User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
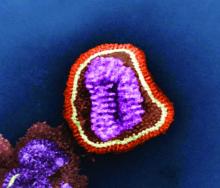
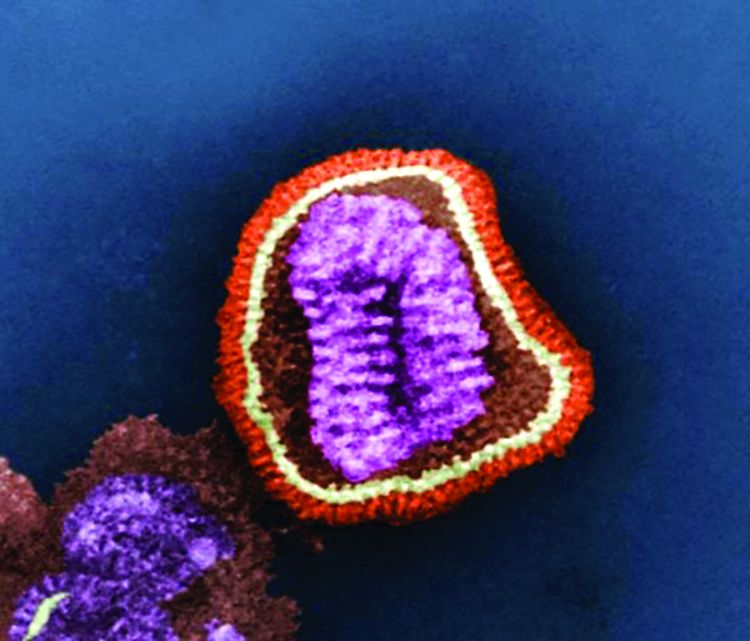
FDA grants emergency use authorization to Lilly’s antibody COVID-19 therapy
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
New eGFR equation ‘less biased’ by age, kidney function; some disagree
The European Kidney Function Consortium (EKFC) equation surpasses existing equations by “resulting in generally lower bias across the spectrum of age and kidney function,” its developers wrote in an article published online Nov. 9 in Annals of Internal Medicine.
“The new EKFC equation may have helpful properties and perform better in estimating GFR, compared with the current KDIGO [Kidney Disease: Improving Global Outcomes]-recommended equations,” they added.
The primary KDIGO-recommended equation in its most recent guideline was the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, designed for adults, and a companion equation, the CKiD, covers children and adolescents.
“Key in our [new] equation is the adjustment for differences in serum creatinine generation between children and adults, or between men and women,” lead author Hans Pottel, PhD, KU Leuven (Belgium), said in an interview.
In an accompanying editorial, Andrew M. Levey, MD, and associates wrote: “We agree that a single eGFR equation that can be used in children and adults and performs well in the transition from adolescence to young adulthood is a worthy goal.”
“But the claim of equivalent or superior performance, compared with the CKD-EPI equation is not conclusive,” claimed Dr. Levey, who led the research team that developed the CKD-EPI equation, and coauthors.
Dr. Levey is professor of medicine at Tufts University, Boston.
What’s new is Q
Dr. Pottel and codevelopers devised what they call Q values: age- and sex-dependent median creatinine levels in normal individuals.
Q values act to “normalize or rescale creatinine before entering it into the equation, because we know that creatinine generation is different” based on factors that include age, sex, and muscle mass.
The EKFC equation extends the CKD-EPI equation and first eGFR equation by using Q values and applying across age ranges, like the full-age spectrum (FAS) equation, first reported in 2016 by a team led by Dr. Pottel.
“Although the FAS equation was designed to overcome the challenge in measuring GFR in patients transitioning from adolescence to adult nephrology care, it also underestimates GFR at low serum creatinine values and in patients with chronic kidney disease,” wrote Dr. Pottel and coauthors.
Hence, their intent to tweak the FAS equation to overcome this limitation and create the EKFC equation.
“The new equation combines the strengths of the CKD-EPI and FAS equations,” they woite.
However, “we acknowledge that lack of precision is still a major problem with all eGFR equations,” including the new EKFC, they added.
Editorialists dispute better performance of EKFC over CKD-EPI
In their editorial, Dr. Levey and coauthors noted the EKFC equations and other adapted equations in development “represent a conceptual advance over the FAS equations,” but they dispute the claims of better performance, compared with the CKD-EPI.
“We compared the performance of the EKFC and CKD-EPI equations in a different, large external validation population of Black and non-Black adults,” the external population used to validate the CKD-EPI equation, the editorialists reported.
The upshot was “our results did not confirm the author’s conclusions” about the EKFC equation.
In response, Dr. Pottel highlighted that the EKFC equation is currently not designed for use in Black patients.
“With its derivation and validation now reported in the new article, the EKFC equation is fully validated and ready for routine use in Whites,” he said. “We plan to evaluate and possibly fine tune our equation for its application in other ethnicities.”
Regarding the inferior performance, compared with the CKD-EPI equation in the non-Black population tested by the editorialists, Dr. Pottel cited “calibration issues for serum creatinine” that some experts have found in the datasets compiled by developers of the CKI-EPI equation that could limit the utility of these data.
Still room for improvement; app hopefully coming next year
Dr. Pottel and coauthors developed and validated the EKFC equation with data from 19,629 patients drawn from 13 cohorts. This included 11,251 patients from seven cohorts for development and internal validation, and 8378 from six cohorts for external validation. The EKFC effort received endorsement from the European Renal Association–European Dialysis and Transplant Association.
However, “We acknowledge that there is still room for improvement,” Dr. Pottel said.
Although the new report presents the EKFC equations (actually two slightly different equations depending on whether a patient’s serum creatinine is higher or lower than the relevant Q value), most potential users will likely find the equations easier to work with once they’re in an app form that allows someone to simply plug in age, sex, and serum creatinine level. That app currently doesn’t exist but is coming soon, promised Dr. Pottel.
“I hope to have an electronic tool by the beginning of 2021,” he said. “I have to find a programmer who can do this for me.”
The EKFC project has received no commercial funding. Dr. Pottel reported no relevant financial relationships. Dr. Levey has reported receiving research funding from AstraZeneca.
A version of this article originally appeared on Medscape.com.
The European Kidney Function Consortium (EKFC) equation surpasses existing equations by “resulting in generally lower bias across the spectrum of age and kidney function,” its developers wrote in an article published online Nov. 9 in Annals of Internal Medicine.
“The new EKFC equation may have helpful properties and perform better in estimating GFR, compared with the current KDIGO [Kidney Disease: Improving Global Outcomes]-recommended equations,” they added.
The primary KDIGO-recommended equation in its most recent guideline was the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, designed for adults, and a companion equation, the CKiD, covers children and adolescents.
“Key in our [new] equation is the adjustment for differences in serum creatinine generation between children and adults, or between men and women,” lead author Hans Pottel, PhD, KU Leuven (Belgium), said in an interview.
In an accompanying editorial, Andrew M. Levey, MD, and associates wrote: “We agree that a single eGFR equation that can be used in children and adults and performs well in the transition from adolescence to young adulthood is a worthy goal.”
“But the claim of equivalent or superior performance, compared with the CKD-EPI equation is not conclusive,” claimed Dr. Levey, who led the research team that developed the CKD-EPI equation, and coauthors.
Dr. Levey is professor of medicine at Tufts University, Boston.
What’s new is Q
Dr. Pottel and codevelopers devised what they call Q values: age- and sex-dependent median creatinine levels in normal individuals.
Q values act to “normalize or rescale creatinine before entering it into the equation, because we know that creatinine generation is different” based on factors that include age, sex, and muscle mass.
The EKFC equation extends the CKD-EPI equation and first eGFR equation by using Q values and applying across age ranges, like the full-age spectrum (FAS) equation, first reported in 2016 by a team led by Dr. Pottel.
“Although the FAS equation was designed to overcome the challenge in measuring GFR in patients transitioning from adolescence to adult nephrology care, it also underestimates GFR at low serum creatinine values and in patients with chronic kidney disease,” wrote Dr. Pottel and coauthors.
Hence, their intent to tweak the FAS equation to overcome this limitation and create the EKFC equation.
“The new equation combines the strengths of the CKD-EPI and FAS equations,” they woite.
However, “we acknowledge that lack of precision is still a major problem with all eGFR equations,” including the new EKFC, they added.
Editorialists dispute better performance of EKFC over CKD-EPI
In their editorial, Dr. Levey and coauthors noted the EKFC equations and other adapted equations in development “represent a conceptual advance over the FAS equations,” but they dispute the claims of better performance, compared with the CKD-EPI.
“We compared the performance of the EKFC and CKD-EPI equations in a different, large external validation population of Black and non-Black adults,” the external population used to validate the CKD-EPI equation, the editorialists reported.
The upshot was “our results did not confirm the author’s conclusions” about the EKFC equation.
In response, Dr. Pottel highlighted that the EKFC equation is currently not designed for use in Black patients.
“With its derivation and validation now reported in the new article, the EKFC equation is fully validated and ready for routine use in Whites,” he said. “We plan to evaluate and possibly fine tune our equation for its application in other ethnicities.”
Regarding the inferior performance, compared with the CKD-EPI equation in the non-Black population tested by the editorialists, Dr. Pottel cited “calibration issues for serum creatinine” that some experts have found in the datasets compiled by developers of the CKI-EPI equation that could limit the utility of these data.
Still room for improvement; app hopefully coming next year
Dr. Pottel and coauthors developed and validated the EKFC equation with data from 19,629 patients drawn from 13 cohorts. This included 11,251 patients from seven cohorts for development and internal validation, and 8378 from six cohorts for external validation. The EKFC effort received endorsement from the European Renal Association–European Dialysis and Transplant Association.
However, “We acknowledge that there is still room for improvement,” Dr. Pottel said.
Although the new report presents the EKFC equations (actually two slightly different equations depending on whether a patient’s serum creatinine is higher or lower than the relevant Q value), most potential users will likely find the equations easier to work with once they’re in an app form that allows someone to simply plug in age, sex, and serum creatinine level. That app currently doesn’t exist but is coming soon, promised Dr. Pottel.
“I hope to have an electronic tool by the beginning of 2021,” he said. “I have to find a programmer who can do this for me.”
The EKFC project has received no commercial funding. Dr. Pottel reported no relevant financial relationships. Dr. Levey has reported receiving research funding from AstraZeneca.
A version of this article originally appeared on Medscape.com.
The European Kidney Function Consortium (EKFC) equation surpasses existing equations by “resulting in generally lower bias across the spectrum of age and kidney function,” its developers wrote in an article published online Nov. 9 in Annals of Internal Medicine.
“The new EKFC equation may have helpful properties and perform better in estimating GFR, compared with the current KDIGO [Kidney Disease: Improving Global Outcomes]-recommended equations,” they added.
The primary KDIGO-recommended equation in its most recent guideline was the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, designed for adults, and a companion equation, the CKiD, covers children and adolescents.
“Key in our [new] equation is the adjustment for differences in serum creatinine generation between children and adults, or between men and women,” lead author Hans Pottel, PhD, KU Leuven (Belgium), said in an interview.
In an accompanying editorial, Andrew M. Levey, MD, and associates wrote: “We agree that a single eGFR equation that can be used in children and adults and performs well in the transition from adolescence to young adulthood is a worthy goal.”
“But the claim of equivalent or superior performance, compared with the CKD-EPI equation is not conclusive,” claimed Dr. Levey, who led the research team that developed the CKD-EPI equation, and coauthors.
Dr. Levey is professor of medicine at Tufts University, Boston.
What’s new is Q
Dr. Pottel and codevelopers devised what they call Q values: age- and sex-dependent median creatinine levels in normal individuals.
Q values act to “normalize or rescale creatinine before entering it into the equation, because we know that creatinine generation is different” based on factors that include age, sex, and muscle mass.
The EKFC equation extends the CKD-EPI equation and first eGFR equation by using Q values and applying across age ranges, like the full-age spectrum (FAS) equation, first reported in 2016 by a team led by Dr. Pottel.
“Although the FAS equation was designed to overcome the challenge in measuring GFR in patients transitioning from adolescence to adult nephrology care, it also underestimates GFR at low serum creatinine values and in patients with chronic kidney disease,” wrote Dr. Pottel and coauthors.
Hence, their intent to tweak the FAS equation to overcome this limitation and create the EKFC equation.
“The new equation combines the strengths of the CKD-EPI and FAS equations,” they woite.
However, “we acknowledge that lack of precision is still a major problem with all eGFR equations,” including the new EKFC, they added.
Editorialists dispute better performance of EKFC over CKD-EPI
In their editorial, Dr. Levey and coauthors noted the EKFC equations and other adapted equations in development “represent a conceptual advance over the FAS equations,” but they dispute the claims of better performance, compared with the CKD-EPI.
“We compared the performance of the EKFC and CKD-EPI equations in a different, large external validation population of Black and non-Black adults,” the external population used to validate the CKD-EPI equation, the editorialists reported.
The upshot was “our results did not confirm the author’s conclusions” about the EKFC equation.
In response, Dr. Pottel highlighted that the EKFC equation is currently not designed for use in Black patients.
“With its derivation and validation now reported in the new article, the EKFC equation is fully validated and ready for routine use in Whites,” he said. “We plan to evaluate and possibly fine tune our equation for its application in other ethnicities.”
Regarding the inferior performance, compared with the CKD-EPI equation in the non-Black population tested by the editorialists, Dr. Pottel cited “calibration issues for serum creatinine” that some experts have found in the datasets compiled by developers of the CKI-EPI equation that could limit the utility of these data.
Still room for improvement; app hopefully coming next year
Dr. Pottel and coauthors developed and validated the EKFC equation with data from 19,629 patients drawn from 13 cohorts. This included 11,251 patients from seven cohorts for development and internal validation, and 8378 from six cohorts for external validation. The EKFC effort received endorsement from the European Renal Association–European Dialysis and Transplant Association.
However, “We acknowledge that there is still room for improvement,” Dr. Pottel said.
Although the new report presents the EKFC equations (actually two slightly different equations depending on whether a patient’s serum creatinine is higher or lower than the relevant Q value), most potential users will likely find the equations easier to work with once they’re in an app form that allows someone to simply plug in age, sex, and serum creatinine level. That app currently doesn’t exist but is coming soon, promised Dr. Pottel.
“I hope to have an electronic tool by the beginning of 2021,” he said. “I have to find a programmer who can do this for me.”
The EKFC project has received no commercial funding. Dr. Pottel reported no relevant financial relationships. Dr. Levey has reported receiving research funding from AstraZeneca.
A version of this article originally appeared on Medscape.com.
Study IDs microbial signature of celiac disease in children
Eleven operational taxonomic units (OTUs) of fecal bacteria were less abundant in children with celiac disease than in healthy children, according to the findings of a study published in Gastroenterology.
This microbial signature correctly identified approximately four out of five cases of celiac disease, regardless of whether children were newly diagnosed or had already modified their diet, reported Konstantina Zafeiropoulou and Ben Nichols, PhD, of the Glasgow Royal Infirmary. “It is not clear whether the microbes identified [in this study] contribute to the pathogenesis of celiac disease or are the result of it. Future research should explore the role of the disease-specific species identified here,” the researchers wrote in Gastroenterology.
Celiac disease is multifactorial. While up to 40% of people are genetically predisposed, only a small proportion develop it, suggesting that environmental factors are key to pathogenesis. Recent studies have linked celiac disease with alterations in the gut microbiome, but it is unclear whether dysbiosis is pathogenic or a secondary effect of disease processes such as nutrient malabsorption, or whether dysbiosis is present at disease onset or results from a gluten-free diet.
For the study, the researchers performed gas chromatography and 16S ribosomal RNA sequencing of fecal samples from 141 children, including 20 with newly biopsy-confirmed, previously untreated celiac disease, 45 who were previously diagnosed and on a gluten-free diet, 19 unaffected siblings, and 57 healthy children who were not on regular medications and had no history of chronic gastrointestinal symptoms. A single fecal sample was tested for all but the previously untreated children, who were tested at baseline and then after 6 and 12 months on a gluten-free diet.
Children with new-onset celiac disease showed no evidence of dysbiosis, while a gluten-free diet explained up to 2.8% of variation in microbiota between patients and controls. Microbial alpha diversity, a measure of species-level diversity, was generally similar among groups, but between 3% and 5% of all taxa differed. Irrespective of treatment, the decreased abundance of the 11 OTUs was diagnostic for celiac disease with an error rate of 21.5% (P < .001 vs. random classification). Notably, most of these 11 discrepant OTUs were associated with nutrient or food group intake and with biomarkers of gluten ingestion, the researchers said. Gas chromatography showed that, after patients started a gluten-free diet, fecal levels of butyrate and ammonia decreased.
“Even though we identified differences in the abundance of a few species between patients with untreated celiac disease and healthy controls, the profound microbial dysbiosis noted in Crohn’s disease was not observed, at least using crude diversity indices,” the investigators commented. “Although several alterations in the intestinal microbiota of children with established celiac disease appear to be effects of a gluten-free diet, there are specific bacteria that are distinct biomarkers of celiac disease.”
Future research might involve performing in vitro tests of “candidate” bacteria, coculturing these bacteria with human immune cells, and studying whether dietary interventions alter the relative abundance of these bacteria in the gut microbiome, the researchers said.
