User login
June 2015 Quiz 1
Q1: ANSWER: D
Critique
The patient has had complicated recurrent Crohn’s disease and two resections. If this were his first resection of a short-segment ileal stricture, then choice (a) would be a reasonable alternative to starting medication at this time. This is based on the endoscopic scoring system of Rutgeerts in which endoscopic findings in the neoterminal ileum at 6-12 months postoperatively are somewhat predictive of clinical recurrence over the next 5 years. Short-term antibiotic therapy with ciprofloxacin has not been shown to be a good long-term solution to prevention of postoperative recurrence. Both azathioprine and 6-mercaptopurine have a modest effect on the prevention of postoperative recurrence of Crohn’s disease. Azathioprine at 1 mg/kg per day would be less than an optimal dose for this purpose. The D’Haens study used 1.5-2 mg/kg of azathioprine in combination with 3 months of metronidazole. At 1 year the endoscopic recurrence rate (i2-i4 lesions) was greater in the placebo group at 69% compared with 44% in the azathioprine-treated group. Infliximab has been shown in a small randomized trial to be very effective in preventing postoperative recurrence of Crohn’s disease. In a 24-subject randomized trial, 91% of the infliximab treated patients were free of endoscopic recurrence, compared to 9% of patients receiving placebo. In this patient, who is at a high risk for recurrence with recurrent inflammation, multiple surgeries, and continued smoking, anti-TNF therapy should be initiated within 4 weeks after surgery. Mesalamine has minimal postoperative preventative effects and would not be appropriate monotherapy to use in this high-risk patient.
References
1. D’Haens, G.R., Vermeire, S., Van Assche G., et al. Therapy of metronidazole with azathioprine to prevent postoperative recurrence of Crohn’s disease: a controlled randomized trial. Gastroenterology 2008;135:1123-9.
2. Regueiro, M., Schraut, W., Baidoo, L., et al. Infliximab prevents Crohn’s disease recurrence after ileal resection. Gastroenterology 2009;136:441-50.e1;quiz 716. Epub 2008 Oct 31.
3. Rutgeerts P, Geboes K, Vantrappen G, Beyls J, Kerremans R, Hiele M. Predictability of the postoperative course of Crohn’s disease. Gastroenterology 1990;99:956-63.
4. Schwartz M, Regueiro M. Prevention and treatment of postoperative Crohn’s disease recurrence: an update for a new decade. Curr. Gastroenterol. Rep. 2011;13:95-100.
Q1: ANSWER: D
Critique
The patient has had complicated recurrent Crohn’s disease and two resections. If this were his first resection of a short-segment ileal stricture, then choice (a) would be a reasonable alternative to starting medication at this time. This is based on the endoscopic scoring system of Rutgeerts in which endoscopic findings in the neoterminal ileum at 6-12 months postoperatively are somewhat predictive of clinical recurrence over the next 5 years. Short-term antibiotic therapy with ciprofloxacin has not been shown to be a good long-term solution to prevention of postoperative recurrence. Both azathioprine and 6-mercaptopurine have a modest effect on the prevention of postoperative recurrence of Crohn’s disease. Azathioprine at 1 mg/kg per day would be less than an optimal dose for this purpose. The D’Haens study used 1.5-2 mg/kg of azathioprine in combination with 3 months of metronidazole. At 1 year the endoscopic recurrence rate (i2-i4 lesions) was greater in the placebo group at 69% compared with 44% in the azathioprine-treated group. Infliximab has been shown in a small randomized trial to be very effective in preventing postoperative recurrence of Crohn’s disease. In a 24-subject randomized trial, 91% of the infliximab treated patients were free of endoscopic recurrence, compared to 9% of patients receiving placebo. In this patient, who is at a high risk for recurrence with recurrent inflammation, multiple surgeries, and continued smoking, anti-TNF therapy should be initiated within 4 weeks after surgery. Mesalamine has minimal postoperative preventative effects and would not be appropriate monotherapy to use in this high-risk patient.
References
1. D’Haens, G.R., Vermeire, S., Van Assche G., et al. Therapy of metronidazole with azathioprine to prevent postoperative recurrence of Crohn’s disease: a controlled randomized trial. Gastroenterology 2008;135:1123-9.
2. Regueiro, M., Schraut, W., Baidoo, L., et al. Infliximab prevents Crohn’s disease recurrence after ileal resection. Gastroenterology 2009;136:441-50.e1;quiz 716. Epub 2008 Oct 31.
3. Rutgeerts P, Geboes K, Vantrappen G, Beyls J, Kerremans R, Hiele M. Predictability of the postoperative course of Crohn’s disease. Gastroenterology 1990;99:956-63.
4. Schwartz M, Regueiro M. Prevention and treatment of postoperative Crohn’s disease recurrence: an update for a new decade. Curr. Gastroenterol. Rep. 2011;13:95-100.
Q1: ANSWER: D
Critique
The patient has had complicated recurrent Crohn’s disease and two resections. If this were his first resection of a short-segment ileal stricture, then choice (a) would be a reasonable alternative to starting medication at this time. This is based on the endoscopic scoring system of Rutgeerts in which endoscopic findings in the neoterminal ileum at 6-12 months postoperatively are somewhat predictive of clinical recurrence over the next 5 years. Short-term antibiotic therapy with ciprofloxacin has not been shown to be a good long-term solution to prevention of postoperative recurrence. Both azathioprine and 6-mercaptopurine have a modest effect on the prevention of postoperative recurrence of Crohn’s disease. Azathioprine at 1 mg/kg per day would be less than an optimal dose for this purpose. The D’Haens study used 1.5-2 mg/kg of azathioprine in combination with 3 months of metronidazole. At 1 year the endoscopic recurrence rate (i2-i4 lesions) was greater in the placebo group at 69% compared with 44% in the azathioprine-treated group. Infliximab has been shown in a small randomized trial to be very effective in preventing postoperative recurrence of Crohn’s disease. In a 24-subject randomized trial, 91% of the infliximab treated patients were free of endoscopic recurrence, compared to 9% of patients receiving placebo. In this patient, who is at a high risk for recurrence with recurrent inflammation, multiple surgeries, and continued smoking, anti-TNF therapy should be initiated within 4 weeks after surgery. Mesalamine has minimal postoperative preventative effects and would not be appropriate monotherapy to use in this high-risk patient.
References
1. D’Haens, G.R., Vermeire, S., Van Assche G., et al. Therapy of metronidazole with azathioprine to prevent postoperative recurrence of Crohn’s disease: a controlled randomized trial. Gastroenterology 2008;135:1123-9.
2. Regueiro, M., Schraut, W., Baidoo, L., et al. Infliximab prevents Crohn’s disease recurrence after ileal resection. Gastroenterology 2009;136:441-50.e1;quiz 716. Epub 2008 Oct 31.
3. Rutgeerts P, Geboes K, Vantrappen G, Beyls J, Kerremans R, Hiele M. Predictability of the postoperative course of Crohn’s disease. Gastroenterology 1990;99:956-63.
4. Schwartz M, Regueiro M. Prevention and treatment of postoperative Crohn’s disease recurrence: an update for a new decade. Curr. Gastroenterol. Rep. 2011;13:95-100.
Nanotherapy appears effective against MM

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work. ![]()

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work. ![]()

Photo by Aaron Logan
A nanoparticle-based therapy can effectively fight multiple myeloma (MM), according to preclinical research published in Molecular Cancer Therapy.
The nanoparticles protected their therapeutic cargo from degradation in the bloodstream and enhanced drug delivery into MM cells, thereby improving survival in mouse models of MM.
The nanoparticles carried an Sn 2 lipase-labile prodrug inhibitor of MYC-MAX dimerization (MI1-PD).
Researchers designed this prodrug for use in MM because MM pathogenesis is driven by the MYC oncoprotein, its dimerization with MAX, and the binding of this heterodimer to E-Boxes in the vicinity of target genes.
Previous research has shown that MYC inhibitors are extremely potent in vitro, but, when injected into the blood, they degrade immediately. The current study is the first to show that a MYC inhibitor can be effective in animals with cancer, as long as the drug has a vehicle to protect and deliver it into cancer cells.
“The nanoparticles serve as vehicles that protect the drug from the harsh environment of the blood,” said study author Gregory M. Lanza, MD, PhD, of the Washington University School of Medicine in St Louis, Missouri.
“In this case, the drug is modified into a prodrug and actually locked into the outer membrane of the particle.”
