the hymen, since the patient rarely reports symptoms in these cases.
Another challenge involves the use of allografts or xenografts, which have not undergone sufficient study to determine their long-term benefit or risks in comparison with traditional repairs.
This article reviews anatomy of the anterior vaginal wall and its supports, as well as surgical technique and outcomes.
Why the anterior wall is more susceptible to prolapse
One theory is that, in comparison with the posterior compartment, the anterior wall is not as well supported by the levator plate, which counters the effects of gravity and abdominal pressure. Normally, the anterior wall rests horizontally on the posterior wall, which in turn rests on the levator plate. When the levator muscles weaken, the anterior wall is the first to fall as increasing force is placed on the connective tissue supports.
Other possibilities: The anterior compartment’s attachments to the pelvic sidewall or apex may be weaker, the anterior wall may be more elastic or less dense than the posterior wall, and the anterior wall may be more susceptible to damage during childbirth or to the effects of age and loss of estrogen.
If only some defects are addressed at surgery, failure may be more likely. Some experts believe pelvic surgeons have focused too much attention on the urethrovesical junction in patients with concomitant urinary incontinence and not enough attention on suspending the anterior wall at the apex.
For most women, it is probably a combination of many of these factors that renders the anterior compartment so vulnerable.
Anatomy of the pelvic floor
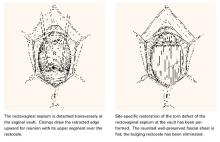
The anterior vaginal wall resembles a trapezoidal plane due to ventral and more medial attachments near the pubic symphysis, and dorsal and more lateral attachments near the ischial spine (FIGURE 1).1 This helps explain the many appearances of the cystocele. The type of cystocele is defined by the location of the break in the fascial attachments.
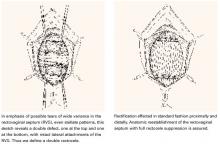
Paravaginal defects. The trapezoidal anterior wall is suspended on both sides from the parietal fascia overlying the levator ani muscles at the arcus tendineus fascia pelvis (ATFP). Prolapse can occur when there is loss of attachment to the pelvic sidewall at any point between the pubis and ischial spine.
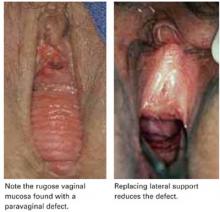
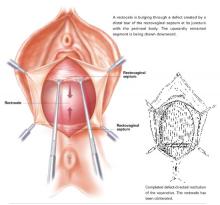
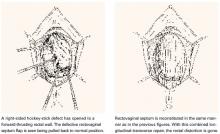
First described by White2 and characterized later by Richardson et al,3 this loss of lateral attachment is called a paravaginal defect or displacement cystocele (FIGURE 2). The goal of paravaginal repair is to reattach the lateral vaginal walls to the ATFP, either abdominally, laparoscopically, or vaginally.
Central defects, the rarest type of anterior wall prolapse, involve a loss of support near the pubis and tend to be smaller. The most common manifestation is urethral hypermobility.
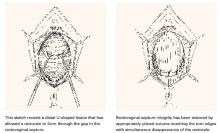
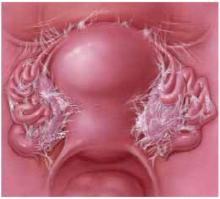
Transverse defects occur when the top of the pubocervical fascia detaches from the cervix or vaginal apex, both of which are suspended from the uterosacral-cardinal ligament complex. A transverse cystocele is evidenced by loss of the anterior fornix. The anterior wall appears to be attenuated in the midline, and the vaginal mucosa is pale, thin, and smooth (FIGURE 3).
Goals of traditional repair. The traditional anterior colporrhaphy aims to excise or reinforce the attenuated transverse defect with plication of the “endopelvic fascia” in the midline of the anterior vaginal wall. The endopelvic fascia is not true fascia but the muscularis of the vaginal wall. It is comprised of smooth muscle and elastin along with the collagenous adventitia layer.4
The importance of restoring apical wall support becomes apparent when one considers the trapezoidal anatomy. The most common sites of defects or detachments of the anterior wall are near the ischial spines laterally. In an operative case series of paravaginal defects, DeLancey1 found the site of defect to be near the ischial spine in 96% of cases. The reattachment of the apex near the level of the spine becomes the highest point of support for the anterior vaginal wall.
This cephalad apical attachment can be accomplished in a variety of ways, by suspending the vaginal apex from the uterosacral ligaments, from the sacrospinous ligament, or via abdominal sacrocolpopexy.
FIGURE 1 Anatomy of the anterior wall
The anterior vaginal wall resembles a trapezoidal plane, with ventral and more medial attachments near the pubic symphysis, and dorsal and more lateral attachments to the ischial spine. Detachment from the pelvic sidewall and ischial spine results in anterior wall prolapse (right).
FIGURE 2 Paravaginal defect
FIGURE 3 Transverse defect
A transverse defect with loss of the anterior fornix. The loss of cephalad apical attachment at the level of the ischial spine leads to anterior wall prolapse. Suspending the upper vagina from shortened cardinal/uterosacral ligaments, the sacrospinous ligament, or via abdominal sacrocolpopexy is as important as plication.
Symptoms of anterior wall prolapse
As with other forms of pelvic organ prolapse, many patients complain of a bulge or feeling of pelvic pressure when the anterior vaginal wall has come through the introitus. However, some symptoms of anterior wall prolapse are unique.
Incontinence is not universal. A common misperception is that most patients with cystocele also experience stress urinary incontinence (SUI), which can develop when there is loss of urethral support and descent of the lower vaginal wall along with urethral hypermobility. However, there is no defining degree of hypermobility that links anterior wall prolapse with SUI. That is because the continence mechanism relies not only on urethral position and lateral attachments, but also on the neuromuscular function of the pelvis and lower urinary tract.
In fact, descent of the midvagina under the bladder base may actually reduce the chance of SUI. The reason: As a woman strains, the increased abdominal pressure pushes the cystocele farther and farther out. As the cystocele enlarges, it creates a functional outlet obstruction by kinking the urethra shut. When this is the case, patients may complain of prolonged voiding, an intermittent urine stream, and/or urinary retention. The woman may have to elevate the vaginal wall to empty her bladder. Patients with chronic urinary retention are at risk of developing recurrent urinary tract infections.
Bladder outlet obstruction and detrusor dysfunction. Women with grade 3 or 4 cystoceles often have evidence of bladder outlet obstruction on urodynamic testing, according to a study that found such evidence in 57% of subjects.5 After reduction of the prolapse with a pessary, obstructed flow reverted to normal in 94% of these women.
A large proportion (52%) of women with cystoceles also have detrusor instability, as well as evidence of impaired detrusor contractility. Many complain of urinary frequency and urgency and difficulty emptying the bladder.5
Again, this phenomenon is complex, related not only to anatomy but to altered neuromuscular function of the lower urinary tract. Incomplete emptying, frequency, and urgency may arise from stretching of the bladder base as it prolapses through the vaginal introitus, resulting in urinary retention. These symptoms often are less pronounced at night when the patient is supine.
We reviewed 35 cases of anterior wall prolapse greater than 1 cm outside the hymen, with elevated postvoid residuals exceeding 100 cc on 2 separate occasions.6 Thirty-one (89%) had normal postvoid residuals following reconstructive surgery and correction of their anterior wall prolapse.
Preoperative assessment
A careful physical exam is a prerequisite for all surgical repairs of pelvic organ prolapse. During this exam, identify the sites of defects and detachments.
Maximize the defect. Have the patient perform the Valsalva maneuver, cough, and/or strain while sitting upright or standing. As she is performing these maneuvers, ask her if this feels like her maximum prolapse. A split speculum often aids in visualizing the anterior and posterior compartments without pressure from the opposite vaginal wall.
Assess the apex. Place a large swab in the vagina, hold it gently against the apex, and ask the patient to strain. If the swab is pushed out, the apex needs suspension.
This technique can help identify apical relaxation that may be masked by a large anterior or posterior wall defect. A standardized staging system, such as the Pelvic Organ Prolapse Quantitative Examination (POP-Q) or Baden-Walker, aids in communicating and documenting the prolapse. In addition, it allows the surgeon to track anatomical outcomes after surgery.
Look for paravaginal defects by supporting the lateral anterior walls with a ring forceps at the level of the ATFP. Barber et al7 found this maneuver to be highly sensitive (90–94%): If no paravaginal defect was suspected clinically, none was found intraoperatively. However, the positive predictive value was poor (57%), in that defects suspected preoperatively were confirmed during surgery in less than two thirds of patients.
These findings point to the importance of careful intraoperative assessment, both before and during the repair procedure.
Limited utility of imaging studies. The use of radiologic studies such as defecography or dynamic magnetic resonance imaging of the pelvis may aid in the evaluation of defecatory disorders or suspected sigmoidocele or rectal prolapse, but have not been studied sufficiently to determine the impact on surgical outcome.
Unmasking SUI
As mentioned above, women with anterior wall prolapse do not always complain of stress incontinence. However, correction of the cystocele can relieve their obstructive voiding and unmask “occult” SUI. Various techniques have been described to elevate the anterior wall with pessaries, swabs, etc, during urodynamic testing to predict which women should have an incontinence procedure performed at the time of reconstructive surgery.
Conflicting rates of occult SUI have been reported, with estimates ranging from 36% to 80%.8 Although preoperative urodynamic testing indicates a high rate of occult stress incontinence, a study by Borstad et al9 suggests that the rate of de novo incontinence may be lower and that preoperative urodynamic findings are not predictive of postoperative continence status. In that study, 16 of 73 women (22%) developed stress incontinence following surgery for prolapse when no incontinence procedure was performed. Advanced age increased the risk of incontinence after surgery.
Contrast these findings with those of Chaikin and colleagues,10 who prospectively followed 24 patients with grade 3 or 4 cystoceles. Preoperative urodynamics showed a 58% rate of occult stress incontinence. All these patients were also defined as having intrinsic sphincter deficiency with leak point pressures below 60 cm water. The incontinent group underwent anterior colporrhaphy and concomitant pubovaginal sling, compared with anterior colporrhaphy alone for those without incontinence. Postoperatively, 2 patients who had the pubovaginal sling procedure reported continued stress incontinence (14%). No new symptoms of incontinence were reported in the patients without leakage on preoperative urodynamics. Thus, preoperative urodynamics were 100% accurate in determining which women did not need additional surgery for SUI.
Implications of a negative stress test. Our experience has shown that, despite our best attempts, a negative stress test with the prolapse reduced prior to surgery is less than 100% predictive. Occasionally, new SUI occurs after reconstructive surgery. It is unclear whether this incontinence is caused by straightening the urethra and reducing the bulge or secondary to the dissection of surgery.
Tips on technique
Anterior colporrhaphy traditionally is performed with plication of the “endopelvic fascia” or fibromuscular layer at the bladder neck with a Kelly plication stitch. Using “3-point” traction aids in dissecting the muscularis (FIGURE 4). Repair the remainder of the cystocele using vertical mattress stitches (1 or 2 layers) from the bladder neck to the apex.
Avoid creating weak areas. Using this technique, the repair frequently stops short of the apex, leaving a “gap” or weak area. One way to avoid this is to begin plication at the apex instead of the bladder neck (FIGURE 5).
Next, excise the excess vaginal tissue and close with interrupted fine absorbable sutures (FIGURE 6).
Recreate apical support. Another problem with traditional repairs is that they do not reestablish apical support. In many patients with anterior wall prolapse, reattachment of the apex reduces the cystocele. Therefore, it often is necessary to combine anterior colporrhaphy with an apical repair procedure such as uterosacral ligament suspension or sacrospinous ligament suspension.
Sutures for the apical repair should be placed and held prior to initiating the anterior colporrhaphy. At the end of the anterior repair, incorporate the apical sutures into the vaginal cuff.
Careful attention to the integrity and strength of the tissue is crucial. Regardless of the type of transvaginal suspension, we advocate bringing 1 arm of the suspension suture through the anterior wall of the cuff. Then place the other suture arm through the posterior cuff so that, when tied, anterior and posterior walls are brought together and suspended.
Using prolonged-delayed absorbable suture allows for a full-thickness bite, ensuring scarring to the suspensory ligament. If permanent suture is used for the uterosacral suspension, place the stitches along the inside surface of the anterior wall with a strong, broad bite that incorporates the muscularis or “endopelvic fascia.”
The occasional enterocele. When a transverse cystocele occurs following hysterectomy, the surgeon should be on the lookout for an enterocele, which sometimes accompanies anterior wall prolapse. The enterocele should be corrected at the time of surgery by closing the defect and suspending the cuff.
FIGURE 4 Three-point traction
Three-point traction using Allis clamps. The assistant retracts with DeBackey forceps to allow dissection of the muscularis. An index finger placed firmly against the vaginal mucosa enables the surgeon to judge depth of dissection.
FIGURE 5 Begin plication at the apex
Plication begins at the apex with vertical mattress stitches. Use 3-0 prolonged delayed absorbable or permanent suture in the anterior wall.
FIGURE 6 The reduced cystocele
The cystocele reduced following midline plication of the vaginal muscularis. The excess vagina is then trimmed and closed with interrupted 3-0 absorbable suture.
Functional surgical outcomes
Because of the long association between anterior wall prolapse and SUI, most surgeons evaluate patients preoperatively to determine the need for concomitant incontinence procedures. As a result, the literature reporting surgical cystocele repair via anterior colporrhaphy frequently uses continence of urine as the functional outcome. This is not surprising considering that anterior repair and Kelly plication, as reported by Howard Kelly more than 75 years ago, have been the gold standard for surgical correction of anterior wall prolapse and SUI.
In a series of 194 SUI patients who underwent anterior colporrhaphy, Beck11 found that adding a modified Kelly plication, including a vaginal retropubic urethropexy, increased the cure rate for SUI from 75% to 94%. Unfortunately, he did not report the anatomic success of the anterior colporrhaphy.
Kohli et al12 also retrospectively examined patients who had undergone anterior colporrhaphy with and without needle bladder-neck suspension. Although the cure rate for SUI was not reported, patients who underwent concomitant needle suspension had a higher rate of recurrent cystocele: 33% (n = 40) versus 7% (n = 27). Investigators theorized that retropubic dissection at the time of transvaginal needle suspension resulted in an iatrogenic paravaginal defect and denervation of the anterior vaginal wall.
The risks of needle suspension. A randomized controlled trial by Bump et al8 also suggests that needle suspension should be avoided. In that trial, 29 patients with stage 3 and 4 prolapse were randomized to needle colposuspension or endopelvic fascia plication. They, too, found that needle colposuspension carried a higher rate of recurrent anterior prolapse. Further, it did not reduce the rates of SUI compared with fascia plication.
Although incontinence surgery performed at the time of cystocele repair will reduce the rates of de novo incontinence, the higher rates of cystocele recurrence associated with some procedures warrants judicious preoperative planning. Clearly, needle suspension should not be performed as an incontinence procedure or repair of anterior wall prolapse. Whether other vaginal incontinence procedures, eg, midurethral slings, lead to recurrence of anterior wall prolapse deserves further investigation.
Anatomic outcomes
Midline colporrhaphy. Because anterior colporrhaphy is rarely performed alone, few series describe patients having undergone simply an anterior repair. Stanton et al13 followed 54 women for up to 2 years after they underwent traditional midline plication with vaginal hysterectomy for prolapse. Eight (15%) of the women had recurrent anterior wall prolapse.
Colombo et al14 randomized 71 women with clinical SUI and stage 2 or 3 prolapse to Burch colposuspension or anterior colporrhaphy with Kelly plication. All women were followed for at least 8 years. The cure rate for SUI was 86% for the Burch procedure, compared with 52% for anterior repair and Kelly plication. However, 12 (34%) women treated with Burch colposuspension and 1 (3%) treated with anterior colporrhaphy had recurrent cystocele of grade 2 or 3 with or without prolapse at other vaginal sites.
Abdominal paravaginal repair. Shull15 followed 149 women for 6 months to 4 years after they underwent abdominal paravaginal repair with the urethrovesical stitch brought through Cooper’s ligament for treatment of SUI and paravaginal cystocele. He reported a 5% recurrence of anterior wall prolapse.
In another series, Shull16 reported on 62 women who were followed for a mean of 18 months after abdominal paravaginal repair. Four of 57 (7%) had recurrent vaginal prolapse to the hymen.
In a cohort of 102 patients evaluated with the Pelvic Organ Prolapse Quantitative Examination (POP-Q) at our institution, the maximal point of prolapse was the anterior wall in 60% of cases; the apex and posterior wall each accounted for roughly half the remaining cases (unpublished data).
Ellerkmann et al30 reported on 237 consecutive patients who presented with symptoms of pelvic organ prolapse. In 77 women (33%), anterior compartment pelvic organ prolapse predominated; 46 patients (19%) had posterior compartment prolapse; and 22 patients (11%) had apical prolapse.
Hendrix et al31 analyzed patients from the Women’s Health Initiative (WHI) and found that the anterior compartment predominated over the posterior compartment. In a follow-up study from the WHI, Handa32 reported on 412 women followed for 2 to 8 years. Among those who entered the WHI protocol without cystocele, 1 in 4 was diagnosed with it at some point in the study. This compares to 1 in 6 for rectocele and 1 in 100 for uterine prolapse. The majority of all defects were grade 1 or relaxation above the hymen.
Vaginal paravaginal repair. Mallipeddi et al17 reported on 45 patients undergoing vaginal paravaginal repair over 2 years, with 35 women followed for a mean of 1.6 years. Incontinent patients had a Kelly plication performed at the time of vaginal paravaginal repair. Recurrence rates were 3% for cystocele, 14% for rectocele, and 20% for enterocele.
Young et al18 followed 100 women for as long as 36 months after bilateral paravaginal repair using 1 to 6 expanded polytetrafluoroethylene (Gore-Tex) CV-0 sutures and midline colporrhaphy. Two patients had grade 1 or 2 failure at the lateral fixation points, but 21 patients had recurrent midline defects, all but 1 inside the hymen. Several patients had bloody discharge from the permanent sutures.
Sacrocolpopexy. Brubaker19 retrospectively reviewed 65 women who underwent sacrocolpopexy for apical prolapse. Three months postoperatively 19 patients (29%) had persistent anterior wall defects.
Uterosacral suspension. Shull et al20 also found the anterior segment to have the most recurrent defects. In that study, which had an average follow-up of a little over a year, 289 patients underwent vaginal uterosacral ligament repair of the apex, and 264 had an anterior wall defect preoperatively. At the time of furthest follow-up, 26 patients (9%) had failure at this site. This study confirmed that the anterior compartment is the most likely site to fail, and also that it fails the quickest.
Sacrospinous ligament suspension (SSLS). Morley and DeLancey21 found a 22% cystocele recurrence rate in 71 women 1 year after SSLS, with most of them asymptomatic. Shull22 reported a 30% incidence of cystoceles after SSLS. Paraiso and colleagues23 reported on 243 women undergoing SSLS and pelvic reconstructive surgery. Of these, 217 patients underwent concomitant anterior colporrhaphy. Follow-up at 74 months found 37% with symptomatic recurrence at the anterior wall, 13% at the posterior wall, and 8% at the apex.
Vaginal versus abdominal repair
Few studies have compared vaginal and abdominal repair of pelvic organ prolapse, including anterior wall prolapse.
In a trial by Benson et al,24 women with prolapse to or beyond the hymen were randomized to bilateral sacrospinous vault suspension and vaginal paravaginal repair (n = 48) or abdominal sacrocolpopexy with abdominal paravaginal repair (n = 40). One third of patients in each group also underwent anterior colporrhaphy. After a mean follow-up of 2.5 years, 16 of 20 women required reoperation for recurrent cystocele—12 (29%) from the vaginal group and 4 (10.5%) from the abdominal group. Vaginal vault eversion recurred in 5 women from the vaginal group and 1 from the abdominal group.
Investigators concluded that the anterior wall was the most likely site of failure because of the posterior placement of the vaginal apex with SSLS, predisposing the anterior wall to greater pressures and to neuropathy caused by lateral dissection of the anterior wall. Earlier studies have demonstrated that neuropathy may occur after extensive dissection of the vaginal wall and may affect the strength and integrity of the muscular support tissues.25,26
Allografts and xenografts
The difficulty of repairing anterior wall prolapse has led some pelvic surgeons to use mesh for cystocele repair. When Julian27 randomized 24 patients with recurrent cystocele to transvaginal repair with and without polypropylene (Marlex) mesh, 4 patients in the control group and no patients in the mesh group had recurrences (P <.05). However, 3 patients (25%) had mesh-related complications.
Weber et al28 randomized patients to standard midline plication, plication of the paravaginal tissue more laterally, or standard plication plus polyglactin 910 (Vicryl) mesh. Among 83 patients who returned for follow-up, there were no differences in anatomic outcome. Weber and colleagues concluded that there is little benefit to using mesh to correct cystoceles.
Still, although the overall cure rate was low (30–46%), most patients had cystocele to the hymen and not beyond, with significant improvement of symptoms. Although this cannot be defined as an anatomic cure, it is encouraging that the majority of patients appear to have benefited from surgery.
Sand et al29 randomized 161 women with the anterior wall to or beyond the hymen to traditional anterior colporrhaphy with or without Vicryl. The 2-inch square mesh was not placed over the repair as described above, but was folded into the anterior colporrhaphy stitches. At 1 year, 16 (22%) of 73 women with mesh and 28 (40%) of the 88 women without mesh had recurrent central cystoceles beyond the midvagina (P = .02). No women had cystoceles beyond the hymen or vaginal erosions.
Difficulty of interpreting the evidence
Because of the broad range of study designs, small number of patients per series, variety of concomitant procedures, and wide range of variables used to describe recurrence and success, it is difficult to draw conclusions from the literature. The evidence does suggest that the risks of wide vaginal dissection required for vaginal paravaginal repair outweigh the benefits. As a result, we have abandoned this technique. As mentioned above, it remains unclear whether graft materials will prove to be of long-term benefit for either midline plication or paravaginal repair.
The gold standard, for now
Prolapse of the anterior vaginal wall remains a challenge for the gynecologic surgeon. Careful preoperative and intraoperative evaluation and identification of support defects should guide repairs.
Randomized, controlled trials of midline versus paravaginal repair, as well as use of various graft materials, are greatly needed. These studies should not only address recurrence of prolapse symptoms, but the impact of surgery on sexual and lower urinary tract function.
At this time, the traditional anterior colporrhaphy with attention to apical suspension remains the gold standard.
The authors report no financial relationships relevant to this article.
1. DeLancey JOL. Fascial and muscular abnormalities in women with urethral hypermobility and anterior vaginal wall prolapse. Am J Obstet Gynecol. 2002;187:93-98.
2. White GR. Cystocele: a radical cure by suturing lateral sulci of vagina to white line of pelvic fascia. JAMA. 1909;21:1707-1710.
3. Richardson AC, Lyon JB, Williams NL. A new look at pelvic relaxation. Am J Obstet Gynecol. 1976;126:568-571.
4. Weber AM, Walters MD. Anterior vaginal prolapse: review of anatomy and techniques of surgical repair. Obstet Gynecol. 1997;89:311-318.
5. Romanzi LJ, Chaikin DC, Blaivas JG. The effect of genital prolapse on voiding. J Urol. 1999;161:581-586.
6. FitzGerald MP, Kulkarni N, Fenner D. Postoperative resolution of urinary retention in patients with advanced pelvic organ prolapse. Am J Obstet Gynecol. 2000;183:1361-1364.
7. Barber MD, Cundiff GW, Weidner AC, Cotes KW, Bump RC, Addison WA. Accuracy of clinical assessment of paravaginal defects in women with anterior vaginal wall proloapse. Am J Obstet Gynecol. 1999;181:1-7.
8. Bump RC, Hurt WG, Theofrastous JP, et al. Randomized prospective comparison of needle colposuspension versus endopelvic fascia plication for potential stress incontinence prophylaxis in women undergoing vaginal reconstruction for stage III or IV pelvic organ prolapse. The Continence Program for Women Research Group. Am J Obstet Gynecol. 1996;175:326-333.
9. Borstad E, Rud T. The risk of developing urinary stress incontinence after vaginal repair in continent women. Acta Obstet Gynecol Scand. 1989;68:545-549.
10. Chaikin DC, Groutz A, Blaivas JG. Predicting the need for anti-incontinence surgery in continent women undergoing repair of severe urogenital prolapse. J Urol. 2000;163:531-534.
11. Beck RP, McCormick S, Nordstrom L. A 25-year experience with 519 anterior colporrhaphy procedures. Obstet Gynecol. 1991;78:1011-1018.
12. Kohli N, Sze EHM, Roat TW, Karram M. Incidence of recurrent cystocele after anterior colporrhaphy with and without concomitant transvaginal needle suspension. Am J Obstet Gynecol. 1996;175:1476-1482.
13. Stanton SL, Hilton P, Norton C, Cardozo L. Clinical and urodynamics effect of anterior colporrhaphy and vaginal hysterectomy for prolapse with and without incontinence. Br J Obstet Gynecol. 1982;89:459-463.
14. Colombo M, Vitobello D, Proiette F, Milani R. Randomized comparison of Burch colposuspension versus anterior colporrhaphy in women with stress urinary incontinence and anterior vaginal wall prolapse. BJOG. 2000;107:544-551.
15. Shull BL, Baden WF. A six-year experience with paravaginal defect repair for stress urinary incontinence. Am J Obstet Gynecol. 1989;160:1432-1440.
16. Shull BL, Benn SJ, Kuehl TJ. Surgical management of prolapse of the anterior vaginal segment: an analysis of support defects, operative morbidity, and anatomic outcome. Am J Obstet Gynecol. 1994;171:1429-1439.
17. Mallipeddi PK, Steele AC, Kohli N, Karram MM. Anatomic and functional outcome of vaginal paravaginal repair in the correction of anterior wall prolapse. Int Urogynecol J. 2001;12:83-88.
18. Young SB, Daman JJ, Bony LG. Vaginal paravaginal repair: one-year outcomes. Am J Obstet Gynecol. 2001;185:1360-1367.
19. Brubaker L. Sacrocolpopexy and the anterior compartment: support and function. Am J Obstet Gynecol. 1995;173:1690-1694.
20. Shull BL, Bachofen C, Coates KW, Kuehl TJ. A transvaginal approach to repair of apical and other associated sites of pelvic organ prolapse with uterosacral ligaments. Am J Obstet Gynecol. 2000;183:1365-1374.
21. Morley GW, DeLancey JOL. Sacrospinous ligament fixation for eversion of the vagina. Am J Obstet Gynecol. 1988;158:872-881.
22. Shull BL, Capen CV, Riggs MW, Kuehl TJ. Preoperative and postoperative analysis of site-specific pelvic support defects in 81 women treated with sacrospinous ligament suspension and pelvic reconstruction. Am J Obstet Gynecol. 1992;166:1764-1771.
23. Paraiso MFR, Ballard LA, Walter MD, et al. Pelvic support defects and visceral and sexual function in women treated with sacrospinous ligament suspension and pelvic reconstruction. Am J Obstet Gynecol. 1996;175:1423-1431.
24. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421.
25. Benson JT, McClellan E. The effect of vaginal dissection of the pudendal nerve. Obstet Gynecol. 1993;82:387-389.
26. Zivkovic F, Tamussino K, Ralph G, et al. Long-term effects of vaginal dissection on the innervation of the striated urethral sphincter. Obstet Gynecol. 1996;87:257-260.
27. Julian T. The efficacy of Marlex mesh in the repair of severe, recurrent vaginal prolapse of the anterior midvaginal wall. Am J Obstet Gynecol. 1996;175:1472-1475.
28. Weber AM, Walters MD, Piedmonte MR, et al. Anterior colporrhaphy: a randomized trial of three surgical techniques. Am J Obstet Gynecol. 2001;185:1299-1306.
29. Sand PK, et al. Prospective randomized trial of polyglactin 910 mesh to prevent recurrence of cystocele and rectocele. Am J Obstet Gynecol. 2001;184:1357-1364.
30. Ellerkmann RM, Cundiff GW, Melick CF, Nihira MA, Leffler K, Bent AE. Correlation of symptoms with location and severity of pelvic organ prolapse. Am J Obstet Gynecol. 2001;185:1332-1337.
31. Hendrix SL, Clark A, Nygaard I, Aragaki A, Barnabei V, McTiernan A. Pelvic organ prolapse in the Women’s Health Initiative: gravity and gravidity. Am J Obstet Gynecol. 2002;186:1160-1166.
32. Handa VL, Garrett E, Hendrix S, Gold E, Robbins J. Progression and remission of pelvic organ prolapse: a longitudinal study of menopausal women. Am J Obstet Gynecol. 2004;190:27-32.
- At this time, the traditional anterior colporrhaphy with attention to apical suspension remains the gold standard.
- If only some defects of the anterior wall are addressed at the time of reconstructive surgery, failure may be more likely.
- Women with grade 3 or 4 cystoceles often have evidence of bladder outlet obstruction on urodynamic testing.
- In 52% of cases, cystoceles coexist with detrusor instability and evidence of impaired detrusor contractility.
- A thorough preoperative evaluation includes assessing the apex, having the patient strain to maximize the defect, looking for paravaginal detachments, and making every effort to “unmask” occult stress urinary incontinence.
Ask a pelvic reconstructive surgeon to name the most difficult challenge, and the answer is likely to be anterior vaginal wall prolapse. The reason: The anterior wall usually is the leading edge of prolapse and the most common site of relaxation or failure following reconstructive surgery. This appears to hold true regardless of surgical route or technique.
Short-term success rates of anterior wall repairs appear promising, but long-term outcomes are not as encouraging. Success usually is claimed as long as the anterior wall is kept above the hymen, since the patient rarely reports symptoms in these cases.
Another challenge involves the use of allografts or xenografts, which have not undergone sufficient study to determine their long-term benefit or risks in comparison with traditional repairs.
This article reviews anatomy of the anterior vaginal wall and its supports, as well as surgical technique and outcomes.
Why the anterior wall is more susceptible to prolapse
One theory is that, in comparison with the posterior compartment, the anterior wall is not as well supported by the levator plate, which counters the effects of gravity and abdominal pressure. Normally, the anterior wall rests horizontally on the posterior wall, which in turn rests on the levator plate. When the levator muscles weaken, the anterior wall is the first to fall as increasing force is placed on the connective tissue supports.
Other possibilities: The anterior compartment’s attachments to the pelvic sidewall or apex may be weaker, the anterior wall may be more elastic or less dense than the posterior wall, and the anterior wall may be more susceptible to damage during childbirth or to the effects of age and loss of estrogen.
If only some defects are addressed at surgery, failure may be more likely. Some experts believe pelvic surgeons have focused too much attention on the urethrovesical junction in patients with concomitant urinary incontinence and not enough attention on suspending the anterior wall at the apex.
For most women, it is probably a combination of many of these factors that renders the anterior compartment so vulnerable.
Anatomy of the pelvic floor
The anterior vaginal wall resembles a trapezoidal plane due to ventral and more medial attachments near the pubic symphysis, and dorsal and more lateral attachments near the ischial spine (FIGURE 1).1 This helps explain the many appearances of the cystocele. The type of cystocele is defined by the location of the break in the fascial attachments.
Paravaginal defects. The trapezoidal anterior wall is suspended on both sides from the parietal fascia overlying the levator ani muscles at the arcus tendineus fascia pelvis (ATFP). Prolapse can occur when there is loss of attachment to the pelvic sidewall at any point between the pubis and ischial spine.
First described by White2 and characterized later by Richardson et al,3 this loss of lateral attachment is called a paravaginal defect or displacement cystocele (FIGURE 2). The goal of paravaginal repair is to reattach the lateral vaginal walls to the ATFP, either abdominally, laparoscopically, or vaginally.
Central defects, the rarest type of anterior wall prolapse, involve a loss of support near the pubis and tend to be smaller. The most common manifestation is urethral hypermobility.
Transverse defects occur when the top of the pubocervical fascia detaches from the cervix or vaginal apex, both of which are suspended from the uterosacral-cardinal ligament complex. A transverse cystocele is evidenced by loss of the anterior fornix. The anterior wall appears to be attenuated in the midline, and the vaginal mucosa is pale, thin, and smooth (FIGURE 3).
Goals of traditional repair. The traditional anterior colporrhaphy aims to excise or reinforce the attenuated transverse defect with plication of the “endopelvic fascia” in the midline of the anterior vaginal wall. The endopelvic fascia is not true fascia but the muscularis of the vaginal wall. It is comprised of smooth muscle and elastin along with the collagenous adventitia layer.4
The importance of restoring apical wall support becomes apparent when one considers the trapezoidal anatomy. The most common sites of defects or detachments of the anterior wall are near the ischial spines laterally. In an operative case series of paravaginal defects, DeLancey1 found the site of defect to be near the ischial spine in 96% of cases. The reattachment of the apex near the level of the spine becomes the highest point of support for the anterior vaginal wall.
This cephalad apical attachment can be accomplished in a variety of ways, by suspending the vaginal apex from the uterosacral ligaments, from the sacrospinous ligament, or via abdominal sacrocolpopexy.
FIGURE 1 Anatomy of the anterior wall
The anterior vaginal wall resembles a trapezoidal plane, with ventral and more medial attachments near the pubic symphysis, and dorsal and more lateral attachments to the ischial spine. Detachment from the pelvic sidewall and ischial spine results in anterior wall prolapse (right).
FIGURE 2 Paravaginal defect
FIGURE 3 Transverse defect
A transverse defect with loss of the anterior fornix. The loss of cephalad apical attachment at the level of the ischial spine leads to anterior wall prolapse. Suspending the upper vagina from shortened cardinal/uterosacral ligaments, the sacrospinous ligament, or via abdominal sacrocolpopexy is as important as plication.
Symptoms of anterior wall prolapse
As with other forms of pelvic organ prolapse, many patients complain of a bulge or feeling of pelvic pressure when the anterior vaginal wall has come through the introitus. However, some symptoms of anterior wall prolapse are unique.
Incontinence is not universal. A common misperception is that most patients with cystocele also experience stress urinary incontinence (SUI), which can develop when there is loss of urethral support and descent of the lower vaginal wall along with urethral hypermobility. However, there is no defining degree of hypermobility that links anterior wall prolapse with SUI. That is because the continence mechanism relies not only on urethral position and lateral attachments, but also on the neuromuscular function of the pelvis and lower urinary tract.
In fact, descent of the midvagina under the bladder base may actually reduce the chance of SUI. The reason: As a woman strains, the increased abdominal pressure pushes the cystocele farther and farther out. As the cystocele enlarges, it creates a functional outlet obstruction by kinking the urethra shut. When this is the case, patients may complain of prolonged voiding, an intermittent urine stream, and/or urinary retention. The woman may have to elevate the vaginal wall to empty her bladder. Patients with chronic urinary retention are at risk of developing recurrent urinary tract infections.
Bladder outlet obstruction and detrusor dysfunction. Women with grade 3 or 4 cystoceles often have evidence of bladder outlet obstruction on urodynamic testing, according to a study that found such evidence in 57% of subjects.5 After reduction of the prolapse with a pessary, obstructed flow reverted to normal in 94% of these women.
A large proportion (52%) of women with cystoceles also have detrusor instability, as well as evidence of impaired detrusor contractility. Many complain of urinary frequency and urgency and difficulty emptying the bladder.5
Again, this phenomenon is complex, related not only to anatomy but to altered neuromuscular function of the lower urinary tract. Incomplete emptying, frequency, and urgency may arise from stretching of the bladder base as it prolapses through the vaginal introitus, resulting in urinary retention. These symptoms often are less pronounced at night when the patient is supine.
We reviewed 35 cases of anterior wall prolapse greater than 1 cm outside the hymen, with elevated postvoid residuals exceeding 100 cc on 2 separate occasions.6 Thirty-one (89%) had normal postvoid residuals following reconstructive surgery and correction of their anterior wall prolapse.
Preoperative assessment
A careful physical exam is a prerequisite for all surgical repairs of pelvic organ prolapse. During this exam, identify the sites of defects and detachments.
Maximize the defect. Have the patient perform the Valsalva maneuver, cough, and/or strain while sitting upright or standing. As she is performing these maneuvers, ask her if this feels like her maximum prolapse. A split speculum often aids in visualizing the anterior and posterior compartments without pressure from the opposite vaginal wall.
Assess the apex. Place a large swab in the vagina, hold it gently against the apex, and ask the patient to strain. If the swab is pushed out, the apex needs suspension.
This technique can help identify apical relaxation that may be masked by a large anterior or posterior wall defect. A standardized staging system, such as the Pelvic Organ Prolapse Quantitative Examination (POP-Q) or Baden-Walker, aids in communicating and documenting the prolapse. In addition, it allows the surgeon to track anatomical outcomes after surgery.
Look for paravaginal defects by supporting the lateral anterior walls with a ring forceps at the level of the ATFP. Barber et al7 found this maneuver to be highly sensitive (90–94%): If no paravaginal defect was suspected clinically, none was found intraoperatively. However, the positive predictive value was poor (57%), in that defects suspected preoperatively were confirmed during surgery in less than two thirds of patients.
These findings point to the importance of careful intraoperative assessment, both before and during the repair procedure.
Limited utility of imaging studies. The use of radiologic studies such as defecography or dynamic magnetic resonance imaging of the pelvis may aid in the evaluation of defecatory disorders or suspected sigmoidocele or rectal prolapse, but have not been studied sufficiently to determine the impact on surgical outcome.
Unmasking SUI
As mentioned above, women with anterior wall prolapse do not always complain of stress incontinence. However, correction of the cystocele can relieve their obstructive voiding and unmask “occult” SUI. Various techniques have been described to elevate the anterior wall with pessaries, swabs, etc, during urodynamic testing to predict which women should have an incontinence procedure performed at the time of reconstructive surgery.
Conflicting rates of occult SUI have been reported, with estimates ranging from 36% to 80%.8 Although preoperative urodynamic testing indicates a high rate of occult stress incontinence, a study by Borstad et al9 suggests that the rate of de novo incontinence may be lower and that preoperative urodynamic findings are not predictive of postoperative continence status. In that study, 16 of 73 women (22%) developed stress incontinence following surgery for prolapse when no incontinence procedure was performed. Advanced age increased the risk of incontinence after surgery.
Contrast these findings with those of Chaikin and colleagues,10 who prospectively followed 24 patients with grade 3 or 4 cystoceles. Preoperative urodynamics showed a 58% rate of occult stress incontinence. All these patients were also defined as having intrinsic sphincter deficiency with leak point pressures below 60 cm water. The incontinent group underwent anterior colporrhaphy and concomitant pubovaginal sling, compared with anterior colporrhaphy alone for those without incontinence. Postoperatively, 2 patients who had the pubovaginal sling procedure reported continued stress incontinence (14%). No new symptoms of incontinence were reported in the patients without leakage on preoperative urodynamics. Thus, preoperative urodynamics were 100% accurate in determining which women did not need additional surgery for SUI.
Implications of a negative stress test. Our experience has shown that, despite our best attempts, a negative stress test with the prolapse reduced prior to surgery is less than 100% predictive. Occasionally, new SUI occurs after reconstructive surgery. It is unclear whether this incontinence is caused by straightening the urethra and reducing the bulge or secondary to the dissection of surgery.
Tips on technique
Anterior colporrhaphy traditionally is performed with plication of the “endopelvic fascia” or fibromuscular layer at the bladder neck with a Kelly plication stitch. Using “3-point” traction aids in dissecting the muscularis (FIGURE 4). Repair the remainder of the cystocele using vertical mattress stitches (1 or 2 layers) from the bladder neck to the apex.
Avoid creating weak areas. Using this technique, the repair frequently stops short of the apex, leaving a “gap” or weak area. One way to avoid this is to begin plication at the apex instead of the bladder neck (FIGURE 5).
Next, excise the excess vaginal tissue and close with interrupted fine absorbable sutures (FIGURE 6).
Recreate apical support. Another problem with traditional repairs is that they do not reestablish apical support. In many patients with anterior wall prolapse, reattachment of the apex reduces the cystocele. Therefore, it often is necessary to combine anterior colporrhaphy with an apical repair procedure such as uterosacral ligament suspension or sacrospinous ligament suspension.
Sutures for the apical repair should be placed and held prior to initiating the anterior colporrhaphy. At the end of the anterior repair, incorporate the apical sutures into the vaginal cuff.
Careful attention to the integrity and strength of the tissue is crucial. Regardless of the type of transvaginal suspension, we advocate bringing 1 arm of the suspension suture through the anterior wall of the cuff. Then place the other suture arm through the posterior cuff so that, when tied, anterior and posterior walls are brought together and suspended.
Using prolonged-delayed absorbable suture allows for a full-thickness bite, ensuring scarring to the suspensory ligament. If permanent suture is used for the uterosacral suspension, place the stitches along the inside surface of the anterior wall with a strong, broad bite that incorporates the muscularis or “endopelvic fascia.”
The occasional enterocele. When a transverse cystocele occurs following hysterectomy, the surgeon should be on the lookout for an enterocele, which sometimes accompanies anterior wall prolapse. The enterocele should be corrected at the time of surgery by closing the defect and suspending the cuff.
FIGURE 4 Three-point traction
Three-point traction using Allis clamps. The assistant retracts with DeBackey forceps to allow dissection of the muscularis. An index finger placed firmly against the vaginal mucosa enables the surgeon to judge depth of dissection.
FIGURE 5 Begin plication at the apex
Plication begins at the apex with vertical mattress stitches. Use 3-0 prolonged delayed absorbable or permanent suture in the anterior wall.
FIGURE 6 The reduced cystocele
The cystocele reduced following midline plication of the vaginal muscularis. The excess vagina is then trimmed and closed with interrupted 3-0 absorbable suture.
Functional surgical outcomes
Because of the long association between anterior wall prolapse and SUI, most surgeons evaluate patients preoperatively to determine the need for concomitant incontinence procedures. As a result, the literature reporting surgical cystocele repair via anterior colporrhaphy frequently uses continence of urine as the functional outcome. This is not surprising considering that anterior repair and Kelly plication, as reported by Howard Kelly more than 75 years ago, have been the gold standard for surgical correction of anterior wall prolapse and SUI.
In a series of 194 SUI patients who underwent anterior colporrhaphy, Beck11 found that adding a modified Kelly plication, including a vaginal retropubic urethropexy, increased the cure rate for SUI from 75% to 94%. Unfortunately, he did not report the anatomic success of the anterior colporrhaphy.
Kohli et al12 also retrospectively examined patients who had undergone anterior colporrhaphy with and without needle bladder-neck suspension. Although the cure rate for SUI was not reported, patients who underwent concomitant needle suspension had a higher rate of recurrent cystocele: 33% (n = 40) versus 7% (n = 27). Investigators theorized that retropubic dissection at the time of transvaginal needle suspension resulted in an iatrogenic paravaginal defect and denervation of the anterior vaginal wall.
The risks of needle suspension. A randomized controlled trial by Bump et al8 also suggests that needle suspension should be avoided. In that trial, 29 patients with stage 3 and 4 prolapse were randomized to needle colposuspension or endopelvic fascia plication. They, too, found that needle colposuspension carried a higher rate of recurrent anterior prolapse. Further, it did not reduce the rates of SUI compared with fascia plication.
Although incontinence surgery performed at the time of cystocele repair will reduce the rates of de novo incontinence, the higher rates of cystocele recurrence associated with some procedures warrants judicious preoperative planning. Clearly, needle suspension should not be performed as an incontinence procedure or repair of anterior wall prolapse. Whether other vaginal incontinence procedures, eg, midurethral slings, lead to recurrence of anterior wall prolapse deserves further investigation.
Anatomic outcomes
Midline colporrhaphy. Because anterior colporrhaphy is rarely performed alone, few series describe patients having undergone simply an anterior repair. Stanton et al13 followed 54 women for up to 2 years after they underwent traditional midline plication with vaginal hysterectomy for prolapse. Eight (15%) of the women had recurrent anterior wall prolapse.
Colombo et al14 randomized 71 women with clinical SUI and stage 2 or 3 prolapse to Burch colposuspension or anterior colporrhaphy with Kelly plication. All women were followed for at least 8 years. The cure rate for SUI was 86% for the Burch procedure, compared with 52% for anterior repair and Kelly plication. However, 12 (34%) women treated with Burch colposuspension and 1 (3%) treated with anterior colporrhaphy had recurrent cystocele of grade 2 or 3 with or without prolapse at other vaginal sites.
Abdominal paravaginal repair. Shull15 followed 149 women for 6 months to 4 years after they underwent abdominal paravaginal repair with the urethrovesical stitch brought through Cooper’s ligament for treatment of SUI and paravaginal cystocele. He reported a 5% recurrence of anterior wall prolapse.
In another series, Shull16 reported on 62 women who were followed for a mean of 18 months after abdominal paravaginal repair. Four of 57 (7%) had recurrent vaginal prolapse to the hymen.
In a cohort of 102 patients evaluated with the Pelvic Organ Prolapse Quantitative Examination (POP-Q) at our institution, the maximal point of prolapse was the anterior wall in 60% of cases; the apex and posterior wall each accounted for roughly half the remaining cases (unpublished data).
Ellerkmann et al30 reported on 237 consecutive patients who presented with symptoms of pelvic organ prolapse. In 77 women (33%), anterior compartment pelvic organ prolapse predominated; 46 patients (19%) had posterior compartment prolapse; and 22 patients (11%) had apical prolapse.
Hendrix et al31 analyzed patients from the Women’s Health Initiative (WHI) and found that the anterior compartment predominated over the posterior compartment. In a follow-up study from the WHI, Handa32 reported on 412 women followed for 2 to 8 years. Among those who entered the WHI protocol without cystocele, 1 in 4 was diagnosed with it at some point in the study. This compares to 1 in 6 for rectocele and 1 in 100 for uterine prolapse. The majority of all defects were grade 1 or relaxation above the hymen.
Vaginal paravaginal repair. Mallipeddi et al17 reported on 45 patients undergoing vaginal paravaginal repair over 2 years, with 35 women followed for a mean of 1.6 years. Incontinent patients had a Kelly plication performed at the time of vaginal paravaginal repair. Recurrence rates were 3% for cystocele, 14% for rectocele, and 20% for enterocele.
Young et al18 followed 100 women for as long as 36 months after bilateral paravaginal repair using 1 to 6 expanded polytetrafluoroethylene (Gore-Tex) CV-0 sutures and midline colporrhaphy. Two patients had grade 1 or 2 failure at the lateral fixation points, but 21 patients had recurrent midline defects, all but 1 inside the hymen. Several patients had bloody discharge from the permanent sutures.
Sacrocolpopexy. Brubaker19 retrospectively reviewed 65 women who underwent sacrocolpopexy for apical prolapse. Three months postoperatively 19 patients (29%) had persistent anterior wall defects.
Uterosacral suspension. Shull et al20 also found the anterior segment to have the most recurrent defects. In that study, which had an average follow-up of a little over a year, 289 patients underwent vaginal uterosacral ligament repair of the apex, and 264 had an anterior wall defect preoperatively. At the time of furthest follow-up, 26 patients (9%) had failure at this site. This study confirmed that the anterior compartment is the most likely site to fail, and also that it fails the quickest.
Sacrospinous ligament suspension (SSLS). Morley and DeLancey21 found a 22% cystocele recurrence rate in 71 women 1 year after SSLS, with most of them asymptomatic. Shull22 reported a 30% incidence of cystoceles after SSLS. Paraiso and colleagues23 reported on 243 women undergoing SSLS and pelvic reconstructive surgery. Of these, 217 patients underwent concomitant anterior colporrhaphy. Follow-up at 74 months found 37% with symptomatic recurrence at the anterior wall, 13% at the posterior wall, and 8% at the apex.
Vaginal versus abdominal repair
Few studies have compared vaginal and abdominal repair of pelvic organ prolapse, including anterior wall prolapse.
In a trial by Benson et al,24 women with prolapse to or beyond the hymen were randomized to bilateral sacrospinous vault suspension and vaginal paravaginal repair (n = 48) or abdominal sacrocolpopexy with abdominal paravaginal repair (n = 40). One third of patients in each group also underwent anterior colporrhaphy. After a mean follow-up of 2.5 years, 16 of 20 women required reoperation for recurrent cystocele—12 (29%) from the vaginal group and 4 (10.5%) from the abdominal group. Vaginal vault eversion recurred in 5 women from the vaginal group and 1 from the abdominal group.
Investigators concluded that the anterior wall was the most likely site of failure because of the posterior placement of the vaginal apex with SSLS, predisposing the anterior wall to greater pressures and to neuropathy caused by lateral dissection of the anterior wall. Earlier studies have demonstrated that neuropathy may occur after extensive dissection of the vaginal wall and may affect the strength and integrity of the muscular support tissues.25,26
Allografts and xenografts
The difficulty of repairing anterior wall prolapse has led some pelvic surgeons to use mesh for cystocele repair. When Julian27 randomized 24 patients with recurrent cystocele to transvaginal repair with and without polypropylene (Marlex) mesh, 4 patients in the control group and no patients in the mesh group had recurrences (P <.05). However, 3 patients (25%) had mesh-related complications.
Weber et al28 randomized patients to standard midline plication, plication of the paravaginal tissue more laterally, or standard plication plus polyglactin 910 (Vicryl) mesh. Among 83 patients who returned for follow-up, there were no differences in anatomic outcome. Weber and colleagues concluded that there is little benefit to using mesh to correct cystoceles.
Still, although the overall cure rate was low (30–46%), most patients had cystocele to the hymen and not beyond, with significant improvement of symptoms. Although this cannot be defined as an anatomic cure, it is encouraging that the majority of patients appear to have benefited from surgery.
Sand et al29 randomized 161 women with the anterior wall to or beyond the hymen to traditional anterior colporrhaphy with or without Vicryl. The 2-inch square mesh was not placed over the repair as described above, but was folded into the anterior colporrhaphy stitches. At 1 year, 16 (22%) of 73 women with mesh and 28 (40%) of the 88 women without mesh had recurrent central cystoceles beyond the midvagina (P = .02). No women had cystoceles beyond the hymen or vaginal erosions.
Difficulty of interpreting the evidence
Because of the broad range of study designs, small number of patients per series, variety of concomitant procedures, and wide range of variables used to describe recurrence and success, it is difficult to draw conclusions from the literature. The evidence does suggest that the risks of wide vaginal dissection required for vaginal paravaginal repair outweigh the benefits. As a result, we have abandoned this technique. As mentioned above, it remains unclear whether graft materials will prove to be of long-term benefit for either midline plication or paravaginal repair.
The gold standard, for now
Prolapse of the anterior vaginal wall remains a challenge for the gynecologic surgeon. Careful preoperative and intraoperative evaluation and identification of support defects should guide repairs.
Randomized, controlled trials of midline versus paravaginal repair, as well as use of various graft materials, are greatly needed. These studies should not only address recurrence of prolapse symptoms, but the impact of surgery on sexual and lower urinary tract function.
At this time, the traditional anterior colporrhaphy with attention to apical suspension remains the gold standard.
The authors report no financial relationships relevant to this article.
- At this time, the traditional anterior colporrhaphy with attention to apical suspension remains the gold standard.
- If only some defects of the anterior wall are addressed at the time of reconstructive surgery, failure may be more likely.
- Women with grade 3 or 4 cystoceles often have evidence of bladder outlet obstruction on urodynamic testing.
- In 52% of cases, cystoceles coexist with detrusor instability and evidence of impaired detrusor contractility.
- A thorough preoperative evaluation includes assessing the apex, having the patient strain to maximize the defect, looking for paravaginal detachments, and making every effort to “unmask” occult stress urinary incontinence.
Ask a pelvic reconstructive surgeon to name the most difficult challenge, and the answer is likely to be anterior vaginal wall prolapse. The reason: The anterior wall usually is the leading edge of prolapse and the most common site of relaxation or failure following reconstructive surgery. This appears to hold true regardless of surgical route or technique.
Short-term success rates of anterior wall repairs appear promising, but long-term outcomes are not as encouraging. Success usually is claimed as long as the anterior wall is kept above the hymen, since the patient rarely reports symptoms in these cases.
Another challenge involves the use of allografts or xenografts, which have not undergone sufficient study to determine their long-term benefit or risks in comparison with traditional repairs.
This article reviews anatomy of the anterior vaginal wall and its supports, as well as surgical technique and outcomes.
Why the anterior wall is more susceptible to prolapse
One theory is that, in comparison with the posterior compartment, the anterior wall is not as well supported by the levator plate, which counters the effects of gravity and abdominal pressure. Normally, the anterior wall rests horizontally on the posterior wall, which in turn rests on the levator plate. When the levator muscles weaken, the anterior wall is the first to fall as increasing force is placed on the connective tissue supports.
Other possibilities: The anterior compartment’s attachments to the pelvic sidewall or apex may be weaker, the anterior wall may be more elastic or less dense than the posterior wall, and the anterior wall may be more susceptible to damage during childbirth or to the effects of age and loss of estrogen.
If only some defects are addressed at surgery, failure may be more likely. Some experts believe pelvic surgeons have focused too much attention on the urethrovesical junction in patients with concomitant urinary incontinence and not enough attention on suspending the anterior wall at the apex.
For most women, it is probably a combination of many of these factors that renders the anterior compartment so vulnerable.
Anatomy of the pelvic floor
The anterior vaginal wall resembles a trapezoidal plane due to ventral and more medial attachments near the pubic symphysis, and dorsal and more lateral attachments near the ischial spine (FIGURE 1).1 This helps explain the many appearances of the cystocele. The type of cystocele is defined by the location of the break in the fascial attachments.
Paravaginal defects. The trapezoidal anterior wall is suspended on both sides from the parietal fascia overlying the levator ani muscles at the arcus tendineus fascia pelvis (ATFP). Prolapse can occur when there is loss of attachment to the pelvic sidewall at any point between the pubis and ischial spine.
First described by White2 and characterized later by Richardson et al,3 this loss of lateral attachment is called a paravaginal defect or displacement cystocele (FIGURE 2). The goal of paravaginal repair is to reattach the lateral vaginal walls to the ATFP, either abdominally, laparoscopically, or vaginally.
Central defects, the rarest type of anterior wall prolapse, involve a loss of support near the pubis and tend to be smaller. The most common manifestation is urethral hypermobility.
Transverse defects occur when the top of the pubocervical fascia detaches from the cervix or vaginal apex, both of which are suspended from the uterosacral-cardinal ligament complex. A transverse cystocele is evidenced by loss of the anterior fornix. The anterior wall appears to be attenuated in the midline, and the vaginal mucosa is pale, thin, and smooth (FIGURE 3).
Goals of traditional repair. The traditional anterior colporrhaphy aims to excise or reinforce the attenuated transverse defect with plication of the “endopelvic fascia” in the midline of the anterior vaginal wall. The endopelvic fascia is not true fascia but the muscularis of the vaginal wall. It is comprised of smooth muscle and elastin along with the collagenous adventitia layer.4
The importance of restoring apical wall support becomes apparent when one considers the trapezoidal anatomy. The most common sites of defects or detachments of the anterior wall are near the ischial spines laterally. In an operative case series of paravaginal defects, DeLancey1 found the site of defect to be near the ischial spine in 96% of cases. The reattachment of the apex near the level of the spine becomes the highest point of support for the anterior vaginal wall.
This cephalad apical attachment can be accomplished in a variety of ways, by suspending the vaginal apex from the uterosacral ligaments, from the sacrospinous ligament, or via abdominal sacrocolpopexy.
FIGURE 1 Anatomy of the anterior wall
The anterior vaginal wall resembles a trapezoidal plane, with ventral and more medial attachments near the pubic symphysis, and dorsal and more lateral attachments to the ischial spine. Detachment from the pelvic sidewall and ischial spine results in anterior wall prolapse (right).
FIGURE 2 Paravaginal defect
FIGURE 3 Transverse defect
A transverse defect with loss of the anterior fornix. The loss of cephalad apical attachment at the level of the ischial spine leads to anterior wall prolapse. Suspending the upper vagina from shortened cardinal/uterosacral ligaments, the sacrospinous ligament, or via abdominal sacrocolpopexy is as important as plication.
Symptoms of anterior wall prolapse
As with other forms of pelvic organ prolapse, many patients complain of a bulge or feeling of pelvic pressure when the anterior vaginal wall has come through the introitus. However, some symptoms of anterior wall prolapse are unique.
Incontinence is not universal. A common misperception is that most patients with cystocele also experience stress urinary incontinence (SUI), which can develop when there is loss of urethral support and descent of the lower vaginal wall along with urethral hypermobility. However, there is no defining degree of hypermobility that links anterior wall prolapse with SUI. That is because the continence mechanism relies not only on urethral position and lateral attachments, but also on the neuromuscular function of the pelvis and lower urinary tract.
In fact, descent of the midvagina under the bladder base may actually reduce the chance of SUI. The reason: As a woman strains, the increased abdominal pressure pushes the cystocele farther and farther out. As the cystocele enlarges, it creates a functional outlet obstruction by kinking the urethra shut. When this is the case, patients may complain of prolonged voiding, an intermittent urine stream, and/or urinary retention. The woman may have to elevate the vaginal wall to empty her bladder. Patients with chronic urinary retention are at risk of developing recurrent urinary tract infections.
Bladder outlet obstruction and detrusor dysfunction. Women with grade 3 or 4 cystoceles often have evidence of bladder outlet obstruction on urodynamic testing, according to a study that found such evidence in 57% of subjects.5 After reduction of the prolapse with a pessary, obstructed flow reverted to normal in 94% of these women.
A large proportion (52%) of women with cystoceles also have detrusor instability, as well as evidence of impaired detrusor contractility. Many complain of urinary frequency and urgency and difficulty emptying the bladder.5
Again, this phenomenon is complex, related not only to anatomy but to altered neuromuscular function of the lower urinary tract. Incomplete emptying, frequency, and urgency may arise from stretching of the bladder base as it prolapses through the vaginal introitus, resulting in urinary retention. These symptoms often are less pronounced at night when the patient is supine.
We reviewed 35 cases of anterior wall prolapse greater than 1 cm outside the hymen, with elevated postvoid residuals exceeding 100 cc on 2 separate occasions.6 Thirty-one (89%) had normal postvoid residuals following reconstructive surgery and correction of their anterior wall prolapse.
Preoperative assessment
A careful physical exam is a prerequisite for all surgical repairs of pelvic organ prolapse. During this exam, identify the sites of defects and detachments.
Maximize the defect. Have the patient perform the Valsalva maneuver, cough, and/or strain while sitting upright or standing. As she is performing these maneuvers, ask her if this feels like her maximum prolapse. A split speculum often aids in visualizing the anterior and posterior compartments without pressure from the opposite vaginal wall.
Assess the apex. Place a large swab in the vagina, hold it gently against the apex, and ask the patient to strain. If the swab is pushed out, the apex needs suspension.
This technique can help identify apical relaxation that may be masked by a large anterior or posterior wall defect. A standardized staging system, such as the Pelvic Organ Prolapse Quantitative Examination (POP-Q) or Baden-Walker, aids in communicating and documenting the prolapse. In addition, it allows the surgeon to track anatomical outcomes after surgery.
Look for paravaginal defects by supporting the lateral anterior walls with a ring forceps at the level of the ATFP. Barber et al7 found this maneuver to be highly sensitive (90–94%): If no paravaginal defect was suspected clinically, none was found intraoperatively. However, the positive predictive value was poor (57%), in that defects suspected preoperatively were confirmed during surgery in less than two thirds of patients.
These findings point to the importance of careful intraoperative assessment, both before and during the repair procedure.
Limited utility of imaging studies. The use of radiologic studies such as defecography or dynamic magnetic resonance imaging of the pelvis may aid in the evaluation of defecatory disorders or suspected sigmoidocele or rectal prolapse, but have not been studied sufficiently to determine the impact on surgical outcome.
Unmasking SUI
As mentioned above, women with anterior wall prolapse do not always complain of stress incontinence. However, correction of the cystocele can relieve their obstructive voiding and unmask “occult” SUI. Various techniques have been described to elevate the anterior wall with pessaries, swabs, etc, during urodynamic testing to predict which women should have an incontinence procedure performed at the time of reconstructive surgery.
Conflicting rates of occult SUI have been reported, with estimates ranging from 36% to 80%.8 Although preoperative urodynamic testing indicates a high rate of occult stress incontinence, a study by Borstad et al9 suggests that the rate of de novo incontinence may be lower and that preoperative urodynamic findings are not predictive of postoperative continence status. In that study, 16 of 73 women (22%) developed stress incontinence following surgery for prolapse when no incontinence procedure was performed. Advanced age increased the risk of incontinence after surgery.
Contrast these findings with those of Chaikin and colleagues,10 who prospectively followed 24 patients with grade 3 or 4 cystoceles. Preoperative urodynamics showed a 58% rate of occult stress incontinence. All these patients were also defined as having intrinsic sphincter deficiency with leak point pressures below 60 cm water. The incontinent group underwent anterior colporrhaphy and concomitant pubovaginal sling, compared with anterior colporrhaphy alone for those without incontinence. Postoperatively, 2 patients who had the pubovaginal sling procedure reported continued stress incontinence (14%). No new symptoms of incontinence were reported in the patients without leakage on preoperative urodynamics. Thus, preoperative urodynamics were 100% accurate in determining which women did not need additional surgery for SUI.
Implications of a negative stress test. Our experience has shown that, despite our best attempts, a negative stress test with the prolapse reduced prior to surgery is less than 100% predictive. Occasionally, new SUI occurs after reconstructive surgery. It is unclear whether this incontinence is caused by straightening the urethra and reducing the bulge or secondary to the dissection of surgery.
Tips on technique
Anterior colporrhaphy traditionally is performed with plication of the “endopelvic fascia” or fibromuscular layer at the bladder neck with a Kelly plication stitch. Using “3-point” traction aids in dissecting the muscularis (FIGURE 4). Repair the remainder of the cystocele using vertical mattress stitches (1 or 2 layers) from the bladder neck to the apex.
Avoid creating weak areas. Using this technique, the repair frequently stops short of the apex, leaving a “gap” or weak area. One way to avoid this is to begin plication at the apex instead of the bladder neck (FIGURE 5).
Next, excise the excess vaginal tissue and close with interrupted fine absorbable sutures (FIGURE 6).
Recreate apical support. Another problem with traditional repairs is that they do not reestablish apical support. In many patients with anterior wall prolapse, reattachment of the apex reduces the cystocele. Therefore, it often is necessary to combine anterior colporrhaphy with an apical repair procedure such as uterosacral ligament suspension or sacrospinous ligament suspension.
Sutures for the apical repair should be placed and held prior to initiating the anterior colporrhaphy. At the end of the anterior repair, incorporate the apical sutures into the vaginal cuff.
Careful attention to the integrity and strength of the tissue is crucial. Regardless of the type of transvaginal suspension, we advocate bringing 1 arm of the suspension suture through the anterior wall of the cuff. Then place the other suture arm through the posterior cuff so that, when tied, anterior and posterior walls are brought together and suspended.
Using prolonged-delayed absorbable suture allows for a full-thickness bite, ensuring scarring to the suspensory ligament. If permanent suture is used for the uterosacral suspension, place the stitches along the inside surface of the anterior wall with a strong, broad bite that incorporates the muscularis or “endopelvic fascia.”
The occasional enterocele. When a transverse cystocele occurs following hysterectomy, the surgeon should be on the lookout for an enterocele, which sometimes accompanies anterior wall prolapse. The enterocele should be corrected at the time of surgery by closing the defect and suspending the cuff.
FIGURE 4 Three-point traction
Three-point traction using Allis clamps. The assistant retracts with DeBackey forceps to allow dissection of the muscularis. An index finger placed firmly against the vaginal mucosa enables the surgeon to judge depth of dissection.
