User login
Xyloglucan-pea protein a possible steroid-sparing alternative for treating pediatric AD
Key clinical point: Xyloglucan and pea protein (XG-PP)-based topical treatment shows safety and efficacy outcomes comparable with those of hydrocortisone in pediatric patients with atopic dermatitis (AD).
Major finding: At 8 and 15 days of treatment, both XG-PP and hydrocortisone led to significant decreases in the AD Severity Index (ADSI) score (all P = .00001). Both treatment arms showed similar decrease in ADSI scores at 8 (P = .91) and 15 (P = .92) days. No adverse events were reported in the XG-PP treatment arm.
Study details: Findings are from a prospective multicenter study including 42 pediatric patients with mild-to-moderate AD (age 6 months-12 years) who were randomly assigned to receive either topical XG-PP-based cream or hydrocortisone twice daily for 14 consecutive days.
Disclosures: This study was sponsored by Novintethical Pharma SA. The authors declared no conflicts of interest.
Source: Sowlati M et al. Efficacy and tolerability of a novel topical treatment containing pea protein and xyloglucan in the management of atopic dermatitis in children: A prospective, multicenter clinical study. Dermatol Ther (Heidelb). 2023 (Sep 23). doi: 10.1007/s13555-023-01035-6
Key clinical point: Xyloglucan and pea protein (XG-PP)-based topical treatment shows safety and efficacy outcomes comparable with those of hydrocortisone in pediatric patients with atopic dermatitis (AD).
Major finding: At 8 and 15 days of treatment, both XG-PP and hydrocortisone led to significant decreases in the AD Severity Index (ADSI) score (all P = .00001). Both treatment arms showed similar decrease in ADSI scores at 8 (P = .91) and 15 (P = .92) days. No adverse events were reported in the XG-PP treatment arm.
Study details: Findings are from a prospective multicenter study including 42 pediatric patients with mild-to-moderate AD (age 6 months-12 years) who were randomly assigned to receive either topical XG-PP-based cream or hydrocortisone twice daily for 14 consecutive days.
Disclosures: This study was sponsored by Novintethical Pharma SA. The authors declared no conflicts of interest.
Source: Sowlati M et al. Efficacy and tolerability of a novel topical treatment containing pea protein and xyloglucan in the management of atopic dermatitis in children: A prospective, multicenter clinical study. Dermatol Ther (Heidelb). 2023 (Sep 23). doi: 10.1007/s13555-023-01035-6
Key clinical point: Xyloglucan and pea protein (XG-PP)-based topical treatment shows safety and efficacy outcomes comparable with those of hydrocortisone in pediatric patients with atopic dermatitis (AD).
Major finding: At 8 and 15 days of treatment, both XG-PP and hydrocortisone led to significant decreases in the AD Severity Index (ADSI) score (all P = .00001). Both treatment arms showed similar decrease in ADSI scores at 8 (P = .91) and 15 (P = .92) days. No adverse events were reported in the XG-PP treatment arm.
Study details: Findings are from a prospective multicenter study including 42 pediatric patients with mild-to-moderate AD (age 6 months-12 years) who were randomly assigned to receive either topical XG-PP-based cream or hydrocortisone twice daily for 14 consecutive days.
Disclosures: This study was sponsored by Novintethical Pharma SA. The authors declared no conflicts of interest.
Source: Sowlati M et al. Efficacy and tolerability of a novel topical treatment containing pea protein and xyloglucan in the management of atopic dermatitis in children: A prospective, multicenter clinical study. Dermatol Ther (Heidelb). 2023 (Sep 23). doi: 10.1007/s13555-023-01035-6
Increased risk for neuropsychiatric disorders in adults with AD
Key clinical point: Patients with atopic dermatitis (AD) have an increased risk for multiple neuropsychiatric conditions; however, the risk profiles for specific neuropsychiatric conditions differ with AD severity.
Major finding: Adults with AD (of any severity level) vs without AD had a higher risk for anxiety (adjusted hazard ratio [aHR] 1.14, 95% CI 1.13-1.15), depression (aHR 1.14, 95% CI 1.13-1.15), and obsessive-compulsive disorder (aHR 1.48, 95% CI 1.38-1.58); the risk for autism increased in patients with mild (aHR 1.55; 95% CI 1.26-1.89) and moderate (aHR 1.40; 95% CI 1.07-1.83) AD and that for attention-deficit/hyperactivity disorder increased in those with mild AD (aHR 1.27; 95% CI 1.03-1.55].
Study details: This population-based cohort study included 625,083 adults with AD who were matched with 2,678,888 control adults without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. Some authors declared receiving research or fellowship funding or consultation honoraria from various sources, including Pfizer. AR Lemeshow declared being an employee of Pfizer.
Source: Wan J et al. Neuropsychiatric disorders in adults with atopic dermatitis: A population-based cohort study. J Eur Acad Dermatol Venereol. 2023 (Sep 20). doi: 10.1111/jdv.19518
Key clinical point: Patients with atopic dermatitis (AD) have an increased risk for multiple neuropsychiatric conditions; however, the risk profiles for specific neuropsychiatric conditions differ with AD severity.
Major finding: Adults with AD (of any severity level) vs without AD had a higher risk for anxiety (adjusted hazard ratio [aHR] 1.14, 95% CI 1.13-1.15), depression (aHR 1.14, 95% CI 1.13-1.15), and obsessive-compulsive disorder (aHR 1.48, 95% CI 1.38-1.58); the risk for autism increased in patients with mild (aHR 1.55; 95% CI 1.26-1.89) and moderate (aHR 1.40; 95% CI 1.07-1.83) AD and that for attention-deficit/hyperactivity disorder increased in those with mild AD (aHR 1.27; 95% CI 1.03-1.55].
Study details: This population-based cohort study included 625,083 adults with AD who were matched with 2,678,888 control adults without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. Some authors declared receiving research or fellowship funding or consultation honoraria from various sources, including Pfizer. AR Lemeshow declared being an employee of Pfizer.
Source: Wan J et al. Neuropsychiatric disorders in adults with atopic dermatitis: A population-based cohort study. J Eur Acad Dermatol Venereol. 2023 (Sep 20). doi: 10.1111/jdv.19518
Key clinical point: Patients with atopic dermatitis (AD) have an increased risk for multiple neuropsychiatric conditions; however, the risk profiles for specific neuropsychiatric conditions differ with AD severity.
Major finding: Adults with AD (of any severity level) vs without AD had a higher risk for anxiety (adjusted hazard ratio [aHR] 1.14, 95% CI 1.13-1.15), depression (aHR 1.14, 95% CI 1.13-1.15), and obsessive-compulsive disorder (aHR 1.48, 95% CI 1.38-1.58); the risk for autism increased in patients with mild (aHR 1.55; 95% CI 1.26-1.89) and moderate (aHR 1.40; 95% CI 1.07-1.83) AD and that for attention-deficit/hyperactivity disorder increased in those with mild AD (aHR 1.27; 95% CI 1.03-1.55].
Study details: This population-based cohort study included 625,083 adults with AD who were matched with 2,678,888 control adults without AD.
Disclosures: This study was supported by a contract from Pfizer, Inc. Some authors declared receiving research or fellowship funding or consultation honoraria from various sources, including Pfizer. AR Lemeshow declared being an employee of Pfizer.
Source: Wan J et al. Neuropsychiatric disorders in adults with atopic dermatitis: A population-based cohort study. J Eur Acad Dermatol Venereol. 2023 (Sep 20). doi: 10.1111/jdv.19518
Methotrexate is a safe and efficacious alternative to ciclosporin in children with severe AD
Key clinical point: Both ciclosporin and methotrexate were effective against severe atopic dermatitis (AD) in children, but ciclosporin resulted in a more rapid response whereas methotrexate led to more sustained disease control even after treatment discontinuation.
Major finding: At 12 weeks, a significantly higher proportion of patients achieved 50% improvement in Objective Severity Scoring of Atopic Dermatitis scores (o-SCORAD-50) with ciclosporin vs methotrexate (P = .012). However, at 60 weeks, the proportion of patients who achieved o-SCORAD-50 was higher with methotrexate vs ciclosporin (P = .022). Adverse event rates were comparable in both groups.
Study details: The TREatment of severe Atopic Eczema Trial included 103 children with severe AD (age 2-16 years) who were unresponsive to topical treatments and were randomly assigned to receive ciclosporin or methotrexate.
Disclosures: This study was funded by the UK Medical Research Council/National Institute for Health Research (NIHR). Some authors, including the lead author, declared receiving consulting fees, advisory fees, or research funding from various sources, including UK NIHR.
Source: Flohr C et al and the TREAT Trial Investigators. Efficacy and safety of ciclosporin versus methotrexate in the treatment of severe atopic dermatitis in children and young people (TREAT): A multicentre, parallel group, assessor-blinded clinical trial. Br J Dermatol. 2023 (Sep 19). doi: 10.1093/bjd/ljad281
Key clinical point: Both ciclosporin and methotrexate were effective against severe atopic dermatitis (AD) in children, but ciclosporin resulted in a more rapid response whereas methotrexate led to more sustained disease control even after treatment discontinuation.
Major finding: At 12 weeks, a significantly higher proportion of patients achieved 50% improvement in Objective Severity Scoring of Atopic Dermatitis scores (o-SCORAD-50) with ciclosporin vs methotrexate (P = .012). However, at 60 weeks, the proportion of patients who achieved o-SCORAD-50 was higher with methotrexate vs ciclosporin (P = .022). Adverse event rates were comparable in both groups.
Study details: The TREatment of severe Atopic Eczema Trial included 103 children with severe AD (age 2-16 years) who were unresponsive to topical treatments and were randomly assigned to receive ciclosporin or methotrexate.
Disclosures: This study was funded by the UK Medical Research Council/National Institute for Health Research (NIHR). Some authors, including the lead author, declared receiving consulting fees, advisory fees, or research funding from various sources, including UK NIHR.
Source: Flohr C et al and the TREAT Trial Investigators. Efficacy and safety of ciclosporin versus methotrexate in the treatment of severe atopic dermatitis in children and young people (TREAT): A multicentre, parallel group, assessor-blinded clinical trial. Br J Dermatol. 2023 (Sep 19). doi: 10.1093/bjd/ljad281
Key clinical point: Both ciclosporin and methotrexate were effective against severe atopic dermatitis (AD) in children, but ciclosporin resulted in a more rapid response whereas methotrexate led to more sustained disease control even after treatment discontinuation.
Major finding: At 12 weeks, a significantly higher proportion of patients achieved 50% improvement in Objective Severity Scoring of Atopic Dermatitis scores (o-SCORAD-50) with ciclosporin vs methotrexate (P = .012). However, at 60 weeks, the proportion of patients who achieved o-SCORAD-50 was higher with methotrexate vs ciclosporin (P = .022). Adverse event rates were comparable in both groups.
Study details: The TREatment of severe Atopic Eczema Trial included 103 children with severe AD (age 2-16 years) who were unresponsive to topical treatments and were randomly assigned to receive ciclosporin or methotrexate.
Disclosures: This study was funded by the UK Medical Research Council/National Institute for Health Research (NIHR). Some authors, including the lead author, declared receiving consulting fees, advisory fees, or research funding from various sources, including UK NIHR.
Source: Flohr C et al and the TREAT Trial Investigators. Efficacy and safety of ciclosporin versus methotrexate in the treatment of severe atopic dermatitis in children and young people (TREAT): A multicentre, parallel group, assessor-blinded clinical trial. Br J Dermatol. 2023 (Sep 19). doi: 10.1093/bjd/ljad281
Children with atopic dermatitis more prone to allergic contact dermatitis
Key clinical point: Compared with children without atopic dermatitis (AD), those with AD are significantly more likely to have positive patch tests (PPT) and respond to ≥1 allergen on patch testing.
Major finding: Children with vs without AD were significantly more likely to have a longer duration of dermatitis (P < .0001), >1 PPT result (P = .005), a greater number of PPT overall (P = .012), and a more generalized distribution of dermatitis (P = .001) as well as PPT to bacitracin (P = .030), carba mix (diphenylguanidine, zinc dibutyldithiocarbamate, and zinc diethyldithiocarbamate) (P = .025), and cocamidopropyl betaine (P = .0007).
Study details: This retrospective case-control study included 615 children with AD and 297 children without AD.
Disclosures: This study was supported by the Dermatology Foundation, Evanston, IL. The authors declared no conflicts of interest.