The nanoparticles are designed to carry targeting molecules that home in on and bind to MM cells that carry the complementary receptor. When a nanoparticle binds to an MM cell, their membranes fuse together, transferring MI1-PD to the malignant cell. When safely inside, MI1-PD is enzymatically freed to do its job, blocking MYC from activation.
In theory, noncancerous cells are unlikely to be adversely affected by MYC inhibition because healthy cells shouldn’t have highly activated MYC proteins to begin with, according to the researchers.
The team injected 2 sizes of MI1-PD-containing nanoparticles into mice with MM and found that the nanoparticles increased the animals’ median survival.
The median survival was 46 days for mice that received MI1-PD-containing, 200-nm nanoparticles and 28 days for mice that received control, 200-nm nanoparticles (P<0.05). For mice that received 20 nm nanoparticles, the median survival was 52 days and 29 days, respectively (P=0.001).
The researchers also pointed out that neither MI1 nor MI1-PD conferred a survival benefit for the mice when injected without nanoparticle protection.
“We’re excited about our results because there was no guarantee the nanotherapy would increase survival,” said study author Michael H. Tomasson, MD, of the Washington University School of Medicine.
“We injected the nanoparticles intravenously, and they found the tumors throughout the body, whether they were in the bone marrow, the spleen, or elsewhere.”
Drs Tomasson and Lanza said this technology is still years away from clinical trials, but they are optimistic about its future potential and are eager to begin that work. ![]()
CTLs captured on video destroying cancer cells
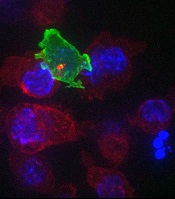
cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()
Approach could overcome drug resistance

Photo courtesy of the CDC
When treating patients with malaria and other infectious diseases, doctors should consider using drug combinations that reach similar parts of the body, according to researchers.
The group found that imperfect drug penetration—when drugs don’t reach all parts of the body—accelerates treatment resistance.
When there is a “pocket” of the body where only one drug is present, such as the brain or the digestive system, a pathogen can quickly develop resistance to one drug at a time.
“If there is a space where there is only one drug, that’s the place where the pathogen can start its escape,” said Pleuni Pennings, PhD, of San Francisco State University in California.
“Once it no longer has the first drug to deal with, it’s very easy for it to quickly become resistant to a second drug.”
Dr Pennings and her colleagues reported these findings in PNAS.
The team believes this research could have major implications for how treatment plans are designed and prescribed to patients with malaria, HIV, tuberculosis, and other ailments.
Because pathogens can quickly develop resistance to a single drug, providers often prescribe multiple drugs to increase their effectiveness.
The results of the study suggest that, when doing so, doctors should carefully consider which parts of the body each drug will reach and whether selecting medications with imperfect but similar penetrations might be the best treatment option.
“It may be better, in some cases, to leave a pocket of the body without any drugs instead of leaving a pocket with just one drug,” Dr Pennings said.
The study is the first to look at the connection between drug penetration and multidrug resistance. Dr Pennings and her colleagues ran computer simulations to observe the behavior of pathogens in response to changes in drugs and their levels of penetration.
The team found that, in instances where even small parts of the body could only be reached by one drug, the pathogen’s ability to build resistance to both drugs was accelerated compared to situations where no such pockets existed.
“This requires a new way of thinking about drug combinations that is a bit counterintuitive,” Dr Pennings said. “Suppose that drug A does not reach the brain, but drug B does. You’ll see the pathogen evolving resistance to drug B and assume that’s where the problem lies. But, in fact, it is drug A that is not doing its job because it’s not reaching the brain, and that’s the drug you may have to actually fix.”
With future research, Dr Pennings and her colleagues hope to outline the most effective drug combinations by exploring which parts of the body cannot be reached by specific drugs and where and how quickly specific pathogens are able to develop resistance. ![]()

Photo courtesy of the CDC
When treating patients with malaria and other infectious diseases, doctors should consider using drug combinations that reach similar parts of the body, according to researchers.
The group found that imperfect drug penetration—when drugs don’t reach all parts of the body—accelerates treatment resistance.
When there is a “pocket” of the body where only one drug is present, such as the brain or the digestive system, a pathogen can quickly develop resistance to one drug at a time.
“If there is a space where there is only one drug, that’s the place where the pathogen can start its escape,” said Pleuni Pennings, PhD, of San Francisco State University in California.
“Once it no longer has the first drug to deal with, it’s very easy for it to quickly become resistant to a second drug.”
Dr Pennings and her colleagues reported these findings in PNAS.
The team believes this research could have major implications for how treatment plans are designed and prescribed to patients with malaria, HIV, tuberculosis, and other ailments.
Because pathogens can quickly develop resistance to a single drug, providers often prescribe multiple drugs to increase their effectiveness.
The results of the study suggest that, when doing so, doctors should carefully consider which parts of the body each drug will reach and whether selecting medications with imperfect but similar penetrations might be the best treatment option.
“It may be better, in some cases, to leave a pocket of the body without any drugs instead of leaving a pocket with just one drug,” Dr Pennings said.
The study is the first to look at the connection between drug penetration and multidrug resistance. Dr Pennings and her colleagues ran computer simulations to observe the behavior of pathogens in response to changes in drugs and their levels of penetration.
The team found that, in instances where even small parts of the body could only be reached by one drug, the pathogen’s ability to build resistance to both drugs was accelerated compared to situations where no such pockets existed.
“This requires a new way of thinking about drug combinations that is a bit counterintuitive,” Dr Pennings said. “Suppose that drug A does not reach the brain, but drug B does. You’ll see the pathogen evolving resistance to drug B and assume that’s where the problem lies. But, in fact, it is drug A that is not doing its job because it’s not reaching the brain, and that’s the drug you may have to actually fix.”
With future research, Dr Pennings and her colleagues hope to outline the most effective drug combinations by exploring which parts of the body cannot be reached by specific drugs and where and how quickly specific pathogens are able to develop resistance. ![]()

Photo courtesy of the CDC
When treating patients with malaria and other infectious diseases, doctors should consider using drug combinations that reach similar parts of the body, according to researchers.
The group found that imperfect drug penetration—when drugs don’t reach all parts of the body—accelerates treatment resistance.
When there is a “pocket” of the body where only one drug is present, such as the brain or the digestive system, a pathogen can quickly develop resistance to one drug at a time.
“If there is a space where there is only one drug, that’s the place where the pathogen can start its escape,” said Pleuni Pennings, PhD, of San Francisco State University in California.
“Once it no longer has the first drug to deal with, it’s very easy for it to quickly become resistant to a second drug.”
Dr Pennings and her colleagues reported these findings in PNAS.
The team believes this research could have major implications for how treatment plans are designed and prescribed to patients with malaria, HIV, tuberculosis, and other ailments.
Because pathogens can quickly develop resistance to a single drug, providers often prescribe multiple drugs to increase their effectiveness.
The results of the study suggest that, when doing so, doctors should carefully consider which parts of the body each drug will reach and whether selecting medications with imperfect but similar penetrations might be the best treatment option.
“It may be better, in some cases, to leave a pocket of the body without any drugs instead of leaving a pocket with just one drug,” Dr Pennings said.
The study is the first to look at the connection between drug penetration and multidrug resistance. Dr Pennings and her colleagues ran computer simulations to observe the behavior of pathogens in response to changes in drugs and their levels of penetration.
The team found that, in instances where even small parts of the body could only be reached by one drug, the pathogen’s ability to build resistance to both drugs was accelerated compared to situations where no such pockets existed.
“This requires a new way of thinking about drug combinations that is a bit counterintuitive,” Dr Pennings said. “Suppose that drug A does not reach the brain, but drug B does. You’ll see the pathogen evolving resistance to drug B and assume that’s where the problem lies. But, in fact, it is drug A that is not doing its job because it’s not reaching the brain, and that’s the drug you may have to actually fix.”
With future research, Dr Pennings and her colleagues hope to outline the most effective drug combinations by exploring which parts of the body cannot be reached by specific drugs and where and how quickly specific pathogens are able to develop resistance. ![]()
Readmissions for severe sepsis fairly common

Photo courtesy of the CDC
DENVER—Severe sepsis is a significant cause of rehospitalization, along the lines of more commonly discussed conditions such as heart failure and pneumonia, according to research presented at the 2015 American Thoracic Society International Conference.
