User login
Trump lays out principles for ACA replacement
Flexibility and choice were key themes in the health care reform vision President Trump outlined in his first speech to a joint session of Congress on Feb. 28.
Specifically, Americans should be able to “purchase their own coverage through the use of tax credits and expanded health savings accounts, but it must be the plan they want, not the plan forced on them by our government,” Mr. Trump said. They also should be able to purchase insurance across state lines, which “will create a truly competitive national marketplace that will bring costs way down and provide far better care.”
He also advocated providing more flexibility to states to improve Medicaid but did not provide any specifics on how that would be accomplished.
Finally, Mr. Trump called for “legal reforms that protect patients and doctors from unnecessary costs that drive up the price of insurance and work to bring down the artificially high price of drugs and bring them down immediately,” he said, adding that the Food and Drug Administration needs to slash the time to approval for drugs and other medical treatments.
Some of these concepts were mirrored in a talking-points memo from the House Republican leadership earlier in February.
According to the memo, Republican efforts to repeal and replace the ACA will focus on the following areas:
• Repealing the ACA’s Medicaid expansion and allowing states to choose between block grands or per capita grants for Medicaid funding. States could choose to use those grants and per capita grants to keep expansion.
• Rebranding high-risk pools as “state innovation grants” to provide states with flexibility to design coverage that meets the needs of their populations. States could use the innovation grants to reduce patient out-of-pocket costs, lower the cost of providing care, stabilize the individual and small-group markets, provide access to preventive care, and promote participation in private health care plans.
• Promoting health savings accounts tied to high-deductible plans through increasing maximum contribution limits and other administrative fixes to allow for greater flexibility in their use.
• Replacing ACA premium subsides with refundable flat tax credits. Credit would be age-based, with younger recipients receiving smaller credits and older taxpayers being eligible for more. Tax credits would be available to those who are not eligible to receive coverage through other sources, such as an employer or government program.
Many of these provisions were included in a health reform plan known as A Better Way, which was announced in June 2016 by House Speaker Paul Ryan (R-Wis.).
Like Mr. Trump’s outline to Congress, the GOP talking-points memo was light on specifics, including how the bill will be paid for, how much money will be distributed to states for Medicaid, and how these provisions would alter current insurance coverage rates, which government actuaries project would reach 91.5% by 2025 under current law.
The GOP talking-points memo followed a Feb. 10 discussion draft that included the legislative language required to implement these concepts and allowed insurers to charge higher premiums to people with coverage lapses; repealed a number of taxes imposed on insurers, pharmaceutical companies, and device manufacturers; eliminated many of the ACA’s essential benefits; and ended tax penalties on companies that do not provide coverage to their employees.
This proposal, however, is already running into opposition from House Republicans, with reports stating that blocks of Republicans would not approve of the provisions.
It also could run aground in the Senate, where Sen. Lamar Alexander (R-Tenn.), chairman of the Committee on Health, Education, Labor, and Pensions, has expressed his intent to tackle heath care reform in pieces (public programs, the individual market, and employer market) to avoid getting bogged down in one overarching piece of legislation.
Getting Republicans on the same page is going to be a huge hurdle to get any legislation passed long before it comes down to trying to secure any Democratic votes to help pass any replacement legislation.
As former House Speaker John Boehner told physicians and health care IT leaders at the HIMSS annual conference on Feb. 23, “In the 25 years that I served in the Unites States Congress, Republicans never ever one time agreed on what a health care proposal should look like. Not once. … If you repeal without replace, anything that happens is your fault. You broke it. … If you pass repeal without replace, you’ll never pass replace because they will never ever agree on what the [replacement] bill should be.”
Flexibility and choice were key themes in the health care reform vision President Trump outlined in his first speech to a joint session of Congress on Feb. 28.
Specifically, Americans should be able to “purchase their own coverage through the use of tax credits and expanded health savings accounts, but it must be the plan they want, not the plan forced on them by our government,” Mr. Trump said. They also should be able to purchase insurance across state lines, which “will create a truly competitive national marketplace that will bring costs way down and provide far better care.”
He also advocated providing more flexibility to states to improve Medicaid but did not provide any specifics on how that would be accomplished.
Finally, Mr. Trump called for “legal reforms that protect patients and doctors from unnecessary costs that drive up the price of insurance and work to bring down the artificially high price of drugs and bring them down immediately,” he said, adding that the Food and Drug Administration needs to slash the time to approval for drugs and other medical treatments.
Some of these concepts were mirrored in a talking-points memo from the House Republican leadership earlier in February.
According to the memo, Republican efforts to repeal and replace the ACA will focus on the following areas:
• Repealing the ACA’s Medicaid expansion and allowing states to choose between block grands or per capita grants for Medicaid funding. States could choose to use those grants and per capita grants to keep expansion.
• Rebranding high-risk pools as “state innovation grants” to provide states with flexibility to design coverage that meets the needs of their populations. States could use the innovation grants to reduce patient out-of-pocket costs, lower the cost of providing care, stabilize the individual and small-group markets, provide access to preventive care, and promote participation in private health care plans.
• Promoting health savings accounts tied to high-deductible plans through increasing maximum contribution limits and other administrative fixes to allow for greater flexibility in their use.
• Replacing ACA premium subsides with refundable flat tax credits. Credit would be age-based, with younger recipients receiving smaller credits and older taxpayers being eligible for more. Tax credits would be available to those who are not eligible to receive coverage through other sources, such as an employer or government program.
Many of these provisions were included in a health reform plan known as A Better Way, which was announced in June 2016 by House Speaker Paul Ryan (R-Wis.).
Like Mr. Trump’s outline to Congress, the GOP talking-points memo was light on specifics, including how the bill will be paid for, how much money will be distributed to states for Medicaid, and how these provisions would alter current insurance coverage rates, which government actuaries project would reach 91.5% by 2025 under current law.
The GOP talking-points memo followed a Feb. 10 discussion draft that included the legislative language required to implement these concepts and allowed insurers to charge higher premiums to people with coverage lapses; repealed a number of taxes imposed on insurers, pharmaceutical companies, and device manufacturers; eliminated many of the ACA’s essential benefits; and ended tax penalties on companies that do not provide coverage to their employees.
This proposal, however, is already running into opposition from House Republicans, with reports stating that blocks of Republicans would not approve of the provisions.
It also could run aground in the Senate, where Sen. Lamar Alexander (R-Tenn.), chairman of the Committee on Health, Education, Labor, and Pensions, has expressed his intent to tackle heath care reform in pieces (public programs, the individual market, and employer market) to avoid getting bogged down in one overarching piece of legislation.
Getting Republicans on the same page is going to be a huge hurdle to get any legislation passed long before it comes down to trying to secure any Democratic votes to help pass any replacement legislation.
As former House Speaker John Boehner told physicians and health care IT leaders at the HIMSS annual conference on Feb. 23, “In the 25 years that I served in the Unites States Congress, Republicans never ever one time agreed on what a health care proposal should look like. Not once. … If you repeal without replace, anything that happens is your fault. You broke it. … If you pass repeal without replace, you’ll never pass replace because they will never ever agree on what the [replacement] bill should be.”
Flexibility and choice were key themes in the health care reform vision President Trump outlined in his first speech to a joint session of Congress on Feb. 28.
Specifically, Americans should be able to “purchase their own coverage through the use of tax credits and expanded health savings accounts, but it must be the plan they want, not the plan forced on them by our government,” Mr. Trump said. They also should be able to purchase insurance across state lines, which “will create a truly competitive national marketplace that will bring costs way down and provide far better care.”
He also advocated providing more flexibility to states to improve Medicaid but did not provide any specifics on how that would be accomplished.
Finally, Mr. Trump called for “legal reforms that protect patients and doctors from unnecessary costs that drive up the price of insurance and work to bring down the artificially high price of drugs and bring them down immediately,” he said, adding that the Food and Drug Administration needs to slash the time to approval for drugs and other medical treatments.
Some of these concepts were mirrored in a talking-points memo from the House Republican leadership earlier in February.
According to the memo, Republican efforts to repeal and replace the ACA will focus on the following areas:
• Repealing the ACA’s Medicaid expansion and allowing states to choose between block grands or per capita grants for Medicaid funding. States could choose to use those grants and per capita grants to keep expansion.
• Rebranding high-risk pools as “state innovation grants” to provide states with flexibility to design coverage that meets the needs of their populations. States could use the innovation grants to reduce patient out-of-pocket costs, lower the cost of providing care, stabilize the individual and small-group markets, provide access to preventive care, and promote participation in private health care plans.
• Promoting health savings accounts tied to high-deductible plans through increasing maximum contribution limits and other administrative fixes to allow for greater flexibility in their use.
• Replacing ACA premium subsides with refundable flat tax credits. Credit would be age-based, with younger recipients receiving smaller credits and older taxpayers being eligible for more. Tax credits would be available to those who are not eligible to receive coverage through other sources, such as an employer or government program.
