User login
For MD-IQ use only
So you want to be an expert witness?
Acting as an expert witness in a legal matter can be a nice way to compliment your practice. However, it is important to understand the role of experts, as well as their duties and obligations. Expert witnesses are called to testify on the basis of their specialized knowledge, not necessarily their direct knowledge of events and issues in the case.
Medical experts often play an important role in the evaluation, development, and preparation of a case long before it ever goes to trial. In some states, to even file a medical malpractice complaint a plaintiff is required to have the case evaluated by an expert and obtain a written report outlining why the plaintiff has a reasonable and meritorious cause for filing such an action.
There are different types of expert witness testimony. Experts can give opinion testimony as a physician who provided treatment to the plaintiff and whose conduct is not at issue. The second type of expert witness is a retained or controlled expert witness. This is a person giving opinion testimony after being retained by a lawyer on behalf of one of the parties to the lawsuit.
Before you give deposition or trial testimony, your opinions must be disclosed in writing and provided to the other parties in the case. In federal court, this is governed by Federal Rule of Civil Procedure 26. If the case is pending in state court, your written opinions are governed by local court rules. In both cases, the written opinions should be thorough and complete because you will not be allowed to testify to new opinions at the time of trial but will generally be allowed to expand upon those disclosed in writing at your deposition trial.
In order for a jury to hear your opinions at trial, your opinions must be reliable. In federal court, expert testimony is governed by Federal Rule of Evidence 702, which states:
A witness who is qualified as an expert by knowledge, skill, experience, training, or education may testify in the form of an opinion or otherwise if:
a) the expert’s scientific, technical, or other specialized knowledge will help the trier of fact to understand the evidence or to determine a fact in issue;
b) the testimony is based on sufficient facts or data;
c) the testimony is the product of reliable principles and methods; and
d) the expert has reliably applied the principles and methods to the facts of the case.
This means, that if a fact or evidence at issue involves scientific, technical, or specialized knowledge that is outside the scope of an ordinary layman’s experience, or involves complex issues challenging a layman’s comprehension, expert testimony is required. The scientific evidence must not just be relevant but also reliable. Expert opinions will be scrutinized to see if they are based on scientific testing or review of scientific data rather than just assumptions or speculation. Additionally, the experts must be qualified by their knowledge, skill, experience, training, or education. Given these parameters, it should come as no surprise that expert trial testimony is required for all medical malpractice cases.
Some states follow the “new or novel rule” which dictates that expert testimony is only admissible if the methodology or scientific principal on which the opinion is based is sufficiently established to have gained general acceptance in the particular field in which it belongs. This means that the evidence must be generally accepted as reliable in the relevant scientific community. New or novel techniques will be placed under the scrutiny of this standard. Courts will look at papers, books, journals, and case law to make a determination as to the reliability and general acceptance. Failure to meet the requisite standards may render a physician ineligible to testify.
If you are considering acting as an expert witness there are a few basic dos and don’ts to keep in mind:
Do be mindful of your criticism. If testifying in a medical malpractice case, you will be giving sworn testimony as to whether another physician deviated from the standard of care. Be aware that your testimony can later be used against you if your conduct is ever at issue, or if you contradict yourself in another case. Attorneys often look for prior testimony to use when questioning you at deposition and trial.
Do be aware of any applicable professional society guidelines. Many professional societies publish ethical guidelines as it relates to expert medical testimony. Be aware of those and know that you may be asked about them, especially if you are a member of that society.
Do be prepared for basic areas of cross-examination. There are a few tried and true areas that will always be the subject of cross-examination. Any perceived bias you may have, your fees, and whether you do more work for plaintiffs versus defendants are a just few examples. You should also be prepared to be cross-examined on the differences between personal practice (what you do) and an actual deviation from the standard of care.
Do keep written communication to a minimum. All communication between the expert physician and the attorney is potentially discoverable by the other side. The rules differ for state and federal courts. Emails, draft reports, and written questions all cause the creation of unnecessary side issues and areas of cross-examination. The best practice is for all substantive communication to be done by phone.
Do be clear in what you are charging. It is not unusual for an expert to charge one hourly rate for record review, and a different rate for testimony. Your fee schedule should also note that any travel expenses you incur will also be invoiced. Your hourly rate should be appropriate for your area of practice. In our experience, gastroenterologists typically charge $400.00-$600.00 an hour for record review, and $550.00-$700.00 an hour for testimony.
Do not submit an invoice until after your deposition. Submitting invoices before your deposition creates unnecessary cross-examination issues. At the time of retention, speak to the attorney and ask if you will be able to submit invoices as you work. Most attorneys prefer invoices be submitted after your deposition. Because the wheels of justice often turn slowly, you could be waiting an equally long time to submit an invoice and get paid. One way to avoid this dilemma is to require a retainer at the time of retention.
Do not sign up with an expert finder service. Resist the urge to sign up with an expert finder service. The best medical experts come from referrals from other attorneys or physicians. Expert retention via an expert finder service creates the impression that you are a “hired gun” in the business of being a professional expert and can diminish your credibility. The finder services also charge a commission or fee.
As a gastroenterologist, you have the specialized knowledge to provide expert testimony regarding the cause of an injury and extent of damages in cases where you have treated a patient. You also have the type of education and training necessary to serve as an independent expert. Doing so is a serious task that can be time consuming and stressful. However, it can also be rewarding and allow you to make sure a fair and just outcome occurs.
This article is for general informational purposes only. Please consult your own attorney if you have questions. This information is not intended to create an attorney-client relationship.
Mr. Mills is an equity partner at Cunningham, Meyer & Vedrine PC in Chicago. Ms. Lindbert is a partner at Cunningham, Meyer & Vedrine PC. Both focus their practices on defending doctors and hospitals in medical malpractice actions.
Acting as an expert witness in a legal matter can be a nice way to compliment your practice. However, it is important to understand the role of experts, as well as their duties and obligations. Expert witnesses are called to testify on the basis of their specialized knowledge, not necessarily their direct knowledge of events and issues in the case.
Medical experts often play an important role in the evaluation, development, and preparation of a case long before it ever goes to trial. In some states, to even file a medical malpractice complaint a plaintiff is required to have the case evaluated by an expert and obtain a written report outlining why the plaintiff has a reasonable and meritorious cause for filing such an action.
There are different types of expert witness testimony. Experts can give opinion testimony as a physician who provided treatment to the plaintiff and whose conduct is not at issue. The second type of expert witness is a retained or controlled expert witness. This is a person giving opinion testimony after being retained by a lawyer on behalf of one of the parties to the lawsuit.
Before you give deposition or trial testimony, your opinions must be disclosed in writing and provided to the other parties in the case. In federal court, this is governed by Federal Rule of Civil Procedure 26. If the case is pending in state court, your written opinions are governed by local court rules. In both cases, the written opinions should be thorough and complete because you will not be allowed to testify to new opinions at the time of trial but will generally be allowed to expand upon those disclosed in writing at your deposition trial.
In order for a jury to hear your opinions at trial, your opinions must be reliable. In federal court, expert testimony is governed by Federal Rule of Evidence 702, which states:
A witness who is qualified as an expert by knowledge, skill, experience, training, or education may testify in the form of an opinion or otherwise if:
a) the expert’s scientific, technical, or other specialized knowledge will help the trier of fact to understand the evidence or to determine a fact in issue;
b) the testimony is based on sufficient facts or data;
c) the testimony is the product of reliable principles and methods; and
d) the expert has reliably applied the principles and methods to the facts of the case.
This means, that if a fact or evidence at issue involves scientific, technical, or specialized knowledge that is outside the scope of an ordinary layman’s experience, or involves complex issues challenging a layman’s comprehension, expert testimony is required. The scientific evidence must not just be relevant but also reliable. Expert opinions will be scrutinized to see if they are based on scientific testing or review of scientific data rather than just assumptions or speculation. Additionally, the experts must be qualified by their knowledge, skill, experience, training, or education. Given these parameters, it should come as no surprise that expert trial testimony is required for all medical malpractice cases.
Some states follow the “new or novel rule” which dictates that expert testimony is only admissible if the methodology or scientific principal on which the opinion is based is sufficiently established to have gained general acceptance in the particular field in which it belongs. This means that the evidence must be generally accepted as reliable in the relevant scientific community. New or novel techniques will be placed under the scrutiny of this standard. Courts will look at papers, books, journals, and case law to make a determination as to the reliability and general acceptance. Failure to meet the requisite standards may render a physician ineligible to testify.
If you are considering acting as an expert witness there are a few basic dos and don’ts to keep in mind:
Do be mindful of your criticism. If testifying in a medical malpractice case, you will be giving sworn testimony as to whether another physician deviated from the standard of care. Be aware that your testimony can later be used against you if your conduct is ever at issue, or if you contradict yourself in another case. Attorneys often look for prior testimony to use when questioning you at deposition and trial.
Do be aware of any applicable professional society guidelines. Many professional societies publish ethical guidelines as it relates to expert medical testimony. Be aware of those and know that you may be asked about them, especially if you are a member of that society.
Do be prepared for basic areas of cross-examination. There are a few tried and true areas that will always be the subject of cross-examination. Any perceived bias you may have, your fees, and whether you do more work for plaintiffs versus defendants are a just few examples. You should also be prepared to be cross-examined on the differences between personal practice (what you do) and an actual deviation from the standard of care.
Do keep written communication to a minimum. All communication between the expert physician and the attorney is potentially discoverable by the other side. The rules differ for state and federal courts. Emails, draft reports, and written questions all cause the creation of unnecessary side issues and areas of cross-examination. The best practice is for all substantive communication to be done by phone.
Do be clear in what you are charging. It is not unusual for an expert to charge one hourly rate for record review, and a different rate for testimony. Your fee schedule should also note that any travel expenses you incur will also be invoiced. Your hourly rate should be appropriate for your area of practice. In our experience, gastroenterologists typically charge $400.00-$600.00 an hour for record review, and $550.00-$700.00 an hour for testimony.
Do not submit an invoice until after your deposition. Submitting invoices before your deposition creates unnecessary cross-examination issues. At the time of retention, speak to the attorney and ask if you will be able to submit invoices as you work. Most attorneys prefer invoices be submitted after your deposition. Because the wheels of justice often turn slowly, you could be waiting an equally long time to submit an invoice and get paid. One way to avoid this dilemma is to require a retainer at the time of retention.
Do not sign up with an expert finder service. Resist the urge to sign up with an expert finder service. The best medical experts come from referrals from other attorneys or physicians. Expert retention via an expert finder service creates the impression that you are a “hired gun” in the business of being a professional expert and can diminish your credibility. The finder services also charge a commission or fee.
As a gastroenterologist, you have the specialized knowledge to provide expert testimony regarding the cause of an injury and extent of damages in cases where you have treated a patient. You also have the type of education and training necessary to serve as an independent expert. Doing so is a serious task that can be time consuming and stressful. However, it can also be rewarding and allow you to make sure a fair and just outcome occurs.
This article is for general informational purposes only. Please consult your own attorney if you have questions. This information is not intended to create an attorney-client relationship.
Mr. Mills is an equity partner at Cunningham, Meyer & Vedrine PC in Chicago. Ms. Lindbert is a partner at Cunningham, Meyer & Vedrine PC. Both focus their practices on defending doctors and hospitals in medical malpractice actions.
Acting as an expert witness in a legal matter can be a nice way to compliment your practice. However, it is important to understand the role of experts, as well as their duties and obligations. Expert witnesses are called to testify on the basis of their specialized knowledge, not necessarily their direct knowledge of events and issues in the case.
Medical experts often play an important role in the evaluation, development, and preparation of a case long before it ever goes to trial. In some states, to even file a medical malpractice complaint a plaintiff is required to have the case evaluated by an expert and obtain a written report outlining why the plaintiff has a reasonable and meritorious cause for filing such an action.
There are different types of expert witness testimony. Experts can give opinion testimony as a physician who provided treatment to the plaintiff and whose conduct is not at issue. The second type of expert witness is a retained or controlled expert witness. This is a person giving opinion testimony after being retained by a lawyer on behalf of one of the parties to the lawsuit.
Before you give deposition or trial testimony, your opinions must be disclosed in writing and provided to the other parties in the case. In federal court, this is governed by Federal Rule of Civil Procedure 26. If the case is pending in state court, your written opinions are governed by local court rules. In both cases, the written opinions should be thorough and complete because you will not be allowed to testify to new opinions at the time of trial but will generally be allowed to expand upon those disclosed in writing at your deposition trial.
In order for a jury to hear your opinions at trial, your opinions must be reliable. In federal court, expert testimony is governed by Federal Rule of Evidence 702, which states:
A witness who is qualified as an expert by knowledge, skill, experience, training, or education may testify in the form of an opinion or otherwise if:
a) the expert’s scientific, technical, or other specialized knowledge will help the trier of fact to understand the evidence or to determine a fact in issue;
b) the testimony is based on sufficient facts or data;
c) the testimony is the product of reliable principles and methods; and
d) the expert has reliably applied the principles and methods to the facts of the case.
This means, that if a fact or evidence at issue involves scientific, technical, or specialized knowledge that is outside the scope of an ordinary layman’s experience, or involves complex issues challenging a layman’s comprehension, expert testimony is required. The scientific evidence must not just be relevant but also reliable. Expert opinions will be scrutinized to see if they are based on scientific testing or review of scientific data rather than just assumptions or speculation. Additionally, the experts must be qualified by their knowledge, skill, experience, training, or education. Given these parameters, it should come as no surprise that expert trial testimony is required for all medical malpractice cases.
Some states follow the “new or novel rule” which dictates that expert testimony is only admissible if the methodology or scientific principal on which the opinion is based is sufficiently established to have gained general acceptance in the particular field in which it belongs. This means that the evidence must be generally accepted as reliable in the relevant scientific community. New or novel techniques will be placed under the scrutiny of this standard. Courts will look at papers, books, journals, and case law to make a determination as to the reliability and general acceptance. Failure to meet the requisite standards may render a physician ineligible to testify.
If you are considering acting as an expert witness there are a few basic dos and don’ts to keep in mind:
Do be mindful of your criticism. If testifying in a medical malpractice case, you will be giving sworn testimony as to whether another physician deviated from the standard of care. Be aware that your testimony can later be used against you if your conduct is ever at issue, or if you contradict yourself in another case. Attorneys often look for prior testimony to use when questioning you at deposition and trial.
Do be aware of any applicable professional society guidelines. Many professional societies publish ethical guidelines as it relates to expert medical testimony. Be aware of those and know that you may be asked about them, especially if you are a member of that society.
Do be prepared for basic areas of cross-examination. There are a few tried and true areas that will always be the subject of cross-examination. Any perceived bias you may have, your fees, and whether you do more work for plaintiffs versus defendants are a just few examples. You should also be prepared to be cross-examined on the differences between personal practice (what you do) and an actual deviation from the standard of care.
Do keep written communication to a minimum. All communication between the expert physician and the attorney is potentially discoverable by the other side. The rules differ for state and federal courts. Emails, draft reports, and written questions all cause the creation of unnecessary side issues and areas of cross-examination. The best practice is for all substantive communication to be done by phone.
Do be clear in what you are charging. It is not unusual for an expert to charge one hourly rate for record review, and a different rate for testimony. Your fee schedule should also note that any travel expenses you incur will also be invoiced. Your hourly rate should be appropriate for your area of practice. In our experience, gastroenterologists typically charge $400.00-$600.00 an hour for record review, and $550.00-$700.00 an hour for testimony.
Do not submit an invoice until after your deposition. Submitting invoices before your deposition creates unnecessary cross-examination issues. At the time of retention, speak to the attorney and ask if you will be able to submit invoices as you work. Most attorneys prefer invoices be submitted after your deposition. Because the wheels of justice often turn slowly, you could be waiting an equally long time to submit an invoice and get paid. One way to avoid this dilemma is to require a retainer at the time of retention.
Do not sign up with an expert finder service. Resist the urge to sign up with an expert finder service. The best medical experts come from referrals from other attorneys or physicians. Expert retention via an expert finder service creates the impression that you are a “hired gun” in the business of being a professional expert and can diminish your credibility. The finder services also charge a commission or fee.
As a gastroenterologist, you have the specialized knowledge to provide expert testimony regarding the cause of an injury and extent of damages in cases where you have treated a patient. You also have the type of education and training necessary to serve as an independent expert. Doing so is a serious task that can be time consuming and stressful. However, it can also be rewarding and allow you to make sure a fair and just outcome occurs.
This article is for general informational purposes only. Please consult your own attorney if you have questions. This information is not intended to create an attorney-client relationship.
Mr. Mills is an equity partner at Cunningham, Meyer & Vedrine PC in Chicago. Ms. Lindbert is a partner at Cunningham, Meyer & Vedrine PC. Both focus their practices on defending doctors and hospitals in medical malpractice actions.
Tandem transplantation, long-term maintenance may extend MM remission
Tandem autologous hematopoietic stem cell transplantation (HSCT) could extend progression-free survival (PFS) for some patients with newly diagnosed multiple myeloma, based on long-term data from the phase 3 STaMINA trial.
While the intent-to-treat analysis showed no difference in 6-year PFS rate between single versus tandem HSCT, the as-treated analysis showed that patients who received two transplants had a 6-year PFS rate that was approximately 10% higher than those who received just one transplant, reported lead author Parameswaran Hari, MD, of the Medical College of Wisconsin, Milwaukee, who presented the findings as part of the American Society of Clinical Oncology virtual scientific program.
The STaMINA trial, also known as BMT CTN 0702, involved 758 patients who were randomized to receive one of three treatment regimens followed by 3 years of maintenance lenalidomide: tandem HSCT (auto/auto), single HSCT plus consolidation with lenalidomide/bortezomib/dexamethasone (auto/RVD), and single HSCT (auto/len).
“At the time, we intended the study to stop approximately 38 months from randomization, allowing for the time for transplant, and then 3 years of maintenance,” Dr. Hari said. However, as the results of lenalidomide maintenance in CALGB 00104 study were reported, they allowed for a follow-on protocol, which provided patients who are progression-free at the completion of the original STaMINA trial to go on to a second follow-on trial, which allowed lenalidomide maintenance on an indefinite basis, he added.
The present analysis looked at the long-term results of this follow-on trial, including the impact of discontinuing lenalidomide.
Aligning with the original study, the present intent-to-treat analysis showed no significant difference between treatment arms for 6-year PFS rate or overall survival. Respectively, PFS rates for auto/auto, auto/RVD, and auto/len were 43.9%, 39.7%, and 40.9% (P = .6).
But 32% of patients in the tandem group never underwent second HSCT, Dr. Hari noted, prompting the as-treated analysis. Although overall survival remained similar between groups, the 6-year PFS was significantly higher for patients who underwent tandem HSCT, at 49.4%, compared with 39.7% for auto/RVD and 38.6% for auto/len (P = .03).
Subgroup analysis showed the statistical benefit of tandem HSCT was driven by high-risk patients, who had a significantly better PFS after tandem transplant, compared with standard-risk patients, who showed no significant benefit.
Dr. Hari called the findings “provocative.”
“The tandem auto approach may still be relevant in high-risk multiple myeloma patients,” he said.
Dr. Hari and his colleagues also found that patients who stayed on maintenance lenalidomide after 38 months had a better 5-year PFS rate than those who discontinued maintenance therapy (79.5% vs. 61%; P = .0004). Subgroup analysis showed this benefit was statistically significant among patients with standard-risk disease (86.3% vs. 66%; P less than .001) but not among those in the high-risk subgroup (86.7% vs. 67.8%; P = .2).
However, Dr. Hari suggested that, based on the similarity of proportions between subgroups, the lack of significance in the high-risk subgroup was likely because of small sample size, suggesting the benefit of maintenance was actually shared across risk strata.
“Lenalidomide maintenance becomes a significant factor for preventing patients from progression,” Dr. Hari said, noting that the tandem transplant approach requires further study, and that he and his colleagues would soon publish minimal residual disease data.
He finished his presentation with a clear clinical recommendation. “Preplanned lenalidomide discontinuation at 3 years is not recommended based on inferior progression-free survival among those who stopped such therapy,” he said.
Invited discussant Joshua R. Richter, MD, of the Icahn School of Medicine at Mount Sinai, New York, said the findings encourage high-dose maintenance therapy, and for some, tandem HSCT.
“The STaMINA study presented today supports the notion that some patients with high-risk disease still may benefit and have further tumor burden reduction with the second transplant that leads to deeper remissions and hopefully abrogates diminished outcomes,” Dr. Richter said during a virtual presentation.
But improvements are needed to better identify such patients, Dr. Richter added. He highlighted a lack of standardization in risk modeling, with various factors currently employed, such as patient characteristics and genomic markers, among several others.
“Better definitions will allow us to cross compare and make true analyses about how to manage these patients,” Dr. Richter said. “Despite the improvements across the board that we’ve seen in myeloma patients, high-risk disease continues to represent a more complicated arena. And patients continue to suffer from worse outcomes, despite all of the other advances.”
The study was funded by the National Institutes of Health. The investigators disclosed additional relationships with Amgen, Celgene, Novartis, and others. Dr. Richter disclosed affiliations with Takeda, Sanofi, Janssen, and others.
SOURCE: Hari et al. ASCO 2020. Abstract 8506.
Tandem autologous hematopoietic stem cell transplantation (HSCT) could extend progression-free survival (PFS) for some patients with newly diagnosed multiple myeloma, based on long-term data from the phase 3 STaMINA trial.
While the intent-to-treat analysis showed no difference in 6-year PFS rate between single versus tandem HSCT, the as-treated analysis showed that patients who received two transplants had a 6-year PFS rate that was approximately 10% higher than those who received just one transplant, reported lead author Parameswaran Hari, MD, of the Medical College of Wisconsin, Milwaukee, who presented the findings as part of the American Society of Clinical Oncology virtual scientific program.
The STaMINA trial, also known as BMT CTN 0702, involved 758 patients who were randomized to receive one of three treatment regimens followed by 3 years of maintenance lenalidomide: tandem HSCT (auto/auto), single HSCT plus consolidation with lenalidomide/bortezomib/dexamethasone (auto/RVD), and single HSCT (auto/len).
“At the time, we intended the study to stop approximately 38 months from randomization, allowing for the time for transplant, and then 3 years of maintenance,” Dr. Hari said. However, as the results of lenalidomide maintenance in CALGB 00104 study were reported, they allowed for a follow-on protocol, which provided patients who are progression-free at the completion of the original STaMINA trial to go on to a second follow-on trial, which allowed lenalidomide maintenance on an indefinite basis, he added.
The present analysis looked at the long-term results of this follow-on trial, including the impact of discontinuing lenalidomide.
Aligning with the original study, the present intent-to-treat analysis showed no significant difference between treatment arms for 6-year PFS rate or overall survival. Respectively, PFS rates for auto/auto, auto/RVD, and auto/len were 43.9%, 39.7%, and 40.9% (P = .6).
But 32% of patients in the tandem group never underwent second HSCT, Dr. Hari noted, prompting the as-treated analysis. Although overall survival remained similar between groups, the 6-year PFS was significantly higher for patients who underwent tandem HSCT, at 49.4%, compared with 39.7% for auto/RVD and 38.6% for auto/len (P = .03).
Subgroup analysis showed the statistical benefit of tandem HSCT was driven by high-risk patients, who had a significantly better PFS after tandem transplant, compared with standard-risk patients, who showed no significant benefit.
Dr. Hari called the findings “provocative.”
“The tandem auto approach may still be relevant in high-risk multiple myeloma patients,” he said.
Dr. Hari and his colleagues also found that patients who stayed on maintenance lenalidomide after 38 months had a better 5-year PFS rate than those who discontinued maintenance therapy (79.5% vs. 61%; P = .0004). Subgroup analysis showed this benefit was statistically significant among patients with standard-risk disease (86.3% vs. 66%; P less than .001) but not among those in the high-risk subgroup (86.7% vs. 67.8%; P = .2).
However, Dr. Hari suggested that, based on the similarity of proportions between subgroups, the lack of significance in the high-risk subgroup was likely because of small sample size, suggesting the benefit of maintenance was actually shared across risk strata.
“Lenalidomide maintenance becomes a significant factor for preventing patients from progression,” Dr. Hari said, noting that the tandem transplant approach requires further study, and that he and his colleagues would soon publish minimal residual disease data.
