User login
For MD-IQ use only
Common brain parasite linked to attractiveness, new study
That Toxoplasma gondii looks good on you
Parasite and attractiveness don’t usually go together, but it appears that nobody told Toxoplasma gondii. The world’s most successful parasite affects 30%-50% of the world’s population, and it’s mainly thought to go after the brain in humans, possibly changing behavior and leading to neurological disorders and mental illness.
Now, are you ready to be super confused? According to a recent study, those affected with T. gondii were seen as more attractive and healthy looking, compared with noninfected people. It doesn’t make much sense to us, but it could be an evolutionary thing: The more attractive the parasite makes a person or animal, the more likely it is to spread.
“Some sexually transmitted parasites, such as T. gondii, may produce changes in the appearance and behavior of the human host, either as a by-product of the infection or as the result of the manipulation of the parasite to increase its spread to new hosts,” Javier Borráz-León, PhD, of the University of Turku (Finland), and associates wrote in PeerJ.
Previous research has suggested that men with more testosterone are more likely to become infected because of their behavior with the extra hormones. It’s also been noted that the parasite may manipulate hormones for its own gain, but that’s not proven. Infected women were found to have a lower BMI, more confidence in their appearance, and more partners. Dr. Borráz-León and associates also found that “Toxoplasma-infected subjects had significantly lower facial fluctuating asymmetry than the noninfected people,” ScienceAlert said.
We usually perceive parasites as a bad thing, but honestly this one isn’t sounding too bad. It seems to help with some confidence boosters, and who doesn’t want that? We’re thinking that T. gondii could be the Next Big Thing. All it needs is some marketing and … what if it was covered with nonpareils?
Give it to me straight, Doc. Don’t sugar coat it.
Okay, so he’s not a doctor – not a medical doctor, anyway – but that’s exactly what he did. William H. Grover, PhD, has sugar coated drugs in the name of fraud prevention. We will explain.
The sugar coating comes in the form of nonpareils, the tiny and colorful round sprinkles often found covering small discs of chocolate. Dr. Grover, a bioengineering professor at the University of California, Riverside, who has been working on ways to ensure the authenticity of pharmaceuticals, “started wondering how many different patterns of colored nonpareils were possible on these candies,” he said in a statement from the university.
With just eight colors and an average of 92 individual nonpareils on each candy, the combinations, he found out, are almost endless. Could the same thing be done with a pill? Could the nonpareils be applied as a coating to a pill, giving it a unique pattern that could be stored by the manufacturer and used later as identification?
After much time and effort involving edible cake-decorating glue, Tylenol capsules, smartphones, and computer simulations, he produced CandyCode, an algorithm that converts a photo of a nonpareil-covered pill “into a set of text strings suitable for storing in a computer database and querying by consumers,” the statement explained.
Dr. Grover also mentioned a side benefit: “Anecdotally, I found that CandyCoded caplets were more pleasant to swallow than plain caplets, confirming Mary Poppins’ classic observation about the relationship between sugar and medicine.”
First of all, we can’t believe we just used a Mary Poppins reference. Not exactly what you’d call MDedgey, is it? Second of all, what about the children? We’re talking about drugs that, literally, have been turned into candy. Are the kids going to love them, too? Sounds more like a job for Mr. Yuk.
So you want to be a superhero?
Be honest, who didn’t want to be a superhero when they were a kid? There’s a reason every other movie released in the past decade has been a superhero movie. That’s how we’ve ended up with the millionth Batman reboot and Marvel scraping the bottom of the C-list hero barrel. (Seriously, who’d actually heard of Moon Knight before now?)
Point is, we all like to fantasize, and now a meta-analysis from researchers in Germany and the United States has given us all a reason to strike those dashing superhero poses. Through evaluation of 130 studies and over 10,000 people, the researchers found that power posing (and perfect posture) was strongly associated with increased confidence and self-worth. It was also associated with improved behavior, though the connection was less strong.
Sadly though, the research found no connection with power posing and changes in testosterone or cortisol levels. Standing like a superhero may make you feel good, but it won’t give your body any cool powers or superhuman abilities. But don’t despair, because we’re not finished yet. In fact, it may be the biggest news we’ve ever reported for LOTME: A group of scientists from the University of Kentucky has assembled the full genome of a salamander.
Wait, we have more! Beyond having a genome ten times bigger than a human, this salamander, the axolotl from Mexico, is the model of natural regeneration. Name a body part, and the axolotl can grow it back. It can even regenerate portions of its brain. And now that we have access to the complete genome, it’s possible that one day we could use the axolotl’s regeneration for ourselves. Growing back limbs, regenerating spinal cords, the sky’s the limit. And if Wolverine and Deadpool are anything to go by, it’s all you need to get that superhero career off the ground. Salamander powers may not have the cachet of a radioactive spider, but we’ll take what we can get.
Post your way to financial hardship
After you pump your gas at the gas station, how do you pay? At the pump or inside? How frequently do you post to your social media pages? What kind of content are you posting?
That kind of nontraditional credit data hasn’t been considered by lenders and credit agencies, but that is changing. The reasoning? It’s opening more opportunities for those without much credit history. But according to a paper published by Janine S. Hiller of Virginia Tech and Lindsay Sain Jones, a financial regulation researcher at the University of Georgia, this just opens a can of worms.
Why is this so dangerous? Well, alternative credit scoring isn’t covered by the Fair Credit Reporting Act or Equal Opportunity Act, so the consumer doesn’t have the ability to dispute any data the credit agencies or lenders receive. Then there’s the “credit boost,” which some companies offer to gain access to the consumer’s data. Often there are no limitations on how long it’s kept. That purchase you made 2 years ago can come back to haunt you.
It also creates a cause for the possibility of discrimination based on “lifestyle-related data points,” which some lenders use to determine creditworthiness: zip code, age, gender, race, socioeconomic status. Even where the consumer went to college is a factor taken under consideration.
“There are all kinds of factors that can be correlated with creditworthiness, but that doesn’t mean they should be used,” Ms. Jones said in the EurekAlert statement.
Let’s say someone applies for a loan needed for a medical procedure. They could be denied because the lender or a credit-reporting agency didn’t like the data they received (most times without the consumer’s consent). Talk about a broken system.
That Toxoplasma gondii looks good on you
Parasite and attractiveness don’t usually go together, but it appears that nobody told Toxoplasma gondii. The world’s most successful parasite affects 30%-50% of the world’s population, and it’s mainly thought to go after the brain in humans, possibly changing behavior and leading to neurological disorders and mental illness.
Now, are you ready to be super confused? According to a recent study, those affected with T. gondii were seen as more attractive and healthy looking, compared with noninfected people. It doesn’t make much sense to us, but it could be an evolutionary thing: The more attractive the parasite makes a person or animal, the more likely it is to spread.
“Some sexually transmitted parasites, such as T. gondii, may produce changes in the appearance and behavior of the human host, either as a by-product of the infection or as the result of the manipulation of the parasite to increase its spread to new hosts,” Javier Borráz-León, PhD, of the University of Turku (Finland), and associates wrote in PeerJ.
Previous research has suggested that men with more testosterone are more likely to become infected because of their behavior with the extra hormones. It’s also been noted that the parasite may manipulate hormones for its own gain, but that’s not proven. Infected women were found to have a lower BMI, more confidence in their appearance, and more partners. Dr. Borráz-León and associates also found that “Toxoplasma-infected subjects had significantly lower facial fluctuating asymmetry than the noninfected people,” ScienceAlert said.
We usually perceive parasites as a bad thing, but honestly this one isn’t sounding too bad. It seems to help with some confidence boosters, and who doesn’t want that? We’re thinking that T. gondii could be the Next Big Thing. All it needs is some marketing and … what if it was covered with nonpareils?
Give it to me straight, Doc. Don’t sugar coat it.
Okay, so he’s not a doctor – not a medical doctor, anyway – but that’s exactly what he did. William H. Grover, PhD, has sugar coated drugs in the name of fraud prevention. We will explain.
The sugar coating comes in the form of nonpareils, the tiny and colorful round sprinkles often found covering small discs of chocolate. Dr. Grover, a bioengineering professor at the University of California, Riverside, who has been working on ways to ensure the authenticity of pharmaceuticals, “started wondering how many different patterns of colored nonpareils were possible on these candies,” he said in a statement from the university.
With just eight colors and an average of 92 individual nonpareils on each candy, the combinations, he found out, are almost endless. Could the same thing be done with a pill? Could the nonpareils be applied as a coating to a pill, giving it a unique pattern that could be stored by the manufacturer and used later as identification?
After much time and effort involving edible cake-decorating glue, Tylenol capsules, smartphones, and computer simulations, he produced CandyCode, an algorithm that converts a photo of a nonpareil-covered pill “into a set of text strings suitable for storing in a computer database and querying by consumers,” the statement explained.
Dr. Grover also mentioned a side benefit: “Anecdotally, I found that CandyCoded caplets were more pleasant to swallow than plain caplets, confirming Mary Poppins’ classic observation about the relationship between sugar and medicine.”
First of all, we can’t believe we just used a Mary Poppins reference. Not exactly what you’d call MDedgey, is it? Second of all, what about the children? We’re talking about drugs that, literally, have been turned into candy. Are the kids going to love them, too? Sounds more like a job for Mr. Yuk.
So you want to be a superhero?
Be honest, who didn’t want to be a superhero when they were a kid? There’s a reason every other movie released in the past decade has been a superhero movie. That’s how we’ve ended up with the millionth Batman reboot and Marvel scraping the bottom of the C-list hero barrel. (Seriously, who’d actually heard of Moon Knight before now?)
Point is, we all like to fantasize, and now a meta-analysis from researchers in Germany and the United States has given us all a reason to strike those dashing superhero poses. Through evaluation of 130 studies and over 10,000 people, the researchers found that power posing (and perfect posture) was strongly associated with increased confidence and self-worth. It was also associated with improved behavior, though the connection was less strong.
Sadly though, the research found no connection with power posing and changes in testosterone or cortisol levels. Standing like a superhero may make you feel good, but it won’t give your body any cool powers or superhuman abilities. But don’t despair, because we’re not finished yet. In fact, it may be the biggest news we’ve ever reported for LOTME: A group of scientists from the University of Kentucky has assembled the full genome of a salamander.
Wait, we have more! Beyond having a genome ten times bigger than a human, this salamander, the axolotl from Mexico, is the model of natural regeneration. Name a body part, and the axolotl can grow it back. It can even regenerate portions of its brain. And now that we have access to the complete genome, it’s possible that one day we could use the axolotl’s regeneration for ourselves. Growing back limbs, regenerating spinal cords, the sky’s the limit. And if Wolverine and Deadpool are anything to go by, it’s all you need to get that superhero career off the ground. Salamander powers may not have the cachet of a radioactive spider, but we’ll take what we can get.
Post your way to financial hardship
After you pump your gas at the gas station, how do you pay? At the pump or inside? How frequently do you post to your social media pages? What kind of content are you posting?
That kind of nontraditional credit data hasn’t been considered by lenders and credit agencies, but that is changing. The reasoning? It’s opening more opportunities for those without much credit history. But according to a paper published by Janine S. Hiller of Virginia Tech and Lindsay Sain Jones, a financial regulation researcher at the University of Georgia, this just opens a can of worms.
Why is this so dangerous? Well, alternative credit scoring isn’t covered by the Fair Credit Reporting Act or Equal Opportunity Act, so the consumer doesn’t have the ability to dispute any data the credit agencies or lenders receive. Then there’s the “credit boost,” which some companies offer to gain access to the consumer’s data. Often there are no limitations on how long it’s kept. That purchase you made 2 years ago can come back to haunt you.
It also creates a cause for the possibility of discrimination based on “lifestyle-related data points,” which some lenders use to determine creditworthiness: zip code, age, gender, race, socioeconomic status. Even where the consumer went to college is a factor taken under consideration.
“There are all kinds of factors that can be correlated with creditworthiness, but that doesn’t mean they should be used,” Ms. Jones said in the EurekAlert statement.
Let’s say someone applies for a loan needed for a medical procedure. They could be denied because the lender or a credit-reporting agency didn’t like the data they received (most times without the consumer’s consent). Talk about a broken system.
That Toxoplasma gondii looks good on you
Parasite and attractiveness don’t usually go together, but it appears that nobody told Toxoplasma gondii. The world’s most successful parasite affects 30%-50% of the world’s population, and it’s mainly thought to go after the brain in humans, possibly changing behavior and leading to neurological disorders and mental illness.
Now, are you ready to be super confused? According to a recent study, those affected with T. gondii were seen as more attractive and healthy looking, compared with noninfected people. It doesn’t make much sense to us, but it could be an evolutionary thing: The more attractive the parasite makes a person or animal, the more likely it is to spread.
“Some sexually transmitted parasites, such as T. gondii, may produce changes in the appearance and behavior of the human host, either as a by-product of the infection or as the result of the manipulation of the parasite to increase its spread to new hosts,” Javier Borráz-León, PhD, of the University of Turku (Finland), and associates wrote in PeerJ.
Previous research has suggested that men with more testosterone are more likely to become infected because of their behavior with the extra hormones. It’s also been noted that the parasite may manipulate hormones for its own gain, but that’s not proven. Infected women were found to have a lower BMI, more confidence in their appearance, and more partners. Dr. Borráz-León and associates also found that “Toxoplasma-infected subjects had significantly lower facial fluctuating asymmetry than the noninfected people,” ScienceAlert said.
We usually perceive parasites as a bad thing, but honestly this one isn’t sounding too bad. It seems to help with some confidence boosters, and who doesn’t want that? We’re thinking that T. gondii could be the Next Big Thing. All it needs is some marketing and … what if it was covered with nonpareils?
Give it to me straight, Doc. Don’t sugar coat it.
Okay, so he’s not a doctor – not a medical doctor, anyway – but that’s exactly what he did. William H. Grover, PhD, has sugar coated drugs in the name of fraud prevention. We will explain.
The sugar coating comes in the form of nonpareils, the tiny and colorful round sprinkles often found covering small discs of chocolate. Dr. Grover, a bioengineering professor at the University of California, Riverside, who has been working on ways to ensure the authenticity of pharmaceuticals, “started wondering how many different patterns of colored nonpareils were possible on these candies,” he said in a statement from the university.
With just eight colors and an average of 92 individual nonpareils on each candy, the combinations, he found out, are almost endless. Could the same thing be done with a pill? Could the nonpareils be applied as a coating to a pill, giving it a unique pattern that could be stored by the manufacturer and used later as identification?
After much time and effort involving edible cake-decorating glue, Tylenol capsules, smartphones, and computer simulations, he produced CandyCode, an algorithm that converts a photo of a nonpareil-covered pill “into a set of text strings suitable for storing in a computer database and querying by consumers,” the statement explained.
Dr. Grover also mentioned a side benefit: “Anecdotally, I found that CandyCoded caplets were more pleasant to swallow than plain caplets, confirming Mary Poppins’ classic observation about the relationship between sugar and medicine.”
First of all, we can’t believe we just used a Mary Poppins reference. Not exactly what you’d call MDedgey, is it? Second of all, what about the children? We’re talking about drugs that, literally, have been turned into candy. Are the kids going to love them, too? Sounds more like a job for Mr. Yuk.
So you want to be a superhero?
Be honest, who didn’t want to be a superhero when they were a kid? There’s a reason every other movie released in the past decade has been a superhero movie. That’s how we’ve ended up with the millionth Batman reboot and Marvel scraping the bottom of the C-list hero barrel. (Seriously, who’d actually heard of Moon Knight before now?)
Point is, we all like to fantasize, and now a meta-analysis from researchers in Germany and the United States has given us all a reason to strike those dashing superhero poses. Through evaluation of 130 studies and over 10,000 people, the researchers found that power posing (and perfect posture) was strongly associated with increased confidence and self-worth. It was also associated with improved behavior, though the connection was less strong.
Sadly though, the research found no connection with power posing and changes in testosterone or cortisol levels. Standing like a superhero may make you feel good, but it won’t give your body any cool powers or superhuman abilities. But don’t despair, because we’re not finished yet. In fact, it may be the biggest news we’ve ever reported for LOTME: A group of scientists from the University of Kentucky has assembled the full genome of a salamander.
Wait, we have more! Beyond having a genome ten times bigger than a human, this salamander, the axolotl from Mexico, is the model of natural regeneration. Name a body part, and the axolotl can grow it back. It can even regenerate portions of its brain. And now that we have access to the complete genome, it’s possible that one day we could use the axolotl’s regeneration for ourselves. Growing back limbs, regenerating spinal cords, the sky’s the limit. And if Wolverine and Deadpool are anything to go by, it’s all you need to get that superhero career off the ground. Salamander powers may not have the cachet of a radioactive spider, but we’ll take what we can get.
Post your way to financial hardship
After you pump your gas at the gas station, how do you pay? At the pump or inside? How frequently do you post to your social media pages? What kind of content are you posting?
That kind of nontraditional credit data hasn’t been considered by lenders and credit agencies, but that is changing. The reasoning? It’s opening more opportunities for those without much credit history. But according to a paper published by Janine S. Hiller of Virginia Tech and Lindsay Sain Jones, a financial regulation researcher at the University of Georgia, this just opens a can of worms.
Why is this so dangerous? Well, alternative credit scoring isn’t covered by the Fair Credit Reporting Act or Equal Opportunity Act, so the consumer doesn’t have the ability to dispute any data the credit agencies or lenders receive. Then there’s the “credit boost,” which some companies offer to gain access to the consumer’s data. Often there are no limitations on how long it’s kept. That purchase you made 2 years ago can come back to haunt you.
It also creates a cause for the possibility of discrimination based on “lifestyle-related data points,” which some lenders use to determine creditworthiness: zip code, age, gender, race, socioeconomic status. Even where the consumer went to college is a factor taken under consideration.
“There are all kinds of factors that can be correlated with creditworthiness, but that doesn’t mean they should be used,” Ms. Jones said in the EurekAlert statement.
Let’s say someone applies for a loan needed for a medical procedure. They could be denied because the lender or a credit-reporting agency didn’t like the data they received (most times without the consumer’s consent). Talk about a broken system.
Is benzophenone safe in skin care? Part 1: Risks to humans
Benzophenones are a family of compounds that include dixoxybenzone, sulisobenzone, and benzophenone-3, or oxybenzone. These . Benzophenones (BPs) act as penetration enhancers, as they modify the structure of the skin and facilitate the absorption of other chemical ingredients into the body. The best known uses of these compounds are as perfume fixatives and sunscreen agents.
Sunscreens and benzophenones
BP-2, -3 and -4 are used as sunscreens but have many downsides. They are well known photoallergens, are toxic to aquatic animals (especially BP-3), and are found in urine. BP-2 has weak estrogenic effects, and some studies suggest that it decreases fertility in men. BP-4 can increase absorption of pesticides. BP-3 is banned in Hawaii because of the risk to coral and is the most worrisome.
In particular, BP-3 is known to protect skin and hair from UV radiation-induced harm.1 Unfortunately, BPs are also associated with photocontact allergies, hypersensitivity, hives, contact urticaria, anaphylaxis, hormone disruption, and DNA damage.2,3 BP-3 has also been implicated as an environmental contaminant. This column will focus on recent studies pertaining to effects on humans, primarily, and on the role of BPs in sunscreen agents.
