User login
Daptomycin/fosfomycin: A new standard for MRSA bacteremia?
SAN FRANCISCO – Daptomycin plus fosfomycin is more effective than daptomycin alone for methicillin-resistant Staphylococcus aureus bacteremia, according to a multicenter, randomized trial from Spain.
“I think this is really an important study; I think it will change clinical practice for this infection” once it’s published, said lead investigator Miquel Pujol, MD, PhD, clinical head of infectious diseases at Bellvitge University Hospital in Barcelona.
The current standard for MRSA bacteremia is daptomycin (Cubicin) or vancomycin (Vancocin) monotherapy on both sides of the Atlantic, but mortality rates are way too high, more than 30% in some reviews. Dr. Pujol and his colleagues wanted to find something better.
Their lab work showed that daptomycin and fosfomycin (Monurol) were synergistic and rapidly bactericidal against MRSA, and anecdotal experience in Spain suggested the drugs improved bacteremia outcomes, so they decided to put the combination to the test.
They randomized 74 MRSA bacteremia patients to the combination, daptomycin 10mg/kg IV daily plus fosfomycin 2g IV q 6h. They randomized 81 other subjects to standard of care with daptomycin monotherapy, also at 10mg/kg IV daily. Treatment was 10-14 days for uncomplicated and 28-42 days for complicated bacteremia.
The open-label trial was conducted at 18 medical centers in Spain, where fosfomycin was discovered in dirt samples in the late 1960s and remains a matter of pride.
At day 7, 69 of the 74 combination patients (93.2%) were alive with clinical improvement, clearance of bacteremia, and no subsequent relapse, versus 62 of 81 patients (76.5%) on monotherapy (absolute difference 16.7%; 95% confidence interval, 5.4%-27.7%). Three people in the combination arm (4.1%) had died by day 7, versus six on monotherapy (7.4%).
Six weeks after the end of treatment at the test-of-cure visit, 40 of 74 combination patients (54.1%) were alive with resolution of all clinical signs and symptoms, negative blood cultures, and no previous or subsequent relapses; just 34 of 81 patients (42%) in the monotherapy arm hit that mark. The 12.1% difference was not statistically significant, nor was the difference in 12-week survival.
However, patients in the combination arm were 70% less likely to have complicated bacteremia at the test-of-cure visit (9.5% vs. 28.4%; relative risk 0.3; 95% CI, 0.2-0.7). There were no cases of persistent or recurrent infection in the combination arm, but nine persistent (11.1%) and five recurrent (6.2%) cases with daptomycin monotherapy. The differences were statistically significant.
The subjects all had at least one positive MRSA blood culture within 72 hours of randomization. Exclusion criteria included MRSA pneumonia, prosthetic valve endocarditis, end-stage liver disease, and moderate to severe heart failure.
There were no significant baseline differences between the groups. About half the subjects were men, and the mean age was about 73 years. The mean Charlson Comorbidity Index score was a bit under 4, and the mean Pitt bacteremia score a bit over 1. The leading source of infection was vascular catheter; acquisition was thought to be nosocomial in more than 40% of patients.
There were no discontinuations from drug side effects in the daptomycin arm, but there were five in the combination arm, including two for heart failure, two for respiratory insufficiency, and one for GI bleeding. Even so, the benefit outweighed the risk, Dr. Pujol said.
Intravenous fosfomycin is available in Europe, but the drug is approved in the United States only as an oral formulation. That could change soon; Nabriva Therapeutics plans to file its IV formulation (Contepo) for Food and Drug Administration approval in late 2018.
Though it is not standard of practice yet, the combination is increasingly being used in Spain for MRSA bacteremia, according to Dr. Pujol. “Patients probably need the combination [at least] initially, especially if they have complicated bacteremia” or fail monotherapy, he said at ID week, an annual scientific meeting on infectious diseases.
The work was funded by the Spanish government. Dr. Pujol said he had no relevant disclosures.
SOURCE: Pujol M et al. 2018 ID Week abstract LB3
SAN FRANCISCO – Daptomycin plus fosfomycin is more effective than daptomycin alone for methicillin-resistant Staphylococcus aureus bacteremia, according to a multicenter, randomized trial from Spain.
“I think this is really an important study; I think it will change clinical practice for this infection” once it’s published, said lead investigator Miquel Pujol, MD, PhD, clinical head of infectious diseases at Bellvitge University Hospital in Barcelona.
The current standard for MRSA bacteremia is daptomycin (Cubicin) or vancomycin (Vancocin) monotherapy on both sides of the Atlantic, but mortality rates are way too high, more than 30% in some reviews. Dr. Pujol and his colleagues wanted to find something better.
Their lab work showed that daptomycin and fosfomycin (Monurol) were synergistic and rapidly bactericidal against MRSA, and anecdotal experience in Spain suggested the drugs improved bacteremia outcomes, so they decided to put the combination to the test.
They randomized 74 MRSA bacteremia patients to the combination, daptomycin 10mg/kg IV daily plus fosfomycin 2g IV q 6h. They randomized 81 other subjects to standard of care with daptomycin monotherapy, also at 10mg/kg IV daily. Treatment was 10-14 days for uncomplicated and 28-42 days for complicated bacteremia.
The open-label trial was conducted at 18 medical centers in Spain, where fosfomycin was discovered in dirt samples in the late 1960s and remains a matter of pride.
At day 7, 69 of the 74 combination patients (93.2%) were alive with clinical improvement, clearance of bacteremia, and no subsequent relapse, versus 62 of 81 patients (76.5%) on monotherapy (absolute difference 16.7%; 95% confidence interval, 5.4%-27.7%). Three people in the combination arm (4.1%) had died by day 7, versus six on monotherapy (7.4%).
Six weeks after the end of treatment at the test-of-cure visit, 40 of 74 combination patients (54.1%) were alive with resolution of all clinical signs and symptoms, negative blood cultures, and no previous or subsequent relapses; just 34 of 81 patients (42%) in the monotherapy arm hit that mark. The 12.1% difference was not statistically significant, nor was the difference in 12-week survival.
However, patients in the combination arm were 70% less likely to have complicated bacteremia at the test-of-cure visit (9.5% vs. 28.4%; relative risk 0.3; 95% CI, 0.2-0.7). There were no cases of persistent or recurrent infection in the combination arm, but nine persistent (11.1%) and five recurrent (6.2%) cases with daptomycin monotherapy. The differences were statistically significant.
The subjects all had at least one positive MRSA blood culture within 72 hours of randomization. Exclusion criteria included MRSA pneumonia, prosthetic valve endocarditis, end-stage liver disease, and moderate to severe heart failure.
There were no significant baseline differences between the groups. About half the subjects were men, and the mean age was about 73 years. The mean Charlson Comorbidity Index score was a bit under 4, and the mean Pitt bacteremia score a bit over 1. The leading source of infection was vascular catheter; acquisition was thought to be nosocomial in more than 40% of patients.
There were no discontinuations from drug side effects in the daptomycin arm, but there were five in the combination arm, including two for heart failure, two for respiratory insufficiency, and one for GI bleeding. Even so, the benefit outweighed the risk, Dr. Pujol said.
Intravenous fosfomycin is available in Europe, but the drug is approved in the United States only as an oral formulation. That could change soon; Nabriva Therapeutics plans to file its IV formulation (Contepo) for Food and Drug Administration approval in late 2018.
Though it is not standard of practice yet, the combination is increasingly being used in Spain for MRSA bacteremia, according to Dr. Pujol. “Patients probably need the combination [at least] initially, especially if they have complicated bacteremia” or fail monotherapy, he said at ID week, an annual scientific meeting on infectious diseases.
The work was funded by the Spanish government. Dr. Pujol said he had no relevant disclosures.
SOURCE: Pujol M et al. 2018 ID Week abstract LB3
SAN FRANCISCO – Daptomycin plus fosfomycin is more effective than daptomycin alone for methicillin-resistant Staphylococcus aureus bacteremia, according to a multicenter, randomized trial from Spain.
“I think this is really an important study; I think it will change clinical practice for this infection” once it’s published, said lead investigator Miquel Pujol, MD, PhD, clinical head of infectious diseases at Bellvitge University Hospital in Barcelona.
The current standard for MRSA bacteremia is daptomycin (Cubicin) or vancomycin (Vancocin) monotherapy on both sides of the Atlantic, but mortality rates are way too high, more than 30% in some reviews. Dr. Pujol and his colleagues wanted to find something better.
Their lab work showed that daptomycin and fosfomycin (Monurol) were synergistic and rapidly bactericidal against MRSA, and anecdotal experience in Spain suggested the drugs improved bacteremia outcomes, so they decided to put the combination to the test.
They randomized 74 MRSA bacteremia patients to the combination, daptomycin 10mg/kg IV daily plus fosfomycin 2g IV q 6h. They randomized 81 other subjects to standard of care with daptomycin monotherapy, also at 10mg/kg IV daily. Treatment was 10-14 days for uncomplicated and 28-42 days for complicated bacteremia.
The open-label trial was conducted at 18 medical centers in Spain, where fosfomycin was discovered in dirt samples in the late 1960s and remains a matter of pride.
At day 7, 69 of the 74 combination patients (93.2%) were alive with clinical improvement, clearance of bacteremia, and no subsequent relapse, versus 62 of 81 patients (76.5%) on monotherapy (absolute difference 16.7%; 95% confidence interval, 5.4%-27.7%). Three people in the combination arm (4.1%) had died by day 7, versus six on monotherapy (7.4%).
Six weeks after the end of treatment at the test-of-cure visit, 40 of 74 combination patients (54.1%) were alive with resolution of all clinical signs and symptoms, negative blood cultures, and no previous or subsequent relapses; just 34 of 81 patients (42%) in the monotherapy arm hit that mark. The 12.1% difference was not statistically significant, nor was the difference in 12-week survival.
