User login
For MD-IQ use only
ED staff speak out about workplace violence, ask for mitigation
WASHINGTON – Speaker after speaker, veteran emergency department physicians and nurses approached the podium for a May 4 press conference on the U.S. Capitol lawn across from the East Senate steps to describe violent incidents – being bitten, punched, slapped, kicked, choked, spat on, threatened – that they have both observed and have been subject to while working in EDs.
The press conference was cosponsored by the American College of Emergency Physicians and the Emergency Nurses Association, which have partnered since 2019 on the No Silence on ED Violence campaign.
The numbers confirm their experience. A 2018 poll of 3,500 ED physicians nationwide, which was conducted by Marketing General and was reported at ACEP’s annual meeting, found that nearly half of respondents had been assaulted at work; 27% of them were injured from the assault. Nurses, who spend more time with patients, may face even higher rates.
Incidence was reported to be increasing in 2018, and that was before the social and psychological upheavals imposed by the COVID pandemic caused assaults on staff in the hospital to go up an estimated 200%-300%.
But what really grated was that more than 95% of such cases, mostly perpetrated by patients, were never prosecuted, said Jennifer Casaletto, MD, FACEP, a North Carolina emergency physician and president of the state’s ACEP chapter. “Hospital and law enforcement see violence as just part of the job in our EDs.”
It’s no secret that workplace violence is increasing, Dr. Casaletto said. Four weeks ago, she stitched up the face of a charge nurse who had been assaulted. The nurse didn’t report the incident because she didn’t believe anything would change.
“Listening to my colleagues, I know the terror they have felt in the moment – for themselves, their colleagues, their patients. I know that raw fear of being attacked, and the complex emotions that follow. I’ve been hit, bit, and punched and watched colleagues getting choked.”
Dr. Casaletto was present in the ED when an out-of-control patient clubbed a nurse with an IV pole as she tried to close the doors to other patients’ rooms. “Instinctively, I pulled my stethoscope from around my neck, hoping I wouldn’t be strangled with it.”
Tennessee emergency nurse Todd Haines, MSN, RN, AEMT, CEN, said he has stepped in to help pull patients off coworkers. “I’ve seen some staff so severely injured they could not return to the bedside. I’ve been verbally threatened. My family has been threatened by patients and their families,” he reported. “We’ve all seen it. And COVID has made some people even meaner. They just lose their minds, and ED staff take the brunt of their aggression. But then to report these incidents and hear: ‘It’s just part of your job,’ well, it’s not part of my job.”
Mr. Haines spent 10 years in law enforcement with a sheriff’s department in middle Tennessee and was on its special tactical response team before becoming an ED nurse. He said he saw many more verbal and physical assaults in 11 years in the ED than during his police career.
“I love emergency nursing at the bedside, but it got to the point where I took the first chance to leave the bedside. And I’m not alone. Other nurses are leaving in droves.” Mr. Haines now has a job directing a trauma program, and he volunteers on policy issues for the Tennessee ENA. But he worries about the toll of this violence on the ED workforce, with so many professionals already mulling over leaving the field because of job stress and burnout.
“We have to do something to keep experienced hospital emergency staff at the bedside.”
What’s the answer?
Also speaking at the press conference was Senator Tammy Baldwin (D-Wis.), who pledged to introduce the Workplace Violence Prevention for Health Care and Social Services Workers Act, which passed the House in April. This bill would direct the Occupational Health and Safety Administration to issue a standard requiring employers in health care and social services to develop and implement workplace violence prevention plans. It would cover a variety of health facilities but not doctor’s offices or home-based services.
An interim final standard would be due within a year of enactment, with a final version to follow. Covered employers would have 6 months to develop and implement their own comprehensive workplace violence prevention plans, with the meaningful participation of direct care employees, tailored for and specific to the conditions and hazards of their facility, informed by past violent incidents, and subject to the size and complexity of the setting.
The plan would also name an individual responsible for its implementation, would include staff training and education, and would require facilities to track incidents and prohibit retaliation against employees who reported incidents of workplace violence.
On Wednesday, Sen. Baldwin called for unanimous consent on the Senate floor to fast-track this bill, but that was opposed by Senator Mike Braun (R-Ind.). She will soon introduce legislation similar to HR 1195, which the House passed.
“This bill will provide long overdue protections and safety standards,” she said. It will ensure that workplaces adopt proven protection techniques, such as those in OSHA’s 2015 guideline for preventing health care workplace violence. The American Hospital Association opposed the House bill on the grounds that hospitals have already implemented policies and programs specifically tailored to address workplace violence, so the OSHA standards required by the bill are not warranted.
Another speaker at the press conference, Aisha Terry, MD, MPH, FACEP, an emergency physician for George Washington University and Veterans Affairs in Washington, D.C., and current vice president of ACEP, described an incident that occurred when she was at work. A patient punched the nurse caring for him in the face, knocking her unconscious to the floor. “I’ll never forget that sound,” Dr. Terry said. “To this day, it has impacted her career. She hasn’t known what to do.”
Many people don’t realize how bad workplace violence really is, Dr. Terry added. “You assume you can serve as the safety net of this country, taking care of patients in the context of the pandemic, and feel safe – and not have to worry about your own safety. It’s past due that we put an end to this.”
Biggest win
Mr. Haines called the workplace violence bill a game changer for ED professionals, now and into the future. “We’re not going to totally eliminate violence in the emergency department. That is part of our business. But this legislation will support us and give a safer environment for us to do the work we love,” he said.
“The biggest win for this legislation is that it will create a supportive, nonretaliatory environment. It will give us as nurses a structured way to report things.” And, when these incidents do get reported, staff will get the help they need, Mr. Haines said. “The legislation will help show the importance of implementing systems and processes in emergency settings to address the risks and hazards that makes us all vulnerable to violence.”
No relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
WASHINGTON – Speaker after speaker, veteran emergency department physicians and nurses approached the podium for a May 4 press conference on the U.S. Capitol lawn across from the East Senate steps to describe violent incidents – being bitten, punched, slapped, kicked, choked, spat on, threatened – that they have both observed and have been subject to while working in EDs.
The press conference was cosponsored by the American College of Emergency Physicians and the Emergency Nurses Association, which have partnered since 2019 on the No Silence on ED Violence campaign.
The numbers confirm their experience. A 2018 poll of 3,500 ED physicians nationwide, which was conducted by Marketing General and was reported at ACEP’s annual meeting, found that nearly half of respondents had been assaulted at work; 27% of them were injured from the assault. Nurses, who spend more time with patients, may face even higher rates.
Incidence was reported to be increasing in 2018, and that was before the social and psychological upheavals imposed by the COVID pandemic caused assaults on staff in the hospital to go up an estimated 200%-300%.
But what really grated was that more than 95% of such cases, mostly perpetrated by patients, were never prosecuted, said Jennifer Casaletto, MD, FACEP, a North Carolina emergency physician and president of the state’s ACEP chapter. “Hospital and law enforcement see violence as just part of the job in our EDs.”
It’s no secret that workplace violence is increasing, Dr. Casaletto said. Four weeks ago, she stitched up the face of a charge nurse who had been assaulted. The nurse didn’t report the incident because she didn’t believe anything would change.
“Listening to my colleagues, I know the terror they have felt in the moment – for themselves, their colleagues, their patients. I know that raw fear of being attacked, and the complex emotions that follow. I’ve been hit, bit, and punched and watched colleagues getting choked.”
Dr. Casaletto was present in the ED when an out-of-control patient clubbed a nurse with an IV pole as she tried to close the doors to other patients’ rooms. “Instinctively, I pulled my stethoscope from around my neck, hoping I wouldn’t be strangled with it.”
Tennessee emergency nurse Todd Haines, MSN, RN, AEMT, CEN, said he has stepped in to help pull patients off coworkers. “I’ve seen some staff so severely injured they could not return to the bedside. I’ve been verbally threatened. My family has been threatened by patients and their families,” he reported. “We’ve all seen it. And COVID has made some people even meaner. They just lose their minds, and ED staff take the brunt of their aggression. But then to report these incidents and hear: ‘It’s just part of your job,’ well, it’s not part of my job.”
Mr. Haines spent 10 years in law enforcement with a sheriff’s department in middle Tennessee and was on its special tactical response team before becoming an ED nurse. He said he saw many more verbal and physical assaults in 11 years in the ED than during his police career.
“I love emergency nursing at the bedside, but it got to the point where I took the first chance to leave the bedside. And I’m not alone. Other nurses are leaving in droves.” Mr. Haines now has a job directing a trauma program, and he volunteers on policy issues for the Tennessee ENA. But he worries about the toll of this violence on the ED workforce, with so many professionals already mulling over leaving the field because of job stress and burnout.
“We have to do something to keep experienced hospital emergency staff at the bedside.”
What’s the answer?
Also speaking at the press conference was Senator Tammy Baldwin (D-Wis.), who pledged to introduce the Workplace Violence Prevention for Health Care and Social Services Workers Act, which passed the House in April. This bill would direct the Occupational Health and Safety Administration to issue a standard requiring employers in health care and social services to develop and implement workplace violence prevention plans. It would cover a variety of health facilities but not doctor’s offices or home-based services.
An interim final standard would be due within a year of enactment, with a final version to follow. Covered employers would have 6 months to develop and implement their own comprehensive workplace violence prevention plans, with the meaningful participation of direct care employees, tailored for and specific to the conditions and hazards of their facility, informed by past violent incidents, and subject to the size and complexity of the setting.
The plan would also name an individual responsible for its implementation, would include staff training and education, and would require facilities to track incidents and prohibit retaliation against employees who reported incidents of workplace violence.
On Wednesday, Sen. Baldwin called for unanimous consent on the Senate floor to fast-track this bill, but that was opposed by Senator Mike Braun (R-Ind.). She will soon introduce legislation similar to HR 1195, which the House passed.
“This bill will provide long overdue protections and safety standards,” she said. It will ensure that workplaces adopt proven protection techniques, such as those in OSHA’s 2015 guideline for preventing health care workplace violence. The American Hospital Association opposed the House bill on the grounds that hospitals have already implemented policies and programs specifically tailored to address workplace violence, so the OSHA standards required by the bill are not warranted.
Another speaker at the press conference, Aisha Terry, MD, MPH, FACEP, an emergency physician for George Washington University and Veterans Affairs in Washington, D.C., and current vice president of ACEP, described an incident that occurred when she was at work. A patient punched the nurse caring for him in the face, knocking her unconscious to the floor. “I’ll never forget that sound,” Dr. Terry said. “To this day, it has impacted her career. She hasn’t known what to do.”
Many people don’t realize how bad workplace violence really is, Dr. Terry added. “You assume you can serve as the safety net of this country, taking care of patients in the context of the pandemic, and feel safe – and not have to worry about your own safety. It’s past due that we put an end to this.”
Biggest win
Mr. Haines called the workplace violence bill a game changer for ED professionals, now and into the future. “We’re not going to totally eliminate violence in the emergency department. That is part of our business. But this legislation will support us and give a safer environment for us to do the work we love,” he said.
“The biggest win for this legislation is that it will create a supportive, nonretaliatory environment. It will give us as nurses a structured way to report things.” And, when these incidents do get reported, staff will get the help they need, Mr. Haines said. “The legislation will help show the importance of implementing systems and processes in emergency settings to address the risks and hazards that makes us all vulnerable to violence.”
No relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
WASHINGTON – Speaker after speaker, veteran emergency department physicians and nurses approached the podium for a May 4 press conference on the U.S. Capitol lawn across from the East Senate steps to describe violent incidents – being bitten, punched, slapped, kicked, choked, spat on, threatened – that they have both observed and have been subject to while working in EDs.
The press conference was cosponsored by the American College of Emergency Physicians and the Emergency Nurses Association, which have partnered since 2019 on the No Silence on ED Violence campaign.
The numbers confirm their experience. A 2018 poll of 3,500 ED physicians nationwide, which was conducted by Marketing General and was reported at ACEP’s annual meeting, found that nearly half of respondents had been assaulted at work; 27% of them were injured from the assault. Nurses, who spend more time with patients, may face even higher rates.
Incidence was reported to be increasing in 2018, and that was before the social and psychological upheavals imposed by the COVID pandemic caused assaults on staff in the hospital to go up an estimated 200%-300%.
But what really grated was that more than 95% of such cases, mostly perpetrated by patients, were never prosecuted, said Jennifer Casaletto, MD, FACEP, a North Carolina emergency physician and president of the state’s ACEP chapter. “Hospital and law enforcement see violence as just part of the job in our EDs.”
It’s no secret that workplace violence is increasing, Dr. Casaletto said. Four weeks ago, she stitched up the face of a charge nurse who had been assaulted. The nurse didn’t report the incident because she didn’t believe anything would change.
“Listening to my colleagues, I know the terror they have felt in the moment – for themselves, their colleagues, their patients. I know that raw fear of being attacked, and the complex emotions that follow. I’ve been hit, bit, and punched and watched colleagues getting choked.”
Dr. Casaletto was present in the ED when an out-of-control patient clubbed a nurse with an IV pole as she tried to close the doors to other patients’ rooms. “Instinctively, I pulled my stethoscope from around my neck, hoping I wouldn’t be strangled with it.”
Tennessee emergency nurse Todd Haines, MSN, RN, AEMT, CEN, said he has stepped in to help pull patients off coworkers. “I’ve seen some staff so severely injured they could not return to the bedside. I’ve been verbally threatened. My family has been threatened by patients and their families,” he reported. “We’ve all seen it. And COVID has made some people even meaner. They just lose their minds, and ED staff take the brunt of their aggression. But then to report these incidents and hear: ‘It’s just part of your job,’ well, it’s not part of my job.”
Mr. Haines spent 10 years in law enforcement with a sheriff’s department in middle Tennessee and was on its special tactical response team before becoming an ED nurse. He said he saw many more verbal and physical assaults in 11 years in the ED than during his police career.
“I love emergency nursing at the bedside, but it got to the point where I took the first chance to leave the bedside. And I’m not alone. Other nurses are leaving in droves.” Mr. Haines now has a job directing a trauma program, and he volunteers on policy issues for the Tennessee ENA. But he worries about the toll of this violence on the ED workforce, with so many professionals already mulling over leaving the field because of job stress and burnout.
“We have to do something to keep experienced hospital emergency staff at the bedside.”
What’s the answer?
Also speaking at the press conference was Senator Tammy Baldwin (D-Wis.), who pledged to introduce the Workplace Violence Prevention for Health Care and Social Services Workers Act, which passed the House in April. This bill would direct the Occupational Health and Safety Administration to issue a standard requiring employers in health care and social services to develop and implement workplace violence prevention plans. It would cover a variety of health facilities but not doctor’s offices or home-based services.
An interim final standard would be due within a year of enactment, with a final version to follow. Covered employers would have 6 months to develop and implement their own comprehensive workplace violence prevention plans, with the meaningful participation of direct care employees, tailored for and specific to the conditions and hazards of their facility, informed by past violent incidents, and subject to the size and complexity of the setting.
The plan would also name an individual responsible for its implementation, would include staff training and education, and would require facilities to track incidents and prohibit retaliation against employees who reported incidents of workplace violence.
On Wednesday, Sen. Baldwin called for unanimous consent on the Senate floor to fast-track this bill, but that was opposed by Senator Mike Braun (R-Ind.). She will soon introduce legislation similar to HR 1195, which the House passed.
“This bill will provide long overdue protections and safety standards,” she said. It will ensure that workplaces adopt proven protection techniques, such as those in OSHA’s 2015 guideline for preventing health care workplace violence. The American Hospital Association opposed the House bill on the grounds that hospitals have already implemented policies and programs specifically tailored to address workplace violence, so the OSHA standards required by the bill are not warranted.
Another speaker at the press conference, Aisha Terry, MD, MPH, FACEP, an emergency physician for George Washington University and Veterans Affairs in Washington, D.C., and current vice president of ACEP, described an incident that occurred when she was at work. A patient punched the nurse caring for him in the face, knocking her unconscious to the floor. “I’ll never forget that sound,” Dr. Terry said. “To this day, it has impacted her career. She hasn’t known what to do.”
Many people don’t realize how bad workplace violence really is, Dr. Terry added. “You assume you can serve as the safety net of this country, taking care of patients in the context of the pandemic, and feel safe – and not have to worry about your own safety. It’s past due that we put an end to this.”
Biggest win
Mr. Haines called the workplace violence bill a game changer for ED professionals, now and into the future. “We’re not going to totally eliminate violence in the emergency department. That is part of our business. But this legislation will support us and give a safer environment for us to do the work we love,” he said.
“The biggest win for this legislation is that it will create a supportive, nonretaliatory environment. It will give us as nurses a structured way to report things.” And, when these incidents do get reported, staff will get the help they need, Mr. Haines said. “The legislation will help show the importance of implementing systems and processes in emergency settings to address the risks and hazards that makes us all vulnerable to violence.”
No relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
TikTok challenge hits Taco Bell right in its ‘Stuft Nacho’
Losing weight for TikTok: Taco Bell edition
There are many reasons why a person would want to lose weight. Too numerous to list. Losing weight to improve your health, however, doesn’t bring in a few hundred thousand TikTok subscribers. Losing weight to convince Taco Bell to bring back an obscure menu item, on the other hand ...
Chris Sandberg, a 37-year-old man from San Francisco, has struggled with his weight for years, losing and gaining hundreds of pounds in an endless cycle of feast and famine. In an unrelated development, at the start of the pandemic he also started making videos on TikTok. As the pandemic wore on, he realized that his excess weight put him at increased risk for severe COVID, as well as other chronic diseases, and he resolved to lose weight. He decided to turn his weight-loss journey into a TikTok challenge but, as we said, losing weight for its own sake isn’t enough for the almighty algorithm. He needed a different goal, preferably something offbeat and a little silly.
Back in 2013, Taco Bell introduced the Grilled Stuft Nacho, “a flour tortilla, shaped like a nacho, stuffed with beef, cheesy jalapeño sauce, sour cream and crunchy red strips,” according to its website. Mr. Sandberg discovered the item in 2015 and instantly fell in love, purchasing one every day for a week. After that first week, however, he discovered, to his horror, that the Grilled Stuft Nacho had been discontinued.
That loss haunted him for years, until inspiration struck in 2021. He pledged to work out every day on TikTok until Taco Bell brought back the Grilled Stuft Nacho. A bit incongruous, exercising for notoriously unhealthy fast food, but that’s kind of the point. He began the challenge on Jan. 4, 2021, and has continued it every day since, nearly 500 days. Over that time, he’s lost 87 pounds (from 275 at the start to under 190) and currently has 450,000 TikTok subscribers.
A year into the challenge, a local Taco Bell made Mr. Sandberg his beloved Grilled Stuft Nacho, but since the challenge was to exercise until Taco Bell brings the item back to all its restaurants, not just for him, the great journey continues. And we admire him for it. In fact, he’s inspired us: We will write a LOTME every week until it receives a Pulitzer Prize. This is important journalism we do here. Don’t deny it!
Episode XIX: COVID strikes back
So what’s next for COVID? Is Disney going to turn it into a series? Can it support a spin-off? Did James Cameron really buy the movie rights? Can it compete against the NFL in the all-important 18-34 demographic? When are Star Wars characters going to get involved?
COVID’s motivations and negotiations are pretty much a mystery to us, but we can answer that last question. They already are involved. Well, one of them anyway.
The Chinese government has been enforcing a COVID lockdown in Shanghai for over a month now, but authorities had started letting people out of their homes for short periods of time. A recent push to bring down transmission, however, has made residents increasingly frustrated and argumentative, according to Reuters.
A now-unavailable video, which Reuters could not verify, surfaced on Chinese social media showing police in hazmat suits arguing with people who were being told that they were going to be quarantined because a neighbor had tested positive.
That’s when the Force kicks in, and this next bit comes directly from the Reuters report: “This is so that we can thoroughly remove any positive cases,” one of the officers is heard saying. “Stop asking me why, there is no why.”
There is no why? Does that remind you of someone? Someone short and green, with an odd syntax? That’s right. Clearly, Yoda it is. Yoda is alive and working for the Chinese government in Shanghai. You read it here first.
Your coffee may be guilty of sexual discrimination
How do you take your coffee? Espresso, drip, instant, or brewed from a regular old coffee machine? Well, a recent study published in Open Heart suggests that gender and brewing method can alter your coffee’s effect on cholesterol levels.
Besides caffeine, coffee beans have naturally occurring chemicals such as diterpenes, cafestol, and kahweol that raise cholesterol levels in the blood. And then there are the various brewing methods, which are going to release different amounts of chemicals from the beans. According to Consumer Reports, an ounce of espresso has 63 mg of caffeine and an ounce of regular coffee has 12-16 mg. That’s a bit deceiving, though, since no one ever drinks an ounce of regular coffee, so figure 96-128 mg of caffeine for an 8-ounce cup. That’s enough to make anyone’s heart race.
Data from 21,083 participants in the seventh survey of the Tromsø Study who were aged 40 and older showed that women drank a mean of 3.8 cups per day while men drank 4.9 cups. Drinking six or more cups of plunger-brewed coffee was associated with increased cholesterol in both genders, but drinking three to five cups of espresso was significantly associated with high cholesterol in men only. Having six or more cups of filtered coffee daily raised cholesterol in women, but instant coffee increased cholesterol levels in both genders, regardless of how many cups they drank.
People all over the planet drink coffee, some of us like our lives depend on it. Since “coffee is the most frequently consumed central stimulant worldwide,” the investigators said, “even small health effects can have considerable health consequences.”
We’ll drink to that.
Have you ever dreamed of having a clone?
When will science grace us with the ability to clone ourselves? It sounds like a dream come true. Our clones can do the stuff that we don’t want to do, like sit in on that 3-hour meeting or do our grocery shopping – really just all the boring stuff we don’t want to do.
In 1996, when a sheep named Dolly became the first mammal cloned successfully, people thought it was the start of an amazing cloning era, but, alas, we haven’t made it to cloning humans yet, as LiveScience discovered when it took a look at the subject.
The idea of cloning was quite exciting for science, as people looked forward to eradicating genetic diseases and birth defects. Research done in 1999, however, countered those hopes by suggesting that cloning might increase birth defects.
So why do you think we haven’t advanced to truly cloning humans? Ethics? Time and effort? Technological barriers? “Human cloning is a particularly dramatic action, and was one of the topics that helped launch American bioethics,” Hank Greely, professor of law and genetics at Stanford (Calif.) University, told LiveScience.
What if the clones turned evil and were bent on destroying the world?
We might imagine a clone of ourselves being completely identical to us in our thoughts, actions, and physical looks. However, that’s not necessarily true; a clone would be its own person even if it looks exactly like you.
So what do the professionals think? Is it worth giving human cloning a shot? Are there benefits? Mr. Greely said that “there are none that we should be willing to consider.”
The dream of having a clone to help your son with his math homework may have gone down the drain, but maybe it’s best not to open doors that could lead to drastic changes in our world.
Losing weight for TikTok: Taco Bell edition
There are many reasons why a person would want to lose weight. Too numerous to list. Losing weight to improve your health, however, doesn’t bring in a few hundred thousand TikTok subscribers. Losing weight to convince Taco Bell to bring back an obscure menu item, on the other hand ...
Chris Sandberg, a 37-year-old man from San Francisco, has struggled with his weight for years, losing and gaining hundreds of pounds in an endless cycle of feast and famine. In an unrelated development, at the start of the pandemic he also started making videos on TikTok. As the pandemic wore on, he realized that his excess weight put him at increased risk for severe COVID, as well as other chronic diseases, and he resolved to lose weight. He decided to turn his weight-loss journey into a TikTok challenge but, as we said, losing weight for its own sake isn’t enough for the almighty algorithm. He needed a different goal, preferably something offbeat and a little silly.
Back in 2013, Taco Bell introduced the Grilled Stuft Nacho, “a flour tortilla, shaped like a nacho, stuffed with beef, cheesy jalapeño sauce, sour cream and crunchy red strips,” according to its website. Mr. Sandberg discovered the item in 2015 and instantly fell in love, purchasing one every day for a week. After that first week, however, he discovered, to his horror, that the Grilled Stuft Nacho had been discontinued.
That loss haunted him for years, until inspiration struck in 2021. He pledged to work out every day on TikTok until Taco Bell brought back the Grilled Stuft Nacho. A bit incongruous, exercising for notoriously unhealthy fast food, but that’s kind of the point. He began the challenge on Jan. 4, 2021, and has continued it every day since, nearly 500 days. Over that time, he’s lost 87 pounds (from 275 at the start to under 190) and currently has 450,000 TikTok subscribers.
A year into the challenge, a local Taco Bell made Mr. Sandberg his beloved Grilled Stuft Nacho, but since the challenge was to exercise until Taco Bell brings the item back to all its restaurants, not just for him, the great journey continues. And we admire him for it. In fact, he’s inspired us: We will write a LOTME every week until it receives a Pulitzer Prize. This is important journalism we do here. Don’t deny it!
Episode XIX: COVID strikes back
So what’s next for COVID? Is Disney going to turn it into a series? Can it support a spin-off? Did James Cameron really buy the movie rights? Can it compete against the NFL in the all-important 18-34 demographic? When are Star Wars characters going to get involved?
COVID’s motivations and negotiations are pretty much a mystery to us, but we can answer that last question. They already are involved. Well, one of them anyway.
The Chinese government has been enforcing a COVID lockdown in Shanghai for over a month now, but authorities had started letting people out of their homes for short periods of time. A recent push to bring down transmission, however, has made residents increasingly frustrated and argumentative, according to Reuters.
A now-unavailable video, which Reuters could not verify, surfaced on Chinese social media showing police in hazmat suits arguing with people who were being told that they were going to be quarantined because a neighbor had tested positive.
That’s when the Force kicks in, and this next bit comes directly from the Reuters report: “This is so that we can thoroughly remove any positive cases,” one of the officers is heard saying. “Stop asking me why, there is no why.”
There is no why? Does that remind you of someone? Someone short and green, with an odd syntax? That’s right. Clearly, Yoda it is. Yoda is alive and working for the Chinese government in Shanghai. You read it here first.
Your coffee may be guilty of sexual discrimination
How do you take your coffee? Espresso, drip, instant, or brewed from a regular old coffee machine? Well, a recent study published in Open Heart suggests that gender and brewing method can alter your coffee’s effect on cholesterol levels.
Besides caffeine, coffee beans have naturally occurring chemicals such as diterpenes, cafestol, and kahweol that raise cholesterol levels in the blood. And then there are the various brewing methods, which are going to release different amounts of chemicals from the beans. According to Consumer Reports, an ounce of espresso has 63 mg of caffeine and an ounce of regular coffee has 12-16 mg. That’s a bit deceiving, though, since no one ever drinks an ounce of regular coffee, so figure 96-128 mg of caffeine for an 8-ounce cup. That’s enough to make anyone’s heart race.
Data from 21,083 participants in the seventh survey of the Tromsø Study who were aged 40 and older showed that women drank a mean of 3.8 cups per day while men drank 4.9 cups. Drinking six or more cups of plunger-brewed coffee was associated with increased cholesterol in both genders, but drinking three to five cups of espresso was significantly associated with high cholesterol in men only. Having six or more cups of filtered coffee daily raised cholesterol in women, but instant coffee increased cholesterol levels in both genders, regardless of how many cups they drank.
People all over the planet drink coffee, some of us like our lives depend on it. Since “coffee is the most frequently consumed central stimulant worldwide,” the investigators said, “even small health effects can have considerable health consequences.”
We’ll drink to that.
Have you ever dreamed of having a clone?
When will science grace us with the ability to clone ourselves? It sounds like a dream come true. Our clones can do the stuff that we don’t want to do, like sit in on that 3-hour meeting or do our grocery shopping – really just all the boring stuff we don’t want to do.
In 1996, when a sheep named Dolly became the first mammal cloned successfully, people thought it was the start of an amazing cloning era, but, alas, we haven’t made it to cloning humans yet, as LiveScience discovered when it took a look at the subject.
The idea of cloning was quite exciting for science, as people looked forward to eradicating genetic diseases and birth defects. Research done in 1999, however, countered those hopes by suggesting that cloning might increase birth defects.
So why do you think we haven’t advanced to truly cloning humans? Ethics? Time and effort? Technological barriers? “Human cloning is a particularly dramatic action, and was one of the topics that helped launch American bioethics,” Hank Greely, professor of law and genetics at Stanford (Calif.) University, told LiveScience.
What if the clones turned evil and were bent on destroying the world?
We might imagine a clone of ourselves being completely identical to us in our thoughts, actions, and physical looks. However, that’s not necessarily true; a clone would be its own person even if it looks exactly like you.
So what do the professionals think? Is it worth giving human cloning a shot? Are there benefits? Mr. Greely said that “there are none that we should be willing to consider.”
The dream of having a clone to help your son with his math homework may have gone down the drain, but maybe it’s best not to open doors that could lead to drastic changes in our world.
Losing weight for TikTok: Taco Bell edition
There are many reasons why a person would want to lose weight. Too numerous to list. Losing weight to improve your health, however, doesn’t bring in a few hundred thousand TikTok subscribers. Losing weight to convince Taco Bell to bring back an obscure menu item, on the other hand ...
Chris Sandberg, a 37-year-old man from San Francisco, has struggled with his weight for years, losing and gaining hundreds of pounds in an endless cycle of feast and famine. In an unrelated development, at the start of the pandemic he also started making videos on TikTok. As the pandemic wore on, he realized that his excess weight put him at increased risk for severe COVID, as well as other chronic diseases, and he resolved to lose weight. He decided to turn his weight-loss journey into a TikTok challenge but, as we said, losing weight for its own sake isn’t enough for the almighty algorithm. He needed a different goal, preferably something offbeat and a little silly.
Back in 2013, Taco Bell introduced the Grilled Stuft Nacho, “a flour tortilla, shaped like a nacho, stuffed with beef, cheesy jalapeño sauce, sour cream and crunchy red strips,” according to its website. Mr. Sandberg discovered the item in 2015 and instantly fell in love, purchasing one every day for a week. After that first week, however, he discovered, to his horror, that the Grilled Stuft Nacho had been discontinued.
That loss haunted him for years, until inspiration struck in 2021. He pledged to work out every day on TikTok until Taco Bell brought back the Grilled Stuft Nacho. A bit incongruous, exercising for notoriously unhealthy fast food, but that’s kind of the point. He began the challenge on Jan. 4, 2021, and has continued it every day since, nearly 500 days. Over that time, he’s lost 87 pounds (from 275 at the start to under 190) and currently has 450,000 TikTok subscribers.
A year into the challenge, a local Taco Bell made Mr. Sandberg his beloved Grilled Stuft Nacho, but since the challenge was to exercise until Taco Bell brings the item back to all its restaurants, not just for him, the great journey continues. And we admire him for it. In fact, he’s inspired us: We will write a LOTME every week until it receives a Pulitzer Prize. This is important journalism we do here. Don’t deny it!
Episode XIX: COVID strikes back
So what’s next for COVID? Is Disney going to turn it into a series? Can it support a spin-off? Did James Cameron really buy the movie rights? Can it compete against the NFL in the all-important 18-34 demographic? When are Star Wars characters going to get involved?
COVID’s motivations and negotiations are pretty much a mystery to us, but we can answer that last question. They already are involved. Well, one of them anyway.
The Chinese government has been enforcing a COVID lockdown in Shanghai for over a month now, but authorities had started letting people out of their homes for short periods of time. A recent push to bring down transmission, however, has made residents increasingly frustrated and argumentative, according to Reuters.
A now-unavailable video, which Reuters could not verify, surfaced on Chinese social media showing police in hazmat suits arguing with people who were being told that they were going to be quarantined because a neighbor had tested positive.
That’s when the Force kicks in, and this next bit comes directly from the Reuters report: “This is so that we can thoroughly remove any positive cases,” one of the officers is heard saying. “Stop asking me why, there is no why.”
There is no why? Does that remind you of someone? Someone short and green, with an odd syntax? That’s right. Clearly, Yoda it is. Yoda is alive and working for the Chinese government in Shanghai. You read it here first.
Your coffee may be guilty of sexual discrimination
How do you take your coffee? Espresso, drip, instant, or brewed from a regular old coffee machine? Well, a recent study published in Open Heart suggests that gender and brewing method can alter your coffee’s effect on cholesterol levels.
Besides caffeine, coffee beans have naturally occurring chemicals such as diterpenes, cafestol, and kahweol that raise cholesterol levels in the blood. And then there are the various brewing methods, which are going to release different amounts of chemicals from the beans. According to Consumer Reports, an ounce of espresso has 63 mg of caffeine and an ounce of regular coffee has 12-16 mg. That’s a bit deceiving, though, since no one ever drinks an ounce of regular coffee, so figure 96-128 mg of caffeine for an 8-ounce cup. That’s enough to make anyone’s heart race.
