User login
The COVID-19 pandemic and changes in pediatric respiratory and nonrespiratory illnesses
The COVID-19 pandemic upended the U.S. health care market and disrupted much of what was thought to be consistent and necessary hospital-based care for children. Early in the pandemic, clinics closed, elective surgeries were delayed, and well visits were postponed. Mitigation strategies were launched nationwide to limit the spread of SARS-CoV-2 including mask mandates, social distancing, shelter-in-place orders, and school closures. While these measures were enacted to target COVID-19, a potential off-target effect was reductions in transmission of other respiratory illness, and potentially nonrespiratory infectious illnesses and conditions exacerbated by acute infections.1 These measures have heavily impacted the pediatric population, wherein respiratory infections are common, and also because daycares and school can be hubs for disease transmission.2
To evaluate the effect of the COVID-19 pandemic on pediatric health care utilization, we performed a multicenter, cross-sectional study of 44 children’s hospitals using the Pediatric Health Information System (PHIS) database.3 Children aged 2 months to 18 years discharged from a PHIS hospital with nonsurgical diagnoses from Jan. 1 to Sept. 30 over a 4-year period (2017-2020) were included in the study. The primary exposure was the 2020 COVID-19 pandemic, which was divided into three study periods: pre–COVID-19 (January–February 2020), early COVID-19 (March-April 2020), and COVID-19 (May-September 2020). The primary outcomes were the observed-to-expected ratio of respiratory and nonrespiratory illness encounters of the study period, compared with the 3 years prior to the pandemic. For these calculations, the expected encounters for each period was derived from the same calendar periods from prepandemic years (2017-2019).
A total of 9,051,980 pediatric encounters were included in the analyses: 6,811,799 with nonrespiratory illnesses and 2,240,181 with respiratory illnesses. We found a 42% reduction in overall encounters during the COVID-19 period, compared with the 3 years prior to the pandemic, with a greater reduction in respiratory, compared with nonrespiratory illnesses, which decreased 62% and 38%, respectively. These reductions were consistent across geographic and encounter type (ED vs. hospitalization). The frequency of hospital-based encounters for common pediatric respiratory illnesses was substantially reduced, with reductions in asthma exacerbations (down 76%), pneumonia (down 81%), croup (down 84%), influenza (down 87%) and bronchiolitis (down 91%). Differences in both respiratory and nonrespiratory illnesses varied by age, with larger reductions found in children aged less than 12 years. While adolescent (children aged over 12 years) encounters diminished during the early COVID period for both respiratory and nonrespiratory illnesses, their encounters returned to previous levels faster than those from younger children. For respiratory illnesses, hospital-based adolescents encounters had returned to prepandemic levels by the end of the study period (September 2020).
These findings warrant consideration as relaxation of SARS-CoV-2 mitigation are contemplated. Encounters for respiratory and nonrespiratory illnesses declined less and recovered faster in adolescents, compared with younger children. The underlying contributors to this trend are likely multifactorial. For example, respiratory illnesses such as croup and bronchiolitis are more common in younger children and adolescents may be more likely to transmit SARS-CoV-2, compared with younger age groups.4,5 However, adolescents may have had less strict adherence to social distancing measures.6 Future efforts to halt transmission of SARS-CoV-2, as well as other respiratory pathogens, should inform mitigation efforts in the adolescent population with considerations of the intensity of social mixing in different pediatric age groups.
While reductions in encounters caused by respiratory illnesses were substantial, more modest but similar age-based trends were seen in nonrespiratory illnesses. Yet, reduced transmission of infectious agents may not fully explain these findings. For example, it is possible that families sought care for mild to moderate nonrespiratory illness in clinics or via telehealth rather than the EDs.7 Provided there were no unintended negative consequences, such transition of care to non-ED settings would suggest there was overutilization of hospital resources prior to the pandemic. Additional assessments would be helpful to examine this more closely and to clarify the long-term impact of those transitions.
It is also possible that the pandemic effects on financial, social, and family stress may have led to increases in some pediatric health care encounters, such as those for mental health conditions,8 nonaccidental trauma or inability to adhere to treatment because of lack of resources.9,10 Additional study on the evolution and distribution of social and stress-related illnesses is critical to maintain and improve the health of children and adolescents.
The COVID-19 pandemic resulted in rapid and marked changes to both communicable and noncommunicable illnesses and care-seeking behaviors. Some of these findings are encouraging, such as large reductions in respiratory and nonrespiratory illnesses. However, other trends may be harbingers of negative health consequences of the pandemic, such as increases in health care utilization later in the pandemic. Further study of the evolving pandemic’s effects on disease and health care utilization is needed to benefit our children now and during the next pandemic.
Dr. Antoon is an assistant professor of pediatrics at Vanderbilt University and a pediatric hospitalist at the Monroe Carroll Jr. Children’s Hospital at Vanderbilt, both in Nashville, Tenn.
References
1. Kenyon CC et al. Initial effects of the COVID-19 pandemic on pediatric asthma emergency department utilization. J Allergy Clin Immunol Pract. 2020 Sep;8(8):2774-6.e1. doi: 10.1016/j.jaip.2020.05.045.
2. Luca G et al. The impact of regular school closure on seasonal influenza epidemics: A data-driven spatial transmission model for Belgium. BMC Infect Dis. 2018;18(1):29. doi: 10.1186/s12879-017-2934-3.
3. Antoon JW et al. The COVID-19 Pandemic and changes in healthcare utilization for pediatric respiratory and nonrespiratory illnesses in the United States. J Hosp Med. 2021 Mar 8. doi: 10.12788/jhm.3608.
4. Park YJ et al. Contact tracing during coronavirus disease outbreak, South Korea, 2020. Emerg Infect Dis. 2020 Oct;26(10):2465-8. doi: 10.3201/eid2610.201315.
5. Davies NG et al. Age-dependent effects in the transmission and control of COVID-19 epidemics. Nat Med. 2020 Aug;26(8):1205-11. doi: 10.1038/s41591-020-0962-9.
6. Andrews JL et al. Peer influence in adolescence: Public health implications for COVID-19. Trends Cogn Sci. 2020;24(8):585-7. doi: 10.1016/j.tics.2020.05.001.
7. Taquechel K et al. Pediatric asthma healthcare utilization, viral testing, and air pollution changes during the COVID-19 pandemic. J Allergy Clin Immunol Pract. 2020 Nov-Dec;8(10):3378-87.e11. doi: 10.1016/j.jaip.2020.07.057.
8. Hill RM et al. Suicide ideation and attempts in a pediatric emergency department before and during COVID-19. Pediatrics. 2021;147(3):e2020029280. doi: 10.1542/peds.2020-029280.
9. Sharma S et al. COVID-19: Differences in sentinel injury and child abuse reporting during a pandemic. Child Abuse Negl. 2020 Dec;110:104709. doi: 10.1016/j.chiabu.2020.104709.
10. Lauren BN et al. Predictors of households at risk for food insecurity in the United States during the COVID-19 pandemic. Public Health Nutr. 2021 Jan 27. doi: 10.1017/S1368980021000355.
The COVID-19 pandemic upended the U.S. health care market and disrupted much of what was thought to be consistent and necessary hospital-based care for children. Early in the pandemic, clinics closed, elective surgeries were delayed, and well visits were postponed. Mitigation strategies were launched nationwide to limit the spread of SARS-CoV-2 including mask mandates, social distancing, shelter-in-place orders, and school closures. While these measures were enacted to target COVID-19, a potential off-target effect was reductions in transmission of other respiratory illness, and potentially nonrespiratory infectious illnesses and conditions exacerbated by acute infections.1 These measures have heavily impacted the pediatric population, wherein respiratory infections are common, and also because daycares and school can be hubs for disease transmission.2
To evaluate the effect of the COVID-19 pandemic on pediatric health care utilization, we performed a multicenter, cross-sectional study of 44 children’s hospitals using the Pediatric Health Information System (PHIS) database.3 Children aged 2 months to 18 years discharged from a PHIS hospital with nonsurgical diagnoses from Jan. 1 to Sept. 30 over a 4-year period (2017-2020) were included in the study. The primary exposure was the 2020 COVID-19 pandemic, which was divided into three study periods: pre–COVID-19 (January–February 2020), early COVID-19 (March-April 2020), and COVID-19 (May-September 2020). The primary outcomes were the observed-to-expected ratio of respiratory and nonrespiratory illness encounters of the study period, compared with the 3 years prior to the pandemic. For these calculations, the expected encounters for each period was derived from the same calendar periods from prepandemic years (2017-2019).
A total of 9,051,980 pediatric encounters were included in the analyses: 6,811,799 with nonrespiratory illnesses and 2,240,181 with respiratory illnesses. We found a 42% reduction in overall encounters during the COVID-19 period, compared with the 3 years prior to the pandemic, with a greater reduction in respiratory, compared with nonrespiratory illnesses, which decreased 62% and 38%, respectively. These reductions were consistent across geographic and encounter type (ED vs. hospitalization). The frequency of hospital-based encounters for common pediatric respiratory illnesses was substantially reduced, with reductions in asthma exacerbations (down 76%), pneumonia (down 81%), croup (down 84%), influenza (down 87%) and bronchiolitis (down 91%). Differences in both respiratory and nonrespiratory illnesses varied by age, with larger reductions found in children aged less than 12 years. While adolescent (children aged over 12 years) encounters diminished during the early COVID period for both respiratory and nonrespiratory illnesses, their encounters returned to previous levels faster than those from younger children. For respiratory illnesses, hospital-based adolescents encounters had returned to prepandemic levels by the end of the study period (September 2020).
These findings warrant consideration as relaxation of SARS-CoV-2 mitigation are contemplated. Encounters for respiratory and nonrespiratory illnesses declined less and recovered faster in adolescents, compared with younger children. The underlying contributors to this trend are likely multifactorial. For example, respiratory illnesses such as croup and bronchiolitis are more common in younger children and adolescents may be more likely to transmit SARS-CoV-2, compared with younger age groups.4,5 However, adolescents may have had less strict adherence to social distancing measures.6 Future efforts to halt transmission of SARS-CoV-2, as well as other respiratory pathogens, should inform mitigation efforts in the adolescent population with considerations of the intensity of social mixing in different pediatric age groups.
While reductions in encounters caused by respiratory illnesses were substantial, more modest but similar age-based trends were seen in nonrespiratory illnesses. Yet, reduced transmission of infectious agents may not fully explain these findings. For example, it is possible that families sought care for mild to moderate nonrespiratory illness in clinics or via telehealth rather than the EDs.7 Provided there were no unintended negative consequences, such transition of care to non-ED settings would suggest there was overutilization of hospital resources prior to the pandemic. Additional assessments would be helpful to examine this more closely and to clarify the long-term impact of those transitions.
It is also possible that the pandemic effects on financial, social, and family stress may have led to increases in some pediatric health care encounters, such as those for mental health conditions,8 nonaccidental trauma or inability to adhere to treatment because of lack of resources.9,10 Additional study on the evolution and distribution of social and stress-related illnesses is critical to maintain and improve the health of children and adolescents.
The COVID-19 pandemic resulted in rapid and marked changes to both communicable and noncommunicable illnesses and care-seeking behaviors. Some of these findings are encouraging, such as large reductions in respiratory and nonrespiratory illnesses. However, other trends may be harbingers of negative health consequences of the pandemic, such as increases in health care utilization later in the pandemic. Further study of the evolving pandemic’s effects on disease and health care utilization is needed to benefit our children now and during the next pandemic.
Dr. Antoon is an assistant professor of pediatrics at Vanderbilt University and a pediatric hospitalist at the Monroe Carroll Jr. Children’s Hospital at Vanderbilt, both in Nashville, Tenn.
References
1. Kenyon CC et al. Initial effects of the COVID-19 pandemic on pediatric asthma emergency department utilization. J Allergy Clin Immunol Pract. 2020 Sep;8(8):2774-6.e1. doi: 10.1016/j.jaip.2020.05.045.
2. Luca G et al. The impact of regular school closure on seasonal influenza epidemics: A data-driven spatial transmission model for Belgium. BMC Infect Dis. 2018;18(1):29. doi: 10.1186/s12879-017-2934-3.
3. Antoon JW et al. The COVID-19 Pandemic and changes in healthcare utilization for pediatric respiratory and nonrespiratory illnesses in the United States. J Hosp Med. 2021 Mar 8. doi: 10.12788/jhm.3608.
4. Park YJ et al. Contact tracing during coronavirus disease outbreak, South Korea, 2020. Emerg Infect Dis. 2020 Oct;26(10):2465-8. doi: 10.3201/eid2610.201315.
5. Davies NG et al. Age-dependent effects in the transmission and control of COVID-19 epidemics. Nat Med. 2020 Aug;26(8):1205-11. doi: 10.1038/s41591-020-0962-9.
6. Andrews JL et al. Peer influence in adolescence: Public health implications for COVID-19. Trends Cogn Sci. 2020;24(8):585-7. doi: 10.1016/j.tics.2020.05.001.
7. Taquechel K et al. Pediatric asthma healthcare utilization, viral testing, and air pollution changes during the COVID-19 pandemic. J Allergy Clin Immunol Pract. 2020 Nov-Dec;8(10):3378-87.e11. doi: 10.1016/j.jaip.2020.07.057.
8. Hill RM et al. Suicide ideation and attempts in a pediatric emergency department before and during COVID-19. Pediatrics. 2021;147(3):e2020029280. doi: 10.1542/peds.2020-029280.
9. Sharma S et al. COVID-19: Differences in sentinel injury and child abuse reporting during a pandemic. Child Abuse Negl. 2020 Dec;110:104709. doi: 10.1016/j.chiabu.2020.104709.
10. Lauren BN et al. Predictors of households at risk for food insecurity in the United States during the COVID-19 pandemic. Public Health Nutr. 2021 Jan 27. doi: 10.1017/S1368980021000355.
The COVID-19 pandemic upended the U.S. health care market and disrupted much of what was thought to be consistent and necessary hospital-based care for children. Early in the pandemic, clinics closed, elective surgeries were delayed, and well visits were postponed. Mitigation strategies were launched nationwide to limit the spread of SARS-CoV-2 including mask mandates, social distancing, shelter-in-place orders, and school closures. While these measures were enacted to target COVID-19, a potential off-target effect was reductions in transmission of other respiratory illness, and potentially nonrespiratory infectious illnesses and conditions exacerbated by acute infections.1 These measures have heavily impacted the pediatric population, wherein respiratory infections are common, and also because daycares and school can be hubs for disease transmission.2
To evaluate the effect of the COVID-19 pandemic on pediatric health care utilization, we performed a multicenter, cross-sectional study of 44 children’s hospitals using the Pediatric Health Information System (PHIS) database.3 Children aged 2 months to 18 years discharged from a PHIS hospital with nonsurgical diagnoses from Jan. 1 to Sept. 30 over a 4-year period (2017-2020) were included in the study. The primary exposure was the 2020 COVID-19 pandemic, which was divided into three study periods: pre–COVID-19 (January–February 2020), early COVID-19 (March-April 2020), and COVID-19 (May-September 2020). The primary outcomes were the observed-to-expected ratio of respiratory and nonrespiratory illness encounters of the study period, compared with the 3 years prior to the pandemic. For these calculations, the expected encounters for each period was derived from the same calendar periods from prepandemic years (2017-2019).
A total of 9,051,980 pediatric encounters were included in the analyses: 6,811,799 with nonrespiratory illnesses and 2,240,181 with respiratory illnesses. We found a 42% reduction in overall encounters during the COVID-19 period, compared with the 3 years prior to the pandemic, with a greater reduction in respiratory, compared with nonrespiratory illnesses, which decreased 62% and 38%, respectively. These reductions were consistent across geographic and encounter type (ED vs. hospitalization). The frequency of hospital-based encounters for common pediatric respiratory illnesses was substantially reduced, with reductions in asthma exacerbations (down 76%), pneumonia (down 81%), croup (down 84%), influenza (down 87%) and bronchiolitis (down 91%). Differences in both respiratory and nonrespiratory illnesses varied by age, with larger reductions found in children aged less than 12 years. While adolescent (children aged over 12 years) encounters diminished during the early COVID period for both respiratory and nonrespiratory illnesses, their encounters returned to previous levels faster than those from younger children. For respiratory illnesses, hospital-based adolescents encounters had returned to prepandemic levels by the end of the study period (September 2020).