FIGURE 5 Begin plication at the apex
Plication begins at the apex with vertical mattress stitches. Use 3-0 prolonged delayed absorbable or permanent suture in the anterior wall.
FIGURE 6 The reduced cystocele
The cystocele reduced following midline plication of the vaginal muscularis. The excess vagina is then trimmed and closed with interrupted 3-0 absorbable suture.
Functional surgical outcomes
Because of the long association between anterior wall prolapse and SUI, most surgeons evaluate patients preoperatively to determine the need for concomitant incontinence procedures. As a result, the literature reporting surgical cystocele repair via anterior colporrhaphy frequently uses continence of urine as the functional outcome. This is not surprising considering that anterior repair and Kelly plication, as reported by Howard Kelly more than 75 years ago, have been the gold standard for surgical correction of anterior wall prolapse and SUI.
In a series of 194 SUI patients who underwent anterior colporrhaphy, Beck11 found that adding a modified Kelly plication, including a vaginal retropubic urethropexy, increased the cure rate for SUI from 75% to 94%. Unfortunately, he did not report the anatomic success of the anterior colporrhaphy.
Kohli et al12 also retrospectively examined patients who had undergone anterior colporrhaphy with and without needle bladder-neck suspension. Although the cure rate for SUI was not reported, patients who underwent concomitant needle suspension had a higher rate of recurrent cystocele: 33% (n = 40) versus 7% (n = 27). Investigators theorized that retropubic dissection at the time of transvaginal needle suspension resulted in an iatrogenic paravaginal defect and denervation of the anterior vaginal wall.
The risks of needle suspension. A randomized controlled trial by Bump et al8 also suggests that needle suspension should be avoided. In that trial, 29 patients with stage 3 and 4 prolapse were randomized to needle colposuspension or endopelvic fascia plication. They, too, found that needle colposuspension carried a higher rate of recurrent anterior prolapse. Further, it did not reduce the rates of SUI compared with fascia plication.
Although incontinence surgery performed at the time of cystocele repair will reduce the rates of de novo incontinence, the higher rates of cystocele recurrence associated with some procedures warrants judicious preoperative planning. Clearly, needle suspension should not be performed as an incontinence procedure or repair of anterior wall prolapse. Whether other vaginal incontinence procedures, eg, midurethral slings, lead to recurrence of anterior wall prolapse deserves further investigation.
Anatomic outcomes
Midline colporrhaphy. Because anterior colporrhaphy is rarely performed alone, few series describe patients having undergone simply an anterior repair. Stanton et al13 followed 54 women for up to 2 years after they underwent traditional midline plication with vaginal hysterectomy for prolapse. Eight (15%) of the women had recurrent anterior wall prolapse.
Colombo et al14 randomized 71 women with clinical SUI and stage 2 or 3 prolapse to Burch colposuspension or anterior colporrhaphy with Kelly plication. All women were followed for at least 8 years. The cure rate for SUI was 86% for the Burch procedure, compared with 52% for anterior repair and Kelly plication. However, 12 (34%) women treated with Burch colposuspension and 1 (3%) treated with anterior colporrhaphy had recurrent cystocele of grade 2 or 3 with or without prolapse at other vaginal sites.
Abdominal paravaginal repair. Shull15 followed 149 women for 6 months to 4 years after they underwent abdominal paravaginal repair with the urethrovesical stitch brought through Cooper’s ligament for treatment of SUI and paravaginal cystocele. He reported a 5% recurrence of anterior wall prolapse.
In another series, Shull16 reported on 62 women who were followed for a mean of 18 months after abdominal paravaginal repair. Four of 57 (7%) had recurrent vaginal prolapse to the hymen.
In a cohort of 102 patients evaluated with the Pelvic Organ Prolapse Quantitative Examination (POP-Q) at our institution, the maximal point of prolapse was the anterior wall in 60% of cases; the apex and posterior wall each accounted for roughly half the remaining cases (unpublished data).
Ellerkmann et al30 reported on 237 consecutive patients who presented with symptoms of pelvic organ prolapse. In 77 women (33%), anterior compartment pelvic organ prolapse predominated; 46 patients (19%) had posterior compartment prolapse; and 22 patients (11%) had apical prolapse.
Hendrix et al31 analyzed patients from the Women’s Health Initiative (WHI) and found that the anterior compartment predominated over the posterior compartment. In a follow-up study from the WHI, Handa32 reported on 412 women followed for 2 to 8 years. Among those who entered the WHI protocol without cystocele, 1 in 4 was diagnosed with it at some point in the study. This compares to 1 in 6 for rectocele and 1 in 100 for uterine prolapse. The majority of all defects were grade 1 or relaxation above the hymen.
Vaginal paravaginal repair. Mallipeddi et al17 reported on 45 patients undergoing vaginal paravaginal repair over 2 years, with 35 women followed for a mean of 1.6 years. Incontinent patients had a Kelly plication performed at the time of vaginal paravaginal repair. Recurrence rates were 3% for cystocele, 14% for rectocele, and 20% for enterocele.
Young et al18 followed 100 women for as long as 36 months after bilateral paravaginal repair using 1 to 6 expanded polytetrafluoroethylene (Gore-Tex) CV-0 sutures and midline colporrhaphy. Two patients had grade 1 or 2 failure at the lateral fixation points, but 21 patients had recurrent midline defects, all but 1 inside the hymen. Several patients had bloody discharge from the permanent sutures.
Sacrocolpopexy. Brubaker19 retrospectively reviewed 65 women who underwent sacrocolpopexy for apical prolapse. Three months postoperatively 19 patients (29%) had persistent anterior wall defects.
Uterosacral suspension. Shull et al20 also found the anterior segment to have the most recurrent defects. In that study, which had an average follow-up of a little over a year, 289 patients underwent vaginal uterosacral ligament repair of the apex, and 264 had an anterior wall defect preoperatively. At the time of furthest follow-up, 26 patients (9%) had failure at this site. This study confirmed that the anterior compartment is the most likely site to fail, and also that it fails the quickest.
Sacrospinous ligament suspension (SSLS). Morley and DeLancey21 found a 22% cystocele recurrence rate in 71 women 1 year after SSLS, with most of them asymptomatic. Shull22 reported a 30% incidence of cystoceles after SSLS. Paraiso and colleagues23 reported on 243 women undergoing SSLS and pelvic reconstructive surgery. Of these, 217 patients underwent concomitant anterior colporrhaphy. Follow-up at 74 months found 37% with symptomatic recurrence at the anterior wall, 13% at the posterior wall, and 8% at the apex.
Vaginal versus abdominal repair
Few studies have compared vaginal and abdominal repair of pelvic organ prolapse, including anterior wall prolapse.
In a trial by Benson et al,24 women with prolapse to or beyond the hymen were randomized to bilateral sacrospinous vault suspension and vaginal paravaginal repair (n = 48) or abdominal sacrocolpopexy with abdominal paravaginal repair (n = 40). One third of patients in each group also underwent anterior colporrhaphy. After a mean follow-up of 2.5 years, 16 of 20 women required reoperation for recurrent cystocele—12 (29%) from the vaginal group and 4 (10.5%) from the abdominal group. Vaginal vault eversion recurred in 5 women from the vaginal group and 1 from the abdominal group.
Investigators concluded that the anterior wall was the most likely site of failure because of the posterior placement of the vaginal apex with SSLS, predisposing the anterior wall to greater pressures and to neuropathy caused by lateral dissection of the anterior wall. Earlier studies have demonstrated that neuropathy may occur after extensive dissection of the vaginal wall and may affect the strength and integrity of the muscular support tissues.25,26
Allografts and xenografts
The difficulty of repairing anterior wall prolapse has led some pelvic surgeons to use mesh for cystocele repair. When Julian27 randomized 24 patients with recurrent cystocele to transvaginal repair with and without polypropylene (Marlex) mesh, 4 patients in the control group and no patients in the mesh group had recurrences (P <.05). However, 3 patients (25%) had mesh-related complications.
Weber et al28 randomized patients to standard midline plication, plication of the paravaginal tissue more laterally, or standard plication plus polyglactin 910 (Vicryl) mesh. Among 83 patients who returned for follow-up, there were no differences in anatomic outcome. Weber and colleagues concluded that there is little benefit to using mesh to correct cystoceles.
Still, although the overall cure rate was low (30–46%), most patients had cystocele to the hymen and not beyond, with significant improvement of symptoms. Although this cannot be defined as an anatomic cure, it is encouraging that the majority of patients appear to have benefited from surgery.
Sand et al29 randomized 161 women with the anterior wall to or beyond the hymen to traditional anterior colporrhaphy with or without Vicryl. The 2-inch square mesh was not placed over the repair as described above, but was folded into the anterior colporrhaphy stitches. At 1 year, 16 (22%) of 73 women with mesh and 28 (40%) of the 88 women without mesh had recurrent central cystoceles beyond the midvagina (P = .02). No women had cystoceles beyond the hymen or vaginal erosions.
Difficulty of interpreting the evidence
Because of the broad range of study designs, small number of patients per series, variety of concomitant procedures, and wide range of variables used to describe recurrence and success, it is difficult to draw conclusions from the literature. The evidence does suggest that the risks of wide vaginal dissection required for vaginal paravaginal repair outweigh the benefits. As a result, we have abandoned this technique. As mentioned above, it remains unclear whether graft materials will prove to be of long-term benefit for either midline plication or paravaginal repair.
The gold standard, for now
Prolapse of the anterior vaginal wall remains a challenge for the gynecologic surgeon. Careful preoperative and intraoperative evaluation and identification of support defects should guide repairs.
Randomized, controlled trials of midline versus paravaginal repair, as well as use of various graft materials, are greatly needed. These studies should not only address recurrence of prolapse symptoms, but the impact of surgery on sexual and lower urinary tract function.
At this time, the traditional anterior colporrhaphy with attention to apical suspension remains the gold standard.
The authors report no financial relationships relevant to this article.
1. DeLancey JOL. Fascial and muscular abnormalities in women with urethral hypermobility and anterior vaginal wall prolapse. Am J Obstet Gynecol. 2002;187:93-98.
2. White GR. Cystocele: a radical cure by suturing lateral sulci of vagina to white line of pelvic fascia. JAMA. 1909;21:1707-1710.
3. Richardson AC, Lyon JB, Williams NL. A new look at pelvic relaxation. Am J Obstet Gynecol. 1976;126:568-571.
4. Weber AM, Walters MD. Anterior vaginal prolapse: review of anatomy and techniques of surgical repair. Obstet Gynecol. 1997;89:311-318.
5. Romanzi LJ, Chaikin DC, Blaivas JG. The effect of genital prolapse on voiding. J Urol. 1999;161:581-586.
6. FitzGerald MP, Kulkarni N, Fenner D. Postoperative resolution of urinary retention in patients with advanced pelvic organ prolapse. Am J Obstet Gynecol. 2000;183:1361-1364.
7. Barber MD, Cundiff GW, Weidner AC, Cotes KW, Bump RC, Addison WA. Accuracy of clinical assessment of paravaginal defects in women with anterior vaginal wall proloapse. Am J Obstet Gynecol. 1999;181:1-7.
8. Bump RC, Hurt WG, Theofrastous JP, et al. Randomized prospective comparison of needle colposuspension versus endopelvic fascia plication for potential stress incontinence prophylaxis in women undergoing vaginal reconstruction for stage III or IV pelvic organ prolapse. The Continence Program for Women Research Group. Am J Obstet Gynecol. 1996;175:326-333.
9. Borstad E, Rud T. The risk of developing urinary stress incontinence after vaginal repair in continent women. Acta Obstet Gynecol Scand. 1989;68:545-549.
10. Chaikin DC, Groutz A, Blaivas JG. Predicting the need for anti-incontinence surgery in continent women undergoing repair of severe urogenital prolapse. J Urol. 2000;163:531-534.
11. Beck RP, McCormick S, Nordstrom L. A 25-year experience with 519 anterior colporrhaphy procedures. Obstet Gynecol. 1991;78:1011-1018.
12. Kohli N, Sze EHM, Roat TW, Karram M. Incidence of recurrent cystocele after anterior colporrhaphy with and without concomitant transvaginal needle suspension. Am J Obstet Gynecol. 1996;175:1476-1482.
13. Stanton SL, Hilton P, Norton C, Cardozo L. Clinical and urodynamics effect of anterior colporrhaphy and vaginal hysterectomy for prolapse with and without incontinence. Br J Obstet Gynecol. 1982;89:459-463.
14. Colombo M, Vitobello D, Proiette F, Milani R. Randomized comparison of Burch colposuspension versus anterior colporrhaphy in women with stress urinary incontinence and anterior vaginal wall prolapse. BJOG. 2000;107:544-551.
15. Shull BL, Baden WF. A six-year experience with paravaginal defect repair for stress urinary incontinence. Am J Obstet Gynecol. 1989;160:1432-1440.
16. Shull BL, Benn SJ, Kuehl TJ. Surgical management of prolapse of the anterior vaginal segment: an analysis of support defects, operative morbidity, and anatomic outcome. Am J Obstet Gynecol. 1994;171:1429-1439.
17. Mallipeddi PK, Steele AC, Kohli N, Karram MM. Anatomic and functional outcome of vaginal paravaginal repair in the correction of anterior wall prolapse. Int Urogynecol J. 2001;12:83-88.
18. Young SB, Daman JJ, Bony LG. Vaginal paravaginal repair: one-year outcomes. Am J Obstet Gynecol. 2001;185:1360-1367.
19. Brubaker L. Sacrocolpopexy and the anterior compartment: support and function. Am J Obstet Gynecol. 1995;173:1690-1694.
20. Shull BL, Bachofen C, Coates KW, Kuehl TJ. A transvaginal approach to repair of apical and other associated sites of pelvic organ prolapse with uterosacral ligaments. Am J Obstet Gynecol. 2000;183:1365-1374.
21. Morley GW, DeLancey JOL. Sacrospinous ligament fixation for eversion of the vagina. Am J Obstet Gynecol. 1988;158:872-881.
22. Shull BL, Capen CV, Riggs MW, Kuehl TJ. Preoperative and postoperative analysis of site-specific pelvic support defects in 81 women treated with sacrospinous ligament suspension and pelvic reconstruction. Am J Obstet Gynecol. 1992;166:1764-1771.
23. Paraiso MFR, Ballard LA, Walter MD, et al. Pelvic support defects and visceral and sexual function in women treated with sacrospinous ligament suspension and pelvic reconstruction. Am J Obstet Gynecol. 1996;175:1423-1431.
24. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421.
25. Benson JT, McClellan E. The effect of vaginal dissection of the pudendal nerve. Obstet Gynecol. 1993;82:387-389.
26. Zivkovic F, Tamussino K, Ralph G, et al. Long-term effects of vaginal dissection on the innervation of the striated urethral sphincter. Obstet Gynecol. 1996;87:257-260.
27. Julian T. The efficacy of Marlex mesh in the repair of severe, recurrent vaginal prolapse of the anterior midvaginal wall. Am J Obstet Gynecol. 1996;175:1472-1475.
28. Weber AM, Walters MD, Piedmonte MR, et al. Anterior colporrhaphy: a randomized trial of three surgical techniques. Am J Obstet Gynecol. 2001;185:1299-1306.
29. Sand PK, et al. Prospective randomized trial of polyglactin 910 mesh to prevent recurrence of cystocele and rectocele. Am J Obstet Gynecol. 2001;184:1357-1364.
30. Ellerkmann RM, Cundiff GW, Melick CF, Nihira MA, Leffler K, Bent AE. Correlation of symptoms with location and severity of pelvic organ prolapse. Am J Obstet Gynecol. 2001;185:1332-1337.
31. Hendrix SL, Clark A, Nygaard I, Aragaki A, Barnabei V, McTiernan A. Pelvic organ prolapse in the Women’s Health Initiative: gravity and gravidity. Am J Obstet Gynecol. 2002;186:1160-1166.
32. Handa VL, Garrett E, Hendrix S, Gold E, Robbins J. Progression and remission of pelvic organ prolapse: a longitudinal study of menopausal women. Am J Obstet Gynecol. 2004;190:27-32.
1. DeLancey JOL. Fascial and muscular abnormalities in women with urethral hypermobility and anterior vaginal wall prolapse. Am J Obstet Gynecol. 2002;187:93-98.
2. White GR. Cystocele: a radical cure by suturing lateral sulci of vagina to white line of pelvic fascia. JAMA. 1909;21:1707-1710.
3. Richardson AC, Lyon JB, Williams NL. A new look at pelvic relaxation. Am J Obstet Gynecol. 1976;126:568-571.
4. Weber AM, Walters MD. Anterior vaginal prolapse: review of anatomy and techniques of surgical repair. Obstet Gynecol. 1997;89:311-318.
5. Romanzi LJ, Chaikin DC, Blaivas JG. The effect of genital prolapse on voiding. J Urol. 1999;161:581-586.
6. FitzGerald MP, Kulkarni N, Fenner D. Postoperative resolution of urinary retention in patients with advanced pelvic organ prolapse. Am J Obstet Gynecol. 2000;183:1361-1364.
7. Barber MD, Cundiff GW, Weidner AC, Cotes KW, Bump RC, Addison WA. Accuracy of clinical assessment of paravaginal defects in women with anterior vaginal wall proloapse. Am J Obstet Gynecol. 1999;181:1-7.
8. Bump RC, Hurt WG, Theofrastous JP, et al. Randomized prospective comparison of needle colposuspension versus endopelvic fascia plication for potential stress incontinence prophylaxis in women undergoing vaginal reconstruction for stage III or IV pelvic organ prolapse. The Continence Program for Women Research Group. Am J Obstet Gynecol. 1996;175:326-333.
9. Borstad E, Rud T. The risk of developing urinary stress incontinence after vaginal repair in continent women. Acta Obstet Gynecol Scand. 1989;68:545-549.
10. Chaikin DC, Groutz A, Blaivas JG. Predicting the need for anti-incontinence surgery in continent women undergoing repair of severe urogenital prolapse. J Urol. 2000;163:531-534.
11. Beck RP, McCormick S, Nordstrom L. A 25-year experience with 519 anterior colporrhaphy procedures. Obstet Gynecol. 1991;78:1011-1018.
12. Kohli N, Sze EHM, Roat TW, Karram M. Incidence of recurrent cystocele after anterior colporrhaphy with and without concomitant transvaginal needle suspension. Am J Obstet Gynecol. 1996;175:1476-1482.
13. Stanton SL, Hilton P, Norton C, Cardozo L. Clinical and urodynamics effect of anterior colporrhaphy and vaginal hysterectomy for prolapse with and without incontinence. Br J Obstet Gynecol. 1982;89:459-463.
14. Colombo M, Vitobello D, Proiette F, Milani R. Randomized comparison of Burch colposuspension versus anterior colporrhaphy in women with stress urinary incontinence and anterior vaginal wall prolapse. BJOG. 2000;107:544-551.
15. Shull BL, Baden WF. A six-year experience with paravaginal defect repair for stress urinary incontinence. Am J Obstet Gynecol. 1989;160:1432-1440.
16. Shull BL, Benn SJ, Kuehl TJ. Surgical management of prolapse of the anterior vaginal segment: an analysis of support defects, operative morbidity, and anatomic outcome. Am J Obstet Gynecol. 1994;171:1429-1439.
17. Mallipeddi PK, Steele AC, Kohli N, Karram MM. Anatomic and functional outcome of vaginal paravaginal repair in the correction of anterior wall prolapse. Int Urogynecol J. 2001;12:83-88.
18. Young SB, Daman JJ, Bony LG. Vaginal paravaginal repair: one-year outcomes. Am J Obstet Gynecol. 2001;185:1360-1367.
19. Brubaker L. Sacrocolpopexy and the anterior compartment: support and function. Am J Obstet Gynecol. 1995;173:1690-1694.
20. Shull BL, Bachofen C, Coates KW, Kuehl TJ. A transvaginal approach to repair of apical and other associated sites of pelvic organ prolapse with uterosacral ligaments. Am J Obstet Gynecol. 2000;183:1365-1374.
21. Morley GW, DeLancey JOL. Sacrospinous ligament fixation for eversion of the vagina. Am J Obstet Gynecol. 1988;158:872-881.
22. Shull BL, Capen CV, Riggs MW, Kuehl TJ. Preoperative and postoperative analysis of site-specific pelvic support defects in 81 women treated with sacrospinous ligament suspension and pelvic reconstruction. Am J Obstet Gynecol. 1992;166:1764-1771.
23. Paraiso MFR, Ballard LA, Walter MD, et al. Pelvic support defects and visceral and sexual function in women treated with sacrospinous ligament suspension and pelvic reconstruction. Am J Obstet Gynecol. 1996;175:1423-1431.
24. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421.
25. Benson JT, McClellan E. The effect of vaginal dissection of the pudendal nerve. Obstet Gynecol. 1993;82:387-389.
26. Zivkovic F, Tamussino K, Ralph G, et al. Long-term effects of vaginal dissection on the innervation of the striated urethral sphincter. Obstet Gynecol. 1996;87:257-260.
27. Julian T. The efficacy of Marlex mesh in the repair of severe, recurrent vaginal prolapse of the anterior midvaginal wall. Am J Obstet Gynecol. 1996;175:1472-1475.
28. Weber AM, Walters MD, Piedmonte MR, et al. Anterior colporrhaphy: a randomized trial of three surgical techniques. Am J Obstet Gynecol. 2001;185:1299-1306.
29. Sand PK, et al. Prospective randomized trial of polyglactin 910 mesh to prevent recurrence of cystocele and rectocele. Am J Obstet Gynecol. 2001;184:1357-1364.
30. Ellerkmann RM, Cundiff GW, Melick CF, Nihira MA, Leffler K, Bent AE. Correlation of symptoms with location and severity of pelvic organ prolapse. Am J Obstet Gynecol. 2001;185:1332-1337.
31. Hendrix SL, Clark A, Nygaard I, Aragaki A, Barnabei V, McTiernan A. Pelvic organ prolapse in the Women’s Health Initiative: gravity and gravidity. Am J Obstet Gynecol. 2002;186:1160-1166.
32. Handa VL, Garrett E, Hendrix S, Gold E, Robbins J. Progression and remission of pelvic organ prolapse: a longitudinal study of menopausal women. Am J Obstet Gynecol. 2004;190:27-32.
Preventing BRCA-related cancers: The case for oophorectomy
- Mutations in BRCA1 and BRCA2 may be responsible for more than 90% of inherited predisposition to ovarian cancer.
- BRCA1 and BRCA2 mutations are associated with a lifetime risk of breast cancer of up to 85% and a 15% to 45% lifetime risk of ovarian cancer.
- The only prospective trial to date found risk-reducing salpingo-oophorectomy (RRSO) was associated with an 85% reduction in ovarian cancer and a 68% reduction in breast cancer.
- Because microscopic cancer may be found in 2% to 4% of RRSO specimens upon careful pathologic review, the ovaries and fallopian tubes should be sectioned in their entirety and examined by an experienced gynecologic pathologist.
When A.M. Liber encountered a family of 5 sisters and their mother with histologically confirmed papillary adenocarcinoma of the ovary, he recommended frequent gynecologic cancer screening for all family members and suggested prophylactic oophorectomy as an option.1 The year was 1950.
Flash forward half a century or so, and prophylactic oophorectomy has gained wider acceptance for the prevention of hereditary ovarian and breast cancer, with the only prospective trial to date confirming its overall efficacy for women with BRCA1 and BRCA2 mutations. These mutations are related to the vast majority of inherited ovarian cancers.
Using the evidence published thus far, including the recently published prospective trial, we discuss surgical technique, post-oophorectomy estrogens, psychosocial impact, insurance reimbursement, and other issues.
Three hereditary syndromes
The single biggest risk factor for ovarian cancer is a family history, although only about 10% of cases are believed to be due to an inherited predisposition. Three syndromes are associated with such a predisposition:
- Hereditary breast-ovarian cancer syndrome, caused by mutations in BRCA1 and BRCA2, is thought to be responsible for more than 90% of inherited predisposition to ovarian cancer.
- Hereditary nonpolyposis colon cancer (HNPCC) syndrome is associated with mutations in the mismatch repair genes and a greatly increased risk of cancers of the colon, endometrium, ovaries, and urinary tract. HNPCC accounts for about 2% of inherited ovarian cancers.
- A syndrome of site-specific ovarian cancer also has been proposed, though we lack conclusive evidence that it exists as a separate entity at the genetic level.
How BRCA mutations lead to cancer
BRCA1 and BRCA2 are tumor suppressor genes that play a role in genomic stability and double-stranded DNA break repair. BRCA1 is located on chromosome 17; BRCA2 on chromosome 13. Both genes function as classic tumor suppressors, as described by Knudson.2 Only a single working copy of each gene is needed for the genes to effectively suppress tumors.
In patients with no inherited mutation in these genes, carcinogenesis caused by dysfunction of this pathway can occur only if both working copies of the gene are lost. In contrast, women with an inherited mutation in BRCA1 or BRCA2 start out with only a single working copy of the gene. If any cell loses this single copy, DNA repair cannot occur via this pathway, and cancer can develop.
These repair pathways seem to be particularly important in dividing breast and ovarian cells. This explains why women with inherited mutations in these genes develop cancers more frequently and at an earlier age.
Quantifying the risk
Specific risks associated with BRCA1 and BRCA2 mutations include:
- a lifetime risk of breast cancer of up to 85%, with half of these cancers occurring prior to age 50
- a 15% to 45% lifetime risk of ovarian cancer3,4
Mutations in these genes can be inherited from a mother or father. In the general population, between 1 in 385 and 1 in 800 individuals carry a deleterious mutation in either BRCA1 or BRCA2.
In certain populations, such as Icelandic, French Canadian, or Eastern European Jewish populations, founder effects can contribute to a greatly increased frequency of mutation. For example, the Eastern European Jewish population, from which approximately 90% of North American Jews are descended, has one of the highest known frequencies of BRCA1 and BRCA2 mutation: 1 in 40 individuals carries a deleterious mutation in 1 of these 2 genes.5,6
Most evidence is historical or retrospective
Liber was not the first to suggest oophorectomy to impact the risk of breast or ovarian cancer: The procedure was initially proposed by Schinziner in 1889 as a treatment for breast cancer.7 However, the earliest evidence that oophorectomy was performed as adjuvant therapy did not appear until 7 years later, in 1896 (reviewed by Love and Philips).8
In 1968, Feinleib9 reported that premenopausal oophorectomy decreased the rate of subsequent breast cancer. Twenty years later, Brinton suggested that prophylactic oophorectomy might reduce breast cancer risk in women with a family history of the disease.10
In the sole prospective trial, salpingo-oophorectomy was associated with a 75% reduction in breast and gynecologic cancer.
Post-oophorectomy cancers identified. Possible limitations to the strategy became apparent in the early 1980s, when Tobacman and colleagues11 reported adenocarcinoma histologically indistinguishable from ovarian cancer after oophorectomy in a series of women with a strong family history.
In 1993, Piver et al12 reported a series of 6 cases of primary peritoneal cancer after prophylactic oophorectomy in 324 women from hereditary ovarian cancer families.
In 1997, the Cancer Genetics Studies Consortium reviewed all available data and concluded: “There is insufficient evidence to recommend for or against prophylactic oophorectomy as a measure for reducing ovarian cancer risk. Women with BRCA1 mutations should be counseled that this is an option available to them. Those considering prophylactic oophorectomy should be counseled that cancer has been documented to occur after the procedure.”13
Although the Cancer Genetics Studies Consortium did not specifically comment on prophylactic oophorectomy in carriers of BRCA2 mutations, most authorities interpreted these recommendations to apply to these women as well.
Predicting life expectancy. After these findings, several groups undertook decision analyses to evaluate the effect of prophylactic oophorectomy on life expectancy in women with BRCA mutations. Schrag et al14 reported that prophylactic oophorectomy in a 30-year-old with a BRCA mutation increased life expectancy by 0.3 to 1.7 years. This compares to 0.9 years for adjuvant chemotherapy in node-negative breast cancer.
A subsequent report by Grann and colleagues15 also suggested that prophylactic oophorectomy was associated with an increased life expectancy of 0.4 to 2.6 years. However, surgery was not cost-effective for quality-adjusted life-years saved.
Investigators cite need for prospective studies. In 1999, Rebbeck and colleagues16 conducted a retrospective case-control study of 43 women with BRCA1 mutations who underwent oophorectomy and 79 age-matched women with BRCA1 mutations who had ovaries in situ. In this series, oophorectomy was associated with a 47% decreased risk of subsequent breast cancer (hazard ratio 0.53). However, several investigators cited the need for prospective studies before incorporating oophorectomy into routine clinical practice for the prevention of cancer.17
The first prospective look at risk-reducing surgery
It was in this setting that our group launched a prospective trial to determine whether salpingo-oophorectomy offers any benefit over surveillance in preventing breast and gynecologic (ovarian, fallopian tube, and peritoneal) cancers in women with BRCA mutations.18
Proportional hazard analysis demonstrated that salpingo-oophorectomy was associated with a 75% reduction in subsequent breast and gynecologic cancer incidence in women with BRCA mutations (hazard ratio 0.25, 95% confidence interval 0.08 to 0.74). When the individual endpoints of breast and gynecologic cancer were observed, risk-reducing salpingo-oophorectomy (RRSO) was associated with an 85% reduction in subsequent ovarian cancer and a 68% reduction in subsequent breast cancer.
Methods. From June 1995 through May 2001, we enrolled 265 women with documented BRCA1 or BRCA2 mutations. Patients were followed by annual questionnaire, telephone contact, and medical-record review. Pathology reports were obtained for all new cancers diagnosed during follow-up.
After excluding women who underwent bilateral salpingo-oophorectomy before genetic testing, who were younger than 35 years at the time of testing, or who did not provide any follow-up information, 173 women with ovaries at risk and a documented BRCA mutation remained. These women participated in formal pre- and post-test genetic counseling and received uniform recommendations for cancer risk reduction, as detailed in the TABLE.
During follow-up, we calculated the incidence of new breast and gynecologic cancers diagnosed in the cohort who elected RRSO and compared it with the incidence of these cancers in women who chose surveillance.
Salpingo-oophorectomy was elected by 101 of the 173 women.
Findings. In 3 of these women, early-stage ovarian or fallopian-tube cancer that had not been detected during preoperative evaluation was found at the time of surgery. In the remaining 98 patients who underwent RRSO, 1 peritoneal cancer and 3 breast cancers were diagnosed during a mean 23 months of follow-up. In the 72 women who chose surveillance, 5 ovarian or peritoneal cancers and 8 breast cancers were diagnosed in a mean 25 months of follow-up.
Kaplan-Meier analysis of time to breast or BRCA-related gynecologic cancer is illustrated in FIGURE 1.
Other studies confirm findings. A second retrospective study by Rebbeck et al19 was released simultaneously with our findings and showed similar benefits. They found a 53% reduction in subsequent breast cancer risk and a 96% reduction in subsequent ovarian cancer risk. In the summer of 2003, a study from Israel by Rutter et al provided further confirmation of the substantially decreased incidence of cancer following risk-reducing surgery.20
TABLE
Breast and ovarian cancer risk-reduction strategies for women with BRCA1 or BRCA2 mutations
| TYPE OF CANCER | STRATEGY | ALSO CONSIDER … |
|---|---|---|
| Breast | Monthly self-examination beginning at age 18 | Imaging Breast ultrasound or magnetic resonance imaging |
| 2-4 physician examinations per year, starting at age 25 | Risk–reducing surgery Mastectomy, no earlier than mid-20s | |
| Annual mammography beginning at age 25 | Salpingo-oophorectomy, after age 35 and completion of childbearing | |
| Chemoprevention Tamoxifen. Need to discuss conflicting reports on efficacy | ||
| Ovarian | CA 125 and ultrasound twice yearly, starting at age 35 | Salpingo-oophorectomy After age 35 and the completion of childbearing |
| Chemoprevention Oral contraceptives, though they may be associated with an increased risk of breast cancer | ||
| Source: Adapted from Scheuer et al28 | ||
FIGURE 1 Reduction in cancer cases associated with salpingo-oophorectomy
Reprinted with permission from Kauff ND et al.18
Copyright 2002 Massachusetts Medical Society. All rights reserved.
Good technique and pathologic review may prevent post-oophorectomy cancer
There are 3 theories about the origin of primary peritoneal cancer after oophorectomy:
- The cancer represents undetected occult cancer present at the time of risk-reducing surgery.
- It represents cancer arising in an ovarian remnant left behind after risk-reducing surgery.
- The peritoneal cancer arises de novo from the peritoneal surface epithelium.
Reasonable evidence supports each of these theories; thus, each may play some role in the incidence of “peritoneal” cancer after risk-reducing surgery.21-23
While surgical technique and detailed pathologic review are unlikely to decrease the incidence of de novo peritoneal cancer, they may play a substantial role in reducing ovarian and related cancers after risk-reducing surgery.
Surgical requirements. Obviously, if a surgery is to be risk-reducing, as much as possible of the tissue at risk should be removed. To do so effectively, the surgeon should be comfortable operating in the retroperitoneum so that the infundibulopelvic ligament can be ligated sufficiently proximal from the ovarian hilum to minimize the possibility of an ovarian remnant. Similarly, if a salpingo-oophorectomy without hysterectomy is to be performed, the fallopian tube should be amputated as close as possible to the uterine cornua (FIGURE 2).
Laparoscopy versus open surgery. RRSO can be performed using either a laparoscopic or open approach. The appropriate choice is best determined by the patient’s history, associated comorbid conditions, need for additional procedures, and experience of the surgeon. At our institution, in the absence of contraindications, we generally offer a laparoscopic approach due to its decreased morbidity.
Concomitant hysterectomy? An area of substantial controversy is whether the uterus should be removed at the time of RRSO. In most studies exploring this issue, hereditary breast-ovarian cancer syndrome does not appear to be associated with an increased risk of uterine cancer.24 However, there is concern that the portion of interstitial fallopian tube left behind after salpingo-oophorectomy may be at risk for malignant transformation.25,26
In our series, almost 90% of risk-reducing procedures were salpingo-oophorectomies without hysterectomy. If there is an additional benefit to concomitant hysterectomy, it has yet to be demonstrated by clinical trials.
In several studies, a patient’s level of anxiety was more important than objective cancer risk in the choice of RRSO.
Close pathologic scrutiny advised. Microscopic cancer may be found in 2% to 4% of RRSO specimens upon careful pathologic review.21,27,28 Thus, it is essential that the ovaries and fallopian tubes are sectioned in their entirety and examined by an experienced gynecologic pathologist to minimize the chance that microscopic cancer goes undetected.
It is not clear whether cytology should be routinely done at the time of risk-reducing surgery. A single report documents malignant cells in a woman with a BRCA1 mutation and no obvious foci of malignancy despite hysterectomy with bilateral salpingo-oophorectomy and staging.29 Pending further studies, we routinely send cytology for review.
FIGURE 2 Careful surgical ligation and division to eliminate residual tissue
Source: Devita VT Jr., Hellman S, Rosenberg SA, eds. Progress in Oncology 2003. 2004: Jones and Bartlett Publishers; Sudbury, Mass. Reprinted with permission.
When no BRCA mutation is present
Most of the data cited thus far apply to women with documented BRCA mutations. There is much less information about the relative risks and benefits of RRSO in women with a personal or family history of breast or ovarian cancer who lack a documented BRCA mutation.
Although RRSO may be appropriate for some of these women, in 2004 it is not the standard of care to recommend RRSO to all individuals with a personal or family history suggestive of an inherited predisposition to ovarian cancer. These patients are best managed by an interdisciplinary team of gynecologists, gynecologic oncologists, and clinical geneticists, all with experience caring for women who may have an inherited predisposition.
Is anxiety a factor?
We have limited information about the psychosocial impact of RRSO. Several studies have found that a patient’s level of anxiety is a more important factor than objective cancer risk in the decision to undergo RRSO.30,31 Unfortunately, we do not yet know whether the surgery successfully reduces these patients’ subjective concerns.
A recent study showed that risk-reducing surgery did not impair women’s overall health or psychological well-being.32 However, 20.7% of the women reported substantial cancer-related anxiety despite the risk-reducing surgery. This issue requires further investigation.
Is estrogen the best option for surgical menopause?
The role of hormone replacement after RRSO is unclear. The issue is important because many women considering salpingo-oophorectomy are in their late 30s or early 40s, when premature surgical menopause is a predictable result. Consequences can include considerable vasomotor and pelvic symptoms.
Preliminary data suggest that a woman’s satisfaction with RRSO depends in large part on its impact on sexual functioning.32 Urogenital symptoms that adversely affected sexual function, such as vaginal dryness and dyspareunia, were the most significant predictors of dissatisfaction with surgery.
Premature surgical menopause also has a substantial impact on osteoporosis risk, while its effect on heart disease remains uncertain.
While nonhormonal therapies can address each of these issues, we need more data on their long-term use. We counsel women considering RRSO that hormone replacement may be an option. We believe it is unlikely to reduce the efficacy of RRSO in preventing ovarian cancer, but it may reduce the protective effect of RRSO against subsequent breast cancer. Until further studies are available, we recommend that decisions regarding hormone replacement be individualized to the patient’s specific symptoms and personal history.
Not all insurers cover RRSO
One study explored insurance carriers’ policies about reimbursing risk-reducing surgical procedures and found that 10% to 11% of private insurers and 48% to 50% of governmental carriers had policies specifically denying coverage for such operations.
An additional 40% to 64% of insurers had no identifiable policy regarding these procedures in women with BRCA mutations.33 The authors speculated that, without identifiable policies, this critical health-care decision may be subject to arbitrary criteria that result in substantial variation.
When we recently investigated the reimbursement experience of women with BRCA mutations undergoing RRSO at our institution, we found that 97% of the procedures were reimbursed in full, less any applicable coinsurance and deductibles.34 Two important limitations of our study: It was conducted at a tertiary cancer center and was retrospective. It is not known if the findings reflect the experience of women with BRCA mutation who have risk-reducing surgery in other settings.
Unresolved issues
RRSO clearly has a role in preventing breast and ovarian cancer in women at inherited risk. However, several questions remain unanswered:
- Who is the best candidate?
- What is optimal timing of the procedure?
- What, if any, concomitant procedures should be performed?
- What is the role of hormone replacement after the surgery?
These issues will be best addressed through multicenter prospective trials, such as the one now being conducted by the Gynecologic Oncology Group.
Hope also remains that further research will improve serum and radiological detection of early ovarian cancer, and that basic research on the molecular etiology and progression of these cancers will ultimately render it unnecessary to remove organs at risk.
The authors report no financial relationships relevant to this article.
1. Liber AM. Ovarian cancer in a mother and five daughters. Arch Pathol. 1950;49:280-290.
2. Knudson AG, Jr. Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA. 1971;68:820-823.
3. Ford D, Easton DF, Stratton M, et al. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am J Hum Genet. 1998;62:676-689.
4. Risch HA, McLaughlin JR, Cole DE, et al. Prevalence and penetrance of germline BRCA1 and BRCA2 mutations in a population series of 649 women with ovarian cancer. Am J Hum Genet. 2001;68:700-710.
5. Oddoux C, Struewing JP, Clayton CM, et al. The carrier frequency of the BRCA2 6174delT mutation among Ashkenazi Jewish individuals is approximately 1%. Nat Genet. 1996;14:188-190.
6. Tonin P, Weber B, Offit K, et al. Frequency of recurrent BRCA1 and BRCA2 mutations in Ashkenazi Jewish breast cancer families. Nat Med. 1996;2:1179-1183.
7. Schinzinger A. Ueber Carcinoma Mammae. Verhandlungen der Deutschen Gesellschaft fur Chirurgie. 18th Kongress, Berlin, Apr 24–27, 1889. Berlin, Germany: Hirschwald; 1889:28[abstract].
8. Love RR, Philips J. Oophorectomy for breast cancer: history revisited. J Natl Cancer Inst. 2002;94:1433-1434.
9. Feinleib M. Breast cancer and artificial menopause: a cohort study. J Natl Cancer Inst. 1968;41:315-329.
10. Brinton LA, Schairer C, Hoover RN, Fraumeni JF, Jr. Menstrual factors and risk of breast cancer. Cancer Invest. 1988;6:245-254.
11. Tobacman JK, Greene MH, Tucker MA, Costa J, Kase R, Fraumeni JF, Jr. Intraabdominal carcinomatosis after prophylactic oophorectomy in ovarian-cancer-prone families. Lancet. 1982;2:795-797.
12. Piver MS, Jishi MF, Tsukada Y, Nava G. Primary peritoneal carcinoma after prophylactic oophorectomy in women with a family history of ovarian cancer. A report of the Gilda Radner Familial Ovarian Cancer Registry. Cancer. 1993;71:2751-2755.
13. Burke W, Daly M, Garber J, et al. Recommendations for follow-up care of individuals with an inherited predisposition to cancer. II. BRCA1 and BRCA2. Cancer Genetics Studies Consortium. JAMA. 1997;277:997-1003.
14. Schrag D, Kuntz KM, Garber JE, Weeks JC. Decision analysis—effects of prophylactic mastectomy and oophorectomy on life expectancy among women with BRCA1 or BRCA2 mutations. N Engl J Med. 1997;336:1465-1471.
15. Grann VR, Panageas KS, Whang W, Antman KH, Neugut AI. Decision analysis of prophylactic mastectomy and oophorectomy in BRCA1-positive or BRCA2-positive patients. J Clin Oncol. 1998;16:979-985.
16. Rebbeck TR, Levin AM, Eisen A, et al. Breast cancer risk after bilateral prophylactic oophorectomy in BRCA1 mutation carriers. J Natl Cancer Inst. 1999;91:1475-1479.
17. Eisen A, Rebbeck TR, Wood WC, Weber BL. Prophylactic surgery in women with a hereditary predisposition to breast and ovarian cancer. J Clin Oncol. 2000;18:1980-1995.
18. Kauff ND, Satagopan JM, Robson ME, et al. Risk-reducing salpingo-oophorectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med. 2002;346:1609-1615.
19. Rebbeck TR, Lynch HT, Neuhausen SL, et al. Prophylactic oophorectomy in carriers of BRCA1 or BRCA2 mutations. N Engl J Med. 2002;346:1616-1622.
20. Rutter JL, Wacholder S, Chetrit A, et al. Gynecologic surgeries and risk of ovarian cancer in women with BRCA1 and BRCA2 Ashkenazi founder mutations: an Israeli population-based case-control study. J Natl Cancer Inst. 2003;95:1072-1078.
21. Lu KH, Garber JE, Cramer DW, et al. Occult ovarian tumors in women with BRCA1 or BRCA2 mutations undergoing prophylactic oophorectomy. J Clin Oncol. 2000;18:2728-2732.
22. Lafferty HW, Angioli R, Rudolph J, Penalver MA. Ovarian remnant syndrome: experience at Jackson Memorial Hospital, University of Miami, 1985 through 1993. Am J Obstet Gynecol. 1996;174:641-645.
23. Schorge JO, Muto MG, Welch WR, et al. Molecular evidence for multifocal papillary serous carcinoma of the peritoneum in patients with germline BRCA1 mutations. J Natl Cancer Inst. 1998;90:841-845.
24. Levine DA, Lin O, Barakat RR, et al. Risk of endometrial carcinoma associated with BRCA mutation. Gynecol Oncol. 2001;80:395-398.
25. Paley PJ, Swisher EM, Garcia RL, et al. Occult cancer of the fallopian tube in BRCA-1 germline mutation carriers at prophylactic oophorectomy: a case for recommending hysterectomy at surgical prophylaxis. Gynecol Oncol. 2001;80:176-180.
26. Aziz S, Kuperstein G, Rosen B, et al. A genetic epidemiological study of carcinoma of the fallopian tube. Gynecol Oncol. 2001;80:341-345.
27. Colgan TJ, Murphy J, Cole DE, Narod S, Rosen B. Occult carcinoma in prophylactic oophorectomy specimens: prevalence and association with BRCA germline mutation status. Am J Surg Pathol. 2001;25:1283-1289.
28. Scheuer L, Kauff N, Robson M, et al. Outcome of preventive surgery and screening for breast and ovarian cancer in BRCA mutation carriers. J Clin Oncol. 2002;20:1260-1268.
29. Colgan TJ, Boerner SL, Murphy J, Cole DE, Narod S, Rosen B. Peritoneal lavage cytology: an assessment of its value during prophylactic oophorectomy. Gynecol Oncol. 2002;85:397-403.
30. Meiser B, Butow P, Barratt A, et al. Attitudes toward prophylactic oophorectomy and screening utilization in women at increased risk of developing hereditary breast/ovarian cancer. Gynecol Oncol. 1999;75:122-129.
31. Hurley KE, Miller SM, Costalas JW, Gillespie D, Daly MB. Anxiety/uncertainty reduction as a motivation for interest in prophylactic oophorectomy in women with a family history of ovarian cancer. J Womens Health Gend Based Med. 2001;10:189-199.
32. Robson M, Hensley M, Barakat R, et al. Quality of life in women at risk for ovarian cancer who have undergone risk-reducing oophorectomy. Gynecol Oncol. 2003;89:281-287.
33. Kuerer HM, Hwang ES, Anthony JP, et al. Current national health insurance coverage policies for breast and ovarian cancer prophylactic surgery. Ann Surg Oncol. 2000;7:325-332.
34. Kauff ND, Scheuer L, Robson ME, et al. Insurance reimbursement for risk-reducing mastectomy and oophorectomy in women with BRCA1 or BRCA2 mutations. Genet Med. 2001;3:422-425.
- Mutations in BRCA1 and BRCA2 may be responsible for more than 90% of inherited predisposition to ovarian cancer.
- BRCA1 and BRCA2 mutations are associated with a lifetime risk of breast cancer of up to 85% and a 15% to 45% lifetime risk of ovarian cancer.
- The only prospective trial to date found risk-reducing salpingo-oophorectomy (RRSO) was associated with an 85% reduction in ovarian cancer and a 68% reduction in breast cancer.
- Because microscopic cancer may be found in 2% to 4% of RRSO specimens upon careful pathologic review, the ovaries and fallopian tubes should be sectioned in their entirety and examined by an experienced gynecologic pathologist.
When A.M. Liber encountered a family of 5 sisters and their mother with histologically confirmed papillary adenocarcinoma of the ovary, he recommended frequent gynecologic cancer screening for all family members and suggested prophylactic oophorectomy as an option.1 The year was 1950.
Flash forward half a century or so, and prophylactic oophorectomy has gained wider acceptance for the prevention of hereditary ovarian and breast cancer, with the only prospective trial to date confirming its overall efficacy for women with BRCA1 and BRCA2 mutations. These mutations are related to the vast majority of inherited ovarian cancers.
Using the evidence published thus far, including the recently published prospective trial, we discuss surgical technique, post-oophorectomy estrogens, psychosocial impact, insurance reimbursement, and other issues.
Three hereditary syndromes
The single biggest risk factor for ovarian cancer is a family history, although only about 10% of cases are believed to be due to an inherited predisposition. Three syndromes are associated with such a predisposition:
- Hereditary breast-ovarian cancer syndrome, caused by mutations in BRCA1 and BRCA2, is thought to be responsible for more than 90% of inherited predisposition to ovarian cancer.
- Hereditary nonpolyposis colon cancer (HNPCC) syndrome is associated with mutations in the mismatch repair genes and a greatly increased risk of cancers of the colon, endometrium, ovaries, and urinary tract. HNPCC accounts for about 2% of inherited ovarian cancers.
- A syndrome of site-specific ovarian cancer also has been proposed, though we lack conclusive evidence that it exists as a separate entity at the genetic level.
How BRCA mutations lead to cancer
BRCA1 and BRCA2 are tumor suppressor genes that play a role in genomic stability and double-stranded DNA break repair. BRCA1 is located on chromosome 17; BRCA2 on chromosome 13. Both genes function as classic tumor suppressors, as described by Knudson.2 Only a single working copy of each gene is needed for the genes to effectively suppress tumors.
In patients with no inherited mutation in these genes, carcinogenesis caused by dysfunction of this pathway can occur only if both working copies of the gene are lost. In contrast, women with an inherited mutation in BRCA1 or BRCA2 start out with only a single working copy of the gene. If any cell loses this single copy, DNA repair cannot occur via this pathway, and cancer can develop.
These repair pathways seem to be particularly important in dividing breast and ovarian cells. This explains why women with inherited mutations in these genes develop cancers more frequently and at an earlier age.
Quantifying the risk
Specific risks associated with BRCA1 and BRCA2 mutations include:
- a lifetime risk of breast cancer of up to 85%, with half of these cancers occurring prior to age 50
- a 15% to 45% lifetime risk of ovarian cancer3,4
Mutations in these genes can be inherited from a mother or father. In the general population, between 1 in 385 and 1 in 800 individuals carry a deleterious mutation in either BRCA1 or BRCA2.
In certain populations, such as Icelandic, French Canadian, or Eastern European Jewish populations, founder effects can contribute to a greatly increased frequency of mutation. For example, the Eastern European Jewish population, from which approximately 90% of North American Jews are descended, has one of the highest known frequencies of BRCA1 and BRCA2 mutation: 1 in 40 individuals carries a deleterious mutation in 1 of these 2 genes.5,6
Most evidence is historical or retrospective
Liber was not the first to suggest oophorectomy to impact the risk of breast or ovarian cancer: The procedure was initially proposed by Schinziner in 1889 as a treatment for breast cancer.7 However, the earliest evidence that oophorectomy was performed as adjuvant therapy did not appear until 7 years later, in 1896 (reviewed by Love and Philips).8
In 1968, Feinleib9 reported that premenopausal oophorectomy decreased the rate of subsequent breast cancer. Twenty years later, Brinton suggested that prophylactic oophorectomy might reduce breast cancer risk in women with a family history of the disease.10
In the sole prospective trial, salpingo-oophorectomy was associated with a 75% reduction in breast and gynecologic cancer.
Post-oophorectomy cancers identified. Possible limitations to the strategy became apparent in the early 1980s, when Tobacman and colleagues11 reported adenocarcinoma histologically indistinguishable from ovarian cancer after oophorectomy in a series of women with a strong family history.
In 1993, Piver et al12 reported a series of 6 cases of primary peritoneal cancer after prophylactic oophorectomy in 324 women from hereditary ovarian cancer families.
In 1997, the Cancer Genetics Studies Consortium reviewed all available data and concluded: “There is insufficient evidence to recommend for or against prophylactic oophorectomy as a measure for reducing ovarian cancer risk. Women with BRCA1 mutations should be counseled that this is an option available to them. Those considering prophylactic oophorectomy should be counseled that cancer has been documented to occur after the procedure.”13
Although the Cancer Genetics Studies Consortium did not specifically comment on prophylactic oophorectomy in carriers of BRCA2 mutations, most authorities interpreted these recommendations to apply to these women as well.
Predicting life expectancy. After these findings, several groups undertook decision analyses to evaluate the effect of prophylactic oophorectomy on life expectancy in women with BRCA mutations. Schrag et al14 reported that prophylactic oophorectomy in a 30-year-old with a BRCA mutation increased life expectancy by 0.3 to 1.7 years. This compares to 0.9 years for adjuvant chemotherapy in node-negative breast cancer.
A subsequent report by Grann and colleagues15 also suggested that prophylactic oophorectomy was associated with an increased life expectancy of 0.4 to 2.6 years. However, surgery was not cost-effective for quality-adjusted life-years saved.
Investigators cite need for prospective studies. In 1999, Rebbeck and colleagues16 conducted a retrospective case-control study of 43 women with BRCA1 mutations who underwent oophorectomy and 79 age-matched women with BRCA1 mutations who had ovaries in situ. In this series, oophorectomy was associated with a 47% decreased risk of subsequent breast cancer (hazard ratio 0.53). However, several investigators cited the need for prospective studies before incorporating oophorectomy into routine clinical practice for the prevention of cancer.17
The first prospective look at risk-reducing surgery
It was in this setting that our group launched a prospective trial to determine whether salpingo-oophorectomy offers any benefit over surveillance in preventing breast and gynecologic (ovarian, fallopian tube, and peritoneal) cancers in women with BRCA mutations.18
Proportional hazard analysis demonstrated that salpingo-oophorectomy was associated with a 75% reduction in subsequent breast and gynecologic cancer incidence in women with BRCA mutations (hazard ratio 0.25, 95% confidence interval 0.08 to 0.74). When the individual endpoints of breast and gynecologic cancer were observed, risk-reducing salpingo-oophorectomy (RRSO) was associated with an 85% reduction in subsequent ovarian cancer and a 68% reduction in subsequent breast cancer.
Methods. From June 1995 through May 2001, we enrolled 265 women with documented BRCA1 or BRCA2 mutations. Patients were followed by annual questionnaire, telephone contact, and medical-record review. Pathology reports were obtained for all new cancers diagnosed during follow-up.
After excluding women who underwent bilateral salpingo-oophorectomy before genetic testing, who were younger than 35 years at the time of testing, or who did not provide any follow-up information, 173 women with ovaries at risk and a documented BRCA mutation remained. These women participated in formal pre- and post-test genetic counseling and received uniform recommendations for cancer risk reduction, as detailed in the TABLE.
During follow-up, we calculated the incidence of new breast and gynecologic cancers diagnosed in the cohort who elected RRSO and compared it with the incidence of these cancers in women who chose surveillance.
Salpingo-oophorectomy was elected by 101 of the 173 women.
Findings. In 3 of these women, early-stage ovarian or fallopian-tube cancer that had not been detected during preoperative evaluation was found at the time of surgery. In the remaining 98 patients who underwent RRSO, 1 peritoneal cancer and 3 breast cancers were diagnosed during a mean 23 months of follow-up. In the 72 women who chose surveillance, 5 ovarian or peritoneal cancers and 8 breast cancers were diagnosed in a mean 25 months of follow-up.
Kaplan-Meier analysis of time to breast or BRCA-related gynecologic cancer is illustrated in FIGURE 1.
Other studies confirm findings. A second retrospective study by Rebbeck et al19 was released simultaneously with our findings and showed similar benefits. They found a 53% reduction in subsequent breast cancer risk and a 96% reduction in subsequent ovarian cancer risk. In the summer of 2003, a study from Israel by Rutter et al provided further confirmation of the substantially decreased incidence of cancer following risk-reducing surgery.20
TABLE
Breast and ovarian cancer risk-reduction strategies for women with BRCA1 or BRCA2 mutations
| TYPE OF CANCER | STRATEGY | ALSO CONSIDER … |
|---|---|---|
| Breast | Monthly self-examination beginning at age 18 | Imaging Breast ultrasound or magnetic resonance imaging |
| 2-4 physician examinations per year, starting at age 25 | Risk–reducing surgery Mastectomy, no earlier than mid-20s | |
| Annual mammography beginning at age 25 | Salpingo-oophorectomy, after age 35 and completion of childbearing | |
| Chemoprevention Tamoxifen. Need to discuss conflicting reports on efficacy | ||
| Ovarian | CA 125 and ultrasound twice yearly, starting at age 35 | Salpingo-oophorectomy After age 35 and the completion of childbearing |
| Chemoprevention Oral contraceptives, though they may be associated with an increased risk of breast cancer | ||
| Source: Adapted from Scheuer et al28 | ||
FIGURE 1 Reduction in cancer cases associated with salpingo-oophorectomy
Reprinted with permission from Kauff ND et al.18
Copyright 2002 Massachusetts Medical Society. All rights reserved.
Good technique and pathologic review may prevent post-oophorectomy cancer
There are 3 theories about the origin of primary peritoneal cancer after oophorectomy:
- The cancer represents undetected occult cancer present at the time of risk-reducing surgery.
- It represents cancer arising in an ovarian remnant left behind after risk-reducing surgery.
- The peritoneal cancer arises de novo from the peritoneal surface epithelium.
Reasonable evidence supports each of these theories; thus, each may play some role in the incidence of “peritoneal” cancer after risk-reducing surgery.21-23
While surgical technique and detailed pathologic review are unlikely to decrease the incidence of de novo peritoneal cancer, they may play a substantial role in reducing ovarian and related cancers after risk-reducing surgery.
Surgical requirements. Obviously, if a surgery is to be risk-reducing, as much as possible of the tissue at risk should be removed. To do so effectively, the surgeon should be comfortable operating in the retroperitoneum so that the infundibulopelvic ligament can be ligated sufficiently proximal from the ovarian hilum to minimize the possibility of an ovarian remnant. Similarly, if a salpingo-oophorectomy without hysterectomy is to be performed, the fallopian tube should be amputated as close as possible to the uterine cornua (FIGURE 2).
Laparoscopy versus open surgery. RRSO can be performed using either a laparoscopic or open approach. The appropriate choice is best determined by the patient’s history, associated comorbid conditions, need for additional procedures, and experience of the surgeon. At our institution, in the absence of contraindications, we generally offer a laparoscopic approach due to its decreased morbidity.
Concomitant hysterectomy? An area of substantial controversy is whether the uterus should be removed at the time of RRSO. In most studies exploring this issue, hereditary breast-ovarian cancer syndrome does not appear to be associated with an increased risk of uterine cancer.24 However, there is concern that the portion of interstitial fallopian tube left behind after salpingo-oophorectomy may be at risk for malignant transformation.25,26
In our series, almost 90% of risk-reducing procedures were salpingo-oophorectomies without hysterectomy. If there is an additional benefit to concomitant hysterectomy, it has yet to be demonstrated by clinical trials.
In several studies, a patient’s level of anxiety was more important than objective cancer risk in the choice of RRSO.
Close pathologic scrutiny advised. Microscopic cancer may be found in 2% to 4% of RRSO specimens upon careful pathologic review.21,27,28 Thus, it is essential that the ovaries and fallopian tubes are sectioned in their entirety and examined by an experienced gynecologic pathologist to minimize the chance that microscopic cancer goes undetected.
It is not clear whether cytology should be routinely done at the time of risk-reducing surgery. A single report documents malignant cells in a woman with a BRCA1 mutation and no obvious foci of malignancy despite hysterectomy with bilateral salpingo-oophorectomy and staging.29 Pending further studies, we routinely send cytology for review.
FIGURE 2 Careful surgical ligation and division to eliminate residual tissue
Source: Devita VT Jr., Hellman S, Rosenberg SA, eds. Progress in Oncology 2003. 2004: Jones and Bartlett Publishers; Sudbury, Mass. Reprinted with permission.
When no BRCA mutation is present
Most of the data cited thus far apply to women with documented BRCA mutations. There is much less information about the relative risks and benefits of RRSO in women with a personal or family history of breast or ovarian cancer who lack a documented BRCA mutation.
Although RRSO may be appropriate for some of these women, in 2004 it is not the standard of care to recommend RRSO to all individuals with a personal or family history suggestive of an inherited predisposition to ovarian cancer. These patients are best managed by an interdisciplinary team of gynecologists, gynecologic oncologists, and clinical geneticists, all with experience caring for women who may have an inherited predisposition.
Is anxiety a factor?
We have limited information about the psychosocial impact of RRSO. Several studies have found that a patient’s level of anxiety is a more important factor than objective cancer risk in the decision to undergo RRSO.30,31 Unfortunately, we do not yet know whether the surgery successfully reduces these patients’ subjective concerns.
A recent study showed that risk-reducing surgery did not impair women’s overall health or psychological well-being.32 However, 20.7% of the women reported substantial cancer-related anxiety despite the risk-reducing surgery. This issue requires further investigation.
Is estrogen the best option for surgical menopause?
The role of hormone replacement after RRSO is unclear. The issue is important because many women considering salpingo-oophorectomy are in their late 30s or early 40s, when premature surgical menopause is a predictable result. Consequences can include considerable vasomotor and pelvic symptoms.
Preliminary data suggest that a woman’s satisfaction with RRSO depends in large part on its impact on sexual functioning.32 Urogenital symptoms that adversely affected sexual function, such as vaginal dryness and dyspareunia, were the most significant predictors of dissatisfaction with surgery.
Premature surgical menopause also has a substantial impact on osteoporosis risk, while its effect on heart disease remains uncertain.
While nonhormonal therapies can address each of these issues, we need more data on their long-term use. We counsel women considering RRSO that hormone replacement may be an option. We believe it is unlikely to reduce the efficacy of RRSO in preventing ovarian cancer, but it may reduce the protective effect of RRSO against subsequent breast cancer. Until further studies are available, we recommend that decisions regarding hormone replacement be individualized to the patient’s specific symptoms and personal history.
Not all insurers cover RRSO
One study explored insurance carriers’ policies about reimbursing risk-reducing surgical procedures and found that 10% to 11% of private insurers and 48% to 50% of governmental carriers had policies specifically denying coverage for such operations.
An additional 40% to 64% of insurers had no identifiable policy regarding these procedures in women with BRCA mutations.33 The authors speculated that, without identifiable policies, this critical health-care decision may be subject to arbitrary criteria that result in substantial variation.
When we recently investigated the reimbursement experience of women with BRCA mutations undergoing RRSO at our institution, we found that 97% of the procedures were reimbursed in full, less any applicable coinsurance and deductibles.34 Two important limitations of our study: It was conducted at a tertiary cancer center and was retrospective. It is not known if the findings reflect the experience of women with BRCA mutation who have risk-reducing surgery in other settings.
Unresolved issues
RRSO clearly has a role in preventing breast and ovarian cancer in women at inherited risk. However, several questions remain unanswered:
- Who is the best candidate?
- What is optimal timing of the procedure?
- What, if any, concomitant procedures should be performed?
- What is the role of hormone replacement after the surgery?
These issues will be best addressed through multicenter prospective trials, such as the one now being conducted by the Gynecologic Oncology Group.
Hope also remains that further research will improve serum and radiological detection of early ovarian cancer, and that basic research on the molecular etiology and progression of these cancers will ultimately render it unnecessary to remove organs at risk.
The authors report no financial relationships relevant to this article.
- Mutations in BRCA1 and BRCA2 may be responsible for more than 90% of inherited predisposition to ovarian cancer.
- BRCA1 and BRCA2 mutations are associated with a lifetime risk of breast cancer of up to 85% and a 15% to 45% lifetime risk of ovarian cancer.
- The only prospective trial to date found risk-reducing salpingo-oophorectomy (RRSO) was associated with an 85% reduction in ovarian cancer and a 68% reduction in breast cancer.
- Because microscopic cancer may be found in 2% to 4% of RRSO specimens upon careful pathologic review, the ovaries and fallopian tubes should be sectioned in their entirety and examined by an experienced gynecologic pathologist.
When A.M. Liber encountered a family of 5 sisters and their mother with histologically confirmed papillary adenocarcinoma of the ovary, he recommended frequent gynecologic cancer screening for all family members and suggested prophylactic oophorectomy as an option.1 The year was 1950.
Flash forward half a century or so, and prophylactic oophorectomy has gained wider acceptance for the prevention of hereditary ovarian and breast cancer, with the only prospective trial to date confirming its overall efficacy for women with BRCA1 and BRCA2 mutations. These mutations are related to the vast majority of inherited ovarian cancers.
Using the evidence published thus far, including the recently published prospective trial, we discuss surgical technique, post-oophorectomy estrogens, psychosocial impact, insurance reimbursement, and other issues.
Three hereditary syndromes
The single biggest risk factor for ovarian cancer is a family history, although only about 10% of cases are believed to be due to an inherited predisposition. Three syndromes are associated with such a predisposition:
- Hereditary breast-ovarian cancer syndrome, caused by mutations in BRCA1 and BRCA2, is thought to be responsible for more than 90% of inherited predisposition to ovarian cancer.
- Hereditary nonpolyposis colon cancer (HNPCC) syndrome is associated with mutations in the mismatch repair genes and a greatly increased risk of cancers of the colon, endometrium, ovaries, and urinary tract. HNPCC accounts for about 2% of inherited ovarian cancers.
