User login
Report Offers Practice Management Roadmap for Hospital Medicine Groups
A new white paper from the Society of Hospital Medicine (SHM) is the specialty's first formal blueprint on best practices for running a hospital medicine group (HMG).
"The Key Principles and Characteristics of an Effective Hospital Medicine Group: An Assessment Guide for Hospitals and Hospitalists" report published this week in the Journal of Hospital Medicine, is the culmination of a two-year effort to give group leaders and hospital executives a self-assessment tool, says former SHM President John Nelson, MD, MHM, a practicing hospitalist and principal in Nelson Flores Hospital Medicine Consultants in Bellevue, Wash.
Dr. Nelson, one of the report's authors, says the 10 guiding principles and 47 individual characteristics are a launching point for group leaders, C-suite administrators, and others to discuss what ideals apply to their respective practices.
"It's like reading any other thing about how to get fit, how to eat a healthy diet, how to lead a good life," he adds. "It's pretty hard to rigidly pursue everything that an expert or well—considered document might recommend, but you tend to adapt it to your own circumstances and some things resonate."
Culled from more than 200 stakeholders, the report's 10 principles focus on:
Dr. Nelson says it's important for group leaders and hospital executives not to negatively view their practice in light of the report. Because characteristics recommended by the paper won’t necessarily apply everywhere, he suggests instead that readers pick out a handful of principles that apply most specifically to them.
"Two or three might be the right number to zero in on, but those are likely to be different for different groups," he adds. "I don't think there's any way for this to be used in the same manner at each group."
Likewise, Patrick Cawley, MD, MHM, chief executive officer at the Medical University of South Carolina (MUSC) Medical Center in Charleston, S.C. and author of the paper, says it shouldn't be viewed as a scientific conclusion, but as an aspirational approach to improvement.
"We feel that hospital medicine can be better than it is today," says Dr. Cawley, a former SHM president, "by laying out a road map not only [for HMGs] but [also for] hospital medicine leaders and hospital leaders about the things they should concentrate on raises the bar for everybody."
Dr. Cawley says the report is a first step for physicians and executives looking to benchmark their practices. In the future, SHM could follow up with other assessment tools that help groups improve themselves further.
"There are leaders and hospitals trying to improve their group. They're looking for something to measure themselves against," he adds. "This is instantly available for them, and we think this will be self-fulfilling. If people aren't using it, we haven't done our job right."
Richard Quinn is a freelance author in New Jersey.
Visit our website for more information on hospital medicine improvement initiatives.
A new white paper from the Society of Hospital Medicine (SHM) is the specialty's first formal blueprint on best practices for running a hospital medicine group (HMG).
"The Key Principles and Characteristics of an Effective Hospital Medicine Group: An Assessment Guide for Hospitals and Hospitalists" report published this week in the Journal of Hospital Medicine, is the culmination of a two-year effort to give group leaders and hospital executives a self-assessment tool, says former SHM President John Nelson, MD, MHM, a practicing hospitalist and principal in Nelson Flores Hospital Medicine Consultants in Bellevue, Wash.
Dr. Nelson, one of the report's authors, says the 10 guiding principles and 47 individual characteristics are a launching point for group leaders, C-suite administrators, and others to discuss what ideals apply to their respective practices.
"It's like reading any other thing about how to get fit, how to eat a healthy diet, how to lead a good life," he adds. "It's pretty hard to rigidly pursue everything that an expert or well—considered document might recommend, but you tend to adapt it to your own circumstances and some things resonate."
Culled from more than 200 stakeholders, the report's 10 principles focus on:
Dr. Nelson says it's important for group leaders and hospital executives not to negatively view their practice in light of the report. Because characteristics recommended by the paper won’t necessarily apply everywhere, he suggests instead that readers pick out a handful of principles that apply most specifically to them.
"Two or three might be the right number to zero in on, but those are likely to be different for different groups," he adds. "I don't think there's any way for this to be used in the same manner at each group."
Likewise, Patrick Cawley, MD, MHM, chief executive officer at the Medical University of South Carolina (MUSC) Medical Center in Charleston, S.C. and author of the paper, says it shouldn't be viewed as a scientific conclusion, but as an aspirational approach to improvement.
"We feel that hospital medicine can be better than it is today," says Dr. Cawley, a former SHM president, "by laying out a road map not only [for HMGs] but [also for] hospital medicine leaders and hospital leaders about the things they should concentrate on raises the bar for everybody."
Dr. Cawley says the report is a first step for physicians and executives looking to benchmark their practices. In the future, SHM could follow up with other assessment tools that help groups improve themselves further.
"There are leaders and hospitals trying to improve their group. They're looking for something to measure themselves against," he adds. "This is instantly available for them, and we think this will be self-fulfilling. If people aren't using it, we haven't done our job right."
Richard Quinn is a freelance author in New Jersey.
Visit our website for more information on hospital medicine improvement initiatives.
A new white paper from the Society of Hospital Medicine (SHM) is the specialty's first formal blueprint on best practices for running a hospital medicine group (HMG).
"The Key Principles and Characteristics of an Effective Hospital Medicine Group: An Assessment Guide for Hospitals and Hospitalists" report published this week in the Journal of Hospital Medicine, is the culmination of a two-year effort to give group leaders and hospital executives a self-assessment tool, says former SHM President John Nelson, MD, MHM, a practicing hospitalist and principal in Nelson Flores Hospital Medicine Consultants in Bellevue, Wash.
Dr. Nelson, one of the report's authors, says the 10 guiding principles and 47 individual characteristics are a launching point for group leaders, C-suite administrators, and others to discuss what ideals apply to their respective practices.
"It's like reading any other thing about how to get fit, how to eat a healthy diet, how to lead a good life," he adds. "It's pretty hard to rigidly pursue everything that an expert or well—considered document might recommend, but you tend to adapt it to your own circumstances and some things resonate."
Culled from more than 200 stakeholders, the report's 10 principles focus on:
Dr. Nelson says it's important for group leaders and hospital executives not to negatively view their practice in light of the report. Because characteristics recommended by the paper won’t necessarily apply everywhere, he suggests instead that readers pick out a handful of principles that apply most specifically to them.
"Two or three might be the right number to zero in on, but those are likely to be different for different groups," he adds. "I don't think there's any way for this to be used in the same manner at each group."
Likewise, Patrick Cawley, MD, MHM, chief executive officer at the Medical University of South Carolina (MUSC) Medical Center in Charleston, S.C. and author of the paper, says it shouldn't be viewed as a scientific conclusion, but as an aspirational approach to improvement.
"We feel that hospital medicine can be better than it is today," says Dr. Cawley, a former SHM president, "by laying out a road map not only [for HMGs] but [also for] hospital medicine leaders and hospital leaders about the things they should concentrate on raises the bar for everybody."
Dr. Cawley says the report is a first step for physicians and executives looking to benchmark their practices. In the future, SHM could follow up with other assessment tools that help groups improve themselves further.
"There are leaders and hospitals trying to improve their group. They're looking for something to measure themselves against," he adds. "This is instantly available for them, and we think this will be self-fulfilling. If people aren't using it, we haven't done our job right."
Richard Quinn is a freelance author in New Jersey.
Visit our website for more information on hospital medicine improvement initiatives.
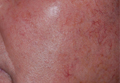
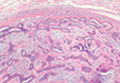
Ulerythema Ophryogenes: Updates and Insights
Test your knowledge on ulerythema ophryogenes with MD-IQ: the medical intelligence quiz. Click here to answer 5 questions.
Consensus Recommendations From the American Acne & Rosacea Society on the Management of Rosacea, Part 4: A Status Report on Physical Modalities and Devices
Chondroid Syringoma
What's Eating You? Turkey Mite and Lone Star Tick (Amblyomma americanum)
Polymer could improve blood cryopreservation

Credit: UAB Hospital
Scientists have found that a common polymer can help red blood cells (RBCs) survive storage at freezing temperatures, and it may offer benefits over current methods of cryopreservation.
The polymer, polyvinyl alcohol, mimics antifreeze properties found in cold-acclimatized fish, such as arctic cod.
And experiments revealed that using polyvinyl alcohol in blood cryopreservation can inhibit the growth of ice crystals, which damage RBCs and make them unusable.
Matthew Gibson, PhD, of the University of Warwick in the UK, and his colleagues conducted these experiments and reported the results in Nature Communications.
“We know that certain types of fish survive perfectly well in sub-zero sea temperatures without their blood freezing,” Dr Gibson said. “We used this as a starting point to search for synthetic substances which reflect what nature already does so well.”
“On closer examination, it turns out that polyvinyl alcohol, which is actually a derivative of wood glue, mimics the properties of the antifreeze proteins found in these kinds of fish.”
So Dr Gibson and his colleagues decided to see how polyvinyl alcohol fared in blood cryopreservation.
The team tested RBCs from sheep and humans and found that polyvinyl alcohol could inhibit ice crystal growth, even when used at concentrations as low as 0.01wt%.
The polymer was most effective at 0.1wt%, which allowed for 40% RBC recovery. Higher concentrations of polyvinyl alcohol did reduce the growth of ice crystals, but the benefits were counteracted by the secondary effects of ice shaping, which can pierce cell membranes.
The researchers noted that the current method of cryopreservation typically requires more than 20wt% of organic solvents to prevent ice formation. And the solvents must be removed before the blood can be used.
“Polyvinyl alcohol has 3 things in its favor when applied to freezing blood,” Dr Gibson said. “Firstly, it reduces the growth of ice crystals during thawing. Secondly, it reduces the need for organic solvents, and, crucially, it reduces the time between defrosting and having transfusion-ready blood by eliminating the need to remove solvent.”
Dr Gibson pointed out that, although polyvinyl alcohol appears to be a promising option for cryopreservation, additional research is needed. But if the polymer proves effective in subsequent studies, it could be used on other cell types as well. ![]()

Credit: UAB Hospital
Scientists have found that a common polymer can help red blood cells (RBCs) survive storage at freezing temperatures, and it may offer benefits over current methods of cryopreservation.
The polymer, polyvinyl alcohol, mimics antifreeze properties found in cold-acclimatized fish, such as arctic cod.
And experiments revealed that using polyvinyl alcohol in blood cryopreservation can inhibit the growth of ice crystals, which damage RBCs and make them unusable.
Matthew Gibson, PhD, of the University of Warwick in the UK, and his colleagues conducted these experiments and reported the results in Nature Communications.
“We know that certain types of fish survive perfectly well in sub-zero sea temperatures without their blood freezing,” Dr Gibson said. “We used this as a starting point to search for synthetic substances which reflect what nature already does so well.”
“On closer examination, it turns out that polyvinyl alcohol, which is actually a derivative of wood glue, mimics the properties of the antifreeze proteins found in these kinds of fish.”
So Dr Gibson and his colleagues decided to see how polyvinyl alcohol fared in blood cryopreservation.
The team tested RBCs from sheep and humans and found that polyvinyl alcohol could inhibit ice crystal growth, even when used at concentrations as low as 0.01wt%.
The polymer was most effective at 0.1wt%, which allowed for 40% RBC recovery. Higher concentrations of polyvinyl alcohol did reduce the growth of ice crystals, but the benefits were counteracted by the secondary effects of ice shaping, which can pierce cell membranes.
The researchers noted that the current method of cryopreservation typically requires more than 20wt% of organic solvents to prevent ice formation. And the solvents must be removed before the blood can be used.
“Polyvinyl alcohol has 3 things in its favor when applied to freezing blood,” Dr Gibson said. “Firstly, it reduces the growth of ice crystals during thawing. Secondly, it reduces the need for organic solvents, and, crucially, it reduces the time between defrosting and having transfusion-ready blood by eliminating the need to remove solvent.”
