User login
Starting a blog
. Health information is one of the most popular topics people search for online. Starting a physician blog can provide your practice with promotional and marketing benefits that you may have a difficult time finding elsewhere. A blog can be an effective way to drive traffic to your website, establish yourself as an authority or expert in a particular area, and stay on the radar with your patients. However, there are a few things you should think about before you start.
Start by determining what you want to accomplish. Do you want to reach quantitative milestones, like a certain number of followers, or are you looking to increase your website traffic from potential patients? One goal will probably be to augment the health knowledge of your patients. Decide early on what your benchmarks will be and how you will track them.
Next, determine who your potential readers are. Initially, most will probably be local (your existing patient base and their family and friends), but your audience may expand geographically as your blog gains in popularity.
By now, you probably realize that blogging will require a significant commitment, over and above the time needed to write the content. Decide whether you have the time and energy to take this on yourself, or whether help will be needed. Ideally, you should have one person in charge of all your social media efforts, so that everything is consistent and has the same voice. That person can be in-house, or you can outsource to any of the many companies that administer blogs and other media functions. (As always, I have no financial interest in any company or service mentioned in this column.)
The advantage of hiring an outside administrator is that a professionally designed blog will be far more attractive and polished than anything you could build yourself. Furthermore, an experienced designer will employ “search engine optimization” (SEO), meaning that content will be created using key words and phrases that will make it readily visible to search engine users.
You can leave design and SEO to the pros, but don’t delegate the content itself; as captain of the ship you are responsible for all the facts and opinions on your blog. You may not be up to writing everything yourself, but anything you don’t write personally needs to be scrutinized by you personally to make sure that it is factually accurate and reflects your personal view. And remember that, once it’s online, it’s online forever; consider the ramifications of anything you post on any site – yours or others – before hitting the “send” button. “The most damaging item about you,” one consultant told me, “could well be something you post yourself.” Just ask any of several prominent politicians who have famously sabotaged their own careers online.
That said, don’t be shy about creating content. Patients appreciate factual information, but they value your opinions too. Give people content that will be of interest or benefit to them. This can include health-related tips, reminders, suggestions, whatever. If they are interested in it, they will keep reading and may even share it with others. You should also write about subjects – medical and otherwise – that interest you personally. If you have expertise in a particular field, be sure to write about that.
Your practice is a local business, so localize your blog to attract people from your area. Be sure to include local city keywords in your writing. You may also want to post about local events in which your practice is involved.
Try to avoid political diatribes. While most physicians have strong political opinions, and some are not shy about expressing them, there are many venues that are more appropriate for those discussions than medical blogs. Also avoid outright sales pitches. It’s fine to describe procedures that you offer, but aggressive solicitation will only turn readers off.
Keep any medical advice in general terms; don’t use any specific examples that might make a patient identifiable and generate a HIPAA violation.
If you are having trouble growing your readership, use your practice’s Facebook page to push blog updates into patients’ feeds. Additionally, track Twitter hashtags that are relevant to your practice, and use them to find existing online communities with an interest in your blog’s topics.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
*This article was updated 10/17/2022.
. Health information is one of the most popular topics people search for online. Starting a physician blog can provide your practice with promotional and marketing benefits that you may have a difficult time finding elsewhere. A blog can be an effective way to drive traffic to your website, establish yourself as an authority or expert in a particular area, and stay on the radar with your patients. However, there are a few things you should think about before you start.
Start by determining what you want to accomplish. Do you want to reach quantitative milestones, like a certain number of followers, or are you looking to increase your website traffic from potential patients? One goal will probably be to augment the health knowledge of your patients. Decide early on what your benchmarks will be and how you will track them.
Next, determine who your potential readers are. Initially, most will probably be local (your existing patient base and their family and friends), but your audience may expand geographically as your blog gains in popularity.
By now, you probably realize that blogging will require a significant commitment, over and above the time needed to write the content. Decide whether you have the time and energy to take this on yourself, or whether help will be needed. Ideally, you should have one person in charge of all your social media efforts, so that everything is consistent and has the same voice. That person can be in-house, or you can outsource to any of the many companies that administer blogs and other media functions. (As always, I have no financial interest in any company or service mentioned in this column.)
The advantage of hiring an outside administrator is that a professionally designed blog will be far more attractive and polished than anything you could build yourself. Furthermore, an experienced designer will employ “search engine optimization” (SEO), meaning that content will be created using key words and phrases that will make it readily visible to search engine users.
You can leave design and SEO to the pros, but don’t delegate the content itself; as captain of the ship you are responsible for all the facts and opinions on your blog. You may not be up to writing everything yourself, but anything you don’t write personally needs to be scrutinized by you personally to make sure that it is factually accurate and reflects your personal view. And remember that, once it’s online, it’s online forever; consider the ramifications of anything you post on any site – yours or others – before hitting the “send” button. “The most damaging item about you,” one consultant told me, “could well be something you post yourself.” Just ask any of several prominent politicians who have famously sabotaged their own careers online.
That said, don’t be shy about creating content. Patients appreciate factual information, but they value your opinions too. Give people content that will be of interest or benefit to them. This can include health-related tips, reminders, suggestions, whatever. If they are interested in it, they will keep reading and may even share it with others. You should also write about subjects – medical and otherwise – that interest you personally. If you have expertise in a particular field, be sure to write about that.
Your practice is a local business, so localize your blog to attract people from your area. Be sure to include local city keywords in your writing. You may also want to post about local events in which your practice is involved.
Try to avoid political diatribes. While most physicians have strong political opinions, and some are not shy about expressing them, there are many venues that are more appropriate for those discussions than medical blogs. Also avoid outright sales pitches. It’s fine to describe procedures that you offer, but aggressive solicitation will only turn readers off.
Keep any medical advice in general terms; don’t use any specific examples that might make a patient identifiable and generate a HIPAA violation.
If you are having trouble growing your readership, use your practice’s Facebook page to push blog updates into patients’ feeds. Additionally, track Twitter hashtags that are relevant to your practice, and use them to find existing online communities with an interest in your blog’s topics.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
*This article was updated 10/17/2022.
. Health information is one of the most popular topics people search for online. Starting a physician blog can provide your practice with promotional and marketing benefits that you may have a difficult time finding elsewhere. A blog can be an effective way to drive traffic to your website, establish yourself as an authority or expert in a particular area, and stay on the radar with your patients. However, there are a few things you should think about before you start.
Start by determining what you want to accomplish. Do you want to reach quantitative milestones, like a certain number of followers, or are you looking to increase your website traffic from potential patients? One goal will probably be to augment the health knowledge of your patients. Decide early on what your benchmarks will be and how you will track them.
Next, determine who your potential readers are. Initially, most will probably be local (your existing patient base and their family and friends), but your audience may expand geographically as your blog gains in popularity.
By now, you probably realize that blogging will require a significant commitment, over and above the time needed to write the content. Decide whether you have the time and energy to take this on yourself, or whether help will be needed. Ideally, you should have one person in charge of all your social media efforts, so that everything is consistent and has the same voice. That person can be in-house, or you can outsource to any of the many companies that administer blogs and other media functions. (As always, I have no financial interest in any company or service mentioned in this column.)
The advantage of hiring an outside administrator is that a professionally designed blog will be far more attractive and polished than anything you could build yourself. Furthermore, an experienced designer will employ “search engine optimization” (SEO), meaning that content will be created using key words and phrases that will make it readily visible to search engine users.
You can leave design and SEO to the pros, but don’t delegate the content itself; as captain of the ship you are responsible for all the facts and opinions on your blog. You may not be up to writing everything yourself, but anything you don’t write personally needs to be scrutinized by you personally to make sure that it is factually accurate and reflects your personal view. And remember that, once it’s online, it’s online forever; consider the ramifications of anything you post on any site – yours or others – before hitting the “send” button. “The most damaging item about you,” one consultant told me, “could well be something you post yourself.” Just ask any of several prominent politicians who have famously sabotaged their own careers online.
That said, don’t be shy about creating content. Patients appreciate factual information, but they value your opinions too. Give people content that will be of interest or benefit to them. This can include health-related tips, reminders, suggestions, whatever. If they are interested in it, they will keep reading and may even share it with others. You should also write about subjects – medical and otherwise – that interest you personally. If you have expertise in a particular field, be sure to write about that.
Your practice is a local business, so localize your blog to attract people from your area. Be sure to include local city keywords in your writing. You may also want to post about local events in which your practice is involved.
Try to avoid political diatribes. While most physicians have strong political opinions, and some are not shy about expressing them, there are many venues that are more appropriate for those discussions than medical blogs. Also avoid outright sales pitches. It’s fine to describe procedures that you offer, but aggressive solicitation will only turn readers off.
Keep any medical advice in general terms; don’t use any specific examples that might make a patient identifiable and generate a HIPAA violation.
If you are having trouble growing your readership, use your practice’s Facebook page to push blog updates into patients’ feeds. Additionally, track Twitter hashtags that are relevant to your practice, and use them to find existing online communities with an interest in your blog’s topics.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
*This article was updated 10/17/2022.
Tourette syndrome: Diagnosis is key for best care
Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), and autism spectrum disorder (ASD) share significant overlap in symptomatology, and it can be challenging at times to distinguish between these conditions. Being able to do so, however, can help guide more targeted interventions and accommodations to optimize a patient’s level of functioning.
Case example
A healthy, bright 6-year-old boy is referred by his family doctor to an academic medical center for a full team evaluation because of suspicion of ASD, after having already been diagnosed with ADHD at the age of 5. His difficulties with inattention, impulsivity, and hyperactivity, as well as his behavioral rigidities and sensory avoidant and sensory seeking behaviors have caused functional impairments for him in his kindergarten classroom. He has been penalized with removal of recess on more than one occasion. A low dose of a stimulant had been tried but resulted in a perceived increase in disruptive behaviors.
The boy, while hyperkinetic and often paying poor attention, is quite capable of high-quality and well-modulated eye contact paired with typical social referencing and reciprocity when actively engaging with the examiner and his parents. He does have a reported history of serial fixated interests and some repetitive behaviors but is also noted to be flexible in his interpersonal style, maintains other varied and typical interests, easily directs affect, utilizes a wide array of fluid gestures paired naturally with verbal communication, and shares enjoyment with smoothly coordinated gaze. He has mild articulation errors but uses pronouns appropriately and has no scripted speech or echolalia, though does engage in some whispered palilalia intermittently.
He is generally quite cooperative and redirectable when focused and has a completely normal physical and neurologic examination. During the visit, the doctor notices the boy making an intermittent honking sound, which parents report as an attention-seeking strategy during times of stress. Further physician-guided information gathering around other repetitive noises and movements elicits a history of engagement in repetitive hand-to-groin movements, some exaggerated blinking, and a number of other waxing and waning subtle motor and phonic tics with onset in preschool. These noises and movements have generally been identified as “fidgeting” and “misbehaving” by well-meaning caregivers in the home and school environments.
Both Tourette syndrome and ASD are more common in males, with stereotyped patterns of movements and behaviors; anxious, obsessive, and compulsive behaviors resulting in behavioral rigidities; sensory sensitivities; and increased rates of hyperkinesis with decreased impulse control which result in increased sensory-seeking behaviors. Diagnostic criteria for Tourette syndrome are met when a child has had multiple motor tics and at least one phonic tic present for at least 1 year, with tic-free intervals lasting no longer than 3 months, and with onset before the age of 18. Typically, tics emerge in late preschool and early grade school, and some children even develop repetitive movements as early as toddlerhood. Tics tend to worsen around the peripubertal era, then often generally improve in the teen years. Tic types, frequency, and severity general fluctuate over time.
