User login
A new ultrabrief screening scale for pediatric OCD
Obsessive-compulsive disorder (OCD) affects 1-2% of the population. The disorder is characterized by recurrent intrusive unwanted thoughts (obsessions) that cause significant distress and anxiety, and behavioral or mental rituals (compulsions) that are performed to reduce distress stemming from obsessions. OCD may onset at any time in life, but most commonly begins in childhood or in early adulthood.
Cognitive behavioral therapy (CBT) with exposure and response prevention is an empirically based and highly effective treatment for OCD. However, most youth with OCD do not receive any treatment, which is related to a shortage of mental health care providers with expertise in assessment and treatment of the disorder, and misdiagnosis of the disorder is all too prevalent.
Aside from the subjective emotional toll associated with OCD, individuals living with this disorder frequently experience interpersonal, academic, and vocational impairments. Nevertheless, OCD is often overlooked or misdiagnosed. This may be more pronounced in youth with OCD, particularly in primary health care settings and large nonspecialized medical institutions. In fact, research indicates that pediatric OCD is often underrecognized even among mental health professionals. This situation is not new, and in fact the National Institute for Health and Care Excellence (NICE) in the United Kingdom stated that there is an urgent need to develop brief reliable screeners for OCD nearly 20 years ago.
Although there were several attempts to develop brief screening scales for adults and youth with OCD, none of them were found to be suitable for use as rapid screening tools in nonspecialized settings. One of the primary reasons is that OCD is associated with different “themes” or dimensions. For example, a child with OCD may engage in cleaning rituals because the context (or dimension) of their obsessions is contamination concerns. Another child with OCD, who may suffer from similar overall symptom severity, may primarily engage in checking rituals which are related with obsessions associated with fear of being responsible for harm. Therefore, one child with OCD may score very high on items assessing one dimension (e.g., contamination concerns), but very low on another dimension (e.g., harm obsessions).
This results in a known challenge in the assessment and psychometrics of self-report (as opposed to clinician administered) measures of OCD. Secondly, development of such measures requires very large carefully screened samples of individuals with OCD, with other disorders, and those without a known psychological disorder – which may be more challenging than requiring adult participants.
To accomplish this, we harmonized data from several sites that included three samples of carefully screened youths with OCD, with other disorders, and without known disorders who completed multiple self-report questionnaires, including the 21-item Obsessive-Compulsive Inventory – Child Version (OCI-CV).
Utilizing psychometric analyses including factor analyses, invariance analyses, and item response theory methodologies, we were able to develop an ultrabrief measure extracted from the OCI-CV: the 5-item Obsessive-Compulsive Inventory – Child Version (OCI-CV-5). This very brief self-report measure was found to have very good psychometric properties including a sensitive and specific clinical cutoff score. Youth who score at or above the cutoff score are nearly 21 times more likely to meet criteria for OCD.
This measure corresponds to a need to rapidly screen for OCD in children in nonspecialized settings, including community mental health clinics, primary care settings, and pediatric treatment facilities. However, it is important to note it is not a diagnostic measure. The measure is intended to identify youth who should be referred to a mental health care professional to conduct a diagnostic interview.
Dr. Abramovitch is a clinical psychologist and neuropsychologist based in Austin, Tex., and an associate professor at Texas State University. Dr. Abramowitz is professor and director of clinical training in the Anxiety and Stress Lab at University of North Carolina at Chapel Hill. Dr. McKay is professor of psychology at Fordham University, Bronx, N.Y.
Obsessive-compulsive disorder (OCD) affects 1-2% of the population. The disorder is characterized by recurrent intrusive unwanted thoughts (obsessions) that cause significant distress and anxiety, and behavioral or mental rituals (compulsions) that are performed to reduce distress stemming from obsessions. OCD may onset at any time in life, but most commonly begins in childhood or in early adulthood.
Cognitive behavioral therapy (CBT) with exposure and response prevention is an empirically based and highly effective treatment for OCD. However, most youth with OCD do not receive any treatment, which is related to a shortage of mental health care providers with expertise in assessment and treatment of the disorder, and misdiagnosis of the disorder is all too prevalent.
Aside from the subjective emotional toll associated with OCD, individuals living with this disorder frequently experience interpersonal, academic, and vocational impairments. Nevertheless, OCD is often overlooked or misdiagnosed. This may be more pronounced in youth with OCD, particularly in primary health care settings and large nonspecialized medical institutions. In fact, research indicates that pediatric OCD is often underrecognized even among mental health professionals. This situation is not new, and in fact the National Institute for Health and Care Excellence (NICE) in the United Kingdom stated that there is an urgent need to develop brief reliable screeners for OCD nearly 20 years ago.
Although there were several attempts to develop brief screening scales for adults and youth with OCD, none of them were found to be suitable for use as rapid screening tools in nonspecialized settings. One of the primary reasons is that OCD is associated with different “themes” or dimensions. For example, a child with OCD may engage in cleaning rituals because the context (or dimension) of their obsessions is contamination concerns. Another child with OCD, who may suffer from similar overall symptom severity, may primarily engage in checking rituals which are related with obsessions associated with fear of being responsible for harm. Therefore, one child with OCD may score very high on items assessing one dimension (e.g., contamination concerns), but very low on another dimension (e.g., harm obsessions).
This results in a known challenge in the assessment and psychometrics of self-report (as opposed to clinician administered) measures of OCD. Secondly, development of such measures requires very large carefully screened samples of individuals with OCD, with other disorders, and those without a known psychological disorder – which may be more challenging than requiring adult participants.
To accomplish this, we harmonized data from several sites that included three samples of carefully screened youths with OCD, with other disorders, and without known disorders who completed multiple self-report questionnaires, including the 21-item Obsessive-Compulsive Inventory – Child Version (OCI-CV).
Utilizing psychometric analyses including factor analyses, invariance analyses, and item response theory methodologies, we were able to develop an ultrabrief measure extracted from the OCI-CV: the 5-item Obsessive-Compulsive Inventory – Child Version (OCI-CV-5). This very brief self-report measure was found to have very good psychometric properties including a sensitive and specific clinical cutoff score. Youth who score at or above the cutoff score are nearly 21 times more likely to meet criteria for OCD.
This measure corresponds to a need to rapidly screen for OCD in children in nonspecialized settings, including community mental health clinics, primary care settings, and pediatric treatment facilities. However, it is important to note it is not a diagnostic measure. The measure is intended to identify youth who should be referred to a mental health care professional to conduct a diagnostic interview.
Dr. Abramovitch is a clinical psychologist and neuropsychologist based in Austin, Tex., and an associate professor at Texas State University. Dr. Abramowitz is professor and director of clinical training in the Anxiety and Stress Lab at University of North Carolina at Chapel Hill. Dr. McKay is professor of psychology at Fordham University, Bronx, N.Y.
Obsessive-compulsive disorder (OCD) affects 1-2% of the population. The disorder is characterized by recurrent intrusive unwanted thoughts (obsessions) that cause significant distress and anxiety, and behavioral or mental rituals (compulsions) that are performed to reduce distress stemming from obsessions. OCD may onset at any time in life, but most commonly begins in childhood or in early adulthood.
Cognitive behavioral therapy (CBT) with exposure and response prevention is an empirically based and highly effective treatment for OCD. However, most youth with OCD do not receive any treatment, which is related to a shortage of mental health care providers with expertise in assessment and treatment of the disorder, and misdiagnosis of the disorder is all too prevalent.
Aside from the subjective emotional toll associated with OCD, individuals living with this disorder frequently experience interpersonal, academic, and vocational impairments. Nevertheless, OCD is often overlooked or misdiagnosed. This may be more pronounced in youth with OCD, particularly in primary health care settings and large nonspecialized medical institutions. In fact, research indicates that pediatric OCD is often underrecognized even among mental health professionals. This situation is not new, and in fact the National Institute for Health and Care Excellence (NICE) in the United Kingdom stated that there is an urgent need to develop brief reliable screeners for OCD nearly 20 years ago.
Although there were several attempts to develop brief screening scales for adults and youth with OCD, none of them were found to be suitable for use as rapid screening tools in nonspecialized settings. One of the primary reasons is that OCD is associated with different “themes” or dimensions. For example, a child with OCD may engage in cleaning rituals because the context (or dimension) of their obsessions is contamination concerns. Another child with OCD, who may suffer from similar overall symptom severity, may primarily engage in checking rituals which are related with obsessions associated with fear of being responsible for harm. Therefore, one child with OCD may score very high on items assessing one dimension (e.g., contamination concerns), but very low on another dimension (e.g., harm obsessions).
This results in a known challenge in the assessment and psychometrics of self-report (as opposed to clinician administered) measures of OCD. Secondly, development of such measures requires very large carefully screened samples of individuals with OCD, with other disorders, and those without a known psychological disorder – which may be more challenging than requiring adult participants.
To accomplish this, we harmonized data from several sites that included three samples of carefully screened youths with OCD, with other disorders, and without known disorders who completed multiple self-report questionnaires, including the 21-item Obsessive-Compulsive Inventory – Child Version (OCI-CV).
Utilizing psychometric analyses including factor analyses, invariance analyses, and item response theory methodologies, we were able to develop an ultrabrief measure extracted from the OCI-CV: the 5-item Obsessive-Compulsive Inventory – Child Version (OCI-CV-5). This very brief self-report measure was found to have very good psychometric properties including a sensitive and specific clinical cutoff score. Youth who score at or above the cutoff score are nearly 21 times more likely to meet criteria for OCD.
This measure corresponds to a need to rapidly screen for OCD in children in nonspecialized settings, including community mental health clinics, primary care settings, and pediatric treatment facilities. However, it is important to note it is not a diagnostic measure. The measure is intended to identify youth who should be referred to a mental health care professional to conduct a diagnostic interview.
Dr. Abramovitch is a clinical psychologist and neuropsychologist based in Austin, Tex., and an associate professor at Texas State University. Dr. Abramowitz is professor and director of clinical training in the Anxiety and Stress Lab at University of North Carolina at Chapel Hill. Dr. McKay is professor of psychology at Fordham University, Bronx, N.Y.
Children from poorer ZIP codes often untreated for ear infections
Children from socially disadvantaged backgrounds are less likely to be treated for middle ear infections and are likely to experience serious complications from the condition – potentially with lifelong economic consequences – researchers have found.
Problems such as hearing loss and chronic ear infections were more common for children who lived in areas marked by difficult socioeconomic circumstances, according to the researchers, who linked the complications to a lack of adequate treatment in this population.
“We are treating socially disadvantaged kids differently than we are treating more advantaged kids,” said Jason Qian, MD, a resident in otolaryngology and head and neck surgery at Stanford (Calif.) University, who helped conduct the new study. “We have to think about social inequalities so we can ensure all kids are receiving the same level and type of care.”
In the United States, 80% of children will experience otitis media during their lifetime. Untreated ear infections can lead to symptoms ranging from mild discharge from the ear to life-threatening conditions like mastoiditis and intracranial abscesses.
For the new study, published online in JAMA Otolaryngology–Head & Neck Surgery, Dr. Qian and colleagues looked at 4.8 million children with private health insurance across the United States using a database with information on inpatient and outpatient visits and medication use. The researchers identified patients between January 2003 and March 2021 who received treatment for recurrent and suppurative otitis media, those who received tympanostomy tubes, and children who experienced severe complications from undertreated ear infections.
Social disadvantage was assessed using the Social Deprivation Index (SDI), a tool used to measure indicators of poverty throughout the United States based on seven demographic factors including level of educational attainment, the number of single-parent households, the share of people living in overcrowded homes, and other factors.
Every point increase in the SDI score was associated with a 14% lower likelihood of being treated for recurrent ear infections despite having them and a 28% greater chance of being hospitalized for severe ear infections, according to the researchers.
Previous research established that children with government health insurance or no coverage have more difficulty receiving proper treatment for ear infections. Although people with commercial insurance are generally wealthier than those without private coverage, Dr. Qian said, the new data indicate that significant social disparities in care exist even within this group.
Although some studies have found that wealthier children are more likely to develop otitis media, Dr. Qian’s group said that association likely reflects the better access to health care money affords.
“We found that socially disadvantaged children not only have a higher burden of otitis media but are also undertreated both medically and surgically for [ear infections]. Because chronic and complicated forms of otitis media can cause childhood hearing loss, which in turn limits academic and economic potential, undertreatment of [otitis media] in socially disadvantaged populations can contribute to generational cycles of poverty, unemployment, and low pay,” they write.
“The biggest take home is that we are not treating children equitably when it comes to ear infections,” Dr. Qian added. “In order to give children equal access to care, we as health care providers need to find strategies to do better.”
The study was supported by the Stanford Center for Population Health Science Data Core, which is supported by a grant from the National Institutes of Health and internal funding. Dr. Qian has reported receiving grant funding from Merck.
A version of this article first appeared on Medscape.com.
Children from socially disadvantaged backgrounds are less likely to be treated for middle ear infections and are likely to experience serious complications from the condition – potentially with lifelong economic consequences – researchers have found.
Problems such as hearing loss and chronic ear infections were more common for children who lived in areas marked by difficult socioeconomic circumstances, according to the researchers, who linked the complications to a lack of adequate treatment in this population.
“We are treating socially disadvantaged kids differently than we are treating more advantaged kids,” said Jason Qian, MD, a resident in otolaryngology and head and neck surgery at Stanford (Calif.) University, who helped conduct the new study. “We have to think about social inequalities so we can ensure all kids are receiving the same level and type of care.”
In the United States, 80% of children will experience otitis media during their lifetime. Untreated ear infections can lead to symptoms ranging from mild discharge from the ear to life-threatening conditions like mastoiditis and intracranial abscesses.
For the new study, published online in JAMA Otolaryngology–Head & Neck Surgery, Dr. Qian and colleagues looked at 4.8 million children with private health insurance across the United States using a database with information on inpatient and outpatient visits and medication use. The researchers identified patients between January 2003 and March 2021 who received treatment for recurrent and suppurative otitis media, those who received tympanostomy tubes, and children who experienced severe complications from undertreated ear infections.
Social disadvantage was assessed using the Social Deprivation Index (SDI), a tool used to measure indicators of poverty throughout the United States based on seven demographic factors including level of educational attainment, the number of single-parent households, the share of people living in overcrowded homes, and other factors.
Every point increase in the SDI score was associated with a 14% lower likelihood of being treated for recurrent ear infections despite having them and a 28% greater chance of being hospitalized for severe ear infections, according to the researchers.
Previous research established that children with government health insurance or no coverage have more difficulty receiving proper treatment for ear infections. Although people with commercial insurance are generally wealthier than those without private coverage, Dr. Qian said, the new data indicate that significant social disparities in care exist even within this group.
Although some studies have found that wealthier children are more likely to develop otitis media, Dr. Qian’s group said that association likely reflects the better access to health care money affords.
“We found that socially disadvantaged children not only have a higher burden of otitis media but are also undertreated both medically and surgically for [ear infections]. Because chronic and complicated forms of otitis media can cause childhood hearing loss, which in turn limits academic and economic potential, undertreatment of [otitis media] in socially disadvantaged populations can contribute to generational cycles of poverty, unemployment, and low pay,” they write.
