User login
For MD-IQ use only
More evidence hydroxychloroquine is ineffective, harmful in COVID-19
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Today’s top news highlights: Remdesivir data dive, FDA approves contraceptive gel
:
Remdesivir trial data published
Weeks after topline remdesivir data appeared in the press, investigators published their full experience using the drug to treat COVID-19 patients. The study, published in the New England Journal of Medicine, showed the drug reduced recovery time from 15 to 11 days, compared with placebo. Patients receiving oxygen seemed to fare best from treatment with remdesivir. “There is clear and consistent evidence of clinically significant benefit for those hospitalized on oxygen but not yet requiring mechanical ventilation,” Daniel Kaul, MD, a professor of infectious diseases at the University of Michigan, Ann Arbor, said after seeing the published results. “Surprisingly, early dosing as measured from time to onset of symptoms did not seem to make a difference.” READ MORE.
FDA approves contraceptive gel
The Food and Drug Administration approved Phexxi (lactic acid, citric acid, and potassium bitartrate) vaginal gel to prevent pregnancy in women of reproductive potential. It’s the first nonhormonal, on-demand, vaginal pH regulator contraceptive designed to maintain vaginal pH within the range of 3.5-4.5. READ MORE.
COVID-19 lessons from one cancer center
Physicians at Levine Cancer Institute in Charlotte, N.C., largely have been able to keep hematologic oncology patients on their treatment regimens and continue to care for inpatients during the early months of the COVID-19 pandemic. How have they kept the situation managable? Strict infection control, liberal testing, and a proactive plan to defer and temporarily replace infusion care when medically appropriate were all part of the strategy. “My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious, even before the coronavirus, using distancing, masking, and meticulous hand hygiene,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in Levine Cancer Institute’s Department of Hematologic Oncology and Blood Disorders. READ MORE.
Convalescent plasma: Hope or hype?
There are currently more than two dozen trials of convalescent plasma in the United States and elsewhere but most are single-arm trials to determine if one infusion can decrease the need for intubation or help patients on a ventilator to improve. Others researchers are investigating whether convalescent plasma might be used before severe disease sets in. Meanwhile, about 2,200 hospitals are participating in an expanded access program being led by the Mayo Clinic nationwide. The National Institutes of Health recently said that “there are insufficient clinical data to recommend either for or against” its use for COVID-19. READ MORE.
New rosacea treatment guidelines
Patients with rosacea should receive treatments based on their phenotype and specific symptoms, rather than being assigned into distinct subtype categories, according to updated guidance published in the Journal of the American Academy of Dermatology. The update comes from the National Rosacea Society Expert Committee and is based on a review of the evidence. Patients “shouldn’t be classified as having a certain subtype of rosacea” since “many patients have features that overlap more than one subtype,” said Diane Thiboutot, MD, lead author of the update and a professor of dermatology and associate dean of clinical and translational research education at Penn State University, Hershey. READ MORE.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
:
Remdesivir trial data published
Weeks after topline remdesivir data appeared in the press, investigators published their full experience using the drug to treat COVID-19 patients. The study, published in the New England Journal of Medicine, showed the drug reduced recovery time from 15 to 11 days, compared with placebo. Patients receiving oxygen seemed to fare best from treatment with remdesivir. “There is clear and consistent evidence of clinically significant benefit for those hospitalized on oxygen but not yet requiring mechanical ventilation,” Daniel Kaul, MD, a professor of infectious diseases at the University of Michigan, Ann Arbor, said after seeing the published results. “Surprisingly, early dosing as measured from time to onset of symptoms did not seem to make a difference.” READ MORE.
FDA approves contraceptive gel
The Food and Drug Administration approved Phexxi (lactic acid, citric acid, and potassium bitartrate) vaginal gel to prevent pregnancy in women of reproductive potential. It’s the first nonhormonal, on-demand, vaginal pH regulator contraceptive designed to maintain vaginal pH within the range of 3.5-4.5. READ MORE.
COVID-19 lessons from one cancer center
Physicians at Levine Cancer Institute in Charlotte, N.C., largely have been able to keep hematologic oncology patients on their treatment regimens and continue to care for inpatients during the early months of the COVID-19 pandemic. How have they kept the situation managable? Strict infection control, liberal testing, and a proactive plan to defer and temporarily replace infusion care when medically appropriate were all part of the strategy. “My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious, even before the coronavirus, using distancing, masking, and meticulous hand hygiene,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in Levine Cancer Institute’s Department of Hematologic Oncology and Blood Disorders. READ MORE.
Convalescent plasma: Hope or hype?
There are currently more than two dozen trials of convalescent plasma in the United States and elsewhere but most are single-arm trials to determine if one infusion can decrease the need for intubation or help patients on a ventilator to improve. Others researchers are investigating whether convalescent plasma might be used before severe disease sets in. Meanwhile, about 2,200 hospitals are participating in an expanded access program being led by the Mayo Clinic nationwide. The National Institutes of Health recently said that “there are insufficient clinical data to recommend either for or against” its use for COVID-19. READ MORE.
New rosacea treatment guidelines
Patients with rosacea should receive treatments based on their phenotype and specific symptoms, rather than being assigned into distinct subtype categories, according to updated guidance published in the Journal of the American Academy of Dermatology. The update comes from the National Rosacea Society Expert Committee and is based on a review of the evidence. Patients “shouldn’t be classified as having a certain subtype of rosacea” since “many patients have features that overlap more than one subtype,” said Diane Thiboutot, MD, lead author of the update and a professor of dermatology and associate dean of clinical and translational research education at Penn State University, Hershey. READ MORE.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
:
Remdesivir trial data published
Weeks after topline remdesivir data appeared in the press, investigators published their full experience using the drug to treat COVID-19 patients. The study, published in the New England Journal of Medicine, showed the drug reduced recovery time from 15 to 11 days, compared with placebo. Patients receiving oxygen seemed to fare best from treatment with remdesivir. “There is clear and consistent evidence of clinically significant benefit for those hospitalized on oxygen but not yet requiring mechanical ventilation,” Daniel Kaul, MD, a professor of infectious diseases at the University of Michigan, Ann Arbor, said after seeing the published results. “Surprisingly, early dosing as measured from time to onset of symptoms did not seem to make a difference.” READ MORE.
FDA approves contraceptive gel
The Food and Drug Administration approved Phexxi (lactic acid, citric acid, and potassium bitartrate) vaginal gel to prevent pregnancy in women of reproductive potential. It’s the first nonhormonal, on-demand, vaginal pH regulator contraceptive designed to maintain vaginal pH within the range of 3.5-4.5. READ MORE.
COVID-19 lessons from one cancer center
Physicians at Levine Cancer Institute in Charlotte, N.C., largely have been able to keep hematologic oncology patients on their treatment regimens and continue to care for inpatients during the early months of the COVID-19 pandemic. How have they kept the situation managable? Strict infection control, liberal testing, and a proactive plan to defer and temporarily replace infusion care when medically appropriate were all part of the strategy. “My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious, even before the coronavirus, using distancing, masking, and meticulous hand hygiene,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in Levine Cancer Institute’s Department of Hematologic Oncology and Blood Disorders. READ MORE.
Convalescent plasma: Hope or hype?
There are currently more than two dozen trials of convalescent plasma in the United States and elsewhere but most are single-arm trials to determine if one infusion can decrease the need for intubation or help patients on a ventilator to improve. Others researchers are investigating whether convalescent plasma might be used before severe disease sets in. Meanwhile, about 2,200 hospitals are participating in an expanded access program being led by the Mayo Clinic nationwide. The National Institutes of Health recently said that “there are insufficient clinical data to recommend either for or against” its use for COVID-19. READ MORE.
New rosacea treatment guidelines
Patients with rosacea should receive treatments based on their phenotype and specific symptoms, rather than being assigned into distinct subtype categories, according to updated guidance published in the Journal of the American Academy of Dermatology. The update comes from the National Rosacea Society Expert Committee and is based on a review of the evidence. Patients “shouldn’t be classified as having a certain subtype of rosacea” since “many patients have features that overlap more than one subtype,” said Diane Thiboutot, MD, lead author of the update and a professor of dermatology and associate dean of clinical and translational research education at Penn State University, Hershey. READ MORE.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Patient-focused precautions, testing help blunt pandemic effects on heme-onc unit
Keeping hematologic oncology patients on their treatment regimens and caring for inpatients with hematologic malignancies remained “manageable” during the first 2 months of the COVID-19 pandemic at Levine Cancer Institute in Charlotte, N.C.
That level of manageability has partly been because a surge in cases so far hasn’t arrived at Levine or in most of the surrounding North Carolina and South Carolina communities it serves. As of May 15, 2020, the total number of confirmed and reported COVID-19 cases had reached about 19,000 in North Carolina, and just under 9,000 in South Carolina, out of a total population in the two states of close to 16 million. What’s happened instead at Levine Cancer Institute (LCI) has been a steady but low drumbeat of cases that, by mid-May 2020, totaled fewer than 10 patients with hematologic malignancies diagnosed with COVID-19.
“For a large system with multiple sites throughout North and South Carolina that saw 17,200 new patients in 2019 – including solid tumor, benign hematology, and malignant hematology patients – with 198,000 total patient visits, it is safe to say that we are off to a good start. However, we remain in the early throes of the pandemic and we will need to remain vigilant going forward,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in LCI’s Department of Hematologic Oncology and Blood Disorders.
The limited effects to date of COVID-19 at LCI has been thanks to a regimen of great caution for preventing infections that’s been consistently conveyed to LCI patients from before the pandemic’s onset, liberal testing that started early, a proactive plan to defer and temporarily replace infusion care when medically appropriate, a novel staffing approach designed to minimize and contain potential staff outbreaks, and an early pivot to virtual patient contact when feasible.
COVID-19 has had limited penetration into the LCI case load because patients have, in general, “been very careful,” said Dr. Voorhees.
“My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious even before the coronavirus using distancing, masking, and meticulous hand hygiene,” he said in an interview that reviewed the steps LCI took starting in March to confront and manage the effects of the then-nascent pandemic. “Since we started screening asymptomatic patients in the inpatient and outpatient settings we have identified only one patient with COVID-19 infection, which supports the low rate of infection in our patient population thus far.”
Another key step was the launch of “robust” testing for the COVID-19 virus starting on March 9, using an in-house assay from LCI’s parent health system, Atrium Health, that delivered results within 24 hours. Testing became available at LCI “earlier than at many other health systems.” At first, testing was limited to patients or staff presenting with symptoms, but in the following weeks, it expanded to more patients, including those without symptoms who were scheduled for treatment at the apheresis center, cell donors and cell recipients, patients arriving for inpatient chemotherapy or cellular therapy, patients arriving from a skilled nursing facility or similar environments, and more recently, outpatient chemotherapy patients. “We’re now doing a lot of screening,” Dr. Voorhees said. “In general, screening has been well received because patients recognize that it’s for their own safety.”
Another piece of COVID-19 preparedness was a move toward technology as an alternative to face-to-face encounters between patients and staff. “We adopted virtual technology early.” When medically appropriate, they provided either video consultations with more tech-savvy patients or telephone-based virtual visits for patients who preferred a more familiar interface. As LCI starts the process of reentry for patients whose face-to-face encounters were deferred, virtual visits will remain an important facet of maintaining care while limiting exposure for appropriate patients and facilitating adequate space for social distancing in the clinics and infusion centers.
Atrium Health also launched a “virtual hospital” geared to intensified remote management of COVID-19 patients who aren’t sick enough for hospitalization. “People who test positive automatically enter the virtual hospital and have regular interactions with their team of providers,” with LCI providing additional support for their patients who get infected. Patients receive an equipment kit that lets them monitor and transmit their vital signs. The virtual hospital program also helps expedite personal needs like delivery of prescriptions and food. “It helps patients manage at home, and has been incredibly useful,” said Dr. Voorhees.
Perhaps the most challenging step LCI clinicians took to preclude a potential COVID-19 case surge was to review all patients receiving infusional therapy or planned cellular therapy and triage those who could potentially tolerate a temporary change to either an oral, at-home regimen or to a brief hold on their treatment. Some patients on maintenance, outpatient infusion-therapy regimens “expressed concern about coming to the clinic. We looked at the patients scheduled to come for infusions and decided which visits were essential and which were deferrable without disrupting care by briefly using a noninfusional approach,” said Dr. Voorhees. The number of patients who had their regimens modified or held was “relatively small,” and with the recent recognition that a surge of infections has not occurred, “we’re now rolling out cautious reentry of those patients back to their originally prescribed chemotherapy.”
In addition to concerns of exposure at infusion clinics, there are concerns about the heightened susceptibility of immunosuppressed hematologic oncology patients to COVID-19 and their risk for more severe infection. “Our view is that, if patients tested positive, continuing immunosuppressive treatment would likely be detrimental,” so when possible treatment is temporarily suspended and then resumed when the infection has cleared. “When patients test positive for a prolonged period, a decision to resume treatment must be in the best interests of the patient and weigh the benefits of resuming therapy against the risks of incurring a more severe infection by restarting potentially immunosuppressive therapy,” Dr. Voorhees said.
The enhanced risk that cancer patients face if they develop COVID-19 was documented in a recent review of 218 cancer patients hospitalized for COVID-19 during parts of March and April in a large New York health system. The results showed an overall mortality rate of 28%, including a 37% rate among 54 patients with hematologic malignancies and a 25% rate among 164 patients with solid tumors. The mortality rate “may not be quite as high as they reported because that depends on how many patients you test, but there is no question that patients with more comorbidities are at higher risk. Patients with active cancer on chemotherapy are a particularly vulnerable population, and many have expressed concerns about their vulnerability,” he observed.
For the few LCI patients who developed COVID-19 infection, the medical staff has had several therapeutic options they could match to each patient’s needs, with help from the Atrium Health infectious disease team. LCI and Atrium Health are participating in several COVID-19 clinical treatment trials, including an investigational convalescent plasma protocol spearheaded by the Mayo Clinic. They have also opened a randomized, phase 2 trial evaluating the safety and efficacy of selinexor (Xpovio), an oral drug that’s Food and Drug Administration approved for patients with multiple myeloma, for treatment of moderate or severe COVID-19 infection. Additional studies evaluating blockade of granulocyte-macrophage colony-stimulating factor, as well as inhaled antiviral therapy, have recently launched, and several additional studies are poised to open in the coming weeks.
The LCI and Atrium Health team also has a supply of the antiviral agent remdesivir as part of the FDA’s expanded access protocol and emergency use authorization. They also have a supply of and experience administering the interleukin-6 receptor inhibitor tocilizumab (Actemra), which showed some suggestion of efficacy in limited experience treating patients with severe or critical COVID-19 infections. Clinicians at LCI have not used the investigational and unproven agents hydroxychloroquine, chloroquine, and azithromycin to either prevent or treat COVID-19.
LCI also instituted measures to try to minimize the risk that staff members could become infected and transmit the virus while asymptomatic. Following conversations held early on with COVID-19–experienced health authorities in China and Italy, the patient-facing LCI staff split into two teams starting on March 23 that alternated responsibility for direct patient interactions every 2 weeks. When one of these teams was off from direct patient contact they continued to care for patients remotely through virtual technologies. The concept was that, if a staffer became infected while remaining asymptomatic during their contact with patients, their status would either become diagnosable or resolve during their 2 weeks away from seeing any patients. Perhaps in part because of this approach infections among staff members “have not been a big issue. We’ve had an incredibly low infection rate among the LCI staff,” Dr. Voorhees noted.
By mid-May, with the imminent threat of a sudden CODIV-19 surge moderated, heme-onc operations at LCI began to cautiously revert to more normal operations. “We’re continuing patient screening for signs and symptoms of COVID-19 infection, testing for asymptomatic infections, and requiring masking and social distancing in the clinics and hospitals, but we’re starting to slowly restore the number of patients at our clinics [virtual and face to face[ and infusion centers,” and the staff’s division into two teams ended. “The idea was to get past a surge and make sure our system was not overwhelmed. We anticipated a local surge in late April, but then it kept getting pushed back. Current projections are for the infection rate among LCI patients to remain low provided that community spread remains stable or, ideally, decreases.” The LCI infectious disease staff is closely monitoring infection rates for early recognition of an outbreak, with plans to follow any new cases with contact tracing. So far, the COVID-19 pandemic at LCI “has been very manageable,” Dr. Voorhees concluded.
“We’re now better positioned to deal with a case surge if it were to happen. We could resume the two-team approach, hospital-wide plans are now in place for a future surge, and we are now up and running with robust testing and inpatient and outpatient virtual technology. The first time, we were all learning on the fly.”
The LCI biostatistics team has been prospectively collecting the Institutes’s COVID-19 patient data, with plans to report their findings.
Dr. Voorhees has had financial relationships with Bristol-Myers Squibb/Celgene, Janssen, Novartis, and Oncopeptides, none of which are relevant to this article.
Keeping hematologic oncology patients on their treatment regimens and caring for inpatients with hematologic malignancies remained “manageable” during the first 2 months of the COVID-19 pandemic at Levine Cancer Institute in Charlotte, N.C.
That level of manageability has partly been because a surge in cases so far hasn’t arrived at Levine or in most of the surrounding North Carolina and South Carolina communities it serves. As of May 15, 2020, the total number of confirmed and reported COVID-19 cases had reached about 19,000 in North Carolina, and just under 9,000 in South Carolina, out of a total population in the two states of close to 16 million. What’s happened instead at Levine Cancer Institute (LCI) has been a steady but low drumbeat of cases that, by mid-May 2020, totaled fewer than 10 patients with hematologic malignancies diagnosed with COVID-19.
“For a large system with multiple sites throughout North and South Carolina that saw 17,200 new patients in 2019 – including solid tumor, benign hematology, and malignant hematology patients – with 198,000 total patient visits, it is safe to say that we are off to a good start. However, we remain in the early throes of the pandemic and we will need to remain vigilant going forward,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in LCI’s Department of Hematologic Oncology and Blood Disorders.
The limited effects to date of COVID-19 at LCI has been thanks to a regimen of great caution for preventing infections that’s been consistently conveyed to LCI patients from before the pandemic’s onset, liberal testing that started early, a proactive plan to defer and temporarily replace infusion care when medically appropriate, a novel staffing approach designed to minimize and contain potential staff outbreaks, and an early pivot to virtual patient contact when feasible.
COVID-19 has had limited penetration into the LCI case load because patients have, in general, “been very careful,” said Dr. Voorhees.
“My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious even before the coronavirus using distancing, masking, and meticulous hand hygiene,” he said in an interview that reviewed the steps LCI took starting in March to confront and manage the effects of the then-nascent pandemic. “Since we started screening asymptomatic patients in the inpatient and outpatient settings we have identified only one patient with COVID-19 infection, which supports the low rate of infection in our patient population thus far.”
Another key step was the launch of “robust” testing for the COVID-19 virus starting on March 9, using an in-house assay from LCI’s parent health system, Atrium Health, that delivered results within 24 hours. Testing became available at LCI “earlier than at many other health systems.” At first, testing was limited to patients or staff presenting with symptoms, but in the following weeks, it expanded to more patients, including those without symptoms who were scheduled for treatment at the apheresis center, cell donors and cell recipients, patients arriving for inpatient chemotherapy or cellular therapy, patients arriving from a skilled nursing facility or similar environments, and more recently, outpatient chemotherapy patients. “We’re now doing a lot of screening,” Dr. Voorhees said. “In general, screening has been well received because patients recognize that it’s for their own safety.”