Forty percent of children with Tourette syndrome also meet criteria for OCD, with many more having OCD traits, and about 65% of children with Tourette syndrome also meet criteria for ADHD, with many more having ADHD traits. OCD can lead to more rigid and directive social interactions in children as well as obsessive interests, just as ADHD can lead to less socially attuned and less cooperative behaviors, even in children who do not meet criteria for ASD.
For example, a child with OCD in the absence of ASD may still “police” other kids in class and be overly focused on the rules of a game, which may become a social liability. Likewise, a child with ADHD in the absence of ASD may be so distractible that focusing on what other kids are saying and their paired facial expressions is compromised, leading to poor-quality social reciprocity during interactions with peers. Given the remarkable overlap in shared symptoms, it is essential for pediatric providers to consider Tourette syndrome in the differential for any child with repetitive movements and behaviors in addition to ASD and a wide array of other neurodevelopment differences, including global developmental delays and intellectual disabilities. This is of particular importance as the diagnosis of Tourette syndrome can be used to gain access to developmental disability services if the condition has resulted in true adaptive impairments.
It is determined that the boy does in fact meet criteria for ADHD, but also for OCD and Tourette syndrome. Both his Autism Diagnostic Observation Schedule and DSM-5–influenced autism interview are found to be in the nonclinical ranges, given his quality of communication, social engagement, imaginative play, and varied interests. A diagnosis of ASD is not felt to be an appropriate conceptualization of his neurodevelopmental differences. He is started on a low dose of guanfacine, which induces a decline in tics, impulsivity, and hyperkinesis. He is given a 504 plan in school that includes scheduled “tic breaks,” sensory fidgets for use in the classroom, extra movement opportunities as needed, and utilization of a gentle cueing system between him and his teacher for low-key redirection of disruptive behaviors. He is no longer penalized for inattention or tics, and his 504 plan protects him from the use of recess removal as a behavioral modification strategy.
His parents enroll him in the community swim program for extra exercise, focus on decreasing screen time, and give him an earlier bedtime to help decrease his tics and rigidities, while improving his ability to self-regulate. Eventually, a low dose of a newer-generation stimulant is added to his guanfacine, with excellent results and only a mild increase in tolerable tics.
The child in the vignette did well with a 504 plan based on his medical diagnoses, though if related learning difficulties had persisted, eligibility under Other Health Impaired could be used to provide eligibility for an Individualized Education Plan. Alpha-agonists can be helpful for symptom control in those with Tourette syndrome by simultaneously treating tics, hyperkinesis, and impulsivity, while decreasing the risk of tic exacerbation with use of stimulants. Overall, understanding the neurodiversity related to Tourette syndrome can help providers advocate for home and community-based supports to optimize general functioning and quality of life.
Dr. Roth is a developmental and behavioral pediatrician in Eugene, Ore. She has no conflicts of interest.
References
Darrow S et al. J Am Acad Child Adolescent Psych. 2017;56(7):610-7.
AAP Section on Developmental and Behavioral Pediatrics. Developmental and Behavioral Pediatrics. Voigt RG et al, eds. 2018: American Academy of Pediatrics.
Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), and autism spectrum disorder (ASD) share significant overlap in symptomatology, and it can be challenging at times to distinguish between these conditions. Being able to do so, however, can help guide more targeted interventions and accommodations to optimize a patient’s level of functioning.
Case example
A healthy, bright 6-year-old boy is referred by his family doctor to an academic medical center for a full team evaluation because of suspicion of ASD, after having already been diagnosed with ADHD at the age of 5. His difficulties with inattention, impulsivity, and hyperactivity, as well as his behavioral rigidities and sensory avoidant and sensory seeking behaviors have caused functional impairments for him in his kindergarten classroom. He has been penalized with removal of recess on more than one occasion. A low dose of a stimulant had been tried but resulted in a perceived increase in disruptive behaviors.
The boy, while hyperkinetic and often paying poor attention, is quite capable of high-quality and well-modulated eye contact paired with typical social referencing and reciprocity when actively engaging with the examiner and his parents. He does have a reported history of serial fixated interests and some repetitive behaviors but is also noted to be flexible in his interpersonal style, maintains other varied and typical interests, easily directs affect, utilizes a wide array of fluid gestures paired naturally with verbal communication, and shares enjoyment with smoothly coordinated gaze. He has mild articulation errors but uses pronouns appropriately and has no scripted speech or echolalia, though does engage in some whispered palilalia intermittently.
He is generally quite cooperative and redirectable when focused and has a completely normal physical and neurologic examination. During the visit, the doctor notices the boy making an intermittent honking sound, which parents report as an attention-seeking strategy during times of stress. Further physician-guided information gathering around other repetitive noises and movements elicits a history of engagement in repetitive hand-to-groin movements, some exaggerated blinking, and a number of other waxing and waning subtle motor and phonic tics with onset in preschool. These noises and movements have generally been identified as “fidgeting” and “misbehaving” by well-meaning caregivers in the home and school environments.
Both Tourette syndrome and ASD are more common in males, with stereotyped patterns of movements and behaviors; anxious, obsessive, and compulsive behaviors resulting in behavioral rigidities; sensory sensitivities; and increased rates of hyperkinesis with decreased impulse control which result in increased sensory-seeking behaviors. Diagnostic criteria for Tourette syndrome are met when a child has had multiple motor tics and at least one phonic tic present for at least 1 year, with tic-free intervals lasting no longer than 3 months, and with onset before the age of 18. Typically, tics emerge in late preschool and early grade school, and some children even develop repetitive movements as early as toddlerhood. Tics tend to worsen around the peripubertal era, then often generally improve in the teen years. Tic types, frequency, and severity general fluctuate over time.
Forty percent of children with Tourette syndrome also meet criteria for OCD, with many more having OCD traits, and about 65% of children with Tourette syndrome also meet criteria for ADHD, with many more having ADHD traits. OCD can lead to more rigid and directive social interactions in children as well as obsessive interests, just as ADHD can lead to less socially attuned and less cooperative behaviors, even in children who do not meet criteria for ASD.
For example, a child with OCD in the absence of ASD may still “police” other kids in class and be overly focused on the rules of a game, which may become a social liability. Likewise, a child with ADHD in the absence of ASD may be so distractible that focusing on what other kids are saying and their paired facial expressions is compromised, leading to poor-quality social reciprocity during interactions with peers. Given the remarkable overlap in shared symptoms, it is essential for pediatric providers to consider Tourette syndrome in the differential for any child with repetitive movements and behaviors in addition to ASD and a wide array of other neurodevelopment differences, including global developmental delays and intellectual disabilities. This is of particular importance as the diagnosis of Tourette syndrome can be used to gain access to developmental disability services if the condition has resulted in true adaptive impairments.
It is determined that the boy does in fact meet criteria for ADHD, but also for OCD and Tourette syndrome. Both his Autism Diagnostic Observation Schedule and DSM-5–influenced autism interview are found to be in the nonclinical ranges, given his quality of communication, social engagement, imaginative play, and varied interests. A diagnosis of ASD is not felt to be an appropriate conceptualization of his neurodevelopmental differences. He is started on a low dose of guanfacine, which induces a decline in tics, impulsivity, and hyperkinesis. He is given a 504 plan in school that includes scheduled “tic breaks,” sensory fidgets for use in the classroom, extra movement opportunities as needed, and utilization of a gentle cueing system between him and his teacher for low-key redirection of disruptive behaviors. He is no longer penalized for inattention or tics, and his 504 plan protects him from the use of recess removal as a behavioral modification strategy.
His parents enroll him in the community swim program for extra exercise, focus on decreasing screen time, and give him an earlier bedtime to help decrease his tics and rigidities, while improving his ability to self-regulate. Eventually, a low dose of a newer-generation stimulant is added to his guanfacine, with excellent results and only a mild increase in tolerable tics.
The child in the vignette did well with a 504 plan based on his medical diagnoses, though if related learning difficulties had persisted, eligibility under Other Health Impaired could be used to provide eligibility for an Individualized Education Plan. Alpha-agonists can be helpful for symptom control in those with Tourette syndrome by simultaneously treating tics, hyperkinesis, and impulsivity, while decreasing the risk of tic exacerbation with use of stimulants. Overall, understanding the neurodiversity related to Tourette syndrome can help providers advocate for home and community-based supports to optimize general functioning and quality of life.
Dr. Roth is a developmental and behavioral pediatrician in Eugene, Ore. She has no conflicts of interest.
References
Darrow S et al. J Am Acad Child Adolescent Psych. 2017;56(7):610-7.
AAP Section on Developmental and Behavioral Pediatrics. Developmental and Behavioral Pediatrics. Voigt RG et al, eds. 2018: American Academy of Pediatrics.
Tourette syndrome, attention-deficit/hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), and autism spectrum disorder (ASD) share significant overlap in symptomatology, and it can be challenging at times to distinguish between these conditions. Being able to do so, however, can help guide more targeted interventions and accommodations to optimize a patient’s level of functioning.
Case example
A healthy, bright 6-year-old boy is referred by his family doctor to an academic medical center for a full team evaluation because of suspicion of ASD, after having already been diagnosed with ADHD at the age of 5. His difficulties with inattention, impulsivity, and hyperactivity, as well as his behavioral rigidities and sensory avoidant and sensory seeking behaviors have caused functional impairments for him in his kindergarten classroom. He has been penalized with removal of recess on more than one occasion. A low dose of a stimulant had been tried but resulted in a perceived increase in disruptive behaviors.
The boy, while hyperkinetic and often paying poor attention, is quite capable of high-quality and well-modulated eye contact paired with typical social referencing and reciprocity when actively engaging with the examiner and his parents. He does have a reported history of serial fixated interests and some repetitive behaviors but is also noted to be flexible in his interpersonal style, maintains other varied and typical interests, easily directs affect, utilizes a wide array of fluid gestures paired naturally with verbal communication, and shares enjoyment with smoothly coordinated gaze. He has mild articulation errors but uses pronouns appropriately and has no scripted speech or echolalia, though does engage in some whispered palilalia intermittently.
He is generally quite cooperative and redirectable when focused and has a completely normal physical and neurologic examination. During the visit, the doctor notices the boy making an intermittent honking sound, which parents report as an attention-seeking strategy during times of stress. Further physician-guided information gathering around other repetitive noises and movements elicits a history of engagement in repetitive hand-to-groin movements, some exaggerated blinking, and a number of other waxing and waning subtle motor and phonic tics with onset in preschool. These noises and movements have generally been identified as “fidgeting” and “misbehaving” by well-meaning caregivers in the home and school environments.
Both Tourette syndrome and ASD are more common in males, with stereotyped patterns of movements and behaviors; anxious, obsessive, and compulsive behaviors resulting in behavioral rigidities; sensory sensitivities; and increased rates of hyperkinesis with decreased impulse control which result in increased sensory-seeking behaviors. Diagnostic criteria for Tourette syndrome are met when a child has had multiple motor tics and at least one phonic tic present for at least 1 year, with tic-free intervals lasting no longer than 3 months, and with onset before the age of 18. Typically, tics emerge in late preschool and early grade school, and some children even develop repetitive movements as early as toddlerhood. Tics tend to worsen around the peripubertal era, then often generally improve in the teen years. Tic types, frequency, and severity general fluctuate over time.
Forty percent of children with Tourette syndrome also meet criteria for OCD, with many more having OCD traits, and about 65% of children with Tourette syndrome also meet criteria for ADHD, with many more having ADHD traits. OCD can lead to more rigid and directive social interactions in children as well as obsessive interests, just as ADHD can lead to less socially attuned and less cooperative behaviors, even in children who do not meet criteria for ASD.