However, patients in the combination arm were 70% less likely to have complicated bacteremia at the test-of-cure visit (9.5% vs. 28.4%; relative risk 0.3; 95% CI, 0.2-0.7). There were no cases of persistent or recurrent infection in the combination arm, but nine persistent (11.1%) and five recurrent (6.2%) cases with daptomycin monotherapy. The differences were statistically significant.
The subjects all had at least one positive MRSA blood culture within 72 hours of randomization. Exclusion criteria included MRSA pneumonia, prosthetic valve endocarditis, end-stage liver disease, and moderate to severe heart failure.
There were no significant baseline differences between the groups. About half the subjects were men, and the mean age was about 73 years. The mean Charlson Comorbidity Index score was a bit under 4, and the mean Pitt bacteremia score a bit over 1. The leading source of infection was vascular catheter; acquisition was thought to be nosocomial in more than 40% of patients.
There were no discontinuations from drug side effects in the daptomycin arm, but there were five in the combination arm, including two for heart failure, two for respiratory insufficiency, and one for GI bleeding. Even so, the benefit outweighed the risk, Dr. Pujol said.
Intravenous fosfomycin is available in Europe, but the drug is approved in the United States only as an oral formulation. That could change soon; Nabriva Therapeutics plans to file its IV formulation (Contepo) for Food and Drug Administration approval in late 2018.
Though it is not standard of practice yet, the combination is increasingly being used in Spain for MRSA bacteremia, according to Dr. Pujol. “Patients probably need the combination [at least] initially, especially if they have complicated bacteremia” or fail monotherapy, he said at ID week, an annual scientific meeting on infectious diseases.
The work was funded by the Spanish government. Dr. Pujol said he had no relevant disclosures.
SOURCE: Pujol M et al. 2018 ID Week abstract LB3
REPORTING FROM ID WEEK 2018
Key clinical point:
Major finding: At day 93% of the combination patients were alive with clinical improvement, clearance of bacteremia, and no subsequent relapse, vs. 77% on monotherapy.
Study details: Randomized, open label trial in 155 patients with MRSA bacteremia.
Disclosures: The work was funded by the Spanish government. The lead investigator said he had no relevant disclosures.
Source: Pujol M et al. 2018 ID Week, Abstract LB3
What infectious disease should parents be most worried about?
I think the question was intended as polite, dinner party chit chat ... maybe an attempt by a gracious hostess to make sure everyone was engaged in conversation.
“So what pediatric infectious disease should parents be most worried about?” she asked me.
I’ll admit that a couple of perfectly respectable and noncontroversial possibilities crossed my mind before I answered.
Acute flaccid myelitis? Measles?
When I replied, “gonorrhea,” conversation at the table pretty much stopped.
Let me explain. Acute flaccid myelitis is a polio-like neurologic condition that has been grabbing headlines. Yes, it is concerning that most cases have occurred in children and some affected children are left with long-term deficits. Technically though, AFM is a neurologic rather than an infectious disease. When cases occur, we suspect a viral infection but according to the Centers for Disease Control and Prevention, no pathogen has been consistently identified from the spinal fluid of infected patients. From August 2014 to September 2018, the CDC received information on 368 confirmed cases, so AFM fortunately is still rare.
News reports describe measles outbreaks raging in Europe – more than 41,000 cases so far this year, and 40 deaths – and warn that the United States could be next. But let’s be honest: We have a safe and effective vaccine for measles and outbreaks like this don’t happen when individuals are appropriately immunized. Parents, immunize your children. If you are lucky enough to be traveling to Europe with your baby, remember that MMR vaccine is indicated for 6- to 11-month olds, but it doesn’t count in the 2-dose series.
But gonorrhea?
In 2017, the World Health Organization included Neisseria gonorrhoeae on its list of bacteria that pose the greatest threat to human health and for which new antibiotics are urgently needed. The popular media are calling N. gonorrhoeae one of the new “superbugs.” Globally, patients are being diagnosed with strains of gonorrhea that are resistant to all commonly used antibiotics. As reported during IDWeek 2018 this October, patients also are being diagnosed in the United States.
Sancta St. Cyr, MD, of the Centers for Disease Control and Prevention, and her colleagues reported data from the Gonococcal Isolate Surveillance Project (GISP) and trends in multidrug resistant (MDR) and extensively-drug resistant (XDR) gonorrhea in the United States. A gonococcal isolate with resistance or elevated minimum inhibitory concentrations (MIC) to greater than or equal to two classes of antimicrobials is classified as MDR and an isolate with elevated MICs to greater than or equal to three classes of antimicrobials is classified as XDR. The MIC is the lowest antimicrobial concentration that inhibits growth of bacteria in the laboratory and rising MICs – evidence that higher levels of an antibiotic are needed to stop bacterial growth – can be an early indicator that resistance is emerging.
More than 150,000 gonococcal isolates were tested between 1987 and 2016. The first isolates with elevated MICs to cephalosporins and macrolides were identified in 1998, and since 2011, MDR resistance rates have hovered around 1%. In 2016, the rate was 1.1%, down from 1.3% in 2011. A single XDR isolate with resistance to fluoroquinolones with elevated MICs to both cephalosporins and macrolides was identified in 2011.
One could look at these data and ask if this is a “glass half full or half empty” situation, but I propose that clinicians and public health officials should not look at these data and be reassured that rates of MDR-gonorrhea remained stable between 2010 and 2016. According to a recent surveillance report released by the CDC, the absolute number of cases of gonorrhea has continued to rise. In 2017, there were 555,608 cases reported in the United States, a 67% increase since 2013. If we assume that rates of resistance in 2017 were similar to those in 2016, that’s more than 5,000 cases of MDR-gonorrhea in a single year.
“That’s bad,” one of my dining companions agreed. “But is gonorrhea really a pediatric issue?”
To answer that question, we just have to look at the numbers. According to the 2017 Youth Risk Behavior Survey, the percentage of high school students who had ever had sex was approximately 40% and about 10% of students had four or more lifetime partners. More than 45% of sexually active students denied the use of a condom during the last sexual intercourse. Certainly, that puts many teenagers at risk for sexually transmitted infections (STIs). Perhaps it shouldn’t be surprising that public health authorities report that half of all new STIs occur in individuals aged 15-24 years. Moreover, 25% of sexually active adolescent girls contract at least one STI.
Gonorrhea is the second most commonly reported notifiable disease in the United States, and according to the CDC, rates of disease in 2017 were highest among adolescents and young adults. In females specifically, the highest rates of gonorrhea were observed among those aged 20-24 years (684.8 cases per 100,000 females) and 15-19 years (557.4 cases per 100,000 females).
It makes sense that pediatricians and parents advocate for making the reduction of gonorrhea transmission rates a public health priority. We also need to recognize that prompt diagnosis and appropriate treatment are critical. Since 2015, dual therapy with ceftriaxone and azithromycin is the only CDC-recommended treatment for gonorrhea.
At that dinner party, my closest friend, who also happens to be a pediatrician, rolled her eyes and shot me look that I’m sure meant, “Nobody really wants to talk about gonorrhea over dessert.” Still, because she is a good friend she said, “So basically you’re saying that and if this keeps up, we may see kids with untreatable infection. Now that is scary.”
I kept quiet after that but I wanted to mention that in 2017, less than 85% of patients diagnosed with gonorrhea at selected surveillance sites received the recommended treatment with two antibiotics. Patients with inadequately treated gonorrhea are at risk for a host of sequelae. Women can develop pelvic inflammatory disease, abscesses, chronic pelvic pain, and damage of the fallopian tubes that can lead to infertility. Men can develop epididymitis, which occasionally results in infertility. Rarely, N. gonorrhoeae can spread to the blood and cause life-threatening infection. Of course, patients who aren’t treated appropriately may continue to spread the bacteria. Scary? You bet.
For pediatricians who need a refresher course in the treatment of STIs, there are free resources available. The CDC’s 2015 STD Treatment Guidelines are available in a free app; the app contains a nice refresher on taking a sexual history. There also is a print version, wall chart, and pocket guide. Providers also may want to check out the National STD Curriculum offered by the University of Washington STD Prevention Training Center and the University of Washington. Visit https://www.std.uw.edu/.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She said she had no relevant financial disclosures. Email her at [email protected].
I think the question was intended as polite, dinner party chit chat ... maybe an attempt by a gracious hostess to make sure everyone was engaged in conversation.
“So what pediatric infectious disease should parents be most worried about?” she asked me.
I’ll admit that a couple of perfectly respectable and noncontroversial possibilities crossed my mind before I answered.
Acute flaccid myelitis? Measles?
When I replied, “gonorrhea,” conversation at the table pretty much stopped.
Let me explain. Acute flaccid myelitis is a polio-like neurologic condition that has been grabbing headlines. Yes, it is concerning that most cases have occurred in children and some affected children are left with long-term deficits. Technically though, AFM is a neurologic rather than an infectious disease. When cases occur, we suspect a viral infection but according to the Centers for Disease Control and Prevention, no pathogen has been consistently identified from the spinal fluid of infected patients. From August 2014 to September 2018, the CDC received information on 368 confirmed cases, so AFM fortunately is still rare.
News reports describe measles outbreaks raging in Europe – more than 41,000 cases so far this year, and 40 deaths – and warn that the United States could be next. But let’s be honest: We have a safe and effective vaccine for measles and outbreaks like this don’t happen when individuals are appropriately immunized. Parents, immunize your children. If you are lucky enough to be traveling to Europe with your baby, remember that MMR vaccine is indicated for 6- to 11-month olds, but it doesn’t count in the 2-dose series.
But gonorrhea?
In 2017, the World Health Organization included Neisseria gonorrhoeae on its list of bacteria that pose the greatest threat to human health and for which new antibiotics are urgently needed. The popular media are calling N. gonorrhoeae one of the new “superbugs.” Globally, patients are being diagnosed with strains of gonorrhea that are resistant to all commonly used antibiotics. As reported during IDWeek 2018 this October, patients also are being diagnosed in the United States.
