User login
Predictors of Suboptimal Glycemic Control for Hospitalized Patients with Diabetes: Targets for Clinical Action
From Sharp HealthCare, San Diego, CA (Ms. Thompson, Mr. Koucheki, Dr. Holdy), National University, San Diego, CA (Dr. Smith), and University of California, Irvine (Dr. Bender).
Abstract
- Objective: Suboptimal glycemic control (SGC) puts hospitalized patients with diabetes at risk for poor outcomes. The purpose of this study was to quantify factors with predictive capacity to identify patients at risk for SGC during hospitalization.
- Methods: 32 baseline and demographic variables were extracted from the electronic records of 23,100 patients with diabetes hospitalized between 2009 and 2012. The rate of blood glucose values between 70 and 180 mg/dL was calculated for each patient. A predictive model for SGC was developed using regression modeling, standardized coefficients, and classification tree analysis. Odds ratios (ORs) were calculated to isolate adjusted odds of SGC for top predictors.
- Results: The final predictive model included 13 variables (C statistic = 0.88). HbA1c (OR, 0.60 [95% confidence interval {CI}, 0.58–0.61]), admission blood glucose (OR, 0.91 [CI, 0.91–0.92]), and steroid use (OR, 0.06 [CI 0.04–0.08]) were the highest-ranking predictors of SGC. HbA1c and SGC had a strong linear relationship (R2 = 0.99), with increasing odds for SGC as HbA1c increased. Admission blood glucose and SGC had a polynomial relationship (R2 = 0.95); increasing odds for SGC until 240 mg/dL; then odds started decreasing. Steroid use showed a steady threefold increase in odds for SGC across all rates of use.
- Conclusions: Poor preadmission diabetes control and inpatient steroid use strongly predict SGC. A range of thresholds for these predictors was empirically determined, providing a basis for targeted therapies on admission. Guidelines incorporating empirically derived thresholds should enhance the ability to achieve optimal glycemic control for hospitalized patients with diabetes.
Current recommendations for glycemic control of hospitalized patients include use of multidisciplinary diabetic care teams and standardized insulin order sets [1,2], yet there is still uncertainty how best to target such protocols for patients with diabetes at risk for suboptimal glycemic control [2,3]. Many factors are theorized to hinder optimal inpatient glycemic control, such as steroid use [4,5],comorbid states [6],severity of illness [7,8], and preadmission glycemic control [9,10]. However, there currently exists little evidence of these factors’ ability to predict suboptimal glycemic control (SGC). Identifying straightforward predictive factors of SGC would provide a clinically meaningful basis for targeted therapy. The purpose of this study was to describe the prevalence of a range of potential risk factors in a diverse hospitalized patient population with a secondary diagnosis of diabetes (types 1 and 2), and to determine which factors were predictive of SGC.
Methods
A retrospective cohort study design was used to identify factors predictive of inpatient SGC for patients admitted to any of 3 hospitals aligned with Sharp HealthCare (“Sharp”), a community-based, nonprofit integrated health system headquartered in San Diego, California, that serves more than 27% of the county’s 3 million-plus residents each year.
Inclusion Criteria
We extracted data for 23,100 patients hospitalized between January 2009 and December 2012 with a secondary diagnosis of diabetes (types 1 and 2), a length of stay (LOS) ≥ 3 days, and a minimum of 2 point-of-care (POC) blood glucose tests per day. The LOS and blood glucose minimum are standard criteria for Sharp glycemic monitoring to ensure a minimum quantity of blood glucose monitoring for glycemic management.
Glycemic Control (Independent Variable)
Glycemic control was defined as POC blood glucose values within the target range of 70 to 180 mg/dL during hospitalization. POC tests outside that range were defined as SGC. This range was determined based on current Sharp benchmark targets and was not adjusted for total number of blood glucose values.
Predictive Variables
Age, gender, race, ethnicity, payor, facility and LOS were extracted from Sharp’s data warehouse. Medical vs. surgical stay and major diagnostic category was determined from administrative diagnosis coding. Body mass index (BMI) was extracted from Sharp’s electronic health record. Risk of mortality and severity of illness were calculated using 3M APR-DRG proprietary software using administrative diagnosis coding. Comorbidities were determined based on administrative diagnosis codes per published guidelines [11]. Glycosylated hemoglobin (HbA1c) was obtained on admission for patients with a secondary diagnosis of diabetes as part of Sharp's multidisciplinary diabetes care management program and extracted from the electronic health record. Admission blood glucose was defined as the first documented POC blood glucose after admission. ICU stay was calculated as a continuous variable: the percent of LOS spent in the ICU. Steroid use was similarly calculated, and defined as oral or intravenous administration of any quantity or dosage of the following corticosteroids during each day of hospitalization: dexamethasone, hydrocortisone, prednisone, and/or methylprednisone. Adherence to Sharp's multidisciplinary diabetes care management program was measured by use of standardized insulin order sets. Sharp uses evidence-based order sets for continuous infusion and subcutaneous insulin management; subcutaneous orders include basal and rapid-acting insulin. We calculated the total time a person was on an order set during hospitalization by subtracting any time a patient did not have insulin ordered from the total LOS. This was transformed into a variable documenting the percent of LOS the patient was on an insulin order set. Average blood glucose for admission was calculated for all documented POC blood glucoses during admission, omitting the admission blood glucose (the first POC blood glucose of the admission).
Analysis
Univariate analyses including t tests and chi-square tests were conducted to investigate the unadjusted association between variables and glycemic control. Good glycemic control was defined as 90% of all POC blood glucose tests between 70 and 180 mg/dL based on empirical distribution and organization targets. A predictive model of inpatient glycemic control was then developed using a backward stepwise multivariable logistic regression approach. The data were split into a model building and validation set. Variables were included that represented both baseline and transitional state during the hospital stay to account for potentially mediating effects and a sensitivity analysis was conducted with them in and out of the final model to assess impact. Standardized coefficients were calculated to rank order variables in the model allowing indication of the variables with the greatest predictive impact on the outcome. Further investigation of the optimal classification points for the variables was conducted to indicate best differentiation of good glycemic control. Significant variables from the multivariable logistic regression were included in an exploratory classification tree analysis that recursively partitioned data in order to improve the fit, with optimal splitting identified over all variables at all possible split points. Classification tree cut-off points were used to further develop models identifying odds ratios for various thresholds for the top three predictive variables. A series of logistic regression models were then run with differing cut points of top 3 predictors to isolate adjusted odds of good glycemic control. Analytic data set building and statistical analyses were completed using SAS 9.4.
Results
Patient Characteristics
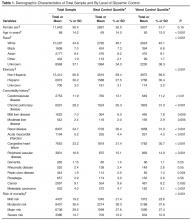
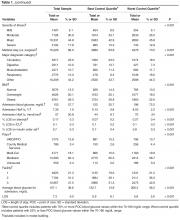
Table 1 shows patient demographic and clinical characteristics for the entire sample and for the top quartile (76% or greater POC blood glucose values within target range) and bottom quartile (25% or less POC blood glucose values within target range). Unadjusted results show a significant difference across quartiles for all factors except age, gender, dementia, rheumatic disease and paraplegia. Patients in the bottom 25th percentile (ie, the poorest control) were more likely than the total population to have a higher admission blood glucose (198 mg/dL vs. 153 mg/dL), higher HbA1c (8.53 [70mmol/mol] vs. 7.35 [57mmol/mol), a medical (74% vs. 66%) and/or respiratory (18% vs. 12%) diagnosis, corticosteroid use (17% vs. 27%), an insulin order set use (80% vs. 70%) and higher mean blood glucose during hospitalization (206.3 vs. 157.1 mg/dL). Patients with poorest control were less likely that the total population to have a high risk of mortality (11% vs. 15%) and severity of illness (13% vs. 18%). They also had less ICU care (8% vs. 13%), and a shorter LOS (5.82 vs. 7.82 days).
Predictive Modeling
Classification tree analysis resulted in the same top 3 predictors, but in a different order. The analysis also provided cut-off values that predict suboptimal glycemic control. Classification tree analysis showed admission blood glucose was the most influential predictor, with 164.5 mg/dL indicating the optimal cut-point for prediction of SGC, followed by HbA1c with an optimal cut-off point of 6.65% indicating prediction of SGC, followed by treatment with corticosteroids, with an optimal cut-off point of 24% of the LOS on corticosteroids indicating prediction of SGC.
Discussion
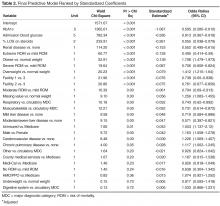
Evidence of patient characteristics that consistently predict suboptimal glycemic control during hospitalization is needed to better inform clinical decisions for inpatient glycemic management. Hospitalized patients would greatly benefit from glycemic protocols that incorporate risk stratification tools based evidence-based risk factors for poor glycemic control [12]. The science of inpatient glycemic management is in its infancy, however, and currently there is limited evidence to help identify at-risk populations and guide effective management for at-risk patient populations [13]. This study provides important data that can be used to develop risk stratification tools with implications for improved glycemic management of hospitalized patients with diabetes. Among the 32 factors included in the final multivariate logistic model (Table 2), 10 were statistically significant predictors of SGC. The top 3 predictors of SGC were HbA1c, admission blood glucose, and steroid use. These are straightforward, easily accessed factors that can become the basis for effective risk stratification and targeted clinical therapies.
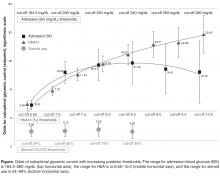
This study showed that the degree of diabetes control prior to admission, as measured by HbA1c, is one of the strongest predictors of inpatient SGC. Patients with poorly controlled diabetes pre-admission had significantly higher rates of SGC than patients admitted with good diabetes control. Furthermore, there was a strong linear relationship between degree of pre-admission diabetes control and glycemic control during hospitalization: the higher the HbA1c on admission, the higher the odds are for poor glycemic control during hospitalization (Figure). The odds of SGC increased more than fivefold at an HbA1c of just 6.7% (50 mmol/mol) and continued to increase linearly as HbA1c increased. This increase occurred despite the fact that patients with poorly controlled pre-admission diabetes were found to have a significantly greater rate of insulin treatment using standardized order sets (which included basal and rapid-acting insulin) than the total sample in this study. So although patients with poorly controlled diabetes pre-admission were actively managed using evidence-based insulin order sets to control glycemia throughout their hospitalization, this did not translate to better glycemic control. Boord et al [14] and Neubauer et al [15] found similar results in their evaluation of glycemic control in hospitalized patients: while insulin use was high, glycemic control remained suboptimal. Similarly, Schnipper et al [16] found that adherence to insulin orders per se were not associated with better glucose control. They noted that the majority of patients with continued elevated blood glucose values did not have changes made to their insulin orders in response to suboptimal blood glucose values. These observations and this study’s findings suggest that fixed standardized subcutaneous orders may be more effective for well-controlled patients with diabetes than for patients with poorly controlled diabetes on admission. These patients need more frequent modifications to standard order sets based on clinical response during hospitalization to ensure good glycemic control.