These findings warrant consideration as relaxation of SARS-CoV-2 mitigation are contemplated. Encounters for respiratory and nonrespiratory illnesses declined less and recovered faster in adolescents, compared with younger children. The underlying contributors to this trend are likely multifactorial. For example, respiratory illnesses such as croup and bronchiolitis are more common in younger children and adolescents may be more likely to transmit SARS-CoV-2, compared with younger age groups.4,5 However, adolescents may have had less strict adherence to social distancing measures.6 Future efforts to halt transmission of SARS-CoV-2, as well as other respiratory pathogens, should inform mitigation efforts in the adolescent population with considerations of the intensity of social mixing in different pediatric age groups.
While reductions in encounters caused by respiratory illnesses were substantial, more modest but similar age-based trends were seen in nonrespiratory illnesses. Yet, reduced transmission of infectious agents may not fully explain these findings. For example, it is possible that families sought care for mild to moderate nonrespiratory illness in clinics or via telehealth rather than the EDs.7 Provided there were no unintended negative consequences, such transition of care to non-ED settings would suggest there was overutilization of hospital resources prior to the pandemic. Additional assessments would be helpful to examine this more closely and to clarify the long-term impact of those transitions.
It is also possible that the pandemic effects on financial, social, and family stress may have led to increases in some pediatric health care encounters, such as those for mental health conditions,8 nonaccidental trauma or inability to adhere to treatment because of lack of resources.9,10 Additional study on the evolution and distribution of social and stress-related illnesses is critical to maintain and improve the health of children and adolescents.
The COVID-19 pandemic resulted in rapid and marked changes to both communicable and noncommunicable illnesses and care-seeking behaviors. Some of these findings are encouraging, such as large reductions in respiratory and nonrespiratory illnesses. However, other trends may be harbingers of negative health consequences of the pandemic, such as increases in health care utilization later in the pandemic. Further study of the evolving pandemic’s effects on disease and health care utilization is needed to benefit our children now and during the next pandemic.
Dr. Antoon is an assistant professor of pediatrics at Vanderbilt University and a pediatric hospitalist at the Monroe Carroll Jr. Children’s Hospital at Vanderbilt, both in Nashville, Tenn.
References
1. Kenyon CC et al. Initial effects of the COVID-19 pandemic on pediatric asthma emergency department utilization. J Allergy Clin Immunol Pract. 2020 Sep;8(8):2774-6.e1. doi: 10.1016/j.jaip.2020.05.045.
2. Luca G et al. The impact of regular school closure on seasonal influenza epidemics: A data-driven spatial transmission model for Belgium. BMC Infect Dis. 2018;18(1):29. doi: 10.1186/s12879-017-2934-3.
3. Antoon JW et al. The COVID-19 Pandemic and changes in healthcare utilization for pediatric respiratory and nonrespiratory illnesses in the United States. J Hosp Med. 2021 Mar 8. doi: 10.12788/jhm.3608.
4. Park YJ et al. Contact tracing during coronavirus disease outbreak, South Korea, 2020. Emerg Infect Dis. 2020 Oct;26(10):2465-8. doi: 10.3201/eid2610.201315.
5. Davies NG et al. Age-dependent effects in the transmission and control of COVID-19 epidemics. Nat Med. 2020 Aug;26(8):1205-11. doi: 10.1038/s41591-020-0962-9.
6. Andrews JL et al. Peer influence in adolescence: Public health implications for COVID-19. Trends Cogn Sci. 2020;24(8):585-7. doi: 10.1016/j.tics.2020.05.001.
7. Taquechel K et al. Pediatric asthma healthcare utilization, viral testing, and air pollution changes during the COVID-19 pandemic. J Allergy Clin Immunol Pract. 2020 Nov-Dec;8(10):3378-87.e11. doi: 10.1016/j.jaip.2020.07.057.
8. Hill RM et al. Suicide ideation and attempts in a pediatric emergency department before and during COVID-19. Pediatrics. 2021;147(3):e2020029280. doi: 10.1542/peds.2020-029280.
9. Sharma S et al. COVID-19: Differences in sentinel injury and child abuse reporting during a pandemic. Child Abuse Negl. 2020 Dec;110:104709. doi: 10.1016/j.chiabu.2020.104709.
10. Lauren BN et al. Predictors of households at risk for food insecurity in the United States during the COVID-19 pandemic. Public Health Nutr. 2021 Jan 27. doi: 10.1017/S1368980021000355.
Pandemic colonoscopy restrictions may lead to worse CRC outcomes
For veterans, changes in colonoscopy screening caused by the COVID-19 pandemic may have increased risks of delayed colorectal cancer (CRC) diagnosis and could lead to worse CRC outcomes, based on data from more than 33,000 patients in the Veterans Health Administration.
After COVID-19 screening policies were implemented, a significantly lower rate of veterans with red-flag signs or symptoms for CRC underwent colonoscopy, lead author Joshua Demb, PhD, a cancer epidemiologist at the University of California, San Diego, reported at the annual Digestive Disease Week® (DDW).
“As a result of the COVID-19 pandemic, the Veterans Health Administration enacted risk mitigation and management strategies in March 2020, including postponement of nearly all colonoscopies,” the investigators reported. “Notably, this included veterans with red flag signs or symptoms for CRC, among whom delays in workup could increase risk for later-stage and fatal CRC, if present.”
To measure the effects of this policy change, Dr. Demb and colleagues performed a cohort study involving 33,804 veterans with red-flag signs or symptoms for CRC, including hematochezia, iron deficiency anemia, or abnormal guaiac fecal occult blood test or fecal immunochemical test (FIT). Veterans were divided into two cohorts based on date of first red flag diagnosis: either before the COVID-19 policy was implemented (April to October 2019; n = 19,472) or after (April to October 2020; n = 14,332), with an intervening 6-month washout period.
Primary outcomes were proportion completing colonoscopy and time to colonoscopy completion. Multivariable logistic regression incorporated a number of demographic and medical covariates, including race/ethnicity, sex, age, number of red-flag signs/symptoms, first red-flag sign/symptom, and others.
Before the COVID-19 policy change, 44% of individuals with red-flag signs or symptoms received a colonoscopy, compared with 32% after the policy was introduced (P < .01). Adjusted models showed that veterans in the COVID policy group were 42% less likely to receive a diagnostic colonoscopy than those in the prepolicy group (odds ratio, 0.58; 95% confidence interval, 0.55-0.61). While these findings showed greater likelihood of receiving a screening before the pandemic, postpolicy colonoscopies were conducted sooner, with a median time to procedure of 41 days, compared with 65 days before the pandemic (P < .01). Similar differences in screening rates between pre- and postpandemic groups were observed across all types of red flag signs and symptoms.
“Lower colonoscopy uptake was observed among individuals with red-flag signs/symptoms for CRC post- versus preimplementation of COVID-19 policies, suggesting increased future risk for delayed CRC diagnosis and adverse CRC outcomes,” the investigators concluded.
Prioritization may be needed to overcome backlog of colonoscopies
Jill Tinmouth, MD, PhD, lead scientist for ColonCancerCheck, Ontario’s organized colorectal cancer screening program, and a gastroenterologist and scientist at Sunnybrook Health Sciences Centre, Toronto, shared similar concerns about delayed diagnoses.
“We might expect these cancers to present ... at a more advanced stage, and that, as a result, the outcomes from these cancers could be worse,” Dr. Tinmouth said in an interview.
She also noted the change in colonoscopy timing.
“A particularly interesting finding was that, when a colonoscopy occurred, the time to colonoscopy was shorter during the COVID era than in the pre-COVID era,” Dr. Tinmouth said. “The authors suggested that this might be as a result of Veterans Health Administration policies implemented as a result of the pandemic that led to prioritization of more urgent procedures.”
According to Dr. Tinmouth, similar prioritization may be needed to catch up with the backlog of colonoscopies created by pandemic-related policy changes. In a recent study comparing two backlog management techniques, Dr. Tinmouth and colleagues concluded that redirecting low-yield colonoscopies to FIT without increasing hospital colonoscopy capacity could reduce time to recovery by more than half.
Even so, screening programs may be facing a long road to recovery.
“Recovery of the colonoscopy backlog is going to be a challenge that will take a while – maybe even years – to resolve,” Dr. Tinmouth said. “Jurisdictions/institutions that have a strong centralized intake or triage will likely be most successful in resolving the backlog quickly as they will be able to prioritize the most urgent cases, such as persons with an abnormal FIT or with symptoms, and to redirect persons scheduled for a ‘low-yield’ colonoscopy to have a FIT instead.” Ontario defines low-yield colonoscopies as primary screening for average-risk individuals and follow-up colonoscopies for patients with low-risk adenomas at baseline.
When asked about strategies to address future pandemics, Dr. Tinmouth said, “I think that two key learnings for me from this [pandemic] are: one, not to let our guard down, and to remain vigilant and prepared – in terms of monitoring, supply chain, equipment, etc.] ... and two to create a nimble and agile health system so that we are able to assess the challenges that the next pandemic brings and address them as quickly as possible.”The investigators and Dr. Tinmouth reported no conflicts of interest.
For veterans, changes in colonoscopy screening caused by the COVID-19 pandemic may have increased risks of delayed colorectal cancer (CRC) diagnosis and could lead to worse CRC outcomes, based on data from more than 33,000 patients in the Veterans Health Administration.
After COVID-19 screening policies were implemented, a significantly lower rate of veterans with red-flag signs or symptoms for CRC underwent colonoscopy, lead author Joshua Demb, PhD, a cancer epidemiologist at the University of California, San Diego, reported at the annual Digestive Disease Week® (DDW).
“As a result of the COVID-19 pandemic, the Veterans Health Administration enacted risk mitigation and management strategies in March 2020, including postponement of nearly all colonoscopies,” the investigators reported. “Notably, this included veterans with red flag signs or symptoms for CRC, among whom delays in workup could increase risk for later-stage and fatal CRC, if present.”
To measure the effects of this policy change, Dr. Demb and colleagues performed a cohort study involving 33,804 veterans with red-flag signs or symptoms for CRC, including hematochezia, iron deficiency anemia, or abnormal guaiac fecal occult blood test or fecal immunochemical test (FIT). Veterans were divided into two cohorts based on date of first red flag diagnosis: either before the COVID-19 policy was implemented (April to October 2019; n = 19,472) or after (April to October 2020; n = 14,332), with an intervening 6-month washout period.
Primary outcomes were proportion completing colonoscopy and time to colonoscopy completion. Multivariable logistic regression incorporated a number of demographic and medical covariates, including race/ethnicity, sex, age, number of red-flag signs/symptoms, first red-flag sign/symptom, and others.
Before the COVID-19 policy change, 44% of individuals with red-flag signs or symptoms received a colonoscopy, compared with 32% after the policy was introduced (P < .01). Adjusted models showed that veterans in the COVID policy group were 42% less likely to receive a diagnostic colonoscopy than those in the prepolicy group (odds ratio, 0.58; 95% confidence interval, 0.55-0.61). While these findings showed greater likelihood of receiving a screening before the pandemic, postpolicy colonoscopies were conducted sooner, with a median time to procedure of 41 days, compared with 65 days before the pandemic (P < .01). Similar differences in screening rates between pre- and postpandemic groups were observed across all types of red flag signs and symptoms.
“Lower colonoscopy uptake was observed among individuals with red-flag signs/symptoms for CRC post- versus preimplementation of COVID-19 policies, suggesting increased future risk for delayed CRC diagnosis and adverse CRC outcomes,” the investigators concluded.
Prioritization may be needed to overcome backlog of colonoscopies
Jill Tinmouth, MD, PhD, lead scientist for ColonCancerCheck, Ontario’s organized colorectal cancer screening program, and a gastroenterologist and scientist at Sunnybrook Health Sciences Centre, Toronto, shared similar concerns about delayed diagnoses.
“We might expect these cancers to present ... at a more advanced stage, and that, as a result, the outcomes from these cancers could be worse,” Dr. Tinmouth said in an interview.
She also noted the change in colonoscopy timing.
“A particularly interesting finding was that, when a colonoscopy occurred, the time to colonoscopy was shorter during the COVID era than in the pre-COVID era,” Dr. Tinmouth said. “The authors suggested that this might be as a result of Veterans Health Administration policies implemented as a result of the pandemic that led to prioritization of more urgent procedures.”
According to Dr. Tinmouth, similar prioritization may be needed to catch up with the backlog of colonoscopies created by pandemic-related policy changes. In a recent study comparing two backlog management techniques, Dr. Tinmouth and colleagues concluded that redirecting low-yield colonoscopies to FIT without increasing hospital colonoscopy capacity could reduce time to recovery by more than half.
Even so, screening programs may be facing a long road to recovery.
“Recovery of the colonoscopy backlog is going to be a challenge that will take a while – maybe even years – to resolve,” Dr. Tinmouth said. “Jurisdictions/institutions that have a strong centralized intake or triage will likely be most successful in resolving the backlog quickly as they will be able to prioritize the most urgent cases, such as persons with an abnormal FIT or with symptoms, and to redirect persons scheduled for a ‘low-yield’ colonoscopy to have a FIT instead.” Ontario defines low-yield colonoscopies as primary screening for average-risk individuals and follow-up colonoscopies for patients with low-risk adenomas at baseline.
When asked about strategies to address future pandemics, Dr. Tinmouth said, “I think that two key learnings for me from this [pandemic] are: one, not to let our guard down, and to remain vigilant and prepared – in terms of monitoring, supply chain, equipment, etc.] ... and two to create a nimble and agile health system so that we are able to assess the challenges that the next pandemic brings and address them as quickly as possible.”The investigators and Dr. Tinmouth reported no conflicts of interest.
For veterans, changes in colonoscopy screening caused by the COVID-19 pandemic may have increased risks of delayed colorectal cancer (CRC) diagnosis and could lead to worse CRC outcomes, based on data from more than 33,000 patients in the Veterans Health Administration.
After COVID-19 screening policies were implemented, a significantly lower rate of veterans with red-flag signs or symptoms for CRC underwent colonoscopy, lead author Joshua Demb, PhD, a cancer epidemiologist at the University of California, San Diego, reported at the annual Digestive Disease Week® (DDW).
“As a result of the COVID-19 pandemic, the Veterans Health Administration enacted risk mitigation and management strategies in March 2020, including postponement of nearly all colonoscopies,” the investigators reported. “Notably, this included veterans with red flag signs or symptoms for CRC, among whom delays in workup could increase risk for later-stage and fatal CRC, if present.”
To measure the effects of this policy change, Dr. Demb and colleagues performed a cohort study involving 33,804 veterans with red-flag signs or symptoms for CRC, including hematochezia, iron deficiency anemia, or abnormal guaiac fecal occult blood test or fecal immunochemical test (FIT). Veterans were divided into two cohorts based on date of first red flag diagnosis: either before the COVID-19 policy was implemented (April to October 2019; n = 19,472) or after (April to October 2020; n = 14,332), with an intervening 6-month washout period.
Primary outcomes were proportion completing colonoscopy and time to colonoscopy completion. Multivariable logistic regression incorporated a number of demographic and medical covariates, including race/ethnicity, sex, age, number of red-flag signs/symptoms, first red-flag sign/symptom, and others.
Before the COVID-19 policy change, 44% of individuals with red-flag signs or symptoms received a colonoscopy, compared with 32% after the policy was introduced (P < .01). Adjusted models showed that veterans in the COVID policy group were 42% less likely to receive a diagnostic colonoscopy than those in the prepolicy group (odds ratio, 0.58; 95% confidence interval, 0.55-0.61). While these findings showed greater likelihood of receiving a screening before the pandemic, postpolicy colonoscopies were conducted sooner, with a median time to procedure of 41 days, compared with 65 days before the pandemic (P < .01). Similar differences in screening rates between pre- and postpandemic groups were observed across all types of red flag signs and symptoms.
“Lower colonoscopy uptake was observed among individuals with red-flag signs/symptoms for CRC post- versus preimplementation of COVID-19 policies, suggesting increased future risk for delayed CRC diagnosis and adverse CRC outcomes,” the investigators concluded.
Prioritization may be needed to overcome backlog of colonoscopies
Jill Tinmouth, MD, PhD, lead scientist for ColonCancerCheck, Ontario’s organized colorectal cancer screening program, and a gastroenterologist and scientist at Sunnybrook Health Sciences Centre, Toronto, shared similar concerns about delayed diagnoses.
“We might expect these cancers to present ... at a more advanced stage, and that, as a result, the outcomes from these cancers could be worse,” Dr. Tinmouth said in an interview.
She also noted the change in colonoscopy timing.
“A particularly interesting finding was that, when a colonoscopy occurred, the time to colonoscopy was shorter during the COVID era than in the pre-COVID era,” Dr. Tinmouth said. “The authors suggested that this might be as a result of Veterans Health Administration policies implemented as a result of the pandemic that led to prioritization of more urgent procedures.”