Data from 21,083 participants in the seventh survey of the Tromsø Study who were aged 40 and older showed that women drank a mean of 3.8 cups per day while men drank 4.9 cups. Drinking six or more cups of plunger-brewed coffee was associated with increased cholesterol in both genders, but drinking three to five cups of espresso was significantly associated with high cholesterol in men only. Having six or more cups of filtered coffee daily raised cholesterol in women, but instant coffee increased cholesterol levels in both genders, regardless of how many cups they drank.
People all over the planet drink coffee, some of us like our lives depend on it. Since “coffee is the most frequently consumed central stimulant worldwide,” the investigators said, “even small health effects can have considerable health consequences.”
We’ll drink to that.
Have you ever dreamed of having a clone?
When will science grace us with the ability to clone ourselves? It sounds like a dream come true. Our clones can do the stuff that we don’t want to do, like sit in on that 3-hour meeting or do our grocery shopping – really just all the boring stuff we don’t want to do.
In 1996, when a sheep named Dolly became the first mammal cloned successfully, people thought it was the start of an amazing cloning era, but, alas, we haven’t made it to cloning humans yet, as LiveScience discovered when it took a look at the subject.
The idea of cloning was quite exciting for science, as people looked forward to eradicating genetic diseases and birth defects. Research done in 1999, however, countered those hopes by suggesting that cloning might increase birth defects.
So why do you think we haven’t advanced to truly cloning humans? Ethics? Time and effort? Technological barriers? “Human cloning is a particularly dramatic action, and was one of the topics that helped launch American bioethics,” Hank Greely, professor of law and genetics at Stanford (Calif.) University, told LiveScience.
What if the clones turned evil and were bent on destroying the world?
We might imagine a clone of ourselves being completely identical to us in our thoughts, actions, and physical looks. However, that’s not necessarily true; a clone would be its own person even if it looks exactly like you.
So what do the professionals think? Is it worth giving human cloning a shot? Are there benefits? Mr. Greely said that “there are none that we should be willing to consider.”
The dream of having a clone to help your son with his math homework may have gone down the drain, but maybe it’s best not to open doors that could lead to drastic changes in our world.
Inverted Appendix in a Patient With Weakness and Occult Bleeding
Appendiceal mucinous neoplasms (AMNs) are rare tumors of the appendix that can be asymptomatic or present with acute right lower quadrant (RLQ) pain mimicking appendicitis. Due to their potential to cause either no symptoms or nonspecific symptoms, such as abdominal pain, nausea, or vomiting, AMNs are often found incidentally during appendectomies or, even more rarely, colonoscopies. Most AMNs grow slowly and have little metastatic potential. However, due to potential complications, such as bowel obstruction and rupture, timely detection and removal of AMN is essential. We describe the case of a patient who appeared to have acute appendicitis complicated by rupture on imaging who was found instead to have a perforated low-grade AMN during surgery.
Case Presentation
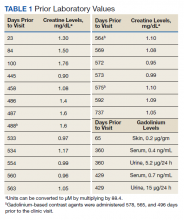
A male patient aged 72 years with a history of type 2 diabetes mellitus, hypertension, and aortic stenosis, but no prior abdominal surgery, presented with a chief concern of generalized weakness. As part of the workup for his weakness, a computed tomography (CT) scan of the abdomen was performed which showed an RLQ phlegmon and mild fat stranding in the area. Imaging also revealed an asymptomatic gallstone measuring 1.5 cm with no evidence of cholecystitis. The patient had no fever and reported no abdominal pain, nausea, vomiting, or change in bowel habits. On physical examination, the patient’s abdomen was soft, nontender, and nondistended with normoactive bowel sounds and no rebound or guarding.
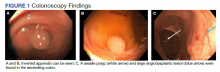
To manage the appendicitis, the patient started a 2-week course of amoxicillin clavulanate 875 mg twice daily and was instructed to schedule an interval appendectomy in the coming months. Four days later, during a follow-up with his primary care physician, he was found to be asymptomatic. However, at this visit his stool was found to be positive for occult blood. Given this finding and the lack of a previous colonoscopy, the patient underwent a colonoscopy, which revealed bulging at the appendiceal orifice, consistent with an inverted appendix. Portions of the appendix were biopsied (Figure 1). Histologic analysis of the appendiceal biopsies revealed no dysplasia or malignancy. The colonoscopy also revealed an 8-mm sessile polyp in the ascending colon which was resected, and histologic analysis of this polyp revealed a low-grade tubular adenoma. Additionally, a large angiodysplastic lesion was found in the ascending colon as well as external and medium-sized internal hemorrhoids.
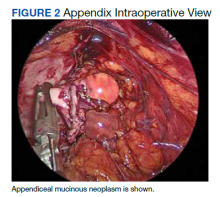
Six weeks after the colonoscopy, the patient was taken to the operating room for a laparoscopic appendectomy. Upon entry of the abdomen, extensive adhesions throughout the RLQ were found which required adhesiolysis. A calcified fecalith adherent to the mesentery of the small intestine in the RLQ was also found and resected. After lysis of the adhesions, the appendix and fibrotic tissue surrounding it could be seen (Figure 2). The appendix was dilated and the tip showed perforation. During dissection of the appendix, clear gelatinous material was found coming from the appendiceal lumen as well as from the fibrotic tissue around the appendix. On postoperative day 1 the appendix was resected and the patient was discharged.
Histologic specimens of the appendix were notable for evidence of perforation and neoplasia leading to a diagnosis of low-grade AMN. The presence of atypical mucinous epithelial cells on the serosal surface of the appendix, confirmed with a positive pancytokeratin stain, provided histologic evidence of appendiceal perforation (Figure 3). The presence of nuclear atypia demonstrated that the appendix was involved by a neoplastic process. Additionally, attenuation of the normal appendiceal epithelium, evidence of a chronic process, further helped to differentiate the AMN from complicated appendicitis. The presence of mucin involving the serosa of the appendix led to the classification of this patient’s neoplasm as grade pT4a. Of note, histologic examination demonstrated that the surgical margins contained tumor cells.
Given the positive margins of the resected AMN and the relatively large size of the neoplasm, a laparoscopic right hemicolectomy was performed 2 months later. Although multiple adhesions were found in the terminal ileum, cecum, and ascending colon during the hemicolectomy, no mucinous lesions were observed grossly. Histologic analysis showed no residual neoplasm as well as no lymph node involvement. On postoperative day 3 the patient was discharged and had an uneventful recovery. At his first surveillance visit 6 months after his hemicolectomy, the patient appeared to be doing well and reported no abdominal pain, nausea, vomiting, change in bowel habits, or any blood in the stool.
Discussion
AMNs are rare tumors with an annual age-adjusted incidence of approximately 0.12 per 1,000,000 people.1 These neoplasms can present as acute or chronic abdominal pain, gastrointestinal bleeding, intestinal obstruction, or acute abdomen.2-4 Most AMNs, however, are asymptomatic and are usually found incidentally during appendectomies for appendicitis, and can even be found during colonoscopies,such as in this case.5,6
Low-grade AMNs are distinguished from appendiceal mucinous adenocarcinomas by their lack of wall invasion.7 Additionally, low-grade AMNs have a very good prognosis as even neoplasms that have spread outside of the appendix have a 5-year overall survival rate of 79 to 86%.8 These low-grade neoplasms also have extremely low rates of recurrence after resection.9 In contrast, appendiceal mucinous adenocarcinomas have a much worse prognosis with a 5-year overall survival rate of 53.6%.10
Treatment of AMNs depends on the extent of their spread. Neoplasms that are confined to the appendix can typically be treated with appendectomy alone, while those that have spread beyond the appendix may require cytoreductive surgery and chemotherapy, namely, hyperthermic intraperitoneal chemotherapy (HIPEC), in addition to appendectomy.11 Cases in which neoplasms are not confined to the appendix also require more frequent surveillance for recurrence as compared to appendix-restricted neoplasms.11
Appendiceal inversion is a rare finding in adults with an estimated prevalence of 0.01%.6 Not only is appendiceal inversion rare in and of itself, it is even more rarely found in combination with appendiceal neoplasms.6 Other causes of appendiceal inversion include intussusception, acute appendicitis, appendiceal nodule, or even iatrogenic due to appendectomy.12-14 While appendiceal inversion can be completely benign, because these morphological changes of the appendix can resemble a polyp, these lesions are often biopsied and/or resected.15 However, lesion resection may be quite problematic due to high risk of bleeding and perforation.15 In order to avoid the risks associated with resection of a potentially benign finding, biopsy should be performed prior to any attempted resection of inverted appendices.15
Another interesting aspect of this case is the finding of fecal occult blood. The differential for fecal occult blood is quite broad and the patient had multiple conditions that could have led to the finding of occult blood in his stool. Hemorrhoids can cause a positive result on a fecal occult blood test (FOBT) although this is relatively uncommon, and hemorrhoids are more likely to cause frank blood to be seen.16 The sessile polyp found in the patient’s colon may also have caused the FOBT to be positive. This patient was also found to have an angiodysplasia (a finding that is associated with aortic stenosis, which this patient has a history of) which can also cause gastrointestinal bleeding.17 Lastly, AMNs may also cause gastrointestinal bleeding and thus a positive FOBT, although bleeding is a relatively uncommon presentation of AMNs, especially those that are low-grade as in this case.18
This case also highlights the association between appendiceal neoplasms and colonic neoplastic lesions. Patients with appendiceal neoplasms are more likely to have colonic neoplastic lesions than patients without appendiceal neoplasms.19 Studies have found that approximately 13 to 42% of patients with appendiceal neoplasms also have colonic neoplastic lesions.19 The majority of these lesions in the colon were right-sided and this finding was also seen in this case as the patient’s polyp was located in the ascending colon.19 Due to this association between appendiceal and colorectal neoplasia, the American Society of Colon and Rectal Surgeons strongly recommends that patients with appendiceal neoplasms or who are suspected of having them receive a colonoscopy.19
Additionally, perforation of an AMN, as was seen in this case, is a finding that should raise significant concern. Perforation of an AMN allows for the spread of malignant mucinous epithelial cells throughout the abdomen. The finding of extensive adhesions throughout the patient’s RLQ was unexpected as abdominal adhesions are most often seen in patients with a history of abdominal surgeries. Considering the lack of any prior abdominal surgeries in this patient, these adhesions were most likely the result of the spread and proliferation of malignant mucinous epithelial cells from the perforated AMN in the RLQ.20 The adhesiolysis performed in this case was thus not only important in order to visualize the appendix, but also for preventing future complications of abdominal adhesions such as bowel obstruction.20 Perforated AMN is also so concerning because it can potentially lead to pseudomyxoma peritonei—a condition in which malignant mucinous epithelial cells accumulate in the abdomen.21 Pseudomyxoma peritonei is extremely rare with an incidence of approximately 1 to 2 cases per million per year.22 Early recognition of AMNs and surgical referral are critically important as pseudomyxoma peritonei is difficult to treat, has a high rate of recurrence, and can be fatal.23
Lastly, this case highlights how findings of a ruptured appendix and/or mucin surrounding the appendix on imaging should warrant laparoscopy because only pathologic analysis of the appendix can definitively rule out AMNs. The utility of laparoscopic evaluation of the appendix is especially apparent as nonsurgical treatment of appendicitis using antibiotics is gaining favor for treating even complicated appendicitis.24 Appendicitis is much more common than AMNs. However, had the patient in this case only been given antibiotics for his suspected complicated appendicitis without any colonoscopy or appendectomy, the neoplasm in his appendix would have gone undetected and continued to grow, causing significant complications. The patient’s age at presentation in this case also necessitated laparoscopic evaluation of the appendix as the incidence of AMNs is highest among patients aged > 60 years.25 Additionally, because appendiceal inversion may be seen with AMNs,the patient’s inverted appendix seen during his colonoscopy was another compelling reason for laparoscopic evaluation of his appendix.6
Conclusions
AMNs can present with nonspecific symptoms or can be completely asymptomatic and are often found incidentally during colonoscopies or appendectomies for acute appendicitis. While it is true that AMNs have low metastatic potential and grow slowly, AMNs can rupture leading to pseudomyxoma peritonei or even cause bowel obstruction warranting timely identification and removal of these neoplasms. Laparoscopic evaluation in cases of ruptured appendices is critical not only for treatment, but also for determining the presence of a potential underlying appendiceal malignancy. Although AMNs are a rare pathology, physicians should still consider the possibility of these neoplasms even when imaging findings suggest appendicitis. Having AMNs as part of the differential diagnosis is especially necessary in cases, such as this one, in which the patient has appendiceal inversion, is aged > 50 years, and has concurrent colorectal neoplasms.
1. Shaib WL, Goodman M, Chen Z, et al. Incidence and survival of appendiceal mucinous neoplasms: a SEER analysis. Am J Clin Oncol. 2017;40(6):569-573. doi:10.1097/COC.0000000000000210
2. Kehagias I, Zygomalas A, Markopoulos G, Papandreou T, Kraniotis P. Diagnosis and treatment of mucinous appendiceal neoplasm presented as acute appendicitis. Case Rep Oncol Med. 2016;3:1-6. doi:10.1155/2016/2161952
3. Karatas M, Simsek C, Gunay S, et al. Acute lower gastrointestinal bleeding due to low-grade mucinous neoplasm of appendix. Acta Chir Belg. 2020;120(4):1-4. doi:10.1080/00015458.2020.1860397
4. Mourad FH, Hussein M, Bahlawan M, Haddad M, Tawil A. Intestinal obstruction secondary to appendiceal mucocele. Dig Dis Sci. 1999;44(8):1594-1599. doi:10.1023/a:1026615010989
5. Benabe SH, Leeman R, Brady AC, Hirzel A, Langshaw AH. Low-grade appendiceal mucinous neoplasm in an adolescent patient with untreated Crohn’s disease. ACG Case Reports J. 2020;7(3). doi:10.14309/crj.0000000000000338
6. Liu X, Liu G, Liu Y, et al. Complete appendiceal inversion with local high-grade intraepithelial neoplasia in an adult female: A case report. BMC Surg. 2019;19(1). doi:10.1186/s12893-019-0632-3
7. Gündog˘ar ÖS, Kımılog˘lu ES, Komut NS, et al. The evaluation of appendiceal mucinous neoplasms with a new classification system. Turk J Gastroenterol. 2018;29(5):532-542. doi:10.5152/tjg.2018.17605
8. Misdraji J, Yantiss RK, Graeme-Cook FM, Balis UJ, Young RH. Appendiceal mucinous neoplasms: a clinicopathologic analysis of 107 cases. Am J Surg Pathol. 2003;27(8):1089-1103. doi:10.1097/00000478-200308000-00006
9. Pai RK, Beck AH, Norton JA, Longacre TA. Appendiceal mucinous neoplasms: clinicopathologic study of 116 cases with analysis of factors predicting recurrence. Am J Surg Pathol. 2009;33(10):1425-1439. doi:10.1097/PAS.0b013e3181af6067
10. Asare EA, Compton CC, Hanna NN, et al. The impact of stage, grade, and mucinous histology on the efficacy of systemic chemotherapy in adenocarcinomas of the appendix: analysis of the National Cancer Data Base. Cancer. 2015;122(2):213-221. doi:10.1002/cncr.29744
11. Shaib WL, Assi R, Shamseddine A, et al. Appendiceal mucinous neoplasms: diagnosis and management. Oncologist. 2018;23(1):137. doi:10.1634/theoncologist.2017-0081erratum
12. Tran C, Sakioka J, Nguyen E, Beutler BD, Hsu J. An inverted appendix found on routine colonoscopy: a case report with discussion of imaging findings. Radiol Case Rep. 2019;14(8):952-955. doi:10.1016/j.radcr.2019.05.022
13. Shafi A, Azab M. A case of everted appendix with benign appendiceal nodule masquerading as appendiceal mucocele: a case report. Am J Gastroenterol. 2018;113:S1436. doi:10.14309/00000434-201810001-02585
14. Pokhrel B, Chang M, Anand G, Savides T, Fehmi S. Appendiceal mucinous neoplasm in an inverted appendix found on prior colonoscopy. VideoGIE. 2020;5(1):34-36. doi:10.1016/j.vgie.2019.09.013
15. Johnson EK, Arcila ME, Steele SR. Appendiceal inversion: a diagnostic and therapeutic dilemma. JSLS. 2009;13(1):92-95.
16. van Turenhout ST, Oort FA, sive Droste JST, et al. Hemorrhoids detected at colonoscopy: an infrequent cause of false-positive fecal immunochemical test results. Gastrointest Endosc. 2012;76(1):136-143. doi:10.1016/j.gie.2012.03.169
17. Hudzik B, Wilczek K, Gasior M. Heyde syndrome: Gastrointestinal bleeding and aortic stenosis. CMAJ. 2016;188(2):135-138. doi:10.1503/cmaj.150194
18. Leonards LM, Pahwa A, Patel MK, Petersen J, Nguyen MJ, Jude CM. Neoplasms of the appendix: pictorial review with clinical and pathologic correlation. RadioGraphics. 2017;37(4):1059-1083. doi:10.1148/rg.2017160150
19. Glasgow SC, Gaertner W, Stewart D, et al. The American Society of Colon and Rectal Surgeons, clinical practice guidelines for the management of appendiceal neoplasms. Dis Colon Rectum. 2019;62(12):1425-1438. doi:10.1097/DCR.0000000000001530
20. Panagopoulos P, Tsokaki T, Misiakos E, et al. Low-grade appendiceal mucinous neoplasm presenting as an adnexal mass. Case Reports in Obstetrics and Gynecology. 2017;2017:1-3. doi:10.1155/2017/7165321
21. Ramaswamy V. Pathology of mucinous appendiceal tumors and pseudomyxoma peritonei. Indian J Surg Oncol. 2016;7(2):258-267. doi:10.1007/s13193-016-0516-2.
22. Bevan KE, Mohamed F, Moran BJ. Pseudomyxoma peritonei. World J Gastrointest Oncol. 2010;2(1):44-50. doi:10.4251/wjgo.v2.i1.44
23. Mercier F, Dagbert F, Pocard M, et al. Recurrence of pseudomyxoma peritonei after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. BJS Open. 2018;3(2):195-202. doi:10.1002/bjs5.97
24. David A, Dodgion C, Eddine SBZ, Davila D, Webb TP, Trevino CM. Perforated appendicitis: Short duration antibiotics are noninferior to traditional long duration antibiotics. Surgery. 2020;167(2):475-477. doi:10.1016/j.surg.2019.08.007
25. Raijman I, Leong S, Hassaram S, Marcon NE. Appendiceal mucocele: Endoscopic appearance. Endoscopy. 1994;26(3):326-328. doi:10.1055/s-2007-1008979
Appendiceal mucinous neoplasms (AMNs) are rare tumors of the appendix that can be asymptomatic or present with acute right lower quadrant (RLQ) pain mimicking appendicitis. Due to their potential to cause either no symptoms or nonspecific symptoms, such as abdominal pain, nausea, or vomiting, AMNs are often found incidentally during appendectomies or, even more rarely, colonoscopies. Most AMNs grow slowly and have little metastatic potential. However, due to potential complications, such as bowel obstruction and rupture, timely detection and removal of AMN is essential. We describe the case of a patient who appeared to have acute appendicitis complicated by rupture on imaging who was found instead to have a perforated low-grade AMN during surgery.
Case Presentation
A male patient aged 72 years with a history of type 2 diabetes mellitus, hypertension, and aortic stenosis, but no prior abdominal surgery, presented with a chief concern of generalized weakness. As part of the workup for his weakness, a computed tomography (CT) scan of the abdomen was performed which showed an RLQ phlegmon and mild fat stranding in the area. Imaging also revealed an asymptomatic gallstone measuring 1.5 cm with no evidence of cholecystitis. The patient had no fever and reported no abdominal pain, nausea, vomiting, or change in bowel habits. On physical examination, the patient’s abdomen was soft, nontender, and nondistended with normoactive bowel sounds and no rebound or guarding.
To manage the appendicitis, the patient started a 2-week course of amoxicillin clavulanate 875 mg twice daily and was instructed to schedule an interval appendectomy in the coming months. Four days later, during a follow-up with his primary care physician, he was found to be asymptomatic. However, at this visit his stool was found to be positive for occult blood. Given this finding and the lack of a previous colonoscopy, the patient underwent a colonoscopy, which revealed bulging at the appendiceal orifice, consistent with an inverted appendix. Portions of the appendix were biopsied (Figure 1). Histologic analysis of the appendiceal biopsies revealed no dysplasia or malignancy. The colonoscopy also revealed an 8-mm sessile polyp in the ascending colon which was resected, and histologic analysis of this polyp revealed a low-grade tubular adenoma. Additionally, a large angiodysplastic lesion was found in the ascending colon as well as external and medium-sized internal hemorrhoids.
Six weeks after the colonoscopy, the patient was taken to the operating room for a laparoscopic appendectomy. Upon entry of the abdomen, extensive adhesions throughout the RLQ were found which required adhesiolysis. A calcified fecalith adherent to the mesentery of the small intestine in the RLQ was also found and resected. After lysis of the adhesions, the appendix and fibrotic tissue surrounding it could be seen (Figure 2). The appendix was dilated and the tip showed perforation. During dissection of the appendix, clear gelatinous material was found coming from the appendiceal lumen as well as from the fibrotic tissue around the appendix. On postoperative day 1 the appendix was resected and the patient was discharged.
Histologic specimens of the appendix were notable for evidence of perforation and neoplasia leading to a diagnosis of low-grade AMN. The presence of atypical mucinous epithelial cells on the serosal surface of the appendix, confirmed with a positive pancytokeratin stain, provided histologic evidence of appendiceal perforation (Figure 3). The presence of nuclear atypia demonstrated that the appendix was involved by a neoplastic process. Additionally, attenuation of the normal appendiceal epithelium, evidence of a chronic process, further helped to differentiate the AMN from complicated appendicitis. The presence of mucin involving the serosa of the appendix led to the classification of this patient’s neoplasm as grade pT4a. Of note, histologic examination demonstrated that the surgical margins contained tumor cells.
Given the positive margins of the resected AMN and the relatively large size of the neoplasm, a laparoscopic right hemicolectomy was performed 2 months later. Although multiple adhesions were found in the terminal ileum, cecum, and ascending colon during the hemicolectomy, no mucinous lesions were observed grossly. Histologic analysis showed no residual neoplasm as well as no lymph node involvement. On postoperative day 3 the patient was discharged and had an uneventful recovery. At his first surveillance visit 6 months after his hemicolectomy, the patient appeared to be doing well and reported no abdominal pain, nausea, vomiting, change in bowel habits, or any blood in the stool.
Discussion
AMNs are rare tumors with an annual age-adjusted incidence of approximately 0.12 per 1,000,000 people.1 These neoplasms can present as acute or chronic abdominal pain, gastrointestinal bleeding, intestinal obstruction, or acute abdomen.2-4 Most AMNs, however, are asymptomatic and are usually found incidentally during appendectomies for appendicitis, and can even be found during colonoscopies,such as in this case.5,6
Low-grade AMNs are distinguished from appendiceal mucinous adenocarcinomas by their lack of wall invasion.7 Additionally, low-grade AMNs have a very good prognosis as even neoplasms that have spread outside of the appendix have a 5-year overall survival rate of 79 to 86%.8 These low-grade neoplasms also have extremely low rates of recurrence after resection.9 In contrast, appendiceal mucinous adenocarcinomas have a much worse prognosis with a 5-year overall survival rate of 53.6%.10
Treatment of AMNs depends on the extent of their spread. Neoplasms that are confined to the appendix can typically be treated with appendectomy alone, while those that have spread beyond the appendix may require cytoreductive surgery and chemotherapy, namely, hyperthermic intraperitoneal chemotherapy (HIPEC), in addition to appendectomy.11 Cases in which neoplasms are not confined to the appendix also require more frequent surveillance for recurrence as compared to appendix-restricted neoplasms.11
Appendiceal inversion is a rare finding in adults with an estimated prevalence of 0.01%.6 Not only is appendiceal inversion rare in and of itself, it is even more rarely found in combination with appendiceal neoplasms.6 Other causes of appendiceal inversion include intussusception, acute appendicitis, appendiceal nodule, or even iatrogenic due to appendectomy.12-14 While appendiceal inversion can be completely benign, because these morphological changes of the appendix can resemble a polyp, these lesions are often biopsied and/or resected.15 However, lesion resection may be quite problematic due to high risk of bleeding and perforation.15 In order to avoid the risks associated with resection of a potentially benign finding, biopsy should be performed prior to any attempted resection of inverted appendices.15
Another interesting aspect of this case is the finding of fecal occult blood. The differential for fecal occult blood is quite broad and the patient had multiple conditions that could have led to the finding of occult blood in his stool. Hemorrhoids can cause a positive result on a fecal occult blood test (FOBT) although this is relatively uncommon, and hemorrhoids are more likely to cause frank blood to be seen.16 The sessile polyp found in the patient’s colon may also have caused the FOBT to be positive. This patient was also found to have an angiodysplasia (a finding that is associated with aortic stenosis, which this patient has a history of) which can also cause gastrointestinal bleeding.17 Lastly, AMNs may also cause gastrointestinal bleeding and thus a positive FOBT, although bleeding is a relatively uncommon presentation of AMNs, especially those that are low-grade as in this case.18
This case also highlights the association between appendiceal neoplasms and colonic neoplastic lesions. Patients with appendiceal neoplasms are more likely to have colonic neoplastic lesions than patients without appendiceal neoplasms.19 Studies have found that approximately 13 to 42% of patients with appendiceal neoplasms also have colonic neoplastic lesions.19 The majority of these lesions in the colon were right-sided and this finding was also seen in this case as the patient’s polyp was located in the ascending colon.19 Due to this association between appendiceal and colorectal neoplasia, the American Society of Colon and Rectal Surgeons strongly recommends that patients with appendiceal neoplasms or who are suspected of having them receive a colonoscopy.19
Additionally, perforation of an AMN, as was seen in this case, is a finding that should raise significant concern. Perforation of an AMN allows for the spread of malignant mucinous epithelial cells throughout the abdomen. The finding of extensive adhesions throughout the patient’s RLQ was unexpected as abdominal adhesions are most often seen in patients with a history of abdominal surgeries. Considering the lack of any prior abdominal surgeries in this patient, these adhesions were most likely the result of the spread and proliferation of malignant mucinous epithelial cells from the perforated AMN in the RLQ.20 The adhesiolysis performed in this case was thus not only important in order to visualize the appendix, but also for preventing future complications of abdominal adhesions such as bowel obstruction.20 Perforated AMN is also so concerning because it can potentially lead to pseudomyxoma peritonei—a condition in which malignant mucinous epithelial cells accumulate in the abdomen.21 Pseudomyxoma peritonei is extremely rare with an incidence of approximately 1 to 2 cases per million per year.22 Early recognition of AMNs and surgical referral are critically important as pseudomyxoma peritonei is difficult to treat, has a high rate of recurrence, and can be fatal.23
Lastly, this case highlights how findings of a ruptured appendix and/or mucin surrounding the appendix on imaging should warrant laparoscopy because only pathologic analysis of the appendix can definitively rule out AMNs. The utility of laparoscopic evaluation of the appendix is especially apparent as nonsurgical treatment of appendicitis using antibiotics is gaining favor for treating even complicated appendicitis.24 Appendicitis is much more common than AMNs. However, had the patient in this case only been given antibiotics for his suspected complicated appendicitis without any colonoscopy or appendectomy, the neoplasm in his appendix would have gone undetected and continued to grow, causing significant complications. The patient’s age at presentation in this case also necessitated laparoscopic evaluation of the appendix as the incidence of AMNs is highest among patients aged > 60 years.25 Additionally, because appendiceal inversion may be seen with AMNs,the patient’s inverted appendix seen during his colonoscopy was another compelling reason for laparoscopic evaluation of his appendix.6
Conclusions
AMNs can present with nonspecific symptoms or can be completely asymptomatic and are often found incidentally during colonoscopies or appendectomies for acute appendicitis. While it is true that AMNs have low metastatic potential and grow slowly, AMNs can rupture leading to pseudomyxoma peritonei or even cause bowel obstruction warranting timely identification and removal of these neoplasms. Laparoscopic evaluation in cases of ruptured appendices is critical not only for treatment, but also for determining the presence of a potential underlying appendiceal malignancy. Although AMNs are a rare pathology, physicians should still consider the possibility of these neoplasms even when imaging findings suggest appendicitis. Having AMNs as part of the differential diagnosis is especially necessary in cases, such as this one, in which the patient has appendiceal inversion, is aged > 50 years, and has concurrent colorectal neoplasms.
Appendiceal mucinous neoplasms (AMNs) are rare tumors of the appendix that can be asymptomatic or present with acute right lower quadrant (RLQ) pain mimicking appendicitis. Due to their potential to cause either no symptoms or nonspecific symptoms, such as abdominal pain, nausea, or vomiting, AMNs are often found incidentally during appendectomies or, even more rarely, colonoscopies. Most AMNs grow slowly and have little metastatic potential. However, due to potential complications, such as bowel obstruction and rupture, timely detection and removal of AMN is essential. We describe the case of a patient who appeared to have acute appendicitis complicated by rupture on imaging who was found instead to have a perforated low-grade AMN during surgery.
Case Presentation
A male patient aged 72 years with a history of type 2 diabetes mellitus, hypertension, and aortic stenosis, but no prior abdominal surgery, presented with a chief concern of generalized weakness. As part of the workup for his weakness, a computed tomography (CT) scan of the abdomen was performed which showed an RLQ phlegmon and mild fat stranding in the area. Imaging also revealed an asymptomatic gallstone measuring 1.5 cm with no evidence of cholecystitis. The patient had no fever and reported no abdominal pain, nausea, vomiting, or change in bowel habits. On physical examination, the patient’s abdomen was soft, nontender, and nondistended with normoactive bowel sounds and no rebound or guarding.
To manage the appendicitis, the patient started a 2-week course of amoxicillin clavulanate 875 mg twice daily and was instructed to schedule an interval appendectomy in the coming months. Four days later, during a follow-up with his primary care physician, he was found to be asymptomatic. However, at this visit his stool was found to be positive for occult blood. Given this finding and the lack of a previous colonoscopy, the patient underwent a colonoscopy, which revealed bulging at the appendiceal orifice, consistent with an inverted appendix. Portions of the appendix were biopsied (Figure 1). Histologic analysis of the appendiceal biopsies revealed no dysplasia or malignancy. The colonoscopy also revealed an 8-mm sessile polyp in the ascending colon which was resected, and histologic analysis of this polyp revealed a low-grade tubular adenoma. Additionally, a large angiodysplastic lesion was found in the ascending colon as well as external and medium-sized internal hemorrhoids.
Six weeks after the colonoscopy, the patient was taken to the operating room for a laparoscopic appendectomy. Upon entry of the abdomen, extensive adhesions throughout the RLQ were found which required adhesiolysis. A calcified fecalith adherent to the mesentery of the small intestine in the RLQ was also found and resected. After lysis of the adhesions, the appendix and fibrotic tissue surrounding it could be seen (Figure 2). The appendix was dilated and the tip showed perforation. During dissection of the appendix, clear gelatinous material was found coming from the appendiceal lumen as well as from the fibrotic tissue around the appendix. On postoperative day 1 the appendix was resected and the patient was discharged.
Histologic specimens of the appendix were notable for evidence of perforation and neoplasia leading to a diagnosis of low-grade AMN. The presence of atypical mucinous epithelial cells on the serosal surface of the appendix, confirmed with a positive pancytokeratin stain, provided histologic evidence of appendiceal perforation (Figure 3). The presence of nuclear atypia demonstrated that the appendix was involved by a neoplastic process. Additionally, attenuation of the normal appendiceal epithelium, evidence of a chronic process, further helped to differentiate the AMN from complicated appendicitis. The presence of mucin involving the serosa of the appendix led to the classification of this patient’s neoplasm as grade pT4a. Of note, histologic examination demonstrated that the surgical margins contained tumor cells.
Given the positive margins of the resected AMN and the relatively large size of the neoplasm, a laparoscopic right hemicolectomy was performed 2 months later. Although multiple adhesions were found in the terminal ileum, cecum, and ascending colon during the hemicolectomy, no mucinous lesions were observed grossly. Histologic analysis showed no residual neoplasm as well as no lymph node involvement. On postoperative day 3 the patient was discharged and had an uneventful recovery. At his first surveillance visit 6 months after his hemicolectomy, the patient appeared to be doing well and reported no abdominal pain, nausea, vomiting, change in bowel habits, or any blood in the stool.