“Severe sepsis continues to be a common cause of hospitalization and has the associated high costs,” said Darya Rudym, MD, of New York University School of Medicine in New York.
She presented this finding at the meeting as publication number A3699.
Some quality measures associated with sepsis, such as length of hospital stay, have been studied previously. But Dr Rudym and her colleagues wanted to look at the rate of readmission within 30 days after discharge among patients with a diagnosis of severe sepsis.
The researchers also wanted to compare this rate to the readmission rates for outcomes reported by the Centers for Medicare and Medicaid Services, such as acute myocardial infarction, heart failure, and pneumonia.
The team examined inpatient discharges from Bellevue Hospital in New York City between July 2011 and July 2014 and identified subsequent readmissions to the hospital within 30 days.
During the 3-year study period, there were 22,712 discharges and an overall readmission rate of 15.31%, or 3477 patients.
The researchers used 3 different methods to identify patients with severe sepsis: Angus implementation, Martin implementation, and explicit ICD-9-CM sepsis codes.
There were 1801 cases of severe sepsis according to Angus implementation, 798 cases according to Martin implementation, and 579 cases according to ICD-9-CM sepsis codes.
In all, 266, 119, and 71 of these patients, respectively, were readmitted to the hospital within 30 days. This accounted for 14.77%, 14.91%, and 12.26% of cases, respectively.
The readmission rate for myocardial infarction was 8.67%. It was 14.46% for pneumonia, and 15% for heart failure.
“Readmission rates in severe sepsis are shown not to be significantly different from readmission rates in heart failure and pneumonia,” Dr Rudym noted. ![]()

Photo courtesy of the CDC
DENVER—Severe sepsis is a significant cause of rehospitalization, along the lines of more commonly discussed conditions such as heart failure and pneumonia, according to research presented at the 2015 American Thoracic Society International Conference.
“Severe sepsis continues to be a common cause of hospitalization and has the associated high costs,” said Darya Rudym, MD, of New York University School of Medicine in New York.
She presented this finding at the meeting as publication number A3699.
Some quality measures associated with sepsis, such as length of hospital stay, have been studied previously. But Dr Rudym and her colleagues wanted to look at the rate of readmission within 30 days after discharge among patients with a diagnosis of severe sepsis.
The researchers also wanted to compare this rate to the readmission rates for outcomes reported by the Centers for Medicare and Medicaid Services, such as acute myocardial infarction, heart failure, and pneumonia.
The team examined inpatient discharges from Bellevue Hospital in New York City between July 2011 and July 2014 and identified subsequent readmissions to the hospital within 30 days.
During the 3-year study period, there were 22,712 discharges and an overall readmission rate of 15.31%, or 3477 patients.
The researchers used 3 different methods to identify patients with severe sepsis: Angus implementation, Martin implementation, and explicit ICD-9-CM sepsis codes.
There were 1801 cases of severe sepsis according to Angus implementation, 798 cases according to Martin implementation, and 579 cases according to ICD-9-CM sepsis codes.
In all, 266, 119, and 71 of these patients, respectively, were readmitted to the hospital within 30 days. This accounted for 14.77%, 14.91%, and 12.26% of cases, respectively.
The readmission rate for myocardial infarction was 8.67%. It was 14.46% for pneumonia, and 15% for heart failure.
“Readmission rates in severe sepsis are shown not to be significantly different from readmission rates in heart failure and pneumonia,” Dr Rudym noted. ![]()

Photo courtesy of the CDC
DENVER—Severe sepsis is a significant cause of rehospitalization, along the lines of more commonly discussed conditions such as heart failure and pneumonia, according to research presented at the 2015 American Thoracic Society International Conference.
“Severe sepsis continues to be a common cause of hospitalization and has the associated high costs,” said Darya Rudym, MD, of New York University School of Medicine in New York.
She presented this finding at the meeting as publication number A3699.
Some quality measures associated with sepsis, such as length of hospital stay, have been studied previously. But Dr Rudym and her colleagues wanted to look at the rate of readmission within 30 days after discharge among patients with a diagnosis of severe sepsis.
The researchers also wanted to compare this rate to the readmission rates for outcomes reported by the Centers for Medicare and Medicaid Services, such as acute myocardial infarction, heart failure, and pneumonia.
The team examined inpatient discharges from Bellevue Hospital in New York City between July 2011 and July 2014 and identified subsequent readmissions to the hospital within 30 days.
During the 3-year study period, there were 22,712 discharges and an overall readmission rate of 15.31%, or 3477 patients.
The researchers used 3 different methods to identify patients with severe sepsis: Angus implementation, Martin implementation, and explicit ICD-9-CM sepsis codes.
There were 1801 cases of severe sepsis according to Angus implementation, 798 cases according to Martin implementation, and 579 cases according to ICD-9-CM sepsis codes.
In all, 266, 119, and 71 of these patients, respectively, were readmitted to the hospital within 30 days. This accounted for 14.77%, 14.91%, and 12.26% of cases, respectively.
The readmission rate for myocardial infarction was 8.67%. It was 14.46% for pneumonia, and 15% for heart failure.
“Readmission rates in severe sepsis are shown not to be significantly different from readmission rates in heart failure and pneumonia,” Dr Rudym noted. ![]()
Newest SHM Board Member Eager to Make Positive Impact
Christopher Frost, MD, FHM, has accomplished a lot in his career. He retired as a major from the U.S. Air Force Reserve. He's been an ED physician. And now he is the chief medical officer of hospital-based physicians for Hospital Corporation of America.
Yet, he considers being the newest member of SHM's Board of Directors a distinct honor.
"I am truly humbled to be participating with this group of energetic, intelligent, and wise individuals that comprise the SHM board," Dr. Frost says in an e-mail interview with The Hospitalist. "[I'm] very eager to participate with this group, and ultimately, I would like for us, not me, to be able to say we had some positive impact on the specialty, whether that be for our providers, patients, families, or some combination thereof."
Question: What drew you to hospital medicine?
Answer: During my chief [residency] year, I started moonlighting as a hospitalist for a large hematology/oncology group. I enjoyed the experience but did not really consider hospital medicine as a career option until I received a call asking if I knew of any residents who might be interested in joining a hospital medicine group undergoing rapid growth. Up until that moment, hospital medicine was just a moonlighting gig. It was still a relatively nascent specialty, and I did not realize it could be a career path. The call served as a catalyst to rethink my options.
Q: What keeps you engaged in the specialty?
A: The specialty attracts change agents. Individuals that are not necessarily satisfied with the status quo but instead are interested in collaborating with others to affect change. From a local level, in our community hospitals where hospitalists chair committees or serve as chiefs of staff, all the way to the national level where hospitalists are serving in such roles as the CMO of the Centers for Medicare & Medicaid Services or as the U.S. Surgeon General.
Q: As a new board member, what do you now realize that you didn’t before?
A: One is what it means to be a part of a “big tent” organization, and the other is an appreciation of the breadth and depth of the talent found among the SHM staff. I knew that SHM was involved in a lot of different activities and that several other specialty societies and organizations seek out SHM based on the collaborative nature of our specialty society; however, I didn't appreciate just how broad the scope of involvement extends. TH
Visit our website for more information on leadership.
Christopher Frost, MD, FHM, has accomplished a lot in his career. He retired as a major from the U.S. Air Force Reserve. He's been an ED physician. And now he is the chief medical officer of hospital-based physicians for Hospital Corporation of America.
Yet, he considers being the newest member of SHM's Board of Directors a distinct honor.
"I am truly humbled to be participating with this group of energetic, intelligent, and wise individuals that comprise the SHM board," Dr. Frost says in an e-mail interview with The Hospitalist. "[I'm] very eager to participate with this group, and ultimately, I would like for us, not me, to be able to say we had some positive impact on the specialty, whether that be for our providers, patients, families, or some combination thereof."
Question: What drew you to hospital medicine?
Answer: During my chief [residency] year, I started moonlighting as a hospitalist for a large hematology/oncology group. I enjoyed the experience but did not really consider hospital medicine as a career option until I received a call asking if I knew of any residents who might be interested in joining a hospital medicine group undergoing rapid growth. Up until that moment, hospital medicine was just a moonlighting gig. It was still a relatively nascent specialty, and I did not realize it could be a career path. The call served as a catalyst to rethink my options.
Q: What keeps you engaged in the specialty?