Source: Johnson H et al. Prevalence of allergic contact dermatitis in children with and without atopic dermatitis: A multicenter retrospective case-control study. J Am Acad Dermatol. 2023;89(5):1007-1014 (Sep 25). doi: 10.1016/j.jaad.2023.06.048
Key clinical point: Compared with children without atopic dermatitis (AD), those with AD are significantly more likely to have positive patch tests (PPT) and respond to ≥1 allergen on patch testing.
Major finding: Children with vs without AD were significantly more likely to have a longer duration of dermatitis (P < .0001), >1 PPT result (P = .005), a greater number of PPT overall (P = .012), and a more generalized distribution of dermatitis (P = .001) as well as PPT to bacitracin (P = .030), carba mix (diphenylguanidine, zinc dibutyldithiocarbamate, and zinc diethyldithiocarbamate) (P = .025), and cocamidopropyl betaine (P = .0007).
Study details: This retrospective case-control study included 615 children with AD and 297 children without AD.
Disclosures: This study was supported by the Dermatology Foundation, Evanston, IL. The authors declared no conflicts of interest.
Source: Johnson H et al. Prevalence of allergic contact dermatitis in children with and without atopic dermatitis: A multicenter retrospective case-control study. J Am Acad Dermatol. 2023;89(5):1007-1014 (Sep 25). doi: 10.1016/j.jaad.2023.06.048
Key clinical point: Compared with children without atopic dermatitis (AD), those with AD are significantly more likely to have positive patch tests (PPT) and respond to ≥1 allergen on patch testing.
Major finding: Children with vs without AD were significantly more likely to have a longer duration of dermatitis (P < .0001), >1 PPT result (P = .005), a greater number of PPT overall (P = .012), and a more generalized distribution of dermatitis (P = .001) as well as PPT to bacitracin (P = .030), carba mix (diphenylguanidine, zinc dibutyldithiocarbamate, and zinc diethyldithiocarbamate) (P = .025), and cocamidopropyl betaine (P = .0007).
Study details: This retrospective case-control study included 615 children with AD and 297 children without AD.
Disclosures: This study was supported by the Dermatology Foundation, Evanston, IL. The authors declared no conflicts of interest.
Source: Johnson H et al. Prevalence of allergic contact dermatitis in children with and without atopic dermatitis: A multicenter retrospective case-control study. J Am Acad Dermatol. 2023;89(5):1007-1014 (Sep 25). doi: 10.1016/j.jaad.2023.06.048
Maternal perinatal mortality: A pediatric issue
Checking on the well-being of mothers is one of the important acknowledged aspects of primary pediatric care. “How are you doing?” directed to the child’s mother has long been considered an appropriate question. The AAP recommends several checks in the Bright Futures Guidelines, including conducting several formal screens for depression and asking about “getting time alone with your partner” as well as other supports.
But I have recently become aware of new data that changes my ideas about what we pediatricians need to be doing as part of our care for children and their families, especially in the first year: Considering the risks to the mother of dying.
Maternal mortality increased by 26.6% from 2000 to 2014 across the United States such that it is higher now than it was for our own mothers. The U.S. now has the highest rates of maternal mortality among high-income nations, especially for Black, American Indian, or Alaska Native women, those of lower socioeconomic status, and those under 18 or over 35 years old.
You may be thinking, well, that is an issue for ob.gyns. Indeed, the most common reasons for maternal death are cardiovascular: hemorrhage, hypertensive disorders, deep vein thrombosis, and stroke, all usually occurring at or in the first week after birth. You may have heard about sudden unexpected heart failure from postpartum cardiomyopathy, although rare (1 in 1,000-4,000), presenting from 1 month pre birth to 5 months post delivery, which is when we may be the main clinicians seeing the mother, not the ob.gyns. This can be easily missed since it presents with shortness of breath and decreased exercise tolerance, fatigue, palpitations, and/or leg swelling. Serious eclampsia may have only symptoms of headache or abdominal pain. All of these may easily be mistaken for lingering pregnancy symptoms. But in higher income countries, such as the U.S., 38% of maternal deaths occur from 8 to 42 days after birth, the period for fatal infections as well as cardiac complications. Elevated risk for all of these causes of mortality include Black race, obesity, tobacco use, congenital heart disease, and being older than 40.
As pediatric providers, we may see mothers along with their infants as newborns in the hospital, at day 2, at 2 weeks, or even at 1-2 months after birth, potentially before their one recommended postnatal obstetric visit at 3-8 weeks. Asking the mother how she is feeling at those times should not just be a social nicety but rather an additional check for serious postnatal complications.
Additional concerns
But wait, it gets worse.
Did you know that the leading cause of maternal death from pregnancy up to 1 year after a birth is homicide?
Maternal perinatal mortality figures have not usually included “perinatal-associated” deaths, a maternal death attributable to a condition that is unaffected by the pregnancy and occurring within 1 year of delivery (that I will cite as perinatal henceforth). While half of maternal deaths occur during pregnancy, another half occur in the year following. There were 3.62 homicides per 100,000 live births among females who were pregnant or within 1 year postpartum, 16% more than for similarly aged nonpregnant and nonpostpartum women (3.12 deaths/100,000 population, P < .05). Homicides made up 8.4% of reported perinatal maternal deaths from all causes, with a rate of 1.7 per 100,000 live births, twice the rate of any one of the other leading causes noted above. Black women had seven times the risk of perinatal homicide as that of White women. Females under 20, many of them our own pediatric patients, had a greater than six times higher risk and those aged 20-24 had a 65% higher risk of pregnancy-associated homicide across race and ethnic groups. Homicide is most likely before 21 weeks of pregnancy, decreases in the third trimester, but increases again after birth. Two-thirds of pregnancy-associated homicide deaths occurred in the home, with the perpetrator a current or prior partner (> 59%, with 98% being male), 45%-50% were associated with reported intimate partner violence (IPV), and the most common method was a firearm (55%). Often the same women had histories of substance abuse, serious mental illness, and/or prior IPV, all risk factors for pregnancy-associated deaths, including from homicide.
Homicide? “Not the mothers in my practice,” you may say, but, if not homicide, drug-related deaths (3.68 per 100,000 person-years) and suicide (1.42 per 100,000 person-years) together comprise 18% of all maternal deaths. Non-Hispanic White women, Medicaid-insured women, and women residing in smaller cities were especially likely to die from drugs or suicide. More than half (54.3%) of perinatal suicides involve intimate partner conflict, which increases the risk ninefold. Perinatal mood disorders, affecting up to 15% of pregnant and postpartum U.S. women, is also a risk factor in substance abuse, opioid overdose death, and suicide.
And substance use has gotten more dangerous with the increase in fentanyl lacing. Pregnancy-associated deaths (4%-10% of deaths) involving opioids more than doubled between 2007 and 2016, and, although the rates are higher for Black women, the increase has been greater for non-Hispanic White women. Two-thirds of those deaths occur between 6 and 12 months postpartum, on our watch. Although many women decrease substance use during pregnancy, they may fall back into substance use (rates increase 4 times by 7-12 months after delivery) and not continue to receive treatment. Although pharmacotherapy (e.g., methadone, buprenorphine treatment) is the current standard of care for opioid use disorder (OUD) during pregnancy, nearly half receiving treatment in publicly funded centers are not receiving these medications and others may lose insurance or access to pregnancy-related treatment programs after delivery, increasing risk of relapse. Stigma, and punitive or discriminatory approaches to pregnant women with OUD (e.g., jail, removal of children) can dissuade them from participating in treatment, increasing overdose risk.
It is important to note that in more than half of the 41 deaths from violent trauma in one study (including 22 homicides), obstetrical providers knew of or suspected IPV. Also, the vast majority (74%) of those who died by drugs or suicide had made one or more emergency department or hospital visit between their delivery and death, and 39% had made three or more visits. Without knowing if anything was done in those cases, we also know that, in addition to thorough, compassionate providers, there is sometimes segmentation of responsibility, insensitivity, discrimination, racism, stigma, inequity, lack of resources, lack of access, lack of payment mechanisms, legal issues for immigrants, time constraints, and other systemic deficits that may hinder effective care for these and subsequent women.
Awareness and action
What should we, who are primary care pediatric providers, do about these threats to the mothers and pregnant young women we care for? Clearly, their children, our main patients, would be terribly and permanently hurt by harm coming to their mothers – the extreme adverse childhood experiences and social determinants of health to which we are already committed.
I hope this article will help alert pediatric providers to what is being published, mainly as women’s health and public health issues.
First, we need awareness of the physical symptoms that may come up in our interactions with pregnant and postpartum women so that we can educate them and expedite any indicated emergency care.
Next, we need to expand our routine screening of mothers and pregnant women from just the most impactful social determinants of health (including depression, substance use, and IPV) to include anxiety, past suicide attempts and current suicidal ideation, and the presence of firearms, early and repeatedly in the first year of the child’s life. Adults and teens are more likely to disclose risk for sensitive issues through questionnaires than through interviews, perhaps even more so when the identified patient is their child rather than themselves. Any screen can have false negatives, so asking directly when risk is suspected is important. The reason for screening could be framed as caring for the caregiver who is the most important person for the child. It could be accompanied by acknowledging that pregnancy and the first year of life can be difficult for mothers and their partners and that we want to support them and connect them to resources, if needed. When substance use disorder is acknowledged, we should prescribe and teach about Narcan for overdose. When there is IPV, we should discuss firearm removal/locking as well as counseling on a personal safety plan.
Working as part of an on-site or virtual team that includes professionals who know about community resources and can coordinate care is essential, in addition to educating about 211 for services and 988 for suicide risk.
Finally, we can advocate and vote for programs, people, and laws that support and safeguard women and families, address substance use, and reduce access to firearms.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
Checking on the well-being of mothers is one of the important acknowledged aspects of primary pediatric care. “How are you doing?” directed to the child’s mother has long been considered an appropriate question. The AAP recommends several checks in the Bright Futures Guidelines, including conducting several formal screens for depression and asking about “getting time alone with your partner” as well as other supports.
But I have recently become aware of new data that changes my ideas about what we pediatricians need to be doing as part of our care for children and their families, especially in the first year: Considering the risks to the mother of dying.
Maternal mortality increased by 26.6% from 2000 to 2014 across the United States such that it is higher now than it was for our own mothers. The U.S. now has the highest rates of maternal mortality among high-income nations, especially for Black, American Indian, or Alaska Native women, those of lower socioeconomic status, and those under 18 or over 35 years old.
You may be thinking, well, that is an issue for ob.gyns. Indeed, the most common reasons for maternal death are cardiovascular: hemorrhage, hypertensive disorders, deep vein thrombosis, and stroke, all usually occurring at or in the first week after birth. You may have heard about sudden unexpected heart failure from postpartum cardiomyopathy, although rare (1 in 1,000-4,000), presenting from 1 month pre birth to 5 months post delivery, which is when we may be the main clinicians seeing the mother, not the ob.gyns. This can be easily missed since it presents with shortness of breath and decreased exercise tolerance, fatigue, palpitations, and/or leg swelling. Serious eclampsia may have only symptoms of headache or abdominal pain. All of these may easily be mistaken for lingering pregnancy symptoms. But in higher income countries, such as the U.S., 38% of maternal deaths occur from 8 to 42 days after birth, the period for fatal infections as well as cardiac complications. Elevated risk for all of these causes of mortality include Black race, obesity, tobacco use, congenital heart disease, and being older than 40.
As pediatric providers, we may see mothers along with their infants as newborns in the hospital, at day 2, at 2 weeks, or even at 1-2 months after birth, potentially before their one recommended postnatal obstetric visit at 3-8 weeks. Asking the mother how she is feeling at those times should not just be a social nicety but rather an additional check for serious postnatal complications.
Additional concerns
But wait, it gets worse.
Did you know that the leading cause of maternal death from pregnancy up to 1 year after a birth is homicide?
Maternal perinatal mortality figures have not usually included “perinatal-associated” deaths, a maternal death attributable to a condition that is unaffected by the pregnancy and occurring within 1 year of delivery (that I will cite as perinatal henceforth). While half of maternal deaths occur during pregnancy, another half occur in the year following. There were 3.62 homicides per 100,000 live births among females who were pregnant or within 1 year postpartum, 16% more than for similarly aged nonpregnant and nonpostpartum women (3.12 deaths/100,000 population, P < .05). Homicides made up 8.4% of reported perinatal maternal deaths from all causes, with a rate of 1.7 per 100,000 live births, twice the rate of any one of the other leading causes noted above. Black women had seven times the risk of perinatal homicide as that of White women. Females under 20, many of them our own pediatric patients, had a greater than six times higher risk and those aged 20-24 had a 65% higher risk of pregnancy-associated homicide across race and ethnic groups. Homicide is most likely before 21 weeks of pregnancy, decreases in the third trimester, but increases again after birth. Two-thirds of pregnancy-associated homicide deaths occurred in the home, with the perpetrator a current or prior partner (> 59%, with 98% being male), 45%-50% were associated with reported intimate partner violence (IPV), and the most common method was a firearm (55%). Often the same women had histories of substance abuse, serious mental illness, and/or prior IPV, all risk factors for pregnancy-associated deaths, including from homicide.