He finished his presentation with a clear clinical recommendation. “Preplanned lenalidomide discontinuation at 3 years is not recommended based on inferior progression-free survival among those who stopped such therapy,” he said.
Invited discussant Joshua R. Richter, MD, of the Icahn School of Medicine at Mount Sinai, New York, said the findings encourage high-dose maintenance therapy, and for some, tandem HSCT.
“The STaMINA study presented today supports the notion that some patients with high-risk disease still may benefit and have further tumor burden reduction with the second transplant that leads to deeper remissions and hopefully abrogates diminished outcomes,” Dr. Richter said during a virtual presentation.
But improvements are needed to better identify such patients, Dr. Richter added. He highlighted a lack of standardization in risk modeling, with various factors currently employed, such as patient characteristics and genomic markers, among several others.
“Better definitions will allow us to cross compare and make true analyses about how to manage these patients,” Dr. Richter said. “Despite the improvements across the board that we’ve seen in myeloma patients, high-risk disease continues to represent a more complicated arena. And patients continue to suffer from worse outcomes, despite all of the other advances.”
The study was funded by the National Institutes of Health. The investigators disclosed additional relationships with Amgen, Celgene, Novartis, and others. Dr. Richter disclosed affiliations with Takeda, Sanofi, Janssen, and others.
SOURCE: Hari et al. ASCO 2020. Abstract 8506.
Tandem autologous hematopoietic stem cell transplantation (HSCT) could extend progression-free survival (PFS) for some patients with newly diagnosed multiple myeloma, based on long-term data from the phase 3 STaMINA trial.
While the intent-to-treat analysis showed no difference in 6-year PFS rate between single versus tandem HSCT, the as-treated analysis showed that patients who received two transplants had a 6-year PFS rate that was approximately 10% higher than those who received just one transplant, reported lead author Parameswaran Hari, MD, of the Medical College of Wisconsin, Milwaukee, who presented the findings as part of the American Society of Clinical Oncology virtual scientific program.
The STaMINA trial, also known as BMT CTN 0702, involved 758 patients who were randomized to receive one of three treatment regimens followed by 3 years of maintenance lenalidomide: tandem HSCT (auto/auto), single HSCT plus consolidation with lenalidomide/bortezomib/dexamethasone (auto/RVD), and single HSCT (auto/len).
“At the time, we intended the study to stop approximately 38 months from randomization, allowing for the time for transplant, and then 3 years of maintenance,” Dr. Hari said. However, as the results of lenalidomide maintenance in CALGB 00104 study were reported, they allowed for a follow-on protocol, which provided patients who are progression-free at the completion of the original STaMINA trial to go on to a second follow-on trial, which allowed lenalidomide maintenance on an indefinite basis, he added.
The present analysis looked at the long-term results of this follow-on trial, including the impact of discontinuing lenalidomide.
Aligning with the original study, the present intent-to-treat analysis showed no significant difference between treatment arms for 6-year PFS rate or overall survival. Respectively, PFS rates for auto/auto, auto/RVD, and auto/len were 43.9%, 39.7%, and 40.9% (P = .6).
But 32% of patients in the tandem group never underwent second HSCT, Dr. Hari noted, prompting the as-treated analysis. Although overall survival remained similar between groups, the 6-year PFS was significantly higher for patients who underwent tandem HSCT, at 49.4%, compared with 39.7% for auto/RVD and 38.6% for auto/len (P = .03).
Subgroup analysis showed the statistical benefit of tandem HSCT was driven by high-risk patients, who had a significantly better PFS after tandem transplant, compared with standard-risk patients, who showed no significant benefit.
Dr. Hari called the findings “provocative.”
“The tandem auto approach may still be relevant in high-risk multiple myeloma patients,” he said.
Dr. Hari and his colleagues also found that patients who stayed on maintenance lenalidomide after 38 months had a better 5-year PFS rate than those who discontinued maintenance therapy (79.5% vs. 61%; P = .0004). Subgroup analysis showed this benefit was statistically significant among patients with standard-risk disease (86.3% vs. 66%; P less than .001) but not among those in the high-risk subgroup (86.7% vs. 67.8%; P = .2).
However, Dr. Hari suggested that, based on the similarity of proportions between subgroups, the lack of significance in the high-risk subgroup was likely because of small sample size, suggesting the benefit of maintenance was actually shared across risk strata.
“Lenalidomide maintenance becomes a significant factor for preventing patients from progression,” Dr. Hari said, noting that the tandem transplant approach requires further study, and that he and his colleagues would soon publish minimal residual disease data.
He finished his presentation with a clear clinical recommendation. “Preplanned lenalidomide discontinuation at 3 years is not recommended based on inferior progression-free survival among those who stopped such therapy,” he said.
Invited discussant Joshua R. Richter, MD, of the Icahn School of Medicine at Mount Sinai, New York, said the findings encourage high-dose maintenance therapy, and for some, tandem HSCT.
“The STaMINA study presented today supports the notion that some patients with high-risk disease still may benefit and have further tumor burden reduction with the second transplant that leads to deeper remissions and hopefully abrogates diminished outcomes,” Dr. Richter said during a virtual presentation.
But improvements are needed to better identify such patients, Dr. Richter added. He highlighted a lack of standardization in risk modeling, with various factors currently employed, such as patient characteristics and genomic markers, among several others.
“Better definitions will allow us to cross compare and make true analyses about how to manage these patients,” Dr. Richter said. “Despite the improvements across the board that we’ve seen in myeloma patients, high-risk disease continues to represent a more complicated arena. And patients continue to suffer from worse outcomes, despite all of the other advances.”
The study was funded by the National Institutes of Health. The investigators disclosed additional relationships with Amgen, Celgene, Novartis, and others. Dr. Richter disclosed affiliations with Takeda, Sanofi, Janssen, and others.
SOURCE: Hari et al. ASCO 2020. Abstract 8506.
FROM ASCO 2020
The CDI APP adviser
A novel approach to APP documentation engagement
As hospitals and clinicians, we are facing increased scrutiny of the care we provide to our patients. There is increased demand for more transparency of our outcomes and a need for increased efficiency of the care we provide in the setting of already significant documentation burden and its known impact on provider burnout.
Clinical documentation integrity (CDI) is an instrumental department which supports complete and accurate documentation, serves as a bridge between physicians and hospital coders such that hospital reimbursement is appropriate and quality metrics are attributed appropriately to the hospital, service lines, and individual providers. Complete and accurate documentation also leads to the submission of coded/claims-based data reflecting provider true intent and to clinically valid data for research and patient centric purposes. For this reason, the physician adviser role as a liaison between physicians and CDI and coding, in addition to utilization management and case management, has become more commonplace. The physician adviser role has been a mainstay of CDI programs across the United States since as early as 2012.
At the University of Colorado Health (UCHealth), the physician adviser role first began in 2015 at our major academic medical center, the University of Colorado Hospital (UCH). That physician adviser, after the additional physician adviser FTE at UCH, having established relationships with physicians across service lines, began to focus on CDI-related education and communication as it pertained to inpatient documentation.
At our institution we have approximately 500 advance practice providers (APPs). Approximately two-thirds of the APPs care for inpatients on a myriad of different service lines and, along with physician learners from interns to fellows, complete the bulk of the documentation in the electronic health record.
In early 2018, the UCHealth office of advanced practice collaborated with CDI in its mission to optimize documentation with the aim to have a positive impact on reimbursement and quality metrics while highlighting APP value. In the relatively early stages of the collaboration it became evident that an APP adviser could be an innovative and effective approach in engaging our many APPs with CDI as faculty members who are generally service line based and, as such, invested in hospital and service line outcomes.
A business case for a new position of APP adviser for CDI was formulated based on not only the number of APP faculty and learners at our institution, but also on the premise that the level of consistency APPs provide would increase reliability in the adoption and adaptation of documentation practices as medicine and coding rules evolve. In addition, APP documentation can stand alone without physician attestation or signature, unlike physicians in training, further making them ideally suited collaborators. The position was approved by hospital leadership and the first APP adviser for CDI in the country (of whom we are aware) was hired at UCH in July 2019.
A dedicated APP CDI adviser facilitates the success of a CDI/APP collaboration through a better understanding of APP engagement needs largely by creating new and/or fostering existing relationships between the APP adviser and the APPs for each service line. The APP CDI adviser identifies the needs of the team in order to maximally enhance their documentation while illustrating how the work/collaboration can positively contribute to APP clinical and/or academic goals. The APP CDI adviser possesses a deeper knowledge of APP clinical work flow and how that work flow might be impacting the documentation. He or she utilizes information gathered from the APP team to create more efficient note templates, provide lunch and learns with different service line APPs, and offering 1:1 drop-in documentation support, allowing for more feedback flexibility in context of their clinical work flow.
This real time input may be received more positively and be perceived as less intimidating in the peer-to-peer context. The APP adviser also attends various educational forums to which the physician advisers may not have access. For example, the APP adviser attends monthly APP orientation to meet new APPs for the institution, attends APP council, is a member of the APP steering committee, and provides documentation tips for the APP monthly newsletter.
At this point we are in the process of collecting pre- and post data to illustrate the benefit of a CDI APP adviser (and the CDI APP collaboration as a whole) through metrics such as CC/MCC capture rate, case mix index, and mortality and length of stay as influenced by the level of complexity in documentation. We hope to add APPs as advisers across the UCHealth system over time and to continue to highlight and publish the experience and outcomes related to this innovative role as it evolves such that other institutions across the country will consider this type of collaboration.
Dr. Anoff is associate professor of clinical practice in the division of hospital medicine and medical director of clinical documentation integrity at University of Colorado Health, Denver. Ms. Brill is senior instructor in the department of neurosurgery and APP adviser of clinical documentation integrity at UCHealth Denver Metro.
A novel approach to APP documentation engagement
A novel approach to APP documentation engagement
As hospitals and clinicians, we are facing increased scrutiny of the care we provide to our patients. There is increased demand for more transparency of our outcomes and a need for increased efficiency of the care we provide in the setting of already significant documentation burden and its known impact on provider burnout.
Clinical documentation integrity (CDI) is an instrumental department which supports complete and accurate documentation, serves as a bridge between physicians and hospital coders such that hospital reimbursement is appropriate and quality metrics are attributed appropriately to the hospital, service lines, and individual providers. Complete and accurate documentation also leads to the submission of coded/claims-based data reflecting provider true intent and to clinically valid data for research and patient centric purposes. For this reason, the physician adviser role as a liaison between physicians and CDI and coding, in addition to utilization management and case management, has become more commonplace. The physician adviser role has been a mainstay of CDI programs across the United States since as early as 2012.
At the University of Colorado Health (UCHealth), the physician adviser role first began in 2015 at our major academic medical center, the University of Colorado Hospital (UCH). That physician adviser, after the additional physician adviser FTE at UCH, having established relationships with physicians across service lines, began to focus on CDI-related education and communication as it pertained to inpatient documentation.
At our institution we have approximately 500 advance practice providers (APPs). Approximately two-thirds of the APPs care for inpatients on a myriad of different service lines and, along with physician learners from interns to fellows, complete the bulk of the documentation in the electronic health record.
In early 2018, the UCHealth office of advanced practice collaborated with CDI in its mission to optimize documentation with the aim to have a positive impact on reimbursement and quality metrics while highlighting APP value. In the relatively early stages of the collaboration it became evident that an APP adviser could be an innovative and effective approach in engaging our many APPs with CDI as faculty members who are generally service line based and, as such, invested in hospital and service line outcomes.
A business case for a new position of APP adviser for CDI was formulated based on not only the number of APP faculty and learners at our institution, but also on the premise that the level of consistency APPs provide would increase reliability in the adoption and adaptation of documentation practices as medicine and coding rules evolve. In addition, APP documentation can stand alone without physician attestation or signature, unlike physicians in training, further making them ideally suited collaborators. The position was approved by hospital leadership and the first APP adviser for CDI in the country (of whom we are aware) was hired at UCH in July 2019.
A dedicated APP CDI adviser facilitates the success of a CDI/APP collaboration through a better understanding of APP engagement needs largely by creating new and/or fostering existing relationships between the APP adviser and the APPs for each service line. The APP CDI adviser identifies the needs of the team in order to maximally enhance their documentation while illustrating how the work/collaboration can positively contribute to APP clinical and/or academic goals. The APP CDI adviser possesses a deeper knowledge of APP clinical work flow and how that work flow might be impacting the documentation. He or she utilizes information gathered from the APP team to create more efficient note templates, provide lunch and learns with different service line APPs, and offering 1:1 drop-in documentation support, allowing for more feedback flexibility in context of their clinical work flow.
This real time input may be received more positively and be perceived as less intimidating in the peer-to-peer context. The APP adviser also attends various educational forums to which the physician advisers may not have access. For example, the APP adviser attends monthly APP orientation to meet new APPs for the institution, attends APP council, is a member of the APP steering committee, and provides documentation tips for the APP monthly newsletter.
At this point we are in the process of collecting pre- and post data to illustrate the benefit of a CDI APP adviser (and the CDI APP collaboration as a whole) through metrics such as CC/MCC capture rate, case mix index, and mortality and length of stay as influenced by the level of complexity in documentation. We hope to add APPs as advisers across the UCHealth system over time and to continue to highlight and publish the experience and outcomes related to this innovative role as it evolves such that other institutions across the country will consider this type of collaboration.
Dr. Anoff is associate professor of clinical practice in the division of hospital medicine and medical director of clinical documentation integrity at University of Colorado Health, Denver. Ms. Brill is senior instructor in the department of neurosurgery and APP adviser of clinical documentation integrity at UCHealth Denver Metro.
As hospitals and clinicians, we are facing increased scrutiny of the care we provide to our patients. There is increased demand for more transparency of our outcomes and a need for increased efficiency of the care we provide in the setting of already significant documentation burden and its known impact on provider burnout.
Clinical documentation integrity (CDI) is an instrumental department which supports complete and accurate documentation, serves as a bridge between physicians and hospital coders such that hospital reimbursement is appropriate and quality metrics are attributed appropriately to the hospital, service lines, and individual providers. Complete and accurate documentation also leads to the submission of coded/claims-based data reflecting provider true intent and to clinically valid data for research and patient centric purposes. For this reason, the physician adviser role as a liaison between physicians and CDI and coding, in addition to utilization management and case management, has become more commonplace. The physician adviser role has been a mainstay of CDI programs across the United States since as early as 2012.
At the University of Colorado Health (UCHealth), the physician adviser role first began in 2015 at our major academic medical center, the University of Colorado Hospital (UCH). That physician adviser, after the additional physician adviser FTE at UCH, having established relationships with physicians across service lines, began to focus on CDI-related education and communication as it pertained to inpatient documentation.
At our institution we have approximately 500 advance practice providers (APPs). Approximately two-thirds of the APPs care for inpatients on a myriad of different service lines and, along with physician learners from interns to fellows, complete the bulk of the documentation in the electronic health record.
In early 2018, the UCHealth office of advanced practice collaborated with CDI in its mission to optimize documentation with the aim to have a positive impact on reimbursement and quality metrics while highlighting APP value. In the relatively early stages of the collaboration it became evident that an APP adviser could be an innovative and effective approach in engaging our many APPs with CDI as faculty members who are generally service line based and, as such, invested in hospital and service line outcomes.
A business case for a new position of APP adviser for CDI was formulated based on not only the number of APP faculty and learners at our institution, but also on the premise that the level of consistency APPs provide would increase reliability in the adoption and adaptation of documentation practices as medicine and coding rules evolve. In addition, APP documentation can stand alone without physician attestation or signature, unlike physicians in training, further making them ideally suited collaborators. The position was approved by hospital leadership and the first APP adviser for CDI in the country (of whom we are aware) was hired at UCH in July 2019.
A dedicated APP CDI adviser facilitates the success of a CDI/APP collaboration through a better understanding of APP engagement needs largely by creating new and/or fostering existing relationships between the APP adviser and the APPs for each service line. The APP CDI adviser identifies the needs of the team in order to maximally enhance their documentation while illustrating how the work/collaboration can positively contribute to APP clinical and/or academic goals. The APP CDI adviser possesses a deeper knowledge of APP clinical work flow and how that work flow might be impacting the documentation. He or she utilizes information gathered from the APP team to create more efficient note templates, provide lunch and learns with different service line APPs, and offering 1:1 drop-in documentation support, allowing for more feedback flexibility in context of their clinical work flow.
This real time input may be received more positively and be perceived as less intimidating in the peer-to-peer context. The APP adviser also attends various educational forums to which the physician advisers may not have access. For example, the APP adviser attends monthly APP orientation to meet new APPs for the institution, attends APP council, is a member of the APP steering committee, and provides documentation tips for the APP monthly newsletter.
At this point we are in the process of collecting pre- and post data to illustrate the benefit of a CDI APP adviser (and the CDI APP collaboration as a whole) through metrics such as CC/MCC capture rate, case mix index, and mortality and length of stay as influenced by the level of complexity in documentation. We hope to add APPs as advisers across the UCHealth system over time and to continue to highlight and publish the experience and outcomes related to this innovative role as it evolves such that other institutions across the country will consider this type of collaboration.
Dr. Anoff is associate professor of clinical practice in the division of hospital medicine and medical director of clinical documentation integrity at University of Colorado Health, Denver. Ms. Brill is senior instructor in the department of neurosurgery and APP adviser of clinical documentation integrity at UCHealth Denver Metro.
One-fifth of stem cell transplantation patients develop PTSD
Approximately one-fifth of patients undergoing hematopoietic stem cell transplantation (HSCT) develop posttraumatic stress disorder (PTSD), based on a retrospective analysis.
Patient factors at time of transplantation, such as low quality of life and high anxiety, predicted PTSD 6 months later, reported lead author Sarah Griffith, MD, of Massachusetts General Hospital, Boston, who presented findings as part of the American Society of Clinical Oncology virtual scientific program.
“We know that patients admitted for HSCT are often isolated in the hospital for a prolonged period of time, usually about 3-4 weeks, and that they endure substantial toxicities that impact both their physical and psychological well-being,” Dr. Griffith said. “We also know from the literature that HSCT can be considered a traumatic event and that it may lead to clinically significant PTSD symptoms.” But studies evaluating the prevalence and characteristics of PTSD in this patient population have been lacking, she noted.
Dr. Griffith and her colleagues therefore conducted a retrospective analysis involving 250 adults with hematologic malignancies who underwent autologous or allogeneic HSCT during clinical trials conducted from 2014 to 2016. Median patient age was 56 years.
The first objective of the study was to measure the prevalence of PTSD. The second was to characterize features of PTSD such as intrusion, which entails reliving experiences in the form of nightmares or flashbacks, and hypervigilance, which encompasses insomnia, irritability, and hyperarousal for threat. The third objective was to determine risk factors at baseline.
At time of admission for HSCT, after 2 weeks of hospitalization, and again 6 months after transplantation, patients were evaluated using the Functional Assessment of Cancer Therapy–Bone Marrow Transplant (FACT-BMT), and the Hospital Anxiety and Depression Scale (HADS), which measured of quality of life, anxiety, and depression. Six months after HSCT, patients also underwent screening for PTSD with the Post-Traumatic Stress Checklist (PTSD-CL). Multivariate regression models were used to determine predictive risk factors.
Six months after HSCT, 18.9% of patients had clinically significant PTSD symptoms; most common were symptoms of avoidance (92.3%), hypervigilance (92.3%), and intrusion (76.9%). Among those who did not have clinically significant PTSD, almost one-quarter (24.5%) demonstrated significant hypervigilance, while 13.7% showed symptoms of avoidance.
“Clinically significant PTSD symptoms are common in the transplant population,” Dr. Griffith said.
Baseline predictors of PTSD included single status and lower quality of life. More severe PTSD was predicted by single status, younger age, higher baseline scores for anxiety or depression, and increased anxiety during hospitalization.
Concluding her presentation, Dr. Griffith said that the findings, while correlative and not causative, should prompt concern and intervention.
“It is very important to be aware of and to manage PTSD symptoms in these patients,” she said. “There are several baseline factors that can be identified prior to HSCT that may illuminate patients at risk for developing worse PTSD symptoms down the road, and these patients may benefit from tailored supportive care interventions.”
Specifically, Dr. Griffith recommended integrating palliative care into hospitalization, as this has been shown to reduce anxiety.
In a virtual presentation, invited discussant Nirali N. Shah, MD, of the National Cancer Institute, Bethesda, Md., highlighted the importance of the findings, while also noting that the impact of palliative care on risk of PTSD has yet to be demonstrated.
Dr. Shah suggested that future research may be improved through use of a formal diagnosis for PTSD, instead of a PTSD checklist, as was used in the present study.
“And certainly long-term follow-up would be important to evaluate the utility of this tool looking at symptoms beyond 6 months,” she said.
Dr. Shah went on to discuss the relevance of the findings for pediatric populations, as children may face unique risk factors and consequences related to PTSD.
“[PTSD in children] may be impacted by family dynamics and structure,” Dr. Shah said. “Children may also have significant neurocognitive implications as a result of their underlying disease or prior therapy. They may experience chronic pain as they go out into adulthood and long-term survivorship, and may also struggle with symptoms of anxiety and depression.”
According to Dr. Shah, one previous study involving more than 6,000 adult survivors of childhood cancer found that PTSD was relatively common, with prevalence rate of 9%, suggesting that interventional work is necessary.
“Applying the data in the study from Griffith et al. suggests that evaluation in the more proximal posttransplant period for children is needed to specifically evaluate PTSD and symptoms thereof, and to try to use this to identify an opportunity for intervention,” Dr. Shah said.
“Pediatric-specific assessments are essential to optimally capture disease and/or age-specific considerations,” she added.
The study was funded by the Lymphoma and Leukemia Society. The investigators disclosed additional relationships with Vector Oncology, Pfizer, AstraZeneca, and Gaido Health/BCG Digital Ventures.
SOURCE: Griffith et al. ASCO 2020. Abstract # 7505.
Approximately one-fifth of patients undergoing hematopoietic stem cell transplantation (HSCT) develop posttraumatic stress disorder (PTSD), based on a retrospective analysis.
Patient factors at time of transplantation, such as low quality of life and high anxiety, predicted PTSD 6 months later, reported lead author Sarah Griffith, MD, of Massachusetts General Hospital, Boston, who presented findings as part of the American Society of Clinical Oncology virtual scientific program.
“We know that patients admitted for HSCT are often isolated in the hospital for a prolonged period of time, usually about 3-4 weeks, and that they endure substantial toxicities that impact both their physical and psychological well-being,” Dr. Griffith said. “We also know from the literature that HSCT can be considered a traumatic event and that it may lead to clinically significant PTSD symptoms.” But studies evaluating the prevalence and characteristics of PTSD in this patient population have been lacking, she noted.
Dr. Griffith and her colleagues therefore conducted a retrospective analysis involving 250 adults with hematologic malignancies who underwent autologous or allogeneic HSCT during clinical trials conducted from 2014 to 2016. Median patient age was 56 years.
The first objective of the study was to measure the prevalence of PTSD. The second was to characterize features of PTSD such as intrusion, which entails reliving experiences in the form of nightmares or flashbacks, and hypervigilance, which encompasses insomnia, irritability, and hyperarousal for threat. The third objective was to determine risk factors at baseline.
At time of admission for HSCT, after 2 weeks of hospitalization, and again 6 months after transplantation, patients were evaluated using the Functional Assessment of Cancer Therapy–Bone Marrow Transplant (FACT-BMT), and the Hospital Anxiety and Depression Scale (HADS), which measured of quality of life, anxiety, and depression. Six months after HSCT, patients also underwent screening for PTSD with the Post-Traumatic Stress Checklist (PTSD-CL). Multivariate regression models were used to determine predictive risk factors.
Six months after HSCT, 18.9% of patients had clinically significant PTSD symptoms; most common were symptoms of avoidance (92.3%), hypervigilance (92.3%), and intrusion (76.9%). Among those who did not have clinically significant PTSD, almost one-quarter (24.5%) demonstrated significant hypervigilance, while 13.7% showed symptoms of avoidance.
“Clinically significant PTSD symptoms are common in the transplant population,” Dr. Griffith said.
Baseline predictors of PTSD included single status and lower quality of life. More severe PTSD was predicted by single status, younger age, higher baseline scores for anxiety or depression, and increased anxiety during hospitalization.
Concluding her presentation, Dr. Griffith said that the findings, while correlative and not causative, should prompt concern and intervention.