Nutricia Research Foundation, the Biotechnology and Biological Sciences Research Council, and The Catherine McEwan Foundation provided funding. Three coinvestigators disclosed ties to Nutricia, 4D Pharma, AbbVie, Celltrion, Janssen, Takeda, and several other pharmaceutical companies. One coinvestigator reported chairing the working group for ISLI Europe. The remaining investigators reported having no conflicts of interest.
SOURCE: Zafeiropoulou K et al. Gastroenterology. 2020 Aug 10;S0016-5085(20)35023-X. doi: 10.1053/j.gastro.2020.08.007.
It is well known that gluten ingestion in genetically susceptible individuals does not guarantee celiac disease, and research over the past decade has searched for environmental triggers. Gut microbiota play a role in activation of innate immunity, which leads to the adaptive immune response and the small bowel damage that is characteristic of celiac disease. The authors of this study sought to identify whether there is a distinct microbial pattern among celiac disease patients, both those with treated and untreated disease, in comparison with healthy controls and healthy siblings.
A significantly different microbial profile and metabolites were identified in subjects on gluten-free diets. The consequences of the gluten-free diet are an important consideration when committing a patient to this life-long therapy. The microbiome changes may play a role in persistent symptoms and the increased health conditions we see in treated celiac disease. Those on a gluten-free diet have other micronutrient deficiencies in addition to microbiome changes and the health sequelae of this are not fully understood. A gluten-free diet focused on restoring the normal gut flora through probiotic or gluten-free prebiotic or fiber supplementation in celiac disease patients could prove beneficial.
Dawn Wiese Adams, MD, MS, is assistant professor and medical director, Center for Human Nutrition, department of gastroenterology, hepatology, and nutrition, Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
It is well known that gluten ingestion in genetically susceptible individuals does not guarantee celiac disease, and research over the past decade has searched for environmental triggers. Gut microbiota play a role in activation of innate immunity, which leads to the adaptive immune response and the small bowel damage that is characteristic of celiac disease. The authors of this study sought to identify whether there is a distinct microbial pattern among celiac disease patients, both those with treated and untreated disease, in comparison with healthy controls and healthy siblings.
A significantly different microbial profile and metabolites were identified in subjects on gluten-free diets. The consequences of the gluten-free diet are an important consideration when committing a patient to this life-long therapy. The microbiome changes may play a role in persistent symptoms and the increased health conditions we see in treated celiac disease. Those on a gluten-free diet have other micronutrient deficiencies in addition to microbiome changes and the health sequelae of this are not fully understood. A gluten-free diet focused on restoring the normal gut flora through probiotic or gluten-free prebiotic or fiber supplementation in celiac disease patients could prove beneficial.
Dawn Wiese Adams, MD, MS, is assistant professor and medical director, Center for Human Nutrition, department of gastroenterology, hepatology, and nutrition, Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
It is well known that gluten ingestion in genetically susceptible individuals does not guarantee celiac disease, and research over the past decade has searched for environmental triggers. Gut microbiota play a role in activation of innate immunity, which leads to the adaptive immune response and the small bowel damage that is characteristic of celiac disease. The authors of this study sought to identify whether there is a distinct microbial pattern among celiac disease patients, both those with treated and untreated disease, in comparison with healthy controls and healthy siblings.
A significantly different microbial profile and metabolites were identified in subjects on gluten-free diets. The consequences of the gluten-free diet are an important consideration when committing a patient to this life-long therapy. The microbiome changes may play a role in persistent symptoms and the increased health conditions we see in treated celiac disease. Those on a gluten-free diet have other micronutrient deficiencies in addition to microbiome changes and the health sequelae of this are not fully understood. A gluten-free diet focused on restoring the normal gut flora through probiotic or gluten-free prebiotic or fiber supplementation in celiac disease patients could prove beneficial.
Dawn Wiese Adams, MD, MS, is assistant professor and medical director, Center for Human Nutrition, department of gastroenterology, hepatology, and nutrition, Vanderbilt University Medical Center, Nashville, Tenn. She has no conflicts of interest.
Eleven operational taxonomic units (OTUs) of fecal bacteria were less abundant in children with celiac disease than in healthy children, according to the findings of a study published in Gastroenterology.
This microbial signature correctly identified approximately four out of five cases of celiac disease, regardless of whether children were newly diagnosed or had already modified their diet, reported Konstantina Zafeiropoulou and Ben Nichols, PhD, of the Glasgow Royal Infirmary. “It is not clear whether the microbes identified [in this study] contribute to the pathogenesis of celiac disease or are the result of it. Future research should explore the role of the disease-specific species identified here,” the researchers wrote in Gastroenterology.
Celiac disease is multifactorial. While up to 40% of people are genetically predisposed, only a small proportion develop it, suggesting that environmental factors are key to pathogenesis. Recent studies have linked celiac disease with alterations in the gut microbiome, but it is unclear whether dysbiosis is pathogenic or a secondary effect of disease processes such as nutrient malabsorption, or whether dysbiosis is present at disease onset or results from a gluten-free diet.
For the study, the researchers performed gas chromatography and 16S ribosomal RNA sequencing of fecal samples from 141 children, including 20 with newly biopsy-confirmed, previously untreated celiac disease, 45 who were previously diagnosed and on a gluten-free diet, 19 unaffected siblings, and 57 healthy children who were not on regular medications and had no history of chronic gastrointestinal symptoms. A single fecal sample was tested for all but the previously untreated children, who were tested at baseline and then after 6 and 12 months on a gluten-free diet.
Children with new-onset celiac disease showed no evidence of dysbiosis, while a gluten-free diet explained up to 2.8% of variation in microbiota between patients and controls. Microbial alpha diversity, a measure of species-level diversity, was generally similar among groups, but between 3% and 5% of all taxa differed. Irrespective of treatment, the decreased abundance of the 11 OTUs was diagnostic for celiac disease with an error rate of 21.5% (P < .001 vs. random classification). Notably, most of these 11 discrepant OTUs were associated with nutrient or food group intake and with biomarkers of gluten ingestion, the researchers said. Gas chromatography showed that, after patients started a gluten-free diet, fecal levels of butyrate and ammonia decreased.
“Even though we identified differences in the abundance of a few species between patients with untreated celiac disease and healthy controls, the profound microbial dysbiosis noted in Crohn’s disease was not observed, at least using crude diversity indices,” the investigators commented. “Although several alterations in the intestinal microbiota of children with established celiac disease appear to be effects of a gluten-free diet, there are specific bacteria that are distinct biomarkers of celiac disease.”
Future research might involve performing in vitro tests of “candidate” bacteria, coculturing these bacteria with human immune cells, and studying whether dietary interventions alter the relative abundance of these bacteria in the gut microbiome, the researchers said.
Nutricia Research Foundation, the Biotechnology and Biological Sciences Research Council, and The Catherine McEwan Foundation provided funding. Three coinvestigators disclosed ties to Nutricia, 4D Pharma, AbbVie, Celltrion, Janssen, Takeda, and several other pharmaceutical companies. One coinvestigator reported chairing the working group for ISLI Europe. The remaining investigators reported having no conflicts of interest.
SOURCE: Zafeiropoulou K et al. Gastroenterology. 2020 Aug 10;S0016-5085(20)35023-X. doi: 10.1053/j.gastro.2020.08.007.
Eleven operational taxonomic units (OTUs) of fecal bacteria were less abundant in children with celiac disease than in healthy children, according to the findings of a study published in Gastroenterology.
This microbial signature correctly identified approximately four out of five cases of celiac disease, regardless of whether children were newly diagnosed or had already modified their diet, reported Konstantina Zafeiropoulou and Ben Nichols, PhD, of the Glasgow Royal Infirmary. “It is not clear whether the microbes identified [in this study] contribute to the pathogenesis of celiac disease or are the result of it. Future research should explore the role of the disease-specific species identified here,” the researchers wrote in Gastroenterology.
Celiac disease is multifactorial. While up to 40% of people are genetically predisposed, only a small proportion develop it, suggesting that environmental factors are key to pathogenesis. Recent studies have linked celiac disease with alterations in the gut microbiome, but it is unclear whether dysbiosis is pathogenic or a secondary effect of disease processes such as nutrient malabsorption, or whether dysbiosis is present at disease onset or results from a gluten-free diet.
For the study, the researchers performed gas chromatography and 16S ribosomal RNA sequencing of fecal samples from 141 children, including 20 with newly biopsy-confirmed, previously untreated celiac disease, 45 who were previously diagnosed and on a gluten-free diet, 19 unaffected siblings, and 57 healthy children who were not on regular medications and had no history of chronic gastrointestinal symptoms. A single fecal sample was tested for all but the previously untreated children, who were tested at baseline and then after 6 and 12 months on a gluten-free diet.
Children with new-onset celiac disease showed no evidence of dysbiosis, while a gluten-free diet explained up to 2.8% of variation in microbiota between patients and controls. Microbial alpha diversity, a measure of species-level diversity, was generally similar among groups, but between 3% and 5% of all taxa differed. Irrespective of treatment, the decreased abundance of the 11 OTUs was diagnostic for celiac disease with an error rate of 21.5% (P < .001 vs. random classification). Notably, most of these 11 discrepant OTUs were associated with nutrient or food group intake and with biomarkers of gluten ingestion, the researchers said. Gas chromatography showed that, after patients started a gluten-free diet, fecal levels of butyrate and ammonia decreased.
“Even though we identified differences in the abundance of a few species between patients with untreated celiac disease and healthy controls, the profound microbial dysbiosis noted in Crohn’s disease was not observed, at least using crude diversity indices,” the investigators commented. “Although several alterations in the intestinal microbiota of children with established celiac disease appear to be effects of a gluten-free diet, there are specific bacteria that are distinct biomarkers of celiac disease.”
Future research might involve performing in vitro tests of “candidate” bacteria, coculturing these bacteria with human immune cells, and studying whether dietary interventions alter the relative abundance of these bacteria in the gut microbiome, the researchers said.
Nutricia Research Foundation, the Biotechnology and Biological Sciences Research Council, and The Catherine McEwan Foundation provided funding. Three coinvestigators disclosed ties to Nutricia, 4D Pharma, AbbVie, Celltrion, Janssen, Takeda, and several other pharmaceutical companies. One coinvestigator reported chairing the working group for ISLI Europe. The remaining investigators reported having no conflicts of interest.
SOURCE: Zafeiropoulou K et al. Gastroenterology. 2020 Aug 10;S0016-5085(20)35023-X. doi: 10.1053/j.gastro.2020.08.007.
FROM GASTROENTEROLOGY
Key clinical point: A novel microbial signature distinguished children with celiac disease from healthy controls.
Major finding: Eleven operational taxonomic units (OTUs) were less abundant in fecal samples from children with treated and untreated celiac disease than in healthy controls. The microbial signature was diagnostic for celiac disease with an error rate of 21.5% (P < .001 compared with random classification).
Study details: Gas chromatography and 16S ribosomal RNA sequencing of fecal samples from 141 children: 20 with new-onset celiac disease, 45 with an established diagnosis who were on a gluten-free diet, 19 unaffected siblings, and 57 healthy children. Also, a prospective study of fecal samples from 13 newly diagnosed children after 6 and 12 months on a gluten-free diet.
Disclosures: Nutricia Research Foundation, the Biotechnology and Biological Sciences Research Council, and The Catherine McEwan Foundation provided funding. Three coinvestigators disclosed ties to Nutricia, 4D Pharma, Abbvie, Janssen, Takeda, Celltrion, and several other pharmaceutical companies. One coinvestigator reported chairing the working group for ISLI Europe. The remaining investigators reported having no conflicts of interest.
Source: Zafeiropoulou K et al. Gastroenterology. 2020 Aug 10;S0016-5085(20)35023-X. doi: 10.1053/j.gastro.2020.08.007.
Great Barrington coauthor backs off strict reliance on herd immunity
A coauthor of the Great Barrington Declaration says that he and colleagues have never argued against using mitigation strategies to keep COVID-19 from spreading, and that critics have mischaracterized the document as a “let it rip” strategy.
Jay Bhattacharya, MD, PhD, a professor and public health policy expert in infectious diseases at Stanford University in California, spoke on a JAMA Livestream debate on November 6. Marc Lipsitch, MD, an epidemiology professor at the Harvard T.H. Chan School of Public Health in Boston, Massachusetts, represented the 6900 signatories of the John Snow Memorandum, a rebuttal to the Great Barrington document.
The Great Barrington approach of “Focused Protection” advocates isolation and protection of people who are most vulnerable to COVID-19 while avoiding what they characterize as lockdowns. “The most compassionate approach that balances the risks and benefits of reaching herd immunity, is to allow those who are at minimal risk of death to live their lives normally to build up immunity to the virus through natural infection, while better protecting those who are at highest risk,” the document reads.
The Infectious Diseases Society of America (IDSA) and its HIV Medicine Association denounced the declaration, as reported by Medscape Medical News, and the World Health Organization (WHO) Director General Tedros Adhanom Ghebreyesus called the proposal “unethical.” But the idea has gained some traction at the White House, where Coronavirus Task Force Member and Stanford professor Scott Atlas, MD, has been advising President Donald J. Trump.
On the JAMA debate, Bhattacharya said, “I think all of the mitigation measures are really important,” listing social distancing, hand washing, and masks when distancing is not possible as chief among those strategies for the less vulnerable. “I don’t want to create infections intentionally, but I want us to allow people to go back to their lives as best they can, understanding of the risks they are taking when they do it,” he said, claiming that 99.95% of the population will survive infection.
“The harmful lockdowns are worse for many, many people,” Bhattacharya said.
“I think Jay is moving towards a middle ground which is not really what the Great Barrington Declaration seems to promote,” countered Lipsitch. The declaration does not say use masks or social distance, he said. “It just says we need to go back to a normal life.”
Bhattacharya’s statements to JAMA mean that “maybe we are approaching some common ground,” Lipsitch said.
Definition of a lockdown
Both men were asked to give their definition of a “lockdown.” To Lipsitch, it means people are not allowed out except for essential services and that most businesses are closed, with exceptions for those deemed essential.
Bhattacharya, however, said he views that as a quarantine. Lockdowns “are what we’re currently doing,” he said. Schools, churches, businesses, and arts and culture organizations are shuttered, and “almost every aspect of society is restricted in some way,” Bhattacharya said.
He blamed these lockdowns for most of the excess deaths over and above the COVID-19 deaths and said they had failed to control the pandemic.
Lipsitch said that “it feels to me that Jay is describing as lockdown everything that causes harm, even when it’s not locked down.” He noted that the country was truly closed down for 2 months or so in the spring.
“All of these harms I agree are real,” said Lipsitch. “But they are because the normal life of our society is being interfered with by viral transmission and by people’s inability to live their normal lives.”
Closures and lockdowns are essential to delaying cases and deaths, said Lipsitch. “A case today is worse than a case tomorrow and a lot worse than a case 6 months from now,” he said, noting that a vaccine or improved therapeutics could evolve.
“Delay is not nothing,” Lipsitch added. “It’s actually the goal as I see it, and as the John Snow memo says, we want to keep the virus under control in such a way as that the vulnerable people are not at risk.”
He predicted that cases will continue to grow exponentially because the nation is “not even close to herd immunity.” And, if intensive care units fill up, “there will be a responsive lockdown,” he said, adding that he did not endorse that as a general matter or favor it as a default position.
Bhattacharya claimed that Sweden has tallied only 1800 excess deaths since the pandemic began. “That’s lockdown harm avoided,” he said, advocating a similar strategy for the United States. But, infections have been on the rise in Sweden, and the nation has a higher COVID-19 death rate — with 6000 deaths — than other Nordic countries.
“If we keep this policy of lockdown we will have the same kind of outcomes we’ve already had — high excess deaths and sort of indifferent control of COVID,” Bhattacharya said.
“We’re still going to have misery and death going forward until we reach a point where there’s sufficient immunity either though a vaccine or through natural infection,” he said.
This article first appeared on Medscape.com.
A coauthor of the Great Barrington Declaration says that he and colleagues have never argued against using mitigation strategies to keep COVID-19 from spreading, and that critics have mischaracterized the document as a “let it rip” strategy.
Jay Bhattacharya, MD, PhD, a professor and public health policy expert in infectious diseases at Stanford University in California, spoke on a JAMA Livestream debate on November 6. Marc Lipsitch, MD, an epidemiology professor at the Harvard T.H. Chan School of Public Health in Boston, Massachusetts, represented the 6900 signatories of the John Snow Memorandum, a rebuttal to the Great Barrington document.
The Great Barrington approach of “Focused Protection” advocates isolation and protection of people who are most vulnerable to COVID-19 while avoiding what they characterize as lockdowns. “The most compassionate approach that balances the risks and benefits of reaching herd immunity, is to allow those who are at minimal risk of death to live their lives normally to build up immunity to the virus through natural infection, while better protecting those who are at highest risk,” the document reads.
The Infectious Diseases Society of America (IDSA) and its HIV Medicine Association denounced the declaration, as reported by Medscape Medical News, and the World Health Organization (WHO) Director General Tedros Adhanom Ghebreyesus called the proposal “unethical.” But the idea has gained some traction at the White House, where Coronavirus Task Force Member and Stanford professor Scott Atlas, MD, has been advising President Donald J. Trump.
On the JAMA debate, Bhattacharya said, “I think all of the mitigation measures are really important,” listing social distancing, hand washing, and masks when distancing is not possible as chief among those strategies for the less vulnerable. “I don’t want to create infections intentionally, but I want us to allow people to go back to their lives as best they can, understanding of the risks they are taking when they do it,” he said, claiming that 99.95% of the population will survive infection.
