User login
Opioid Use Disorder Numbers Among Pregnant Women Are on the Rise
The number of women with opioid use disorder (OUD) at labor and delivery more than quadrupled between 1999 and 2014, according to a first-ever multistate analysis of trends by the CDC.
Researchers found that the national prevalence rate of OUD rose from 1.5 per 1,000 delivery hospitalizations in 1999 to 6.5 in 2014. On average, the national prevalence rate grew by 0.39 cases per 1,000 each year.
The increases were significant and seen in all of the 28 states with at least 3 years of data available for analysis. The average increases were lowest in California and Hawaii and highest in Maine, New Mexico, Vermont, and West Virginia.
Opioid use disorder during pregnancy has been associated with a range of negative health outcomes, including maternal death, preterm birth, stillbirth, and neonatal abstinence syndrome (NAS).
The CDC’s recommended strategies include:
- Implementing universal substance use screening at the first prenatal visit;
- Ensuring pregnant women with OUD have access to medication-assisted therapy and related addiction services; and
- Ensuring that mothers with OUD receive adequate patient-centered postpartum care, including mental health and substance use treatment, relapse-prevention programs, and family planning services
The CDC also is supporting state-based perinatal quality cooperatives, networks of teams working to better identify women with OUD during pregnancy and to standardize care for mothers and NAS-affected infants.
The number of women with opioid use disorder (OUD) at labor and delivery more than quadrupled between 1999 and 2014, according to a first-ever multistate analysis of trends by the CDC.
Researchers found that the national prevalence rate of OUD rose from 1.5 per 1,000 delivery hospitalizations in 1999 to 6.5 in 2014. On average, the national prevalence rate grew by 0.39 cases per 1,000 each year.
The increases were significant and seen in all of the 28 states with at least 3 years of data available for analysis. The average increases were lowest in California and Hawaii and highest in Maine, New Mexico, Vermont, and West Virginia.
Opioid use disorder during pregnancy has been associated with a range of negative health outcomes, including maternal death, preterm birth, stillbirth, and neonatal abstinence syndrome (NAS).
The CDC’s recommended strategies include:
- Implementing universal substance use screening at the first prenatal visit;
- Ensuring pregnant women with OUD have access to medication-assisted therapy and related addiction services; and
- Ensuring that mothers with OUD receive adequate patient-centered postpartum care, including mental health and substance use treatment, relapse-prevention programs, and family planning services
The CDC also is supporting state-based perinatal quality cooperatives, networks of teams working to better identify women with OUD during pregnancy and to standardize care for mothers and NAS-affected infants.
The number of women with opioid use disorder (OUD) at labor and delivery more than quadrupled between 1999 and 2014, according to a first-ever multistate analysis of trends by the CDC.
Researchers found that the national prevalence rate of OUD rose from 1.5 per 1,000 delivery hospitalizations in 1999 to 6.5 in 2014. On average, the national prevalence rate grew by 0.39 cases per 1,000 each year.
The increases were significant and seen in all of the 28 states with at least 3 years of data available for analysis. The average increases were lowest in California and Hawaii and highest in Maine, New Mexico, Vermont, and West Virginia.
Opioid use disorder during pregnancy has been associated with a range of negative health outcomes, including maternal death, preterm birth, stillbirth, and neonatal abstinence syndrome (NAS).
The CDC’s recommended strategies include:
- Implementing universal substance use screening at the first prenatal visit;
- Ensuring pregnant women with OUD have access to medication-assisted therapy and related addiction services; and
- Ensuring that mothers with OUD receive adequate patient-centered postpartum care, including mental health and substance use treatment, relapse-prevention programs, and family planning services
The CDC also is supporting state-based perinatal quality cooperatives, networks of teams working to better identify women with OUD during pregnancy and to standardize care for mothers and NAS-affected infants.
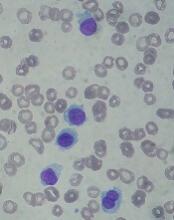
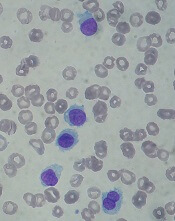
Kids with BCP-ALL exhibit immunological disparities at birth
Patients who develop B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in childhood may have dysregulated immune function at birth, according to a study published in Cancer Research.
Investigators evaluated neonatal concentrations of inflammatory markers and found significant differences between children who were later diagnosed with BCP-ALL and leukemia-free control subjects.
“Our findings suggest that children who develop ALL are immunologically disparate already at birth,” said study author Signe Holst Søegaard, a PhD student at Statens Serum Institut in Copenhagen, Denmark.
“This may link to other observations suggesting that children who develop ALL respond differently to infections in early childhood, potentially promoting subsequent genetic events required for transformation to ALL, or speculations that they are unable to eliminate preleukemic cells.”
“Importantly, our study does not inform about the nature of the associations observed—i.e., whether they are causal or consequential. Accordingly, further studies are needed both to confirm the findings and to identify the underlying mechanisms.”
For this study, Søegaard and her colleagues measured concentrations of 10 inflammatory markers on neonatal dried blood spots from 178 patients with BCP-ALL and 178 matched controls. The patients were diagnosed with BCP-ALL at ages 1 to 9.
The inflammatory markers assessed were interleukin (IL)-6, its soluble receptor sIL-6Rα, IL-8, IL-10, IL-12, IL-17, IL-18, transforming growth factor (TGF)-β1, monocyte chemotactic protein (MCP)-1, and C-reactive protein (CRP).
Results
Compared to controls, children who later developed BCP-ALL had significantly different neonatal concentrations of eight inflammatory markers.
Concentrations of sIL-6Rα, IL-8, TGF-β1, MCP-1, and CRP were significantly lower among the BCP-ALL patients. The adjusted odds ratios (adjusted for birth weight and maternal age) of BCP-ALL were 0.82 for sIL-6Rα, 0.84 for IL-8, 0.83 for TGF-β1, 0.68 for MCP-1, and 0.83 for CRP.
On the other hand, concentrations of IL-6, IL-17, and IL-18 were significantly higher among BCP-ALL patients than controls. The adjusted odds ratios were 1.19 for IL-6, 1.12 for IL-17, and 1.08 for IL-18.
The investigators noted that IL-10 concentrations were too low for accurate measurement in all patients and controls. Additionally, a “large proportion” of patients and controls (31% to 61%) had IL-6 and IL-17 concentrations that were below the limit of detection.
“We also demonstrated that several previously shown ALL risk factors—namely, birth order, gestational age, and sex—were associated with the neonatal concentrations of inflammatory markers,” Søegaard said. “These findings raise the interesting possibility that the effects of some known ALL risk factors partly act through prenatal programming of immune function.”
The investigators found that increasing birth order was associated with significantly higher IL-18 and lower CRP concentrations.
Increasing gestational age was associated with significantly lower sIL-6Rα and TGF-β1 concentrations and higher CRP concentrations. And males had significantly lower sIL-6Rα and IL-8 concentrations and higher CRP concentrations than females.
However, none of the following factors were significantly associated with concentrations of inflammatory biomarkers: maternal age at delivery, maternal hospital contact due to infection during pregnancy, maternal prescription for antimicrobials during pregnancy, birth weight, and mode of delivery.
“Our findings underline the role the child’s baseline immune characteristics may play in the development of ALL,” Søegaard said. “However, we cannot yet use our research results to predict who will develop childhood ALL. In future studies, we will further characterize the relation between immune constitution at birth and risk of childhood ALL with the ultimate goal of developing preventive strategies targeting predisposed children.”
Søegaard noted that this study had its limitations, including the small number of inflammatory markers studied. In addition, the limited sample size made it impossible to detect potential differences between BCP-ALL subtypes.
The study was sponsored by the Dagmar Marshall Foundation, the A.P. Møller Foundation, the Danish Childhood Cancer Foundation, the Arvid Nilsson Foundation, and the Danish Cancer Research Foundation. There were no conflicts of interest disclosed.
Patients who develop B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in childhood may have dysregulated immune function at birth, according to a study published in Cancer Research.
Investigators evaluated neonatal concentrations of inflammatory markers and found significant differences between children who were later diagnosed with BCP-ALL and leukemia-free control subjects.
“Our findings suggest that children who develop ALL are immunologically disparate already at birth,” said study author Signe Holst Søegaard, a PhD student at Statens Serum Institut in Copenhagen, Denmark.
“This may link to other observations suggesting that children who develop ALL respond differently to infections in early childhood, potentially promoting subsequent genetic events required for transformation to ALL, or speculations that they are unable to eliminate preleukemic cells.”
“Importantly, our study does not inform about the nature of the associations observed—i.e., whether they are causal or consequential. Accordingly, further studies are needed both to confirm the findings and to identify the underlying mechanisms.”
For this study, Søegaard and her colleagues measured concentrations of 10 inflammatory markers on neonatal dried blood spots from 178 patients with BCP-ALL and 178 matched controls. The patients were diagnosed with BCP-ALL at ages 1 to 9.
The inflammatory markers assessed were interleukin (IL)-6, its soluble receptor sIL-6Rα, IL-8, IL-10, IL-12, IL-17, IL-18, transforming growth factor (TGF)-β1, monocyte chemotactic protein (MCP)-1, and C-reactive protein (CRP).
Results
Compared to controls, children who later developed BCP-ALL had significantly different neonatal concentrations of eight inflammatory markers.
Concentrations of sIL-6Rα, IL-8, TGF-β1, MCP-1, and CRP were significantly lower among the BCP-ALL patients. The adjusted odds ratios (adjusted for birth weight and maternal age) of BCP-ALL were 0.82 for sIL-6Rα, 0.84 for IL-8, 0.83 for TGF-β1, 0.68 for MCP-1, and 0.83 for CRP.
On the other hand, concentrations of IL-6, IL-17, and IL-18 were significantly higher among BCP-ALL patients than controls. The adjusted odds ratios were 1.19 for IL-6, 1.12 for IL-17, and 1.08 for IL-18.
The investigators noted that IL-10 concentrations were too low for accurate measurement in all patients and controls. Additionally, a “large proportion” of patients and controls (31% to 61%) had IL-6 and IL-17 concentrations that were below the limit of detection.
“We also demonstrated that several previously shown ALL risk factors—namely, birth order, gestational age, and sex—were associated with the neonatal concentrations of inflammatory markers,” Søegaard said. “These findings raise the interesting possibility that the effects of some known ALL risk factors partly act through prenatal programming of immune function.”
The investigators found that increasing birth order was associated with significantly higher IL-18 and lower CRP concentrations.
Increasing gestational age was associated with significantly lower sIL-6Rα and TGF-β1 concentrations and higher CRP concentrations. And males had significantly lower sIL-6Rα and IL-8 concentrations and higher CRP concentrations than females.
However, none of the following factors were significantly associated with concentrations of inflammatory biomarkers: maternal age at delivery, maternal hospital contact due to infection during pregnancy, maternal prescription for antimicrobials during pregnancy, birth weight, and mode of delivery.
“Our findings underline the role the child’s baseline immune characteristics may play in the development of ALL,” Søegaard said. “However, we cannot yet use our research results to predict who will develop childhood ALL. In future studies, we will further characterize the relation between immune constitution at birth and risk of childhood ALL with the ultimate goal of developing preventive strategies targeting predisposed children.”
Søegaard noted that this study had its limitations, including the small number of inflammatory markers studied. In addition, the limited sample size made it impossible to detect potential differences between BCP-ALL subtypes.
The study was sponsored by the Dagmar Marshall Foundation, the A.P. Møller Foundation, the Danish Childhood Cancer Foundation, the Arvid Nilsson Foundation, and the Danish Cancer Research Foundation. There were no conflicts of interest disclosed.
Patients who develop B-cell precursor acute lymphoblastic leukemia (BCP-ALL) in childhood may have dysregulated immune function at birth, according to a study published in Cancer Research.
Investigators evaluated neonatal concentrations of inflammatory markers and found significant differences between children who were later diagnosed with BCP-ALL and leukemia-free control subjects.
“Our findings suggest that children who develop ALL are immunologically disparate already at birth,” said study author Signe Holst Søegaard, a PhD student at Statens Serum Institut in Copenhagen, Denmark.
“This may link to other observations suggesting that children who develop ALL respond differently to infections in early childhood, potentially promoting subsequent genetic events required for transformation to ALL, or speculations that they are unable to eliminate preleukemic cells.”
“Importantly, our study does not inform about the nature of the associations observed—i.e., whether they are causal or consequential. Accordingly, further studies are needed both to confirm the findings and to identify the underlying mechanisms.”
For this study, Søegaard and her colleagues measured concentrations of 10 inflammatory markers on neonatal dried blood spots from 178 patients with BCP-ALL and 178 matched controls. The patients were diagnosed with BCP-ALL at ages 1 to 9.
The inflammatory markers assessed were interleukin (IL)-6, its soluble receptor sIL-6Rα, IL-8, IL-10, IL-12, IL-17, IL-18, transforming growth factor (TGF)-β1, monocyte chemotactic protein (MCP)-1, and C-reactive protein (CRP).
Results
Compared to controls, children who later developed BCP-ALL had significantly different neonatal concentrations of eight inflammatory markers.
Concentrations of sIL-6Rα, IL-8, TGF-β1, MCP-1, and CRP were significantly lower among the BCP-ALL patients. The adjusted odds ratios (adjusted for birth weight and maternal age) of BCP-ALL were 0.82 for sIL-6Rα, 0.84 for IL-8, 0.83 for TGF-β1, 0.68 for MCP-1, and 0.83 for CRP.