Homicide? “Not the mothers in my practice,” you may say, but, if not homicide, drug-related deaths (3.68 per 100,000 person-years) and suicide (1.42 per 100,000 person-years) together comprise 18% of all maternal deaths. Non-Hispanic White women, Medicaid-insured women, and women residing in smaller cities were especially likely to die from drugs or suicide. More than half (54.3%) of perinatal suicides involve intimate partner conflict, which increases the risk ninefold. Perinatal mood disorders, affecting up to 15% of pregnant and postpartum U.S. women, is also a risk factor in substance abuse, opioid overdose death, and suicide.
And substance use has gotten more dangerous with the increase in fentanyl lacing. Pregnancy-associated deaths (4%-10% of deaths) involving opioids more than doubled between 2007 and 2016, and, although the rates are higher for Black women, the increase has been greater for non-Hispanic White women. Two-thirds of those deaths occur between 6 and 12 months postpartum, on our watch. Although many women decrease substance use during pregnancy, they may fall back into substance use (rates increase 4 times by 7-12 months after delivery) and not continue to receive treatment. Although pharmacotherapy (e.g., methadone, buprenorphine treatment) is the current standard of care for opioid use disorder (OUD) during pregnancy, nearly half receiving treatment in publicly funded centers are not receiving these medications and others may lose insurance or access to pregnancy-related treatment programs after delivery, increasing risk of relapse. Stigma, and punitive or discriminatory approaches to pregnant women with OUD (e.g., jail, removal of children) can dissuade them from participating in treatment, increasing overdose risk.
It is important to note that in more than half of the 41 deaths from violent trauma in one study (including 22 homicides), obstetrical providers knew of or suspected IPV. Also, the vast majority (74%) of those who died by drugs or suicide had made one or more emergency department or hospital visit between their delivery and death, and 39% had made three or more visits. Without knowing if anything was done in those cases, we also know that, in addition to thorough, compassionate providers, there is sometimes segmentation of responsibility, insensitivity, discrimination, racism, stigma, inequity, lack of resources, lack of access, lack of payment mechanisms, legal issues for immigrants, time constraints, and other systemic deficits that may hinder effective care for these and subsequent women.
Awareness and action
What should we, who are primary care pediatric providers, do about these threats to the mothers and pregnant young women we care for? Clearly, their children, our main patients, would be terribly and permanently hurt by harm coming to their mothers – the extreme adverse childhood experiences and social determinants of health to which we are already committed.
I hope this article will help alert pediatric providers to what is being published, mainly as women’s health and public health issues.
First, we need awareness of the physical symptoms that may come up in our interactions with pregnant and postpartum women so that we can educate them and expedite any indicated emergency care.
Next, we need to expand our routine screening of mothers and pregnant women from just the most impactful social determinants of health (including depression, substance use, and IPV) to include anxiety, past suicide attempts and current suicidal ideation, and the presence of firearms, early and repeatedly in the first year of the child’s life. Adults and teens are more likely to disclose risk for sensitive issues through questionnaires than through interviews, perhaps even more so when the identified patient is their child rather than themselves. Any screen can have false negatives, so asking directly when risk is suspected is important. The reason for screening could be framed as caring for the caregiver who is the most important person for the child. It could be accompanied by acknowledging that pregnancy and the first year of life can be difficult for mothers and their partners and that we want to support them and connect them to resources, if needed. When substance use disorder is acknowledged, we should prescribe and teach about Narcan for overdose. When there is IPV, we should discuss firearm removal/locking as well as counseling on a personal safety plan.
Working as part of an on-site or virtual team that includes professionals who know about community resources and can coordinate care is essential, in addition to educating about 211 for services and 988 for suicide risk.
Finally, we can advocate and vote for programs, people, and laws that support and safeguard women and families, address substance use, and reduce access to firearms.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
Checking on the well-being of mothers is one of the important acknowledged aspects of primary pediatric care. “How are you doing?” directed to the child’s mother has long been considered an appropriate question. The AAP recommends several checks in the Bright Futures Guidelines, including conducting several formal screens for depression and asking about “getting time alone with your partner” as well as other supports.
But I have recently become aware of new data that changes my ideas about what we pediatricians need to be doing as part of our care for children and their families, especially in the first year: Considering the risks to the mother of dying.
Maternal mortality increased by 26.6% from 2000 to 2014 across the United States such that it is higher now than it was for our own mothers. The U.S. now has the highest rates of maternal mortality among high-income nations, especially for Black, American Indian, or Alaska Native women, those of lower socioeconomic status, and those under 18 or over 35 years old.
You may be thinking, well, that is an issue for ob.gyns. Indeed, the most common reasons for maternal death are cardiovascular: hemorrhage, hypertensive disorders, deep vein thrombosis, and stroke, all usually occurring at or in the first week after birth. You may have heard about sudden unexpected heart failure from postpartum cardiomyopathy, although rare (1 in 1,000-4,000), presenting from 1 month pre birth to 5 months post delivery, which is when we may be the main clinicians seeing the mother, not the ob.gyns. This can be easily missed since it presents with shortness of breath and decreased exercise tolerance, fatigue, palpitations, and/or leg swelling. Serious eclampsia may have only symptoms of headache or abdominal pain. All of these may easily be mistaken for lingering pregnancy symptoms. But in higher income countries, such as the U.S., 38% of maternal deaths occur from 8 to 42 days after birth, the period for fatal infections as well as cardiac complications. Elevated risk for all of these causes of mortality include Black race, obesity, tobacco use, congenital heart disease, and being older than 40.
As pediatric providers, we may see mothers along with their infants as newborns in the hospital, at day 2, at 2 weeks, or even at 1-2 months after birth, potentially before their one recommended postnatal obstetric visit at 3-8 weeks. Asking the mother how she is feeling at those times should not just be a social nicety but rather an additional check for serious postnatal complications.
Additional concerns
But wait, it gets worse.
Did you know that the leading cause of maternal death from pregnancy up to 1 year after a birth is homicide?
Maternal perinatal mortality figures have not usually included “perinatal-associated” deaths, a maternal death attributable to a condition that is unaffected by the pregnancy and occurring within 1 year of delivery (that I will cite as perinatal henceforth). While half of maternal deaths occur during pregnancy, another half occur in the year following. There were 3.62 homicides per 100,000 live births among females who were pregnant or within 1 year postpartum, 16% more than for similarly aged nonpregnant and nonpostpartum women (3.12 deaths/100,000 population, P < .05). Homicides made up 8.4% of reported perinatal maternal deaths from all causes, with a rate of 1.7 per 100,000 live births, twice the rate of any one of the other leading causes noted above. Black women had seven times the risk of perinatal homicide as that of White women. Females under 20, many of them our own pediatric patients, had a greater than six times higher risk and those aged 20-24 had a 65% higher risk of pregnancy-associated homicide across race and ethnic groups. Homicide is most likely before 21 weeks of pregnancy, decreases in the third trimester, but increases again after birth. Two-thirds of pregnancy-associated homicide deaths occurred in the home, with the perpetrator a current or prior partner (> 59%, with 98% being male), 45%-50% were associated with reported intimate partner violence (IPV), and the most common method was a firearm (55%). Often the same women had histories of substance abuse, serious mental illness, and/or prior IPV, all risk factors for pregnancy-associated deaths, including from homicide.
Homicide? “Not the mothers in my practice,” you may say, but, if not homicide, drug-related deaths (3.68 per 100,000 person-years) and suicide (1.42 per 100,000 person-years) together comprise 18% of all maternal deaths. Non-Hispanic White women, Medicaid-insured women, and women residing in smaller cities were especially likely to die from drugs or suicide. More than half (54.3%) of perinatal suicides involve intimate partner conflict, which increases the risk ninefold. Perinatal mood disorders, affecting up to 15% of pregnant and postpartum U.S. women, is also a risk factor in substance abuse, opioid overdose death, and suicide.
And substance use has gotten more dangerous with the increase in fentanyl lacing. Pregnancy-associated deaths (4%-10% of deaths) involving opioids more than doubled between 2007 and 2016, and, although the rates are higher for Black women, the increase has been greater for non-Hispanic White women. Two-thirds of those deaths occur between 6 and 12 months postpartum, on our watch. Although many women decrease substance use during pregnancy, they may fall back into substance use (rates increase 4 times by 7-12 months after delivery) and not continue to receive treatment. Although pharmacotherapy (e.g., methadone, buprenorphine treatment) is the current standard of care for opioid use disorder (OUD) during pregnancy, nearly half receiving treatment in publicly funded centers are not receiving these medications and others may lose insurance or access to pregnancy-related treatment programs after delivery, increasing risk of relapse. Stigma, and punitive or discriminatory approaches to pregnant women with OUD (e.g., jail, removal of children) can dissuade them from participating in treatment, increasing overdose risk.
It is important to note that in more than half of the 41 deaths from violent trauma in one study (including 22 homicides), obstetrical providers knew of or suspected IPV. Also, the vast majority (74%) of those who died by drugs or suicide had made one or more emergency department or hospital visit between their delivery and death, and 39% had made three or more visits. Without knowing if anything was done in those cases, we also know that, in addition to thorough, compassionate providers, there is sometimes segmentation of responsibility, insensitivity, discrimination, racism, stigma, inequity, lack of resources, lack of access, lack of payment mechanisms, legal issues for immigrants, time constraints, and other systemic deficits that may hinder effective care for these and subsequent women.
Awareness and action
What should we, who are primary care pediatric providers, do about these threats to the mothers and pregnant young women we care for? Clearly, their children, our main patients, would be terribly and permanently hurt by harm coming to their mothers – the extreme adverse childhood experiences and social determinants of health to which we are already committed.
I hope this article will help alert pediatric providers to what is being published, mainly as women’s health and public health issues.
First, we need awareness of the physical symptoms that may come up in our interactions with pregnant and postpartum women so that we can educate them and expedite any indicated emergency care.
Next, we need to expand our routine screening of mothers and pregnant women from just the most impactful social determinants of health (including depression, substance use, and IPV) to include anxiety, past suicide attempts and current suicidal ideation, and the presence of firearms, early and repeatedly in the first year of the child’s life. Adults and teens are more likely to disclose risk for sensitive issues through questionnaires than through interviews, perhaps even more so when the identified patient is their child rather than themselves. Any screen can have false negatives, so asking directly when risk is suspected is important. The reason for screening could be framed as caring for the caregiver who is the most important person for the child. It could be accompanied by acknowledging that pregnancy and the first year of life can be difficult for mothers and their partners and that we want to support them and connect them to resources, if needed. When substance use disorder is acknowledged, we should prescribe and teach about Narcan for overdose. When there is IPV, we should discuss firearm removal/locking as well as counseling on a personal safety plan.
Working as part of an on-site or virtual team that includes professionals who know about community resources and can coordinate care is essential, in addition to educating about 211 for services and 988 for suicide risk.
Finally, we can advocate and vote for programs, people, and laws that support and safeguard women and families, address substance use, and reduce access to firearms.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
New kids on the block for migraine treatment and prophylaxis
This transcript has been edited for clarity.
Dear colleagues, I’m Hans-Christoph Diener from the Faculty of Medicine at the University of Duisburg-Essen in Germany.
CGRP receptor agonists
Let me start with the treatment of acute migraine attacks. Until recently, we had analgesics, nonsteroidal anti-inflammatory drugs like ibuprofen, ergot alkaloids, and triptans. There are new developments, which are small molecules that are antagonists at the calcitonin gene-related peptide (CGRP) receptor. At the moment, we have three of them: rimegepant 75 mg, ubrogepant 50 mg or 100 mg, and zavegepant (a nasal spray) 10 mg.
These are all effective and superior to placebo. The 2-hour pain-free rate is somewhere between 25% and 30%. They have very few side effects; these include a little bit of nausea, somnolence, nasopharyngitis, and for zavegepant, the nasal spray, taste disturbance. In indirect comparisons, the so-called gepants are about as effective as ibuprofen and aspirin, and they seem to be less effective than sumatriptan 100 mg.
