User login
Two VAM Scholarship, Fellowship deadlines are Feb. 1
- VAM travel scholarships (Feb. 1): The scholarships (General Surgery Resident/Medical Student Travel Scholarship and Diversity Medical Student Travel Scholarship) are for medical students and residents for the Vascular Annual Meeting. Recipients will be able to meet other students and residents plus talk with members and leaders of the vascular surgical community. Applicants get complimentary meeting registration plus financial resources to help defray travel costs.
- Student Research Fellowship (Feb. 1): Sponsored by the SVS Foundation, this award seeks to introduce the student to the application of rigorous scientific methods to clinical problems and underlying biologic processes important to patients with vascular disease.
- VAM travel scholarships (Feb. 1): The scholarships (General Surgery Resident/Medical Student Travel Scholarship and Diversity Medical Student Travel Scholarship) are for medical students and residents for the Vascular Annual Meeting. Recipients will be able to meet other students and residents plus talk with members and leaders of the vascular surgical community. Applicants get complimentary meeting registration plus financial resources to help defray travel costs.
- Student Research Fellowship (Feb. 1): Sponsored by the SVS Foundation, this award seeks to introduce the student to the application of rigorous scientific methods to clinical problems and underlying biologic processes important to patients with vascular disease.
- VAM travel scholarships (Feb. 1): The scholarships (General Surgery Resident/Medical Student Travel Scholarship and Diversity Medical Student Travel Scholarship) are for medical students and residents for the Vascular Annual Meeting. Recipients will be able to meet other students and residents plus talk with members and leaders of the vascular surgical community. Applicants get complimentary meeting registration plus financial resources to help defray travel costs.
- Student Research Fellowship (Feb. 1): Sponsored by the SVS Foundation, this award seeks to introduce the student to the application of rigorous scientific methods to clinical problems and underlying biologic processes important to patients with vascular disease.
Two Important VAM Deadlines are Wednesday
The deadline is Wednesday, Jan. 17, for two important research endeavors related to the 2018 Vascular Annual Meeting: abstract submissions and the Resident Research Award. This year's VAM will be June 20 to 23 at the Hynes Convention Center in Boston. Following a full day of programming on Wednesday, June 20, plenary sessions are set for June 21-23. Exhibits will be June 21-22. VAM registration and housing are scheduled to open in early March 2018.
Abstract Submission: Abstracts must be submitted by 3 p.m. Wednesday (CST), Jan. 17.
Resident Research Award (Jan. 17): The recipient will showcase his or her work at the 2018 Vascular Annual Meeting. This award is an excellent opportunity for surgical trainees in vascular research laboratories to be recognized and rewarded for their research efforts. The winner receives a $5,000 award and the VAM presentation opportunity.
The deadline is Wednesday, Jan. 17, for two important research endeavors related to the 2018 Vascular Annual Meeting: abstract submissions and the Resident Research Award. This year's VAM will be June 20 to 23 at the Hynes Convention Center in Boston. Following a full day of programming on Wednesday, June 20, plenary sessions are set for June 21-23. Exhibits will be June 21-22. VAM registration and housing are scheduled to open in early March 2018.
Abstract Submission: Abstracts must be submitted by 3 p.m. Wednesday (CST), Jan. 17.
Resident Research Award (Jan. 17): The recipient will showcase his or her work at the 2018 Vascular Annual Meeting. This award is an excellent opportunity for surgical trainees in vascular research laboratories to be recognized and rewarded for their research efforts. The winner receives a $5,000 award and the VAM presentation opportunity.
The deadline is Wednesday, Jan. 17, for two important research endeavors related to the 2018 Vascular Annual Meeting: abstract submissions and the Resident Research Award. This year's VAM will be June 20 to 23 at the Hynes Convention Center in Boston. Following a full day of programming on Wednesday, June 20, plenary sessions are set for June 21-23. Exhibits will be June 21-22. VAM registration and housing are scheduled to open in early March 2018.
Abstract Submission: Abstracts must be submitted by 3 p.m. Wednesday (CST), Jan. 17.
Resident Research Award (Jan. 17): The recipient will showcase his or her work at the 2018 Vascular Annual Meeting. This award is an excellent opportunity for surgical trainees in vascular research laboratories to be recognized and rewarded for their research efforts. The winner receives a $5,000 award and the VAM presentation opportunity.
Trump administration clears way to require work for some Medicaid enrollees
The Trump administration early on Jan. 11 initiated a pivotal change in the Medicaid program, announcing that
The announcement came in a 10-page memo with detailed directions about how states can reshape the federal-state health program for low-income people.
The document says who should be excluded from the new work requirements – including children and people being treated for opioid abuse – and offers suggestions as to what counts as “work.” Besides employment, it can include job training, volunteering, or caring for a close relative.
“Medicaid needs to be more flexible so that states can best address the needs of this population,” Seema Verma, administrator of the Centers for Medicare & Medicaid Services, said in a press release. “Our fundamental goal is to make a positive and lasting difference in the health and wellness of our beneficiaries.”
Adding a work requirement to Medicaid would mark one of the biggest changes to the program since its inception in 1966. It is likely to prompt a lawsuit from patient advocacy groups, which claim the requirement is inconsistent with Medicaid’s objectives and would require an act of Congress.
Republicans have been pushing for the change since the Affordable Care Act added millions of so called “able-bodied” adults to Medicaid. It allowed states to provide coverage to anyone earning up to 138% of the federal poverty level (about $16,600 for an individual).
The Obama administration turned down several state requests to add a work requirement.