Sancta St. Cyr, MD, of the Centers for Disease Control and Prevention, and her colleagues reported data from the Gonococcal Isolate Surveillance Project (GISP) and trends in multidrug resistant (MDR) and extensively-drug resistant (XDR) gonorrhea in the United States. A gonococcal isolate with resistance or elevated minimum inhibitory concentrations (MIC) to greater than or equal to two classes of antimicrobials is classified as MDR and an isolate with elevated MICs to greater than or equal to three classes of antimicrobials is classified as XDR. The MIC is the lowest antimicrobial concentration that inhibits growth of bacteria in the laboratory and rising MICs – evidence that higher levels of an antibiotic are needed to stop bacterial growth – can be an early indicator that resistance is emerging.
More than 150,000 gonococcal isolates were tested between 1987 and 2016. The first isolates with elevated MICs to cephalosporins and macrolides were identified in 1998, and since 2011, MDR resistance rates have hovered around 1%. In 2016, the rate was 1.1%, down from 1.3% in 2011. A single XDR isolate with resistance to fluoroquinolones with elevated MICs to both cephalosporins and macrolides was identified in 2011.
One could look at these data and ask if this is a “glass half full or half empty” situation, but I propose that clinicians and public health officials should not look at these data and be reassured that rates of MDR-gonorrhea remained stable between 2010 and 2016. According to a recent surveillance report released by the CDC, the absolute number of cases of gonorrhea has continued to rise. In 2017, there were 555,608 cases reported in the United States, a 67% increase since 2013. If we assume that rates of resistance in 2017 were similar to those in 2016, that’s more than 5,000 cases of MDR-gonorrhea in a single year.
“That’s bad,” one of my dining companions agreed. “But is gonorrhea really a pediatric issue?”
To answer that question, we just have to look at the numbers. According to the 2017 Youth Risk Behavior Survey, the percentage of high school students who had ever had sex was approximately 40% and about 10% of students had four or more lifetime partners. More than 45% of sexually active students denied the use of a condom during the last sexual intercourse. Certainly, that puts many teenagers at risk for sexually transmitted infections (STIs). Perhaps it shouldn’t be surprising that public health authorities report that half of all new STIs occur in individuals aged 15-24 years. Moreover, 25% of sexually active adolescent girls contract at least one STI.
Gonorrhea is the second most commonly reported notifiable disease in the United States, and according to the CDC, rates of disease in 2017 were highest among adolescents and young adults. In females specifically, the highest rates of gonorrhea were observed among those aged 20-24 years (684.8 cases per 100,000 females) and 15-19 years (557.4 cases per 100,000 females).
It makes sense that pediatricians and parents advocate for making the reduction of gonorrhea transmission rates a public health priority. We also need to recognize that prompt diagnosis and appropriate treatment are critical. Since 2015, dual therapy with ceftriaxone and azithromycin is the only CDC-recommended treatment for gonorrhea.
At that dinner party, my closest friend, who also happens to be a pediatrician, rolled her eyes and shot me look that I’m sure meant, “Nobody really wants to talk about gonorrhea over dessert.” Still, because she is a good friend she said, “So basically you’re saying that and if this keeps up, we may see kids with untreatable infection. Now that is scary.”
I kept quiet after that but I wanted to mention that in 2017, less than 85% of patients diagnosed with gonorrhea at selected surveillance sites received the recommended treatment with two antibiotics. Patients with inadequately treated gonorrhea are at risk for a host of sequelae. Women can develop pelvic inflammatory disease, abscesses, chronic pelvic pain, and damage of the fallopian tubes that can lead to infertility. Men can develop epididymitis, which occasionally results in infertility. Rarely, N. gonorrhoeae can spread to the blood and cause life-threatening infection. Of course, patients who aren’t treated appropriately may continue to spread the bacteria. Scary? You bet.
For pediatricians who need a refresher course in the treatment of STIs, there are free resources available. The CDC’s 2015 STD Treatment Guidelines are available in a free app; the app contains a nice refresher on taking a sexual history. There also is a print version, wall chart, and pocket guide. Providers also may want to check out the National STD Curriculum offered by the University of Washington STD Prevention Training Center and the University of Washington. Visit https://www.std.uw.edu/.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She said she had no relevant financial disclosures. Email her at [email protected].
I think the question was intended as polite, dinner party chit chat ... maybe an attempt by a gracious hostess to make sure everyone was engaged in conversation.
“So what pediatric infectious disease should parents be most worried about?” she asked me.
I’ll admit that a couple of perfectly respectable and noncontroversial possibilities crossed my mind before I answered.
Acute flaccid myelitis? Measles?
When I replied, “gonorrhea,” conversation at the table pretty much stopped.
Let me explain. Acute flaccid myelitis is a polio-like neurologic condition that has been grabbing headlines. Yes, it is concerning that most cases have occurred in children and some affected children are left with long-term deficits. Technically though, AFM is a neurologic rather than an infectious disease. When cases occur, we suspect a viral infection but according to the Centers for Disease Control and Prevention, no pathogen has been consistently identified from the spinal fluid of infected patients. From August 2014 to September 2018, the CDC received information on 368 confirmed cases, so AFM fortunately is still rare.
News reports describe measles outbreaks raging in Europe – more than 41,000 cases so far this year, and 40 deaths – and warn that the United States could be next. But let’s be honest: We have a safe and effective vaccine for measles and outbreaks like this don’t happen when individuals are appropriately immunized. Parents, immunize your children. If you are lucky enough to be traveling to Europe with your baby, remember that MMR vaccine is indicated for 6- to 11-month olds, but it doesn’t count in the 2-dose series.
But gonorrhea?
In 2017, the World Health Organization included Neisseria gonorrhoeae on its list of bacteria that pose the greatest threat to human health and for which new antibiotics are urgently needed. The popular media are calling N. gonorrhoeae one of the new “superbugs.” Globally, patients are being diagnosed with strains of gonorrhea that are resistant to all commonly used antibiotics. As reported during IDWeek 2018 this October, patients also are being diagnosed in the United States.
Sancta St. Cyr, MD, of the Centers for Disease Control and Prevention, and her colleagues reported data from the Gonococcal Isolate Surveillance Project (GISP) and trends in multidrug resistant (MDR) and extensively-drug resistant (XDR) gonorrhea in the United States. A gonococcal isolate with resistance or elevated minimum inhibitory concentrations (MIC) to greater than or equal to two classes of antimicrobials is classified as MDR and an isolate with elevated MICs to greater than or equal to three classes of antimicrobials is classified as XDR. The MIC is the lowest antimicrobial concentration that inhibits growth of bacteria in the laboratory and rising MICs – evidence that higher levels of an antibiotic are needed to stop bacterial growth – can be an early indicator that resistance is emerging.
More than 150,000 gonococcal isolates were tested between 1987 and 2016. The first isolates with elevated MICs to cephalosporins and macrolides were identified in 1998, and since 2011, MDR resistance rates have hovered around 1%. In 2016, the rate was 1.1%, down from 1.3% in 2011. A single XDR isolate with resistance to fluoroquinolones with elevated MICs to both cephalosporins and macrolides was identified in 2011.
One could look at these data and ask if this is a “glass half full or half empty” situation, but I propose that clinicians and public health officials should not look at these data and be reassured that rates of MDR-gonorrhea remained stable between 2010 and 2016. According to a recent surveillance report released by the CDC, the absolute number of cases of gonorrhea has continued to rise. In 2017, there were 555,608 cases reported in the United States, a 67% increase since 2013. If we assume that rates of resistance in 2017 were similar to those in 2016, that’s more than 5,000 cases of MDR-gonorrhea in a single year.
“That’s bad,” one of my dining companions agreed. “But is gonorrhea really a pediatric issue?”
To answer that question, we just have to look at the numbers. According to the 2017 Youth Risk Behavior Survey, the percentage of high school students who had ever had sex was approximately 40% and about 10% of students had four or more lifetime partners. More than 45% of sexually active students denied the use of a condom during the last sexual intercourse. Certainly, that puts many teenagers at risk for sexually transmitted infections (STIs). Perhaps it shouldn’t be surprising that public health authorities report that half of all new STIs occur in individuals aged 15-24 years. Moreover, 25% of sexually active adolescent girls contract at least one STI.
Gonorrhea is the second most commonly reported notifiable disease in the United States, and according to the CDC, rates of disease in 2017 were highest among adolescents and young adults. In females specifically, the highest rates of gonorrhea were observed among those aged 20-24 years (684.8 cases per 100,000 females) and 15-19 years (557.4 cases per 100,000 females).
It makes sense that pediatricians and parents advocate for making the reduction of gonorrhea transmission rates a public health priority. We also need to recognize that prompt diagnosis and appropriate treatment are critical. Since 2015, dual therapy with ceftriaxone and azithromycin is the only CDC-recommended treatment for gonorrhea.
At that dinner party, my closest friend, who also happens to be a pediatrician, rolled her eyes and shot me look that I’m sure meant, “Nobody really wants to talk about gonorrhea over dessert.” Still, because she is a good friend she said, “So basically you’re saying that and if this keeps up, we may see kids with untreatable infection. Now that is scary.”
I kept quiet after that but I wanted to mention that in 2017, less than 85% of patients diagnosed with gonorrhea at selected surveillance sites received the recommended treatment with two antibiotics. Patients with inadequately treated gonorrhea are at risk for a host of sequelae. Women can develop pelvic inflammatory disease, abscesses, chronic pelvic pain, and damage of the fallopian tubes that can lead to infertility. Men can develop epididymitis, which occasionally results in infertility. Rarely, N. gonorrhoeae can spread to the blood and cause life-threatening infection. Of course, patients who aren’t treated appropriately may continue to spread the bacteria. Scary? You bet.
For pediatricians who need a refresher course in the treatment of STIs, there are free resources available. The CDC’s 2015 STD Treatment Guidelines are available in a free app; the app contains a nice refresher on taking a sexual history. There also is a print version, wall chart, and pocket guide. Providers also may want to check out the National STD Curriculum offered by the University of Washington STD Prevention Training Center and the University of Washington. Visit https://www.std.uw.edu/.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She said she had no relevant financial disclosures. Email her at [email protected].
