User login
Simple changes shorten door-to-insulin time for diabetic ketoacidosis
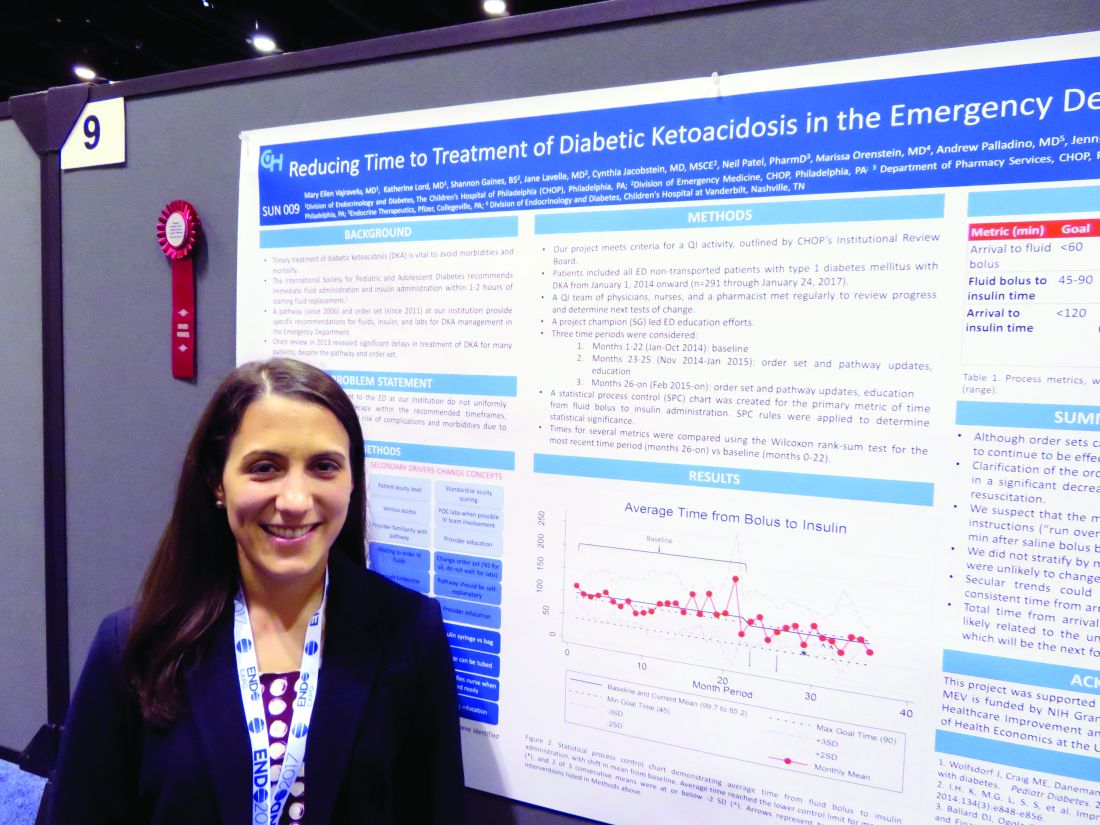
ORLANDO – A few simple adjustments in standing orders shaved 20 minutes off the time one emergency department took to deliver insulin to pediatric patients with diabetic ketoacidosis, according to Mary Ellen Vajravelu, MD.
Door-to-insulin time decreased from a mean of 168 minutes to 146 minutes after the changes went into effect. Most of the time was saved in the period between starting the initial fluid bolus and starting the insulin, she said during an interview at the annual meeting of the Endocrine Society.
A single patient’s experience inspired the quality improvement project.
“A couple of years ago, we saw a patient in the emergency department with diabetic ketoacidosis who had to wait more than 6 hours before insulin was started,” she explained. “Fortunately, there were no adverse clinical outcomes from that,” but the case signaled that the treatment process needed some streamlining.
The International Society for Pediatric and Adolescent Diabetes recommends immediate fluid administration and insulin within 1-2 hours of starting fluid replacement.
The hospital had been following in-house recommendations for fluids, insulin, and labs for patients with diabetic ketoacidosis that were established in 2006 and 2011. When they examined a baseline cohort of patients treated from January to October 2014, they immediately saw that those order sets were not achieving the recommended times.
“We went back and looked at our cases and saw that overall, it was taking more than 2 hours from the time of arrival for these patients to get their insulin,” Dr. Vajravelu said.
She and her colleagues formed a quality improvement team consisting of physicians, nurses, and a pharmacist. They broke down the wait time into three components: arrival to fluid bolus, fluid bolus start to insulin start, and the overall arrival-to-insulin time. They then set specific time goals for each of those periods: giving fluids within 60 minutes of arrival, starting insulin 45-90 minutes from the bolus, and an overall door-to-insulin time of less than 120 minutes.
The team tried to determine key clinical drivers that were affecting these times. One of the first findings was a delay in diagnosing diabetic ketoacidosis (DKA). This was driven by lack of consistency in scoring patient acuity and problems with venous access.
“To address this, we standardized acuity scoring, got an IV team involved, and used point-of-care labs when possible, and educated our providers,” Dr. Vajravelu said.
The team also found that there were delays in ordering the insulin. These were driven by long wait times for intravenous fluids, by waiting for endocrinology consults, and by providers not being familiar with the DKA order sets.
To address these issues, the team changed the order set to use normal saline for all suspected DKA patients without waiting for lab results. The old orders called for delivering the fluid over a 1-hour period; that was changed to running it over 30 minutes, Dr. Vajravelu said. “That shaved off half an hour right there.”
The team examined reasons that insulin infusion was delayed. Pharmacy orders were a big part of this problem, one of which was the way the insulin was sent up from the pharmacy in a bag. “We changed that to being delivered in a syringe and tube system,” she explained.
They also changed the standing order for when insulin was started, from 60 minutes after fluid to 45 minutes.
These changes were instituted in November 2014, and the team trained clinicians on them through January 2015. Since then, the team has reassessed the situation several times, tracking 291 patients in all. They saw improvement in all three time periods, which was significant in two of them.
Arrival to fluid bolus time dropped significantly, from 55 minutes to 53 minutes – an improvement, even though the baseline time was still within the goal. Fluid bolus-to-insulin time fell significantly as well, from 96 minutes to 74 minutes – well within the new goal.
Overall arrival-to-insulin time thus decreased, from 168 minutes to 146 minutes. However, Dr. Vajravelu said, this is still longer than their goal of less than 120 minutes from door to treatment.
“We are now going back to try and see just where the delay still is,” she said. “It may be getting IV access; it may be labs. We need to tease that out.”
Fortunately, the consistent delays in treatment haven’t resulted in any adverse clinical outcomes.
“I think the biggest benefit we will see will be saving time and money in the emergency department,” Dr. Vajravelu predicted. “We hope to get patients out of there faster, getting them admitted into the hospital and treated in the appropriate department. Speeding up care and reducing waste go together in that sense.”
Dr. Vajravelu had no relevant financial disclosures.
[email protected]
On Twitter @Alz_gal
ORLANDO – A few simple adjustments in standing orders shaved 20 minutes off the time one emergency department took to deliver insulin to pediatric patients with diabetic ketoacidosis, according to Mary Ellen Vajravelu, MD.
Door-to-insulin time decreased from a mean of 168 minutes to 146 minutes after the changes went into effect. Most of the time was saved in the period between starting the initial fluid bolus and starting the insulin, she said during an interview at the annual meeting of the Endocrine Society.
A single patient’s experience inspired the quality improvement project.
“A couple of years ago, we saw a patient in the emergency department with diabetic ketoacidosis who had to wait more than 6 hours before insulin was started,” she explained. “Fortunately, there were no adverse clinical outcomes from that,” but the case signaled that the treatment process needed some streamlining.
The International Society for Pediatric and Adolescent Diabetes recommends immediate fluid administration and insulin within 1-2 hours of starting fluid replacement.
The hospital had been following in-house recommendations for fluids, insulin, and labs for patients with diabetic ketoacidosis that were established in 2006 and 2011. When they examined a baseline cohort of patients treated from January to October 2014, they immediately saw that those order sets were not achieving the recommended times.
“We went back and looked at our cases and saw that overall, it was taking more than 2 hours from the time of arrival for these patients to get their insulin,” Dr. Vajravelu said.
She and her colleagues formed a quality improvement team consisting of physicians, nurses, and a pharmacist. They broke down the wait time into three components: arrival to fluid bolus, fluid bolus start to insulin start, and the overall arrival-to-insulin time. They then set specific time goals for each of those periods: giving fluids within 60 minutes of arrival, starting insulin 45-90 minutes from the bolus, and an overall door-to-insulin time of less than 120 minutes.
The team tried to determine key clinical drivers that were affecting these times. One of the first findings was a delay in diagnosing diabetic ketoacidosis (DKA). This was driven by lack of consistency in scoring patient acuity and problems with venous access.
“To address this, we standardized acuity scoring, got an IV team involved, and used point-of-care labs when possible, and educated our providers,” Dr. Vajravelu said.
The team also found that there were delays in ordering the insulin. These were driven by long wait times for intravenous fluids, by waiting for endocrinology consults, and by providers not being familiar with the DKA order sets.
To address these issues, the team changed the order set to use normal saline for all suspected DKA patients without waiting for lab results. The old orders called for delivering the fluid over a 1-hour period; that was changed to running it over 30 minutes, Dr. Vajravelu said. “That shaved off half an hour right there.”
The team examined reasons that insulin infusion was delayed. Pharmacy orders were a big part of this problem, one of which was the way the insulin was sent up from the pharmacy in a bag. “We changed that to being delivered in a syringe and tube system,” she explained.
They also changed the standing order for when insulin was started, from 60 minutes after fluid to 45 minutes.
These changes were instituted in November 2014, and the team trained clinicians on them through January 2015. Since then, the team has reassessed the situation several times, tracking 291 patients in all. They saw improvement in all three time periods, which was significant in two of them.
Arrival to fluid bolus time dropped significantly, from 55 minutes to 53 minutes – an improvement, even though the baseline time was still within the goal. Fluid bolus-to-insulin time fell significantly as well, from 96 minutes to 74 minutes – well within the new goal.