“It is very important to be aware of and to manage PTSD symptoms in these patients,” she said. “There are several baseline factors that can be identified prior to HSCT that may illuminate patients at risk for developing worse PTSD symptoms down the road, and these patients may benefit from tailored supportive care interventions.”
Specifically, Dr. Griffith recommended integrating palliative care into hospitalization, as this has been shown to reduce anxiety.
In a virtual presentation, invited discussant Nirali N. Shah, MD, of the National Cancer Institute, Bethesda, Md., highlighted the importance of the findings, while also noting that the impact of palliative care on risk of PTSD has yet to be demonstrated.
Dr. Shah suggested that future research may be improved through use of a formal diagnosis for PTSD, instead of a PTSD checklist, as was used in the present study.
“And certainly long-term follow-up would be important to evaluate the utility of this tool looking at symptoms beyond 6 months,” she said.
Dr. Shah went on to discuss the relevance of the findings for pediatric populations, as children may face unique risk factors and consequences related to PTSD.
“[PTSD in children] may be impacted by family dynamics and structure,” Dr. Shah said. “Children may also have significant neurocognitive implications as a result of their underlying disease or prior therapy. They may experience chronic pain as they go out into adulthood and long-term survivorship, and may also struggle with symptoms of anxiety and depression.”
According to Dr. Shah, one previous study involving more than 6,000 adult survivors of childhood cancer found that PTSD was relatively common, with prevalence rate of 9%, suggesting that interventional work is necessary.
“Applying the data in the study from Griffith et al. suggests that evaluation in the more proximal posttransplant period for children is needed to specifically evaluate PTSD and symptoms thereof, and to try to use this to identify an opportunity for intervention,” Dr. Shah said.
“Pediatric-specific assessments are essential to optimally capture disease and/or age-specific considerations,” she added.
The study was funded by the Lymphoma and Leukemia Society. The investigators disclosed additional relationships with Vector Oncology, Pfizer, AstraZeneca, and Gaido Health/BCG Digital Ventures.
SOURCE: Griffith et al. ASCO 2020. Abstract # 7505.
Approximately one-fifth of patients undergoing hematopoietic stem cell transplantation (HSCT) develop posttraumatic stress disorder (PTSD), based on a retrospective analysis.
Patient factors at time of transplantation, such as low quality of life and high anxiety, predicted PTSD 6 months later, reported lead author Sarah Griffith, MD, of Massachusetts General Hospital, Boston, who presented findings as part of the American Society of Clinical Oncology virtual scientific program.
“We know that patients admitted for HSCT are often isolated in the hospital for a prolonged period of time, usually about 3-4 weeks, and that they endure substantial toxicities that impact both their physical and psychological well-being,” Dr. Griffith said. “We also know from the literature that HSCT can be considered a traumatic event and that it may lead to clinically significant PTSD symptoms.” But studies evaluating the prevalence and characteristics of PTSD in this patient population have been lacking, she noted.
Dr. Griffith and her colleagues therefore conducted a retrospective analysis involving 250 adults with hematologic malignancies who underwent autologous or allogeneic HSCT during clinical trials conducted from 2014 to 2016. Median patient age was 56 years.
The first objective of the study was to measure the prevalence of PTSD. The second was to characterize features of PTSD such as intrusion, which entails reliving experiences in the form of nightmares or flashbacks, and hypervigilance, which encompasses insomnia, irritability, and hyperarousal for threat. The third objective was to determine risk factors at baseline.
At time of admission for HSCT, after 2 weeks of hospitalization, and again 6 months after transplantation, patients were evaluated using the Functional Assessment of Cancer Therapy–Bone Marrow Transplant (FACT-BMT), and the Hospital Anxiety and Depression Scale (HADS), which measured of quality of life, anxiety, and depression. Six months after HSCT, patients also underwent screening for PTSD with the Post-Traumatic Stress Checklist (PTSD-CL). Multivariate regression models were used to determine predictive risk factors.
Six months after HSCT, 18.9% of patients had clinically significant PTSD symptoms; most common were symptoms of avoidance (92.3%), hypervigilance (92.3%), and intrusion (76.9%). Among those who did not have clinically significant PTSD, almost one-quarter (24.5%) demonstrated significant hypervigilance, while 13.7% showed symptoms of avoidance.
“Clinically significant PTSD symptoms are common in the transplant population,” Dr. Griffith said.
Baseline predictors of PTSD included single status and lower quality of life. More severe PTSD was predicted by single status, younger age, higher baseline scores for anxiety or depression, and increased anxiety during hospitalization.
Concluding her presentation, Dr. Griffith said that the findings, while correlative and not causative, should prompt concern and intervention.
“It is very important to be aware of and to manage PTSD symptoms in these patients,” she said. “There are several baseline factors that can be identified prior to HSCT that may illuminate patients at risk for developing worse PTSD symptoms down the road, and these patients may benefit from tailored supportive care interventions.”
Specifically, Dr. Griffith recommended integrating palliative care into hospitalization, as this has been shown to reduce anxiety.
In a virtual presentation, invited discussant Nirali N. Shah, MD, of the National Cancer Institute, Bethesda, Md., highlighted the importance of the findings, while also noting that the impact of palliative care on risk of PTSD has yet to be demonstrated.
Dr. Shah suggested that future research may be improved through use of a formal diagnosis for PTSD, instead of a PTSD checklist, as was used in the present study.
“And certainly long-term follow-up would be important to evaluate the utility of this tool looking at symptoms beyond 6 months,” she said.
Dr. Shah went on to discuss the relevance of the findings for pediatric populations, as children may face unique risk factors and consequences related to PTSD.
“[PTSD in children] may be impacted by family dynamics and structure,” Dr. Shah said. “Children may also have significant neurocognitive implications as a result of their underlying disease or prior therapy. They may experience chronic pain as they go out into adulthood and long-term survivorship, and may also struggle with symptoms of anxiety and depression.”
According to Dr. Shah, one previous study involving more than 6,000 adult survivors of childhood cancer found that PTSD was relatively common, with prevalence rate of 9%, suggesting that interventional work is necessary.
“Applying the data in the study from Griffith et al. suggests that evaluation in the more proximal posttransplant period for children is needed to specifically evaluate PTSD and symptoms thereof, and to try to use this to identify an opportunity for intervention,” Dr. Shah said.
“Pediatric-specific assessments are essential to optimally capture disease and/or age-specific considerations,” she added.
The study was funded by the Lymphoma and Leukemia Society. The investigators disclosed additional relationships with Vector Oncology, Pfizer, AstraZeneca, and Gaido Health/BCG Digital Ventures.
SOURCE: Griffith et al. ASCO 2020. Abstract # 7505.
FROM ASCO 2020
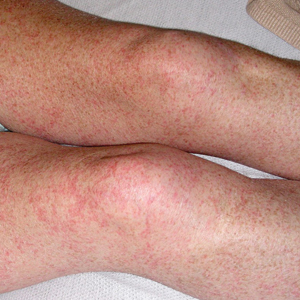
Petechial Rash on the Thighs in an Immunosuppressed Patient
The Diagnosis: Disseminated Strongyloidiasis
Strongyloidiasis is a parasitic infection caused by Strongyloides stercoralis. In the United States it is most prevalent in the Appalachian region. During the filariform larval stage of the parasite's life cycle, larvae from contaminated soil infect the human skin and spread to the intestinal epithelium,1 then the larvae mature into adult female worms that can produce eggs asexually. Rhabditiform larvae hatch from the eggs and are either excreted in the stool or develop into infectious filariform larvae. The latter can cause autoinfection of the intestinal mucosa or nearby skin; in addition, if the larvae enter the bloodstream, they can spread throughout the body and lead to disseminated strongyloidiasis and hyperinfection syndrome.2 This often fatal progression most commonly occurs in immunosuppressed individuals.3 The mortality rate has been reported to be up to 87%.2,4
Fever, abdominal pain, nausea, and diarrhea are clinically common in disseminated strongyloidiasis and hyperinfection syndrome.5 Patients also may exhibit dyspnea, cough, wheezing, and hemoptysis.2 Cutaneous manifestations are rare and typically include pruritus and petechiae.6 Eosinophilia may be present but is not a reliable indicator.1
Our patient displayed several risk factors and an early clinical presentation for disseminated strongyloidiasis and hyperinfection syndrome, which evolved over the course of hospitalization. Clues to the diagnosis included an immunosuppressed state; erythematous pruritic macules at presentation that later developed into reticulated petechial patches; and fever, general abdominal symptoms, and dyspnea. However, the patient's overall physical examination findings were subtle and nonspecific. Additionally, the patient did not display the classic larva currens for strongyloidiasis or the pathognomonic periumbilical thumbprint purpura of disseminated infection,6,7 which may indicate that the latter is a later-stage finding. Although graft-vs-host disease initially was suspected, a third skin biopsy revealed basophilic Strongyloides larvae, extravasated erythrocytes, and mild perivascular inflammation (Figure).
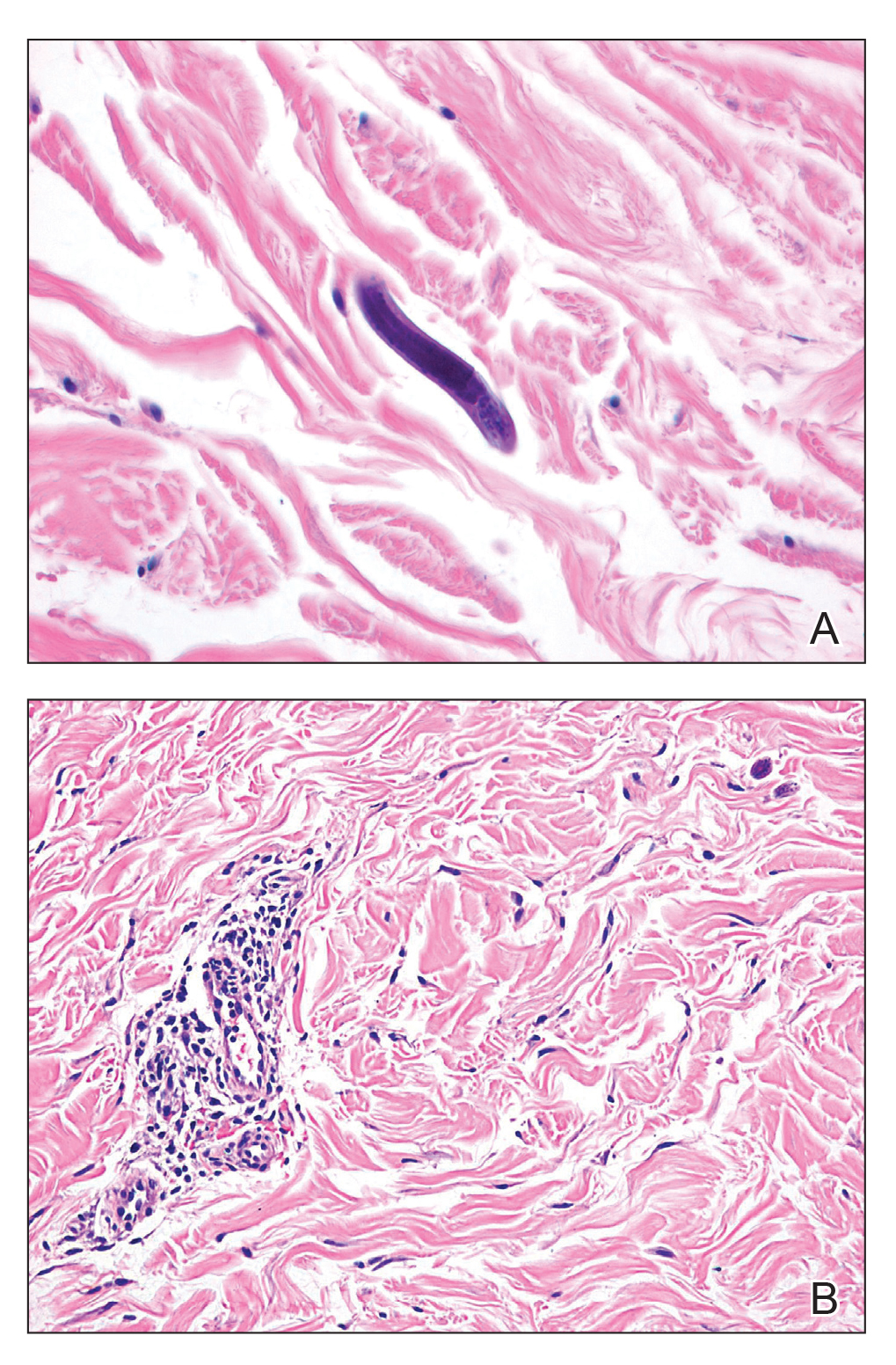
Subsequent gastric aspirates and stool cultures revealed S stercoralis. A bronchoalveolar lavage specimen and serum enzyme-linked immunosorbent assay for Strongyloides antibody were negative. The patient was treated with an extended 16-day course of ivermectin 12 mg daily until gastric aspirates and stool cultures were negative for the parasite. The rash receded by the end of the patient's 32-day hospital stay.
Because of the high mortality rate of untreated disseminated strongyloidiasis and hyperinfection syndrome, early diagnosis and initiation of anthelmintic treatment is vital in improving patient outcomes. As such, the diagnosis of disseminated strongyloidiasis should be considered in any immunosuppressed patient with multisystemic symptoms and/or petechiae. The differential diagnosis includes graft-vs-host disease, drug-induced urticaria, disseminated intravascular coagulation, and other opportunistic parasites.6,8,9
- Concha R, Harrington W Jr, Rogers AI. Intestinal strongyloidiasis: recognition, management, and determinants of outcome. J Clin Gastroenterol. 2005;39:203-211.
- Vadlamudi RS, Chi DS, Krishnaswamy G. Intestinal strongyloidiasis and hyperinfection syndrome. Clin Mol Allergy. 2006;4:8.
- Keiser PB, Nutman TB. Strongyloides stercoralis in the immunocompromised population. Clin Microbiol Rev. 2004;17:208-217.
- Chan FLY, Kennedy B, Nelson R. Fatal Strongyloides hyperinfection syndrome in an immunocompetent adult with review of the literature. Intern Med J. 2018;48:872-875.
- Scowden EB, Schaffner W, Stone WJ. Overwhelming strongyloidiasis: an unappreciated opportunistic infection. Medicine (Baltimore). 1978;57:527-544.
- von Kuster LC, Genta RM. Cutaneous manifestations of strongyloidiasis. Arch Dermatol. 1988;124:1826-1830.
- Weiser JA, Scully BE, Bulman WA, et al. Periumbilical parasitic thumbprint purpura: Strongyloides hyperinfection syndrome acquired from a cadaveric renal transplant. Transpl Infect Dis. 2011;13:58-62.
- Berenson CS, Dobuler KJ, Bia FJ. Fever, petechiae, and pulmonary infiltrates in an immunocompromised Peruvian man. Yale J Biol Med. 1987;60:437-445.
- Ly MN, Bethel SL, Usmani AS, et al. Cutaneous Strongyloides stercoralis infection: an unusual presentation. J Am Acad Dermatol. 2003;49(2 suppl case reports):S157-S160.
The Diagnosis: Disseminated Strongyloidiasis
Strongyloidiasis is a parasitic infection caused by Strongyloides stercoralis. In the United States it is most prevalent in the Appalachian region. During the filariform larval stage of the parasite's life cycle, larvae from contaminated soil infect the human skin and spread to the intestinal epithelium,1 then the larvae mature into adult female worms that can produce eggs asexually. Rhabditiform larvae hatch from the eggs and are either excreted in the stool or develop into infectious filariform larvae. The latter can cause autoinfection of the intestinal mucosa or nearby skin; in addition, if the larvae enter the bloodstream, they can spread throughout the body and lead to disseminated strongyloidiasis and hyperinfection syndrome.2 This often fatal progression most commonly occurs in immunosuppressed individuals.3 The mortality rate has been reported to be up to 87%.2,4
Fever, abdominal pain, nausea, and diarrhea are clinically common in disseminated strongyloidiasis and hyperinfection syndrome.5 Patients also may exhibit dyspnea, cough, wheezing, and hemoptysis.2 Cutaneous manifestations are rare and typically include pruritus and petechiae.6 Eosinophilia may be present but is not a reliable indicator.1
Our patient displayed several risk factors and an early clinical presentation for disseminated strongyloidiasis and hyperinfection syndrome, which evolved over the course of hospitalization. Clues to the diagnosis included an immunosuppressed state; erythematous pruritic macules at presentation that later developed into reticulated petechial patches; and fever, general abdominal symptoms, and dyspnea. However, the patient's overall physical examination findings were subtle and nonspecific. Additionally, the patient did not display the classic larva currens for strongyloidiasis or the pathognomonic periumbilical thumbprint purpura of disseminated infection,6,7 which may indicate that the latter is a later-stage finding. Although graft-vs-host disease initially was suspected, a third skin biopsy revealed basophilic Strongyloides larvae, extravasated erythrocytes, and mild perivascular inflammation (Figure).

Subsequent gastric aspirates and stool cultures revealed S stercoralis. A bronchoalveolar lavage specimen and serum enzyme-linked immunosorbent assay for Strongyloides antibody were negative. The patient was treated with an extended 16-day course of ivermectin 12 mg daily until gastric aspirates and stool cultures were negative for the parasite. The rash receded by the end of the patient's 32-day hospital stay.
Because of the high mortality rate of untreated disseminated strongyloidiasis and hyperinfection syndrome, early diagnosis and initiation of anthelmintic treatment is vital in improving patient outcomes. As such, the diagnosis of disseminated strongyloidiasis should be considered in any immunosuppressed patient with multisystemic symptoms and/or petechiae. The differential diagnosis includes graft-vs-host disease, drug-induced urticaria, disseminated intravascular coagulation, and other opportunistic parasites.6,8,9
The Diagnosis: Disseminated Strongyloidiasis
Strongyloidiasis is a parasitic infection caused by Strongyloides stercoralis. In the United States it is most prevalent in the Appalachian region. During the filariform larval stage of the parasite's life cycle, larvae from contaminated soil infect the human skin and spread to the intestinal epithelium,1 then the larvae mature into adult female worms that can produce eggs asexually. Rhabditiform larvae hatch from the eggs and are either excreted in the stool or develop into infectious filariform larvae. The latter can cause autoinfection of the intestinal mucosa or nearby skin; in addition, if the larvae enter the bloodstream, they can spread throughout the body and lead to disseminated strongyloidiasis and hyperinfection syndrome.2 This often fatal progression most commonly occurs in immunosuppressed individuals.3 The mortality rate has been reported to be up to 87%.2,4
Fever, abdominal pain, nausea, and diarrhea are clinically common in disseminated strongyloidiasis and hyperinfection syndrome.5 Patients also may exhibit dyspnea, cough, wheezing, and hemoptysis.2 Cutaneous manifestations are rare and typically include pruritus and petechiae.6 Eosinophilia may be present but is not a reliable indicator.1
Our patient displayed several risk factors and an early clinical presentation for disseminated strongyloidiasis and hyperinfection syndrome, which evolved over the course of hospitalization. Clues to the diagnosis included an immunosuppressed state; erythematous pruritic macules at presentation that later developed into reticulated petechial patches; and fever, general abdominal symptoms, and dyspnea. However, the patient's overall physical examination findings were subtle and nonspecific. Additionally, the patient did not display the classic larva currens for strongyloidiasis or the pathognomonic periumbilical thumbprint purpura of disseminated infection,6,7 which may indicate that the latter is a later-stage finding. Although graft-vs-host disease initially was suspected, a third skin biopsy revealed basophilic Strongyloides larvae, extravasated erythrocytes, and mild perivascular inflammation (Figure).

Subsequent gastric aspirates and stool cultures revealed S stercoralis. A bronchoalveolar lavage specimen and serum enzyme-linked immunosorbent assay for Strongyloides antibody were negative. The patient was treated with an extended 16-day course of ivermectin 12 mg daily until gastric aspirates and stool cultures were negative for the parasite. The rash receded by the end of the patient's 32-day hospital stay.
Because of the high mortality rate of untreated disseminated strongyloidiasis and hyperinfection syndrome, early diagnosis and initiation of anthelmintic treatment is vital in improving patient outcomes. As such, the diagnosis of disseminated strongyloidiasis should be considered in any immunosuppressed patient with multisystemic symptoms and/or petechiae. The differential diagnosis includes graft-vs-host disease, drug-induced urticaria, disseminated intravascular coagulation, and other opportunistic parasites.6,8,9
- Concha R, Harrington W Jr, Rogers AI. Intestinal strongyloidiasis: recognition, management, and determinants of outcome. J Clin Gastroenterol. 2005;39:203-211.
- Vadlamudi RS, Chi DS, Krishnaswamy G. Intestinal strongyloidiasis and hyperinfection syndrome. Clin Mol Allergy. 2006;4:8.
- Keiser PB, Nutman TB. Strongyloides stercoralis in the immunocompromised population. Clin Microbiol Rev. 2004;17:208-217.
- Chan FLY, Kennedy B, Nelson R. Fatal Strongyloides hyperinfection syndrome in an immunocompetent adult with review of the literature. Intern Med J. 2018;48:872-875.
- Scowden EB, Schaffner W, Stone WJ. Overwhelming strongyloidiasis: an unappreciated opportunistic infection. Medicine (Baltimore). 1978;57:527-544.
- von Kuster LC, Genta RM. Cutaneous manifestations of strongyloidiasis. Arch Dermatol. 1988;124:1826-1830.
- Weiser JA, Scully BE, Bulman WA, et al. Periumbilical parasitic thumbprint purpura: Strongyloides hyperinfection syndrome acquired from a cadaveric renal transplant. Transpl Infect Dis. 2011;13:58-62.
- Berenson CS, Dobuler KJ, Bia FJ. Fever, petechiae, and pulmonary infiltrates in an immunocompromised Peruvian man. Yale J Biol Med. 1987;60:437-445.
- Ly MN, Bethel SL, Usmani AS, et al. Cutaneous Strongyloides stercoralis infection: an unusual presentation. J Am Acad Dermatol. 2003;49(2 suppl case reports):S157-S160.
- Concha R, Harrington W Jr, Rogers AI. Intestinal strongyloidiasis: recognition, management, and determinants of outcome. J Clin Gastroenterol. 2005;39:203-211.
- Vadlamudi RS, Chi DS, Krishnaswamy G. Intestinal strongyloidiasis and hyperinfection syndrome. Clin Mol Allergy. 2006;4:8.
- Keiser PB, Nutman TB. Strongyloides stercoralis in the immunocompromised population. Clin Microbiol Rev. 2004;17:208-217.
- Chan FLY, Kennedy B, Nelson R. Fatal Strongyloides hyperinfection syndrome in an immunocompetent adult with review of the literature. Intern Med J. 2018;48:872-875.
- Scowden EB, Schaffner W, Stone WJ. Overwhelming strongyloidiasis: an unappreciated opportunistic infection. Medicine (Baltimore). 1978;57:527-544.
- von Kuster LC, Genta RM. Cutaneous manifestations of strongyloidiasis. Arch Dermatol. 1988;124:1826-1830.
- Weiser JA, Scully BE, Bulman WA, et al. Periumbilical parasitic thumbprint purpura: Strongyloides hyperinfection syndrome acquired from a cadaveric renal transplant. Transpl Infect Dis. 2011;13:58-62.
- Berenson CS, Dobuler KJ, Bia FJ. Fever, petechiae, and pulmonary infiltrates in an immunocompromised Peruvian man. Yale J Biol Med. 1987;60:437-445.
- Ly MN, Bethel SL, Usmani AS, et al. Cutaneous Strongyloides stercoralis infection: an unusual presentation. J Am Acad Dermatol. 2003;49(2 suppl case reports):S157-S160.