FDA clears Abbott’s Influenza A & B 2, Strep A 2 assays
The Food and Drug Administration has cleared Abbott Laboratories’ next-generation Influenza A & B 2 and Strep A 2 molecular assays for point-of-care testing.
The Influenza A & B 2 assay can detect and differentiate influenza A and B in 13 minutes, with a call-out of positive results at 5 minutes. It can be stored at room temperature, simplifying storage and ordering. The Strep A 2 assay detects group A streptococcus bacterial nucleic acid in 6 minutes, with a call-out of positive results at 2 minutes. Both will be the fastest tests currently on the market in their respective fields, according to a corporate press release.
The assays will be available in a variety of inpatient and outpatient settings, particularly in locations where patients commonly access health care services, such as EDs, physician offices, walk-in clinics, and urgent care centers. This will allow health care providers to make a fast, informed diagnosis and provide appropriate treatment within the span of a single patient visit.
“The ability to obtain early call outs for positive test results with molecular accuracy in as little as 5 minutes for influenza and 2 minutes for strep A is a game-changing development that allows prompt treatment decisions at the point of care. Rapid testing may also help reduce improper antibiotic usage, which can occur when treatment is based exclusively on a patient’s symptoms, and contributes to antibiotic resistance,” Gregory J. Berry, PhD, director of molecular diagnostics at Northwell Health Laboratories in Lake Success, N.Y., said in the press release.
Find the full press release on the Abbott Laboratories website.
The Food and Drug Administration has cleared Abbott Laboratories’ next-generation Influenza A & B 2 and Strep A 2 molecular assays for point-of-care testing.
The Influenza A & B 2 assay can detect and differentiate influenza A and B in 13 minutes, with a call-out of positive results at 5 minutes. It can be stored at room temperature, simplifying storage and ordering. The Strep A 2 assay detects group A streptococcus bacterial nucleic acid in 6 minutes, with a call-out of positive results at 2 minutes. Both will be the fastest tests currently on the market in their respective fields, according to a corporate press release.
The assays will be available in a variety of inpatient and outpatient settings, particularly in locations where patients commonly access health care services, such as EDs, physician offices, walk-in clinics, and urgent care centers. This will allow health care providers to make a fast, informed diagnosis and provide appropriate treatment within the span of a single patient visit.
“The ability to obtain early call outs for positive test results with molecular accuracy in as little as 5 minutes for influenza and 2 minutes for strep A is a game-changing development that allows prompt treatment decisions at the point of care. Rapid testing may also help reduce improper antibiotic usage, which can occur when treatment is based exclusively on a patient’s symptoms, and contributes to antibiotic resistance,” Gregory J. Berry, PhD, director of molecular diagnostics at Northwell Health Laboratories in Lake Success, N.Y., said in the press release.
Find the full press release on the Abbott Laboratories website.
The Food and Drug Administration has cleared Abbott Laboratories’ next-generation Influenza A & B 2 and Strep A 2 molecular assays for point-of-care testing.
The Influenza A & B 2 assay can detect and differentiate influenza A and B in 13 minutes, with a call-out of positive results at 5 minutes. It can be stored at room temperature, simplifying storage and ordering. The Strep A 2 assay detects group A streptococcus bacterial nucleic acid in 6 minutes, with a call-out of positive results at 2 minutes. Both will be the fastest tests currently on the market in their respective fields, according to a corporate press release.
The assays will be available in a variety of inpatient and outpatient settings, particularly in locations where patients commonly access health care services, such as EDs, physician offices, walk-in clinics, and urgent care centers. This will allow health care providers to make a fast, informed diagnosis and provide appropriate treatment within the span of a single patient visit.
“The ability to obtain early call outs for positive test results with molecular accuracy in as little as 5 minutes for influenza and 2 minutes for strep A is a game-changing development that allows prompt treatment decisions at the point of care. Rapid testing may also help reduce improper antibiotic usage, which can occur when treatment is based exclusively on a patient’s symptoms, and contributes to antibiotic resistance,” Gregory J. Berry, PhD, director of molecular diagnostics at Northwell Health Laboratories in Lake Success, N.Y., said in the press release.
Find the full press release on the Abbott Laboratories website.
ACIP votes unanimously in favor of immunization schedule update and redesign
Clinicians consulting the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices vaccination schedules for children, adolescents, and adults in 2019 will find a simpler design and more useful product, according to David Kim, MD, of the Immunization Services Division of the Centers for Disease Control and Prevention, Atlanta.
In a single vote to cover both adult and child/adolescent schedules, the committee voted unanimously in favor of a redesign of the schedules and several clinical updates.
In 2016, the working group for vaccination schedules conducted an ad hoc evaluation of the adult schedule to assess its usability, Dr. Kim said at a meeting of the CDC’s ACIP.
The design of the adult schedule was fully evaluated in 2018 via a three-step process – interviews with 48 health care providers, a redesign of the schedule, and a survey after the redesign. Design changes to the child/adolescent schedule were harmonized with the adult schedule, Dr. Kim explained.
The adult vaccination schedule itself includes several updates in ACIP recommendations in addition to the aesthetic design changes.
The 2019 Adult Immunization Schedule includes the option of the live attenuated influenza vaccine (LAIV) for influenza, the addition of homelessness as an indication for hepatitis A vaccination, and the use of CpG-adjuvanted hepatitis B vaccine, Dr. Kim said.
The additions to the 2019 Child and Adolescent Immunization Schedule are the optional use of the LAIV for influenza, the addition of homelessness as an indication for hepatitis A vaccination, the use of CpG-adjuvanted hepatitis B vaccine (a cytosine phosphoguanosine oligodeoxynucleotide adjuvant), and the addition of the Tdap vaccination of individuals who received Tdap at age 7-10 years.
Some of the key design changes include the use of bright purple on the child/adolescent schedule to more easily distinguish it from the adult version, said Dr. Kim.
Other changes to both schedules include shorter titles, lists of vaccines and trade names, and compartmentalized information for easier reference. Figures have been replaced by tables, and footnotes are simply “Notes” at the end of the schedule, compartmentalized for easier reading, he said. In addition, the schedules include resources for vaccination in outbreak situations and a section on how to report vaccine preventable disease outbreaks.
The ACIP committee members had no relevant financial conflicts to disclose.
Clinicians consulting the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices vaccination schedules for children, adolescents, and adults in 2019 will find a simpler design and more useful product, according to David Kim, MD, of the Immunization Services Division of the Centers for Disease Control and Prevention, Atlanta.
In a single vote to cover both adult and child/adolescent schedules, the committee voted unanimously in favor of a redesign of the schedules and several clinical updates.
In 2016, the working group for vaccination schedules conducted an ad hoc evaluation of the adult schedule to assess its usability, Dr. Kim said at a meeting of the CDC’s ACIP.
The design of the adult schedule was fully evaluated in 2018 via a three-step process – interviews with 48 health care providers, a redesign of the schedule, and a survey after the redesign. Design changes to the child/adolescent schedule were harmonized with the adult schedule, Dr. Kim explained.
The adult vaccination schedule itself includes several updates in ACIP recommendations in addition to the aesthetic design changes.
The 2019 Adult Immunization Schedule includes the option of the live attenuated influenza vaccine (LAIV) for influenza, the addition of homelessness as an indication for hepatitis A vaccination, and the use of CpG-adjuvanted hepatitis B vaccine, Dr. Kim said.
The additions to the 2019 Child and Adolescent Immunization Schedule are the optional use of the LAIV for influenza, the addition of homelessness as an indication for hepatitis A vaccination, the use of CpG-adjuvanted hepatitis B vaccine (a cytosine phosphoguanosine oligodeoxynucleotide adjuvant), and the addition of the Tdap vaccination of individuals who received Tdap at age 7-10 years.
Some of the key design changes include the use of bright purple on the child/adolescent schedule to more easily distinguish it from the adult version, said Dr. Kim.
Other changes to both schedules include shorter titles, lists of vaccines and trade names, and compartmentalized information for easier reference. Figures have been replaced by tables, and footnotes are simply “Notes” at the end of the schedule, compartmentalized for easier reading, he said. In addition, the schedules include resources for vaccination in outbreak situations and a section on how to report vaccine preventable disease outbreaks.
The ACIP committee members had no relevant financial conflicts to disclose.
Clinicians consulting the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices vaccination schedules for children, adolescents, and adults in 2019 will find a simpler design and more useful product, according to David Kim, MD, of the Immunization Services Division of the Centers for Disease Control and Prevention, Atlanta.
In a single vote to cover both adult and child/adolescent schedules, the committee voted unanimously in favor of a redesign of the schedules and several clinical updates.
In 2016, the working group for vaccination schedules conducted an ad hoc evaluation of the adult schedule to assess its usability, Dr. Kim said at a meeting of the CDC’s ACIP.
The design of the adult schedule was fully evaluated in 2018 via a three-step process – interviews with 48 health care providers, a redesign of the schedule, and a survey after the redesign. Design changes to the child/adolescent schedule were harmonized with the adult schedule, Dr. Kim explained.
The adult vaccination schedule itself includes several updates in ACIP recommendations in addition to the aesthetic design changes.
The 2019 Adult Immunization Schedule includes the option of the live attenuated influenza vaccine (LAIV) for influenza, the addition of homelessness as an indication for hepatitis A vaccination, and the use of CpG-adjuvanted hepatitis B vaccine, Dr. Kim said.
The additions to the 2019 Child and Adolescent Immunization Schedule are the optional use of the LAIV for influenza, the addition of homelessness as an indication for hepatitis A vaccination, the use of CpG-adjuvanted hepatitis B vaccine (a cytosine phosphoguanosine oligodeoxynucleotide adjuvant), and the addition of the Tdap vaccination of individuals who received Tdap at age 7-10 years.
Some of the key design changes include the use of bright purple on the child/adolescent schedule to more easily distinguish it from the adult version, said Dr. Kim.