Many of these provisions were included in a health reform plan known as A Better Way, which was announced in June 2016 by House Speaker Paul Ryan (R-Wis.).
Like Mr. Trump’s outline to Congress, the GOP talking-points memo was light on specifics, including how the bill will be paid for, how much money will be distributed to states for Medicaid, and how these provisions would alter current insurance coverage rates, which government actuaries project would reach 91.5% by 2025 under current law.
The GOP talking-points memo followed a Feb. 10 discussion draft that included the legislative language required to implement these concepts and allowed insurers to charge higher premiums to people with coverage lapses; repealed a number of taxes imposed on insurers, pharmaceutical companies, and device manufacturers; eliminated many of the ACA’s essential benefits; and ended tax penalties on companies that do not provide coverage to their employees.
This proposal, however, is already running into opposition from House Republicans, with reports stating that blocks of Republicans would not approve of the provisions.
It also could run aground in the Senate, where Sen. Lamar Alexander (R-Tenn.), chairman of the Committee on Health, Education, Labor, and Pensions, has expressed his intent to tackle heath care reform in pieces (public programs, the individual market, and employer market) to avoid getting bogged down in one overarching piece of legislation.
Getting Republicans on the same page is going to be a huge hurdle to get any legislation passed long before it comes down to trying to secure any Democratic votes to help pass any replacement legislation.
As former House Speaker John Boehner told physicians and health care IT leaders at the HIMSS annual conference on Feb. 23, “In the 25 years that I served in the Unites States Congress, Republicans never ever one time agreed on what a health care proposal should look like. Not once. … If you repeal without replace, anything that happens is your fault. You broke it. … If you pass repeal without replace, you’ll never pass replace because they will never ever agree on what the [replacement] bill should be.”
ACIP debates adding third dose to current mumps recommendation
Because of a spate of mumps outbreaks over the last decade, adding a third dose of the mumps vaccine to the currently standard two-dose series was debated during a meeting of the Center for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP).
“Data [on recent outbreaks] were presented to ACIP in 2012, and ACIP determined that the data were insufficient to recommend for or against the use of a third dose of MMR vaccine for mumps outbreak control,” explained Mona Marin, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. “Subsequently, CDC issued guidance for consideration for use of a third dose in specifically identified target populations, along with criteria for public health departments to consider for decision-making. That includes settings with high two-dose coverage, intense exposure, and ongoing transmission.”
Recent data, explained Dr. Marin, have “raised the question of the short- and long-term benefits of a third dose, and implications for routine use versus outbreak policy recommendations.” However, the efficacy of a third vaccine dose has not been verified against cell memory, cell-mediated response, and other factors. These will need to be evaluated before a third dose can be debated further, let alone approved.
The mumps work group, therefore, will continue to assess the benefits and potential harms of adding a third dose to the immunization schedule. Dr. Marin explained that they hope to be able to discuss this further, and perhaps vote on it, during the next ACIP meeting, which is scheduled to take place on June 21 and 22 of this year.
“The current two-dose schedule is sufficient for mumps control in the general population, but outbreaks can occur in well-vaccinated populations in specific settings,” Dr. Marin said. “Intense exposure settings and waning immunity appear to be risk factors for secondary vaccine failure. The benefit of a third MMR dose still needs to be assessed.”
Dr. Marin said she had no relevant financial disclosures.
Because of a spate of mumps outbreaks over the last decade, adding a third dose of the mumps vaccine to the currently standard two-dose series was debated during a meeting of the Center for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP).
“Data [on recent outbreaks] were presented to ACIP in 2012, and ACIP determined that the data were insufficient to recommend for or against the use of a third dose of MMR vaccine for mumps outbreak control,” explained Mona Marin, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. “Subsequently, CDC issued guidance for consideration for use of a third dose in specifically identified target populations, along with criteria for public health departments to consider for decision-making. That includes settings with high two-dose coverage, intense exposure, and ongoing transmission.”
Recent data, explained Dr. Marin, have “raised the question of the short- and long-term benefits of a third dose, and implications for routine use versus outbreak policy recommendations.” However, the efficacy of a third vaccine dose has not been verified against cell memory, cell-mediated response, and other factors. These will need to be evaluated before a third dose can be debated further, let alone approved.
The mumps work group, therefore, will continue to assess the benefits and potential harms of adding a third dose to the immunization schedule. Dr. Marin explained that they hope to be able to discuss this further, and perhaps vote on it, during the next ACIP meeting, which is scheduled to take place on June 21 and 22 of this year.
“The current two-dose schedule is sufficient for mumps control in the general population, but outbreaks can occur in well-vaccinated populations in specific settings,” Dr. Marin said. “Intense exposure settings and waning immunity appear to be risk factors for secondary vaccine failure. The benefit of a third MMR dose still needs to be assessed.”
Dr. Marin said she had no relevant financial disclosures.
Because of a spate of mumps outbreaks over the last decade, adding a third dose of the mumps vaccine to the currently standard two-dose series was debated during a meeting of the Center for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP).
“Data [on recent outbreaks] were presented to ACIP in 2012, and ACIP determined that the data were insufficient to recommend for or against the use of a third dose of MMR vaccine for mumps outbreak control,” explained Mona Marin, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. “Subsequently, CDC issued guidance for consideration for use of a third dose in specifically identified target populations, along with criteria for public health departments to consider for decision-making. That includes settings with high two-dose coverage, intense exposure, and ongoing transmission.”
Recent data, explained Dr. Marin, have “raised the question of the short- and long-term benefits of a third dose, and implications for routine use versus outbreak policy recommendations.” However, the efficacy of a third vaccine dose has not been verified against cell memory, cell-mediated response, and other factors. These will need to be evaluated before a third dose can be debated further, let alone approved.
The mumps work group, therefore, will continue to assess the benefits and potential harms of adding a third dose to the immunization schedule. Dr. Marin explained that they hope to be able to discuss this further, and perhaps vote on it, during the next ACIP meeting, which is scheduled to take place on June 21 and 22 of this year.
“The current two-dose schedule is sufficient for mumps control in the general population, but outbreaks can occur in well-vaccinated populations in specific settings,” Dr. Marin said. “Intense exposure settings and waning immunity appear to be risk factors for secondary vaccine failure. The benefit of a third MMR dose still needs to be assessed.”
Dr. Marin said she had no relevant financial disclosures.
FROM AN ACIP MEETING
Single dose of ZA pre-transplant prevents bone loss
ORLANDO, FL—A single, 4 mg-dose of the bisphosphonate zoledronic acid (ZA) prior to allogeneic hematopoietic stem cell transplant (HSCT) prevents femoral neck (FN) bone loss at day 100 in patients with lymphoid or myeloid malignancies, according to new research.
The study also suggests that patients who receive risk-adapted ZA therapy after transplant can experience significant reductions in bone loss between days 100 and 365.
However, patients with acute and chronic graft-versus-host disease (GVHD) continue to be at risk of bone loss.
Eric Wong, of the Royal Melbourne Hospital in Parkville, Australia, presented these findings at the 2017 BMT Tandem Meetings (abstract 53) on behalf of the Australasian Leukaemia and Lymphoma Group.
“Previous studies have demonstrated that efforts to prevent bone loss through calcium and vitamin D supplementation as well as hormone-replacement therapy alone have been ineffective in preventing bone loss,” Wong explained.
And monthly pamidronate begun prior to HSCT reduces, but does not prevent, FN bone density loss.
So Wong and his colleagues began a trial of ZA, which is approximately 100-fold more potent than pamidronate.
Study design
The researchers enrolled 82 patients into the phase 2 ALLG BM07 trial. All patients received a single shot of ZA prior to HSCT conditioning.
All patients also received vitamin D and calcium supplements, and pre-menopausal women received hormone replacement therapy.
Depending on their risk of bone loss, patients received individualized ZA therapy after transplant. Researchers assessed the patients’ bone density at days 100, 180, 270, and 365 post-HSCT. Patients at high risk of bone loss received additional doses of ZA.
Risks for bone loss included bone mineral density (BMD) reduction of 5% or greater compared to baseline, prednisolone exposure of 1 mg/kg/d or greater for 2 weeks, or prednisolone exposure of 10 mg/d or more for 6 weeks
The primary endpoint of the study was the change in FN BMD at days 100 and 365 after HSCT compared to baseline.
The researchers also compared patients’ FN BMD with 35 untreated historical controls assessed at the same time points.
Patient characteristics
Seventy patients were alive and had not relapsed at day 100. Most (87%) were 60 years or younger, 60% were male, 53% had myeloid disease, 43% lymphoid, and 4% other disease.
“The most common indications for transplant were acute myeloid leukemia and acute lymphoblastic leukemia,” Wong said, “which, together, formed over 50% of the entire cohort.”
Seventy percent of patients were ECOG status 0 or 1, and 30% were 2 or greater.
