User login
More than 30 experts question validity of serotonin/depression study
The authors of the article, however, stand by their conclusion.
“The methodology doesn’t conform to a conventional umbrella review,” said the commentary’s lead author, Sameer Jauhar, MD, PhD, first author of the commentary criticizing the review, which was published online in Molecular Psychiatry.
In addition, preeminent psychiatrist David J. Nutt, MD, PhD, Edmond J. Safra Professor of Neuropsychopharmacology, Imperial College London, is calling for the review to be retracted. In an interview with The Daily Mail, he said the article is “full of flaws and it should never have been published in the first place. Yet it has been frequently cited and people believe it is true. It’s essentially misinformation. That’s why I’m calling on the journal to retract it.” Dr. Nutt is one of the authors of the published commentary.
‘No convincing evidence’
Led by Joanna Moncrieff, MD, professor of clinical and social psychiatry, University College London, the authors analyzed systematic reviews and meta-analyses to determine whether low serotonin levels are, in fact, associated with depression.
Of 361 potential studies, 17 were selected for the review, including meta-analyses, systematic reviews, and a genetic association study.
The review included examinations of 5-HT and its metabolite 5-hydroxyindoleacetic acid (5-HIAA) in “body fluids,” 5HT1A receptor and serotonin transporter protein (SERT) availability in imaging and postmortem studies, investigations of SERT gene polymorphisms, interactions between SERT and stress in depression, and effects of tryptophan depletion on mood.
The tryptophan hypothesis suggests depression occurs through tryptophan depletion, which lowers available serotonin. According to the review, two crossover studies of patients with depression who were currently receiving or had recently received antidepressant treatment did not show substantial effects of depletion, and data from studies involving volunteers largely showed no effect.
Ultimately, Dr. Moncrieff and colleagues concluded that “there is no convincing evidence that depression is associated with, or caused by, lower serotonin concentrations or activity.”
‘Unconventional, odd’ methodology
However, Dr. Jauhar and the commentary’s coauthors disagree with the study’s conclusion. The researchers claim that “we don’t see depression symptoms in healthy volunteers when given tryptophan depletion; everyone knows that and agrees with that; it’s only in people vulnerable to depression who will have it.”
Furthermore, he said, the study’s conclusion does not consider that experimental medicine studies of tryptophan depletion are difficult to conduct. “You’re not going to have huge sample sizes as you would in a genetic study or big epidemiological studies.
Dr. Jauhar said he found it “unconventional” and “odd” that the review included individual tryptophan depletion studies that were not in the prespecified protocol.
For studies involving molecular imaging, Dr. Jauhar said the review’s inferences were “simplistic” and the review authors are “basically shaping the argument” to fit their desired narrative.
He also noted factual errors in the review. “They make a mistake when they talk about serotonin transporter imaging; they say there are no consistent findings across studies when, in fact, there are.”
With both tryptophan depletion and molecular imaging studies, the review “glosses over findings” from the original studies, said Dr. Jauhar.
For tryptophan depletion, “a more accurate, constructive conclusion would be that acute tryptophan depletion and decreased plasma tryptophan in depression indicate a role for 5-HT in those vulnerable to or suffering from depression, and that molecular imaging suggests the system is perturbed,” the commentators wrote.
“The proven efficacy of SSRIs in a proportion of people with depression lends credibility to this position,” they added.
Dr. Jauhar also took issue with criteria for certainty of finding of these and other studies used in the review. “If you’re setting the criteria yourself, it’s arbitrary.”
No new data
An umbrella review is supposed to be of the highest quality and should entail “taking out the studies and analysing them yourself,” but here, “all they have done is put a synthesis forward of other people’s reviews, so essentially there’s no new data there,” said Dr. Jauhar.
And sometimes the review’s findings differ from the original research. “When you have people who haven’t conducted original research themselves quoting someone else’s work and ignoring what those people say, we’re all in trouble,” said Dr. Jauhar.
In an additional commentary also published in Molecular Psychiatry, Jacob Pade Ramsøe Jacobsen, Evecxia Therapeutics, Durham, N.C., also criticized the review by Dr. Moncrieff and colleagues.
Its authors appear unfamiliar with serotonin biology and pharmacology, Dr. Jacobsen wrote.
“The review contains factual errors, makes conclusions serotonin neurobiology may not support, and quotes the cited literature in a selective manner,” he added.
“Most troubling, they misinterpret some data reviewed and intimate that serotonin reuptake inhibitor antidepressants, e.g., SSRIs, may decrease, rather than increase, serotonin function.”
If accepted by general practitioners and the public, the review’s conclusions “could lead to reduced use of antidepressants among patients in need and increased morbidity related to depression.”
Dr. Moncrieff pushes back
Responding to the torrent of criticism of her study, Dr. Moncrieff told this news organization via email that they stand by the review, adding that Dr. Jauhar and others “don’t want to let the cat out of the bag” that there’s no good evidence to support the hypothesis that low serotonin causes depression because it challenges antidepressant use.
“The idea that antidepressants work by correcting an underlying chemical imbalance or serotonin abnormality has led research up a blind alley and meant scientists have not taken the harmful effects of these drugs seriously enough.”
These critics, she added, “want business as usual – which means people will continue to be misinformed and exposed to harmful effects of drugs that have minimal and uncertain benefits.”
In a letter to the editor of Molecular Psychiatry, Dr. Moncrieff and her fellow authors maintain that they used approved and well-accepted methods for the umbrella review, including preregistering the protocol and using recommended search methods and quality assessments, and that they did not miss certain studies, as has been claimed.
In her blog, Dr. Moncrieff wrote that the “marginal differences between antidepressants and placebo that are apparent in clinical trials are likely to be produced by alternative, more plausible mechanisms like the emotional blunting effects of the drugs or by amplified placebo effects, rather than by targeting underlying biological mechanisms (since these have not been demonstrated).”
It also highlights “how we don’t know what antidepressants do to the brain exactly, which is a cause for concern,” she adds.
Dr. Jauhar has received honoraria for nonpromotional educational talks on antipsychotics from Janssen, Sunovion, and Lundbeck and on causes of schizophrenia for Boehringer-Ingelheim. He has also received honoraria for consulting on antipsychotics for LB Pharmaceuticals. He sits on Council for the British Association for Psychopharmacology and was a recent panel member for the Wellcome Trust.
A version of this article originally appeared on Medscape.com.
The authors of the article, however, stand by their conclusion.
“The methodology doesn’t conform to a conventional umbrella review,” said the commentary’s lead author, Sameer Jauhar, MD, PhD, first author of the commentary criticizing the review, which was published online in Molecular Psychiatry.
In addition, preeminent psychiatrist David J. Nutt, MD, PhD, Edmond J. Safra Professor of Neuropsychopharmacology, Imperial College London, is calling for the review to be retracted. In an interview with The Daily Mail, he said the article is “full of flaws and it should never have been published in the first place. Yet it has been frequently cited and people believe it is true. It’s essentially misinformation. That’s why I’m calling on the journal to retract it.” Dr. Nutt is one of the authors of the published commentary.
‘No convincing evidence’
Led by Joanna Moncrieff, MD, professor of clinical and social psychiatry, University College London, the authors analyzed systematic reviews and meta-analyses to determine whether low serotonin levels are, in fact, associated with depression.
Of 361 potential studies, 17 were selected for the review, including meta-analyses, systematic reviews, and a genetic association study.
The review included examinations of 5-HT and its metabolite 5-hydroxyindoleacetic acid (5-HIAA) in “body fluids,” 5HT1A receptor and serotonin transporter protein (SERT) availability in imaging and postmortem studies, investigations of SERT gene polymorphisms, interactions between SERT and stress in depression, and effects of tryptophan depletion on mood.
The tryptophan hypothesis suggests depression occurs through tryptophan depletion, which lowers available serotonin. According to the review, two crossover studies of patients with depression who were currently receiving or had recently received antidepressant treatment did not show substantial effects of depletion, and data from studies involving volunteers largely showed no effect.
Ultimately, Dr. Moncrieff and colleagues concluded that “there is no convincing evidence that depression is associated with, or caused by, lower serotonin concentrations or activity.”
‘Unconventional, odd’ methodology
However, Dr. Jauhar and the commentary’s coauthors disagree with the study’s conclusion. The researchers claim that “we don’t see depression symptoms in healthy volunteers when given tryptophan depletion; everyone knows that and agrees with that; it’s only in people vulnerable to depression who will have it.”
Furthermore, he said, the study’s conclusion does not consider that experimental medicine studies of tryptophan depletion are difficult to conduct. “You’re not going to have huge sample sizes as you would in a genetic study or big epidemiological studies.
Dr. Jauhar said he found it “unconventional” and “odd” that the review included individual tryptophan depletion studies that were not in the prespecified protocol.
For studies involving molecular imaging, Dr. Jauhar said the review’s inferences were “simplistic” and the review authors are “basically shaping the argument” to fit their desired narrative.
He also noted factual errors in the review. “They make a mistake when they talk about serotonin transporter imaging; they say there are no consistent findings across studies when, in fact, there are.”
With both tryptophan depletion and molecular imaging studies, the review “glosses over findings” from the original studies, said Dr. Jauhar.
For tryptophan depletion, “a more accurate, constructive conclusion would be that acute tryptophan depletion and decreased plasma tryptophan in depression indicate a role for 5-HT in those vulnerable to or suffering from depression, and that molecular imaging suggests the system is perturbed,” the commentators wrote.
“The proven efficacy of SSRIs in a proportion of people with depression lends credibility to this position,” they added.
Dr. Jauhar also took issue with criteria for certainty of finding of these and other studies used in the review. “If you’re setting the criteria yourself, it’s arbitrary.”
No new data
An umbrella review is supposed to be of the highest quality and should entail “taking out the studies and analysing them yourself,” but here, “all they have done is put a synthesis forward of other people’s reviews, so essentially there’s no new data there,” said Dr. Jauhar.
And sometimes the review’s findings differ from the original research. “When you have people who haven’t conducted original research themselves quoting someone else’s work and ignoring what those people say, we’re all in trouble,” said Dr. Jauhar.
In an additional commentary also published in Molecular Psychiatry, Jacob Pade Ramsøe Jacobsen, Evecxia Therapeutics, Durham, N.C., also criticized the review by Dr. Moncrieff and colleagues.
Its authors appear unfamiliar with serotonin biology and pharmacology, Dr. Jacobsen wrote.
“The review contains factual errors, makes conclusions serotonin neurobiology may not support, and quotes the cited literature in a selective manner,” he added.
“Most troubling, they misinterpret some data reviewed and intimate that serotonin reuptake inhibitor antidepressants, e.g., SSRIs, may decrease, rather than increase, serotonin function.”
If accepted by general practitioners and the public, the review’s conclusions “could lead to reduced use of antidepressants among patients in need and increased morbidity related to depression.”
Dr. Moncrieff pushes back
Responding to the torrent of criticism of her study, Dr. Moncrieff told this news organization via email that they stand by the review, adding that Dr. Jauhar and others “don’t want to let the cat out of the bag” that there’s no good evidence to support the hypothesis that low serotonin causes depression because it challenges antidepressant use.
“The idea that antidepressants work by correcting an underlying chemical imbalance or serotonin abnormality has led research up a blind alley and meant scientists have not taken the harmful effects of these drugs seriously enough.”
These critics, she added, “want business as usual – which means people will continue to be misinformed and exposed to harmful effects of drugs that have minimal and uncertain benefits.”
In a letter to the editor of Molecular Psychiatry, Dr. Moncrieff and her fellow authors maintain that they used approved and well-accepted methods for the umbrella review, including preregistering the protocol and using recommended search methods and quality assessments, and that they did not miss certain studies, as has been claimed.
In her blog, Dr. Moncrieff wrote that the “marginal differences between antidepressants and placebo that are apparent in clinical trials are likely to be produced by alternative, more plausible mechanisms like the emotional blunting effects of the drugs or by amplified placebo effects, rather than by targeting underlying biological mechanisms (since these have not been demonstrated).”
It also highlights “how we don’t know what antidepressants do to the brain exactly, which is a cause for concern,” she adds.
Dr. Jauhar has received honoraria for nonpromotional educational talks on antipsychotics from Janssen, Sunovion, and Lundbeck and on causes of schizophrenia for Boehringer-Ingelheim. He has also received honoraria for consulting on antipsychotics for LB Pharmaceuticals. He sits on Council for the British Association for Psychopharmacology and was a recent panel member for the Wellcome Trust.
A version of this article originally appeared on Medscape.com.
The authors of the article, however, stand by their conclusion.
“The methodology doesn’t conform to a conventional umbrella review,” said the commentary’s lead author, Sameer Jauhar, MD, PhD, first author of the commentary criticizing the review, which was published online in Molecular Psychiatry.
In addition, preeminent psychiatrist David J. Nutt, MD, PhD, Edmond J. Safra Professor of Neuropsychopharmacology, Imperial College London, is calling for the review to be retracted. In an interview with The Daily Mail, he said the article is “full of flaws and it should never have been published in the first place. Yet it has been frequently cited and people believe it is true. It’s essentially misinformation. That’s why I’m calling on the journal to retract it.” Dr. Nutt is one of the authors of the published commentary.
‘No convincing evidence’
Led by Joanna Moncrieff, MD, professor of clinical and social psychiatry, University College London, the authors analyzed systematic reviews and meta-analyses to determine whether low serotonin levels are, in fact, associated with depression.
Of 361 potential studies, 17 were selected for the review, including meta-analyses, systematic reviews, and a genetic association study.
The review included examinations of 5-HT and its metabolite 5-hydroxyindoleacetic acid (5-HIAA) in “body fluids,” 5HT1A receptor and serotonin transporter protein (SERT) availability in imaging and postmortem studies, investigations of SERT gene polymorphisms, interactions between SERT and stress in depression, and effects of tryptophan depletion on mood.
The tryptophan hypothesis suggests depression occurs through tryptophan depletion, which lowers available serotonin. According to the review, two crossover studies of patients with depression who were currently receiving or had recently received antidepressant treatment did not show substantial effects of depletion, and data from studies involving volunteers largely showed no effect.
Ultimately, Dr. Moncrieff and colleagues concluded that “there is no convincing evidence that depression is associated with, or caused by, lower serotonin concentrations or activity.”
‘Unconventional, odd’ methodology
However, Dr. Jauhar and the commentary’s coauthors disagree with the study’s conclusion. The researchers claim that “we don’t see depression symptoms in healthy volunteers when given tryptophan depletion; everyone knows that and agrees with that; it’s only in people vulnerable to depression who will have it.”