Ten states have applied for a federal waiver to add a work requirement – Arizona, Arkansas, Indiana, Kansas, Kentucky, Maine, New Hampshire, North Carolina, Utah and Wisconsin. Officials in several other states have said they are interested in the idea.
An HHS official, who spoke on the condition of anonymity because the official had not been authorized to discuss the developments, said the agency may approve Kentucky’s request as early asJan. 12. Gov. Matt Bevin, a Republican, first sought to add such a provision in 2016. The current request would require able-bodied adults without dependents to work at least 20 hours a week.
Kentucky, which has some of the poorest counties in the country, has seen its Medicaid enrollment double in the past 3 years after the state expanded eligibility under the ACA.
While more than 74 million people are enrolled in Medicaid, only a small fraction would be affected by the work requirement. That’s because children – who make up nearly half of Medicaid enrollees – are excluded. So are the more than 10 million people on Medicaid because they have a disability.
More than 4 in 10 adults with Medicaid coverage already work full time, and most others either go to school, take care of a relative, or are too sick to work.
Still, critics fear a work requirement could have a chilling effect on people signing up for Medicaid or make it harder for people to get coverage.
But work requirements have strong public backing. About 70% of Americans say they support states imposing a work requirement on non-disabled adults, according to a Kaiser Family Foundation poll last year.
The Trump administration, along with many Republican leaders in Congress, has long supported such a move. The failed efforts in the House to replace Obamacare included a work requirement for Medicaid.
In its guidance to states, CMS said they should consider how some communities have high unemployment rates and whether enrollees need to care for young children and elderly families.
CMS also advised states to make work requirements for Medicaid similar than those used with food stamps to “reduce the burden on both states and beneficiaries.”
“This new guidance paves the way for states to demonstrate how their ideas will improve the health of Medicaid beneficiaries, as well as potentially improve their economic well-being,” Brian Neale, CMS deputy administrator and director for the Center for Medicaid and CHIP Services, said in the press release.
Ms. Verma, who has said she doesn’t think Medicaid should become a way of life for people who are not disabled, said the new guidance shows how the administration is trying to give states more flexibility in running Medicaid.
“Our policy guidance was in response to states that asked us for the flexibility they need to improve their programs and to help people in achieving greater well-being and self-sufficiency,” she said.
Ms. Verma, who worked with Kentucky and Indiana on their work requirement waivers as a health consultant before joining the Trump administration, recused herself from the decision on those states’ waiver requests.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
The Trump administration early on Jan. 11 initiated a pivotal change in the Medicaid program, announcing that
The announcement came in a 10-page memo with detailed directions about how states can reshape the federal-state health program for low-income people.
The document says who should be excluded from the new work requirements – including children and people being treated for opioid abuse – and offers suggestions as to what counts as “work.” Besides employment, it can include job training, volunteering, or caring for a close relative.
“Medicaid needs to be more flexible so that states can best address the needs of this population,” Seema Verma, administrator of the Centers for Medicare & Medicaid Services, said in a press release. “Our fundamental goal is to make a positive and lasting difference in the health and wellness of our beneficiaries.”
Adding a work requirement to Medicaid would mark one of the biggest changes to the program since its inception in 1966. It is likely to prompt a lawsuit from patient advocacy groups, which claim the requirement is inconsistent with Medicaid’s objectives and would require an act of Congress.
Republicans have been pushing for the change since the Affordable Care Act added millions of so called “able-bodied” adults to Medicaid. It allowed states to provide coverage to anyone earning up to 138% of the federal poverty level (about $16,600 for an individual).
The Obama administration turned down several state requests to add a work requirement.
Ten states have applied for a federal waiver to add a work requirement – Arizona, Arkansas, Indiana, Kansas, Kentucky, Maine, New Hampshire, North Carolina, Utah and Wisconsin. Officials in several other states have said they are interested in the idea.
An HHS official, who spoke on the condition of anonymity because the official had not been authorized to discuss the developments, said the agency may approve Kentucky’s request as early asJan. 12. Gov. Matt Bevin, a Republican, first sought to add such a provision in 2016. The current request would require able-bodied adults without dependents to work at least 20 hours a week.
Kentucky, which has some of the poorest counties in the country, has seen its Medicaid enrollment double in the past 3 years after the state expanded eligibility under the ACA.
While more than 74 million people are enrolled in Medicaid, only a small fraction would be affected by the work requirement. That’s because children – who make up nearly half of Medicaid enrollees – are excluded. So are the more than 10 million people on Medicaid because they have a disability.
More than 4 in 10 adults with Medicaid coverage already work full time, and most others either go to school, take care of a relative, or are too sick to work.
Still, critics fear a work requirement could have a chilling effect on people signing up for Medicaid or make it harder for people to get coverage.
But work requirements have strong public backing. About 70% of Americans say they support states imposing a work requirement on non-disabled adults, according to a Kaiser Family Foundation poll last year.
The Trump administration, along with many Republican leaders in Congress, has long supported such a move. The failed efforts in the House to replace Obamacare included a work requirement for Medicaid.
In its guidance to states, CMS said they should consider how some communities have high unemployment rates and whether enrollees need to care for young children and elderly families.
CMS also advised states to make work requirements for Medicaid similar than those used with food stamps to “reduce the burden on both states and beneficiaries.”
“This new guidance paves the way for states to demonstrate how their ideas will improve the health of Medicaid beneficiaries, as well as potentially improve their economic well-being,” Brian Neale, CMS deputy administrator and director for the Center for Medicaid and CHIP Services, said in the press release.
Ms. Verma, who has said she doesn’t think Medicaid should become a way of life for people who are not disabled, said the new guidance shows how the administration is trying to give states more flexibility in running Medicaid.