Dr Gibson pointed out that, although polyvinyl alcohol appears to be a promising option for cryopreservation, additional research is needed. But if the polymer proves effective in subsequent studies, it could be used on other cell types as well. ![]()

Credit: UAB Hospital
Scientists have found that a common polymer can help red blood cells (RBCs) survive storage at freezing temperatures, and it may offer benefits over current methods of cryopreservation.
The polymer, polyvinyl alcohol, mimics antifreeze properties found in cold-acclimatized fish, such as arctic cod.
And experiments revealed that using polyvinyl alcohol in blood cryopreservation can inhibit the growth of ice crystals, which damage RBCs and make them unusable.
Matthew Gibson, PhD, of the University of Warwick in the UK, and his colleagues conducted these experiments and reported the results in Nature Communications.
“We know that certain types of fish survive perfectly well in sub-zero sea temperatures without their blood freezing,” Dr Gibson said. “We used this as a starting point to search for synthetic substances which reflect what nature already does so well.”
“On closer examination, it turns out that polyvinyl alcohol, which is actually a derivative of wood glue, mimics the properties of the antifreeze proteins found in these kinds of fish.”
So Dr Gibson and his colleagues decided to see how polyvinyl alcohol fared in blood cryopreservation.
The team tested RBCs from sheep and humans and found that polyvinyl alcohol could inhibit ice crystal growth, even when used at concentrations as low as 0.01wt%.
The polymer was most effective at 0.1wt%, which allowed for 40% RBC recovery. Higher concentrations of polyvinyl alcohol did reduce the growth of ice crystals, but the benefits were counteracted by the secondary effects of ice shaping, which can pierce cell membranes.
The researchers noted that the current method of cryopreservation typically requires more than 20wt% of organic solvents to prevent ice formation. And the solvents must be removed before the blood can be used.
“Polyvinyl alcohol has 3 things in its favor when applied to freezing blood,” Dr Gibson said. “Firstly, it reduces the growth of ice crystals during thawing. Secondly, it reduces the need for organic solvents, and, crucially, it reduces the time between defrosting and having transfusion-ready blood by eliminating the need to remove solvent.”
Dr Gibson pointed out that, although polyvinyl alcohol appears to be a promising option for cryopreservation, additional research is needed. But if the polymer proves effective in subsequent studies, it could be used on other cell types as well. ![]()
ASCO explains how ACA impacts trial participation

patient and her father
Credit: Rhoda Baer
The Affordable Care Act (ACA) includes provisions that may make it easier for US cancer patients to participate in clinical trials.
But this aspect of the law has not received much attention and is not well understood, according to the American Society of Clinical Oncology (ASCO).
So the organization has created educational materials for providers and patients that explain ACA coverage as it relates to clinical trials.
“ASCO and other groups fought long and hard for this law requiring insurers nationwide to cover the routine costs of care for individuals participating in clinical trials,” said ASCO President Clifford A. Hudis, MD, FACP.
“Healthcare providers, financial counselors, and others involved in helping patients need to understand the law’s provisions so that their patients can benefit and we can make scientific progress.”
The law, which is effective for health plans newly issued or renewed after January 1, 2014, prohibits health plans or insurance issuers from:
- Denying participation of beneficiaries in clinical trials
- Denying or limiting coverage of routine patient care costs, subject to the plan’s out-of-network coverage policy
- Discriminating against the individual on the basis of participation in a trial.
The federal government has not yet issued regulations to guide implementation of the law. While much of the statutory language is clear, in the absence of federal guidance, payers will likely vary on the legal interpretation of each element of the provision.
Therefore, understanding the provisions can help patients and their healthcare providers seek the best available treatment options, which may include participation in a clinical trial.
Provider materials are available on ASCO’s Clinical Trials Coverage web page at www.ASCO.org/ClinicalTrialsCoverage.
Patient materials available on Cancer.Net include a detailed article explaining health insurance coverage of clinical trials at www.cancer.net/clinicaltrials and a fact sheet that provides an overview of the ACA provision at www.cancer.net/factsheets. ![]()

patient and her father
Credit: Rhoda Baer
The Affordable Care Act (ACA) includes provisions that may make it easier for US cancer patients to participate in clinical trials.
But this aspect of the law has not received much attention and is not well understood, according to the American Society of Clinical Oncology (ASCO).
So the organization has created educational materials for providers and patients that explain ACA coverage as it relates to clinical trials.
“ASCO and other groups fought long and hard for this law requiring insurers nationwide to cover the routine costs of care for individuals participating in clinical trials,” said ASCO President Clifford A. Hudis, MD, FACP.
“Healthcare providers, financial counselors, and others involved in helping patients need to understand the law’s provisions so that their patients can benefit and we can make scientific progress.”
The law, which is effective for health plans newly issued or renewed after January 1, 2014, prohibits health plans or insurance issuers from:
- Denying participation of beneficiaries in clinical trials
- Denying or limiting coverage of routine patient care costs, subject to the plan’s out-of-network coverage policy
- Discriminating against the individual on the basis of participation in a trial.
The federal government has not yet issued regulations to guide implementation of the law. While much of the statutory language is clear, in the absence of federal guidance, payers will likely vary on the legal interpretation of each element of the provision.
Therefore, understanding the provisions can help patients and their healthcare providers seek the best available treatment options, which may include participation in a clinical trial.
Provider materials are available on ASCO’s Clinical Trials Coverage web page at www.ASCO.org/ClinicalTrialsCoverage.
Patient materials available on Cancer.Net include a detailed article explaining health insurance coverage of clinical trials at www.cancer.net/clinicaltrials and a fact sheet that provides an overview of the ACA provision at www.cancer.net/factsheets. ![]()

patient and her father
Credit: Rhoda Baer
The Affordable Care Act (ACA) includes provisions that may make it easier for US cancer patients to participate in clinical trials.
But this aspect of the law has not received much attention and is not well understood, according to the American Society of Clinical Oncology (ASCO).
So the organization has created educational materials for providers and patients that explain ACA coverage as it relates to clinical trials.
“ASCO and other groups fought long and hard for this law requiring insurers nationwide to cover the routine costs of care for individuals participating in clinical trials,” said ASCO President Clifford A. Hudis, MD, FACP.
“Healthcare providers, financial counselors, and others involved in helping patients need to understand the law’s provisions so that their patients can benefit and we can make scientific progress.”
The law, which is effective for health plans newly issued or renewed after January 1, 2014, prohibits health plans or insurance issuers from:
- Denying participation of beneficiaries in clinical trials
- Denying or limiting coverage of routine patient care costs, subject to the plan’s out-of-network coverage policy
- Discriminating against the individual on the basis of participation in a trial.
The federal government has not yet issued regulations to guide implementation of the law. While much of the statutory language is clear, in the absence of federal guidance, payers will likely vary on the legal interpretation of each element of the provision.
Therefore, understanding the provisions can help patients and their healthcare providers seek the best available treatment options, which may include participation in a clinical trial.
Provider materials are available on ASCO’s Clinical Trials Coverage web page at www.ASCO.org/ClinicalTrialsCoverage.
Patient materials available on Cancer.Net include a detailed article explaining health insurance coverage of clinical trials at www.cancer.net/clinicaltrials and a fact sheet that provides an overview of the ACA provision at www.cancer.net/factsheets. ![]()
Affluence seems to affect CML survival in the UK

Credit: CDC
Results of population-based research suggest that financial status may affect survival in patients with chronic myeloid leukemia (CML) living in the UK.
The study showed that, despite equal access to the same clinical care and treatment, survival rates were significantly lower for patients living in more deprived areas.
The researchers said this difference might be explained by lower rates of treatment compliance in the less affluent population.
“These findings highlight the importance of conducting comprehensive, population-based studies to examine treatment pathways across the entire patient population, rather than solely concentrating on findings from clinical trials,” said study author Alexandra Smith, PhD, of the University of York in the UK.
She and her colleagues recounted their findings in BMJ Open.
The team analyzed data from 242 patients who were diagnosed with CML from September 2004 to August 2011. Ninety-seven percent of patients had chronic-phase disease at presentation, and 86% were Ph-positive.
Fifty-five percent of patients were younger than 60 at diagnosis, and 60% were male. Fifty-nine percent lived in deprivation quintiles 1 to 3, and 41% lived in the less affluent quintiles 4 and 5.
Ninety-seven percent of patients received treatment with tyrosine kinase inhibitors (TKIs)—94% imatinib and the rest dasatinib. Three percent of patients were not treated with TKIs due to death, relocation, refusal, a more serious competing comorbidity, or the use of supportive care alone.
Factors affecting survival
The minimum follow-up was 1.5 years, and the maximum was 8.5 years. The overall 5-year survival was 79%. And the relative survival, which took into account the background mortality in the general population, was 89%.
The relative survival curves did not differ significantly between the 2 age groups. Five-year relative survival was 90% for patients younger than 60 and 87% for those older than 60.
Gender also had little impact on relative survival. The 5-year rates were 90% for men and 89% for women.
However, relative survival differed significantly according to affluence. The 5-year relative survival was 95% for the most affluent patients (quintiles 1 to 3) and 80% for the least affluent (quintiles 4 and 5).
Although 41% of all patients lived in the less affluent areas, this group accounted for about 60% of the deaths.
The researchers said this finding could not be attributed to biological features of disease or access to therapy. But they believe a lack of treatment compliance could be the cause.
“We suspect a major factor is that we are not supporting patients sufficiently to allow them to be fully compliant with a treatment that needs to be taken every day to be effective,” said Russell Patmore, MD, of Castle Hill Hospital in the UK.
“We would encourage all teams treating patients with CML to use these findings to focus their resource where it is likely to be most beneficial. This includes helping patients to manage their CML by explaining fully the importance of daily treatment and providing easy access to ongoing support.” ![]()

Credit: CDC
Results of population-based research suggest that financial status may affect survival in patients with chronic myeloid leukemia (CML) living in the UK.
The study showed that, despite equal access to the same clinical care and treatment, survival rates were significantly lower for patients living in more deprived areas.
The researchers said this difference might be explained by lower rates of treatment compliance in the less affluent population.
“These findings highlight the importance of conducting comprehensive, population-based studies to examine treatment pathways across the entire patient population, rather than solely concentrating on findings from clinical trials,” said study author Alexandra Smith, PhD, of the University of York in the UK.
She and her colleagues recounted their findings in BMJ Open.
The team analyzed data from 242 patients who were diagnosed with CML from September 2004 to August 2011. Ninety-seven percent of patients had chronic-phase disease at presentation, and 86% were Ph-positive.
Fifty-five percent of patients were younger than 60 at diagnosis, and 60% were male. Fifty-nine percent lived in deprivation quintiles 1 to 3, and 41% lived in the less affluent quintiles 4 and 5.
Ninety-seven percent of patients received treatment with tyrosine kinase inhibitors (TKIs)—94% imatinib and the rest dasatinib. Three percent of patients were not treated with TKIs due to death, relocation, refusal, a more serious competing comorbidity, or the use of supportive care alone.
Factors affecting survival
The minimum follow-up was 1.5 years, and the maximum was 8.5 years. The overall 5-year survival was 79%. And the relative survival, which took into account the background mortality in the general population, was 89%.
The relative survival curves did not differ significantly between the 2 age groups. Five-year relative survival was 90% for patients younger than 60 and 87% for those older than 60.
Gender also had little impact on relative survival. The 5-year rates were 90% for men and 89% for women.
However, relative survival differed significantly according to affluence. The 5-year relative survival was 95% for the most affluent patients (quintiles 1 to 3) and 80% for the least affluent (quintiles 4 and 5).
Although 41% of all patients lived in the less affluent areas, this group accounted for about 60% of the deaths.