“The harmful lockdowns are worse for many, many people,” Bhattacharya said.
“I think Jay is moving towards a middle ground which is not really what the Great Barrington Declaration seems to promote,” countered Lipsitch. The declaration does not say use masks or social distance, he said. “It just says we need to go back to a normal life.”
Bhattacharya’s statements to JAMA mean that “maybe we are approaching some common ground,” Lipsitch said.
Definition of a lockdown
Both men were asked to give their definition of a “lockdown.” To Lipsitch, it means people are not allowed out except for essential services and that most businesses are closed, with exceptions for those deemed essential.
Bhattacharya, however, said he views that as a quarantine. Lockdowns “are what we’re currently doing,” he said. Schools, churches, businesses, and arts and culture organizations are shuttered, and “almost every aspect of society is restricted in some way,” Bhattacharya said.
He blamed these lockdowns for most of the excess deaths over and above the COVID-19 deaths and said they had failed to control the pandemic.
Lipsitch said that “it feels to me that Jay is describing as lockdown everything that causes harm, even when it’s not locked down.” He noted that the country was truly closed down for 2 months or so in the spring.
“All of these harms I agree are real,” said Lipsitch. “But they are because the normal life of our society is being interfered with by viral transmission and by people’s inability to live their normal lives.”
Closures and lockdowns are essential to delaying cases and deaths, said Lipsitch. “A case today is worse than a case tomorrow and a lot worse than a case 6 months from now,” he said, noting that a vaccine or improved therapeutics could evolve.
“Delay is not nothing,” Lipsitch added. “It’s actually the goal as I see it, and as the John Snow memo says, we want to keep the virus under control in such a way as that the vulnerable people are not at risk.”
He predicted that cases will continue to grow exponentially because the nation is “not even close to herd immunity.” And, if intensive care units fill up, “there will be a responsive lockdown,” he said, adding that he did not endorse that as a general matter or favor it as a default position.
Bhattacharya claimed that Sweden has tallied only 1800 excess deaths since the pandemic began. “That’s lockdown harm avoided,” he said, advocating a similar strategy for the United States. But, infections have been on the rise in Sweden, and the nation has a higher COVID-19 death rate — with 6000 deaths — than other Nordic countries.
“If we keep this policy of lockdown we will have the same kind of outcomes we’ve already had — high excess deaths and sort of indifferent control of COVID,” Bhattacharya said.
“We’re still going to have misery and death going forward until we reach a point where there’s sufficient immunity either though a vaccine or through natural infection,” he said.
This article first appeared on Medscape.com.
A coauthor of the Great Barrington Declaration says that he and colleagues have never argued against using mitigation strategies to keep COVID-19 from spreading, and that critics have mischaracterized the document as a “let it rip” strategy.
Jay Bhattacharya, MD, PhD, a professor and public health policy expert in infectious diseases at Stanford University in California, spoke on a JAMA Livestream debate on November 6. Marc Lipsitch, MD, an epidemiology professor at the Harvard T.H. Chan School of Public Health in Boston, Massachusetts, represented the 6900 signatories of the John Snow Memorandum, a rebuttal to the Great Barrington document.
The Great Barrington approach of “Focused Protection” advocates isolation and protection of people who are most vulnerable to COVID-19 while avoiding what they characterize as lockdowns. “The most compassionate approach that balances the risks and benefits of reaching herd immunity, is to allow those who are at minimal risk of death to live their lives normally to build up immunity to the virus through natural infection, while better protecting those who are at highest risk,” the document reads.
The Infectious Diseases Society of America (IDSA) and its HIV Medicine Association denounced the declaration, as reported by Medscape Medical News, and the World Health Organization (WHO) Director General Tedros Adhanom Ghebreyesus called the proposal “unethical.” But the idea has gained some traction at the White House, where Coronavirus Task Force Member and Stanford professor Scott Atlas, MD, has been advising President Donald J. Trump.
On the JAMA debate, Bhattacharya said, “I think all of the mitigation measures are really important,” listing social distancing, hand washing, and masks when distancing is not possible as chief among those strategies for the less vulnerable. “I don’t want to create infections intentionally, but I want us to allow people to go back to their lives as best they can, understanding of the risks they are taking when they do it,” he said, claiming that 99.95% of the population will survive infection.
“The harmful lockdowns are worse for many, many people,” Bhattacharya said.
“I think Jay is moving towards a middle ground which is not really what the Great Barrington Declaration seems to promote,” countered Lipsitch. The declaration does not say use masks or social distance, he said. “It just says we need to go back to a normal life.”
Bhattacharya’s statements to JAMA mean that “maybe we are approaching some common ground,” Lipsitch said.
Definition of a lockdown
Both men were asked to give their definition of a “lockdown.” To Lipsitch, it means people are not allowed out except for essential services and that most businesses are closed, with exceptions for those deemed essential.
Bhattacharya, however, said he views that as a quarantine. Lockdowns “are what we’re currently doing,” he said. Schools, churches, businesses, and arts and culture organizations are shuttered, and “almost every aspect of society is restricted in some way,” Bhattacharya said.
He blamed these lockdowns for most of the excess deaths over and above the COVID-19 deaths and said they had failed to control the pandemic.
Lipsitch said that “it feels to me that Jay is describing as lockdown everything that causes harm, even when it’s not locked down.” He noted that the country was truly closed down for 2 months or so in the spring.
“All of these harms I agree are real,” said Lipsitch. “But they are because the normal life of our society is being interfered with by viral transmission and by people’s inability to live their normal lives.”
Closures and lockdowns are essential to delaying cases and deaths, said Lipsitch. “A case today is worse than a case tomorrow and a lot worse than a case 6 months from now,” he said, noting that a vaccine or improved therapeutics could evolve.
“Delay is not nothing,” Lipsitch added. “It’s actually the goal as I see it, and as the John Snow memo says, we want to keep the virus under control in such a way as that the vulnerable people are not at risk.”
He predicted that cases will continue to grow exponentially because the nation is “not even close to herd immunity.” And, if intensive care units fill up, “there will be a responsive lockdown,” he said, adding that he did not endorse that as a general matter or favor it as a default position.
Bhattacharya claimed that Sweden has tallied only 1800 excess deaths since the pandemic began. “That’s lockdown harm avoided,” he said, advocating a similar strategy for the United States. But, infections have been on the rise in Sweden, and the nation has a higher COVID-19 death rate — with 6000 deaths — than other Nordic countries.
“If we keep this policy of lockdown we will have the same kind of outcomes we’ve already had — high excess deaths and sort of indifferent control of COVID,” Bhattacharya said.
“We’re still going to have misery and death going forward until we reach a point where there’s sufficient immunity either though a vaccine or through natural infection,” he said.
This article first appeared on Medscape.com.
Hospitals poised to launch first COVID-19 vaccines in clinicians
At first, when news spread of a 28-year-old doctor on the COVID-19 front lines in Brazil who died after receiving an experimental vaccine, doubts arose about the safety of one of the most promising coronavirus vaccine candidates. But then the story flipped. Although the vaccine maker wouldn’t confirm it, the doctor appeared to have been in the control group and had received a dose of an established meningitis vaccine. The danger came from exposure to the coronavirus itself.
That tragedy underscores the ongoing risk of COVID-19 to healthcare workers, who have been designated by US advisory panels as part of phase 1A – the first to receive doses of any approved vaccine. The Centers for Disease Control and Prevention (CDC) recently reported that 6% of adults hospitalized with COVID from March to May were healthcare workers. The report was based on surveillance data from 13 states. The average age of the patients was 49 years. The agency set a November 15 vaccination “readiness date” for jurisdictions, such as state health departments, even though a vaccine isn’t likely to be authorized by then.
As hospitals scramble to prepare, their watchword is flexibility. They don’t yet know how many initial doses they will get, of which vaccine, or in what time frame. They have a sophisticated infrastructure to deliver flu vaccines each fall, but that framework doesn’t align with the likely scenarios of limited supply, additional reporting requirements, two-dose regimens, and differing storage needs.
“Healthcare organizations have consistently risen to the challenge. I wholeheartedly believe in their potential to do this,” Anna Legreid Dopp, PharmD, senior director of quality improvement and guidelines for the American Society of Health-System Pharmacists, told Medscape Medical News.
Healthcare workers won’t face a vaccine mandate
Even after months of caring for COVID patients, most clinicians remain vulnerable to infection – at work and in their communities. That was what occupational medicine physician Kevin Smith, MD, realized when his health system, Toledo, Ohio–based ProMedica, offered antibody testing to all its 50,000 employees. About 2% of the 6933 tests given came back positive, he says.
Yet many physicians, nurses, and other healthcare workers share the public’s skepticism about the safety and effectiveness of a vaccine that receives swift US Food and Drug Administration (FDA) approval for emergency use. About half of nurses (47%) and almost 1 in 3 physicians (30%) say that they don’t want to get the vaccine when it first becomes available or that they’re unsure about vaccination, according to a Medscape survey.
Because vaccination of healthcare workers will set the stage for public acceptance of the vaccine, hospital epidemiologists are concerned. “We know that there will be some hesitancy in the healthcare workforce, just as there will be in the broader public,” said Marci Drees, MD, chief infection prevention officer and hospital epidemiologist for ChristianaCare in Newark, Delaware, and liaison from the Society for Healthcare Epidemiology of America to the CDC’s Advisory Committee on Immunization Practices.* “I do not think we can expect anyone to be vaccinated if we’re not willing to vaccinate ourselves.”
Healthcare workers are typically required to receive a range of vaccines, including measles, mumps, and rubella (MMR) and pertussis shots. Each year, close to half of US healthcare workers receive a flu vaccine under a workplace mandate. But COVID-19 will be different. The FDA requires anyone given products under an emergency use authorization (EUA) to receive information about risks and benefits and to have the option to decline. Hospitals instead will rely on education as they offer a novel vaccine (or more than one) that will have a minimum effectiveness of 50%.
ProMedica doesn’t require employees to be vaccinated against flu, but employees who decline must get a note from a doctor indicating that they have talked about the risks and benefits of the vaccine. A similar approach may be used with a COVID-19 vaccine, in which employees may be required to learn about the vaccine before they decline, Smith says. “I do believe some people will say they don’t want to get it,” he added.
Like colleagues across the country, Smith is identifying healthcare workers who are involved in direct care of COVID-19 patients and are at highest risk for exposure. Even within the top tier, those performing the riskiest tasks, such as respiratory therapists who provide breathing treatments that spread aerosols and droplets, will be tagged as a priority group, he says. Healthcare workers who spend the most time in proximity to COVID patients, such as nurses in a COVID unit, also are likely to get the first doses, he says.
Swirl, don’t shake, the vaccine
Hospitals are adept at ramping up vaccination campaigns. For example, last year, Vanderbilt University Medical Center, in Nashville, Tennessee, vaccinated nearly 16,000 employees against influenza in their 1-day “Flulapalooza” event. The medical center even earned a Guinness world record in 2011 at the first Flulapalooza for giving the most vaccinations ever within 8 hours.
The 10th anniversary of the event was canceled this year because of COVID restrictions. Instead, nurses, pharmacists, and other clinicians pitched in to vaccinate their coworkers against influenza. Now, plans for COVID-19 vaccination move forward amid uncertainty.
Instead of holding a mass event, “the delivery mechanisms will need to be more targeted and focused,” said Lori Rolando, MD, MPH, director of the Vanderbilt Occupational Health Clinic. In the CDC’s most recent version of its vaccination program “playbook,” the agency recommends giving the vaccines in an area that allows people to remain 6 feet apart and for them to wait for 15 minutes after receiving the shot to make sure they don’t faint, a potential risk common to almost all vaccines.
That’s the easy part. Planning becomes more complex, given the uncertainty as to which vaccines will receive approval and which one a hospital will receive.
If the Pfizer/BioNTech vaccine receives EUA in 2020, about 10 to 20 million doses could be available in November and 20 to 30 million doses in December. The ultracold containers used to ship the vaccines have to be replenished with dry ice within 24 hours of receipt and every 5 days thereafter. Hospitals will need temperature probes to monitor storage in the containers. The five-dose vials can be refrigerated before administering, but only for 5 days. The product must be diluted, and it then must be used within 6 hours.
The Moderna vaccine will be somewhat less plentiful at first. About 10 million doses are expected in November and 15 million doses by the end of December. The 10-dose vials are stored in a freezer. Once they are placed in a refrigerator to thaw, they have to be used within 7 days, and once they’re removed from the refrigerator, they have to be used within 12 hours. The pharmacist or other vaccinator must swirl – but not shake! – the vial before delivering a dose, according to the CDC playbook.
As more information emerges about the vaccines, instructions may change, and Smith is steeled for shifting scenarios. “These are all draft plans. We’re going to modify as we go along,” he says.
The Pfizer vaccine requires a second dose at 21 days, and the Moderna vaccine targets the second dose at 28 days. In addition to using information systems to track vaccinations and any adverse effects, hospitals will give employees a card indicating what vaccine they received, the date it was administered, and the date on which they need to return. (At this point, the time frame for the second dose doesn’t appear to be flexible.)
Regardless of the vaccine, one message stays the same: COVID precautions must continue. That means mask wearing, social distancing, and hand washing – practices that also must be followed by healthcare workers who test positive for naturally acquired antibodies.
“I don’t think anyone expects the COVID vaccine to be 100% effective at preventing COVID,” says Rolando. “So all of the other tools in our toolbox are going to need to be continued to be used as well.”
*Correction, 11/12/20: An earlier version of this article misstated the name of Dr. Drees' institution.
This article first appeared on Medscape.com.
At first, when news spread of a 28-year-old doctor on the COVID-19 front lines in Brazil who died after receiving an experimental vaccine, doubts arose about the safety of one of the most promising coronavirus vaccine candidates. But then the story flipped. Although the vaccine maker wouldn’t confirm it, the doctor appeared to have been in the control group and had received a dose of an established meningitis vaccine. The danger came from exposure to the coronavirus itself.
That tragedy underscores the ongoing risk of COVID-19 to healthcare workers, who have been designated by US advisory panels as part of phase 1A – the first to receive doses of any approved vaccine. The Centers for Disease Control and Prevention (CDC) recently reported that 6% of adults hospitalized with COVID from March to May were healthcare workers. The report was based on surveillance data from 13 states. The average age of the patients was 49 years. The agency set a November 15 vaccination “readiness date” for jurisdictions, such as state health departments, even though a vaccine isn’t likely to be authorized by then.
As hospitals scramble to prepare, their watchword is flexibility. They don’t yet know how many initial doses they will get, of which vaccine, or in what time frame. They have a sophisticated infrastructure to deliver flu vaccines each fall, but that framework doesn’t align with the likely scenarios of limited supply, additional reporting requirements, two-dose regimens, and differing storage needs.
“Healthcare organizations have consistently risen to the challenge. I wholeheartedly believe in their potential to do this,” Anna Legreid Dopp, PharmD, senior director of quality improvement and guidelines for the American Society of Health-System Pharmacists, told Medscape Medical News.
Healthcare workers won’t face a vaccine mandate
Even after months of caring for COVID patients, most clinicians remain vulnerable to infection – at work and in their communities. That was what occupational medicine physician Kevin Smith, MD, realized when his health system, Toledo, Ohio–based ProMedica, offered antibody testing to all its 50,000 employees. About 2% of the 6933 tests given came back positive, he says.
Yet many physicians, nurses, and other healthcare workers share the public’s skepticism about the safety and effectiveness of a vaccine that receives swift US Food and Drug Administration (FDA) approval for emergency use. About half of nurses (47%) and almost 1 in 3 physicians (30%) say that they don’t want to get the vaccine when it first becomes available or that they’re unsure about vaccination, according to a Medscape survey.
Because vaccination of healthcare workers will set the stage for public acceptance of the vaccine, hospital epidemiologists are concerned. “We know that there will be some hesitancy in the healthcare workforce, just as there will be in the broader public,” said Marci Drees, MD, chief infection prevention officer and hospital epidemiologist for ChristianaCare in Newark, Delaware, and liaison from the Society for Healthcare Epidemiology of America to the CDC’s Advisory Committee on Immunization Practices.* “I do not think we can expect anyone to be vaccinated if we’re not willing to vaccinate ourselves.”
Healthcare workers are typically required to receive a range of vaccines, including measles, mumps, and rubella (MMR) and pertussis shots. Each year, close to half of US healthcare workers receive a flu vaccine under a workplace mandate. But COVID-19 will be different. The FDA requires anyone given products under an emergency use authorization (EUA) to receive information about risks and benefits and to have the option to decline. Hospitals instead will rely on education as they offer a novel vaccine (or more than one) that will have a minimum effectiveness of 50%.
ProMedica doesn’t require employees to be vaccinated against flu, but employees who decline must get a note from a doctor indicating that they have talked about the risks and benefits of the vaccine. A similar approach may be used with a COVID-19 vaccine, in which employees may be required to learn about the vaccine before they decline, Smith says. “I do believe some people will say they don’t want to get it,” he added.
Like colleagues across the country, Smith is identifying healthcare workers who are involved in direct care of COVID-19 patients and are at highest risk for exposure. Even within the top tier, those performing the riskiest tasks, such as respiratory therapists who provide breathing treatments that spread aerosols and droplets, will be tagged as a priority group, he says. Healthcare workers who spend the most time in proximity to COVID patients, such as nurses in a COVID unit, also are likely to get the first doses, he says.