Unfortunately, until now, we have no direct comparison with triptans and we have no data demonstrating whether they are effective in people where triptans do not work. The major shortcoming is the cost in the United States. The cost per tablet or nasal spray is somewhere between $80 and $200. This means we definitely need more studies for these gepants.
Migraine prophylaxis
Let me move to the prophylaxis of migraine with drugs. Previously and still, we have all medications like beta-blockers, flunarizine, topiramate, valproic acid, amitriptyline, and candesartan, and for chronic migraine, onabotulinumtoxinA. We have now 5 years’ experience with the monoclonal antibodies against CGRP or the CGRP receptor like eptinezumab, erenumab, fremanezumab, and galcanezumab.
These are all equally effective. They reduce migraine-days between 3 and 7 per month. They are effective both in episodic and chronic migraine, and most importantly, they are effective in people with medication overuse headaches. The 50% responder rates are somewhere between 40% and 60%, and there are no significant differences between the four monoclonal antibodies.
The major advantage is a very good tolerability profile; very few patients terminate treatment because of adverse events. There has been, with one exception, no direct comparison of the monoclonal antibodies with traditional migraine preventive drugs or onabotulinumtoxinA. The only exception is a trial that compared topiramate and erenumab, showing that erenumab was definitely more effective and better tolerated.
At the moment, the recommendation is to use these monoclonal antibodies for 12 months in episodic migraine and 24 months in chronic migraine and then pause. It usually turns out that between 50% and 70% of these patients need to continue the treatment. If they are not working, there is a possibility to switch between the monoclonal antibodies, and the success rate after this is somewhere between 15% and 30%.
Gepants were also developed for the prevention of migraine. Here, we have rimegepant 75 mg every other day or atogepant 60 mg daily. They are effective, but in indirect comparisons, they are less effective than the monoclonal antibodies. At present, we have no comparative trials with monoclonal antibodies or the traditional migraine preventive drugs.
Potential patients are those who have needle phobia or patients who do not respond to monoclonal antibodies. Again, the biggest shortcoming is cost in the United States. The cost per year for migraine prevention or prophylaxis is between $12,000 and $20,000.
Finally, we also had very exciting news. There is a new therapeutic approach via PACAP. PACAP is pituitary adenylate cyclase-activating polypeptide, which has similar biological actions as CGRP but with additional actions. It could very well be that people who do not respond to a monoclonal antibody would respond to a monoclonal antibody against PACAP.
At the congress, the first randomized, placebo-controlled trial with a monoclonal antibody against PACAP was presented. This monoclonal antibody was effective in a population of people in whom prior preventive therapy had failed. A phase 3 study is planned, and most probably the PACAP monoclonal could work in people who do not respond to monoclonal antibodies against CGRP.
Dear colleagues, we have now many choices for the acute treatment of migraine and migraine prophylaxis. We have new kids on the block, and we have to learn more about how to use these drugs, their benefits, and their shortcomings.
He has disclosed the following relevant financial relationships:Received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Abbott; Addex Pharma; Alder; Allergan; Almirall; Amgen; Autonomic Technology; AstraZeneca; Bayer Vital; Berlin Chemie; Bristol-Myers Squibb; Boehringer Ingelheim; Chordate; CoAxia; Corimmun; Covidien; Coherex; CoLucid; Daiichi-Sankyo; D-Pharm; Electrocore; Fresenius; GlaxoSmithKline; Grunenthal; Janssen-Cilag; Labrys Biologics Lilly; La Roche; 3M Medica; MSD; Medtronic; Menarini; MindFrame; Minster; Neuroscore; Neurobiological Technologies; Novartis; Novo Nordisk; Johnson & Johnson; Knoll; Paion; Parke-Davis; Pierre Fabre; Pfizer; Schaper and Brummer; Sanofi-Aventis; Schering-Plough; Servier; Solvay; Syngis; St. Jude; Talecris; Thrombogenics; WebMD Global; Weber and Weber; Wyeth; and Yamanouchi.
Dr. Diener is professor, department of neurology, Stroke Center-Headache Center, University Duisburg-Essen (Germany).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Dear colleagues, I’m Hans-Christoph Diener from the Faculty of Medicine at the University of Duisburg-Essen in Germany.
CGRP receptor agonists
Let me start with the treatment of acute migraine attacks. Until recently, we had analgesics, nonsteroidal anti-inflammatory drugs like ibuprofen, ergot alkaloids, and triptans. There are new developments, which are small molecules that are antagonists at the calcitonin gene-related peptide (CGRP) receptor. At the moment, we have three of them: rimegepant 75 mg, ubrogepant 50 mg or 100 mg, and zavegepant (a nasal spray) 10 mg.
These are all effective and superior to placebo. The 2-hour pain-free rate is somewhere between 25% and 30%. They have very few side effects; these include a little bit of nausea, somnolence, nasopharyngitis, and for zavegepant, the nasal spray, taste disturbance. In indirect comparisons, the so-called gepants are about as effective as ibuprofen and aspirin, and they seem to be less effective than sumatriptan 100 mg.
Unfortunately, until now, we have no direct comparison with triptans and we have no data demonstrating whether they are effective in people where triptans do not work. The major shortcoming is the cost in the United States. The cost per tablet or nasal spray is somewhere between $80 and $200. This means we definitely need more studies for these gepants.
Migraine prophylaxis
Let me move to the prophylaxis of migraine with drugs. Previously and still, we have all medications like beta-blockers, flunarizine, topiramate, valproic acid, amitriptyline, and candesartan, and for chronic migraine, onabotulinumtoxinA. We have now 5 years’ experience with the monoclonal antibodies against CGRP or the CGRP receptor like eptinezumab, erenumab, fremanezumab, and galcanezumab.
These are all equally effective. They reduce migraine-days between 3 and 7 per month. They are effective both in episodic and chronic migraine, and most importantly, they are effective in people with medication overuse headaches. The 50% responder rates are somewhere between 40% and 60%, and there are no significant differences between the four monoclonal antibodies.
The major advantage is a very good tolerability profile; very few patients terminate treatment because of adverse events. There has been, with one exception, no direct comparison of the monoclonal antibodies with traditional migraine preventive drugs or onabotulinumtoxinA. The only exception is a trial that compared topiramate and erenumab, showing that erenumab was definitely more effective and better tolerated.
At the moment, the recommendation is to use these monoclonal antibodies for 12 months in episodic migraine and 24 months in chronic migraine and then pause. It usually turns out that between 50% and 70% of these patients need to continue the treatment. If they are not working, there is a possibility to switch between the monoclonal antibodies, and the success rate after this is somewhere between 15% and 30%.
Gepants were also developed for the prevention of migraine. Here, we have rimegepant 75 mg every other day or atogepant 60 mg daily. They are effective, but in indirect comparisons, they are less effective than the monoclonal antibodies. At present, we have no comparative trials with monoclonal antibodies or the traditional migraine preventive drugs.
Potential patients are those who have needle phobia or patients who do not respond to monoclonal antibodies. Again, the biggest shortcoming is cost in the United States. The cost per year for migraine prevention or prophylaxis is between $12,000 and $20,000.
Finally, we also had very exciting news. There is a new therapeutic approach via PACAP. PACAP is pituitary adenylate cyclase-activating polypeptide, which has similar biological actions as CGRP but with additional actions. It could very well be that people who do not respond to a monoclonal antibody would respond to a monoclonal antibody against PACAP.
At the congress, the first randomized, placebo-controlled trial with a monoclonal antibody against PACAP was presented. This monoclonal antibody was effective in a population of people in whom prior preventive therapy had failed. A phase 3 study is planned, and most probably the PACAP monoclonal could work in people who do not respond to monoclonal antibodies against CGRP.
Dear colleagues, we have now many choices for the acute treatment of migraine and migraine prophylaxis. We have new kids on the block, and we have to learn more about how to use these drugs, their benefits, and their shortcomings.
He has disclosed the following relevant financial relationships:Received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Abbott; Addex Pharma; Alder; Allergan; Almirall; Amgen; Autonomic Technology; AstraZeneca; Bayer Vital; Berlin Chemie; Bristol-Myers Squibb; Boehringer Ingelheim; Chordate; CoAxia; Corimmun; Covidien; Coherex; CoLucid; Daiichi-Sankyo; D-Pharm; Electrocore; Fresenius; GlaxoSmithKline; Grunenthal; Janssen-Cilag; Labrys Biologics Lilly; La Roche; 3M Medica; MSD; Medtronic; Menarini; MindFrame; Minster; Neuroscore; Neurobiological Technologies; Novartis; Novo Nordisk; Johnson & Johnson; Knoll; Paion; Parke-Davis; Pierre Fabre; Pfizer; Schaper and Brummer; Sanofi-Aventis; Schering-Plough; Servier; Solvay; Syngis; St. Jude; Talecris; Thrombogenics; WebMD Global; Weber and Weber; Wyeth; and Yamanouchi.
Dr. Diener is professor, department of neurology, Stroke Center-Headache Center, University Duisburg-Essen (Germany).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Dear colleagues, I’m Hans-Christoph Diener from the Faculty of Medicine at the University of Duisburg-Essen in Germany.
CGRP receptor agonists
Let me start with the treatment of acute migraine attacks. Until recently, we had analgesics, nonsteroidal anti-inflammatory drugs like ibuprofen, ergot alkaloids, and triptans. There are new developments, which are small molecules that are antagonists at the calcitonin gene-related peptide (CGRP) receptor. At the moment, we have three of them: rimegepant 75 mg, ubrogepant 50 mg or 100 mg, and zavegepant (a nasal spray) 10 mg.
These are all effective and superior to placebo. The 2-hour pain-free rate is somewhere between 25% and 30%. They have very few side effects; these include a little bit of nausea, somnolence, nasopharyngitis, and for zavegepant, the nasal spray, taste disturbance. In indirect comparisons, the so-called gepants are about as effective as ibuprofen and aspirin, and they seem to be less effective than sumatriptan 100 mg.
Unfortunately, until now, we have no direct comparison with triptans and we have no data demonstrating whether they are effective in people where triptans do not work. The major shortcoming is the cost in the United States. The cost per tablet or nasal spray is somewhere between $80 and $200. This means we definitely need more studies for these gepants.
Migraine prophylaxis
Let me move to the prophylaxis of migraine with drugs. Previously and still, we have all medications like beta-blockers, flunarizine, topiramate, valproic acid, amitriptyline, and candesartan, and for chronic migraine, onabotulinumtoxinA. We have now 5 years’ experience with the monoclonal antibodies against CGRP or the CGRP receptor like eptinezumab, erenumab, fremanezumab, and galcanezumab.
These are all equally effective. They reduce migraine-days between 3 and 7 per month. They are effective both in episodic and chronic migraine, and most importantly, they are effective in people with medication overuse headaches. The 50% responder rates are somewhere between 40% and 60%, and there are no significant differences between the four monoclonal antibodies.
The major advantage is a very good tolerability profile; very few patients terminate treatment because of adverse events. There has been, with one exception, no direct comparison of the monoclonal antibodies with traditional migraine preventive drugs or onabotulinumtoxinA. The only exception is a trial that compared topiramate and erenumab, showing that erenumab was definitely more effective and better tolerated.
At the moment, the recommendation is to use these monoclonal antibodies for 12 months in episodic migraine and 24 months in chronic migraine and then pause. It usually turns out that between 50% and 70% of these patients need to continue the treatment. If they are not working, there is a possibility to switch between the monoclonal antibodies, and the success rate after this is somewhere between 15% and 30%.
Gepants were also developed for the prevention of migraine. Here, we have rimegepant 75 mg every other day or atogepant 60 mg daily. They are effective, but in indirect comparisons, they are less effective than the monoclonal antibodies. At present, we have no comparative trials with monoclonal antibodies or the traditional migraine preventive drugs.
Potential patients are those who have needle phobia or patients who do not respond to monoclonal antibodies. Again, the biggest shortcoming is cost in the United States. The cost per year for migraine prevention or prophylaxis is between $12,000 and $20,000.
Finally, we also had very exciting news. There is a new therapeutic approach via PACAP. PACAP is pituitary adenylate cyclase-activating polypeptide, which has similar biological actions as CGRP but with additional actions. It could very well be that people who do not respond to a monoclonal antibody would respond to a monoclonal antibody against PACAP.