The researchers said this finding could not be attributed to biological features of disease or access to therapy. But they believe a lack of treatment compliance could be the cause.
“We suspect a major factor is that we are not supporting patients sufficiently to allow them to be fully compliant with a treatment that needs to be taken every day to be effective,” said Russell Patmore, MD, of Castle Hill Hospital in the UK.
“We would encourage all teams treating patients with CML to use these findings to focus their resource where it is likely to be most beneficial. This includes helping patients to manage their CML by explaining fully the importance of daily treatment and providing easy access to ongoing support.” ![]()

Credit: CDC
Results of population-based research suggest that financial status may affect survival in patients with chronic myeloid leukemia (CML) living in the UK.
The study showed that, despite equal access to the same clinical care and treatment, survival rates were significantly lower for patients living in more deprived areas.
The researchers said this difference might be explained by lower rates of treatment compliance in the less affluent population.
“These findings highlight the importance of conducting comprehensive, population-based studies to examine treatment pathways across the entire patient population, rather than solely concentrating on findings from clinical trials,” said study author Alexandra Smith, PhD, of the University of York in the UK.
She and her colleagues recounted their findings in BMJ Open.
The team analyzed data from 242 patients who were diagnosed with CML from September 2004 to August 2011. Ninety-seven percent of patients had chronic-phase disease at presentation, and 86% were Ph-positive.
Fifty-five percent of patients were younger than 60 at diagnosis, and 60% were male. Fifty-nine percent lived in deprivation quintiles 1 to 3, and 41% lived in the less affluent quintiles 4 and 5.
Ninety-seven percent of patients received treatment with tyrosine kinase inhibitors (TKIs)—94% imatinib and the rest dasatinib. Three percent of patients were not treated with TKIs due to death, relocation, refusal, a more serious competing comorbidity, or the use of supportive care alone.
Factors affecting survival
The minimum follow-up was 1.5 years, and the maximum was 8.5 years. The overall 5-year survival was 79%. And the relative survival, which took into account the background mortality in the general population, was 89%.
The relative survival curves did not differ significantly between the 2 age groups. Five-year relative survival was 90% for patients younger than 60 and 87% for those older than 60.
Gender also had little impact on relative survival. The 5-year rates were 90% for men and 89% for women.
However, relative survival differed significantly according to affluence. The 5-year relative survival was 95% for the most affluent patients (quintiles 1 to 3) and 80% for the least affluent (quintiles 4 and 5).
Although 41% of all patients lived in the less affluent areas, this group accounted for about 60% of the deaths.
The researchers said this finding could not be attributed to biological features of disease or access to therapy. But they believe a lack of treatment compliance could be the cause.
“We suspect a major factor is that we are not supporting patients sufficiently to allow them to be fully compliant with a treatment that needs to be taken every day to be effective,” said Russell Patmore, MD, of Castle Hill Hospital in the UK.
“We would encourage all teams treating patients with CML to use these findings to focus their resource where it is likely to be most beneficial. This includes helping patients to manage their CML by explaining fully the importance of daily treatment and providing easy access to ongoing support.” ![]()
Drug gets breakthrough designation for SAA

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for the thrombopoietin receptor agonist eltrombopag (Promacta/Revolade) to treat patients with severe aplastic anemia (SAA) who have had an insufficient response to immunosuppressive therapy (IST).
Eltrombopag is not approved in this setting anywhere in the world, and there are no approved therapies for SAA patients who are unresponsive to initial IST.
Of those patients who are unresponsive to initial IST, approximately 40% die from infection or bleeding within 5 years of their diagnosis.
Breakthrough therapy designation is the newest of the FDA’s programs aimed at accelerating the development and review of drugs for serious or life-threatening conditions. A drug receives the designation when preliminary clinical evidence suggests it may offer substantial improvement over available therapies on at least one clinically significant endpoint.
Eltrombopag was granted breakthrough designation based on results from an open-label, phase 2 study in 43 heavily pretreated SAA patients with an insufficient response to IST. Updated results of this trial were published in December (Desmond et al, Blood 2013).
Patients received varying doses of eltrombopag to improve blood counts. At 3 to 4 months of follow-up, the overall response rate was 40% (17/43), which included 1 tri-lineage and 5 bi-lineage responses.
Most of the 17 patients who remained on eltrombopag in an extension study continued to show improvement, but 3 lost their response. Ultimately, 7 patients had significant increases in neutrophil, red cell, and platelet counts.
Five patients who experienced robust near-normalization of blood counts discontinued the drug and maintained stable counts a median of 13 months after discontinuation (range, 1-15).
Eight patients, including 2 who responded, developed new cytogenetic abnormalities while on eltrombopag. But none have evolved to acute myeloid leukemia to date.
The only dose-limiting toxicity was reversible transaminitis. Two patients had reversible transaminitis related to treatment, and both required dose interruption.
There were no thrombotic events while patients were on treatment, but 1 responding patient developed deep-vein thrombosis 14 months after treatment discontinuation. ![]()

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for the thrombopoietin receptor agonist eltrombopag (Promacta/Revolade) to treat patients with severe aplastic anemia (SAA) who have had an insufficient response to immunosuppressive therapy (IST).
Eltrombopag is not approved in this setting anywhere in the world, and there are no approved therapies for SAA patients who are unresponsive to initial IST.
Of those patients who are unresponsive to initial IST, approximately 40% die from infection or bleeding within 5 years of their diagnosis.
Breakthrough therapy designation is the newest of the FDA’s programs aimed at accelerating the development and review of drugs for serious or life-threatening conditions. A drug receives the designation when preliminary clinical evidence suggests it may offer substantial improvement over available therapies on at least one clinically significant endpoint.
Eltrombopag was granted breakthrough designation based on results from an open-label, phase 2 study in 43 heavily pretreated SAA patients with an insufficient response to IST. Updated results of this trial were published in December (Desmond et al, Blood 2013).
Patients received varying doses of eltrombopag to improve blood counts. At 3 to 4 months of follow-up, the overall response rate was 40% (17/43), which included 1 tri-lineage and 5 bi-lineage responses.
Most of the 17 patients who remained on eltrombopag in an extension study continued to show improvement, but 3 lost their response. Ultimately, 7 patients had significant increases in neutrophil, red cell, and platelet counts.
Five patients who experienced robust near-normalization of blood counts discontinued the drug and maintained stable counts a median of 13 months after discontinuation (range, 1-15).
Eight patients, including 2 who responded, developed new cytogenetic abnormalities while on eltrombopag. But none have evolved to acute myeloid leukemia to date.
The only dose-limiting toxicity was reversible transaminitis. Two patients had reversible transaminitis related to treatment, and both required dose interruption.
There were no thrombotic events while patients were on treatment, but 1 responding patient developed deep-vein thrombosis 14 months after treatment discontinuation. ![]()

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for the thrombopoietin receptor agonist eltrombopag (Promacta/Revolade) to treat patients with severe aplastic anemia (SAA) who have had an insufficient response to immunosuppressive therapy (IST).
Eltrombopag is not approved in this setting anywhere in the world, and there are no approved therapies for SAA patients who are unresponsive to initial IST.
Of those patients who are unresponsive to initial IST, approximately 40% die from infection or bleeding within 5 years of their diagnosis.
Breakthrough therapy designation is the newest of the FDA’s programs aimed at accelerating the development and review of drugs for serious or life-threatening conditions. A drug receives the designation when preliminary clinical evidence suggests it may offer substantial improvement over available therapies on at least one clinically significant endpoint.
Eltrombopag was granted breakthrough designation based on results from an open-label, phase 2 study in 43 heavily pretreated SAA patients with an insufficient response to IST. Updated results of this trial were published in December (Desmond et al, Blood 2013).
Patients received varying doses of eltrombopag to improve blood counts. At 3 to 4 months of follow-up, the overall response rate was 40% (17/43), which included 1 tri-lineage and 5 bi-lineage responses.
Most of the 17 patients who remained on eltrombopag in an extension study continued to show improvement, but 3 lost their response. Ultimately, 7 patients had significant increases in neutrophil, red cell, and platelet counts.
Five patients who experienced robust near-normalization of blood counts discontinued the drug and maintained stable counts a median of 13 months after discontinuation (range, 1-15).
Eight patients, including 2 who responded, developed new cytogenetic abnormalities while on eltrombopag. But none have evolved to acute myeloid leukemia to date.
The only dose-limiting toxicity was reversible transaminitis. Two patients had reversible transaminitis related to treatment, and both required dose interruption.
There were no thrombotic events while patients were on treatment, but 1 responding patient developed deep-vein thrombosis 14 months after treatment discontinuation. ![]()
VTE Codes in Academic Medical Centers
Pulmonary embolism (PE) and deep venous thrombosis (DVT), historically referred to together as venous thromboembolism (VTE), are common, treatable, sometimes fatal, and potentially preventable medical problems.[1] Such thromboses can both precipitate a hospitalization as well as complicate it (either during or soon after discharge). Preventing such thrombosis as a complication of medical care has become a national imperative. Landmark studies such as Prophylaxis in Medical Patients With Enoxaparin (MEDENOX)[2] and Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT)[3] demonstrated both a high incidence of thrombosis in a hospitalized high‐risk medical population (15% and 5% in the 2 trials' placebo arms, respectively) as well as significant relative risk reduction through venous thromboembolism pharmacoprophylaxis (VTEP)63% and 45%, respectively. The Joint Commission,[4] the Society of Hospital Medicine,[5] and the American College of Chest Physicians[6, 7] have thus all strived to ensure the appropriate provision of VTEP in order to reduce the morbidity and mortality associated with thrombosis in hospitalized patients, including those on medical services.
Ideally, the global success of these efforts would be assessed by measuring the rate of hospital‐associated VTE (potentially including superficial venous thrombosis [SVT], which, like upper‐extremity deep venous thrombosis [UE‐DVT], is commonly a central venous catheter [CVC]‐associated, or peripherally inserted central catheter [PICC]‐associated, complication)thrombosis acquired and diagnosed during either the index hospitalization (hospital‐acquired, or HA‐VTE/SVT) or up to 30 days postdischarge. Unfortunately, postdischarge VTE/SVT is difficult to measure because patients developing it may not present to the original hospital, or at all (eg, if they do not seek care, are treated as outpatients, or, in the most extreme case, die at home). In this context, despite being far less comprehensive, HA‐VTE/SVT is a useful subset of hospital‐associated VTE/SVT, for several reasons. First, the Centers for Medicare & Medicaid Services (CMS) have mandated hospitals to qualify all medical diagnoses as present‐on‐admission (POA = Y) or not (POA = N) since 2008, such that all medical diagnoses coded POA = N can be considered hospital acquired.[8] Second, refinements made to the International Classification of Diseases, 9th Revision (ICD‐9) codes now allow differentiation of UE‐DVT and SVT from lower‐extremity (LE) DVT/PE, whereas the former were sometimes obscured by nonspecific coding.[9] Third, recent studies have shown that medical diagnoses administratively coded as HA‐VTE/SVT correlated well with HA‐VTE/SVT ascertained through chart review.[9, 10] Finally, previous work has estimated that approximately half of all hospital‐associated VTE are HA‐VTE and the other half are postdischarge VTE.[11] Thus, HA‐VTE, though comprising only approximately half of all hospital‐associated VTE, is often used as a surrogate for measuring the success of ongoing VTE prevention programs.[12]
Our study aimed to assess the incidence of HA‐VTE plus HA‐SVT in the era of mandatory POA coding and newer ICD‐9 codes for VTE.