“The biggest take home is that we are not treating children equitably when it comes to ear infections,” Dr. Qian added. “In order to give children equal access to care, we as health care providers need to find strategies to do better.”
The study was supported by the Stanford Center for Population Health Science Data Core, which is supported by a grant from the National Institutes of Health and internal funding. Dr. Qian has reported receiving grant funding from Merck.
A version of this article first appeared on Medscape.com.
Children from socially disadvantaged backgrounds are less likely to be treated for middle ear infections and are likely to experience serious complications from the condition – potentially with lifelong economic consequences – researchers have found.
Problems such as hearing loss and chronic ear infections were more common for children who lived in areas marked by difficult socioeconomic circumstances, according to the researchers, who linked the complications to a lack of adequate treatment in this population.
“We are treating socially disadvantaged kids differently than we are treating more advantaged kids,” said Jason Qian, MD, a resident in otolaryngology and head and neck surgery at Stanford (Calif.) University, who helped conduct the new study. “We have to think about social inequalities so we can ensure all kids are receiving the same level and type of care.”
In the United States, 80% of children will experience otitis media during their lifetime. Untreated ear infections can lead to symptoms ranging from mild discharge from the ear to life-threatening conditions like mastoiditis and intracranial abscesses.
For the new study, published online in JAMA Otolaryngology–Head & Neck Surgery, Dr. Qian and colleagues looked at 4.8 million children with private health insurance across the United States using a database with information on inpatient and outpatient visits and medication use. The researchers identified patients between January 2003 and March 2021 who received treatment for recurrent and suppurative otitis media, those who received tympanostomy tubes, and children who experienced severe complications from undertreated ear infections.
Social disadvantage was assessed using the Social Deprivation Index (SDI), a tool used to measure indicators of poverty throughout the United States based on seven demographic factors including level of educational attainment, the number of single-parent households, the share of people living in overcrowded homes, and other factors.
Every point increase in the SDI score was associated with a 14% lower likelihood of being treated for recurrent ear infections despite having them and a 28% greater chance of being hospitalized for severe ear infections, according to the researchers.
Previous research established that children with government health insurance or no coverage have more difficulty receiving proper treatment for ear infections. Although people with commercial insurance are generally wealthier than those without private coverage, Dr. Qian said, the new data indicate that significant social disparities in care exist even within this group.
Although some studies have found that wealthier children are more likely to develop otitis media, Dr. Qian’s group said that association likely reflects the better access to health care money affords.
“We found that socially disadvantaged children not only have a higher burden of otitis media but are also undertreated both medically and surgically for [ear infections]. Because chronic and complicated forms of otitis media can cause childhood hearing loss, which in turn limits academic and economic potential, undertreatment of [otitis media] in socially disadvantaged populations can contribute to generational cycles of poverty, unemployment, and low pay,” they write.
“The biggest take home is that we are not treating children equitably when it comes to ear infections,” Dr. Qian added. “In order to give children equal access to care, we as health care providers need to find strategies to do better.”
The study was supported by the Stanford Center for Population Health Science Data Core, which is supported by a grant from the National Institutes of Health and internal funding. Dr. Qian has reported receiving grant funding from Merck.
A version of this article first appeared on Medscape.com.
Monkeypox in children appears rare and relatively mild
Monkeypox virus infections in children and adolescents in the United States are rare, and young patients with known infections have all recovered, according to a study from the Centers for Disease Control and Prevention.
In addition, evidence suggests that secondary transmission in schools or childcare facilities may be unlikely.
The study was the first comprehensive study on the impact of monkeypox on children during the 2022 outbreak, according to a statement emailed to this news organization from the California Department of Public Health, one of the state health departments that partnered with the CDC to share information.
News of low infection rates and relatively mild disease was welcome to clinicians, who had braced for severe findings on the basis of sparse prior data, according to Peter Chin-Hong, MD, a professor of medicine and an infectious diseases physician at the University of California, San Francisco.
“We were on heightened alert that kids may do poorly,” said Dr. Chin-Hong, who was not involved in the study but who cared for monkeypox patients during the outbreak. “I think this study is reassuring.
“The other silver lining about it is that most of the kids got infected in the household setting from ways that you would expect them to get [infected],” Dr. Chin-Hong said in an interview.
However, Black and Hispanic children were more likely to contract the disease, underscoring troubling inequities.
“Early on, individuals of color were much less likely to be able to successfully access vaccination,” said first author Ian Hennessee, PhD, MPH, an epidemic intelligence service officer with the CDC and a member of the Special Case Investigation Unit of the Multinational Monkeypox Response Team at the CDC. “We think those kinds of structural inequities really trickled down towards the children and adolescents that have been affected by this outbreak.”
The study was published in Morbidity and Mortality Weekly Report.
A nationwide look at the data
The researchers discussed 83 children and adolescents with monkeypox who came to the CDC’s attention between May 17 and Sept. 24, 2022.
The 83 cases represent 0.3% of the 25,038 reported monkeypox cases in the United States over that period. Of the 28 children aged 12 years or younger, 18 (64%) were boys. Sixteen children were younger than 4 years.
Exposure data were available for 20 (71%) of those aged 0-12. In that group, 19 were exposed at home; 17 cases were due to routine skin-to-skin contact with a household caregiver; and one case was suspected to be caused by fomites (such as a shared towel). Exposure information was unavailable for the remaining case.
Most of the children experienced lesions on the trunk. No lesions were anogenital. Two patients in the youngest age group were hospitalized because of widespread rash that involved the eyelids, and a patient in the 5- to 12-year-old group was hospitalized because of periorbital cellulitis and conjunctivitis.
Among those aged 13-17, there were 55 cases. Of these patients, 89% were boys. Exposure data were available for 35 (64%). In 32 of these patients, the infection occurred from presumed sexual contact. Twenty-three of those adolescents reported male-to-male sexual contact. No case was found to be connected with sexual abuse.
Lesions in the adolescents were mostly truncal or anogenital. Six in this group were hospitalized, and all of them recovered. One adolescent was found to be HIV positive.
Black and Hispanic children accounted for 47% and 35% of all cases, respectively.
Eleven percent of all the children and adolescents were hospitalized, and none received intensive care.
Treatments, when given, included the antiviral drug tecovirimat, intravenous vaccinia immune globulin, and topical trifluridine. There were no deaths.
Ten symptomatic patients attended school or daycare. Among these patients, no secondary transmissions were found to have occurred. Some contacts were offered the JYNNEOS monkeypox vaccine as postexposure prophylaxis.
Limitations of the study included potentially overlooked cases. Data were collected through routine surveillance, children frequently experience rashes, and access to testing has been a challenge, Dr. Hennessee explained.
In addition, data on exposure characteristics were missing for some children.
Inequities and the risks of being judged
The outbreak in the United States has eased in recent months. However, though uncommon in children, monkeypox has affected some racial groups disproportionately.
“Especially in the later course of the outbreak, the majority of cases were among Black and Hispanic individuals,” said co-author Rachel E. Harold, MD, an infectious diseases specialist and supervisory medical officer with the District of Columbia Department of Health’s HIV/AIDS, Hepatitis, STDs, and TB Administration.
“Unfortunately, the pediatric cases do reflect the outbreak overall,” she told this news organization.
Dr. Harold noted there have been efforts in D.C. and other jurisdictions, as well as by the White House monkeypox response team, to reach populations at greatest risk and that they were “really trying to make vaccine available to people of color.”
Vaccination clinics often popped up in unexpected locations at short notice, and that made it hard for some people to get to them, Dr. Chin-Hong pointed out.
Another factor was “the public aspect of accessing diagnostics and vaccines and the way that that’s linked to potential judgment or sexual risk,” he added.
“Not everybody’s out,” Dr. Chin-Hong said, referring to members of the LGBTQ community. “In many communities of color, going to get a test or going to get a vaccine essentially means that you’re out.”
For clinicians who suspect monkeypox in a child, Dr. Harold suggests keeping a broad differential diagnosis, looking for an epidemiologic link, and contacting the CDC for assistance. Infected children should be encouraged to avoid touching their own eyes or mucous membranes, she added.
In addition, she said, tecovirimat is a reasonable treatment and is well tolerated by pediatric monkeypox patients with eczema, an underlying condition that could lead to severe disease.
For infected caregivers, Dr. Hennessee said, measures to prevent infecting children at home include isolation, contact precautions, and in some cases, postexposure prophylaxis via vaccination.
For sexually active adolescents, he advised that clinicians offer vaccination, education on sexual health, and testing for HIV and other sexually transmitted infections.
“It’s important to remember that adolescents may be sexually active, and clinicians should do a thorough and nonjudgmental sexual history,” Dr. Harold added. “That is always true, but especially if there is concern for [monkeypox].”
Dr. Hennessee, Dr. Chin-Hong, and Dr. Harold have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Monkeypox virus infections in children and adolescents in the United States are rare, and young patients with known infections have all recovered, according to a study from the Centers for Disease Control and Prevention.
In addition, evidence suggests that secondary transmission in schools or childcare facilities may be unlikely.
The study was the first comprehensive study on the impact of monkeypox on children during the 2022 outbreak, according to a statement emailed to this news organization from the California Department of Public Health, one of the state health departments that partnered with the CDC to share information.
News of low infection rates and relatively mild disease was welcome to clinicians, who had braced for severe findings on the basis of sparse prior data, according to Peter Chin-Hong, MD, a professor of medicine and an infectious diseases physician at the University of California, San Francisco.
“We were on heightened alert that kids may do poorly,” said Dr. Chin-Hong, who was not involved in the study but who cared for monkeypox patients during the outbreak. “I think this study is reassuring.
“The other silver lining about it is that most of the kids got infected in the household setting from ways that you would expect them to get [infected],” Dr. Chin-Hong said in an interview.
However, Black and Hispanic children were more likely to contract the disease, underscoring troubling inequities.
“Early on, individuals of color were much less likely to be able to successfully access vaccination,” said first author Ian Hennessee, PhD, MPH, an epidemic intelligence service officer with the CDC and a member of the Special Case Investigation Unit of the Multinational Monkeypox Response Team at the CDC. “We think those kinds of structural inequities really trickled down towards the children and adolescents that have been affected by this outbreak.”
The study was published in Morbidity and Mortality Weekly Report.
A nationwide look at the data
The researchers discussed 83 children and adolescents with monkeypox who came to the CDC’s attention between May 17 and Sept. 24, 2022.
The 83 cases represent 0.3% of the 25,038 reported monkeypox cases in the United States over that period. Of the 28 children aged 12 years or younger, 18 (64%) were boys. Sixteen children were younger than 4 years.
Exposure data were available for 20 (71%) of those aged 0-12. In that group, 19 were exposed at home; 17 cases were due to routine skin-to-skin contact with a household caregiver; and one case was suspected to be caused by fomites (such as a shared towel). Exposure information was unavailable for the remaining case.
Most of the children experienced lesions on the trunk. No lesions were anogenital. Two patients in the youngest age group were hospitalized because of widespread rash that involved the eyelids, and a patient in the 5- to 12-year-old group was hospitalized because of periorbital cellulitis and conjunctivitis.
Among those aged 13-17, there were 55 cases. Of these patients, 89% were boys. Exposure data were available for 35 (64%). In 32 of these patients, the infection occurred from presumed sexual contact. Twenty-three of those adolescents reported male-to-male sexual contact. No case was found to be connected with sexual abuse.
Lesions in the adolescents were mostly truncal or anogenital. Six in this group were hospitalized, and all of them recovered. One adolescent was found to be HIV positive.
Black and Hispanic children accounted for 47% and 35% of all cases, respectively.
Eleven percent of all the children and adolescents were hospitalized, and none received intensive care.
Treatments, when given, included the antiviral drug tecovirimat, intravenous vaccinia immune globulin, and topical trifluridine. There were no deaths.
Ten symptomatic patients attended school or daycare. Among these patients, no secondary transmissions were found to have occurred. Some contacts were offered the JYNNEOS monkeypox vaccine as postexposure prophylaxis.
Limitations of the study included potentially overlooked cases. Data were collected through routine surveillance, children frequently experience rashes, and access to testing has been a challenge, Dr. Hennessee explained.
In addition, data on exposure characteristics were missing for some children.
Inequities and the risks of being judged
The outbreak in the United States has eased in recent months. However, though uncommon in children, monkeypox has affected some racial groups disproportionately.
“Especially in the later course of the outbreak, the majority of cases were among Black and Hispanic individuals,” said co-author Rachel E. Harold, MD, an infectious diseases specialist and supervisory medical officer with the District of Columbia Department of Health’s HIV/AIDS, Hepatitis, STDs, and TB Administration.
“Unfortunately, the pediatric cases do reflect the outbreak overall,” she told this news organization.
Dr. Harold noted there have been efforts in D.C. and other jurisdictions, as well as by the White House monkeypox response team, to reach populations at greatest risk and that they were “really trying to make vaccine available to people of color.”
Vaccination clinics often popped up in unexpected locations at short notice, and that made it hard for some people to get to them, Dr. Chin-Hong pointed out.
Another factor was “the public aspect of accessing diagnostics and vaccines and the way that that’s linked to potential judgment or sexual risk,” he added.
“Not everybody’s out,” Dr. Chin-Hong said, referring to members of the LGBTQ community. “In many communities of color, going to get a test or going to get a vaccine essentially means that you’re out.”
For clinicians who suspect monkeypox in a child, Dr. Harold suggests keeping a broad differential diagnosis, looking for an epidemiologic link, and contacting the CDC for assistance. Infected children should be encouraged to avoid touching their own eyes or mucous membranes, she added.
In addition, she said, tecovirimat is a reasonable treatment and is well tolerated by pediatric monkeypox patients with eczema, an underlying condition that could lead to severe disease.
For infected caregivers, Dr. Hennessee said, measures to prevent infecting children at home include isolation, contact precautions, and in some cases, postexposure prophylaxis via vaccination.
For sexually active adolescents, he advised that clinicians offer vaccination, education on sexual health, and testing for HIV and other sexually transmitted infections.
“It’s important to remember that adolescents may be sexually active, and clinicians should do a thorough and nonjudgmental sexual history,” Dr. Harold added. “That is always true, but especially if there is concern for [monkeypox].”
Dr. Hennessee, Dr. Chin-Hong, and Dr. Harold have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Monkeypox virus infections in children and adolescents in the United States are rare, and young patients with known infections have all recovered, according to a study from the Centers for Disease Control and Prevention.
In addition, evidence suggests that secondary transmission in schools or childcare facilities may be unlikely.
The study was the first comprehensive study on the impact of monkeypox on children during the 2022 outbreak, according to a statement emailed to this news organization from the California Department of Public Health, one of the state health departments that partnered with the CDC to share information.
News of low infection rates and relatively mild disease was welcome to clinicians, who had braced for severe findings on the basis of sparse prior data, according to Peter Chin-Hong, MD, a professor of medicine and an infectious diseases physician at the University of California, San Francisco.
“We were on heightened alert that kids may do poorly,” said Dr. Chin-Hong, who was not involved in the study but who cared for monkeypox patients during the outbreak. “I think this study is reassuring.