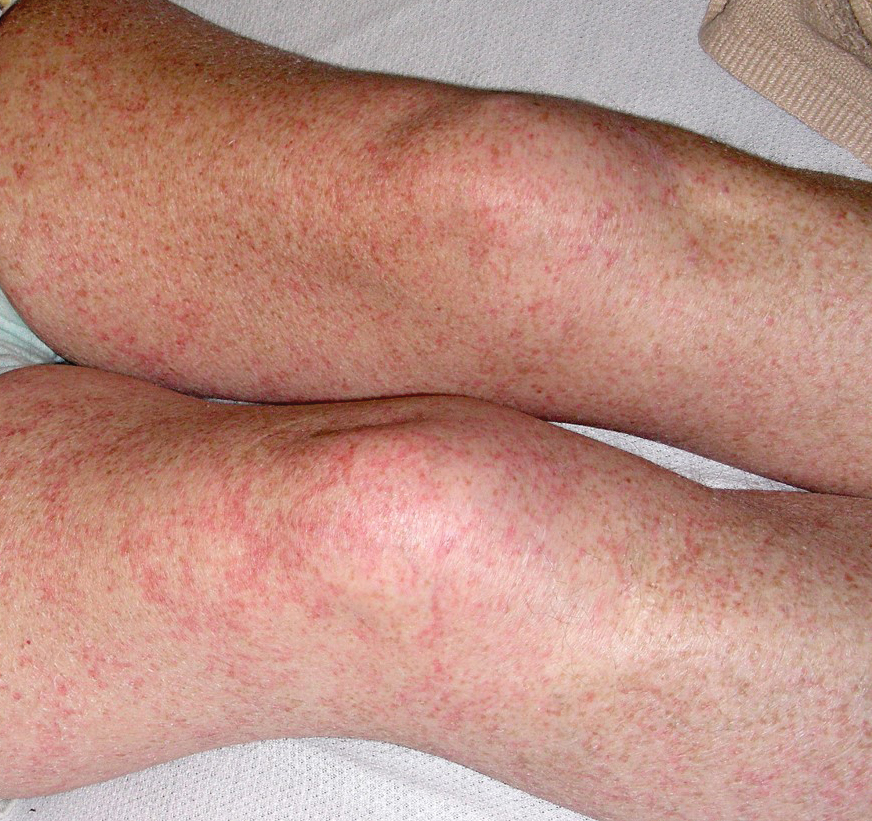
A 48-year-old woman from rural Virginia presented with centrifugally spreading, pruritic, blanchable macules over the lower abdomen and upper thighs noted 4 months after a pancreas transplant. After 3 weeks, the macules coalesced into reticulated nonblanching petechial patches. Fever, dyspnea, increasing xerosis, abdominal pain, and constipation were present. The patient had a medical history of type 1 diabetes mellitus requiring a pancreas transplant. Initial skin biopsy and fluorescence in situ hybridization to test for immune reaction to the XY-donor pancreas were negative. Mild transient eosinophilia was present at admission.
Lessons From the COVID-19 Pandemic: It’s Time to Invest in Public Health
What have you been doing since you left the US Public Health Service?
RADM Boris D. Lushniak, MD, MPH. I retired in 2015 and spent a year at the Uniformed Services University for the Health Sciences in Bethesda, Maryland as the Chair of Preventive Medicine and Biostatistics before I took the opportunity to become the Dean of the School of Public Health at the University of Maryland in College Park. I was very intrigued with that position. It’s a large and young school of public health—just 13 years since its inception. And it functions at both the undergraduate and graduate school levels. We have 2,400 undergraduates in 4 different degree paths. The intriguing part of this is the ability to influence a young person’s educational pathway, and for them to look at all the opportunities in public health, and to focus on a mission, which falls into the mission of the US Public Health Service (PHS) Commissioned Corps: Protect, promote and advance the health and safety of our nation.
It has been a very intriguing transition; I have been the Dean there for 3 years. Who would have predicted that things would change drastically in that time, both at the academic level (ie, moving a school from being a normal college environment to an online environment) and now moving into the realm of preparing for the near future of that university in terms of a potential reopening. It is using all of my public health experiences and putting it at that culmination point, which is my community of 52,000 people—40,000 students at the University in College Park, and 12,000 faculty and staff members.
We are responsible for making sure that the return is as safe as possible. With so many unknowns in the world of COVID-19 and so many unpredictable components, it is quite an undertaking to be able to determine for that community of 52,000 whether it’s time to return, and under what circumstances do we return.
In addition, we’re part of a larger community. The University of Maryland in College Park is in Prince George’s County, which is the epicenter of disease and death in Maryland. The School of Public Health is working closely with county authorities. Some of our students are now contact tracers. It’s been interesting to see our faculty, staff, and students standing up as a volunteer support structure for Public Health.
We have incredible research going on at the school. One of my prime research physicians, Don Milton, MD, DrPH, has been studying the transmission of influenza. Now his work is priming on not just influenza, but also COVID-19. Our hope is to establish a community that will be safe and healthy for everyone, and so it’s been an incredible amount of work.
How would you describe the federal/ local public health cooperation?
RADM Lushniak. First and foremost, we have seen a major issue in terms of state and local response to the COVID-19 pandemic. I have to congratulate the state and the local officials for doing as best as they can under the strained circumstances that they’re in.
The first strained circumstance is that local and state health departments have lost nearly a quarter of their workforce: 50,000 jobs across the country since the recession of 2008. Part of the answer why it’s been such a struggle is that our nation as a whole hasn’t looked at public health and hasn’t looked at prevention as a key component of how our country works. We have seen a lack of support at the state and the local level, the shedding of jobs, and the lack of foresight in terms of saying that prevention works and public health is important for our cities, states, regions, and the nation. We need to reemphasize that in terms of public health.
In the State of Maryland, in general, the counties are doing as best as they can under the circumstances. They certainly started out with trying to do as much testing as possible. Testing is a critical component to this response, and obviously, we have a situation nationwide with the testing still trying to be put online to the extent that it needs to be. We need to be able to test more and more individuals to be able to determine the people who are positive. The curve ball that COVID-19 threw us is that 25 to 50% of individuals who may have a positive test may be asymptomatic. So, this isn’t simple. It’s not a matter of just saying, “Okay, you’re sick. You may then have it.” It may be: “Hey, you’re feeling healthy, you still may have it.”
But just as important as testing is what you do with those individuals who are tested. You need to have health departments turning to these individuals and providing them directions of what needs to be done. If one is COVID-19-positive, one goes into isolation for at least 14 days. And if ill, they need to be connected with a medical care system. That’s an important part of the state and local response is making sure the individuals are properly directed to the right pathway.
In addition, contact tracing is critical. The way we’re going to fight COVID-19 is the ability for us to go out there and determine if you are a positive, who did you come in contact with, and did you potentially spread this to others? You need to direct individuals who may have been in contact with the person who is now COVID- 19-positive, saying “You may have to quarantine yourself, watch out for symptoms, and you have to be really careful in the meantime.”
State and local officials took up the burden of making decisions in terms of communicating the directions given to the population. Is stay at home required? Is it the closure of businesses? Is it the wearing of masks? Certainly, the issue of physical distancing plays a role.
All that was implemented at the state and local level. Under the circumstances, it has been done as well as possible, but that now reflects on the issue of the federal response. And the federal response, I’ll admit, has been less than I had hoped for on several realms.
Number one, coordination and direction from the federal level has been rather piecemeal. State and local officials, I think, were waiting for further directions. What did federal officials think; what did they want us to do? State and local officials want independence to implement things, but what’s the right answer? I think this has been not handled well at the highest levels of the US government.
Secondly, obviously, there was an issue with testing, and the responsibility here lays with the Centers of Disease Control and Prevention (CDC), which had problems from the get-go with setting up their testing caches and getting them out. We’re still catching up from there. Now it’s unfolding that the tie in between the federal government and the private sector and academic centers are at least making some headway on that testing front.
Third, people rely on the federal officials not only for action but also for communication. It really boils down to: Who’s in charge, who’s telling me the information that I need to know, who’s honest with me and telling me what they don’t know, and who has the insight to say, “Here’s how we’re going to find out the things that we don’t know?” Who’s there empathizing with the population?
The reality is there’s been a mismatch between the communication channels for the federal government and getting down not just to the state and locals but, also, to the general population in this country.
How would you characterize the US Public Health Service Response?
RADM Lushniak. I’ll first start off with kudos and congratulations to the Commissioned Corps of the PHS for their response to date. I think the latest numbers that ADM Brett Giroir, MD, Assistant Secretary of Health, told Congress in May, was that at the time more than 3,100 of the 6,100 current officers at the PHS have been deployed over the last several months. The reality is that the Commissioned Corps is out there doing service to our nation and to the world. PHS teams were deployed initially to Japan and the Diamond Princess cruise ship. The Corps been out there internationally.
Nationally, the Corps was at the Javits Center in New York assisting in setting up that medical response. They have been assisting at the military bases initially where some of the individuals who were coming in from China and other places were being held in quarantine. They have been assisting with investigations at nursing homes across the country and meat packing plants where there have been outbreaks occurring. The Commissioned Corps has been out there, so that’s the good news.
The bad news is that the Corps is a small uniformed service. The reality is nobody still is seeing the Corps or knows about the Corps as they’re out there doing their thing. It was very nice that ADM Giroir put a plug in for them in his recent congressional testimony. That’s great that our leadership is out there acknowledging the Corps. But to a large extent, I still have an issue with the Commissioned Corps being an underfunded uniformed service of this country. The Commissioned Corps is the only uniformed service in the world whose only mission is public health. But, lack of support reflects the idea of the lack of importance that public health plays in the minds of policy makers.
To a large extent, we have had no dollars in the Corps recently for training of officers to prepare for this. For 10 years we’ve waited for a Ready Reserve to be set up. The Ready Reserve component was part of the Affordable Care Act. I was in the office of the Surgeon General as we were told to ramp this up. Now 10 years later, in the midst of this COVID-19 pandemic, Congress finally has passed legislation that sets a pathway for a Ready Reserve.
Why is the Ready Reserve important? In essence, we have incredible public health professionals out there in the civilian ranks who would be willing to assist the Commissioned Corps in their mission, either to backfill critical positions where Corps officers are currently stationed and need to be deployed, or as a Ready Reserve that’s ready to deploy itself. All this is happening right now. I hope for better days, and I hope this COVID-19 pandemic will wake our nation up to the need of a Public Health Service Commissioned Corps, a uniformed service, that's out there doing good.
What lessons are we learning about public health in this pandemic?
RADM Lushniak. We’ve just developed a new space force, the 8th uniformed US service. In reality they are talking about tens of thousands of people assigned to it. Excuse me if I’m going to be assertive. I’m a big fan of space exploration. I realize that space is the final frontier and that perhaps we have to be able to defend our country in that regard. But we’re already saying that space is worth investing in. Where is the wisdom that we’re not investing in battling on this planet against emerging threats like COVID-19? And why is it that to this date the Commissioned Corps of the Public Health Service does not have its own budget; does not have a line item anywhere; does not have money directed for training; and, in essence, only serves because its officers are stationed at other agencies who pay for these officers? It’s a personnel system and not really treated as a key and critical uniformed service of this country. That’s point number one in terms of lessons learned and what needs to be done.
In addition, it’s not just the people in uniform who serve at the federal level, civilians serve as well. These civilians work at the CDC, at the US Food and Drug Administration, at the National Institute of Health, at the Indian Health Service, and at many, many other agencies throughout the US government. Within those realms, we need to show support of those federal practitioners who are working very diligently and in a devoted fashion to fight this pandemic as well. Part of it is the moral support to recognize that there are multiple fronts to fighting this pandemic and the federal practitioner who is working out there, is a key component to this.
I don’t want everything to be money, money, money, but the fact is that CDC’s budget has been decreasing over the years. How are we supposed to set up the laboratories, how are we supposed to demand the high level of expertise when, in fact, everything has to be done on a shoestring?
Finally, we notice public health in the midst of a crisis, but public health matters each and every day. The idea that the pandemic certainly brings to light what needs to get done, but without a pandemic, what do we have? We still have cigarette smoking, the number 1 killer in this country. That’s a public health issue. We have cardiovascular diseases as an extreme killer in this country. That’s a public health issue. We have diabetes mellitus that is rampant. We have substance abuse, including the opioid epidemic. Those are public health issues. We have hypertension, we have overweight and obesity. Those are all public health issues that public health battles each and every day without the recognition.
What we need is a major shift in the philosophy of this country to really take the health and wellness of our society as a key component of how you’ll raise that on to a pedestal—the idea that health and wellness is critical to the functioning of this country.
How have recent public health emergencies influenced the Commissioned Corps?
RADM Lushniak. The key feature is that the Public Health Service Commissioned Corps has been growing in its mission over the years. The pre-9/11 Commissioned Corps, was a different life. The post-9/11 world is the first time that the Commissioned Corps really fell into this idea of being America’s public health responders. I think that we ramped it up; we started out strong.
This was shown not only in the World Trade Center and the 9/11 disasters that occurred, but in the anthrax scenario that unfolded shortly afterwards. We saw it further continue in Hurricane Katrina and the multiple hurricane responses.
Then the Ebola response, in my last year of serving in uniform, was another action of both the civilian sector of federal responders as well as the uniformed sector. The beauty of that in terms of what we learned from Ebola was that coordination is key. That was the first time that the PHS worked so closely with the US Department of Defense and our sister services to basically have an international mission unfold with that level of coordination.
We can use those changes that have gone on, the metamorphoses that have happened over the years, as a jumping off point, but they need to be fulfilled with further growth and support of the Commissioned Corps of the US Public Health Service. The numbers are the lowest they’ve been in recent times in terms of active duty officers. That’s not a good thing. As the mission expands, the idea of recruiting and retaining remains a problem. We have to deal with it.
Was your interest in taking the position at the University of Maryland in part to help build the future of public health?
RADM Lushniak. Certainly, I was so excited to be at the University of Maryland College Park exactly for that reason. The undergraduates are coming in from high school and their eyes are wide open. Two things are important at that stage. One is to teach them about the beauty of public health. That it’s a bold and noble mission. As I always tell our students, it’s about the 3 Ps: Promoting health and wellbeing, preventing disease and injury, and prolonging a high quality of life.
When you put all those things together, that’s an incredible mission. I want to tell them at that young age, “Be a part of this, figure out where you fit in.” But it’s not for everyone. I tell my students that one of the major attributes that I need to see in a student is optimism. Public health does not deal well with pessimism. If your character is pessimistic, I actually dissuade you from becoming a public health person because there are a lot of barriers in this incredible bold and noble mission, and optimism needs to be a key feature that keeps us all going.
Next is the realization that there’s so many different public health issues in our world, so many different problems to deal with. I mentioned some of them previously in terms of the public health issues we see each and every day.
Let me talk about one that’s, in particular, shining through in the midst of COVID-19, but also shines through each and every day. That’s the issue of health equity in our communities. A young person, who usually comes in and wants to help their community, needs to realize that part of the battle of public health is to make sure that we deal with the disparities that exist. We must make health equity a key component of our jobs. We are here to serve others.
There’s a saying at the University of Maryland College Park that we’re a “Do good university.” I would say that public health is a do-good profession. It is about compassion, it’s about love, it’s about caring. Those are the types of people that I try to bring into the school, and I try to mentor and support.
What have you been doing since you left the US Public Health Service?
RADM Boris D. Lushniak, MD, MPH. I retired in 2015 and spent a year at the Uniformed Services University for the Health Sciences in Bethesda, Maryland as the Chair of Preventive Medicine and Biostatistics before I took the opportunity to become the Dean of the School of Public Health at the University of Maryland in College Park. I was very intrigued with that position. It’s a large and young school of public health—just 13 years since its inception. And it functions at both the undergraduate and graduate school levels. We have 2,400 undergraduates in 4 different degree paths. The intriguing part of this is the ability to influence a young person’s educational pathway, and for them to look at all the opportunities in public health, and to focus on a mission, which falls into the mission of the US Public Health Service (PHS) Commissioned Corps: Protect, promote and advance the health and safety of our nation.
It has been a very intriguing transition; I have been the Dean there for 3 years. Who would have predicted that things would change drastically in that time, both at the academic level (ie, moving a school from being a normal college environment to an online environment) and now moving into the realm of preparing for the near future of that university in terms of a potential reopening. It is using all of my public health experiences and putting it at that culmination point, which is my community of 52,000 people—40,000 students at the University in College Park, and 12,000 faculty and staff members.
We are responsible for making sure that the return is as safe as possible. With so many unknowns in the world of COVID-19 and so many unpredictable components, it is quite an undertaking to be able to determine for that community of 52,000 whether it’s time to return, and under what circumstances do we return.
In addition, we’re part of a larger community. The University of Maryland in College Park is in Prince George’s County, which is the epicenter of disease and death in Maryland. The School of Public Health is working closely with county authorities. Some of our students are now contact tracers. It’s been interesting to see our faculty, staff, and students standing up as a volunteer support structure for Public Health.
We have incredible research going on at the school. One of my prime research physicians, Don Milton, MD, DrPH, has been studying the transmission of influenza. Now his work is priming on not just influenza, but also COVID-19. Our hope is to establish a community that will be safe and healthy for everyone, and so it’s been an incredible amount of work.
How would you describe the federal/ local public health cooperation?
RADM Lushniak. First and foremost, we have seen a major issue in terms of state and local response to the COVID-19 pandemic. I have to congratulate the state and the local officials for doing as best as they can under the strained circumstances that they’re in.
The first strained circumstance is that local and state health departments have lost nearly a quarter of their workforce: 50,000 jobs across the country since the recession of 2008. Part of the answer why it’s been such a struggle is that our nation as a whole hasn’t looked at public health and hasn’t looked at prevention as a key component of how our country works. We have seen a lack of support at the state and the local level, the shedding of jobs, and the lack of foresight in terms of saying that prevention works and public health is important for our cities, states, regions, and the nation. We need to reemphasize that in terms of public health.
In the State of Maryland, in general, the counties are doing as best as they can under the circumstances. They certainly started out with trying to do as much testing as possible. Testing is a critical component to this response, and obviously, we have a situation nationwide with the testing still trying to be put online to the extent that it needs to be. We need to be able to test more and more individuals to be able to determine the people who are positive. The curve ball that COVID-19 threw us is that 25 to 50% of individuals who may have a positive test may be asymptomatic. So, this isn’t simple. It’s not a matter of just saying, “Okay, you’re sick. You may then have it.” It may be: “Hey, you’re feeling healthy, you still may have it.”
But just as important as testing is what you do with those individuals who are tested. You need to have health departments turning to these individuals and providing them directions of what needs to be done. If one is COVID-19-positive, one goes into isolation for at least 14 days. And if ill, they need to be connected with a medical care system. That’s an important part of the state and local response is making sure the individuals are properly directed to the right pathway.
In addition, contact tracing is critical. The way we’re going to fight COVID-19 is the ability for us to go out there and determine if you are a positive, who did you come in contact with, and did you potentially spread this to others? You need to direct individuals who may have been in contact with the person who is now COVID- 19-positive, saying “You may have to quarantine yourself, watch out for symptoms, and you have to be really careful in the meantime.”
State and local officials took up the burden of making decisions in terms of communicating the directions given to the population. Is stay at home required? Is it the closure of businesses? Is it the wearing of masks? Certainly, the issue of physical distancing plays a role.
All that was implemented at the state and local level. Under the circumstances, it has been done as well as possible, but that now reflects on the issue of the federal response. And the federal response, I’ll admit, has been less than I had hoped for on several realms.
Number one, coordination and direction from the federal level has been rather piecemeal. State and local officials, I think, were waiting for further directions. What did federal officials think; what did they want us to do? State and local officials want independence to implement things, but what’s the right answer? I think this has been not handled well at the highest levels of the US government.
Secondly, obviously, there was an issue with testing, and the responsibility here lays with the Centers of Disease Control and Prevention (CDC), which had problems from the get-go with setting up their testing caches and getting them out. We’re still catching up from there. Now it’s unfolding that the tie in between the federal government and the private sector and academic centers are at least making some headway on that testing front.
Third, people rely on the federal officials not only for action but also for communication. It really boils down to: Who’s in charge, who’s telling me the information that I need to know, who’s honest with me and telling me what they don’t know, and who has the insight to say, “Here’s how we’re going to find out the things that we don’t know?” Who’s there empathizing with the population?
The reality is there’s been a mismatch between the communication channels for the federal government and getting down not just to the state and locals but, also, to the general population in this country.
How would you characterize the US Public Health Service Response?
RADM Lushniak. I’ll first start off with kudos and congratulations to the Commissioned Corps of the PHS for their response to date. I think the latest numbers that ADM Brett Giroir, MD, Assistant Secretary of Health, told Congress in May, was that at the time more than 3,100 of the 6,100 current officers at the PHS have been deployed over the last several months. The reality is that the Commissioned Corps is out there doing service to our nation and to the world. PHS teams were deployed initially to Japan and the Diamond Princess cruise ship. The Corps been out there internationally.
Nationally, the Corps was at the Javits Center in New York assisting in setting up that medical response. They have been assisting at the military bases initially where some of the individuals who were coming in from China and other places were being held in quarantine. They have been assisting with investigations at nursing homes across the country and meat packing plants where there have been outbreaks occurring. The Commissioned Corps has been out there, so that’s the good news.
The bad news is that the Corps is a small uniformed service. The reality is nobody still is seeing the Corps or knows about the Corps as they’re out there doing their thing. It was very nice that ADM Giroir put a plug in for them in his recent congressional testimony. That’s great that our leadership is out there acknowledging the Corps. But to a large extent, I still have an issue with the Commissioned Corps being an underfunded uniformed service of this country. The Commissioned Corps is the only uniformed service in the world whose only mission is public health. But, lack of support reflects the idea of the lack of importance that public health plays in the minds of policy makers.
To a large extent, we have had no dollars in the Corps recently for training of officers to prepare for this. For 10 years we’ve waited for a Ready Reserve to be set up. The Ready Reserve component was part of the Affordable Care Act. I was in the office of the Surgeon General as we were told to ramp this up. Now 10 years later, in the midst of this COVID-19 pandemic, Congress finally has passed legislation that sets a pathway for a Ready Reserve.
Why is the Ready Reserve important? In essence, we have incredible public health professionals out there in the civilian ranks who would be willing to assist the Commissioned Corps in their mission, either to backfill critical positions where Corps officers are currently stationed and need to be deployed, or as a Ready Reserve that’s ready to deploy itself. All this is happening right now. I hope for better days, and I hope this COVID-19 pandemic will wake our nation up to the need of a Public Health Service Commissioned Corps, a uniformed service, that's out there doing good.
What lessons are we learning about public health in this pandemic?
RADM Lushniak. We’ve just developed a new space force, the 8th uniformed US service. In reality they are talking about tens of thousands of people assigned to it. Excuse me if I’m going to be assertive. I’m a big fan of space exploration. I realize that space is the final frontier and that perhaps we have to be able to defend our country in that regard. But we’re already saying that space is worth investing in. Where is the wisdom that we’re not investing in battling on this planet against emerging threats like COVID-19? And why is it that to this date the Commissioned Corps of the Public Health Service does not have its own budget; does not have a line item anywhere; does not have money directed for training; and, in essence, only serves because its officers are stationed at other agencies who pay for these officers? It’s a personnel system and not really treated as a key and critical uniformed service of this country. That’s point number one in terms of lessons learned and what needs to be done.
In addition, it’s not just the people in uniform who serve at the federal level, civilians serve as well. These civilians work at the CDC, at the US Food and Drug Administration, at the National Institute of Health, at the Indian Health Service, and at many, many other agencies throughout the US government. Within those realms, we need to show support of those federal practitioners who are working very diligently and in a devoted fashion to fight this pandemic as well. Part of it is the moral support to recognize that there are multiple fronts to fighting this pandemic and the federal practitioner who is working out there, is a key component to this.
I don’t want everything to be money, money, money, but the fact is that CDC’s budget has been decreasing over the years. How are we supposed to set up the laboratories, how are we supposed to demand the high level of expertise when, in fact, everything has to be done on a shoestring?
Finally, we notice public health in the midst of a crisis, but public health matters each and every day. The idea that the pandemic certainly brings to light what needs to get done, but without a pandemic, what do we have? We still have cigarette smoking, the number 1 killer in this country. That’s a public health issue. We have cardiovascular diseases as an extreme killer in this country. That’s a public health issue. We have diabetes mellitus that is rampant. We have substance abuse, including the opioid epidemic. Those are public health issues. We have hypertension, we have overweight and obesity. Those are all public health issues that public health battles each and every day without the recognition.
What we need is a major shift in the philosophy of this country to really take the health and wellness of our society as a key component of how you’ll raise that on to a pedestal—the idea that health and wellness is critical to the functioning of this country.
How have recent public health emergencies influenced the Commissioned Corps?