METHODS
Setting and Cases
We conducted a retrospective analysis of discharges from the 83 academic medical centers belonging to the UHC (formerly, the University HealthSystem Consortium,
Patients in our analysis were age 18 years and discharged with a medical medical severity diagnostic‐related group (MS‐DRG) code, hospitalized for 48 hours, and did not have a surgical or obstetric MS‐DRG code (except when assigned a surgical MS‐DRG code solely due to insertion of an inferior vena cava filter, with no other major procedures performed). Cases excluded discharges with a principal diagnosis of acute VTE/SVT (defined here as including PE, LE‐DVT, UE‐DVT, SVT, chronic VTE, and thrombosis not otherwise specified), as coding guidelines prohibit assigning a HA‐VTE as the principal diagnosis for the index hospitalization.[14]
Hospital‐Acquired Venous Thromboembolism or Superficial Venous Thrombosis
Cases were classified as having a HA‐VTE/SVT if there was 1 VTE/SVT coded in a secondary diagnosis position (other diagnosis) with a corresponding POA indicator equal to either N (not POA) or U (documentation insufficient to clarify whether VTE was POA or not). This usage corresponds to CMS guidelines and reimbursement policies for hospital‐acquired conditions.[15] Among cases with 1 HA‐VTE (or SVT), we assigned 1 HA‐VTE diagnosis using a hierarchy based on the highest level of clinical importance: first, PE; then LE‐DVT; then UE‐DVT; then SVT; then chronic VTE; then, finally, unspecified VTE. We subsequently excluded cases with primarily chronic VTE from our analysis because these were likely miscodes (ie, it is unclear how a chronic VTE could not be POA) and there were only 30 such cases. Cases with HA‐PE or HA‐LE DVT were analyzed separately as an important subset of HA‐VTE (plus SVT), because HA‐PE/LE‐DVT is both life‐threatening and theoretically preventable with VTEP.
Severity of Illness and Other Measures of Comorbidity
For each case we used proprietary software (3M Health Information Systems, Murray UT) to classify severity of illness (SOI). The SOI scale, based on physiologic derangement and organ system loss of function,[16] has 4 levels: minor, major, severe, and extreme. Defined within specific disease groups (All Patient Refined DRGs), it is often compared across diseases as well.[17] We also assessed whether patients had a cancer diagnosis, spent time in the intensive care unit (ICU), and died in the hospital.
Central Venous Catheter Use in Patients With Upper‐Extremity Deep Venous Thrombosis or Superficial Venous Thrombosis
Because UE‐DVT and SVT are frequently associated with a CVC or PICC, we assessed central venous catheterization among patients with an UE‐DVT or SVT of the cephalic, basilic, or antecubital veins using diagnosis codes for complications related to dialysis devices, implants, and grafts.
Pharmacologic Thromboprophylaxis
Pharmacy records of the subset of HA‐VTE/SVT cases with PE or LE‐DVT were analyzed to determine if VTEP was administered on hospital day 1 or 2, as per Joint Commission performance requirements.[4] Medications that met criteria as VTEP included unfractionated heparin, 5000 IU, given 2 or 3 a day; enoxaparin, 40 mg, given daily; dalteparin, 2500 or 5000 IU, given daily; fondaparinux, 2.5 mg, given daily; and warfarin. We could not reliably determine if VTEP was used throughout the entire hospitalization, or whether mechanical prophylaxis was used at all.
Statistical Analysis
This was a descriptive analysis to determine the incidence of HA‐VTE/SVT and describe the demographic and clinical characteristics of this population. We calculated means and standard deviations (SD) for continuous variables and proportions for binary variables (including HA‐VTE/SVT incidence). All comparisons between populations were performed as either 2‐tailed t tests or 2 analyses. All analysis was conducted using SAS software, version 9.2 (SAS Institute, Inc., Cary, NC).
RESULTS
For the 18‐month period between October 1, 2009, and March 31, 2011, across 83 UHC hospitals, there were 2,525,068 cases. Among these, 12,847 (0.51%) had 1 HA‐VTE/SVT coded. As per the clinical importance hierarchy described above, 2449 (19.1%) cases had at least a PE coded; 3848 (30%) had at least a LE‐DVT (but not a PE) coded; 2893 (22.5%) had at least an UE‐DVT coded; 3248 (25.3%) had at least an SVT coded; 30 had at least a chronic VTE coded; and 379 had at least a VTE coded with no specified location. Of those with SVT, 192 (5.8%) were LE‐SVT codes, whereas the rest were SVT/thrombophlebitis of the upper extremities or not otherwise specified. There were 11,882 (92.5%) hospitalizations with a single HA‐VTE/SVT code and an additional 965 (7.5%) with multiple codes, for a total of 13,749 HA‐VTE/SVT events (see Supporting Information, Table S1, in the online version of this article for more specific data for the individual ICD‐9 codes used to specify HA‐VTE events).
Compared with those who did not develop any HA‐VTE/SVT, patients with HA‐PE/LE‐DVT were more likely to be Caucasian (65% vs 58%, P < 0.001) and were older (age 62 vs 48 years, P < 0.001) and sicker (79.9% vs 44.9% with a severe or extreme SOI, P < 0.001). They also were more likely to have cancer, have longer lengths of stay, be more likely to stay in the ICU, and die in the hospital (P < 0.001 for all comparisons; Table 1).
| Characteristic | No HA‐VTE, n = 2,512,221 | HA‐PE/LE DVT, n = 6,297a | P Valueb |
|---|---|---|---|
| |||
| Proportion of hospitalizations, % | 99.49 | 0.25 | |
| Age, y | 48.2 27.1 | 62.5 20.0 | <0.001 |
| Female sex | 1,347,219 (53.6) | 3,104 (49.3) | <0.001 |
| Race | <0.001 | ||
| Caucasian | 1,455,215 (57.9) | 3,963 (64.7) | |
| Black | 600,991 (23.9) | 1,425 (23.3) | |
| Hispanic | 206,553 (8.2) | 263 (4.3) | |
| API | 59,560 (2.4) | 88 (1.4) | |
| Other | 189,902 (7.6) | 389 (6.4) | |
| Admission SOI | <0.001 | ||
| Minor | 461,411 (18.4) | 181 (2.9) | |
| Major | 922,734 (36.7) | 1,081 (17.2) | |
| Severe | 880,542 (35.1) | 2,975 (47.2) | |
| Extreme | 247,244 (9.8) | 2,060 (32.7) | |
| Unknown | 290 (0.01) | 0 (0.0) | |
| Had an active diagnosis of cancer | 331,705 (13.2) | 2,162 (34.3) | <0.001 |
| Length of stay, d | 7.31 9.31 | 18.7 19.5 | <0.001 |
| Spent time in the ICU | 441,412 (17.6) | 3,011 (47.8) | <0.001 |
| Died in hospital | 57,954 (2.3) | 1,036 (16.5) | <0.001 |
| Received prophylaxisc | c | 3,454 (54.9) | c |
Among cases with a code for UE‐DVT (22.5% of all patients with HA‐VTE), 74% were noted to also have a code for a CVC, as did 60% of cases with a HA‐SVT of the antecubital, basilic, or cephalic veins (71% of SVT events; see Supporting Information, Table S1, in the online version of this article).
Of those with HA‐PE/LE‐DVT, 54.9% received pharmacologic prophylaxis on hospital day 1 or 2 (mostly with low‐molecular‐weight heparin or unfractionated heparin).
DISCUSSION
In this study of medical patients admitted to academic medical centers throughout the United States, we found that HA‐VTE/SVT was coded in approximately 0.51% of discharges, and the incidence of HA‐PE/LE‐DVT was 0.25%. Patients with a HA‐PE/LE‐DVT code were, in general, older and sicker than those who did not develop VTE. We further found that close to half of all HA‐VTE/SVT occurred in the upper extremity, with the majority of these occurring in patients who had CVCs. Finally, the majority of patients diagnosed with HA‐PE/LE‐DVT were started on VTEP on the first or second hospital day.
The overall incidence of HA‐VTE/SVT we discovered corresponds well to other studies, even those with disparate populations. A single‐institution study found a HA‐VTE/SVT incidence of approximately 0.6% among hospitalized patients on medical and nonmedical services.[12] The study by Barba found a rate of 0.93%,[18] whereas the study by Lederle found a rate of approximately 1%.[19] Spyropolous found an HA‐VTE incidence of 0.55%.[11] Rothberg found a lower rate of 0.25% in his risk‐stratification study, though in the pre‐POA and preupdated code era.[20] Our findings extend and provide context for, in a much larger population, the results of these prior studies, and represent the first national examination of HA‐VTE/SVT in the setting of numerous quality‐improvement and other efforts to reduce hospital‐associated VTE.
The incidence of HA‐VTE/SVT codes we observed likely underestimates the incidence of hospital‐associated VTE/SVT by a factor of approximately 4, for 2 reasons. First, although VTE/SVT codes with a POA flag set to No are truly hospital‐acquired events on chart review approximately 75% of the time, and thus overestimate HA‐VTE/SVT, 25% of POA = Yes codes are actually HA‐VTE/SVT events on chart review, and therefore lead to underestimation of HA‐VTE/SVT.[9] Because VTE/SVT codes with a POA flag set to Yes outnumber those flagged No by 3 or 4 to 1, events mis‐flagged Yes contribute a much greater number of undercounted HA‐VTE/SVT, elevating the actual HA‐VTE/SVT event rate by a factor of approximately 2. Second, HA‐VTE events do not include hospital‐associated VTE events that are diagnosed after the index hospitalization. In the Spyropolous study, 45% of hospital‐associated VTE events occurred after discharge, so translating HA‐VTE/SVT events to hospital‐associated VTE/SVT events would again involve multiplying by a factor of 2.[11] Thus, the overall incidence of hospital‐associated VTE/SVT events in our sample may have been approximately 2% (0.51% 4), and the overall incidence of hospital‐associated PE or LE‐DVT events may have been approximately 1%, though there may be significant variation around these estimates given that individual institutions were themselves quite variable in their POA flag accuracy in our study.[9] There is additionally the possibility that hospitals may have deliberately left some VTE/SVT uncoded, but in the absence of financial incentives to do so for anything other than postsurgical VTE, and in the presence of penalties from CMS for undercoding, we believe this to be unlikely, at least at present.
Despite these upward extrapolations, the estimated incidence of hospital‐associated VTE/SVT in our study may seem low compared with that reported in the MEDENOX[2] and PREVENT studies.[3] Much of this discrepancy vanishes on closer examination. In the large randomized trials, patients were uniformly and routinely assessed for LE‐DVT using vascular ultrasound; in contrast, in our population of hospitalizations patients may have only had diagnostic studies done for signs or symptoms. Clinically apparent hospital‐associated VTE is less common than all hospital‐associated VTE, as it was even in PREVENT,[3] and increased surveillance may even be partially driving increased hospital‐associated VTE/SVT at some hospitals.[21] Our findings suggest that success or failure in preventing administratively coded, clinically apparent HA LE‐VTE/PE should be judged, broadly, against numbers in the range established in our study (eg, 0.25%), not the 5% or 15% of chart‐abstracted, aggressively ascertained (and sometimes clinically silent) hospital‐associated VTE in the large randomized controlled trials. That is, 0.25% is not an achievement, but rather the average, expected value.