Another piece of COVID-19 preparedness was a move toward technology as an alternative to face-to-face encounters between patients and staff. “We adopted virtual technology early.” When medically appropriate, they provided either video consultations with more tech-savvy patients or telephone-based virtual visits for patients who preferred a more familiar interface. As LCI starts the process of reentry for patients whose face-to-face encounters were deferred, virtual visits will remain an important facet of maintaining care while limiting exposure for appropriate patients and facilitating adequate space for social distancing in the clinics and infusion centers.
Atrium Health also launched a “virtual hospital” geared to intensified remote management of COVID-19 patients who aren’t sick enough for hospitalization. “People who test positive automatically enter the virtual hospital and have regular interactions with their team of providers,” with LCI providing additional support for their patients who get infected. Patients receive an equipment kit that lets them monitor and transmit their vital signs. The virtual hospital program also helps expedite personal needs like delivery of prescriptions and food. “It helps patients manage at home, and has been incredibly useful,” said Dr. Voorhees.
Perhaps the most challenging step LCI clinicians took to preclude a potential COVID-19 case surge was to review all patients receiving infusional therapy or planned cellular therapy and triage those who could potentially tolerate a temporary change to either an oral, at-home regimen or to a brief hold on their treatment. Some patients on maintenance, outpatient infusion-therapy regimens “expressed concern about coming to the clinic. We looked at the patients scheduled to come for infusions and decided which visits were essential and which were deferrable without disrupting care by briefly using a noninfusional approach,” said Dr. Voorhees. The number of patients who had their regimens modified or held was “relatively small,” and with the recent recognition that a surge of infections has not occurred, “we’re now rolling out cautious reentry of those patients back to their originally prescribed chemotherapy.”
In addition to concerns of exposure at infusion clinics, there are concerns about the heightened susceptibility of immunosuppressed hematologic oncology patients to COVID-19 and their risk for more severe infection. “Our view is that, if patients tested positive, continuing immunosuppressive treatment would likely be detrimental,” so when possible treatment is temporarily suspended and then resumed when the infection has cleared. “When patients test positive for a prolonged period, a decision to resume treatment must be in the best interests of the patient and weigh the benefits of resuming therapy against the risks of incurring a more severe infection by restarting potentially immunosuppressive therapy,” Dr. Voorhees said.
The enhanced risk that cancer patients face if they develop COVID-19 was documented in a recent review of 218 cancer patients hospitalized for COVID-19 during parts of March and April in a large New York health system. The results showed an overall mortality rate of 28%, including a 37% rate among 54 patients with hematologic malignancies and a 25% rate among 164 patients with solid tumors. The mortality rate “may not be quite as high as they reported because that depends on how many patients you test, but there is no question that patients with more comorbidities are at higher risk. Patients with active cancer on chemotherapy are a particularly vulnerable population, and many have expressed concerns about their vulnerability,” he observed.
For the few LCI patients who developed COVID-19 infection, the medical staff has had several therapeutic options they could match to each patient’s needs, with help from the Atrium Health infectious disease team. LCI and Atrium Health are participating in several COVID-19 clinical treatment trials, including an investigational convalescent plasma protocol spearheaded by the Mayo Clinic. They have also opened a randomized, phase 2 trial evaluating the safety and efficacy of selinexor (Xpovio), an oral drug that’s Food and Drug Administration approved for patients with multiple myeloma, for treatment of moderate or severe COVID-19 infection. Additional studies evaluating blockade of granulocyte-macrophage colony-stimulating factor, as well as inhaled antiviral therapy, have recently launched, and several additional studies are poised to open in the coming weeks.
The LCI and Atrium Health team also has a supply of the antiviral agent remdesivir as part of the FDA’s expanded access protocol and emergency use authorization. They also have a supply of and experience administering the interleukin-6 receptor inhibitor tocilizumab (Actemra), which showed some suggestion of efficacy in limited experience treating patients with severe or critical COVID-19 infections. Clinicians at LCI have not used the investigational and unproven agents hydroxychloroquine, chloroquine, and azithromycin to either prevent or treat COVID-19.
LCI also instituted measures to try to minimize the risk that staff members could become infected and transmit the virus while asymptomatic. Following conversations held early on with COVID-19–experienced health authorities in China and Italy, the patient-facing LCI staff split into two teams starting on March 23 that alternated responsibility for direct patient interactions every 2 weeks. When one of these teams was off from direct patient contact they continued to care for patients remotely through virtual technologies. The concept was that, if a staffer became infected while remaining asymptomatic during their contact with patients, their status would either become diagnosable or resolve during their 2 weeks away from seeing any patients. Perhaps in part because of this approach infections among staff members “have not been a big issue. We’ve had an incredibly low infection rate among the LCI staff,” Dr. Voorhees noted.
By mid-May, with the imminent threat of a sudden CODIV-19 surge moderated, heme-onc operations at LCI began to cautiously revert to more normal operations. “We’re continuing patient screening for signs and symptoms of COVID-19 infection, testing for asymptomatic infections, and requiring masking and social distancing in the clinics and hospitals, but we’re starting to slowly restore the number of patients at our clinics [virtual and face to face[ and infusion centers,” and the staff’s division into two teams ended. “The idea was to get past a surge and make sure our system was not overwhelmed. We anticipated a local surge in late April, but then it kept getting pushed back. Current projections are for the infection rate among LCI patients to remain low provided that community spread remains stable or, ideally, decreases.” The LCI infectious disease staff is closely monitoring infection rates for early recognition of an outbreak, with plans to follow any new cases with contact tracing. So far, the COVID-19 pandemic at LCI “has been very manageable,” Dr. Voorhees concluded.
“We’re now better positioned to deal with a case surge if it were to happen. We could resume the two-team approach, hospital-wide plans are now in place for a future surge, and we are now up and running with robust testing and inpatient and outpatient virtual technology. The first time, we were all learning on the fly.”
The LCI biostatistics team has been prospectively collecting the Institutes’s COVID-19 patient data, with plans to report their findings.
Dr. Voorhees has had financial relationships with Bristol-Myers Squibb/Celgene, Janssen, Novartis, and Oncopeptides, none of which are relevant to this article.
Keeping hematologic oncology patients on their treatment regimens and caring for inpatients with hematologic malignancies remained “manageable” during the first 2 months of the COVID-19 pandemic at Levine Cancer Institute in Charlotte, N.C.
That level of manageability has partly been because a surge in cases so far hasn’t arrived at Levine or in most of the surrounding North Carolina and South Carolina communities it serves. As of May 15, 2020, the total number of confirmed and reported COVID-19 cases had reached about 19,000 in North Carolina, and just under 9,000 in South Carolina, out of a total population in the two states of close to 16 million. What’s happened instead at Levine Cancer Institute (LCI) has been a steady but low drumbeat of cases that, by mid-May 2020, totaled fewer than 10 patients with hematologic malignancies diagnosed with COVID-19.
“For a large system with multiple sites throughout North and South Carolina that saw 17,200 new patients in 2019 – including solid tumor, benign hematology, and malignant hematology patients – with 198,000 total patient visits, it is safe to say that we are off to a good start. However, we remain in the early throes of the pandemic and we will need to remain vigilant going forward,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in LCI’s Department of Hematologic Oncology and Blood Disorders.
The limited effects to date of COVID-19 at LCI has been thanks to a regimen of great caution for preventing infections that’s been consistently conveyed to LCI patients from before the pandemic’s onset, liberal testing that started early, a proactive plan to defer and temporarily replace infusion care when medically appropriate, a novel staffing approach designed to minimize and contain potential staff outbreaks, and an early pivot to virtual patient contact when feasible.
COVID-19 has had limited penetration into the LCI case load because patients have, in general, “been very careful,” said Dr. Voorhees.
“My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious even before the coronavirus using distancing, masking, and meticulous hand hygiene,” he said in an interview that reviewed the steps LCI took starting in March to confront and manage the effects of the then-nascent pandemic. “Since we started screening asymptomatic patients in the inpatient and outpatient settings we have identified only one patient with COVID-19 infection, which supports the low rate of infection in our patient population thus far.”
Another key step was the launch of “robust” testing for the COVID-19 virus starting on March 9, using an in-house assay from LCI’s parent health system, Atrium Health, that delivered results within 24 hours. Testing became available at LCI “earlier than at many other health systems.” At first, testing was limited to patients or staff presenting with symptoms, but in the following weeks, it expanded to more patients, including those without symptoms who were scheduled for treatment at the apheresis center, cell donors and cell recipients, patients arriving for inpatient chemotherapy or cellular therapy, patients arriving from a skilled nursing facility or similar environments, and more recently, outpatient chemotherapy patients. “We’re now doing a lot of screening,” Dr. Voorhees said. “In general, screening has been well received because patients recognize that it’s for their own safety.”
Another piece of COVID-19 preparedness was a move toward technology as an alternative to face-to-face encounters between patients and staff. “We adopted virtual technology early.” When medically appropriate, they provided either video consultations with more tech-savvy patients or telephone-based virtual visits for patients who preferred a more familiar interface. As LCI starts the process of reentry for patients whose face-to-face encounters were deferred, virtual visits will remain an important facet of maintaining care while limiting exposure for appropriate patients and facilitating adequate space for social distancing in the clinics and infusion centers.
Atrium Health also launched a “virtual hospital” geared to intensified remote management of COVID-19 patients who aren’t sick enough for hospitalization. “People who test positive automatically enter the virtual hospital and have regular interactions with their team of providers,” with LCI providing additional support for their patients who get infected. Patients receive an equipment kit that lets them monitor and transmit their vital signs. The virtual hospital program also helps expedite personal needs like delivery of prescriptions and food. “It helps patients manage at home, and has been incredibly useful,” said Dr. Voorhees.
Perhaps the most challenging step LCI clinicians took to preclude a potential COVID-19 case surge was to review all patients receiving infusional therapy or planned cellular therapy and triage those who could potentially tolerate a temporary change to either an oral, at-home regimen or to a brief hold on their treatment. Some patients on maintenance, outpatient infusion-therapy regimens “expressed concern about coming to the clinic. We looked at the patients scheduled to come for infusions and decided which visits were essential and which were deferrable without disrupting care by briefly using a noninfusional approach,” said Dr. Voorhees. The number of patients who had their regimens modified or held was “relatively small,” and with the recent recognition that a surge of infections has not occurred, “we’re now rolling out cautious reentry of those patients back to their originally prescribed chemotherapy.”
In addition to concerns of exposure at infusion clinics, there are concerns about the heightened susceptibility of immunosuppressed hematologic oncology patients to COVID-19 and their risk for more severe infection. “Our view is that, if patients tested positive, continuing immunosuppressive treatment would likely be detrimental,” so when possible treatment is temporarily suspended and then resumed when the infection has cleared. “When patients test positive for a prolonged period, a decision to resume treatment must be in the best interests of the patient and weigh the benefits of resuming therapy against the risks of incurring a more severe infection by restarting potentially immunosuppressive therapy,” Dr. Voorhees said.
The enhanced risk that cancer patients face if they develop COVID-19 was documented in a recent review of 218 cancer patients hospitalized for COVID-19 during parts of March and April in a large New York health system. The results showed an overall mortality rate of 28%, including a 37% rate among 54 patients with hematologic malignancies and a 25% rate among 164 patients with solid tumors. The mortality rate “may not be quite as high as they reported because that depends on how many patients you test, but there is no question that patients with more comorbidities are at higher risk. Patients with active cancer on chemotherapy are a particularly vulnerable population, and many have expressed concerns about their vulnerability,” he observed.
For the few LCI patients who developed COVID-19 infection, the medical staff has had several therapeutic options they could match to each patient’s needs, with help from the Atrium Health infectious disease team. LCI and Atrium Health are participating in several COVID-19 clinical treatment trials, including an investigational convalescent plasma protocol spearheaded by the Mayo Clinic. They have also opened a randomized, phase 2 trial evaluating the safety and efficacy of selinexor (Xpovio), an oral drug that’s Food and Drug Administration approved for patients with multiple myeloma, for treatment of moderate or severe COVID-19 infection. Additional studies evaluating blockade of granulocyte-macrophage colony-stimulating factor, as well as inhaled antiviral therapy, have recently launched, and several additional studies are poised to open in the coming weeks.
The LCI and Atrium Health team also has a supply of the antiviral agent remdesivir as part of the FDA’s expanded access protocol and emergency use authorization. They also have a supply of and experience administering the interleukin-6 receptor inhibitor tocilizumab (Actemra), which showed some suggestion of efficacy in limited experience treating patients with severe or critical COVID-19 infections. Clinicians at LCI have not used the investigational and unproven agents hydroxychloroquine, chloroquine, and azithromycin to either prevent or treat COVID-19.
LCI also instituted measures to try to minimize the risk that staff members could become infected and transmit the virus while asymptomatic. Following conversations held early on with COVID-19–experienced health authorities in China and Italy, the patient-facing LCI staff split into two teams starting on March 23 that alternated responsibility for direct patient interactions every 2 weeks. When one of these teams was off from direct patient contact they continued to care for patients remotely through virtual technologies. The concept was that, if a staffer became infected while remaining asymptomatic during their contact with patients, their status would either become diagnosable or resolve during their 2 weeks away from seeing any patients. Perhaps in part because of this approach infections among staff members “have not been a big issue. We’ve had an incredibly low infection rate among the LCI staff,” Dr. Voorhees noted.
By mid-May, with the imminent threat of a sudden CODIV-19 surge moderated, heme-onc operations at LCI began to cautiously revert to more normal operations. “We’re continuing patient screening for signs and symptoms of COVID-19 infection, testing for asymptomatic infections, and requiring masking and social distancing in the clinics and hospitals, but we’re starting to slowly restore the number of patients at our clinics [virtual and face to face[ and infusion centers,” and the staff’s division into two teams ended. “The idea was to get past a surge and make sure our system was not overwhelmed. We anticipated a local surge in late April, but then it kept getting pushed back. Current projections are for the infection rate among LCI patients to remain low provided that community spread remains stable or, ideally, decreases.” The LCI infectious disease staff is closely monitoring infection rates for early recognition of an outbreak, with plans to follow any new cases with contact tracing. So far, the COVID-19 pandemic at LCI “has been very manageable,” Dr. Voorhees concluded.
“We’re now better positioned to deal with a case surge if it were to happen. We could resume the two-team approach, hospital-wide plans are now in place for a future surge, and we are now up and running with robust testing and inpatient and outpatient virtual technology. The first time, we were all learning on the fly.”
The LCI biostatistics team has been prospectively collecting the Institutes’s COVID-19 patient data, with plans to report their findings.
Dr. Voorhees has had financial relationships with Bristol-Myers Squibb/Celgene, Janssen, Novartis, and Oncopeptides, none of which are relevant to this article.
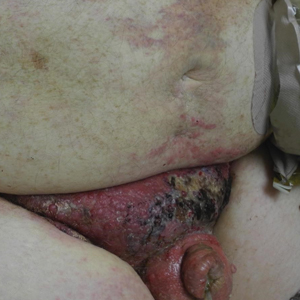
Painful Indurated Plaque on the Groin
The Diagnosis: Cutaneous Metastasis
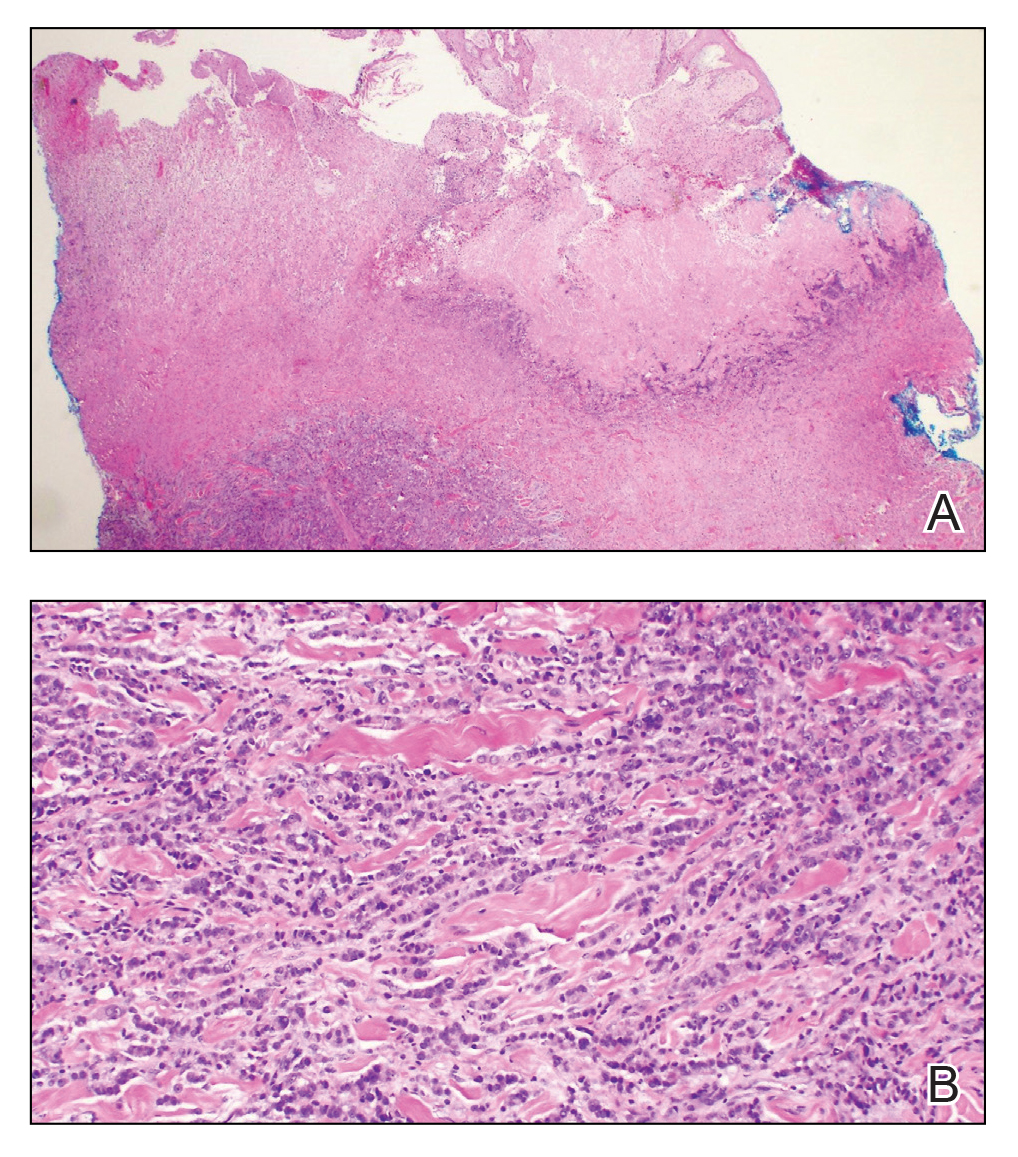
Histopathology demonstrated ulceration of the epidermis with necrosis of the papillary dermis. There was a diffuse infiltration of pleomorphic and atypical epithelioid cells in the reticular dermis (Figure). Focally there was ductal and glandular differentiation. The stroma was sclerotic. At the deep aspect of the biopsy specimen, tumor cells intercalated between collagen bundles in linear strands. Atypical mitoses were common, and necrosis en masse was seen. An immunohistochemical panel also was performed. Tissue from the biopsy was strongly positive for CDX-2 and cytokeratin 20 and diffusely negative for cytokeratin 7, gross cystic disease fluid protein 15, and prostate-specific antigen. The other biopsy was sent for cultures and grew no organisms, which confirmed the diagnosis of cutaneous metastasis from the patient's primary colonic adenocarcinoma. Due to the poor prognosis and his overall poor health, our patient opted for palliative care.