“The other silver lining about it is that most of the kids got infected in the household setting from ways that you would expect them to get [infected],” Dr. Chin-Hong said in an interview.
However, Black and Hispanic children were more likely to contract the disease, underscoring troubling inequities.
“Early on, individuals of color were much less likely to be able to successfully access vaccination,” said first author Ian Hennessee, PhD, MPH, an epidemic intelligence service officer with the CDC and a member of the Special Case Investigation Unit of the Multinational Monkeypox Response Team at the CDC. “We think those kinds of structural inequities really trickled down towards the children and adolescents that have been affected by this outbreak.”
The study was published in Morbidity and Mortality Weekly Report.
A nationwide look at the data
The researchers discussed 83 children and adolescents with monkeypox who came to the CDC’s attention between May 17 and Sept. 24, 2022.
The 83 cases represent 0.3% of the 25,038 reported monkeypox cases in the United States over that period. Of the 28 children aged 12 years or younger, 18 (64%) were boys. Sixteen children were younger than 4 years.
Exposure data were available for 20 (71%) of those aged 0-12. In that group, 19 were exposed at home; 17 cases were due to routine skin-to-skin contact with a household caregiver; and one case was suspected to be caused by fomites (such as a shared towel). Exposure information was unavailable for the remaining case.
Most of the children experienced lesions on the trunk. No lesions were anogenital. Two patients in the youngest age group were hospitalized because of widespread rash that involved the eyelids, and a patient in the 5- to 12-year-old group was hospitalized because of periorbital cellulitis and conjunctivitis.
Among those aged 13-17, there were 55 cases. Of these patients, 89% were boys. Exposure data were available for 35 (64%). In 32 of these patients, the infection occurred from presumed sexual contact. Twenty-three of those adolescents reported male-to-male sexual contact. No case was found to be connected with sexual abuse.
Lesions in the adolescents were mostly truncal or anogenital. Six in this group were hospitalized, and all of them recovered. One adolescent was found to be HIV positive.
Black and Hispanic children accounted for 47% and 35% of all cases, respectively.
Eleven percent of all the children and adolescents were hospitalized, and none received intensive care.
Treatments, when given, included the antiviral drug tecovirimat, intravenous vaccinia immune globulin, and topical trifluridine. There were no deaths.
Ten symptomatic patients attended school or daycare. Among these patients, no secondary transmissions were found to have occurred. Some contacts were offered the JYNNEOS monkeypox vaccine as postexposure prophylaxis.
Limitations of the study included potentially overlooked cases. Data were collected through routine surveillance, children frequently experience rashes, and access to testing has been a challenge, Dr. Hennessee explained.
In addition, data on exposure characteristics were missing for some children.
Inequities and the risks of being judged
The outbreak in the United States has eased in recent months. However, though uncommon in children, monkeypox has affected some racial groups disproportionately.
“Especially in the later course of the outbreak, the majority of cases were among Black and Hispanic individuals,” said co-author Rachel E. Harold, MD, an infectious diseases specialist and supervisory medical officer with the District of Columbia Department of Health’s HIV/AIDS, Hepatitis, STDs, and TB Administration.
“Unfortunately, the pediatric cases do reflect the outbreak overall,” she told this news organization.
Dr. Harold noted there have been efforts in D.C. and other jurisdictions, as well as by the White House monkeypox response team, to reach populations at greatest risk and that they were “really trying to make vaccine available to people of color.”
Vaccination clinics often popped up in unexpected locations at short notice, and that made it hard for some people to get to them, Dr. Chin-Hong pointed out.
Another factor was “the public aspect of accessing diagnostics and vaccines and the way that that’s linked to potential judgment or sexual risk,” he added.
“Not everybody’s out,” Dr. Chin-Hong said, referring to members of the LGBTQ community. “In many communities of color, going to get a test or going to get a vaccine essentially means that you’re out.”
For clinicians who suspect monkeypox in a child, Dr. Harold suggests keeping a broad differential diagnosis, looking for an epidemiologic link, and contacting the CDC for assistance. Infected children should be encouraged to avoid touching their own eyes or mucous membranes, she added.
In addition, she said, tecovirimat is a reasonable treatment and is well tolerated by pediatric monkeypox patients with eczema, an underlying condition that could lead to severe disease.
For infected caregivers, Dr. Hennessee said, measures to prevent infecting children at home include isolation, contact precautions, and in some cases, postexposure prophylaxis via vaccination.
For sexually active adolescents, he advised that clinicians offer vaccination, education on sexual health, and testing for HIV and other sexually transmitted infections.
“It’s important to remember that adolescents may be sexually active, and clinicians should do a thorough and nonjudgmental sexual history,” Dr. Harold added. “That is always true, but especially if there is concern for [monkeypox].”
Dr. Hennessee, Dr. Chin-Hong, and Dr. Harold have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Yellow Papules and Plaques on a Child
The Diagnosis: Tuberous Xanthoma
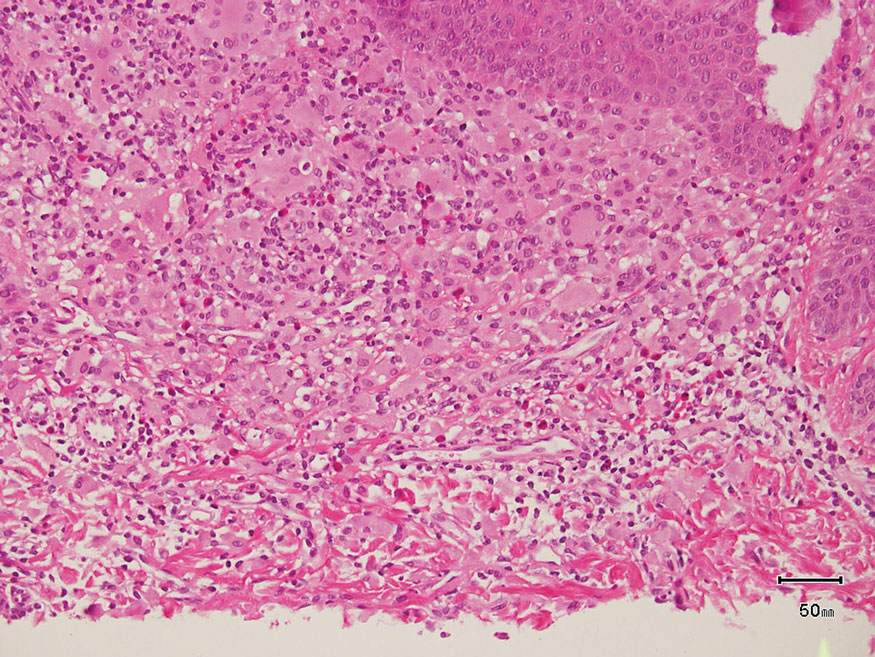
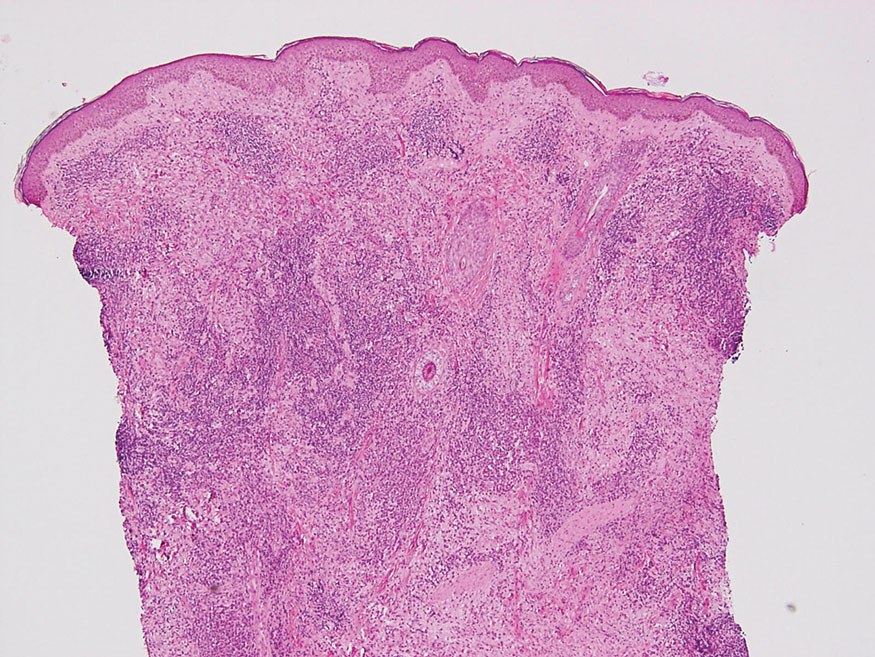
The skin biopsy revealed a nodular collection of foam cells (quiz image [bottom]). Tuberous xanthoma was the most likely diagnosis based on the patient’s history as well as the clinical and histologic findings. Tuberous xanthomas are flat or elevated nodules in the dermis and subcutaneous tissue, commonly occurring on the skin over the joints.1 Smaller nodules and papules often are referred to as tuberoeruptive xanthomas and exist on a continuum with the larger tuberous xanthomas. All xanthomas appear histologically similar, with collections of foam cells present within the dermis.2 Foam cells form when serum lipoproteins diffuse through capillary walls, deposit in the skin or tendons, and are scavenged by monocytes.3 Tuberous xanthomas, along with tendinous, eruptive, and planar xanthomas, are the most likely to be associated with hyperlipidemia.4 They may indicate an underlying disorder of lipid metabolism, such as familial hypercholesterolemia.1,3 This is the most common cause of inheritable cardiovascular disease, with a prevalence of approximately 1:250.2 Premature cardiovascular disease risk increases 2 to 4 times in patients with familial hypercholesterolemia and tendinous xanthomas,1 illustrating that recognition of cutaneous lesions can lead to earlier diagnosis and prevention of patient morbidity and mortality.
Juvenile xanthogranuloma typically presents as smooth yellow papules or nodules on the head and neck, with a characteristic “setting-sun” appearance (ie, yellow center with an erythematous halo) on dermoscopy.5 Histologically, juvenile xanthogranulomas are composed of foam cells and a mixed lymphohistiocytic infiltrate with eosinophils within the dermis. Giant cells with a ring of nuclei surrounded by cytoplasm containing lipid vacuoles (called Touton giant cells) are characteristic (Figure 1). In contrast to tuberous xanthomas, juvenile xanthogranulomas often present within the first year of life.6
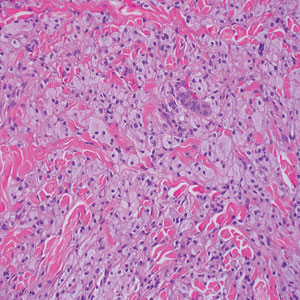
Keloid scars are more prevalent in patients with skin of color. They are characterized by eosinophilic keloidal collagen with a whorled proliferation of fibroblasts on histology (Figure 2).7 They occur spontaneously or at sites of injury and present as bluish-red or flesh-colored firm papules or nodules.8 In our patient, keloid scars were an unlikely diagnosis due to the lack of trauma and the absence of keloidal collagen on histology.
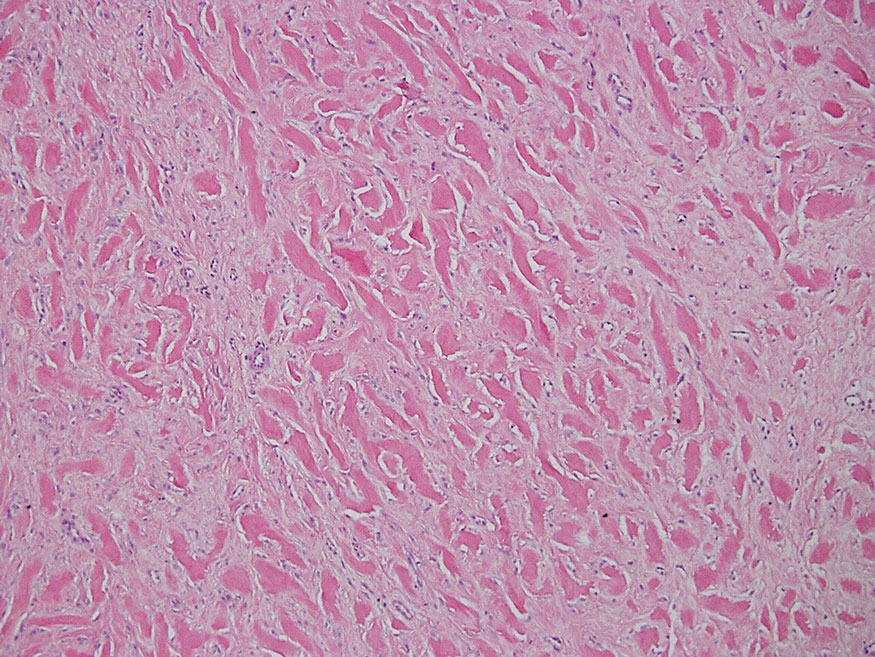
Necrobiosis lipoidica diabeticorum typically presents as an erythematous, yellow-brown, circular plaque on the anterior lower leg in patients with diabetes mellitus; it rarely occurs in children.9 Microscopy shows palisaded granulomas surrounding necrobiotic collagen arranged horizontally in a layer cake–like fashion (Figure 3).9,10 The etiology of necrobiosis lipoidica diabeticorum currently is unknown, though immune complex deposition may contribute to its pathology. It has been associated with type 1 diabetes mellitus, though severity of the lesions is not associated with extent of glycemic control.10
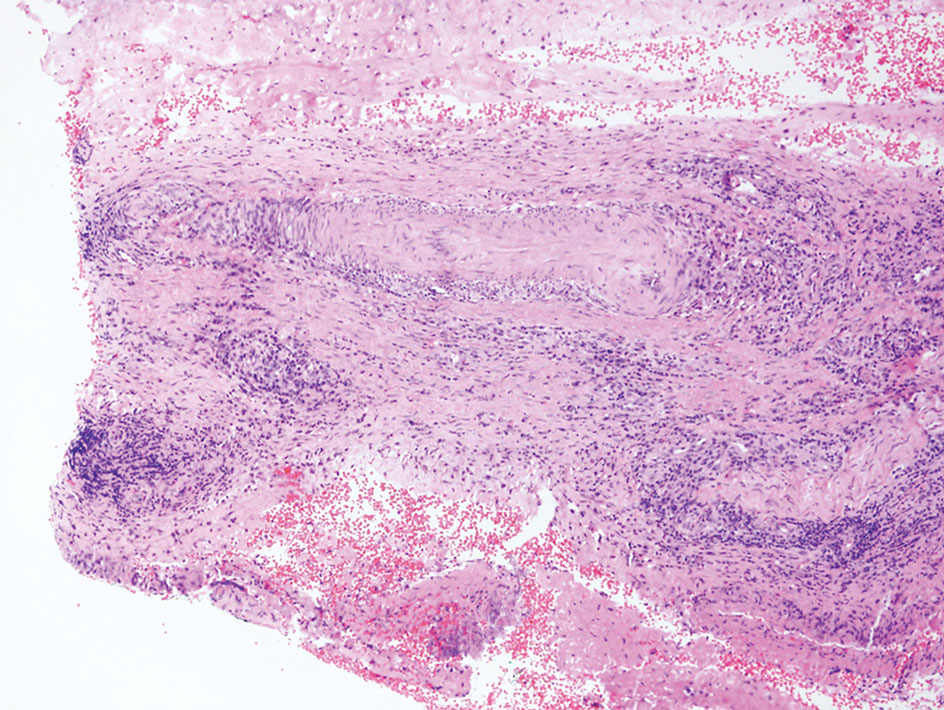
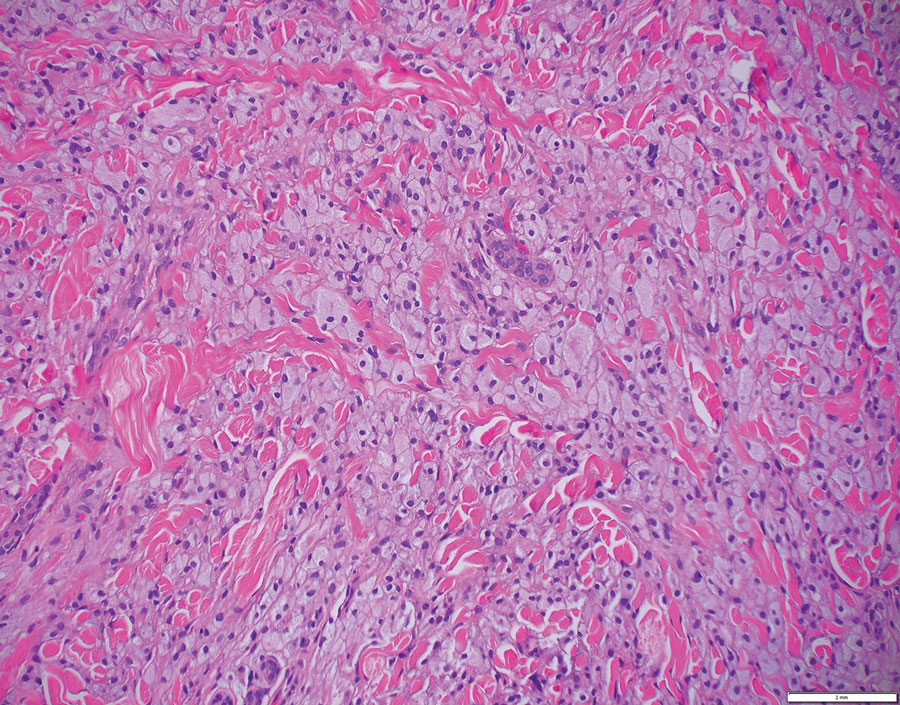
Rosai-Dorfman disease is an uncommon disorder characterized by a proliferation of histiocytes that most often presents as bilateral cervical lymphadenopathy in children and young adults but rarely can present with cutaneous lesions when extranodal involvement is present.11,12 The cutaneous form most commonly presents as red papules or nodules. On histology, the lesions exhibit a nodular dermal proliferation of histiocytes and smaller lymphocytoid cells with a marbled or starry sky–like appearance on low power (Figure 4). On higher magnification, the characteristic finding of emperipolesis can be seen.11 On immunohistochemistry, the histiocytes stain positively for CD68 and S-100. Although the pathogenesis currently is unknown, evidence of clonality indicates the disease may be related to a neoplastic process.12
- Zak A, Zeman M, Slaby A, et al. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158:181-188. doi:10.5507/bp.2014.016
- Ison HE, Clarke SL, Knowles JW. Familial hypercholesterolemia. In: Adam MP, Everman DB, Mirzaa GM, et al, eds. GeneReviews. University of Washington, Seattle; 1993-2022. https://www.ncbi.nlm.nih.gov/books/NBK174884/
- Sathiyakumar V, Jones SR, Martin SS. Xanthomas and lipoprotein disorders. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019.