According to Dr. Tinmouth, similar prioritization may be needed to catch up with the backlog of colonoscopies created by pandemic-related policy changes. In a recent study comparing two backlog management techniques, Dr. Tinmouth and colleagues concluded that redirecting low-yield colonoscopies to FIT without increasing hospital colonoscopy capacity could reduce time to recovery by more than half.
Even so, screening programs may be facing a long road to recovery.
“Recovery of the colonoscopy backlog is going to be a challenge that will take a while – maybe even years – to resolve,” Dr. Tinmouth said. “Jurisdictions/institutions that have a strong centralized intake or triage will likely be most successful in resolving the backlog quickly as they will be able to prioritize the most urgent cases, such as persons with an abnormal FIT or with symptoms, and to redirect persons scheduled for a ‘low-yield’ colonoscopy to have a FIT instead.” Ontario defines low-yield colonoscopies as primary screening for average-risk individuals and follow-up colonoscopies for patients with low-risk adenomas at baseline.
When asked about strategies to address future pandemics, Dr. Tinmouth said, “I think that two key learnings for me from this [pandemic] are: one, not to let our guard down, and to remain vigilant and prepared – in terms of monitoring, supply chain, equipment, etc.] ... and two to create a nimble and agile health system so that we are able to assess the challenges that the next pandemic brings and address them as quickly as possible.”The investigators and Dr. Tinmouth reported no conflicts of interest.
FROM DDW 2021
Lower SARS-CoV-2 vaccine responses seen in patients with immune-mediated inflammatory diseases
Ten percent of patients with immune-mediated inflammatory diseases (IMIDs) fail to respond properly to COVID-19 vaccinations regardless of medication, researchers report, and small new studies suggest those on methotrexate and rituximab may be especially vulnerable to vaccine failure.
Even so, it’s still crucially vital for patients with IMIDs to get vaccinated and for clinicians to follow recommendations to temporarily withhold certain medications around the time of vaccination, rheumatologist Anne R. Bass, MD, of Weill Cornell Medicine and the Hospital for Special Surgery, New York, said in an interview. “We’re not making any significant adjustments,” added Dr. Bass, a coauthor of the American College of Rheumatology’s COVID-19 vaccination guidelines for patients with rheumatic and musculoskeletal diseases.
The findings appear in a trio of studies in Annals of the Rheumatic Diseases. The most recent study, which appeared May 25, 2021, found that more than one-third of patients with IMIDs who took methotrexate didn’t produce adequate antibody levels after vaccination versus 10% of those in other groups. (P < .001) A May 11 study found that 20 of 30 patients with rheumatic diseases on rituximab failed to respond to vaccination. And a May 6 study reported that immune responses against SARS-CoV-2 are “somewhat delayed and reduced” in patients with IMID, with 99.5% of a control group developing neutralizing antibody activity after vaccination versus 90% of those with IMID (P = .0008).
Development of neutralizing antibodies somewhat delayed and reduced
Team members were surprised by the high number of vaccine nonresponders in the May 6 IMID study, coauthor Georg Schett, MD, of Germany’s Friedrich-Alexander University Erlangen-Nuremberg and University Hospital Erlangen, said in an interview.
The researchers compared two groups of patients who had no history of COVID-19 and received COVID-19 vaccinations, mostly two shots of the Pfizer-BioNTech vaccine (96%): 84 with IMID (mean age, 53.1 years; 65.5% females) and 182 healthy controls (mean age, 40.8 years; 57.1% females).
The patients with IMID most commonly had spondyloarthritis (32.1%), RA (29.8%), inflammatory bowel disease (9.5%), and psoriasis (9.5%). Nearly 43% of the patients were treated with biologic and targeted synthetic disease-modifying antirheumatic drugs and 23.9% with conventional synthetic DMARDSs. Another 29% were not treated.
All of the controls developed anti–SARS-CoV-2 IgG, but 6% of the patients with IMID did not (P = .003). The gap in development of neutralizing antibodies was even higher: 99.5% of the controls developed neutralizing antibody activity versus 90% of the IMID group. “Neutralizing antibodies are more relevant because the test shows how much the antibodies interfere with the binding of SARS-CoV-2 proteins to the receptor,” Dr. Schett said.
The study authors concluded that “our study provides evidence that, while vaccination against SARS-CoV-2 is well tolerated and even associated with lower incidence of side effects in patients with IMID, its efficacy is somewhat delayed and reduced. Nonetheless, the data also show that, in principle, patients with IMID respond to SARS-CoV-2 vaccination, supporting an aggressive vaccination strategy.”
Lowered antibody response to vaccination for some methotrexate users
In the newer study, led by Rebecca H. Haberman, MD, of New York University Langone Health, researchers examined COVID-19 vaccine response in cohorts in New York City and Erlangen, Germany.
The New York cohort included 25 patients with IMID who were taking methotrexate by itself or with other immunomodulatory medications (mean age, 63.2 years), 26 with IMID who were on anticytokine therapy and/or other oral immunomodulators (mean age, 49.1 years) and 26 healthy controls (mean age, 49.2 years). Most patients with IMID had psoriasis/psoriatic arthritis or RA.
The German validation cohort included 182 healthy subjects (mean age, 45.0 years), 11 subjects with IMID who received TNF inhibitor monotherapy (mean age, 40.8 years), and 20 subjects with IMID on methotrexate monotherapy (mean age, 54.5 years).
In the New York cohort, 96.1% of healthy controls showed “adequate humoral immune response,” along with 92.3% of patients with IMID who weren’t taking methotrexate. However, those on methotrexate had a lower rate of adequate response (72.0%), and the gap persisted even after researchers removed those who showed signs of previous COVID-19 infection (P = .045).
In the German cohort, 98.3% of healthy cohorts and 90.9% of patients with IMID who didn’t receive methotrexate reached an “adequate” humoral response versus just half (50.0%) of those who were taking methotrexate.
When both cohorts are combined, over 90% of the healthy subjects and the patients with IMID on biologic treatments (mainly TNF blockers, n = 37) showed “robust” antibody response. However, only 62% of patients with IMID who took methotrexate (n = 45) reached an “adequate” level of response. The methotrexate gap remained after researchers accounted for differences in age among the cohorts.
What’s going on? “We think that the underlying chronic immune stimulation in autoimmune patients may cause T-cell exhaustion and thus blunts the immune response,” said Dr. Schett, who’s also a coauthor of this study. “In addition, specific drugs such as methotrexate could additionally impair the immune response.”
Still, the findings “reiterate that vaccinations are safe and effective, which is what the recommendations state,” he said, adding that more testing of vaccination immune response is wise.
Insights into vaccine response while on rituximab
Two more reports, also published in Annals of the Rheumatic Diseases, offer insight into vaccine response in patients with IMID who take rituximab.
In one report, published May 11, U.S. researchers retrospectively tracked 89 rheumatic disease patients (76% female; mean age, 61) at a single clinic who’d received at least one dose of a COVID-19 vaccine. Of those, 21 patients showed no sign of vaccine antibody response, and 20 of them were in the group taking rituximab. (The other patient was taking belimumab.) Another 10 patients taking rituximab did show a response.
“Longer duration from most recent rituximab exposure was associated with a greater likelihood of response,” the report’s authors wrote. “The results suggest that time from last rituximab exposure is an important consideration in maximizing the likelihood of a serological response, but this likely is related to the substantial variation in the period of B-cell depletion following rituximab.”
Finally, an Austrian report published May 6 examined COVID-19 vaccine immune response in five patients who were taking rituximab (four with other drugs such as methotrexate and prednisone). Researchers compared them with eight healthy controls, half who’d been vaccinated.
The researchers found evidence that rituximab “may not have to preclude SARS-CoV-2 vaccination, since a cellular immune response will be mounted even in the absence of circulating B cells. Alternatively, in patients with stable disease, delaying [rituximab] treatment until after the second vaccination may be warranted and, therefore, vaccines with a short interval between first and second vaccination or those showing full protection after a single vaccination may be preferable. Importantly, in the presence of circulating B cells also a humoral immune response may be expected despite prior [rituximab] therapy.”
Dr. Bass said the findings reflect growing awareness that “patients with autoimmune disease, especially when they’re on immunosuppressant medications, don’t quite have as optimal responses to the vaccinations.” However, she said, the vaccines are so potent that they’re likely to still have significant efficacy in these patients even if there’s a reduction in response.
What’s next? Dr. Schett said “testing immune response to vaccination is important for patients with autoimmune disease. Some of them may need a third vaccination.”
The American College of Rheumatology’s COVID-19 vaccination guidelines do not recommend third vaccinations or postvaccination immune testing at this time. However, Dr. Bass, one of the coauthors of the recommendations, said it’s likely that postvaccination immune testing and booster shots will become routine.
Dr. Bass reported no relevant disclosures. Dr. Schett reported receiving consulting fees from AbbVie. The May 6 German vaccine study was funded by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung und Forschung, the ERC Synergy grant 4D Nanoscope, the IMI funded project RTCure, the Emerging Fields Initiative MIRACLE of the Friedrich-Alexander-Universität Erlangen-Nürnberg, the Schreiber Stiftung, and the Else Kröner-Memorial Scholarship. The study authors reported no disclosures. The May 25 study of German and American cohorts was funded by the National Institute of Arthritis and Musculoskletal and Skin Diseases, National Institute of Allergy and Infectious Diseases, Rheumatology Research Foundation, Bloomberg Philanthropies COVID-19 Initiative, Pfizer COVID-19 Competitive Grant Program, Beatrice Snyder Foundation, Riley Family Foundation, National Psoriasis Foundation, and Deutsche Forschungsgemeinschaft. The authors reported a range of financial relationships with pharmaceutical companies. No specific funding was reported for the other two studies mentioned.
Ten percent of patients with immune-mediated inflammatory diseases (IMIDs) fail to respond properly to COVID-19 vaccinations regardless of medication, researchers report, and small new studies suggest those on methotrexate and rituximab may be especially vulnerable to vaccine failure.
Even so, it’s still crucially vital for patients with IMIDs to get vaccinated and for clinicians to follow recommendations to temporarily withhold certain medications around the time of vaccination, rheumatologist Anne R. Bass, MD, of Weill Cornell Medicine and the Hospital for Special Surgery, New York, said in an interview. “We’re not making any significant adjustments,” added Dr. Bass, a coauthor of the American College of Rheumatology’s COVID-19 vaccination guidelines for patients with rheumatic and musculoskeletal diseases.
The findings appear in a trio of studies in Annals of the Rheumatic Diseases. The most recent study, which appeared May 25, 2021, found that more than one-third of patients with IMIDs who took methotrexate didn’t produce adequate antibody levels after vaccination versus 10% of those in other groups. (P < .001) A May 11 study found that 20 of 30 patients with rheumatic diseases on rituximab failed to respond to vaccination. And a May 6 study reported that immune responses against SARS-CoV-2 are “somewhat delayed and reduced” in patients with IMID, with 99.5% of a control group developing neutralizing antibody activity after vaccination versus 90% of those with IMID (P = .0008).
Development of neutralizing antibodies somewhat delayed and reduced
Team members were surprised by the high number of vaccine nonresponders in the May 6 IMID study, coauthor Georg Schett, MD, of Germany’s Friedrich-Alexander University Erlangen-Nuremberg and University Hospital Erlangen, said in an interview.
The researchers compared two groups of patients who had no history of COVID-19 and received COVID-19 vaccinations, mostly two shots of the Pfizer-BioNTech vaccine (96%): 84 with IMID (mean age, 53.1 years; 65.5% females) and 182 healthy controls (mean age, 40.8 years; 57.1% females).
The patients with IMID most commonly had spondyloarthritis (32.1%), RA (29.8%), inflammatory bowel disease (9.5%), and psoriasis (9.5%). Nearly 43% of the patients were treated with biologic and targeted synthetic disease-modifying antirheumatic drugs and 23.9% with conventional synthetic DMARDSs. Another 29% were not treated.
All of the controls developed anti–SARS-CoV-2 IgG, but 6% of the patients with IMID did not (P = .003). The gap in development of neutralizing antibodies was even higher: 99.5% of the controls developed neutralizing antibody activity versus 90% of the IMID group. “Neutralizing antibodies are more relevant because the test shows how much the antibodies interfere with the binding of SARS-CoV-2 proteins to the receptor,” Dr. Schett said.
The study authors concluded that “our study provides evidence that, while vaccination against SARS-CoV-2 is well tolerated and even associated with lower incidence of side effects in patients with IMID, its efficacy is somewhat delayed and reduced. Nonetheless, the data also show that, in principle, patients with IMID respond to SARS-CoV-2 vaccination, supporting an aggressive vaccination strategy.”
Lowered antibody response to vaccination for some methotrexate users
In the newer study, led by Rebecca H. Haberman, MD, of New York University Langone Health, researchers examined COVID-19 vaccine response in cohorts in New York City and Erlangen, Germany.
The New York cohort included 25 patients with IMID who were taking methotrexate by itself or with other immunomodulatory medications (mean age, 63.2 years), 26 with IMID who were on anticytokine therapy and/or other oral immunomodulators (mean age, 49.1 years) and 26 healthy controls (mean age, 49.2 years). Most patients with IMID had psoriasis/psoriatic arthritis or RA.
The German validation cohort included 182 healthy subjects (mean age, 45.0 years), 11 subjects with IMID who received TNF inhibitor monotherapy (mean age, 40.8 years), and 20 subjects with IMID on methotrexate monotherapy (mean age, 54.5 years).
In the New York cohort, 96.1% of healthy controls showed “adequate humoral immune response,” along with 92.3% of patients with IMID who weren’t taking methotrexate. However, those on methotrexate had a lower rate of adequate response (72.0%), and the gap persisted even after researchers removed those who showed signs of previous COVID-19 infection (P = .045).
In the German cohort, 98.3% of healthy cohorts and 90.9% of patients with IMID who didn’t receive methotrexate reached an “adequate” humoral response versus just half (50.0%) of those who were taking methotrexate.
When both cohorts are combined, over 90% of the healthy subjects and the patients with IMID on biologic treatments (mainly TNF blockers, n = 37) showed “robust” antibody response. However, only 62% of patients with IMID who took methotrexate (n = 45) reached an “adequate” level of response. The methotrexate gap remained after researchers accounted for differences in age among the cohorts.
What’s going on? “We think that the underlying chronic immune stimulation in autoimmune patients may cause T-cell exhaustion and thus blunts the immune response,” said Dr. Schett, who’s also a coauthor of this study. “In addition, specific drugs such as methotrexate could additionally impair the immune response.”
Still, the findings “reiterate that vaccinations are safe and effective, which is what the recommendations state,” he said, adding that more testing of vaccination immune response is wise.
Insights into vaccine response while on rituximab
Two more reports, also published in Annals of the Rheumatic Diseases, offer insight into vaccine response in patients with IMID who take rituximab.
In one report, published May 11, U.S. researchers retrospectively tracked 89 rheumatic disease patients (76% female; mean age, 61) at a single clinic who’d received at least one dose of a COVID-19 vaccine. Of those, 21 patients showed no sign of vaccine antibody response, and 20 of them were in the group taking rituximab. (The other patient was taking belimumab.) Another 10 patients taking rituximab did show a response.
“Longer duration from most recent rituximab exposure was associated with a greater likelihood of response,” the report’s authors wrote. “The results suggest that time from last rituximab exposure is an important consideration in maximizing the likelihood of a serological response, but this likely is related to the substantial variation in the period of B-cell depletion following rituximab.”
Finally, an Austrian report published May 6 examined COVID-19 vaccine immune response in five patients who were taking rituximab (four with other drugs such as methotrexate and prednisone). Researchers compared them with eight healthy controls, half who’d been vaccinated.
The researchers found evidence that rituximab “may not have to preclude SARS-CoV-2 vaccination, since a cellular immune response will be mounted even in the absence of circulating B cells. Alternatively, in patients with stable disease, delaying [rituximab] treatment until after the second vaccination may be warranted and, therefore, vaccines with a short interval between first and second vaccination or those showing full protection after a single vaccination may be preferable. Importantly, in the presence of circulating B cells also a humoral immune response may be expected despite prior [rituximab] therapy.”
Dr. Bass said the findings reflect growing awareness that “patients with autoimmune disease, especially when they’re on immunosuppressant medications, don’t quite have as optimal responses to the vaccinations.” However, she said, the vaccines are so potent that they’re likely to still have significant efficacy in these patients even if there’s a reduction in response.
What’s next? Dr. Schett said “testing immune response to vaccination is important for patients with autoimmune disease. Some of them may need a third vaccination.”