Swirl, don’t shake, the vaccine
Hospitals are adept at ramping up vaccination campaigns. For example, last year, Vanderbilt University Medical Center, in Nashville, Tennessee, vaccinated nearly 16,000 employees against influenza in their 1-day “Flulapalooza” event. The medical center even earned a Guinness world record in 2011 at the first Flulapalooza for giving the most vaccinations ever within 8 hours.
The 10th anniversary of the event was canceled this year because of COVID restrictions. Instead, nurses, pharmacists, and other clinicians pitched in to vaccinate their coworkers against influenza. Now, plans for COVID-19 vaccination move forward amid uncertainty.
Instead of holding a mass event, “the delivery mechanisms will need to be more targeted and focused,” said Lori Rolando, MD, MPH, director of the Vanderbilt Occupational Health Clinic. In the CDC’s most recent version of its vaccination program “playbook,” the agency recommends giving the vaccines in an area that allows people to remain 6 feet apart and for them to wait for 15 minutes after receiving the shot to make sure they don’t faint, a potential risk common to almost all vaccines.
That’s the easy part. Planning becomes more complex, given the uncertainty as to which vaccines will receive approval and which one a hospital will receive.
If the Pfizer/BioNTech vaccine receives EUA in 2020, about 10 to 20 million doses could be available in November and 20 to 30 million doses in December. The ultracold containers used to ship the vaccines have to be replenished with dry ice within 24 hours of receipt and every 5 days thereafter. Hospitals will need temperature probes to monitor storage in the containers. The five-dose vials can be refrigerated before administering, but only for 5 days. The product must be diluted, and it then must be used within 6 hours.
The Moderna vaccine will be somewhat less plentiful at first. About 10 million doses are expected in November and 15 million doses by the end of December. The 10-dose vials are stored in a freezer. Once they are placed in a refrigerator to thaw, they have to be used within 7 days, and once they’re removed from the refrigerator, they have to be used within 12 hours. The pharmacist or other vaccinator must swirl – but not shake! – the vial before delivering a dose, according to the CDC playbook.
As more information emerges about the vaccines, instructions may change, and Smith is steeled for shifting scenarios. “These are all draft plans. We’re going to modify as we go along,” he says.
The Pfizer vaccine requires a second dose at 21 days, and the Moderna vaccine targets the second dose at 28 days. In addition to using information systems to track vaccinations and any adverse effects, hospitals will give employees a card indicating what vaccine they received, the date it was administered, and the date on which they need to return. (At this point, the time frame for the second dose doesn’t appear to be flexible.)
Regardless of the vaccine, one message stays the same: COVID precautions must continue. That means mask wearing, social distancing, and hand washing – practices that also must be followed by healthcare workers who test positive for naturally acquired antibodies.
“I don’t think anyone expects the COVID vaccine to be 100% effective at preventing COVID,” says Rolando. “So all of the other tools in our toolbox are going to need to be continued to be used as well.”
*Correction, 11/12/20: An earlier version of this article misstated the name of Dr. Drees' institution.
This article first appeared on Medscape.com.
At first, when news spread of a 28-year-old doctor on the COVID-19 front lines in Brazil who died after receiving an experimental vaccine, doubts arose about the safety of one of the most promising coronavirus vaccine candidates. But then the story flipped. Although the vaccine maker wouldn’t confirm it, the doctor appeared to have been in the control group and had received a dose of an established meningitis vaccine. The danger came from exposure to the coronavirus itself.
That tragedy underscores the ongoing risk of COVID-19 to healthcare workers, who have been designated by US advisory panels as part of phase 1A – the first to receive doses of any approved vaccine. The Centers for Disease Control and Prevention (CDC) recently reported that 6% of adults hospitalized with COVID from March to May were healthcare workers. The report was based on surveillance data from 13 states. The average age of the patients was 49 years. The agency set a November 15 vaccination “readiness date” for jurisdictions, such as state health departments, even though a vaccine isn’t likely to be authorized by then.
As hospitals scramble to prepare, their watchword is flexibility. They don’t yet know how many initial doses they will get, of which vaccine, or in what time frame. They have a sophisticated infrastructure to deliver flu vaccines each fall, but that framework doesn’t align with the likely scenarios of limited supply, additional reporting requirements, two-dose regimens, and differing storage needs.
“Healthcare organizations have consistently risen to the challenge. I wholeheartedly believe in their potential to do this,” Anna Legreid Dopp, PharmD, senior director of quality improvement and guidelines for the American Society of Health-System Pharmacists, told Medscape Medical News.
Healthcare workers won’t face a vaccine mandate
Even after months of caring for COVID patients, most clinicians remain vulnerable to infection – at work and in their communities. That was what occupational medicine physician Kevin Smith, MD, realized when his health system, Toledo, Ohio–based ProMedica, offered antibody testing to all its 50,000 employees. About 2% of the 6933 tests given came back positive, he says.
Yet many physicians, nurses, and other healthcare workers share the public’s skepticism about the safety and effectiveness of a vaccine that receives swift US Food and Drug Administration (FDA) approval for emergency use. About half of nurses (47%) and almost 1 in 3 physicians (30%) say that they don’t want to get the vaccine when it first becomes available or that they’re unsure about vaccination, according to a Medscape survey.
Because vaccination of healthcare workers will set the stage for public acceptance of the vaccine, hospital epidemiologists are concerned. “We know that there will be some hesitancy in the healthcare workforce, just as there will be in the broader public,” said Marci Drees, MD, chief infection prevention officer and hospital epidemiologist for ChristianaCare in Newark, Delaware, and liaison from the Society for Healthcare Epidemiology of America to the CDC’s Advisory Committee on Immunization Practices.* “I do not think we can expect anyone to be vaccinated if we’re not willing to vaccinate ourselves.”
Healthcare workers are typically required to receive a range of vaccines, including measles, mumps, and rubella (MMR) and pertussis shots. Each year, close to half of US healthcare workers receive a flu vaccine under a workplace mandate. But COVID-19 will be different. The FDA requires anyone given products under an emergency use authorization (EUA) to receive information about risks and benefits and to have the option to decline. Hospitals instead will rely on education as they offer a novel vaccine (or more than one) that will have a minimum effectiveness of 50%.
ProMedica doesn’t require employees to be vaccinated against flu, but employees who decline must get a note from a doctor indicating that they have talked about the risks and benefits of the vaccine. A similar approach may be used with a COVID-19 vaccine, in which employees may be required to learn about the vaccine before they decline, Smith says. “I do believe some people will say they don’t want to get it,” he added.
Like colleagues across the country, Smith is identifying healthcare workers who are involved in direct care of COVID-19 patients and are at highest risk for exposure. Even within the top tier, those performing the riskiest tasks, such as respiratory therapists who provide breathing treatments that spread aerosols and droplets, will be tagged as a priority group, he says. Healthcare workers who spend the most time in proximity to COVID patients, such as nurses in a COVID unit, also are likely to get the first doses, he says.
Swirl, don’t shake, the vaccine
Hospitals are adept at ramping up vaccination campaigns. For example, last year, Vanderbilt University Medical Center, in Nashville, Tennessee, vaccinated nearly 16,000 employees against influenza in their 1-day “Flulapalooza” event. The medical center even earned a Guinness world record in 2011 at the first Flulapalooza for giving the most vaccinations ever within 8 hours.
The 10th anniversary of the event was canceled this year because of COVID restrictions. Instead, nurses, pharmacists, and other clinicians pitched in to vaccinate their coworkers against influenza. Now, plans for COVID-19 vaccination move forward amid uncertainty.
Instead of holding a mass event, “the delivery mechanisms will need to be more targeted and focused,” said Lori Rolando, MD, MPH, director of the Vanderbilt Occupational Health Clinic. In the CDC’s most recent version of its vaccination program “playbook,” the agency recommends giving the vaccines in an area that allows people to remain 6 feet apart and for them to wait for 15 minutes after receiving the shot to make sure they don’t faint, a potential risk common to almost all vaccines.
That’s the easy part. Planning becomes more complex, given the uncertainty as to which vaccines will receive approval and which one a hospital will receive.
If the Pfizer/BioNTech vaccine receives EUA in 2020, about 10 to 20 million doses could be available in November and 20 to 30 million doses in December. The ultracold containers used to ship the vaccines have to be replenished with dry ice within 24 hours of receipt and every 5 days thereafter. Hospitals will need temperature probes to monitor storage in the containers. The five-dose vials can be refrigerated before administering, but only for 5 days. The product must be diluted, and it then must be used within 6 hours.
The Moderna vaccine will be somewhat less plentiful at first. About 10 million doses are expected in November and 15 million doses by the end of December. The 10-dose vials are stored in a freezer. Once they are placed in a refrigerator to thaw, they have to be used within 7 days, and once they’re removed from the refrigerator, they have to be used within 12 hours. The pharmacist or other vaccinator must swirl – but not shake! – the vial before delivering a dose, according to the CDC playbook.
As more information emerges about the vaccines, instructions may change, and Smith is steeled for shifting scenarios. “These are all draft plans. We’re going to modify as we go along,” he says.
The Pfizer vaccine requires a second dose at 21 days, and the Moderna vaccine targets the second dose at 28 days. In addition to using information systems to track vaccinations and any adverse effects, hospitals will give employees a card indicating what vaccine they received, the date it was administered, and the date on which they need to return. (At this point, the time frame for the second dose doesn’t appear to be flexible.)
Regardless of the vaccine, one message stays the same: COVID precautions must continue. That means mask wearing, social distancing, and hand washing – practices that also must be followed by healthcare workers who test positive for naturally acquired antibodies.
“I don’t think anyone expects the COVID vaccine to be 100% effective at preventing COVID,” says Rolando. “So all of the other tools in our toolbox are going to need to be continued to be used as well.”
*Correction, 11/12/20: An earlier version of this article misstated the name of Dr. Drees' institution.
This article first appeared on Medscape.com.
United States adds nearly 74,000 more children with COVID-19
The new weekly high for COVID-19 cases in children announced last week has been surpassed already, as the United States experienced almost 74,000 new pediatric cases for the week ending Nov. 5, according to the American Academy of Pediatrics and the Children’s Hospital Association.
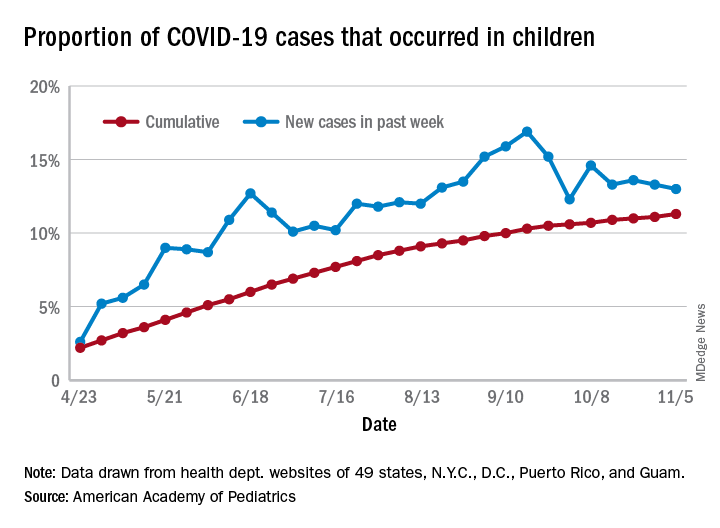
The total number of COVID-19 cases in children is now 927,518 in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA said in their weekly report.
Cumulatively, children represent 11.3% of all COVID-19 cases in those jurisdictions, up from 11.1% a week ago. For just the past week, those 73,883 children represent 13.0% of the 567,672 new cases reported among all ages. That proportion peaked at 16.9% in mid-September, the AAP/CHA data show.
Dropping down to the state level, cumulative proportions as of Nov. 5 range from 5.2% in New Jersey to 23.3% in Wyoming, with 11 other states over 15%. California has had more cases, 100,856, than any other state, and Vermont the fewest at 329, the AAP and CHA said.
The national rate per 100,000 children is now 1,232, up from 1,134 the previous week and more than doubled since mid-August (582.2 per 100,000 on Aug. 20). North Dakota’s rate of 3,990 per 100,000 children is the highest of any state (South Dakota is next at 2,779), while Vermont is again the lowest at 245 per 100,000, based on data collected from state health department websites.
Two COVID-19–related deaths in children were reported during the week ending Nov. 5, bringing the total to 123 but leaving the overall proportion of deaths in children unchanged at 0.06% of all deaths. Texas has reported the most COVID-19 deaths in children with 29, while 15 states have recorded no deaths so far (mortality data in children reported by 42 states and New York City), the AAP and CHA said.
The new weekly high for COVID-19 cases in children announced last week has been surpassed already, as the United States experienced almost 74,000 new pediatric cases for the week ending Nov. 5, according to the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of COVID-19 cases in children is now 927,518 in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA said in their weekly report.
Cumulatively, children represent 11.3% of all COVID-19 cases in those jurisdictions, up from 11.1% a week ago. For just the past week, those 73,883 children represent 13.0% of the 567,672 new cases reported among all ages. That proportion peaked at 16.9% in mid-September, the AAP/CHA data show.
Dropping down to the state level, cumulative proportions as of Nov. 5 range from 5.2% in New Jersey to 23.3% in Wyoming, with 11 other states over 15%. California has had more cases, 100,856, than any other state, and Vermont the fewest at 329, the AAP and CHA said.
The national rate per 100,000 children is now 1,232, up from 1,134 the previous week and more than doubled since mid-August (582.2 per 100,000 on Aug. 20). North Dakota’s rate of 3,990 per 100,000 children is the highest of any state (South Dakota is next at 2,779), while Vermont is again the lowest at 245 per 100,000, based on data collected from state health department websites.
Two COVID-19–related deaths in children were reported during the week ending Nov. 5, bringing the total to 123 but leaving the overall proportion of deaths in children unchanged at 0.06% of all deaths. Texas has reported the most COVID-19 deaths in children with 29, while 15 states have recorded no deaths so far (mortality data in children reported by 42 states and New York City), the AAP and CHA said.
The new weekly high for COVID-19 cases in children announced last week has been surpassed already, as the United States experienced almost 74,000 new pediatric cases for the week ending Nov. 5, according to the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of COVID-19 cases in children is now 927,518 in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA said in their weekly report.
Cumulatively, children represent 11.3% of all COVID-19 cases in those jurisdictions, up from 11.1% a week ago. For just the past week, those 73,883 children represent 13.0% of the 567,672 new cases reported among all ages. That proportion peaked at 16.9% in mid-September, the AAP/CHA data show.
Dropping down to the state level, cumulative proportions as of Nov. 5 range from 5.2% in New Jersey to 23.3% in Wyoming, with 11 other states over 15%. California has had more cases, 100,856, than any other state, and Vermont the fewest at 329, the AAP and CHA said.
The national rate per 100,000 children is now 1,232, up from 1,134 the previous week and more than doubled since mid-August (582.2 per 100,000 on Aug. 20). North Dakota’s rate of 3,990 per 100,000 children is the highest of any state (South Dakota is next at 2,779), while Vermont is again the lowest at 245 per 100,000, based on data collected from state health department websites.
Two COVID-19–related deaths in children were reported during the week ending Nov. 5, bringing the total to 123 but leaving the overall proportion of deaths in children unchanged at 0.06% of all deaths. Texas has reported the most COVID-19 deaths in children with 29, while 15 states have recorded no deaths so far (mortality data in children reported by 42 states and New York City), the AAP and CHA said.
Food insecurity called urgent issue you must address
and advocate on behalf of those experiencing or at risk of food insecurity, according to Kofi Essel, MD, MPH, a pediatrician at Children’s National Hospital in Washington.
More than one in four adults are dealing with food access hardships during the pandemic, Dr. Essel said at the virtual annual meeting of the American Academy of Pediatrics. Food insecurity is often interchangeable with hunger and refers to limited or uncertain availability of foods that are nutritious and safe.
“Food insecurity is as much about the threat of deprivation as it is about deprivation itself: A food-insecure life means a life lived in fear of hunger, and the psychological toll that takes,” according to a 2020 New York Times photo feature on food insecurity by Brenda Ann Kenneally that Dr. Essel quoted.
The lived experience of food insecure households includes food anxiety, a preoccupation with being able to get enough food that takes up cognitive bandwidth and prevents people from being able to focus on other important things. Another feature of food-insecure homes is a monotony of diet, which often involves an increase in caloric density and decrease in nutritional quality. As food insecurity grows more dire, adults’ food intake decreases, and then children’s intake decreases as adults seek out any way to get food, including “socially unacceptable” ways, which can include food pantries and bartering for food.
Food insecurity is associated with a wide range of negative outcomes even after accounting for other confounders, including decreased overall health, mental health, and educational outcomes. It’s also associated with an increase in developmental delays, hospitalizations, iron deficiency, asthma, and birth defects, among other problems. Somewhat paradoxically, it’s associated with both an increase and a decrease in obesity in the research.
Megan J. Gray, MD, MPH, assistant professor of pediatrics and population health at Dell Medical School at the The University of Texas at Austin, attended Dr. Essel’s session because food insecurity during COVID-19 now affects about half her patients, according to screening research she’s conducted.
“I wanted to learn more about the nuances of screening and using language and talking points that are helpful with families and with staff in building a culture of discussing food insecurity in our clinics,” Dr. Gray said in an interview. “What I’ve learned in my clinic is that if we don’t ask about it, families aren’t telling us – food insecurity is hiding in plain sight.”