- A syndrome of site-specific ovarian cancer also has been proposed, though we lack conclusive evidence that it exists as a separate entity at the genetic level.
How BRCA mutations lead to cancer
BRCA1 and BRCA2 are tumor suppressor genes that play a role in genomic stability and double-stranded DNA break repair. BRCA1 is located on chromosome 17; BRCA2 on chromosome 13. Both genes function as classic tumor suppressors, as described by Knudson.2 Only a single working copy of each gene is needed for the genes to effectively suppress tumors.
In patients with no inherited mutation in these genes, carcinogenesis caused by dysfunction of this pathway can occur only if both working copies of the gene are lost. In contrast, women with an inherited mutation in BRCA1 or BRCA2 start out with only a single working copy of the gene. If any cell loses this single copy, DNA repair cannot occur via this pathway, and cancer can develop.
These repair pathways seem to be particularly important in dividing breast and ovarian cells. This explains why women with inherited mutations in these genes develop cancers more frequently and at an earlier age.
Quantifying the risk
Specific risks associated with BRCA1 and BRCA2 mutations include:
- a lifetime risk of breast cancer of up to 85%, with half of these cancers occurring prior to age 50
- a 15% to 45% lifetime risk of ovarian cancer3,4
Mutations in these genes can be inherited from a mother or father. In the general population, between 1 in 385 and 1 in 800 individuals carry a deleterious mutation in either BRCA1 or BRCA2.
In certain populations, such as Icelandic, French Canadian, or Eastern European Jewish populations, founder effects can contribute to a greatly increased frequency of mutation. For example, the Eastern European Jewish population, from which approximately 90% of North American Jews are descended, has one of the highest known frequencies of BRCA1 and BRCA2 mutation: 1 in 40 individuals carries a deleterious mutation in 1 of these 2 genes.5,6
Most evidence is historical or retrospective
Liber was not the first to suggest oophorectomy to impact the risk of breast or ovarian cancer: The procedure was initially proposed by Schinziner in 1889 as a treatment for breast cancer.7 However, the earliest evidence that oophorectomy was performed as adjuvant therapy did not appear until 7 years later, in 1896 (reviewed by Love and Philips).8
In 1968, Feinleib9 reported that premenopausal oophorectomy decreased the rate of subsequent breast cancer. Twenty years later, Brinton suggested that prophylactic oophorectomy might reduce breast cancer risk in women with a family history of the disease.10
In the sole prospective trial, salpingo-oophorectomy was associated with a 75% reduction in breast and gynecologic cancer.
Post-oophorectomy cancers identified. Possible limitations to the strategy became apparent in the early 1980s, when Tobacman and colleagues11 reported adenocarcinoma histologically indistinguishable from ovarian cancer after oophorectomy in a series of women with a strong family history.
In 1993, Piver et al12 reported a series of 6 cases of primary peritoneal cancer after prophylactic oophorectomy in 324 women from hereditary ovarian cancer families.
In 1997, the Cancer Genetics Studies Consortium reviewed all available data and concluded: “There is insufficient evidence to recommend for or against prophylactic oophorectomy as a measure for reducing ovarian cancer risk. Women with BRCA1 mutations should be counseled that this is an option available to them. Those considering prophylactic oophorectomy should be counseled that cancer has been documented to occur after the procedure.”13
Although the Cancer Genetics Studies Consortium did not specifically comment on prophylactic oophorectomy in carriers of BRCA2 mutations, most authorities interpreted these recommendations to apply to these women as well.
Predicting life expectancy. After these findings, several groups undertook decision analyses to evaluate the effect of prophylactic oophorectomy on life expectancy in women with BRCA mutations. Schrag et al14 reported that prophylactic oophorectomy in a 30-year-old with a BRCA mutation increased life expectancy by 0.3 to 1.7 years. This compares to 0.9 years for adjuvant chemotherapy in node-negative breast cancer.
A subsequent report by Grann and colleagues15 also suggested that prophylactic oophorectomy was associated with an increased life expectancy of 0.4 to 2.6 years. However, surgery was not cost-effective for quality-adjusted life-years saved.
Investigators cite need for prospective studies. In 1999, Rebbeck and colleagues16 conducted a retrospective case-control study of 43 women with BRCA1 mutations who underwent oophorectomy and 79 age-matched women with BRCA1 mutations who had ovaries in situ. In this series, oophorectomy was associated with a 47% decreased risk of subsequent breast cancer (hazard ratio 0.53). However, several investigators cited the need for prospective studies before incorporating oophorectomy into routine clinical practice for the prevention of cancer.17
The first prospective look at risk-reducing surgery
It was in this setting that our group launched a prospective trial to determine whether salpingo-oophorectomy offers any benefit over surveillance in preventing breast and gynecologic (ovarian, fallopian tube, and peritoneal) cancers in women with BRCA mutations.18
Proportional hazard analysis demonstrated that salpingo-oophorectomy was associated with a 75% reduction in subsequent breast and gynecologic cancer incidence in women with BRCA mutations (hazard ratio 0.25, 95% confidence interval 0.08 to 0.74). When the individual endpoints of breast and gynecologic cancer were observed, risk-reducing salpingo-oophorectomy (RRSO) was associated with an 85% reduction in subsequent ovarian cancer and a 68% reduction in subsequent breast cancer.
Methods. From June 1995 through May 2001, we enrolled 265 women with documented BRCA1 or BRCA2 mutations. Patients were followed by annual questionnaire, telephone contact, and medical-record review. Pathology reports were obtained for all new cancers diagnosed during follow-up.
After excluding women who underwent bilateral salpingo-oophorectomy before genetic testing, who were younger than 35 years at the time of testing, or who did not provide any follow-up information, 173 women with ovaries at risk and a documented BRCA mutation remained. These women participated in formal pre- and post-test genetic counseling and received uniform recommendations for cancer risk reduction, as detailed in the TABLE.
During follow-up, we calculated the incidence of new breast and gynecologic cancers diagnosed in the cohort who elected RRSO and compared it with the incidence of these cancers in women who chose surveillance.
Salpingo-oophorectomy was elected by 101 of the 173 women.
Findings. In 3 of these women, early-stage ovarian or fallopian-tube cancer that had not been detected during preoperative evaluation was found at the time of surgery. In the remaining 98 patients who underwent RRSO, 1 peritoneal cancer and 3 breast cancers were diagnosed during a mean 23 months of follow-up. In the 72 women who chose surveillance, 5 ovarian or peritoneal cancers and 8 breast cancers were diagnosed in a mean 25 months of follow-up.
Kaplan-Meier analysis of time to breast or BRCA-related gynecologic cancer is illustrated in FIGURE 1.
Other studies confirm findings. A second retrospective study by Rebbeck et al19 was released simultaneously with our findings and showed similar benefits. They found a 53% reduction in subsequent breast cancer risk and a 96% reduction in subsequent ovarian cancer risk. In the summer of 2003, a study from Israel by Rutter et al provided further confirmation of the substantially decreased incidence of cancer following risk-reducing surgery.20
TABLE
Breast and ovarian cancer risk-reduction strategies for women with BRCA1 or BRCA2 mutations
| TYPE OF CANCER | STRATEGY | ALSO CONSIDER … |
|---|---|---|
| Breast | Monthly self-examination beginning at age 18 | Imaging Breast ultrasound or magnetic resonance imaging |
| 2-4 physician examinations per year, starting at age 25 | Risk–reducing surgery Mastectomy, no earlier than mid-20s | |
| Annual mammography beginning at age 25 | Salpingo-oophorectomy, after age 35 and completion of childbearing | |
| Chemoprevention Tamoxifen. Need to discuss conflicting reports on efficacy | ||
| Ovarian | CA 125 and ultrasound twice yearly, starting at age 35 | Salpingo-oophorectomy After age 35 and the completion of childbearing |
| Chemoprevention Oral contraceptives, though they may be associated with an increased risk of breast cancer | ||
| Source: Adapted from Scheuer et al28 | ||
FIGURE 1 Reduction in cancer cases associated with salpingo-oophorectomy
Reprinted with permission from Kauff ND et al.18
Copyright 2002 Massachusetts Medical Society. All rights reserved.
Good technique and pathologic review may prevent post-oophorectomy cancer
There are 3 theories about the origin of primary peritoneal cancer after oophorectomy:
- The cancer represents undetected occult cancer present at the time of risk-reducing surgery.
- It represents cancer arising in an ovarian remnant left behind after risk-reducing surgery.
- The peritoneal cancer arises de novo from the peritoneal surface epithelium.
Reasonable evidence supports each of these theories; thus, each may play some role in the incidence of “peritoneal” cancer after risk-reducing surgery.21-23
While surgical technique and detailed pathologic review are unlikely to decrease the incidence of de novo peritoneal cancer, they may play a substantial role in reducing ovarian and related cancers after risk-reducing surgery.
Surgical requirements. Obviously, if a surgery is to be risk-reducing, as much as possible of the tissue at risk should be removed. To do so effectively, the surgeon should be comfortable operating in the retroperitoneum so that the infundibulopelvic ligament can be ligated sufficiently proximal from the ovarian hilum to minimize the possibility of an ovarian remnant. Similarly, if a salpingo-oophorectomy without hysterectomy is to be performed, the fallopian tube should be amputated as close as possible to the uterine cornua (FIGURE 2).
Laparoscopy versus open surgery. RRSO can be performed using either a laparoscopic or open approach. The appropriate choice is best determined by the patient’s history, associated comorbid conditions, need for additional procedures, and experience of the surgeon. At our institution, in the absence of contraindications, we generally offer a laparoscopic approach due to its decreased morbidity.
Concomitant hysterectomy? An area of substantial controversy is whether the uterus should be removed at the time of RRSO. In most studies exploring this issue, hereditary breast-ovarian cancer syndrome does not appear to be associated with an increased risk of uterine cancer.24 However, there is concern that the portion of interstitial fallopian tube left behind after salpingo-oophorectomy may be at risk for malignant transformation.25,26
In our series, almost 90% of risk-reducing procedures were salpingo-oophorectomies without hysterectomy. If there is an additional benefit to concomitant hysterectomy, it has yet to be demonstrated by clinical trials.
In several studies, a patient’s level of anxiety was more important than objective cancer risk in the choice of RRSO.
Close pathologic scrutiny advised. Microscopic cancer may be found in 2% to 4% of RRSO specimens upon careful pathologic review.21,27,28 Thus, it is essential that the ovaries and fallopian tubes are sectioned in their entirety and examined by an experienced gynecologic pathologist to minimize the chance that microscopic cancer goes undetected.
It is not clear whether cytology should be routinely done at the time of risk-reducing surgery. A single report documents malignant cells in a woman with a BRCA1 mutation and no obvious foci of malignancy despite hysterectomy with bilateral salpingo-oophorectomy and staging.29 Pending further studies, we routinely send cytology for review.
FIGURE 2 Careful surgical ligation and division to eliminate residual tissue
Source: Devita VT Jr., Hellman S, Rosenberg SA, eds. Progress in Oncology 2003. 2004: Jones and Bartlett Publishers; Sudbury, Mass. Reprinted with permission.
When no BRCA mutation is present
Most of the data cited thus far apply to women with documented BRCA mutations. There is much less information about the relative risks and benefits of RRSO in women with a personal or family history of breast or ovarian cancer who lack a documented BRCA mutation.
Although RRSO may be appropriate for some of these women, in 2004 it is not the standard of care to recommend RRSO to all individuals with a personal or family history suggestive of an inherited predisposition to ovarian cancer. These patients are best managed by an interdisciplinary team of gynecologists, gynecologic oncologists, and clinical geneticists, all with experience caring for women who may have an inherited predisposition.
Is anxiety a factor?
We have limited information about the psychosocial impact of RRSO. Several studies have found that a patient’s level of anxiety is a more important factor than objective cancer risk in the decision to undergo RRSO.30,31 Unfortunately, we do not yet know whether the surgery successfully reduces these patients’ subjective concerns.
A recent study showed that risk-reducing surgery did not impair women’s overall health or psychological well-being.32 However, 20.7% of the women reported substantial cancer-related anxiety despite the risk-reducing surgery. This issue requires further investigation.
Is estrogen the best option for surgical menopause?
The role of hormone replacement after RRSO is unclear. The issue is important because many women considering salpingo-oophorectomy are in their late 30s or early 40s, when premature surgical menopause is a predictable result. Consequences can include considerable vasomotor and pelvic symptoms.
Preliminary data suggest that a woman’s satisfaction with RRSO depends in large part on its impact on sexual functioning.32 Urogenital symptoms that adversely affected sexual function, such as vaginal dryness and dyspareunia, were the most significant predictors of dissatisfaction with surgery.
Premature surgical menopause also has a substantial impact on osteoporosis risk, while its effect on heart disease remains uncertain.
While nonhormonal therapies can address each of these issues, we need more data on their long-term use. We counsel women considering RRSO that hormone replacement may be an option. We believe it is unlikely to reduce the efficacy of RRSO in preventing ovarian cancer, but it may reduce the protective effect of RRSO against subsequent breast cancer. Until further studies are available, we recommend that decisions regarding hormone replacement be individualized to the patient’s specific symptoms and personal history.
Not all insurers cover RRSO
One study explored insurance carriers’ policies about reimbursing risk-reducing surgical procedures and found that 10% to 11% of private insurers and 48% to 50% of governmental carriers had policies specifically denying coverage for such operations.
An additional 40% to 64% of insurers had no identifiable policy regarding these procedures in women with BRCA mutations.33 The authors speculated that, without identifiable policies, this critical health-care decision may be subject to arbitrary criteria that result in substantial variation.
When we recently investigated the reimbursement experience of women with BRCA mutations undergoing RRSO at our institution, we found that 97% of the procedures were reimbursed in full, less any applicable coinsurance and deductibles.34 Two important limitations of our study: It was conducted at a tertiary cancer center and was retrospective. It is not known if the findings reflect the experience of women with BRCA mutation who have risk-reducing surgery in other settings.
Unresolved issues
RRSO clearly has a role in preventing breast and ovarian cancer in women at inherited risk. However, several questions remain unanswered:
- Who is the best candidate?
- What is optimal timing of the procedure?
- What, if any, concomitant procedures should be performed?
- What is the role of hormone replacement after the surgery?
These issues will be best addressed through multicenter prospective trials, such as the one now being conducted by the Gynecologic Oncology Group.
Hope also remains that further research will improve serum and radiological detection of early ovarian cancer, and that basic research on the molecular etiology and progression of these cancers will ultimately render it unnecessary to remove organs at risk.
The authors report no financial relationships relevant to this article.
1. Liber AM. Ovarian cancer in a mother and five daughters. Arch Pathol. 1950;49:280-290.
2. Knudson AG, Jr. Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA. 1971;68:820-823.
3. Ford D, Easton DF, Stratton M, et al. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am J Hum Genet. 1998;62:676-689.
4. Risch HA, McLaughlin JR, Cole DE, et al. Prevalence and penetrance of germline BRCA1 and BRCA2 mutations in a population series of 649 women with ovarian cancer. Am J Hum Genet. 2001;68:700-710.
5. Oddoux C, Struewing JP, Clayton CM, et al. The carrier frequency of the BRCA2 6174delT mutation among Ashkenazi Jewish individuals is approximately 1%. Nat Genet. 1996;14:188-190.
6. Tonin P, Weber B, Offit K, et al. Frequency of recurrent BRCA1 and BRCA2 mutations in Ashkenazi Jewish breast cancer families. Nat Med. 1996;2:1179-1183.
7. Schinzinger A. Ueber Carcinoma Mammae. Verhandlungen der Deutschen Gesellschaft fur Chirurgie. 18th Kongress, Berlin, Apr 24–27, 1889. Berlin, Germany: Hirschwald; 1889:28[abstract].
8. Love RR, Philips J. Oophorectomy for breast cancer: history revisited. J Natl Cancer Inst. 2002;94:1433-1434.
9. Feinleib M. Breast cancer and artificial menopause: a cohort study. J Natl Cancer Inst. 1968;41:315-329.
10. Brinton LA, Schairer C, Hoover RN, Fraumeni JF, Jr. Menstrual factors and risk of breast cancer. Cancer Invest. 1988;6:245-254.
11. Tobacman JK, Greene MH, Tucker MA, Costa J, Kase R, Fraumeni JF, Jr. Intraabdominal carcinomatosis after prophylactic oophorectomy in ovarian-cancer-prone families. Lancet. 1982;2:795-797.
12. Piver MS, Jishi MF, Tsukada Y, Nava G. Primary peritoneal carcinoma after prophylactic oophorectomy in women with a family history of ovarian cancer. A report of the Gilda Radner Familial Ovarian Cancer Registry. Cancer. 1993;71:2751-2755.
13. Burke W, Daly M, Garber J, et al. Recommendations for follow-up care of individuals with an inherited predisposition to cancer. II. BRCA1 and BRCA2. Cancer Genetics Studies Consortium. JAMA. 1997;277:997-1003.
14. Schrag D, Kuntz KM, Garber JE, Weeks JC. Decision analysis—effects of prophylactic mastectomy and oophorectomy on life expectancy among women with BRCA1 or BRCA2 mutations. N Engl J Med. 1997;336:1465-1471.
15. Grann VR, Panageas KS, Whang W, Antman KH, Neugut AI. Decision analysis of prophylactic mastectomy and oophorectomy in BRCA1-positive or BRCA2-positive patients. J Clin Oncol. 1998;16:979-985.
16. Rebbeck TR, Levin AM, Eisen A, et al. Breast cancer risk after bilateral prophylactic oophorectomy in BRCA1 mutation carriers. J Natl Cancer Inst. 1999;91:1475-1479.
17. Eisen A, Rebbeck TR, Wood WC, Weber BL. Prophylactic surgery in women with a hereditary predisposition to breast and ovarian cancer. J Clin Oncol. 2000;18:1980-1995.
18. Kauff ND, Satagopan JM, Robson ME, et al. Risk-reducing salpingo-oophorectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med. 2002;346:1609-1615.
19. Rebbeck TR, Lynch HT, Neuhausen SL, et al. Prophylactic oophorectomy in carriers of BRCA1 or BRCA2 mutations. N Engl J Med. 2002;346:1616-1622.
20. Rutter JL, Wacholder S, Chetrit A, et al. Gynecologic surgeries and risk of ovarian cancer in women with BRCA1 and BRCA2 Ashkenazi founder mutations: an Israeli population-based case-control study. J Natl Cancer Inst. 2003;95:1072-1078.
21. Lu KH, Garber JE, Cramer DW, et al. Occult ovarian tumors in women with BRCA1 or BRCA2 mutations undergoing prophylactic oophorectomy. J Clin Oncol. 2000;18:2728-2732.
22. Lafferty HW, Angioli R, Rudolph J, Penalver MA. Ovarian remnant syndrome: experience at Jackson Memorial Hospital, University of Miami, 1985 through 1993. Am J Obstet Gynecol. 1996;174:641-645.
23. Schorge JO, Muto MG, Welch WR, et al. Molecular evidence for multifocal papillary serous carcinoma of the peritoneum in patients with germline BRCA1 mutations. J Natl Cancer Inst. 1998;90:841-845.
24. Levine DA, Lin O, Barakat RR, et al. Risk of endometrial carcinoma associated with BRCA mutation. Gynecol Oncol. 2001;80:395-398.
25. Paley PJ, Swisher EM, Garcia RL, et al. Occult cancer of the fallopian tube in BRCA-1 germline mutation carriers at prophylactic oophorectomy: a case for recommending hysterectomy at surgical prophylaxis. Gynecol Oncol. 2001;80:176-180.
26. Aziz S, Kuperstein G, Rosen B, et al. A genetic epidemiological study of carcinoma of the fallopian tube. Gynecol Oncol. 2001;80:341-345.
27. Colgan TJ, Murphy J, Cole DE, Narod S, Rosen B. Occult carcinoma in prophylactic oophorectomy specimens: prevalence and association with BRCA germline mutation status. Am J Surg Pathol. 2001;25:1283-1289.
28. Scheuer L, Kauff N, Robson M, et al. Outcome of preventive surgery and screening for breast and ovarian cancer in BRCA mutation carriers. J Clin Oncol. 2002;20:1260-1268.
29. Colgan TJ, Boerner SL, Murphy J, Cole DE, Narod S, Rosen B. Peritoneal lavage cytology: an assessment of its value during prophylactic oophorectomy. Gynecol Oncol. 2002;85:397-403.
30. Meiser B, Butow P, Barratt A, et al. Attitudes toward prophylactic oophorectomy and screening utilization in women at increased risk of developing hereditary breast/ovarian cancer. Gynecol Oncol. 1999;75:122-129.
31. Hurley KE, Miller SM, Costalas JW, Gillespie D, Daly MB. Anxiety/uncertainty reduction as a motivation for interest in prophylactic oophorectomy in women with a family history of ovarian cancer. J Womens Health Gend Based Med. 2001;10:189-199.
32. Robson M, Hensley M, Barakat R, et al. Quality of life in women at risk for ovarian cancer who have undergone risk-reducing oophorectomy. Gynecol Oncol. 2003;89:281-287.
33. Kuerer HM, Hwang ES, Anthony JP, et al. Current national health insurance coverage policies for breast and ovarian cancer prophylactic surgery. Ann Surg Oncol. 2000;7:325-332.
34. Kauff ND, Scheuer L, Robson ME, et al. Insurance reimbursement for risk-reducing mastectomy and oophorectomy in women with BRCA1 or BRCA2 mutations. Genet Med. 2001;3:422-425.
1. Liber AM. Ovarian cancer in a mother and five daughters. Arch Pathol. 1950;49:280-290.
2. Knudson AG, Jr. Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA. 1971;68:820-823.
3. Ford D, Easton DF, Stratton M, et al. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am J Hum Genet. 1998;62:676-689.
4. Risch HA, McLaughlin JR, Cole DE, et al. Prevalence and penetrance of germline BRCA1 and BRCA2 mutations in a population series of 649 women with ovarian cancer. Am J Hum Genet. 2001;68:700-710.
5. Oddoux C, Struewing JP, Clayton CM, et al. The carrier frequency of the BRCA2 6174delT mutation among Ashkenazi Jewish individuals is approximately 1%. Nat Genet. 1996;14:188-190.
6. Tonin P, Weber B, Offit K, et al. Frequency of recurrent BRCA1 and BRCA2 mutations in Ashkenazi Jewish breast cancer families. Nat Med. 1996;2:1179-1183.
7. Schinzinger A. Ueber Carcinoma Mammae. Verhandlungen der Deutschen Gesellschaft fur Chirurgie. 18th Kongress, Berlin, Apr 24–27, 1889. Berlin, Germany: Hirschwald; 1889:28[abstract].
8. Love RR, Philips J. Oophorectomy for breast cancer: history revisited. J Natl Cancer Inst. 2002;94:1433-1434.
9. Feinleib M. Breast cancer and artificial menopause: a cohort study. J Natl Cancer Inst. 1968;41:315-329.
10. Brinton LA, Schairer C, Hoover RN, Fraumeni JF, Jr. Menstrual factors and risk of breast cancer. Cancer Invest. 1988;6:245-254.
11. Tobacman JK, Greene MH, Tucker MA, Costa J, Kase R, Fraumeni JF, Jr. Intraabdominal carcinomatosis after prophylactic oophorectomy in ovarian-cancer-prone families. Lancet. 1982;2:795-797.
12. Piver MS, Jishi MF, Tsukada Y, Nava G. Primary peritoneal carcinoma after prophylactic oophorectomy in women with a family history of ovarian cancer. A report of the Gilda Radner Familial Ovarian Cancer Registry. Cancer. 1993;71:2751-2755.
13. Burke W, Daly M, Garber J, et al. Recommendations for follow-up care of individuals with an inherited predisposition to cancer. II. BRCA1 and BRCA2. Cancer Genetics Studies Consortium. JAMA. 1997;277:997-1003.
14. Schrag D, Kuntz KM, Garber JE, Weeks JC. Decision analysis—effects of prophylactic mastectomy and oophorectomy on life expectancy among women with BRCA1 or BRCA2 mutations. N Engl J Med. 1997;336:1465-1471.
15. Grann VR, Panageas KS, Whang W, Antman KH, Neugut AI. Decision analysis of prophylactic mastectomy and oophorectomy in BRCA1-positive or BRCA2-positive patients. J Clin Oncol. 1998;16:979-985.
16. Rebbeck TR, Levin AM, Eisen A, et al. Breast cancer risk after bilateral prophylactic oophorectomy in BRCA1 mutation carriers. J Natl Cancer Inst. 1999;91:1475-1479.
17. Eisen A, Rebbeck TR, Wood WC, Weber BL. Prophylactic surgery in women with a hereditary predisposition to breast and ovarian cancer. J Clin Oncol. 2000;18:1980-1995.
18. Kauff ND, Satagopan JM, Robson ME, et al. Risk-reducing salpingo-oophorectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med. 2002;346:1609-1615.
19. Rebbeck TR, Lynch HT, Neuhausen SL, et al. Prophylactic oophorectomy in carriers of BRCA1 or BRCA2 mutations. N Engl J Med. 2002;346:1616-1622.
20. Rutter JL, Wacholder S, Chetrit A, et al. Gynecologic surgeries and risk of ovarian cancer in women with BRCA1 and BRCA2 Ashkenazi founder mutations: an Israeli population-based case-control study. J Natl Cancer Inst. 2003;95:1072-1078.
21. Lu KH, Garber JE, Cramer DW, et al. Occult ovarian tumors in women with BRCA1 or BRCA2 mutations undergoing prophylactic oophorectomy. J Clin Oncol. 2000;18:2728-2732.
22. Lafferty HW, Angioli R, Rudolph J, Penalver MA. Ovarian remnant syndrome: experience at Jackson Memorial Hospital, University of Miami, 1985 through 1993. Am J Obstet Gynecol. 1996;174:641-645.
23. Schorge JO, Muto MG, Welch WR, et al. Molecular evidence for multifocal papillary serous carcinoma of the peritoneum in patients with germline BRCA1 mutations. J Natl Cancer Inst. 1998;90:841-845.
24. Levine DA, Lin O, Barakat RR, et al. Risk of endometrial carcinoma associated with BRCA mutation. Gynecol Oncol. 2001;80:395-398.
25. Paley PJ, Swisher EM, Garcia RL, et al. Occult cancer of the fallopian tube in BRCA-1 germline mutation carriers at prophylactic oophorectomy: a case for recommending hysterectomy at surgical prophylaxis. Gynecol Oncol. 2001;80:176-180.
26. Aziz S, Kuperstein G, Rosen B, et al. A genetic epidemiological study of carcinoma of the fallopian tube. Gynecol Oncol. 2001;80:341-345.
27. Colgan TJ, Murphy J, Cole DE, Narod S, Rosen B. Occult carcinoma in prophylactic oophorectomy specimens: prevalence and association with BRCA germline mutation status. Am J Surg Pathol. 2001;25:1283-1289.
28. Scheuer L, Kauff N, Robson M, et al. Outcome of preventive surgery and screening for breast and ovarian cancer in BRCA mutation carriers. J Clin Oncol. 2002;20:1260-1268.
29. Colgan TJ, Boerner SL, Murphy J, Cole DE, Narod S, Rosen B. Peritoneal lavage cytology: an assessment of its value during prophylactic oophorectomy. Gynecol Oncol. 2002;85:397-403.
30. Meiser B, Butow P, Barratt A, et al. Attitudes toward prophylactic oophorectomy and screening utilization in women at increased risk of developing hereditary breast/ovarian cancer. Gynecol Oncol. 1999;75:122-129.
31. Hurley KE, Miller SM, Costalas JW, Gillespie D, Daly MB. Anxiety/uncertainty reduction as a motivation for interest in prophylactic oophorectomy in women with a family history of ovarian cancer. J Womens Health Gend Based Med. 2001;10:189-199.
32. Robson M, Hensley M, Barakat R, et al. Quality of life in women at risk for ovarian cancer who have undergone risk-reducing oophorectomy. Gynecol Oncol. 2003;89:281-287.
33. Kuerer HM, Hwang ES, Anthony JP, et al. Current national health insurance coverage policies for breast and ovarian cancer prophylactic surgery. Ann Surg Oncol. 2000;7:325-332.
34. Kauff ND, Scheuer L, Robson ME, et al. Insurance reimbursement for risk-reducing mastectomy and oophorectomy in women with BRCA1 or BRCA2 mutations. Genet Med. 2001;3:422-425.
Laparoscopic tissue extraction: Pros and cons of 4 techniques
- In appropriately selected patients, the ability to easily and skillfully remove tissue during laparoscopy facilitates patient recovery and healing and limits hospitalization time.
- Even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators, and the size of masses is less significant than with “manual” approaches.
Novel surgical devices and techniques have transformed operative laparoscopy by improving the inefficiencies in tissue extraction that limited early acceptance.
In the beginning, it was relatively easy to isolate a myoma and dissect it from the underlying myometrium, but it took hours to extract the tissue using a hand-held morcellator. This article focuses on the 4 techniques commonly used today, as well as the products that make them possible.
In appropriately selected patients, the ability to remove tissue through any of these methods facilitates patient recovery and healing and limits hospitalization time.
Posterior colpotomy
In the 1980s and early 1990s, it was common for tissue to be extracted through a posterior colpotomy. This is not surprising given that gynecologists were trained to perform transvaginal tubal ligation and to use colpotomies when incising and draining tubo-ovarian abscesses—not to mention our ease in entering the posterior cul-de-sac during vaginal surgery.
The elasticity of the vagina facilitates removal of fairly sizeable masses. Large ovarian cysts or masses can be brought to the cul-de-sac and incised and drained in a manner that markedly reduces the risk of intraperitoneal spillage.
There are disadvantages, however. For example, if the surgeon wants to maintain laparoscopic visualization once the colpotomy has been made, the tissue to be removed must be grasped and brought toward the opening to plug the defect and maintain pneumoperitoneum.
This may not be particularly problematic if there is only 1 mass to be removed, but it can be troublesome if there are several. An option is to place the masses in the posterior cul-de-sac so they can be readily grasped once the posterior colpotomy has been made.
One conceptual concern is the issue of subsequent adhesion formation, especially in patients desiring fertility. Unfortunately, no substantive trials exist to better answer this question.
Removal through the trocar or trocar site
Although some physicians still remove tissue through a posterior colpotomy, most have abandoned that approach in favor of extraction through a primary or lateral laparoscopic port. Indeed, this is the simplest technique for extracting tissue. I often change from a 10-mm laparoscope to a 5-mm instrument, placing the smaller endoscope in one of the lower ports and removing tissue under direct visualization through the 10- or 11-mm infraumbilical port.
If a cystic mass placed in a laparoscopic bag is too large to be removed, carefully aspirate it with a large-gauge needle.
Trapped tissue. One potential problem is the trapping of tissue in trocars that contain a flap valve. If this occurs, remove the trocar, clear the tissue, and replace the trocar in the original site using a blunt instrument such as the 10-mm laparoscope. Do not use the sharp inner blade to replace this port, as it is unduly risky.
For large masses, remove the port to create extra space. It also may be necessary to enlarge the skin or the fascial incision using a blunt instrument such as forceps.
Before the widespread availability of laparoscopic bags, tissue extraction was generally performed in this manner.
Risks include spillage of cyst contents during extraction and development of a hernia secondary to the wider disruption of fascia. This risk is particularly high in the infraumbilical area, which is inherently weak to begin with. It is thus critical—in any methodology—that the fascia be appropriately closed.
Laparoscopic bags
Many of the laparoscopic bags now widely available are easily opened once they have been placed in the abdomen, though some must be opened with graspers after the bag is positioned in the peritoneal cavity. Laparoscopic bags have greater utility when the extracted tissue is soft, such as with a dermoid cyst or ovary. Dense tissue is more difficult to manage.
Some surgeons fashion their own bags using sterile gloves or baggies.
Durability. The bags vary in their ability to withstand manipulation and puncture. For example, one type of nylon bag has a polyurethane inner coating and drawstring closure, making it quite durable. It also comes in a range of sizes, allowing the surgeon to choose the bag most suitable for the mass being removed.
To use a laparoscopic bag, insert it through the infraumbilical trocar and place the mass inside it. Then remove the trocar to provide maximal room for the mass to be extracted.
If the mass is cystic and too large to be removed, carefully aspirate it with a largegauge needle, taking care not to puncture the bag. Otherwise, morcellate the mass in the sac and remove it piecemeal, allowing no spillage of contents.
This may be performed under laparoscopic visualization through the lower ancillary trocars or trocar site. If a larger port has been placed—or there is a clinical need for one—tissue extraction also could be performed through the lower port.
Risks include bag breakage and potential spillage. In addition, it sometimes is necessary to change to a larger bag.
Morcellators
Early morcellators were hand-held, requiring the operator to continuously bite into the tissue and remove the small fragments. While this approach was effective for soft tissues and small myomas, it was ineffective for larger or more solid masses.
“Orange peel” technique. Scissors have been used to achieve the same effect as the handheld morcellator. Harrith Hasson described the “orange peel” technique, in which the surgeon uses scissors to peel away the tissue as one would peel an orange.1 The long, thin strips of tissue then can be extracted through the trocar. Unfortunately, laparoscopic scissors are often too small or dull to adequately incise larger fibroids.
Automatic morcellators have markedly enhanced our ability to perform laparoscopic myomectomy and similar procedures. They also have had a strong impact on nongynecologic procedures such as splenectomy or nephrectomy, in which large amounts of tissue must be removed. Although these devices are costly, the time savings associated with their use are significant. Current devices range from disposable to semidisposable and are available in a wide variety of sizes.
Even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators.
Hasson’s orange-peel technique also can be employed with automatic morcellators. This allows long, thin strips of tissue to be removed while facilitating constant visualization of anatomy surrounding the tissue being extracted.
An alternative is making multiple “through-passes” into the myoma using the morcellator. In this method, the strips of tissue obtained will be smaller and the myoma will develop a Swiss-cheese appearance. Note that this approach takes longer and may increase the number of myoma fragments that fall into the pelvis and need to be removed.
Effective for a range of masses. Not surprisingly, even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators, and the size of masses is less significant than with “manual” approaches. Nevertheless, it is critical that the surgeon maintain constant visualization and that tissue be brought toward the morcellator and away from underlying structures (FIGURE).
Do not move the morcellator toward the tissue. Because of its sharpness, the automatic morcellator will cut through vital structures as easily as it penetrates fibroids.
FIGURE Automatic morcellation: Move excised tissue toward device
To prevent injury, tissue should be brought toward the morcellator and away from underlying structures. Do not move the morcellator toward the tissue or vital structures may be cut.
Spillage
An early and continuing concern regarding ovarian cystectomy or oophorectomy is spillage of the mass’s contents into the peritoneal cavity. This is more of an issue in the case of borderline or malignant ovarian lesions or mucinous or dermoid ovarian cysts. In fact, nowhere is there more contention than over the clinical ramifications of spillage in the case of malignancy.
Mixed data on impact of spillage. Clinical data suggest that the impact of spillage is inconsequential, whereas other evidence suggests a worsening prognosis.2-5 In the event of spillage, most gynecologic oncologists would convert an ovarian cancer patient with a 1A or 1B staged lesion to stage 1C and would likely administer chemotherapy.
Concern about spillage of a mucinous or dermoid cyst centers on the theoretical risk of pseudomyxoma peritonei or, in the case of a teratoma, chemical peritonitis. Some surgeons routinely enter dermoids and intentionally spill the contents.6,7 Of note, we lack significant case series of ensuing infections or problems with this technique. Still, removing an intact cyst negates this issue and expedites surgery, eliminating the need to irrigate the abdomen and pelvis with large quantities of fluid.
Ectopic pregnancy has also been a concern, as there have been reports of chorionic tissue being disseminated in the abdomen and pelvis during laparoscopic procedures.8
Nowhere is there more contention than over the clinical ramifications of spillage in the case of malignancy.
Patient selection
Preoperative evaluation is a critical component of patient selection. A thorough ultrasound examination can help determine who is and who is not an appropriate candidate for laparoscopic management.
Cases that suggest a high risk of ovarian malignancy may be best managed in the traditional manner, as may patients with a large number of myomas or other compounding factors.
Dr. Bieber reports no financial relationships relevant to this article.
1. Hasson HM, Rotman C, Rana N, Sistos F, Dmowski WP. Laparoscopic myomectomy. Obstet Gynecol. 1992;80:884-888.
2. Mayer C, Miller DM, Ehlen TG. Peritoneal implantation of squamous cell carcinoma following rupture of a dermoid cyst during laparoscopic removal. Gynecol Oncol. 2002;84:180-183.
3. Kodama S, Tanaka K, Tokunaga A, et al. Multivariate analysis of prognostic fators in patients with ovarian cancer stage I and II. Int J Gynaecol Obstet. 1997;56:147-153.
4. Mizuno M, Kikkawa F, Shibata K, et al. Long-term prognosis of stage I ovarian carcinoma. Prognostic importance of intraoperative rupture. Oncology. 2003;65:29-36.
5. Vergote I, De Brabanter J, Fyles A, et al. Prognostic importance of degree of differentiation and cyst rupture in stage I invasive epithelial ovarian carcinoma. Lancet. 2001;357:176-182.
6. Zanetta G, Ferrari L, Mignini-Renzini M, Vignali M, Fadini R. Laparoscopic excision of ovarian dermoid cysts with controlled intraoperative spillage. Safety andeffectiveness. J Reprod Med. 1999;44:815-820.
7. Mecke H, Sawas V. Laparoscopic surgery of dermoid cysts—intraoperative spillage and complications. Eur J Obstet Gynecol Reprod Biol. 2001;96:80-84.
8. Billieux MH, Petignat P, Anguenot JL, Campana A, Bischof P. Early and late halflife of human chorionic gonadotropin as a predictor of persistent trophoblast after laparoscopic conservative surgery for tubal pregnancy. Acta Obstet Gynecol Scand. 2003;82:550-555.
- In appropriately selected patients, the ability to easily and skillfully remove tissue during laparoscopy facilitates patient recovery and healing and limits hospitalization time.
- Even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators, and the size of masses is less significant than with “manual” approaches.
Novel surgical devices and techniques have transformed operative laparoscopy by improving the inefficiencies in tissue extraction that limited early acceptance.
In the beginning, it was relatively easy to isolate a myoma and dissect it from the underlying myometrium, but it took hours to extract the tissue using a hand-held morcellator. This article focuses on the 4 techniques commonly used today, as well as the products that make them possible.
In appropriately selected patients, the ability to remove tissue through any of these methods facilitates patient recovery and healing and limits hospitalization time.
Posterior colpotomy
In the 1980s and early 1990s, it was common for tissue to be extracted through a posterior colpotomy. This is not surprising given that gynecologists were trained to perform transvaginal tubal ligation and to use colpotomies when incising and draining tubo-ovarian abscesses—not to mention our ease in entering the posterior cul-de-sac during vaginal surgery.
The elasticity of the vagina facilitates removal of fairly sizeable masses. Large ovarian cysts or masses can be brought to the cul-de-sac and incised and drained in a manner that markedly reduces the risk of intraperitoneal spillage.
There are disadvantages, however. For example, if the surgeon wants to maintain laparoscopic visualization once the colpotomy has been made, the tissue to be removed must be grasped and brought toward the opening to plug the defect and maintain pneumoperitoneum.
This may not be particularly problematic if there is only 1 mass to be removed, but it can be troublesome if there are several. An option is to place the masses in the posterior cul-de-sac so they can be readily grasped once the posterior colpotomy has been made.
One conceptual concern is the issue of subsequent adhesion formation, especially in patients desiring fertility. Unfortunately, no substantive trials exist to better answer this question.
Removal through the trocar or trocar site
Although some physicians still remove tissue through a posterior colpotomy, most have abandoned that approach in favor of extraction through a primary or lateral laparoscopic port. Indeed, this is the simplest technique for extracting tissue. I often change from a 10-mm laparoscope to a 5-mm instrument, placing the smaller endoscope in one of the lower ports and removing tissue under direct visualization through the 10- or 11-mm infraumbilical port.
If a cystic mass placed in a laparoscopic bag is too large to be removed, carefully aspirate it with a large-gauge needle.
Trapped tissue. One potential problem is the trapping of tissue in trocars that contain a flap valve. If this occurs, remove the trocar, clear the tissue, and replace the trocar in the original site using a blunt instrument such as the 10-mm laparoscope. Do not use the sharp inner blade to replace this port, as it is unduly risky.
For large masses, remove the port to create extra space. It also may be necessary to enlarge the skin or the fascial incision using a blunt instrument such as forceps.
Before the widespread availability of laparoscopic bags, tissue extraction was generally performed in this manner.
Risks include spillage of cyst contents during extraction and development of a hernia secondary to the wider disruption of fascia. This risk is particularly high in the infraumbilical area, which is inherently weak to begin with. It is thus critical—in any methodology—that the fascia be appropriately closed.
Laparoscopic bags
Many of the laparoscopic bags now widely available are easily opened once they have been placed in the abdomen, though some must be opened with graspers after the bag is positioned in the peritoneal cavity. Laparoscopic bags have greater utility when the extracted tissue is soft, such as with a dermoid cyst or ovary. Dense tissue is more difficult to manage.
Some surgeons fashion their own bags using sterile gloves or baggies.
Durability. The bags vary in their ability to withstand manipulation and puncture. For example, one type of nylon bag has a polyurethane inner coating and drawstring closure, making it quite durable. It also comes in a range of sizes, allowing the surgeon to choose the bag most suitable for the mass being removed.
To use a laparoscopic bag, insert it through the infraumbilical trocar and place the mass inside it. Then remove the trocar to provide maximal room for the mass to be extracted.
If the mass is cystic and too large to be removed, carefully aspirate it with a largegauge needle, taking care not to puncture the bag. Otherwise, morcellate the mass in the sac and remove it piecemeal, allowing no spillage of contents.
This may be performed under laparoscopic visualization through the lower ancillary trocars or trocar site. If a larger port has been placed—or there is a clinical need for one—tissue extraction also could be performed through the lower port.
Risks include bag breakage and potential spillage. In addition, it sometimes is necessary to change to a larger bag.
Morcellators
Early morcellators were hand-held, requiring the operator to continuously bite into the tissue and remove the small fragments. While this approach was effective for soft tissues and small myomas, it was ineffective for larger or more solid masses.
“Orange peel” technique. Scissors have been used to achieve the same effect as the handheld morcellator. Harrith Hasson described the “orange peel” technique, in which the surgeon uses scissors to peel away the tissue as one would peel an orange.1 The long, thin strips of tissue then can be extracted through the trocar. Unfortunately, laparoscopic scissors are often too small or dull to adequately incise larger fibroids.
Automatic morcellators have markedly enhanced our ability to perform laparoscopic myomectomy and similar procedures. They also have had a strong impact on nongynecologic procedures such as splenectomy or nephrectomy, in which large amounts of tissue must be removed. Although these devices are costly, the time savings associated with their use are significant. Current devices range from disposable to semidisposable and are available in a wide variety of sizes.
Even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators.
Hasson’s orange-peel technique also can be employed with automatic morcellators. This allows long, thin strips of tissue to be removed while facilitating constant visualization of anatomy surrounding the tissue being extracted.
An alternative is making multiple “through-passes” into the myoma using the morcellator. In this method, the strips of tissue obtained will be smaller and the myoma will develop a Swiss-cheese appearance. Note that this approach takes longer and may increase the number of myoma fragments that fall into the pelvis and need to be removed.
Effective for a range of masses. Not surprisingly, even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators, and the size of masses is less significant than with “manual” approaches. Nevertheless, it is critical that the surgeon maintain constant visualization and that tissue be brought toward the morcellator and away from underlying structures (FIGURE).
Do not move the morcellator toward the tissue. Because of its sharpness, the automatic morcellator will cut through vital structures as easily as it penetrates fibroids.
FIGURE Automatic morcellation: Move excised tissue toward device
To prevent injury, tissue should be brought toward the morcellator and away from underlying structures. Do not move the morcellator toward the tissue or vital structures may be cut.
Spillage
An early and continuing concern regarding ovarian cystectomy or oophorectomy is spillage of the mass’s contents into the peritoneal cavity. This is more of an issue in the case of borderline or malignant ovarian lesions or mucinous or dermoid ovarian cysts. In fact, nowhere is there more contention than over the clinical ramifications of spillage in the case of malignancy.
Mixed data on impact of spillage. Clinical data suggest that the impact of spillage is inconsequential, whereas other evidence suggests a worsening prognosis.2-5 In the event of spillage, most gynecologic oncologists would convert an ovarian cancer patient with a 1A or 1B staged lesion to stage 1C and would likely administer chemotherapy.
Concern about spillage of a mucinous or dermoid cyst centers on the theoretical risk of pseudomyxoma peritonei or, in the case of a teratoma, chemical peritonitis. Some surgeons routinely enter dermoids and intentionally spill the contents.6,7 Of note, we lack significant case series of ensuing infections or problems with this technique. Still, removing an intact cyst negates this issue and expedites surgery, eliminating the need to irrigate the abdomen and pelvis with large quantities of fluid.
Ectopic pregnancy has also been a concern, as there have been reports of chorionic tissue being disseminated in the abdomen and pelvis during laparoscopic procedures.8
Nowhere is there more contention than over the clinical ramifications of spillage in the case of malignancy.
Patient selection
Preoperative evaluation is a critical component of patient selection. A thorough ultrasound examination can help determine who is and who is not an appropriate candidate for laparoscopic management.
Cases that suggest a high risk of ovarian malignancy may be best managed in the traditional manner, as may patients with a large number of myomas or other compounding factors.
Dr. Bieber reports no financial relationships relevant to this article.
- In appropriately selected patients, the ability to easily and skillfully remove tissue during laparoscopy facilitates patient recovery and healing and limits hospitalization time.
- Even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators, and the size of masses is less significant than with “manual” approaches.
Novel surgical devices and techniques have transformed operative laparoscopy by improving the inefficiencies in tissue extraction that limited early acceptance.
In the beginning, it was relatively easy to isolate a myoma and dissect it from the underlying myometrium, but it took hours to extract the tissue using a hand-held morcellator. This article focuses on the 4 techniques commonly used today, as well as the products that make them possible.
In appropriately selected patients, the ability to remove tissue through any of these methods facilitates patient recovery and healing and limits hospitalization time.
Posterior colpotomy
In the 1980s and early 1990s, it was common for tissue to be extracted through a posterior colpotomy. This is not surprising given that gynecologists were trained to perform transvaginal tubal ligation and to use colpotomies when incising and draining tubo-ovarian abscesses—not to mention our ease in entering the posterior cul-de-sac during vaginal surgery.
The elasticity of the vagina facilitates removal of fairly sizeable masses. Large ovarian cysts or masses can be brought to the cul-de-sac and incised and drained in a manner that markedly reduces the risk of intraperitoneal spillage.
There are disadvantages, however. For example, if the surgeon wants to maintain laparoscopic visualization once the colpotomy has been made, the tissue to be removed must be grasped and brought toward the opening to plug the defect and maintain pneumoperitoneum.
This may not be particularly problematic if there is only 1 mass to be removed, but it can be troublesome if there are several. An option is to place the masses in the posterior cul-de-sac so they can be readily grasped once the posterior colpotomy has been made.
One conceptual concern is the issue of subsequent adhesion formation, especially in patients desiring fertility. Unfortunately, no substantive trials exist to better answer this question.
Removal through the trocar or trocar site
Although some physicians still remove tissue through a posterior colpotomy, most have abandoned that approach in favor of extraction through a primary or lateral laparoscopic port. Indeed, this is the simplest technique for extracting tissue. I often change from a 10-mm laparoscope to a 5-mm instrument, placing the smaller endoscope in one of the lower ports and removing tissue under direct visualization through the 10- or 11-mm infraumbilical port.
If a cystic mass placed in a laparoscopic bag is too large to be removed, carefully aspirate it with a large-gauge needle.
Trapped tissue. One potential problem is the trapping of tissue in trocars that contain a flap valve. If this occurs, remove the trocar, clear the tissue, and replace the trocar in the original site using a blunt instrument such as the 10-mm laparoscope. Do not use the sharp inner blade to replace this port, as it is unduly risky.
For large masses, remove the port to create extra space. It also may be necessary to enlarge the skin or the fascial incision using a blunt instrument such as forceps.
Before the widespread availability of laparoscopic bags, tissue extraction was generally performed in this manner.
Risks include spillage of cyst contents during extraction and development of a hernia secondary to the wider disruption of fascia. This risk is particularly high in the infraumbilical area, which is inherently weak to begin with. It is thus critical—in any methodology—that the fascia be appropriately closed.
Laparoscopic bags
Many of the laparoscopic bags now widely available are easily opened once they have been placed in the abdomen, though some must be opened with graspers after the bag is positioned in the peritoneal cavity. Laparoscopic bags have greater utility when the extracted tissue is soft, such as with a dermoid cyst or ovary. Dense tissue is more difficult to manage.
Some surgeons fashion their own bags using sterile gloves or baggies.
Durability. The bags vary in their ability to withstand manipulation and puncture. For example, one type of nylon bag has a polyurethane inner coating and drawstring closure, making it quite durable. It also comes in a range of sizes, allowing the surgeon to choose the bag most suitable for the mass being removed.
To use a laparoscopic bag, insert it through the infraumbilical trocar and place the mass inside it. Then remove the trocar to provide maximal room for the mass to be extracted.
If the mass is cystic and too large to be removed, carefully aspirate it with a largegauge needle, taking care not to puncture the bag. Otherwise, morcellate the mass in the sac and remove it piecemeal, allowing no spillage of contents.
This may be performed under laparoscopic visualization through the lower ancillary trocars or trocar site. If a larger port has been placed—or there is a clinical need for one—tissue extraction also could be performed through the lower port.
Risks include bag breakage and potential spillage. In addition, it sometimes is necessary to change to a larger bag.
Morcellators
Early morcellators were hand-held, requiring the operator to continuously bite into the tissue and remove the small fragments. While this approach was effective for soft tissues and small myomas, it was ineffective for larger or more solid masses.
“Orange peel” technique. Scissors have been used to achieve the same effect as the handheld morcellator. Harrith Hasson described the “orange peel” technique, in which the surgeon uses scissors to peel away the tissue as one would peel an orange.1 The long, thin strips of tissue then can be extracted through the trocar. Unfortunately, laparoscopic scissors are often too small or dull to adequately incise larger fibroids.
Automatic morcellators have markedly enhanced our ability to perform laparoscopic myomectomy and similar procedures. They also have had a strong impact on nongynecologic procedures such as splenectomy or nephrectomy, in which large amounts of tissue must be removed. Although these devices are costly, the time savings associated with their use are significant. Current devices range from disposable to semidisposable and are available in a wide variety of sizes.
Even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators.
Hasson’s orange-peel technique also can be employed with automatic morcellators. This allows long, thin strips of tissue to be removed while facilitating constant visualization of anatomy surrounding the tissue being extracted.
An alternative is making multiple “through-passes” into the myoma using the morcellator. In this method, the strips of tissue obtained will be smaller and the myoma will develop a Swiss-cheese appearance. Note that this approach takes longer and may increase the number of myoma fragments that fall into the pelvis and need to be removed.
Effective for a range of masses. Not surprisingly, even dense tissues such as partially calcified leiomyomata are readily removed with automatic morcellators, and the size of masses is less significant than with “manual” approaches. Nevertheless, it is critical that the surgeon maintain constant visualization and that tissue be brought toward the morcellator and away from underlying structures (FIGURE).
Do not move the morcellator toward the tissue. Because of its sharpness, the automatic morcellator will cut through vital structures as easily as it penetrates fibroids.
FIGURE Automatic morcellation: Move excised tissue toward device
To prevent injury, tissue should be brought toward the morcellator and away from underlying structures. Do not move the morcellator toward the tissue or vital structures may be cut.
Spillage
An early and continuing concern regarding ovarian cystectomy or oophorectomy is spillage of the mass’s contents into the peritoneal cavity. This is more of an issue in the case of borderline or malignant ovarian lesions or mucinous or dermoid ovarian cysts. In fact, nowhere is there more contention than over the clinical ramifications of spillage in the case of malignancy.
Mixed data on impact of spillage. Clinical data suggest that the impact of spillage is inconsequential, whereas other evidence suggests a worsening prognosis.2-5 In the event of spillage, most gynecologic oncologists would convert an ovarian cancer patient with a 1A or 1B staged lesion to stage 1C and would likely administer chemotherapy.
Concern about spillage of a mucinous or dermoid cyst centers on the theoretical risk of pseudomyxoma peritonei or, in the case of a teratoma, chemical peritonitis. Some surgeons routinely enter dermoids and intentionally spill the contents.6,7 Of note, we lack significant case series of ensuing infections or problems with this technique. Still, removing an intact cyst negates this issue and expedites surgery, eliminating the need to irrigate the abdomen and pelvis with large quantities of fluid.
Ectopic pregnancy has also been a concern, as there have been reports of chorionic tissue being disseminated in the abdomen and pelvis during laparoscopic procedures.8
Nowhere is there more contention than over the clinical ramifications of spillage in the case of malignancy.
Patient selection
Preoperative evaluation is a critical component of patient selection. A thorough ultrasound examination can help determine who is and who is not an appropriate candidate for laparoscopic management.
Cases that suggest a high risk of ovarian malignancy may be best managed in the traditional manner, as may patients with a large number of myomas or other compounding factors.
Dr. Bieber reports no financial relationships relevant to this article.
1. Hasson HM, Rotman C, Rana N, Sistos F, Dmowski WP. Laparoscopic myomectomy. Obstet Gynecol. 1992;80:884-888.
2. Mayer C, Miller DM, Ehlen TG. Peritoneal implantation of squamous cell carcinoma following rupture of a dermoid cyst during laparoscopic removal. Gynecol Oncol. 2002;84:180-183.
3. Kodama S, Tanaka K, Tokunaga A, et al. Multivariate analysis of prognostic fators in patients with ovarian cancer stage I and II. Int J Gynaecol Obstet. 1997;56:147-153.
4. Mizuno M, Kikkawa F, Shibata K, et al. Long-term prognosis of stage I ovarian carcinoma. Prognostic importance of intraoperative rupture. Oncology. 2003;65:29-36.
5. Vergote I, De Brabanter J, Fyles A, et al. Prognostic importance of degree of differentiation and cyst rupture in stage I invasive epithelial ovarian carcinoma. Lancet. 2001;357:176-182.
6. Zanetta G, Ferrari L, Mignini-Renzini M, Vignali M, Fadini R. Laparoscopic excision of ovarian dermoid cysts with controlled intraoperative spillage. Safety andeffectiveness. J Reprod Med. 1999;44:815-820.
7. Mecke H, Sawas V. Laparoscopic surgery of dermoid cysts—intraoperative spillage and complications. Eur J Obstet Gynecol Reprod Biol. 2001;96:80-84.
8. Billieux MH, Petignat P, Anguenot JL, Campana A, Bischof P. Early and late halflife of human chorionic gonadotropin as a predictor of persistent trophoblast after laparoscopic conservative surgery for tubal pregnancy. Acta Obstet Gynecol Scand. 2003;82:550-555.
1. Hasson HM, Rotman C, Rana N, Sistos F, Dmowski WP. Laparoscopic myomectomy. Obstet Gynecol. 1992;80:884-888.
2. Mayer C, Miller DM, Ehlen TG. Peritoneal implantation of squamous cell carcinoma following rupture of a dermoid cyst during laparoscopic removal. Gynecol Oncol. 2002;84:180-183.
3. Kodama S, Tanaka K, Tokunaga A, et al. Multivariate analysis of prognostic fators in patients with ovarian cancer stage I and II. Int J Gynaecol Obstet. 1997;56:147-153.
4. Mizuno M, Kikkawa F, Shibata K, et al. Long-term prognosis of stage I ovarian carcinoma. Prognostic importance of intraoperative rupture. Oncology. 2003;65:29-36.
5. Vergote I, De Brabanter J, Fyles A, et al. Prognostic importance of degree of differentiation and cyst rupture in stage I invasive epithelial ovarian carcinoma. Lancet. 2001;357:176-182.
6. Zanetta G, Ferrari L, Mignini-Renzini M, Vignali M, Fadini R. Laparoscopic excision of ovarian dermoid cysts with controlled intraoperative spillage. Safety andeffectiveness. J Reprod Med. 1999;44:815-820.
7. Mecke H, Sawas V. Laparoscopic surgery of dermoid cysts—intraoperative spillage and complications. Eur J Obstet Gynecol Reprod Biol. 2001;96:80-84.
8. Billieux MH, Petignat P, Anguenot JL, Campana A, Bischof P. Early and late halflife of human chorionic gonadotropin as a predictor of persistent trophoblast after laparoscopic conservative surgery for tubal pregnancy. Acta Obstet Gynecol Scand. 2003;82:550-555.
A novel minilaparotomy approach for large ovarian cysts
- Make a cruciate incision by incising the skin transversely and the anterior rectus fascia vertically.
- Insert a soft, sleeved, self-retaining retractor.
- Using a surgical adhesive, glue a large plastic wound dressing to the surface of the cyst to prevent leakage of contents into the abdominal cavity.
- Aspirate the cyst until it collapses and can be delivered, with the ovary, through the abdominal incision.
- After performing an extracorporeal cystectomy and/or adnexectomy, return the repaired ovary to the abdominal cavity.
Although laparotomy is still considered the standard for ovarian cyst removal, over the past 15 years minimally invasive surgery has gained wider acceptance in cases where preoperative assessment suggests an adnexal mass is benign.
Unfortunately, minimally invasive management of a large ovarian cyst (greater than 10 cm) is particularly challenging for several reasons:
- The cyst can rupture and spill its contents into the peritoneum,
- the cyst’s size limits the surgical field, and
- an unexpected malignancy may be revealed.
An innovative minilaparotomy technique for the removal of benign ovarian cysts offers the advantages of laparoscopic and laparoscopic-assisted procedures while bypassing the major disadvantages: the necessity for specialized and expensive equipment, lengthy operative time, and long learning curves.1 (The minimally invasive procedures currently available for the treatment of ovarian cysts include laparoscopic cystectomy, laparoscopic-assisted minilaparotomy cystectomy, laparoscopic-assisted vaginal cystectomy, combined percutaneous ultrasound cyst aspiration and laparoscopic cystectomy, transvaginal cystectomy, and the traditional minilaparotomy cystectomy.2-10)
The procedure is faster, less expensive, carries fewer potential risks than traditional alternatives, and offers these advantages:
- can be performed under regional anesthesia
- relies on standard open techniques
- uses inexpensive instrumentation
- is easy to learn
- can be used for very large cysts
- eliminates the risk of intraperitoneal spillage of cyst contents
- offers similar postoperative convalescence and mean time to return to work as laparoscopic or laparoscopic-assisted management of large ovarian cysts
General Ob/Gyns—not gynecologic oncologists—perform most surgeries on patients with adnexal masses, since ovarian cancer is relatively uncommon in the absence of preoperative risk factors for malignancy. Our approach offers an appealing option to Ob/Gyns reluctant to abandon routine traditional laparotomy for such ovarian cysts.
Selecting the right patient
Adequate preoperative assessment diminishes the risk of unexpected malignancy in a patient undergoing surgery for an ovarian mass to less than 1%.4 At this time, the combination of menopausal status, cancer antigen (CA) 125 level, physical examination, and ultrasound is the best strategy for evaluating the patient with an ovarian cyst.11
Signs of malignancy. Ultrasound features that suggest malignancy include irregular borders, thick septa, solid areas, internal and external excrescences, matted bowel, and ascites. Benign cysts, on the other hand, are usually unilateral and have regular borders, thin septa, no solid areas, and no internal excrescences.4 The measurement of blood flow within the mass by color Doppler may improve the accuracy of ultrasound in differentiating benign from malignant cysts.4,12
On physical examination, an adnexal mass that is fixed, irregular, or solid also suggests a neoplasm. An elevated CA 125 combined with a complex adnexal mass is likely to be associated with malignancy. The test is even more specific in postmenopausal women with adnexal masses.4,12
However, plasma levels of CA 125 also can be elevated in several benign gynecologic conditions such as endometriosis, simple ovarian cysts, pelvic inflammatory disease, ovarian torsion, fibroids, and in physiologic conditions such as menstruation and pregnancy.13
Anticipate the need to convert to laparotomy. Every patient’s surgical consent should include a possible conversion to laparotomy. To avoid incomplete surgical treatment and significant delays in proper therapy, a gynecologic surgeon experienced in the management of ovarian cancer should be readily available, in the event an unexpected malignancy is encountered. Ideally, the staging surgery and definite treatment should be performed at the time of initial minilaparotomy. Comprehensive surgical staging and treatment include thorough exploration of the pelvis and abdomen, omentectomy, pelvic and paraaortic lymph node sampling, multiple peritoneal biopsies and washings, bilateral salpingo-oophorectomy, hysterectomy, and debulking, when indicated.
Prepare for surgery with position, incision, and retraction
Before beginning, it is crucial to correctly position the patient, make the appropriate incision, and insert the right retractor.1
Position. After administering regional or general anesthesia, place the patient in a modified lithotomy, as for laparoscopic surgery. Tuck the arms alongside the torso and place the legs in boot stirrups. Avoid hip flexion and allow adequate thigh abduction to expose the vagina. Perform a careful pelvic examination to determine the size and mobility of the adnexal mass.
When properly placed, this retractor creates an atraumatic, circular area of self-retraction, enabling superior exposure
Place an indwelling transurethral catheter, and pass a sturdy, hinged uterine manipulator such as the Pelosi Uterine Manipulator (Apple Medical Corp, Marlboro, Mass) transcervically into the uterine cavity (FIGURE 1).
The cruciate incision. With a conventional scalpel make a small suprapubic transverse incision through the skin and subcutaneous tissue (FIGURE 2). After clearing subcutaneous fat from the midline, incise the rectus fascia and the peritoneum in a vertical direction.
A vertical skin incision can be selected if the preoperative workup suggests a later extension of the original minilaparotomy incision may be required, or if there is a prior vertical incision.
Retraction. Use a soft sleeve-type self-retaining plastic retractor, such as Mobius (Apple Medical Corp) (FIGURE 3 ). When properly placed, this retractor creates an atraumatic, circular area of self-retraction, enabling superior exposure of the pelvis (FIGURE 4).
During surgery it may be necessary to adjust the outer ring if the sleeve loosens. Narrow Deaver or Richardson retractors, if required, provide additional retraction. The bowel may be gently packed, if necessary, but typically it is adequately displaced by the large ovarian cyst.
The atraumatic retraction provided by the soft, self-retaining abdominal retractor minimizes the possibility of tissue trauma, nerve damage, bruising, and postoperative pain. At the same time, the continuous 360° retraction force on the incision maximizes surgical exposure, providing a significantly larger working area than conventional retractors. For example, when applied to a 6-cm incision, the self-retaining retractor creates a 28-cm2 exposed working area, compared with only 18 cm2 provided by a conventional 4-point metal retractor.
The adjustable height of the self-retaining retractor adapts to wounds of varying depth and works on virtually any tissue thickness—a feature that makes the device effective for obese patients. Further, by lining the abdominal incision, the retractor’s plastic sleeve protects the wound’s edges from contamination and potential implantation of malignant cells, making the device ideal for managing ovarian cysts.
FIGURE 1 Hinged uterine manipulator
The manipulator facilitates exposure of the cyst and contralateral adnexa, as well as uterine elevation/rotation.
