User login
“I didn’t want to meet you.” Dispelling myths about palliative care
The names of health care professionals and patients cited within the dialogue text have been changed to protect their privacy.
but over the years I have come to realize that she was right – most people, including many within health care, don’t have a good appreciation of what palliative care is or how it can help patients and health care teams.
A recent national survey about cancer-related health information found that of more than 1,000 surveyed Americans, less than 30% professed any knowledge of palliative care. Of those who had some knowledge of palliative care, around 30% believed palliative care was synonymous with hospice.1 Another 15% believed that a patient would have to give up cancer-directed treatments to receive palliative care.1
It’s not giving up
This persistent belief that palliative care is equivalent to hospice, or is tantamount to “giving up,” is one of the most commonly held myths I encounter in everyday practice.
I knock on the exam door and walk in.
A small, trim woman in her late 50s is sitting in a chair, arms folded across her chest, face drawn in.
“Hi,” I start. “I’m Sarah, the palliative care nurse practitioner who works in this clinic. I work closely with Dr. Smith.”
Dr. Smith is the patient’s oncologist.
“I really didn’t want to meet you,” she says in a quiet voice, her eyes large with concern.
I don’t take it personally. Few patients really want to be in the position of needing to meet the palliative care team.
“I looked up palliative care on Google and saw the word hospice.”
“Yeah,” I say. “I hear that a lot. Well, I can reassure you that this isn’t hospice.
In this clinic, our focus is on your cancer symptoms, your treatment side effects, and your quality of life.”
She looks visibly relieved. “Quality of life,” she echoes. “I need more of that.”
“OK,” I say. “So, tell me what you’re struggling with the most right now.”
That’s how many palliative care visits start. I actually prefer if patients haven’t heard of palliative care because it allows me to frame it for them, rather than having to start by addressing a myth or a prior negative experience. Even when patients haven’t had a negative experience with palliative care per se, typically, if they’ve interacted with palliative care in the past, it’s usually because someone they loved died in a hospital setting and it is the memory of that terrible loss that becomes synonymous with their recollection of palliative care.
Many patients I meet have never seen another outpatient palliative care practitioner – and this makes sense – we are still too few and far between. Most established palliative care teams are hospital based and many patients seen in the community do not have easy access to palliative care teams where they receive oncologic care.2 As an embedded practitioner, I see patients in the same exam rooms and infusion centers where they receive their cancer therapies, so I’m effectively woven into the fabric of their oncology experience. Just being there in the cancer center allows me to be in the right place at the right time for the right patients and their care teams.
More than pain management
Another myth I tend to dispel a lot is that palliative care is just a euphemism for “pain management.” I have seen this less lately, but still occasionally in the chart I’ll see documented in a note, “patient is seeing palliative/pain management,” when a patient is seeing me or one of my colleagues. Unfortunately, when providers have limited or outdated views of what palliative care is or the value it brings to patient-centered cancer care, referrals to palliative care tend to be delayed.3
“I really think Ms. Lopez could benefit from seeing palliative care,” an oncology nurse practitioner says to an oncologist.
I’m standing nearby, about to see another patient in one of the exam rooms in our clinic.
“But I don’t think she’s ready. And besides, she doesn’t have any pain,” he says.
He turns to me quizzically. “What do you think?”
“Tell me about the patient,” I ask, taking a few steps in their direction.
“Well, she’s a 64-year-old woman with metastatic cancer.
She has a really poor appetite and is losing some weight.
Seems a bit down, kind of pessimistic about things.
Her scan showed some new growth, so guess I’m not surprised by that.”
“I might be able to help her with the appetite and the mood changes.
I can at least talk with her and see where she’s at,” I offer.
“Alright,” he says. “We’ll put the palliative referral in.”
He hesitates. “But are you sure you want to see her?
She doesn’t have any pain.” He sounds skeptical.
“Yeah, I mean, it sounds like she has symptoms that are bothering her, so I’d be happy to see her. She sounds completely appropriate for palliative care.”
I hear this assumption a lot – that palliative care is somehow equivalent to pain management and that unless a patient’s pain is severe, it’s not worth referring the patient to palliative care. Don’t get me wrong – we do a lot of pain management, but at its heart, palliative care is an interdisciplinary specialty focused on improving or maintaining quality of life for people with serious illness. Because the goal is so broad, care can take many shapes.4
In addition to pain, palliative care clinicians commonly treat nausea, shortness of breath, constipation or diarrhea, poor appetite, fatigue, anxiety, depression, and insomnia.
Palliative care is more than medical or nursing care
A related misconception about palliative care held by many lay people and health care workers alike is that palliative care is primarily medical or nursing care focused mostly on alleviating physical symptoms such as pain or nausea. This couldn’t be further from the truth.
We’ve been talking for a while.
Ms. Lopez tells me about her struggles to maintain her weight while undergoing chemotherapy. She has low-grade nausea that is impacting her ability and desire to eat more and didn’t think that her weight loss was severe enough to warrant taking medication.
We talk about how she may be able to use antinausea medication sparingly to alleviate nausea while also limiting side effects from the medications—which was a big concern for her.
I ask her what else is bothering her.
She tells me that she has always been a strong Catholic and even when life has gotten tough, her faith was never shaken – until now.
She is struggling to understand why she ended up with metastatic cancer at such a relatively young age—why would God do this to her?
She had plans for retirement that have since evaporated in the face of a foreshortened life.
Why did this happen to her of all people? She was completely healthy until her diagnosis.
Her face is wet with tears.
We talk a little about how a diagnosis like this can change so much of a person’s life and identity. I try to validate her experience. She’s clearly suffering from a sense that her life is not what she expected, and she is struggling to integrate how her future looks at this point.
I ask her what conversations with her priest have been like.
At this point you may be wondering where this conversation is going. Why are we talking about Ms. Lopez’s religion? Palliative care is best delivered through high functioning interdisciplinary teams that can include other supportive people in a patient’s life. We work in concert to try to bring comfort to a patient and their family.4 That support network can include nurses, physicians, social workers, and chaplains. In this case, Ms. Lopez had not yet reached out to her priest. She hasn’t had the time or energy to contact her priest given her symptoms.
“Can I contact your priest for you?
Maybe he can visit or call and chat with you?”
She nods and wipes tears away.
“That would be really nice,” she says. “I’d love it if he could pray with me.”
A few hours after the visit, I call Ms. Lopez’s priest.
I ask him to reach out to her and about her request for prayer.
He says he’s been thinking about her and that her presence has been missed at weekly Mass. He thanks me for the call and says he’ll call her tomorrow.
I say my own small prayer for Ms. Lopez and head home, the day’s work completed.
Sarah D'Ambruoso was born and raised in Maine. She completed her undergraduate and graduate nursing education at New York University and UCLA, respectively, and currently works as a palliative care nurse practitioner in an oncology clinic in Los Angeles.
References
1. Cheng BT et al. Patterns of palliative care beliefs among adults in the U.S.: Analysis of a National Cancer Database. J Pain Symptom Manage. 2019 Aug 10. doi: 10.1016/j.jpainsymman.2019.07.030.
2. Finlay E et al. Filling the gap: Creating an outpatient palliative care program in your institution. Am Soc Clin Oncol Educ Book. 2018 May 23. doi: 10.1200/EDBK_200775.
3. Von Roenn JH et al. Barriers and approaches to the successful integration of palliative care and oncology practice. J Natl Compr Canc Netw. 2013 Mar. doi: 10.6004/jnccn.2013.0209.
4. Ferrell BR et al. Integration of palliative care into standard oncology care: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2016 Oct 31. doi: 10.1200/JCO.2016.70.1474.
The names of health care professionals and patients cited within the dialogue text have been changed to protect their privacy.
but over the years I have come to realize that she was right – most people, including many within health care, don’t have a good appreciation of what palliative care is or how it can help patients and health care teams.
A recent national survey about cancer-related health information found that of more than 1,000 surveyed Americans, less than 30% professed any knowledge of palliative care. Of those who had some knowledge of palliative care, around 30% believed palliative care was synonymous with hospice.1 Another 15% believed that a patient would have to give up cancer-directed treatments to receive palliative care.1
It’s not giving up
This persistent belief that palliative care is equivalent to hospice, or is tantamount to “giving up,” is one of the most commonly held myths I encounter in everyday practice.
I knock on the exam door and walk in.
A small, trim woman in her late 50s is sitting in a chair, arms folded across her chest, face drawn in.
“Hi,” I start. “I’m Sarah, the palliative care nurse practitioner who works in this clinic. I work closely with Dr. Smith.”
Dr. Smith is the patient’s oncologist.
“I really didn’t want to meet you,” she says in a quiet voice, her eyes large with concern.
I don’t take it personally. Few patients really want to be in the position of needing to meet the palliative care team.
“I looked up palliative care on Google and saw the word hospice.”
“Yeah,” I say. “I hear that a lot. Well, I can reassure you that this isn’t hospice.
In this clinic, our focus is on your cancer symptoms, your treatment side effects, and your quality of life.”
She looks visibly relieved. “Quality of life,” she echoes. “I need more of that.”
“OK,” I say. “So, tell me what you’re struggling with the most right now.”
That’s how many palliative care visits start. I actually prefer if patients haven’t heard of palliative care because it allows me to frame it for them, rather than having to start by addressing a myth or a prior negative experience. Even when patients haven’t had a negative experience with palliative care per se, typically, if they’ve interacted with palliative care in the past, it’s usually because someone they loved died in a hospital setting and it is the memory of that terrible loss that becomes synonymous with their recollection of palliative care.
Many patients I meet have never seen another outpatient palliative care practitioner – and this makes sense – we are still too few and far between. Most established palliative care teams are hospital based and many patients seen in the community do not have easy access to palliative care teams where they receive oncologic care.2 As an embedded practitioner, I see patients in the same exam rooms and infusion centers where they receive their cancer therapies, so I’m effectively woven into the fabric of their oncology experience. Just being there in the cancer center allows me to be in the right place at the right time for the right patients and their care teams.
More than pain management
Another myth I tend to dispel a lot is that palliative care is just a euphemism for “pain management.” I have seen this less lately, but still occasionally in the chart I’ll see documented in a note, “patient is seeing palliative/pain management,” when a patient is seeing me or one of my colleagues. Unfortunately, when providers have limited or outdated views of what palliative care is or the value it brings to patient-centered cancer care, referrals to palliative care tend to be delayed.3
“I really think Ms. Lopez could benefit from seeing palliative care,” an oncology nurse practitioner says to an oncologist.
I’m standing nearby, about to see another patient in one of the exam rooms in our clinic.
“But I don’t think she’s ready. And besides, she doesn’t have any pain,” he says.
He turns to me quizzically. “What do you think?”
“Tell me about the patient,” I ask, taking a few steps in their direction.
“Well, she’s a 64-year-old woman with metastatic cancer.
She has a really poor appetite and is losing some weight.
Seems a bit down, kind of pessimistic about things.
Her scan showed some new growth, so guess I’m not surprised by that.”
“I might be able to help her with the appetite and the mood changes.
I can at least talk with her and see where she’s at,” I offer.
“Alright,” he says. “We’ll put the palliative referral in.”
He hesitates. “But are you sure you want to see her?
She doesn’t have any pain.” He sounds skeptical.
“Yeah, I mean, it sounds like she has symptoms that are bothering her, so I’d be happy to see her. She sounds completely appropriate for palliative care.”
I hear this assumption a lot – that palliative care is somehow equivalent to pain management and that unless a patient’s pain is severe, it’s not worth referring the patient to palliative care. Don’t get me wrong – we do a lot of pain management, but at its heart, palliative care is an interdisciplinary specialty focused on improving or maintaining quality of life for people with serious illness. Because the goal is so broad, care can take many shapes.4
In addition to pain, palliative care clinicians commonly treat nausea, shortness of breath, constipation or diarrhea, poor appetite, fatigue, anxiety, depression, and insomnia.
Palliative care is more than medical or nursing care
A related misconception about palliative care held by many lay people and health care workers alike is that palliative care is primarily medical or nursing care focused mostly on alleviating physical symptoms such as pain or nausea. This couldn’t be further from the truth.
We’ve been talking for a while.
Ms. Lopez tells me about her struggles to maintain her weight while undergoing chemotherapy. She has low-grade nausea that is impacting her ability and desire to eat more and didn’t think that her weight loss was severe enough to warrant taking medication.
We talk about how she may be able to use antinausea medication sparingly to alleviate nausea while also limiting side effects from the medications—which was a big concern for her.
I ask her what else is bothering her.
She tells me that she has always been a strong Catholic and even when life has gotten tough, her faith was never shaken – until now.
She is struggling to understand why she ended up with metastatic cancer at such a relatively young age—why would God do this to her?
She had plans for retirement that have since evaporated in the face of a foreshortened life.
Why did this happen to her of all people? She was completely healthy until her diagnosis.
Her face is wet with tears.
We talk a little about how a diagnosis like this can change so much of a person’s life and identity. I try to validate her experience. She’s clearly suffering from a sense that her life is not what she expected, and she is struggling to integrate how her future looks at this point.
I ask her what conversations with her priest have been like.
At this point you may be wondering where this conversation is going. Why are we talking about Ms. Lopez’s religion? Palliative care is best delivered through high functioning interdisciplinary teams that can include other supportive people in a patient’s life. We work in concert to try to bring comfort to a patient and their family.4 That support network can include nurses, physicians, social workers, and chaplains. In this case, Ms. Lopez had not yet reached out to her priest. She hasn’t had the time or energy to contact her priest given her symptoms.
“Can I contact your priest for you?
Maybe he can visit or call and chat with you?”
She nods and wipes tears away.
“That would be really nice,” she says. “I’d love it if he could pray with me.”
A few hours after the visit, I call Ms. Lopez’s priest.
I ask him to reach out to her and about her request for prayer.
He says he’s been thinking about her and that her presence has been missed at weekly Mass. He thanks me for the call and says he’ll call her tomorrow.
I say my own small prayer for Ms. Lopez and head home, the day’s work completed.
Sarah D'Ambruoso was born and raised in Maine. She completed her undergraduate and graduate nursing education at New York University and UCLA, respectively, and currently works as a palliative care nurse practitioner in an oncology clinic in Los Angeles.
References
1. Cheng BT et al. Patterns of palliative care beliefs among adults in the U.S.: Analysis of a National Cancer Database. J Pain Symptom Manage. 2019 Aug 10. doi: 10.1016/j.jpainsymman.2019.07.030.
2. Finlay E et al. Filling the gap: Creating an outpatient palliative care program in your institution. Am Soc Clin Oncol Educ Book. 2018 May 23. doi: 10.1200/EDBK_200775.
3. Von Roenn JH et al. Barriers and approaches to the successful integration of palliative care and oncology practice. J Natl Compr Canc Netw. 2013 Mar. doi: 10.6004/jnccn.2013.0209.
4. Ferrell BR et al. Integration of palliative care into standard oncology care: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2016 Oct 31. doi: 10.1200/JCO.2016.70.1474.
The names of health care professionals and patients cited within the dialogue text have been changed to protect their privacy.
but over the years I have come to realize that she was right – most people, including many within health care, don’t have a good appreciation of what palliative care is or how it can help patients and health care teams.
A recent national survey about cancer-related health information found that of more than 1,000 surveyed Americans, less than 30% professed any knowledge of palliative care. Of those who had some knowledge of palliative care, around 30% believed palliative care was synonymous with hospice.1 Another 15% believed that a patient would have to give up cancer-directed treatments to receive palliative care.1
It’s not giving up
This persistent belief that palliative care is equivalent to hospice, or is tantamount to “giving up,” is one of the most commonly held myths I encounter in everyday practice.
I knock on the exam door and walk in.
A small, trim woman in her late 50s is sitting in a chair, arms folded across her chest, face drawn in.
“Hi,” I start. “I’m Sarah, the palliative care nurse practitioner who works in this clinic. I work closely with Dr. Smith.”
Dr. Smith is the patient’s oncologist.
“I really didn’t want to meet you,” she says in a quiet voice, her eyes large with concern.
I don’t take it personally. Few patients really want to be in the position of needing to meet the palliative care team.
“I looked up palliative care on Google and saw the word hospice.”
“Yeah,” I say. “I hear that a lot. Well, I can reassure you that this isn’t hospice.
In this clinic, our focus is on your cancer symptoms, your treatment side effects, and your quality of life.”
She looks visibly relieved. “Quality of life,” she echoes. “I need more of that.”
“OK,” I say. “So, tell me what you’re struggling with the most right now.”
That’s how many palliative care visits start. I actually prefer if patients haven’t heard of palliative care because it allows me to frame it for them, rather than having to start by addressing a myth or a prior negative experience. Even when patients haven’t had a negative experience with palliative care per se, typically, if they’ve interacted with palliative care in the past, it’s usually because someone they loved died in a hospital setting and it is the memory of that terrible loss that becomes synonymous with their recollection of palliative care.
Many patients I meet have never seen another outpatient palliative care practitioner – and this makes sense – we are still too few and far between. Most established palliative care teams are hospital based and many patients seen in the community do not have easy access to palliative care teams where they receive oncologic care.2 As an embedded practitioner, I see patients in the same exam rooms and infusion centers where they receive their cancer therapies, so I’m effectively woven into the fabric of their oncology experience. Just being there in the cancer center allows me to be in the right place at the right time for the right patients and their care teams.
More than pain management
Another myth I tend to dispel a lot is that palliative care is just a euphemism for “pain management.” I have seen this less lately, but still occasionally in the chart I’ll see documented in a note, “patient is seeing palliative/pain management,” when a patient is seeing me or one of my colleagues. Unfortunately, when providers have limited or outdated views of what palliative care is or the value it brings to patient-centered cancer care, referrals to palliative care tend to be delayed.3
“I really think Ms. Lopez could benefit from seeing palliative care,” an oncology nurse practitioner says to an oncologist.
I’m standing nearby, about to see another patient in one of the exam rooms in our clinic.
“But I don’t think she’s ready. And besides, she doesn’t have any pain,” he says.
He turns to me quizzically. “What do you think?”
“Tell me about the patient,” I ask, taking a few steps in their direction.
“Well, she’s a 64-year-old woman with metastatic cancer.
She has a really poor appetite and is losing some weight.
Seems a bit down, kind of pessimistic about things.
Her scan showed some new growth, so guess I’m not surprised by that.”
“I might be able to help her with the appetite and the mood changes.
I can at least talk with her and see where she’s at,” I offer.
“Alright,” he says. “We’ll put the palliative referral in.”
He hesitates. “But are you sure you want to see her?
She doesn’t have any pain.” He sounds skeptical.
“Yeah, I mean, it sounds like she has symptoms that are bothering her, so I’d be happy to see her. She sounds completely appropriate for palliative care.”
I hear this assumption a lot – that palliative care is somehow equivalent to pain management and that unless a patient’s pain is severe, it’s not worth referring the patient to palliative care. Don’t get me wrong – we do a lot of pain management, but at its heart, palliative care is an interdisciplinary specialty focused on improving or maintaining quality of life for people with serious illness. Because the goal is so broad, care can take many shapes.4
In addition to pain, palliative care clinicians commonly treat nausea, shortness of breath, constipation or diarrhea, poor appetite, fatigue, anxiety, depression, and insomnia.
Palliative care is more than medical or nursing care
A related misconception about palliative care held by many lay people and health care workers alike is that palliative care is primarily medical or nursing care focused mostly on alleviating physical symptoms such as pain or nausea. This couldn’t be further from the truth.
We’ve been talking for a while.
Ms. Lopez tells me about her struggles to maintain her weight while undergoing chemotherapy. She has low-grade nausea that is impacting her ability and desire to eat more and didn’t think that her weight loss was severe enough to warrant taking medication.
We talk about how she may be able to use antinausea medication sparingly to alleviate nausea while also limiting side effects from the medications—which was a big concern for her.
I ask her what else is bothering her.
She tells me that she has always been a strong Catholic and even when life has gotten tough, her faith was never shaken – until now.
She is struggling to understand why she ended up with metastatic cancer at such a relatively young age—why would God do this to her?
She had plans for retirement that have since evaporated in the face of a foreshortened life.
Why did this happen to her of all people? She was completely healthy until her diagnosis.
Her face is wet with tears.
We talk a little about how a diagnosis like this can change so much of a person’s life and identity. I try to validate her experience. She’s clearly suffering from a sense that her life is not what she expected, and she is struggling to integrate how her future looks at this point.
I ask her what conversations with her priest have been like.
At this point you may be wondering where this conversation is going. Why are we talking about Ms. Lopez’s religion? Palliative care is best delivered through high functioning interdisciplinary teams that can include other supportive people in a patient’s life. We work in concert to try to bring comfort to a patient and their family.4 That support network can include nurses, physicians, social workers, and chaplains. In this case, Ms. Lopez had not yet reached out to her priest. She hasn’t had the time or energy to contact her priest given her symptoms.
“Can I contact your priest for you?
Maybe he can visit or call and chat with you?”
She nods and wipes tears away.
“That would be really nice,” she says. “I’d love it if he could pray with me.”
A few hours after the visit, I call Ms. Lopez’s priest.
I ask him to reach out to her and about her request for prayer.
He says he’s been thinking about her and that her presence has been missed at weekly Mass. He thanks me for the call and says he’ll call her tomorrow.
I say my own small prayer for Ms. Lopez and head home, the day’s work completed.
Sarah D'Ambruoso was born and raised in Maine. She completed her undergraduate and graduate nursing education at New York University and UCLA, respectively, and currently works as a palliative care nurse practitioner in an oncology clinic in Los Angeles.
References
1. Cheng BT et al. Patterns of palliative care beliefs among adults in the U.S.: Analysis of a National Cancer Database. J Pain Symptom Manage. 2019 Aug 10. doi: 10.1016/j.jpainsymman.2019.07.030.
2. Finlay E et al. Filling the gap: Creating an outpatient palliative care program in your institution. Am Soc Clin Oncol Educ Book. 2018 May 23. doi: 10.1200/EDBK_200775.
3. Von Roenn JH et al. Barriers and approaches to the successful integration of palliative care and oncology practice. J Natl Compr Canc Netw. 2013 Mar. doi: 10.6004/jnccn.2013.0209.