For example, a child with OCD in the absence of ASD may still “police” other kids in class and be overly focused on the rules of a game, which may become a social liability. Likewise, a child with ADHD in the absence of ASD may be so distractible that focusing on what other kids are saying and their paired facial expressions is compromised, leading to poor-quality social reciprocity during interactions with peers. Given the remarkable overlap in shared symptoms, it is essential for pediatric providers to consider Tourette syndrome in the differential for any child with repetitive movements and behaviors in addition to ASD and a wide array of other neurodevelopment differences, including global developmental delays and intellectual disabilities. This is of particular importance as the diagnosis of Tourette syndrome can be used to gain access to developmental disability services if the condition has resulted in true adaptive impairments.
It is determined that the boy does in fact meet criteria for ADHD, but also for OCD and Tourette syndrome. Both his Autism Diagnostic Observation Schedule and DSM-5–influenced autism interview are found to be in the nonclinical ranges, given his quality of communication, social engagement, imaginative play, and varied interests. A diagnosis of ASD is not felt to be an appropriate conceptualization of his neurodevelopmental differences. He is started on a low dose of guanfacine, which induces a decline in tics, impulsivity, and hyperkinesis. He is given a 504 plan in school that includes scheduled “tic breaks,” sensory fidgets for use in the classroom, extra movement opportunities as needed, and utilization of a gentle cueing system between him and his teacher for low-key redirection of disruptive behaviors. He is no longer penalized for inattention or tics, and his 504 plan protects him from the use of recess removal as a behavioral modification strategy.
His parents enroll him in the community swim program for extra exercise, focus on decreasing screen time, and give him an earlier bedtime to help decrease his tics and rigidities, while improving his ability to self-regulate. Eventually, a low dose of a newer-generation stimulant is added to his guanfacine, with excellent results and only a mild increase in tolerable tics.
The child in the vignette did well with a 504 plan based on his medical diagnoses, though if related learning difficulties had persisted, eligibility under Other Health Impaired could be used to provide eligibility for an Individualized Education Plan. Alpha-agonists can be helpful for symptom control in those with Tourette syndrome by simultaneously treating tics, hyperkinesis, and impulsivity, while decreasing the risk of tic exacerbation with use of stimulants. Overall, understanding the neurodiversity related to Tourette syndrome can help providers advocate for home and community-based supports to optimize general functioning and quality of life.
Dr. Roth is a developmental and behavioral pediatrician in Eugene, Ore. She has no conflicts of interest.
References
Darrow S et al. J Am Acad Child Adolescent Psych. 2017;56(7):610-7.
AAP Section on Developmental and Behavioral Pediatrics. Developmental and Behavioral Pediatrics. Voigt RG et al, eds. 2018: American Academy of Pediatrics.
The WPATH guidelines for treatment of adolescents with gender dysphoria have changed
The World Professional Association for Transgender Health (WPATH) is an interdisciplinary professional and educational organization devoted to transgender health. One of their activities is to produce the Standards of Care (SOC) for treatment of individuals with gender dysphoria. According to WPATH, the SOC “articulate a professional consensus about the psychiatric, psychological, medical, and surgical management of gender dysphoria and help professionals understand the parameters within which they may offer assistance to those with these conditions.” Many clinicians around the world use these guidelines to help them care for patients with gender dysphoria and diverse gender expressions.
The most recent SOC, version 8, were released on Sept. 15, 2022, after a 2-year postponement because of the pandemic. These new standards represent the first update to the SOC since version 7, which was released in 2012. Given how recent this update is, this column will attempt to summarize the changes in the new guidelines that affect children and adolescents.
One of the major differences between SOC versions 7 and 8 is that version 8 now includes a chapter specifically dedicated to the care of adolescents. Version 7 lumped children and adolescents together into one chapter. This is an important distinction for SOC 8, as it highlights that care for prepubertal youth is simply social in nature and distinct from that of pubertal adolescents. Social transition includes things such as using an affirmed name/pronouns and changing hair style and clothes. It does not include medications of any kind. Allowing these youth the time and space to explore the natural gender diversity of childhood leads to improved psychological outcomes over time and reduces adversity. Psychological support, where indicated, should be offered to gender-diverse children and their families to explore the persistence, consistence, and insistence of that child’s gender identity.
Once a child reaches puberty, medications may come into play as part of an adolescent’s transition. SOC 7 had established a minimum age of 16 before any partially reversible medications (testosterone, estrogen) were started as part of a patient’s medical transition. Starting with SOC 8, a minimum age has been removed for the initiation of gender-affirming hormone therapy. However, a patient must still have begun their natal puberty before any medication is started. A specific age was removed to acknowledge that maturity in adolescents occurs on a continuum and at different ages. SOC 8 guidelines continue to recommend that the individual’s emotional, cognitive, and psychosocial development be taken into account when determining their ability to provide consent for treatment. These individuals should still undergo a comprehensive assessment, as described below.
Similar to SOC 7, SOC 8 continues to stress the importance of a comprehensive, multidisciplinary evaluation of those adolescents who seek medical therapy as part of their transition. This allows for the exploration of additional coexisting causes of gender dysphoria, such as anxiety, depression, or other mental health conditions. If these exist, then they must be appropriately treated before any gender-affirming medical treatment is initiated. Assessments should be performed by clinicians who have training and expertise with the developmental trajectory of adolescents, as well as with common mental health conditions. These assessments are also critical, as SOC 8 acknowledges a rise in the number of adolescents who may not have had gender-diverse expression in childhood.
SOC 8 and the Endocrine Society Guidelines (see references) provide physicians and other health care professionals with a road map for addressing the needs of transgender and gender-diverse persons. By referencing these guidelines when taking care of these patients, physicians and other health care professionals will know that they are providing the most up-to-date, evidence-based care.
Dr. M. Brett Cooper is an assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
SOC 8: https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
SOC 7: https://www.wpath.org/media/cms/Documents/SOC%20v7/SOC%20V7_English2012.pdf?_t=1613669341
Endocrine Society Gender Affirming Care Guidelines: https://academic.oup.com/jcem/article/102/11/3869/4157558?login=false
The World Professional Association for Transgender Health (WPATH) is an interdisciplinary professional and educational organization devoted to transgender health. One of their activities is to produce the Standards of Care (SOC) for treatment of individuals with gender dysphoria. According to WPATH, the SOC “articulate a professional consensus about the psychiatric, psychological, medical, and surgical management of gender dysphoria and help professionals understand the parameters within which they may offer assistance to those with these conditions.” Many clinicians around the world use these guidelines to help them care for patients with gender dysphoria and diverse gender expressions.
The most recent SOC, version 8, were released on Sept. 15, 2022, after a 2-year postponement because of the pandemic. These new standards represent the first update to the SOC since version 7, which was released in 2012. Given how recent this update is, this column will attempt to summarize the changes in the new guidelines that affect children and adolescents.
One of the major differences between SOC versions 7 and 8 is that version 8 now includes a chapter specifically dedicated to the care of adolescents. Version 7 lumped children and adolescents together into one chapter. This is an important distinction for SOC 8, as it highlights that care for prepubertal youth is simply social in nature and distinct from that of pubertal adolescents. Social transition includes things such as using an affirmed name/pronouns and changing hair style and clothes. It does not include medications of any kind. Allowing these youth the time and space to explore the natural gender diversity of childhood leads to improved psychological outcomes over time and reduces adversity. Psychological support, where indicated, should be offered to gender-diverse children and their families to explore the persistence, consistence, and insistence of that child’s gender identity.
Once a child reaches puberty, medications may come into play as part of an adolescent’s transition. SOC 7 had established a minimum age of 16 before any partially reversible medications (testosterone, estrogen) were started as part of a patient’s medical transition. Starting with SOC 8, a minimum age has been removed for the initiation of gender-affirming hormone therapy. However, a patient must still have begun their natal puberty before any medication is started. A specific age was removed to acknowledge that maturity in adolescents occurs on a continuum and at different ages. SOC 8 guidelines continue to recommend that the individual’s emotional, cognitive, and psychosocial development be taken into account when determining their ability to provide consent for treatment. These individuals should still undergo a comprehensive assessment, as described below.
Similar to SOC 7, SOC 8 continues to stress the importance of a comprehensive, multidisciplinary evaluation of those adolescents who seek medical therapy as part of their transition. This allows for the exploration of additional coexisting causes of gender dysphoria, such as anxiety, depression, or other mental health conditions. If these exist, then they must be appropriately treated before any gender-affirming medical treatment is initiated. Assessments should be performed by clinicians who have training and expertise with the developmental trajectory of adolescents, as well as with common mental health conditions. These assessments are also critical, as SOC 8 acknowledges a rise in the number of adolescents who may not have had gender-diverse expression in childhood.
SOC 8 and the Endocrine Society Guidelines (see references) provide physicians and other health care professionals with a road map for addressing the needs of transgender and gender-diverse persons. By referencing these guidelines when taking care of these patients, physicians and other health care professionals will know that they are providing the most up-to-date, evidence-based care.
Dr. M. Brett Cooper is an assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
SOC 8: https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
SOC 7: https://www.wpath.org/media/cms/Documents/SOC%20v7/SOC%20V7_English2012.pdf?_t=1613669341
Endocrine Society Gender Affirming Care Guidelines: https://academic.oup.com/jcem/article/102/11/3869/4157558?login=false
The World Professional Association for Transgender Health (WPATH) is an interdisciplinary professional and educational organization devoted to transgender health. One of their activities is to produce the Standards of Care (SOC) for treatment of individuals with gender dysphoria. According to WPATH, the SOC “articulate a professional consensus about the psychiatric, psychological, medical, and surgical management of gender dysphoria and help professionals understand the parameters within which they may offer assistance to those with these conditions.” Many clinicians around the world use these guidelines to help them care for patients with gender dysphoria and diverse gender expressions.
The most recent SOC, version 8, were released on Sept. 15, 2022, after a 2-year postponement because of the pandemic. These new standards represent the first update to the SOC since version 7, which was released in 2012. Given how recent this update is, this column will attempt to summarize the changes in the new guidelines that affect children and adolescents.
One of the major differences between SOC versions 7 and 8 is that version 8 now includes a chapter specifically dedicated to the care of adolescents. Version 7 lumped children and adolescents together into one chapter. This is an important distinction for SOC 8, as it highlights that care for prepubertal youth is simply social in nature and distinct from that of pubertal adolescents. Social transition includes things such as using an affirmed name/pronouns and changing hair style and clothes. It does not include medications of any kind. Allowing these youth the time and space to explore the natural gender diversity of childhood leads to improved psychological outcomes over time and reduces adversity. Psychological support, where indicated, should be offered to gender-diverse children and their families to explore the persistence, consistence, and insistence of that child’s gender identity.
Once a child reaches puberty, medications may come into play as part of an adolescent’s transition. SOC 7 had established a minimum age of 16 before any partially reversible medications (testosterone, estrogen) were started as part of a patient’s medical transition. Starting with SOC 8, a minimum age has been removed for the initiation of gender-affirming hormone therapy. However, a patient must still have begun their natal puberty before any medication is started. A specific age was removed to acknowledge that maturity in adolescents occurs on a continuum and at different ages. SOC 8 guidelines continue to recommend that the individual’s emotional, cognitive, and psychosocial development be taken into account when determining their ability to provide consent for treatment. These individuals should still undergo a comprehensive assessment, as described below.
Similar to SOC 7, SOC 8 continues to stress the importance of a comprehensive, multidisciplinary evaluation of those adolescents who seek medical therapy as part of their transition. This allows for the exploration of additional coexisting causes of gender dysphoria, such as anxiety, depression, or other mental health conditions. If these exist, then they must be appropriately treated before any gender-affirming medical treatment is initiated. Assessments should be performed by clinicians who have training and expertise with the developmental trajectory of adolescents, as well as with common mental health conditions. These assessments are also critical, as SOC 8 acknowledges a rise in the number of adolescents who may not have had gender-diverse expression in childhood.
SOC 8 and the Endocrine Society Guidelines (see references) provide physicians and other health care professionals with a road map for addressing the needs of transgender and gender-diverse persons. By referencing these guidelines when taking care of these patients, physicians and other health care professionals will know that they are providing the most up-to-date, evidence-based care.