Effects of BPs in animals
A recent study on the cytotoxicity of BP-3 against thymocytes in rats revealed that cell mortality increased significantly after 3 hours of exposure to 300 μM BP-3, but the membrane potential of thymocytes was unchanged by BP-3 exposure. In a concentration-dependent fashion, intracellular Zn2+ levels increased significantly after administration of at least 30 μM BP-3. The investigators concluded that the cytotoxicity engendered by BP-3 could be the result of oxidative stress linked to elevated intracellular Zn2+ levels.1
Effects of BPs in humans and systemic absorption
In multiple studies, exposure to BP-3, as well as to octinoxate, has been linked to endocrine and hormonal disruptions in humans and animals.4,5 Motivated by several notable observations (global increase in the use of sunscreens with UV filters; rapid rise in malignant melanoma, against which sunscreens should protect; increase in reported experimental findings of UV filters acting as endocrine disruptors), Krause et al. in 2012 reviewed animal and human data on the UV filters BP-3, 3-benzylidene camphor (3-BC), 3-(4-methyl-benzylidene) camphor (4-MBC), 2-ethylhexyl 4-methoxy cinnamate (OMC), homosalate (HMS), 2-ethylhexyl 4-dimethylaminobenzoate, and 4-aminobenzoic acid (PABA). Importantly, BP-3 was present in 96% of human urine samples in the United States, and various filters were found in 85% of the human breast milk samples in Switzerland.6
A 2019 analysis by Wang and Ganley reported that systemic absorption of the active sunscreen ingredient BP-3 can be substantial, justifying the assessment and understanding of systemic exposure to characterize the risks of long-term usage.7
Between January and February 2019, Matta et al. conducted a randomized clinical trial with 48 healthy participants to evaluate the systemic absorption and pharmacokinetics of six active ingredients in four sunscreen formulations, including avobenzone and BP-3. The researchers found that all ingredients were systemically absorbed, with plasma concentrations exceeding the Food and Drug Administration threshold for considering the waiving of further safety studies. They concluded that these results did not warrant discontinuing the use of the tested sunscreen ingredients.8 Yeager and Lim add that, while BP-3 has been incorporated into sunscreen formulations for sale in the United States since 1978, there have been no reports of adverse systemic reactions in human beings.3
However, topical reactions have elicited a different assessment. That is, in 2014, the American Contact Dermatitis Society labeled BPs the Contact Allergen of the Year, as they were identified as the most common source of photoallergic and contact allergic reactions of all UV filters.3,9
Risks of BPs in sunscreens and other skincare products
In 2015, Amar et al. investigated the photogenotoxicity and apoptotic effects in human keratinocytes (HaCaT cells) of BP-1, which is used as a UV blocker in sunscreens. They found that BP-1, when exposed to UV radiation, photosensitized cells and yielded intracellular reactive oxygen species. Significant reductions in cell viability were also seen with exposure to sunlight, UVA, and UVB. The researchers also confirmed genotoxic activity, with BP-1 augmenting lipid peroxidation and upregulating apoptotic proteins. They concluded that sunscreen users should be advised to avoid products that contain BP-1.10
In 2019, Amar et al. evaluated the effects of BPs on the differential expression of proteins in HaCaT cells exposed to UVA. Their findings indicated the expression of novel proteins that helped to initiate or promote apoptosis. They concluded that, because of the predilection to render such effects in human skin keratinocytes, consumers should avoid the use of sunscreens that contain BPs as UV blocking ingredients.11
Still widely used as an effective filter against UVA2 and UVB, BP-3 was believed to be present in two thirds of nonmineral sunscreens in the United States in 2018.3,12
Notably, BP-1 and BP-3 were found in small proportions (3.7% and 4.9%, respectively) among a total of 283 products culled from various stores in Lecce, Italy, in a survey of the potentially dangerous chemicals found in rinse-off, leave-on, and makeup products in 2019.13 The authors added that the International Agency for Research on Cancer, in 2010, classified BP as potentially carcinogenic to humans (2B group).13,14
Promising use of nanocapsules
The widespread concern about the phototoxicity of BP has prompted some interesting research into workarounds. Specifically, in 2019, Barbosa et al. reported on the creation of a new sunscreen formulation using polymeric nanocapsules loading BP-3. The nanocapsules are made of poly(ε-caprolactone) carrot oil and Pluronic F68 (nonionic surfactant used in suspension cultures), and the BP-3–loaded capsules were found to be noncytotoxic in L929 fibroblast cell lines with a sun protection factor of 8.64. The researchers concluded that this promising nanocapsule may be an effective and safe way to use lipophilic sunscreen ingredients such as BP-3.15
Conclusion
The body of evidence is weighted against the use of BPs. Luckily, we have safe sunscreen choices that allow us to protect our skin without using these compounds.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Utsunomiya H et al. Chem Biol Interact. 2019 Jan 25;298:52-6.
2. Schneider SL and Lim HW. J Am Acad Dermatol. 2019 Jan;80(1):266-71.
3. Yeager DG and Lim HW. Dermatol Clin. 2019 Apr;37(2):149-57.
4. Ramos S et al. Sci Total Environ. 2015 Sep 1;526:278-311.
5. Siller A et al. Plast Surg Nur. 2019 Oct/Dec;39(4):157-60.
6. Krause M et al. Int J Androl. 2012 Jun;35(3):424-36.
7. Wang J and Ganley CJ. Clin Pharmacol Ther. 2019 Jan;105(1):161-7.
8. Matta MK et al. JAMA. 2020 Jan 21;323(3):256-67.
9. Warshaw EM et al. Dermatitis. 2013 Jul-Aug;24(4):176-82.
10. Amar SK et al. Toxicol Lett. 2015 Dec 15;239(3):182-93.
11. Amar SK et al. Toxicol Ind Health. 2019 Jul;35(7):457-65.
12. EWG. The trouble with ingredients in sunscreens. Accessed on 4 April 2020.
13. Panico A et al. J Prev Med Hyg. 2019 Mar 29;60(1):E50-7.
14. International Agency for Research on Cancer (IARC). Benzophenone. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. WHO, IARC Press, Lyon, France. 2010;101:285-304.
15. Barbosa TC et al. Toxics. 2019 Sep 22;7(4):51.
Benzophenones are a family of compounds that include dixoxybenzone, sulisobenzone, and benzophenone-3, or oxybenzone. These . Benzophenones (BPs) act as penetration enhancers, as they modify the structure of the skin and facilitate the absorption of other chemical ingredients into the body. The best known uses of these compounds are as perfume fixatives and sunscreen agents.
Sunscreens and benzophenones
BP-2, -3 and -4 are used as sunscreens but have many downsides. They are well known photoallergens, are toxic to aquatic animals (especially BP-3), and are found in urine. BP-2 has weak estrogenic effects, and some studies suggest that it decreases fertility in men. BP-4 can increase absorption of pesticides. BP-3 is banned in Hawaii because of the risk to coral and is the most worrisome.
In particular, BP-3 is known to protect skin and hair from UV radiation-induced harm.1 Unfortunately, BPs are also associated with photocontact allergies, hypersensitivity, hives, contact urticaria, anaphylaxis, hormone disruption, and DNA damage.2,3 BP-3 has also been implicated as an environmental contaminant. This column will focus on recent studies pertaining to effects on humans, primarily, and on the role of BPs in sunscreen agents.
Effects of BPs in animals
A recent study on the cytotoxicity of BP-3 against thymocytes in rats revealed that cell mortality increased significantly after 3 hours of exposure to 300 μM BP-3, but the membrane potential of thymocytes was unchanged by BP-3 exposure. In a concentration-dependent fashion, intracellular Zn2+ levels increased significantly after administration of at least 30 μM BP-3. The investigators concluded that the cytotoxicity engendered by BP-3 could be the result of oxidative stress linked to elevated intracellular Zn2+ levels.1
Effects of BPs in humans and systemic absorption
In multiple studies, exposure to BP-3, as well as to octinoxate, has been linked to endocrine and hormonal disruptions in humans and animals.4,5 Motivated by several notable observations (global increase in the use of sunscreens with UV filters; rapid rise in malignant melanoma, against which sunscreens should protect; increase in reported experimental findings of UV filters acting as endocrine disruptors), Krause et al. in 2012 reviewed animal and human data on the UV filters BP-3, 3-benzylidene camphor (3-BC), 3-(4-methyl-benzylidene) camphor (4-MBC), 2-ethylhexyl 4-methoxy cinnamate (OMC), homosalate (HMS), 2-ethylhexyl 4-dimethylaminobenzoate, and 4-aminobenzoic acid (PABA). Importantly, BP-3 was present in 96% of human urine samples in the United States, and various filters were found in 85% of the human breast milk samples in Switzerland.6
A 2019 analysis by Wang and Ganley reported that systemic absorption of the active sunscreen ingredient BP-3 can be substantial, justifying the assessment and understanding of systemic exposure to characterize the risks of long-term usage.7
Between January and February 2019, Matta et al. conducted a randomized clinical trial with 48 healthy participants to evaluate the systemic absorption and pharmacokinetics of six active ingredients in four sunscreen formulations, including avobenzone and BP-3. The researchers found that all ingredients were systemically absorbed, with plasma concentrations exceeding the Food and Drug Administration threshold for considering the waiving of further safety studies. They concluded that these results did not warrant discontinuing the use of the tested sunscreen ingredients.8 Yeager and Lim add that, while BP-3 has been incorporated into sunscreen formulations for sale in the United States since 1978, there have been no reports of adverse systemic reactions in human beings.3
However, topical reactions have elicited a different assessment. That is, in 2014, the American Contact Dermatitis Society labeled BPs the Contact Allergen of the Year, as they were identified as the most common source of photoallergic and contact allergic reactions of all UV filters.3,9
Risks of BPs in sunscreens and other skincare products
In 2015, Amar et al. investigated the photogenotoxicity and apoptotic effects in human keratinocytes (HaCaT cells) of BP-1, which is used as a UV blocker in sunscreens. They found that BP-1, when exposed to UV radiation, photosensitized cells and yielded intracellular reactive oxygen species. Significant reductions in cell viability were also seen with exposure to sunlight, UVA, and UVB. The researchers also confirmed genotoxic activity, with BP-1 augmenting lipid peroxidation and upregulating apoptotic proteins. They concluded that sunscreen users should be advised to avoid products that contain BP-1.10
In 2019, Amar et al. evaluated the effects of BPs on the differential expression of proteins in HaCaT cells exposed to UVA. Their findings indicated the expression of novel proteins that helped to initiate or promote apoptosis. They concluded that, because of the predilection to render such effects in human skin keratinocytes, consumers should avoid the use of sunscreens that contain BPs as UV blocking ingredients.11
Still widely used as an effective filter against UVA2 and UVB, BP-3 was believed to be present in two thirds of nonmineral sunscreens in the United States in 2018.3,12
Notably, BP-1 and BP-3 were found in small proportions (3.7% and 4.9%, respectively) among a total of 283 products culled from various stores in Lecce, Italy, in a survey of the potentially dangerous chemicals found in rinse-off, leave-on, and makeup products in 2019.13 The authors added that the International Agency for Research on Cancer, in 2010, classified BP as potentially carcinogenic to humans (2B group).13,14
Promising use of nanocapsules
The widespread concern about the phototoxicity of BP has prompted some interesting research into workarounds. Specifically, in 2019, Barbosa et al. reported on the creation of a new sunscreen formulation using polymeric nanocapsules loading BP-3. The nanocapsules are made of poly(ε-caprolactone) carrot oil and Pluronic F68 (nonionic surfactant used in suspension cultures), and the BP-3–loaded capsules were found to be noncytotoxic in L929 fibroblast cell lines with a sun protection factor of 8.64. The researchers concluded that this promising nanocapsule may be an effective and safe way to use lipophilic sunscreen ingredients such as BP-3.15
Conclusion
The body of evidence is weighted against the use of BPs. Luckily, we have safe sunscreen choices that allow us to protect our skin without using these compounds.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Utsunomiya H et al. Chem Biol Interact. 2019 Jan 25;298:52-6.
2. Schneider SL and Lim HW. J Am Acad Dermatol. 2019 Jan;80(1):266-71.
3. Yeager DG and Lim HW. Dermatol Clin. 2019 Apr;37(2):149-57.
4. Ramos S et al. Sci Total Environ. 2015 Sep 1;526:278-311.
5. Siller A et al. Plast Surg Nur. 2019 Oct/Dec;39(4):157-60.
6. Krause M et al. Int J Androl. 2012 Jun;35(3):424-36.
7. Wang J and Ganley CJ. Clin Pharmacol Ther. 2019 Jan;105(1):161-7.
8. Matta MK et al. JAMA. 2020 Jan 21;323(3):256-67.
9. Warshaw EM et al. Dermatitis. 2013 Jul-Aug;24(4):176-82.
10. Amar SK et al. Toxicol Lett. 2015 Dec 15;239(3):182-93.
11. Amar SK et al. Toxicol Ind Health. 2019 Jul;35(7):457-65.
12. EWG. The trouble with ingredients in sunscreens. Accessed on 4 April 2020.
13. Panico A et al. J Prev Med Hyg. 2019 Mar 29;60(1):E50-7.
14. International Agency for Research on Cancer (IARC). Benzophenone. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. WHO, IARC Press, Lyon, France. 2010;101:285-304.
15. Barbosa TC et al. Toxics. 2019 Sep 22;7(4):51.
Benzophenones are a family of compounds that include dixoxybenzone, sulisobenzone, and benzophenone-3, or oxybenzone. These . Benzophenones (BPs) act as penetration enhancers, as they modify the structure of the skin and facilitate the absorption of other chemical ingredients into the body. The best known uses of these compounds are as perfume fixatives and sunscreen agents.
Sunscreens and benzophenones
BP-2, -3 and -4 are used as sunscreens but have many downsides. They are well known photoallergens, are toxic to aquatic animals (especially BP-3), and are found in urine. BP-2 has weak estrogenic effects, and some studies suggest that it decreases fertility in men. BP-4 can increase absorption of pesticides. BP-3 is banned in Hawaii because of the risk to coral and is the most worrisome.
In particular, BP-3 is known to protect skin and hair from UV radiation-induced harm.1 Unfortunately, BPs are also associated with photocontact allergies, hypersensitivity, hives, contact urticaria, anaphylaxis, hormone disruption, and DNA damage.2,3 BP-3 has also been implicated as an environmental contaminant. This column will focus on recent studies pertaining to effects on humans, primarily, and on the role of BPs in sunscreen agents.
Effects of BPs in animals
A recent study on the cytotoxicity of BP-3 against thymocytes in rats revealed that cell mortality increased significantly after 3 hours of exposure to 300 μM BP-3, but the membrane potential of thymocytes was unchanged by BP-3 exposure. In a concentration-dependent fashion, intracellular Zn2+ levels increased significantly after administration of at least 30 μM BP-3. The investigators concluded that the cytotoxicity engendered by BP-3 could be the result of oxidative stress linked to elevated intracellular Zn2+ levels.1
Effects of BPs in humans and systemic absorption
In multiple studies, exposure to BP-3, as well as to octinoxate, has been linked to endocrine and hormonal disruptions in humans and animals.4,5 Motivated by several notable observations (global increase in the use of sunscreens with UV filters; rapid rise in malignant melanoma, against which sunscreens should protect; increase in reported experimental findings of UV filters acting as endocrine disruptors), Krause et al. in 2012 reviewed animal and human data on the UV filters BP-3, 3-benzylidene camphor (3-BC), 3-(4-methyl-benzylidene) camphor (4-MBC), 2-ethylhexyl 4-methoxy cinnamate (OMC), homosalate (HMS), 2-ethylhexyl 4-dimethylaminobenzoate, and 4-aminobenzoic acid (PABA). Importantly, BP-3 was present in 96% of human urine samples in the United States, and various filters were found in 85% of the human breast milk samples in Switzerland.6
A 2019 analysis by Wang and Ganley reported that systemic absorption of the active sunscreen ingredient BP-3 can be substantial, justifying the assessment and understanding of systemic exposure to characterize the risks of long-term usage.7
Between January and February 2019, Matta et al. conducted a randomized clinical trial with 48 healthy participants to evaluate the systemic absorption and pharmacokinetics of six active ingredients in four sunscreen formulations, including avobenzone and BP-3. The researchers found that all ingredients were systemically absorbed, with plasma concentrations exceeding the Food and Drug Administration threshold for considering the waiving of further safety studies. They concluded that these results did not warrant discontinuing the use of the tested sunscreen ingredients.8 Yeager and Lim add that, while BP-3 has been incorporated into sunscreen formulations for sale in the United States since 1978, there have been no reports of adverse systemic reactions in human beings.3
However, topical reactions have elicited a different assessment. That is, in 2014, the American Contact Dermatitis Society labeled BPs the Contact Allergen of the Year, as they were identified as the most common source of photoallergic and contact allergic reactions of all UV filters.3,9
Risks of BPs in sunscreens and other skincare products
In 2015, Amar et al. investigated the photogenotoxicity and apoptotic effects in human keratinocytes (HaCaT cells) of BP-1, which is used as a UV blocker in sunscreens. They found that BP-1, when exposed to UV radiation, photosensitized cells and yielded intracellular reactive oxygen species. Significant reductions in cell viability were also seen with exposure to sunlight, UVA, and UVB. The researchers also confirmed genotoxic activity, with BP-1 augmenting lipid peroxidation and upregulating apoptotic proteins. They concluded that sunscreen users should be advised to avoid products that contain BP-1.10
In 2019, Amar et al. evaluated the effects of BPs on the differential expression of proteins in HaCaT cells exposed to UVA. Their findings indicated the expression of novel proteins that helped to initiate or promote apoptosis. They concluded that, because of the predilection to render such effects in human skin keratinocytes, consumers should avoid the use of sunscreens that contain BPs as UV blocking ingredients.11
Still widely used as an effective filter against UVA2 and UVB, BP-3 was believed to be present in two thirds of nonmineral sunscreens in the United States in 2018.3,12
Notably, BP-1 and BP-3 were found in small proportions (3.7% and 4.9%, respectively) among a total of 283 products culled from various stores in Lecce, Italy, in a survey of the potentially dangerous chemicals found in rinse-off, leave-on, and makeup products in 2019.13 The authors added that the International Agency for Research on Cancer, in 2010, classified BP as potentially carcinogenic to humans (2B group).13,14
Promising use of nanocapsules
The widespread concern about the phototoxicity of BP has prompted some interesting research into workarounds. Specifically, in 2019, Barbosa et al. reported on the creation of a new sunscreen formulation using polymeric nanocapsules loading BP-3. The nanocapsules are made of poly(ε-caprolactone) carrot oil and Pluronic F68 (nonionic surfactant used in suspension cultures), and the BP-3–loaded capsules were found to be noncytotoxic in L929 fibroblast cell lines with a sun protection factor of 8.64. The researchers concluded that this promising nanocapsule may be an effective and safe way to use lipophilic sunscreen ingredients such as BP-3.15
Conclusion
The body of evidence is weighted against the use of BPs. Luckily, we have safe sunscreen choices that allow us to protect our skin without using these compounds.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions Inc., a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Utsunomiya H et al. Chem Biol Interact. 2019 Jan 25;298:52-6.