She particularly appreciated Dr. Essel’s slides on the progression of food insecurity and how they acknowledged the mental health burden of food insecurity among parents.
“Right now during COVID-19, I see more patients I would call ‘socially complex’ rather than ‘medically complex,’ ” she said. “We all need to get a crash course in social work and Dr. Essel’s presentation is a great starting place.”
Screening for food insecurity
Beginning in 2015, an AAP policy statement charged pediatricians to “screen and intervene” with regard to food insecurity and their patients, Dr. Essel said. The statement also called for pediatricians to advocate for programs and policies that end childhood food insecurity.
The policy statement recommended a validated two-question screening tool called the Hunger Vital Sign:
1. “Within the past 12 months, we worried whether our food would run out before we got money to buy more.”
2. “Within the past 12 months, the food that we bought just didn’t last and we didn’t have money to get more.”
But in screening, you need to be conscious of how dignity intersects with food insecurity concerns, Dr. Essel said.
“We need to create dignity for our families,” he said. “We need to create a safe environment for our families and use appropriate tools when necessary to be able to identify families that are struggling with food insecurity.”
That need is seen in research on food screening. The Hunger Vital Signs questions can be asked with a dichotomous variable, as a yes/no question, or on a Likert scale, though the latter is a more complex way to ask.
A 2017 study found, however, that asking with “yes/no” answers missed more than a quarter of at-risk families. In the AAP survey using “yes/no” answers, 31% of families screened positive for being at risk of food insecurity, compared with 46% when the same question was asked on a Likert scale. It seems the ability to answer with “sometimes” feels “safer” than answering “yes,” Dr. Essel said.
Another factor that potentially affects answers is how doctors ask. In a March 2020 study at a single primary care practice, 16% of families screened positive with yes/no responses to a food insecurity screen when the questions were written, compared with 10% of positive screens with verbal responses (P < .001).
Epidemiology of food insecurity
The most updated United States Department of Agriculture report on food insecurity released in September shows the United States finally reached prerecession levels in 2019, with 11% of families designated as “food insecure.” But 2019 data cannot show what has occurred since the pandemic.
Further, the numbers are higher in households with children: Fourteen percent, or one in seven households with children, are experiencing food insecurity. Racial and ethnic disparities in food insecurity have remained consistent over the past 2 decades, with about twice as many Black and Hispanic homes experiencing food insecurity as White homes.
More recent research using Census Household Pulse Surveys has found a tremendous increase in food insecurity for children in 2020. One in three Black children and one in four Hispanic children are food insecure, according to these surveys. The rates are one in six for Asian households and one in ten for White households.
“The disparity is consistent,” Dr. Essel said. “We see what COVID has done. We once may have described it as a great equalizer – everyone is touched in the same way – but the reality is, this is actually a great magnifier. It’s revealing to us and magnifying disparities that have existed for far too long and has really allowed us to see it in a new way.”
A big part of disparities in food insecurity is disparities in wealth, “the safety net or cushion for families when things go wrong,” Dr. Essel said. The median wealth of White Americans in 2016 was $171,000, compared to $20,700 among Latinx Americans and $17,600 among Black Americans, according to the Federal Reserve Board Survey of Consumer Finances.
Food insecurity interventions
Federal nutrition programs – such as Supplemental Nutrition Assistance Program (SNAP), the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), and school meal programs – are key to addressing food insecurity, Dr. Essel said.
“They have a long track record of rescuing families out of poverty, of rescuing families from food security and improving overall health of families,” he said.
But emergency food relief programs are important as well. Four in 10 families currently coming into food pantries are new recipients, and these resources have seen a 60% increase in clients, he said.
“This is utterly unreasonable for them to be able to manage,” he said. “Food pantries are essential but inadequate to compensate for large numbers of families,” even while they also may be the only option for families unable or unwilling to access federal programs. For example, for every one meal that food banks can provide, SNAP can provide nine meals, Dr. Essel said. Further, during times of economic downtown, every SNAP $1 spent generates $1.50 to $2 in economic activity.
Currently, the Pandemic Electronic Benefit Transfer (P-EBT) program provides benefits to families for school breakfast and lunch and has been extended through December 2021. Another federal pandemic response was to increase SNAP to the maximum household benefit for families, about $646 for a family of four, although 40% of households were already receiving the maximum benefit.
Food insecurity advocacy
You can advocate for any one of multiple pillars when it comes to food insecurity, Dr. Essel said. “Food cannot solve food insecurity by itself,” he said. “We have to think about root causes – systemic causes – and think about unemployment, livable wage, systemic racism, oppression, an inequitable food system. All of these things are pillars that any of you can advocate for when recognizing a family that is struggling with food insecurity.”
He offered several suggestions for advocacy:
- Join your local AAP chapter and prioritize food insecurity.
- Join a local antihunger task force.
- Make your clinical environment as safe as possible for families to respond to questions about food insecurity.
- Know what’s happening in your community immigrant populations.
- Provide up-to-date information to families about eligibility for federal programs.
- Share stories through op-eds and letters to the editor, and by contacting congressional representatives and providing expert testimony to school boards and city councils.
- Educate others about food insecurity through the above channels and on social media.
Jessica Lazerov, MD, a general pediatrician at Children’s National Anacostia and assistant professor of pediatrics at George Washington University, Washington, said the session was fantastic.
“Dr. Essel went beyond the basics of food insecurity, delving into the root causes, potential solutions, and important considerations when screening for food insecurity in practice,” Dr. Lazerov said in an interview. “I enjoyed his focus on advocacy, as well as the fact that he spent a bit of time reviewing how the COVID pandemic has affected food insecurity. I truly felt empowered to take my advocacy efforts a step further as Dr. Essel laid out concrete, actionable next steps, as well as a review of the most relevant and current information about food insecurity.”
Dr. Essel, Dr. Lazerov, and Dr. Gray have no relevant financial disclosures.
and advocate on behalf of those experiencing or at risk of food insecurity, according to Kofi Essel, MD, MPH, a pediatrician at Children’s National Hospital in Washington.
More than one in four adults are dealing with food access hardships during the pandemic, Dr. Essel said at the virtual annual meeting of the American Academy of Pediatrics. Food insecurity is often interchangeable with hunger and refers to limited or uncertain availability of foods that are nutritious and safe.
“Food insecurity is as much about the threat of deprivation as it is about deprivation itself: A food-insecure life means a life lived in fear of hunger, and the psychological toll that takes,” according to a 2020 New York Times photo feature on food insecurity by Brenda Ann Kenneally that Dr. Essel quoted.
The lived experience of food insecure households includes food anxiety, a preoccupation with being able to get enough food that takes up cognitive bandwidth and prevents people from being able to focus on other important things. Another feature of food-insecure homes is a monotony of diet, which often involves an increase in caloric density and decrease in nutritional quality. As food insecurity grows more dire, adults’ food intake decreases, and then children’s intake decreases as adults seek out any way to get food, including “socially unacceptable” ways, which can include food pantries and bartering for food.
Food insecurity is associated with a wide range of negative outcomes even after accounting for other confounders, including decreased overall health, mental health, and educational outcomes. It’s also associated with an increase in developmental delays, hospitalizations, iron deficiency, asthma, and birth defects, among other problems. Somewhat paradoxically, it’s associated with both an increase and a decrease in obesity in the research.
Megan J. Gray, MD, MPH, assistant professor of pediatrics and population health at Dell Medical School at the The University of Texas at Austin, attended Dr. Essel’s session because food insecurity during COVID-19 now affects about half her patients, according to screening research she’s conducted.
“I wanted to learn more about the nuances of screening and using language and talking points that are helpful with families and with staff in building a culture of discussing food insecurity in our clinics,” Dr. Gray said in an interview. “What I’ve learned in my clinic is that if we don’t ask about it, families aren’t telling us – food insecurity is hiding in plain sight.”
She particularly appreciated Dr. Essel’s slides on the progression of food insecurity and how they acknowledged the mental health burden of food insecurity among parents.
“Right now during COVID-19, I see more patients I would call ‘socially complex’ rather than ‘medically complex,’ ” she said. “We all need to get a crash course in social work and Dr. Essel’s presentation is a great starting place.”
Screening for food insecurity
Beginning in 2015, an AAP policy statement charged pediatricians to “screen and intervene” with regard to food insecurity and their patients, Dr. Essel said. The statement also called for pediatricians to advocate for programs and policies that end childhood food insecurity.
The policy statement recommended a validated two-question screening tool called the Hunger Vital Sign:
1. “Within the past 12 months, we worried whether our food would run out before we got money to buy more.”
2. “Within the past 12 months, the food that we bought just didn’t last and we didn’t have money to get more.”
But in screening, you need to be conscious of how dignity intersects with food insecurity concerns, Dr. Essel said.
“We need to create dignity for our families,” he said. “We need to create a safe environment for our families and use appropriate tools when necessary to be able to identify families that are struggling with food insecurity.”
That need is seen in research on food screening. The Hunger Vital Signs questions can be asked with a dichotomous variable, as a yes/no question, or on a Likert scale, though the latter is a more complex way to ask.
A 2017 study found, however, that asking with “yes/no” answers missed more than a quarter of at-risk families. In the AAP survey using “yes/no” answers, 31% of families screened positive for being at risk of food insecurity, compared with 46% when the same question was asked on a Likert scale. It seems the ability to answer with “sometimes” feels “safer” than answering “yes,” Dr. Essel said.
Another factor that potentially affects answers is how doctors ask. In a March 2020 study at a single primary care practice, 16% of families screened positive with yes/no responses to a food insecurity screen when the questions were written, compared with 10% of positive screens with verbal responses (P < .001).
Epidemiology of food insecurity
The most updated United States Department of Agriculture report on food insecurity released in September shows the United States finally reached prerecession levels in 2019, with 11% of families designated as “food insecure.” But 2019 data cannot show what has occurred since the pandemic.
Further, the numbers are higher in households with children: Fourteen percent, or one in seven households with children, are experiencing food insecurity. Racial and ethnic disparities in food insecurity have remained consistent over the past 2 decades, with about twice as many Black and Hispanic homes experiencing food insecurity as White homes.
More recent research using Census Household Pulse Surveys has found a tremendous increase in food insecurity for children in 2020. One in three Black children and one in four Hispanic children are food insecure, according to these surveys. The rates are one in six for Asian households and one in ten for White households.
“The disparity is consistent,” Dr. Essel said. “We see what COVID has done. We once may have described it as a great equalizer – everyone is touched in the same way – but the reality is, this is actually a great magnifier. It’s revealing to us and magnifying disparities that have existed for far too long and has really allowed us to see it in a new way.”
A big part of disparities in food insecurity is disparities in wealth, “the safety net or cushion for families when things go wrong,” Dr. Essel said. The median wealth of White Americans in 2016 was $171,000, compared to $20,700 among Latinx Americans and $17,600 among Black Americans, according to the Federal Reserve Board Survey of Consumer Finances.
Food insecurity interventions
Federal nutrition programs – such as Supplemental Nutrition Assistance Program (SNAP), the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), and school meal programs – are key to addressing food insecurity, Dr. Essel said.
“They have a long track record of rescuing families out of poverty, of rescuing families from food security and improving overall health of families,” he said.
But emergency food relief programs are important as well. Four in 10 families currently coming into food pantries are new recipients, and these resources have seen a 60% increase in clients, he said.
“This is utterly unreasonable for them to be able to manage,” he said. “Food pantries are essential but inadequate to compensate for large numbers of families,” even while they also may be the only option for families unable or unwilling to access federal programs. For example, for every one meal that food banks can provide, SNAP can provide nine meals, Dr. Essel said. Further, during times of economic downtown, every SNAP $1 spent generates $1.50 to $2 in economic activity.
Currently, the Pandemic Electronic Benefit Transfer (P-EBT) program provides benefits to families for school breakfast and lunch and has been extended through December 2021. Another federal pandemic response was to increase SNAP to the maximum household benefit for families, about $646 for a family of four, although 40% of households were already receiving the maximum benefit.
Food insecurity advocacy
You can advocate for any one of multiple pillars when it comes to food insecurity, Dr. Essel said. “Food cannot solve food insecurity by itself,” he said. “We have to think about root causes – systemic causes – and think about unemployment, livable wage, systemic racism, oppression, an inequitable food system. All of these things are pillars that any of you can advocate for when recognizing a family that is struggling with food insecurity.”
He offered several suggestions for advocacy:
- Join your local AAP chapter and prioritize food insecurity.
- Join a local antihunger task force.
- Make your clinical environment as safe as possible for families to respond to questions about food insecurity.
- Know what’s happening in your community immigrant populations.
- Provide up-to-date information to families about eligibility for federal programs.
- Share stories through op-eds and letters to the editor, and by contacting congressional representatives and providing expert testimony to school boards and city councils.
- Educate others about food insecurity through the above channels and on social media.
Jessica Lazerov, MD, a general pediatrician at Children’s National Anacostia and assistant professor of pediatrics at George Washington University, Washington, said the session was fantastic.
“Dr. Essel went beyond the basics of food insecurity, delving into the root causes, potential solutions, and important considerations when screening for food insecurity in practice,” Dr. Lazerov said in an interview. “I enjoyed his focus on advocacy, as well as the fact that he spent a bit of time reviewing how the COVID pandemic has affected food insecurity. I truly felt empowered to take my advocacy efforts a step further as Dr. Essel laid out concrete, actionable next steps, as well as a review of the most relevant and current information about food insecurity.”
Dr. Essel, Dr. Lazerov, and Dr. Gray have no relevant financial disclosures.
and advocate on behalf of those experiencing or at risk of food insecurity, according to Kofi Essel, MD, MPH, a pediatrician at Children’s National Hospital in Washington.
More than one in four adults are dealing with food access hardships during the pandemic, Dr. Essel said at the virtual annual meeting of the American Academy of Pediatrics. Food insecurity is often interchangeable with hunger and refers to limited or uncertain availability of foods that are nutritious and safe.
“Food insecurity is as much about the threat of deprivation as it is about deprivation itself: A food-insecure life means a life lived in fear of hunger, and the psychological toll that takes,” according to a 2020 New York Times photo feature on food insecurity by Brenda Ann Kenneally that Dr. Essel quoted.
The lived experience of food insecure households includes food anxiety, a preoccupation with being able to get enough food that takes up cognitive bandwidth and prevents people from being able to focus on other important things. Another feature of food-insecure homes is a monotony of diet, which often involves an increase in caloric density and decrease in nutritional quality. As food insecurity grows more dire, adults’ food intake decreases, and then children’s intake decreases as adults seek out any way to get food, including “socially unacceptable” ways, which can include food pantries and bartering for food.
Food insecurity is associated with a wide range of negative outcomes even after accounting for other confounders, including decreased overall health, mental health, and educational outcomes. It’s also associated with an increase in developmental delays, hospitalizations, iron deficiency, asthma, and birth defects, among other problems. Somewhat paradoxically, it’s associated with both an increase and a decrease in obesity in the research.
Megan J. Gray, MD, MPH, assistant professor of pediatrics and population health at Dell Medical School at the The University of Texas at Austin, attended Dr. Essel’s session because food insecurity during COVID-19 now affects about half her patients, according to screening research she’s conducted.
“I wanted to learn more about the nuances of screening and using language and talking points that are helpful with families and with staff in building a culture of discussing food insecurity in our clinics,” Dr. Gray said in an interview. “What I’ve learned in my clinic is that if we don’t ask about it, families aren’t telling us – food insecurity is hiding in plain sight.”
She particularly appreciated Dr. Essel’s slides on the progression of food insecurity and how they acknowledged the mental health burden of food insecurity among parents.
“Right now during COVID-19, I see more patients I would call ‘socially complex’ rather than ‘medically complex,’ ” she said. “We all need to get a crash course in social work and Dr. Essel’s presentation is a great starting place.”
Screening for food insecurity
Beginning in 2015, an AAP policy statement charged pediatricians to “screen and intervene” with regard to food insecurity and their patients, Dr. Essel said. The statement also called for pediatricians to advocate for programs and policies that end childhood food insecurity.
The policy statement recommended a validated two-question screening tool called the Hunger Vital Sign:
1. “Within the past 12 months, we worried whether our food would run out before we got money to buy more.”
2. “Within the past 12 months, the food that we bought just didn’t last and we didn’t have money to get more.”
But in screening, you need to be conscious of how dignity intersects with food insecurity concerns, Dr. Essel said.
“We need to create dignity for our families,” he said. “We need to create a safe environment for our families and use appropriate tools when necessary to be able to identify families that are struggling with food insecurity.”
That need is seen in research on food screening. The Hunger Vital Signs questions can be asked with a dichotomous variable, as a yes/no question, or on a Likert scale, though the latter is a more complex way to ask.
A 2017 study found, however, that asking with “yes/no” answers missed more than a quarter of at-risk families. In the AAP survey using “yes/no” answers, 31% of families screened positive for being at risk of food insecurity, compared with 46% when the same question was asked on a Likert scale. It seems the ability to answer with “sometimes” feels “safer” than answering “yes,” Dr. Essel said.
Another factor that potentially affects answers is how doctors ask. In a March 2020 study at a single primary care practice, 16% of families screened positive with yes/no responses to a food insecurity screen when the questions were written, compared with 10% of positive screens with verbal responses (P < .001).
Epidemiology of food insecurity
The most updated United States Department of Agriculture report on food insecurity released in September shows the United States finally reached prerecession levels in 2019, with 11% of families designated as “food insecure.” But 2019 data cannot show what has occurred since the pandemic.