A: The specialty attracts change agents. Individuals that are not necessarily satisfied with the status quo but instead are interested in collaborating with others to affect change. From a local level, in our community hospitals where hospitalists chair committees or serve as chiefs of staff, all the way to the national level where hospitalists are serving in such roles as the CMO of the Centers for Medicare & Medicaid Services or as the U.S. Surgeon General.
Q: As a new board member, what do you now realize that you didn’t before?
A: One is what it means to be a part of a “big tent” organization, and the other is an appreciation of the breadth and depth of the talent found among the SHM staff. I knew that SHM was involved in a lot of different activities and that several other specialty societies and organizations seek out SHM based on the collaborative nature of our specialty society; however, I didn't appreciate just how broad the scope of involvement extends. TH
Visit our website for more information on leadership.
Christopher Frost, MD, FHM, has accomplished a lot in his career. He retired as a major from the U.S. Air Force Reserve. He's been an ED physician. And now he is the chief medical officer of hospital-based physicians for Hospital Corporation of America.
Yet, he considers being the newest member of SHM's Board of Directors a distinct honor.
"I am truly humbled to be participating with this group of energetic, intelligent, and wise individuals that comprise the SHM board," Dr. Frost says in an e-mail interview with The Hospitalist. "[I'm] very eager to participate with this group, and ultimately, I would like for us, not me, to be able to say we had some positive impact on the specialty, whether that be for our providers, patients, families, or some combination thereof."
Question: What drew you to hospital medicine?
Answer: During my chief [residency] year, I started moonlighting as a hospitalist for a large hematology/oncology group. I enjoyed the experience but did not really consider hospital medicine as a career option until I received a call asking if I knew of any residents who might be interested in joining a hospital medicine group undergoing rapid growth. Up until that moment, hospital medicine was just a moonlighting gig. It was still a relatively nascent specialty, and I did not realize it could be a career path. The call served as a catalyst to rethink my options.
Q: What keeps you engaged in the specialty?
A: The specialty attracts change agents. Individuals that are not necessarily satisfied with the status quo but instead are interested in collaborating with others to affect change. From a local level, in our community hospitals where hospitalists chair committees or serve as chiefs of staff, all the way to the national level where hospitalists are serving in such roles as the CMO of the Centers for Medicare & Medicaid Services or as the U.S. Surgeon General.
Q: As a new board member, what do you now realize that you didn’t before?
A: One is what it means to be a part of a “big tent” organization, and the other is an appreciation of the breadth and depth of the talent found among the SHM staff. I knew that SHM was involved in a lot of different activities and that several other specialty societies and organizations seek out SHM based on the collaborative nature of our specialty society; however, I didn't appreciate just how broad the scope of involvement extends. TH
Visit our website for more information on leadership.
Antibiotic Regimens Compared for Treating Uncomplicated Skin Infections
Clinical question: What are the efficacy and safety of clindamycin and trimethoprim-sulfamethoxazole (TMP-SMX) for treatment of uncomplicated soft tissue infections in adults and children?
Background: Clindamycin and TMP-SMX are commonly used treatments for uncomplicated skin infections in adults and children, but no head-to-head comparison of efficacy or side effect profile for these medications exists.
Study design: Multi-center, prospective, double-blind, randomized clinical trial of superiority.
Setting: Four academic medical centers.
Synopsis: A group of 524 adults (n=369) and children with uncomplicated cellulitis, abscess, or both was enrolled. For patients with abscess, only those with larger lesions (based on age) were included. All abscesses were incised and drained, and patients were randomly treated with either clindamycin or TMP-SMX for 10 days. A total of 41% of cultures from suppurative wounds grew Staphylococcus aureus; 77% of these were methicillin-resistant.
The primary outcome was clinical cure assessed at 7 to 10 days and one month after completing treatment. No significant difference in cure or reported side effects was seen between drug treatment groups, age groups, lesion types, or isolates cultured. Some 80.3% of patients in the clindamycin group and 77.3% of patients in the TMP-SMX group were cured. Side effect profiles assessed by patient questionnaires showed similar rates of self-limited gastrointestinal (~19%) and dermatologic (~1%) complaints. No cases of Clostridium difficile–associated diarrhea were found.
Limitations include exclusion of patients with significant comorbidities and hospitalized patients. Also, other antibiotic regimens were not compared. Patients were followed for only one month to assess recurrence. Finally, no attempt was made to optimize antibiotic dose.
Bottom line: Clindamycin and TMP-SMX had similar cure rates and side effect profiles in otherwise healthy patients with uncomplicated skin infections.
Citation: Miller LG, Daum RS, Creech CB, et al. Clindamycin versus trimethoprim-sulfamethoxazole for uncomplicated skin infections. N Engl J Med. 2015;372:1093–1103.
Visit our website for more hospitalist-focused literature reviews.
Clinical question: What are the efficacy and safety of clindamycin and trimethoprim-sulfamethoxazole (TMP-SMX) for treatment of uncomplicated soft tissue infections in adults and children?
Background: Clindamycin and TMP-SMX are commonly used treatments for uncomplicated skin infections in adults and children, but no head-to-head comparison of efficacy or side effect profile for these medications exists.
Study design: Multi-center, prospective, double-blind, randomized clinical trial of superiority.
Setting: Four academic medical centers.
Synopsis: A group of 524 adults (n=369) and children with uncomplicated cellulitis, abscess, or both was enrolled. For patients with abscess, only those with larger lesions (based on age) were included. All abscesses were incised and drained, and patients were randomly treated with either clindamycin or TMP-SMX for 10 days. A total of 41% of cultures from suppurative wounds grew Staphylococcus aureus; 77% of these were methicillin-resistant.
The primary outcome was clinical cure assessed at 7 to 10 days and one month after completing treatment. No significant difference in cure or reported side effects was seen between drug treatment groups, age groups, lesion types, or isolates cultured. Some 80.3% of patients in the clindamycin group and 77.3% of patients in the TMP-SMX group were cured. Side effect profiles assessed by patient questionnaires showed similar rates of self-limited gastrointestinal (~19%) and dermatologic (~1%) complaints. No cases of Clostridium difficile–associated diarrhea were found.
Limitations include exclusion of patients with significant comorbidities and hospitalized patients. Also, other antibiotic regimens were not compared. Patients were followed for only one month to assess recurrence. Finally, no attempt was made to optimize antibiotic dose.
Bottom line: Clindamycin and TMP-SMX had similar cure rates and side effect profiles in otherwise healthy patients with uncomplicated skin infections.
Citation: Miller LG, Daum RS, Creech CB, et al. Clindamycin versus trimethoprim-sulfamethoxazole for uncomplicated skin infections. N Engl J Med. 2015;372:1093–1103.
Visit our website for more hospitalist-focused literature reviews.
Clinical question: What are the efficacy and safety of clindamycin and trimethoprim-sulfamethoxazole (TMP-SMX) for treatment of uncomplicated soft tissue infections in adults and children?
Background: Clindamycin and TMP-SMX are commonly used treatments for uncomplicated skin infections in adults and children, but no head-to-head comparison of efficacy or side effect profile for these medications exists.
Study design: Multi-center, prospective, double-blind, randomized clinical trial of superiority.
Setting: Four academic medical centers.
Synopsis: A group of 524 adults (n=369) and children with uncomplicated cellulitis, abscess, or both was enrolled. For patients with abscess, only those with larger lesions (based on age) were included. All abscesses were incised and drained, and patients were randomly treated with either clindamycin or TMP-SMX for 10 days. A total of 41% of cultures from suppurative wounds grew Staphylococcus aureus; 77% of these were methicillin-resistant.
The primary outcome was clinical cure assessed at 7 to 10 days and one month after completing treatment. No significant difference in cure or reported side effects was seen between drug treatment groups, age groups, lesion types, or isolates cultured. Some 80.3% of patients in the clindamycin group and 77.3% of patients in the TMP-SMX group were cured. Side effect profiles assessed by patient questionnaires showed similar rates of self-limited gastrointestinal (~19%) and dermatologic (~1%) complaints. No cases of Clostridium difficile–associated diarrhea were found.
Limitations include exclusion of patients with significant comorbidities and hospitalized patients. Also, other antibiotic regimens were not compared. Patients were followed for only one month to assess recurrence. Finally, no attempt was made to optimize antibiotic dose.
Bottom line: Clindamycin and TMP-SMX had similar cure rates and side effect profiles in otherwise healthy patients with uncomplicated skin infections.