Furthermore, he said, the study’s conclusion does not consider that experimental medicine studies of tryptophan depletion are difficult to conduct. “You’re not going to have huge sample sizes as you would in a genetic study or big epidemiological studies.
Dr. Jauhar said he found it “unconventional” and “odd” that the review included individual tryptophan depletion studies that were not in the prespecified protocol.
For studies involving molecular imaging, Dr. Jauhar said the review’s inferences were “simplistic” and the review authors are “basically shaping the argument” to fit their desired narrative.
He also noted factual errors in the review. “They make a mistake when they talk about serotonin transporter imaging; they say there are no consistent findings across studies when, in fact, there are.”
With both tryptophan depletion and molecular imaging studies, the review “glosses over findings” from the original studies, said Dr. Jauhar.
For tryptophan depletion, “a more accurate, constructive conclusion would be that acute tryptophan depletion and decreased plasma tryptophan in depression indicate a role for 5-HT in those vulnerable to or suffering from depression, and that molecular imaging suggests the system is perturbed,” the commentators wrote.
“The proven efficacy of SSRIs in a proportion of people with depression lends credibility to this position,” they added.
Dr. Jauhar also took issue with criteria for certainty of finding of these and other studies used in the review. “If you’re setting the criteria yourself, it’s arbitrary.”
No new data
An umbrella review is supposed to be of the highest quality and should entail “taking out the studies and analysing them yourself,” but here, “all they have done is put a synthesis forward of other people’s reviews, so essentially there’s no new data there,” said Dr. Jauhar.
And sometimes the review’s findings differ from the original research. “When you have people who haven’t conducted original research themselves quoting someone else’s work and ignoring what those people say, we’re all in trouble,” said Dr. Jauhar.
In an additional commentary also published in Molecular Psychiatry, Jacob Pade Ramsøe Jacobsen, Evecxia Therapeutics, Durham, N.C., also criticized the review by Dr. Moncrieff and colleagues.
Its authors appear unfamiliar with serotonin biology and pharmacology, Dr. Jacobsen wrote.
“The review contains factual errors, makes conclusions serotonin neurobiology may not support, and quotes the cited literature in a selective manner,” he added.
“Most troubling, they misinterpret some data reviewed and intimate that serotonin reuptake inhibitor antidepressants, e.g., SSRIs, may decrease, rather than increase, serotonin function.”
If accepted by general practitioners and the public, the review’s conclusions “could lead to reduced use of antidepressants among patients in need and increased morbidity related to depression.”
Dr. Moncrieff pushes back
Responding to the torrent of criticism of her study, Dr. Moncrieff told this news organization via email that they stand by the review, adding that Dr. Jauhar and others “don’t want to let the cat out of the bag” that there’s no good evidence to support the hypothesis that low serotonin causes depression because it challenges antidepressant use.
“The idea that antidepressants work by correcting an underlying chemical imbalance or serotonin abnormality has led research up a blind alley and meant scientists have not taken the harmful effects of these drugs seriously enough.”
These critics, she added, “want business as usual – which means people will continue to be misinformed and exposed to harmful effects of drugs that have minimal and uncertain benefits.”
In a letter to the editor of Molecular Psychiatry, Dr. Moncrieff and her fellow authors maintain that they used approved and well-accepted methods for the umbrella review, including preregistering the protocol and using recommended search methods and quality assessments, and that they did not miss certain studies, as has been claimed.
In her blog, Dr. Moncrieff wrote that the “marginal differences between antidepressants and placebo that are apparent in clinical trials are likely to be produced by alternative, more plausible mechanisms like the emotional blunting effects of the drugs or by amplified placebo effects, rather than by targeting underlying biological mechanisms (since these have not been demonstrated).”
It also highlights “how we don’t know what antidepressants do to the brain exactly, which is a cause for concern,” she adds.
Dr. Jauhar has received honoraria for nonpromotional educational talks on antipsychotics from Janssen, Sunovion, and Lundbeck and on causes of schizophrenia for Boehringer-Ingelheim. He has also received honoraria for consulting on antipsychotics for LB Pharmaceuticals. He sits on Council for the British Association for Psychopharmacology and was a recent panel member for the Wellcome Trust.
A version of this article originally appeared on Medscape.com.
Multidisciplinary Approach to Stage III NSCLC
Stage III non–small cell lung cancer, depending on tumor stage and histology, may be managed in multiple ways. Because of the range of treatments available and the numerous factors that inform the optimal approach, management by a multidisciplinary team is considered advantageous.
Weekly meetings of the multidisciplinary team facilitate the efficient review of tumor histology and molecular status, surgical and radiation options, and pre- and post-surgery care for individual patients.
Pulmonologist Dr Anne Gonzalez, from McGill University Health Centre; thoracic oncologist Dr Jyoti D. Patel, of Northwestern University; and thoracic surgeon Dr John Howington, from Virginia Mason Franciscan Health, discuss the vital roles that specialists play in the coordinated treatment of a patient. The panelists also consider factors in treatment selection and how multidisciplinary care is managed at each of their institutions.
--
Anne V. Gonzalez, MD, MSc, FCCP, Associate Professor of Medicine, McGill University Health Centre, Montreal, Quebec, Canada
Anne V. Gonzalez, MD, MSc, FCCP, has disclosed the following relevant financial relationships:
Received research grant from: Lung Cancer Canada
DSMB for: Laurent Pharmaceuticals; GSK; Idorsia; Janssen; Pfizer
Jyoti D. Patel, MD, Professor of Medicine, Feinberg School of Medicine, Northwestern University, Chicago, Illinois
Jyoti Patel, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Astra Zeneca; AnHeart; Takeda; Lilly
John Howington, MD, MBA, FCCP, Thoracic Surgeon, Virginia Mason Franciscan Health, Franciscan Cardiothoracic Surgery Associates at Saint Michael, Silverdale, Washington
John Howington, MD, MBA, FCCP, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: President Designate of American College of Chest Physicians
Stage III non–small cell lung cancer, depending on tumor stage and histology, may be managed in multiple ways. Because of the range of treatments available and the numerous factors that inform the optimal approach, management by a multidisciplinary team is considered advantageous.
Weekly meetings of the multidisciplinary team facilitate the efficient review of tumor histology and molecular status, surgical and radiation options, and pre- and post-surgery care for individual patients.
Pulmonologist Dr Anne Gonzalez, from McGill University Health Centre; thoracic oncologist Dr Jyoti D. Patel, of Northwestern University; and thoracic surgeon Dr John Howington, from Virginia Mason Franciscan Health, discuss the vital roles that specialists play in the coordinated treatment of a patient. The panelists also consider factors in treatment selection and how multidisciplinary care is managed at each of their institutions.
--
Anne V. Gonzalez, MD, MSc, FCCP, Associate Professor of Medicine, McGill University Health Centre, Montreal, Quebec, Canada
Anne V. Gonzalez, MD, MSc, FCCP, has disclosed the following relevant financial relationships:
Received research grant from: Lung Cancer Canada
DSMB for: Laurent Pharmaceuticals; GSK; Idorsia; Janssen; Pfizer
Jyoti D. Patel, MD, Professor of Medicine, Feinberg School of Medicine, Northwestern University, Chicago, Illinois
Jyoti Patel, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Astra Zeneca; AnHeart; Takeda; Lilly
John Howington, MD, MBA, FCCP, Thoracic Surgeon, Virginia Mason Franciscan Health, Franciscan Cardiothoracic Surgery Associates at Saint Michael, Silverdale, Washington
John Howington, MD, MBA, FCCP, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: President Designate of American College of Chest Physicians
Stage III non–small cell lung cancer, depending on tumor stage and histology, may be managed in multiple ways. Because of the range of treatments available and the numerous factors that inform the optimal approach, management by a multidisciplinary team is considered advantageous.
Weekly meetings of the multidisciplinary team facilitate the efficient review of tumor histology and molecular status, surgical and radiation options, and pre- and post-surgery care for individual patients.
Pulmonologist Dr Anne Gonzalez, from McGill University Health Centre; thoracic oncologist Dr Jyoti D. Patel, of Northwestern University; and thoracic surgeon Dr John Howington, from Virginia Mason Franciscan Health, discuss the vital roles that specialists play in the coordinated treatment of a patient. The panelists also consider factors in treatment selection and how multidisciplinary care is managed at each of their institutions.
--
Anne V. Gonzalez, MD, MSc, FCCP, Associate Professor of Medicine, McGill University Health Centre, Montreal, Quebec, Canada
Anne V. Gonzalez, MD, MSc, FCCP, has disclosed the following relevant financial relationships:
Received research grant from: Lung Cancer Canada
DSMB for: Laurent Pharmaceuticals; GSK; Idorsia; Janssen; Pfizer
Jyoti D. Patel, MD, Professor of Medicine, Feinberg School of Medicine, Northwestern University, Chicago, Illinois
Jyoti Patel, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: Astra Zeneca; AnHeart; Takeda; Lilly
John Howington, MD, MBA, FCCP, Thoracic Surgeon, Virginia Mason Franciscan Health, Franciscan Cardiothoracic Surgery Associates at Saint Michael, Silverdale, Washington
John Howington, MD, MBA, FCCP, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: President Designate of American College of Chest Physicians

Study points to need to improve identification of culprit in SJS/TEN cases
demonstrated.
“Prompt identification and discontinuation of a culprit drug is critical to improving patient outcomes and preventing recurrence,” researchers led by Sherrie J. Divito, MD, PhD, of the department of dermatology at Brigham and Women’s Hospital, Boston, wrote in the study, which was published online in JAMA Dermatology. “Identification is difficult because there is no laboratory test or other criterion standard (in the absence of rechallenge) to identify a culprit drug, and patients take on average six medications at the time of their reaction. Consequently, many patients may be labeled as allergic to multiple agents.”
Although failing to identify the culprit drug can have severe consequences, they added, “overlabeling” (labeling a patient as allergic to a drug or drugs that they can tolerate safely) “is not insignificant.” As a result of overlabeling, “the patient may receive a less efficacious, more toxic, and/or more expensive agent than necessary, and in some cases may be left without treatment for their underlying disease.”
To evaluate the outcomes of patients’ allergy lists, current approaches to identify culprit drugs such as the Algorithm for Drug Causality for Epidermal Necrolysis (ALDEN), which was published in 2010, and potential methods of improving culprit drug identification, the researchers performed a retrospective cohort study of 48 patients at Brigham and Women’s Hospital and Massachusetts General Hospital, with clinically and histologically confirmed cases of SJS/TEN between January 2000 and July 2018. Of the 48 patients, 26 had SJS/TEN overlap and 22 had TEN. Their median age at diagnosis was 40 years; 60.4% were female; and 52.1% were white, 12.5% were Black, 10.4% were Hispanic, and 8.3% were Asian. They took a median of 6.5 drugs in the 3 months prior to disease onset.
The researchers observed that all patients had at least one drug labeled as an allergy. A single culprit drug was labeled in 17 cases, but physicians communicated certainty in only 7 of those cases. Among all 48 patients, 104 drugs were labeled as allergies.
To identify a culprit drug, physicians appeared to mainly rely on two factors: drug notoriety and timing of exposure, compared with the onset of SJS/TEN. “Identifying high-risk medications seemed heuristic, with one or more drugs in question noted in the record as a common culprit without reference to published or vetted data regarding risk,” the researchers wrote. “Regarding timing, drug charts when present in medical records were incomplete, as they focused predominantly on high-notoriety drugs.”
In other findings, ALDEN scoring was discordant with physician-labeled lists in 28 cases. It labeled an additional 9 drugs missed by physicians and scored 43 drugs labeled as allergens by physicians as “unlikely.” The researchers also reported that 20 cases could have potentially benefited from human leukocyte antigen testing.
Their results “underscore the need for a laboratory test to identify culprit drugs,” but without such a test, “a systematic unbiased approach, such as ALDEN or the RegiSCAR database, with possibly HLA testing, should be considered to ensure the true culprit drug is not missed and exonerate as many nonculprit drugs as possible,” Dr. Divito and colleagues concluded.
They acknowledged certain limitations of the analysis, including its retrospective design and that many cases predated research advances in the topic area that took place during the 18-year study period.
Karl Saardi, MD, director of the inpatient dermatology service at George Washington University Hospital, Washington, who was asked to comment on the study, said that the findings “are consistent with clinical practice in that drug causality is usually determined by ‘gestalt’ rather than objective tools like the ALDEN algorithm.”
“The main limitation is the small size, which suggests the study sites are low-volume centers for SJS/TEN. The fact that the ALDEN score wasn’t developed until 2010 means that all the cases included prior to 2010 would not have applied the ALDEN algorithm, so I think the metric about how infrequently ALDEN was applied is not very meaningful.”
Still, he said that he was “surprised” by the number of medications that were added as allergies based on clinical impression, “and I’m glad this article does cast some light on the issue. In my experience, beta-lactam antibiotics are often – incorrectly – deemed to be the cause of SJS/TEN when further review of the patient’s medication history clearly shows they have tolerated these drugs multiple times in the past.”
Since 2000, he added, “our understanding of SJS/TEN has grown substantially including the identification of MIRM [mycoplasma-induced rash and mucositis]/RIME [reactive infections mucocutaneous eruptions] and GBFDE [generalized bullous fixed drug eruption] as mimickers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was also asked to comment on the study, agreed that a limitation of the study is that it partially preceded development of the unbiased approaches to determining the cause of a medication reaction, such as the ALDEN system. “A strength of this study is the examination of heuristics in dermatology and how they relate to patient safety,” she added.
The study was funded by grants from the National Institutes of Health, a Dermatology Foundation Diversity Research Supplement Award, and by the German Research Foundation. Dr. Divito reported receiving personal fees from Adaptimmune and MEI Pharma and a provisional patent issued from Brigham and Women’s Hospital outside the submitted work. Neither Dr. Saardi nor Dr. Ko reported having relevant disclosures.
demonstrated.
“Prompt identification and discontinuation of a culprit drug is critical to improving patient outcomes and preventing recurrence,” researchers led by Sherrie J. Divito, MD, PhD, of the department of dermatology at Brigham and Women’s Hospital, Boston, wrote in the study, which was published online in JAMA Dermatology. “Identification is difficult because there is no laboratory test or other criterion standard (in the absence of rechallenge) to identify a culprit drug, and patients take on average six medications at the time of their reaction. Consequently, many patients may be labeled as allergic to multiple agents.”