4. Ferrell BR et al. Integration of palliative care into standard oncology care: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2016 Oct 31. doi: 10.1200/JCO.2016.70.1474.
Withholding anticoagulation for isolated subsegmental pulmonary embolism – Houston, we have a problem
All else being equal, I’d prefer to do nothing. Whether this is nihilism, laziness, or experience is a matter of debate. The American College of Chest Physicians (CHEST) Guidelines on therapy for venous thromboembolism (VTE) opened a door for withholding treatment for isolated subsegmental pulmonary embolism (ISSPE) in 2016 and kept it open in 2021. I was happy to walk through it and withhold therapy if it wasn’t indicated.
ISSPE is truly a conundrum. With advances in technology, the distal vessels in the lung became visible on commercial CT a little more than 10 years ago. The subsegmental branches are located after the fourth bifurcation of the pulmonary arterial system, and the new technology offered resolution adequate to identify clot in these vessels. But the new technology told us nothing about how to manage clot isolated to the subsegmental vasculature.
Autopsy data say clot in these vessels is common, even in patients who were never diagnosed with VTE while they were alive. To some degree then, the pulmonary arterial system is thought to serve as a filter to prevent clot from crossing to the systemic circulation and causing stroke. This led some to speculate that the subsegmental pulmonary arteries are supposed to contain clot and that we simply couldn’t see it before now. If this theory is correct, the practice of providing anticoagulation for ISSPE could increase bleeding without reducing the risk for VTE recurrence.
Management studies generally supported this concept. In 2007, a trial that was published in JAMA randomized patients to two different diagnostic strategies: ventilation-perfusion (VQ) and CT. CT detected more clot than VQ did, so more anticoagulation was given in the CT arm. Yet, the VTE rate during follow-up was not significantly different between arms. The implication? Some of the clots detected by CT were of lesser clinical significance and didn’t need to be treated.
Meta-analytic data from management trials also suggested that some pulmonary emboli (PE) need not be treated. Data also show when compared with patients who have more proximal PE, those with ISSPE have lower pretest probability for VTE, are less symptomatic, and have a lower burden of coexistent lower extremity thrombosis (deep vein thrombosis [DVT]).
In response to this data, the CHEST Guidelines began cautiously providing the option for withholding therapy in patients who were diagnosed with ISSPE in 2016. Their recommendations stated that patients should be stratified for recurrence risk and have lower extremity ultrasonography performed to rule out DVT. A patient with ISSPE, a low recurrence risk, and a negative ultrasound can have anticoagulation withheld. This made perfect sense to me based on what I thought I knew at the time.
Recently published data cast doubt on my nihilism. The first prospective study designed specifically to assess the safety of withholding therapy for ISSPE suggests that this practice could be dangerous. How did this happen? The trial was very well done, and the authors enrolled the right population. All of the patients had ISSPE, low recurrence risk, and negative lower extremity ultrasound. The authors were anticipating a 1% VTE rate at 90 days based on prior data but instead found a rate of 3.1% (1.6%-6.1%). They point out that this rate is not different from those seen in patients with more proximal PE who are treated with anticoagulation. However, they acknowledge that it is higher than what’s considered acceptable and warrants therapeutic anticoagulation.
So what should we do now? We treat ISSPE, that’s what. All the arguments for withholding therapy remain valid, the recurrence rate is reasonably low, and none of the recurrent VTEs in the new study were fatal. There’s still no doubt that some patients with PE won’t benefit from anticoagulation. Unfortunately, we currently lack the tools to identify them. The risk-benefit ratio for recurrence versus bleeding will be tighter with ISSPE, particularly when there’s only one clot. Unless the bleeding risk is elevated though, the ratio still favors treatment.
Aaron B. Holley, MD, is an associate professor of medicine at Uniformed Services University and program director of pulmonary and critical care medicine at Walter Reed National Military Medical Center.
A version of this article first appeared on Medscape.com.
All else being equal, I’d prefer to do nothing. Whether this is nihilism, laziness, or experience is a matter of debate. The American College of Chest Physicians (CHEST) Guidelines on therapy for venous thromboembolism (VTE) opened a door for withholding treatment for isolated subsegmental pulmonary embolism (ISSPE) in 2016 and kept it open in 2021. I was happy to walk through it and withhold therapy if it wasn’t indicated.
ISSPE is truly a conundrum. With advances in technology, the distal vessels in the lung became visible on commercial CT a little more than 10 years ago. The subsegmental branches are located after the fourth bifurcation of the pulmonary arterial system, and the new technology offered resolution adequate to identify clot in these vessels. But the new technology told us nothing about how to manage clot isolated to the subsegmental vasculature.
Autopsy data say clot in these vessels is common, even in patients who were never diagnosed with VTE while they were alive. To some degree then, the pulmonary arterial system is thought to serve as a filter to prevent clot from crossing to the systemic circulation and causing stroke. This led some to speculate that the subsegmental pulmonary arteries are supposed to contain clot and that we simply couldn’t see it before now. If this theory is correct, the practice of providing anticoagulation for ISSPE could increase bleeding without reducing the risk for VTE recurrence.
Management studies generally supported this concept. In 2007, a trial that was published in JAMA randomized patients to two different diagnostic strategies: ventilation-perfusion (VQ) and CT. CT detected more clot than VQ did, so more anticoagulation was given in the CT arm. Yet, the VTE rate during follow-up was not significantly different between arms. The implication? Some of the clots detected by CT were of lesser clinical significance and didn’t need to be treated.
Meta-analytic data from management trials also suggested that some pulmonary emboli (PE) need not be treated. Data also show when compared with patients who have more proximal PE, those with ISSPE have lower pretest probability for VTE, are less symptomatic, and have a lower burden of coexistent lower extremity thrombosis (deep vein thrombosis [DVT]).
In response to this data, the CHEST Guidelines began cautiously providing the option for withholding therapy in patients who were diagnosed with ISSPE in 2016. Their recommendations stated that patients should be stratified for recurrence risk and have lower extremity ultrasonography performed to rule out DVT. A patient with ISSPE, a low recurrence risk, and a negative ultrasound can have anticoagulation withheld. This made perfect sense to me based on what I thought I knew at the time.
Recently published data cast doubt on my nihilism. The first prospective study designed specifically to assess the safety of withholding therapy for ISSPE suggests that this practice could be dangerous. How did this happen? The trial was very well done, and the authors enrolled the right population. All of the patients had ISSPE, low recurrence risk, and negative lower extremity ultrasound. The authors were anticipating a 1% VTE rate at 90 days based on prior data but instead found a rate of 3.1% (1.6%-6.1%). They point out that this rate is not different from those seen in patients with more proximal PE who are treated with anticoagulation. However, they acknowledge that it is higher than what’s considered acceptable and warrants therapeutic anticoagulation.
So what should we do now? We treat ISSPE, that’s what. All the arguments for withholding therapy remain valid, the recurrence rate is reasonably low, and none of the recurrent VTEs in the new study were fatal. There’s still no doubt that some patients with PE won’t benefit from anticoagulation. Unfortunately, we currently lack the tools to identify them. The risk-benefit ratio for recurrence versus bleeding will be tighter with ISSPE, particularly when there’s only one clot. Unless the bleeding risk is elevated though, the ratio still favors treatment.
Aaron B. Holley, MD, is an associate professor of medicine at Uniformed Services University and program director of pulmonary and critical care medicine at Walter Reed National Military Medical Center.
A version of this article first appeared on Medscape.com.
All else being equal, I’d prefer to do nothing. Whether this is nihilism, laziness, or experience is a matter of debate. The American College of Chest Physicians (CHEST) Guidelines on therapy for venous thromboembolism (VTE) opened a door for withholding treatment for isolated subsegmental pulmonary embolism (ISSPE) in 2016 and kept it open in 2021. I was happy to walk through it and withhold therapy if it wasn’t indicated.
ISSPE is truly a conundrum. With advances in technology, the distal vessels in the lung became visible on commercial CT a little more than 10 years ago. The subsegmental branches are located after the fourth bifurcation of the pulmonary arterial system, and the new technology offered resolution adequate to identify clot in these vessels. But the new technology told us nothing about how to manage clot isolated to the subsegmental vasculature.
Autopsy data say clot in these vessels is common, even in patients who were never diagnosed with VTE while they were alive. To some degree then, the pulmonary arterial system is thought to serve as a filter to prevent clot from crossing to the systemic circulation and causing stroke. This led some to speculate that the subsegmental pulmonary arteries are supposed to contain clot and that we simply couldn’t see it before now. If this theory is correct, the practice of providing anticoagulation for ISSPE could increase bleeding without reducing the risk for VTE recurrence.
Management studies generally supported this concept. In 2007, a trial that was published in JAMA randomized patients to two different diagnostic strategies: ventilation-perfusion (VQ) and CT. CT detected more clot than VQ did, so more anticoagulation was given in the CT arm. Yet, the VTE rate during follow-up was not significantly different between arms. The implication? Some of the clots detected by CT were of lesser clinical significance and didn’t need to be treated.
Meta-analytic data from management trials also suggested that some pulmonary emboli (PE) need not be treated. Data also show when compared with patients who have more proximal PE, those with ISSPE have lower pretest probability for VTE, are less symptomatic, and have a lower burden of coexistent lower extremity thrombosis (deep vein thrombosis [DVT]).
In response to this data, the CHEST Guidelines began cautiously providing the option for withholding therapy in patients who were diagnosed with ISSPE in 2016. Their recommendations stated that patients should be stratified for recurrence risk and have lower extremity ultrasonography performed to rule out DVT. A patient with ISSPE, a low recurrence risk, and a negative ultrasound can have anticoagulation withheld. This made perfect sense to me based on what I thought I knew at the time.
Recently published data cast doubt on my nihilism. The first prospective study designed specifically to assess the safety of withholding therapy for ISSPE suggests that this practice could be dangerous. How did this happen? The trial was very well done, and the authors enrolled the right population. All of the patients had ISSPE, low recurrence risk, and negative lower extremity ultrasound. The authors were anticipating a 1% VTE rate at 90 days based on prior data but instead found a rate of 3.1% (1.6%-6.1%). They point out that this rate is not different from those seen in patients with more proximal PE who are treated with anticoagulation. However, they acknowledge that it is higher than what’s considered acceptable and warrants therapeutic anticoagulation.
So what should we do now? We treat ISSPE, that’s what. All the arguments for withholding therapy remain valid, the recurrence rate is reasonably low, and none of the recurrent VTEs in the new study were fatal. There’s still no doubt that some patients with PE won’t benefit from anticoagulation. Unfortunately, we currently lack the tools to identify them. The risk-benefit ratio for recurrence versus bleeding will be tighter with ISSPE, particularly when there’s only one clot. Unless the bleeding risk is elevated though, the ratio still favors treatment.
Aaron B. Holley, MD, is an associate professor of medicine at Uniformed Services University and program director of pulmonary and critical care medicine at Walter Reed National Military Medical Center.
A version of this article first appeared on Medscape.com.
Structural Ableism: Defining Standards of Care Amid Crisis and Inequity
Equitable Standards for All Patients in a Crisis
Health care delivered during a pandemic instantiates medicine’s perspectives on the value of human life in clinical scenarios where resource allocation is limited. The COVID-19 pandemic has fostered dialogue and debate around the ethical principles that underly such resource allocation, which generally balance (1) utilitarian optimization of resources, (2) equality or equity in health access, (3) the instrumental value of individuals as agents in society, and (4) prioritizing the “worst off” in their natural history of disease.1,2 State legislatures and health systems have responded to the challeges posed by COVID-19 by considering both the scarcity of intensive care resources, such as mechanical ventilation and hemodialysis, and the clinical criteria to be used for determining which patients should receive said resources. These crisis guidelines have yielded several concerning themes vis-à-vis equitable distribution of health care resources, particularly when the disability status of patients is considered alongside life-expectancy or quality of life.3
Crisis standards of care (CSC) prioritize population-level health under a utilitarian paradigm, explicitly maximizing “life-years” within a population of patients rather than the life of any individual patient.4 Debated during initial COVID surges, these CSC guidelines have recently been enacted at the state level in several settings, including Alaska and Idaho.5 In a setting with scarce intensive care resources, balancing health equity in access to these resources against population-based survival metrics has been a challenge for commissions considering CSC.6,7 This need for balance has further promoted systemic views of “disability,” raising concern for structural “ableism” and highlighting the need for greater “ability awareness” in clinicians’ continued professional learning.
Structural Ableism: Defining Perspectives to Address Health Equity
Ableism has been defined as “a system that places value on people’s bodies and minds, based on societally constructed ideas of normalcy, intelligence, excellence, and productivity…[and] leads to people and society determining who is valuable and worthy based on their appearance and/or their ability to satisfactorily [re]produce, excel, and ‘behave.’”8 Regarding CSC, concerns about systemic bias in guideline design were raised early by disability advocacy groups during comment periods.9,10 More broadly, concerns about ableism sit alongside many deeply rooted societal perspectives of disabled individuals as pitiable or, conversely, heroic for having “overcome” their disability in some way. As a physician who sits in a manual wheelchair with paraplegia and mobility impairment, I have equally been subject to inappropriate bias and inappropriate praise for living in a wheelchair. I have also wondered, alongside my patients living with different levels of mobility or ability, why others often view us as “worse off.” Addressing directly whether disabled individuals are “worse off,” disability rights attorney and advocate Harriet McBryde Johnson has articulated a predominant sentiment among persons living with unique or different abilities:
Are we “worse off”? I don’t think so. Not in any meaningful way. There are too many variables. For those of us with congenital conditions, disability shapes all we are. Those disabled later in life adapt. We take constraints that no one would choose and build rich and satisfying lives within them. We enjoy pleasures other people enjoy and pleasures peculiarly our own. We have something the world needs.11
Many physician colleagues have common, invisible diseases such as diabetes and heart disease; fewer colleagues share conditions that are as visible as my spinal cord injury, as readily apparent to patients upon my entry to their hospital rooms. This simultaneous and inescapable identity as both patient and provider has afforded me wonderful doctor-patient interactions, particularly with those patients who appreciate how my patient experience impacts my ability to partially understand theirs. However, this simultaneous identity as doctor and patient also informed my personal and professional concerns regarding structural ableism as I considered scoring my own acutely ill hospital medicine patients with CSC triage scores in April 2020.
As a practicing hospital medicine physician, I have been emboldened by the efforts of my fellow clinicians amid COVID-19; their efforts have reaffirmed all the reasons I pursued a career in medicine. However, when I heard my clinical colleagues’ first explanation of the Massachusetts CSC guidelines in April 2020, I raised my hand to ask whether the “life-years” to which the guidelines referred were quality-adjusted. My concern regarding the implicit use of quality-adjusted life years (QALY) or disability-adjusted life years in clinical decision-making and implementation of these guidelines was validated when no clinical leaders could address this question directly. Sitting on the CSC committee for my hospital during this time was an honor. However, it was disconcerting to hear many clinicians’ unease when estimating mean survival for common chronic diseases, ranging from end-stage renal disease to advanced heart failure. If my expert colleagues, clinical specialists in kidney and heart disease, could not confidently apply mean survival estimates to multimorbid hospital patients, then idiosyncratic clinical judgment was sure to have a heavy hand in any calculation of “life-years.” Thus, my primary concern was that clinicians using triage heuristics would be subject to bias, regardless of their intention, and negatively adjust for the quality of a disabled life in their CSC triage scoring. My secondary concern was that the CSC guidelines themselves included systemic bias against disabled individuals.
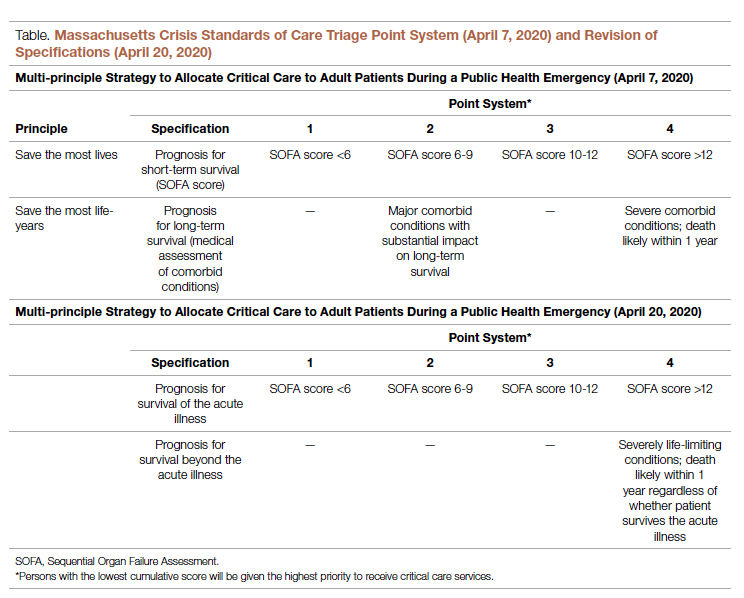
According to CSC schema, triage scores index heavily on Sequential Organ Failure Assessment (SOFA) scores to define short-term survival; SOFA scores are partially driven by the Glasgow Coma Scale (GCS). Following professional and public comment periods, CSC guidelines in Massachusetts were revised to, among other critical points of revision, change prognostic estimation via “life years” in favor of generic estimation of short-term survival (Table). I wondered, if I presented to an emergency department with severe COVID-19 and was scored with the GCS for the purpose of making a CSC ventilator triage decision, how would my complete paraplegia and lower-extremity motor impairment be accounted for by a clinician assessing “best motor response” in the GCS? The purpose of these scores is to act algorithmically, to guide clinicians whose cognitive load and time limitations may not allow for adjustment of these algorithms based on the individual patient in front of them. Individualization of clinical decisions is part of medicine’s art, but is difficult in the best of times and no easier during a crisis in care delivery. As CSC triage scores were amended and addended throughout 2020, I returned to the COVID wards, time and again wondering, “What have we learned about systemic bias and health inequity in the CSC process and the pandemic broadly, with specific regard to disability?”
Ability Awareness: Room for Our Improvement
Unfortunately, there is reason to believe that clinical judgment is impaired by structural ableism. In seminal work on this topic, Gerhart et al12 demonstrated that clinicians considered spinal cord injury (SCI) survivors to have low self-perceptions of worthiness, overall negative attitudes, and low self-esteem as compared to able-bodied individuals. However, surveyed SCI survivors generally had similar self-perceptions of worth and positivity as compared to ”able-bodied” clinicians.12 For providers who care for persons with disabilities, the majority (82.4%) have rated their disabled patients’ quality of life as worse.13 It is no wonder that patients with disabilities are more likely to feel that their doctor-patient relationship is impacted by lack of understanding, negative sentiment, or simple lack of listening.14 Generally, this poor doctor-patient relationship with disabled patients is exacerbated by poor exposure of medical trainees to disability education; only 34.2% of internal medicine residents recall any form of disability education in medical school, while only 52% of medical school deans report having disability educational content in their curricula.15,16 There is a similar lack of disability representation in the population of medical trainees themselves. While approximately 20% of the American population lives with a disability, less than 2% of American medical students have a disability.17-19
While representation of disabled populations in medical practice remains poor, disabled patients are generally less likely to receive age-appropriate prevention, appropriate access to care, and equal access to treatment.20-22 “Diagnostic overshadowing” refers to clinicians’ attribution of nonspecific signs or symptoms to a patient’s chronic disability as opposed to acute illness.23 This phenomenon has led to higher rates of preventable malignancy in disabled patients and misattribution of common somatic symptoms to intellectual disability.24,25 With this disparity in place as status quo for health care delivery to disabled populations, it is no surprise that certain portions of the disabled population have accounted for disproportionate mortality due to COVID-19.26,27Disability advocates have called for “nothing about us without us,” a phrase associated with the United Nations Convention on the Rights of Persons with Disabilities. Understanding the profound neurodiversity among several forms of sensory and cognitive disabilities, as well as the functional difference between cognitive disabilities, mobility impairment, and inability to meet one’s instrumental activities of daily living independently, others have proposed a unique approach to certain disabled populations in COVID care.28 My own perspective is that definite progress may require a more general understanding of the prevalence of disability by clinicians, both via medical training and by directly addressing health equity for disabled populations in such calculations as the CSC. Systemic ableism is apparent in our most common clinical scoring systems, ranging from the GCS and Functional Assessment Staging Table to the Eastern Cooperative Oncology Group and Karnofsky Performance Status scales. I have reexamined these scoring systems in my own understanding given their general equation of ambulation with ability or normalcy. As a doctor in a manual wheelchair who values greatly my personal quality of life and professional contribution to patient care, I worry that these scoring systems inherently discount my own equitable access to care. Individualization of patients’ particular abilities in the context of these scales must occur alongside evidence-based, guideline-directed management via these scoring systems.
Conclusion: Future Orientation
Updated CSC guidelines have accounted for the unique considerations of disabled patients by effectively caveating their scoring algorithms, directing clinicians via disclaimers to uniquely consider their disabled patients in clinical judgement. This is a first step, but it is also one that erodes the value of algorithms, which generally obviate more deliberative thinking and individualization. For our patients who lack certain abilities, as CSC continue to be activated in several states, we have an opportunity to pursue more inherently equitable solutions before further suffering accrues.29 By way of example, adaptations to scoring systems that leverage QALYs for value-based drug pricing indices have been proposed by organizations like the Institute for Clinical and Economic Review, which proposed the Equal-Value-of Life-Years-Gained framework to inform QALY-based arbitration of drug pricing.30 This is not a perfect rubric but instead represents an attempt to balance consideration of drugs, as has been done with ventilators during the pandemic, as a scare and expensive resource while addressing the just concerns of advocacy groups in structural ableism.
Resource stewardship during a crisis should not discount those states of human life that are perceived to be less desirable, particularly if they are not experienced as less desirable but are experienced uniquely. Instead, we should consider equitably measuring our intervention to match a patient’s needs, as we would dose-adjust a medication for renal function or consider minimally invasive procedures for multimorbid patients. COVID-19 has reflected our profession’s ethical adaptation during crisis as resources have become scarce; there is no better time to define solutions for health equity. We should now be concerned equally by the influence our personal biases have on our clinical practice and by the way in which these crisis standards will influence patients’ perception of and trust in their care providers during periods of perceived plentiful resources in the future. Health care resources are always limited, allocated according to societal values; if we value health equity for people of all abilities, then we will consider these abilities equitably as we pursue new standards for health care delivery.