- Massangale WT. Xanthomas. In: Bolognia JL, Schaffer JV, Cerroni L, et al, eds. Dermatology. Elsevier; 2018:1634-1643.
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. StatPearls. StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/
- Hernández-San Martín MJ, Vargas-Mora P, Aranibar L. Juvenile xanthogranuloma: an entity with a wide clinical spectrum. Actas Dermosifiliogr (Engl Ed). 2020;111:725-733. doi:10.1016/j.ad.2020.07.004
- Lee JY, Yang C, Chao S, et al. Histopathological differential diagnosis of keloid and hypertrophic scar. Am J Dermatopathology. 2004;26:379-384.
- Wolff K, Johnson R, Saavedra AP, et al. Benign neoplasms and hyperplasias. In: Wolff K, Johnson R, Saavedra AP, et al, eds. Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. 8th ed. McGraw Hill; 2017:141-188.
- Bonura C, Frontino G, Rigamonti A, et al. Necrobiosis lipoidica diabeticorum: a pediatric case report. Dermatoendocrinol. 2014;6:E27790. doi:10.4161/derm.27790
- Lepe K, Riley CA, Salazar FJ. Necrobiosis lipoidica. StatPearls. StatPearls Publishing; 2021. https://www-ncbi-nlm-nih-gov.proxy.kumc.edu/books/NBK459318/
- Parrent T, Clark T, Hall D. Cutaneous Rosai-Dorfman disease. Cutis. 2012;90:237-238.
- Bruce-Brand C, Schneider JW, Schubert P. Rosai-Dorfman disease: an overview. J Clin Pathol. 2020;73:697-705. doi:10.1136/jclinpath-2020-206733
The Diagnosis: Tuberous Xanthoma
The skin biopsy revealed a nodular collection of foam cells (quiz image [bottom]). Tuberous xanthoma was the most likely diagnosis based on the patient’s history as well as the clinical and histologic findings. Tuberous xanthomas are flat or elevated nodules in the dermis and subcutaneous tissue, commonly occurring on the skin over the joints.1 Smaller nodules and papules often are referred to as tuberoeruptive xanthomas and exist on a continuum with the larger tuberous xanthomas. All xanthomas appear histologically similar, with collections of foam cells present within the dermis.2 Foam cells form when serum lipoproteins diffuse through capillary walls, deposit in the skin or tendons, and are scavenged by monocytes.3 Tuberous xanthomas, along with tendinous, eruptive, and planar xanthomas, are the most likely to be associated with hyperlipidemia.4 They may indicate an underlying disorder of lipid metabolism, such as familial hypercholesterolemia.1,3 This is the most common cause of inheritable cardiovascular disease, with a prevalence of approximately 1:250.2 Premature cardiovascular disease risk increases 2 to 4 times in patients with familial hypercholesterolemia and tendinous xanthomas,1 illustrating that recognition of cutaneous lesions can lead to earlier diagnosis and prevention of patient morbidity and mortality.
Juvenile xanthogranuloma typically presents as smooth yellow papules or nodules on the head and neck, with a characteristic “setting-sun” appearance (ie, yellow center with an erythematous halo) on dermoscopy.5 Histologically, juvenile xanthogranulomas are composed of foam cells and a mixed lymphohistiocytic infiltrate with eosinophils within the dermis. Giant cells with a ring of nuclei surrounded by cytoplasm containing lipid vacuoles (called Touton giant cells) are characteristic (Figure 1). In contrast to tuberous xanthomas, juvenile xanthogranulomas often present within the first year of life.6

Keloid scars are more prevalent in patients with skin of color. They are characterized by eosinophilic keloidal collagen with a whorled proliferation of fibroblasts on histology (Figure 2).7 They occur spontaneously or at sites of injury and present as bluish-red or flesh-colored firm papules or nodules.8 In our patient, keloid scars were an unlikely diagnosis due to the lack of trauma and the absence of keloidal collagen on histology.

Necrobiosis lipoidica diabeticorum typically presents as an erythematous, yellow-brown, circular plaque on the anterior lower leg in patients with diabetes mellitus; it rarely occurs in children.9 Microscopy shows palisaded granulomas surrounding necrobiotic collagen arranged horizontally in a layer cake–like fashion (Figure 3).9,10 The etiology of necrobiosis lipoidica diabeticorum currently is unknown, though immune complex deposition may contribute to its pathology. It has been associated with type 1 diabetes mellitus, though severity of the lesions is not associated with extent of glycemic control.10

Rosai-Dorfman disease is an uncommon disorder characterized by a proliferation of histiocytes that most often presents as bilateral cervical lymphadenopathy in children and young adults but rarely can present with cutaneous lesions when extranodal involvement is present.11,12 The cutaneous form most commonly presents as red papules or nodules. On histology, the lesions exhibit a nodular dermal proliferation of histiocytes and smaller lymphocytoid cells with a marbled or starry sky–like appearance on low power (Figure 4). On higher magnification, the characteristic finding of emperipolesis can be seen.11 On immunohistochemistry, the histiocytes stain positively for CD68 and S-100. Although the pathogenesis currently is unknown, evidence of clonality indicates the disease may be related to a neoplastic process.12

The Diagnosis: Tuberous Xanthoma
The skin biopsy revealed a nodular collection of foam cells (quiz image [bottom]). Tuberous xanthoma was the most likely diagnosis based on the patient’s history as well as the clinical and histologic findings. Tuberous xanthomas are flat or elevated nodules in the dermis and subcutaneous tissue, commonly occurring on the skin over the joints.1 Smaller nodules and papules often are referred to as tuberoeruptive xanthomas and exist on a continuum with the larger tuberous xanthomas. All xanthomas appear histologically similar, with collections of foam cells present within the dermis.2 Foam cells form when serum lipoproteins diffuse through capillary walls, deposit in the skin or tendons, and are scavenged by monocytes.3 Tuberous xanthomas, along with tendinous, eruptive, and planar xanthomas, are the most likely to be associated with hyperlipidemia.4 They may indicate an underlying disorder of lipid metabolism, such as familial hypercholesterolemia.1,3 This is the most common cause of inheritable cardiovascular disease, with a prevalence of approximately 1:250.2 Premature cardiovascular disease risk increases 2 to 4 times in patients with familial hypercholesterolemia and tendinous xanthomas,1 illustrating that recognition of cutaneous lesions can lead to earlier diagnosis and prevention of patient morbidity and mortality.
Juvenile xanthogranuloma typically presents as smooth yellow papules or nodules on the head and neck, with a characteristic “setting-sun” appearance (ie, yellow center with an erythematous halo) on dermoscopy.5 Histologically, juvenile xanthogranulomas are composed of foam cells and a mixed lymphohistiocytic infiltrate with eosinophils within the dermis. Giant cells with a ring of nuclei surrounded by cytoplasm containing lipid vacuoles (called Touton giant cells) are characteristic (Figure 1). In contrast to tuberous xanthomas, juvenile xanthogranulomas often present within the first year of life.6

Keloid scars are more prevalent in patients with skin of color. They are characterized by eosinophilic keloidal collagen with a whorled proliferation of fibroblasts on histology (Figure 2).7 They occur spontaneously or at sites of injury and present as bluish-red or flesh-colored firm papules or nodules.8 In our patient, keloid scars were an unlikely diagnosis due to the lack of trauma and the absence of keloidal collagen on histology.

Necrobiosis lipoidica diabeticorum typically presents as an erythematous, yellow-brown, circular plaque on the anterior lower leg in patients with diabetes mellitus; it rarely occurs in children.9 Microscopy shows palisaded granulomas surrounding necrobiotic collagen arranged horizontally in a layer cake–like fashion (Figure 3).9,10 The etiology of necrobiosis lipoidica diabeticorum currently is unknown, though immune complex deposition may contribute to its pathology. It has been associated with type 1 diabetes mellitus, though severity of the lesions is not associated with extent of glycemic control.10

Rosai-Dorfman disease is an uncommon disorder characterized by a proliferation of histiocytes that most often presents as bilateral cervical lymphadenopathy in children and young adults but rarely can present with cutaneous lesions when extranodal involvement is present.11,12 The cutaneous form most commonly presents as red papules or nodules. On histology, the lesions exhibit a nodular dermal proliferation of histiocytes and smaller lymphocytoid cells with a marbled or starry sky–like appearance on low power (Figure 4). On higher magnification, the characteristic finding of emperipolesis can be seen.11 On immunohistochemistry, the histiocytes stain positively for CD68 and S-100. Although the pathogenesis currently is unknown, evidence of clonality indicates the disease may be related to a neoplastic process.12

- Zak A, Zeman M, Slaby A, et al. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158:181-188. doi:10.5507/bp.2014.016
- Ison HE, Clarke SL, Knowles JW. Familial hypercholesterolemia. In: Adam MP, Everman DB, Mirzaa GM, et al, eds. GeneReviews. University of Washington, Seattle; 1993-2022. https://www.ncbi.nlm.nih.gov/books/NBK174884/
- Sathiyakumar V, Jones SR, Martin SS. Xanthomas and lipoprotein disorders. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019.
- Massangale WT. Xanthomas. In: Bolognia JL, Schaffer JV, Cerroni L, et al, eds. Dermatology. Elsevier; 2018:1634-1643.
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. StatPearls. StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/
- Hernández-San Martín MJ, Vargas-Mora P, Aranibar L. Juvenile xanthogranuloma: an entity with a wide clinical spectrum. Actas Dermosifiliogr (Engl Ed). 2020;111:725-733. doi:10.1016/j.ad.2020.07.004
- Lee JY, Yang C, Chao S, et al. Histopathological differential diagnosis of keloid and hypertrophic scar. Am J Dermatopathology. 2004;26:379-384.
- Wolff K, Johnson R, Saavedra AP, et al. Benign neoplasms and hyperplasias. In: Wolff K, Johnson R, Saavedra AP, et al, eds. Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. 8th ed. McGraw Hill; 2017:141-188.
- Bonura C, Frontino G, Rigamonti A, et al. Necrobiosis lipoidica diabeticorum: a pediatric case report. Dermatoendocrinol. 2014;6:E27790. doi:10.4161/derm.27790
- Lepe K, Riley CA, Salazar FJ. Necrobiosis lipoidica. StatPearls. StatPearls Publishing; 2021. https://www-ncbi-nlm-nih-gov.proxy.kumc.edu/books/NBK459318/
- Parrent T, Clark T, Hall D. Cutaneous Rosai-Dorfman disease. Cutis. 2012;90:237-238.
- Bruce-Brand C, Schneider JW, Schubert P. Rosai-Dorfman disease: an overview. J Clin Pathol. 2020;73:697-705. doi:10.1136/jclinpath-2020-206733
- Zak A, Zeman M, Slaby A, et al. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158:181-188. doi:10.5507/bp.2014.016
- Ison HE, Clarke SL, Knowles JW. Familial hypercholesterolemia. In: Adam MP, Everman DB, Mirzaa GM, et al, eds. GeneReviews. University of Washington, Seattle; 1993-2022. https://www.ncbi.nlm.nih.gov/books/NBK174884/
- Sathiyakumar V, Jones SR, Martin SS. Xanthomas and lipoprotein disorders. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw Hill; 2019.
- Massangale WT. Xanthomas. In: Bolognia JL, Schaffer JV, Cerroni L, et al, eds. Dermatology. Elsevier; 2018:1634-1643.