The American College of Rheumatology’s COVID-19 vaccination guidelines do not recommend third vaccinations or postvaccination immune testing at this time. However, Dr. Bass, one of the coauthors of the recommendations, said it’s likely that postvaccination immune testing and booster shots will become routine.
Dr. Bass reported no relevant disclosures. Dr. Schett reported receiving consulting fees from AbbVie. The May 6 German vaccine study was funded by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung und Forschung, the ERC Synergy grant 4D Nanoscope, the IMI funded project RTCure, the Emerging Fields Initiative MIRACLE of the Friedrich-Alexander-Universität Erlangen-Nürnberg, the Schreiber Stiftung, and the Else Kröner-Memorial Scholarship. The study authors reported no disclosures. The May 25 study of German and American cohorts was funded by the National Institute of Arthritis and Musculoskletal and Skin Diseases, National Institute of Allergy and Infectious Diseases, Rheumatology Research Foundation, Bloomberg Philanthropies COVID-19 Initiative, Pfizer COVID-19 Competitive Grant Program, Beatrice Snyder Foundation, Riley Family Foundation, National Psoriasis Foundation, and Deutsche Forschungsgemeinschaft. The authors reported a range of financial relationships with pharmaceutical companies. No specific funding was reported for the other two studies mentioned.
Ten percent of patients with immune-mediated inflammatory diseases (IMIDs) fail to respond properly to COVID-19 vaccinations regardless of medication, researchers report, and small new studies suggest those on methotrexate and rituximab may be especially vulnerable to vaccine failure.
Even so, it’s still crucially vital for patients with IMIDs to get vaccinated and for clinicians to follow recommendations to temporarily withhold certain medications around the time of vaccination, rheumatologist Anne R. Bass, MD, of Weill Cornell Medicine and the Hospital for Special Surgery, New York, said in an interview. “We’re not making any significant adjustments,” added Dr. Bass, a coauthor of the American College of Rheumatology’s COVID-19 vaccination guidelines for patients with rheumatic and musculoskeletal diseases.
The findings appear in a trio of studies in Annals of the Rheumatic Diseases. The most recent study, which appeared May 25, 2021, found that more than one-third of patients with IMIDs who took methotrexate didn’t produce adequate antibody levels after vaccination versus 10% of those in other groups. (P < .001) A May 11 study found that 20 of 30 patients with rheumatic diseases on rituximab failed to respond to vaccination. And a May 6 study reported that immune responses against SARS-CoV-2 are “somewhat delayed and reduced” in patients with IMID, with 99.5% of a control group developing neutralizing antibody activity after vaccination versus 90% of those with IMID (P = .0008).
Development of neutralizing antibodies somewhat delayed and reduced
Team members were surprised by the high number of vaccine nonresponders in the May 6 IMID study, coauthor Georg Schett, MD, of Germany’s Friedrich-Alexander University Erlangen-Nuremberg and University Hospital Erlangen, said in an interview.
The researchers compared two groups of patients who had no history of COVID-19 and received COVID-19 vaccinations, mostly two shots of the Pfizer-BioNTech vaccine (96%): 84 with IMID (mean age, 53.1 years; 65.5% females) and 182 healthy controls (mean age, 40.8 years; 57.1% females).
The patients with IMID most commonly had spondyloarthritis (32.1%), RA (29.8%), inflammatory bowel disease (9.5%), and psoriasis (9.5%). Nearly 43% of the patients were treated with biologic and targeted synthetic disease-modifying antirheumatic drugs and 23.9% with conventional synthetic DMARDSs. Another 29% were not treated.
All of the controls developed anti–SARS-CoV-2 IgG, but 6% of the patients with IMID did not (P = .003). The gap in development of neutralizing antibodies was even higher: 99.5% of the controls developed neutralizing antibody activity versus 90% of the IMID group. “Neutralizing antibodies are more relevant because the test shows how much the antibodies interfere with the binding of SARS-CoV-2 proteins to the receptor,” Dr. Schett said.
The study authors concluded that “our study provides evidence that, while vaccination against SARS-CoV-2 is well tolerated and even associated with lower incidence of side effects in patients with IMID, its efficacy is somewhat delayed and reduced. Nonetheless, the data also show that, in principle, patients with IMID respond to SARS-CoV-2 vaccination, supporting an aggressive vaccination strategy.”
Lowered antibody response to vaccination for some methotrexate users
In the newer study, led by Rebecca H. Haberman, MD, of New York University Langone Health, researchers examined COVID-19 vaccine response in cohorts in New York City and Erlangen, Germany.
The New York cohort included 25 patients with IMID who were taking methotrexate by itself or with other immunomodulatory medications (mean age, 63.2 years), 26 with IMID who were on anticytokine therapy and/or other oral immunomodulators (mean age, 49.1 years) and 26 healthy controls (mean age, 49.2 years). Most patients with IMID had psoriasis/psoriatic arthritis or RA.
The German validation cohort included 182 healthy subjects (mean age, 45.0 years), 11 subjects with IMID who received TNF inhibitor monotherapy (mean age, 40.8 years), and 20 subjects with IMID on methotrexate monotherapy (mean age, 54.5 years).
In the New York cohort, 96.1% of healthy controls showed “adequate humoral immune response,” along with 92.3% of patients with IMID who weren’t taking methotrexate. However, those on methotrexate had a lower rate of adequate response (72.0%), and the gap persisted even after researchers removed those who showed signs of previous COVID-19 infection (P = .045).
In the German cohort, 98.3% of healthy cohorts and 90.9% of patients with IMID who didn’t receive methotrexate reached an “adequate” humoral response versus just half (50.0%) of those who were taking methotrexate.
When both cohorts are combined, over 90% of the healthy subjects and the patients with IMID on biologic treatments (mainly TNF blockers, n = 37) showed “robust” antibody response. However, only 62% of patients with IMID who took methotrexate (n = 45) reached an “adequate” level of response. The methotrexate gap remained after researchers accounted for differences in age among the cohorts.
What’s going on? “We think that the underlying chronic immune stimulation in autoimmune patients may cause T-cell exhaustion and thus blunts the immune response,” said Dr. Schett, who’s also a coauthor of this study. “In addition, specific drugs such as methotrexate could additionally impair the immune response.”
Still, the findings “reiterate that vaccinations are safe and effective, which is what the recommendations state,” he said, adding that more testing of vaccination immune response is wise.
Insights into vaccine response while on rituximab
Two more reports, also published in Annals of the Rheumatic Diseases, offer insight into vaccine response in patients with IMID who take rituximab.
In one report, published May 11, U.S. researchers retrospectively tracked 89 rheumatic disease patients (76% female; mean age, 61) at a single clinic who’d received at least one dose of a COVID-19 vaccine. Of those, 21 patients showed no sign of vaccine antibody response, and 20 of them were in the group taking rituximab. (The other patient was taking belimumab.) Another 10 patients taking rituximab did show a response.
“Longer duration from most recent rituximab exposure was associated with a greater likelihood of response,” the report’s authors wrote. “The results suggest that time from last rituximab exposure is an important consideration in maximizing the likelihood of a serological response, but this likely is related to the substantial variation in the period of B-cell depletion following rituximab.”
Finally, an Austrian report published May 6 examined COVID-19 vaccine immune response in five patients who were taking rituximab (four with other drugs such as methotrexate and prednisone). Researchers compared them with eight healthy controls, half who’d been vaccinated.
The researchers found evidence that rituximab “may not have to preclude SARS-CoV-2 vaccination, since a cellular immune response will be mounted even in the absence of circulating B cells. Alternatively, in patients with stable disease, delaying [rituximab] treatment until after the second vaccination may be warranted and, therefore, vaccines with a short interval between first and second vaccination or those showing full protection after a single vaccination may be preferable. Importantly, in the presence of circulating B cells also a humoral immune response may be expected despite prior [rituximab] therapy.”
Dr. Bass said the findings reflect growing awareness that “patients with autoimmune disease, especially when they’re on immunosuppressant medications, don’t quite have as optimal responses to the vaccinations.” However, she said, the vaccines are so potent that they’re likely to still have significant efficacy in these patients even if there’s a reduction in response.
What’s next? Dr. Schett said “testing immune response to vaccination is important for patients with autoimmune disease. Some of them may need a third vaccination.”
The American College of Rheumatology’s COVID-19 vaccination guidelines do not recommend third vaccinations or postvaccination immune testing at this time. However, Dr. Bass, one of the coauthors of the recommendations, said it’s likely that postvaccination immune testing and booster shots will become routine.
Dr. Bass reported no relevant disclosures. Dr. Schett reported receiving consulting fees from AbbVie. The May 6 German vaccine study was funded by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung und Forschung, the ERC Synergy grant 4D Nanoscope, the IMI funded project RTCure, the Emerging Fields Initiative MIRACLE of the Friedrich-Alexander-Universität Erlangen-Nürnberg, the Schreiber Stiftung, and the Else Kröner-Memorial Scholarship. The study authors reported no disclosures. The May 25 study of German and American cohorts was funded by the National Institute of Arthritis and Musculoskletal and Skin Diseases, National Institute of Allergy and Infectious Diseases, Rheumatology Research Foundation, Bloomberg Philanthropies COVID-19 Initiative, Pfizer COVID-19 Competitive Grant Program, Beatrice Snyder Foundation, Riley Family Foundation, National Psoriasis Foundation, and Deutsche Forschungsgemeinschaft. The authors reported a range of financial relationships with pharmaceutical companies. No specific funding was reported for the other two studies mentioned.
FROM ANNALS OF THE RHEUMATIC DISEASES
COVID-19 vaccination rate rising quickly among adolescents
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.
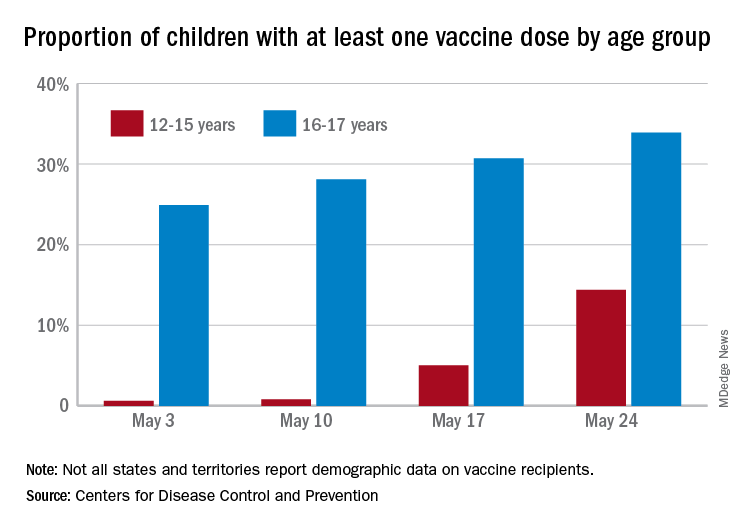
As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.

As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
With nearly half of all Americans having received at least one dose of a COVID-19 vaccine, the youngest eligible group is beginning to overcome its late start, according to data from the Centers for Disease Control and Prevention.

As of May 24, 49.4% of the U.S. population – that’s almost 164 million people – has received at least one dose of vaccine. The corresponding figure for children aged 12-15 years is 14.4%, but that’s up from only 0.6% just 3 weeks before. Among children aged 16-17, who’ve been getting vaccinated since early April in some states, the proportion receiving at least one dose went from 24.9% to 33.9% over those same 3 weeks, the CDC said on its COVID Data Tracker site.
The comparatively rapid increase among the younger group of eligible children can be seen over the last 14 days. To put that into perspective, only those aged 25-39 years were higher at 21.9%, while 18-24 (12.1%), 40-49 (13.4%), 50-64 (18.2%), 65-74 (5.3%), and ≥75 (2.9%) were all lower.
The 12- to 15-year-olds are further behind when it comes to full vaccination status, however, with just 0.6% having received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 21.6% for those aged 16-17 years. Children aged 12-15 make up 5% of the total U.S. population but just 0.1% of all those who have been fully vaccinated versus 2.5% and 1.4%, respectively, for those aged 16-17, the CDC reported.
COVID-19 vaccination and pregnancy: Benefits outweigh the risks, for now
Vaccines have been a lifesaving public health measure since 1000 CE, when the Chinese first used smallpox inoculations to induce immunity.1 Work by pioneers such as Edward Jenner, Louis Pasteur, and Maurice Hilleman has averted countless millions of vaccine-preventable illnesses and deaths, and vaccines have become a routine part of health maintenance throughout the human life cycle.
Pregnant patients who receive vaccines often have an added benefit of protection provided to their infants through passive transfer of antibodies. Several vaccine platforms have been utilized in pregnancy with well-documented improvements in maternal and obstetric outcomes as well as improved neonatal outcomes in the first several months of life.
Risks of COVID-19 in pregnancy
The COVID-19 pandemic placed a spotlight on medically at-risk groups. Pregnant women are 3 times more likely to require admission to the intensive care unit, have increased requirement for extracorporeal membrane oxygenation treatment, and are up to 70% more likely to die than nonpregnant peers—and this risk increases with the presence of additional comorbidities.
In the case of COVID-19, vaccination trials that have shaped worldwide clinical practice unfortunately followed the historical trend of excluding pregnant patients from participation. This has required clinicians to guide their patients through the decision of whether or not to accept vaccination without having the same reassurances regarding safety and effectiveness afforded to their nonpregnant counterparts. With more than 86,000 pregnant women infected with COVID-19 through April 19, 2021, this lack of information regarding vaccine safety in pregnancy is a significant public health gap.2
COVID-19 vaccines
The current COVID-19 vaccines approved for use in the United States under an Emergency Use Authorization issued by the US Food and Drug Administration are nonreplicating and thus cannot cause infection in the mother or fetus. These are the Pfizer-BioNTech mRNA vaccine, the Moderna mRNA-1273 vaccine, and the Janssen Biotech Inc. monovalent vaccine. Furthermore, in animal studies that included the Pfizer-BioNTech, Moderna, or Janssen COVID-19 vaccines, no fetal, embryonal, female reproductive, or postnatal development safety concerns were demonstrated.
As of April 19, 2021, 94,335 pregnant women had received a COVID-19 vaccination, and 4,622 of these enrolled in the Centers for Disease Control and Prevention’s (CDC) V-safe Vaccine Pregnancy Registry.3 The data reported noted no unexpected pregnancy or infant outcomes related to COVID-19 vaccination in pregnancy. Adverse effects of the vaccine were similar to those in nonpregnant cohorts. Additionally, emerging data suggest passage of immunity to neonates, with maternal antibodies demonstrated in cord blood at time of delivery as well as in breast milk.4 To date, these data mainly have come from women immunized with the Moderna and Pfizer-BioNTech mRNA vaccines.
Counseling pregnant patients
Our counseling aligns with that of the American College of Obstetricians and Gynecologists, the Society for Maternal-Fetal Medicine, and the CDC’s Advisory Committee on Immunization Practices in that COVID-19 vaccination should not be withheld from pregnant patients or patients who want to become pregnant. In pregnant patients with comorbidities that place them at higher risk for severe COVID-19 infection, all available formulations of the COVID-19 vaccination should be strongly considered.
As evidence for vaccination safety continues to emerge, patients should continue to discuss their individual needs for vaccination in a shared decision-making format with their obstetric providers.
Boylston A. The origins of inoculation. J R Soc Med. 2012;105:309-313.
Centers for Disease Control and Prevention. COVID data tracker. Data on COVID-19 during pregnancy: severity of maternal illness. https://covid.cdc.gov/covid-data-tracker/#pregnant-population. Accessed April 19, 2021.
Centers for Disease Control and Prevention. V-safe COVID-19 Vaccine Pregnancy Registry. https://www.cdc.gov/coronavirus/2019- ncov/vaccines/safety/vsafepregnancyregistry.html. Updated May 3, 2021. Accessed April 19, 2021.
Gray KJ, Bordt EA, Atyeo C, et al. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. 2021;S0002-9378(21)00187-3. doi: 10.1016/j.ajog.2021.03.023
Vaccines have been a lifesaving public health measure since 1000 CE, when the Chinese first used smallpox inoculations to induce immunity.1 Work by pioneers such as Edward Jenner, Louis Pasteur, and Maurice Hilleman has averted countless millions of vaccine-preventable illnesses and deaths, and vaccines have become a routine part of health maintenance throughout the human life cycle.
Pregnant patients who receive vaccines often have an added benefit of protection provided to their infants through passive transfer of antibodies. Several vaccine platforms have been utilized in pregnancy with well-documented improvements in maternal and obstetric outcomes as well as improved neonatal outcomes in the first several months of life.