Overall arrival-to-insulin time thus decreased, from 168 minutes to 146 minutes. However, Dr. Vajravelu said, this is still longer than their goal of less than 120 minutes from door to treatment.
“We are now going back to try and see just where the delay still is,” she said. “It may be getting IV access; it may be labs. We need to tease that out.”
Fortunately, the consistent delays in treatment haven’t resulted in any adverse clinical outcomes.
“I think the biggest benefit we will see will be saving time and money in the emergency department,” Dr. Vajravelu predicted. “We hope to get patients out of there faster, getting them admitted into the hospital and treated in the appropriate department. Speeding up care and reducing waste go together in that sense.”
Dr. Vajravelu had no relevant financial disclosures.
[email protected]
On Twitter @Alz_gal
ORLANDO – A few simple adjustments in standing orders shaved 20 minutes off the time one emergency department took to deliver insulin to pediatric patients with diabetic ketoacidosis, according to Mary Ellen Vajravelu, MD.
Door-to-insulin time decreased from a mean of 168 minutes to 146 minutes after the changes went into effect. Most of the time was saved in the period between starting the initial fluid bolus and starting the insulin, she said during an interview at the annual meeting of the Endocrine Society.
A single patient’s experience inspired the quality improvement project.
“A couple of years ago, we saw a patient in the emergency department with diabetic ketoacidosis who had to wait more than 6 hours before insulin was started,” she explained. “Fortunately, there were no adverse clinical outcomes from that,” but the case signaled that the treatment process needed some streamlining.
The International Society for Pediatric and Adolescent Diabetes recommends immediate fluid administration and insulin within 1-2 hours of starting fluid replacement.
The hospital had been following in-house recommendations for fluids, insulin, and labs for patients with diabetic ketoacidosis that were established in 2006 and 2011. When they examined a baseline cohort of patients treated from January to October 2014, they immediately saw that those order sets were not achieving the recommended times.
“We went back and looked at our cases and saw that overall, it was taking more than 2 hours from the time of arrival for these patients to get their insulin,” Dr. Vajravelu said.
She and her colleagues formed a quality improvement team consisting of physicians, nurses, and a pharmacist. They broke down the wait time into three components: arrival to fluid bolus, fluid bolus start to insulin start, and the overall arrival-to-insulin time. They then set specific time goals for each of those periods: giving fluids within 60 minutes of arrival, starting insulin 45-90 minutes from the bolus, and an overall door-to-insulin time of less than 120 minutes.
The team tried to determine key clinical drivers that were affecting these times. One of the first findings was a delay in diagnosing diabetic ketoacidosis (DKA). This was driven by lack of consistency in scoring patient acuity and problems with venous access.
“To address this, we standardized acuity scoring, got an IV team involved, and used point-of-care labs when possible, and educated our providers,” Dr. Vajravelu said.
The team also found that there were delays in ordering the insulin. These were driven by long wait times for intravenous fluids, by waiting for endocrinology consults, and by providers not being familiar with the DKA order sets.
To address these issues, the team changed the order set to use normal saline for all suspected DKA patients without waiting for lab results. The old orders called for delivering the fluid over a 1-hour period; that was changed to running it over 30 minutes, Dr. Vajravelu said. “That shaved off half an hour right there.”
The team examined reasons that insulin infusion was delayed. Pharmacy orders were a big part of this problem, one of which was the way the insulin was sent up from the pharmacy in a bag. “We changed that to being delivered in a syringe and tube system,” she explained.
They also changed the standing order for when insulin was started, from 60 minutes after fluid to 45 minutes.
These changes were instituted in November 2014, and the team trained clinicians on them through January 2015. Since then, the team has reassessed the situation several times, tracking 291 patients in all. They saw improvement in all three time periods, which was significant in two of them.
Arrival to fluid bolus time dropped significantly, from 55 minutes to 53 minutes – an improvement, even though the baseline time was still within the goal. Fluid bolus-to-insulin time fell significantly as well, from 96 minutes to 74 minutes – well within the new goal.
Overall arrival-to-insulin time thus decreased, from 168 minutes to 146 minutes. However, Dr. Vajravelu said, this is still longer than their goal of less than 120 minutes from door to treatment.
“We are now going back to try and see just where the delay still is,” she said. “It may be getting IV access; it may be labs. We need to tease that out.”
Fortunately, the consistent delays in treatment haven’t resulted in any adverse clinical outcomes.
“I think the biggest benefit we will see will be saving time and money in the emergency department,” Dr. Vajravelu predicted. “We hope to get patients out of there faster, getting them admitted into the hospital and treated in the appropriate department. Speeding up care and reducing waste go together in that sense.”
Dr. Vajravelu had no relevant financial disclosures.
[email protected]
On Twitter @Alz_gal
AT ENDO 2017
Key clinical point:
Major finding: Overall door-to-insulin time decreased from 168 minutes to 146 minutes.
Data source: A 2-year quality improvement project involving 291 patients.
Disclosures: Dr. Vajravelu had no relevant financial disclosures.
Using genetics to predict CRC outcomes
BOSTON – Gastroenterologists and GI surgeons had the opportunity to learn more about “Predicting Clinical Outcomes in CRC with Genetics” at the 2017 AGA Tech Summit, sponsored by the AGA Center for GI Innovation and Technology.
“The topic is extremely significant for two reasons,” said presenter C. Richard Boland, MD, of the University of California in San Diego. “First, we are now able to test colorectal cancer (CRC) for genetic alternations – allowing us to gauge prognosis and, eventually, target specific therapies to the actual tumor,” he said in an interview in advance of the meeting. “Second, the issue of detecting germline mutations in familial lines of CRC is an important factor in diagnosis.”
“All cancers are genetic diseases, developing as a consequence of cumulative mutations. Each cancer is different, both in terms of total mutational burden, as well as the specific mutations that ‘drive’ the tumor,” explained Dr. Boland. “We will be able to stop tumor growth by inhibiting the driver mutation, once we know what that driver is. This will allow us to make more accurate prognoses and predict tumor responses to specific therapies. The hard part will be having effective therapies for each driver mutation.”
The presentation began with a discussion of optimizing CRC care using genetics, predictive tests (for defective DNA mismatch repair – dMMR), specific tumor mutations and blood tests (for miRNAs or other noncoding RNAs). It also covered the sensitivity of immunohistochemistry (IHC) for dMMR and potential problems with IHC.
Then it detailed an innovative technical proposal whose elements include developing a next-generation sequencing platform for CRC and performing microsatellite instability and other mutational testing for specific genes as test-guided therapies emerge. The session also provided an overview of germline mutation testing, including a description of genes that cause hereditary CRC, are on Myriad’s myRisk panel, and are included on Ambry’s 8 Test Panels.
“We think about 3% or so of CRCs have a strong genetic basis, but probably a lot more have weaker genetic bases,” said Dr. Boland. “When we find strongly penetrant germline mutations, it permits case finding and preventive intervention in family members. It also tells us a lot about how to care for the index individual, since all of these familial syndromes have a wide range of effects on patients.
“The technology driving this type of patient assessment is rapidly developing, and making it easier to find these mutations” he continued. “But it is also confusing when we find unexpected mutations in people who don’t seem to fit the classic disease descriptions.”
Attendees were taught why genetic test of CRCs can help direct patient care, how the tests can be efficiently done using a new diagnostic platform, and the state of germline testing and its interpretation.
Dr. Boland has given a lecture for Ambry Genetics (a genetics testing company) and may give others in the future.
BOSTON – Gastroenterologists and GI surgeons had the opportunity to learn more about “Predicting Clinical Outcomes in CRC with Genetics” at the 2017 AGA Tech Summit, sponsored by the AGA Center for GI Innovation and Technology.
“The topic is extremely significant for two reasons,” said presenter C. Richard Boland, MD, of the University of California in San Diego. “First, we are now able to test colorectal cancer (CRC) for genetic alternations – allowing us to gauge prognosis and, eventually, target specific therapies to the actual tumor,” he said in an interview in advance of the meeting. “Second, the issue of detecting germline mutations in familial lines of CRC is an important factor in diagnosis.”
“All cancers are genetic diseases, developing as a consequence of cumulative mutations. Each cancer is different, both in terms of total mutational burden, as well as the specific mutations that ‘drive’ the tumor,” explained Dr. Boland. “We will be able to stop tumor growth by inhibiting the driver mutation, once we know what that driver is. This will allow us to make more accurate prognoses and predict tumor responses to specific therapies. The hard part will be having effective therapies for each driver mutation.”
The presentation began with a discussion of optimizing CRC care using genetics, predictive tests (for defective DNA mismatch repair – dMMR), specific tumor mutations and blood tests (for miRNAs or other noncoding RNAs). It also covered the sensitivity of immunohistochemistry (IHC) for dMMR and potential problems with IHC.
Then it detailed an innovative technical proposal whose elements include developing a next-generation sequencing platform for CRC and performing microsatellite instability and other mutational testing for specific genes as test-guided therapies emerge. The session also provided an overview of germline mutation testing, including a description of genes that cause hereditary CRC, are on Myriad’s myRisk panel, and are included on Ambry’s 8 Test Panels.
“We think about 3% or so of CRCs have a strong genetic basis, but probably a lot more have weaker genetic bases,” said Dr. Boland. “When we find strongly penetrant germline mutations, it permits case finding and preventive intervention in family members. It also tells us a lot about how to care for the index individual, since all of these familial syndromes have a wide range of effects on patients.
“The technology driving this type of patient assessment is rapidly developing, and making it easier to find these mutations” he continued. “But it is also confusing when we find unexpected mutations in people who don’t seem to fit the classic disease descriptions.”
Attendees were taught why genetic test of CRCs can help direct patient care, how the tests can be efficiently done using a new diagnostic platform, and the state of germline testing and its interpretation.
Dr. Boland has given a lecture for Ambry Genetics (a genetics testing company) and may give others in the future.