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. StatPearls. StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/
- Hernández-San Martín MJ, Vargas-Mora P, Aranibar L. Juvenile xanthogranuloma: an entity with a wide clinical spectrum. Actas Dermosifiliogr (Engl Ed). 2020;111:725-733. doi:10.1016/j.ad.2020.07.004
- Lee JY, Yang C, Chao S, et al. Histopathological differential diagnosis of keloid and hypertrophic scar. Am J Dermatopathology. 2004;26:379-384.
- Wolff K, Johnson R, Saavedra AP, et al. Benign neoplasms and hyperplasias. In: Wolff K, Johnson R, Saavedra AP, et al, eds. Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. 8th ed. McGraw Hill; 2017:141-188.
- Bonura C, Frontino G, Rigamonti A, et al. Necrobiosis lipoidica diabeticorum: a pediatric case report. Dermatoendocrinol. 2014;6:E27790. doi:10.4161/derm.27790
- Lepe K, Riley CA, Salazar FJ. Necrobiosis lipoidica. StatPearls. StatPearls Publishing; 2021. https://www-ncbi-nlm-nih-gov.proxy.kumc.edu/books/NBK459318/
- Parrent T, Clark T, Hall D. Cutaneous Rosai-Dorfman disease. Cutis. 2012;90:237-238.
- Bruce-Brand C, Schneider JW, Schubert P. Rosai-Dorfman disease: an overview. J Clin Pathol. 2020;73:697-705. doi:10.1136/jclinpath-2020-206733
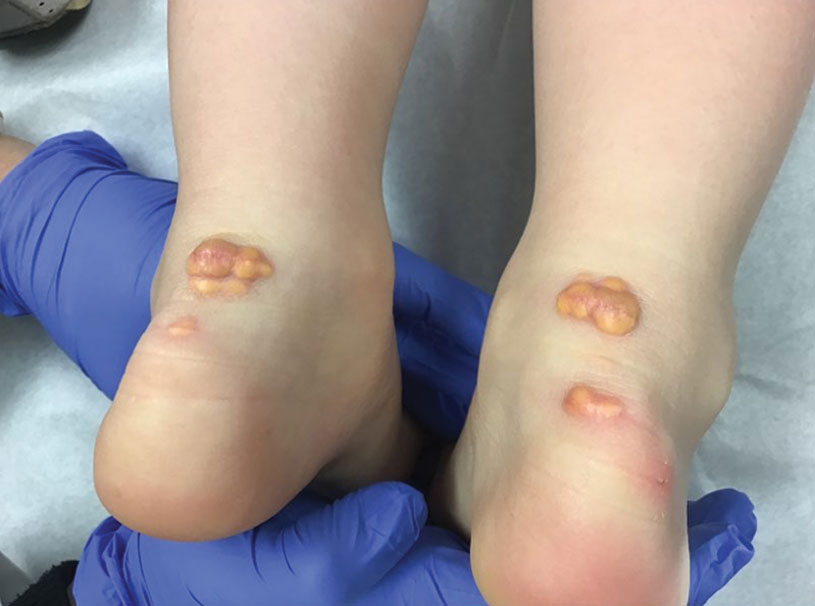
A 3-year-old girl presented with raised, firm, enlarging, asymptomatic, well-defined, subcutaneous papules, plaques, and nodules on the hands, knees, and posterior ankles of 1 year’s duration. The patient’s mother stated that the lesions began on the ankles (top), and she initially believed them to be due to friction from the child’s shoes until the more recent involvement of the knees and hands. The patient’s father, paternal grandfather, and paternal great-grandfather had a history of elevated cholesterol levels. A shave biopsy was performed (bottom).
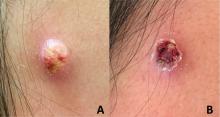
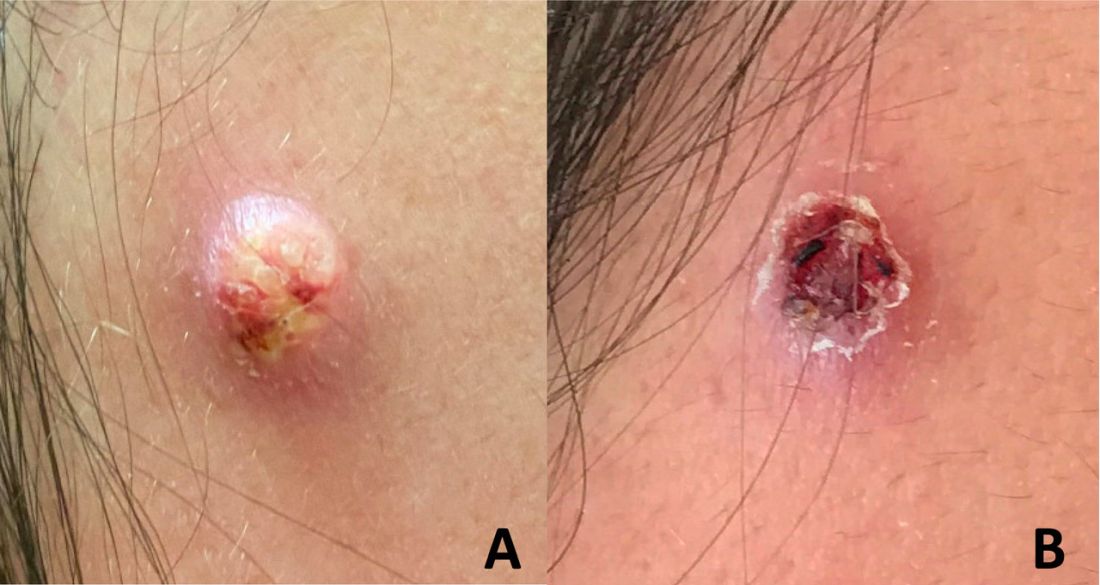
An adolescent male presents with an eroded bump on the temple
The correct answer is (D), molluscum contagiosum. Upon surgical excision, the pathology indicated the lesion was consistent with molluscum contagiosum.
Molluscum contagiosum is a benign skin disorder caused by a pox virus and is frequently seen in children. This disease is transmitted primarily through direct skin contact with an infected individual.1 Contaminated fomites have been suggested as another source of infection.2 The typical lesion appears dome-shaped, round, and pinkish-purple in color.1 The incubation period ranges from 2 weeks to 6 months and is typically self-limited in immunocompetent hosts; however, in immunocompromised persons, molluscum contagiosum lesions may present atypically such that they are larger in size and/or resemble malignancies, such as basal cell carcinoma or keratoacanthoma (for single lesions), or other infectious diseases, such as cryptococcosis and histoplasmosis (for more numerous lesions).3,4 A giant atypical molluscum contagiosum is rarely seen in healthy individuals.
What’s on the differential?
The recent episode of bleeding raises concern for other neoplastic processes of the skin including squamous cell carcinoma or basal cell carcinoma as well as cutaneous metastatic rhabdoid tumor, given the patient’s history.
Eruptive keratoacanthomas are also reported in patients taking nivolumab, an anti-PD-1 immunotherapy, which the patient has received for treatment of his recurrent metastatic rhabdoid tumor.5 More common entities such as a pyogenic granuloma or verruca are also included on the differential. The initial presentation of the lesion, however, is more consistent with the pearly umbilicated papules associated with molluscum contagiosum.
Comments from Dr. Eichenfield
This is a very hard diagnosis to make with the clinical findings and history.
Molluscum contagiosum infections are common, but with this patient’s medical history, biopsy and excision with pathologic examination was an appropriate approach to make a certain diagnosis.
Ms. Moyal is a research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego.
References
1. Brown J et al. Int J Dermatol. 2006 Feb;45(2):93-9.
2. Hanson D and Diven DG. Dermatol Online J. 2003 Mar;9(2).
3. Badri T and Gandhi GR. Molluscum contagiosum. 2022. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing.
4. Schwartz JJ and Myskowski PL. J Am Acad Dermatol. 1992 Oct 1;27(4):583-8.
5. Antonov NK et al. JAAD Case Rep. 2019 Apr 5;5(4):342-5.
The correct answer is (D), molluscum contagiosum. Upon surgical excision, the pathology indicated the lesion was consistent with molluscum contagiosum.
Molluscum contagiosum is a benign skin disorder caused by a pox virus and is frequently seen in children. This disease is transmitted primarily through direct skin contact with an infected individual.1 Contaminated fomites have been suggested as another source of infection.2 The typical lesion appears dome-shaped, round, and pinkish-purple in color.1 The incubation period ranges from 2 weeks to 6 months and is typically self-limited in immunocompetent hosts; however, in immunocompromised persons, molluscum contagiosum lesions may present atypically such that they are larger in size and/or resemble malignancies, such as basal cell carcinoma or keratoacanthoma (for single lesions), or other infectious diseases, such as cryptococcosis and histoplasmosis (for more numerous lesions).3,4 A giant atypical molluscum contagiosum is rarely seen in healthy individuals.
What’s on the differential?
The recent episode of bleeding raises concern for other neoplastic processes of the skin including squamous cell carcinoma or basal cell carcinoma as well as cutaneous metastatic rhabdoid tumor, given the patient’s history.
Eruptive keratoacanthomas are also reported in patients taking nivolumab, an anti-PD-1 immunotherapy, which the patient has received for treatment of his recurrent metastatic rhabdoid tumor.5 More common entities such as a pyogenic granuloma or verruca are also included on the differential. The initial presentation of the lesion, however, is more consistent with the pearly umbilicated papules associated with molluscum contagiosum.
Comments from Dr. Eichenfield
This is a very hard diagnosis to make with the clinical findings and history.
Molluscum contagiosum infections are common, but with this patient’s medical history, biopsy and excision with pathologic examination was an appropriate approach to make a certain diagnosis.
Ms. Moyal is a research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego.
References
1. Brown J et al. Int J Dermatol. 2006 Feb;45(2):93-9.
2. Hanson D and Diven DG. Dermatol Online J. 2003 Mar;9(2).
3. Badri T and Gandhi GR. Molluscum contagiosum. 2022. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing.
4. Schwartz JJ and Myskowski PL. J Am Acad Dermatol. 1992 Oct 1;27(4):583-8.
5. Antonov NK et al. JAAD Case Rep. 2019 Apr 5;5(4):342-5.
The correct answer is (D), molluscum contagiosum. Upon surgical excision, the pathology indicated the lesion was consistent with molluscum contagiosum.
Molluscum contagiosum is a benign skin disorder caused by a pox virus and is frequently seen in children. This disease is transmitted primarily through direct skin contact with an infected individual.1 Contaminated fomites have been suggested as another source of infection.2 The typical lesion appears dome-shaped, round, and pinkish-purple in color.1 The incubation period ranges from 2 weeks to 6 months and is typically self-limited in immunocompetent hosts; however, in immunocompromised persons, molluscum contagiosum lesions may present atypically such that they are larger in size and/or resemble malignancies, such as basal cell carcinoma or keratoacanthoma (for single lesions), or other infectious diseases, such as cryptococcosis and histoplasmosis (for more numerous lesions).3,4 A giant atypical molluscum contagiosum is rarely seen in healthy individuals.
What’s on the differential?
The recent episode of bleeding raises concern for other neoplastic processes of the skin including squamous cell carcinoma or basal cell carcinoma as well as cutaneous metastatic rhabdoid tumor, given the patient’s history.
Eruptive keratoacanthomas are also reported in patients taking nivolumab, an anti-PD-1 immunotherapy, which the patient has received for treatment of his recurrent metastatic rhabdoid tumor.5 More common entities such as a pyogenic granuloma or verruca are also included on the differential. The initial presentation of the lesion, however, is more consistent with the pearly umbilicated papules associated with molluscum contagiosum.
Comments from Dr. Eichenfield
This is a very hard diagnosis to make with the clinical findings and history.
Molluscum contagiosum infections are common, but with this patient’s medical history, biopsy and excision with pathologic examination was an appropriate approach to make a certain diagnosis.
Ms. Moyal is a research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego.
References
1. Brown J et al. Int J Dermatol. 2006 Feb;45(2):93-9.
2. Hanson D and Diven DG. Dermatol Online J. 2003 Mar;9(2).
3. Badri T and Gandhi GR. Molluscum contagiosum. 2022. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing.
4. Schwartz JJ and Myskowski PL. J Am Acad Dermatol. 1992 Oct 1;27(4):583-8.
5. Antonov NK et al. JAAD Case Rep. 2019 Apr 5;5(4):342-5.
Breaking the itch-scratch cycle with mindfulness
Apple A. Bodemer, MD, a dermatologist at the University of Wisconsin, Madison, teaches patients how to breathe mindfully. So does Kathy Farah, MD, an integrative family physician who practices in Roberts, Wis.
, they said at the annual Integrative Dermatology Symposium.
“As with any integrative modality, if it’s safe and effective, then let’s use it,” Dr. Farah said in a presentation on the mind-body approach to pain and itch.
“A breathwork session can literally take 1 minute,” said Dr. Bodemer, associate professor of dermatology at the University of Wisconsin and director of an integrative dermatology clinic. Dr. Bodemer, who completed a fellowship in integrative medicine at the Andrew Weil Center for Integrative Medicine at the University of Arizona and sits on the American Board of Integrative Medicine, spoke on a mindfulness panel at the meeting.
Her favorite breathing practice is the “4-7-8” breath taught by Andrew Weil, MD, founder and director of the center. This involves inhaling through the nose for a count of 4, holding for 7, and exhaling through the mouth for a count of 8. “It doesn’t matter how slow or fast, it’s the tempo that matters ... On exhale, squeeze your abs in to engage your core and get air out of your lungs as much as you can,” she said, advising a cycle of three at a time.
A technique known as “square breathing” (breath in 4, hold for 4, breath out for 4, hold for 4) is another helpful technique to “reset the nervous system” said Dr. Farah, who worked for many years in a children’s hospital. With children, she said, “I often do five finger breathing.”
For five finger breathing, the children spread their fingers apart in front of them or on the ground and use the pointer finger of the opposite hand to trace each finger, inhaling while tracing upward, and exhaling while tracing down.
Dr. Farah, associate clinical director of The Center for Mind-Body Medicine in Washington, DC, said her commitment to mindfulness was influenced by a “seminal” study published over 20 years ago showing that patients with moderate to severe psoriasis who used a meditation-based, audiotape-guided stress reduction intervention during phototherapy sessions had more rapid resolution of psoriatic lesions than did patients who didn’t use the mindfulness exercise.
Among more recent findings: A cross-sectional study of 120 adult dermatology patients, published in the British Journal of Dermatology in 2016, assessed skin shame, social anxiety, anxiety, depression, dermatological quality of life, and levels of mindfulness, and found that higher levels of mindfulness were associated with lower levels of psychosocial distress.
Another cross-sectional questionnaire study looked at mindfulness and “itch catastrophizing” in 155 adult patients with atopic dermatitis. Higher levels of a specific facet of mindfulness termed “acting with awareness” were associated with lower levels of itch catastrophizing, the researchers found. “Catastrophizing is a negative way of thinking, this itching will never stop,” Dr. Farah explained. The study shows that “mindfulness can actually help reduce some of the automatic scratching and response to itch. So it’s a great adjunct to pharmaceuticals.”