Based on large retrospective studies, the frequency of cutaneous metastasis for patients diagnosed with any malignancy is 0.7% to 9.0%.1-4 The third most common malignancy in both sexes is colorectal cancer, affecting approximately 5% of the US population.3 The frequency of cutaneous metastases from colorectal cancer is 0.81% to 3.9%.1,2,4,5 Generally, cutaneous metastases present within 2 to 3 years from diagnosis of primary malignancy.6,7 The most common sites for cutaneous metastases in a patient with colorectal cancer are the abdomen and pelvic region, often at surgical sites.1-4,6-9
The clinical presentation of cutaneous metastases varies greatly, and as a result, they commonly are misdiagnosed.6,7 Although treatment with many antibiotics and antifungals had failed in our patient, the examination still was concerning for a possible granulomatous infection vs malignancy. With the history of colon cancer, radiation treatment, and chemotherapy, the possible malignancy diagnoses included primary skin cancers, viral tumors, and cutaneous metastasis. The initial evaluations had focused on infectious causes and resulted in 6 weeks of misdiagnosis and inappropriate therapy. Despite cutaneous metastases being uncommon, there should be a high index of suspicion for lesions in patients who have a history of cancer, especially if the lesion does not respond to treatment.2,6,7
Physical examination in our patient showed a high tumor burden as well as evidence of carcinoma erysipeloides on the lower abdomen and thighs, in addition to carcinoma en cuirasse throughout the pubic region. Carcinoma erysipeloides was first described in 1893 in a patient with breast cancer: "The erythematous infiltration of the skin was very superficial, and was attended simply by redness with a slight degree of induration. Until touched by the finger the condition might easily have been taken for a slightly-marked form of erysipelas."10 The clinical findings are a result of lymphatic and vascular obstruction.3,9 The breast is the most common location to find carcinoma erysipeloides.3 It is an unusual occurrence to find it on the abdomen from colonic adenocarcinoma. The term cancer en cuirasse was coined in 1838 to describe the cutaneous manifestation of breast cancer that caused the skin to resemble the metal breastplate of a cuirasser.4 Similar to carcinoma erysipeloides, carcinoma en cuirasse most commonly is found as cutaneous metastasis from breast cancer, not from colonic adenocarcinoma.3
The histologic characteristics of cutaneous metastases in general are similar to the primary malignancy but can be more poorly differentiated.7 Generally, neoplastic cells are seen in the lymphatic and blood vessels, and a large portion of the tumor is confined to the deep dermis and in the subcutaneous fat.3,6 Histologic features of colonic adenocarcinoma metastases can demonstrate a well-differentiated, glandular architecture with mucin-secreting cells.3,8,9 There also is a histologic pattern of neoplastic cells arranging themselves between collagen bundles in linear strands; this finding more commonly is seen in adenocarcinoma of the breast but also was seen in our patient.3,9 With immunohistochemical staining, a truncated panel of cytokeratin 7, cytokeratin 20, and S-100 had a diagnostic accuracy of 100% for cutaneous metastases from colonic adenocarcinoma in one study. The pattern of all colonic adenocarcinomas was cytokeratin 20 positive and cytokeratin 7 and S-100 negative.6
Cutaneous metastases typically demonstrate widespread and rapidly progressive disease.3,9 Survival studies of cutaneous metastases showed that 48% to 66% of patients died within the first 6 months.3,6 Specifically, cutaneous metastases from colorectal cancers showed a median survival of 3 to 5 months.6,7 Currently there are no treatment guidelines for cutaneous metastases.
- Lookingbill DP, Spangler N, Helm K. Cutaneous metastases in patients with metastatic carcinoma: a retrospective study of 4020 patients. J Am Acad Dermatol. 1993;29(2 pt 1):228-236.
- Gul U, Kilic A, Gonul M, et al. Spectrum of cutaneous metastases in 1287 cases of internal malignancies: a study from Turkey. Acta Derm Venereol. 2007;87:160-162.
- Hussein MR. Skin metastasis: a pathologist's perspective. J Cutan Pathol. 2010;37:E1-E20.
- Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33(2 pt 1):161-182; quiz 183-186.
- Hu S, Chen G, Wu C, et al. Rates of cutaneous metastases from different internal malignancies: experience from a Taiwanese medical center. J Am Acad Dermatol. 2009;60:379-387.
- Saeed S, Keehn C, Morgan M. Cutaneous metastasis: a clinical, pathological, and immunohistochemical appraisal. J Cutan Pathol. 2004;31:419-430.
- Sariya D, Ruth K, Adams-McDonnell R. Clinicopathologic correlation of cutaneous metastases: experience of a cancer center. Arch Dermatol. 2007;143:613-620.
- Brownstein M, Helwig E. Metastatic tumors of the skin. Cancer. 1972;29:1298-1307.
- McKee PH. Cutaneous metastases. J Cutan Pathol. 1985;12:239-250.
- Hutchinson J. Notes from congresses and continental hospitals: erythema-scirrhus of the skin in association with cancer of the breast. Arch Surg (London). 1893;4:220-222
The Diagnosis: Cutaneous Metastasis
Histopathology demonstrated ulceration of the epidermis with necrosis of the papillary dermis. There was a diffuse infiltration of pleomorphic and atypical epithelioid cells in the reticular dermis (Figure). Focally there was ductal and glandular differentiation. The stroma was sclerotic. At the deep aspect of the biopsy specimen, tumor cells intercalated between collagen bundles in linear strands. Atypical mitoses were common, and necrosis en masse was seen. An immunohistochemical panel also was performed. Tissue from the biopsy was strongly positive for CDX-2 and cytokeratin 20 and diffusely negative for cytokeratin 7, gross cystic disease fluid protein 15, and prostate-specific antigen. The other biopsy was sent for cultures and grew no organisms, which confirmed the diagnosis of cutaneous metastasis from the patient's primary colonic adenocarcinoma. Due to the poor prognosis and his overall poor health, our patient opted for palliative care.

Based on large retrospective studies, the frequency of cutaneous metastasis for patients diagnosed with any malignancy is 0.7% to 9.0%.1-4 The third most common malignancy in both sexes is colorectal cancer, affecting approximately 5% of the US population.3 The frequency of cutaneous metastases from colorectal cancer is 0.81% to 3.9%.1,2,4,5 Generally, cutaneous metastases present within 2 to 3 years from diagnosis of primary malignancy.6,7 The most common sites for cutaneous metastases in a patient with colorectal cancer are the abdomen and pelvic region, often at surgical sites.1-4,6-9
The clinical presentation of cutaneous metastases varies greatly, and as a result, they commonly are misdiagnosed.6,7 Although treatment with many antibiotics and antifungals had failed in our patient, the examination still was concerning for a possible granulomatous infection vs malignancy. With the history of colon cancer, radiation treatment, and chemotherapy, the possible malignancy diagnoses included primary skin cancers, viral tumors, and cutaneous metastasis. The initial evaluations had focused on infectious causes and resulted in 6 weeks of misdiagnosis and inappropriate therapy. Despite cutaneous metastases being uncommon, there should be a high index of suspicion for lesions in patients who have a history of cancer, especially if the lesion does not respond to treatment.2,6,7
Physical examination in our patient showed a high tumor burden as well as evidence of carcinoma erysipeloides on the lower abdomen and thighs, in addition to carcinoma en cuirasse throughout the pubic region. Carcinoma erysipeloides was first described in 1893 in a patient with breast cancer: "The erythematous infiltration of the skin was very superficial, and was attended simply by redness with a slight degree of induration. Until touched by the finger the condition might easily have been taken for a slightly-marked form of erysipelas."10 The clinical findings are a result of lymphatic and vascular obstruction.3,9 The breast is the most common location to find carcinoma erysipeloides.3 It is an unusual occurrence to find it on the abdomen from colonic adenocarcinoma. The term cancer en cuirasse was coined in 1838 to describe the cutaneous manifestation of breast cancer that caused the skin to resemble the metal breastplate of a cuirasser.4 Similar to carcinoma erysipeloides, carcinoma en cuirasse most commonly is found as cutaneous metastasis from breast cancer, not from colonic adenocarcinoma.3
The histologic characteristics of cutaneous metastases in general are similar to the primary malignancy but can be more poorly differentiated.7 Generally, neoplastic cells are seen in the lymphatic and blood vessels, and a large portion of the tumor is confined to the deep dermis and in the subcutaneous fat.3,6 Histologic features of colonic adenocarcinoma metastases can demonstrate a well-differentiated, glandular architecture with mucin-secreting cells.3,8,9 There also is a histologic pattern of neoplastic cells arranging themselves between collagen bundles in linear strands; this finding more commonly is seen in adenocarcinoma of the breast but also was seen in our patient.3,9 With immunohistochemical staining, a truncated panel of cytokeratin 7, cytokeratin 20, and S-100 had a diagnostic accuracy of 100% for cutaneous metastases from colonic adenocarcinoma in one study. The pattern of all colonic adenocarcinomas was cytokeratin 20 positive and cytokeratin 7 and S-100 negative.6
Cutaneous metastases typically demonstrate widespread and rapidly progressive disease.3,9 Survival studies of cutaneous metastases showed that 48% to 66% of patients died within the first 6 months.3,6 Specifically, cutaneous metastases from colorectal cancers showed a median survival of 3 to 5 months.6,7 Currently there are no treatment guidelines for cutaneous metastases.
The Diagnosis: Cutaneous Metastasis
Histopathology demonstrated ulceration of the epidermis with necrosis of the papillary dermis. There was a diffuse infiltration of pleomorphic and atypical epithelioid cells in the reticular dermis (Figure). Focally there was ductal and glandular differentiation. The stroma was sclerotic. At the deep aspect of the biopsy specimen, tumor cells intercalated between collagen bundles in linear strands. Atypical mitoses were common, and necrosis en masse was seen. An immunohistochemical panel also was performed. Tissue from the biopsy was strongly positive for CDX-2 and cytokeratin 20 and diffusely negative for cytokeratin 7, gross cystic disease fluid protein 15, and prostate-specific antigen. The other biopsy was sent for cultures and grew no organisms, which confirmed the diagnosis of cutaneous metastasis from the patient's primary colonic adenocarcinoma. Due to the poor prognosis and his overall poor health, our patient opted for palliative care.

Based on large retrospective studies, the frequency of cutaneous metastasis for patients diagnosed with any malignancy is 0.7% to 9.0%.1-4 The third most common malignancy in both sexes is colorectal cancer, affecting approximately 5% of the US population.3 The frequency of cutaneous metastases from colorectal cancer is 0.81% to 3.9%.1,2,4,5 Generally, cutaneous metastases present within 2 to 3 years from diagnosis of primary malignancy.6,7 The most common sites for cutaneous metastases in a patient with colorectal cancer are the abdomen and pelvic region, often at surgical sites.1-4,6-9
The clinical presentation of cutaneous metastases varies greatly, and as a result, they commonly are misdiagnosed.6,7 Although treatment with many antibiotics and antifungals had failed in our patient, the examination still was concerning for a possible granulomatous infection vs malignancy. With the history of colon cancer, radiation treatment, and chemotherapy, the possible malignancy diagnoses included primary skin cancers, viral tumors, and cutaneous metastasis. The initial evaluations had focused on infectious causes and resulted in 6 weeks of misdiagnosis and inappropriate therapy. Despite cutaneous metastases being uncommon, there should be a high index of suspicion for lesions in patients who have a history of cancer, especially if the lesion does not respond to treatment.2,6,7
Physical examination in our patient showed a high tumor burden as well as evidence of carcinoma erysipeloides on the lower abdomen and thighs, in addition to carcinoma en cuirasse throughout the pubic region. Carcinoma erysipeloides was first described in 1893 in a patient with breast cancer: "The erythematous infiltration of the skin was very superficial, and was attended simply by redness with a slight degree of induration. Until touched by the finger the condition might easily have been taken for a slightly-marked form of erysipelas."10 The clinical findings are a result of lymphatic and vascular obstruction.3,9 The breast is the most common location to find carcinoma erysipeloides.3 It is an unusual occurrence to find it on the abdomen from colonic adenocarcinoma. The term cancer en cuirasse was coined in 1838 to describe the cutaneous manifestation of breast cancer that caused the skin to resemble the metal breastplate of a cuirasser.4 Similar to carcinoma erysipeloides, carcinoma en cuirasse most commonly is found as cutaneous metastasis from breast cancer, not from colonic adenocarcinoma.3
The histologic characteristics of cutaneous metastases in general are similar to the primary malignancy but can be more poorly differentiated.7 Generally, neoplastic cells are seen in the lymphatic and blood vessels, and a large portion of the tumor is confined to the deep dermis and in the subcutaneous fat.3,6 Histologic features of colonic adenocarcinoma metastases can demonstrate a well-differentiated, glandular architecture with mucin-secreting cells.3,8,9 There also is a histologic pattern of neoplastic cells arranging themselves between collagen bundles in linear strands; this finding more commonly is seen in adenocarcinoma of the breast but also was seen in our patient.3,9 With immunohistochemical staining, a truncated panel of cytokeratin 7, cytokeratin 20, and S-100 had a diagnostic accuracy of 100% for cutaneous metastases from colonic adenocarcinoma in one study. The pattern of all colonic adenocarcinomas was cytokeratin 20 positive and cytokeratin 7 and S-100 negative.6
Cutaneous metastases typically demonstrate widespread and rapidly progressive disease.3,9 Survival studies of cutaneous metastases showed that 48% to 66% of patients died within the first 6 months.3,6 Specifically, cutaneous metastases from colorectal cancers showed a median survival of 3 to 5 months.6,7 Currently there are no treatment guidelines for cutaneous metastases.
- Lookingbill DP, Spangler N, Helm K. Cutaneous metastases in patients with metastatic carcinoma: a retrospective study of 4020 patients. J Am Acad Dermatol. 1993;29(2 pt 1):228-236.
- Gul U, Kilic A, Gonul M, et al. Spectrum of cutaneous metastases in 1287 cases of internal malignancies: a study from Turkey. Acta Derm Venereol. 2007;87:160-162.
- Hussein MR. Skin metastasis: a pathologist's perspective. J Cutan Pathol. 2010;37:E1-E20.
- Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33(2 pt 1):161-182; quiz 183-186.
- Hu S, Chen G, Wu C, et al. Rates of cutaneous metastases from different internal malignancies: experience from a Taiwanese medical center. J Am Acad Dermatol. 2009;60:379-387.
- Saeed S, Keehn C, Morgan M. Cutaneous metastasis: a clinical, pathological, and immunohistochemical appraisal. J Cutan Pathol. 2004;31:419-430.
- Sariya D, Ruth K, Adams-McDonnell R. Clinicopathologic correlation of cutaneous metastases: experience of a cancer center. Arch Dermatol. 2007;143:613-620.
- Brownstein M, Helwig E. Metastatic tumors of the skin. Cancer. 1972;29:1298-1307.
- McKee PH. Cutaneous metastases. J Cutan Pathol. 1985;12:239-250.
- Hutchinson J. Notes from congresses and continental hospitals: erythema-scirrhus of the skin in association with cancer of the breast. Arch Surg (London). 1893;4:220-222
- Lookingbill DP, Spangler N, Helm K. Cutaneous metastases in patients with metastatic carcinoma: a retrospective study of 4020 patients. J Am Acad Dermatol. 1993;29(2 pt 1):228-236.
- Gul U, Kilic A, Gonul M, et al. Spectrum of cutaneous metastases in 1287 cases of internal malignancies: a study from Turkey. Acta Derm Venereol. 2007;87:160-162.
- Hussein MR. Skin metastasis: a pathologist's perspective. J Cutan Pathol. 2010;37:E1-E20.
- Schwartz RA. Cutaneous metastatic disease. J Am Acad Dermatol. 1995;33(2 pt 1):161-182; quiz 183-186.
- Hu S, Chen G, Wu C, et al. Rates of cutaneous metastases from different internal malignancies: experience from a Taiwanese medical center. J Am Acad Dermatol. 2009;60:379-387.
- Saeed S, Keehn C, Morgan M. Cutaneous metastasis: a clinical, pathological, and immunohistochemical appraisal. J Cutan Pathol. 2004;31:419-430.
- Sariya D, Ruth K, Adams-McDonnell R. Clinicopathologic correlation of cutaneous metastases: experience of a cancer center. Arch Dermatol. 2007;143:613-620.
- Brownstein M, Helwig E. Metastatic tumors of the skin. Cancer. 1972;29:1298-1307.
- McKee PH. Cutaneous metastases. J Cutan Pathol. 1985;12:239-250.