On the other hand, concentrations of IL-6, IL-17, and IL-18 were significantly higher among BCP-ALL patients than controls. The adjusted odds ratios were 1.19 for IL-6, 1.12 for IL-17, and 1.08 for IL-18.
The investigators noted that IL-10 concentrations were too low for accurate measurement in all patients and controls. Additionally, a “large proportion” of patients and controls (31% to 61%) had IL-6 and IL-17 concentrations that were below the limit of detection.
“We also demonstrated that several previously shown ALL risk factors—namely, birth order, gestational age, and sex—were associated with the neonatal concentrations of inflammatory markers,” Søegaard said. “These findings raise the interesting possibility that the effects of some known ALL risk factors partly act through prenatal programming of immune function.”
The investigators found that increasing birth order was associated with significantly higher IL-18 and lower CRP concentrations.
Increasing gestational age was associated with significantly lower sIL-6Rα and TGF-β1 concentrations and higher CRP concentrations. And males had significantly lower sIL-6Rα and IL-8 concentrations and higher CRP concentrations than females.
However, none of the following factors were significantly associated with concentrations of inflammatory biomarkers: maternal age at delivery, maternal hospital contact due to infection during pregnancy, maternal prescription for antimicrobials during pregnancy, birth weight, and mode of delivery.
“Our findings underline the role the child’s baseline immune characteristics may play in the development of ALL,” Søegaard said. “However, we cannot yet use our research results to predict who will develop childhood ALL. In future studies, we will further characterize the relation between immune constitution at birth and risk of childhood ALL with the ultimate goal of developing preventive strategies targeting predisposed children.”
Søegaard noted that this study had its limitations, including the small number of inflammatory markers studied. In addition, the limited sample size made it impossible to detect potential differences between BCP-ALL subtypes.
The study was sponsored by the Dagmar Marshall Foundation, the A.P. Møller Foundation, the Danish Childhood Cancer Foundation, the Arvid Nilsson Foundation, and the Danish Cancer Research Foundation. There were no conflicts of interest disclosed.
FDA approves drug for hairy cell leukemia
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
EC approves product for von Willebrand disease
The European Commission (EC) has granted marketing authorization for vonicog alfa (Veyvondi), a recombinant von Willebrand factor (rVWF) product.
The EC approved vonicog alfa for the treatment of bleeding events and treatment/prevention of surgical bleeding in adults (age 18 and older) with von Willebrand disease (VWD) when desmopressin treatment alone is ineffective or not indicated.
The approval means Shire is authorized to market vonicog alfa in the European Union as well as in Iceland, Lichtenstein, and Norway.
The EC’s approval of vonicog alfa was based on outcomes from three clinical trials. This includes a phase 1 study and a pair of phase 3 trials—one in a surgical setting and one in a non-surgical setting.
Data from these studies are available in the Summary of Product Characteristics for vonicog alfa.
Phase 1 trial
This trial (NCT00816660, 070701) enrolled patients with type 3 or severe type 1 VWD.
The goal was to assess the safety and pharmacokinetics of vonicog alfa (rVWF) combined at a fixed ratio with recombinant factor VIII (rFVIII)—referred to as “rVWF-rFVIII.” The researchers compared rVWF-rFVIII to plasma-derived (pd) VWF combined with pdFVIII (pdVWF-pdFVIII).
The safety analysis included 32 patients who received rVWF-rFVIII. There were no thrombotic events, serious adverse events, or new cases of inhibitors to VWF or FVIII in these patients.
The pharmacokinetic analysis included 19 patients. The researchers said the pharmacokinetics of rVWF ristocetin cofactor activity, VWF antigen, and collagen-binding activity were similar with rVWF-rFVIII and pdVWF-pdFVIII.
FVIII levels were higher after infusion with rVWF-rFVIII than with pdVWF-pdFVIII, even after 72 hours. The researchers said this suggests that rVWF alone “could maintain sufficient FVIII activity to treat a bleeding episode once the initial FVIII level has reached a therapeutic threshold.”
These results were published in Blood in 2013.
Phase 3: Non-surgical
This study (NCT01410227, 071001) included 49 patients with VWD who received vonicog alfa with or without rFVIII.
All participants had successful treatment of bleeding episodes. Most (96.9%) treated bleeds (192 bleeds in 22 patients) were given an “excellent” efficacy rating (as good as or better than expected).
Most bleeds (81.8%) were resolved with a single infusion of vonicog alfa, and the treatment had a mean half-life of 21.9 hours.
There were 8 adverse events considered related to vonicog alfa, and 2 were serious. One patient experienced 2 simultaneous serious events—chest discomfort and increased heart rate—but these were resolved.
There were no thrombotic events in this trial, no treatment-related binding or neutralizing antibodies against VWF, and no neutralizing antibodies against FVIII.
These results were published in Blood in 2015.
Phase 3: Surgical setting
This trial (NCT02283268, 071101) enrolled 15 adults with severe VWD who were undergoing elective surgical procedures (10 of them major procedures).
Patients received vonicog alfa at 40 to 60 IU per kg of body weight 12 to 24 hours before surgery. Within 3 hours of surgery, each patient’s FVIII level (FVIII:C) was assessed, with a target of 30 IU/dL for minor surgeries and 60 IU/dL for major surgeries.
Within an hour of surgery, patients received a dose of vonicog alfa, with or without rFVIII, depending on the target FVIII:C levels at the 3-hour assessment.
Ten patients received rVWF alone, 12 did not receive any preoperative FVIII, and 2 did not receive rVWF postoperatively.
Vonicog alfa demonstrated overall hemostatic efficacy, as assessed 24 hours after the last perioperative infusion or the completion of the study visit, whichever occurred earlier.
Intra- and post-operative hemostasis was rated as “excellent” (as good as or better than expected) in 60% of patients and “good” (probably as good as expected) in 40% of patients.
One patient developed deep vein thrombosis 3 days after undergoing hip replacement surgery.
One patient tested positive for binding antibodies to VWF. None of the patients developed binding antibodies against potential impurities such as rFurin, CHO-protein, or mouse IgG.
These results were presented at the WFH 2018 World Congress.
The European Commission (EC) has granted marketing authorization for vonicog alfa (Veyvondi), a recombinant von Willebrand factor (rVWF) product.
The EC approved vonicog alfa for the treatment of bleeding events and treatment/prevention of surgical bleeding in adults (age 18 and older) with von Willebrand disease (VWD) when desmopressin treatment alone is ineffective or not indicated.
The approval means Shire is authorized to market vonicog alfa in the European Union as well as in Iceland, Lichtenstein, and Norway.
The EC’s approval of vonicog alfa was based on outcomes from three clinical trials. This includes a phase 1 study and a pair of phase 3 trials—one in a surgical setting and one in a non-surgical setting.
Data from these studies are available in the Summary of Product Characteristics for vonicog alfa.
Phase 1 trial
This trial (NCT00816660, 070701) enrolled patients with type 3 or severe type 1 VWD.
The goal was to assess the safety and pharmacokinetics of vonicog alfa (rVWF) combined at a fixed ratio with recombinant factor VIII (rFVIII)—referred to as “rVWF-rFVIII.” The researchers compared rVWF-rFVIII to plasma-derived (pd) VWF combined with pdFVIII (pdVWF-pdFVIII).
The safety analysis included 32 patients who received rVWF-rFVIII. There were no thrombotic events, serious adverse events, or new cases of inhibitors to VWF or FVIII in these patients.
The pharmacokinetic analysis included 19 patients. The researchers said the pharmacokinetics of rVWF ristocetin cofactor activity, VWF antigen, and collagen-binding activity were similar with rVWF-rFVIII and pdVWF-pdFVIII.
FVIII levels were higher after infusion with rVWF-rFVIII than with pdVWF-pdFVIII, even after 72 hours. The researchers said this suggests that rVWF alone “could maintain sufficient FVIII activity to treat a bleeding episode once the initial FVIII level has reached a therapeutic threshold.”
These results were published in Blood in 2013.
Phase 3: Non-surgical
This study (NCT01410227, 071001) included 49 patients with VWD who received vonicog alfa with or without rFVIII.
All participants had successful treatment of bleeding episodes. Most (96.9%) treated bleeds (192 bleeds in 22 patients) were given an “excellent” efficacy rating (as good as or better than expected).
Most bleeds (81.8%) were resolved with a single infusion of vonicog alfa, and the treatment had a mean half-life of 21.9 hours.
There were 8 adverse events considered related to vonicog alfa, and 2 were serious. One patient experienced 2 simultaneous serious events—chest discomfort and increased heart rate—but these were resolved.
There were no thrombotic events in this trial, no treatment-related binding or neutralizing antibodies against VWF, and no neutralizing antibodies against FVIII.
These results were published in Blood in 2015.
Phase 3: Surgical setting
This trial (NCT02283268, 071101) enrolled 15 adults with severe VWD who were undergoing elective surgical procedures (10 of them major procedures).
Patients received vonicog alfa at 40 to 60 IU per kg of body weight 12 to 24 hours before surgery. Within 3 hours of surgery, each patient’s FVIII level (FVIII:C) was assessed, with a target of 30 IU/dL for minor surgeries and 60 IU/dL for major surgeries.
Within an hour of surgery, patients received a dose of vonicog alfa, with or without rFVIII, depending on the target FVIII:C levels at the 3-hour assessment.
Ten patients received rVWF alone, 12 did not receive any preoperative FVIII, and 2 did not receive rVWF postoperatively.
Vonicog alfa demonstrated overall hemostatic efficacy, as assessed 24 hours after the last perioperative infusion or the completion of the study visit, whichever occurred earlier.
Intra- and post-operative hemostasis was rated as “excellent” (as good as or better than expected) in 60% of patients and “good” (probably as good as expected) in 40% of patients.
One patient developed deep vein thrombosis 3 days after undergoing hip replacement surgery.
One patient tested positive for binding antibodies to VWF. None of the patients developed binding antibodies against potential impurities such as rFurin, CHO-protein, or mouse IgG.
These results were presented at the WFH 2018 World Congress.
The European Commission (EC) has granted marketing authorization for vonicog alfa (Veyvondi), a recombinant von Willebrand factor (rVWF) product.
The EC approved vonicog alfa for the treatment of bleeding events and treatment/prevention of surgical bleeding in adults (age 18 and older) with von Willebrand disease (VWD) when desmopressin treatment alone is ineffective or not indicated.
The approval means Shire is authorized to market vonicog alfa in the European Union as well as in Iceland, Lichtenstein, and Norway.
The EC’s approval of vonicog alfa was based on outcomes from three clinical trials. This includes a phase 1 study and a pair of phase 3 trials—one in a surgical setting and one in a non-surgical setting.
Data from these studies are available in the Summary of Product Characteristics for vonicog alfa.
Phase 1 trial
This trial (NCT00816660, 070701) enrolled patients with type 3 or severe type 1 VWD.
The goal was to assess the safety and pharmacokinetics of vonicog alfa (rVWF) combined at a fixed ratio with recombinant factor VIII (rFVIII)—referred to as “rVWF-rFVIII.” The researchers compared rVWF-rFVIII to plasma-derived (pd) VWF combined with pdFVIII (pdVWF-pdFVIII).
The safety analysis included 32 patients who received rVWF-rFVIII. There were no thrombotic events, serious adverse events, or new cases of inhibitors to VWF or FVIII in these patients.
The pharmacokinetic analysis included 19 patients. The researchers said the pharmacokinetics of rVWF ristocetin cofactor activity, VWF antigen, and collagen-binding activity were similar with rVWF-rFVIII and pdVWF-pdFVIII.
FVIII levels were higher after infusion with rVWF-rFVIII than with pdVWF-pdFVIII, even after 72 hours. The researchers said this suggests that rVWF alone “could maintain sufficient FVIII activity to treat a bleeding episode once the initial FVIII level has reached a therapeutic threshold.”
These results were published in Blood in 2013.
Phase 3: Non-surgical
This study (NCT01410227, 071001) included 49 patients with VWD who received vonicog alfa with or without rFVIII.
All participants had successful treatment of bleeding episodes. Most (96.9%) treated bleeds (192 bleeds in 22 patients) were given an “excellent” efficacy rating (as good as or better than expected).
Most bleeds (81.8%) were resolved with a single infusion of vonicog alfa, and the treatment had a mean half-life of 21.9 hours.
There were 8 adverse events considered related to vonicog alfa, and 2 were serious. One patient experienced 2 simultaneous serious events—chest discomfort and increased heart rate—but these were resolved.
There were no thrombotic events in this trial, no treatment-related binding or neutralizing antibodies against VWF, and no neutralizing antibodies against FVIII.
These results were published in Blood in 2015.
Phase 3: Surgical setting
This trial (NCT02283268, 071101) enrolled 15 adults with severe VWD who were undergoing elective surgical procedures (10 of them major procedures).
Patients received vonicog alfa at 40 to 60 IU per kg of body weight 12 to 24 hours before surgery. Within 3 hours of surgery, each patient’s FVIII level (FVIII:C) was assessed, with a target of 30 IU/dL for minor surgeries and 60 IU/dL for major surgeries.
Within an hour of surgery, patients received a dose of vonicog alfa, with or without rFVIII, depending on the target FVIII:C levels at the 3-hour assessment.
Ten patients received rVWF alone, 12 did not receive any preoperative FVIII, and 2 did not receive rVWF postoperatively.
Vonicog alfa demonstrated overall hemostatic efficacy, as assessed 24 hours after the last perioperative infusion or the completion of the study visit, whichever occurred earlier.
Intra- and post-operative hemostasis was rated as “excellent” (as good as or better than expected) in 60% of patients and “good” (probably as good as expected) in 40% of patients.