Citation: Miller LG, Daum RS, Creech CB, et al. Clindamycin versus trimethoprim-sulfamethoxazole for uncomplicated skin infections. N Engl J Med. 2015;372:1093–1103.
Visit our website for more hospitalist-focused literature reviews.
Food recalls highlight risk of listeriosis
Recently, after a long day at the hospital, I stopped at the grocery store to pick up something for a quick dinner. I drifted to the frozen food case in the organic food section, but pulled up short when I saw empty shelves. A paper sign announced that Amy’s Kitchen, a manufacturer of organic and natural frozen foods, had become the latest company to recall its products because of concern about Listeria monocytogenes contamination.
According to information posted on the Food and Drug Administration website, this facultative, anaerobic gram-positive bacillus has been the impetus behind 10 national recalls of food products between April 1 and May 8, 2015 alone. Implicated food products have ranged from gourmet ice cream to soybean sprouts to frozen vegetables. Unlike some other bacterial causes of food-borne illness, Listeria organisms can thrive at cold temperatures. Historically, outbreaks of disease have been linked to a variety of foods, including raw produce, contaminated ready-to-eat foods such as deli meats and prepared salads, and unpasteurized milk and milk products.
Clinical manifestations of listeriosis range from febrile gastroenteritis to bacteremia and meningitis, with severe disease seen primarily in immunocompromised individuals and adults 65 and older.
Pregnant women are especially susceptible, with incidence rates 13 times higher than in the general population. Probably as a result of food choices, Hispanic women are disproportionately affected, with rates up to 24 times higher. Maternal infection may be asymptomatic or may manifest with flulike symptoms that include fever, myalgias, headache, and backache, with or without a preceding diarrhea illness. Even mild maternal illness may result in adverse pregnancy outcomes such as fetal loss, premature labor, and severe neonatal infection.
While medical students and residents are still taught to think of Listeria infection as one of the “big three” causes of neonatal sepsis along with group B streptococcus and Escherichia coli, many pediatricians have never seen a case of this rare, but potentially devastating disease. As with group B streptococcus, both early-onset and late-onset disease occur. Sepsis is the most common presentation of disease in the first week of life, while meningitis predominates in late-onset disease. Pneumonia and myocarditis are occasionally seen. Granulomatosis infantisepticum is an uncommon manifestation of severe, disseminated Listeria infection. Granuloma can occur in nearly every organ, although involvement of the liver and skin is most common.
In 2002, investigators from the Centers for Disease Control and Prevention and the American College of Obstetricians and Gynecologists surveyed more than 400 pregnant women from across the United States about their knowledge of the transmission, risk factors, symptoms, and prevention of listeriosis (Infect. Dis. Obstet. Gyn. 2005;13:11-15). A year later, the Minnesota Department of Health surveyed an additional 286 pregnant women from their state using the same survey instrument.
More than 80% of survey respondents had never heard of the disease, and knowledge about prevention strategies was therefore predictably limited. Only 33% of respondents in the national survey and 17% of respondents in the Minnesota survey knew, for example, that infection could be prevented by avoiding delicatessen meats and soft cheeses. Investigators concluded that “timely and appropriate education” of pregnant women about listeriosis could reduce cases of perinatal infection.
Data from the CDC suggest we have more work to do. The Listeria Initiative is an enhanced national surveillance system that collects laboratory, clinical, and food exposure data about listeriosis cases in the United States. Between 2009 and 2011, 14% of the 1,651 invasive Listeria infections reported were classified as pregnancy associated. Morbidity and mortality were significant, with 40 fetal losses and 6 neonatal deaths (MMWR 2013;62:448-52).
The CDC offers some common sense tips for preventing listeriosis and other food-borne illness. Raw fruits and vegetables should be thoroughly rinsed with tap water and dried with a clean cloth or paper towel before being eaten or cooked. Even foods that are typically peeled first should be washed, and firm produce, such as cantaloupe, should be scrubbed with a produce brush to reduce surface contamination. Uncooked meats and poultry should never come in contact with other food. Hands, knives, cutting boards, and other food preparation surfaces should be washed thoroughly after uncooked food is handled.
Pregnant women and others at increased risk for listeriosis should not eat hot dogs or deli meats unless they are cooked to steaming. Soft cheeses, including feta, brie, Camembert, queso blanco, or anything blue veined, should be avoided unless the label clearly states that the product has been made with pasteurized milk. Even then, it might not be safe. Pasteurized Mexican-style cheeses, such as queso fresco, have been linked to Listeria infections, likely as a result of contamination during the cheese-making process.
Physicians should be prepared to field calls from concerned parents who believe their children may have consumed a product potentially contaminated with Listeria. In general, someone who has eaten a recalled food product but has no symptoms doesn’t need a laboratory evaluation or treatment. Screening blood cultures is not indicated, and routine tests such as a complete blood count are unlikely to be helpful. Instead, patients should be counseled about the symptoms of listeriosis and undergo prompt evaluation if any develop within 2 months of exposure. The typical interval between exposure and the development of symptoms is 1 day to 3 weeks, but may be as long as 70 days.
Although Listeria infection may result in gastrointestinal symptoms, stool cultures are not recommended for diagnosis. According to the CDC, ingestion of food contaminated with Listeria occurs frequently because the organisms are commonly found in the environment. Although uncommon, intermittent fecal carriage and shedding have been observed in asymptomatic individuals.
Back at the grocery, I sighed and resigned myself to a grilled cheese sandwich for dinner. I turned and saw another woman in the aisle stop and read the sign on the freezer case.
“It’s a little scary,” she said with a sigh. “It seems like there is another recall every week, and I’m wondering what’s safe to eat.”
The parents of our patients may have similar questions. Although the Food and Drug Administration offers detailed guidance for food manufacturers about reducing Listeria contamination, perfect compliance wouldn’t eliminate the risk for consumers because L. monocytogenes is widespread in the environment. The organisms are found in water, soil, sewage, and decaying vegetation, and can be isolated from a variety of animals. Fresh fruits and vegetables are “healthy” choices as long as they are handled and prepared appropriately. Conversely, unpasteurized milk and milk products can never be considered safe.
That’s food for thought.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Kosair Children’s Hospital, also in Louisville. She had no relevant financial disclosures.
Recently, after a long day at the hospital, I stopped at the grocery store to pick up something for a quick dinner. I drifted to the frozen food case in the organic food section, but pulled up short when I saw empty shelves. A paper sign announced that Amy’s Kitchen, a manufacturer of organic and natural frozen foods, had become the latest company to recall its products because of concern about Listeria monocytogenes contamination.
According to information posted on the Food and Drug Administration website, this facultative, anaerobic gram-positive bacillus has been the impetus behind 10 national recalls of food products between April 1 and May 8, 2015 alone. Implicated food products have ranged from gourmet ice cream to soybean sprouts to frozen vegetables. Unlike some other bacterial causes of food-borne illness, Listeria organisms can thrive at cold temperatures. Historically, outbreaks of disease have been linked to a variety of foods, including raw produce, contaminated ready-to-eat foods such as deli meats and prepared salads, and unpasteurized milk and milk products.
Clinical manifestations of listeriosis range from febrile gastroenteritis to bacteremia and meningitis, with severe disease seen primarily in immunocompromised individuals and adults 65 and older.
Pregnant women are especially susceptible, with incidence rates 13 times higher than in the general population. Probably as a result of food choices, Hispanic women are disproportionately affected, with rates up to 24 times higher. Maternal infection may be asymptomatic or may manifest with flulike symptoms that include fever, myalgias, headache, and backache, with or without a preceding diarrhea illness. Even mild maternal illness may result in adverse pregnancy outcomes such as fetal loss, premature labor, and severe neonatal infection.
While medical students and residents are still taught to think of Listeria infection as one of the “big three” causes of neonatal sepsis along with group B streptococcus and Escherichia coli, many pediatricians have never seen a case of this rare, but potentially devastating disease. As with group B streptococcus, both early-onset and late-onset disease occur. Sepsis is the most common presentation of disease in the first week of life, while meningitis predominates in late-onset disease. Pneumonia and myocarditis are occasionally seen. Granulomatosis infantisepticum is an uncommon manifestation of severe, disseminated Listeria infection. Granuloma can occur in nearly every organ, although involvement of the liver and skin is most common.