Corresponding author: Gregory D. Snyder, MD, MBA, 2014 Washington Street, Newton, MA 02462; [email protected].
Disclosures: None.
1. Emanuel EJ, Persad G, Upshur R, et al. Fair Allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055. doi:10.1056/NEJMsb2005114
2. Savulescu J, Persson I, Wilkinson D. Utilitarianism and the pandemic. Bioethics. 2020;34(6):620-632. doi:10.1111/bioe.12771
3. Mello MM, Persad G, White DB. Respecting disability rights - toward improved crisis standards of care. N Engl J Med. 2020;383(5):e26. doi: 10.1056/NEJMp2011997
4. The Commonwealth of Massachusetts Executive Office of Health and Human Services Department of Public Health. Crisis Standards of Care Planning Guidance for the COVID-19 Pandemic. April 7, 2020. https://d279m997dpfwgl.cloudfront.net/wp/2020/04/CSC_April-7_2020.pdf
5. Knowles H. Hospitals overwhelmed by covid are turning to ‘crisis standards of care.’ What does that mean? The Washington Post. September 21, 2021. Accessed January 24, 2022. https://www.washingtonpost.com/health/2021/09/22/crisis-standards-of-care/
6. Hick JL, Hanfling D, Wynia MK, Toner E. Crisis standards of care and COVID-19: What did we learn? How do we ensure equity? What should we do? NAM Perspect. 2021;2021:10.31478/202108e. doi:10.31478/202108e
7. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
8. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
9. Kukla E. My life is more ‘disposable’ during this pandemic. The New York Times. March 19, 2020. Accessed January 24, 2022. https://www.nytimes.com/2020/03/19/opinion/coronavirus-disabled-health-care.html
10. CPR and Coalition Partners Secure Important Changes in Massachusetts’ Crisis Standards of Care. Center for Public Representation. December 1, 2020. Accessed January 24, 2022. https://www.centerforpublicrep.org/news/cpr-and-coalition-partners-secure-important-changes-in-massachusetts-crisis-standards-of-care/
11. Johnson HM. Unspeakable conversations. The New York Times. February 16, 2003. Accessed January 24, 2022. https://www.nytimes.com/2003/02/16/magazine/unspeakable-conversations.html
12. Gerhart KA, Koziol-McLain J, Lowenstein SR, Whiteneck GG. Quality of life following spinal cord injury: knowledge and attitudes of emergency care providers. Ann Emerg Med. 1994;23(4):807-812. doi:10.1016/s0196-0644(94)70318-3
13. Iezzoni LI, Rao SR, Ressalam J, et al. Physicians’ perceptions of people with disability and their health care. Health Aff (Millwood). 2021;40(2):297-306. doi:10.1377/hlthaff.2020.01452
14. Smith DL. Disparities in patient-physician communication for persons with a disability from the 2006 Medical Expenditure Panel Survey (MEPS). Disabil Health J. 2009;2(4):206-215. doi:10.1016/j.dhjo.2009.06.002
15. Stillman MD, Ankam N, Mallow M, Capron M, Williams S. A survey of internal and family medicine residents: Assessment of disability-specific education and knowledge. Disabil Health J. 2021;14(2):101011. doi:10.1016/j.dhjo.2020.101011
16. Seidel E, Crowe S. The state of disability awareness in American medical schools. Am J Phys Med Rehabil. 2017;96(9):673-676. doi:10.1097/PHM.0000000000000719
17. Okoro CA, Hollis ND, Cyrus AC, Griffin-Blake S. Prevalence of disabilities and health care access by disability status and type among adults - United States, 2016. MMWR Morb Mortal Wkly Rep. 2018;67(32):882-887. doi:10.15585/mmwr.mm6732a3
18. Peacock G, Iezzoni LI, Harkin TR. Health care for Americans with disabilities--25 years after the ADA. N Engl J Med. 2015;373(10):892-893. doi:10.1056/NEJMp1508854
19. DeLisa JA, Thomas P. Physicians with disabilities and the physician workforce: a need to reassess our policies. Am J Phys Med Rehabil. 2005;84(1):5-11. doi:10.1097/01.phm.0000153323.28396.de
20. Disability and Health. Healthy People 2020. Accessed January 24, 2022. https://www.healthypeople.gov/2020/topics-objectives/topic/disability-and-health
21. Lagu T, Hannon NS, Rothberg MB, et al. Access to subspecialty care for patients with mobility impairment: a survey. Ann Intern Med. 2013;158(6):441-446. doi: 10.7326/0003-4819-158-6-201303190-00003
22. McCarthy EP, Ngo LH, Roetzheim RG, et al. Disparities in breast cancer treatment and survival for women with disabilities. Ann Intern Med. 2006;145(9):637-645. doi: 10.7326/0003-4819-145-9-200611070-00005
23. Javaid A, Nakata V, Michael D. Diagnostic overshadowing in learning disability: think beyond the disability. Prog Neurol Psychiatry. 2019;23:8-10.
24. Iezzoni LI, Rao SR, Agaronnik ND, El-Jawahri A. Cross-sectional analysis of the associations between four common cancers and disability. J Natl Compr Canc Netw. 2020;18(8):1031-1044. doi:10.6004/jnccn.2020.7551
25. Sanders JS, Keller S, Aravamuthan BR. Caring for individuals with intellectual and developmental disabilities in the COVID-19 crisis. Neurol Clin Pract. 2021;11(2):e174-e178. doi:10.1212/CPJ.0000000000000886
26. Landes SD, Turk MA, Formica MK, McDonald KE, Stevens JD. COVID-19 outcomes among people with intellectual and developmental disability living in residential group homes in New York State. Disabil Health J. 2020;13(4):100969. doi:10.1016/j.dhjo.2020.100969
27. Gleason J, Ross W, Fossi A, Blonksy H, Tobias J, Stephens M. The devastating impact of Covid-19 on individuals with intellectual disabilities in the United States. NEJM Catalyst. 2021.doi.org/10.1056/CAT.21.0051
28. Nankervis K, Chan J. Applying the CRPD to people with intellectual and developmental disability with behaviors of concern during COVID-19. J Policy Pract Intellect Disabil. 2021:10.1111/jppi.12374. doi:10.1111/jppi.12374
29. Alaska Department of Health and Social Services, Division of Public Health, Rural and Community Health Systems. Patient care strategies for scarce resource situations. Version 1. August 2021. Accessed November 11, 2021, https://dhss.alaska.gov/dph/Epi/id/SiteAssets/Pages/HumanCoV/SOA_DHSS_CrisisStandardsOfCare.pdf
30. Cost-effectiveness, the QALY, and the evlyg. ICER. May 21, 2021. Accessed January 24, 2022. https://icer.org/our-approach/methods-process/cost-effectiveness-the-qaly-and-the-evlyg/
Equitable Standards for All Patients in a Crisis
Health care delivered during a pandemic instantiates medicine’s perspectives on the value of human life in clinical scenarios where resource allocation is limited. The COVID-19 pandemic has fostered dialogue and debate around the ethical principles that underly such resource allocation, which generally balance (1) utilitarian optimization of resources, (2) equality or equity in health access, (3) the instrumental value of individuals as agents in society, and (4) prioritizing the “worst off” in their natural history of disease.1,2 State legislatures and health systems have responded to the challeges posed by COVID-19 by considering both the scarcity of intensive care resources, such as mechanical ventilation and hemodialysis, and the clinical criteria to be used for determining which patients should receive said resources. These crisis guidelines have yielded several concerning themes vis-à-vis equitable distribution of health care resources, particularly when the disability status of patients is considered alongside life-expectancy or quality of life.3
Crisis standards of care (CSC) prioritize population-level health under a utilitarian paradigm, explicitly maximizing “life-years” within a population of patients rather than the life of any individual patient.4 Debated during initial COVID surges, these CSC guidelines have recently been enacted at the state level in several settings, including Alaska and Idaho.5 In a setting with scarce intensive care resources, balancing health equity in access to these resources against population-based survival metrics has been a challenge for commissions considering CSC.6,7 This need for balance has further promoted systemic views of “disability,” raising concern for structural “ableism” and highlighting the need for greater “ability awareness” in clinicians’ continued professional learning.
Structural Ableism: Defining Perspectives to Address Health Equity
Ableism has been defined as “a system that places value on people’s bodies and minds, based on societally constructed ideas of normalcy, intelligence, excellence, and productivity…[and] leads to people and society determining who is valuable and worthy based on their appearance and/or their ability to satisfactorily [re]produce, excel, and ‘behave.’”8 Regarding CSC, concerns about systemic bias in guideline design were raised early by disability advocacy groups during comment periods.9,10 More broadly, concerns about ableism sit alongside many deeply rooted societal perspectives of disabled individuals as pitiable or, conversely, heroic for having “overcome” their disability in some way. As a physician who sits in a manual wheelchair with paraplegia and mobility impairment, I have equally been subject to inappropriate bias and inappropriate praise for living in a wheelchair. I have also wondered, alongside my patients living with different levels of mobility or ability, why others often view us as “worse off.” Addressing directly whether disabled individuals are “worse off,” disability rights attorney and advocate Harriet McBryde Johnson has articulated a predominant sentiment among persons living with unique or different abilities:
Are we “worse off”? I don’t think so. Not in any meaningful way. There are too many variables. For those of us with congenital conditions, disability shapes all we are. Those disabled later in life adapt. We take constraints that no one would choose and build rich and satisfying lives within them. We enjoy pleasures other people enjoy and pleasures peculiarly our own. We have something the world needs.11
Many physician colleagues have common, invisible diseases such as diabetes and heart disease; fewer colleagues share conditions that are as visible as my spinal cord injury, as readily apparent to patients upon my entry to their hospital rooms. This simultaneous and inescapable identity as both patient and provider has afforded me wonderful doctor-patient interactions, particularly with those patients who appreciate how my patient experience impacts my ability to partially understand theirs. However, this simultaneous identity as doctor and patient also informed my personal and professional concerns regarding structural ableism as I considered scoring my own acutely ill hospital medicine patients with CSC triage scores in April 2020.
As a practicing hospital medicine physician, I have been emboldened by the efforts of my fellow clinicians amid COVID-19; their efforts have reaffirmed all the reasons I pursued a career in medicine. However, when I heard my clinical colleagues’ first explanation of the Massachusetts CSC guidelines in April 2020, I raised my hand to ask whether the “life-years” to which the guidelines referred were quality-adjusted. My concern regarding the implicit use of quality-adjusted life years (QALY) or disability-adjusted life years in clinical decision-making and implementation of these guidelines was validated when no clinical leaders could address this question directly. Sitting on the CSC committee for my hospital during this time was an honor. However, it was disconcerting to hear many clinicians’ unease when estimating mean survival for common chronic diseases, ranging from end-stage renal disease to advanced heart failure. If my expert colleagues, clinical specialists in kidney and heart disease, could not confidently apply mean survival estimates to multimorbid hospital patients, then idiosyncratic clinical judgment was sure to have a heavy hand in any calculation of “life-years.” Thus, my primary concern was that clinicians using triage heuristics would be subject to bias, regardless of their intention, and negatively adjust for the quality of a disabled life in their CSC triage scoring. My secondary concern was that the CSC guidelines themselves included systemic bias against disabled individuals.
According to CSC schema, triage scores index heavily on Sequential Organ Failure Assessment (SOFA) scores to define short-term survival; SOFA scores are partially driven by the Glasgow Coma Scale (GCS). Following professional and public comment periods, CSC guidelines in Massachusetts were revised to, among other critical points of revision, change prognostic estimation via “life years” in favor of generic estimation of short-term survival (Table). I wondered, if I presented to an emergency department with severe COVID-19 and was scored with the GCS for the purpose of making a CSC ventilator triage decision, how would my complete paraplegia and lower-extremity motor impairment be accounted for by a clinician assessing “best motor response” in the GCS? The purpose of these scores is to act algorithmically, to guide clinicians whose cognitive load and time limitations may not allow for adjustment of these algorithms based on the individual patient in front of them. Individualization of clinical decisions is part of medicine’s art, but is difficult in the best of times and no easier during a crisis in care delivery. As CSC triage scores were amended and addended throughout 2020, I returned to the COVID wards, time and again wondering, “What have we learned about systemic bias and health inequity in the CSC process and the pandemic broadly, with specific regard to disability?”

Ability Awareness: Room for Our Improvement
Unfortunately, there is reason to believe that clinical judgment is impaired by structural ableism. In seminal work on this topic, Gerhart et al12 demonstrated that clinicians considered spinal cord injury (SCI) survivors to have low self-perceptions of worthiness, overall negative attitudes, and low self-esteem as compared to able-bodied individuals. However, surveyed SCI survivors generally had similar self-perceptions of worth and positivity as compared to ”able-bodied” clinicians.12 For providers who care for persons with disabilities, the majority (82.4%) have rated their disabled patients’ quality of life as worse.13 It is no wonder that patients with disabilities are more likely to feel that their doctor-patient relationship is impacted by lack of understanding, negative sentiment, or simple lack of listening.14 Generally, this poor doctor-patient relationship with disabled patients is exacerbated by poor exposure of medical trainees to disability education; only 34.2% of internal medicine residents recall any form of disability education in medical school, while only 52% of medical school deans report having disability educational content in their curricula.15,16 There is a similar lack of disability representation in the population of medical trainees themselves. While approximately 20% of the American population lives with a disability, less than 2% of American medical students have a disability.17-19
While representation of disabled populations in medical practice remains poor, disabled patients are generally less likely to receive age-appropriate prevention, appropriate access to care, and equal access to treatment.20-22 “Diagnostic overshadowing” refers to clinicians’ attribution of nonspecific signs or symptoms to a patient’s chronic disability as opposed to acute illness.23 This phenomenon has led to higher rates of preventable malignancy in disabled patients and misattribution of common somatic symptoms to intellectual disability.24,25 With this disparity in place as status quo for health care delivery to disabled populations, it is no surprise that certain portions of the disabled population have accounted for disproportionate mortality due to COVID-19.26,27Disability advocates have called for “nothing about us without us,” a phrase associated with the United Nations Convention on the Rights of Persons with Disabilities. Understanding the profound neurodiversity among several forms of sensory and cognitive disabilities, as well as the functional difference between cognitive disabilities, mobility impairment, and inability to meet one’s instrumental activities of daily living independently, others have proposed a unique approach to certain disabled populations in COVID care.28 My own perspective is that definite progress may require a more general understanding of the prevalence of disability by clinicians, both via medical training and by directly addressing health equity for disabled populations in such calculations as the CSC. Systemic ableism is apparent in our most common clinical scoring systems, ranging from the GCS and Functional Assessment Staging Table to the Eastern Cooperative Oncology Group and Karnofsky Performance Status scales. I have reexamined these scoring systems in my own understanding given their general equation of ambulation with ability or normalcy. As a doctor in a manual wheelchair who values greatly my personal quality of life and professional contribution to patient care, I worry that these scoring systems inherently discount my own equitable access to care. Individualization of patients’ particular abilities in the context of these scales must occur alongside evidence-based, guideline-directed management via these scoring systems.
Conclusion: Future Orientation
Updated CSC guidelines have accounted for the unique considerations of disabled patients by effectively caveating their scoring algorithms, directing clinicians via disclaimers to uniquely consider their disabled patients in clinical judgement. This is a first step, but it is also one that erodes the value of algorithms, which generally obviate more deliberative thinking and individualization. For our patients who lack certain abilities, as CSC continue to be activated in several states, we have an opportunity to pursue more inherently equitable solutions before further suffering accrues.29 By way of example, adaptations to scoring systems that leverage QALYs for value-based drug pricing indices have been proposed by organizations like the Institute for Clinical and Economic Review, which proposed the Equal-Value-of Life-Years-Gained framework to inform QALY-based arbitration of drug pricing.30 This is not a perfect rubric but instead represents an attempt to balance consideration of drugs, as has been done with ventilators during the pandemic, as a scare and expensive resource while addressing the just concerns of advocacy groups in structural ableism.
Resource stewardship during a crisis should not discount those states of human life that are perceived to be less desirable, particularly if they are not experienced as less desirable but are experienced uniquely. Instead, we should consider equitably measuring our intervention to match a patient’s needs, as we would dose-adjust a medication for renal function or consider minimally invasive procedures for multimorbid patients. COVID-19 has reflected our profession’s ethical adaptation during crisis as resources have become scarce; there is no better time to define solutions for health equity. We should now be concerned equally by the influence our personal biases have on our clinical practice and by the way in which these crisis standards will influence patients’ perception of and trust in their care providers during periods of perceived plentiful resources in the future. Health care resources are always limited, allocated according to societal values; if we value health equity for people of all abilities, then we will consider these abilities equitably as we pursue new standards for health care delivery.
Corresponding author: Gregory D. Snyder, MD, MBA, 2014 Washington Street, Newton, MA 02462; [email protected].
Disclosures: None.
Equitable Standards for All Patients in a Crisis
Health care delivered during a pandemic instantiates medicine’s perspectives on the value of human life in clinical scenarios where resource allocation is limited. The COVID-19 pandemic has fostered dialogue and debate around the ethical principles that underly such resource allocation, which generally balance (1) utilitarian optimization of resources, (2) equality or equity in health access, (3) the instrumental value of individuals as agents in society, and (4) prioritizing the “worst off” in their natural history of disease.1,2 State legislatures and health systems have responded to the challeges posed by COVID-19 by considering both the scarcity of intensive care resources, such as mechanical ventilation and hemodialysis, and the clinical criteria to be used for determining which patients should receive said resources. These crisis guidelines have yielded several concerning themes vis-à-vis equitable distribution of health care resources, particularly when the disability status of patients is considered alongside life-expectancy or quality of life.3
Crisis standards of care (CSC) prioritize population-level health under a utilitarian paradigm, explicitly maximizing “life-years” within a population of patients rather than the life of any individual patient.4 Debated during initial COVID surges, these CSC guidelines have recently been enacted at the state level in several settings, including Alaska and Idaho.5 In a setting with scarce intensive care resources, balancing health equity in access to these resources against population-based survival metrics has been a challenge for commissions considering CSC.6,7 This need for balance has further promoted systemic views of “disability,” raising concern for structural “ableism” and highlighting the need for greater “ability awareness” in clinicians’ continued professional learning.
Structural Ableism: Defining Perspectives to Address Health Equity
Ableism has been defined as “a system that places value on people’s bodies and minds, based on societally constructed ideas of normalcy, intelligence, excellence, and productivity…[and] leads to people and society determining who is valuable and worthy based on their appearance and/or their ability to satisfactorily [re]produce, excel, and ‘behave.’”8 Regarding CSC, concerns about systemic bias in guideline design were raised early by disability advocacy groups during comment periods.9,10 More broadly, concerns about ableism sit alongside many deeply rooted societal perspectives of disabled individuals as pitiable or, conversely, heroic for having “overcome” their disability in some way. As a physician who sits in a manual wheelchair with paraplegia and mobility impairment, I have equally been subject to inappropriate bias and inappropriate praise for living in a wheelchair. I have also wondered, alongside my patients living with different levels of mobility or ability, why others often view us as “worse off.” Addressing directly whether disabled individuals are “worse off,” disability rights attorney and advocate Harriet McBryde Johnson has articulated a predominant sentiment among persons living with unique or different abilities:
Are we “worse off”? I don’t think so. Not in any meaningful way. There are too many variables. For those of us with congenital conditions, disability shapes all we are. Those disabled later in life adapt. We take constraints that no one would choose and build rich and satisfying lives within them. We enjoy pleasures other people enjoy and pleasures peculiarly our own. We have something the world needs.11
Many physician colleagues have common, invisible diseases such as diabetes and heart disease; fewer colleagues share conditions that are as visible as my spinal cord injury, as readily apparent to patients upon my entry to their hospital rooms. This simultaneous and inescapable identity as both patient and provider has afforded me wonderful doctor-patient interactions, particularly with those patients who appreciate how my patient experience impacts my ability to partially understand theirs. However, this simultaneous identity as doctor and patient also informed my personal and professional concerns regarding structural ableism as I considered scoring my own acutely ill hospital medicine patients with CSC triage scores in April 2020.
As a practicing hospital medicine physician, I have been emboldened by the efforts of my fellow clinicians amid COVID-19; their efforts have reaffirmed all the reasons I pursued a career in medicine. However, when I heard my clinical colleagues’ first explanation of the Massachusetts CSC guidelines in April 2020, I raised my hand to ask whether the “life-years” to which the guidelines referred were quality-adjusted. My concern regarding the implicit use of quality-adjusted life years (QALY) or disability-adjusted life years in clinical decision-making and implementation of these guidelines was validated when no clinical leaders could address this question directly. Sitting on the CSC committee for my hospital during this time was an honor. However, it was disconcerting to hear many clinicians’ unease when estimating mean survival for common chronic diseases, ranging from end-stage renal disease to advanced heart failure. If my expert colleagues, clinical specialists in kidney and heart disease, could not confidently apply mean survival estimates to multimorbid hospital patients, then idiosyncratic clinical judgment was sure to have a heavy hand in any calculation of “life-years.” Thus, my primary concern was that clinicians using triage heuristics would be subject to bias, regardless of their intention, and negatively adjust for the quality of a disabled life in their CSC triage scoring. My secondary concern was that the CSC guidelines themselves included systemic bias against disabled individuals.
According to CSC schema, triage scores index heavily on Sequential Organ Failure Assessment (SOFA) scores to define short-term survival; SOFA scores are partially driven by the Glasgow Coma Scale (GCS). Following professional and public comment periods, CSC guidelines in Massachusetts were revised to, among other critical points of revision, change prognostic estimation via “life years” in favor of generic estimation of short-term survival (Table). I wondered, if I presented to an emergency department with severe COVID-19 and was scored with the GCS for the purpose of making a CSC ventilator triage decision, how would my complete paraplegia and lower-extremity motor impairment be accounted for by a clinician assessing “best motor response” in the GCS? The purpose of these scores is to act algorithmically, to guide clinicians whose cognitive load and time limitations may not allow for adjustment of these algorithms based on the individual patient in front of them. Individualization of clinical decisions is part of medicine’s art, but is difficult in the best of times and no easier during a crisis in care delivery. As CSC triage scores were amended and addended throughout 2020, I returned to the COVID wards, time and again wondering, “What have we learned about systemic bias and health inequity in the CSC process and the pandemic broadly, with specific regard to disability?”