Dr. M. Brett Cooper is an assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
SOC 8: https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
SOC 7: https://www.wpath.org/media/cms/Documents/SOC%20v7/SOC%20V7_English2012.pdf?_t=1613669341
Endocrine Society Gender Affirming Care Guidelines: https://academic.oup.com/jcem/article/102/11/3869/4157558?login=false
Vitamin D: Recent findings and implications for clinical practice
This transcript has been edited for clarity.
Hello. This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. As a director of the Vitamin D and Omega-3 trial (VITAL), the largest randomized clinical trial in the world, I’m often asked, “How much vitamin D do we need, and should I take a vitamin D supplement?” I want to review the findings from recent randomized clinical trials and the implications for practice.
For a long time, vitamin D has been perceived as a magic bullet, a panacea, and a cure-all for many chronic health conditions such as cancer, cardiovascular disease, diabetes, bone fractures, cognitive decline, and depression. Many of the findings, though, have been from observational studies where a higher blood level of 25-hydroxy vitamin D has been linked to a lower risk for these health conditions.
We know in epidemiology that correlation doesn’t prove causation. Other factors could be involved; for example, people who have higher blood levels of vitamin D may have healthier diets, or they may be spending more time outdoors, being physically active and exposed to the sun. Some of these other factors could be lowering their risk.
When the randomized trials began to emerge, in many of these large-scale trials, the findings were generally neutral or null for cardiovascular disease, total cancer, diabetes, cognitive decline, depression, and many other health outcomes, including fracture. So, the question was asked, does this mean that vitamin D is not important to health?
To the contrary, these findings suggest that vitamin D is so essential to health that we need only small to moderate amounts of vitamin D. Vitamin D is very tightly regulated in the body – the metabolism and function of vitamin D. Even small to moderate amounts will meet the requirements for vitamin D and bone health and many other outcomes.
This is what the National Academy of Medicine, U.S. Preventive Services Task Force, and many other professional organizations have advised, that widespread screening for vitamin D deficiency and blanket universal supplementation with vitamin D would not be indicated.
The randomized trials of vitamin D, including the VITAL study, have generally not shown reductions in the major health outcomes. We found two exceptions in VITAL. We saw promising signals, including a 22% reduction in autoimmune conditions (rheumatoid arthritis and psoriasis) and a 17% reduction in advanced (metastatic or fatal) cancers. In meta-analyses of other large-scale randomized trials, the findings were a signal for a reduction in advanced cancers, even with very small doses of vitamin D (400-800 IUs daily). We tested 2,000 IUs daily in VITAL.
Overall, it’s recommended that small to moderate amounts of vitamin D are adequate, and among the healthy population, most people do not need screening or supplements.
The reduction in autoimmune diseases suggests that vitamin D may play a role in tamping down inflammation. The question has been raised about whether vitamin D is beneficial in reducing the severity of COVID illness, the need for hospitalization, and long COVID. We are looking at this question in a separate trial called VIVID (Vitamin D for COVID Trial) which tests a higher dose (> 3,000 IUs daily) of vitamin D. Those results will be available at the end of this year or early next year.
In other randomized trials of COVID and vitamin D, the results have been mixed and inconsistent, with no clear answer. During the COVID pandemic, I have generally advised that it’s reasonable to take 1,000-2,000 IUs of vitamin D daily as a form of insurance. This dose is known to be very safe. Over 5.3 years in the VITAL trial we saw that a dose of 2,000 IUs was very safe.
But it’s not essential to take a supplement. And overall, aside from some high-risk groups, most people do not need a supplement. The high-risk groups include patients in nursing homes who may have restricted diets and limited time out of doors. For people with malabsorption conditions such as Crohn’s disease, celiac disease, post–gastric bypass surgery, and those with osteoporosis who are on medications for osteoporosis, it’s still quite reasonable to prescribe calcium and vitamin D.
Recommendations for vitamin D in the generally healthy population really should focus on a healthy diet. The United States has a fortified food supply. Vitamin D is added to many foods, dairy products, and cereals, as well as beverages. Natural sources of vitamin D include fatty fish and wild mushrooms.
We should be looking at food labels (which now include vitamin D content) and try to get adequate vitamin D from our diet, and also do our best to spend time outdoors, being physically active, because it is of great benefit to our health. The general principle is that a dietary supplement will never be a substitute for a healthy diet or healthy lifestyle. And those other behaviors really should be the focus at this time.
Dr. Manson is professor of medicine and the Michael and Lee Bell Professor of Women’s Health, Harvard Medical School, and chief of the division of preventive medicine at Brigham and Women’s Hospital, both in Boston. She has received infrastructure support from Mars Symbioscience for the COSMOS trial.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hello. This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. As a director of the Vitamin D and Omega-3 trial (VITAL), the largest randomized clinical trial in the world, I’m often asked, “How much vitamin D do we need, and should I take a vitamin D supplement?” I want to review the findings from recent randomized clinical trials and the implications for practice.
For a long time, vitamin D has been perceived as a magic bullet, a panacea, and a cure-all for many chronic health conditions such as cancer, cardiovascular disease, diabetes, bone fractures, cognitive decline, and depression. Many of the findings, though, have been from observational studies where a higher blood level of 25-hydroxy vitamin D has been linked to a lower risk for these health conditions.
We know in epidemiology that correlation doesn’t prove causation. Other factors could be involved; for example, people who have higher blood levels of vitamin D may have healthier diets, or they may be spending more time outdoors, being physically active and exposed to the sun. Some of these other factors could be lowering their risk.
When the randomized trials began to emerge, in many of these large-scale trials, the findings were generally neutral or null for cardiovascular disease, total cancer, diabetes, cognitive decline, depression, and many other health outcomes, including fracture. So, the question was asked, does this mean that vitamin D is not important to health?
To the contrary, these findings suggest that vitamin D is so essential to health that we need only small to moderate amounts of vitamin D. Vitamin D is very tightly regulated in the body – the metabolism and function of vitamin D. Even small to moderate amounts will meet the requirements for vitamin D and bone health and many other outcomes.
This is what the National Academy of Medicine, U.S. Preventive Services Task Force, and many other professional organizations have advised, that widespread screening for vitamin D deficiency and blanket universal supplementation with vitamin D would not be indicated.
The randomized trials of vitamin D, including the VITAL study, have generally not shown reductions in the major health outcomes. We found two exceptions in VITAL. We saw promising signals, including a 22% reduction in autoimmune conditions (rheumatoid arthritis and psoriasis) and a 17% reduction in advanced (metastatic or fatal) cancers. In meta-analyses of other large-scale randomized trials, the findings were a signal for a reduction in advanced cancers, even with very small doses of vitamin D (400-800 IUs daily). We tested 2,000 IUs daily in VITAL.
Overall, it’s recommended that small to moderate amounts of vitamin D are adequate, and among the healthy population, most people do not need screening or supplements.
The reduction in autoimmune diseases suggests that vitamin D may play a role in tamping down inflammation. The question has been raised about whether vitamin D is beneficial in reducing the severity of COVID illness, the need for hospitalization, and long COVID. We are looking at this question in a separate trial called VIVID (Vitamin D for COVID Trial) which tests a higher dose (> 3,000 IUs daily) of vitamin D. Those results will be available at the end of this year or early next year.
In other randomized trials of COVID and vitamin D, the results have been mixed and inconsistent, with no clear answer. During the COVID pandemic, I have generally advised that it’s reasonable to take 1,000-2,000 IUs of vitamin D daily as a form of insurance. This dose is known to be very safe. Over 5.3 years in the VITAL trial we saw that a dose of 2,000 IUs was very safe.
But it’s not essential to take a supplement. And overall, aside from some high-risk groups, most people do not need a supplement. The high-risk groups include patients in nursing homes who may have restricted diets and limited time out of doors. For people with malabsorption conditions such as Crohn’s disease, celiac disease, post–gastric bypass surgery, and those with osteoporosis who are on medications for osteoporosis, it’s still quite reasonable to prescribe calcium and vitamin D.
Recommendations for vitamin D in the generally healthy population really should focus on a healthy diet. The United States has a fortified food supply. Vitamin D is added to many foods, dairy products, and cereals, as well as beverages. Natural sources of vitamin D include fatty fish and wild mushrooms.
We should be looking at food labels (which now include vitamin D content) and try to get adequate vitamin D from our diet, and also do our best to spend time outdoors, being physically active, because it is of great benefit to our health. The general principle is that a dietary supplement will never be a substitute for a healthy diet or healthy lifestyle. And those other behaviors really should be the focus at this time.
Dr. Manson is professor of medicine and the Michael and Lee Bell Professor of Women’s Health, Harvard Medical School, and chief of the division of preventive medicine at Brigham and Women’s Hospital, both in Boston. She has received infrastructure support from Mars Symbioscience for the COSMOS trial.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hello. This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. As a director of the Vitamin D and Omega-3 trial (VITAL), the largest randomized clinical trial in the world, I’m often asked, “How much vitamin D do we need, and should I take a vitamin D supplement?” I want to review the findings from recent randomized clinical trials and the implications for practice.
For a long time, vitamin D has been perceived as a magic bullet, a panacea, and a cure-all for many chronic health conditions such as cancer, cardiovascular disease, diabetes, bone fractures, cognitive decline, and depression. Many of the findings, though, have been from observational studies where a higher blood level of 25-hydroxy vitamin D has been linked to a lower risk for these health conditions.
We know in epidemiology that correlation doesn’t prove causation. Other factors could be involved; for example, people who have higher blood levels of vitamin D may have healthier diets, or they may be spending more time outdoors, being physically active and exposed to the sun. Some of these other factors could be lowering their risk.
When the randomized trials began to emerge, in many of these large-scale trials, the findings were generally neutral or null for cardiovascular disease, total cancer, diabetes, cognitive decline, depression, and many other health outcomes, including fracture. So, the question was asked, does this mean that vitamin D is not important to health?
To the contrary, these findings suggest that vitamin D is so essential to health that we need only small to moderate amounts of vitamin D. Vitamin D is very tightly regulated in the body – the metabolism and function of vitamin D. Even small to moderate amounts will meet the requirements for vitamin D and bone health and many other outcomes.
This is what the National Academy of Medicine, U.S. Preventive Services Task Force, and many other professional organizations have advised, that widespread screening for vitamin D deficiency and blanket universal supplementation with vitamin D would not be indicated.
The randomized trials of vitamin D, including the VITAL study, have generally not shown reductions in the major health outcomes. We found two exceptions in VITAL. We saw promising signals, including a 22% reduction in autoimmune conditions (rheumatoid arthritis and psoriasis) and a 17% reduction in advanced (metastatic or fatal) cancers. In meta-analyses of other large-scale randomized trials, the findings were a signal for a reduction in advanced cancers, even with very small doses of vitamin D (400-800 IUs daily). We tested 2,000 IUs daily in VITAL.
Overall, it’s recommended that small to moderate amounts of vitamin D are adequate, and among the healthy population, most people do not need screening or supplements.
The reduction in autoimmune diseases suggests that vitamin D may play a role in tamping down inflammation. The question has been raised about whether vitamin D is beneficial in reducing the severity of COVID illness, the need for hospitalization, and long COVID. We are looking at this question in a separate trial called VIVID (Vitamin D for COVID Trial) which tests a higher dose (> 3,000 IUs daily) of vitamin D. Those results will be available at the end of this year or early next year.
In other randomized trials of COVID and vitamin D, the results have been mixed and inconsistent, with no clear answer. During the COVID pandemic, I have generally advised that it’s reasonable to take 1,000-2,000 IUs of vitamin D daily as a form of insurance. This dose is known to be very safe. Over 5.3 years in the VITAL trial we saw that a dose of 2,000 IUs was very safe.