This study confirmed the well known, highly correlated relationship between POC blood glucose and HbA1c. Rohlfing et al [17] previously documented a strong relationship (R = 0.82) between blood glucose and HbA1c in patients enrolled in the Diabetes Control and Complications Trial. Nathan et al [18]also documented a strong relationship (R= 0.84) between HbA1c and average blood glucose that was consistent across diverse populations. This study’s findings show odds for SGC based on admission blood glucose followed a very similar trend as HbA1c, and both factors showed much greater odds for SGC than steroid use, independent of other factors.
Admission blood glucose was the second best predictor of SBC using regression modeling (Table 2) and the strongest predictor using classification tree analysis. Odds for SGC were both very high and remarkably similar for a concomitant range of admission blood glucose and HbA1c values (Figure). The overlapping admission blood glucose range was 165 to 240 mg/dL, which corresponded to HbA1c values between 6.65 and 8.5 (49 mmol/mol–69 mmol/mol). The odds for SGC increased from a 6.5-fold increase in odds at 165 mg/dL to 22-fold increased odds at 240 mg/dL. This corresponded to a 5.5-fold increase in odds with HbA1c of 6.65 (49 mmol/mol) to a 19-fold increase in odds with HbA1c of 8.5 (69 mmol/mol).
Notably, an admission blood glucose as low as 164 mg/dL significantly increased the odds of SGC. This relatively mild hyperglycemia is not typically considered a signifier for difficult inpatient glycemic management. Furthermore, the odds of SGC continue to increase until admission blood glucose reached approximately 240 mg/dL, at which point the odds start declining (Figure). This suggests that only exceptionally elevated admission blood glucoses triggered prompt insulin treatment on admission. The data from this study suggests targeted action for an admission blood glucose as low as 164 mg/dL is just as necessary as for those admitted with a much higher blood glucose to ensure optimal glycemic control throughout hospitalization. Implications are that admission blood glucose may be an inexpensive, straightforward, and readily available predictor of SGC and marker for targeted clinical action, especially for hospitals that do not routinely order HbA1c labs during hospitalization.
Steroid treatment was the third strongest predictor of SGC. Additional analysis showed that any proportion of hospital stay with steroid administration resulted in a stable threefold increased odds for SGC, adjusting for other predictive factors (Figure). Developing insulin treatment therapies that are tailored to patients that will be administered steroids at any point during their hospitalization may be a reasonable strategy to reduce SGC in this population. Based on the results of this study and other Sharp data, Sharp is currently piloting a steroid insulin order set that is available in the electronic health record to hospital physicians for use with any patient that is administered steroids. The order set includes eating and non-eating standards, intensified meal dose coverage, a lower blood glucose threshold for starting correction dosing, and a diabetic nurse educator consult. Evaluation will include appropriate order set usage, rate of glycemic control and extreme blood glucose values.
Limitations
A limitation of this study is that it used observational data and was conducted within a single health care system, thus potentially reducing generalizability. Nevertheless, the sample size was large, there were many clinical and demographic characteristics to be leveraged in the analysis, the statistical approach utilized a complementary regression and classification approach to adjust and present the findings, and the sample included patients from three hospitals across San Diego County with diverse patient populations.
Conclusion
While much progress has been made understanding the need for appropriate glycemic management for patient with diabetes to reduce their risk for adverse outcomes, the knowledge base is still quite limited, especially regarding optimal glycemic limits for diverse patient populations. There is a need to identify predictors of SGC if risk stratification tools are to be built that can help target therapies with the potential to reducing the risk of poor glycemic control and adverse patient outcomes. This study identified 3 readily available factors—admission blood glucose, HbA1c and steroid use—that strongly predict SGC, controlling for other patient risk factors. In general, poor pre-admission diabetes control and inpatient steroid use strongly predict SGC, and the data suggests that earlier and frequently calibrated intervention may improve inpatient glycemic control for these patient populations. We identified a range of thresholds for these variables that may provide a basis for targeted treatment on admission. In conclusion, this study has important implications for meaningful use of readily available factors to identify patients at risk for SGC. Clinical therapies and guidelines incorporating empirically derived risk-stratification tools should enhance the ability to achieve the triple aim of better health, better care quality and more efficient care costs for hospitalized patient with diabetes.
Corresponding author: Miriam Bender, PhD, RN, University of California, Irvine, 252 Berk Hall, Irvine, CA 92697, [email protected].
Financial disclosures: None.
Author contributions: Author contributions: conception and design, MB, TCS, JT, KH; analysis and interpretation of data, MB, TCS, JT, KH; drafting of article, MB, TCS; critical revision of the article, MB, TCS, JT, KH; administrative or technical support, JT, KH; collection and assembly of data, MB, TCS, AK.
1. Umpierrez GE, Hellman R, Korytkowski MT, et al; Endocrine Society. Management of hyperglycemia in hospitalized patients in non-critical care setting: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2012;97:16–38.
2. Moghissi ES, Korytkowski MT, DiNardo M, et al; American Association of Clinical Endocrinologists; American Diabetes Association. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care2009;32:1119–31.
3. Coursin DB, Connery LE, Ketzler JT. Perioperative diabetic and hyperglycemic management issues. Crit Care Med 2004;32(4 Suppl):S116–25.
4. Pandit MK, Burke J, Gustafson AB, et al. Drug-induced disorders of glucose tolerance. Ann Intern Med 1993;118:529–39.
5. Donihi AC, Raval D, Saul M, et al. Prevalence and predictors of corticosteroid-related hyperglycemia in hospitalized patients. Endocr Pract 2006;12:358–62.
6. McHugh MD, Shang J, Sloane DM, Aiken LH. Risk factors for hospital-acquired ‘poor glycemic control’: a case-control study. Int J Qual Health Care 2011;23:44–51.
7. McCowen KC, Malhotra A, Bistrian BR. Stress-induced hyperglycemia. Crit Care Clin 2001;17:107–24.
8. Rady MY, Johnson DJ, Patel BM, et al. Influence of individual characteristics on outcome of glycemic control in intensive care unit patients with or without diabetes mellitus. Mayo Clin Proc 2005;80:1558–67.
9. Selvin E, Steffes MW, Zhu H, et al. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med 2010;362:800–11.
10. Kim KS, Kim SK, Lee YK, et al. Diagnostic value of glycated haemoglobin HbA(1c) for the early detection of diabetes in high-risk subjects. Diabet Med 2008;25:997–1000.
11. Quan H, Li B, Couris CM, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol 2011;173:676–82.
12. Kirk JK, Oldham EC. Hyperglycemia management using insulin in the acute care setting: therapies and strategies for care in the non-critically ill patient. Ann Pharmacother 2010;44:1222–30.
13. Draznin B, Gilden J, Golden SH, et al. Pathways to quality inpatient management of hyperglycemia and diabetes: a call to action. Diabetes Care 2013;36:1807–14.
14. Boord JB, Greevy RA, Braithwaite SS, et al. Evaluation of hospital glycemic control at US academic medical centers. J Hosp Med 2009;4:35–44.
15. Neubauer KM, Schaupp L, Plank J, et al. Failure to control hyperglycemia in noncritically ill diabetes patients despite standard glycemic management in a hospital setting. J Diabetes Sci Technol 2013;7:402–9.
16. Schnipper JL, Barsky EE, Shaykevich S, et al. Inpatient management of diabetes and hyperglycemia among general medicine patients at a large teaching hospital. J Hosp Med 2006;1:145–50.
17. Rohlfing CL, Wiedmeyer HM, Little RR, et al. Defining the relationship between plasma glucose and HbA(1c): analysis of glucose profiles and HbA(1c) in the Diabetes Control and Complications Trial. Diabetes Care 2002;25:275–8.
18. Nathan DM, Kuenen J, Borg R, et al; A1c-Derived Average Glucose Study Group. Translating the A1C assay into estimated average glucose values. Diabetes Care 2008;31:1473–8.
From Sharp HealthCare, San Diego, CA (Ms. Thompson, Mr. Koucheki, Dr. Holdy), National University, San Diego, CA (Dr. Smith), and University of California, Irvine (Dr. Bender).
Abstract
- Objective: Suboptimal glycemic control (SGC) puts hospitalized patients with diabetes at risk for poor outcomes. The purpose of this study was to quantify factors with predictive capacity to identify patients at risk for SGC during hospitalization.
- Methods: 32 baseline and demographic variables were extracted from the electronic records of 23,100 patients with diabetes hospitalized between 2009 and 2012. The rate of blood glucose values between 70 and 180 mg/dL was calculated for each patient. A predictive model for SGC was developed using regression modeling, standardized coefficients, and classification tree analysis. Odds ratios (ORs) were calculated to isolate adjusted odds of SGC for top predictors.
- Results: The final predictive model included 13 variables (C statistic = 0.88). HbA1c (OR, 0.60 [95% confidence interval {CI}, 0.58–0.61]), admission blood glucose (OR, 0.91 [CI, 0.91–0.92]), and steroid use (OR, 0.06 [CI 0.04–0.08]) were the highest-ranking predictors of SGC. HbA1c and SGC had a strong linear relationship (R2 = 0.99), with increasing odds for SGC as HbA1c increased. Admission blood glucose and SGC had a polynomial relationship (R2 = 0.95); increasing odds for SGC until 240 mg/dL; then odds started decreasing. Steroid use showed a steady threefold increase in odds for SGC across all rates of use.
- Conclusions: Poor preadmission diabetes control and inpatient steroid use strongly predict SGC. A range of thresholds for these predictors was empirically determined, providing a basis for targeted therapies on admission. Guidelines incorporating empirically derived thresholds should enhance the ability to achieve optimal glycemic control for hospitalized patients with diabetes.
Current recommendations for glycemic control of hospitalized patients include use of multidisciplinary diabetic care teams and standardized insulin order sets [1,2], yet there is still uncertainty how best to target such protocols for patients with diabetes at risk for suboptimal glycemic control [2,3]. Many factors are theorized to hinder optimal inpatient glycemic control, such as steroid use [4,5],comorbid states [6],severity of illness [7,8], and preadmission glycemic control [9,10]. However, there currently exists little evidence of these factors’ ability to predict suboptimal glycemic control (SGC). Identifying straightforward predictive factors of SGC would provide a clinically meaningful basis for targeted therapy. The purpose of this study was to describe the prevalence of a range of potential risk factors in a diverse hospitalized patient population with a secondary diagnosis of diabetes (types 1 and 2), and to determine which factors were predictive of SGC.
Methods
A retrospective cohort study design was used to identify factors predictive of inpatient SGC for patients admitted to any of 3 hospitals aligned with Sharp HealthCare (“Sharp”), a community-based, nonprofit integrated health system headquartered in San Diego, California, that serves more than 27% of the county’s 3 million-plus residents each year.