Discussion
AMNs are rare tumors with an annual age-adjusted incidence of approximately 0.12 per 1,000,000 people.1 These neoplasms can present as acute or chronic abdominal pain, gastrointestinal bleeding, intestinal obstruction, or acute abdomen.2-4 Most AMNs, however, are asymptomatic and are usually found incidentally during appendectomies for appendicitis, and can even be found during colonoscopies,such as in this case.5,6
Low-grade AMNs are distinguished from appendiceal mucinous adenocarcinomas by their lack of wall invasion.7 Additionally, low-grade AMNs have a very good prognosis as even neoplasms that have spread outside of the appendix have a 5-year overall survival rate of 79 to 86%.8 These low-grade neoplasms also have extremely low rates of recurrence after resection.9 In contrast, appendiceal mucinous adenocarcinomas have a much worse prognosis with a 5-year overall survival rate of 53.6%.10
Treatment of AMNs depends on the extent of their spread. Neoplasms that are confined to the appendix can typically be treated with appendectomy alone, while those that have spread beyond the appendix may require cytoreductive surgery and chemotherapy, namely, hyperthermic intraperitoneal chemotherapy (HIPEC), in addition to appendectomy.11 Cases in which neoplasms are not confined to the appendix also require more frequent surveillance for recurrence as compared to appendix-restricted neoplasms.11
Appendiceal inversion is a rare finding in adults with an estimated prevalence of 0.01%.6 Not only is appendiceal inversion rare in and of itself, it is even more rarely found in combination with appendiceal neoplasms.6 Other causes of appendiceal inversion include intussusception, acute appendicitis, appendiceal nodule, or even iatrogenic due to appendectomy.12-14 While appendiceal inversion can be completely benign, because these morphological changes of the appendix can resemble a polyp, these lesions are often biopsied and/or resected.15 However, lesion resection may be quite problematic due to high risk of bleeding and perforation.15 In order to avoid the risks associated with resection of a potentially benign finding, biopsy should be performed prior to any attempted resection of inverted appendices.15
Another interesting aspect of this case is the finding of fecal occult blood. The differential for fecal occult blood is quite broad and the patient had multiple conditions that could have led to the finding of occult blood in his stool. Hemorrhoids can cause a positive result on a fecal occult blood test (FOBT) although this is relatively uncommon, and hemorrhoids are more likely to cause frank blood to be seen.16 The sessile polyp found in the patient’s colon may also have caused the FOBT to be positive. This patient was also found to have an angiodysplasia (a finding that is associated with aortic stenosis, which this patient has a history of) which can also cause gastrointestinal bleeding.17 Lastly, AMNs may also cause gastrointestinal bleeding and thus a positive FOBT, although bleeding is a relatively uncommon presentation of AMNs, especially those that are low-grade as in this case.18
This case also highlights the association between appendiceal neoplasms and colonic neoplastic lesions. Patients with appendiceal neoplasms are more likely to have colonic neoplastic lesions than patients without appendiceal neoplasms.19 Studies have found that approximately 13 to 42% of patients with appendiceal neoplasms also have colonic neoplastic lesions.19 The majority of these lesions in the colon were right-sided and this finding was also seen in this case as the patient’s polyp was located in the ascending colon.19 Due to this association between appendiceal and colorectal neoplasia, the American Society of Colon and Rectal Surgeons strongly recommends that patients with appendiceal neoplasms or who are suspected of having them receive a colonoscopy.19
Additionally, perforation of an AMN, as was seen in this case, is a finding that should raise significant concern. Perforation of an AMN allows for the spread of malignant mucinous epithelial cells throughout the abdomen. The finding of extensive adhesions throughout the patient’s RLQ was unexpected as abdominal adhesions are most often seen in patients with a history of abdominal surgeries. Considering the lack of any prior abdominal surgeries in this patient, these adhesions were most likely the result of the spread and proliferation of malignant mucinous epithelial cells from the perforated AMN in the RLQ.20 The adhesiolysis performed in this case was thus not only important in order to visualize the appendix, but also for preventing future complications of abdominal adhesions such as bowel obstruction.20 Perforated AMN is also so concerning because it can potentially lead to pseudomyxoma peritonei—a condition in which malignant mucinous epithelial cells accumulate in the abdomen.21 Pseudomyxoma peritonei is extremely rare with an incidence of approximately 1 to 2 cases per million per year.22 Early recognition of AMNs and surgical referral are critically important as pseudomyxoma peritonei is difficult to treat, has a high rate of recurrence, and can be fatal.23
Lastly, this case highlights how findings of a ruptured appendix and/or mucin surrounding the appendix on imaging should warrant laparoscopy because only pathologic analysis of the appendix can definitively rule out AMNs. The utility of laparoscopic evaluation of the appendix is especially apparent as nonsurgical treatment of appendicitis using antibiotics is gaining favor for treating even complicated appendicitis.24 Appendicitis is much more common than AMNs. However, had the patient in this case only been given antibiotics for his suspected complicated appendicitis without any colonoscopy or appendectomy, the neoplasm in his appendix would have gone undetected and continued to grow, causing significant complications. The patient’s age at presentation in this case also necessitated laparoscopic evaluation of the appendix as the incidence of AMNs is highest among patients aged > 60 years.25 Additionally, because appendiceal inversion may be seen with AMNs,the patient’s inverted appendix seen during his colonoscopy was another compelling reason for laparoscopic evaluation of his appendix.6
Conclusions
AMNs can present with nonspecific symptoms or can be completely asymptomatic and are often found incidentally during colonoscopies or appendectomies for acute appendicitis. While it is true that AMNs have low metastatic potential and grow slowly, AMNs can rupture leading to pseudomyxoma peritonei or even cause bowel obstruction warranting timely identification and removal of these neoplasms. Laparoscopic evaluation in cases of ruptured appendices is critical not only for treatment, but also for determining the presence of a potential underlying appendiceal malignancy. Although AMNs are a rare pathology, physicians should still consider the possibility of these neoplasms even when imaging findings suggest appendicitis. Having AMNs as part of the differential diagnosis is especially necessary in cases, such as this one, in which the patient has appendiceal inversion, is aged > 50 years, and has concurrent colorectal neoplasms.
1. Shaib WL, Goodman M, Chen Z, et al. Incidence and survival of appendiceal mucinous neoplasms: a SEER analysis. Am J Clin Oncol. 2017;40(6):569-573. doi:10.1097/COC.0000000000000210
2. Kehagias I, Zygomalas A, Markopoulos G, Papandreou T, Kraniotis P. Diagnosis and treatment of mucinous appendiceal neoplasm presented as acute appendicitis. Case Rep Oncol Med. 2016;3:1-6. doi:10.1155/2016/2161952
3. Karatas M, Simsek C, Gunay S, et al. Acute lower gastrointestinal bleeding due to low-grade mucinous neoplasm of appendix. Acta Chir Belg. 2020;120(4):1-4. doi:10.1080/00015458.2020.1860397
4. Mourad FH, Hussein M, Bahlawan M, Haddad M, Tawil A. Intestinal obstruction secondary to appendiceal mucocele. Dig Dis Sci. 1999;44(8):1594-1599. doi:10.1023/a:1026615010989
5. Benabe SH, Leeman R, Brady AC, Hirzel A, Langshaw AH. Low-grade appendiceal mucinous neoplasm in an adolescent patient with untreated Crohn’s disease. ACG Case Reports J. 2020;7(3). doi:10.14309/crj.0000000000000338
6. Liu X, Liu G, Liu Y, et al. Complete appendiceal inversion with local high-grade intraepithelial neoplasia in an adult female: A case report. BMC Surg. 2019;19(1). doi:10.1186/s12893-019-0632-3
7. Gündog˘ar ÖS, Kımılog˘lu ES, Komut NS, et al. The evaluation of appendiceal mucinous neoplasms with a new classification system. Turk J Gastroenterol. 2018;29(5):532-542. doi:10.5152/tjg.2018.17605
8. Misdraji J, Yantiss RK, Graeme-Cook FM, Balis UJ, Young RH. Appendiceal mucinous neoplasms: a clinicopathologic analysis of 107 cases. Am J Surg Pathol. 2003;27(8):1089-1103. doi:10.1097/00000478-200308000-00006
9. Pai RK, Beck AH, Norton JA, Longacre TA. Appendiceal mucinous neoplasms: clinicopathologic study of 116 cases with analysis of factors predicting recurrence. Am J Surg Pathol. 2009;33(10):1425-1439. doi:10.1097/PAS.0b013e3181af6067
10. Asare EA, Compton CC, Hanna NN, et al. The impact of stage, grade, and mucinous histology on the efficacy of systemic chemotherapy in adenocarcinomas of the appendix: analysis of the National Cancer Data Base. Cancer. 2015;122(2):213-221. doi:10.1002/cncr.29744
11. Shaib WL, Assi R, Shamseddine A, et al. Appendiceal mucinous neoplasms: diagnosis and management. Oncologist. 2018;23(1):137. doi:10.1634/theoncologist.2017-0081erratum
12. Tran C, Sakioka J, Nguyen E, Beutler BD, Hsu J. An inverted appendix found on routine colonoscopy: a case report with discussion of imaging findings. Radiol Case Rep. 2019;14(8):952-955. doi:10.1016/j.radcr.2019.05.022
13. Shafi A, Azab M. A case of everted appendix with benign appendiceal nodule masquerading as appendiceal mucocele: a case report. Am J Gastroenterol. 2018;113:S1436. doi:10.14309/00000434-201810001-02585
14. Pokhrel B, Chang M, Anand G, Savides T, Fehmi S. Appendiceal mucinous neoplasm in an inverted appendix found on prior colonoscopy. VideoGIE. 2020;5(1):34-36. doi:10.1016/j.vgie.2019.09.013
15. Johnson EK, Arcila ME, Steele SR. Appendiceal inversion: a diagnostic and therapeutic dilemma. JSLS. 2009;13(1):92-95.
16. van Turenhout ST, Oort FA, sive Droste JST, et al. Hemorrhoids detected at colonoscopy: an infrequent cause of false-positive fecal immunochemical test results. Gastrointest Endosc. 2012;76(1):136-143. doi:10.1016/j.gie.2012.03.169
17. Hudzik B, Wilczek K, Gasior M. Heyde syndrome: Gastrointestinal bleeding and aortic stenosis. CMAJ. 2016;188(2):135-138. doi:10.1503/cmaj.150194
18. Leonards LM, Pahwa A, Patel MK, Petersen J, Nguyen MJ, Jude CM. Neoplasms of the appendix: pictorial review with clinical and pathologic correlation. RadioGraphics. 2017;37(4):1059-1083. doi:10.1148/rg.2017160150
19. Glasgow SC, Gaertner W, Stewart D, et al. The American Society of Colon and Rectal Surgeons, clinical practice guidelines for the management of appendiceal neoplasms. Dis Colon Rectum. 2019;62(12):1425-1438. doi:10.1097/DCR.0000000000001530
20. Panagopoulos P, Tsokaki T, Misiakos E, et al. Low-grade appendiceal mucinous neoplasm presenting as an adnexal mass. Case Reports in Obstetrics and Gynecology. 2017;2017:1-3. doi:10.1155/2017/7165321
21. Ramaswamy V. Pathology of mucinous appendiceal tumors and pseudomyxoma peritonei. Indian J Surg Oncol. 2016;7(2):258-267. doi:10.1007/s13193-016-0516-2.
22. Bevan KE, Mohamed F, Moran BJ. Pseudomyxoma peritonei. World J Gastrointest Oncol. 2010;2(1):44-50. doi:10.4251/wjgo.v2.i1.44
23. Mercier F, Dagbert F, Pocard M, et al. Recurrence of pseudomyxoma peritonei after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. BJS Open. 2018;3(2):195-202. doi:10.1002/bjs5.97
24. David A, Dodgion C, Eddine SBZ, Davila D, Webb TP, Trevino CM. Perforated appendicitis: Short duration antibiotics are noninferior to traditional long duration antibiotics. Surgery. 2020;167(2):475-477. doi:10.1016/j.surg.2019.08.007
25. Raijman I, Leong S, Hassaram S, Marcon NE. Appendiceal mucocele: Endoscopic appearance. Endoscopy. 1994;26(3):326-328. doi:10.1055/s-2007-1008979
1. Shaib WL, Goodman M, Chen Z, et al. Incidence and survival of appendiceal mucinous neoplasms: a SEER analysis. Am J Clin Oncol. 2017;40(6):569-573. doi:10.1097/COC.0000000000000210
2. Kehagias I, Zygomalas A, Markopoulos G, Papandreou T, Kraniotis P. Diagnosis and treatment of mucinous appendiceal neoplasm presented as acute appendicitis. Case Rep Oncol Med. 2016;3:1-6. doi:10.1155/2016/2161952
3. Karatas M, Simsek C, Gunay S, et al. Acute lower gastrointestinal bleeding due to low-grade mucinous neoplasm of appendix. Acta Chir Belg. 2020;120(4):1-4. doi:10.1080/00015458.2020.1860397
4. Mourad FH, Hussein M, Bahlawan M, Haddad M, Tawil A. Intestinal obstruction secondary to appendiceal mucocele. Dig Dis Sci. 1999;44(8):1594-1599. doi:10.1023/a:1026615010989
5. Benabe SH, Leeman R, Brady AC, Hirzel A, Langshaw AH. Low-grade appendiceal mucinous neoplasm in an adolescent patient with untreated Crohn’s disease. ACG Case Reports J. 2020;7(3). doi:10.14309/crj.0000000000000338
6. Liu X, Liu G, Liu Y, et al. Complete appendiceal inversion with local high-grade intraepithelial neoplasia in an adult female: A case report. BMC Surg. 2019;19(1). doi:10.1186/s12893-019-0632-3
7. Gündog˘ar ÖS, Kımılog˘lu ES, Komut NS, et al. The evaluation of appendiceal mucinous neoplasms with a new classification system. Turk J Gastroenterol. 2018;29(5):532-542. doi:10.5152/tjg.2018.17605
8. Misdraji J, Yantiss RK, Graeme-Cook FM, Balis UJ, Young RH. Appendiceal mucinous neoplasms: a clinicopathologic analysis of 107 cases. Am J Surg Pathol. 2003;27(8):1089-1103. doi:10.1097/00000478-200308000-00006
9. Pai RK, Beck AH, Norton JA, Longacre TA. Appendiceal mucinous neoplasms: clinicopathologic study of 116 cases with analysis of factors predicting recurrence. Am J Surg Pathol. 2009;33(10):1425-1439. doi:10.1097/PAS.0b013e3181af6067
10. Asare EA, Compton CC, Hanna NN, et al. The impact of stage, grade, and mucinous histology on the efficacy of systemic chemotherapy in adenocarcinomas of the appendix: analysis of the National Cancer Data Base. Cancer. 2015;122(2):213-221. doi:10.1002/cncr.29744
11. Shaib WL, Assi R, Shamseddine A, et al. Appendiceal mucinous neoplasms: diagnosis and management. Oncologist. 2018;23(1):137. doi:10.1634/theoncologist.2017-0081erratum
12. Tran C, Sakioka J, Nguyen E, Beutler BD, Hsu J. An inverted appendix found on routine colonoscopy: a case report with discussion of imaging findings. Radiol Case Rep. 2019;14(8):952-955. doi:10.1016/j.radcr.2019.05.022
13. Shafi A, Azab M. A case of everted appendix with benign appendiceal nodule masquerading as appendiceal mucocele: a case report. Am J Gastroenterol. 2018;113:S1436. doi:10.14309/00000434-201810001-02585
14. Pokhrel B, Chang M, Anand G, Savides T, Fehmi S. Appendiceal mucinous neoplasm in an inverted appendix found on prior colonoscopy. VideoGIE. 2020;5(1):34-36. doi:10.1016/j.vgie.2019.09.013
15. Johnson EK, Arcila ME, Steele SR. Appendiceal inversion: a diagnostic and therapeutic dilemma. JSLS. 2009;13(1):92-95.
16. van Turenhout ST, Oort FA, sive Droste JST, et al. Hemorrhoids detected at colonoscopy: an infrequent cause of false-positive fecal immunochemical test results. Gastrointest Endosc. 2012;76(1):136-143. doi:10.1016/j.gie.2012.03.169
17. Hudzik B, Wilczek K, Gasior M. Heyde syndrome: Gastrointestinal bleeding and aortic stenosis. CMAJ. 2016;188(2):135-138. doi:10.1503/cmaj.150194
18. Leonards LM, Pahwa A, Patel MK, Petersen J, Nguyen MJ, Jude CM. Neoplasms of the appendix: pictorial review with clinical and pathologic correlation. RadioGraphics. 2017;37(4):1059-1083. doi:10.1148/rg.2017160150
19. Glasgow SC, Gaertner W, Stewart D, et al. The American Society of Colon and Rectal Surgeons, clinical practice guidelines for the management of appendiceal neoplasms. Dis Colon Rectum. 2019;62(12):1425-1438. doi:10.1097/DCR.0000000000001530
20. Panagopoulos P, Tsokaki T, Misiakos E, et al. Low-grade appendiceal mucinous neoplasm presenting as an adnexal mass. Case Reports in Obstetrics and Gynecology. 2017;2017:1-3. doi:10.1155/2017/7165321
21. Ramaswamy V. Pathology of mucinous appendiceal tumors and pseudomyxoma peritonei. Indian J Surg Oncol. 2016;7(2):258-267. doi:10.1007/s13193-016-0516-2.
22. Bevan KE, Mohamed F, Moran BJ. Pseudomyxoma peritonei. World J Gastrointest Oncol. 2010;2(1):44-50. doi:10.4251/wjgo.v2.i1.44
23. Mercier F, Dagbert F, Pocard M, et al. Recurrence of pseudomyxoma peritonei after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. BJS Open. 2018;3(2):195-202. doi:10.1002/bjs5.97
24. David A, Dodgion C, Eddine SBZ, Davila D, Webb TP, Trevino CM. Perforated appendicitis: Short duration antibiotics are noninferior to traditional long duration antibiotics. Surgery. 2020;167(2):475-477. doi:10.1016/j.surg.2019.08.007
25. Raijman I, Leong S, Hassaram S, Marcon NE. Appendiceal mucocele: Endoscopic appearance. Endoscopy. 1994;26(3):326-328. doi:10.1055/s-2007-1008979
Gadolinium Deposition Disease: A Case Report and the Prevalence of Enhanced MRI Procedures Within the Veterans Health Administration
Gadolinium (Gd)-based contrast agents are frequently used in health care for enhancing magnetic resonance image (MRI) signals at low concentrations. Contrary to popular opinion, this widely used heavy metal is not biologically inert. Once notable for its safety profile, there is mounting evidence for Gd deposition in various organ systems of the body, even in those with normal renal function. A large knowledge gap remains concerning the potential harms of Gd deposition and the factors determining its elimination from the body. However, the findings of deposited Gd throughout various organs and their intracellular compartments even years after the initial exposure have been established. Here, we describe a case of a Vietnam-era veteran whose presentation, clinical, and laboratory findings were consistent within the spectrum of Gd deposition disease.
Case Presentation
A Vietnam-era veteran aged > 70 years presented for evaluation of Gd-based contrast agent–induced chronic multisymptomatic illness His medical history was significant for chronic low back pain, chronic hypertension, type 2 diabetes mellitus, and hypogonadism. Surgical history was notable for back surgery (24 years prior), laminectomy (2 years prior), shoulder replacement (2 years prior), and an epidural complicated by a hematoma (1 year prior). His presenting concerns included a painful and pruritic rash that worsened with showering, pain originating at the right Achilles tendon with migration to the knee, and shoulder pain. His symptoms started shortly after receiving multiple exposures to Gd-based contrast agents to enhance MRIs during his clinical care (Omniscan 20 mL, Omniscan 20 mL, and Gadovist 10 mL, administered 578, 565, and 496 days prior to the clinic visit, respectively). New onset headaches coincided with the timeline of symptom onset, in addition to hoarseness and liberation of an “oily substance” from the skin. More than one year prior to this clinic visit, he was considered for having polymyalgia rheumatica given the ambiguity of symptoms. Functional status remained impaired despite treatment with prednisone and methotrexate.
The patient’s military service was in the mid-1960s. He was deployed to Japan and had no knowledge of an Agent Orange exposure. His tobacco history was distant, and he reported no tattoos, prior transfusions, or occupational metal exposure
Clinical Findings
The patient was afebrile, normotensive (146/88 mmHg), and normocardic. His weight was 100 kg. He was well nourished and in no acute distress. The thought process was attentive, and his affect pleasant. Ocular examination was notable for arcus senilus. The fundoscopic examination was limited on the left, but there was no neovascularization on the right. Jugular venous pulsation was normal at 8 cm. Right ventricular impulse was slightly hyperdynamic, the rhythm was regular, and there was no abnormal splitting of S2. A soft-grade I/VI crescendo/decrescendo murmur was auscultated along the apex. Radial pulses were 2/2. He was not in respiratory distress, with equally resonant fields bilaterally. Lung sounds were clear bilaterally. A papular, erythematous rash was present in a general distribution over the chest, with few telangiectasias and some varicosity along his left arm. The skin had normal elasticity, although the skin of the hands and legs was papyraceous.
Gd levels were measured in the blood and urine (Table 1). Gd was detectable in the skin (0.2 µg/g) nearly 400 days after the last exposure. Gd was still detectable in the patient’s blood and urine (0.2 ng/mL and 0.5 µg/24 h, respectively) more than 3 years after his last exposure.
Discussion
In the United States, there are 40.44 MRI units per million people and 40 million MRIs are conducted annually. From 30 to 50% of these are enhanced with Gd-based contrast agents. In the past 30 years, there have been > 450 million contrast-enhanced MRI procedures.1
Gd is a rare earth metal. Among commercially available elements Gd has exceptional properties for enhancing MRI signals at low concentrations.1 The nonphysiologic metal is detoxified by chelation with proprietary multidentate formulations that enhance (primarily renal) elimination while retaining the paramagnetic and chemical properties for imaging. Gd exposure was found to be associated to iatrogenic nephrogenic systemic fibrosis in 2006 and later confirmed via multiple systematic reviews.2 Gd is retained in every vital organ after exposure.3 Gd-based contrast agents stimulate bone marrow–derived fibrocytes in mediating fibrosis, and bone marrow develop a memory of prior contrast exposure (Figure 1).4-6 Systemic fibrosis is mediated by the monocyte chemoattractant protein 1/C-C chemokine receptor 2.6,7 Even in the setting of normal renal function, Gd-based contrast induces the formation of Gd-rich nanoparticles in the skin and kidney.7,8 Far from being inert, Gd-based contrast agents induce systemic metabolic changes such as hypertriglyceridemia, elevations in low-density lipoprotein cholesterol, insulin resistance, and the Warburg effect (glycolytic/energy switching) in the renal cortex concomitant with profound mitochondrial abnormalities.8
We have discovered that the rate of Gd-enhanced procedures has increased immensely within the Veterans Health Administration (VHA) system in a subset of patients with designated kidney disease (Table 2). Although a substantial number of procedures are dedicated to head and brain imaging within the VHA, the indications for Gd-enhanced diagnoses (eg, cardiac) are increasing (Figure 2).
Retention of Gd can be modeled as a function of time (t) by the half-lives of the fast, intermediate, and slow phases of elimination (Ta, Tb, and Tc, respectively):9
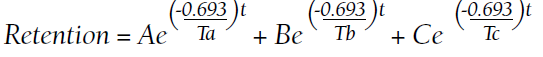
A, B, and C are the proportions (adding to 100%) that represent each of the compartments:
Numerous patients with normal renal function developed similar or novel symptoms that have been attributed to Gd concomitant with detectable urinary Gd years after exposure.11 Gd-based contrast agents are increasingly associated with cutaneous abnormalities even outside of nephrogenic systemic fibrosis. Gd-associated plaques develop in patients without kidney disease—these range from asymptomatic, pruritic, to burning.13 Histologic specimens reveal CD68 and factor XIIIa–positive spindle-shaped myeloid cells (the same mediators of iatrogenic systemic fibrosis) or CD34-positive cells. CD68 and factor XIIIa are distinctive for histologic specimens from patients with systemic fibrosis, and these markers have been detected in our preclinical models that demonstrated that bone marrow–derived cells are involved in mediating fibrosis.3,4,14-19 Similarly, CD34-positive cells have been historically associated with systemic fibrosis lesions.15,16,18-23 Plump osteocyte-appearing cells have also been noted (note that extraosseous metaplasia makes the histologic diagnosis of systemic fibrosis).14 Nephrogenic systemic fibrosis is an iatrogenic disease that can manifest years after exposure to Gd.5 Gd induces the recruitment of bone marrow–derived cells to the affected sites.4
The VA Health Service Research and Development Evidence Synthesis Program reviewed the safety of Gd-based contrast agents in patients with impaired kidney function.24,25 The group found only a single study of Gd and veterans. “Awareness and concern are growing about the long-term deposition of gadolinium in [the] brain and other tissues among patients with normal kidney function,” according to Lunyera and colleagues.25 The largest knowledge gap was that a comprehensive review “of all potential harms associated with gadolinium exposure” was not addressed. Furthermore, the group advised “caution in the use of [Gd-based contrast agents] in patients with severely impaired kidney function and acute kidney injury remains prudent, because the exact clinical factors contributing to [nephrogenic systemic fibrosis] risk in these subpopulations are still unknown.”25
Gd-based contrast agents—contrary to a widely held misconception—are not biologically inert.1 Gd-based contrast agents have a long history of association with acute renal injury. We have demonstrated that systemic treatment with MRI contrast agents leads to vacuolization of the proximal tubule and tubular injury.7,8 Kidney injury may be mediated by the generation of reactive oxygen species from NADPH oxidase 4 (Nox4).26
Gd retention, Gd-induced multisymptomatic illnesses, Gd-associated plaques, Gd-induced neurotoxicity, and nephrogenic systemic fibrosis are part of a continuum (with Gd as the common thread)—a theme of the September 8, 2017, US Food and Drug Administration (FDA) Medical Imaging Drugs Advisory Committee meeting.27 Patients, patient advocacy groups, and regulating agencies are concerned about long-term retention of a nonphysiologic rare earth element such as Gd.28-30 A patient advocacy group, The Lighthouse Project, collected information from patients linking the last date of Gd-based contrast agent exposure and urinary Gd.11 Data from their report suggest that the rate constants (valuable for the elimination equation above) are obtainable from 24-hour urine collections. Conceptually, Gd-induced diseases may represent a continuum that results from the retention of a nonphysiologic, toxic heavy rare earth metal.
As a heavy metal, Gd is not a natural physiologic trace element. Similar to numerous nonphysiologic metals, Gd is toxic. Inhaled Gd oxide (Gd2O3) dust leads to a number of time-dependent pathologies. Animal lung studies demonstrate reduced elasticity, enlarged cells, thickened lung walls, and recruitment of immune cells.31 Symptoms of acute IV Gd toxicity include decreased respiration, lethargy, abdominal cramps, and diarrhea.32 Pharmacologically, Gd concentrates in the liver and kidney and accumulates in the bone.32 Animals demonstrate intestinal depression and low blood pressure in response to Gd and, with higher doses, cardiovascular collapse.32 IV Gd chloride leads to metal deposition in the small blood vessels diffusely throughout the body, particularly in the lung and kidney and the metal is absorbed by the scavenging white blood cells.33 Gd chloride induces severe damage to the liver, spleen, and the digestive tract.33 Furthermore, this form of the toxicant metal markedly impacted functions associated with bleeding and clotting, ie, decreased platelet numbers and an increase in the laboratory-measured coagulation parameters.33 Semelka and colleagues have characterized chronic symptoms attributed to Gd-based contrast agents (not limited to chronic pain, headache, bone pain, skin thickening, and clouded mentation).34,35 Because Gd-induced conditions are underrecognized and ill-defined, disinherited patients often resort to untested (and potentially dangerous) chelation therapies.36
This patient presented with numerous symptoms that arose after Gd exposure. It is well established that Gd-based contrast agents (of any class) are retained in multiple organs (including the brain), for months to years. Gd-based contrast agents enter the cerebrospinal fluid within minutes of IV administration.37 Gd was found in the cerebrospinal fluid 9 months after administration in a case presented to the FDA Medical Imaging Drugs Advisory Committee.38 We know from intentional and accidental intrathecal administrations that Gd-based contrast agents are neurotoxic.39 Runge and colleagues demonstrated that Gd-based contrast agents exert mitochondrial toxicity in cultured neurons in vitro.40 McDonald and his team found Gd-rich nanoparticles within the brain neurons (cytoplasm and nuclei) from patients exposed to MRI contrast in the normal course of care.41 These nanoparticles are similar to what we have found in rodent models of Gd-induced disease.7,8,42
Prolonged elimination of Gd after MRI contrast administration (months to years) may be universal.10 Gd compartmentalizes into leukocytes and erythrocytes and into the cerebrospinal fluid within minutes.37,43 Patients with multisymptomatic illnesses attributed to Gd (Gd deposition disease) have perturbations in cytokine levels, many inflammatory.44,45 The results are concerning: Gd is retained intracellularly in vital organs, including brain neurons. It is inarguable that Gd is an alien, nonphysiologic element. With mounting evidence that Gd retention has clinical consequences, patients should be provided proper informed consent. Complications of renal insufficiency (ie, hyperkalemia, hyperphosphatemia, renal osteodystrophy, hyponatremia, anemia, immunosuppression, etc) follow a smooth, curvilinear slope as the true (not estimated) glomerular filtration declines; the worst iatrogenic complication from Gd—systemic fibrosis—is likely no different.
Patient Perspective
“Seems like it’s one thing after another. My family doctor said that once I had the gadolinium exposures, I have had problems ever since that I don’t recover from.” This includes chronic numbness from the rectum to the bilateral lower extremities and an indolent worsening kidney function; “I have already developed stage 3B chronic kidney disease.” Similar to many suffering with gadolinium retention, the patient was concerned about the long-term consequences. Gadolinium “is a toxic metal that is going through my body for 4 years. That has to be a problem. How come we don’t have that answer?” Clinician ignorance of Gd-induced complications and long-term retention is frustrating. “Not one of my doctors has taken gadolinium retention seriously. Where else are patients supposed to go?”
Conclusions
Health care professionals should be considering subclinical manifestations of nephrogenic systemic fibrosis or open to considering that intracellular neuronal retention of Gd may correlate with symptoms arising after MRI contrast exposures. The science concerning the mechanisms of how Gd exerts its pathologic effects is lagging behind the commercialization of enhancing Gd elimination (ie, chelation therapies) and other untested remedies. Practitioners need to acknowledge the unknown potential consequences of Gd and listen to patients who suspect chronic adverse effects.