Although failing to identify the culprit drug can have severe consequences, they added, “overlabeling” (labeling a patient as allergic to a drug or drugs that they can tolerate safely) “is not insignificant.” As a result of overlabeling, “the patient may receive a less efficacious, more toxic, and/or more expensive agent than necessary, and in some cases may be left without treatment for their underlying disease.”
To evaluate the outcomes of patients’ allergy lists, current approaches to identify culprit drugs such as the Algorithm for Drug Causality for Epidermal Necrolysis (ALDEN), which was published in 2010, and potential methods of improving culprit drug identification, the researchers performed a retrospective cohort study of 48 patients at Brigham and Women’s Hospital and Massachusetts General Hospital, with clinically and histologically confirmed cases of SJS/TEN between January 2000 and July 2018. Of the 48 patients, 26 had SJS/TEN overlap and 22 had TEN. Their median age at diagnosis was 40 years; 60.4% were female; and 52.1% were white, 12.5% were Black, 10.4% were Hispanic, and 8.3% were Asian. They took a median of 6.5 drugs in the 3 months prior to disease onset.
The researchers observed that all patients had at least one drug labeled as an allergy. A single culprit drug was labeled in 17 cases, but physicians communicated certainty in only 7 of those cases. Among all 48 patients, 104 drugs were labeled as allergies.
To identify a culprit drug, physicians appeared to mainly rely on two factors: drug notoriety and timing of exposure, compared with the onset of SJS/TEN. “Identifying high-risk medications seemed heuristic, with one or more drugs in question noted in the record as a common culprit without reference to published or vetted data regarding risk,” the researchers wrote. “Regarding timing, drug charts when present in medical records were incomplete, as they focused predominantly on high-notoriety drugs.”
In other findings, ALDEN scoring was discordant with physician-labeled lists in 28 cases. It labeled an additional 9 drugs missed by physicians and scored 43 drugs labeled as allergens by physicians as “unlikely.” The researchers also reported that 20 cases could have potentially benefited from human leukocyte antigen testing.
Their results “underscore the need for a laboratory test to identify culprit drugs,” but without such a test, “a systematic unbiased approach, such as ALDEN or the RegiSCAR database, with possibly HLA testing, should be considered to ensure the true culprit drug is not missed and exonerate as many nonculprit drugs as possible,” Dr. Divito and colleagues concluded.
They acknowledged certain limitations of the analysis, including its retrospective design and that many cases predated research advances in the topic area that took place during the 18-year study period.
Karl Saardi, MD, director of the inpatient dermatology service at George Washington University Hospital, Washington, who was asked to comment on the study, said that the findings “are consistent with clinical practice in that drug causality is usually determined by ‘gestalt’ rather than objective tools like the ALDEN algorithm.”
“The main limitation is the small size, which suggests the study sites are low-volume centers for SJS/TEN. The fact that the ALDEN score wasn’t developed until 2010 means that all the cases included prior to 2010 would not have applied the ALDEN algorithm, so I think the metric about how infrequently ALDEN was applied is not very meaningful.”
Still, he said that he was “surprised” by the number of medications that were added as allergies based on clinical impression, “and I’m glad this article does cast some light on the issue. In my experience, beta-lactam antibiotics are often – incorrectly – deemed to be the cause of SJS/TEN when further review of the patient’s medication history clearly shows they have tolerated these drugs multiple times in the past.”
Since 2000, he added, “our understanding of SJS/TEN has grown substantially including the identification of MIRM [mycoplasma-induced rash and mucositis]/RIME [reactive infections mucocutaneous eruptions] and GBFDE [generalized bullous fixed drug eruption] as mimickers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was also asked to comment on the study, agreed that a limitation of the study is that it partially preceded development of the unbiased approaches to determining the cause of a medication reaction, such as the ALDEN system. “A strength of this study is the examination of heuristics in dermatology and how they relate to patient safety,” she added.
The study was funded by grants from the National Institutes of Health, a Dermatology Foundation Diversity Research Supplement Award, and by the German Research Foundation. Dr. Divito reported receiving personal fees from Adaptimmune and MEI Pharma and a provisional patent issued from Brigham and Women’s Hospital outside the submitted work. Neither Dr. Saardi nor Dr. Ko reported having relevant disclosures.
demonstrated.
“Prompt identification and discontinuation of a culprit drug is critical to improving patient outcomes and preventing recurrence,” researchers led by Sherrie J. Divito, MD, PhD, of the department of dermatology at Brigham and Women’s Hospital, Boston, wrote in the study, which was published online in JAMA Dermatology. “Identification is difficult because there is no laboratory test or other criterion standard (in the absence of rechallenge) to identify a culprit drug, and patients take on average six medications at the time of their reaction. Consequently, many patients may be labeled as allergic to multiple agents.”
Although failing to identify the culprit drug can have severe consequences, they added, “overlabeling” (labeling a patient as allergic to a drug or drugs that they can tolerate safely) “is not insignificant.” As a result of overlabeling, “the patient may receive a less efficacious, more toxic, and/or more expensive agent than necessary, and in some cases may be left without treatment for their underlying disease.”
To evaluate the outcomes of patients’ allergy lists, current approaches to identify culprit drugs such as the Algorithm for Drug Causality for Epidermal Necrolysis (ALDEN), which was published in 2010, and potential methods of improving culprit drug identification, the researchers performed a retrospective cohort study of 48 patients at Brigham and Women’s Hospital and Massachusetts General Hospital, with clinically and histologically confirmed cases of SJS/TEN between January 2000 and July 2018. Of the 48 patients, 26 had SJS/TEN overlap and 22 had TEN. Their median age at diagnosis was 40 years; 60.4% were female; and 52.1% were white, 12.5% were Black, 10.4% were Hispanic, and 8.3% were Asian. They took a median of 6.5 drugs in the 3 months prior to disease onset.
The researchers observed that all patients had at least one drug labeled as an allergy. A single culprit drug was labeled in 17 cases, but physicians communicated certainty in only 7 of those cases. Among all 48 patients, 104 drugs were labeled as allergies.
To identify a culprit drug, physicians appeared to mainly rely on two factors: drug notoriety and timing of exposure, compared with the onset of SJS/TEN. “Identifying high-risk medications seemed heuristic, with one or more drugs in question noted in the record as a common culprit without reference to published or vetted data regarding risk,” the researchers wrote. “Regarding timing, drug charts when present in medical records were incomplete, as they focused predominantly on high-notoriety drugs.”
In other findings, ALDEN scoring was discordant with physician-labeled lists in 28 cases. It labeled an additional 9 drugs missed by physicians and scored 43 drugs labeled as allergens by physicians as “unlikely.” The researchers also reported that 20 cases could have potentially benefited from human leukocyte antigen testing.
Their results “underscore the need for a laboratory test to identify culprit drugs,” but without such a test, “a systematic unbiased approach, such as ALDEN or the RegiSCAR database, with possibly HLA testing, should be considered to ensure the true culprit drug is not missed and exonerate as many nonculprit drugs as possible,” Dr. Divito and colleagues concluded.
They acknowledged certain limitations of the analysis, including its retrospective design and that many cases predated research advances in the topic area that took place during the 18-year study period.
Karl Saardi, MD, director of the inpatient dermatology service at George Washington University Hospital, Washington, who was asked to comment on the study, said that the findings “are consistent with clinical practice in that drug causality is usually determined by ‘gestalt’ rather than objective tools like the ALDEN algorithm.”
“The main limitation is the small size, which suggests the study sites are low-volume centers for SJS/TEN. The fact that the ALDEN score wasn’t developed until 2010 means that all the cases included prior to 2010 would not have applied the ALDEN algorithm, so I think the metric about how infrequently ALDEN was applied is not very meaningful.”
Still, he said that he was “surprised” by the number of medications that were added as allergies based on clinical impression, “and I’m glad this article does cast some light on the issue. In my experience, beta-lactam antibiotics are often – incorrectly – deemed to be the cause of SJS/TEN when further review of the patient’s medication history clearly shows they have tolerated these drugs multiple times in the past.”
Since 2000, he added, “our understanding of SJS/TEN has grown substantially including the identification of MIRM [mycoplasma-induced rash and mucositis]/RIME [reactive infections mucocutaneous eruptions] and GBFDE [generalized bullous fixed drug eruption] as mimickers.”
Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was also asked to comment on the study, agreed that a limitation of the study is that it partially preceded development of the unbiased approaches to determining the cause of a medication reaction, such as the ALDEN system. “A strength of this study is the examination of heuristics in dermatology and how they relate to patient safety,” she added.
The study was funded by grants from the National Institutes of Health, a Dermatology Foundation Diversity Research Supplement Award, and by the German Research Foundation. Dr. Divito reported receiving personal fees from Adaptimmune and MEI Pharma and a provisional patent issued from Brigham and Women’s Hospital outside the submitted work. Neither Dr. Saardi nor Dr. Ko reported having relevant disclosures.
FROM JAMA DERMATOLOGY
Understanding, Diagnosing, and Treating Long COVID
As the pandemic wanes, the public is clamoring for a return to normal. But individuals with long COVID face a challenging journey to get back to their baseline. Here’s what clinicians need to know to help patients with long COVID.
The COVID-19 pandemic is waning. The official federal public health emergency ended on May 11, 2023. Moreover, the public is ready to move on 3 years after the beginning of a pandemic that resulted in over a million deaths in the United States.
But not everyone can go back to normal. The Centers for Disease Control and Prevention (CDC) estimates that 1 in 13 US adults (7.5%) have long COVID symptoms. Many of these people feel as if they are the forgotten patients. While everyone else is moving on, a significant number of people have not returned to their baseline.
A group from Yale School of Medicine, myself included, reviewed a number of studies to gain a better understanding of 1) how long COVID manifests and 2) potential treatment options. Highlights of our evaluation are presented here.
Long COVID: The Basics
What exactly is long COVID-19, and how is it thought to develop?
The World Health Organization (WHO) defines long COVID as symptoms that persist 3 months postinfection, last for ≥ 2 months, and are not attributable to another cause.
Hypotheses about the mechanisms of long COVID include the presence of a persistent viral reservoir, an imbalance in the viral and microbial ecosystems, reactivation of latent DNA viruses, and endothelial dysfunction.
Who is most at risk?
Females
Older individuals
Individuals with preexisting conditions, including hypertension, diabetes, obesity, and lung disease
Individuals who experienced > 5 symptoms within the first week of COVID-19 illness
Individuals with breakthrough infections after vaccination against COVID-19 appear to be at increased risk of at least 1 postacute condition
Additionally, as the risk of contracting COVID-19 is demonstrably higher in certain racial and ethnic populations, it stands to reason that more of these individuals will experience long COVID.
Long COVID symptoms: how long is long?
Long COVID symptoms may persist for 2 years after initial infection. One analysis from China showed that nearly 7 in 10 patients experienced at least 1 ongoing symptom 6 months following infection, with more than half reporting symptoms at 24 months. Dyspnea, anxiety, and depression are especially persistent.
In another analysis, 90% of individuals reported symptoms 35 weeks postinfection. Symptoms did not only occur in people who were hospitalized; they also occurred in people who had a “mild case.”
Clinical Manifestations of Long COVID
More than 50 symptoms have been identified as potentially associated with long COVID. The most common manifestations involve pulmonary, cardiac, and neuropsychiatric sequelae. There is no single test to determine if symptoms are due to long COVID.
Pulmonary
How it manifests
Chronic cough
Shortness of breath
Interstitial lung disease
Treatment options
Treatment options are variable and depend on predominant symptoms. Chronic cough should be managed based on primary etiology. Treatment for interstitial lung disease depends on whether the process continues to evolve or stabilizes. The role of antifibrotics in these patients is being investigated. Lung transplantation has largely been reserved for unresolved acute injury.
Cardiac
How it manifests
Postacute sequelae cardiovascular disease, where cardiovascular disorders are uncovered during diagnostic testing
Postacute sequelae cardiovascular syndrome, such as exercise intolerance, tachycardia and chest pain, and dyspnea
Other important considerations:
Cardiac symptoms can occur independent of preexisting conditions, severity, course of acute illness, and time from original diagnosis
Cardiac involvement can occur in any age group
One analysis revealed increased risk of stroke, arrythmias, pericarditis, myocarditis, and ischemic heart disease 1 year after COVID-19 infection
Postural orthostatic tachycardia syndrome (POTS) and neurogenic orthostatic hypotension have also been observed
Treatment options
Treatment options are dictated by clinical manifestations and course. Patients who have autonomic dysfunction can be advised to increase salt and water intake since hypovolemia can worsen symptoms. Consider fludrocortisone and midodrine along with recumbent and semirecumbent exercises as tolerated, as exercise can sometimes worsen symptoms.
Neuropsychiatric
How it manifests
Patients can present with fatigue, memory disorders, headache, vertigo, myalgia, neuropathy, and smell and taste disorders, and there have been reports of cognitive decline postinfection.
Other important considerations:
A retrospective cohort study revealed that 34% of individuals had a new neurological or psychiatric diagnosis in the first 6 months after infection, including intracranial hemorrhage, ischemic stroke, parkinsonism, and dementia. Many COVID survivors experienced critical illness requiring mechanical ventilation, sedation, and paralytics, increasing the odds of developing postintensive care syndrome
Treatment options
Use of standard of care treatments, as well as neurocognitive rehabilitation and psychosocial support, is recommended for specific neuropsychiatric conditions. Patients with headache may benefit from treatment with amitriptyline or similar medications. Olfactory training and intranasal treatments can benefit those with loss of smell.
Future Directions
Two medications that may hold promise for treating individuals long COVID symptoms are currently undergoing early investigation.
Pyridostigmine may help improve peak exercise capacity
Pyridostigmine improved peak exercise oxygen uptake in patients with chronic fatigue syndrome in a randomized, double-blind, placebo-controlled trial involving 45 individuals. Participants were assigned to receive either pyridostigmine 60 mg orally or placebo, and the pyridostigmine group showed an improved peak exercise uptake via increased cardiac output and right ventricular filling pressures.
An investigational compound may improve fatigue-based symptoms
A 4-week protocol using the compound AXA1125 improved fatigue-based symptoms in patients with long COVID in a double-blind, randomized, controlled phase 2a pilot study involving 41 individuals. Investigators looked at average change in postexertional skeletal muscle phosphocreatine (PCr) recovery rate from baseline to day 28 after moderate exercise as well as fatigue levels. Although PCr recovery rate did not differ significantly between groups, use of the compound was linked with significant reduction in fatigue-based symptoms.