Inclusion Criteria
We extracted data for 23,100 patients hospitalized between January 2009 and December 2012 with a secondary diagnosis of diabetes (types 1 and 2), a length of stay (LOS) ≥ 3 days, and a minimum of 2 point-of-care (POC) blood glucose tests per day. The LOS and blood glucose minimum are standard criteria for Sharp glycemic monitoring to ensure a minimum quantity of blood glucose monitoring for glycemic management.
Glycemic Control (Independent Variable)
Glycemic control was defined as POC blood glucose values within the target range of 70 to 180 mg/dL during hospitalization. POC tests outside that range were defined as SGC. This range was determined based on current Sharp benchmark targets and was not adjusted for total number of blood glucose values.
Predictive Variables
Age, gender, race, ethnicity, payor, facility and LOS were extracted from Sharp’s data warehouse. Medical vs. surgical stay and major diagnostic category was determined from administrative diagnosis coding. Body mass index (BMI) was extracted from Sharp’s electronic health record. Risk of mortality and severity of illness were calculated using 3M APR-DRG proprietary software using administrative diagnosis coding. Comorbidities were determined based on administrative diagnosis codes per published guidelines [11]. Glycosylated hemoglobin (HbA1c) was obtained on admission for patients with a secondary diagnosis of diabetes as part of Sharp's multidisciplinary diabetes care management program and extracted from the electronic health record. Admission blood glucose was defined as the first documented POC blood glucose after admission. ICU stay was calculated as a continuous variable: the percent of LOS spent in the ICU. Steroid use was similarly calculated, and defined as oral or intravenous administration of any quantity or dosage of the following corticosteroids during each day of hospitalization: dexamethasone, hydrocortisone, prednisone, and/or methylprednisone. Adherence to Sharp's multidisciplinary diabetes care management program was measured by use of standardized insulin order sets. Sharp uses evidence-based order sets for continuous infusion and subcutaneous insulin management; subcutaneous orders include basal and rapid-acting insulin. We calculated the total time a person was on an order set during hospitalization by subtracting any time a patient did not have insulin ordered from the total LOS. This was transformed into a variable documenting the percent of LOS the patient was on an insulin order set. Average blood glucose for admission was calculated for all documented POC blood glucoses during admission, omitting the admission blood glucose (the first POC blood glucose of the admission).
Analysis
Univariate analyses including t tests and chi-square tests were conducted to investigate the unadjusted association between variables and glycemic control. Good glycemic control was defined as 90% of all POC blood glucose tests between 70 and 180 mg/dL based on empirical distribution and organization targets. A predictive model of inpatient glycemic control was then developed using a backward stepwise multivariable logistic regression approach. The data were split into a model building and validation set. Variables were included that represented both baseline and transitional state during the hospital stay to account for potentially mediating effects and a sensitivity analysis was conducted with them in and out of the final model to assess impact. Standardized coefficients were calculated to rank order variables in the model allowing indication of the variables with the greatest predictive impact on the outcome. Further investigation of the optimal classification points for the variables was conducted to indicate best differentiation of good glycemic control. Significant variables from the multivariable logistic regression were included in an exploratory classification tree analysis that recursively partitioned data in order to improve the fit, with optimal splitting identified over all variables at all possible split points. Classification tree cut-off points were used to further develop models identifying odds ratios for various thresholds for the top three predictive variables. A series of logistic regression models were then run with differing cut points of top 3 predictors to isolate adjusted odds of good glycemic control. Analytic data set building and statistical analyses were completed using SAS 9.4.
Results
Patient Characteristics
Table 1 shows patient demographic and clinical characteristics for the entire sample and for the top quartile (76% or greater POC blood glucose values within target range) and bottom quartile (25% or less POC blood glucose values within target range). Unadjusted results show a significant difference across quartiles for all factors except age, gender, dementia, rheumatic disease and paraplegia. Patients in the bottom 25th percentile (ie, the poorest control) were more likely than the total population to have a higher admission blood glucose (198 mg/dL vs. 153 mg/dL), higher HbA1c (8.53 [70mmol/mol] vs. 7.35 [57mmol/mol), a medical (74% vs. 66%) and/or respiratory (18% vs. 12%) diagnosis, corticosteroid use (17% vs. 27%), an insulin order set use (80% vs. 70%) and higher mean blood glucose during hospitalization (206.3 vs. 157.1 mg/dL). Patients with poorest control were less likely that the total population to have a high risk of mortality (11% vs. 15%) and severity of illness (13% vs. 18%). They also had less ICU care (8% vs. 13%), and a shorter LOS (5.82 vs. 7.82 days).
Predictive Modeling
Classification tree analysis resulted in the same top 3 predictors, but in a different order. The analysis also provided cut-off values that predict suboptimal glycemic control. Classification tree analysis showed admission blood glucose was the most influential predictor, with 164.5 mg/dL indicating the optimal cut-point for prediction of SGC, followed by HbA1c with an optimal cut-off point of 6.65% indicating prediction of SGC, followed by treatment with corticosteroids, with an optimal cut-off point of 24% of the LOS on corticosteroids indicating prediction of SGC.
Discussion
Evidence of patient characteristics that consistently predict suboptimal glycemic control during hospitalization is needed to better inform clinical decisions for inpatient glycemic management. Hospitalized patients would greatly benefit from glycemic protocols that incorporate risk stratification tools based evidence-based risk factors for poor glycemic control [12]. The science of inpatient glycemic management is in its infancy, however, and currently there is limited evidence to help identify at-risk populations and guide effective management for at-risk patient populations [13]. This study provides important data that can be used to develop risk stratification tools with implications for improved glycemic management of hospitalized patients with diabetes. Among the 32 factors included in the final multivariate logistic model (Table 2), 10 were statistically significant predictors of SGC. The top 3 predictors of SGC were HbA1c, admission blood glucose, and steroid use. These are straightforward, easily accessed factors that can become the basis for effective risk stratification and targeted clinical therapies.
This study showed that the degree of diabetes control prior to admission, as measured by HbA1c, is one of the strongest predictors of inpatient SGC. Patients with poorly controlled diabetes pre-admission had significantly higher rates of SGC than patients admitted with good diabetes control. Furthermore, there was a strong linear relationship between degree of pre-admission diabetes control and glycemic control during hospitalization: the higher the HbA1c on admission, the higher the odds are for poor glycemic control during hospitalization (Figure). The odds of SGC increased more than fivefold at an HbA1c of just 6.7% (50 mmol/mol) and continued to increase linearly as HbA1c increased. This increase occurred despite the fact that patients with poorly controlled pre-admission diabetes were found to have a significantly greater rate of insulin treatment using standardized order sets (which included basal and rapid-acting insulin) than the total sample in this study. So although patients with poorly controlled diabetes pre-admission were actively managed using evidence-based insulin order sets to control glycemia throughout their hospitalization, this did not translate to better glycemic control. Boord et al [14] and Neubauer et al [15] found similar results in their evaluation of glycemic control in hospitalized patients: while insulin use was high, glycemic control remained suboptimal. Similarly, Schnipper et al [16] found that adherence to insulin orders per se were not associated with better glucose control. They noted that the majority of patients with continued elevated blood glucose values did not have changes made to their insulin orders in response to suboptimal blood glucose values. These observations and this study’s findings suggest that fixed standardized subcutaneous orders may be more effective for well-controlled patients with diabetes than for patients with poorly controlled diabetes on admission. These patients need more frequent modifications to standard order sets based on clinical response during hospitalization to ensure good glycemic control.
This study confirmed the well known, highly correlated relationship between POC blood glucose and HbA1c. Rohlfing et al [17] previously documented a strong relationship (R = 0.82) between blood glucose and HbA1c in patients enrolled in the Diabetes Control and Complications Trial. Nathan et al [18]also documented a strong relationship (R= 0.84) between HbA1c and average blood glucose that was consistent across diverse populations. This study’s findings show odds for SGC based on admission blood glucose followed a very similar trend as HbA1c, and both factors showed much greater odds for SGC than steroid use, independent of other factors.
Admission blood glucose was the second best predictor of SBC using regression modeling (Table 2) and the strongest predictor using classification tree analysis. Odds for SGC were both very high and remarkably similar for a concomitant range of admission blood glucose and HbA1c values (Figure). The overlapping admission blood glucose range was 165 to 240 mg/dL, which corresponded to HbA1c values between 6.65 and 8.5 (49 mmol/mol–69 mmol/mol). The odds for SGC increased from a 6.5-fold increase in odds at 165 mg/dL to 22-fold increased odds at 240 mg/dL. This corresponded to a 5.5-fold increase in odds with HbA1c of 6.65 (49 mmol/mol) to a 19-fold increase in odds with HbA1c of 8.5 (69 mmol/mol).
Notably, an admission blood glucose as low as 164 mg/dL significantly increased the odds of SGC. This relatively mild hyperglycemia is not typically considered a signifier for difficult inpatient glycemic management. Furthermore, the odds of SGC continue to increase until admission blood glucose reached approximately 240 mg/dL, at which point the odds start declining (Figure). This suggests that only exceptionally elevated admission blood glucoses triggered prompt insulin treatment on admission. The data from this study suggests targeted action for an admission blood glucose as low as 164 mg/dL is just as necessary as for those admitted with a much higher blood glucose to ensure optimal glycemic control throughout hospitalization. Implications are that admission blood glucose may be an inexpensive, straightforward, and readily available predictor of SGC and marker for targeted clinical action, especially for hospitals that do not routinely order HbA1c labs during hospitalization.
Steroid treatment was the third strongest predictor of SGC. Additional analysis showed that any proportion of hospital stay with steroid administration resulted in a stable threefold increased odds for SGC, adjusting for other predictive factors (Figure). Developing insulin treatment therapies that are tailored to patients that will be administered steroids at any point during their hospitalization may be a reasonable strategy to reduce SGC in this population. Based on the results of this study and other Sharp data, Sharp is currently piloting a steroid insulin order set that is available in the electronic health record to hospital physicians for use with any patient that is administered steroids. The order set includes eating and non-eating standards, intensified meal dose coverage, a lower blood glucose threshold for starting correction dosing, and a diabetic nurse educator consult. Evaluation will include appropriate order set usage, rate of glycemic control and extreme blood glucose values.
Limitations
A limitation of this study is that it used observational data and was conducted within a single health care system, thus potentially reducing generalizability. Nevertheless, the sample size was large, there were many clinical and demographic characteristics to be leveraged in the analysis, the statistical approach utilized a complementary regression and classification approach to adjust and present the findings, and the sample included patients from three hospitals across San Diego County with diverse patient populations.