2. Schneider SL and Lim HW. J Am Acad Dermatol. 2019 Jan;80(1):266-71.
3. Yeager DG and Lim HW. Dermatol Clin. 2019 Apr;37(2):149-57.
4. Ramos S et al. Sci Total Environ. 2015 Sep 1;526:278-311.
5. Siller A et al. Plast Surg Nur. 2019 Oct/Dec;39(4):157-60.
6. Krause M et al. Int J Androl. 2012 Jun;35(3):424-36.
7. Wang J and Ganley CJ. Clin Pharmacol Ther. 2019 Jan;105(1):161-7.
8. Matta MK et al. JAMA. 2020 Jan 21;323(3):256-67.
9. Warshaw EM et al. Dermatitis. 2013 Jul-Aug;24(4):176-82.
10. Amar SK et al. Toxicol Lett. 2015 Dec 15;239(3):182-93.
11. Amar SK et al. Toxicol Ind Health. 2019 Jul;35(7):457-65.
12. EWG. The trouble with ingredients in sunscreens. Accessed on 4 April 2020.
13. Panico A et al. J Prev Med Hyg. 2019 Mar 29;60(1):E50-7.
14. International Agency for Research on Cancer (IARC). Benzophenone. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. WHO, IARC Press, Lyon, France. 2010;101:285-304.
15. Barbosa TC et al. Toxics. 2019 Sep 22;7(4):51.
HPV strains covered by the vaccine have declined greatly in the U.S.
Twelve years after the human papillomavirus (HPV) vaccination program was introduced in the United States, the overall prevalence of cancer-causing HPV strains covered by the vaccine dropped by 85% among females – 90% among vaccinated females and 74% among unvaccinated females – a strong sign of herd immunity, a new analysis of a nationally representative database is showing.
“HPV vaccination is working well,” Hannah Rosenblum, MD, Centers for Disease Control and Prevention, Atlanta, told this news organization in an email.
“Twelve years after introduction of HPV vaccination in the United States, national data demonstrate increasing impact among females and strong herd effects among unvaccinated females,” she added. “[Although] vaccination coverage and completion of the recommended dose in the United States is lower than coverage with other adolescent vaccinations, HPV vaccination is the best way to prevent HPV infections that can lead to several cancers in both females and males.”
The study was published online in Annals of Internal Medicine.
NHANES survey
The authors used data from the National Health and Nutrition Examination Survey (NHANES) to examine the four HPV types in the quadrivalent vaccine before 2003 and 2006 (the pre-vaccine era) and then again between 2007-2010, 2011-2014, and 2015-2018 (the vaccine era). For females, they analyzed demographic and HPV prevalence data across each 4-year era.
“Analyses were limited to sexually experienced participants, to ensure that all those included had an opportunity for HPV exposure, and to participants aged 14-24 years with adequate self-collected cervicovaginal specimens,” the authors explain.
This resulted in a sample size of 3,197 females. Demographic and HPV prevalence data were also collected from males but only during the 2013-2016 era, because those are the only years for which male HPV typing data are available in NHANES. Again, analyses were limited to sexually experienced males aged 14-24 years with adequate self-collected penile specimens, which resulted in a sample size of 661 males.
Over the 12 years of follow-up for females, there was a steady increase in females reporting having received at least one dose of the HPV vaccine – from slightly over 25% during 2007-12 to 59% during 2015-2018. The percentage of males who reported having at least one HPV dose also increased, from 29.5% in 2016 to 34.5% in 2018.
During the earliest vaccine era (2007-2010), the prevalence of the four HPV strains covered by the vaccine was 7.3% among vaccinated females, compared with 20.4% among unvaccinated females. “By 2015 to 2018, the prevalence was 2.8% (prevalence ratio, 0.16; 95% confidence interval, 0.07-0.39). The prevalence of the four vaccine-covered types was only 1.9% in vaccinated females, compared with 4.8% in unvaccinated females (PR, 0.40; 95% CI, 0.11-1.41).
In contrast, the prevalence of HPV types that were not covered by the vaccine showed little change – from 51.1% in the pre-vaccine era to 47.6% during 2015-2018 (PR, 0.93; 95% CI, 0.80-1.08). The authors considered this a good sign because it indicates that vaccine-type HPV infections are not being replaced with other oncogenic HPV infections. Between 2013 and 2016, the difference in the prevalence of the four HPV vaccine types was smaller at 1.8% among vaccinated males and 3.5% among unvaccinated males (PR, 0.49; 95% CI, 0.11-2.20).
Again, the prevalence of non-HPV vaccine types was not significantly different between vaccinated and unvaccinated males: 30.7% versus 34.3%.
During the vaccine era, effectiveness for females ranged from 60% to 84%. For males, vaccine effectiveness could only be evaluated for the single 4-year period from 2013 to 2016, and it was estimated at 51%. Dr. Rosenblum noted that vaccine efficacy estimates were lower on this national survey than the almost 100% efficacy rates observed in clinical trials in both males and females.
“This might be due in part to many participants receiving the vaccine at an older age than is recommended when they could have been infected [with HPV] at the time of vaccination,” Dr. Rosenblum said. She also noted that because males were incorporated into the HPV vaccination program years after females, they likely also experienced strong herd effects from the vaccine, making it challenging to estimate vaccine effectiveness.
Dr. Rosenblum also noted that there have already been documented declines in cervical precancers and high-grade vulvar and vaginal precancers, as well as genital warts and juvenile-onset recurrent respiratory papillomatosis. At the same time, the incidence of cervical precancers has recently declined among U.S. females in their late teens and early 20s – “likely reflecting the impact of vaccination,” she said.
“This study is good news for the United States HPV vaccination program, and all efforts are needed to ensure that children and adolescents receive routinely recommended vaccinations [including vaccination against HPV],” Dr. Rosenblum added.
Editorial comment
Commenting on the findings, Rebecca Perkins, MD, Boston University School of Medicine, and colleagues point out that the COVID-19 pandemic has led to disruptions in HPV vaccination programs and has reversed much of the progress made in recent years. “During the pandemic, providers and health systems have deprioritized adolescent vaccination and particularly HPV vaccination, which in turn has led to more severe drops for HPV vaccination than for other adolescent vaccinations, and for adolescent vaccination, compared with early childhood and adult vaccinations,” Dr. Perkins and colleagues write in an accompanying editorial.
Thus, the need to compensate for the cumulative deficit of missed vaccinations over the past 2 years has created a “serious and urgent threat” to cancer prevention efforts – “a shortfall from which it may take a decade to recover,” the editorialists predict. To try and reverse this trend, several practices have been shown to improve HPV vaccination rates.
The first is a strong provider recommendation such as, “Your child is due for an HPV vaccine today.” The second is to give standing orders to allow nurses and medical assistants to administer vaccinations without requiring intervention by a physician. Lastly, programs to remind patients when vaccines are due and to recall them for appointments also work well.
“Using evidence-based methods and redoubling our efforts to prioritize HPV vaccination will be crucial to ensuring that we do not lose a generation to preventable HPV-associated cancer,” write Dr. Perkins and colleagues.
The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Twelve years after the human papillomavirus (HPV) vaccination program was introduced in the United States, the overall prevalence of cancer-causing HPV strains covered by the vaccine dropped by 85% among females – 90% among vaccinated females and 74% among unvaccinated females – a strong sign of herd immunity, a new analysis of a nationally representative database is showing.
“HPV vaccination is working well,” Hannah Rosenblum, MD, Centers for Disease Control and Prevention, Atlanta, told this news organization in an email.
“Twelve years after introduction of HPV vaccination in the United States, national data demonstrate increasing impact among females and strong herd effects among unvaccinated females,” she added. “[Although] vaccination coverage and completion of the recommended dose in the United States is lower than coverage with other adolescent vaccinations, HPV vaccination is the best way to prevent HPV infections that can lead to several cancers in both females and males.”
The study was published online in Annals of Internal Medicine.
NHANES survey
The authors used data from the National Health and Nutrition Examination Survey (NHANES) to examine the four HPV types in the quadrivalent vaccine before 2003 and 2006 (the pre-vaccine era) and then again between 2007-2010, 2011-2014, and 2015-2018 (the vaccine era). For females, they analyzed demographic and HPV prevalence data across each 4-year era.
“Analyses were limited to sexually experienced participants, to ensure that all those included had an opportunity for HPV exposure, and to participants aged 14-24 years with adequate self-collected cervicovaginal specimens,” the authors explain.
This resulted in a sample size of 3,197 females. Demographic and HPV prevalence data were also collected from males but only during the 2013-2016 era, because those are the only years for which male HPV typing data are available in NHANES. Again, analyses were limited to sexually experienced males aged 14-24 years with adequate self-collected penile specimens, which resulted in a sample size of 661 males.
Over the 12 years of follow-up for females, there was a steady increase in females reporting having received at least one dose of the HPV vaccine – from slightly over 25% during 2007-12 to 59% during 2015-2018. The percentage of males who reported having at least one HPV dose also increased, from 29.5% in 2016 to 34.5% in 2018.
During the earliest vaccine era (2007-2010), the prevalence of the four HPV strains covered by the vaccine was 7.3% among vaccinated females, compared with 20.4% among unvaccinated females. “By 2015 to 2018, the prevalence was 2.8% (prevalence ratio, 0.16; 95% confidence interval, 0.07-0.39). The prevalence of the four vaccine-covered types was only 1.9% in vaccinated females, compared with 4.8% in unvaccinated females (PR, 0.40; 95% CI, 0.11-1.41).
In contrast, the prevalence of HPV types that were not covered by the vaccine showed little change – from 51.1% in the pre-vaccine era to 47.6% during 2015-2018 (PR, 0.93; 95% CI, 0.80-1.08). The authors considered this a good sign because it indicates that vaccine-type HPV infections are not being replaced with other oncogenic HPV infections. Between 2013 and 2016, the difference in the prevalence of the four HPV vaccine types was smaller at 1.8% among vaccinated males and 3.5% among unvaccinated males (PR, 0.49; 95% CI, 0.11-2.20).
Again, the prevalence of non-HPV vaccine types was not significantly different between vaccinated and unvaccinated males: 30.7% versus 34.3%.
During the vaccine era, effectiveness for females ranged from 60% to 84%. For males, vaccine effectiveness could only be evaluated for the single 4-year period from 2013 to 2016, and it was estimated at 51%. Dr. Rosenblum noted that vaccine efficacy estimates were lower on this national survey than the almost 100% efficacy rates observed in clinical trials in both males and females.
“This might be due in part to many participants receiving the vaccine at an older age than is recommended when they could have been infected [with HPV] at the time of vaccination,” Dr. Rosenblum said. She also noted that because males were incorporated into the HPV vaccination program years after females, they likely also experienced strong herd effects from the vaccine, making it challenging to estimate vaccine effectiveness.
Dr. Rosenblum also noted that there have already been documented declines in cervical precancers and high-grade vulvar and vaginal precancers, as well as genital warts and juvenile-onset recurrent respiratory papillomatosis. At the same time, the incidence of cervical precancers has recently declined among U.S. females in their late teens and early 20s – “likely reflecting the impact of vaccination,” she said.
“This study is good news for the United States HPV vaccination program, and all efforts are needed to ensure that children and adolescents receive routinely recommended vaccinations [including vaccination against HPV],” Dr. Rosenblum added.
Editorial comment
Commenting on the findings, Rebecca Perkins, MD, Boston University School of Medicine, and colleagues point out that the COVID-19 pandemic has led to disruptions in HPV vaccination programs and has reversed much of the progress made in recent years. “During the pandemic, providers and health systems have deprioritized adolescent vaccination and particularly HPV vaccination, which in turn has led to more severe drops for HPV vaccination than for other adolescent vaccinations, and for adolescent vaccination, compared with early childhood and adult vaccinations,” Dr. Perkins and colleagues write in an accompanying editorial.
Thus, the need to compensate for the cumulative deficit of missed vaccinations over the past 2 years has created a “serious and urgent threat” to cancer prevention efforts – “a shortfall from which it may take a decade to recover,” the editorialists predict. To try and reverse this trend, several practices have been shown to improve HPV vaccination rates.
The first is a strong provider recommendation such as, “Your child is due for an HPV vaccine today.” The second is to give standing orders to allow nurses and medical assistants to administer vaccinations without requiring intervention by a physician. Lastly, programs to remind patients when vaccines are due and to recall them for appointments also work well.
“Using evidence-based methods and redoubling our efforts to prioritize HPV vaccination will be crucial to ensuring that we do not lose a generation to preventable HPV-associated cancer,” write Dr. Perkins and colleagues.
The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Twelve years after the human papillomavirus (HPV) vaccination program was introduced in the United States, the overall prevalence of cancer-causing HPV strains covered by the vaccine dropped by 85% among females – 90% among vaccinated females and 74% among unvaccinated females – a strong sign of herd immunity, a new analysis of a nationally representative database is showing.
“HPV vaccination is working well,” Hannah Rosenblum, MD, Centers for Disease Control and Prevention, Atlanta, told this news organization in an email.
“Twelve years after introduction of HPV vaccination in the United States, national data demonstrate increasing impact among females and strong herd effects among unvaccinated females,” she added. “[Although] vaccination coverage and completion of the recommended dose in the United States is lower than coverage with other adolescent vaccinations, HPV vaccination is the best way to prevent HPV infections that can lead to several cancers in both females and males.”
The study was published online in Annals of Internal Medicine.
NHANES survey
The authors used data from the National Health and Nutrition Examination Survey (NHANES) to examine the four HPV types in the quadrivalent vaccine before 2003 and 2006 (the pre-vaccine era) and then again between 2007-2010, 2011-2014, and 2015-2018 (the vaccine era). For females, they analyzed demographic and HPV prevalence data across each 4-year era.
“Analyses were limited to sexually experienced participants, to ensure that all those included had an opportunity for HPV exposure, and to participants aged 14-24 years with adequate self-collected cervicovaginal specimens,” the authors explain.
This resulted in a sample size of 3,197 females. Demographic and HPV prevalence data were also collected from males but only during the 2013-2016 era, because those are the only years for which male HPV typing data are available in NHANES. Again, analyses were limited to sexually experienced males aged 14-24 years with adequate self-collected penile specimens, which resulted in a sample size of 661 males.
Over the 12 years of follow-up for females, there was a steady increase in females reporting having received at least one dose of the HPV vaccine – from slightly over 25% during 2007-12 to 59% during 2015-2018. The percentage of males who reported having at least one HPV dose also increased, from 29.5% in 2016 to 34.5% in 2018.
During the earliest vaccine era (2007-2010), the prevalence of the four HPV strains covered by the vaccine was 7.3% among vaccinated females, compared with 20.4% among unvaccinated females. “By 2015 to 2018, the prevalence was 2.8% (prevalence ratio, 0.16; 95% confidence interval, 0.07-0.39). The prevalence of the four vaccine-covered types was only 1.9% in vaccinated females, compared with 4.8% in unvaccinated females (PR, 0.40; 95% CI, 0.11-1.41).
In contrast, the prevalence of HPV types that were not covered by the vaccine showed little change – from 51.1% in the pre-vaccine era to 47.6% during 2015-2018 (PR, 0.93; 95% CI, 0.80-1.08). The authors considered this a good sign because it indicates that vaccine-type HPV infections are not being replaced with other oncogenic HPV infections. Between 2013 and 2016, the difference in the prevalence of the four HPV vaccine types was smaller at 1.8% among vaccinated males and 3.5% among unvaccinated males (PR, 0.49; 95% CI, 0.11-2.20).
Again, the prevalence of non-HPV vaccine types was not significantly different between vaccinated and unvaccinated males: 30.7% versus 34.3%.
During the vaccine era, effectiveness for females ranged from 60% to 84%. For males, vaccine effectiveness could only be evaluated for the single 4-year period from 2013 to 2016, and it was estimated at 51%. Dr. Rosenblum noted that vaccine efficacy estimates were lower on this national survey than the almost 100% efficacy rates observed in clinical trials in both males and females.
“This might be due in part to many participants receiving the vaccine at an older age than is recommended when they could have been infected [with HPV] at the time of vaccination,” Dr. Rosenblum said. She also noted that because males were incorporated into the HPV vaccination program years after females, they likely also experienced strong herd effects from the vaccine, making it challenging to estimate vaccine effectiveness.
Dr. Rosenblum also noted that there have already been documented declines in cervical precancers and high-grade vulvar and vaginal precancers, as well as genital warts and juvenile-onset recurrent respiratory papillomatosis. At the same time, the incidence of cervical precancers has recently declined among U.S. females in their late teens and early 20s – “likely reflecting the impact of vaccination,” she said.
“This study is good news for the United States HPV vaccination program, and all efforts are needed to ensure that children and adolescents receive routinely recommended vaccinations [including vaccination against HPV],” Dr. Rosenblum added.
Editorial comment
Commenting on the findings, Rebecca Perkins, MD, Boston University School of Medicine, and colleagues point out that the COVID-19 pandemic has led to disruptions in HPV vaccination programs and has reversed much of the progress made in recent years. “During the pandemic, providers and health systems have deprioritized adolescent vaccination and particularly HPV vaccination, which in turn has led to more severe drops for HPV vaccination than for other adolescent vaccinations, and for adolescent vaccination, compared with early childhood and adult vaccinations,” Dr. Perkins and colleagues write in an accompanying editorial.
Thus, the need to compensate for the cumulative deficit of missed vaccinations over the past 2 years has created a “serious and urgent threat” to cancer prevention efforts – “a shortfall from which it may take a decade to recover,” the editorialists predict. To try and reverse this trend, several practices have been shown to improve HPV vaccination rates.
The first is a strong provider recommendation such as, “Your child is due for an HPV vaccine today.” The second is to give standing orders to allow nurses and medical assistants to administer vaccinations without requiring intervention by a physician. Lastly, programs to remind patients when vaccines are due and to recall them for appointments also work well.
“Using evidence-based methods and redoubling our efforts to prioritize HPV vaccination will be crucial to ensuring that we do not lose a generation to preventable HPV-associated cancer,” write Dr. Perkins and colleagues.
The study authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
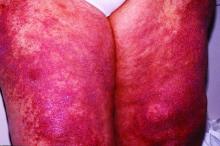
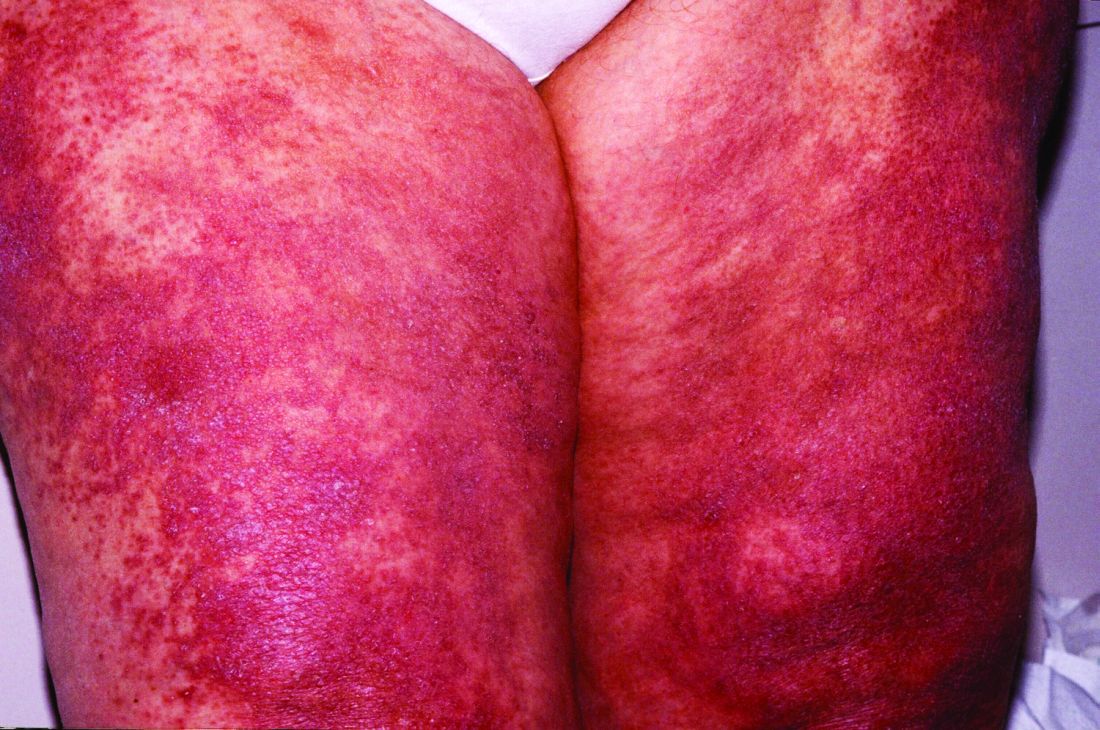
Lenabasum improved skin symptoms in dermatomyositis, but future is uncertain
An – some of it statistically significant – in a phase 2, double-blind, randomized, controlled study.