One patient developed deep vein thrombosis 3 days after undergoing hip replacement surgery.
One patient tested positive for binding antibodies to VWF. None of the patients developed binding antibodies against potential impurities such as rFurin, CHO-protein, or mouse IgG.
These results were presented at the WFH 2018 World Congress.
The benzodiazepine dilemma
As clinicians, we are faced with a conflict when deciding whether or not to prescribe a benzodiazepine. If we prescribe one of these agents, we might be putting our patients at risk for dependence and abuse. However, if we do not prescribe them, we risk providing inadequate treatment, especially for patients with panic disorder.
Benzodiazepine dependence and abuse can take many forms. Dependence can be psychological as well as physiologic. While many patients will adhere to their prescribing regimen, some may sell their benzodiazepines, falsely claim that they have “panic attacks,” or take a fatal overdose of an opioid and benzodiazepine combination.
Here I discuss the pros and cons of restricting benzodiazepines use to low doses and/or combination therapy with antidepressants.
_
Weighing the benefits of restricted prescribing
Some double-blind studies referenced in the American Psychiatric Association (APA) 2010 Practice Guideline for the Treatment of Patients with Panic Disorder1 suggest that benzodiazepine duration of treatment and dosages should be severely restricted. These studies found that:
- Although the combination of a selective serotonin reuptake inhibitor (SSRI) and a benzodiazepine initially decreased the number of panic attacks more quickly than SSRI monotherapy, the 2 treatments are equally effective after 4 or 5 weeks.2,3
- For the treatment of panic disorder, a low dosage of a benzodiazepine (clonazepam 1 mg/d or alprazolam 2 mg/d) was as effective as a higher dosage (clonazepam 2 mg/d or alprazolam 6 mg/d).4,5
However, these studies could be misleading. They all excluded patients with a comorbid condition, such as bipolar disorder or depression, that was more severe than their panic disorder. Severe comorbidity is associated with more severe panic symptoms,6,7 which might require an SSRI/benzodiazepine combination or a higher benzodiazepine dosage.
The APA Practice Guideline suggests the following possible options:
- benzodiazepine augmentation if there is a partial response to an SSRI
- substitution with a different SSRI or a serotonin-norepinephrine reuptake inhibitor (SNRI) if there is no response to an SSRI
- benzodiazepine augmentation or substitution if there is still no therapeutic response.
Continue to: The APA Practice Guideline also states...
The APA Practice Guideline also states that although the highest “usual therapeutic dose” for panic disorder is clonazepam 2 mg/d or alprazolam 4 mg/d, “higher doses are sometimes used for patients who do not respond to the usual therapeutic dose.”1
Presumably, an SSRI/benzodiazepine combination should be considered if an SSRI alleviates major depressive disorder but does not alleviate a comorbid panic disorder. However, the APA Practice Guideline does not include studies that investigated this clinical scenario.
Monitor carefully for dependency/abuse
Restricting benzodiazepine use to low doses over a short period of time may decrease the risk of dependence and abuse. However, this practice may also limit or prevent effective treatment for adherent patients with panic disorder who do not adequately respond to SSRI or SNRI monotherapy.
Therefore, clinicians need to carefully differentiate between patients who are adherent to their prescribed dosages and those who may be at risk for benzodiazepine dependence and abuse. Consider using prescription drug monitoring programs and drug screens to help detect patients who “doctor shop” for benzodiazepines, or who could be abusing opioids, alcohol, marijuana, or other substances while taking a benzodiazepine.
1. American Psychiatric Association. Practice guideline for the treatment of patients with panic disorder, 2nd edition. Washington DC: American Psychiatric Association. 2010. https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/panicdisorder.pdf. Accessed March 7, 2018.
2. Goddard AW, Brouette T, Almai A, et al. Early coadministration of clonazepam with sertraline for panic disorder. Arch Gen Psychiatry. 2001;58(7):681-686.
3. Pollack MH, Simon NM, Worthington JJ, et al. Combined paroxetine and clonazepam treatment strategies compared to paroxetine monotherapy for panic disorder. J Psychopharmacol. 2003;17(3):276-282.
4. Lydiard RB, Lesser IM, Ballenger JC, et al. A fixed-dose study of alprazolam 2 mg, alprazolam 6 mg, and placebo in panic disorder. J Clin Psychopharmacol. 1992;12(2):966-103.
5. Rosenbaum JF, Moroz G, Bowden CL. Clonazepam in the treatment of panic disorder with or without agoraphobia: a dose-response study of efficacy, safety, and discontinuance. Clonazepam Panic Disorder Dose-Response Study Group. J Clin Psychopharmacol. 1997;17(5):390-400.
6. Goodwin RD, Hoven CW. Bipolar-panic comorbidity in the general population: prevalence and associated morbidity. J Affect Disord. 2002;70(1):27-33.
7. Roy-Byrne PP, Stang P, Wittchen HU, et al. Lifetime panic-depression comorbidity in the National Comorbidity Survey. Association with symptoms, impairment, course and help-seeking. Br J Psychiatry. 2000;176:229-235.
As clinicians, we are faced with a conflict when deciding whether or not to prescribe a benzodiazepine. If we prescribe one of these agents, we might be putting our patients at risk for dependence and abuse. However, if we do not prescribe them, we risk providing inadequate treatment, especially for patients with panic disorder.
Benzodiazepine dependence and abuse can take many forms. Dependence can be psychological as well as physiologic. While many patients will adhere to their prescribing regimen, some may sell their benzodiazepines, falsely claim that they have “panic attacks,” or take a fatal overdose of an opioid and benzodiazepine combination.
Here I discuss the pros and cons of restricting benzodiazepines use to low doses and/or combination therapy with antidepressants.
_
Weighing the benefits of restricted prescribing
Some double-blind studies referenced in the American Psychiatric Association (APA) 2010 Practice Guideline for the Treatment of Patients with Panic Disorder1 suggest that benzodiazepine duration of treatment and dosages should be severely restricted. These studies found that:
- Although the combination of a selective serotonin reuptake inhibitor (SSRI) and a benzodiazepine initially decreased the number of panic attacks more quickly than SSRI monotherapy, the 2 treatments are equally effective after 4 or 5 weeks.2,3
- For the treatment of panic disorder, a low dosage of a benzodiazepine (clonazepam 1 mg/d or alprazolam 2 mg/d) was as effective as a higher dosage (clonazepam 2 mg/d or alprazolam 6 mg/d).4,5
However, these studies could be misleading. They all excluded patients with a comorbid condition, such as bipolar disorder or depression, that was more severe than their panic disorder. Severe comorbidity is associated with more severe panic symptoms,6,7 which might require an SSRI/benzodiazepine combination or a higher benzodiazepine dosage.
The APA Practice Guideline suggests the following possible options:
- benzodiazepine augmentation if there is a partial response to an SSRI
- substitution with a different SSRI or a serotonin-norepinephrine reuptake inhibitor (SNRI) if there is no response to an SSRI
- benzodiazepine augmentation or substitution if there is still no therapeutic response.
Continue to: The APA Practice Guideline also states...
The APA Practice Guideline also states that although the highest “usual therapeutic dose” for panic disorder is clonazepam 2 mg/d or alprazolam 4 mg/d, “higher doses are sometimes used for patients who do not respond to the usual therapeutic dose.”1
Presumably, an SSRI/benzodiazepine combination should be considered if an SSRI alleviates major depressive disorder but does not alleviate a comorbid panic disorder. However, the APA Practice Guideline does not include studies that investigated this clinical scenario.
Monitor carefully for dependency/abuse
Restricting benzodiazepine use to low doses over a short period of time may decrease the risk of dependence and abuse. However, this practice may also limit or prevent effective treatment for adherent patients with panic disorder who do not adequately respond to SSRI or SNRI monotherapy.
Therefore, clinicians need to carefully differentiate between patients who are adherent to their prescribed dosages and those who may be at risk for benzodiazepine dependence and abuse. Consider using prescription drug monitoring programs and drug screens to help detect patients who “doctor shop” for benzodiazepines, or who could be abusing opioids, alcohol, marijuana, or other substances while taking a benzodiazepine.
As clinicians, we are faced with a conflict when deciding whether or not to prescribe a benzodiazepine. If we prescribe one of these agents, we might be putting our patients at risk for dependence and abuse. However, if we do not prescribe them, we risk providing inadequate treatment, especially for patients with panic disorder.
Benzodiazepine dependence and abuse can take many forms. Dependence can be psychological as well as physiologic. While many patients will adhere to their prescribing regimen, some may sell their benzodiazepines, falsely claim that they have “panic attacks,” or take a fatal overdose of an opioid and benzodiazepine combination.
Here I discuss the pros and cons of restricting benzodiazepines use to low doses and/or combination therapy with antidepressants.
_
Weighing the benefits of restricted prescribing
Some double-blind studies referenced in the American Psychiatric Association (APA) 2010 Practice Guideline for the Treatment of Patients with Panic Disorder1 suggest that benzodiazepine duration of treatment and dosages should be severely restricted. These studies found that:
- Although the combination of a selective serotonin reuptake inhibitor (SSRI) and a benzodiazepine initially decreased the number of panic attacks more quickly than SSRI monotherapy, the 2 treatments are equally effective after 4 or 5 weeks.2,3
- For the treatment of panic disorder, a low dosage of a benzodiazepine (clonazepam 1 mg/d or alprazolam 2 mg/d) was as effective as a higher dosage (clonazepam 2 mg/d or alprazolam 6 mg/d).4,5
However, these studies could be misleading. They all excluded patients with a comorbid condition, such as bipolar disorder or depression, that was more severe than their panic disorder. Severe comorbidity is associated with more severe panic symptoms,6,7 which might require an SSRI/benzodiazepine combination or a higher benzodiazepine dosage.
The APA Practice Guideline suggests the following possible options:
- benzodiazepine augmentation if there is a partial response to an SSRI
- substitution with a different SSRI or a serotonin-norepinephrine reuptake inhibitor (SNRI) if there is no response to an SSRI
- benzodiazepine augmentation or substitution if there is still no therapeutic response.
Continue to: The APA Practice Guideline also states...
The APA Practice Guideline also states that although the highest “usual therapeutic dose” for panic disorder is clonazepam 2 mg/d or alprazolam 4 mg/d, “higher doses are sometimes used for patients who do not respond to the usual therapeutic dose.”1
Presumably, an SSRI/benzodiazepine combination should be considered if an SSRI alleviates major depressive disorder but does not alleviate a comorbid panic disorder. However, the APA Practice Guideline does not include studies that investigated this clinical scenario.
Monitor carefully for dependency/abuse
Restricting benzodiazepine use to low doses over a short period of time may decrease the risk of dependence and abuse. However, this practice may also limit or prevent effective treatment for adherent patients with panic disorder who do not adequately respond to SSRI or SNRI monotherapy.
Therefore, clinicians need to carefully differentiate between patients who are adherent to their prescribed dosages and those who may be at risk for benzodiazepine dependence and abuse. Consider using prescription drug monitoring programs and drug screens to help detect patients who “doctor shop” for benzodiazepines, or who could be abusing opioids, alcohol, marijuana, or other substances while taking a benzodiazepine.
1. American Psychiatric Association. Practice guideline for the treatment of patients with panic disorder, 2nd edition. Washington DC: American Psychiatric Association. 2010. https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/panicdisorder.pdf. Accessed March 7, 2018.
2. Goddard AW, Brouette T, Almai A, et al. Early coadministration of clonazepam with sertraline for panic disorder. Arch Gen Psychiatry. 2001;58(7):681-686.
3. Pollack MH, Simon NM, Worthington JJ, et al. Combined paroxetine and clonazepam treatment strategies compared to paroxetine monotherapy for panic disorder. J Psychopharmacol. 2003;17(3):276-282.
4. Lydiard RB, Lesser IM, Ballenger JC, et al. A fixed-dose study of alprazolam 2 mg, alprazolam 6 mg, and placebo in panic disorder. J Clin Psychopharmacol. 1992;12(2):966-103.
5. Rosenbaum JF, Moroz G, Bowden CL. Clonazepam in the treatment of panic disorder with or without agoraphobia: a dose-response study of efficacy, safety, and discontinuance. Clonazepam Panic Disorder Dose-Response Study Group. J Clin Psychopharmacol. 1997;17(5):390-400.
6. Goodwin RD, Hoven CW. Bipolar-panic comorbidity in the general population: prevalence and associated morbidity. J Affect Disord. 2002;70(1):27-33.
7. Roy-Byrne PP, Stang P, Wittchen HU, et al. Lifetime panic-depression comorbidity in the National Comorbidity Survey. Association with symptoms, impairment, course and help-seeking. Br J Psychiatry. 2000;176:229-235.
1. American Psychiatric Association. Practice guideline for the treatment of patients with panic disorder, 2nd edition. Washington DC: American Psychiatric Association. 2010. https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/panicdisorder.pdf. Accessed March 7, 2018.
2. Goddard AW, Brouette T, Almai A, et al. Early coadministration of clonazepam with sertraline for panic disorder. Arch Gen Psychiatry. 2001;58(7):681-686.
3. Pollack MH, Simon NM, Worthington JJ, et al. Combined paroxetine and clonazepam treatment strategies compared to paroxetine monotherapy for panic disorder. J Psychopharmacol. 2003;17(3):276-282.
4. Lydiard RB, Lesser IM, Ballenger JC, et al. A fixed-dose study of alprazolam 2 mg, alprazolam 6 mg, and placebo in panic disorder. J Clin Psychopharmacol. 1992;12(2):966-103.