At the congress, the first randomized, placebo-controlled trial with a monoclonal antibody against PACAP was presented. This monoclonal antibody was effective in a population of people in whom prior preventive therapy had failed. A phase 3 study is planned, and most probably the PACAP monoclonal could work in people who do not respond to monoclonal antibodies against CGRP.
Dear colleagues, we have now many choices for the acute treatment of migraine and migraine prophylaxis. We have new kids on the block, and we have to learn more about how to use these drugs, their benefits, and their shortcomings.
He has disclosed the following relevant financial relationships:Received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Abbott; Addex Pharma; Alder; Allergan; Almirall; Amgen; Autonomic Technology; AstraZeneca; Bayer Vital; Berlin Chemie; Bristol-Myers Squibb; Boehringer Ingelheim; Chordate; CoAxia; Corimmun; Covidien; Coherex; CoLucid; Daiichi-Sankyo; D-Pharm; Electrocore; Fresenius; GlaxoSmithKline; Grunenthal; Janssen-Cilag; Labrys Biologics Lilly; La Roche; 3M Medica; MSD; Medtronic; Menarini; MindFrame; Minster; Neuroscore; Neurobiological Technologies; Novartis; Novo Nordisk; Johnson & Johnson; Knoll; Paion; Parke-Davis; Pierre Fabre; Pfizer; Schaper and Brummer; Sanofi-Aventis; Schering-Plough; Servier; Solvay; Syngis; St. Jude; Talecris; Thrombogenics; WebMD Global; Weber and Weber; Wyeth; and Yamanouchi.
Dr. Diener is professor, department of neurology, Stroke Center-Headache Center, University Duisburg-Essen (Germany).
A version of this article appeared on Medscape.com.
Docs weigh in on insurance coverage for obesity medications
You can’t argue with success, unless you are an insurance company faced with covering medications shown to improve obesity.
The ability of drugs originally designed for diabetes management to reduce body weight has spiked demand and taxed supplies, according to the U.S. Food and Drug Administration, which included semaglutide (both Wegovy and Ozempic) on its Drug Shortages List as of May 31, 2023.
Meanwhile, clinicians and patients report that insurance companies are pushing back against coverage of these medications that mimic glucagon-like peptide 1 (GLP-1) because of the costs. A recent study conducted by Prime Therapeutics, a pharmacy benefit management organization, showed that individuals who started GLP-1 drugs for weight loss and who were adherent to the treatment averaged a 59% increase in health care costs after 1 year; for those in a subgroup analysis who were treatment adherent, the increase in health care costs was 98%.
“Insurance coverage for obesity treatment is challenging, particularly regarding medications,” said Scott Kahan, MD, director of the National Center for Weight and Wellness at George Washington University, Washington, in an interview. Employers must opt in for patients to have coverage for these medications; therefore, relatively few patients have had access at reasonable out-of-pocket costs, he said.
For example, the University of Texas stated on its website that its prescription drug plans will no longer cover drugs with the active ingredients semaglutide (Wegovy) or liraglutide (Saxenda) for weight loss as of Sept. 1, 2023. Both products are FDA-approved for weight management, whereas the equally popular Ozempic is currently approved only as a treatment for diabetes. The school’s website noted that the current price of the drugs, which cost the plan more than $5 million per month as of May 2023, outstrips the most expensive cancer agents.
The University of Texas also found that among its patients, the compliance rate for those who began Wegovy or Saxenda for weight loss was only 46%, which was not enough to justify continued coverage. The plan advised patients to approach their insurers directly.
Eventually, more information may prompt more support from insurance across a range of medications, Dr. Kahan noted. “Most insurers are wanting cost-effectiveness data in order to support their investments in broader coverage,” he said.
However, costs do vary with and without insurance; some medications are less expensive than others without significant differences in outcomes, so encourage patients to explore all the options and not just one brand, Dr. Kahan said.
Educate patients on plan details
Clinicians can’t guarantee coverage, but they can offer guidance to their patients, according to said Andrew Kraftson, MD, an endocrinologist and internal medicine physician at the University of Michigan, Ann Arbor, who specializes in the care of people with obesity.
Unfortunately, some of the challenges to obtaining insurance coverage for weight-loss medications lie in the plan details because some insurers have a blanket prohibition against the use of weight-loss medications, he said.
If patients did not look for this particular aspect of coverage at the time of enrollment in their chosen plan, they may not have known about this exclusion, and they are disappointed to find that they are ineligible for weight-loss medications despite medical circumstances, Dr. Kraftson said in an interview.
If weight-loss medications are covered, prior authorization often is required, Dr. Kraftson added.
“Unfortunately, the requirements vary from insurer to insurer, and this can present challenges for the busy clinicians who may not have dedicated staff to assist with these authorizations. Sometimes, the requirements are exactingly particular, and denials can commonly occur,” he said.
Some insurers will cover weight-loss medications for an initial period then require a certain degree of weight loss before renewing the approval, Dr. Kraftson said.
“While this is reasonable, sometimes it is necessary to titrate a medication more slowly to help a patient get used to the medicine, so they may not reach the required weight loss in the time required by the insurer,” he said. “As such, the medical professional is ‘punished’ for trying to be safe and patient-sensitive, and the patient may lose coverage of the medicine.”
Clinicians can help patients increase their chances for insurance coverage by providing a patient instruction guide to walk them through the steps that allow the patient to make inquiries with their own insurer, Dr. Kraftson said.
This guide should instruct patients on how to read their prescription coverage card to correctly contact their insurer, along with a guide to medical coverage terminology.
Lauren Oshman, MD, also of the University of Michigan, heads a collaborative quality initiative in the state known as Michigan Collaborative for Type 2 Diabetes (MCT2D). Dr. Oshman and her colleagues created a user-friendly list of terms to help patients understand their plans and better advocate for coverage (see below). The list was designed to guide patients with diabetes but applies to any medication.
Learn the lingo (common insurance terms and definitions)
- Deductible: Predetermined amount that must be paid annually before insurance pays for anything.
- Copayment: Set amount paid for a prescription.
- Coinsurance: Amount you pay after your deductible is met. Your insurance pays their portion. Coinsurance only applies to prescriptions and services covered under your health plan.
- Medication tier: Levels of insurance medication coverage; you play a smaller amount for a lower tier and a higher amount for a higher tier.
- Out-of-pocket max: Annual limit on what you pay before insurance covers 100% of covered services. Deductibles, copayment, and coinsurance all apply toward your out-of-pocket maximum.
- Prior authorization: Request made by your doctor to insurance company for coverage of a medication.
- Quantity limit: Limitation on the number of pills covered for a period of time.
- Step therapy: Medication you must have tried prior to approval of a nonpreferred medication, typically prior to trying a more expensive medication.
(Source: Learn the Lingo: A Guide to Common Insurance Terms and Definitions, courtesy of Lauren Oshman, MD, and MCT2D)
Also, make sure patients understand that they need to find out whether they have a deductible and if so, how much it is, Dr. Kraftson said.
Pros and cons of compounding
Compounded drugs are not approved by the FDA; however, that does not mean they are not available, and patients may pursue them as an option for weight-loss drugs.
In a statement issued on May 31, 2023, the FDA cited reports of adverse events associated with the use of compounded weight-loss drugs as a lower-cost alternative to the approved product. The FDA emphasized that the agency does not review compounded versions of weight-loss drugs for safety, efficacy, or quality.
Dr. Kraftson cited the lack of quality control, transparency, and safety data as reasons to discourage his patients from pursuing compounded medications.
“If a patient insists on pursuing it, then I review the position statement from the Obesity Medicine Association,” he said. The OMA statement recommends that anti-obesity medications undergo clinical trials and noted the lack of FDA oversight on these products. The OMA statement also advises compounded peptides to be “legally produced by companies whose identities are readily disclosed, and who have documented manufacturing processes compliant with oversight by applicable regulatory agencies.”
Tracking outcomes might boost coverage
Robust data on the long-term cost-effectiveness of weight-loss medications are lacking, although this is changing, Dr. Kraftson said. A 2022 study published last year in the Journal of Managed Care and Specialty Pharmacy showed that a 2.4-mg dose of semaglutide was cost-effective, compared with no treatment, diet and exercise, and other anti-obesity medications based on gains in quality of life.
“Regardless, insurers are not as motivated by long-term cost effectiveness,” Dr. Kraftson said. Insurers are accustomed to employee turnover and are more likely to be motivated by short-term costs and benefits, he said. “Obesity treatment provides some short-term benefit, but the majority of the benefit can be experienced when we look at the long-term horizon,” he said.
Looking ahead, “We need better ways to account for the myriad benefits experienced by patients with successful weight control beyond what is currently measured as metrics of success, including better ways to qualify and quantify quality-of-life benefits,” Dr. Kraftson said.
Also, clinicians should address the stigma associated with obesity, Dr. Kraftson said.
“We would not see the spate of coverage restrictions if we were talking about heart disease or cancer; insurers can get away with this because obesity is held to a different standard and patients with obesity are used to being undertreated and mistreated by the medical community and society,” he said. “We need to better account for the true costs of excess weight/obesity beyond what is traditionally accepted. This would help make the case for the cost-effective nature of treatment.”
Dr. Kraftson and Dr. Oshman disclosed no relevant financial relationships. Dr. Kahan had no financial conflicts and serves on the Medscape Editorial Advisory Board.
You can’t argue with success, unless you are an insurance company faced with covering medications shown to improve obesity.
The ability of drugs originally designed for diabetes management to reduce body weight has spiked demand and taxed supplies, according to the U.S. Food and Drug Administration, which included semaglutide (both Wegovy and Ozempic) on its Drug Shortages List as of May 31, 2023.
Meanwhile, clinicians and patients report that insurance companies are pushing back against coverage of these medications that mimic glucagon-like peptide 1 (GLP-1) because of the costs. A recent study conducted by Prime Therapeutics, a pharmacy benefit management organization, showed that individuals who started GLP-1 drugs for weight loss and who were adherent to the treatment averaged a 59% increase in health care costs after 1 year; for those in a subgroup analysis who were treatment adherent, the increase in health care costs was 98%.
“Insurance coverage for obesity treatment is challenging, particularly regarding medications,” said Scott Kahan, MD, director of the National Center for Weight and Wellness at George Washington University, Washington, in an interview. Employers must opt in for patients to have coverage for these medications; therefore, relatively few patients have had access at reasonable out-of-pocket costs, he said.
For example, the University of Texas stated on its website that its prescription drug plans will no longer cover drugs with the active ingredients semaglutide (Wegovy) or liraglutide (Saxenda) for weight loss as of Sept. 1, 2023. Both products are FDA-approved for weight management, whereas the equally popular Ozempic is currently approved only as a treatment for diabetes. The school’s website noted that the current price of the drugs, which cost the plan more than $5 million per month as of May 2023, outstrips the most expensive cancer agents.
The University of Texas also found that among its patients, the compliance rate for those who began Wegovy or Saxenda for weight loss was only 46%, which was not enough to justify continued coverage. The plan advised patients to approach their insurers directly.
Eventually, more information may prompt more support from insurance across a range of medications, Dr. Kahan noted. “Most insurers are wanting cost-effectiveness data in order to support their investments in broader coverage,” he said.
However, costs do vary with and without insurance; some medications are less expensive than others without significant differences in outcomes, so encourage patients to explore all the options and not just one brand, Dr. Kahan said.
Educate patients on plan details
Clinicians can’t guarantee coverage, but they can offer guidance to their patients, according to said Andrew Kraftson, MD, an endocrinologist and internal medicine physician at the University of Michigan, Ann Arbor, who specializes in the care of people with obesity.
Unfortunately, some of the challenges to obtaining insurance coverage for weight-loss medications lie in the plan details because some insurers have a blanket prohibition against the use of weight-loss medications, he said.
If patients did not look for this particular aspect of coverage at the time of enrollment in their chosen plan, they may not have known about this exclusion, and they are disappointed to find that they are ineligible for weight-loss medications despite medical circumstances, Dr. Kraftson said in an interview.
If weight-loss medications are covered, prior authorization often is required, Dr. Kraftson added.
“Unfortunately, the requirements vary from insurer to insurer, and this can present challenges for the busy clinicians who may not have dedicated staff to assist with these authorizations. Sometimes, the requirements are exactingly particular, and denials can commonly occur,” he said.
Some insurers will cover weight-loss medications for an initial period then require a certain degree of weight loss before renewing the approval, Dr. Kraftson said.
“While this is reasonable, sometimes it is necessary to titrate a medication more slowly to help a patient get used to the medicine, so they may not reach the required weight loss in the time required by the insurer,” he said. “As such, the medical professional is ‘punished’ for trying to be safe and patient-sensitive, and the patient may lose coverage of the medicine.”