Most (59%) had received myeloablative conditioning, the predominant regimens being busulfan/cyclophosphamide or cyclophosphamide/total-body irradiation. And the most common reduced-intensity conditioning regimen was fludarabine/melphalan.
Fifty-six percent of patients had a sibling donor, and 43% had a matched unrelated donor.
Thirty-eight percent of patients developed acute GVHD—19% grade 1, and 19% grade 2 to 3.
“Of note,” Wong said, “no patient developed grade 4 acute GVHD.”
Patients received a median of 2 ZA doses (range, 1–4), including the pre-transplant dose.
Sixty patients received at least 1 dose of ZA between day 100 and day 365, including 33% who received multiple doses.
At day 100, 33 patients received additional ZA. At day 180, 27 patients received additional ZA, including 8 patients who did not have it at day 100. And at day 270, 18 patients had additional ZA, including 1 patient who had no additional ZA at earlier time points.
Results
At day 100, there was no statistically significant change in FN bone density compared with baseline. The mean change was -2.6% (range, -6.6% to 1.4%).
For patients with acute GVHD, however, the change in bone density was significant (P=0.03). Patients with grade 1-2 GVHD had a mean change of -1.6% ± 3.7%, and patients with grade 3-4 GVHD had a mean change of -8.5% ± 11.2%.
Sixty-five patients were available for the day 365 efficacy analysis.
Bone density did not change significantly between day 100 and 365 for the entire group.
“By day 365,” Wong noted, “there was a net loss of bone density of -2.9%.”
But by day 365, patients with extensive chronic GVHD had significantly more bone loss compared with patients who had no chronic GVHD (P=0.03).
Age, sex, duration of cyclosporine, and mean steroid dose were not associated with a change in bone density at day 100 or 365, although there was a trend for an association between high steroid exposure and increased bone loss (P=0.07).
When the researchers compared the patients to untreated historical controls, patients who received ZA had significantly less bone loss at day 100 (P=0.001) and day 365 (P<0.0001).
The researchers observed no serious adverse events with ZA.
Wong concluded that patients with extensive GVHD are a “high-risk cohort that needs augmented therapies.” ![]()
ORLANDO, FL—A single, 4 mg-dose of the bisphosphonate zoledronic acid (ZA) prior to allogeneic hematopoietic stem cell transplant (HSCT) prevents femoral neck (FN) bone loss at day 100 in patients with lymphoid or myeloid malignancies, according to new research.
The study also suggests that patients who receive risk-adapted ZA therapy after transplant can experience significant reductions in bone loss between days 100 and 365.
However, patients with acute and chronic graft-versus-host disease (GVHD) continue to be at risk of bone loss.
Eric Wong, of the Royal Melbourne Hospital in Parkville, Australia, presented these findings at the 2017 BMT Tandem Meetings (abstract 53) on behalf of the Australasian Leukaemia and Lymphoma Group.
“Previous studies have demonstrated that efforts to prevent bone loss through calcium and vitamin D supplementation as well as hormone-replacement therapy alone have been ineffective in preventing bone loss,” Wong explained.
And monthly pamidronate begun prior to HSCT reduces, but does not prevent, FN bone density loss.
So Wong and his colleagues began a trial of ZA, which is approximately 100-fold more potent than pamidronate.
Study design
The researchers enrolled 82 patients into the phase 2 ALLG BM07 trial. All patients received a single shot of ZA prior to HSCT conditioning.
All patients also received vitamin D and calcium supplements, and pre-menopausal women received hormone replacement therapy.
Depending on their risk of bone loss, patients received individualized ZA therapy after transplant. Researchers assessed the patients’ bone density at days 100, 180, 270, and 365 post-HSCT. Patients at high risk of bone loss received additional doses of ZA.
Risks for bone loss included bone mineral density (BMD) reduction of 5% or greater compared to baseline, prednisolone exposure of 1 mg/kg/d or greater for 2 weeks, or prednisolone exposure of 10 mg/d or more for 6 weeks
The primary endpoint of the study was the change in FN BMD at days 100 and 365 after HSCT compared to baseline.
The researchers also compared patients’ FN BMD with 35 untreated historical controls assessed at the same time points.
Patient characteristics
Seventy patients were alive and had not relapsed at day 100. Most (87%) were 60 years or younger, 60% were male, 53% had myeloid disease, 43% lymphoid, and 4% other disease.
“The most common indications for transplant were acute myeloid leukemia and acute lymphoblastic leukemia,” Wong said, “which, together, formed over 50% of the entire cohort.”
Seventy percent of patients were ECOG status 0 or 1, and 30% were 2 or greater.
Most (59%) had received myeloablative conditioning, the predominant regimens being busulfan/cyclophosphamide or cyclophosphamide/total-body irradiation. And the most common reduced-intensity conditioning regimen was fludarabine/melphalan.
Fifty-six percent of patients had a sibling donor, and 43% had a matched unrelated donor.
Thirty-eight percent of patients developed acute GVHD—19% grade 1, and 19% grade 2 to 3.
“Of note,” Wong said, “no patient developed grade 4 acute GVHD.”
Patients received a median of 2 ZA doses (range, 1–4), including the pre-transplant dose.
Sixty patients received at least 1 dose of ZA between day 100 and day 365, including 33% who received multiple doses.
At day 100, 33 patients received additional ZA. At day 180, 27 patients received additional ZA, including 8 patients who did not have it at day 100. And at day 270, 18 patients had additional ZA, including 1 patient who had no additional ZA at earlier time points.
Results
At day 100, there was no statistically significant change in FN bone density compared with baseline. The mean change was -2.6% (range, -6.6% to 1.4%).
For patients with acute GVHD, however, the change in bone density was significant (P=0.03). Patients with grade 1-2 GVHD had a mean change of -1.6% ± 3.7%, and patients with grade 3-4 GVHD had a mean change of -8.5% ± 11.2%.
Sixty-five patients were available for the day 365 efficacy analysis.
Bone density did not change significantly between day 100 and 365 for the entire group.
“By day 365,” Wong noted, “there was a net loss of bone density of -2.9%.”
But by day 365, patients with extensive chronic GVHD had significantly more bone loss compared with patients who had no chronic GVHD (P=0.03).
Age, sex, duration of cyclosporine, and mean steroid dose were not associated with a change in bone density at day 100 or 365, although there was a trend for an association between high steroid exposure and increased bone loss (P=0.07).
When the researchers compared the patients to untreated historical controls, patients who received ZA had significantly less bone loss at day 100 (P=0.001) and day 365 (P<0.0001).
The researchers observed no serious adverse events with ZA.
Wong concluded that patients with extensive GVHD are a “high-risk cohort that needs augmented therapies.” ![]()
ORLANDO, FL—A single, 4 mg-dose of the bisphosphonate zoledronic acid (ZA) prior to allogeneic hematopoietic stem cell transplant (HSCT) prevents femoral neck (FN) bone loss at day 100 in patients with lymphoid or myeloid malignancies, according to new research.
The study also suggests that patients who receive risk-adapted ZA therapy after transplant can experience significant reductions in bone loss between days 100 and 365.
However, patients with acute and chronic graft-versus-host disease (GVHD) continue to be at risk of bone loss.
Eric Wong, of the Royal Melbourne Hospital in Parkville, Australia, presented these findings at the 2017 BMT Tandem Meetings (abstract 53) on behalf of the Australasian Leukaemia and Lymphoma Group.
“Previous studies have demonstrated that efforts to prevent bone loss through calcium and vitamin D supplementation as well as hormone-replacement therapy alone have been ineffective in preventing bone loss,” Wong explained.
And monthly pamidronate begun prior to HSCT reduces, but does not prevent, FN bone density loss.
So Wong and his colleagues began a trial of ZA, which is approximately 100-fold more potent than pamidronate.
Study design
The researchers enrolled 82 patients into the phase 2 ALLG BM07 trial. All patients received a single shot of ZA prior to HSCT conditioning.
All patients also received vitamin D and calcium supplements, and pre-menopausal women received hormone replacement therapy.
Depending on their risk of bone loss, patients received individualized ZA therapy after transplant. Researchers assessed the patients’ bone density at days 100, 180, 270, and 365 post-HSCT. Patients at high risk of bone loss received additional doses of ZA.
Risks for bone loss included bone mineral density (BMD) reduction of 5% or greater compared to baseline, prednisolone exposure of 1 mg/kg/d or greater for 2 weeks, or prednisolone exposure of 10 mg/d or more for 6 weeks
The primary endpoint of the study was the change in FN BMD at days 100 and 365 after HSCT compared to baseline.
The researchers also compared patients’ FN BMD with 35 untreated historical controls assessed at the same time points.
Patient characteristics
Seventy patients were alive and had not relapsed at day 100. Most (87%) were 60 years or younger, 60% were male, 53% had myeloid disease, 43% lymphoid, and 4% other disease.