In 2002, investigators from the Centers for Disease Control and Prevention and the American College of Obstetricians and Gynecologists surveyed more than 400 pregnant women from across the United States about their knowledge of the transmission, risk factors, symptoms, and prevention of listeriosis (Infect. Dis. Obstet. Gyn. 2005;13:11-15). A year later, the Minnesota Department of Health surveyed an additional 286 pregnant women from their state using the same survey instrument.
More than 80% of survey respondents had never heard of the disease, and knowledge about prevention strategies was therefore predictably limited. Only 33% of respondents in the national survey and 17% of respondents in the Minnesota survey knew, for example, that infection could be prevented by avoiding delicatessen meats and soft cheeses. Investigators concluded that “timely and appropriate education” of pregnant women about listeriosis could reduce cases of perinatal infection.
Data from the CDC suggest we have more work to do. The Listeria Initiative is an enhanced national surveillance system that collects laboratory, clinical, and food exposure data about listeriosis cases in the United States. Between 2009 and 2011, 14% of the 1,651 invasive Listeria infections reported were classified as pregnancy associated. Morbidity and mortality were significant, with 40 fetal losses and 6 neonatal deaths (MMWR 2013;62:448-52).
The CDC offers some common sense tips for preventing listeriosis and other food-borne illness. Raw fruits and vegetables should be thoroughly rinsed with tap water and dried with a clean cloth or paper towel before being eaten or cooked. Even foods that are typically peeled first should be washed, and firm produce, such as cantaloupe, should be scrubbed with a produce brush to reduce surface contamination. Uncooked meats and poultry should never come in contact with other food. Hands, knives, cutting boards, and other food preparation surfaces should be washed thoroughly after uncooked food is handled.
Pregnant women and others at increased risk for listeriosis should not eat hot dogs or deli meats unless they are cooked to steaming. Soft cheeses, including feta, brie, Camembert, queso blanco, or anything blue veined, should be avoided unless the label clearly states that the product has been made with pasteurized milk. Even then, it might not be safe. Pasteurized Mexican-style cheeses, such as queso fresco, have been linked to Listeria infections, likely as a result of contamination during the cheese-making process.
Physicians should be prepared to field calls from concerned parents who believe their children may have consumed a product potentially contaminated with Listeria. In general, someone who has eaten a recalled food product but has no symptoms doesn’t need a laboratory evaluation or treatment. Screening blood cultures is not indicated, and routine tests such as a complete blood count are unlikely to be helpful. Instead, patients should be counseled about the symptoms of listeriosis and undergo prompt evaluation if any develop within 2 months of exposure. The typical interval between exposure and the development of symptoms is 1 day to 3 weeks, but may be as long as 70 days.
Although Listeria infection may result in gastrointestinal symptoms, stool cultures are not recommended for diagnosis. According to the CDC, ingestion of food contaminated with Listeria occurs frequently because the organisms are commonly found in the environment. Although uncommon, intermittent fecal carriage and shedding have been observed in asymptomatic individuals.
Back at the grocery, I sighed and resigned myself to a grilled cheese sandwich for dinner. I turned and saw another woman in the aisle stop and read the sign on the freezer case.
“It’s a little scary,” she said with a sigh. “It seems like there is another recall every week, and I’m wondering what’s safe to eat.”
The parents of our patients may have similar questions. Although the Food and Drug Administration offers detailed guidance for food manufacturers about reducing Listeria contamination, perfect compliance wouldn’t eliminate the risk for consumers because L. monocytogenes is widespread in the environment. The organisms are found in water, soil, sewage, and decaying vegetation, and can be isolated from a variety of animals. Fresh fruits and vegetables are “healthy” choices as long as they are handled and prepared appropriately. Conversely, unpasteurized milk and milk products can never be considered safe.
That’s food for thought.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Kosair Children’s Hospital, also in Louisville. She had no relevant financial disclosures.
Recently, after a long day at the hospital, I stopped at the grocery store to pick up something for a quick dinner. I drifted to the frozen food case in the organic food section, but pulled up short when I saw empty shelves. A paper sign announced that Amy’s Kitchen, a manufacturer of organic and natural frozen foods, had become the latest company to recall its products because of concern about Listeria monocytogenes contamination.
According to information posted on the Food and Drug Administration website, this facultative, anaerobic gram-positive bacillus has been the impetus behind 10 national recalls of food products between April 1 and May 8, 2015 alone. Implicated food products have ranged from gourmet ice cream to soybean sprouts to frozen vegetables. Unlike some other bacterial causes of food-borne illness, Listeria organisms can thrive at cold temperatures. Historically, outbreaks of disease have been linked to a variety of foods, including raw produce, contaminated ready-to-eat foods such as deli meats and prepared salads, and unpasteurized milk and milk products.
Clinical manifestations of listeriosis range from febrile gastroenteritis to bacteremia and meningitis, with severe disease seen primarily in immunocompromised individuals and adults 65 and older.
Pregnant women are especially susceptible, with incidence rates 13 times higher than in the general population. Probably as a result of food choices, Hispanic women are disproportionately affected, with rates up to 24 times higher. Maternal infection may be asymptomatic or may manifest with flulike symptoms that include fever, myalgias, headache, and backache, with or without a preceding diarrhea illness. Even mild maternal illness may result in adverse pregnancy outcomes such as fetal loss, premature labor, and severe neonatal infection.
While medical students and residents are still taught to think of Listeria infection as one of the “big three” causes of neonatal sepsis along with group B streptococcus and Escherichia coli, many pediatricians have never seen a case of this rare, but potentially devastating disease. As with group B streptococcus, both early-onset and late-onset disease occur. Sepsis is the most common presentation of disease in the first week of life, while meningitis predominates in late-onset disease. Pneumonia and myocarditis are occasionally seen. Granulomatosis infantisepticum is an uncommon manifestation of severe, disseminated Listeria infection. Granuloma can occur in nearly every organ, although involvement of the liver and skin is most common.
In 2002, investigators from the Centers for Disease Control and Prevention and the American College of Obstetricians and Gynecologists surveyed more than 400 pregnant women from across the United States about their knowledge of the transmission, risk factors, symptoms, and prevention of listeriosis (Infect. Dis. Obstet. Gyn. 2005;13:11-15). A year later, the Minnesota Department of Health surveyed an additional 286 pregnant women from their state using the same survey instrument.
More than 80% of survey respondents had never heard of the disease, and knowledge about prevention strategies was therefore predictably limited. Only 33% of respondents in the national survey and 17% of respondents in the Minnesota survey knew, for example, that infection could be prevented by avoiding delicatessen meats and soft cheeses. Investigators concluded that “timely and appropriate education” of pregnant women about listeriosis could reduce cases of perinatal infection.
Data from the CDC suggest we have more work to do. The Listeria Initiative is an enhanced national surveillance system that collects laboratory, clinical, and food exposure data about listeriosis cases in the United States. Between 2009 and 2011, 14% of the 1,651 invasive Listeria infections reported were classified as pregnancy associated. Morbidity and mortality were significant, with 40 fetal losses and 6 neonatal deaths (MMWR 2013;62:448-52).
The CDC offers some common sense tips for preventing listeriosis and other food-borne illness. Raw fruits and vegetables should be thoroughly rinsed with tap water and dried with a clean cloth or paper towel before being eaten or cooked. Even foods that are typically peeled first should be washed, and firm produce, such as cantaloupe, should be scrubbed with a produce brush to reduce surface contamination. Uncooked meats and poultry should never come in contact with other food. Hands, knives, cutting boards, and other food preparation surfaces should be washed thoroughly after uncooked food is handled.
Pregnant women and others at increased risk for listeriosis should not eat hot dogs or deli meats unless they are cooked to steaming. Soft cheeses, including feta, brie, Camembert, queso blanco, or anything blue veined, should be avoided unless the label clearly states that the product has been made with pasteurized milk. Even then, it might not be safe. Pasteurized Mexican-style cheeses, such as queso fresco, have been linked to Listeria infections, likely as a result of contamination during the cheese-making process.
Physicians should be prepared to field calls from concerned parents who believe their children may have consumed a product potentially contaminated with Listeria. In general, someone who has eaten a recalled food product but has no symptoms doesn’t need a laboratory evaluation or treatment. Screening blood cultures is not indicated, and routine tests such as a complete blood count are unlikely to be helpful. Instead, patients should be counseled about the symptoms of listeriosis and undergo prompt evaluation if any develop within 2 months of exposure. The typical interval between exposure and the development of symptoms is 1 day to 3 weeks, but may be as long as 70 days.