“Our policy guidance was in response to states that asked us for the flexibility they need to improve their programs and to help people in achieving greater well-being and self-sufficiency,” she said.
Ms. Verma, who worked with Kentucky and Indiana on their work requirement waivers as a health consultant before joining the Trump administration, recused herself from the decision on those states’ waiver requests.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
The Trump administration early on Jan. 11 initiated a pivotal change in the Medicaid program, announcing that
The announcement came in a 10-page memo with detailed directions about how states can reshape the federal-state health program for low-income people.
The document says who should be excluded from the new work requirements – including children and people being treated for opioid abuse – and offers suggestions as to what counts as “work.” Besides employment, it can include job training, volunteering, or caring for a close relative.
“Medicaid needs to be more flexible so that states can best address the needs of this population,” Seema Verma, administrator of the Centers for Medicare & Medicaid Services, said in a press release. “Our fundamental goal is to make a positive and lasting difference in the health and wellness of our beneficiaries.”
Adding a work requirement to Medicaid would mark one of the biggest changes to the program since its inception in 1966. It is likely to prompt a lawsuit from patient advocacy groups, which claim the requirement is inconsistent with Medicaid’s objectives and would require an act of Congress.
Republicans have been pushing for the change since the Affordable Care Act added millions of so called “able-bodied” adults to Medicaid. It allowed states to provide coverage to anyone earning up to 138% of the federal poverty level (about $16,600 for an individual).
The Obama administration turned down several state requests to add a work requirement.
Ten states have applied for a federal waiver to add a work requirement – Arizona, Arkansas, Indiana, Kansas, Kentucky, Maine, New Hampshire, North Carolina, Utah and Wisconsin. Officials in several other states have said they are interested in the idea.
An HHS official, who spoke on the condition of anonymity because the official had not been authorized to discuss the developments, said the agency may approve Kentucky’s request as early asJan. 12. Gov. Matt Bevin, a Republican, first sought to add such a provision in 2016. The current request would require able-bodied adults without dependents to work at least 20 hours a week.
Kentucky, which has some of the poorest counties in the country, has seen its Medicaid enrollment double in the past 3 years after the state expanded eligibility under the ACA.
While more than 74 million people are enrolled in Medicaid, only a small fraction would be affected by the work requirement. That’s because children – who make up nearly half of Medicaid enrollees – are excluded. So are the more than 10 million people on Medicaid because they have a disability.
More than 4 in 10 adults with Medicaid coverage already work full time, and most others either go to school, take care of a relative, or are too sick to work.
Still, critics fear a work requirement could have a chilling effect on people signing up for Medicaid or make it harder for people to get coverage.
But work requirements have strong public backing. About 70% of Americans say they support states imposing a work requirement on non-disabled adults, according to a Kaiser Family Foundation poll last year.
The Trump administration, along with many Republican leaders in Congress, has long supported such a move. The failed efforts in the House to replace Obamacare included a work requirement for Medicaid.
In its guidance to states, CMS said they should consider how some communities have high unemployment rates and whether enrollees need to care for young children and elderly families.
CMS also advised states to make work requirements for Medicaid similar than those used with food stamps to “reduce the burden on both states and beneficiaries.”
“This new guidance paves the way for states to demonstrate how their ideas will improve the health of Medicaid beneficiaries, as well as potentially improve their economic well-being,” Brian Neale, CMS deputy administrator and director for the Center for Medicaid and CHIP Services, said in the press release.
Ms. Verma, who has said she doesn’t think Medicaid should become a way of life for people who are not disabled, said the new guidance shows how the administration is trying to give states more flexibility in running Medicaid.
“Our policy guidance was in response to states that asked us for the flexibility they need to improve their programs and to help people in achieving greater well-being and self-sufficiency,” she said.
Ms. Verma, who worked with Kentucky and Indiana on their work requirement waivers as a health consultant before joining the Trump administration, recused herself from the decision on those states’ waiver requests.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Does maternal sleep position affect risk of stillbirth?
WHAT DOES THIS MEAN FOR PRACTICE?
Encourage pregnant patients to not go to sleep in the supine position, especially those who:
- are obese
- have medical complications of pregnancy
- have a history of prior stillbirth
- smoke
- are of advanced maternal age
Normal prepregnancy weight becoming less normal
according to the Centers for Disease Control and Prevention.
The overall prevalence of normal prepregnancy weight declined from 47.3% to 45.1% over that period in 36 states, the District of Columbia, and New York City, which reports natality data separately from New York state. The decreases were statistically significant in 26 states and New York City, the CDC investigators reported (MMWR. 2018 Jan 5;66[51-2]:1402-7).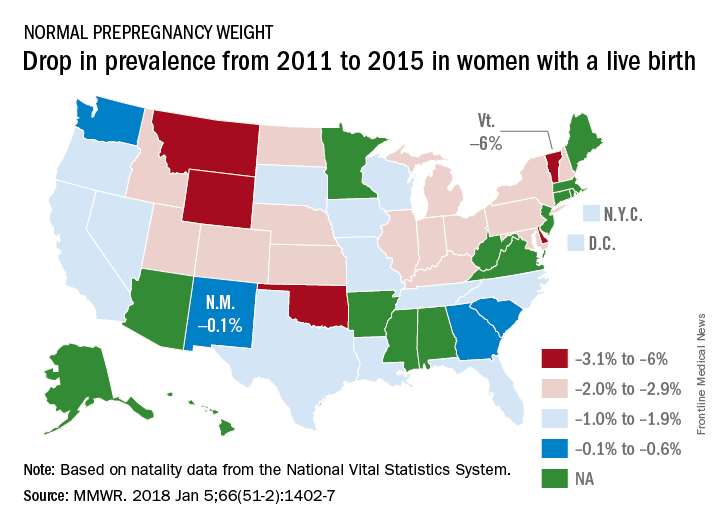
Based on data from 48 states, D.C., and New York City, the distribution of prevalence for the BMI categories in 2015 was 3.6% underweight, 45% normal weight, 25.8% overweight, and 25.6% obese, the investigators said.