5. Rosenbaum JF, Moroz G, Bowden CL. Clonazepam in the treatment of panic disorder with or without agoraphobia: a dose-response study of efficacy, safety, and discontinuance. Clonazepam Panic Disorder Dose-Response Study Group. J Clin Psychopharmacol. 1997;17(5):390-400.
6. Goodwin RD, Hoven CW. Bipolar-panic comorbidity in the general population: prevalence and associated morbidity. J Affect Disord. 2002;70(1):27-33.
7. Roy-Byrne PP, Stang P, Wittchen HU, et al. Lifetime panic-depression comorbidity in the National Comorbidity Survey. Association with symptoms, impairment, course and help-seeking. Br J Psychiatry. 2000;176:229-235.
From the Editors: Surgical M&M – a lost art?
Surgeons have a time-honored institution of reflection, learning, and responsibility in the Morbidity & Mortality conference. The M&M is unique in the medical profession and we should look critically at efforts by hospitals and bureaucracies to change its character and its purpose.
A story in this issue (“Little overlap between surgical M&M and AHRQ” on adverse events, p. xx) covers a published study (Anderson J et al. J Am Coll Surg. 2018 Jul 5. doi: 10.1016/j.jamcollsurg.2018.06.008) of the ways in which surgeon-reported M&M cases do (but mostly, do not) overlap with those captured in the ubiquitous Patient Safety Indicators (PSIs).
I am reminded once again of the long road we have traveled from the M&M conference of my student and resident days in the 1970s to today’s version of this event.
At our institution, the M&M has evolved over the years to serve not only an educational venue from which all in attendance learn from the misadventures of others and (hopefully) avoid similar errors in the future, but also as a component of the institution’s overall patient safety and quality improvement program. The authors point out the inherent difference between the clinician-generated M&M cases, which may possess selection bias in the cases they identify as complications, and the strictly defined PSIs that are captured from an administrative database and often include cases overlooked by the clinicians as not relevant to the surgical M&M process.
Both kinds of data are valuable, but for surgeons, the M&M is the one venue where they can focus in the company of their colleagues on those instances where the best intentions go off the rails: erroneous decisions, faulty assessments, and unanticipated complications.
The surgical M&M conference traces its origins to the admirable practice of early 20th century Boston surgeon Ernest Codman, who tracked the treatment outcome of all of his patients on “End Result Cards” for at least a year with the goal of identifying errors to improve the care of his future patients. He established the first M&M conference at Harvard and was one of the founders of the American College of Surgeons and the forerunner of the Joint Commission. His idea that surgeon and hospital outcomes should be made public so that patients could make an informed choice about where and from whom to seek care was, however, vigorously resisted by Harvard and Dr. Codman lost his privileges there.
I would offer that the M&M conference and the PSIs are apples and oranges: both good, but different. Each serves a different purpose. But too much integration of the PSI into the M&M format could end up creating a formulaic adverse events conference that answers to bureaucratic needs of the hospital, but loses some of its value as a forum for learning.
It is worth reflecting on how and why the traditional M&M has such value to surgeons.
To begin with, the quality of the leadership matters. A good leader knows how to avoid blaming, shaming, or embarrassing the presenter, who likely feels bad enough about the complication without being tortured about it. The goal is that all salient factors that contributed to the complication are elicited and that everyone present comes away from the conference armed with alternative ways to prevent a repeat of the same complication.
As a resident I learned more at the Saturday morning M&M than I did at any other conference. I was there every Saturday morning almost without fail for 15 years as a medical student, resident, and faculty member. There I learned not only how to avoid errors and benefit from the accumulated wisdom of many gray hairs in the audience, but also how to present an embarrassing complication both honestly and even with some self-deprecating humor.
Chief residents such as Don Trunkey, Brent Eastman, and Theodore Schrock were gifted in being accountable for mistakes while simultaneously deflecting ire with some well-chosen props. I remember one vivid example: Ted Schrock stepping up to the podium to present a “case gone bad” while holding a garbage-can lid in front of his chest like a shield, ready to defend himself. I don’t remember the case, but the picture is still etched in my mind 45 years later.
Our chairman, Dr. J. Englebert Dunphy, was a master at zeroing in on the critical errors in decision-making or operative conduct that had led to a poor outcome. When the presenter was honest and well-meaning but lacking in sophisticated insight, Dr. Dunphy would calmly ask probing questions that guided the resident to understand why a complication had occurred and how it might have been avoided. If the complication was exceptionally egregious, or the resident was not forthright or was evasive in his “mea culpa,” Dr. Dunphy would turn to one of his staunch faculty allies in the front row and inquire, “Brodie, what do you think about that?” to which Brodie Stephens would typically reply, “Bert, I thought we were here to CURE disease, not CAUSE it!” (To add some colorful football lore to the story, Howard Brodie Stephens was the All-American end who caught a 53-yard pass from “Brick” Muller in the 1921 Rose Bowl victory of Cal Berkeley over Ohio State, the Pacific Coast Conference’s last win against a Big Ten team until 1953.)
Certain resident shortcomings were sure to raise Dr. Dunphy’s ire. These included failing to take responsibility for your mistake and attempting to blame the error on someone else or on another discipline or not adequately supervising an intern or junior resident if you were the chief resident. The latter crime was the subject of one of the most clever and resourceful chief resident M&M presentations of all time, that of past ACS President Brent Eastman as his final presentation from his vascular surgery rotation, the last of his chief year. This one took some moxie, considerable preparation, and the involvement of colleagues near and far. Brent enlisted his good friend and later distinguished cardiac surgeon Dr. Larry Cohn, then junior faculty at Harvard, to find a list of Dr. Dunphy’s complications while he was a resident in Boston in the 1930s. Although records were no longer available, Dr. Cohn mentioned the issue to Dr. Hartwell Harrison, who had been chief resident at the Peter Bent Brigham Hospital when Dr. Dunphy was a junior resident. Dr. Harrison remembered a case that Dr. Dunphy had performed in the outpatient clinic without supervision in which he encountered uncontrolled bleeding.
Armed with the perfect case to present, Brent coached Dr. Edwin (Jack) Wylie to be his “plant” in the audience. At M&M, Brent sheepishly admitted that the case he had to present was that of an unsupervised junior resident who incurred uncontrolled bleeding in the outpatient setting. On cue, Dr. Wylie asked, “Who the hell WAS that resident?” Dr. Eastman then shuffled through his papers to find the correct sheet and announced, “Dr. John E. Dunphy, Peter Bent Brigham Hospital, 1937.” The room exploded in uproarious laughter, joined heartily by Dr. Dunphy.
That was then, and this is now. I can’t envision such a spectacle ever occurring these days. The M&M conference of 2018 has become far more standardized and endowed with greater scientific rigor. Its evolution has likely made M&M more precise and valuable as an educational tool for surgeons to learn from the mistakes of others, but of course, it has lost an element of surprise and hilarity that kept all of us sleep-deprived residents awake and alert. The lessons learned from the traditional M&M lasted this surgeon’s lifetime, and we should consider preserving some of the give-and-take, admission of failure, and reflection that made the M&M so unforgettable.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery at Oregon Health & Science University, Portland. She is the coeditor of ACS Surgery News.
Surgeons have a time-honored institution of reflection, learning, and responsibility in the Morbidity & Mortality conference. The M&M is unique in the medical profession and we should look critically at efforts by hospitals and bureaucracies to change its character and its purpose.
A story in this issue (“Little overlap between surgical M&M and AHRQ” on adverse events, p. xx) covers a published study (Anderson J et al. J Am Coll Surg. 2018 Jul 5. doi: 10.1016/j.jamcollsurg.2018.06.008) of the ways in which surgeon-reported M&M cases do (but mostly, do not) overlap with those captured in the ubiquitous Patient Safety Indicators (PSIs).
I am reminded once again of the long road we have traveled from the M&M conference of my student and resident days in the 1970s to today’s version of this event.
At our institution, the M&M has evolved over the years to serve not only an educational venue from which all in attendance learn from the misadventures of others and (hopefully) avoid similar errors in the future, but also as a component of the institution’s overall patient safety and quality improvement program. The authors point out the inherent difference between the clinician-generated M&M cases, which may possess selection bias in the cases they identify as complications, and the strictly defined PSIs that are captured from an administrative database and often include cases overlooked by the clinicians as not relevant to the surgical M&M process.
Both kinds of data are valuable, but for surgeons, the M&M is the one venue where they can focus in the company of their colleagues on those instances where the best intentions go off the rails: erroneous decisions, faulty assessments, and unanticipated complications.
The surgical M&M conference traces its origins to the admirable practice of early 20th century Boston surgeon Ernest Codman, who tracked the treatment outcome of all of his patients on “End Result Cards” for at least a year with the goal of identifying errors to improve the care of his future patients. He established the first M&M conference at Harvard and was one of the founders of the American College of Surgeons and the forerunner of the Joint Commission. His idea that surgeon and hospital outcomes should be made public so that patients could make an informed choice about where and from whom to seek care was, however, vigorously resisted by Harvard and Dr. Codman lost his privileges there.
I would offer that the M&M conference and the PSIs are apples and oranges: both good, but different. Each serves a different purpose. But too much integration of the PSI into the M&M format could end up creating a formulaic adverse events conference that answers to bureaucratic needs of the hospital, but loses some of its value as a forum for learning.
It is worth reflecting on how and why the traditional M&M has such value to surgeons.
To begin with, the quality of the leadership matters. A good leader knows how to avoid blaming, shaming, or embarrassing the presenter, who likely feels bad enough about the complication without being tortured about it. The goal is that all salient factors that contributed to the complication are elicited and that everyone present comes away from the conference armed with alternative ways to prevent a repeat of the same complication.
As a resident I learned more at the Saturday morning M&M than I did at any other conference. I was there every Saturday morning almost without fail for 15 years as a medical student, resident, and faculty member. There I learned not only how to avoid errors and benefit from the accumulated wisdom of many gray hairs in the audience, but also how to present an embarrassing complication both honestly and even with some self-deprecating humor.
Chief residents such as Don Trunkey, Brent Eastman, and Theodore Schrock were gifted in being accountable for mistakes while simultaneously deflecting ire with some well-chosen props. I remember one vivid example: Ted Schrock stepping up to the podium to present a “case gone bad” while holding a garbage-can lid in front of his chest like a shield, ready to defend himself. I don’t remember the case, but the picture is still etched in my mind 45 years later.
Our chairman, Dr. J. Englebert Dunphy, was a master at zeroing in on the critical errors in decision-making or operative conduct that had led to a poor outcome. When the presenter was honest and well-meaning but lacking in sophisticated insight, Dr. Dunphy would calmly ask probing questions that guided the resident to understand why a complication had occurred and how it might have been avoided. If the complication was exceptionally egregious, or the resident was not forthright or was evasive in his “mea culpa,” Dr. Dunphy would turn to one of his staunch faculty allies in the front row and inquire, “Brodie, what do you think about that?” to which Brodie Stephens would typically reply, “Bert, I thought we were here to CURE disease, not CAUSE it!” (To add some colorful football lore to the story, Howard Brodie Stephens was the All-American end who caught a 53-yard pass from “Brick” Muller in the 1921 Rose Bowl victory of Cal Berkeley over Ohio State, the Pacific Coast Conference’s last win against a Big Ten team until 1953.)
Certain resident shortcomings were sure to raise Dr. Dunphy’s ire. These included failing to take responsibility for your mistake and attempting to blame the error on someone else or on another discipline or not adequately supervising an intern or junior resident if you were the chief resident. The latter crime was the subject of one of the most clever and resourceful chief resident M&M presentations of all time, that of past ACS President Brent Eastman as his final presentation from his vascular surgery rotation, the last of his chief year. This one took some moxie, considerable preparation, and the involvement of colleagues near and far. Brent enlisted his good friend and later distinguished cardiac surgeon Dr. Larry Cohn, then junior faculty at Harvard, to find a list of Dr. Dunphy’s complications while he was a resident in Boston in the 1930s. Although records were no longer available, Dr. Cohn mentioned the issue to Dr. Hartwell Harrison, who had been chief resident at the Peter Bent Brigham Hospital when Dr. Dunphy was a junior resident. Dr. Harrison remembered a case that Dr. Dunphy had performed in the outpatient clinic without supervision in which he encountered uncontrolled bleeding.
Armed with the perfect case to present, Brent coached Dr. Edwin (Jack) Wylie to be his “plant” in the audience. At M&M, Brent sheepishly admitted that the case he had to present was that of an unsupervised junior resident who incurred uncontrolled bleeding in the outpatient setting. On cue, Dr. Wylie asked, “Who the hell WAS that resident?” Dr. Eastman then shuffled through his papers to find the correct sheet and announced, “Dr. John E. Dunphy, Peter Bent Brigham Hospital, 1937.” The room exploded in uproarious laughter, joined heartily by Dr. Dunphy.
That was then, and this is now. I can’t envision such a spectacle ever occurring these days. The M&M conference of 2018 has become far more standardized and endowed with greater scientific rigor. Its evolution has likely made M&M more precise and valuable as an educational tool for surgeons to learn from the mistakes of others, but of course, it has lost an element of surprise and hilarity that kept all of us sleep-deprived residents awake and alert. The lessons learned from the traditional M&M lasted this surgeon’s lifetime, and we should consider preserving some of the give-and-take, admission of failure, and reflection that made the M&M so unforgettable.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery at Oregon Health & Science University, Portland. She is the coeditor of ACS Surgery News.
Surgeons have a time-honored institution of reflection, learning, and responsibility in the Morbidity & Mortality conference. The M&M is unique in the medical profession and we should look critically at efforts by hospitals and bureaucracies to change its character and its purpose.