BOSTON – Gastroenterologists and GI surgeons had the opportunity to learn more about “Predicting Clinical Outcomes in CRC with Genetics” at the 2017 AGA Tech Summit, sponsored by the AGA Center for GI Innovation and Technology.
“The topic is extremely significant for two reasons,” said presenter C. Richard Boland, MD, of the University of California in San Diego. “First, we are now able to test colorectal cancer (CRC) for genetic alternations – allowing us to gauge prognosis and, eventually, target specific therapies to the actual tumor,” he said in an interview in advance of the meeting. “Second, the issue of detecting germline mutations in familial lines of CRC is an important factor in diagnosis.”
“All cancers are genetic diseases, developing as a consequence of cumulative mutations. Each cancer is different, both in terms of total mutational burden, as well as the specific mutations that ‘drive’ the tumor,” explained Dr. Boland. “We will be able to stop tumor growth by inhibiting the driver mutation, once we know what that driver is. This will allow us to make more accurate prognoses and predict tumor responses to specific therapies. The hard part will be having effective therapies for each driver mutation.”
The presentation began with a discussion of optimizing CRC care using genetics, predictive tests (for defective DNA mismatch repair – dMMR), specific tumor mutations and blood tests (for miRNAs or other noncoding RNAs). It also covered the sensitivity of immunohistochemistry (IHC) for dMMR and potential problems with IHC.
Then it detailed an innovative technical proposal whose elements include developing a next-generation sequencing platform for CRC and performing microsatellite instability and other mutational testing for specific genes as test-guided therapies emerge. The session also provided an overview of germline mutation testing, including a description of genes that cause hereditary CRC, are on Myriad’s myRisk panel, and are included on Ambry’s 8 Test Panels.
“We think about 3% or so of CRCs have a strong genetic basis, but probably a lot more have weaker genetic bases,” said Dr. Boland. “When we find strongly penetrant germline mutations, it permits case finding and preventive intervention in family members. It also tells us a lot about how to care for the index individual, since all of these familial syndromes have a wide range of effects on patients.
“The technology driving this type of patient assessment is rapidly developing, and making it easier to find these mutations” he continued. “But it is also confusing when we find unexpected mutations in people who don’t seem to fit the classic disease descriptions.”
Attendees were taught why genetic test of CRCs can help direct patient care, how the tests can be efficiently done using a new diagnostic platform, and the state of germline testing and its interpretation.
Dr. Boland has given a lecture for Ambry Genetics (a genetics testing company) and may give others in the future.
FROM THE 2017 AGA TECH SUMMIT
Crohn’s hospitalizations up significantly … or not
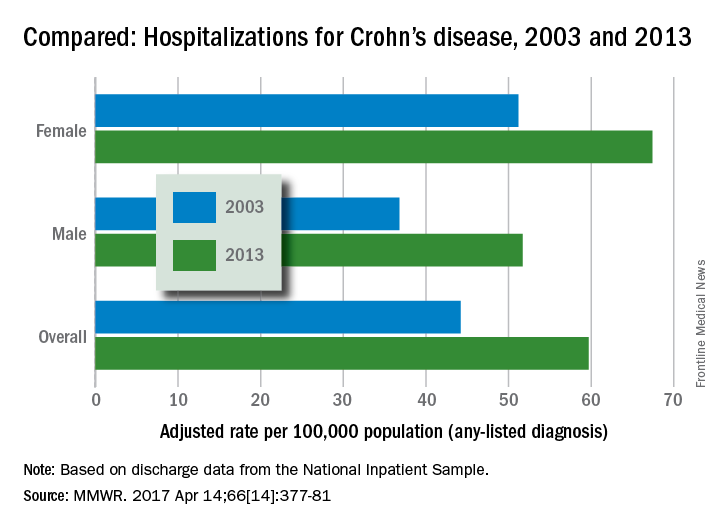
Hospitalizations for Crohn’s disease were up by a statistically significant 35% from 2003 to 2013 … or they were up just 5%, according to the Centers for Disease Control and Prevention.
It depends on how you look at it. In 2013, age-adjusted hospitalization was 59.7 stays per 100,000 population for Crohn’s as any-listed diagnosis – indicating that patients had Crohn’s disease but that it was not necessarily the main reason they were being hospitalized – compared with 44.2 per 100,000 in 2003. That’s an increase of 35%, the CDC said (MMWR. 2017 Apr 14;66[14]:377-81).
Hospitalizations with Crohn’s as the first-listed diagnosis – making it the main reason for the admission – were up 5% over the same period: 18.2 stays per 100,000 pop. in 2003 and 19.1 per 100,000 in 2013. That increase is not statistically significant, the CDC noted.
The increase in any-listed diagnosis “might represent greater physician awareness and diagnosis of Crohn’s disease or more complete coding of secondary diagnoses by physicians,” CDC investigators suggested.
Increases in hospitalizations for any-listed Crohn’s were significant for both men (40.5%) and women (31.6%). For first-listed Crohn’s, women saw a slight but not significant decrease of 1% while men had a significant 14.5% increase, according to the report, which was based on data from the National Inpatient Sample.
Hospitalizations for Crohn’s disease were up by a statistically significant 35% from 2003 to 2013 … or they were up just 5%, according to the Centers for Disease Control and Prevention.
It depends on how you look at it. In 2013, age-adjusted hospitalization was 59.7 stays per 100,000 population for Crohn’s as any-listed diagnosis – indicating that patients had Crohn’s disease but that it was not necessarily the main reason they were being hospitalized – compared with 44.2 per 100,000 in 2003. That’s an increase of 35%, the CDC said (MMWR. 2017 Apr 14;66[14]:377-81).
Hospitalizations with Crohn’s as the first-listed diagnosis – making it the main reason for the admission – were up 5% over the same period: 18.2 stays per 100,000 pop. in 2003 and 19.1 per 100,000 in 2013. That increase is not statistically significant, the CDC noted.
The increase in any-listed diagnosis “might represent greater physician awareness and diagnosis of Crohn’s disease or more complete coding of secondary diagnoses by physicians,” CDC investigators suggested.
Increases in hospitalizations for any-listed Crohn’s were significant for both men (40.5%) and women (31.6%). For first-listed Crohn’s, women saw a slight but not significant decrease of 1% while men had a significant 14.5% increase, according to the report, which was based on data from the National Inpatient Sample.
Hospitalizations for Crohn’s disease were up by a statistically significant 35% from 2003 to 2013 … or they were up just 5%, according to the Centers for Disease Control and Prevention.
It depends on how you look at it. In 2013, age-adjusted hospitalization was 59.7 stays per 100,000 population for Crohn’s as any-listed diagnosis – indicating that patients had Crohn’s disease but that it was not necessarily the main reason they were being hospitalized – compared with 44.2 per 100,000 in 2003. That’s an increase of 35%, the CDC said (MMWR. 2017 Apr 14;66[14]:377-81).
Hospitalizations with Crohn’s as the first-listed diagnosis – making it the main reason for the admission – were up 5% over the same period: 18.2 stays per 100,000 pop. in 2003 and 19.1 per 100,000 in 2013. That increase is not statistically significant, the CDC noted.
The increase in any-listed diagnosis “might represent greater physician awareness and diagnosis of Crohn’s disease or more complete coding of secondary diagnoses by physicians,” CDC investigators suggested.
Increases in hospitalizations for any-listed Crohn’s were significant for both men (40.5%) and women (31.6%). For first-listed Crohn’s, women saw a slight but not significant decrease of 1% while men had a significant 14.5% increase, according to the report, which was based on data from the National Inpatient Sample.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Medicaid reform: Work-based waivers may not fly
The Trump administration may not be able to successfully implement the work requirements and other Medicaid eligibility caveats proffered by Health and Human Services Secretary Tom Price, MD, according to Jane Perkins, legal director for the National Health Law Program.
In March, Secretary Price and Seema Verma, administrator of the Centers for Medicare & Medicaid Services, wrote to state governors, letting them know that the HHS would support states’ efforts to increase employment, community engagement, and work requirements for Medicaid recipients. The letter also was supportive of aligning Medicaid benefits with private insurance via alternative benefits, cost-sharing, and premium payments.
High mandatory premiums, cost sharing, lifetime limits, and drug testing “are of concern to us,” Ms. Perkins said at an April 13 press briefing. “They really change the complexion of Medicaid and Medicaid coverage for low-income people.”
These requirements “are not typically seen in Medicaid programs,” she said.
While Section 1115 of the Social Security Act “allows states to test novel approaches to providing medical assistance” via Medicaid waivers, it does not allow the HHS or the states to “ignore congressional mandates; to cut eligibility, services, or provider payments; or to use section 1115 to save money,” according to an issue brief by Ms. Perkins.
When asked how work requirements harm people, Ms. Perkins responded that adding a work requirement to Medicaid eligibility gets things “backwards,” because a sick person needs health care before being able to return to work.
The work requirement would not save states much money, as nearly 8 in 10 adults on Medicaid are in a household that includes a worker and 59% of recipients work themselves, according to a Kaiser Family Foundation study. The adults affected by the work requirement would make up only a drop in the ocean of Medicaid spending. About two-thirds of that spending goes toward senior citizens, people with disabilities, children, and people in long term care, according to projections from the Congressional Budget Office.
The National Health Law program, which advocates for low-income Medicaid recipients, is following the waivers state-by-state with a network of lawyers who work with people with disabilities in each state.
“With this new openness to flexibility, we are certainly watching what is going on in the states,” Ms. Perkins said.
The Trump administration may not be able to successfully implement the work requirements and other Medicaid eligibility caveats proffered by Health and Human Services Secretary Tom Price, MD, according to Jane Perkins, legal director for the National Health Law Program.
In March, Secretary Price and Seema Verma, administrator of the Centers for Medicare & Medicaid Services, wrote to state governors, letting them know that the HHS would support states’ efforts to increase employment, community engagement, and work requirements for Medicaid recipients. The letter also was supportive of aligning Medicaid benefits with private insurance via alternative benefits, cost-sharing, and premium payments.
High mandatory premiums, cost sharing, lifetime limits, and drug testing “are of concern to us,” Ms. Perkins said at an April 13 press briefing. “They really change the complexion of Medicaid and Medicaid coverage for low-income people.”