- Hutchinson J. Notes from congresses and continental hospitals: erythema-scirrhus of the skin in association with cancer of the breast. Arch Surg (London). 1893;4:220-222
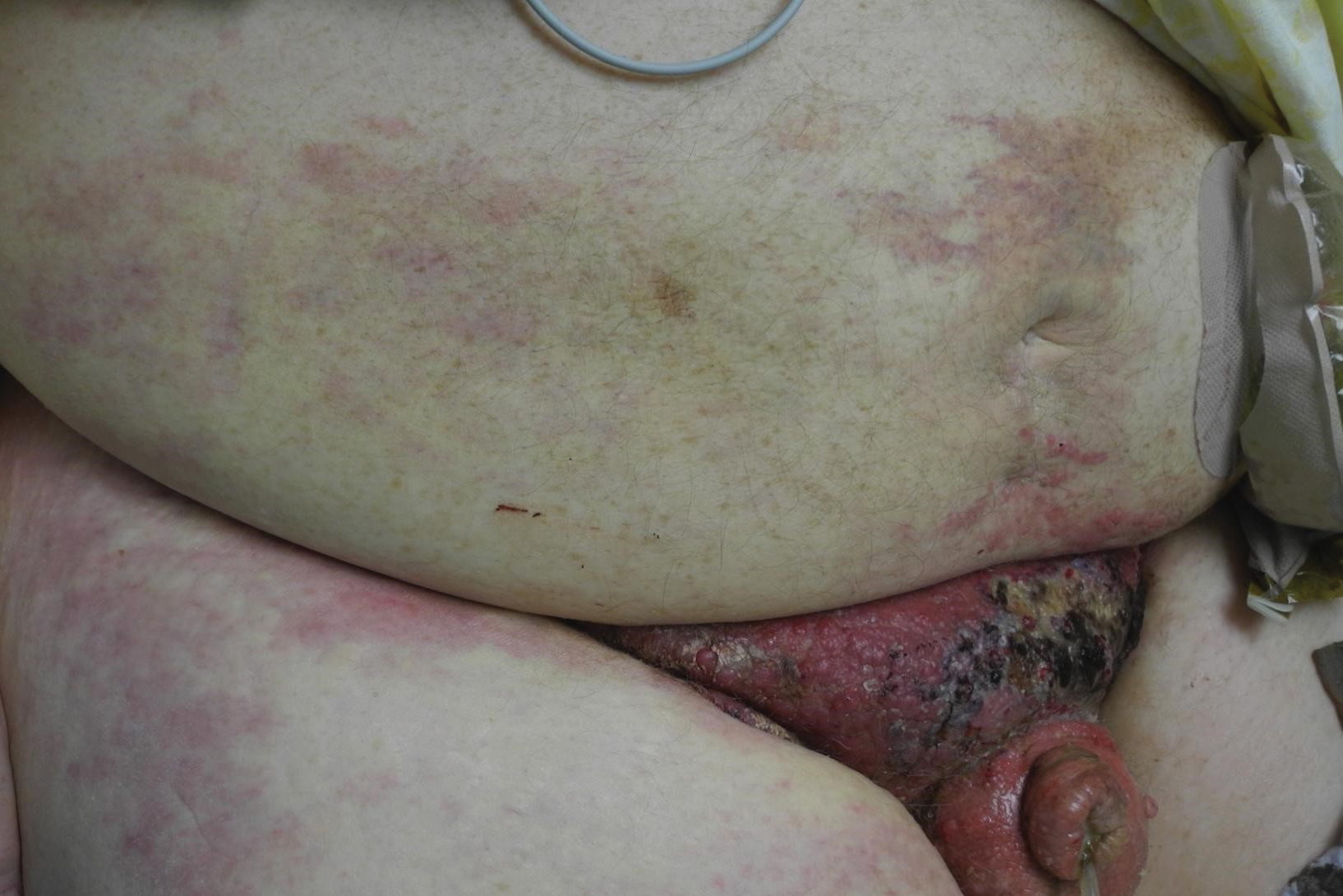
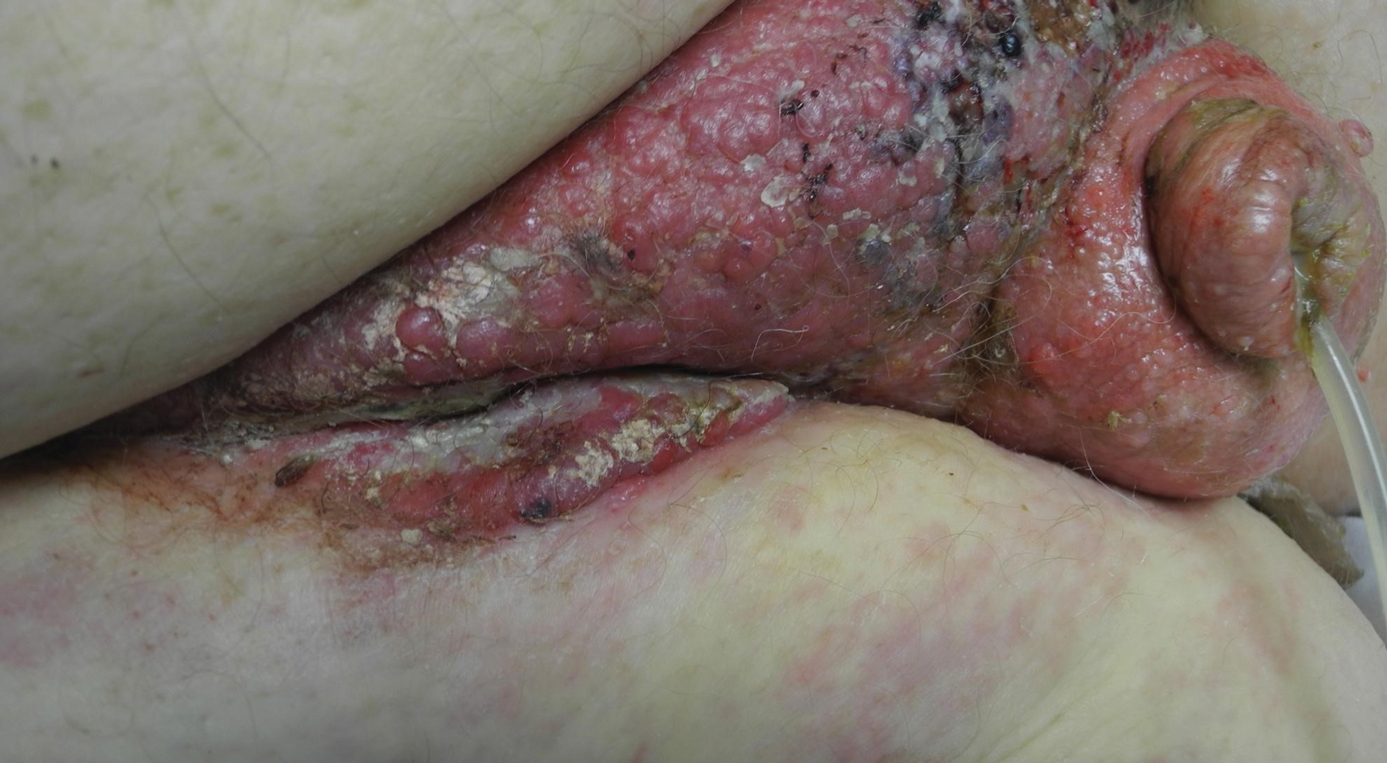
A 67-year-old man presented with a chronic lesion on the groin of 6 weeks' duration. The patient had a history of type 2 diabetes mellitus and colonic adenocarcinoma diagnosed 4 years prior that was treated with a colectomy, radiation therapy, and chemotherapy. Six weeks prior to the current presentation, the patient first sought treatment of swelling, redness, pain, and a bumpy texture on the groin. He was unsuccessfully managed by several physicians including at a long-term care facility where he was admitted and treated for presumed cellulitis. Attempted treatments included a topical antifungal, fluconazole, ciprofloxacin, metronidazole, cefepime, clindamycin, daptomycin, and vancomycin. The affected area continued to worsen along with the patient's overall health. He was transferred to the hospital for more advanced care and was evaluated by inpatient dermatology. Physical examination revealed firm, pink to red-brown, ulcerating papulonodules that coalesced into a large indurated plaque over the pubis, scrotum, penis, and inguinal folds (top). There also were red-violet, indurated plaques on the lower abdomen and bilateral proximal thighs (bottom). Punch biopsies were taken from the indurated area on the left side of the pubis--one for histopathologic evaluation and the other for bacterial, fungal, atypical mycobacterial, and Nocardia tissue cultures.
Today’s top news highlights: ACE inhibitors in COVID patients, fewer AMI admissions, and more
Here are the stories our MDedge editors across specialties think you need to know about today:
Are ACE inhibitors protective in COVID-19?
Older patients with COVID-19 had a lower risk of developing severe illness if they were taking ACE inhibitors, according to a large observational U.S. study. ACE inhibitor use was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. Senior investigator Harlan M. Krumholz, MD, said that while he and his associates think this finding is worthy of further study, “We don’t believe this is enough info to change practice.” The study was published on the MedRxiv preprint server and has not yet been peer reviewed.
READ MORE.
AMI: Admissions drop, deaths rise
In Italy, sharp nationwide decreases in hospitalizations for acute myocardial infarctions (AMIs) during the height of COVID-19 were offset by higher mortality for patients who did present. The study counted AMIs at 54 hospitals nationwide for the week of March 12-19, 2020, and compared that with an equivalent week in 2019 – 319 vs. 618 AMIs, respectively, representing a 48% reduction in hospitalizations. Mortality for ST-segment elevation MI cases more than tripled to 14% during the outbreak, compared with 4% in 2019. “The concern is fewer MIs most likely means people are dying at home or presenting later as this study suggests,” commented Martha Gulati, MD, chief of cardiology at the University of Arizona, Phoenix, who was not involved with the study.
READ MORE.
Prenatal, postpartum screening for depression falls short
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Researchers analyzed self-reported data on postpartum depressive symptoms collected in 2018 by the Pregnancy Risk Assessment Monitoring System. Mental health conditions play a role in approximately 9% of pregnancy-related deaths and not asking about depression represents “missed opportunities to potentially identify and treat women with depression,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Are ACE inhibitors protective in COVID-19?
Older patients with COVID-19 had a lower risk of developing severe illness if they were taking ACE inhibitors, according to a large observational U.S. study. ACE inhibitor use was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. Senior investigator Harlan M. Krumholz, MD, said that while he and his associates think this finding is worthy of further study, “We don’t believe this is enough info to change practice.” The study was published on the MedRxiv preprint server and has not yet been peer reviewed.
READ MORE.
AMI: Admissions drop, deaths rise
In Italy, sharp nationwide decreases in hospitalizations for acute myocardial infarctions (AMIs) during the height of COVID-19 were offset by higher mortality for patients who did present. The study counted AMIs at 54 hospitals nationwide for the week of March 12-19, 2020, and compared that with an equivalent week in 2019 – 319 vs. 618 AMIs, respectively, representing a 48% reduction in hospitalizations. Mortality for ST-segment elevation MI cases more than tripled to 14% during the outbreak, compared with 4% in 2019. “The concern is fewer MIs most likely means people are dying at home or presenting later as this study suggests,” commented Martha Gulati, MD, chief of cardiology at the University of Arizona, Phoenix, who was not involved with the study.
READ MORE.
Prenatal, postpartum screening for depression falls short
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Researchers analyzed self-reported data on postpartum depressive symptoms collected in 2018 by the Pregnancy Risk Assessment Monitoring System. Mental health conditions play a role in approximately 9% of pregnancy-related deaths and not asking about depression represents “missed opportunities to potentially identify and treat women with depression,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Are ACE inhibitors protective in COVID-19?
Older patients with COVID-19 had a lower risk of developing severe illness if they were taking ACE inhibitors, according to a large observational U.S. study. ACE inhibitor use was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. Senior investigator Harlan M. Krumholz, MD, said that while he and his associates think this finding is worthy of further study, “We don’t believe this is enough info to change practice.” The study was published on the MedRxiv preprint server and has not yet been peer reviewed.
READ MORE.
AMI: Admissions drop, deaths rise
In Italy, sharp nationwide decreases in hospitalizations for acute myocardial infarctions (AMIs) during the height of COVID-19 were offset by higher mortality for patients who did present. The study counted AMIs at 54 hospitals nationwide for the week of March 12-19, 2020, and compared that with an equivalent week in 2019 – 319 vs. 618 AMIs, respectively, representing a 48% reduction in hospitalizations. Mortality for ST-segment elevation MI cases more than tripled to 14% during the outbreak, compared with 4% in 2019. “The concern is fewer MIs most likely means people are dying at home or presenting later as this study suggests,” commented Martha Gulati, MD, chief of cardiology at the University of Arizona, Phoenix, who was not involved with the study.
READ MORE.
Prenatal, postpartum screening for depression falls short
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Researchers analyzed self-reported data on postpartum depressive symptoms collected in 2018 by the Pregnancy Risk Assessment Monitoring System. Mental health conditions play a role in approximately 9% of pregnancy-related deaths and not asking about depression represents “missed opportunities to potentially identify and treat women with depression,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
COVID-19 and Mental Health Awareness Month
#howareyoureally challenge seeks to increase access to care
We are months into the COVID-19 crisis, and mental health issues are proving to be rampant. In every crisis, there is opportunity, and this one is no different. The opportunity is clear. For Mental Health Awareness Month and beyond, we must convey a powerful message that mental health is key to our well-being and must be actively addressed. Because almost everyone has felt excess anxiety these last months, we have a unique chance to engage a wider audience.
To address the urgent need, the Mental Health Coalition was formed with the understanding that the mental health crisis is fueled by a pervasive and devastating stigma, preventing millions of individuals from being able to seek the critical treatment they need. Spearheaded by social activist and fashion designer, Kenneth Cole, it is a coalition of leading mental health organizations, brands, celebrities, and advocates who have joined forces to end the stigma surrounding mental health and to change the way people talk about, and care for, mental illness. The group’s mission listed on its website states: “We must increase the conversation around mental health. We must act to end silence, reduce stigma, and engage our community to inspire hope at this essential moment.”
As most of the United States has been under stay-at-home orders, our traditional relationships have been radically disrupted. New types of relationships are forming as we are relying even more on technology to connect us. Social media seems to be on the only “social” we can now safely engage in.
The coalition’s campaign, “#howareyoureally?” is harnessing the power of social media and creating a storytelling platform to allow users to more genuinely share their feelings in these unprecedented times. Celebrities include Whoopi Goldberg, Kendall Jenner, Chris Cuomo, Deepak Chopra, Kesha, and many more have already shared their stories.
“How Are You, Really?” challenges people to answer this question using social media in an open and honest fashion while still providing hope.
The second component of the initiative is to increase access to care, and they have a long list of collaborators, including leading mental health organizations such as the American Foundation for Suicide Prevention, Anxiety and Depression Association of America, Child Mind Institute, Depression and Bipolar Support Alliance, Didi Hirsch Mental Health Services, National Alliance on Mental Illness, and many more.
We have a unique opportunity this Mental Health Awareness Month, and As a community, we must be prepared to meet the escalating needs of our population.
Dr. Ritvo, a psychiatrist with more than 25 years’ experience, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018) and is the founder of the Bekindr Global Initiative, a movement aimed at cultivating kindness in the world. Dr. Ritvo also is the cofounder of the Bold Beauty Project, a nonprofit group that pairs women with disabilities with photographers who create art exhibitions to raise awareness.
#howareyoureally challenge seeks to increase access to care
#howareyoureally challenge seeks to increase access to care
We are months into the COVID-19 crisis, and mental health issues are proving to be rampant. In every crisis, there is opportunity, and this one is no different. The opportunity is clear. For Mental Health Awareness Month and beyond, we must convey a powerful message that mental health is key to our well-being and must be actively addressed. Because almost everyone has felt excess anxiety these last months, we have a unique chance to engage a wider audience.
To address the urgent need, the Mental Health Coalition was formed with the understanding that the mental health crisis is fueled by a pervasive and devastating stigma, preventing millions of individuals from being able to seek the critical treatment they need. Spearheaded by social activist and fashion designer, Kenneth Cole, it is a coalition of leading mental health organizations, brands, celebrities, and advocates who have joined forces to end the stigma surrounding mental health and to change the way people talk about, and care for, mental illness. The group’s mission listed on its website states: “We must increase the conversation around mental health. We must act to end silence, reduce stigma, and engage our community to inspire hope at this essential moment.”
As most of the United States has been under stay-at-home orders, our traditional relationships have been radically disrupted. New types of relationships are forming as we are relying even more on technology to connect us. Social media seems to be on the only “social” we can now safely engage in.
The coalition’s campaign, “#howareyoureally?” is harnessing the power of social media and creating a storytelling platform to allow users to more genuinely share their feelings in these unprecedented times. Celebrities include Whoopi Goldberg, Kendall Jenner, Chris Cuomo, Deepak Chopra, Kesha, and many more have already shared their stories.
“How Are You, Really?” challenges people to answer this question using social media in an open and honest fashion while still providing hope.
The second component of the initiative is to increase access to care, and they have a long list of collaborators, including leading mental health organizations such as the American Foundation for Suicide Prevention, Anxiety and Depression Association of America, Child Mind Institute, Depression and Bipolar Support Alliance, Didi Hirsch Mental Health Services, National Alliance on Mental Illness, and many more.
We have a unique opportunity this Mental Health Awareness Month, and As a community, we must be prepared to meet the escalating needs of our population.
Dr. Ritvo, a psychiatrist with more than 25 years’ experience, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018) and is the founder of the Bekindr Global Initiative, a movement aimed at cultivating kindness in the world. Dr. Ritvo also is the cofounder of the Bold Beauty Project, a nonprofit group that pairs women with disabilities with photographers who create art exhibitions to raise awareness.
We are months into the COVID-19 crisis, and mental health issues are proving to be rampant. In every crisis, there is opportunity, and this one is no different. The opportunity is clear. For Mental Health Awareness Month and beyond, we must convey a powerful message that mental health is key to our well-being and must be actively addressed. Because almost everyone has felt excess anxiety these last months, we have a unique chance to engage a wider audience.
To address the urgent need, the Mental Health Coalition was formed with the understanding that the mental health crisis is fueled by a pervasive and devastating stigma, preventing millions of individuals from being able to seek the critical treatment they need. Spearheaded by social activist and fashion designer, Kenneth Cole, it is a coalition of leading mental health organizations, brands, celebrities, and advocates who have joined forces to end the stigma surrounding mental health and to change the way people talk about, and care for, mental illness. The group’s mission listed on its website states: “We must increase the conversation around mental health. We must act to end silence, reduce stigma, and engage our community to inspire hope at this essential moment.”
As most of the United States has been under stay-at-home orders, our traditional relationships have been radically disrupted. New types of relationships are forming as we are relying even more on technology to connect us. Social media seems to be on the only “social” we can now safely engage in.
The coalition’s campaign, “#howareyoureally?” is harnessing the power of social media and creating a storytelling platform to allow users to more genuinely share their feelings in these unprecedented times. Celebrities include Whoopi Goldberg, Kendall Jenner, Chris Cuomo, Deepak Chopra, Kesha, and many more have already shared their stories.
“How Are You, Really?” challenges people to answer this question using social media in an open and honest fashion while still providing hope.
The second component of the initiative is to increase access to care, and they have a long list of collaborators, including leading mental health organizations such as the American Foundation for Suicide Prevention, Anxiety and Depression Association of America, Child Mind Institute, Depression and Bipolar Support Alliance, Didi Hirsch Mental Health Services, National Alliance on Mental Illness, and many more.
We have a unique opportunity this Mental Health Awareness Month, and As a community, we must be prepared to meet the escalating needs of our population.
Dr. Ritvo, a psychiatrist with more than 25 years’ experience, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018) and is the founder of the Bekindr Global Initiative, a movement aimed at cultivating kindness in the world. Dr. Ritvo also is the cofounder of the Bold Beauty Project, a nonprofit group that pairs women with disabilities with photographers who create art exhibitions to raise awareness.
Oncologists’ income and satisfaction are up
Oncologists continue to rank above the middle range for all specialties in annual compensation for physicians, according to findings from the newly released Medscape Oncologist Compensation Report 2020.
The average earnings for oncologists who participated in the survey was $377,000, which was a 5% increase from the $359,000 reported for 2018.
Just over two-thirds (67%) of oncologists reported that they felt that they were fairly compensated, which is quite a jump from 53% last year.
In addition, oncologists appear to be very satisfied with their profession. Similar to last year’s findings, 84% said they would choose medicine again, and 96% said they would choose the specialty of oncology again.
Earning in top third of all specialties
The average annual earnings reported by oncologists put this specialty in eleventh place among 29 specialties. Orthopedic specialists remain at the head of the list, with estimated earnings of $511,000, followed by plastic surgeons ($479,000), otolaryngologists ($455,000), and cardiologists ($438,000), according to Medscape’s compensation report, which included responses from 17,461 physicians in over 30 specialties.
At the bottom of the estimated earnings list were public health and preventive medicine doctors and pediatricians. For both specialties, the reported annual earnings was $232,000. Family medicine specialists were only marginally higher at $234,000.
Radiologists ($427,000), gastroenterologists ($419,000), and urologists ($417,000) all reported higher earnings than oncologists, whereas neurologists, at $280,000, rheumatologists, at $262,000, and internal medicine physicians, at $251,000, earned less.