A story in this issue (“Little overlap between surgical M&M and AHRQ” on adverse events, p. xx) covers a published study (Anderson J et al. J Am Coll Surg. 2018 Jul 5. doi: 10.1016/j.jamcollsurg.2018.06.008) of the ways in which surgeon-reported M&M cases do (but mostly, do not) overlap with those captured in the ubiquitous Patient Safety Indicators (PSIs).
I am reminded once again of the long road we have traveled from the M&M conference of my student and resident days in the 1970s to today’s version of this event.
At our institution, the M&M has evolved over the years to serve not only an educational venue from which all in attendance learn from the misadventures of others and (hopefully) avoid similar errors in the future, but also as a component of the institution’s overall patient safety and quality improvement program. The authors point out the inherent difference between the clinician-generated M&M cases, which may possess selection bias in the cases they identify as complications, and the strictly defined PSIs that are captured from an administrative database and often include cases overlooked by the clinicians as not relevant to the surgical M&M process.
Both kinds of data are valuable, but for surgeons, the M&M is the one venue where they can focus in the company of their colleagues on those instances where the best intentions go off the rails: erroneous decisions, faulty assessments, and unanticipated complications.
The surgical M&M conference traces its origins to the admirable practice of early 20th century Boston surgeon Ernest Codman, who tracked the treatment outcome of all of his patients on “End Result Cards” for at least a year with the goal of identifying errors to improve the care of his future patients. He established the first M&M conference at Harvard and was one of the founders of the American College of Surgeons and the forerunner of the Joint Commission. His idea that surgeon and hospital outcomes should be made public so that patients could make an informed choice about where and from whom to seek care was, however, vigorously resisted by Harvard and Dr. Codman lost his privileges there.
I would offer that the M&M conference and the PSIs are apples and oranges: both good, but different. Each serves a different purpose. But too much integration of the PSI into the M&M format could end up creating a formulaic adverse events conference that answers to bureaucratic needs of the hospital, but loses some of its value as a forum for learning.
It is worth reflecting on how and why the traditional M&M has such value to surgeons.
To begin with, the quality of the leadership matters. A good leader knows how to avoid blaming, shaming, or embarrassing the presenter, who likely feels bad enough about the complication without being tortured about it. The goal is that all salient factors that contributed to the complication are elicited and that everyone present comes away from the conference armed with alternative ways to prevent a repeat of the same complication.
As a resident I learned more at the Saturday morning M&M than I did at any other conference. I was there every Saturday morning almost without fail for 15 years as a medical student, resident, and faculty member. There I learned not only how to avoid errors and benefit from the accumulated wisdom of many gray hairs in the audience, but also how to present an embarrassing complication both honestly and even with some self-deprecating humor.
Chief residents such as Don Trunkey, Brent Eastman, and Theodore Schrock were gifted in being accountable for mistakes while simultaneously deflecting ire with some well-chosen props. I remember one vivid example: Ted Schrock stepping up to the podium to present a “case gone bad” while holding a garbage-can lid in front of his chest like a shield, ready to defend himself. I don’t remember the case, but the picture is still etched in my mind 45 years later.
Our chairman, Dr. J. Englebert Dunphy, was a master at zeroing in on the critical errors in decision-making or operative conduct that had led to a poor outcome. When the presenter was honest and well-meaning but lacking in sophisticated insight, Dr. Dunphy would calmly ask probing questions that guided the resident to understand why a complication had occurred and how it might have been avoided. If the complication was exceptionally egregious, or the resident was not forthright or was evasive in his “mea culpa,” Dr. Dunphy would turn to one of his staunch faculty allies in the front row and inquire, “Brodie, what do you think about that?” to which Brodie Stephens would typically reply, “Bert, I thought we were here to CURE disease, not CAUSE it!” (To add some colorful football lore to the story, Howard Brodie Stephens was the All-American end who caught a 53-yard pass from “Brick” Muller in the 1921 Rose Bowl victory of Cal Berkeley over Ohio State, the Pacific Coast Conference’s last win against a Big Ten team until 1953.)
Certain resident shortcomings were sure to raise Dr. Dunphy’s ire. These included failing to take responsibility for your mistake and attempting to blame the error on someone else or on another discipline or not adequately supervising an intern or junior resident if you were the chief resident. The latter crime was the subject of one of the most clever and resourceful chief resident M&M presentations of all time, that of past ACS President Brent Eastman as his final presentation from his vascular surgery rotation, the last of his chief year. This one took some moxie, considerable preparation, and the involvement of colleagues near and far. Brent enlisted his good friend and later distinguished cardiac surgeon Dr. Larry Cohn, then junior faculty at Harvard, to find a list of Dr. Dunphy’s complications while he was a resident in Boston in the 1930s. Although records were no longer available, Dr. Cohn mentioned the issue to Dr. Hartwell Harrison, who had been chief resident at the Peter Bent Brigham Hospital when Dr. Dunphy was a junior resident. Dr. Harrison remembered a case that Dr. Dunphy had performed in the outpatient clinic without supervision in which he encountered uncontrolled bleeding.
Armed with the perfect case to present, Brent coached Dr. Edwin (Jack) Wylie to be his “plant” in the audience. At M&M, Brent sheepishly admitted that the case he had to present was that of an unsupervised junior resident who incurred uncontrolled bleeding in the outpatient setting. On cue, Dr. Wylie asked, “Who the hell WAS that resident?” Dr. Eastman then shuffled through his papers to find the correct sheet and announced, “Dr. John E. Dunphy, Peter Bent Brigham Hospital, 1937.” The room exploded in uproarious laughter, joined heartily by Dr. Dunphy.
That was then, and this is now. I can’t envision such a spectacle ever occurring these days. The M&M conference of 2018 has become far more standardized and endowed with greater scientific rigor. Its evolution has likely made M&M more precise and valuable as an educational tool for surgeons to learn from the mistakes of others, but of course, it has lost an element of surprise and hilarity that kept all of us sleep-deprived residents awake and alert. The lessons learned from the traditional M&M lasted this surgeon’s lifetime, and we should consider preserving some of the give-and-take, admission of failure, and reflection that made the M&M so unforgettable.
Dr. Deveney is professor of surgery and vice chair of education in the department of surgery at Oregon Health & Science University, Portland. She is the coeditor of ACS Surgery News.
ACS hosts briefing on military health care
The American College of Surgeons (ACS) hosted a Capitol Hill briefing July 23 featuring a panel of experts on military surgical care—John H. Armstrong, MD, FACS; Arthur Kellerman, MD, MPH; CAPT Eric Elster, MD, FACS, USN; and COL Frederick Lough, MD, FACS, USA. The panelists focused on themes from the recent publication, Out of the Crucible: How the U.S. Military Transformed Combat Casualty Care in Iraq and Afghanistan, and the role military-civilian trauma partnerships play in maintaining trauma care readiness.
Attendees learned that the prolonged conflicts in Operation Iraqi Freedom and Operation Enduring Freedom saw an unprecedented improvement in military combat casualty care and the creation of the Joint Trauma System (JTS), allowing for the development and dissemination of best trauma practices across the Department of Defense. Preserving the JTS and establishing a fully integrated military-civilian trauma system are among the primary efforts of the Military Health System Strategic Partnership American College of Surgeons.
For more information about this briefing, contact Carrie Zlatos, ACS Senior Congressional Lobbyist, at [email protected] or 202-672-1508.
The American College of Surgeons (ACS) hosted a Capitol Hill briefing July 23 featuring a panel of experts on military surgical care—John H. Armstrong, MD, FACS; Arthur Kellerman, MD, MPH; CAPT Eric Elster, MD, FACS, USN; and COL Frederick Lough, MD, FACS, USA. The panelists focused on themes from the recent publication, Out of the Crucible: How the U.S. Military Transformed Combat Casualty Care in Iraq and Afghanistan, and the role military-civilian trauma partnerships play in maintaining trauma care readiness.
Attendees learned that the prolonged conflicts in Operation Iraqi Freedom and Operation Enduring Freedom saw an unprecedented improvement in military combat casualty care and the creation of the Joint Trauma System (JTS), allowing for the development and dissemination of best trauma practices across the Department of Defense. Preserving the JTS and establishing a fully integrated military-civilian trauma system are among the primary efforts of the Military Health System Strategic Partnership American College of Surgeons.
For more information about this briefing, contact Carrie Zlatos, ACS Senior Congressional Lobbyist, at [email protected] or 202-672-1508.
The American College of Surgeons (ACS) hosted a Capitol Hill briefing July 23 featuring a panel of experts on military surgical care—John H. Armstrong, MD, FACS; Arthur Kellerman, MD, MPH; CAPT Eric Elster, MD, FACS, USN; and COL Frederick Lough, MD, FACS, USA. The panelists focused on themes from the recent publication, Out of the Crucible: How the U.S. Military Transformed Combat Casualty Care in Iraq and Afghanistan, and the role military-civilian trauma partnerships play in maintaining trauma care readiness.
Attendees learned that the prolonged conflicts in Operation Iraqi Freedom and Operation Enduring Freedom saw an unprecedented improvement in military combat casualty care and the creation of the Joint Trauma System (JTS), allowing for the development and dissemination of best trauma practices across the Department of Defense. Preserving the JTS and establishing a fully integrated military-civilian trauma system are among the primary efforts of the Military Health System Strategic Partnership American College of Surgeons.
For more information about this briefing, contact Carrie Zlatos, ACS Senior Congressional Lobbyist, at [email protected] or 202-672-1508.
Become a member at Clinical Congress
If you are not already a member of the American College of Surgeons (ACS) or you know an interested non-member colleague who is attending the Clinical Congress 2018 in Boston, MA, visit the Become a Member booth to apply for ACS membership. The membership application fee will be waived for individuals who apply at the meeting.
The Become a Member booth will be located in the Registration Area of the Boston Convention & Exhibition Center and will be open the same hours as registration, which are as follows:
• Sunday, October 21: 7:00 am–6:00 pm
• Monday, October 22: 6:30 am–5:00 pm
• Tuesday, October 23: 7:00 am–4:00 pm
• Wednesday, October 24: 7:00 am–4:00 pm
• Thursday, October 25: 7:00–10:00 am
Plan ahead to apply at the meeting for one of the following membership categories and have access to the documents listed to support and speed up your application process:
• Fellows (U.S. and Canada): Copy of board certification document, current curriculum vitae (CV), and names of five ACS Fellows to serve as references. Watch the video about the domestic application process at www.youtube.com/watch?v=2VUqvveNYf8&.
• International Fellows: Copy of medical license, board/country certification document, current CV, and names of three ACS Fellows to serve as references. Watch the video about the international application process at www.youtube.com/watch?v=djoMUAUTA4I.
• Associate Fellows: Copy of residency completion document or an official board/country certification document.
• Residents: Letter verifying training status.
• Medical Students: Letter verifying educational status.
• Affiliate Members: The name of a Fellow as a reference.
Familiarize yourself with the ACS Member Benefits (facs.org/member-services/benefits) and make time to apply for membership while at the meeting. For additional information, contact Member Services at [email protected] or 800-621-4111. We look forward to assisting you.
If you are not already a member of the American College of Surgeons (ACS) or you know an interested non-member colleague who is attending the Clinical Congress 2018 in Boston, MA, visit the Become a Member booth to apply for ACS membership. The membership application fee will be waived for individuals who apply at the meeting.
The Become a Member booth will be located in the Registration Area of the Boston Convention & Exhibition Center and will be open the same hours as registration, which are as follows:
• Sunday, October 21: 7:00 am–6:00 pm
• Monday, October 22: 6:30 am–5:00 pm
• Tuesday, October 23: 7:00 am–4:00 pm
• Wednesday, October 24: 7:00 am–4:00 pm
• Thursday, October 25: 7:00–10:00 am
Plan ahead to apply at the meeting for one of the following membership categories and have access to the documents listed to support and speed up your application process:
• Fellows (U.S. and Canada): Copy of board certification document, current curriculum vitae (CV), and names of five ACS Fellows to serve as references. Watch the video about the domestic application process at www.youtube.com/watch?v=2VUqvveNYf8&.
• International Fellows: Copy of medical license, board/country certification document, current CV, and names of three ACS Fellows to serve as references. Watch the video about the international application process at www.youtube.com/watch?v=djoMUAUTA4I.
• Associate Fellows: Copy of residency completion document or an official board/country certification document.
• Residents: Letter verifying training status.
• Medical Students: Letter verifying educational status.
• Affiliate Members: The name of a Fellow as a reference.
Familiarize yourself with the ACS Member Benefits (facs.org/member-services/benefits) and make time to apply for membership while at the meeting. For additional information, contact Member Services at [email protected] or 800-621-4111. We look forward to assisting you.
If you are not already a member of the American College of Surgeons (ACS) or you know an interested non-member colleague who is attending the Clinical Congress 2018 in Boston, MA, visit the Become a Member booth to apply for ACS membership. The membership application fee will be waived for individuals who apply at the meeting.
The Become a Member booth will be located in the Registration Area of the Boston Convention & Exhibition Center and will be open the same hours as registration, which are as follows:
• Sunday, October 21: 7:00 am–6:00 pm
• Monday, October 22: 6:30 am–5:00 pm
• Tuesday, October 23: 7:00 am–4:00 pm
• Wednesday, October 24: 7:00 am–4:00 pm
• Thursday, October 25: 7:00–10:00 am
Plan ahead to apply at the meeting for one of the following membership categories and have access to the documents listed to support and speed up your application process:
• Fellows (U.S. and Canada): Copy of board certification document, current curriculum vitae (CV), and names of five ACS Fellows to serve as references. Watch the video about the domestic application process at www.youtube.com/watch?v=2VUqvveNYf8&.