These requirements “are not typically seen in Medicaid programs,” she said.
While Section 1115 of the Social Security Act “allows states to test novel approaches to providing medical assistance” via Medicaid waivers, it does not allow the HHS or the states to “ignore congressional mandates; to cut eligibility, services, or provider payments; or to use section 1115 to save money,” according to an issue brief by Ms. Perkins.
When asked how work requirements harm people, Ms. Perkins responded that adding a work requirement to Medicaid eligibility gets things “backwards,” because a sick person needs health care before being able to return to work.
The work requirement would not save states much money, as nearly 8 in 10 adults on Medicaid are in a household that includes a worker and 59% of recipients work themselves, according to a Kaiser Family Foundation study. The adults affected by the work requirement would make up only a drop in the ocean of Medicaid spending. About two-thirds of that spending goes toward senior citizens, people with disabilities, children, and people in long term care, according to projections from the Congressional Budget Office.
The National Health Law program, which advocates for low-income Medicaid recipients, is following the waivers state-by-state with a network of lawyers who work with people with disabilities in each state.
“With this new openness to flexibility, we are certainly watching what is going on in the states,” Ms. Perkins said.
The Trump administration may not be able to successfully implement the work requirements and other Medicaid eligibility caveats proffered by Health and Human Services Secretary Tom Price, MD, according to Jane Perkins, legal director for the National Health Law Program.
In March, Secretary Price and Seema Verma, administrator of the Centers for Medicare & Medicaid Services, wrote to state governors, letting them know that the HHS would support states’ efforts to increase employment, community engagement, and work requirements for Medicaid recipients. The letter also was supportive of aligning Medicaid benefits with private insurance via alternative benefits, cost-sharing, and premium payments.
High mandatory premiums, cost sharing, lifetime limits, and drug testing “are of concern to us,” Ms. Perkins said at an April 13 press briefing. “They really change the complexion of Medicaid and Medicaid coverage for low-income people.”
These requirements “are not typically seen in Medicaid programs,” she said.
While Section 1115 of the Social Security Act “allows states to test novel approaches to providing medical assistance” via Medicaid waivers, it does not allow the HHS or the states to “ignore congressional mandates; to cut eligibility, services, or provider payments; or to use section 1115 to save money,” according to an issue brief by Ms. Perkins.
When asked how work requirements harm people, Ms. Perkins responded that adding a work requirement to Medicaid eligibility gets things “backwards,” because a sick person needs health care before being able to return to work.
The work requirement would not save states much money, as nearly 8 in 10 adults on Medicaid are in a household that includes a worker and 59% of recipients work themselves, according to a Kaiser Family Foundation study. The adults affected by the work requirement would make up only a drop in the ocean of Medicaid spending. About two-thirds of that spending goes toward senior citizens, people with disabilities, children, and people in long term care, according to projections from the Congressional Budget Office.
The National Health Law program, which advocates for low-income Medicaid recipients, is following the waivers state-by-state with a network of lawyers who work with people with disabilities in each state.
“With this new openness to flexibility, we are certainly watching what is going on in the states,” Ms. Perkins said.
PTSD linked to cognitive decline in middle-aged women
SAN FRANCISCO – Posttraumatic stress disorder is associated with worse cognitive function in middle-aged women, Jennifer A. Sumner, PhD, reported at the annual conference of the Anxiety and Depression Association of America.
The strongest link between PTSD and impaired cognitive function found in this new analysis from the Nurses’ Health Study 2 was observed among those women with elevated PTSD symptoms indicative of probable PTSD and comorbid depression, added Dr. Sumner, a clinical psychologist affiliated with Columbia University in New York.
The findings are important, in part because they help fill a gender gap in PTSD research. The great majority of studies of PTSD and its long-term consequences for physical and mental health have focused on male military veterans. Yet PTSD is twice as common in women as in men and is on average more severe and causes more impairment in women as well.
Growing evidence suggests that brain aging and cognitive decline begin in midlife, long before the dysfunction becomes manifest as dementia. The Nurses’ Health Study 2 findings raise the testable hypothesis that PTSD might be a modifiable risk factor for early cognitive decline, Dr. Sumner observed.
The Nurses’ Health Study 2 is one of the largest ongoing longitudinal studies of women’s health in the world. Dr. Sumner’s analysis focused on 14,029 participants who in 2008 underwent assessment of lifetime exposure to 16 types of trauma included in the Brief Trauma Questionnaire, lifetime PTSD symptoms using the Short Screening Scale for PTSD, and current depressive symptoms via the Center for Epidemiologic Studies Depression Scale. The women then completed the Cogstate Brief Battery in 2014-2016. The Cogstate is a validated, sensitive, self-administered online cognitive battery that measures psychomotor speed, attention, working memory, and learning. It takes 10-15 minutes to complete.
Seventeen percent of women had both a lifetime history of exposure to trauma and probable PTSD based upon having four or more of the seven symptoms listed on the Short Screening Scale for PTSD. These women had, on average, a lower educational level, income, and self-assessed social standing, more medical comorbidities, and more depressive symptoms than the other subjects. However, in a multivariate analysis controlling for these potential confounders, the group with both trauma and PTSD performed significantly worse on the Cogstate measures of psychomotor speed/attention and learning/working memory than women with no trauma and no PTSD symptoms.
Similarly, the 17% of women with both probable PTSD and probable depression as defined by a score of 10 or more on the Center for Epidemiologic Studies Depression Scale demonstrated significantly worse cognitive function on the Cogstate, compared with women with neither condition.
In regard to potential mechanisms that might explain the observed association between PTSD and worse cognitive function, Dr. Sumner noted that other investigators have reported that PTSD is characterized by oxidative stress, systemic inflammation, and hypothalamic-pituitary-adrenal axis and neuroendocrine dysfunction. This could promote neuronal death. Moreover, PTSD also is associated with increased risk of poor health behaviors, including sleep disturbances, physical inactivity, obesity, and smoking, which also could contribute to cognitive decline down the line.
Dr. Sumner’s study, which was supported by the National Institutes of Health, recently has been published (Depress Anxiety. 2017 Apr;34[4]:356-66). She reported having no financial conflicts.
SAN FRANCISCO – Posttraumatic stress disorder is associated with worse cognitive function in middle-aged women, Jennifer A. Sumner, PhD, reported at the annual conference of the Anxiety and Depression Association of America.
The strongest link between PTSD and impaired cognitive function found in this new analysis from the Nurses’ Health Study 2 was observed among those women with elevated PTSD symptoms indicative of probable PTSD and comorbid depression, added Dr. Sumner, a clinical psychologist affiliated with Columbia University in New York.
The findings are important, in part because they help fill a gender gap in PTSD research. The great majority of studies of PTSD and its long-term consequences for physical and mental health have focused on male military veterans. Yet PTSD is twice as common in women as in men and is on average more severe and causes more impairment in women as well.
Growing evidence suggests that brain aging and cognitive decline begin in midlife, long before the dysfunction becomes manifest as dementia. The Nurses’ Health Study 2 findings raise the testable hypothesis that PTSD might be a modifiable risk factor for early cognitive decline, Dr. Sumner observed.
The Nurses’ Health Study 2 is one of the largest ongoing longitudinal studies of women’s health in the world. Dr. Sumner’s analysis focused on 14,029 participants who in 2008 underwent assessment of lifetime exposure to 16 types of trauma included in the Brief Trauma Questionnaire, lifetime PTSD symptoms using the Short Screening Scale for PTSD, and current depressive symptoms via the Center for Epidemiologic Studies Depression Scale. The women then completed the Cogstate Brief Battery in 2014-2016. The Cogstate is a validated, sensitive, self-administered online cognitive battery that measures psychomotor speed, attention, working memory, and learning. It takes 10-15 minutes to complete.
Seventeen percent of women had both a lifetime history of exposure to trauma and probable PTSD based upon having four or more of the seven symptoms listed on the Short Screening Scale for PTSD. These women had, on average, a lower educational level, income, and self-assessed social standing, more medical comorbidities, and more depressive symptoms than the other subjects. However, in a multivariate analysis controlling for these potential confounders, the group with both trauma and PTSD performed significantly worse on the Cogstate measures of psychomotor speed/attention and learning/working memory than women with no trauma and no PTSD symptoms.
Similarly, the 17% of women with both probable PTSD and probable depression as defined by a score of 10 or more on the Center for Epidemiologic Studies Depression Scale demonstrated significantly worse cognitive function on the Cogstate, compared with women with neither condition.
In regard to potential mechanisms that might explain the observed association between PTSD and worse cognitive function, Dr. Sumner noted that other investigators have reported that PTSD is characterized by oxidative stress, systemic inflammation, and hypothalamic-pituitary-adrenal axis and neuroendocrine dysfunction. This could promote neuronal death. Moreover, PTSD also is associated with increased risk of poor health behaviors, including sleep disturbances, physical inactivity, obesity, and smoking, which also could contribute to cognitive decline down the line.
Dr. Sumner’s study, which was supported by the National Institutes of Health, recently has been published (Depress Anxiety. 2017 Apr;34[4]:356-66). She reported having no financial conflicts.
SAN FRANCISCO – Posttraumatic stress disorder is associated with worse cognitive function in middle-aged women, Jennifer A. Sumner, PhD, reported at the annual conference of the Anxiety and Depression Association of America.
The strongest link between PTSD and impaired cognitive function found in this new analysis from the Nurses’ Health Study 2 was observed among those women with elevated PTSD symptoms indicative of probable PTSD and comorbid depression, added Dr. Sumner, a clinical psychologist affiliated with Columbia University in New York.
The findings are important, in part because they help fill a gender gap in PTSD research. The great majority of studies of PTSD and its long-term consequences for physical and mental health have focused on male military veterans. Yet PTSD is twice as common in women as in men and is on average more severe and causes more impairment in women as well.
Growing evidence suggests that brain aging and cognitive decline begin in midlife, long before the dysfunction becomes manifest as dementia. The Nurses’ Health Study 2 findings raise the testable hypothesis that PTSD might be a modifiable risk factor for early cognitive decline, Dr. Sumner observed.