Almost 25% of the observed HA‐VTE/SVTs coded were UE‐DVT, with roughly 75% of these being likely related to central venous catheterization (including those peripherally inserted). An additional 1/5 were upper‐extremity SVT of the antecubital, cephalic, and basilic veins, with the majority of these (60%) also listed as catheter‐related. Such thrombosis is best prevented by decreased use of central catheters or perhaps by using smaller‐caliber catheters.[22] It is unclear if VTEP can prevent such clots, though in cancer patients at least one recent trial seems promising.[23]
We found that patients with a coded HA‐PE/LE‐DVT were remarkably different from those not developing HA‐VTE/SVT. Patients with HA‐PE or HA‐LE‐DVT were older, sicker, more likely to have cancer, significantly more likely to spend time in the ICU, and much more likely to die in the hospital; risk factors for HA‐VTE overlap significantly with risk factors for death in the hospital. A small majority (55%) of patients in the HA‐PE/LE‐DVT group had actually received VTEP on at least day 1 or 2 of hospitalization. It may be the case that the dose of VTEP was insufficient to suppress clot formation in these patients, or that HA‐PE/LE‐DVT in patients with this degree of comorbidity is difficult to prevent.
There are a number of limitations to our study. We analyzed administrative codes, which underestimate hospital‐associated VTE/SVT events as noted above. This was a descriptive study, cross‐sectional across each hospitalization, and we were unable to draw any causal inference for differences in HA‐VTE/SVT incidence that might exist between subpopulations. We estimated VTEP from medication usage in just the first 2 days of hospitalization; we could not assess mechanical prophylaxis in this dataset; and we did not have any VTEP data for the first 2 days of hospitalization on the patients who did not develop a HA‐VTE/SVT, which made it impossible to compare the 2 populations on this measure. For those who did not receive VTEP, we were unable to obtain data regarding possible contraindications to VTEP, such as ongoing gastrointestinal or intracerebral hemorrhage. Additionally, our data are based on academic hospitals only and may not generalize to nonacademic settings. Extrapolating from HA‐VTE/SVT to hospital‐associated VTE/SVT may not be possible due to heterogeneity of clotting events and perhaps variability in whether patients would return to the hospital for all of them (eg, superficial or UE VTE may not result in readmission). Finally, it is unclear whether a switch to ICD Tenth Revision (ICD‐10) codes will impact our measured baseline in the coming year. The strengths of our analysis included stratification by type of HA‐VTE/SVT and our ability to assess the incidence of HA‐VTE/SVT in a large national population, and the provision of a baseline for VTE incidenceeasily usable by any individual hospital, network, or researcher with access to administrative datagoing forward.
In conclusion, among patients hospitalized in academic medical centers, HA‐VTE/SVT was coded in approximately 0.51% of patients with a medical illness staying >2 days, with approximately half of the events due to HA‐PE/LE‐DVT. Patients who developed HA‐PE/LE‐DVT were more acutely ill than those who did not, and VTE developed despite 55% of these patients receiving VTEP on day 1 or 2. Hospitals can reasonably treat the 0.25% figure as the baseline around which to assess their own performance in preventing HA‐PE/LE‐DVT, and can measure their own performance using administrative data. Further research is needed to determine how best to achieve further reductions in HA‐VTE/SVT through risk stratification and/or through other interventions.
Disclosures
Nothing to report.
- , , , , ;American College of Chest Physicians AntithromboticTherapy and Prevention of Thrombosis Panel. Executive summary: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines [published corrections appear in Chest. 2012;141(4):1129 and 2012;142(6):1698]. Chest. 2012;141(2 suppl):7S–47S.
- , , , et al;Prophylaxis in Medical Patients with Enoxaparin Study Group. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. N Engl J Med. 1999;341(11):793–800.
- , , , , , . Randomized, placebo‐controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110(7):874–879.
- The Joint Commission.Specifications Manual for National Hospital Inpatient Quality Measures. Available at: http://www.jointcommission.org/specifications_manual_for_national_hospital_inpatient_quality_measures.aspx. Accessed July 18, 2012.
- , . Preventing Hospital‐Acquired Venous Thromboembolism: A Guide for Effective Quality Improvement. Prepared by the Society of Hospital Medicine. Rockville, MD: Agency for Healthcare Research and Quality; AHRQ Publication No. 08‐0075.
- , , , et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 suppl):338S–400S.
- , , , et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e195S–e226S.
- Centers for Medicare 46(6 part 1):1946–1962.
- , , , et al. Predictive and associative models to identify hospitalized medical patients at risk for VTE. Chest. 2011;140(3):706–714.
- , , , . Unintended consequences of a standard admission order set on venous thromboembolism prophylaxis and patient outcomes. J Gen Intern Med. 2012;27(3):318–324.
- United Health Consortium Website. Available at: https://www.uhc.edu. Accessed March 8, 2012.
- ICD‐9‐CM Official Guidelines for Coding and Reporting. Available at: http://www.cdc.gov/nchs/data/icd9/icdguide10.pdf. Published 2010. Accessed June 4, 2013.
- Centers for Medicare 27(5):587–612.
- Overview of Disease Severity Measures Disseminated with the Nationwide Inpatient Sample (NIS) and Kids' Inpatient Database (KID). Available at: http://www.hcup‐us.ahrq.gov/db/nation/nis/OverviewofSeveritySystems.pdf. Published December 9, 2005. Accessed June 4, 2013.
- , , , et al. Venous thromboembolism in acutely ill hospitalized medical patients. Thromb Res. 2010;126(4):276–279.
- , , , . Venous thromboembolism prophylaxis in hospitalized medical patients and those with stroke: a background review for an American College of Physicians Clinical Practice Guideline. Ann Intern Med. 2011;155(9):602–615.
- , , , , . Risk factor model to predict venous thromboembolism in hospitalized medical patients. J Hosp Med. 2011;6(4):202–209.
- , , , et al. Evaluation of surveillance bias and the validity of the venous thromboembolism quality measure. JAMA. 2013;310(14):1482–1489.
- , , , et al. Reduction of peripherally inserted central catheter‐associated DVT. Chest. 2013;143(3):627–633.
- , , , et al. Prophylaxis of catheter‐related deep vein thrombosis in cancer patients with low‐dose warfarin, low molecular weight heparin, or control: a randomized, controlled, phase III study. Cancer Chemother Pharmacol. 2013;72(1):65–73.
Pulmonary embolism (PE) and deep venous thrombosis (DVT), historically referred to together as venous thromboembolism (VTE), are common, treatable, sometimes fatal, and potentially preventable medical problems.[1] Such thromboses can both precipitate a hospitalization as well as complicate it (either during or soon after discharge). Preventing such thrombosis as a complication of medical care has become a national imperative. Landmark studies such as Prophylaxis in Medical Patients With Enoxaparin (MEDENOX)[2] and Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT)[3] demonstrated both a high incidence of thrombosis in a hospitalized high‐risk medical population (15% and 5% in the 2 trials' placebo arms, respectively) as well as significant relative risk reduction through venous thromboembolism pharmacoprophylaxis (VTEP)63% and 45%, respectively. The Joint Commission,[4] the Society of Hospital Medicine,[5] and the American College of Chest Physicians[6, 7] have thus all strived to ensure the appropriate provision of VTEP in order to reduce the morbidity and mortality associated with thrombosis in hospitalized patients, including those on medical services.
Ideally, the global success of these efforts would be assessed by measuring the rate of hospital‐associated VTE (potentially including superficial venous thrombosis [SVT], which, like upper‐extremity deep venous thrombosis [UE‐DVT], is commonly a central venous catheter [CVC]‐associated, or peripherally inserted central catheter [PICC]‐associated, complication)thrombosis acquired and diagnosed during either the index hospitalization (hospital‐acquired, or HA‐VTE/SVT) or up to 30 days postdischarge. Unfortunately, postdischarge VTE/SVT is difficult to measure because patients developing it may not present to the original hospital, or at all (eg, if they do not seek care, are treated as outpatients, or, in the most extreme case, die at home). In this context, despite being far less comprehensive, HA‐VTE/SVT is a useful subset of hospital‐associated VTE/SVT, for several reasons. First, the Centers for Medicare & Medicaid Services (CMS) have mandated hospitals to qualify all medical diagnoses as present‐on‐admission (POA = Y) or not (POA = N) since 2008, such that all medical diagnoses coded POA = N can be considered hospital acquired.[8] Second, refinements made to the International Classification of Diseases, 9th Revision (ICD‐9) codes now allow differentiation of UE‐DVT and SVT from lower‐extremity (LE) DVT/PE, whereas the former were sometimes obscured by nonspecific coding.[9] Third, recent studies have shown that medical diagnoses administratively coded as HA‐VTE/SVT correlated well with HA‐VTE/SVT ascertained through chart review.[9, 10] Finally, previous work has estimated that approximately half of all hospital‐associated VTE are HA‐VTE and the other half are postdischarge VTE.[11] Thus, HA‐VTE, though comprising only approximately half of all hospital‐associated VTE, is often used as a surrogate for measuring the success of ongoing VTE prevention programs.[12]
Our study aimed to assess the incidence of HA‐VTE plus HA‐SVT in the era of mandatory POA coding and newer ICD‐9 codes for VTE.
METHODS
Setting and Cases
We conducted a retrospective analysis of discharges from the 83 academic medical centers belonging to the UHC (formerly, the University HealthSystem Consortium,
Patients in our analysis were age 18 years and discharged with a medical medical severity diagnostic‐related group (MS‐DRG) code, hospitalized for 48 hours, and did not have a surgical or obstetric MS‐DRG code (except when assigned a surgical MS‐DRG code solely due to insertion of an inferior vena cava filter, with no other major procedures performed). Cases excluded discharges with a principal diagnosis of acute VTE/SVT (defined here as including PE, LE‐DVT, UE‐DVT, SVT, chronic VTE, and thrombosis not otherwise specified), as coding guidelines prohibit assigning a HA‐VTE as the principal diagnosis for the index hospitalization.[14]
Hospital‐Acquired Venous Thromboembolism or Superficial Venous Thrombosis
Cases were classified as having a HA‐VTE/SVT if there was 1 VTE/SVT coded in a secondary diagnosis position (other diagnosis) with a corresponding POA indicator equal to either N (not POA) or U (documentation insufficient to clarify whether VTE was POA or not). This usage corresponds to CMS guidelines and reimbursement policies for hospital‐acquired conditions.[15] Among cases with 1 HA‐VTE (or SVT), we assigned 1 HA‐VTE diagnosis using a hierarchy based on the highest level of clinical importance: first, PE; then LE‐DVT; then UE‐DVT; then SVT; then chronic VTE; then, finally, unspecified VTE. We subsequently excluded cases with primarily chronic VTE from our analysis because these were likely miscodes (ie, it is unclear how a chronic VTE could not be POA) and there were only 30 such cases. Cases with HA‐PE or HA‐LE DVT were analyzed separately as an important subset of HA‐VTE (plus SVT), because HA‐PE/LE‐DVT is both life‐threatening and theoretically preventable with VTEP.
Severity of Illness and Other Measures of Comorbidity
For each case we used proprietary software (3M Health Information Systems, Murray UT) to classify severity of illness (SOI). The SOI scale, based on physiologic derangement and organ system loss of function,[16] has 4 levels: minor, major, severe, and extreme. Defined within specific disease groups (All Patient Refined DRGs), it is often compared across diseases as well.[17] We also assessed whether patients had a cancer diagnosis, spent time in the intensive care unit (ICU), and died in the hospital.
Central Venous Catheter Use in Patients With Upper‐Extremity Deep Venous Thrombosis or Superficial Venous Thrombosis
Because UE‐DVT and SVT are frequently associated with a CVC or PICC, we assessed central venous catheterization among patients with an UE‐DVT or SVT of the cephalic, basilic, or antecubital veins using diagnosis codes for complications related to dialysis devices, implants, and grafts.
Pharmacologic Thromboprophylaxis
Pharmacy records of the subset of HA‐VTE/SVT cases with PE or LE‐DVT were analyzed to determine if VTEP was administered on hospital day 1 or 2, as per Joint Commission performance requirements.[4] Medications that met criteria as VTEP included unfractionated heparin, 5000 IU, given 2 or 3 a day; enoxaparin, 40 mg, given daily; dalteparin, 2500 or 5000 IU, given daily; fondaparinux, 2.5 mg, given daily; and warfarin. We could not reliably determine if VTEP was used throughout the entire hospitalization, or whether mechanical prophylaxis was used at all.