Other changes to both schedules include shorter titles, lists of vaccines and trade names, and compartmentalized information for easier reference. Figures have been replaced by tables, and footnotes are simply “Notes” at the end of the schedule, compartmentalized for easier reading, he said. In addition, the schedules include resources for vaccination in outbreak situations and a section on how to report vaccine preventable disease outbreaks.
The ACIP committee members had no relevant financial conflicts to disclose.
AT AN ACIP MEETING
ACIP supports hepatitis A vaccine for homeless individuals
Homeless individuals aged 1 year and older should be vaccinated against hepatitis A, based on a unanimous vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
“It is important that we take a national approach to vaccinating homeless” people, Noele Nelson, MD, PhD, MPH, of the CDC’s Division of Viral Hepatitis, said in a presentation prior to the vote, in which all 11 committee members voted in favor of hepatitis A vaccination for the homeless population.
Even limited vaccination will increase the herd immunity of the homeless population over time, she said.
Dr. Nelson presented data on the pros and cons of routine hepatitis A vaccination for homeless individuals aged 1 year and older. The Hepatitis Vaccines Work Group convened four meetings in advance of the October ACIP meeting and reached a consensus that homelessness is an independent indication for hepatitis A vaccination, she said.
If the hepatitis A vaccine is included as an ACIP recommendation, “it is more likely to be considered by homeless service providers,” noted Dr. Nelson. She also cited a low quality of evidence for adverse events associated with hepatitis A vaccination.
The work group considerations in the wake of a nationwide hepatitis A outbreak earlier in 2018 included the challenges of controlling outbreaks, which can spread quickly among the homeless population because of poor personal hygiene, limited sanitation, and tight living quarters. These factors make the homeless population more reliant on a vaccine for protection. An outbreak in San Diego, Calif., in particular, occurred largely in the homeless population.
“Routine vaccination is a more feasible approach to reach the homeless over time through regular homeless care providers,” Dr. Nelson said. As for costs, integrating vaccination into routine care for the homeless is cheaper and much less disruptive than the cost of responding to an outbreak.
The “cons” of recommending routine hepatitis A vaccination for the homeless population included the challenges of administrative record keeping. However, during the public comment period, Mae Morgan, MD, an internist who is medical director of Mercy Care Decatur Street & City of Refuge in Atlanta, emphasized that local homeless care organizations have procedures to manage routine vaccination. “If anyone is concerned that there is not a network in place, there are health centers to do this [that] would implement the vaccine.”
The ACIP committee members had no financial conflicts to disclose.
Homeless individuals aged 1 year and older should be vaccinated against hepatitis A, based on a unanimous vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
“It is important that we take a national approach to vaccinating homeless” people, Noele Nelson, MD, PhD, MPH, of the CDC’s Division of Viral Hepatitis, said in a presentation prior to the vote, in which all 11 committee members voted in favor of hepatitis A vaccination for the homeless population.
Even limited vaccination will increase the herd immunity of the homeless population over time, she said.
Dr. Nelson presented data on the pros and cons of routine hepatitis A vaccination for homeless individuals aged 1 year and older. The Hepatitis Vaccines Work Group convened four meetings in advance of the October ACIP meeting and reached a consensus that homelessness is an independent indication for hepatitis A vaccination, she said.
If the hepatitis A vaccine is included as an ACIP recommendation, “it is more likely to be considered by homeless service providers,” noted Dr. Nelson. She also cited a low quality of evidence for adverse events associated with hepatitis A vaccination.
The work group considerations in the wake of a nationwide hepatitis A outbreak earlier in 2018 included the challenges of controlling outbreaks, which can spread quickly among the homeless population because of poor personal hygiene, limited sanitation, and tight living quarters. These factors make the homeless population more reliant on a vaccine for protection. An outbreak in San Diego, Calif., in particular, occurred largely in the homeless population.
“Routine vaccination is a more feasible approach to reach the homeless over time through regular homeless care providers,” Dr. Nelson said. As for costs, integrating vaccination into routine care for the homeless is cheaper and much less disruptive than the cost of responding to an outbreak.
The “cons” of recommending routine hepatitis A vaccination for the homeless population included the challenges of administrative record keeping. However, during the public comment period, Mae Morgan, MD, an internist who is medical director of Mercy Care Decatur Street & City of Refuge in Atlanta, emphasized that local homeless care organizations have procedures to manage routine vaccination. “If anyone is concerned that there is not a network in place, there are health centers to do this [that] would implement the vaccine.”
The ACIP committee members had no financial conflicts to disclose.
Homeless individuals aged 1 year and older should be vaccinated against hepatitis A, based on a unanimous vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
“It is important that we take a national approach to vaccinating homeless” people, Noele Nelson, MD, PhD, MPH, of the CDC’s Division of Viral Hepatitis, said in a presentation prior to the vote, in which all 11 committee members voted in favor of hepatitis A vaccination for the homeless population.
Even limited vaccination will increase the herd immunity of the homeless population over time, she said.
Dr. Nelson presented data on the pros and cons of routine hepatitis A vaccination for homeless individuals aged 1 year and older. The Hepatitis Vaccines Work Group convened four meetings in advance of the October ACIP meeting and reached a consensus that homelessness is an independent indication for hepatitis A vaccination, she said.
If the hepatitis A vaccine is included as an ACIP recommendation, “it is more likely to be considered by homeless service providers,” noted Dr. Nelson. She also cited a low quality of evidence for adverse events associated with hepatitis A vaccination.
The work group considerations in the wake of a nationwide hepatitis A outbreak earlier in 2018 included the challenges of controlling outbreaks, which can spread quickly among the homeless population because of poor personal hygiene, limited sanitation, and tight living quarters. These factors make the homeless population more reliant on a vaccine for protection. An outbreak in San Diego, Calif., in particular, occurred largely in the homeless population.
“Routine vaccination is a more feasible approach to reach the homeless over time through regular homeless care providers,” Dr. Nelson said. As for costs, integrating vaccination into routine care for the homeless is cheaper and much less disruptive than the cost of responding to an outbreak.
The “cons” of recommending routine hepatitis A vaccination for the homeless population included the challenges of administrative record keeping. However, during the public comment period, Mae Morgan, MD, an internist who is medical director of Mercy Care Decatur Street & City of Refuge in Atlanta, emphasized that local homeless care organizations have procedures to manage routine vaccination. “If anyone is concerned that there is not a network in place, there are health centers to do this [that] would implement the vaccine.”
The ACIP committee members had no financial conflicts to disclose.
FROM AN ACIP MEETING
Swollen knee in a kid? Above 9, treat for Lyme
SAN FRANCISCO – There’s no need to wait for western blot results to differentiate Lyme arthritis from septic arthritis in children, as long as your lab, like many, uses the Liaison Borrelia burgdorferi assay, according to investigators at the University of Minnesota, Minneapolis.
Acute, isolated monoarthritis presents with a single swollen joint and pain whether it’s due to Lyme disease or infection, so it’s hard to tell them apart. Current guidelines recommend a two-tier approach to diagnose Lyme arthritis, an initial blood screen followed by western blot confirmation. Screening results come back in a few hours, but western blot confirmation can take days.
In the meantime, children are treated presumptively for the more concerning diagnosis – septic arthritis – which means hospitalization, surgical drainage, and IV antibiotics. Those who turn out to have Lyme are exposed to the risks and costs of unnecessary treatment and delays to proper diagnosis and doxycycline.
When “kids come in with a swollen knee, maybe 10% or 15% end up in the hospital being treated for septic arthritis that they never had. I wanted to see if we can diagnose Lyme arthritis more quickly,” said lead investigator Bazak Sharon, MD, a pediatric infectious disease specialist at the university’s Masonic Children’s Hospital.
Masonic and its affiliated health system use the Liaison Borrelia burgdorferi assay (DiaSorin) to screen for Lyme, and a careful parsing of the results seems to solve the problem.
Liaison is a chemiluminescence immunoassay that uses light to measure IgM and IgG antibodies to a B. burgdorferi surface protein in serum samples. Results are reported as relative light units (RLUs); below 0.9 RLUs is negative; 0.9-1.1 is equivocal, and over 1.1 is positive.
It’s where patients fall in the range of positivity that matters when it comes to differentiating Lyme from septic arthritis, Dr. Sharon said at ID Week, an annual scientific meeting on infectious diseases (Clin Vaccine Immunol. 2008 Dec;15[12]:1796-804).
He and his team reviewed 60 cases of acute, isolated monoarthritis culled from more than 700 children who presented with joint complaints from 2011 to 2016; 47 had Lyme arthritis confirmed by western blot; 13 had septic arthritis.
It turned out that “every single patient with a” Liaison value of 9 RLUs or higher was confirmed on western blot for Lyme. “Under 9, there was not a single case of Lyme arthritis,” Dr. Sharon said. Three other patients with acute arthritis also tested positive on the screen, but their RLU values were below 4; two turned out to be trauma related and one was ultimately diagnosed with juvenile idiopathic arthritis. Western blots were negative in all three.
The RLU number reported on the screening test “appears to correlate very well with Lyme arthritis. In an otherwise healthy child presenting with acute joint swelling, utilizing this screening test can confirm clinical suspicion of Lyme arthritis within hours, and prevent the potential harmful interventions accompanying a misdiagnosis of septic arthritis. Just do the screening. If it comes up above 9, you’ve got Lyme arthritis,” and don’t need to wait for western blot results to treat, Dr. Sharon said.
In other words, above 9, treat for Lyme.
The investigators plan to delve further into their results with sensitivity/specificity and other analyses before publishing. Ultimately, “my goal is to have a better diagnosis algorithm for kids who present with acute, isolated monoarthritis,” Dr. Sharon said.
There was no industry funding for the work, and the investigators didn’t have any disclosures.
SOURCE: Sharon B et al. 2018 ID Week abstract 286.