Patients taking lenabasum experienced greater reductions in the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) – a validated outcome designed to assess inflammatory skin involvement in the rare autoimmune disease – and improvements in patient-reported and biomarker outcomes, compared with those on placebo, dermatologist Victoria P. Werth, MD, and coinvestigators reported.
And in a recently completed phase 3 trial, reported by the manufacturer, a subpopulation of patients with active skin disease and no active muscle disease again showed greater reductions in CDASI activity scores – a secondary outcome in the trial.
However, the phase 3 DETERMINE trial produced negative findings overall. It enrolled a more heterogeneous group of patients – including those with both muscle weakness and skin involvement – and its primary outcome measure was a broader composite measure, the Total Improvement Score. The trial failed to meet this primary endpoint, Corbus Pharmaceuticals, the developer of lenabasum, announced in a press release in June 2021.
The phase 3 results are “frustrating” for patients with symptomatic and refractory skin manifestations of dermatomyositis (DM), given the promising findings from the phase 2 trial and from an open-label extension study, said Dr. Werth, professor of dermatology and medicine, University of Pennsylvania, Philadelphia, and principal investigator and coprincipal investigator of the phase 2 and phase 3 studies, respectively.
Dr. Werth is scheduled to present the results from the phase 3 trial at the annual European Alliance of Associations for Rheumatology meeting in June.
“With lenabasum, we have a therapy that doesn’t work for every patient, but does work for quite a number of them,” Dr. Werth said in an interview. “It’s oral, it’s not really that immunosuppressing, and there aren’t many side effects. Right now, patients are often being managed with steroids ... we really need treatments that are not as toxic.”
Robert Spiera, MD, a rheumatologist who led trials of lenabasum for treatment of diffuse cutaneous systemic sclerosis (dcSSc), agreed. “The CB2 agonist strategy is appealing because it’s nonimmunosuppressing and has both anti-inflammatory and antifibrotic properties,” he said in an interview. “I wouldn’t want to give up on it ... especially [for patients] with scleroderma and dermatomyositis who are treated with substantial drugs that are associated with morbidity.”
Lenabasum, he said, has proven to be “incredibly safe, and incredibly safe in the long term.”
While the phase 2 trial of the drug for dcSSc showed clear benefit over placebo, the phase 3 trial did not meet its primary endpoint using the American College of Rheumatology Combined Response Index in Diffuse Cutaneous Systemic Sclerosis.
It allowed background immunosuppressant therapy to reflect real-world clinical practice, and “there was such a high response rate to [that therapy, largely mycophenolate] that there was little room to show benefit beyond that,” said Dr. Spiera, director of the vasculitis and scleroderma program, Hospital for Special Surgery, New York.
The drug led to more improvement in the small subset of participants who were not receiving background immunotherapy during the trial, he noted.
Corbus is currently “seeking a partnership to further explore the drug” for treatment in different subpopulations, according to a company spokesperson. Results of a phase 2 trial of lenabasum for the treatment of systemic lupus erythematosus – with a pain rating as the primary outcome measure – are expected soon.
Phase 2 findings
The single-center phase 2 trial of lenabasum for DM enrolled 22 adults with minimal muscle involvement as evidenced by normal maximal resistance on muscle testing at entry and throughout the study. Most were taking immunosuppressant medication, and all had CDASI scores of at least 20, with mean scores in the severe range (> 26). Symptoms registered on patient-reported outcome measures were moderate to severe.
Patients received a half-dose of lenabasum (20 mg daily) for 1 month and a full dose (20 mg twice daily) for 2 months, or placebo, and were followed for an additional month without dosing.
Starting at day 43 – approximately 2 weeks after the dose was increased – there was “a trend for the change from baseline CDASI to be greater” in the lenabasum group, compared with those on placebo, Dr. Werth and colleagues reported. The differences reached statistical significance on day 113 (P = .038), a month after patients discontinued lenabasum, “suggesting that the modulation of the inflammatory response by lenabasum continued beyond its last dose.”
Five of the 11 patients treated with lenabasum (45%), and none of those on placebo, achieved at least a 40% reduction in the CDASI activity score by the end of the study.
Patients in the lenabasum group also had greater improvement in the Skindex-29 Symptoms scores – an objective measure of itch – and improvements in other secondary efficacy outcomes, including pain, though these did not reach statistical significance.
Skin biopsies before and after treatment showed significant reductions in inflammatory cytokines relevant to DM pathogenesis. Patients treated with the CB2 agonist had a downward trend in the CD4+ T cell population, which correlated with decreased CDASI activity scores, for instance, and a decrease in IL-31 protein expression, which correlated with decreased Skindex-29 Symptoms scores, the investigators reported.
There were no serious adverse events related to the CB2 agonist, and no treatment discontinuations.
The main part of the phase 2 trial, conducted from 2015 to 2017, was followed by a 3-year, open-label extension, in which 20 of the 22 patients took lenabasum 20 mg twice a day. The drug continued to be safe and well tolerated, and the CDASI activity score and other outcomes improved through year 1 and remained stable thereafter, according to a poster presented by Dr. Werth at the 2021 EULAR meeting.
After 1 year in the open-label extension, 60%-70% of patients had achieved mild skin disease, and 75% had achieved at least a 40% reduction in CDASI activity.
“A lot of patients, even if they weren’t completely cleared, were much happier in terms of their itch,” said Dr. Werth, also chief of dermatology, Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia. “It’s been difficult for a lot of them now that they’re off the long-term extension ... a lot of them are flaring.”
The future
In the lab, with funding from the National Institutes of Health, Dr. Werth is continuing to investigate how lenabasum may be working in DM. A paper just published in the open access journal Arthritis Research & Therapy describes CB2 receptor distribution and up-regulation on key immune cells in the skin and blood, and how, in DM skin, its highest expression is on dendritic cells.
Through both mechanistic and more clinical research, “it’s important to understand the characteristics of the people [lenabasum] worked in or didn’t work in,” she said.
And in clinical trials, it’s important to capture meaningful improvement from the patient perspective. “It may be,” she noted, “that more global, systemic assessments are not the way to go for autoimmune skin disease.”
For dcSSc, Dr. Spiera said, it’s possible that a CB2 agonist may be helpful for patients who have been on immunosuppressants, particularly mycophenolate, for more than 6 months “and are still struggling.”
The phase 2 trial in DM was funded by the National Institutes of Health, the Department of Veterans Affairs, and Corbus Pharmaceuticals. The phase 3 trials in DM and in dcSSc were funded by Corbus. Dr. Werth disclosed grant support from Corbus and several other pharmaceutical companies. Dr. Spiera disclosed that he has received grant support or consulting fees from Roche-Genentech, GlaxoSmithKline, and several other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
An – some of it statistically significant – in a phase 2, double-blind, randomized, controlled study.
Patients taking lenabasum experienced greater reductions in the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) – a validated outcome designed to assess inflammatory skin involvement in the rare autoimmune disease – and improvements in patient-reported and biomarker outcomes, compared with those on placebo, dermatologist Victoria P. Werth, MD, and coinvestigators reported.
And in a recently completed phase 3 trial, reported by the manufacturer, a subpopulation of patients with active skin disease and no active muscle disease again showed greater reductions in CDASI activity scores – a secondary outcome in the trial.
However, the phase 3 DETERMINE trial produced negative findings overall. It enrolled a more heterogeneous group of patients – including those with both muscle weakness and skin involvement – and its primary outcome measure was a broader composite measure, the Total Improvement Score. The trial failed to meet this primary endpoint, Corbus Pharmaceuticals, the developer of lenabasum, announced in a press release in June 2021.
The phase 3 results are “frustrating” for patients with symptomatic and refractory skin manifestations of dermatomyositis (DM), given the promising findings from the phase 2 trial and from an open-label extension study, said Dr. Werth, professor of dermatology and medicine, University of Pennsylvania, Philadelphia, and principal investigator and coprincipal investigator of the phase 2 and phase 3 studies, respectively.
Dr. Werth is scheduled to present the results from the phase 3 trial at the annual European Alliance of Associations for Rheumatology meeting in June.
“With lenabasum, we have a therapy that doesn’t work for every patient, but does work for quite a number of them,” Dr. Werth said in an interview. “It’s oral, it’s not really that immunosuppressing, and there aren’t many side effects. Right now, patients are often being managed with steroids ... we really need treatments that are not as toxic.”
Robert Spiera, MD, a rheumatologist who led trials of lenabasum for treatment of diffuse cutaneous systemic sclerosis (dcSSc), agreed. “The CB2 agonist strategy is appealing because it’s nonimmunosuppressing and has both anti-inflammatory and antifibrotic properties,” he said in an interview. “I wouldn’t want to give up on it ... especially [for patients] with scleroderma and dermatomyositis who are treated with substantial drugs that are associated with morbidity.”
Lenabasum, he said, has proven to be “incredibly safe, and incredibly safe in the long term.”
While the phase 2 trial of the drug for dcSSc showed clear benefit over placebo, the phase 3 trial did not meet its primary endpoint using the American College of Rheumatology Combined Response Index in Diffuse Cutaneous Systemic Sclerosis.
It allowed background immunosuppressant therapy to reflect real-world clinical practice, and “there was such a high response rate to [that therapy, largely mycophenolate] that there was little room to show benefit beyond that,” said Dr. Spiera, director of the vasculitis and scleroderma program, Hospital for Special Surgery, New York.
The drug led to more improvement in the small subset of participants who were not receiving background immunotherapy during the trial, he noted.
Corbus is currently “seeking a partnership to further explore the drug” for treatment in different subpopulations, according to a company spokesperson. Results of a phase 2 trial of lenabasum for the treatment of systemic lupus erythematosus – with a pain rating as the primary outcome measure – are expected soon.
Phase 2 findings
The single-center phase 2 trial of lenabasum for DM enrolled 22 adults with minimal muscle involvement as evidenced by normal maximal resistance on muscle testing at entry and throughout the study. Most were taking immunosuppressant medication, and all had CDASI scores of at least 20, with mean scores in the severe range (> 26). Symptoms registered on patient-reported outcome measures were moderate to severe.
Patients received a half-dose of lenabasum (20 mg daily) for 1 month and a full dose (20 mg twice daily) for 2 months, or placebo, and were followed for an additional month without dosing.
Starting at day 43 – approximately 2 weeks after the dose was increased – there was “a trend for the change from baseline CDASI to be greater” in the lenabasum group, compared with those on placebo, Dr. Werth and colleagues reported. The differences reached statistical significance on day 113 (P = .038), a month after patients discontinued lenabasum, “suggesting that the modulation of the inflammatory response by lenabasum continued beyond its last dose.”
Five of the 11 patients treated with lenabasum (45%), and none of those on placebo, achieved at least a 40% reduction in the CDASI activity score by the end of the study.
Patients in the lenabasum group also had greater improvement in the Skindex-29 Symptoms scores – an objective measure of itch – and improvements in other secondary efficacy outcomes, including pain, though these did not reach statistical significance.
Skin biopsies before and after treatment showed significant reductions in inflammatory cytokines relevant to DM pathogenesis. Patients treated with the CB2 agonist had a downward trend in the CD4+ T cell population, which correlated with decreased CDASI activity scores, for instance, and a decrease in IL-31 protein expression, which correlated with decreased Skindex-29 Symptoms scores, the investigators reported.
There were no serious adverse events related to the CB2 agonist, and no treatment discontinuations.
The main part of the phase 2 trial, conducted from 2015 to 2017, was followed by a 3-year, open-label extension, in which 20 of the 22 patients took lenabasum 20 mg twice a day. The drug continued to be safe and well tolerated, and the CDASI activity score and other outcomes improved through year 1 and remained stable thereafter, according to a poster presented by Dr. Werth at the 2021 EULAR meeting.
After 1 year in the open-label extension, 60%-70% of patients had achieved mild skin disease, and 75% had achieved at least a 40% reduction in CDASI activity.
“A lot of patients, even if they weren’t completely cleared, were much happier in terms of their itch,” said Dr. Werth, also chief of dermatology, Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia. “It’s been difficult for a lot of them now that they’re off the long-term extension ... a lot of them are flaring.”
The future
In the lab, with funding from the National Institutes of Health, Dr. Werth is continuing to investigate how lenabasum may be working in DM. A paper just published in the open access journal Arthritis Research & Therapy describes CB2 receptor distribution and up-regulation on key immune cells in the skin and blood, and how, in DM skin, its highest expression is on dendritic cells.
Through both mechanistic and more clinical research, “it’s important to understand the characteristics of the people [lenabasum] worked in or didn’t work in,” she said.
And in clinical trials, it’s important to capture meaningful improvement from the patient perspective. “It may be,” she noted, “that more global, systemic assessments are not the way to go for autoimmune skin disease.”
For dcSSc, Dr. Spiera said, it’s possible that a CB2 agonist may be helpful for patients who have been on immunosuppressants, particularly mycophenolate, for more than 6 months “and are still struggling.”
The phase 2 trial in DM was funded by the National Institutes of Health, the Department of Veterans Affairs, and Corbus Pharmaceuticals. The phase 3 trials in DM and in dcSSc were funded by Corbus. Dr. Werth disclosed grant support from Corbus and several other pharmaceutical companies. Dr. Spiera disclosed that he has received grant support or consulting fees from Roche-Genentech, GlaxoSmithKline, and several other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
An – some of it statistically significant – in a phase 2, double-blind, randomized, controlled study.
Patients taking lenabasum experienced greater reductions in the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) – a validated outcome designed to assess inflammatory skin involvement in the rare autoimmune disease – and improvements in patient-reported and biomarker outcomes, compared with those on placebo, dermatologist Victoria P. Werth, MD, and coinvestigators reported.
And in a recently completed phase 3 trial, reported by the manufacturer, a subpopulation of patients with active skin disease and no active muscle disease again showed greater reductions in CDASI activity scores – a secondary outcome in the trial.
However, the phase 3 DETERMINE trial produced negative findings overall. It enrolled a more heterogeneous group of patients – including those with both muscle weakness and skin involvement – and its primary outcome measure was a broader composite measure, the Total Improvement Score. The trial failed to meet this primary endpoint, Corbus Pharmaceuticals, the developer of lenabasum, announced in a press release in June 2021.
The phase 3 results are “frustrating” for patients with symptomatic and refractory skin manifestations of dermatomyositis (DM), given the promising findings from the phase 2 trial and from an open-label extension study, said Dr. Werth, professor of dermatology and medicine, University of Pennsylvania, Philadelphia, and principal investigator and coprincipal investigator of the phase 2 and phase 3 studies, respectively.
Dr. Werth is scheduled to present the results from the phase 3 trial at the annual European Alliance of Associations for Rheumatology meeting in June.
“With lenabasum, we have a therapy that doesn’t work for every patient, but does work for quite a number of them,” Dr. Werth said in an interview. “It’s oral, it’s not really that immunosuppressing, and there aren’t many side effects. Right now, patients are often being managed with steroids ... we really need treatments that are not as toxic.”
Robert Spiera, MD, a rheumatologist who led trials of lenabasum for treatment of diffuse cutaneous systemic sclerosis (dcSSc), agreed. “The CB2 agonist strategy is appealing because it’s nonimmunosuppressing and has both anti-inflammatory and antifibrotic properties,” he said in an interview. “I wouldn’t want to give up on it ... especially [for patients] with scleroderma and dermatomyositis who are treated with substantial drugs that are associated with morbidity.”
Lenabasum, he said, has proven to be “incredibly safe, and incredibly safe in the long term.”
While the phase 2 trial of the drug for dcSSc showed clear benefit over placebo, the phase 3 trial did not meet its primary endpoint using the American College of Rheumatology Combined Response Index in Diffuse Cutaneous Systemic Sclerosis.
It allowed background immunosuppressant therapy to reflect real-world clinical practice, and “there was such a high response rate to [that therapy, largely mycophenolate] that there was little room to show benefit beyond that,” said Dr. Spiera, director of the vasculitis and scleroderma program, Hospital for Special Surgery, New York.
The drug led to more improvement in the small subset of participants who were not receiving background immunotherapy during the trial, he noted.
Corbus is currently “seeking a partnership to further explore the drug” for treatment in different subpopulations, according to a company spokesperson. Results of a phase 2 trial of lenabasum for the treatment of systemic lupus erythematosus – with a pain rating as the primary outcome measure – are expected soon.
Phase 2 findings
The single-center phase 2 trial of lenabasum for DM enrolled 22 adults with minimal muscle involvement as evidenced by normal maximal resistance on muscle testing at entry and throughout the study. Most were taking immunosuppressant medication, and all had CDASI scores of at least 20, with mean scores in the severe range (> 26). Symptoms registered on patient-reported outcome measures were moderate to severe.
Patients received a half-dose of lenabasum (20 mg daily) for 1 month and a full dose (20 mg twice daily) for 2 months, or placebo, and were followed for an additional month without dosing.
Starting at day 43 – approximately 2 weeks after the dose was increased – there was “a trend for the change from baseline CDASI to be greater” in the lenabasum group, compared with those on placebo, Dr. Werth and colleagues reported. The differences reached statistical significance on day 113 (P = .038), a month after patients discontinued lenabasum, “suggesting that the modulation of the inflammatory response by lenabasum continued beyond its last dose.”
Five of the 11 patients treated with lenabasum (45%), and none of those on placebo, achieved at least a 40% reduction in the CDASI activity score by the end of the study.
Patients in the lenabasum group also had greater improvement in the Skindex-29 Symptoms scores – an objective measure of itch – and improvements in other secondary efficacy outcomes, including pain, though these did not reach statistical significance.
Skin biopsies before and after treatment showed significant reductions in inflammatory cytokines relevant to DM pathogenesis. Patients treated with the CB2 agonist had a downward trend in the CD4+ T cell population, which correlated with decreased CDASI activity scores, for instance, and a decrease in IL-31 protein expression, which correlated with decreased Skindex-29 Symptoms scores, the investigators reported.
There were no serious adverse events related to the CB2 agonist, and no treatment discontinuations.
The main part of the phase 2 trial, conducted from 2015 to 2017, was followed by a 3-year, open-label extension, in which 20 of the 22 patients took lenabasum 20 mg twice a day. The drug continued to be safe and well tolerated, and the CDASI activity score and other outcomes improved through year 1 and remained stable thereafter, according to a poster presented by Dr. Werth at the 2021 EULAR meeting.
After 1 year in the open-label extension, 60%-70% of patients had achieved mild skin disease, and 75% had achieved at least a 40% reduction in CDASI activity.
“A lot of patients, even if they weren’t completely cleared, were much happier in terms of their itch,” said Dr. Werth, also chief of dermatology, Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia. “It’s been difficult for a lot of them now that they’re off the long-term extension ... a lot of them are flaring.”