The CDC analysis was based on natality data from the National Vital Statistics System. The standard birth certificate was revised in 2003 to include maternal height and prepregnancy weight, but only 38 jurisdictions were using it by 2011. By 2015, all states except Connecticut and New Jersey had adopted its use.
according to the Centers for Disease Control and Prevention.
The overall prevalence of normal prepregnancy weight declined from 47.3% to 45.1% over that period in 36 states, the District of Columbia, and New York City, which reports natality data separately from New York state. The decreases were statistically significant in 26 states and New York City, the CDC investigators reported (MMWR. 2018 Jan 5;66[51-2]:1402-7).
Based on data from 48 states, D.C., and New York City, the distribution of prevalence for the BMI categories in 2015 was 3.6% underweight, 45% normal weight, 25.8% overweight, and 25.6% obese, the investigators said.
The CDC analysis was based on natality data from the National Vital Statistics System. The standard birth certificate was revised in 2003 to include maternal height and prepregnancy weight, but only 38 jurisdictions were using it by 2011. By 2015, all states except Connecticut and New Jersey had adopted its use.
according to the Centers for Disease Control and Prevention.
The overall prevalence of normal prepregnancy weight declined from 47.3% to 45.1% over that period in 36 states, the District of Columbia, and New York City, which reports natality data separately from New York state. The decreases were statistically significant in 26 states and New York City, the CDC investigators reported (MMWR. 2018 Jan 5;66[51-2]:1402-7).
Based on data from 48 states, D.C., and New York City, the distribution of prevalence for the BMI categories in 2015 was 3.6% underweight, 45% normal weight, 25.8% overweight, and 25.6% obese, the investigators said.
The CDC analysis was based on natality data from the National Vital Statistics System. The standard birth certificate was revised in 2003 to include maternal height and prepregnancy weight, but only 38 jurisdictions were using it by 2011. By 2015, all states except Connecticut and New Jersey had adopted its use.
FROM MMWR
Role of Obstructive Sleep Apnea in HTN
Heart disease and stroke are leading causes of death and disability. High blood pressure (BP) is a major risk factor for both.
The 2017 guidelines regarding “Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure” (JNC 7) were recently published, which is an update incorporating new information from studies regarding BP-related risk of cardiovascular disease (CVD) and strategies to improve hypertension (HTN) treatment and control.
Screening for secondary causes of HTN is necessary for new-onset or uncontrolled HTN in adults, including drug-resistant HTN. Screening includes testing for obstructive sleep apnea, which is highly prevalent in this population.
Obstructive sleep apnea is a common chronic condition characterized by recurrent collapse of upper airways during sleep, inducing intermittent episodes of apnea/hypopnea, hypoxemia, and sleep disruption (Pedrosa RP, et al. Chest. 2013;144[5]:1487).
It is estimated to affect 17% of US adults but is overwhelmingly underrecognized and untreated (JAMA. 2012;307[20]:2169). The prevalence is higher in men than women. The major risk factors for OSA are obesity, male sex, and advancing age. Since these conditions oftentimes predispose to and are concomitant with HTN, it can be challenging to determine the independent effects of OSA on the development of HTN.
The relationship between obstructive sleep apnea (OSA) and HTN has been a point of interest for decades, with untreated OSA being associated with an increased risk for developing new-onset HTN (JAMA. 2012;307[20]:2169).
There have been several landmark trials that have sought to determine the extent of a causal relationship between OSAS and HTN. Sleep Heart Health Study (Sleep. 2006;29;1009) was one such study, which was limited by the inability to prove that OSA preceded the onset of HTN.
Wisconsin Sleep Cohort (N Engl J Med. 2000;342:1378) was another landmark prospective longitudinal study that implicates OSA as a possible causal factor in HTN. The notable limitation of the study was the presence of HTN after initial assessment was found to be dependent upon the severity of OSA at baseline.
While these two cohort studies found an association between OSA and HTN, the Vitoria Sleep Cohort out of Spain (Am J Respir Crit Care Med. 2011;184[11]:1299), the third and most recent longitudinal cohort study, looked at younger and thinner patients than the SHHS and the Wisconsin Sleep Cohort, failed to show a significant association between OSA and incident HTN. Methodologic differences may help to explain the disparity in results.
NREM sleep has normal circadian variation of BP, causing “dipping” of both systolic and diastolic BP at night due to decreased sympathetic and increased parasympathetic activity. REM sleep has predominant sympathetic activity and transient nocturnal BP surges.
OSA results in hypoxemia, which causes nocturnal catecholamine surges, resulting in nocturnal increase in heart rate and BP that is most prominent during post-apneic hyperventilation.
Reduced nocturnal BP (nondipping) or even higher nocturnal BP than daytime BP is an undoubted risk factor for hypertensive patients due to the end-organ damage and subsequent cardiovascular events. With sleep apnea, sleep quality is decreased due to frequent arousal from sleep (Hypertension. 2006;47[5]:833).
Sleep duration of less than or equal to 5 hours per night was shown to significantly increase risk for HTN in patients less than or equal to 60 years of age, even after controlling for obesity and diabetes.