Conclusion
While much progress has been made understanding the need for appropriate glycemic management for patient with diabetes to reduce their risk for adverse outcomes, the knowledge base is still quite limited, especially regarding optimal glycemic limits for diverse patient populations. There is a need to identify predictors of SGC if risk stratification tools are to be built that can help target therapies with the potential to reducing the risk of poor glycemic control and adverse patient outcomes. This study identified 3 readily available factors—admission blood glucose, HbA1c and steroid use—that strongly predict SGC, controlling for other patient risk factors. In general, poor pre-admission diabetes control and inpatient steroid use strongly predict SGC, and the data suggests that earlier and frequently calibrated intervention may improve inpatient glycemic control for these patient populations. We identified a range of thresholds for these variables that may provide a basis for targeted treatment on admission. In conclusion, this study has important implications for meaningful use of readily available factors to identify patients at risk for SGC. Clinical therapies and guidelines incorporating empirically derived risk-stratification tools should enhance the ability to achieve the triple aim of better health, better care quality and more efficient care costs for hospitalized patient with diabetes.
Corresponding author: Miriam Bender, PhD, RN, University of California, Irvine, 252 Berk Hall, Irvine, CA 92697, [email protected].
Financial disclosures: None.
Author contributions: Author contributions: conception and design, MB, TCS, JT, KH; analysis and interpretation of data, MB, TCS, JT, KH; drafting of article, MB, TCS; critical revision of the article, MB, TCS, JT, KH; administrative or technical support, JT, KH; collection and assembly of data, MB, TCS, AK.
From Sharp HealthCare, San Diego, CA (Ms. Thompson, Mr. Koucheki, Dr. Holdy), National University, San Diego, CA (Dr. Smith), and University of California, Irvine (Dr. Bender).
Abstract
- Objective: Suboptimal glycemic control (SGC) puts hospitalized patients with diabetes at risk for poor outcomes. The purpose of this study was to quantify factors with predictive capacity to identify patients at risk for SGC during hospitalization.
- Methods: 32 baseline and demographic variables were extracted from the electronic records of 23,100 patients with diabetes hospitalized between 2009 and 2012. The rate of blood glucose values between 70 and 180 mg/dL was calculated for each patient. A predictive model for SGC was developed using regression modeling, standardized coefficients, and classification tree analysis. Odds ratios (ORs) were calculated to isolate adjusted odds of SGC for top predictors.
- Results: The final predictive model included 13 variables (C statistic = 0.88). HbA1c (OR, 0.60 [95% confidence interval {CI}, 0.58–0.61]), admission blood glucose (OR, 0.91 [CI, 0.91–0.92]), and steroid use (OR, 0.06 [CI 0.04–0.08]) were the highest-ranking predictors of SGC. HbA1c and SGC had a strong linear relationship (R2 = 0.99), with increasing odds for SGC as HbA1c increased. Admission blood glucose and SGC had a polynomial relationship (R2 = 0.95); increasing odds for SGC until 240 mg/dL; then odds started decreasing. Steroid use showed a steady threefold increase in odds for SGC across all rates of use.
- Conclusions: Poor preadmission diabetes control and inpatient steroid use strongly predict SGC. A range of thresholds for these predictors was empirically determined, providing a basis for targeted therapies on admission. Guidelines incorporating empirically derived thresholds should enhance the ability to achieve optimal glycemic control for hospitalized patients with diabetes.
Current recommendations for glycemic control of hospitalized patients include use of multidisciplinary diabetic care teams and standardized insulin order sets [1,2], yet there is still uncertainty how best to target such protocols for patients with diabetes at risk for suboptimal glycemic control [2,3]. Many factors are theorized to hinder optimal inpatient glycemic control, such as steroid use [4,5],comorbid states [6],severity of illness [7,8], and preadmission glycemic control [9,10]. However, there currently exists little evidence of these factors’ ability to predict suboptimal glycemic control (SGC). Identifying straightforward predictive factors of SGC would provide a clinically meaningful basis for targeted therapy. The purpose of this study was to describe the prevalence of a range of potential risk factors in a diverse hospitalized patient population with a secondary diagnosis of diabetes (types 1 and 2), and to determine which factors were predictive of SGC.
Methods
A retrospective cohort study design was used to identify factors predictive of inpatient SGC for patients admitted to any of 3 hospitals aligned with Sharp HealthCare (“Sharp”), a community-based, nonprofit integrated health system headquartered in San Diego, California, that serves more than 27% of the county’s 3 million-plus residents each year.
Inclusion Criteria
We extracted data for 23,100 patients hospitalized between January 2009 and December 2012 with a secondary diagnosis of diabetes (types 1 and 2), a length of stay (LOS) ≥ 3 days, and a minimum of 2 point-of-care (POC) blood glucose tests per day. The LOS and blood glucose minimum are standard criteria for Sharp glycemic monitoring to ensure a minimum quantity of blood glucose monitoring for glycemic management.
Glycemic Control (Independent Variable)
Glycemic control was defined as POC blood glucose values within the target range of 70 to 180 mg/dL during hospitalization. POC tests outside that range were defined as SGC. This range was determined based on current Sharp benchmark targets and was not adjusted for total number of blood glucose values.
Predictive Variables
Age, gender, race, ethnicity, payor, facility and LOS were extracted from Sharp’s data warehouse. Medical vs. surgical stay and major diagnostic category was determined from administrative diagnosis coding. Body mass index (BMI) was extracted from Sharp’s electronic health record. Risk of mortality and severity of illness were calculated using 3M APR-DRG proprietary software using administrative diagnosis coding. Comorbidities were determined based on administrative diagnosis codes per published guidelines [11]. Glycosylated hemoglobin (HbA1c) was obtained on admission for patients with a secondary diagnosis of diabetes as part of Sharp's multidisciplinary diabetes care management program and extracted from the electronic health record. Admission blood glucose was defined as the first documented POC blood glucose after admission. ICU stay was calculated as a continuous variable: the percent of LOS spent in the ICU. Steroid use was similarly calculated, and defined as oral or intravenous administration of any quantity or dosage of the following corticosteroids during each day of hospitalization: dexamethasone, hydrocortisone, prednisone, and/or methylprednisone. Adherence to Sharp's multidisciplinary diabetes care management program was measured by use of standardized insulin order sets. Sharp uses evidence-based order sets for continuous infusion and subcutaneous insulin management; subcutaneous orders include basal and rapid-acting insulin. We calculated the total time a person was on an order set during hospitalization by subtracting any time a patient did not have insulin ordered from the total LOS. This was transformed into a variable documenting the percent of LOS the patient was on an insulin order set. Average blood glucose for admission was calculated for all documented POC blood glucoses during admission, omitting the admission blood glucose (the first POC blood glucose of the admission).
Analysis
Univariate analyses including t tests and chi-square tests were conducted to investigate the unadjusted association between variables and glycemic control. Good glycemic control was defined as 90% of all POC blood glucose tests between 70 and 180 mg/dL based on empirical distribution and organization targets. A predictive model of inpatient glycemic control was then developed using a backward stepwise multivariable logistic regression approach. The data were split into a model building and validation set. Variables were included that represented both baseline and transitional state during the hospital stay to account for potentially mediating effects and a sensitivity analysis was conducted with them in and out of the final model to assess impact. Standardized coefficients were calculated to rank order variables in the model allowing indication of the variables with the greatest predictive impact on the outcome. Further investigation of the optimal classification points for the variables was conducted to indicate best differentiation of good glycemic control. Significant variables from the multivariable logistic regression were included in an exploratory classification tree analysis that recursively partitioned data in order to improve the fit, with optimal splitting identified over all variables at all possible split points. Classification tree cut-off points were used to further develop models identifying odds ratios for various thresholds for the top three predictive variables. A series of logistic regression models were then run with differing cut points of top 3 predictors to isolate adjusted odds of good glycemic control. Analytic data set building and statistical analyses were completed using SAS 9.4.
Results
Patient Characteristics
Table 1 shows patient demographic and clinical characteristics for the entire sample and for the top quartile (76% or greater POC blood glucose values within target range) and bottom quartile (25% or less POC blood glucose values within target range). Unadjusted results show a significant difference across quartiles for all factors except age, gender, dementia, rheumatic disease and paraplegia. Patients in the bottom 25th percentile (ie, the poorest control) were more likely than the total population to have a higher admission blood glucose (198 mg/dL vs. 153 mg/dL), higher HbA1c (8.53 [70mmol/mol] vs. 7.35 [57mmol/mol), a medical (74% vs. 66%) and/or respiratory (18% vs. 12%) diagnosis, corticosteroid use (17% vs. 27%), an insulin order set use (80% vs. 70%) and higher mean blood glucose during hospitalization (206.3 vs. 157.1 mg/dL). Patients with poorest control were less likely that the total population to have a high risk of mortality (11% vs. 15%) and severity of illness (13% vs. 18%). They also had less ICU care (8% vs. 13%), and a shorter LOS (5.82 vs. 7.82 days).
Predictive Modeling
Classification tree analysis resulted in the same top 3 predictors, but in a different order. The analysis also provided cut-off values that predict suboptimal glycemic control. Classification tree analysis showed admission blood glucose was the most influential predictor, with 164.5 mg/dL indicating the optimal cut-point for prediction of SGC, followed by HbA1c with an optimal cut-off point of 6.65% indicating prediction of SGC, followed by treatment with corticosteroids, with an optimal cut-off point of 24% of the LOS on corticosteroids indicating prediction of SGC.
Discussion
Evidence of patient characteristics that consistently predict suboptimal glycemic control during hospitalization is needed to better inform clinical decisions for inpatient glycemic management. Hospitalized patients would greatly benefit from glycemic protocols that incorporate risk stratification tools based evidence-based risk factors for poor glycemic control [12]. The science of inpatient glycemic management is in its infancy, however, and currently there is limited evidence to help identify at-risk populations and guide effective management for at-risk patient populations [13]. This study provides important data that can be used to develop risk stratification tools with implications for improved glycemic management of hospitalized patients with diabetes. Among the 32 factors included in the final multivariate logistic model (Table 2), 10 were statistically significant predictors of SGC. The top 3 predictors of SGC were HbA1c, admission blood glucose, and steroid use. These are straightforward, easily accessed factors that can become the basis for effective risk stratification and targeted clinical therapies.