Ability Awareness: Room for Our Improvement
Unfortunately, there is reason to believe that clinical judgment is impaired by structural ableism. In seminal work on this topic, Gerhart et al12 demonstrated that clinicians considered spinal cord injury (SCI) survivors to have low self-perceptions of worthiness, overall negative attitudes, and low self-esteem as compared to able-bodied individuals. However, surveyed SCI survivors generally had similar self-perceptions of worth and positivity as compared to ”able-bodied” clinicians.12 For providers who care for persons with disabilities, the majority (82.4%) have rated their disabled patients’ quality of life as worse.13 It is no wonder that patients with disabilities are more likely to feel that their doctor-patient relationship is impacted by lack of understanding, negative sentiment, or simple lack of listening.14 Generally, this poor doctor-patient relationship with disabled patients is exacerbated by poor exposure of medical trainees to disability education; only 34.2% of internal medicine residents recall any form of disability education in medical school, while only 52% of medical school deans report having disability educational content in their curricula.15,16 There is a similar lack of disability representation in the population of medical trainees themselves. While approximately 20% of the American population lives with a disability, less than 2% of American medical students have a disability.17-19
While representation of disabled populations in medical practice remains poor, disabled patients are generally less likely to receive age-appropriate prevention, appropriate access to care, and equal access to treatment.20-22 “Diagnostic overshadowing” refers to clinicians’ attribution of nonspecific signs or symptoms to a patient’s chronic disability as opposed to acute illness.23 This phenomenon has led to higher rates of preventable malignancy in disabled patients and misattribution of common somatic symptoms to intellectual disability.24,25 With this disparity in place as status quo for health care delivery to disabled populations, it is no surprise that certain portions of the disabled population have accounted for disproportionate mortality due to COVID-19.26,27Disability advocates have called for “nothing about us without us,” a phrase associated with the United Nations Convention on the Rights of Persons with Disabilities. Understanding the profound neurodiversity among several forms of sensory and cognitive disabilities, as well as the functional difference between cognitive disabilities, mobility impairment, and inability to meet one’s instrumental activities of daily living independently, others have proposed a unique approach to certain disabled populations in COVID care.28 My own perspective is that definite progress may require a more general understanding of the prevalence of disability by clinicians, both via medical training and by directly addressing health equity for disabled populations in such calculations as the CSC. Systemic ableism is apparent in our most common clinical scoring systems, ranging from the GCS and Functional Assessment Staging Table to the Eastern Cooperative Oncology Group and Karnofsky Performance Status scales. I have reexamined these scoring systems in my own understanding given their general equation of ambulation with ability or normalcy. As a doctor in a manual wheelchair who values greatly my personal quality of life and professional contribution to patient care, I worry that these scoring systems inherently discount my own equitable access to care. Individualization of patients’ particular abilities in the context of these scales must occur alongside evidence-based, guideline-directed management via these scoring systems.
Conclusion: Future Orientation
Updated CSC guidelines have accounted for the unique considerations of disabled patients by effectively caveating their scoring algorithms, directing clinicians via disclaimers to uniquely consider their disabled patients in clinical judgement. This is a first step, but it is also one that erodes the value of algorithms, which generally obviate more deliberative thinking and individualization. For our patients who lack certain abilities, as CSC continue to be activated in several states, we have an opportunity to pursue more inherently equitable solutions before further suffering accrues.29 By way of example, adaptations to scoring systems that leverage QALYs for value-based drug pricing indices have been proposed by organizations like the Institute for Clinical and Economic Review, which proposed the Equal-Value-of Life-Years-Gained framework to inform QALY-based arbitration of drug pricing.30 This is not a perfect rubric but instead represents an attempt to balance consideration of drugs, as has been done with ventilators during the pandemic, as a scare and expensive resource while addressing the just concerns of advocacy groups in structural ableism.
Resource stewardship during a crisis should not discount those states of human life that are perceived to be less desirable, particularly if they are not experienced as less desirable but are experienced uniquely. Instead, we should consider equitably measuring our intervention to match a patient’s needs, as we would dose-adjust a medication for renal function or consider minimally invasive procedures for multimorbid patients. COVID-19 has reflected our profession’s ethical adaptation during crisis as resources have become scarce; there is no better time to define solutions for health equity. We should now be concerned equally by the influence our personal biases have on our clinical practice and by the way in which these crisis standards will influence patients’ perception of and trust in their care providers during periods of perceived plentiful resources in the future. Health care resources are always limited, allocated according to societal values; if we value health equity for people of all abilities, then we will consider these abilities equitably as we pursue new standards for health care delivery.
Corresponding author: Gregory D. Snyder, MD, MBA, 2014 Washington Street, Newton, MA 02462; [email protected].
Disclosures: None.
1. Emanuel EJ, Persad G, Upshur R, et al. Fair Allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055. doi:10.1056/NEJMsb2005114
2. Savulescu J, Persson I, Wilkinson D. Utilitarianism and the pandemic. Bioethics. 2020;34(6):620-632. doi:10.1111/bioe.12771
3. Mello MM, Persad G, White DB. Respecting disability rights - toward improved crisis standards of care. N Engl J Med. 2020;383(5):e26. doi: 10.1056/NEJMp2011997
4. The Commonwealth of Massachusetts Executive Office of Health and Human Services Department of Public Health. Crisis Standards of Care Planning Guidance for the COVID-19 Pandemic. April 7, 2020. https://d279m997dpfwgl.cloudfront.net/wp/2020/04/CSC_April-7_2020.pdf
5. Knowles H. Hospitals overwhelmed by covid are turning to ‘crisis standards of care.’ What does that mean? The Washington Post. September 21, 2021. Accessed January 24, 2022. https://www.washingtonpost.com/health/2021/09/22/crisis-standards-of-care/
6. Hick JL, Hanfling D, Wynia MK, Toner E. Crisis standards of care and COVID-19: What did we learn? How do we ensure equity? What should we do? NAM Perspect. 2021;2021:10.31478/202108e. doi:10.31478/202108e
7. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
8. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
9. Kukla E. My life is more ‘disposable’ during this pandemic. The New York Times. March 19, 2020. Accessed January 24, 2022. https://www.nytimes.com/2020/03/19/opinion/coronavirus-disabled-health-care.html
10. CPR and Coalition Partners Secure Important Changes in Massachusetts’ Crisis Standards of Care. Center for Public Representation. December 1, 2020. Accessed January 24, 2022. https://www.centerforpublicrep.org/news/cpr-and-coalition-partners-secure-important-changes-in-massachusetts-crisis-standards-of-care/
11. Johnson HM. Unspeakable conversations. The New York Times. February 16, 2003. Accessed January 24, 2022. https://www.nytimes.com/2003/02/16/magazine/unspeakable-conversations.html
12. Gerhart KA, Koziol-McLain J, Lowenstein SR, Whiteneck GG. Quality of life following spinal cord injury: knowledge and attitudes of emergency care providers. Ann Emerg Med. 1994;23(4):807-812. doi:10.1016/s0196-0644(94)70318-3
13. Iezzoni LI, Rao SR, Ressalam J, et al. Physicians’ perceptions of people with disability and their health care. Health Aff (Millwood). 2021;40(2):297-306. doi:10.1377/hlthaff.2020.01452
14. Smith DL. Disparities in patient-physician communication for persons with a disability from the 2006 Medical Expenditure Panel Survey (MEPS). Disabil Health J. 2009;2(4):206-215. doi:10.1016/j.dhjo.2009.06.002
15. Stillman MD, Ankam N, Mallow M, Capron M, Williams S. A survey of internal and family medicine residents: Assessment of disability-specific education and knowledge. Disabil Health J. 2021;14(2):101011. doi:10.1016/j.dhjo.2020.101011
16. Seidel E, Crowe S. The state of disability awareness in American medical schools. Am J Phys Med Rehabil. 2017;96(9):673-676. doi:10.1097/PHM.0000000000000719
17. Okoro CA, Hollis ND, Cyrus AC, Griffin-Blake S. Prevalence of disabilities and health care access by disability status and type among adults - United States, 2016. MMWR Morb Mortal Wkly Rep. 2018;67(32):882-887. doi:10.15585/mmwr.mm6732a3
18. Peacock G, Iezzoni LI, Harkin TR. Health care for Americans with disabilities--25 years after the ADA. N Engl J Med. 2015;373(10):892-893. doi:10.1056/NEJMp1508854
19. DeLisa JA, Thomas P. Physicians with disabilities and the physician workforce: a need to reassess our policies. Am J Phys Med Rehabil. 2005;84(1):5-11. doi:10.1097/01.phm.0000153323.28396.de
20. Disability and Health. Healthy People 2020. Accessed January 24, 2022. https://www.healthypeople.gov/2020/topics-objectives/topic/disability-and-health
21. Lagu T, Hannon NS, Rothberg MB, et al. Access to subspecialty care for patients with mobility impairment: a survey. Ann Intern Med. 2013;158(6):441-446. doi: 10.7326/0003-4819-158-6-201303190-00003
22. McCarthy EP, Ngo LH, Roetzheim RG, et al. Disparities in breast cancer treatment and survival for women with disabilities. Ann Intern Med. 2006;145(9):637-645. doi: 10.7326/0003-4819-145-9-200611070-00005
23. Javaid A, Nakata V, Michael D. Diagnostic overshadowing in learning disability: think beyond the disability. Prog Neurol Psychiatry. 2019;23:8-10.
24. Iezzoni LI, Rao SR, Agaronnik ND, El-Jawahri A. Cross-sectional analysis of the associations between four common cancers and disability. J Natl Compr Canc Netw. 2020;18(8):1031-1044. doi:10.6004/jnccn.2020.7551
25. Sanders JS, Keller S, Aravamuthan BR. Caring for individuals with intellectual and developmental disabilities in the COVID-19 crisis. Neurol Clin Pract. 2021;11(2):e174-e178. doi:10.1212/CPJ.0000000000000886
26. Landes SD, Turk MA, Formica MK, McDonald KE, Stevens JD. COVID-19 outcomes among people with intellectual and developmental disability living in residential group homes in New York State. Disabil Health J. 2020;13(4):100969. doi:10.1016/j.dhjo.2020.100969
27. Gleason J, Ross W, Fossi A, Blonksy H, Tobias J, Stephens M. The devastating impact of Covid-19 on individuals with intellectual disabilities in the United States. NEJM Catalyst. 2021.doi.org/10.1056/CAT.21.0051
28. Nankervis K, Chan J. Applying the CRPD to people with intellectual and developmental disability with behaviors of concern during COVID-19. J Policy Pract Intellect Disabil. 2021:10.1111/jppi.12374. doi:10.1111/jppi.12374
29. Alaska Department of Health and Social Services, Division of Public Health, Rural and Community Health Systems. Patient care strategies for scarce resource situations. Version 1. August 2021. Accessed November 11, 2021, https://dhss.alaska.gov/dph/Epi/id/SiteAssets/Pages/HumanCoV/SOA_DHSS_CrisisStandardsOfCare.pdf
30. Cost-effectiveness, the QALY, and the evlyg. ICER. May 21, 2021. Accessed January 24, 2022. https://icer.org/our-approach/methods-process/cost-effectiveness-the-qaly-and-the-evlyg/
1. Emanuel EJ, Persad G, Upshur R, et al. Fair Allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055. doi:10.1056/NEJMsb2005114
2. Savulescu J, Persson I, Wilkinson D. Utilitarianism and the pandemic. Bioethics. 2020;34(6):620-632. doi:10.1111/bioe.12771
3. Mello MM, Persad G, White DB. Respecting disability rights - toward improved crisis standards of care. N Engl J Med. 2020;383(5):e26. doi: 10.1056/NEJMp2011997
4. The Commonwealth of Massachusetts Executive Office of Health and Human Services Department of Public Health. Crisis Standards of Care Planning Guidance for the COVID-19 Pandemic. April 7, 2020. https://d279m997dpfwgl.cloudfront.net/wp/2020/04/CSC_April-7_2020.pdf
5. Knowles H. Hospitals overwhelmed by covid are turning to ‘crisis standards of care.’ What does that mean? The Washington Post. September 21, 2021. Accessed January 24, 2022. https://www.washingtonpost.com/health/2021/09/22/crisis-standards-of-care/
6. Hick JL, Hanfling D, Wynia MK, Toner E. Crisis standards of care and COVID-19: What did we learn? How do we ensure equity? What should we do? NAM Perspect. 2021;2021:10.31478/202108e. doi:10.31478/202108e
7. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
8. Cleveland Manchanda EC, Sanky C, Appel JM. Crisis standards of care in the USA: a systematic review and implications for equity amidst COVID-19. J Racial Ethn Health Disparities. 2021;8(4):824-836. doi:10.1007/s40615-020-00840-5
9. Kukla E. My life is more ‘disposable’ during this pandemic. The New York Times. March 19, 2020. Accessed January 24, 2022. https://www.nytimes.com/2020/03/19/opinion/coronavirus-disabled-health-care.html
10. CPR and Coalition Partners Secure Important Changes in Massachusetts’ Crisis Standards of Care. Center for Public Representation. December 1, 2020. Accessed January 24, 2022. https://www.centerforpublicrep.org/news/cpr-and-coalition-partners-secure-important-changes-in-massachusetts-crisis-standards-of-care/
11. Johnson HM. Unspeakable conversations. The New York Times. February 16, 2003. Accessed January 24, 2022. https://www.nytimes.com/2003/02/16/magazine/unspeakable-conversations.html
12. Gerhart KA, Koziol-McLain J, Lowenstein SR, Whiteneck GG. Quality of life following spinal cord injury: knowledge and attitudes of emergency care providers. Ann Emerg Med. 1994;23(4):807-812. doi:10.1016/s0196-0644(94)70318-3
13. Iezzoni LI, Rao SR, Ressalam J, et al. Physicians’ perceptions of people with disability and their health care. Health Aff (Millwood). 2021;40(2):297-306. doi:10.1377/hlthaff.2020.01452
14. Smith DL. Disparities in patient-physician communication for persons with a disability from the 2006 Medical Expenditure Panel Survey (MEPS). Disabil Health J. 2009;2(4):206-215. doi:10.1016/j.dhjo.2009.06.002
15. Stillman MD, Ankam N, Mallow M, Capron M, Williams S. A survey of internal and family medicine residents: Assessment of disability-specific education and knowledge. Disabil Health J. 2021;14(2):101011. doi:10.1016/j.dhjo.2020.101011
16. Seidel E, Crowe S. The state of disability awareness in American medical schools. Am J Phys Med Rehabil. 2017;96(9):673-676. doi:10.1097/PHM.0000000000000719
17. Okoro CA, Hollis ND, Cyrus AC, Griffin-Blake S. Prevalence of disabilities and health care access by disability status and type among adults - United States, 2016. MMWR Morb Mortal Wkly Rep. 2018;67(32):882-887. doi:10.15585/mmwr.mm6732a3
18. Peacock G, Iezzoni LI, Harkin TR. Health care for Americans with disabilities--25 years after the ADA. N Engl J Med. 2015;373(10):892-893. doi:10.1056/NEJMp1508854
19. DeLisa JA, Thomas P. Physicians with disabilities and the physician workforce: a need to reassess our policies. Am J Phys Med Rehabil. 2005;84(1):5-11. doi:10.1097/01.phm.0000153323.28396.de
20. Disability and Health. Healthy People 2020. Accessed January 24, 2022. https://www.healthypeople.gov/2020/topics-objectives/topic/disability-and-health
21. Lagu T, Hannon NS, Rothberg MB, et al. Access to subspecialty care for patients with mobility impairment: a survey. Ann Intern Med. 2013;158(6):441-446. doi: 10.7326/0003-4819-158-6-201303190-00003
22. McCarthy EP, Ngo LH, Roetzheim RG, et al. Disparities in breast cancer treatment and survival for women with disabilities. Ann Intern Med. 2006;145(9):637-645. doi: 10.7326/0003-4819-145-9-200611070-00005
23. Javaid A, Nakata V, Michael D. Diagnostic overshadowing in learning disability: think beyond the disability. Prog Neurol Psychiatry. 2019;23:8-10.
24. Iezzoni LI, Rao SR, Agaronnik ND, El-Jawahri A. Cross-sectional analysis of the associations between four common cancers and disability. J Natl Compr Canc Netw. 2020;18(8):1031-1044. doi:10.6004/jnccn.2020.7551
25. Sanders JS, Keller S, Aravamuthan BR. Caring for individuals with intellectual and developmental disabilities in the COVID-19 crisis. Neurol Clin Pract. 2021;11(2):e174-e178. doi:10.1212/CPJ.0000000000000886
26. Landes SD, Turk MA, Formica MK, McDonald KE, Stevens JD. COVID-19 outcomes among people with intellectual and developmental disability living in residential group homes in New York State. Disabil Health J. 2020;13(4):100969. doi:10.1016/j.dhjo.2020.100969
27. Gleason J, Ross W, Fossi A, Blonksy H, Tobias J, Stephens M. The devastating impact of Covid-19 on individuals with intellectual disabilities in the United States. NEJM Catalyst. 2021.doi.org/10.1056/CAT.21.0051
28. Nankervis K, Chan J. Applying the CRPD to people with intellectual and developmental disability with behaviors of concern during COVID-19. J Policy Pract Intellect Disabil. 2021:10.1111/jppi.12374. doi:10.1111/jppi.12374
29. Alaska Department of Health and Social Services, Division of Public Health, Rural and Community Health Systems. Patient care strategies for scarce resource situations. Version 1. August 2021. Accessed November 11, 2021, https://dhss.alaska.gov/dph/Epi/id/SiteAssets/Pages/HumanCoV/SOA_DHSS_CrisisStandardsOfCare.pdf
30. Cost-effectiveness, the QALY, and the evlyg. ICER. May 21, 2021. Accessed January 24, 2022. https://icer.org/our-approach/methods-process/cost-effectiveness-the-qaly-and-the-evlyg/
Another winter for our discontent
Here we are. Again. It’s cold and it’s gray. The sun rises late and sets early, so that it feels like midnight by 8 p.m. Indoor venues are risky with the highly contagious Omicron variant, and I feel like we are all pushing the replay button on 2021’s miserable winter.
In some ways, it’s worse: In 2021 we had the hope that vaccines would pull us out of the pandemic and we had guidance on all that we should not be doing. In January, we were gaming the various Internet sites to get a coveted vaccine for ourselves or our family and friends, then lining up to get jabbed. We did not yet know that it wouldn’t be enough – that we’d need boosters, that Delta and Omicron would defy the vaccines. Yes, the vaccines work miracles to prevent severe disease and death, but the worry of passing the virus to someone who is vulnerable or unvaccinated(!), or both, remains – and now we can wonder how we’ll ever get out of this mess with hopeful talk of an endemic, while we wait on the next variant. I like certainty, and this pandemic is one big screaming reminder that certainty about anything is just a pleasant notion, death and taxes excluded, of course.
Kris Lukish, vice president of human resources at Johns Hopkins Hospital in Baltimore, started an update to the hospital employees with: “As we begin 2022, it feels like we are experiencing dejà vu, or ‘Groundhog Day,’ or ‘50 First Dates.’ In ‘50 First Dates,’ Drew Barrymore wakes up each day reliving one specific day. It never changes. I realize our world may seem a little like that right now. We thought we’d turned a corner with COVID, and instead we saw a rapid rise in cases and hospitalizations due to the Omicron variant, higher than in previous surges.”
In 2021, many of us skipped holiday travel and ate outdoors. My morning coffee group moved to Zoom and it wasn’t until late spring, when community rates of COVID nose-dived, that I began seeing patients in my office for the first time in over a year. Since many of my patients are over 60, I tested myself with a home antigen test before going into the office. I changed my schedule so sessions began on the half-hour to be sure the suite’s waiting room would be empty, and I purchased an air purifier, cracked the window open, and figured everyone was as safe as we could reasonably be.
By the first Monday in January 2022, the positivity rate in Maryland was just shy of 30%. Twitter circulated anecdotes about false negatives with the home antigen test kits, and I decided it was safest to return to all-virtual appointments.
Mona Masood, DO, is cofounder of the Physician Support Line, a call-in service for doctors that started in March 2020. She has noted a change in the problems physicians face.
“We’re seeing a lot of empathy fatigue,” Dr. Masood said. “It’s not unexpected with a prolonged situation like this – the trauma has doctors in survival mode and they need to be present for themselves, their families, and their patients. People are emotionally drained, and we’re stretching them to the limit. Now at the front lines, doctors are getting a lot of backlash. There are the conspiracy theories, and people who challenge their knowledge and training and it leads them to ask if they should be doing this work. and these are large decisions that are being made in a specific context.
“The other thing we’re hearing is from trainees – residents and fellows – who are expected to carry a lot of work on the COVID units. Some are being told that they can’t graduate because they haven’t finished their other training requirements. This type of systemic issue produces moral injury.”
Dr. Masood talked about what running the support line has been like for her. “I know people want to give more in a catastrophe, and I was realistic that the enthusiasm might die off. I would go as long as psychiatrists volunteer, and the most incredible thing is that it hasn’t stopped. Some of the original people are no longer with us, but others have come aboard, and it’s been incredible to be a part of this.”
In her Jan. 26, 2022, newsletter, epidemiologist Katelyn Jetelina, PhD, MPH, tried to be reassuring about the future. “In order to know how this will end, we need to look at how other pandemics ended,” Dr. Jetelina wrote. “First, recognize the last part of that sentence ... pandemics end. Every epi curve comes down. This pandemic will end, too. Hold that fact close to you.”
She wrote about the three ways that pandemics end. The SARS pandemic of 2002 lasted 1.5 years as public health measures were effective, in large part because the disease was spread only by symptomatic patients. Vaccines offer a second way to end pandemics, as they have for polio and smallpox. “If the globe works together, we could possibly eradicate SARS-CoV-2 with vaccines. [Now that we have numerous animal reservoirs, though, this is close to impossible.]”