• International Fellows: Copy of medical license, board/country certification document, current CV, and names of three ACS Fellows to serve as references. Watch the video about the international application process at www.youtube.com/watch?v=djoMUAUTA4I.
• Associate Fellows: Copy of residency completion document or an official board/country certification document.
• Residents: Letter verifying training status.
• Medical Students: Letter verifying educational status.
• Affiliate Members: The name of a Fellow as a reference.
Familiarize yourself with the ACS Member Benefits (facs.org/member-services/benefits) and make time to apply for membership while at the meeting. For additional information, contact Member Services at [email protected] or 800-621-4111. We look forward to assisting you.
AMA House of Delegates meeting: Setting policy for organized medicine
The American Medical Association (AMA) Annual House of Delegates (HOD) meeting took place June 9–13 in Chicago, IL. Approximately 600 delegates representing state medical societies, national specialty societies, and national medical societies gathered to discuss a variety of policies and issues that affect patients and physicians.
The American College of Surgeons (ACS) sent a six-member delegation to the meeting (see sidebar, page 84). In addition to serving on the College’s delegation, some of the ACS delegates serve in other capacities in the HOD, on the AMA Council on Medical Education, in the AMA Young Physicians Section, and in the AMA Resident and Fellow Section. All three of these entities convene during the broader HOD meeting, providing further opportunities to influence the adoption of health care policy.
Issue highlights
During the HOD meeting, delegates discuss and adopt policies as presented in reports and resolutions. The HOD considered more than 64 reports and 200 resolutions at the June meeting, including the following:
• Colorectal cancer screening: A joint report of the Council on Medical Service and the Council on Science and Public Health focused on policy updates related to coverage for colorectal cancer screening. As adopted, this report directs the AMA to seek to eliminate cost-sharing in all health care plans for the full range of colorectal cancer screening and all associated costs, including colonoscopies that include a diagnostic intervention (such as the removal of a polyp or biopsy of a mass), as defined by Medicare. In addition, the AMA now recognizes colon cancer as a leading cause of cancer deaths in the U.S. and encourages appropriate screening programs to detect colorectal cancer.
• Obesity treatment: The ACS cosponsored Resolution 201, Barriers to Obesity Treatment, which the HOD adopted. It directs the AMA to work with appropriate stakeholders to remove out-of-date restrictions at the state and federal level that prohibit health care providers from providing the accepted standard of care to patients affected by obesity.
• Kidney transplantation: The ACS cosponsored Resolution 219, which asked the AMA to work with professional and patient-centered organizations to advance patient- and physician-directed coordinated care for end-stage renal disease (ESRD) patients and to actively oppose the Dialysis Patient Access to Integrated-care, Empowerment, Nephrologists, and Treatment Services (PATIENTS) Demonstration Act of 2017. The College and other organizations opposed this federal legislation as introduced, partly due to infringement on the patient-physician relationship and the disruption of care by prohibiting nephrologists who are not part of preferred networks from caring for their patients receiving care in units owned by an ESRD integrated care organization participating in the model. The resolution was referred to the AMA Board of Trustees.
The HOD also amended AMA policy regarding living organ donation. The amended policy supports removing financial barriers to living organ donation, such as provisions for expenses incurred after the donation as a consequence of donation, and prohibiting the use of living donor status to limit disability and long-term care coverage. In addition, the AMA will advocate that live organ donation operations be classified as a serious health condition under the Family and Medical Leave Act.
• Firearms: After extensive debate, the HOD adopted a new policy regarding firearms. The new policy supports a ban on the sale of firearms and ammunition from licensed and unlicensed dealers to those younger than 21 years old (excluding certain categories of individuals, such as military and law enforcement personnel); opposes “concealed carry reciprocity” federal legislation; and supports a ban on the sale and ownership of all assault-type weapons, bump stocks and related devices, high-capacity magazines, and armor-piercing bullets.
• Prostate cancer screening: Resolution 226 proposed that the AMA develop model state legislation for screening asymptomatic men ages 55–69 for prostate cancer after informed discussion between patients and their physician without annual deductible or copayment. Testimony both for and against this resolution was offered, and the HOD referred the resolution so that the various facets of the issue can be studied.
• Maintenance of Certification: The AMA Council on Medical Education submitted its annual report on Maintenance of Certification (MOC), detailing the status of revisions by specialty boards to the MOC process and discussing council interactions with the boards and the American Board of Medical Specialties. The HOD adopted this report.
Two additional MOC-related resolutions were introduced. Resolution 320 asked the AMA to support young physician involvement in MOC, and Resolution 316 asked the AMA to oppose the Part 4 Improvement in Medical Practice requirement for MOC. The HOD adopted Resolution 320 and referred Resolution 316.
AMA elections
During the annual meeting of the HOD, officers and councils are elected. Patrice Harris, MD, a psychiatrist from Atlanta, GA, was elected president-elect, and Russell Kridel, MD, FACS, a facial plastic surgeon from Houston, TX, who was endorsed by the ACS, was reelected to the Board of Trustees. Following the conclusion of the HOD, Dr. Kridel also was elected to serve as Secretary of the Board. For a summary of the AMA election results, go to the AMA Wire article at wire.ama-assn.org/ama-news/patrice-harris-md-wins-office-ama-president-elect.
Surgical Caucus
Founded almost 30 years ago, the Surgical Caucus gives surgeon delegates and alternates the opportunity to meet to discuss HOD business from the surgical perspective. Part of the regular business of the caucus involves sponsoring an educational session. Titled Ready or Not, Here We Come: Transitioning to Practice in a Modern Healthcare System, this program focused on the transition from surgical residency to practice and how the profession can enhance this process, as well as the effects of current training program requirements on preparedness for independent practice.
Future meeting
The Interim Meeting of the HOD will take place November 10–13 at National Harbor, MD, and the ACS delegation will be well-prepared to represent surgical perspectives. Surgeons with thoughts about potential resolutions or questions about the HOD may contact [email protected] or visit the HOD web page at www.ama-assn.org/hod-annual-overview.
ACS Delegation at the AMA HOD
• Patricia L. Turner, MD, FACS (Delegation Chair), general surgery, Chicago, IL; Director, ACS Division of Member Services; member and immediate past-chair, AMA Council on Medical Education
• David B. Hoyt, MD, FACS, general surgery, Chicago, IL; ACS Executive Director
• Brian J. Gavitt, MD, MPH (also Young Physicians Section delegate), general surgery, Cincinnati, OH
• Jacob Moalem, MD, FACS, general surgery, Rochester, NY
• Leigh A. Neumayer, MD, FACS, general surgery, Tucson, AZ; Chair, ACS Board of Regents
• Naveen F. Sangji, MD (also Resident and Fellow Section delegate), general surgery resident, Boston, MA
Dr. Turner is Director, ACS Division of Member Services, Chicago, IL.
Mr. Sutton is Manager, State Affairs, ACS Division of Advocacy and Health Policy, Washington, DC.
The American Medical Association (AMA) Annual House of Delegates (HOD) meeting took place June 9–13 in Chicago, IL. Approximately 600 delegates representing state medical societies, national specialty societies, and national medical societies gathered to discuss a variety of policies and issues that affect patients and physicians.
The American College of Surgeons (ACS) sent a six-member delegation to the meeting (see sidebar, page 84). In addition to serving on the College’s delegation, some of the ACS delegates serve in other capacities in the HOD, on the AMA Council on Medical Education, in the AMA Young Physicians Section, and in the AMA Resident and Fellow Section. All three of these entities convene during the broader HOD meeting, providing further opportunities to influence the adoption of health care policy.
Issue highlights
During the HOD meeting, delegates discuss and adopt policies as presented in reports and resolutions. The HOD considered more than 64 reports and 200 resolutions at the June meeting, including the following:
• Colorectal cancer screening: A joint report of the Council on Medical Service and the Council on Science and Public Health focused on policy updates related to coverage for colorectal cancer screening. As adopted, this report directs the AMA to seek to eliminate cost-sharing in all health care plans for the full range of colorectal cancer screening and all associated costs, including colonoscopies that include a diagnostic intervention (such as the removal of a polyp or biopsy of a mass), as defined by Medicare. In addition, the AMA now recognizes colon cancer as a leading cause of cancer deaths in the U.S. and encourages appropriate screening programs to detect colorectal cancer.
• Obesity treatment: The ACS cosponsored Resolution 201, Barriers to Obesity Treatment, which the HOD adopted. It directs the AMA to work with appropriate stakeholders to remove out-of-date restrictions at the state and federal level that prohibit health care providers from providing the accepted standard of care to patients affected by obesity.
• Kidney transplantation: The ACS cosponsored Resolution 219, which asked the AMA to work with professional and patient-centered organizations to advance patient- and physician-directed coordinated care for end-stage renal disease (ESRD) patients and to actively oppose the Dialysis Patient Access to Integrated-care, Empowerment, Nephrologists, and Treatment Services (PATIENTS) Demonstration Act of 2017. The College and other organizations opposed this federal legislation as introduced, partly due to infringement on the patient-physician relationship and the disruption of care by prohibiting nephrologists who are not part of preferred networks from caring for their patients receiving care in units owned by an ESRD integrated care organization participating in the model. The resolution was referred to the AMA Board of Trustees.
The HOD also amended AMA policy regarding living organ donation. The amended policy supports removing financial barriers to living organ donation, such as provisions for expenses incurred after the donation as a consequence of donation, and prohibiting the use of living donor status to limit disability and long-term care coverage. In addition, the AMA will advocate that live organ donation operations be classified as a serious health condition under the Family and Medical Leave Act.
• Firearms: After extensive debate, the HOD adopted a new policy regarding firearms. The new policy supports a ban on the sale of firearms and ammunition from licensed and unlicensed dealers to those younger than 21 years old (excluding certain categories of individuals, such as military and law enforcement personnel); opposes “concealed carry reciprocity” federal legislation; and supports a ban on the sale and ownership of all assault-type weapons, bump stocks and related devices, high-capacity magazines, and armor-piercing bullets.
• Prostate cancer screening: Resolution 226 proposed that the AMA develop model state legislation for screening asymptomatic men ages 55–69 for prostate cancer after informed discussion between patients and their physician without annual deductible or copayment. Testimony both for and against this resolution was offered, and the HOD referred the resolution so that the various facets of the issue can be studied.
• Maintenance of Certification: The AMA Council on Medical Education submitted its annual report on Maintenance of Certification (MOC), detailing the status of revisions by specialty boards to the MOC process and discussing council interactions with the boards and the American Board of Medical Specialties. The HOD adopted this report.
Two additional MOC-related resolutions were introduced. Resolution 320 asked the AMA to support young physician involvement in MOC, and Resolution 316 asked the AMA to oppose the Part 4 Improvement in Medical Practice requirement for MOC. The HOD adopted Resolution 320 and referred Resolution 316.
AMA elections
During the annual meeting of the HOD, officers and councils are elected. Patrice Harris, MD, a psychiatrist from Atlanta, GA, was elected president-elect, and Russell Kridel, MD, FACS, a facial plastic surgeon from Houston, TX, who was endorsed by the ACS, was reelected to the Board of Trustees. Following the conclusion of the HOD, Dr. Kridel also was elected to serve as Secretary of the Board. For a summary of the AMA election results, go to the AMA Wire article at wire.ama-assn.org/ama-news/patrice-harris-md-wins-office-ama-president-elect.
Surgical Caucus
Founded almost 30 years ago, the Surgical Caucus gives surgeon delegates and alternates the opportunity to meet to discuss HOD business from the surgical perspective. Part of the regular business of the caucus involves sponsoring an educational session. Titled Ready or Not, Here We Come: Transitioning to Practice in a Modern Healthcare System, this program focused on the transition from surgical residency to practice and how the profession can enhance this process, as well as the effects of current training program requirements on preparedness for independent practice.
Future meeting
The Interim Meeting of the HOD will take place November 10–13 at National Harbor, MD, and the ACS delegation will be well-prepared to represent surgical perspectives. Surgeons with thoughts about potential resolutions or questions about the HOD may contact [email protected] or visit the HOD web page at www.ama-assn.org/hod-annual-overview.
ACS Delegation at the AMA HOD
• Patricia L. Turner, MD, FACS (Delegation Chair), general surgery, Chicago, IL; Director, ACS Division of Member Services; member and immediate past-chair, AMA Council on Medical Education
• David B. Hoyt, MD, FACS, general surgery, Chicago, IL; ACS Executive Director
• Brian J. Gavitt, MD, MPH (also Young Physicians Section delegate), general surgery, Cincinnati, OH
• Jacob Moalem, MD, FACS, general surgery, Rochester, NY
• Leigh A. Neumayer, MD, FACS, general surgery, Tucson, AZ; Chair, ACS Board of Regents
• Naveen F. Sangji, MD (also Resident and Fellow Section delegate), general surgery resident, Boston, MA
Dr. Turner is Director, ACS Division of Member Services, Chicago, IL.
Mr. Sutton is Manager, State Affairs, ACS Division of Advocacy and Health Policy, Washington, DC.
The American Medical Association (AMA) Annual House of Delegates (HOD) meeting took place June 9–13 in Chicago, IL. Approximately 600 delegates representing state medical societies, national specialty societies, and national medical societies gathered to discuss a variety of policies and issues that affect patients and physicians.