RADM Lushniak. The key feature is that the Public Health Service Commissioned Corps has been growing in its mission over the years. The pre-9/11 Commissioned Corps, was a different life. The post-9/11 world is the first time that the Commissioned Corps really fell into this idea of being America’s public health responders. I think that we ramped it up; we started out strong.
This was shown not only in the World Trade Center and the 9/11 disasters that occurred, but in the anthrax scenario that unfolded shortly afterwards. We saw it further continue in Hurricane Katrina and the multiple hurricane responses.
Then the Ebola response, in my last year of serving in uniform, was another action of both the civilian sector of federal responders as well as the uniformed sector. The beauty of that in terms of what we learned from Ebola was that coordination is key. That was the first time that the PHS worked so closely with the US Department of Defense and our sister services to basically have an international mission unfold with that level of coordination.
We can use those changes that have gone on, the metamorphoses that have happened over the years, as a jumping off point, but they need to be fulfilled with further growth and support of the Commissioned Corps of the US Public Health Service. The numbers are the lowest they’ve been in recent times in terms of active duty officers. That’s not a good thing. As the mission expands, the idea of recruiting and retaining remains a problem. We have to deal with it.
Was your interest in taking the position at the University of Maryland in part to help build the future of public health?
RADM Lushniak. Certainly, I was so excited to be at the University of Maryland College Park exactly for that reason. The undergraduates are coming in from high school and their eyes are wide open. Two things are important at that stage. One is to teach them about the beauty of public health. That it’s a bold and noble mission. As I always tell our students, it’s about the 3 Ps: Promoting health and wellbeing, preventing disease and injury, and prolonging a high quality of life.
When you put all those things together, that’s an incredible mission. I want to tell them at that young age, “Be a part of this, figure out where you fit in.” But it’s not for everyone. I tell my students that one of the major attributes that I need to see in a student is optimism. Public health does not deal well with pessimism. If your character is pessimistic, I actually dissuade you from becoming a public health person because there are a lot of barriers in this incredible bold and noble mission, and optimism needs to be a key feature that keeps us all going.
Next is the realization that there’s so many different public health issues in our world, so many different problems to deal with. I mentioned some of them previously in terms of the public health issues we see each and every day.
Let me talk about one that’s, in particular, shining through in the midst of COVID-19, but also shines through each and every day. That’s the issue of health equity in our communities. A young person, who usually comes in and wants to help their community, needs to realize that part of the battle of public health is to make sure that we deal with the disparities that exist. We must make health equity a key component of our jobs. We are here to serve others.
There’s a saying at the University of Maryland College Park that we’re a “Do good university.” I would say that public health is a do-good profession. It is about compassion, it’s about love, it’s about caring. Those are the types of people that I try to bring into the school, and I try to mentor and support.
What have you been doing since you left the US Public Health Service?
RADM Boris D. Lushniak, MD, MPH. I retired in 2015 and spent a year at the Uniformed Services University for the Health Sciences in Bethesda, Maryland as the Chair of Preventive Medicine and Biostatistics before I took the opportunity to become the Dean of the School of Public Health at the University of Maryland in College Park. I was very intrigued with that position. It’s a large and young school of public health—just 13 years since its inception. And it functions at both the undergraduate and graduate school levels. We have 2,400 undergraduates in 4 different degree paths. The intriguing part of this is the ability to influence a young person’s educational pathway, and for them to look at all the opportunities in public health, and to focus on a mission, which falls into the mission of the US Public Health Service (PHS) Commissioned Corps: Protect, promote and advance the health and safety of our nation.
It has been a very intriguing transition; I have been the Dean there for 3 years. Who would have predicted that things would change drastically in that time, both at the academic level (ie, moving a school from being a normal college environment to an online environment) and now moving into the realm of preparing for the near future of that university in terms of a potential reopening. It is using all of my public health experiences and putting it at that culmination point, which is my community of 52,000 people—40,000 students at the University in College Park, and 12,000 faculty and staff members.
We are responsible for making sure that the return is as safe as possible. With so many unknowns in the world of COVID-19 and so many unpredictable components, it is quite an undertaking to be able to determine for that community of 52,000 whether it’s time to return, and under what circumstances do we return.
In addition, we’re part of a larger community. The University of Maryland in College Park is in Prince George’s County, which is the epicenter of disease and death in Maryland. The School of Public Health is working closely with county authorities. Some of our students are now contact tracers. It’s been interesting to see our faculty, staff, and students standing up as a volunteer support structure for Public Health.
We have incredible research going on at the school. One of my prime research physicians, Don Milton, MD, DrPH, has been studying the transmission of influenza. Now his work is priming on not just influenza, but also COVID-19. Our hope is to establish a community that will be safe and healthy for everyone, and so it’s been an incredible amount of work.
How would you describe the federal/ local public health cooperation?
RADM Lushniak. First and foremost, we have seen a major issue in terms of state and local response to the COVID-19 pandemic. I have to congratulate the state and the local officials for doing as best as they can under the strained circumstances that they’re in.
The first strained circumstance is that local and state health departments have lost nearly a quarter of their workforce: 50,000 jobs across the country since the recession of 2008. Part of the answer why it’s been such a struggle is that our nation as a whole hasn’t looked at public health and hasn’t looked at prevention as a key component of how our country works. We have seen a lack of support at the state and the local level, the shedding of jobs, and the lack of foresight in terms of saying that prevention works and public health is important for our cities, states, regions, and the nation. We need to reemphasize that in terms of public health.
In the State of Maryland, in general, the counties are doing as best as they can under the circumstances. They certainly started out with trying to do as much testing as possible. Testing is a critical component to this response, and obviously, we have a situation nationwide with the testing still trying to be put online to the extent that it needs to be. We need to be able to test more and more individuals to be able to determine the people who are positive. The curve ball that COVID-19 threw us is that 25 to 50% of individuals who may have a positive test may be asymptomatic. So, this isn’t simple. It’s not a matter of just saying, “Okay, you’re sick. You may then have it.” It may be: “Hey, you’re feeling healthy, you still may have it.”
But just as important as testing is what you do with those individuals who are tested. You need to have health departments turning to these individuals and providing them directions of what needs to be done. If one is COVID-19-positive, one goes into isolation for at least 14 days. And if ill, they need to be connected with a medical care system. That’s an important part of the state and local response is making sure the individuals are properly directed to the right pathway.
In addition, contact tracing is critical. The way we’re going to fight COVID-19 is the ability for us to go out there and determine if you are a positive, who did you come in contact with, and did you potentially spread this to others? You need to direct individuals who may have been in contact with the person who is now COVID- 19-positive, saying “You may have to quarantine yourself, watch out for symptoms, and you have to be really careful in the meantime.”
State and local officials took up the burden of making decisions in terms of communicating the directions given to the population. Is stay at home required? Is it the closure of businesses? Is it the wearing of masks? Certainly, the issue of physical distancing plays a role.
All that was implemented at the state and local level. Under the circumstances, it has been done as well as possible, but that now reflects on the issue of the federal response. And the federal response, I’ll admit, has been less than I had hoped for on several realms.
Number one, coordination and direction from the federal level has been rather piecemeal. State and local officials, I think, were waiting for further directions. What did federal officials think; what did they want us to do? State and local officials want independence to implement things, but what’s the right answer? I think this has been not handled well at the highest levels of the US government.
Secondly, obviously, there was an issue with testing, and the responsibility here lays with the Centers of Disease Control and Prevention (CDC), which had problems from the get-go with setting up their testing caches and getting them out. We’re still catching up from there. Now it’s unfolding that the tie in between the federal government and the private sector and academic centers are at least making some headway on that testing front.
Third, people rely on the federal officials not only for action but also for communication. It really boils down to: Who’s in charge, who’s telling me the information that I need to know, who’s honest with me and telling me what they don’t know, and who has the insight to say, “Here’s how we’re going to find out the things that we don’t know?” Who’s there empathizing with the population?
The reality is there’s been a mismatch between the communication channels for the federal government and getting down not just to the state and locals but, also, to the general population in this country.
How would you characterize the US Public Health Service Response?
RADM Lushniak. I’ll first start off with kudos and congratulations to the Commissioned Corps of the PHS for their response to date. I think the latest numbers that ADM Brett Giroir, MD, Assistant Secretary of Health, told Congress in May, was that at the time more than 3,100 of the 6,100 current officers at the PHS have been deployed over the last several months. The reality is that the Commissioned Corps is out there doing service to our nation and to the world. PHS teams were deployed initially to Japan and the Diamond Princess cruise ship. The Corps been out there internationally.
Nationally, the Corps was at the Javits Center in New York assisting in setting up that medical response. They have been assisting at the military bases initially where some of the individuals who were coming in from China and other places were being held in quarantine. They have been assisting with investigations at nursing homes across the country and meat packing plants where there have been outbreaks occurring. The Commissioned Corps has been out there, so that’s the good news.
The bad news is that the Corps is a small uniformed service. The reality is nobody still is seeing the Corps or knows about the Corps as they’re out there doing their thing. It was very nice that ADM Giroir put a plug in for them in his recent congressional testimony. That’s great that our leadership is out there acknowledging the Corps. But to a large extent, I still have an issue with the Commissioned Corps being an underfunded uniformed service of this country. The Commissioned Corps is the only uniformed service in the world whose only mission is public health. But, lack of support reflects the idea of the lack of importance that public health plays in the minds of policy makers.
To a large extent, we have had no dollars in the Corps recently for training of officers to prepare for this. For 10 years we’ve waited for a Ready Reserve to be set up. The Ready Reserve component was part of the Affordable Care Act. I was in the office of the Surgeon General as we were told to ramp this up. Now 10 years later, in the midst of this COVID-19 pandemic, Congress finally has passed legislation that sets a pathway for a Ready Reserve.
Why is the Ready Reserve important? In essence, we have incredible public health professionals out there in the civilian ranks who would be willing to assist the Commissioned Corps in their mission, either to backfill critical positions where Corps officers are currently stationed and need to be deployed, or as a Ready Reserve that’s ready to deploy itself. All this is happening right now. I hope for better days, and I hope this COVID-19 pandemic will wake our nation up to the need of a Public Health Service Commissioned Corps, a uniformed service, that's out there doing good.
What lessons are we learning about public health in this pandemic?
RADM Lushniak. We’ve just developed a new space force, the 8th uniformed US service. In reality they are talking about tens of thousands of people assigned to it. Excuse me if I’m going to be assertive. I’m a big fan of space exploration. I realize that space is the final frontier and that perhaps we have to be able to defend our country in that regard. But we’re already saying that space is worth investing in. Where is the wisdom that we’re not investing in battling on this planet against emerging threats like COVID-19? And why is it that to this date the Commissioned Corps of the Public Health Service does not have its own budget; does not have a line item anywhere; does not have money directed for training; and, in essence, only serves because its officers are stationed at other agencies who pay for these officers? It’s a personnel system and not really treated as a key and critical uniformed service of this country. That’s point number one in terms of lessons learned and what needs to be done.
In addition, it’s not just the people in uniform who serve at the federal level, civilians serve as well. These civilians work at the CDC, at the US Food and Drug Administration, at the National Institute of Health, at the Indian Health Service, and at many, many other agencies throughout the US government. Within those realms, we need to show support of those federal practitioners who are working very diligently and in a devoted fashion to fight this pandemic as well. Part of it is the moral support to recognize that there are multiple fronts to fighting this pandemic and the federal practitioner who is working out there, is a key component to this.
I don’t want everything to be money, money, money, but the fact is that CDC’s budget has been decreasing over the years. How are we supposed to set up the laboratories, how are we supposed to demand the high level of expertise when, in fact, everything has to be done on a shoestring?
Finally, we notice public health in the midst of a crisis, but public health matters each and every day. The idea that the pandemic certainly brings to light what needs to get done, but without a pandemic, what do we have? We still have cigarette smoking, the number 1 killer in this country. That’s a public health issue. We have cardiovascular diseases as an extreme killer in this country. That’s a public health issue. We have diabetes mellitus that is rampant. We have substance abuse, including the opioid epidemic. Those are public health issues. We have hypertension, we have overweight and obesity. Those are all public health issues that public health battles each and every day without the recognition.
What we need is a major shift in the philosophy of this country to really take the health and wellness of our society as a key component of how you’ll raise that on to a pedestal—the idea that health and wellness is critical to the functioning of this country.
How have recent public health emergencies influenced the Commissioned Corps?
RADM Lushniak. The key feature is that the Public Health Service Commissioned Corps has been growing in its mission over the years. The pre-9/11 Commissioned Corps, was a different life. The post-9/11 world is the first time that the Commissioned Corps really fell into this idea of being America’s public health responders. I think that we ramped it up; we started out strong.
This was shown not only in the World Trade Center and the 9/11 disasters that occurred, but in the anthrax scenario that unfolded shortly afterwards. We saw it further continue in Hurricane Katrina and the multiple hurricane responses.
Then the Ebola response, in my last year of serving in uniform, was another action of both the civilian sector of federal responders as well as the uniformed sector. The beauty of that in terms of what we learned from Ebola was that coordination is key. That was the first time that the PHS worked so closely with the US Department of Defense and our sister services to basically have an international mission unfold with that level of coordination.
We can use those changes that have gone on, the metamorphoses that have happened over the years, as a jumping off point, but they need to be fulfilled with further growth and support of the Commissioned Corps of the US Public Health Service. The numbers are the lowest they’ve been in recent times in terms of active duty officers. That’s not a good thing. As the mission expands, the idea of recruiting and retaining remains a problem. We have to deal with it.
Was your interest in taking the position at the University of Maryland in part to help build the future of public health?
RADM Lushniak. Certainly, I was so excited to be at the University of Maryland College Park exactly for that reason. The undergraduates are coming in from high school and their eyes are wide open. Two things are important at that stage. One is to teach them about the beauty of public health. That it’s a bold and noble mission. As I always tell our students, it’s about the 3 Ps: Promoting health and wellbeing, preventing disease and injury, and prolonging a high quality of life.
When you put all those things together, that’s an incredible mission. I want to tell them at that young age, “Be a part of this, figure out where you fit in.” But it’s not for everyone. I tell my students that one of the major attributes that I need to see in a student is optimism. Public health does not deal well with pessimism. If your character is pessimistic, I actually dissuade you from becoming a public health person because there are a lot of barriers in this incredible bold and noble mission, and optimism needs to be a key feature that keeps us all going.
Next is the realization that there’s so many different public health issues in our world, so many different problems to deal with. I mentioned some of them previously in terms of the public health issues we see each and every day.
Let me talk about one that’s, in particular, shining through in the midst of COVID-19, but also shines through each and every day. That’s the issue of health equity in our communities. A young person, who usually comes in and wants to help their community, needs to realize that part of the battle of public health is to make sure that we deal with the disparities that exist. We must make health equity a key component of our jobs. We are here to serve others.
There’s a saying at the University of Maryland College Park that we’re a “Do good university.” I would say that public health is a do-good profession. It is about compassion, it’s about love, it’s about caring. Those are the types of people that I try to bring into the school, and I try to mentor and support.
Open Clinical Trials for Patients With COVID-19
Finding effective treatment or a vaccine for COVID-19, the disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has placed significant strains on the global health care system. The National Library of Medicine database lists > 1,800 trials that are aimed at addressing COVID-19-related health care. Already, trials developed by the US Department of Veterans Affairs (VA), US Department of Defense (DoD), and the National Institute of Allergy and Infectious Diseases have provided important data on effective treatment options. The clinical trials listed below are all open as of May 31, 2020 and have trial sites at VA and DoD facilities. For additional information and full inclusion/exclusion criteria, please consult clinicaltrials.gov.
Adaptive COVID-19 Treatment Trial (ACTT)
This study is an adaptive, randomized, double-blind, placebo-controlled trial to evaluate the safety and efficacy of novel therapeutic agents in hospitalized adults diagnosed with COVID-19. The study will compare different investigational therapeutic agents to a control arm. ID: NCT04280705
Sponsor: National Institute of Allergy and Infectious Diseases
Contact: Central Contact ([email protected])
Locations: VA Palo Alto Health Care System, California; Naval Medical Center San Diego, California; Southeast Louisiana Veterans Health Care System, New Orleans; Walter Reed National Military Medical Center, Bethesda, Maryland; National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Bethesda, Maryland; Brooke Army Medical Center, Fort Sam Houston, Texas; Madigan Army Medical Center, Tacoma, Washington
Study to Evaluate the Safety and Antiviral Activity of Remdesivir (GS-5734) in Participants With Severe Coronavirus Disease (COVID-19)
The primary objective of this study is to evaluate the efficacy of 2 remdesivir (RDV) regimens with respect to clinical status assessed by a 7-point ordinal scale on Day 11 (NCT04292730) or Day 14 (NCT04292899).
ID: NCT04292730/NCT04292899
Sponsor: Gilead Sciences
Contact: Gilead Clinical Study Information Center (833-445-3230)
Location: James J. Peters VA Medical Center, Bronx, New York
Expanded Access Remdesivir (RDV; GS-5734)
The treatment of communicable Novel Coronavirus of 2019 with Remdesivir (RDV; GS-5734) also known as severe acute respiratory syndrome coronavirus 2.
ID: NCT04302766
Sponsor: US Army Medical Research and Development Command
Contact: Sandi Parriott ([email protected])
A Study to Evaluate the Safety and Efficacy of Tocilizumab in Patients With Severe COVID-19 Pneumonia (COVACTA)
This study will evaluate the efficacy, safety, pharmacodynamics, and pharmacokinetics of tocilizumab (TCZ) compared with a matching placebo in combination with standard of care (SOC) in hospitalized patients with severe COVID-19 pneumonia.
ID: NCT04320615
Sponsor: Hoffmann-La Roche
Location: James J Peters VA Medical Center, Bronx, New York
Administration of Intravenous Vitamin C in Novel Coronavirus Infection (COVID-19) and Decreased Oxygenation (AVoCaDO)
Previous research has shown that high dose intravenous vitamin C (HDIVC) may benefit patients with sepsis, acute lung injury (ALI), and the acute respiratory distress syndrome (ARDS). However, it is not known if early administration of HDIVC could prevent progression to ARDS. We hypothesize that HDIVC is safe and tolerable in COVID-19 subjects given early or late in the disease course and may reduce the risk of respiratory failure requiring mechanical ventilation and development of ARDS along with reductions in supplemental oxygen demand and inflammatory markers.
ID: NCT04357782
Sponsor: Hunter Holmes Mcguire VA Medical CenterContact: Brian Davis ([email protected])
Location: Hunter Holmes Mcguire VA Medical Center, Richmond, Virginia
Treatment Of CORONAVIRUS DISEASE 2019 (COVID-19) With Anti-Sars-CoV-2 Convalescent Plasma (ASCoV2CP)
This is an expanded access open-label, single-arm, multi-site protocol to provide convalescent plasma as a treatment for patients diagnosed with severe, or life-threatening COVID-19.
ID: NCT04360486
Sponsor: US Army Medical Research and Development Command
Contact: Andrew Cap ([email protected])
VA Remote and Equitable Access to COVID-19 Healthcare Delivery (VA-REACH TRIAL) (VA-REACH)
We propose a 3-arm randomized control trial to determine the efficacy of hydroxychloroquine or azithromycin in treating mild to moderate COVID-19 among veterans in the outpatient setting.
ID: NCT04363203
Sponsor: Salomeh Keyhani
Location: San Francisco VA Health Care System, California
A Study to Evaluate the Safety and Efficacy of MSTT1041A (Astegolimab) or UTTR1147A in Patients With Severe COVID-19 Pneumonia (COVASTIL)
This is a Phase II, randomized, double-blind, placebo-controlled, multicenter study to assess the efficacy and safety of MSTT1041A (astegolimab) or UTTR1147A in combination with standard of care (SOC) compared with matching placebo in combination with SOC in patients hospitalized with severe coronavirus disease 2019 (COVID-19) pneumonia.
ID: NCT04386616
Sponsor: Genentech
Contact: Study ID Number: GA42469 ([email protected])
Location: Southeast Louisiana Veterans Health Care System, New Orleans
Hormonal Intervention for the Treatment in Veterans With COVID-19 Requiring Hospitalization (HITCH)
The purpose of this study is to determine if temporary androgen suppression improves the clinical outcomes of veterans who are hospitalized to an acute care ward due to COVID-19.ID: NCT04397718
Sponsor: VA Office of Research and Development
Contact: Matthew B Rettig ([email protected]), Nicholas Nickols ([email protected])
Locations: VA Greater Los Angeles Healthcare System, California; VA NY Harbor Healthcare System, New York; VA Puget Sound Health Care System, Seattle, Washington
Adaptive COVID-19 Treatment Trial 2 (ACTT-II)
ACTT-II will evaluate the combination of baricitinib and remdesivir compared to remdesivir alone. Subjects will be assessed daily while hospitalized. If the subjects are discharged from the hospital, they will have a study visit at Days 15, 22, and 29.
ID: NCT04401579
Sponsor: National Institute of Allergy and Infectious Diseases (NIAID)
Contact: Central Contact ([email protected])
Locations: VA Palo Alto Health Care System, California; Naval Medical Center San Diego, California; Rocky Mountain Regional Veteran Affairs Medical Center, Aurora, Colorado; Southeast Louisiana Veterans Health Care System, New Orleans; Walter Reed National Military Medical Center, Bethesda, Maryland; National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Bethesda, Maryland; Brooke Army Medical Center, Fort Sam Houston, Texas; Madigan Army Medical Center, Tacoma, Washington
Finding effective treatment or a vaccine for COVID-19, the disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has placed significant strains on the global health care system. The National Library of Medicine database lists > 1,800 trials that are aimed at addressing COVID-19-related health care. Already, trials developed by the US Department of Veterans Affairs (VA), US Department of Defense (DoD), and the National Institute of Allergy and Infectious Diseases have provided important data on effective treatment options. The clinical trials listed below are all open as of May 31, 2020 and have trial sites at VA and DoD facilities. For additional information and full inclusion/exclusion criteria, please consult clinicaltrials.gov.
Adaptive COVID-19 Treatment Trial (ACTT)
This study is an adaptive, randomized, double-blind, placebo-controlled trial to evaluate the safety and efficacy of novel therapeutic agents in hospitalized adults diagnosed with COVID-19. The study will compare different investigational therapeutic agents to a control arm. ID: NCT04280705
Sponsor: National Institute of Allergy and Infectious Diseases
Contact: Central Contact ([email protected])
Locations: VA Palo Alto Health Care System, California; Naval Medical Center San Diego, California; Southeast Louisiana Veterans Health Care System, New Orleans; Walter Reed National Military Medical Center, Bethesda, Maryland; National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Bethesda, Maryland; Brooke Army Medical Center, Fort Sam Houston, Texas; Madigan Army Medical Center, Tacoma, Washington
Study to Evaluate the Safety and Antiviral Activity of Remdesivir (GS-5734) in Participants With Severe Coronavirus Disease (COVID-19)
The primary objective of this study is to evaluate the efficacy of 2 remdesivir (RDV) regimens with respect to clinical status assessed by a 7-point ordinal scale on Day 11 (NCT04292730) or Day 14 (NCT04292899).
ID: NCT04292730/NCT04292899
Sponsor: Gilead Sciences
Contact: Gilead Clinical Study Information Center (833-445-3230)
Location: James J. Peters VA Medical Center, Bronx, New York
Expanded Access Remdesivir (RDV; GS-5734)
The treatment of communicable Novel Coronavirus of 2019 with Remdesivir (RDV; GS-5734) also known as severe acute respiratory syndrome coronavirus 2.
ID: NCT04302766
Sponsor: US Army Medical Research and Development Command
Contact: Sandi Parriott ([email protected])
A Study to Evaluate the Safety and Efficacy of Tocilizumab in Patients With Severe COVID-19 Pneumonia (COVACTA)
This study will evaluate the efficacy, safety, pharmacodynamics, and pharmacokinetics of tocilizumab (TCZ) compared with a matching placebo in combination with standard of care (SOC) in hospitalized patients with severe COVID-19 pneumonia.
ID: NCT04320615
Sponsor: Hoffmann-La Roche
Location: James J Peters VA Medical Center, Bronx, New York
Administration of Intravenous Vitamin C in Novel Coronavirus Infection (COVID-19) and Decreased Oxygenation (AVoCaDO)
Previous research has shown that high dose intravenous vitamin C (HDIVC) may benefit patients with sepsis, acute lung injury (ALI), and the acute respiratory distress syndrome (ARDS). However, it is not known if early administration of HDIVC could prevent progression to ARDS. We hypothesize that HDIVC is safe and tolerable in COVID-19 subjects given early or late in the disease course and may reduce the risk of respiratory failure requiring mechanical ventilation and development of ARDS along with reductions in supplemental oxygen demand and inflammatory markers.