This study showed that the degree of diabetes control prior to admission, as measured by HbA1c, is one of the strongest predictors of inpatient SGC. Patients with poorly controlled diabetes pre-admission had significantly higher rates of SGC than patients admitted with good diabetes control. Furthermore, there was a strong linear relationship between degree of pre-admission diabetes control and glycemic control during hospitalization: the higher the HbA1c on admission, the higher the odds are for poor glycemic control during hospitalization (Figure). The odds of SGC increased more than fivefold at an HbA1c of just 6.7% (50 mmol/mol) and continued to increase linearly as HbA1c increased. This increase occurred despite the fact that patients with poorly controlled pre-admission diabetes were found to have a significantly greater rate of insulin treatment using standardized order sets (which included basal and rapid-acting insulin) than the total sample in this study. So although patients with poorly controlled diabetes pre-admission were actively managed using evidence-based insulin order sets to control glycemia throughout their hospitalization, this did not translate to better glycemic control. Boord et al [14] and Neubauer et al [15] found similar results in their evaluation of glycemic control in hospitalized patients: while insulin use was high, glycemic control remained suboptimal. Similarly, Schnipper et al [16] found that adherence to insulin orders per se were not associated with better glucose control. They noted that the majority of patients with continued elevated blood glucose values did not have changes made to their insulin orders in response to suboptimal blood glucose values. These observations and this study’s findings suggest that fixed standardized subcutaneous orders may be more effective for well-controlled patients with diabetes than for patients with poorly controlled diabetes on admission. These patients need more frequent modifications to standard order sets based on clinical response during hospitalization to ensure good glycemic control.
This study confirmed the well known, highly correlated relationship between POC blood glucose and HbA1c. Rohlfing et al [17] previously documented a strong relationship (R = 0.82) between blood glucose and HbA1c in patients enrolled in the Diabetes Control and Complications Trial. Nathan et al [18]also documented a strong relationship (R= 0.84) between HbA1c and average blood glucose that was consistent across diverse populations. This study’s findings show odds for SGC based on admission blood glucose followed a very similar trend as HbA1c, and both factors showed much greater odds for SGC than steroid use, independent of other factors.
Admission blood glucose was the second best predictor of SBC using regression modeling (Table 2) and the strongest predictor using classification tree analysis. Odds for SGC were both very high and remarkably similar for a concomitant range of admission blood glucose and HbA1c values (Figure). The overlapping admission blood glucose range was 165 to 240 mg/dL, which corresponded to HbA1c values between 6.65 and 8.5 (49 mmol/mol–69 mmol/mol). The odds for SGC increased from a 6.5-fold increase in odds at 165 mg/dL to 22-fold increased odds at 240 mg/dL. This corresponded to a 5.5-fold increase in odds with HbA1c of 6.65 (49 mmol/mol) to a 19-fold increase in odds with HbA1c of 8.5 (69 mmol/mol).
Notably, an admission blood glucose as low as 164 mg/dL significantly increased the odds of SGC. This relatively mild hyperglycemia is not typically considered a signifier for difficult inpatient glycemic management. Furthermore, the odds of SGC continue to increase until admission blood glucose reached approximately 240 mg/dL, at which point the odds start declining (Figure). This suggests that only exceptionally elevated admission blood glucoses triggered prompt insulin treatment on admission. The data from this study suggests targeted action for an admission blood glucose as low as 164 mg/dL is just as necessary as for those admitted with a much higher blood glucose to ensure optimal glycemic control throughout hospitalization. Implications are that admission blood glucose may be an inexpensive, straightforward, and readily available predictor of SGC and marker for targeted clinical action, especially for hospitals that do not routinely order HbA1c labs during hospitalization.
Steroid treatment was the third strongest predictor of SGC. Additional analysis showed that any proportion of hospital stay with steroid administration resulted in a stable threefold increased odds for SGC, adjusting for other predictive factors (Figure). Developing insulin treatment therapies that are tailored to patients that will be administered steroids at any point during their hospitalization may be a reasonable strategy to reduce SGC in this population. Based on the results of this study and other Sharp data, Sharp is currently piloting a steroid insulin order set that is available in the electronic health record to hospital physicians for use with any patient that is administered steroids. The order set includes eating and non-eating standards, intensified meal dose coverage, a lower blood glucose threshold for starting correction dosing, and a diabetic nurse educator consult. Evaluation will include appropriate order set usage, rate of glycemic control and extreme blood glucose values.
Limitations
A limitation of this study is that it used observational data and was conducted within a single health care system, thus potentially reducing generalizability. Nevertheless, the sample size was large, there were many clinical and demographic characteristics to be leveraged in the analysis, the statistical approach utilized a complementary regression and classification approach to adjust and present the findings, and the sample included patients from three hospitals across San Diego County with diverse patient populations.
Conclusion
While much progress has been made understanding the need for appropriate glycemic management for patient with diabetes to reduce their risk for adverse outcomes, the knowledge base is still quite limited, especially regarding optimal glycemic limits for diverse patient populations. There is a need to identify predictors of SGC if risk stratification tools are to be built that can help target therapies with the potential to reducing the risk of poor glycemic control and adverse patient outcomes. This study identified 3 readily available factors—admission blood glucose, HbA1c and steroid use—that strongly predict SGC, controlling for other patient risk factors. In general, poor pre-admission diabetes control and inpatient steroid use strongly predict SGC, and the data suggests that earlier and frequently calibrated intervention may improve inpatient glycemic control for these patient populations. We identified a range of thresholds for these variables that may provide a basis for targeted treatment on admission. In conclusion, this study has important implications for meaningful use of readily available factors to identify patients at risk for SGC. Clinical therapies and guidelines incorporating empirically derived risk-stratification tools should enhance the ability to achieve the triple aim of better health, better care quality and more efficient care costs for hospitalized patient with diabetes.
Corresponding author: Miriam Bender, PhD, RN, University of California, Irvine, 252 Berk Hall, Irvine, CA 92697, [email protected].
Financial disclosures: None.
Author contributions: Author contributions: conception and design, MB, TCS, JT, KH; analysis and interpretation of data, MB, TCS, JT, KH; drafting of article, MB, TCS; critical revision of the article, MB, TCS, JT, KH; administrative or technical support, JT, KH; collection and assembly of data, MB, TCS, AK.
1. Umpierrez GE, Hellman R, Korytkowski MT, et al; Endocrine Society. Management of hyperglycemia in hospitalized patients in non-critical care setting: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2012;97:16–38.
2. Moghissi ES, Korytkowski MT, DiNardo M, et al; American Association of Clinical Endocrinologists; American Diabetes Association. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care2009;32:1119–31.
3. Coursin DB, Connery LE, Ketzler JT. Perioperative diabetic and hyperglycemic management issues. Crit Care Med 2004;32(4 Suppl):S116–25.
4. Pandit MK, Burke J, Gustafson AB, et al. Drug-induced disorders of glucose tolerance. Ann Intern Med 1993;118:529–39.
5. Donihi AC, Raval D, Saul M, et al. Prevalence and predictors of corticosteroid-related hyperglycemia in hospitalized patients. Endocr Pract 2006;12:358–62.
6. McHugh MD, Shang J, Sloane DM, Aiken LH. Risk factors for hospital-acquired ‘poor glycemic control’: a case-control study. Int J Qual Health Care 2011;23:44–51.
7. McCowen KC, Malhotra A, Bistrian BR. Stress-induced hyperglycemia. Crit Care Clin 2001;17:107–24.
8. Rady MY, Johnson DJ, Patel BM, et al. Influence of individual characteristics on outcome of glycemic control in intensive care unit patients with or without diabetes mellitus. Mayo Clin Proc 2005;80:1558–67.
9. Selvin E, Steffes MW, Zhu H, et al. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med 2010;362:800–11.
10. Kim KS, Kim SK, Lee YK, et al. Diagnostic value of glycated haemoglobin HbA(1c) for the early detection of diabetes in high-risk subjects. Diabet Med 2008;25:997–1000.
11. Quan H, Li B, Couris CM, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol 2011;173:676–82.
12. Kirk JK, Oldham EC. Hyperglycemia management using insulin in the acute care setting: therapies and strategies for care in the non-critically ill patient. Ann Pharmacother 2010;44:1222–30.
13. Draznin B, Gilden J, Golden SH, et al. Pathways to quality inpatient management of hyperglycemia and diabetes: a call to action. Diabetes Care 2013;36:1807–14.
14. Boord JB, Greevy RA, Braithwaite SS, et al. Evaluation of hospital glycemic control at US academic medical centers. J Hosp Med 2009;4:35–44.
15. Neubauer KM, Schaupp L, Plank J, et al. Failure to control hyperglycemia in noncritically ill diabetes patients despite standard glycemic management in a hospital setting. J Diabetes Sci Technol 2013;7:402–9.
16. Schnipper JL, Barsky EE, Shaykevich S, et al. Inpatient management of diabetes and hyperglycemia among general medicine patients at a large teaching hospital. J Hosp Med 2006;1:145–50.
17. Rohlfing CL, Wiedmeyer HM, Little RR, et al. Defining the relationship between plasma glucose and HbA(1c): analysis of glucose profiles and HbA(1c) in the Diabetes Control and Complications Trial. Diabetes Care 2002;25:275–8.
18. Nathan DM, Kuenen J, Borg R, et al; A1c-Derived Average Glucose Study Group. Translating the A1C assay into estimated average glucose values. Diabetes Care 2008;31:1473–8.
1. Umpierrez GE, Hellman R, Korytkowski MT, et al; Endocrine Society. Management of hyperglycemia in hospitalized patients in non-critical care setting: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2012;97:16–38.
2. Moghissi ES, Korytkowski MT, DiNardo M, et al; American Association of Clinical Endocrinologists; American Diabetes Association. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care2009;32:1119–31.
3. Coursin DB, Connery LE, Ketzler JT. Perioperative diabetic and hyperglycemic management issues. Crit Care Med 2004;32(4 Suppl):S116–25.
4. Pandit MK, Burke J, Gustafson AB, et al. Drug-induced disorders of glucose tolerance. Ann Intern Med 1993;118:529–39.
5. Donihi AC, Raval D, Saul M, et al. Prevalence and predictors of corticosteroid-related hyperglycemia in hospitalized patients. Endocr Pract 2006;12:358–62.
6. McHugh MD, Shang J, Sloane DM, Aiken LH. Risk factors for hospital-acquired ‘poor glycemic control’: a case-control study. Int J Qual Health Care 2011;23:44–51.
7. McCowen KC, Malhotra A, Bistrian BR. Stress-induced hyperglycemia. Crit Care Clin 2001;17:107–24.
8. Rady MY, Johnson DJ, Patel BM, et al. Influence of individual characteristics on outcome of glycemic control in intensive care unit patients with or without diabetes mellitus. Mayo Clin Proc 2005;80:1558–67.
9. Selvin E, Steffes MW, Zhu H, et al. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med 2010;362:800–11.
10. Kim KS, Kim SK, Lee YK, et al. Diagnostic value of glycated haemoglobin HbA(1c) for the early detection of diabetes in high-risk subjects. Diabet Med 2008;25:997–1000.
11. Quan H, Li B, Couris CM, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol 2011;173:676–82.
12. Kirk JK, Oldham EC. Hyperglycemia management using insulin in the acute care setting: therapies and strategies for care in the non-critically ill patient. Ann Pharmacother 2010;44:1222–30.
13. Draznin B, Gilden J, Golden SH, et al. Pathways to quality inpatient management of hyperglycemia and diabetes: a call to action. Diabetes Care 2013;36:1807–14.