Sleep Heart Health Study suggests that sleep duration above or below a median of 7 to 8 hours per night is associated with a higher prevalence of HTN (Sleep. 2006;29:1009). Thus, improving duration and quality of sleep in sleep apnea patients may help decrease the risk of developing HTN.
Key question: Will treatment of OSA appreciably alter BP?
Continuous positive airway pressure (CPAP) is an efficacious treatment of choice for OSA. Interventional trials, though limited by issues related to compliance, have shown CPAP to acutely reduce sympathetic drive and BP during sleep. However, this improvement in BP control is not entirely consistent in all patients with the data being less clear-cut regarding nighttime CPAP therapy and impact on daytime BP.
A randomized controlled trial from Barbe et al suggests that normotensive subjects with severe OSA but without demonstrable daytime sleepiness are immune to the BP-reducing effects of CPAP (Ann Intern Med. 2001;134:1015); those who were objectively sleepy had a more robust response to the BP lowering effects of CPAP with better cardiovascular outcomes among patients who were adherent to CPAP therapy (≥4 hours per night).
Sleep Apnea Cardiovascular Endpoints (SAVE) study looked at CPAP for Prevention of Cardiovascular Events in Obstructive Sleep Apnea (N Engl J Med. 2016;375:919). CPAP significantly reduced snoring and daytime sleepiness and improved health-related quality of life and mood, but the risk of serious cardiovascular events was not lower among patients who received treatment with CPAP in addition to usual care compared with usual care alone. This study was not powered to provide definitive answers regarding the effects of CPAP on secondary cardiovascular end points, and the use of PAP was less than 4 hours.
A recent systematic review and meta-analysis looked at “Association of Positive Airway Pressure with Cardiovascular Events and Death in Adults with Sleep Apnea” (JAMA. 2017;318(2):156). No significant associations between PAP treatment and a range of cardiovascular events were noted in this meta-analysis.
It is possible that the limited adherence to therapy in many trials was insufficient to drive protection, along with short follow-up duration of most trials that may have given insufficient time for PAP to have affected vascular outcomes.
In a cross-over study of valsartan and CPAP, combining drug treatment with CPAP appeared to have a more synergistic effect in reducing BP than either agent alone (Am J Respir Crit Care Med. 2010;182:954).
The beneficial effect of CPAP remains an open question. Considering the multifactorial pathophysiology of OSA-associated HTN, proven therapies, such as BP lowering, lipid lowering, and antiplatelet therapy, along with PAP therapy, should be utilized. This combination strategy is likely to be more effective in improving both nocturnal and daytime BP control in OSA.
Dr. Singh is Director, Sleep Disorder and Research Center, Michael E. DeBakey VA Medical Center; and Dr. Singh is Assistant Professor and Dr. Velamuri is Associate Professor, Pulmonary, Critical Care and Sleep Medicine, Baylor College of Medicine. Houston, Texas.
Heart disease and stroke are leading causes of death and disability. High blood pressure (BP) is a major risk factor for both.
The 2017 guidelines regarding “Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure” (JNC 7) were recently published, which is an update incorporating new information from studies regarding BP-related risk of cardiovascular disease (CVD) and strategies to improve hypertension (HTN) treatment and control.
Screening for secondary causes of HTN is necessary for new-onset or uncontrolled HTN in adults, including drug-resistant HTN. Screening includes testing for obstructive sleep apnea, which is highly prevalent in this population.
Obstructive sleep apnea is a common chronic condition characterized by recurrent collapse of upper airways during sleep, inducing intermittent episodes of apnea/hypopnea, hypoxemia, and sleep disruption (Pedrosa RP, et al. Chest. 2013;144[5]:1487).
It is estimated to affect 17% of US adults but is overwhelmingly underrecognized and untreated (JAMA. 2012;307[20]:2169). The prevalence is higher in men than women. The major risk factors for OSA are obesity, male sex, and advancing age. Since these conditions oftentimes predispose to and are concomitant with HTN, it can be challenging to determine the independent effects of OSA on the development of HTN.
The relationship between obstructive sleep apnea (OSA) and HTN has been a point of interest for decades, with untreated OSA being associated with an increased risk for developing new-onset HTN (JAMA. 2012;307[20]:2169).
There have been several landmark trials that have sought to determine the extent of a causal relationship between OSAS and HTN. Sleep Heart Health Study (Sleep. 2006;29;1009) was one such study, which was limited by the inability to prove that OSA preceded the onset of HTN.
Wisconsin Sleep Cohort (N Engl J Med. 2000;342:1378) was another landmark prospective longitudinal study that implicates OSA as a possible causal factor in HTN. The notable limitation of the study was the presence of HTN after initial assessment was found to be dependent upon the severity of OSA at baseline.
While these two cohort studies found an association between OSA and HTN, the Vitoria Sleep Cohort out of Spain (Am J Respir Crit Care Med. 2011;184[11]:1299), the third and most recent longitudinal cohort study, looked at younger and thinner patients than the SHHS and the Wisconsin Sleep Cohort, failed to show a significant association between OSA and incident HTN. Methodologic differences may help to explain the disparity in results.
NREM sleep has normal circadian variation of BP, causing “dipping” of both systolic and diastolic BP at night due to decreased sympathetic and increased parasympathetic activity. REM sleep has predominant sympathetic activity and transient nocturnal BP surges.
OSA results in hypoxemia, which causes nocturnal catecholamine surges, resulting in nocturnal increase in heart rate and BP that is most prominent during post-apneic hyperventilation.