Further, the numbers are higher in households with children: Fourteen percent, or one in seven households with children, are experiencing food insecurity. Racial and ethnic disparities in food insecurity have remained consistent over the past 2 decades, with about twice as many Black and Hispanic homes experiencing food insecurity as White homes.
More recent research using Census Household Pulse Surveys has found a tremendous increase in food insecurity for children in 2020. One in three Black children and one in four Hispanic children are food insecure, according to these surveys. The rates are one in six for Asian households and one in ten for White households.
“The disparity is consistent,” Dr. Essel said. “We see what COVID has done. We once may have described it as a great equalizer – everyone is touched in the same way – but the reality is, this is actually a great magnifier. It’s revealing to us and magnifying disparities that have existed for far too long and has really allowed us to see it in a new way.”
A big part of disparities in food insecurity is disparities in wealth, “the safety net or cushion for families when things go wrong,” Dr. Essel said. The median wealth of White Americans in 2016 was $171,000, compared to $20,700 among Latinx Americans and $17,600 among Black Americans, according to the Federal Reserve Board Survey of Consumer Finances.
Food insecurity interventions
Federal nutrition programs – such as Supplemental Nutrition Assistance Program (SNAP), the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), and school meal programs – are key to addressing food insecurity, Dr. Essel said.
“They have a long track record of rescuing families out of poverty, of rescuing families from food security and improving overall health of families,” he said.
But emergency food relief programs are important as well. Four in 10 families currently coming into food pantries are new recipients, and these resources have seen a 60% increase in clients, he said.
“This is utterly unreasonable for them to be able to manage,” he said. “Food pantries are essential but inadequate to compensate for large numbers of families,” even while they also may be the only option for families unable or unwilling to access federal programs. For example, for every one meal that food banks can provide, SNAP can provide nine meals, Dr. Essel said. Further, during times of economic downtown, every SNAP $1 spent generates $1.50 to $2 in economic activity.
Currently, the Pandemic Electronic Benefit Transfer (P-EBT) program provides benefits to families for school breakfast and lunch and has been extended through December 2021. Another federal pandemic response was to increase SNAP to the maximum household benefit for families, about $646 for a family of four, although 40% of households were already receiving the maximum benefit.
Food insecurity advocacy
You can advocate for any one of multiple pillars when it comes to food insecurity, Dr. Essel said. “Food cannot solve food insecurity by itself,” he said. “We have to think about root causes – systemic causes – and think about unemployment, livable wage, systemic racism, oppression, an inequitable food system. All of these things are pillars that any of you can advocate for when recognizing a family that is struggling with food insecurity.”
He offered several suggestions for advocacy:
- Join your local AAP chapter and prioritize food insecurity.
- Join a local antihunger task force.
- Make your clinical environment as safe as possible for families to respond to questions about food insecurity.
- Know what’s happening in your community immigrant populations.
- Provide up-to-date information to families about eligibility for federal programs.
- Share stories through op-eds and letters to the editor, and by contacting congressional representatives and providing expert testimony to school boards and city councils.
- Educate others about food insecurity through the above channels and on social media.
Jessica Lazerov, MD, a general pediatrician at Children’s National Anacostia and assistant professor of pediatrics at George Washington University, Washington, said the session was fantastic.
“Dr. Essel went beyond the basics of food insecurity, delving into the root causes, potential solutions, and important considerations when screening for food insecurity in practice,” Dr. Lazerov said in an interview. “I enjoyed his focus on advocacy, as well as the fact that he spent a bit of time reviewing how the COVID pandemic has affected food insecurity. I truly felt empowered to take my advocacy efforts a step further as Dr. Essel laid out concrete, actionable next steps, as well as a review of the most relevant and current information about food insecurity.”
Dr. Essel, Dr. Lazerov, and Dr. Gray have no relevant financial disclosures.
EXPERT ANALYSIS FROM AAP 2020
Infectious disease is an increasing threat from climate change
“I would argue that the most important reason to care about climate change is because of our children,” Saul Hymes, MD, said at the annual meeting of the American Academy of Pediatrics, held virtually this year.
“Being able to point out to people how climate change harms the health of their children and affects their children’s risk of infections is a particularly effective argument to make,” said Dr. Hymes, a pediatric infectious diseases specialist at Stony Brook (N.Y.) University.
Rachel Boykan, MD, a pediatrician at the university, found Dr. Hymes’ presentation excellent and highly relevant to issues all health care workers treating children face, even beyond infectious disease.
“It was data focused but also understandable for a broad audience,” Dr. Boykan, who was not involved in the presentation, said in an interview. “He explained the science of climate change in a way that all physicians, but especially pediatricians, would find relevant. I suspect if people who were listening didn’t already prioritize the issues of climate change, they certainly did after hearing the talk.”
She also appreciated that Dr. Hymes addressed how climate change affects everyone in both their professional and personal lives.
“We need to be prepared to address the clinical issues that ensue after a natural disaster, and we need to be advocates for change so that we can slow down the climate changes we are all dealing with,” said Dr. Boykan, adding that the presentation was also inspiring. “He presented many different viewpoints and many ways to be involved and to be an advocate. I would think that a good number of people who were there would be energized to do something differently to combat climate change.”
The multitudinous impacts of climate change
The impact of climate change on human health is broad and far-reaching, Dr. Hymes said. It doesn’t require much imagination to recognize that rising global temperatures can lead to prolonged extreme heat waves that can cause heat-related deaths and illnesses. But other effects can be more gradual or subtle. Changes in outdoor air quality can affect weather patterns, pollen counts, and air pollution that can increase risk of asthma, allergies, as well as acute and chronic respiratory and cardiovascular disease.
Sea level rise, more frequent and severe hurricanes, storm surges, and extreme precipitation all can lead to contaminated water and destruction of essential infrastructure. In addition to drowning and injuries from the storms themselves, these changes have mental health consequences, and can lead to gastrointestinal and other illnesses, including water-borne infectious disease. The distribution and prevalence of vector-borne diseases also will shift with changes in temperature, precipitation, and other weather patterns.
Distribution, prevalence of vector-borne diseases shift with climate change
One of the most common bacteria transmitted by vectors in the United States is Borrelia burgdorferi, the cause of Lyme disease. Transmitted by deer ticks, Lyme disease is listed by the Environmental Protection Agency as an indicator of climate change’s impact on human health and is becoming more common every year. Cases doubled from 1990 to 2014, from 4 to 8 cases per 100,000 people.
Increases were most dramatic in the Northeast, where Lyme disease is endemic. States such as Maine, Vermont, and New Hampshire all saw increases of 80-100 more cases per 100,000 people. Evidence now shows that Lyme disease is moving north as the climate warms. Toronto, for example, has seen more than a 400% increase in cases in less than a decade, from 128 cases per 100,000 people in 2009 to 700 cases per 100,000 in 2015.
“It’s a known phenomenon that climate change affects more northerly latitudes disproportionately to more than southerly latitudes,” Dr. Hymes said. He shared a 2013 study providing evidence that climate change is expanding the range of Lyme disease. Even when controlling for other confounding factors, the research found that areas being warmed proportionately more by climate change also are experiencing greater Lyme incidence. While Lyme cases declined in several Western and Deep South states, it significantly increased in nearly every Northeast state as well as Idaho, Arizona, and states in the northern Midwest near the Great Lakes.
“We find that this impact of climate change on the movement of vectors like ticks affects more than just Lyme disease,” Dr. Hymes said. Amblyomma americanum, the Lone Star tick, has historically been restricted to the southern United States but is now found further north, even up to New England. It carries bacteria that can cause multiple illnesses, including ehrlichiosis, heartland virus, and tularemia.
An alpha-gal meat allergy associated with this tick can lead to anaphylaxis about 6 hours after a person eats red meat or pork. Prevalence of this allergy, first reported in Georgia in 1989-1991, has been increasing and moving further north, and the Lone Star tick is a particularly heat-tolerant and heat-loving tick.
Climate change also affects how long during the year people are at risk. Lyme disease, for example, typically lasted from April/May to October, when ticks then hibernated during the cold weather. But the warming climate has expanded Lyme season: Local Lyme cases have begun occurring into November through January on Long Island over the past 5 years.
The impact of seasonal changes on infectious diseases overall is difficult to predict. The seasons for cold weather diseases such as influenza and respiratory syncytial virus, for example, may become shorter or milder while viruses more common in the summer, such as enteroviruses, may become a risk year-round.
Natural disasters pose multiple risks
Natural disasters can pose immediate dangers to families and have a significant impact on mental health, but that’s not their only potential impact.
“Severe weather events such as hurricanes, floods, and tornadoes are well established in the climate change literature as an effect of increased temperatures and more volatile weather systems, but they also have a significant effect on infectious diseases and on children in particular,” Dr. Hymes said. “Hurricanes and flash floods can cause increases in infectious disease outbreaks through a variety of different ways.”
They can bring saltwater, freshwater, and sometimes soil organisms into the food and water supplies, and lead to sewage contamination from overloaded sewers, overflowing storm drains, and loss of power or pumps. Displaced animal vectors, such as rats, can lead to spread of other diseases, such as plague, hantavirus, typhus, and rabies.
Examples of saltwater organisms include Vibrio, Aeromonas, and Mycobacterium marinum, all of which can cause infections in wounds and/or diarrheal illness or bacteremia. Similarly, organisms from freshwater and soil that can cause serious illness or death include Aeromonas, Pseudomonas, Amebiasis, Giardia, and Legionella. Without access to clean water, or with contamination from overflowing sewage, cryptosporidium, Escherichia coli, salmonella, typhoid, norovirus, hepatitis A and E, and even cholera can also become problems as well.
In Houston following Hurricane Harvey, for example, cellulitis cases doubled and included infections from organisms different from the usual suspects. Scrapes and cuts that occurred during the storm also festered sooner.
Cases of disease linked to Hurricane Katrina in a Centers for Disease Control and Prevention report included 6 cases of cholera, 17 cases of other vibrio – including five that resulted in death – and reported cases of norovirus, Escherichia coli, salmonella, and influenza and pneumonia from overcrowding of evacuees.
You can help in a variety of ways
You can play several key roles as the world’s climate changes, starting with preparing for the changes. You should familiarize themselves with new and emerging infections, or those that have been around a while but not seen in your areas, such as Lyme, Zika, and Dengue.
“If you haven’t seen them already, you likely will due to movements of vector-borne infections that can occur due to climate change,” Dr. Hymes said. “You also want to expect the usual common diseases, but maybe at unsuspected times,” he added. “If you have a pediatric patient who looks like they have Coxsackie virus but it’s February, if it’s been a warm February, it may very well be Coxsackie virus.”
Following natural disasters such as floods, hurricanes and tornadoes, consider who your patients are. If they’re evacuees, are they living in overcrowded conditions? Do they have access to clean water? If not, explain the need to boil water if they can, or to use iodine tablets or a portable pump filter. Consider that some infections may involve unexpected or odd organisms, such as legionella pneumonia or vibrio cellulitis, and contact your local infectious disease doctor as needed.
You also can make personal lifestyle changes that, while small, can add up in the aggregate in reducing carbon footprints, such as purchasing an electric or hybrid car and converting their homes to solar power.
“For very little money, you can purchase carbon offsets,” Dr. Hymes said, such as $10-$15 a month for wind power offsets with home electricity or $5-$10 a month for car or plane travel.
“But really, the most important thing we can do as pediatricians is educate,” Dr. Hymes said. “Taking opportunities every day in your office to educate your patients and educate your colleagues about the importance of climate change in our patients’ health and our own children’s health is super, super important.”
Dr. Hymes and Dr. Boykan had no relevant financial disclosures.
“I would argue that the most important reason to care about climate change is because of our children,” Saul Hymes, MD, said at the annual meeting of the American Academy of Pediatrics, held virtually this year.
“Being able to point out to people how climate change harms the health of their children and affects their children’s risk of infections is a particularly effective argument to make,” said Dr. Hymes, a pediatric infectious diseases specialist at Stony Brook (N.Y.) University.
Rachel Boykan, MD, a pediatrician at the university, found Dr. Hymes’ presentation excellent and highly relevant to issues all health care workers treating children face, even beyond infectious disease.
“It was data focused but also understandable for a broad audience,” Dr. Boykan, who was not involved in the presentation, said in an interview. “He explained the science of climate change in a way that all physicians, but especially pediatricians, would find relevant. I suspect if people who were listening didn’t already prioritize the issues of climate change, they certainly did after hearing the talk.”
She also appreciated that Dr. Hymes addressed how climate change affects everyone in both their professional and personal lives.
“We need to be prepared to address the clinical issues that ensue after a natural disaster, and we need to be advocates for change so that we can slow down the climate changes we are all dealing with,” said Dr. Boykan, adding that the presentation was also inspiring. “He presented many different viewpoints and many ways to be involved and to be an advocate. I would think that a good number of people who were there would be energized to do something differently to combat climate change.”
The multitudinous impacts of climate change
The impact of climate change on human health is broad and far-reaching, Dr. Hymes said. It doesn’t require much imagination to recognize that rising global temperatures can lead to prolonged extreme heat waves that can cause heat-related deaths and illnesses. But other effects can be more gradual or subtle. Changes in outdoor air quality can affect weather patterns, pollen counts, and air pollution that can increase risk of asthma, allergies, as well as acute and chronic respiratory and cardiovascular disease.
Sea level rise, more frequent and severe hurricanes, storm surges, and extreme precipitation all can lead to contaminated water and destruction of essential infrastructure. In addition to drowning and injuries from the storms themselves, these changes have mental health consequences, and can lead to gastrointestinal and other illnesses, including water-borne infectious disease. The distribution and prevalence of vector-borne diseases also will shift with changes in temperature, precipitation, and other weather patterns.
Distribution, prevalence of vector-borne diseases shift with climate change
One of the most common bacteria transmitted by vectors in the United States is Borrelia burgdorferi, the cause of Lyme disease. Transmitted by deer ticks, Lyme disease is listed by the Environmental Protection Agency as an indicator of climate change’s impact on human health and is becoming more common every year. Cases doubled from 1990 to 2014, from 4 to 8 cases per 100,000 people.
Increases were most dramatic in the Northeast, where Lyme disease is endemic. States such as Maine, Vermont, and New Hampshire all saw increases of 80-100 more cases per 100,000 people. Evidence now shows that Lyme disease is moving north as the climate warms. Toronto, for example, has seen more than a 400% increase in cases in less than a decade, from 128 cases per 100,000 people in 2009 to 700 cases per 100,000 in 2015.
“It’s a known phenomenon that climate change affects more northerly latitudes disproportionately to more than southerly latitudes,” Dr. Hymes said. He shared a 2013 study providing evidence that climate change is expanding the range of Lyme disease. Even when controlling for other confounding factors, the research found that areas being warmed proportionately more by climate change also are experiencing greater Lyme incidence. While Lyme cases declined in several Western and Deep South states, it significantly increased in nearly every Northeast state as well as Idaho, Arizona, and states in the northern Midwest near the Great Lakes.
“We find that this impact of climate change on the movement of vectors like ticks affects more than just Lyme disease,” Dr. Hymes said. Amblyomma americanum, the Lone Star tick, has historically been restricted to the southern United States but is now found further north, even up to New England. It carries bacteria that can cause multiple illnesses, including ehrlichiosis, heartland virus, and tularemia.
An alpha-gal meat allergy associated with this tick can lead to anaphylaxis about 6 hours after a person eats red meat or pork. Prevalence of this allergy, first reported in Georgia in 1989-1991, has been increasing and moving further north, and the Lone Star tick is a particularly heat-tolerant and heat-loving tick.
Climate change also affects how long during the year people are at risk. Lyme disease, for example, typically lasted from April/May to October, when ticks then hibernated during the cold weather. But the warming climate has expanded Lyme season: Local Lyme cases have begun occurring into November through January on Long Island over the past 5 years.
The impact of seasonal changes on infectious diseases overall is difficult to predict. The seasons for cold weather diseases such as influenza and respiratory syncytial virus, for example, may become shorter or milder while viruses more common in the summer, such as enteroviruses, may become a risk year-round.
Natural disasters pose multiple risks
Natural disasters can pose immediate dangers to families and have a significant impact on mental health, but that’s not their only potential impact.
“Severe weather events such as hurricanes, floods, and tornadoes are well established in the climate change literature as an effect of increased temperatures and more volatile weather systems, but they also have a significant effect on infectious diseases and on children in particular,” Dr. Hymes said. “Hurricanes and flash floods can cause increases in infectious disease outbreaks through a variety of different ways.”
They can bring saltwater, freshwater, and sometimes soil organisms into the food and water supplies, and lead to sewage contamination from overloaded sewers, overflowing storm drains, and loss of power or pumps. Displaced animal vectors, such as rats, can lead to spread of other diseases, such as plague, hantavirus, typhus, and rabies.
Examples of saltwater organisms include Vibrio, Aeromonas, and Mycobacterium marinum, all of which can cause infections in wounds and/or diarrheal illness or bacteremia. Similarly, organisms from freshwater and soil that can cause serious illness or death include Aeromonas, Pseudomonas, Amebiasis, Giardia, and Legionella. Without access to clean water, or with contamination from overflowing sewage, cryptosporidium, Escherichia coli, salmonella, typhoid, norovirus, hepatitis A and E, and even cholera can also become problems as well.
In Houston following Hurricane Harvey, for example, cellulitis cases doubled and included infections from organisms different from the usual suspects. Scrapes and cuts that occurred during the storm also festered sooner.