FIGURE 2 Cruciate incision
Make a 2.5- to 5-cm suprapubic transverse skin incision. Using the Bovie device, incise the subcutaneous fat transversely along the full length of the skin incision down to the level of the anterior rectus fascia. Clear the subcutaneous fat from the midline superiorly and inferiorly to expose 5 to 6 cm of the rectus fascia in the vertical axis. Use blunt digital dissection to assist in mobilizing the subcutaneous fat. Incise the anterior rectus fascia vertically through the full length of the cleared area. Retract the rectus muscles from the midline to expose the underlying transversalis fascia and the peritoneum. Control small bleeding points with the Bovie device. Enter the peritoneum either digitally or with scissors above the bladder dome and extend the peritoneal incision to the full length of the fascial incision.
FIGURE 3 Self-retaining retractor
The retractor consists of a flexible plastic inner ring and a firmer outer ring connected by a soft plastic sleeve.
FIGURE 4360° retraction force
Squeeze the inner ring of the soft, sleeve-type, self-retaining retractor into the peritoneal cavity through the cruciate abdominal incision and allow it to spring open against the parietal peritoneum. Make a digital assessment to ensure that viscera is not trapped between the inner ring and the abdominal wall. Place the entire sleeve on traction by lifting the outer ring. Then roll the outer ring onto the sleeve, collecting excess length, until it sits firmly against the skin. The atraumatic retraction provided by the sleeve-type retractor maximizes exposure of the pelvis. Note how a portion of the large cyst is clearly seen through the atraumatic, circular area of retraction.
Cyst assessment
Visually and digitally inspect the cyst and carefully evaluate the uterus, pelvis, and contralateral adnexa. Determine the extent of adhesions and any unexpected pelvic pathology. When needed, use traditional small retractors or gentle packing to gain additional exposure. If the cystic mass appears suspicious (internal and external excrescences on the cyst, ovaries, or peritoneal surfaces, or ascites), obtain pelvic washings with a suction-irrigation cannula, send the fluid for cytologic examination, and convert the minilaparotomy to a standard exploratory laparotomy. Extensive adhesions to the bowel, broad ligament, or pelvic sidewall and unexpected extensive endometriosis may also require a conversion to standard laparotomy.
Reduce cyst size by decompression
Simple aspiration is inadvisable. To remove a large ovarian cyst using the Pelosi minilaparotomy, reduce the size of the cyst to permit safe and effective mobilization and resection through the small abdominal incision. Simple aspiration of the cyst, with its potential for spilling the contents, is not a wise strategy for several reasons. First, many ovarian cysts contain functional epithelium with a high recurrence rate (8% to 67%). Second, studies show that 10% to 66% of ovarian cyst fluid aspirates initially diagnosed as benign actually are malignant. Further, relying on negative cytology from the aspirate of an ovarian cyst without tissue biopsy may delay appropriate surgery, and the puncture of an unexpected malignant cyst may seed the peritoneal cavity and possibly worsen the patient’s prognosis.11,12
This technique, suitable for all ovarian cysts, makes it possible to aspirate a large cyst without leakage.
Whether spillage of cancer cells actually worsens the prognosis of a patient with a neoplastic cyst remains controversial because of conflicting study results. Nonetheless, the possibility of intraperitoneal dissemination of neoplastic cells from a ruptured cyst cannot be considered innocuous, and the potential negative effect on a patient’s prognosis should not be ignored.14 Make every attempt, therefore, to avoid rupturing the cyst and spilling the fluid into the peritoneal cavity.
A shared flaw plagues aspiration devices. Different devices are available for intraoperative cyst aspiration during laparoscopic, transvaginal, or laparotomy approaches.3,4,6 In addition to long needles, drainage trocars, suction cannulas, and suprapubic bladder catheters, special aspiration instruments have been developed.15 They include a metal vacuum system with an aspirator trocar that seals the surface of the cyst, and a catheter system that pinches the punctured cyst wall between double balloons to prevent spillage.16 In addition, several commercial bags are available to prevent intraperitoneal spillage during removal of ovarian cysts.
All these devices have a universal flaw, however: After a thin-walled cyst initially is punctured, none of these products can prevent the spontaneous dehiscence of the cyst and the resulting spillage of its contents into the abdominal cavity. Vacuum systems work well for large cysts with smooth, round surfaces, but in those with irregular surfaces, both application and maintaining the seal are difficult.17 Fortunately, our technique makes it possible to aspirate a large ovarian cyst without leakage, and the method is suitable for all ovarian cysts regardless of surface type or wall thickness.
Glue the dressing to the cyst to capture leakage. Using a gauze pad, carefully dry the area of the ovarian cyst that is visible through the self-retaining retractor. Then generously spread sterile surgical glue such as Dermabond (Ethicon, Somerville, NJ) on the cyst wall surface (FIGURE 5A). Dermabond is the commercial name for 2-octyl cyanoacrylate, a sterile skin adhesive used as an alternative to stitches to close the edges of small wounds. It is similar to commercial adhesives such as Super Glue and Krazy Glue.
Remove the paper cover of a transparent plastic surgical dressing and place the adhesive side directly onto the glued cyst surface until you are sure the adhesive is completely fixed (FIGURES 5B AND 5C). The 35 cm x 35 cm Steri-Drape or the transparent Tegaderm (both from 3M Health Care, St. Paul, Minn) dressing is effective. A standard nonadhesive plastic dressing or a sterile plastic bag also can be used, as long as the free edges extend beyond the outer rim of the self-retaining retractor.
With a needle aspirator, pierce the cyst through the glued plastic dressing and carefully aspirate the fluid (FIGURES 6A AND 6B). Any leakage is trapped inside the plastic dressing rather than the abdominal cavity. Continue the aspiration until the collapsed cyst and ovary can be gradually delivered through the abdominal incision (FIGURE 6C). Note that the selfretaining retractor also protects the abdominal incision from potential contamination and implantation of neoplastic cells.
Once the cyst and ovary are extracted, you can readily perform an extracorporeal cystectomy, after which the repaired ovary is returned to the abdominal cavity. Be careful to avoid letting any fluid flow back into the peritoneal cavity during cyst removal. When indicated, an extracorporeal adnexectomy can readily be performed.
The presence of oily material or hairs in the aspirated fluid and on the needle tip readily identifies a dermoid cyst.
Dermoid cysts require extra care. Preventing intraperitoneal spillage is especially important when removing a large dermoid cyst, to avoid the possibility of chemical peritonitis, dense adhesions, and fistulas. The presence of oily material or hairs in the aspirated fluid and on the needle tip readily identifies a dermoid cyst. Quite frequently, a large-diameter suction cannula is required to remove the waxy contents. If the dermoid cyst is very large, aspiration alone will not empty it entirely. Complete emptying is not necessary, however. The primary goal of aspiration is to reduce the size and tension of the cyst to permit delivery through the abdominal incision.
Closing the cruciate incision is quicker and requires less exposure than a scaled-down Pfannenstiel’s incision.
Before concluding the procedure, irrigate the peritoneal cavity to remove any remnants of cyst contents that may have spilled—especially important with a dermoid cyst. Perform any additional indicated procedures through the minilaparotomy incision, such as a contralateral ovarian cystectomy, salpingo-oophorectomy, or hysterectomy. After the surgery is completed, remove the self-retaining retractor by hooking a finger through the bottom ring and pulling it gently out of the incision. Closing the cruciate incision is quicker and requires less exposure to complete than a scaled-down Pfannenstiel’s incision. Apply a vertical pressure dressing over the incision to prevent postoperative wound hematoma or seroma formation. Remove the dressing 24 hours later.
FIGURE 5A ‘Leak-proofing’ the aspiration
After drying the surface of the cyst, apply sterile surgical glue to the cyst wall (2 to 3 ampules are usually required to cover the cyst surface).
FIGURE 5B ‘Leak-proofing’ the aspiration
Apply the adhesive side of a clear plastic wound dressing directly onto the cyst’s surface, making sure that the dressing is large enough to fully cover the self-retaining retractor. Using either a piece of folded gauze or your fingers, press the plastic dressing against the adhesive-coated cyst for about 3 to 5 minutes until the adhesive is completely fixed.
FIGURE 5C ‘Leak-proofing’ the aspiration
Remove the paper cover of the wound dressing.
FIGURE 6A Aspirating the cyst
Pierce the dressing and carefully aspirate the fluid until the cyst is partially collapsed.
FIGURE 6B Aspirating the cyst
Place atraumatic clamps on the cyst wall to further control drainage. Any leakage is trapped inside the plastic dressing rather than draining into the abdominal cavity.
FIGURE 6C Aspirating the cyst
Continue the aspiration until the collapsed cyst and ovary can be delivered gradually through the abdominal incision, after detaching the adhesive plastic dressing from the edges of the self-retaining retractor. The portion of the plastic sleeve that is glued to the cyst surface remains attached to the cyst until the cyst is extracted. Following extraction, perform an extracorporeal cystectomy and return the repaired ovary to the abdominal cavity.
Good results
Using our approach, we have treated 38 patients with ovarian cysts of diameters greater than 20 cm thought to be benign by preoperative workup (FIGURE 7). We encountered no malignancies. All surgeries were successfully completed without laparoscopy or conversion to traditional laparotomy and with good cosmesis (FIGURE 8).
Other procedures performed in some of these patients using the same technique included contralateral cystectomy, salpingo-oophorectomy, subtotal and total hysterectomy, adhesiolysis, and appendectomy. We encountered no intraoperative or postoperative complications. Operating times ranged from 18 to 65 minutes. All patients were discharged home within 36 hours and returned to work in a mean of 12 days. Pathology findings of the ovarian cysts included endometrioma, dermoid cyst, serous cystadenoma, and mucinous cystadenoma.
FIGURE 7 Effective for large cysts
Following removal, the collapsed cyst was refilled with 1,300 mL of normal saline to demonstrate its actual size.
FIGURE 8 Good cosmetic results
Cosmetic appearance of the small cruciate abdominal incision 10 days after surgery.Dr. Pelosi II reports that he is a consultant for Apple Medical Corporation. Dr. Pelosi III reports no affiliations or financial arrangements with any companies whose products are mentioned in this article.
1. Pelosi MA, II, Pelosi MA, III. Pelosi minilaparotomy hysterectomy: Effective alternative to laparoscopy and laparotomy. OBG Management. 2003;15(4):16-33.
2. Havrilesky LJ, Peterson BL, Dryden DK, et al. Predictors of clinical outcomes in the laparoscopic management of adnexal masses. Obstet Gynecol. 2003;102:243-251.
3. Pelosi MA, II, Pelosi MA, III. Laparoscopic removal of a 103 pound ovarian tumor. J Am Assoc Gynecol Laparosc. 1996;3:413-417.
4. Eltabbaku GH. Laparoscopic management of ovarian cysts. Contemporary OB/GYN. 2003;48(8):37-50.
5. Ou C, Liu Y, Zabriskie V, et al. Alternate method for laparoscopic management of adnexal masses greater than 10 cm in diameter. J Laparoendosc Adv Surg Tech. 2001;11:125-132.
6. Jeong E, Kim H, Ahn C, et al. Successful laparoscopic removal of huge ovarian cysts. J Am Gynecol Laparosc. 1997;4:609-614.
7. Nagele F, Magos AL. Combined ultrasonographically guided drainage and laparoscopic excision of a large ovarian cyst. Am J Obstet Gynecol. 1996;175:1377-1378.
8. Sheth S. Adnexal pathology. In: Sheth S, Studd J, eds. Vaginal Hysterectomy. London, England: Martin Dunitz; 2002;165-178.
9. Flynn MK, Niloff JM. Outpatient minilaparotomy for ovarian cysts. J Reprod Med. 1999;44:399-404.
10. Benedetti P, Panicci P, Maneschi F, et al. Surgery by minilaparotomy in benign gynecological disease. Obstet Gynecol. 1996;87:456-459.
11. Jansen FW, Tanahotoe S, Veselie M, et al. Laparoscopic aspiration of ovarian cysts: an unreliable technique in primary diagnosis of sonographically benign ovarian lesions. Gynecol Endosc. 1997;6:363-367.
12. Parker WH. Laparoscopic management of the adnexal mass in postmenopausal women. J Gynecol Tech. 1995;1:3-5.
13. Guerriero S, Ajossa S, Mais V, et al. Prelaparoscopic assessment of ovarian cysts in reproductive-age women. Gynecol Endosc. 1997;6:157-167.
14. Fowler JM, Carter JR. Laparoscopic management of the adnexal mass in postmenopausal women. J Gynecol Tech. 1995;1:7-10.
15. McCormick JB, Fitzgibbons JP. Instrument for aspiration of large ovarian cysts. Obstet Gynecol. 1967;29:869-870.
16. Yamada T, Okamoto Y, Kasematsu H. Use of the Sand balloon catheter for the laparoscopic surgery of benign ovarian cysts. Gynecol Endosc. 1999;9:51-54.
17. Shozu M, Segawa T, Sumitani H, et al. Leak-proof puncture of ovarian cysts: instant mounting of plastic bag using cyanoacrylate adhesive. Obstet Gynecol. 2001;97:1007-1010.
- Make a cruciate incision by incising the skin transversely and the anterior rectus fascia vertically.
- Insert a soft, sleeved, self-retaining retractor.
- Using a surgical adhesive, glue a large plastic wound dressing to the surface of the cyst to prevent leakage of contents into the abdominal cavity.
- Aspirate the cyst until it collapses and can be delivered, with the ovary, through the abdominal incision.
- After performing an extracorporeal cystectomy and/or adnexectomy, return the repaired ovary to the abdominal cavity.
Although laparotomy is still considered the standard for ovarian cyst removal, over the past 15 years minimally invasive surgery has gained wider acceptance in cases where preoperative assessment suggests an adnexal mass is benign.
Unfortunately, minimally invasive management of a large ovarian cyst (greater than 10 cm) is particularly challenging for several reasons:
- The cyst can rupture and spill its contents into the peritoneum,
- the cyst’s size limits the surgical field, and
- an unexpected malignancy may be revealed.
An innovative minilaparotomy technique for the removal of benign ovarian cysts offers the advantages of laparoscopic and laparoscopic-assisted procedures while bypassing the major disadvantages: the necessity for specialized and expensive equipment, lengthy operative time, and long learning curves.1 (The minimally invasive procedures currently available for the treatment of ovarian cysts include laparoscopic cystectomy, laparoscopic-assisted minilaparotomy cystectomy, laparoscopic-assisted vaginal cystectomy, combined percutaneous ultrasound cyst aspiration and laparoscopic cystectomy, transvaginal cystectomy, and the traditional minilaparotomy cystectomy.2-10)
The procedure is faster, less expensive, carries fewer potential risks than traditional alternatives, and offers these advantages:
- can be performed under regional anesthesia
- relies on standard open techniques
- uses inexpensive instrumentation
- is easy to learn
- can be used for very large cysts
- eliminates the risk of intraperitoneal spillage of cyst contents
- offers similar postoperative convalescence and mean time to return to work as laparoscopic or laparoscopic-assisted management of large ovarian cysts
General Ob/Gyns—not gynecologic oncologists—perform most surgeries on patients with adnexal masses, since ovarian cancer is relatively uncommon in the absence of preoperative risk factors for malignancy. Our approach offers an appealing option to Ob/Gyns reluctant to abandon routine traditional laparotomy for such ovarian cysts.
Selecting the right patient
Adequate preoperative assessment diminishes the risk of unexpected malignancy in a patient undergoing surgery for an ovarian mass to less than 1%.4 At this time, the combination of menopausal status, cancer antigen (CA) 125 level, physical examination, and ultrasound is the best strategy for evaluating the patient with an ovarian cyst.11
Signs of malignancy. Ultrasound features that suggest malignancy include irregular borders, thick septa, solid areas, internal and external excrescences, matted bowel, and ascites. Benign cysts, on the other hand, are usually unilateral and have regular borders, thin septa, no solid areas, and no internal excrescences.4 The measurement of blood flow within the mass by color Doppler may improve the accuracy of ultrasound in differentiating benign from malignant cysts.4,12
On physical examination, an adnexal mass that is fixed, irregular, or solid also suggests a neoplasm. An elevated CA 125 combined with a complex adnexal mass is likely to be associated with malignancy. The test is even more specific in postmenopausal women with adnexal masses.4,12
However, plasma levels of CA 125 also can be elevated in several benign gynecologic conditions such as endometriosis, simple ovarian cysts, pelvic inflammatory disease, ovarian torsion, fibroids, and in physiologic conditions such as menstruation and pregnancy.13
Anticipate the need to convert to laparotomy. Every patient’s surgical consent should include a possible conversion to laparotomy. To avoid incomplete surgical treatment and significant delays in proper therapy, a gynecologic surgeon experienced in the management of ovarian cancer should be readily available, in the event an unexpected malignancy is encountered. Ideally, the staging surgery and definite treatment should be performed at the time of initial minilaparotomy. Comprehensive surgical staging and treatment include thorough exploration of the pelvis and abdomen, omentectomy, pelvic and paraaortic lymph node sampling, multiple peritoneal biopsies and washings, bilateral salpingo-oophorectomy, hysterectomy, and debulking, when indicated.
Prepare for surgery with position, incision, and retraction
Before beginning, it is crucial to correctly position the patient, make the appropriate incision, and insert the right retractor.1
Position. After administering regional or general anesthesia, place the patient in a modified lithotomy, as for laparoscopic surgery. Tuck the arms alongside the torso and place the legs in boot stirrups. Avoid hip flexion and allow adequate thigh abduction to expose the vagina. Perform a careful pelvic examination to determine the size and mobility of the adnexal mass.
When properly placed, this retractor creates an atraumatic, circular area of self-retraction, enabling superior exposure
Place an indwelling transurethral catheter, and pass a sturdy, hinged uterine manipulator such as the Pelosi Uterine Manipulator (Apple Medical Corp, Marlboro, Mass) transcervically into the uterine cavity (FIGURE 1).
The cruciate incision. With a conventional scalpel make a small suprapubic transverse incision through the skin and subcutaneous tissue (FIGURE 2). After clearing subcutaneous fat from the midline, incise the rectus fascia and the peritoneum in a vertical direction.
A vertical skin incision can be selected if the preoperative workup suggests a later extension of the original minilaparotomy incision may be required, or if there is a prior vertical incision.
Retraction. Use a soft sleeve-type self-retaining plastic retractor, such as Mobius (Apple Medical Corp) (FIGURE 3 ). When properly placed, this retractor creates an atraumatic, circular area of self-retraction, enabling superior exposure of the pelvis (FIGURE 4).
During surgery it may be necessary to adjust the outer ring if the sleeve loosens. Narrow Deaver or Richardson retractors, if required, provide additional retraction. The bowel may be gently packed, if necessary, but typically it is adequately displaced by the large ovarian cyst.
The atraumatic retraction provided by the soft, self-retaining abdominal retractor minimizes the possibility of tissue trauma, nerve damage, bruising, and postoperative pain. At the same time, the continuous 360° retraction force on the incision maximizes surgical exposure, providing a significantly larger working area than conventional retractors. For example, when applied to a 6-cm incision, the self-retaining retractor creates a 28-cm2 exposed working area, compared with only 18 cm2 provided by a conventional 4-point metal retractor.
The adjustable height of the self-retaining retractor adapts to wounds of varying depth and works on virtually any tissue thickness—a feature that makes the device effective for obese patients. Further, by lining the abdominal incision, the retractor’s plastic sleeve protects the wound’s edges from contamination and potential implantation of malignant cells, making the device ideal for managing ovarian cysts.
FIGURE 1 Hinged uterine manipulator
The manipulator facilitates exposure of the cyst and contralateral adnexa, as well as uterine elevation/rotation.
FIGURE 2 Cruciate incision
Make a 2.5- to 5-cm suprapubic transverse skin incision. Using the Bovie device, incise the subcutaneous fat transversely along the full length of the skin incision down to the level of the anterior rectus fascia. Clear the subcutaneous fat from the midline superiorly and inferiorly to expose 5 to 6 cm of the rectus fascia in the vertical axis. Use blunt digital dissection to assist in mobilizing the subcutaneous fat. Incise the anterior rectus fascia vertically through the full length of the cleared area. Retract the rectus muscles from the midline to expose the underlying transversalis fascia and the peritoneum. Control small bleeding points with the Bovie device. Enter the peritoneum either digitally or with scissors above the bladder dome and extend the peritoneal incision to the full length of the fascial incision.
FIGURE 3 Self-retaining retractor
The retractor consists of a flexible plastic inner ring and a firmer outer ring connected by a soft plastic sleeve.
FIGURE 4360° retraction force
Squeeze the inner ring of the soft, sleeve-type, self-retaining retractor into the peritoneal cavity through the cruciate abdominal incision and allow it to spring open against the parietal peritoneum. Make a digital assessment to ensure that viscera is not trapped between the inner ring and the abdominal wall. Place the entire sleeve on traction by lifting the outer ring. Then roll the outer ring onto the sleeve, collecting excess length, until it sits firmly against the skin. The atraumatic retraction provided by the sleeve-type retractor maximizes exposure of the pelvis. Note how a portion of the large cyst is clearly seen through the atraumatic, circular area of retraction.
Cyst assessment
Visually and digitally inspect the cyst and carefully evaluate the uterus, pelvis, and contralateral adnexa. Determine the extent of adhesions and any unexpected pelvic pathology. When needed, use traditional small retractors or gentle packing to gain additional exposure. If the cystic mass appears suspicious (internal and external excrescences on the cyst, ovaries, or peritoneal surfaces, or ascites), obtain pelvic washings with a suction-irrigation cannula, send the fluid for cytologic examination, and convert the minilaparotomy to a standard exploratory laparotomy. Extensive adhesions to the bowel, broad ligament, or pelvic sidewall and unexpected extensive endometriosis may also require a conversion to standard laparotomy.
Reduce cyst size by decompression
Simple aspiration is inadvisable. To remove a large ovarian cyst using the Pelosi minilaparotomy, reduce the size of the cyst to permit safe and effective mobilization and resection through the small abdominal incision. Simple aspiration of the cyst, with its potential for spilling the contents, is not a wise strategy for several reasons. First, many ovarian cysts contain functional epithelium with a high recurrence rate (8% to 67%). Second, studies show that 10% to 66% of ovarian cyst fluid aspirates initially diagnosed as benign actually are malignant. Further, relying on negative cytology from the aspirate of an ovarian cyst without tissue biopsy may delay appropriate surgery, and the puncture of an unexpected malignant cyst may seed the peritoneal cavity and possibly worsen the patient’s prognosis.11,12
This technique, suitable for all ovarian cysts, makes it possible to aspirate a large cyst without leakage.
Whether spillage of cancer cells actually worsens the prognosis of a patient with a neoplastic cyst remains controversial because of conflicting study results. Nonetheless, the possibility of intraperitoneal dissemination of neoplastic cells from a ruptured cyst cannot be considered innocuous, and the potential negative effect on a patient’s prognosis should not be ignored.14 Make every attempt, therefore, to avoid rupturing the cyst and spilling the fluid into the peritoneal cavity.
A shared flaw plagues aspiration devices. Different devices are available for intraoperative cyst aspiration during laparoscopic, transvaginal, or laparotomy approaches.3,4,6 In addition to long needles, drainage trocars, suction cannulas, and suprapubic bladder catheters, special aspiration instruments have been developed.15 They include a metal vacuum system with an aspirator trocar that seals the surface of the cyst, and a catheter system that pinches the punctured cyst wall between double balloons to prevent spillage.16 In addition, several commercial bags are available to prevent intraperitoneal spillage during removal of ovarian cysts.
All these devices have a universal flaw, however: After a thin-walled cyst initially is punctured, none of these products can prevent the spontaneous dehiscence of the cyst and the resulting spillage of its contents into the abdominal cavity. Vacuum systems work well for large cysts with smooth, round surfaces, but in those with irregular surfaces, both application and maintaining the seal are difficult.17 Fortunately, our technique makes it possible to aspirate a large ovarian cyst without leakage, and the method is suitable for all ovarian cysts regardless of surface type or wall thickness.
Glue the dressing to the cyst to capture leakage. Using a gauze pad, carefully dry the area of the ovarian cyst that is visible through the self-retaining retractor. Then generously spread sterile surgical glue such as Dermabond (Ethicon, Somerville, NJ) on the cyst wall surface (FIGURE 5A). Dermabond is the commercial name for 2-octyl cyanoacrylate, a sterile skin adhesive used as an alternative to stitches to close the edges of small wounds. It is similar to commercial adhesives such as Super Glue and Krazy Glue.
Remove the paper cover of a transparent plastic surgical dressing and place the adhesive side directly onto the glued cyst surface until you are sure the adhesive is completely fixed (FIGURES 5B AND 5C). The 35 cm x 35 cm Steri-Drape or the transparent Tegaderm (both from 3M Health Care, St. Paul, Minn) dressing is effective. A standard nonadhesive plastic dressing or a sterile plastic bag also can be used, as long as the free edges extend beyond the outer rim of the self-retaining retractor.
With a needle aspirator, pierce the cyst through the glued plastic dressing and carefully aspirate the fluid (FIGURES 6A AND 6B). Any leakage is trapped inside the plastic dressing rather than the abdominal cavity. Continue the aspiration until the collapsed cyst and ovary can be gradually delivered through the abdominal incision (FIGURE 6C). Note that the selfretaining retractor also protects the abdominal incision from potential contamination and implantation of neoplastic cells.
Once the cyst and ovary are extracted, you can readily perform an extracorporeal cystectomy, after which the repaired ovary is returned to the abdominal cavity. Be careful to avoid letting any fluid flow back into the peritoneal cavity during cyst removal. When indicated, an extracorporeal adnexectomy can readily be performed.
The presence of oily material or hairs in the aspirated fluid and on the needle tip readily identifies a dermoid cyst.
Dermoid cysts require extra care. Preventing intraperitoneal spillage is especially important when removing a large dermoid cyst, to avoid the possibility of chemical peritonitis, dense adhesions, and fistulas. The presence of oily material or hairs in the aspirated fluid and on the needle tip readily identifies a dermoid cyst. Quite frequently, a large-diameter suction cannula is required to remove the waxy contents. If the dermoid cyst is very large, aspiration alone will not empty it entirely. Complete emptying is not necessary, however. The primary goal of aspiration is to reduce the size and tension of the cyst to permit delivery through the abdominal incision.
Closing the cruciate incision is quicker and requires less exposure than a scaled-down Pfannenstiel’s incision.
Before concluding the procedure, irrigate the peritoneal cavity to remove any remnants of cyst contents that may have spilled—especially important with a dermoid cyst. Perform any additional indicated procedures through the minilaparotomy incision, such as a contralateral ovarian cystectomy, salpingo-oophorectomy, or hysterectomy. After the surgery is completed, remove the self-retaining retractor by hooking a finger through the bottom ring and pulling it gently out of the incision. Closing the cruciate incision is quicker and requires less exposure to complete than a scaled-down Pfannenstiel’s incision. Apply a vertical pressure dressing over the incision to prevent postoperative wound hematoma or seroma formation. Remove the dressing 24 hours later.
FIGURE 5A ‘Leak-proofing’ the aspiration
After drying the surface of the cyst, apply sterile surgical glue to the cyst wall (2 to 3 ampules are usually required to cover the cyst surface).
FIGURE 5B ‘Leak-proofing’ the aspiration
Apply the adhesive side of a clear plastic wound dressing directly onto the cyst’s surface, making sure that the dressing is large enough to fully cover the self-retaining retractor. Using either a piece of folded gauze or your fingers, press the plastic dressing against the adhesive-coated cyst for about 3 to 5 minutes until the adhesive is completely fixed.
FIGURE 5C ‘Leak-proofing’ the aspiration
Remove the paper cover of the wound dressing.
FIGURE 6A Aspirating the cyst
Pierce the dressing and carefully aspirate the fluid until the cyst is partially collapsed.
FIGURE 6B Aspirating the cyst
Place atraumatic clamps on the cyst wall to further control drainage. Any leakage is trapped inside the plastic dressing rather than draining into the abdominal cavity.
FIGURE 6C Aspirating the cyst
Continue the aspiration until the collapsed cyst and ovary can be delivered gradually through the abdominal incision, after detaching the adhesive plastic dressing from the edges of the self-retaining retractor. The portion of the plastic sleeve that is glued to the cyst surface remains attached to the cyst until the cyst is extracted. Following extraction, perform an extracorporeal cystectomy and return the repaired ovary to the abdominal cavity.
Good results
Using our approach, we have treated 38 patients with ovarian cysts of diameters greater than 20 cm thought to be benign by preoperative workup (FIGURE 7). We encountered no malignancies. All surgeries were successfully completed without laparoscopy or conversion to traditional laparotomy and with good cosmesis (FIGURE 8).
Other procedures performed in some of these patients using the same technique included contralateral cystectomy, salpingo-oophorectomy, subtotal and total hysterectomy, adhesiolysis, and appendectomy. We encountered no intraoperative or postoperative complications. Operating times ranged from 18 to 65 minutes. All patients were discharged home within 36 hours and returned to work in a mean of 12 days. Pathology findings of the ovarian cysts included endometrioma, dermoid cyst, serous cystadenoma, and mucinous cystadenoma.
FIGURE 7 Effective for large cysts
Following removal, the collapsed cyst was refilled with 1,300 mL of normal saline to demonstrate its actual size.
FIGURE 8 Good cosmetic results
Cosmetic appearance of the small cruciate abdominal incision 10 days after surgery.Dr. Pelosi II reports that he is a consultant for Apple Medical Corporation. Dr. Pelosi III reports no affiliations or financial arrangements with any companies whose products are mentioned in this article.
- Make a cruciate incision by incising the skin transversely and the anterior rectus fascia vertically.
- Insert a soft, sleeved, self-retaining retractor.
- Using a surgical adhesive, glue a large plastic wound dressing to the surface of the cyst to prevent leakage of contents into the abdominal cavity.
- Aspirate the cyst until it collapses and can be delivered, with the ovary, through the abdominal incision.
- After performing an extracorporeal cystectomy and/or adnexectomy, return the repaired ovary to the abdominal cavity.
Although laparotomy is still considered the standard for ovarian cyst removal, over the past 15 years minimally invasive surgery has gained wider acceptance in cases where preoperative assessment suggests an adnexal mass is benign.
Unfortunately, minimally invasive management of a large ovarian cyst (greater than 10 cm) is particularly challenging for several reasons:
- The cyst can rupture and spill its contents into the peritoneum,
- the cyst’s size limits the surgical field, and
- an unexpected malignancy may be revealed.
An innovative minilaparotomy technique for the removal of benign ovarian cysts offers the advantages of laparoscopic and laparoscopic-assisted procedures while bypassing the major disadvantages: the necessity for specialized and expensive equipment, lengthy operative time, and long learning curves.1 (The minimally invasive procedures currently available for the treatment of ovarian cysts include laparoscopic cystectomy, laparoscopic-assisted minilaparotomy cystectomy, laparoscopic-assisted vaginal cystectomy, combined percutaneous ultrasound cyst aspiration and laparoscopic cystectomy, transvaginal cystectomy, and the traditional minilaparotomy cystectomy.2-10)
The procedure is faster, less expensive, carries fewer potential risks than traditional alternatives, and offers these advantages:
- can be performed under regional anesthesia
- relies on standard open techniques
- uses inexpensive instrumentation
- is easy to learn
- can be used for very large cysts
- eliminates the risk of intraperitoneal spillage of cyst contents
- offers similar postoperative convalescence and mean time to return to work as laparoscopic or laparoscopic-assisted management of large ovarian cysts
General Ob/Gyns—not gynecologic oncologists—perform most surgeries on patients with adnexal masses, since ovarian cancer is relatively uncommon in the absence of preoperative risk factors for malignancy. Our approach offers an appealing option to Ob/Gyns reluctant to abandon routine traditional laparotomy for such ovarian cysts.
Selecting the right patient
Adequate preoperative assessment diminishes the risk of unexpected malignancy in a patient undergoing surgery for an ovarian mass to less than 1%.4 At this time, the combination of menopausal status, cancer antigen (CA) 125 level, physical examination, and ultrasound is the best strategy for evaluating the patient with an ovarian cyst.11
Signs of malignancy. Ultrasound features that suggest malignancy include irregular borders, thick septa, solid areas, internal and external excrescences, matted bowel, and ascites. Benign cysts, on the other hand, are usually unilateral and have regular borders, thin septa, no solid areas, and no internal excrescences.4 The measurement of blood flow within the mass by color Doppler may improve the accuracy of ultrasound in differentiating benign from malignant cysts.4,12
On physical examination, an adnexal mass that is fixed, irregular, or solid also suggests a neoplasm. An elevated CA 125 combined with a complex adnexal mass is likely to be associated with malignancy. The test is even more specific in postmenopausal women with adnexal masses.4,12
However, plasma levels of CA 125 also can be elevated in several benign gynecologic conditions such as endometriosis, simple ovarian cysts, pelvic inflammatory disease, ovarian torsion, fibroids, and in physiologic conditions such as menstruation and pregnancy.13
Anticipate the need to convert to laparotomy. Every patient’s surgical consent should include a possible conversion to laparotomy. To avoid incomplete surgical treatment and significant delays in proper therapy, a gynecologic surgeon experienced in the management of ovarian cancer should be readily available, in the event an unexpected malignancy is encountered. Ideally, the staging surgery and definite treatment should be performed at the time of initial minilaparotomy. Comprehensive surgical staging and treatment include thorough exploration of the pelvis and abdomen, omentectomy, pelvic and paraaortic lymph node sampling, multiple peritoneal biopsies and washings, bilateral salpingo-oophorectomy, hysterectomy, and debulking, when indicated.
Prepare for surgery with position, incision, and retraction
Before beginning, it is crucial to correctly position the patient, make the appropriate incision, and insert the right retractor.1
Position. After administering regional or general anesthesia, place the patient in a modified lithotomy, as for laparoscopic surgery. Tuck the arms alongside the torso and place the legs in boot stirrups. Avoid hip flexion and allow adequate thigh abduction to expose the vagina. Perform a careful pelvic examination to determine the size and mobility of the adnexal mass.
When properly placed, this retractor creates an atraumatic, circular area of self-retraction, enabling superior exposure
Place an indwelling transurethral catheter, and pass a sturdy, hinged uterine manipulator such as the Pelosi Uterine Manipulator (Apple Medical Corp, Marlboro, Mass) transcervically into the uterine cavity (FIGURE 1).
The cruciate incision. With a conventional scalpel make a small suprapubic transverse incision through the skin and subcutaneous tissue (FIGURE 2). After clearing subcutaneous fat from the midline, incise the rectus fascia and the peritoneum in a vertical direction.
A vertical skin incision can be selected if the preoperative workup suggests a later extension of the original minilaparotomy incision may be required, or if there is a prior vertical incision.
Retraction. Use a soft sleeve-type self-retaining plastic retractor, such as Mobius (Apple Medical Corp) (FIGURE 3 ). When properly placed, this retractor creates an atraumatic, circular area of self-retraction, enabling superior exposure of the pelvis (FIGURE 4).
During surgery it may be necessary to adjust the outer ring if the sleeve loosens. Narrow Deaver or Richardson retractors, if required, provide additional retraction. The bowel may be gently packed, if necessary, but typically it is adequately displaced by the large ovarian cyst.
The atraumatic retraction provided by the soft, self-retaining abdominal retractor minimizes the possibility of tissue trauma, nerve damage, bruising, and postoperative pain. At the same time, the continuous 360° retraction force on the incision maximizes surgical exposure, providing a significantly larger working area than conventional retractors. For example, when applied to a 6-cm incision, the self-retaining retractor creates a 28-cm2 exposed working area, compared with only 18 cm2 provided by a conventional 4-point metal retractor.
The adjustable height of the self-retaining retractor adapts to wounds of varying depth and works on virtually any tissue thickness—a feature that makes the device effective for obese patients. Further, by lining the abdominal incision, the retractor’s plastic sleeve protects the wound’s edges from contamination and potential implantation of malignant cells, making the device ideal for managing ovarian cysts.
FIGURE 1 Hinged uterine manipulator
The manipulator facilitates exposure of the cyst and contralateral adnexa, as well as uterine elevation/rotation.
FIGURE 2 Cruciate incision
Make a 2.5- to 5-cm suprapubic transverse skin incision. Using the Bovie device, incise the subcutaneous fat transversely along the full length of the skin incision down to the level of the anterior rectus fascia. Clear the subcutaneous fat from the midline superiorly and inferiorly to expose 5 to 6 cm of the rectus fascia in the vertical axis. Use blunt digital dissection to assist in mobilizing the subcutaneous fat. Incise the anterior rectus fascia vertically through the full length of the cleared area. Retract the rectus muscles from the midline to expose the underlying transversalis fascia and the peritoneum. Control small bleeding points with the Bovie device. Enter the peritoneum either digitally or with scissors above the bladder dome and extend the peritoneal incision to the full length of the fascial incision.
FIGURE 3 Self-retaining retractor
The retractor consists of a flexible plastic inner ring and a firmer outer ring connected by a soft plastic sleeve.
FIGURE 4360° retraction force
Squeeze the inner ring of the soft, sleeve-type, self-retaining retractor into the peritoneal cavity through the cruciate abdominal incision and allow it to spring open against the parietal peritoneum. Make a digital assessment to ensure that viscera is not trapped between the inner ring and the abdominal wall. Place the entire sleeve on traction by lifting the outer ring. Then roll the outer ring onto the sleeve, collecting excess length, until it sits firmly against the skin. The atraumatic retraction provided by the sleeve-type retractor maximizes exposure of the pelvis. Note how a portion of the large cyst is clearly seen through the atraumatic, circular area of retraction.
Cyst assessment
Visually and digitally inspect the cyst and carefully evaluate the uterus, pelvis, and contralateral adnexa. Determine the extent of adhesions and any unexpected pelvic pathology. When needed, use traditional small retractors or gentle packing to gain additional exposure. If the cystic mass appears suspicious (internal and external excrescences on the cyst, ovaries, or peritoneal surfaces, or ascites), obtain pelvic washings with a suction-irrigation cannula, send the fluid for cytologic examination, and convert the minilaparotomy to a standard exploratory laparotomy. Extensive adhesions to the bowel, broad ligament, or pelvic sidewall and unexpected extensive endometriosis may also require a conversion to standard laparotomy.
Reduce cyst size by decompression
Simple aspiration is inadvisable. To remove a large ovarian cyst using the Pelosi minilaparotomy, reduce the size of the cyst to permit safe and effective mobilization and resection through the small abdominal incision. Simple aspiration of the cyst, with its potential for spilling the contents, is not a wise strategy for several reasons. First, many ovarian cysts contain functional epithelium with a high recurrence rate (8% to 67%). Second, studies show that 10% to 66% of ovarian cyst fluid aspirates initially diagnosed as benign actually are malignant. Further, relying on negative cytology from the aspirate of an ovarian cyst without tissue biopsy may delay appropriate surgery, and the puncture of an unexpected malignant cyst may seed the peritoneal cavity and possibly worsen the patient’s prognosis.11,12
This technique, suitable for all ovarian cysts, makes it possible to aspirate a large cyst without leakage.
Whether spillage of cancer cells actually worsens the prognosis of a patient with a neoplastic cyst remains controversial because of conflicting study results. Nonetheless, the possibility of intraperitoneal dissemination of neoplastic cells from a ruptured cyst cannot be considered innocuous, and the potential negative effect on a patient’s prognosis should not be ignored.14 Make every attempt, therefore, to avoid rupturing the cyst and spilling the fluid into the peritoneal cavity.
A shared flaw plagues aspiration devices. Different devices are available for intraoperative cyst aspiration during laparoscopic, transvaginal, or laparotomy approaches.3,4,6 In addition to long needles, drainage trocars, suction cannulas, and suprapubic bladder catheters, special aspiration instruments have been developed.15 They include a metal vacuum system with an aspirator trocar that seals the surface of the cyst, and a catheter system that pinches the punctured cyst wall between double balloons to prevent spillage.16 In addition, several commercial bags are available to prevent intraperitoneal spillage during removal of ovarian cysts.
All these devices have a universal flaw, however: After a thin-walled cyst initially is punctured, none of these products can prevent the spontaneous dehiscence of the cyst and the resulting spillage of its contents into the abdominal cavity. Vacuum systems work well for large cysts with smooth, round surfaces, but in those with irregular surfaces, both application and maintaining the seal are difficult.17 Fortunately, our technique makes it possible to aspirate a large ovarian cyst without leakage, and the method is suitable for all ovarian cysts regardless of surface type or wall thickness.
Glue the dressing to the cyst to capture leakage. Using a gauze pad, carefully dry the area of the ovarian cyst that is visible through the self-retaining retractor. Then generously spread sterile surgical glue such as Dermabond (Ethicon, Somerville, NJ) on the cyst wall surface (FIGURE 5A). Dermabond is the commercial name for 2-octyl cyanoacrylate, a sterile skin adhesive used as an alternative to stitches to close the edges of small wounds. It is similar to commercial adhesives such as Super Glue and Krazy Glue.
Remove the paper cover of a transparent plastic surgical dressing and place the adhesive side directly onto the glued cyst surface until you are sure the adhesive is completely fixed (FIGURES 5B AND 5C). The 35 cm x 35 cm Steri-Drape or the transparent Tegaderm (both from 3M Health Care, St. Paul, Minn) dressing is effective. A standard nonadhesive plastic dressing or a sterile plastic bag also can be used, as long as the free edges extend beyond the outer rim of the self-retaining retractor.
With a needle aspirator, pierce the cyst through the glued plastic dressing and carefully aspirate the fluid (FIGURES 6A AND 6B). Any leakage is trapped inside the plastic dressing rather than the abdominal cavity. Continue the aspiration until the collapsed cyst and ovary can be gradually delivered through the abdominal incision (FIGURE 6C). Note that the selfretaining retractor also protects the abdominal incision from potential contamination and implantation of neoplastic cells.
Once the cyst and ovary are extracted, you can readily perform an extracorporeal cystectomy, after which the repaired ovary is returned to the abdominal cavity. Be careful to avoid letting any fluid flow back into the peritoneal cavity during cyst removal. When indicated, an extracorporeal adnexectomy can readily be performed.
The presence of oily material or hairs in the aspirated fluid and on the needle tip readily identifies a dermoid cyst.
Dermoid cysts require extra care. Preventing intraperitoneal spillage is especially important when removing a large dermoid cyst, to avoid the possibility of chemical peritonitis, dense adhesions, and fistulas. The presence of oily material or hairs in the aspirated fluid and on the needle tip readily identifies a dermoid cyst. Quite frequently, a large-diameter suction cannula is required to remove the waxy contents. If the dermoid cyst is very large, aspiration alone will not empty it entirely. Complete emptying is not necessary, however. The primary goal of aspiration is to reduce the size and tension of the cyst to permit delivery through the abdominal incision.
Closing the cruciate incision is quicker and requires less exposure than a scaled-down Pfannenstiel’s incision.
Before concluding the procedure, irrigate the peritoneal cavity to remove any remnants of cyst contents that may have spilled—especially important with a dermoid cyst. Perform any additional indicated procedures through the minilaparotomy incision, such as a contralateral ovarian cystectomy, salpingo-oophorectomy, or hysterectomy. After the surgery is completed, remove the self-retaining retractor by hooking a finger through the bottom ring and pulling it gently out of the incision. Closing the cruciate incision is quicker and requires less exposure to complete than a scaled-down Pfannenstiel’s incision. Apply a vertical pressure dressing over the incision to prevent postoperative wound hematoma or seroma formation. Remove the dressing 24 hours later.
FIGURE 5A ‘Leak-proofing’ the aspiration
After drying the surface of the cyst, apply sterile surgical glue to the cyst wall (2 to 3 ampules are usually required to cover the cyst surface).
FIGURE 5B ‘Leak-proofing’ the aspiration
Apply the adhesive side of a clear plastic wound dressing directly onto the cyst’s surface, making sure that the dressing is large enough to fully cover the self-retaining retractor. Using either a piece of folded gauze or your fingers, press the plastic dressing against the adhesive-coated cyst for about 3 to 5 minutes until the adhesive is completely fixed.
FIGURE 5C ‘Leak-proofing’ the aspiration
Remove the paper cover of the wound dressing.
FIGURE 6A Aspirating the cyst
Pierce the dressing and carefully aspirate the fluid until the cyst is partially collapsed.
FIGURE 6B Aspirating the cyst
Place atraumatic clamps on the cyst wall to further control drainage. Any leakage is trapped inside the plastic dressing rather than draining into the abdominal cavity.
FIGURE 6C Aspirating the cyst
Continue the aspiration until the collapsed cyst and ovary can be delivered gradually through the abdominal incision, after detaching the adhesive plastic dressing from the edges of the self-retaining retractor. The portion of the plastic sleeve that is glued to the cyst surface remains attached to the cyst until the cyst is extracted. Following extraction, perform an extracorporeal cystectomy and return the repaired ovary to the abdominal cavity.
Good results
Using our approach, we have treated 38 patients with ovarian cysts of diameters greater than 20 cm thought to be benign by preoperative workup (FIGURE 7). We encountered no malignancies. All surgeries were successfully completed without laparoscopy or conversion to traditional laparotomy and with good cosmesis (FIGURE 8).
Other procedures performed in some of these patients using the same technique included contralateral cystectomy, salpingo-oophorectomy, subtotal and total hysterectomy, adhesiolysis, and appendectomy. We encountered no intraoperative or postoperative complications. Operating times ranged from 18 to 65 minutes. All patients were discharged home within 36 hours and returned to work in a mean of 12 days. Pathology findings of the ovarian cysts included endometrioma, dermoid cyst, serous cystadenoma, and mucinous cystadenoma.
FIGURE 7 Effective for large cysts
Following removal, the collapsed cyst was refilled with 1,300 mL of normal saline to demonstrate its actual size.
FIGURE 8 Good cosmetic results
Cosmetic appearance of the small cruciate abdominal incision 10 days after surgery.Dr. Pelosi II reports that he is a consultant for Apple Medical Corporation. Dr. Pelosi III reports no affiliations or financial arrangements with any companies whose products are mentioned in this article.
1. Pelosi MA, II, Pelosi MA, III. Pelosi minilaparotomy hysterectomy: Effective alternative to laparoscopy and laparotomy. OBG Management. 2003;15(4):16-33.
2. Havrilesky LJ, Peterson BL, Dryden DK, et al. Predictors of clinical outcomes in the laparoscopic management of adnexal masses. Obstet Gynecol. 2003;102:243-251.
3. Pelosi MA, II, Pelosi MA, III. Laparoscopic removal of a 103 pound ovarian tumor. J Am Assoc Gynecol Laparosc. 1996;3:413-417.
4. Eltabbaku GH. Laparoscopic management of ovarian cysts. Contemporary OB/GYN. 2003;48(8):37-50.
5. Ou C, Liu Y, Zabriskie V, et al. Alternate method for laparoscopic management of adnexal masses greater than 10 cm in diameter. J Laparoendosc Adv Surg Tech. 2001;11:125-132.
6. Jeong E, Kim H, Ahn C, et al. Successful laparoscopic removal of huge ovarian cysts. J Am Gynecol Laparosc. 1997;4:609-614.
7. Nagele F, Magos AL. Combined ultrasonographically guided drainage and laparoscopic excision of a large ovarian cyst. Am J Obstet Gynecol. 1996;175:1377-1378.
8. Sheth S. Adnexal pathology. In: Sheth S, Studd J, eds. Vaginal Hysterectomy. London, England: Martin Dunitz; 2002;165-178.
9. Flynn MK, Niloff JM. Outpatient minilaparotomy for ovarian cysts. J Reprod Med. 1999;44:399-404.
10. Benedetti P, Panicci P, Maneschi F, et al. Surgery by minilaparotomy in benign gynecological disease. Obstet Gynecol. 1996;87:456-459.
11. Jansen FW, Tanahotoe S, Veselie M, et al. Laparoscopic aspiration of ovarian cysts: an unreliable technique in primary diagnosis of sonographically benign ovarian lesions. Gynecol Endosc. 1997;6:363-367.
12. Parker WH. Laparoscopic management of the adnexal mass in postmenopausal women. J Gynecol Tech. 1995;1:3-5.
13. Guerriero S, Ajossa S, Mais V, et al. Prelaparoscopic assessment of ovarian cysts in reproductive-age women. Gynecol Endosc. 1997;6:157-167.
14. Fowler JM, Carter JR. Laparoscopic management of the adnexal mass in postmenopausal women. J Gynecol Tech. 1995;1:7-10.
15. McCormick JB, Fitzgibbons JP. Instrument for aspiration of large ovarian cysts. Obstet Gynecol. 1967;29:869-870.
16. Yamada T, Okamoto Y, Kasematsu H. Use of the Sand balloon catheter for the laparoscopic surgery of benign ovarian cysts. Gynecol Endosc. 1999;9:51-54.
17. Shozu M, Segawa T, Sumitani H, et al. Leak-proof puncture of ovarian cysts: instant mounting of plastic bag using cyanoacrylate adhesive. Obstet Gynecol. 2001;97:1007-1010.
1. Pelosi MA, II, Pelosi MA, III. Pelosi minilaparotomy hysterectomy: Effective alternative to laparoscopy and laparotomy. OBG Management. 2003;15(4):16-33.
2. Havrilesky LJ, Peterson BL, Dryden DK, et al. Predictors of clinical outcomes in the laparoscopic management of adnexal masses. Obstet Gynecol. 2003;102:243-251.
3. Pelosi MA, II, Pelosi MA, III. Laparoscopic removal of a 103 pound ovarian tumor. J Am Assoc Gynecol Laparosc. 1996;3:413-417.
4. Eltabbaku GH. Laparoscopic management of ovarian cysts. Contemporary OB/GYN. 2003;48(8):37-50.
5. Ou C, Liu Y, Zabriskie V, et al. Alternate method for laparoscopic management of adnexal masses greater than 10 cm in diameter. J Laparoendosc Adv Surg Tech. 2001;11:125-132.
6. Jeong E, Kim H, Ahn C, et al. Successful laparoscopic removal of huge ovarian cysts. J Am Gynecol Laparosc. 1997;4:609-614.
7. Nagele F, Magos AL. Combined ultrasonographically guided drainage and laparoscopic excision of a large ovarian cyst. Am J Obstet Gynecol. 1996;175:1377-1378.
8. Sheth S. Adnexal pathology. In: Sheth S, Studd J, eds. Vaginal Hysterectomy. London, England: Martin Dunitz; 2002;165-178.
9. Flynn MK, Niloff JM. Outpatient minilaparotomy for ovarian cysts. J Reprod Med. 1999;44:399-404.
10. Benedetti P, Panicci P, Maneschi F, et al. Surgery by minilaparotomy in benign gynecological disease. Obstet Gynecol. 1996;87:456-459.
11. Jansen FW, Tanahotoe S, Veselie M, et al. Laparoscopic aspiration of ovarian cysts: an unreliable technique in primary diagnosis of sonographically benign ovarian lesions. Gynecol Endosc. 1997;6:363-367.
12. Parker WH. Laparoscopic management of the adnexal mass in postmenopausal women. J Gynecol Tech. 1995;1:3-5.
13. Guerriero S, Ajossa S, Mais V, et al. Prelaparoscopic assessment of ovarian cysts in reproductive-age women. Gynecol Endosc. 1997;6:157-167.
14. Fowler JM, Carter JR. Laparoscopic management of the adnexal mass in postmenopausal women. J Gynecol Tech. 1995;1:7-10.
15. McCormick JB, Fitzgibbons JP. Instrument for aspiration of large ovarian cysts. Obstet Gynecol. 1967;29:869-870.
16. Yamada T, Okamoto Y, Kasematsu H. Use of the Sand balloon catheter for the laparoscopic surgery of benign ovarian cysts. Gynecol Endosc. 1999;9:51-54.
17. Shozu M, Segawa T, Sumitani H, et al. Leak-proof puncture of ovarian cysts: instant mounting of plastic bag using cyanoacrylate adhesive. Obstet Gynecol. 2001;97:1007-1010.
Defect-directed reconstruction: The common-sense technique for rectocele repair
- Tears can almost always be identified without difficulty with the aid of edge-grasping clamps at surgery and a rectal examining finger.
- We can restore the natural anatomic integrity of the rectovaginal septum by reuniting the torn edges, thus eliminating the rectocele.
- The most common tears are transverse. Others include U-shaped, linear, and combined tears.
Why did it take us 2 centuries to learn how to do it right? Posterior pelvic repair for the correction of rectocele combined with restoration of the perineal body has been on gynecologic OR schedules since time immemorial, so one would think we knew what we were doing. Yet only in the past 10 years has there been progressive general recognition of the true nature of the anatomic lesions responsible for rectocele formation, thereby finally pointing us in the right direction.
Although it was always assumed, correctly, that the major factor initiating rectoceles is tissue trauma sustained in vaginal birth, traditionally we were taught, and we believed, that the end result was attenuation and deterioration of the rectovaginal septum (RVS) connective tissue. Similar tissue changes also initiated at vaginal delivery were ascribed to the proximal and distal connective tissue attachments of the RVS, respectively; the cardinal-uterosacral ligament complex; and the perineal body—but tears were never entertained in our perceptions. We know, as emphasized by Nichols in the 1960s and 1970s,1-3 that all of these elements, together with the bilateral RVS connection to levator fascia plus the levator plate of the pelvic floor, constitute the normal vaginal axis when all elements are intact.
To be concise, the disrupted vaginal axis, including its 2 major anatomic abnormalities, rectocele and perineal body defects, was thought to be the direct result of disintegration over time after the initiating shock of vaginal delivery. Specific tissue tears were, until now, an unrealized concept. As a result, we routinely resorted to makeshift methods in attempts to restore posterior pelvic configuration. Today, thanks to A. Cullen Richardson, we are much better informed.4,5
Sutured reunion of RVS tears key to rectocele eradication
This article centers on today’s concepts of the defects isolated within the RVS, a critical part of the vaginal axis. No longer is there any question about the key role of sutured reunion of RVS tears in eradicating rectocele. We must acknowledge, however, that such anatomic restoration alone is insufficient without simultaneous fixed suspension of the posterior vaginal vault above and perineal body reconstitution below, as Nichols emphasized.3
The overall goal in corrective posterior pelvic surgery must always be restoration of the normal vaginal axis in order to preserve the pelvic valve mechanism, as described by Porges.6
The primary reason for connective tissue defects is tears, not attenuation. Richardson dramatically and conclusively demonstrated this fact in the early 1990s, after prolonged and intensive investigation of the anatomy.4,5,7 This astonishing disclosure was nowhere more obvious than in the defective RVS, where discrete separations in otherwise intact tissue could be uncovered and reattached by simple standard stitching technique. We had simply never looked for these torn segments (which were never obvious), so we never found them. Once we were convinced of Richardson’s concepts, the torn edges became easily identifiable in practically every case of rectocele. Suddenly, we were able to restore the natural anatomic integrity of the RVS by reuniting these torn edges. With absolute confidence, we eliminated the rectocele.8-16
The critical digital technique
The key to this logical operative technique was the rectal examining finger. Using this digital strategy, not only could we definitively outline the rectocele on outpatient examination, but we could delineate the exact extent of the rectocele intraoperatively. This is accomplished by performing the first rectal exam just before vaginal mucosal dissection, and then, most accurately, after full dissection and exposure of the entire operative field, thus clearly exposing the torn edges in the RVS. A final anorectal exam is performed to check the restored anatomic configuration of the RVS before vaginal mucosal reunification.8
Transverse tears
Transverse tears are the most common: either detachment from below, adjacent to the perineal body, or separation at the top in juxtaposition to the fibrous uterosacral extensions in the area of the cul-de-sac. FIGURES 1 AND 2 convey the “before and after” aspects, showing Allis clamps drawing the retracted edge to the site of the tear followed by the revelation of the actual repair accomplished by interrupted O-caliber sutures, either delayed absorbable or nonabsorbable. A rectally inserted index finger initially demonstrates that, except for scraps of areolar tissue, the anorectal wall is the only layer that prevents the examining finger from falling into the operative field as it produces rectocele simulation with forward pressure. When the clamps draw the long-torn RVS segments into normal anatomic positions, the anorectal finger cannot advance at all, despite strong effort, because of the natural firm barrier effect of the anatomically restored RVS.8
FIGURE 1 Lower transverse tear
FIGURE 2 Upper transverse tear
U-shaped tears
Another not uncommon type of defect can be U-shaped, either at the bottom, as depicted in FIGURE 3, or at the top of the posterior pelvic compartment. Similarly linear (longitudinal) tears near one side or the other directly adjacent to the pelvic sidewalls, rarely in the midline, are not unusual and can be repaired in the same manner.
FIGURE 3 U-shaped tear
Hockey-stick tears
Occasionally, a hockey-stick lesion, combining a longitudinal tear and a transverse tear in continuity, as shown in FIGURE 4, is discovered.
FIGURE 4 Hockey-stick tear
Double defect
A more uncommon type of combined or double defect, in which the Denonvilliers fascia (RVS) has been torn both adjacent to the vault and in the perineal area but retains strong attachments bilaterally, is shown in FIGURE 5.
FIGURE 5 Double defect
A contented vaginal environment
The major breakthrough is the concept that tears in the Denonvilliers fascia, not attenuation, are the cause of rectoceles. These tears—transverse, longitudinal, U-shaped, multidirectional, even stellate—can be identified almost always without difficulty with the aid of a rectal examining finger and edge-grasping clamps at surgery. The repair itself is not only anatomically logical but also much easier and more confidence-inspiring than the traditional, now archaic, method of fishing around for scraps of levator fascia and muscle to approximate, under tension, in the midline.
This defect-directed repair, born of common sense (ie, comprehension and application of normal anatomy), is structurally nonconstrictive and functionally nonrestrictive—truly a contented vaginal environment.
All sketches from TeLinde’s Operative Gynecology, 9th edition, with permission from Lippincott, Williams, and Wilkins, Publishers.
Dr. Grody reports no financial relationship with any companies whose products are mentioned in this article.
1. Nichols DH. Posterior colporrhaphy and perineorrhaphy: separate and distinct operations. Am J Obstet Gynecol. 1991;164:714-721.
2. Nichols DH, Milley PS, Randall CL. Significance of restoration of normal vaginal depth and axis. Obstet Gynecol. 1970;36:251-256.
3. Nichols DH, Randall CS. Vaginal Surgery. 3rd ed. Baltimore, Md: Williams & Wilkins; 1989;21.-
4. Richardson AC. The rectovaginal septum revisited: its relationship to rectocele and its importance to rectocele repair. Clin Obstet Gynecol. 1993;36:976-983.
5. Richardson AC. The anatomic defects in rectocele and enterocele. J Pelv Surg. 1995;1:214.-
6. Porges RF, Sinden SW. Long-term analysis of the surgical management of pelvic support defects. Am J Obstet Gynecol. 1994;171:1518-1528.
7. Edmonds PB, Richardson AC. Anatomical Approach to Rectocele Repair [videotape]. St. Louis, Mo: Society of Gynecologic Surgeons; 1993.
8. Grody MHT. Posterior compartment defects. In: Rock JA, Jones III HW, eds. TeLinde’s Operative Gynecology. 9th ed. Philadelphia, Pa: Lippincott, Williams and Wilkins; 2003;966-985.
9. Cundiff GW, Weidner AC, Visco AG, Addison WA, Bump RC. An anatomic and functional assessment of the discrete defect rectocele repair. Am J Obstet Gynecol. 1998;179:1456-1457.
10. Kenton K, Shott S, Brubaker L. Outcome after rectovaginal fascia reattachment for rectocele repair. Am J Obstet Gynecol. 1999;181:360-364.
11. Porter WE, Steele A, Walsh P, Kohli N, Karram MM. The anatomic and functional outcomes of defect-specific rectocele repairs. Am J Obstet Gynecol. 1999;181:1353-13549.
12. Glavind K, Madsen H. A prospective study of the discrete fascial defect rectocele repair. Acta Obstet Gynecol Scand. 2000;79:145-147.
13. Cundiff GW. Defect Directed Rectocele Repairs: Restorative and Compensatory Techniques [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2001.
14. Maccarone JL, Caraballo R, Holzberg A, Grody MHT. Innovative Defect-specific Posterior Pelvic Surgery: Triggered Ligament Sutures and Collagen Graft [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2003.
15. Caraballo R, Maccarone JL, Holzberg AS, Grody MHT. New Concepts in Reconstructive Pelvic Surgery: Slings, Collagen Matrix Grafts, Triggered Sutures [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2003.
16. Singh K, Cortes E, Reid WMN. Evaluation of the fascial technique for surgical repair of isolated posterior vaginal wall prolapse. Obstet Gynecol. 2003;101:320-324.
- Tears can almost always be identified without difficulty with the aid of edge-grasping clamps at surgery and a rectal examining finger.
- We can restore the natural anatomic integrity of the rectovaginal septum by reuniting the torn edges, thus eliminating the rectocele.
- The most common tears are transverse. Others include U-shaped, linear, and combined tears.
Why did it take us 2 centuries to learn how to do it right? Posterior pelvic repair for the correction of rectocele combined with restoration of the perineal body has been on gynecologic OR schedules since time immemorial, so one would think we knew what we were doing. Yet only in the past 10 years has there been progressive general recognition of the true nature of the anatomic lesions responsible for rectocele formation, thereby finally pointing us in the right direction.
Although it was always assumed, correctly, that the major factor initiating rectoceles is tissue trauma sustained in vaginal birth, traditionally we were taught, and we believed, that the end result was attenuation and deterioration of the rectovaginal septum (RVS) connective tissue. Similar tissue changes also initiated at vaginal delivery were ascribed to the proximal and distal connective tissue attachments of the RVS, respectively; the cardinal-uterosacral ligament complex; and the perineal body—but tears were never entertained in our perceptions. We know, as emphasized by Nichols in the 1960s and 1970s,1-3 that all of these elements, together with the bilateral RVS connection to levator fascia plus the levator plate of the pelvic floor, constitute the normal vaginal axis when all elements are intact.
To be concise, the disrupted vaginal axis, including its 2 major anatomic abnormalities, rectocele and perineal body defects, was thought to be the direct result of disintegration over time after the initiating shock of vaginal delivery. Specific tissue tears were, until now, an unrealized concept. As a result, we routinely resorted to makeshift methods in attempts to restore posterior pelvic configuration. Today, thanks to A. Cullen Richardson, we are much better informed.4,5
Sutured reunion of RVS tears key to rectocele eradication
This article centers on today’s concepts of the defects isolated within the RVS, a critical part of the vaginal axis. No longer is there any question about the key role of sutured reunion of RVS tears in eradicating rectocele. We must acknowledge, however, that such anatomic restoration alone is insufficient without simultaneous fixed suspension of the posterior vaginal vault above and perineal body reconstitution below, as Nichols emphasized.3
The overall goal in corrective posterior pelvic surgery must always be restoration of the normal vaginal axis in order to preserve the pelvic valve mechanism, as described by Porges.6
The primary reason for connective tissue defects is tears, not attenuation. Richardson dramatically and conclusively demonstrated this fact in the early 1990s, after prolonged and intensive investigation of the anatomy.4,5,7 This astonishing disclosure was nowhere more obvious than in the defective RVS, where discrete separations in otherwise intact tissue could be uncovered and reattached by simple standard stitching technique. We had simply never looked for these torn segments (which were never obvious), so we never found them. Once we were convinced of Richardson’s concepts, the torn edges became easily identifiable in practically every case of rectocele. Suddenly, we were able to restore the natural anatomic integrity of the RVS by reuniting these torn edges. With absolute confidence, we eliminated the rectocele.8-16
The critical digital technique
The key to this logical operative technique was the rectal examining finger. Using this digital strategy, not only could we definitively outline the rectocele on outpatient examination, but we could delineate the exact extent of the rectocele intraoperatively. This is accomplished by performing the first rectal exam just before vaginal mucosal dissection, and then, most accurately, after full dissection and exposure of the entire operative field, thus clearly exposing the torn edges in the RVS. A final anorectal exam is performed to check the restored anatomic configuration of the RVS before vaginal mucosal reunification.8
Transverse tears
Transverse tears are the most common: either detachment from below, adjacent to the perineal body, or separation at the top in juxtaposition to the fibrous uterosacral extensions in the area of the cul-de-sac. FIGURES 1 AND 2 convey the “before and after” aspects, showing Allis clamps drawing the retracted edge to the site of the tear followed by the revelation of the actual repair accomplished by interrupted O-caliber sutures, either delayed absorbable or nonabsorbable. A rectally inserted index finger initially demonstrates that, except for scraps of areolar tissue, the anorectal wall is the only layer that prevents the examining finger from falling into the operative field as it produces rectocele simulation with forward pressure. When the clamps draw the long-torn RVS segments into normal anatomic positions, the anorectal finger cannot advance at all, despite strong effort, because of the natural firm barrier effect of the anatomically restored RVS.8
FIGURE 1 Lower transverse tear
FIGURE 2 Upper transverse tear
U-shaped tears
Another not uncommon type of defect can be U-shaped, either at the bottom, as depicted in FIGURE 3, or at the top of the posterior pelvic compartment. Similarly linear (longitudinal) tears near one side or the other directly adjacent to the pelvic sidewalls, rarely in the midline, are not unusual and can be repaired in the same manner.
