User login
HT, even short-term use, linked to dementia risk in women
Short-term and cyclical use of estrogen and progestin therapy for menopausal symptoms is linked to an increased risk of dementia, results of a large observational study show.
(AD) 20 years later, compared with those who didn’t use HT. The risk was present even in women who used HT for brief periods at menopause onset.
However, both the investigators and experts not involved in the research caution that further studies are needed to explore whether the increased risk of dementia stems from HT use or whether women in need of HT have other underlying dementia risk factors.
“We cannot guarantee that our findings illustrate a causal relationship or if they represent underlying disposition to dementia in women in need of [HT],” lead investigator Nelsan Pourhadi, MD, from the Danish Dementia Research Centre at Copenhagen University Hospital Rigshospitalet, told this news organization.
Still, he added, the findings supported evidence from the Women’s Health Initiative Memory Study (WHIMS), the largest randomized trial on menopausal hormone therapy and dementia.
The findings were published online in BMJ.
Conflicting findings
Before WHIMS was published in 2003, HT was widely prescribed to relieve menopausal symptoms. However, WHIMS, which included more than 4,000 women aged 65 years or older, revealed that HT was associated with a twofold increased risk of dementia.
Studies published since then have yielded mixed results, adding to the controversy surrounding the safety of HT.
To discover whether age of initiation or length of duration of HT affects health outcomes, Dr. Pourhadi and his team undertook the observational study.
Between 2000 and 2018, the researchers tracked more than 60,000 Danish women aged 50-60 years using diagnosis and prescription information from Denmark’s National Registry of Patients.
The registry records showed that nearly 5,600 women developed dementia and 56,000 did not develop dementia. Of the 5,600 women with dementia, 1,460 had a diagnosis of AD.
Nearly 18,000 participants in the study sample received HT – 1,790 (29%) in the dementia group and 16,150 (32%) in the control group. Half started treatment before age 53 years and half stopped within 4 years. Roughly 90% used oral medications, which included a combination of estrogen and progestin.
The median age at which participants started HT was 53 years for both cases and controls, and the median duration of use was 4 years.
Longer use equals greater risk
Compared with those who did not use HT, those who used estrogen-progestin therapy had a 24% increased risk of developing all-cause dementia (hazard ratio, 1.24; 95% confidence interval, 1.17-1.44).
The increased dementia risk was similar between continuous (estrogen and progestin taken daily) and cyclic (daily estrogen with progestin taken 10-14 days a month) treatment regimens.
Longer durations of HT use were associated with increased risk, ranging from a 21% increased risk (HR, 1.21; 95% CI, 1.09-1.35) for those who used it for 1 year or less to a 74% increased risk (HR, 1.74; 95% CI, 1.45-2.10) for use lasting 12 years or more.
Women who started HT between the age of 45 and 50 had a 26% increased risk of developing all-cause dementia (HR, 1.26; 95% CI, 1.13-1.41) while women who initiated HT between age 51 and 60 had a 21% greater risk (HR, 1.21; 95% CI, 1.12-1.29).
Progestin-only or vaginal-estrogen-only therapy was not associated with the development of dementia.
The investigators noted that because this is an observational study, “further studies are warranted to explore if the observed association in this study between menopausal hormone therapy use and increased risk of dementia illustrates a causal effect.”
No causal relationship
In an accompanying editorial, Kejal Kantarci, MD, a professor of radiology at the Mayo Clinic, Rochester, Minn., noted that three clinical trials, including the WHIMS of Younger Women (WHIMS-Y) in 2013, did not show a link between cognitive function and HT.
“Although [Dr.] Pourhadi and colleagues’ study was done carefully using national registries, the observed associations could be artefactual and should not be used to infer a causal relationship between hormone therapy and dementia risk. These findings cannot inform shared decision-making about use of hormone therapy for menopausal symptoms,” she states in the editorial.
Also commenting on the findings, Amanda Heslegrave, PhD, a senior research fellow at the U.K. Dementia Research Institute, London, said in a release from the U.K.’s Science Media Centre that while the study “may cause alarm for women taking [HT], it highlights just how much we still don’t know about the effects of hormones on women’s brain health, and with promising treatments on the horizon, it should be a call to action to make this a priority area of research.”
There was no specific funding for the study. Dr. Kantarci reported working on an unpaid educational activity on Alzheimer’s disease for Biogen and is the PI on a study of a PET imaging ligand for Alzheimer’s disease, to which Eli Lilly and Avid Radiopharmaceuticals donated supplies.
A version of this article originally appeared on Medscape.com.
Short-term and cyclical use of estrogen and progestin therapy for menopausal symptoms is linked to an increased risk of dementia, results of a large observational study show.
(AD) 20 years later, compared with those who didn’t use HT. The risk was present even in women who used HT for brief periods at menopause onset.
However, both the investigators and experts not involved in the research caution that further studies are needed to explore whether the increased risk of dementia stems from HT use or whether women in need of HT have other underlying dementia risk factors.
“We cannot guarantee that our findings illustrate a causal relationship or if they represent underlying disposition to dementia in women in need of [HT],” lead investigator Nelsan Pourhadi, MD, from the Danish Dementia Research Centre at Copenhagen University Hospital Rigshospitalet, told this news organization.
Still, he added, the findings supported evidence from the Women’s Health Initiative Memory Study (WHIMS), the largest randomized trial on menopausal hormone therapy and dementia.
The findings were published online in BMJ.
Conflicting findings
Before WHIMS was published in 2003, HT was widely prescribed to relieve menopausal symptoms. However, WHIMS, which included more than 4,000 women aged 65 years or older, revealed that HT was associated with a twofold increased risk of dementia.
Studies published since then have yielded mixed results, adding to the controversy surrounding the safety of HT.
To discover whether age of initiation or length of duration of HT affects health outcomes, Dr. Pourhadi and his team undertook the observational study.
Between 2000 and 2018, the researchers tracked more than 60,000 Danish women aged 50-60 years using diagnosis and prescription information from Denmark’s National Registry of Patients.
The registry records showed that nearly 5,600 women developed dementia and 56,000 did not develop dementia. Of the 5,600 women with dementia, 1,460 had a diagnosis of AD.
Nearly 18,000 participants in the study sample received HT – 1,790 (29%) in the dementia group and 16,150 (32%) in the control group. Half started treatment before age 53 years and half stopped within 4 years. Roughly 90% used oral medications, which included a combination of estrogen and progestin.
The median age at which participants started HT was 53 years for both cases and controls, and the median duration of use was 4 years.
Longer use equals greater risk
Compared with those who did not use HT, those who used estrogen-progestin therapy had a 24% increased risk of developing all-cause dementia (hazard ratio, 1.24; 95% confidence interval, 1.17-1.44).
The increased dementia risk was similar between continuous (estrogen and progestin taken daily) and cyclic (daily estrogen with progestin taken 10-14 days a month) treatment regimens.
Longer durations of HT use were associated with increased risk, ranging from a 21% increased risk (HR, 1.21; 95% CI, 1.09-1.35) for those who used it for 1 year or less to a 74% increased risk (HR, 1.74; 95% CI, 1.45-2.10) for use lasting 12 years or more.
Women who started HT between the age of 45 and 50 had a 26% increased risk of developing all-cause dementia (HR, 1.26; 95% CI, 1.13-1.41) while women who initiated HT between age 51 and 60 had a 21% greater risk (HR, 1.21; 95% CI, 1.12-1.29).
Progestin-only or vaginal-estrogen-only therapy was not associated with the development of dementia.
The investigators noted that because this is an observational study, “further studies are warranted to explore if the observed association in this study between menopausal hormone therapy use and increased risk of dementia illustrates a causal effect.”
No causal relationship
In an accompanying editorial, Kejal Kantarci, MD, a professor of radiology at the Mayo Clinic, Rochester, Minn., noted that three clinical trials, including the WHIMS of Younger Women (WHIMS-Y) in 2013, did not show a link between cognitive function and HT.
“Although [Dr.] Pourhadi and colleagues’ study was done carefully using national registries, the observed associations could be artefactual and should not be used to infer a causal relationship between hormone therapy and dementia risk. These findings cannot inform shared decision-making about use of hormone therapy for menopausal symptoms,” she states in the editorial.
Also commenting on the findings, Amanda Heslegrave, PhD, a senior research fellow at the U.K. Dementia Research Institute, London, said in a release from the U.K.’s Science Media Centre that while the study “may cause alarm for women taking [HT], it highlights just how much we still don’t know about the effects of hormones on women’s brain health, and with promising treatments on the horizon, it should be a call to action to make this a priority area of research.”
There was no specific funding for the study. Dr. Kantarci reported working on an unpaid educational activity on Alzheimer’s disease for Biogen and is the PI on a study of a PET imaging ligand for Alzheimer’s disease, to which Eli Lilly and Avid Radiopharmaceuticals donated supplies.
A version of this article originally appeared on Medscape.com.
Short-term and cyclical use of estrogen and progestin therapy for menopausal symptoms is linked to an increased risk of dementia, results of a large observational study show.
(AD) 20 years later, compared with those who didn’t use HT. The risk was present even in women who used HT for brief periods at menopause onset.
However, both the investigators and experts not involved in the research caution that further studies are needed to explore whether the increased risk of dementia stems from HT use or whether women in need of HT have other underlying dementia risk factors.
“We cannot guarantee that our findings illustrate a causal relationship or if they represent underlying disposition to dementia in women in need of [HT],” lead investigator Nelsan Pourhadi, MD, from the Danish Dementia Research Centre at Copenhagen University Hospital Rigshospitalet, told this news organization.
Still, he added, the findings supported evidence from the Women’s Health Initiative Memory Study (WHIMS), the largest randomized trial on menopausal hormone therapy and dementia.
The findings were published online in BMJ.
Conflicting findings
Before WHIMS was published in 2003, HT was widely prescribed to relieve menopausal symptoms. However, WHIMS, which included more than 4,000 women aged 65 years or older, revealed that HT was associated with a twofold increased risk of dementia.
Studies published since then have yielded mixed results, adding to the controversy surrounding the safety of HT.
To discover whether age of initiation or length of duration of HT affects health outcomes, Dr. Pourhadi and his team undertook the observational study.
Between 2000 and 2018, the researchers tracked more than 60,000 Danish women aged 50-60 years using diagnosis and prescription information from Denmark’s National Registry of Patients.
The registry records showed that nearly 5,600 women developed dementia and 56,000 did not develop dementia. Of the 5,600 women with dementia, 1,460 had a diagnosis of AD.
Nearly 18,000 participants in the study sample received HT – 1,790 (29%) in the dementia group and 16,150 (32%) in the control group. Half started treatment before age 53 years and half stopped within 4 years. Roughly 90% used oral medications, which included a combination of estrogen and progestin.
The median age at which participants started HT was 53 years for both cases and controls, and the median duration of use was 4 years.
Longer use equals greater risk
Compared with those who did not use HT, those who used estrogen-progestin therapy had a 24% increased risk of developing all-cause dementia (hazard ratio, 1.24; 95% confidence interval, 1.17-1.44).
The increased dementia risk was similar between continuous (estrogen and progestin taken daily) and cyclic (daily estrogen with progestin taken 10-14 days a month) treatment regimens.
Longer durations of HT use were associated with increased risk, ranging from a 21% increased risk (HR, 1.21; 95% CI, 1.09-1.35) for those who used it for 1 year or less to a 74% increased risk (HR, 1.74; 95% CI, 1.45-2.10) for use lasting 12 years or more.
Women who started HT between the age of 45 and 50 had a 26% increased risk of developing all-cause dementia (HR, 1.26; 95% CI, 1.13-1.41) while women who initiated HT between age 51 and 60 had a 21% greater risk (HR, 1.21; 95% CI, 1.12-1.29).
Progestin-only or vaginal-estrogen-only therapy was not associated with the development of dementia.
The investigators noted that because this is an observational study, “further studies are warranted to explore if the observed association in this study between menopausal hormone therapy use and increased risk of dementia illustrates a causal effect.”
No causal relationship
In an accompanying editorial, Kejal Kantarci, MD, a professor of radiology at the Mayo Clinic, Rochester, Minn., noted that three clinical trials, including the WHIMS of Younger Women (WHIMS-Y) in 2013, did not show a link between cognitive function and HT.
“Although [Dr.] Pourhadi and colleagues’ study was done carefully using national registries, the observed associations could be artefactual and should not be used to infer a causal relationship between hormone therapy and dementia risk. These findings cannot inform shared decision-making about use of hormone therapy for menopausal symptoms,” she states in the editorial.
Also commenting on the findings, Amanda Heslegrave, PhD, a senior research fellow at the U.K. Dementia Research Institute, London, said in a release from the U.K.’s Science Media Centre that while the study “may cause alarm for women taking [HT], it highlights just how much we still don’t know about the effects of hormones on women’s brain health, and with promising treatments on the horizon, it should be a call to action to make this a priority area of research.”
There was no specific funding for the study. Dr. Kantarci reported working on an unpaid educational activity on Alzheimer’s disease for Biogen and is the PI on a study of a PET imaging ligand for Alzheimer’s disease, to which Eli Lilly and Avid Radiopharmaceuticals donated supplies.
A version of this article originally appeared on Medscape.com.
Med students, doctor groups react to SCOTUS affirmative action ban
The U.S. Supreme Court ruled on June 29 that using race as a factor in college admissions is unconstitutional, rolling back more than 40 years of affirmative action standards and changing how medical schools evaluate applicants to attract students from diverse backgrounds.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, said in a prepared statement that the Supreme Court ruling will result in a less diverse physician workforce, which is “bad for health care, bad for medicine, and undermines the health of our nation.” He cited the AMA’s recent adoption of a policy advising medical schools to increase enrollment of people from racial and ethnic groups traditionally underrepresented in medicine – even if that means considering race as a factor in admissions criteria.
“Supporting racial and ethnic diversity in the health professions – spanning classrooms, labs, and clinical settings – enriches the educational experiences of all medical and health professions students and the teaching experiences of faculty, and it is essential to improving the overall health of our nation,” the Association of American Medical Colleges (AAMC) said in a prepared statement.
The American Medical Student Association also denounced the Supreme Court decision. “As future physicians committed to justice and equality, we are profoundly outraged ... We strongly support increased representation of minority students in all levels of education, including colleges and medical schools. By fostering diversity and inclusion, institutions have the power to create more empathetic and inclusive learning environments,” the organization said in a press release.
“Diversity in the health care workforce not only benefits underserved patients but improves care for all patients” by increasing understanding and empathy for people of various cultures, Omar T. Atiq, MD, president of the American College of Physicians, said in a press release.
The Supreme Court ruling stems from a lawsuit by the Students for Fair Admissions against Harvard University and the University of North Carolina. The lawsuit alleges that considering race in the college admission process constitutes discrimination and violates the Equal Protection Clause.
Chief Justice John Roberts, who delivered the court’s decision, stated that an applicant’s personal experiences should carry the most weight in admission decisions and that historically, universities have “wrongly concluded that the touchstone of an individual’s identity is not challenges bested, skills built, or lessons learned, but the color of their skin. Our constitutional history does not tolerate that choice.”
Still, Justice Roberts said the opinion does not prohibit universities from considering how race has affected an applicant’s life, “be it through discrimination, inspiration, or otherwise.”
Diversity in medical schools increased last year, with more Black, Hispanic, and female students applying and enrolling. But continued diversity efforts were expected to prove challenging with affirmative action off the table, according to an amicus brief filed last year by the AMA, the AAMC, and dozens of other professional health care organizations.
The brief supported continued use of race in college admissions, stating that eliminating that factor could slow efforts to achieve greater health equity because fewer doctors would be training and working with colleagues from diverse backgrounds.
Several universities with medical programs, such as Yale and Johns Hopkins universities, filed a separate brief citing similar concerns. After the June 29 decision, Harvard and the University of North Carolina released statements stating they would comply with the ruling.
A version of this article first appeared on Medscape.com.
The U.S. Supreme Court ruled on June 29 that using race as a factor in college admissions is unconstitutional, rolling back more than 40 years of affirmative action standards and changing how medical schools evaluate applicants to attract students from diverse backgrounds.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, said in a prepared statement that the Supreme Court ruling will result in a less diverse physician workforce, which is “bad for health care, bad for medicine, and undermines the health of our nation.” He cited the AMA’s recent adoption of a policy advising medical schools to increase enrollment of people from racial and ethnic groups traditionally underrepresented in medicine – even if that means considering race as a factor in admissions criteria.
“Supporting racial and ethnic diversity in the health professions – spanning classrooms, labs, and clinical settings – enriches the educational experiences of all medical and health professions students and the teaching experiences of faculty, and it is essential to improving the overall health of our nation,” the Association of American Medical Colleges (AAMC) said in a prepared statement.
The American Medical Student Association also denounced the Supreme Court decision. “As future physicians committed to justice and equality, we are profoundly outraged ... We strongly support increased representation of minority students in all levels of education, including colleges and medical schools. By fostering diversity and inclusion, institutions have the power to create more empathetic and inclusive learning environments,” the organization said in a press release.
“Diversity in the health care workforce not only benefits underserved patients but improves care for all patients” by increasing understanding and empathy for people of various cultures, Omar T. Atiq, MD, president of the American College of Physicians, said in a press release.
The Supreme Court ruling stems from a lawsuit by the Students for Fair Admissions against Harvard University and the University of North Carolina. The lawsuit alleges that considering race in the college admission process constitutes discrimination and violates the Equal Protection Clause.
Chief Justice John Roberts, who delivered the court’s decision, stated that an applicant’s personal experiences should carry the most weight in admission decisions and that historically, universities have “wrongly concluded that the touchstone of an individual’s identity is not challenges bested, skills built, or lessons learned, but the color of their skin. Our constitutional history does not tolerate that choice.”
Still, Justice Roberts said the opinion does not prohibit universities from considering how race has affected an applicant’s life, “be it through discrimination, inspiration, or otherwise.”
Diversity in medical schools increased last year, with more Black, Hispanic, and female students applying and enrolling. But continued diversity efforts were expected to prove challenging with affirmative action off the table, according to an amicus brief filed last year by the AMA, the AAMC, and dozens of other professional health care organizations.
The brief supported continued use of race in college admissions, stating that eliminating that factor could slow efforts to achieve greater health equity because fewer doctors would be training and working with colleagues from diverse backgrounds.
Several universities with medical programs, such as Yale and Johns Hopkins universities, filed a separate brief citing similar concerns. After the June 29 decision, Harvard and the University of North Carolina released statements stating they would comply with the ruling.
A version of this article first appeared on Medscape.com.
The U.S. Supreme Court ruled on June 29 that using race as a factor in college admissions is unconstitutional, rolling back more than 40 years of affirmative action standards and changing how medical schools evaluate applicants to attract students from diverse backgrounds.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, said in a prepared statement that the Supreme Court ruling will result in a less diverse physician workforce, which is “bad for health care, bad for medicine, and undermines the health of our nation.” He cited the AMA’s recent adoption of a policy advising medical schools to increase enrollment of people from racial and ethnic groups traditionally underrepresented in medicine – even if that means considering race as a factor in admissions criteria.
“Supporting racial and ethnic diversity in the health professions – spanning classrooms, labs, and clinical settings – enriches the educational experiences of all medical and health professions students and the teaching experiences of faculty, and it is essential to improving the overall health of our nation,” the Association of American Medical Colleges (AAMC) said in a prepared statement.
The American Medical Student Association also denounced the Supreme Court decision. “As future physicians committed to justice and equality, we are profoundly outraged ... We strongly support increased representation of minority students in all levels of education, including colleges and medical schools. By fostering diversity and inclusion, institutions have the power to create more empathetic and inclusive learning environments,” the organization said in a press release.
“Diversity in the health care workforce not only benefits underserved patients but improves care for all patients” by increasing understanding and empathy for people of various cultures, Omar T. Atiq, MD, president of the American College of Physicians, said in a press release.
The Supreme Court ruling stems from a lawsuit by the Students for Fair Admissions against Harvard University and the University of North Carolina. The lawsuit alleges that considering race in the college admission process constitutes discrimination and violates the Equal Protection Clause.
Chief Justice John Roberts, who delivered the court’s decision, stated that an applicant’s personal experiences should carry the most weight in admission decisions and that historically, universities have “wrongly concluded that the touchstone of an individual’s identity is not challenges bested, skills built, or lessons learned, but the color of their skin. Our constitutional history does not tolerate that choice.”
Still, Justice Roberts said the opinion does not prohibit universities from considering how race has affected an applicant’s life, “be it through discrimination, inspiration, or otherwise.”
Diversity in medical schools increased last year, with more Black, Hispanic, and female students applying and enrolling. But continued diversity efforts were expected to prove challenging with affirmative action off the table, according to an amicus brief filed last year by the AMA, the AAMC, and dozens of other professional health care organizations.
The brief supported continued use of race in college admissions, stating that eliminating that factor could slow efforts to achieve greater health equity because fewer doctors would be training and working with colleagues from diverse backgrounds.
Several universities with medical programs, such as Yale and Johns Hopkins universities, filed a separate brief citing similar concerns. After the June 29 decision, Harvard and the University of North Carolina released statements stating they would comply with the ruling.
A version of this article first appeared on Medscape.com.
New AHA statement on ischemia after cardiac surgery
The American Heart Association outlines “considerations” on the management of acute postoperative myocardial ischemia (PMI) after cardiac surgery in a scientific statement.
Although an infrequent event, acute PMI following cardiac surgery can rapidly evolve and become a potentially life-threatening complication, the writing group, led by Mario Gaudino, MD, PhD, with Weill Cornell Medicine, New York, points out.
The new statement was published online in Circulation.
Data show that the incidence of postoperative myocardial infarction after cardiac surgery ranges from 0.3% to 9.8% after isolated coronary artery bypass graft (CABG) surgery and 0.7% to 11.8% after concomitant valvular surgery. For isolated mitral valve surgery, incidence ranges from 1.7% to 2.2%.
Short-term mortality is elevated among patients with acute PMI, irrespective of the type of surgery. Reported mortality rates range from 5.1% to 24%; the evidence on long-term mortality has been mixed.
Graft-related factors are the most common cause of PMI after CABG, but other factors may contribute, including technical factors, competitive flow, suture entrapment, or coronary artery distortion, as well as non–graft related factors.
Prompt diagnosis and treatment important
Currently, there is no consensus definition of PMI. Elevations in cardiac biomarkers may not be reliable for diagnosis after surgery, and pain management regimens may mask symptoms of ischemia, the writing group notes.
because timely diagnosis and treatment are key to a good clinical outcome,” they write.
Delay in urgent angiography has been associated with higher mortality; thus, a low threshold for action is encouraged for patients with suspected acute PMI.
Indications for urgent angiography include new ECG changes, chest pain with ongoing signs of ischemia, cardiac imaging abnormalities, cardiac rhythm abnormalities, significant elevations in cardiac biomarkers, and low cardiac output syndrome despite postoperative pressor support.
Patients with acute PMI and low cardiac output syndrome may require mechanical support when first-line treatment fails.
The writing group says fast and effective reperfusion of the ischemic zone, which is generally achieved by percutaneous intervention and, less often, by repeat surgery, is the key to a good clinical outcome.
The statement was prepared by the volunteer writing group on behalf of the AHA Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Stroke Council.
The research had no commercial funding. Disclosures for the writing group are listed with the original article.
A version of this article originally appeared on Medscape.com.
The American Heart Association outlines “considerations” on the management of acute postoperative myocardial ischemia (PMI) after cardiac surgery in a scientific statement.
Although an infrequent event, acute PMI following cardiac surgery can rapidly evolve and become a potentially life-threatening complication, the writing group, led by Mario Gaudino, MD, PhD, with Weill Cornell Medicine, New York, points out.
The new statement was published online in Circulation.
Data show that the incidence of postoperative myocardial infarction after cardiac surgery ranges from 0.3% to 9.8% after isolated coronary artery bypass graft (CABG) surgery and 0.7% to 11.8% after concomitant valvular surgery. For isolated mitral valve surgery, incidence ranges from 1.7% to 2.2%.
Short-term mortality is elevated among patients with acute PMI, irrespective of the type of surgery. Reported mortality rates range from 5.1% to 24%; the evidence on long-term mortality has been mixed.
Graft-related factors are the most common cause of PMI after CABG, but other factors may contribute, including technical factors, competitive flow, suture entrapment, or coronary artery distortion, as well as non–graft related factors.
Prompt diagnosis and treatment important
Currently, there is no consensus definition of PMI. Elevations in cardiac biomarkers may not be reliable for diagnosis after surgery, and pain management regimens may mask symptoms of ischemia, the writing group notes.
because timely diagnosis and treatment are key to a good clinical outcome,” they write.
Delay in urgent angiography has been associated with higher mortality; thus, a low threshold for action is encouraged for patients with suspected acute PMI.
Indications for urgent angiography include new ECG changes, chest pain with ongoing signs of ischemia, cardiac imaging abnormalities, cardiac rhythm abnormalities, significant elevations in cardiac biomarkers, and low cardiac output syndrome despite postoperative pressor support.
Patients with acute PMI and low cardiac output syndrome may require mechanical support when first-line treatment fails.
The writing group says fast and effective reperfusion of the ischemic zone, which is generally achieved by percutaneous intervention and, less often, by repeat surgery, is the key to a good clinical outcome.
The statement was prepared by the volunteer writing group on behalf of the AHA Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Stroke Council.
The research had no commercial funding. Disclosures for the writing group are listed with the original article.
A version of this article originally appeared on Medscape.com.
The American Heart Association outlines “considerations” on the management of acute postoperative myocardial ischemia (PMI) after cardiac surgery in a scientific statement.
Although an infrequent event, acute PMI following cardiac surgery can rapidly evolve and become a potentially life-threatening complication, the writing group, led by Mario Gaudino, MD, PhD, with Weill Cornell Medicine, New York, points out.
The new statement was published online in Circulation.
Data show that the incidence of postoperative myocardial infarction after cardiac surgery ranges from 0.3% to 9.8% after isolated coronary artery bypass graft (CABG) surgery and 0.7% to 11.8% after concomitant valvular surgery. For isolated mitral valve surgery, incidence ranges from 1.7% to 2.2%.
Short-term mortality is elevated among patients with acute PMI, irrespective of the type of surgery. Reported mortality rates range from 5.1% to 24%; the evidence on long-term mortality has been mixed.
Graft-related factors are the most common cause of PMI after CABG, but other factors may contribute, including technical factors, competitive flow, suture entrapment, or coronary artery distortion, as well as non–graft related factors.
Prompt diagnosis and treatment important
Currently, there is no consensus definition of PMI. Elevations in cardiac biomarkers may not be reliable for diagnosis after surgery, and pain management regimens may mask symptoms of ischemia, the writing group notes.
because timely diagnosis and treatment are key to a good clinical outcome,” they write.