1. Leyba K, Wagner B. Gadolinium-based contrast agents: why nephrologists need to be concerned. Curr Opin Nephrol Hypertens. 2019;28(2):154-162. doi:10.1097/MNH.0000000000000475
2. Grobner T. Gadolinium—a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis?. Nephrol Dial Transplant. 2006;21(4):1104-1108. doi:10.1093/ndt/gfk062
3. Do C, Barnes JL, Tan C, Wagner B. Type of MRI contrast, tissue gadolinium, and fibrosis. Am J Physiol Renal Physiol. 2014;307(7):F844-F855. doi:10.1152/ajprenal.00379.2014
4. Wagner B, Tan C, Barnes JL, et al. Nephrogenic systemic fibrosis: evidence for oxidative stress and bone marrow-derived fibrocytes in skin, liver, and heart lesions using a 5/6 nephrectomy rodent model. Am J Pathol. 2012;181(6):1941-1952. doi:10.1016/j.ajpath.2012.08.026
5. Wagner B, Drel V, Gorin Y. Pathophysiology of gadolinium-associated systemic fibrosis. Am J Physiol Renal Physiol. 2016;311(1):F1-F11. doi:10.1152/ajprenal.00166.2016
6. Drel VR, Tan C, Barnes JL, Gorin Y, Lee DY, Wagner B. Centrality of bone marrow in the severity of gadolinium-based contrast-induced systemic fibrosis. FASEB J. 2016;30(9):3026-3038. doi:10.1096/fj.201500188R
7. Do C, Drel V, Tan C, Lee D, Wagner B. Nephrogenic systemic fibrosis is mediated by myeloid C-C chemokine receptor 2. J Invest Dermatol. 2019;139(10):2134-2143.e2. doi:10.1016/j.jid.2019.03.1145
8. Do C, Ford B, Lee DY, Tan C, Escobar P, Wagner B. Gadolinium-based contrast agents: stimulators of myeloid-induced renal fibrosis and major metabolic disruptors. Toxicol Appl Pharmacol. 2019;375:32-45. doi:10.1016/j.taap.2019.05.009
9. Hirano S, Suzuki KT. Exposure, metabolism, and toxicity of rare earths and related compounds. Environ Health Perspect. 1996;104(suppl 1):85-95. doi:10.1289/ehp.96104s185
10. Alwasiyah D, Murphy C, Jannetto P, Hogg M, Beuhler MC. Urinary gadolinium levels after contrast-enhanced MRI in individuals with normal renal function: a pilot study. J Med Toxicol. 2019;15(2):121-127. doi:10.1007/s13181-018-0693-1
11. Williams S, Grimm H. gadolinium toxicity: shedding light on the effects of retained gadolinium from contrast MRI. Accessed April 11, 2022. https://gdtoxicity.files.wordpress.com/2018/12/gadolinium-clearance-times-for-135-contrast-mri-cases-final-v1-1.pdf
12. DeBevits JJ, Reshma M, Bageac D, et al. Gray matter nucleus hyperintensity after monthly triple-dose gadopentetate dimeglumine with long-term magnetic resonance imaging. Invest Radiol. 2020;55(10):629-635. doi:10.1097/RLI.0000000000000663
13. Gathings RM, Reddy R, Santa Cruz D, Brodell RT. Gadolinium-associated plaques: a new, distinctive clinical entity. JAMA Dermatol. 2015;151(3):316-319. doi:10.1001/jamadermatol.2014.2660
14. Girardi M, Kay J, Elston DM, Leboit PE, Abu-Alfa A, Cowper SE. Nephrogenic systemic fibrosis: clinicopathological definition and workup recommendations. J Am Acad Dermatol. 2011;65(6):1095-1106 e7. doi:10.1016/j.jaad.2010.08.041
15. Daram SR, Cortese CM, Bastani B. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis: report of a new case with literature review. Am J Kidney Dis. 2005;46(4):754-759. doi:10.1053/j.ajkd.2005.06.024
16. Ortonne N, Lipsker D, Chantrel F, Boehm N, Grosshans E, Cribier B. Presence of CD45RO+ CD34+ cells with collagen synthesis activity in nephrogenic fibrosing dermopathy: a new pathogenic hypothesis. Br J Dermatol. 2004;150(5):1050-1052. doi:10.1111/j.1365-2133.2004.05900.x
17. Mendoza FA, Artlett CM, Sandorfi N, Latinis K, Piera-Velazquez S, Jimenez SA. Description of 12 cases of nephrogenic fibrosing dermopathy and review of the literature. Semin Arthritis Rheum. 2006;35(4):238-49. doi:10.1016/j.semarthrit.2005.08.002
18. Lewis KG, Lester BW, Pan TD, Robinson-Bostom L. Nephrogenic fibrosing dermopathyand calciphylaxis with pseudoxanthoma elasticum-like changes. J Cutan Pathol. 2006;33(10):695-700. doi:10.1111/j.1600-0560.2006.00490.x
19. Gibson SE, Farver CF, Prayson RA. Multiorgan involvement in nephrogenic fibrosing dermopathy: an autopsy case and review of the literature. Arch Pathol Lab Med. 2006;130(2):209-212. doi:10.5858/2006-130-209-MIINFD
20. Cassis TB, Jackson JM, Sonnier GB, Callen JP. Nephrogenic fibrosing dermopathy in a patient with acute renal failure never requiring dialysis. Int J Dermatol. 2006;45(1):56-59. doi:10.1111/j.1365-4632.2005.02701.x
21. Kucher C, Steere J, Elenitsas R, Siegel DL, Xu X. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis with diaphragmatic involvement in a patient with respiratory failure. J Am Acad Dermatol. 2006;54(suppl 2):S31-S34. doi:10.1016/j.jaad.2005.04.024
22. Sanyal S, Marckmann P, Scherer S, Abraham JL. Multiorgan gadolinium (Gd) deposition and fibrosis in a patient with nephrogenic systemic fibrosis—an autopsy-based review. Nephrol Dial Transplant. 2011;26(11):3616-3626. doi:10.1093/ndt/gfr085
23. Kucher C, Xu X, Pasha T, Elenitsas R. Histopathologic comparison of nephrogenic fibrosing dermopathy and scleromyxedema. J Cutan Pathol. 2005;32(7):484-490. doi:10.1111/j.0303-6987.2005.00365.x
24. Goldstein KM, Lunyera J, Mohottige D, et al. Risk of Nephrogenic Systemic Fibrosis after Exposure to Newer Gadolinium Agents. Washington (DC): Department of Veterans Affairs (US); October 2019. https://www.ncbi.nlm.nih.gov/books/NBK559376/25. Lunyera J, Mohottige D, Alexopoulos AS, et al. Risk for nephrogenic systemic fibrosis after exposure to newer gadolinium agents: a systematic review. Ann Intern Med. 2020;173(2):110-119. doi:10.7326/M20-0299
26. Bruno F, DeAguero J, Do C, et al. Overlapping roles of NADPH Oxidase 4 (Nox4) for diabetic and gadolinium-based contrast agent-induced systemic fibrosis. Am J Physiol Renal Physiol. 2021;320(4):F617-F627. doi:10.1152/ajprenal.00456.2020
27. Wagner B. The pathophysiology and retention of gadolinium. United States Food & Drug Administration Medical Imaging Drugs Advisory Committee. 2017:1-23. https://www.fda.gov/advisory-committees/medical-imaging-drugs-advisory-committee/2017-meeting-materials-medical-imaging-drugs-advisory-committee?msclkid=6b5764ccbaa611ec95e35dddf8db57af
28. Runge VM. Critical questions regarding gadolinium deposition in the brain and body after injections of the gadolinium-based contrast agents, safety, and clinical recommendations in consideration of the EMA’s pharmacovigilance and risk assessment committee recommendation for suspension of the marketing authorizations for 4 linear agents. Invest Radiol. 2017;52(6):317-323. doi:10.1097/RLI.0000000000000374
29. Wagner B. Scared to the marrow: pitfalls and pearls in renal imaging. Adv Chronic Kidney Dis. 2017;24(3):136-137. doi:10.1053/j.ackd.2017.03.008
30. US Food and Drug Administration. Transcript for the September 8, 2017 Meeting of the Medical Imaging Drugs Advisory Committee (MIDAC). September 8, 2017. Accessed April 11, 2022. https://www.fda.gov/media/108935/download
31. Abel M, Talbot RB. Gadolinium oxide inhalation by guinea pigs: a correlative functional and histopathologic study. J Pharmacol Exp Ther. 1967;157(1):207-213.
32. Haley TJ, Raymond K, Komesu N, Upham HC. Toxicological and pharmacological effects of gadolinium and samarium chlorides. Br J Pharmacol Chemother. 1961;17(3):526-532. doi:10.1111/j.1476-5381.1961.tb01139.x

33. Spencer AJ, Wilson SA, Batchelor J, Reid A, Rees J, Harpur E. Gadolinium chloride toxicity in the rat. Toxicol Pathol. 1997;25(3):245-255. doi:10.1177/019262339702500301
34. Semelka RC, Ramalho M, AlObaidy M, Ramalho J. Gadolinium in humans: a family of disorders. AJR Am J Roentgenol. 2016;207(2):229-233. doi:10.2214/AJR.15.15842
35. Semelka RC, Ramalho M. Physicians with self-diagnosed gadolinium deposition disease: a case series. Radiol Bras. 2021;54(4):238-242. doi:10.1590/0100-3984.2020.0073
36. Layne KA, Wood DM, Dargan PI. Gadolinium-based contrast agents—what is the evidence for ‘gadolinium deposition disease’ and the use of chelation therapy? Clin Toxicol (Phila). 2020;58(3):151-160. doi:10.1080/15563650.2019.1681442
37. Nehra AK, McDonald RJ, Bluhm AM, et al. Accumulation of gadolinium in human cerebrospinal fluid after gadobutrol-enhanced MR imaging: a prospective observational cohort study. Radiology. 2018;288(2):416-423. doi:10.1148/radiol.2018171105
38. US Food and Drug Administration. Medical Imaging Drugs Advisory Committee Meeting. Gadolinium retention after gadolinium based contrast magnetic resonance imaging in patients with normal renal function. Briefing document. 2017. Accessed April 12, 2022. https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/MedicalImagingDrugsAdvisoryCommittee/UCM572848.pdf
39. Calvo N, Jamil M, Feldman S, Shah A, Nauman F, Ferrara J. Neurotoxicity from intrathecal gadolinium administration: case presentation and brief review. Neurol Clin Pract. 2020;10(1):e7-e10. doi:10.1212/CPJ.0000000000000696
40. Bower DV, Richter JK, von Tengg-Kobligk H, Heverhagen JT, Runge VM. Gadolinium-based MRI contrast agents induce mitochondrial toxicity and cell death in human neurons, and toxicity increases with reduced kinetic stability of the agent. Invest Radiol. 2019;54(8):453-463. doi:10.1097/RLI.0000000000000567
41. McDonald RJ, McDonald JS, Kallmes DF, et al. Gadolinium deposition in human brain tissues after contrast-enhanced MR imaging in adult patients without intracranial abnormalities. Radiology. 2017;285(2):546-554. doi:10.1148/radiol.2017161595
42. Do C, DeAguero J, Brearley A, et al. Gadolinium-based contrast agent use, their safety, and practice evolution. Kidney360. 2020;1(6):561-568. doi:10.34067/KID.0000272019
43. Di Gregorio E, Furlan C, Atlante S, Stefania R, Gianolio E, Aime S. Gadolinium retention in erythrocytes and leukocytes from human and murine blood upon treatment with gadolinium-based contrast agents for magnetic resonance imaging. Invest Radiol. 2020;55(1):30-37. doi:10.1097/RLI.0000000000000608
44. Maecker HT, Siebert JC, Rosenberg-Hasson Y, Koran LM, Ramalho M, Semelka RC. Acute chelation therapy-associated changes in urine gadolinium, self-reported flare severity, and serum cytokines in gadolinium deposition disease. Invest Radiol. 2021;56(6):374-384. doi:10.1097/RLI.0000000000000752
45. Maecker HT, Wang W, Rosenberg-Hasson Y, Semelka RC, Hickey J, Koran LM. An initial investigation of serum cytokine levels in patients with gadolinium retention. Radiol Bras. 2020;53(5):306-313. doi:10.1590/0100-3984.2019.0075
46. Birka M, Wentker KS, Lusmöller E, et al. Diagnosis of nephrogenic systemic fibrosis by means of elemental bioimaging and speciation analysis. Anal Chem. 2015;87(6):3321-3328. doi:10.1021/ac504488k
Gadolinium (Gd)-based contrast agents are frequently used in health care for enhancing magnetic resonance image (MRI) signals at low concentrations. Contrary to popular opinion, this widely used heavy metal is not biologically inert. Once notable for its safety profile, there is mounting evidence for Gd deposition in various organ systems of the body, even in those with normal renal function. A large knowledge gap remains concerning the potential harms of Gd deposition and the factors determining its elimination from the body. However, the findings of deposited Gd throughout various organs and their intracellular compartments even years after the initial exposure have been established. Here, we describe a case of a Vietnam-era veteran whose presentation, clinical, and laboratory findings were consistent within the spectrum of Gd deposition disease.
Case Presentation
A Vietnam-era veteran aged > 70 years presented for evaluation of Gd-based contrast agent–induced chronic multisymptomatic illness His medical history was significant for chronic low back pain, chronic hypertension, type 2 diabetes mellitus, and hypogonadism. Surgical history was notable for back surgery (24 years prior), laminectomy (2 years prior), shoulder replacement (2 years prior), and an epidural complicated by a hematoma (1 year prior). His presenting concerns included a painful and pruritic rash that worsened with showering, pain originating at the right Achilles tendon with migration to the knee, and shoulder pain. His symptoms started shortly after receiving multiple exposures to Gd-based contrast agents to enhance MRIs during his clinical care (Omniscan 20 mL, Omniscan 20 mL, and Gadovist 10 mL, administered 578, 565, and 496 days prior to the clinic visit, respectively). New onset headaches coincided with the timeline of symptom onset, in addition to hoarseness and liberation of an “oily substance” from the skin. More than one year prior to this clinic visit, he was considered for having polymyalgia rheumatica given the ambiguity of symptoms. Functional status remained impaired despite treatment with prednisone and methotrexate.
The patient’s military service was in the mid-1960s. He was deployed to Japan and had no knowledge of an Agent Orange exposure. His tobacco history was distant, and he reported no tattoos, prior transfusions, or occupational metal exposure
Clinical Findings
The patient was afebrile, normotensive (146/88 mmHg), and normocardic. His weight was 100 kg. He was well nourished and in no acute distress. The thought process was attentive, and his affect pleasant. Ocular examination was notable for arcus senilus. The fundoscopic examination was limited on the left, but there was no neovascularization on the right. Jugular venous pulsation was normal at 8 cm. Right ventricular impulse was slightly hyperdynamic, the rhythm was regular, and there was no abnormal splitting of S2. A soft-grade I/VI crescendo/decrescendo murmur was auscultated along the apex. Radial pulses were 2/2. He was not in respiratory distress, with equally resonant fields bilaterally. Lung sounds were clear bilaterally. A papular, erythematous rash was present in a general distribution over the chest, with few telangiectasias and some varicosity along his left arm. The skin had normal elasticity, although the skin of the hands and legs was papyraceous.
Gd levels were measured in the blood and urine (Table 1). Gd was detectable in the skin (0.2 µg/g) nearly 400 days after the last exposure. Gd was still detectable in the patient’s blood and urine (0.2 ng/mL and 0.5 µg/24 h, respectively) more than 3 years after his last exposure.
Discussion
In the United States, there are 40.44 MRI units per million people and 40 million MRIs are conducted annually. From 30 to 50% of these are enhanced with Gd-based contrast agents. In the past 30 years, there have been > 450 million contrast-enhanced MRI procedures.1
Gd is a rare earth metal. Among commercially available elements Gd has exceptional properties for enhancing MRI signals at low concentrations.1 The nonphysiologic metal is detoxified by chelation with proprietary multidentate formulations that enhance (primarily renal) elimination while retaining the paramagnetic and chemical properties for imaging. Gd exposure was found to be associated to iatrogenic nephrogenic systemic fibrosis in 2006 and later confirmed via multiple systematic reviews.2 Gd is retained in every vital organ after exposure.3 Gd-based contrast agents stimulate bone marrow–derived fibrocytes in mediating fibrosis, and bone marrow develop a memory of prior contrast exposure (Figure 1).4-6 Systemic fibrosis is mediated by the monocyte chemoattractant protein 1/C-C chemokine receptor 2.6,7 Even in the setting of normal renal function, Gd-based contrast induces the formation of Gd-rich nanoparticles in the skin and kidney.7,8 Far from being inert, Gd-based contrast agents induce systemic metabolic changes such as hypertriglyceridemia, elevations in low-density lipoprotein cholesterol, insulin resistance, and the Warburg effect (glycolytic/energy switching) in the renal cortex concomitant with profound mitochondrial abnormalities.8
We have discovered that the rate of Gd-enhanced procedures has increased immensely within the Veterans Health Administration (VHA) system in a subset of patients with designated kidney disease (Table 2). Although a substantial number of procedures are dedicated to head and brain imaging within the VHA, the indications for Gd-enhanced diagnoses (eg, cardiac) are increasing (Figure 2).
Retention of Gd can be modeled as a function of time (t) by the half-lives of the fast, intermediate, and slow phases of elimination (Ta, Tb, and Tc, respectively):9
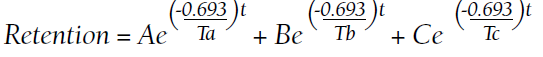
A, B, and C are the proportions (adding to 100%) that represent each of the compartments:
Numerous patients with normal renal function developed similar or novel symptoms that have been attributed to Gd concomitant with detectable urinary Gd years after exposure.11 Gd-based contrast agents are increasingly associated with cutaneous abnormalities even outside of nephrogenic systemic fibrosis. Gd-associated plaques develop in patients without kidney disease—these range from asymptomatic, pruritic, to burning.13 Histologic specimens reveal CD68 and factor XIIIa–positive spindle-shaped myeloid cells (the same mediators of iatrogenic systemic fibrosis) or CD34-positive cells. CD68 and factor XIIIa are distinctive for histologic specimens from patients with systemic fibrosis, and these markers have been detected in our preclinical models that demonstrated that bone marrow–derived cells are involved in mediating fibrosis.3,4,14-19 Similarly, CD34-positive cells have been historically associated with systemic fibrosis lesions.15,16,18-23 Plump osteocyte-appearing cells have also been noted (note that extraosseous metaplasia makes the histologic diagnosis of systemic fibrosis).14 Nephrogenic systemic fibrosis is an iatrogenic disease that can manifest years after exposure to Gd.5 Gd induces the recruitment of bone marrow–derived cells to the affected sites.4
The VA Health Service Research and Development Evidence Synthesis Program reviewed the safety of Gd-based contrast agents in patients with impaired kidney function.24,25 The group found only a single study of Gd and veterans. “Awareness and concern are growing about the long-term deposition of gadolinium in [the] brain and other tissues among patients with normal kidney function,” according to Lunyera and colleagues.25 The largest knowledge gap was that a comprehensive review “of all potential harms associated with gadolinium exposure” was not addressed. Furthermore, the group advised “caution in the use of [Gd-based contrast agents] in patients with severely impaired kidney function and acute kidney injury remains prudent, because the exact clinical factors contributing to [nephrogenic systemic fibrosis] risk in these subpopulations are still unknown.”25
Gd-based contrast agents—contrary to a widely held misconception—are not biologically inert.1 Gd-based contrast agents have a long history of association with acute renal injury. We have demonstrated that systemic treatment with MRI contrast agents leads to vacuolization of the proximal tubule and tubular injury.7,8 Kidney injury may be mediated by the generation of reactive oxygen species from NADPH oxidase 4 (Nox4).26
Gd retention, Gd-induced multisymptomatic illnesses, Gd-associated plaques, Gd-induced neurotoxicity, and nephrogenic systemic fibrosis are part of a continuum (with Gd as the common thread)—a theme of the September 8, 2017, US Food and Drug Administration (FDA) Medical Imaging Drugs Advisory Committee meeting.27 Patients, patient advocacy groups, and regulating agencies are concerned about long-term retention of a nonphysiologic rare earth element such as Gd.28-30 A patient advocacy group, The Lighthouse Project, collected information from patients linking the last date of Gd-based contrast agent exposure and urinary Gd.11 Data from their report suggest that the rate constants (valuable for the elimination equation above) are obtainable from 24-hour urine collections. Conceptually, Gd-induced diseases may represent a continuum that results from the retention of a nonphysiologic, toxic heavy rare earth metal.
As a heavy metal, Gd is not a natural physiologic trace element. Similar to numerous nonphysiologic metals, Gd is toxic. Inhaled Gd oxide (Gd2O3) dust leads to a number of time-dependent pathologies. Animal lung studies demonstrate reduced elasticity, enlarged cells, thickened lung walls, and recruitment of immune cells.31 Symptoms of acute IV Gd toxicity include decreased respiration, lethargy, abdominal cramps, and diarrhea.32 Pharmacologically, Gd concentrates in the liver and kidney and accumulates in the bone.32 Animals demonstrate intestinal depression and low blood pressure in response to Gd and, with higher doses, cardiovascular collapse.32 IV Gd chloride leads to metal deposition in the small blood vessels diffusely throughout the body, particularly in the lung and kidney and the metal is absorbed by the scavenging white blood cells.33 Gd chloride induces severe damage to the liver, spleen, and the digestive tract.33 Furthermore, this form of the toxicant metal markedly impacted functions associated with bleeding and clotting, ie, decreased platelet numbers and an increase in the laboratory-measured coagulation parameters.33 Semelka and colleagues have characterized chronic symptoms attributed to Gd-based contrast agents (not limited to chronic pain, headache, bone pain, skin thickening, and clouded mentation).34,35 Because Gd-induced conditions are underrecognized and ill-defined, disinherited patients often resort to untested (and potentially dangerous) chelation therapies.36
This patient presented with numerous symptoms that arose after Gd exposure. It is well established that Gd-based contrast agents (of any class) are retained in multiple organs (including the brain), for months to years. Gd-based contrast agents enter the cerebrospinal fluid within minutes of IV administration.37 Gd was found in the cerebrospinal fluid 9 months after administration in a case presented to the FDA Medical Imaging Drugs Advisory Committee.38 We know from intentional and accidental intrathecal administrations that Gd-based contrast agents are neurotoxic.39 Runge and colleagues demonstrated that Gd-based contrast agents exert mitochondrial toxicity in cultured neurons in vitro.40 McDonald and his team found Gd-rich nanoparticles within the brain neurons (cytoplasm and nuclei) from patients exposed to MRI contrast in the normal course of care.41 These nanoparticles are similar to what we have found in rodent models of Gd-induced disease.7,8,42
Prolonged elimination of Gd after MRI contrast administration (months to years) may be universal.10 Gd compartmentalizes into leukocytes and erythrocytes and into the cerebrospinal fluid within minutes.37,43 Patients with multisymptomatic illnesses attributed to Gd (Gd deposition disease) have perturbations in cytokine levels, many inflammatory.44,45 The results are concerning: Gd is retained intracellularly in vital organs, including brain neurons. It is inarguable that Gd is an alien, nonphysiologic element. With mounting evidence that Gd retention has clinical consequences, patients should be provided proper informed consent. Complications of renal insufficiency (ie, hyperkalemia, hyperphosphatemia, renal osteodystrophy, hyponatremia, anemia, immunosuppression, etc) follow a smooth, curvilinear slope as the true (not estimated) glomerular filtration declines; the worst iatrogenic complication from Gd—systemic fibrosis—is likely no different.
Patient Perspective
“Seems like it’s one thing after another. My family doctor said that once I had the gadolinium exposures, I have had problems ever since that I don’t recover from.” This includes chronic numbness from the rectum to the bilateral lower extremities and an indolent worsening kidney function; “I have already developed stage 3B chronic kidney disease.” Similar to many suffering with gadolinium retention, the patient was concerned about the long-term consequences. Gadolinium “is a toxic metal that is going through my body for 4 years. That has to be a problem. How come we don’t have that answer?” Clinician ignorance of Gd-induced complications and long-term retention is frustrating. “Not one of my doctors has taken gadolinium retention seriously. Where else are patients supposed to go?”
Conclusions
Health care professionals should be considering subclinical manifestations of nephrogenic systemic fibrosis or open to considering that intracellular neuronal retention of Gd may correlate with symptoms arising after MRI contrast exposures. The science concerning the mechanisms of how Gd exerts its pathologic effects is lagging behind the commercialization of enhancing Gd elimination (ie, chelation therapies) and other untested remedies. Practitioners need to acknowledge the unknown potential consequences of Gd and listen to patients who suspect chronic adverse effects.
Gadolinium (Gd)-based contrast agents are frequently used in health care for enhancing magnetic resonance image (MRI) signals at low concentrations. Contrary to popular opinion, this widely used heavy metal is not biologically inert. Once notable for its safety profile, there is mounting evidence for Gd deposition in various organ systems of the body, even in those with normal renal function. A large knowledge gap remains concerning the potential harms of Gd deposition and the factors determining its elimination from the body. However, the findings of deposited Gd throughout various organs and their intracellular compartments even years after the initial exposure have been established. Here, we describe a case of a Vietnam-era veteran whose presentation, clinical, and laboratory findings were consistent within the spectrum of Gd deposition disease.
Case Presentation
A Vietnam-era veteran aged > 70 years presented for evaluation of Gd-based contrast agent–induced chronic multisymptomatic illness His medical history was significant for chronic low back pain, chronic hypertension, type 2 diabetes mellitus, and hypogonadism. Surgical history was notable for back surgery (24 years prior), laminectomy (2 years prior), shoulder replacement (2 years prior), and an epidural complicated by a hematoma (1 year prior). His presenting concerns included a painful and pruritic rash that worsened with showering, pain originating at the right Achilles tendon with migration to the knee, and shoulder pain. His symptoms started shortly after receiving multiple exposures to Gd-based contrast agents to enhance MRIs during his clinical care (Omniscan 20 mL, Omniscan 20 mL, and Gadovist 10 mL, administered 578, 565, and 496 days prior to the clinic visit, respectively). New onset headaches coincided with the timeline of symptom onset, in addition to hoarseness and liberation of an “oily substance” from the skin. More than one year prior to this clinic visit, he was considered for having polymyalgia rheumatica given the ambiguity of symptoms. Functional status remained impaired despite treatment with prednisone and methotrexate.
The patient’s military service was in the mid-1960s. He was deployed to Japan and had no knowledge of an Agent Orange exposure. His tobacco history was distant, and he reported no tattoos, prior transfusions, or occupational metal exposure
Clinical Findings
The patient was afebrile, normotensive (146/88 mmHg), and normocardic. His weight was 100 kg. He was well nourished and in no acute distress. The thought process was attentive, and his affect pleasant. Ocular examination was notable for arcus senilus. The fundoscopic examination was limited on the left, but there was no neovascularization on the right. Jugular venous pulsation was normal at 8 cm. Right ventricular impulse was slightly hyperdynamic, the rhythm was regular, and there was no abnormal splitting of S2. A soft-grade I/VI crescendo/decrescendo murmur was auscultated along the apex. Radial pulses were 2/2. He was not in respiratory distress, with equally resonant fields bilaterally. Lung sounds were clear bilaterally. A papular, erythematous rash was present in a general distribution over the chest, with few telangiectasias and some varicosity along his left arm. The skin had normal elasticity, although the skin of the hands and legs was papyraceous.
Gd levels were measured in the blood and urine (Table 1). Gd was detectable in the skin (0.2 µg/g) nearly 400 days after the last exposure. Gd was still detectable in the patient’s blood and urine (0.2 ng/mL and 0.5 µg/24 h, respectively) more than 3 years after his last exposure.
Discussion
In the United States, there are 40.44 MRI units per million people and 40 million MRIs are conducted annually. From 30 to 50% of these are enhanced with Gd-based contrast agents. In the past 30 years, there have been > 450 million contrast-enhanced MRI procedures.1
Gd is a rare earth metal. Among commercially available elements Gd has exceptional properties for enhancing MRI signals at low concentrations.1 The nonphysiologic metal is detoxified by chelation with proprietary multidentate formulations that enhance (primarily renal) elimination while retaining the paramagnetic and chemical properties for imaging. Gd exposure was found to be associated to iatrogenic nephrogenic systemic fibrosis in 2006 and later confirmed via multiple systematic reviews.2 Gd is retained in every vital organ after exposure.3 Gd-based contrast agents stimulate bone marrow–derived fibrocytes in mediating fibrosis, and bone marrow develop a memory of prior contrast exposure (Figure 1).4-6 Systemic fibrosis is mediated by the monocyte chemoattractant protein 1/C-C chemokine receptor 2.6,7 Even in the setting of normal renal function, Gd-based contrast induces the formation of Gd-rich nanoparticles in the skin and kidney.7,8 Far from being inert, Gd-based contrast agents induce systemic metabolic changes such as hypertriglyceridemia, elevations in low-density lipoprotein cholesterol, insulin resistance, and the Warburg effect (glycolytic/energy switching) in the renal cortex concomitant with profound mitochondrial abnormalities.8
We have discovered that the rate of Gd-enhanced procedures has increased immensely within the Veterans Health Administration (VHA) system in a subset of patients with designated kidney disease (Table 2). Although a substantial number of procedures are dedicated to head and brain imaging within the VHA, the indications for Gd-enhanced diagnoses (eg, cardiac) are increasing (Figure 2).
Retention of Gd can be modeled as a function of time (t) by the half-lives of the fast, intermediate, and slow phases of elimination (Ta, Tb, and Tc, respectively):9
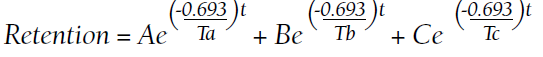
A, B, and C are the proportions (adding to 100%) that represent each of the compartments:
Numerous patients with normal renal function developed similar or novel symptoms that have been attributed to Gd concomitant with detectable urinary Gd years after exposure.11 Gd-based contrast agents are increasingly associated with cutaneous abnormalities even outside of nephrogenic systemic fibrosis. Gd-associated plaques develop in patients without kidney disease—these range from asymptomatic, pruritic, to burning.13 Histologic specimens reveal CD68 and factor XIIIa–positive spindle-shaped myeloid cells (the same mediators of iatrogenic systemic fibrosis) or CD34-positive cells. CD68 and factor XIIIa are distinctive for histologic specimens from patients with systemic fibrosis, and these markers have been detected in our preclinical models that demonstrated that bone marrow–derived cells are involved in mediating fibrosis.3,4,14-19 Similarly, CD34-positive cells have been historically associated with systemic fibrosis lesions.15,16,18-23 Plump osteocyte-appearing cells have also been noted (note that extraosseous metaplasia makes the histologic diagnosis of systemic fibrosis).14 Nephrogenic systemic fibrosis is an iatrogenic disease that can manifest years after exposure to Gd.5 Gd induces the recruitment of bone marrow–derived cells to the affected sites.4
The VA Health Service Research and Development Evidence Synthesis Program reviewed the safety of Gd-based contrast agents in patients with impaired kidney function.24,25 The group found only a single study of Gd and veterans. “Awareness and concern are growing about the long-term deposition of gadolinium in [the] brain and other tissues among patients with normal kidney function,” according to Lunyera and colleagues.25 The largest knowledge gap was that a comprehensive review “of all potential harms associated with gadolinium exposure” was not addressed. Furthermore, the group advised “caution in the use of [Gd-based contrast agents] in patients with severely impaired kidney function and acute kidney injury remains prudent, because the exact clinical factors contributing to [nephrogenic systemic fibrosis] risk in these subpopulations are still unknown.”25
Gd-based contrast agents—contrary to a widely held misconception—are not biologically inert.1 Gd-based contrast agents have a long history of association with acute renal injury. We have demonstrated that systemic treatment with MRI contrast agents leads to vacuolization of the proximal tubule and tubular injury.7,8 Kidney injury may be mediated by the generation of reactive oxygen species from NADPH oxidase 4 (Nox4).26
Gd retention, Gd-induced multisymptomatic illnesses, Gd-associated plaques, Gd-induced neurotoxicity, and nephrogenic systemic fibrosis are part of a continuum (with Gd as the common thread)—a theme of the September 8, 2017, US Food and Drug Administration (FDA) Medical Imaging Drugs Advisory Committee meeting.27 Patients, patient advocacy groups, and regulating agencies are concerned about long-term retention of a nonphysiologic rare earth element such as Gd.28-30 A patient advocacy group, The Lighthouse Project, collected information from patients linking the last date of Gd-based contrast agent exposure and urinary Gd.11 Data from their report suggest that the rate constants (valuable for the elimination equation above) are obtainable from 24-hour urine collections. Conceptually, Gd-induced diseases may represent a continuum that results from the retention of a nonphysiologic, toxic heavy rare earth metal.
As a heavy metal, Gd is not a natural physiologic trace element. Similar to numerous nonphysiologic metals, Gd is toxic. Inhaled Gd oxide (Gd2O3) dust leads to a number of time-dependent pathologies. Animal lung studies demonstrate reduced elasticity, enlarged cells, thickened lung walls, and recruitment of immune cells.31 Symptoms of acute IV Gd toxicity include decreased respiration, lethargy, abdominal cramps, and diarrhea.32 Pharmacologically, Gd concentrates in the liver and kidney and accumulates in the bone.32 Animals demonstrate intestinal depression and low blood pressure in response to Gd and, with higher doses, cardiovascular collapse.32 IV Gd chloride leads to metal deposition in the small blood vessels diffusely throughout the body, particularly in the lung and kidney and the metal is absorbed by the scavenging white blood cells.33 Gd chloride induces severe damage to the liver, spleen, and the digestive tract.33 Furthermore, this form of the toxicant metal markedly impacted functions associated with bleeding and clotting, ie, decreased platelet numbers and an increase in the laboratory-measured coagulation parameters.33 Semelka and colleagues have characterized chronic symptoms attributed to Gd-based contrast agents (not limited to chronic pain, headache, bone pain, skin thickening, and clouded mentation).34,35 Because Gd-induced conditions are underrecognized and ill-defined, disinherited patients often resort to untested (and potentially dangerous) chelation therapies.36
This patient presented with numerous symptoms that arose after Gd exposure. It is well established that Gd-based contrast agents (of any class) are retained in multiple organs (including the brain), for months to years. Gd-based contrast agents enter the cerebrospinal fluid within minutes of IV administration.37 Gd was found in the cerebrospinal fluid 9 months after administration in a case presented to the FDA Medical Imaging Drugs Advisory Committee.38 We know from intentional and accidental intrathecal administrations that Gd-based contrast agents are neurotoxic.39 Runge and colleagues demonstrated that Gd-based contrast agents exert mitochondrial toxicity in cultured neurons in vitro.40 McDonald and his team found Gd-rich nanoparticles within the brain neurons (cytoplasm and nuclei) from patients exposed to MRI contrast in the normal course of care.41 These nanoparticles are similar to what we have found in rodent models of Gd-induced disease.7,8,42
Prolonged elimination of Gd after MRI contrast administration (months to years) may be universal.10 Gd compartmentalizes into leukocytes and erythrocytes and into the cerebrospinal fluid within minutes.37,43 Patients with multisymptomatic illnesses attributed to Gd (Gd deposition disease) have perturbations in cytokine levels, many inflammatory.44,45 The results are concerning: Gd is retained intracellularly in vital organs, including brain neurons. It is inarguable that Gd is an alien, nonphysiologic element. With mounting evidence that Gd retention has clinical consequences, patients should be provided proper informed consent. Complications of renal insufficiency (ie, hyperkalemia, hyperphosphatemia, renal osteodystrophy, hyponatremia, anemia, immunosuppression, etc) follow a smooth, curvilinear slope as the true (not estimated) glomerular filtration declines; the worst iatrogenic complication from Gd—systemic fibrosis—is likely no different.