FIGURE 3 U-shaped tear
Hockey-stick tears
Occasionally, a hockey-stick lesion, combining a longitudinal tear and a transverse tear in continuity, as shown in FIGURE 4, is discovered.
FIGURE 4 Hockey-stick tear
Double defect
A more uncommon type of combined or double defect, in which the Denonvilliers fascia (RVS) has been torn both adjacent to the vault and in the perineal area but retains strong attachments bilaterally, is shown in FIGURE 5.
FIGURE 5 Double defect
A contented vaginal environment
The major breakthrough is the concept that tears in the Denonvilliers fascia, not attenuation, are the cause of rectoceles. These tears—transverse, longitudinal, U-shaped, multidirectional, even stellate—can be identified almost always without difficulty with the aid of a rectal examining finger and edge-grasping clamps at surgery. The repair itself is not only anatomically logical but also much easier and more confidence-inspiring than the traditional, now archaic, method of fishing around for scraps of levator fascia and muscle to approximate, under tension, in the midline.
This defect-directed repair, born of common sense (ie, comprehension and application of normal anatomy), is structurally nonconstrictive and functionally nonrestrictive—truly a contented vaginal environment.
All sketches from TeLinde’s Operative Gynecology, 9th edition, with permission from Lippincott, Williams, and Wilkins, Publishers.
Dr. Grody reports no financial relationship with any companies whose products are mentioned in this article.
- Tears can almost always be identified without difficulty with the aid of edge-grasping clamps at surgery and a rectal examining finger.
- We can restore the natural anatomic integrity of the rectovaginal septum by reuniting the torn edges, thus eliminating the rectocele.
- The most common tears are transverse. Others include U-shaped, linear, and combined tears.
Why did it take us 2 centuries to learn how to do it right? Posterior pelvic repair for the correction of rectocele combined with restoration of the perineal body has been on gynecologic OR schedules since time immemorial, so one would think we knew what we were doing. Yet only in the past 10 years has there been progressive general recognition of the true nature of the anatomic lesions responsible for rectocele formation, thereby finally pointing us in the right direction.
Although it was always assumed, correctly, that the major factor initiating rectoceles is tissue trauma sustained in vaginal birth, traditionally we were taught, and we believed, that the end result was attenuation and deterioration of the rectovaginal septum (RVS) connective tissue. Similar tissue changes also initiated at vaginal delivery were ascribed to the proximal and distal connective tissue attachments of the RVS, respectively; the cardinal-uterosacral ligament complex; and the perineal body—but tears were never entertained in our perceptions. We know, as emphasized by Nichols in the 1960s and 1970s,1-3 that all of these elements, together with the bilateral RVS connection to levator fascia plus the levator plate of the pelvic floor, constitute the normal vaginal axis when all elements are intact.
To be concise, the disrupted vaginal axis, including its 2 major anatomic abnormalities, rectocele and perineal body defects, was thought to be the direct result of disintegration over time after the initiating shock of vaginal delivery. Specific tissue tears were, until now, an unrealized concept. As a result, we routinely resorted to makeshift methods in attempts to restore posterior pelvic configuration. Today, thanks to A. Cullen Richardson, we are much better informed.4,5
Sutured reunion of RVS tears key to rectocele eradication
This article centers on today’s concepts of the defects isolated within the RVS, a critical part of the vaginal axis. No longer is there any question about the key role of sutured reunion of RVS tears in eradicating rectocele. We must acknowledge, however, that such anatomic restoration alone is insufficient without simultaneous fixed suspension of the posterior vaginal vault above and perineal body reconstitution below, as Nichols emphasized.3
The overall goal in corrective posterior pelvic surgery must always be restoration of the normal vaginal axis in order to preserve the pelvic valve mechanism, as described by Porges.6
The primary reason for connective tissue defects is tears, not attenuation. Richardson dramatically and conclusively demonstrated this fact in the early 1990s, after prolonged and intensive investigation of the anatomy.4,5,7 This astonishing disclosure was nowhere more obvious than in the defective RVS, where discrete separations in otherwise intact tissue could be uncovered and reattached by simple standard stitching technique. We had simply never looked for these torn segments (which were never obvious), so we never found them. Once we were convinced of Richardson’s concepts, the torn edges became easily identifiable in practically every case of rectocele. Suddenly, we were able to restore the natural anatomic integrity of the RVS by reuniting these torn edges. With absolute confidence, we eliminated the rectocele.8-16
The critical digital technique
The key to this logical operative technique was the rectal examining finger. Using this digital strategy, not only could we definitively outline the rectocele on outpatient examination, but we could delineate the exact extent of the rectocele intraoperatively. This is accomplished by performing the first rectal exam just before vaginal mucosal dissection, and then, most accurately, after full dissection and exposure of the entire operative field, thus clearly exposing the torn edges in the RVS. A final anorectal exam is performed to check the restored anatomic configuration of the RVS before vaginal mucosal reunification.8
Transverse tears
Transverse tears are the most common: either detachment from below, adjacent to the perineal body, or separation at the top in juxtaposition to the fibrous uterosacral extensions in the area of the cul-de-sac. FIGURES 1 AND 2 convey the “before and after” aspects, showing Allis clamps drawing the retracted edge to the site of the tear followed by the revelation of the actual repair accomplished by interrupted O-caliber sutures, either delayed absorbable or nonabsorbable. A rectally inserted index finger initially demonstrates that, except for scraps of areolar tissue, the anorectal wall is the only layer that prevents the examining finger from falling into the operative field as it produces rectocele simulation with forward pressure. When the clamps draw the long-torn RVS segments into normal anatomic positions, the anorectal finger cannot advance at all, despite strong effort, because of the natural firm barrier effect of the anatomically restored RVS.8
FIGURE 1 Lower transverse tear
FIGURE 2 Upper transverse tear
U-shaped tears
Another not uncommon type of defect can be U-shaped, either at the bottom, as depicted in FIGURE 3, or at the top of the posterior pelvic compartment. Similarly linear (longitudinal) tears near one side or the other directly adjacent to the pelvic sidewalls, rarely in the midline, are not unusual and can be repaired in the same manner.
FIGURE 3 U-shaped tear
Hockey-stick tears
Occasionally, a hockey-stick lesion, combining a longitudinal tear and a transverse tear in continuity, as shown in FIGURE 4, is discovered.
FIGURE 4 Hockey-stick tear
Double defect
A more uncommon type of combined or double defect, in which the Denonvilliers fascia (RVS) has been torn both adjacent to the vault and in the perineal area but retains strong attachments bilaterally, is shown in FIGURE 5.
FIGURE 5 Double defect
A contented vaginal environment
The major breakthrough is the concept that tears in the Denonvilliers fascia, not attenuation, are the cause of rectoceles. These tears—transverse, longitudinal, U-shaped, multidirectional, even stellate—can be identified almost always without difficulty with the aid of a rectal examining finger and edge-grasping clamps at surgery. The repair itself is not only anatomically logical but also much easier and more confidence-inspiring than the traditional, now archaic, method of fishing around for scraps of levator fascia and muscle to approximate, under tension, in the midline.
This defect-directed repair, born of common sense (ie, comprehension and application of normal anatomy), is structurally nonconstrictive and functionally nonrestrictive—truly a contented vaginal environment.
All sketches from TeLinde’s Operative Gynecology, 9th edition, with permission from Lippincott, Williams, and Wilkins, Publishers.
Dr. Grody reports no financial relationship with any companies whose products are mentioned in this article.
1. Nichols DH. Posterior colporrhaphy and perineorrhaphy: separate and distinct operations. Am J Obstet Gynecol. 1991;164:714-721.
2. Nichols DH, Milley PS, Randall CL. Significance of restoration of normal vaginal depth and axis. Obstet Gynecol. 1970;36:251-256.
3. Nichols DH, Randall CS. Vaginal Surgery. 3rd ed. Baltimore, Md: Williams & Wilkins; 1989;21.-
4. Richardson AC. The rectovaginal septum revisited: its relationship to rectocele and its importance to rectocele repair. Clin Obstet Gynecol. 1993;36:976-983.
5. Richardson AC. The anatomic defects in rectocele and enterocele. J Pelv Surg. 1995;1:214.-
6. Porges RF, Sinden SW. Long-term analysis of the surgical management of pelvic support defects. Am J Obstet Gynecol. 1994;171:1518-1528.
7. Edmonds PB, Richardson AC. Anatomical Approach to Rectocele Repair [videotape]. St. Louis, Mo: Society of Gynecologic Surgeons; 1993.
8. Grody MHT. Posterior compartment defects. In: Rock JA, Jones III HW, eds. TeLinde’s Operative Gynecology. 9th ed. Philadelphia, Pa: Lippincott, Williams and Wilkins; 2003;966-985.
9. Cundiff GW, Weidner AC, Visco AG, Addison WA, Bump RC. An anatomic and functional assessment of the discrete defect rectocele repair. Am J Obstet Gynecol. 1998;179:1456-1457.
10. Kenton K, Shott S, Brubaker L. Outcome after rectovaginal fascia reattachment for rectocele repair. Am J Obstet Gynecol. 1999;181:360-364.
11. Porter WE, Steele A, Walsh P, Kohli N, Karram MM. The anatomic and functional outcomes of defect-specific rectocele repairs. Am J Obstet Gynecol. 1999;181:1353-13549.
12. Glavind K, Madsen H. A prospective study of the discrete fascial defect rectocele repair. Acta Obstet Gynecol Scand. 2000;79:145-147.
13. Cundiff GW. Defect Directed Rectocele Repairs: Restorative and Compensatory Techniques [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2001.
14. Maccarone JL, Caraballo R, Holzberg A, Grody MHT. Innovative Defect-specific Posterior Pelvic Surgery: Triggered Ligament Sutures and Collagen Graft [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2003.
15. Caraballo R, Maccarone JL, Holzberg AS, Grody MHT. New Concepts in Reconstructive Pelvic Surgery: Slings, Collagen Matrix Grafts, Triggered Sutures [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2003.
16. Singh K, Cortes E, Reid WMN. Evaluation of the fascial technique for surgical repair of isolated posterior vaginal wall prolapse. Obstet Gynecol. 2003;101:320-324.
1. Nichols DH. Posterior colporrhaphy and perineorrhaphy: separate and distinct operations. Am J Obstet Gynecol. 1991;164:714-721.
2. Nichols DH, Milley PS, Randall CL. Significance of restoration of normal vaginal depth and axis. Obstet Gynecol. 1970;36:251-256.
3. Nichols DH, Randall CS. Vaginal Surgery. 3rd ed. Baltimore, Md: Williams & Wilkins; 1989;21.-
4. Richardson AC. The rectovaginal septum revisited: its relationship to rectocele and its importance to rectocele repair. Clin Obstet Gynecol. 1993;36:976-983.
5. Richardson AC. The anatomic defects in rectocele and enterocele. J Pelv Surg. 1995;1:214.-
6. Porges RF, Sinden SW. Long-term analysis of the surgical management of pelvic support defects. Am J Obstet Gynecol. 1994;171:1518-1528.
7. Edmonds PB, Richardson AC. Anatomical Approach to Rectocele Repair [videotape]. St. Louis, Mo: Society of Gynecologic Surgeons; 1993.
8. Grody MHT. Posterior compartment defects. In: Rock JA, Jones III HW, eds. TeLinde’s Operative Gynecology. 9th ed. Philadelphia, Pa: Lippincott, Williams and Wilkins; 2003;966-985.
9. Cundiff GW, Weidner AC, Visco AG, Addison WA, Bump RC. An anatomic and functional assessment of the discrete defect rectocele repair. Am J Obstet Gynecol. 1998;179:1456-1457.
10. Kenton K, Shott S, Brubaker L. Outcome after rectovaginal fascia reattachment for rectocele repair. Am J Obstet Gynecol. 1999;181:360-364.
11. Porter WE, Steele A, Walsh P, Kohli N, Karram MM. The anatomic and functional outcomes of defect-specific rectocele repairs. Am J Obstet Gynecol. 1999;181:1353-13549.
12. Glavind K, Madsen H. A prospective study of the discrete fascial defect rectocele repair. Acta Obstet Gynecol Scand. 2000;79:145-147.
13. Cundiff GW. Defect Directed Rectocele Repairs: Restorative and Compensatory Techniques [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2001.
14. Maccarone JL, Caraballo R, Holzberg A, Grody MHT. Innovative Defect-specific Posterior Pelvic Surgery: Triggered Ligament Sutures and Collagen Graft [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2003.
15. Caraballo R, Maccarone JL, Holzberg AS, Grody MHT. New Concepts in Reconstructive Pelvic Surgery: Slings, Collagen Matrix Grafts, Triggered Sutures [videotape]. Washington, DC: American College of Obstetricians and Gynecologists; 2003.
16. Singh K, Cortes E, Reid WMN. Evaluation of the fascial technique for surgical repair of isolated posterior vaginal wall prolapse. Obstet Gynecol. 2003;101:320-324.
Cutting the risk of hysteroscopic complications
- Preoperative treatment with a gonadotropinreleasing hormone agonist increases the odds of operative complications by a factor of 4 to 7.
- Preoperative cervical ripening reduced the need for cervical dilation, minimized cervical complications, and reduced operative time.
- CO2 should never be used for operative hysteroscopic procedures because of the high risk of CO2 embolism.
- Ultrasound guidance may improve outcomes in selected hysteroscopic procedures.
It is a valuable tool in the evaluation and treatment of infertility, recurrent pregnancy loss, and abnormal and postmenopausal uterine bleeding, and is useful when saline infusion sonography findings are equivocal.
Further, if a global ablation device fails, the surgeon can convert to hysteroscopic ablation rather than abandon the procedure altogether. This is not as unusual as it might appear: In US Food and Drug Administration trials, there was a staggering 10% to 22% malfunction of global ablation technology.1
Safe, easily learned
Although gynecologists are beginning to embrace this modality, many physicians avoid it because of inadequate training or exaggerated fears of complications. In reality, hysteroscopy is one of the safest and most easily acquired surgical skills in gynecology. For example, in a prospective evaluation of 13,600 diagnostic and operative hysteroscopic procedures performed at 63 hospitals in the Netherlands—which involved both established surgeons and residents—Jansen et al2 found an astonishingly low complication rate of 0.28%, with no deaths.
Proper selection and treatment of patients and prompt intervention minimize complications as well as legal risks. Surgical misadventures and lawsuits occur with delayed intervention, failure to recognize pathology or risky conditions, and inadequate preventive maneuvers.
Overall, emphasis on safety is vital to success, and thorough awareness of potential complications is just as important.
Three types of complications
Complications fall into 3 categories (TABLE):
- Procedure-related
- Media-related
- Postoperative
PROCEDURE-RELATED COMPLICATIONS
Complication rates
In a retrospective investigation, Propst et al3 determined the rate of complications associated with specific hysteroscopic procedures. Demographic data and medical histories were collected for 925 women who had operative hysteroscopy in 1995 and 1996. The overall complication rate was 2.7%. Myomectomy and resection of uterine septa carried the greatest odds of complications; polypectomy and endometrial ablation had the lowest. Preoperative treatment with a gonadotropinreleasing hormone (GnRH) agonist increased the odds of complications by a factor of 4 to 7. Women under age 50 were more likely to experience complications than those over 50.
In the study by Jansen et al,2 38 complica-tions occurred in the 13,600 procedures. The greatest risk of complications occurred with adhesiolysis (4.48%), followed by endometrial resection (0.81%), myomectomy (0.75%), and polypectomy (0.38%).
Cervical entry requires special attention
Almost half of the complications in the Jansen study were related to cervical entry, so caution and, perhaps, preoperative cervical ripening are advised. Many premenopausal subjects were given GnRH analogues, which may render the cervix more resistant to dilation. Complications associated with a stenotic cervix include a cervical tear, creation of a false cervical passage, and uterine perforation.
Cervical ripening may help prevent uterine perforation. The most common complication, occurring in 14.2 cases per thousand, is uterine perforation.2 The risk of this is highest in postpartum procedures, followed by procedures in postmenopausal, then perimenopausal, women. Patients with endometrial cancer also have a higher rate of perforation.
Risk factors for uterine perforation include:
- nulliparity
- menopause
- use of GnRH agonists
- prior cone biopsy
- markedly retroverted uterus
- undue force
Vaginal or oral misoprostol for cervical ripening prior to operative hysteroscopy was evaluated in a randomized trial.4 Researchers found a reduced need for cervical dilation, a minimum of cervical complications, and reduced operative time in study patients compared with controls.
When 400 μg oral misoprostol is given 12 and 24 hours before surgery, it also softens the cervix and eases dilation.5 Although misoprostol has several bothersome side effects (such as lower abdominal pain and slight vaginal bleeding), few if any prevent its use.
Signs of perforation. Patients who sustain uterine perforation with subsequent intraperitoneal bleeding often complain of pain in the abdomen and shoulder, and experience hemodynamic instability. A quick sonographic survey of the abdomen will demonstrate free intraperitoneal fluid. (It is rare for much intraperitoneal fluid to accumulate by transtubal regurgitation during operative hysteroscopy, despite the quantity of fluid used.)
If perforation is suspected, laparoscopy or laparotomy is necessary to clarify the cause of pain, unstable vital signs, or free fluid visualized by ultrasound.6
Exercise extra care and precautions in women who have had a prior cesarean section, myomectomy, or uterine perforation. Complete visualization of uterine landmarks is necessary during operative hysteroscopy to exclude uterine dehiscence, sacculation, and perforation. Prior uterine surgery may cause myometrial weakness and lead to possible perforation. Do not proceed if abnormal uterine morphology is detected. If uterine perforation occurs, injury to bladder and bowel is possible when electrical energy is applied to a uterine wall compromised by prior surgery. Strict visualization of uterine anatomy is critical in this population so that bowel or bladder burns can be avoided.
MEDIA-RELATED COMPLICATIONS
Notorious complications and several recent lawsuits have stemmed from fluid overload. A common element has been the physician’s lack of awareness of how rapidly complications can arise, and what signs and symptoms are specific to the fluid used.
Monitor fluids vigilantly
Operative hysteroscopy must be performed in a fluid medium. The type of fluid depends on the surgeon’s preference and the instrument utilized, but any fluid can be associated with complications. Fluid choices with monopolar instruments include glycine 1.5%, a mixture of sorbitol 3% and mannitol 0.54%, and mannitol 5%. These are frequently used with the continuous-flow resectoscope. Bipolar operative hysteroscopy can be performed using saline.
The solution to media-related complications is basic: vigilant monitoring of fluids. A cavalier attitude, poor fluid documentation, and failure to respond to complications can lead to trouble. If fluid overload occurs, comanagement and consultation with an intensive care specialist is advised.
Distention media
Among the options for distention media in operative and diagnostic hysteroscopy are high-viscosity dextran 70 and low-viscosity fluids such as hypotonic, electrolyte-free and isotonic, electrolyte-containing solutions. The popularity of dextran 70 is waning, however. While it is immiscible with blood, significant complications have been reported.
Signs of anaphylactic reactions to dextran 70 include acute hypotension, hypoxia, pulmonary edema, fluid overload, fulminant coagulopathies, and anemia. The surgeon must operate quickly, minimize endometrial trauma, use continuous pulse oximetry, and obtain a preoperative coagulation panel.
Dextran 70 also can ruin operative hysteroscopes if they are not cleaned promptly and thoroughly after use.
Hypotonic, electrolyte-free solutions. With hypotonic, electrolyte-free solutions such as glycine 1.5%, early recognition of possible complications, including hyponatremic hypervolemia, is vital. For example, when glycine and sorbitol are metabolized, free water accumulates and the body attempts to achieve homeostasis through compensatory mechanisms such as osmosis, which moves free water into extracellular and intracellular spaces. This can lead to increased free water in the brain, resulting in cerebral edema, rising intracranial pressure, and cellular necrosis.
The cerebral cation pump normally pumps osmotically active cations into the extracellular space, thereby minimizing cerebral edema. However, this pump is inhibited by estrogen, so the compensatory mechanism is diminished.
Classic clinical features of hyponatremic hypervolemia include apprehension, confusion, fatigue, headache, mental agitation, nausea, visual disturbances (including blindness), vomiting, and weakness. These complications are more readily apparent when regional anesthesia is used rather than general anesthesia.
If hyponatremic hypervolemia goes unrecognized, bradycardia and hypertension can ensue, followed rapidly by cerebral and pulmonary edema and cardiovascular collapse. In addition, glycine 1.5% is metabolized to glycolic acid and ammonia. Free ammonia is associated with central nervous system disorders. Recognition and prompt treatment by an intensivist may prevent permanent neurologic sequelae, death, and lawsuits.7
Isotonic, electrolyte-containing solutions. Mannitol 5% is electrolyte poor but isotonic, creating less risk for hypo-osmolality. However, dilutional hyponatremia (ie, low sodium levels) can still occur.
Advantages of bipolar instruments. To minimize complications from hypotonic, electrolyte-free solutions, manufacturers developed operative hysteroscopes that can function in a bipolar environment. Bipolar instruments can operate in isotonic, physiologic, electrolyte-containing media. Hyponatremia and hypo-osmolality cannot occur with normal saline or Ringer’s lactate, but fluid overload can. (Fluid overload with saline can cause pulmonary edema and congestive heart failure.)
How much fluid will be absorbed? The answer depends on factors including surface area of the surgical field, duration of surgery, opened venous channels, type of irrigation fluid used, and pressure of the delivery system. Modern gynecologic suites employ fluid irrigation systems that continuously measure input and output, with alarms that signal a predetermined fluid deficit. The alarm indicates the need to halt the procedure and quickly evaluate the patient. Careful attention to the recommendations of Loffer et al8 would lead to fewer complications from fluid mismanagement.
Appropriate use of CO2
High risk of embolism with CO2 in operative procedures. Although diagnostic hysteroscopic procedures often are performed with carbon dioxide (CO2), operative procedures never should be. The reason: the high risk of CO2 embolism that occurs with open venous channels and vascular endometrium. The choice between CO2 and fluid medium for diagnostic hysteroscopy often is determined by physician preference and the presence of uterine bleeding. Many gynecologists prefer CO2 for its optical clarity and patient comfort during insufflation.9
Purge tubing of room air before each procedure. Embolic complications with CO2 have been recorded with use of the neodymium: yttrium aluminum garnet (Nd:YAG) laser and during operative procedures. Less well known are the adverse sequelae that can occur with room air prior to beginning the procedure. It is critical to purge the entire tubal system with CO2 prior to instrumentation, since up to 40 cm3 of room air may be insufflated into a patient when 200 cm of connective tubing with a 0.5-cm lumen is used.10 Wait for several minutes before starting the procedure so that the whole system is purged.
One of the greatest concerns about endometrial ablation is that diagnosis of endometrial cancer will be delayed because the endometrial cavity has been obliterated. Vilos19 recently reviewed the salient characteristics and findings in women treated by endometrial ablation who subsequently developed endometrial cancer. A review of the individual cases revealed that most of these patients had numerous risk factors for endometrial cancer.
Review risk factors, chronic conditions
Many patients with abnormal bleeding also have risk factors for endometrial cancer, as well as medical conditions that increase the likelihood of morbidity with surgery, such as obesity, hypertension, diabetes, and advanced age. In these cases, hysterectomy may be a better option than endometrial ablation. It would be far better to have such high-risk patients cleared for hysterectomy than to chance their becoming an endometrial-ablation “statistic.” If endometrial ablation is performed in these cases, we prevent the egress of blood, foster development of synechiae, render endometrial biopsy difficult or impossible and, potentially, “bury” endometrial cells deeper within the myometrium—all of which contribute to a delayed and “upstaged” diagnosis of endometrial cancer.
Patients at risk of endometrial cancer should undergo a scrupulous and unambiguous work-up and evaluation. Indeterminate endometrial echo and office evaluation that generates biopsy samples designated as “insufficient for diagnosis,” “no endometrial tissue seen,” or “atrophy” should raise suspicion. These patients require full visualization of the endometrium.
Heightened risk during perimenopause
Newer ablation techniques that utilize global therapy make it paramount that perimenopausal women undergo scrupulous evaluation. Until much more information is available, endometrial ablation should be avoided in patients with endometrial hyperplasia, particularly with atypia. While some gynecologists may be persuaded to consider endometrial ablation as a minimally invasive procedure compared to hysterectomy, the risk of delayed diagnosis of endometrial cancer is of paramount concern. The treatment of choice for these patients remains medical therapy with oral progesterone and, possibly, longterm use of a levonorgestrel-releasing intrauterine system. If this fails, hysterectomy is advisable. As Cooper20 aptly states, “Conservative, nonextirpative procedures offer no life raft” compared with hysterectomy, which covers many missed diagnoses.
No risk of spreading cancer cells
Some gynecologists have worried about the risk of disseminating endometrial cancer cells during hysteroscopy. However, Kudela and Pilka21 studied the true risk in women undergoing blind dilation and curettage and hysteroscopy performed with a fluid medium. Cul-de-sac aspiration prior to instrumentation and at the conclusion of the procedure demonstrated no increased risk of positive cytology. They are continuing a Phase II trial comparing outcomes of both groups over 5 years.
6 most common symptoms of venous or air emboli. Anesthesiologists and gynecologists must be vigilant to prevent venous or air emboli. Munro et al12 succinctly outline the 6 most common symptoms:
- pulmonary hypertension
- hypercarbia
- hypoxia
- arrhythmias
- tachypnea
- systemic hypotension
Beware of a drop in end-tidal CO2. The most common sign of impending cardiovascular collapse is a sudden decrease in end-tidal CO2, when the right cardiac outflow tract is obstructed by CO2, which leads to arterial oxygen (O2) desaturation. If such a decrease is suspected, stop the procedure immediately and administer 100% O2. (Also stop nitrous oxide, if used.) Turn the patient to the left lateral decubitis position and use a central venous catheter to aspirate gas, if necessary. Cardiac massage and a precordial thump may dislodge CO2; unfortunately, high false-positive rates of pre-cordial Doppler make its use impractical.
How to minimize risks
- Avoid coaxial gas cooling tips associated with Nd:YAG crystal lasers
- Avoid a steep Trendelenburg position
- Keep cervix covered with sponge or dilator when operative hysteroscope is removed to minimize air embolism
- Deaerate the equipment prior to surgery Use a low-pressure hysteroscopic CO2 insufflator
- Carefully monitor the patient
- Be highly suspicious when vital signs are unstable
POSTOPERATIVE COMPLICATIONS
Some complications of hysteroscopy may not become clinically evident for months or even years. The most common complications of hysteroscopic endometrial ablation include pregnancy, postablation tubal sterilization syndrome, new or worsening dysmenorrhea, hematometra, endometrial cancer, and failure to completely treat symptoms.
Patients scheduled for hysteroscopy must be informed of potential delayed risks of the procedure. In addition, all reproductive-aged women should be advised that pregnancy is possible after endometrial ablation or operative removal of an intracavitary mass; thus, contraception is crucial. The endometrial tissue is resilient and may regenerate after ablation.
Hematometra: Avoid cervical canal
Hematometra is an infrequent late complication of operative hysteroscopy. If menstruating women or those taking hormone replacement therapy experience cyclic or chronic lower pelvic pain after surgery, scarring or narrowing of the endometrial cavity may be the cause. Approximately 1% to 2% of women who undergo operative hysteroscopy experience this phenomenon. Most cases can be treated with cervical dilation alone.
Since the cervical canal contains no endometrial glands, there is no need to treat this area in women undergoing endometrial ablation. In fact, avoiding this area during treatment is a critical component of successful surgery.
Tubal sterilization syndrome possible after endometrial ablation
Consider this syndrome when a patient undergoing endometrial ablation complains of crampy, cyclic, unilateral or bilateral pelvic pain, possibly accompanied by vaginal spotting. Sometimes a unilateral mass can be palpated, but more commonly tenderness is elicited on pelvic examination.
Ultrasound may demonstrate fluid near the cornual region. Laparoscopy confirms the diagnosis by visualizing a swollen, edematous proximal fallopian tube. Salpingectomy may confirm hematosalpinx, chronic or acute inflammation, or hemosiderin deposits.
Treatment includes bilateral cornual resection and reablation of proximal endometrium, or hysterectomy.14
Pregnancy complications
Endometrial ablation is not to be regarded as a method of contraception. Patients needing birth control should consider concurrent tubal ligation or other reliable methods after this procedure.
The frequency of pregnancy after endometrial ablation ranges from 0.2% to 1.6%, though this data may represent underreporting. Pregnancy outcomes have been dismal in women conceiving after endometrial ablation. Complications include preterm labor, premature delivery, intrauterine growth retardation, prenatal death, postpartum hemorrhage, and placentation problems such as placenta accreta, increta, or percreta, as well as placental abruption.15
Uterine dehiscence and sacculation and extremely thin myometrium have been reported after uterine adhesiolysis, uterine perforation during operative hysteroscopy, and with myoma resections. A high index of suspicion is vital when a gravida presents with pelvic pain, decreased fetal movement, vaginal bleeding, or abnormal uterine masses detected ultrasonographically.
Signs of uterine rupture. Pregnancyrelated complications of operative hysteroscopy can be dramatic and fatal if not recognized quickly, as in the case of uterine rupture. Kerimis et al16 describe uterine rupture in a term pregnancy after hysteroscopic resection of a uterine septum. Severe fetal distress, maternal shoulder pain, and abdominal pain led to an emergency cesarean section. Intraoperative findings included a 7-cm tear from left cornua to right cornua. The original metroplasty, performed with cutting diathermy and laparoscopy, was not accompanied by complications or perforation.
Patients who experience intraoperative complications during metroplasty or deep resection of intramural fibroids should be informed of the risk of uterine rupture so they may consider elective cesarean. Regardless of the mode of delivery, prompt attention is vital if fetal distress is suspected.
Postablation warning signs
Patients undergoing endometrial ablation generally have a quick postoperative return to activity, minimal need for postoperative pain medication, and limited complaints. Beware of patients who make frequent postoperative phone calls and have escalating requirements for pain medication. While bowel and bladder injuries are infrequent—as is postoperative endometritis—these must be vigilantly considered and evaluated when patients complain of persistent pain, fever, and general malaise. Office evaluation is necessary, including thorough abdominal and pelvic examinations. Laboratory testing should include electrolytes, complete blood count, sedimentation rate, ultrasound, and a flat plate of the abdomen (kidneys, ureter, and bladder; upright) may be required. Sometimes a computed tomography scan of the pelvis/abdomen may be needed if perforation with bowel or bladder injury is suspected.
Hysteroscopic fibroid removal may be necessary after UAE
Uterine artery embolization (UAE) is gaining popularity for the treatment of symptomatic uterine fibroids. Transcatheter embolization of the uterine artery leads to occlusion of the fibroid, ischemic shrinkage of the fibroid, and shrinkage of residual myometrial tissue. Fibroids may migrate weeks to months after the procedure as the myometrium contracts and the treated fibroid degenerates, leading to delayed discharge, passage of necrotic fibroids, cramps, and heavy bleeding if the fibroid migrates to a submucosal location. Hysteroscopic removal is an obvious option.
Recently, De Iaco et al17 reported the development of a uterine fistula and discontinuity of the myometrium after hysteroscopic resection of an embolized migrated fibroid. They speculated this was due to the development of an avascular myometrium after UAE. The patient was asymptomatic, but routine diagnostic hysteroscopy revealed a 2-cm discontinuity of the uterine wall at the site of the previous resectoscopic myomectomy. The myometrium was white and less than the full thickness.
Ultrasound guidance improves outcomes
Coccia et al18 described the benefits of intraoperative ultrasound guidance during operative hysteroscopy in fibroid treatment and uterine septum removal. Prospective evaluation of 81 patients involved an experienced ultrasonographer who mapped the limits of treatment. Patients were compared to 45 historical controls who had been similarly treated with laparoscopic monitoring. Satisfactory outcomes included relief of menorrhagia, complete resection of fibroids (including full resection of intramural fibroids), and thorough metroplasty of uterine septum.
Ultrasound guidance made it possible to extend the resection beyond the limit conventionally defined by hysteroscopy; none of the patients in the ultrasound group required reintervention. Among controls, a second operation was necessary in 4 cases. Investigators concluded that a wider resection (10 to 15 mm distance from the external surface of the uterus) of fibroids was achieved using ultrasound guidance.
Dr. Bradley reports that she serves as a consultant to Karl Storz, ACMI, Olympus, and Gynecare, and as a lecturer for Novacept.
1. Gurtcheff SE, Sharp HT. Complications associated with global endometrial ablation: the utility of the MAUDE database. Obstet Gynecol. 2003;102:1278-1282.
2. Jansen FW, Vredevoogd CB, Ulzen K, et al. Complications of hysteroscopy: a prospective, multicenter study. Obstet Gynecol. 2000;96:266-270.
3. Propst AM, Liberman RF, Harlow BL, Ginsburg ES. Complications of hysteroscopic surgery: predicting patients at risk. Obstet Gynecol. 2000;96:517-520.
4. Preutthipan S, Herabutya Y. Vaginal misoprostol for cervical priming before operative hysteroscopy: a randomized controlled trial. Obstet Gynecol. 2000;96:890-894.
5. Thomas JA, Leyland N, Durand N, Windrim RD. The use of oral misoprostol as a cervical ripening agent in operative hysteroscopy: a double-blind, placebo-controlled trial. Am J Obstet Gynecol. 2002;186:876-879.
6. Stotz M, Lampart A, Kochli OR, Schneider M. Intraabdominal bleeding masked by hemodilution after hysteroscopy. Anesthesiology. 2000;93:569-570.
7. Cooper JM, Brady RM. Intraoperative and early postoperative complications of operative hysteroscopy. Obstet Gynecol Clin North Am. 2000;27:347-366.
8. Loffer FD, Bradley LD, Brill AL, Brooks PG, Cooper JM. Hysteroscopic fluid monitoring guidelines. J Am Assoc Gynecol Laparosc. 2000;7:167-168.
9. Bradley LD, Widrich T. Flexible hysteroscopy: a state-of-the-art procedure for gynecologic evaluation. J Am Assoc Gynecol Laparosc. 1995;2:263-267.
10. Neis KJ, Brandner P, Lindemann HJ. Room air as a cause of gas embolism in diagnostic CO2hysteroscopy. Zentralbl Gynakol. 2000;122:222-225.
11. Bradner P, Neis KJ, Ehmer C. The etiology, frequency, and prevention of gas embolism during CO2hysteroscopy. J Am Assoc Gynecol Laparosc. 1999;6:421-428.
12. Munro MG, Weisberg M, Rubinstein E. Gas and air embolization during hysteroscopic electrosurgical vaporization: comparison of gas generation using bipolar and monopolar electrodes in an experimental model. J Am Assoc Gynecol Laparosc. 2001;8:488-494.
13. Murdoch JAC, Gan TJ. Anesthesia for hysteroscopy. Anesthesiol Clin North Am. 2001;1:125-140.
14. Cooper JM, Brady RM. Late complications of operative hysteroscopy. Obstet Gynecol Clin North Am. 2000;27:367-374.
15. Rogerson L, Gannon B, O’Donovan P. Outcome of pregnancy following endometrial ablation. J Gynecol Surg. 1997;13:155-160.
16. Kerimis P, Zolti M, Sinwany G, Mashiach S, Carp H. Uterine rupture after hysteroscopic resection of uterine septum. Fertil Steril. 2002;77:618-620.
17. De Iaco P, Golfieri R, Ghi T, Muzzupapa G, Ceccarini M, Bovicelli L. Uterine fistula induced by hysteroscopic resection of an embolized migrated fibroid: a rare complication after embolization of uterine fibroids. Fertil Steril. 2001;75:818-820.
18. Coccia ME, Becattini C, Bracco GL, et al. Intraoperative ultrasound guidance for operative hysteroscopy. J Reprod Med. 2000;45:413-418.
19. Brooks-Carter GN, Killackey MA, Neuwirth RS. Adenocarcinoma of the endometrium after endometrial ablation. Obstet Gynecol. 2000;96:836-837.
20. Cooper JM. Swimming lessons: check the water before jumping in. J Am Assoc Gynecol Laparosc. 1998;5:87-90.
21. Kudela M, Pilka R. Is there a real risk in patients with endometrial carcinoma undergoing diagnostic hysteroscopy (HSC)? Eur J Gynecol Oncol. 2001;22:342-344.
- Preoperative treatment with a gonadotropinreleasing hormone agonist increases the odds of operative complications by a factor of 4 to 7.
- Preoperative cervical ripening reduced the need for cervical dilation, minimized cervical complications, and reduced operative time.
- CO2 should never be used for operative hysteroscopic procedures because of the high risk of CO2 embolism.
- Ultrasound guidance may improve outcomes in selected hysteroscopic procedures.
It is a valuable tool in the evaluation and treatment of infertility, recurrent pregnancy loss, and abnormal and postmenopausal uterine bleeding, and is useful when saline infusion sonography findings are equivocal.
Further, if a global ablation device fails, the surgeon can convert to hysteroscopic ablation rather than abandon the procedure altogether. This is not as unusual as it might appear: In US Food and Drug Administration trials, there was a staggering 10% to 22% malfunction of global ablation technology.1
Safe, easily learned
Although gynecologists are beginning to embrace this modality, many physicians avoid it because of inadequate training or exaggerated fears of complications. In reality, hysteroscopy is one of the safest and most easily acquired surgical skills in gynecology. For example, in a prospective evaluation of 13,600 diagnostic and operative hysteroscopic procedures performed at 63 hospitals in the Netherlands—which involved both established surgeons and residents—Jansen et al2 found an astonishingly low complication rate of 0.28%, with no deaths.
Proper selection and treatment of patients and prompt intervention minimize complications as well as legal risks. Surgical misadventures and lawsuits occur with delayed intervention, failure to recognize pathology or risky conditions, and inadequate preventive maneuvers.
Overall, emphasis on safety is vital to success, and thorough awareness of potential complications is just as important.
Three types of complications
Complications fall into 3 categories (TABLE):
- Procedure-related
- Media-related
- Postoperative
PROCEDURE-RELATED COMPLICATIONS
Complication rates
In a retrospective investigation, Propst et al3 determined the rate of complications associated with specific hysteroscopic procedures. Demographic data and medical histories were collected for 925 women who had operative hysteroscopy in 1995 and 1996. The overall complication rate was 2.7%. Myomectomy and resection of uterine septa carried the greatest odds of complications; polypectomy and endometrial ablation had the lowest. Preoperative treatment with a gonadotropinreleasing hormone (GnRH) agonist increased the odds of complications by a factor of 4 to 7. Women under age 50 were more likely to experience complications than those over 50.
In the study by Jansen et al,2 38 complica-tions occurred in the 13,600 procedures. The greatest risk of complications occurred with adhesiolysis (4.48%), followed by endometrial resection (0.81%), myomectomy (0.75%), and polypectomy (0.38%).
Cervical entry requires special attention
Almost half of the complications in the Jansen study were related to cervical entry, so caution and, perhaps, preoperative cervical ripening are advised. Many premenopausal subjects were given GnRH analogues, which may render the cervix more resistant to dilation. Complications associated with a stenotic cervix include a cervical tear, creation of a false cervical passage, and uterine perforation.
Cervical ripening may help prevent uterine perforation. The most common complication, occurring in 14.2 cases per thousand, is uterine perforation.2 The risk of this is highest in postpartum procedures, followed by procedures in postmenopausal, then perimenopausal, women. Patients with endometrial cancer also have a higher rate of perforation.
Risk factors for uterine perforation include:
- nulliparity
- menopause
- use of GnRH agonists
- prior cone biopsy
- markedly retroverted uterus
- undue force
Vaginal or oral misoprostol for cervical ripening prior to operative hysteroscopy was evaluated in a randomized trial.4 Researchers found a reduced need for cervical dilation, a minimum of cervical complications, and reduced operative time in study patients compared with controls.
When 400 μg oral misoprostol is given 12 and 24 hours before surgery, it also softens the cervix and eases dilation.5 Although misoprostol has several bothersome side effects (such as lower abdominal pain and slight vaginal bleeding), few if any prevent its use.
Signs of perforation. Patients who sustain uterine perforation with subsequent intraperitoneal bleeding often complain of pain in the abdomen and shoulder, and experience hemodynamic instability. A quick sonographic survey of the abdomen will demonstrate free intraperitoneal fluid. (It is rare for much intraperitoneal fluid to accumulate by transtubal regurgitation during operative hysteroscopy, despite the quantity of fluid used.)
If perforation is suspected, laparoscopy or laparotomy is necessary to clarify the cause of pain, unstable vital signs, or free fluid visualized by ultrasound.6
Exercise extra care and precautions in women who have had a prior cesarean section, myomectomy, or uterine perforation. Complete visualization of uterine landmarks is necessary during operative hysteroscopy to exclude uterine dehiscence, sacculation, and perforation. Prior uterine surgery may cause myometrial weakness and lead to possible perforation. Do not proceed if abnormal uterine morphology is detected. If uterine perforation occurs, injury to bladder and bowel is possible when electrical energy is applied to a uterine wall compromised by prior surgery. Strict visualization of uterine anatomy is critical in this population so that bowel or bladder burns can be avoided.
MEDIA-RELATED COMPLICATIONS
Notorious complications and several recent lawsuits have stemmed from fluid overload. A common element has been the physician’s lack of awareness of how rapidly complications can arise, and what signs and symptoms are specific to the fluid used.
Monitor fluids vigilantly
Operative hysteroscopy must be performed in a fluid medium. The type of fluid depends on the surgeon’s preference and the instrument utilized, but any fluid can be associated with complications. Fluid choices with monopolar instruments include glycine 1.5%, a mixture of sorbitol 3% and mannitol 0.54%, and mannitol 5%. These are frequently used with the continuous-flow resectoscope. Bipolar operative hysteroscopy can be performed using saline.
The solution to media-related complications is basic: vigilant monitoring of fluids. A cavalier attitude, poor fluid documentation, and failure to respond to complications can lead to trouble. If fluid overload occurs, comanagement and consultation with an intensive care specialist is advised.
Distention media
Among the options for distention media in operative and diagnostic hysteroscopy are high-viscosity dextran 70 and low-viscosity fluids such as hypotonic, electrolyte-free and isotonic, electrolyte-containing solutions. The popularity of dextran 70 is waning, however. While it is immiscible with blood, significant complications have been reported.
Signs of anaphylactic reactions to dextran 70 include acute hypotension, hypoxia, pulmonary edema, fluid overload, fulminant coagulopathies, and anemia. The surgeon must operate quickly, minimize endometrial trauma, use continuous pulse oximetry, and obtain a preoperative coagulation panel.
Dextran 70 also can ruin operative hysteroscopes if they are not cleaned promptly and thoroughly after use.
Hypotonic, electrolyte-free solutions. With hypotonic, electrolyte-free solutions such as glycine 1.5%, early recognition of possible complications, including hyponatremic hypervolemia, is vital. For example, when glycine and sorbitol are metabolized, free water accumulates and the body attempts to achieve homeostasis through compensatory mechanisms such as osmosis, which moves free water into extracellular and intracellular spaces. This can lead to increased free water in the brain, resulting in cerebral edema, rising intracranial pressure, and cellular necrosis.
The cerebral cation pump normally pumps osmotically active cations into the extracellular space, thereby minimizing cerebral edema. However, this pump is inhibited by estrogen, so the compensatory mechanism is diminished.
Classic clinical features of hyponatremic hypervolemia include apprehension, confusion, fatigue, headache, mental agitation, nausea, visual disturbances (including blindness), vomiting, and weakness. These complications are more readily apparent when regional anesthesia is used rather than general anesthesia.
If hyponatremic hypervolemia goes unrecognized, bradycardia and hypertension can ensue, followed rapidly by cerebral and pulmonary edema and cardiovascular collapse. In addition, glycine 1.5% is metabolized to glycolic acid and ammonia. Free ammonia is associated with central nervous system disorders. Recognition and prompt treatment by an intensivist may prevent permanent neurologic sequelae, death, and lawsuits.7
Isotonic, electrolyte-containing solutions. Mannitol 5% is electrolyte poor but isotonic, creating less risk for hypo-osmolality. However, dilutional hyponatremia (ie, low sodium levels) can still occur.
Advantages of bipolar instruments. To minimize complications from hypotonic, electrolyte-free solutions, manufacturers developed operative hysteroscopes that can function in a bipolar environment. Bipolar instruments can operate in isotonic, physiologic, electrolyte-containing media. Hyponatremia and hypo-osmolality cannot occur with normal saline or Ringer’s lactate, but fluid overload can. (Fluid overload with saline can cause pulmonary edema and congestive heart failure.)
How much fluid will be absorbed? The answer depends on factors including surface area of the surgical field, duration of surgery, opened venous channels, type of irrigation fluid used, and pressure of the delivery system. Modern gynecologic suites employ fluid irrigation systems that continuously measure input and output, with alarms that signal a predetermined fluid deficit. The alarm indicates the need to halt the procedure and quickly evaluate the patient. Careful attention to the recommendations of Loffer et al8 would lead to fewer complications from fluid mismanagement.
Appropriate use of CO2
High risk of embolism with CO2 in operative procedures. Although diagnostic hysteroscopic procedures often are performed with carbon dioxide (CO2), operative procedures never should be. The reason: the high risk of CO2 embolism that occurs with open venous channels and vascular endometrium. The choice between CO2 and fluid medium for diagnostic hysteroscopy often is determined by physician preference and the presence of uterine bleeding. Many gynecologists prefer CO2 for its optical clarity and patient comfort during insufflation.9
Purge tubing of room air before each procedure. Embolic complications with CO2 have been recorded with use of the neodymium: yttrium aluminum garnet (Nd:YAG) laser and during operative procedures. Less well known are the adverse sequelae that can occur with room air prior to beginning the procedure. It is critical to purge the entire tubal system with CO2 prior to instrumentation, since up to 40 cm3 of room air may be insufflated into a patient when 200 cm of connective tubing with a 0.5-cm lumen is used.10 Wait for several minutes before starting the procedure so that the whole system is purged.
One of the greatest concerns about endometrial ablation is that diagnosis of endometrial cancer will be delayed because the endometrial cavity has been obliterated. Vilos19 recently reviewed the salient characteristics and findings in women treated by endometrial ablation who subsequently developed endometrial cancer. A review of the individual cases revealed that most of these patients had numerous risk factors for endometrial cancer.
Review risk factors, chronic conditions
Many patients with abnormal bleeding also have risk factors for endometrial cancer, as well as medical conditions that increase the likelihood of morbidity with surgery, such as obesity, hypertension, diabetes, and advanced age. In these cases, hysterectomy may be a better option than endometrial ablation. It would be far better to have such high-risk patients cleared for hysterectomy than to chance their becoming an endometrial-ablation “statistic.” If endometrial ablation is performed in these cases, we prevent the egress of blood, foster development of synechiae, render endometrial biopsy difficult or impossible and, potentially, “bury” endometrial cells deeper within the myometrium—all of which contribute to a delayed and “upstaged” diagnosis of endometrial cancer.
Patients at risk of endometrial cancer should undergo a scrupulous and unambiguous work-up and evaluation. Indeterminate endometrial echo and office evaluation that generates biopsy samples designated as “insufficient for diagnosis,” “no endometrial tissue seen,” or “atrophy” should raise suspicion. These patients require full visualization of the endometrium.
Heightened risk during perimenopause
Newer ablation techniques that utilize global therapy make it paramount that perimenopausal women undergo scrupulous evaluation. Until much more information is available, endometrial ablation should be avoided in patients with endometrial hyperplasia, particularly with atypia. While some gynecologists may be persuaded to consider endometrial ablation as a minimally invasive procedure compared to hysterectomy, the risk of delayed diagnosis of endometrial cancer is of paramount concern. The treatment of choice for these patients remains medical therapy with oral progesterone and, possibly, longterm use of a levonorgestrel-releasing intrauterine system. If this fails, hysterectomy is advisable. As Cooper20 aptly states, “Conservative, nonextirpative procedures offer no life raft” compared with hysterectomy, which covers many missed diagnoses.
No risk of spreading cancer cells
Some gynecologists have worried about the risk of disseminating endometrial cancer cells during hysteroscopy. However, Kudela and Pilka21 studied the true risk in women undergoing blind dilation and curettage and hysteroscopy performed with a fluid medium. Cul-de-sac aspiration prior to instrumentation and at the conclusion of the procedure demonstrated no increased risk of positive cytology. They are continuing a Phase II trial comparing outcomes of both groups over 5 years.
6 most common symptoms of venous or air emboli. Anesthesiologists and gynecologists must be vigilant to prevent venous or air emboli. Munro et al12 succinctly outline the 6 most common symptoms:
- pulmonary hypertension
- hypercarbia
- hypoxia
- arrhythmias
- tachypnea
- systemic hypotension
Beware of a drop in end-tidal CO2. The most common sign of impending cardiovascular collapse is a sudden decrease in end-tidal CO2, when the right cardiac outflow tract is obstructed by CO2, which leads to arterial oxygen (O2) desaturation. If such a decrease is suspected, stop the procedure immediately and administer 100% O2. (Also stop nitrous oxide, if used.) Turn the patient to the left lateral decubitis position and use a central venous catheter to aspirate gas, if necessary. Cardiac massage and a precordial thump may dislodge CO2; unfortunately, high false-positive rates of pre-cordial Doppler make its use impractical.
How to minimize risks
- Avoid coaxial gas cooling tips associated with Nd:YAG crystal lasers
- Avoid a steep Trendelenburg position
- Keep cervix covered with sponge or dilator when operative hysteroscope is removed to minimize air embolism
- Deaerate the equipment prior to surgery Use a low-pressure hysteroscopic CO2 insufflator
- Carefully monitor the patient
- Be highly suspicious when vital signs are unstable
POSTOPERATIVE COMPLICATIONS
Some complications of hysteroscopy may not become clinically evident for months or even years. The most common complications of hysteroscopic endometrial ablation include pregnancy, postablation tubal sterilization syndrome, new or worsening dysmenorrhea, hematometra, endometrial cancer, and failure to completely treat symptoms.
Patients scheduled for hysteroscopy must be informed of potential delayed risks of the procedure. In addition, all reproductive-aged women should be advised that pregnancy is possible after endometrial ablation or operative removal of an intracavitary mass; thus, contraception is crucial. The endometrial tissue is resilient and may regenerate after ablation.
Hematometra: Avoid cervical canal
Hematometra is an infrequent late complication of operative hysteroscopy. If menstruating women or those taking hormone replacement therapy experience cyclic or chronic lower pelvic pain after surgery, scarring or narrowing of the endometrial cavity may be the cause. Approximately 1% to 2% of women who undergo operative hysteroscopy experience this phenomenon. Most cases can be treated with cervical dilation alone.
Since the cervical canal contains no endometrial glands, there is no need to treat this area in women undergoing endometrial ablation. In fact, avoiding this area during treatment is a critical component of successful surgery.
Tubal sterilization syndrome possible after endometrial ablation
Consider this syndrome when a patient undergoing endometrial ablation complains of crampy, cyclic, unilateral or bilateral pelvic pain, possibly accompanied by vaginal spotting. Sometimes a unilateral mass can be palpated, but more commonly tenderness is elicited on pelvic examination.
Ultrasound may demonstrate fluid near the cornual region. Laparoscopy confirms the diagnosis by visualizing a swollen, edematous proximal fallopian tube. Salpingectomy may confirm hematosalpinx, chronic or acute inflammation, or hemosiderin deposits.
Treatment includes bilateral cornual resection and reablation of proximal endometrium, or hysterectomy.14
Pregnancy complications
Endometrial ablation is not to be regarded as a method of contraception. Patients needing birth control should consider concurrent tubal ligation or other reliable methods after this procedure.
The frequency of pregnancy after endometrial ablation ranges from 0.2% to 1.6%, though this data may represent underreporting. Pregnancy outcomes have been dismal in women conceiving after endometrial ablation. Complications include preterm labor, premature delivery, intrauterine growth retardation, prenatal death, postpartum hemorrhage, and placentation problems such as placenta accreta, increta, or percreta, as well as placental abruption.15
Uterine dehiscence and sacculation and extremely thin myometrium have been reported after uterine adhesiolysis, uterine perforation during operative hysteroscopy, and with myoma resections. A high index of suspicion is vital when a gravida presents with pelvic pain, decreased fetal movement, vaginal bleeding, or abnormal uterine masses detected ultrasonographically.
Signs of uterine rupture. Pregnancyrelated complications of operative hysteroscopy can be dramatic and fatal if not recognized quickly, as in the case of uterine rupture. Kerimis et al16 describe uterine rupture in a term pregnancy after hysteroscopic resection of a uterine septum. Severe fetal distress, maternal shoulder pain, and abdominal pain led to an emergency cesarean section. Intraoperative findings included a 7-cm tear from left cornua to right cornua. The original metroplasty, performed with cutting diathermy and laparoscopy, was not accompanied by complications or perforation.
Patients who experience intraoperative complications during metroplasty or deep resection of intramural fibroids should be informed of the risk of uterine rupture so they may consider elective cesarean. Regardless of the mode of delivery, prompt attention is vital if fetal distress is suspected.
Postablation warning signs
Patients undergoing endometrial ablation generally have a quick postoperative return to activity, minimal need for postoperative pain medication, and limited complaints. Beware of patients who make frequent postoperative phone calls and have escalating requirements for pain medication. While bowel and bladder injuries are infrequent—as is postoperative endometritis—these must be vigilantly considered and evaluated when patients complain of persistent pain, fever, and general malaise. Office evaluation is necessary, including thorough abdominal and pelvic examinations. Laboratory testing should include electrolytes, complete blood count, sedimentation rate, ultrasound, and a flat plate of the abdomen (kidneys, ureter, and bladder; upright) may be required. Sometimes a computed tomography scan of the pelvis/abdomen may be needed if perforation with bowel or bladder injury is suspected.
Hysteroscopic fibroid removal may be necessary after UAE
Uterine artery embolization (UAE) is gaining popularity for the treatment of symptomatic uterine fibroids. Transcatheter embolization of the uterine artery leads to occlusion of the fibroid, ischemic shrinkage of the fibroid, and shrinkage of residual myometrial tissue. Fibroids may migrate weeks to months after the procedure as the myometrium contracts and the treated fibroid degenerates, leading to delayed discharge, passage of necrotic fibroids, cramps, and heavy bleeding if the fibroid migrates to a submucosal location. Hysteroscopic removal is an obvious option.
Recently, De Iaco et al17 reported the development of a uterine fistula and discontinuity of the myometrium after hysteroscopic resection of an embolized migrated fibroid. They speculated this was due to the development of an avascular myometrium after UAE. The patient was asymptomatic, but routine diagnostic hysteroscopy revealed a 2-cm discontinuity of the uterine wall at the site of the previous resectoscopic myomectomy. The myometrium was white and less than the full thickness.
Ultrasound guidance improves outcomes
Coccia et al18 described the benefits of intraoperative ultrasound guidance during operative hysteroscopy in fibroid treatment and uterine septum removal. Prospective evaluation of 81 patients involved an experienced ultrasonographer who mapped the limits of treatment. Patients were compared to 45 historical controls who had been similarly treated with laparoscopic monitoring. Satisfactory outcomes included relief of menorrhagia, complete resection of fibroids (including full resection of intramural fibroids), and thorough metroplasty of uterine septum.
Ultrasound guidance made it possible to extend the resection beyond the limit conventionally defined by hysteroscopy; none of the patients in the ultrasound group required reintervention. Among controls, a second operation was necessary in 4 cases. Investigators concluded that a wider resection (10 to 15 mm distance from the external surface of the uterus) of fibroids was achieved using ultrasound guidance.
Dr. Bradley reports that she serves as a consultant to Karl Storz, ACMI, Olympus, and Gynecare, and as a lecturer for Novacept.
- Preoperative treatment with a gonadotropinreleasing hormone agonist increases the odds of operative complications by a factor of 4 to 7.
- Preoperative cervical ripening reduced the need for cervical dilation, minimized cervical complications, and reduced operative time.
- CO2 should never be used for operative hysteroscopic procedures because of the high risk of CO2 embolism.
- Ultrasound guidance may improve outcomes in selected hysteroscopic procedures.
It is a valuable tool in the evaluation and treatment of infertility, recurrent pregnancy loss, and abnormal and postmenopausal uterine bleeding, and is useful when saline infusion sonography findings are equivocal.
Further, if a global ablation device fails, the surgeon can convert to hysteroscopic ablation rather than abandon the procedure altogether. This is not as unusual as it might appear: In US Food and Drug Administration trials, there was a staggering 10% to 22% malfunction of global ablation technology.1
Safe, easily learned
Although gynecologists are beginning to embrace this modality, many physicians avoid it because of inadequate training or exaggerated fears of complications. In reality, hysteroscopy is one of the safest and most easily acquired surgical skills in gynecology. For example, in a prospective evaluation of 13,600 diagnostic and operative hysteroscopic procedures performed at 63 hospitals in the Netherlands—which involved both established surgeons and residents—Jansen et al2 found an astonishingly low complication rate of 0.28%, with no deaths.
Proper selection and treatment of patients and prompt intervention minimize complications as well as legal risks. Surgical misadventures and lawsuits occur with delayed intervention, failure to recognize pathology or risky conditions, and inadequate preventive maneuvers.
Overall, emphasis on safety is vital to success, and thorough awareness of potential complications is just as important.
Three types of complications
Complications fall into 3 categories (TABLE):
- Procedure-related
- Media-related
- Postoperative
PROCEDURE-RELATED COMPLICATIONS
Complication rates
In a retrospective investigation, Propst et al3 determined the rate of complications associated with specific hysteroscopic procedures. Demographic data and medical histories were collected for 925 women who had operative hysteroscopy in 1995 and 1996. The overall complication rate was 2.7%. Myomectomy and resection of uterine septa carried the greatest odds of complications; polypectomy and endometrial ablation had the lowest. Preoperative treatment with a gonadotropinreleasing hormone (GnRH) agonist increased the odds of complications by a factor of 4 to 7. Women under age 50 were more likely to experience complications than those over 50.
In the study by Jansen et al,2 38 complica-tions occurred in the 13,600 procedures. The greatest risk of complications occurred with adhesiolysis (4.48%), followed by endometrial resection (0.81%), myomectomy (0.75%), and polypectomy (0.38%).
Cervical entry requires special attention
Almost half of the complications in the Jansen study were related to cervical entry, so caution and, perhaps, preoperative cervical ripening are advised. Many premenopausal subjects were given GnRH analogues, which may render the cervix more resistant to dilation. Complications associated with a stenotic cervix include a cervical tear, creation of a false cervical passage, and uterine perforation.
Cervical ripening may help prevent uterine perforation. The most common complication, occurring in 14.2 cases per thousand, is uterine perforation.2 The risk of this is highest in postpartum procedures, followed by procedures in postmenopausal, then perimenopausal, women. Patients with endometrial cancer also have a higher rate of perforation.
Risk factors for uterine perforation include:
- nulliparity
- menopause
- use of GnRH agonists
- prior cone biopsy
- markedly retroverted uterus
- undue force
Vaginal or oral misoprostol for cervical ripening prior to operative hysteroscopy was evaluated in a randomized trial.4 Researchers found a reduced need for cervical dilation, a minimum of cervical complications, and reduced operative time in study patients compared with controls.
When 400 μg oral misoprostol is given 12 and 24 hours before surgery, it also softens the cervix and eases dilation.5 Although misoprostol has several bothersome side effects (such as lower abdominal pain and slight vaginal bleeding), few if any prevent its use.
Signs of perforation. Patients who sustain uterine perforation with subsequent intraperitoneal bleeding often complain of pain in the abdomen and shoulder, and experience hemodynamic instability. A quick sonographic survey of the abdomen will demonstrate free intraperitoneal fluid. (It is rare for much intraperitoneal fluid to accumulate by transtubal regurgitation during operative hysteroscopy, despite the quantity of fluid used.)
If perforation is suspected, laparoscopy or laparotomy is necessary to clarify the cause of pain, unstable vital signs, or free fluid visualized by ultrasound.6
Exercise extra care and precautions in women who have had a prior cesarean section, myomectomy, or uterine perforation. Complete visualization of uterine landmarks is necessary during operative hysteroscopy to exclude uterine dehiscence, sacculation, and perforation. Prior uterine surgery may cause myometrial weakness and lead to possible perforation. Do not proceed if abnormal uterine morphology is detected. If uterine perforation occurs, injury to bladder and bowel is possible when electrical energy is applied to a uterine wall compromised by prior surgery. Strict visualization of uterine anatomy is critical in this population so that bowel or bladder burns can be avoided.
MEDIA-RELATED COMPLICATIONS
Notorious complications and several recent lawsuits have stemmed from fluid overload. A common element has been the physician’s lack of awareness of how rapidly complications can arise, and what signs and symptoms are specific to the fluid used.
Monitor fluids vigilantly
Operative hysteroscopy must be performed in a fluid medium. The type of fluid depends on the surgeon’s preference and the instrument utilized, but any fluid can be associated with complications. Fluid choices with monopolar instruments include glycine 1.5%, a mixture of sorbitol 3% and mannitol 0.54%, and mannitol 5%. These are frequently used with the continuous-flow resectoscope. Bipolar operative hysteroscopy can be performed using saline.
The solution to media-related complications is basic: vigilant monitoring of fluids. A cavalier attitude, poor fluid documentation, and failure to respond to complications can lead to trouble. If fluid overload occurs, comanagement and consultation with an intensive care specialist is advised.
Distention media
Among the options for distention media in operative and diagnostic hysteroscopy are high-viscosity dextran 70 and low-viscosity fluids such as hypotonic, electrolyte-free and isotonic, electrolyte-containing solutions. The popularity of dextran 70 is waning, however. While it is immiscible with blood, significant complications have been reported.
Signs of anaphylactic reactions to dextran 70 include acute hypotension, hypoxia, pulmonary edema, fluid overload, fulminant coagulopathies, and anemia. The surgeon must operate quickly, minimize endometrial trauma, use continuous pulse oximetry, and obtain a preoperative coagulation panel.
Dextran 70 also can ruin operative hysteroscopes if they are not cleaned promptly and thoroughly after use.
Hypotonic, electrolyte-free solutions. With hypotonic, electrolyte-free solutions such as glycine 1.5%, early recognition of possible complications, including hyponatremic hypervolemia, is vital. For example, when glycine and sorbitol are metabolized, free water accumulates and the body attempts to achieve homeostasis through compensatory mechanisms such as osmosis, which moves free water into extracellular and intracellular spaces. This can lead to increased free water in the brain, resulting in cerebral edema, rising intracranial pressure, and cellular necrosis.