Delay in urgent angiography has been associated with higher mortality; thus, a low threshold for action is encouraged for patients with suspected acute PMI.
Indications for urgent angiography include new ECG changes, chest pain with ongoing signs of ischemia, cardiac imaging abnormalities, cardiac rhythm abnormalities, significant elevations in cardiac biomarkers, and low cardiac output syndrome despite postoperative pressor support.
Patients with acute PMI and low cardiac output syndrome may require mechanical support when first-line treatment fails.
The writing group says fast and effective reperfusion of the ischemic zone, which is generally achieved by percutaneous intervention and, less often, by repeat surgery, is the key to a good clinical outcome.
The statement was prepared by the volunteer writing group on behalf of the AHA Council on Cardiovascular Surgery and Anesthesia; Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Stroke Council.
The research had no commercial funding. Disclosures for the writing group are listed with the original article.
A version of this article originally appeared on Medscape.com.
FROM CIRCULATION
Hairy moles may contain the cure for baldness: Study
.
The researchers found that a specific molecule in those hairy moles “causes normally dormant and diminutive hair follicles to activate their stem cells for robust growth of long and thick hairs,” lead researcher Maksim Plikus, PhD, professor of developmental and cell biology at the University of California, Irvine, said in a statement.
The findings could lead to new treatments for the hair loss condition known as androgenetic alopecia, which researchers said occurs in both men and women. It is also known as male-pattern baldness in men.
The global team led by researchers at the university analyzed hair follicle stem cells and discovered that a molecule called osteopontin drives accelerated hair growth. Stem cells can develop into different kinds of cells, whether they are in the body or in a laboratory, and are often involved in regenerative or repair processes, according to the Mayo Clinic.
This latest study, published in the journal Nature, was done on mice. A drug company cofounded by Dr. Plikus said in a news release that it had further tested the hair growth technique on human hair follicles, and “the researchers were able to induce new growth by human hair follicles in a robust preclinical model.” The company, Amplifica, said in the release that it has an exclusive licensing agreement with the university for the new hair growth “inventions” described in the newly published findings.
Hair loss from androgenetic alopecia occurs in two out of every three men, according to the Cleveland Clinic. Amplifica said the condition affects an estimated 50 million men and 30 million women in the United States.
The hair loss and thinning can begin as early as the late teens, the Cleveland Clinic says. The condition is progressive and can follow a specific pattern, such as the hairline creating an “M” or “U” shape midway through the process toward complete baldness on the top of the head, with a remaining thin band of hair around the sides of the head.
A version of this article first appeared on WebMD.com.
.
The researchers found that a specific molecule in those hairy moles “causes normally dormant and diminutive hair follicles to activate their stem cells for robust growth of long and thick hairs,” lead researcher Maksim Plikus, PhD, professor of developmental and cell biology at the University of California, Irvine, said in a statement.
The findings could lead to new treatments for the hair loss condition known as androgenetic alopecia, which researchers said occurs in both men and women. It is also known as male-pattern baldness in men.
The global team led by researchers at the university analyzed hair follicle stem cells and discovered that a molecule called osteopontin drives accelerated hair growth. Stem cells can develop into different kinds of cells, whether they are in the body or in a laboratory, and are often involved in regenerative or repair processes, according to the Mayo Clinic.
This latest study, published in the journal Nature, was done on mice. A drug company cofounded by Dr. Plikus said in a news release that it had further tested the hair growth technique on human hair follicles, and “the researchers were able to induce new growth by human hair follicles in a robust preclinical model.” The company, Amplifica, said in the release that it has an exclusive licensing agreement with the university for the new hair growth “inventions” described in the newly published findings.
Hair loss from androgenetic alopecia occurs in two out of every three men, according to the Cleveland Clinic. Amplifica said the condition affects an estimated 50 million men and 30 million women in the United States.
The hair loss and thinning can begin as early as the late teens, the Cleveland Clinic says. The condition is progressive and can follow a specific pattern, such as the hairline creating an “M” or “U” shape midway through the process toward complete baldness on the top of the head, with a remaining thin band of hair around the sides of the head.
A version of this article first appeared on WebMD.com.
.
The researchers found that a specific molecule in those hairy moles “causes normally dormant and diminutive hair follicles to activate their stem cells for robust growth of long and thick hairs,” lead researcher Maksim Plikus, PhD, professor of developmental and cell biology at the University of California, Irvine, said in a statement.
The findings could lead to new treatments for the hair loss condition known as androgenetic alopecia, which researchers said occurs in both men and women. It is also known as male-pattern baldness in men.
The global team led by researchers at the university analyzed hair follicle stem cells and discovered that a molecule called osteopontin drives accelerated hair growth. Stem cells can develop into different kinds of cells, whether they are in the body or in a laboratory, and are often involved in regenerative or repair processes, according to the Mayo Clinic.
This latest study, published in the journal Nature, was done on mice. A drug company cofounded by Dr. Plikus said in a news release that it had further tested the hair growth technique on human hair follicles, and “the researchers were able to induce new growth by human hair follicles in a robust preclinical model.” The company, Amplifica, said in the release that it has an exclusive licensing agreement with the university for the new hair growth “inventions” described in the newly published findings.
Hair loss from androgenetic alopecia occurs in two out of every three men, according to the Cleveland Clinic. Amplifica said the condition affects an estimated 50 million men and 30 million women in the United States.
The hair loss and thinning can begin as early as the late teens, the Cleveland Clinic says. The condition is progressive and can follow a specific pattern, such as the hairline creating an “M” or “U” shape midway through the process toward complete baldness on the top of the head, with a remaining thin band of hair around the sides of the head.
A version of this article first appeared on WebMD.com.
FROM NATURE
Depression in People With Multiple Sclerosis
Many people believe that a diagnosis of MS and its progression can bring on depression. But is that really true?
This supplement examines the two-way connection between MS and depression. It gives you tools to actively look for signs of depression and develop techniques to more effectively communicate with and manage these individuals.
Many people believe that a diagnosis of MS and its progression can bring on depression. But is that really true?
This supplement examines the two-way connection between MS and depression. It gives you tools to actively look for signs of depression and develop techniques to more effectively communicate with and manage these individuals.
Many people believe that a diagnosis of MS and its progression can bring on depression. But is that really true?
This supplement examines the two-way connection between MS and depression. It gives you tools to actively look for signs of depression and develop techniques to more effectively communicate with and manage these individuals.
Is there a link between esophageal risk and sleep habits?
, suggest researchers at Washington University, St. Louis.
In their prospective study, the team found that individuals who slept more than 9 hours a night had double the risk of getting EAC after 2 or more years (hazard ratio, 2.06) compared with a person sleeping a “standard” 7 hours a night.
Conversely, people who slept fewer than 6 hours were 21% more likely to be diagnosed with EAC and 63% more likely to get ESCC after 2 years.
The research team, led by Yin Cao, ScD, associate professor of surgery, mined UK Biobank data between 2006 and 2016 for the sleep habits of 393,114 people, then followed them for up to 10 years to see how many developed esophageal cancer. During this time, 294 people developed EAC, and 95 were diagnosed with ESCC.
The study was published in Cancer, Epidemiology Biomarkers and Prevention.
Dr. Cao said that she and her team decided to conduct the study because they were intrigued by evidence suggesting a link between shorter sleep duration with ESCC and Barrett esophagus, the precursor of EAC. “Yet other sleep behaviours [had] not been comprehensively evaluated,” Dr. Cao said.
The team also assessed daytime sleep behavior. They found that taking daytime naps or experiencing sleepiness did not bode well down the road with regard to esophageal cancer, depending on the type of cancer. Individuals who “sometimes” took daytime naps were at 39% increased risk of EAC, and those who “usually” experienced sleepiness during the day had twice the risk of ESCC after 2 years.
Chronotype, a person’s natural rhythm of daytime sleepiness, also appeared to play a role. For example, being an “evening person” increased the risk of EAC by 32% and almost tripled the risk of ESCC compared to being a “morning person.”
Neither snoring nor insomnia affected the risk of either type of esophageal cancer.
Dr. Cao was surprised that both short and long sleep duration, napping, and daytime sleepiness turned out to be risk factors for esophageal cancer.
The mechanisms underlying these associations are “likely complex,” she said, adding that different aspects of sleep behavior might be driving cancer through differing routes.
“Disruption of circadian physiology could result in gastroesophageal reflux disease (GERD), the most important risk factor for EAC, via altering the expression of circadian-clock genes in esophagus tissue or reducing the expression of melatonin,” said Dr. Cao. “Both excess sleep and sleep deprivation might relate to immune dysfunction, which might lead to an increased susceptibility to esophageal cancer.”
In an interview, when asked whether other, simpler explanations were possible, Dr. Cao acknowledged that other associations were a potential source of confusion. For example, obesity is known to affect both quality of sleep and cancer risk. Dr. Cao said in their analyses, the team adjusted for a myriad of likely confounders, such as sex, smoking status, and shift-work status, in addition to body mass index and the Townsend Deprivation Index, a poverty measure.
Another factor that could have muddied the results was reverse causation, the possibility that already-present esophageal cancer was causing fatigue and poor sleep patterns instead of the other way around. For this reason, Dr. Cao’s team conducted a separate analysis that excluded patients who were diagnosed with esophageal cancer during the first 2 years of follow-up. (Data from this separate analysis, available in the supplementary tables, were used for the current article.)
Dr. Cao concluded that her team’s data “may serve as modifying risk factors for esophageal adenoma carcinoma” but that “additional validation and mechanistic studies are needed.”
The study was supported by a National Institutes of Health grant. Dr. Cao has received personal fees from Geneoscopy outside the published work.
A version of this article originally appeared on Medscape.com.
, suggest researchers at Washington University, St. Louis.
In their prospective study, the team found that individuals who slept more than 9 hours a night had double the risk of getting EAC after 2 or more years (hazard ratio, 2.06) compared with a person sleeping a “standard” 7 hours a night.
Conversely, people who slept fewer than 6 hours were 21% more likely to be diagnosed with EAC and 63% more likely to get ESCC after 2 years.
The research team, led by Yin Cao, ScD, associate professor of surgery, mined UK Biobank data between 2006 and 2016 for the sleep habits of 393,114 people, then followed them for up to 10 years to see how many developed esophageal cancer. During this time, 294 people developed EAC, and 95 were diagnosed with ESCC.
The study was published in Cancer, Epidemiology Biomarkers and Prevention.
Dr. Cao said that she and her team decided to conduct the study because they were intrigued by evidence suggesting a link between shorter sleep duration with ESCC and Barrett esophagus, the precursor of EAC. “Yet other sleep behaviours [had] not been comprehensively evaluated,” Dr. Cao said.
The team also assessed daytime sleep behavior. They found that taking daytime naps or experiencing sleepiness did not bode well down the road with regard to esophageal cancer, depending on the type of cancer. Individuals who “sometimes” took daytime naps were at 39% increased risk of EAC, and those who “usually” experienced sleepiness during the day had twice the risk of ESCC after 2 years.
Chronotype, a person’s natural rhythm of daytime sleepiness, also appeared to play a role. For example, being an “evening person” increased the risk of EAC by 32% and almost tripled the risk of ESCC compared to being a “morning person.”
Neither snoring nor insomnia affected the risk of either type of esophageal cancer.
Dr. Cao was surprised that both short and long sleep duration, napping, and daytime sleepiness turned out to be risk factors for esophageal cancer.
The mechanisms underlying these associations are “likely complex,” she said, adding that different aspects of sleep behavior might be driving cancer through differing routes.
“Disruption of circadian physiology could result in gastroesophageal reflux disease (GERD), the most important risk factor for EAC, via altering the expression of circadian-clock genes in esophagus tissue or reducing the expression of melatonin,” said Dr. Cao. “Both excess sleep and sleep deprivation might relate to immune dysfunction, which might lead to an increased susceptibility to esophageal cancer.”
In an interview, when asked whether other, simpler explanations were possible, Dr. Cao acknowledged that other associations were a potential source of confusion. For example, obesity is known to affect both quality of sleep and cancer risk. Dr. Cao said in their analyses, the team adjusted for a myriad of likely confounders, such as sex, smoking status, and shift-work status, in addition to body mass index and the Townsend Deprivation Index, a poverty measure.
Another factor that could have muddied the results was reverse causation, the possibility that already-present esophageal cancer was causing fatigue and poor sleep patterns instead of the other way around. For this reason, Dr. Cao’s team conducted a separate analysis that excluded patients who were diagnosed with esophageal cancer during the first 2 years of follow-up. (Data from this separate analysis, available in the supplementary tables, were used for the current article.)
Dr. Cao concluded that her team’s data “may serve as modifying risk factors for esophageal adenoma carcinoma” but that “additional validation and mechanistic studies are needed.”
The study was supported by a National Institutes of Health grant. Dr. Cao has received personal fees from Geneoscopy outside the published work.
A version of this article originally appeared on Medscape.com.
, suggest researchers at Washington University, St. Louis.
In their prospective study, the team found that individuals who slept more than 9 hours a night had double the risk of getting EAC after 2 or more years (hazard ratio, 2.06) compared with a person sleeping a “standard” 7 hours a night.
Conversely, people who slept fewer than 6 hours were 21% more likely to be diagnosed with EAC and 63% more likely to get ESCC after 2 years.
The research team, led by Yin Cao, ScD, associate professor of surgery, mined UK Biobank data between 2006 and 2016 for the sleep habits of 393,114 people, then followed them for up to 10 years to see how many developed esophageal cancer. During this time, 294 people developed EAC, and 95 were diagnosed with ESCC.
The study was published in Cancer, Epidemiology Biomarkers and Prevention.
Dr. Cao said that she and her team decided to conduct the study because they were intrigued by evidence suggesting a link between shorter sleep duration with ESCC and Barrett esophagus, the precursor of EAC. “Yet other sleep behaviours [had] not been comprehensively evaluated,” Dr. Cao said.
The team also assessed daytime sleep behavior. They found that taking daytime naps or experiencing sleepiness did not bode well down the road with regard to esophageal cancer, depending on the type of cancer. Individuals who “sometimes” took daytime naps were at 39% increased risk of EAC, and those who “usually” experienced sleepiness during the day had twice the risk of ESCC after 2 years.
Chronotype, a person’s natural rhythm of daytime sleepiness, also appeared to play a role. For example, being an “evening person” increased the risk of EAC by 32% and almost tripled the risk of ESCC compared to being a “morning person.”
Neither snoring nor insomnia affected the risk of either type of esophageal cancer.
Dr. Cao was surprised that both short and long sleep duration, napping, and daytime sleepiness turned out to be risk factors for esophageal cancer.
The mechanisms underlying these associations are “likely complex,” she said, adding that different aspects of sleep behavior might be driving cancer through differing routes.
“Disruption of circadian physiology could result in gastroesophageal reflux disease (GERD), the most important risk factor for EAC, via altering the expression of circadian-clock genes in esophagus tissue or reducing the expression of melatonin,” said Dr. Cao. “Both excess sleep and sleep deprivation might relate to immune dysfunction, which might lead to an increased susceptibility to esophageal cancer.”
In an interview, when asked whether other, simpler explanations were possible, Dr. Cao acknowledged that other associations were a potential source of confusion. For example, obesity is known to affect both quality of sleep and cancer risk. Dr. Cao said in their analyses, the team adjusted for a myriad of likely confounders, such as sex, smoking status, and shift-work status, in addition to body mass index and the Townsend Deprivation Index, a poverty measure.
Another factor that could have muddied the results was reverse causation, the possibility that already-present esophageal cancer was causing fatigue and poor sleep patterns instead of the other way around. For this reason, Dr. Cao’s team conducted a separate analysis that excluded patients who were diagnosed with esophageal cancer during the first 2 years of follow-up. (Data from this separate analysis, available in the supplementary tables, were used for the current article.)
Dr. Cao concluded that her team’s data “may serve as modifying risk factors for esophageal adenoma carcinoma” but that “additional validation and mechanistic studies are needed.”
The study was supported by a National Institutes of Health grant. Dr. Cao has received personal fees from Geneoscopy outside the published work.
A version of this article originally appeared on Medscape.com.
Improving Diagnostic Accuracy in Skin of Color Using an Educational Module
Dermatologic disparities disproportionately affect patients with skin of color (SOC). Two studies assessing the diagnostic accuracy of medical students have shown disparities in diagnosing common skin conditions presenting in darker skin compared to lighter skin at early stages of training.1,2 This knowledge gap could be attributed to the underrepresentation of SOC in dermatologic textbooks, journals, and educational curricula.3-6 It is important for dermatologists as well as physicians in other specialties and ancillary health care workers involved in treating or triaging dermatologic diseases to recognize common skin conditions presenting in SOC. We sought to evaluate the effectiveness of a focused educational module for improving diagnostic accuracy and confidence in treating SOC among interprofessional health care providers.
Methods
Interprofessional health care providers—medical students, residents/fellows, attending physicians, advanced practice providers (APPs), and nurses practicing across various medical specialties—at The University of Texas at Austin Dell Medical School and Ascension Medical Group (both in Austin, Texas) were invited to participate in an institutional review board–exempt study involving a virtual SOC educational module from February through May 2021. The 1-hour module involved a pretest, a 15-minute lecture, an immediate posttest, and a 3-month posttest. All tests included the same 40 multiple-choice questions of 20 dermatologic conditions portrayed in lighter and darker skin types from VisualDx.com, and participants were asked to identify the condition in each photograph. Questions appeared one at a time in a randomized order, and answers could not be changed once submitted.
For analysis, the dermatologic conditions were categorized into 4 groups: cancerous, infectious, inflammatory, and SOC-associated conditions. Cancerous conditions included basal cell carcinoma, squamous cell carcinoma, and melanoma. Infectious conditions included herpes zoster, tinea corporis, tinea versicolor, staphylococcal scalded skin syndrome, and verruca vulgaris. Inflammatory conditions included acne, atopic dermatitis, pityriasis rosea, psoriasis, seborrheic dermatitis, contact dermatitis, lichen planus, and urticaria. Skin of color–associated conditions included hidradenitis suppurativa, acanthosis nigricans, keloid, and melasma. Two questions utilizing a 5-point Likert scale assessing confidence in diagnosing light and dark skin also were included.
The pre-recorded 15-minute video lecture was given by 2 dermatology residents (P.L.K. and C.P.), and the learning objectives covered morphologic differences in lighter skin and darker skin, comparisons of common dermatologic diseases in lighter skin and darker skin, diseases more commonly affecting patients with SOC, and treatment considerations for conditions affecting skin and hair in patients with SOC. Photographs from the diagnostic accuracy assessment were not reused in the lecture. Detailed explanations on morphology, diagnostic pearls, and treatment options for all conditions tested were provided to participants upon completion of the 3-month posttest.
Statistical Analysis—Test scores were compared between conditions shown in lighter and darker skin types and from the pretest to the immediate posttest and 3-month posttest. Multiple linear regression was used to assess for intervention effects on lighter and darker skin scores controlling for provider type and specialty. All tests were 2-sided with significance at P<.05. Analyses were conducted using Stata 17.
Results
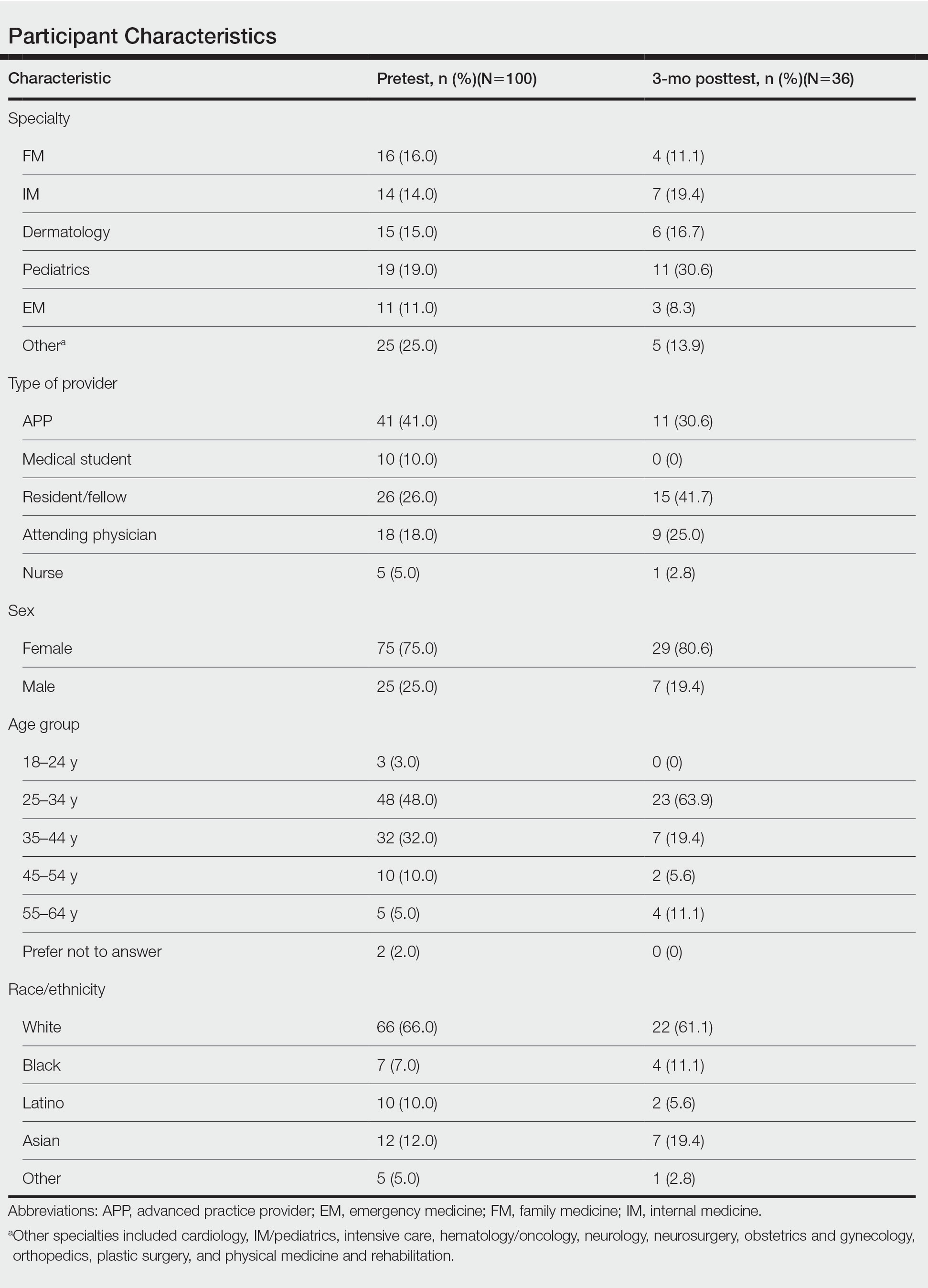
One hundred participants completed the pretest and immediate posttest, 36 of whom also completed the 3-month posttest (Table). There was no significant difference in baseline characteristics between the pretest and 3-month posttest groups.
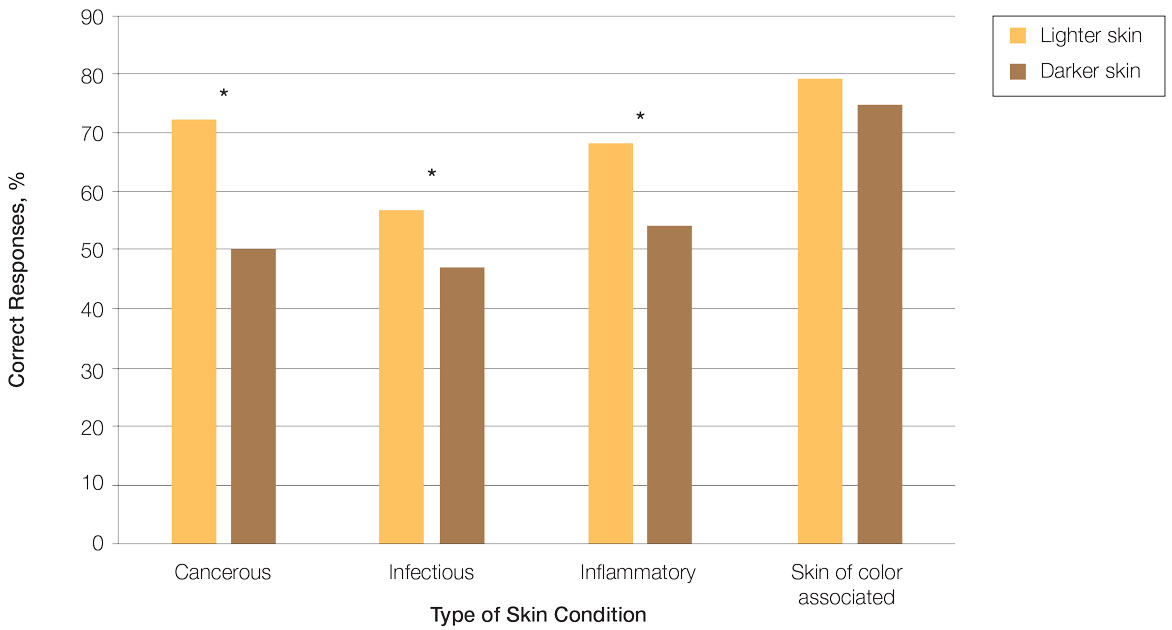
Test scores were correlated with provider type and specialty but not age, sex, or race/ethnicity. Specializing in dermatology and being a resident or attending physician were independently associated with higher test scores. Mean pretest diagnostic accuracy and confidence scores were higher for skin conditions shown in lighter skin compared with those shown in darker skin (13.6 vs 11.3 and 2.7 vs 1.9, respectively; both P<.001). Pretest diagnostic accuracy was significantly higher for skin conditions shown in lighter skin compared with darker skin for cancerous, inflammatory, and infectious conditions (72% vs 50%, 68% vs 55%, and 57% vs 47%, respectively; P<.001 for all)(Figure 1). Skin of color–associated conditions were not associated with significantly different scores for lighter skin compared with darker skin (79% vs 75%; P=.059).
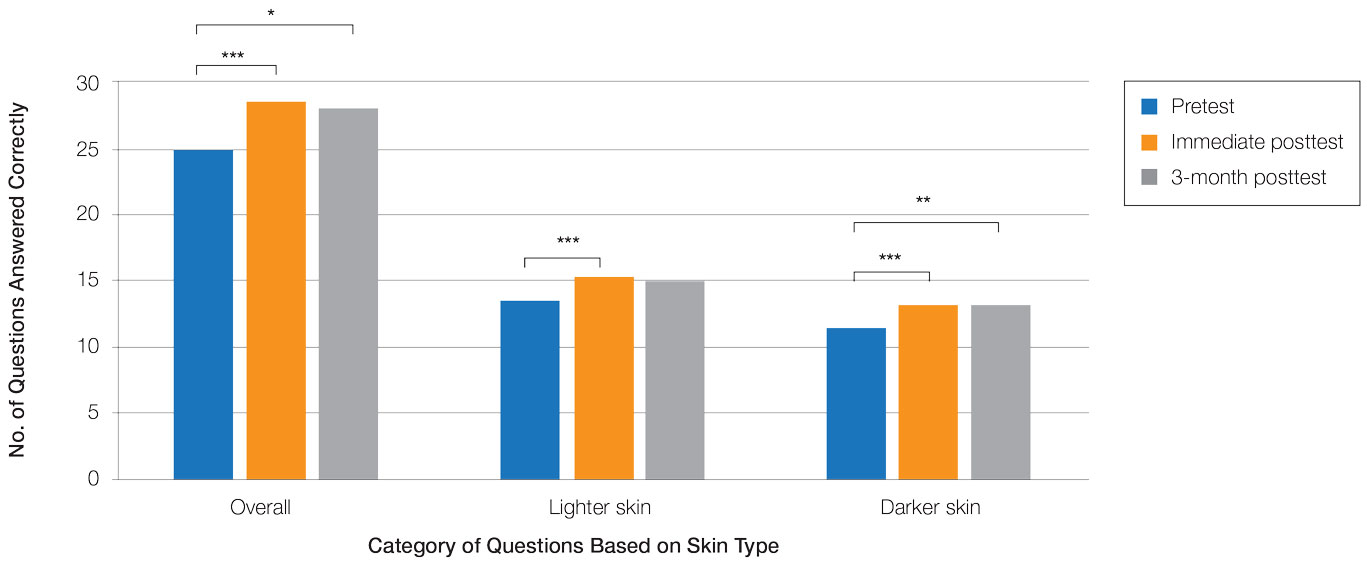
Controlling for provider type and specialty, significantly improved diagnostic accuracy was seen in immediate posttest scores compared with pretest scores for conditions shown in both lighter and darker skin types (lighter: 15.2 vs 13.6; darker: 13.3 vs 11.3; both P<.001)(Figure 2). The immediate posttest demonstrated higher mean diagnostic accuracy and confidence scores for skin conditions shown in lighter skin compared with darker skin (diagnostic accuracy: 15.2 vs 13.3; confidence: 3.0 vs 2.6; both P<.001), but the disparity between scores was less than in the pretest.
Following the 3-month posttest, improvement in diagnostic accuracy was noted among both lighter and darker skin types compared with the pretest, but the difference remained significant only for conditions shown in darker skin (mean scores, 11.3 vs 13.3; P<.01). Similarly, confidence in diagnosing conditions in both lighter and darker skin improved following the immediate posttest (mean scores, 2.7 vs 3.0 and 1.9 vs 2.6; both P<.001), and this improvement remained significant for only darker skin following the 3-month posttest (mean scores, 1.9 vs 2.3; P<.001). Despite these improvements, diagnostic accuracy and confidence remained higher for skin conditions shown in lighter skin compared with darker skin (diagnostic accuracy: 14.7 vs 13.3; P<.01; confidence: 2.8 vs 2.3; P<.001), though the disparity between scores was again less than in the pretest.
Comment
Our study showed that there are diagnostic disparities between lighter and darker skin types among interprofessional health care providers. Education on SOC should extend to interprofessional health care providers and other medical specialties involved in treating or triaging dermatologic diseases. A focused educational module may provide long-term improvements in diagnostic accuracy and confidence for conditions presenting in SOC. Differences in diagnostic accuracy between conditions shown in lighter and darker skin types were noted for the disease categories of infectious, cancerous, and inflammatory conditions, with the exception of conditions more frequently seen in patients with SOC. Learning resources for SOC-associated conditions are more likely to have greater representation of images depicting darker skin types.7 Future educational interventions may need to focus on dermatologic conditions that are not preferentially seen in patients with SOC. In our study, the pretest scores for conditions shown in darker skin were lowest among infectious and cancerous conditions. For infections, certain morphologic clues such as erythema are important for diagnosis but may be more subtle or difficult to discern in darker skin. It also is possible that providers may be less likely to suspect skin cancer in patients with SOC given that the morphologic presentation and/or anatomic site of involvement for skin cancers in SOC differs from those in lighter skin. Future educational interventions targeting disparities in diagnostic accuracy should focus on conditions that are not specifically associated with SOC.
Limitations of our study included the small number of participants, the study population came from a single institution, and a possible selection bias for providers interested in dermatology.
Conclusion
Disparities exist among interprofessional health care providers when treating conditions in patients with lighter skin compared to darker skin. An educational module for health care providers may provide long-term improvements in diagnostic accuracy and confidence for conditions presenting in patients with SOC.
- Fenton A, Elliott E, Shahbandi A, et al. Medical students’ ability to diagnose common dermatologic conditions in skin of color. J Am Acad Dermatol. 2020;83:957-958. doi:10.1016/j.jaad.2019.12.078
- Mamo A, Szeto MD, Rietcheck H, et al. Evaluating medical student assessment of common dermatologic conditions across Fitzpatrick phototypes and skin of color. J Am Acad Dermatol. 2022;87:167-169. doi:10.1016/j.jaad.2021.06.868
- Guda VA, Paek SY. Skin of color representation in commonly utilized medical student dermatology resources. J Drugs Dermatol. 2021;20:799. doi:10.36849/JDD.5726
- Wilson BN, Sun M, Ashbaugh AG, et al. Assessment of skin of color and diversity and inclusion content of dermatologic published literature: an analysis and call to action. Int J Womens Dermatol. 2021;7:391-397. doi:10.1016/j.ijwd.2021.04.001
- Ibraheim MK, Gupta R, Dao H, et al. Evaluating skin of color education in dermatology residency programs: data from a national survey. Clin Dermatol. 2022;40:228-233. doi:10.1016/j.clindermatol.2021.11.015
- Gupta R, Ibraheim MK, Dao H Jr, et al. Assessing dermatology resident confidence in caring for patients with skin of color. Clin Dermatol. 2021;39:873-878. doi:10.1016/j.clindermatol.2021.08.019
- Chang MJ, Lipner SR. Analysis of skin color on the American Academy of Dermatology public education website. J Drugs Dermatol. 2020;19:1236-1237. doi:10.36849/JDD.2020.5545
Dermatologic disparities disproportionately affect patients with skin of color (SOC). Two studies assessing the diagnostic accuracy of medical students have shown disparities in diagnosing common skin conditions presenting in darker skin compared to lighter skin at early stages of training.1,2 This knowledge gap could be attributed to the underrepresentation of SOC in dermatologic textbooks, journals, and educational curricula.3-6 It is important for dermatologists as well as physicians in other specialties and ancillary health care workers involved in treating or triaging dermatologic diseases to recognize common skin conditions presenting in SOC. We sought to evaluate the effectiveness of a focused educational module for improving diagnostic accuracy and confidence in treating SOC among interprofessional health care providers.
Methods
Interprofessional health care providers—medical students, residents/fellows, attending physicians, advanced practice providers (APPs), and nurses practicing across various medical specialties—at The University of Texas at Austin Dell Medical School and Ascension Medical Group (both in Austin, Texas) were invited to participate in an institutional review board–exempt study involving a virtual SOC educational module from February through May 2021. The 1-hour module involved a pretest, a 15-minute lecture, an immediate posttest, and a 3-month posttest. All tests included the same 40 multiple-choice questions of 20 dermatologic conditions portrayed in lighter and darker skin types from VisualDx.com, and participants were asked to identify the condition in each photograph. Questions appeared one at a time in a randomized order, and answers could not be changed once submitted.
For analysis, the dermatologic conditions were categorized into 4 groups: cancerous, infectious, inflammatory, and SOC-associated conditions. Cancerous conditions included basal cell carcinoma, squamous cell carcinoma, and melanoma. Infectious conditions included herpes zoster, tinea corporis, tinea versicolor, staphylococcal scalded skin syndrome, and verruca vulgaris. Inflammatory conditions included acne, atopic dermatitis, pityriasis rosea, psoriasis, seborrheic dermatitis, contact dermatitis, lichen planus, and urticaria. Skin of color–associated conditions included hidradenitis suppurativa, acanthosis nigricans, keloid, and melasma. Two questions utilizing a 5-point Likert scale assessing confidence in diagnosing light and dark skin also were included.
The pre-recorded 15-minute video lecture was given by 2 dermatology residents (P.L.K. and C.P.), and the learning objectives covered morphologic differences in lighter skin and darker skin, comparisons of common dermatologic diseases in lighter skin and darker skin, diseases more commonly affecting patients with SOC, and treatment considerations for conditions affecting skin and hair in patients with SOC. Photographs from the diagnostic accuracy assessment were not reused in the lecture. Detailed explanations on morphology, diagnostic pearls, and treatment options for all conditions tested were provided to participants upon completion of the 3-month posttest.
Statistical Analysis—Test scores were compared between conditions shown in lighter and darker skin types and from the pretest to the immediate posttest and 3-month posttest. Multiple linear regression was used to assess for intervention effects on lighter and darker skin scores controlling for provider type and specialty. All tests were 2-sided with significance at P<.05. Analyses were conducted using Stata 17.
Results
One hundred participants completed the pretest and immediate posttest, 36 of whom also completed the 3-month posttest (Table). There was no significant difference in baseline characteristics between the pretest and 3-month posttest groups.