Reduced nocturnal BP (nondipping) or even higher nocturnal BP than daytime BP is an undoubted risk factor for hypertensive patients due to the end-organ damage and subsequent cardiovascular events. With sleep apnea, sleep quality is decreased due to frequent arousal from sleep (Hypertension. 2006;47[5]:833).
Sleep duration of less than or equal to 5 hours per night was shown to significantly increase risk for HTN in patients less than or equal to 60 years of age, even after controlling for obesity and diabetes.
Sleep Heart Health Study suggests that sleep duration above or below a median of 7 to 8 hours per night is associated with a higher prevalence of HTN (Sleep. 2006;29:1009). Thus, improving duration and quality of sleep in sleep apnea patients may help decrease the risk of developing HTN.
Key question: Will treatment of OSA appreciably alter BP?
Continuous positive airway pressure (CPAP) is an efficacious treatment of choice for OSA. Interventional trials, though limited by issues related to compliance, have shown CPAP to acutely reduce sympathetic drive and BP during sleep. However, this improvement in BP control is not entirely consistent in all patients with the data being less clear-cut regarding nighttime CPAP therapy and impact on daytime BP.
A randomized controlled trial from Barbe et al suggests that normotensive subjects with severe OSA but without demonstrable daytime sleepiness are immune to the BP-reducing effects of CPAP (Ann Intern Med. 2001;134:1015); those who were objectively sleepy had a more robust response to the BP lowering effects of CPAP with better cardiovascular outcomes among patients who were adherent to CPAP therapy (≥4 hours per night).
Sleep Apnea Cardiovascular Endpoints (SAVE) study looked at CPAP for Prevention of Cardiovascular Events in Obstructive Sleep Apnea (N Engl J Med. 2016;375:919). CPAP significantly reduced snoring and daytime sleepiness and improved health-related quality of life and mood, but the risk of serious cardiovascular events was not lower among patients who received treatment with CPAP in addition to usual care compared with usual care alone. This study was not powered to provide definitive answers regarding the effects of CPAP on secondary cardiovascular end points, and the use of PAP was less than 4 hours.
A recent systematic review and meta-analysis looked at “Association of Positive Airway Pressure with Cardiovascular Events and Death in Adults with Sleep Apnea” (JAMA. 2017;318(2):156). No significant associations between PAP treatment and a range of cardiovascular events were noted in this meta-analysis.
It is possible that the limited adherence to therapy in many trials was insufficient to drive protection, along with short follow-up duration of most trials that may have given insufficient time for PAP to have affected vascular outcomes.
In a cross-over study of valsartan and CPAP, combining drug treatment with CPAP appeared to have a more synergistic effect in reducing BP than either agent alone (Am J Respir Crit Care Med. 2010;182:954).
The beneficial effect of CPAP remains an open question. Considering the multifactorial pathophysiology of OSA-associated HTN, proven therapies, such as BP lowering, lipid lowering, and antiplatelet therapy, along with PAP therapy, should be utilized. This combination strategy is likely to be more effective in improving both nocturnal and daytime BP control in OSA.
Dr. Singh is Director, Sleep Disorder and Research Center, Michael E. DeBakey VA Medical Center; and Dr. Singh is Assistant Professor and Dr. Velamuri is Associate Professor, Pulmonary, Critical Care and Sleep Medicine, Baylor College of Medicine. Houston, Texas.
Heart disease and stroke are leading causes of death and disability. High blood pressure (BP) is a major risk factor for both.
The 2017 guidelines regarding “Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure” (JNC 7) were recently published, which is an update incorporating new information from studies regarding BP-related risk of cardiovascular disease (CVD) and strategies to improve hypertension (HTN) treatment and control.
Screening for secondary causes of HTN is necessary for new-onset or uncontrolled HTN in adults, including drug-resistant HTN. Screening includes testing for obstructive sleep apnea, which is highly prevalent in this population.
Obstructive sleep apnea is a common chronic condition characterized by recurrent collapse of upper airways during sleep, inducing intermittent episodes of apnea/hypopnea, hypoxemia, and sleep disruption (Pedrosa RP, et al. Chest. 2013;144[5]:1487).
It is estimated to affect 17% of US adults but is overwhelmingly underrecognized and untreated (JAMA. 2012;307[20]:2169). The prevalence is higher in men than women. The major risk factors for OSA are obesity, male sex, and advancing age. Since these conditions oftentimes predispose to and are concomitant with HTN, it can be challenging to determine the independent effects of OSA on the development of HTN.
The relationship between obstructive sleep apnea (OSA) and HTN has been a point of interest for decades, with untreated OSA being associated with an increased risk for developing new-onset HTN (JAMA. 2012;307[20]:2169).
There have been several landmark trials that have sought to determine the extent of a causal relationship between OSAS and HTN. Sleep Heart Health Study (Sleep. 2006;29;1009) was one such study, which was limited by the inability to prove that OSA preceded the onset of HTN.
Wisconsin Sleep Cohort (N Engl J Med. 2000;342:1378) was another landmark prospective longitudinal study that implicates OSA as a possible causal factor in HTN. The notable limitation of the study was the presence of HTN after initial assessment was found to be dependent upon the severity of OSA at baseline.
While these two cohort studies found an association between OSA and HTN, the Vitoria Sleep Cohort out of Spain (Am J Respir Crit Care Med. 2011;184[11]:1299), the third and most recent longitudinal cohort study, looked at younger and thinner patients than the SHHS and the Wisconsin Sleep Cohort, failed to show a significant association between OSA and incident HTN. Methodologic differences may help to explain the disparity in results.
NREM sleep has normal circadian variation of BP, causing “dipping” of both systolic and diastolic BP at night due to decreased sympathetic and increased parasympathetic activity. REM sleep has predominant sympathetic activity and transient nocturnal BP surges.