Risks of COVID-19 in pregnancy
The COVID-19 pandemic placed a spotlight on medically at-risk groups. Pregnant women are 3 times more likely to require admission to the intensive care unit, have increased requirement for extracorporeal membrane oxygenation treatment, and are up to 70% more likely to die than nonpregnant peers—and this risk increases with the presence of additional comorbidities.
In the case of COVID-19, vaccination trials that have shaped worldwide clinical practice unfortunately followed the historical trend of excluding pregnant patients from participation. This has required clinicians to guide their patients through the decision of whether or not to accept vaccination without having the same reassurances regarding safety and effectiveness afforded to their nonpregnant counterparts. With more than 86,000 pregnant women infected with COVID-19 through April 19, 2021, this lack of information regarding vaccine safety in pregnancy is a significant public health gap.2
COVID-19 vaccines
The current COVID-19 vaccines approved for use in the United States under an Emergency Use Authorization issued by the US Food and Drug Administration are nonreplicating and thus cannot cause infection in the mother or fetus. These are the Pfizer-BioNTech mRNA vaccine, the Moderna mRNA-1273 vaccine, and the Janssen Biotech Inc. monovalent vaccine. Furthermore, in animal studies that included the Pfizer-BioNTech, Moderna, or Janssen COVID-19 vaccines, no fetal, embryonal, female reproductive, or postnatal development safety concerns were demonstrated.
As of April 19, 2021, 94,335 pregnant women had received a COVID-19 vaccination, and 4,622 of these enrolled in the Centers for Disease Control and Prevention’s (CDC) V-safe Vaccine Pregnancy Registry.3 The data reported noted no unexpected pregnancy or infant outcomes related to COVID-19 vaccination in pregnancy. Adverse effects of the vaccine were similar to those in nonpregnant cohorts. Additionally, emerging data suggest passage of immunity to neonates, with maternal antibodies demonstrated in cord blood at time of delivery as well as in breast milk.4 To date, these data mainly have come from women immunized with the Moderna and Pfizer-BioNTech mRNA vaccines.
Counseling pregnant patients
Our counseling aligns with that of the American College of Obstetricians and Gynecologists, the Society for Maternal-Fetal Medicine, and the CDC’s Advisory Committee on Immunization Practices in that COVID-19 vaccination should not be withheld from pregnant patients or patients who want to become pregnant. In pregnant patients with comorbidities that place them at higher risk for severe COVID-19 infection, all available formulations of the COVID-19 vaccination should be strongly considered.
As evidence for vaccination safety continues to emerge, patients should continue to discuss their individual needs for vaccination in a shared decision-making format with their obstetric providers.
Vaccines have been a lifesaving public health measure since 1000 CE, when the Chinese first used smallpox inoculations to induce immunity.1 Work by pioneers such as Edward Jenner, Louis Pasteur, and Maurice Hilleman has averted countless millions of vaccine-preventable illnesses and deaths, and vaccines have become a routine part of health maintenance throughout the human life cycle.
Pregnant patients who receive vaccines often have an added benefit of protection provided to their infants through passive transfer of antibodies. Several vaccine platforms have been utilized in pregnancy with well-documented improvements in maternal and obstetric outcomes as well as improved neonatal outcomes in the first several months of life.
Risks of COVID-19 in pregnancy
The COVID-19 pandemic placed a spotlight on medically at-risk groups. Pregnant women are 3 times more likely to require admission to the intensive care unit, have increased requirement for extracorporeal membrane oxygenation treatment, and are up to 70% more likely to die than nonpregnant peers—and this risk increases with the presence of additional comorbidities.
In the case of COVID-19, vaccination trials that have shaped worldwide clinical practice unfortunately followed the historical trend of excluding pregnant patients from participation. This has required clinicians to guide their patients through the decision of whether or not to accept vaccination without having the same reassurances regarding safety and effectiveness afforded to their nonpregnant counterparts. With more than 86,000 pregnant women infected with COVID-19 through April 19, 2021, this lack of information regarding vaccine safety in pregnancy is a significant public health gap.2
COVID-19 vaccines
The current COVID-19 vaccines approved for use in the United States under an Emergency Use Authorization issued by the US Food and Drug Administration are nonreplicating and thus cannot cause infection in the mother or fetus. These are the Pfizer-BioNTech mRNA vaccine, the Moderna mRNA-1273 vaccine, and the Janssen Biotech Inc. monovalent vaccine. Furthermore, in animal studies that included the Pfizer-BioNTech, Moderna, or Janssen COVID-19 vaccines, no fetal, embryonal, female reproductive, or postnatal development safety concerns were demonstrated.
As of April 19, 2021, 94,335 pregnant women had received a COVID-19 vaccination, and 4,622 of these enrolled in the Centers for Disease Control and Prevention’s (CDC) V-safe Vaccine Pregnancy Registry.3 The data reported noted no unexpected pregnancy or infant outcomes related to COVID-19 vaccination in pregnancy. Adverse effects of the vaccine were similar to those in nonpregnant cohorts. Additionally, emerging data suggest passage of immunity to neonates, with maternal antibodies demonstrated in cord blood at time of delivery as well as in breast milk.4 To date, these data mainly have come from women immunized with the Moderna and Pfizer-BioNTech mRNA vaccines.
Counseling pregnant patients
Our counseling aligns with that of the American College of Obstetricians and Gynecologists, the Society for Maternal-Fetal Medicine, and the CDC’s Advisory Committee on Immunization Practices in that COVID-19 vaccination should not be withheld from pregnant patients or patients who want to become pregnant. In pregnant patients with comorbidities that place them at higher risk for severe COVID-19 infection, all available formulations of the COVID-19 vaccination should be strongly considered.
As evidence for vaccination safety continues to emerge, patients should continue to discuss their individual needs for vaccination in a shared decision-making format with their obstetric providers.
Boylston A. The origins of inoculation. J R Soc Med. 2012;105:309-313.
Centers for Disease Control and Prevention. COVID data tracker. Data on COVID-19 during pregnancy: severity of maternal illness. https://covid.cdc.gov/covid-data-tracker/#pregnant-population. Accessed April 19, 2021.
Centers for Disease Control and Prevention. V-safe COVID-19 Vaccine Pregnancy Registry. https://www.cdc.gov/coronavirus/2019- ncov/vaccines/safety/vsafepregnancyregistry.html. Updated May 3, 2021. Accessed April 19, 2021.
Gray KJ, Bordt EA, Atyeo C, et al. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. 2021;S0002-9378(21)00187-3. doi: 10.1016/j.ajog.2021.03.023
Boylston A. The origins of inoculation. J R Soc Med. 2012;105:309-313.
Centers for Disease Control and Prevention. COVID data tracker. Data on COVID-19 during pregnancy: severity of maternal illness. https://covid.cdc.gov/covid-data-tracker/#pregnant-population. Accessed April 19, 2021.
Centers for Disease Control and Prevention. V-safe COVID-19 Vaccine Pregnancy Registry. https://www.cdc.gov/coronavirus/2019- ncov/vaccines/safety/vsafepregnancyregistry.html. Updated May 3, 2021. Accessed April 19, 2021.
Gray KJ, Bordt EA, Atyeo C, et al. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. 2021;S0002-9378(21)00187-3. doi: 10.1016/j.ajog.2021.03.023
Dengue may double the risk of symptomatic COVID-19
In a surprising study from the Amazon rain forest, Brazilian scientists found that symptomatic COVID-19 infections were twice as likely to occur in people who had prior dengue.
The study, led by Marcelo Urbano Ferreira, MD, PhD, of the University of São Paulo Biomedical Sciences Institute, was conducted in Mâncio Lima, a town in the Amazon region of Brazil, and published May 6, 2021, in Clinical Infectious Diseases.
In the study, supported by the São Paulo Research Foundation, Ferreira’s team looked at sequential blood samples from 1,285 residents of Mâncio Lima.
An earlier study by Miguel Nicolelis, MD, PhD, and colleagues (published as a preprint) had analyzed data from the first COVID-19 wave in Brazil in 2020. It was an “ecological study” and examined dengue cases in different geographic regions of Brazil. That study concluded that dengue actually seemed to protect people from later developing COVID-19.
Dr. Ferreira anticipated finding a similar effect. Instead, he found the opposite effect. Although dengue did not increase the risk of subsequent COVID-19 infection, symptomatic COVID-19 became twice as likely in people with prior dengue. His study was longitudinal, following a single group of patients in Mâncio Lima over time.
Dr. Ferreira explained that ecological studies are inherently less accurate, as they look at populations in different places. “All the older cases are diagnosed on clinical grounds ... Because most infections are either asymptomatic or symptoms can be easily confused with” other diseases, many cases are missed. So, during the dengue transmission season, “We have some overestimation of the actual number of cases, and outside the transmission season, we have underestimation of the cases.”
On the apparent discrepancy with the earlier study by Dr. Nicolelis, Dr. Ferreira commented, “It’s a wonderful study because it’s something you can do quickly and test a hypothesis [in a] very, very timely [manner], but the problem is if your diagnosis is not very reliable.”
Dr. Ferreira had another advantage: Knowing from sequential blood samples that his patients were exposed to dengue within the past 5 years. He also could tell serologically when they became infected with the SARS-CoV-2 virus, which causes COVID-19.
Dr. Ferreira told this news organization that very few of their patients became seriously ill or required hospitalization. Because their sample size was too small, he could not say if prior dengue made the COVID-19 infection worse.
The type of interaction between two infections like dengue and COVID-19 is called a “syndemic,” which the CDC defines as “synergistically interacting epidemics.” Dr. Ferreira hypothesized about some of the factors that might be at play but does not yet have enough data. For example, he speculated about a biologic basis, such as a link to autoimmunity or vasculitis from prior dengue, but “has no real data to either support or reject these things.”
Dr. Ferreira added that perhaps there are social factors that put certain people at higher risk of infection; for example, maybe some people are “more exposed to high viral loads.”
In Brazil’s first wave of COVID-19, Dr. Ferreira’s team calculated dengue seroconversion as about 10%; many cases of dengue were asymptomatic. Ferreira expects they will “have a very different clinical spectrum during the second wave,” with young people becoming much more ill from the P1 variant of concern.
Scott O’Neill, PhD, founder and director of the World Mosquito Program, told this news organization that, while he found the Brazil results intriguing, at present they are not sufficient to say that there’s a causal relationship between dengue and COVID-19. He expressed concern that the results seem counterintuitive and doubts there’s a biological or mechanistic cause. Instead, Dr. O’Neill wondered if “there could be something about social or economic conditions or living conditions” that might account for the correlation. For example, perhaps poverty increases exposure to both dengue and COVID-19.
Furthermore, Dr. O’Neill said in an interview that he suspects that, with the COVID-19 lockdowns, “You might expect to see more dengue.” This is because “most transmission occurs around the house, and so [with] having more people confined to houses, you might expect to see more dengue.” Such appears to be the case in Singapore.
In an article in the Journal of Infectious Diseases, Jue Tao Lim and colleagues described increased dengue in Singapore during COVID-19. They noted that most employees in Singapore work in air-conditioned settings. With social distancing enforced to try to reduce COVID-19, people stayed at home. The mosquito that transmits dengue, Aedes aegypti, gathers in wet spots in residential areas and bites during the daytime. The authors hypothesized that the spike in dengue was because of this change in habits, which shifted people’s exposure.
The syndemic in Brazil is complicated, with malaria and multiple arboviral diseases (chikungunya, dengue, Zika) overlapping with COVID-19 in areas of high population density, poverty, and poor sanitation, among other social ills. Such overlapping factors make it harder to distinguish correlations from causations. Prospective longitudinal studies might be needed to provide definitive answers.
Dr. Ferreira and Dr. O’Neill have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a surprising study from the Amazon rain forest, Brazilian scientists found that symptomatic COVID-19 infections were twice as likely to occur in people who had prior dengue.
The study, led by Marcelo Urbano Ferreira, MD, PhD, of the University of São Paulo Biomedical Sciences Institute, was conducted in Mâncio Lima, a town in the Amazon region of Brazil, and published May 6, 2021, in Clinical Infectious Diseases.
In the study, supported by the São Paulo Research Foundation, Ferreira’s team looked at sequential blood samples from 1,285 residents of Mâncio Lima.
An earlier study by Miguel Nicolelis, MD, PhD, and colleagues (published as a preprint) had analyzed data from the first COVID-19 wave in Brazil in 2020. It was an “ecological study” and examined dengue cases in different geographic regions of Brazil. That study concluded that dengue actually seemed to protect people from later developing COVID-19.
Dr. Ferreira anticipated finding a similar effect. Instead, he found the opposite effect. Although dengue did not increase the risk of subsequent COVID-19 infection, symptomatic COVID-19 became twice as likely in people with prior dengue. His study was longitudinal, following a single group of patients in Mâncio Lima over time.
Dr. Ferreira explained that ecological studies are inherently less accurate, as they look at populations in different places. “All the older cases are diagnosed on clinical grounds ... Because most infections are either asymptomatic or symptoms can be easily confused with” other diseases, many cases are missed. So, during the dengue transmission season, “We have some overestimation of the actual number of cases, and outside the transmission season, we have underestimation of the cases.”
On the apparent discrepancy with the earlier study by Dr. Nicolelis, Dr. Ferreira commented, “It’s a wonderful study because it’s something you can do quickly and test a hypothesis [in a] very, very timely [manner], but the problem is if your diagnosis is not very reliable.”
Dr. Ferreira had another advantage: Knowing from sequential blood samples that his patients were exposed to dengue within the past 5 years. He also could tell serologically when they became infected with the SARS-CoV-2 virus, which causes COVID-19.
Dr. Ferreira told this news organization that very few of their patients became seriously ill or required hospitalization. Because their sample size was too small, he could not say if prior dengue made the COVID-19 infection worse.
The type of interaction between two infections like dengue and COVID-19 is called a “syndemic,” which the CDC defines as “synergistically interacting epidemics.” Dr. Ferreira hypothesized about some of the factors that might be at play but does not yet have enough data. For example, he speculated about a biologic basis, such as a link to autoimmunity or vasculitis from prior dengue, but “has no real data to either support or reject these things.”
Dr. Ferreira added that perhaps there are social factors that put certain people at higher risk of infection; for example, maybe some people are “more exposed to high viral loads.”
In Brazil’s first wave of COVID-19, Dr. Ferreira’s team calculated dengue seroconversion as about 10%; many cases of dengue were asymptomatic. Ferreira expects they will “have a very different clinical spectrum during the second wave,” with young people becoming much more ill from the P1 variant of concern.
Scott O’Neill, PhD, founder and director of the World Mosquito Program, told this news organization that, while he found the Brazil results intriguing, at present they are not sufficient to say that there’s a causal relationship between dengue and COVID-19. He expressed concern that the results seem counterintuitive and doubts there’s a biological or mechanistic cause. Instead, Dr. O’Neill wondered if “there could be something about social or economic conditions or living conditions” that might account for the correlation. For example, perhaps poverty increases exposure to both dengue and COVID-19.
Furthermore, Dr. O’Neill said in an interview that he suspects that, with the COVID-19 lockdowns, “You might expect to see more dengue.” This is because “most transmission occurs around the house, and so [with] having more people confined to houses, you might expect to see more dengue.” Such appears to be the case in Singapore.
In an article in the Journal of Infectious Diseases, Jue Tao Lim and colleagues described increased dengue in Singapore during COVID-19. They noted that most employees in Singapore work in air-conditioned settings. With social distancing enforced to try to reduce COVID-19, people stayed at home. The mosquito that transmits dengue, Aedes aegypti, gathers in wet spots in residential areas and bites during the daytime. The authors hypothesized that the spike in dengue was because of this change in habits, which shifted people’s exposure.
The syndemic in Brazil is complicated, with malaria and multiple arboviral diseases (chikungunya, dengue, Zika) overlapping with COVID-19 in areas of high population density, poverty, and poor sanitation, among other social ills. Such overlapping factors make it harder to distinguish correlations from causations. Prospective longitudinal studies might be needed to provide definitive answers.
Dr. Ferreira and Dr. O’Neill have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a surprising study from the Amazon rain forest, Brazilian scientists found that symptomatic COVID-19 infections were twice as likely to occur in people who had prior dengue.
The study, led by Marcelo Urbano Ferreira, MD, PhD, of the University of São Paulo Biomedical Sciences Institute, was conducted in Mâncio Lima, a town in the Amazon region of Brazil, and published May 6, 2021, in Clinical Infectious Diseases.
In the study, supported by the São Paulo Research Foundation, Ferreira’s team looked at sequential blood samples from 1,285 residents of Mâncio Lima.