The cerebral cation pump normally pumps osmotically active cations into the extracellular space, thereby minimizing cerebral edema. However, this pump is inhibited by estrogen, so the compensatory mechanism is diminished.
Classic clinical features of hyponatremic hypervolemia include apprehension, confusion, fatigue, headache, mental agitation, nausea, visual disturbances (including blindness), vomiting, and weakness. These complications are more readily apparent when regional anesthesia is used rather than general anesthesia.
If hyponatremic hypervolemia goes unrecognized, bradycardia and hypertension can ensue, followed rapidly by cerebral and pulmonary edema and cardiovascular collapse. In addition, glycine 1.5% is metabolized to glycolic acid and ammonia. Free ammonia is associated with central nervous system disorders. Recognition and prompt treatment by an intensivist may prevent permanent neurologic sequelae, death, and lawsuits.7
Isotonic, electrolyte-containing solutions. Mannitol 5% is electrolyte poor but isotonic, creating less risk for hypo-osmolality. However, dilutional hyponatremia (ie, low sodium levels) can still occur.
Advantages of bipolar instruments. To minimize complications from hypotonic, electrolyte-free solutions, manufacturers developed operative hysteroscopes that can function in a bipolar environment. Bipolar instruments can operate in isotonic, physiologic, electrolyte-containing media. Hyponatremia and hypo-osmolality cannot occur with normal saline or Ringer’s lactate, but fluid overload can. (Fluid overload with saline can cause pulmonary edema and congestive heart failure.)
How much fluid will be absorbed? The answer depends on factors including surface area of the surgical field, duration of surgery, opened venous channels, type of irrigation fluid used, and pressure of the delivery system. Modern gynecologic suites employ fluid irrigation systems that continuously measure input and output, with alarms that signal a predetermined fluid deficit. The alarm indicates the need to halt the procedure and quickly evaluate the patient. Careful attention to the recommendations of Loffer et al8 would lead to fewer complications from fluid mismanagement.
Appropriate use of CO2
High risk of embolism with CO2 in operative procedures. Although diagnostic hysteroscopic procedures often are performed with carbon dioxide (CO2), operative procedures never should be. The reason: the high risk of CO2 embolism that occurs with open venous channels and vascular endometrium. The choice between CO2 and fluid medium for diagnostic hysteroscopy often is determined by physician preference and the presence of uterine bleeding. Many gynecologists prefer CO2 for its optical clarity and patient comfort during insufflation.9
Purge tubing of room air before each procedure. Embolic complications with CO2 have been recorded with use of the neodymium: yttrium aluminum garnet (Nd:YAG) laser and during operative procedures. Less well known are the adverse sequelae that can occur with room air prior to beginning the procedure. It is critical to purge the entire tubal system with CO2 prior to instrumentation, since up to 40 cm3 of room air may be insufflated into a patient when 200 cm of connective tubing with a 0.5-cm lumen is used.10 Wait for several minutes before starting the procedure so that the whole system is purged.
One of the greatest concerns about endometrial ablation is that diagnosis of endometrial cancer will be delayed because the endometrial cavity has been obliterated. Vilos19 recently reviewed the salient characteristics and findings in women treated by endometrial ablation who subsequently developed endometrial cancer. A review of the individual cases revealed that most of these patients had numerous risk factors for endometrial cancer.
Review risk factors, chronic conditions
Many patients with abnormal bleeding also have risk factors for endometrial cancer, as well as medical conditions that increase the likelihood of morbidity with surgery, such as obesity, hypertension, diabetes, and advanced age. In these cases, hysterectomy may be a better option than endometrial ablation. It would be far better to have such high-risk patients cleared for hysterectomy than to chance their becoming an endometrial-ablation “statistic.” If endometrial ablation is performed in these cases, we prevent the egress of blood, foster development of synechiae, render endometrial biopsy difficult or impossible and, potentially, “bury” endometrial cells deeper within the myometrium—all of which contribute to a delayed and “upstaged” diagnosis of endometrial cancer.
Patients at risk of endometrial cancer should undergo a scrupulous and unambiguous work-up and evaluation. Indeterminate endometrial echo and office evaluation that generates biopsy samples designated as “insufficient for diagnosis,” “no endometrial tissue seen,” or “atrophy” should raise suspicion. These patients require full visualization of the endometrium.
Heightened risk during perimenopause
Newer ablation techniques that utilize global therapy make it paramount that perimenopausal women undergo scrupulous evaluation. Until much more information is available, endometrial ablation should be avoided in patients with endometrial hyperplasia, particularly with atypia. While some gynecologists may be persuaded to consider endometrial ablation as a minimally invasive procedure compared to hysterectomy, the risk of delayed diagnosis of endometrial cancer is of paramount concern. The treatment of choice for these patients remains medical therapy with oral progesterone and, possibly, longterm use of a levonorgestrel-releasing intrauterine system. If this fails, hysterectomy is advisable. As Cooper20 aptly states, “Conservative, nonextirpative procedures offer no life raft” compared with hysterectomy, which covers many missed diagnoses.
No risk of spreading cancer cells
Some gynecologists have worried about the risk of disseminating endometrial cancer cells during hysteroscopy. However, Kudela and Pilka21 studied the true risk in women undergoing blind dilation and curettage and hysteroscopy performed with a fluid medium. Cul-de-sac aspiration prior to instrumentation and at the conclusion of the procedure demonstrated no increased risk of positive cytology. They are continuing a Phase II trial comparing outcomes of both groups over 5 years.
6 most common symptoms of venous or air emboli. Anesthesiologists and gynecologists must be vigilant to prevent venous or air emboli. Munro et al12 succinctly outline the 6 most common symptoms:
- pulmonary hypertension
- hypercarbia
- hypoxia
- arrhythmias
- tachypnea
- systemic hypotension
Beware of a drop in end-tidal CO2. The most common sign of impending cardiovascular collapse is a sudden decrease in end-tidal CO2, when the right cardiac outflow tract is obstructed by CO2, which leads to arterial oxygen (O2) desaturation. If such a decrease is suspected, stop the procedure immediately and administer 100% O2. (Also stop nitrous oxide, if used.) Turn the patient to the left lateral decubitis position and use a central venous catheter to aspirate gas, if necessary. Cardiac massage and a precordial thump may dislodge CO2; unfortunately, high false-positive rates of pre-cordial Doppler make its use impractical.
How to minimize risks
- Avoid coaxial gas cooling tips associated with Nd:YAG crystal lasers
- Avoid a steep Trendelenburg position
- Keep cervix covered with sponge or dilator when operative hysteroscope is removed to minimize air embolism
- Deaerate the equipment prior to surgery Use a low-pressure hysteroscopic CO2 insufflator
- Carefully monitor the patient
- Be highly suspicious when vital signs are unstable
POSTOPERATIVE COMPLICATIONS
Some complications of hysteroscopy may not become clinically evident for months or even years. The most common complications of hysteroscopic endometrial ablation include pregnancy, postablation tubal sterilization syndrome, new or worsening dysmenorrhea, hematometra, endometrial cancer, and failure to completely treat symptoms.
Patients scheduled for hysteroscopy must be informed of potential delayed risks of the procedure. In addition, all reproductive-aged women should be advised that pregnancy is possible after endometrial ablation or operative removal of an intracavitary mass; thus, contraception is crucial. The endometrial tissue is resilient and may regenerate after ablation.
Hematometra: Avoid cervical canal
Hematometra is an infrequent late complication of operative hysteroscopy. If menstruating women or those taking hormone replacement therapy experience cyclic or chronic lower pelvic pain after surgery, scarring or narrowing of the endometrial cavity may be the cause. Approximately 1% to 2% of women who undergo operative hysteroscopy experience this phenomenon. Most cases can be treated with cervical dilation alone.
Since the cervical canal contains no endometrial glands, there is no need to treat this area in women undergoing endometrial ablation. In fact, avoiding this area during treatment is a critical component of successful surgery.
Tubal sterilization syndrome possible after endometrial ablation
Consider this syndrome when a patient undergoing endometrial ablation complains of crampy, cyclic, unilateral or bilateral pelvic pain, possibly accompanied by vaginal spotting. Sometimes a unilateral mass can be palpated, but more commonly tenderness is elicited on pelvic examination.
Ultrasound may demonstrate fluid near the cornual region. Laparoscopy confirms the diagnosis by visualizing a swollen, edematous proximal fallopian tube. Salpingectomy may confirm hematosalpinx, chronic or acute inflammation, or hemosiderin deposits.
Treatment includes bilateral cornual resection and reablation of proximal endometrium, or hysterectomy.14
Pregnancy complications
Endometrial ablation is not to be regarded as a method of contraception. Patients needing birth control should consider concurrent tubal ligation or other reliable methods after this procedure.
The frequency of pregnancy after endometrial ablation ranges from 0.2% to 1.6%, though this data may represent underreporting. Pregnancy outcomes have been dismal in women conceiving after endometrial ablation. Complications include preterm labor, premature delivery, intrauterine growth retardation, prenatal death, postpartum hemorrhage, and placentation problems such as placenta accreta, increta, or percreta, as well as placental abruption.15
Uterine dehiscence and sacculation and extremely thin myometrium have been reported after uterine adhesiolysis, uterine perforation during operative hysteroscopy, and with myoma resections. A high index of suspicion is vital when a gravida presents with pelvic pain, decreased fetal movement, vaginal bleeding, or abnormal uterine masses detected ultrasonographically.
Signs of uterine rupture. Pregnancyrelated complications of operative hysteroscopy can be dramatic and fatal if not recognized quickly, as in the case of uterine rupture. Kerimis et al16 describe uterine rupture in a term pregnancy after hysteroscopic resection of a uterine septum. Severe fetal distress, maternal shoulder pain, and abdominal pain led to an emergency cesarean section. Intraoperative findings included a 7-cm tear from left cornua to right cornua. The original metroplasty, performed with cutting diathermy and laparoscopy, was not accompanied by complications or perforation.
Patients who experience intraoperative complications during metroplasty or deep resection of intramural fibroids should be informed of the risk of uterine rupture so they may consider elective cesarean. Regardless of the mode of delivery, prompt attention is vital if fetal distress is suspected.
Postablation warning signs
Patients undergoing endometrial ablation generally have a quick postoperative return to activity, minimal need for postoperative pain medication, and limited complaints. Beware of patients who make frequent postoperative phone calls and have escalating requirements for pain medication. While bowel and bladder injuries are infrequent—as is postoperative endometritis—these must be vigilantly considered and evaluated when patients complain of persistent pain, fever, and general malaise. Office evaluation is necessary, including thorough abdominal and pelvic examinations. Laboratory testing should include electrolytes, complete blood count, sedimentation rate, ultrasound, and a flat plate of the abdomen (kidneys, ureter, and bladder; upright) may be required. Sometimes a computed tomography scan of the pelvis/abdomen may be needed if perforation with bowel or bladder injury is suspected.
Hysteroscopic fibroid removal may be necessary after UAE
Uterine artery embolization (UAE) is gaining popularity for the treatment of symptomatic uterine fibroids. Transcatheter embolization of the uterine artery leads to occlusion of the fibroid, ischemic shrinkage of the fibroid, and shrinkage of residual myometrial tissue. Fibroids may migrate weeks to months after the procedure as the myometrium contracts and the treated fibroid degenerates, leading to delayed discharge, passage of necrotic fibroids, cramps, and heavy bleeding if the fibroid migrates to a submucosal location. Hysteroscopic removal is an obvious option.
Recently, De Iaco et al17 reported the development of a uterine fistula and discontinuity of the myometrium after hysteroscopic resection of an embolized migrated fibroid. They speculated this was due to the development of an avascular myometrium after UAE. The patient was asymptomatic, but routine diagnostic hysteroscopy revealed a 2-cm discontinuity of the uterine wall at the site of the previous resectoscopic myomectomy. The myometrium was white and less than the full thickness.
Ultrasound guidance improves outcomes
Coccia et al18 described the benefits of intraoperative ultrasound guidance during operative hysteroscopy in fibroid treatment and uterine septum removal. Prospective evaluation of 81 patients involved an experienced ultrasonographer who mapped the limits of treatment. Patients were compared to 45 historical controls who had been similarly treated with laparoscopic monitoring. Satisfactory outcomes included relief of menorrhagia, complete resection of fibroids (including full resection of intramural fibroids), and thorough metroplasty of uterine septum.
Ultrasound guidance made it possible to extend the resection beyond the limit conventionally defined by hysteroscopy; none of the patients in the ultrasound group required reintervention. Among controls, a second operation was necessary in 4 cases. Investigators concluded that a wider resection (10 to 15 mm distance from the external surface of the uterus) of fibroids was achieved using ultrasound guidance.
Dr. Bradley reports that she serves as a consultant to Karl Storz, ACMI, Olympus, and Gynecare, and as a lecturer for Novacept.
1. Gurtcheff SE, Sharp HT. Complications associated with global endometrial ablation: the utility of the MAUDE database. Obstet Gynecol. 2003;102:1278-1282.
2. Jansen FW, Vredevoogd CB, Ulzen K, et al. Complications of hysteroscopy: a prospective, multicenter study. Obstet Gynecol. 2000;96:266-270.
3. Propst AM, Liberman RF, Harlow BL, Ginsburg ES. Complications of hysteroscopic surgery: predicting patients at risk. Obstet Gynecol. 2000;96:517-520.
4. Preutthipan S, Herabutya Y. Vaginal misoprostol for cervical priming before operative hysteroscopy: a randomized controlled trial. Obstet Gynecol. 2000;96:890-894.
5. Thomas JA, Leyland N, Durand N, Windrim RD. The use of oral misoprostol as a cervical ripening agent in operative hysteroscopy: a double-blind, placebo-controlled trial. Am J Obstet Gynecol. 2002;186:876-879.
6. Stotz M, Lampart A, Kochli OR, Schneider M. Intraabdominal bleeding masked by hemodilution after hysteroscopy. Anesthesiology. 2000;93:569-570.
7. Cooper JM, Brady RM. Intraoperative and early postoperative complications of operative hysteroscopy. Obstet Gynecol Clin North Am. 2000;27:347-366.
8. Loffer FD, Bradley LD, Brill AL, Brooks PG, Cooper JM. Hysteroscopic fluid monitoring guidelines. J Am Assoc Gynecol Laparosc. 2000;7:167-168.
9. Bradley LD, Widrich T. Flexible hysteroscopy: a state-of-the-art procedure for gynecologic evaluation. J Am Assoc Gynecol Laparosc. 1995;2:263-267.
10. Neis KJ, Brandner P, Lindemann HJ. Room air as a cause of gas embolism in diagnostic CO2hysteroscopy. Zentralbl Gynakol. 2000;122:222-225.
11. Bradner P, Neis KJ, Ehmer C. The etiology, frequency, and prevention of gas embolism during CO2hysteroscopy. J Am Assoc Gynecol Laparosc. 1999;6:421-428.
12. Munro MG, Weisberg M, Rubinstein E. Gas and air embolization during hysteroscopic electrosurgical vaporization: comparison of gas generation using bipolar and monopolar electrodes in an experimental model. J Am Assoc Gynecol Laparosc. 2001;8:488-494.
13. Murdoch JAC, Gan TJ. Anesthesia for hysteroscopy. Anesthesiol Clin North Am. 2001;1:125-140.
14. Cooper JM, Brady RM. Late complications of operative hysteroscopy. Obstet Gynecol Clin North Am. 2000;27:367-374.
15. Rogerson L, Gannon B, O’Donovan P. Outcome of pregnancy following endometrial ablation. J Gynecol Surg. 1997;13:155-160.
16. Kerimis P, Zolti M, Sinwany G, Mashiach S, Carp H. Uterine rupture after hysteroscopic resection of uterine septum. Fertil Steril. 2002;77:618-620.
17. De Iaco P, Golfieri R, Ghi T, Muzzupapa G, Ceccarini M, Bovicelli L. Uterine fistula induced by hysteroscopic resection of an embolized migrated fibroid: a rare complication after embolization of uterine fibroids. Fertil Steril. 2001;75:818-820.
18. Coccia ME, Becattini C, Bracco GL, et al. Intraoperative ultrasound guidance for operative hysteroscopy. J Reprod Med. 2000;45:413-418.
19. Brooks-Carter GN, Killackey MA, Neuwirth RS. Adenocarcinoma of the endometrium after endometrial ablation. Obstet Gynecol. 2000;96:836-837.
20. Cooper JM. Swimming lessons: check the water before jumping in. J Am Assoc Gynecol Laparosc. 1998;5:87-90.
21. Kudela M, Pilka R. Is there a real risk in patients with endometrial carcinoma undergoing diagnostic hysteroscopy (HSC)? Eur J Gynecol Oncol. 2001;22:342-344.
1. Gurtcheff SE, Sharp HT. Complications associated with global endometrial ablation: the utility of the MAUDE database. Obstet Gynecol. 2003;102:1278-1282.
2. Jansen FW, Vredevoogd CB, Ulzen K, et al. Complications of hysteroscopy: a prospective, multicenter study. Obstet Gynecol. 2000;96:266-270.
3. Propst AM, Liberman RF, Harlow BL, Ginsburg ES. Complications of hysteroscopic surgery: predicting patients at risk. Obstet Gynecol. 2000;96:517-520.
4. Preutthipan S, Herabutya Y. Vaginal misoprostol for cervical priming before operative hysteroscopy: a randomized controlled trial. Obstet Gynecol. 2000;96:890-894.
5. Thomas JA, Leyland N, Durand N, Windrim RD. The use of oral misoprostol as a cervical ripening agent in operative hysteroscopy: a double-blind, placebo-controlled trial. Am J Obstet Gynecol. 2002;186:876-879.
6. Stotz M, Lampart A, Kochli OR, Schneider M. Intraabdominal bleeding masked by hemodilution after hysteroscopy. Anesthesiology. 2000;93:569-570.
7. Cooper JM, Brady RM. Intraoperative and early postoperative complications of operative hysteroscopy. Obstet Gynecol Clin North Am. 2000;27:347-366.
8. Loffer FD, Bradley LD, Brill AL, Brooks PG, Cooper JM. Hysteroscopic fluid monitoring guidelines. J Am Assoc Gynecol Laparosc. 2000;7:167-168.
9. Bradley LD, Widrich T. Flexible hysteroscopy: a state-of-the-art procedure for gynecologic evaluation. J Am Assoc Gynecol Laparosc. 1995;2:263-267.
10. Neis KJ, Brandner P, Lindemann HJ. Room air as a cause of gas embolism in diagnostic CO2hysteroscopy. Zentralbl Gynakol. 2000;122:222-225.
11. Bradner P, Neis KJ, Ehmer C. The etiology, frequency, and prevention of gas embolism during CO2hysteroscopy. J Am Assoc Gynecol Laparosc. 1999;6:421-428.
12. Munro MG, Weisberg M, Rubinstein E. Gas and air embolization during hysteroscopic electrosurgical vaporization: comparison of gas generation using bipolar and monopolar electrodes in an experimental model. J Am Assoc Gynecol Laparosc. 2001;8:488-494.
13. Murdoch JAC, Gan TJ. Anesthesia for hysteroscopy. Anesthesiol Clin North Am. 2001;1:125-140.
14. Cooper JM, Brady RM. Late complications of operative hysteroscopy. Obstet Gynecol Clin North Am. 2000;27:367-374.
15. Rogerson L, Gannon B, O’Donovan P. Outcome of pregnancy following endometrial ablation. J Gynecol Surg. 1997;13:155-160.
16. Kerimis P, Zolti M, Sinwany G, Mashiach S, Carp H. Uterine rupture after hysteroscopic resection of uterine septum. Fertil Steril. 2002;77:618-620.
17. De Iaco P, Golfieri R, Ghi T, Muzzupapa G, Ceccarini M, Bovicelli L. Uterine fistula induced by hysteroscopic resection of an embolized migrated fibroid: a rare complication after embolization of uterine fibroids. Fertil Steril. 2001;75:818-820.
18. Coccia ME, Becattini C, Bracco GL, et al. Intraoperative ultrasound guidance for operative hysteroscopy. J Reprod Med. 2000;45:413-418.
19. Brooks-Carter GN, Killackey MA, Neuwirth RS. Adenocarcinoma of the endometrium after endometrial ablation. Obstet Gynecol. 2000;96:836-837.
20. Cooper JM. Swimming lessons: check the water before jumping in. J Am Assoc Gynecol Laparosc. 1998;5:87-90.
21. Kudela M, Pilka R. Is there a real risk in patients with endometrial carcinoma undergoing diagnostic hysteroscopy (HSC)? Eur J Gynecol Oncol. 2001;22:342-344.
Expert Panel: Techniques and tools to prevent pelvic adhesions


-

-

- Approximately 40% of people who undergo primary surgery develop adhesions and reformation occurs in 80% to 90% of cases.
- Microsurgical techniques such as gentle handling of tissues, careful hemostasis, and avoidance of heat may help reduce the incidence.
- Laparoscopy appears to be less likely to produce adhesions than laparotomy.
- Ob/Gyns should be aware of the potential for adhesion-related bowel obstruction and take steps to prevent it.
Are adhesions a pathologic response to injury or a normal aspect of healing? Can they be avoided, or are preventive efforts part of the problem? How useful are the different barriers in gynecologic surgery? What is the ideal adjuvant?
OBG Management convened a panel of experts to explore these and other questions.
Common problem, high recurrence rate
DECHERNEY: Adhesion formation is serious because it is associated with clinical entities such as infertility, pelvic pain, and bowel obstruction. We all agree that approximately 40% of people who undergo primary surgery develop adhesions and that 80% to 90% of patients who undergo lysis develop recurrent adhesions.
SANFILIPPO: One study several years ago explored adhesion formation.1 Unfortunately, no matter how meticulous the surgeon is, adhesions will form, even with microsurgical techniques and carefully ensured hemostasis.
HURD: The number of patients with significant adhesion formation after some gynecologic procedures has been reported to be greater than 90%.2
DECHERNEY: That higher incidence usually occurs after general surgery—and there’s a reason it is so high: General surgeons don’t use adjunctive therapy. They are critical of it. It is to our credit as gynecologic surgeons that we adopted adjunctive therapies about 15 years ago with the introduction of dextran 70 (Hyskon; Medisan Pharmaceuticals, Parsippany, NJ).
PAGIDAS: If anything, the pelvis seems to have even more of a predilection for adhesion formation than the abdomen, probably because of the close proximity of structures.
How and why adhesions form
DECHERNEY: What is the pathophysiology of adhesion formation? Let’s say you have 2 raw surface areas. What happens?
The process
PAGIDAS: The increase in leukotrienes and prostaglandins and the decrease in plasminogen activity (which actually initiates the inflammation) appear to be significant.
HURD: Vessel permeability also increases, and inflammatory cells leak through the vessels and set up a matrix for adhesion formation.
DECHERNEY: So we have 2 raw surface areas with fibrin leaking out and forming bridges between them.
PAGIDAS: The key is that it takes 2 surfaces to form these bridges. As I mentioned, the greater proximity of pelvic structures—particularly around the tube and ovary—probably contributes to adhesion formation.
DECHERNEY: Macrophage activity also is important. The macrophage “migrates” along these fibrin bridges and lays down collagen over a period of time. Then the collagen becomes organized and, eventually, vascular.
Window of opportunity
SANFILIPPO: Adhesion formation probably occurs and is pretty well established within 5 to 7 days of the precipitating event—usually surgery. Once that process is under way, attempts to halt it yield diminishing returns. Unfortunately, we don’t know how to interfere with it in a positive way.
HURD: Under normal conditions, there seems to be a balance between fibrin deposition and fibrinolysis. In some tissues, however, these functions become imbalanced. This disparity may contribute more to adhesions than the actual laying down of fibrin—especially in tissue that is hypoxic.
DECHERNEY: Would you say that adhesion formation represents normal or abnormal healing?
HURD: It is one of the body’s normal protective mechanisms and an important part of healing. Without it, any abdominal injury would likely result in death.
SANFILIPPO: I don’t think it differs that much from processes that occur externally. For example, if you get cut deeply enough, you develop a scar. Is that scar part of the normal healing process? It is.
PAGIDAS: Right. It is a normal process of tissue remodeling. The question is: What allows it to go astray?
What surgical techniques help prevent adhesions?
DECHERNEY: Let’s review the aspects of surgical technique that are important for adhesion prevention.
PAGIDAS: I emphasize the value of microsurgical techniques, which help to minimize severe tissue handling. It also is important to keep surfaces moistened so they don’t desiccate.
SANFILIPPO: I agree with Dr. Pagidas about microsurgical techniques such as gentle tissue handling, careful hemostasis, and keeping tissues moist. If we follow these principles, we create an environment that minimizes the potential for adhesion formation.
HURD: The findings of many well-controlled animal studies have been surprising. For example, it is difficult to demonstrate that drying of tissue increases adhesions.3 Probably the greatest contributor to adhesions in these models was abrasion.4 One way that laparoscopic surgery decreases adhesions is by avoiding abrasion of the bowel mucosa, which occurs specifically with packing.
A 38-year-old mother of 2 undergoes myomectomy for menorrhagia.
SANFILIPPO: The initial question is: Can this case be managed laparoscopically? I do myomectomies laparoscopically whenever possible, although I do close the uterus with a minilaparotomy incision. The reason is my strong concern about reapproximating the myometrium, since wound dehiscence sometimes occurs at the site of myoma removal.
In this case, depending on the size of the myomas, I would do as much as possible laparoscopically and then reapproximate the myometrium. I would plan my incisions carefully, to maximize the number of myomas that can be removed. I would end with meticulous hemostasis and, assuming it is successful, use a barrier over the incision—in this case, Interceed.
HURD: Does the patient desire future childbearing? If so, I would avoid the laparoscopic approach because of the possibly increased risk of uterine rupture during pregnancy. If she isn’t planning pregnancy, there are more options.
The next question is: How many myomas are there, and where are they located? If they are intrauterine, a hysteroscopic approach would avoid extrauterine adhesions. If they are multiple and large, I am pretty much limited to laparotomy. If there is 1 or only a few myomas, a laparoscopic approach would be best.
I have not used Interceed. In laparoscopic cases, I worry that it would create more problems because, as you allow the carbon dioxide to decrease at the end of a case, oozing begins. Instead of a barrier, I would use limited hydroflotation.
SANFILIPPO: That’s a good point. At the end of the myomectomy, with the laparoscope in place, I decrease the insufflation, eliminating the tamponade effect. Then, assuming good hemostasis, I apply Interceed.
HURD: With open cases, I use Seprafilm, which takes practice because, as it gets wet, it sticks to anything, including gloves and instruments. But if you can put it down dry on the uterus, it sticks and stays in place. If oozing occurs, it seems to block or stop it.
PAGIDAS: I want to reiterate the importance of determining whether childbearing is an issue. In this case, the biggest concern is the risk of adhesions developing on our incision, so I would use a barrier. My preference would be Seprafilm or GoreTex. If we can limit adhesions at the incision site, then hopefully we can minimize bowel and tuboovarian adhesions, too.
In open cases, one thing we can do to minimize the risk of adhesions is to pack gently when needed. Also, we should avoid using packing to reposition the bowel.
Another factor frequently overlooked is the application of heat, which appears to be a very effective way to create adhesions. This probably isn’t an issue for laparoscopic cases, but when you use irrigation fluid in an open case, watch the temperature. If it feels hot to you, you need to worry about potential injury to the bowel surfaces.
PAGIDAS: That is critical. In abdominal cases you want to make sure irrigation fluid is warm, but not too warm, because heat increases the vascular permeability of vessels and leads to more macrophages, more prostaglandins, and more leukotrienes.
HURD: Another important element is the type of suture material used.
DECHERNEY: Overall, we need to minimize the use of sutures. For example, when I am operating laparoscopically on an ovarian cyst, I try to apply bipolar energy to the edges so that they will coapt without a stitch.
HURD: When it first became clear that suturing ovaries increased adhesion formation, we conducted a controlled trial of different kinds of sutures in animals. Not surprisingly, we found that the less reactive the suture, the fewer adhesions.5 Sutures that are absorbed more slowly, such as polydioxanone, seem to be less reactive.
Obviously, inert sutures like nylon are the least reactive, but they are permanent. It is assumed that animal-protein sutures such as chromic and plain gut are the most reactive, although I am not sure there are sufficient data to support that conclusion.
Multiple clinical studies have shown laparoscopy to be associated with a lower adhesion rate, although it isn’t clear why. It may be related to decreased suturing.
DECHERNEY: Bulk is important, too—that is, the number of throws in the suture. When Vicryl (polyglactin 910) became available, we conducted a study in mice using proportionately small Vicryl plaques to determine whether this would be good a barrier (A. DeCherney, MD, unpublished data). It caused a tremendous amount of adhesion because so much foreign matter was applied.
We also did a study using human-size titanium clips in rats (A. DeCherney, MD, unpublished data). Not surprisingly, there was a lot of adhesion formation.
FIGURE 1 Pelvic adhesions: How they develop, problems they cause
Adhesions occur when 2 or more raw surfaces are exposed to leaking fibrin, which forms a bridge between the surfaces. Macrophages “migrate” along these bridges, depositing collagen.
The pelvis has a greater predilection for adhesions than the abdomen because of the close proximity of structures.
Although adhesions represent one of the body’s protective mechanisms, they may also cause pain or interfere with fertility, bowel function, or other processes.
Laparoscopy versus laparotomy: More adhesions in open cases?
DECHERNEY: Based on all the techniques we have learned from microsurgery—with the exception of magnification—it appears that laparoscopic procedures are less likely to cause adhesions than laparotomy. Do you agree?
PAGIDAS: I think so. As Dr. Hurd noted, a main reason is the diminished tissue handling, because there is no packing.
HURD: Multiple clinical studies have shown laparoscopy to be associated with a lower adhesion rate, although it isn’t clear why.6,7 It may be related to decreased suturing.
DECHERNEY: Less bleeding occurs because surgeons are less aggressive laparoscopically than in laparotomy.
A 15-year-old undergoes removal of a dermoid cyst, which was shelled out laparoscopically.
PAGIDAS: This case is easier because the cyst has been successfully shelled out. It is not the spill of a cyst’s contents at surgery that creates adhesions, but a chronic leak, which can occur if you do not remove the cyst in its entirety.
Once the cyst has been excised completely, I would ensure hemostasis with bipolar cautery and reapproximate the edges. I would not suture. There seems to be no clear advantage to suturing. I would use hydroflotation. Although Ringer’s lactate solution has not been shown to be effective, it is safe and has no toxicity.
DECHERNEY: Would you remove the cyst via laparotomy?
PAGIDAS: I would do it laparoscopically, using the endobag to minimize spillage, even though we know that a spill doesn’t necessarily change the outcome. If spillage does occur, I would perform copious irrigation to ensure that nothing is left behind.
HURD: My priority would be minimizing the use of power on the ovary. Studies of ovarian drilling have demonstrated that burning an ovary stimulates adhesion formation.21,22 If the dermoid cyst spills, as happens occasionally, I perform copious rinsing until no more oil is visible on the surface of the peritoneal fluid.
I also would minimize the amount of ovarian capsule that is removed. Good studies of endometriomas have shown that the more capsulate that is removed, the more adhesions. Even if the capsule looks redundant and floppy, the concern should be to achieve hemostasis with bipolar cautery and then leave it alone.
DECHERNEY: Would you use crystalloids in this case?
HURD: Yes.
SANFILIPPO: If spillage occurs, I would ensure that the patient is taken out of the Trendelenburg position. I want to emphasize the importance of thorough irrigation to eliminate any material that could produce chemical peritonitis.
DECHERNEY: Over the years, I have seen a fair number of cases of Fitz-Hugh and Curtis syndrome. You rigorously lavage a ruptured dermoid cyst, which sometimes presents with low-grade fever, but always with pain.
I’m surprised that none of you would use Interceed, since wrapping the ovary is the only thing for which it has been clearly shown to be effective. Since the cortex is relatively avascular, you don’t get a lot of bleeding. Unfortunately, it is not technically easy to wrap the ovary.
HURD: Since we do not know the effect on future fertility of changing the ovarian surface, less would seem to be better in patients this young.
I have stopped doing difficult cases laparoscopically. For example, it is rare for me to operate laparoscopically on a patient with stage IV endometriosis, at least when it comes to infertility—I might consider laparoscopy for pain.
I think case selection plays a role as well, although there are few data to back that up. It is purely clinical opinion.
SANFILIPPO: We need a well-designed prospective study to explore the effects of laparotomy versus laparoscopy. Existing data are not clear. You would assume laparoscopy would be associated with less adhesion formation. But genetic or other factors may explain why patient A is more prone to adhesions than patient B.
Does anybody think carbon dioxide plays a role in adhesion formation?
HURD: In the laboratory, carbon dioxide increases cell growth.8 Without an increased carbon dioxide concentration in the atmosphere, cell cultures don’t grow well. This might suggest that the carbon dioxide used for laparoscopy could actually enhance adhesion growth. Fortunately, this does not appear to be the case clinically.
With myomectomy, the surgeon needs to plan ahead to maximize the number of myomas removed from a single incision.
DECHERNEY: What about second-look laparoscopies? Do any of you perform them after a patient has undergone lysis of adhesions?
SANFILIPPO: Only as part of a research protocol. It amazes me how rapidly adhesions can form and how dense they are 2 or more weeks after the initial laparoscopic surgery.
PAGIDAS: We tend to limit second-look laparoscopy to a research protocol, although it is sometimes valuable after laparoscopic or abdominal myomectomy, which has the highest incidence of adhesions. If the surgeon can perform a second look and lyse adhesions, he or she may potentially alter the reproductive outcome. However, with assisted reproductive techniques becoming integral to every infertility case, that approach has begun to go out of style.
SANFILIPPO: That’s a good point. With myomectomy, the surgeon needs to plan ahead to maximize the number of myomas removed from a single incision. If adhesions do occur, it is best if they occur toward the bladder rather than in the area of the tubes and ovaries.
HURD: For second-look laparoscopy, we must keep in mind the cost and the small but real risks of surgery. Until good controlled studies show a reasonable clinical advantage, this approach probably should remain a research protocol.
SANFILIPPO: The literature suggests it is helpful, but does not help fertility, so second-look laparoscopy is used mainly to evaluate adjunctive therapies. I don’t think anybody uses it as a primary therapy anymore.
Bowel obstruction still a risk, though rarely seen by Ob/Gyns
DECHERNEY: Although bowel obstruction is fairly common, Ob/Gyns do not often encounter it because it occurs relatively distant from the index surgery. Even though a patient may not experience bowel obstruction in the first year, an obstruction related to the index surgery is just as likely to develop 20 years later as 2 years later. These patients usually are treated by general surgeons. Still, we should beware of the potential for bowel obstruction and take steps to prevent it, if at all possible. Do you agree?
PAGIDAS: Yes. We tend to forget about bowel obstruction because we rarely follow patients past pregnancy or the first trimester if they are seeking infertility treatment.
HURD: The primary problem seems to be the abdominal wall incision. Fortunately, cesarean section seems to carry a decreased risk of abdominal wall adhesions, probably because the uterus serves as a splint over the incision.
The Pfannenstiel incision also appears to have some advantage. Both human and animal models suggest little advantage or disadvantage when peritoneal closure is compared to nonclosure.9,10
SANFILIPPO: I’m curious about how the panelists manage loose clips. If you are using an EndoGIA (US Surgical, Norwalk, Conn) or other stapling device and you have free-floating clips, do you make a concerted effort to find them? In some cases, they have been implicated in bowel adhesion and obstruction. I try to retrieve loose clips, whether open or closed.
HURD: The advantage of those devices is minimal tissue damage, and the clips are inert. In general, inert, nonreactive clips have not been implicated as much in adhesion formation. I retrieve them if I see them, but I don’t search them out.
PAGIDAS: I do the same. If the clips are visible, I remove them. But I would not repack the bowel or do anything more heroic than look in locations where they might be.
What drugs may inhibit inflammatory response?
DECHERNEY: What about use of pharmacologic agents to prevent adhesions? Is there reason to think research should focus on inhibiting the inflammatory response? How important is polymorphonuclear cell infiltration?
Cyclooxygenase (COX) 2 agents could be helpful for inhibition of platelet function, since they are low in side effects. Thus, high doses of these drugs might be effective. At one time, aspirin was proposed, but you’d have to give a human so much aspirin that her ears would ring.
PAGIDAS: Pharmacologic agents have a role, especially for dampening the inflammatory immune response. But you don’t want to dampen it completely because, as we observed, it is an important part of healing. The difficulty is finding a balance between allowing the tissue to heal and preventing adhesions.
HURD: We studied the ability of a water-soluble prostaglandin inhibitor to prevent postoperative adhesion formation. Like many other agents, we found only a partial response.11
DECHERNEY: With current options, the best you can aim for is a 50% reduction.
Adjunctive therapy likely to limit adhesion rate Hydroflotation
DECHERNEY: The original adjunctive therapy was 20 mg dexamethasone and 25 mg femergin in 200 cc of Ringer’s lactate, with an equivalent amount of dexamethasone and femergin every 4 hours for a total of 6 doses. I prescribed that regimen because I was trained to do so. I stopped after it became clear that hydroflotation from the fluid—not the medication itself—was responsible for the improvement.
I must admit I gave it up reluctantly; patients felt fabulous with those higher load doses of glucocorticoids after surgery.
Do any of you use crystalloids as adjunctive therapy?
SANFILIPPO: In the animal model, they are so rapidly absorbed that they aren’t effective. I was a strong advocate, but now I don’t use them at all.
HURD: A lot depends on the kind of case. For instance, at the end of an open myomectomy, the patient often is oozing, so you want to use a barrier that blood won’t affect.
For ovarian surgery, you might want to specifically target the ovaries with some kind of coverage. But when you are doing a broad lysis of adhesions, you have few choices to cover the pelvis. In those cases I use hydroflotation with Ringer’s lactate. Both human and animal studies have shown some benefit in preventing adhesions, and it appears to have little risk.12,13
It’s better than nothing, in my opinion.
DECHERNEY: Do you use dextran 70 or crystalloids?
HURD: I use Ringer’s lactate solution. I was trained in the dextran 70 era, and there were certain problems with that approach. Since studies have failed to show a consistent effect of dextran 70, I no longer use this solution.14,15
DECHERNEY: Another problem with crystalloids is that they leak, which is disconcerting to the patient.
HURD: They also can mask an injury to the bladder in difficult cases.
DECHERNEY: I agree that dextran 70 is only appropriate in certain cases, but it is a good hydroflotation agent. Every cubic centimeter of dextran 70 brings in 1.2 cc of transudate, so it hangs around for at least 4 days.
It is appropriate only for certain surface areas—mainly the cul-de-sac. It is harmful on raw surface areas on the lateral pelvic sidewall because it tends to push the ovary and tube to those areas. Unless you are doing a lot of work in the deep pelvis, I would avoid dextran 70.
A 45-year-old woman undergoes an abdominal hysterectomy. The cuff is closed and the ovaries are intact.
PAGIDAS: I would do nothing other than ensure adequate hemostasis, check that I have left no round surfaces and, probably, use hydroflotation. I see no advantage to barriers.
HURD: One of the main causes of adhesions is devascularized tissue, and the perfect devascularized tissue might be the vaginal cuff. Re-“peritonealizing” the cuff might be advantageous. Thus, I would use minimal sutures—probably a slowly absorbable, light polydioxanone suture to place the peritoneum over the cuff so there are no pedicles.
DECHERNEY: All the pedicles are exteriorized.
HURD: Yes, that could be. We don’t bring all of it down like we used to years ago, but we do cover the cuff.
PAGIDAS: I agree that closing the cuff and reperitonealizing may actually minimize formation of hematomas—clearly an advantage.
SANFILIPPO: I agree. I guess I’m old fashioned. If it looks good, then hopefully it will stimulate less adhesion formation, so peritonealization is important.
As far as the abdominal incision is concerned, I would not close that peritoneum. I’m convinced now that there is no advantage.
DECHERNEY: Reperitonealizing the cuff is controversial. Most gynecologic surgeons do not do it, the theory being that the peritoneum is being stretched, attenuating the vessels that go through it and thereby creating an ischemic barrier that contributes to adhesions. Personally, I like to do it because it looks better—and that is certainly an important aspect of a surgery. No evidence shows that it is bad or good, either way.
In addition, there have been allergic reactions, most of which seem to occur in patients with fluid overload; a lot of the dextran 70 is absorbed.
With infertility patients, even if you lyse dense adhesions, you do not render the ovarian surface normal.
SANFILIPPO: Dextran 70 is not recommended for patients with sugar beet allergy, either.
We completed a study in a rabbit model, in which the peritoneal cavity was lavaged with either chlorhexidine or iodine.16 At the time of second-look surgery, the rate of adhesion formation was decreased, especially with the iodine preparation. I would hope that this has potential in humans.
DECHERNEY: In your lavage procedures to prevent adhesions, do any of you use heparin?
HURD: No.
PAGIDAS: I don’t think any evidence suggests that local administration changes the outcome.
DECHERNEY: I agree. When heparin has been used, the doses have been so low that it was not terribly helpful. And when you consider that hemorrhage can be a problem, heparin is probably deleterious rather than helpful.
Barriers
DECHERNEY: What about barriers? The first to become available, Interceed (Gynecare, a division of Ethicon, Somerville, NJ), is oxidized cellulose, similar to Surgicell (Johnson & Johnson, New Brunswick, NJ). Since it gelates quickly, there is no fenestration, so the fibrin is unable to penetrate. However, if the patient has bleeding by capillary action, the raw surface just moves from one side of the Interceed barrier to the other.
What has been your experience? Do you use it?
PAGIDAS: I do not use Interceed, although prospective randomized trials and a meta-analysis confirmed its benefits in de novo formation and reformation.17-19 I don’t use it because it requires complete hemostasis. Also, with the surfaces we work on—notably, the ovary and tube—it is difficult to apply to just 1 surface area. From a clinical perspective, I appreciate the data, but it is hard to ensure a good application to optimize its effectiveness.
A 29-year-old woman is undergoing her third cesarean section, although adhesions cause difficulty getting through anteriorly.
DECHERNEY: In this case, will you use barriers or re-peritonealize the surface? Will you do a 1- or a 2-layered closure?
HURD: In these cases, I have not been doing anything, since there is no peritoneum to reperitonealize—just old scar tissue. I assume that the uterus will immediately readhere to the anterior peritoneum where it was before.
DECHERNEY: You would use a barrier?
HURD: No. I would not.
PAGIDAS: I would take the same approach, although I have not performed cesarean sections in about 4 years. I don’t think any intervention would change the outcome. And, as Dr. Hurd mentioned, that is pretty much old scar tissue anyway.
SANFILIPPO: I use Interceed, but I agree with you about its limitations. Meticulous hemostasis is a prerequisite.
HURD: With infertility patients, even if you lyse dense adhesions, you do not render the ovarian surface normal. If those patients have dense adhesions of the ovary or the sidewall, I generally leave them alone, and I try to avoid putting Interceed around the ovaries. No study has shown that using Interceed improves pregnancy.
In contrast, when a chronic pain patient’s ovaries are densely adherent to the cul-de-sac, which appears to be highly associated with dyspareunia, I lyse the adhesions, achieve meticulous hemostasis, and then use Interceed. It is hard to demonstrate in a study that this approach decreases the chance of pain. Even so, it certainly does decrease the chance of the ovaries being adherent.
DECHERNEY: One issue with Interceed is that we don’t know what happens to it once the abdomen is closed. It may migrate significantly.
Psychological issues may also be involved. For example, patients with multiple somatic complaints may be less likely to benefit from lysis of adhesions.
PAGIDAS: Right. Interestingly, a meta-analysis of all the randomized trials involving mechanical barriers found no correlation to pregnancy outcome or pelvic pain.18 If we were to consider new trials, the psychological aspect would be worth looking into.
Seprafilm
DECHERNEY: Let’s move on to Seprafilm (Genzyme, Cambridge, Mass). What is it and how useful is it?
HURD: Seprafilm is modified hyaluronic acid, which forms a brittle, thin plastic layer. It is somewhat difficult to work with but, once it is in place, seems to adhere well. The presence of blood does not appear to be a problem, since the Seprafilm forms an impermeable barrier—unless it breaks. I have found it especially useful in myomectomies, which produce postoperative oozing through the incisions no matter how hard you try to prevent it.
In addition, in open cases, surfaces can easily be covered with this material. Unfortunately, it can’t be used laparoscopically because it is so brittle.
DECHERNEY: In my opinion, that is its major drawback.
PAGIDAS: In cardiac surgery, Seprafilm appears to work quite effectively.
SANFILIPPO: The manufacturer initially focused on surgeons in the context of sigmoid colon surgery, and it seems to work well in that setting.
Intergel
DECHERNEY: That brings us to the current state of the art: gels. I’m sure you all are familiar with Intergel (Lifecore Biomedical, Chaska, Minn), which is a ferrous derivative of hyaluronic acid that works by coating the raw surface areas. It also has the theoretical advantage of ease of use. Have any of you used Intergel?
HURD: As you know it was only recently approved by the US Food and Drug Administration, but not for laparoscopic use. It may work best on abraded bowel, which is avoided by laparoscopic surgery.
As you are probably also aware, the manufacturer recently took it off the market because of unusual side effects, namely a chemical peritonitis. Although peritonitis was cited as being rare, we encountered it in probably half the patients we operated on.
PAGIDAS: When we used it on hospitalized patients, the peritonitis wasn’t that obvious, since there was an expectation of significant pain. However, when we used Intergel on short-stay patients, we had to readmit them and do a full workup because we were concerned about bowel perforations. I’m surprised the manufacturer didn’t take it off the market sooner.
DECHERNEY: It seems strange, since Intergel has been used in Europe for a while now. I’ve used it in only 1 case and didn’t have adverse effects. It seemed to work well.
SANFILIPPO: It had all the right ingredients for success. It is unfortunate that these side effects have prohibited its use.
There is no question that adhesion prevention is one of the unmet challenges in all surgeries, especially reproductive surgery.
Gels and the cost factor
DECHERNEY: Other gels are in the pipeline. I’m reminded of plasminogen activator, which is a powerful antiadhesive agent that lyses fibrin effectively. Unfortunately, it is prohibitively expensive.
The next phase likely will involve the so-called polymers. If you spray them on your hand, they are activated by light or another chemical and become a cellophane-like substance. The problem is viscosity. If sprayed on the sidewall, for instance, they run halfway down before they are activated, so the entire surface does not get covered.
PAGIDAS: One concern with polymers is that they could actually bring surfaces together when they polymerize. We still have a lot to learn.
DECHERNEY: Let’s say a new gel comes on the market that takes reformation adhesions from 90% to 10%, as opposed to 40% recurrence. Would you use it in all 4 of the cases we discuss here?
HURD: If it was that effective and had no adverse effects, it would be wonderful.
The cesarean-delivery case is different, as healing in a pregnant patient is 1 concern; the size of the uterus also has an effect. If the patient is breastfeeding, you would want to make sure the gel didn’t interfere.
PAGIDAS: We desperately need a product that can minimize adhesions regardless of the route of access or type of procedure. Even though we lack data on outcomes, I predict wide use of such a product, assuming it is nontoxic and effective.
DECHERNEY: What if it costs $1,000 a case?
SANFILIPPO: If it prevents 1 bowel obstruction, it still would be cost-effective.
DECHERNEY: The incidence of bowel obstruction for total abdominal hysterectomy is 2%, and 5% for radical hysterectomy.
HURD: We must be careful of the cost-benefit ratio. Bowel obstruction after gynecologic surgery is uncommon.
Is the gel worth $100? $1,000? $3,000? It’s difficult to say, but the more expensive it is, the less likely it will find widespread use.
PAGIDAS: I agree. We should remember that we still need to maintain microsurgical techniques and appropriate tissue handling, as well as avoid ischemia and infection.
Looking for the magic bullet
DECHERNEY: What is the future of adjunctive therapy?
SANFILIPPO: I would focus on noxythiolin; it has potential. Calcium channel blockers for adhesion prevention have also been studied.20 In 1 investigation involving a rat model, the calcium channel blocker verapamil as well as several other agents—including vitamin E, carboxymethylcellulose, cyclosporin, aprotinin, and tenoxicam—were compared with respect to tissue effects. A beneficial effect was noted with all agents except cyclosporin and carboxymethylcellulose.
Whoever succeeds in manufacturing an effective preventive will be a winner.
HURD: There is no question that adhesion prevention is one of the unmet challenges in all surgeries, especially reproductive surgery. The most effective agent would be applied intraabdominally, since any systemic agent that stops adhesion formation would probably decrease wound healing as well.
I hope the most effective agents can be used in both laparoscopy and laparotomy, and that they will decrease the adhesion-formation rate by more than 50%. We need to find the magic bullet that can cover the entire pelvis—if not the entire abdomen.
Dr. DeCherney reports small holdings with Lifecore Biomedical and Johnson & Johnson. Drs. Hurd and Pagidas report no financial relationship with any companies whose products are mentioned in this article. Dr. Sanfilippo serves on the speaker’s bureau for Berlex, Ortho Pharmaceuticals, and Wyeth, and receives grant support from Eli Lilly and Wyeth.
1. Operative Laparoscopy Study Group Postoperative adhesion development after operative laparoscopy: evaluation at early second look procedures. Fertil Steril. 1991;55:700-704.
2. Tulandi T, Murray C, Guralnick M. Adhesion formation and reproductive outcome after myomectomy and second-look laparoscopy. Obstet Gynecol. 1993;82:213-215.
3. Larsson B, Perbeck L. The possible advantage of keeping the uterine and intestinal serosa irrigated with saline to prevent intraabdominal adhesions in operations for infertility. An experimental study in rats. Acta Chir Scand Suppl. 1985;530:15-18.
4. Zamir G, Bloom AI, Reissman P. Prevention of intestinal adhesions after laparotomy in a rat model—a randomized prospective study. Res Exp Med (Berl). 1998;197:349-353.
5. Hurd WW, Himebaugh KS, Cofer KF, Gauvin JM, Elkins TE. The etiology of closure-related adhesion formation after wedge resection of the rabbit ovary. J Reprod Med. 1993;38:465-468.
6. Chen MD, Teigen GA, Reynolds HT, Johnson PR, Fowler JM. Laparoscopy versus laparotomy: an evaluation of adhesion formation after pelvic and paraaortic lymphadenectomy in a porcine model. Am J Obstet Gynecol. 1998;178:499-503.
7. Polymeneas G, Theodosopoulos T, Stamatiadis A, Kourias E. A comparative study of postoperative adhesion formation after laparoscopic vs. open cholecystectomy. Surg Endosc. 2001;15:41-43.
8. Smidt VJ, Singh DM, Hurteau JA, Hurd WW. Effect of carbon dioxide on human ovarian carcinoma cell growth. Am J Obstet Gynecol. 2001;185:1314-1317.
9. Tulandi T, Hum HS, Gelfand MM. Closure of laparotomy incisions with or without peritoneal suturing and second look laparoscopy. Am J Obstet Gynecol. 1988;158:536-537.
10. Kapur ML, Daneswar A, Chopra P. Evaluation of peritoneal closure at laparotomy. Am J Surg. 1979;137:650-652.
11. Cofer KF, Himebaugh KS, Gauvin JM, Hurd WW. Inhibition of adhesion reformation in the rabbit model by meclofenamate: an inhibitor of both prostaglandin and leukotriene production. Fertil Steril. 1994;62:1262-1265.
12. Elkelani OA, Molinas CR, Mynbaev O, Koninckx PR. Prevention of adhesions with crystalloids during laparoscopic surgery in mice. J Am Assoc Gynecol Laparosc. 2002;9:447-452.
13. Chan KL, Marino T, Qu WM, Tulandi T. Effects of intraperitoneal Ringer’s lactate instillation and infusion on postsurgical adhesion formation. J Gynecol Surg. 1995;11:241-243.
14. Watson A, Vandekerckhove P, Lilford R. Pharmacological adjuvants during infertility surgery: a systematic review of evidence derived from randomized controlled trial. Hum Fertil (Camb). 1999;2:149-157.
15. Larson B. Dextran—later clinical studies. clinical and experimental evaluation of different adjuvant therapies. Prog Clin Biol Res. 1990;358:165-175.
16. Roberts L, Sanfilippo JS, Ehrlich G, Raab S. Adhesion formation, peritoneal effects and the presence of bacterial biofilms after laparoscopic lavage in an animal model of pelvic inflammatory disease. J Am Assoc Gynecol Laparosc. 2002;9:4.-
17. Barrier agents for preventing adhesions after surgery for subfertility Cochrane Database Syst Rev. 2000;CD000475.-
18. Wiseman DM, Trout JR, Franklin RR, Diamond MP. Metaanalysis of the safety and efficacy of an adhesion barrier (Interceed TC7) in laparotomy. J Reprod Med. 1999;44:325-331.
19. Sawada T, Nishizawa H, Nishio E, Kadowaki M. Postoperative adhesion prevention with an oxidized regenerated cellulose adhesion barrier in infertile women. J Reprod. Med. 2000;45:387-389.
20. Uzunkoy A, Akinci OF, Coskun A, Aslan O, Kocyigit A. Effects of antiadhesive agents on the healing of intestinal anastomosis. Dis Colon Rectum. 2000;43:370-375.
21. Farquhar C, Vandekerckhove P, Arnot M, Lilford R. Laparoscopic “drilling” by diathermy or laser for ovulation induction in anovulatory polycystic ovary syndrome. Cochrane Database Syst Rev. 2000(2);CD001122.-
22. Greenblatt EM, Casper RF. Laparoscopic ovarian drilling in women with polycystic ovarian syndrome. Prog Clin Biol Res. 1993;381:129-138.


-

-

- Approximately 40% of people who undergo primary surgery develop adhesions and reformation occurs in 80% to 90% of cases.
- Microsurgical techniques such as gentle handling of tissues, careful hemostasis, and avoidance of heat may help reduce the incidence.
- Laparoscopy appears to be less likely to produce adhesions than laparotomy.
- Ob/Gyns should be aware of the potential for adhesion-related bowel obstruction and take steps to prevent it.
Are adhesions a pathologic response to injury or a normal aspect of healing? Can they be avoided, or are preventive efforts part of the problem? How useful are the different barriers in gynecologic surgery? What is the ideal adjuvant?
OBG Management convened a panel of experts to explore these and other questions.
Common problem, high recurrence rate
DECHERNEY: Adhesion formation is serious because it is associated with clinical entities such as infertility, pelvic pain, and bowel obstruction. We all agree that approximately 40% of people who undergo primary surgery develop adhesions and that 80% to 90% of patients who undergo lysis develop recurrent adhesions.
SANFILIPPO: One study several years ago explored adhesion formation.1 Unfortunately, no matter how meticulous the surgeon is, adhesions will form, even with microsurgical techniques and carefully ensured hemostasis.
HURD: The number of patients with significant adhesion formation after some gynecologic procedures has been reported to be greater than 90%.2
DECHERNEY: That higher incidence usually occurs after general surgery—and there’s a reason it is so high: General surgeons don’t use adjunctive therapy. They are critical of it. It is to our credit as gynecologic surgeons that we adopted adjunctive therapies about 15 years ago with the introduction of dextran 70 (Hyskon; Medisan Pharmaceuticals, Parsippany, NJ).
PAGIDAS: If anything, the pelvis seems to have even more of a predilection for adhesion formation than the abdomen, probably because of the close proximity of structures.
How and why adhesions form
DECHERNEY: What is the pathophysiology of adhesion formation? Let’s say you have 2 raw surface areas. What happens?
The process
PAGIDAS: The increase in leukotrienes and prostaglandins and the decrease in plasminogen activity (which actually initiates the inflammation) appear to be significant.
HURD: Vessel permeability also increases, and inflammatory cells leak through the vessels and set up a matrix for adhesion formation.
DECHERNEY: So we have 2 raw surface areas with fibrin leaking out and forming bridges between them.
PAGIDAS: The key is that it takes 2 surfaces to form these bridges. As I mentioned, the greater proximity of pelvic structures—particularly around the tube and ovary—probably contributes to adhesion formation.
DECHERNEY: Macrophage activity also is important. The macrophage “migrates” along these fibrin bridges and lays down collagen over a period of time. Then the collagen becomes organized and, eventually, vascular.
Window of opportunity
SANFILIPPO: Adhesion formation probably occurs and is pretty well established within 5 to 7 days of the precipitating event—usually surgery. Once that process is under way, attempts to halt it yield diminishing returns. Unfortunately, we don’t know how to interfere with it in a positive way.
HURD: Under normal conditions, there seems to be a balance between fibrin deposition and fibrinolysis. In some tissues, however, these functions become imbalanced. This disparity may contribute more to adhesions than the actual laying down of fibrin—especially in tissue that is hypoxic.
DECHERNEY: Would you say that adhesion formation represents normal or abnormal healing?
HURD: It is one of the body’s normal protective mechanisms and an important part of healing. Without it, any abdominal injury would likely result in death.
SANFILIPPO: I don’t think it differs that much from processes that occur externally. For example, if you get cut deeply enough, you develop a scar. Is that scar part of the normal healing process? It is.
PAGIDAS: Right. It is a normal process of tissue remodeling. The question is: What allows it to go astray?
What surgical techniques help prevent adhesions?
DECHERNEY: Let’s review the aspects of surgical technique that are important for adhesion prevention.
PAGIDAS: I emphasize the value of microsurgical techniques, which help to minimize severe tissue handling. It also is important to keep surfaces moistened so they don’t desiccate.
SANFILIPPO: I agree with Dr. Pagidas about microsurgical techniques such as gentle tissue handling, careful hemostasis, and keeping tissues moist. If we follow these principles, we create an environment that minimizes the potential for adhesion formation.
HURD: The findings of many well-controlled animal studies have been surprising. For example, it is difficult to demonstrate that drying of tissue increases adhesions.3 Probably the greatest contributor to adhesions in these models was abrasion.4 One way that laparoscopic surgery decreases adhesions is by avoiding abrasion of the bowel mucosa, which occurs specifically with packing.
A 38-year-old mother of 2 undergoes myomectomy for menorrhagia.
SANFILIPPO: The initial question is: Can this case be managed laparoscopically? I do myomectomies laparoscopically whenever possible, although I do close the uterus with a minilaparotomy incision. The reason is my strong concern about reapproximating the myometrium, since wound dehiscence sometimes occurs at the site of myoma removal.
In this case, depending on the size of the myomas, I would do as much as possible laparoscopically and then reapproximate the myometrium. I would plan my incisions carefully, to maximize the number of myomas that can be removed. I would end with meticulous hemostasis and, assuming it is successful, use a barrier over the incision—in this case, Interceed.
HURD: Does the patient desire future childbearing? If so, I would avoid the laparoscopic approach because of the possibly increased risk of uterine rupture during pregnancy. If she isn’t planning pregnancy, there are more options.
The next question is: How many myomas are there, and where are they located? If they are intrauterine, a hysteroscopic approach would avoid extrauterine adhesions. If they are multiple and large, I am pretty much limited to laparotomy. If there is 1 or only a few myomas, a laparoscopic approach would be best.
I have not used Interceed. In laparoscopic cases, I worry that it would create more problems because, as you allow the carbon dioxide to decrease at the end of a case, oozing begins. Instead of a barrier, I would use limited hydroflotation.
SANFILIPPO: That’s a good point. At the end of the myomectomy, with the laparoscope in place, I decrease the insufflation, eliminating the tamponade effect. Then, assuming good hemostasis, I apply Interceed.
HURD: With open cases, I use Seprafilm, which takes practice because, as it gets wet, it sticks to anything, including gloves and instruments. But if you can put it down dry on the uterus, it sticks and stays in place. If oozing occurs, it seems to block or stop it.
PAGIDAS: I want to reiterate the importance of determining whether childbearing is an issue. In this case, the biggest concern is the risk of adhesions developing on our incision, so I would use a barrier. My preference would be Seprafilm or GoreTex. If we can limit adhesions at the incision site, then hopefully we can minimize bowel and tuboovarian adhesions, too.
In open cases, one thing we can do to minimize the risk of adhesions is to pack gently when needed. Also, we should avoid using packing to reposition the bowel.
Another factor frequently overlooked is the application of heat, which appears to be a very effective way to create adhesions. This probably isn’t an issue for laparoscopic cases, but when you use irrigation fluid in an open case, watch the temperature. If it feels hot to you, you need to worry about potential injury to the bowel surfaces.
PAGIDAS: That is critical. In abdominal cases you want to make sure irrigation fluid is warm, but not too warm, because heat increases the vascular permeability of vessels and leads to more macrophages, more prostaglandins, and more leukotrienes.
HURD: Another important element is the type of suture material used.
DECHERNEY: Overall, we need to minimize the use of sutures. For example, when I am operating laparoscopically on an ovarian cyst, I try to apply bipolar energy to the edges so that they will coapt without a stitch.
HURD: When it first became clear that suturing ovaries increased adhesion formation, we conducted a controlled trial of different kinds of sutures in animals. Not surprisingly, we found that the less reactive the suture, the fewer adhesions.5 Sutures that are absorbed more slowly, such as polydioxanone, seem to be less reactive.
Obviously, inert sutures like nylon are the least reactive, but they are permanent. It is assumed that animal-protein sutures such as chromic and plain gut are the most reactive, although I am not sure there are sufficient data to support that conclusion.
Multiple clinical studies have shown laparoscopy to be associated with a lower adhesion rate, although it isn’t clear why. It may be related to decreased suturing.
DECHERNEY: Bulk is important, too—that is, the number of throws in the suture. When Vicryl (polyglactin 910) became available, we conducted a study in mice using proportionately small Vicryl plaques to determine whether this would be good a barrier (A. DeCherney, MD, unpublished data). It caused a tremendous amount of adhesion because so much foreign matter was applied.
We also did a study using human-size titanium clips in rats (A. DeCherney, MD, unpublished data). Not surprisingly, there was a lot of adhesion formation.
FIGURE 1 Pelvic adhesions: How they develop, problems they cause
Adhesions occur when 2 or more raw surfaces are exposed to leaking fibrin, which forms a bridge between the surfaces. Macrophages “migrate” along these bridges, depositing collagen.
The pelvis has a greater predilection for adhesions than the abdomen because of the close proximity of structures.
Although adhesions represent one of the body’s protective mechanisms, they may also cause pain or interfere with fertility, bowel function, or other processes.
Laparoscopy versus laparotomy: More adhesions in open cases?
DECHERNEY: Based on all the techniques we have learned from microsurgery—with the exception of magnification—it appears that laparoscopic procedures are less likely to cause adhesions than laparotomy. Do you agree?
PAGIDAS: I think so. As Dr. Hurd noted, a main reason is the diminished tissue handling, because there is no packing.
HURD: Multiple clinical studies have shown laparoscopy to be associated with a lower adhesion rate, although it isn’t clear why.6,7 It may be related to decreased suturing.
DECHERNEY: Less bleeding occurs because surgeons are less aggressive laparoscopically than in laparotomy.
A 15-year-old undergoes removal of a dermoid cyst, which was shelled out laparoscopically.
PAGIDAS: This case is easier because the cyst has been successfully shelled out. It is not the spill of a cyst’s contents at surgery that creates adhesions, but a chronic leak, which can occur if you do not remove the cyst in its entirety.
Once the cyst has been excised completely, I would ensure hemostasis with bipolar cautery and reapproximate the edges. I would not suture. There seems to be no clear advantage to suturing. I would use hydroflotation. Although Ringer’s lactate solution has not been shown to be effective, it is safe and has no toxicity.
DECHERNEY: Would you remove the cyst via laparotomy?