But it’s not essential to take a supplement. And overall, aside from some high-risk groups, most people do not need a supplement. The high-risk groups include patients in nursing homes who may have restricted diets and limited time out of doors. For people with malabsorption conditions such as Crohn’s disease, celiac disease, post–gastric bypass surgery, and those with osteoporosis who are on medications for osteoporosis, it’s still quite reasonable to prescribe calcium and vitamin D.
Recommendations for vitamin D in the generally healthy population really should focus on a healthy diet. The United States has a fortified food supply. Vitamin D is added to many foods, dairy products, and cereals, as well as beverages. Natural sources of vitamin D include fatty fish and wild mushrooms.
We should be looking at food labels (which now include vitamin D content) and try to get adequate vitamin D from our diet, and also do our best to spend time outdoors, being physically active, because it is of great benefit to our health. The general principle is that a dietary supplement will never be a substitute for a healthy diet or healthy lifestyle. And those other behaviors really should be the focus at this time.
Dr. Manson is professor of medicine and the Michael and Lee Bell Professor of Women’s Health, Harvard Medical School, and chief of the division of preventive medicine at Brigham and Women’s Hospital, both in Boston. She has received infrastructure support from Mars Symbioscience for the COSMOS trial.
A version of this article first appeared on Medscape.com.
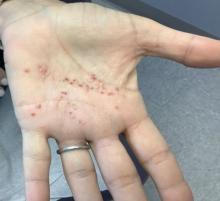
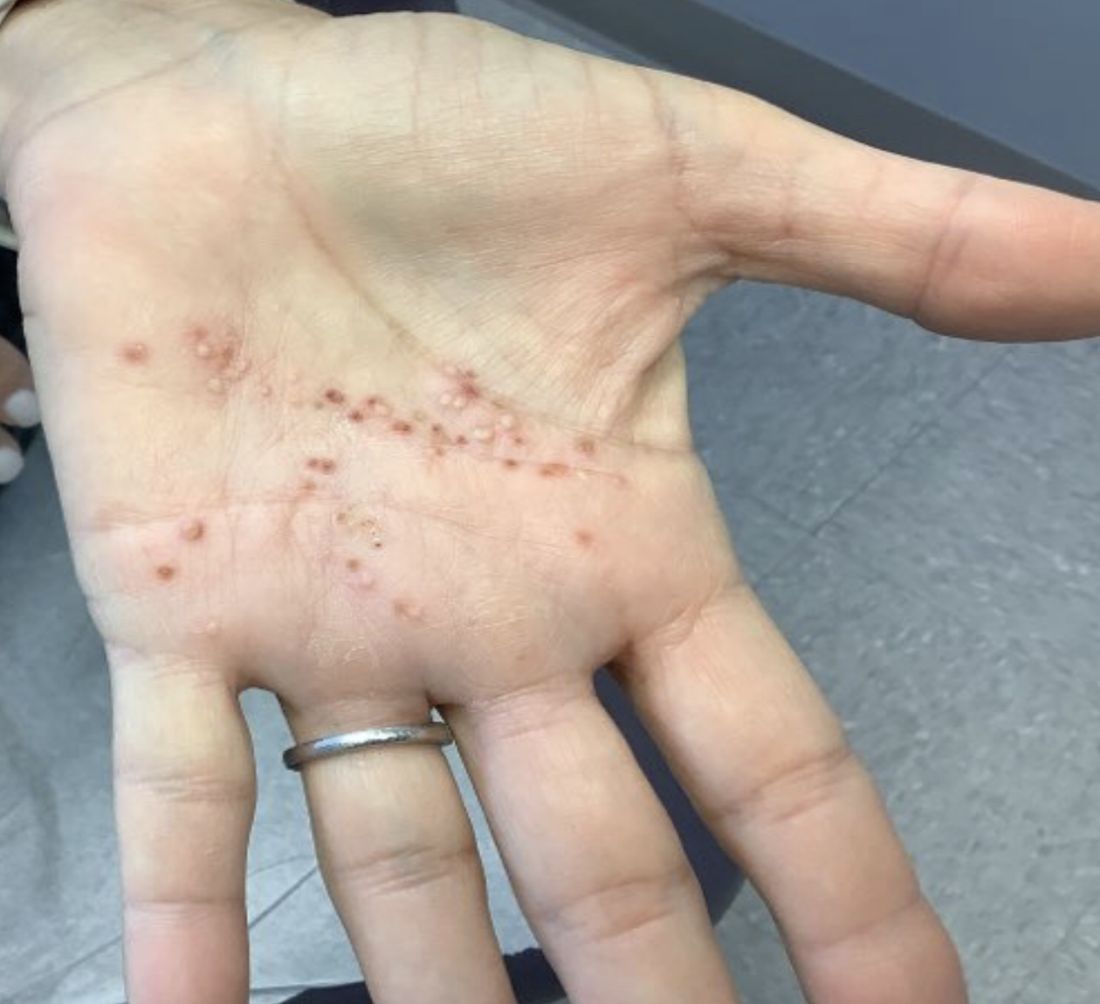
A White female presented with pustules and erythematous macules on the left palm
Psoriasis is an immune-mediated chronic inflammatory disease characterized by well-demarcated, scaly, erythematous plaques. Those who present with the condition often have a family history, which supports recent research uncovering various genes implicated in its pathogenesis. The disease is also associated with other systemic complications, most notably cardiovascular disease.
This condition is found in a small percentage of patients with psoriasis and presentation varies from hyperkeratotic plaques to pustular lesions. The pustular form is known as palmoplantar pustulosis and is within the spectrum of palmoplantar psoriasis.
Psoriasis is typically a clinical diagnosis and its severity can be measured using the Psoriasis Area and Severity Index. If biopsy is performed, the histology demonstrates parakeratosis, orthokeratosis, loss of the stratum granulosum, and dilated vasculature with an inflammatory cell infiltrate. The keratinocytes present with abnormal differentiation and hyperplasia, and the presence of foci of neutrophils known as “Munro’s microabscesses” in the stratum corneum serve as the hallmark of histological diagnosis. However, it is important to note that appearance can vary based on the stage of the lesion and the subtype of psoriasis present.
Palmoplantar psoriasis can be especially limiting and difficult to treat because of its distribution. Topical steroids, topical vitamin D analogues, and narrow band ultraviolet light therapy can be effective for less severe cases. Methotrexate, biologic treatments, and apremilast can be used for more extensive disease.
This patient is HLA-B27 positive and has uveitis. The presence of the HLA-B27 allele has been associated with inflammatory bowel disease, uveitis, psoriatic arthritis, and reactive arthritis. It has also been reported to be associated with pustular psoriasis. She responded well to topical steroids and vitamin D analogues.
This case and photo were submitted by Mr. Shapiro at Nova Southeastern University College of Osteopathic Medicine, Davie, Fla., and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Psoriasis: Overview and Diagnosis, in “Evidence-Based Psoriasis. Updates in Clinical Dermatology.” (Cham, Switzerland: Springer International, 2018).
2. Merola JF et al. Dermatol Ther. 2018 May;31(3):e12589.
3. Chung J et al. J Am Acad Dermatol. 2014 Oct;71(4):623-32.
Psoriasis is an immune-mediated chronic inflammatory disease characterized by well-demarcated, scaly, erythematous plaques. Those who present with the condition often have a family history, which supports recent research uncovering various genes implicated in its pathogenesis. The disease is also associated with other systemic complications, most notably cardiovascular disease.
This condition is found in a small percentage of patients with psoriasis and presentation varies from hyperkeratotic plaques to pustular lesions. The pustular form is known as palmoplantar pustulosis and is within the spectrum of palmoplantar psoriasis.
Psoriasis is typically a clinical diagnosis and its severity can be measured using the Psoriasis Area and Severity Index. If biopsy is performed, the histology demonstrates parakeratosis, orthokeratosis, loss of the stratum granulosum, and dilated vasculature with an inflammatory cell infiltrate. The keratinocytes present with abnormal differentiation and hyperplasia, and the presence of foci of neutrophils known as “Munro’s microabscesses” in the stratum corneum serve as the hallmark of histological diagnosis. However, it is important to note that appearance can vary based on the stage of the lesion and the subtype of psoriasis present.
Palmoplantar psoriasis can be especially limiting and difficult to treat because of its distribution. Topical steroids, topical vitamin D analogues, and narrow band ultraviolet light therapy can be effective for less severe cases. Methotrexate, biologic treatments, and apremilast can be used for more extensive disease.
This patient is HLA-B27 positive and has uveitis. The presence of the HLA-B27 allele has been associated with inflammatory bowel disease, uveitis, psoriatic arthritis, and reactive arthritis. It has also been reported to be associated with pustular psoriasis. She responded well to topical steroids and vitamin D analogues.
This case and photo were submitted by Mr. Shapiro at Nova Southeastern University College of Osteopathic Medicine, Davie, Fla., and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Psoriasis: Overview and Diagnosis, in “Evidence-Based Psoriasis. Updates in Clinical Dermatology.” (Cham, Switzerland: Springer International, 2018).
2. Merola JF et al. Dermatol Ther. 2018 May;31(3):e12589.
3. Chung J et al. J Am Acad Dermatol. 2014 Oct;71(4):623-32.
Psoriasis is an immune-mediated chronic inflammatory disease characterized by well-demarcated, scaly, erythematous plaques. Those who present with the condition often have a family history, which supports recent research uncovering various genes implicated in its pathogenesis. The disease is also associated with other systemic complications, most notably cardiovascular disease.
This condition is found in a small percentage of patients with psoriasis and presentation varies from hyperkeratotic plaques to pustular lesions. The pustular form is known as palmoplantar pustulosis and is within the spectrum of palmoplantar psoriasis.
Psoriasis is typically a clinical diagnosis and its severity can be measured using the Psoriasis Area and Severity Index. If biopsy is performed, the histology demonstrates parakeratosis, orthokeratosis, loss of the stratum granulosum, and dilated vasculature with an inflammatory cell infiltrate. The keratinocytes present with abnormal differentiation and hyperplasia, and the presence of foci of neutrophils known as “Munro’s microabscesses” in the stratum corneum serve as the hallmark of histological diagnosis. However, it is important to note that appearance can vary based on the stage of the lesion and the subtype of psoriasis present.
Palmoplantar psoriasis can be especially limiting and difficult to treat because of its distribution. Topical steroids, topical vitamin D analogues, and narrow band ultraviolet light therapy can be effective for less severe cases. Methotrexate, biologic treatments, and apremilast can be used for more extensive disease.
This patient is HLA-B27 positive and has uveitis. The presence of the HLA-B27 allele has been associated with inflammatory bowel disease, uveitis, psoriatic arthritis, and reactive arthritis. It has also been reported to be associated with pustular psoriasis. She responded well to topical steroids and vitamin D analogues.
This case and photo were submitted by Mr. Shapiro at Nova Southeastern University College of Osteopathic Medicine, Davie, Fla., and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Psoriasis: Overview and Diagnosis, in “Evidence-Based Psoriasis. Updates in Clinical Dermatology.” (Cham, Switzerland: Springer International, 2018).
2. Merola JF et al. Dermatol Ther. 2018 May;31(3):e12589.
3. Chung J et al. J Am Acad Dermatol. 2014 Oct;71(4):623-32.
Loan forgiveness and med school debt: What about me?
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I run the division of medical ethics at New York University Grossman School of Medicine.
Many of you know that President Biden created a loan forgiveness program, forgiving up to $10,000 against federal student loans, including graduate and undergraduate education. The Department of Education is supposed to provide up to $20,000 in debt cancellation to Pell Grant recipients who have loans that are held by the Department of Education. Borrowers can get this relief if their income is less than $125,000 for an individual or $250,000 for married couples.