Summary
It is important to exercise caution when interpreting data involving individuals with long COVID. Most studies to date are retrospective and observational, definitions and assessments are not yet standardized, and long-term follow-up is lacking, among other factors.
Clinicians should remain vigilant, keeping the following in mind as they see patients who may be experiencing long COVID:
Those most at risk include females, older individuals, those with obesity, people with preexisting conditions, individuals who experienced multiple symptoms early in their COVID-19 illness, and those who had breakthrough infections after COVID-19 vaccination
Symptoms may persist up to 2 years after acute infection
The most common manifestations of long COVID involve pulmonary, cardiac, and neuropsychiatric complications
Two medications, pyridostigmine and the compound AXA1125, are under investigation and may hold promise in treating some symptoms
As the pandemic wanes, the public is clamoring for a return to normal. But individuals with long COVID face a challenging journey to get back to their baseline. Here’s what clinicians need to know to help patients with long COVID.
The COVID-19 pandemic is waning. The official federal public health emergency ended on May 11, 2023. Moreover, the public is ready to move on 3 years after the beginning of a pandemic that resulted in over a million deaths in the United States.
But not everyone can go back to normal. The Centers for Disease Control and Prevention (CDC) estimates that 1 in 13 US adults (7.5%) have long COVID symptoms. Many of these people feel as if they are the forgotten patients. While everyone else is moving on, a significant number of people have not returned to their baseline.
A group from Yale School of Medicine, myself included, reviewed a number of studies to gain a better understanding of 1) how long COVID manifests and 2) potential treatment options. Highlights of our evaluation are presented here.
Long COVID: The Basics
What exactly is long COVID-19, and how is it thought to develop?
The World Health Organization (WHO) defines long COVID as symptoms that persist 3 months postinfection, last for ≥ 2 months, and are not attributable to another cause.
Hypotheses about the mechanisms of long COVID include the presence of a persistent viral reservoir, an imbalance in the viral and microbial ecosystems, reactivation of latent DNA viruses, and endothelial dysfunction.
Who is most at risk?
Females
Older individuals
Individuals with preexisting conditions, including hypertension, diabetes, obesity, and lung disease
Individuals who experienced > 5 symptoms within the first week of COVID-19 illness
Individuals with breakthrough infections after vaccination against COVID-19 appear to be at increased risk of at least 1 postacute condition
Additionally, as the risk of contracting COVID-19 is demonstrably higher in certain racial and ethnic populations, it stands to reason that more of these individuals will experience long COVID.
Long COVID symptoms: how long is long?
Long COVID symptoms may persist for 2 years after initial infection. One analysis from China showed that nearly 7 in 10 patients experienced at least 1 ongoing symptom 6 months following infection, with more than half reporting symptoms at 24 months. Dyspnea, anxiety, and depression are especially persistent.
In another analysis, 90% of individuals reported symptoms 35 weeks postinfection. Symptoms did not only occur in people who were hospitalized; they also occurred in people who had a “mild case.”
Clinical Manifestations of Long COVID
More than 50 symptoms have been identified as potentially associated with long COVID. The most common manifestations involve pulmonary, cardiac, and neuropsychiatric sequelae. There is no single test to determine if symptoms are due to long COVID.
Pulmonary
How it manifests
Chronic cough
Shortness of breath
Interstitial lung disease
Treatment options
Treatment options are variable and depend on predominant symptoms. Chronic cough should be managed based on primary etiology. Treatment for interstitial lung disease depends on whether the process continues to evolve or stabilizes. The role of antifibrotics in these patients is being investigated. Lung transplantation has largely been reserved for unresolved acute injury.
Cardiac
How it manifests
Postacute sequelae cardiovascular disease, where cardiovascular disorders are uncovered during diagnostic testing
Postacute sequelae cardiovascular syndrome, such as exercise intolerance, tachycardia and chest pain, and dyspnea
Other important considerations:
Cardiac symptoms can occur independent of preexisting conditions, severity, course of acute illness, and time from original diagnosis
Cardiac involvement can occur in any age group
One analysis revealed increased risk of stroke, arrythmias, pericarditis, myocarditis, and ischemic heart disease 1 year after COVID-19 infection
Postural orthostatic tachycardia syndrome (POTS) and neurogenic orthostatic hypotension have also been observed
Treatment options
Treatment options are dictated by clinical manifestations and course. Patients who have autonomic dysfunction can be advised to increase salt and water intake since hypovolemia can worsen symptoms. Consider fludrocortisone and midodrine along with recumbent and semirecumbent exercises as tolerated, as exercise can sometimes worsen symptoms.
Neuropsychiatric
How it manifests
Patients can present with fatigue, memory disorders, headache, vertigo, myalgia, neuropathy, and smell and taste disorders, and there have been reports of cognitive decline postinfection.
Other important considerations:
A retrospective cohort study revealed that 34% of individuals had a new neurological or psychiatric diagnosis in the first 6 months after infection, including intracranial hemorrhage, ischemic stroke, parkinsonism, and dementia. Many COVID survivors experienced critical illness requiring mechanical ventilation, sedation, and paralytics, increasing the odds of developing postintensive care syndrome
Treatment options
Use of standard of care treatments, as well as neurocognitive rehabilitation and psychosocial support, is recommended for specific neuropsychiatric conditions. Patients with headache may benefit from treatment with amitriptyline or similar medications. Olfactory training and intranasal treatments can benefit those with loss of smell.
Future Directions
Two medications that may hold promise for treating individuals long COVID symptoms are currently undergoing early investigation.
Pyridostigmine may help improve peak exercise capacity
Pyridostigmine improved peak exercise oxygen uptake in patients with chronic fatigue syndrome in a randomized, double-blind, placebo-controlled trial involving 45 individuals. Participants were assigned to receive either pyridostigmine 60 mg orally or placebo, and the pyridostigmine group showed an improved peak exercise uptake via increased cardiac output and right ventricular filling pressures.
An investigational compound may improve fatigue-based symptoms
A 4-week protocol using the compound AXA1125 improved fatigue-based symptoms in patients with long COVID in a double-blind, randomized, controlled phase 2a pilot study involving 41 individuals. Investigators looked at average change in postexertional skeletal muscle phosphocreatine (PCr) recovery rate from baseline to day 28 after moderate exercise as well as fatigue levels. Although PCr recovery rate did not differ significantly between groups, use of the compound was linked with significant reduction in fatigue-based symptoms.
Summary
It is important to exercise caution when interpreting data involving individuals with long COVID. Most studies to date are retrospective and observational, definitions and assessments are not yet standardized, and long-term follow-up is lacking, among other factors.
Clinicians should remain vigilant, keeping the following in mind as they see patients who may be experiencing long COVID:
Those most at risk include females, older individuals, those with obesity, people with preexisting conditions, individuals who experienced multiple symptoms early in their COVID-19 illness, and those who had breakthrough infections after COVID-19 vaccination
Symptoms may persist up to 2 years after acute infection
The most common manifestations of long COVID involve pulmonary, cardiac, and neuropsychiatric complications
Two medications, pyridostigmine and the compound AXA1125, are under investigation and may hold promise in treating some symptoms
As the pandemic wanes, the public is clamoring for a return to normal. But individuals with long COVID face a challenging journey to get back to their baseline. Here’s what clinicians need to know to help patients with long COVID.
The COVID-19 pandemic is waning. The official federal public health emergency ended on May 11, 2023. Moreover, the public is ready to move on 3 years after the beginning of a pandemic that resulted in over a million deaths in the United States.
But not everyone can go back to normal. The Centers for Disease Control and Prevention (CDC) estimates that 1 in 13 US adults (7.5%) have long COVID symptoms. Many of these people feel as if they are the forgotten patients. While everyone else is moving on, a significant number of people have not returned to their baseline.
A group from Yale School of Medicine, myself included, reviewed a number of studies to gain a better understanding of 1) how long COVID manifests and 2) potential treatment options. Highlights of our evaluation are presented here.
Long COVID: The Basics
What exactly is long COVID-19, and how is it thought to develop?
The World Health Organization (WHO) defines long COVID as symptoms that persist 3 months postinfection, last for ≥ 2 months, and are not attributable to another cause.
Hypotheses about the mechanisms of long COVID include the presence of a persistent viral reservoir, an imbalance in the viral and microbial ecosystems, reactivation of latent DNA viruses, and endothelial dysfunction.
Who is most at risk?
Females
Older individuals
Individuals with preexisting conditions, including hypertension, diabetes, obesity, and lung disease
Individuals who experienced > 5 symptoms within the first week of COVID-19 illness
Individuals with breakthrough infections after vaccination against COVID-19 appear to be at increased risk of at least 1 postacute condition
Additionally, as the risk of contracting COVID-19 is demonstrably higher in certain racial and ethnic populations, it stands to reason that more of these individuals will experience long COVID.
Long COVID symptoms: how long is long?
Long COVID symptoms may persist for 2 years after initial infection. One analysis from China showed that nearly 7 in 10 patients experienced at least 1 ongoing symptom 6 months following infection, with more than half reporting symptoms at 24 months. Dyspnea, anxiety, and depression are especially persistent.
In another analysis, 90% of individuals reported symptoms 35 weeks postinfection. Symptoms did not only occur in people who were hospitalized; they also occurred in people who had a “mild case.”
Clinical Manifestations of Long COVID
More than 50 symptoms have been identified as potentially associated with long COVID. The most common manifestations involve pulmonary, cardiac, and neuropsychiatric sequelae. There is no single test to determine if symptoms are due to long COVID.
Pulmonary
How it manifests
Chronic cough
Shortness of breath
Interstitial lung disease
Treatment options
Treatment options are variable and depend on predominant symptoms. Chronic cough should be managed based on primary etiology. Treatment for interstitial lung disease depends on whether the process continues to evolve or stabilizes. The role of antifibrotics in these patients is being investigated. Lung transplantation has largely been reserved for unresolved acute injury.
Cardiac
How it manifests
Postacute sequelae cardiovascular disease, where cardiovascular disorders are uncovered during diagnostic testing
Postacute sequelae cardiovascular syndrome, such as exercise intolerance, tachycardia and chest pain, and dyspnea
Other important considerations:
Cardiac symptoms can occur independent of preexisting conditions, severity, course of acute illness, and time from original diagnosis
Cardiac involvement can occur in any age group
One analysis revealed increased risk of stroke, arrythmias, pericarditis, myocarditis, and ischemic heart disease 1 year after COVID-19 infection
Postural orthostatic tachycardia syndrome (POTS) and neurogenic orthostatic hypotension have also been observed
Treatment options
Treatment options are dictated by clinical manifestations and course. Patients who have autonomic dysfunction can be advised to increase salt and water intake since hypovolemia can worsen symptoms. Consider fludrocortisone and midodrine along with recumbent and semirecumbent exercises as tolerated, as exercise can sometimes worsen symptoms.
Neuropsychiatric
How it manifests
Patients can present with fatigue, memory disorders, headache, vertigo, myalgia, neuropathy, and smell and taste disorders, and there have been reports of cognitive decline postinfection.
Other important considerations:
A retrospective cohort study revealed that 34% of individuals had a new neurological or psychiatric diagnosis in the first 6 months after infection, including intracranial hemorrhage, ischemic stroke, parkinsonism, and dementia. Many COVID survivors experienced critical illness requiring mechanical ventilation, sedation, and paralytics, increasing the odds of developing postintensive care syndrome
Treatment options
Use of standard of care treatments, as well as neurocognitive rehabilitation and psychosocial support, is recommended for specific neuropsychiatric conditions. Patients with headache may benefit from treatment with amitriptyline or similar medications. Olfactory training and intranasal treatments can benefit those with loss of smell.
Future Directions
Two medications that may hold promise for treating individuals long COVID symptoms are currently undergoing early investigation.
Pyridostigmine may help improve peak exercise capacity
Pyridostigmine improved peak exercise oxygen uptake in patients with chronic fatigue syndrome in a randomized, double-blind, placebo-controlled trial involving 45 individuals. Participants were assigned to receive either pyridostigmine 60 mg orally or placebo, and the pyridostigmine group showed an improved peak exercise uptake via increased cardiac output and right ventricular filling pressures.
An investigational compound may improve fatigue-based symptoms
A 4-week protocol using the compound AXA1125 improved fatigue-based symptoms in patients with long COVID in a double-blind, randomized, controlled phase 2a pilot study involving 41 individuals. Investigators looked at average change in postexertional skeletal muscle phosphocreatine (PCr) recovery rate from baseline to day 28 after moderate exercise as well as fatigue levels. Although PCr recovery rate did not differ significantly between groups, use of the compound was linked with significant reduction in fatigue-based symptoms.
Summary
It is important to exercise caution when interpreting data involving individuals with long COVID. Most studies to date are retrospective and observational, definitions and assessments are not yet standardized, and long-term follow-up is lacking, among other factors.
Clinicians should remain vigilant, keeping the following in mind as they see patients who may be experiencing long COVID:
Those most at risk include females, older individuals, those with obesity, people with preexisting conditions, individuals who experienced multiple symptoms early in their COVID-19 illness, and those who had breakthrough infections after COVID-19 vaccination
Symptoms may persist up to 2 years after acute infection
The most common manifestations of long COVID involve pulmonary, cardiac, and neuropsychiatric complications
Two medications, pyridostigmine and the compound AXA1125, are under investigation and may hold promise in treating some symptoms
Limiting social media use in youths brings challenges, benefits
Amelia Kennedy, 19, of Royersford, Pa., a point guard on the Catholic University of America, Washington, basketball team who will begin her sophomore year in the fall, uses TikTok, Snapchat, Instagram, and YouTube regularly.
How regularly? She estimates 7 hours a day and about 9 on weekends. She’s aware of the time-wasting potential. “If my mom says, ‘Do dishes,’ and I say, ‘5 more minutes,’ it can be longer,’’ she said.
Now imagine the challenge of cutting that 7 or 9 hours a day of social media use down to 30 minutes.
A very tall order, considering a 2022 Pew Research Center survey of more than 1,300 teens found 35% are “nearly constantly” on at least one of the top five social media platforms: YouTube, TikTok, Instagram, Snapchat, and Facebook.
Researchers from Iowa State University, Ames, recently took on that daunting challenge, limiting a group of students to only 30 minutes of social media a day to see what happens. Two weeks into the study, the students reported improvement in psychological well-being and other important measures, including sleep quality, compared with a control group assigned to continue using social media as usual.
And the dreaded FOMO, or fear of missing out, didn’t happen, the researchers said. At the end, the students were rethinking their social media use and feeling positive about it.