Test scores were correlated with provider type and specialty but not age, sex, or race/ethnicity. Specializing in dermatology and being a resident or attending physician were independently associated with higher test scores. Mean pretest diagnostic accuracy and confidence scores were higher for skin conditions shown in lighter skin compared with those shown in darker skin (13.6 vs 11.3 and 2.7 vs 1.9, respectively; both P<.001). Pretest diagnostic accuracy was significantly higher for skin conditions shown in lighter skin compared with darker skin for cancerous, inflammatory, and infectious conditions (72% vs 50%, 68% vs 55%, and 57% vs 47%, respectively; P<.001 for all)(Figure 1). Skin of color–associated conditions were not associated with significantly different scores for lighter skin compared with darker skin (79% vs 75%; P=.059).

Controlling for provider type and specialty, significantly improved diagnostic accuracy was seen in immediate posttest scores compared with pretest scores for conditions shown in both lighter and darker skin types (lighter: 15.2 vs 13.6; darker: 13.3 vs 11.3; both P<.001)(Figure 2). The immediate posttest demonstrated higher mean diagnostic accuracy and confidence scores for skin conditions shown in lighter skin compared with darker skin (diagnostic accuracy: 15.2 vs 13.3; confidence: 3.0 vs 2.6; both P<.001), but the disparity between scores was less than in the pretest.