Patient Perspective
“Seems like it’s one thing after another. My family doctor said that once I had the gadolinium exposures, I have had problems ever since that I don’t recover from.” This includes chronic numbness from the rectum to the bilateral lower extremities and an indolent worsening kidney function; “I have already developed stage 3B chronic kidney disease.” Similar to many suffering with gadolinium retention, the patient was concerned about the long-term consequences. Gadolinium “is a toxic metal that is going through my body for 4 years. That has to be a problem. How come we don’t have that answer?” Clinician ignorance of Gd-induced complications and long-term retention is frustrating. “Not one of my doctors has taken gadolinium retention seriously. Where else are patients supposed to go?”
Conclusions
Health care professionals should be considering subclinical manifestations of nephrogenic systemic fibrosis or open to considering that intracellular neuronal retention of Gd may correlate with symptoms arising after MRI contrast exposures. The science concerning the mechanisms of how Gd exerts its pathologic effects is lagging behind the commercialization of enhancing Gd elimination (ie, chelation therapies) and other untested remedies. Practitioners need to acknowledge the unknown potential consequences of Gd and listen to patients who suspect chronic adverse effects.
1. Leyba K, Wagner B. Gadolinium-based contrast agents: why nephrologists need to be concerned. Curr Opin Nephrol Hypertens. 2019;28(2):154-162. doi:10.1097/MNH.0000000000000475
2. Grobner T. Gadolinium—a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis?. Nephrol Dial Transplant. 2006;21(4):1104-1108. doi:10.1093/ndt/gfk062
3. Do C, Barnes JL, Tan C, Wagner B. Type of MRI contrast, tissue gadolinium, and fibrosis. Am J Physiol Renal Physiol. 2014;307(7):F844-F855. doi:10.1152/ajprenal.00379.2014
4. Wagner B, Tan C, Barnes JL, et al. Nephrogenic systemic fibrosis: evidence for oxidative stress and bone marrow-derived fibrocytes in skin, liver, and heart lesions using a 5/6 nephrectomy rodent model. Am J Pathol. 2012;181(6):1941-1952. doi:10.1016/j.ajpath.2012.08.026
5. Wagner B, Drel V, Gorin Y. Pathophysiology of gadolinium-associated systemic fibrosis. Am J Physiol Renal Physiol. 2016;311(1):F1-F11. doi:10.1152/ajprenal.00166.2016
6. Drel VR, Tan C, Barnes JL, Gorin Y, Lee DY, Wagner B. Centrality of bone marrow in the severity of gadolinium-based contrast-induced systemic fibrosis. FASEB J. 2016;30(9):3026-3038. doi:10.1096/fj.201500188R
7. Do C, Drel V, Tan C, Lee D, Wagner B. Nephrogenic systemic fibrosis is mediated by myeloid C-C chemokine receptor 2. J Invest Dermatol. 2019;139(10):2134-2143.e2. doi:10.1016/j.jid.2019.03.1145
8. Do C, Ford B, Lee DY, Tan C, Escobar P, Wagner B. Gadolinium-based contrast agents: stimulators of myeloid-induced renal fibrosis and major metabolic disruptors. Toxicol Appl Pharmacol. 2019;375:32-45. doi:10.1016/j.taap.2019.05.009
9. Hirano S, Suzuki KT. Exposure, metabolism, and toxicity of rare earths and related compounds. Environ Health Perspect. 1996;104(suppl 1):85-95. doi:10.1289/ehp.96104s185
10. Alwasiyah D, Murphy C, Jannetto P, Hogg M, Beuhler MC. Urinary gadolinium levels after contrast-enhanced MRI in individuals with normal renal function: a pilot study. J Med Toxicol. 2019;15(2):121-127. doi:10.1007/s13181-018-0693-1
11. Williams S, Grimm H. gadolinium toxicity: shedding light on the effects of retained gadolinium from contrast MRI. Accessed April 11, 2022. https://gdtoxicity.files.wordpress.com/2018/12/gadolinium-clearance-times-for-135-contrast-mri-cases-final-v1-1.pdf
12. DeBevits JJ, Reshma M, Bageac D, et al. Gray matter nucleus hyperintensity after monthly triple-dose gadopentetate dimeglumine with long-term magnetic resonance imaging. Invest Radiol. 2020;55(10):629-635. doi:10.1097/RLI.0000000000000663
13. Gathings RM, Reddy R, Santa Cruz D, Brodell RT. Gadolinium-associated plaques: a new, distinctive clinical entity. JAMA Dermatol. 2015;151(3):316-319. doi:10.1001/jamadermatol.2014.2660
14. Girardi M, Kay J, Elston DM, Leboit PE, Abu-Alfa A, Cowper SE. Nephrogenic systemic fibrosis: clinicopathological definition and workup recommendations. J Am Acad Dermatol. 2011;65(6):1095-1106 e7. doi:10.1016/j.jaad.2010.08.041
15. Daram SR, Cortese CM, Bastani B. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis: report of a new case with literature review. Am J Kidney Dis. 2005;46(4):754-759. doi:10.1053/j.ajkd.2005.06.024
16. Ortonne N, Lipsker D, Chantrel F, Boehm N, Grosshans E, Cribier B. Presence of CD45RO+ CD34+ cells with collagen synthesis activity in nephrogenic fibrosing dermopathy: a new pathogenic hypothesis. Br J Dermatol. 2004;150(5):1050-1052. doi:10.1111/j.1365-2133.2004.05900.x
17. Mendoza FA, Artlett CM, Sandorfi N, Latinis K, Piera-Velazquez S, Jimenez SA. Description of 12 cases of nephrogenic fibrosing dermopathy and review of the literature. Semin Arthritis Rheum. 2006;35(4):238-49. doi:10.1016/j.semarthrit.2005.08.002
18. Lewis KG, Lester BW, Pan TD, Robinson-Bostom L. Nephrogenic fibrosing dermopathyand calciphylaxis with pseudoxanthoma elasticum-like changes. J Cutan Pathol. 2006;33(10):695-700. doi:10.1111/j.1600-0560.2006.00490.x
19. Gibson SE, Farver CF, Prayson RA. Multiorgan involvement in nephrogenic fibrosing dermopathy: an autopsy case and review of the literature. Arch Pathol Lab Med. 2006;130(2):209-212. doi:10.5858/2006-130-209-MIINFD
20. Cassis TB, Jackson JM, Sonnier GB, Callen JP. Nephrogenic fibrosing dermopathy in a patient with acute renal failure never requiring dialysis. Int J Dermatol. 2006;45(1):56-59. doi:10.1111/j.1365-4632.2005.02701.x
21. Kucher C, Steere J, Elenitsas R, Siegel DL, Xu X. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis with diaphragmatic involvement in a patient with respiratory failure. J Am Acad Dermatol. 2006;54(suppl 2):S31-S34. doi:10.1016/j.jaad.2005.04.024
22. Sanyal S, Marckmann P, Scherer S, Abraham JL. Multiorgan gadolinium (Gd) deposition and fibrosis in a patient with nephrogenic systemic fibrosis—an autopsy-based review. Nephrol Dial Transplant. 2011;26(11):3616-3626. doi:10.1093/ndt/gfr085
23. Kucher C, Xu X, Pasha T, Elenitsas R. Histopathologic comparison of nephrogenic fibrosing dermopathy and scleromyxedema. J Cutan Pathol. 2005;32(7):484-490. doi:10.1111/j.0303-6987.2005.00365.x
24. Goldstein KM, Lunyera J, Mohottige D, et al. Risk of Nephrogenic Systemic Fibrosis after Exposure to Newer Gadolinium Agents. Washington (DC): Department of Veterans Affairs (US); October 2019. https://www.ncbi.nlm.nih.gov/books/NBK559376/25. Lunyera J, Mohottige D, Alexopoulos AS, et al. Risk for nephrogenic systemic fibrosis after exposure to newer gadolinium agents: a systematic review. Ann Intern Med. 2020;173(2):110-119. doi:10.7326/M20-0299
26. Bruno F, DeAguero J, Do C, et al. Overlapping roles of NADPH Oxidase 4 (Nox4) for diabetic and gadolinium-based contrast agent-induced systemic fibrosis. Am J Physiol Renal Physiol. 2021;320(4):F617-F627. doi:10.1152/ajprenal.00456.2020
27. Wagner B. The pathophysiology and retention of gadolinium. United States Food & Drug Administration Medical Imaging Drugs Advisory Committee. 2017:1-23. https://www.fda.gov/advisory-committees/medical-imaging-drugs-advisory-committee/2017-meeting-materials-medical-imaging-drugs-advisory-committee?msclkid=6b5764ccbaa611ec95e35dddf8db57af
28. Runge VM. Critical questions regarding gadolinium deposition in the brain and body after injections of the gadolinium-based contrast agents, safety, and clinical recommendations in consideration of the EMA’s pharmacovigilance and risk assessment committee recommendation for suspension of the marketing authorizations for 4 linear agents. Invest Radiol. 2017;52(6):317-323. doi:10.1097/RLI.0000000000000374
29. Wagner B. Scared to the marrow: pitfalls and pearls in renal imaging. Adv Chronic Kidney Dis. 2017;24(3):136-137. doi:10.1053/j.ackd.2017.03.008
30. US Food and Drug Administration. Transcript for the September 8, 2017 Meeting of the Medical Imaging Drugs Advisory Committee (MIDAC). September 8, 2017. Accessed April 11, 2022. https://www.fda.gov/media/108935/download
31. Abel M, Talbot RB. Gadolinium oxide inhalation by guinea pigs: a correlative functional and histopathologic study. J Pharmacol Exp Ther. 1967;157(1):207-213.
32. Haley TJ, Raymond K, Komesu N, Upham HC. Toxicological and pharmacological effects of gadolinium and samarium chlorides. Br J Pharmacol Chemother. 1961;17(3):526-532. doi:10.1111/j.1476-5381.1961.tb01139.x

33. Spencer AJ, Wilson SA, Batchelor J, Reid A, Rees J, Harpur E. Gadolinium chloride toxicity in the rat. Toxicol Pathol. 1997;25(3):245-255. doi:10.1177/019262339702500301
34. Semelka RC, Ramalho M, AlObaidy M, Ramalho J. Gadolinium in humans: a family of disorders. AJR Am J Roentgenol. 2016;207(2):229-233. doi:10.2214/AJR.15.15842
35. Semelka RC, Ramalho M. Physicians with self-diagnosed gadolinium deposition disease: a case series. Radiol Bras. 2021;54(4):238-242. doi:10.1590/0100-3984.2020.0073
36. Layne KA, Wood DM, Dargan PI. Gadolinium-based contrast agents—what is the evidence for ‘gadolinium deposition disease’ and the use of chelation therapy? Clin Toxicol (Phila). 2020;58(3):151-160. doi:10.1080/15563650.2019.1681442
37. Nehra AK, McDonald RJ, Bluhm AM, et al. Accumulation of gadolinium in human cerebrospinal fluid after gadobutrol-enhanced MR imaging: a prospective observational cohort study. Radiology. 2018;288(2):416-423. doi:10.1148/radiol.2018171105
38. US Food and Drug Administration. Medical Imaging Drugs Advisory Committee Meeting. Gadolinium retention after gadolinium based contrast magnetic resonance imaging in patients with normal renal function. Briefing document. 2017. Accessed April 12, 2022. https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/MedicalImagingDrugsAdvisoryCommittee/UCM572848.pdf
39. Calvo N, Jamil M, Feldman S, Shah A, Nauman F, Ferrara J. Neurotoxicity from intrathecal gadolinium administration: case presentation and brief review. Neurol Clin Pract. 2020;10(1):e7-e10. doi:10.1212/CPJ.0000000000000696
40. Bower DV, Richter JK, von Tengg-Kobligk H, Heverhagen JT, Runge VM. Gadolinium-based MRI contrast agents induce mitochondrial toxicity and cell death in human neurons, and toxicity increases with reduced kinetic stability of the agent. Invest Radiol. 2019;54(8):453-463. doi:10.1097/RLI.0000000000000567
41. McDonald RJ, McDonald JS, Kallmes DF, et al. Gadolinium deposition in human brain tissues after contrast-enhanced MR imaging in adult patients without intracranial abnormalities. Radiology. 2017;285(2):546-554. doi:10.1148/radiol.2017161595
42. Do C, DeAguero J, Brearley A, et al. Gadolinium-based contrast agent use, their safety, and practice evolution. Kidney360. 2020;1(6):561-568. doi:10.34067/KID.0000272019
43. Di Gregorio E, Furlan C, Atlante S, Stefania R, Gianolio E, Aime S. Gadolinium retention in erythrocytes and leukocytes from human and murine blood upon treatment with gadolinium-based contrast agents for magnetic resonance imaging. Invest Radiol. 2020;55(1):30-37. doi:10.1097/RLI.0000000000000608
44. Maecker HT, Siebert JC, Rosenberg-Hasson Y, Koran LM, Ramalho M, Semelka RC. Acute chelation therapy-associated changes in urine gadolinium, self-reported flare severity, and serum cytokines in gadolinium deposition disease. Invest Radiol. 2021;56(6):374-384. doi:10.1097/RLI.0000000000000752
45. Maecker HT, Wang W, Rosenberg-Hasson Y, Semelka RC, Hickey J, Koran LM. An initial investigation of serum cytokine levels in patients with gadolinium retention. Radiol Bras. 2020;53(5):306-313. doi:10.1590/0100-3984.2019.0075
46. Birka M, Wentker KS, Lusmöller E, et al. Diagnosis of nephrogenic systemic fibrosis by means of elemental bioimaging and speciation analysis. Anal Chem. 2015;87(6):3321-3328. doi:10.1021/ac504488k
1. Leyba K, Wagner B. Gadolinium-based contrast agents: why nephrologists need to be concerned. Curr Opin Nephrol Hypertens. 2019;28(2):154-162. doi:10.1097/MNH.0000000000000475
2. Grobner T. Gadolinium—a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis?. Nephrol Dial Transplant. 2006;21(4):1104-1108. doi:10.1093/ndt/gfk062
3. Do C, Barnes JL, Tan C, Wagner B. Type of MRI contrast, tissue gadolinium, and fibrosis. Am J Physiol Renal Physiol. 2014;307(7):F844-F855. doi:10.1152/ajprenal.00379.2014
4. Wagner B, Tan C, Barnes JL, et al. Nephrogenic systemic fibrosis: evidence for oxidative stress and bone marrow-derived fibrocytes in skin, liver, and heart lesions using a 5/6 nephrectomy rodent model. Am J Pathol. 2012;181(6):1941-1952. doi:10.1016/j.ajpath.2012.08.026
5. Wagner B, Drel V, Gorin Y. Pathophysiology of gadolinium-associated systemic fibrosis. Am J Physiol Renal Physiol. 2016;311(1):F1-F11. doi:10.1152/ajprenal.00166.2016
6. Drel VR, Tan C, Barnes JL, Gorin Y, Lee DY, Wagner B. Centrality of bone marrow in the severity of gadolinium-based contrast-induced systemic fibrosis. FASEB J. 2016;30(9):3026-3038. doi:10.1096/fj.201500188R
7. Do C, Drel V, Tan C, Lee D, Wagner B. Nephrogenic systemic fibrosis is mediated by myeloid C-C chemokine receptor 2. J Invest Dermatol. 2019;139(10):2134-2143.e2. doi:10.1016/j.jid.2019.03.1145
8. Do C, Ford B, Lee DY, Tan C, Escobar P, Wagner B. Gadolinium-based contrast agents: stimulators of myeloid-induced renal fibrosis and major metabolic disruptors. Toxicol Appl Pharmacol. 2019;375:32-45. doi:10.1016/j.taap.2019.05.009
9. Hirano S, Suzuki KT. Exposure, metabolism, and toxicity of rare earths and related compounds. Environ Health Perspect. 1996;104(suppl 1):85-95. doi:10.1289/ehp.96104s185
10. Alwasiyah D, Murphy C, Jannetto P, Hogg M, Beuhler MC. Urinary gadolinium levels after contrast-enhanced MRI in individuals with normal renal function: a pilot study. J Med Toxicol. 2019;15(2):121-127. doi:10.1007/s13181-018-0693-1
11. Williams S, Grimm H. gadolinium toxicity: shedding light on the effects of retained gadolinium from contrast MRI. Accessed April 11, 2022. https://gdtoxicity.files.wordpress.com/2018/12/gadolinium-clearance-times-for-135-contrast-mri-cases-final-v1-1.pdf
12. DeBevits JJ, Reshma M, Bageac D, et al. Gray matter nucleus hyperintensity after monthly triple-dose gadopentetate dimeglumine with long-term magnetic resonance imaging. Invest Radiol. 2020;55(10):629-635. doi:10.1097/RLI.0000000000000663
13. Gathings RM, Reddy R, Santa Cruz D, Brodell RT. Gadolinium-associated plaques: a new, distinctive clinical entity. JAMA Dermatol. 2015;151(3):316-319. doi:10.1001/jamadermatol.2014.2660
14. Girardi M, Kay J, Elston DM, Leboit PE, Abu-Alfa A, Cowper SE. Nephrogenic systemic fibrosis: clinicopathological definition and workup recommendations. J Am Acad Dermatol. 2011;65(6):1095-1106 e7. doi:10.1016/j.jaad.2010.08.041
15. Daram SR, Cortese CM, Bastani B. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis: report of a new case with literature review. Am J Kidney Dis. 2005;46(4):754-759. doi:10.1053/j.ajkd.2005.06.024
16. Ortonne N, Lipsker D, Chantrel F, Boehm N, Grosshans E, Cribier B. Presence of CD45RO+ CD34+ cells with collagen synthesis activity in nephrogenic fibrosing dermopathy: a new pathogenic hypothesis. Br J Dermatol. 2004;150(5):1050-1052. doi:10.1111/j.1365-2133.2004.05900.x
17. Mendoza FA, Artlett CM, Sandorfi N, Latinis K, Piera-Velazquez S, Jimenez SA. Description of 12 cases of nephrogenic fibrosing dermopathy and review of the literature. Semin Arthritis Rheum. 2006;35(4):238-49. doi:10.1016/j.semarthrit.2005.08.002
18. Lewis KG, Lester BW, Pan TD, Robinson-Bostom L. Nephrogenic fibrosing dermopathyand calciphylaxis with pseudoxanthoma elasticum-like changes. J Cutan Pathol. 2006;33(10):695-700. doi:10.1111/j.1600-0560.2006.00490.x
19. Gibson SE, Farver CF, Prayson RA. Multiorgan involvement in nephrogenic fibrosing dermopathy: an autopsy case and review of the literature. Arch Pathol Lab Med. 2006;130(2):209-212. doi:10.5858/2006-130-209-MIINFD
20. Cassis TB, Jackson JM, Sonnier GB, Callen JP. Nephrogenic fibrosing dermopathy in a patient with acute renal failure never requiring dialysis. Int J Dermatol. 2006;45(1):56-59. doi:10.1111/j.1365-4632.2005.02701.x
21. Kucher C, Steere J, Elenitsas R, Siegel DL, Xu X. Nephrogenic fibrosing dermopathy/nephrogenic systemic fibrosis with diaphragmatic involvement in a patient with respiratory failure. J Am Acad Dermatol. 2006;54(suppl 2):S31-S34. doi:10.1016/j.jaad.2005.04.024
22. Sanyal S, Marckmann P, Scherer S, Abraham JL. Multiorgan gadolinium (Gd) deposition and fibrosis in a patient with nephrogenic systemic fibrosis—an autopsy-based review. Nephrol Dial Transplant. 2011;26(11):3616-3626. doi:10.1093/ndt/gfr085
23. Kucher C, Xu X, Pasha T, Elenitsas R. Histopathologic comparison of nephrogenic fibrosing dermopathy and scleromyxedema. J Cutan Pathol. 2005;32(7):484-490. doi:10.1111/j.0303-6987.2005.00365.x
24. Goldstein KM, Lunyera J, Mohottige D, et al. Risk of Nephrogenic Systemic Fibrosis after Exposure to Newer Gadolinium Agents. Washington (DC): Department of Veterans Affairs (US); October 2019. https://www.ncbi.nlm.nih.gov/books/NBK559376/25. Lunyera J, Mohottige D, Alexopoulos AS, et al. Risk for nephrogenic systemic fibrosis after exposure to newer gadolinium agents: a systematic review. Ann Intern Med. 2020;173(2):110-119. doi:10.7326/M20-0299
26. Bruno F, DeAguero J, Do C, et al. Overlapping roles of NADPH Oxidase 4 (Nox4) for diabetic and gadolinium-based contrast agent-induced systemic fibrosis. Am J Physiol Renal Physiol. 2021;320(4):F617-F627. doi:10.1152/ajprenal.00456.2020
27. Wagner B. The pathophysiology and retention of gadolinium. United States Food & Drug Administration Medical Imaging Drugs Advisory Committee. 2017:1-23. https://www.fda.gov/advisory-committees/medical-imaging-drugs-advisory-committee/2017-meeting-materials-medical-imaging-drugs-advisory-committee?msclkid=6b5764ccbaa611ec95e35dddf8db57af
28. Runge VM. Critical questions regarding gadolinium deposition in the brain and body after injections of the gadolinium-based contrast agents, safety, and clinical recommendations in consideration of the EMA’s pharmacovigilance and risk assessment committee recommendation for suspension of the marketing authorizations for 4 linear agents. Invest Radiol. 2017;52(6):317-323. doi:10.1097/RLI.0000000000000374
29. Wagner B. Scared to the marrow: pitfalls and pearls in renal imaging. Adv Chronic Kidney Dis. 2017;24(3):136-137. doi:10.1053/j.ackd.2017.03.008
30. US Food and Drug Administration. Transcript for the September 8, 2017 Meeting of the Medical Imaging Drugs Advisory Committee (MIDAC). September 8, 2017. Accessed April 11, 2022. https://www.fda.gov/media/108935/download
31. Abel M, Talbot RB. Gadolinium oxide inhalation by guinea pigs: a correlative functional and histopathologic study. J Pharmacol Exp Ther. 1967;157(1):207-213.
32. Haley TJ, Raymond K, Komesu N, Upham HC. Toxicological and pharmacological effects of gadolinium and samarium chlorides. Br J Pharmacol Chemother. 1961;17(3):526-532. doi:10.1111/j.1476-5381.1961.tb01139.x

33. Spencer AJ, Wilson SA, Batchelor J, Reid A, Rees J, Harpur E. Gadolinium chloride toxicity in the rat. Toxicol Pathol. 1997;25(3):245-255. doi:10.1177/019262339702500301
34. Semelka RC, Ramalho M, AlObaidy M, Ramalho J. Gadolinium in humans: a family of disorders. AJR Am J Roentgenol. 2016;207(2):229-233. doi:10.2214/AJR.15.15842
35. Semelka RC, Ramalho M. Physicians with self-diagnosed gadolinium deposition disease: a case series. Radiol Bras. 2021;54(4):238-242. doi:10.1590/0100-3984.2020.0073
36. Layne KA, Wood DM, Dargan PI. Gadolinium-based contrast agents—what is the evidence for ‘gadolinium deposition disease’ and the use of chelation therapy? Clin Toxicol (Phila). 2020;58(3):151-160. doi:10.1080/15563650.2019.1681442
37. Nehra AK, McDonald RJ, Bluhm AM, et al. Accumulation of gadolinium in human cerebrospinal fluid after gadobutrol-enhanced MR imaging: a prospective observational cohort study. Radiology. 2018;288(2):416-423. doi:10.1148/radiol.2018171105
38. US Food and Drug Administration. Medical Imaging Drugs Advisory Committee Meeting. Gadolinium retention after gadolinium based contrast magnetic resonance imaging in patients with normal renal function. Briefing document. 2017. Accessed April 12, 2022. https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/MedicalImagingDrugsAdvisoryCommittee/UCM572848.pdf
39. Calvo N, Jamil M, Feldman S, Shah A, Nauman F, Ferrara J. Neurotoxicity from intrathecal gadolinium administration: case presentation and brief review. Neurol Clin Pract. 2020;10(1):e7-e10. doi:10.1212/CPJ.0000000000000696
40. Bower DV, Richter JK, von Tengg-Kobligk H, Heverhagen JT, Runge VM. Gadolinium-based MRI contrast agents induce mitochondrial toxicity and cell death in human neurons, and toxicity increases with reduced kinetic stability of the agent. Invest Radiol. 2019;54(8):453-463. doi:10.1097/RLI.0000000000000567
41. McDonald RJ, McDonald JS, Kallmes DF, et al. Gadolinium deposition in human brain tissues after contrast-enhanced MR imaging in adult patients without intracranial abnormalities. Radiology. 2017;285(2):546-554. doi:10.1148/radiol.2017161595
42. Do C, DeAguero J, Brearley A, et al. Gadolinium-based contrast agent use, their safety, and practice evolution. Kidney360. 2020;1(6):561-568. doi:10.34067/KID.0000272019
43. Di Gregorio E, Furlan C, Atlante S, Stefania R, Gianolio E, Aime S. Gadolinium retention in erythrocytes and leukocytes from human and murine blood upon treatment with gadolinium-based contrast agents for magnetic resonance imaging. Invest Radiol. 2020;55(1):30-37. doi:10.1097/RLI.0000000000000608
44. Maecker HT, Siebert JC, Rosenberg-Hasson Y, Koran LM, Ramalho M, Semelka RC. Acute chelation therapy-associated changes in urine gadolinium, self-reported flare severity, and serum cytokines in gadolinium deposition disease. Invest Radiol. 2021;56(6):374-384. doi:10.1097/RLI.0000000000000752
45. Maecker HT, Wang W, Rosenberg-Hasson Y, Semelka RC, Hickey J, Koran LM. An initial investigation of serum cytokine levels in patients with gadolinium retention. Radiol Bras. 2020;53(5):306-313. doi:10.1590/0100-3984.2019.0075
46. Birka M, Wentker KS, Lusmöller E, et al. Diagnosis of nephrogenic systemic fibrosis by means of elemental bioimaging and speciation analysis. Anal Chem. 2015;87(6):3321-3328. doi:10.1021/ac504488k
Is There a Relationship Between Facility Peer Review Findings and Quality in the Veterans Health Administration?
Hospital leaders report the most common aim of peer review (PR) is to improve quality and patient safety, thus it is a potentially powerful quality improvement (QI) driver.1 “When conducted systematically and credibly, peer review for quality management can result in both short-term and long-term improvements in patient care by revealing areas for improvement in the provision of care,” Veterans Health Administration (VHA) Directive 1190 states. “This ultimately contributes to organizational improvements.” At the same time, there are anecdotal concerns that PR may be used punitively and driven by case outcomes rather than by accepted best practices supporting QI.
Studies of the PR process suggest these concerns are valid. A key tenet of QI is standardization. PR is problematic in that regard; studies show poor interrater reliability for judgments on care, as well as hindsight bias—the fact that raters are strongly influenced by the outcome of care, not the process of care.2-5 There are concerns that case selection or review process when not standardized may be wielded as punitive too.6 In this study, we sought to identify the relationship between PR findings and subsequent institution quality metrics. If PR does lead to an improvement in quality, or if quality concerns are managed within the PR committee, it should be possible to identify a measurable relationship between the PR process and a facility’s subsequent quality measures.
A handful of studies describe the association between PR and quality of care. Itri and colleagues noted that random, not standardized PR in radiology does not achieve reductions in diagnostic error rate.7 However, adoption of just culture principles in PR resulted in a significant improvement in facility leaders’ self-reports of quality measures at surveyed institutions.8 The same author reported that increases in PR standardization and integration with performance improvement activities could explain up to 18% of objective quality measure variation.9
We sought to determine whether a specific aspect of the PR process, the PR committee judgment of quality of care by clinicians, was related to medical center quality in a cross-sectional study of 136 Veterans Health Administration (VHA) medical centers. The VHA is a good source of study because there are standardized PR processes and training for committee members and reviewers. Our hypothesis was that medical centers with a higher number of Level 2 (“most experienced and competent clinicians might have managed the case differently”) and Level 3 (“most experienced and competent providers would have managed the case differently”) PR findings would also have lower quality metric scores for processes and outcomes of care.
Methods
We used PR data from fiscal year 2018 and 2019. VHA PR data are available quarterly and are self-reported by each facility to the VHA Office of Clinical Risk Management. These data are broken down by facility. The following data, when available in both fiscal years 2018 and 2019, were used for this analysis: percent and number of PR that are ranked as level 1, 2, or 3; medical center group (MCG) acuity measure assigned by the VHA (1 is highest, 3 is lowest); and number of PR per 100,000 unique veteran encounters in 2019. Measures of facility quality are drawn from Strategic Analytics for Improvement and Learning (SAIL) data from 2019, which are available quarterly by facility and are rolling for 12 months. SAIL measures processes and outcomes of care. Table 1 indicates which measures are focused on outcomes vs quality processes.
SAS Version 9.2 was used to perform statistical analyses. We used Spearman correlation to estimate the PR and quality relationship.
Results
There were 136 facilities with 2 years of PR data available. The majority of these facilities (89) were highest complexity MCG 1 facilities; 19 were MCG 2, and 28 were MCG 3. Of 13,515 PRs, most of the 9555 PR findings were level 1 (70.7%). The between-facility range of level 2 and 3 findings was large, varying from 3.5% to nearly 70% in 2019 (Table 2). Findings were similar in 2018; facilities level 2 and 3 ratings ranged from 3.6% to 73.5% of all PR findings.
There was no correlation between most quality measures and facility PR findings (Table 3). The only exception was for Global Measures (GM90), an inpatient process of care measure. Unexpectedly, the correlation was positive—facilities with a higher percentage of level 2 and 3 PR findings had better inpatient processes of care SAIL score. The strongest correlation was between 2018 and 2019 PR findings.
Discussion
We hypothesized that a high percentage of level 2 and 3 PR findings would be negatively associated with objective facility measures of care processes in SAIL but we did not see this association. The only quality measure associated with PR findings was GM90, a score of inpatient care processes. However, the association was positive, with better performance associated with more level 2 and 3 PR findings.
The best predictor of the proportion of a facility’s PR findings is the previous year’s PR findings. With an R = 0.59, the previous year findings explain about 35% of the variability in level assignment. Our analysis may describe a new bias in PR, in which committees consistently assign either low or high proportions of level 2 and 3 findings. This correlation could be due to individual PR committee culture or composition, but it does not relate to objective quality measures.
Strengths
For this study we use objective measures of PR processes, the assignment of levels of care.
Limitations
Facilities self-report PR outcomes, so there could be errors in reporting. In addition, this study was cross sectional and not longitudinal and it is possible that change in quality measures over time are correlated with PR findings. Future studies using the VHA PR and SAIL data could evaluate whether changes over time, and perhaps in response to level 2 and 3 findings, would be a more sensitive indicator of the impact of the PR process on quality metrics. Future studies could incorporate the relationship between findings from the All Employee Survey, which is conducted annually, such as psychologic safety, as well as the distance the facility has gone on the high reliability organization journey, with PR findings and SAIL metrics. Finally, PR is focused on the practice of an individual clinician, while SAIL quality metrics reflect facility performance. Interventions possibly stay at the clinician level and do not drive subsequent QI processes.