OSA results in hypoxemia, which causes nocturnal catecholamine surges, resulting in nocturnal increase in heart rate and BP that is most prominent during post-apneic hyperventilation.
Reduced nocturnal BP (nondipping) or even higher nocturnal BP than daytime BP is an undoubted risk factor for hypertensive patients due to the end-organ damage and subsequent cardiovascular events. With sleep apnea, sleep quality is decreased due to frequent arousal from sleep (Hypertension. 2006;47[5]:833).
Sleep duration of less than or equal to 5 hours per night was shown to significantly increase risk for HTN in patients less than or equal to 60 years of age, even after controlling for obesity and diabetes.
Sleep Heart Health Study suggests that sleep duration above or below a median of 7 to 8 hours per night is associated with a higher prevalence of HTN (Sleep. 2006;29:1009). Thus, improving duration and quality of sleep in sleep apnea patients may help decrease the risk of developing HTN.
Key question: Will treatment of OSA appreciably alter BP?
Continuous positive airway pressure (CPAP) is an efficacious treatment of choice for OSA. Interventional trials, though limited by issues related to compliance, have shown CPAP to acutely reduce sympathetic drive and BP during sleep. However, this improvement in BP control is not entirely consistent in all patients with the data being less clear-cut regarding nighttime CPAP therapy and impact on daytime BP.
A randomized controlled trial from Barbe et al suggests that normotensive subjects with severe OSA but without demonstrable daytime sleepiness are immune to the BP-reducing effects of CPAP (Ann Intern Med. 2001;134:1015); those who were objectively sleepy had a more robust response to the BP lowering effects of CPAP with better cardiovascular outcomes among patients who were adherent to CPAP therapy (≥4 hours per night).
Sleep Apnea Cardiovascular Endpoints (SAVE) study looked at CPAP for Prevention of Cardiovascular Events in Obstructive Sleep Apnea (N Engl J Med. 2016;375:919). CPAP significantly reduced snoring and daytime sleepiness and improved health-related quality of life and mood, but the risk of serious cardiovascular events was not lower among patients who received treatment with CPAP in addition to usual care compared with usual care alone. This study was not powered to provide definitive answers regarding the effects of CPAP on secondary cardiovascular end points, and the use of PAP was less than 4 hours.
A recent systematic review and meta-analysis looked at “Association of Positive Airway Pressure with Cardiovascular Events and Death in Adults with Sleep Apnea” (JAMA. 2017;318(2):156). No significant associations between PAP treatment and a range of cardiovascular events were noted in this meta-analysis.
It is possible that the limited adherence to therapy in many trials was insufficient to drive protection, along with short follow-up duration of most trials that may have given insufficient time for PAP to have affected vascular outcomes.
In a cross-over study of valsartan and CPAP, combining drug treatment with CPAP appeared to have a more synergistic effect in reducing BP than either agent alone (Am J Respir Crit Care Med. 2010;182:954).
The beneficial effect of CPAP remains an open question. Considering the multifactorial pathophysiology of OSA-associated HTN, proven therapies, such as BP lowering, lipid lowering, and antiplatelet therapy, along with PAP therapy, should be utilized. This combination strategy is likely to be more effective in improving both nocturnal and daytime BP control in OSA.
Dr. Singh is Director, Sleep Disorder and Research Center, Michael E. DeBakey VA Medical Center; and Dr. Singh is Assistant Professor and Dr. Velamuri is Associate Professor, Pulmonary, Critical Care and Sleep Medicine, Baylor College of Medicine. Houston, Texas.
Check orthostatic vital signs within 1 minute
Clinical question: What is the relationship between timing of measurement of postural blood pressure (BP) and adverse clinical outcomes?
Background: Guidelines recommend measuring postural BP after 3 minutes of standing to avoid potentially false-positive readings obtained before that interval. In SPRINT, orthostatic hypotension (OH) determined at 1 minute was associated with higher risk of emergency department visits for OH and syncope. Whether that finding was because of the shortened interval of measurement is uncertain.
Setting: Four U.S. communities over 2 decades.
Synopsis: In a cohort of 11,429 middle-aged patients, upright BP was measured every 25 seconds over a 5-minute interval after participants had been supine for 20 minutes. About 2-3 seconds elapsed between the end of one BP measurement and the initiation of the next. OH was defined as a 20–mm Hg drop in systolic BP. After researchers adjusted for covariates, OH at 30 seconds and 1 minute were associated with higher odds of dizziness, fracture, syncope, death, and motor vehicle crashes recorded over a median follow-up of 23 years. Measurements after 1 minute were not reliably associated with any adverse outcomes.
Bottom line: Measuring OH at 30 seconds and 1 minute reliably identifies patients at risk for associated adverse clinical outcomes.
Citation: Juraschek SP et al. Association of history of dizziness and long-term adverse outcomes with early vs. later orthostatic hypotension times in middle-aged adults. JAMA Intern Med. 2017 Sep 1;177(9):1316-23.
Dr. Anderson is an associate program director in the internal medicine residency training program at the University of Colorado School of Medicine and a hospitalist at the VA Eastern Colorado Health Care System in Denver.
Clinical question: What is the relationship between timing of measurement of postural blood pressure (BP) and adverse clinical outcomes?
Background: Guidelines recommend measuring postural BP after 3 minutes of standing to avoid potentially false-positive readings obtained before that interval. In SPRINT, orthostatic hypotension (OH) determined at 1 minute was associated with higher risk of emergency department visits for OH and syncope. Whether that finding was because of the shortened interval of measurement is uncertain.