The future
In the lab, with funding from the National Institutes of Health, Dr. Werth is continuing to investigate how lenabasum may be working in DM. A paper just published in the open access journal Arthritis Research & Therapy describes CB2 receptor distribution and up-regulation on key immune cells in the skin and blood, and how, in DM skin, its highest expression is on dendritic cells.
Through both mechanistic and more clinical research, “it’s important to understand the characteristics of the people [lenabasum] worked in or didn’t work in,” she said.
And in clinical trials, it’s important to capture meaningful improvement from the patient perspective. “It may be,” she noted, “that more global, systemic assessments are not the way to go for autoimmune skin disease.”
For dcSSc, Dr. Spiera said, it’s possible that a CB2 agonist may be helpful for patients who have been on immunosuppressants, particularly mycophenolate, for more than 6 months “and are still struggling.”
The phase 2 trial in DM was funded by the National Institutes of Health, the Department of Veterans Affairs, and Corbus Pharmaceuticals. The phase 3 trials in DM and in dcSSc were funded by Corbus. Dr. Werth disclosed grant support from Corbus and several other pharmaceutical companies. Dr. Spiera disclosed that he has received grant support or consulting fees from Roche-Genentech, GlaxoSmithKline, and several other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Stroke in the young: Cancer in disguise?
The data were presented by Jamie Verhoeven, MD, Radboud University Medical Centre, the Netherlands, at the recent European Stroke Organisation Conference 2022.
Dr. Verhoeven noted that 10% of all stroke cases occur in individuals younger than 50 years. During the past few decades, the incidence of stroke in the young has steadily increased, whereas the incidence of stroke in older adults has stabilized or decreased.
“Stroke in the young differs from stroke in older patients, and one of the major differences is that stroke in the young has a higher proportion of cryptogenic stroke, with no clear cause found in over one-third of patients,” she said.
Also, having an active cancer is known to be a risk factor for thrombosis. This association is strongest in venous thrombosis and has been less well investigated in arterial thrombosis, Dr. Verhoeven reported.
Her group aimed to investigate whether in some patients with cryptogenic stroke, this may be the first manifestation of an underlying cancer. “If this hypothesis is true, then it would be more obvious in young patients who have a higher incidence of cryptogenic stroke,” she said.
They performed a population-based observational cohort study using diagnostic ICD codes from the national Hospital Discharge Registry in the Netherlands and the Dutch Population Registry from 1998 to 2019.
Patients with a history of cancer before their first stroke and those with central nervous system cancers at the time of stroke or nonmelanoma skin cancers (which have been shown to have no systemic effects) were excluded.
Reference data came from the Netherlands Comprehensive Cancer Organisation, which collects data on all cancer diagnoses in the country.
The researchers identified 27,616 young stroke patients (age range, 15-49 years; median age, 45 years) and 362,782 older stroke patients (age range, 50 years and older; median age, 76 years).
The cumulative incidence of any cancer at 10 years was 3.7% in the younger group and 8.5% in the older group.
The data were compared with matched peers from the general population. The main outcome measures were cumulative incidence of first-ever cancer after stroke (stratified by stroke subtype, age and sex) and standardized incidence rates.
Results showed that the risk for cancer was higher in the younger age group than in the matched general population.
In this age group, the 1-year risk of any new cancer was 2.6 times higher (95% confidence interval, 2.2-3.1) after ischemic stroke and 5.4 times (95% CI, 3.8-7.3) after intracerebral hemorrhage than in matched peers from the general population.
In contrast, in stroke patients older than 50 years, the 1-year risk for any new cancer was 1.2 times higher than the general population after either ischemic or hemorrhagic stroke.
“The younger patients have a higher risk increase of cancer than older patients, and this risk increase is most evident in the first 1 to 2 years after stroke but remains statistically significant for up to 5 to 8 years later,” Dr. Verhoeven said.
The cancers that were most involved in this risk increase were those of the lower respiratory tract, hematologic cancers, and gastrointestinal cancers.
The main strength of this study was the use of national databases that allowed for a very large sample size, but this brings with it the danger of misclassification of events and the lack of clinical data, Dr. Verhoeven noted.
“Young stroke patients are at increased risk of developing a new cancer in the years following their stroke compared to peers from the general population, but this risk is only marginally increased in the older stroke population,” she concluded.
She pointed out that it is not possible to confirm any causal relation from this study design, but a clear association has been shown.
“We need more studies into this field. We need a large clinical dataset to examine which clinical phenotypes are associated with possible underlying cancers to identify which patients are most at risk. We are already working on this,” she said. “Then it remains to be investigated whether screening for an underlying cancer should be added to the diagnostic workup in young stroke patients.”
Commenting on the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh, Scotland, and a consultant neurologist in NHS Lothian, said it was difficult to know whether the link shown between stroke and cancer was causal, but the effect size in this study was “quite large.”
He pointed out that the associations with bowel and lung cancer could be due to shared risk factors, such as smoking, but he said the finding on a link with hematologic cancers is “interesting.”
Noting that there are links between hematologic cancers and thrombotic events, he said: “People have wondered if that is because of clonal expansion, which has been shown to increase the risk of atherosclerosis, so the question is whether this is some kind of common risk factor here.”
Dr. Verhoeven said she did not believe that shared risk factors fully explained the difference in increased risks between young and older patients.
“It does not fully explain why the risk of cancer is specifically higher in the first 1 to 2 years after the stroke diagnosis. I would think if it was just shared risk factors, the risk increase should remain relatively stable, or even increase due to the build-up of exposure to risk factors over the years,” she said.
Dr. Whiteley said that data like these are “really useful in trying to estimate these associations and it gives us some hypotheses to investigate in smaller mechanistic studies.”
Asked whether these data justify screening younger cryptogenic stroke patients more systematically for cancer, Dr. Whiteley replied: “I think we need some absolute risk estimates for that; for example, what proportion of younger patients would be at risk over the next few years when that screening would make a difference.”
Dr. Verhoeven reports no disclosures.
A version of this article first appeared on Medscape.com.
The data were presented by Jamie Verhoeven, MD, Radboud University Medical Centre, the Netherlands, at the recent European Stroke Organisation Conference 2022.
Dr. Verhoeven noted that 10% of all stroke cases occur in individuals younger than 50 years. During the past few decades, the incidence of stroke in the young has steadily increased, whereas the incidence of stroke in older adults has stabilized or decreased.
“Stroke in the young differs from stroke in older patients, and one of the major differences is that stroke in the young has a higher proportion of cryptogenic stroke, with no clear cause found in over one-third of patients,” she said.
Also, having an active cancer is known to be a risk factor for thrombosis. This association is strongest in venous thrombosis and has been less well investigated in arterial thrombosis, Dr. Verhoeven reported.
Her group aimed to investigate whether in some patients with cryptogenic stroke, this may be the first manifestation of an underlying cancer. “If this hypothesis is true, then it would be more obvious in young patients who have a higher incidence of cryptogenic stroke,” she said.
They performed a population-based observational cohort study using diagnostic ICD codes from the national Hospital Discharge Registry in the Netherlands and the Dutch Population Registry from 1998 to 2019.
Patients with a history of cancer before their first stroke and those with central nervous system cancers at the time of stroke or nonmelanoma skin cancers (which have been shown to have no systemic effects) were excluded.
Reference data came from the Netherlands Comprehensive Cancer Organisation, which collects data on all cancer diagnoses in the country.
The researchers identified 27,616 young stroke patients (age range, 15-49 years; median age, 45 years) and 362,782 older stroke patients (age range, 50 years and older; median age, 76 years).
The cumulative incidence of any cancer at 10 years was 3.7% in the younger group and 8.5% in the older group.
The data were compared with matched peers from the general population. The main outcome measures were cumulative incidence of first-ever cancer after stroke (stratified by stroke subtype, age and sex) and standardized incidence rates.
Results showed that the risk for cancer was higher in the younger age group than in the matched general population.
In this age group, the 1-year risk of any new cancer was 2.6 times higher (95% confidence interval, 2.2-3.1) after ischemic stroke and 5.4 times (95% CI, 3.8-7.3) after intracerebral hemorrhage than in matched peers from the general population.
In contrast, in stroke patients older than 50 years, the 1-year risk for any new cancer was 1.2 times higher than the general population after either ischemic or hemorrhagic stroke.
“The younger patients have a higher risk increase of cancer than older patients, and this risk increase is most evident in the first 1 to 2 years after stroke but remains statistically significant for up to 5 to 8 years later,” Dr. Verhoeven said.
The cancers that were most involved in this risk increase were those of the lower respiratory tract, hematologic cancers, and gastrointestinal cancers.
The main strength of this study was the use of national databases that allowed for a very large sample size, but this brings with it the danger of misclassification of events and the lack of clinical data, Dr. Verhoeven noted.
“Young stroke patients are at increased risk of developing a new cancer in the years following their stroke compared to peers from the general population, but this risk is only marginally increased in the older stroke population,” she concluded.
She pointed out that it is not possible to confirm any causal relation from this study design, but a clear association has been shown.
“We need more studies into this field. We need a large clinical dataset to examine which clinical phenotypes are associated with possible underlying cancers to identify which patients are most at risk. We are already working on this,” she said. “Then it remains to be investigated whether screening for an underlying cancer should be added to the diagnostic workup in young stroke patients.”
Commenting on the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh, Scotland, and a consultant neurologist in NHS Lothian, said it was difficult to know whether the link shown between stroke and cancer was causal, but the effect size in this study was “quite large.”
He pointed out that the associations with bowel and lung cancer could be due to shared risk factors, such as smoking, but he said the finding on a link with hematologic cancers is “interesting.”
Noting that there are links between hematologic cancers and thrombotic events, he said: “People have wondered if that is because of clonal expansion, which has been shown to increase the risk of atherosclerosis, so the question is whether this is some kind of common risk factor here.”
Dr. Verhoeven said she did not believe that shared risk factors fully explained the difference in increased risks between young and older patients.
“It does not fully explain why the risk of cancer is specifically higher in the first 1 to 2 years after the stroke diagnosis. I would think if it was just shared risk factors, the risk increase should remain relatively stable, or even increase due to the build-up of exposure to risk factors over the years,” she said.
Dr. Whiteley said that data like these are “really useful in trying to estimate these associations and it gives us some hypotheses to investigate in smaller mechanistic studies.”
Asked whether these data justify screening younger cryptogenic stroke patients more systematically for cancer, Dr. Whiteley replied: “I think we need some absolute risk estimates for that; for example, what proportion of younger patients would be at risk over the next few years when that screening would make a difference.”
Dr. Verhoeven reports no disclosures.
A version of this article first appeared on Medscape.com.
The data were presented by Jamie Verhoeven, MD, Radboud University Medical Centre, the Netherlands, at the recent European Stroke Organisation Conference 2022.
Dr. Verhoeven noted that 10% of all stroke cases occur in individuals younger than 50 years. During the past few decades, the incidence of stroke in the young has steadily increased, whereas the incidence of stroke in older adults has stabilized or decreased.
“Stroke in the young differs from stroke in older patients, and one of the major differences is that stroke in the young has a higher proportion of cryptogenic stroke, with no clear cause found in over one-third of patients,” she said.
Also, having an active cancer is known to be a risk factor for thrombosis. This association is strongest in venous thrombosis and has been less well investigated in arterial thrombosis, Dr. Verhoeven reported.
Her group aimed to investigate whether in some patients with cryptogenic stroke, this may be the first manifestation of an underlying cancer. “If this hypothesis is true, then it would be more obvious in young patients who have a higher incidence of cryptogenic stroke,” she said.
They performed a population-based observational cohort study using diagnostic ICD codes from the national Hospital Discharge Registry in the Netherlands and the Dutch Population Registry from 1998 to 2019.
Patients with a history of cancer before their first stroke and those with central nervous system cancers at the time of stroke or nonmelanoma skin cancers (which have been shown to have no systemic effects) were excluded.
Reference data came from the Netherlands Comprehensive Cancer Organisation, which collects data on all cancer diagnoses in the country.
The researchers identified 27,616 young stroke patients (age range, 15-49 years; median age, 45 years) and 362,782 older stroke patients (age range, 50 years and older; median age, 76 years).
The cumulative incidence of any cancer at 10 years was 3.7% in the younger group and 8.5% in the older group.
The data were compared with matched peers from the general population. The main outcome measures were cumulative incidence of first-ever cancer after stroke (stratified by stroke subtype, age and sex) and standardized incidence rates.
Results showed that the risk for cancer was higher in the younger age group than in the matched general population.
In this age group, the 1-year risk of any new cancer was 2.6 times higher (95% confidence interval, 2.2-3.1) after ischemic stroke and 5.4 times (95% CI, 3.8-7.3) after intracerebral hemorrhage than in matched peers from the general population.
In contrast, in stroke patients older than 50 years, the 1-year risk for any new cancer was 1.2 times higher than the general population after either ischemic or hemorrhagic stroke.
“The younger patients have a higher risk increase of cancer than older patients, and this risk increase is most evident in the first 1 to 2 years after stroke but remains statistically significant for up to 5 to 8 years later,” Dr. Verhoeven said.
The cancers that were most involved in this risk increase were those of the lower respiratory tract, hematologic cancers, and gastrointestinal cancers.
The main strength of this study was the use of national databases that allowed for a very large sample size, but this brings with it the danger of misclassification of events and the lack of clinical data, Dr. Verhoeven noted.
“Young stroke patients are at increased risk of developing a new cancer in the years following their stroke compared to peers from the general population, but this risk is only marginally increased in the older stroke population,” she concluded.
She pointed out that it is not possible to confirm any causal relation from this study design, but a clear association has been shown.
“We need more studies into this field. We need a large clinical dataset to examine which clinical phenotypes are associated with possible underlying cancers to identify which patients are most at risk. We are already working on this,” she said. “Then it remains to be investigated whether screening for an underlying cancer should be added to the diagnostic workup in young stroke patients.”
Commenting on the study after the presentation, William Whiteley, BM, PhD, a clinical epidemiologist at the University of Edinburgh, Scotland, and a consultant neurologist in NHS Lothian, said it was difficult to know whether the link shown between stroke and cancer was causal, but the effect size in this study was “quite large.”
He pointed out that the associations with bowel and lung cancer could be due to shared risk factors, such as smoking, but he said the finding on a link with hematologic cancers is “interesting.”
Noting that there are links between hematologic cancers and thrombotic events, he said: “People have wondered if that is because of clonal expansion, which has been shown to increase the risk of atherosclerosis, so the question is whether this is some kind of common risk factor here.”
Dr. Verhoeven said she did not believe that shared risk factors fully explained the difference in increased risks between young and older patients.
“It does not fully explain why the risk of cancer is specifically higher in the first 1 to 2 years after the stroke diagnosis. I would think if it was just shared risk factors, the risk increase should remain relatively stable, or even increase due to the build-up of exposure to risk factors over the years,” she said.
Dr. Whiteley said that data like these are “really useful in trying to estimate these associations and it gives us some hypotheses to investigate in smaller mechanistic studies.”
Asked whether these data justify screening younger cryptogenic stroke patients more systematically for cancer, Dr. Whiteley replied: “I think we need some absolute risk estimates for that; for example, what proportion of younger patients would be at risk over the next few years when that screening would make a difference.”
Dr. Verhoeven reports no disclosures.
A version of this article first appeared on Medscape.com.
FROM ESOC 2022
IBD after age 60: More evidence antibiotics play a role
Most studies to date have assessed a link between antibiotics and IBD in younger patients, lead researcher Adam S. Faye, MD, said during a media briefing that previewed select research for the annual Digestive Disease Week® (DDW).
The impact of antibiotic use on the incidence of IBD in older adults is really unknown, he added.
In contrast to younger people with IBD, who tend to have a strong family history or genetic predisposition to developing Crohn’s disease or ulcerative colitis, the cause is likely different in older populations.
“There’s clearly something in the environment that’s driving this new older-onset IBD,” said Dr. Faye, who is an assistant professor of medicine and population health at New York University.
Antibiotics as a contributing link
Dr. Faye and colleagues took a closer look at antibiotics as contributing to this link. They studied 2.3 million patient records in Denmark’s national medical registry from 2000 to 2018. They identified people aged 60 years and older who were newly diagnosed with IBD, and they then assessed the number, frequency, and timing of any antibiotic prescriptions.
They found that IBD was 27% more likely in this age group if the patients had received any antibiotic prescription.
They also found that the chance of developing IBD was higher as the number of antibiotic prescriptions increased. For example, IBD was 55% more likely if a person had received two prescriptions, and it was 96% more likely with four prescriptions. The risk really jumped with five or more antibiotic prescriptions – a person with this many prescriptions was more than 2.3 times (236%) more likely to be diagnosed with IBD than those who had not been prescribed antibiotics in the prior 5 years.
Not all antibiotics were equal, however. For example, the investigators found no link with nitrofurantoin, an antibiotic commonly prescribed for urinary tract infections. In contrast, all other antibiotic agents that were evaluated, and especially fluoroquinolones, nitroimidazoles, and macrolides, were associated with IBD.
Timing made some difference.
“The risk was highest if antibiotics were prescribed within the 1- to 2-year period before diagnosis, and it declined as you go farther out. But the risks persist,” Dr. Faye said. He noted that they even found that risk was elevated 10 years out.
The investigators also considered whether the antibiotic agent or infection was behind the association.
Dr. Faye cited previous research, again in younger people with IBD, that revealed that “infections plus antibiotics substantially increase the odds or risk of developing IBD more than the infection alone.
“So there really does seem to be something that the antibiotics are doing here,” Dr. Faye said.
A leading theory is that antibiotics disrupt the gut microbiota and increase the risk for developing IBD. “But, obviously, it’s quite complicated,” he said.
Clinical implications
The findings suggest that older people who may have IBD should be screened for prior antibiotic use, Dr. Faye said.
“This is a result that really has important implications for diagnosing older adults with new gastrointestinal symptoms,” he said. “Inflammatory bowel disease often can be overlooked in older adults because there’s a lot of different diagnoses you’re thinking of.”
IBD “should be considered, especially if you have a patient who’s reporting multiple courses of antibiotics in the last few years,” he added.
The results suggest another reason that antimicrobial stewardship programs should promote judicial use of these agents beyond concerns about resistance.
“We think of antibiotic stewardship to prevent the development of multidrug-resistant organisms, but we should be thinking about it to also prevent the development of inflammatory bowel disease,” Dr. Faye said.
Although this study adds to the evidence implicating antibiotics and expands the concept to an older population, “we really don’t have a great handle on what all of the environmental and other factors are,” he said.
Some researchers point to smoking and diet, among other factors, but the interplay remains unknown, Dr. Faye added.
The study is important because the incidence of IBD is increasing within the older population, “and this is one of the first studies to look at it,” he said.
Dr. Faye and colleagues plan to start a new study to evaluate other environmental factors.
“Hopefully, we’ll have more within the next few years to report,” he said.
Shedding more light on older-onset IBD worldwide
“It’s a well-done study,” Aline Charabaty, MD, said in a comment. “We are seeing that there’s an increase of incidence of IBD in the entire population, but even more so in the elderly.”
IBD is likely caused by a combination of factors, including genetics, environmental influences, and dysfunction of the gut immune system, agreed Dr. Charabaty, who is an assistant clinical professor in the division of gastroenterology at Johns Hopkins University, Baltimore.
The research “goes along with other studies that we’ve done in the pediatric and adult populations that show antibiotics exposure increases the risk of developing inflammatory bowel disease,” she said.
For a broader perspective of the study’s findings, Dr. Faye was asked during the media briefing if the results of this Danish registry study would be generalizable to the U.S. population.