An earlier study by Miguel Nicolelis, MD, PhD, and colleagues (published as a preprint) had analyzed data from the first COVID-19 wave in Brazil in 2020. It was an “ecological study” and examined dengue cases in different geographic regions of Brazil. That study concluded that dengue actually seemed to protect people from later developing COVID-19.
Dr. Ferreira anticipated finding a similar effect. Instead, he found the opposite effect. Although dengue did not increase the risk of subsequent COVID-19 infection, symptomatic COVID-19 became twice as likely in people with prior dengue. His study was longitudinal, following a single group of patients in Mâncio Lima over time.
Dr. Ferreira explained that ecological studies are inherently less accurate, as they look at populations in different places. “All the older cases are diagnosed on clinical grounds ... Because most infections are either asymptomatic or symptoms can be easily confused with” other diseases, many cases are missed. So, during the dengue transmission season, “We have some overestimation of the actual number of cases, and outside the transmission season, we have underestimation of the cases.”
On the apparent discrepancy with the earlier study by Dr. Nicolelis, Dr. Ferreira commented, “It’s a wonderful study because it’s something you can do quickly and test a hypothesis [in a] very, very timely [manner], but the problem is if your diagnosis is not very reliable.”
Dr. Ferreira had another advantage: Knowing from sequential blood samples that his patients were exposed to dengue within the past 5 years. He also could tell serologically when they became infected with the SARS-CoV-2 virus, which causes COVID-19.
Dr. Ferreira told this news organization that very few of their patients became seriously ill or required hospitalization. Because their sample size was too small, he could not say if prior dengue made the COVID-19 infection worse.
The type of interaction between two infections like dengue and COVID-19 is called a “syndemic,” which the CDC defines as “synergistically interacting epidemics.” Dr. Ferreira hypothesized about some of the factors that might be at play but does not yet have enough data. For example, he speculated about a biologic basis, such as a link to autoimmunity or vasculitis from prior dengue, but “has no real data to either support or reject these things.”
Dr. Ferreira added that perhaps there are social factors that put certain people at higher risk of infection; for example, maybe some people are “more exposed to high viral loads.”
In Brazil’s first wave of COVID-19, Dr. Ferreira’s team calculated dengue seroconversion as about 10%; many cases of dengue were asymptomatic. Ferreira expects they will “have a very different clinical spectrum during the second wave,” with young people becoming much more ill from the P1 variant of concern.
Scott O’Neill, PhD, founder and director of the World Mosquito Program, told this news organization that, while he found the Brazil results intriguing, at present they are not sufficient to say that there’s a causal relationship between dengue and COVID-19. He expressed concern that the results seem counterintuitive and doubts there’s a biological or mechanistic cause. Instead, Dr. O’Neill wondered if “there could be something about social or economic conditions or living conditions” that might account for the correlation. For example, perhaps poverty increases exposure to both dengue and COVID-19.
Furthermore, Dr. O’Neill said in an interview that he suspects that, with the COVID-19 lockdowns, “You might expect to see more dengue.” This is because “most transmission occurs around the house, and so [with] having more people confined to houses, you might expect to see more dengue.” Such appears to be the case in Singapore.
In an article in the Journal of Infectious Diseases, Jue Tao Lim and colleagues described increased dengue in Singapore during COVID-19. They noted that most employees in Singapore work in air-conditioned settings. With social distancing enforced to try to reduce COVID-19, people stayed at home. The mosquito that transmits dengue, Aedes aegypti, gathers in wet spots in residential areas and bites during the daytime. The authors hypothesized that the spike in dengue was because of this change in habits, which shifted people’s exposure.
The syndemic in Brazil is complicated, with malaria and multiple arboviral diseases (chikungunya, dengue, Zika) overlapping with COVID-19 in areas of high population density, poverty, and poor sanitation, among other social ills. Such overlapping factors make it harder to distinguish correlations from causations. Prospective longitudinal studies might be needed to provide definitive answers.
Dr. Ferreira and Dr. O’Neill have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
How to help vaccinated patients navigate FOGO (fear of going out)
Remember FOMO (fear of missing out)? The pandemic cured most of us of that! In its place, many are suffering from a new syndrome that has been coined “FOGO” (fear of going out). As the COVID-19 vaccines roll out, restrictions lessen, and cases decline, we face new challenges. The pandemic showed us that “we are all in it together.” Now our patients, family, friends – and even we, ourselves – may face similar anxieties as we transition back.
Our brains love routines. They save energy as we transverse the same pathway with ease. We created new patterns in the first 30 days of quarantine, and we spent more than a year engraining them. Many people are feeling even more anxiety as restrictions are lifting and expectations are rising. Those with preexisting anxiety disorders may have an even more difficult time resuming routine activities.
Since the virus is still among us, we need to maintain caution, so some degree of FOGO is wise. But when we limit our activities too much, we create a whole new host of issues. The pandemic gave us all a taste of the agoraphobic lifestyle. It is difficult to know where exactly to draw the line right now between healthy anxiety and anxiety that becomes the disease for ourselves, our families and friends – and our patients.
Recommendations for FOGO
- Talk to your families, friends, and patients about what activities you recommend, which they might resume and which they should continue to avoid. People should make plans to optimize their physical and mental health while continuing to protect themselves from COVID-19. If anxiety is becoming the main problem, psychotherapy or medication may be necessary to treat their symptoms.
- Continue to encourage those with FOGO to practice techniques to be calm. Suggest that they take deep breaths with long exhales. This breathing pattern activates the parasympathetic nervous system and will help them feel calmer. We have all been under chronic stress, and our sympathetic nervous system has been in overdrive. We need to be calm to make the best decisions so our frontal lobe can be in charge rather than our primitive, fear-based brain that has been running the show for more a year. Encourage calming activities, such as yoga, meditation, warm baths, spending time in nature, hugging a pet, and more.
- Advise sufferers to start slowly. They should resume activities where they feel the safest. Walking outside with a friend is a good way to start. We now know that transmission is remarkably low or nonexistent if both parties are vaccinated. Exercise is a great way to combat many psychological issues, including FOGO.
- FOGO sufferers should build confidence gradually. Recommend taking one day at a time and trying to find ways to enjoy new ventures out. Soon, our brains will adapt to the new routines and the days of COVID-19 will recede from our thoughts.
- Respect whatever feelings emerge. The closer we and our patients were to trauma, the more challenging it may be to recover. If you or your patients suffered from COVID-19 or had a close family member or friend who did, be prepared to reemerge more slowly. Don’t feel pressured by what others are doing. Go at your own pace. Only you can decide what is the right way to move forward in these times.
- Look for signs of substance overuse or misuse. FOGO sufferers may turn to drugs or alcohol to mask their anxiety. This is a common pothole and should be avoided. Be alert for this problem and discuss it with patients, friends, or family members who may be making unhealthy choices.
Time is a great healer, and remind others that “this too shall pass.” FOGO will give rise to another yet-to-be named syndrome. We seem to be moving in a very positive direction at a remarkable pace. As Alexander Pope so wisely wrote, “Hope springs eternal.” Better times are ahead.
Dr. Ritvo, who has almost 30 years’ experience in psychiatry, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa. Momosa Publishing, 2018). Dr. Ritvo has no disclosures.
Remember FOMO (fear of missing out)? The pandemic cured most of us of that! In its place, many are suffering from a new syndrome that has been coined “FOGO” (fear of going out). As the COVID-19 vaccines roll out, restrictions lessen, and cases decline, we face new challenges. The pandemic showed us that “we are all in it together.” Now our patients, family, friends – and even we, ourselves – may face similar anxieties as we transition back.
Our brains love routines. They save energy as we transverse the same pathway with ease. We created new patterns in the first 30 days of quarantine, and we spent more than a year engraining them. Many people are feeling even more anxiety as restrictions are lifting and expectations are rising. Those with preexisting anxiety disorders may have an even more difficult time resuming routine activities.
Since the virus is still among us, we need to maintain caution, so some degree of FOGO is wise. But when we limit our activities too much, we create a whole new host of issues. The pandemic gave us all a taste of the agoraphobic lifestyle. It is difficult to know where exactly to draw the line right now between healthy anxiety and anxiety that becomes the disease for ourselves, our families and friends – and our patients.
Recommendations for FOGO
- Talk to your families, friends, and patients about what activities you recommend, which they might resume and which they should continue to avoid. People should make plans to optimize their physical and mental health while continuing to protect themselves from COVID-19. If anxiety is becoming the main problem, psychotherapy or medication may be necessary to treat their symptoms.
- Continue to encourage those with FOGO to practice techniques to be calm. Suggest that they take deep breaths with long exhales. This breathing pattern activates the parasympathetic nervous system and will help them feel calmer. We have all been under chronic stress, and our sympathetic nervous system has been in overdrive. We need to be calm to make the best decisions so our frontal lobe can be in charge rather than our primitive, fear-based brain that has been running the show for more a year. Encourage calming activities, such as yoga, meditation, warm baths, spending time in nature, hugging a pet, and more.
- Advise sufferers to start slowly. They should resume activities where they feel the safest. Walking outside with a friend is a good way to start. We now know that transmission is remarkably low or nonexistent if both parties are vaccinated. Exercise is a great way to combat many psychological issues, including FOGO.
- FOGO sufferers should build confidence gradually. Recommend taking one day at a time and trying to find ways to enjoy new ventures out. Soon, our brains will adapt to the new routines and the days of COVID-19 will recede from our thoughts.
- Respect whatever feelings emerge. The closer we and our patients were to trauma, the more challenging it may be to recover. If you or your patients suffered from COVID-19 or had a close family member or friend who did, be prepared to reemerge more slowly. Don’t feel pressured by what others are doing. Go at your own pace. Only you can decide what is the right way to move forward in these times.
- Look for signs of substance overuse or misuse. FOGO sufferers may turn to drugs or alcohol to mask their anxiety. This is a common pothole and should be avoided. Be alert for this problem and discuss it with patients, friends, or family members who may be making unhealthy choices.
Time is a great healer, and remind others that “this too shall pass.” FOGO will give rise to another yet-to-be named syndrome. We seem to be moving in a very positive direction at a remarkable pace. As Alexander Pope so wisely wrote, “Hope springs eternal.” Better times are ahead.
Dr. Ritvo, who has almost 30 years’ experience in psychiatry, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa. Momosa Publishing, 2018). Dr. Ritvo has no disclosures.
Remember FOMO (fear of missing out)? The pandemic cured most of us of that! In its place, many are suffering from a new syndrome that has been coined “FOGO” (fear of going out). As the COVID-19 vaccines roll out, restrictions lessen, and cases decline, we face new challenges. The pandemic showed us that “we are all in it together.” Now our patients, family, friends – and even we, ourselves – may face similar anxieties as we transition back.
Our brains love routines. They save energy as we transverse the same pathway with ease. We created new patterns in the first 30 days of quarantine, and we spent more than a year engraining them. Many people are feeling even more anxiety as restrictions are lifting and expectations are rising. Those with preexisting anxiety disorders may have an even more difficult time resuming routine activities.
Since the virus is still among us, we need to maintain caution, so some degree of FOGO is wise. But when we limit our activities too much, we create a whole new host of issues. The pandemic gave us all a taste of the agoraphobic lifestyle. It is difficult to know where exactly to draw the line right now between healthy anxiety and anxiety that becomes the disease for ourselves, our families and friends – and our patients.
Recommendations for FOGO
- Talk to your families, friends, and patients about what activities you recommend, which they might resume and which they should continue to avoid. People should make plans to optimize their physical and mental health while continuing to protect themselves from COVID-19. If anxiety is becoming the main problem, psychotherapy or medication may be necessary to treat their symptoms.
- Continue to encourage those with FOGO to practice techniques to be calm. Suggest that they take deep breaths with long exhales. This breathing pattern activates the parasympathetic nervous system and will help them feel calmer. We have all been under chronic stress, and our sympathetic nervous system has been in overdrive. We need to be calm to make the best decisions so our frontal lobe can be in charge rather than our primitive, fear-based brain that has been running the show for more a year. Encourage calming activities, such as yoga, meditation, warm baths, spending time in nature, hugging a pet, and more.
- Advise sufferers to start slowly. They should resume activities where they feel the safest. Walking outside with a friend is a good way to start. We now know that transmission is remarkably low or nonexistent if both parties are vaccinated. Exercise is a great way to combat many psychological issues, including FOGO.
- FOGO sufferers should build confidence gradually. Recommend taking one day at a time and trying to find ways to enjoy new ventures out. Soon, our brains will adapt to the new routines and the days of COVID-19 will recede from our thoughts.
- Respect whatever feelings emerge. The closer we and our patients were to trauma, the more challenging it may be to recover. If you or your patients suffered from COVID-19 or had a close family member or friend who did, be prepared to reemerge more slowly. Don’t feel pressured by what others are doing. Go at your own pace. Only you can decide what is the right way to move forward in these times.
- Look for signs of substance overuse or misuse. FOGO sufferers may turn to drugs or alcohol to mask their anxiety. This is a common pothole and should be avoided. Be alert for this problem and discuss it with patients, friends, or family members who may be making unhealthy choices.
Time is a great healer, and remind others that “this too shall pass.” FOGO will give rise to another yet-to-be named syndrome. We seem to be moving in a very positive direction at a remarkable pace. As Alexander Pope so wisely wrote, “Hope springs eternal.” Better times are ahead.
Dr. Ritvo, who has almost 30 years’ experience in psychiatry, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa. Momosa Publishing, 2018). Dr. Ritvo has no disclosures.
AHA reassures myocarditis rare after COVID vaccination, benefits overwhelm risks
The benefits of COVID-19 vaccination “enormously outweigh” the rare possible risk for heart-related complications, including myocarditis, the American Heart Association/American Stroke Association (ASA) says in new statement.
The message follows a Centers for Disease Control and Prevention report that the agency is monitoring the Vaccine Adverse Events Reporting System (VAERS) and the Vaccine Safety Datalink (VSD) for cases of myocarditis that have been associated with the mRNA vaccines against SARS-CoV-2 from Pfizer and Moderna.
The “relatively few” reported cases myocarditis in adolescents or young adults have involved males more often than females, more often followed the second dose rather than the first, and were usually seen in the 4 days after vaccination, the CDC’s COVID-19 Vaccine Safety Technical Work Group (VaST) found.
“Most cases appear to be mild, and follow-up of cases is ongoing,” the CDC says. “Within CDC safety monitoring systems, rates of myocarditis reports in the window following COVID-19 vaccination have not differed from expected baseline rates.”
In their statement, the AHA/ASA “strongly urge” all adults and children 12 years and older to receive a COVID-19 vaccine as soon as possible.
“The evidence continues to indicate that the COVID-19 vaccines are nearly 100% effective at preventing death and hospitalization due to COVID-19 infection,” the groups say.
Although the investigation of cases of myocarditis related to COVID-19 vaccination is ongoing, the AHA/ASA notes that myocarditis is typically the result of an actual viral infection, “and it is yet to be determined if these cases have any correlation to receiving a COVID-19 vaccine.”
“We’ve lost hundreds of children, and there have been thousands who have been hospitalized, thousands who developed an inflammatory syndrome, and one of the pieces of that can be myocarditis,” Richard Besser, MD, president and CEO of the Robert Wood Johnson Foundation (RWJF), said today on ABC’s Good Morning America.
Still, “from my perspective, the risk of COVID is so much greater than any theoretical risk from the vaccine,” said Dr. Besser, former acting director of the CDC.
The symptoms that can occur after COVID-19 vaccination include tiredness, headache, muscle pain, chills, fever, and nausea, reminds the AHA/ASA statement. Such symptoms would “typically appear within 24-48 hours and usually pass within 36-48 hours after receiving the vaccine.”
All health care providers should be aware of the “very rare” adverse events that could be related to a COVID-19 vaccine, including myocarditis, blood clots, low platelets, and symptoms of severe inflammation, it says.
“Health care professionals should strongly consider inquiring about the timing of any recent COVID vaccination among patients presenting with these conditions, as needed, in order to provide appropriate treatment quickly,” the statement advises.
A version of this article first appeared on Medscape.com.
The benefits of COVID-19 vaccination “enormously outweigh” the rare possible risk for heart-related complications, including myocarditis, the American Heart Association/American Stroke Association (ASA) says in new statement.
The message follows a Centers for Disease Control and Prevention report that the agency is monitoring the Vaccine Adverse Events Reporting System (VAERS) and the Vaccine Safety Datalink (VSD) for cases of myocarditis that have been associated with the mRNA vaccines against SARS-CoV-2 from Pfizer and Moderna.
The “relatively few” reported cases myocarditis in adolescents or young adults have involved males more often than females, more often followed the second dose rather than the first, and were usually seen in the 4 days after vaccination, the CDC’s COVID-19 Vaccine Safety Technical Work Group (VaST) found.