SAN FRANCISCO – There’s no need to wait for western blot results to differentiate Lyme arthritis from septic arthritis in children, as long as your lab, like many, uses the Liaison Borrelia burgdorferi assay, according to investigators at the University of Minnesota, Minneapolis.
Acute, isolated monoarthritis presents with a single swollen joint and pain whether it’s due to Lyme disease or infection, so it’s hard to tell them apart. Current guidelines recommend a two-tier approach to diagnose Lyme arthritis, an initial blood screen followed by western blot confirmation. Screening results come back in a few hours, but western blot confirmation can take days.
In the meantime, children are treated presumptively for the more concerning diagnosis – septic arthritis – which means hospitalization, surgical drainage, and IV antibiotics. Those who turn out to have Lyme are exposed to the risks and costs of unnecessary treatment and delays to proper diagnosis and doxycycline.
When “kids come in with a swollen knee, maybe 10% or 15% end up in the hospital being treated for septic arthritis that they never had. I wanted to see if we can diagnose Lyme arthritis more quickly,” said lead investigator Bazak Sharon, MD, a pediatric infectious disease specialist at the university’s Masonic Children’s Hospital.
Masonic and its affiliated health system use the Liaison Borrelia burgdorferi assay (DiaSorin) to screen for Lyme, and a careful parsing of the results seems to solve the problem.
Liaison is a chemiluminescence immunoassay that uses light to measure IgM and IgG antibodies to a B. burgdorferi surface protein in serum samples. Results are reported as relative light units (RLUs); below 0.9 RLUs is negative; 0.9-1.1 is equivocal, and over 1.1 is positive.
It’s where patients fall in the range of positivity that matters when it comes to differentiating Lyme from septic arthritis, Dr. Sharon said at ID Week, an annual scientific meeting on infectious diseases (Clin Vaccine Immunol. 2008 Dec;15[12]:1796-804).
He and his team reviewed 60 cases of acute, isolated monoarthritis culled from more than 700 children who presented with joint complaints from 2011 to 2016; 47 had Lyme arthritis confirmed by western blot; 13 had septic arthritis.
It turned out that “every single patient with a” Liaison value of 9 RLUs or higher was confirmed on western blot for Lyme. “Under 9, there was not a single case of Lyme arthritis,” Dr. Sharon said. Three other patients with acute arthritis also tested positive on the screen, but their RLU values were below 4; two turned out to be trauma related and one was ultimately diagnosed with juvenile idiopathic arthritis. Western blots were negative in all three.
The RLU number reported on the screening test “appears to correlate very well with Lyme arthritis. In an otherwise healthy child presenting with acute joint swelling, utilizing this screening test can confirm clinical suspicion of Lyme arthritis within hours, and prevent the potential harmful interventions accompanying a misdiagnosis of septic arthritis. Just do the screening. If it comes up above 9, you’ve got Lyme arthritis,” and don’t need to wait for western blot results to treat, Dr. Sharon said.
In other words, above 9, treat for Lyme.
The investigators plan to delve further into their results with sensitivity/specificity and other analyses before publishing. Ultimately, “my goal is to have a better diagnosis algorithm for kids who present with acute, isolated monoarthritis,” Dr. Sharon said.
There was no industry funding for the work, and the investigators didn’t have any disclosures.
SOURCE: Sharon B et al. 2018 ID Week abstract 286.
SAN FRANCISCO – There’s no need to wait for western blot results to differentiate Lyme arthritis from septic arthritis in children, as long as your lab, like many, uses the Liaison Borrelia burgdorferi assay, according to investigators at the University of Minnesota, Minneapolis.
Acute, isolated monoarthritis presents with a single swollen joint and pain whether it’s due to Lyme disease or infection, so it’s hard to tell them apart. Current guidelines recommend a two-tier approach to diagnose Lyme arthritis, an initial blood screen followed by western blot confirmation. Screening results come back in a few hours, but western blot confirmation can take days.
In the meantime, children are treated presumptively for the more concerning diagnosis – septic arthritis – which means hospitalization, surgical drainage, and IV antibiotics. Those who turn out to have Lyme are exposed to the risks and costs of unnecessary treatment and delays to proper diagnosis and doxycycline.
When “kids come in with a swollen knee, maybe 10% or 15% end up in the hospital being treated for septic arthritis that they never had. I wanted to see if we can diagnose Lyme arthritis more quickly,” said lead investigator Bazak Sharon, MD, a pediatric infectious disease specialist at the university’s Masonic Children’s Hospital.
Masonic and its affiliated health system use the Liaison Borrelia burgdorferi assay (DiaSorin) to screen for Lyme, and a careful parsing of the results seems to solve the problem.
Liaison is a chemiluminescence immunoassay that uses light to measure IgM and IgG antibodies to a B. burgdorferi surface protein in serum samples. Results are reported as relative light units (RLUs); below 0.9 RLUs is negative; 0.9-1.1 is equivocal, and over 1.1 is positive.
It’s where patients fall in the range of positivity that matters when it comes to differentiating Lyme from septic arthritis, Dr. Sharon said at ID Week, an annual scientific meeting on infectious diseases (Clin Vaccine Immunol. 2008 Dec;15[12]:1796-804).
He and his team reviewed 60 cases of acute, isolated monoarthritis culled from more than 700 children who presented with joint complaints from 2011 to 2016; 47 had Lyme arthritis confirmed by western blot; 13 had septic arthritis.
It turned out that “every single patient with a” Liaison value of 9 RLUs or higher was confirmed on western blot for Lyme. “Under 9, there was not a single case of Lyme arthritis,” Dr. Sharon said. Three other patients with acute arthritis also tested positive on the screen, but their RLU values were below 4; two turned out to be trauma related and one was ultimately diagnosed with juvenile idiopathic arthritis. Western blots were negative in all three.
The RLU number reported on the screening test “appears to correlate very well with Lyme arthritis. In an otherwise healthy child presenting with acute joint swelling, utilizing this screening test can confirm clinical suspicion of Lyme arthritis within hours, and prevent the potential harmful interventions accompanying a misdiagnosis of septic arthritis. Just do the screening. If it comes up above 9, you’ve got Lyme arthritis,” and don’t need to wait for western blot results to treat, Dr. Sharon said.
In other words, above 9, treat for Lyme.
The investigators plan to delve further into their results with sensitivity/specificity and other analyses before publishing. Ultimately, “my goal is to have a better diagnosis algorithm for kids who present with acute, isolated monoarthritis,” Dr. Sharon said.
There was no industry funding for the work, and the investigators didn’t have any disclosures.
SOURCE: Sharon B et al. 2018 ID Week abstract 286.
REPORTING FROM IDWEEK 2018
Key clinical point:
Major finding: There was not a single case of Lyme arthritis under 9 RLUs on the screening test.
Study details: Review of 60 children with acute, isolated monoarthritis, culled from more than 700 with joint complaints.
Disclosures: There was no industry funding for the work, and the investigators didn’t have any disclosures.
Source: Sharon B et al. 2018 ID Week abstract 286.
FDA approves Xofluza for treatment of influenza
The Food and Drug Administration has approved Xofluza (baloxavir marboxil) for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for 48 hours or less.
The FDA approval is based on results from two randomized, clinical trials. In both trials, patients who received Xofluza experienced a shorter duration until alleviation of symptoms, compared with patients who received a placebo. In the second trial, patients who received Xofluza and patients who received another approved antiviral influenza medication experienced similar durations until symptom alleviation.
“When treatment is started within 48 hours of becoming sick with flu symptoms, antiviral drugs can lessen symptoms and shorten the time patients feel sick. Having more treatment options that work in different ways to attack the virus is important because flu viruses can become resistant to antiviral drugs,” Debra Birnkrant, MD, director of the Division of Antiviral Products in the FDA’s Center for Drug Evaluation and Research, said in a press release.
The most common adverse events associated with Xofluza were diarrhea and bronchitis.
“This is the first new antiviral flu treatment with a novel mechanism of action approved by the FDA in nearly 20 years,” FDA Commissioner Scott Gottlieb, MD, added. “With thousands of people getting the flu every year, and many people becoming seriously ill, having safe and effective treatment alternatives is critical. This novel drug provides an important, additional treatment option.”
Find the full press release on the FDA website.
The Food and Drug Administration has approved Xofluza (baloxavir marboxil) for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for 48 hours or less.
The FDA approval is based on results from two randomized, clinical trials. In both trials, patients who received Xofluza experienced a shorter duration until alleviation of symptoms, compared with patients who received a placebo. In the second trial, patients who received Xofluza and patients who received another approved antiviral influenza medication experienced similar durations until symptom alleviation.
“When treatment is started within 48 hours of becoming sick with flu symptoms, antiviral drugs can lessen symptoms and shorten the time patients feel sick. Having more treatment options that work in different ways to attack the virus is important because flu viruses can become resistant to antiviral drugs,” Debra Birnkrant, MD, director of the Division of Antiviral Products in the FDA’s Center for Drug Evaluation and Research, said in a press release.
The most common adverse events associated with Xofluza were diarrhea and bronchitis.
“This is the first new antiviral flu treatment with a novel mechanism of action approved by the FDA in nearly 20 years,” FDA Commissioner Scott Gottlieb, MD, added. “With thousands of people getting the flu every year, and many people becoming seriously ill, having safe and effective treatment alternatives is critical. This novel drug provides an important, additional treatment option.”
Find the full press release on the FDA website.
The Food and Drug Administration has approved Xofluza (baloxavir marboxil) for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for 48 hours or less.
The FDA approval is based on results from two randomized, clinical trials. In both trials, patients who received Xofluza experienced a shorter duration until alleviation of symptoms, compared with patients who received a placebo. In the second trial, patients who received Xofluza and patients who received another approved antiviral influenza medication experienced similar durations until symptom alleviation.
“When treatment is started within 48 hours of becoming sick with flu symptoms, antiviral drugs can lessen symptoms and shorten the time patients feel sick. Having more treatment options that work in different ways to attack the virus is important because flu viruses can become resistant to antiviral drugs,” Debra Birnkrant, MD, director of the Division of Antiviral Products in the FDA’s Center for Drug Evaluation and Research, said in a press release.