As social media becomes more common and youth mental health more endangered, experts are sounding the alarm. In late May, U.S. Surgeon General Vivek Murthy, MD, issued an advisory on social media and youth mental health, calling for tech companies to do better, policymakers to strengthen safety, and researchers to get more information, among other actions.
After that, the Biden administration took actions including the launch of a task force on kids’ online health and safety. The American Psychological Association has issued recommendations on social media use in youths. And the Social Media Victims Law Center in Seattle has sued numerous social media companies for online activity resulting in death and other tragedies.
While experts acknowledge that much more research is needed to sort out how to balance social media’s risks and benefits to preserve youth mental health and prevent such disasters, the new Iowa State study, as well as other recent research, suggests that
Goal: 30 minutes a day
In the Iowa State study, 230 undergraduate students were assigned to one of two groups, with 99 in the 30-minute-a-day social media use group and 131 in the “usual” or control group, which made no changes. For those in the intervention group, “we sent a daily reminder email,” said Ella Faulhaber, a PhD candidate at Iowa State and the study’s lead author. It simply reminded them to limit social medial use to the 30-minute maximum.
At the study start and end, all participants provided a screenshot of their weekly social media usage time. The researchers gave both groups a battery of tests to assess anxiety, depression, loneliness, fear of missing out, and negative and positive feelings.
“By limiting their social media time, that resulted in less anxiety, less depression, less FOMO, fewer negative emotions, and greater positive emotions,” said Douglas Gentile, PhD, a distinguished professor of psychology at Iowa State and a study coauthor. “We know that it is the limiting [of] the social media that is causing that.”
Ms. Faulhaber recalled one participant who mentioned having trouble at first adjusting to the 30-minute time frame, but once sleep improved, it was easier to stick to that guidance. Another who gave up phone use at bedtime found: “Instead of looking at my phone, it was much easier to go straight to bed.”
Sleep improvements, of course, affect many parts of physical and mental health, Dr. Gentile said. And the study also showed that even with reduced screen time, “we can still get the benefit of being connected.” Those who didn’t make the 30-minute mark, but cut back, got benefits, too, the researchers said.
‘Youth are aware’
Self-monitoring works, agreed Jane Harness, DO, an adjunct clinical assistant professor of psychiatry at the University of Michigan, Ann Arbor, because “having that insight is often the first step.”
In a study she conducted, Dr. Harness aimed to gather youths’ insights about how their social media use affected them. With her colleagues, she asked more than 1,100 youths, aged 14-24, what advice they would give to those new to social media, if they ever felt they needed to change social media habits, and if they have deleted or considered deleting social media accounts.
From the 871 responses, Harness found that youths were especially concerned about safety online, that most had thought about deleting a social media app and some had, and that youths were more likely to say they wanted to change the amount of time spent on social media, compared with the content they view.
“Users responded with great advice for each other,” she said. “Safety was brought up,” with users reminding others to keep accounts private and to be aware of location tracking links and content that seems to promote eating disorders, suicide, and other harms.
In the study report, Dr. Harness concluded: “Youth are aware of ways in which social media could be negatively impacting them and they have employed methods to modulate their use because of this awareness.”
Less FOMO, less anxiety
In an earlier study, University of Pennsylvania, Philadelphia, researchers had 143 college students self-monitor social media for a week, then randomly assigned them either to a group told to limit Facebook, Instagram, and Snapchat use to 10 minutes per platform, per day, or to a group told to use social media as usual for 3 weeks.
At the end of the study, the researchers evaluated both groups and found “significant reductions in loneliness and depression over 3 weeks’’ in the limited-use group, compared with the usual-use group, according to study researcher Melissa G. Hunt, PhD, associate director of clinical training at the University of Pennsylvania.
And both groups showed declines in anxiety and fear of missing out, suggesting a benefit tied to self-monitoring itself, she said.
While Dr. Hunt’s study focused on 30 minutes a day, she said “about an hour a day seems to be the sweet spot for maximizing the positive effects of connecting, but limiting the negative effects of social media use.”
She also suggested that smartphones have no place in middle or high school classrooms. Instead, they should be on lockdown during classes.
“Parents need to set real limits of cellphone use during meals and in bedrooms,” Dr. Hunt said. At mealtime, for instance, all phones should be absent from the table. And after 10 p.m., “all family phones remain in the kitchen.”
Be ‘more mindful’
These recent study findings about self-monitoring and limiting social media time may not work the same for everyone, especially among those who aren’t as motivated, said psychiatrist Elizabeth Ortiz-Schwartz, MD, team lead for the adolescent inpatient unit at Silver Hill Hospital in New Canaan, Conn.
But “the bigger take-home piece is that being intentional and attempting to decrease the use in these individuals, even if they were not always successful, was clearly beneficial,” she said.
As we await clearer guidelines about what is the “right” amount of use in terms of social media content and time, Dr. Ortiz-Schwartz said, “becoming more mindful and aware of the risks and benefits can hopefully help individuals become more mindful and deliberate about its use.”
Real-world strategies
Max Schwandt, 23, is an outlier, but a happy one. He works as a sales clerk at a Los Angeles–area recreational gear shop, and he uses no social media. Why not? “It takes up too much time,” he explained. As simple as that.
But for many other teens and young adults, the struggle to stay off social media is real.
Amelia Kennedy, the Catholic University of America student, is trying to reduce her screen time. One way is to track it on her phone. These days, her summer job at a restaurant serving breakfast gets her up early. “If I have to work, I still go on my phone, but not that long.” And once at work, she only has time for quick checks between work responsibilities. “I definitely am more productive,” she said about days when she has work.
Last December, Lauren Young, 25, whose father was a researcher on the Iowa State study, was finishing law school at Georgetown University, Washington, and decided to take a break from social media for the entire month. “I can’t say I was always successful in avoiding it,” she said. But cutting down greatly “made me a lot more present in my day-to-day life, and it was easier to concentrate.”
She could even get through a meal, out with friends, without her phone, keeping it in her purse. That was a definite change from the norm. “I noticed I would go out to dinner and the standard for people my age is having the phone on the table. If you are being polite, you turn it over.”
During her social media “blackout,” Ms. Young had deleted TikTok, Instagram, and Facebook apps. Then, when she graduated, she had to reinstall to post a picture. But now, she is back to minimal social media use.
“I’m studying for the bar, so it’s kind of necessary, but it always makes me happier.” She figures she can always text family and friends if necessary, instead of posting. “I felt for a while I was missing out on things, but not now,” she said.
Others, including Sarah Goldstein, 22, of Chatsworth, Calif., a supermarket courtesy clerk who is thinking of returning to college, said she has developed a healthier attitude toward social media as she has gotten older.
“In middle and early high school, I would see parties, things I wasn’t invited to, on Snapchat and Instagram.” While she realized there could be legitimate reasons for not being included, she said it was easy to internalize those feelings of being left out.
These days, she said she doesn’t let it affect her mental health that way. She enjoys social media – especially TikTok and Instagram – for its benefits. “It kills time, gives you something to watch, can make you laugh and feel like you have a connection with other people.”
A version of this article first appeared on WebMD.com.
Amelia Kennedy, 19, of Royersford, Pa., a point guard on the Catholic University of America, Washington, basketball team who will begin her sophomore year in the fall, uses TikTok, Snapchat, Instagram, and YouTube regularly.
How regularly? She estimates 7 hours a day and about 9 on weekends. She’s aware of the time-wasting potential. “If my mom says, ‘Do dishes,’ and I say, ‘5 more minutes,’ it can be longer,’’ she said.
Now imagine the challenge of cutting that 7 or 9 hours a day of social media use down to 30 minutes.
A very tall order, considering a 2022 Pew Research Center survey of more than 1,300 teens found 35% are “nearly constantly” on at least one of the top five social media platforms: YouTube, TikTok, Instagram, Snapchat, and Facebook.
Researchers from Iowa State University, Ames, recently took on that daunting challenge, limiting a group of students to only 30 minutes of social media a day to see what happens. Two weeks into the study, the students reported improvement in psychological well-being and other important measures, including sleep quality, compared with a control group assigned to continue using social media as usual.
And the dreaded FOMO, or fear of missing out, didn’t happen, the researchers said. At the end, the students were rethinking their social media use and feeling positive about it.
As social media becomes more common and youth mental health more endangered, experts are sounding the alarm. In late May, U.S. Surgeon General Vivek Murthy, MD, issued an advisory on social media and youth mental health, calling for tech companies to do better, policymakers to strengthen safety, and researchers to get more information, among other actions.
After that, the Biden administration took actions including the launch of a task force on kids’ online health and safety. The American Psychological Association has issued recommendations on social media use in youths. And the Social Media Victims Law Center in Seattle has sued numerous social media companies for online activity resulting in death and other tragedies.
While experts acknowledge that much more research is needed to sort out how to balance social media’s risks and benefits to preserve youth mental health and prevent such disasters, the new Iowa State study, as well as other recent research, suggests that
Goal: 30 minutes a day
In the Iowa State study, 230 undergraduate students were assigned to one of two groups, with 99 in the 30-minute-a-day social media use group and 131 in the “usual” or control group, which made no changes. For those in the intervention group, “we sent a daily reminder email,” said Ella Faulhaber, a PhD candidate at Iowa State and the study’s lead author. It simply reminded them to limit social medial use to the 30-minute maximum.
At the study start and end, all participants provided a screenshot of their weekly social media usage time. The researchers gave both groups a battery of tests to assess anxiety, depression, loneliness, fear of missing out, and negative and positive feelings.
“By limiting their social media time, that resulted in less anxiety, less depression, less FOMO, fewer negative emotions, and greater positive emotions,” said Douglas Gentile, PhD, a distinguished professor of psychology at Iowa State and a study coauthor. “We know that it is the limiting [of] the social media that is causing that.”
Ms. Faulhaber recalled one participant who mentioned having trouble at first adjusting to the 30-minute time frame, but once sleep improved, it was easier to stick to that guidance. Another who gave up phone use at bedtime found: “Instead of looking at my phone, it was much easier to go straight to bed.”
Sleep improvements, of course, affect many parts of physical and mental health, Dr. Gentile said. And the study also showed that even with reduced screen time, “we can still get the benefit of being connected.” Those who didn’t make the 30-minute mark, but cut back, got benefits, too, the researchers said.
‘Youth are aware’
Self-monitoring works, agreed Jane Harness, DO, an adjunct clinical assistant professor of psychiatry at the University of Michigan, Ann Arbor, because “having that insight is often the first step.”
In a study she conducted, Dr. Harness aimed to gather youths’ insights about how their social media use affected them. With her colleagues, she asked more than 1,100 youths, aged 14-24, what advice they would give to those new to social media, if they ever felt they needed to change social media habits, and if they have deleted or considered deleting social media accounts.
From the 871 responses, Harness found that youths were especially concerned about safety online, that most had thought about deleting a social media app and some had, and that youths were more likely to say they wanted to change the amount of time spent on social media, compared with the content they view.
“Users responded with great advice for each other,” she said. “Safety was brought up,” with users reminding others to keep accounts private and to be aware of location tracking links and content that seems to promote eating disorders, suicide, and other harms.
In the study report, Dr. Harness concluded: “Youth are aware of ways in which social media could be negatively impacting them and they have employed methods to modulate their use because of this awareness.”
Less FOMO, less anxiety
In an earlier study, University of Pennsylvania, Philadelphia, researchers had 143 college students self-monitor social media for a week, then randomly assigned them either to a group told to limit Facebook, Instagram, and Snapchat use to 10 minutes per platform, per day, or to a group told to use social media as usual for 3 weeks.
At the end of the study, the researchers evaluated both groups and found “significant reductions in loneliness and depression over 3 weeks’’ in the limited-use group, compared with the usual-use group, according to study researcher Melissa G. Hunt, PhD, associate director of clinical training at the University of Pennsylvania.
And both groups showed declines in anxiety and fear of missing out, suggesting a benefit tied to self-monitoring itself, she said.
While Dr. Hunt’s study focused on 30 minutes a day, she said “about an hour a day seems to be the sweet spot for maximizing the positive effects of connecting, but limiting the negative effects of social media use.”
She also suggested that smartphones have no place in middle or high school classrooms. Instead, they should be on lockdown during classes.
“Parents need to set real limits of cellphone use during meals and in bedrooms,” Dr. Hunt said. At mealtime, for instance, all phones should be absent from the table. And after 10 p.m., “all family phones remain in the kitchen.”
Be ‘more mindful’
These recent study findings about self-monitoring and limiting social media time may not work the same for everyone, especially among those who aren’t as motivated, said psychiatrist Elizabeth Ortiz-Schwartz, MD, team lead for the adolescent inpatient unit at Silver Hill Hospital in New Canaan, Conn.
But “the bigger take-home piece is that being intentional and attempting to decrease the use in these individuals, even if they were not always successful, was clearly beneficial,” she said.
As we await clearer guidelines about what is the “right” amount of use in terms of social media content and time, Dr. Ortiz-Schwartz said, “becoming more mindful and aware of the risks and benefits can hopefully help individuals become more mindful and deliberate about its use.”
Real-world strategies
Max Schwandt, 23, is an outlier, but a happy one. He works as a sales clerk at a Los Angeles–area recreational gear shop, and he uses no social media. Why not? “It takes up too much time,” he explained. As simple as that.
But for many other teens and young adults, the struggle to stay off social media is real.
Amelia Kennedy, the Catholic University of America student, is trying to reduce her screen time. One way is to track it on her phone. These days, her summer job at a restaurant serving breakfast gets her up early. “If I have to work, I still go on my phone, but not that long.” And once at work, she only has time for quick checks between work responsibilities. “I definitely am more productive,” she said about days when she has work.
Last December, Lauren Young, 25, whose father was a researcher on the Iowa State study, was finishing law school at Georgetown University, Washington, and decided to take a break from social media for the entire month. “I can’t say I was always successful in avoiding it,” she said. But cutting down greatly “made me a lot more present in my day-to-day life, and it was easier to concentrate.”
She could even get through a meal, out with friends, without her phone, keeping it in her purse. That was a definite change from the norm. “I noticed I would go out to dinner and the standard for people my age is having the phone on the table. If you are being polite, you turn it over.”
During her social media “blackout,” Ms. Young had deleted TikTok, Instagram, and Facebook apps. Then, when she graduated, she had to reinstall to post a picture. But now, she is back to minimal social media use.