Although Listeria infection may result in gastrointestinal symptoms, stool cultures are not recommended for diagnosis. According to the CDC, ingestion of food contaminated with Listeria occurs frequently because the organisms are commonly found in the environment. Although uncommon, intermittent fecal carriage and shedding have been observed in asymptomatic individuals.
Back at the grocery, I sighed and resigned myself to a grilled cheese sandwich for dinner. I turned and saw another woman in the aisle stop and read the sign on the freezer case.
“It’s a little scary,” she said with a sigh. “It seems like there is another recall every week, and I’m wondering what’s safe to eat.”
The parents of our patients may have similar questions. Although the Food and Drug Administration offers detailed guidance for food manufacturers about reducing Listeria contamination, perfect compliance wouldn’t eliminate the risk for consumers because L. monocytogenes is widespread in the environment. The organisms are found in water, soil, sewage, and decaying vegetation, and can be isolated from a variety of animals. Fresh fruits and vegetables are “healthy” choices as long as they are handled and prepared appropriately. Conversely, unpasteurized milk and milk products can never be considered safe.
That’s food for thought.
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Kosair Children’s Hospital, also in Louisville. She had no relevant financial disclosures.
Ischemia a bigger concern than PE recurrence?
Among patients taking anticoagulants after venous thromboembolism, mortality due to ischemic events is twice that due to recurrent pulmonary embolism, according to a report published in the Journal of Vascular Surgery: Venous and Lymphatic Disorders. “In clinical practice in patients with VTE, most of the physician’s attention is often focused on the resolution of VTE signs and symptoms, whereas less attention is paid to the prevention of ischemic events,” said Dr. Olga Madridano of Hospital Universitario Infanta Sofia, Madrid, and her associates.
These study findings show that when prescribing anticoagulation for these patients, it is crucial to identify those who may also require concomitant antiplatelet therapy to prevent ischemic events, the investigators noted.
Dr. Madridano and her colleagues analyzed data from an international registry of VTE cases to determine how often major ischemic events – stroke, MI, limb amputation, or mesenteric ischemia – occur during anticoagulant therapy. They assessed the case reports of 23,370 consecutive patients in 10 European countries, Canada, and Ecuador who were enrolled in the registry during a 5-year period.
During a mean of 9.2 months of anticoagulation treatment, 597 patients developed recurrent VTE: 267 had pulmonary embolism and 330 had deep vein thrombosis. A total of 162 developed major ischemic events: 86 had stroke, 53 had MI, 13 required limb amputation, and 11 had mesenteric ischemia. There were 21 deaths from PE recurrences and 53 from ischemic events.
Thus, the number of PE recurrences was greater than that of ischemic events, but the mortality from PE recurrences was only half that from ischemic events. “We hypothesize that some patients who subsequently died of MI or stroke might have benefited from concomitant therapy with antiplatelets,” Dr. Madridano and her associates said (J. Vasc. Surg.: Venous Lymph. Dis. 2015;3:135-41). However, it is important to note that mortality due to bleeding complications (83 deaths) was even higher than mortality due to PE recurrence and ischemic events put together (74 deaths). Earlier discontinuation of anticoagulant therapy might have reduced the rate of major bleeding, they added.
The VTE registry is supported by an unrestricted grant from Sanofi Spain and by Bayer Pharma AG. Dr. Madridano and her associates reported having no financial disclosures.
Among patients taking anticoagulants after venous thromboembolism, mortality due to ischemic events is twice that due to recurrent pulmonary embolism, according to a report published in the Journal of Vascular Surgery: Venous and Lymphatic Disorders. “In clinical practice in patients with VTE, most of the physician’s attention is often focused on the resolution of VTE signs and symptoms, whereas less attention is paid to the prevention of ischemic events,” said Dr. Olga Madridano of Hospital Universitario Infanta Sofia, Madrid, and her associates.
These study findings show that when prescribing anticoagulation for these patients, it is crucial to identify those who may also require concomitant antiplatelet therapy to prevent ischemic events, the investigators noted.
Dr. Madridano and her colleagues analyzed data from an international registry of VTE cases to determine how often major ischemic events – stroke, MI, limb amputation, or mesenteric ischemia – occur during anticoagulant therapy. They assessed the case reports of 23,370 consecutive patients in 10 European countries, Canada, and Ecuador who were enrolled in the registry during a 5-year period.
During a mean of 9.2 months of anticoagulation treatment, 597 patients developed recurrent VTE: 267 had pulmonary embolism and 330 had deep vein thrombosis. A total of 162 developed major ischemic events: 86 had stroke, 53 had MI, 13 required limb amputation, and 11 had mesenteric ischemia. There were 21 deaths from PE recurrences and 53 from ischemic events.
Thus, the number of PE recurrences was greater than that of ischemic events, but the mortality from PE recurrences was only half that from ischemic events. “We hypothesize that some patients who subsequently died of MI or stroke might have benefited from concomitant therapy with antiplatelets,” Dr. Madridano and her associates said (J. Vasc. Surg.: Venous Lymph. Dis. 2015;3:135-41). However, it is important to note that mortality due to bleeding complications (83 deaths) was even higher than mortality due to PE recurrence and ischemic events put together (74 deaths). Earlier discontinuation of anticoagulant therapy might have reduced the rate of major bleeding, they added.
The VTE registry is supported by an unrestricted grant from Sanofi Spain and by Bayer Pharma AG. Dr. Madridano and her associates reported having no financial disclosures.
Among patients taking anticoagulants after venous thromboembolism, mortality due to ischemic events is twice that due to recurrent pulmonary embolism, according to a report published in the Journal of Vascular Surgery: Venous and Lymphatic Disorders. “In clinical practice in patients with VTE, most of the physician’s attention is often focused on the resolution of VTE signs and symptoms, whereas less attention is paid to the prevention of ischemic events,” said Dr. Olga Madridano of Hospital Universitario Infanta Sofia, Madrid, and her associates.
These study findings show that when prescribing anticoagulation for these patients, it is crucial to identify those who may also require concomitant antiplatelet therapy to prevent ischemic events, the investigators noted.
Dr. Madridano and her colleagues analyzed data from an international registry of VTE cases to determine how often major ischemic events – stroke, MI, limb amputation, or mesenteric ischemia – occur during anticoagulant therapy. They assessed the case reports of 23,370 consecutive patients in 10 European countries, Canada, and Ecuador who were enrolled in the registry during a 5-year period.
During a mean of 9.2 months of anticoagulation treatment, 597 patients developed recurrent VTE: 267 had pulmonary embolism and 330 had deep vein thrombosis. A total of 162 developed major ischemic events: 86 had stroke, 53 had MI, 13 required limb amputation, and 11 had mesenteric ischemia. There were 21 deaths from PE recurrences and 53 from ischemic events.
Thus, the number of PE recurrences was greater than that of ischemic events, but the mortality from PE recurrences was only half that from ischemic events. “We hypothesize that some patients who subsequently died of MI or stroke might have benefited from concomitant therapy with antiplatelets,” Dr. Madridano and her associates said (J. Vasc. Surg.: Venous Lymph. Dis. 2015;3:135-41). However, it is important to note that mortality due to bleeding complications (83 deaths) was even higher than mortality due to PE recurrence and ischemic events put together (74 deaths). Earlier discontinuation of anticoagulant therapy might have reduced the rate of major bleeding, they added.
The VTE registry is supported by an unrestricted grant from Sanofi Spain and by Bayer Pharma AG. Dr. Madridano and her associates reported having no financial disclosures.
Key clinical point: Among patients receiving anticoagulation after VTE, mortality from ischemic events is twice that from recurrent pulmonary embolism.
Major finding: During a mean of 9.2 months of anticoagulation treatment, there were 21 deaths from PE recurrences and 53 from ischemic events.
Data source: An observational cohort study involving 23,370 consecutive patients entered into an international registry of acute VTE cases during a 5-year period.
Disclosures: The VTE registry is supported by an unrestricted grant from Sanofi Spain and by Bayer Pharma AG. Dr. Madridano and her associates reported having no financial disclosures.
Statins, fibrates lower stroke risk in elderly
Both statin and fibrate therapies taken to improve lipid profiles decreased the risk of stroke by 30% in a community-dwelling population of elderly people, according to prospective European study published online May 19 in the British Medical Journal.