Finally, Dr. Jetelina noted that the 1918 flu changed from a pandemic situation to being endemic. “Over time, the virus attenuated, it became less severe.” Society acclimates to a virus with a low mortality rate. “The vast majority of scientists think an endemic state is the future of SARS-CoV-2. I agree.” And she goes on to define endemic as a steady state, but not the absence of suffering. She likens it to malaria and tuberculosis, illnesses with high global mortality.
“An endemic will come without an announcement or headlines, we won’t know we’re there until well after we’ve arrived.” She wrote of the uncertainty that faces us moving forward: We don’t know how much, or how long, immunity from Omicron infections will last, or if future variants will cause more or less severe disease. She casted her vote for global vaccinations, boosters, masks, better ventilation, communication, empathy, and tolerance to end the pandemic.
In Maryland, hospitalizations and positivity are starting to decline from the postholiday surge. I have figured out that I am not good at predicting what will happen next, and the experts don’t seem to be much better. I’d like a headline ending, the kind we looked to be heading toward last June.
I’ve told my patients who want to come in person that I will reassess in March. We have written our own rules, and mine are somewhere in the middle – I don’t go to public indoor spaces unmasked, but I do see vaccinated family and friends in our homes without masks. I don’t want to be responsible for transmitting a potentially fatal illness to a vulnerable patient. Honestly, this makes no sense, but since there is a video option, I feel I should not risk passing a potentially lethal virus to my patients. I just hope I’m not writing this same article again in January 2023.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins. Dr. Miller has no conflicts of interest.
Here we are. Again. It’s cold and it’s gray. The sun rises late and sets early, so that it feels like midnight by 8 p.m. Indoor venues are risky with the highly contagious Omicron variant, and I feel like we are all pushing the replay button on 2021’s miserable winter.
In some ways, it’s worse: In 2021 we had the hope that vaccines would pull us out of the pandemic and we had guidance on all that we should not be doing. In January, we were gaming the various Internet sites to get a coveted vaccine for ourselves or our family and friends, then lining up to get jabbed. We did not yet know that it wouldn’t be enough – that we’d need boosters, that Delta and Omicron would defy the vaccines. Yes, the vaccines work miracles to prevent severe disease and death, but the worry of passing the virus to someone who is vulnerable or unvaccinated(!), or both, remains – and now we can wonder how we’ll ever get out of this mess with hopeful talk of an endemic, while we wait on the next variant. I like certainty, and this pandemic is one big screaming reminder that certainty about anything is just a pleasant notion, death and taxes excluded, of course.
Kris Lukish, vice president of human resources at Johns Hopkins Hospital in Baltimore, started an update to the hospital employees with: “As we begin 2022, it feels like we are experiencing dejà vu, or ‘Groundhog Day,’ or ‘50 First Dates.’ In ‘50 First Dates,’ Drew Barrymore wakes up each day reliving one specific day. It never changes. I realize our world may seem a little like that right now. We thought we’d turned a corner with COVID, and instead we saw a rapid rise in cases and hospitalizations due to the Omicron variant, higher than in previous surges.”
In 2021, many of us skipped holiday travel and ate outdoors. My morning coffee group moved to Zoom and it wasn’t until late spring, when community rates of COVID nose-dived, that I began seeing patients in my office for the first time in over a year. Since many of my patients are over 60, I tested myself with a home antigen test before going into the office. I changed my schedule so sessions began on the half-hour to be sure the suite’s waiting room would be empty, and I purchased an air purifier, cracked the window open, and figured everyone was as safe as we could reasonably be.
By the first Monday in January 2022, the positivity rate in Maryland was just shy of 30%. Twitter circulated anecdotes about false negatives with the home antigen test kits, and I decided it was safest to return to all-virtual appointments.
Mona Masood, DO, is cofounder of the Physician Support Line, a call-in service for doctors that started in March 2020. She has noted a change in the problems physicians face.
“We’re seeing a lot of empathy fatigue,” Dr. Masood said. “It’s not unexpected with a prolonged situation like this – the trauma has doctors in survival mode and they need to be present for themselves, their families, and their patients. People are emotionally drained, and we’re stretching them to the limit. Now at the front lines, doctors are getting a lot of backlash. There are the conspiracy theories, and people who challenge their knowledge and training and it leads them to ask if they should be doing this work. and these are large decisions that are being made in a specific context.
“The other thing we’re hearing is from trainees – residents and fellows – who are expected to carry a lot of work on the COVID units. Some are being told that they can’t graduate because they haven’t finished their other training requirements. This type of systemic issue produces moral injury.”
Dr. Masood talked about what running the support line has been like for her. “I know people want to give more in a catastrophe, and I was realistic that the enthusiasm might die off. I would go as long as psychiatrists volunteer, and the most incredible thing is that it hasn’t stopped. Some of the original people are no longer with us, but others have come aboard, and it’s been incredible to be a part of this.”
In her Jan. 26, 2022, newsletter, epidemiologist Katelyn Jetelina, PhD, MPH, tried to be reassuring about the future. “In order to know how this will end, we need to look at how other pandemics ended,” Dr. Jetelina wrote. “First, recognize the last part of that sentence ... pandemics end. Every epi curve comes down. This pandemic will end, too. Hold that fact close to you.”
She wrote about the three ways that pandemics end. The SARS pandemic of 2002 lasted 1.5 years as public health measures were effective, in large part because the disease was spread only by symptomatic patients. Vaccines offer a second way to end pandemics, as they have for polio and smallpox. “If the globe works together, we could possibly eradicate SARS-CoV-2 with vaccines. [Now that we have numerous animal reservoirs, though, this is close to impossible.]”
Finally, Dr. Jetelina noted that the 1918 flu changed from a pandemic situation to being endemic. “Over time, the virus attenuated, it became less severe.” Society acclimates to a virus with a low mortality rate. “The vast majority of scientists think an endemic state is the future of SARS-CoV-2. I agree.” And she goes on to define endemic as a steady state, but not the absence of suffering. She likens it to malaria and tuberculosis, illnesses with high global mortality.
“An endemic will come without an announcement or headlines, we won’t know we’re there until well after we’ve arrived.” She wrote of the uncertainty that faces us moving forward: We don’t know how much, or how long, immunity from Omicron infections will last, or if future variants will cause more or less severe disease. She casted her vote for global vaccinations, boosters, masks, better ventilation, communication, empathy, and tolerance to end the pandemic.
In Maryland, hospitalizations and positivity are starting to decline from the postholiday surge. I have figured out that I am not good at predicting what will happen next, and the experts don’t seem to be much better. I’d like a headline ending, the kind we looked to be heading toward last June.
I’ve told my patients who want to come in person that I will reassess in March. We have written our own rules, and mine are somewhere in the middle – I don’t go to public indoor spaces unmasked, but I do see vaccinated family and friends in our homes without masks. I don’t want to be responsible for transmitting a potentially fatal illness to a vulnerable patient. Honestly, this makes no sense, but since there is a video option, I feel I should not risk passing a potentially lethal virus to my patients. I just hope I’m not writing this same article again in January 2023.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins. Dr. Miller has no conflicts of interest.
Here we are. Again. It’s cold and it’s gray. The sun rises late and sets early, so that it feels like midnight by 8 p.m. Indoor venues are risky with the highly contagious Omicron variant, and I feel like we are all pushing the replay button on 2021’s miserable winter.
In some ways, it’s worse: In 2021 we had the hope that vaccines would pull us out of the pandemic and we had guidance on all that we should not be doing. In January, we were gaming the various Internet sites to get a coveted vaccine for ourselves or our family and friends, then lining up to get jabbed. We did not yet know that it wouldn’t be enough – that we’d need boosters, that Delta and Omicron would defy the vaccines. Yes, the vaccines work miracles to prevent severe disease and death, but the worry of passing the virus to someone who is vulnerable or unvaccinated(!), or both, remains – and now we can wonder how we’ll ever get out of this mess with hopeful talk of an endemic, while we wait on the next variant. I like certainty, and this pandemic is one big screaming reminder that certainty about anything is just a pleasant notion, death and taxes excluded, of course.
Kris Lukish, vice president of human resources at Johns Hopkins Hospital in Baltimore, started an update to the hospital employees with: “As we begin 2022, it feels like we are experiencing dejà vu, or ‘Groundhog Day,’ or ‘50 First Dates.’ In ‘50 First Dates,’ Drew Barrymore wakes up each day reliving one specific day. It never changes. I realize our world may seem a little like that right now. We thought we’d turned a corner with COVID, and instead we saw a rapid rise in cases and hospitalizations due to the Omicron variant, higher than in previous surges.”
In 2021, many of us skipped holiday travel and ate outdoors. My morning coffee group moved to Zoom and it wasn’t until late spring, when community rates of COVID nose-dived, that I began seeing patients in my office for the first time in over a year. Since many of my patients are over 60, I tested myself with a home antigen test before going into the office. I changed my schedule so sessions began on the half-hour to be sure the suite’s waiting room would be empty, and I purchased an air purifier, cracked the window open, and figured everyone was as safe as we could reasonably be.
By the first Monday in January 2022, the positivity rate in Maryland was just shy of 30%. Twitter circulated anecdotes about false negatives with the home antigen test kits, and I decided it was safest to return to all-virtual appointments.
Mona Masood, DO, is cofounder of the Physician Support Line, a call-in service for doctors that started in March 2020. She has noted a change in the problems physicians face.
“We’re seeing a lot of empathy fatigue,” Dr. Masood said. “It’s not unexpected with a prolonged situation like this – the trauma has doctors in survival mode and they need to be present for themselves, their families, and their patients. People are emotionally drained, and we’re stretching them to the limit. Now at the front lines, doctors are getting a lot of backlash. There are the conspiracy theories, and people who challenge their knowledge and training and it leads them to ask if they should be doing this work. and these are large decisions that are being made in a specific context.
“The other thing we’re hearing is from trainees – residents and fellows – who are expected to carry a lot of work on the COVID units. Some are being told that they can’t graduate because they haven’t finished their other training requirements. This type of systemic issue produces moral injury.”
Dr. Masood talked about what running the support line has been like for her. “I know people want to give more in a catastrophe, and I was realistic that the enthusiasm might die off. I would go as long as psychiatrists volunteer, and the most incredible thing is that it hasn’t stopped. Some of the original people are no longer with us, but others have come aboard, and it’s been incredible to be a part of this.”
In her Jan. 26, 2022, newsletter, epidemiologist Katelyn Jetelina, PhD, MPH, tried to be reassuring about the future. “In order to know how this will end, we need to look at how other pandemics ended,” Dr. Jetelina wrote. “First, recognize the last part of that sentence ... pandemics end. Every epi curve comes down. This pandemic will end, too. Hold that fact close to you.”
She wrote about the three ways that pandemics end. The SARS pandemic of 2002 lasted 1.5 years as public health measures were effective, in large part because the disease was spread only by symptomatic patients. Vaccines offer a second way to end pandemics, as they have for polio and smallpox. “If the globe works together, we could possibly eradicate SARS-CoV-2 with vaccines. [Now that we have numerous animal reservoirs, though, this is close to impossible.]”
Finally, Dr. Jetelina noted that the 1918 flu changed from a pandemic situation to being endemic. “Over time, the virus attenuated, it became less severe.” Society acclimates to a virus with a low mortality rate. “The vast majority of scientists think an endemic state is the future of SARS-CoV-2. I agree.” And she goes on to define endemic as a steady state, but not the absence of suffering. She likens it to malaria and tuberculosis, illnesses with high global mortality.
“An endemic will come without an announcement or headlines, we won’t know we’re there until well after we’ve arrived.” She wrote of the uncertainty that faces us moving forward: We don’t know how much, or how long, immunity from Omicron infections will last, or if future variants will cause more or less severe disease. She casted her vote for global vaccinations, boosters, masks, better ventilation, communication, empathy, and tolerance to end the pandemic.
In Maryland, hospitalizations and positivity are starting to decline from the postholiday surge. I have figured out that I am not good at predicting what will happen next, and the experts don’t seem to be much better. I’d like a headline ending, the kind we looked to be heading toward last June.
I’ve told my patients who want to come in person that I will reassess in March. We have written our own rules, and mine are somewhere in the middle – I don’t go to public indoor spaces unmasked, but I do see vaccinated family and friends in our homes without masks. I don’t want to be responsible for transmitting a potentially fatal illness to a vulnerable patient. Honestly, this makes no sense, but since there is a video option, I feel I should not risk passing a potentially lethal virus to my patients. I just hope I’m not writing this same article again in January 2023.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins. Dr. Miller has no conflicts of interest.
Primary care docs have role to play in hypertension prevention and treatment for women of reproductive age
The American Heart Association recently released a scientific statement concerning hypertension in pregnancy, which laid out the variety of disorders, the epidemiology, the future impact of pregnant persons, and the current debates regarding treatment and diagnosis.
This statement addresses all stages from preconception through post pregnancy and outlines the many prevention and treatment options available. Although family physicians were not specifically called out to be partners in the statement, we have a large role to play for both our pregnant patients and those of reproductive age who are not pregnant.
Preconception health
One of the first things pointed out was preconception health. Regardless of whether each individual family physician provides prenatal care, we can all focus on preconception health for those of reproductive age.
The statement from the AHA points out that “lifestyle changes before and during pregnancy may ameliorate both maternal and fetal risks.”
As many already do, family physicians should focus on encouraging their patients to practice healthy eating and exercise prior to pregnancy to help establish routines that will decrease the risk of hypertensive disorders in pregnancy.
Focusing on care prior to pregnancy also allows the primary care provider to be involved in quickly linking patients to prenatal care, as it is well established that early and complete prenatal care is important for improving outcomes.
Later-in-life pregnancy
The AHA also highlights that many are choosing to have pregnancies at older ages and with greater comorbidities than in past years. This is another area in which family physicians can provide important care.
We can help by first identifying the chronic conditions, such as hypertension and diabetes, that make the hypertensive disorders of pregnancy more likely. We should then focus on the treatment of these conditions during the preconception time so that they are well controlled prior to pregnancy.
We should also preferentially choose medications that our patients will be able to continue in pregnancy, so that control may be maintained throughout pregnancy.
The statement particularly highlights the avoidance of antihypertensives that are renin-angiotensin system blockers.
We can also help prepare our patients for the additional medications, testing, and precautions they will likely require during their pregnancy so that they know what to expect.
Family physicians are also already starting to utilize home blood pressure monitoring and can introduce this method so that patients may continue to monitor their blood pressures during pregnancy.
Throughout pregnancy, the new statement calls in the current debates of when prenatal care providers should be diagnosing hypertensive disorders and the goals of treatment.
Prenatal care providers can use shared decision-making for medication choices and blood pressure goals. They can also continue to encourage the healthy lifestyle choices such as diet and exercise to reduce the risk of poor outcomes.
This AHA also indicates that prenatal care providers can integrate the use of home blood pressure monitoring as they monitor the blood pressure for patients with hypertensive disorders of pregnancy.
Postpartum care
The postpartum period is another crucial time for family physicians and other primary care providers to greatly impact their patients with hypertensive diseases of pregnancy.
They can work to ensure that blood pressure is closely monitored and controlled, including by prescribing diuretics, which are typically not used during pregnancy.
If a patient’s blood pressure does not go down on its own, the primary care provider can begin treatment for hypertension outside of pregnancy. This can decrease their long-term cardiac risk factors and provide control prior to any future potential pregnancies.
Providing care during this postpartum time also offers a great opportunity to again encourage lifestyle options that may decrease risk.
Family physicians and other primary care providers can also encourage their patient to be involved in registries that gather data on hypertensive disorders in pregnancy.
In the new statement, the AHA acknowledges the great number of things that are not yet known or fully understood and the health inequities that many face.
Family physicians are positioned to help advocate for their patients and utilize a team-based approach to help provide resources to patients. We must continue to be there for our patients at every stage of their lives to help them live their healthiest lives possible.
The statement also indicates that there may be genetic factors at play more than social determinants of health. It is important to identify what those are for the best care of our patients while ensuring we are doing our best to provide our patients with the resources they need.
Dr. Wheat is a family physician at Erie Family Health Center and program director of Northwestern University’s McGaw Family Medicine residency program, both in Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
The American Heart Association recently released a scientific statement concerning hypertension in pregnancy, which laid out the variety of disorders, the epidemiology, the future impact of pregnant persons, and the current debates regarding treatment and diagnosis.
This statement addresses all stages from preconception through post pregnancy and outlines the many prevention and treatment options available. Although family physicians were not specifically called out to be partners in the statement, we have a large role to play for both our pregnant patients and those of reproductive age who are not pregnant.
Preconception health
One of the first things pointed out was preconception health. Regardless of whether each individual family physician provides prenatal care, we can all focus on preconception health for those of reproductive age.
The statement from the AHA points out that “lifestyle changes before and during pregnancy may ameliorate both maternal and fetal risks.”
As many already do, family physicians should focus on encouraging their patients to practice healthy eating and exercise prior to pregnancy to help establish routines that will decrease the risk of hypertensive disorders in pregnancy.
Focusing on care prior to pregnancy also allows the primary care provider to be involved in quickly linking patients to prenatal care, as it is well established that early and complete prenatal care is important for improving outcomes.
Later-in-life pregnancy
The AHA also highlights that many are choosing to have pregnancies at older ages and with greater comorbidities than in past years. This is another area in which family physicians can provide important care.
We can help by first identifying the chronic conditions, such as hypertension and diabetes, that make the hypertensive disorders of pregnancy more likely. We should then focus on the treatment of these conditions during the preconception time so that they are well controlled prior to pregnancy.
We should also preferentially choose medications that our patients will be able to continue in pregnancy, so that control may be maintained throughout pregnancy.
The statement particularly highlights the avoidance of antihypertensives that are renin-angiotensin system blockers.
We can also help prepare our patients for the additional medications, testing, and precautions they will likely require during their pregnancy so that they know what to expect.
Family physicians are also already starting to utilize home blood pressure monitoring and can introduce this method so that patients may continue to monitor their blood pressures during pregnancy.
Throughout pregnancy, the new statement calls in the current debates of when prenatal care providers should be diagnosing hypertensive disorders and the goals of treatment.
Prenatal care providers can use shared decision-making for medication choices and blood pressure goals. They can also continue to encourage the healthy lifestyle choices such as diet and exercise to reduce the risk of poor outcomes.
This AHA also indicates that prenatal care providers can integrate the use of home blood pressure monitoring as they monitor the blood pressure for patients with hypertensive disorders of pregnancy.
Postpartum care
The postpartum period is another crucial time for family physicians and other primary care providers to greatly impact their patients with hypertensive diseases of pregnancy.
They can work to ensure that blood pressure is closely monitored and controlled, including by prescribing diuretics, which are typically not used during pregnancy.
If a patient’s blood pressure does not go down on its own, the primary care provider can begin treatment for hypertension outside of pregnancy. This can decrease their long-term cardiac risk factors and provide control prior to any future potential pregnancies.
Providing care during this postpartum time also offers a great opportunity to again encourage lifestyle options that may decrease risk.
Family physicians and other primary care providers can also encourage their patient to be involved in registries that gather data on hypertensive disorders in pregnancy.
In the new statement, the AHA acknowledges the great number of things that are not yet known or fully understood and the health inequities that many face.
Family physicians are positioned to help advocate for their patients and utilize a team-based approach to help provide resources to patients. We must continue to be there for our patients at every stage of their lives to help them live their healthiest lives possible.
The statement also indicates that there may be genetic factors at play more than social determinants of health. It is important to identify what those are for the best care of our patients while ensuring we are doing our best to provide our patients with the resources they need.
Dr. Wheat is a family physician at Erie Family Health Center and program director of Northwestern University’s McGaw Family Medicine residency program, both in Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
The American Heart Association recently released a scientific statement concerning hypertension in pregnancy, which laid out the variety of disorders, the epidemiology, the future impact of pregnant persons, and the current debates regarding treatment and diagnosis.
This statement addresses all stages from preconception through post pregnancy and outlines the many prevention and treatment options available. Although family physicians were not specifically called out to be partners in the statement, we have a large role to play for both our pregnant patients and those of reproductive age who are not pregnant.
Preconception health
One of the first things pointed out was preconception health. Regardless of whether each individual family physician provides prenatal care, we can all focus on preconception health for those of reproductive age.
The statement from the AHA points out that “lifestyle changes before and during pregnancy may ameliorate both maternal and fetal risks.”
As many already do, family physicians should focus on encouraging their patients to practice healthy eating and exercise prior to pregnancy to help establish routines that will decrease the risk of hypertensive disorders in pregnancy.
Focusing on care prior to pregnancy also allows the primary care provider to be involved in quickly linking patients to prenatal care, as it is well established that early and complete prenatal care is important for improving outcomes.
Later-in-life pregnancy
The AHA also highlights that many are choosing to have pregnancies at older ages and with greater comorbidities than in past years. This is another area in which family physicians can provide important care.
We can help by first identifying the chronic conditions, such as hypertension and diabetes, that make the hypertensive disorders of pregnancy more likely. We should then focus on the treatment of these conditions during the preconception time so that they are well controlled prior to pregnancy.
We should also preferentially choose medications that our patients will be able to continue in pregnancy, so that control may be maintained throughout pregnancy.
The statement particularly highlights the avoidance of antihypertensives that are renin-angiotensin system blockers.
We can also help prepare our patients for the additional medications, testing, and precautions they will likely require during their pregnancy so that they know what to expect.
Family physicians are also already starting to utilize home blood pressure monitoring and can introduce this method so that patients may continue to monitor their blood pressures during pregnancy.
Throughout pregnancy, the new statement calls in the current debates of when prenatal care providers should be diagnosing hypertensive disorders and the goals of treatment.
Prenatal care providers can use shared decision-making for medication choices and blood pressure goals. They can also continue to encourage the healthy lifestyle choices such as diet and exercise to reduce the risk of poor outcomes.
This AHA also indicates that prenatal care providers can integrate the use of home blood pressure monitoring as they monitor the blood pressure for patients with hypertensive disorders of pregnancy.