“I’m studying for the bar, so it’s kind of necessary, but it always makes me happier.” She figures she can always text family and friends if necessary, instead of posting. “I felt for a while I was missing out on things, but not now,” she said.
Others, including Sarah Goldstein, 22, of Chatsworth, Calif., a supermarket courtesy clerk who is thinking of returning to college, said she has developed a healthier attitude toward social media as she has gotten older.
“In middle and early high school, I would see parties, things I wasn’t invited to, on Snapchat and Instagram.” While she realized there could be legitimate reasons for not being included, she said it was easy to internalize those feelings of being left out.
These days, she said she doesn’t let it affect her mental health that way. She enjoys social media – especially TikTok and Instagram – for its benefits. “It kills time, gives you something to watch, can make you laugh and feel like you have a connection with other people.”
A version of this article first appeared on WebMD.com.
Amelia Kennedy, 19, of Royersford, Pa., a point guard on the Catholic University of America, Washington, basketball team who will begin her sophomore year in the fall, uses TikTok, Snapchat, Instagram, and YouTube regularly.
How regularly? She estimates 7 hours a day and about 9 on weekends. She’s aware of the time-wasting potential. “If my mom says, ‘Do dishes,’ and I say, ‘5 more minutes,’ it can be longer,’’ she said.
Now imagine the challenge of cutting that 7 or 9 hours a day of social media use down to 30 minutes.
A very tall order, considering a 2022 Pew Research Center survey of more than 1,300 teens found 35% are “nearly constantly” on at least one of the top five social media platforms: YouTube, TikTok, Instagram, Snapchat, and Facebook.
Researchers from Iowa State University, Ames, recently took on that daunting challenge, limiting a group of students to only 30 minutes of social media a day to see what happens. Two weeks into the study, the students reported improvement in psychological well-being and other important measures, including sleep quality, compared with a control group assigned to continue using social media as usual.
And the dreaded FOMO, or fear of missing out, didn’t happen, the researchers said. At the end, the students were rethinking their social media use and feeling positive about it.
As social media becomes more common and youth mental health more endangered, experts are sounding the alarm. In late May, U.S. Surgeon General Vivek Murthy, MD, issued an advisory on social media and youth mental health, calling for tech companies to do better, policymakers to strengthen safety, and researchers to get more information, among other actions.
After that, the Biden administration took actions including the launch of a task force on kids’ online health and safety. The American Psychological Association has issued recommendations on social media use in youths. And the Social Media Victims Law Center in Seattle has sued numerous social media companies for online activity resulting in death and other tragedies.
While experts acknowledge that much more research is needed to sort out how to balance social media’s risks and benefits to preserve youth mental health and prevent such disasters, the new Iowa State study, as well as other recent research, suggests that
Goal: 30 minutes a day
In the Iowa State study, 230 undergraduate students were assigned to one of two groups, with 99 in the 30-minute-a-day social media use group and 131 in the “usual” or control group, which made no changes. For those in the intervention group, “we sent a daily reminder email,” said Ella Faulhaber, a PhD candidate at Iowa State and the study’s lead author. It simply reminded them to limit social medial use to the 30-minute maximum.
At the study start and end, all participants provided a screenshot of their weekly social media usage time. The researchers gave both groups a battery of tests to assess anxiety, depression, loneliness, fear of missing out, and negative and positive feelings.
“By limiting their social media time, that resulted in less anxiety, less depression, less FOMO, fewer negative emotions, and greater positive emotions,” said Douglas Gentile, PhD, a distinguished professor of psychology at Iowa State and a study coauthor. “We know that it is the limiting [of] the social media that is causing that.”
Ms. Faulhaber recalled one participant who mentioned having trouble at first adjusting to the 30-minute time frame, but once sleep improved, it was easier to stick to that guidance. Another who gave up phone use at bedtime found: “Instead of looking at my phone, it was much easier to go straight to bed.”
Sleep improvements, of course, affect many parts of physical and mental health, Dr. Gentile said. And the study also showed that even with reduced screen time, “we can still get the benefit of being connected.” Those who didn’t make the 30-minute mark, but cut back, got benefits, too, the researchers said.
‘Youth are aware’
Self-monitoring works, agreed Jane Harness, DO, an adjunct clinical assistant professor of psychiatry at the University of Michigan, Ann Arbor, because “having that insight is often the first step.”
In a study she conducted, Dr. Harness aimed to gather youths’ insights about how their social media use affected them. With her colleagues, she asked more than 1,100 youths, aged 14-24, what advice they would give to those new to social media, if they ever felt they needed to change social media habits, and if they have deleted or considered deleting social media accounts.
From the 871 responses, Harness found that youths were especially concerned about safety online, that most had thought about deleting a social media app and some had, and that youths were more likely to say they wanted to change the amount of time spent on social media, compared with the content they view.
“Users responded with great advice for each other,” she said. “Safety was brought up,” with users reminding others to keep accounts private and to be aware of location tracking links and content that seems to promote eating disorders, suicide, and other harms.
In the study report, Dr. Harness concluded: “Youth are aware of ways in which social media could be negatively impacting them and they have employed methods to modulate their use because of this awareness.”
Less FOMO, less anxiety
In an earlier study, University of Pennsylvania, Philadelphia, researchers had 143 college students self-monitor social media for a week, then randomly assigned them either to a group told to limit Facebook, Instagram, and Snapchat use to 10 minutes per platform, per day, or to a group told to use social media as usual for 3 weeks.
At the end of the study, the researchers evaluated both groups and found “significant reductions in loneliness and depression over 3 weeks’’ in the limited-use group, compared with the usual-use group, according to study researcher Melissa G. Hunt, PhD, associate director of clinical training at the University of Pennsylvania.
And both groups showed declines in anxiety and fear of missing out, suggesting a benefit tied to self-monitoring itself, she said.
While Dr. Hunt’s study focused on 30 minutes a day, she said “about an hour a day seems to be the sweet spot for maximizing the positive effects of connecting, but limiting the negative effects of social media use.”
She also suggested that smartphones have no place in middle or high school classrooms. Instead, they should be on lockdown during classes.
“Parents need to set real limits of cellphone use during meals and in bedrooms,” Dr. Hunt said. At mealtime, for instance, all phones should be absent from the table. And after 10 p.m., “all family phones remain in the kitchen.”
Be ‘more mindful’
These recent study findings about self-monitoring and limiting social media time may not work the same for everyone, especially among those who aren’t as motivated, said psychiatrist Elizabeth Ortiz-Schwartz, MD, team lead for the adolescent inpatient unit at Silver Hill Hospital in New Canaan, Conn.
But “the bigger take-home piece is that being intentional and attempting to decrease the use in these individuals, even if they were not always successful, was clearly beneficial,” she said.
As we await clearer guidelines about what is the “right” amount of use in terms of social media content and time, Dr. Ortiz-Schwartz said, “becoming more mindful and aware of the risks and benefits can hopefully help individuals become more mindful and deliberate about its use.”
Real-world strategies
Max Schwandt, 23, is an outlier, but a happy one. He works as a sales clerk at a Los Angeles–area recreational gear shop, and he uses no social media. Why not? “It takes up too much time,” he explained. As simple as that.
But for many other teens and young adults, the struggle to stay off social media is real.
Amelia Kennedy, the Catholic University of America student, is trying to reduce her screen time. One way is to track it on her phone. These days, her summer job at a restaurant serving breakfast gets her up early. “If I have to work, I still go on my phone, but not that long.” And once at work, she only has time for quick checks between work responsibilities. “I definitely am more productive,” she said about days when she has work.
Last December, Lauren Young, 25, whose father was a researcher on the Iowa State study, was finishing law school at Georgetown University, Washington, and decided to take a break from social media for the entire month. “I can’t say I was always successful in avoiding it,” she said. But cutting down greatly “made me a lot more present in my day-to-day life, and it was easier to concentrate.”
She could even get through a meal, out with friends, without her phone, keeping it in her purse. That was a definite change from the norm. “I noticed I would go out to dinner and the standard for people my age is having the phone on the table. If you are being polite, you turn it over.”
During her social media “blackout,” Ms. Young had deleted TikTok, Instagram, and Facebook apps. Then, when she graduated, she had to reinstall to post a picture. But now, she is back to minimal social media use.
“I’m studying for the bar, so it’s kind of necessary, but it always makes me happier.” She figures she can always text family and friends if necessary, instead of posting. “I felt for a while I was missing out on things, but not now,” she said.
Others, including Sarah Goldstein, 22, of Chatsworth, Calif., a supermarket courtesy clerk who is thinking of returning to college, said she has developed a healthier attitude toward social media as she has gotten older.
“In middle and early high school, I would see parties, things I wasn’t invited to, on Snapchat and Instagram.” While she realized there could be legitimate reasons for not being included, she said it was easy to internalize those feelings of being left out.
These days, she said she doesn’t let it affect her mental health that way. She enjoys social media – especially TikTok and Instagram – for its benefits. “It kills time, gives you something to watch, can make you laugh and feel like you have a connection with other people.”
A version of this article first appeared on WebMD.com.
Agency issues advisory on mental health symptoms of long COVID
The nine mental health symptoms highlighted in the advisory are fatigue; cognitive impairment, including brain fog; anxiety; depression; obsessive-compulsive disorder; sleep disorders; PTSD; psychotic disorder; and start of a substance use disorder.
The advisory noted that social factors can contribute to the mental health problems for racial and ethnic minorities; people with limited access to health care; people who already have behavioral health conditions and physical disabilities; and people who are lesbian, gay, bisexual, transgender, queer, or intersex.
“Long COVID has a range of burdensome physical symptoms and can take a toll on a person’s mental health. It can be very challenging for a person, whether they are impacted themselves, or they are a caregiver for someone who is affected,” Health and Human Services Secretary Xavier Becerra said in a statement. “This advisory helps to raise awareness, especially among primary care practitioners and clinicians who are often the ones treating patients with long COVID.”
The department says about 10% of people infected with COVID have at least one long COVID symptom. Physical symptoms include dizziness, stomach upset, heart palpitations, issues with sexual desire or capacity, loss of smell or taste, thirst, chronic coughing, chest pain, and abnormal movements.
“We know that people living with long COVID need help today, and providers need help understanding what long COVID is and how to treat it,” Admiral Rachel Levine, MD, assistant secretary for health, said in the statement. “This advisory helps bridge that gap for the behavioral health impacts of long COVID.”
A version of this article first appeared on WebMD.com.
The nine mental health symptoms highlighted in the advisory are fatigue; cognitive impairment, including brain fog; anxiety; depression; obsessive-compulsive disorder; sleep disorders; PTSD; psychotic disorder; and start of a substance use disorder.
The advisory noted that social factors can contribute to the mental health problems for racial and ethnic minorities; people with limited access to health care; people who already have behavioral health conditions and physical disabilities; and people who are lesbian, gay, bisexual, transgender, queer, or intersex.
“Long COVID has a range of burdensome physical symptoms and can take a toll on a person’s mental health. It can be very challenging for a person, whether they are impacted themselves, or they are a caregiver for someone who is affected,” Health and Human Services Secretary Xavier Becerra said in a statement. “This advisory helps to raise awareness, especially among primary care practitioners and clinicians who are often the ones treating patients with long COVID.”
The department says about 10% of people infected with COVID have at least one long COVID symptom. Physical symptoms include dizziness, stomach upset, heart palpitations, issues with sexual desire or capacity, loss of smell or taste, thirst, chronic coughing, chest pain, and abnormal movements.
“We know that people living with long COVID need help today, and providers need help understanding what long COVID is and how to treat it,” Admiral Rachel Levine, MD, assistant secretary for health, said in the statement. “This advisory helps bridge that gap for the behavioral health impacts of long COVID.”
A version of this article first appeared on WebMD.com.
The nine mental health symptoms highlighted in the advisory are fatigue; cognitive impairment, including brain fog; anxiety; depression; obsessive-compulsive disorder; sleep disorders; PTSD; psychotic disorder; and start of a substance use disorder.
The advisory noted that social factors can contribute to the mental health problems for racial and ethnic minorities; people with limited access to health care; people who already have behavioral health conditions and physical disabilities; and people who are lesbian, gay, bisexual, transgender, queer, or intersex.
“Long COVID has a range of burdensome physical symptoms and can take a toll on a person’s mental health. It can be very challenging for a person, whether they are impacted themselves, or they are a caregiver for someone who is affected,” Health and Human Services Secretary Xavier Becerra said in a statement. “This advisory helps to raise awareness, especially among primary care practitioners and clinicians who are often the ones treating patients with long COVID.”
The department says about 10% of people infected with COVID have at least one long COVID symptom. Physical symptoms include dizziness, stomach upset, heart palpitations, issues with sexual desire or capacity, loss of smell or taste, thirst, chronic coughing, chest pain, and abnormal movements.
“We know that people living with long COVID need help today, and providers need help understanding what long COVID is and how to treat it,” Admiral Rachel Levine, MD, assistant secretary for health, said in the statement. “This advisory helps bridge that gap for the behavioral health impacts of long COVID.”
A version of this article first appeared on WebMD.com.
Myasthenia gravis drug gets FDA nod
, the drug’s manufacturer, UCB, has announced.
Rozanolixizumab is a subcutaneous-infused humanized IgG4 monoclonal antibody that binds to the neonatal Fc receptor (FcRn), reducing the concentration of pathogenic IgG autoantibodies.
U.S. approval is based on results of the phase 3 MycarinG study involving 200 patients with AChR or MuSK autoantibody-positive gMG. Patients were randomly assigned to one of two rozanolixizumab groups (7 mg/kg or 10 mg/kg) or placebo for 6 weeks.
As reported last month in Lancet Neurology, rozanolixizumab led to statistically significant improvements in gMG-specific outcomes, including everyday activities such as breathing, talking, swallowing, and being able to rise from a chair.
“There is a significant need for new, innovative treatment options to reduce the day-to-day burden of gMG,” lead investigator Vera Bril, MD, professor of medicine (neurology), University of Toronto, said in a news release.
Rozanolixizumab is “a new treatment option, targeting one of the mechanisms of disease to provide symptom improvement in patient- and physician-reported outcomes at day 43,” Dr. Bril added.
The most common adverse reactions (reported in at least 10% of patients treated with rozanolixizumab) were headache, infections, diarrhea, pyrexia, hypersensitivity reactions, and nausea.
The company expects rozanolixizumab to be available in the United States during the third quarter of 2023.
The FDA granted the application for rozanolixizumab in gMG priority review.
A version of this article first appeared on Medscape.com.
, the drug’s manufacturer, UCB, has announced.