“The most common indications for transplant were acute myeloid leukemia and acute lymphoblastic leukemia,” Wong said, “which, together, formed over 50% of the entire cohort.”
Seventy percent of patients were ECOG status 0 or 1, and 30% were 2 or greater.
Most (59%) had received myeloablative conditioning, the predominant regimens being busulfan/cyclophosphamide or cyclophosphamide/total-body irradiation. And the most common reduced-intensity conditioning regimen was fludarabine/melphalan.
Fifty-six percent of patients had a sibling donor, and 43% had a matched unrelated donor.
Thirty-eight percent of patients developed acute GVHD—19% grade 1, and 19% grade 2 to 3.
“Of note,” Wong said, “no patient developed grade 4 acute GVHD.”
Patients received a median of 2 ZA doses (range, 1–4), including the pre-transplant dose.
Sixty patients received at least 1 dose of ZA between day 100 and day 365, including 33% who received multiple doses.
At day 100, 33 patients received additional ZA. At day 180, 27 patients received additional ZA, including 8 patients who did not have it at day 100. And at day 270, 18 patients had additional ZA, including 1 patient who had no additional ZA at earlier time points.
Results
At day 100, there was no statistically significant change in FN bone density compared with baseline. The mean change was -2.6% (range, -6.6% to 1.4%).
For patients with acute GVHD, however, the change in bone density was significant (P=0.03). Patients with grade 1-2 GVHD had a mean change of -1.6% ± 3.7%, and patients with grade 3-4 GVHD had a mean change of -8.5% ± 11.2%.
Sixty-five patients were available for the day 365 efficacy analysis.
Bone density did not change significantly between day 100 and 365 for the entire group.
“By day 365,” Wong noted, “there was a net loss of bone density of -2.9%.”
But by day 365, patients with extensive chronic GVHD had significantly more bone loss compared with patients who had no chronic GVHD (P=0.03).
Age, sex, duration of cyclosporine, and mean steroid dose were not associated with a change in bone density at day 100 or 365, although there was a trend for an association between high steroid exposure and increased bone loss (P=0.07).
When the researchers compared the patients to untreated historical controls, patients who received ZA had significantly less bone loss at day 100 (P=0.001) and day 365 (P<0.0001).
The researchers observed no serious adverse events with ZA.
Wong concluded that patients with extensive GVHD are a “high-risk cohort that needs augmented therapies.” ![]()
Antithrombotic drugs may raise risk of subdural hematoma
Use of antithrombotic drugs is associated with a higher risk of subdural hematoma, according to a case-control study of more than 400,000 individuals in Denmark.
The highest risk of subdural hematoma was observed in patients who were taking both a vitamin K antagonist (VKA) and clopidogrel.
The study also showed that the incidence of subdural hematoma increased from 2000 to 2015, which appeared to be associated with the increased use of antithrombotic drugs over that time period.
David Gaist, MD, PhD, of Odense University Hospital and the University of Southern Denmark, and his colleagues conducted this research and reported the results in JAMA.
The researchers evaluated 10,010 patients, ages 20 to 89, with a first-ever subdural hematoma diagnosis from 2000 to 2015. The patients were matched to 400,380 individuals from the general population.
Among the patients with subdural hematoma (average age, 69), 47% were taking antithrombotic medications.
The researchers used conditional logistic regression models to estimate the association between antithrombotic drugs and subdural hematoma risk, adjusting for a range of factors. With the following data, they adjusted for age, sex, calendar period, comorbidity, education level, and income level.
The team found that current low-dose aspirin use was associated with a low risk of subdural hematoma (adjusted odds ratio [OR]=1.24).
Current clopidogrel (OR=1.80) and direct oral anticoagulant (OR=1.55) use were both associated with a moderate risk.
And current VKA use was associated with the highest risk of all the agents when given alone (OR=3.69).
Concurrent use of more than one antithrombotic drug was also associated with a high subdural hematoma risk:
- Low-dose aspirin and clopidogrel (OR=2.45)
- Low-dose aspirin and direct oral anticoagulant (OR=2.52)
- Low-dose aspirin and VKA (OR=4.00)
- Clopidogrel and VKA (OR=7.93).
There was one exception to this. Low-dose aspirin combined with the antiplatelet drug dipyridamole was associated with a risk similar to that of low-dose aspirin alone (OR=1.17).
The researchers noted that the prevalence of antithrombotic drug use increased in the general population over the time period studied, from 31.0 per 1000 individuals in 2000 to 76.9 per 1000 individuals in 2015 (P<0.001 for trend).
The overall subdural hematoma incidence rate increased as well, from 10.9 per 100,000 person-years in 2000 to 19.0 per 100,000 person-years in 2015 (P<0.001 for trend).
The largest increase in subdural hematoma incidence was among patients older than 75 years of age—from 55.1 per 100,000 person-years to 99.7 per 100,000 person-years (P<0.001 for trend). ![]()
Use of antithrombotic drugs is associated with a higher risk of subdural hematoma, according to a case-control study of more than 400,000 individuals in Denmark.
The highest risk of subdural hematoma was observed in patients who were taking both a vitamin K antagonist (VKA) and clopidogrel.
The study also showed that the incidence of subdural hematoma increased from 2000 to 2015, which appeared to be associated with the increased use of antithrombotic drugs over that time period.
David Gaist, MD, PhD, of Odense University Hospital and the University of Southern Denmark, and his colleagues conducted this research and reported the results in JAMA.
The researchers evaluated 10,010 patients, ages 20 to 89, with a first-ever subdural hematoma diagnosis from 2000 to 2015. The patients were matched to 400,380 individuals from the general population.
Among the patients with subdural hematoma (average age, 69), 47% were taking antithrombotic medications.
The researchers used conditional logistic regression models to estimate the association between antithrombotic drugs and subdural hematoma risk, adjusting for a range of factors. With the following data, they adjusted for age, sex, calendar period, comorbidity, education level, and income level.
The team found that current low-dose aspirin use was associated with a low risk of subdural hematoma (adjusted odds ratio [OR]=1.24).
Current clopidogrel (OR=1.80) and direct oral anticoagulant (OR=1.55) use were both associated with a moderate risk.
And current VKA use was associated with the highest risk of all the agents when given alone (OR=3.69).
Concurrent use of more than one antithrombotic drug was also associated with a high subdural hematoma risk:
- Low-dose aspirin and clopidogrel (OR=2.45)
- Low-dose aspirin and direct oral anticoagulant (OR=2.52)
- Low-dose aspirin and VKA (OR=4.00)
- Clopidogrel and VKA (OR=7.93).
There was one exception to this. Low-dose aspirin combined with the antiplatelet drug dipyridamole was associated with a risk similar to that of low-dose aspirin alone (OR=1.17).
The researchers noted that the prevalence of antithrombotic drug use increased in the general population over the time period studied, from 31.0 per 1000 individuals in 2000 to 76.9 per 1000 individuals in 2015 (P<0.001 for trend).
The overall subdural hematoma incidence rate increased as well, from 10.9 per 100,000 person-years in 2000 to 19.0 per 100,000 person-years in 2015 (P<0.001 for trend).
The largest increase in subdural hematoma incidence was among patients older than 75 years of age—from 55.1 per 100,000 person-years to 99.7 per 100,000 person-years (P<0.001 for trend). ![]()
Use of antithrombotic drugs is associated with a higher risk of subdural hematoma, according to a case-control study of more than 400,000 individuals in Denmark.
The highest risk of subdural hematoma was observed in patients who were taking both a vitamin K antagonist (VKA) and clopidogrel.
The study also showed that the incidence of subdural hematoma increased from 2000 to 2015, which appeared to be associated with the increased use of antithrombotic drugs over that time period.
David Gaist, MD, PhD, of Odense University Hospital and the University of Southern Denmark, and his colleagues conducted this research and reported the results in JAMA.
The researchers evaluated 10,010 patients, ages 20 to 89, with a first-ever subdural hematoma diagnosis from 2000 to 2015. The patients were matched to 400,380 individuals from the general population.
Among the patients with subdural hematoma (average age, 69), 47% were taking antithrombotic medications.
The researchers used conditional logistic regression models to estimate the association between antithrombotic drugs and subdural hematoma risk, adjusting for a range of factors. With the following data, they adjusted for age, sex, calendar period, comorbidity, education level, and income level.
The team found that current low-dose aspirin use was associated with a low risk of subdural hematoma (adjusted odds ratio [OR]=1.24).
Current clopidogrel (OR=1.80) and direct oral anticoagulant (OR=1.55) use were both associated with a moderate risk.
And current VKA use was associated with the highest risk of all the agents when given alone (OR=3.69).
Concurrent use of more than one antithrombotic drug was also associated with a high subdural hematoma risk:
- Low-dose aspirin and clopidogrel (OR=2.45)
- Low-dose aspirin and direct oral anticoagulant (OR=2.52)
- Low-dose aspirin and VKA (OR=4.00)
- Clopidogrel and VKA (OR=7.93).