Clinicians can help patients increase their chances for insurance coverage by providing a patient instruction guide to walk them through the steps that allow the patient to make inquiries with their own insurer, Dr. Kraftson said.
This guide should instruct patients on how to read their prescription coverage card to correctly contact their insurer, along with a guide to medical coverage terminology.
Lauren Oshman, MD, also of the University of Michigan, heads a collaborative quality initiative in the state known as Michigan Collaborative for Type 2 Diabetes (MCT2D). Dr. Oshman and her colleagues created a user-friendly list of terms to help patients understand their plans and better advocate for coverage (see below). The list was designed to guide patients with diabetes but applies to any medication.
Learn the lingo (common insurance terms and definitions)
- Deductible: Predetermined amount that must be paid annually before insurance pays for anything.
- Copayment: Set amount paid for a prescription.
- Coinsurance: Amount you pay after your deductible is met. Your insurance pays their portion. Coinsurance only applies to prescriptions and services covered under your health plan.
- Medication tier: Levels of insurance medication coverage; you play a smaller amount for a lower tier and a higher amount for a higher tier.
- Out-of-pocket max: Annual limit on what you pay before insurance covers 100% of covered services. Deductibles, copayment, and coinsurance all apply toward your out-of-pocket maximum.
- Prior authorization: Request made by your doctor to insurance company for coverage of a medication.
- Quantity limit: Limitation on the number of pills covered for a period of time.
- Step therapy: Medication you must have tried prior to approval of a nonpreferred medication, typically prior to trying a more expensive medication.
(Source: Learn the Lingo: A Guide to Common Insurance Terms and Definitions, courtesy of Lauren Oshman, MD, and MCT2D)
Also, make sure patients understand that they need to find out whether they have a deductible and if so, how much it is, Dr. Kraftson said.
Pros and cons of compounding
Compounded drugs are not approved by the FDA; however, that does not mean they are not available, and patients may pursue them as an option for weight-loss drugs.
In a statement issued on May 31, 2023, the FDA cited reports of adverse events associated with the use of compounded weight-loss drugs as a lower-cost alternative to the approved product. The FDA emphasized that the agency does not review compounded versions of weight-loss drugs for safety, efficacy, or quality.
Dr. Kraftson cited the lack of quality control, transparency, and safety data as reasons to discourage his patients from pursuing compounded medications.
“If a patient insists on pursuing it, then I review the position statement from the Obesity Medicine Association,” he said. The OMA statement recommends that anti-obesity medications undergo clinical trials and noted the lack of FDA oversight on these products. The OMA statement also advises compounded peptides to be “legally produced by companies whose identities are readily disclosed, and who have documented manufacturing processes compliant with oversight by applicable regulatory agencies.”
Tracking outcomes might boost coverage
Robust data on the long-term cost-effectiveness of weight-loss medications are lacking, although this is changing, Dr. Kraftson said. A 2022 study published last year in the Journal of Managed Care and Specialty Pharmacy showed that a 2.4-mg dose of semaglutide was cost-effective, compared with no treatment, diet and exercise, and other anti-obesity medications based on gains in quality of life.
“Regardless, insurers are not as motivated by long-term cost effectiveness,” Dr. Kraftson said. Insurers are accustomed to employee turnover and are more likely to be motivated by short-term costs and benefits, he said. “Obesity treatment provides some short-term benefit, but the majority of the benefit can be experienced when we look at the long-term horizon,” he said.
Looking ahead, “We need better ways to account for the myriad benefits experienced by patients with successful weight control beyond what is currently measured as metrics of success, including better ways to qualify and quantify quality-of-life benefits,” Dr. Kraftson said.
Also, clinicians should address the stigma associated with obesity, Dr. Kraftson said.
“We would not see the spate of coverage restrictions if we were talking about heart disease or cancer; insurers can get away with this because obesity is held to a different standard and patients with obesity are used to being undertreated and mistreated by the medical community and society,” he said. “We need to better account for the true costs of excess weight/obesity beyond what is traditionally accepted. This would help make the case for the cost-effective nature of treatment.”
Dr. Kraftson and Dr. Oshman disclosed no relevant financial relationships. Dr. Kahan had no financial conflicts and serves on the Medscape Editorial Advisory Board.
You can’t argue with success, unless you are an insurance company faced with covering medications shown to improve obesity.
The ability of drugs originally designed for diabetes management to reduce body weight has spiked demand and taxed supplies, according to the U.S. Food and Drug Administration, which included semaglutide (both Wegovy and Ozempic) on its Drug Shortages List as of May 31, 2023.
Meanwhile, clinicians and patients report that insurance companies are pushing back against coverage of these medications that mimic glucagon-like peptide 1 (GLP-1) because of the costs. A recent study conducted by Prime Therapeutics, a pharmacy benefit management organization, showed that individuals who started GLP-1 drugs for weight loss and who were adherent to the treatment averaged a 59% increase in health care costs after 1 year; for those in a subgroup analysis who were treatment adherent, the increase in health care costs was 98%.
“Insurance coverage for obesity treatment is challenging, particularly regarding medications,” said Scott Kahan, MD, director of the National Center for Weight and Wellness at George Washington University, Washington, in an interview. Employers must opt in for patients to have coverage for these medications; therefore, relatively few patients have had access at reasonable out-of-pocket costs, he said.
For example, the University of Texas stated on its website that its prescription drug plans will no longer cover drugs with the active ingredients semaglutide (Wegovy) or liraglutide (Saxenda) for weight loss as of Sept. 1, 2023. Both products are FDA-approved for weight management, whereas the equally popular Ozempic is currently approved only as a treatment for diabetes. The school’s website noted that the current price of the drugs, which cost the plan more than $5 million per month as of May 2023, outstrips the most expensive cancer agents.
The University of Texas also found that among its patients, the compliance rate for those who began Wegovy or Saxenda for weight loss was only 46%, which was not enough to justify continued coverage. The plan advised patients to approach their insurers directly.
Eventually, more information may prompt more support from insurance across a range of medications, Dr. Kahan noted. “Most insurers are wanting cost-effectiveness data in order to support their investments in broader coverage,” he said.
However, costs do vary with and without insurance; some medications are less expensive than others without significant differences in outcomes, so encourage patients to explore all the options and not just one brand, Dr. Kahan said.
Educate patients on plan details
Clinicians can’t guarantee coverage, but they can offer guidance to their patients, according to said Andrew Kraftson, MD, an endocrinologist and internal medicine physician at the University of Michigan, Ann Arbor, who specializes in the care of people with obesity.
Unfortunately, some of the challenges to obtaining insurance coverage for weight-loss medications lie in the plan details because some insurers have a blanket prohibition against the use of weight-loss medications, he said.
If patients did not look for this particular aspect of coverage at the time of enrollment in their chosen plan, they may not have known about this exclusion, and they are disappointed to find that they are ineligible for weight-loss medications despite medical circumstances, Dr. Kraftson said in an interview.
If weight-loss medications are covered, prior authorization often is required, Dr. Kraftson added.
“Unfortunately, the requirements vary from insurer to insurer, and this can present challenges for the busy clinicians who may not have dedicated staff to assist with these authorizations. Sometimes, the requirements are exactingly particular, and denials can commonly occur,” he said.
Some insurers will cover weight-loss medications for an initial period then require a certain degree of weight loss before renewing the approval, Dr. Kraftson said.
“While this is reasonable, sometimes it is necessary to titrate a medication more slowly to help a patient get used to the medicine, so they may not reach the required weight loss in the time required by the insurer,” he said. “As such, the medical professional is ‘punished’ for trying to be safe and patient-sensitive, and the patient may lose coverage of the medicine.”
Clinicians can help patients increase their chances for insurance coverage by providing a patient instruction guide to walk them through the steps that allow the patient to make inquiries with their own insurer, Dr. Kraftson said.
This guide should instruct patients on how to read their prescription coverage card to correctly contact their insurer, along with a guide to medical coverage terminology.
Lauren Oshman, MD, also of the University of Michigan, heads a collaborative quality initiative in the state known as Michigan Collaborative for Type 2 Diabetes (MCT2D). Dr. Oshman and her colleagues created a user-friendly list of terms to help patients understand their plans and better advocate for coverage (see below). The list was designed to guide patients with diabetes but applies to any medication.
Learn the lingo (common insurance terms and definitions)
- Deductible: Predetermined amount that must be paid annually before insurance pays for anything.
- Copayment: Set amount paid for a prescription.
- Coinsurance: Amount you pay after your deductible is met. Your insurance pays their portion. Coinsurance only applies to prescriptions and services covered under your health plan.
- Medication tier: Levels of insurance medication coverage; you play a smaller amount for a lower tier and a higher amount for a higher tier.
- Out-of-pocket max: Annual limit on what you pay before insurance covers 100% of covered services. Deductibles, copayment, and coinsurance all apply toward your out-of-pocket maximum.
- Prior authorization: Request made by your doctor to insurance company for coverage of a medication.
- Quantity limit: Limitation on the number of pills covered for a period of time.
- Step therapy: Medication you must have tried prior to approval of a nonpreferred medication, typically prior to trying a more expensive medication.
(Source: Learn the Lingo: A Guide to Common Insurance Terms and Definitions, courtesy of Lauren Oshman, MD, and MCT2D)
Also, make sure patients understand that they need to find out whether they have a deductible and if so, how much it is, Dr. Kraftson said.
Pros and cons of compounding
Compounded drugs are not approved by the FDA; however, that does not mean they are not available, and patients may pursue them as an option for weight-loss drugs.
In a statement issued on May 31, 2023, the FDA cited reports of adverse events associated with the use of compounded weight-loss drugs as a lower-cost alternative to the approved product. The FDA emphasized that the agency does not review compounded versions of weight-loss drugs for safety, efficacy, or quality.
Dr. Kraftson cited the lack of quality control, transparency, and safety data as reasons to discourage his patients from pursuing compounded medications.
“If a patient insists on pursuing it, then I review the position statement from the Obesity Medicine Association,” he said. The OMA statement recommends that anti-obesity medications undergo clinical trials and noted the lack of FDA oversight on these products. The OMA statement also advises compounded peptides to be “legally produced by companies whose identities are readily disclosed, and who have documented manufacturing processes compliant with oversight by applicable regulatory agencies.”
Tracking outcomes might boost coverage
Robust data on the long-term cost-effectiveness of weight-loss medications are lacking, although this is changing, Dr. Kraftson said. A 2022 study published last year in the Journal of Managed Care and Specialty Pharmacy showed that a 2.4-mg dose of semaglutide was cost-effective, compared with no treatment, diet and exercise, and other anti-obesity medications based on gains in quality of life.
“Regardless, insurers are not as motivated by long-term cost effectiveness,” Dr. Kraftson said. Insurers are accustomed to employee turnover and are more likely to be motivated by short-term costs and benefits, he said. “Obesity treatment provides some short-term benefit, but the majority of the benefit can be experienced when we look at the long-term horizon,” he said.
Looking ahead, “We need better ways to account for the myriad benefits experienced by patients with successful weight control beyond what is currently measured as metrics of success, including better ways to qualify and quantify quality-of-life benefits,” Dr. Kraftson said.
Also, clinicians should address the stigma associated with obesity, Dr. Kraftson said.
“We would not see the spate of coverage restrictions if we were talking about heart disease or cancer; insurers can get away with this because obesity is held to a different standard and patients with obesity are used to being undertreated and mistreated by the medical community and society,” he said. “We need to better account for the true costs of excess weight/obesity beyond what is traditionally accepted. This would help make the case for the cost-effective nature of treatment.”
Dr. Kraftson and Dr. Oshman disclosed no relevant financial relationships. Dr. Kahan had no financial conflicts and serves on the Medscape Editorial Advisory Board.
Testosterone replacement benefits men with type 2 diabetes
HAMBURG, GERMANY – Testosterone replacement therapy was associated with significant reductions in hemoglobin A1c at 1 and 2 years among men with type 2 diabetes, a multinational audit shows.
“If you have a patient with type 2 diabetes, sexual dysfunction, or fatigue, please consider checking their testosterone level. And if they fulfill criteria for testosterone deficiency and have had their [prostate-specific antigen] checked, consider a trial of treatment and follow them,” study lead author T. Hugh Jones, MD, consultant physician and endocrinologist at Barnsley (England) Hospital NHS Foundation Trust advised, speaking with this news organization.