Statistical Analysis
This was a descriptive analysis to determine the incidence of HA‐VTE/SVT and describe the demographic and clinical characteristics of this population. We calculated means and standard deviations (SD) for continuous variables and proportions for binary variables (including HA‐VTE/SVT incidence). All comparisons between populations were performed as either 2‐tailed t tests or 2 analyses. All analysis was conducted using SAS software, version 9.2 (SAS Institute, Inc., Cary, NC).
RESULTS
For the 18‐month period between October 1, 2009, and March 31, 2011, across 83 UHC hospitals, there were 2,525,068 cases. Among these, 12,847 (0.51%) had 1 HA‐VTE/SVT coded. As per the clinical importance hierarchy described above, 2449 (19.1%) cases had at least a PE coded; 3848 (30%) had at least a LE‐DVT (but not a PE) coded; 2893 (22.5%) had at least an UE‐DVT coded; 3248 (25.3%) had at least an SVT coded; 30 had at least a chronic VTE coded; and 379 had at least a VTE coded with no specified location. Of those with SVT, 192 (5.8%) were LE‐SVT codes, whereas the rest were SVT/thrombophlebitis of the upper extremities or not otherwise specified. There were 11,882 (92.5%) hospitalizations with a single HA‐VTE/SVT code and an additional 965 (7.5%) with multiple codes, for a total of 13,749 HA‐VTE/SVT events (see Supporting Information, Table S1, in the online version of this article for more specific data for the individual ICD‐9 codes used to specify HA‐VTE events).
Compared with those who did not develop any HA‐VTE/SVT, patients with HA‐PE/LE‐DVT were more likely to be Caucasian (65% vs 58%, P < 0.001) and were older (age 62 vs 48 years, P < 0.001) and sicker (79.9% vs 44.9% with a severe or extreme SOI, P < 0.001). They also were more likely to have cancer, have longer lengths of stay, be more likely to stay in the ICU, and die in the hospital (P < 0.001 for all comparisons; Table 1).
| Characteristic | No HA‐VTE, n = 2,512,221 | HA‐PE/LE DVT, n = 6,297a | P Valueb |
|---|---|---|---|
| |||
| Proportion of hospitalizations, % | 99.49 | 0.25 | |
| Age, y | 48.2 27.1 | 62.5 20.0 | <0.001 |
| Female sex | 1,347,219 (53.6) | 3,104 (49.3) | <0.001 |
| Race | <0.001 | ||
| Caucasian | 1,455,215 (57.9) | 3,963 (64.7) | |
| Black | 600,991 (23.9) | 1,425 (23.3) | |
| Hispanic | 206,553 (8.2) | 263 (4.3) | |
| API | 59,560 (2.4) | 88 (1.4) | |
| Other | 189,902 (7.6) | 389 (6.4) | |
| Admission SOI | <0.001 | ||
| Minor | 461,411 (18.4) | 181 (2.9) | |
| Major | 922,734 (36.7) | 1,081 (17.2) | |
| Severe | 880,542 (35.1) | 2,975 (47.2) | |
| Extreme | 247,244 (9.8) | 2,060 (32.7) | |
| Unknown | 290 (0.01) | 0 (0.0) | |
| Had an active diagnosis of cancer | 331,705 (13.2) | 2,162 (34.3) | <0.001 |
| Length of stay, d | 7.31 9.31 | 18.7 19.5 | <0.001 |
| Spent time in the ICU | 441,412 (17.6) | 3,011 (47.8) | <0.001 |
| Died in hospital | 57,954 (2.3) | 1,036 (16.5) | <0.001 |
| Received prophylaxisc | c | 3,454 (54.9) | c |
Among cases with a code for UE‐DVT (22.5% of all patients with HA‐VTE), 74% were noted to also have a code for a CVC, as did 60% of cases with a HA‐SVT of the antecubital, basilic, or cephalic veins (71% of SVT events; see Supporting Information, Table S1, in the online version of this article).
Of those with HA‐PE/LE‐DVT, 54.9% received pharmacologic prophylaxis on hospital day 1 or 2 (mostly with low‐molecular‐weight heparin or unfractionated heparin).
DISCUSSION
In this study of medical patients admitted to academic medical centers throughout the United States, we found that HA‐VTE/SVT was coded in approximately 0.51% of discharges, and the incidence of HA‐PE/LE‐DVT was 0.25%. Patients with a HA‐PE/LE‐DVT code were, in general, older and sicker than those who did not develop VTE. We further found that close to half of all HA‐VTE/SVT occurred in the upper extremity, with the majority of these occurring in patients who had CVCs. Finally, the majority of patients diagnosed with HA‐PE/LE‐DVT were started on VTEP on the first or second hospital day.
The overall incidence of HA‐VTE/SVT we discovered corresponds well to other studies, even those with disparate populations. A single‐institution study found a HA‐VTE/SVT incidence of approximately 0.6% among hospitalized patients on medical and nonmedical services.[12] The study by Barba found a rate of 0.93%,[18] whereas the study by Lederle found a rate of approximately 1%.[19] Spyropolous found an HA‐VTE incidence of 0.55%.[11] Rothberg found a lower rate of 0.25% in his risk‐stratification study, though in the pre‐POA and preupdated code era.[20] Our findings extend and provide context for, in a much larger population, the results of these prior studies, and represent the first national examination of HA‐VTE/SVT in the setting of numerous quality‐improvement and other efforts to reduce hospital‐associated VTE.
The incidence of HA‐VTE/SVT codes we observed likely underestimates the incidence of hospital‐associated VTE/SVT by a factor of approximately 4, for 2 reasons. First, although VTE/SVT codes with a POA flag set to No are truly hospital‐acquired events on chart review approximately 75% of the time, and thus overestimate HA‐VTE/SVT, 25% of POA = Yes codes are actually HA‐VTE/SVT events on chart review, and therefore lead to underestimation of HA‐VTE/SVT.[9] Because VTE/SVT codes with a POA flag set to Yes outnumber those flagged No by 3 or 4 to 1, events mis‐flagged Yes contribute a much greater number of undercounted HA‐VTE/SVT, elevating the actual HA‐VTE/SVT event rate by a factor of approximately 2. Second, HA‐VTE events do not include hospital‐associated VTE events that are diagnosed after the index hospitalization. In the Spyropolous study, 45% of hospital‐associated VTE events occurred after discharge, so translating HA‐VTE/SVT events to hospital‐associated VTE/SVT events would again involve multiplying by a factor of 2.[11] Thus, the overall incidence of hospital‐associated VTE/SVT events in our sample may have been approximately 2% (0.51% 4), and the overall incidence of hospital‐associated PE or LE‐DVT events may have been approximately 1%, though there may be significant variation around these estimates given that individual institutions were themselves quite variable in their POA flag accuracy in our study.[9] There is additionally the possibility that hospitals may have deliberately left some VTE/SVT uncoded, but in the absence of financial incentives to do so for anything other than postsurgical VTE, and in the presence of penalties from CMS for undercoding, we believe this to be unlikely, at least at present.
Despite these upward extrapolations, the estimated incidence of hospital‐associated VTE/SVT in our study may seem low compared with that reported in the MEDENOX[2] and PREVENT studies.[3] Much of this discrepancy vanishes on closer examination. In the large randomized trials, patients were uniformly and routinely assessed for LE‐DVT using vascular ultrasound; in contrast, in our population of hospitalizations patients may have only had diagnostic studies done for signs or symptoms. Clinically apparent hospital‐associated VTE is less common than all hospital‐associated VTE, as it was even in PREVENT,[3] and increased surveillance may even be partially driving increased hospital‐associated VTE/SVT at some hospitals.[21] Our findings suggest that success or failure in preventing administratively coded, clinically apparent HA LE‐VTE/PE should be judged, broadly, against numbers in the range established in our study (eg, 0.25%), not the 5% or 15% of chart‐abstracted, aggressively ascertained (and sometimes clinically silent) hospital‐associated VTE in the large randomized controlled trials. That is, 0.25% is not an achievement, but rather the average, expected value.
Almost 25% of the observed HA‐VTE/SVTs coded were UE‐DVT, with roughly 75% of these being likely related to central venous catheterization (including those peripherally inserted). An additional 1/5 were upper‐extremity SVT of the antecubital, cephalic, and basilic veins, with the majority of these (60%) also listed as catheter‐related. Such thrombosis is best prevented by decreased use of central catheters or perhaps by using smaller‐caliber catheters.[22] It is unclear if VTEP can prevent such clots, though in cancer patients at least one recent trial seems promising.[23]
We found that patients with a coded HA‐PE/LE‐DVT were remarkably different from those not developing HA‐VTE/SVT. Patients with HA‐PE or HA‐LE‐DVT were older, sicker, more likely to have cancer, significantly more likely to spend time in the ICU, and much more likely to die in the hospital; risk factors for HA‐VTE overlap significantly with risk factors for death in the hospital. A small majority (55%) of patients in the HA‐PE/LE‐DVT group had actually received VTEP on at least day 1 or 2 of hospitalization. It may be the case that the dose of VTEP was insufficient to suppress clot formation in these patients, or that HA‐PE/LE‐DVT in patients with this degree of comorbidity is difficult to prevent.
There are a number of limitations to our study. We analyzed administrative codes, which underestimate hospital‐associated VTE/SVT events as noted above. This was a descriptive study, cross‐sectional across each hospitalization, and we were unable to draw any causal inference for differences in HA‐VTE/SVT incidence that might exist between subpopulations. We estimated VTEP from medication usage in just the first 2 days of hospitalization; we could not assess mechanical prophylaxis in this dataset; and we did not have any VTEP data for the first 2 days of hospitalization on the patients who did not develop a HA‐VTE/SVT, which made it impossible to compare the 2 populations on this measure. For those who did not receive VTEP, we were unable to obtain data regarding possible contraindications to VTEP, such as ongoing gastrointestinal or intracerebral hemorrhage. Additionally, our data are based on academic hospitals only and may not generalize to nonacademic settings. Extrapolating from HA‐VTE/SVT to hospital‐associated VTE/SVT may not be possible due to heterogeneity of clotting events and perhaps variability in whether patients would return to the hospital for all of them (eg, superficial or UE VTE may not result in readmission). Finally, it is unclear whether a switch to ICD Tenth Revision (ICD‐10) codes will impact our measured baseline in the coming year. The strengths of our analysis included stratification by type of HA‐VTE/SVT and our ability to assess the incidence of HA‐VTE/SVT in a large national population, and the provision of a baseline for VTE incidenceeasily usable by any individual hospital, network, or researcher with access to administrative datagoing forward.
In conclusion, among patients hospitalized in academic medical centers, HA‐VTE/SVT was coded in approximately 0.51% of patients with a medical illness staying >2 days, with approximately half of the events due to HA‐PE/LE‐DVT. Patients who developed HA‐PE/LE‐DVT were more acutely ill than those who did not, and VTE developed despite 55% of these patients receiving VTEP on day 1 or 2. Hospitals can reasonably treat the 0.25% figure as the baseline around which to assess their own performance in preventing HA‐PE/LE‐DVT, and can measure their own performance using administrative data. Further research is needed to determine how best to achieve further reductions in HA‐VTE/SVT through risk stratification and/or through other interventions.
Disclosures
Nothing to report.