Following the 3-month posttest, improvement in diagnostic accuracy was noted among both lighter and darker skin types compared with the pretest, but the difference remained significant only for conditions shown in darker skin (mean scores, 11.3 vs 13.3; P<.01). Similarly, confidence in diagnosing conditions in both lighter and darker skin improved following the immediate posttest (mean scores, 2.7 vs 3.0 and 1.9 vs 2.6; both P<.001), and this improvement remained significant for only darker skin following the 3-month posttest (mean scores, 1.9 vs 2.3; P<.001). Despite these improvements, diagnostic accuracy and confidence remained higher for skin conditions shown in lighter skin compared with darker skin (diagnostic accuracy: 14.7 vs 13.3; P<.01; confidence: 2.8 vs 2.3; P<.001), though the disparity between scores was again less than in the pretest.
Comment
Our study showed that there are diagnostic disparities between lighter and darker skin types among interprofessional health care providers. Education on SOC should extend to interprofessional health care providers and other medical specialties involved in treating or triaging dermatologic diseases. A focused educational module may provide long-term improvements in diagnostic accuracy and confidence for conditions presenting in SOC. Differences in diagnostic accuracy between conditions shown in lighter and darker skin types were noted for the disease categories of infectious, cancerous, and inflammatory conditions, with the exception of conditions more frequently seen in patients with SOC. Learning resources for SOC-associated conditions are more likely to have greater representation of images depicting darker skin types.7 Future educational interventions may need to focus on dermatologic conditions that are not preferentially seen in patients with SOC. In our study, the pretest scores for conditions shown in darker skin were lowest among infectious and cancerous conditions. For infections, certain morphologic clues such as erythema are important for diagnosis but may be more subtle or difficult to discern in darker skin. It also is possible that providers may be less likely to suspect skin cancer in patients with SOC given that the morphologic presentation and/or anatomic site of involvement for skin cancers in SOC differs from those in lighter skin. Future educational interventions targeting disparities in diagnostic accuracy should focus on conditions that are not specifically associated with SOC.
Limitations of our study included the small number of participants, the study population came from a single institution, and a possible selection bias for providers interested in dermatology.
Conclusion
Disparities exist among interprofessional health care providers when treating conditions in patients with lighter skin compared to darker skin. An educational module for health care providers may provide long-term improvements in diagnostic accuracy and confidence for conditions presenting in patients with SOC.
Dermatologic disparities disproportionately affect patients with skin of color (SOC). Two studies assessing the diagnostic accuracy of medical students have shown disparities in diagnosing common skin conditions presenting in darker skin compared to lighter skin at early stages of training.1,2 This knowledge gap could be attributed to the underrepresentation of SOC in dermatologic textbooks, journals, and educational curricula.3-6 It is important for dermatologists as well as physicians in other specialties and ancillary health care workers involved in treating or triaging dermatologic diseases to recognize common skin conditions presenting in SOC. We sought to evaluate the effectiveness of a focused educational module for improving diagnostic accuracy and confidence in treating SOC among interprofessional health care providers.
Methods
Interprofessional health care providers—medical students, residents/fellows, attending physicians, advanced practice providers (APPs), and nurses practicing across various medical specialties—at The University of Texas at Austin Dell Medical School and Ascension Medical Group (both in Austin, Texas) were invited to participate in an institutional review board–exempt study involving a virtual SOC educational module from February through May 2021. The 1-hour module involved a pretest, a 15-minute lecture, an immediate posttest, and a 3-month posttest. All tests included the same 40 multiple-choice questions of 20 dermatologic conditions portrayed in lighter and darker skin types from VisualDx.com, and participants were asked to identify the condition in each photograph. Questions appeared one at a time in a randomized order, and answers could not be changed once submitted.
For analysis, the dermatologic conditions were categorized into 4 groups: cancerous, infectious, inflammatory, and SOC-associated conditions. Cancerous conditions included basal cell carcinoma, squamous cell carcinoma, and melanoma. Infectious conditions included herpes zoster, tinea corporis, tinea versicolor, staphylococcal scalded skin syndrome, and verruca vulgaris. Inflammatory conditions included acne, atopic dermatitis, pityriasis rosea, psoriasis, seborrheic dermatitis, contact dermatitis, lichen planus, and urticaria. Skin of color–associated conditions included hidradenitis suppurativa, acanthosis nigricans, keloid, and melasma. Two questions utilizing a 5-point Likert scale assessing confidence in diagnosing light and dark skin also were included.
The pre-recorded 15-minute video lecture was given by 2 dermatology residents (P.L.K. and C.P.), and the learning objectives covered morphologic differences in lighter skin and darker skin, comparisons of common dermatologic diseases in lighter skin and darker skin, diseases more commonly affecting patients with SOC, and treatment considerations for conditions affecting skin and hair in patients with SOC. Photographs from the diagnostic accuracy assessment were not reused in the lecture. Detailed explanations on morphology, diagnostic pearls, and treatment options for all conditions tested were provided to participants upon completion of the 3-month posttest.
Statistical Analysis—Test scores were compared between conditions shown in lighter and darker skin types and from the pretest to the immediate posttest and 3-month posttest. Multiple linear regression was used to assess for intervention effects on lighter and darker skin scores controlling for provider type and specialty. All tests were 2-sided with significance at P<.05. Analyses were conducted using Stata 17.
Results
One hundred participants completed the pretest and immediate posttest, 36 of whom also completed the 3-month posttest (Table). There was no significant difference in baseline characteristics between the pretest and 3-month posttest groups.