The report also found that gender disparities in income persist, with male oncologists earning 17% more than their female colleagues. The gender gap in oncology is somewhat less than that seen for all specialties combined, in which men earned 31% more than women, similar to last year’s figure of 33%.
Male oncologists reported spending 38.8 hours per week seeing patients, compared with 34.9 hours reported by female oncologists. This could be a factor contributing to the gender pay disparity. Overall, the average amount of time seeing patients was 37.9 hours per week.
Frustrations with paperwork and denied claims
Surveyed oncologists cited some of the frustrations they are facing, such as spending nearly 17 hours a week on paperwork and administrative tasks. They reported that 16% of claims are denied or have to be resubmitted. As for the most challenging part of the job, oncologists (22%), similar to physicians overall (26%), found that having so many rules and regulations takes first place, followed by working with electronic health record systems (20%), difficulties getting fair reimbursement (19%), having to work long hours (12%), and dealing with difficult patients (8%). Few oncologists were concerned about lawsuits (4%), and 4% reported that there were no challenges.
Oncologists reported that the most rewarding part of their job was gratitude/relationships with patients (31%), followed by knowing that they are making the world a better place (27%). After that, oncologists agreed with statements about being very good at what they do/finding answers/diagnoses (22%), having pride in being a doctor (9%), and making good money at a job they like (8%).
Other key findings
Other key findings from the Medscape Oncologist Compensation Report 2020 included the following:
- Regarding payment models, 80% take insurance, 41% are in fee-for-service arrangements, and 18% are in accountable care organizations (21%). Only 3% are in direct primary care, and 1% are cash-only practices or have a concierge practice.
- 65% of oncologists state that they will continue taking new and current Medicare/Medicaid patients. None said that they would not take on new Medicare/Medicaid patients, and 35% remain undecided. These numbers differed from physicians overall; 73% of all physicians surveyed said they would continue taking new/current Medicare/Medicaid patients, 6% said that will not take on new Medicare patients, and 4% said they will not take new Medicaid patients. In addition, 3% and 2% said that they would stop treating some or all of their Medicare and Medicaid patients, respectively.
- About half (51%) of oncologists use nurse practitioners, about a third (34%) use physician assistants, and 37% use neither. This was about the same as physicians overall.
- A larger percentage of oncologists (38%) expect to participate in MIPS (merit-based incentive payment system), and only 8% expect to participate in APMs (alternative payment models). This was similar to the findings for physicians overall, with more than one-third (37%) expecting to participate in MIPS and 9% planning to take part in APMs.
Impact of COVID-19 pandemic
The Medscape compensation reports also gives a glimpse of the impact the COVID-19 pandemic is having on physician compensation.
Since the beginning of the pandemic, practices have reported a 55% decrease in revenue and a 60% drop in patient volume. Physician practices and hospitals have laid off or furloughed personnel and have cut pay, and 9% of practices have closed their doors, at least for the time being.
A total of 43,000 health care workers were laid off in March, the report notes.
The findings tie in with those reported elsewhere. For example, a survey conducted by the Medical Group Management Association, which was reported by Medscape Medical News, found that 97% of physician practices have experienced negative financial effects directly or indirectly related to COVID-19.
Specialties were hard hit, especially those that rely on elective procedures, such as dermatology and cardiology. Oncology care has also been disrupted. For example, a survey conducted by the American Cancer Society Cancer Action Network found that half of the cancer patients and survivors who responded reported changes, delays, or disruptions to the care they were receiving.
This article first appeared on Medscape.com.
Oncologists continue to rank above the middle range for all specialties in annual compensation for physicians, according to findings from the newly released Medscape Oncologist Compensation Report 2020.
The average earnings for oncologists who participated in the survey was $377,000, which was a 5% increase from the $359,000 reported for 2018.
Just over two-thirds (67%) of oncologists reported that they felt that they were fairly compensated, which is quite a jump from 53% last year.
In addition, oncologists appear to be very satisfied with their profession. Similar to last year’s findings, 84% said they would choose medicine again, and 96% said they would choose the specialty of oncology again.
Earning in top third of all specialties
The average annual earnings reported by oncologists put this specialty in eleventh place among 29 specialties. Orthopedic specialists remain at the head of the list, with estimated earnings of $511,000, followed by plastic surgeons ($479,000), otolaryngologists ($455,000), and cardiologists ($438,000), according to Medscape’s compensation report, which included responses from 17,461 physicians in over 30 specialties.

At the bottom of the estimated earnings list were public health and preventive medicine doctors and pediatricians. For both specialties, the reported annual earnings was $232,000. Family medicine specialists were only marginally higher at $234,000.
Radiologists ($427,000), gastroenterologists ($419,000), and urologists ($417,000) all reported higher earnings than oncologists, whereas neurologists, at $280,000, rheumatologists, at $262,000, and internal medicine physicians, at $251,000, earned less.
The report also found that gender disparities in income persist, with male oncologists earning 17% more than their female colleagues. The gender gap in oncology is somewhat less than that seen for all specialties combined, in which men earned 31% more than women, similar to last year’s figure of 33%.
Male oncologists reported spending 38.8 hours per week seeing patients, compared with 34.9 hours reported by female oncologists. This could be a factor contributing to the gender pay disparity. Overall, the average amount of time seeing patients was 37.9 hours per week.
Frustrations with paperwork and denied claims
Surveyed oncologists cited some of the frustrations they are facing, such as spending nearly 17 hours a week on paperwork and administrative tasks. They reported that 16% of claims are denied or have to be resubmitted. As for the most challenging part of the job, oncologists (22%), similar to physicians overall (26%), found that having so many rules and regulations takes first place, followed by working with electronic health record systems (20%), difficulties getting fair reimbursement (19%), having to work long hours (12%), and dealing with difficult patients (8%). Few oncologists were concerned about lawsuits (4%), and 4% reported that there were no challenges.
Oncologists reported that the most rewarding part of their job was gratitude/relationships with patients (31%), followed by knowing that they are making the world a better place (27%). After that, oncologists agreed with statements about being very good at what they do/finding answers/diagnoses (22%), having pride in being a doctor (9%), and making good money at a job they like (8%).
Other key findings
Other key findings from the Medscape Oncologist Compensation Report 2020 included the following:
- Regarding payment models, 80% take insurance, 41% are in fee-for-service arrangements, and 18% are in accountable care organizations (21%). Only 3% are in direct primary care, and 1% are cash-only practices or have a concierge practice.
- 65% of oncologists state that they will continue taking new and current Medicare/Medicaid patients. None said that they would not take on new Medicare/Medicaid patients, and 35% remain undecided. These numbers differed from physicians overall; 73% of all physicians surveyed said they would continue taking new/current Medicare/Medicaid patients, 6% said that will not take on new Medicare patients, and 4% said they will not take new Medicaid patients. In addition, 3% and 2% said that they would stop treating some or all of their Medicare and Medicaid patients, respectively.
- About half (51%) of oncologists use nurse practitioners, about a third (34%) use physician assistants, and 37% use neither. This was about the same as physicians overall.
- A larger percentage of oncologists (38%) expect to participate in MIPS (merit-based incentive payment system), and only 8% expect to participate in APMs (alternative payment models). This was similar to the findings for physicians overall, with more than one-third (37%) expecting to participate in MIPS and 9% planning to take part in APMs.
Impact of COVID-19 pandemic
The Medscape compensation reports also gives a glimpse of the impact the COVID-19 pandemic is having on physician compensation.
Since the beginning of the pandemic, practices have reported a 55% decrease in revenue and a 60% drop in patient volume. Physician practices and hospitals have laid off or furloughed personnel and have cut pay, and 9% of practices have closed their doors, at least for the time being.
A total of 43,000 health care workers were laid off in March, the report notes.
The findings tie in with those reported elsewhere. For example, a survey conducted by the Medical Group Management Association, which was reported by Medscape Medical News, found that 97% of physician practices have experienced negative financial effects directly or indirectly related to COVID-19.
Specialties were hard hit, especially those that rely on elective procedures, such as dermatology and cardiology. Oncology care has also been disrupted. For example, a survey conducted by the American Cancer Society Cancer Action Network found that half of the cancer patients and survivors who responded reported changes, delays, or disruptions to the care they were receiving.
This article first appeared on Medscape.com.
Oncologists continue to rank above the middle range for all specialties in annual compensation for physicians, according to findings from the newly released Medscape Oncologist Compensation Report 2020.
The average earnings for oncologists who participated in the survey was $377,000, which was a 5% increase from the $359,000 reported for 2018.
Just over two-thirds (67%) of oncologists reported that they felt that they were fairly compensated, which is quite a jump from 53% last year.
In addition, oncologists appear to be very satisfied with their profession. Similar to last year’s findings, 84% said they would choose medicine again, and 96% said they would choose the specialty of oncology again.
Earning in top third of all specialties
The average annual earnings reported by oncologists put this specialty in eleventh place among 29 specialties. Orthopedic specialists remain at the head of the list, with estimated earnings of $511,000, followed by plastic surgeons ($479,000), otolaryngologists ($455,000), and cardiologists ($438,000), according to Medscape’s compensation report, which included responses from 17,461 physicians in over 30 specialties.

At the bottom of the estimated earnings list were public health and preventive medicine doctors and pediatricians. For both specialties, the reported annual earnings was $232,000. Family medicine specialists were only marginally higher at $234,000.
Radiologists ($427,000), gastroenterologists ($419,000), and urologists ($417,000) all reported higher earnings than oncologists, whereas neurologists, at $280,000, rheumatologists, at $262,000, and internal medicine physicians, at $251,000, earned less.
The report also found that gender disparities in income persist, with male oncologists earning 17% more than their female colleagues. The gender gap in oncology is somewhat less than that seen for all specialties combined, in which men earned 31% more than women, similar to last year’s figure of 33%.
Male oncologists reported spending 38.8 hours per week seeing patients, compared with 34.9 hours reported by female oncologists. This could be a factor contributing to the gender pay disparity. Overall, the average amount of time seeing patients was 37.9 hours per week.
Frustrations with paperwork and denied claims
Surveyed oncologists cited some of the frustrations they are facing, such as spending nearly 17 hours a week on paperwork and administrative tasks. They reported that 16% of claims are denied or have to be resubmitted. As for the most challenging part of the job, oncologists (22%), similar to physicians overall (26%), found that having so many rules and regulations takes first place, followed by working with electronic health record systems (20%), difficulties getting fair reimbursement (19%), having to work long hours (12%), and dealing with difficult patients (8%). Few oncologists were concerned about lawsuits (4%), and 4% reported that there were no challenges.
Oncologists reported that the most rewarding part of their job was gratitude/relationships with patients (31%), followed by knowing that they are making the world a better place (27%). After that, oncologists agreed with statements about being very good at what they do/finding answers/diagnoses (22%), having pride in being a doctor (9%), and making good money at a job they like (8%).
Other key findings
Other key findings from the Medscape Oncologist Compensation Report 2020 included the following:
- Regarding payment models, 80% take insurance, 41% are in fee-for-service arrangements, and 18% are in accountable care organizations (21%). Only 3% are in direct primary care, and 1% are cash-only practices or have a concierge practice.
- 65% of oncologists state that they will continue taking new and current Medicare/Medicaid patients. None said that they would not take on new Medicare/Medicaid patients, and 35% remain undecided. These numbers differed from physicians overall; 73% of all physicians surveyed said they would continue taking new/current Medicare/Medicaid patients, 6% said that will not take on new Medicare patients, and 4% said they will not take new Medicaid patients. In addition, 3% and 2% said that they would stop treating some or all of their Medicare and Medicaid patients, respectively.
- About half (51%) of oncologists use nurse practitioners, about a third (34%) use physician assistants, and 37% use neither. This was about the same as physicians overall.
- A larger percentage of oncologists (38%) expect to participate in MIPS (merit-based incentive payment system), and only 8% expect to participate in APMs (alternative payment models). This was similar to the findings for physicians overall, with more than one-third (37%) expecting to participate in MIPS and 9% planning to take part in APMs.
Impact of COVID-19 pandemic
The Medscape compensation reports also gives a glimpse of the impact the COVID-19 pandemic is having on physician compensation.
Since the beginning of the pandemic, practices have reported a 55% decrease in revenue and a 60% drop in patient volume. Physician practices and hospitals have laid off or furloughed personnel and have cut pay, and 9% of practices have closed their doors, at least for the time being.
A total of 43,000 health care workers were laid off in March, the report notes.
The findings tie in with those reported elsewhere. For example, a survey conducted by the Medical Group Management Association, which was reported by Medscape Medical News, found that 97% of physician practices have experienced negative financial effects directly or indirectly related to COVID-19.
Specialties were hard hit, especially those that rely on elective procedures, such as dermatology and cardiology. Oncology care has also been disrupted. For example, a survey conducted by the American Cancer Society Cancer Action Network found that half of the cancer patients and survivors who responded reported changes, delays, or disruptions to the care they were receiving.
This article first appeared on Medscape.com.
FDA expands approval of atezolizumab in NSCLC
The Food and Drug Administration has expanded the approved indication for atezolizumab (Tecentriq) in patients with non–small cell lung cancer (NSCLC).
Atezolizumab is now approved as first-line monotherapy for adults with metastatic NSCLC whose tumors are EGFR and ALK wild-type but have high PD-L1 expression (PD-L1 stained ≥50% of tumor cells or PD-L1 stained tumor-infiltrating immune cells covering ≥10% of the tumor area).
The FDA also approved the VENTANA PD-L1 (SP142) Assay as a companion diagnostic to identify patients with NSCLC who are eligible for treatment with atezolizumab.
The drug was evaluated in the IMpower110 trial (NCT02409342), which enrolled patients with stage IV, PD-L1–positive (tumor cells [TC] ≥1% or immune cells [IC] ≥1%) NSCLC who had received no prior chemotherapy for metastatic disease.
The patients were randomized to receive atezolizumab at 1,200 mg every 3 weeks (n = 286) or platinum-based chemotherapy (n = 263), which consisted of carboplatin or cisplatin with either pemetrexed or gemcitabine, until disease progression or unacceptable toxicity.
Overall survival was superior in the atezolizumab arm, but only among patients with high PD-L1 expression (TC ≥50% or IC ≥10%). The median overall survival was 20.2 months among PD-L1–high patients in the atezolizumab arm and 13.1 months among PD-L1–high patients in the chemotherapy arm (hazard ratio, 0.59; P = .0106). There was no significant difference in overall survival between the treatment arms for patients in the other two PD-L1 subgroups – TC ≥5% or IC ≥5% and TC ≥1% or IC ≥1%.
Serious adverse events occurred in 28% of patients receiving atezolizumab. The most frequent of these were pneumonia (2.8%), chronic obstructive pulmonary disease (2.1%), and pneumonitis (2.1%). Fatal adverse events in the atezolizumab arm included unexplained death, aspiration, chronic obstructive pulmonary disease, pulmonary embolism, acute myocardial infarction, cardiac arrest, mechanical ileus, sepsis, cerebral infraction, and device occlusion (one patient each).
For more details on atezolizumab, see the full prescribing information.
The FDA has granted the approval of atezolizumab to Genentech and the approval of the VENTANA PD-L1 (SP142) Assay to Ventana Medical Systems.
The Food and Drug Administration has expanded the approved indication for atezolizumab (Tecentriq) in patients with non–small cell lung cancer (NSCLC).
Atezolizumab is now approved as first-line monotherapy for adults with metastatic NSCLC whose tumors are EGFR and ALK wild-type but have high PD-L1 expression (PD-L1 stained ≥50% of tumor cells or PD-L1 stained tumor-infiltrating immune cells covering ≥10% of the tumor area).
The FDA also approved the VENTANA PD-L1 (SP142) Assay as a companion diagnostic to identify patients with NSCLC who are eligible for treatment with atezolizumab.
The drug was evaluated in the IMpower110 trial (NCT02409342), which enrolled patients with stage IV, PD-L1–positive (tumor cells [TC] ≥1% or immune cells [IC] ≥1%) NSCLC who had received no prior chemotherapy for metastatic disease.
The patients were randomized to receive atezolizumab at 1,200 mg every 3 weeks (n = 286) or platinum-based chemotherapy (n = 263), which consisted of carboplatin or cisplatin with either pemetrexed or gemcitabine, until disease progression or unacceptable toxicity.
Overall survival was superior in the atezolizumab arm, but only among patients with high PD-L1 expression (TC ≥50% or IC ≥10%). The median overall survival was 20.2 months among PD-L1–high patients in the atezolizumab arm and 13.1 months among PD-L1–high patients in the chemotherapy arm (hazard ratio, 0.59; P = .0106). There was no significant difference in overall survival between the treatment arms for patients in the other two PD-L1 subgroups – TC ≥5% or IC ≥5% and TC ≥1% or IC ≥1%.
Serious adverse events occurred in 28% of patients receiving atezolizumab. The most frequent of these were pneumonia (2.8%), chronic obstructive pulmonary disease (2.1%), and pneumonitis (2.1%). Fatal adverse events in the atezolizumab arm included unexplained death, aspiration, chronic obstructive pulmonary disease, pulmonary embolism, acute myocardial infarction, cardiac arrest, mechanical ileus, sepsis, cerebral infraction, and device occlusion (one patient each).
For more details on atezolizumab, see the full prescribing information.
The FDA has granted the approval of atezolizumab to Genentech and the approval of the VENTANA PD-L1 (SP142) Assay to Ventana Medical Systems.
The Food and Drug Administration has expanded the approved indication for atezolizumab (Tecentriq) in patients with non–small cell lung cancer (NSCLC).
Atezolizumab is now approved as first-line monotherapy for adults with metastatic NSCLC whose tumors are EGFR and ALK wild-type but have high PD-L1 expression (PD-L1 stained ≥50% of tumor cells or PD-L1 stained tumor-infiltrating immune cells covering ≥10% of the tumor area).
The FDA also approved the VENTANA PD-L1 (SP142) Assay as a companion diagnostic to identify patients with NSCLC who are eligible for treatment with atezolizumab.
The drug was evaluated in the IMpower110 trial (NCT02409342), which enrolled patients with stage IV, PD-L1–positive (tumor cells [TC] ≥1% or immune cells [IC] ≥1%) NSCLC who had received no prior chemotherapy for metastatic disease.
The patients were randomized to receive atezolizumab at 1,200 mg every 3 weeks (n = 286) or platinum-based chemotherapy (n = 263), which consisted of carboplatin or cisplatin with either pemetrexed or gemcitabine, until disease progression or unacceptable toxicity.
Overall survival was superior in the atezolizumab arm, but only among patients with high PD-L1 expression (TC ≥50% or IC ≥10%). The median overall survival was 20.2 months among PD-L1–high patients in the atezolizumab arm and 13.1 months among PD-L1–high patients in the chemotherapy arm (hazard ratio, 0.59; P = .0106). There was no significant difference in overall survival between the treatment arms for patients in the other two PD-L1 subgroups – TC ≥5% or IC ≥5% and TC ≥1% or IC ≥1%.
Serious adverse events occurred in 28% of patients receiving atezolizumab. The most frequent of these were pneumonia (2.8%), chronic obstructive pulmonary disease (2.1%), and pneumonitis (2.1%). Fatal adverse events in the atezolizumab arm included unexplained death, aspiration, chronic obstructive pulmonary disease, pulmonary embolism, acute myocardial infarction, cardiac arrest, mechanical ileus, sepsis, cerebral infraction, and device occlusion (one patient each).