Many people have looked at this and said, “Hey, wait a minute. I paid off my loans. I didn’t get any reimbursement. That isn’t fair.”
who often still have huge amounts of debt, and either because of the income limits or because they don’t qualify because this debt was accrued long in the past, they’re saying, “What about me? Don’t you want to give any relief to me?”
This is a topic near and dear to my heart because I happen to be at a medical school, NYU, that has decided for the two medical schools it runs – our main campus, NYU in Manhattan and NYU Langone out on Long Island – that we’re going to go tuition free. We’ve done it for a couple of years.
We did it because I think all the administrators and faculty understood the tremendous burden that debt poses on people who both carry forward their undergraduate debt and then have medical school debt. This really leads to very difficult situations – which we have great empathy for – about what specialty you’re going to go into, whether you have to moonlight, and how you’re going to manage a huge burden of debt.
Many people don’t have sympathy out in the public. They say doctors make a large amount of money and they live a nice lifestyle, so we’re not going to relieve their debt. The reality is that, whoever you are, short of Bill Gates or Elon Musk, having hundreds of thousands of dollars of debt is no easy task to live with and to work off.
Still, when we created free tuition at NYU for our medical school, there were many people who paid high tuition fees in the past. Some of them said to us, “What about me?” We decided not to try to do anything retrospectively. The plan was to build up enough money so that we could handle no-cost tuition going forward. We didn’t really have it in our pocketbook to help people who’d already paid their debts or were saddled with NYU debt. Is it fair? No, it’s probably not fair, but it’s an improvement.
That’s what I want people to think about who are saying, “What about my medical school debt? What about my undergraduate plus medical school debt?” I think we should be grateful when efforts are being made to reduce very burdensome student loans that people have. It’s good to give that benefit and move it forward.
Does that mean no one should get anything unless everyone with any kind of debt from school is covered? I don’t think so. I don’t think that’s fair either.
It is possible that we could continue to agitate politically and say, let’s go after some of the health care debt. Let’s go after some of the things that are still driving people to have to work more than they would or to choose specialties that they really don’t want to be in because they have to make up that debt.
It doesn’t mean the last word has been said about the politics of debt relief or, for that matter, the price of going to medical school in the first place and trying to see whether that can be driven down.
I don’t think it’s right to say, “If I can’t benefit, given the huge burden that I’m carrying, then I’m not going to try to give relief to others.” I think we’re relieving debt to the extent that we can do it. The nation can afford it. Going forward is a good thing. It’s wrong to create those gigantic debts in the first place.
What are we going to do about the past? We may decide that we need some sort of forgiveness or reparations for loans that were built up for others going backwards. I wouldn’t hold hostage the future and our children to what was probably a very poor, unethical practice about saddling doctors and others in the past with huge debt.
I’m Art Caplan at the division of medical ethics at New York University Grossman School of Medicine. Thank you for watching.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I run the division of medical ethics at New York University Grossman School of Medicine.
Many of you know that President Biden created a loan forgiveness program, forgiving up to $10,000 against federal student loans, including graduate and undergraduate education. The Department of Education is supposed to provide up to $20,000 in debt cancellation to Pell Grant recipients who have loans that are held by the Department of Education. Borrowers can get this relief if their income is less than $125,000 for an individual or $250,000 for married couples.
Many people have looked at this and said, “Hey, wait a minute. I paid off my loans. I didn’t get any reimbursement. That isn’t fair.”
who often still have huge amounts of debt, and either because of the income limits or because they don’t qualify because this debt was accrued long in the past, they’re saying, “What about me? Don’t you want to give any relief to me?”
This is a topic near and dear to my heart because I happen to be at a medical school, NYU, that has decided for the two medical schools it runs – our main campus, NYU in Manhattan and NYU Langone out on Long Island – that we’re going to go tuition free. We’ve done it for a couple of years.
We did it because I think all the administrators and faculty understood the tremendous burden that debt poses on people who both carry forward their undergraduate debt and then have medical school debt. This really leads to very difficult situations – which we have great empathy for – about what specialty you’re going to go into, whether you have to moonlight, and how you’re going to manage a huge burden of debt.
Many people don’t have sympathy out in the public. They say doctors make a large amount of money and they live a nice lifestyle, so we’re not going to relieve their debt. The reality is that, whoever you are, short of Bill Gates or Elon Musk, having hundreds of thousands of dollars of debt is no easy task to live with and to work off.
Still, when we created free tuition at NYU for our medical school, there were many people who paid high tuition fees in the past. Some of them said to us, “What about me?” We decided not to try to do anything retrospectively. The plan was to build up enough money so that we could handle no-cost tuition going forward. We didn’t really have it in our pocketbook to help people who’d already paid their debts or were saddled with NYU debt. Is it fair? No, it’s probably not fair, but it’s an improvement.
That’s what I want people to think about who are saying, “What about my medical school debt? What about my undergraduate plus medical school debt?” I think we should be grateful when efforts are being made to reduce very burdensome student loans that people have. It’s good to give that benefit and move it forward.
Does that mean no one should get anything unless everyone with any kind of debt from school is covered? I don’t think so. I don’t think that’s fair either.
It is possible that we could continue to agitate politically and say, let’s go after some of the health care debt. Let’s go after some of the things that are still driving people to have to work more than they would or to choose specialties that they really don’t want to be in because they have to make up that debt.
It doesn’t mean the last word has been said about the politics of debt relief or, for that matter, the price of going to medical school in the first place and trying to see whether that can be driven down.
I don’t think it’s right to say, “If I can’t benefit, given the huge burden that I’m carrying, then I’m not going to try to give relief to others.” I think we’re relieving debt to the extent that we can do it. The nation can afford it. Going forward is a good thing. It’s wrong to create those gigantic debts in the first place.
What are we going to do about the past? We may decide that we need some sort of forgiveness or reparations for loans that were built up for others going backwards. I wouldn’t hold hostage the future and our children to what was probably a very poor, unethical practice about saddling doctors and others in the past with huge debt.
I’m Art Caplan at the division of medical ethics at New York University Grossman School of Medicine. Thank you for watching.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I run the division of medical ethics at New York University Grossman School of Medicine.
Many of you know that President Biden created a loan forgiveness program, forgiving up to $10,000 against federal student loans, including graduate and undergraduate education. The Department of Education is supposed to provide up to $20,000 in debt cancellation to Pell Grant recipients who have loans that are held by the Department of Education. Borrowers can get this relief if their income is less than $125,000 for an individual or $250,000 for married couples.
Many people have looked at this and said, “Hey, wait a minute. I paid off my loans. I didn’t get any reimbursement. That isn’t fair.”
who often still have huge amounts of debt, and either because of the income limits or because they don’t qualify because this debt was accrued long in the past, they’re saying, “What about me? Don’t you want to give any relief to me?”
This is a topic near and dear to my heart because I happen to be at a medical school, NYU, that has decided for the two medical schools it runs – our main campus, NYU in Manhattan and NYU Langone out on Long Island – that we’re going to go tuition free. We’ve done it for a couple of years.
We did it because I think all the administrators and faculty understood the tremendous burden that debt poses on people who both carry forward their undergraduate debt and then have medical school debt. This really leads to very difficult situations – which we have great empathy for – about what specialty you’re going to go into, whether you have to moonlight, and how you’re going to manage a huge burden of debt.
Many people don’t have sympathy out in the public. They say doctors make a large amount of money and they live a nice lifestyle, so we’re not going to relieve their debt. The reality is that, whoever you are, short of Bill Gates or Elon Musk, having hundreds of thousands of dollars of debt is no easy task to live with and to work off.
Still, when we created free tuition at NYU for our medical school, there were many people who paid high tuition fees in the past. Some of them said to us, “What about me?” We decided not to try to do anything retrospectively. The plan was to build up enough money so that we could handle no-cost tuition going forward. We didn’t really have it in our pocketbook to help people who’d already paid their debts or were saddled with NYU debt. Is it fair? No, it’s probably not fair, but it’s an improvement.
That’s what I want people to think about who are saying, “What about my medical school debt? What about my undergraduate plus medical school debt?” I think we should be grateful when efforts are being made to reduce very burdensome student loans that people have. It’s good to give that benefit and move it forward.
Does that mean no one should get anything unless everyone with any kind of debt from school is covered? I don’t think so. I don’t think that’s fair either.
It is possible that we could continue to agitate politically and say, let’s go after some of the health care debt. Let’s go after some of the things that are still driving people to have to work more than they would or to choose specialties that they really don’t want to be in because they have to make up that debt.
It doesn’t mean the last word has been said about the politics of debt relief or, for that matter, the price of going to medical school in the first place and trying to see whether that can be driven down.
I don’t think it’s right to say, “If I can’t benefit, given the huge burden that I’m carrying, then I’m not going to try to give relief to others.” I think we’re relieving debt to the extent that we can do it. The nation can afford it. Going forward is a good thing. It’s wrong to create those gigantic debts in the first place.
What are we going to do about the past? We may decide that we need some sort of forgiveness or reparations for loans that were built up for others going backwards. I wouldn’t hold hostage the future and our children to what was probably a very poor, unethical practice about saddling doctors and others in the past with huge debt.
I’m Art Caplan at the division of medical ethics at New York University Grossman School of Medicine. Thank you for watching.
A version of this article first appeared on Medscape.com.
The marked contrast in pandemic outcomes between Japan and the United States
This article was originally published Oct. 8 on Medscape Editor-In-Chief Eric Topol’s “Ground Truths” column on Substack.
Over time it has the least cumulative deaths per capita of any major country in the world. That’s without a zero-Covid policy or any national lockdowns, which is why I have not included China as a comparator.
Before we get into that data, let’s take a look at the age pyramids for Japan and the United States. The No. 1 risk factor for death from COVID-19 is advanced age, and you can see that in Japan about 25% of the population is age 65 and older, whereas in the United States that proportion is substantially reduced at 15%. Sure there are differences in comorbidities such as obesity and diabetes, but there is also the trade-off of a much higher population density in Japan.
Besides masks, which were distributed early on by the government to the population in Japan, there was the “Avoid the 3Cs” cluster-busting strategy, widely disseminated in the spring of 2020, leveraging Pareto’s 80-20 principle, long before there were any vaccines available. For a good portion of the pandemic, the Ministry of Foreign Affairs of Japan maintained a strict policy for border control, which while hard to quantify, may certainly have contributed to its success.
Besides these factors, once vaccines became available, Japan got the population with the primary series to 83% rapidly, even after getting a late start by many months compared with the United States, which has peaked at 68%. That’s a big gap.
But that gap got much worse when it came to boosters. Ninety-five percent of Japanese eligible compared with 40.8% of Americans have had a booster shot. Of note, that 95% in Japan pertains to the whole population. In the United States the percentage of people age 65 and older who have had two boosters is currently only 42%. I’ve previously reviewed the important lifesaving impact of two boosters among people age 65 and older from five independent studies during Omicron waves throughout the world.
Now let’s turn to cumulative fatalities in the two countries. There’s a huge, nearly ninefold difference, per capita. Using today’s Covid-19 Dashboard, there are cumulatively 45,533 deaths in Japan and 1,062,560 American deaths. That translates to 1 in 2,758 people in Japan compared with 1 in 315 Americans dying of COVID.
And if we look at excess mortality instead of confirmed COVID deaths, that enormous gap doesn’t change.
Obviously it would be good to have data for other COVID outcomes, such as hospitalizations, ICUs, and Long COVID, but they are not accessible.
Comparing Japan, the country that has fared the best, with the United States, one of the worst pandemic outcome results, leaves us with a sense that Prof Ian MacKay’s “Swiss cheese model” is the best explanation. It’s not just one thing. Masks, consistent evidence-based communication (3Cs) with attention to ventilation and air quality, and the outstanding uptake of vaccines and boosters all contributed to Japan’s success.