The American College of Surgeons (ACS) sent a six-member delegation to the meeting (see sidebar, page 84). In addition to serving on the College’s delegation, some of the ACS delegates serve in other capacities in the HOD, on the AMA Council on Medical Education, in the AMA Young Physicians Section, and in the AMA Resident and Fellow Section. All three of these entities convene during the broader HOD meeting, providing further opportunities to influence the adoption of health care policy.
Issue highlights
During the HOD meeting, delegates discuss and adopt policies as presented in reports and resolutions. The HOD considered more than 64 reports and 200 resolutions at the June meeting, including the following:
• Colorectal cancer screening: A joint report of the Council on Medical Service and the Council on Science and Public Health focused on policy updates related to coverage for colorectal cancer screening. As adopted, this report directs the AMA to seek to eliminate cost-sharing in all health care plans for the full range of colorectal cancer screening and all associated costs, including colonoscopies that include a diagnostic intervention (such as the removal of a polyp or biopsy of a mass), as defined by Medicare. In addition, the AMA now recognizes colon cancer as a leading cause of cancer deaths in the U.S. and encourages appropriate screening programs to detect colorectal cancer.
• Obesity treatment: The ACS cosponsored Resolution 201, Barriers to Obesity Treatment, which the HOD adopted. It directs the AMA to work with appropriate stakeholders to remove out-of-date restrictions at the state and federal level that prohibit health care providers from providing the accepted standard of care to patients affected by obesity.
• Kidney transplantation: The ACS cosponsored Resolution 219, which asked the AMA to work with professional and patient-centered organizations to advance patient- and physician-directed coordinated care for end-stage renal disease (ESRD) patients and to actively oppose the Dialysis Patient Access to Integrated-care, Empowerment, Nephrologists, and Treatment Services (PATIENTS) Demonstration Act of 2017. The College and other organizations opposed this federal legislation as introduced, partly due to infringement on the patient-physician relationship and the disruption of care by prohibiting nephrologists who are not part of preferred networks from caring for their patients receiving care in units owned by an ESRD integrated care organization participating in the model. The resolution was referred to the AMA Board of Trustees.
The HOD also amended AMA policy regarding living organ donation. The amended policy supports removing financial barriers to living organ donation, such as provisions for expenses incurred after the donation as a consequence of donation, and prohibiting the use of living donor status to limit disability and long-term care coverage. In addition, the AMA will advocate that live organ donation operations be classified as a serious health condition under the Family and Medical Leave Act.
• Firearms: After extensive debate, the HOD adopted a new policy regarding firearms. The new policy supports a ban on the sale of firearms and ammunition from licensed and unlicensed dealers to those younger than 21 years old (excluding certain categories of individuals, such as military and law enforcement personnel); opposes “concealed carry reciprocity” federal legislation; and supports a ban on the sale and ownership of all assault-type weapons, bump stocks and related devices, high-capacity magazines, and armor-piercing bullets.
• Prostate cancer screening: Resolution 226 proposed that the AMA develop model state legislation for screening asymptomatic men ages 55–69 for prostate cancer after informed discussion between patients and their physician without annual deductible or copayment. Testimony both for and against this resolution was offered, and the HOD referred the resolution so that the various facets of the issue can be studied.
• Maintenance of Certification: The AMA Council on Medical Education submitted its annual report on Maintenance of Certification (MOC), detailing the status of revisions by specialty boards to the MOC process and discussing council interactions with the boards and the American Board of Medical Specialties. The HOD adopted this report.
Two additional MOC-related resolutions were introduced. Resolution 320 asked the AMA to support young physician involvement in MOC, and Resolution 316 asked the AMA to oppose the Part 4 Improvement in Medical Practice requirement for MOC. The HOD adopted Resolution 320 and referred Resolution 316.
AMA elections
During the annual meeting of the HOD, officers and councils are elected. Patrice Harris, MD, a psychiatrist from Atlanta, GA, was elected president-elect, and Russell Kridel, MD, FACS, a facial plastic surgeon from Houston, TX, who was endorsed by the ACS, was reelected to the Board of Trustees. Following the conclusion of the HOD, Dr. Kridel also was elected to serve as Secretary of the Board. For a summary of the AMA election results, go to the AMA Wire article at wire.ama-assn.org/ama-news/patrice-harris-md-wins-office-ama-president-elect.
Surgical Caucus
Founded almost 30 years ago, the Surgical Caucus gives surgeon delegates and alternates the opportunity to meet to discuss HOD business from the surgical perspective. Part of the regular business of the caucus involves sponsoring an educational session. Titled Ready or Not, Here We Come: Transitioning to Practice in a Modern Healthcare System, this program focused on the transition from surgical residency to practice and how the profession can enhance this process, as well as the effects of current training program requirements on preparedness for independent practice.
Future meeting
The Interim Meeting of the HOD will take place November 10–13 at National Harbor, MD, and the ACS delegation will be well-prepared to represent surgical perspectives. Surgeons with thoughts about potential resolutions or questions about the HOD may contact [email protected] or visit the HOD web page at www.ama-assn.org/hod-annual-overview.
ACS Delegation at the AMA HOD
• Patricia L. Turner, MD, FACS (Delegation Chair), general surgery, Chicago, IL; Director, ACS Division of Member Services; member and immediate past-chair, AMA Council on Medical Education
• David B. Hoyt, MD, FACS, general surgery, Chicago, IL; ACS Executive Director
• Brian J. Gavitt, MD, MPH (also Young Physicians Section delegate), general surgery, Cincinnati, OH
• Jacob Moalem, MD, FACS, general surgery, Rochester, NY
• Leigh A. Neumayer, MD, FACS, general surgery, Tucson, AZ; Chair, ACS Board of Regents
• Naveen F. Sangji, MD (also Resident and Fellow Section delegate), general surgery resident, Boston, MA
Dr. Turner is Director, ACS Division of Member Services, Chicago, IL.
Mr. Sutton is Manager, State Affairs, ACS Division of Advocacy and Health Policy, Washington, DC.
Severe maternal morbidity climbs; racial disparities persist
The rate of severe maternal morbidity in the United States has climbed steadily since 2006, increasing 45% overall in the decade ending in 2015, according to a new report from the Agency for Healthcare Research and Quality that also found large ethnic and racial, geographic, and socioeconomic variation in rates of severe maternal morbidity.
The longstanding increased risk for severe maternal morbidity for black women, compared with white women, continued essentially unchanged, with black women 112%-115% more likely to experience any of 21 conditions and procedures that defined severe maternal morbidity in the report. Disparities also existed between white women and those of Hispanic or Asian/Pacific Islander origin, but those gaps are narrowing, according to the report.
“Black women, Hispanic women, and women of other races/ethnicities were overrepresented among deliveries involving severe maternal morbidity, as compared with white women,” wrote Kathryn Fingar, PhD, and her coauthors. “White women constituted a lower percentage of deliveries with any severe maternal morbidity than they did other deliveries” – 23% lower.
The 21 indicators, developed by the Centers for Disease Control and Prevention, range from conditions such as renal failure and sepsis to in-hospital procedures such as blood transfusion and hysterectomy. Women were considered to have severe maternal morbidity if any of the indicators were present, regardless of whether in-hospital death occurred.

Dr. Fingar of IBM Watson Health, Cambridge, Mass., and her collaborators summarized data from AHRQ’s Healthcare Cost and Utilization Project (H-CUP) in the statistical brief. The two most common procedures that indicate severe maternal morbidity are blood transfusion and hysterectomy, and these indicators were tracked in deliveries where women had a condition that served as one of the severe maternal morbidity indicators.
Dr. Fingar and her colleagues noted that they excluded data for the final quarter of 2015, because that is when the transition from the 9th to the 10th edition of the International Classifications of Diseases was made.
In addition to the overall increase from 101.3 to 146.6 women per 10,000 experiencing any severe maternal morbidity, the incidence of blood transfusion during a delivery hospitalization, either alone or in conjunction with other indicators, rose from 78.9 to 121.1 women per 10,000, an increase of 54%.
Showing that increased blood transfusions were a major driver of the jump in severe maternal morbidity, the composite increase in the rates of all other indicators went from 34 to 42 per delivery hospitalization, an increase of just 24%, or less than half the increase in blood transfusions.
There was significant variation in trends over time for the rates of the other indicators: acute renal failure, shock, the need for mechanical ventilation, and sepsis all increased by at least 100% (range, 104%-134%), and the rates of aneurysm increased by 99%. Rates of other indicators fell; pulmonary edema, embolism, eclampsia, myocardial infarction, cerebrovascular disorders, serious anesthesia complications, and intraoperative heart failure and arrest all declined by 25%-53% during the data-gathering period.
Looking at the data another way, 78% of cases of severe maternal morbidity in 2006 and 83% in 2015 involved a blood transfusion, making it the most common indicator. Far fewer delivery hospitalizations involved disseminated intravascular coagulation (DIC) and hysterectomy, the next most common indicators, which were seen in just 8% of cases.
Blood transfusions were most likely in women with shock (72%), amniotic fluid embolism (63%), sickle cell disease with crisis (54%), and DIC (51%).
One in three women who experienced shock during delivery had a hysterectomy, as did more than 20% of women who experienced adult respiratory distress syndrome or cardiac arrest (or ventricular fibrillation).
Women delivering at the youngest and oldest ends of the age spectrum were more likely to experience severe maternal morbidity. For women younger than 20 years of age, the rate was 206 per 10,000. In the group of women aged 40 years and over, the rate of any severe maternal morbidity was 248 per 10,000 delivery hospitalizations. Women in their 20s and 30s had rates of 136 and 143 per 10,000, respectively.
Besides race/ethnicity and age, a variety of other patient characteristics were associated with increased rates of severe maternal morbidity and mortality, with higher rates seen in women from the poorest quartile than the wealthiest (177 versus 122 per 10,000, respectively). Women with Medicaid were more likely than those with private insurance to experience severe maternal morbidity during a delivery hospitalization (175 versus 121 per 10,000, respectively).
Similarly, severe maternal morbidity was more common in safety net hospitals and in minority-serving hospitals (182 versus 128 and 176 versus 123 per 10,000, respectively) than other hospitals.
Regionally, severe maternal morbidity was most common in the Northeast and the South, at 165 and 164 per 10,000, respectively, compared with rates of 132 and 116 in the West and Midwest, respectively.
Hospital deaths per 100,000 delivery hospitalizations were 248% higher for black than white women in 2006. By the end of 2015, that figure declined modestly to 193%, with absolute rates of 19 versus 5.5 deaths per 100,000 delivery hospitalizations for black versus white women in 2006. In 2015, the absolute rates were 11 versus 4 per 100,000.
The study was conducted by the Agency for Healthcare Research and Quality. There were no reported conflicts of interest.
SOURCE: Fingar K et al. Agency for Healthcare Research and Quality Statistical Brief #243.
The data here confirm, again, that we have a crisis in maternal health care in the United States. Not only is severe maternal morbidity increasing, but disparities in outcomes between white and black women persist, such that severe maternal morbidity is twice as common among black women as it is among white women.
It’s tempting to attribute the gap to social determinants of health that are outside the control of the obstetrician-gynecologist. But education doesn’t protect black women: the New York City Department of Health and Mental Hygiene found that non-Hispanic black women with a college degree have higher rates of severe maternal morbidity than women of other race/ethnicities who never graduated high school. It’s a stark finding that’s illustrated by the heartbreaking story of Shalon Irving, a PhD epidemiologist at the Centers for Disease Control and Prevention, whose postpartum death was reported by ProPublica last winter.
One contributing factor is likely to be implicit biases that affect how well we see and hear patients who are different from us. I was mortified to see my own implicit biases when I took the Project Implicit Social Attitudes test (https://implicit.harvard.edu/implicit/). These aren’t the beliefs that we choose – they are the patterns that we absorb from our day-to-day lives. If you search Google images for “pregnancy,” “motherhood,” or “breastfeeding,” the overwhelming majority of images feature white women, whereas the phrase “welfare queen” is used to denigrate the reproduction of women of color. That’s the world we live in, and these patterns reinforce mental short cuts that are more likely to drive our behavior when we’re pressed for time, as we are every day in clinical practice. In one study in a pediatric emergency department, the busier the shift, the higher the providers’ implicit bias at the end of the shift.
We might start by looking at the images on our web sites and hanging in our offices and hospital corridors – do they look like the women we care for? Do they celebrate pregnancy and parenthood for diverse families? And how can we make sure that we pause, before we walk into a patient’s room, so that we can be fully present to her individual strengths and vulnerabilities, and tailor our care to her as an individual?
Alison Stuebe, MD, of the University of North Carolina, Chapel Hill, is medical director of lactation services and on the steering committee for Moms Rising North Carolina, the Breastfeeding Expert Work Group for the American College of Obstetricians and Gynecologists, and a board member of the Society for Maternal-Fetal Medicine. She was asked to comment on the AHRQ report by this newspaper.
The data here confirm, again, that we have a crisis in maternal health care in the United States. Not only is severe maternal morbidity increasing, but disparities in outcomes between white and black women persist, such that severe maternal morbidity is twice as common among black women as it is among white women.
It’s tempting to attribute the gap to social determinants of health that are outside the control of the obstetrician-gynecologist. But education doesn’t protect black women: the New York City Department of Health and Mental Hygiene found that non-Hispanic black women with a college degree have higher rates of severe maternal morbidity than women of other race/ethnicities who never graduated high school. It’s a stark finding that’s illustrated by the heartbreaking story of Shalon Irving, a PhD epidemiologist at the Centers for Disease Control and Prevention, whose postpartum death was reported by ProPublica last winter.