Postpartum care
The postpartum period is another crucial time for family physicians and other primary care providers to greatly impact their patients with hypertensive diseases of pregnancy.
They can work to ensure that blood pressure is closely monitored and controlled, including by prescribing diuretics, which are typically not used during pregnancy.
If a patient’s blood pressure does not go down on its own, the primary care provider can begin treatment for hypertension outside of pregnancy. This can decrease their long-term cardiac risk factors and provide control prior to any future potential pregnancies.
Providing care during this postpartum time also offers a great opportunity to again encourage lifestyle options that may decrease risk.
Family physicians and other primary care providers can also encourage their patient to be involved in registries that gather data on hypertensive disorders in pregnancy.
In the new statement, the AHA acknowledges the great number of things that are not yet known or fully understood and the health inequities that many face.
Family physicians are positioned to help advocate for their patients and utilize a team-based approach to help provide resources to patients. We must continue to be there for our patients at every stage of their lives to help them live their healthiest lives possible.
The statement also indicates that there may be genetic factors at play more than social determinants of health. It is important to identify what those are for the best care of our patients while ensuring we are doing our best to provide our patients with the resources they need.
Dr. Wheat is a family physician at Erie Family Health Center and program director of Northwestern University’s McGaw Family Medicine residency program, both in Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
Should we always offer CPR?
Some details have been changed to protect the patient’s identity.
The first thing I noticed about Mr. Barry as I entered the intensive care unit was his left foot: Half of it was black, shriveled, and gangrenous, jutting out from under the white blanket. The soft rays of the morning sun illuminated his gaunt, unshaven, hollow cheeks. Sedated on propofol, with a green endotracheal tube sticking out of his chapped lips, he looked frail. His nurse, Becky, had just cleaned him after he passed tarry, maroon-colored stool. As she turned him over, I saw that the skin over his tailbone was broken. He had a large decubitus ulcer, the edges of which were now dried and black. The Foley bag, hanging next to his bed, was empty; there had been no urine for several hours now.
No one knew much about Mr. Barry. I don’t mean his current medical status – I mean what he did in life, who he loved, whether he had kids, what he valued. All we knew was that he was 83 years old and lived alone. No prior records in our system. No advanced directives. No information on any family. One of his neighbors called 911 after he was not seen for at least 10 days. Emergency medical services found Mr. Barry in bed, nearly lifeless. In the emergency room, he was noted to be in shock, with a dangerously low blood pressure. He was dry as a bone with markedly elevated sodium levels. His laboratory makers for kidney and liver function were deranged. He was admitted to the medical ICU with a diagnosis of hypovolemic shock and/or septic shock with multiorgan dysfunction. With 48 hours of supportive management with intravenous fluids and antibiotics, he did not improve. Blood cultures were positive for gram-positive cocci. The doses for medications used to maintain the blood pressure increased steadily. He also developed gastrointestinal bleeding.
Futile vs. potentially inappropriate
I was called for a cardiology consult because he had transient ST elevation in inferolateral leads on the monitor. Given his clinical scenario, the likelihood of type 1 myocardial infarction from plaque rupture was low; the ST elevations were probably related to vasospasm from increasing pressor requirement. Diagnostic cardiac catheterization showed clean coronary arteries. Continuous renal replacement therapy was soon started. Given Mr Barry’s multiorgan dysfunction and extremely poor prognosis, I recommended making all efforts to find his family or surrogate decision-maker to discuss goals of care or having a two-physician sign-off to place a DNR order.
Despite all efforts, we could not trace the family. We physicians vary individually on how we define value as related to life. We also vary on the degree of uncertainty about prognostication that we are comfortable with. This is one of the reasons the term “futility” is controversial and there is a push to use “potentially inappropriate” instead. The primary team had a different threshold for placing a DNR order and did not do it. That night, after I left the hospital, Mr Barry had a PEA (pulseless electrical activity) arrest and was resuscitated after 10 minutes of CPR. The next day, I noticed his bruised chest. He was on multiple medications to support his blood pressure.
My patient and a Hemingway protagonist
Whether by coincidence or irony, I started reading Ernest Hemingway’s short story “The Snows of Kilimanjaro” the same day that I met Mr. Barry. He reminded me of the story’s protagonist, Harry, lying on the cot with a gangrenous leg, waiting to die. Harry could sense death approaching. He reminisced about his past. All he wanted was to drink his “whiskey-soda.” “Darling, don’t drink that. We have to do everything we can,” his wife said. “You do it. I am tired,” Harry said, and continued to drink his whiskey-soda.
Mr. Barry looked tired. Tired of life? I can’t say with certainty. However, if I had to guess, the medical team’s heroics meant nothing to him. Unfortunately, he was not awake like Harry and could not do what he wished. I wondered what snippets of his life flashed before him as he lay on his bed at home for days. Did he want to have a whiskey-soda before dying? But we are not letting him die. Not easily anyway. We have to do everything we can: medications, coronary angiogram, dialysis, multiple rounds of CPR. Why?
In this country, we need permission to forgo CPR. If there are no advanced directives or next of kin available to discuss end-of-life care, performing CPR is the default status for all hospitalized patients, irrespective of the underlying severity of the illness. A unilateral DNR order written by a physician in good conscience (in a medically futile situation), but to which the patient has not consented, is generally invalid in most U.S. states. If health directives are not available, CPR will be administered on the presumption that the patient would want us to “do everything we can.” The medicolegal consequences and fear of not administering CPR is more profound than being found wrong and defying a patient’s wishes against CPR.
In patients with outside-hospital cardiac arrest, especially if related to ventricular fibrillation, early bystander CPR improves the survival rate. Hence, it makes sense for first responders and paramedics to administer CPR as the default option, focusing on the technique, rather than thinking about its utility based on the patient’s underlying comorbidities.
In the inpatient setting, however, physicians have enough information to comprehensively evaluate the patient. In a cohort of 5,690 critically ill ICU patients, obtained from a U.S. registry, the rate of survival to discharge after inpatient cardiac arrest is very low at 12.5%. Chronic health conditions, malignancy, end-stage renal disease, multiorgan dysfunction, need for vasopressor support, prior CPR, initial rhythm of asystole, or PEA advanced age were all associated with a less than 10% survival rate after CPR.
Dying is a process. Administering CPR to a dying patient is of little to no value. For Mr. Barry, it resulted in a bruised chest and broken ribs. James R. Jude, MD, one of the pioneers of closed chest compression, or modern-day CPR, wrote in 1965 that “resuscitation of the dying patient with irreparable damage to lungs, heart, kidneys, brain or any other vital system of the body has no medical, ethical, or moral justification. The techniques described in this monograph were designed to resuscitate the victim of acute insult, whether be it from drowning, electrical shock, untoward effect of drugs, anesthetic accident, heart block, acute myocardial infarction, or surgery.”
Yet, doctors continue to provide futile treatments at end of life for a variety of reasons: concerns about medico-legal risks, discomfort or inexperience with death and dying, uncertainty in prognostication, family requests, and organizational barriers such as lack of palliative services that can help lead end-of-life care discussions. Despite knowing that CPR has little benefit in critically ill patients with terminal illness and multiorgan dysfunction, we often ask the patient and their surrogate decision-makers: “If your heart stops, do you want us to restore your heart by pressing on the chest and giving electric shocks?” The very act of asking the question implies that CPR may be beneficial. We often do not go over the risks or offer an opinion on whether CPR should be performed. We take a neutral stance.
Anoxic brain injury, pain from broken ribs, and low likelihood of survival to discharge with acceptable neurologic recovery are rarely discussed in detail. Laypeople may overestimate the chances of survival after CPR and they may not comprehend that it does not reverse the dying process in patients with a terminal illness. When you ask about CPR, most families hear: “Do you want your loved one to live?” and the answer is nearly always “Yes.” We then administer CPR, thinking that we are respecting the patient’s autonomy in the medical decision-making process. However, in end-of-life care, elderly patients or surrogates may not fully understand the complexities involved or the outcomes of CPR. So, are we truly respecting their autonomy?
When to offer CPR?
In 2011, Billings and Krakauer, palliative care specialists from Massachusetts General Hospital, Boston, suggested that we focus on understanding our patient’s values and goals of care, and then decide whether to offer CPR, rather than taking a neutral stance. With this approach, we continue to respect the patient’s autonomy and also affirm our responsibility in providing care consistent with medical reality. We need to have the humility to accept that death is inevitable.
It has been 10 years since a group of physicians from Columbia University Medical Center, Harvard Medical School, MGH, and Boston Children’s Hospital proposed changes to how we determine resuscitation status. Instead of assuming that CPR is always wanted, they suggested three distinct approaches: consider CPR when the benefits versus risks are uncertain, and the patient is not end stage; recommend against CPR when there is a low likelihood of benefit and high likelihood of harm (e.g., patients with anoxic brain injury, advanced incurable cancer, or end-stage multiorgan dysfunction); and do not offer CPR to patients who will die imminently and have no chance of surviving CPR (e.g., patients with multiorgan dysfunction, increasing pressor requirements, and those who are actively dying without a single immediately reversible cause). I agree with their proposal.
Mr. Barry was actively dying. Unfortunately, we had neither his advanced directives nor access to family members or surrogates to discuss values and goals of care. Given the futility of administering CPR again, and based on our humanitarian principles, a moral and ethical responsibility to ensure a peaceful dying process, I and another ICU attending placed the DNR order. He passed away, peacefully, within a few hours.
That evening, as I was sitting on my porch reading the last page of “The Snows of Kilimanjaro,” my phone pinged. It was an email asking me to complete the final attestation for the death certificate. I imagined that Mr. Barry knew where he was going. He probably had his own special place – something beautiful and majestic, great and tall, dazzlingly white in the hot sun, like the snow-capped mountain of Kilimanjaro that Harry saw at the time of his death.
Dr. Mallidi is a general cardiologist at Zuckerberg San Francisco General Hospital, UCSF. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Some details have been changed to protect the patient’s identity.
The first thing I noticed about Mr. Barry as I entered the intensive care unit was his left foot: Half of it was black, shriveled, and gangrenous, jutting out from under the white blanket. The soft rays of the morning sun illuminated his gaunt, unshaven, hollow cheeks. Sedated on propofol, with a green endotracheal tube sticking out of his chapped lips, he looked frail. His nurse, Becky, had just cleaned him after he passed tarry, maroon-colored stool. As she turned him over, I saw that the skin over his tailbone was broken. He had a large decubitus ulcer, the edges of which were now dried and black. The Foley bag, hanging next to his bed, was empty; there had been no urine for several hours now.
No one knew much about Mr. Barry. I don’t mean his current medical status – I mean what he did in life, who he loved, whether he had kids, what he valued. All we knew was that he was 83 years old and lived alone. No prior records in our system. No advanced directives. No information on any family. One of his neighbors called 911 after he was not seen for at least 10 days. Emergency medical services found Mr. Barry in bed, nearly lifeless. In the emergency room, he was noted to be in shock, with a dangerously low blood pressure. He was dry as a bone with markedly elevated sodium levels. His laboratory makers for kidney and liver function were deranged. He was admitted to the medical ICU with a diagnosis of hypovolemic shock and/or septic shock with multiorgan dysfunction. With 48 hours of supportive management with intravenous fluids and antibiotics, he did not improve. Blood cultures were positive for gram-positive cocci. The doses for medications used to maintain the blood pressure increased steadily. He also developed gastrointestinal bleeding.
Futile vs. potentially inappropriate
I was called for a cardiology consult because he had transient ST elevation in inferolateral leads on the monitor. Given his clinical scenario, the likelihood of type 1 myocardial infarction from plaque rupture was low; the ST elevations were probably related to vasospasm from increasing pressor requirement. Diagnostic cardiac catheterization showed clean coronary arteries. Continuous renal replacement therapy was soon started. Given Mr Barry’s multiorgan dysfunction and extremely poor prognosis, I recommended making all efforts to find his family or surrogate decision-maker to discuss goals of care or having a two-physician sign-off to place a DNR order.
Despite all efforts, we could not trace the family. We physicians vary individually on how we define value as related to life. We also vary on the degree of uncertainty about prognostication that we are comfortable with. This is one of the reasons the term “futility” is controversial and there is a push to use “potentially inappropriate” instead. The primary team had a different threshold for placing a DNR order and did not do it. That night, after I left the hospital, Mr Barry had a PEA (pulseless electrical activity) arrest and was resuscitated after 10 minutes of CPR. The next day, I noticed his bruised chest. He was on multiple medications to support his blood pressure.
My patient and a Hemingway protagonist
Whether by coincidence or irony, I started reading Ernest Hemingway’s short story “The Snows of Kilimanjaro” the same day that I met Mr. Barry. He reminded me of the story’s protagonist, Harry, lying on the cot with a gangrenous leg, waiting to die. Harry could sense death approaching. He reminisced about his past. All he wanted was to drink his “whiskey-soda.” “Darling, don’t drink that. We have to do everything we can,” his wife said. “You do it. I am tired,” Harry said, and continued to drink his whiskey-soda.
Mr. Barry looked tired. Tired of life? I can’t say with certainty. However, if I had to guess, the medical team’s heroics meant nothing to him. Unfortunately, he was not awake like Harry and could not do what he wished. I wondered what snippets of his life flashed before him as he lay on his bed at home for days. Did he want to have a whiskey-soda before dying? But we are not letting him die. Not easily anyway. We have to do everything we can: medications, coronary angiogram, dialysis, multiple rounds of CPR. Why?
In this country, we need permission to forgo CPR. If there are no advanced directives or next of kin available to discuss end-of-life care, performing CPR is the default status for all hospitalized patients, irrespective of the underlying severity of the illness. A unilateral DNR order written by a physician in good conscience (in a medically futile situation), but to which the patient has not consented, is generally invalid in most U.S. states. If health directives are not available, CPR will be administered on the presumption that the patient would want us to “do everything we can.” The medicolegal consequences and fear of not administering CPR is more profound than being found wrong and defying a patient’s wishes against CPR.
In patients with outside-hospital cardiac arrest, especially if related to ventricular fibrillation, early bystander CPR improves the survival rate. Hence, it makes sense for first responders and paramedics to administer CPR as the default option, focusing on the technique, rather than thinking about its utility based on the patient’s underlying comorbidities.
In the inpatient setting, however, physicians have enough information to comprehensively evaluate the patient. In a cohort of 5,690 critically ill ICU patients, obtained from a U.S. registry, the rate of survival to discharge after inpatient cardiac arrest is very low at 12.5%. Chronic health conditions, malignancy, end-stage renal disease, multiorgan dysfunction, need for vasopressor support, prior CPR, initial rhythm of asystole, or PEA advanced age were all associated with a less than 10% survival rate after CPR.
Dying is a process. Administering CPR to a dying patient is of little to no value. For Mr. Barry, it resulted in a bruised chest and broken ribs. James R. Jude, MD, one of the pioneers of closed chest compression, or modern-day CPR, wrote in 1965 that “resuscitation of the dying patient with irreparable damage to lungs, heart, kidneys, brain or any other vital system of the body has no medical, ethical, or moral justification. The techniques described in this monograph were designed to resuscitate the victim of acute insult, whether be it from drowning, electrical shock, untoward effect of drugs, anesthetic accident, heart block, acute myocardial infarction, or surgery.”
Yet, doctors continue to provide futile treatments at end of life for a variety of reasons: concerns about medico-legal risks, discomfort or inexperience with death and dying, uncertainty in prognostication, family requests, and organizational barriers such as lack of palliative services that can help lead end-of-life care discussions. Despite knowing that CPR has little benefit in critically ill patients with terminal illness and multiorgan dysfunction, we often ask the patient and their surrogate decision-makers: “If your heart stops, do you want us to restore your heart by pressing on the chest and giving electric shocks?” The very act of asking the question implies that CPR may be beneficial. We often do not go over the risks or offer an opinion on whether CPR should be performed. We take a neutral stance.
Anoxic brain injury, pain from broken ribs, and low likelihood of survival to discharge with acceptable neurologic recovery are rarely discussed in detail. Laypeople may overestimate the chances of survival after CPR and they may not comprehend that it does not reverse the dying process in patients with a terminal illness. When you ask about CPR, most families hear: “Do you want your loved one to live?” and the answer is nearly always “Yes.” We then administer CPR, thinking that we are respecting the patient’s autonomy in the medical decision-making process. However, in end-of-life care, elderly patients or surrogates may not fully understand the complexities involved or the outcomes of CPR. So, are we truly respecting their autonomy?
When to offer CPR?
In 2011, Billings and Krakauer, palliative care specialists from Massachusetts General Hospital, Boston, suggested that we focus on understanding our patient’s values and goals of care, and then decide whether to offer CPR, rather than taking a neutral stance. With this approach, we continue to respect the patient’s autonomy and also affirm our responsibility in providing care consistent with medical reality. We need to have the humility to accept that death is inevitable.
It has been 10 years since a group of physicians from Columbia University Medical Center, Harvard Medical School, MGH, and Boston Children’s Hospital proposed changes to how we determine resuscitation status. Instead of assuming that CPR is always wanted, they suggested three distinct approaches: consider CPR when the benefits versus risks are uncertain, and the patient is not end stage; recommend against CPR when there is a low likelihood of benefit and high likelihood of harm (e.g., patients with anoxic brain injury, advanced incurable cancer, or end-stage multiorgan dysfunction); and do not offer CPR to patients who will die imminently and have no chance of surviving CPR (e.g., patients with multiorgan dysfunction, increasing pressor requirements, and those who are actively dying without a single immediately reversible cause). I agree with their proposal.
Mr. Barry was actively dying. Unfortunately, we had neither his advanced directives nor access to family members or surrogates to discuss values and goals of care. Given the futility of administering CPR again, and based on our humanitarian principles, a moral and ethical responsibility to ensure a peaceful dying process, I and another ICU attending placed the DNR order. He passed away, peacefully, within a few hours.
That evening, as I was sitting on my porch reading the last page of “The Snows of Kilimanjaro,” my phone pinged. It was an email asking me to complete the final attestation for the death certificate. I imagined that Mr. Barry knew where he was going. He probably had his own special place – something beautiful and majestic, great and tall, dazzlingly white in the hot sun, like the snow-capped mountain of Kilimanjaro that Harry saw at the time of his death.
Dr. Mallidi is a general cardiologist at Zuckerberg San Francisco General Hospital, UCSF. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Some details have been changed to protect the patient’s identity.
The first thing I noticed about Mr. Barry as I entered the intensive care unit was his left foot: Half of it was black, shriveled, and gangrenous, jutting out from under the white blanket. The soft rays of the morning sun illuminated his gaunt, unshaven, hollow cheeks. Sedated on propofol, with a green endotracheal tube sticking out of his chapped lips, he looked frail. His nurse, Becky, had just cleaned him after he passed tarry, maroon-colored stool. As she turned him over, I saw that the skin over his tailbone was broken. He had a large decubitus ulcer, the edges of which were now dried and black. The Foley bag, hanging next to his bed, was empty; there had been no urine for several hours now.
No one knew much about Mr. Barry. I don’t mean his current medical status – I mean what he did in life, who he loved, whether he had kids, what he valued. All we knew was that he was 83 years old and lived alone. No prior records in our system. No advanced directives. No information on any family. One of his neighbors called 911 after he was not seen for at least 10 days. Emergency medical services found Mr. Barry in bed, nearly lifeless. In the emergency room, he was noted to be in shock, with a dangerously low blood pressure. He was dry as a bone with markedly elevated sodium levels. His laboratory makers for kidney and liver function were deranged. He was admitted to the medical ICU with a diagnosis of hypovolemic shock and/or septic shock with multiorgan dysfunction. With 48 hours of supportive management with intravenous fluids and antibiotics, he did not improve. Blood cultures were positive for gram-positive cocci. The doses for medications used to maintain the blood pressure increased steadily. He also developed gastrointestinal bleeding.
Futile vs. potentially inappropriate
I was called for a cardiology consult because he had transient ST elevation in inferolateral leads on the monitor. Given his clinical scenario, the likelihood of type 1 myocardial infarction from plaque rupture was low; the ST elevations were probably related to vasospasm from increasing pressor requirement. Diagnostic cardiac catheterization showed clean coronary arteries. Continuous renal replacement therapy was soon started. Given Mr Barry’s multiorgan dysfunction and extremely poor prognosis, I recommended making all efforts to find his family or surrogate decision-maker to discuss goals of care or having a two-physician sign-off to place a DNR order.
Despite all efforts, we could not trace the family. We physicians vary individually on how we define value as related to life. We also vary on the degree of uncertainty about prognostication that we are comfortable with. This is one of the reasons the term “futility” is controversial and there is a push to use “potentially inappropriate” instead. The primary team had a different threshold for placing a DNR order and did not do it. That night, after I left the hospital, Mr Barry had a PEA (pulseless electrical activity) arrest and was resuscitated after 10 minutes of CPR. The next day, I noticed his bruised chest. He was on multiple medications to support his blood pressure.
My patient and a Hemingway protagonist
Whether by coincidence or irony, I started reading Ernest Hemingway’s short story “The Snows of Kilimanjaro” the same day that I met Mr. Barry. He reminded me of the story’s protagonist, Harry, lying on the cot with a gangrenous leg, waiting to die. Harry could sense death approaching. He reminisced about his past. All he wanted was to drink his “whiskey-soda.” “Darling, don’t drink that. We have to do everything we can,” his wife said. “You do it. I am tired,” Harry said, and continued to drink his whiskey-soda.
Mr. Barry looked tired. Tired of life? I can’t say with certainty. However, if I had to guess, the medical team’s heroics meant nothing to him. Unfortunately, he was not awake like Harry and could not do what he wished. I wondered what snippets of his life flashed before him as he lay on his bed at home for days. Did he want to have a whiskey-soda before dying? But we are not letting him die. Not easily anyway. We have to do everything we can: medications, coronary angiogram, dialysis, multiple rounds of CPR. Why?