Test scores were correlated with provider type and specialty but not age, sex, or race/ethnicity. Specializing in dermatology and being a resident or attending physician were independently associated with higher test scores. Mean pretest diagnostic accuracy and confidence scores were higher for skin conditions shown in lighter skin compared with those shown in darker skin (13.6 vs 11.3 and 2.7 vs 1.9, respectively; both P<.001). Pretest diagnostic accuracy was significantly higher for skin conditions shown in lighter skin compared with darker skin for cancerous, inflammatory, and infectious conditions (72% vs 50%, 68% vs 55%, and 57% vs 47%, respectively; P<.001 for all)(Figure 1). Skin of color–associated conditions were not associated with significantly different scores for lighter skin compared with darker skin (79% vs 75%; P=.059).

Controlling for provider type and specialty, significantly improved diagnostic accuracy was seen in immediate posttest scores compared with pretest scores for conditions shown in both lighter and darker skin types (lighter: 15.2 vs 13.6; darker: 13.3 vs 11.3; both P<.001)(Figure 2). The immediate posttest demonstrated higher mean diagnostic accuracy and confidence scores for skin conditions shown in lighter skin compared with darker skin (diagnostic accuracy: 15.2 vs 13.3; confidence: 3.0 vs 2.6; both P<.001), but the disparity between scores was less than in the pretest.

Following the 3-month posttest, improvement in diagnostic accuracy was noted among both lighter and darker skin types compared with the pretest, but the difference remained significant only for conditions shown in darker skin (mean scores, 11.3 vs 13.3; P<.01). Similarly, confidence in diagnosing conditions in both lighter and darker skin improved following the immediate posttest (mean scores, 2.7 vs 3.0 and 1.9 vs 2.6; both P<.001), and this improvement remained significant for only darker skin following the 3-month posttest (mean scores, 1.9 vs 2.3; P<.001). Despite these improvements, diagnostic accuracy and confidence remained higher for skin conditions shown in lighter skin compared with darker skin (diagnostic accuracy: 14.7 vs 13.3; P<.01; confidence: 2.8 vs 2.3; P<.001), though the disparity between scores was again less than in the pretest.
Comment
Our study showed that there are diagnostic disparities between lighter and darker skin types among interprofessional health care providers. Education on SOC should extend to interprofessional health care providers and other medical specialties involved in treating or triaging dermatologic diseases. A focused educational module may provide long-term improvements in diagnostic accuracy and confidence for conditions presenting in SOC. Differences in diagnostic accuracy between conditions shown in lighter and darker skin types were noted for the disease categories of infectious, cancerous, and inflammatory conditions, with the exception of conditions more frequently seen in patients with SOC. Learning resources for SOC-associated conditions are more likely to have greater representation of images depicting darker skin types.7 Future educational interventions may need to focus on dermatologic conditions that are not preferentially seen in patients with SOC. In our study, the pretest scores for conditions shown in darker skin were lowest among infectious and cancerous conditions. For infections, certain morphologic clues such as erythema are important for diagnosis but may be more subtle or difficult to discern in darker skin. It also is possible that providers may be less likely to suspect skin cancer in patients with SOC given that the morphologic presentation and/or anatomic site of involvement for skin cancers in SOC differs from those in lighter skin. Future educational interventions targeting disparities in diagnostic accuracy should focus on conditions that are not specifically associated with SOC.
Limitations of our study included the small number of participants, the study population came from a single institution, and a possible selection bias for providers interested in dermatology.
Conclusion
Disparities exist among interprofessional health care providers when treating conditions in patients with lighter skin compared to darker skin. An educational module for health care providers may provide long-term improvements in diagnostic accuracy and confidence for conditions presenting in patients with SOC.
- Fenton A, Elliott E, Shahbandi A, et al. Medical students’ ability to diagnose common dermatologic conditions in skin of color. J Am Acad Dermatol. 2020;83:957-958. doi:10.1016/j.jaad.2019.12.078
- Mamo A, Szeto MD, Rietcheck H, et al. Evaluating medical student assessment of common dermatologic conditions across Fitzpatrick phototypes and skin of color. J Am Acad Dermatol. 2022;87:167-169. doi:10.1016/j.jaad.2021.06.868
- Guda VA, Paek SY. Skin of color representation in commonly utilized medical student dermatology resources. J Drugs Dermatol. 2021;20:799. doi:10.36849/JDD.5726
- Wilson BN, Sun M, Ashbaugh AG, et al. Assessment of skin of color and diversity and inclusion content of dermatologic published literature: an analysis and call to action. Int J Womens Dermatol. 2021;7:391-397. doi:10.1016/j.ijwd.2021.04.001
- Ibraheim MK, Gupta R, Dao H, et al. Evaluating skin of color education in dermatology residency programs: data from a national survey. Clin Dermatol. 2022;40:228-233. doi:10.1016/j.clindermatol.2021.11.015
- Gupta R, Ibraheim MK, Dao H Jr, et al. Assessing dermatology resident confidence in caring for patients with skin of color. Clin Dermatol. 2021;39:873-878. doi:10.1016/j.clindermatol.2021.08.019
- Chang MJ, Lipner SR. Analysis of skin color on the American Academy of Dermatology public education website. J Drugs Dermatol. 2020;19:1236-1237. doi:10.36849/JDD.2020.5545
- Fenton A, Elliott E, Shahbandi A, et al. Medical students’ ability to diagnose common dermatologic conditions in skin of color. J Am Acad Dermatol. 2020;83:957-958. doi:10.1016/j.jaad.2019.12.078
- Mamo A, Szeto MD, Rietcheck H, et al. Evaluating medical student assessment of common dermatologic conditions across Fitzpatrick phototypes and skin of color. J Am Acad Dermatol. 2022;87:167-169. doi:10.1016/j.jaad.2021.06.868
- Guda VA, Paek SY. Skin of color representation in commonly utilized medical student dermatology resources. J Drugs Dermatol. 2021;20:799. doi:10.36849/JDD.5726
- Wilson BN, Sun M, Ashbaugh AG, et al. Assessment of skin of color and diversity and inclusion content of dermatologic published literature: an analysis and call to action. Int J Womens Dermatol. 2021;7:391-397. doi:10.1016/j.ijwd.2021.04.001
- Ibraheim MK, Gupta R, Dao H, et al. Evaluating skin of color education in dermatology residency programs: data from a national survey. Clin Dermatol. 2022;40:228-233. doi:10.1016/j.clindermatol.2021.11.015
- Gupta R, Ibraheim MK, Dao H Jr, et al. Assessing dermatology resident confidence in caring for patients with skin of color. Clin Dermatol. 2021;39:873-878. doi:10.1016/j.clindermatol.2021.08.019
- Chang MJ, Lipner SR. Analysis of skin color on the American Academy of Dermatology public education website. J Drugs Dermatol. 2020;19:1236-1237. doi:10.36849/JDD.2020.5545
Practice Points
- Disparities exist among interprofessional health care providers when diagnosing conditions in patients with lighter and darker skin, specifically for infectious, cancerous, or inflammatory conditions vs conditions that are preferentially seen in patients with skin of color (SOC).
- A focused educational module for health care providers may provide long-term improvements in diagnostic accuracy and confidence for conditions presenting in patients with SOC.
Understanding Medical Standards for Entrance Into Military Service and Disqualifying Dermatologic Conditions
Purpose of Medical Standards in the US Military
Young adults in the United States traditionally have viewed military service as a viable career given its stable salary, career training, opportunities for progression, comprehensive health care coverage, tuition assistance, and other benefits; however, not all who desire to serve in the US Military are eligible to join. The Department of Defense (DoD) maintains fitness and health requirements (ie, accession standards), which are codified in DoD Instruction 6130.03, Volume 1,1 that help ensure potential recruits can safely and fully perform their military duties. These accession standards change over time with the evolving understanding of diseases, medical advances, and accrued experience conducting operations in various environments. Accession standards serve to both preserve the health of the applicant and to ensure military mission success.
Dermatologic diseases have been prevalent in conflicts throughout US military history, representing a considerable source of morbidity to service members, inability of service members to remain on active duty, and costly use of resources. Hospitalizations of US Army soldiers for skin conditions led to the loss of more than 2 million days of service in World War I.2 In World War II, skin diseases made up 25% and 75% of all temperate and tropical climate visits, respectively. Cutaneous diseases were the most frequently addressed category for US service members in Vietnam, representing more than 1.5 million visits and nearly 10% of disease-related evacuations.2 Skin disease remains vital in 21st-century conflict. At a military hospital in Afghanistan, a review of 2421 outpatient medical records from June through July 2007 identified that dermatologic conditions resulted in 20% of military patient evaluations, 7% of nontraumatic hospital admissions, and 2% of total patient evacuations, at an estimated cost of $80,000 per evacuee.3 Between 2003 and 2006, 918 service members were evacuated for dermatologic reasons from combat zones in Afghanistan and Iraq.4
Unpredictable military environments may result in flares of a previously controlled condition, new skin diseases, or infection with endemic diseases. Mild cases of common conditions such as psoriasis or atopic dermatitis can present an unacceptable risk for severe flare in the setting of deployed military operations.5 Personnel may face extremes in temperature and humidity and work long hours under stress with limited or nonexistent opportunities for hygiene or self-care. Shared equipment and close living quarters permit the spread of infectious diseases and complicate the treatment of infestations. Military equipment and supplies such as gas masks and insect repellents can contain compounds that act as irritants or sensitizing agents, leading to contact dermatitis or urticaria. When dermatologic conditions develop or flare, further challenges are associated with evaluation and management. Health care resources vary considerably by location, with potential limitations in the availability of medications; supplies; refrigeration capabilities; and laboratory, microbiology, and histology services. Furthermore, dermatology referrals and services typically are not feasible in most deployed settings,3 though teledermatology has been available in the armed forces since 2002.
Deployed environments compound the consequences of dermatologic conditions and can impact the military mission. Military units deploy with the number of personnel needed to complete a mission and cannot replace members who become ill or injured or are medically evacuated. Something seemingly trivial, such as poor sleep due to pruritic dermatitis, may impair daytime alertness with potentially grave consequences in critical tasks such as guard or flying duties. The evacuation of a service member can compromise those left behind, and losing a service member with a unique required skill set may jeopardize a unit’s chance of success. Additionally, the impact of an evacuation itself extends beyond its direct cost and effects on the service member’s unit. The military does not maintain dedicated medical evacuation aircraft, instead repurposing aircraft in the deployed setting as needed.6 Evacuations can delay flights initially scheduled to move troops, ammunition, food, or other supplies and equipment elsewhere.
Disqualifying Skin and Soft Tissue Conditions
Current accession standards, which are listed in a publicly released document (DoD Instruction 6130.03, Volume 1), are updated based on medical, societal, and technical advances.1 These standards differ from retention standards, which apply to members actively serving in the military. Although the DoD creates a minimum standard for the entire military, the US Army, Navy, and Air Force adopt these standards and adjust as required for each branch’s needs. An updated copy can be found on the DoD Directives Division website (https://www.esd.whs.mil/dd/) or Med Standards, a third-party mobile application (app) available as a free download for Apple iOS and Android devices (https://www.doc-apps.com/). The app also includes each military branch’s interpretation of the requirements.
The accession standards outline medical conditions that, if present or verified in an applicant’s medical history, preclude joining the military (eTable). These standards are organized into general systems, with a section dedicated to dermatologic (skin and soft tissue) conditions.1 When a candidate has a potentially disqualifying medical condition identified by a screening questionnaire, medical record review, or military entrance physical examination, a referral for a determination of fitness for duty may be required. Medical accession standards are not solely driven by the diagnosis but also by the extent, nature, and timing of medical management. Procedures or prescriptions requiring frequent clinical monitoring, special handling, or severe dietary restrictions may deem the applicant’s condition potentially unsuitable. The need for immunosuppressive, anticoagulant, or refrigerated medications can impact a patient’s eligibility due to future deployment requirements and suitability for prolonged service, especially if treated for any substantial length of time. Chronic dermatologic conditions that are unresponsive to treatment, are susceptible to exacerbation despite treatment, require regular follow-up care, or interfere with the wear of military gear may be inconsistent with future deployment standards. Although the dermatologist should primarily focus on the skin and soft tissue conditions section of the accession standards, some dermatologic conditions can overlap with other medical systems and be located in a different section; for example, the section on lower extremity conditions includes a disqualifying condition of “[c]urrent ingrown toenails, if infected or symptomatic.”1
Waiver Process
Medical conditions listed in the accession standards are deemed ineligible for military service; however, applicants can apply for a waiver.1 The goal is for service members to be well controlled without treatment or with treatment widely available at military clinics and hospitals. Waivers ensure that service members are “[m]edically capable of performing duties without aggravating physical defects or medical conditions,” are “[m]edically adaptable to the military environment without geographical area limitations,” and are “free of medical conditions or physical defects that may reasonably be expected to require excessive time lost from duty for necessary treatment or hospitalization, or may result in separation from the Military Service for unfitness.”1 The waiver process requires an evaluation from specialists with verification and documentation but does not guarantee approval. Although each military branch follows the same guidelines for disqualifying medical conditions, the evaluation and waiver process varies.
Considerations for Civilian Dermatologists
For several reasons, accurate and detailed medical documentation is essential for patients who pursue military service. Applicants must complete detailed health questionnaires and may need to provide copies of health records. The military electronic health record connects to large civilian health information exchanges and pulls primary documentation from records at many hospitals and clinics. Although applicants may request supportive clarification from their dermatologists, the military relies on primary medical documentation throughout the recruitment process. Accurate diagnostic codes reduce ambiguity, as accession standards are organized by diagnosis; for example, an unspecified history of psoriasis disqualifies applicants unless documentation supports nonrecurrent childhood guttate psoriasis.1 Clear documentation of symptom severity, response to treatment, or resolution of a condition may elucidate suitability for service when matching a potentially disqualifying condition to a standard is not straightforward. Correct documentation will ensure that potential service members achieve a waiver when it is appropriate. If they are found to be unfit, it may save a patient from a bad outcome or a military unit from mission failure.
Dermatologists in the United States can reference current military medical accession standards to guide patients when needed. For example, a prospective recruit may be hesitant to start isotretinoin for severe nodulocystic acne, concerned that this medication may preclude them from joining the military. The current standards state that “[a]pplicants under treatment with systemic retinoids . . . do not meet the standard until 4 weeks after completing therapy,” while active severe nodulocystic acne is a disqualifying condition.1 Therefore, the patient could proceed with isotretinoin therapy and, pending clinical response, meet accession standards as soon as 4 weeks after treatment. A clear understanding of the purpose of these standards, including protecting the applicant’s health and maximizing the chance of combat mission accomplishment, helps to reinforce responsibilities when caring for patients who wish to serve.