One contributing factor is likely to be implicit biases that affect how well we see and hear patients who are different from us. I was mortified to see my own implicit biases when I took the Project Implicit Social Attitudes test (https://implicit.harvard.edu/implicit/). These aren’t the beliefs that we choose – they are the patterns that we absorb from our day-to-day lives. If you search Google images for “pregnancy,” “motherhood,” or “breastfeeding,” the overwhelming majority of images feature white women, whereas the phrase “welfare queen” is used to denigrate the reproduction of women of color. That’s the world we live in, and these patterns reinforce mental short cuts that are more likely to drive our behavior when we’re pressed for time, as we are every day in clinical practice. In one study in a pediatric emergency department, the busier the shift, the higher the providers’ implicit bias at the end of the shift.
We might start by looking at the images on our web sites and hanging in our offices and hospital corridors – do they look like the women we care for? Do they celebrate pregnancy and parenthood for diverse families? And how can we make sure that we pause, before we walk into a patient’s room, so that we can be fully present to her individual strengths and vulnerabilities, and tailor our care to her as an individual?
Alison Stuebe, MD, of the University of North Carolina, Chapel Hill, is medical director of lactation services and on the steering committee for Moms Rising North Carolina, the Breastfeeding Expert Work Group for the American College of Obstetricians and Gynecologists, and a board member of the Society for Maternal-Fetal Medicine. She was asked to comment on the AHRQ report by this newspaper.
The data here confirm, again, that we have a crisis in maternal health care in the United States. Not only is severe maternal morbidity increasing, but disparities in outcomes between white and black women persist, such that severe maternal morbidity is twice as common among black women as it is among white women.
It’s tempting to attribute the gap to social determinants of health that are outside the control of the obstetrician-gynecologist. But education doesn’t protect black women: the New York City Department of Health and Mental Hygiene found that non-Hispanic black women with a college degree have higher rates of severe maternal morbidity than women of other race/ethnicities who never graduated high school. It’s a stark finding that’s illustrated by the heartbreaking story of Shalon Irving, a PhD epidemiologist at the Centers for Disease Control and Prevention, whose postpartum death was reported by ProPublica last winter.
One contributing factor is likely to be implicit biases that affect how well we see and hear patients who are different from us. I was mortified to see my own implicit biases when I took the Project Implicit Social Attitudes test (https://implicit.harvard.edu/implicit/). These aren’t the beliefs that we choose – they are the patterns that we absorb from our day-to-day lives. If you search Google images for “pregnancy,” “motherhood,” or “breastfeeding,” the overwhelming majority of images feature white women, whereas the phrase “welfare queen” is used to denigrate the reproduction of women of color. That’s the world we live in, and these patterns reinforce mental short cuts that are more likely to drive our behavior when we’re pressed for time, as we are every day in clinical practice. In one study in a pediatric emergency department, the busier the shift, the higher the providers’ implicit bias at the end of the shift.
We might start by looking at the images on our web sites and hanging in our offices and hospital corridors – do they look like the women we care for? Do they celebrate pregnancy and parenthood for diverse families? And how can we make sure that we pause, before we walk into a patient’s room, so that we can be fully present to her individual strengths and vulnerabilities, and tailor our care to her as an individual?
Alison Stuebe, MD, of the University of North Carolina, Chapel Hill, is medical director of lactation services and on the steering committee for Moms Rising North Carolina, the Breastfeeding Expert Work Group for the American College of Obstetricians and Gynecologists, and a board member of the Society for Maternal-Fetal Medicine. She was asked to comment on the AHRQ report by this newspaper.
The rate of severe maternal morbidity in the United States has climbed steadily since 2006, increasing 45% overall in the decade ending in 2015, according to a new report from the Agency for Healthcare Research and Quality that also found large ethnic and racial, geographic, and socioeconomic variation in rates of severe maternal morbidity.
The longstanding increased risk for severe maternal morbidity for black women, compared with white women, continued essentially unchanged, with black women 112%-115% more likely to experience any of 21 conditions and procedures that defined severe maternal morbidity in the report. Disparities also existed between white women and those of Hispanic or Asian/Pacific Islander origin, but those gaps are narrowing, according to the report.
“Black women, Hispanic women, and women of other races/ethnicities were overrepresented among deliveries involving severe maternal morbidity, as compared with white women,” wrote Kathryn Fingar, PhD, and her coauthors. “White women constituted a lower percentage of deliveries with any severe maternal morbidity than they did other deliveries” – 23% lower.
The 21 indicators, developed by the Centers for Disease Control and Prevention, range from conditions such as renal failure and sepsis to in-hospital procedures such as blood transfusion and hysterectomy. Women were considered to have severe maternal morbidity if any of the indicators were present, regardless of whether in-hospital death occurred.

Dr. Fingar of IBM Watson Health, Cambridge, Mass., and her collaborators summarized data from AHRQ’s Healthcare Cost and Utilization Project (H-CUP) in the statistical brief. The two most common procedures that indicate severe maternal morbidity are blood transfusion and hysterectomy, and these indicators were tracked in deliveries where women had a condition that served as one of the severe maternal morbidity indicators.
Dr. Fingar and her colleagues noted that they excluded data for the final quarter of 2015, because that is when the transition from the 9th to the 10th edition of the International Classifications of Diseases was made.
In addition to the overall increase from 101.3 to 146.6 women per 10,000 experiencing any severe maternal morbidity, the incidence of blood transfusion during a delivery hospitalization, either alone or in conjunction with other indicators, rose from 78.9 to 121.1 women per 10,000, an increase of 54%.
Showing that increased blood transfusions were a major driver of the jump in severe maternal morbidity, the composite increase in the rates of all other indicators went from 34 to 42 per delivery hospitalization, an increase of just 24%, or less than half the increase in blood transfusions.
There was significant variation in trends over time for the rates of the other indicators: acute renal failure, shock, the need for mechanical ventilation, and sepsis all increased by at least 100% (range, 104%-134%), and the rates of aneurysm increased by 99%. Rates of other indicators fell; pulmonary edema, embolism, eclampsia, myocardial infarction, cerebrovascular disorders, serious anesthesia complications, and intraoperative heart failure and arrest all declined by 25%-53% during the data-gathering period.
Looking at the data another way, 78% of cases of severe maternal morbidity in 2006 and 83% in 2015 involved a blood transfusion, making it the most common indicator. Far fewer delivery hospitalizations involved disseminated intravascular coagulation (DIC) and hysterectomy, the next most common indicators, which were seen in just 8% of cases.
Blood transfusions were most likely in women with shock (72%), amniotic fluid embolism (63%), sickle cell disease with crisis (54%), and DIC (51%).
One in three women who experienced shock during delivery had a hysterectomy, as did more than 20% of women who experienced adult respiratory distress syndrome or cardiac arrest (or ventricular fibrillation).
Women delivering at the youngest and oldest ends of the age spectrum were more likely to experience severe maternal morbidity. For women younger than 20 years of age, the rate was 206 per 10,000. In the group of women aged 40 years and over, the rate of any severe maternal morbidity was 248 per 10,000 delivery hospitalizations. Women in their 20s and 30s had rates of 136 and 143 per 10,000, respectively.
Besides race/ethnicity and age, a variety of other patient characteristics were associated with increased rates of severe maternal morbidity and mortality, with higher rates seen in women from the poorest quartile than the wealthiest (177 versus 122 per 10,000, respectively). Women with Medicaid were more likely than those with private insurance to experience severe maternal morbidity during a delivery hospitalization (175 versus 121 per 10,000, respectively).
Similarly, severe maternal morbidity was more common in safety net hospitals and in minority-serving hospitals (182 versus 128 and 176 versus 123 per 10,000, respectively) than other hospitals.
Regionally, severe maternal morbidity was most common in the Northeast and the South, at 165 and 164 per 10,000, respectively, compared with rates of 132 and 116 in the West and Midwest, respectively.
Hospital deaths per 100,000 delivery hospitalizations were 248% higher for black than white women in 2006. By the end of 2015, that figure declined modestly to 193%, with absolute rates of 19 versus 5.5 deaths per 100,000 delivery hospitalizations for black versus white women in 2006. In 2015, the absolute rates were 11 versus 4 per 100,000.
The study was conducted by the Agency for Healthcare Research and Quality. There were no reported conflicts of interest.
SOURCE: Fingar K et al. Agency for Healthcare Research and Quality Statistical Brief #243.
The rate of severe maternal morbidity in the United States has climbed steadily since 2006, increasing 45% overall in the decade ending in 2015, according to a new report from the Agency for Healthcare Research and Quality that also found large ethnic and racial, geographic, and socioeconomic variation in rates of severe maternal morbidity.
The longstanding increased risk for severe maternal morbidity for black women, compared with white women, continued essentially unchanged, with black women 112%-115% more likely to experience any of 21 conditions and procedures that defined severe maternal morbidity in the report. Disparities also existed between white women and those of Hispanic or Asian/Pacific Islander origin, but those gaps are narrowing, according to the report.
“Black women, Hispanic women, and women of other races/ethnicities were overrepresented among deliveries involving severe maternal morbidity, as compared with white women,” wrote Kathryn Fingar, PhD, and her coauthors. “White women constituted a lower percentage of deliveries with any severe maternal morbidity than they did other deliveries” – 23% lower.
The 21 indicators, developed by the Centers for Disease Control and Prevention, range from conditions such as renal failure and sepsis to in-hospital procedures such as blood transfusion and hysterectomy. Women were considered to have severe maternal morbidity if any of the indicators were present, regardless of whether in-hospital death occurred.

Dr. Fingar of IBM Watson Health, Cambridge, Mass., and her collaborators summarized data from AHRQ’s Healthcare Cost and Utilization Project (H-CUP) in the statistical brief. The two most common procedures that indicate severe maternal morbidity are blood transfusion and hysterectomy, and these indicators were tracked in deliveries where women had a condition that served as one of the severe maternal morbidity indicators.
Dr. Fingar and her colleagues noted that they excluded data for the final quarter of 2015, because that is when the transition from the 9th to the 10th edition of the International Classifications of Diseases was made.
In addition to the overall increase from 101.3 to 146.6 women per 10,000 experiencing any severe maternal morbidity, the incidence of blood transfusion during a delivery hospitalization, either alone or in conjunction with other indicators, rose from 78.9 to 121.1 women per 10,000, an increase of 54%.
Showing that increased blood transfusions were a major driver of the jump in severe maternal morbidity, the composite increase in the rates of all other indicators went from 34 to 42 per delivery hospitalization, an increase of just 24%, or less than half the increase in blood transfusions.
There was significant variation in trends over time for the rates of the other indicators: acute renal failure, shock, the need for mechanical ventilation, and sepsis all increased by at least 100% (range, 104%-134%), and the rates of aneurysm increased by 99%. Rates of other indicators fell; pulmonary edema, embolism, eclampsia, myocardial infarction, cerebrovascular disorders, serious anesthesia complications, and intraoperative heart failure and arrest all declined by 25%-53% during the data-gathering period.
Looking at the data another way, 78% of cases of severe maternal morbidity in 2006 and 83% in 2015 involved a blood transfusion, making it the most common indicator. Far fewer delivery hospitalizations involved disseminated intravascular coagulation (DIC) and hysterectomy, the next most common indicators, which were seen in just 8% of cases.
Blood transfusions were most likely in women with shock (72%), amniotic fluid embolism (63%), sickle cell disease with crisis (54%), and DIC (51%).
One in three women who experienced shock during delivery had a hysterectomy, as did more than 20% of women who experienced adult respiratory distress syndrome or cardiac arrest (or ventricular fibrillation).
Women delivering at the youngest and oldest ends of the age spectrum were more likely to experience severe maternal morbidity. For women younger than 20 years of age, the rate was 206 per 10,000. In the group of women aged 40 years and over, the rate of any severe maternal morbidity was 248 per 10,000 delivery hospitalizations. Women in their 20s and 30s had rates of 136 and 143 per 10,000, respectively.
Besides race/ethnicity and age, a variety of other patient characteristics were associated with increased rates of severe maternal morbidity and mortality, with higher rates seen in women from the poorest quartile than the wealthiest (177 versus 122 per 10,000, respectively). Women with Medicaid were more likely than those with private insurance to experience severe maternal morbidity during a delivery hospitalization (175 versus 121 per 10,000, respectively).
Similarly, severe maternal morbidity was more common in safety net hospitals and in minority-serving hospitals (182 versus 128 and 176 versus 123 per 10,000, respectively) than other hospitals.
Regionally, severe maternal morbidity was most common in the Northeast and the South, at 165 and 164 per 10,000, respectively, compared with rates of 132 and 116 in the West and Midwest, respectively.
Hospital deaths per 100,000 delivery hospitalizations were 248% higher for black than white women in 2006. By the end of 2015, that figure declined modestly to 193%, with absolute rates of 19 versus 5.5 deaths per 100,000 delivery hospitalizations for black versus white women in 2006. In 2015, the absolute rates were 11 versus 4 per 100,000.
The study was conducted by the Agency for Healthcare Research and Quality. There were no reported conflicts of interest.
SOURCE: Fingar K et al. Agency for Healthcare Research and Quality Statistical Brief #243.
FROM AN AHRQ STATISTICAL BRIEF
Key clinical point:
Major finding: Black women were over 100% more likely to experience severe maternal morbidity, compared with white women.
Study details: Statistical analysis of Healthcare Cost and Utilization Project (H-CUP) data.
Disclosures: The study was funded by the Agency for Healthcare Research and Quality. The authors reported no conflicts of interest.
Source: Fingar K et al. Agency for Healthcare Research and Quality Statistical Brief #243.