In this country, we need permission to forgo CPR. If there are no advanced directives or next of kin available to discuss end-of-life care, performing CPR is the default status for all hospitalized patients, irrespective of the underlying severity of the illness. A unilateral DNR order written by a physician in good conscience (in a medically futile situation), but to which the patient has not consented, is generally invalid in most U.S. states. If health directives are not available, CPR will be administered on the presumption that the patient would want us to “do everything we can.” The medicolegal consequences and fear of not administering CPR is more profound than being found wrong and defying a patient’s wishes against CPR.
In patients with outside-hospital cardiac arrest, especially if related to ventricular fibrillation, early bystander CPR improves the survival rate. Hence, it makes sense for first responders and paramedics to administer CPR as the default option, focusing on the technique, rather than thinking about its utility based on the patient’s underlying comorbidities.
In the inpatient setting, however, physicians have enough information to comprehensively evaluate the patient. In a cohort of 5,690 critically ill ICU patients, obtained from a U.S. registry, the rate of survival to discharge after inpatient cardiac arrest is very low at 12.5%. Chronic health conditions, malignancy, end-stage renal disease, multiorgan dysfunction, need for vasopressor support, prior CPR, initial rhythm of asystole, or PEA advanced age were all associated with a less than 10% survival rate after CPR.
Dying is a process. Administering CPR to a dying patient is of little to no value. For Mr. Barry, it resulted in a bruised chest and broken ribs. James R. Jude, MD, one of the pioneers of closed chest compression, or modern-day CPR, wrote in 1965 that “resuscitation of the dying patient with irreparable damage to lungs, heart, kidneys, brain or any other vital system of the body has no medical, ethical, or moral justification. The techniques described in this monograph were designed to resuscitate the victim of acute insult, whether be it from drowning, electrical shock, untoward effect of drugs, anesthetic accident, heart block, acute myocardial infarction, or surgery.”
Yet, doctors continue to provide futile treatments at end of life for a variety of reasons: concerns about medico-legal risks, discomfort or inexperience with death and dying, uncertainty in prognostication, family requests, and organizational barriers such as lack of palliative services that can help lead end-of-life care discussions. Despite knowing that CPR has little benefit in critically ill patients with terminal illness and multiorgan dysfunction, we often ask the patient and their surrogate decision-makers: “If your heart stops, do you want us to restore your heart by pressing on the chest and giving electric shocks?” The very act of asking the question implies that CPR may be beneficial. We often do not go over the risks or offer an opinion on whether CPR should be performed. We take a neutral stance.
Anoxic brain injury, pain from broken ribs, and low likelihood of survival to discharge with acceptable neurologic recovery are rarely discussed in detail. Laypeople may overestimate the chances of survival after CPR and they may not comprehend that it does not reverse the dying process in patients with a terminal illness. When you ask about CPR, most families hear: “Do you want your loved one to live?” and the answer is nearly always “Yes.” We then administer CPR, thinking that we are respecting the patient’s autonomy in the medical decision-making process. However, in end-of-life care, elderly patients or surrogates may not fully understand the complexities involved or the outcomes of CPR. So, are we truly respecting their autonomy?
When to offer CPR?
In 2011, Billings and Krakauer, palliative care specialists from Massachusetts General Hospital, Boston, suggested that we focus on understanding our patient’s values and goals of care, and then decide whether to offer CPR, rather than taking a neutral stance. With this approach, we continue to respect the patient’s autonomy and also affirm our responsibility in providing care consistent with medical reality. We need to have the humility to accept that death is inevitable.
It has been 10 years since a group of physicians from Columbia University Medical Center, Harvard Medical School, MGH, and Boston Children’s Hospital proposed changes to how we determine resuscitation status. Instead of assuming that CPR is always wanted, they suggested three distinct approaches: consider CPR when the benefits versus risks are uncertain, and the patient is not end stage; recommend against CPR when there is a low likelihood of benefit and high likelihood of harm (e.g., patients with anoxic brain injury, advanced incurable cancer, or end-stage multiorgan dysfunction); and do not offer CPR to patients who will die imminently and have no chance of surviving CPR (e.g., patients with multiorgan dysfunction, increasing pressor requirements, and those who are actively dying without a single immediately reversible cause). I agree with their proposal.
Mr. Barry was actively dying. Unfortunately, we had neither his advanced directives nor access to family members or surrogates to discuss values and goals of care. Given the futility of administering CPR again, and based on our humanitarian principles, a moral and ethical responsibility to ensure a peaceful dying process, I and another ICU attending placed the DNR order. He passed away, peacefully, within a few hours.
That evening, as I was sitting on my porch reading the last page of “The Snows of Kilimanjaro,” my phone pinged. It was an email asking me to complete the final attestation for the death certificate. I imagined that Mr. Barry knew where he was going. He probably had his own special place – something beautiful and majestic, great and tall, dazzlingly white in the hot sun, like the snow-capped mountain of Kilimanjaro that Harry saw at the time of his death.
Dr. Mallidi is a general cardiologist at Zuckerberg San Francisco General Hospital, UCSF. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Optimizing ‘optimal’ in ovarian cancer cytoreduction
The goal of advanced ovarian cancer surgery is to remove all gross disease, or all visible and palpable disease implants. This became the established standard when improved survival was consistently observed among patients who had undergone complete surgical resection. Traditionally, definitions of no gross residual disease have been left in the hands, and eyes, of the surgeon. However, new technology has emerged which affords surgeons the ability to visualize ovarian cancer deposits that are imperceptible to the naked eye. But will this improve upon the poor cure rates for advanced ovarian cancer?
Many are familiar with the traditional definitions of “optimal” (less than 1 cm–sized deposits at any one location) and “suboptimal” (greater than 1 cm–sized deposits remaining) when referring to surgical cytoreduction of ovarian cancer. This nomenclature was introduced to define, categorize, and prognosticate patient groups after surgery. In recent years we have moved away from these descriptive definitions of ovarian cancer resection, borrowing from surgical oncology measures of surgical outcomes where “R0” defines surgical resection with negative margins, “R1” includes resection with positive microscopic margins (negative for tumor intraoperatively, but positive on microscopic pathology), and “R2” refers to macroscopic residual disease remaining.1
In ovarian cancer, surgeons have adopted the expression R0 to include patients in whom there is no gross visible or palpable residual disease, a special, favorable subgrouping of the previous “optimal” group. R1 is applied to patients with macroscopic, residual disease that fits within the traditional “optimal” cytoreduction classification (<1 cm in any one location). Obviously, these are significant variations to the traditional surgical oncology definitions, but not without supporting data. For example, patients with no gross residual disease (now defined as “R0”) have been observed to have improved survival, compared with patients who are “optimally” debulked but with R1 (<1 cm) residual disease.2 Therefore, this new goal of complete surgical resection has replaced the previous standard of “optimal” cytoreduction in which small macroscopic residual disease was acceptable.
Whether or not a surgery is completed with no gross residual disease is a subjective assessment made by the surgeon, and in practice, highly inaccurate. When a posttrial ad hoc analysis of 1,873 patients with advanced ovarian cancer who had been enrolled in a Gynecologic Oncology Group cooperative trial correlated surgeons’ assessments of “optimal” cytoreduction with objective postoperative radiographic findings (performed, on average, less than 1 month postoperatively) they found that postoperative CT scans identified lesions >1 cm in 40% of cases that had been characterized by surgeons as an “optimal” cytoreduction.3 Most commonly, discrepant lesions were identified in the upper abdominal quadrants and retroperitoneal aortic nodal regions. Therefore, surgeons’ subjective assessment of cytoreduction is prone to error, and given how important the completeness of cytoreduction is for clinical outcomes, there is interest in discovering methods to improve upon surgeons’ ability to discriminate volume of disease.
Pafolacianine (Cytalux, On Target Laboratories) is a novel drug that binds a fluorescent molecule to folic acid targeting the folate alpha receptors which are overexpressed on nonmucinous epithelial ovarian cancer cells compared with adjacent nonmalignant tissues.4 The drug is intravenously infused preoperatively and then visualized with companion near-infrared imaging devices during surgery to visualize its fluorescent signal where it is bound to ovarian cancer implants. In a phase 2 study of 178 patients with confirmed or suspected ovarian cancer, pafolacianine was able to detect implants of ovarian cancer in 26.9% of cases where the surgeon’s visual inspection was negative.5 Of note, the false-positive rate of this drug was not trivial, at 20%. Based on this efficacy data, the drug has been granted FDA approved for use in ovarian cancer surgery to augment the surgeon’s visualization of cancer. However, important questions remain unanswered by these preliminary data.
Will removal of additional microscopic ovarian cancer implants, only seen by pafolacianine, improve the survival of patients with ovarian cancer, and what effect will the addition of this extra surgery have on their surgical morbidity and risk? The use of pafolacianine to augment ovarian cancer debulking surgeries pivots on the premise that ovarian cancer outcomes are determined by surgical “effort” more than the biology of the disease. Otherwise said: The more we surgically remove, the more we cure. But this seems an old-fashioned notion, increasingly challenged by data. It has been shown that, when ovarian cancer debulking surgeries are necessarily more radical because of extensive disease distribution, prognosis is worse, compared with those patients with less extensive disease distribution.6 The effect of surgical effort contributes less than that of predetermined patterns of disease presentation. Additionally, genomic traits are different in tumors that are objectively determined to be not amenable to optimal cytoreduction, compared with resectable tumors.7 These data suggest that it is the disease, more than the surgeon, that most influences outcomes.
Additionally, the question of whether surgical removal of microscopic disease improves ovarian cancer survival has already been addressed with negative findings. The LION trial randomized 647 women with advanced ovarian cancer to primary cytoreductive surgery either with or without routine lymphadenectomy of clinically negative nodes.8 This study found no survival benefit to resecting clinically negative, microscopically positive nodes. In light of these data, it is difficult to imagine that there would be different results with the resection of microscopic peritoneal disease implants identified by pafolacianine.
While pafolacianine promises to move us closer to a true “R0” (negative margins) resection of ovarian cancer, is this even a feasible goal in a disease that is widely metastatic, particularly in the peritoneal cavity? What do “negative margins” mean in the peritoneal cavity? The sensitivity of pafolacianine in detecting microscopic disease is obviously not so high that it can guarantee patients a complete resection of a disseminated disease, and we still do not know what absolute benefit is derived from moving a little bit further on the continuum of surgical resection.
Perhaps augmentation of debulking is not the only, or best, use of pafolacianine for ovarian cancer surgery. Perhaps it might serve a role in diagnostics or staging of the disease rather than for a therapeutic purpose. In the meantime, we await ongoing clinical trials in this space to better inform clinicians what benefits, or harms, they might expect from the addition of this new drug as we continue to define the “optimal” surgical procedure for advanced ovarian cancer.
Dr. Emma Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no conflicts of interest.
References
1. Hermanek P, Wittekind C. Semin Surg Oncol 1994;10:12-20.
2. Elattar A et al. Cochrane Database Syst Rev 2011 Aug 10;2011(8):CD007565.
3. Eskander RN et al. Gynecol Oncol 2018;149:525-30.
4. Randall LM et al. Gynecol Oncol 2019;155:63-8.
5. Food and Drug Administration. FDA approves pafolacianine for identifying malignant ovarian cancer lesions. 2021 Dec 1.
6. Horowitz NS et al. J Clin Oncol 2015;33:937-43.
7. Lee S et al. Cell Rep. 2020;31:107502.
8. Harter P et al. N Engl J Med 2019;380:822-32.
The goal of advanced ovarian cancer surgery is to remove all gross disease, or all visible and palpable disease implants. This became the established standard when improved survival was consistently observed among patients who had undergone complete surgical resection. Traditionally, definitions of no gross residual disease have been left in the hands, and eyes, of the surgeon. However, new technology has emerged which affords surgeons the ability to visualize ovarian cancer deposits that are imperceptible to the naked eye. But will this improve upon the poor cure rates for advanced ovarian cancer?
Many are familiar with the traditional definitions of “optimal” (less than 1 cm–sized deposits at any one location) and “suboptimal” (greater than 1 cm–sized deposits remaining) when referring to surgical cytoreduction of ovarian cancer. This nomenclature was introduced to define, categorize, and prognosticate patient groups after surgery. In recent years we have moved away from these descriptive definitions of ovarian cancer resection, borrowing from surgical oncology measures of surgical outcomes where “R0” defines surgical resection with negative margins, “R1” includes resection with positive microscopic margins (negative for tumor intraoperatively, but positive on microscopic pathology), and “R2” refers to macroscopic residual disease remaining.1
In ovarian cancer, surgeons have adopted the expression R0 to include patients in whom there is no gross visible or palpable residual disease, a special, favorable subgrouping of the previous “optimal” group. R1 is applied to patients with macroscopic, residual disease that fits within the traditional “optimal” cytoreduction classification (<1 cm in any one location). Obviously, these are significant variations to the traditional surgical oncology definitions, but not without supporting data. For example, patients with no gross residual disease (now defined as “R0”) have been observed to have improved survival, compared with patients who are “optimally” debulked but with R1 (<1 cm) residual disease.2 Therefore, this new goal of complete surgical resection has replaced the previous standard of “optimal” cytoreduction in which small macroscopic residual disease was acceptable.
Whether or not a surgery is completed with no gross residual disease is a subjective assessment made by the surgeon, and in practice, highly inaccurate. When a posttrial ad hoc analysis of 1,873 patients with advanced ovarian cancer who had been enrolled in a Gynecologic Oncology Group cooperative trial correlated surgeons’ assessments of “optimal” cytoreduction with objective postoperative radiographic findings (performed, on average, less than 1 month postoperatively) they found that postoperative CT scans identified lesions >1 cm in 40% of cases that had been characterized by surgeons as an “optimal” cytoreduction.3 Most commonly, discrepant lesions were identified in the upper abdominal quadrants and retroperitoneal aortic nodal regions. Therefore, surgeons’ subjective assessment of cytoreduction is prone to error, and given how important the completeness of cytoreduction is for clinical outcomes, there is interest in discovering methods to improve upon surgeons’ ability to discriminate volume of disease.
Pafolacianine (Cytalux, On Target Laboratories) is a novel drug that binds a fluorescent molecule to folic acid targeting the folate alpha receptors which are overexpressed on nonmucinous epithelial ovarian cancer cells compared with adjacent nonmalignant tissues.4 The drug is intravenously infused preoperatively and then visualized with companion near-infrared imaging devices during surgery to visualize its fluorescent signal where it is bound to ovarian cancer implants. In a phase 2 study of 178 patients with confirmed or suspected ovarian cancer, pafolacianine was able to detect implants of ovarian cancer in 26.9% of cases where the surgeon’s visual inspection was negative.5 Of note, the false-positive rate of this drug was not trivial, at 20%. Based on this efficacy data, the drug has been granted FDA approved for use in ovarian cancer surgery to augment the surgeon’s visualization of cancer. However, important questions remain unanswered by these preliminary data.
Will removal of additional microscopic ovarian cancer implants, only seen by pafolacianine, improve the survival of patients with ovarian cancer, and what effect will the addition of this extra surgery have on their surgical morbidity and risk? The use of pafolacianine to augment ovarian cancer debulking surgeries pivots on the premise that ovarian cancer outcomes are determined by surgical “effort” more than the biology of the disease. Otherwise said: The more we surgically remove, the more we cure. But this seems an old-fashioned notion, increasingly challenged by data. It has been shown that, when ovarian cancer debulking surgeries are necessarily more radical because of extensive disease distribution, prognosis is worse, compared with those patients with less extensive disease distribution.6 The effect of surgical effort contributes less than that of predetermined patterns of disease presentation. Additionally, genomic traits are different in tumors that are objectively determined to be not amenable to optimal cytoreduction, compared with resectable tumors.7 These data suggest that it is the disease, more than the surgeon, that most influences outcomes.
Additionally, the question of whether surgical removal of microscopic disease improves ovarian cancer survival has already been addressed with negative findings. The LION trial randomized 647 women with advanced ovarian cancer to primary cytoreductive surgery either with or without routine lymphadenectomy of clinically negative nodes.8 This study found no survival benefit to resecting clinically negative, microscopically positive nodes. In light of these data, it is difficult to imagine that there would be different results with the resection of microscopic peritoneal disease implants identified by pafolacianine.
While pafolacianine promises to move us closer to a true “R0” (negative margins) resection of ovarian cancer, is this even a feasible goal in a disease that is widely metastatic, particularly in the peritoneal cavity? What do “negative margins” mean in the peritoneal cavity? The sensitivity of pafolacianine in detecting microscopic disease is obviously not so high that it can guarantee patients a complete resection of a disseminated disease, and we still do not know what absolute benefit is derived from moving a little bit further on the continuum of surgical resection.
Perhaps augmentation of debulking is not the only, or best, use of pafolacianine for ovarian cancer surgery. Perhaps it might serve a role in diagnostics or staging of the disease rather than for a therapeutic purpose. In the meantime, we await ongoing clinical trials in this space to better inform clinicians what benefits, or harms, they might expect from the addition of this new drug as we continue to define the “optimal” surgical procedure for advanced ovarian cancer.
Dr. Emma Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no conflicts of interest.
References
1. Hermanek P, Wittekind C. Semin Surg Oncol 1994;10:12-20.
2. Elattar A et al. Cochrane Database Syst Rev 2011 Aug 10;2011(8):CD007565.
3. Eskander RN et al. Gynecol Oncol 2018;149:525-30.
4. Randall LM et al. Gynecol Oncol 2019;155:63-8.
5. Food and Drug Administration. FDA approves pafolacianine for identifying malignant ovarian cancer lesions. 2021 Dec 1.
6. Horowitz NS et al. J Clin Oncol 2015;33:937-43.
7. Lee S et al. Cell Rep. 2020;31:107502.
8. Harter P et al. N Engl J Med 2019;380:822-32.
The goal of advanced ovarian cancer surgery is to remove all gross disease, or all visible and palpable disease implants. This became the established standard when improved survival was consistently observed among patients who had undergone complete surgical resection. Traditionally, definitions of no gross residual disease have been left in the hands, and eyes, of the surgeon. However, new technology has emerged which affords surgeons the ability to visualize ovarian cancer deposits that are imperceptible to the naked eye. But will this improve upon the poor cure rates for advanced ovarian cancer?
Many are familiar with the traditional definitions of “optimal” (less than 1 cm–sized deposits at any one location) and “suboptimal” (greater than 1 cm–sized deposits remaining) when referring to surgical cytoreduction of ovarian cancer. This nomenclature was introduced to define, categorize, and prognosticate patient groups after surgery. In recent years we have moved away from these descriptive definitions of ovarian cancer resection, borrowing from surgical oncology measures of surgical outcomes where “R0” defines surgical resection with negative margins, “R1” includes resection with positive microscopic margins (negative for tumor intraoperatively, but positive on microscopic pathology), and “R2” refers to macroscopic residual disease remaining.1
In ovarian cancer, surgeons have adopted the expression R0 to include patients in whom there is no gross visible or palpable residual disease, a special, favorable subgrouping of the previous “optimal” group. R1 is applied to patients with macroscopic, residual disease that fits within the traditional “optimal” cytoreduction classification (<1 cm in any one location). Obviously, these are significant variations to the traditional surgical oncology definitions, but not without supporting data. For example, patients with no gross residual disease (now defined as “R0”) have been observed to have improved survival, compared with patients who are “optimally” debulked but with R1 (<1 cm) residual disease.2 Therefore, this new goal of complete surgical resection has replaced the previous standard of “optimal” cytoreduction in which small macroscopic residual disease was acceptable.
Whether or not a surgery is completed with no gross residual disease is a subjective assessment made by the surgeon, and in practice, highly inaccurate. When a posttrial ad hoc analysis of 1,873 patients with advanced ovarian cancer who had been enrolled in a Gynecologic Oncology Group cooperative trial correlated surgeons’ assessments of “optimal” cytoreduction with objective postoperative radiographic findings (performed, on average, less than 1 month postoperatively) they found that postoperative CT scans identified lesions >1 cm in 40% of cases that had been characterized by surgeons as an “optimal” cytoreduction.3 Most commonly, discrepant lesions were identified in the upper abdominal quadrants and retroperitoneal aortic nodal regions. Therefore, surgeons’ subjective assessment of cytoreduction is prone to error, and given how important the completeness of cytoreduction is for clinical outcomes, there is interest in discovering methods to improve upon surgeons’ ability to discriminate volume of disease.
Pafolacianine (Cytalux, On Target Laboratories) is a novel drug that binds a fluorescent molecule to folic acid targeting the folate alpha receptors which are overexpressed on nonmucinous epithelial ovarian cancer cells compared with adjacent nonmalignant tissues.4 The drug is intravenously infused preoperatively and then visualized with companion near-infrared imaging devices during surgery to visualize its fluorescent signal where it is bound to ovarian cancer implants. In a phase 2 study of 178 patients with confirmed or suspected ovarian cancer, pafolacianine was able to detect implants of ovarian cancer in 26.9% of cases where the surgeon’s visual inspection was negative.5 Of note, the false-positive rate of this drug was not trivial, at 20%. Based on this efficacy data, the drug has been granted FDA approved for use in ovarian cancer surgery to augment the surgeon’s visualization of cancer. However, important questions remain unanswered by these preliminary data.
Will removal of additional microscopic ovarian cancer implants, only seen by pafolacianine, improve the survival of patients with ovarian cancer, and what effect will the addition of this extra surgery have on their surgical morbidity and risk? The use of pafolacianine to augment ovarian cancer debulking surgeries pivots on the premise that ovarian cancer outcomes are determined by surgical “effort” more than the biology of the disease. Otherwise said: The more we surgically remove, the more we cure. But this seems an old-fashioned notion, increasingly challenged by data. It has been shown that, when ovarian cancer debulking surgeries are necessarily more radical because of extensive disease distribution, prognosis is worse, compared with those patients with less extensive disease distribution.6 The effect of surgical effort contributes less than that of predetermined patterns of disease presentation. Additionally, genomic traits are different in tumors that are objectively determined to be not amenable to optimal cytoreduction, compared with resectable tumors.7 These data suggest that it is the disease, more than the surgeon, that most influences outcomes.
Additionally, the question of whether surgical removal of microscopic disease improves ovarian cancer survival has already been addressed with negative findings. The LION trial randomized 647 women with advanced ovarian cancer to primary cytoreductive surgery either with or without routine lymphadenectomy of clinically negative nodes.8 This study found no survival benefit to resecting clinically negative, microscopically positive nodes. In light of these data, it is difficult to imagine that there would be different results with the resection of microscopic peritoneal disease implants identified by pafolacianine.