There is another factor to add to that model – Paxlovid. Its benefit of reducing hospitalizations and deaths for people over age 65 is unquestionable.
That’s why I had previously modified the Swiss cheese model to add Paxlovid.
But in the United States, where 15% of the population is 65 and older, they account for over 75% of the daily death toll, still in the range of 400 per day. Here, with a very high proportion of people age 65 and older left vulnerable without boosters, or primary vaccines, Paxlovid is only being given to less than 25% of the eligible (age 50+), and less people age 80 and older are getting Paxlovid than those age 45. The reasons that doctors are not prescribing it – worried about interactions for a 5-day course and rebound – are not substantiated.
Bottom line: In the United States we are not protecting our population anywhere near as well as Japan, as grossly evident by the fatalities among people at the highest risk. There needs to be far better uptake of boosters and use of Paxlovid in the age 65+ group, but the need for amped up protection is not at all restricted to this age subgroup. Across all age groups age 18 and over there is an 81% reduction of hospitalizations with two boosters with the most updated CDC data available, through the Omicron BA.5 wave.
No less the previous data through May 2022 showing protection from death across all ages with two boosters
And please don’t forget that around the world, over 20 million lives were saved, just in 2021, the first year of vaccines.
We can learn so much from a model country like Japan. Yes, we need nasal and variant-proof vaccines to effectively deal with the new variants that are already getting legs in places like XBB in Singapore and ones not on the radar yet. But right now we’ve got to do far better for people getting boosters and, when a person age 65 or older gets COVID, Paxlovid. Take a look at the Chris Hayes video segment when he pleaded for Americans to get a booster shot. Every day that vaccine waning of the U.S. population exceeds the small percentage of people who get a booster, our vulnerability increases. If we don’t get that on track, it’s likely going to be a rough winter ahead.
Dr. Topol is director of the Scripps Translational Science Institute in La Jolla, Calif. He has received research grants from the National Institutes of Health and reported conflicts of interest involving Dexcom, Illumina, Molecular Stethoscope, Quest Diagnostics, and Blue Cross Blue Shield Association. A version of this article appeared on Medscape.com.
This article was originally published Oct. 8 on Medscape Editor-In-Chief Eric Topol’s “Ground Truths” column on Substack.
Over time it has the least cumulative deaths per capita of any major country in the world. That’s without a zero-Covid policy or any national lockdowns, which is why I have not included China as a comparator.
Before we get into that data, let’s take a look at the age pyramids for Japan and the United States. The No. 1 risk factor for death from COVID-19 is advanced age, and you can see that in Japan about 25% of the population is age 65 and older, whereas in the United States that proportion is substantially reduced at 15%. Sure there are differences in comorbidities such as obesity and diabetes, but there is also the trade-off of a much higher population density in Japan.
Besides masks, which were distributed early on by the government to the population in Japan, there was the “Avoid the 3Cs” cluster-busting strategy, widely disseminated in the spring of 2020, leveraging Pareto’s 80-20 principle, long before there were any vaccines available. For a good portion of the pandemic, the Ministry of Foreign Affairs of Japan maintained a strict policy for border control, which while hard to quantify, may certainly have contributed to its success.
Besides these factors, once vaccines became available, Japan got the population with the primary series to 83% rapidly, even after getting a late start by many months compared with the United States, which has peaked at 68%. That’s a big gap.
But that gap got much worse when it came to boosters. Ninety-five percent of Japanese eligible compared with 40.8% of Americans have had a booster shot. Of note, that 95% in Japan pertains to the whole population. In the United States the percentage of people age 65 and older who have had two boosters is currently only 42%. I’ve previously reviewed the important lifesaving impact of two boosters among people age 65 and older from five independent studies during Omicron waves throughout the world.
Now let’s turn to cumulative fatalities in the two countries. There’s a huge, nearly ninefold difference, per capita. Using today’s Covid-19 Dashboard, there are cumulatively 45,533 deaths in Japan and 1,062,560 American deaths. That translates to 1 in 2,758 people in Japan compared with 1 in 315 Americans dying of COVID.
And if we look at excess mortality instead of confirmed COVID deaths, that enormous gap doesn’t change.
Obviously it would be good to have data for other COVID outcomes, such as hospitalizations, ICUs, and Long COVID, but they are not accessible.
Comparing Japan, the country that has fared the best, with the United States, one of the worst pandemic outcome results, leaves us with a sense that Prof Ian MacKay’s “Swiss cheese model” is the best explanation. It’s not just one thing. Masks, consistent evidence-based communication (3Cs) with attention to ventilation and air quality, and the outstanding uptake of vaccines and boosters all contributed to Japan’s success.
There is another factor to add to that model – Paxlovid. Its benefit of reducing hospitalizations and deaths for people over age 65 is unquestionable.
That’s why I had previously modified the Swiss cheese model to add Paxlovid.
But in the United States, where 15% of the population is 65 and older, they account for over 75% of the daily death toll, still in the range of 400 per day. Here, with a very high proportion of people age 65 and older left vulnerable without boosters, or primary vaccines, Paxlovid is only being given to less than 25% of the eligible (age 50+), and less people age 80 and older are getting Paxlovid than those age 45. The reasons that doctors are not prescribing it – worried about interactions for a 5-day course and rebound – are not substantiated.
Bottom line: In the United States we are not protecting our population anywhere near as well as Japan, as grossly evident by the fatalities among people at the highest risk. There needs to be far better uptake of boosters and use of Paxlovid in the age 65+ group, but the need for amped up protection is not at all restricted to this age subgroup. Across all age groups age 18 and over there is an 81% reduction of hospitalizations with two boosters with the most updated CDC data available, through the Omicron BA.5 wave.
No less the previous data through May 2022 showing protection from death across all ages with two boosters
And please don’t forget that around the world, over 20 million lives were saved, just in 2021, the first year of vaccines.
We can learn so much from a model country like Japan. Yes, we need nasal and variant-proof vaccines to effectively deal with the new variants that are already getting legs in places like XBB in Singapore and ones not on the radar yet. But right now we’ve got to do far better for people getting boosters and, when a person age 65 or older gets COVID, Paxlovid. Take a look at the Chris Hayes video segment when he pleaded for Americans to get a booster shot. Every day that vaccine waning of the U.S. population exceeds the small percentage of people who get a booster, our vulnerability increases. If we don’t get that on track, it’s likely going to be a rough winter ahead.
Dr. Topol is director of the Scripps Translational Science Institute in La Jolla, Calif. He has received research grants from the National Institutes of Health and reported conflicts of interest involving Dexcom, Illumina, Molecular Stethoscope, Quest Diagnostics, and Blue Cross Blue Shield Association. A version of this article appeared on Medscape.com.
This article was originally published Oct. 8 on Medscape Editor-In-Chief Eric Topol’s “Ground Truths” column on Substack.
Over time it has the least cumulative deaths per capita of any major country in the world. That’s without a zero-Covid policy or any national lockdowns, which is why I have not included China as a comparator.
Before we get into that data, let’s take a look at the age pyramids for Japan and the United States. The No. 1 risk factor for death from COVID-19 is advanced age, and you can see that in Japan about 25% of the population is age 65 and older, whereas in the United States that proportion is substantially reduced at 15%. Sure there are differences in comorbidities such as obesity and diabetes, but there is also the trade-off of a much higher population density in Japan.
Besides masks, which were distributed early on by the government to the population in Japan, there was the “Avoid the 3Cs” cluster-busting strategy, widely disseminated in the spring of 2020, leveraging Pareto’s 80-20 principle, long before there were any vaccines available. For a good portion of the pandemic, the Ministry of Foreign Affairs of Japan maintained a strict policy for border control, which while hard to quantify, may certainly have contributed to its success.
Besides these factors, once vaccines became available, Japan got the population with the primary series to 83% rapidly, even after getting a late start by many months compared with the United States, which has peaked at 68%. That’s a big gap.
But that gap got much worse when it came to boosters. Ninety-five percent of Japanese eligible compared with 40.8% of Americans have had a booster shot. Of note, that 95% in Japan pertains to the whole population. In the United States the percentage of people age 65 and older who have had two boosters is currently only 42%. I’ve previously reviewed the important lifesaving impact of two boosters among people age 65 and older from five independent studies during Omicron waves throughout the world.
Now let’s turn to cumulative fatalities in the two countries. There’s a huge, nearly ninefold difference, per capita. Using today’s Covid-19 Dashboard, there are cumulatively 45,533 deaths in Japan and 1,062,560 American deaths. That translates to 1 in 2,758 people in Japan compared with 1 in 315 Americans dying of COVID.
And if we look at excess mortality instead of confirmed COVID deaths, that enormous gap doesn’t change.
Obviously it would be good to have data for other COVID outcomes, such as hospitalizations, ICUs, and Long COVID, but they are not accessible.
Comparing Japan, the country that has fared the best, with the United States, one of the worst pandemic outcome results, leaves us with a sense that Prof Ian MacKay’s “Swiss cheese model” is the best explanation. It’s not just one thing. Masks, consistent evidence-based communication (3Cs) with attention to ventilation and air quality, and the outstanding uptake of vaccines and boosters all contributed to Japan’s success.
There is another factor to add to that model – Paxlovid. Its benefit of reducing hospitalizations and deaths for people over age 65 is unquestionable.
That’s why I had previously modified the Swiss cheese model to add Paxlovid.
But in the United States, where 15% of the population is 65 and older, they account for over 75% of the daily death toll, still in the range of 400 per day. Here, with a very high proportion of people age 65 and older left vulnerable without boosters, or primary vaccines, Paxlovid is only being given to less than 25% of the eligible (age 50+), and less people age 80 and older are getting Paxlovid than those age 45. The reasons that doctors are not prescribing it – worried about interactions for a 5-day course and rebound – are not substantiated.
Bottom line: In the United States we are not protecting our population anywhere near as well as Japan, as grossly evident by the fatalities among people at the highest risk. There needs to be far better uptake of boosters and use of Paxlovid in the age 65+ group, but the need for amped up protection is not at all restricted to this age subgroup. Across all age groups age 18 and over there is an 81% reduction of hospitalizations with two boosters with the most updated CDC data available, through the Omicron BA.5 wave.
No less the previous data through May 2022 showing protection from death across all ages with two boosters
And please don’t forget that around the world, over 20 million lives were saved, just in 2021, the first year of vaccines.
We can learn so much from a model country like Japan. Yes, we need nasal and variant-proof vaccines to effectively deal with the new variants that are already getting legs in places like XBB in Singapore and ones not on the radar yet. But right now we’ve got to do far better for people getting boosters and, when a person age 65 or older gets COVID, Paxlovid. Take a look at the Chris Hayes video segment when he pleaded for Americans to get a booster shot. Every day that vaccine waning of the U.S. population exceeds the small percentage of people who get a booster, our vulnerability increases. If we don’t get that on track, it’s likely going to be a rough winter ahead.
Dr. Topol is director of the Scripps Translational Science Institute in La Jolla, Calif. He has received research grants from the National Institutes of Health and reported conflicts of interest involving Dexcom, Illumina, Molecular Stethoscope, Quest Diagnostics, and Blue Cross Blue Shield Association. A version of this article appeared on Medscape.com.
Playing the fat shame game in medicine: It needs to stop
I will remember that there is art to medicine as well as science, and that warmth, sympathy, and understanding may outweigh the surgeon’s knife or the chemist’s drug.
Upon finishing medical school, many of us recited this passage from a modernized version of the Hippocratic Oath. Though there has been controversy regarding the current relevancy of this oath, it can still serve as a reminder of the promises we made on behalf of our patients: To treat them ethically, with empathy and respect, and without pretension. Though I hadn’t thought about the Hippocratic Oath in ages, it came to mind recently after I read an article about weight trends in adults during the COVID pandemic.