Affirmations – phrases and statements that are repeated to oneself to help challenge negative thoughts – can also help reverse itch catastrophizing. Statements such as “I can breathe through this feeling of itching,” or “I can move to feel comfortable and relaxed” encourage positive change, she said.
“I teach [mindfulness skills like breathing] a lot, without any expectations. I’ll say ‘give it a try and see what you think.’ If patients feel even a micron better, then they’re invested” and can then find numerous tools online, Dr. Farah said. “Can I do this [in a busy schedule] with every patient? Absolutely not. But can I do it with every 10th patient? Maybe.”
Dr. Bodemer’s experience has shown her that “breathing with your patient builds rapport,” she said. “There’s something very powerful in that in terms of building trust. ... I’ll just do it [during a visit, to show them] and almost always, patients start breathing with me, with an invitation or without.”
For her own health, 4-7-8 breathing has “been a gateway to meditation and deeper practices,” she said. “But even without going very deep, it has a long history of being able to modulate the stress response. It’s the parasympathetic-sympathetic rebalancing I’m interested in.”
Mindful breathing and other mind-body practices also can be helpful for parents of children with eczema, she and Dr. Farah said.
Dr. Bodemer and Dr. Farah reported no financial relationships to disclose.
Apple A. Bodemer, MD, a dermatologist at the University of Wisconsin, Madison, teaches patients how to breathe mindfully. So does Kathy Farah, MD, an integrative family physician who practices in Roberts, Wis.
, they said at the annual Integrative Dermatology Symposium.
“As with any integrative modality, if it’s safe and effective, then let’s use it,” Dr. Farah said in a presentation on the mind-body approach to pain and itch.
“A breathwork session can literally take 1 minute,” said Dr. Bodemer, associate professor of dermatology at the University of Wisconsin and director of an integrative dermatology clinic. Dr. Bodemer, who completed a fellowship in integrative medicine at the Andrew Weil Center for Integrative Medicine at the University of Arizona and sits on the American Board of Integrative Medicine, spoke on a mindfulness panel at the meeting.
Her favorite breathing practice is the “4-7-8” breath taught by Andrew Weil, MD, founder and director of the center. This involves inhaling through the nose for a count of 4, holding for 7, and exhaling through the mouth for a count of 8. “It doesn’t matter how slow or fast, it’s the tempo that matters ... On exhale, squeeze your abs in to engage your core and get air out of your lungs as much as you can,” she said, advising a cycle of three at a time.
A technique known as “square breathing” (breath in 4, hold for 4, breath out for 4, hold for 4) is another helpful technique to “reset the nervous system” said Dr. Farah, who worked for many years in a children’s hospital. With children, she said, “I often do five finger breathing.”
For five finger breathing, the children spread their fingers apart in front of them or on the ground and use the pointer finger of the opposite hand to trace each finger, inhaling while tracing upward, and exhaling while tracing down.
Dr. Farah, associate clinical director of The Center for Mind-Body Medicine in Washington, DC, said her commitment to mindfulness was influenced by a “seminal” study published over 20 years ago showing that patients with moderate to severe psoriasis who used a meditation-based, audiotape-guided stress reduction intervention during phototherapy sessions had more rapid resolution of psoriatic lesions than did patients who didn’t use the mindfulness exercise.
Among more recent findings: A cross-sectional study of 120 adult dermatology patients, published in the British Journal of Dermatology in 2016, assessed skin shame, social anxiety, anxiety, depression, dermatological quality of life, and levels of mindfulness, and found that higher levels of mindfulness were associated with lower levels of psychosocial distress.
Another cross-sectional questionnaire study looked at mindfulness and “itch catastrophizing” in 155 adult patients with atopic dermatitis. Higher levels of a specific facet of mindfulness termed “acting with awareness” were associated with lower levels of itch catastrophizing, the researchers found. “Catastrophizing is a negative way of thinking, this itching will never stop,” Dr. Farah explained. The study shows that “mindfulness can actually help reduce some of the automatic scratching and response to itch. So it’s a great adjunct to pharmaceuticals.”
Affirmations – phrases and statements that are repeated to oneself to help challenge negative thoughts – can also help reverse itch catastrophizing. Statements such as “I can breathe through this feeling of itching,” or “I can move to feel comfortable and relaxed” encourage positive change, she said.
“I teach [mindfulness skills like breathing] a lot, without any expectations. I’ll say ‘give it a try and see what you think.’ If patients feel even a micron better, then they’re invested” and can then find numerous tools online, Dr. Farah said. “Can I do this [in a busy schedule] with every patient? Absolutely not. But can I do it with every 10th patient? Maybe.”
Dr. Bodemer’s experience has shown her that “breathing with your patient builds rapport,” she said. “There’s something very powerful in that in terms of building trust. ... I’ll just do it [during a visit, to show them] and almost always, patients start breathing with me, with an invitation or without.”
For her own health, 4-7-8 breathing has “been a gateway to meditation and deeper practices,” she said. “But even without going very deep, it has a long history of being able to modulate the stress response. It’s the parasympathetic-sympathetic rebalancing I’m interested in.”
Mindful breathing and other mind-body practices also can be helpful for parents of children with eczema, she and Dr. Farah said.
Dr. Bodemer and Dr. Farah reported no financial relationships to disclose.
Apple A. Bodemer, MD, a dermatologist at the University of Wisconsin, Madison, teaches patients how to breathe mindfully. So does Kathy Farah, MD, an integrative family physician who practices in Roberts, Wis.
, they said at the annual Integrative Dermatology Symposium.
“As with any integrative modality, if it’s safe and effective, then let’s use it,” Dr. Farah said in a presentation on the mind-body approach to pain and itch.
“A breathwork session can literally take 1 minute,” said Dr. Bodemer, associate professor of dermatology at the University of Wisconsin and director of an integrative dermatology clinic. Dr. Bodemer, who completed a fellowship in integrative medicine at the Andrew Weil Center for Integrative Medicine at the University of Arizona and sits on the American Board of Integrative Medicine, spoke on a mindfulness panel at the meeting.
Her favorite breathing practice is the “4-7-8” breath taught by Andrew Weil, MD, founder and director of the center. This involves inhaling through the nose for a count of 4, holding for 7, and exhaling through the mouth for a count of 8. “It doesn’t matter how slow or fast, it’s the tempo that matters ... On exhale, squeeze your abs in to engage your core and get air out of your lungs as much as you can,” she said, advising a cycle of three at a time.
A technique known as “square breathing” (breath in 4, hold for 4, breath out for 4, hold for 4) is another helpful technique to “reset the nervous system” said Dr. Farah, who worked for many years in a children’s hospital. With children, she said, “I often do five finger breathing.”
For five finger breathing, the children spread their fingers apart in front of them or on the ground and use the pointer finger of the opposite hand to trace each finger, inhaling while tracing upward, and exhaling while tracing down.
Dr. Farah, associate clinical director of The Center for Mind-Body Medicine in Washington, DC, said her commitment to mindfulness was influenced by a “seminal” study published over 20 years ago showing that patients with moderate to severe psoriasis who used a meditation-based, audiotape-guided stress reduction intervention during phototherapy sessions had more rapid resolution of psoriatic lesions than did patients who didn’t use the mindfulness exercise.
Among more recent findings: A cross-sectional study of 120 adult dermatology patients, published in the British Journal of Dermatology in 2016, assessed skin shame, social anxiety, anxiety, depression, dermatological quality of life, and levels of mindfulness, and found that higher levels of mindfulness were associated with lower levels of psychosocial distress.
Another cross-sectional questionnaire study looked at mindfulness and “itch catastrophizing” in 155 adult patients with atopic dermatitis. Higher levels of a specific facet of mindfulness termed “acting with awareness” were associated with lower levels of itch catastrophizing, the researchers found. “Catastrophizing is a negative way of thinking, this itching will never stop,” Dr. Farah explained. The study shows that “mindfulness can actually help reduce some of the automatic scratching and response to itch. So it’s a great adjunct to pharmaceuticals.”
Affirmations – phrases and statements that are repeated to oneself to help challenge negative thoughts – can also help reverse itch catastrophizing. Statements such as “I can breathe through this feeling of itching,” or “I can move to feel comfortable and relaxed” encourage positive change, she said.
“I teach [mindfulness skills like breathing] a lot, without any expectations. I’ll say ‘give it a try and see what you think.’ If patients feel even a micron better, then they’re invested” and can then find numerous tools online, Dr. Farah said. “Can I do this [in a busy schedule] with every patient? Absolutely not. But can I do it with every 10th patient? Maybe.”
Dr. Bodemer’s experience has shown her that “breathing with your patient builds rapport,” she said. “There’s something very powerful in that in terms of building trust. ... I’ll just do it [during a visit, to show them] and almost always, patients start breathing with me, with an invitation or without.”
For her own health, 4-7-8 breathing has “been a gateway to meditation and deeper practices,” she said. “But even without going very deep, it has a long history of being able to modulate the stress response. It’s the parasympathetic-sympathetic rebalancing I’m interested in.”
Mindful breathing and other mind-body practices also can be helpful for parents of children with eczema, she and Dr. Farah said.
Dr. Bodemer and Dr. Farah reported no financial relationships to disclose.
FROM IDS 2022
Treating deadly disease in utero called ‘revolutionary’ advance
The successful treatment of Pompe disease in utero for the first time may be the start of a new chapter for fetal therapy, researchers said.
A report published online in the New England Journal of Medicine describes in utero enzyme-replacement therapy (ERT) for infantile-onset Pompe disease.
The patient, now a toddler, is thriving, according to the researchers. Her parents previously had children with the same disorder who died.
“This treatment expands the repertoire of fetal therapies in a new direction,” Tippi MacKenzie, MD, a pediatric surgeon with University of California, San Francisco, Benioff Children’s Hospitals and a coauthor of the report, said in a news release. “As new treatments become available for children with genetic conditions, we are developing protocols to apply them before birth.”
Dr. MacKenzie codirects the University of California, San Francisco’s center for maternal-fetal precision medicine and directs the Eli and Edythe Broad Center of Regeneration Medicine and Stem Cell Research.
Pompe disease is caused by mutations in a gene that makes acid alpha-glucosidase. With limited amounts of this enzyme, dangerous amounts of glycogen accumulate in the body. Babies with infantile-onset disease typically have enlarged hearts and die by age 2 years.
The condition, which occurs in an estimated 1 in 40,000 births, is one of several early-onset lysosomal storage disorders. Patients with these diseases “are ideal candidates for prenatal therapy because organ damage starts in utero,” the researchers said.
Newborn screening can lead to early initiation of treatment with recombinant enzymes, “but this strategy does not completely prevent irreversible organ damage,” the authors said.
The patient in the new report received six prenatal ERT treatments at the Ottawa Hospital and is receiving postnatal enzyme therapy at CHEO, a pediatric hospital and research center in Ottawa.
Investigators administered alglucosidase alfa through the umbilical vein. They delivered the first infusion to the fetus at 24 weeks 5 days of gestation. They continued providing infusions at 2-week intervals through 34 weeks 5 days of gestation.
She is doing well at age 16 months, with normal cardiac and motor function, and is meeting developmental milestones, according to the news release.
The successful treatment involved collaboration among the University of California, San Francisco, where researchers are conducting a clinical trial of this treatment approach; CHEO and the Ottawa Hospital; and Duke University, Durham, N.C.
Under normal circumstances, the patient’s family would have traveled to Benioff Children’s Hospitals fetal treatment center to participate in the clinical trial, but COVID-19 restrictions led the researchers to deliver the therapy to Ottawa as part of the trial.
The University of California, San Francisco, has received U.S. Food and Drug Administration approval to treat Pompe disease and several other lysosomal storage disorders in utero as part of a phase 1 clinical trial with 10 patients. The other diseases are mucopolysaccharidosis types 1, 2, 4a, 6, and 7; Gaucher disease types 2 and 3; and Wolman disease.
Patients with Pompe disease might typically be diagnosed clinically at age 3-6 months, said study coauthor Paul Harmatz, MD, with the University of California, San Francisco. With newborn screening, the disease might be diagnosed at 1 week. But intervening before birth may be optimal, Dr. Harmatz said.
Fetal treatment appears to be “revolutionary at this point,” Dr. Harmatz said.
The research was supported by a grant from the National Institutes of Health. Sanofi Genzyme provided the enzyme for the patient.
A version of this article first appeared on Medscape.com.
The successful treatment of Pompe disease in utero for the first time may be the start of a new chapter for fetal therapy, researchers said.
A report published online in the New England Journal of Medicine describes in utero enzyme-replacement therapy (ERT) for infantile-onset Pompe disease.
The patient, now a toddler, is thriving, according to the researchers. Her parents previously had children with the same disorder who died.
“This treatment expands the repertoire of fetal therapies in a new direction,” Tippi MacKenzie, MD, a pediatric surgeon with University of California, San Francisco, Benioff Children’s Hospitals and a coauthor of the report, said in a news release. “As new treatments become available for children with genetic conditions, we are developing protocols to apply them before birth.”
Dr. MacKenzie codirects the University of California, San Francisco’s center for maternal-fetal precision medicine and directs the Eli and Edythe Broad Center of Regeneration Medicine and Stem Cell Research.
Pompe disease is caused by mutations in a gene that makes acid alpha-glucosidase. With limited amounts of this enzyme, dangerous amounts of glycogen accumulate in the body. Babies with infantile-onset disease typically have enlarged hearts and die by age 2 years.
The condition, which occurs in an estimated 1 in 40,000 births, is one of several early-onset lysosomal storage disorders. Patients with these diseases “are ideal candidates for prenatal therapy because organ damage starts in utero,” the researchers said.
Newborn screening can lead to early initiation of treatment with recombinant enzymes, “but this strategy does not completely prevent irreversible organ damage,” the authors said.
The patient in the new report received six prenatal ERT treatments at the Ottawa Hospital and is receiving postnatal enzyme therapy at CHEO, a pediatric hospital and research center in Ottawa.
Investigators administered alglucosidase alfa through the umbilical vein. They delivered the first infusion to the fetus at 24 weeks 5 days of gestation. They continued providing infusions at 2-week intervals through 34 weeks 5 days of gestation.
She is doing well at age 16 months, with normal cardiac and motor function, and is meeting developmental milestones, according to the news release.
The successful treatment involved collaboration among the University of California, San Francisco, where researchers are conducting a clinical trial of this treatment approach; CHEO and the Ottawa Hospital; and Duke University, Durham, N.C.
Under normal circumstances, the patient’s family would have traveled to Benioff Children’s Hospitals fetal treatment center to participate in the clinical trial, but COVID-19 restrictions led the researchers to deliver the therapy to Ottawa as part of the trial.
The University of California, San Francisco, has received U.S. Food and Drug Administration approval to treat Pompe disease and several other lysosomal storage disorders in utero as part of a phase 1 clinical trial with 10 patients. The other diseases are mucopolysaccharidosis types 1, 2, 4a, 6, and 7; Gaucher disease types 2 and 3; and Wolman disease.
Patients with Pompe disease might typically be diagnosed clinically at age 3-6 months, said study coauthor Paul Harmatz, MD, with the University of California, San Francisco. With newborn screening, the disease might be diagnosed at 1 week. But intervening before birth may be optimal, Dr. Harmatz said.