For more details on atezolizumab, see the full prescribing information.
The FDA has granted the approval of atezolizumab to Genentech and the approval of the VENTANA PD-L1 (SP142) Assay to Ventana Medical Systems.
The SHM 2019 Chapter Excellence Awards
The Society of Hospital Medicine is proud to recognize its chapters for their hard work and dedication in 2019 through Chapter Excellence Awards. Each year, chapters strive to demonstrate growth, sustenance, and innovation within their chapter activities, which are then applauded for their successes throughout the subsequent year. In 2019, a new Bronze category was established, for a total of four Status Awards that chapters can earn.
Please join SHM in congratulating the following chapters on their year of success in 2019!
Outstanding Chapter of the Year
The Outstanding Chapter of the Year Award goes to one chapter who exemplifies high performance, going above and beyond the basic chapter requirements. The recipient of the Outstanding Chapter of the Year Award for 2019 is the Wiregrass Chapter of SHM. The chapter has a strong and engaged leadership which includes representation at all levels of the hospital medicine team, including physician hospitalists, advanced care provider hospitalists, practice administrators, nurses, residents, and medical students.
In the last year, the Wiregrass leadership team has organized programs and events to cater to and engage all the chapter’s members. This includes a variety of innovative ideas that catered toward medical education, health care provider well-being, engagement, mentorship, and community involvement.
The SHM Wiregrass Chapter’s biggest accomplishment in 2019 was the creation of an exchange program for physician and advanced practice provider hospitalists between the SHM New Mexico Chapter and the SHM Wiregrass Chapter. This idea first arose at HM19, where the chapter leaders had met during a networking event and debated the role of clinician wellbeing, quality of medical education, and faculty development to individual hospital medicine group (HMG) practice styles.
Clinician well-being is the prerequisite to the triple aim of improving the health of populations, enhancing the patient experience, and reducing the cost of care. Each HMG faces similar challenges but approaches to solving them vary. Professional challenges can affect the well-being of the individual clinicians. Having interinstitutional exchange programs provides a platform to exchange ideas and establish mentors. Also, the quality of medical education is directly linked to the quality of faculty development. Improving the quality of medical education requires a multifaceted approach by highly developed faculty. The complex factors affecting medical education and faculty development are further complicated by geographic location, patient characteristics, and professional growth opportunities. Overcoming these obstacles requires an innovative and collaborative approach. Although faculty exchanges are common in academic medicine, they are not commonly attempted with HMGs.
Hospitalists are responsible for a significant part of inpatient training for residents, medical students, and nurse practitioners/physician assistants (NPs/PAs), but their faculty training can vary based on location. Being a young specialty, only 2 decades old, hospital medicine is still evolving and incorporating NP/PA and physician hospitalists in varied practice models. Each HMG addresses common obstacles differently based on their culture and practice styles. The chapter leaders determined an exchange program would afford the opportunity for visiting faculty members to experience these differences. This emphasized the role and importance of exchanging ideas and contemplated a solution to benefit more practicing hospitalists.
The chapter leaders researched the characteristics of individual academic HMGs and structured a tailored faculty exchange involving physicians and NPs/PAs. During the exchange program planning, the visiting faculty itinerary was tailored to a well-planned agenda for 1 week, with separate tracks for physicians and NPs/PAs giving increased access to their individual peer practice styles. Additionally, the visiting faculty had meetings and discussions with HMG and hospital leadership, to specifically address each visiting faculty institution’s challenges. The overall goal of this exchange program was to promote cross-institutional collaboration, increase engagement, improve medical education through faculty development, and improve the quality of care. The focus of the exchange program was to share ideas and innovation and learn the approaches to unique challenges at each institution. Out of this also came collaboration and mentoring opportunities.
The evaluation process of the exchange involved interviews, a survey, and the establishment of shared QI projects in mutual areas of challenge. The survey provided feedback, lessons learned from the exchange, and areas to be improved. Collaborative QI projects currently underway as a result of the exchange include paging etiquette, quality of sleep for hospitalized patients, and onboarding of NPs/PAs in HMGs.
This innovation addressed faculty development and medical education via clinician well-being. The physician and NP/PA Faculty Exchange was an essential and meaningful innovation that resulted in increased SHM member engagement, cross-institutional collaboration, networking, and mentorship.
Additional projects that the SHM Wiregrass Chapter successfully implemented in 2019 include a “Women in Medicine” event that recognized women physician and advanced practice provider hospitalist leaders, a poster competition that expanded its research, clinical vignettes, and quality categories to include a fourth category of innovation, featuring 75 posters. Additionally, the chapter held a policy meeting with six Alabama state legislators, creating new channels of collaboration between the legislators and the chapter. Lastly, the chapter held a successful community event and launched a mentor program targeting medical students and residents.
Rising Star Chapter
The Rising Star Chapter Award goes to one chapter who has been active for 2 years or less, who in the past 12 months have made improvements to their leadership, stability and growth, and membership. The recipient of the Rising Star Chapter Award for 2019 is the Blue Ridge Chapter of SHM, which has made significant strides to develop since its launch in the fall of 2018. The chapter represents counties in northwest Tennessee, southwest Virginia, and western North Carolina.
The chapter held three meetings in 2019 which were well attended by hospitalists, residents/fellows, administrators, advanced practice providers, and nurses. On average, attendees from five to six different hospitalist groups are represented. The chapter hosted both Dr. Chris Frost, immediate past president of the SHM board of directors, and Dr. Ron Greeno, a past president of the SHM board of directors.
The SHM Blue Ridge Chapter has collaborated with both the ACP Tennessee Chapter and the Healthcare MBA program at Haslam College of Business at the University of Tennessee.
The chapter leadership regularly attends local medical residency programs at noon conferences to attract and recruit young physicians into chapter activities. Overall, the chapter has seen a growth in membership in 2019. The Blue Ridge Chapter is an active, enthusiastic chapter that is rapidly growing and thriving.
Outstanding Membership Recruitment and Retention
The Outstanding Membership Recruitment and Retention Award is a new exemplary award for 2019 that goes to one chapter who has gone above and beyond to implement initiatives to recruit and retain SHM members in their chapter. The recipient of the Outstanding Membership Recruitment and Retention Award for 2019 is the Western Massachusetts Chapter of SHM, which has done outstanding work to recruit and retain the membership. In 2019, the SHM membership in the chapter grew by 24%. The chapter utilized Chapter Development Funds to launch new initiatives to conduct outreach to nonmember hospitalists in the community and invite them to meetings to obtain the SHM experience. Additionally, the chapter encouraged residents to join and get involved by hosting a poster competition.
The Western Massachusetts Chapter focused on being innovative, inclusive, and creative to retain their existing meetings. For example, the chapter hosted a new “Jeopardy Session” event that featured a nontraditional jeopardy game that attracted a large attendance including local residents. Additionally, the chapter insured that all clinical and nonclinical members of the hospital medicine team were included and encouraged to participate in all chapter meetings. Lastly, the chapter launched a local awards program to recognize senior hospitalist and early career hospitalist who contributed to chapter development.
Most Engaged Chapter Leader
The Most Engaged Chapter Leader Award is a new exemplary award for 2019 that goes to one chapter leader or district chair who is either nominated or self-nominated and has demonstrated how they or their nominee has gone above and beyond in the past year to grow and sustain their chapter and/or district and continues to carry out the SHM mission. The recipient of the Most Engaged Chapter Leader Award for 2019 goes to Thérèse Franco, MD, SFHM, president of the Pacific Northwest Chapter.
Dr. Franco has served as the chapter’s president for 2 years and has served on the SHM Chapter Support Committee for 3 years. She has previously participated as a mentor in the glycemic control mentored implementation program, and as chair and cochair of the RIV contest. She continues to review abstracts, volunteer as a judge and offer local education on glycemic control through the Washington State Hospital Association, promoting SHM’s work there. One of Dr. Franco’s core strengths has been effective collaboration with past leaders (such as Rachel Thompson, MD, and Kimberly Bell, MD), future leaders, and other organizations (such as the Washington State Medical Association and the King County Medical Association). Dr. Franco has recruited an outstanding leadership team and new advisory committee for the Pacific Northwest Chapter, resulting a fantastic year of growth, innovation, and development.
The Society of Hospital Medicine is proud to recognize its chapters for their hard work and dedication in 2019 through Chapter Excellence Awards. Each year, chapters strive to demonstrate growth, sustenance, and innovation within their chapter activities, which are then applauded for their successes throughout the subsequent year. In 2019, a new Bronze category was established, for a total of four Status Awards that chapters can earn.
Please join SHM in congratulating the following chapters on their year of success in 2019!
Outstanding Chapter of the Year
The Outstanding Chapter of the Year Award goes to one chapter who exemplifies high performance, going above and beyond the basic chapter requirements. The recipient of the Outstanding Chapter of the Year Award for 2019 is the Wiregrass Chapter of SHM. The chapter has a strong and engaged leadership which includes representation at all levels of the hospital medicine team, including physician hospitalists, advanced care provider hospitalists, practice administrators, nurses, residents, and medical students.
In the last year, the Wiregrass leadership team has organized programs and events to cater to and engage all the chapter’s members. This includes a variety of innovative ideas that catered toward medical education, health care provider well-being, engagement, mentorship, and community involvement.
The SHM Wiregrass Chapter’s biggest accomplishment in 2019 was the creation of an exchange program for physician and advanced practice provider hospitalists between the SHM New Mexico Chapter and the SHM Wiregrass Chapter. This idea first arose at HM19, where the chapter leaders had met during a networking event and debated the role of clinician wellbeing, quality of medical education, and faculty development to individual hospital medicine group (HMG) practice styles.
Clinician well-being is the prerequisite to the triple aim of improving the health of populations, enhancing the patient experience, and reducing the cost of care. Each HMG faces similar challenges but approaches to solving them vary. Professional challenges can affect the well-being of the individual clinicians. Having interinstitutional exchange programs provides a platform to exchange ideas and establish mentors. Also, the quality of medical education is directly linked to the quality of faculty development. Improving the quality of medical education requires a multifaceted approach by highly developed faculty. The complex factors affecting medical education and faculty development are further complicated by geographic location, patient characteristics, and professional growth opportunities. Overcoming these obstacles requires an innovative and collaborative approach. Although faculty exchanges are common in academic medicine, they are not commonly attempted with HMGs.
Hospitalists are responsible for a significant part of inpatient training for residents, medical students, and nurse practitioners/physician assistants (NPs/PAs), but their faculty training can vary based on location. Being a young specialty, only 2 decades old, hospital medicine is still evolving and incorporating NP/PA and physician hospitalists in varied practice models. Each HMG addresses common obstacles differently based on their culture and practice styles. The chapter leaders determined an exchange program would afford the opportunity for visiting faculty members to experience these differences. This emphasized the role and importance of exchanging ideas and contemplated a solution to benefit more practicing hospitalists.
The chapter leaders researched the characteristics of individual academic HMGs and structured a tailored faculty exchange involving physicians and NPs/PAs. During the exchange program planning, the visiting faculty itinerary was tailored to a well-planned agenda for 1 week, with separate tracks for physicians and NPs/PAs giving increased access to their individual peer practice styles. Additionally, the visiting faculty had meetings and discussions with HMG and hospital leadership, to specifically address each visiting faculty institution’s challenges. The overall goal of this exchange program was to promote cross-institutional collaboration, increase engagement, improve medical education through faculty development, and improve the quality of care. The focus of the exchange program was to share ideas and innovation and learn the approaches to unique challenges at each institution. Out of this also came collaboration and mentoring opportunities.
The evaluation process of the exchange involved interviews, a survey, and the establishment of shared QI projects in mutual areas of challenge. The survey provided feedback, lessons learned from the exchange, and areas to be improved. Collaborative QI projects currently underway as a result of the exchange include paging etiquette, quality of sleep for hospitalized patients, and onboarding of NPs/PAs in HMGs.
This innovation addressed faculty development and medical education via clinician well-being. The physician and NP/PA Faculty Exchange was an essential and meaningful innovation that resulted in increased SHM member engagement, cross-institutional collaboration, networking, and mentorship.
Additional projects that the SHM Wiregrass Chapter successfully implemented in 2019 include a “Women in Medicine” event that recognized women physician and advanced practice provider hospitalist leaders, a poster competition that expanded its research, clinical vignettes, and quality categories to include a fourth category of innovation, featuring 75 posters. Additionally, the chapter held a policy meeting with six Alabama state legislators, creating new channels of collaboration between the legislators and the chapter. Lastly, the chapter held a successful community event and launched a mentor program targeting medical students and residents.
Rising Star Chapter
The Rising Star Chapter Award goes to one chapter who has been active for 2 years or less, who in the past 12 months have made improvements to their leadership, stability and growth, and membership. The recipient of the Rising Star Chapter Award for 2019 is the Blue Ridge Chapter of SHM, which has made significant strides to develop since its launch in the fall of 2018. The chapter represents counties in northwest Tennessee, southwest Virginia, and western North Carolina.
The chapter held three meetings in 2019 which were well attended by hospitalists, residents/fellows, administrators, advanced practice providers, and nurses. On average, attendees from five to six different hospitalist groups are represented. The chapter hosted both Dr. Chris Frost, immediate past president of the SHM board of directors, and Dr. Ron Greeno, a past president of the SHM board of directors.
The SHM Blue Ridge Chapter has collaborated with both the ACP Tennessee Chapter and the Healthcare MBA program at Haslam College of Business at the University of Tennessee.
The chapter leadership regularly attends local medical residency programs at noon conferences to attract and recruit young physicians into chapter activities. Overall, the chapter has seen a growth in membership in 2019. The Blue Ridge Chapter is an active, enthusiastic chapter that is rapidly growing and thriving.
Outstanding Membership Recruitment and Retention
The Outstanding Membership Recruitment and Retention Award is a new exemplary award for 2019 that goes to one chapter who has gone above and beyond to implement initiatives to recruit and retain SHM members in their chapter. The recipient of the Outstanding Membership Recruitment and Retention Award for 2019 is the Western Massachusetts Chapter of SHM, which has done outstanding work to recruit and retain the membership. In 2019, the SHM membership in the chapter grew by 24%. The chapter utilized Chapter Development Funds to launch new initiatives to conduct outreach to nonmember hospitalists in the community and invite them to meetings to obtain the SHM experience. Additionally, the chapter encouraged residents to join and get involved by hosting a poster competition.
The Western Massachusetts Chapter focused on being innovative, inclusive, and creative to retain their existing meetings. For example, the chapter hosted a new “Jeopardy Session” event that featured a nontraditional jeopardy game that attracted a large attendance including local residents. Additionally, the chapter insured that all clinical and nonclinical members of the hospital medicine team were included and encouraged to participate in all chapter meetings. Lastly, the chapter launched a local awards program to recognize senior hospitalist and early career hospitalist who contributed to chapter development.
Most Engaged Chapter Leader
The Most Engaged Chapter Leader Award is a new exemplary award for 2019 that goes to one chapter leader or district chair who is either nominated or self-nominated and has demonstrated how they or their nominee has gone above and beyond in the past year to grow and sustain their chapter and/or district and continues to carry out the SHM mission. The recipient of the Most Engaged Chapter Leader Award for 2019 goes to Thérèse Franco, MD, SFHM, president of the Pacific Northwest Chapter.
Dr. Franco has served as the chapter’s president for 2 years and has served on the SHM Chapter Support Committee for 3 years. She has previously participated as a mentor in the glycemic control mentored implementation program, and as chair and cochair of the RIV contest. She continues to review abstracts, volunteer as a judge and offer local education on glycemic control through the Washington State Hospital Association, promoting SHM’s work there. One of Dr. Franco’s core strengths has been effective collaboration with past leaders (such as Rachel Thompson, MD, and Kimberly Bell, MD), future leaders, and other organizations (such as the Washington State Medical Association and the King County Medical Association). Dr. Franco has recruited an outstanding leadership team and new advisory committee for the Pacific Northwest Chapter, resulting a fantastic year of growth, innovation, and development.
The Society of Hospital Medicine is proud to recognize its chapters for their hard work and dedication in 2019 through Chapter Excellence Awards. Each year, chapters strive to demonstrate growth, sustenance, and innovation within their chapter activities, which are then applauded for their successes throughout the subsequent year. In 2019, a new Bronze category was established, for a total of four Status Awards that chapters can earn.
Please join SHM in congratulating the following chapters on their year of success in 2019!
Outstanding Chapter of the Year
The Outstanding Chapter of the Year Award goes to one chapter who exemplifies high performance, going above and beyond the basic chapter requirements. The recipient of the Outstanding Chapter of the Year Award for 2019 is the Wiregrass Chapter of SHM. The chapter has a strong and engaged leadership which includes representation at all levels of the hospital medicine team, including physician hospitalists, advanced care provider hospitalists, practice administrators, nurses, residents, and medical students.
In the last year, the Wiregrass leadership team has organized programs and events to cater to and engage all the chapter’s members. This includes a variety of innovative ideas that catered toward medical education, health care provider well-being, engagement, mentorship, and community involvement.
The SHM Wiregrass Chapter’s biggest accomplishment in 2019 was the creation of an exchange program for physician and advanced practice provider hospitalists between the SHM New Mexico Chapter and the SHM Wiregrass Chapter. This idea first arose at HM19, where the chapter leaders had met during a networking event and debated the role of clinician wellbeing, quality of medical education, and faculty development to individual hospital medicine group (HMG) practice styles.
Clinician well-being is the prerequisite to the triple aim of improving the health of populations, enhancing the patient experience, and reducing the cost of care. Each HMG faces similar challenges but approaches to solving them vary. Professional challenges can affect the well-being of the individual clinicians. Having interinstitutional exchange programs provides a platform to exchange ideas and establish mentors. Also, the quality of medical education is directly linked to the quality of faculty development. Improving the quality of medical education requires a multifaceted approach by highly developed faculty. The complex factors affecting medical education and faculty development are further complicated by geographic location, patient characteristics, and professional growth opportunities. Overcoming these obstacles requires an innovative and collaborative approach. Although faculty exchanges are common in academic medicine, they are not commonly attempted with HMGs.
Hospitalists are responsible for a significant part of inpatient training for residents, medical students, and nurse practitioners/physician assistants (NPs/PAs), but their faculty training can vary based on location. Being a young specialty, only 2 decades old, hospital medicine is still evolving and incorporating NP/PA and physician hospitalists in varied practice models. Each HMG addresses common obstacles differently based on their culture and practice styles. The chapter leaders determined an exchange program would afford the opportunity for visiting faculty members to experience these differences. This emphasized the role and importance of exchanging ideas and contemplated a solution to benefit more practicing hospitalists.
The chapter leaders researched the characteristics of individual academic HMGs and structured a tailored faculty exchange involving physicians and NPs/PAs. During the exchange program planning, the visiting faculty itinerary was tailored to a well-planned agenda for 1 week, with separate tracks for physicians and NPs/PAs giving increased access to their individual peer practice styles. Additionally, the visiting faculty had meetings and discussions with HMG and hospital leadership, to specifically address each visiting faculty institution’s challenges. The overall goal of this exchange program was to promote cross-institutional collaboration, increase engagement, improve medical education through faculty development, and improve the quality of care. The focus of the exchange program was to share ideas and innovation and learn the approaches to unique challenges at each institution. Out of this also came collaboration and mentoring opportunities.