“Most cases appear to be mild, and follow-up of cases is ongoing,” the CDC says. “Within CDC safety monitoring systems, rates of myocarditis reports in the window following COVID-19 vaccination have not differed from expected baseline rates.”
In their statement, the AHA/ASA “strongly urge” all adults and children 12 years and older to receive a COVID-19 vaccine as soon as possible.
“The evidence continues to indicate that the COVID-19 vaccines are nearly 100% effective at preventing death and hospitalization due to COVID-19 infection,” the groups say.
Although the investigation of cases of myocarditis related to COVID-19 vaccination is ongoing, the AHA/ASA notes that myocarditis is typically the result of an actual viral infection, “and it is yet to be determined if these cases have any correlation to receiving a COVID-19 vaccine.”
“We’ve lost hundreds of children, and there have been thousands who have been hospitalized, thousands who developed an inflammatory syndrome, and one of the pieces of that can be myocarditis,” Richard Besser, MD, president and CEO of the Robert Wood Johnson Foundation (RWJF), said today on ABC’s Good Morning America.
Still, “from my perspective, the risk of COVID is so much greater than any theoretical risk from the vaccine,” said Dr. Besser, former acting director of the CDC.
The symptoms that can occur after COVID-19 vaccination include tiredness, headache, muscle pain, chills, fever, and nausea, reminds the AHA/ASA statement. Such symptoms would “typically appear within 24-48 hours and usually pass within 36-48 hours after receiving the vaccine.”
All health care providers should be aware of the “very rare” adverse events that could be related to a COVID-19 vaccine, including myocarditis, blood clots, low platelets, and symptoms of severe inflammation, it says.
“Health care professionals should strongly consider inquiring about the timing of any recent COVID vaccination among patients presenting with these conditions, as needed, in order to provide appropriate treatment quickly,” the statement advises.
A version of this article first appeared on Medscape.com.
The benefits of COVID-19 vaccination “enormously outweigh” the rare possible risk for heart-related complications, including myocarditis, the American Heart Association/American Stroke Association (ASA) says in new statement.
The message follows a Centers for Disease Control and Prevention report that the agency is monitoring the Vaccine Adverse Events Reporting System (VAERS) and the Vaccine Safety Datalink (VSD) for cases of myocarditis that have been associated with the mRNA vaccines against SARS-CoV-2 from Pfizer and Moderna.
The “relatively few” reported cases myocarditis in adolescents or young adults have involved males more often than females, more often followed the second dose rather than the first, and were usually seen in the 4 days after vaccination, the CDC’s COVID-19 Vaccine Safety Technical Work Group (VaST) found.
“Most cases appear to be mild, and follow-up of cases is ongoing,” the CDC says. “Within CDC safety monitoring systems, rates of myocarditis reports in the window following COVID-19 vaccination have not differed from expected baseline rates.”
In their statement, the AHA/ASA “strongly urge” all adults and children 12 years and older to receive a COVID-19 vaccine as soon as possible.
“The evidence continues to indicate that the COVID-19 vaccines are nearly 100% effective at preventing death and hospitalization due to COVID-19 infection,” the groups say.
Although the investigation of cases of myocarditis related to COVID-19 vaccination is ongoing, the AHA/ASA notes that myocarditis is typically the result of an actual viral infection, “and it is yet to be determined if these cases have any correlation to receiving a COVID-19 vaccine.”
“We’ve lost hundreds of children, and there have been thousands who have been hospitalized, thousands who developed an inflammatory syndrome, and one of the pieces of that can be myocarditis,” Richard Besser, MD, president and CEO of the Robert Wood Johnson Foundation (RWJF), said today on ABC’s Good Morning America.
Still, “from my perspective, the risk of COVID is so much greater than any theoretical risk from the vaccine,” said Dr. Besser, former acting director of the CDC.
The symptoms that can occur after COVID-19 vaccination include tiredness, headache, muscle pain, chills, fever, and nausea, reminds the AHA/ASA statement. Such symptoms would “typically appear within 24-48 hours and usually pass within 36-48 hours after receiving the vaccine.”
All health care providers should be aware of the “very rare” adverse events that could be related to a COVID-19 vaccine, including myocarditis, blood clots, low platelets, and symptoms of severe inflammation, it says.
“Health care professionals should strongly consider inquiring about the timing of any recent COVID vaccination among patients presenting with these conditions, as needed, in order to provide appropriate treatment quickly,” the statement advises.
A version of this article first appeared on Medscape.com.
First issue vs. April 2021 issue: Much has changed since 1971
For the first readers of Family Practice News, it started with this: “How safe is the pill? An extensive epidemiologic study being carried out in the United Kingdom by the Royal College of General Practitioners is expected to provide definitive answers to this question within the next few years.”

That was the first paragraph of the lead article on the front page of our very first issue, published in October 1971. The report on oral contraceptives, coming from the annual meeting of the British Medical Association in Leicester, largely focused on recruiting – noting that “all women in the study are married” – and data-gathering methods.
That first issue also covered such topics as the effect of “early and frequent coitus” on cervical dysplasia incidence (p. 4), breast cancer in men (p. 13), and treatment of prostate inflammation in patients with impotence (p. 34).
Our April 2021 issue included five articles related to the COVID-19 pandemic, starting on the front page and featuring a photo – a physician sitting at a computer, wearing a mask – and a topic – vaccine-hesitant patients.
Business of medicine today vs. in 1971
At the time of publication of our first issue the United States was in the midst of a 90-day freeze on wages and prices ordered by President Richard Nixon. Two articles in that first issue discussed the subject: “Freeze clouds future of health insurance plans” and “Freeze lets physicians ‘stabilize’ office fees.”
Besides COVID-19, here are some other topics covered in April 2021 but not in 1971: lessons learned from an electronic health records conversion, competition for physicians in the form of a “virtual primary care service” offered by United Healthcare, and the sleep effects of smartphone “addiction.”
Technology, clearly, plays a much larger role in physicians’ lives these days.
Similarities between issues
Not everything has changed, of course. We were informing physicians about heart disease in 1971 with “Primary MD can treat most vascular cases” and “Job satisfaction can help prevent heart disease.”
A look at the latest issue uncovered “Link clinched between high-glycemic index diets and cardiovascular disease events” and “Ultraprocessed ‘healthy’ foods raise cardiovascular disease events risk.” Diabetes is another topic that we have began covering since day one and continue to consider to be relevant to practicing family medicine. “Family attitude key to diabetic’s state” was published in our first issue and “Type 1 diabetes prevention moves toward reality as studies published” ran in our April 2021 issue.
The photos in that first issue, however, present a somewhat jarring counterpoint to our latest issue. The faces that look back from 50 years ago are men’s faces: 29 men, to be exact. There were no photos of women physicians in that issue.
That was not the case in April of 2021. Of the 26 physicians or research scientists who appeared in photos in that issue, 8 were women. Plus, three of those women appeared on the cover.
Among the photos from 1971 were 6 of the 14 founding members of our editorial advisory board, who were, again, all men. Our current board consists of 13 men and 8 women.
Times have changed.
This article was updated 5/27/21.
For the first readers of Family Practice News, it started with this: “How safe is the pill? An extensive epidemiologic study being carried out in the United Kingdom by the Royal College of General Practitioners is expected to provide definitive answers to this question within the next few years.”

That was the first paragraph of the lead article on the front page of our very first issue, published in October 1971. The report on oral contraceptives, coming from the annual meeting of the British Medical Association in Leicester, largely focused on recruiting – noting that “all women in the study are married” – and data-gathering methods.
That first issue also covered such topics as the effect of “early and frequent coitus” on cervical dysplasia incidence (p. 4), breast cancer in men (p. 13), and treatment of prostate inflammation in patients with impotence (p. 34).
Our April 2021 issue included five articles related to the COVID-19 pandemic, starting on the front page and featuring a photo – a physician sitting at a computer, wearing a mask – and a topic – vaccine-hesitant patients.
Business of medicine today vs. in 1971
At the time of publication of our first issue the United States was in the midst of a 90-day freeze on wages and prices ordered by President Richard Nixon. Two articles in that first issue discussed the subject: “Freeze clouds future of health insurance plans” and “Freeze lets physicians ‘stabilize’ office fees.”
Besides COVID-19, here are some other topics covered in April 2021 but not in 1971: lessons learned from an electronic health records conversion, competition for physicians in the form of a “virtual primary care service” offered by United Healthcare, and the sleep effects of smartphone “addiction.”
Technology, clearly, plays a much larger role in physicians’ lives these days.
Similarities between issues
Not everything has changed, of course. We were informing physicians about heart disease in 1971 with “Primary MD can treat most vascular cases” and “Job satisfaction can help prevent heart disease.”
A look at the latest issue uncovered “Link clinched between high-glycemic index diets and cardiovascular disease events” and “Ultraprocessed ‘healthy’ foods raise cardiovascular disease events risk.” Diabetes is another topic that we have began covering since day one and continue to consider to be relevant to practicing family medicine. “Family attitude key to diabetic’s state” was published in our first issue and “Type 1 diabetes prevention moves toward reality as studies published” ran in our April 2021 issue.
The photos in that first issue, however, present a somewhat jarring counterpoint to our latest issue. The faces that look back from 50 years ago are men’s faces: 29 men, to be exact. There were no photos of women physicians in that issue.
That was not the case in April of 2021. Of the 26 physicians or research scientists who appeared in photos in that issue, 8 were women. Plus, three of those women appeared on the cover.
Among the photos from 1971 were 6 of the 14 founding members of our editorial advisory board, who were, again, all men. Our current board consists of 13 men and 8 women.
Times have changed.
This article was updated 5/27/21.
For the first readers of Family Practice News, it started with this: “How safe is the pill? An extensive epidemiologic study being carried out in the United Kingdom by the Royal College of General Practitioners is expected to provide definitive answers to this question within the next few years.”

That was the first paragraph of the lead article on the front page of our very first issue, published in October 1971. The report on oral contraceptives, coming from the annual meeting of the British Medical Association in Leicester, largely focused on recruiting – noting that “all women in the study are married” – and data-gathering methods.
That first issue also covered such topics as the effect of “early and frequent coitus” on cervical dysplasia incidence (p. 4), breast cancer in men (p. 13), and treatment of prostate inflammation in patients with impotence (p. 34).
Our April 2021 issue included five articles related to the COVID-19 pandemic, starting on the front page and featuring a photo – a physician sitting at a computer, wearing a mask – and a topic – vaccine-hesitant patients.
Business of medicine today vs. in 1971
At the time of publication of our first issue the United States was in the midst of a 90-day freeze on wages and prices ordered by President Richard Nixon. Two articles in that first issue discussed the subject: “Freeze clouds future of health insurance plans” and “Freeze lets physicians ‘stabilize’ office fees.”
Besides COVID-19, here are some other topics covered in April 2021 but not in 1971: lessons learned from an electronic health records conversion, competition for physicians in the form of a “virtual primary care service” offered by United Healthcare, and the sleep effects of smartphone “addiction.”
Technology, clearly, plays a much larger role in physicians’ lives these days.
Similarities between issues
Not everything has changed, of course. We were informing physicians about heart disease in 1971 with “Primary MD can treat most vascular cases” and “Job satisfaction can help prevent heart disease.”
A look at the latest issue uncovered “Link clinched between high-glycemic index diets and cardiovascular disease events” and “Ultraprocessed ‘healthy’ foods raise cardiovascular disease events risk.” Diabetes is another topic that we have began covering since day one and continue to consider to be relevant to practicing family medicine. “Family attitude key to diabetic’s state” was published in our first issue and “Type 1 diabetes prevention moves toward reality as studies published” ran in our April 2021 issue.
The photos in that first issue, however, present a somewhat jarring counterpoint to our latest issue. The faces that look back from 50 years ago are men’s faces: 29 men, to be exact. There were no photos of women physicians in that issue.
That was not the case in April of 2021. Of the 26 physicians or research scientists who appeared in photos in that issue, 8 were women. Plus, three of those women appeared on the cover.
Among the photos from 1971 were 6 of the 14 founding members of our editorial advisory board, who were, again, all men. Our current board consists of 13 men and 8 women.
Times have changed.
This article was updated 5/27/21.
Cultivating emotional awareness
A path to resilience and joy in the hospital
Approaching the nursing unit, I heard the anxiety in my masked colleagues’ voices. I was starting another rotation on our COVID unit; this week I was trying to develop my emotional awareness in an effort to help with the stress of the job and, just as importantly, take in the moments of positive emotions when they arose. I was making a conscious effort to take in all I saw and felt in the same way I approached my patient examinations: my mind quiet, receptive, and curious.
Seeing my nursing teammates covered with personal protective equipment, I felt a little reverence at the purpose they bring to work. Thinking of our patients, isolated and scared in noisy, ventilated rooms, there was compassion welling up in my chest. Thinking about my role on the team, I felt humbled by the challenges of treating this new disease and meeting the needs of staff and patient.
A few years ago, a period of frustration and disaffectedness had led me to apply my diagnostic eye to myself: I was burning out. Developing a mindfulness practice has transformed my experience at work. Now, the pandemic pushed me to go beyond a few minutes of quieting the mind before work. I was developing my emotional awareness. A growing body of research suggests that emotional awareness helps temper the negative experiences and savor the good. This week on the COVID unit was an opportunity to put this idea to the test.
Across the hall from the desk was Ms. A, 85-year-old woman who always clutched her rosary. My Spanish is not great, but I understood her prayer when I entered the room. She had tested positive for COVID about 7 days before – so had all the people in her multigenerational home. Over the din of the negative-pressure machine, with damp eyes she kept saying she wanted to go home. I felt my body soften and, in my chest, it felt as if my heart moved towards her which is the manifestation of compassion. “I will do my best to get you there soon,” I said in an effort to comfort her.
We often resist strong emotions, especially at work, because they can increase stress in situations where we need to be in control. In high-emotion situations, our brain’s warning centers alert both body and brain. This has helped our ancestors to action over the millennia, but in the hospital, these responses hurt more than help. Our bodies amplifying the emotion, our mind races for solutions and we can feel overwhelmed.
Simply recognizing the emotion and naming it puts the brakes on this process. fMRI data demonstrate that naming the emotions moves the brain activity away from the emotion centers to the appraisal centers in the frontal lobe. Just the perspective to see the emotional process calms it down.
Name it to tame it – this is what those in the field call this act. “This is sadness,” I said to myself as I left Ms. A’s room.
Down the hall was Mr. D; he was an 81-year-old former Vietnamese refugee. He had come in 3 days prior to my coming on service. While he didn’t talk, even with an interpreter, he ate well and had looked comfortable for days on 50% O2.
Ms. A’s O2 needs crept up each day as did her anxiety, the plaintive tenor of her prayers and inquiries about going home. I got a priest to visit, not for last rites but just for some support. Over the phone, I updated the family on the prognosis.
A couple of days later, she needed 95% O2 and with PO2 was only 70. I told her family it seemed she was losing her battle with the virus. I said we could see how she did on 60% – that’s the max she could get at home with hospice. I called them after 2 hours on 60% to tell them she was up eating and despite slight increased resp rate, she looked okay. “Can you guarantee that she would not make it if she stayed in the hospital? “
My body vibrating with uncertainty – an emotional mix of fear and sadness – I said, “I am sorry, but this is such a new disease, I can’t say that for certain.” On the call, family members voiced different opinions, but in the end, they were unable to give up hope, so we agreed to keep her in hospital.
Down the hall, Mr. D had stopped eating and his sats dropped as did his blood pressure. A nurse exited his room; despite the mask and steamed-up glasses, I could read her body language. “That poor man is dying,” she said. I told her I agreed and called the family with the news and to offer them a chance to visit and to talk about home hospice.
“He has not seen any of us in 10 months,” said his daughter over the phone, “We would love to visit and talk about bringing him home on hospice.” The next morning four of his nine kids showed up with a quart of jook, an Asian rice porridge, for him and pastries for the staff.
They left the room smiling an hour later. “He ate all the jook and he smiled! Yes, let’s work on home with hospice.” That night his blood pressure was better, and we were able to move him to 8 liters oxymizer; the staff was excited by his improvement, too.
The next day Ms. A was less responsive with sats in the 80’s on 100% FiO2, but she still had this great sense of warmth and dignity about her. When I entered the room, Spanish Catholic hymns were playing, two of her kids stood leaning over the bed and on an iPod, there was a chorus of tears. 20 family members were all crying on a Zoom call. Together this made the most beautiful soundtrack to an end of life I have ever heard. I tried hard not to join the chorus as we talked about turning off the oxygen to help limit her suffering.