“The simplest answer is we’ll need to redo this study within the U.S. to make absolutely sure,” Dr. Faye said. She noted that prior studies in the United States and elsewhere have found a risk associated with antibiotics, although again these studies focused on younger patients.
Dr. Charabaty was more certain that the findings were meaningful outside of Denmark.
“I definitely think this will apply to our U.S. population,” added Dr. Charabaty, who is also the clinical director of the IBD Center at Johns Hopkins–Sibley Memorial Hospital, Washington. “We have very similar practices in terms of how we approach antibiotic use.
“This could be one of the risk factors that’s promoting an increase in IBD everywhere,” she added.
The study was conducted in partnership with the Danish National Center of Excellence PREDICT Program. Dr. Faye and Dr. Charabaty did not report any conflicts of interest related to this study.
A version of this article first appeared on Medscape.com.
Most studies to date have assessed a link between antibiotics and IBD in younger patients, lead researcher Adam S. Faye, MD, said during a media briefing that previewed select research for the annual Digestive Disease Week® (DDW).
The impact of antibiotic use on the incidence of IBD in older adults is really unknown, he added.
In contrast to younger people with IBD, who tend to have a strong family history or genetic predisposition to developing Crohn’s disease or ulcerative colitis, the cause is likely different in older populations.
“There’s clearly something in the environment that’s driving this new older-onset IBD,” said Dr. Faye, who is an assistant professor of medicine and population health at New York University.
Antibiotics as a contributing link
Dr. Faye and colleagues took a closer look at antibiotics as contributing to this link. They studied 2.3 million patient records in Denmark’s national medical registry from 2000 to 2018. They identified people aged 60 years and older who were newly diagnosed with IBD, and they then assessed the number, frequency, and timing of any antibiotic prescriptions.
They found that IBD was 27% more likely in this age group if the patients had received any antibiotic prescription.
They also found that the chance of developing IBD was higher as the number of antibiotic prescriptions increased. For example, IBD was 55% more likely if a person had received two prescriptions, and it was 96% more likely with four prescriptions. The risk really jumped with five or more antibiotic prescriptions – a person with this many prescriptions was more than 2.3 times (236%) more likely to be diagnosed with IBD than those who had not been prescribed antibiotics in the prior 5 years.
Not all antibiotics were equal, however. For example, the investigators found no link with nitrofurantoin, an antibiotic commonly prescribed for urinary tract infections. In contrast, all other antibiotic agents that were evaluated, and especially fluoroquinolones, nitroimidazoles, and macrolides, were associated with IBD.
Timing made some difference.
“The risk was highest if antibiotics were prescribed within the 1- to 2-year period before diagnosis, and it declined as you go farther out. But the risks persist,” Dr. Faye said. He noted that they even found that risk was elevated 10 years out.
The investigators also considered whether the antibiotic agent or infection was behind the association.
Dr. Faye cited previous research, again in younger people with IBD, that revealed that “infections plus antibiotics substantially increase the odds or risk of developing IBD more than the infection alone.
“So there really does seem to be something that the antibiotics are doing here,” Dr. Faye said.
A leading theory is that antibiotics disrupt the gut microbiota and increase the risk for developing IBD. “But, obviously, it’s quite complicated,” he said.
Clinical implications
The findings suggest that older people who may have IBD should be screened for prior antibiotic use, Dr. Faye said.
“This is a result that really has important implications for diagnosing older adults with new gastrointestinal symptoms,” he said. “Inflammatory bowel disease often can be overlooked in older adults because there’s a lot of different diagnoses you’re thinking of.”
IBD “should be considered, especially if you have a patient who’s reporting multiple courses of antibiotics in the last few years,” he added.
The results suggest another reason that antimicrobial stewardship programs should promote judicial use of these agents beyond concerns about resistance.
“We think of antibiotic stewardship to prevent the development of multidrug-resistant organisms, but we should be thinking about it to also prevent the development of inflammatory bowel disease,” Dr. Faye said.
Although this study adds to the evidence implicating antibiotics and expands the concept to an older population, “we really don’t have a great handle on what all of the environmental and other factors are,” he said.
Some researchers point to smoking and diet, among other factors, but the interplay remains unknown, Dr. Faye added.
The study is important because the incidence of IBD is increasing within the older population, “and this is one of the first studies to look at it,” he said.
Dr. Faye and colleagues plan to start a new study to evaluate other environmental factors.
“Hopefully, we’ll have more within the next few years to report,” he said.
Shedding more light on older-onset IBD worldwide
“It’s a well-done study,” Aline Charabaty, MD, said in a comment. “We are seeing that there’s an increase of incidence of IBD in the entire population, but even more so in the elderly.”
IBD is likely caused by a combination of factors, including genetics, environmental influences, and dysfunction of the gut immune system, agreed Dr. Charabaty, who is an assistant clinical professor in the division of gastroenterology at Johns Hopkins University, Baltimore.
The research “goes along with other studies that we’ve done in the pediatric and adult populations that show antibiotics exposure increases the risk of developing inflammatory bowel disease,” she said.
For a broader perspective of the study’s findings, Dr. Faye was asked during the media briefing if the results of this Danish registry study would be generalizable to the U.S. population.
“The simplest answer is we’ll need to redo this study within the U.S. to make absolutely sure,” Dr. Faye said. She noted that prior studies in the United States and elsewhere have found a risk associated with antibiotics, although again these studies focused on younger patients.
Dr. Charabaty was more certain that the findings were meaningful outside of Denmark.
“I definitely think this will apply to our U.S. population,” added Dr. Charabaty, who is also the clinical director of the IBD Center at Johns Hopkins–Sibley Memorial Hospital, Washington. “We have very similar practices in terms of how we approach antibiotic use.
“This could be one of the risk factors that’s promoting an increase in IBD everywhere,” she added.
The study was conducted in partnership with the Danish National Center of Excellence PREDICT Program. Dr. Faye and Dr. Charabaty did not report any conflicts of interest related to this study.
A version of this article first appeared on Medscape.com.
Most studies to date have assessed a link between antibiotics and IBD in younger patients, lead researcher Adam S. Faye, MD, said during a media briefing that previewed select research for the annual Digestive Disease Week® (DDW).
The impact of antibiotic use on the incidence of IBD in older adults is really unknown, he added.
In contrast to younger people with IBD, who tend to have a strong family history or genetic predisposition to developing Crohn’s disease or ulcerative colitis, the cause is likely different in older populations.
“There’s clearly something in the environment that’s driving this new older-onset IBD,” said Dr. Faye, who is an assistant professor of medicine and population health at New York University.
Antibiotics as a contributing link
Dr. Faye and colleagues took a closer look at antibiotics as contributing to this link. They studied 2.3 million patient records in Denmark’s national medical registry from 2000 to 2018. They identified people aged 60 years and older who were newly diagnosed with IBD, and they then assessed the number, frequency, and timing of any antibiotic prescriptions.
They found that IBD was 27% more likely in this age group if the patients had received any antibiotic prescription.
They also found that the chance of developing IBD was higher as the number of antibiotic prescriptions increased. For example, IBD was 55% more likely if a person had received two prescriptions, and it was 96% more likely with four prescriptions. The risk really jumped with five or more antibiotic prescriptions – a person with this many prescriptions was more than 2.3 times (236%) more likely to be diagnosed with IBD than those who had not been prescribed antibiotics in the prior 5 years.
Not all antibiotics were equal, however. For example, the investigators found no link with nitrofurantoin, an antibiotic commonly prescribed for urinary tract infections. In contrast, all other antibiotic agents that were evaluated, and especially fluoroquinolones, nitroimidazoles, and macrolides, were associated with IBD.
Timing made some difference.
“The risk was highest if antibiotics were prescribed within the 1- to 2-year period before diagnosis, and it declined as you go farther out. But the risks persist,” Dr. Faye said. He noted that they even found that risk was elevated 10 years out.
The investigators also considered whether the antibiotic agent or infection was behind the association.
Dr. Faye cited previous research, again in younger people with IBD, that revealed that “infections plus antibiotics substantially increase the odds or risk of developing IBD more than the infection alone.
“So there really does seem to be something that the antibiotics are doing here,” Dr. Faye said.
A leading theory is that antibiotics disrupt the gut microbiota and increase the risk for developing IBD. “But, obviously, it’s quite complicated,” he said.
Clinical implications
The findings suggest that older people who may have IBD should be screened for prior antibiotic use, Dr. Faye said.
“This is a result that really has important implications for diagnosing older adults with new gastrointestinal symptoms,” he said. “Inflammatory bowel disease often can be overlooked in older adults because there’s a lot of different diagnoses you’re thinking of.”
IBD “should be considered, especially if you have a patient who’s reporting multiple courses of antibiotics in the last few years,” he added.
The results suggest another reason that antimicrobial stewardship programs should promote judicial use of these agents beyond concerns about resistance.
“We think of antibiotic stewardship to prevent the development of multidrug-resistant organisms, but we should be thinking about it to also prevent the development of inflammatory bowel disease,” Dr. Faye said.
Although this study adds to the evidence implicating antibiotics and expands the concept to an older population, “we really don’t have a great handle on what all of the environmental and other factors are,” he said.
Some researchers point to smoking and diet, among other factors, but the interplay remains unknown, Dr. Faye added.
The study is important because the incidence of IBD is increasing within the older population, “and this is one of the first studies to look at it,” he said.
Dr. Faye and colleagues plan to start a new study to evaluate other environmental factors.
“Hopefully, we’ll have more within the next few years to report,” he said.
Shedding more light on older-onset IBD worldwide
“It’s a well-done study,” Aline Charabaty, MD, said in a comment. “We are seeing that there’s an increase of incidence of IBD in the entire population, but even more so in the elderly.”
IBD is likely caused by a combination of factors, including genetics, environmental influences, and dysfunction of the gut immune system, agreed Dr. Charabaty, who is an assistant clinical professor in the division of gastroenterology at Johns Hopkins University, Baltimore.
The research “goes along with other studies that we’ve done in the pediatric and adult populations that show antibiotics exposure increases the risk of developing inflammatory bowel disease,” she said.
For a broader perspective of the study’s findings, Dr. Faye was asked during the media briefing if the results of this Danish registry study would be generalizable to the U.S. population.
“The simplest answer is we’ll need to redo this study within the U.S. to make absolutely sure,” Dr. Faye said. She noted that prior studies in the United States and elsewhere have found a risk associated with antibiotics, although again these studies focused on younger patients.
Dr. Charabaty was more certain that the findings were meaningful outside of Denmark.
“I definitely think this will apply to our U.S. population,” added Dr. Charabaty, who is also the clinical director of the IBD Center at Johns Hopkins–Sibley Memorial Hospital, Washington. “We have very similar practices in terms of how we approach antibiotic use.
“This could be one of the risk factors that’s promoting an increase in IBD everywhere,” she added.
The study was conducted in partnership with the Danish National Center of Excellence PREDICT Program. Dr. Faye and Dr. Charabaty did not report any conflicts of interest related to this study.
A version of this article first appeared on Medscape.com.
FROM DDW 2022
How to make visits run more smoothly and be more productive
We all feel pressure from not having enough time to care for our patients the way we want to.
Organ recital
Some of our patients need to share an update on all their symptoms each visit, old and new, including those that are minor or possibly concerning. I have learned over the years that, for many patients, this allows them to release the worry about symptoms.
Some symptoms are so distressing and severe that symptomatic treatment is needed, but most aren’t.
I am very honest with patients when I have no idea what is causing their symptoms. I tell them, we will watch for other clues to see if the symptom needs a workup.
One thing I don’t do, and I strongly recommend against, is doing a review of systems. This leads a patient to believe you are concerned about exploring each possible symptom, ones that they didn’t even bring up! The yield is very low, and the time commitment is great.
The angry patient
Imagine a scenario when you are running 15 minutes behind and, when you step into the room, your patient is angry. You are already behind, and helping the patient navigate their anger will be part of your clinic visit.
In these situations, I always address the patient’s anger immediately. Problems with getting appointments with specialists, delays in diagnostic tests, or a broken entry to the parking garage have all been sources of my patients’ frustrations.
When we have limited time, using much of the clinic visit to process frustration leads to empty clinic visits. I listen and work to empathize with the patient, often agreeing that there are so many messed up aspects of the health care system. I do not like to use the corporate “I am sad you feel that way” response, because I feel it is not helpful. Instead, I tell them how much I want to help them today in any way I can at this visit.
The Internet sleuth
When our patients have new symptoms, some of them will go to the Internet to try to self-diagnose. Sometimes they make a correct diagnosis, but other times consider scary diagnoses we would not consider based on their symptoms and risk factors.
In these scenarios, I always ask the patient why they think their diagnosis is accurate. Their response to this question gives me insight into where their beliefs come from and helps me understand what information I need to provide.
McMullan said physicians can be defensive, collaborative, and informative when they interact with patients about information they have found on the Internet. In the first model, the physician is authoritative. The second involves working with the patient and obtaining and analyzing information. In the third model, the physician provides reputable internet sites to patients for obtaining information.
‘Oh, by the way’
Patients frequently bring up sensitive topics or complicated requests after the visit has wrapped up. Topics such as insomnia, erectile dysfunction, and anxiety are often brought up with the assumption that a quick prescription is the answer. For many years, I would add time to the appointment and try to address these issues as quickly as I could. But I invariably did a poor job at helping with these problems. Now, I offer to see the patient back soon to spend an entire visit discussing the newly brought up concern. I tell them that the problem is too important to not have my full attention and focus.
Pearls
- Empathetically listen to descriptions of symptoms, but don’t focus on fixing them.
- Empathize with the angry patient, and move on to taking care of their medical problems.
- Avoid the urge to address newly raised problems at the end of the visit.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose.
We all feel pressure from not having enough time to care for our patients the way we want to.
Organ recital
Some of our patients need to share an update on all their symptoms each visit, old and new, including those that are minor or possibly concerning. I have learned over the years that, for many patients, this allows them to release the worry about symptoms.
Some symptoms are so distressing and severe that symptomatic treatment is needed, but most aren’t.
I am very honest with patients when I have no idea what is causing their symptoms. I tell them, we will watch for other clues to see if the symptom needs a workup.
One thing I don’t do, and I strongly recommend against, is doing a review of systems. This leads a patient to believe you are concerned about exploring each possible symptom, ones that they didn’t even bring up! The yield is very low, and the time commitment is great.
The angry patient
Imagine a scenario when you are running 15 minutes behind and, when you step into the room, your patient is angry. You are already behind, and helping the patient navigate their anger will be part of your clinic visit.
In these situations, I always address the patient’s anger immediately. Problems with getting appointments with specialists, delays in diagnostic tests, or a broken entry to the parking garage have all been sources of my patients’ frustrations.
When we have limited time, using much of the clinic visit to process frustration leads to empty clinic visits. I listen and work to empathize with the patient, often agreeing that there are so many messed up aspects of the health care system. I do not like to use the corporate “I am sad you feel that way” response, because I feel it is not helpful. Instead, I tell them how much I want to help them today in any way I can at this visit.
The Internet sleuth
When our patients have new symptoms, some of them will go to the Internet to try to self-diagnose. Sometimes they make a correct diagnosis, but other times consider scary diagnoses we would not consider based on their symptoms and risk factors.
In these scenarios, I always ask the patient why they think their diagnosis is accurate. Their response to this question gives me insight into where their beliefs come from and helps me understand what information I need to provide.
McMullan said physicians can be defensive, collaborative, and informative when they interact with patients about information they have found on the Internet. In the first model, the physician is authoritative. The second involves working with the patient and obtaining and analyzing information. In the third model, the physician provides reputable internet sites to patients for obtaining information.
‘Oh, by the way’
Patients frequently bring up sensitive topics or complicated requests after the visit has wrapped up. Topics such as insomnia, erectile dysfunction, and anxiety are often brought up with the assumption that a quick prescription is the answer. For many years, I would add time to the appointment and try to address these issues as quickly as I could. But I invariably did a poor job at helping with these problems. Now, I offer to see the patient back soon to spend an entire visit discussing the newly brought up concern. I tell them that the problem is too important to not have my full attention and focus.
Pearls
- Empathetically listen to descriptions of symptoms, but don’t focus on fixing them.
- Empathize with the angry patient, and move on to taking care of their medical problems.
- Avoid the urge to address newly raised problems at the end of the visit.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose.
We all feel pressure from not having enough time to care for our patients the way we want to.
Organ recital
Some of our patients need to share an update on all their symptoms each visit, old and new, including those that are minor or possibly concerning. I have learned over the years that, for many patients, this allows them to release the worry about symptoms.
Some symptoms are so distressing and severe that symptomatic treatment is needed, but most aren’t.
I am very honest with patients when I have no idea what is causing their symptoms. I tell them, we will watch for other clues to see if the symptom needs a workup.
One thing I don’t do, and I strongly recommend against, is doing a review of systems. This leads a patient to believe you are concerned about exploring each possible symptom, ones that they didn’t even bring up! The yield is very low, and the time commitment is great.
The angry patient
Imagine a scenario when you are running 15 minutes behind and, when you step into the room, your patient is angry. You are already behind, and helping the patient navigate their anger will be part of your clinic visit.
In these situations, I always address the patient’s anger immediately. Problems with getting appointments with specialists, delays in diagnostic tests, or a broken entry to the parking garage have all been sources of my patients’ frustrations.
When we have limited time, using much of the clinic visit to process frustration leads to empty clinic visits. I listen and work to empathize with the patient, often agreeing that there are so many messed up aspects of the health care system. I do not like to use the corporate “I am sad you feel that way” response, because I feel it is not helpful. Instead, I tell them how much I want to help them today in any way I can at this visit.
The Internet sleuth
When our patients have new symptoms, some of them will go to the Internet to try to self-diagnose. Sometimes they make a correct diagnosis, but other times consider scary diagnoses we would not consider based on their symptoms and risk factors.
In these scenarios, I always ask the patient why they think their diagnosis is accurate. Their response to this question gives me insight into where their beliefs come from and helps me understand what information I need to provide.
McMullan said physicians can be defensive, collaborative, and informative when they interact with patients about information they have found on the Internet. In the first model, the physician is authoritative. The second involves working with the patient and obtaining and analyzing information. In the third model, the physician provides reputable internet sites to patients for obtaining information.
‘Oh, by the way’
Patients frequently bring up sensitive topics or complicated requests after the visit has wrapped up. Topics such as insomnia, erectile dysfunction, and anxiety are often brought up with the assumption that a quick prescription is the answer. For many years, I would add time to the appointment and try to address these issues as quickly as I could. But I invariably did a poor job at helping with these problems. Now, I offer to see the patient back soon to spend an entire visit discussing the newly brought up concern. I tell them that the problem is too important to not have my full attention and focus.
Pearls
- Empathetically listen to descriptions of symptoms, but don’t focus on fixing them.
- Empathize with the angry patient, and move on to taking care of their medical problems.
- Avoid the urge to address newly raised problems at the end of the visit.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose.
Lyme disease may cost U.S. nearly $970 million per year
The annual cost of Lyme disease in the United States could be nearly $970 million, according to new research. But not all cases of Lyme disease are equally costly. Costs for patients with disseminated Lyme disease were more than twice as high as those for patients with localized disease.
“These findings emphasize the importance of early and accurate diagnosis to reduce both illness and its associated personal and societal costs,” the study’s authors write. The analysis was published in Emerging Infectious Diseases. The authors are from the Centers for Disease Control and Prevention, the Yale School of Public Health, and the Departments of Health of Minnesota, Maryland, and New York State.