The Nurses’ Health Study 2 is one of the largest ongoing longitudinal studies of women’s health in the world. Dr. Sumner’s analysis focused on 14,029 participants who in 2008 underwent assessment of lifetime exposure to 16 types of trauma included in the Brief Trauma Questionnaire, lifetime PTSD symptoms using the Short Screening Scale for PTSD, and current depressive symptoms via the Center for Epidemiologic Studies Depression Scale. The women then completed the Cogstate Brief Battery in 2014-2016. The Cogstate is a validated, sensitive, self-administered online cognitive battery that measures psychomotor speed, attention, working memory, and learning. It takes 10-15 minutes to complete.
Seventeen percent of women had both a lifetime history of exposure to trauma and probable PTSD based upon having four or more of the seven symptoms listed on the Short Screening Scale for PTSD. These women had, on average, a lower educational level, income, and self-assessed social standing, more medical comorbidities, and more depressive symptoms than the other subjects. However, in a multivariate analysis controlling for these potential confounders, the group with both trauma and PTSD performed significantly worse on the Cogstate measures of psychomotor speed/attention and learning/working memory than women with no trauma and no PTSD symptoms.
Similarly, the 17% of women with both probable PTSD and probable depression as defined by a score of 10 or more on the Center for Epidemiologic Studies Depression Scale demonstrated significantly worse cognitive function on the Cogstate, compared with women with neither condition.
In regard to potential mechanisms that might explain the observed association between PTSD and worse cognitive function, Dr. Sumner noted that other investigators have reported that PTSD is characterized by oxidative stress, systemic inflammation, and hypothalamic-pituitary-adrenal axis and neuroendocrine dysfunction. This could promote neuronal death. Moreover, PTSD also is associated with increased risk of poor health behaviors, including sleep disturbances, physical inactivity, obesity, and smoking, which also could contribute to cognitive decline down the line.
Dr. Sumner’s study, which was supported by the National Institutes of Health, recently has been published (Depress Anxiety. 2017 Apr;34[4]:356-66). She reported having no financial conflicts.
AT ANXIETY AND DEPRESSION CONFERENCE 2017
Key clinical point:
Major finding: A large group of middle-aged civilian women with a history of exposure to trauma at any point in their lives along with elevated PTSD symptoms scored significantly worse on a cognitive function battery than women with no history of trauma or PTSD symptoms.
Data source: This was an analysis of more than 14,000 middle-aged women participating in the ongoing longitudinal Nurses’ Health Study 2.
Disclosures: The study was supported by the National Institutes of Health. Dr. Sumner reported having no financial conflicts.
Measuring Patient Satisfaction: How Do Patients Perceive Quality of Care Delivered by Dermatologists?
Why the Affordable Care Act will be greatly modified
Health care rationing is an uncomfortable topic, but one that responsible Americans should discuss. In particular, physicians should consider this topic, because our typical response to health care is that “everything should be covered.”
Health care has always been rationed. Historically, in the United States, health care has been apportioned capitalistically; that is, those who could afford health care got it and the rest – particularly the poor – scrambled. In Europe and Canada, universal or government health care is rationed by long wait times, limited range of services, and formulary restrictions. It is important to note that in almost all countries with universal health care, there is still the option of quick access and more comprehensive service via a neighboring country (to the United States from Canada, for example) or a separate additional private insurance option (England, Germany, and most of Europe).
In the United States, the Affordable Care Act (ACA) changed the dynamic regarding insurance by adding mandates to cover preexisting conditions, mental health and substance abuse disorders, behavioral health treatment, prescription drugs, rehabilitative services, preventive care, and pediatric services including oral and vision care for children. In addition, adult children now can remain on their parents’ insurance plans until age 26. Also, when the Cadillac tax on expensive health insurance coverage was delayed until 2020, the excise payments were classified as tax deductible to the employer. Whew, did I leave anything out?
Advocates for the poor and those previously considered uninsurable were delighted by the ACA. The middle class was mollified with false reassurances that their premiums would go down, and that they could keep their current insurance plans and doctors.
Currently under the ACA, “poor” is generally defined as a reported income of 150% or less of the federal poverty level ($22,000-$45,000). The poor are eligible for health care that is very nearly free, according to Medicaid.gov. They are also protected from claims against their property. They effectively have first-dollar coverage, though they usually can’t see a specialist unless they go through the emergency department.
At age 65, Medicare takes over. Medicare is an insurance benefit workers contribute to for all their working lives. It doesn’t cover everything, but the relatively low $183 Part B deductible for office-based care sure takes the edge off. The average retirees collect three times what they put in, but they arguably could have made more if they had invested anywhere other than in the federal government.
So, the real crunch hits the middle class under age 65. Under the ACA, those earning up to $97,000 for a family of four get insurance subsidies to buy insurance on the exchanges. The real problem arises when a high deductible – the maximum for 2017 is between $13,000 and $14,000 – is combined with that level of income. There are over 20 million health care savings accounts in the United States, which ameliorate the cost for some, but health care savings accounts do not cover the entire deductible, and many cannot afford to self-fund them if their employer does not fund it for them.People simply do not have that kind of money put away in a disaster fund, and a major medical expense can easily wipe them out. If possible, they will ignore a medical problem; and if they can’t, they could risk financial toxicity by running up high-interest credit cards – with the predictable spiral of debt, collection agencies, credit ratings ruined, wages garnished, and even their homes repossessed. They aren’t going to use their health care coverage unless forced to by a disaster – or worse for the system, they have been saving their elective surgeries up and are ready for a major tune-up. The long list of mandated benefits has triggered a meltdown in the exchange markets and huge increases in premiums and deductibles for all insurance buyers. Who is going to pay for it? To quote the late Russell B. Long, former U.S. Senator:
Don’t tax you,
Don’t tax me,
Tax that fellow behind the tree.
Raising taxes and pouring even more money into the system might be a solution, but this is not likely to happen.
In the United States, the rationing of health care has been shifted from the poor to the middle class, and the middle class doesn’t like it. The situation will only change when the howling from the middle class is louder than the wailing from the formerly uninsurable and poor. As insurance premiums and deductibles continue to rocket up, I expect the howling will get louder.
So, either we agree to let insurance premiums continue to increase, our state budgets to explode, our taxes to go up, or we pare down the list of mandated benefits.
I am going on the record stating that we simply cannot afford “medical everything” for everybody. I think we should tighten up the benefit list considerably. What do you think?
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
Health care rationing is an uncomfortable topic, but one that responsible Americans should discuss. In particular, physicians should consider this topic, because our typical response to health care is that “everything should be covered.”
Health care has always been rationed. Historically, in the United States, health care has been apportioned capitalistically; that is, those who could afford health care got it and the rest – particularly the poor – scrambled. In Europe and Canada, universal or government health care is rationed by long wait times, limited range of services, and formulary restrictions. It is important to note that in almost all countries with universal health care, there is still the option of quick access and more comprehensive service via a neighboring country (to the United States from Canada, for example) or a separate additional private insurance option (England, Germany, and most of Europe).
In the United States, the Affordable Care Act (ACA) changed the dynamic regarding insurance by adding mandates to cover preexisting conditions, mental health and substance abuse disorders, behavioral health treatment, prescription drugs, rehabilitative services, preventive care, and pediatric services including oral and vision care for children. In addition, adult children now can remain on their parents’ insurance plans until age 26. Also, when the Cadillac tax on expensive health insurance coverage was delayed until 2020, the excise payments were classified as tax deductible to the employer. Whew, did I leave anything out?
Advocates for the poor and those previously considered uninsurable were delighted by the ACA. The middle class was mollified with false reassurances that their premiums would go down, and that they could keep their current insurance plans and doctors.
Currently under the ACA, “poor” is generally defined as a reported income of 150% or less of the federal poverty level ($22,000-$45,000). The poor are eligible for health care that is very nearly free, according to Medicaid.gov. They are also protected from claims against their property. They effectively have first-dollar coverage, though they usually can’t see a specialist unless they go through the emergency department.
At age 65, Medicare takes over. Medicare is an insurance benefit workers contribute to for all their working lives. It doesn’t cover everything, but the relatively low $183 Part B deductible for office-based care sure takes the edge off. The average retirees collect three times what they put in, but they arguably could have made more if they had invested anywhere other than in the federal government.
So, the real crunch hits the middle class under age 65. Under the ACA, those earning up to $97,000 for a family of four get insurance subsidies to buy insurance on the exchanges. The real problem arises when a high deductible – the maximum for 2017 is between $13,000 and $14,000 – is combined with that level of income. There are over 20 million health care savings accounts in the United States, which ameliorate the cost for some, but health care savings accounts do not cover the entire deductible, and many cannot afford to self-fund them if their employer does not fund it for them.People simply do not have that kind of money put away in a disaster fund, and a major medical expense can easily wipe them out. If possible, they will ignore a medical problem; and if they can’t, they could risk financial toxicity by running up high-interest credit cards – with the predictable spiral of debt, collection agencies, credit ratings ruined, wages garnished, and even their homes repossessed. They aren’t going to use their health care coverage unless forced to by a disaster – or worse for the system, they have been saving their elective surgeries up and are ready for a major tune-up. The long list of mandated benefits has triggered a meltdown in the exchange markets and huge increases in premiums and deductibles for all insurance buyers. Who is going to pay for it? To quote the late Russell B. Long, former U.S. Senator:
Don’t tax you,
Don’t tax me,
Tax that fellow behind the tree.
Raising taxes and pouring even more money into the system might be a solution, but this is not likely to happen.
In the United States, the rationing of health care has been shifted from the poor to the middle class, and the middle class doesn’t like it. The situation will only change when the howling from the middle class is louder than the wailing from the formerly uninsurable and poor. As insurance premiums and deductibles continue to rocket up, I expect the howling will get louder.
So, either we agree to let insurance premiums continue to increase, our state budgets to explode, our taxes to go up, or we pare down the list of mandated benefits.