Cases of disease linked to Hurricane Katrina in a Centers for Disease Control and Prevention report included 6 cases of cholera, 17 cases of other vibrio – including five that resulted in death – and reported cases of norovirus, Escherichia coli, salmonella, and influenza and pneumonia from overcrowding of evacuees.
You can help in a variety of ways
You can play several key roles as the world’s climate changes, starting with preparing for the changes. You should familiarize themselves with new and emerging infections, or those that have been around a while but not seen in your areas, such as Lyme, Zika, and Dengue.
“If you haven’t seen them already, you likely will due to movements of vector-borne infections that can occur due to climate change,” Dr. Hymes said. “You also want to expect the usual common diseases, but maybe at unsuspected times,” he added. “If you have a pediatric patient who looks like they have Coxsackie virus but it’s February, if it’s been a warm February, it may very well be Coxsackie virus.”
Following natural disasters such as floods, hurricanes and tornadoes, consider who your patients are. If they’re evacuees, are they living in overcrowded conditions? Do they have access to clean water? If not, explain the need to boil water if they can, or to use iodine tablets or a portable pump filter. Consider that some infections may involve unexpected or odd organisms, such as legionella pneumonia or vibrio cellulitis, and contact your local infectious disease doctor as needed.
You also can make personal lifestyle changes that, while small, can add up in the aggregate in reducing carbon footprints, such as purchasing an electric or hybrid car and converting their homes to solar power.
“For very little money, you can purchase carbon offsets,” Dr. Hymes said, such as $10-$15 a month for wind power offsets with home electricity or $5-$10 a month for car or plane travel.
“But really, the most important thing we can do as pediatricians is educate,” Dr. Hymes said. “Taking opportunities every day in your office to educate your patients and educate your colleagues about the importance of climate change in our patients’ health and our own children’s health is super, super important.”
Dr. Hymes and Dr. Boykan had no relevant financial disclosures.
“I would argue that the most important reason to care about climate change is because of our children,” Saul Hymes, MD, said at the annual meeting of the American Academy of Pediatrics, held virtually this year.
“Being able to point out to people how climate change harms the health of their children and affects their children’s risk of infections is a particularly effective argument to make,” said Dr. Hymes, a pediatric infectious diseases specialist at Stony Brook (N.Y.) University.
Rachel Boykan, MD, a pediatrician at the university, found Dr. Hymes’ presentation excellent and highly relevant to issues all health care workers treating children face, even beyond infectious disease.
“It was data focused but also understandable for a broad audience,” Dr. Boykan, who was not involved in the presentation, said in an interview. “He explained the science of climate change in a way that all physicians, but especially pediatricians, would find relevant. I suspect if people who were listening didn’t already prioritize the issues of climate change, they certainly did after hearing the talk.”
She also appreciated that Dr. Hymes addressed how climate change affects everyone in both their professional and personal lives.
“We need to be prepared to address the clinical issues that ensue after a natural disaster, and we need to be advocates for change so that we can slow down the climate changes we are all dealing with,” said Dr. Boykan, adding that the presentation was also inspiring. “He presented many different viewpoints and many ways to be involved and to be an advocate. I would think that a good number of people who were there would be energized to do something differently to combat climate change.”
The multitudinous impacts of climate change
The impact of climate change on human health is broad and far-reaching, Dr. Hymes said. It doesn’t require much imagination to recognize that rising global temperatures can lead to prolonged extreme heat waves that can cause heat-related deaths and illnesses. But other effects can be more gradual or subtle. Changes in outdoor air quality can affect weather patterns, pollen counts, and air pollution that can increase risk of asthma, allergies, as well as acute and chronic respiratory and cardiovascular disease.
Sea level rise, more frequent and severe hurricanes, storm surges, and extreme precipitation all can lead to contaminated water and destruction of essential infrastructure. In addition to drowning and injuries from the storms themselves, these changes have mental health consequences, and can lead to gastrointestinal and other illnesses, including water-borne infectious disease. The distribution and prevalence of vector-borne diseases also will shift with changes in temperature, precipitation, and other weather patterns.
Distribution, prevalence of vector-borne diseases shift with climate change
One of the most common bacteria transmitted by vectors in the United States is Borrelia burgdorferi, the cause of Lyme disease. Transmitted by deer ticks, Lyme disease is listed by the Environmental Protection Agency as an indicator of climate change’s impact on human health and is becoming more common every year. Cases doubled from 1990 to 2014, from 4 to 8 cases per 100,000 people.
Increases were most dramatic in the Northeast, where Lyme disease is endemic. States such as Maine, Vermont, and New Hampshire all saw increases of 80-100 more cases per 100,000 people. Evidence now shows that Lyme disease is moving north as the climate warms. Toronto, for example, has seen more than a 400% increase in cases in less than a decade, from 128 cases per 100,000 people in 2009 to 700 cases per 100,000 in 2015.
“It’s a known phenomenon that climate change affects more northerly latitudes disproportionately to more than southerly latitudes,” Dr. Hymes said. He shared a 2013 study providing evidence that climate change is expanding the range of Lyme disease. Even when controlling for other confounding factors, the research found that areas being warmed proportionately more by climate change also are experiencing greater Lyme incidence. While Lyme cases declined in several Western and Deep South states, it significantly increased in nearly every Northeast state as well as Idaho, Arizona, and states in the northern Midwest near the Great Lakes.
“We find that this impact of climate change on the movement of vectors like ticks affects more than just Lyme disease,” Dr. Hymes said. Amblyomma americanum, the Lone Star tick, has historically been restricted to the southern United States but is now found further north, even up to New England. It carries bacteria that can cause multiple illnesses, including ehrlichiosis, heartland virus, and tularemia.
An alpha-gal meat allergy associated with this tick can lead to anaphylaxis about 6 hours after a person eats red meat or pork. Prevalence of this allergy, first reported in Georgia in 1989-1991, has been increasing and moving further north, and the Lone Star tick is a particularly heat-tolerant and heat-loving tick.
Climate change also affects how long during the year people are at risk. Lyme disease, for example, typically lasted from April/May to October, when ticks then hibernated during the cold weather. But the warming climate has expanded Lyme season: Local Lyme cases have begun occurring into November through January on Long Island over the past 5 years.
The impact of seasonal changes on infectious diseases overall is difficult to predict. The seasons for cold weather diseases such as influenza and respiratory syncytial virus, for example, may become shorter or milder while viruses more common in the summer, such as enteroviruses, may become a risk year-round.
Natural disasters pose multiple risks
Natural disasters can pose immediate dangers to families and have a significant impact on mental health, but that’s not their only potential impact.
“Severe weather events such as hurricanes, floods, and tornadoes are well established in the climate change literature as an effect of increased temperatures and more volatile weather systems, but they also have a significant effect on infectious diseases and on children in particular,” Dr. Hymes said. “Hurricanes and flash floods can cause increases in infectious disease outbreaks through a variety of different ways.”
They can bring saltwater, freshwater, and sometimes soil organisms into the food and water supplies, and lead to sewage contamination from overloaded sewers, overflowing storm drains, and loss of power or pumps. Displaced animal vectors, such as rats, can lead to spread of other diseases, such as plague, hantavirus, typhus, and rabies.
Examples of saltwater organisms include Vibrio, Aeromonas, and Mycobacterium marinum, all of which can cause infections in wounds and/or diarrheal illness or bacteremia. Similarly, organisms from freshwater and soil that can cause serious illness or death include Aeromonas, Pseudomonas, Amebiasis, Giardia, and Legionella. Without access to clean water, or with contamination from overflowing sewage, cryptosporidium, Escherichia coli, salmonella, typhoid, norovirus, hepatitis A and E, and even cholera can also become problems as well.
In Houston following Hurricane Harvey, for example, cellulitis cases doubled and included infections from organisms different from the usual suspects. Scrapes and cuts that occurred during the storm also festered sooner.
Cases of disease linked to Hurricane Katrina in a Centers for Disease Control and Prevention report included 6 cases of cholera, 17 cases of other vibrio – including five that resulted in death – and reported cases of norovirus, Escherichia coli, salmonella, and influenza and pneumonia from overcrowding of evacuees.
You can help in a variety of ways
You can play several key roles as the world’s climate changes, starting with preparing for the changes. You should familiarize themselves with new and emerging infections, or those that have been around a while but not seen in your areas, such as Lyme, Zika, and Dengue.
“If you haven’t seen them already, you likely will due to movements of vector-borne infections that can occur due to climate change,” Dr. Hymes said. “You also want to expect the usual common diseases, but maybe at unsuspected times,” he added. “If you have a pediatric patient who looks like they have Coxsackie virus but it’s February, if it’s been a warm February, it may very well be Coxsackie virus.”
Following natural disasters such as floods, hurricanes and tornadoes, consider who your patients are. If they’re evacuees, are they living in overcrowded conditions? Do they have access to clean water? If not, explain the need to boil water if they can, or to use iodine tablets or a portable pump filter. Consider that some infections may involve unexpected or odd organisms, such as legionella pneumonia or vibrio cellulitis, and contact your local infectious disease doctor as needed.
You also can make personal lifestyle changes that, while small, can add up in the aggregate in reducing carbon footprints, such as purchasing an electric or hybrid car and converting their homes to solar power.
“For very little money, you can purchase carbon offsets,” Dr. Hymes said, such as $10-$15 a month for wind power offsets with home electricity or $5-$10 a month for car or plane travel.
“But really, the most important thing we can do as pediatricians is educate,” Dr. Hymes said. “Taking opportunities every day in your office to educate your patients and educate your colleagues about the importance of climate change in our patients’ health and our own children’s health is super, super important.”
Dr. Hymes and Dr. Boykan had no relevant financial disclosures.
FROM AAP 2020
A 4-year-old presented to our pediatric dermatology clinic for evaluation of asymptomatic "brown spots."
Capillary malformation-arteriovenous malformation syndrome
with or without arteriovenous malformations, as well as arteriovenous fistulas (AVFs). CM-AVM is an autosomal dominant disorder.1 CM-AVM type 1 is caused by mutations in the RASA1 gene, and CM-AVM type 2 is caused by mutations in the EPHB4 gene.2 Approximately 70% of patients with RASA1-associated CM-AVM syndrome and 80% of patients with EPHB4-associated CM-AVM syndrome have an affected parent, while the remainder have de novo variants.1
In patients with CM-AVM syndrome, CMs are often present at birth and more are typically acquired over time. CMs are characteristically 1-3 cm in diameter, round or oval, dull red or red-brown macules and patches with a blanched halo.3 Some CMs may be warm to touch indicating a possible underlying AVM or AVF.4 This can be confirmed by Doppler ultrasound, which would demonstrate increased arterial flow.4 CMs are most commonly located on the face and limbs and may present in isolation, but approximately one-third of patients have associated AVMs and AVFs.1,5 These high-flow vascular malformations may be present in skin, muscle, bone, brain, and/or spine and may be asymptomatic or lead to serious sequelae, including bleeding, congestive heart failure, and neurologic complications, such as migraine headaches, seizures, or even stroke.5 Symptoms from intracranial and spinal high-flow lesions usually present in early childhood and affect approximately 7% of patients.3
The diagnosis of CM-AVM should be suspected in an individual with numerous characteristic CMs and may be supported by the presence of AVMs and AVFs, family history of CM-AVM, and/or identification of RASA1 or EPHB4 mutation by molecular genetic testing.1,3 Although there are no consensus protocols for imaging CM-AVM patients, MRI of the brain and spine is recommended at diagnosis to identify underlying high-flow lesions.1 This may allow for early treatment before the development of symptoms.1 Any lesions identified on screening imaging may require regular surveillance, which is best determined by discussion with the radiologist.1 Although there are no reports of patients with negative results on screening imaging who later develop AVMs or AVFs, there should be a low threshold for repeat imaging in patients who develop new symptoms or physical exam findings.3,4
It has previously been suggested that the CMs in CM-AVM may actually represent early or small AVMs and pulsed-dye laser (PDL) treatment was not recommended because of concern for potential progression of lesions.4 However, a recent study demonstrated good response to PDL in patients with CM-AVM with no evidence of worsening or recurrence of lesions with long-term follow-up.6 Treatment of CMs that cause cosmetic concerns may be considered following discussion of risks and benefits with a dermatologist. Management of AVMs and AVFs requires a multidisciplinary team that, depending on location and symptoms of these features, may require the expertise of specialists such as neurosurgery, surgery, orthopedics, cardiology, and/or interventional radiology.1
Given the suspicion for CM-AVM in our patient, further workup was completed. A skin biopsy was consistent with CM. Genetic testing with the Vascular Malformations Panel, Sequencing and Deletion/Duplication revealed a pathogenic variant in the RASA1 gene and a variant of unknown clinical significance in the TEK gene. Parental genetic testing for the RASA1 mutation was negative, supporting a de novo mutation in the patient. CNS imaging showed a small developmental venous malformation in the brain that neurosurgery did not think was clinically significant. At the most recent follow-up at age 8 years, our patient had developed a few new small CMs but was otherwise well.
Dr. Leszczynska is trained in pediatrics and is the current dermatology research fellow at the University of Texas at Austin. Ms. Croce is a dermatology-trained pediatric nurse practitioner and PhD student at the University of Texas at Austin School of Nursing. Dr. Diaz is chief of pediatric dermatology at Dell Children’s Medical Center, Austin, assistant professor of pediatrics and medicine (dermatology), and dermatology residency associate program director at University of Texas at Austin . The authors have no relevant conflicts of interest to disclose. Donna Bilu Martin, MD, is the editor of this column.
References
1. Bayrak-Toydemir P, Stevenson D. Capillary Malformation-Arteriovenous Malformation Syndrome. In: Adam MP, Ardinger HH, Pagon RA, et al., eds. GeneReviews®. Seattle: University of Washington, Seattle; February 22, 2011.
2.Yu J et al. Pediatr Dermatol. 2017 Sep;34(5):e227-30.
3. Orme CM et al. Pediatr Dermatol. 2013 Jul-Aug;30(4):409-15.
4. Weitz NA et al. Pediatr Dermatol. 2015 Jan-Feb;32(1):76-84.
5. Revencu N et al. Hum Mutat. 2013 Dec;34(12):1632-41.
6. Iznardo H et al. Pediatr Dermatol. 2020 Mar;37(2):342-44.
Capillary malformation-arteriovenous malformation syndrome
with or without arteriovenous malformations, as well as arteriovenous fistulas (AVFs). CM-AVM is an autosomal dominant disorder.1 CM-AVM type 1 is caused by mutations in the RASA1 gene, and CM-AVM type 2 is caused by mutations in the EPHB4 gene.2 Approximately 70% of patients with RASA1-associated CM-AVM syndrome and 80% of patients with EPHB4-associated CM-AVM syndrome have an affected parent, while the remainder have de novo variants.1
In patients with CM-AVM syndrome, CMs are often present at birth and more are typically acquired over time. CMs are characteristically 1-3 cm in diameter, round or oval, dull red or red-brown macules and patches with a blanched halo.3 Some CMs may be warm to touch indicating a possible underlying AVM or AVF.4 This can be confirmed by Doppler ultrasound, which would demonstrate increased arterial flow.4 CMs are most commonly located on the face and limbs and may present in isolation, but approximately one-third of patients have associated AVMs and AVFs.1,5 These high-flow vascular malformations may be present in skin, muscle, bone, brain, and/or spine and may be asymptomatic or lead to serious sequelae, including bleeding, congestive heart failure, and neurologic complications, such as migraine headaches, seizures, or even stroke.5 Symptoms from intracranial and spinal high-flow lesions usually present in early childhood and affect approximately 7% of patients.3
The diagnosis of CM-AVM should be suspected in an individual with numerous characteristic CMs and may be supported by the presence of AVMs and AVFs, family history of CM-AVM, and/or identification of RASA1 or EPHB4 mutation by molecular genetic testing.1,3 Although there are no consensus protocols for imaging CM-AVM patients, MRI of the brain and spine is recommended at diagnosis to identify underlying high-flow lesions.1 This may allow for early treatment before the development of symptoms.1 Any lesions identified on screening imaging may require regular surveillance, which is best determined by discussion with the radiologist.1 Although there are no reports of patients with negative results on screening imaging who later develop AVMs or AVFs, there should be a low threshold for repeat imaging in patients who develop new symptoms or physical exam findings.3,4
It has previously been suggested that the CMs in CM-AVM may actually represent early or small AVMs and pulsed-dye laser (PDL) treatment was not recommended because of concern for potential progression of lesions.4 However, a recent study demonstrated good response to PDL in patients with CM-AVM with no evidence of worsening or recurrence of lesions with long-term follow-up.6 Treatment of CMs that cause cosmetic concerns may be considered following discussion of risks and benefits with a dermatologist. Management of AVMs and AVFs requires a multidisciplinary team that, depending on location and symptoms of these features, may require the expertise of specialists such as neurosurgery, surgery, orthopedics, cardiology, and/or interventional radiology.1
Given the suspicion for CM-AVM in our patient, further workup was completed. A skin biopsy was consistent with CM. Genetic testing with the Vascular Malformations Panel, Sequencing and Deletion/Duplication revealed a pathogenic variant in the RASA1 gene and a variant of unknown clinical significance in the TEK gene. Parental genetic testing for the RASA1 mutation was negative, supporting a de novo mutation in the patient. CNS imaging showed a small developmental venous malformation in the brain that neurosurgery did not think was clinically significant. At the most recent follow-up at age 8 years, our patient had developed a few new small CMs but was otherwise well.