There was one exception to this. Low-dose aspirin combined with the antiplatelet drug dipyridamole was associated with a risk similar to that of low-dose aspirin alone (OR=1.17).
The researchers noted that the prevalence of antithrombotic drug use increased in the general population over the time period studied, from 31.0 per 1000 individuals in 2000 to 76.9 per 1000 individuals in 2015 (P<0.001 for trend).
The overall subdural hematoma incidence rate increased as well, from 10.9 per 100,000 person-years in 2000 to 19.0 per 100,000 person-years in 2015 (P<0.001 for trend).
The largest increase in subdural hematoma incidence was among patients older than 75 years of age—from 55.1 per 100,000 person-years to 99.7 per 100,000 person-years (P<0.001 for trend). ![]()
Hemophilia repository open to US scientists
A new resource has been made available for US-based scientists studying hemophilia A and B.
The resource—known as the My Life, Our Future Research Repository—is a collection of genetic data and blood samples that are linked to phenotypic data from more than 5000 hemophilia patients in the US.
Academic and industry scientists within the US can now access the repository but must apply to do so.
Scientists outside of the US should have access to the repository in 2018.
My Life, Our Future is a program founded by the American Thrombosis and Hemostasis Network, Bloodworks Northwest, the National Hemophilia Foundation, and Bioverativ.
My Life, Our Future was launched in 2012 to increase genotyping among individuals with hemophilia A and B, as well as potential and confirmed carriers of the disorder. The program will be available at participating hemophilia treatment centers through the end of 2017.
My Life, Our Future provides hemophilia patients (and potential/confirmed carriers) with free genotyping, and patients can then consent to add their de-identified genetic data and blood sample to the My Life, Our Future Research Repository.
Eighty-three percent of genotyped patients have contributed their data and blood samples. This is more than 5000 individuals representing more than 25% of the US hemophilia population.
The information provided by these individuals has led to the discovery of more than 600 new genetic variants that cause hemophilia.
“Hemophilia is a complex disorder, and, prior to today, there was no genetic and phenotypic database of this scale aiding scientific innovation,” said Barbara Konkle, MD, associate chief scientific officer of Bloodworks Northwest and principal investigator for My Life, Our Future.
“With this new resource, researchers now have one source for the genetic data, repository samples, and clinical data needed to investigate the many unanswered questions about hemophilia.”
Accessing the repository
To access the samples and data in the repository, scientists must first submit a letter of intent to the American Thrombosis and Hemostasis Network.
Scientists may then be asked to submit a full research proposal, which will be evaluated by a research review committee—a group of experts including a molecular pathologist, genetic counselor, research scientist, molecular biologist, genetic epidemiologist, molecular epidemiologist, geneticist, patient representative, and hematologists.
Approved scientists will be selected based on the scientific merit of their proposals and level of benefit to those with bleeding disorders.
For details on how to apply, visit ATHN.org/MLOF. ![]()
A new resource has been made available for US-based scientists studying hemophilia A and B.
The resource—known as the My Life, Our Future Research Repository—is a collection of genetic data and blood samples that are linked to phenotypic data from more than 5000 hemophilia patients in the US.
Academic and industry scientists within the US can now access the repository but must apply to do so.
Scientists outside of the US should have access to the repository in 2018.
My Life, Our Future is a program founded by the American Thrombosis and Hemostasis Network, Bloodworks Northwest, the National Hemophilia Foundation, and Bioverativ.
My Life, Our Future was launched in 2012 to increase genotyping among individuals with hemophilia A and B, as well as potential and confirmed carriers of the disorder. The program will be available at participating hemophilia treatment centers through the end of 2017.
My Life, Our Future provides hemophilia patients (and potential/confirmed carriers) with free genotyping, and patients can then consent to add their de-identified genetic data and blood sample to the My Life, Our Future Research Repository.
Eighty-three percent of genotyped patients have contributed their data and blood samples. This is more than 5000 individuals representing more than 25% of the US hemophilia population.
The information provided by these individuals has led to the discovery of more than 600 new genetic variants that cause hemophilia.
“Hemophilia is a complex disorder, and, prior to today, there was no genetic and phenotypic database of this scale aiding scientific innovation,” said Barbara Konkle, MD, associate chief scientific officer of Bloodworks Northwest and principal investigator for My Life, Our Future.
“With this new resource, researchers now have one source for the genetic data, repository samples, and clinical data needed to investigate the many unanswered questions about hemophilia.”
Accessing the repository
To access the samples and data in the repository, scientists must first submit a letter of intent to the American Thrombosis and Hemostasis Network.
Scientists may then be asked to submit a full research proposal, which will be evaluated by a research review committee—a group of experts including a molecular pathologist, genetic counselor, research scientist, molecular biologist, genetic epidemiologist, molecular epidemiologist, geneticist, patient representative, and hematologists.
Approved scientists will be selected based on the scientific merit of their proposals and level of benefit to those with bleeding disorders.
For details on how to apply, visit ATHN.org/MLOF. ![]()
A new resource has been made available for US-based scientists studying hemophilia A and B.
The resource—known as the My Life, Our Future Research Repository—is a collection of genetic data and blood samples that are linked to phenotypic data from more than 5000 hemophilia patients in the US.
Academic and industry scientists within the US can now access the repository but must apply to do so.
Scientists outside of the US should have access to the repository in 2018.
My Life, Our Future is a program founded by the American Thrombosis and Hemostasis Network, Bloodworks Northwest, the National Hemophilia Foundation, and Bioverativ.
My Life, Our Future was launched in 2012 to increase genotyping among individuals with hemophilia A and B, as well as potential and confirmed carriers of the disorder. The program will be available at participating hemophilia treatment centers through the end of 2017.
My Life, Our Future provides hemophilia patients (and potential/confirmed carriers) with free genotyping, and patients can then consent to add their de-identified genetic data and blood sample to the My Life, Our Future Research Repository.
Eighty-three percent of genotyped patients have contributed their data and blood samples. This is more than 5000 individuals representing more than 25% of the US hemophilia population.
The information provided by these individuals has led to the discovery of more than 600 new genetic variants that cause hemophilia.
“Hemophilia is a complex disorder, and, prior to today, there was no genetic and phenotypic database of this scale aiding scientific innovation,” said Barbara Konkle, MD, associate chief scientific officer of Bloodworks Northwest and principal investigator for My Life, Our Future.
“With this new resource, researchers now have one source for the genetic data, repository samples, and clinical data needed to investigate the many unanswered questions about hemophilia.”
Accessing the repository
To access the samples and data in the repository, scientists must first submit a letter of intent to the American Thrombosis and Hemostasis Network.
Scientists may then be asked to submit a full research proposal, which will be evaluated by a research review committee—a group of experts including a molecular pathologist, genetic counselor, research scientist, molecular biologist, genetic epidemiologist, molecular epidemiologist, geneticist, patient representative, and hematologists.
Approved scientists will be selected based on the scientific merit of their proposals and level of benefit to those with bleeding disorders.
For details on how to apply, visit ATHN.org/MLOF. ![]()
CCSs’ subsequent cancer risk decreased from ’70s to ’90s
Childhood cancer survivors (CCSs) who were diagnosed in the 1990s have a lower risk of subsequent malignancies than CCSs diagnosed in the 1970s, according to research published in JAMA.
The data suggest this outcome is associated with a reduction in the overall use and median dose of therapeutic radiation over time.
Past research has shown an association between therapeutic radiation and the development of subsequent neoplasms in CCSs. Studies have also linked specific chemotherapeutic agents to subsequent neoplasms.
This information has been used to modify childhood cancer treatment over time, with the hope of reducing the risk of subsequent neoplasms while maintaining or improving 5-year survival.
To assess the effects of these treatment modifications, Gregory Armstrong, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted a study of CCSs.
The researchers evaluated 23,603 five-year CCSs (mean age at diagnosis, 7.7 years) treated at pediatric hospitals in the US and Canada from 1970 through 1999, with follow-up through December 2015.
The most common initial diagnoses were acute lymphoblastic leukemia (35.1%), Hodgkin lymphoma (11.1%), and astrocytoma (9.6%).
Subsequent neoplasms, malignancies
At a mean follow-up of 20.5 years, 1639 CCSs had experienced 3115 subsequent neoplasms, including 1026 malignancies, 233 benign meningiomas, and 1856 non-melanoma skin cancers. The most common neoplasms were breast and thyroid cancers.
The 15-year cumulative incidence of subsequent neoplasms decreased by decade of diagnosis. The incidence was 2.9% for patients diagnosed in the 1970s, 2.4% for those diagnosed in the ’80s, and 1.5% for those diagnosed in the ’90s. For the 1970s vs 1980s, the P value was 0.02. For the 1970s vs 1990s and for the 1980s vs 1990s, the P value was <0.001.