Pulmonary embolism (PE) and deep venous thrombosis (DVT), historically referred to together as venous thromboembolism (VTE), are common, treatable, sometimes fatal, and potentially preventable medical problems.[1] Such thromboses can both precipitate a hospitalization as well as complicate it (either during or soon after discharge). Preventing such thrombosis as a complication of medical care has become a national imperative. Landmark studies such as Prophylaxis in Medical Patients With Enoxaparin (MEDENOX)[2] and Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT)[3] demonstrated both a high incidence of thrombosis in a hospitalized high‐risk medical population (15% and 5% in the 2 trials' placebo arms, respectively) as well as significant relative risk reduction through venous thromboembolism pharmacoprophylaxis (VTEP)63% and 45%, respectively. The Joint Commission,[4] the Society of Hospital Medicine,[5] and the American College of Chest Physicians[6, 7] have thus all strived to ensure the appropriate provision of VTEP in order to reduce the morbidity and mortality associated with thrombosis in hospitalized patients, including those on medical services.
Ideally, the global success of these efforts would be assessed by measuring the rate of hospital‐associated VTE (potentially including superficial venous thrombosis [SVT], which, like upper‐extremity deep venous thrombosis [UE‐DVT], is commonly a central venous catheter [CVC]‐associated, or peripherally inserted central catheter [PICC]‐associated, complication)thrombosis acquired and diagnosed during either the index hospitalization (hospital‐acquired, or HA‐VTE/SVT) or up to 30 days postdischarge. Unfortunately, postdischarge VTE/SVT is difficult to measure because patients developing it may not present to the original hospital, or at all (eg, if they do not seek care, are treated as outpatients, or, in the most extreme case, die at home). In this context, despite being far less comprehensive, HA‐VTE/SVT is a useful subset of hospital‐associated VTE/SVT, for several reasons. First, the Centers for Medicare & Medicaid Services (CMS) have mandated hospitals to qualify all medical diagnoses as present‐on‐admission (POA = Y) or not (POA = N) since 2008, such that all medical diagnoses coded POA = N can be considered hospital acquired.[8] Second, refinements made to the International Classification of Diseases, 9th Revision (ICD‐9) codes now allow differentiation of UE‐DVT and SVT from lower‐extremity (LE) DVT/PE, whereas the former were sometimes obscured by nonspecific coding.[9] Third, recent studies have shown that medical diagnoses administratively coded as HA‐VTE/SVT correlated well with HA‐VTE/SVT ascertained through chart review.[9, 10] Finally, previous work has estimated that approximately half of all hospital‐associated VTE are HA‐VTE and the other half are postdischarge VTE.[11] Thus, HA‐VTE, though comprising only approximately half of all hospital‐associated VTE, is often used as a surrogate for measuring the success of ongoing VTE prevention programs.[12]
Our study aimed to assess the incidence of HA‐VTE plus HA‐SVT in the era of mandatory POA coding and newer ICD‐9 codes for VTE.
METHODS
Setting and Cases
We conducted a retrospective analysis of discharges from the 83 academic medical centers belonging to the UHC (formerly, the University HealthSystem Consortium,
Patients in our analysis were age 18 years and discharged with a medical medical severity diagnostic‐related group (MS‐DRG) code, hospitalized for 48 hours, and did not have a surgical or obstetric MS‐DRG code (except when assigned a surgical MS‐DRG code solely due to insertion of an inferior vena cava filter, with no other major procedures performed). Cases excluded discharges with a principal diagnosis of acute VTE/SVT (defined here as including PE, LE‐DVT, UE‐DVT, SVT, chronic VTE, and thrombosis not otherwise specified), as coding guidelines prohibit assigning a HA‐VTE as the principal diagnosis for the index hospitalization.[14]
Hospital‐Acquired Venous Thromboembolism or Superficial Venous Thrombosis
Cases were classified as having a HA‐VTE/SVT if there was 1 VTE/SVT coded in a secondary diagnosis position (other diagnosis) with a corresponding POA indicator equal to either N (not POA) or U (documentation insufficient to clarify whether VTE was POA or not). This usage corresponds to CMS guidelines and reimbursement policies for hospital‐acquired conditions.[15] Among cases with 1 HA‐VTE (or SVT), we assigned 1 HA‐VTE diagnosis using a hierarchy based on the highest level of clinical importance: first, PE; then LE‐DVT; then UE‐DVT; then SVT; then chronic VTE; then, finally, unspecified VTE. We subsequently excluded cases with primarily chronic VTE from our analysis because these were likely miscodes (ie, it is unclear how a chronic VTE could not be POA) and there were only 30 such cases. Cases with HA‐PE or HA‐LE DVT were analyzed separately as an important subset of HA‐VTE (plus SVT), because HA‐PE/LE‐DVT is both life‐threatening and theoretically preventable with VTEP.
Severity of Illness and Other Measures of Comorbidity
For each case we used proprietary software (3M Health Information Systems, Murray UT) to classify severity of illness (SOI). The SOI scale, based on physiologic derangement and organ system loss of function,[16] has 4 levels: minor, major, severe, and extreme. Defined within specific disease groups (All Patient Refined DRGs), it is often compared across diseases as well.[17] We also assessed whether patients had a cancer diagnosis, spent time in the intensive care unit (ICU), and died in the hospital.
Central Venous Catheter Use in Patients With Upper‐Extremity Deep Venous Thrombosis or Superficial Venous Thrombosis
Because UE‐DVT and SVT are frequently associated with a CVC or PICC, we assessed central venous catheterization among patients with an UE‐DVT or SVT of the cephalic, basilic, or antecubital veins using diagnosis codes for complications related to dialysis devices, implants, and grafts.
Pharmacologic Thromboprophylaxis
Pharmacy records of the subset of HA‐VTE/SVT cases with PE or LE‐DVT were analyzed to determine if VTEP was administered on hospital day 1 or 2, as per Joint Commission performance requirements.[4] Medications that met criteria as VTEP included unfractionated heparin, 5000 IU, given 2 or 3 a day; enoxaparin, 40 mg, given daily; dalteparin, 2500 or 5000 IU, given daily; fondaparinux, 2.5 mg, given daily; and warfarin. We could not reliably determine if VTEP was used throughout the entire hospitalization, or whether mechanical prophylaxis was used at all.
Statistical Analysis
This was a descriptive analysis to determine the incidence of HA‐VTE/SVT and describe the demographic and clinical characteristics of this population. We calculated means and standard deviations (SD) for continuous variables and proportions for binary variables (including HA‐VTE/SVT incidence). All comparisons between populations were performed as either 2‐tailed t tests or 2 analyses. All analysis was conducted using SAS software, version 9.2 (SAS Institute, Inc., Cary, NC).
RESULTS
For the 18‐month period between October 1, 2009, and March 31, 2011, across 83 UHC hospitals, there were 2,525,068 cases. Among these, 12,847 (0.51%) had 1 HA‐VTE/SVT coded. As per the clinical importance hierarchy described above, 2449 (19.1%) cases had at least a PE coded; 3848 (30%) had at least a LE‐DVT (but not a PE) coded; 2893 (22.5%) had at least an UE‐DVT coded; 3248 (25.3%) had at least an SVT coded; 30 had at least a chronic VTE coded; and 379 had at least a VTE coded with no specified location. Of those with SVT, 192 (5.8%) were LE‐SVT codes, whereas the rest were SVT/thrombophlebitis of the upper extremities or not otherwise specified. There were 11,882 (92.5%) hospitalizations with a single HA‐VTE/SVT code and an additional 965 (7.5%) with multiple codes, for a total of 13,749 HA‐VTE/SVT events (see Supporting Information, Table S1, in the online version of this article for more specific data for the individual ICD‐9 codes used to specify HA‐VTE events).
Compared with those who did not develop any HA‐VTE/SVT, patients with HA‐PE/LE‐DVT were more likely to be Caucasian (65% vs 58%, P < 0.001) and were older (age 62 vs 48 years, P < 0.001) and sicker (79.9% vs 44.9% with a severe or extreme SOI, P < 0.001). They also were more likely to have cancer, have longer lengths of stay, be more likely to stay in the ICU, and die in the hospital (P < 0.001 for all comparisons; Table 1).
| Characteristic | No HA‐VTE, n = 2,512,221 | HA‐PE/LE DVT, n = 6,297a | P Valueb |
|---|---|---|---|
| |||
| Proportion of hospitalizations, % | 99.49 | 0.25 | |
| Age, y | 48.2 27.1 | 62.5 20.0 | <0.001 |
| Female sex | 1,347,219 (53.6) | 3,104 (49.3) | <0.001 |
| Race | <0.001 | ||
| Caucasian | 1,455,215 (57.9) | 3,963 (64.7) | |
| Black | 600,991 (23.9) | 1,425 (23.3) | |
| Hispanic | 206,553 (8.2) | 263 (4.3) | |
| API | 59,560 (2.4) | 88 (1.4) | |
| Other | 189,902 (7.6) | 389 (6.4) | |
| Admission SOI | <0.001 | ||
| Minor | 461,411 (18.4) | 181 (2.9) | |
| Major | 922,734 (36.7) | 1,081 (17.2) | |
| Severe | 880,542 (35.1) | 2,975 (47.2) | |
| Extreme | 247,244 (9.8) | 2,060 (32.7) | |
| Unknown | 290 (0.01) | 0 (0.0) | |
| Had an active diagnosis of cancer | 331,705 (13.2) | 2,162 (34.3) | <0.001 |
| Length of stay, d | 7.31 9.31 | 18.7 19.5 | <0.001 |
| Spent time in the ICU | 441,412 (17.6) | 3,011 (47.8) | <0.001 |
| Died in hospital | 57,954 (2.3) | 1,036 (16.5) | <0.001 |
| Received prophylaxisc | c | 3,454 (54.9) | c |
Among cases with a code for UE‐DVT (22.5% of all patients with HA‐VTE), 74% were noted to also have a code for a CVC, as did 60% of cases with a HA‐SVT of the antecubital, basilic, or cephalic veins (71% of SVT events; see Supporting Information, Table S1, in the online version of this article).
Of those with HA‐PE/LE‐DVT, 54.9% received pharmacologic prophylaxis on hospital day 1 or 2 (mostly with low‐molecular‐weight heparin or unfractionated heparin).
DISCUSSION
In this study of medical patients admitted to academic medical centers throughout the United States, we found that HA‐VTE/SVT was coded in approximately 0.51% of discharges, and the incidence of HA‐PE/LE‐DVT was 0.25%. Patients with a HA‐PE/LE‐DVT code were, in general, older and sicker than those who did not develop VTE. We further found that close to half of all HA‐VTE/SVT occurred in the upper extremity, with the majority of these occurring in patients who had CVCs. Finally, the majority of patients diagnosed with HA‐PE/LE‐DVT were started on VTEP on the first or second hospital day.
The overall incidence of HA‐VTE/SVT we discovered corresponds well to other studies, even those with disparate populations. A single‐institution study found a HA‐VTE/SVT incidence of approximately 0.6% among hospitalized patients on medical and nonmedical services.[12] The study by Barba found a rate of 0.93%,[18] whereas the study by Lederle found a rate of approximately 1%.[19] Spyropolous found an HA‐VTE incidence of 0.55%.[11] Rothberg found a lower rate of 0.25% in his risk‐stratification study, though in the pre‐POA and preupdated code era.[20] Our findings extend and provide context for, in a much larger population, the results of these prior studies, and represent the first national examination of HA‐VTE/SVT in the setting of numerous quality‐improvement and other efforts to reduce hospital‐associated VTE.