Dr. Jones also urges clinicians worldwide to enter their patients’ data into the ABCD Testosterone Audit, which aims to identify long-term outcomes and predictors of response to testosterone replacement therapy.
Dr. Jones, who is also professor of andrology at the University of Sheffield, presented the preliminary data analysis at the annual meeting of the European Association for the Study of Diabetes.
Thus far, a total of 428 men with type 2 diabetes and hypogonadism are entered into the audit, from 34 centers in eight countries: the United Kingdom, Germany, Canada, Brazil, South Africa, New Zealand, Malaysia, and Vietnam. Among 121 of the men at 12 months, there was a drop in A1c from a baseline level of 71.27 mmol/mol (8.7%) to 61.26 mmol/mol (7.8%). Among 104 men at 24 months, the drop was from 71.4 mmol/mol (8.7%) to 55.97 mmol/mol (7.3%). Both decreases were significant (P < .001).
Prior data from Dr. Jones’ group showed that about 40% of men with type 2 diabetes have symptomatic testosterone deficiency. Testosterone deficiency is also associated with adverse effects on cardiovascular risk factors, bone health, muscle strength, sexual function, and psychological well-being, yet it is often overlooked, Dr. Jones noted.
“It’s not typically measured in routine clinical practice. ... Deficiency is very common, but a lot of practitioners don’t treat it and don’t ask about it. But in fact, treatment has very significant benefits for patients. ... We know from sildenafil (Viagra) studies that 60%of people who didn’t respond were testosterone deficient. After being given testosterone, they converted to Viagra responders,” he noted.
Regarding safety concerns, the recent findings from the TRAVERSE study, in which about 70% of participants had type 2 diabetes, demonstrated no increased cardiovascular risk. There was also no association with prostate cancer, although it’s important to monitor prostate-specific antigen in patients for the first year on testosterone replacement, Dr. Jones said.
Asked to comment, endocrinologist Bradley D. Anawalt, MD, chief of medicine at the University of Washington Medical Center, Seattle, told this news organization, “This ‘worldwide survey’ confirms many studies from around the world over the past 20 years. ... [T]he association is due to ‘reverse causation,’ in that diabetes type 2 and obesity lower testosterone concentrations. Weight loss of 5%-10% may raise testosterone concentrations in men with high body mass indices, large waist circumferences, and low blood testosterone concentrations.”
At the same time, Dr. Anawalt pointed to data suggesting that “[t]reatment of androgen deficiency may facilitate lifestyle measures in men with high [body mass indexes] and high risk of type 2 diabetes to prevent, or more likely delay, the development of type 2 diabetes.”
However, both Dr. Jones and Dr. Anawalt emphasized that testosterone therapy would not be expected to affect blood glucose levels or any other cardiometabolic parameters in men who are not testosterone deficient, regardless of diabetes status.
“It’s important when you give testosterone to replace it to the normal level. Adequate treatment gives the greatest benefit,”Dr. Jones said.
As more centers contribute data to the ABCD audit, Jones anticipates collecting clinical practice data on a variety of clinical parameters, including complications, total insulin dose, kidney function, and eventually cardiovascular outcomes.
In the meantime, he said, giving testosterone replacement to men with deficiency can be very rewarding for many reasons. “People feel better. Individual patients come back and say ‘thank you doctor, you’ve given me my life back.’ It’s not often you get that. And the compliance is excellent.”
Dr. Jones is a speaker for, advisory board member for, and/or travel grant recipient of Besins Healthcare, Grantss, Grunenthal, and Simple Pharma. Dr. Anawalt has no disclosures.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – Testosterone replacement therapy was associated with significant reductions in hemoglobin A1c at 1 and 2 years among men with type 2 diabetes, a multinational audit shows.
“If you have a patient with type 2 diabetes, sexual dysfunction, or fatigue, please consider checking their testosterone level. And if they fulfill criteria for testosterone deficiency and have had their [prostate-specific antigen] checked, consider a trial of treatment and follow them,” study lead author T. Hugh Jones, MD, consultant physician and endocrinologist at Barnsley (England) Hospital NHS Foundation Trust advised, speaking with this news organization.
Dr. Jones also urges clinicians worldwide to enter their patients’ data into the ABCD Testosterone Audit, which aims to identify long-term outcomes and predictors of response to testosterone replacement therapy.
Dr. Jones, who is also professor of andrology at the University of Sheffield, presented the preliminary data analysis at the annual meeting of the European Association for the Study of Diabetes.
Thus far, a total of 428 men with type 2 diabetes and hypogonadism are entered into the audit, from 34 centers in eight countries: the United Kingdom, Germany, Canada, Brazil, South Africa, New Zealand, Malaysia, and Vietnam. Among 121 of the men at 12 months, there was a drop in A1c from a baseline level of 71.27 mmol/mol (8.7%) to 61.26 mmol/mol (7.8%). Among 104 men at 24 months, the drop was from 71.4 mmol/mol (8.7%) to 55.97 mmol/mol (7.3%). Both decreases were significant (P < .001).
Prior data from Dr. Jones’ group showed that about 40% of men with type 2 diabetes have symptomatic testosterone deficiency. Testosterone deficiency is also associated with adverse effects on cardiovascular risk factors, bone health, muscle strength, sexual function, and psychological well-being, yet it is often overlooked, Dr. Jones noted.
“It’s not typically measured in routine clinical practice. ... Deficiency is very common, but a lot of practitioners don’t treat it and don’t ask about it. But in fact, treatment has very significant benefits for patients. ... We know from sildenafil (Viagra) studies that 60%of people who didn’t respond were testosterone deficient. After being given testosterone, they converted to Viagra responders,” he noted.
Regarding safety concerns, the recent findings from the TRAVERSE study, in which about 70% of participants had type 2 diabetes, demonstrated no increased cardiovascular risk. There was also no association with prostate cancer, although it’s important to monitor prostate-specific antigen in patients for the first year on testosterone replacement, Dr. Jones said.
Asked to comment, endocrinologist Bradley D. Anawalt, MD, chief of medicine at the University of Washington Medical Center, Seattle, told this news organization, “This ‘worldwide survey’ confirms many studies from around the world over the past 20 years. ... [T]he association is due to ‘reverse causation,’ in that diabetes type 2 and obesity lower testosterone concentrations. Weight loss of 5%-10% may raise testosterone concentrations in men with high body mass indices, large waist circumferences, and low blood testosterone concentrations.”
At the same time, Dr. Anawalt pointed to data suggesting that “[t]reatment of androgen deficiency may facilitate lifestyle measures in men with high [body mass indexes] and high risk of type 2 diabetes to prevent, or more likely delay, the development of type 2 diabetes.”
However, both Dr. Jones and Dr. Anawalt emphasized that testosterone therapy would not be expected to affect blood glucose levels or any other cardiometabolic parameters in men who are not testosterone deficient, regardless of diabetes status.
“It’s important when you give testosterone to replace it to the normal level. Adequate treatment gives the greatest benefit,”Dr. Jones said.
As more centers contribute data to the ABCD audit, Jones anticipates collecting clinical practice data on a variety of clinical parameters, including complications, total insulin dose, kidney function, and eventually cardiovascular outcomes.
In the meantime, he said, giving testosterone replacement to men with deficiency can be very rewarding for many reasons. “People feel better. Individual patients come back and say ‘thank you doctor, you’ve given me my life back.’ It’s not often you get that. And the compliance is excellent.”
Dr. Jones is a speaker for, advisory board member for, and/or travel grant recipient of Besins Healthcare, Grantss, Grunenthal, and Simple Pharma. Dr. Anawalt has no disclosures.
A version of this article first appeared on Medscape.com.
HAMBURG, GERMANY – Testosterone replacement therapy was associated with significant reductions in hemoglobin A1c at 1 and 2 years among men with type 2 diabetes, a multinational audit shows.
“If you have a patient with type 2 diabetes, sexual dysfunction, or fatigue, please consider checking their testosterone level. And if they fulfill criteria for testosterone deficiency and have had their [prostate-specific antigen] checked, consider a trial of treatment and follow them,” study lead author T. Hugh Jones, MD, consultant physician and endocrinologist at Barnsley (England) Hospital NHS Foundation Trust advised, speaking with this news organization.
Dr. Jones also urges clinicians worldwide to enter their patients’ data into the ABCD Testosterone Audit, which aims to identify long-term outcomes and predictors of response to testosterone replacement therapy.
Dr. Jones, who is also professor of andrology at the University of Sheffield, presented the preliminary data analysis at the annual meeting of the European Association for the Study of Diabetes.
Thus far, a total of 428 men with type 2 diabetes and hypogonadism are entered into the audit, from 34 centers in eight countries: the United Kingdom, Germany, Canada, Brazil, South Africa, New Zealand, Malaysia, and Vietnam. Among 121 of the men at 12 months, there was a drop in A1c from a baseline level of 71.27 mmol/mol (8.7%) to 61.26 mmol/mol (7.8%). Among 104 men at 24 months, the drop was from 71.4 mmol/mol (8.7%) to 55.97 mmol/mol (7.3%). Both decreases were significant (P < .001).
Prior data from Dr. Jones’ group showed that about 40% of men with type 2 diabetes have symptomatic testosterone deficiency. Testosterone deficiency is also associated with adverse effects on cardiovascular risk factors, bone health, muscle strength, sexual function, and psychological well-being, yet it is often overlooked, Dr. Jones noted.
“It’s not typically measured in routine clinical practice. ... Deficiency is very common, but a lot of practitioners don’t treat it and don’t ask about it. But in fact, treatment has very significant benefits for patients. ... We know from sildenafil (Viagra) studies that 60%of people who didn’t respond were testosterone deficient. After being given testosterone, they converted to Viagra responders,” he noted.
Regarding safety concerns, the recent findings from the TRAVERSE study, in which about 70% of participants had type 2 diabetes, demonstrated no increased cardiovascular risk. There was also no association with prostate cancer, although it’s important to monitor prostate-specific antigen in patients for the first year on testosterone replacement, Dr. Jones said.
Asked to comment, endocrinologist Bradley D. Anawalt, MD, chief of medicine at the University of Washington Medical Center, Seattle, told this news organization, “This ‘worldwide survey’ confirms many studies from around the world over the past 20 years. ... [T]he association is due to ‘reverse causation,’ in that diabetes type 2 and obesity lower testosterone concentrations. Weight loss of 5%-10% may raise testosterone concentrations in men with high body mass indices, large waist circumferences, and low blood testosterone concentrations.”
At the same time, Dr. Anawalt pointed to data suggesting that “[t]reatment of androgen deficiency may facilitate lifestyle measures in men with high [body mass indexes] and high risk of type 2 diabetes to prevent, or more likely delay, the development of type 2 diabetes.”
However, both Dr. Jones and Dr. Anawalt emphasized that testosterone therapy would not be expected to affect blood glucose levels or any other cardiometabolic parameters in men who are not testosterone deficient, regardless of diabetes status.
“It’s important when you give testosterone to replace it to the normal level. Adequate treatment gives the greatest benefit,”Dr. Jones said.
As more centers contribute data to the ABCD audit, Jones anticipates collecting clinical practice data on a variety of clinical parameters, including complications, total insulin dose, kidney function, and eventually cardiovascular outcomes.
In the meantime, he said, giving testosterone replacement to men with deficiency can be very rewarding for many reasons. “People feel better. Individual patients come back and say ‘thank you doctor, you’ve given me my life back.’ It’s not often you get that. And the compliance is excellent.”
Dr. Jones is a speaker for, advisory board member for, and/or travel grant recipient of Besins Healthcare, Grantss, Grunenthal, and Simple Pharma. Dr. Anawalt has no disclosures.
A version of this article first appeared on Medscape.com.
AT EASD 2023
Where do you stand on the Middle East conflict?
“What do you think about the whole Israel thing?”
That question came at the end of an otherwise routine appointment.
Maybe she was just chatting. Maybe she wanted something deeper. I have no idea. I just said, “I don’t discuss those things with patients.”
My answer surprised her, but she didn’t push it. She paid her copay, scheduled a follow-up for 3 months, and left.
As I’ve written before, I try to avoid all news except the local weather. The sad reality is that most of it is bad and there’s nothing I can really do about it. It only upsets me, which isn’t good for my mental health and blood pressure, and if I can’t change it, what’s the point of knowing? It falls under the serenity prayer.
Of course, some news stories are too big not to hear something. I pass TVs in the doctors lounge or coffee house, hear others talking as I stand in line for the elevator, or see blurbs go by when checking the weather. It’s not entirely unavoidable.