Lyme disease is the most reported vector-borne disease in the United States; an estimated 476,000 Americans are diagnosed with the infection every year. Though the highest incidences are in the Northeast and in Minnesota and Wisconsin, the CDC has recorded cases in all 50 states. Over the past 20 years, estimates of the total cost of treating Lyme disease have varied from $203 million to nearly $1.3 billion. A major limitation to these studies, however, is that they may not accurately identify patients with Lyme disease or capture many of the nonmedical costs associated with treatment, the authors write, such as costs associated with time taken off work and travel to appointments.
To better capture the comprehensive cost of Lyme disease in the United States, researchers conducted a prospective study from September 2014 to January 2016 and followed reported cases in four states with high rates of Lyme disease: Connecticut, Maryland, Minnesota, and New York. The team conducted monthly patient surveys and queried billing codes from participants’ clinicians to estimate both the personal expenses and societal costs – such as total costs to the health care system – of Lyme disease.
Participants were variously assigned to one of three categories: those with confirmed local disease (such as patients with erythema migrans); those with confirmed disseminated disease, with symptoms including arthritis, lymphocytic meningitis, and encephalomyelitis; and those with probable cases with reported symptoms.
The researchers surveyed 901 participants with clinician-diagnosed Lyme disease: 402 with localized disease, 238 with disseminated disease, and 261 with probable cases. Nearly all participants (94%) were White, 57% were men, and 71% had an annual household income above $60,000. About 28% of participants were younger than 18 years, 16% were aged 18-45 years, 36% were aged 46-65, and 19% were older than 65 years. For most participants (68%), complete medical cost data were available. Those data were used to calculate societal costs.
Costs per patient varied dramatically, from $0.46 to $30,628. On average, patients spent $1,252 during the study period on expenses related to Lyme disease treatment, including expenses associated with travel and time taken off work. The median cost to patients was notably smaller, at $244. Patients with disseminated disease had the highest median ($358) and mean ($1,692) costs, followed by participants with probable disease (median, $315; mean, $1,277). Participants with localized disease had the lowest median ($170) and mean ($1,070) costs.
Societal costs ranged from $54 to $122,766 per patient; the median was $690, and the mean was $2,032. Multiplying these numbers by the estimated cases diagnosed nationally each year, researchers determined that the total cost of Lyme disease in the United States is between $345 million and $968 million.
The study “pointed out the tremendous variability in costs” associated with Lyme disease treatment, Brian Fallon, MD, MPH, director of the Lyme and Tick-Borne Diseases Research Center at the Columbia University Irving Medical Center, New York, told this news organization. He was not involved with the research. Some patients continued having health expenses after the end of the study, which means their costs would be even higher, he noted. Though most patients fully recovered after 1 year, about 4% still reported symptoms, and 3% were still incurring costs.
And while the calculated total cost is “significant,” it is likely an underestimate, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Clinical Research Center, Baltimore, in an interview. Dr. Aucott is also unaffiliated with the work. The researchers included confirmed or probable Lyme disease cases, he said, but that does not capture patients who may have had Lyme disease but were initially misdiagnosed. “Those poor patients end up going to see many different doctors to try to figure out what their diagnosis is,” added Dr. Fallon. “If they do have Lyme, they may not get started on treatment until much later than those who had the classic manifestations [of the disease].” These patients are more likely to have relapsing symptoms, he said, and their cost of care would be much higher, as highlighted in the research findings.
“The take-home point is that the cost to the patient and society is much less if you make an early diagnosis,” Dr. Aucott said. “It highlights the need for rapid diagnosis and rapid identification,” added Dr. Fallon, “as well as the potential importance and value of better means of prevention.”
A version of this article first appeared on Medscape.com.
The annual cost of Lyme disease in the United States could be nearly $970 million, according to new research. But not all cases of Lyme disease are equally costly. Costs for patients with disseminated Lyme disease were more than twice as high as those for patients with localized disease.
“These findings emphasize the importance of early and accurate diagnosis to reduce both illness and its associated personal and societal costs,” the study’s authors write. The analysis was published in Emerging Infectious Diseases. The authors are from the Centers for Disease Control and Prevention, the Yale School of Public Health, and the Departments of Health of Minnesota, Maryland, and New York State.
Lyme disease is the most reported vector-borne disease in the United States; an estimated 476,000 Americans are diagnosed with the infection every year. Though the highest incidences are in the Northeast and in Minnesota and Wisconsin, the CDC has recorded cases in all 50 states. Over the past 20 years, estimates of the total cost of treating Lyme disease have varied from $203 million to nearly $1.3 billion. A major limitation to these studies, however, is that they may not accurately identify patients with Lyme disease or capture many of the nonmedical costs associated with treatment, the authors write, such as costs associated with time taken off work and travel to appointments.
To better capture the comprehensive cost of Lyme disease in the United States, researchers conducted a prospective study from September 2014 to January 2016 and followed reported cases in four states with high rates of Lyme disease: Connecticut, Maryland, Minnesota, and New York. The team conducted monthly patient surveys and queried billing codes from participants’ clinicians to estimate both the personal expenses and societal costs – such as total costs to the health care system – of Lyme disease.
Participants were variously assigned to one of three categories: those with confirmed local disease (such as patients with erythema migrans); those with confirmed disseminated disease, with symptoms including arthritis, lymphocytic meningitis, and encephalomyelitis; and those with probable cases with reported symptoms.
The researchers surveyed 901 participants with clinician-diagnosed Lyme disease: 402 with localized disease, 238 with disseminated disease, and 261 with probable cases. Nearly all participants (94%) were White, 57% were men, and 71% had an annual household income above $60,000. About 28% of participants were younger than 18 years, 16% were aged 18-45 years, 36% were aged 46-65, and 19% were older than 65 years. For most participants (68%), complete medical cost data were available. Those data were used to calculate societal costs.
Costs per patient varied dramatically, from $0.46 to $30,628. On average, patients spent $1,252 during the study period on expenses related to Lyme disease treatment, including expenses associated with travel and time taken off work. The median cost to patients was notably smaller, at $244. Patients with disseminated disease had the highest median ($358) and mean ($1,692) costs, followed by participants with probable disease (median, $315; mean, $1,277). Participants with localized disease had the lowest median ($170) and mean ($1,070) costs.
Societal costs ranged from $54 to $122,766 per patient; the median was $690, and the mean was $2,032. Multiplying these numbers by the estimated cases diagnosed nationally each year, researchers determined that the total cost of Lyme disease in the United States is between $345 million and $968 million.
The study “pointed out the tremendous variability in costs” associated with Lyme disease treatment, Brian Fallon, MD, MPH, director of the Lyme and Tick-Borne Diseases Research Center at the Columbia University Irving Medical Center, New York, told this news organization. He was not involved with the research. Some patients continued having health expenses after the end of the study, which means their costs would be even higher, he noted. Though most patients fully recovered after 1 year, about 4% still reported symptoms, and 3% were still incurring costs.
And while the calculated total cost is “significant,” it is likely an underestimate, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Clinical Research Center, Baltimore, in an interview. Dr. Aucott is also unaffiliated with the work. The researchers included confirmed or probable Lyme disease cases, he said, but that does not capture patients who may have had Lyme disease but were initially misdiagnosed. “Those poor patients end up going to see many different doctors to try to figure out what their diagnosis is,” added Dr. Fallon. “If they do have Lyme, they may not get started on treatment until much later than those who had the classic manifestations [of the disease].” These patients are more likely to have relapsing symptoms, he said, and their cost of care would be much higher, as highlighted in the research findings.
“The take-home point is that the cost to the patient and society is much less if you make an early diagnosis,” Dr. Aucott said. “It highlights the need for rapid diagnosis and rapid identification,” added Dr. Fallon, “as well as the potential importance and value of better means of prevention.”
A version of this article first appeared on Medscape.com.
The annual cost of Lyme disease in the United States could be nearly $970 million, according to new research. But not all cases of Lyme disease are equally costly. Costs for patients with disseminated Lyme disease were more than twice as high as those for patients with localized disease.
“These findings emphasize the importance of early and accurate diagnosis to reduce both illness and its associated personal and societal costs,” the study’s authors write. The analysis was published in Emerging Infectious Diseases. The authors are from the Centers for Disease Control and Prevention, the Yale School of Public Health, and the Departments of Health of Minnesota, Maryland, and New York State.
Lyme disease is the most reported vector-borne disease in the United States; an estimated 476,000 Americans are diagnosed with the infection every year. Though the highest incidences are in the Northeast and in Minnesota and Wisconsin, the CDC has recorded cases in all 50 states. Over the past 20 years, estimates of the total cost of treating Lyme disease have varied from $203 million to nearly $1.3 billion. A major limitation to these studies, however, is that they may not accurately identify patients with Lyme disease or capture many of the nonmedical costs associated with treatment, the authors write, such as costs associated with time taken off work and travel to appointments.
To better capture the comprehensive cost of Lyme disease in the United States, researchers conducted a prospective study from September 2014 to January 2016 and followed reported cases in four states with high rates of Lyme disease: Connecticut, Maryland, Minnesota, and New York. The team conducted monthly patient surveys and queried billing codes from participants’ clinicians to estimate both the personal expenses and societal costs – such as total costs to the health care system – of Lyme disease.
Participants were variously assigned to one of three categories: those with confirmed local disease (such as patients with erythema migrans); those with confirmed disseminated disease, with symptoms including arthritis, lymphocytic meningitis, and encephalomyelitis; and those with probable cases with reported symptoms.
The researchers surveyed 901 participants with clinician-diagnosed Lyme disease: 402 with localized disease, 238 with disseminated disease, and 261 with probable cases. Nearly all participants (94%) were White, 57% were men, and 71% had an annual household income above $60,000. About 28% of participants were younger than 18 years, 16% were aged 18-45 years, 36% were aged 46-65, and 19% were older than 65 years. For most participants (68%), complete medical cost data were available. Those data were used to calculate societal costs.
Costs per patient varied dramatically, from $0.46 to $30,628. On average, patients spent $1,252 during the study period on expenses related to Lyme disease treatment, including expenses associated with travel and time taken off work. The median cost to patients was notably smaller, at $244. Patients with disseminated disease had the highest median ($358) and mean ($1,692) costs, followed by participants with probable disease (median, $315; mean, $1,277). Participants with localized disease had the lowest median ($170) and mean ($1,070) costs.
Societal costs ranged from $54 to $122,766 per patient; the median was $690, and the mean was $2,032. Multiplying these numbers by the estimated cases diagnosed nationally each year, researchers determined that the total cost of Lyme disease in the United States is between $345 million and $968 million.
The study “pointed out the tremendous variability in costs” associated with Lyme disease treatment, Brian Fallon, MD, MPH, director of the Lyme and Tick-Borne Diseases Research Center at the Columbia University Irving Medical Center, New York, told this news organization. He was not involved with the research. Some patients continued having health expenses after the end of the study, which means their costs would be even higher, he noted. Though most patients fully recovered after 1 year, about 4% still reported symptoms, and 3% were still incurring costs.
And while the calculated total cost is “significant,” it is likely an underestimate, said John Aucott, MD, director of the Johns Hopkins Lyme Disease Clinical Research Center, Baltimore, in an interview. Dr. Aucott is also unaffiliated with the work. The researchers included confirmed or probable Lyme disease cases, he said, but that does not capture patients who may have had Lyme disease but were initially misdiagnosed. “Those poor patients end up going to see many different doctors to try to figure out what their diagnosis is,” added Dr. Fallon. “If they do have Lyme, they may not get started on treatment until much later than those who had the classic manifestations [of the disease].” These patients are more likely to have relapsing symptoms, he said, and their cost of care would be much higher, as highlighted in the research findings.
“The take-home point is that the cost to the patient and society is much less if you make an early diagnosis,” Dr. Aucott said. “It highlights the need for rapid diagnosis and rapid identification,” added Dr. Fallon, “as well as the potential importance and value of better means of prevention.”
A version of this article first appeared on Medscape.com.
FROM EMERGING INFECTIOUS DISEASES
First fatty liver guidelines for endocrinology, primary care
New clinical practice guidelines for the diagnosis and management of nonalcoholic fatty liver disease (NAFLD) are the first to be targeted specifically to primary care and endocrinology clinical settings.
They include 34 evidence-based clinical practice recommendations for screening, diagnosis, management, and referral, presented in a table and an algorithm flow chart as well as detailed text.
The new guidelines are by the American Association of Clinical Endocrinology and cosponsored by the American Association for the Study of Liver Diseases. They were presented at the annual scientific & clinical congress of the AACE and simultaneously published in Endocrine Practice.
These are “the first of this type for this field of medicine. The vast majority of patients with NAFLD are being seen in the primary care and endocrinology settings. Only when they get to the more advanced disease are they being referred to the liver specialists. So, we need to be the ones who are diagnosing and managing these patients because there just aren’t enough liver specialists to do that,” Scott Isaacs, MD, cochair of the writing panel for the guidelines, said in an interview.
80 million Americans have NAFLD, but very few are aware
The spectrum of NAFLD ranges from nonprogressive steatosis to the progressive conditions nonalcoholic steatohepatitis, fibrotic NASH, and end-stage NASH cirrhosis. And NASH, in turn, is a major cause of liver cancer. NAFLD is also strongly associated with insulin resistance, type 2 diabetes, atherogenesis, and myocardial dysfunction.
The global prevalence of NAFLD is about 25% and NASH, about 12%-14%. However, a recent study found that, among patients in endocrine and primary care clinics, more than 70% of patients with type 2 diabetes and more than 90% with type 2 diabetes who had a body mass index above 35 kg/m2 also had NAFLD, and more than 20% of those patients had significant liver fibrosis.
Problematically, very few people are aware they have either. “It’s so common. At least 80 million Americans have this but only about 6% know they have it. We talk about it a lot, but it’s not talked about enough,” said Dr. Isaacs, an endocrinologist who practices in Atlanta.
In fact, most cases of NAFLD are diagnosed incidentally when people undergo an ultrasound or a CT scan for another reason. And, in about 70% of cases the liver enzymes are normal, and those patients rarely undergo liver workups, Dr. Isaacs noted.
In an accompanying editorial, Suthat Liangpunsakul, MD, wrote: “In my perspective, as a hepatologist, this AACE guideline is very practical and easy to incorporate into routine practice in primary care and endocrinology settings. ... Early identification and risk stratification of patients with NAFLD, especially the degree of hepatic fibrosis, are required to reduce downstream health care costs and triage unwarranted specialty care referrals.”
And “an effective screening strategy may also identify those in primary care and endocrinology settings who may benefit from an appropriate referral to hepatologists before the development of portal hypertension complications, decompensated liver disease, and hepatocellular carcinoma,” added Dr. Liangpunsakul, professor of medicine in the division of gastroenterology and hepatology at Indiana University, Indianapolis.
Screening advised using new FIB-4 test
The guideline calls for screening all patients at high risk for NAFLD, including those with prediabetes, type 2 diabetes, obesity, and/or two or more cardiometabolic risk factors, or those with hepatic steatosis found on imaging, and/or persistently elevated plasma aminotransferase levels (that is, for more than 6 months).
The recommended screening test is the Fibrosis-4 (FIB-4) index, calculated using the patient’s age, AST level, platelet count, and ALT level: FIB-4 score = age (years) x AST (U/L)/PLT (109/L) x ALT ½ (U/L).
Recently approved by the Food and Drug Administration, the FIB-4 has been demonstrated to help identify liver disease in primary care settings.
“We really want to encourage clinicians to do the screening. The first step is the FIB-4 test. It’s a mathematical calculation using blood tests that we do anyway,” Dr. Isaacs said in an interview.
The FIB-4 stratifies patients as being low, intermediate, or high risk for liver fibrosis. Those at low risk can be managed in primary care or endocrinology settings with a focus on obesity management and cardiovascular disease prevention. “Those at low risk on FIB-4 still have a high cardiovascular disease risk. They still need to be managed,” Dr. Isaacs observed.
For those at intermediate risk, a second noninvasive test – either a liver stiffness measurement by elastography or an enhanced liver fibrosis test – is advised. If the patient is found to be at high risk or is still indeterminant after two noninvasive tests, referral to a liver specialist for further testing, including possible biopsy, is advised.
Those found to be at high risk with the FIB-4 should also be referred to hepatology. In both the intermediate- and high-risk groups, management should be multidisciplinary, including a hepatologist, endocrinologist, and other professionals to prevent both cardiovascular disease and progression to cirrhosis, the guidelines say.
“The diagnosis isn’t about diagnosing liver fat. It’s about diagnosing fibrosis, or the risk for clinically significant fibrosis. That’s really where the challenge lies,” Dr. Isaacs commented.
NAFLD treatment in endocrinology and primary care: CVD prevention
During the presentation at the AACE meeting, guideline panel cochair Kenneth Cusi, MD, chief of endocrinology, diabetes, and metabolism at the University of Florida, Gainesville, summarized current and future treatments for NAFLD.
Lifestyle intervention, cardiovascular risk reduction, and weight loss for those who are overweight or obese are recommended for all patients with NAFLD, including structured weight-loss programs, antiobesity medications, and bariatric surgery if indicated.
There are currently no FDA-approved medications specifically for NASH, but pioglitazone, approved for type 2 diabetes, and glucagonlike peptide–1 agonists, approved for type 2 diabetes and weight loss, have been shown to be effective in treating the condition and preventing progression. Other treatments are in development, Dr. Cusi said.
The guideline also includes a section on diagnosis and management of NAFLD in children and adolescents. Here, the FIB-4 is not recommended because it isn’t accurate due to the age part of the equation, so liver enzyme tests are used in pediatric patients considered at high risk because of clinical factors. Management is similar to adults, except not all medications used in adults are approved for use in children.
In the editorial, Dr. Liangpunsakul cautioned that “the level of uptake and usage of the guideline may be an obstacle.”
To remedy that, he advised that “the next effort should gear toward distributing this guideline to the targeted providers and developing the ‘feedback platforms’ on its execution in the real-world. ... The successful implementation of this AACE guideline by the primary care providers and endocrinologists, hopefully, will deescalate the future burden of NAFLD-related morbidity and mortality.”
Dr. Isaacs and Dr. Liangpunsakul have reported no relevant financial relationships. Dr. Cusi has reported receiving research support towards the University of Florida as principal investigator from the National Institute of Health, Echosens, Inventiva, Nordic Bioscience, Novo Nordisk, Poxel, Labcorp, and Zydus, and is a consultant for Altimmune, Akero, Arrowhead, AstraZeneca, 89Bio, Bristol-Myers Squibb, Coherus, Intercept, Lilly, Madrigal, Merck, Novo Nordisk, Quest, Sagimet, Sonic Incytes, Terns, and Thera Technologies.
A version of this article first appeared on Medscape.com.
New clinical practice guidelines for the diagnosis and management of nonalcoholic fatty liver disease (NAFLD) are the first to be targeted specifically to primary care and endocrinology clinical settings.
They include 34 evidence-based clinical practice recommendations for screening, diagnosis, management, and referral, presented in a table and an algorithm flow chart as well as detailed text.
The new guidelines are by the American Association of Clinical Endocrinology and cosponsored by the American Association for the Study of Liver Diseases. They were presented at the annual scientific & clinical congress of the AACE and simultaneously published in Endocrine Practice.
These are “the first of this type for this field of medicine. The vast majority of patients with NAFLD are being seen in the primary care and endocrinology settings. Only when they get to the more advanced disease are they being referred to the liver specialists. So, we need to be the ones who are diagnosing and managing these patients because there just aren’t enough liver specialists to do that,” Scott Isaacs, MD, cochair of the writing panel for the guidelines, said in an interview.