The incidence of HA‐VTE/SVT codes we observed likely underestimates the incidence of hospital‐associated VTE/SVT by a factor of approximately 4, for 2 reasons. First, although VTE/SVT codes with a POA flag set to No are truly hospital‐acquired events on chart review approximately 75% of the time, and thus overestimate HA‐VTE/SVT, 25% of POA = Yes codes are actually HA‐VTE/SVT events on chart review, and therefore lead to underestimation of HA‐VTE/SVT.[9] Because VTE/SVT codes with a POA flag set to Yes outnumber those flagged No by 3 or 4 to 1, events mis‐flagged Yes contribute a much greater number of undercounted HA‐VTE/SVT, elevating the actual HA‐VTE/SVT event rate by a factor of approximately 2. Second, HA‐VTE events do not include hospital‐associated VTE events that are diagnosed after the index hospitalization. In the Spyropolous study, 45% of hospital‐associated VTE events occurred after discharge, so translating HA‐VTE/SVT events to hospital‐associated VTE/SVT events would again involve multiplying by a factor of 2.[11] Thus, the overall incidence of hospital‐associated VTE/SVT events in our sample may have been approximately 2% (0.51% 4), and the overall incidence of hospital‐associated PE or LE‐DVT events may have been approximately 1%, though there may be significant variation around these estimates given that individual institutions were themselves quite variable in their POA flag accuracy in our study.[9] There is additionally the possibility that hospitals may have deliberately left some VTE/SVT uncoded, but in the absence of financial incentives to do so for anything other than postsurgical VTE, and in the presence of penalties from CMS for undercoding, we believe this to be unlikely, at least at present.
Despite these upward extrapolations, the estimated incidence of hospital‐associated VTE/SVT in our study may seem low compared with that reported in the MEDENOX[2] and PREVENT studies.[3] Much of this discrepancy vanishes on closer examination. In the large randomized trials, patients were uniformly and routinely assessed for LE‐DVT using vascular ultrasound; in contrast, in our population of hospitalizations patients may have only had diagnostic studies done for signs or symptoms. Clinically apparent hospital‐associated VTE is less common than all hospital‐associated VTE, as it was even in PREVENT,[3] and increased surveillance may even be partially driving increased hospital‐associated VTE/SVT at some hospitals.[21] Our findings suggest that success or failure in preventing administratively coded, clinically apparent HA LE‐VTE/PE should be judged, broadly, against numbers in the range established in our study (eg, 0.25%), not the 5% or 15% of chart‐abstracted, aggressively ascertained (and sometimes clinically silent) hospital‐associated VTE in the large randomized controlled trials. That is, 0.25% is not an achievement, but rather the average, expected value.
Almost 25% of the observed HA‐VTE/SVTs coded were UE‐DVT, with roughly 75% of these being likely related to central venous catheterization (including those peripherally inserted). An additional 1/5 were upper‐extremity SVT of the antecubital, cephalic, and basilic veins, with the majority of these (60%) also listed as catheter‐related. Such thrombosis is best prevented by decreased use of central catheters or perhaps by using smaller‐caliber catheters.[22] It is unclear if VTEP can prevent such clots, though in cancer patients at least one recent trial seems promising.[23]
We found that patients with a coded HA‐PE/LE‐DVT were remarkably different from those not developing HA‐VTE/SVT. Patients with HA‐PE or HA‐LE‐DVT were older, sicker, more likely to have cancer, significantly more likely to spend time in the ICU, and much more likely to die in the hospital; risk factors for HA‐VTE overlap significantly with risk factors for death in the hospital. A small majority (55%) of patients in the HA‐PE/LE‐DVT group had actually received VTEP on at least day 1 or 2 of hospitalization. It may be the case that the dose of VTEP was insufficient to suppress clot formation in these patients, or that HA‐PE/LE‐DVT in patients with this degree of comorbidity is difficult to prevent.
There are a number of limitations to our study. We analyzed administrative codes, which underestimate hospital‐associated VTE/SVT events as noted above. This was a descriptive study, cross‐sectional across each hospitalization, and we were unable to draw any causal inference for differences in HA‐VTE/SVT incidence that might exist between subpopulations. We estimated VTEP from medication usage in just the first 2 days of hospitalization; we could not assess mechanical prophylaxis in this dataset; and we did not have any VTEP data for the first 2 days of hospitalization on the patients who did not develop a HA‐VTE/SVT, which made it impossible to compare the 2 populations on this measure. For those who did not receive VTEP, we were unable to obtain data regarding possible contraindications to VTEP, such as ongoing gastrointestinal or intracerebral hemorrhage. Additionally, our data are based on academic hospitals only and may not generalize to nonacademic settings. Extrapolating from HA‐VTE/SVT to hospital‐associated VTE/SVT may not be possible due to heterogeneity of clotting events and perhaps variability in whether patients would return to the hospital for all of them (eg, superficial or UE VTE may not result in readmission). Finally, it is unclear whether a switch to ICD Tenth Revision (ICD‐10) codes will impact our measured baseline in the coming year. The strengths of our analysis included stratification by type of HA‐VTE/SVT and our ability to assess the incidence of HA‐VTE/SVT in a large national population, and the provision of a baseline for VTE incidenceeasily usable by any individual hospital, network, or researcher with access to administrative datagoing forward.
In conclusion, among patients hospitalized in academic medical centers, HA‐VTE/SVT was coded in approximately 0.51% of patients with a medical illness staying >2 days, with approximately half of the events due to HA‐PE/LE‐DVT. Patients who developed HA‐PE/LE‐DVT were more acutely ill than those who did not, and VTE developed despite 55% of these patients receiving VTEP on day 1 or 2. Hospitals can reasonably treat the 0.25% figure as the baseline around which to assess their own performance in preventing HA‐PE/LE‐DVT, and can measure their own performance using administrative data. Further research is needed to determine how best to achieve further reductions in HA‐VTE/SVT through risk stratification and/or through other interventions.
Disclosures
Nothing to report.
- , , , , ;American College of Chest Physicians AntithromboticTherapy and Prevention of Thrombosis Panel. Executive summary: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines [published corrections appear in Chest. 2012;141(4):1129 and 2012;142(6):1698]. Chest. 2012;141(2 suppl):7S–47S.
- , , , et al;Prophylaxis in Medical Patients with Enoxaparin Study Group. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. N Engl J Med. 1999;341(11):793–800.
- , , , , , . Randomized, placebo‐controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110(7):874–879.
- The Joint Commission.Specifications Manual for National Hospital Inpatient Quality Measures. Available at: http://www.jointcommission.org/specifications_manual_for_national_hospital_inpatient_quality_measures.aspx. Accessed July 18, 2012.
- , . Preventing Hospital‐Acquired Venous Thromboembolism: A Guide for Effective Quality Improvement. Prepared by the Society of Hospital Medicine. Rockville, MD: Agency for Healthcare Research and Quality; AHRQ Publication No. 08‐0075.
- , , , et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 suppl):338S–400S.
- , , , et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e195S–e226S.
- Centers for Medicare 46(6 part 1):1946–1962.
- , , , et al. Predictive and associative models to identify hospitalized medical patients at risk for VTE. Chest. 2011;140(3):706–714.
- , , , . Unintended consequences of a standard admission order set on venous thromboembolism prophylaxis and patient outcomes. J Gen Intern Med. 2012;27(3):318–324.
- United Health Consortium Website. Available at: https://www.uhc.edu. Accessed March 8, 2012.
- ICD‐9‐CM Official Guidelines for Coding and Reporting. Available at: http://www.cdc.gov/nchs/data/icd9/icdguide10.pdf. Published 2010. Accessed June 4, 2013.
- Centers for Medicare 27(5):587–612.
- Overview of Disease Severity Measures Disseminated with the Nationwide Inpatient Sample (NIS) and Kids' Inpatient Database (KID). Available at: http://www.hcup‐us.ahrq.gov/db/nation/nis/OverviewofSeveritySystems.pdf. Published December 9, 2005. Accessed June 4, 2013.
- , , , et al. Venous thromboembolism in acutely ill hospitalized medical patients. Thromb Res. 2010;126(4):276–279.
- , , , . Venous thromboembolism prophylaxis in hospitalized medical patients and those with stroke: a background review for an American College of Physicians Clinical Practice Guideline. Ann Intern Med. 2011;155(9):602–615.
- , , , , . Risk factor model to predict venous thromboembolism in hospitalized medical patients. J Hosp Med. 2011;6(4):202–209.
- , , , et al. Evaluation of surveillance bias and the validity of the venous thromboembolism quality measure. JAMA. 2013;310(14):1482–1489.
- , , , et al. Reduction of peripherally inserted central catheter‐associated DVT. Chest. 2013;143(3):627–633.
- , , , et al. Prophylaxis of catheter‐related deep vein thrombosis in cancer patients with low‐dose warfarin, low molecular weight heparin, or control: a randomized, controlled, phase III study. Cancer Chemother Pharmacol. 2013;72(1):65–73.
- , , , , ;American College of Chest Physicians AntithromboticTherapy and Prevention of Thrombosis Panel. Executive summary: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines [published corrections appear in Chest. 2012;141(4):1129 and 2012;142(6):1698]. Chest. 2012;141(2 suppl):7S–47S.
- , , , et al;Prophylaxis in Medical Patients with Enoxaparin Study Group. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. N Engl J Med. 1999;341(11):793–800.
- , , , , , . Randomized, placebo‐controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110(7):874–879.
- The Joint Commission.Specifications Manual for National Hospital Inpatient Quality Measures. Available at: http://www.jointcommission.org/specifications_manual_for_national_hospital_inpatient_quality_measures.aspx. Accessed July 18, 2012.
- , . Preventing Hospital‐Acquired Venous Thromboembolism: A Guide for Effective Quality Improvement. Prepared by the Society of Hospital Medicine. Rockville, MD: Agency for Healthcare Research and Quality; AHRQ Publication No. 08‐0075.
- , , , et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 suppl):338S–400S.
- , , , et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e195S–e226S.
- Centers for Medicare 46(6 part 1):1946–1962.
- , , , et al. Predictive and associative models to identify hospitalized medical patients at risk for VTE. Chest. 2011;140(3):706–714.
- , , , . Unintended consequences of a standard admission order set on venous thromboembolism prophylaxis and patient outcomes. J Gen Intern Med. 2012;27(3):318–324.
- United Health Consortium Website. Available at: https://www.uhc.edu. Accessed March 8, 2012.
- ICD‐9‐CM Official Guidelines for Coding and Reporting. Available at: http://www.cdc.gov/nchs/data/icd9/icdguide10.pdf. Published 2010. Accessed June 4, 2013.
- Centers for Medicare 27(5):587–612.
- Overview of Disease Severity Measures Disseminated with the Nationwide Inpatient Sample (NIS) and Kids' Inpatient Database (KID). Available at: http://www.hcup‐us.ahrq.gov/db/nation/nis/OverviewofSeveritySystems.pdf. Published December 9, 2005. Accessed June 4, 2013.
- , , , et al. Venous thromboembolism in acutely ill hospitalized medical patients. Thromb Res. 2010;126(4):276–279.
- , , , . Venous thromboembolism prophylaxis in hospitalized medical patients and those with stroke: a background review for an American College of Physicians Clinical Practice Guideline. Ann Intern Med. 2011;155(9):602–615.
- , , , , . Risk factor model to predict venous thromboembolism in hospitalized medical patients. J Hosp Med. 2011;6(4):202–209.
- , , , et al. Evaluation of surveillance bias and the validity of the venous thromboembolism quality measure. JAMA. 2013;310(14):1482–1489.
- , , , et al. Reduction of peripherally inserted central catheter‐associated DVT. Chest. 2013;143(3):627–633.
- , , , et al. Prophylaxis of catheter‐related deep vein thrombosis in cancer patients with low‐dose warfarin, low molecular weight heparin, or control: a randomized, controlled, phase III study. Cancer Chemother Pharmacol. 2013;72(1):65–73.
© 2014 Society of Hospital Medicine