14. Boord JB, Greevy RA, Braithwaite SS, et al. Evaluation of hospital glycemic control at US academic medical centers. J Hosp Med 2009;4:35–44.
15. Neubauer KM, Schaupp L, Plank J, et al. Failure to control hyperglycemia in noncritically ill diabetes patients despite standard glycemic management in a hospital setting. J Diabetes Sci Technol 2013;7:402–9.
16. Schnipper JL, Barsky EE, Shaykevich S, et al. Inpatient management of diabetes and hyperglycemia among general medicine patients at a large teaching hospital. J Hosp Med 2006;1:145–50.
17. Rohlfing CL, Wiedmeyer HM, Little RR, et al. Defining the relationship between plasma glucose and HbA(1c): analysis of glucose profiles and HbA(1c) in the Diabetes Control and Complications Trial. Diabetes Care 2002;25:275–8.
18. Nathan DM, Kuenen J, Borg R, et al; A1c-Derived Average Glucose Study Group. Translating the A1C assay into estimated average glucose values. Diabetes Care 2008;31:1473–8.
Mouse model could aid study of malaria

Plasmodium vivax infection
Image by Mae Melvin
Researchers say they have developed a human-chimeric mouse model that can advance the study of the malaria parasite Plasmodium vivax.
The model is engineered to grow human livers that can be infected with P vivax and used for investigations into parasite development, dormancy, and activation, as well as the effect certain drugs have on each aspect of the infection.
The researchers described this model in Cell Host & Microbe.
Stefan Kappe, PhD, of Seattle Biomedical Research Institute in Washington, and his colleagues noted that P vivax malaria parasites are more resistant to control and elimination than other malaria parasites because, after infection, they fall dormant within the liver for months to years.
When these dormant parasites eventually become activated, they replicate, infect the bloodstream, and cause relapsing malaria.
The lack of tractable P vivax animal models has made it difficult to examine P vivax liver-stage infection, but Dr Kappe and his colleagues found a solution in a model known as FRG KO huHep.
To create the FRG KO huHep model, the team transplanted human hepatocytes in the severely immunocompromised FRG KO mouse, which has deletions in fumarylacetoacetate hydrolase (FAH), recombinationactivating gene 2 (Rag2), and interleukin-2 receptor subunit gamma (Il2rg).
The researchers found that FRG KO huHep mice support P vivax sporozoite infection, liver-stage development, and the formation and persistence of dormant liver-stage parasites.
In addition, experiments with this model revealed that the antimalarial drug primaquine could prevent and eliminate liver-stage infection.
The researchers therefore concluded that P vivax-infected FRG KO huHep mice are a suitable model for studying liver-stage development and dormancy and may facilitate the discovery of drugs targeting relapsing malaria.
“This model is a real game-changer,” said Ivo Mueller, PhD, a malaria expert at the Walter and Eliza Hall Institute in Melbourne, Victoria, Australia, who was not involved in this research.
“For the first time, we now have a realistic opportunity that not only allows us to directly study this normally hidden P vivax life-stage, but also to test new drugs and vaccines. This new model is an essential resource in our quest to develop the new anti-vivax interventions that we will need to eliminate P vivax malaria.” ![]()

Plasmodium vivax infection
Image by Mae Melvin
Researchers say they have developed a human-chimeric mouse model that can advance the study of the malaria parasite Plasmodium vivax.
The model is engineered to grow human livers that can be infected with P vivax and used for investigations into parasite development, dormancy, and activation, as well as the effect certain drugs have on each aspect of the infection.
The researchers described this model in Cell Host & Microbe.
Stefan Kappe, PhD, of Seattle Biomedical Research Institute in Washington, and his colleagues noted that P vivax malaria parasites are more resistant to control and elimination than other malaria parasites because, after infection, they fall dormant within the liver for months to years.
When these dormant parasites eventually become activated, they replicate, infect the bloodstream, and cause relapsing malaria.
The lack of tractable P vivax animal models has made it difficult to examine P vivax liver-stage infection, but Dr Kappe and his colleagues found a solution in a model known as FRG KO huHep.
To create the FRG KO huHep model, the team transplanted human hepatocytes in the severely immunocompromised FRG KO mouse, which has deletions in fumarylacetoacetate hydrolase (FAH), recombinationactivating gene 2 (Rag2), and interleukin-2 receptor subunit gamma (Il2rg).
The researchers found that FRG KO huHep mice support P vivax sporozoite infection, liver-stage development, and the formation and persistence of dormant liver-stage parasites.
In addition, experiments with this model revealed that the antimalarial drug primaquine could prevent and eliminate liver-stage infection.
The researchers therefore concluded that P vivax-infected FRG KO huHep mice are a suitable model for studying liver-stage development and dormancy and may facilitate the discovery of drugs targeting relapsing malaria.
“This model is a real game-changer,” said Ivo Mueller, PhD, a malaria expert at the Walter and Eliza Hall Institute in Melbourne, Victoria, Australia, who was not involved in this research.
“For the first time, we now have a realistic opportunity that not only allows us to directly study this normally hidden P vivax life-stage, but also to test new drugs and vaccines. This new model is an essential resource in our quest to develop the new anti-vivax interventions that we will need to eliminate P vivax malaria.” ![]()

Plasmodium vivax infection
Image by Mae Melvin
Researchers say they have developed a human-chimeric mouse model that can advance the study of the malaria parasite Plasmodium vivax.
The model is engineered to grow human livers that can be infected with P vivax and used for investigations into parasite development, dormancy, and activation, as well as the effect certain drugs have on each aspect of the infection.
The researchers described this model in Cell Host & Microbe.
Stefan Kappe, PhD, of Seattle Biomedical Research Institute in Washington, and his colleagues noted that P vivax malaria parasites are more resistant to control and elimination than other malaria parasites because, after infection, they fall dormant within the liver for months to years.
When these dormant parasites eventually become activated, they replicate, infect the bloodstream, and cause relapsing malaria.
The lack of tractable P vivax animal models has made it difficult to examine P vivax liver-stage infection, but Dr Kappe and his colleagues found a solution in a model known as FRG KO huHep.
To create the FRG KO huHep model, the team transplanted human hepatocytes in the severely immunocompromised FRG KO mouse, which has deletions in fumarylacetoacetate hydrolase (FAH), recombinationactivating gene 2 (Rag2), and interleukin-2 receptor subunit gamma (Il2rg).
The researchers found that FRG KO huHep mice support P vivax sporozoite infection, liver-stage development, and the formation and persistence of dormant liver-stage parasites.
In addition, experiments with this model revealed that the antimalarial drug primaquine could prevent and eliminate liver-stage infection.
The researchers therefore concluded that P vivax-infected FRG KO huHep mice are a suitable model for studying liver-stage development and dormancy and may facilitate the discovery of drugs targeting relapsing malaria.
“This model is a real game-changer,” said Ivo Mueller, PhD, a malaria expert at the Walter and Eliza Hall Institute in Melbourne, Victoria, Australia, who was not involved in this research.
“For the first time, we now have a realistic opportunity that not only allows us to directly study this normally hidden P vivax life-stage, but also to test new drugs and vaccines. This new model is an essential resource in our quest to develop the new anti-vivax interventions that we will need to eliminate P vivax malaria.” ![]()
Medicare Standard Practical Solution to Medical Coding Complexity
In the article “Common Coding Mistakes Hospitalists Should Avoid” in the August 2014 issue of The Hospitalist, the author states:
“For inpatient care, an established problem is defined as one in which a care plan has been generated by the physician (or same specialty group practice member) during the current hospitalization.”
This definition contradicts what I have been told in other coding courses regarding new vs. established problems relative to the examiner. It has been my understanding that when [I am] rotating on service and I have not seen that particular patient during the current admission, all of the current problems are new to me, even if previously identified by another member of my group. This [situation] results in a higher complexity of medical decision-making, which is reflective of the increased time spent learning a new patient when coming on service. I would appreciate clarification from the author.
–Matt George, MD,
medical director, MBHS Hospitalists
Billing and coding expert Carol Pohlig, BSN, RN, CPC, ACS, explains:
Be mindful when attending coding courses that are not contractor sponsored, as they may not validate the geographical interpretations of the rules for providers. There are several factors to consider when crediting the physician with “new” or “established” problems.
CMS documentation guidelines state: “Decision making with respect to a diagnosed problem is easier than that for an identified but undiagnosed problem.1
- For a presenting problem with an established diagnosis, the record should reflect whether the problem is:
- Improved, well-controlled, resolving, or resolved or
- Inadequately controlled, worsening, or failing to change as expected.
- For a presenting problem without an established diagnosis, the assessment or clinical impression may be stated in the form of differential diagnoses or as a “possible,” “probable,” or “rule out” diagnosis.
Although Medicare contractors utilize the Marshfield Clinic Scoring Tool when reviewing evaluation and management (E/M) services, a tool that historically refers to the “examiner” when considering the patient’s diagnoses, not all accept this tool-inspired standard, particularly in the advent of electronic health record accessibility and the idea that same-specialty physicians in a group practice are viewed as an individual physician.2,3
Reviewing information and familiarization of patients is often considered pre-service work and factored into the payment for E/M services. More importantly, the feasibility of an auditor being able to distinguish new vs. established problems at the level of the “examiner” is decreased when auditing a single date of service. Non-Medicare payers who audit E/M services do not necessarily follow contractor-specific guidelines but, rather, general CMS guidelines.
Therefore, without knowing the insurer or their interpretation at the time of service or visit level selection, the CMS-developed standard is the most practical application when considering the complexity of the encounter.
References
- Centers for Medicare and Medicaid Services. Department of Health and Human Services. Evaluation and management services guide. Available at: http://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/downloads//eval_mgmt_serv_guide-ICN006764.pdf. Accessed November 13, 2014.
- National Government Services. Evaluation and management documentation training too. Available at: http://www.ngsmedicare.com/ngs/wcm/connect/3632a905-b697-4266-8fc0-2aa2a84fedb2/1074_0514_EM_Documentation_Training_Tool_508.pdf?MOD=AJPERES. Accessed November 13, 2014.
- Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual. Chapter 12 – Physicians/nonphysician practitioners. Available at: http://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c12.pdf. Accessed November 13, 2014.
In the article “Common Coding Mistakes Hospitalists Should Avoid” in the August 2014 issue of The Hospitalist, the author states:
“For inpatient care, an established problem is defined as one in which a care plan has been generated by the physician (or same specialty group practice member) during the current hospitalization.”
This definition contradicts what I have been told in other coding courses regarding new vs. established problems relative to the examiner. It has been my understanding that when [I am] rotating on service and I have not seen that particular patient during the current admission, all of the current problems are new to me, even if previously identified by another member of my group. This [situation] results in a higher complexity of medical decision-making, which is reflective of the increased time spent learning a new patient when coming on service. I would appreciate clarification from the author.