The evaluation process of the exchange involved interviews, a survey, and the establishment of shared QI projects in mutual areas of challenge. The survey provided feedback, lessons learned from the exchange, and areas to be improved. Collaborative QI projects currently underway as a result of the exchange include paging etiquette, quality of sleep for hospitalized patients, and onboarding of NPs/PAs in HMGs.
This innovation addressed faculty development and medical education via clinician well-being. The physician and NP/PA Faculty Exchange was an essential and meaningful innovation that resulted in increased SHM member engagement, cross-institutional collaboration, networking, and mentorship.
Additional projects that the SHM Wiregrass Chapter successfully implemented in 2019 include a “Women in Medicine” event that recognized women physician and advanced practice provider hospitalist leaders, a poster competition that expanded its research, clinical vignettes, and quality categories to include a fourth category of innovation, featuring 75 posters. Additionally, the chapter held a policy meeting with six Alabama state legislators, creating new channels of collaboration between the legislators and the chapter. Lastly, the chapter held a successful community event and launched a mentor program targeting medical students and residents.
Rising Star Chapter
The Rising Star Chapter Award goes to one chapter who has been active for 2 years or less, who in the past 12 months have made improvements to their leadership, stability and growth, and membership. The recipient of the Rising Star Chapter Award for 2019 is the Blue Ridge Chapter of SHM, which has made significant strides to develop since its launch in the fall of 2018. The chapter represents counties in northwest Tennessee, southwest Virginia, and western North Carolina.
The chapter held three meetings in 2019 which were well attended by hospitalists, residents/fellows, administrators, advanced practice providers, and nurses. On average, attendees from five to six different hospitalist groups are represented. The chapter hosted both Dr. Chris Frost, immediate past president of the SHM board of directors, and Dr. Ron Greeno, a past president of the SHM board of directors.
The SHM Blue Ridge Chapter has collaborated with both the ACP Tennessee Chapter and the Healthcare MBA program at Haslam College of Business at the University of Tennessee.
The chapter leadership regularly attends local medical residency programs at noon conferences to attract and recruit young physicians into chapter activities. Overall, the chapter has seen a growth in membership in 2019. The Blue Ridge Chapter is an active, enthusiastic chapter that is rapidly growing and thriving.
Outstanding Membership Recruitment and Retention
The Outstanding Membership Recruitment and Retention Award is a new exemplary award for 2019 that goes to one chapter who has gone above and beyond to implement initiatives to recruit and retain SHM members in their chapter. The recipient of the Outstanding Membership Recruitment and Retention Award for 2019 is the Western Massachusetts Chapter of SHM, which has done outstanding work to recruit and retain the membership. In 2019, the SHM membership in the chapter grew by 24%. The chapter utilized Chapter Development Funds to launch new initiatives to conduct outreach to nonmember hospitalists in the community and invite them to meetings to obtain the SHM experience. Additionally, the chapter encouraged residents to join and get involved by hosting a poster competition.
The Western Massachusetts Chapter focused on being innovative, inclusive, and creative to retain their existing meetings. For example, the chapter hosted a new “Jeopardy Session” event that featured a nontraditional jeopardy game that attracted a large attendance including local residents. Additionally, the chapter insured that all clinical and nonclinical members of the hospital medicine team were included and encouraged to participate in all chapter meetings. Lastly, the chapter launched a local awards program to recognize senior hospitalist and early career hospitalist who contributed to chapter development.
Most Engaged Chapter Leader
The Most Engaged Chapter Leader Award is a new exemplary award for 2019 that goes to one chapter leader or district chair who is either nominated or self-nominated and has demonstrated how they or their nominee has gone above and beyond in the past year to grow and sustain their chapter and/or district and continues to carry out the SHM mission. The recipient of the Most Engaged Chapter Leader Award for 2019 goes to Thérèse Franco, MD, SFHM, president of the Pacific Northwest Chapter.
Dr. Franco has served as the chapter’s president for 2 years and has served on the SHM Chapter Support Committee for 3 years. She has previously participated as a mentor in the glycemic control mentored implementation program, and as chair and cochair of the RIV contest. She continues to review abstracts, volunteer as a judge and offer local education on glycemic control through the Washington State Hospital Association, promoting SHM’s work there. One of Dr. Franco’s core strengths has been effective collaboration with past leaders (such as Rachel Thompson, MD, and Kimberly Bell, MD), future leaders, and other organizations (such as the Washington State Medical Association and the King County Medical Association). Dr. Franco has recruited an outstanding leadership team and new advisory committee for the Pacific Northwest Chapter, resulting a fantastic year of growth, innovation, and development.
COVID-19 exacerbating challenges for Latino patients
Disproportionate burden of pandemic complicates mental health care
Pamela Montano, MD, recalls the recent case of a patient with bipolar II disorder who was improving after treatment with medication and therapy when her life was upended by the COVID-19 pandemic.
The patient, who is Puerto Rican, lost two cousins to the virus, two of her brothers fell ill, and her sister became sick with coronavirus, said Dr. Montano, director of the Latino Bicultural Clinic at Gouverneur Health in New York. The patient was then left to care for her sister’s toddlers along with the patient’s own children, one of whom has special needs.
“After this happened, it increased her anxiety,” Dr. Montano said in an interview. “She’s not sleeping, and she started having panic attacks. My main concern was how to help her cope.”
Across the country, clinicians who treat mental illness and behavioral disorders in Latino patients are facing similar experiences and challenges associated with COVID-19 and the ensuing pandemic response. Current data suggest a disproportionate burden of illness and death from the novel coronavirus among racial and ethnic groups, particularly black and Hispanic patients. The disparities are likely attributable to economic and social conditions more common among such populations, compared with non-Hispanic whites, in addition to isolation from resources, according to the Centers for Disease Control and Prevention.
A recent New York City Department of Health study based on data that were available in late April found that deaths from COVID-19 were substantially higher for black and Hispanic/Latino patients than for white and Asian patients. The death rate per 100,000 population was 209.4 for blacks, 195.3 for Hispanics/Latinos, 107.7 for whites, and 90.8 for Asians.
“The COVID pandemic has highlighted the structural inequities that affect the Latino population [both] immigrant and nonimmigrant,” said Dr. Montano, a board member of the American Society of Hispanic Psychiatry and the officer of infrastructure and advocacy for the Hispanic Caucus of the American Psychiatric Association. “This includes income inequality, poor nutrition, history of trauma and discrimination, employment issues, quality education, access to technology, and overall access to appropriate cultural linguistic health care.”
Navigating challenges
For mental health professionals treating Latino patients, COVID-19 and the pandemic response have generated a range of treatment obstacles.
The transition to telehealth for example, has not been easy for some patients, said Jacqueline Posada, MD, consultation-liaison psychiatry fellow at the Inova Fairfax Hospital–George Washington University program in Falls Church, Va., and an APA Substance Abuse and Mental Health Services Administration minority fellow. Some patients lack Internet services, others forget virtual visits, and some do not have working phones, she said.
“I’ve had to be very flexible,” she said in an interview. “Ideally, I’d love to see everybody via video chat, but a lot of people either don’t have a stable Internet connection or Internet, so I meet the patient where they are. Whatever they have available, that’s what I’m going to use. If they don’t answer on the first call, I will call again at least three to five times in the first 15 minutes to make sure I’m giving them an opportunity to pick up the phone.”
In addition, Dr. Posada has encountered disconnected phones when calling patients for appointments. In such cases, Dr. Posada contacts the patient’s primary care physician to relay medication recommendations in case the patient resurfaces at the clinic.
In other instances, patients are not familiar with video technology, or they must travel to a friend or neighbor’s house to access the technology, said Hector Colón-Rivera, MD, an addiction psychiatrist and medical director of the Asociación Puertorriqueños en Marcha Behavioral Health Program, a nonprofit organization based in the Philadelphia area. Telehealth visits frequently include appearances by children, family members, barking dogs, and other distractions, said Dr. Colón-Rivera, president of the APA Hispanic Caucus.
“We’re seeing things that we didn’t used to see when they came to our office – for good or for bad,” said Dr. Colón-Rivera, an attending telemedicine physician at the University of Pittsburgh Medical Center. “It could be a good chance to meet our patient in a different way. Of course, it creates different stressors. If you have five kids on top of you and you’re the only one at home, it’s hard to do therapy.”
Psychiatrists are also seeing prior health conditions in patients exacerbated by COVID-19 fears and new health problems arising from the current pandemic environment. Dr. Posada recalls a patient whom she successfully treated for premenstrual dysphoric disorder who recently descended into severe clinical depression. The patient, from Colombia, was attending school in the United States on a student visa and supporting herself through child care jobs.
“So much of her depression was based on her social circumstance,” Dr. Posada said. “She had lost her job, her sister had lost her job so they were scraping by on her sister’s husband’s income, and the thing that brought her joy, which was going to school and studying so she could make a different life for herself than what her parents had in Colombia, also seemed like it was out of reach.”
Dr. Colón-Rivera recently received a call from a hospital where one of his patients was admitted after becoming delusional and psychotic. The patient was correctly taking medication prescribed by Dr. Colón-Rivera, but her diabetes had become uncontrolled because she was unable to reach her primary care doctor and couldn’t access the pharmacy. Her blood sugar level became elevated, leading to the delusions.
“A patient that was perfectly stable now is unstable,” he said. “Her diet has not been good enough through the pandemic, exacerbating her diabetes. She was admitted to the hospital for delirium.
Compounding of traumas
For many Latino patients, the adverse impacts of the pandemic comes on top of multiple prior traumas, such as violence exposures, discrimination, and economic issues, said Lisa Fortuna, MD, MPH, MDiv, chief of psychiatry and vice chair at Zuckerberg San Francisco General Hospital. A 2017 analysis found that nearly four in five Latino youth face at least one traumatic childhood experience, like poverty or abuse, and that about 29% of Latino youth experience four or more of these traumas.
Immigrants in particular, may have faced trauma in their home country and/or immigration trauma, Dr. Fortuna added. A 2013 study on immigrant Latino adolescents for example, found that 29% of foreign-born adolescents and 34% of foreign-born parents experienced trauma during the migration process (Int Migr Rev. 2013 Dec;47(4):10).
“All of these things are cumulative,” Dr. Fortuna said. “Then when you’re hit with a pandemic, all of the disparities that you already have and all the stress that you already have are compounded. This is for the kids, too, who have been exposed to a lot of stressors and now maybe have family members that have been ill or have died. All of these things definitely put people at risk for increased depression [and] the worsening of any preexisting posttraumatic stress disorder. We’ve seen this in previous disasters, and I expect that’s what we’re going to see more of with the COVID-19 pandemic.”
At the same time, a central cultural value of many Latinos is family unity, Dr. Montano said, a foundation that is now being strained by social distancing and severed connections.
“This has separated many families,” she said. “There has been a lot of loneliness and grief.”
Mistrust and fear toward the government, public agencies, and even the health system itself act as further hurdles for some Latinos in the face of COVID-19. In areas with large immigrant populations such as San Francisco, Dr. Fortuna noted, it’s not uncommon for undocumented patients to avoid accessing medical care and social services, or visiting emergency departments for needed care for fear of drawing attention to themselves or possible detainment.
“The fact that so many people showed up at our hospital so ill and ended up in the ICU – that could be a combination of factors. Because the population has high rates of diabetes and hypertension, that might have put people at increased risk for severe illness,” she said. “But some people may have been holding out for care because they wanted to avoid being in places out of fear of immigration scrutiny.”
Overcoming language barriers
Compounding the challenging pandemic landscape for Latino patients is the fact that many state resources about COVID-19 have not been translated to Spanish, Dr. Colón-Rivera said. He was troubled recently when he went to several state websites and found limited to no information in Spanish about the coronavirus. Some data about COVID-19 from the federal government were not translated to Spanish until officials received pushback, he added. Even now, press releases and other information disseminated by the federal government about the virus appear to be translated by an automated service – and lack sense and context.
The state agencies in Pennsylvania have been alerted to the absence of Spanish information, but change has been slow, he noted.
“In Philadelphia, 23% speaks a language other than English,” he said. “So we missed a lot of critical information that could have helped to avoid spreading the illness and access support.”
Dr. Fortuna said that California has done better with providing COVID-19–related information in Spanish, compared with some other states, but misinformation about the virus and lingering myths have still been a problem among the Latino community. The University of California, San Francisco, recently launched a Latino Task Force resource website for the Latino community that includes information in English, Spanish, and Yucatec Maya about COVID-19, health and wellness tips, and resources for various assistance needs.
The concerning lack of COVID-19 information translated to Spanish led Dr. Montano to start a Facebook page in Spanish about mental health tips and guidance for managing COVID-19–related issues. She and her team of clinicians share information, videos, relaxation exercises, and community resources on the page, among other posts. “There is also general info and recommendations about COVID-19 that I think can be useful for the community,” she said. “The idea is that patients, the general community, and providers can have share information, hope messages, and ask questions in Spanish.”
Feeling ‘helpless’
A central part of caring for Latino patients during the COVID-19 crisis has been referring them to outside agencies and social services, psychiatrists say. But finding the right resources amid a pandemic and ensuring that patients connect with the correct aid has been an uphill battle.
“We sometimes feel like our hands are tied,” Dr. Colón-Rivera said. “Sometimes, we need to call a place to bring food. Some of the state agencies and nonprofits don’t have delivery systems, so the patient has to go pick up for food or medication. Some of our patients don’t want to go outside. Some do not have cars.”
As a clinician, it can be easy to feel helpless when trying to navigate new challenges posed by the pandemic in addition to other longstanding barriers, Dr. Posada said.
“Already, mental health disorders are so influenced by social situations like poverty, job insecurity, or family issues, and now it just seems those obstacles are even more insurmountable,” she said. “At the end of the day, I can feel like: ‘Did I make a difference?’ That’s a big struggle.”
Dr. Montano’s team, which includes psychiatrists, psychologists, and social workers, have come to rely on virtual debriefings to vent, express frustrations, and support one another, she said. She also recently joined a virtual mind-body skills group as a participant.
“I recognize the importance of getting additional support and ways to alleviate burnout,” she said. “We need to take care of ourselves or we won’t be able to help others.”
Focusing on resilience during the current crisis can be beneficial for both patients and providers in coping and drawing strength, Dr. Posada said.
“When it comes to fostering resilience during times of hardship, I think it’s most helpful to reflect on what skills or attributes have helped during past crises and apply those now – whether it’s turning to comfort from close relationships, looking to religion and spirituality, practicing self-care like rest or exercise, or really tapping into one’s purpose and reason for practicing psychiatry and being a physician,” she said. “The same advice goes for clinicians: We’ve all been through hard times in the past, it’s part of the human condition and we’ve also witnessed a lot of suffering in our patients, so now is the time to practice those skills that have gotten us through hard times in the past.”
Learning lessons from COVID-19
Despite the challenges with moving to telehealth, Dr. Fortuna said the tool has proved beneficial overall for mental health care. For Dr. Fortuna’s team for example, telehealth by phone has decreased the no-show rate, compared with clinic visits, and improved care access.
“We need to figure out how to maintain that,” she said. “If we can build ways for equity and access to Internet, especially equipment, I think that’s going to help.”
In addition, more data are needed about the ways in which COVID-19 is affecting Latino patients, Dr. Colón-Rivera said. Mortality statistics have been published, but information is needed about the rates of infection and manifestation of illness.
Most importantly, the COVID-19 crisis has emphasized the critical need to address and improve the underlying inequity issues among Latino patients, psychiatrists say.
“We really need to think about how there can be partnerships, in terms of community-based Latino business and leaders, multisector resources, trying to think about how we can improve conditions both work and safety for Latinos,” Dr. Fortuna said. “How can schools get support in integrating mental health and support for families, especially now after COVID-19? And really looking at some of these underlying inequities that are the underpinnings of why people were at risk for the disproportionate effects of the COVID-19 pandemic.”
Disproportionate burden of pandemic complicates mental health care
Disproportionate burden of pandemic complicates mental health care
Pamela Montano, MD, recalls the recent case of a patient with bipolar II disorder who was improving after treatment with medication and therapy when her life was upended by the COVID-19 pandemic.
The patient, who is Puerto Rican, lost two cousins to the virus, two of her brothers fell ill, and her sister became sick with coronavirus, said Dr. Montano, director of the Latino Bicultural Clinic at Gouverneur Health in New York. The patient was then left to care for her sister’s toddlers along with the patient’s own children, one of whom has special needs.
“After this happened, it increased her anxiety,” Dr. Montano said in an interview. “She’s not sleeping, and she started having panic attacks. My main concern was how to help her cope.”
Across the country, clinicians who treat mental illness and behavioral disorders in Latino patients are facing similar experiences and challenges associated with COVID-19 and the ensuing pandemic response. Current data suggest a disproportionate burden of illness and death from the novel coronavirus among racial and ethnic groups, particularly black and Hispanic patients. The disparities are likely attributable to economic and social conditions more common among such populations, compared with non-Hispanic whites, in addition to isolation from resources, according to the Centers for Disease Control and Prevention.
A recent New York City Department of Health study based on data that were available in late April found that deaths from COVID-19 were substantially higher for black and Hispanic/Latino patients than for white and Asian patients. The death rate per 100,000 population was 209.4 for blacks, 195.3 for Hispanics/Latinos, 107.7 for whites, and 90.8 for Asians.
“The COVID pandemic has highlighted the structural inequities that affect the Latino population [both] immigrant and nonimmigrant,” said Dr. Montano, a board member of the American Society of Hispanic Psychiatry and the officer of infrastructure and advocacy for the Hispanic Caucus of the American Psychiatric Association. “This includes income inequality, poor nutrition, history of trauma and discrimination, employment issues, quality education, access to technology, and overall access to appropriate cultural linguistic health care.”
Navigating challenges
For mental health professionals treating Latino patients, COVID-19 and the pandemic response have generated a range of treatment obstacles.
The transition to telehealth for example, has not been easy for some patients, said Jacqueline Posada, MD, consultation-liaison psychiatry fellow at the Inova Fairfax Hospital–George Washington University program in Falls Church, Va., and an APA Substance Abuse and Mental Health Services Administration minority fellow. Some patients lack Internet services, others forget virtual visits, and some do not have working phones, she said.
“I’ve had to be very flexible,” she said in an interview. “Ideally, I’d love to see everybody via video chat, but a lot of people either don’t have a stable Internet connection or Internet, so I meet the patient where they are. Whatever they have available, that’s what I’m going to use. If they don’t answer on the first call, I will call again at least three to five times in the first 15 minutes to make sure I’m giving them an opportunity to pick up the phone.”
In addition, Dr. Posada has encountered disconnected phones when calling patients for appointments. In such cases, Dr. Posada contacts the patient’s primary care physician to relay medication recommendations in case the patient resurfaces at the clinic.
In other instances, patients are not familiar with video technology, or they must travel to a friend or neighbor’s house to access the technology, said Hector Colón-Rivera, MD, an addiction psychiatrist and medical director of the Asociación Puertorriqueños en Marcha Behavioral Health Program, a nonprofit organization based in the Philadelphia area. Telehealth visits frequently include appearances by children, family members, barking dogs, and other distractions, said Dr. Colón-Rivera, president of the APA Hispanic Caucus.