80 million Americans have NAFLD, but very few are aware
The spectrum of NAFLD ranges from nonprogressive steatosis to the progressive conditions nonalcoholic steatohepatitis, fibrotic NASH, and end-stage NASH cirrhosis. And NASH, in turn, is a major cause of liver cancer. NAFLD is also strongly associated with insulin resistance, type 2 diabetes, atherogenesis, and myocardial dysfunction.
The global prevalence of NAFLD is about 25% and NASH, about 12%-14%. However, a recent study found that, among patients in endocrine and primary care clinics, more than 70% of patients with type 2 diabetes and more than 90% with type 2 diabetes who had a body mass index above 35 kg/m2 also had NAFLD, and more than 20% of those patients had significant liver fibrosis.
Problematically, very few people are aware they have either. “It’s so common. At least 80 million Americans have this but only about 6% know they have it. We talk about it a lot, but it’s not talked about enough,” said Dr. Isaacs, an endocrinologist who practices in Atlanta.
In fact, most cases of NAFLD are diagnosed incidentally when people undergo an ultrasound or a CT scan for another reason. And, in about 70% of cases the liver enzymes are normal, and those patients rarely undergo liver workups, Dr. Isaacs noted.
In an accompanying editorial, Suthat Liangpunsakul, MD, wrote: “In my perspective, as a hepatologist, this AACE guideline is very practical and easy to incorporate into routine practice in primary care and endocrinology settings. ... Early identification and risk stratification of patients with NAFLD, especially the degree of hepatic fibrosis, are required to reduce downstream health care costs and triage unwarranted specialty care referrals.”
And “an effective screening strategy may also identify those in primary care and endocrinology settings who may benefit from an appropriate referral to hepatologists before the development of portal hypertension complications, decompensated liver disease, and hepatocellular carcinoma,” added Dr. Liangpunsakul, professor of medicine in the division of gastroenterology and hepatology at Indiana University, Indianapolis.
Screening advised using new FIB-4 test
The guideline calls for screening all patients at high risk for NAFLD, including those with prediabetes, type 2 diabetes, obesity, and/or two or more cardiometabolic risk factors, or those with hepatic steatosis found on imaging, and/or persistently elevated plasma aminotransferase levels (that is, for more than 6 months).
The recommended screening test is the Fibrosis-4 (FIB-4) index, calculated using the patient’s age, AST level, platelet count, and ALT level: FIB-4 score = age (years) x AST (U/L)/PLT (109/L) x ALT ½ (U/L).
Recently approved by the Food and Drug Administration, the FIB-4 has been demonstrated to help identify liver disease in primary care settings.
“We really want to encourage clinicians to do the screening. The first step is the FIB-4 test. It’s a mathematical calculation using blood tests that we do anyway,” Dr. Isaacs said in an interview.
The FIB-4 stratifies patients as being low, intermediate, or high risk for liver fibrosis. Those at low risk can be managed in primary care or endocrinology settings with a focus on obesity management and cardiovascular disease prevention. “Those at low risk on FIB-4 still have a high cardiovascular disease risk. They still need to be managed,” Dr. Isaacs observed.
For those at intermediate risk, a second noninvasive test – either a liver stiffness measurement by elastography or an enhanced liver fibrosis test – is advised. If the patient is found to be at high risk or is still indeterminant after two noninvasive tests, referral to a liver specialist for further testing, including possible biopsy, is advised.
Those found to be at high risk with the FIB-4 should also be referred to hepatology. In both the intermediate- and high-risk groups, management should be multidisciplinary, including a hepatologist, endocrinologist, and other professionals to prevent both cardiovascular disease and progression to cirrhosis, the guidelines say.
“The diagnosis isn’t about diagnosing liver fat. It’s about diagnosing fibrosis, or the risk for clinically significant fibrosis. That’s really where the challenge lies,” Dr. Isaacs commented.
NAFLD treatment in endocrinology and primary care: CVD prevention
During the presentation at the AACE meeting, guideline panel cochair Kenneth Cusi, MD, chief of endocrinology, diabetes, and metabolism at the University of Florida, Gainesville, summarized current and future treatments for NAFLD.
Lifestyle intervention, cardiovascular risk reduction, and weight loss for those who are overweight or obese are recommended for all patients with NAFLD, including structured weight-loss programs, antiobesity medications, and bariatric surgery if indicated.
There are currently no FDA-approved medications specifically for NASH, but pioglitazone, approved for type 2 diabetes, and glucagonlike peptide–1 agonists, approved for type 2 diabetes and weight loss, have been shown to be effective in treating the condition and preventing progression. Other treatments are in development, Dr. Cusi said.
The guideline also includes a section on diagnosis and management of NAFLD in children and adolescents. Here, the FIB-4 is not recommended because it isn’t accurate due to the age part of the equation, so liver enzyme tests are used in pediatric patients considered at high risk because of clinical factors. Management is similar to adults, except not all medications used in adults are approved for use in children.
In the editorial, Dr. Liangpunsakul cautioned that “the level of uptake and usage of the guideline may be an obstacle.”
To remedy that, he advised that “the next effort should gear toward distributing this guideline to the targeted providers and developing the ‘feedback platforms’ on its execution in the real-world. ... The successful implementation of this AACE guideline by the primary care providers and endocrinologists, hopefully, will deescalate the future burden of NAFLD-related morbidity and mortality.”
Dr. Isaacs and Dr. Liangpunsakul have reported no relevant financial relationships. Dr. Cusi has reported receiving research support towards the University of Florida as principal investigator from the National Institute of Health, Echosens, Inventiva, Nordic Bioscience, Novo Nordisk, Poxel, Labcorp, and Zydus, and is a consultant for Altimmune, Akero, Arrowhead, AstraZeneca, 89Bio, Bristol-Myers Squibb, Coherus, Intercept, Lilly, Madrigal, Merck, Novo Nordisk, Quest, Sagimet, Sonic Incytes, Terns, and Thera Technologies.
A version of this article first appeared on Medscape.com.
New clinical practice guidelines for the diagnosis and management of nonalcoholic fatty liver disease (NAFLD) are the first to be targeted specifically to primary care and endocrinology clinical settings.
They include 34 evidence-based clinical practice recommendations for screening, diagnosis, management, and referral, presented in a table and an algorithm flow chart as well as detailed text.
The new guidelines are by the American Association of Clinical Endocrinology and cosponsored by the American Association for the Study of Liver Diseases. They were presented at the annual scientific & clinical congress of the AACE and simultaneously published in Endocrine Practice.
These are “the first of this type for this field of medicine. The vast majority of patients with NAFLD are being seen in the primary care and endocrinology settings. Only when they get to the more advanced disease are they being referred to the liver specialists. So, we need to be the ones who are diagnosing and managing these patients because there just aren’t enough liver specialists to do that,” Scott Isaacs, MD, cochair of the writing panel for the guidelines, said in an interview.
80 million Americans have NAFLD, but very few are aware
The spectrum of NAFLD ranges from nonprogressive steatosis to the progressive conditions nonalcoholic steatohepatitis, fibrotic NASH, and end-stage NASH cirrhosis. And NASH, in turn, is a major cause of liver cancer. NAFLD is also strongly associated with insulin resistance, type 2 diabetes, atherogenesis, and myocardial dysfunction.
The global prevalence of NAFLD is about 25% and NASH, about 12%-14%. However, a recent study found that, among patients in endocrine and primary care clinics, more than 70% of patients with type 2 diabetes and more than 90% with type 2 diabetes who had a body mass index above 35 kg/m2 also had NAFLD, and more than 20% of those patients had significant liver fibrosis.
Problematically, very few people are aware they have either. “It’s so common. At least 80 million Americans have this but only about 6% know they have it. We talk about it a lot, but it’s not talked about enough,” said Dr. Isaacs, an endocrinologist who practices in Atlanta.
In fact, most cases of NAFLD are diagnosed incidentally when people undergo an ultrasound or a CT scan for another reason. And, in about 70% of cases the liver enzymes are normal, and those patients rarely undergo liver workups, Dr. Isaacs noted.
In an accompanying editorial, Suthat Liangpunsakul, MD, wrote: “In my perspective, as a hepatologist, this AACE guideline is very practical and easy to incorporate into routine practice in primary care and endocrinology settings. ... Early identification and risk stratification of patients with NAFLD, especially the degree of hepatic fibrosis, are required to reduce downstream health care costs and triage unwarranted specialty care referrals.”
And “an effective screening strategy may also identify those in primary care and endocrinology settings who may benefit from an appropriate referral to hepatologists before the development of portal hypertension complications, decompensated liver disease, and hepatocellular carcinoma,” added Dr. Liangpunsakul, professor of medicine in the division of gastroenterology and hepatology at Indiana University, Indianapolis.
Screening advised using new FIB-4 test
The guideline calls for screening all patients at high risk for NAFLD, including those with prediabetes, type 2 diabetes, obesity, and/or two or more cardiometabolic risk factors, or those with hepatic steatosis found on imaging, and/or persistently elevated plasma aminotransferase levels (that is, for more than 6 months).
The recommended screening test is the Fibrosis-4 (FIB-4) index, calculated using the patient’s age, AST level, platelet count, and ALT level: FIB-4 score = age (years) x AST (U/L)/PLT (109/L) x ALT ½ (U/L).
Recently approved by the Food and Drug Administration, the FIB-4 has been demonstrated to help identify liver disease in primary care settings.
“We really want to encourage clinicians to do the screening. The first step is the FIB-4 test. It’s a mathematical calculation using blood tests that we do anyway,” Dr. Isaacs said in an interview.
The FIB-4 stratifies patients as being low, intermediate, or high risk for liver fibrosis. Those at low risk can be managed in primary care or endocrinology settings with a focus on obesity management and cardiovascular disease prevention. “Those at low risk on FIB-4 still have a high cardiovascular disease risk. They still need to be managed,” Dr. Isaacs observed.
For those at intermediate risk, a second noninvasive test – either a liver stiffness measurement by elastography or an enhanced liver fibrosis test – is advised. If the patient is found to be at high risk or is still indeterminant after two noninvasive tests, referral to a liver specialist for further testing, including possible biopsy, is advised.
Those found to be at high risk with the FIB-4 should also be referred to hepatology. In both the intermediate- and high-risk groups, management should be multidisciplinary, including a hepatologist, endocrinologist, and other professionals to prevent both cardiovascular disease and progression to cirrhosis, the guidelines say.
“The diagnosis isn’t about diagnosing liver fat. It’s about diagnosing fibrosis, or the risk for clinically significant fibrosis. That’s really where the challenge lies,” Dr. Isaacs commented.
NAFLD treatment in endocrinology and primary care: CVD prevention
During the presentation at the AACE meeting, guideline panel cochair Kenneth Cusi, MD, chief of endocrinology, diabetes, and metabolism at the University of Florida, Gainesville, summarized current and future treatments for NAFLD.
Lifestyle intervention, cardiovascular risk reduction, and weight loss for those who are overweight or obese are recommended for all patients with NAFLD, including structured weight-loss programs, antiobesity medications, and bariatric surgery if indicated.
There are currently no FDA-approved medications specifically for NASH, but pioglitazone, approved for type 2 diabetes, and glucagonlike peptide–1 agonists, approved for type 2 diabetes and weight loss, have been shown to be effective in treating the condition and preventing progression. Other treatments are in development, Dr. Cusi said.
The guideline also includes a section on diagnosis and management of NAFLD in children and adolescents. Here, the FIB-4 is not recommended because it isn’t accurate due to the age part of the equation, so liver enzyme tests are used in pediatric patients considered at high risk because of clinical factors. Management is similar to adults, except not all medications used in adults are approved for use in children.
In the editorial, Dr. Liangpunsakul cautioned that “the level of uptake and usage of the guideline may be an obstacle.”
To remedy that, he advised that “the next effort should gear toward distributing this guideline to the targeted providers and developing the ‘feedback platforms’ on its execution in the real-world. ... The successful implementation of this AACE guideline by the primary care providers and endocrinologists, hopefully, will deescalate the future burden of NAFLD-related morbidity and mortality.”
Dr. Isaacs and Dr. Liangpunsakul have reported no relevant financial relationships. Dr. Cusi has reported receiving research support towards the University of Florida as principal investigator from the National Institute of Health, Echosens, Inventiva, Nordic Bioscience, Novo Nordisk, Poxel, Labcorp, and Zydus, and is a consultant for Altimmune, Akero, Arrowhead, AstraZeneca, 89Bio, Bristol-Myers Squibb, Coherus, Intercept, Lilly, Madrigal, Merck, Novo Nordisk, Quest, Sagimet, Sonic Incytes, Terns, and Thera Technologies.
A version of this article first appeared on Medscape.com.
FROM AACE 2022
Can fecal transplants help reverse aging?
Transplanting fecal microbiota from young mice into older mice can reverse signs of aging in the gut, brain, and eyes, a team of scientists from the United Kingdom has found. Conversely, transplanting microbiota from old mice to young mice has the opposite effect.
This research provides “tantalizing evidence for the direct involvement of gut microbes in aging and the functional decline of brain function and vision and offers a potential solution in the form of gut microbe replacement therapy,” Simon Carding, PhD, who heads the gut microbes and health research program at the Quadram Institute in Norwich, England, said in a news release.
The study was published online in the journal Microbiome.
The fountain of youth?
Age-related changes in diversity, composition, and function of the gut microbiota are associated with low-grade systemic inflammation, declining tissue function, and increased susceptibility to age-related chronic diseases.
Dr. Carding and colleagues at the Quadram Institute and the University of East Anglia used fecal microbiota transplant (FMT) to exchange the intestinal microbiota of young mice and aged mice.
Young mice who received aged microbiota showed increased intestinal barrier permeability (leaky gut) coupled with upregulated inflammation in the brain and retina, as well as loss of a key functional protein in the eye, they report.
Conversely, these detrimental effects were reversed when microbiota from young mice was transferred to aged mice. FMT with young microbiota also led to enrichment of beneficial taxa in aged mice.
“Our data support the suggestion that altered gut microbiota in old age contributes to intestinal and systemic inflammation, and so may contribute to driving inflammatory pathologies of aged organs,” the study team wrote.
“Targeting the gut-brain axis in aging, by modification of microbial composition to modulate immune and metabolic pathways, may therefore be a potential avenue for therapeutic approaches to age-associated inflammatory and functional decline,” they suggested.
In ongoing studies, the study team are working to understand how long the beneficial effects of young donor microbiota last, which will establish whether FMT can promote long-term health benefits in aged individuals and ameliorate age-associated neurodegeneration and retinal functional deterioration.
“Our results provide more evidence of the important links between microbes in the gut and healthy aging of tissues and organs around the body,” lead author Aimée Parker, PhD, from the Quadram Institute, said in the release.
“We hope that our findings will contribute ultimately to understanding how we can manipulate our diet and our gut bacteria to maximize good health in later life,” she added.
Support for this research was provided by the Biotechnology and Biological Sciences Research Council. The authors report no relevant financial relationships .
A version of this article first appeared on Medscape.com.
Transplanting fecal microbiota from young mice into older mice can reverse signs of aging in the gut, brain, and eyes, a team of scientists from the United Kingdom has found. Conversely, transplanting microbiota from old mice to young mice has the opposite effect.
This research provides “tantalizing evidence for the direct involvement of gut microbes in aging and the functional decline of brain function and vision and offers a potential solution in the form of gut microbe replacement therapy,” Simon Carding, PhD, who heads the gut microbes and health research program at the Quadram Institute in Norwich, England, said in a news release.
The study was published online in the journal Microbiome.
The fountain of youth?
Age-related changes in diversity, composition, and function of the gut microbiota are associated with low-grade systemic inflammation, declining tissue function, and increased susceptibility to age-related chronic diseases.
Dr. Carding and colleagues at the Quadram Institute and the University of East Anglia used fecal microbiota transplant (FMT) to exchange the intestinal microbiota of young mice and aged mice.
Young mice who received aged microbiota showed increased intestinal barrier permeability (leaky gut) coupled with upregulated inflammation in the brain and retina, as well as loss of a key functional protein in the eye, they report.
Conversely, these detrimental effects were reversed when microbiota from young mice was transferred to aged mice. FMT with young microbiota also led to enrichment of beneficial taxa in aged mice.
“Our data support the suggestion that altered gut microbiota in old age contributes to intestinal and systemic inflammation, and so may contribute to driving inflammatory pathologies of aged organs,” the study team wrote.
“Targeting the gut-brain axis in aging, by modification of microbial composition to modulate immune and metabolic pathways, may therefore be a potential avenue for therapeutic approaches to age-associated inflammatory and functional decline,” they suggested.
In ongoing studies, the study team are working to understand how long the beneficial effects of young donor microbiota last, which will establish whether FMT can promote long-term health benefits in aged individuals and ameliorate age-associated neurodegeneration and retinal functional deterioration.
“Our results provide more evidence of the important links between microbes in the gut and healthy aging of tissues and organs around the body,” lead author Aimée Parker, PhD, from the Quadram Institute, said in the release.
“We hope that our findings will contribute ultimately to understanding how we can manipulate our diet and our gut bacteria to maximize good health in later life,” she added.
Support for this research was provided by the Biotechnology and Biological Sciences Research Council. The authors report no relevant financial relationships .
A version of this article first appeared on Medscape.com.
Transplanting fecal microbiota from young mice into older mice can reverse signs of aging in the gut, brain, and eyes, a team of scientists from the United Kingdom has found. Conversely, transplanting microbiota from old mice to young mice has the opposite effect.
This research provides “tantalizing evidence for the direct involvement of gut microbes in aging and the functional decline of brain function and vision and offers a potential solution in the form of gut microbe replacement therapy,” Simon Carding, PhD, who heads the gut microbes and health research program at the Quadram Institute in Norwich, England, said in a news release.
The study was published online in the journal Microbiome.
The fountain of youth?
Age-related changes in diversity, composition, and function of the gut microbiota are associated with low-grade systemic inflammation, declining tissue function, and increased susceptibility to age-related chronic diseases.
Dr. Carding and colleagues at the Quadram Institute and the University of East Anglia used fecal microbiota transplant (FMT) to exchange the intestinal microbiota of young mice and aged mice.
Young mice who received aged microbiota showed increased intestinal barrier permeability (leaky gut) coupled with upregulated inflammation in the brain and retina, as well as loss of a key functional protein in the eye, they report.
Conversely, these detrimental effects were reversed when microbiota from young mice was transferred to aged mice. FMT with young microbiota also led to enrichment of beneficial taxa in aged mice.
“Our data support the suggestion that altered gut microbiota in old age contributes to intestinal and systemic inflammation, and so may contribute to driving inflammatory pathologies of aged organs,” the study team wrote.
“Targeting the gut-brain axis in aging, by modification of microbial composition to modulate immune and metabolic pathways, may therefore be a potential avenue for therapeutic approaches to age-associated inflammatory and functional decline,” they suggested.
In ongoing studies, the study team are working to understand how long the beneficial effects of young donor microbiota last, which will establish whether FMT can promote long-term health benefits in aged individuals and ameliorate age-associated neurodegeneration and retinal functional deterioration.
“Our results provide more evidence of the important links between microbes in the gut and healthy aging of tissues and organs around the body,” lead author Aimée Parker, PhD, from the Quadram Institute, said in the release.
“We hope that our findings will contribute ultimately to understanding how we can manipulate our diet and our gut bacteria to maximize good health in later life,” she added.
Support for this research was provided by the Biotechnology and Biological Sciences Research Council. The authors report no relevant financial relationships .
A version of this article first appeared on Medscape.com.