What does this mean for PR? Since the early 1990s, there have been exhortations from experts to improve PR, by adopting a QI model, or for a deeper integration of PR and QI.1,2,10 Just culture tools, which include QI, are promoted as a means to improve PR.8,11,12 Other studies show PR remains problematic in terms of standardization, incorporation of best practices, redesigning systems of care, or demonstrable improvements to facility safety and care quality.1,4,6,8 Several publications have described interventions to improve PR. Deyo-Svedson discussed a program with standardized training and triggers, much like VHA.13 Itri and colleagues standardized PR in radiology to target areas of known diagnostic error, as well as use the issues assessed in PR to perform QI and education. One example of a successful QI effort involved changing the radiology reporting template to make sure areas that are prone to diagnostic error are addressed.7
Conclusions
Since 35% of PR level variance is correlated with prior year’s results, PR committees should look at increased standardization in reviews and findings. We endorse a strong focus on standardization, application of just culture tools to case reviews, and tighter linkage between process and outcome metrics measured by SAIL and PR case finding. Studies should be performed to pilot interventions to improve the linkage between PR and quality, so that greater and faster gains can be made in quality processes and, leading from this, outcomes. Additionally, future research should investigate why some facilities consistently choose higher or lower PR ratings.
Acknowledgments
We acknowledge Dr. George “Web” Ross for his helpful edits.
1. Edwards MT. In pursuit of quality and safety: an 8-year study of clinical peer review best practices in US hospitals. Int J Qual Health Care. 2018;30(8):602-607. doi:10.1093/intqhc/mzy069
2. Dans PE. Clinical peer Review: burnishing a tarnished icon. Ann Intern Med. 1993;118(7):566-568. doi:10.7326/0003-4819-118-7-199304010-00014
3. Goldman RL. The reliability of peer assessments of quality of care. JAMA. 1992;267(7):958-960. doi:10.1001/jama.1992.03480070074034
4. Swaroop R. Disrupting physician clinical practice peer review. Perm J. 2019;23:18-207. doi:10.7812/TPP/18-207
5. Caplan RA, Posner KL, Cheney FW. Effect of outcome on physician judgments of appropriateness of care. JAMA. 1991;265(15):1957–1960. doi:10.1001/jama.1991.03460150061024
6. Vyas D, Hozain AE. Clinical peer review in the United States: history, legal development and subsequent abuse. World J Gastroenterol. 2014;20(21):6357-6363. doi:10.3748/wjg.v20.i21.6357
7. Itri JN, Donithan A, Patel SH. Random versus nonrandom peer review: a case for more meaningful peer review. J Am Coll Radiol. 2018;15(7):1045-1052. doi:10.1016/j.jacr.2018.03.054
8. Edwards MT. An assessment of the impact of just culture on quality and safety in US hospitals. Am J Med Qual. 2018; 33(5):502-508. doi:10.1177/1062860618768057
9. Edwards MT. The objective impact of clinical peer review on hospital quality and safety. Am J Med Qual. 2011;26(2);110-119. doi:10.1177/1062860610380732
10. Berwick DM. Peer review and quality management: are they compatible?. QRB Qual Rev Bull. 1990;16(7):246-251. doi:10.1016/s0097-5990(16)30377-3
11. Volkar JK, Phrampus P, English D, et al. Institution of just culture physician peer review in an academic medical center. J Patient Saf. 2021;17(7):e689-e693. doi:10.1097/PTS.0000000000000449
12. Burns J, Miller T, Weiss JM, Erdfarb A, Silber D, Goldberg-Stein S. Just culture: practical implementation for radiologist peer review. J Am Coll Radiol. 2019;16(3):384-388. doi:10.1016/j.jacr.2018.10.021
13. Deyo-Svendsen ME, Phillips MR, Albright JK, et al. A systematic approach to clinical peer review in a critical access hospital. Qual Manag Health Care. 2016;25(4):213-218. doi:10.1097/QMH.0000000000000113
Hospital leaders report the most common aim of peer review (PR) is to improve quality and patient safety, thus it is a potentially powerful quality improvement (QI) driver.1 “When conducted systematically and credibly, peer review for quality management can result in both short-term and long-term improvements in patient care by revealing areas for improvement in the provision of care,” Veterans Health Administration (VHA) Directive 1190 states. “This ultimately contributes to organizational improvements.” At the same time, there are anecdotal concerns that PR may be used punitively and driven by case outcomes rather than by accepted best practices supporting QI.
Studies of the PR process suggest these concerns are valid. A key tenet of QI is standardization. PR is problematic in that regard; studies show poor interrater reliability for judgments on care, as well as hindsight bias—the fact that raters are strongly influenced by the outcome of care, not the process of care.2-5 There are concerns that case selection or review process when not standardized may be wielded as punitive too.6 In this study, we sought to identify the relationship between PR findings and subsequent institution quality metrics. If PR does lead to an improvement in quality, or if quality concerns are managed within the PR committee, it should be possible to identify a measurable relationship between the PR process and a facility’s subsequent quality measures.
A handful of studies describe the association between PR and quality of care. Itri and colleagues noted that random, not standardized PR in radiology does not achieve reductions in diagnostic error rate.7 However, adoption of just culture principles in PR resulted in a significant improvement in facility leaders’ self-reports of quality measures at surveyed institutions.8 The same author reported that increases in PR standardization and integration with performance improvement activities could explain up to 18% of objective quality measure variation.9
We sought to determine whether a specific aspect of the PR process, the PR committee judgment of quality of care by clinicians, was related to medical center quality in a cross-sectional study of 136 Veterans Health Administration (VHA) medical centers. The VHA is a good source of study because there are standardized PR processes and training for committee members and reviewers. Our hypothesis was that medical centers with a higher number of Level 2 (“most experienced and competent clinicians might have managed the case differently”) and Level 3 (“most experienced and competent providers would have managed the case differently”) PR findings would also have lower quality metric scores for processes and outcomes of care.
Methods
We used PR data from fiscal year 2018 and 2019. VHA PR data are available quarterly and are self-reported by each facility to the VHA Office of Clinical Risk Management. These data are broken down by facility. The following data, when available in both fiscal years 2018 and 2019, were used for this analysis: percent and number of PR that are ranked as level 1, 2, or 3; medical center group (MCG) acuity measure assigned by the VHA (1 is highest, 3 is lowest); and number of PR per 100,000 unique veteran encounters in 2019. Measures of facility quality are drawn from Strategic Analytics for Improvement and Learning (SAIL) data from 2019, which are available quarterly by facility and are rolling for 12 months. SAIL measures processes and outcomes of care. Table 1 indicates which measures are focused on outcomes vs quality processes.
SAS Version 9.2 was used to perform statistical analyses. We used Spearman correlation to estimate the PR and quality relationship.
Results
There were 136 facilities with 2 years of PR data available. The majority of these facilities (89) were highest complexity MCG 1 facilities; 19 were MCG 2, and 28 were MCG 3. Of 13,515 PRs, most of the 9555 PR findings were level 1 (70.7%). The between-facility range of level 2 and 3 findings was large, varying from 3.5% to nearly 70% in 2019 (Table 2). Findings were similar in 2018; facilities level 2 and 3 ratings ranged from 3.6% to 73.5% of all PR findings.
There was no correlation between most quality measures and facility PR findings (Table 3). The only exception was for Global Measures (GM90), an inpatient process of care measure. Unexpectedly, the correlation was positive—facilities with a higher percentage of level 2 and 3 PR findings had better inpatient processes of care SAIL score. The strongest correlation was between 2018 and 2019 PR findings.
Discussion
We hypothesized that a high percentage of level 2 and 3 PR findings would be negatively associated with objective facility measures of care processes in SAIL but we did not see this association. The only quality measure associated with PR findings was GM90, a score of inpatient care processes. However, the association was positive, with better performance associated with more level 2 and 3 PR findings.
The best predictor of the proportion of a facility’s PR findings is the previous year’s PR findings. With an R = 0.59, the previous year findings explain about 35% of the variability in level assignment. Our analysis may describe a new bias in PR, in which committees consistently assign either low or high proportions of level 2 and 3 findings. This correlation could be due to individual PR committee culture or composition, but it does not relate to objective quality measures.
Strengths
For this study we use objective measures of PR processes, the assignment of levels of care.
Limitations
Facilities self-report PR outcomes, so there could be errors in reporting. In addition, this study was cross sectional and not longitudinal and it is possible that change in quality measures over time are correlated with PR findings. Future studies using the VHA PR and SAIL data could evaluate whether changes over time, and perhaps in response to level 2 and 3 findings, would be a more sensitive indicator of the impact of the PR process on quality metrics. Future studies could incorporate the relationship between findings from the All Employee Survey, which is conducted annually, such as psychologic safety, as well as the distance the facility has gone on the high reliability organization journey, with PR findings and SAIL metrics. Finally, PR is focused on the practice of an individual clinician, while SAIL quality metrics reflect facility performance. Interventions possibly stay at the clinician level and do not drive subsequent QI processes.
What does this mean for PR? Since the early 1990s, there have been exhortations from experts to improve PR, by adopting a QI model, or for a deeper integration of PR and QI.1,2,10 Just culture tools, which include QI, are promoted as a means to improve PR.8,11,12 Other studies show PR remains problematic in terms of standardization, incorporation of best practices, redesigning systems of care, or demonstrable improvements to facility safety and care quality.1,4,6,8 Several publications have described interventions to improve PR. Deyo-Svedson discussed a program with standardized training and triggers, much like VHA.13 Itri and colleagues standardized PR in radiology to target areas of known diagnostic error, as well as use the issues assessed in PR to perform QI and education. One example of a successful QI effort involved changing the radiology reporting template to make sure areas that are prone to diagnostic error are addressed.7
Conclusions
Since 35% of PR level variance is correlated with prior year’s results, PR committees should look at increased standardization in reviews and findings. We endorse a strong focus on standardization, application of just culture tools to case reviews, and tighter linkage between process and outcome metrics measured by SAIL and PR case finding. Studies should be performed to pilot interventions to improve the linkage between PR and quality, so that greater and faster gains can be made in quality processes and, leading from this, outcomes. Additionally, future research should investigate why some facilities consistently choose higher or lower PR ratings.
Acknowledgments
We acknowledge Dr. George “Web” Ross for his helpful edits.
Hospital leaders report the most common aim of peer review (PR) is to improve quality and patient safety, thus it is a potentially powerful quality improvement (QI) driver.1 “When conducted systematically and credibly, peer review for quality management can result in both short-term and long-term improvements in patient care by revealing areas for improvement in the provision of care,” Veterans Health Administration (VHA) Directive 1190 states. “This ultimately contributes to organizational improvements.” At the same time, there are anecdotal concerns that PR may be used punitively and driven by case outcomes rather than by accepted best practices supporting QI.
Studies of the PR process suggest these concerns are valid. A key tenet of QI is standardization. PR is problematic in that regard; studies show poor interrater reliability for judgments on care, as well as hindsight bias—the fact that raters are strongly influenced by the outcome of care, not the process of care.2-5 There are concerns that case selection or review process when not standardized may be wielded as punitive too.6 In this study, we sought to identify the relationship between PR findings and subsequent institution quality metrics. If PR does lead to an improvement in quality, or if quality concerns are managed within the PR committee, it should be possible to identify a measurable relationship between the PR process and a facility’s subsequent quality measures.
A handful of studies describe the association between PR and quality of care. Itri and colleagues noted that random, not standardized PR in radiology does not achieve reductions in diagnostic error rate.7 However, adoption of just culture principles in PR resulted in a significant improvement in facility leaders’ self-reports of quality measures at surveyed institutions.8 The same author reported that increases in PR standardization and integration with performance improvement activities could explain up to 18% of objective quality measure variation.9
We sought to determine whether a specific aspect of the PR process, the PR committee judgment of quality of care by clinicians, was related to medical center quality in a cross-sectional study of 136 Veterans Health Administration (VHA) medical centers. The VHA is a good source of study because there are standardized PR processes and training for committee members and reviewers. Our hypothesis was that medical centers with a higher number of Level 2 (“most experienced and competent clinicians might have managed the case differently”) and Level 3 (“most experienced and competent providers would have managed the case differently”) PR findings would also have lower quality metric scores for processes and outcomes of care.
Methods
We used PR data from fiscal year 2018 and 2019. VHA PR data are available quarterly and are self-reported by each facility to the VHA Office of Clinical Risk Management. These data are broken down by facility. The following data, when available in both fiscal years 2018 and 2019, were used for this analysis: percent and number of PR that are ranked as level 1, 2, or 3; medical center group (MCG) acuity measure assigned by the VHA (1 is highest, 3 is lowest); and number of PR per 100,000 unique veteran encounters in 2019. Measures of facility quality are drawn from Strategic Analytics for Improvement and Learning (SAIL) data from 2019, which are available quarterly by facility and are rolling for 12 months. SAIL measures processes and outcomes of care. Table 1 indicates which measures are focused on outcomes vs quality processes.
SAS Version 9.2 was used to perform statistical analyses. We used Spearman correlation to estimate the PR and quality relationship.
Results
There were 136 facilities with 2 years of PR data available. The majority of these facilities (89) were highest complexity MCG 1 facilities; 19 were MCG 2, and 28 were MCG 3. Of 13,515 PRs, most of the 9555 PR findings were level 1 (70.7%). The between-facility range of level 2 and 3 findings was large, varying from 3.5% to nearly 70% in 2019 (Table 2). Findings were similar in 2018; facilities level 2 and 3 ratings ranged from 3.6% to 73.5% of all PR findings.
There was no correlation between most quality measures and facility PR findings (Table 3). The only exception was for Global Measures (GM90), an inpatient process of care measure. Unexpectedly, the correlation was positive—facilities with a higher percentage of level 2 and 3 PR findings had better inpatient processes of care SAIL score. The strongest correlation was between 2018 and 2019 PR findings.
Discussion
We hypothesized that a high percentage of level 2 and 3 PR findings would be negatively associated with objective facility measures of care processes in SAIL but we did not see this association. The only quality measure associated with PR findings was GM90, a score of inpatient care processes. However, the association was positive, with better performance associated with more level 2 and 3 PR findings.
The best predictor of the proportion of a facility’s PR findings is the previous year’s PR findings. With an R = 0.59, the previous year findings explain about 35% of the variability in level assignment. Our analysis may describe a new bias in PR, in which committees consistently assign either low or high proportions of level 2 and 3 findings. This correlation could be due to individual PR committee culture or composition, but it does not relate to objective quality measures.
Strengths
For this study we use objective measures of PR processes, the assignment of levels of care.
Limitations
Facilities self-report PR outcomes, so there could be errors in reporting. In addition, this study was cross sectional and not longitudinal and it is possible that change in quality measures over time are correlated with PR findings. Future studies using the VHA PR and SAIL data could evaluate whether changes over time, and perhaps in response to level 2 and 3 findings, would be a more sensitive indicator of the impact of the PR process on quality metrics. Future studies could incorporate the relationship between findings from the All Employee Survey, which is conducted annually, such as psychologic safety, as well as the distance the facility has gone on the high reliability organization journey, with PR findings and SAIL metrics. Finally, PR is focused on the practice of an individual clinician, while SAIL quality metrics reflect facility performance. Interventions possibly stay at the clinician level and do not drive subsequent QI processes.
What does this mean for PR? Since the early 1990s, there have been exhortations from experts to improve PR, by adopting a QI model, or for a deeper integration of PR and QI.1,2,10 Just culture tools, which include QI, are promoted as a means to improve PR.8,11,12 Other studies show PR remains problematic in terms of standardization, incorporation of best practices, redesigning systems of care, or demonstrable improvements to facility safety and care quality.1,4,6,8 Several publications have described interventions to improve PR. Deyo-Svedson discussed a program with standardized training and triggers, much like VHA.13 Itri and colleagues standardized PR in radiology to target areas of known diagnostic error, as well as use the issues assessed in PR to perform QI and education. One example of a successful QI effort involved changing the radiology reporting template to make sure areas that are prone to diagnostic error are addressed.7
Conclusions
Since 35% of PR level variance is correlated with prior year’s results, PR committees should look at increased standardization in reviews and findings. We endorse a strong focus on standardization, application of just culture tools to case reviews, and tighter linkage between process and outcome metrics measured by SAIL and PR case finding. Studies should be performed to pilot interventions to improve the linkage between PR and quality, so that greater and faster gains can be made in quality processes and, leading from this, outcomes. Additionally, future research should investigate why some facilities consistently choose higher or lower PR ratings.
Acknowledgments
We acknowledge Dr. George “Web” Ross for his helpful edits.
1. Edwards MT. In pursuit of quality and safety: an 8-year study of clinical peer review best practices in US hospitals. Int J Qual Health Care. 2018;30(8):602-607. doi:10.1093/intqhc/mzy069
2. Dans PE. Clinical peer Review: burnishing a tarnished icon. Ann Intern Med. 1993;118(7):566-568. doi:10.7326/0003-4819-118-7-199304010-00014
3. Goldman RL. The reliability of peer assessments of quality of care. JAMA. 1992;267(7):958-960. doi:10.1001/jama.1992.03480070074034
4. Swaroop R. Disrupting physician clinical practice peer review. Perm J. 2019;23:18-207. doi:10.7812/TPP/18-207
5. Caplan RA, Posner KL, Cheney FW. Effect of outcome on physician judgments of appropriateness of care. JAMA. 1991;265(15):1957–1960. doi:10.1001/jama.1991.03460150061024
6. Vyas D, Hozain AE. Clinical peer review in the United States: history, legal development and subsequent abuse. World J Gastroenterol. 2014;20(21):6357-6363. doi:10.3748/wjg.v20.i21.6357
7. Itri JN, Donithan A, Patel SH. Random versus nonrandom peer review: a case for more meaningful peer review. J Am Coll Radiol. 2018;15(7):1045-1052. doi:10.1016/j.jacr.2018.03.054
8. Edwards MT. An assessment of the impact of just culture on quality and safety in US hospitals. Am J Med Qual. 2018; 33(5):502-508. doi:10.1177/1062860618768057
9. Edwards MT. The objective impact of clinical peer review on hospital quality and safety. Am J Med Qual. 2011;26(2);110-119. doi:10.1177/1062860610380732
10. Berwick DM. Peer review and quality management: are they compatible?. QRB Qual Rev Bull. 1990;16(7):246-251. doi:10.1016/s0097-5990(16)30377-3
11. Volkar JK, Phrampus P, English D, et al. Institution of just culture physician peer review in an academic medical center. J Patient Saf. 2021;17(7):e689-e693. doi:10.1097/PTS.0000000000000449
12. Burns J, Miller T, Weiss JM, Erdfarb A, Silber D, Goldberg-Stein S. Just culture: practical implementation for radiologist peer review. J Am Coll Radiol. 2019;16(3):384-388. doi:10.1016/j.jacr.2018.10.021
13. Deyo-Svendsen ME, Phillips MR, Albright JK, et al. A systematic approach to clinical peer review in a critical access hospital. Qual Manag Health Care. 2016;25(4):213-218. doi:10.1097/QMH.0000000000000113
1. Edwards MT. In pursuit of quality and safety: an 8-year study of clinical peer review best practices in US hospitals. Int J Qual Health Care. 2018;30(8):602-607. doi:10.1093/intqhc/mzy069
2. Dans PE. Clinical peer Review: burnishing a tarnished icon. Ann Intern Med. 1993;118(7):566-568. doi:10.7326/0003-4819-118-7-199304010-00014
3. Goldman RL. The reliability of peer assessments of quality of care. JAMA. 1992;267(7):958-960. doi:10.1001/jama.1992.03480070074034
4. Swaroop R. Disrupting physician clinical practice peer review. Perm J. 2019;23:18-207. doi:10.7812/TPP/18-207
5. Caplan RA, Posner KL, Cheney FW. Effect of outcome on physician judgments of appropriateness of care. JAMA. 1991;265(15):1957–1960. doi:10.1001/jama.1991.03460150061024
6. Vyas D, Hozain AE. Clinical peer review in the United States: history, legal development and subsequent abuse. World J Gastroenterol. 2014;20(21):6357-6363. doi:10.3748/wjg.v20.i21.6357
7. Itri JN, Donithan A, Patel SH. Random versus nonrandom peer review: a case for more meaningful peer review. J Am Coll Radiol. 2018;15(7):1045-1052. doi:10.1016/j.jacr.2018.03.054
8. Edwards MT. An assessment of the impact of just culture on quality and safety in US hospitals. Am J Med Qual. 2018; 33(5):502-508. doi:10.1177/1062860618768057
9. Edwards MT. The objective impact of clinical peer review on hospital quality and safety. Am J Med Qual. 2011;26(2);110-119. doi:10.1177/1062860610380732
10. Berwick DM. Peer review and quality management: are they compatible?. QRB Qual Rev Bull. 1990;16(7):246-251. doi:10.1016/s0097-5990(16)30377-3
11. Volkar JK, Phrampus P, English D, et al. Institution of just culture physician peer review in an academic medical center. J Patient Saf. 2021;17(7):e689-e693. doi:10.1097/PTS.0000000000000449
12. Burns J, Miller T, Weiss JM, Erdfarb A, Silber D, Goldberg-Stein S. Just culture: practical implementation for radiologist peer review. J Am Coll Radiol. 2019;16(3):384-388. doi:10.1016/j.jacr.2018.10.021
13. Deyo-Svendsen ME, Phillips MR, Albright JK, et al. A systematic approach to clinical peer review in a critical access hospital. Qual Manag Health Care. 2016;25(4):213-218. doi:10.1097/QMH.0000000000000113
Ex–hospital porter a neglected giant of cancer research
We have a half-forgotten Indian immigrant to thank – a hospital night porter turned biochemist –for revolutionizing treatment of leukemia, the once deadly childhood scourge that is still the most common pediatric cancer.
Dr. Yellapragada SubbaRow has been called the “father of chemotherapy” for developing methotrexate, a powerful, inexpensive therapy for leukemia and other diseases, and he is celebrated for additional scientific achievements. Yet Dr. SubbaRow’s life was marked more by struggle than glory.
Born poor in southeastern India, he nearly succumbed to a tropical disease that killed two older brothers, and he didn’t focus on schoolwork until his father died. Later, prejudice dogged his years as an immigrant to the United States, and a blood clot took his life at the age of 53.
Scientifically, however, Dr. SubbaRow (pronounced sue-buh-rao) triumphed, despite mammoth challenges and a lack of recognition that persists to this day. National Cancer Research Month is a fitting time to look back on his extraordinary life and work and pay tribute to his accomplishments.
‘Yella,’ folic acid, and a paradigm shift
No one appreciates Dr. SubbaRow more than a cadre of Indian-born physicians who have kept his legacy alive in journal articles, presentations, and a Pulitzer Prize-winning book. Among them is author and oncologist Siddhartha Mukherjee, MD, who chronicled Dr. SubbaRow’s achievements in his New York Times No. 1 bestseller, “The Emperor of All Maladies: A Biography of Cancer.”
As Dr. Mukherjee wrote, Dr. SubbaRow was a “pioneer in many ways, a physician turned cellular physiologist, a chemist who had accidentally wandered into biology.” (Per Indian tradition, SubbaRow is the doctor’s first name, and Yellapragada is his surname, but medical literature uses SubbaRow as his cognomen, with some variations in spelling. Dr. Mukherjee wrote that his friends called him “Yella.”)
Dr. SubbaRow came to the United States in 1923, after enduring a difficult childhood and young adulthood. He’d survived bouts of religious fervor, childhood rebellion (including a bid to run away from home and become a banana trader), and a failed arranged marriage. His wife bore him a child who died in infancy. He left it all behind.
In Boston, medical officials rejected his degree. Broke, he worked for a time as a night porter at Brigham and Women’s Hospital in Boston, changing sheets and cleaning urinals. To a poor but proud high-caste Indian Brahmin, the culture shock of carrying out these tasks must have been especially jarring.
Dr. SubbaRow went on to earn a diploma from Harvard Medical School, also in Boston, and became a junior faculty member. As a foreigner, Dr. Mukherjee wrote, Dr. SubbaRow was a “reclusive, nocturnal, heavily accented vegetarian,” so different from his colleagues that advancement seemed impossible. Despite his pioneering biochemistry work, Harvard later declined to offer Dr. SubbaRow a tenured faculty position.
By the early 1940s, he took a job at an upstate New York pharmaceutical company called Lederle Labs (later purchased by Pfizer). At Lederle, Dr. SubbaRow strove to synthesize the vitamin known as folic acid. He ended up creating a kind of antivitamin, a lookalike that acted like folic acid but only succeeded in gumming up the works in receptors. But what good would it do to stop the body from absorbing folic acid? Plenty, it turned out.
Discoveries pile up, but credit and fame prove elusive
Dr. SubbaRow was no stranger to producing landmark biological work. He’d previously codiscovered phosphocreatine and ATP, which are crucial to muscular contractions. However, “in 1935, he had to disown the extent of his role in the discovery of the color test related to phosphorus, instead giving the credit to his co-author, who was being considered for promotion to a full professorship at Harvard,” wrote author Gerald Posner in his 2020 book, “Pharma: Greed, Lies and the Poisoning of America.”
Houston-area oncologist Kirtan Nautiyal, MD, who paid tribute to Dr. SubbaRow in a 2018 article, contended that “with his Indian instinct for self-effacement, he had irreparably sabotaged his own career.”
Dr. SubbaRow and his team also developed “the first effective treatment of filariasis, which causes elephantiasis of the lower limbs and genitals in millions of people, mainly in tropical countries,” Dr. Nautiyal wrote. “Later in the decade, his antibiotic program generated polymyxin, the first effective treatment against the class of bacteria called Gram negatives, and aureomycin, the first “broad-spectrum’ antibiotic.” (Aureomycin is also the first tetracycline antibiotic.)
Dr. SubbaRow’s discovery of a folic acid antagonist would again go largely unheralded. But first came the realization that folic acid made childhood leukemia worse, not better, and the prospect that this process could potentially be reversed.
Rise of methotrexate and fall of leukemia
In Boston, Sidney Farber, MD, a Boston pathologist, was desperate to help Robert Sandler, a 2-year-old leukemia patient. Dr. Farber contacted his ex-colleague Dr. SubbaRow to request a supply of aminopterin, an early version of methotrexate that Dr. SubbaRow and his team had developed. Dr. Farber injected Robert with the substance and within 3 days, the toddler’s white blood count started falling – fast. He stopped bleeding, resumed eating, and once again seemed almost identical to his twin brother, as Dr. Mukherjee wrote in his book.
Leukemia had never gone into remission before. Unfortunately, the treatment only worked temporarily. Robert, like other children treated with the drug, relapsed and died within months. But Dr. Farber “saw a door open” – a chemical, a kind of chemotherapy, that could turn back cancer. In the case of folic acid antagonists, they do so by stopping cancer cells from replicating.
Methotrexate, a related agent synthesized by Dr. SubbaRow, would become a mainstay of leukemia treatment and begin to produce long-term remission from acute lymphoblastic leukemia in 1970, when combination chemotherapy was developed.
Other cancers fell to methotrexate treatment. “Previous assumptions that cancer was nearly always fatal were revised, and the field of medical oncology (treatment of cancer with chemotherapy), which had not previously existed, was formally established in 1971,” according to the National Cancer Institute’s history of methotrexate. This account does not mention Dr. SubbaRow.
Death takes the doctor, but his legacy remains
In biographies, as well as his own words, Dr. SubbaRow comes across as a prickly, hard-driving workaholic who had little interest in intimate human connections. “It is not good to ask in every letter when I will be back,” he wrote to his wife back in India, before cutting off ties completely in the early 1930s. “I will come as early as possible. ... I do not want to write anything more.”
It seems, as his biographer S.P.K. Gupta noted, that “he was quite determined that the time allotted to him on Earth should be completely devoted to finding cures for ailments that plagued mankind.”
Still, Dr. SubbaRow’s research team was devoted to him, and he had plenty of reasons to be bitter, such as the prejudice and isolation he encountered in the United States and earlier, in British-run India. According to Mr. Posner’s book, even as a young medical student, Dr. SubbaRow heeded the call of Indian independence activist Mohandas Gandhi. He “refused the British surgical gown given him at school and instead donned a traditional and simple cotton Khadi. That act of defiance cost SubbaRow the college degree that was necessary for him to get into the State Medical College.”
During the last year of his life, Dr. SubbaRow faced yet another humiliation: In his landmark 1948 study about aminopterin as a treatment for leukemia, his colleague Dr. Farber failed to credit him, an “astonishing omission” as Yaddanapudi Ravindranath, MBBS, a pediatric hematologist/oncologist at Wayne State University, Detroit, put it. “From everything I know, Dr. Farber spent the rest of his career apologizing and trying to make amends for it,” Dr. Ravindranath said in an interview.
A career cut short, and a lasting legacy
In 1948, at the age of 53, Dr. SubbaRow suddenly died. “Many think Dr. SubbaRow would have won [the] Nobel Prize had he lived a few years longer,” said Dr. Ravindranath.
Like Dr. SubbaRow, Dr. Ravindranath was born in Andhra Pradesh state, near the city of Chennai formerly known as Madras. “Being a compatriot, in a way I continue his legacy, and I am obviously proud of him,” said Dr. Ravindranath, who has conducted his own landmark research regarding methotrexate and leukemia.
Nearly 75 years after Dr. SubbaRow’s death, Indian-born physicians like Dr. Ravindranath continue to honor him in print, trying to ensure that he’s not forgotten. Methotrexate remains a crucial treatment for leukemia, along with a long list of other ailments, including psoriasis.
Recognition for “Yella” may have come late and infrequently, but a Lederle Laboratories research library named after him offered Dr. SubbaRow a kind of immortality. A plaque there memorialized him in stone as a scientist, teacher, philosopher, and humanitarian, featuring the quote: “Science simply prolongs life. Religion deepens it.”
By all accounts, Dr. SubbaRow was a man of science and faith who had faith in science.
We have a half-forgotten Indian immigrant to thank – a hospital night porter turned biochemist –for revolutionizing treatment of leukemia, the once deadly childhood scourge that is still the most common pediatric cancer.
Dr. Yellapragada SubbaRow has been called the “father of chemotherapy” for developing methotrexate, a powerful, inexpensive therapy for leukemia and other diseases, and he is celebrated for additional scientific achievements. Yet Dr. SubbaRow’s life was marked more by struggle than glory.
Born poor in southeastern India, he nearly succumbed to a tropical disease that killed two older brothers, and he didn’t focus on schoolwork until his father died. Later, prejudice dogged his years as an immigrant to the United States, and a blood clot took his life at the age of 53.
Scientifically, however, Dr. SubbaRow (pronounced sue-buh-rao) triumphed, despite mammoth challenges and a lack of recognition that persists to this day. National Cancer Research Month is a fitting time to look back on his extraordinary life and work and pay tribute to his accomplishments.
‘Yella,’ folic acid, and a paradigm shift
No one appreciates Dr. SubbaRow more than a cadre of Indian-born physicians who have kept his legacy alive in journal articles, presentations, and a Pulitzer Prize-winning book. Among them is author and oncologist Siddhartha Mukherjee, MD, who chronicled Dr. SubbaRow’s achievements in his New York Times No. 1 bestseller, “The Emperor of All Maladies: A Biography of Cancer.”
As Dr. Mukherjee wrote, Dr. SubbaRow was a “pioneer in many ways, a physician turned cellular physiologist, a chemist who had accidentally wandered into biology.” (Per Indian tradition, SubbaRow is the doctor’s first name, and Yellapragada is his surname, but medical literature uses SubbaRow as his cognomen, with some variations in spelling. Dr. Mukherjee wrote that his friends called him “Yella.”)
Dr. SubbaRow came to the United States in 1923, after enduring a difficult childhood and young adulthood. He’d survived bouts of religious fervor, childhood rebellion (including a bid to run away from home and become a banana trader), and a failed arranged marriage. His wife bore him a child who died in infancy. He left it all behind.
In Boston, medical officials rejected his degree. Broke, he worked for a time as a night porter at Brigham and Women’s Hospital in Boston, changing sheets and cleaning urinals. To a poor but proud high-caste Indian Brahmin, the culture shock of carrying out these tasks must have been especially jarring.
Dr. SubbaRow went on to earn a diploma from Harvard Medical School, also in Boston, and became a junior faculty member. As a foreigner, Dr. Mukherjee wrote, Dr. SubbaRow was a “reclusive, nocturnal, heavily accented vegetarian,” so different from his colleagues that advancement seemed impossible. Despite his pioneering biochemistry work, Harvard later declined to offer Dr. SubbaRow a tenured faculty position.
By the early 1940s, he took a job at an upstate New York pharmaceutical company called Lederle Labs (later purchased by Pfizer). At Lederle, Dr. SubbaRow strove to synthesize the vitamin known as folic acid. He ended up creating a kind of antivitamin, a lookalike that acted like folic acid but only succeeded in gumming up the works in receptors. But what good would it do to stop the body from absorbing folic acid? Plenty, it turned out.
Discoveries pile up, but credit and fame prove elusive
Dr. SubbaRow was no stranger to producing landmark biological work. He’d previously codiscovered phosphocreatine and ATP, which are crucial to muscular contractions. However, “in 1935, he had to disown the extent of his role in the discovery of the color test related to phosphorus, instead giving the credit to his co-author, who was being considered for promotion to a full professorship at Harvard,” wrote author Gerald Posner in his 2020 book, “Pharma: Greed, Lies and the Poisoning of America.”