I am going on the record stating that we simply cannot afford “medical everything” for everybody. I think we should tighten up the benefit list considerably. What do you think?
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
Health care rationing is an uncomfortable topic, but one that responsible Americans should discuss. In particular, physicians should consider this topic, because our typical response to health care is that “everything should be covered.”
Health care has always been rationed. Historically, in the United States, health care has been apportioned capitalistically; that is, those who could afford health care got it and the rest – particularly the poor – scrambled. In Europe and Canada, universal or government health care is rationed by long wait times, limited range of services, and formulary restrictions. It is important to note that in almost all countries with universal health care, there is still the option of quick access and more comprehensive service via a neighboring country (to the United States from Canada, for example) or a separate additional private insurance option (England, Germany, and most of Europe).
In the United States, the Affordable Care Act (ACA) changed the dynamic regarding insurance by adding mandates to cover preexisting conditions, mental health and substance abuse disorders, behavioral health treatment, prescription drugs, rehabilitative services, preventive care, and pediatric services including oral and vision care for children. In addition, adult children now can remain on their parents’ insurance plans until age 26. Also, when the Cadillac tax on expensive health insurance coverage was delayed until 2020, the excise payments were classified as tax deductible to the employer. Whew, did I leave anything out?
Advocates for the poor and those previously considered uninsurable were delighted by the ACA. The middle class was mollified with false reassurances that their premiums would go down, and that they could keep their current insurance plans and doctors.
Currently under the ACA, “poor” is generally defined as a reported income of 150% or less of the federal poverty level ($22,000-$45,000). The poor are eligible for health care that is very nearly free, according to Medicaid.gov. They are also protected from claims against their property. They effectively have first-dollar coverage, though they usually can’t see a specialist unless they go through the emergency department.
At age 65, Medicare takes over. Medicare is an insurance benefit workers contribute to for all their working lives. It doesn’t cover everything, but the relatively low $183 Part B deductible for office-based care sure takes the edge off. The average retirees collect three times what they put in, but they arguably could have made more if they had invested anywhere other than in the federal government.
So, the real crunch hits the middle class under age 65. Under the ACA, those earning up to $97,000 for a family of four get insurance subsidies to buy insurance on the exchanges. The real problem arises when a high deductible – the maximum for 2017 is between $13,000 and $14,000 – is combined with that level of income. There are over 20 million health care savings accounts in the United States, which ameliorate the cost for some, but health care savings accounts do not cover the entire deductible, and many cannot afford to self-fund them if their employer does not fund it for them.People simply do not have that kind of money put away in a disaster fund, and a major medical expense can easily wipe them out. If possible, they will ignore a medical problem; and if they can’t, they could risk financial toxicity by running up high-interest credit cards – with the predictable spiral of debt, collection agencies, credit ratings ruined, wages garnished, and even their homes repossessed. They aren’t going to use their health care coverage unless forced to by a disaster – or worse for the system, they have been saving their elective surgeries up and are ready for a major tune-up. The long list of mandated benefits has triggered a meltdown in the exchange markets and huge increases in premiums and deductibles for all insurance buyers. Who is going to pay for it? To quote the late Russell B. Long, former U.S. Senator:
Don’t tax you,
Don’t tax me,
Tax that fellow behind the tree.
Raising taxes and pouring even more money into the system might be a solution, but this is not likely to happen.
In the United States, the rationing of health care has been shifted from the poor to the middle class, and the middle class doesn’t like it. The situation will only change when the howling from the middle class is louder than the wailing from the formerly uninsurable and poor. As insurance premiums and deductibles continue to rocket up, I expect the howling will get louder.
So, either we agree to let insurance premiums continue to increase, our state budgets to explode, our taxes to go up, or we pare down the list of mandated benefits.
I am going on the record stating that we simply cannot afford “medical everything” for everybody. I think we should tighten up the benefit list considerably. What do you think?
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at [email protected].
VIDEO: Making the most of MACRA reimbursement methods
BOSTON – “There’s a hunger out there” among physicians to participate in novel practice models made possible by value-based care, according to Lawrence R. Kosinski, MD, MBA, AGAF, clinical private practice councilor on the American Gastroenterological Association governing board.
In this video interview, conducted during the 2017 AGA Tech Summit, Dr. Kosinski explained how physicians can leverage the reimbursement methods of the recent MACRA legislation in their practices. He discusses how, in his own practice, he and his colleagues created an alternative payment model that uses a proprietary patient portal, SonarMD, to help physicians improve the management of chronic diseases such as inflammatory bowel disease at a lower cost and with markedly improved patient satisfaction.
“We look forward very much to the success of this project going forward, and the AGA will continue to be an integral part of the development of Project Sonar,” he said.
Dr. Kosinski is president of SonarMD.
BOSTON – “There’s a hunger out there” among physicians to participate in novel practice models made possible by value-based care, according to Lawrence R. Kosinski, MD, MBA, AGAF, clinical private practice councilor on the American Gastroenterological Association governing board.
In this video interview, conducted during the 2017 AGA Tech Summit, Dr. Kosinski explained how physicians can leverage the reimbursement methods of the recent MACRA legislation in their practices. He discusses how, in his own practice, he and his colleagues created an alternative payment model that uses a proprietary patient portal, SonarMD, to help physicians improve the management of chronic diseases such as inflammatory bowel disease at a lower cost and with markedly improved patient satisfaction.
“We look forward very much to the success of this project going forward, and the AGA will continue to be an integral part of the development of Project Sonar,” he said.
Dr. Kosinski is president of SonarMD.
BOSTON – “There’s a hunger out there” among physicians to participate in novel practice models made possible by value-based care, according to Lawrence R. Kosinski, MD, MBA, AGAF, clinical private practice councilor on the American Gastroenterological Association governing board.
In this video interview, conducted during the 2017 AGA Tech Summit, Dr. Kosinski explained how physicians can leverage the reimbursement methods of the recent MACRA legislation in their practices. He discusses how, in his own practice, he and his colleagues created an alternative payment model that uses a proprietary patient portal, SonarMD, to help physicians improve the management of chronic diseases such as inflammatory bowel disease at a lower cost and with markedly improved patient satisfaction.
“We look forward very much to the success of this project going forward, and the AGA will continue to be an integral part of the development of Project Sonar,” he said.
Dr. Kosinski is president of SonarMD.
FROM THE 2017 AGA TECH SUMMIT
CMS market stabilization efforts useless without subsidies
New final regulations designed to bring stability to the individual health insurance market may not matter if the White House follows through on a threat to kill subsidies paid to insurers to help reduce deductibles and other out-of-pocket costs for low-income patients.
The final rule from the Centers for Medicare & Medicaid Services grants a number of wishes sought by the insurance industry to help bring a level of predictability and flexibility when designing plans for the individual market. Specifically, it does the following:
• Shortens the open enrollment period for the 2018 plan year to 6 weeks running from Nov. 1 to Dec. 15, so that open enrollment closely aligns with Medicare and other private insurance.
• Requires individuals to submit documentation when seeking coverage through a special enrollment period.
• Allows insurers to collect past-due premiums before issuing coverage for a future year.
• Provides more actuarial flexibility to allow for different plan designs.
• Returns network adequacy oversight to states.
The new rules are not expected to alter the existing market dynamic, according to Kelly Brantley, vice president at Avalere Health.
“I would say the rule is nominally helpful, but it’s really unlikely to persuade anyone, particularly those insurers who are already on their way out. I don’t think this a game-changer for them,” she said in an interview.
The American Medical Association, in comments to the CMS when the rule was proposed as a draft, said that if finalized, the rule “would raise premiums, out-of-pocket costs, or both for millions of moderate-income families and would make it more difficult for eligible individuals to enroll in health coverage and access needed care.”
The potential impact of these regulatory changes could be moot if President Trump makes good on his threat to withhold cost-sharing subsidies to insurers. The subsides already are the subject of a lawsuit brought by the House of Representatives against the Obama administration; they continue to be paid while the suit makes its way through the judicial process. President Trump has threatened to cut off the subsidies in an effort to force Congressional Democrats to the negotiating table regarding the repeal and replacement of the Affordable Care Act.
“My take on this is that the [market stabilization] rule as written is not likely to shift the market, really, in terms of access,” Ms. Brantley said. “The bigger question is whether the cost-sharing reductions are going to be paid. I think that has a bigger likelihood of influencing issuer participation and robustness of the market in 2018.”
Even with the changes made by the market stabilization rule, “there is still too much instability and uncertainty in this market,” Marilyn Tavenner, president and CEO of the industry group America’s Health Insurance Plans, said in a statement. “Most urgently, health plans and the consumers they serve need to know that funding for cost-sharing reduction subsidies will continue uninterrupted.”
Ms. Tavenner noted that without the subsidies, more plans are likely to drop out of the health insurance exchanges, leading to premium increases, and “doctors and hospitals will see even greater strains on their ability to care for people.”
The AMA, in an April 12 letter to President Trump, cosigned by America’s Health Insurance Plans, the American Academy of Family Physicians, the American Hospital Association, the Federation of American Hospitals, the American Benefits Council, the Blue Cross Blue Shield Association, and the U.S. Chamber of Commerce, stated that the “most critical action to help stabilize the individual market for 2017 and 2018 is to remove uncertainty about continued funding for cost sharing reductions.”
Ms. Brantley added that if the subsides were cut, “it makes it more challenging to bring any kind of money back into the system at a later point. I think it would be hard for those cost-sharing reductions to go away at this point and then ever come back, but I do think that it’s a possibility that that could happen.”
The CMS released the final rule April 13, 2017, and it is scheduled for publication in the Federal Register on April 18, 2017.
New final regulations designed to bring stability to the individual health insurance market may not matter if the White House follows through on a threat to kill subsidies paid to insurers to help reduce deductibles and other out-of-pocket costs for low-income patients.
The final rule from the Centers for Medicare & Medicaid Services grants a number of wishes sought by the insurance industry to help bring a level of predictability and flexibility when designing plans for the individual market. Specifically, it does the following:
• Shortens the open enrollment period for the 2018 plan year to 6 weeks running from Nov. 1 to Dec. 15, so that open enrollment closely aligns with Medicare and other private insurance.