PAGIDAS: I would do it laparoscopically, using the endobag to minimize spillage, even though we know that a spill doesn’t necessarily change the outcome. If spillage does occur, I would perform copious irrigation to ensure that nothing is left behind.
HURD: My priority would be minimizing the use of power on the ovary. Studies of ovarian drilling have demonstrated that burning an ovary stimulates adhesion formation.21,22 If the dermoid cyst spills, as happens occasionally, I perform copious rinsing until no more oil is visible on the surface of the peritoneal fluid.
I also would minimize the amount of ovarian capsule that is removed. Good studies of endometriomas have shown that the more capsulate that is removed, the more adhesions. Even if the capsule looks redundant and floppy, the concern should be to achieve hemostasis with bipolar cautery and then leave it alone.
DECHERNEY: Would you use crystalloids in this case?
HURD: Yes.
SANFILIPPO: If spillage occurs, I would ensure that the patient is taken out of the Trendelenburg position. I want to emphasize the importance of thorough irrigation to eliminate any material that could produce chemical peritonitis.
DECHERNEY: Over the years, I have seen a fair number of cases of Fitz-Hugh and Curtis syndrome. You rigorously lavage a ruptured dermoid cyst, which sometimes presents with low-grade fever, but always with pain.
I’m surprised that none of you would use Interceed, since wrapping the ovary is the only thing for which it has been clearly shown to be effective. Since the cortex is relatively avascular, you don’t get a lot of bleeding. Unfortunately, it is not technically easy to wrap the ovary.
HURD: Since we do not know the effect on future fertility of changing the ovarian surface, less would seem to be better in patients this young.
I have stopped doing difficult cases laparoscopically. For example, it is rare for me to operate laparoscopically on a patient with stage IV endometriosis, at least when it comes to infertility—I might consider laparoscopy for pain.
I think case selection plays a role as well, although there are few data to back that up. It is purely clinical opinion.
SANFILIPPO: We need a well-designed prospective study to explore the effects of laparotomy versus laparoscopy. Existing data are not clear. You would assume laparoscopy would be associated with less adhesion formation. But genetic or other factors may explain why patient A is more prone to adhesions than patient B.
Does anybody think carbon dioxide plays a role in adhesion formation?
HURD: In the laboratory, carbon dioxide increases cell growth.8 Without an increased carbon dioxide concentration in the atmosphere, cell cultures don’t grow well. This might suggest that the carbon dioxide used for laparoscopy could actually enhance adhesion growth. Fortunately, this does not appear to be the case clinically.
With myomectomy, the surgeon needs to plan ahead to maximize the number of myomas removed from a single incision.
DECHERNEY: What about second-look laparoscopies? Do any of you perform them after a patient has undergone lysis of adhesions?
SANFILIPPO: Only as part of a research protocol. It amazes me how rapidly adhesions can form and how dense they are 2 or more weeks after the initial laparoscopic surgery.
PAGIDAS: We tend to limit second-look laparoscopy to a research protocol, although it is sometimes valuable after laparoscopic or abdominal myomectomy, which has the highest incidence of adhesions. If the surgeon can perform a second look and lyse adhesions, he or she may potentially alter the reproductive outcome. However, with assisted reproductive techniques becoming integral to every infertility case, that approach has begun to go out of style.
SANFILIPPO: That’s a good point. With myomectomy, the surgeon needs to plan ahead to maximize the number of myomas removed from a single incision. If adhesions do occur, it is best if they occur toward the bladder rather than in the area of the tubes and ovaries.
HURD: For second-look laparoscopy, we must keep in mind the cost and the small but real risks of surgery. Until good controlled studies show a reasonable clinical advantage, this approach probably should remain a research protocol.
SANFILIPPO: The literature suggests it is helpful, but does not help fertility, so second-look laparoscopy is used mainly to evaluate adjunctive therapies. I don’t think anybody uses it as a primary therapy anymore.
Bowel obstruction still a risk, though rarely seen by Ob/Gyns
DECHERNEY: Although bowel obstruction is fairly common, Ob/Gyns do not often encounter it because it occurs relatively distant from the index surgery. Even though a patient may not experience bowel obstruction in the first year, an obstruction related to the index surgery is just as likely to develop 20 years later as 2 years later. These patients usually are treated by general surgeons. Still, we should beware of the potential for bowel obstruction and take steps to prevent it, if at all possible. Do you agree?
PAGIDAS: Yes. We tend to forget about bowel obstruction because we rarely follow patients past pregnancy or the first trimester if they are seeking infertility treatment.
HURD: The primary problem seems to be the abdominal wall incision. Fortunately, cesarean section seems to carry a decreased risk of abdominal wall adhesions, probably because the uterus serves as a splint over the incision.
The Pfannenstiel incision also appears to have some advantage. Both human and animal models suggest little advantage or disadvantage when peritoneal closure is compared to nonclosure.9,10
SANFILIPPO: I’m curious about how the panelists manage loose clips. If you are using an EndoGIA (US Surgical, Norwalk, Conn) or other stapling device and you have free-floating clips, do you make a concerted effort to find them? In some cases, they have been implicated in bowel adhesion and obstruction. I try to retrieve loose clips, whether open or closed.
HURD: The advantage of those devices is minimal tissue damage, and the clips are inert. In general, inert, nonreactive clips have not been implicated as much in adhesion formation. I retrieve them if I see them, but I don’t search them out.
PAGIDAS: I do the same. If the clips are visible, I remove them. But I would not repack the bowel or do anything more heroic than look in locations where they might be.
What drugs may inhibit inflammatory response?
DECHERNEY: What about use of pharmacologic agents to prevent adhesions? Is there reason to think research should focus on inhibiting the inflammatory response? How important is polymorphonuclear cell infiltration?
Cyclooxygenase (COX) 2 agents could be helpful for inhibition of platelet function, since they are low in side effects. Thus, high doses of these drugs might be effective. At one time, aspirin was proposed, but you’d have to give a human so much aspirin that her ears would ring.
PAGIDAS: Pharmacologic agents have a role, especially for dampening the inflammatory immune response. But you don’t want to dampen it completely because, as we observed, it is an important part of healing. The difficulty is finding a balance between allowing the tissue to heal and preventing adhesions.
HURD: We studied the ability of a water-soluble prostaglandin inhibitor to prevent postoperative adhesion formation. Like many other agents, we found only a partial response.11
DECHERNEY: With current options, the best you can aim for is a 50% reduction.
Adjunctive therapy likely to limit adhesion rate Hydroflotation
DECHERNEY: The original adjunctive therapy was 20 mg dexamethasone and 25 mg femergin in 200 cc of Ringer’s lactate, with an equivalent amount of dexamethasone and femergin every 4 hours for a total of 6 doses. I prescribed that regimen because I was trained to do so. I stopped after it became clear that hydroflotation from the fluid—not the medication itself—was responsible for the improvement.
I must admit I gave it up reluctantly; patients felt fabulous with those higher load doses of glucocorticoids after surgery.
Do any of you use crystalloids as adjunctive therapy?
SANFILIPPO: In the animal model, they are so rapidly absorbed that they aren’t effective. I was a strong advocate, but now I don’t use them at all.
HURD: A lot depends on the kind of case. For instance, at the end of an open myomectomy, the patient often is oozing, so you want to use a barrier that blood won’t affect.
For ovarian surgery, you might want to specifically target the ovaries with some kind of coverage. But when you are doing a broad lysis of adhesions, you have few choices to cover the pelvis. In those cases I use hydroflotation with Ringer’s lactate. Both human and animal studies have shown some benefit in preventing adhesions, and it appears to have little risk.12,13
It’s better than nothing, in my opinion.
DECHERNEY: Do you use dextran 70 or crystalloids?
HURD: I use Ringer’s lactate solution. I was trained in the dextran 70 era, and there were certain problems with that approach. Since studies have failed to show a consistent effect of dextran 70, I no longer use this solution.14,15
DECHERNEY: Another problem with crystalloids is that they leak, which is disconcerting to the patient.
HURD: They also can mask an injury to the bladder in difficult cases.
DECHERNEY: I agree that dextran 70 is only appropriate in certain cases, but it is a good hydroflotation agent. Every cubic centimeter of dextran 70 brings in 1.2 cc of transudate, so it hangs around for at least 4 days.
It is appropriate only for certain surface areas—mainly the cul-de-sac. It is harmful on raw surface areas on the lateral pelvic sidewall because it tends to push the ovary and tube to those areas. Unless you are doing a lot of work in the deep pelvis, I would avoid dextran 70.
A 45-year-old woman undergoes an abdominal hysterectomy. The cuff is closed and the ovaries are intact.
PAGIDAS: I would do nothing other than ensure adequate hemostasis, check that I have left no round surfaces and, probably, use hydroflotation. I see no advantage to barriers.
HURD: One of the main causes of adhesions is devascularized tissue, and the perfect devascularized tissue might be the vaginal cuff. Re-“peritonealizing” the cuff might be advantageous. Thus, I would use minimal sutures—probably a slowly absorbable, light polydioxanone suture to place the peritoneum over the cuff so there are no pedicles.
DECHERNEY: All the pedicles are exteriorized.
HURD: Yes, that could be. We don’t bring all of it down like we used to years ago, but we do cover the cuff.
PAGIDAS: I agree that closing the cuff and reperitonealizing may actually minimize formation of hematomas—clearly an advantage.
SANFILIPPO: I agree. I guess I’m old fashioned. If it looks good, then hopefully it will stimulate less adhesion formation, so peritonealization is important.
As far as the abdominal incision is concerned, I would not close that peritoneum. I’m convinced now that there is no advantage.
DECHERNEY: Reperitonealizing the cuff is controversial. Most gynecologic surgeons do not do it, the theory being that the peritoneum is being stretched, attenuating the vessels that go through it and thereby creating an ischemic barrier that contributes to adhesions. Personally, I like to do it because it looks better—and that is certainly an important aspect of a surgery. No evidence shows that it is bad or good, either way.
In addition, there have been allergic reactions, most of which seem to occur in patients with fluid overload; a lot of the dextran 70 is absorbed.
With infertility patients, even if you lyse dense adhesions, you do not render the ovarian surface normal.
SANFILIPPO: Dextran 70 is not recommended for patients with sugar beet allergy, either.
We completed a study in a rabbit model, in which the peritoneal cavity was lavaged with either chlorhexidine or iodine.16 At the time of second-look surgery, the rate of adhesion formation was decreased, especially with the iodine preparation. I would hope that this has potential in humans.
DECHERNEY: In your lavage procedures to prevent adhesions, do any of you use heparin?
HURD: No.
PAGIDAS: I don’t think any evidence suggests that local administration changes the outcome.
DECHERNEY: I agree. When heparin has been used, the doses have been so low that it was not terribly helpful. And when you consider that hemorrhage can be a problem, heparin is probably deleterious rather than helpful.
Barriers
DECHERNEY: What about barriers? The first to become available, Interceed (Gynecare, a division of Ethicon, Somerville, NJ), is oxidized cellulose, similar to Surgicell (Johnson & Johnson, New Brunswick, NJ). Since it gelates quickly, there is no fenestration, so the fibrin is unable to penetrate. However, if the patient has bleeding by capillary action, the raw surface just moves from one side of the Interceed barrier to the other.
What has been your experience? Do you use it?
PAGIDAS: I do not use Interceed, although prospective randomized trials and a meta-analysis confirmed its benefits in de novo formation and reformation.17-19 I don’t use it because it requires complete hemostasis. Also, with the surfaces we work on—notably, the ovary and tube—it is difficult to apply to just 1 surface area. From a clinical perspective, I appreciate the data, but it is hard to ensure a good application to optimize its effectiveness.
A 29-year-old woman is undergoing her third cesarean section, although adhesions cause difficulty getting through anteriorly.
DECHERNEY: In this case, will you use barriers or re-peritonealize the surface? Will you do a 1- or a 2-layered closure?
HURD: In these cases, I have not been doing anything, since there is no peritoneum to reperitonealize—just old scar tissue. I assume that the uterus will immediately readhere to the anterior peritoneum where it was before.
DECHERNEY: You would use a barrier?
HURD: No. I would not.
PAGIDAS: I would take the same approach, although I have not performed cesarean sections in about 4 years. I don’t think any intervention would change the outcome. And, as Dr. Hurd mentioned, that is pretty much old scar tissue anyway.
SANFILIPPO: I use Interceed, but I agree with you about its limitations. Meticulous hemostasis is a prerequisite.
HURD: With infertility patients, even if you lyse dense adhesions, you do not render the ovarian surface normal. If those patients have dense adhesions of the ovary or the sidewall, I generally leave them alone, and I try to avoid putting Interceed around the ovaries. No study has shown that using Interceed improves pregnancy.
In contrast, when a chronic pain patient’s ovaries are densely adherent to the cul-de-sac, which appears to be highly associated with dyspareunia, I lyse the adhesions, achieve meticulous hemostasis, and then use Interceed. It is hard to demonstrate in a study that this approach decreases the chance of pain. Even so, it certainly does decrease the chance of the ovaries being adherent.
DECHERNEY: One issue with Interceed is that we don’t know what happens to it once the abdomen is closed. It may migrate significantly.
Psychological issues may also be involved. For example, patients with multiple somatic complaints may be less likely to benefit from lysis of adhesions.
PAGIDAS: Right. Interestingly, a meta-analysis of all the randomized trials involving mechanical barriers found no correlation to pregnancy outcome or pelvic pain.18 If we were to consider new trials, the psychological aspect would be worth looking into.
Seprafilm
DECHERNEY: Let’s move on to Seprafilm (Genzyme, Cambridge, Mass). What is it and how useful is it?
HURD: Seprafilm is modified hyaluronic acid, which forms a brittle, thin plastic layer. It is somewhat difficult to work with but, once it is in place, seems to adhere well. The presence of blood does not appear to be a problem, since the Seprafilm forms an impermeable barrier—unless it breaks. I have found it especially useful in myomectomies, which produce postoperative oozing through the incisions no matter how hard you try to prevent it.
In addition, in open cases, surfaces can easily be covered with this material. Unfortunately, it can’t be used laparoscopically because it is so brittle.
DECHERNEY: In my opinion, that is its major drawback.
PAGIDAS: In cardiac surgery, Seprafilm appears to work quite effectively.
SANFILIPPO: The manufacturer initially focused on surgeons in the context of sigmoid colon surgery, and it seems to work well in that setting.
Intergel
DECHERNEY: That brings us to the current state of the art: gels. I’m sure you all are familiar with Intergel (Lifecore Biomedical, Chaska, Minn), which is a ferrous derivative of hyaluronic acid that works by coating the raw surface areas. It also has the theoretical advantage of ease of use. Have any of you used Intergel?
HURD: As you know it was only recently approved by the US Food and Drug Administration, but not for laparoscopic use. It may work best on abraded bowel, which is avoided by laparoscopic surgery.
As you are probably also aware, the manufacturer recently took it off the market because of unusual side effects, namely a chemical peritonitis. Although peritonitis was cited as being rare, we encountered it in probably half the patients we operated on.
PAGIDAS: When we used it on hospitalized patients, the peritonitis wasn’t that obvious, since there was an expectation of significant pain. However, when we used Intergel on short-stay patients, we had to readmit them and do a full workup because we were concerned about bowel perforations. I’m surprised the manufacturer didn’t take it off the market sooner.
DECHERNEY: It seems strange, since Intergel has been used in Europe for a while now. I’ve used it in only 1 case and didn’t have adverse effects. It seemed to work well.
SANFILIPPO: It had all the right ingredients for success. It is unfortunate that these side effects have prohibited its use.
There is no question that adhesion prevention is one of the unmet challenges in all surgeries, especially reproductive surgery.
Gels and the cost factor
DECHERNEY: Other gels are in the pipeline. I’m reminded of plasminogen activator, which is a powerful antiadhesive agent that lyses fibrin effectively. Unfortunately, it is prohibitively expensive.
The next phase likely will involve the so-called polymers. If you spray them on your hand, they are activated by light or another chemical and become a cellophane-like substance. The problem is viscosity. If sprayed on the sidewall, for instance, they run halfway down before they are activated, so the entire surface does not get covered.
PAGIDAS: One concern with polymers is that they could actually bring surfaces together when they polymerize. We still have a lot to learn.
DECHERNEY: Let’s say a new gel comes on the market that takes reformation adhesions from 90% to 10%, as opposed to 40% recurrence. Would you use it in all 4 of the cases we discuss here?
HURD: If it was that effective and had no adverse effects, it would be wonderful.
The cesarean-delivery case is different, as healing in a pregnant patient is 1 concern; the size of the uterus also has an effect. If the patient is breastfeeding, you would want to make sure the gel didn’t interfere.
PAGIDAS: We desperately need a product that can minimize adhesions regardless of the route of access or type of procedure. Even though we lack data on outcomes, I predict wide use of such a product, assuming it is nontoxic and effective.
DECHERNEY: What if it costs $1,000 a case?
SANFILIPPO: If it prevents 1 bowel obstruction, it still would be cost-effective.
DECHERNEY: The incidence of bowel obstruction for total abdominal hysterectomy is 2%, and 5% for radical hysterectomy.
HURD: We must be careful of the cost-benefit ratio. Bowel obstruction after gynecologic surgery is uncommon.
Is the gel worth $100? $1,000? $3,000? It’s difficult to say, but the more expensive it is, the less likely it will find widespread use.
PAGIDAS: I agree. We should remember that we still need to maintain microsurgical techniques and appropriate tissue handling, as well as avoid ischemia and infection.
Looking for the magic bullet
DECHERNEY: What is the future of adjunctive therapy?
SANFILIPPO: I would focus on noxythiolin; it has potential. Calcium channel blockers for adhesion prevention have also been studied.20 In 1 investigation involving a rat model, the calcium channel blocker verapamil as well as several other agents—including vitamin E, carboxymethylcellulose, cyclosporin, aprotinin, and tenoxicam—were compared with respect to tissue effects. A beneficial effect was noted with all agents except cyclosporin and carboxymethylcellulose.
Whoever succeeds in manufacturing an effective preventive will be a winner.
HURD: There is no question that adhesion prevention is one of the unmet challenges in all surgeries, especially reproductive surgery. The most effective agent would be applied intraabdominally, since any systemic agent that stops adhesion formation would probably decrease wound healing as well.
I hope the most effective agents can be used in both laparoscopy and laparotomy, and that they will decrease the adhesion-formation rate by more than 50%. We need to find the magic bullet that can cover the entire pelvis—if not the entire abdomen.
Dr. DeCherney reports small holdings with Lifecore Biomedical and Johnson & Johnson. Drs. Hurd and Pagidas report no financial relationship with any companies whose products are mentioned in this article. Dr. Sanfilippo serves on the speaker’s bureau for Berlex, Ortho Pharmaceuticals, and Wyeth, and receives grant support from Eli Lilly and Wyeth.


-

-

- Approximately 40% of people who undergo primary surgery develop adhesions and reformation occurs in 80% to 90% of cases.
- Microsurgical techniques such as gentle handling of tissues, careful hemostasis, and avoidance of heat may help reduce the incidence.
- Laparoscopy appears to be less likely to produce adhesions than laparotomy.
- Ob/Gyns should be aware of the potential for adhesion-related bowel obstruction and take steps to prevent it.
Are adhesions a pathologic response to injury or a normal aspect of healing? Can they be avoided, or are preventive efforts part of the problem? How useful are the different barriers in gynecologic surgery? What is the ideal adjuvant?
OBG Management convened a panel of experts to explore these and other questions.
Common problem, high recurrence rate
DECHERNEY: Adhesion formation is serious because it is associated with clinical entities such as infertility, pelvic pain, and bowel obstruction. We all agree that approximately 40% of people who undergo primary surgery develop adhesions and that 80% to 90% of patients who undergo lysis develop recurrent adhesions.
SANFILIPPO: One study several years ago explored adhesion formation.1 Unfortunately, no matter how meticulous the surgeon is, adhesions will form, even with microsurgical techniques and carefully ensured hemostasis.
HURD: The number of patients with significant adhesion formation after some gynecologic procedures has been reported to be greater than 90%.2
DECHERNEY: That higher incidence usually occurs after general surgery—and there’s a reason it is so high: General surgeons don’t use adjunctive therapy. They are critical of it. It is to our credit as gynecologic surgeons that we adopted adjunctive therapies about 15 years ago with the introduction of dextran 70 (Hyskon; Medisan Pharmaceuticals, Parsippany, NJ).
PAGIDAS: If anything, the pelvis seems to have even more of a predilection for adhesion formation than the abdomen, probably because of the close proximity of structures.
How and why adhesions form
DECHERNEY: What is the pathophysiology of adhesion formation? Let’s say you have 2 raw surface areas. What happens?
The process
PAGIDAS: The increase in leukotrienes and prostaglandins and the decrease in plasminogen activity (which actually initiates the inflammation) appear to be significant.
HURD: Vessel permeability also increases, and inflammatory cells leak through the vessels and set up a matrix for adhesion formation.
DECHERNEY: So we have 2 raw surface areas with fibrin leaking out and forming bridges between them.
PAGIDAS: The key is that it takes 2 surfaces to form these bridges. As I mentioned, the greater proximity of pelvic structures—particularly around the tube and ovary—probably contributes to adhesion formation.
DECHERNEY: Macrophage activity also is important. The macrophage “migrates” along these fibrin bridges and lays down collagen over a period of time. Then the collagen becomes organized and, eventually, vascular.
Window of opportunity
SANFILIPPO: Adhesion formation probably occurs and is pretty well established within 5 to 7 days of the precipitating event—usually surgery. Once that process is under way, attempts to halt it yield diminishing returns. Unfortunately, we don’t know how to interfere with it in a positive way.
HURD: Under normal conditions, there seems to be a balance between fibrin deposition and fibrinolysis. In some tissues, however, these functions become imbalanced. This disparity may contribute more to adhesions than the actual laying down of fibrin—especially in tissue that is hypoxic.
DECHERNEY: Would you say that adhesion formation represents normal or abnormal healing?
HURD: It is one of the body’s normal protective mechanisms and an important part of healing. Without it, any abdominal injury would likely result in death.
SANFILIPPO: I don’t think it differs that much from processes that occur externally. For example, if you get cut deeply enough, you develop a scar. Is that scar part of the normal healing process? It is.
PAGIDAS: Right. It is a normal process of tissue remodeling. The question is: What allows it to go astray?
What surgical techniques help prevent adhesions?
DECHERNEY: Let’s review the aspects of surgical technique that are important for adhesion prevention.
PAGIDAS: I emphasize the value of microsurgical techniques, which help to minimize severe tissue handling. It also is important to keep surfaces moistened so they don’t desiccate.
SANFILIPPO: I agree with Dr. Pagidas about microsurgical techniques such as gentle tissue handling, careful hemostasis, and keeping tissues moist. If we follow these principles, we create an environment that minimizes the potential for adhesion formation.
HURD: The findings of many well-controlled animal studies have been surprising. For example, it is difficult to demonstrate that drying of tissue increases adhesions.3 Probably the greatest contributor to adhesions in these models was abrasion.4 One way that laparoscopic surgery decreases adhesions is by avoiding abrasion of the bowel mucosa, which occurs specifically with packing.
A 38-year-old mother of 2 undergoes myomectomy for menorrhagia.
SANFILIPPO: The initial question is: Can this case be managed laparoscopically? I do myomectomies laparoscopically whenever possible, although I do close the uterus with a minilaparotomy incision. The reason is my strong concern about reapproximating the myometrium, since wound dehiscence sometimes occurs at the site of myoma removal.
In this case, depending on the size of the myomas, I would do as much as possible laparoscopically and then reapproximate the myometrium. I would plan my incisions carefully, to maximize the number of myomas that can be removed. I would end with meticulous hemostasis and, assuming it is successful, use a barrier over the incision—in this case, Interceed.
HURD: Does the patient desire future childbearing? If so, I would avoid the laparoscopic approach because of the possibly increased risk of uterine rupture during pregnancy. If she isn’t planning pregnancy, there are more options.
The next question is: How many myomas are there, and where are they located? If they are intrauterine, a hysteroscopic approach would avoid extrauterine adhesions. If they are multiple and large, I am pretty much limited to laparotomy. If there is 1 or only a few myomas, a laparoscopic approach would be best.
I have not used Interceed. In laparoscopic cases, I worry that it would create more problems because, as you allow the carbon dioxide to decrease at the end of a case, oozing begins. Instead of a barrier, I would use limited hydroflotation.
SANFILIPPO: That’s a good point. At the end of the myomectomy, with the laparoscope in place, I decrease the insufflation, eliminating the tamponade effect. Then, assuming good hemostasis, I apply Interceed.
HURD: With open cases, I use Seprafilm, which takes practice because, as it gets wet, it sticks to anything, including gloves and instruments. But if you can put it down dry on the uterus, it sticks and stays in place. If oozing occurs, it seems to block or stop it.
PAGIDAS: I want to reiterate the importance of determining whether childbearing is an issue. In this case, the biggest concern is the risk of adhesions developing on our incision, so I would use a barrier. My preference would be Seprafilm or GoreTex. If we can limit adhesions at the incision site, then hopefully we can minimize bowel and tuboovarian adhesions, too.
In open cases, one thing we can do to minimize the risk of adhesions is to pack gently when needed. Also, we should avoid using packing to reposition the bowel.
Another factor frequently overlooked is the application of heat, which appears to be a very effective way to create adhesions. This probably isn’t an issue for laparoscopic cases, but when you use irrigation fluid in an open case, watch the temperature. If it feels hot to you, you need to worry about potential injury to the bowel surfaces.
PAGIDAS: That is critical. In abdominal cases you want to make sure irrigation fluid is warm, but not too warm, because heat increases the vascular permeability of vessels and leads to more macrophages, more prostaglandins, and more leukotrienes.
HURD: Another important element is the type of suture material used.
DECHERNEY: Overall, we need to minimize the use of sutures. For example, when I am operating laparoscopically on an ovarian cyst, I try to apply bipolar energy to the edges so that they will coapt without a stitch.
HURD: When it first became clear that suturing ovaries increased adhesion formation, we conducted a controlled trial of different kinds of sutures in animals. Not surprisingly, we found that the less reactive the suture, the fewer adhesions.5 Sutures that are absorbed more slowly, such as polydioxanone, seem to be less reactive.
Obviously, inert sutures like nylon are the least reactive, but they are permanent. It is assumed that animal-protein sutures such as chromic and plain gut are the most reactive, although I am not sure there are sufficient data to support that conclusion.
Multiple clinical studies have shown laparoscopy to be associated with a lower adhesion rate, although it isn’t clear why. It may be related to decreased suturing.
DECHERNEY: Bulk is important, too—that is, the number of throws in the suture. When Vicryl (polyglactin 910) became available, we conducted a study in mice using proportionately small Vicryl plaques to determine whether this would be good a barrier (A. DeCherney, MD, unpublished data). It caused a tremendous amount of adhesion because so much foreign matter was applied.
We also did a study using human-size titanium clips in rats (A. DeCherney, MD, unpublished data). Not surprisingly, there was a lot of adhesion formation.
FIGURE 1 Pelvic adhesions: How they develop, problems they cause
Adhesions occur when 2 or more raw surfaces are exposed to leaking fibrin, which forms a bridge between the surfaces. Macrophages “migrate” along these bridges, depositing collagen.
The pelvis has a greater predilection for adhesions than the abdomen because of the close proximity of structures.
Although adhesions represent one of the body’s protective mechanisms, they may also cause pain or interfere with fertility, bowel function, or other processes.
Laparoscopy versus laparotomy: More adhesions in open cases?
DECHERNEY: Based on all the techniques we have learned from microsurgery—with the exception of magnification—it appears that laparoscopic procedures are less likely to cause adhesions than laparotomy. Do you agree?
PAGIDAS: I think so. As Dr. Hurd noted, a main reason is the diminished tissue handling, because there is no packing.
HURD: Multiple clinical studies have shown laparoscopy to be associated with a lower adhesion rate, although it isn’t clear why.6,7 It may be related to decreased suturing.
DECHERNEY: Less bleeding occurs because surgeons are less aggressive laparoscopically than in laparotomy.
A 15-year-old undergoes removal of a dermoid cyst, which was shelled out laparoscopically.
PAGIDAS: This case is easier because the cyst has been successfully shelled out. It is not the spill of a cyst’s contents at surgery that creates adhesions, but a chronic leak, which can occur if you do not remove the cyst in its entirety.
Once the cyst has been excised completely, I would ensure hemostasis with bipolar cautery and reapproximate the edges. I would not suture. There seems to be no clear advantage to suturing. I would use hydroflotation. Although Ringer’s lactate solution has not been shown to be effective, it is safe and has no toxicity.
DECHERNEY: Would you remove the cyst via laparotomy?
PAGIDAS: I would do it laparoscopically, using the endobag to minimize spillage, even though we know that a spill doesn’t necessarily change the outcome. If spillage does occur, I would perform copious irrigation to ensure that nothing is left behind.
HURD: My priority would be minimizing the use of power on the ovary. Studies of ovarian drilling have demonstrated that burning an ovary stimulates adhesion formation.21,22 If the dermoid cyst spills, as happens occasionally, I perform copious rinsing until no more oil is visible on the surface of the peritoneal fluid.
I also would minimize the amount of ovarian capsule that is removed. Good studies of endometriomas have shown that the more capsulate that is removed, the more adhesions. Even if the capsule looks redundant and floppy, the concern should be to achieve hemostasis with bipolar cautery and then leave it alone.
DECHERNEY: Would you use crystalloids in this case?
HURD: Yes.
SANFILIPPO: If spillage occurs, I would ensure that the patient is taken out of the Trendelenburg position. I want to emphasize the importance of thorough irrigation to eliminate any material that could produce chemical peritonitis.
DECHERNEY: Over the years, I have seen a fair number of cases of Fitz-Hugh and Curtis syndrome. You rigorously lavage a ruptured dermoid cyst, which sometimes presents with low-grade fever, but always with pain.
I’m surprised that none of you would use Interceed, since wrapping the ovary is the only thing for which it has been clearly shown to be effective. Since the cortex is relatively avascular, you don’t get a lot of bleeding. Unfortunately, it is not technically easy to wrap the ovary.
HURD: Since we do not know the effect on future fertility of changing the ovarian surface, less would seem to be better in patients this young.
I have stopped doing difficult cases laparoscopically. For example, it is rare for me to operate laparoscopically on a patient with stage IV endometriosis, at least when it comes to infertility—I might consider laparoscopy for pain.
I think case selection plays a role as well, although there are few data to back that up. It is purely clinical opinion.
SANFILIPPO: We need a well-designed prospective study to explore the effects of laparotomy versus laparoscopy. Existing data are not clear. You would assume laparoscopy would be associated with less adhesion formation. But genetic or other factors may explain why patient A is more prone to adhesions than patient B.
Does anybody think carbon dioxide plays a role in adhesion formation?
HURD: In the laboratory, carbon dioxide increases cell growth.8 Without an increased carbon dioxide concentration in the atmosphere, cell cultures don’t grow well. This might suggest that the carbon dioxide used for laparoscopy could actually enhance adhesion growth. Fortunately, this does not appear to be the case clinically.
With myomectomy, the surgeon needs to plan ahead to maximize the number of myomas removed from a single incision.
DECHERNEY: What about second-look laparoscopies? Do any of you perform them after a patient has undergone lysis of adhesions?
SANFILIPPO: Only as part of a research protocol. It amazes me how rapidly adhesions can form and how dense they are 2 or more weeks after the initial laparoscopic surgery.
PAGIDAS: We tend to limit second-look laparoscopy to a research protocol, although it is sometimes valuable after laparoscopic or abdominal myomectomy, which has the highest incidence of adhesions. If the surgeon can perform a second look and lyse adhesions, he or she may potentially alter the reproductive outcome. However, with assisted reproductive techniques becoming integral to every infertility case, that approach has begun to go out of style.
SANFILIPPO: That’s a good point. With myomectomy, the surgeon needs to plan ahead to maximize the number of myomas removed from a single incision. If adhesions do occur, it is best if they occur toward the bladder rather than in the area of the tubes and ovaries.
HURD: For second-look laparoscopy, we must keep in mind the cost and the small but real risks of surgery. Until good controlled studies show a reasonable clinical advantage, this approach probably should remain a research protocol.
SANFILIPPO: The literature suggests it is helpful, but does not help fertility, so second-look laparoscopy is used mainly to evaluate adjunctive therapies. I don’t think anybody uses it as a primary therapy anymore.
Bowel obstruction still a risk, though rarely seen by Ob/Gyns
DECHERNEY: Although bowel obstruction is fairly common, Ob/Gyns do not often encounter it because it occurs relatively distant from the index surgery. Even though a patient may not experience bowel obstruction in the first year, an obstruction related to the index surgery is just as likely to develop 20 years later as 2 years later. These patients usually are treated by general surgeons. Still, we should beware of the potential for bowel obstruction and take steps to prevent it, if at all possible. Do you agree?
PAGIDAS: Yes. We tend to forget about bowel obstruction because we rarely follow patients past pregnancy or the first trimester if they are seeking infertility treatment.
HURD: The primary problem seems to be the abdominal wall incision. Fortunately, cesarean section seems to carry a decreased risk of abdominal wall adhesions, probably because the uterus serves as a splint over the incision.
The Pfannenstiel incision also appears to have some advantage. Both human and animal models suggest little advantage or disadvantage when peritoneal closure is compared to nonclosure.9,10
SANFILIPPO: I’m curious about how the panelists manage loose clips. If you are using an EndoGIA (US Surgical, Norwalk, Conn) or other stapling device and you have free-floating clips, do you make a concerted effort to find them? In some cases, they have been implicated in bowel adhesion and obstruction. I try to retrieve loose clips, whether open or closed.
HURD: The advantage of those devices is minimal tissue damage, and the clips are inert. In general, inert, nonreactive clips have not been implicated as much in adhesion formation. I retrieve them if I see them, but I don’t search them out.
PAGIDAS: I do the same. If the clips are visible, I remove them. But I would not repack the bowel or do anything more heroic than look in locations where they might be.
What drugs may inhibit inflammatory response?
DECHERNEY: What about use of pharmacologic agents to prevent adhesions? Is there reason to think research should focus on inhibiting the inflammatory response? How important is polymorphonuclear cell infiltration?
Cyclooxygenase (COX) 2 agents could be helpful for inhibition of platelet function, since they are low in side effects. Thus, high doses of these drugs might be effective. At one time, aspirin was proposed, but you’d have to give a human so much aspirin that her ears would ring.
PAGIDAS: Pharmacologic agents have a role, especially for dampening the inflammatory immune response. But you don’t want to dampen it completely because, as we observed, it is an important part of healing. The difficulty is finding a balance between allowing the tissue to heal and preventing adhesions.
HURD: We studied the ability of a water-soluble prostaglandin inhibitor to prevent postoperative adhesion formation. Like many other agents, we found only a partial response.11
DECHERNEY: With current options, the best you can aim for is a 50% reduction.
Adjunctive therapy likely to limit adhesion rate Hydroflotation
DECHERNEY: The original adjunctive therapy was 20 mg dexamethasone and 25 mg femergin in 200 cc of Ringer’s lactate, with an equivalent amount of dexamethasone and femergin every 4 hours for a total of 6 doses. I prescribed that regimen because I was trained to do so. I stopped after it became clear that hydroflotation from the fluid—not the medication itself—was responsible for the improvement.
I must admit I gave it up reluctantly; patients felt fabulous with those higher load doses of glucocorticoids after surgery.
Do any of you use crystalloids as adjunctive therapy?
SANFILIPPO: In the animal model, they are so rapidly absorbed that they aren’t effective. I was a strong advocate, but now I don’t use them at all.
HURD: A lot depends on the kind of case. For instance, at the end of an open myomectomy, the patient often is oozing, so you want to use a barrier that blood won’t affect.
For ovarian surgery, you might want to specifically target the ovaries with some kind of coverage. But when you are doing a broad lysis of adhesions, you have few choices to cover the pelvis. In those cases I use hydroflotation with Ringer’s lactate. Both human and animal studies have shown some benefit in preventing adhesions, and it appears to have little risk.12,13
It’s better than nothing, in my opinion.
DECHERNEY: Do you use dextran 70 or crystalloids?
HURD: I use Ringer’s lactate solution. I was trained in the dextran 70 era, and there were certain problems with that approach. Since studies have failed to show a consistent effect of dextran 70, I no longer use this solution.14,15
DECHERNEY: Another problem with crystalloids is that they leak, which is disconcerting to the patient.
HURD: They also can mask an injury to the bladder in difficult cases.
DECHERNEY: I agree that dextran 70 is only appropriate in certain cases, but it is a good hydroflotation agent. Every cubic centimeter of dextran 70 brings in 1.2 cc of transudate, so it hangs around for at least 4 days.
It is appropriate only for certain surface areas—mainly the cul-de-sac. It is harmful on raw surface areas on the lateral pelvic sidewall because it tends to push the ovary and tube to those areas. Unless you are doing a lot of work in the deep pelvis, I would avoid dextran 70.
A 45-year-old woman undergoes an abdominal hysterectomy. The cuff is closed and the ovaries are intact.
PAGIDAS: I would do nothing other than ensure adequate hemostasis, check that I have left no round surfaces and, probably, use hydroflotation. I see no advantage to barriers.
HURD: One of the main causes of adhesions is devascularized tissue, and the perfect devascularized tissue might be the vaginal cuff. Re-“peritonealizing” the cuff might be advantageous. Thus, I would use minimal sutures—probably a slowly absorbable, light polydioxanone suture to place the peritoneum over the cuff so there are no pedicles.
DECHERNEY: All the pedicles are exteriorized.
HURD: Yes, that could be. We don’t bring all of it down like we used to years ago, but we do cover the cuff.
PAGIDAS: I agree that closing the cuff and reperitonealizing may actually minimize formation of hematomas—clearly an advantage.
SANFILIPPO: I agree. I guess I’m old fashioned. If it looks good, then hopefully it will stimulate less adhesion formation, so peritonealization is important.
As far as the abdominal incision is concerned, I would not close that peritoneum. I’m convinced now that there is no advantage.
DECHERNEY: Reperitonealizing the cuff is controversial. Most gynecologic surgeons do not do it, the theory being that the peritoneum is being stretched, attenuating the vessels that go through it and thereby creating an ischemic barrier that contributes to adhesions. Personally, I like to do it because it looks better—and that is certainly an important aspect of a surgery. No evidence shows that it is bad or good, either way.
In addition, there have been allergic reactions, most of which seem to occur in patients with fluid overload; a lot of the dextran 70 is absorbed.
With infertility patients, even if you lyse dense adhesions, you do not render the ovarian surface normal.
SANFILIPPO: Dextran 70 is not recommended for patients with sugar beet allergy, either.
We completed a study in a rabbit model, in which the peritoneal cavity was lavaged with either chlorhexidine or iodine.16 At the time of second-look surgery, the rate of adhesion formation was decreased, especially with the iodine preparation. I would hope that this has potential in humans.
DECHERNEY: In your lavage procedures to prevent adhesions, do any of you use heparin?
HURD: No.
PAGIDAS: I don’t think any evidence suggests that local administration changes the outcome.
DECHERNEY: I agree. When heparin has been used, the doses have been so low that it was not terribly helpful. And when you consider that hemorrhage can be a problem, heparin is probably deleterious rather than helpful.
Barriers
DECHERNEY: What about barriers? The first to become available, Interceed (Gynecare, a division of Ethicon, Somerville, NJ), is oxidized cellulose, similar to Surgicell (Johnson & Johnson, New Brunswick, NJ). Since it gelates quickly, there is no fenestration, so the fibrin is unable to penetrate. However, if the patient has bleeding by capillary action, the raw surface just moves from one side of the Interceed barrier to the other.
What has been your experience? Do you use it?
PAGIDAS: I do not use Interceed, although prospective randomized trials and a meta-analysis confirmed its benefits in de novo formation and reformation.17-19 I don’t use it because it requires complete hemostasis. Also, with the surfaces we work on—notably, the ovary and tube—it is difficult to apply to just 1 surface area. From a clinical perspective, I appreciate the data, but it is hard to ensure a good application to optimize its effectiveness.
A 29-year-old woman is undergoing her third cesarean section, although adhesions cause difficulty getting through anteriorly.
DECHERNEY: In this case, will you use barriers or re-peritonealize the surface? Will you do a 1- or a 2-layered closure?
HURD: In these cases, I have not been doing anything, since there is no peritoneum to reperitonealize—just old scar tissue. I assume that the uterus will immediately readhere to the anterior peritoneum where it was before.
DECHERNEY: You would use a barrier?
HURD: No. I would not.
PAGIDAS: I would take the same approach, although I have not performed cesarean sections in about 4 years. I don’t think any intervention would change the outcome. And, as Dr. Hurd mentioned, that is pretty much old scar tissue anyway.
SANFILIPPO: I use Interceed, but I agree with you about its limitations. Meticulous hemostasis is a prerequisite.
HURD: With infertility patients, even if you lyse dense adhesions, you do not render the ovarian surface normal. If those patients have dense adhesions of the ovary or the sidewall, I generally leave them alone, and I try to avoid putting Interceed around the ovaries. No study has shown that using Interceed improves pregnancy.
In contrast, when a chronic pain patient’s ovaries are densely adherent to the cul-de-sac, which appears to be highly associated with dyspareunia, I lyse the adhesions, achieve meticulous hemostasis, and then use Interceed. It is hard to demonstrate in a study that this approach decreases the chance of pain. Even so, it certainly does decrease the chance of the ovaries being adherent.
DECHERNEY: One issue with Interceed is that we don’t know what happens to it once the abdomen is closed. It may migrate significantly.
Psychological issues may also be involved. For example, patients with multiple somatic complaints may be less likely to benefit from lysis of adhesions.
PAGIDAS: Right. Interestingly, a meta-analysis of all the randomized trials involving mechanical barriers found no correlation to pregnancy outcome or pelvic pain.18 If we were to consider new trials, the psychological aspect would be worth looking into.
Seprafilm
DECHERNEY: Let’s move on to Seprafilm (Genzyme, Cambridge, Mass). What is it and how useful is it?
HURD: Seprafilm is modified hyaluronic acid, which forms a brittle, thin plastic layer. It is somewhat difficult to work with but, once it is in place, seems to adhere well. The presence of blood does not appear to be a problem, since the Seprafilm forms an impermeable barrier—unless it breaks. I have found it especially useful in myomectomies, which produce postoperative oozing through the incisions no matter how hard you try to prevent it.
In addition, in open cases, surfaces can easily be covered with this material. Unfortunately, it can’t be used laparoscopically because it is so brittle.
DECHERNEY: In my opinion, that is its major drawback.
PAGIDAS: In cardiac surgery, Seprafilm appears to work quite effectively.
SANFILIPPO: The manufacturer initially focused on surgeons in the context of sigmoid colon surgery, and it seems to work well in that setting.
Intergel
DECHERNEY: That brings us to the current state of the art: gels. I’m sure you all are familiar with Intergel (Lifecore Biomedical, Chaska, Minn), which is a ferrous derivative of hyaluronic acid that works by coating the raw surface areas. It also has the theoretical advantage of ease of use. Have any of you used Intergel?
HURD: As you know it was only recently approved by the US Food and Drug Administration, but not for laparoscopic use. It may work best on abraded bowel, which is avoided by laparoscopic surgery.
As you are probably also aware, the manufacturer recently took it off the market because of unusual side effects, namely a chemical peritonitis. Although peritonitis was cited as being rare, we encountered it in probably half the patients we operated on.
PAGIDAS: When we used it on hospitalized patients, the peritonitis wasn’t that obvious, since there was an expectation of significant pain. However, when we used Intergel on short-stay patients, we had to readmit them and do a full workup because we were concerned about bowel perforations. I’m surprised the manufacturer didn’t take it off the market sooner.
DECHERNEY: It seems strange, since Intergel has been used in Europe for a while now. I’ve used it in only 1 case and didn’t have adverse effects. It seemed to work well.
SANFILIPPO: It had all the right ingredients for success. It is unfortunate that these side effects have prohibited its use.
There is no question that adhesion prevention is one of the unmet challenges in all surgeries, especially reproductive surgery.
Gels and the cost factor
DECHERNEY: Other gels are in the pipeline. I’m reminded of plasminogen activator, which is a powerful antiadhesive agent that lyses fibrin effectively. Unfortunately, it is prohibitively expensive.
The next phase likely will involve the so-called polymers. If you spray them on your hand, they are activated by light or another chemical and become a cellophane-like substance. The problem is viscosity. If sprayed on the sidewall, for instance, they run halfway down before they are activated, so the entire surface does not get covered.
PAGIDAS: One concern with polymers is that they could actually bring surfaces together when they polymerize. We still have a lot to learn.
DECHERNEY: Let’s say a new gel comes on the market that takes reformation adhesions from 90% to 10%, as opposed to 40% recurrence. Would you use it in all 4 of the cases we discuss here?
HURD: If it was that effective and had no adverse effects, it would be wonderful.
The cesarean-delivery case is different, as healing in a pregnant patient is 1 concern; the size of the uterus also has an effect. If the patient is breastfeeding, you would want to make sure the gel didn’t interfere.
PAGIDAS: We desperately need a product that can minimize adhesions regardless of the route of access or type of procedure. Even though we lack data on outcomes, I predict wide use of such a product, assuming it is nontoxic and effective.
DECHERNEY: What if it costs $1,000 a case?
SANFILIPPO: If it prevents 1 bowel obstruction, it still would be cost-effective.
DECHERNEY: The incidence of bowel obstruction for total abdominal hysterectomy is 2%, and 5% for radical hysterectomy.
HURD: We must be careful of the cost-benefit ratio. Bowel obstruction after gynecologic surgery is uncommon.
Is the gel worth $100? $1,000? $3,000? It’s difficult to say, but the more expensive it is, the less likely it will find widespread use.
PAGIDAS: I agree. We should remember that we still need to maintain microsurgical techniques and appropriate tissue handling, as well as avoid ischemia and infection.
Looking for the magic bullet
DECHERNEY: What is the future of adjunctive therapy?
SANFILIPPO: I would focus on noxythiolin; it has potential. Calcium channel blockers for adhesion prevention have also been studied.20 In 1 investigation involving a rat model, the calcium channel blocker verapamil as well as several other agents—including vitamin E, carboxymethylcellulose, cyclosporin, aprotinin, and tenoxicam—were compared with respect to tissue effects. A beneficial effect was noted with all agents except cyclosporin and carboxymethylcellulose.
Whoever succeeds in manufacturing an effective preventive will be a winner.
HURD: There is no question that adhesion prevention is one of the unmet challenges in all surgeries, especially reproductive surgery. The most effective agent would be applied intraabdominally, since any systemic agent that stops adhesion formation would probably decrease wound healing as well.
I hope the most effective agents can be used in both laparoscopy and laparotomy, and that they will decrease the adhesion-formation rate by more than 50%. We need to find the magic bullet that can cover the entire pelvis—if not the entire abdomen.
Dr. DeCherney reports small holdings with Lifecore Biomedical and Johnson & Johnson. Drs. Hurd and Pagidas report no financial relationship with any companies whose products are mentioned in this article. Dr. Sanfilippo serves on the speaker’s bureau for Berlex, Ortho Pharmaceuticals, and Wyeth, and receives grant support from Eli Lilly and Wyeth.
1. Operative Laparoscopy Study Group Postoperative adhesion development after operative laparoscopy: evaluation at early second look procedures. Fertil Steril. 1991;55:700-704.
2. Tulandi T, Murray C, Guralnick M. Adhesion formation and reproductive outcome after myomectomy and second-look laparoscopy. Obstet Gynecol. 1993;82:213-215.
3. Larsson B, Perbeck L. The possible advantage of keeping the uterine and intestinal serosa irrigated with saline to prevent intraabdominal adhesions in operations for infertility. An experimental study in rats. Acta Chir Scand Suppl. 1985;530:15-18.
4. Zamir G, Bloom AI, Reissman P. Prevention of intestinal adhesions after laparotomy in a rat model—a randomized prospective study. Res Exp Med (Berl). 1998;197:349-353.
5. Hurd WW, Himebaugh KS, Cofer KF, Gauvin JM, Elkins TE. The etiology of closure-related adhesion formation after wedge resection of the rabbit ovary. J Reprod Med. 1993;38:465-468.
6. Chen MD, Teigen GA, Reynolds HT, Johnson PR, Fowler JM. Laparoscopy versus laparotomy: an evaluation of adhesion formation after pelvic and paraaortic lymphadenectomy in a porcine model. Am J Obstet Gynecol. 1998;178:499-503.
7. Polymeneas G, Theodosopoulos T, Stamatiadis A, Kourias E. A comparative study of postoperative adhesion formation after laparoscopic vs. open cholecystectomy. Surg Endosc. 2001;15:41-43.
8. Smidt VJ, Singh DM, Hurteau JA, Hurd WW. Effect of carbon dioxide on human ovarian carcinoma cell growth. Am J Obstet Gynecol. 2001;185:1314-1317.
9. Tulandi T, Hum HS, Gelfand MM. Closure of laparotomy incisions with or without peritoneal suturing and second look laparoscopy. Am J Obstet Gynecol. 1988;158:536-537.
10. Kapur ML, Daneswar A, Chopra P. Evaluation of peritoneal closure at laparotomy. Am J Surg. 1979;137:650-652.
11. Cofer KF, Himebaugh KS, Gauvin JM, Hurd WW. Inhibition of adhesion reformation in the rabbit model by meclofenamate: an inhibitor of both prostaglandin and leukotriene production. Fertil Steril. 1994;62:1262-1265.
12. Elkelani OA, Molinas CR, Mynbaev O, Koninckx PR. Prevention of adhesions with crystalloids during laparoscopic surgery in mice. J Am Assoc Gynecol Laparosc. 2002;9:447-452.
13. Chan KL, Marino T, Qu WM, Tulandi T. Effects of intraperitoneal Ringer’s lactate instillation and infusion on postsurgical adhesion formation. J Gynecol Surg. 1995;11:241-243.
14. Watson A, Vandekerckhove P, Lilford R. Pharmacological adjuvants during infertility surgery: a systematic review of evidence derived from randomized controlled trial. Hum Fertil (Camb). 1999;2:149-157.
15. Larson B. Dextran—later clinical studies. clinical and experimental evaluation of different adjuvant therapies. Prog Clin Biol Res. 1990;358:165-175.
16. Roberts L, Sanfilippo JS, Ehrlich G, Raab S. Adhesion formation, peritoneal effects and the presence of bacterial biofilms after laparoscopic lavage in an animal model of pelvic inflammatory disease. J Am Assoc Gynecol Laparosc. 2002;9:4.-
17. Barrier agents for preventing adhesions after surgery for subfertility Cochrane Database Syst Rev. 2000;CD000475.-
18. Wiseman DM, Trout JR, Franklin RR, Diamond MP. Metaanalysis of the safety and efficacy of an adhesion barrier (Interceed TC7) in laparotomy. J Reprod Med. 1999;44:325-331.
19. Sawada T, Nishizawa H, Nishio E, Kadowaki M. Postoperative adhesion prevention with an oxidized regenerated cellulose adhesion barrier in infertile women. J Reprod. Med. 2000;45:387-389.
20. Uzunkoy A, Akinci OF, Coskun A, Aslan O, Kocyigit A. Effects of antiadhesive agents on the healing of intestinal anastomosis. Dis Colon Rectum. 2000;43:370-375.
21. Farquhar C, Vandekerckhove P, Arnot M, Lilford R. Laparoscopic “drilling” by diathermy or laser for ovulation induction in anovulatory polycystic ovary syndrome. Cochrane Database Syst Rev. 2000(2);CD001122.-
22. Greenblatt EM, Casper RF. Laparoscopic ovarian drilling in women with polycystic ovarian syndrome. Prog Clin Biol Res. 1993;381:129-138.
1. Operative Laparoscopy Study Group Postoperative adhesion development after operative laparoscopy: evaluation at early second look procedures. Fertil Steril. 1991;55:700-704.
2. Tulandi T, Murray C, Guralnick M. Adhesion formation and reproductive outcome after myomectomy and second-look laparoscopy. Obstet Gynecol. 1993;82:213-215.
3. Larsson B, Perbeck L. The possible advantage of keeping the uterine and intestinal serosa irrigated with saline to prevent intraabdominal adhesions in operations for infertility. An experimental study in rats. Acta Chir Scand Suppl. 1985;530:15-18.
4. Zamir G, Bloom AI, Reissman P. Prevention of intestinal adhesions after laparotomy in a rat model—a randomized prospective study. Res Exp Med (Berl). 1998;197:349-353.
5. Hurd WW, Himebaugh KS, Cofer KF, Gauvin JM, Elkins TE. The etiology of closure-related adhesion formation after wedge resection of the rabbit ovary. J Reprod Med. 1993;38:465-468.
6. Chen MD, Teigen GA, Reynolds HT, Johnson PR, Fowler JM. Laparoscopy versus laparotomy: an evaluation of adhesion formation after pelvic and paraaortic lymphadenectomy in a porcine model. Am J Obstet Gynecol. 1998;178:499-503.
7. Polymeneas G, Theodosopoulos T, Stamatiadis A, Kourias E. A comparative study of postoperative adhesion formation after laparoscopic vs. open cholecystectomy. Surg Endosc. 2001;15:41-43.
8. Smidt VJ, Singh DM, Hurteau JA, Hurd WW. Effect of carbon dioxide on human ovarian carcinoma cell growth. Am J Obstet Gynecol. 2001;185:1314-1317.
9. Tulandi T, Hum HS, Gelfand MM. Closure of laparotomy incisions with or without peritoneal suturing and second look laparoscopy. Am J Obstet Gynecol. 1988;158:536-537.
10. Kapur ML, Daneswar A, Chopra P. Evaluation of peritoneal closure at laparotomy. Am J Surg. 1979;137:650-652.
11. Cofer KF, Himebaugh KS, Gauvin JM, Hurd WW. Inhibition of adhesion reformation in the rabbit model by meclofenamate: an inhibitor of both prostaglandin and leukotriene production. Fertil Steril. 1994;62:1262-1265.
12. Elkelani OA, Molinas CR, Mynbaev O, Koninckx PR. Prevention of adhesions with crystalloids during laparoscopic surgery in mice. J Am Assoc Gynecol Laparosc. 2002;9:447-452.
13. Chan KL, Marino T, Qu WM, Tulandi T. Effects of intraperitoneal Ringer’s lactate instillation and infusion on postsurgical adhesion formation. J Gynecol Surg. 1995;11:241-243.
14. Watson A, Vandekerckhove P, Lilford R. Pharmacological adjuvants during infertility surgery: a systematic review of evidence derived from randomized controlled trial. Hum Fertil (Camb). 1999;2:149-157.
15. Larson B. Dextran—later clinical studies. clinical and experimental evaluation of different adjuvant therapies. Prog Clin Biol Res. 1990;358:165-175.
16. Roberts L, Sanfilippo JS, Ehrlich G, Raab S. Adhesion formation, peritoneal effects and the presence of bacterial biofilms after laparoscopic lavage in an animal model of pelvic inflammatory disease. J Am Assoc Gynecol Laparosc. 2002;9:4.-
17. Barrier agents for preventing adhesions after surgery for subfertility Cochrane Database Syst Rev. 2000;CD000475.-
18. Wiseman DM, Trout JR, Franklin RR, Diamond MP. Metaanalysis of the safety and efficacy of an adhesion barrier (Interceed TC7) in laparotomy. J Reprod Med. 1999;44:325-331.
19. Sawada T, Nishizawa H, Nishio E, Kadowaki M. Postoperative adhesion prevention with an oxidized regenerated cellulose adhesion barrier in infertile women. J Reprod. Med. 2000;45:387-389.
20. Uzunkoy A, Akinci OF, Coskun A, Aslan O, Kocyigit A. Effects of antiadhesive agents on the healing of intestinal anastomosis. Dis Colon Rectum. 2000;43:370-375.
21. Farquhar C, Vandekerckhove P, Arnot M, Lilford R. Laparoscopic “drilling” by diathermy or laser for ovulation induction in anovulatory polycystic ovary syndrome. Cochrane Database Syst Rev. 2000(2);CD001122.-
22. Greenblatt EM, Casper RF. Laparoscopic ovarian drilling in women with polycystic ovarian syndrome. Prog Clin Biol Res. 1993;381:129-138.
When is episiotomy warranted? What the evidence shows
- A Cochrane Database review concluded that restrictive episiotomy utilization is preferable to routine utilization. The review of 6 randomized trials found no differences in vaginal or perineal trauma, dyspareunia, or urinary incontinence between patients with and without episiotomy. Patients who had an episiotomy had less risk of anterior perineal trauma but an overall greater risk of posterior perineal trauma and other complications.
- Shoulder dystocia, operative vaginal delivery, and a “short” perineal body have been presumed indications for episiotomy, although data are inadequate to support these claims.
- The rationale for routine prophylactic episiotomy is to protect the pelvic floor, thereby minimizing the risk of urinary incontinence and pelvic floor dysfunction; however, episiotomy’s role in preventing such dysfunction remains to be established.
Although a large body of evidence indicates reassessment is in order, prophylactic episiotomy is a contentious issue. Indeed, it has been controversial ever since the procedure first became “routine” in the United States, in 1920. Still, advocates and dissenters share the same goal: to prevent severe perineal tears and their potential for urinary and fecal incontinence and sexual dysfunction.
This article reviews research findings that indicate:
- Data are inadequate to recommend one method of episiotomy over another.
- Timing of episiotomy to shorten the second stage of labor may be less relevant in an era of decreasing forceps utilization and without evidence of improved neonatal outcomes.5
- Episiotomy, particularly midline episiotomy, remains the single greatest risk that a patient will sustain a third- or fourth-degree laceration. When such lacerations occur spontaneously, recovery is equivalent to episiotomy extension or deliberate proctoepisiotomy.
TABLE
Incidence of third- or fourth-degree laceration with and without episiotomy
| NO. STUDIES COMPILED | NO. PATIENTS | % WITH 3RD- OR 4TH-DEGREE LACERATION | |
|---|---|---|---|
| Midline episiotomy | 12 | 49,395 | 6.5 |
| No episiotomy | 13 | 38,961 | 1.4 |
| Adapted from Thorp JM.3 | |||
Research does not support presumed indications
Episiotomy was first described in 1742 as a procedure that could assist the obstetrician in difficult vaginal deliveries.3 It was not until the work of DeLee6 and Pomeroy7 was published in 1920—coincident with deliveries beginning to move from home to hospital—that the procedure became “routine.” Still, some leaders in the field—specifically, J. Whitridge Williams of Johns Hopkins—vigorously dissented.8
Historically, episiotomy has been used to facilitate delivery in cases of protracted second stage, instrumented vaginal delivery, and suspected fetal compromise. However, data supporting episiotomy as a facilitating procedure are sparse, and evidence endorsing prophylactic episiotomy is largely anecdotal or descriptive.
Agreement is widespread that episiotomy is warranted under certain circumstances: Shoulder dystocia, operative vaginal delivery, and a “short” perineal body have been presumed indications. Data are inadequate to support these claims, however.
Shoulder dystocia. While it might seem to make sense to perform an episiotomy (or more likely, a proctoepisiotomy) in cases of shoulder dystocia, no data from controlled trials support this theory. Given the relative rarity of severe shoulder dystocia and the inability to conduct a truly randomized trial, physicians are left with only their clinical judgment as a guide in this circumstance.
Operative delivery. Many clinicians have advocated routine episiotomy before operative vaginal delivery, particularly with forceps. The intent is to increase the space available for delivery that has been diminished by the introduction of forceps. This rationale does not hold up as well for vacuum extraction; 1 study noted that when episiotomy is performed in cases of vacuum extraction, the likelihood of severe perineal trauma is increased.9
It has been reported10 that the greatest risk factor for both perineal trauma and third- or fourth-degree perineal laceration is episiotomy itself (TABLE), independent of mode of delivery (spontaneous or operative).
Short perineum. Many physicians, myself included, have performed episiotomies because they perceived that the perineum was short and that even a controlled delivery with optimal use of the Ritgen maneuver probably would not prevent a perineal laceration. That said, data on anal and flatus incontinence and postpartum sexual functioning suggest that spontaneous recovery from second-degree lacerations is no worse than recovery from midline episiotomy11,12 and, as stated, episiotomy itself is the leading risk factor for incurring a third- or fourth-degree extension—which imposes significantly greater recovery problems.
Two recent studies11,12 identified episiotomy as a specific, independent risk factor for fecal incontinence and delayed return of sexual activity postpartum. When matched for degree of perineal trauma, episiotomy without extension still resulted in poorer outcomes at 3 and 6 months postpartum than did spontaneous second-degree lacerations, suggesting that routine episiotomy not only fails to prevent, but may actually increase risk of perineal injury and impaired function.
‘Prophylactic’ episiotomy is not preventive
Much debate has centered on optimal utilization of so-called prophylactic episiotomy. The intent of routine prophylactic episiotomy is to protect the pelvic floor, thus minimizing the risk of urinary incontinence and pelvic floor dysfunction. Data have suggested that absence of labor and cesarean delivery may protect against pelvic floor dysfunction; however, the role of episiotomy in preventing such dysfunction remains to be determined.
Cochrane Database review. This review1 found no differences in vaginal or perineal trauma, dyspareunia, or urinary incontinence between patients with and without episiotomy. Patients who had an episiotomy had less risk of anterior perineal trauma but an overall greater risk of posterior perineal trauma and other complications. The reviewers concluded that restrictive episiotomy utilization is preferable to routine utilization.
The reviewers selected a total of 6 randomized trials; these examined:
- restrictive versus routine use of episiotomy;
- restrictive versus mediolateral episiotomy;
- restrictive versus routine midline episiotomy; and
- midline versus mediolateral episiotomy.
Compared with routine use, restrictive episiotomy involved less posterior perineal trauma (relative risk [RR], 0.88; 95% confidence interval [CI], 0.84 to 0.920), less suturing (RR, 0.74; 95% CI, 0.71 to 0.77), and fewer healing complications (RR, 0.69; 95% CI, 0.56 to 0.85). Restrictive episiotomy was associated with more anterior perineal trauma (RR, 1.79; 95% CI, 1.55 to 2.07).
There was no difference in severe vaginal or perineal trauma (RR, 1.11; 95% CI, 0.83 to 1.50), dyspareunia (RR, 1.02; 95% CI, 0.90 to 1.16), urinary incontinence (RR 0.98; 95% CI, 0.79 to 1.20), or several pain measures.
Results for restrictive versus routine mediolateral and midline episiotomies were similar to the overall comparison.
Reviewers concluded that a policy of restrictive episiotomy appears to have several benefits over routine episiotomy: less posterior perineal trauma, less suturing, fewer complications, and no difference for most pain measures and severe vaginal or perineal trauma.
Risk of anterior perineal trauma with restrictive episiotomy was increased, however. Restrictive-use protocols, likely to be institution-specific, essentially curb episiotomy use by stating that the procedure should not be “routinely performed.” Instead, episiotomy is restricted to cases in which the clinician believes it is warranted. Examples of such situations include use of forceps, shoulder dystocia, and an estimated fetal weight above 4,000 g. As discussed, the data cannot support the value of episiotomy use even in these circumstances; however, simply discouraging routine episiotomy would effectively lower the rate to the desired 30% range.
Midline versus mediolateral incision. The most vocal debates focus on which type of episiotomy to perform and whether it should be performed earlier or later in the second stage of labor.
It has been proposed that by abandoning midline episiotomies in favor of the mediolateral technique, physicians can avoid injury to the sphincter and improve immediate birth outcome without compromising long-term function—though pros and cons of this approach are a subject of debate (see “Comparison of mid-line versus mediolateral episiotomy”).