- US Department of Defense. DoD Instruction 6130.03, Volume 1. Medical Standards for Military Service: Appointment, Enlistment, or Induction. Updated November 16, 2022. Accessed May 22, 2023. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_vol1.PDF?ver=7fhqacc0jGX_R9_1iexudA%3D%3D
- Becker LE, James WD. Historical overview and principles of diagnosis. In: Becker LE, James WD. Military Dermatology. Office of the Surgeon General, US Department of the Army; 1994: 1-20.
- Arnold JG, Michener MD. Evaluation of dermatologic conditions by primary care providers in deployed military settings. Mil Med. 2008;173:882-888. doi:10.7205/MILMED.173.9.882
- McGraw TA, Norton SA. Military aeromedical evacuations from central and southwest Asia for ill-defined dermatologic diseases. Arch Dermatol. 2009;145:165-170.
- Gelman AB, Norton SA, Valdes-Rodriguez R, et al. A review of skin conditions in modern warfare and peacekeeping operations. Mil Med. 2015;180:32-37.
- Fang R, Dorlac GR, Allan PF, et al. Intercontinental aeromedical evacuation of patients with traumatic brain injuries during Operations Iraqi Freedom and Enduring Freedom. Neurosurg Focus. 2010;28:E11.
Purpose of Medical Standards in the US Military
Young adults in the United States traditionally have viewed military service as a viable career given its stable salary, career training, opportunities for progression, comprehensive health care coverage, tuition assistance, and other benefits; however, not all who desire to serve in the US Military are eligible to join. The Department of Defense (DoD) maintains fitness and health requirements (ie, accession standards), which are codified in DoD Instruction 6130.03, Volume 1,1 that help ensure potential recruits can safely and fully perform their military duties. These accession standards change over time with the evolving understanding of diseases, medical advances, and accrued experience conducting operations in various environments. Accession standards serve to both preserve the health of the applicant and to ensure military mission success.
Dermatologic diseases have been prevalent in conflicts throughout US military history, representing a considerable source of morbidity to service members, inability of service members to remain on active duty, and costly use of resources. Hospitalizations of US Army soldiers for skin conditions led to the loss of more than 2 million days of service in World War I.2 In World War II, skin diseases made up 25% and 75% of all temperate and tropical climate visits, respectively. Cutaneous diseases were the most frequently addressed category for US service members in Vietnam, representing more than 1.5 million visits and nearly 10% of disease-related evacuations.2 Skin disease remains vital in 21st-century conflict. At a military hospital in Afghanistan, a review of 2421 outpatient medical records from June through July 2007 identified that dermatologic conditions resulted in 20% of military patient evaluations, 7% of nontraumatic hospital admissions, and 2% of total patient evacuations, at an estimated cost of $80,000 per evacuee.3 Between 2003 and 2006, 918 service members were evacuated for dermatologic reasons from combat zones in Afghanistan and Iraq.4
Unpredictable military environments may result in flares of a previously controlled condition, new skin diseases, or infection with endemic diseases. Mild cases of common conditions such as psoriasis or atopic dermatitis can present an unacceptable risk for severe flare in the setting of deployed military operations.5 Personnel may face extremes in temperature and humidity and work long hours under stress with limited or nonexistent opportunities for hygiene or self-care. Shared equipment and close living quarters permit the spread of infectious diseases and complicate the treatment of infestations. Military equipment and supplies such as gas masks and insect repellents can contain compounds that act as irritants or sensitizing agents, leading to contact dermatitis or urticaria. When dermatologic conditions develop or flare, further challenges are associated with evaluation and management. Health care resources vary considerably by location, with potential limitations in the availability of medications; supplies; refrigeration capabilities; and laboratory, microbiology, and histology services. Furthermore, dermatology referrals and services typically are not feasible in most deployed settings,3 though teledermatology has been available in the armed forces since 2002.
Deployed environments compound the consequences of dermatologic conditions and can impact the military mission. Military units deploy with the number of personnel needed to complete a mission and cannot replace members who become ill or injured or are medically evacuated. Something seemingly trivial, such as poor sleep due to pruritic dermatitis, may impair daytime alertness with potentially grave consequences in critical tasks such as guard or flying duties. The evacuation of a service member can compromise those left behind, and losing a service member with a unique required skill set may jeopardize a unit’s chance of success. Additionally, the impact of an evacuation itself extends beyond its direct cost and effects on the service member’s unit. The military does not maintain dedicated medical evacuation aircraft, instead repurposing aircraft in the deployed setting as needed.6 Evacuations can delay flights initially scheduled to move troops, ammunition, food, or other supplies and equipment elsewhere.
Disqualifying Skin and Soft Tissue Conditions
Current accession standards, which are listed in a publicly released document (DoD Instruction 6130.03, Volume 1), are updated based on medical, societal, and technical advances.1 These standards differ from retention standards, which apply to members actively serving in the military. Although the DoD creates a minimum standard for the entire military, the US Army, Navy, and Air Force adopt these standards and adjust as required for each branch’s needs. An updated copy can be found on the DoD Directives Division website (https://www.esd.whs.mil/dd/) or Med Standards, a third-party mobile application (app) available as a free download for Apple iOS and Android devices (https://www.doc-apps.com/). The app also includes each military branch’s interpretation of the requirements.
The accession standards outline medical conditions that, if present or verified in an applicant’s medical history, preclude joining the military (eTable). These standards are organized into general systems, with a section dedicated to dermatologic (skin and soft tissue) conditions.1 When a candidate has a potentially disqualifying medical condition identified by a screening questionnaire, medical record review, or military entrance physical examination, a referral for a determination of fitness for duty may be required. Medical accession standards are not solely driven by the diagnosis but also by the extent, nature, and timing of medical management. Procedures or prescriptions requiring frequent clinical monitoring, special handling, or severe dietary restrictions may deem the applicant’s condition potentially unsuitable. The need for immunosuppressive, anticoagulant, or refrigerated medications can impact a patient’s eligibility due to future deployment requirements and suitability for prolonged service, especially if treated for any substantial length of time. Chronic dermatologic conditions that are unresponsive to treatment, are susceptible to exacerbation despite treatment, require regular follow-up care, or interfere with the wear of military gear may be inconsistent with future deployment standards. Although the dermatologist should primarily focus on the skin and soft tissue conditions section of the accession standards, some dermatologic conditions can overlap with other medical systems and be located in a different section; for example, the section on lower extremity conditions includes a disqualifying condition of “[c]urrent ingrown toenails, if infected or symptomatic.”1
Waiver Process
Medical conditions listed in the accession standards are deemed ineligible for military service; however, applicants can apply for a waiver.1 The goal is for service members to be well controlled without treatment or with treatment widely available at military clinics and hospitals. Waivers ensure that service members are “[m]edically capable of performing duties without aggravating physical defects or medical conditions,” are “[m]edically adaptable to the military environment without geographical area limitations,” and are “free of medical conditions or physical defects that may reasonably be expected to require excessive time lost from duty for necessary treatment or hospitalization, or may result in separation from the Military Service for unfitness.”1 The waiver process requires an evaluation from specialists with verification and documentation but does not guarantee approval. Although each military branch follows the same guidelines for disqualifying medical conditions, the evaluation and waiver process varies.
Considerations for Civilian Dermatologists
For several reasons, accurate and detailed medical documentation is essential for patients who pursue military service. Applicants must complete detailed health questionnaires and may need to provide copies of health records. The military electronic health record connects to large civilian health information exchanges and pulls primary documentation from records at many hospitals and clinics. Although applicants may request supportive clarification from their dermatologists, the military relies on primary medical documentation throughout the recruitment process. Accurate diagnostic codes reduce ambiguity, as accession standards are organized by diagnosis; for example, an unspecified history of psoriasis disqualifies applicants unless documentation supports nonrecurrent childhood guttate psoriasis.1 Clear documentation of symptom severity, response to treatment, or resolution of a condition may elucidate suitability for service when matching a potentially disqualifying condition to a standard is not straightforward. Correct documentation will ensure that potential service members achieve a waiver when it is appropriate. If they are found to be unfit, it may save a patient from a bad outcome or a military unit from mission failure.
Dermatologists in the United States can reference current military medical accession standards to guide patients when needed. For example, a prospective recruit may be hesitant to start isotretinoin for severe nodulocystic acne, concerned that this medication may preclude them from joining the military. The current standards state that “[a]pplicants under treatment with systemic retinoids . . . do not meet the standard until 4 weeks after completing therapy,” while active severe nodulocystic acne is a disqualifying condition.1 Therefore, the patient could proceed with isotretinoin therapy and, pending clinical response, meet accession standards as soon as 4 weeks after treatment. A clear understanding of the purpose of these standards, including protecting the applicant’s health and maximizing the chance of combat mission accomplishment, helps to reinforce responsibilities when caring for patients who wish to serve.