No surprise – we gained weight during the initial surge at a rate of roughly a pound and a half per month following the initial shelter-in-place period. For some of us, that trend in weight gain worsened as the pandemic persisted. A survey conducted in February 2021 suggested that over 40% of adults who experienced undesired weight changes since the start of the pandemic gained an average of 29 pounds (significantly more than the typical gain of 15 pounds, often referred to as the “Quarantine 15” or “COVID-15”).
Updated data, obtained via a review of electronic health records for over 15 million patients, shows that 39% of patients gained weight during the pandemic (10% of them gained more than 12.5 pounds, while 2% gained over 27.5 pounds). Though these recent numbers may be lower than previously reported, they still aren’t reassuring.
Research has already confirmed that sizeism has a negative impact on both a patient’s physical health and psychological well-being, and as medical providers, we’re part of the problem. We cause distress in our patients through disrespectful treatment and medical fat shaming, which can lead to cycles of disordered eating, reduced physical activity, and more weight gain. We discriminate based on weight, causing our patients to delay health care visits and other provider interactions, resulting in increased risks for morbidity and even mortality. We make assumptions that a patient’s presenting complaints are due to weight rather than other causes, resulting in missed diagnoses. And we recommend different treatments for obese patients with the same condition as nonobese patients simply because of their weight.
One study has suggested that over 40% of adults in the United States have suffered from weight stigma, and physicians and coworkers are listed as some of the most common sources. Another study suggests that nearly 70% of overweight or obese patients report feeling stigmatized by physicians, whether through expressed biases or purposeful avoidance (patients have previously reported that their providers addressed weight loss in fewer than 20% of their examinations).
As health care providers, we need to do better. We should all be willing to consider our own biases about body size, and there are self-assessments to help with this, including the Implicit Associations Test: Weight Bias. By becoming more self-aware, hopefully we can change the doctor-patient conversation about weight management.
Studies have shown that meaningful conversations with physicians can have a significant impact on patients’ attempts to change behaviors related to weight. Yet, many medical providers are not trained in how to counsel patients on nutrition, weight loss, and physical activity (if we bring it up at all). We need to better educate ourselves about weight science and treatments.
In the meantime, we can work on how we interact with our patients:
- Make sure that your practice space is accommodating and nondiscriminatory, with appropriately sized furniture in the waiting and exam rooms, large blood pressure cuffs and gowns, and size-inclusive reading materials.
- Ensure that your workplace has an antiharassment policy that includes sizeism.
- Be an ally and speak up against weight discrimination.
- Educate your office staff about weight stigma and ensure that they avoid commenting on the weight or body size of others (being recognized only for losing weight isn’t a compliment, and sharing “fat jokes” isn’t funny).
- Remember that a person’s body size tells you nothing about that person’s health behaviors. Stop assuming that larger body sizes are related to laziness, overeating, or a lack of motivation.
- Ask your overweight or obese patients if they are willing to talk about their weight before jumping into the topic.
- Practice (patients are more likely to report changing their exercise routine and attempting to lose weight with these techniques).
- Be mindful of your word choices; for example, it can be more helpful to focus on comorbidities (such as high blood pressure or prediabetes) rather than body weight, nutrition rather than dieting, and physical activity rather than specific exercises.
Regardless of how you feel about reciting the Hippocratic Oath, our patients, no matter their body size, deserve to be treated with respect and dignity, as others have said in more eloquent ways than I. Let’s stop playing the fat shame game and help fight weight bias in medicine.
Dr. Devlin is president, Locum Infectious Disease Services, and an independent contractor for Weatherby Healthcare. She reported no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
I will remember that there is art to medicine as well as science, and that warmth, sympathy, and understanding may outweigh the surgeon’s knife or the chemist’s drug.
Upon finishing medical school, many of us recited this passage from a modernized version of the Hippocratic Oath. Though there has been controversy regarding the current relevancy of this oath, it can still serve as a reminder of the promises we made on behalf of our patients: To treat them ethically, with empathy and respect, and without pretension. Though I hadn’t thought about the Hippocratic Oath in ages, it came to mind recently after I read an article about weight trends in adults during the COVID pandemic.
No surprise – we gained weight during the initial surge at a rate of roughly a pound and a half per month following the initial shelter-in-place period. For some of us, that trend in weight gain worsened as the pandemic persisted. A survey conducted in February 2021 suggested that over 40% of adults who experienced undesired weight changes since the start of the pandemic gained an average of 29 pounds (significantly more than the typical gain of 15 pounds, often referred to as the “Quarantine 15” or “COVID-15”).
Updated data, obtained via a review of electronic health records for over 15 million patients, shows that 39% of patients gained weight during the pandemic (10% of them gained more than 12.5 pounds, while 2% gained over 27.5 pounds). Though these recent numbers may be lower than previously reported, they still aren’t reassuring.
Research has already confirmed that sizeism has a negative impact on both a patient’s physical health and psychological well-being, and as medical providers, we’re part of the problem. We cause distress in our patients through disrespectful treatment and medical fat shaming, which can lead to cycles of disordered eating, reduced physical activity, and more weight gain. We discriminate based on weight, causing our patients to delay health care visits and other provider interactions, resulting in increased risks for morbidity and even mortality. We make assumptions that a patient’s presenting complaints are due to weight rather than other causes, resulting in missed diagnoses. And we recommend different treatments for obese patients with the same condition as nonobese patients simply because of their weight.
One study has suggested that over 40% of adults in the United States have suffered from weight stigma, and physicians and coworkers are listed as some of the most common sources. Another study suggests that nearly 70% of overweight or obese patients report feeling stigmatized by physicians, whether through expressed biases or purposeful avoidance (patients have previously reported that their providers addressed weight loss in fewer than 20% of their examinations).
As health care providers, we need to do better. We should all be willing to consider our own biases about body size, and there are self-assessments to help with this, including the Implicit Associations Test: Weight Bias. By becoming more self-aware, hopefully we can change the doctor-patient conversation about weight management.
Studies have shown that meaningful conversations with physicians can have a significant impact on patients’ attempts to change behaviors related to weight. Yet, many medical providers are not trained in how to counsel patients on nutrition, weight loss, and physical activity (if we bring it up at all). We need to better educate ourselves about weight science and treatments.
In the meantime, we can work on how we interact with our patients:
- Make sure that your practice space is accommodating and nondiscriminatory, with appropriately sized furniture in the waiting and exam rooms, large blood pressure cuffs and gowns, and size-inclusive reading materials.
- Ensure that your workplace has an antiharassment policy that includes sizeism.
- Be an ally and speak up against weight discrimination.
- Educate your office staff about weight stigma and ensure that they avoid commenting on the weight or body size of others (being recognized only for losing weight isn’t a compliment, and sharing “fat jokes” isn’t funny).
- Remember that a person’s body size tells you nothing about that person’s health behaviors. Stop assuming that larger body sizes are related to laziness, overeating, or a lack of motivation.
- Ask your overweight or obese patients if they are willing to talk about their weight before jumping into the topic.
- Practice (patients are more likely to report changing their exercise routine and attempting to lose weight with these techniques).
- Be mindful of your word choices; for example, it can be more helpful to focus on comorbidities (such as high blood pressure or prediabetes) rather than body weight, nutrition rather than dieting, and physical activity rather than specific exercises.
Regardless of how you feel about reciting the Hippocratic Oath, our patients, no matter their body size, deserve to be treated with respect and dignity, as others have said in more eloquent ways than I. Let’s stop playing the fat shame game and help fight weight bias in medicine.
Dr. Devlin is president, Locum Infectious Disease Services, and an independent contractor for Weatherby Healthcare. She reported no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
I will remember that there is art to medicine as well as science, and that warmth, sympathy, and understanding may outweigh the surgeon’s knife or the chemist’s drug.
Upon finishing medical school, many of us recited this passage from a modernized version of the Hippocratic Oath. Though there has been controversy regarding the current relevancy of this oath, it can still serve as a reminder of the promises we made on behalf of our patients: To treat them ethically, with empathy and respect, and without pretension. Though I hadn’t thought about the Hippocratic Oath in ages, it came to mind recently after I read an article about weight trends in adults during the COVID pandemic.
No surprise – we gained weight during the initial surge at a rate of roughly a pound and a half per month following the initial shelter-in-place period. For some of us, that trend in weight gain worsened as the pandemic persisted. A survey conducted in February 2021 suggested that over 40% of adults who experienced undesired weight changes since the start of the pandemic gained an average of 29 pounds (significantly more than the typical gain of 15 pounds, often referred to as the “Quarantine 15” or “COVID-15”).
Updated data, obtained via a review of electronic health records for over 15 million patients, shows that 39% of patients gained weight during the pandemic (10% of them gained more than 12.5 pounds, while 2% gained over 27.5 pounds). Though these recent numbers may be lower than previously reported, they still aren’t reassuring.
Research has already confirmed that sizeism has a negative impact on both a patient’s physical health and psychological well-being, and as medical providers, we’re part of the problem. We cause distress in our patients through disrespectful treatment and medical fat shaming, which can lead to cycles of disordered eating, reduced physical activity, and more weight gain. We discriminate based on weight, causing our patients to delay health care visits and other provider interactions, resulting in increased risks for morbidity and even mortality. We make assumptions that a patient’s presenting complaints are due to weight rather than other causes, resulting in missed diagnoses. And we recommend different treatments for obese patients with the same condition as nonobese patients simply because of their weight.
One study has suggested that over 40% of adults in the United States have suffered from weight stigma, and physicians and coworkers are listed as some of the most common sources. Another study suggests that nearly 70% of overweight or obese patients report feeling stigmatized by physicians, whether through expressed biases or purposeful avoidance (patients have previously reported that their providers addressed weight loss in fewer than 20% of their examinations).
As health care providers, we need to do better. We should all be willing to consider our own biases about body size, and there are self-assessments to help with this, including the Implicit Associations Test: Weight Bias. By becoming more self-aware, hopefully we can change the doctor-patient conversation about weight management.
Studies have shown that meaningful conversations with physicians can have a significant impact on patients’ attempts to change behaviors related to weight. Yet, many medical providers are not trained in how to counsel patients on nutrition, weight loss, and physical activity (if we bring it up at all). We need to better educate ourselves about weight science and treatments.
In the meantime, we can work on how we interact with our patients:
- Make sure that your practice space is accommodating and nondiscriminatory, with appropriately sized furniture in the waiting and exam rooms, large blood pressure cuffs and gowns, and size-inclusive reading materials.
- Ensure that your workplace has an antiharassment policy that includes sizeism.
- Be an ally and speak up against weight discrimination.
- Educate your office staff about weight stigma and ensure that they avoid commenting on the weight or body size of others (being recognized only for losing weight isn’t a compliment, and sharing “fat jokes” isn’t funny).
- Remember that a person’s body size tells you nothing about that person’s health behaviors. Stop assuming that larger body sizes are related to laziness, overeating, or a lack of motivation.
- Ask your overweight or obese patients if they are willing to talk about their weight before jumping into the topic.
- Practice (patients are more likely to report changing their exercise routine and attempting to lose weight with these techniques).
- Be mindful of your word choices; for example, it can be more helpful to focus on comorbidities (such as high blood pressure or prediabetes) rather than body weight, nutrition rather than dieting, and physical activity rather than specific exercises.
Regardless of how you feel about reciting the Hippocratic Oath, our patients, no matter their body size, deserve to be treated with respect and dignity, as others have said in more eloquent ways than I. Let’s stop playing the fat shame game and help fight weight bias in medicine.
Dr. Devlin is president, Locum Infectious Disease Services, and an independent contractor for Weatherby Healthcare. She reported no relevant conflicts of interest. A version of this article first appeared on Medscape.com.
Congenital syphilis: It’s still a significant public health problem
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
Why people lie about COVID
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.