Houston-area oncologist Kirtan Nautiyal, MD, who paid tribute to Dr. SubbaRow in a 2018 article, contended that “with his Indian instinct for self-effacement, he had irreparably sabotaged his own career.”
Dr. SubbaRow and his team also developed “the first effective treatment of filariasis, which causes elephantiasis of the lower limbs and genitals in millions of people, mainly in tropical countries,” Dr. Nautiyal wrote. “Later in the decade, his antibiotic program generated polymyxin, the first effective treatment against the class of bacteria called Gram negatives, and aureomycin, the first “broad-spectrum’ antibiotic.” (Aureomycin is also the first tetracycline antibiotic.)
Dr. SubbaRow’s discovery of a folic acid antagonist would again go largely unheralded. But first came the realization that folic acid made childhood leukemia worse, not better, and the prospect that this process could potentially be reversed.
Rise of methotrexate and fall of leukemia
In Boston, Sidney Farber, MD, a Boston pathologist, was desperate to help Robert Sandler, a 2-year-old leukemia patient. Dr. Farber contacted his ex-colleague Dr. SubbaRow to request a supply of aminopterin, an early version of methotrexate that Dr. SubbaRow and his team had developed. Dr. Farber injected Robert with the substance and within 3 days, the toddler’s white blood count started falling – fast. He stopped bleeding, resumed eating, and once again seemed almost identical to his twin brother, as Dr. Mukherjee wrote in his book.
Leukemia had never gone into remission before. Unfortunately, the treatment only worked temporarily. Robert, like other children treated with the drug, relapsed and died within months. But Dr. Farber “saw a door open” – a chemical, a kind of chemotherapy, that could turn back cancer. In the case of folic acid antagonists, they do so by stopping cancer cells from replicating.
Methotrexate, a related agent synthesized by Dr. SubbaRow, would become a mainstay of leukemia treatment and begin to produce long-term remission from acute lymphoblastic leukemia in 1970, when combination chemotherapy was developed.
Other cancers fell to methotrexate treatment. “Previous assumptions that cancer was nearly always fatal were revised, and the field of medical oncology (treatment of cancer with chemotherapy), which had not previously existed, was formally established in 1971,” according to the National Cancer Institute’s history of methotrexate. This account does not mention Dr. SubbaRow.
Death takes the doctor, but his legacy remains
In biographies, as well as his own words, Dr. SubbaRow comes across as a prickly, hard-driving workaholic who had little interest in intimate human connections. “It is not good to ask in every letter when I will be back,” he wrote to his wife back in India, before cutting off ties completely in the early 1930s. “I will come as early as possible. ... I do not want to write anything more.”
It seems, as his biographer S.P.K. Gupta noted, that “he was quite determined that the time allotted to him on Earth should be completely devoted to finding cures for ailments that plagued mankind.”
Still, Dr. SubbaRow’s research team was devoted to him, and he had plenty of reasons to be bitter, such as the prejudice and isolation he encountered in the United States and earlier, in British-run India. According to Mr. Posner’s book, even as a young medical student, Dr. SubbaRow heeded the call of Indian independence activist Mohandas Gandhi. He “refused the British surgical gown given him at school and instead donned a traditional and simple cotton Khadi. That act of defiance cost SubbaRow the college degree that was necessary for him to get into the State Medical College.”
During the last year of his life, Dr. SubbaRow faced yet another humiliation: In his landmark 1948 study about aminopterin as a treatment for leukemia, his colleague Dr. Farber failed to credit him, an “astonishing omission” as Yaddanapudi Ravindranath, MBBS, a pediatric hematologist/oncologist at Wayne State University, Detroit, put it. “From everything I know, Dr. Farber spent the rest of his career apologizing and trying to make amends for it,” Dr. Ravindranath said in an interview.
A career cut short, and a lasting legacy
In 1948, at the age of 53, Dr. SubbaRow suddenly died. “Many think Dr. SubbaRow would have won [the] Nobel Prize had he lived a few years longer,” said Dr. Ravindranath.
Like Dr. SubbaRow, Dr. Ravindranath was born in Andhra Pradesh state, near the city of Chennai formerly known as Madras. “Being a compatriot, in a way I continue his legacy, and I am obviously proud of him,” said Dr. Ravindranath, who has conducted his own landmark research regarding methotrexate and leukemia.
Nearly 75 years after Dr. SubbaRow’s death, Indian-born physicians like Dr. Ravindranath continue to honor him in print, trying to ensure that he’s not forgotten. Methotrexate remains a crucial treatment for leukemia, along with a long list of other ailments, including psoriasis.
Recognition for “Yella” may have come late and infrequently, but a Lederle Laboratories research library named after him offered Dr. SubbaRow a kind of immortality. A plaque there memorialized him in stone as a scientist, teacher, philosopher, and humanitarian, featuring the quote: “Science simply prolongs life. Religion deepens it.”
By all accounts, Dr. SubbaRow was a man of science and faith who had faith in science.
We have a half-forgotten Indian immigrant to thank – a hospital night porter turned biochemist –for revolutionizing treatment of leukemia, the once deadly childhood scourge that is still the most common pediatric cancer.
Dr. Yellapragada SubbaRow has been called the “father of chemotherapy” for developing methotrexate, a powerful, inexpensive therapy for leukemia and other diseases, and he is celebrated for additional scientific achievements. Yet Dr. SubbaRow’s life was marked more by struggle than glory.
Born poor in southeastern India, he nearly succumbed to a tropical disease that killed two older brothers, and he didn’t focus on schoolwork until his father died. Later, prejudice dogged his years as an immigrant to the United States, and a blood clot took his life at the age of 53.
Scientifically, however, Dr. SubbaRow (pronounced sue-buh-rao) triumphed, despite mammoth challenges and a lack of recognition that persists to this day. National Cancer Research Month is a fitting time to look back on his extraordinary life and work and pay tribute to his accomplishments.
‘Yella,’ folic acid, and a paradigm shift
No one appreciates Dr. SubbaRow more than a cadre of Indian-born physicians who have kept his legacy alive in journal articles, presentations, and a Pulitzer Prize-winning book. Among them is author and oncologist Siddhartha Mukherjee, MD, who chronicled Dr. SubbaRow’s achievements in his New York Times No. 1 bestseller, “The Emperor of All Maladies: A Biography of Cancer.”
As Dr. Mukherjee wrote, Dr. SubbaRow was a “pioneer in many ways, a physician turned cellular physiologist, a chemist who had accidentally wandered into biology.” (Per Indian tradition, SubbaRow is the doctor’s first name, and Yellapragada is his surname, but medical literature uses SubbaRow as his cognomen, with some variations in spelling. Dr. Mukherjee wrote that his friends called him “Yella.”)
Dr. SubbaRow came to the United States in 1923, after enduring a difficult childhood and young adulthood. He’d survived bouts of religious fervor, childhood rebellion (including a bid to run away from home and become a banana trader), and a failed arranged marriage. His wife bore him a child who died in infancy. He left it all behind.
In Boston, medical officials rejected his degree. Broke, he worked for a time as a night porter at Brigham and Women’s Hospital in Boston, changing sheets and cleaning urinals. To a poor but proud high-caste Indian Brahmin, the culture shock of carrying out these tasks must have been especially jarring.
Dr. SubbaRow went on to earn a diploma from Harvard Medical School, also in Boston, and became a junior faculty member. As a foreigner, Dr. Mukherjee wrote, Dr. SubbaRow was a “reclusive, nocturnal, heavily accented vegetarian,” so different from his colleagues that advancement seemed impossible. Despite his pioneering biochemistry work, Harvard later declined to offer Dr. SubbaRow a tenured faculty position.
By the early 1940s, he took a job at an upstate New York pharmaceutical company called Lederle Labs (later purchased by Pfizer). At Lederle, Dr. SubbaRow strove to synthesize the vitamin known as folic acid. He ended up creating a kind of antivitamin, a lookalike that acted like folic acid but only succeeded in gumming up the works in receptors. But what good would it do to stop the body from absorbing folic acid? Plenty, it turned out.
Discoveries pile up, but credit and fame prove elusive
Dr. SubbaRow was no stranger to producing landmark biological work. He’d previously codiscovered phosphocreatine and ATP, which are crucial to muscular contractions. However, “in 1935, he had to disown the extent of his role in the discovery of the color test related to phosphorus, instead giving the credit to his co-author, who was being considered for promotion to a full professorship at Harvard,” wrote author Gerald Posner in his 2020 book, “Pharma: Greed, Lies and the Poisoning of America.”
Houston-area oncologist Kirtan Nautiyal, MD, who paid tribute to Dr. SubbaRow in a 2018 article, contended that “with his Indian instinct for self-effacement, he had irreparably sabotaged his own career.”
Dr. SubbaRow and his team also developed “the first effective treatment of filariasis, which causes elephantiasis of the lower limbs and genitals in millions of people, mainly in tropical countries,” Dr. Nautiyal wrote. “Later in the decade, his antibiotic program generated polymyxin, the first effective treatment against the class of bacteria called Gram negatives, and aureomycin, the first “broad-spectrum’ antibiotic.” (Aureomycin is also the first tetracycline antibiotic.)
Dr. SubbaRow’s discovery of a folic acid antagonist would again go largely unheralded. But first came the realization that folic acid made childhood leukemia worse, not better, and the prospect that this process could potentially be reversed.
Rise of methotrexate and fall of leukemia
In Boston, Sidney Farber, MD, a Boston pathologist, was desperate to help Robert Sandler, a 2-year-old leukemia patient. Dr. Farber contacted his ex-colleague Dr. SubbaRow to request a supply of aminopterin, an early version of methotrexate that Dr. SubbaRow and his team had developed. Dr. Farber injected Robert with the substance and within 3 days, the toddler’s white blood count started falling – fast. He stopped bleeding, resumed eating, and once again seemed almost identical to his twin brother, as Dr. Mukherjee wrote in his book.
Leukemia had never gone into remission before. Unfortunately, the treatment only worked temporarily. Robert, like other children treated with the drug, relapsed and died within months. But Dr. Farber “saw a door open” – a chemical, a kind of chemotherapy, that could turn back cancer. In the case of folic acid antagonists, they do so by stopping cancer cells from replicating.
Methotrexate, a related agent synthesized by Dr. SubbaRow, would become a mainstay of leukemia treatment and begin to produce long-term remission from acute lymphoblastic leukemia in 1970, when combination chemotherapy was developed.
Other cancers fell to methotrexate treatment. “Previous assumptions that cancer was nearly always fatal were revised, and the field of medical oncology (treatment of cancer with chemotherapy), which had not previously existed, was formally established in 1971,” according to the National Cancer Institute’s history of methotrexate. This account does not mention Dr. SubbaRow.
Death takes the doctor, but his legacy remains
In biographies, as well as his own words, Dr. SubbaRow comes across as a prickly, hard-driving workaholic who had little interest in intimate human connections. “It is not good to ask in every letter when I will be back,” he wrote to his wife back in India, before cutting off ties completely in the early 1930s. “I will come as early as possible. ... I do not want to write anything more.”
It seems, as his biographer S.P.K. Gupta noted, that “he was quite determined that the time allotted to him on Earth should be completely devoted to finding cures for ailments that plagued mankind.”
Still, Dr. SubbaRow’s research team was devoted to him, and he had plenty of reasons to be bitter, such as the prejudice and isolation he encountered in the United States and earlier, in British-run India. According to Mr. Posner’s book, even as a young medical student, Dr. SubbaRow heeded the call of Indian independence activist Mohandas Gandhi. He “refused the British surgical gown given him at school and instead donned a traditional and simple cotton Khadi. That act of defiance cost SubbaRow the college degree that was necessary for him to get into the State Medical College.”
During the last year of his life, Dr. SubbaRow faced yet another humiliation: In his landmark 1948 study about aminopterin as a treatment for leukemia, his colleague Dr. Farber failed to credit him, an “astonishing omission” as Yaddanapudi Ravindranath, MBBS, a pediatric hematologist/oncologist at Wayne State University, Detroit, put it. “From everything I know, Dr. Farber spent the rest of his career apologizing and trying to make amends for it,” Dr. Ravindranath said in an interview.
A career cut short, and a lasting legacy
In 1948, at the age of 53, Dr. SubbaRow suddenly died. “Many think Dr. SubbaRow would have won [the] Nobel Prize had he lived a few years longer,” said Dr. Ravindranath.
Like Dr. SubbaRow, Dr. Ravindranath was born in Andhra Pradesh state, near the city of Chennai formerly known as Madras. “Being a compatriot, in a way I continue his legacy, and I am obviously proud of him,” said Dr. Ravindranath, who has conducted his own landmark research regarding methotrexate and leukemia.
Nearly 75 years after Dr. SubbaRow’s death, Indian-born physicians like Dr. Ravindranath continue to honor him in print, trying to ensure that he’s not forgotten. Methotrexate remains a crucial treatment for leukemia, along with a long list of other ailments, including psoriasis.
Recognition for “Yella” may have come late and infrequently, but a Lederle Laboratories research library named after him offered Dr. SubbaRow a kind of immortality. A plaque there memorialized him in stone as a scientist, teacher, philosopher, and humanitarian, featuring the quote: “Science simply prolongs life. Religion deepens it.”
By all accounts, Dr. SubbaRow was a man of science and faith who had faith in science.
Medical education programs tell how climate change affects health
Ms. Manivannan, copresident of Emory Medical Students for Climate Action, was in the first class of Emory’s medical students to experience the birth of a refined curriculum – lobbied for and partially created by students themselves. The new course of study addresses the myriad ways climate affects health: from air pollution and its effects on the lungs and cardiovascular system to heat-related kidney disease.
“We have known that climate has affected health for decades,” Ms. Manivannan said in a recent interview. “The narrative used to be that icebergs were melting and in 2050 polar bears would be extinct. The piece that’s different now is people are linking climate to increases in asthma and various diseases. We have a way to directly communicate that it’s not a far-off thing. It’s happening to your friends and family right now.”
Hospitals, medical schools, and public health programs are stepping up to educate the next generation of doctors as well as veteran medical workers on one of the most widespread, insidious health threats of our time – climate change – and specific ways it could affect their patients.
Although climate change may seem to many Americans like a distant threat, Marilyn Howarth, MD, a pediatrician in Philadelphia, is trying to make sure physicians are better prepared to treat a growing number of health problems associated with global warming.
“There isn’t a lot of education for pediatricians and internists on environmental health issues. It has not been a standard part of education in medical school or residency training,” Dr. Howarth, deputy director of the new Philadelphia Regional Center for Children’s Environmental Health, said. “With increasing attention on our climate, we really recognize there’s a real gap in physician knowledge, both in pediatric and adult care.”
Scientists have found that climate change can alter just about every system within the human body. Studies show that more extreme weather events, such as heat waves, thunderstorms, and floods, can worsen asthma and produce more pollen and mold, triggering debilitating respiratory problems.
According to the American Lung Association, ultrafine particles of air pollution can be inhaled and then travel throughout the bloodstream, wreaking havoc on organs and increasing risk of heart attack and stroke. Various types of air pollution also cause changes to the climate by trapping heat in the atmosphere, which leads to problems such as rising sea levels and extreme weather. Plus, in a new study published in Nature, scientists warn that warming climates are forcing animals to migrate to different areas, raising the risk that new infectious diseases will hop from animals – such as bats – to humans, a process called “zoonotic spillover” that many researchers believe is responsible for the COVID-19 pandemic.
The Philadelphia Regional Center for Children’s Environmental Health
One of the latest initiatives aimed at disseminating information about children’s health to health care providers is the Philadelphia Regional Center for Children’s Environmental Health, part of Children’s Hospital of Philadelphia and Penn Medicine. CHOP and Penn Medicine are jointly funding this center’s work, which will include educating health care providers on how to better screen for climate-caused health risks and treat related conditions, such as lead poisoning and asthma.
Outreach will focus on providers who treat patients with illnesses that researchers have linked to climate change, Dr. Howarth said. The center will offer clinicians access to seminars and webinars, along with online resources to help doctors treat environmental illnesses. For example, doctors at CHOP’s Poison Control Center are developing a toolkit for physicians to treat patients with elevated levels of lead in the blood. Scientists have linked extreme weather events related to climate change to flooding that pushes metals away from river banks where they were previously contained, allowing them to more easily contaminate homes, soils, and yards.
The initiative builds on CHOP’s Community Asthma Prevention Program (CAPP), which was launched in 1997 by Tyra Bryant-Stephens, MD, its current medical director. CAPP deploys community health workers into homes armed with supplies and tips for managing asthma. The new center will use similar tactics to provide education and resources to patients. The goal is to reach as many at-risk local children as possible.
Future generation of doctors fuel growth in climate change education
Lisa Doggett, MD, cofounder and president of the board of directors of Texas Physicians for Social Responsibility, announced in March that the University of Texas at Austin, Baylor College of Medicine, Houston, and the University of Texas Southwestern in Dallas have all decided to begin offering a course on environmental threats. Emory’s new curriculum has become more comprehensive every year since its start – thanks in part to the input of students like Ms. Manivannan. Faculty members tasked her with approving the new additions to the curriculum on how climate affects health, which in 2019 had consisted of a few slides about issues such as extreme heat exposure and air pollution and their effects on childbirth outcomes.
Material on climate change has now been woven into 13 courses. It is discussed at length in relation to pulmonology, cardiology, and gastropulmonology, for example, said Rebecca Philipsborn, MD, MPA, FAAP, faculty lead for the environmental and health curriculum at Emory.
The curriculum has only been incorporated into Emory’s program for the past 2 years. Dr. Philipsborn said the school plans to expand it to the clinical years to help trainees learn to treat conditions such as pediatric asthma.
“In the past few years, there has been so much momentum, and part of that is a testament to already seeing effects of climate change and how they affect delivery of health care,” she said.
At least one medical journal has recently ramped up its efforts to educate physicians on the links between health issues and climate change. Editors of Family Practice, from Oxford University Press, have announced that they plan to publish a special Climate Crisis and Primary Health Care issue in September.
Of course, not all climate initiatives in medicine are new. A select few have existed for decades.
But only now are physicians widely seeing the links between health and environment, according to Aaron Bernstein, MD, MPH, interim director of the Center for Climate, Health, and the Global Environment (C-CHANGE) at Harvard School of Public Health, Boston.
C-CHANGE, founded in 1996, was the first center in the world to focus on the health effects of environmental change.
“It’s taken 20 years, but what we’re seeing, I think, is the fruits of education,” Dr. Bernstein said. “There’s clearly a wave building here, and I think it really started with education and people younger than the people in charge calling them into account.”
Like the Philadelphia center, Harvard’s program conducts research on climate and health and educates people from high schoolers to health care veterans. Dr. Bernstein helps lead Climate MD, a program that aims to prepare health care workers for climate crises. The Climate MD team has published several articles in peer-reviewed journals on how to better treat patients struggling with environmental health problems. For example, an article on mapping patients in hurricane zones helped shed light on how systems can identify climate-vulnerable patients using public data.
They also developed a tool to help pediatricians provide “climate-informed primary care” – guidance on how to assess whether children are at risk of any harmful environmental exposures, a feature that is not part of standard pediatric visits.
Like the other programs, Climate MD uses community outreach to treat as many local patients as possible. Staff work with providers at more than 100 health clinics, particularly in areas where climate change disproportionately affects residents.
The next major step is to bring some of this into clinical practice, Dr. Bernstein said. In February 2020, C-CHANGE held its first symposium to address that issue.
“The key is to understand climate issues from a provider’s perspective,” he said. “Then those issues can really be brought to the bedside.”
A version of this article first appeared on Medscape.com.
Ms. Manivannan, copresident of Emory Medical Students for Climate Action, was in the first class of Emory’s medical students to experience the birth of a refined curriculum – lobbied for and partially created by students themselves. The new course of study addresses the myriad ways climate affects health: from air pollution and its effects on the lungs and cardiovascular system to heat-related kidney disease.
“We have known that climate has affected health for decades,” Ms. Manivannan said in a recent interview. “The narrative used to be that icebergs were melting and in 2050 polar bears would be extinct. The piece that’s different now is people are linking climate to increases in asthma and various diseases. We have a way to directly communicate that it’s not a far-off thing. It’s happening to your friends and family right now.”
Hospitals, medical schools, and public health programs are stepping up to educate the next generation of doctors as well as veteran medical workers on one of the most widespread, insidious health threats of our time – climate change – and specific ways it could affect their patients.
Although climate change may seem to many Americans like a distant threat, Marilyn Howarth, MD, a pediatrician in Philadelphia, is trying to make sure physicians are better prepared to treat a growing number of health problems associated with global warming.
“There isn’t a lot of education for pediatricians and internists on environmental health issues. It has not been a standard part of education in medical school or residency training,” Dr. Howarth, deputy director of the new Philadelphia Regional Center for Children’s Environmental Health, said. “With increasing attention on our climate, we really recognize there’s a real gap in physician knowledge, both in pediatric and adult care.”
Scientists have found that climate change can alter just about every system within the human body. Studies show that more extreme weather events, such as heat waves, thunderstorms, and floods, can worsen asthma and produce more pollen and mold, triggering debilitating respiratory problems.
According to the American Lung Association, ultrafine particles of air pollution can be inhaled and then travel throughout the bloodstream, wreaking havoc on organs and increasing risk of heart attack and stroke. Various types of air pollution also cause changes to the climate by trapping heat in the atmosphere, which leads to problems such as rising sea levels and extreme weather. Plus, in a new study published in Nature, scientists warn that warming climates are forcing animals to migrate to different areas, raising the risk that new infectious diseases will hop from animals – such as bats – to humans, a process called “zoonotic spillover” that many researchers believe is responsible for the COVID-19 pandemic.
The Philadelphia Regional Center for Children’s Environmental Health
One of the latest initiatives aimed at disseminating information about children’s health to health care providers is the Philadelphia Regional Center for Children’s Environmental Health, part of Children’s Hospital of Philadelphia and Penn Medicine. CHOP and Penn Medicine are jointly funding this center’s work, which will include educating health care providers on how to better screen for climate-caused health risks and treat related conditions, such as lead poisoning and asthma.
Outreach will focus on providers who treat patients with illnesses that researchers have linked to climate change, Dr. Howarth said. The center will offer clinicians access to seminars and webinars, along with online resources to help doctors treat environmental illnesses. For example, doctors at CHOP’s Poison Control Center are developing a toolkit for physicians to treat patients with elevated levels of lead in the blood. Scientists have linked extreme weather events related to climate change to flooding that pushes metals away from river banks where they were previously contained, allowing them to more easily contaminate homes, soils, and yards.
The initiative builds on CHOP’s Community Asthma Prevention Program (CAPP), which was launched in 1997 by Tyra Bryant-Stephens, MD, its current medical director. CAPP deploys community health workers into homes armed with supplies and tips for managing asthma. The new center will use similar tactics to provide education and resources to patients. The goal is to reach as many at-risk local children as possible.
Future generation of doctors fuel growth in climate change education
Lisa Doggett, MD, cofounder and president of the board of directors of Texas Physicians for Social Responsibility, announced in March that the University of Texas at Austin, Baylor College of Medicine, Houston, and the University of Texas Southwestern in Dallas have all decided to begin offering a course on environmental threats. Emory’s new curriculum has become more comprehensive every year since its start – thanks in part to the input of students like Ms. Manivannan. Faculty members tasked her with approving the new additions to the curriculum on how climate affects health, which in 2019 had consisted of a few slides about issues such as extreme heat exposure and air pollution and their effects on childbirth outcomes.
Material on climate change has now been woven into 13 courses. It is discussed at length in relation to pulmonology, cardiology, and gastropulmonology, for example, said Rebecca Philipsborn, MD, MPA, FAAP, faculty lead for the environmental and health curriculum at Emory.
The curriculum has only been incorporated into Emory’s program for the past 2 years. Dr. Philipsborn said the school plans to expand it to the clinical years to help trainees learn to treat conditions such as pediatric asthma.
“In the past few years, there has been so much momentum, and part of that is a testament to already seeing effects of climate change and how they affect delivery of health care,” she said.
At least one medical journal has recently ramped up its efforts to educate physicians on the links between health issues and climate change. Editors of Family Practice, from Oxford University Press, have announced that they plan to publish a special Climate Crisis and Primary Health Care issue in September.
Of course, not all climate initiatives in medicine are new. A select few have existed for decades.
But only now are physicians widely seeing the links between health and environment, according to Aaron Bernstein, MD, MPH, interim director of the Center for Climate, Health, and the Global Environment (C-CHANGE) at Harvard School of Public Health, Boston.
C-CHANGE, founded in 1996, was the first center in the world to focus on the health effects of environmental change.
“It’s taken 20 years, but what we’re seeing, I think, is the fruits of education,” Dr. Bernstein said. “There’s clearly a wave building here, and I think it really started with education and people younger than the people in charge calling them into account.”
Like the Philadelphia center, Harvard’s program conducts research on climate and health and educates people from high schoolers to health care veterans. Dr. Bernstein helps lead Climate MD, a program that aims to prepare health care workers for climate crises. The Climate MD team has published several articles in peer-reviewed journals on how to better treat patients struggling with environmental health problems. For example, an article on mapping patients in hurricane zones helped shed light on how systems can identify climate-vulnerable patients using public data.
They also developed a tool to help pediatricians provide “climate-informed primary care” – guidance on how to assess whether children are at risk of any harmful environmental exposures, a feature that is not part of standard pediatric visits.
Like the other programs, Climate MD uses community outreach to treat as many local patients as possible. Staff work with providers at more than 100 health clinics, particularly in areas where climate change disproportionately affects residents.
The next major step is to bring some of this into clinical practice, Dr. Bernstein said. In February 2020, C-CHANGE held its first symposium to address that issue.
“The key is to understand climate issues from a provider’s perspective,” he said. “Then those issues can really be brought to the bedside.”
A version of this article first appeared on Medscape.com.
Ms. Manivannan, copresident of Emory Medical Students for Climate Action, was in the first class of Emory’s medical students to experience the birth of a refined curriculum – lobbied for and partially created by students themselves. The new course of study addresses the myriad ways climate affects health: from air pollution and its effects on the lungs and cardiovascular system to heat-related kidney disease.
“We have known that climate has affected health for decades,” Ms. Manivannan said in a recent interview. “The narrative used to be that icebergs were melting and in 2050 polar bears would be extinct. The piece that’s different now is people are linking climate to increases in asthma and various diseases. We have a way to directly communicate that it’s not a far-off thing. It’s happening to your friends and family right now.”
Hospitals, medical schools, and public health programs are stepping up to educate the next generation of doctors as well as veteran medical workers on one of the most widespread, insidious health threats of our time – climate change – and specific ways it could affect their patients.
Although climate change may seem to many Americans like a distant threat, Marilyn Howarth, MD, a pediatrician in Philadelphia, is trying to make sure physicians are better prepared to treat a growing number of health problems associated with global warming.
“There isn’t a lot of education for pediatricians and internists on environmental health issues. It has not been a standard part of education in medical school or residency training,” Dr. Howarth, deputy director of the new Philadelphia Regional Center for Children’s Environmental Health, said. “With increasing attention on our climate, we really recognize there’s a real gap in physician knowledge, both in pediatric and adult care.”
Scientists have found that climate change can alter just about every system within the human body. Studies show that more extreme weather events, such as heat waves, thunderstorms, and floods, can worsen asthma and produce more pollen and mold, triggering debilitating respiratory problems.
According to the American Lung Association, ultrafine particles of air pollution can be inhaled and then travel throughout the bloodstream, wreaking havoc on organs and increasing risk of heart attack and stroke. Various types of air pollution also cause changes to the climate by trapping heat in the atmosphere, which leads to problems such as rising sea levels and extreme weather. Plus, in a new study published in Nature, scientists warn that warming climates are forcing animals to migrate to different areas, raising the risk that new infectious diseases will hop from animals – such as bats – to humans, a process called “zoonotic spillover” that many researchers believe is responsible for the COVID-19 pandemic.
The Philadelphia Regional Center for Children’s Environmental Health
One of the latest initiatives aimed at disseminating information about children’s health to health care providers is the Philadelphia Regional Center for Children’s Environmental Health, part of Children’s Hospital of Philadelphia and Penn Medicine. CHOP and Penn Medicine are jointly funding this center’s work, which will include educating health care providers on how to better screen for climate-caused health risks and treat related conditions, such as lead poisoning and asthma.
Outreach will focus on providers who treat patients with illnesses that researchers have linked to climate change, Dr. Howarth said. The center will offer clinicians access to seminars and webinars, along with online resources to help doctors treat environmental illnesses. For example, doctors at CHOP’s Poison Control Center are developing a toolkit for physicians to treat patients with elevated levels of lead in the blood. Scientists have linked extreme weather events related to climate change to flooding that pushes metals away from river banks where they were previously contained, allowing them to more easily contaminate homes, soils, and yards.
The initiative builds on CHOP’s Community Asthma Prevention Program (CAPP), which was launched in 1997 by Tyra Bryant-Stephens, MD, its current medical director. CAPP deploys community health workers into homes armed with supplies and tips for managing asthma. The new center will use similar tactics to provide education and resources to patients. The goal is to reach as many at-risk local children as possible.
Future generation of doctors fuel growth in climate change education
Lisa Doggett, MD, cofounder and president of the board of directors of Texas Physicians for Social Responsibility, announced in March that the University of Texas at Austin, Baylor College of Medicine, Houston, and the University of Texas Southwestern in Dallas have all decided to begin offering a course on environmental threats. Emory’s new curriculum has become more comprehensive every year since its start – thanks in part to the input of students like Ms. Manivannan. Faculty members tasked her with approving the new additions to the curriculum on how climate affects health, which in 2019 had consisted of a few slides about issues such as extreme heat exposure and air pollution and their effects on childbirth outcomes.
Material on climate change has now been woven into 13 courses. It is discussed at length in relation to pulmonology, cardiology, and gastropulmonology, for example, said Rebecca Philipsborn, MD, MPA, FAAP, faculty lead for the environmental and health curriculum at Emory.
The curriculum has only been incorporated into Emory’s program for the past 2 years. Dr. Philipsborn said the school plans to expand it to the clinical years to help trainees learn to treat conditions such as pediatric asthma.
“In the past few years, there has been so much momentum, and part of that is a testament to already seeing effects of climate change and how they affect delivery of health care,” she said.
At least one medical journal has recently ramped up its efforts to educate physicians on the links between health issues and climate change. Editors of Family Practice, from Oxford University Press, have announced that they plan to publish a special Climate Crisis and Primary Health Care issue in September.
Of course, not all climate initiatives in medicine are new. A select few have existed for decades.
But only now are physicians widely seeing the links between health and environment, according to Aaron Bernstein, MD, MPH, interim director of the Center for Climate, Health, and the Global Environment (C-CHANGE) at Harvard School of Public Health, Boston.
C-CHANGE, founded in 1996, was the first center in the world to focus on the health effects of environmental change.
“It’s taken 20 years, but what we’re seeing, I think, is the fruits of education,” Dr. Bernstein said. “There’s clearly a wave building here, and I think it really started with education and people younger than the people in charge calling them into account.”
Like the Philadelphia center, Harvard’s program conducts research on climate and health and educates people from high schoolers to health care veterans. Dr. Bernstein helps lead Climate MD, a program that aims to prepare health care workers for climate crises. The Climate MD team has published several articles in peer-reviewed journals on how to better treat patients struggling with environmental health problems. For example, an article on mapping patients in hurricane zones helped shed light on how systems can identify climate-vulnerable patients using public data.
They also developed a tool to help pediatricians provide “climate-informed primary care” – guidance on how to assess whether children are at risk of any harmful environmental exposures, a feature that is not part of standard pediatric visits.
Like the other programs, Climate MD uses community outreach to treat as many local patients as possible. Staff work with providers at more than 100 health clinics, particularly in areas where climate change disproportionately affects residents.
The next major step is to bring some of this into clinical practice, Dr. Bernstein said. In February 2020, C-CHANGE held its first symposium to address that issue.
“The key is to understand climate issues from a provider’s perspective,” he said. “Then those issues can really be brought to the bedside.”
A version of this article first appeared on Medscape.com.
Are ants the future of cancer detection?
Cancer diagnosis is frightening, invasive, time-consuming, and expensive. And more than 1.6 million people get that cancer diagnosis every year in the United States. That’s a lot of biopsies and a lot of looking at cells under highly sensitive microscopes.
But imagine if detecting cancer in those samples was as simple as taking a whiff.
We know some animals – like dogs and mice – have very sensitive noses that can sniff out disease. Inspired by those studies, French scientists decided to explore whether ants – known for their olfactory prowess – could do the same.