–Matt George, MD,
medical director, MBHS Hospitalists
Billing and coding expert Carol Pohlig, BSN, RN, CPC, ACS, explains:
Be mindful when attending coding courses that are not contractor sponsored, as they may not validate the geographical interpretations of the rules for providers. There are several factors to consider when crediting the physician with “new” or “established” problems.
CMS documentation guidelines state: “Decision making with respect to a diagnosed problem is easier than that for an identified but undiagnosed problem.1
- For a presenting problem with an established diagnosis, the record should reflect whether the problem is:
- Improved, well-controlled, resolving, or resolved or
- Inadequately controlled, worsening, or failing to change as expected.
- For a presenting problem without an established diagnosis, the assessment or clinical impression may be stated in the form of differential diagnoses or as a “possible,” “probable,” or “rule out” diagnosis.
Although Medicare contractors utilize the Marshfield Clinic Scoring Tool when reviewing evaluation and management (E/M) services, a tool that historically refers to the “examiner” when considering the patient’s diagnoses, not all accept this tool-inspired standard, particularly in the advent of electronic health record accessibility and the idea that same-specialty physicians in a group practice are viewed as an individual physician.2,3
Reviewing information and familiarization of patients is often considered pre-service work and factored into the payment for E/M services. More importantly, the feasibility of an auditor being able to distinguish new vs. established problems at the level of the “examiner” is decreased when auditing a single date of service. Non-Medicare payers who audit E/M services do not necessarily follow contractor-specific guidelines but, rather, general CMS guidelines.
Therefore, without knowing the insurer or their interpretation at the time of service or visit level selection, the CMS-developed standard is the most practical application when considering the complexity of the encounter.
References
- Centers for Medicare and Medicaid Services. Department of Health and Human Services. Evaluation and management services guide. Available at: http://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/downloads//eval_mgmt_serv_guide-ICN006764.pdf. Accessed November 13, 2014.
- National Government Services. Evaluation and management documentation training too. Available at: http://www.ngsmedicare.com/ngs/wcm/connect/3632a905-b697-4266-8fc0-2aa2a84fedb2/1074_0514_EM_Documentation_Training_Tool_508.pdf?MOD=AJPERES. Accessed November 13, 2014.
- Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual. Chapter 12 – Physicians/nonphysician practitioners. Available at: http://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c12.pdf. Accessed November 13, 2014.
In the article “Common Coding Mistakes Hospitalists Should Avoid” in the August 2014 issue of The Hospitalist, the author states:
“For inpatient care, an established problem is defined as one in which a care plan has been generated by the physician (or same specialty group practice member) during the current hospitalization.”
This definition contradicts what I have been told in other coding courses regarding new vs. established problems relative to the examiner. It has been my understanding that when [I am] rotating on service and I have not seen that particular patient during the current admission, all of the current problems are new to me, even if previously identified by another member of my group. This [situation] results in a higher complexity of medical decision-making, which is reflective of the increased time spent learning a new patient when coming on service. I would appreciate clarification from the author.
–Matt George, MD,
medical director, MBHS Hospitalists
Billing and coding expert Carol Pohlig, BSN, RN, CPC, ACS, explains:
Be mindful when attending coding courses that are not contractor sponsored, as they may not validate the geographical interpretations of the rules for providers. There are several factors to consider when crediting the physician with “new” or “established” problems.
CMS documentation guidelines state: “Decision making with respect to a diagnosed problem is easier than that for an identified but undiagnosed problem.1
- For a presenting problem with an established diagnosis, the record should reflect whether the problem is:
- Improved, well-controlled, resolving, or resolved or
- Inadequately controlled, worsening, or failing to change as expected.
- For a presenting problem without an established diagnosis, the assessment or clinical impression may be stated in the form of differential diagnoses or as a “possible,” “probable,” or “rule out” diagnosis.
Although Medicare contractors utilize the Marshfield Clinic Scoring Tool when reviewing evaluation and management (E/M) services, a tool that historically refers to the “examiner” when considering the patient’s diagnoses, not all accept this tool-inspired standard, particularly in the advent of electronic health record accessibility and the idea that same-specialty physicians in a group practice are viewed as an individual physician.2,3
Reviewing information and familiarization of patients is often considered pre-service work and factored into the payment for E/M services. More importantly, the feasibility of an auditor being able to distinguish new vs. established problems at the level of the “examiner” is decreased when auditing a single date of service. Non-Medicare payers who audit E/M services do not necessarily follow contractor-specific guidelines but, rather, general CMS guidelines.
Therefore, without knowing the insurer or their interpretation at the time of service or visit level selection, the CMS-developed standard is the most practical application when considering the complexity of the encounter.
References
- Centers for Medicare and Medicaid Services. Department of Health and Human Services. Evaluation and management services guide. Available at: http://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/downloads//eval_mgmt_serv_guide-ICN006764.pdf. Accessed November 13, 2014.
- National Government Services. Evaluation and management documentation training too. Available at: http://www.ngsmedicare.com/ngs/wcm/connect/3632a905-b697-4266-8fc0-2aa2a84fedb2/1074_0514_EM_Documentation_Training_Tool_508.pdf?MOD=AJPERES. Accessed November 13, 2014.
- Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual. Chapter 12 – Physicians/nonphysician practitioners. Available at: http://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c12.pdf. Accessed November 13, 2014.
LISTEN NOW: Daniel Hunt, MD elaborates on recent article on primary care providers
Listen to Daniel Hunt, MD, chief of the hospital medicine unit at Massachusetts General Hospital, Boston, discuss his recent article titled “Perspectives” in the New England Journal of Medicine on consultation visits by primary care providers.
Listen to Daniel Hunt, MD, chief of the hospital medicine unit at Massachusetts General Hospital, Boston, discuss his recent article titled “Perspectives” in the New England Journal of Medicine on consultation visits by primary care providers.
Listen to Daniel Hunt, MD, chief of the hospital medicine unit at Massachusetts General Hospital, Boston, discuss his recent article titled “Perspectives” in the New England Journal of Medicine on consultation visits by primary care providers.
LISTEN NOW: Jasen Gundersen, MD, MBA, SFHM, gives advice on assessing leadership skills
Jasen Gundersen, MD, MBA, SFHM, talks about mentors and assessing your personal qualities to be a good leader.
Jasen Gundersen, MD, MBA, SFHM, talks about mentors and assessing your personal qualities to be a good leader.
Jasen Gundersen, MD, MBA, SFHM, talks about mentors and assessing your personal qualities to be a good leader.
LISTEN NOW: Bob Wachter, MD, MHM discusses the many facets of healthcare IT
Listen to more of our interview with Bob Wachter, MD, MHM, on the gains, losses, and consequences resulting from healthcare IT.
Listen to more of our interview with Bob Wachter, MD, MHM, on the gains, losses, and consequences resulting from healthcare IT.
Listen to more of our interview with Bob Wachter, MD, MHM, on the gains, losses, and consequences resulting from healthcare IT.
LISTEN NOW: Jeffrey Greenwald, MD provides tips on treating endocrine disorders
Jeffrey Greenwald, MD, a hospitalist at Massacusetts General Hospital with 15 years of experience in hospital medicine, discusses his view of the spectrum of endocrine disorders and how hospitalists should approach the question of when to call in an endocrine specialist.
Jeffrey Greenwald, MD, a hospitalist at Massacusetts General Hospital with 15 years of experience in hospital medicine, discusses his view of the spectrum of endocrine disorders and how hospitalists should approach the question of when to call in an endocrine specialist.
Jeffrey Greenwald, MD, a hospitalist at Massacusetts General Hospital with 15 years of experience in hospital medicine, discusses his view of the spectrum of endocrine disorders and how hospitalists should approach the question of when to call in an endocrine specialist.
LISTEN NOW: Amy Boutwell, MD, MPP provides tips on improving care transitions
Amy Boutwell, MD, MPP, a hospitalist and founder of Collaborative Healthcare Strategies, talks about what clinicians can do to help improve care transitions based on her experience directing IHI’s STAAR Initiative (State-Action on Avoidable Re-hospitalizations).
Amy Boutwell, MD, MPP, a hospitalist and founder of Collaborative Healthcare Strategies, talks about what clinicians can do to help improve care transitions based on her experience directing IHI’s STAAR Initiative (State-Action on Avoidable Re-hospitalizations).
Amy Boutwell, MD, MPP, a hospitalist and founder of Collaborative Healthcare Strategies, talks about what clinicians can do to help improve care transitions based on her experience directing IHI’s STAAR Initiative (State-Action on Avoidable Re-hospitalizations).
LISTEN NOW: Eric Howell, MD, SFHM discusses care transitions and readmissions
Johns Hopkins hospitalist Eric Howell, MD, SFHM, discusses connections between SHM, hospitalist practices, handoffs, and successful care transitions.
Johns Hopkins hospitalist Eric Howell, MD, SFHM, discusses connections between SHM, hospitalist practices, handoffs, and successful care transitions.
Johns Hopkins hospitalist Eric Howell, MD, SFHM, discusses connections between SHM, hospitalist practices, handoffs, and successful care transitions.
The Biggest Thing in Hospital Medicine Since Patient Safety?
Editor’s note: First of a two-part series examining bundled payments and hospital medicine. Additionally, Dr. Whitcomb works for a company that is an Awardee Convener in the CMS Bundled Payments for Care Improvement (BPCI) Initiative.
The Centers for Medicare and Medicaid Services’ (CMS) bundled payment initiative was announced in August 2011 and has been “live” since October 2013, when a handful of healthcare systems launched bundled payment programs. In 2014, the CMS initiative grew substantially as a result of large-scale interest on the part of hospitals, physician groups, skilled nursing facilities (SNFs), and others in testing the model, which can be described as a single payment for an episode of care.
The BPCI initiative will be a large-scale program by July 1; it starts with an April 1 cohort launch and will result in the program’s presence in all 50 states, with hundreds of physician practices and hospitals participating. The 2015 cohort will involve a large number of hospitalist practices, participating as “episode initiators” that bear clinical and economic responsibility for the bundle, or as “gainsharers” who are eligible to receive incentive payments if they can reduce costs while maintaining measurable quality for an episode of care.
How Does Bundled Payment Work?
The BPCI initiative is a large-scale, three- to five-year demonstration to test bundled payment in patients with fee-for-service Medicare. The most common model, referred to as Model 2, involves an inpatient hospitalization for one of 48 defined episodes, which include both medical and surgical conditions, followed by a recovery period lasting 30, 60, or 90 days.
Each hospital or physician practice that is considering entering the BPCI program receives prices for all 48 episodes based on a 2009-2012 historical average of Medicare part A and B claims associated with that hospital or physician group. After analyzing those prices, the hospital or physician practice may elect to choose the bundles that have a good chance of being successful—where actual spending comes in under the historical target price—based on care improvement expectations in their local system. In Model 2, CMS takes 2% off the target price for 90-day episodes and 3% off the target price for 30- and 60-day episodes, making it all the more important to choose bundles that demonstrate a high likelihood of success.