The most common adverse events associated with Xofluza were diarrhea and bronchitis.
“This is the first new antiviral flu treatment with a novel mechanism of action approved by the FDA in nearly 20 years,” FDA Commissioner Scott Gottlieb, MD, added. “With thousands of people getting the flu every year, and many people becoming seriously ill, having safe and effective treatment alternatives is critical. This novel drug provides an important, additional treatment option.”
Find the full press release on the FDA website.
Quadrivalent flu vaccine okayed for 6 months and up
The expanded approval now includes persons aged 6-59 months; the quadrivalent vaccine had previously been approved for ages 5 years and up. A trivalent version of the Afluria influenza vaccine also now is indicated for people aged 6 months and up, according to an Oct. 4 communication from the FDA.
A total of 172 pediatric influenza-related deaths occurred in the United States during the 2017-2018 season, representing a new high in nonpandemic influenza seasons. About half of the pediatric influenza deaths occurred in otherwise healthy children, and about 22% of children who died were fully vaccinated, according to the Centers for Disease Control and Prevention, reporting U.S. data from 2010 to 2016.
“As we enter a new flu season, we are reminded of the enormous impact that influenza can have on public health,” Seqirus’ vice president of medical affairs Gregg Sylvester, MD, said in a press release announcing the extended indication. “Having another option to fight this disease can translate to saved lives and fewer flu-related hospitalizations this season and going forward.”
According to the CDC, the 2018-2019 influenza vaccine has been updated to provide a better match – and more protection against – viruses circulating in this influenza season. Specifically, says the CDC, the influenza B Victoria lineage and the influenza A(H3N2) components were updated.
In addition to providing protection against these two strains of influenza, trivalent vaccines for the 2018-2019 season are recommended to include protection against H1N1 influenza as well. Quadrivalent vaccines protect against a second influenza B lineage.
Most people will receive a quadrivalent vaccine this year, according to the CDC.
The expanded approval now includes persons aged 6-59 months; the quadrivalent vaccine had previously been approved for ages 5 years and up. A trivalent version of the Afluria influenza vaccine also now is indicated for people aged 6 months and up, according to an Oct. 4 communication from the FDA.
A total of 172 pediatric influenza-related deaths occurred in the United States during the 2017-2018 season, representing a new high in nonpandemic influenza seasons. About half of the pediatric influenza deaths occurred in otherwise healthy children, and about 22% of children who died were fully vaccinated, according to the Centers for Disease Control and Prevention, reporting U.S. data from 2010 to 2016.
“As we enter a new flu season, we are reminded of the enormous impact that influenza can have on public health,” Seqirus’ vice president of medical affairs Gregg Sylvester, MD, said in a press release announcing the extended indication. “Having another option to fight this disease can translate to saved lives and fewer flu-related hospitalizations this season and going forward.”
According to the CDC, the 2018-2019 influenza vaccine has been updated to provide a better match – and more protection against – viruses circulating in this influenza season. Specifically, says the CDC, the influenza B Victoria lineage and the influenza A(H3N2) components were updated.
In addition to providing protection against these two strains of influenza, trivalent vaccines for the 2018-2019 season are recommended to include protection against H1N1 influenza as well. Quadrivalent vaccines protect against a second influenza B lineage.
Most people will receive a quadrivalent vaccine this year, according to the CDC.
The expanded approval now includes persons aged 6-59 months; the quadrivalent vaccine had previously been approved for ages 5 years and up. A trivalent version of the Afluria influenza vaccine also now is indicated for people aged 6 months and up, according to an Oct. 4 communication from the FDA.
A total of 172 pediatric influenza-related deaths occurred in the United States during the 2017-2018 season, representing a new high in nonpandemic influenza seasons. About half of the pediatric influenza deaths occurred in otherwise healthy children, and about 22% of children who died were fully vaccinated, according to the Centers for Disease Control and Prevention, reporting U.S. data from 2010 to 2016.
“As we enter a new flu season, we are reminded of the enormous impact that influenza can have on public health,” Seqirus’ vice president of medical affairs Gregg Sylvester, MD, said in a press release announcing the extended indication. “Having another option to fight this disease can translate to saved lives and fewer flu-related hospitalizations this season and going forward.”
According to the CDC, the 2018-2019 influenza vaccine has been updated to provide a better match – and more protection against – viruses circulating in this influenza season. Specifically, says the CDC, the influenza B Victoria lineage and the influenza A(H3N2) components were updated.
In addition to providing protection against these two strains of influenza, trivalent vaccines for the 2018-2019 season are recommended to include protection against H1N1 influenza as well. Quadrivalent vaccines protect against a second influenza B lineage.
Most people will receive a quadrivalent vaccine this year, according to the CDC.
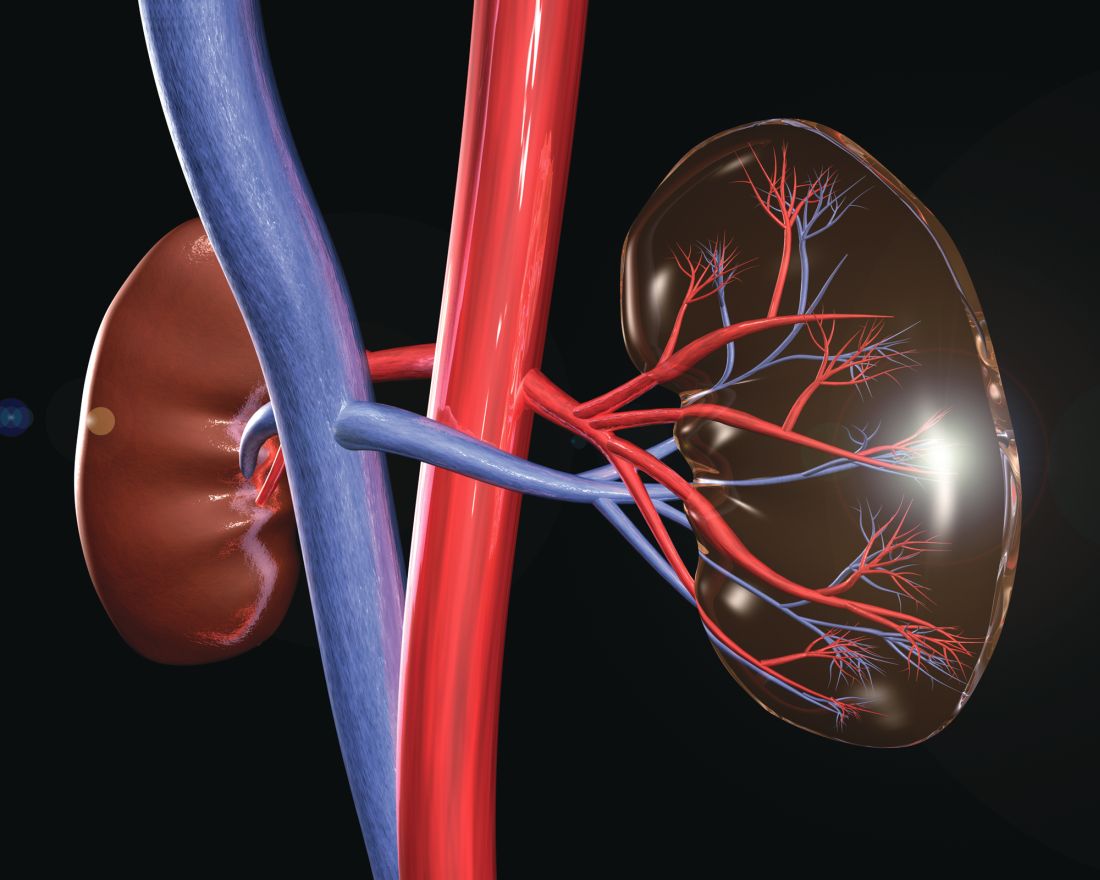
HCV, HBV, and HIV associated with autoimmune kidney diseases
Chronic viral infection can be associated with a variety of autoimmune kidney diseases, according to a review published in Rheumatic Disease Clinics.
In particular, hepatitis C virus (HCV) infection can cause several kidney disorders. These include cryoglobulinemic glomerulonephritis, membranous nephropathy, fibrillary glomerulopathy, immunotactoid glomerulopathy, and IgA nephropathy, wrote Joshua D. Long and his colleagues at Massachusetts General Hospital, Boston.
Similarly, hepatitis B virus (HBV) infection was found to be associated with both membranous nephropathy and polyarteritis nodosa, and human immunodeficiency virus (HIV) infection can cause HIV-associated nephropathy and HIV-associated immune complex diseases, which affect the kidneys.
In their detailed review, the authors discussed the various causal mechanisms and clinical presentations of each of these various autoimmune kidney diseases caused by HCV, HBV, and HIV, along with current treatment modalities.
“Control of the kidney disease relies primarily on treatment of viremia with antiviral agents; however, immunosuppression also may be needed in severe cases,” said the reviewers. However, “more clinical trials are needed to determine first-line therapies for patients who develop autoimmune kidney diseases in the context of chronic viral infections and to define when adjunctive immunosuppressive therapy is warranted,” they concluded.
One of the authors reported grant support and acting as a consultant for various pharmaceutical companies.
[email protected]
SOURCE: Long JD et al. Rheum Dis Clin N Am 2018;44:675-98.
Chronic viral infection can be associated with a variety of autoimmune kidney diseases, according to a review published in Rheumatic Disease Clinics.
In particular, hepatitis C virus (HCV) infection can cause several kidney disorders. These include cryoglobulinemic glomerulonephritis, membranous nephropathy, fibrillary glomerulopathy, immunotactoid glomerulopathy, and IgA nephropathy, wrote Joshua D. Long and his colleagues at Massachusetts General Hospital, Boston.
Similarly, hepatitis B virus (HBV) infection was found to be associated with both membranous nephropathy and polyarteritis nodosa, and human immunodeficiency virus (HIV) infection can cause HIV-associated nephropathy and HIV-associated immune complex diseases, which affect the kidneys.