ID: NCT04357782
Sponsor: Hunter Holmes Mcguire VA Medical CenterContact: Brian Davis ([email protected])
Location: Hunter Holmes Mcguire VA Medical Center, Richmond, Virginia
Treatment Of CORONAVIRUS DISEASE 2019 (COVID-19) With Anti-Sars-CoV-2 Convalescent Plasma (ASCoV2CP)
This is an expanded access open-label, single-arm, multi-site protocol to provide convalescent plasma as a treatment for patients diagnosed with severe, or life-threatening COVID-19.
ID: NCT04360486
Sponsor: US Army Medical Research and Development Command
Contact: Andrew Cap ([email protected])
VA Remote and Equitable Access to COVID-19 Healthcare Delivery (VA-REACH TRIAL) (VA-REACH)
We propose a 3-arm randomized control trial to determine the efficacy of hydroxychloroquine or azithromycin in treating mild to moderate COVID-19 among veterans in the outpatient setting.
ID: NCT04363203
Sponsor: Salomeh Keyhani
Location: San Francisco VA Health Care System, California
A Study to Evaluate the Safety and Efficacy of MSTT1041A (Astegolimab) or UTTR1147A in Patients With Severe COVID-19 Pneumonia (COVASTIL)
This is a Phase II, randomized, double-blind, placebo-controlled, multicenter study to assess the efficacy and safety of MSTT1041A (astegolimab) or UTTR1147A in combination with standard of care (SOC) compared with matching placebo in combination with SOC in patients hospitalized with severe coronavirus disease 2019 (COVID-19) pneumonia.
ID: NCT04386616
Sponsor: Genentech
Contact: Study ID Number: GA42469 ([email protected])
Location: Southeast Louisiana Veterans Health Care System, New Orleans
Hormonal Intervention for the Treatment in Veterans With COVID-19 Requiring Hospitalization (HITCH)
The purpose of this study is to determine if temporary androgen suppression improves the clinical outcomes of veterans who are hospitalized to an acute care ward due to COVID-19.ID: NCT04397718
Sponsor: VA Office of Research and Development
Contact: Matthew B Rettig ([email protected]), Nicholas Nickols ([email protected])
Locations: VA Greater Los Angeles Healthcare System, California; VA NY Harbor Healthcare System, New York; VA Puget Sound Health Care System, Seattle, Washington
Adaptive COVID-19 Treatment Trial 2 (ACTT-II)
ACTT-II will evaluate the combination of baricitinib and remdesivir compared to remdesivir alone. Subjects will be assessed daily while hospitalized. If the subjects are discharged from the hospital, they will have a study visit at Days 15, 22, and 29.
ID: NCT04401579
Sponsor: National Institute of Allergy and Infectious Diseases (NIAID)
Contact: Central Contact ([email protected])
Locations: VA Palo Alto Health Care System, California; Naval Medical Center San Diego, California; Rocky Mountain Regional Veteran Affairs Medical Center, Aurora, Colorado; Southeast Louisiana Veterans Health Care System, New Orleans; Walter Reed National Military Medical Center, Bethesda, Maryland; National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Bethesda, Maryland; Brooke Army Medical Center, Fort Sam Houston, Texas; Madigan Army Medical Center, Tacoma, Washington
Finding effective treatment or a vaccine for COVID-19, the disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has placed significant strains on the global health care system. The National Library of Medicine database lists > 1,800 trials that are aimed at addressing COVID-19-related health care. Already, trials developed by the US Department of Veterans Affairs (VA), US Department of Defense (DoD), and the National Institute of Allergy and Infectious Diseases have provided important data on effective treatment options. The clinical trials listed below are all open as of May 31, 2020 and have trial sites at VA and DoD facilities. For additional information and full inclusion/exclusion criteria, please consult clinicaltrials.gov.
Adaptive COVID-19 Treatment Trial (ACTT)
This study is an adaptive, randomized, double-blind, placebo-controlled trial to evaluate the safety and efficacy of novel therapeutic agents in hospitalized adults diagnosed with COVID-19. The study will compare different investigational therapeutic agents to a control arm. ID: NCT04280705
Sponsor: National Institute of Allergy and Infectious Diseases
Contact: Central Contact ([email protected])
Locations: VA Palo Alto Health Care System, California; Naval Medical Center San Diego, California; Southeast Louisiana Veterans Health Care System, New Orleans; Walter Reed National Military Medical Center, Bethesda, Maryland; National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Bethesda, Maryland; Brooke Army Medical Center, Fort Sam Houston, Texas; Madigan Army Medical Center, Tacoma, Washington
Study to Evaluate the Safety and Antiviral Activity of Remdesivir (GS-5734) in Participants With Severe Coronavirus Disease (COVID-19)
The primary objective of this study is to evaluate the efficacy of 2 remdesivir (RDV) regimens with respect to clinical status assessed by a 7-point ordinal scale on Day 11 (NCT04292730) or Day 14 (NCT04292899).
ID: NCT04292730/NCT04292899
Sponsor: Gilead Sciences
Contact: Gilead Clinical Study Information Center (833-445-3230)
Location: James J. Peters VA Medical Center, Bronx, New York
Expanded Access Remdesivir (RDV; GS-5734)
The treatment of communicable Novel Coronavirus of 2019 with Remdesivir (RDV; GS-5734) also known as severe acute respiratory syndrome coronavirus 2.
ID: NCT04302766
Sponsor: US Army Medical Research and Development Command
Contact: Sandi Parriott ([email protected])
A Study to Evaluate the Safety and Efficacy of Tocilizumab in Patients With Severe COVID-19 Pneumonia (COVACTA)
This study will evaluate the efficacy, safety, pharmacodynamics, and pharmacokinetics of tocilizumab (TCZ) compared with a matching placebo in combination with standard of care (SOC) in hospitalized patients with severe COVID-19 pneumonia.
ID: NCT04320615
Sponsor: Hoffmann-La Roche
Location: James J Peters VA Medical Center, Bronx, New York
Administration of Intravenous Vitamin C in Novel Coronavirus Infection (COVID-19) and Decreased Oxygenation (AVoCaDO)
Previous research has shown that high dose intravenous vitamin C (HDIVC) may benefit patients with sepsis, acute lung injury (ALI), and the acute respiratory distress syndrome (ARDS). However, it is not known if early administration of HDIVC could prevent progression to ARDS. We hypothesize that HDIVC is safe and tolerable in COVID-19 subjects given early or late in the disease course and may reduce the risk of respiratory failure requiring mechanical ventilation and development of ARDS along with reductions in supplemental oxygen demand and inflammatory markers.
ID: NCT04357782
Sponsor: Hunter Holmes Mcguire VA Medical CenterContact: Brian Davis ([email protected])
Location: Hunter Holmes Mcguire VA Medical Center, Richmond, Virginia
Treatment Of CORONAVIRUS DISEASE 2019 (COVID-19) With Anti-Sars-CoV-2 Convalescent Plasma (ASCoV2CP)
This is an expanded access open-label, single-arm, multi-site protocol to provide convalescent plasma as a treatment for patients diagnosed with severe, or life-threatening COVID-19.
ID: NCT04360486
Sponsor: US Army Medical Research and Development Command
Contact: Andrew Cap ([email protected])
VA Remote and Equitable Access to COVID-19 Healthcare Delivery (VA-REACH TRIAL) (VA-REACH)
We propose a 3-arm randomized control trial to determine the efficacy of hydroxychloroquine or azithromycin in treating mild to moderate COVID-19 among veterans in the outpatient setting.
ID: NCT04363203
Sponsor: Salomeh Keyhani
Location: San Francisco VA Health Care System, California
A Study to Evaluate the Safety and Efficacy of MSTT1041A (Astegolimab) or UTTR1147A in Patients With Severe COVID-19 Pneumonia (COVASTIL)
This is a Phase II, randomized, double-blind, placebo-controlled, multicenter study to assess the efficacy and safety of MSTT1041A (astegolimab) or UTTR1147A in combination with standard of care (SOC) compared with matching placebo in combination with SOC in patients hospitalized with severe coronavirus disease 2019 (COVID-19) pneumonia.
ID: NCT04386616
Sponsor: Genentech
Contact: Study ID Number: GA42469 ([email protected])
Location: Southeast Louisiana Veterans Health Care System, New Orleans
Hormonal Intervention for the Treatment in Veterans With COVID-19 Requiring Hospitalization (HITCH)
The purpose of this study is to determine if temporary androgen suppression improves the clinical outcomes of veterans who are hospitalized to an acute care ward due to COVID-19.ID: NCT04397718
Sponsor: VA Office of Research and Development
Contact: Matthew B Rettig ([email protected]), Nicholas Nickols ([email protected])
Locations: VA Greater Los Angeles Healthcare System, California; VA NY Harbor Healthcare System, New York; VA Puget Sound Health Care System, Seattle, Washington
Adaptive COVID-19 Treatment Trial 2 (ACTT-II)
ACTT-II will evaluate the combination of baricitinib and remdesivir compared to remdesivir alone. Subjects will be assessed daily while hospitalized. If the subjects are discharged from the hospital, they will have a study visit at Days 15, 22, and 29.
ID: NCT04401579
Sponsor: National Institute of Allergy and Infectious Diseases (NIAID)
Contact: Central Contact ([email protected])
Locations: VA Palo Alto Health Care System, California; Naval Medical Center San Diego, California; Rocky Mountain Regional Veteran Affairs Medical Center, Aurora, Colorado; Southeast Louisiana Veterans Health Care System, New Orleans; Walter Reed National Military Medical Center, Bethesda, Maryland; National Institutes of Health - Clinical Center, National Institute of Allergy and Infectious Diseases Laboratory Of Immunoregulation, Bethesda, Maryland; Brooke Army Medical Center, Fort Sam Houston, Texas; Madigan Army Medical Center, Tacoma, Washington
Cardiology societies unite to denounce racist violence
The death of George Floyd and other African Americans spurred the Association of Black Cardiologists, the American Heart Association, and the American College of Cardiology to join forces and issue an urgent letter denouncing recent and ongoing events.
Starting off by acknowledging that these are “difficult and disturbing times,” the presidents of the three societies tied the violence into the bigger public health picture. “Like cardiovascular disease, acts of violence and racism are core causes of psychosocial stress that promote poor well-being and cardiovascular health, especially for communities of color.”
“It’s not just one quick solution, one quick letter. It’s more of an ongoing project to raise awareness and have really defined projects. We want to have goals, tactics, and measurable outcomes. We want to make sure it’s not just a banner on the wall,” Athena Poppas, MD, president of the American College of Cardiology and one of three physicians signing the letter, said in an interview.
The Association of Black Cardiologists drafted the statement and asked the AHA and ACC if they wanted to sign on. “It felt important to join them and follow their lead,” she said. “There is a clear link between psychosocial stress and discrimination and health equity in the communities.”
Interestingly, the ABC and ACC have an existing partnership, one that included creating a “Campaign for the Future” a little more than a year ago. One of the focuses is on reducing health disparities and starting a diversity and inclusion task force that later became a committee. The groups held a joint board of trustees meeting at Morehouse University, Atlanta, in January 2020. Thinking about that time, Dr. Poppas added, “who knew what was about to transpire over the next few months?”
The letter is only one component of an ongoing effort to “find concrete ways to make change, both within the college and within our profession,” added Dr. Poppas, chief of cardiology and professor of medicine at Brown University, Providence, R.I., and director of the Lifespan Cardiovascular Institute of Rhode Island, Miriam Hospitals, and Newport Hospitals. “Thereby, there is good data that you affect health equity in the population as well.”
“We DENOUNCE incidents of racism and violence that continue to ravage our communities,” the society leaders wrote in the letter. “Given that heart disease and stroke are the leading causes of death for communities of color, particularly African Americans who have the lowest life expectancy of all racial/ethnic groups living in the United States, we are extremely disturbed by violent acts that cut to the core of the lives of our community.”
Other societies released similar statements. For example, the American College of Physicians expressed “grave concern” about recent events and the American Medical Association released a statement entitled “Police brutality must stop.”
A cardiologist speaks out
“Thank you to my organizations, the Association of Black Cardiologists and the American College of Cardiology, for taking a stand,” Travis C. Batts, MD, said in a video statement posted to YouTube on June 2, 2020.
“As an African American male who has sons, brothers, and friends who are also African American, I oftentimes have angst, particularly with my sons. Despite what I do to create an environment that cultivates education and puts them in the right position, there are some people who would stop just at how they look when they approach them,” Dr. Batts said.
“I always have that fear as a father that at some point they may engage with law enforcement – and it may not turn out the way we want it to,” said Dr. Batts, chairman of medical sub-specialties and medical director of the cardiology clinic at Wilford Hall Ambulatory Surgical Center at Lackland Air Force Base, Tex. He also is an associate professor of cardiovascular medicine for the Uniformed Services University of the Health Sciences, Bethesda, Md., and is an adjunct assistant professor at Texas A&M University. He went on in the video to describe how a personal encounter with police years ago changed his life.
The urgent letter from the cardiology societies speaks to health care disparities, Dr. Batts said, “but it doesn’t stop there. It talks about their goals to balance these issues that we see as a pervasive problem in our community.”
The societies point out that George Floyd’s death is not an isolated incident. “Mr. Floyd’s death comes on the heels of other recent incidents caught on camera. In another 2020 incident, Ahmaud Arbery was shot and killed while jogging in his hometown of Brunswick, Ga. Christian Cooper is fortunately alive and well to speak to the Memorial Day incident in New York’s Central Park where he was accused of threatening the life of a woman while bird watching.” They added that “another senseless death involves officers entering the Louisville, Kent., home of emergency medical technician Breonna Taylor.”
Dr. Batts said this portion of the statement was particularly poignant: “We stand and link arms in solidarity with efforts to dismantle systems that maintain excess morbidity and mortality, especially among vulnerable populations and those historically oppressed. Indeed, our collective vast membership, many of whom are at the front lines of clinical health care, has taken an oath to decisively and with kindness, compassion and grace act to relieve suffering related to ‘I can’t breathe’ in order to preserve life.”
A Positive Response
The response to the urgent letter has been “overwhelmingly positive,” Dr. Poppas said. “This isn’t political, per se. This is really about justice, about health equity, and about being moral and conscious human beings. People I hadn’t heard from in years said, ‘thank you for doing this.’ ” The comments on social media were “almost uniformly positive,” she added. “There is always one or two people who feel this isn’t what cardiology is about.”
“Although making a statement is important, so is doing the hard work to make change,” Dr. Poppas said. The goal involves “rolling up our sleeves and spending the time, the money and the energy to make changes – so 5-10 years from now, it looks different.”
In addition to Dr. Poppas, Michelle A. Albert, MD, MPH, president of the Association of Black Cardiologists and Robert A. Harrington, MD, president of the American Heart Association, signed the letter. Dr. Pappas and Dr. Batts had no relevant disclosures.
The death of George Floyd and other African Americans spurred the Association of Black Cardiologists, the American Heart Association, and the American College of Cardiology to join forces and issue an urgent letter denouncing recent and ongoing events.
Starting off by acknowledging that these are “difficult and disturbing times,” the presidents of the three societies tied the violence into the bigger public health picture. “Like cardiovascular disease, acts of violence and racism are core causes of psychosocial stress that promote poor well-being and cardiovascular health, especially for communities of color.”
“It’s not just one quick solution, one quick letter. It’s more of an ongoing project to raise awareness and have really defined projects. We want to have goals, tactics, and measurable outcomes. We want to make sure it’s not just a banner on the wall,” Athena Poppas, MD, president of the American College of Cardiology and one of three physicians signing the letter, said in an interview.
The Association of Black Cardiologists drafted the statement and asked the AHA and ACC if they wanted to sign on. “It felt important to join them and follow their lead,” she said. “There is a clear link between psychosocial stress and discrimination and health equity in the communities.”
Interestingly, the ABC and ACC have an existing partnership, one that included creating a “Campaign for the Future” a little more than a year ago. One of the focuses is on reducing health disparities and starting a diversity and inclusion task force that later became a committee. The groups held a joint board of trustees meeting at Morehouse University, Atlanta, in January 2020. Thinking about that time, Dr. Poppas added, “who knew what was about to transpire over the next few months?”
The letter is only one component of an ongoing effort to “find concrete ways to make change, both within the college and within our profession,” added Dr. Poppas, chief of cardiology and professor of medicine at Brown University, Providence, R.I., and director of the Lifespan Cardiovascular Institute of Rhode Island, Miriam Hospitals, and Newport Hospitals. “Thereby, there is good data that you affect health equity in the population as well.”
“We DENOUNCE incidents of racism and violence that continue to ravage our communities,” the society leaders wrote in the letter. “Given that heart disease and stroke are the leading causes of death for communities of color, particularly African Americans who have the lowest life expectancy of all racial/ethnic groups living in the United States, we are extremely disturbed by violent acts that cut to the core of the lives of our community.”
Other societies released similar statements. For example, the American College of Physicians expressed “grave concern” about recent events and the American Medical Association released a statement entitled “Police brutality must stop.”
A cardiologist speaks out
“Thank you to my organizations, the Association of Black Cardiologists and the American College of Cardiology, for taking a stand,” Travis C. Batts, MD, said in a video statement posted to YouTube on June 2, 2020.
“As an African American male who has sons, brothers, and friends who are also African American, I oftentimes have angst, particularly with my sons. Despite what I do to create an environment that cultivates education and puts them in the right position, there are some people who would stop just at how they look when they approach them,” Dr. Batts said.
“I always have that fear as a father that at some point they may engage with law enforcement – and it may not turn out the way we want it to,” said Dr. Batts, chairman of medical sub-specialties and medical director of the cardiology clinic at Wilford Hall Ambulatory Surgical Center at Lackland Air Force Base, Tex. He also is an associate professor of cardiovascular medicine for the Uniformed Services University of the Health Sciences, Bethesda, Md., and is an adjunct assistant professor at Texas A&M University. He went on in the video to describe how a personal encounter with police years ago changed his life.
The urgent letter from the cardiology societies speaks to health care disparities, Dr. Batts said, “but it doesn’t stop there. It talks about their goals to balance these issues that we see as a pervasive problem in our community.”
The societies point out that George Floyd’s death is not an isolated incident. “Mr. Floyd’s death comes on the heels of other recent incidents caught on camera. In another 2020 incident, Ahmaud Arbery was shot and killed while jogging in his hometown of Brunswick, Ga. Christian Cooper is fortunately alive and well to speak to the Memorial Day incident in New York’s Central Park where he was accused of threatening the life of a woman while bird watching.” They added that “another senseless death involves officers entering the Louisville, Kent., home of emergency medical technician Breonna Taylor.”
Dr. Batts said this portion of the statement was particularly poignant: “We stand and link arms in solidarity with efforts to dismantle systems that maintain excess morbidity and mortality, especially among vulnerable populations and those historically oppressed. Indeed, our collective vast membership, many of whom are at the front lines of clinical health care, has taken an oath to decisively and with kindness, compassion and grace act to relieve suffering related to ‘I can’t breathe’ in order to preserve life.”
A Positive Response
The response to the urgent letter has been “overwhelmingly positive,” Dr. Poppas said. “This isn’t political, per se. This is really about justice, about health equity, and about being moral and conscious human beings. People I hadn’t heard from in years said, ‘thank you for doing this.’ ” The comments on social media were “almost uniformly positive,” she added. “There is always one or two people who feel this isn’t what cardiology is about.”
“Although making a statement is important, so is doing the hard work to make change,” Dr. Poppas said. The goal involves “rolling up our sleeves and spending the time, the money and the energy to make changes – so 5-10 years from now, it looks different.”
In addition to Dr. Poppas, Michelle A. Albert, MD, MPH, president of the Association of Black Cardiologists and Robert A. Harrington, MD, president of the American Heart Association, signed the letter. Dr. Pappas and Dr. Batts had no relevant disclosures.
The death of George Floyd and other African Americans spurred the Association of Black Cardiologists, the American Heart Association, and the American College of Cardiology to join forces and issue an urgent letter denouncing recent and ongoing events.
Starting off by acknowledging that these are “difficult and disturbing times,” the presidents of the three societies tied the violence into the bigger public health picture. “Like cardiovascular disease, acts of violence and racism are core causes of psychosocial stress that promote poor well-being and cardiovascular health, especially for communities of color.”
“It’s not just one quick solution, one quick letter. It’s more of an ongoing project to raise awareness and have really defined projects. We want to have goals, tactics, and measurable outcomes. We want to make sure it’s not just a banner on the wall,” Athena Poppas, MD, president of the American College of Cardiology and one of three physicians signing the letter, said in an interview.
The Association of Black Cardiologists drafted the statement and asked the AHA and ACC if they wanted to sign on. “It felt important to join them and follow their lead,” she said. “There is a clear link between psychosocial stress and discrimination and health equity in the communities.”
Interestingly, the ABC and ACC have an existing partnership, one that included creating a “Campaign for the Future” a little more than a year ago. One of the focuses is on reducing health disparities and starting a diversity and inclusion task force that later became a committee. The groups held a joint board of trustees meeting at Morehouse University, Atlanta, in January 2020. Thinking about that time, Dr. Poppas added, “who knew what was about to transpire over the next few months?”
The letter is only one component of an ongoing effort to “find concrete ways to make change, both within the college and within our profession,” added Dr. Poppas, chief of cardiology and professor of medicine at Brown University, Providence, R.I., and director of the Lifespan Cardiovascular Institute of Rhode Island, Miriam Hospitals, and Newport Hospitals. “Thereby, there is good data that you affect health equity in the population as well.”
“We DENOUNCE incidents of racism and violence that continue to ravage our communities,” the society leaders wrote in the letter. “Given that heart disease and stroke are the leading causes of death for communities of color, particularly African Americans who have the lowest life expectancy of all racial/ethnic groups living in the United States, we are extremely disturbed by violent acts that cut to the core of the lives of our community.”
Other societies released similar statements. For example, the American College of Physicians expressed “grave concern” about recent events and the American Medical Association released a statement entitled “Police brutality must stop.”
A cardiologist speaks out
“Thank you to my organizations, the Association of Black Cardiologists and the American College of Cardiology, for taking a stand,” Travis C. Batts, MD, said in a video statement posted to YouTube on June 2, 2020.
“As an African American male who has sons, brothers, and friends who are also African American, I oftentimes have angst, particularly with my sons. Despite what I do to create an environment that cultivates education and puts them in the right position, there are some people who would stop just at how they look when they approach them,” Dr. Batts said.
“I always have that fear as a father that at some point they may engage with law enforcement – and it may not turn out the way we want it to,” said Dr. Batts, chairman of medical sub-specialties and medical director of the cardiology clinic at Wilford Hall Ambulatory Surgical Center at Lackland Air Force Base, Tex. He also is an associate professor of cardiovascular medicine for the Uniformed Services University of the Health Sciences, Bethesda, Md., and is an adjunct assistant professor at Texas A&M University. He went on in the video to describe how a personal encounter with police years ago changed his life.
The urgent letter from the cardiology societies speaks to health care disparities, Dr. Batts said, “but it doesn’t stop there. It talks about their goals to balance these issues that we see as a pervasive problem in our community.”
The societies point out that George Floyd’s death is not an isolated incident. “Mr. Floyd’s death comes on the heels of other recent incidents caught on camera. In another 2020 incident, Ahmaud Arbery was shot and killed while jogging in his hometown of Brunswick, Ga. Christian Cooper is fortunately alive and well to speak to the Memorial Day incident in New York’s Central Park where he was accused of threatening the life of a woman while bird watching.” They added that “another senseless death involves officers entering the Louisville, Kent., home of emergency medical technician Breonna Taylor.”
Dr. Batts said this portion of the statement was particularly poignant: “We stand and link arms in solidarity with efforts to dismantle systems that maintain excess morbidity and mortality, especially among vulnerable populations and those historically oppressed. Indeed, our collective vast membership, many of whom are at the front lines of clinical health care, has taken an oath to decisively and with kindness, compassion and grace act to relieve suffering related to ‘I can’t breathe’ in order to preserve life.”