“We’re seeing things that we didn’t used to see when they came to our office – for good or for bad,” said Dr. Colón-Rivera, an attending telemedicine physician at the University of Pittsburgh Medical Center. “It could be a good chance to meet our patient in a different way. Of course, it creates different stressors. If you have five kids on top of you and you’re the only one at home, it’s hard to do therapy.”
Psychiatrists are also seeing prior health conditions in patients exacerbated by COVID-19 fears and new health problems arising from the current pandemic environment. Dr. Posada recalls a patient whom she successfully treated for premenstrual dysphoric disorder who recently descended into severe clinical depression. The patient, from Colombia, was attending school in the United States on a student visa and supporting herself through child care jobs.
“So much of her depression was based on her social circumstance,” Dr. Posada said. “She had lost her job, her sister had lost her job so they were scraping by on her sister’s husband’s income, and the thing that brought her joy, which was going to school and studying so she could make a different life for herself than what her parents had in Colombia, also seemed like it was out of reach.”
Dr. Colón-Rivera recently received a call from a hospital where one of his patients was admitted after becoming delusional and psychotic. The patient was correctly taking medication prescribed by Dr. Colón-Rivera, but her diabetes had become uncontrolled because she was unable to reach her primary care doctor and couldn’t access the pharmacy. Her blood sugar level became elevated, leading to the delusions.
“A patient that was perfectly stable now is unstable,” he said. “Her diet has not been good enough through the pandemic, exacerbating her diabetes. She was admitted to the hospital for delirium.
Compounding of traumas
For many Latino patients, the adverse impacts of the pandemic comes on top of multiple prior traumas, such as violence exposures, discrimination, and economic issues, said Lisa Fortuna, MD, MPH, MDiv, chief of psychiatry and vice chair at Zuckerberg San Francisco General Hospital. A 2017 analysis found that nearly four in five Latino youth face at least one traumatic childhood experience, like poverty or abuse, and that about 29% of Latino youth experience four or more of these traumas.
Immigrants in particular, may have faced trauma in their home country and/or immigration trauma, Dr. Fortuna added. A 2013 study on immigrant Latino adolescents for example, found that 29% of foreign-born adolescents and 34% of foreign-born parents experienced trauma during the migration process (Int Migr Rev. 2013 Dec;47(4):10).
“All of these things are cumulative,” Dr. Fortuna said. “Then when you’re hit with a pandemic, all of the disparities that you already have and all the stress that you already have are compounded. This is for the kids, too, who have been exposed to a lot of stressors and now maybe have family members that have been ill or have died. All of these things definitely put people at risk for increased depression [and] the worsening of any preexisting posttraumatic stress disorder. We’ve seen this in previous disasters, and I expect that’s what we’re going to see more of with the COVID-19 pandemic.”
At the same time, a central cultural value of many Latinos is family unity, Dr. Montano said, a foundation that is now being strained by social distancing and severed connections.
“This has separated many families,” she said. “There has been a lot of loneliness and grief.”
Mistrust and fear toward the government, public agencies, and even the health system itself act as further hurdles for some Latinos in the face of COVID-19. In areas with large immigrant populations such as San Francisco, Dr. Fortuna noted, it’s not uncommon for undocumented patients to avoid accessing medical care and social services, or visiting emergency departments for needed care for fear of drawing attention to themselves or possible detainment.
“The fact that so many people showed up at our hospital so ill and ended up in the ICU – that could be a combination of factors. Because the population has high rates of diabetes and hypertension, that might have put people at increased risk for severe illness,” she said. “But some people may have been holding out for care because they wanted to avoid being in places out of fear of immigration scrutiny.”
Overcoming language barriers
Compounding the challenging pandemic landscape for Latino patients is the fact that many state resources about COVID-19 have not been translated to Spanish, Dr. Colón-Rivera said. He was troubled recently when he went to several state websites and found limited to no information in Spanish about the coronavirus. Some data about COVID-19 from the federal government were not translated to Spanish until officials received pushback, he added. Even now, press releases and other information disseminated by the federal government about the virus appear to be translated by an automated service – and lack sense and context.
The state agencies in Pennsylvania have been alerted to the absence of Spanish information, but change has been slow, he noted.
“In Philadelphia, 23% speaks a language other than English,” he said. “So we missed a lot of critical information that could have helped to avoid spreading the illness and access support.”
Dr. Fortuna said that California has done better with providing COVID-19–related information in Spanish, compared with some other states, but misinformation about the virus and lingering myths have still been a problem among the Latino community. The University of California, San Francisco, recently launched a Latino Task Force resource website for the Latino community that includes information in English, Spanish, and Yucatec Maya about COVID-19, health and wellness tips, and resources for various assistance needs.
The concerning lack of COVID-19 information translated to Spanish led Dr. Montano to start a Facebook page in Spanish about mental health tips and guidance for managing COVID-19–related issues. She and her team of clinicians share information, videos, relaxation exercises, and community resources on the page, among other posts. “There is also general info and recommendations about COVID-19 that I think can be useful for the community,” she said. “The idea is that patients, the general community, and providers can have share information, hope messages, and ask questions in Spanish.”
Feeling ‘helpless’
A central part of caring for Latino patients during the COVID-19 crisis has been referring them to outside agencies and social services, psychiatrists say. But finding the right resources amid a pandemic and ensuring that patients connect with the correct aid has been an uphill battle.
“We sometimes feel like our hands are tied,” Dr. Colón-Rivera said. “Sometimes, we need to call a place to bring food. Some of the state agencies and nonprofits don’t have delivery systems, so the patient has to go pick up for food or medication. Some of our patients don’t want to go outside. Some do not have cars.”
As a clinician, it can be easy to feel helpless when trying to navigate new challenges posed by the pandemic in addition to other longstanding barriers, Dr. Posada said.
“Already, mental health disorders are so influenced by social situations like poverty, job insecurity, or family issues, and now it just seems those obstacles are even more insurmountable,” she said. “At the end of the day, I can feel like: ‘Did I make a difference?’ That’s a big struggle.”
Dr. Montano’s team, which includes psychiatrists, psychologists, and social workers, have come to rely on virtual debriefings to vent, express frustrations, and support one another, she said. She also recently joined a virtual mind-body skills group as a participant.
“I recognize the importance of getting additional support and ways to alleviate burnout,” she said. “We need to take care of ourselves or we won’t be able to help others.”
Focusing on resilience during the current crisis can be beneficial for both patients and providers in coping and drawing strength, Dr. Posada said.
“When it comes to fostering resilience during times of hardship, I think it’s most helpful to reflect on what skills or attributes have helped during past crises and apply those now – whether it’s turning to comfort from close relationships, looking to religion and spirituality, practicing self-care like rest or exercise, or really tapping into one’s purpose and reason for practicing psychiatry and being a physician,” she said. “The same advice goes for clinicians: We’ve all been through hard times in the past, it’s part of the human condition and we’ve also witnessed a lot of suffering in our patients, so now is the time to practice those skills that have gotten us through hard times in the past.”
Learning lessons from COVID-19
Despite the challenges with moving to telehealth, Dr. Fortuna said the tool has proved beneficial overall for mental health care. For Dr. Fortuna’s team for example, telehealth by phone has decreased the no-show rate, compared with clinic visits, and improved care access.
“We need to figure out how to maintain that,” she said. “If we can build ways for equity and access to Internet, especially equipment, I think that’s going to help.”
In addition, more data are needed about the ways in which COVID-19 is affecting Latino patients, Dr. Colón-Rivera said. Mortality statistics have been published, but information is needed about the rates of infection and manifestation of illness.
Most importantly, the COVID-19 crisis has emphasized the critical need to address and improve the underlying inequity issues among Latino patients, psychiatrists say.
“We really need to think about how there can be partnerships, in terms of community-based Latino business and leaders, multisector resources, trying to think about how we can improve conditions both work and safety for Latinos,” Dr. Fortuna said. “How can schools get support in integrating mental health and support for families, especially now after COVID-19? And really looking at some of these underlying inequities that are the underpinnings of why people were at risk for the disproportionate effects of the COVID-19 pandemic.”
Pamela Montano, MD, recalls the recent case of a patient with bipolar II disorder who was improving after treatment with medication and therapy when her life was upended by the COVID-19 pandemic.
The patient, who is Puerto Rican, lost two cousins to the virus, two of her brothers fell ill, and her sister became sick with coronavirus, said Dr. Montano, director of the Latino Bicultural Clinic at Gouverneur Health in New York. The patient was then left to care for her sister’s toddlers along with the patient’s own children, one of whom has special needs.
“After this happened, it increased her anxiety,” Dr. Montano said in an interview. “She’s not sleeping, and she started having panic attacks. My main concern was how to help her cope.”
Across the country, clinicians who treat mental illness and behavioral disorders in Latino patients are facing similar experiences and challenges associated with COVID-19 and the ensuing pandemic response. Current data suggest a disproportionate burden of illness and death from the novel coronavirus among racial and ethnic groups, particularly black and Hispanic patients. The disparities are likely attributable to economic and social conditions more common among such populations, compared with non-Hispanic whites, in addition to isolation from resources, according to the Centers for Disease Control and Prevention.
A recent New York City Department of Health study based on data that were available in late April found that deaths from COVID-19 were substantially higher for black and Hispanic/Latino patients than for white and Asian patients. The death rate per 100,000 population was 209.4 for blacks, 195.3 for Hispanics/Latinos, 107.7 for whites, and 90.8 for Asians.
“The COVID pandemic has highlighted the structural inequities that affect the Latino population [both] immigrant and nonimmigrant,” said Dr. Montano, a board member of the American Society of Hispanic Psychiatry and the officer of infrastructure and advocacy for the Hispanic Caucus of the American Psychiatric Association. “This includes income inequality, poor nutrition, history of trauma and discrimination, employment issues, quality education, access to technology, and overall access to appropriate cultural linguistic health care.”
Navigating challenges
For mental health professionals treating Latino patients, COVID-19 and the pandemic response have generated a range of treatment obstacles.
The transition to telehealth for example, has not been easy for some patients, said Jacqueline Posada, MD, consultation-liaison psychiatry fellow at the Inova Fairfax Hospital–George Washington University program in Falls Church, Va., and an APA Substance Abuse and Mental Health Services Administration minority fellow. Some patients lack Internet services, others forget virtual visits, and some do not have working phones, she said.
“I’ve had to be very flexible,” she said in an interview. “Ideally, I’d love to see everybody via video chat, but a lot of people either don’t have a stable Internet connection or Internet, so I meet the patient where they are. Whatever they have available, that’s what I’m going to use. If they don’t answer on the first call, I will call again at least three to five times in the first 15 minutes to make sure I’m giving them an opportunity to pick up the phone.”
In addition, Dr. Posada has encountered disconnected phones when calling patients for appointments. In such cases, Dr. Posada contacts the patient’s primary care physician to relay medication recommendations in case the patient resurfaces at the clinic.
In other instances, patients are not familiar with video technology, or they must travel to a friend or neighbor’s house to access the technology, said Hector Colón-Rivera, MD, an addiction psychiatrist and medical director of the Asociación Puertorriqueños en Marcha Behavioral Health Program, a nonprofit organization based in the Philadelphia area. Telehealth visits frequently include appearances by children, family members, barking dogs, and other distractions, said Dr. Colón-Rivera, president of the APA Hispanic Caucus.
“We’re seeing things that we didn’t used to see when they came to our office – for good or for bad,” said Dr. Colón-Rivera, an attending telemedicine physician at the University of Pittsburgh Medical Center. “It could be a good chance to meet our patient in a different way. Of course, it creates different stressors. If you have five kids on top of you and you’re the only one at home, it’s hard to do therapy.”
Psychiatrists are also seeing prior health conditions in patients exacerbated by COVID-19 fears and new health problems arising from the current pandemic environment. Dr. Posada recalls a patient whom she successfully treated for premenstrual dysphoric disorder who recently descended into severe clinical depression. The patient, from Colombia, was attending school in the United States on a student visa and supporting herself through child care jobs.
“So much of her depression was based on her social circumstance,” Dr. Posada said. “She had lost her job, her sister had lost her job so they were scraping by on her sister’s husband’s income, and the thing that brought her joy, which was going to school and studying so she could make a different life for herself than what her parents had in Colombia, also seemed like it was out of reach.”
Dr. Colón-Rivera recently received a call from a hospital where one of his patients was admitted after becoming delusional and psychotic. The patient was correctly taking medication prescribed by Dr. Colón-Rivera, but her diabetes had become uncontrolled because she was unable to reach her primary care doctor and couldn’t access the pharmacy. Her blood sugar level became elevated, leading to the delusions.
“A patient that was perfectly stable now is unstable,” he said. “Her diet has not been good enough through the pandemic, exacerbating her diabetes. She was admitted to the hospital for delirium.
Compounding of traumas
For many Latino patients, the adverse impacts of the pandemic comes on top of multiple prior traumas, such as violence exposures, discrimination, and economic issues, said Lisa Fortuna, MD, MPH, MDiv, chief of psychiatry and vice chair at Zuckerberg San Francisco General Hospital. A 2017 analysis found that nearly four in five Latino youth face at least one traumatic childhood experience, like poverty or abuse, and that about 29% of Latino youth experience four or more of these traumas.
Immigrants in particular, may have faced trauma in their home country and/or immigration trauma, Dr. Fortuna added. A 2013 study on immigrant Latino adolescents for example, found that 29% of foreign-born adolescents and 34% of foreign-born parents experienced trauma during the migration process (Int Migr Rev. 2013 Dec;47(4):10).
“All of these things are cumulative,” Dr. Fortuna said. “Then when you’re hit with a pandemic, all of the disparities that you already have and all the stress that you already have are compounded. This is for the kids, too, who have been exposed to a lot of stressors and now maybe have family members that have been ill or have died. All of these things definitely put people at risk for increased depression [and] the worsening of any preexisting posttraumatic stress disorder. We’ve seen this in previous disasters, and I expect that’s what we’re going to see more of with the COVID-19 pandemic.”
At the same time, a central cultural value of many Latinos is family unity, Dr. Montano said, a foundation that is now being strained by social distancing and severed connections.
“This has separated many families,” she said. “There has been a lot of loneliness and grief.”
Mistrust and fear toward the government, public agencies, and even the health system itself act as further hurdles for some Latinos in the face of COVID-19. In areas with large immigrant populations such as San Francisco, Dr. Fortuna noted, it’s not uncommon for undocumented patients to avoid accessing medical care and social services, or visiting emergency departments for needed care for fear of drawing attention to themselves or possible detainment.
“The fact that so many people showed up at our hospital so ill and ended up in the ICU – that could be a combination of factors. Because the population has high rates of diabetes and hypertension, that might have put people at increased risk for severe illness,” she said. “But some people may have been holding out for care because they wanted to avoid being in places out of fear of immigration scrutiny.”
Overcoming language barriers
Compounding the challenging pandemic landscape for Latino patients is the fact that many state resources about COVID-19 have not been translated to Spanish, Dr. Colón-Rivera said. He was troubled recently when he went to several state websites and found limited to no information in Spanish about the coronavirus. Some data about COVID-19 from the federal government were not translated to Spanish until officials received pushback, he added. Even now, press releases and other information disseminated by the federal government about the virus appear to be translated by an automated service – and lack sense and context.
The state agencies in Pennsylvania have been alerted to the absence of Spanish information, but change has been slow, he noted.
“In Philadelphia, 23% speaks a language other than English,” he said. “So we missed a lot of critical information that could have helped to avoid spreading the illness and access support.”
Dr. Fortuna said that California has done better with providing COVID-19–related information in Spanish, compared with some other states, but misinformation about the virus and lingering myths have still been a problem among the Latino community. The University of California, San Francisco, recently launched a Latino Task Force resource website for the Latino community that includes information in English, Spanish, and Yucatec Maya about COVID-19, health and wellness tips, and resources for various assistance needs.
The concerning lack of COVID-19 information translated to Spanish led Dr. Montano to start a Facebook page in Spanish about mental health tips and guidance for managing COVID-19–related issues. She and her team of clinicians share information, videos, relaxation exercises, and community resources on the page, among other posts. “There is also general info and recommendations about COVID-19 that I think can be useful for the community,” she said. “The idea is that patients, the general community, and providers can have share information, hope messages, and ask questions in Spanish.”
Feeling ‘helpless’
A central part of caring for Latino patients during the COVID-19 crisis has been referring them to outside agencies and social services, psychiatrists say. But finding the right resources amid a pandemic and ensuring that patients connect with the correct aid has been an uphill battle.
“We sometimes feel like our hands are tied,” Dr. Colón-Rivera said. “Sometimes, we need to call a place to bring food. Some of the state agencies and nonprofits don’t have delivery systems, so the patient has to go pick up for food or medication. Some of our patients don’t want to go outside. Some do not have cars.”
As a clinician, it can be easy to feel helpless when trying to navigate new challenges posed by the pandemic in addition to other longstanding barriers, Dr. Posada said.
“Already, mental health disorders are so influenced by social situations like poverty, job insecurity, or family issues, and now it just seems those obstacles are even more insurmountable,” she said. “At the end of the day, I can feel like: ‘Did I make a difference?’ That’s a big struggle.”
Dr. Montano’s team, which includes psychiatrists, psychologists, and social workers, have come to rely on virtual debriefings to vent, express frustrations, and support one another, she said. She also recently joined a virtual mind-body skills group as a participant.
“I recognize the importance of getting additional support and ways to alleviate burnout,” she said. “We need to take care of ourselves or we won’t be able to help others.”
Focusing on resilience during the current crisis can be beneficial for both patients and providers in coping and drawing strength, Dr. Posada said.
“When it comes to fostering resilience during times of hardship, I think it’s most helpful to reflect on what skills or attributes have helped during past crises and apply those now – whether it’s turning to comfort from close relationships, looking to religion and spirituality, practicing self-care like rest or exercise, or really tapping into one’s purpose and reason for practicing psychiatry and being a physician,” she said. “The same advice goes for clinicians: We’ve all been through hard times in the past, it’s part of the human condition and we’ve also witnessed a lot of suffering in our patients, so now is the time to practice those skills that have gotten us through hard times in the past.”
Learning lessons from COVID-19
Despite the challenges with moving to telehealth, Dr. Fortuna said the tool has proved beneficial overall for mental health care. For Dr. Fortuna’s team for example, telehealth by phone has decreased the no-show rate, compared with clinic visits, and improved care access.
“We need to figure out how to maintain that,” she said. “If we can build ways for equity and access to Internet, especially equipment, I think that’s going to help.”
In addition, more data are needed about the ways in which COVID-19 is affecting Latino patients, Dr. Colón-Rivera said. Mortality statistics have been published, but information is needed about the rates of infection and manifestation of illness.
Most importantly, the COVID-19 crisis has emphasized the critical need to address and improve the underlying inequity issues among Latino patients, psychiatrists say.
“We really need to think about how there can be partnerships, in terms of community-based Latino business and leaders, multisector resources, trying to think about how we can improve conditions both work and safety for Latinos,” Dr. Fortuna said. “How can schools get support in integrating mental health and support for families, especially now after COVID-19? And really looking at some of these underlying inequities that are the underpinnings of why people were at risk for the disproportionate effects of the COVID-19 pandemic.”