Fetal treatment appears to be “revolutionary at this point,” Dr. Harmatz said.
The research was supported by a grant from the National Institutes of Health. Sanofi Genzyme provided the enzyme for the patient.
A version of this article first appeared on Medscape.com.
The successful treatment of Pompe disease in utero for the first time may be the start of a new chapter for fetal therapy, researchers said.
A report published online in the New England Journal of Medicine describes in utero enzyme-replacement therapy (ERT) for infantile-onset Pompe disease.
The patient, now a toddler, is thriving, according to the researchers. Her parents previously had children with the same disorder who died.
“This treatment expands the repertoire of fetal therapies in a new direction,” Tippi MacKenzie, MD, a pediatric surgeon with University of California, San Francisco, Benioff Children’s Hospitals and a coauthor of the report, said in a news release. “As new treatments become available for children with genetic conditions, we are developing protocols to apply them before birth.”
Dr. MacKenzie codirects the University of California, San Francisco’s center for maternal-fetal precision medicine and directs the Eli and Edythe Broad Center of Regeneration Medicine and Stem Cell Research.
Pompe disease is caused by mutations in a gene that makes acid alpha-glucosidase. With limited amounts of this enzyme, dangerous amounts of glycogen accumulate in the body. Babies with infantile-onset disease typically have enlarged hearts and die by age 2 years.
The condition, which occurs in an estimated 1 in 40,000 births, is one of several early-onset lysosomal storage disorders. Patients with these diseases “are ideal candidates for prenatal therapy because organ damage starts in utero,” the researchers said.
Newborn screening can lead to early initiation of treatment with recombinant enzymes, “but this strategy does not completely prevent irreversible organ damage,” the authors said.
The patient in the new report received six prenatal ERT treatments at the Ottawa Hospital and is receiving postnatal enzyme therapy at CHEO, a pediatric hospital and research center in Ottawa.
Investigators administered alglucosidase alfa through the umbilical vein. They delivered the first infusion to the fetus at 24 weeks 5 days of gestation. They continued providing infusions at 2-week intervals through 34 weeks 5 days of gestation.
She is doing well at age 16 months, with normal cardiac and motor function, and is meeting developmental milestones, according to the news release.
The successful treatment involved collaboration among the University of California, San Francisco, where researchers are conducting a clinical trial of this treatment approach; CHEO and the Ottawa Hospital; and Duke University, Durham, N.C.
Under normal circumstances, the patient’s family would have traveled to Benioff Children’s Hospitals fetal treatment center to participate in the clinical trial, but COVID-19 restrictions led the researchers to deliver the therapy to Ottawa as part of the trial.
The University of California, San Francisco, has received U.S. Food and Drug Administration approval to treat Pompe disease and several other lysosomal storage disorders in utero as part of a phase 1 clinical trial with 10 patients. The other diseases are mucopolysaccharidosis types 1, 2, 4a, 6, and 7; Gaucher disease types 2 and 3; and Wolman disease.
Patients with Pompe disease might typically be diagnosed clinically at age 3-6 months, said study coauthor Paul Harmatz, MD, with the University of California, San Francisco. With newborn screening, the disease might be diagnosed at 1 week. But intervening before birth may be optimal, Dr. Harmatz said.
Fetal treatment appears to be “revolutionary at this point,” Dr. Harmatz said.
The research was supported by a grant from the National Institutes of Health. Sanofi Genzyme provided the enzyme for the patient.
A version of this article first appeared on Medscape.com.
More children should be getting flu vaccines
Cold and flu season came early in 2022.
On Nov. 4, 2022, the Centers for Disease Control and Prevention issued a Health Alert Network Health Advisory about early, elevated respiratory disease incidence caused by multiple viruses other than SARS-CoV-2.
Interseasonal spread of respiratory syncytial virus has continued in 2022, with RSV-associated hospitalizations increasing in the late spring and continuing throughout the summer and into the fall. In October, some regions of the country were seeing RSV activity near the peak seasonal levels typically observed in December and January.
Cases of severe respiratory infection in children who tested positive for rhinovirus or enterovirus spiked in August; further testing confirmed the presence of EV-D68 in some children. Rhinovirus and enterovirus continue to circulate and are isolated in hospitalized children with respiratory illness.
In some parts of the country, influenza cases have rapidly increased ahead of what we normally anticipate. According to preliminary estimates from the CDC, between Oct. 1 and Oct. 22, 880,000 people were sickened with flu, 420,000 people visited a health care provider for flu illness, and 6,900 people were hospitalized for flu. The cumulative hospitalization rate is higher than observed at this time of year in every previous flu season since 2010-2011. Hospitalization rates are highest in children aged 0-4 years and adults 65 years and older.
Of course, this report came as no surprise to pediatric health care providers. Many children’s hospitals had been operating at or over capacity for weeks. While a systematic assessment of the surge on children’s hospitals has not been published, anecdotally, hospitals from around the country have described record emergency department visits and inpatient census numbers. Some have set up tents or other temporary facilities to see ambulatory patients and have canceled elective surgeries because of a lack of beds.
There is no quick or easy solution to stem the tide of RSV-related or enterovirus/rhinovirus admissions, but many flu-related hospitalizations are vaccine preventable. Unfortunately, too few children are receiving influenza vaccine. As of the week ending Oct. 15, only about 22.1% of eligible children had been immunized. The American Academy of Pediatrics and the CDC recommend that all children are vaccinated, preferably by the end of October so they have time to develop immunity before influenza starts circulating. As it stands now, the majority of the nation’s children are facing a flu season without the benefits of vaccine.
There is still time to take steps to prevent this flu season from becoming one of the worst in recent memory. A strong provider recommendation for influenza vaccine is consistently associated with higher rates of vaccine acceptance. We need to recommend influenza vaccine to all eligible patients at every visit and in every setting. It will help if we can say it like we mean it. Some of us are tired of debating the merits of COVID-19 vaccine with families and may be leery of additional debates about flu. Some of us may just be tired, as many practices have already expanded office hours to care for the influx of kids with respiratory illness. On the heels of two atypical flu seasons, a few of us may be quietly complacent about the importance of flu vaccines for children.
Anyone in need of a little motivation should check out a paper recently published in Clinical Infectious Diseases that reinforces the value of flu vaccine, even in a year when there is a poor match between the vaccine and circulating viruses.
The 2019-2020 flu season was a bad flu season for children. Two antigenically drifted influenza viruses predominated and cases of influenza soared, resulting in the largest influenza epidemic in children in the United States since 1992. Pediatric Intensive Care Influenza Study investigators used a test-negative design to estimate the effectiveness of influenza vaccine in preventing critical and life-threatening influenza in children during that season. The good news: vaccination reduced the risk of critical influenza by 78% against H1N1pdm09 viruses that were well-matched to vaccine and by 47% against mismatched viruses. Vaccination was estimated to be 75% protective against antigenically drifted B-Victoria viruses. Overall vaccine effectiveness against critical illness from any influenza virus was 63% (95% confidence interval, 38%-78%).
While it might be tempting to attribute suboptimal immunization rates to vaccine hesitancy, ready availability remains an issue for some families. We need to eliminate barriers to access. While the AAP continues to emphasize immunization in the medical home, especially for the youngest infants, the 2022 policy statement suggests that vaccinating children in schools, pharmacies, and other nontraditional settings could improve immunization rates. To the extent feasible, we need to work with partners to support community-based initiatives and promote these to families who struggle to make it into the office.
Improving access is just one potential way to reduce health disparities related to influenza and influenza vaccination. Over 10 influenza seasons, higher rates of influenza-associated hospitalizations and intensive care unit admissions were observed in Black, Hispanic, and American Indian/Alaska Native people. These disparities were highest in children aged younger than 4 years and influenza-associated in-hospital deaths were three- to fourfold higher in Black, Hispanic, and Asian/Pacific Islander children, compared with White children. The reason for the disparities isn’t completely clear but increasing immunization rates may be part of the solution. During the 2020-2021 influenza season, flu immunization rates in Black children (51.6%) were lower than those seen in White (57.4%) and Hispanic children (58.9%).
The AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023, highlight a variety of evidence-based strategies to increase influenza immunization rates. These may provide a little inspiration for clinicians looking to try a new approach. If you wish to share your experience with increasing influenza immunization rates in your practice setting, please email me at [email protected].
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead.
Cold and flu season came early in 2022.
On Nov. 4, 2022, the Centers for Disease Control and Prevention issued a Health Alert Network Health Advisory about early, elevated respiratory disease incidence caused by multiple viruses other than SARS-CoV-2.
Interseasonal spread of respiratory syncytial virus has continued in 2022, with RSV-associated hospitalizations increasing in the late spring and continuing throughout the summer and into the fall. In October, some regions of the country were seeing RSV activity near the peak seasonal levels typically observed in December and January.
Cases of severe respiratory infection in children who tested positive for rhinovirus or enterovirus spiked in August; further testing confirmed the presence of EV-D68 in some children. Rhinovirus and enterovirus continue to circulate and are isolated in hospitalized children with respiratory illness.
In some parts of the country, influenza cases have rapidly increased ahead of what we normally anticipate. According to preliminary estimates from the CDC, between Oct. 1 and Oct. 22, 880,000 people were sickened with flu, 420,000 people visited a health care provider for flu illness, and 6,900 people were hospitalized for flu. The cumulative hospitalization rate is higher than observed at this time of year in every previous flu season since 2010-2011. Hospitalization rates are highest in children aged 0-4 years and adults 65 years and older.
Of course, this report came as no surprise to pediatric health care providers. Many children’s hospitals had been operating at or over capacity for weeks. While a systematic assessment of the surge on children’s hospitals has not been published, anecdotally, hospitals from around the country have described record emergency department visits and inpatient census numbers. Some have set up tents or other temporary facilities to see ambulatory patients and have canceled elective surgeries because of a lack of beds.
There is no quick or easy solution to stem the tide of RSV-related or enterovirus/rhinovirus admissions, but many flu-related hospitalizations are vaccine preventable. Unfortunately, too few children are receiving influenza vaccine. As of the week ending Oct. 15, only about 22.1% of eligible children had been immunized. The American Academy of Pediatrics and the CDC recommend that all children are vaccinated, preferably by the end of October so they have time to develop immunity before influenza starts circulating. As it stands now, the majority of the nation’s children are facing a flu season without the benefits of vaccine.
There is still time to take steps to prevent this flu season from becoming one of the worst in recent memory. A strong provider recommendation for influenza vaccine is consistently associated with higher rates of vaccine acceptance. We need to recommend influenza vaccine to all eligible patients at every visit and in every setting. It will help if we can say it like we mean it. Some of us are tired of debating the merits of COVID-19 vaccine with families and may be leery of additional debates about flu. Some of us may just be tired, as many practices have already expanded office hours to care for the influx of kids with respiratory illness. On the heels of two atypical flu seasons, a few of us may be quietly complacent about the importance of flu vaccines for children.
Anyone in need of a little motivation should check out a paper recently published in Clinical Infectious Diseases that reinforces the value of flu vaccine, even in a year when there is a poor match between the vaccine and circulating viruses.
The 2019-2020 flu season was a bad flu season for children. Two antigenically drifted influenza viruses predominated and cases of influenza soared, resulting in the largest influenza epidemic in children in the United States since 1992. Pediatric Intensive Care Influenza Study investigators used a test-negative design to estimate the effectiveness of influenza vaccine in preventing critical and life-threatening influenza in children during that season. The good news: vaccination reduced the risk of critical influenza by 78% against H1N1pdm09 viruses that were well-matched to vaccine and by 47% against mismatched viruses. Vaccination was estimated to be 75% protective against antigenically drifted B-Victoria viruses. Overall vaccine effectiveness against critical illness from any influenza virus was 63% (95% confidence interval, 38%-78%).
While it might be tempting to attribute suboptimal immunization rates to vaccine hesitancy, ready availability remains an issue for some families. We need to eliminate barriers to access. While the AAP continues to emphasize immunization in the medical home, especially for the youngest infants, the 2022 policy statement suggests that vaccinating children in schools, pharmacies, and other nontraditional settings could improve immunization rates. To the extent feasible, we need to work with partners to support community-based initiatives and promote these to families who struggle to make it into the office.
Improving access is just one potential way to reduce health disparities related to influenza and influenza vaccination. Over 10 influenza seasons, higher rates of influenza-associated hospitalizations and intensive care unit admissions were observed in Black, Hispanic, and American Indian/Alaska Native people. These disparities were highest in children aged younger than 4 years and influenza-associated in-hospital deaths were three- to fourfold higher in Black, Hispanic, and Asian/Pacific Islander children, compared with White children. The reason for the disparities isn’t completely clear but increasing immunization rates may be part of the solution. During the 2020-2021 influenza season, flu immunization rates in Black children (51.6%) were lower than those seen in White (57.4%) and Hispanic children (58.9%).
The AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023, highlight a variety of evidence-based strategies to increase influenza immunization rates. These may provide a little inspiration for clinicians looking to try a new approach. If you wish to share your experience with increasing influenza immunization rates in your practice setting, please email me at [email protected].
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead.
Cold and flu season came early in 2022.
On Nov. 4, 2022, the Centers for Disease Control and Prevention issued a Health Alert Network Health Advisory about early, elevated respiratory disease incidence caused by multiple viruses other than SARS-CoV-2.
Interseasonal spread of respiratory syncytial virus has continued in 2022, with RSV-associated hospitalizations increasing in the late spring and continuing throughout the summer and into the fall. In October, some regions of the country were seeing RSV activity near the peak seasonal levels typically observed in December and January.
Cases of severe respiratory infection in children who tested positive for rhinovirus or enterovirus spiked in August; further testing confirmed the presence of EV-D68 in some children. Rhinovirus and enterovirus continue to circulate and are isolated in hospitalized children with respiratory illness.
In some parts of the country, influenza cases have rapidly increased ahead of what we normally anticipate. According to preliminary estimates from the CDC, between Oct. 1 and Oct. 22, 880,000 people were sickened with flu, 420,000 people visited a health care provider for flu illness, and 6,900 people were hospitalized for flu. The cumulative hospitalization rate is higher than observed at this time of year in every previous flu season since 2010-2011. Hospitalization rates are highest in children aged 0-4 years and adults 65 years and older.
Of course, this report came as no surprise to pediatric health care providers. Many children’s hospitals had been operating at or over capacity for weeks. While a systematic assessment of the surge on children’s hospitals has not been published, anecdotally, hospitals from around the country have described record emergency department visits and inpatient census numbers. Some have set up tents or other temporary facilities to see ambulatory patients and have canceled elective surgeries because of a lack of beds.
There is no quick or easy solution to stem the tide of RSV-related or enterovirus/rhinovirus admissions, but many flu-related hospitalizations are vaccine preventable. Unfortunately, too few children are receiving influenza vaccine. As of the week ending Oct. 15, only about 22.1% of eligible children had been immunized. The American Academy of Pediatrics and the CDC recommend that all children are vaccinated, preferably by the end of October so they have time to develop immunity before influenza starts circulating. As it stands now, the majority of the nation’s children are facing a flu season without the benefits of vaccine.