A Positive Response
The response to the urgent letter has been “overwhelmingly positive,” Dr. Poppas said. “This isn’t political, per se. This is really about justice, about health equity, and about being moral and conscious human beings. People I hadn’t heard from in years said, ‘thank you for doing this.’ ” The comments on social media were “almost uniformly positive,” she added. “There is always one or two people who feel this isn’t what cardiology is about.”
“Although making a statement is important, so is doing the hard work to make change,” Dr. Poppas said. The goal involves “rolling up our sleeves and spending the time, the money and the energy to make changes – so 5-10 years from now, it looks different.”
In addition to Dr. Poppas, Michelle A. Albert, MD, MPH, president of the Association of Black Cardiologists and Robert A. Harrington, MD, president of the American Heart Association, signed the letter. Dr. Pappas and Dr. Batts had no relevant disclosures.
Microthrombotic Complications of COVID-19 Are Likely Due to Embolism of Circulating Endothelial Derived Ultralarge von Willebrand Factor (eULVWF) Decorated-Platelet Strings
To the Editor: COVID-19 is a pandemic caused by the virus SARS-CoV-2. Serious complications of COVID-19 are characterized by acute respiratory distress syndrome (ARDS), pneumonia and rapidly progressing to multiorgan dysfunction syndrome (MODS).
The pathophysiology of COVID-19 is not fully understood yet and neither vaccine nor clearly effective antiviral treatment is available at this time. Based on the endothelial pathogenesis of viral sepsis, which includes ARDS as seen in severe acute respiratory syndrome (SARS) due to SARS-CoV and Middle East respiratory syndrome due to MERS-CoV,1,2 we believe COVID-19-associated ARDS is also caused by endotheliopathy-associated vascular microthrombotic disease (EA-VMTD), which also involves multiorgan dysfunction syndrome (MODS) that has been reported as the cause of death.3 We suspect these complications are secondary to disequilibrium state (for various reasons4,5) between insufficient ADAMTS13 and excessive exocytosis of ultra large von Willebrand factor multimers (ULVWF) from Weibel-Palade bodies present in endothelial cells due to COVID-19-induced endotheliopathy.
Endothelial-derived ULVWF multimers anchored to the endothelial surface of the vascular wall recruit platelets and initiate microthrombogenesis within the microvasculature, leading to large microthrombi strings composed of platelet and eULVWF complexes like “beads-on-a-string structures”6 where platelets firmly adhere to eULVWF, instead of roll on eULVWF strings.4 Platelets, once adhered to eULVWF strings, are rapidly activated causing platelet aggregation and also recruit leukocytes in a P-selectin dependent manner.4 These aggregates grow until they become sufficiently large and can no longer be held onto the eULVWF strings against the force of blood flow and released from endothelial cells into the circulation.4 It appears to us that in COVID-19 microthrombotic disease, large amounts of circulating complexes of endothelial-derived ULVWF decorated-platelet microthrombi strings are filtered in the microvasculature (embolism) or develops in the microvasculature in situ causing microthrombotic occlusion. During our data search, we have come across several articles published by Chang, including on endotheliopathy causing vascular microthrombotic disease based on a novel concept of “TTP-like syndrome”7
The genesis of EA-VMTD in TTP like syndrome is suspected to be triggered by complement activation and terminal complement complex (C5b-9, membrane attack complex, MAC) may play a key role in producing endotheliopathy.7 Magro and colleagues reported that COVID-19 patients have demonstrated generalized thrombotic microvascular injury involving the lungs and skin showing intense complement activation and C5b-9 deposition in the tissue.8 Also, recent pathology reports of COVID-19 diseased lungs showed extensive platelet-rich clotting with adherent mononuclear cells and extensive fibrin clotting,9 which appear consistent with involvement of NETosis.10 In another case report from Switzerland, a patient with severe COVID-19 had massive elevation of VWF antigen and activity (555% and 520%, respectively) and increased Factor VIII clotting activity (369%).11 These findings support vascular endotheliopathy causing exocytosis of ULVWF and associated increase in Factor VIII causing microthrombotic disease/embolism.
COVID-19 clinical syndrome appears very much consistent with EA-VMTD presenting with ARDS and MODS as well as micro-macro-thrombotic complications, including peripheral ischemia/gangrene involving fingers and toes and skin necrosis.8,12
We believe that an appropriate therapy may not be anticoagulation but should include antimicrothrombotic therapy targeting endotheliopathy and primary hemostasis in the early stages of the disease (platelet adhesion, activation, and aggregation; especially eULVWF) like recombinant CD59 (membrane attack complex inhibition factor [MACIF]), recombinant ADAMTS13, glycoprotein IIb/IIIa receptor blocker, therapeutic plasma exchange, and perhaps anticomplement therapy (in selected cases) and others; these need to be validated in clinical trials prior to clinical application.
Of note, ADAMTS13 is a zinc containing protease. We noted that zinc and calcium concentrations play a significant role (in vitro) in ADAMTS13 activity in citrated plasma and recombinant ADAMTS13 activity with no added chelators (recombinant ADAMTS13 activity can enhance up to 200-fold); whereas in high zinc concentrations, ADAMTS13 gets deactivated.13 We suggest this finding merits an urgent clinical trial since it appears to us as the best possible cost-effective treatment for COVID-19 microthrombotic complications.
In this view of clinical pathophysiology of sepsis in COVID-19, we would like to enlighten the relationship between endothelial pathogenesis of coronaviral sepsis and vascular microthrombotic disease and would urge the medical community to immediately explore appropriate therapeutic options.
N. Varatharajah, MD
Suganthi Rajah, MD
Virginia, US
1. Chang JC. Sepsis and septic shock: endothelial molecular pathogenesis associated with vascular microthrombotic disease. Thromb J. 2019;17:10. Published 2019 May 30. doi:10.1186/s12959-019-0198-4
2. Chang JC. Acute respiratory distress syndrome as an organ phenotype of vascular microthrombotic disease: based on hemostatic theory and endothelial molecular pathogenesis. Clin Appl Thromb Hemost. 2019;25:1076029619887437. doi:10.1177/1076029619887437
3. Zaim S, Chong JH, Sankaranarayanan V, Harky A. COVID-19 and multi-organ response. Curr Probl Cardiol. 2020;100618. In press. doi:10.1016/j.cpcardiol.2020.100618
4. Bernardo A, Ball C, Nolasco L, Choi H, Moake JL, Dong JF. Platelets adhered to endothelial cell-bound ultra-large von Willebrand factor strings support leukocyte tethering and rolling under high shear stress. J Thromb Haemost. 2005;3(3):562‐570. doi:10.1111/j.1538-7836.2005.01122.x https://doi.org/10.1111/j.1538-7836.2005.01122.x
5. Mannucci PM, Canciani MT, Forza I, Lussana F, Lattuada A, Rossi E. Changes in health and disease of the metalloprotease that cleaves von Willebrand factor. Blood. 2001;98(9):2730‐2735. doi:10.1182/blood.v98.9.2730
6. Dong JF, Moake JL, Nolasco L, et al. ADAMTS-13 rapidly cleaves newly secreted ultralarge von Willebrand factor multimers on the endothelial surface under flowing conditions. Blood. 2002;100(12):4033‐4039. doi:10.1182/blood-2002-05-1401
7. Chang JC. TTP-like syndrome: novel concept and molecular pathogenesis of endotheliopathy-associated vascular microthrombotic disease. Thromb J. 2018;16:20. Published 2018 Aug 11. doi:10.1186/s12959-018-0174-4
8. Magro C, Mulvey JJ, Berlin D, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. [Published online ahead of print, 2020 Apr 15.] Transl Res. 2020;S1931-5244(20)30070-0. doi:10.1016/j.trsl.2020.04.007
9. Guang Li, Sharon E. Fox, Brian Summa, et al. Multiscale 3-dimensional pathology findings of COVID-19 diseased lung using high-resolution cleared tissue microscopy. https://www.biorxiv.org/content/10.1101/2020.04.11.037473v1.full.pdf. Posted April 20, 2020. Accessed May 14, 2020. doi: 10.1101/2020.04.11.037473
10. de Bont CM, Boelens WC, Pruijn GJM. NETosis, complement, and coagulation: a triangular relationship. Cell Mol Immunol. 2019;16(1):19‐27. doi:10.1038/s41423-018-0024-0
11. Escher R, Breakey N, Lämmle B. Severe COVID-19 infection associated with endothelial activation. Thromb Res. 2020;190:62. doi:10.1016/j.thromres.2020.04.014 https://doi.org/10.1016/j.thromres.2020.04.014
12. Landa N, Mendieta-Eckert M, Fonda-Pascual P, Aguirre T. Chilblain-like lesions on feet and hands during the COVID-19 Pandemic. Int J Dermatol. 2020;59(6):739‐743. doi:10.1111/ijd.14937
13. Anderson PJ, Kokame K, Sadler JE. Zinc and calcium ions cooperatively modulate ADAMTS13 activity. J Biol Chem. 2006;281(2):850‐857. doi:10.1074/jbc.M504540200
To the Editor: COVID-19 is a pandemic caused by the virus SARS-CoV-2. Serious complications of COVID-19 are characterized by acute respiratory distress syndrome (ARDS), pneumonia and rapidly progressing to multiorgan dysfunction syndrome (MODS).
The pathophysiology of COVID-19 is not fully understood yet and neither vaccine nor clearly effective antiviral treatment is available at this time. Based on the endothelial pathogenesis of viral sepsis, which includes ARDS as seen in severe acute respiratory syndrome (SARS) due to SARS-CoV and Middle East respiratory syndrome due to MERS-CoV,1,2 we believe COVID-19-associated ARDS is also caused by endotheliopathy-associated vascular microthrombotic disease (EA-VMTD), which also involves multiorgan dysfunction syndrome (MODS) that has been reported as the cause of death.3 We suspect these complications are secondary to disequilibrium state (for various reasons4,5) between insufficient ADAMTS13 and excessive exocytosis of ultra large von Willebrand factor multimers (ULVWF) from Weibel-Palade bodies present in endothelial cells due to COVID-19-induced endotheliopathy.
Endothelial-derived ULVWF multimers anchored to the endothelial surface of the vascular wall recruit platelets and initiate microthrombogenesis within the microvasculature, leading to large microthrombi strings composed of platelet and eULVWF complexes like “beads-on-a-string structures”6 where platelets firmly adhere to eULVWF, instead of roll on eULVWF strings.4 Platelets, once adhered to eULVWF strings, are rapidly activated causing platelet aggregation and also recruit leukocytes in a P-selectin dependent manner.4 These aggregates grow until they become sufficiently large and can no longer be held onto the eULVWF strings against the force of blood flow and released from endothelial cells into the circulation.4 It appears to us that in COVID-19 microthrombotic disease, large amounts of circulating complexes of endothelial-derived ULVWF decorated-platelet microthrombi strings are filtered in the microvasculature (embolism) or develops in the microvasculature in situ causing microthrombotic occlusion. During our data search, we have come across several articles published by Chang, including on endotheliopathy causing vascular microthrombotic disease based on a novel concept of “TTP-like syndrome”7
The genesis of EA-VMTD in TTP like syndrome is suspected to be triggered by complement activation and terminal complement complex (C5b-9, membrane attack complex, MAC) may play a key role in producing endotheliopathy.7 Magro and colleagues reported that COVID-19 patients have demonstrated generalized thrombotic microvascular injury involving the lungs and skin showing intense complement activation and C5b-9 deposition in the tissue.8 Also, recent pathology reports of COVID-19 diseased lungs showed extensive platelet-rich clotting with adherent mononuclear cells and extensive fibrin clotting,9 which appear consistent with involvement of NETosis.10 In another case report from Switzerland, a patient with severe COVID-19 had massive elevation of VWF antigen and activity (555% and 520%, respectively) and increased Factor VIII clotting activity (369%).11 These findings support vascular endotheliopathy causing exocytosis of ULVWF and associated increase in Factor VIII causing microthrombotic disease/embolism.
COVID-19 clinical syndrome appears very much consistent with EA-VMTD presenting with ARDS and MODS as well as micro-macro-thrombotic complications, including peripheral ischemia/gangrene involving fingers and toes and skin necrosis.8,12
We believe that an appropriate therapy may not be anticoagulation but should include antimicrothrombotic therapy targeting endotheliopathy and primary hemostasis in the early stages of the disease (platelet adhesion, activation, and aggregation; especially eULVWF) like recombinant CD59 (membrane attack complex inhibition factor [MACIF]), recombinant ADAMTS13, glycoprotein IIb/IIIa receptor blocker, therapeutic plasma exchange, and perhaps anticomplement therapy (in selected cases) and others; these need to be validated in clinical trials prior to clinical application.
Of note, ADAMTS13 is a zinc containing protease. We noted that zinc and calcium concentrations play a significant role (in vitro) in ADAMTS13 activity in citrated plasma and recombinant ADAMTS13 activity with no added chelators (recombinant ADAMTS13 activity can enhance up to 200-fold); whereas in high zinc concentrations, ADAMTS13 gets deactivated.13 We suggest this finding merits an urgent clinical trial since it appears to us as the best possible cost-effective treatment for COVID-19 microthrombotic complications.
In this view of clinical pathophysiology of sepsis in COVID-19, we would like to enlighten the relationship between endothelial pathogenesis of coronaviral sepsis and vascular microthrombotic disease and would urge the medical community to immediately explore appropriate therapeutic options.
N. Varatharajah, MD
Suganthi Rajah, MD
Virginia, US
To the Editor: COVID-19 is a pandemic caused by the virus SARS-CoV-2. Serious complications of COVID-19 are characterized by acute respiratory distress syndrome (ARDS), pneumonia and rapidly progressing to multiorgan dysfunction syndrome (MODS).
The pathophysiology of COVID-19 is not fully understood yet and neither vaccine nor clearly effective antiviral treatment is available at this time. Based on the endothelial pathogenesis of viral sepsis, which includes ARDS as seen in severe acute respiratory syndrome (SARS) due to SARS-CoV and Middle East respiratory syndrome due to MERS-CoV,1,2 we believe COVID-19-associated ARDS is also caused by endotheliopathy-associated vascular microthrombotic disease (EA-VMTD), which also involves multiorgan dysfunction syndrome (MODS) that has been reported as the cause of death.3 We suspect these complications are secondary to disequilibrium state (for various reasons4,5) between insufficient ADAMTS13 and excessive exocytosis of ultra large von Willebrand factor multimers (ULVWF) from Weibel-Palade bodies present in endothelial cells due to COVID-19-induced endotheliopathy.
Endothelial-derived ULVWF multimers anchored to the endothelial surface of the vascular wall recruit platelets and initiate microthrombogenesis within the microvasculature, leading to large microthrombi strings composed of platelet and eULVWF complexes like “beads-on-a-string structures”6 where platelets firmly adhere to eULVWF, instead of roll on eULVWF strings.4 Platelets, once adhered to eULVWF strings, are rapidly activated causing platelet aggregation and also recruit leukocytes in a P-selectin dependent manner.4 These aggregates grow until they become sufficiently large and can no longer be held onto the eULVWF strings against the force of blood flow and released from endothelial cells into the circulation.4 It appears to us that in COVID-19 microthrombotic disease, large amounts of circulating complexes of endothelial-derived ULVWF decorated-platelet microthrombi strings are filtered in the microvasculature (embolism) or develops in the microvasculature in situ causing microthrombotic occlusion. During our data search, we have come across several articles published by Chang, including on endotheliopathy causing vascular microthrombotic disease based on a novel concept of “TTP-like syndrome”7
The genesis of EA-VMTD in TTP like syndrome is suspected to be triggered by complement activation and terminal complement complex (C5b-9, membrane attack complex, MAC) may play a key role in producing endotheliopathy.7 Magro and colleagues reported that COVID-19 patients have demonstrated generalized thrombotic microvascular injury involving the lungs and skin showing intense complement activation and C5b-9 deposition in the tissue.8 Also, recent pathology reports of COVID-19 diseased lungs showed extensive platelet-rich clotting with adherent mononuclear cells and extensive fibrin clotting,9 which appear consistent with involvement of NETosis.10 In another case report from Switzerland, a patient with severe COVID-19 had massive elevation of VWF antigen and activity (555% and 520%, respectively) and increased Factor VIII clotting activity (369%).11 These findings support vascular endotheliopathy causing exocytosis of ULVWF and associated increase in Factor VIII causing microthrombotic disease/embolism.
COVID-19 clinical syndrome appears very much consistent with EA-VMTD presenting with ARDS and MODS as well as micro-macro-thrombotic complications, including peripheral ischemia/gangrene involving fingers and toes and skin necrosis.8,12
We believe that an appropriate therapy may not be anticoagulation but should include antimicrothrombotic therapy targeting endotheliopathy and primary hemostasis in the early stages of the disease (platelet adhesion, activation, and aggregation; especially eULVWF) like recombinant CD59 (membrane attack complex inhibition factor [MACIF]), recombinant ADAMTS13, glycoprotein IIb/IIIa receptor blocker, therapeutic plasma exchange, and perhaps anticomplement therapy (in selected cases) and others; these need to be validated in clinical trials prior to clinical application.
Of note, ADAMTS13 is a zinc containing protease. We noted that zinc and calcium concentrations play a significant role (in vitro) in ADAMTS13 activity in citrated plasma and recombinant ADAMTS13 activity with no added chelators (recombinant ADAMTS13 activity can enhance up to 200-fold); whereas in high zinc concentrations, ADAMTS13 gets deactivated.13 We suggest this finding merits an urgent clinical trial since it appears to us as the best possible cost-effective treatment for COVID-19 microthrombotic complications.
In this view of clinical pathophysiology of sepsis in COVID-19, we would like to enlighten the relationship between endothelial pathogenesis of coronaviral sepsis and vascular microthrombotic disease and would urge the medical community to immediately explore appropriate therapeutic options.
N. Varatharajah, MD
Suganthi Rajah, MD
Virginia, US
1. Chang JC. Sepsis and septic shock: endothelial molecular pathogenesis associated with vascular microthrombotic disease. Thromb J. 2019;17:10. Published 2019 May 30. doi:10.1186/s12959-019-0198-4
2. Chang JC. Acute respiratory distress syndrome as an organ phenotype of vascular microthrombotic disease: based on hemostatic theory and endothelial molecular pathogenesis. Clin Appl Thromb Hemost. 2019;25:1076029619887437. doi:10.1177/1076029619887437
3. Zaim S, Chong JH, Sankaranarayanan V, Harky A. COVID-19 and multi-organ response. Curr Probl Cardiol. 2020;100618. In press. doi:10.1016/j.cpcardiol.2020.100618
4. Bernardo A, Ball C, Nolasco L, Choi H, Moake JL, Dong JF. Platelets adhered to endothelial cell-bound ultra-large von Willebrand factor strings support leukocyte tethering and rolling under high shear stress. J Thromb Haemost. 2005;3(3):562‐570. doi:10.1111/j.1538-7836.2005.01122.x https://doi.org/10.1111/j.1538-7836.2005.01122.x
5. Mannucci PM, Canciani MT, Forza I, Lussana F, Lattuada A, Rossi E. Changes in health and disease of the metalloprotease that cleaves von Willebrand factor. Blood. 2001;98(9):2730‐2735. doi:10.1182/blood.v98.9.2730
6. Dong JF, Moake JL, Nolasco L, et al. ADAMTS-13 rapidly cleaves newly secreted ultralarge von Willebrand factor multimers on the endothelial surface under flowing conditions. Blood. 2002;100(12):4033‐4039. doi:10.1182/blood-2002-05-1401
7. Chang JC. TTP-like syndrome: novel concept and molecular pathogenesis of endotheliopathy-associated vascular microthrombotic disease. Thromb J. 2018;16:20. Published 2018 Aug 11. doi:10.1186/s12959-018-0174-4
8. Magro C, Mulvey JJ, Berlin D, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. [Published online ahead of print, 2020 Apr 15.] Transl Res. 2020;S1931-5244(20)30070-0. doi:10.1016/j.trsl.2020.04.007
9. Guang Li, Sharon E. Fox, Brian Summa, et al. Multiscale 3-dimensional pathology findings of COVID-19 diseased lung using high-resolution cleared tissue microscopy. https://www.biorxiv.org/content/10.1101/2020.04.11.037473v1.full.pdf. Posted April 20, 2020. Accessed May 14, 2020. doi: 10.1101/2020.04.11.037473
10. de Bont CM, Boelens WC, Pruijn GJM. NETosis, complement, and coagulation: a triangular relationship. Cell Mol Immunol. 2019;16(1):19‐27. doi:10.1038/s41423-018-0024-0
11. Escher R, Breakey N, Lämmle B. Severe COVID-19 infection associated with endothelial activation. Thromb Res. 2020;190:62. doi:10.1016/j.thromres.2020.04.014 https://doi.org/10.1016/j.thromres.2020.04.014
12. Landa N, Mendieta-Eckert M, Fonda-Pascual P, Aguirre T. Chilblain-like lesions on feet and hands during the COVID-19 Pandemic. Int J Dermatol. 2020;59(6):739‐743. doi:10.1111/ijd.14937
13. Anderson PJ, Kokame K, Sadler JE. Zinc and calcium ions cooperatively modulate ADAMTS13 activity. J Biol Chem. 2006;281(2):850‐857. doi:10.1074/jbc.M504540200
1. Chang JC. Sepsis and septic shock: endothelial molecular pathogenesis associated with vascular microthrombotic disease. Thromb J. 2019;17:10. Published 2019 May 30. doi:10.1186/s12959-019-0198-4
2. Chang JC. Acute respiratory distress syndrome as an organ phenotype of vascular microthrombotic disease: based on hemostatic theory and endothelial molecular pathogenesis. Clin Appl Thromb Hemost. 2019;25:1076029619887437. doi:10.1177/1076029619887437
3. Zaim S, Chong JH, Sankaranarayanan V, Harky A. COVID-19 and multi-organ response. Curr Probl Cardiol. 2020;100618. In press. doi:10.1016/j.cpcardiol.2020.100618
4. Bernardo A, Ball C, Nolasco L, Choi H, Moake JL, Dong JF. Platelets adhered to endothelial cell-bound ultra-large von Willebrand factor strings support leukocyte tethering and rolling under high shear stress. J Thromb Haemost. 2005;3(3):562‐570. doi:10.1111/j.1538-7836.2005.01122.x https://doi.org/10.1111/j.1538-7836.2005.01122.x
5. Mannucci PM, Canciani MT, Forza I, Lussana F, Lattuada A, Rossi E. Changes in health and disease of the metalloprotease that cleaves von Willebrand factor. Blood. 2001;98(9):2730‐2735. doi:10.1182/blood.v98.9.2730
6. Dong JF, Moake JL, Nolasco L, et al. ADAMTS-13 rapidly cleaves newly secreted ultralarge von Willebrand factor multimers on the endothelial surface under flowing conditions. Blood. 2002;100(12):4033‐4039. doi:10.1182/blood-2002-05-1401
7. Chang JC. TTP-like syndrome: novel concept and molecular pathogenesis of endotheliopathy-associated vascular microthrombotic disease. Thromb J. 2018;16:20. Published 2018 Aug 11. doi:10.1186/s12959-018-0174-4
8. Magro C, Mulvey JJ, Berlin D, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. [Published online ahead of print, 2020 Apr 15.] Transl Res. 2020;S1931-5244(20)30070-0. doi:10.1016/j.trsl.2020.04.007
9. Guang Li, Sharon E. Fox, Brian Summa, et al. Multiscale 3-dimensional pathology findings of COVID-19 diseased lung using high-resolution cleared tissue microscopy. https://www.biorxiv.org/content/10.1101/2020.04.11.037473v1.full.pdf. Posted April 20, 2020. Accessed May 14, 2020. doi: 10.1101/2020.04.11.037473
10. de Bont CM, Boelens WC, Pruijn GJM. NETosis, complement, and coagulation: a triangular relationship. Cell Mol Immunol. 2019;16(1):19‐27. doi:10.1038/s41423-018-0024-0
11. Escher R, Breakey N, Lämmle B. Severe COVID-19 infection associated with endothelial activation. Thromb Res. 2020;190:62. doi:10.1016/j.thromres.2020.04.014 https://doi.org/10.1016/j.thromres.2020.04.014
12. Landa N, Mendieta-Eckert M, Fonda-Pascual P, Aguirre T. Chilblain-like lesions on feet and hands during the COVID-19 Pandemic. Int J Dermatol. 2020;59(6):739‐743. doi:10.1111/ijd.14937
13. Anderson PJ, Kokame K, Sadler JE. Zinc and calcium ions cooperatively modulate ADAMTS13 activity. J Biol Chem. 2006;281(2):850‐857. doi:10.1074/jbc.M504540200
A Tale of 2 Medications: A Desperate Race for Hope
For health care professionals, especially those in the epicenters of the pandemic, among the most distressing aspects of this first wave of COVID-19 has been the absence of any drug to treat the virus. The practitioners on the frontlines have confronted repeated surges of critically ill and dying patients without any effective treatment to offer, resulting in feelings of hopelessness, guilt, moral distress, depression, and in some tragic cases, suicide.2
On May 12th, the Centers of Disease Control and Prevention (CDC) released additional guidance on the antiviral medications that are the subject of this essay. The CDC may have updated its treatment guidelines in part to try and bring a measure of clinical reasoning and scientific order into the impassioned and politicized chaos that surrounded hydrocloroquine and remdesivir in the media.3
In this fourth installment of my series on pandemic ethics, we examine the desperate race for hope in the form of drug treatments for COVID-19. The race has been run faster than any in history thanks to biotechnology, genetic engineering, and artificial intelligence, although many experts believe it will still be a marathon rather than a sprint to a vaccine.4
The first editorial in this series provided a primer of the key differences between public health ethics and clinical ethics. Another crucial distinction is the far more pervasive and powerful influence of nonmedical factors in decision making, including political agendas, economic motives, journalistic hyperbole, and cultural biases and orientations. These competing interests make it even more challenging for scientists of integrity and health care institutions that are trying to uphold core values to make principled judgments about what is best for critically ill patients and the demoralized staff caring for them. In the remainder of this column, I will trace the dynamics of these forces as they impact the use of 2 drugs in federal practice: hydroxychloroquine and remdesivir.