I’m not trivializing the Middle East. But, to me, it’s not part of the doctor-patient relationship any more than my political views are. You run the risk of driving a wedge between you and the person you’re caring for. If you don’t like their opinion, you may find yourself less interested in them and their care. If they don’t like your opinion on news, they may start to question your ability as a doctor.
That’s not what we strive for, but it can be human nature. For better or worse we often reduce things to “us against them,” and learning someone is on the opposite side may, even subconsciously, alter how you treat them.
That’s not good, so to me it’s best not to know.
Some may think I’m being petty, or aloof, to be unwilling to discuss nonmedical issues of significance, but I don’t see it that way. Time is limited at the appointment and is best spent on medical care. Something unrelated to the visit that may alter my objective opinion of a patient – or theirs of me as a doctor – is best left out of it.
I’m here to be your doctor, and to do the best I can for you. I’m not here to be a debate partner. Whenever a patient asks me a question on politics or news I always think of the Monty Python skit “Argument Clinic.” That’s not why you’re here. There are plenty places to discuss such things. My office isn’t one of them.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“What do you think about the whole Israel thing?”
That question came at the end of an otherwise routine appointment.
Maybe she was just chatting. Maybe she wanted something deeper. I have no idea. I just said, “I don’t discuss those things with patients.”
My answer surprised her, but she didn’t push it. She paid her copay, scheduled a follow-up for 3 months, and left.
As I’ve written before, I try to avoid all news except the local weather. The sad reality is that most of it is bad and there’s nothing I can really do about it. It only upsets me, which isn’t good for my mental health and blood pressure, and if I can’t change it, what’s the point of knowing? It falls under the serenity prayer.
Of course, some news stories are too big not to hear something. I pass TVs in the doctors lounge or coffee house, hear others talking as I stand in line for the elevator, or see blurbs go by when checking the weather. It’s not entirely unavoidable.
I’m not trivializing the Middle East. But, to me, it’s not part of the doctor-patient relationship any more than my political views are. You run the risk of driving a wedge between you and the person you’re caring for. If you don’t like their opinion, you may find yourself less interested in them and their care. If they don’t like your opinion on news, they may start to question your ability as a doctor.
That’s not what we strive for, but it can be human nature. For better or worse we often reduce things to “us against them,” and learning someone is on the opposite side may, even subconsciously, alter how you treat them.
That’s not good, so to me it’s best not to know.
Some may think I’m being petty, or aloof, to be unwilling to discuss nonmedical issues of significance, but I don’t see it that way. Time is limited at the appointment and is best spent on medical care. Something unrelated to the visit that may alter my objective opinion of a patient – or theirs of me as a doctor – is best left out of it.
I’m here to be your doctor, and to do the best I can for you. I’m not here to be a debate partner. Whenever a patient asks me a question on politics or news I always think of the Monty Python skit “Argument Clinic.” That’s not why you’re here. There are plenty places to discuss such things. My office isn’t one of them.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“What do you think about the whole Israel thing?”
That question came at the end of an otherwise routine appointment.
Maybe she was just chatting. Maybe she wanted something deeper. I have no idea. I just said, “I don’t discuss those things with patients.”
My answer surprised her, but she didn’t push it. She paid her copay, scheduled a follow-up for 3 months, and left.
As I’ve written before, I try to avoid all news except the local weather. The sad reality is that most of it is bad and there’s nothing I can really do about it. It only upsets me, which isn’t good for my mental health and blood pressure, and if I can’t change it, what’s the point of knowing? It falls under the serenity prayer.
Of course, some news stories are too big not to hear something. I pass TVs in the doctors lounge or coffee house, hear others talking as I stand in line for the elevator, or see blurbs go by when checking the weather. It’s not entirely unavoidable.
I’m not trivializing the Middle East. But, to me, it’s not part of the doctor-patient relationship any more than my political views are. You run the risk of driving a wedge between you and the person you’re caring for. If you don’t like their opinion, you may find yourself less interested in them and their care. If they don’t like your opinion on news, they may start to question your ability as a doctor.
That’s not what we strive for, but it can be human nature. For better or worse we often reduce things to “us against them,” and learning someone is on the opposite side may, even subconsciously, alter how you treat them.
That’s not good, so to me it’s best not to know.
Some may think I’m being petty, or aloof, to be unwilling to discuss nonmedical issues of significance, but I don’t see it that way. Time is limited at the appointment and is best spent on medical care. Something unrelated to the visit that may alter my objective opinion of a patient – or theirs of me as a doctor – is best left out of it.
I’m here to be your doctor, and to do the best I can for you. I’m not here to be a debate partner. Whenever a patient asks me a question on politics or news I always think of the Monty Python skit “Argument Clinic.” That’s not why you’re here. There are plenty places to discuss such things. My office isn’t one of them.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
AI in medicine has a major Cassandra problem
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.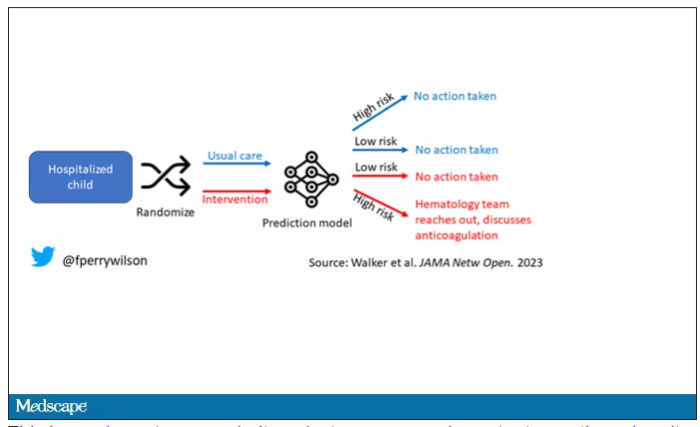
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.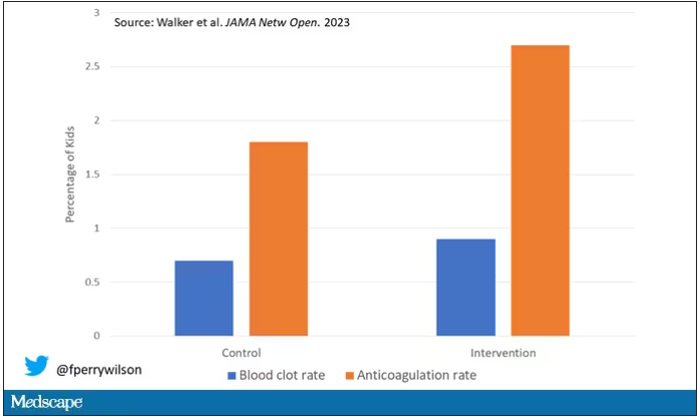
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.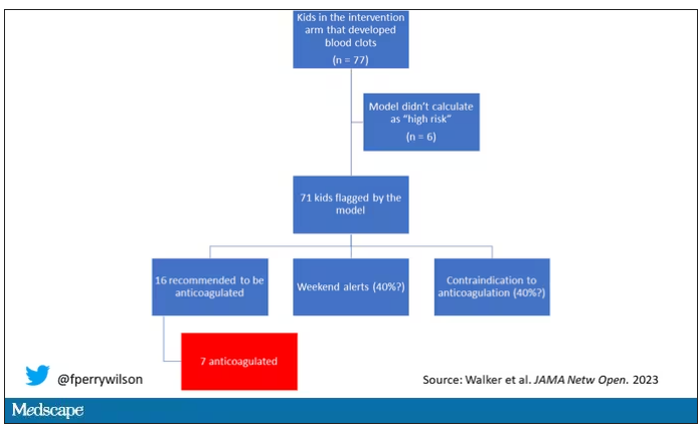
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I’m going to talk to you about a study at the cutting edge of modern medicine, one that uses an artificial intelligence (AI) model to guide care. But before I do, I need to take you back to the late Bronze Age, to a city located on the coast of what is now Turkey.
Troy’s towering walls made it seem unassailable, but that would not stop the Achaeans and their fleet of black ships from making landfall, and, after a siege, destroying the city. The destruction of Troy, as told in the Iliad and the Aeneid, was foretold by Cassandra, the daughter of King Priam and Priestess of Troy.
Cassandra had been given the gift of prophecy by the god Apollo in exchange for her favors. But after the gift was bestowed, she rejected the bright god and, in his rage, he added a curse to her blessing: that no one would ever believe her prophecies.
Thus it was that when her brother Paris set off to Sparta to abduct Helen, she warned him that his actions would lead to the downfall of their great city. He, of course, ignored her.
And you know the rest of the story.
Why am I telling you the story of Cassandra of Troy when we’re supposed to be talking about AI in medicine? Because AI has a major Cassandra problem.
The recent history of AI, and particularly the subset of AI known as machine learning in medicine, has been characterized by an accuracy arms race.
The electronic health record allows for the collection of volumes of data orders of magnitude greater than what we have ever been able to collect before. And all that data can be crunched by various algorithms to make predictions about, well, anything – whether a patient will be transferred to the intensive care unit, whether a GI bleed will need an intervention, whether someone will die in the next year.
Studies in this area tend to rely on retrospective datasets, and as time has gone on, better algorithms and more data have led to better and better predictions. In some simpler cases, machine-learning models have achieved near-perfect accuracy – Cassandra-level accuracy – as in the reading of chest x-rays for pneumonia, for example.
But as Cassandra teaches us, even perfect prediction is useless if no one believes you, if they don’t change their behavior. And this is the central problem of AI in medicine today. Many people are focusing on accuracy of the prediction but have forgotten that high accuracy is just table stakes for an AI model to be useful. It has to not only be accurate, but its use also has to change outcomes for patients. We need to be able to save Troy.
The best way to determine whether an AI model will help patients is to treat a model like we treat a new medication and evaluate it through a randomized trial. That’s what researchers, led by Shannon Walker of Vanderbilt University, Nashville, Tenn., did in a paper appearing in JAMA Network Open.
The model in question was one that predicted venous thromboembolism – blood clots – in hospitalized children. The model took in a variety of data points from the health record: a history of blood clot, history of cancer, presence of a central line, a variety of lab values. And the predictive model was very good – maybe not Cassandra good, but it achieved an AUC of 0.90, which means it had very high accuracy.
But again, accuracy is just table stakes.
The authors deployed the model in the live health record and recorded the results. For half of the kids, that was all that happened; no one actually saw the predictions. For those randomized to the intervention, the hematology team would be notified when the risk for clot was calculated to be greater than 2.5%. The hematology team would then contact the primary team to discuss prophylactic anticoagulation.
This is an elegant approach.
Let’s start with those table stakes – accuracy. The predictions were, by and large, pretty accurate in this trial. Of the 135 kids who developed blood clots, 121 had been flagged by the model in advance. That’s about 90%. The model flagged about 10% of kids who didn’t get a blood clot as well, but that’s not entirely surprising since the threshold for flagging was a 2.5% risk.
Given that the model preidentified almost every kid who would go on to develop a blood clot, it would make sense that kids randomized to the intervention would do better; after all, Cassandra was calling out her warnings.
But those kids didn’t do better. The rate of blood clot was no different between the group that used the accurate prediction model and the group that did not.
Why? Why does the use of an accurate model not necessarily improve outcomes?
First of all, a warning must lead to some change in management. Indeed, the kids in the intervention group were more likely to receive anticoagulation, but barely so. There were lots of reasons for this: physician preference, imminent discharge, active bleeding, and so on.
But let’s take a look at the 77 kids in the intervention arm who developed blood clots, because I think this is an instructive analysis.
Six of them did not meet the 2.5% threshold criteria, a case where the model missed its mark. Again, accuracy is table stakes.
Of the remaining 71, only 16 got a recommendation from the hematologist to start anticoagulation. Why not more? Well, the model identified some of the high-risk kids on the weekend, and it seems that the study team did not contact treatment teams during that time. That may account for about 40% of these cases. The remainder had some contraindication to anticoagulation.
Most tellingly, of the 16 who did get a recommendation to start anticoagulation, the recommendation was followed in only seven patients.
This is the gap between accurate prediction and the ability to change outcomes for patients. A prediction is useless if it is wrong, for sure. But it’s also useless if you don’t tell anyone about it. It’s useless if you tell someone but they can’t do anything about it. And it’s useless if they could do something about it but choose not to.
That’s the gulf that these models need to cross at this point. So, the next time some slick company tells you how accurate their AI model is, ask them if accuracy is really the most important thing. If they say, “Well, yes, of course,” then tell them about Cassandra.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.