There is still time to take steps to prevent this flu season from becoming one of the worst in recent memory. A strong provider recommendation for influenza vaccine is consistently associated with higher rates of vaccine acceptance. We need to recommend influenza vaccine to all eligible patients at every visit and in every setting. It will help if we can say it like we mean it. Some of us are tired of debating the merits of COVID-19 vaccine with families and may be leery of additional debates about flu. Some of us may just be tired, as many practices have already expanded office hours to care for the influx of kids with respiratory illness. On the heels of two atypical flu seasons, a few of us may be quietly complacent about the importance of flu vaccines for children.
Anyone in need of a little motivation should check out a paper recently published in Clinical Infectious Diseases that reinforces the value of flu vaccine, even in a year when there is a poor match between the vaccine and circulating viruses.
The 2019-2020 flu season was a bad flu season for children. Two antigenically drifted influenza viruses predominated and cases of influenza soared, resulting in the largest influenza epidemic in children in the United States since 1992. Pediatric Intensive Care Influenza Study investigators used a test-negative design to estimate the effectiveness of influenza vaccine in preventing critical and life-threatening influenza in children during that season. The good news: vaccination reduced the risk of critical influenza by 78% against H1N1pdm09 viruses that were well-matched to vaccine and by 47% against mismatched viruses. Vaccination was estimated to be 75% protective against antigenically drifted B-Victoria viruses. Overall vaccine effectiveness against critical illness from any influenza virus was 63% (95% confidence interval, 38%-78%).
While it might be tempting to attribute suboptimal immunization rates to vaccine hesitancy, ready availability remains an issue for some families. We need to eliminate barriers to access. While the AAP continues to emphasize immunization in the medical home, especially for the youngest infants, the 2022 policy statement suggests that vaccinating children in schools, pharmacies, and other nontraditional settings could improve immunization rates. To the extent feasible, we need to work with partners to support community-based initiatives and promote these to families who struggle to make it into the office.
Improving access is just one potential way to reduce health disparities related to influenza and influenza vaccination. Over 10 influenza seasons, higher rates of influenza-associated hospitalizations and intensive care unit admissions were observed in Black, Hispanic, and American Indian/Alaska Native people. These disparities were highest in children aged younger than 4 years and influenza-associated in-hospital deaths were three- to fourfold higher in Black, Hispanic, and Asian/Pacific Islander children, compared with White children. The reason for the disparities isn’t completely clear but increasing immunization rates may be part of the solution. During the 2020-2021 influenza season, flu immunization rates in Black children (51.6%) were lower than those seen in White (57.4%) and Hispanic children (58.9%).
The AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023, highlight a variety of evidence-based strategies to increase influenza immunization rates. These may provide a little inspiration for clinicians looking to try a new approach. If you wish to share your experience with increasing influenza immunization rates in your practice setting, please email me at [email protected].
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead.
Is opioid abuse leading to pediatric head trauma?
As a physician in the heart of the opioid epidemic, Pavirthra R. Ellison, MD, has watched for years as her patients have lost parents to overdoses. More than 1,400 adults in West Virginia, where she practices, died of opioid abuse in 2021 alone, government statistics show.
The grim toll made Ellison wonder: What was happening to children in the state? The answer, according to a new study, is not reassuring.
Ellison and her colleagues have found a troubling link between a surge in critical head and neck injuries among youth in West Virginia and a spike in positive tests for opioids and benzodiazepines among children who arrive at emergency departments in the state. They don’t think the pattern is a coincidence.
“What we found was really kind of scary,” said Dr. Ellison, a professor of anesthesiology and pediatrics at West Virginia University, Morgantown. “Children in this region often get exposure to these drugs early on.”
A region in crisis
According to a 2020 report from the Department of Health & Human Services, about 9.9 million Americans abused prescription opioids in 2018. That same year, almost 47,000 died following an overdose of the painkillers. In 2017, Appalachian counties experienced a death rate from opioid overdoses that was 72% higher than that of the rest of the country.
Dr. Ellison and associates who presented their findings recently at the 2022 annual meeting of the American Society of Anesthesiologists, examined rates of pediatric trauma injuries, injury severity, and results of drug screenings throughout West Virginia between 2009 and 2019.
The study included 4,538 children and adolescents younger than 18 years who had been treated for head and neck trauma. The youth were divided into two groups: 3,356 who were treated from 2009 to 2016, and 1,182 who were treated between 2017 and 2019.
The incidence of critical head injuries increased from 3.7% in the period 2009-2016 to 7.2% in the period 2017-2019 (P = .007). The incidence of serious neck injuries increased from 12.2% to 27.1% (P = .007) during that period, according to the researchers. The number of days that these patients spent on ventilators more than doubled, from 3.1 to 6.3 (P < .001), they reported.
At the same time, the rate of positive urine drug tests rose sharply, from 0.8% to 1.8% (P < .001) for benzodiazepines and from 1% to 4.9% for opioids (P < .001).
Drug testing of children hospitalized for trauma rose more than threefold, from 6.9% to 23.2% (P < .001). Dr. Ellison’s group was unable to match positive drug screens with patients who came in with injuries.
Dr. Ellison said her research “warrants further evaluation of current policies and protocols targeting substance use in children and adolescents.” To that end, her team is planning to conduct a prospective study in mid 2023 to further illuminate the trends.
“I hope early next year we can put together a group of physicians, pediatric general surgeons, neurosurgeons, and anesthesiologists,” she said. “I want to look at what we can do to reduce the severity of injury.”
She also wants to reach the population that these findings directly affect.
“The next step that we are currently working on is community awareness of the issue,” Dr. Ellison said. “Our trauma institute is partnering with middle school and high school kids to create material to raise awareness.”
Rural Appalachia faces several other endemic problems that affect the health and well-being of children and families, including limited access to health care, poverty, and minimal community support, according to Dr. Ellison. Children and teens in the region who live with parents who abuse opioids are more likely to experience family conflict, mental health challenges, legal troubles, and negative health effects, including physical trauma.
A call to action
Toufic Jildeh, MD, assistant professor of orthopedics, Michigan State University Health Care, East Lansing, who has studied ways to reduce opioid use among surgery patients, called the new findings “alarming.”
After reviewing the study, Dr. Jildeh said that in his opinion, the results support standardized drug testing of children, particularly in the context of severe trauma.
Bruce Bassi, MD, an addiction psychiatrist and owner of TelepsychHealth, a private, online psychiatric practice, agreed. “The main take-home message is that drug screening should be the standard of care for pediatric patients in this region, because it changes the management of those individuals,” Dr. Bassi said.
But identifying these patients is just the first step. “We should continue to educate and raise awareness, not only in the health care system,” Dr. Bassi said. “We also need to let parents know that the possibility of children obtaining access to medications is high.”
The study was independently supported. Dr. Ellison and Dr. Jildeh reported no relevant financial relationships. Dr. Bassi owns a private psychiatry practice called Telepsychhealth but has no other relevant financial relationships.
A version of this article first appeared on Medscape.com.
As a physician in the heart of the opioid epidemic, Pavirthra R. Ellison, MD, has watched for years as her patients have lost parents to overdoses. More than 1,400 adults in West Virginia, where she practices, died of opioid abuse in 2021 alone, government statistics show.
The grim toll made Ellison wonder: What was happening to children in the state? The answer, according to a new study, is not reassuring.
Ellison and her colleagues have found a troubling link between a surge in critical head and neck injuries among youth in West Virginia and a spike in positive tests for opioids and benzodiazepines among children who arrive at emergency departments in the state. They don’t think the pattern is a coincidence.
“What we found was really kind of scary,” said Dr. Ellison, a professor of anesthesiology and pediatrics at West Virginia University, Morgantown. “Children in this region often get exposure to these drugs early on.”
A region in crisis
According to a 2020 report from the Department of Health & Human Services, about 9.9 million Americans abused prescription opioids in 2018. That same year, almost 47,000 died following an overdose of the painkillers. In 2017, Appalachian counties experienced a death rate from opioid overdoses that was 72% higher than that of the rest of the country.
Dr. Ellison and associates who presented their findings recently at the 2022 annual meeting of the American Society of Anesthesiologists, examined rates of pediatric trauma injuries, injury severity, and results of drug screenings throughout West Virginia between 2009 and 2019.
The study included 4,538 children and adolescents younger than 18 years who had been treated for head and neck trauma. The youth were divided into two groups: 3,356 who were treated from 2009 to 2016, and 1,182 who were treated between 2017 and 2019.
The incidence of critical head injuries increased from 3.7% in the period 2009-2016 to 7.2% in the period 2017-2019 (P = .007). The incidence of serious neck injuries increased from 12.2% to 27.1% (P = .007) during that period, according to the researchers. The number of days that these patients spent on ventilators more than doubled, from 3.1 to 6.3 (P < .001), they reported.
At the same time, the rate of positive urine drug tests rose sharply, from 0.8% to 1.8% (P < .001) for benzodiazepines and from 1% to 4.9% for opioids (P < .001).
Drug testing of children hospitalized for trauma rose more than threefold, from 6.9% to 23.2% (P < .001). Dr. Ellison’s group was unable to match positive drug screens with patients who came in with injuries.
Dr. Ellison said her research “warrants further evaluation of current policies and protocols targeting substance use in children and adolescents.” To that end, her team is planning to conduct a prospective study in mid 2023 to further illuminate the trends.
“I hope early next year we can put together a group of physicians, pediatric general surgeons, neurosurgeons, and anesthesiologists,” she said. “I want to look at what we can do to reduce the severity of injury.”
She also wants to reach the population that these findings directly affect.
“The next step that we are currently working on is community awareness of the issue,” Dr. Ellison said. “Our trauma institute is partnering with middle school and high school kids to create material to raise awareness.”
Rural Appalachia faces several other endemic problems that affect the health and well-being of children and families, including limited access to health care, poverty, and minimal community support, according to Dr. Ellison. Children and teens in the region who live with parents who abuse opioids are more likely to experience family conflict, mental health challenges, legal troubles, and negative health effects, including physical trauma.
A call to action
Toufic Jildeh, MD, assistant professor of orthopedics, Michigan State University Health Care, East Lansing, who has studied ways to reduce opioid use among surgery patients, called the new findings “alarming.”
After reviewing the study, Dr. Jildeh said that in his opinion, the results support standardized drug testing of children, particularly in the context of severe trauma.
Bruce Bassi, MD, an addiction psychiatrist and owner of TelepsychHealth, a private, online psychiatric practice, agreed. “The main take-home message is that drug screening should be the standard of care for pediatric patients in this region, because it changes the management of those individuals,” Dr. Bassi said.
But identifying these patients is just the first step. “We should continue to educate and raise awareness, not only in the health care system,” Dr. Bassi said. “We also need to let parents know that the possibility of children obtaining access to medications is high.”
The study was independently supported. Dr. Ellison and Dr. Jildeh reported no relevant financial relationships. Dr. Bassi owns a private psychiatry practice called Telepsychhealth but has no other relevant financial relationships.
A version of this article first appeared on Medscape.com.
As a physician in the heart of the opioid epidemic, Pavirthra R. Ellison, MD, has watched for years as her patients have lost parents to overdoses. More than 1,400 adults in West Virginia, where she practices, died of opioid abuse in 2021 alone, government statistics show.
The grim toll made Ellison wonder: What was happening to children in the state? The answer, according to a new study, is not reassuring.
Ellison and her colleagues have found a troubling link between a surge in critical head and neck injuries among youth in West Virginia and a spike in positive tests for opioids and benzodiazepines among children who arrive at emergency departments in the state. They don’t think the pattern is a coincidence.
“What we found was really kind of scary,” said Dr. Ellison, a professor of anesthesiology and pediatrics at West Virginia University, Morgantown. “Children in this region often get exposure to these drugs early on.”
A region in crisis
According to a 2020 report from the Department of Health & Human Services, about 9.9 million Americans abused prescription opioids in 2018. That same year, almost 47,000 died following an overdose of the painkillers. In 2017, Appalachian counties experienced a death rate from opioid overdoses that was 72% higher than that of the rest of the country.
Dr. Ellison and associates who presented their findings recently at the 2022 annual meeting of the American Society of Anesthesiologists, examined rates of pediatric trauma injuries, injury severity, and results of drug screenings throughout West Virginia between 2009 and 2019.
The study included 4,538 children and adolescents younger than 18 years who had been treated for head and neck trauma. The youth were divided into two groups: 3,356 who were treated from 2009 to 2016, and 1,182 who were treated between 2017 and 2019.
The incidence of critical head injuries increased from 3.7% in the period 2009-2016 to 7.2% in the period 2017-2019 (P = .007). The incidence of serious neck injuries increased from 12.2% to 27.1% (P = .007) during that period, according to the researchers. The number of days that these patients spent on ventilators more than doubled, from 3.1 to 6.3 (P < .001), they reported.
At the same time, the rate of positive urine drug tests rose sharply, from 0.8% to 1.8% (P < .001) for benzodiazepines and from 1% to 4.9% for opioids (P < .001).
Drug testing of children hospitalized for trauma rose more than threefold, from 6.9% to 23.2% (P < .001). Dr. Ellison’s group was unable to match positive drug screens with patients who came in with injuries.
Dr. Ellison said her research “warrants further evaluation of current policies and protocols targeting substance use in children and adolescents.” To that end, her team is planning to conduct a prospective study in mid 2023 to further illuminate the trends.
“I hope early next year we can put together a group of physicians, pediatric general surgeons, neurosurgeons, and anesthesiologists,” she said. “I want to look at what we can do to reduce the severity of injury.”
She also wants to reach the population that these findings directly affect.
“The next step that we are currently working on is community awareness of the issue,” Dr. Ellison said. “Our trauma institute is partnering with middle school and high school kids to create material to raise awareness.”
Rural Appalachia faces several other endemic problems that affect the health and well-being of children and families, including limited access to health care, poverty, and minimal community support, according to Dr. Ellison. Children and teens in the region who live with parents who abuse opioids are more likely to experience family conflict, mental health challenges, legal troubles, and negative health effects, including physical trauma.
A call to action
Toufic Jildeh, MD, assistant professor of orthopedics, Michigan State University Health Care, East Lansing, who has studied ways to reduce opioid use among surgery patients, called the new findings “alarming.”
After reviewing the study, Dr. Jildeh said that in his opinion, the results support standardized drug testing of children, particularly in the context of severe trauma.
Bruce Bassi, MD, an addiction psychiatrist and owner of TelepsychHealth, a private, online psychiatric practice, agreed. “The main take-home message is that drug screening should be the standard of care for pediatric patients in this region, because it changes the management of those individuals,” Dr. Bassi said.
But identifying these patients is just the first step. “We should continue to educate and raise awareness, not only in the health care system,” Dr. Bassi said. “We also need to let parents know that the possibility of children obtaining access to medications is high.”
The study was independently supported. Dr. Ellison and Dr. Jildeh reported no relevant financial relationships. Dr. Bassi owns a private psychiatry practice called Telepsychhealth but has no other relevant financial relationships.
A version of this article first appeared on Medscape.com.
Lego introduces first character with vitiligo
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.