• Requires individuals to submit documentation when seeking coverage through a special enrollment period.
• Allows insurers to collect past-due premiums before issuing coverage for a future year.
• Provides more actuarial flexibility to allow for different plan designs.
• Returns network adequacy oversight to states.
The new rules are not expected to alter the existing market dynamic, according to Kelly Brantley, vice president at Avalere Health.
“I would say the rule is nominally helpful, but it’s really unlikely to persuade anyone, particularly those insurers who are already on their way out. I don’t think this a game-changer for them,” she said in an interview.
The American Medical Association, in comments to the CMS when the rule was proposed as a draft, said that if finalized, the rule “would raise premiums, out-of-pocket costs, or both for millions of moderate-income families and would make it more difficult for eligible individuals to enroll in health coverage and access needed care.”
The potential impact of these regulatory changes could be moot if President Trump makes good on his threat to withhold cost-sharing subsidies to insurers. The subsides already are the subject of a lawsuit brought by the House of Representatives against the Obama administration; they continue to be paid while the suit makes its way through the judicial process. President Trump has threatened to cut off the subsidies in an effort to force Congressional Democrats to the negotiating table regarding the repeal and replacement of the Affordable Care Act.
“My take on this is that the [market stabilization] rule as written is not likely to shift the market, really, in terms of access,” Ms. Brantley said. “The bigger question is whether the cost-sharing reductions are going to be paid. I think that has a bigger likelihood of influencing issuer participation and robustness of the market in 2018.”
Even with the changes made by the market stabilization rule, “there is still too much instability and uncertainty in this market,” Marilyn Tavenner, president and CEO of the industry group America’s Health Insurance Plans, said in a statement. “Most urgently, health plans and the consumers they serve need to know that funding for cost-sharing reduction subsidies will continue uninterrupted.”
Ms. Tavenner noted that without the subsidies, more plans are likely to drop out of the health insurance exchanges, leading to premium increases, and “doctors and hospitals will see even greater strains on their ability to care for people.”
The AMA, in an April 12 letter to President Trump, cosigned by America’s Health Insurance Plans, the American Academy of Family Physicians, the American Hospital Association, the Federation of American Hospitals, the American Benefits Council, the Blue Cross Blue Shield Association, and the U.S. Chamber of Commerce, stated that the “most critical action to help stabilize the individual market for 2017 and 2018 is to remove uncertainty about continued funding for cost sharing reductions.”
Ms. Brantley added that if the subsides were cut, “it makes it more challenging to bring any kind of money back into the system at a later point. I think it would be hard for those cost-sharing reductions to go away at this point and then ever come back, but I do think that it’s a possibility that that could happen.”
The CMS released the final rule April 13, 2017, and it is scheduled for publication in the Federal Register on April 18, 2017.
New final regulations designed to bring stability to the individual health insurance market may not matter if the White House follows through on a threat to kill subsidies paid to insurers to help reduce deductibles and other out-of-pocket costs for low-income patients.
The final rule from the Centers for Medicare & Medicaid Services grants a number of wishes sought by the insurance industry to help bring a level of predictability and flexibility when designing plans for the individual market. Specifically, it does the following:
• Shortens the open enrollment period for the 2018 plan year to 6 weeks running from Nov. 1 to Dec. 15, so that open enrollment closely aligns with Medicare and other private insurance.
• Requires individuals to submit documentation when seeking coverage through a special enrollment period.
• Allows insurers to collect past-due premiums before issuing coverage for a future year.
• Provides more actuarial flexibility to allow for different plan designs.
• Returns network adequacy oversight to states.
The new rules are not expected to alter the existing market dynamic, according to Kelly Brantley, vice president at Avalere Health.
“I would say the rule is nominally helpful, but it’s really unlikely to persuade anyone, particularly those insurers who are already on their way out. I don’t think this a game-changer for them,” she said in an interview.
The American Medical Association, in comments to the CMS when the rule was proposed as a draft, said that if finalized, the rule “would raise premiums, out-of-pocket costs, or both for millions of moderate-income families and would make it more difficult for eligible individuals to enroll in health coverage and access needed care.”
The potential impact of these regulatory changes could be moot if President Trump makes good on his threat to withhold cost-sharing subsidies to insurers. The subsides already are the subject of a lawsuit brought by the House of Representatives against the Obama administration; they continue to be paid while the suit makes its way through the judicial process. President Trump has threatened to cut off the subsidies in an effort to force Congressional Democrats to the negotiating table regarding the repeal and replacement of the Affordable Care Act.
“My take on this is that the [market stabilization] rule as written is not likely to shift the market, really, in terms of access,” Ms. Brantley said. “The bigger question is whether the cost-sharing reductions are going to be paid. I think that has a bigger likelihood of influencing issuer participation and robustness of the market in 2018.”
Even with the changes made by the market stabilization rule, “there is still too much instability and uncertainty in this market,” Marilyn Tavenner, president and CEO of the industry group America’s Health Insurance Plans, said in a statement. “Most urgently, health plans and the consumers they serve need to know that funding for cost-sharing reduction subsidies will continue uninterrupted.”
Ms. Tavenner noted that without the subsidies, more plans are likely to drop out of the health insurance exchanges, leading to premium increases, and “doctors and hospitals will see even greater strains on their ability to care for people.”
The AMA, in an April 12 letter to President Trump, cosigned by America’s Health Insurance Plans, the American Academy of Family Physicians, the American Hospital Association, the Federation of American Hospitals, the American Benefits Council, the Blue Cross Blue Shield Association, and the U.S. Chamber of Commerce, stated that the “most critical action to help stabilize the individual market for 2017 and 2018 is to remove uncertainty about continued funding for cost sharing reductions.”
Ms. Brantley added that if the subsides were cut, “it makes it more challenging to bring any kind of money back into the system at a later point. I think it would be hard for those cost-sharing reductions to go away at this point and then ever come back, but I do think that it’s a possibility that that could happen.”
The CMS released the final rule April 13, 2017, and it is scheduled for publication in the Federal Register on April 18, 2017.
Palbociclib/fulvestrant works in Asians with HR+/HER2– breast cancer too
Among Asian women with hormone receptor–positive/human epidermal growth factor receptor-2 (HER2)–negative metastatic breast cancer that is resistant to endocrine therapy, a combination of palbociclib (Ibrance) and fulvestrant (Faslodex) was associated with a significant improvement in progression-free survival (PFS), reported investigators from the PALOMA-3 trial.
A preplanned subgroup analysis of 102 pre- and postmenopausal women from Japan, South Korea, and Taiwan who were enrolled in the trial showed that median PFS for 71 women assigned to palbociclib and fulvestrant had not been reached at the trial end. In contrast, median PFS among 31 patients assigned to placebo and fulvestrant was 5.8 months (hazard ratio, 0.485; P = .0065), reported Hiroji Iwata, MD, PhD, of the Aichi Cancer Center Hospital in Nagoya, Japan, and his colleagues.
“Overall, palbociclib plus fulvestrant seems to be a reasonable treatment option in Asians with HR-positive/HER2-negative metastatic breast cancer that has progressed on prior endocrine therapy,” they wrote in the Journal of Global Oncology (2017 Apr 11. doi: 10.1200/JGO.2016.008318).
PALOMA-3 was a multinational randomized double-blind, placebo-controlled phase III trial comparing a combination of fulvestrant, a selective estrogen receptor downregulator, and palbociclib, a small-molecule inhibitor of cyclin-dependent kinases 4 and 6 (CDK4/6) with fulvestrant plus placebo in 521 women with HR-positive/HER2-negative advanced breast cancer.
The trial was halted early for efficacy of the combination. The final overall analysis, published in April 2016 in The Lancet Oncology, showed that the combination significantly improved PFS, compared with fulvestrant and placebo, regardless of the patients’ degree of endocrine resistance, level of hormone receptor expression level, or PIK3CA mutational status.
Dr. Iwata and his associates noted that breast cancer is generally diagnosed at a younger age among Asian women (median, 45-50 years vs. 55-60 years among Western women) and that differences in genetic backgrounds may influence drug metabolism, resulting in potential differences in efficacy and adverse event profiles.
Among patients treated with palbociclib, the most common grade 3 or higher adverse events were neutropenia in 92%, compared with none in the placebo arm, and leukopenia in 29% vs. 0%. In contrast, grade 3/4 neutropenia occurred in 58% of non-Asian patients treated with the combination and 0.7% of non-Asian controls. Leukopenia rates were similar between Asian and non-Asian populations, however.
Comparisons of mean trough concentrations across subgroups showed that palbociclib exposures among Asians and non-Asians were similar. Patient reported outcomes were also similar between the groups, except for significantly more dyspnea among patients who received palbociclib (P = .05).
“This study adds to the limited body of literature assessing a CDK4/6 inhibitor in Asians and represents the largest patient experience with palbociclib in Asians. The present findings show that palbociclib plus fulvestrant improved PFS in Asians with HR-positive/HER2-negative [metastatic breast cancer] who experienced progression on prior endocrine therapy and that the safety profile of palbociclib plus fulvestrant in Asians was generally consistent with that observed in non-Asians. Together, these findings suggest that palbociclib is beneficial in patients who have not previously received endocrine therapy and in Asians and non-Asians who experienced relapse or progression during prior endocrine therapy,” they wrote.
The PALOMA-3 trial was supported by Pfizer. Dr. Iwata disclosed consultations with Chugai Pharma, Eisai, and AstraZeneca. Several coauthors are Pfizer employees and shareholders.
Taking into account all caveats inherent to analyses of subpopulations of large clinical trials (for example, invariably small sample size and multiplicity of testing), the data presented by Iwata et al. support the clinically meaningful efficacy of palbociclib for the end point of progression-free survival (PFS) in Asians. However, this report and others indicate that Asians have a higher risk of adverse events (including grade 3 and 4 neutropenia) despite preserved patient-reported outcomes and quality of life. The reasons for this have yet to be elucidated. In light of growing evidence of interethnic pharmacogenomic and safety discrepancies between Asian and non-Asian populations observed in recently published clinical trials and observational studies, there is a clear need for enhanced enrollment of Asian patients and other ethnic groups into clinical trials of new agents for the treatment of metastatic breast cancer.