“Using olfaction to detect diseases is not a novel idea,” says Baptiste Piqueret, PhD, a researcher at Sorbonne Paris Nord University and lead author of the study. “Knowing how well ants can learn and how they use olfaction, we tested the abilities of ants to learn and detect diseases.”
While this is still far away from real-life clinical use, it could one day lead to a cheaper, more accessible (if not a little weird) alternative for detecting cancer. What would this new diagnostic method look like?
Pavlov’s ant
Cancer cell metabolism produces volatile organic compounds (VOCs) – organic chemicals that smell and can serve as biomarkers for diagnosis.
To train the ants to target VOCs, the researchers placed breast cancer cells and healthy cells in a petri dish – but the cancer cells included a sugary treat. “We associated a reward to the smell of cancer,” Dr. Piqueret says.
It’s a technique scientists call classical, or Pavlovian, conditioning. A neutral stimulus (cancer smell) is associated with a second stimulus (food) that elicits a behavior. After doing this a few times, the ant learns that the first stimulus predicts the second, and it will seek out the odor hoping to find that food.
Once the training was complete, the researchers presented the ant with the learned odor and a novel one – this time without a reward. Sure enough, the ants spent more time investigating the learned odor than the novel one.
“If you are hungry and you smell the odor of fresh bread, you will enter the closest bakery,” says Dr. Piqueret. “This is the same mechanism the ants are using, as you learned that fresh bread odor equals food.”
Dogs can detect VOCs via the same technique but take months and hundreds of trials to condition, the researchers note. F. fusca ants learn fast, requiring only three training trials.
Why ants?
Ants communicate primarily through olfaction or scent, and this sophisticated “language” makes them very sensitive to odors.
“Since ants are already well-attuned to detecting different chemicals, this makes them ideal for scent recognition,” says Corrie Moreau, PhD, an evolutionary biologist and entomologist at Cornell University, Ithaca, N.Y.
In their tiny ant worlds, the little creatures use chemicals, called pheromones, to convey information to other members of their nest.
“There are alarm pheromones to signal an intruder, trail pheromones so an ant knows which way to walk to a food source, and colony-level odors that signal another ant is a member of the same colony,” Dr. Moreau says.
But on closer inspection, you won’t see a nose on an ant. They “smell” with their antennas.
“These specialized structures are covered with highly sensitive receptors to be able to discern even small chemical differences,” Dr. Moreau says.
There are over 14,000 species of ants and as far as scientists like Dr. Moreau know, all of them use chemical communication, though some are better than others at detecting compounds, such as those scientists are interested in using to detect disease.
Diagnostic ants: Realistic or a curiosity?
Whether or not the new research findings could lead to a real tool for diagnosing cancer is difficult to say, says Dr. Moreau. The study only focused on pure cancer cells in a lab and not those growing inside a human body.
Anna Wanda Komorowski, MD, a medical oncologist-hematologist at Northwell Health in New York, found the study interesting and was impressed with how the researchers trained the ants. But she notes more research would be needed to parse out things like how long the ants would remember their training, and how long they could be kept in a lab for testing.
One of the attractive aspects of the research is that if it worked it might be a cheaper alternative to normal lab practices for detecting cancer cells, and possibly useful in some low-income settings where labs do not have access to cell stain technologies used to detect cancer cells.
Another glitch with the study, notes Dr. Komorowski: “The cells we’d expose them to probably would not be the same cells as those used in the study. They exposed the ants to live cell cultures. Usually, we collect material from biopsy and drop it into formaldehyde, which has such a strong odor. So the lab protocol for cancer detection would have to be different. It could be kind of tricky.”
And while ants are cheaper than stains and dyes and formaldehyde, you’d have to hire someone to train the ants – there’d still be a human factor and related costs.
“It would take much more research to figure out cost, and how applicable and reproducible it would be,” Dr. Komorowski says.
And then there’s the question of whether the ants would do their cancer-detecting work in the lab only, or if direct patient interaction might lead to a diagnosis more swiftly.
Ant expert Dr. Moreau adds, “The human body emits many other odors, so the question is whether the ants would be able to ignore all the other scents and focus only on the target scent.”
“But these results are promising,” she continues. “I guess the question is whether a patient would be willing to have trained ants crawl all over their body looking for potential cancer cells.”
A version of this article first appeared on WebMD.com.
Cancer diagnosis is frightening, invasive, time-consuming, and expensive. And more than 1.6 million people get that cancer diagnosis every year in the United States. That’s a lot of biopsies and a lot of looking at cells under highly sensitive microscopes.
But imagine if detecting cancer in those samples was as simple as taking a whiff.
We know some animals – like dogs and mice – have very sensitive noses that can sniff out disease. Inspired by those studies, French scientists decided to explore whether ants – known for their olfactory prowess – could do the same.
“Using olfaction to detect diseases is not a novel idea,” says Baptiste Piqueret, PhD, a researcher at Sorbonne Paris Nord University and lead author of the study. “Knowing how well ants can learn and how they use olfaction, we tested the abilities of ants to learn and detect diseases.”
While this is still far away from real-life clinical use, it could one day lead to a cheaper, more accessible (if not a little weird) alternative for detecting cancer. What would this new diagnostic method look like?
Pavlov’s ant
Cancer cell metabolism produces volatile organic compounds (VOCs) – organic chemicals that smell and can serve as biomarkers for diagnosis.
To train the ants to target VOCs, the researchers placed breast cancer cells and healthy cells in a petri dish – but the cancer cells included a sugary treat. “We associated a reward to the smell of cancer,” Dr. Piqueret says.
It’s a technique scientists call classical, or Pavlovian, conditioning. A neutral stimulus (cancer smell) is associated with a second stimulus (food) that elicits a behavior. After doing this a few times, the ant learns that the first stimulus predicts the second, and it will seek out the odor hoping to find that food.
Once the training was complete, the researchers presented the ant with the learned odor and a novel one – this time without a reward. Sure enough, the ants spent more time investigating the learned odor than the novel one.
“If you are hungry and you smell the odor of fresh bread, you will enter the closest bakery,” says Dr. Piqueret. “This is the same mechanism the ants are using, as you learned that fresh bread odor equals food.”
Dogs can detect VOCs via the same technique but take months and hundreds of trials to condition, the researchers note. F. fusca ants learn fast, requiring only three training trials.
Why ants?
Ants communicate primarily through olfaction or scent, and this sophisticated “language” makes them very sensitive to odors.
“Since ants are already well-attuned to detecting different chemicals, this makes them ideal for scent recognition,” says Corrie Moreau, PhD, an evolutionary biologist and entomologist at Cornell University, Ithaca, N.Y.
In their tiny ant worlds, the little creatures use chemicals, called pheromones, to convey information to other members of their nest.
“There are alarm pheromones to signal an intruder, trail pheromones so an ant knows which way to walk to a food source, and colony-level odors that signal another ant is a member of the same colony,” Dr. Moreau says.
But on closer inspection, you won’t see a nose on an ant. They “smell” with their antennas.
“These specialized structures are covered with highly sensitive receptors to be able to discern even small chemical differences,” Dr. Moreau says.
There are over 14,000 species of ants and as far as scientists like Dr. Moreau know, all of them use chemical communication, though some are better than others at detecting compounds, such as those scientists are interested in using to detect disease.
Diagnostic ants: Realistic or a curiosity?
Whether or not the new research findings could lead to a real tool for diagnosing cancer is difficult to say, says Dr. Moreau. The study only focused on pure cancer cells in a lab and not those growing inside a human body.
Anna Wanda Komorowski, MD, a medical oncologist-hematologist at Northwell Health in New York, found the study interesting and was impressed with how the researchers trained the ants. But she notes more research would be needed to parse out things like how long the ants would remember their training, and how long they could be kept in a lab for testing.
One of the attractive aspects of the research is that if it worked it might be a cheaper alternative to normal lab practices for detecting cancer cells, and possibly useful in some low-income settings where labs do not have access to cell stain technologies used to detect cancer cells.
Another glitch with the study, notes Dr. Komorowski: “The cells we’d expose them to probably would not be the same cells as those used in the study. They exposed the ants to live cell cultures. Usually, we collect material from biopsy and drop it into formaldehyde, which has such a strong odor. So the lab protocol for cancer detection would have to be different. It could be kind of tricky.”
And while ants are cheaper than stains and dyes and formaldehyde, you’d have to hire someone to train the ants – there’d still be a human factor and related costs.
“It would take much more research to figure out cost, and how applicable and reproducible it would be,” Dr. Komorowski says.
And then there’s the question of whether the ants would do their cancer-detecting work in the lab only, or if direct patient interaction might lead to a diagnosis more swiftly.
Ant expert Dr. Moreau adds, “The human body emits many other odors, so the question is whether the ants would be able to ignore all the other scents and focus only on the target scent.”
“But these results are promising,” she continues. “I guess the question is whether a patient would be willing to have trained ants crawl all over their body looking for potential cancer cells.”
A version of this article first appeared on WebMD.com.
Cancer diagnosis is frightening, invasive, time-consuming, and expensive. And more than 1.6 million people get that cancer diagnosis every year in the United States. That’s a lot of biopsies and a lot of looking at cells under highly sensitive microscopes.
But imagine if detecting cancer in those samples was as simple as taking a whiff.
We know some animals – like dogs and mice – have very sensitive noses that can sniff out disease. Inspired by those studies, French scientists decided to explore whether ants – known for their olfactory prowess – could do the same.
“Using olfaction to detect diseases is not a novel idea,” says Baptiste Piqueret, PhD, a researcher at Sorbonne Paris Nord University and lead author of the study. “Knowing how well ants can learn and how they use olfaction, we tested the abilities of ants to learn and detect diseases.”
While this is still far away from real-life clinical use, it could one day lead to a cheaper, more accessible (if not a little weird) alternative for detecting cancer. What would this new diagnostic method look like?
Pavlov’s ant
Cancer cell metabolism produces volatile organic compounds (VOCs) – organic chemicals that smell and can serve as biomarkers for diagnosis.
To train the ants to target VOCs, the researchers placed breast cancer cells and healthy cells in a petri dish – but the cancer cells included a sugary treat. “We associated a reward to the smell of cancer,” Dr. Piqueret says.
It’s a technique scientists call classical, or Pavlovian, conditioning. A neutral stimulus (cancer smell) is associated with a second stimulus (food) that elicits a behavior. After doing this a few times, the ant learns that the first stimulus predicts the second, and it will seek out the odor hoping to find that food.
Once the training was complete, the researchers presented the ant with the learned odor and a novel one – this time without a reward. Sure enough, the ants spent more time investigating the learned odor than the novel one.
“If you are hungry and you smell the odor of fresh bread, you will enter the closest bakery,” says Dr. Piqueret. “This is the same mechanism the ants are using, as you learned that fresh bread odor equals food.”
Dogs can detect VOCs via the same technique but take months and hundreds of trials to condition, the researchers note. F. fusca ants learn fast, requiring only three training trials.
Why ants?
Ants communicate primarily through olfaction or scent, and this sophisticated “language” makes them very sensitive to odors.
“Since ants are already well-attuned to detecting different chemicals, this makes them ideal for scent recognition,” says Corrie Moreau, PhD, an evolutionary biologist and entomologist at Cornell University, Ithaca, N.Y.
In their tiny ant worlds, the little creatures use chemicals, called pheromones, to convey information to other members of their nest.
“There are alarm pheromones to signal an intruder, trail pheromones so an ant knows which way to walk to a food source, and colony-level odors that signal another ant is a member of the same colony,” Dr. Moreau says.
But on closer inspection, you won’t see a nose on an ant. They “smell” with their antennas.
“These specialized structures are covered with highly sensitive receptors to be able to discern even small chemical differences,” Dr. Moreau says.
There are over 14,000 species of ants and as far as scientists like Dr. Moreau know, all of them use chemical communication, though some are better than others at detecting compounds, such as those scientists are interested in using to detect disease.
Diagnostic ants: Realistic or a curiosity?
Whether or not the new research findings could lead to a real tool for diagnosing cancer is difficult to say, says Dr. Moreau. The study only focused on pure cancer cells in a lab and not those growing inside a human body.
Anna Wanda Komorowski, MD, a medical oncologist-hematologist at Northwell Health in New York, found the study interesting and was impressed with how the researchers trained the ants. But she notes more research would be needed to parse out things like how long the ants would remember their training, and how long they could be kept in a lab for testing.
One of the attractive aspects of the research is that if it worked it might be a cheaper alternative to normal lab practices for detecting cancer cells, and possibly useful in some low-income settings where labs do not have access to cell stain technologies used to detect cancer cells.
Another glitch with the study, notes Dr. Komorowski: “The cells we’d expose them to probably would not be the same cells as those used in the study. They exposed the ants to live cell cultures. Usually, we collect material from biopsy and drop it into formaldehyde, which has such a strong odor. So the lab protocol for cancer detection would have to be different. It could be kind of tricky.”
And while ants are cheaper than stains and dyes and formaldehyde, you’d have to hire someone to train the ants – there’d still be a human factor and related costs.
“It would take much more research to figure out cost, and how applicable and reproducible it would be,” Dr. Komorowski says.
And then there’s the question of whether the ants would do their cancer-detecting work in the lab only, or if direct patient interaction might lead to a diagnosis more swiftly.
Ant expert Dr. Moreau adds, “The human body emits many other odors, so the question is whether the ants would be able to ignore all the other scents and focus only on the target scent.”
“But these results are promising,” she continues. “I guess the question is whether a patient would be willing to have trained ants crawl all over their body looking for potential cancer cells.”
A version of this article first appeared on WebMD.com.
The importance of toxin testing in C. difficile infection: Understanding the results
Clostridioides difficile infection is often confirmed through toxin testing, yet toxin tests alone may not be sufficient for diagnosing and guiding treatment decisions for patients with CDI.
“The presence of a toxigenic strain does not always equal disease,” said David Lyerly, PhD, during a session on C. difficile toxin testing at the Peggy Lillis Foundation 2022 National C. diff Advocacy Summit.
Dr. Lyerly, the chief science officer at Techlab, explained that exotoxins A and B are produced by specific strains of C. difficile and are involved in infections, but some patients who test positive for these toxins by polymerase chain reaction or other tests do not have CDI or they are not appropriate candidates for CDI treatment.
Several studies conducted during the past decade, however, support the importance of toxin detection. Some research has suggested that toxin-positive patients tend to have more clinically severe disease than those who test negative, he noted.
Although its use is limited when it is used alone, toxin testing is needed to confirm a CDI diagnosis and to ensure antibiotic stewardship, Dr. Lyerly said.
He suggested that, in addition to toxin testing, there is a need for molecular measures and other improved diagnostics to identify candidates most likely to benefit from CDI treatment.
“Because we generally detect toxin genes instead of toxin proteins, you can identify persons colonized with toxigenic C. difficile who do not actually have CDI,” Kevin W. Garey, PharmD, from the University of Houston, said in an interview.
Dr. Garey added that a person could likewise have low levels of toxins that aren’t detected by toxin tests but could still have CDI.
“Given this, better diagnostics that incorporate active toxin production and your body’s response to those toxins are needed,” he said, especially since C. difficile toxins are responsible for disease sequelae, including gastroenteritis, colonic perforation, sepsis, and death.
Toxin testing a ‘controversial area’
“C. difficile toxin testing has been a controversial area for almost a decade or more,” Shruti K. Gohil, MD, from University of California, Irvine, Health Epidemiology and Infection Prevention, said in an interview.
Dr. Gohil noted that toxin testing is a better test for clinical C. difficile colitis but by itself can miss C. difficile. “So, we are in this conundrum nationally,” she said.
“Many facilities will use a double- or triple-test strategy to make sure that you have a true C. difficile case mandating the use of antibiotics,” she explained. “The reason we test specifically with the enzyme immunoassay or toxin test is to know whether or not you have real C. difficile that’s actively producing the toxin for colitis.”
A patient with C. difficile who has been treated and is in recovery may still test positive on a C. difficile toxin test, added Dr. Gohil. “It would be great if we had a test that could really judge an active, clinical C. difficile infection. This [test] would help in identifying the right patients who need treatment and would also be able to tell if a patient has been cleared of C. difficile.”
Dr. Lyerly is an employee of Techlab. Dr. Garey and Dr. Gohil reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clostridioides difficile infection is often confirmed through toxin testing, yet toxin tests alone may not be sufficient for diagnosing and guiding treatment decisions for patients with CDI.
“The presence of a toxigenic strain does not always equal disease,” said David Lyerly, PhD, during a session on C. difficile toxin testing at the Peggy Lillis Foundation 2022 National C. diff Advocacy Summit.
Dr. Lyerly, the chief science officer at Techlab, explained that exotoxins A and B are produced by specific strains of C. difficile and are involved in infections, but some patients who test positive for these toxins by polymerase chain reaction or other tests do not have CDI or they are not appropriate candidates for CDI treatment.
Several studies conducted during the past decade, however, support the importance of toxin detection. Some research has suggested that toxin-positive patients tend to have more clinically severe disease than those who test negative, he noted.
Although its use is limited when it is used alone, toxin testing is needed to confirm a CDI diagnosis and to ensure antibiotic stewardship, Dr. Lyerly said.
He suggested that, in addition to toxin testing, there is a need for molecular measures and other improved diagnostics to identify candidates most likely to benefit from CDI treatment.
“Because we generally detect toxin genes instead of toxin proteins, you can identify persons colonized with toxigenic C. difficile who do not actually have CDI,” Kevin W. Garey, PharmD, from the University of Houston, said in an interview.
Dr. Garey added that a person could likewise have low levels of toxins that aren’t detected by toxin tests but could still have CDI.
“Given this, better diagnostics that incorporate active toxin production and your body’s response to those toxins are needed,” he said, especially since C. difficile toxins are responsible for disease sequelae, including gastroenteritis, colonic perforation, sepsis, and death.
Toxin testing a ‘controversial area’
“C. difficile toxin testing has been a controversial area for almost a decade or more,” Shruti K. Gohil, MD, from University of California, Irvine, Health Epidemiology and Infection Prevention, said in an interview.
Dr. Gohil noted that toxin testing is a better test for clinical C. difficile colitis but by itself can miss C. difficile. “So, we are in this conundrum nationally,” she said.
“Many facilities will use a double- or triple-test strategy to make sure that you have a true C. difficile case mandating the use of antibiotics,” she explained. “The reason we test specifically with the enzyme immunoassay or toxin test is to know whether or not you have real C. difficile that’s actively producing the toxin for colitis.”
A patient with C. difficile who has been treated and is in recovery may still test positive on a C. difficile toxin test, added Dr. Gohil. “It would be great if we had a test that could really judge an active, clinical C. difficile infection. This [test] would help in identifying the right patients who need treatment and would also be able to tell if a patient has been cleared of C. difficile.”
Dr. Lyerly is an employee of Techlab. Dr. Garey and Dr. Gohil reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clostridioides difficile infection is often confirmed through toxin testing, yet toxin tests alone may not be sufficient for diagnosing and guiding treatment decisions for patients with CDI.
“The presence of a toxigenic strain does not always equal disease,” said David Lyerly, PhD, during a session on C. difficile toxin testing at the Peggy Lillis Foundation 2022 National C. diff Advocacy Summit.
Dr. Lyerly, the chief science officer at Techlab, explained that exotoxins A and B are produced by specific strains of C. difficile and are involved in infections, but some patients who test positive for these toxins by polymerase chain reaction or other tests do not have CDI or they are not appropriate candidates for CDI treatment.
Several studies conducted during the past decade, however, support the importance of toxin detection. Some research has suggested that toxin-positive patients tend to have more clinically severe disease than those who test negative, he noted.
Although its use is limited when it is used alone, toxin testing is needed to confirm a CDI diagnosis and to ensure antibiotic stewardship, Dr. Lyerly said.
He suggested that, in addition to toxin testing, there is a need for molecular measures and other improved diagnostics to identify candidates most likely to benefit from CDI treatment.
“Because we generally detect toxin genes instead of toxin proteins, you can identify persons colonized with toxigenic C. difficile who do not actually have CDI,” Kevin W. Garey, PharmD, from the University of Houston, said in an interview.
Dr. Garey added that a person could likewise have low levels of toxins that aren’t detected by toxin tests but could still have CDI.
“Given this, better diagnostics that incorporate active toxin production and your body’s response to those toxins are needed,” he said, especially since C. difficile toxins are responsible for disease sequelae, including gastroenteritis, colonic perforation, sepsis, and death.
Toxin testing a ‘controversial area’
“C. difficile toxin testing has been a controversial area for almost a decade or more,” Shruti K. Gohil, MD, from University of California, Irvine, Health Epidemiology and Infection Prevention, said in an interview.
Dr. Gohil noted that toxin testing is a better test for clinical C. difficile colitis but by itself can miss C. difficile. “So, we are in this conundrum nationally,” she said.
“Many facilities will use a double- or triple-test strategy to make sure that you have a true C. difficile case mandating the use of antibiotics,” she explained. “The reason we test specifically with the enzyme immunoassay or toxin test is to know whether or not you have real C. difficile that’s actively producing the toxin for colitis.”
A patient with C. difficile who has been treated and is in recovery may still test positive on a C. difficile toxin test, added Dr. Gohil. “It would be great if we had a test that could really judge an active, clinical C. difficile infection. This [test] would help in identifying the right patients who need treatment and would also be able to tell if a patient has been cleared of C. difficile.”
Dr. Lyerly is an employee of Techlab. Dr. Garey and Dr. Gohil reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Medical ‘myths’ persist despite evidence, says professor of medicine
These long-held pieces of dogma – or “medical myths” – were engraved during training or early in the careers of many physicians, and are difficult to overcome, noted Douglas Paauw, MD, professor of medicine at the University of Washington, Seattle.
“I think that myths persist because medical professionals get taught one way in training, given a ‘truth’ or ‘This is the way we do it,’ and then do not ever rethink, ‘Is it true?’ ” he said in an interview. “Studies pop up to question conventional wisdom, but unless the studies get highly publicized, they aren’t noticed.”
During his presentation, Dr. Paauw discussed three of what he considers to be some of the some of the medical myths that are in greatest need of being dispelled.
Shellfish allergy and radiocontrast
A myth persists that people with a shellfish allergy could have an allergic reaction when a contrast agent is used for a scan, he said.
This belief arose, because fish and shellfish contain iodine, and allergic reactions to seafood are fairly common, and contrast agents contain iodine, too, Dr. Paauw said.
The belief is widespread, with 65% of radiologists and 88.9% of interventional cardiologists saying they ask about seafood or shellfish allergies before administering contrast. And a third of radiologists and 50% of cardiologists said they would withhold contrast media or recommend a premedication for patients with such an allergy.
But the belief makes no sense, Dr. Pauuw said. Iodine is present in many other foods, including milk and bread, and allergies to shellfish are because of parvalbumin protein and tropomyosins, not iodine.
Colonoscopy dogma
It’s been long believed that people need to be on a clear, liquid diet for 1 or 2 days and need to drink a bowel-prep liquid before a colonoscopy, noted Dr. Paauw.
But the evidence shows this isn’t necessary, he said.
A 2020 study found that a low-residual diet, allowing foods such as meat, eggs, dairy, and bread, were comparable to the clear liquid diet in terms of bowel prep and detection of polyps during the exam. The patients on the low-residual diet had less nausea, less vomiting, and less hunger, and expressed more willingness to have a repeat colonoscopy.
“Let them eat,” Dr. Paauw said in his presentation.
Metronidazole and alcohol
There is a belief that patients shouldn’t drink alcohol if they are taking metronidazole, because of concerns about nausea, vomiting, flushing and other symptoms – also known as a disulfiramlike reaction, Dr. Paauw explained.
Case reports have been published, but the cases were presented as though a metronidazole-ethanol reaction was an established fact, and the authors didn’t provide evidence to justify this, Dr. Paauw said.
But it’s been shown in rat models that metronidazole can increase levels of acetaldehyde, the trigger of symptoms, in the colon, but not in the blood. And in a small placebo-controlled, randomized trial, six people were given metronidazole and ethanol and, after regular blood testing, no difference was seen in acetaldehyde blood levels, vital signs, or symptoms.
The Centers for Disease Control and Prevention has said that avoiding alcohol while taking metronidazole is unnecessary, said Dr. Paauw.
Sinus headaches
Contrary to common belief, headaches thought to be “sinus headaches” are usually migraine headaches, Dr. Paauw said.
In one study, 2,991 patients with six headaches in the previous 6 months were self-diagnosed or were physician-diagnosed with sinus headaches. But 88% of these headaches met the International Headache Society criteria for migraine headache.
Dr. Paauw said he hopes that clinicians reconsider the evidence regularly when deciding how to treat their patients, and not rely on bits of dogma.
“They stay with us,” he said, “and sometimes there are other ways to do it.”
Shien Tze, MD, an internist in Fargo, N,D,, said that patients sometimes also hold misconceptions, based on outdated dogma, that he needs to dispel.
“I try to convince them that this is a myth that is not based on evidence, not based on science,” he said. “I think it depends on the way you say it. If you say it in a calm, firm, not wishy-washy way, the patients believe you.”
Dr. Paauw reported no relevant financial disclosures. He serves on the editorial advisory board of Internal Medicine News, and he contributes “Myth of the Month” and “Pearl of the Month” columns to this publication.
These long-held pieces of dogma – or “medical myths” – were engraved during training or early in the careers of many physicians, and are difficult to overcome, noted Douglas Paauw, MD, professor of medicine at the University of Washington, Seattle.
“I think that myths persist because medical professionals get taught one way in training, given a ‘truth’ or ‘This is the way we do it,’ and then do not ever rethink, ‘Is it true?’ ” he said in an interview. “Studies pop up to question conventional wisdom, but unless the studies get highly publicized, they aren’t noticed.”
During his presentation, Dr. Paauw discussed three of what he considers to be some of the some of the medical myths that are in greatest need of being dispelled.
Shellfish allergy and radiocontrast
A myth persists that people with a shellfish allergy could have an allergic reaction when a contrast agent is used for a scan, he said.
This belief arose, because fish and shellfish contain iodine, and allergic reactions to seafood are fairly common, and contrast agents contain iodine, too, Dr. Paauw said.
The belief is widespread, with 65% of radiologists and 88.9% of interventional cardiologists saying they ask about seafood or shellfish allergies before administering contrast. And a third of radiologists and 50% of cardiologists said they would withhold contrast media or recommend a premedication for patients with such an allergy.
But the belief makes no sense, Dr. Pauuw said. Iodine is present in many other foods, including milk and bread, and allergies to shellfish are because of parvalbumin protein and tropomyosins, not iodine.
Colonoscopy dogma
It’s been long believed that people need to be on a clear, liquid diet for 1 or 2 days and need to drink a bowel-prep liquid before a colonoscopy, noted Dr. Paauw.
But the evidence shows this isn’t necessary, he said.
A 2020 study found that a low-residual diet, allowing foods such as meat, eggs, dairy, and bread, were comparable to the clear liquid diet in terms of bowel prep and detection of polyps during the exam. The patients on the low-residual diet had less nausea, less vomiting, and less hunger, and expressed more willingness to have a repeat colonoscopy.
“Let them eat,” Dr. Paauw said in his presentation.
Metronidazole and alcohol
There is a belief that patients shouldn’t drink alcohol if they are taking metronidazole, because of concerns about nausea, vomiting, flushing and other symptoms – also known as a disulfiramlike reaction, Dr. Paauw explained.
Case reports have been published, but the cases were presented as though a metronidazole-ethanol reaction was an established fact, and the authors didn’t provide evidence to justify this, Dr. Paauw said.
But it’s been shown in rat models that metronidazole can increase levels of acetaldehyde, the trigger of symptoms, in the colon, but not in the blood. And in a small placebo-controlled, randomized trial, six people were given metronidazole and ethanol and, after regular blood testing, no difference was seen in acetaldehyde blood levels, vital signs, or symptoms.
The Centers for Disease Control and Prevention has said that avoiding alcohol while taking metronidazole is unnecessary, said Dr. Paauw.
Sinus headaches
Contrary to common belief, headaches thought to be “sinus headaches” are usually migraine headaches, Dr. Paauw said.
In one study, 2,991 patients with six headaches in the previous 6 months were self-diagnosed or were physician-diagnosed with sinus headaches. But 88% of these headaches met the International Headache Society criteria for migraine headache.
Dr. Paauw said he hopes that clinicians reconsider the evidence regularly when deciding how to treat their patients, and not rely on bits of dogma.
“They stay with us,” he said, “and sometimes there are other ways to do it.”
Shien Tze, MD, an internist in Fargo, N,D,, said that patients sometimes also hold misconceptions, based on outdated dogma, that he needs to dispel.
“I try to convince them that this is a myth that is not based on evidence, not based on science,” he said. “I think it depends on the way you say it. If you say it in a calm, firm, not wishy-washy way, the patients believe you.”
Dr. Paauw reported no relevant financial disclosures. He serves on the editorial advisory board of Internal Medicine News, and he contributes “Myth of the Month” and “Pearl of the Month” columns to this publication.
These long-held pieces of dogma – or “medical myths” – were engraved during training or early in the careers of many physicians, and are difficult to overcome, noted Douglas Paauw, MD, professor of medicine at the University of Washington, Seattle.
“I think that myths persist because medical professionals get taught one way in training, given a ‘truth’ or ‘This is the way we do it,’ and then do not ever rethink, ‘Is it true?’ ” he said in an interview. “Studies pop up to question conventional wisdom, but unless the studies get highly publicized, they aren’t noticed.”
During his presentation, Dr. Paauw discussed three of what he considers to be some of the some of the medical myths that are in greatest need of being dispelled.
Shellfish allergy and radiocontrast
A myth persists that people with a shellfish allergy could have an allergic reaction when a contrast agent is used for a scan, he said.
This belief arose, because fish and shellfish contain iodine, and allergic reactions to seafood are fairly common, and contrast agents contain iodine, too, Dr. Paauw said.
The belief is widespread, with 65% of radiologists and 88.9% of interventional cardiologists saying they ask about seafood or shellfish allergies before administering contrast. And a third of radiologists and 50% of cardiologists said they would withhold contrast media or recommend a premedication for patients with such an allergy.
But the belief makes no sense, Dr. Pauuw said. Iodine is present in many other foods, including milk and bread, and allergies to shellfish are because of parvalbumin protein and tropomyosins, not iodine.
Colonoscopy dogma
It’s been long believed that people need to be on a clear, liquid diet for 1 or 2 days and need to drink a bowel-prep liquid before a colonoscopy, noted Dr. Paauw.
But the evidence shows this isn’t necessary, he said.
A 2020 study found that a low-residual diet, allowing foods such as meat, eggs, dairy, and bread, were comparable to the clear liquid diet in terms of bowel prep and detection of polyps during the exam. The patients on the low-residual diet had less nausea, less vomiting, and less hunger, and expressed more willingness to have a repeat colonoscopy.
“Let them eat,” Dr. Paauw said in his presentation.
Metronidazole and alcohol
There is a belief that patients shouldn’t drink alcohol if they are taking metronidazole, because of concerns about nausea, vomiting, flushing and other symptoms – also known as a disulfiramlike reaction, Dr. Paauw explained.
Case reports have been published, but the cases were presented as though a metronidazole-ethanol reaction was an established fact, and the authors didn’t provide evidence to justify this, Dr. Paauw said.
But it’s been shown in rat models that metronidazole can increase levels of acetaldehyde, the trigger of symptoms, in the colon, but not in the blood. And in a small placebo-controlled, randomized trial, six people were given metronidazole and ethanol and, after regular blood testing, no difference was seen in acetaldehyde blood levels, vital signs, or symptoms.
The Centers for Disease Control and Prevention has said that avoiding alcohol while taking metronidazole is unnecessary, said Dr. Paauw.
Sinus headaches
Contrary to common belief, headaches thought to be “sinus headaches” are usually migraine headaches, Dr. Paauw said.
In one study, 2,991 patients with six headaches in the previous 6 months were self-diagnosed or were physician-diagnosed with sinus headaches. But 88% of these headaches met the International Headache Society criteria for migraine headache.
Dr. Paauw said he hopes that clinicians reconsider the evidence regularly when deciding how to treat their patients, and not rely on bits of dogma.
“They stay with us,” he said, “and sometimes there are other ways to do it.”
Shien Tze, MD, an internist in Fargo, N,D,, said that patients sometimes also hold misconceptions, based on outdated dogma, that he needs to dispel.
“I try to convince them that this is a myth that is not based on evidence, not based on science,” he said. “I think it depends on the way you say it. If you say it in a calm, firm, not wishy-washy way, the patients believe you.”
Dr. Paauw reported no relevant financial disclosures. He serves on the editorial advisory board of Internal Medicine News, and he contributes “Myth of the Month” and “Pearl of the Month” columns to this publication.
AT INTERNAL MEDICINE 2022