Still, the data suggest that, when properly performed, median and mediolateral episiotomy have equivalent rates of satisfactory recovery,13 though the latter technique may require more technical skill for both its performance and repair.
Early versus late incision. Proponents argue that an episiotomy at the time the presenting part is crowning is “too little, too late.” They maintain that for the procedure to be truly protective, it should be utilized earlier in the second stage of labor.
Data are insufficient to confirm or refute the efficacy of early episiotomy. One would do well to remember, however, that early episiotomy was endorsed as a method to help shorten the second stage of labor when used in conjunction with prophylactic forceps delivery—a method that is now less prevalent in obstetric practice.
The category of obstetric provider—midwife, faculty, or private provider—may be the most reliable predictor of episiotomy. Interestingly, use of episiotomy increased in the 1920s as delivery moved from home to hospital and birth attendants shifted from midwives to physicians.
In a study of demographic variables and obstetric factors associated with episiotomy in spontaneous vaginal delivery, researchers examined 1,576 term, singleton, spontaneous vaginal deliveries in nulliparas. They found that midwives had the lowest episiotomy rate (21.4%), compared with residents and full-time faculty (33.3%) and private physicians (55.6%).15
After controlling for confounding factors with logistic regression, the authors determined that private practice provider was the strongest predictor of episiotomy, followed by faculty provider, prolonged second stage of labor, fetal macrosomia, and epidural analgesia.
The study concluded that the obstetric and demographic factors evaluated did not readily explain the link between type of provider and episiotomy rate. Numerous theories have been proposed, but factors that would clearly explain the differences have yet to be identified.
Does vaginal birth trauma cause pelvic floor dysfunction?
The relationship between vaginal birth trauma, irrespective of episiotomy, and pelvic floor dysfunction remains a topic of investigation. A recent report generated much interest in the potentially protective role of prophylactic cesarean section, particularly if performed prior to the onset of labor.14
Dr. Repke reports no financial relationship with any companies whose products are mentioned in this article.
1. Carroli G, Belizan J. Episiotomy for vaginal birth [Cochrane Review]. In: The Cochrane Library Issue 3, 2003. Oxford: Update Software.
2. Lede RL, Belizan JM, Caroli G. Is routine use of episiotomy justified? Am J Obstet Gynecol. 1996;174:1399-1402.
3. Thorp JM. Episiotomy. In: Repke JT, ed. Intrapartum Obstetrics. New York, NY: Churchill Livingstone; 1996;489-499.
4. Thacker SB, Banta HD. Benefits and risks of episiotomy: an interpretive review of the English language literature, 1860-1980. Obstet Gynecol Surv. 1983;38:322-338.
5. Eason E, Feldman P. Much ado about a little cut: is episiotomy worthwhile? Obstet Gynecol. 2000;95:616-618.
6. DeLee JB. The prophylactic forceps operation. Am J Obstet Gynecol. 1920;1:34-44.
7. Pomeroy RH. Shall we cut and reconstruct the perineum for every primipara? Am J Obstet Dis Women Child. 1918;78:211-219.
8. Taylor ES. Comment on episiotomy and third degree tears. Obstet Gynecol Surg. 1985;41:229.-
9. Robinson JN, Norwitz ER, Cohen AP, McElrath TF, Lieberman ES. Episiotomy, operative vaginal delivery and significant perinatal trauma in nulliparous women. Am J Obstet Gynecol. 1999;181:1180-1184.
10. Robinson JN, Norwitz ER, Cohen AP, McElrath TF, Lieberman ES. Epidural analgesia and the occurrence of third and fourth degree laceration in nulliparas. Obstet Gynecol. 1999;94:259-262.
11. Signorello LB, Harlow BL, Chekos AK, Repke JT. Midline episiotomy and anal incontinence: a retrospective cohort study. BMJ. 2000;320:86-90.
12. Signorello LB, Harlow BL, Chekos AK, Repke JT. Postpartum sexual functioning and its relationship to perineal trauma: a retrospective cohort study of primiparous women. Am J Obstet Gynecol. 2001;184:881-890.
13. Coats PM, Chan KK, Wilkins M, Beard RJ. A comparison between midline and mediolateral episiotomies. Br J Obstet Gynaecol. 1980;87:408-412.
14. Dietz HP, Bennett MJ. The effect of childbirth on pelvic organ mobility. Obstet Gynecol. 2003;102:223-228.
15. Robinson JN, Norwitz ER, Cohen AP, Lieberman E. Predictors of episiotomy use at first spontaneous vaginal delivery. Obstet Gynecol. 2000;96:214-218
- A Cochrane Database review concluded that restrictive episiotomy utilization is preferable to routine utilization. The review of 6 randomized trials found no differences in vaginal or perineal trauma, dyspareunia, or urinary incontinence between patients with and without episiotomy. Patients who had an episiotomy had less risk of anterior perineal trauma but an overall greater risk of posterior perineal trauma and other complications.
- Shoulder dystocia, operative vaginal delivery, and a “short” perineal body have been presumed indications for episiotomy, although data are inadequate to support these claims.
- The rationale for routine prophylactic episiotomy is to protect the pelvic floor, thereby minimizing the risk of urinary incontinence and pelvic floor dysfunction; however, episiotomy’s role in preventing such dysfunction remains to be established.
Although a large body of evidence indicates reassessment is in order, prophylactic episiotomy is a contentious issue. Indeed, it has been controversial ever since the procedure first became “routine” in the United States, in 1920. Still, advocates and dissenters share the same goal: to prevent severe perineal tears and their potential for urinary and fecal incontinence and sexual dysfunction.
This article reviews research findings that indicate:
- Data are inadequate to recommend one method of episiotomy over another.
- Timing of episiotomy to shorten the second stage of labor may be less relevant in an era of decreasing forceps utilization and without evidence of improved neonatal outcomes.5
- Episiotomy, particularly midline episiotomy, remains the single greatest risk that a patient will sustain a third- or fourth-degree laceration. When such lacerations occur spontaneously, recovery is equivalent to episiotomy extension or deliberate proctoepisiotomy.
TABLE
Incidence of third- or fourth-degree laceration with and without episiotomy
| NO. STUDIES COMPILED | NO. PATIENTS | % WITH 3RD- OR 4TH-DEGREE LACERATION | |
|---|---|---|---|
| Midline episiotomy | 12 | 49,395 | 6.5 |
| No episiotomy | 13 | 38,961 | 1.4 |
| Adapted from Thorp JM.3 | |||
Research does not support presumed indications
Episiotomy was first described in 1742 as a procedure that could assist the obstetrician in difficult vaginal deliveries.3 It was not until the work of DeLee6 and Pomeroy7 was published in 1920—coincident with deliveries beginning to move from home to hospital—that the procedure became “routine.” Still, some leaders in the field—specifically, J. Whitridge Williams of Johns Hopkins—vigorously dissented.8
Historically, episiotomy has been used to facilitate delivery in cases of protracted second stage, instrumented vaginal delivery, and suspected fetal compromise. However, data supporting episiotomy as a facilitating procedure are sparse, and evidence endorsing prophylactic episiotomy is largely anecdotal or descriptive.
Agreement is widespread that episiotomy is warranted under certain circumstances: Shoulder dystocia, operative vaginal delivery, and a “short” perineal body have been presumed indications. Data are inadequate to support these claims, however.
Shoulder dystocia. While it might seem to make sense to perform an episiotomy (or more likely, a proctoepisiotomy) in cases of shoulder dystocia, no data from controlled trials support this theory. Given the relative rarity of severe shoulder dystocia and the inability to conduct a truly randomized trial, physicians are left with only their clinical judgment as a guide in this circumstance.
Operative delivery. Many clinicians have advocated routine episiotomy before operative vaginal delivery, particularly with forceps. The intent is to increase the space available for delivery that has been diminished by the introduction of forceps. This rationale does not hold up as well for vacuum extraction; 1 study noted that when episiotomy is performed in cases of vacuum extraction, the likelihood of severe perineal trauma is increased.9
It has been reported10 that the greatest risk factor for both perineal trauma and third- or fourth-degree perineal laceration is episiotomy itself (TABLE), independent of mode of delivery (spontaneous or operative).
Short perineum. Many physicians, myself included, have performed episiotomies because they perceived that the perineum was short and that even a controlled delivery with optimal use of the Ritgen maneuver probably would not prevent a perineal laceration. That said, data on anal and flatus incontinence and postpartum sexual functioning suggest that spontaneous recovery from second-degree lacerations is no worse than recovery from midline episiotomy11,12 and, as stated, episiotomy itself is the leading risk factor for incurring a third- or fourth-degree extension—which imposes significantly greater recovery problems.
Two recent studies11,12 identified episiotomy as a specific, independent risk factor for fecal incontinence and delayed return of sexual activity postpartum. When matched for degree of perineal trauma, episiotomy without extension still resulted in poorer outcomes at 3 and 6 months postpartum than did spontaneous second-degree lacerations, suggesting that routine episiotomy not only fails to prevent, but may actually increase risk of perineal injury and impaired function.
‘Prophylactic’ episiotomy is not preventive
Much debate has centered on optimal utilization of so-called prophylactic episiotomy. The intent of routine prophylactic episiotomy is to protect the pelvic floor, thus minimizing the risk of urinary incontinence and pelvic floor dysfunction. Data have suggested that absence of labor and cesarean delivery may protect against pelvic floor dysfunction; however, the role of episiotomy in preventing such dysfunction remains to be determined.
Cochrane Database review. This review1 found no differences in vaginal or perineal trauma, dyspareunia, or urinary incontinence between patients with and without episiotomy. Patients who had an episiotomy had less risk of anterior perineal trauma but an overall greater risk of posterior perineal trauma and other complications. The reviewers concluded that restrictive episiotomy utilization is preferable to routine utilization.
The reviewers selected a total of 6 randomized trials; these examined:
- restrictive versus routine use of episiotomy;
- restrictive versus mediolateral episiotomy;
- restrictive versus routine midline episiotomy; and
- midline versus mediolateral episiotomy.
Compared with routine use, restrictive episiotomy involved less posterior perineal trauma (relative risk [RR], 0.88; 95% confidence interval [CI], 0.84 to 0.920), less suturing (RR, 0.74; 95% CI, 0.71 to 0.77), and fewer healing complications (RR, 0.69; 95% CI, 0.56 to 0.85). Restrictive episiotomy was associated with more anterior perineal trauma (RR, 1.79; 95% CI, 1.55 to 2.07).
There was no difference in severe vaginal or perineal trauma (RR, 1.11; 95% CI, 0.83 to 1.50), dyspareunia (RR, 1.02; 95% CI, 0.90 to 1.16), urinary incontinence (RR 0.98; 95% CI, 0.79 to 1.20), or several pain measures.
Results for restrictive versus routine mediolateral and midline episiotomies were similar to the overall comparison.
Reviewers concluded that a policy of restrictive episiotomy appears to have several benefits over routine episiotomy: less posterior perineal trauma, less suturing, fewer complications, and no difference for most pain measures and severe vaginal or perineal trauma.
Risk of anterior perineal trauma with restrictive episiotomy was increased, however. Restrictive-use protocols, likely to be institution-specific, essentially curb episiotomy use by stating that the procedure should not be “routinely performed.” Instead, episiotomy is restricted to cases in which the clinician believes it is warranted. Examples of such situations include use of forceps, shoulder dystocia, and an estimated fetal weight above 4,000 g. As discussed, the data cannot support the value of episiotomy use even in these circumstances; however, simply discouraging routine episiotomy would effectively lower the rate to the desired 30% range.
Midline versus mediolateral incision. The most vocal debates focus on which type of episiotomy to perform and whether it should be performed earlier or later in the second stage of labor.
It has been proposed that by abandoning midline episiotomies in favor of the mediolateral technique, physicians can avoid injury to the sphincter and improve immediate birth outcome without compromising long-term function—though pros and cons of this approach are a subject of debate (see “Comparison of mid-line versus mediolateral episiotomy”).
Still, the data suggest that, when properly performed, median and mediolateral episiotomy have equivalent rates of satisfactory recovery,13 though the latter technique may require more technical skill for both its performance and repair.
Early versus late incision. Proponents argue that an episiotomy at the time the presenting part is crowning is “too little, too late.” They maintain that for the procedure to be truly protective, it should be utilized earlier in the second stage of labor.
Data are insufficient to confirm or refute the efficacy of early episiotomy. One would do well to remember, however, that early episiotomy was endorsed as a method to help shorten the second stage of labor when used in conjunction with prophylactic forceps delivery—a method that is now less prevalent in obstetric practice.
The category of obstetric provider—midwife, faculty, or private provider—may be the most reliable predictor of episiotomy. Interestingly, use of episiotomy increased in the 1920s as delivery moved from home to hospital and birth attendants shifted from midwives to physicians.
In a study of demographic variables and obstetric factors associated with episiotomy in spontaneous vaginal delivery, researchers examined 1,576 term, singleton, spontaneous vaginal deliveries in nulliparas. They found that midwives had the lowest episiotomy rate (21.4%), compared with residents and full-time faculty (33.3%) and private physicians (55.6%).15
After controlling for confounding factors with logistic regression, the authors determined that private practice provider was the strongest predictor of episiotomy, followed by faculty provider, prolonged second stage of labor, fetal macrosomia, and epidural analgesia.
The study concluded that the obstetric and demographic factors evaluated did not readily explain the link between type of provider and episiotomy rate. Numerous theories have been proposed, but factors that would clearly explain the differences have yet to be identified.
Does vaginal birth trauma cause pelvic floor dysfunction?
The relationship between vaginal birth trauma, irrespective of episiotomy, and pelvic floor dysfunction remains a topic of investigation. A recent report generated much interest in the potentially protective role of prophylactic cesarean section, particularly if performed prior to the onset of labor.14
Dr. Repke reports no financial relationship with any companies whose products are mentioned in this article.
- A Cochrane Database review concluded that restrictive episiotomy utilization is preferable to routine utilization. The review of 6 randomized trials found no differences in vaginal or perineal trauma, dyspareunia, or urinary incontinence between patients with and without episiotomy. Patients who had an episiotomy had less risk of anterior perineal trauma but an overall greater risk of posterior perineal trauma and other complications.
- Shoulder dystocia, operative vaginal delivery, and a “short” perineal body have been presumed indications for episiotomy, although data are inadequate to support these claims.
- The rationale for routine prophylactic episiotomy is to protect the pelvic floor, thereby minimizing the risk of urinary incontinence and pelvic floor dysfunction; however, episiotomy’s role in preventing such dysfunction remains to be established.
Although a large body of evidence indicates reassessment is in order, prophylactic episiotomy is a contentious issue. Indeed, it has been controversial ever since the procedure first became “routine” in the United States, in 1920. Still, advocates and dissenters share the same goal: to prevent severe perineal tears and their potential for urinary and fecal incontinence and sexual dysfunction.
This article reviews research findings that indicate:
- Data are inadequate to recommend one method of episiotomy over another.
- Timing of episiotomy to shorten the second stage of labor may be less relevant in an era of decreasing forceps utilization and without evidence of improved neonatal outcomes.5
- Episiotomy, particularly midline episiotomy, remains the single greatest risk that a patient will sustain a third- or fourth-degree laceration. When such lacerations occur spontaneously, recovery is equivalent to episiotomy extension or deliberate proctoepisiotomy.
TABLE
Incidence of third- or fourth-degree laceration with and without episiotomy
| NO. STUDIES COMPILED | NO. PATIENTS | % WITH 3RD- OR 4TH-DEGREE LACERATION | |
|---|---|---|---|
| Midline episiotomy | 12 | 49,395 | 6.5 |
| No episiotomy | 13 | 38,961 | 1.4 |
| Adapted from Thorp JM.3 | |||
Research does not support presumed indications
Episiotomy was first described in 1742 as a procedure that could assist the obstetrician in difficult vaginal deliveries.3 It was not until the work of DeLee6 and Pomeroy7 was published in 1920—coincident with deliveries beginning to move from home to hospital—that the procedure became “routine.” Still, some leaders in the field—specifically, J. Whitridge Williams of Johns Hopkins—vigorously dissented.8
Historically, episiotomy has been used to facilitate delivery in cases of protracted second stage, instrumented vaginal delivery, and suspected fetal compromise. However, data supporting episiotomy as a facilitating procedure are sparse, and evidence endorsing prophylactic episiotomy is largely anecdotal or descriptive.
Agreement is widespread that episiotomy is warranted under certain circumstances: Shoulder dystocia, operative vaginal delivery, and a “short” perineal body have been presumed indications. Data are inadequate to support these claims, however.
Shoulder dystocia. While it might seem to make sense to perform an episiotomy (or more likely, a proctoepisiotomy) in cases of shoulder dystocia, no data from controlled trials support this theory. Given the relative rarity of severe shoulder dystocia and the inability to conduct a truly randomized trial, physicians are left with only their clinical judgment as a guide in this circumstance.
Operative delivery. Many clinicians have advocated routine episiotomy before operative vaginal delivery, particularly with forceps. The intent is to increase the space available for delivery that has been diminished by the introduction of forceps. This rationale does not hold up as well for vacuum extraction; 1 study noted that when episiotomy is performed in cases of vacuum extraction, the likelihood of severe perineal trauma is increased.9
It has been reported10 that the greatest risk factor for both perineal trauma and third- or fourth-degree perineal laceration is episiotomy itself (TABLE), independent of mode of delivery (spontaneous or operative).
Short perineum. Many physicians, myself included, have performed episiotomies because they perceived that the perineum was short and that even a controlled delivery with optimal use of the Ritgen maneuver probably would not prevent a perineal laceration. That said, data on anal and flatus incontinence and postpartum sexual functioning suggest that spontaneous recovery from second-degree lacerations is no worse than recovery from midline episiotomy11,12 and, as stated, episiotomy itself is the leading risk factor for incurring a third- or fourth-degree extension—which imposes significantly greater recovery problems.
Two recent studies11,12 identified episiotomy as a specific, independent risk factor for fecal incontinence and delayed return of sexual activity postpartum. When matched for degree of perineal trauma, episiotomy without extension still resulted in poorer outcomes at 3 and 6 months postpartum than did spontaneous second-degree lacerations, suggesting that routine episiotomy not only fails to prevent, but may actually increase risk of perineal injury and impaired function.
‘Prophylactic’ episiotomy is not preventive
Much debate has centered on optimal utilization of so-called prophylactic episiotomy. The intent of routine prophylactic episiotomy is to protect the pelvic floor, thus minimizing the risk of urinary incontinence and pelvic floor dysfunction. Data have suggested that absence of labor and cesarean delivery may protect against pelvic floor dysfunction; however, the role of episiotomy in preventing such dysfunction remains to be determined.
Cochrane Database review. This review1 found no differences in vaginal or perineal trauma, dyspareunia, or urinary incontinence between patients with and without episiotomy. Patients who had an episiotomy had less risk of anterior perineal trauma but an overall greater risk of posterior perineal trauma and other complications. The reviewers concluded that restrictive episiotomy utilization is preferable to routine utilization.
The reviewers selected a total of 6 randomized trials; these examined:
- restrictive versus routine use of episiotomy;
- restrictive versus mediolateral episiotomy;
- restrictive versus routine midline episiotomy; and
- midline versus mediolateral episiotomy.
Compared with routine use, restrictive episiotomy involved less posterior perineal trauma (relative risk [RR], 0.88; 95% confidence interval [CI], 0.84 to 0.920), less suturing (RR, 0.74; 95% CI, 0.71 to 0.77), and fewer healing complications (RR, 0.69; 95% CI, 0.56 to 0.85). Restrictive episiotomy was associated with more anterior perineal trauma (RR, 1.79; 95% CI, 1.55 to 2.07).
There was no difference in severe vaginal or perineal trauma (RR, 1.11; 95% CI, 0.83 to 1.50), dyspareunia (RR, 1.02; 95% CI, 0.90 to 1.16), urinary incontinence (RR 0.98; 95% CI, 0.79 to 1.20), or several pain measures.
Results for restrictive versus routine mediolateral and midline episiotomies were similar to the overall comparison.
Reviewers concluded that a policy of restrictive episiotomy appears to have several benefits over routine episiotomy: less posterior perineal trauma, less suturing, fewer complications, and no difference for most pain measures and severe vaginal or perineal trauma.
Risk of anterior perineal trauma with restrictive episiotomy was increased, however. Restrictive-use protocols, likely to be institution-specific, essentially curb episiotomy use by stating that the procedure should not be “routinely performed.” Instead, episiotomy is restricted to cases in which the clinician believes it is warranted. Examples of such situations include use of forceps, shoulder dystocia, and an estimated fetal weight above 4,000 g. As discussed, the data cannot support the value of episiotomy use even in these circumstances; however, simply discouraging routine episiotomy would effectively lower the rate to the desired 30% range.
Midline versus mediolateral incision. The most vocal debates focus on which type of episiotomy to perform and whether it should be performed earlier or later in the second stage of labor.
It has been proposed that by abandoning midline episiotomies in favor of the mediolateral technique, physicians can avoid injury to the sphincter and improve immediate birth outcome without compromising long-term function—though pros and cons of this approach are a subject of debate (see “Comparison of mid-line versus mediolateral episiotomy”).
Still, the data suggest that, when properly performed, median and mediolateral episiotomy have equivalent rates of satisfactory recovery,13 though the latter technique may require more technical skill for both its performance and repair.
Early versus late incision. Proponents argue that an episiotomy at the time the presenting part is crowning is “too little, too late.” They maintain that for the procedure to be truly protective, it should be utilized earlier in the second stage of labor.
Data are insufficient to confirm or refute the efficacy of early episiotomy. One would do well to remember, however, that early episiotomy was endorsed as a method to help shorten the second stage of labor when used in conjunction with prophylactic forceps delivery—a method that is now less prevalent in obstetric practice.
The category of obstetric provider—midwife, faculty, or private provider—may be the most reliable predictor of episiotomy. Interestingly, use of episiotomy increased in the 1920s as delivery moved from home to hospital and birth attendants shifted from midwives to physicians.
In a study of demographic variables and obstetric factors associated with episiotomy in spontaneous vaginal delivery, researchers examined 1,576 term, singleton, spontaneous vaginal deliveries in nulliparas. They found that midwives had the lowest episiotomy rate (21.4%), compared with residents and full-time faculty (33.3%) and private physicians (55.6%).15
After controlling for confounding factors with logistic regression, the authors determined that private practice provider was the strongest predictor of episiotomy, followed by faculty provider, prolonged second stage of labor, fetal macrosomia, and epidural analgesia.
The study concluded that the obstetric and demographic factors evaluated did not readily explain the link between type of provider and episiotomy rate. Numerous theories have been proposed, but factors that would clearly explain the differences have yet to be identified.
Does vaginal birth trauma cause pelvic floor dysfunction?
The relationship between vaginal birth trauma, irrespective of episiotomy, and pelvic floor dysfunction remains a topic of investigation. A recent report generated much interest in the potentially protective role of prophylactic cesarean section, particularly if performed prior to the onset of labor.14
Dr. Repke reports no financial relationship with any companies whose products are mentioned in this article.
1. Carroli G, Belizan J. Episiotomy for vaginal birth [Cochrane Review]. In: The Cochrane Library Issue 3, 2003. Oxford: Update Software.
2. Lede RL, Belizan JM, Caroli G. Is routine use of episiotomy justified? Am J Obstet Gynecol. 1996;174:1399-1402.
3. Thorp JM. Episiotomy. In: Repke JT, ed. Intrapartum Obstetrics. New York, NY: Churchill Livingstone; 1996;489-499.
4. Thacker SB, Banta HD. Benefits and risks of episiotomy: an interpretive review of the English language literature, 1860-1980. Obstet Gynecol Surv. 1983;38:322-338.
5. Eason E, Feldman P. Much ado about a little cut: is episiotomy worthwhile? Obstet Gynecol. 2000;95:616-618.
6. DeLee JB. The prophylactic forceps operation. Am J Obstet Gynecol. 1920;1:34-44.
7. Pomeroy RH. Shall we cut and reconstruct the perineum for every primipara? Am J Obstet Dis Women Child. 1918;78:211-219.
8. Taylor ES. Comment on episiotomy and third degree tears. Obstet Gynecol Surg. 1985;41:229.-
9. Robinson JN, Norwitz ER, Cohen AP, McElrath TF, Lieberman ES. Episiotomy, operative vaginal delivery and significant perinatal trauma in nulliparous women. Am J Obstet Gynecol. 1999;181:1180-1184.
10. Robinson JN, Norwitz ER, Cohen AP, McElrath TF, Lieberman ES. Epidural analgesia and the occurrence of third and fourth degree laceration in nulliparas. Obstet Gynecol. 1999;94:259-262.
11. Signorello LB, Harlow BL, Chekos AK, Repke JT. Midline episiotomy and anal incontinence: a retrospective cohort study. BMJ. 2000;320:86-90.
12. Signorello LB, Harlow BL, Chekos AK, Repke JT. Postpartum sexual functioning and its relationship to perineal trauma: a retrospective cohort study of primiparous women. Am J Obstet Gynecol. 2001;184:881-890.
13. Coats PM, Chan KK, Wilkins M, Beard RJ. A comparison between midline and mediolateral episiotomies. Br J Obstet Gynaecol. 1980;87:408-412.
14. Dietz HP, Bennett MJ. The effect of childbirth on pelvic organ mobility. Obstet Gynecol. 2003;102:223-228.
15. Robinson JN, Norwitz ER, Cohen AP, Lieberman E. Predictors of episiotomy use at first spontaneous vaginal delivery. Obstet Gynecol. 2000;96:214-218
1. Carroli G, Belizan J. Episiotomy for vaginal birth [Cochrane Review]. In: The Cochrane Library Issue 3, 2003. Oxford: Update Software.
2. Lede RL, Belizan JM, Caroli G. Is routine use of episiotomy justified? Am J Obstet Gynecol. 1996;174:1399-1402.
3. Thorp JM. Episiotomy. In: Repke JT, ed. Intrapartum Obstetrics. New York, NY: Churchill Livingstone; 1996;489-499.
4. Thacker SB, Banta HD. Benefits and risks of episiotomy: an interpretive review of the English language literature, 1860-1980. Obstet Gynecol Surv. 1983;38:322-338.
5. Eason E, Feldman P. Much ado about a little cut: is episiotomy worthwhile? Obstet Gynecol. 2000;95:616-618.
6. DeLee JB. The prophylactic forceps operation. Am J Obstet Gynecol. 1920;1:34-44.
7. Pomeroy RH. Shall we cut and reconstruct the perineum for every primipara? Am J Obstet Dis Women Child. 1918;78:211-219.
8. Taylor ES. Comment on episiotomy and third degree tears. Obstet Gynecol Surg. 1985;41:229.-
9. Robinson JN, Norwitz ER, Cohen AP, McElrath TF, Lieberman ES. Episiotomy, operative vaginal delivery and significant perinatal trauma in nulliparous women. Am J Obstet Gynecol. 1999;181:1180-1184.
10. Robinson JN, Norwitz ER, Cohen AP, McElrath TF, Lieberman ES. Epidural analgesia and the occurrence of third and fourth degree laceration in nulliparas. Obstet Gynecol. 1999;94:259-262.
11. Signorello LB, Harlow BL, Chekos AK, Repke JT. Midline episiotomy and anal incontinence: a retrospective cohort study. BMJ. 2000;320:86-90.
12. Signorello LB, Harlow BL, Chekos AK, Repke JT. Postpartum sexual functioning and its relationship to perineal trauma: a retrospective cohort study of primiparous women. Am J Obstet Gynecol. 2001;184:881-890.
13. Coats PM, Chan KK, Wilkins M, Beard RJ. A comparison between midline and mediolateral episiotomies. Br J Obstet Gynaecol. 1980;87:408-412.
14. Dietz HP, Bennett MJ. The effect of childbirth on pelvic organ mobility. Obstet Gynecol. 2003;102:223-228.
15. Robinson JN, Norwitz ER, Cohen AP, Lieberman E. Predictors of episiotomy use at first spontaneous vaginal delivery. Obstet Gynecol. 2000;96:214-218
3 steps to reduce postoperative ileus
- Rather than contributing to ileus, early postoperative feeding now appears to help diminish its occurrence.
- There is no justification for routine postoperative placement of nasogastric tubes in asymptomatic patients.
- Thoracic epidurals block the reflex that causes postoperative ileus and can be used to prevent its occurrence.
Traditionally, the routine approach to avoiding this complication consisted of placing a nasogastric (NG) tube to decompress the bowel and delaying feeding until bowel function resumed.
More recent studies indicate that a different tactic may be preferable. These suggest that postoperative ileus—which has an estimated annual cost of $750 million1—can be significantly reduced with a simple 3-step process:
- withholding the nasogastric (NG) tube,
- feeding the patient early in the recovery process, and
- continuing epidural local anesthesia postoperatively.
Pathogenesis of ileus
We now know that the return of bowel function following surgery is an orderly event. The return of the small intestine’s action begins first, usually 4 to 8 hours postoperatively, and generally becomes complete around 24 hours. The colon resumes its function between 48 and 72 hours postoperatively.2
Very little has been written about the pathogenesis of postoperative ileus, but multiple causes have been suggested: sympathetic reflexes; inhibitory humoral agents; release of norepinephrine from the bowel wall; and the effects of anesthesia agents, opiates, and inflammation.3 The 2 most frequently mentioned etiologies are:
- the inhibitory neural reflex and
- inflammatory mediators released from the site of injury. (Inflammation is thought to trigger the release of macrophages, cytokines, and other inflammatory mediators, causing neutrophil infiltration.4)
Controversy remains as to what actually initiates the ileus. Is it manipulation of the bowel or the rigors of surgery and anesthesia? Kalff et al6 conducted bowel muscle studies in rats and concluded that manipulation of the bowel—and not the laparotomy per se—causes a failure of gut circular muscle function 24 hours later. They also noted an increase of phagocytes and mast cells. Their data support the hypothesis that abdominal surgery initiates a cascade of inflammatory events that leads to postoperative ileus.2
The case for early feeding
In the past 10 years, several studies have demonstrated that—rather than reduce the incidence of ileus—inserting an NG tube and withholding regular feeding following abdominal surgery can cause an ileus or prolong a preexisting one. Other trials have shown that feeding a patient early in the postoperative period can actually prevent ileus.7,8
Physiologic studies have shown that neither electrical activity of the bowel nor motor activities in the stomach are affected by surgery.8 Schilder et al9 reported bowel activity before the passage of flatus, indicating that the bowel is on its way to recovery much earlier than had been assumed. Thus, early postoperative feeding is well tolerated in most patients and associated with reduced discomfort and a more rapid recovery.7
For example, MacMillan et al studied 139 women undergoing gynecologic surgery for benign conditions; 67 were randomized to “early” feeding and 72 to traditional management. Early feeding involved a low-residue diet given 6 hours postoperatively, while traditional feeding consisted of clear liquids, which were withheld until the return of normal bowel sounds. Patients progressed to a regular diet with the passage of flatus. No increase in gastrointestinal complaints occurred in the early feeding group.7
Pearl et al8 compared similar groups of patients (TABLE 1). Patients in the first group were fed a clear liquid diet on the first postoperative day and progressed to a regular diet as soon as it could be tolerated. The traditional group was not fed until the return of bowel function, which was defined as the passage of flatus and no abdominal distension or vomiting; they were then started on clear liquids and, later, solid foods. While the incidence of complications was the same in both groups, hospitalization was shorter in the early feeding group.
A Cochrane review compared early versus delayed oral fluids and food after cesarean delivery. Of 12 studies considered, 6 were included in the review. No evidence was found to justify a policy of withholding oral fluids after uncomplicated cesarean sections.5
Simple versus complex procedures. Early feeding is not only safe in standardized surgeries such as cesarean section, but extends to complicated surgeries as well, as demonstrated in the trials by MacMillan et al7 and Pearl et al,8 which involved major gynecologic surgery. Trials with gynecologic oncology patients have shown the same result.2,8 Even patients undergoing colorectal surgery can tolerate oral feeding very early in their postoperative course without bowel complications.2,8
Not all studies have reported similar findings. Several concluded that other, nonmedical reasons, such as insurance requirements, accounted for the shorter hospital stays in many patients receiving early feeding.
TABLE 1
Complication rates associated with early feeding versus traditional management
| COMPLICATION | EARLY FEEDING* (N=92) | TRADITIONAL MANAGEMENT· (N= 103) |
|---|---|---|
| Nausea | 43.5% | 24.3% |
| Nasogastric tube use | 3.3% | 6.7% |
| Febrile morbidity | 54.3% | 55.3% |
| Pneumonia | 0% | 1.9% |
| Wound complications | 21.7% | 21.4% |
| Atelectasis | 8.7% | 10.7% |
| Length of hospital stay (mean±standard deviation) | 4.6±2.1 | 5.8±2.7 |
| *Clear liquid diet on postoperative day 1 | ||
| †Feeding delayed until return of bowel function | ||
| Reprinted from Obstet Gyn; vol 92; Pearl ML, Valea FA, Fischer M, Mahler L, Chalas E. A randomized controlled trial of early postoperative feeding in gynecologic oncology patients undergoing intraabdominal surgery; pages 94-97; copyright 1998; with permission from American College of Obstetricians and Gynecologists. | ||
Nasogastric tubes only when indicated
The use of NG tubes after laparotomy has been studied extensively. A review of the literature suggests that routine placement of the tubes in asymptomatic patients is not justified and may possibly be harmful.10
In their meta-analysis of the issue, Cheatham et al11 showed that although abdominal distension and vomiting are more frequent in patients who forgo NG tubes postoperatively, fever, atelectasis, and pneumonia are less common, and the interval between surgery and oral feeding is reduced (TABLE 2).
The authors concluded that for every NG tube inserted after abdominal surgery, at least 20 patients can be managed without it.
Forgoing an NG tube also lowers the risk of pulmonary complications, which increases 10-fold when a tube is inserted.12
TABLE 2
Complications associated with selective versus routine NG tube placement
| SELECTIVE PLACEMENT (N) | ROUTINE PLACEMENT (N) | P VALUE | RELATIVE RISK | |
|---|---|---|---|---|
| Patients | 1,986 | 1,978 | ||
| Tubes placed/replaced | 103 | 36 | <.001> | 2.9 |
| Complications | 833 | 1,084 | .03 | 0.76 |
| Deaths | 13 | 25 | .22 | 0.36 |
| Pneumonia | 53 | 119 | <.0001> | 0.49 |
| Atelectasis | 44 | 94 | .001 | 0.46 |
| Fever | 108 | 212 | .02 | 0.51 |
| Vomiting | 201 | 168 | .11 | 1.19 |
| Oral feedings (days) | 3.53 | 4.59 | .04 | |
| Length of stay (days) | 9.32 | 10.1 | .22 | |
| NG=nasogastric | ||||
| Reprinted with permission from Cheatham ML, Chapman WC, Key SP, Sawyer JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg. 1995:221:469-478. | ||||
Continue the epidural anesthetic
A number of experts believe that postoperative ileus is caused by stimulation of neural reflexes, which appear to be of 2 kinds: afferent stimuli to the spinal cord and efferent stimuli to the intestines through the sympathetic nervous system. The latter inhibits motility of the intestinal tract. Numerous studies demonstrate that this sympathetic reflex can be blocked by the use of epidural anesthesia.10
For example, Holte et al4 found that postoperative administration of thoracic epidural blockade with local anesthesia significantly reduced both ileus and pulmonary complications. They concluded that continuous epidural anesthesia with local anesthesia and minimally invasive surgery are the 2 most critical events in reducing postoperative ileus.
In a Cochrane review, Jorgensen and colleagues12 compared the effects of epidural local anesthesia and opioid-based analgesic regimens on postoperative gastrointestinal paralysis, nausea and vomiting, and pain after abdominal surgery (TABLE 3). Epidural local anesthetics reduced gastrointestinal paralysis, as compared with systemic or epidural opioids, but provided the same postoperative pain relief. They also found that the addition of opioids to local epidural anesthesia provided superior postoperative analgesia—compared with epidural local anesthetics alone—without increasing the likelihood of ileus.
A study10 of patients undergoing colectomy found postoperative ileus was prevented or decreased with a 2-day regimen that included:
- continuous thoracic epidural anesthesia for 48 hours;
- withholding NG tubes;
- having the patient drink a liter of fluid on the day of surgery;
- initiating feeding after 24 hours;
- administering milk of magnesia; and
- mobilization after 8 hours, if possible.
Physicians in this study also used transverse surgical incisions to reduce pain and pulmonary problems.
TABLE 3
Anesthetic effect on GI function, postoperative pain, and nausea and vomiting: A comparison
| ANESTHETIC | GI FUNCTION RETURNS | PAIN RELIEF | NAUSEA AND VOMITING |
|---|---|---|---|
| Epidural plus local | 24 hr | Comparable | No significant difference |
| Epidural plus opioids | 37 hr | Comparable | No significant difference |
| Systemic plus opioids | 37 hr | Comparable | No significant difference |
| GI=gastrointestinal | |||
| Data from Jorgensen H, et al12 | |||
Clinical recommendations
The deregulation of the autonomic nervous system during surgery alters the gastrointestinal tract postoperatively, with neurotransmitters, local factors, and hormones playing a large role. Some forms of anesthesia also contribute to postoperative ileus, as does the use of narcotic analgesia after surgery.
The most efficient ways to activate the bowel postoperatively are:
- Continuing the thoracic epidural from 24 to 48 hours, which increases the splanchnic blood flow and blocks afferent and efferent sympathetic inhibitory nerve impulses. Note, however, that comparative studies of thoracic epidural anesthesia with local anesthesia are needed to quantify its impact.
- Hydrating the patient with a large amount of fluid in the first 24 hours after surgery.
Following these steps routinely can significantly decrease the risk of postoperative ileus and thus its resulting complications.
Dr. Rosenman reports no financial relationship with any companies whose products are mentioned in this article.
1. Groudine S. Use of inexpensive anesthesia during surgery may shorten hospital stay. Anesth Analg. 1998;87:1212-1213.
2. Cutillo G, Maneschi F, Franchi M, Giannice R, Scambia G, Benedetti-Panici P. Early feeding compared with nasogastric decompression after major gynecologic surgery: a randomized study. Obstet Gynecol. 1999;93:41-45.
3. Luckey A, Livingston E, Tache Y. Mechanisms and treatment of postoperative ileus. Arch Surg. 2000;138:206-214.
4. Holte K, Kehlet H. Postoperative ileus: a preventable event. Br J Surg. 2000;87:1480-1493.
5. Mangesi L, Hofmeyr GJ. Early compared with delayed oral fluids and food after caesarean section. Cochrane Database Syst Rev. 2002;(3):CD003516.-
6. Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg. 1998;228,5:652-663.
7. MacMillan SL, Kammerer-Doak D, Rogers RG, Parker KM. Early feeding and the incidence of gastrointestinal symptoms after major gynecologic surgery. Obstet Gynecol. 2000;96:604-608.
8. Pearl ML, Valea FA, Fischer M, Mahler L, Chalas E. A randomized controlled trial of early postoperative feeding in gynecologic oncology patients undergoing intraabdominal surgery. Obstet Gynecol. 1998;92:94-97.
9. Schilder JM, Hurteau JA, Look KY, Moore DH, Raff G, Stehman FB, Sutton GP. A prospective controlled trial of early postoperative oral intake following major abdominal gynecologic surgery. Gynecol Oncol. 1997;67:235-240.
10. Kehlet H, Holte K. Review of postoperative ileus. Am J Surg. 2001;182(5a Suppl):3S-10S.
11. Cheatham ML, Chapman WC, Key SP, Sawyer JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg. 1995;221:469-478.
12. Jorgensen H, Wetterslev J, Moiniche S, Dahl JB. Epidural local anesthetics versus opioid-based analgesic regimens on postoperative gastrointestinal paralysis, PONV and pain after abdominal surgery. Cochrane Database Syst Rev. 2000;(4):CD001893.-
- Rather than contributing to ileus, early postoperative feeding now appears to help diminish its occurrence.
- There is no justification for routine postoperative placement of nasogastric tubes in asymptomatic patients.
- Thoracic epidurals block the reflex that causes postoperative ileus and can be used to prevent its occurrence.
Traditionally, the routine approach to avoiding this complication consisted of placing a nasogastric (NG) tube to decompress the bowel and delaying feeding until bowel function resumed.
More recent studies indicate that a different tactic may be preferable. These suggest that postoperative ileus—which has an estimated annual cost of $750 million1—can be significantly reduced with a simple 3-step process:
- withholding the nasogastric (NG) tube,
- feeding the patient early in the recovery process, and
- continuing epidural local anesthesia postoperatively.
Pathogenesis of ileus
We now know that the return of bowel function following surgery is an orderly event. The return of the small intestine’s action begins first, usually 4 to 8 hours postoperatively, and generally becomes complete around 24 hours. The colon resumes its function between 48 and 72 hours postoperatively.2
Very little has been written about the pathogenesis of postoperative ileus, but multiple causes have been suggested: sympathetic reflexes; inhibitory humoral agents; release of norepinephrine from the bowel wall; and the effects of anesthesia agents, opiates, and inflammation.3 The 2 most frequently mentioned etiologies are:
- the inhibitory neural reflex and
- inflammatory mediators released from the site of injury. (Inflammation is thought to trigger the release of macrophages, cytokines, and other inflammatory mediators, causing neutrophil infiltration.4)
Controversy remains as to what actually initiates the ileus. Is it manipulation of the bowel or the rigors of surgery and anesthesia? Kalff et al6 conducted bowel muscle studies in rats and concluded that manipulation of the bowel—and not the laparotomy per se—causes a failure of gut circular muscle function 24 hours later. They also noted an increase of phagocytes and mast cells. Their data support the hypothesis that abdominal surgery initiates a cascade of inflammatory events that leads to postoperative ileus.2
The case for early feeding
In the past 10 years, several studies have demonstrated that—rather than reduce the incidence of ileus—inserting an NG tube and withholding regular feeding following abdominal surgery can cause an ileus or prolong a preexisting one. Other trials have shown that feeding a patient early in the postoperative period can actually prevent ileus.7,8
Physiologic studies have shown that neither electrical activity of the bowel nor motor activities in the stomach are affected by surgery.8 Schilder et al9 reported bowel activity before the passage of flatus, indicating that the bowel is on its way to recovery much earlier than had been assumed. Thus, early postoperative feeding is well tolerated in most patients and associated with reduced discomfort and a more rapid recovery.7
For example, MacMillan et al studied 139 women undergoing gynecologic surgery for benign conditions; 67 were randomized to “early” feeding and 72 to traditional management. Early feeding involved a low-residue diet given 6 hours postoperatively, while traditional feeding consisted of clear liquids, which were withheld until the return of normal bowel sounds. Patients progressed to a regular diet with the passage of flatus. No increase in gastrointestinal complaints occurred in the early feeding group.7
Pearl et al8 compared similar groups of patients (TABLE 1). Patients in the first group were fed a clear liquid diet on the first postoperative day and progressed to a regular diet as soon as it could be tolerated. The traditional group was not fed until the return of bowel function, which was defined as the passage of flatus and no abdominal distension or vomiting; they were then started on clear liquids and, later, solid foods. While the incidence of complications was the same in both groups, hospitalization was shorter in the early feeding group.
A Cochrane review compared early versus delayed oral fluids and food after cesarean delivery. Of 12 studies considered, 6 were included in the review. No evidence was found to justify a policy of withholding oral fluids after uncomplicated cesarean sections.5
Simple versus complex procedures. Early feeding is not only safe in standardized surgeries such as cesarean section, but extends to complicated surgeries as well, as demonstrated in the trials by MacMillan et al7 and Pearl et al,8 which involved major gynecologic surgery. Trials with gynecologic oncology patients have shown the same result.2,8 Even patients undergoing colorectal surgery can tolerate oral feeding very early in their postoperative course without bowel complications.2,8
Not all studies have reported similar findings. Several concluded that other, nonmedical reasons, such as insurance requirements, accounted for the shorter hospital stays in many patients receiving early feeding.
TABLE 1
Complication rates associated with early feeding versus traditional management
| COMPLICATION | EARLY FEEDING* (N=92) | TRADITIONAL MANAGEMENT· (N= 103) |
|---|---|---|
| Nausea | 43.5% | 24.3% |
| Nasogastric tube use | 3.3% | 6.7% |
| Febrile morbidity | 54.3% | 55.3% |
| Pneumonia | 0% | 1.9% |
| Wound complications | 21.7% | 21.4% |
| Atelectasis | 8.7% | 10.7% |
| Length of hospital stay (mean±standard deviation) | 4.6±2.1 | 5.8±2.7 |
| *Clear liquid diet on postoperative day 1 | ||
| †Feeding delayed until return of bowel function | ||
| Reprinted from Obstet Gyn; vol 92; Pearl ML, Valea FA, Fischer M, Mahler L, Chalas E. A randomized controlled trial of early postoperative feeding in gynecologic oncology patients undergoing intraabdominal surgery; pages 94-97; copyright 1998; with permission from American College of Obstetricians and Gynecologists. | ||
Nasogastric tubes only when indicated
The use of NG tubes after laparotomy has been studied extensively. A review of the literature suggests that routine placement of the tubes in asymptomatic patients is not justified and may possibly be harmful.10
In their meta-analysis of the issue, Cheatham et al11 showed that although abdominal distension and vomiting are more frequent in patients who forgo NG tubes postoperatively, fever, atelectasis, and pneumonia are less common, and the interval between surgery and oral feeding is reduced (TABLE 2).
The authors concluded that for every NG tube inserted after abdominal surgery, at least 20 patients can be managed without it.
Forgoing an NG tube also lowers the risk of pulmonary complications, which increases 10-fold when a tube is inserted.12
TABLE 2
Complications associated with selective versus routine NG tube placement
| SELECTIVE PLACEMENT (N) | ROUTINE PLACEMENT (N) | P VALUE | RELATIVE RISK | |
|---|---|---|---|---|
| Patients | 1,986 | 1,978 | ||
| Tubes placed/replaced | 103 | 36 | <.001> | 2.9 |
| Complications | 833 | 1,084 | .03 | 0.76 |
| Deaths | 13 | 25 | .22 | 0.36 |
| Pneumonia | 53 | 119 | <.0001> | 0.49 |
| Atelectasis | 44 | 94 | .001 | 0.46 |
| Fever | 108 | 212 | .02 | 0.51 |
| Vomiting | 201 | 168 | .11 | 1.19 |
| Oral feedings (days) | 3.53 | 4.59 | .04 | |
| Length of stay (days) | 9.32 | 10.1 | .22 | |
| NG=nasogastric | ||||
| Reprinted with permission from Cheatham ML, Chapman WC, Key SP, Sawyer JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg. 1995:221:469-478. | ||||
Continue the epidural anesthetic
A number of experts believe that postoperative ileus is caused by stimulation of neural reflexes, which appear to be of 2 kinds: afferent stimuli to the spinal cord and efferent stimuli to the intestines through the sympathetic nervous system. The latter inhibits motility of the intestinal tract. Numerous studies demonstrate that this sympathetic reflex can be blocked by the use of epidural anesthesia.10
For example, Holte et al4 found that postoperative administration of thoracic epidural blockade with local anesthesia significantly reduced both ileus and pulmonary complications. They concluded that continuous epidural anesthesia with local anesthesia and minimally invasive surgery are the 2 most critical events in reducing postoperative ileus.
In a Cochrane review, Jorgensen and colleagues12 compared the effects of epidural local anesthesia and opioid-based analgesic regimens on postoperative gastrointestinal paralysis, nausea and vomiting, and pain after abdominal surgery (TABLE 3). Epidural local anesthetics reduced gastrointestinal paralysis, as compared with systemic or epidural opioids, but provided the same postoperative pain relief. They also found that the addition of opioids to local epidural anesthesia provided superior postoperative analgesia—compared with epidural local anesthetics alone—without increasing the likelihood of ileus.
A study10 of patients undergoing colectomy found postoperative ileus was prevented or decreased with a 2-day regimen that included:
- continuous thoracic epidural anesthesia for 48 hours;
- withholding NG tubes;
- having the patient drink a liter of fluid on the day of surgery;
- initiating feeding after 24 hours;
- administering milk of magnesia; and
- mobilization after 8 hours, if possible.
Physicians in this study also used transverse surgical incisions to reduce pain and pulmonary problems.
TABLE 3
Anesthetic effect on GI function, postoperative pain, and nausea and vomiting: A comparison
| ANESTHETIC | GI FUNCTION RETURNS | PAIN RELIEF | NAUSEA AND VOMITING |
|---|---|---|---|
| Epidural plus local | 24 hr | Comparable | No significant difference |
| Epidural plus opioids | 37 hr | Comparable | No significant difference |
| Systemic plus opioids | 37 hr | Comparable | No significant difference |
| GI=gastrointestinal | |||
| Data from Jorgensen H, et al12 | |||
Clinical recommendations
The deregulation of the autonomic nervous system during surgery alters the gastrointestinal tract postoperatively, with neurotransmitters, local factors, and hormones playing a large role. Some forms of anesthesia also contribute to postoperative ileus, as does the use of narcotic analgesia after surgery.
The most efficient ways to activate the bowel postoperatively are:
- Continuing the thoracic epidural from 24 to 48 hours, which increases the splanchnic blood flow and blocks afferent and efferent sympathetic inhibitory nerve impulses. Note, however, that comparative studies of thoracic epidural anesthesia with local anesthesia are needed to quantify its impact.
- Hydrating the patient with a large amount of fluid in the first 24 hours after surgery.
Following these steps routinely can significantly decrease the risk of postoperative ileus and thus its resulting complications.
Dr. Rosenman reports no financial relationship with any companies whose products are mentioned in this article.
- Rather than contributing to ileus, early postoperative feeding now appears to help diminish its occurrence.
- There is no justification for routine postoperative placement of nasogastric tubes in asymptomatic patients.
- Thoracic epidurals block the reflex that causes postoperative ileus and can be used to prevent its occurrence.
Traditionally, the routine approach to avoiding this complication consisted of placing a nasogastric (NG) tube to decompress the bowel and delaying feeding until bowel function resumed.
More recent studies indicate that a different tactic may be preferable. These suggest that postoperative ileus—which has an estimated annual cost of $750 million1—can be significantly reduced with a simple 3-step process:
- withholding the nasogastric (NG) tube,
- feeding the patient early in the recovery process, and
- continuing epidural local anesthesia postoperatively.
Pathogenesis of ileus
We now know that the return of bowel function following surgery is an orderly event. The return of the small intestine’s action begins first, usually 4 to 8 hours postoperatively, and generally becomes complete around 24 hours. The colon resumes its function between 48 and 72 hours postoperatively.2
Very little has been written about the pathogenesis of postoperative ileus, but multiple causes have been suggested: sympathetic reflexes; inhibitory humoral agents; release of norepinephrine from the bowel wall; and the effects of anesthesia agents, opiates, and inflammation.3 The 2 most frequently mentioned etiologies are:
- the inhibitory neural reflex and
- inflammatory mediators released from the site of injury. (Inflammation is thought to trigger the release of macrophages, cytokines, and other inflammatory mediators, causing neutrophil infiltration.4)
Controversy remains as to what actually initiates the ileus. Is it manipulation of the bowel or the rigors of surgery and anesthesia? Kalff et al6 conducted bowel muscle studies in rats and concluded that manipulation of the bowel—and not the laparotomy per se—causes a failure of gut circular muscle function 24 hours later. They also noted an increase of phagocytes and mast cells. Their data support the hypothesis that abdominal surgery initiates a cascade of inflammatory events that leads to postoperative ileus.2
The case for early feeding
In the past 10 years, several studies have demonstrated that—rather than reduce the incidence of ileus—inserting an NG tube and withholding regular feeding following abdominal surgery can cause an ileus or prolong a preexisting one. Other trials have shown that feeding a patient early in the postoperative period can actually prevent ileus.7,8
Physiologic studies have shown that neither electrical activity of the bowel nor motor activities in the stomach are affected by surgery.8 Schilder et al9 reported bowel activity before the passage of flatus, indicating that the bowel is on its way to recovery much earlier than had been assumed. Thus, early postoperative feeding is well tolerated in most patients and associated with reduced discomfort and a more rapid recovery.7
For example, MacMillan et al studied 139 women undergoing gynecologic surgery for benign conditions; 67 were randomized to “early” feeding and 72 to traditional management. Early feeding involved a low-residue diet given 6 hours postoperatively, while traditional feeding consisted of clear liquids, which were withheld until the return of normal bowel sounds. Patients progressed to a regular diet with the passage of flatus. No increase in gastrointestinal complaints occurred in the early feeding group.7
Pearl et al8 compared similar groups of patients (TABLE 1). Patients in the first group were fed a clear liquid diet on the first postoperative day and progressed to a regular diet as soon as it could be tolerated. The traditional group was not fed until the return of bowel function, which was defined as the passage of flatus and no abdominal distension or vomiting; they were then started on clear liquids and, later, solid foods. While the incidence of complications was the same in both groups, hospitalization was shorter in the early feeding group.
A Cochrane review compared early versus delayed oral fluids and food after cesarean delivery. Of 12 studies considered, 6 were included in the review. No evidence was found to justify a policy of withholding oral fluids after uncomplicated cesarean sections.5
Simple versus complex procedures. Early feeding is not only safe in standardized surgeries such as cesarean section, but extends to complicated surgeries as well, as demonstrated in the trials by MacMillan et al7 and Pearl et al,8 which involved major gynecologic surgery. Trials with gynecologic oncology patients have shown the same result.2,8 Even patients undergoing colorectal surgery can tolerate oral feeding very early in their postoperative course without bowel complications.2,8
Not all studies have reported similar findings. Several concluded that other, nonmedical reasons, such as insurance requirements, accounted for the shorter hospital stays in many patients receiving early feeding.
TABLE 1
Complication rates associated with early feeding versus traditional management
| COMPLICATION | EARLY FEEDING* (N=92) | TRADITIONAL MANAGEMENT· (N= 103) |
|---|---|---|
| Nausea | 43.5% | 24.3% |
| Nasogastric tube use | 3.3% | 6.7% |
| Febrile morbidity | 54.3% | 55.3% |
| Pneumonia | 0% | 1.9% |
| Wound complications | 21.7% | 21.4% |
| Atelectasis | 8.7% | 10.7% |
| Length of hospital stay (mean±standard deviation) | 4.6±2.1 | 5.8±2.7 |
| *Clear liquid diet on postoperative day 1 | ||
| †Feeding delayed until return of bowel function | ||
| Reprinted from Obstet Gyn; vol 92; Pearl ML, Valea FA, Fischer M, Mahler L, Chalas E. A randomized controlled trial of early postoperative feeding in gynecologic oncology patients undergoing intraabdominal surgery; pages 94-97; copyright 1998; with permission from American College of Obstetricians and Gynecologists. | ||
Nasogastric tubes only when indicated
The use of NG tubes after laparotomy has been studied extensively. A review of the literature suggests that routine placement of the tubes in asymptomatic patients is not justified and may possibly be harmful.10
In their meta-analysis of the issue, Cheatham et al11 showed that although abdominal distension and vomiting are more frequent in patients who forgo NG tubes postoperatively, fever, atelectasis, and pneumonia are less common, and the interval between surgery and oral feeding is reduced (TABLE 2).
The authors concluded that for every NG tube inserted after abdominal surgery, at least 20 patients can be managed without it.
Forgoing an NG tube also lowers the risk of pulmonary complications, which increases 10-fold when a tube is inserted.12
TABLE 2
Complications associated with selective versus routine NG tube placement
| SELECTIVE PLACEMENT (N) | ROUTINE PLACEMENT (N) | P VALUE | RELATIVE RISK | |
|---|---|---|---|---|
| Patients | 1,986 | 1,978 | ||
| Tubes placed/replaced | 103 | 36 | <.001> | 2.9 |
| Complications | 833 | 1,084 | .03 | 0.76 |
| Deaths | 13 | 25 | .22 | 0.36 |
| Pneumonia | 53 | 119 | <.0001> | 0.49 |
| Atelectasis | 44 | 94 | .001 | 0.46 |
| Fever | 108 | 212 | .02 | 0.51 |
| Vomiting | 201 | 168 | .11 | 1.19 |
| Oral feedings (days) | 3.53 | 4.59 | .04 | |
| Length of stay (days) | 9.32 | 10.1 | .22 | |
| NG=nasogastric | ||||
| Reprinted with permission from Cheatham ML, Chapman WC, Key SP, Sawyer JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg. 1995:221:469-478. | ||||
Continue the epidural anesthetic
A number of experts believe that postoperative ileus is caused by stimulation of neural reflexes, which appear to be of 2 kinds: afferent stimuli to the spinal cord and efferent stimuli to the intestines through the sympathetic nervous system. The latter inhibits motility of the intestinal tract. Numerous studies demonstrate that this sympathetic reflex can be blocked by the use of epidural anesthesia.10
For example, Holte et al4 found that postoperative administration of thoracic epidural blockade with local anesthesia significantly reduced both ileus and pulmonary complications. They concluded that continuous epidural anesthesia with local anesthesia and minimally invasive surgery are the 2 most critical events in reducing postoperative ileus.
In a Cochrane review, Jorgensen and colleagues12 compared the effects of epidural local anesthesia and opioid-based analgesic regimens on postoperative gastrointestinal paralysis, nausea and vomiting, and pain after abdominal surgery (TABLE 3). Epidural local anesthetics reduced gastrointestinal paralysis, as compared with systemic or epidural opioids, but provided the same postoperative pain relief. They also found that the addition of opioids to local epidural anesthesia provided superior postoperative analgesia—compared with epidural local anesthetics alone—without increasing the likelihood of ileus.
A study10 of patients undergoing colectomy found postoperative ileus was prevented or decreased with a 2-day regimen that included:
- continuous thoracic epidural anesthesia for 48 hours;
- withholding NG tubes;
- having the patient drink a liter of fluid on the day of surgery;
- initiating feeding after 24 hours;
- administering milk of magnesia; and
- mobilization after 8 hours, if possible.
Physicians in this study also used transverse surgical incisions to reduce pain and pulmonary problems.
TABLE 3
Anesthetic effect on GI function, postoperative pain, and nausea and vomiting: A comparison
| ANESTHETIC | GI FUNCTION RETURNS | PAIN RELIEF | NAUSEA AND VOMITING |
|---|---|---|---|
| Epidural plus local | 24 hr | Comparable | No significant difference |
| Epidural plus opioids | 37 hr | Comparable | No significant difference |
| Systemic plus opioids | 37 hr | Comparable | No significant difference |
| GI=gastrointestinal | |||
| Data from Jorgensen H, et al12 | |||
Clinical recommendations
The deregulation of the autonomic nervous system during surgery alters the gastrointestinal tract postoperatively, with neurotransmitters, local factors, and hormones playing a large role. Some forms of anesthesia also contribute to postoperative ileus, as does the use of narcotic analgesia after surgery.
The most efficient ways to activate the bowel postoperatively are:
- Continuing the thoracic epidural from 24 to 48 hours, which increases the splanchnic blood flow and blocks afferent and efferent sympathetic inhibitory nerve impulses. Note, however, that comparative studies of thoracic epidural anesthesia with local anesthesia are needed to quantify its impact.
- Hydrating the patient with a large amount of fluid in the first 24 hours after surgery.
Following these steps routinely can significantly decrease the risk of postoperative ileus and thus its resulting complications.
Dr. Rosenman reports no financial relationship with any companies whose products are mentioned in this article.
1. Groudine S. Use of inexpensive anesthesia during surgery may shorten hospital stay. Anesth Analg. 1998;87:1212-1213.
2. Cutillo G, Maneschi F, Franchi M, Giannice R, Scambia G, Benedetti-Panici P. Early feeding compared with nasogastric decompression after major gynecologic surgery: a randomized study. Obstet Gynecol. 1999;93:41-45.
3. Luckey A, Livingston E, Tache Y. Mechanisms and treatment of postoperative ileus. Arch Surg. 2000;138:206-214.
4. Holte K, Kehlet H. Postoperative ileus: a preventable event. Br J Surg. 2000;87:1480-1493.
5. Mangesi L, Hofmeyr GJ. Early compared with delayed oral fluids and food after caesarean section. Cochrane Database Syst Rev. 2002;(3):CD003516.-
6. Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg. 1998;228,5:652-663.
7. MacMillan SL, Kammerer-Doak D, Rogers RG, Parker KM. Early feeding and the incidence of gastrointestinal symptoms after major gynecologic surgery. Obstet Gynecol. 2000;96:604-608.
8. Pearl ML, Valea FA, Fischer M, Mahler L, Chalas E. A randomized controlled trial of early postoperative feeding in gynecologic oncology patients undergoing intraabdominal surgery. Obstet Gynecol. 1998;92:94-97.
9. Schilder JM, Hurteau JA, Look KY, Moore DH, Raff G, Stehman FB, Sutton GP. A prospective controlled trial of early postoperative oral intake following major abdominal gynecologic surgery. Gynecol Oncol. 1997;67:235-240.
10. Kehlet H, Holte K. Review of postoperative ileus. Am J Surg. 2001;182(5a Suppl):3S-10S.
11. Cheatham ML, Chapman WC, Key SP, Sawyer JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg. 1995;221:469-478.
12. Jorgensen H, Wetterslev J, Moiniche S, Dahl JB. Epidural local anesthetics versus opioid-based analgesic regimens on postoperative gastrointestinal paralysis, PONV and pain after abdominal surgery. Cochrane Database Syst Rev. 2000;(4):CD001893.-
1. Groudine S. Use of inexpensive anesthesia during surgery may shorten hospital stay. Anesth Analg. 1998;87:1212-1213.
2. Cutillo G, Maneschi F, Franchi M, Giannice R, Scambia G, Benedetti-Panici P. Early feeding compared with nasogastric decompression after major gynecologic surgery: a randomized study. Obstet Gynecol. 1999;93:41-45.
3. Luckey A, Livingston E, Tache Y. Mechanisms and treatment of postoperative ileus. Arch Surg. 2000;138:206-214.
4. Holte K, Kehlet H. Postoperative ileus: a preventable event. Br J Surg. 2000;87:1480-1493.
5. Mangesi L, Hofmeyr GJ. Early compared with delayed oral fluids and food after caesarean section. Cochrane Database Syst Rev. 2002;(3):CD003516.-
6. Kalff JC, Schraut WH, Simmons RL, Bauer AJ. Surgical manipulation of the gut elicits an intestinal muscularis inflammatory response resulting in postsurgical ileus. Ann Surg. 1998;228,5:652-663.
7. MacMillan SL, Kammerer-Doak D, Rogers RG, Parker KM. Early feeding and the incidence of gastrointestinal symptoms after major gynecologic surgery. Obstet Gynecol. 2000;96:604-608.
8. Pearl ML, Valea FA, Fischer M, Mahler L, Chalas E. A randomized controlled trial of early postoperative feeding in gynecologic oncology patients undergoing intraabdominal surgery. Obstet Gynecol. 1998;92:94-97.
9. Schilder JM, Hurteau JA, Look KY, Moore DH, Raff G, Stehman FB, Sutton GP. A prospective controlled trial of early postoperative oral intake following major abdominal gynecologic surgery. Gynecol Oncol. 1997;67:235-240.
10. Kehlet H, Holte K. Review of postoperative ileus. Am J Surg. 2001;182(5a Suppl):3S-10S.
11. Cheatham ML, Chapman WC, Key SP, Sawyer JL. A meta-analysis of selective versus routine nasogastric decompression after elective laparotomy. Ann Surg. 1995;221:469-478.
12. Jorgensen H, Wetterslev J, Moiniche S, Dahl JB. Epidural local anesthetics versus opioid-based analgesic regimens on postoperative gastrointestinal paralysis, PONV and pain after abdominal surgery. Cochrane Database Syst Rev. 2000;(4):CD001893.-