Rozanolixizumab is a subcutaneous-infused humanized IgG4 monoclonal antibody that binds to the neonatal Fc receptor (FcRn), reducing the concentration of pathogenic IgG autoantibodies.
U.S. approval is based on results of the phase 3 MycarinG study involving 200 patients with AChR or MuSK autoantibody-positive gMG. Patients were randomly assigned to one of two rozanolixizumab groups (7 mg/kg or 10 mg/kg) or placebo for 6 weeks.
As reported last month in Lancet Neurology, rozanolixizumab led to statistically significant improvements in gMG-specific outcomes, including everyday activities such as breathing, talking, swallowing, and being able to rise from a chair.
“There is a significant need for new, innovative treatment options to reduce the day-to-day burden of gMG,” lead investigator Vera Bril, MD, professor of medicine (neurology), University of Toronto, said in a news release.
Rozanolixizumab is “a new treatment option, targeting one of the mechanisms of disease to provide symptom improvement in patient- and physician-reported outcomes at day 43,” Dr. Bril added.
The most common adverse reactions (reported in at least 10% of patients treated with rozanolixizumab) were headache, infections, diarrhea, pyrexia, hypersensitivity reactions, and nausea.
The company expects rozanolixizumab to be available in the United States during the third quarter of 2023.
The FDA granted the application for rozanolixizumab in gMG priority review.
A version of this article first appeared on Medscape.com.
, the drug’s manufacturer, UCB, has announced.
Rozanolixizumab is a subcutaneous-infused humanized IgG4 monoclonal antibody that binds to the neonatal Fc receptor (FcRn), reducing the concentration of pathogenic IgG autoantibodies.
U.S. approval is based on results of the phase 3 MycarinG study involving 200 patients with AChR or MuSK autoantibody-positive gMG. Patients were randomly assigned to one of two rozanolixizumab groups (7 mg/kg or 10 mg/kg) or placebo for 6 weeks.
As reported last month in Lancet Neurology, rozanolixizumab led to statistically significant improvements in gMG-specific outcomes, including everyday activities such as breathing, talking, swallowing, and being able to rise from a chair.
“There is a significant need for new, innovative treatment options to reduce the day-to-day burden of gMG,” lead investigator Vera Bril, MD, professor of medicine (neurology), University of Toronto, said in a news release.
Rozanolixizumab is “a new treatment option, targeting one of the mechanisms of disease to provide symptom improvement in patient- and physician-reported outcomes at day 43,” Dr. Bril added.
The most common adverse reactions (reported in at least 10% of patients treated with rozanolixizumab) were headache, infections, diarrhea, pyrexia, hypersensitivity reactions, and nausea.
The company expects rozanolixizumab to be available in the United States during the third quarter of 2023.
The FDA granted the application for rozanolixizumab in gMG priority review.
A version of this article first appeared on Medscape.com.
FDA clears new biomarker assays for early Alzheimer’s detection
The Elecsys beta-amyloid (1-42) CSF II (Abeta42) and Elecsys total-tau CSF assays (tTau) (used as a tTau/Abeta42 ratio) are for use in adults ages 55 and older being evaluated for AD.
They join the Elecsys beta-amyloid (1-42) CSF II (Abeta42) and Elecsys phospho-tau (181P) CSF (pTau181) assays (used as a pTau181/Abeta42 ratio) that received FDA 510(k) clearance in 2022.
“An early and accurate diagnosis can help patients, caregivers and physicians determine a path forward, and the Elecsys CSF assays support diagnosis at early disease stages, when treatment is most effective,” Brad Moore, president and CEO of Roche Diagnostics North America, said in a statement.
Appropriate use recommendations for new and emerging AD drugs call for confirmation of amyloid pathology. Currently, the only FDA-cleared methods to confirm amyloid pathology are CSF tests and PET scans.
“The Elecsys AD CSF assays are concordant with amyloid PET scan imaging and have the potential to provide a more affordable and accessible routine option to confirm the presence of amyloid pathology in the brain,” Roche said.
“They also offer detection of both amyloid and tau biomarkers from one draw, with no radiation and potential to detect Alzheimer’s pathology in early stages of disease,” the company added.
The previously approved Elecsys pTau181/Abeta42 ratio is currently available and the newly approved Elecsys tTau/Abeta42 ratio will be available in the fourth quarter of 2023.
A version of this article first appeared on Medscape.com.
The Elecsys beta-amyloid (1-42) CSF II (Abeta42) and Elecsys total-tau CSF assays (tTau) (used as a tTau/Abeta42 ratio) are for use in adults ages 55 and older being evaluated for AD.
They join the Elecsys beta-amyloid (1-42) CSF II (Abeta42) and Elecsys phospho-tau (181P) CSF (pTau181) assays (used as a pTau181/Abeta42 ratio) that received FDA 510(k) clearance in 2022.
“An early and accurate diagnosis can help patients, caregivers and physicians determine a path forward, and the Elecsys CSF assays support diagnosis at early disease stages, when treatment is most effective,” Brad Moore, president and CEO of Roche Diagnostics North America, said in a statement.
Appropriate use recommendations for new and emerging AD drugs call for confirmation of amyloid pathology. Currently, the only FDA-cleared methods to confirm amyloid pathology are CSF tests and PET scans.
“The Elecsys AD CSF assays are concordant with amyloid PET scan imaging and have the potential to provide a more affordable and accessible routine option to confirm the presence of amyloid pathology in the brain,” Roche said.
“They also offer detection of both amyloid and tau biomarkers from one draw, with no radiation and potential to detect Alzheimer’s pathology in early stages of disease,” the company added.
The previously approved Elecsys pTau181/Abeta42 ratio is currently available and the newly approved Elecsys tTau/Abeta42 ratio will be available in the fourth quarter of 2023.
A version of this article first appeared on Medscape.com.
The Elecsys beta-amyloid (1-42) CSF II (Abeta42) and Elecsys total-tau CSF assays (tTau) (used as a tTau/Abeta42 ratio) are for use in adults ages 55 and older being evaluated for AD.
They join the Elecsys beta-amyloid (1-42) CSF II (Abeta42) and Elecsys phospho-tau (181P) CSF (pTau181) assays (used as a pTau181/Abeta42 ratio) that received FDA 510(k) clearance in 2022.
“An early and accurate diagnosis can help patients, caregivers and physicians determine a path forward, and the Elecsys CSF assays support diagnosis at early disease stages, when treatment is most effective,” Brad Moore, president and CEO of Roche Diagnostics North America, said in a statement.
Appropriate use recommendations for new and emerging AD drugs call for confirmation of amyloid pathology. Currently, the only FDA-cleared methods to confirm amyloid pathology are CSF tests and PET scans.
“The Elecsys AD CSF assays are concordant with amyloid PET scan imaging and have the potential to provide a more affordable and accessible routine option to confirm the presence of amyloid pathology in the brain,” Roche said.
“They also offer detection of both amyloid and tau biomarkers from one draw, with no radiation and potential to detect Alzheimer’s pathology in early stages of disease,” the company added.
The previously approved Elecsys pTau181/Abeta42 ratio is currently available and the newly approved Elecsys tTau/Abeta42 ratio will be available in the fourth quarter of 2023.
A version of this article first appeared on Medscape.com.
New DEA CME mandate affects 2 million prescribers
The Consolidated Appropriations Act of 2023 mandates that all Drug Enforcement Administration–registered physicians and health care providers complete a one-time, 8-hour CME training on managing and treating opioid and other substance abuse disorders. This requirement goes into effect on June 27, 2023. New DEA registrants must also comply. Veterinarians are exempt.
A DEA registration is required to prescribe any controlled substance. The DEA categorizes these as Schedule I-V, with V being the least likely to be abused (Table 1). For example, opioids like fentanyl, oxycodone, and morphine are Schedule II. Medications without abuse potential are not scheduled.
Will 16 million hours of opioid education save lives?
One should not underestimate the sweeping scope of this new federal requirement. DEA registrants include physicians and other health care providers such as nurse practitioners, physician assistants, and dentists. That is 8 hours per provider x 2 million providers: 16 million hours of CME!
Many states already require 1 or more hours of opioid training and pain management as part of their relicensure requirements (Table 2). To avoid redundancy, the DEA-mandated 8-hour training satisfies the various states’ requirements. 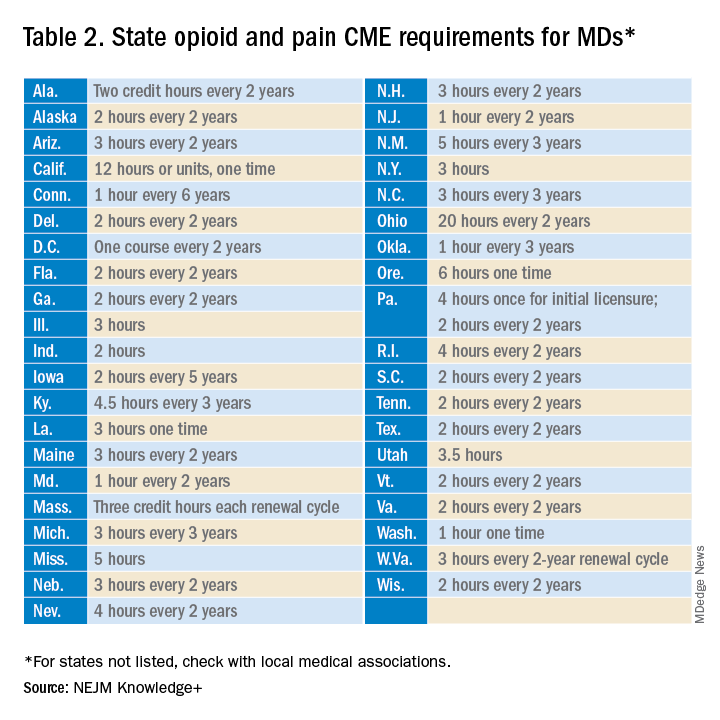
An uncompensated mandate
Physicians are no strangers to lifelong learning and most eagerly pursue educational opportunities. Though some physicians may have CME time and stipends allocated by their employers, many others, such as the approximately 50,000 locum tenens doctors, do not. However, as enthusiastic as these physicians may be about this new CME course, they will likely lose a day of seeing patients (and income) to comply with this new obligation.
Not just pain doctors
The mandate’s broad brush includes many health care providers who hold DEA certificates but do not prescribe opioids. For example, as a general neurologist and epileptologist, I do not treat patients with chronic pain and cannot remember the last time I wrote an opioid prescription. However, I frequently prescribe lacosamide, a Schedule V drug. A surprisingly large number of antiseizure drugs are Schedule III, IV, or V drugs (Table 3).
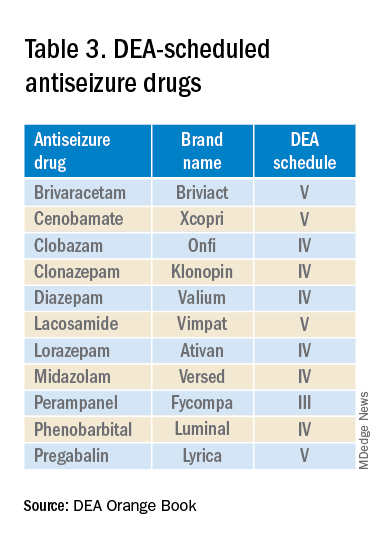
Real-world abuse?
How often scheduled antiseizure drugs are diverted or abused in an epilepsy population is unknown but appears to be infrequent. For example, perampanel abuse has not been reported despite its classification as a Schedule III drug. Anecdotally, in more than 40 years of clinical practice, I have never known a patient with epilepsy to abuse their antiseizure medications.
Take the course
Many organizations are happy to charge for the new 8-hour course. For example, the Tennessee Medical Association offers the training for $299 online or $400 in person. Materials from Elite Learning satisfy the 8-hour requirement for $80. However, NEJM Knowledge+ provides a complimentary 10-hour DEA-compliant course.
I recently completed the NEJM course. The information was thorough and took the whole 10 hours to finish. As excellent as it was, the content was only tangentially relevant to my clinical practice.
Conclusions
To obtain or renew a DEA certificate, neurologists, epilepsy specialists, and many other health care providers must comply with the new 8-hour CME opioid training mandate. Because the course requires 1 day to complete, health care providers would be prudent to obtain their CME well before their DEA certificate expires.
Though efforts to control the morbidity and mortality of the opioid epidemic are laudatory, perhaps the training should be more targeted to physicians who actually prescribe opioids rather than every DEA registrant. In the meantime, whether 16 million CME hours will save lives remains to be seen.
Dr. Wilner is professor of neurology at the University of Tennessee Health Science Center, Memphis. He reported a conflict of interest with Accordant Health Services.
A version of this article first appeared on Medscape.com.
The Consolidated Appropriations Act of 2023 mandates that all Drug Enforcement Administration–registered physicians and health care providers complete a one-time, 8-hour CME training on managing and treating opioid and other substance abuse disorders. This requirement goes into effect on June 27, 2023. New DEA registrants must also comply. Veterinarians are exempt.
A DEA registration is required to prescribe any controlled substance. The DEA categorizes these as Schedule I-V, with V being the least likely to be abused (Table 1). For example, opioids like fentanyl, oxycodone, and morphine are Schedule II. Medications without abuse potential are not scheduled.
Will 16 million hours of opioid education save lives?
One should not underestimate the sweeping scope of this new federal requirement. DEA registrants include physicians and other health care providers such as nurse practitioners, physician assistants, and dentists. That is 8 hours per provider x 2 million providers: 16 million hours of CME!
Many states already require 1 or more hours of opioid training and pain management as part of their relicensure requirements (Table 2). To avoid redundancy, the DEA-mandated 8-hour training satisfies the various states’ requirements. 
An uncompensated mandate
Physicians are no strangers to lifelong learning and most eagerly pursue educational opportunities. Though some physicians may have CME time and stipends allocated by their employers, many others, such as the approximately 50,000 locum tenens doctors, do not. However, as enthusiastic as these physicians may be about this new CME course, they will likely lose a day of seeing patients (and income) to comply with this new obligation.
Not just pain doctors
The mandate’s broad brush includes many health care providers who hold DEA certificates but do not prescribe opioids. For example, as a general neurologist and epileptologist, I do not treat patients with chronic pain and cannot remember the last time I wrote an opioid prescription. However, I frequently prescribe lacosamide, a Schedule V drug. A surprisingly large number of antiseizure drugs are Schedule III, IV, or V drugs (Table 3).