The 15-year cumulative incidence of subsequent malignancies also decreased by decade of diagnosis—2.1% for the ’70s, 1.7% for the ’80s, and 1.3% for the ’90s. The P value was <0.001 for the ’70s vs the ’90s and the ’80s vs the ’90s.
Risk factors
In multivariable analyses, female CCSs had a higher rate of subsequent neoplasms (including malignancies) than males.
In addition, high doses of alkylating agents and platinum agents were associated with increased rates of subsequent malignancies.
The researchers noted that, although there was a decrease in the median cumulative dose of alkylating agents over time, the proportion of CCSs receiving these agents increased. And both the median cumulative dose of platinum agents and the proportion of CCSs receiving these agents increased from the ’70s to the ’90s.
Finally, therapeutic radiation was associated with increased rates of subsequent malignant neoplasms, meningiomas, and non-melanoma skin cancers.
This corresponded with the researchers’ findings that the proportion of individuals receiving radiation and the median dose of radiation both decreased over time.
The proportion of individuals receiving any radiation therapy was 77.7% in the ’70s, 56.7% in the ’80s, and 36.8% in the ’90s. The median dose of radiation was 30.0 Gy in the ’70s, 24.0 Gy in the ’80s, and 26.0 Gy in the ’90s.
“The most ominous late effect of pediatric cancer treatment is a second malignancy,” Dr Armstrong said. “This study shows efforts to reduce the late effects of treatment are paying off. The risk of second cancers for survivors increases with age, so it is good to see the reduction emerging early in survivorship while survivors are still young.” ![]()
Childhood cancer survivors (CCSs) who were diagnosed in the 1990s have a lower risk of subsequent malignancies than CCSs diagnosed in the 1970s, according to research published in JAMA.
The data suggest this outcome is associated with a reduction in the overall use and median dose of therapeutic radiation over time.
Past research has shown an association between therapeutic radiation and the development of subsequent neoplasms in CCSs. Studies have also linked specific chemotherapeutic agents to subsequent neoplasms.
This information has been used to modify childhood cancer treatment over time, with the hope of reducing the risk of subsequent neoplasms while maintaining or improving 5-year survival.
To assess the effects of these treatment modifications, Gregory Armstrong, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted a study of CCSs.
The researchers evaluated 23,603 five-year CCSs (mean age at diagnosis, 7.7 years) treated at pediatric hospitals in the US and Canada from 1970 through 1999, with follow-up through December 2015.
The most common initial diagnoses were acute lymphoblastic leukemia (35.1%), Hodgkin lymphoma (11.1%), and astrocytoma (9.6%).
Subsequent neoplasms, malignancies
At a mean follow-up of 20.5 years, 1639 CCSs had experienced 3115 subsequent neoplasms, including 1026 malignancies, 233 benign meningiomas, and 1856 non-melanoma skin cancers. The most common neoplasms were breast and thyroid cancers.
The 15-year cumulative incidence of subsequent neoplasms decreased by decade of diagnosis. The incidence was 2.9% for patients diagnosed in the 1970s, 2.4% for those diagnosed in the ’80s, and 1.5% for those diagnosed in the ’90s. For the 1970s vs 1980s, the P value was 0.02. For the 1970s vs 1990s and for the 1980s vs 1990s, the P value was <0.001.
The 15-year cumulative incidence of subsequent malignancies also decreased by decade of diagnosis—2.1% for the ’70s, 1.7% for the ’80s, and 1.3% for the ’90s. The P value was <0.001 for the ’70s vs the ’90s and the ’80s vs the ’90s.
Risk factors
In multivariable analyses, female CCSs had a higher rate of subsequent neoplasms (including malignancies) than males.
In addition, high doses of alkylating agents and platinum agents were associated with increased rates of subsequent malignancies.
The researchers noted that, although there was a decrease in the median cumulative dose of alkylating agents over time, the proportion of CCSs receiving these agents increased. And both the median cumulative dose of platinum agents and the proportion of CCSs receiving these agents increased from the ’70s to the ’90s.
Finally, therapeutic radiation was associated with increased rates of subsequent malignant neoplasms, meningiomas, and non-melanoma skin cancers.
This corresponded with the researchers’ findings that the proportion of individuals receiving radiation and the median dose of radiation both decreased over time.
The proportion of individuals receiving any radiation therapy was 77.7% in the ’70s, 56.7% in the ’80s, and 36.8% in the ’90s. The median dose of radiation was 30.0 Gy in the ’70s, 24.0 Gy in the ’80s, and 26.0 Gy in the ’90s.
“The most ominous late effect of pediatric cancer treatment is a second malignancy,” Dr Armstrong said. “This study shows efforts to reduce the late effects of treatment are paying off. The risk of second cancers for survivors increases with age, so it is good to see the reduction emerging early in survivorship while survivors are still young.” ![]()
Childhood cancer survivors (CCSs) who were diagnosed in the 1990s have a lower risk of subsequent malignancies than CCSs diagnosed in the 1970s, according to research published in JAMA.
The data suggest this outcome is associated with a reduction in the overall use and median dose of therapeutic radiation over time.
Past research has shown an association between therapeutic radiation and the development of subsequent neoplasms in CCSs. Studies have also linked specific chemotherapeutic agents to subsequent neoplasms.
This information has been used to modify childhood cancer treatment over time, with the hope of reducing the risk of subsequent neoplasms while maintaining or improving 5-year survival.
To assess the effects of these treatment modifications, Gregory Armstrong, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues conducted a study of CCSs.
The researchers evaluated 23,603 five-year CCSs (mean age at diagnosis, 7.7 years) treated at pediatric hospitals in the US and Canada from 1970 through 1999, with follow-up through December 2015.
The most common initial diagnoses were acute lymphoblastic leukemia (35.1%), Hodgkin lymphoma (11.1%), and astrocytoma (9.6%).
Subsequent neoplasms, malignancies
At a mean follow-up of 20.5 years, 1639 CCSs had experienced 3115 subsequent neoplasms, including 1026 malignancies, 233 benign meningiomas, and 1856 non-melanoma skin cancers. The most common neoplasms were breast and thyroid cancers.
The 15-year cumulative incidence of subsequent neoplasms decreased by decade of diagnosis. The incidence was 2.9% for patients diagnosed in the 1970s, 2.4% for those diagnosed in the ’80s, and 1.5% for those diagnosed in the ’90s. For the 1970s vs 1980s, the P value was 0.02. For the 1970s vs 1990s and for the 1980s vs 1990s, the P value was <0.001.
The 15-year cumulative incidence of subsequent malignancies also decreased by decade of diagnosis—2.1% for the ’70s, 1.7% for the ’80s, and 1.3% for the ’90s. The P value was <0.001 for the ’70s vs the ’90s and the ’80s vs the ’90s.
Risk factors
In multivariable analyses, female CCSs had a higher rate of subsequent neoplasms (including malignancies) than males.
In addition, high doses of alkylating agents and platinum agents were associated with increased rates of subsequent malignancies.
The researchers noted that, although there was a decrease in the median cumulative dose of alkylating agents over time, the proportion of CCSs receiving these agents increased. And both the median cumulative dose of platinum agents and the proportion of CCSs receiving these agents increased from the ’70s to the ’90s.
Finally, therapeutic radiation was associated with increased rates of subsequent malignant neoplasms, meningiomas, and non-melanoma skin cancers.
This corresponded with the researchers’ findings that the proportion of individuals receiving radiation and the median dose of radiation both decreased over time.
The proportion of individuals receiving any radiation therapy was 77.7% in the ’70s, 56.7% in the ’80s, and 36.8% in the ’90s. The median dose of radiation was 30.0 Gy in the ’70s, 24.0 Gy in the ’80s, and 26.0 Gy in the ’90s.
“The most ominous late effect of pediatric cancer treatment is a second malignancy,” Dr Armstrong said. “This study shows efforts to reduce the late effects of treatment are paying off. The risk of second cancers for survivors increases with age, so it is good to see the reduction emerging early in survivorship while survivors are still young.” ![]()
March 2017 Digital Edition
Click here to access the March 2017 Digital Edition.
Table of Contents
- Celebrating Federal Social Work!
- FDA Updates
- Screening for Symptomatic Mefloquine Exposure Among Veterans With Chronic Psychiatric Symptoms
- Medication Adherence and OR Efficiency
- Applying a Time-Out and Standardized Report Form in Anesthesia Handoffs
- Oxybutynin Treatment for Hyperhidrosis in Spinal Cord Injury Patients
- Odontogenic Sinusitis
- Generalized Vaccinia After Smallpox Vaccination With Epstein Barr Infection
- Colonic Diaphragm Disease: An Important NSAID Complication to Know
- The Impact of Obesity on the Efficacy of Simvastatin for Lowering LDL-C
- Improving Caregiver Knowledge of Support Resources
- Walter Reed: A Legacy of Leadership and Service

Click here to access the March 2017 Digital Edition.