In their detailed review, the authors discussed the various causal mechanisms and clinical presentations of each of these various autoimmune kidney diseases caused by HCV, HBV, and HIV, along with current treatment modalities.
“Control of the kidney disease relies primarily on treatment of viremia with antiviral agents; however, immunosuppression also may be needed in severe cases,” said the reviewers. However, “more clinical trials are needed to determine first-line therapies for patients who develop autoimmune kidney diseases in the context of chronic viral infections and to define when adjunctive immunosuppressive therapy is warranted,” they concluded.
One of the authors reported grant support and acting as a consultant for various pharmaceutical companies.
[email protected]
SOURCE: Long JD et al. Rheum Dis Clin N Am 2018;44:675-98.
Chronic viral infection can be associated with a variety of autoimmune kidney diseases, according to a review published in Rheumatic Disease Clinics.
In particular, hepatitis C virus (HCV) infection can cause several kidney disorders. These include cryoglobulinemic glomerulonephritis, membranous nephropathy, fibrillary glomerulopathy, immunotactoid glomerulopathy, and IgA nephropathy, wrote Joshua D. Long and his colleagues at Massachusetts General Hospital, Boston.
Similarly, hepatitis B virus (HBV) infection was found to be associated with both membranous nephropathy and polyarteritis nodosa, and human immunodeficiency virus (HIV) infection can cause HIV-associated nephropathy and HIV-associated immune complex diseases, which affect the kidneys.
In their detailed review, the authors discussed the various causal mechanisms and clinical presentations of each of these various autoimmune kidney diseases caused by HCV, HBV, and HIV, along with current treatment modalities.
“Control of the kidney disease relies primarily on treatment of viremia with antiviral agents; however, immunosuppression also may be needed in severe cases,” said the reviewers. However, “more clinical trials are needed to determine first-line therapies for patients who develop autoimmune kidney diseases in the context of chronic viral infections and to define when adjunctive immunosuppressive therapy is warranted,” they concluded.
One of the authors reported grant support and acting as a consultant for various pharmaceutical companies.
[email protected]
SOURCE: Long JD et al. Rheum Dis Clin N Am 2018;44:675-98.
FROM RHEUMATIC DISEASE CLINICS
Age, insurance type tied to delay in pediatric febrile UTI treatment
even after adjustment for confounders, according to a report in the Journal of Pediatrics.
Stephanie W. Hum and Nader Shaikh, MD, MPH, of the University of Pittsburgh, drew from data provided by two studies, Randomized Intervention for Children With Vesicoureteral Reflux and Careful Urinary Tract Infection Evaluation. Specifically, they extracted data regarding patients’ age, sex, history of UTIs, ethnicity, race, insurance, household size, and duration of fever before initiation of antimicrobial therapy, as well as primary caregivers’ level of education and income. Some factors were analyzed because of associations seen in adult studies, and others because of concerns about access to care. In this analysis, the researchers defined “treatment delay” as the number of hours between onset of fever and initiation of antimicrobial treatment and, after exclusion of afebrile children and those with missing data, included 660 patients.
In univariate analysis, both older age and commercial insurance were found to be significantly associated with delays in treatment. Compared with time to treatment seen with younger children, treatment was delayed by an average of 26.2 hours in children aged 12 months and older (P less than .001). Patients with commercial insurance were treated a mean of 12.6 hours later than were those with noncommercial insurance (P = .002). These associations remained significant even after adjustment in a multivariable regression model for sex, history of UTIs, ethnicity, race, primary caregivers’ level of education, insurance, and income level.
The finding regarding age is consistent with a previous study, and Ms. Hum and Dr. Shaikh suggested it may reflect parents experiencing reduced urgency regarding febrile illnesses among older children. However, the researchers also noted that greater rates of renal scarring are seen in older children, so “it seems important to educate physicians, parents, and triage nurses about the importance of early evaluation of children with fever,” even those older than 12 months.
The finding regarding insurance status, however, is contrary to what studies in adult populations have found, as well as those in pediatric EDs. The researchers suggested that perhaps parents with noncommercial insurance are more likely to take their children to EDs, where testing can be done on-site 24 hours a day, rather than to private clinics, which often have to send out testing to off-site laboratories.
One of the strengths of the study is its relatively large sample size, they said. Among its weaknesses is that treatment delays were self-reported by parents and might be inaccurate and that information regarding location of initial evaluation was not gathered and could not be examined with other factors.
This study was supported by a T35 training grant from the National Institute of Diabetes and Digestive and Kidney Diseases, sponsored by Tom R. Kleyman, MD, chief of the renal-electrolyte division at the University of Pittsburgh. The authors declared no conflicts of interest.
SOURCE: Hum SW et al. J Pediatr. 2018 Oct 16. doi: 10.1016/j.jpeds.2018.09.029.
This article was updated 10/24/18.
even after adjustment for confounders, according to a report in the Journal of Pediatrics.
Stephanie W. Hum and Nader Shaikh, MD, MPH, of the University of Pittsburgh, drew from data provided by two studies, Randomized Intervention for Children With Vesicoureteral Reflux and Careful Urinary Tract Infection Evaluation. Specifically, they extracted data regarding patients’ age, sex, history of UTIs, ethnicity, race, insurance, household size, and duration of fever before initiation of antimicrobial therapy, as well as primary caregivers’ level of education and income. Some factors were analyzed because of associations seen in adult studies, and others because of concerns about access to care. In this analysis, the researchers defined “treatment delay” as the number of hours between onset of fever and initiation of antimicrobial treatment and, after exclusion of afebrile children and those with missing data, included 660 patients.
In univariate analysis, both older age and commercial insurance were found to be significantly associated with delays in treatment. Compared with time to treatment seen with younger children, treatment was delayed by an average of 26.2 hours in children aged 12 months and older (P less than .001). Patients with commercial insurance were treated a mean of 12.6 hours later than were those with noncommercial insurance (P = .002). These associations remained significant even after adjustment in a multivariable regression model for sex, history of UTIs, ethnicity, race, primary caregivers’ level of education, insurance, and income level.
The finding regarding age is consistent with a previous study, and Ms. Hum and Dr. Shaikh suggested it may reflect parents experiencing reduced urgency regarding febrile illnesses among older children. However, the researchers also noted that greater rates of renal scarring are seen in older children, so “it seems important to educate physicians, parents, and triage nurses about the importance of early evaluation of children with fever,” even those older than 12 months.
The finding regarding insurance status, however, is contrary to what studies in adult populations have found, as well as those in pediatric EDs. The researchers suggested that perhaps parents with noncommercial insurance are more likely to take their children to EDs, where testing can be done on-site 24 hours a day, rather than to private clinics, which often have to send out testing to off-site laboratories.
One of the strengths of the study is its relatively large sample size, they said. Among its weaknesses is that treatment delays were self-reported by parents and might be inaccurate and that information regarding location of initial evaluation was not gathered and could not be examined with other factors.
This study was supported by a T35 training grant from the National Institute of Diabetes and Digestive and Kidney Diseases, sponsored by Tom R. Kleyman, MD, chief of the renal-electrolyte division at the University of Pittsburgh. The authors declared no conflicts of interest.
SOURCE: Hum SW et al. J Pediatr. 2018 Oct 16. doi: 10.1016/j.jpeds.2018.09.029.
This article was updated 10/24/18.
even after adjustment for confounders, according to a report in the Journal of Pediatrics.
Stephanie W. Hum and Nader Shaikh, MD, MPH, of the University of Pittsburgh, drew from data provided by two studies, Randomized Intervention for Children With Vesicoureteral Reflux and Careful Urinary Tract Infection Evaluation. Specifically, they extracted data regarding patients’ age, sex, history of UTIs, ethnicity, race, insurance, household size, and duration of fever before initiation of antimicrobial therapy, as well as primary caregivers’ level of education and income. Some factors were analyzed because of associations seen in adult studies, and others because of concerns about access to care. In this analysis, the researchers defined “treatment delay” as the number of hours between onset of fever and initiation of antimicrobial treatment and, after exclusion of afebrile children and those with missing data, included 660 patients.
In univariate analysis, both older age and commercial insurance were found to be significantly associated with delays in treatment. Compared with time to treatment seen with younger children, treatment was delayed by an average of 26.2 hours in children aged 12 months and older (P less than .001). Patients with commercial insurance were treated a mean of 12.6 hours later than were those with noncommercial insurance (P = .002). These associations remained significant even after adjustment in a multivariable regression model for sex, history of UTIs, ethnicity, race, primary caregivers’ level of education, insurance, and income level.
The finding regarding age is consistent with a previous study, and Ms. Hum and Dr. Shaikh suggested it may reflect parents experiencing reduced urgency regarding febrile illnesses among older children. However, the researchers also noted that greater rates of renal scarring are seen in older children, so “it seems important to educate physicians, parents, and triage nurses about the importance of early evaluation of children with fever,” even those older than 12 months.
The finding regarding insurance status, however, is contrary to what studies in adult populations have found, as well as those in pediatric EDs. The researchers suggested that perhaps parents with noncommercial insurance are more likely to take their children to EDs, where testing can be done on-site 24 hours a day, rather than to private clinics, which often have to send out testing to off-site laboratories.
One of the strengths of the study is its relatively large sample size, they said. Among its weaknesses is that treatment delays were self-reported by parents and might be inaccurate and that information regarding location of initial evaluation was not gathered and could not be examined with other factors.
This study was supported by a T35 training grant from the National Institute of Diabetes and Digestive and Kidney Diseases, sponsored by Tom R. Kleyman, MD, chief of the renal-electrolyte division at the University of Pittsburgh. The authors declared no conflicts of interest.
SOURCE: Hum SW et al. J Pediatr. 2018 Oct 16. doi: 10.1016/j.jpeds.2018.09.029.
This article was updated 10/24/18.
FROM THE JOURNAL OF PEDIATRICS