We added a bolus of morphine to her drip and removed the oxygen. She looked more beautiful and peaceful without it. Briefly, she closed her eyes then opened them, her breathing calmer. And with the hymns and the chorus of family crying she lived another 20 minutes in the loving presence of her big family.
Leaving the room, I was flooded with “woulda, coulda, shouldas” that accompany work with so much uncertainty and high stakes. “Maybe I should have tried convalescent plasma. Maybe I should have told them she must go home,” and so on my mind went on looking for solutions when there were none. I turned to my body – my chest ached, and I whispered to myself: “This is how sadness feels.”
By thinking about how the emotion feels in the body, we move the mind away from problem solving that can end up leading to unhealthy ruminations. Such thoughts in times of high emotions lead to that pressurized, tightness feeling we get when overwhelmed. Taking in the universal sensations of the emotions is calming and connects us with these deep human experiences in healthy ways. At the same time, the racing and ruminations stop.
Meanwhile, down the hall, Mr. D’s family arrived in great spirits armed with more food for patient and staff. He was to go home later that day with hospice. When they saw him up in the chair without the oxygen, they said: “It is a miracle, Dr. Hass! He is going home on hospice but having beat COVID! We can’t thank you enough!”
“Don’t thank me! He was cured by love and jook! What a lesson for us all. Sometimes there is no better medicine than food from home and love!” With the explosive expansiveness of joy, we shared some “elbow bumps” and took some pictures before he was wheeled home.
Back at the nurse’s station, there were tears. Sometimes life is so full of emotion that it is hard to give it a name – joy? grief? Our bodies almost pulsing, our minds searching for words, it is as if an ancient process is marking a time and place in our souls. “This is what it is to be a human being living with love and creating meaning,” the experience seems to be telling us.
This is awesome work. In fact, awe was what we were feeling then – that sense of wonder we have in the presence of something beautiful or vast that we cannot easily comprehend. Taking in these moments of awe at the power and depth of the human experience is critical to keep us humble, engaged, and emotionally involved.
Cultivating emotional awareness is a simple technique to maintain equanimity as we do the emotionally turbulent work of caring for vulnerable and seriously ill members of our community. It uses the same techniques of attention and diagnosis we use on those we care for. It is a practice that can be seamlessly incorporated into our workday with no time added. Recognizing it, naming it, and feeling it will give us the resilience to handle the challenges this amazing work inevitably brings.
Dr. Hass is a hospitalist at Sutter East Bay Medical Group in Oakland, Calif. He is a member of the clinical faculty at the University of California, Berkeley–UC San Francisco joint medical program, and an adviser on health and health care at the Greater Good Science Center at UC Berkeley.
A path to resilience and joy in the hospital
A path to resilience and joy in the hospital
Approaching the nursing unit, I heard the anxiety in my masked colleagues’ voices. I was starting another rotation on our COVID unit; this week I was trying to develop my emotional awareness in an effort to help with the stress of the job and, just as importantly, take in the moments of positive emotions when they arose. I was making a conscious effort to take in all I saw and felt in the same way I approached my patient examinations: my mind quiet, receptive, and curious.
Seeing my nursing teammates covered with personal protective equipment, I felt a little reverence at the purpose they bring to work. Thinking of our patients, isolated and scared in noisy, ventilated rooms, there was compassion welling up in my chest. Thinking about my role on the team, I felt humbled by the challenges of treating this new disease and meeting the needs of staff and patient.
A few years ago, a period of frustration and disaffectedness had led me to apply my diagnostic eye to myself: I was burning out. Developing a mindfulness practice has transformed my experience at work. Now, the pandemic pushed me to go beyond a few minutes of quieting the mind before work. I was developing my emotional awareness. A growing body of research suggests that emotional awareness helps temper the negative experiences and savor the good. This week on the COVID unit was an opportunity to put this idea to the test.
Across the hall from the desk was Ms. A, 85-year-old woman who always clutched her rosary. My Spanish is not great, but I understood her prayer when I entered the room. She had tested positive for COVID about 7 days before – so had all the people in her multigenerational home. Over the din of the negative-pressure machine, with damp eyes she kept saying she wanted to go home. I felt my body soften and, in my chest, it felt as if my heart moved towards her which is the manifestation of compassion. “I will do my best to get you there soon,” I said in an effort to comfort her.
We often resist strong emotions, especially at work, because they can increase stress in situations where we need to be in control. In high-emotion situations, our brain’s warning centers alert both body and brain. This has helped our ancestors to action over the millennia, but in the hospital, these responses hurt more than help. Our bodies amplifying the emotion, our mind races for solutions and we can feel overwhelmed.
Simply recognizing the emotion and naming it puts the brakes on this process. fMRI data demonstrate that naming the emotions moves the brain activity away from the emotion centers to the appraisal centers in the frontal lobe. Just the perspective to see the emotional process calms it down.
Name it to tame it – this is what those in the field call this act. “This is sadness,” I said to myself as I left Ms. A’s room.
Down the hall was Mr. D; he was an 81-year-old former Vietnamese refugee. He had come in 3 days prior to my coming on service. While he didn’t talk, even with an interpreter, he ate well and had looked comfortable for days on 50% O2.
Ms. A’s O2 needs crept up each day as did her anxiety, the plaintive tenor of her prayers and inquiries about going home. I got a priest to visit, not for last rites but just for some support. Over the phone, I updated the family on the prognosis.
A couple of days later, she needed 95% O2 and with PO2 was only 70. I told her family it seemed she was losing her battle with the virus. I said we could see how she did on 60% – that’s the max she could get at home with hospice. I called them after 2 hours on 60% to tell them she was up eating and despite slight increased resp rate, she looked okay. “Can you guarantee that she would not make it if she stayed in the hospital? “
My body vibrating with uncertainty – an emotional mix of fear and sadness – I said, “I am sorry, but this is such a new disease, I can’t say that for certain.” On the call, family members voiced different opinions, but in the end, they were unable to give up hope, so we agreed to keep her in hospital.
Down the hall, Mr. D had stopped eating and his sats dropped as did his blood pressure. A nurse exited his room; despite the mask and steamed-up glasses, I could read her body language. “That poor man is dying,” she said. I told her I agreed and called the family with the news and to offer them a chance to visit and to talk about home hospice.
“He has not seen any of us in 10 months,” said his daughter over the phone, “We would love to visit and talk about bringing him home on hospice.” The next morning four of his nine kids showed up with a quart of jook, an Asian rice porridge, for him and pastries for the staff.
They left the room smiling an hour later. “He ate all the jook and he smiled! Yes, let’s work on home with hospice.” That night his blood pressure was better, and we were able to move him to 8 liters oxymizer; the staff was excited by his improvement, too.
The next day Ms. A was less responsive with sats in the 80’s on 100% FiO2, but she still had this great sense of warmth and dignity about her. When I entered the room, Spanish Catholic hymns were playing, two of her kids stood leaning over the bed and on an iPod, there was a chorus of tears. 20 family members were all crying on a Zoom call. Together this made the most beautiful soundtrack to an end of life I have ever heard. I tried hard not to join the chorus as we talked about turning off the oxygen to help limit her suffering.
We added a bolus of morphine to her drip and removed the oxygen. She looked more beautiful and peaceful without it. Briefly, she closed her eyes then opened them, her breathing calmer. And with the hymns and the chorus of family crying she lived another 20 minutes in the loving presence of her big family.
Leaving the room, I was flooded with “woulda, coulda, shouldas” that accompany work with so much uncertainty and high stakes. “Maybe I should have tried convalescent plasma. Maybe I should have told them she must go home,” and so on my mind went on looking for solutions when there were none. I turned to my body – my chest ached, and I whispered to myself: “This is how sadness feels.”
By thinking about how the emotion feels in the body, we move the mind away from problem solving that can end up leading to unhealthy ruminations. Such thoughts in times of high emotions lead to that pressurized, tightness feeling we get when overwhelmed. Taking in the universal sensations of the emotions is calming and connects us with these deep human experiences in healthy ways. At the same time, the racing and ruminations stop.
Meanwhile, down the hall, Mr. D’s family arrived in great spirits armed with more food for patient and staff. He was to go home later that day with hospice. When they saw him up in the chair without the oxygen, they said: “It is a miracle, Dr. Hass! He is going home on hospice but having beat COVID! We can’t thank you enough!”
“Don’t thank me! He was cured by love and jook! What a lesson for us all. Sometimes there is no better medicine than food from home and love!” With the explosive expansiveness of joy, we shared some “elbow bumps” and took some pictures before he was wheeled home.
Back at the nurse’s station, there were tears. Sometimes life is so full of emotion that it is hard to give it a name – joy? grief? Our bodies almost pulsing, our minds searching for words, it is as if an ancient process is marking a time and place in our souls. “This is what it is to be a human being living with love and creating meaning,” the experience seems to be telling us.
This is awesome work. In fact, awe was what we were feeling then – that sense of wonder we have in the presence of something beautiful or vast that we cannot easily comprehend. Taking in these moments of awe at the power and depth of the human experience is critical to keep us humble, engaged, and emotionally involved.
Cultivating emotional awareness is a simple technique to maintain equanimity as we do the emotionally turbulent work of caring for vulnerable and seriously ill members of our community. It uses the same techniques of attention and diagnosis we use on those we care for. It is a practice that can be seamlessly incorporated into our workday with no time added. Recognizing it, naming it, and feeling it will give us the resilience to handle the challenges this amazing work inevitably brings.
Dr. Hass is a hospitalist at Sutter East Bay Medical Group in Oakland, Calif. He is a member of the clinical faculty at the University of California, Berkeley–UC San Francisco joint medical program, and an adviser on health and health care at the Greater Good Science Center at UC Berkeley.
Approaching the nursing unit, I heard the anxiety in my masked colleagues’ voices. I was starting another rotation on our COVID unit; this week I was trying to develop my emotional awareness in an effort to help with the stress of the job and, just as importantly, take in the moments of positive emotions when they arose. I was making a conscious effort to take in all I saw and felt in the same way I approached my patient examinations: my mind quiet, receptive, and curious.
Seeing my nursing teammates covered with personal protective equipment, I felt a little reverence at the purpose they bring to work. Thinking of our patients, isolated and scared in noisy, ventilated rooms, there was compassion welling up in my chest. Thinking about my role on the team, I felt humbled by the challenges of treating this new disease and meeting the needs of staff and patient.
A few years ago, a period of frustration and disaffectedness had led me to apply my diagnostic eye to myself: I was burning out. Developing a mindfulness practice has transformed my experience at work. Now, the pandemic pushed me to go beyond a few minutes of quieting the mind before work. I was developing my emotional awareness. A growing body of research suggests that emotional awareness helps temper the negative experiences and savor the good. This week on the COVID unit was an opportunity to put this idea to the test.
Across the hall from the desk was Ms. A, 85-year-old woman who always clutched her rosary. My Spanish is not great, but I understood her prayer when I entered the room. She had tested positive for COVID about 7 days before – so had all the people in her multigenerational home. Over the din of the negative-pressure machine, with damp eyes she kept saying she wanted to go home. I felt my body soften and, in my chest, it felt as if my heart moved towards her which is the manifestation of compassion. “I will do my best to get you there soon,” I said in an effort to comfort her.
We often resist strong emotions, especially at work, because they can increase stress in situations where we need to be in control. In high-emotion situations, our brain’s warning centers alert both body and brain. This has helped our ancestors to action over the millennia, but in the hospital, these responses hurt more than help. Our bodies amplifying the emotion, our mind races for solutions and we can feel overwhelmed.
Simply recognizing the emotion and naming it puts the brakes on this process. fMRI data demonstrate that naming the emotions moves the brain activity away from the emotion centers to the appraisal centers in the frontal lobe. Just the perspective to see the emotional process calms it down.
Name it to tame it – this is what those in the field call this act. “This is sadness,” I said to myself as I left Ms. A’s room.
Down the hall was Mr. D; he was an 81-year-old former Vietnamese refugee. He had come in 3 days prior to my coming on service. While he didn’t talk, even with an interpreter, he ate well and had looked comfortable for days on 50% O2.
Ms. A’s O2 needs crept up each day as did her anxiety, the plaintive tenor of her prayers and inquiries about going home. I got a priest to visit, not for last rites but just for some support. Over the phone, I updated the family on the prognosis.
A couple of days later, she needed 95% O2 and with PO2 was only 70. I told her family it seemed she was losing her battle with the virus. I said we could see how she did on 60% – that’s the max she could get at home with hospice. I called them after 2 hours on 60% to tell them she was up eating and despite slight increased resp rate, she looked okay. “Can you guarantee that she would not make it if she stayed in the hospital? “
My body vibrating with uncertainty – an emotional mix of fear and sadness – I said, “I am sorry, but this is such a new disease, I can’t say that for certain.” On the call, family members voiced different opinions, but in the end, they were unable to give up hope, so we agreed to keep her in hospital.
Down the hall, Mr. D had stopped eating and his sats dropped as did his blood pressure. A nurse exited his room; despite the mask and steamed-up glasses, I could read her body language. “That poor man is dying,” she said. I told her I agreed and called the family with the news and to offer them a chance to visit and to talk about home hospice.
“He has not seen any of us in 10 months,” said his daughter over the phone, “We would love to visit and talk about bringing him home on hospice.” The next morning four of his nine kids showed up with a quart of jook, an Asian rice porridge, for him and pastries for the staff.
They left the room smiling an hour later. “He ate all the jook and he smiled! Yes, let’s work on home with hospice.” That night his blood pressure was better, and we were able to move him to 8 liters oxymizer; the staff was excited by his improvement, too.
The next day Ms. A was less responsive with sats in the 80’s on 100% FiO2, but she still had this great sense of warmth and dignity about her. When I entered the room, Spanish Catholic hymns were playing, two of her kids stood leaning over the bed and on an iPod, there was a chorus of tears. 20 family members were all crying on a Zoom call. Together this made the most beautiful soundtrack to an end of life I have ever heard. I tried hard not to join the chorus as we talked about turning off the oxygen to help limit her suffering.
We added a bolus of morphine to her drip and removed the oxygen. She looked more beautiful and peaceful without it. Briefly, she closed her eyes then opened them, her breathing calmer. And with the hymns and the chorus of family crying she lived another 20 minutes in the loving presence of her big family.
Leaving the room, I was flooded with “woulda, coulda, shouldas” that accompany work with so much uncertainty and high stakes. “Maybe I should have tried convalescent plasma. Maybe I should have told them she must go home,” and so on my mind went on looking for solutions when there were none. I turned to my body – my chest ached, and I whispered to myself: “This is how sadness feels.”
By thinking about how the emotion feels in the body, we move the mind away from problem solving that can end up leading to unhealthy ruminations. Such thoughts in times of high emotions lead to that pressurized, tightness feeling we get when overwhelmed. Taking in the universal sensations of the emotions is calming and connects us with these deep human experiences in healthy ways. At the same time, the racing and ruminations stop.
Meanwhile, down the hall, Mr. D’s family arrived in great spirits armed with more food for patient and staff. He was to go home later that day with hospice. When they saw him up in the chair without the oxygen, they said: “It is a miracle, Dr. Hass! He is going home on hospice but having beat COVID! We can’t thank you enough!”
“Don’t thank me! He was cured by love and jook! What a lesson for us all. Sometimes there is no better medicine than food from home and love!” With the explosive expansiveness of joy, we shared some “elbow bumps” and took some pictures before he was wheeled home.
Back at the nurse’s station, there were tears. Sometimes life is so full of emotion that it is hard to give it a name – joy? grief? Our bodies almost pulsing, our minds searching for words, it is as if an ancient process is marking a time and place in our souls. “This is what it is to be a human being living with love and creating meaning,” the experience seems to be telling us.
This is awesome work. In fact, awe was what we were feeling then – that sense of wonder we have in the presence of something beautiful or vast that we cannot easily comprehend. Taking in these moments of awe at the power and depth of the human experience is critical to keep us humble, engaged, and emotionally involved.
Cultivating emotional awareness is a simple technique to maintain equanimity as we do the emotionally turbulent work of caring for vulnerable and seriously ill members of our community. It uses the same techniques of attention and diagnosis we use on those we care for. It is a practice that can be seamlessly incorporated into our workday with no time added. Recognizing it, naming it, and feeling it will give us the resilience to handle the challenges this amazing work inevitably brings.
Dr. Hass is a hospitalist at Sutter East Bay Medical Group in Oakland, Calif. He is a member of the clinical faculty at the University of California, Berkeley–UC San Francisco joint medical program, and an adviser on health and health care at the Greater Good Science Center at UC Berkeley.