Purpose of Medical Standards in the US Military
Young adults in the United States traditionally have viewed military service as a viable career given its stable salary, career training, opportunities for progression, comprehensive health care coverage, tuition assistance, and other benefits; however, not all who desire to serve in the US Military are eligible to join. The Department of Defense (DoD) maintains fitness and health requirements (ie, accession standards), which are codified in DoD Instruction 6130.03, Volume 1,1 that help ensure potential recruits can safely and fully perform their military duties. These accession standards change over time with the evolving understanding of diseases, medical advances, and accrued experience conducting operations in various environments. Accession standards serve to both preserve the health of the applicant and to ensure military mission success.
Dermatologic diseases have been prevalent in conflicts throughout US military history, representing a considerable source of morbidity to service members, inability of service members to remain on active duty, and costly use of resources. Hospitalizations of US Army soldiers for skin conditions led to the loss of more than 2 million days of service in World War I.2 In World War II, skin diseases made up 25% and 75% of all temperate and tropical climate visits, respectively. Cutaneous diseases were the most frequently addressed category for US service members in Vietnam, representing more than 1.5 million visits and nearly 10% of disease-related evacuations.2 Skin disease remains vital in 21st-century conflict. At a military hospital in Afghanistan, a review of 2421 outpatient medical records from June through July 2007 identified that dermatologic conditions resulted in 20% of military patient evaluations, 7% of nontraumatic hospital admissions, and 2% of total patient evacuations, at an estimated cost of $80,000 per evacuee.3 Between 2003 and 2006, 918 service members were evacuated for dermatologic reasons from combat zones in Afghanistan and Iraq.4
Unpredictable military environments may result in flares of a previously controlled condition, new skin diseases, or infection with endemic diseases. Mild cases of common conditions such as psoriasis or atopic dermatitis can present an unacceptable risk for severe flare in the setting of deployed military operations.5 Personnel may face extremes in temperature and humidity and work long hours under stress with limited or nonexistent opportunities for hygiene or self-care. Shared equipment and close living quarters permit the spread of infectious diseases and complicate the treatment of infestations. Military equipment and supplies such as gas masks and insect repellents can contain compounds that act as irritants or sensitizing agents, leading to contact dermatitis or urticaria. When dermatologic conditions develop or flare, further challenges are associated with evaluation and management. Health care resources vary considerably by location, with potential limitations in the availability of medications; supplies; refrigeration capabilities; and laboratory, microbiology, and histology services. Furthermore, dermatology referrals and services typically are not feasible in most deployed settings,3 though teledermatology has been available in the armed forces since 2002.
Deployed environments compound the consequences of dermatologic conditions and can impact the military mission. Military units deploy with the number of personnel needed to complete a mission and cannot replace members who become ill or injured or are medically evacuated. Something seemingly trivial, such as poor sleep due to pruritic dermatitis, may impair daytime alertness with potentially grave consequences in critical tasks such as guard or flying duties. The evacuation of a service member can compromise those left behind, and losing a service member with a unique required skill set may jeopardize a unit’s chance of success. Additionally, the impact of an evacuation itself extends beyond its direct cost and effects on the service member’s unit. The military does not maintain dedicated medical evacuation aircraft, instead repurposing aircraft in the deployed setting as needed.6 Evacuations can delay flights initially scheduled to move troops, ammunition, food, or other supplies and equipment elsewhere.
Disqualifying Skin and Soft Tissue Conditions
Current accession standards, which are listed in a publicly released document (DoD Instruction 6130.03, Volume 1), are updated based on medical, societal, and technical advances.1 These standards differ from retention standards, which apply to members actively serving in the military. Although the DoD creates a minimum standard for the entire military, the US Army, Navy, and Air Force adopt these standards and adjust as required for each branch’s needs. An updated copy can be found on the DoD Directives Division website (https://www.esd.whs.mil/dd/) or Med Standards, a third-party mobile application (app) available as a free download for Apple iOS and Android devices (https://www.doc-apps.com/). The app also includes each military branch’s interpretation of the requirements.
The accession standards outline medical conditions that, if present or verified in an applicant’s medical history, preclude joining the military (eTable). These standards are organized into general systems, with a section dedicated to dermatologic (skin and soft tissue) conditions.1 When a candidate has a potentially disqualifying medical condition identified by a screening questionnaire, medical record review, or military entrance physical examination, a referral for a determination of fitness for duty may be required. Medical accession standards are not solely driven by the diagnosis but also by the extent, nature, and timing of medical management. Procedures or prescriptions requiring frequent clinical monitoring, special handling, or severe dietary restrictions may deem the applicant’s condition potentially unsuitable. The need for immunosuppressive, anticoagulant, or refrigerated medications can impact a patient’s eligibility due to future deployment requirements and suitability for prolonged service, especially if treated for any substantial length of time. Chronic dermatologic conditions that are unresponsive to treatment, are susceptible to exacerbation despite treatment, require regular follow-up care, or interfere with the wear of military gear may be inconsistent with future deployment standards. Although the dermatologist should primarily focus on the skin and soft tissue conditions section of the accession standards, some dermatologic conditions can overlap with other medical systems and be located in a different section; for example, the section on lower extremity conditions includes a disqualifying condition of “[c]urrent ingrown toenails, if infected or symptomatic.”1
Waiver Process
Medical conditions listed in the accession standards are deemed ineligible for military service; however, applicants can apply for a waiver.1 The goal is for service members to be well controlled without treatment or with treatment widely available at military clinics and hospitals. Waivers ensure that service members are “[m]edically capable of performing duties without aggravating physical defects or medical conditions,” are “[m]edically adaptable to the military environment without geographical area limitations,” and are “free of medical conditions or physical defects that may reasonably be expected to require excessive time lost from duty for necessary treatment or hospitalization, or may result in separation from the Military Service for unfitness.”1 The waiver process requires an evaluation from specialists with verification and documentation but does not guarantee approval. Although each military branch follows the same guidelines for disqualifying medical conditions, the evaluation and waiver process varies.
Considerations for Civilian Dermatologists
For several reasons, accurate and detailed medical documentation is essential for patients who pursue military service. Applicants must complete detailed health questionnaires and may need to provide copies of health records. The military electronic health record connects to large civilian health information exchanges and pulls primary documentation from records at many hospitals and clinics. Although applicants may request supportive clarification from their dermatologists, the military relies on primary medical documentation throughout the recruitment process. Accurate diagnostic codes reduce ambiguity, as accession standards are organized by diagnosis; for example, an unspecified history of psoriasis disqualifies applicants unless documentation supports nonrecurrent childhood guttate psoriasis.1 Clear documentation of symptom severity, response to treatment, or resolution of a condition may elucidate suitability for service when matching a potentially disqualifying condition to a standard is not straightforward. Correct documentation will ensure that potential service members achieve a waiver when it is appropriate. If they are found to be unfit, it may save a patient from a bad outcome or a military unit from mission failure.
Dermatologists in the United States can reference current military medical accession standards to guide patients when needed. For example, a prospective recruit may be hesitant to start isotretinoin for severe nodulocystic acne, concerned that this medication may preclude them from joining the military. The current standards state that “[a]pplicants under treatment with systemic retinoids . . . do not meet the standard until 4 weeks after completing therapy,” while active severe nodulocystic acne is a disqualifying condition.1 Therefore, the patient could proceed with isotretinoin therapy and, pending clinical response, meet accession standards as soon as 4 weeks after treatment. A clear understanding of the purpose of these standards, including protecting the applicant’s health and maximizing the chance of combat mission accomplishment, helps to reinforce responsibilities when caring for patients who wish to serve.


- US Department of Defense. DoD Instruction 6130.03, Volume 1. Medical Standards for Military Service: Appointment, Enlistment, or Induction. Updated November 16, 2022. Accessed May 22, 2023. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_vol1.PDF?ver=7fhqacc0jGX_R9_1iexudA%3D%3D
- Becker LE, James WD. Historical overview and principles of diagnosis. In: Becker LE, James WD. Military Dermatology. Office of the Surgeon General, US Department of the Army; 1994: 1-20.
- Arnold JG, Michener MD. Evaluation of dermatologic conditions by primary care providers in deployed military settings. Mil Med. 2008;173:882-888. doi:10.7205/MILMED.173.9.882
- McGraw TA, Norton SA. Military aeromedical evacuations from central and southwest Asia for ill-defined dermatologic diseases. Arch Dermatol. 2009;145:165-170.
- Gelman AB, Norton SA, Valdes-Rodriguez R, et al. A review of skin conditions in modern warfare and peacekeeping operations. Mil Med. 2015;180:32-37.
- Fang R, Dorlac GR, Allan PF, et al. Intercontinental aeromedical evacuation of patients with traumatic brain injuries during Operations Iraqi Freedom and Enduring Freedom. Neurosurg Focus. 2010;28:E11.
- US Department of Defense. DoD Instruction 6130.03, Volume 1. Medical Standards for Military Service: Appointment, Enlistment, or Induction. Updated November 16, 2022. Accessed May 22, 2023. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003_vol1.PDF?ver=7fhqacc0jGX_R9_1iexudA%3D%3D
- Becker LE, James WD. Historical overview and principles of diagnosis. In: Becker LE, James WD. Military Dermatology. Office of the Surgeon General, US Department of the Army; 1994: 1-20.
- Arnold JG, Michener MD. Evaluation of dermatologic conditions by primary care providers in deployed military settings. Mil Med. 2008;173:882-888. doi:10.7205/MILMED.173.9.882
- McGraw TA, Norton SA. Military aeromedical evacuations from central and southwest Asia for ill-defined dermatologic diseases. Arch Dermatol. 2009;145:165-170.
- Gelman AB, Norton SA, Valdes-Rodriguez R, et al. A review of skin conditions in modern warfare and peacekeeping operations. Mil Med. 2015;180:32-37.
- Fang R, Dorlac GR, Allan PF, et al. Intercontinental aeromedical evacuation of patients with traumatic brain injuries during Operations Iraqi Freedom and Enduring Freedom. Neurosurg Focus. 2010;28:E11.
Practice Points
- Dermatologic diseases have played a substantial role in conflicts throughout US military history, representing a considerable source of morbidity to service members, loss of active-duty service members trained with necessary skills, and costly use of resources.
- The strict standards are designed to protect the health of the individual and maximize mission success.
- The Department of Defense has a publicly available document (DoD Instruction 6130.03, Volume 1) that details conditions that are disqualifying for entrance into the military. Dermatologists can reference this to provide guidance to adolescents and young adults interested in joining the military.
Use of the Retroauricular Pull-Through Sandwich Flap for Repair of an Extensive Conchal Bowl Defect With Complete Cartilage Loss
Practice Gap
Repair of a conchal defect requires careful consideration to achieve an optimal outcome. Reconstruction should resurface exposed cartilage, restore the natural projection of the auricle, and direct sound into the external auditory meatus. Patients also should be able to wear glasses and a hearing aid.
The reconstructive ladder for most conchal bowl defects includes secondary intention healing, full-thickness skin grafting (FTSG), and either a revolving-door flap or a flip-flop flap. Secondary intention and FTSG are appropriate for superficial defects, in which the loss of cartilage is not substantial.1,2 Revolving-door and flip-flop flaps are single-stage retroauricular approaches used to repair relatively small defects of the conchal bowl.3 However, reconstructive options are limited for a large defect in which there is extensive loss of cartilage; 3-stage retroauricular approaches have been utilized. The anterior pedicled retroauricular flap is a 3-stage repair that can be utilized to reconstruct a through-and-through defect of the central ear:
- Stage 1: an anteriorly based retroauricular pedicle is incised, hinged over, and sutured to the medial aspect of the defect, resurfacing the posterior ear.
- Stage 2: the pedicle is severed and the flap is folded on itself to resurface the anterior ear.
- Stage 3: the folded edge is de-epithelialized and set into the lateral defect.4
The revolving-door flap also uses a 3-stage approach and is utilized for a full-thickness central auricular defect:
- Stage 1: a revolving-door flap is used to resurface the anterior ear.
- Stage 2: a cartilage graft provides structural support.
- Stage 3: division and inset with an FTSG is used to resurface the posterior ear.
The anterior pedicled retroauricular flap and revolving-door flap techniques are useful for defects when there is intact posterior auricular skin but not when there is extensive loss of cartilage. Other downsides to these 3-stage approaches are the time and multiple procedures required.5
We describe the technique of a retroauricular pull-through sandwich flap for repair of a large conchal bowl defect with extensive cartilage loss and intact posterior auricular skin.
Technique
A 62-year-old man presented for treatment of a 2.6×2.4-cm nodular and infiltrative basal cell carcinoma of the right conchal bowl. The tumor was cleared with 3 stages of Mohs micrographic surgery, resulting in a 5.5×4.2-cm defect with complete loss of cartilage throughout the concha, helical crus, and inner rim of the antihelix (Figure 1). A 2-stage repair was performed utilizing a cartilage graft and a pull-through retroauricular interpolation flap.
Stage 1—A cartilage graft was harvested from the left concha and sutured into the central defect for structural support (Figure 2). An incision was then made through the posterior auricular skin, just medial to the residual antihelical cartilage, and a retroauricular interpolation flap was pulled through this incision to resurface the lateral two-thirds of the conchal bowl defect. This created a “sandwich” of tissue, with the following layers (ordered from anterior to posterior): retroauricular interpolation flap, cartilage graft, and intact posterior auricular skin.
A preauricular banner transposition flap was used to repair the medial one-third of the conchal defect. A small area was left to heal by secondary intention (Figure 3).
Stage 2—The patient returned 3 weeks later for division and inset of the retroauricular interpolation flap. The pedicle of the flap was severed and its free edge was sutured into the lateral aspect of the defect. The posterior auricular incision that the flap had been pulled through in stage 1 of the repair was closed in a layered fashion, and the secondary defect of the postauricular scalp was left to heal by secondary intention (Figure 4).
Final Results—At follow-up 1 month later, the patient was noted to have good aesthetic and functional outcomes (Figure 5).
Practice Implications
The retroauricular pull-through sandwich flap combines a cartilage graft and a retroauricular interpolation flap pulled through an incision in the posterior auricular skin to resurface the anterior ear. This repair is most useful for a large conchal bowl defect in which there is extensive missing cartilage but intact posterior auricular skin.
The retroauricular scalp is a substantial tissue reservoir with robust vasculature; an interpolation flap from this area frequently is used to repair an extensive ear defect. The most common use of an interpolation flap is for a large helical defect; however, the flap also can be pulled through an incision in the posterior auricular skin to the front of the ear in a manner similar to revolving-door and flip-flop flaps, thus allowing for increased flap reach.
A cartilage graft provides structural support, helping to maintain auricular projection. The helical arcades provide a robust vascular supply and maintain viability of the helical rim tissue, despite the large aperture created for the pull-through flap.
We recommend this 2-stage repair for large conchal bowl defects with extensive cartilage loss and intact posterior auricular skin.
- Clark DP, Hanke CW. Neoplasms of the conchal bowl: treatment with Mohs micrographic surgery. J Dermatol Surg Oncol. 1988;14:1223-1228. doi:10.1111/j.1524-4725.1988.tb03479.x
- Dessy LA, Figus A, Fioramonti P, et al. Reconstruction of anterior auricular conchal defect after malignancy excision: revolving-door flap versus full-thickness skin graft. J Plast Reconstr Aesthet Surg. 2010;63:746-752. doi:10.1016/j.bjps.2009.01.073
- Golash A, Bera S, Kanoi AV, et al. The revolving door flap: revisiting an elegant but forgotten flap for ear defect reconstruction. Indian J Plast Surg. 2020;53:64-70. doi:10.1055/s-0040-1709531
- Heinz MB, Hölzle F, Ghassemi A. Repairing a non-marginal full-thickness auricular defect using a reversed flap from the postauricular area. J Oral Maxillofac Surg. 2015;73:764-768. doi:10.1016/j.joms.2014.11.005
- Leitenberger JJ, Golden SK. Reconstruction after full-thickness loss of the antihelix, scapha, and triangular fossa. Dermatol Surg. 2016;42:893-896. doi:10.1097/DSS.0000000000000664
Practice Gap
Repair of a conchal defect requires careful consideration to achieve an optimal outcome. Reconstruction should resurface exposed cartilage, restore the natural projection of the auricle, and direct sound into the external auditory meatus. Patients also should be able to wear glasses and a hearing aid.
The reconstructive ladder for most conchal bowl defects includes secondary intention healing, full-thickness skin grafting (FTSG), and either a revolving-door flap or a flip-flop flap. Secondary intention and FTSG are appropriate for superficial defects, in which the loss of cartilage is not substantial.1,2 Revolving-door and flip-flop flaps are single-stage retroauricular approaches used to repair relatively small defects of the conchal bowl.3 However, reconstructive options are limited for a large defect in which there is extensive loss of cartilage; 3-stage retroauricular approaches have been utilized. The anterior pedicled retroauricular flap is a 3-stage repair that can be utilized to reconstruct a through-and-through defect of the central ear:
- Stage 1: an anteriorly based retroauricular pedicle is incised, hinged over, and sutured to the medial aspect of the defect, resurfacing the posterior ear.
- Stage 2: the pedicle is severed and the flap is folded on itself to resurface the anterior ear.
- Stage 3: the folded edge is de-epithelialized and set into the lateral defect.4
The revolving-door flap also uses a 3-stage approach and is utilized for a full-thickness central auricular defect:
- Stage 1: a revolving-door flap is used to resurface the anterior ear.
- Stage 2: a cartilage graft provides structural support.
- Stage 3: division and inset with an FTSG is used to resurface the posterior ear.
The anterior pedicled retroauricular flap and revolving-door flap techniques are useful for defects when there is intact posterior auricular skin but not when there is extensive loss of cartilage. Other downsides to these 3-stage approaches are the time and multiple procedures required.5
We describe the technique of a retroauricular pull-through sandwich flap for repair of a large conchal bowl defect with extensive cartilage loss and intact posterior auricular skin.
Technique
A 62-year-old man presented for treatment of a 2.6×2.4-cm nodular and infiltrative basal cell carcinoma of the right conchal bowl. The tumor was cleared with 3 stages of Mohs micrographic surgery, resulting in a 5.5×4.2-cm defect with complete loss of cartilage throughout the concha, helical crus, and inner rim of the antihelix (Figure 1). A 2-stage repair was performed utilizing a cartilage graft and a pull-through retroauricular interpolation flap.