Table of Contents
- Celebrating Federal Social Work!
- FDA Updates
- Screening for Symptomatic Mefloquine Exposure Among Veterans With Chronic Psychiatric Symptoms
- Medication Adherence and OR Efficiency
- Applying a Time-Out and Standardized Report Form in Anesthesia Handoffs
- Oxybutynin Treatment for Hyperhidrosis in Spinal Cord Injury Patients
- Odontogenic Sinusitis
- Generalized Vaccinia After Smallpox Vaccination With Epstein Barr Infection
- Colonic Diaphragm Disease: An Important NSAID Complication to Know
- The Impact of Obesity on the Efficacy of Simvastatin for Lowering LDL-C
- Improving Caregiver Knowledge of Support Resources
- Walter Reed: A Legacy of Leadership and Service

Click here to access the March 2017 Digital Edition.
Table of Contents
- Celebrating Federal Social Work!
- FDA Updates
- Screening for Symptomatic Mefloquine Exposure Among Veterans With Chronic Psychiatric Symptoms
- Medication Adherence and OR Efficiency
- Applying a Time-Out and Standardized Report Form in Anesthesia Handoffs
- Oxybutynin Treatment for Hyperhidrosis in Spinal Cord Injury Patients
- Odontogenic Sinusitis
- Generalized Vaccinia After Smallpox Vaccination With Epstein Barr Infection
- Colonic Diaphragm Disease: An Important NSAID Complication to Know
- The Impact of Obesity on the Efficacy of Simvastatin for Lowering LDL-C
- Improving Caregiver Knowledge of Support Resources
- Walter Reed: A Legacy of Leadership and Service

A Faster Way to Diagnose Cushing Syndrome
Diagnosing Cushing syndrome (CS) can take 24 hours of complicated and repeated analyses of blood and urine, brain imaging, and tissue samples from sinuses. But that may soon be in the past. The NIH researchers have found that measuring cortisol levels in hair samples can do the same job, faster.
Related: National ALS Biorepository Opens
Patients with CS have a high level of cortisol, perhaps from a tumor of the pituitary or adrenal glands or as an adverse effect from medications. In a study, 36 participants—30 with CS, 6 without—provided hair samples divided into 3 equal segments. The researchers found that the segments closest to the scalp had the most cortisol (96.6 ± 267.7 pg/mg for CS patients, vs 14.1 ± 9.2 pg/mg in control patients). Those segments’ cortisol content correlated closest with the majority of the initial biochemical tests, including in blood taken at night (when cortisol levels normally drop).
Related: Testosterone Replacement Therapy: Playing Catch-up With Patients
The study was small, CS is rare, and it’s hard to recruit large numbers of patients. Still, the researchers believe it is the largest of its kind to compare hair cortisol levels with diagnostic tests in patients with CS. “Our results are encouraging,” said Mihail Zilbermint, MD, the study’s senior author, and an endocrinologist at NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development. “We are hopeful that hair analysis may ultimately prove useful as a less-invasive screening test for Cushing syndrome or in helping to confirm the diagnosis.” The researchers suggest that the test also is a convenient alternative with the “unique ability” for retrospective evaluation of hypercortisolemia over months.
Diagnosing Cushing syndrome (CS) can take 24 hours of complicated and repeated analyses of blood and urine, brain imaging, and tissue samples from sinuses. But that may soon be in the past. The NIH researchers have found that measuring cortisol levels in hair samples can do the same job, faster.
Related: National ALS Biorepository Opens
Patients with CS have a high level of cortisol, perhaps from a tumor of the pituitary or adrenal glands or as an adverse effect from medications. In a study, 36 participants—30 with CS, 6 without—provided hair samples divided into 3 equal segments. The researchers found that the segments closest to the scalp had the most cortisol (96.6 ± 267.7 pg/mg for CS patients, vs 14.1 ± 9.2 pg/mg in control patients). Those segments’ cortisol content correlated closest with the majority of the initial biochemical tests, including in blood taken at night (when cortisol levels normally drop).
Related: Testosterone Replacement Therapy: Playing Catch-up With Patients
The study was small, CS is rare, and it’s hard to recruit large numbers of patients. Still, the researchers believe it is the largest of its kind to compare hair cortisol levels with diagnostic tests in patients with CS. “Our results are encouraging,” said Mihail Zilbermint, MD, the study’s senior author, and an endocrinologist at NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development. “We are hopeful that hair analysis may ultimately prove useful as a less-invasive screening test for Cushing syndrome or in helping to confirm the diagnosis.” The researchers suggest that the test also is a convenient alternative with the “unique ability” for retrospective evaluation of hypercortisolemia over months.
Diagnosing Cushing syndrome (CS) can take 24 hours of complicated and repeated analyses of blood and urine, brain imaging, and tissue samples from sinuses. But that may soon be in the past. The NIH researchers have found that measuring cortisol levels in hair samples can do the same job, faster.
Related: National ALS Biorepository Opens
Patients with CS have a high level of cortisol, perhaps from a tumor of the pituitary or adrenal glands or as an adverse effect from medications. In a study, 36 participants—30 with CS, 6 without—provided hair samples divided into 3 equal segments. The researchers found that the segments closest to the scalp had the most cortisol (96.6 ± 267.7 pg/mg for CS patients, vs 14.1 ± 9.2 pg/mg in control patients). Those segments’ cortisol content correlated closest with the majority of the initial biochemical tests, including in blood taken at night (when cortisol levels normally drop).
Related: Testosterone Replacement Therapy: Playing Catch-up With Patients
The study was small, CS is rare, and it’s hard to recruit large numbers of patients. Still, the researchers believe it is the largest of its kind to compare hair cortisol levels with diagnostic tests in patients with CS. “Our results are encouraging,” said Mihail Zilbermint, MD, the study’s senior author, and an endocrinologist at NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development. “We are hopeful that hair analysis may ultimately prove useful as a less-invasive screening test for Cushing syndrome or in helping to confirm the diagnosis.” The researchers suggest that the test also is a convenient alternative with the “unique ability” for retrospective evaluation of hypercortisolemia over months.
IHS Gives Pharmacy Students Hands-On Experience
The IHS has partnered with 3 top American universities to give pharmacy students an opportunity to get real-life work experience and potentially careers at IHS facilities.
Related: Dangerous Staff Shortages in the IHS
In the IHS Advanced Pharmacy Practice Experience Program, PharmD candidates at Howard University, Purdue University, and the University of Southern California will join students from more than 80 universities in 39 states to complete rotations at IHS direct service facilities. “Many return to start their career in providing quality health care to the American Indian and Alaska Native community,” said Mary Smith, IHS principal deputy director.
“My experience with IHS as a student inspired me to apply to work here when I graduated,” said Fengyee Zhou, now a pharmacist at the IHS Whiteriver Indian Hospital in Arizona. “The level of teamwork among all health care disciplines and the extent to which pharmacists engage in patient care activities brought me back to Whiteriver.”
Related: What s the VA? The Largest Educator of Health Care Professionals in the U.S.
The IHS also offers internships, externships, rotations, and residencies to pharmacy, behavioral health, dentistry, optometry, nursing, and medical students.
The IHS has partnered with 3 top American universities to give pharmacy students an opportunity to get real-life work experience and potentially careers at IHS facilities.
Related: Dangerous Staff Shortages in the IHS
In the IHS Advanced Pharmacy Practice Experience Program, PharmD candidates at Howard University, Purdue University, and the University of Southern California will join students from more than 80 universities in 39 states to complete rotations at IHS direct service facilities. “Many return to start their career in providing quality health care to the American Indian and Alaska Native community,” said Mary Smith, IHS principal deputy director.
“My experience with IHS as a student inspired me to apply to work here when I graduated,” said Fengyee Zhou, now a pharmacist at the IHS Whiteriver Indian Hospital in Arizona. “The level of teamwork among all health care disciplines and the extent to which pharmacists engage in patient care activities brought me back to Whiteriver.”
Related: What s the VA? The Largest Educator of Health Care Professionals in the U.S.
The IHS also offers internships, externships, rotations, and residencies to pharmacy, behavioral health, dentistry, optometry, nursing, and medical students.
The IHS has partnered with 3 top American universities to give pharmacy students an opportunity to get real-life work experience and potentially careers at IHS facilities.
Related: Dangerous Staff Shortages in the IHS
In the IHS Advanced Pharmacy Practice Experience Program, PharmD candidates at Howard University, Purdue University, and the University of Southern California will join students from more than 80 universities in 39 states to complete rotations at IHS direct service facilities. “Many return to start their career in providing quality health care to the American Indian and Alaska Native community,” said Mary Smith, IHS principal deputy director.