While pafolacianine promises to move us closer to a true “R0” (negative margins) resection of ovarian cancer, is this even a feasible goal in a disease that is widely metastatic, particularly in the peritoneal cavity? What do “negative margins” mean in the peritoneal cavity? The sensitivity of pafolacianine in detecting microscopic disease is obviously not so high that it can guarantee patients a complete resection of a disseminated disease, and we still do not know what absolute benefit is derived from moving a little bit further on the continuum of surgical resection.
Perhaps augmentation of debulking is not the only, or best, use of pafolacianine for ovarian cancer surgery. Perhaps it might serve a role in diagnostics or staging of the disease rather than for a therapeutic purpose. In the meantime, we await ongoing clinical trials in this space to better inform clinicians what benefits, or harms, they might expect from the addition of this new drug as we continue to define the “optimal” surgical procedure for advanced ovarian cancer.
Dr. Emma Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She has no conflicts of interest.
References
1. Hermanek P, Wittekind C. Semin Surg Oncol 1994;10:12-20.
2. Elattar A et al. Cochrane Database Syst Rev 2011 Aug 10;2011(8):CD007565.
3. Eskander RN et al. Gynecol Oncol 2018;149:525-30.
4. Randall LM et al. Gynecol Oncol 2019;155:63-8.
5. Food and Drug Administration. FDA approves pafolacianine for identifying malignant ovarian cancer lesions. 2021 Dec 1.
6. Horowitz NS et al. J Clin Oncol 2015;33:937-43.
7. Lee S et al. Cell Rep. 2020;31:107502.
8. Harter P et al. N Engl J Med 2019;380:822-32.
This doc still supports NP/PA-led care ... with caveats
Two years ago, I argued that independent care from nurse practitioners (NPs) and physician assistants (PAs) would not have ill effects on health outcomes. To the surprise of no one, NPs and PAs embraced the argument; physicians clobbered it.
My case had three pegs: One was that medicine isn’t rocket science and clinicians control a lot less than we think we do. The second peg was that technology levels the playing field of clinical care. High-sensitivity troponin assays, for instance, make missing MI a lot less likely. The third peg was empirical: Studies have found little difference in MD versus non–MD-led care. Looking back, I now see empiricism as the weakest part of the argument because the studies had so many limitations.
I update this viewpoint now because health care is increasingly delivered by NPs and PAs. And there are two concerning trends regarding NP education and experience. First is that nurses are turning to advanced practitioner training earlier in their careers – without gathering much bedside experience. And these training programs are increasingly likely to be online, with minimal hands-on clinical tutoring.
Education and experience pop in my head often. Not every day, but many days I think back to my lucky 7 years in Indiana learning under the supervision of master clinicians – at a time when trainees were allowed the leeway to make decisions ... and mistakes. Then, when I joined private practice, I continued to learn from experienced practitioners.
It would be foolish to argue that training and experience aren’t important.
But here’s the thing:
I will make three points: First, I will bolster two of my old arguments as to why we shouldn’t be worried about non-MD clinicians, then I will propose some ideas to increase confidence in NP and PA care.
Health care does not equal health
On the matter of how much clinicians affect outcomes, a recently published randomized controlled trial performed in India found that subsidizing insurance care led to increased utilization of hospital services but had no significant effect on health outcomes. This follows the RAND and Oregon Health Insurance studies in the United States, which largely reported similar results.
We should also not dismiss the fact that – despite the massive technology gains over the past half-century in digital health and artificial intelligence and increased use of quality measures, new drugs and procedures, and mega-medical centers – the average lifespan of Americans is flat to declining (in most ethnic and racial groups). Worse than no gains in longevity, perhaps, is that death from diseases like dementia and Parkinson’s disease are on the rise.
A neutral Martian would look down and wonder why all this health care hasn’t translated to longer and better lives. The causes of this paradox remain speculative, and are for another column, but the point remains that – on average – more health care is clearly not delivering more health. And if that is true, one may deduce that much of U.S. health care is marginal when it comes to affecting major outcomes.
It’s about the delta
Logos trumps pathos. Sure, my physician colleagues can tell scary anecdotes of bad outcomes caused by an inexperienced NP or PA. I would counter that by saying I have sat on our hospital’s peer review committee for 2 decades, including the era before NPs or PAs were practicing, and I have plenty of stories of physician errors. These include, of course, my own errors.
Logos: We must consider the difference between non–MD-led care and MD-led care.
My arguments from 2020 remain relevant today. Most medical problems are not engineering puzzles. Many, perhaps most, patients fall into an easy protocol – say, chest pain, dyspnea, or atrial fibrillation. With basic training, a motivated serious person quickly gains skill in recognizing and treating everyday problems.
And just 2 years on, technology further levels the playing field. Consider radiology in 2022 – it’s easy to take for granted the speed of the CT scan, the fidelity of the MRI, and the easy access to both in the U.S. hospital system. Less experienced clinicians have never had more tools to assist with diagnostics and therapeutics.
The expansion of team-based care has also mitigated the effects of inexperience. It took Americans longer than Canadians to figure out how helpful pharmacists could be. Pharmacists in my hospital now help us dose complicated medicines and protect us against prescribing errors.
Then there is the immediate access to online information. Gone are the days when you had to memorize long-QT syndromes. Book knowledge – that I spent years acquiring – now comes in seconds. The other day an NP corrected me. I asked, Are you sure? Boom, she took out her phone and showed me the evidence.
In sum, if it were even possible to measure the clinical competence of care from NP and PA versus physicians, there would be two bell-shaped curves with a tremendous amount of overlap. And that overlap would steadily increase as a given NP or PA gathered experience. (The NP in our electrophysiology division has more than 25 years’ experience in heart rhythm care, and it is common for colleagues to call her before one of us docs. Rightly so.)
Three basic proposals regarding NP and PA care
To ensure quality of care, I have three proposals.
It has always seemed strange to me that an NP or PA can flip from one field to another without a period of training. I can’t just change practice from electrophysiology to dermatology without doing a residency. But NPs and PAs can.
My first proposal would be that NPs and PAs spend a substantial period of training in a field before practice – a legit apprenticeship. The duration of this period is a matter of debate, but it ought to be standardized.
My second proposal is that, if physicians are required to pass certification exams, so should NPs. (PAs have an exam every 10 years.) The exam should be the same as (or very similar to) the physician exam, and it should be specific to their field of practice.
While I have argued (and still feel) that the American Board of Internal Medicine brand of certification is dubious, the fact remains that physicians must maintain proficiency in their field. Requiring NPs and PAs to do the same would help foster specialization. And while I can’t cite empirical evidence, specialization seems super-important. We have NPs at my hospital who have been in the same area for years, and they exude clinical competence.
Finally, I have come to believe that the best way for nearly any clinician to practice medicine is as part of a team. (The exception being primary care in rural areas where there are clinician shortages.)
On the matter of team care, I’ve practiced for a long time, but nearly every day I run situations by a colleague; often this person is an NP. The economist Friedrich Hayek proposed that dispersed knowledge always outpaces the wisdom of any individual. That notion pertains well to the increasing complexities and specialization of modern medical practice.
A person who commits to learning one area of medicine, enjoys helping people, asks often for help, and has the support of colleagues is set up to be a successful clinician – whether the letters after their name are APRN, PA, DO, or MD.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky. He did not report any relevant financial disclosures. A version of this article first appeared on Medscape.com.
Two years ago, I argued that independent care from nurse practitioners (NPs) and physician assistants (PAs) would not have ill effects on health outcomes. To the surprise of no one, NPs and PAs embraced the argument; physicians clobbered it.
My case had three pegs: One was that medicine isn’t rocket science and clinicians control a lot less than we think we do. The second peg was that technology levels the playing field of clinical care. High-sensitivity troponin assays, for instance, make missing MI a lot less likely. The third peg was empirical: Studies have found little difference in MD versus non–MD-led care. Looking back, I now see empiricism as the weakest part of the argument because the studies had so many limitations.
I update this viewpoint now because health care is increasingly delivered by NPs and PAs. And there are two concerning trends regarding NP education and experience. First is that nurses are turning to advanced practitioner training earlier in their careers – without gathering much bedside experience. And these training programs are increasingly likely to be online, with minimal hands-on clinical tutoring.
Education and experience pop in my head often. Not every day, but many days I think back to my lucky 7 years in Indiana learning under the supervision of master clinicians – at a time when trainees were allowed the leeway to make decisions ... and mistakes. Then, when I joined private practice, I continued to learn from experienced practitioners.
It would be foolish to argue that training and experience aren’t important.
But here’s the thing:
I will make three points: First, I will bolster two of my old arguments as to why we shouldn’t be worried about non-MD clinicians, then I will propose some ideas to increase confidence in NP and PA care.
Health care does not equal health
On the matter of how much clinicians affect outcomes, a recently published randomized controlled trial performed in India found that subsidizing insurance care led to increased utilization of hospital services but had no significant effect on health outcomes. This follows the RAND and Oregon Health Insurance studies in the United States, which largely reported similar results.
We should also not dismiss the fact that – despite the massive technology gains over the past half-century in digital health and artificial intelligence and increased use of quality measures, new drugs and procedures, and mega-medical centers – the average lifespan of Americans is flat to declining (in most ethnic and racial groups). Worse than no gains in longevity, perhaps, is that death from diseases like dementia and Parkinson’s disease are on the rise.
A neutral Martian would look down and wonder why all this health care hasn’t translated to longer and better lives. The causes of this paradox remain speculative, and are for another column, but the point remains that – on average – more health care is clearly not delivering more health. And if that is true, one may deduce that much of U.S. health care is marginal when it comes to affecting major outcomes.
It’s about the delta
Logos trumps pathos. Sure, my physician colleagues can tell scary anecdotes of bad outcomes caused by an inexperienced NP or PA. I would counter that by saying I have sat on our hospital’s peer review committee for 2 decades, including the era before NPs or PAs were practicing, and I have plenty of stories of physician errors. These include, of course, my own errors.
Logos: We must consider the difference between non–MD-led care and MD-led care.
My arguments from 2020 remain relevant today. Most medical problems are not engineering puzzles. Many, perhaps most, patients fall into an easy protocol – say, chest pain, dyspnea, or atrial fibrillation. With basic training, a motivated serious person quickly gains skill in recognizing and treating everyday problems.
And just 2 years on, technology further levels the playing field. Consider radiology in 2022 – it’s easy to take for granted the speed of the CT scan, the fidelity of the MRI, and the easy access to both in the U.S. hospital system. Less experienced clinicians have never had more tools to assist with diagnostics and therapeutics.
The expansion of team-based care has also mitigated the effects of inexperience. It took Americans longer than Canadians to figure out how helpful pharmacists could be. Pharmacists in my hospital now help us dose complicated medicines and protect us against prescribing errors.
Then there is the immediate access to online information. Gone are the days when you had to memorize long-QT syndromes. Book knowledge – that I spent years acquiring – now comes in seconds. The other day an NP corrected me. I asked, Are you sure? Boom, she took out her phone and showed me the evidence.
In sum, if it were even possible to measure the clinical competence of care from NP and PA versus physicians, there would be two bell-shaped curves with a tremendous amount of overlap. And that overlap would steadily increase as a given NP or PA gathered experience. (The NP in our electrophysiology division has more than 25 years’ experience in heart rhythm care, and it is common for colleagues to call her before one of us docs. Rightly so.)
Three basic proposals regarding NP and PA care
To ensure quality of care, I have three proposals.
It has always seemed strange to me that an NP or PA can flip from one field to another without a period of training. I can’t just change practice from electrophysiology to dermatology without doing a residency. But NPs and PAs can.
My first proposal would be that NPs and PAs spend a substantial period of training in a field before practice – a legit apprenticeship. The duration of this period is a matter of debate, but it ought to be standardized.
My second proposal is that, if physicians are required to pass certification exams, so should NPs. (PAs have an exam every 10 years.) The exam should be the same as (or very similar to) the physician exam, and it should be specific to their field of practice.
While I have argued (and still feel) that the American Board of Internal Medicine brand of certification is dubious, the fact remains that physicians must maintain proficiency in their field. Requiring NPs and PAs to do the same would help foster specialization. And while I can’t cite empirical evidence, specialization seems super-important. We have NPs at my hospital who have been in the same area for years, and they exude clinical competence.
Finally, I have come to believe that the best way for nearly any clinician to practice medicine is as part of a team. (The exception being primary care in rural areas where there are clinician shortages.)
On the matter of team care, I’ve practiced for a long time, but nearly every day I run situations by a colleague; often this person is an NP. The economist Friedrich Hayek proposed that dispersed knowledge always outpaces the wisdom of any individual. That notion pertains well to the increasing complexities and specialization of modern medical practice.
A person who commits to learning one area of medicine, enjoys helping people, asks often for help, and has the support of colleagues is set up to be a successful clinician – whether the letters after their name are APRN, PA, DO, or MD.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky. He did not report any relevant financial disclosures. A version of this article first appeared on Medscape.com.
Two years ago, I argued that independent care from nurse practitioners (NPs) and physician assistants (PAs) would not have ill effects on health outcomes. To the surprise of no one, NPs and PAs embraced the argument; physicians clobbered it.
My case had three pegs: One was that medicine isn’t rocket science and clinicians control a lot less than we think we do. The second peg was that technology levels the playing field of clinical care. High-sensitivity troponin assays, for instance, make missing MI a lot less likely. The third peg was empirical: Studies have found little difference in MD versus non–MD-led care. Looking back, I now see empiricism as the weakest part of the argument because the studies had so many limitations.
I update this viewpoint now because health care is increasingly delivered by NPs and PAs. And there are two concerning trends regarding NP education and experience. First is that nurses are turning to advanced practitioner training earlier in their careers – without gathering much bedside experience. And these training programs are increasingly likely to be online, with minimal hands-on clinical tutoring.
Education and experience pop in my head often. Not every day, but many days I think back to my lucky 7 years in Indiana learning under the supervision of master clinicians – at a time when trainees were allowed the leeway to make decisions ... and mistakes. Then, when I joined private practice, I continued to learn from experienced practitioners.
It would be foolish to argue that training and experience aren’t important.
But here’s the thing:
I will make three points: First, I will bolster two of my old arguments as to why we shouldn’t be worried about non-MD clinicians, then I will propose some ideas to increase confidence in NP and PA care.
Health care does not equal health
On the matter of how much clinicians affect outcomes, a recently published randomized controlled trial performed in India found that subsidizing insurance care led to increased utilization of hospital services but had no significant effect on health outcomes. This follows the RAND and Oregon Health Insurance studies in the United States, which largely reported similar results.
We should also not dismiss the fact that – despite the massive technology gains over the past half-century in digital health and artificial intelligence and increased use of quality measures, new drugs and procedures, and mega-medical centers – the average lifespan of Americans is flat to declining (in most ethnic and racial groups). Worse than no gains in longevity, perhaps, is that death from diseases like dementia and Parkinson’s disease are on the rise.
A neutral Martian would look down and wonder why all this health care hasn’t translated to longer and better lives. The causes of this paradox remain speculative, and are for another column, but the point remains that – on average – more health care is clearly not delivering more health. And if that is true, one may deduce that much of U.S. health care is marginal when it comes to affecting major outcomes.
It’s about the delta
Logos trumps pathos. Sure, my physician colleagues can tell scary anecdotes of bad outcomes caused by an inexperienced NP or PA. I would counter that by saying I have sat on our hospital’s peer review committee for 2 decades, including the era before NPs or PAs were practicing, and I have plenty of stories of physician errors. These include, of course, my own errors.
Logos: We must consider the difference between non–MD-led care and MD-led care.
My arguments from 2020 remain relevant today. Most medical problems are not engineering puzzles. Many, perhaps most, patients fall into an easy protocol – say, chest pain, dyspnea, or atrial fibrillation. With basic training, a motivated serious person quickly gains skill in recognizing and treating everyday problems.
And just 2 years on, technology further levels the playing field. Consider radiology in 2022 – it’s easy to take for granted the speed of the CT scan, the fidelity of the MRI, and the easy access to both in the U.S. hospital system. Less experienced clinicians have never had more tools to assist with diagnostics and therapeutics.
The expansion of team-based care has also mitigated the effects of inexperience. It took Americans longer than Canadians to figure out how helpful pharmacists could be. Pharmacists in my hospital now help us dose complicated medicines and protect us against prescribing errors.
Then there is the immediate access to online information. Gone are the days when you had to memorize long-QT syndromes. Book knowledge – that I spent years acquiring – now comes in seconds. The other day an NP corrected me. I asked, Are you sure? Boom, she took out her phone and showed me the evidence.
In sum, if it were even possible to measure the clinical competence of care from NP and PA versus physicians, there would be two bell-shaped curves with a tremendous amount of overlap. And that overlap would steadily increase as a given NP or PA gathered experience. (The NP in our electrophysiology division has more than 25 years’ experience in heart rhythm care, and it is common for colleagues to call her before one of us docs. Rightly so.)
Three basic proposals regarding NP and PA care
To ensure quality of care, I have three proposals.
It has always seemed strange to me that an NP or PA can flip from one field to another without a period of training. I can’t just change practice from electrophysiology to dermatology without doing a residency. But NPs and PAs can.
My first proposal would be that NPs and PAs spend a substantial period of training in a field before practice – a legit apprenticeship. The duration of this period is a matter of debate, but it ought to be standardized.
My second proposal is that, if physicians are required to pass certification exams, so should NPs. (PAs have an exam every 10 years.) The exam should be the same as (or very similar to) the physician exam, and it should be specific to their field of practice.
While I have argued (and still feel) that the American Board of Internal Medicine brand of certification is dubious, the fact remains that physicians must maintain proficiency in their field. Requiring NPs and PAs to do the same would help foster specialization. And while I can’t cite empirical evidence, specialization seems super-important. We have NPs at my hospital who have been in the same area for years, and they exude clinical competence.
Finally, I have come to believe that the best way for nearly any clinician to practice medicine is as part of a team. (The exception being primary care in rural areas where there are clinician shortages.)
On the matter of team care, I’ve practiced for a long time, but nearly every day I run situations by a colleague; often this person is an NP. The economist Friedrich Hayek proposed that dispersed knowledge always outpaces the wisdom of any individual. That notion pertains well to the increasing complexities and specialization of modern medical practice.
A person who commits to learning one area of medicine, enjoys helping people, asks often for help, and has the support of colleagues is set up to be a successful clinician – whether the letters after their name are APRN, PA, DO, or MD.
Dr. Mandrola practices cardiac electrophysiology in Louisville, Ky. He did not report any relevant financial disclosures. A version of this article first appeared on Medscape.com.
Ways to make sure 2022 doesn’t stink for docs
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.
Depending on the data you’re looking at, 40%-60% of physicians are burned out.
Research studies and the eye test reveal the painfully obvious: Colleagues are tired, winded, spent, and at times way past burned out. People aren’t asking me if they’re burned out. They know they’re burned out; heck, they can even recite the Maslach burnout inventory, forward and backward, in a mask, or while completing a COVID quarantine. A fair share of people know the key steps to prevent burnout and promote recovery.
What I’m starting to see more of is, “Why should I even bother to recover from this? Why pick myself up again just to get another occupational stress injury (burnout, demoralization, moral injury, etc.)?” In other words, it’s not just simply about negating burnout; it’s about supporting and facilitating the motivation to work.
We’ve been through so much with COVID that it might be challenging to remember when you saw a truly engaged work environment. No doubt, we have outstanding professionals across medicine who answer the bell every day. However, if you’ve been looking closely, many teams/units have lost a bit of the zip and pep. The synergy and trust aren’t as smooth, and at noon, everyone counts the hours to the end of the shift.
You may be thinking, Well, of course, they are; we’re still amid a pandemic, and people have been through hell. Your observation would be correct, except I’ve personally seen some teams weather the pandemic storm and still remain engaged (some even more involved).
The No. 1 consult result for the GW Resiliency and Well-Being Center, where I work, has been on lectures for burnout. The R&WC has given so many of these lectures that my dreams take the form of a PowerPoint presentation. Overall the talks have gone very well. We’ve added skills sections on practices of whole-person care. We’ve blitzed the daylights out of restorative sleep, yet I know we are still searching for the correct narrative.
Motivated staff, faculty, and students will genuinely take in the information and follow the recommendations; however, they still struggle to find that drive and zest for work. Yes, moving from burnout to neutral is reasonable but likely won’t move the needle of your professional or personal life. We need to have the emotional energy and the clear desire to utilize that energy for a meaningful purpose.
Talking about burnout in specific ways is straightforward and, in my opinion, much easier than talking about engagement. Part of the challenge when trying to discuss engagement is that people can feel invalidated or that you’re telling them to be stoic. Or worse yet, that the problem of burnout primarily lies with them. It’s essential to recognize the role of an organizational factor in burnout (approximately 80%, depending on the study); still, even if you address burnout, people may not be miserable, but it doesn’t mean they will stay at their current job (please cue intro music for the Great Resignation).
Engagement models have existed for some time and certainly have gained much more attention in health care settings over the past 2 decades. Engagement can be described as having three components: dedication, vigor, and absorption. When a person is filling all three of these components over time, presto – you get the much-sought-after state of the supremely engaged professional.
These models definitely give us excellent starting points to approach engagement from a pre-COVID era. In COVID and beyond, I’m not sure how these models will stand up in a hybrid work environment, where autonomy and flexibility could be more valued than ever. Personally, COVID revealed some things I was missing in my work pre-COVID:
- Time to think and process. This was one of the great things about being a consultation-liaison psychiatrist; it was literally feast or famine.
- Doing what I’m talented at and really enjoy.
- Time is short, and I want to be more present in the life of my family.
The list above isn’t exhaustive, but I’ve found them to be my own personal recipe for being engaged. Over the next series of articles, I’m going to focus on engagement and factors related to key resilience. These articles will be informed by a front-line view from my colleagues, and hopefully start to separate the myth from reality on the subject of health professional engagement and resilience.
Everyone be safe and well!
A version of this article first appeared on Medscape.com.