The revenue cycle for hospitals and physicians in the program does not change. They submit claims for their services and receive reimbursement as they always have; however, after the end of each quarter, when the majority of part A and B claims have been processed, a “look back” at actual spending for all participating episodes is reconciled against the baseline price derived from 2009-2012. If there is a net savings compared to the baseline, monies can be distributed to the participating providers—the hospital or physician practice—and those providers may further share some of the savings with other physicians/providers who have signed a gainsharing contract.
Hospitalists and BPCI
Hospitalist practices participate in the CMS program either as episode initiators or gainsharers. As episode initiators, they “own” the bundle, which means they bear economic risk for the program. In this capacity, overall savings will mean the hospitalist practice has a new revenue stream, which could be substantial; however, the practice is also responsible for any losses.
Other hospitalist practices have become gainsharers in the program, which means they have signed an agreement enabling them to receive payments in addition to professional fee revenues for activities that reduce costs while maintaining or improving quality. Such activities are referred to as “care redesign” in the program. Gainsharers do not bear financial risk.
Where Will Savings Come From?
Perhaps ironically for hospitalists, the main source of savings in the BPCI program comes from post-acute care and readmissions. For example, for common conditions like heart failure, COPD, and pneumonia, Medicare spends almost as much on post-acute care and readmissions in the first 30 days after discharge as it does on the index hospitalization.1 As a result, the BPCI program adds further emphasis on preventing readmissions when added to existing pressures, and there is a new premium placed on “right-sizing” the usage of SNF and other post-acute facilities, such as inpatient rehabilitation and long-term acute care hospitals. For hospitalists, this means that new rigor is needed to connect to the post-acute setting, such as determining why a patient is being discharged to a skilled facility.
Another savings pool, called “internal cost savings,” is available to reward decreasing inpatient utilization from, for example, testing, imaging, and implantable devices.
Conclusion
Bundled payment might be the biggest thing to come along for hospitalists since the patient safety movement launched some 16 years ago. Why? Although accountable care organizations have largely focused on ambulatory practice, bundled payment has a major focus on hospital care and on the post-acute care decisions that are made during the hospitalization. If bundled payment proves to be an effective way to pay for—and organize—care, hospitalists will play a central role in the success of this innovation.
In part two of this series, I will explore specific roles hospitalists play in successful bundled payment programs.

Reference
Editor’s note: First of a two-part series examining bundled payments and hospital medicine. Additionally, Dr. Whitcomb works for a company that is an Awardee Convener in the CMS Bundled Payments for Care Improvement (BPCI) Initiative.
The Centers for Medicare and Medicaid Services’ (CMS) bundled payment initiative was announced in August 2011 and has been “live” since October 2013, when a handful of healthcare systems launched bundled payment programs. In 2014, the CMS initiative grew substantially as a result of large-scale interest on the part of hospitals, physician groups, skilled nursing facilities (SNFs), and others in testing the model, which can be described as a single payment for an episode of care.
The BPCI initiative will be a large-scale program by July 1; it starts with an April 1 cohort launch and will result in the program’s presence in all 50 states, with hundreds of physician practices and hospitals participating. The 2015 cohort will involve a large number of hospitalist practices, participating as “episode initiators” that bear clinical and economic responsibility for the bundle, or as “gainsharers” who are eligible to receive incentive payments if they can reduce costs while maintaining measurable quality for an episode of care.
How Does Bundled Payment Work?
The BPCI initiative is a large-scale, three- to five-year demonstration to test bundled payment in patients with fee-for-service Medicare. The most common model, referred to as Model 2, involves an inpatient hospitalization for one of 48 defined episodes, which include both medical and surgical conditions, followed by a recovery period lasting 30, 60, or 90 days.
Each hospital or physician practice that is considering entering the BPCI program receives prices for all 48 episodes based on a 2009-2012 historical average of Medicare part A and B claims associated with that hospital or physician group. After analyzing those prices, the hospital or physician practice may elect to choose the bundles that have a good chance of being successful—where actual spending comes in under the historical target price—based on care improvement expectations in their local system. In Model 2, CMS takes 2% off the target price for 90-day episodes and 3% off the target price for 30- and 60-day episodes, making it all the more important to choose bundles that demonstrate a high likelihood of success.
The revenue cycle for hospitals and physicians in the program does not change. They submit claims for their services and receive reimbursement as they always have; however, after the end of each quarter, when the majority of part A and B claims have been processed, a “look back” at actual spending for all participating episodes is reconciled against the baseline price derived from 2009-2012. If there is a net savings compared to the baseline, monies can be distributed to the participating providers—the hospital or physician practice—and those providers may further share some of the savings with other physicians/providers who have signed a gainsharing contract.
Hospitalists and BPCI
Hospitalist practices participate in the CMS program either as episode initiators or gainsharers. As episode initiators, they “own” the bundle, which means they bear economic risk for the program. In this capacity, overall savings will mean the hospitalist practice has a new revenue stream, which could be substantial; however, the practice is also responsible for any losses.
Other hospitalist practices have become gainsharers in the program, which means they have signed an agreement enabling them to receive payments in addition to professional fee revenues for activities that reduce costs while maintaining or improving quality. Such activities are referred to as “care redesign” in the program. Gainsharers do not bear financial risk.
Where Will Savings Come From?
Perhaps ironically for hospitalists, the main source of savings in the BPCI program comes from post-acute care and readmissions. For example, for common conditions like heart failure, COPD, and pneumonia, Medicare spends almost as much on post-acute care and readmissions in the first 30 days after discharge as it does on the index hospitalization.1 As a result, the BPCI program adds further emphasis on preventing readmissions when added to existing pressures, and there is a new premium placed on “right-sizing” the usage of SNF and other post-acute facilities, such as inpatient rehabilitation and long-term acute care hospitals. For hospitalists, this means that new rigor is needed to connect to the post-acute setting, such as determining why a patient is being discharged to a skilled facility.
Another savings pool, called “internal cost savings,” is available to reward decreasing inpatient utilization from, for example, testing, imaging, and implantable devices.
Conclusion
Bundled payment might be the biggest thing to come along for hospitalists since the patient safety movement launched some 16 years ago. Why? Although accountable care organizations have largely focused on ambulatory practice, bundled payment has a major focus on hospital care and on the post-acute care decisions that are made during the hospitalization. If bundled payment proves to be an effective way to pay for—and organize—care, hospitalists will play a central role in the success of this innovation.
In part two of this series, I will explore specific roles hospitalists play in successful bundled payment programs.

Reference
Editor’s note: First of a two-part series examining bundled payments and hospital medicine. Additionally, Dr. Whitcomb works for a company that is an Awardee Convener in the CMS Bundled Payments for Care Improvement (BPCI) Initiative.
The Centers for Medicare and Medicaid Services’ (CMS) bundled payment initiative was announced in August 2011 and has been “live” since October 2013, when a handful of healthcare systems launched bundled payment programs. In 2014, the CMS initiative grew substantially as a result of large-scale interest on the part of hospitals, physician groups, skilled nursing facilities (SNFs), and others in testing the model, which can be described as a single payment for an episode of care.
The BPCI initiative will be a large-scale program by July 1; it starts with an April 1 cohort launch and will result in the program’s presence in all 50 states, with hundreds of physician practices and hospitals participating. The 2015 cohort will involve a large number of hospitalist practices, participating as “episode initiators” that bear clinical and economic responsibility for the bundle, or as “gainsharers” who are eligible to receive incentive payments if they can reduce costs while maintaining measurable quality for an episode of care.
How Does Bundled Payment Work?
The BPCI initiative is a large-scale, three- to five-year demonstration to test bundled payment in patients with fee-for-service Medicare. The most common model, referred to as Model 2, involves an inpatient hospitalization for one of 48 defined episodes, which include both medical and surgical conditions, followed by a recovery period lasting 30, 60, or 90 days.
Each hospital or physician practice that is considering entering the BPCI program receives prices for all 48 episodes based on a 2009-2012 historical average of Medicare part A and B claims associated with that hospital or physician group. After analyzing those prices, the hospital or physician practice may elect to choose the bundles that have a good chance of being successful—where actual spending comes in under the historical target price—based on care improvement expectations in their local system. In Model 2, CMS takes 2% off the target price for 90-day episodes and 3% off the target price for 30- and 60-day episodes, making it all the more important to choose bundles that demonstrate a high likelihood of success.
The revenue cycle for hospitals and physicians in the program does not change. They submit claims for their services and receive reimbursement as they always have; however, after the end of each quarter, when the majority of part A and B claims have been processed, a “look back” at actual spending for all participating episodes is reconciled against the baseline price derived from 2009-2012. If there is a net savings compared to the baseline, monies can be distributed to the participating providers—the hospital or physician practice—and those providers may further share some of the savings with other physicians/providers who have signed a gainsharing contract.
Hospitalists and BPCI
Hospitalist practices participate in the CMS program either as episode initiators or gainsharers. As episode initiators, they “own” the bundle, which means they bear economic risk for the program. In this capacity, overall savings will mean the hospitalist practice has a new revenue stream, which could be substantial; however, the practice is also responsible for any losses.
Other hospitalist practices have become gainsharers in the program, which means they have signed an agreement enabling them to receive payments in addition to professional fee revenues for activities that reduce costs while maintaining or improving quality. Such activities are referred to as “care redesign” in the program. Gainsharers do not bear financial risk.
Where Will Savings Come From?
Perhaps ironically for hospitalists, the main source of savings in the BPCI program comes from post-acute care and readmissions. For example, for common conditions like heart failure, COPD, and pneumonia, Medicare spends almost as much on post-acute care and readmissions in the first 30 days after discharge as it does on the index hospitalization.1 As a result, the BPCI program adds further emphasis on preventing readmissions when added to existing pressures, and there is a new premium placed on “right-sizing” the usage of SNF and other post-acute facilities, such as inpatient rehabilitation and long-term acute care hospitals. For hospitalists, this means that new rigor is needed to connect to the post-acute setting, such as determining why a patient is being discharged to a skilled facility.
Another savings pool, called “internal cost savings,” is available to reward decreasing inpatient utilization from, for example, testing, imaging, and implantable devices.
Conclusion
Bundled payment might be the biggest thing to come along for hospitalists since the patient safety movement launched some 16 years ago. Why? Although accountable care organizations have largely focused on ambulatory practice, bundled payment has a major focus on hospital care and on the post-acute care decisions that are made during the hospitalization. If bundled payment proves to be an effective way to pay for—and organize—care, hospitalists will play a central role in the success of this innovation.
In part two of this series, I will explore specific roles hospitalists play in successful bundled payment programs.