Real-world abuse?
How often scheduled antiseizure drugs are diverted or abused in an epilepsy population is unknown but appears to be infrequent. For example, perampanel abuse has not been reported despite its classification as a Schedule III drug. Anecdotally, in more than 40 years of clinical practice, I have never known a patient with epilepsy to abuse their antiseizure medications.
Take the course
Many organizations are happy to charge for the new 8-hour course. For example, the Tennessee Medical Association offers the training for $299 online or $400 in person. Materials from Elite Learning satisfy the 8-hour requirement for $80. However, NEJM Knowledge+ provides a complimentary 10-hour DEA-compliant course.
I recently completed the NEJM course. The information was thorough and took the whole 10 hours to finish. As excellent as it was, the content was only tangentially relevant to my clinical practice.
Conclusions
To obtain or renew a DEA certificate, neurologists, epilepsy specialists, and many other health care providers must comply with the new 8-hour CME opioid training mandate. Because the course requires 1 day to complete, health care providers would be prudent to obtain their CME well before their DEA certificate expires.
Though efforts to control the morbidity and mortality of the opioid epidemic are laudatory, perhaps the training should be more targeted to physicians who actually prescribe opioids rather than every DEA registrant. In the meantime, whether 16 million CME hours will save lives remains to be seen.
Dr. Wilner is professor of neurology at the University of Tennessee Health Science Center, Memphis. He reported a conflict of interest with Accordant Health Services.
A version of this article first appeared on Medscape.com.
The Consolidated Appropriations Act of 2023 mandates that all Drug Enforcement Administration–registered physicians and health care providers complete a one-time, 8-hour CME training on managing and treating opioid and other substance abuse disorders. This requirement goes into effect on June 27, 2023. New DEA registrants must also comply. Veterinarians are exempt.
A DEA registration is required to prescribe any controlled substance. The DEA categorizes these as Schedule I-V, with V being the least likely to be abused (Table 1). For example, opioids like fentanyl, oxycodone, and morphine are Schedule II. Medications without abuse potential are not scheduled.
Will 16 million hours of opioid education save lives?
One should not underestimate the sweeping scope of this new federal requirement. DEA registrants include physicians and other health care providers such as nurse practitioners, physician assistants, and dentists. That is 8 hours per provider x 2 million providers: 16 million hours of CME!
Many states already require 1 or more hours of opioid training and pain management as part of their relicensure requirements (Table 2). To avoid redundancy, the DEA-mandated 8-hour training satisfies the various states’ requirements. 
An uncompensated mandate
Physicians are no strangers to lifelong learning and most eagerly pursue educational opportunities. Though some physicians may have CME time and stipends allocated by their employers, many others, such as the approximately 50,000 locum tenens doctors, do not. However, as enthusiastic as these physicians may be about this new CME course, they will likely lose a day of seeing patients (and income) to comply with this new obligation.
Not just pain doctors
The mandate’s broad brush includes many health care providers who hold DEA certificates but do not prescribe opioids. For example, as a general neurologist and epileptologist, I do not treat patients with chronic pain and cannot remember the last time I wrote an opioid prescription. However, I frequently prescribe lacosamide, a Schedule V drug. A surprisingly large number of antiseizure drugs are Schedule III, IV, or V drugs (Table 3).

Real-world abuse?
How often scheduled antiseizure drugs are diverted or abused in an epilepsy population is unknown but appears to be infrequent. For example, perampanel abuse has not been reported despite its classification as a Schedule III drug. Anecdotally, in more than 40 years of clinical practice, I have never known a patient with epilepsy to abuse their antiseizure medications.
Take the course
Many organizations are happy to charge for the new 8-hour course. For example, the Tennessee Medical Association offers the training for $299 online or $400 in person. Materials from Elite Learning satisfy the 8-hour requirement for $80. However, NEJM Knowledge+ provides a complimentary 10-hour DEA-compliant course.
I recently completed the NEJM course. The information was thorough and took the whole 10 hours to finish. As excellent as it was, the content was only tangentially relevant to my clinical practice.
Conclusions
To obtain or renew a DEA certificate, neurologists, epilepsy specialists, and many other health care providers must comply with the new 8-hour CME opioid training mandate. Because the course requires 1 day to complete, health care providers would be prudent to obtain their CME well before their DEA certificate expires.
Though efforts to control the morbidity and mortality of the opioid epidemic are laudatory, perhaps the training should be more targeted to physicians who actually prescribe opioids rather than every DEA registrant. In the meantime, whether 16 million CME hours will save lives remains to be seen.
Dr. Wilner is professor of neurology at the University of Tennessee Health Science Center, Memphis. He reported a conflict of interest with Accordant Health Services.
A version of this article first appeared on Medscape.com.
Fade haircut or something else?
The areas of hair loss and different length hairs in a patient with otherwise normal-density hair was consistent with a diagnosis of trichotillomania. The physician initially thought the patient had a “fade” hairstyle, but then observed him twirling his hair. Further queries confirmed a history of hair pulling (trichotillomania) and ingesting the hair (trichophagia).
In adulthood, trichotillomania affects females about 4 times more than males, although in childhood, the sex distribution appears about equal.1 The typical age of onset is between 10 to 13 years. Several mental health conditions are associated with trichotillomania, such as major depressive disorder, anxiety disorders, and substance use disorder, with trichotillomania generally preceding these comorbid disorders. It is important to rule out obsessive compulsive disorder and consider body dysmorphic disorder when making a diagnosis of trichotillomania.1
It is common to see androgenetic alopecia precipitated by hormone therapy in FTM individuals. This would usually cause thinning of the hair rather than the irregular hair lengths and pattern seen in this patient. Tinea capitis is also part of the differential diagnosis in a case such as this. It can be diagnosed by potassium hydroxide (KOH) preparations in the setting of friable and broken hair shafts (and sometimes erythema and inflammation).
Patients with trichotillomania may have hair with blunt or tapered ends; hair may also look like uneven stubble.2 The scalp is the most commonly involved site (72.8%), followed by eyebrows (50.7%), and the pubic region (50.7%).1 Useful diagnostic clues include an unusual shape of the affected area, accessible location, and a changing pattern from visit to visit.2 If trichotillomania is suspected, it might be useful to ask about hair-playing activities, such as twirling or twisting, or playing with the ends of eyelashes or eyebrows.2
Unwanted medical consequences of trichotillomania include skin damage (if sharp instruments are used for hair pulling), and the formation of gastrointestinal trichobezoars (hairballs) if trichophagia is present. Trichobezoars that cause bowel obstructions may need surgical intervention.1
Behavioral therapy is the first-line treatment for trichotillomania in all age groups.1,3 Treating any coexisting mental health disorders is also essential. There are currently no FDA-approved medications for treatment; however, there is evidence that N-acetylcysteine may be useful in treating adults with trichotillomania and other repetitive skin disorders.3 Antipsychotics and cannabinoid agonists also may be beneficial.1
This patient was continued on his current lithium, naltrexone, and bupropion regimen (rather than adding additional psychiatric medicines) as he was doing better than his previous baseline. He was advised to continue with his cognitive behavioral therapy. His hormone replacement therapy also was continued because it was not thought to be contributory. He was provided with education about symptoms of trichobezoar and red flag symptoms (eg, worsening nausea, vomiting, abdominal pain) that would necessitate emergency follow-up. His GERD symptoms were thought not to be related to his trichophagia. He was scheduled to follow up for routine primary care in 6 months.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Sarasawati Keeni, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Grant JE and Chamberlain SR. Trichotillomania. Am J Psychiatry. 2016;173:868-874. doi: 10.1176/appi.ajp.2016.15111432
2. Sah DE, Koo J, Price VH. Trichotillomania. Dermatol Ther. 2008;21:13-21. doi: 10.1111/j.1529-8019.2008.00165.x
3. Henkel ED, Jaquez SD, Diaz LZ. Pediatric trichotillomania: review of management. Pediatr Dermatol. 2019;36:803-807. doi: 10.1111/pde.13954

The areas of hair loss and different length hairs in a patient with otherwise normal-density hair was consistent with a diagnosis of trichotillomania. The physician initially thought the patient had a “fade” hairstyle, but then observed him twirling his hair. Further queries confirmed a history of hair pulling (trichotillomania) and ingesting the hair (trichophagia).
In adulthood, trichotillomania affects females about 4 times more than males, although in childhood, the sex distribution appears about equal.1 The typical age of onset is between 10 to 13 years. Several mental health conditions are associated with trichotillomania, such as major depressive disorder, anxiety disorders, and substance use disorder, with trichotillomania generally preceding these comorbid disorders. It is important to rule out obsessive compulsive disorder and consider body dysmorphic disorder when making a diagnosis of trichotillomania.1
It is common to see androgenetic alopecia precipitated by hormone therapy in FTM individuals. This would usually cause thinning of the hair rather than the irregular hair lengths and pattern seen in this patient. Tinea capitis is also part of the differential diagnosis in a case such as this. It can be diagnosed by potassium hydroxide (KOH) preparations in the setting of friable and broken hair shafts (and sometimes erythema and inflammation).
Patients with trichotillomania may have hair with blunt or tapered ends; hair may also look like uneven stubble.2 The scalp is the most commonly involved site (72.8%), followed by eyebrows (50.7%), and the pubic region (50.7%).1 Useful diagnostic clues include an unusual shape of the affected area, accessible location, and a changing pattern from visit to visit.2 If trichotillomania is suspected, it might be useful to ask about hair-playing activities, such as twirling or twisting, or playing with the ends of eyelashes or eyebrows.2
Unwanted medical consequences of trichotillomania include skin damage (if sharp instruments are used for hair pulling), and the formation of gastrointestinal trichobezoars (hairballs) if trichophagia is present. Trichobezoars that cause bowel obstructions may need surgical intervention.1
Behavioral therapy is the first-line treatment for trichotillomania in all age groups.1,3 Treating any coexisting mental health disorders is also essential. There are currently no FDA-approved medications for treatment; however, there is evidence that N-acetylcysteine may be useful in treating adults with trichotillomania and other repetitive skin disorders.3 Antipsychotics and cannabinoid agonists also may be beneficial.1
This patient was continued on his current lithium, naltrexone, and bupropion regimen (rather than adding additional psychiatric medicines) as he was doing better than his previous baseline. He was advised to continue with his cognitive behavioral therapy. His hormone replacement therapy also was continued because it was not thought to be contributory. He was provided with education about symptoms of trichobezoar and red flag symptoms (eg, worsening nausea, vomiting, abdominal pain) that would necessitate emergency follow-up. His GERD symptoms were thought not to be related to his trichophagia. He was scheduled to follow up for routine primary care in 6 months.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Sarasawati Keeni, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

The areas of hair loss and different length hairs in a patient with otherwise normal-density hair was consistent with a diagnosis of trichotillomania. The physician initially thought the patient had a “fade” hairstyle, but then observed him twirling his hair. Further queries confirmed a history of hair pulling (trichotillomania) and ingesting the hair (trichophagia).
In adulthood, trichotillomania affects females about 4 times more than males, although in childhood, the sex distribution appears about equal.1 The typical age of onset is between 10 to 13 years. Several mental health conditions are associated with trichotillomania, such as major depressive disorder, anxiety disorders, and substance use disorder, with trichotillomania generally preceding these comorbid disorders. It is important to rule out obsessive compulsive disorder and consider body dysmorphic disorder when making a diagnosis of trichotillomania.1
It is common to see androgenetic alopecia precipitated by hormone therapy in FTM individuals. This would usually cause thinning of the hair rather than the irregular hair lengths and pattern seen in this patient. Tinea capitis is also part of the differential diagnosis in a case such as this. It can be diagnosed by potassium hydroxide (KOH) preparations in the setting of friable and broken hair shafts (and sometimes erythema and inflammation).
Patients with trichotillomania may have hair with blunt or tapered ends; hair may also look like uneven stubble.2 The scalp is the most commonly involved site (72.8%), followed by eyebrows (50.7%), and the pubic region (50.7%).1 Useful diagnostic clues include an unusual shape of the affected area, accessible location, and a changing pattern from visit to visit.2 If trichotillomania is suspected, it might be useful to ask about hair-playing activities, such as twirling or twisting, or playing with the ends of eyelashes or eyebrows.2
Unwanted medical consequences of trichotillomania include skin damage (if sharp instruments are used for hair pulling), and the formation of gastrointestinal trichobezoars (hairballs) if trichophagia is present. Trichobezoars that cause bowel obstructions may need surgical intervention.1
Behavioral therapy is the first-line treatment for trichotillomania in all age groups.1,3 Treating any coexisting mental health disorders is also essential. There are currently no FDA-approved medications for treatment; however, there is evidence that N-acetylcysteine may be useful in treating adults with trichotillomania and other repetitive skin disorders.3 Antipsychotics and cannabinoid agonists also may be beneficial.1
This patient was continued on his current lithium, naltrexone, and bupropion regimen (rather than adding additional psychiatric medicines) as he was doing better than his previous baseline. He was advised to continue with his cognitive behavioral therapy. His hormone replacement therapy also was continued because it was not thought to be contributory. He was provided with education about symptoms of trichobezoar and red flag symptoms (eg, worsening nausea, vomiting, abdominal pain) that would necessitate emergency follow-up. His GERD symptoms were thought not to be related to his trichophagia. He was scheduled to follow up for routine primary care in 6 months.
Photo courtesy of Daniel Stulberg, MD. Text courtesy of Sarasawati Keeni, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Grant JE and Chamberlain SR. Trichotillomania. Am J Psychiatry. 2016;173:868-874. doi: 10.1176/appi.ajp.2016.15111432
2. Sah DE, Koo J, Price VH. Trichotillomania. Dermatol Ther. 2008;21:13-21. doi: 10.1111/j.1529-8019.2008.00165.x
3. Henkel ED, Jaquez SD, Diaz LZ. Pediatric trichotillomania: review of management. Pediatr Dermatol. 2019;36:803-807. doi: 10.1111/pde.13954
1. Grant JE and Chamberlain SR. Trichotillomania. Am J Psychiatry. 2016;173:868-874. doi: 10.1176/appi.ajp.2016.15111432
2. Sah DE, Koo J, Price VH. Trichotillomania. Dermatol Ther. 2008;21:13-21. doi: 10.1111/j.1529-8019.2008.00165.x
3. Henkel ED, Jaquez SD, Diaz LZ. Pediatric trichotillomania: review of management. Pediatr Dermatol. 2019;36:803-807. doi: 10.1111/pde.13954