The trajectory of hydroxychloroquine has been a political and medical roller-coaster since the pandemic hit, as is evident in its US Department of Veterans Affairs (VA) ride. Various media outlets have reported that beginning about March 26, 2020, VA placed orders for up to $400,000 of the antimalarial drug hydroxychloroquine to be given to veterans hospitalized with COVID-19.5 The same day the VA Office of Inspector General (OIG) issued a report critical of VA pandemic readiness and its availability of hydroxychloroquine.6
The VA strongly refuted the report, objecting to the premise of the OIG investigation, which was to determine whether VA facilities had on hand a 14-day supply of chloroquine or hydroxychloroquine. “This is both inaccurate and irresponsible.” Noting that the drugs were still under investigation, the VA insisted that “No conclusions have been made on their effectiveness. To insist that a 14 days’ supply of these drugs is appropriate or not appropriate displays this dangerous lack of expertise on COVID-19 and Pandemic response.”6
In April, National Institutes of Health-sponsored researchers released data that hydroxychloroquine actually increased mortality among VA patients with COVID-19,7 leading veterans’ groups and the Senate minority leader to demand that VA cease to use hydroxychloroquine for COVID-19.8 As recently as May 15, the Associated Press reported that top VA officials have defended their use of the medication and stated they will not stop administering the medication for this indication.9 And VA is not alone, many other health care institutions are still prescribing hydroxychloroquine even amid scientific controversy about its putative benefits. In response to the growing awareness of the potential harms of the drug, the World Health Organization on May 25 announced it was halting all hydroxychloroquine trials.10 Why then do some physicians and health care providers continue to prescribe it? Because when nothing else stands between the patient and certain death even if there are known risks and uncertain benefits, some in health care feel morally obliged to use their best clinical judgment to help a patient.
Remdesivir’s fortunes both scientific and monetary also rose and fell on the tide of mixed results from studies. Military Times reported on March 10, 2020, that the US Army Medical Research and Development Command had made an agreement with Gilead Sciences, the manufacturer of remdesivir, to provide the medication to COVID-19-positive service members.11 The antiviral had failed against Ebola and hepatitis but showed some efficacy for Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS). On April 15, the Secretary of the Army announced that 2 COVID-19-positive soldiers had recovered after being given remdesivir.12 In late April, the National Institute of Allergy and Infectious Diseases reported that in the scientific gold standard randomized placebo-controlled trial, remdesivir did speed the recovery of patients with advanced COVID-19. With the publication of the study in the prestigious New England Journal of Medicine on May 22, 2020, clearly the Army had bet on the right horse.13
This column has not been about quack cures and patent medicines that greed and ignorance breed in almost every American public health crisis—although these are by no means absent in this pandemic. This is about the serious endeavor of the top scientists and physicians in the country and, indeed, the world to discover a new medication or to repurpose an older pharmaceutical that is effective in the battle against COVID-19. The pressure on scientists and physicians to find a magic bullet in the battle against such an implacable enemy is unprecedented and unimaginable and can easily lead to sloppy science and ethical erosion.
In a utopia, pharmaceutical and vaccine research would be a matter of the discoveries of basic science trialed in the proof of concept of clinical care on a methodical, deliberate, and exacting timetable that balanced burdens and benefits.
In our current dystopia, science and medicine are only one of the many considerations affecting drug and vaccine development. As scientists and health care practitioners, we all experience a therapeutic imperative that we must heed with both caution and courage. Without caution we risk causing more harm than the disease we are fighting. Without courage we lose hope, the most potent antidote of all.
1. de Kruif P. Microbe Hunters. San Diego, CA: Harcourt Brace Jovanavick; 1926.
2. Watkins A, Rothfeld M, Rashbaum WK, Rosenthal BM. Top ER doctor who treated patients dies by suicide. New York Times . April 27, 2020. https://www.nytimes.com/2020/04/27/nyregion/new-york-city-doctor-suicide-coronavirus.html. Updated April 29, 2020. Accessed May 26, 2020.
3. National Institutes of Health. https://www.covid19treatmentguidelines.nih.gov/whats-new. Updated May 12, 2020. Accessed June 5, 2020.
4. Doheny K. Finish line unpredictable for COVID-19 vaccine race. https://www.webmd.com/lung/news/20200424/finish-line-unpredictable-for-covid-vaccine-race. Published April 29, 2020. Accessed May 26, 2020.
5. Horton A. What VA isn’t saying about hydroxychloroquine—and everything else related to coronavirus. Washington Post . May 1, 2020. https://www.washingtonpost.com/national-security/2020/05/01/hydroxychloroquine-veterans-trump. Accessed May 27, 2020.
6. US Department of Veterans Affairs, Veterans Health Administration, Office of the Inspector General, Office of Healthcare Inspections. OIG inspection of Veterans Health Administration COVID-19 screening processes and pandemic readiness. https://www.va.gov/oig/pubs/VAOIG-20-02221-120.pdf. Published March 19-24, 2020. Accessed May 26, 2020.
7. Maganoli J, Narendran S, Pereira F, et al. Outcomes of hydroxychloroquine usage in United States veterans hospitalized with Covid-19 [preprint]. doi.org/10.1101/2020.04.16.20065920.
8. Yen H, Balsamo M. Schumer calls on VA to explain use of unproven drug on vets. Associated Press. May10, 2020. https://apnews.com/a2830445e55c6ea324e9a23e4c38f7c3. Accessed May 27, 2020.
9. Yen H. VA says it won’t stop use of unproven drug on vets for now. Associated Press, May 15, 2020. https://apnews.com/2edd19decf58ed921d9b7ba9f6a2b44e. Accessed May 27, 2020.
10. World Health Organization. Coronavirus: WHO halts trials of hydroxychloroquine over safety fears. http://www.bbc.com/news/health-52799120. Accessed May 29, 2020.
11. Kime P. Army signs agreement with drug giant Gilead on experimental COVID-19 treatment. Military Times . March 10, 2020. https://www.militarytimes.com/news/your-military/2020/03/10/army-signs-agreement-with-drug-giant-gilead-on-experimental-covid-19-treatment. Accessed May 27, 2020.
12. Cox M. Two U.S. soldiers with Covid-19 ‘up and walking around’ after taking Ebola drug. https://www.military.com/daily-news/2020/04/15/two-us-soldiers-covid-19-and-walking-around-after-taking-ebola-drug.html. Published April 15, 2020. Accessed May 27, 2020.
13. Beigel JH, Tomashek KM, Dodd LE, et al; ACTT-1 Study Group Members. Remdesivir for the treatment of COVID-19—preliminary report. N Engl J Med. May 22, 2020. doi: 10.1056/NEJMoa2007764
For health care professionals, especially those in the epicenters of the pandemic, among the most distressing aspects of this first wave of COVID-19 has been the absence of any drug to treat the virus. The practitioners on the frontlines have confronted repeated surges of critically ill and dying patients without any effective treatment to offer, resulting in feelings of hopelessness, guilt, moral distress, depression, and in some tragic cases, suicide.2
On May 12th, the Centers of Disease Control and Prevention (CDC) released additional guidance on the antiviral medications that are the subject of this essay. The CDC may have updated its treatment guidelines in part to try and bring a measure of clinical reasoning and scientific order into the impassioned and politicized chaos that surrounded hydrocloroquine and remdesivir in the media.3
In this fourth installment of my series on pandemic ethics, we examine the desperate race for hope in the form of drug treatments for COVID-19. The race has been run faster than any in history thanks to biotechnology, genetic engineering, and artificial intelligence, although many experts believe it will still be a marathon rather than a sprint to a vaccine.4
The first editorial in this series provided a primer of the key differences between public health ethics and clinical ethics. Another crucial distinction is the far more pervasive and powerful influence of nonmedical factors in decision making, including political agendas, economic motives, journalistic hyperbole, and cultural biases and orientations. These competing interests make it even more challenging for scientists of integrity and health care institutions that are trying to uphold core values to make principled judgments about what is best for critically ill patients and the demoralized staff caring for them. In the remainder of this column, I will trace the dynamics of these forces as they impact the use of 2 drugs in federal practice: hydroxychloroquine and remdesivir.
The trajectory of hydroxychloroquine has been a political and medical roller-coaster since the pandemic hit, as is evident in its US Department of Veterans Affairs (VA) ride. Various media outlets have reported that beginning about March 26, 2020, VA placed orders for up to $400,000 of the antimalarial drug hydroxychloroquine to be given to veterans hospitalized with COVID-19.5 The same day the VA Office of Inspector General (OIG) issued a report critical of VA pandemic readiness and its availability of hydroxychloroquine.6
The VA strongly refuted the report, objecting to the premise of the OIG investigation, which was to determine whether VA facilities had on hand a 14-day supply of chloroquine or hydroxychloroquine. “This is both inaccurate and irresponsible.” Noting that the drugs were still under investigation, the VA insisted that “No conclusions have been made on their effectiveness. To insist that a 14 days’ supply of these drugs is appropriate or not appropriate displays this dangerous lack of expertise on COVID-19 and Pandemic response.”6
In April, National Institutes of Health-sponsored researchers released data that hydroxychloroquine actually increased mortality among VA patients with COVID-19,7 leading veterans’ groups and the Senate minority leader to demand that VA cease to use hydroxychloroquine for COVID-19.8 As recently as May 15, the Associated Press reported that top VA officials have defended their use of the medication and stated they will not stop administering the medication for this indication.9 And VA is not alone, many other health care institutions are still prescribing hydroxychloroquine even amid scientific controversy about its putative benefits. In response to the growing awareness of the potential harms of the drug, the World Health Organization on May 25 announced it was halting all hydroxychloroquine trials.10 Why then do some physicians and health care providers continue to prescribe it? Because when nothing else stands between the patient and certain death even if there are known risks and uncertain benefits, some in health care feel morally obliged to use their best clinical judgment to help a patient.
Remdesivir’s fortunes both scientific and monetary also rose and fell on the tide of mixed results from studies. Military Times reported on March 10, 2020, that the US Army Medical Research and Development Command had made an agreement with Gilead Sciences, the manufacturer of remdesivir, to provide the medication to COVID-19-positive service members.11 The antiviral had failed against Ebola and hepatitis but showed some efficacy for Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS). On April 15, the Secretary of the Army announced that 2 COVID-19-positive soldiers had recovered after being given remdesivir.12 In late April, the National Institute of Allergy and Infectious Diseases reported that in the scientific gold standard randomized placebo-controlled trial, remdesivir did speed the recovery of patients with advanced COVID-19. With the publication of the study in the prestigious New England Journal of Medicine on May 22, 2020, clearly the Army had bet on the right horse.13
This column has not been about quack cures and patent medicines that greed and ignorance breed in almost every American public health crisis—although these are by no means absent in this pandemic. This is about the serious endeavor of the top scientists and physicians in the country and, indeed, the world to discover a new medication or to repurpose an older pharmaceutical that is effective in the battle against COVID-19. The pressure on scientists and physicians to find a magic bullet in the battle against such an implacable enemy is unprecedented and unimaginable and can easily lead to sloppy science and ethical erosion.
In a utopia, pharmaceutical and vaccine research would be a matter of the discoveries of basic science trialed in the proof of concept of clinical care on a methodical, deliberate, and exacting timetable that balanced burdens and benefits.
In our current dystopia, science and medicine are only one of the many considerations affecting drug and vaccine development. As scientists and health care practitioners, we all experience a therapeutic imperative that we must heed with both caution and courage. Without caution we risk causing more harm than the disease we are fighting. Without courage we lose hope, the most potent antidote of all.
For health care professionals, especially those in the epicenters of the pandemic, among the most distressing aspects of this first wave of COVID-19 has been the absence of any drug to treat the virus. The practitioners on the frontlines have confronted repeated surges of critically ill and dying patients without any effective treatment to offer, resulting in feelings of hopelessness, guilt, moral distress, depression, and in some tragic cases, suicide.2
On May 12th, the Centers of Disease Control and Prevention (CDC) released additional guidance on the antiviral medications that are the subject of this essay. The CDC may have updated its treatment guidelines in part to try and bring a measure of clinical reasoning and scientific order into the impassioned and politicized chaos that surrounded hydrocloroquine and remdesivir in the media.3
In this fourth installment of my series on pandemic ethics, we examine the desperate race for hope in the form of drug treatments for COVID-19. The race has been run faster than any in history thanks to biotechnology, genetic engineering, and artificial intelligence, although many experts believe it will still be a marathon rather than a sprint to a vaccine.4
The first editorial in this series provided a primer of the key differences between public health ethics and clinical ethics. Another crucial distinction is the far more pervasive and powerful influence of nonmedical factors in decision making, including political agendas, economic motives, journalistic hyperbole, and cultural biases and orientations. These competing interests make it even more challenging for scientists of integrity and health care institutions that are trying to uphold core values to make principled judgments about what is best for critically ill patients and the demoralized staff caring for them. In the remainder of this column, I will trace the dynamics of these forces as they impact the use of 2 drugs in federal practice: hydroxychloroquine and remdesivir.
The trajectory of hydroxychloroquine has been a political and medical roller-coaster since the pandemic hit, as is evident in its US Department of Veterans Affairs (VA) ride. Various media outlets have reported that beginning about March 26, 2020, VA placed orders for up to $400,000 of the antimalarial drug hydroxychloroquine to be given to veterans hospitalized with COVID-19.5 The same day the VA Office of Inspector General (OIG) issued a report critical of VA pandemic readiness and its availability of hydroxychloroquine.6
The VA strongly refuted the report, objecting to the premise of the OIG investigation, which was to determine whether VA facilities had on hand a 14-day supply of chloroquine or hydroxychloroquine. “This is both inaccurate and irresponsible.” Noting that the drugs were still under investigation, the VA insisted that “No conclusions have been made on their effectiveness. To insist that a 14 days’ supply of these drugs is appropriate or not appropriate displays this dangerous lack of expertise on COVID-19 and Pandemic response.”6
In April, National Institutes of Health-sponsored researchers released data that hydroxychloroquine actually increased mortality among VA patients with COVID-19,7 leading veterans’ groups and the Senate minority leader to demand that VA cease to use hydroxychloroquine for COVID-19.8 As recently as May 15, the Associated Press reported that top VA officials have defended their use of the medication and stated they will not stop administering the medication for this indication.9 And VA is not alone, many other health care institutions are still prescribing hydroxychloroquine even amid scientific controversy about its putative benefits. In response to the growing awareness of the potential harms of the drug, the World Health Organization on May 25 announced it was halting all hydroxychloroquine trials.10 Why then do some physicians and health care providers continue to prescribe it? Because when nothing else stands between the patient and certain death even if there are known risks and uncertain benefits, some in health care feel morally obliged to use their best clinical judgment to help a patient.
Remdesivir’s fortunes both scientific and monetary also rose and fell on the tide of mixed results from studies. Military Times reported on March 10, 2020, that the US Army Medical Research and Development Command had made an agreement with Gilead Sciences, the manufacturer of remdesivir, to provide the medication to COVID-19-positive service members.11 The antiviral had failed against Ebola and hepatitis but showed some efficacy for Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS). On April 15, the Secretary of the Army announced that 2 COVID-19-positive soldiers had recovered after being given remdesivir.12 In late April, the National Institute of Allergy and Infectious Diseases reported that in the scientific gold standard randomized placebo-controlled trial, remdesivir did speed the recovery of patients with advanced COVID-19. With the publication of the study in the prestigious New England Journal of Medicine on May 22, 2020, clearly the Army had bet on the right horse.13
This column has not been about quack cures and patent medicines that greed and ignorance breed in almost every American public health crisis—although these are by no means absent in this pandemic. This is about the serious endeavor of the top scientists and physicians in the country and, indeed, the world to discover a new medication or to repurpose an older pharmaceutical that is effective in the battle against COVID-19. The pressure on scientists and physicians to find a magic bullet in the battle against such an implacable enemy is unprecedented and unimaginable and can easily lead to sloppy science and ethical erosion.
In a utopia, pharmaceutical and vaccine research would be a matter of the discoveries of basic science trialed in the proof of concept of clinical care on a methodical, deliberate, and exacting timetable that balanced burdens and benefits.
In our current dystopia, science and medicine are only one of the many considerations affecting drug and vaccine development. As scientists and health care practitioners, we all experience a therapeutic imperative that we must heed with both caution and courage. Without caution we risk causing more harm than the disease we are fighting. Without courage we lose hope, the most potent antidote of all.
1. de Kruif P. Microbe Hunters. San Diego, CA: Harcourt Brace Jovanavick; 1926.
2. Watkins A, Rothfeld M, Rashbaum WK, Rosenthal BM. Top ER doctor who treated patients dies by suicide. New York Times . April 27, 2020. https://www.nytimes.com/2020/04/27/nyregion/new-york-city-doctor-suicide-coronavirus.html. Updated April 29, 2020. Accessed May 26, 2020.
3. National Institutes of Health. https://www.covid19treatmentguidelines.nih.gov/whats-new. Updated May 12, 2020. Accessed June 5, 2020.
4. Doheny K. Finish line unpredictable for COVID-19 vaccine race. https://www.webmd.com/lung/news/20200424/finish-line-unpredictable-for-covid-vaccine-race. Published April 29, 2020. Accessed May 26, 2020.
5. Horton A. What VA isn’t saying about hydroxychloroquine—and everything else related to coronavirus. Washington Post . May 1, 2020. https://www.washingtonpost.com/national-security/2020/05/01/hydroxychloroquine-veterans-trump. Accessed May 27, 2020.
6. US Department of Veterans Affairs, Veterans Health Administration, Office of the Inspector General, Office of Healthcare Inspections. OIG inspection of Veterans Health Administration COVID-19 screening processes and pandemic readiness. https://www.va.gov/oig/pubs/VAOIG-20-02221-120.pdf. Published March 19-24, 2020. Accessed May 26, 2020.
7. Maganoli J, Narendran S, Pereira F, et al. Outcomes of hydroxychloroquine usage in United States veterans hospitalized with Covid-19 [preprint]. doi.org/10.1101/2020.04.16.20065920.
8. Yen H, Balsamo M. Schumer calls on VA to explain use of unproven drug on vets. Associated Press. May10, 2020. https://apnews.com/a2830445e55c6ea324e9a23e4c38f7c3. Accessed May 27, 2020.
9. Yen H. VA says it won’t stop use of unproven drug on vets for now. Associated Press, May 15, 2020. https://apnews.com/2edd19decf58ed921d9b7ba9f6a2b44e. Accessed May 27, 2020.
10. World Health Organization. Coronavirus: WHO halts trials of hydroxychloroquine over safety fears. http://www.bbc.com/news/health-52799120. Accessed May 29, 2020.
11. Kime P. Army signs agreement with drug giant Gilead on experimental COVID-19 treatment. Military Times . March 10, 2020. https://www.militarytimes.com/news/your-military/2020/03/10/army-signs-agreement-with-drug-giant-gilead-on-experimental-covid-19-treatment. Accessed May 27, 2020.
12. Cox M. Two U.S. soldiers with Covid-19 ‘up and walking around’ after taking Ebola drug. https://www.military.com/daily-news/2020/04/15/two-us-soldiers-covid-19-and-walking-around-after-taking-ebola-drug.html. Published April 15, 2020. Accessed May 27, 2020.
13. Beigel JH, Tomashek KM, Dodd LE, et al; ACTT-1 Study Group Members. Remdesivir for the treatment of COVID-19—preliminary report. N Engl J Med. May 22, 2020. doi: 10.1056/NEJMoa2007764
1. de Kruif P. Microbe Hunters. San Diego, CA: Harcourt Brace Jovanavick; 1926.
2. Watkins A, Rothfeld M, Rashbaum WK, Rosenthal BM. Top ER doctor who treated patients dies by suicide. New York Times . April 27, 2020. https://www.nytimes.com/2020/04/27/nyregion/new-york-city-doctor-suicide-coronavirus.html. Updated April 29, 2020. Accessed May 26, 2020.
3. National Institutes of Health. https://www.covid19treatmentguidelines.nih.gov/whats-new. Updated May 12, 2020. Accessed June 5, 2020.
4. Doheny K. Finish line unpredictable for COVID-19 vaccine race. https://www.webmd.com/lung/news/20200424/finish-line-unpredictable-for-covid-vaccine-race. Published April 29, 2020. Accessed May 26, 2020.
5. Horton A. What VA isn’t saying about hydroxychloroquine—and everything else related to coronavirus. Washington Post . May 1, 2020. https://www.washingtonpost.com/national-security/2020/05/01/hydroxychloroquine-veterans-trump. Accessed May 27, 2020.
6. US Department of Veterans Affairs, Veterans Health Administration, Office of the Inspector General, Office of Healthcare Inspections. OIG inspection of Veterans Health Administration COVID-19 screening processes and pandemic readiness. https://www.va.gov/oig/pubs/VAOIG-20-02221-120.pdf. Published March 19-24, 2020. Accessed May 26, 2020.
7. Maganoli J, Narendran S, Pereira F, et al. Outcomes of hydroxychloroquine usage in United States veterans hospitalized with Covid-19 [preprint]. doi.org/10.1101/2020.04.16.20065920.
8. Yen H, Balsamo M. Schumer calls on VA to explain use of unproven drug on vets. Associated Press. May10, 2020. https://apnews.com/a2830445e55c6ea324e9a23e4c38f7c3. Accessed May 27, 2020.
9. Yen H. VA says it won’t stop use of unproven drug on vets for now. Associated Press, May 15, 2020. https://apnews.com/2edd19decf58ed921d9b7ba9f6a2b44e. Accessed May 27, 2020.
10. World Health Organization. Coronavirus: WHO halts trials of hydroxychloroquine over safety fears. http://www.bbc.com/news/health-52799120. Accessed May 29, 2020.
11. Kime P. Army signs agreement with drug giant Gilead on experimental COVID-19 treatment. Military Times . March 10, 2020. https://www.militarytimes.com/news/your-military/2020/03/10/army-signs-agreement-with-drug-giant-gilead-on-experimental-covid-19-treatment. Accessed May 27, 2020.
12. Cox M. Two U.S. soldiers with Covid-19 ‘up and walking around’ after taking Ebola drug. https://www.military.com/daily-news/2020/04/15/two-us-soldiers-covid-19-and-walking-around-after-taking-ebola-drug.html. Published April 15, 2020. Accessed May 27, 2020.
13. Beigel JH, Tomashek KM, Dodd LE, et al; ACTT-1 Study Group Members. Remdesivir for the treatment of COVID-19—preliminary report. N Engl J Med. May 22, 2020. doi: 10.1056/NEJMoa2007764