Dr. Leszczynska is trained in pediatrics and is the current dermatology research fellow at the University of Texas at Austin. Ms. Croce is a dermatology-trained pediatric nurse practitioner and PhD student at the University of Texas at Austin School of Nursing. Dr. Diaz is chief of pediatric dermatology at Dell Children’s Medical Center, Austin, assistant professor of pediatrics and medicine (dermatology), and dermatology residency associate program director at University of Texas at Austin . The authors have no relevant conflicts of interest to disclose. Donna Bilu Martin, MD, is the editor of this column.
References
1. Bayrak-Toydemir P, Stevenson D. Capillary Malformation-Arteriovenous Malformation Syndrome. In: Adam MP, Ardinger HH, Pagon RA, et al., eds. GeneReviews®. Seattle: University of Washington, Seattle; February 22, 2011.
2.Yu J et al. Pediatr Dermatol. 2017 Sep;34(5):e227-30.
3. Orme CM et al. Pediatr Dermatol. 2013 Jul-Aug;30(4):409-15.
4. Weitz NA et al. Pediatr Dermatol. 2015 Jan-Feb;32(1):76-84.
5. Revencu N et al. Hum Mutat. 2013 Dec;34(12):1632-41.
6. Iznardo H et al. Pediatr Dermatol. 2020 Mar;37(2):342-44.
Capillary malformation-arteriovenous malformation syndrome
with or without arteriovenous malformations, as well as arteriovenous fistulas (AVFs). CM-AVM is an autosomal dominant disorder.1 CM-AVM type 1 is caused by mutations in the RASA1 gene, and CM-AVM type 2 is caused by mutations in the EPHB4 gene.2 Approximately 70% of patients with RASA1-associated CM-AVM syndrome and 80% of patients with EPHB4-associated CM-AVM syndrome have an affected parent, while the remainder have de novo variants.1
In patients with CM-AVM syndrome, CMs are often present at birth and more are typically acquired over time. CMs are characteristically 1-3 cm in diameter, round or oval, dull red or red-brown macules and patches with a blanched halo.3 Some CMs may be warm to touch indicating a possible underlying AVM or AVF.4 This can be confirmed by Doppler ultrasound, which would demonstrate increased arterial flow.4 CMs are most commonly located on the face and limbs and may present in isolation, but approximately one-third of patients have associated AVMs and AVFs.1,5 These high-flow vascular malformations may be present in skin, muscle, bone, brain, and/or spine and may be asymptomatic or lead to serious sequelae, including bleeding, congestive heart failure, and neurologic complications, such as migraine headaches, seizures, or even stroke.5 Symptoms from intracranial and spinal high-flow lesions usually present in early childhood and affect approximately 7% of patients.3
The diagnosis of CM-AVM should be suspected in an individual with numerous characteristic CMs and may be supported by the presence of AVMs and AVFs, family history of CM-AVM, and/or identification of RASA1 or EPHB4 mutation by molecular genetic testing.1,3 Although there are no consensus protocols for imaging CM-AVM patients, MRI of the brain and spine is recommended at diagnosis to identify underlying high-flow lesions.1 This may allow for early treatment before the development of symptoms.1 Any lesions identified on screening imaging may require regular surveillance, which is best determined by discussion with the radiologist.1 Although there are no reports of patients with negative results on screening imaging who later develop AVMs or AVFs, there should be a low threshold for repeat imaging in patients who develop new symptoms or physical exam findings.3,4
It has previously been suggested that the CMs in CM-AVM may actually represent early or small AVMs and pulsed-dye laser (PDL) treatment was not recommended because of concern for potential progression of lesions.4 However, a recent study demonstrated good response to PDL in patients with CM-AVM with no evidence of worsening or recurrence of lesions with long-term follow-up.6 Treatment of CMs that cause cosmetic concerns may be considered following discussion of risks and benefits with a dermatologist. Management of AVMs and AVFs requires a multidisciplinary team that, depending on location and symptoms of these features, may require the expertise of specialists such as neurosurgery, surgery, orthopedics, cardiology, and/or interventional radiology.1
Given the suspicion for CM-AVM in our patient, further workup was completed. A skin biopsy was consistent with CM. Genetic testing with the Vascular Malformations Panel, Sequencing and Deletion/Duplication revealed a pathogenic variant in the RASA1 gene and a variant of unknown clinical significance in the TEK gene. Parental genetic testing for the RASA1 mutation was negative, supporting a de novo mutation in the patient. CNS imaging showed a small developmental venous malformation in the brain that neurosurgery did not think was clinically significant. At the most recent follow-up at age 8 years, our patient had developed a few new small CMs but was otherwise well.
Dr. Leszczynska is trained in pediatrics and is the current dermatology research fellow at the University of Texas at Austin. Ms. Croce is a dermatology-trained pediatric nurse practitioner and PhD student at the University of Texas at Austin School of Nursing. Dr. Diaz is chief of pediatric dermatology at Dell Children’s Medical Center, Austin, assistant professor of pediatrics and medicine (dermatology), and dermatology residency associate program director at University of Texas at Austin . The authors have no relevant conflicts of interest to disclose. Donna Bilu Martin, MD, is the editor of this column.
References
1. Bayrak-Toydemir P, Stevenson D. Capillary Malformation-Arteriovenous Malformation Syndrome. In: Adam MP, Ardinger HH, Pagon RA, et al., eds. GeneReviews®. Seattle: University of Washington, Seattle; February 22, 2011.
2.Yu J et al. Pediatr Dermatol. 2017 Sep;34(5):e227-30.
3. Orme CM et al. Pediatr Dermatol. 2013 Jul-Aug;30(4):409-15.
4. Weitz NA et al. Pediatr Dermatol. 2015 Jan-Feb;32(1):76-84.
5. Revencu N et al. Hum Mutat. 2013 Dec;34(12):1632-41.
6. Iznardo H et al. Pediatr Dermatol. 2020 Mar;37(2):342-44.
Whales, seals, and dolphins: Will SARS-CoV-2–contaminated wastewater prove a killer?
Zoonoses are no respecter of biological boundaries and are notorious for crossing genus and even higher taxonomic boundaries. SARS-CoV-2 is no exception, the current outbreak most probably having originated in bats, a common source of human-affecting zoonoses throughout history. But it is not a one-way street, and the virus has been shown to spread from infected humans to a variety of other land mammals, including our domesticated animals and kept zoo species.
A recent troubling report, however, has indicated that sea mammals may be part of a next wave of likely candidates for infection, put at risk by the current human pandemic and environmental degradation on a global scale, according to a the results of a genomic analysis of four major groups of sea mammals.
Researchers Sabateeshan Mathavarajah and colleagues from Dalhousie University, Halifax, N.S., examined the sequences of the ACE2 receptors in the various marine mammal species. The ACE2 receptor has recently been identified as the SARS-CoV-2 receptor, which allows for infection.
The researchers examined genomic databases of the marine species to determine if their ACE2 receptor sequences indicated the potential for high, medium, or low susceptibility to infection, as reported in Science of the Total Environment. Database analysis was performed for four groups: Cetacea (whales and dolphins), Pinnepidia (seals), Sirenia (sea cows), and Fissipedia (sea otters and polar bears).
The researchers defined susceptibility values based on comparable binding with the receptor and came up with the following subgroups: higher than human, high (resembles human ACE2), medium (resembles cat ACE2), and low (resembles dog ACE2). It has yet to be established if these marine mammals actually are infected with SARS-CoV-2 and what the impact of such an infection might have on animal health or humans who come in contact with infected animals.
They also cross-referenced for the level of species endangerment and with maps of potential wastewater contamination for certain areas that species came in contact with, using Alaska as the model.
Populations in danger
The researchers found 15 species that are already at risk globally that fall under the categories of near threatened, vulnerable, endangered, and critically endangered that were predicted to be medium to higher susceptibility to the SARS-CoV-2 virus than humans. Cross infection is of particular concern because other coronaviruses have been shown to have severe and lethal effects among many of these species.
Among the potentially impacted species were the near threatened–status Antarctic Mink whale and the stellar sea lion; the vulnerable sperm whale, northern fur seal, and Atlantic walrus; the endangered northern and southern sea otters, the North Pacific right whale, and the Amazon River dolphin; and the critically threatened Baiji and Vaquita dolphin species.
Pollution risks
In Alaska, as of Aug. 7th, 2020, there were 4,221 confirmed cases of COVID-19 and this number continues to rise, according to the researchers. Since there is a diversity of marine mammals in Alaska and their populations are well documented, they compared this information with available data on the wastewater treatment plants in the state. They were thus able to determine the potential geographic locations and species at high risk for transmission of SARS-CoV-2 via wastewater effluent.
Among their findings, the city of Cold Bay discharges wastewater into Cold Bay, where there are Northern sea otter populations that are predicted to be highly susceptible to the virus. Beluga whales are also predicted to have high susceptibility and they can be found in Bristol Bay near Naknek, a city which relies only on lagoon treatment prior to the discharge of wastewater effluent; the city of Dillingham discharges wastewater into the Nushagak River where beluga whales are found. In Palmer, wastewater effluent flows into the Talkeetna River, which is a tributary to the Susitna River and home to two species predicted to have high susceptibility, beluga whales and harbor seals, the authors added.
Based on these results, the researchers predicted that there was likely a significant risk to sea mammals across the globe, especially where less-adequate treatment facilities and high population densities may lead to greater wastewater contamination.
“Given the proximity of marine animals to high-risk environments where viral spill over is likely, we must act with foresight to protect marine mammal species predicted to be at risk and mitigate the environmental impact of the COVID-19 pandemic,” the researchers concluded.
The authors reported that they had no disclosures.
SOURCE: Mathavarajah S et al. Sci Total Environ. 2020 Oct 29. doi: 10.1016/j.scitotenv.2020.143346.
Zoonoses are no respecter of biological boundaries and are notorious for crossing genus and even higher taxonomic boundaries. SARS-CoV-2 is no exception, the current outbreak most probably having originated in bats, a common source of human-affecting zoonoses throughout history. But it is not a one-way street, and the virus has been shown to spread from infected humans to a variety of other land mammals, including our domesticated animals and kept zoo species.
A recent troubling report, however, has indicated that sea mammals may be part of a next wave of likely candidates for infection, put at risk by the current human pandemic and environmental degradation on a global scale, according to a the results of a genomic analysis of four major groups of sea mammals.
Researchers Sabateeshan Mathavarajah and colleagues from Dalhousie University, Halifax, N.S., examined the sequences of the ACE2 receptors in the various marine mammal species. The ACE2 receptor has recently been identified as the SARS-CoV-2 receptor, which allows for infection.
The researchers examined genomic databases of the marine species to determine if their ACE2 receptor sequences indicated the potential for high, medium, or low susceptibility to infection, as reported in Science of the Total Environment. Database analysis was performed for four groups: Cetacea (whales and dolphins), Pinnepidia (seals), Sirenia (sea cows), and Fissipedia (sea otters and polar bears).
The researchers defined susceptibility values based on comparable binding with the receptor and came up with the following subgroups: higher than human, high (resembles human ACE2), medium (resembles cat ACE2), and low (resembles dog ACE2). It has yet to be established if these marine mammals actually are infected with SARS-CoV-2 and what the impact of such an infection might have on animal health or humans who come in contact with infected animals.
They also cross-referenced for the level of species endangerment and with maps of potential wastewater contamination for certain areas that species came in contact with, using Alaska as the model.
Populations in danger
The researchers found 15 species that are already at risk globally that fall under the categories of near threatened, vulnerable, endangered, and critically endangered that were predicted to be medium to higher susceptibility to the SARS-CoV-2 virus than humans. Cross infection is of particular concern because other coronaviruses have been shown to have severe and lethal effects among many of these species.
Among the potentially impacted species were the near threatened–status Antarctic Mink whale and the stellar sea lion; the vulnerable sperm whale, northern fur seal, and Atlantic walrus; the endangered northern and southern sea otters, the North Pacific right whale, and the Amazon River dolphin; and the critically threatened Baiji and Vaquita dolphin species.
Pollution risks
In Alaska, as of Aug. 7th, 2020, there were 4,221 confirmed cases of COVID-19 and this number continues to rise, according to the researchers. Since there is a diversity of marine mammals in Alaska and their populations are well documented, they compared this information with available data on the wastewater treatment plants in the state. They were thus able to determine the potential geographic locations and species at high risk for transmission of SARS-CoV-2 via wastewater effluent.
Among their findings, the city of Cold Bay discharges wastewater into Cold Bay, where there are Northern sea otter populations that are predicted to be highly susceptible to the virus. Beluga whales are also predicted to have high susceptibility and they can be found in Bristol Bay near Naknek, a city which relies only on lagoon treatment prior to the discharge of wastewater effluent; the city of Dillingham discharges wastewater into the Nushagak River where beluga whales are found. In Palmer, wastewater effluent flows into the Talkeetna River, which is a tributary to the Susitna River and home to two species predicted to have high susceptibility, beluga whales and harbor seals, the authors added.
Based on these results, the researchers predicted that there was likely a significant risk to sea mammals across the globe, especially where less-adequate treatment facilities and high population densities may lead to greater wastewater contamination.
“Given the proximity of marine animals to high-risk environments where viral spill over is likely, we must act with foresight to protect marine mammal species predicted to be at risk and mitigate the environmental impact of the COVID-19 pandemic,” the researchers concluded.
The authors reported that they had no disclosures.
SOURCE: Mathavarajah S et al. Sci Total Environ. 2020 Oct 29. doi: 10.1016/j.scitotenv.2020.143346.
Zoonoses are no respecter of biological boundaries and are notorious for crossing genus and even higher taxonomic boundaries. SARS-CoV-2 is no exception, the current outbreak most probably having originated in bats, a common source of human-affecting zoonoses throughout history. But it is not a one-way street, and the virus has been shown to spread from infected humans to a variety of other land mammals, including our domesticated animals and kept zoo species.
A recent troubling report, however, has indicated that sea mammals may be part of a next wave of likely candidates for infection, put at risk by the current human pandemic and environmental degradation on a global scale, according to a the results of a genomic analysis of four major groups of sea mammals.
Researchers Sabateeshan Mathavarajah and colleagues from Dalhousie University, Halifax, N.S., examined the sequences of the ACE2 receptors in the various marine mammal species. The ACE2 receptor has recently been identified as the SARS-CoV-2 receptor, which allows for infection.
The researchers examined genomic databases of the marine species to determine if their ACE2 receptor sequences indicated the potential for high, medium, or low susceptibility to infection, as reported in Science of the Total Environment. Database analysis was performed for four groups: Cetacea (whales and dolphins), Pinnepidia (seals), Sirenia (sea cows), and Fissipedia (sea otters and polar bears).
The researchers defined susceptibility values based on comparable binding with the receptor and came up with the following subgroups: higher than human, high (resembles human ACE2), medium (resembles cat ACE2), and low (resembles dog ACE2). It has yet to be established if these marine mammals actually are infected with SARS-CoV-2 and what the impact of such an infection might have on animal health or humans who come in contact with infected animals.
They also cross-referenced for the level of species endangerment and with maps of potential wastewater contamination for certain areas that species came in contact with, using Alaska as the model.
Populations in danger
The researchers found 15 species that are already at risk globally that fall under the categories of near threatened, vulnerable, endangered, and critically endangered that were predicted to be medium to higher susceptibility to the SARS-CoV-2 virus than humans. Cross infection is of particular concern because other coronaviruses have been shown to have severe and lethal effects among many of these species.
Among the potentially impacted species were the near threatened–status Antarctic Mink whale and the stellar sea lion; the vulnerable sperm whale, northern fur seal, and Atlantic walrus; the endangered northern and southern sea otters, the North Pacific right whale, and the Amazon River dolphin; and the critically threatened Baiji and Vaquita dolphin species.
Pollution risks
In Alaska, as of Aug. 7th, 2020, there were 4,221 confirmed cases of COVID-19 and this number continues to rise, according to the researchers. Since there is a diversity of marine mammals in Alaska and their populations are well documented, they compared this information with available data on the wastewater treatment plants in the state. They were thus able to determine the potential geographic locations and species at high risk for transmission of SARS-CoV-2 via wastewater effluent.
Among their findings, the city of Cold Bay discharges wastewater into Cold Bay, where there are Northern sea otter populations that are predicted to be highly susceptible to the virus. Beluga whales are also predicted to have high susceptibility and they can be found in Bristol Bay near Naknek, a city which relies only on lagoon treatment prior to the discharge of wastewater effluent; the city of Dillingham discharges wastewater into the Nushagak River where beluga whales are found. In Palmer, wastewater effluent flows into the Talkeetna River, which is a tributary to the Susitna River and home to two species predicted to have high susceptibility, beluga whales and harbor seals, the authors added.
Based on these results, the researchers predicted that there was likely a significant risk to sea mammals across the globe, especially where less-adequate treatment facilities and high population densities may lead to greater wastewater contamination.
“Given the proximity of marine animals to high-risk environments where viral spill over is likely, we must act with foresight to protect marine mammal species predicted to be at risk and mitigate the environmental impact of the COVID-19 pandemic,” the researchers concluded.
The authors reported that they had no disclosures.
SOURCE: Mathavarajah S et al. Sci Total Environ. 2020 Oct 29. doi: 10.1016/j.scitotenv.2020.143346.
FROM SCIENCE OF THE TOTAL ENVIRONMENT