Stage 1—A cartilage graft was harvested from the left concha and sutured into the central defect for structural support (Figure 2). An incision was then made through the posterior auricular skin, just medial to the residual antihelical cartilage, and a retroauricular interpolation flap was pulled through this incision to resurface the lateral two-thirds of the conchal bowl defect. This created a “sandwich” of tissue, with the following layers (ordered from anterior to posterior): retroauricular interpolation flap, cartilage graft, and intact posterior auricular skin.

A preauricular banner transposition flap was used to repair the medial one-third of the conchal defect. A small area was left to heal by secondary intention (Figure 3).

Stage 2—The patient returned 3 weeks later for division and inset of the retroauricular interpolation flap. The pedicle of the flap was severed and its free edge was sutured into the lateral aspect of the defect. The posterior auricular incision that the flap had been pulled through in stage 1 of the repair was closed in a layered fashion, and the secondary defect of the postauricular scalp was left to heal by secondary intention (Figure 4).

Final Results—At follow-up 1 month later, the patient was noted to have good aesthetic and functional outcomes (Figure 5).

Practice Implications
The retroauricular pull-through sandwich flap combines a cartilage graft and a retroauricular interpolation flap pulled through an incision in the posterior auricular skin to resurface the anterior ear. This repair is most useful for a large conchal bowl defect in which there is extensive missing cartilage but intact posterior auricular skin.
The retroauricular scalp is a substantial tissue reservoir with robust vasculature; an interpolation flap from this area frequently is used to repair an extensive ear defect. The most common use of an interpolation flap is for a large helical defect; however, the flap also can be pulled through an incision in the posterior auricular skin to the front of the ear in a manner similar to revolving-door and flip-flop flaps, thus allowing for increased flap reach.
A cartilage graft provides structural support, helping to maintain auricular projection. The helical arcades provide a robust vascular supply and maintain viability of the helical rim tissue, despite the large aperture created for the pull-through flap.
We recommend this 2-stage repair for large conchal bowl defects with extensive cartilage loss and intact posterior auricular skin.
Practice Gap
Repair of a conchal defect requires careful consideration to achieve an optimal outcome. Reconstruction should resurface exposed cartilage, restore the natural projection of the auricle, and direct sound into the external auditory meatus. Patients also should be able to wear glasses and a hearing aid.
The reconstructive ladder for most conchal bowl defects includes secondary intention healing, full-thickness skin grafting (FTSG), and either a revolving-door flap or a flip-flop flap. Secondary intention and FTSG are appropriate for superficial defects, in which the loss of cartilage is not substantial.1,2 Revolving-door and flip-flop flaps are single-stage retroauricular approaches used to repair relatively small defects of the conchal bowl.3 However, reconstructive options are limited for a large defect in which there is extensive loss of cartilage; 3-stage retroauricular approaches have been utilized. The anterior pedicled retroauricular flap is a 3-stage repair that can be utilized to reconstruct a through-and-through defect of the central ear:
- Stage 1: an anteriorly based retroauricular pedicle is incised, hinged over, and sutured to the medial aspect of the defect, resurfacing the posterior ear.
- Stage 2: the pedicle is severed and the flap is folded on itself to resurface the anterior ear.
- Stage 3: the folded edge is de-epithelialized and set into the lateral defect.4
The revolving-door flap also uses a 3-stage approach and is utilized for a full-thickness central auricular defect:
- Stage 1: a revolving-door flap is used to resurface the anterior ear.
- Stage 2: a cartilage graft provides structural support.
- Stage 3: division and inset with an FTSG is used to resurface the posterior ear.
The anterior pedicled retroauricular flap and revolving-door flap techniques are useful for defects when there is intact posterior auricular skin but not when there is extensive loss of cartilage. Other downsides to these 3-stage approaches are the time and multiple procedures required.5
We describe the technique of a retroauricular pull-through sandwich flap for repair of a large conchal bowl defect with extensive cartilage loss and intact posterior auricular skin.
Technique
A 62-year-old man presented for treatment of a 2.6×2.4-cm nodular and infiltrative basal cell carcinoma of the right conchal bowl. The tumor was cleared with 3 stages of Mohs micrographic surgery, resulting in a 5.5×4.2-cm defect with complete loss of cartilage throughout the concha, helical crus, and inner rim of the antihelix (Figure 1). A 2-stage repair was performed utilizing a cartilage graft and a pull-through retroauricular interpolation flap.

Stage 1—A cartilage graft was harvested from the left concha and sutured into the central defect for structural support (Figure 2). An incision was then made through the posterior auricular skin, just medial to the residual antihelical cartilage, and a retroauricular interpolation flap was pulled through this incision to resurface the lateral two-thirds of the conchal bowl defect. This created a “sandwich” of tissue, with the following layers (ordered from anterior to posterior): retroauricular interpolation flap, cartilage graft, and intact posterior auricular skin.

A preauricular banner transposition flap was used to repair the medial one-third of the conchal defect. A small area was left to heal by secondary intention (Figure 3).

Stage 2—The patient returned 3 weeks later for division and inset of the retroauricular interpolation flap. The pedicle of the flap was severed and its free edge was sutured into the lateral aspect of the defect. The posterior auricular incision that the flap had been pulled through in stage 1 of the repair was closed in a layered fashion, and the secondary defect of the postauricular scalp was left to heal by secondary intention (Figure 4).

Final Results—At follow-up 1 month later, the patient was noted to have good aesthetic and functional outcomes (Figure 5).

Practice Implications
The retroauricular pull-through sandwich flap combines a cartilage graft and a retroauricular interpolation flap pulled through an incision in the posterior auricular skin to resurface the anterior ear. This repair is most useful for a large conchal bowl defect in which there is extensive missing cartilage but intact posterior auricular skin.
The retroauricular scalp is a substantial tissue reservoir with robust vasculature; an interpolation flap from this area frequently is used to repair an extensive ear defect. The most common use of an interpolation flap is for a large helical defect; however, the flap also can be pulled through an incision in the posterior auricular skin to the front of the ear in a manner similar to revolving-door and flip-flop flaps, thus allowing for increased flap reach.
A cartilage graft provides structural support, helping to maintain auricular projection. The helical arcades provide a robust vascular supply and maintain viability of the helical rim tissue, despite the large aperture created for the pull-through flap.
We recommend this 2-stage repair for large conchal bowl defects with extensive cartilage loss and intact posterior auricular skin.
- Clark DP, Hanke CW. Neoplasms of the conchal bowl: treatment with Mohs micrographic surgery. J Dermatol Surg Oncol. 1988;14:1223-1228. doi:10.1111/j.1524-4725.1988.tb03479.x
- Dessy LA, Figus A, Fioramonti P, et al. Reconstruction of anterior auricular conchal defect after malignancy excision: revolving-door flap versus full-thickness skin graft. J Plast Reconstr Aesthet Surg. 2010;63:746-752. doi:10.1016/j.bjps.2009.01.073
- Golash A, Bera S, Kanoi AV, et al. The revolving door flap: revisiting an elegant but forgotten flap for ear defect reconstruction. Indian J Plast Surg. 2020;53:64-70. doi:10.1055/s-0040-1709531
- Heinz MB, Hölzle F, Ghassemi A. Repairing a non-marginal full-thickness auricular defect using a reversed flap from the postauricular area. J Oral Maxillofac Surg. 2015;73:764-768. doi:10.1016/j.joms.2014.11.005
- Leitenberger JJ, Golden SK. Reconstruction after full-thickness loss of the antihelix, scapha, and triangular fossa. Dermatol Surg. 2016;42:893-896. doi:10.1097/DSS.0000000000000664
- Clark DP, Hanke CW. Neoplasms of the conchal bowl: treatment with Mohs micrographic surgery. J Dermatol Surg Oncol. 1988;14:1223-1228. doi:10.1111/j.1524-4725.1988.tb03479.x
- Dessy LA, Figus A, Fioramonti P, et al. Reconstruction of anterior auricular conchal defect after malignancy excision: revolving-door flap versus full-thickness skin graft. J Plast Reconstr Aesthet Surg. 2010;63:746-752. doi:10.1016/j.bjps.2009.01.073
- Golash A, Bera S, Kanoi AV, et al. The revolving door flap: revisiting an elegant but forgotten flap for ear defect reconstruction. Indian J Plast Surg. 2020;53:64-70. doi:10.1055/s-0040-1709531
- Heinz MB, Hölzle F, Ghassemi A. Repairing a non-marginal full-thickness auricular defect using a reversed flap from the postauricular area. J Oral Maxillofac Surg. 2015;73:764-768. doi:10.1016/j.joms.2014.11.005
- Leitenberger JJ, Golden SK. Reconstruction after full-thickness loss of the antihelix, scapha, and triangular fossa. Dermatol Surg. 2016;42:893-896. doi:10.1097/DSS.0000000000000664
CLL combo treatment: Phase-3 study inconclusive
The difference in PFS between the IVO arm, 85%, versus 87% in the IO arm was statistically insignificant.
“Due to the early read-out and the futility boundaries being crossed, long-term follow-up will be critical to understand if there are any long-term benefits to IVO,” said study principal investigator Jennifer A. Woyach MD, professor in the division of hematology at The Ohio State University Comprehensive Care Center (OSUCCC – The James) in Columbus.
The 14-month follow-up data includes results from 465 CLL patients aged 65+ (median age 74 years, 67.5% male) who were treatment naive. The IO and IVO arms had 232 and 233 participants respectively, patients across both arms had Eastern Cooperative Oncology Group scores of 0-1 (97%), occurrence of Del (17p) was 13%, and a Rai stage status of III/IV was 55%, slightly more patients in the IO arm had unmutated IGHV 55% vs. 47% in the IVO arm. Researchers noted that, as expected, patients in the IVO group had a greater occurrence of hematologic adverse events graded at 3 or above, 61% VS 48% in the IO arm, P =.006.
The trial was spurred by the fact that many CLL patients on IO therapy must remain on treatment indefinitely, and an earlier phase II trial suggested that IVO therapy could lead to deep remission and therapy discontinuation.
Looking at the complete response (CR) rates and undetectable minimal residual disease (uMRD) rates across both arms suggested that there may be some hope that IVO could help CLL patients achieve deep remissions and discontinue therapy. Patients in the IVO arm had a CR of 68.5% and uMRD of 86.8% while only 31.3% of those in the IO arm had a CR and 33.3% achieved uMRD status.
“Despite the impressive CR and uMRD results, this study demonstrates that IVO is not superior to IO in terms of progression-free survival. However, because many patients in the IVO arm have discontinued treatment while those in the IO arm remain on ibrutinib, we think that it will be very important to continue to follow these patients long term, to see if there are advantages to this time limited therapy, especially in terms of toxicity, that we cannot appreciate with this follow-up,” said Dr. Woyach.
The Alliance for Clinical Trials in Oncology cooperative group, including OSUCCC James, is currently working to design the next frontline CLL study for older patients that builds on this work.
Dr. Woyach disclosed ties with Abbvie, AstraZeneca, Beigene, Genentech, Janssen, Loxo/Lilly, Merck, Newave, Pharmacyclics, and Schrodinger.
The difference in PFS between the IVO arm, 85%, versus 87% in the IO arm was statistically insignificant.
“Due to the early read-out and the futility boundaries being crossed, long-term follow-up will be critical to understand if there are any long-term benefits to IVO,” said study principal investigator Jennifer A. Woyach MD, professor in the division of hematology at The Ohio State University Comprehensive Care Center (OSUCCC – The James) in Columbus.
The 14-month follow-up data includes results from 465 CLL patients aged 65+ (median age 74 years, 67.5% male) who were treatment naive. The IO and IVO arms had 232 and 233 participants respectively, patients across both arms had Eastern Cooperative Oncology Group scores of 0-1 (97%), occurrence of Del (17p) was 13%, and a Rai stage status of III/IV was 55%, slightly more patients in the IO arm had unmutated IGHV 55% vs. 47% in the IVO arm. Researchers noted that, as expected, patients in the IVO group had a greater occurrence of hematologic adverse events graded at 3 or above, 61% VS 48% in the IO arm, P =.006.
The trial was spurred by the fact that many CLL patients on IO therapy must remain on treatment indefinitely, and an earlier phase II trial suggested that IVO therapy could lead to deep remission and therapy discontinuation.
Looking at the complete response (CR) rates and undetectable minimal residual disease (uMRD) rates across both arms suggested that there may be some hope that IVO could help CLL patients achieve deep remissions and discontinue therapy. Patients in the IVO arm had a CR of 68.5% and uMRD of 86.8% while only 31.3% of those in the IO arm had a CR and 33.3% achieved uMRD status.
“Despite the impressive CR and uMRD results, this study demonstrates that IVO is not superior to IO in terms of progression-free survival. However, because many patients in the IVO arm have discontinued treatment while those in the IO arm remain on ibrutinib, we think that it will be very important to continue to follow these patients long term, to see if there are advantages to this time limited therapy, especially in terms of toxicity, that we cannot appreciate with this follow-up,” said Dr. Woyach.
The Alliance for Clinical Trials in Oncology cooperative group, including OSUCCC James, is currently working to design the next frontline CLL study for older patients that builds on this work.
Dr. Woyach disclosed ties with Abbvie, AstraZeneca, Beigene, Genentech, Janssen, Loxo/Lilly, Merck, Newave, Pharmacyclics, and Schrodinger.
The difference in PFS between the IVO arm, 85%, versus 87% in the IO arm was statistically insignificant.
“Due to the early read-out and the futility boundaries being crossed, long-term follow-up will be critical to understand if there are any long-term benefits to IVO,” said study principal investigator Jennifer A. Woyach MD, professor in the division of hematology at The Ohio State University Comprehensive Care Center (OSUCCC – The James) in Columbus.
The 14-month follow-up data includes results from 465 CLL patients aged 65+ (median age 74 years, 67.5% male) who were treatment naive. The IO and IVO arms had 232 and 233 participants respectively, patients across both arms had Eastern Cooperative Oncology Group scores of 0-1 (97%), occurrence of Del (17p) was 13%, and a Rai stage status of III/IV was 55%, slightly more patients in the IO arm had unmutated IGHV 55% vs. 47% in the IVO arm. Researchers noted that, as expected, patients in the IVO group had a greater occurrence of hematologic adverse events graded at 3 or above, 61% VS 48% in the IO arm, P =.006.
The trial was spurred by the fact that many CLL patients on IO therapy must remain on treatment indefinitely, and an earlier phase II trial suggested that IVO therapy could lead to deep remission and therapy discontinuation.
Looking at the complete response (CR) rates and undetectable minimal residual disease (uMRD) rates across both arms suggested that there may be some hope that IVO could help CLL patients achieve deep remissions and discontinue therapy. Patients in the IVO arm had a CR of 68.5% and uMRD of 86.8% while only 31.3% of those in the IO arm had a CR and 33.3% achieved uMRD status.
“Despite the impressive CR and uMRD results, this study demonstrates that IVO is not superior to IO in terms of progression-free survival. However, because many patients in the IVO arm have discontinued treatment while those in the IO arm remain on ibrutinib, we think that it will be very important to continue to follow these patients long term, to see if there are advantages to this time limited therapy, especially in terms of toxicity, that we cannot appreciate with this follow-up,” said Dr. Woyach.
The Alliance for Clinical Trials in Oncology cooperative group, including OSUCCC James, is currently working to design the next frontline CLL study for older patients that builds on this work.
Dr. Woyach disclosed ties with Abbvie, AstraZeneca, Beigene, Genentech, Janssen, Loxo/Lilly, Merck, Newave, Pharmacyclics, and Schrodinger.