Participants in almost all the randomized clinical trials assessing cardiovascular drugs are younger than age 70, so the benefits of these agents in older patients – particularly their effectiveness as primary prevention in people who have no known cardiovascular illness – is uncertain. Nevertheless, “in real life, statins are commonly prescribed to older people without clinical evidence of atherosclerosis,” said Dr. Annick Alperovitch of the University of Bordeaux (France) and her associates.
To assess the effects of statin and fibrate therapies on incident cardiovascular events in an elderly population, the investigators analyzed data from an ongoing cohort study of vascular disease among elderly residents of Bordeaux, Dijon, and Montpellier. Dr. Alperovitch and her associates examined the medical records of a subset of 7,484 men and women (mean age 74 years) who were followed every 2 years for a mean of 9 years. A total of 27% reported using lipid-lowering medications at baseline; roughly half used statins and half used fibrates. There were 292 strokes during follow-up.
The risk of stroke was cut by roughly 30% among statin and fibrate users, compared with nonusers (hazard ratio, 0.66). This decrease was similar between the two medications. All-cause mortality was slightly lower in people who took statins or fibrates, compared with nonusers (HR 0.87), the investigators said (Br. Med. J. 2015 May 19 [doi:10.1136/bmj.h2335]).
This is the first observational study to show a significant association between lipid-lowering drugs and decreased stroke risk, they noted.
The overall incidence of stroke in this study was low (0.47 per 100 person-years), so even a 30% decrease produced “a limited number of avoided cases.” That may be attributable in part to the generally healthy lifestyle, high educational achievement, and high economic status of this urban French study population. But if the findings are confirmed in future studies, they could have an important impact on public health in other populations, Dr. Alperovitch and her associates said.
The findings of a single observational study will not change guidelines regarding cholesterol therapy, but they are sufficiently compelling to warrant further research on primary prevention of stroke in elderly people.
This is the first observational study to describe a significant association between lipid-lowering medications and decreased stroke risk. Previous studies have shown only a weak association in early middle age, and no association in the elderly.
Graeme J. Hankey, M.D., is professor of neurology at the University of Western Australia’s Harry Perkins Institute of Medical Research, Perth, and honorary senior research fellow at Western Australian Neuroscience Research Institute, also in Perth. He reported ties to Sanofi Aventis, Bayer Pharmaceuticals, and Medscape. Dr. Hankey made these remarks in an editorial accompanying Dr. Alperovitch’s report (Br. Med. J. 2015 May 19 [doi:10.1136/bmj.h2568]).
The findings of a single observational study will not change guidelines regarding cholesterol therapy, but they are sufficiently compelling to warrant further research on primary prevention of stroke in elderly people.
This is the first observational study to describe a significant association between lipid-lowering medications and decreased stroke risk. Previous studies have shown only a weak association in early middle age, and no association in the elderly.
Graeme J. Hankey, M.D., is professor of neurology at the University of Western Australia’s Harry Perkins Institute of Medical Research, Perth, and honorary senior research fellow at Western Australian Neuroscience Research Institute, also in Perth. He reported ties to Sanofi Aventis, Bayer Pharmaceuticals, and Medscape. Dr. Hankey made these remarks in an editorial accompanying Dr. Alperovitch’s report (Br. Med. J. 2015 May 19 [doi:10.1136/bmj.h2568]).
The findings of a single observational study will not change guidelines regarding cholesterol therapy, but they are sufficiently compelling to warrant further research on primary prevention of stroke in elderly people.
This is the first observational study to describe a significant association between lipid-lowering medications and decreased stroke risk. Previous studies have shown only a weak association in early middle age, and no association in the elderly.
Graeme J. Hankey, M.D., is professor of neurology at the University of Western Australia’s Harry Perkins Institute of Medical Research, Perth, and honorary senior research fellow at Western Australian Neuroscience Research Institute, also in Perth. He reported ties to Sanofi Aventis, Bayer Pharmaceuticals, and Medscape. Dr. Hankey made these remarks in an editorial accompanying Dr. Alperovitch’s report (Br. Med. J. 2015 May 19 [doi:10.1136/bmj.h2568]).
Both statin and fibrate therapies taken to improve lipid profiles decreased the risk of stroke by 30% in a community-dwelling population of elderly people, according to prospective European study published online May 19 in the British Medical Journal.
Participants in almost all the randomized clinical trials assessing cardiovascular drugs are younger than age 70, so the benefits of these agents in older patients – particularly their effectiveness as primary prevention in people who have no known cardiovascular illness – is uncertain. Nevertheless, “in real life, statins are commonly prescribed to older people without clinical evidence of atherosclerosis,” said Dr. Annick Alperovitch of the University of Bordeaux (France) and her associates.
To assess the effects of statin and fibrate therapies on incident cardiovascular events in an elderly population, the investigators analyzed data from an ongoing cohort study of vascular disease among elderly residents of Bordeaux, Dijon, and Montpellier. Dr. Alperovitch and her associates examined the medical records of a subset of 7,484 men and women (mean age 74 years) who were followed every 2 years for a mean of 9 years. A total of 27% reported using lipid-lowering medications at baseline; roughly half used statins and half used fibrates. There were 292 strokes during follow-up.
The risk of stroke was cut by roughly 30% among statin and fibrate users, compared with nonusers (hazard ratio, 0.66). This decrease was similar between the two medications. All-cause mortality was slightly lower in people who took statins or fibrates, compared with nonusers (HR 0.87), the investigators said (Br. Med. J. 2015 May 19 [doi:10.1136/bmj.h2335]).
This is the first observational study to show a significant association between lipid-lowering drugs and decreased stroke risk, they noted.
The overall incidence of stroke in this study was low (0.47 per 100 person-years), so even a 30% decrease produced “a limited number of avoided cases.” That may be attributable in part to the generally healthy lifestyle, high educational achievement, and high economic status of this urban French study population. But if the findings are confirmed in future studies, they could have an important impact on public health in other populations, Dr. Alperovitch and her associates said.
Both statin and fibrate therapies taken to improve lipid profiles decreased the risk of stroke by 30% in a community-dwelling population of elderly people, according to prospective European study published online May 19 in the British Medical Journal.
Participants in almost all the randomized clinical trials assessing cardiovascular drugs are younger than age 70, so the benefits of these agents in older patients – particularly their effectiveness as primary prevention in people who have no known cardiovascular illness – is uncertain. Nevertheless, “in real life, statins are commonly prescribed to older people without clinical evidence of atherosclerosis,” said Dr. Annick Alperovitch of the University of Bordeaux (France) and her associates.
To assess the effects of statin and fibrate therapies on incident cardiovascular events in an elderly population, the investigators analyzed data from an ongoing cohort study of vascular disease among elderly residents of Bordeaux, Dijon, and Montpellier. Dr. Alperovitch and her associates examined the medical records of a subset of 7,484 men and women (mean age 74 years) who were followed every 2 years for a mean of 9 years. A total of 27% reported using lipid-lowering medications at baseline; roughly half used statins and half used fibrates. There were 292 strokes during follow-up.
The risk of stroke was cut by roughly 30% among statin and fibrate users, compared with nonusers (hazard ratio, 0.66). This decrease was similar between the two medications. All-cause mortality was slightly lower in people who took statins or fibrates, compared with nonusers (HR 0.87), the investigators said (Br. Med. J. 2015 May 19 [doi:10.1136/bmj.h2335]).
This is the first observational study to show a significant association between lipid-lowering drugs and decreased stroke risk, they noted.
The overall incidence of stroke in this study was low (0.47 per 100 person-years), so even a 30% decrease produced “a limited number of avoided cases.” That may be attributable in part to the generally healthy lifestyle, high educational achievement, and high economic status of this urban French study population. But if the findings are confirmed in future studies, they could have an important impact on public health in other populations, Dr. Alperovitch and her associates said.
Key clinical point: Statin and fibrate therapies significantly decreased stroke risk in a large cohort of elderly people.
Major finding: The risk of stroke was cut by 30% among statin and fibrate users, compared with nonusers (HR, 0.66).
Data source: A prospective, population-based cohort study of 7,484 elderly community-dwelling people followed for 9 years.
Disclosures: This study was supported by Institut National de la Sante et de la Recherche Medicale, Victor Segalen-Bordeaux II University, and Sanofi-Aventis. Dr. Alperovitch reported receiving honoraria from the French National Medicines Agency, the Fondation Plan Alzheimer, and Fondation Bettencourt-Schueller.