“My experience with IHS as a student inspired me to apply to work here when I graduated,” said Fengyee Zhou, now a pharmacist at the IHS Whiteriver Indian Hospital in Arizona. “The level of teamwork among all health care disciplines and the extent to which pharmacists engage in patient care activities brought me back to Whiteriver.”
Related: What s the VA? The Largest Educator of Health Care Professionals in the U.S.
The IHS also offers internships, externships, rotations, and residencies to pharmacy, behavioral health, dentistry, optometry, nursing, and medical students.
Understanding the brexpiprazole therapeutic window: Why more isn’t always better
Dosage windows could be difficult to understand pharmacologically, but for a partial agonist the presumed mechanism could be more evident. Clinicians should be aware that more is not always better, meaning that with partial agonist drugs a higher dosage might not lead to greater patient response. With brexpiprazole, a dopamine D2 partial agonist FDA-approved for schizophrenia and an adjunct for major depressive disorder (MDD),1 moderation is best because of
Recommended dosage
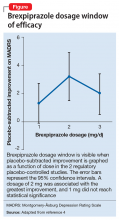
Two placebo-controlled studies2,3 examined brexpiprazole dosages of 1, 2, and 3 mg/d. The recommended dosage of 2 mg/d for MDD was determined by changes in Montgomery-Åsburg Depression Rating Scale scores (Figure).4 Lower dosages of 1 mg/d did not reach statistical significance, and 3 mg/d were less effective than the intermediate dosage of 2 mg/d. This result suggests a window of efficacy for brexpiprazole for MDD. This therapeutic window likely applies to most patients; however, patient-specific variables could alter the optimum dosage.
Dosage window
Brexpiprazole has high affinity for dopamine D2, D3, serotonin 5-HT1A, 5-HT2A, norepinephrine α1B, and α2 Creceptors. At relatively low drug concentrations, brexpiprazole achieves high receptor occupancy. At receptors for which brexpiprazole is a partial agonist (5-HT1A, D2, D3) the drug blocks the receptor and stimulates it at a fraction of the endogenous neurotransmitter. With a very high affinity agent, the endogenous neurotransmitter could be completely excluded from interacting with these receptors if brexpiprazole occupancy is high. At lower dosages, the drug occupies only a fraction of the receptors, allowing the endogenous neurotransmitters to continue interacting with their receptors, thereby magnifying the signal of that receptor above baseline.
1. FDA approves Rexulti (brexpiprazole) as adjunctive treatment for adults with major depressive disorder and as a treatment for adults with schizophrenia [news release]. Valby, Denmark; Tokyo, Japan: H. Lundbeck A/S (Lundbeck); Otsuka Pharmaceutical Co., Ltd; July 11, 2015. http://investor.lundbeck.com/ releasedetail.cfm?Release ID=921621. Accessed October 3, 2015.
2. Thase ME, Youakim JM, Skuban A, et al. Adjunctive brexpiprazole 1 and 3 mg for patients with major depressive disorder following inadequate response to antidepressants: a phase 3, randomized, double-blind study. J Clin Psychiatry. 2015;76(9): 1232-1240.
3. Thase ME, Youakim JM, Skuban A, et al. Efficacy and safety of adjunctive brexpiprazole 2 mg in major depressive disorder: a phase 3, randomized, placebocontrolled study in patients with inadequate response to antidepressants. J Clin Psychiatry. 2015;76(9):1224-1231.
4. Rexulti [package insert]. Rockville, MD: Otsuka; 2015.
Dosage windows could be difficult to understand pharmacologically, but for a partial agonist the presumed mechanism could be more evident. Clinicians should be aware that more is not always better, meaning that with partial agonist drugs a higher dosage might not lead to greater patient response. With brexpiprazole, a dopamine D2 partial agonist FDA-approved for schizophrenia and an adjunct for major depressive disorder (MDD),1 moderation is best because of
Recommended dosage
Two placebo-controlled studies2,3 examined brexpiprazole dosages of 1, 2, and 3 mg/d. The recommended dosage of 2 mg/d for MDD was determined by changes in Montgomery-Åsburg Depression Rating Scale scores (Figure).4 Lower dosages of 1 mg/d did not reach statistical significance, and 3 mg/d were less effective than the intermediate dosage of 2 mg/d. This result suggests a window of efficacy for brexpiprazole for MDD. This therapeutic window likely applies to most patients; however, patient-specific variables could alter the optimum dosage.
Dosage window
Brexpiprazole has high affinity for dopamine D2, D3, serotonin 5-HT1A, 5-HT2A, norepinephrine α1B, and α2 Creceptors. At relatively low drug concentrations, brexpiprazole achieves high receptor occupancy. At receptors for which brexpiprazole is a partial agonist (5-HT1A, D2, D3) the drug blocks the receptor and stimulates it at a fraction of the endogenous neurotransmitter. With a very high affinity agent, the endogenous neurotransmitter could be completely excluded from interacting with these receptors if brexpiprazole occupancy is high. At lower dosages, the drug occupies only a fraction of the receptors, allowing the endogenous neurotransmitters to continue interacting with their receptors, thereby magnifying the signal of that receptor above baseline.
Dosage windows could be difficult to understand pharmacologically, but for a partial agonist the presumed mechanism could be more evident. Clinicians should be aware that more is not always better, meaning that with partial agonist drugs a higher dosage might not lead to greater patient response. With brexpiprazole, a dopamine D2 partial agonist FDA-approved for schizophrenia and an adjunct for major depressive disorder (MDD),1 moderation is best because of
Recommended dosage
Two placebo-controlled studies2,3 examined brexpiprazole dosages of 1, 2, and 3 mg/d. The recommended dosage of 2 mg/d for MDD was determined by changes in Montgomery-Åsburg Depression Rating Scale scores (Figure).4 Lower dosages of 1 mg/d did not reach statistical significance, and 3 mg/d were less effective than the intermediate dosage of 2 mg/d. This result suggests a window of efficacy for brexpiprazole for MDD. This therapeutic window likely applies to most patients; however, patient-specific variables could alter the optimum dosage.
Dosage window
Brexpiprazole has high affinity for dopamine D2, D3, serotonin 5-HT1A, 5-HT2A, norepinephrine α1B, and α2 Creceptors. At relatively low drug concentrations, brexpiprazole achieves high receptor occupancy. At receptors for which brexpiprazole is a partial agonist (5-HT1A, D2, D3) the drug blocks the receptor and stimulates it at a fraction of the endogenous neurotransmitter. With a very high affinity agent, the endogenous neurotransmitter could be completely excluded from interacting with these receptors if brexpiprazole occupancy is high. At lower dosages, the drug occupies only a fraction of the receptors, allowing the endogenous neurotransmitters to continue interacting with their receptors, thereby magnifying the signal of that receptor above baseline.
1. FDA approves Rexulti (brexpiprazole) as adjunctive treatment for adults with major depressive disorder and as a treatment for adults with schizophrenia [news release]. Valby, Denmark; Tokyo, Japan: H. Lundbeck A/S (Lundbeck); Otsuka Pharmaceutical Co., Ltd; July 11, 2015. http://investor.lundbeck.com/ releasedetail.cfm?Release ID=921621. Accessed October 3, 2015.
2. Thase ME, Youakim JM, Skuban A, et al. Adjunctive brexpiprazole 1 and 3 mg for patients with major depressive disorder following inadequate response to antidepressants: a phase 3, randomized, double-blind study. J Clin Psychiatry. 2015;76(9): 1232-1240.
3. Thase ME, Youakim JM, Skuban A, et al. Efficacy and safety of adjunctive brexpiprazole 2 mg in major depressive disorder: a phase 3, randomized, placebocontrolled study in patients with inadequate response to antidepressants. J Clin Psychiatry. 2015;76(9):1224-1231.
4. Rexulti [package insert]. Rockville, MD: Otsuka; 2015.
1. FDA approves Rexulti (brexpiprazole) as adjunctive treatment for adults with major depressive disorder and as a treatment for adults with schizophrenia [news release]. Valby, Denmark; Tokyo, Japan: H. Lundbeck A/S (Lundbeck); Otsuka Pharmaceutical Co., Ltd; July 11, 2015. http://investor.lundbeck.com/ releasedetail.cfm?Release ID=921621. Accessed October 3, 2015.
2. Thase ME, Youakim JM, Skuban A, et al. Adjunctive brexpiprazole 1 and 3 mg for patients with major depressive disorder following inadequate response to antidepressants: a phase 3, randomized, double-blind study. J Clin Psychiatry. 2015;76(9): 1232-1240.
3. Thase ME, Youakim JM, Skuban A, et al. Efficacy and safety of adjunctive brexpiprazole 2 mg in major depressive disorder: a phase 3, randomized, placebocontrolled study in patients with inadequate response to antidepressants. J Clin Psychiatry. 2015;76(9):1224-1231.
4. Rexulti [package insert]. Rockville, MD: Otsuka; 2015.