Ricardo L.B. Costa, MD, and William J. Gradishar, MD, are with the Feinberg School of Medicine and Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago. These comments are excerpted from an editorial accompanying the report by Dr. Iwata and his coauthors (J Glob Oncol. 2017 Apr 11. doi: 10.1200/JGO.2017.009936).
Taking into account all caveats inherent to analyses of subpopulations of large clinical trials (for example, invariably small sample size and multiplicity of testing), the data presented by Iwata et al. support the clinically meaningful efficacy of palbociclib for the end point of progression-free survival (PFS) in Asians. However, this report and others indicate that Asians have a higher risk of adverse events (including grade 3 and 4 neutropenia) despite preserved patient-reported outcomes and quality of life. The reasons for this have yet to be elucidated. In light of growing evidence of interethnic pharmacogenomic and safety discrepancies between Asian and non-Asian populations observed in recently published clinical trials and observational studies, there is a clear need for enhanced enrollment of Asian patients and other ethnic groups into clinical trials of new agents for the treatment of metastatic breast cancer.
Ricardo L.B. Costa, MD, and William J. Gradishar, MD, are with the Feinberg School of Medicine and Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago. These comments are excerpted from an editorial accompanying the report by Dr. Iwata and his coauthors (J Glob Oncol. 2017 Apr 11. doi: 10.1200/JGO.2017.009936).
Taking into account all caveats inherent to analyses of subpopulations of large clinical trials (for example, invariably small sample size and multiplicity of testing), the data presented by Iwata et al. support the clinically meaningful efficacy of palbociclib for the end point of progression-free survival (PFS) in Asians. However, this report and others indicate that Asians have a higher risk of adverse events (including grade 3 and 4 neutropenia) despite preserved patient-reported outcomes and quality of life. The reasons for this have yet to be elucidated. In light of growing evidence of interethnic pharmacogenomic and safety discrepancies between Asian and non-Asian populations observed in recently published clinical trials and observational studies, there is a clear need for enhanced enrollment of Asian patients and other ethnic groups into clinical trials of new agents for the treatment of metastatic breast cancer.
Ricardo L.B. Costa, MD, and William J. Gradishar, MD, are with the Feinberg School of Medicine and Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago. These comments are excerpted from an editorial accompanying the report by Dr. Iwata and his coauthors (J Glob Oncol. 2017 Apr 11. doi: 10.1200/JGO.2017.009936).
Among Asian women with hormone receptor–positive/human epidermal growth factor receptor-2 (HER2)–negative metastatic breast cancer that is resistant to endocrine therapy, a combination of palbociclib (Ibrance) and fulvestrant (Faslodex) was associated with a significant improvement in progression-free survival (PFS), reported investigators from the PALOMA-3 trial.
A preplanned subgroup analysis of 102 pre- and postmenopausal women from Japan, South Korea, and Taiwan who were enrolled in the trial showed that median PFS for 71 women assigned to palbociclib and fulvestrant had not been reached at the trial end. In contrast, median PFS among 31 patients assigned to placebo and fulvestrant was 5.8 months (hazard ratio, 0.485; P = .0065), reported Hiroji Iwata, MD, PhD, of the Aichi Cancer Center Hospital in Nagoya, Japan, and his colleagues.
“Overall, palbociclib plus fulvestrant seems to be a reasonable treatment option in Asians with HR-positive/HER2-negative metastatic breast cancer that has progressed on prior endocrine therapy,” they wrote in the Journal of Global Oncology (2017 Apr 11. doi: 10.1200/JGO.2016.008318).
PALOMA-3 was a multinational randomized double-blind, placebo-controlled phase III trial comparing a combination of fulvestrant, a selective estrogen receptor downregulator, and palbociclib, a small-molecule inhibitor of cyclin-dependent kinases 4 and 6 (CDK4/6) with fulvestrant plus placebo in 521 women with HR-positive/HER2-negative advanced breast cancer.
The trial was halted early for efficacy of the combination. The final overall analysis, published in April 2016 in The Lancet Oncology, showed that the combination significantly improved PFS, compared with fulvestrant and placebo, regardless of the patients’ degree of endocrine resistance, level of hormone receptor expression level, or PIK3CA mutational status.
Dr. Iwata and his associates noted that breast cancer is generally diagnosed at a younger age among Asian women (median, 45-50 years vs. 55-60 years among Western women) and that differences in genetic backgrounds may influence drug metabolism, resulting in potential differences in efficacy and adverse event profiles.
Among patients treated with palbociclib, the most common grade 3 or higher adverse events were neutropenia in 92%, compared with none in the placebo arm, and leukopenia in 29% vs. 0%. In contrast, grade 3/4 neutropenia occurred in 58% of non-Asian patients treated with the combination and 0.7% of non-Asian controls. Leukopenia rates were similar between Asian and non-Asian populations, however.
Comparisons of mean trough concentrations across subgroups showed that palbociclib exposures among Asians and non-Asians were similar. Patient reported outcomes were also similar between the groups, except for significantly more dyspnea among patients who received palbociclib (P = .05).
“This study adds to the limited body of literature assessing a CDK4/6 inhibitor in Asians and represents the largest patient experience with palbociclib in Asians. The present findings show that palbociclib plus fulvestrant improved PFS in Asians with HR-positive/HER2-negative [metastatic breast cancer] who experienced progression on prior endocrine therapy and that the safety profile of palbociclib plus fulvestrant in Asians was generally consistent with that observed in non-Asians. Together, these findings suggest that palbociclib is beneficial in patients who have not previously received endocrine therapy and in Asians and non-Asians who experienced relapse or progression during prior endocrine therapy,” they wrote.
The PALOMA-3 trial was supported by Pfizer. Dr. Iwata disclosed consultations with Chugai Pharma, Eisai, and AstraZeneca. Several coauthors are Pfizer employees and shareholders.
Among Asian women with hormone receptor–positive/human epidermal growth factor receptor-2 (HER2)–negative metastatic breast cancer that is resistant to endocrine therapy, a combination of palbociclib (Ibrance) and fulvestrant (Faslodex) was associated with a significant improvement in progression-free survival (PFS), reported investigators from the PALOMA-3 trial.
A preplanned subgroup analysis of 102 pre- and postmenopausal women from Japan, South Korea, and Taiwan who were enrolled in the trial showed that median PFS for 71 women assigned to palbociclib and fulvestrant had not been reached at the trial end. In contrast, median PFS among 31 patients assigned to placebo and fulvestrant was 5.8 months (hazard ratio, 0.485; P = .0065), reported Hiroji Iwata, MD, PhD, of the Aichi Cancer Center Hospital in Nagoya, Japan, and his colleagues.
“Overall, palbociclib plus fulvestrant seems to be a reasonable treatment option in Asians with HR-positive/HER2-negative metastatic breast cancer that has progressed on prior endocrine therapy,” they wrote in the Journal of Global Oncology (2017 Apr 11. doi: 10.1200/JGO.2016.008318).
PALOMA-3 was a multinational randomized double-blind, placebo-controlled phase III trial comparing a combination of fulvestrant, a selective estrogen receptor downregulator, and palbociclib, a small-molecule inhibitor of cyclin-dependent kinases 4 and 6 (CDK4/6) with fulvestrant plus placebo in 521 women with HR-positive/HER2-negative advanced breast cancer.
The trial was halted early for efficacy of the combination. The final overall analysis, published in April 2016 in The Lancet Oncology, showed that the combination significantly improved PFS, compared with fulvestrant and placebo, regardless of the patients’ degree of endocrine resistance, level of hormone receptor expression level, or PIK3CA mutational status.
Dr. Iwata and his associates noted that breast cancer is generally diagnosed at a younger age among Asian women (median, 45-50 years vs. 55-60 years among Western women) and that differences in genetic backgrounds may influence drug metabolism, resulting in potential differences in efficacy and adverse event profiles.
Among patients treated with palbociclib, the most common grade 3 or higher adverse events were neutropenia in 92%, compared with none in the placebo arm, and leukopenia in 29% vs. 0%. In contrast, grade 3/4 neutropenia occurred in 58% of non-Asian patients treated with the combination and 0.7% of non-Asian controls. Leukopenia rates were similar between Asian and non-Asian populations, however.
Comparisons of mean trough concentrations across subgroups showed that palbociclib exposures among Asians and non-Asians were similar. Patient reported outcomes were also similar between the groups, except for significantly more dyspnea among patients who received palbociclib (P = .05).
“This study adds to the limited body of literature assessing a CDK4/6 inhibitor in Asians and represents the largest patient experience with palbociclib in Asians. The present findings show that palbociclib plus fulvestrant improved PFS in Asians with HR-positive/HER2-negative [metastatic breast cancer] who experienced progression on prior endocrine therapy and that the safety profile of palbociclib plus fulvestrant in Asians was generally consistent with that observed in non-Asians. Together, these findings suggest that palbociclib is beneficial in patients who have not previously received endocrine therapy and in Asians and non-Asians who experienced relapse or progression during prior endocrine therapy,” they wrote.
The PALOMA-3 trial was supported by Pfizer. Dr. Iwata disclosed consultations with Chugai Pharma, Eisai, and AstraZeneca. Several coauthors are Pfizer employees and shareholders.
FROM THE JOURNAL OF GLOBAL ONCOLOGY
Key clinical point: Asian patients with HR+/HER2– metastatic breast cancer derive the same benefits from palbociclib and fulvestrant as non-Asian patients.
Major finding: Median PFS was 5.8 months for patients treated with fulvestrant plus placebo, vs. not reached for patients treated with fulvestrant plus palbociclib.
Data source: Subanalysis of data on 102 Asian patients in the PALOMA-3 randomized, double-blind, placebo-controlled trial.
Disclosures: The PALOMA-3 trial was supported by Pfizer. Dr. Iwata disclosed consultations with Chugai Pharma, Eisai, and AstraZeneca. Several coauthors are Pfizer employees and shareholders.