Setting: Four U.S. communities over 2 decades.
Synopsis: In a cohort of 11,429 middle-aged patients, upright BP was measured every 25 seconds over a 5-minute interval after participants had been supine for 20 minutes. About 2-3 seconds elapsed between the end of one BP measurement and the initiation of the next. OH was defined as a 20–mm Hg drop in systolic BP. After researchers adjusted for covariates, OH at 30 seconds and 1 minute were associated with higher odds of dizziness, fracture, syncope, death, and motor vehicle crashes recorded over a median follow-up of 23 years. Measurements after 1 minute were not reliably associated with any adverse outcomes.
Bottom line: Measuring OH at 30 seconds and 1 minute reliably identifies patients at risk for associated adverse clinical outcomes.
Citation: Juraschek SP et al. Association of history of dizziness and long-term adverse outcomes with early vs. later orthostatic hypotension times in middle-aged adults. JAMA Intern Med. 2017 Sep 1;177(9):1316-23.
Dr. Anderson is an associate program director in the internal medicine residency training program at the University of Colorado School of Medicine and a hospitalist at the VA Eastern Colorado Health Care System in Denver.
Clinical question: What is the relationship between timing of measurement of postural blood pressure (BP) and adverse clinical outcomes?
Background: Guidelines recommend measuring postural BP after 3 minutes of standing to avoid potentially false-positive readings obtained before that interval. In SPRINT, orthostatic hypotension (OH) determined at 1 minute was associated with higher risk of emergency department visits for OH and syncope. Whether that finding was because of the shortened interval of measurement is uncertain.
Setting: Four U.S. communities over 2 decades.
Synopsis: In a cohort of 11,429 middle-aged patients, upright BP was measured every 25 seconds over a 5-minute interval after participants had been supine for 20 minutes. About 2-3 seconds elapsed between the end of one BP measurement and the initiation of the next. OH was defined as a 20–mm Hg drop in systolic BP. After researchers adjusted for covariates, OH at 30 seconds and 1 minute were associated with higher odds of dizziness, fracture, syncope, death, and motor vehicle crashes recorded over a median follow-up of 23 years. Measurements after 1 minute were not reliably associated with any adverse outcomes.
Bottom line: Measuring OH at 30 seconds and 1 minute reliably identifies patients at risk for associated adverse clinical outcomes.
Citation: Juraschek SP et al. Association of history of dizziness and long-term adverse outcomes with early vs. later orthostatic hypotension times in middle-aged adults. JAMA Intern Med. 2017 Sep 1;177(9):1316-23.
Dr. Anderson is an associate program director in the internal medicine residency training program at the University of Colorado School of Medicine and a hospitalist at the VA Eastern Colorado Health Care System in Denver.
Smart Ways to Give More Now
Your gift today truly has an immediate impact that makes a difference now.
We also want you to benefit as much as possible from your generosity.
Gifts of Appreciated Securities, Mutual Funds, and Investments
If you have owned any of these longer than 1 year and they have appreciated in value, they provide a smart option for gifting. You will avoid the capital gains tax, and you also receive a charitable income tax deduction if you itemize your tax return.
The Charitable Individual Retirement Plan Option
If you are 70 1/2, you may distribute funds from your IRA directly to the CHEST Foundation.
You will not pay any income taxes, and it will also qualify for your required minimum withdrawal. You may distribute up to $100,000 per person per year ($200,000 if you are married and both own an IRA).
Retirement Plan Beneficiary Designation
You may also designate a charity as a beneficiary of your IRA, 401K, or 403B.
This will avoid any income tax, so 100% will be directed to the charity of your choice.
For more information on these and other ways to support the CHEST Foundation, confidentially and with no obligation, contact Angela Perillo, CHEST Director of Development & Foundation Operations, at [email protected].
Your gift today truly has an immediate impact that makes a difference now.
We also want you to benefit as much as possible from your generosity.
Gifts of Appreciated Securities, Mutual Funds, and Investments
If you have owned any of these longer than 1 year and they have appreciated in value, they provide a smart option for gifting. You will avoid the capital gains tax, and you also receive a charitable income tax deduction if you itemize your tax return.
The Charitable Individual Retirement Plan Option
If you are 70 1/2, you may distribute funds from your IRA directly to the CHEST Foundation.
You will not pay any income taxes, and it will also qualify for your required minimum withdrawal. You may distribute up to $100,000 per person per year ($200,000 if you are married and both own an IRA).
Retirement Plan Beneficiary Designation
You may also designate a charity as a beneficiary of your IRA, 401K, or 403B.
This will avoid any income tax, so 100% will be directed to the charity of your choice.
For more information on these and other ways to support the CHEST Foundation, confidentially and with no obligation, contact Angela Perillo, CHEST Director of Development & Foundation Operations, at [email protected].
Your gift today truly has an immediate impact that makes a difference now.
We also want you to benefit as much as possible from your generosity.
Gifts of Appreciated Securities, Mutual Funds, and Investments
If you have owned any of these longer than 1 year and they have appreciated in value, they provide a smart option for gifting. You will avoid the capital gains tax, and you also receive a charitable income tax deduction if you itemize your tax return.
The Charitable Individual Retirement Plan Option
If you are 70 1/2, you may distribute funds from your IRA directly to the CHEST Foundation.
You will not pay any income taxes, and it will also qualify for your required minimum withdrawal. You may distribute up to $100,000 per person per year ($200,000 if you are married and both own an IRA).
Retirement Plan Beneficiary Designation
You may also designate a charity as a beneficiary of your IRA, 401K, or 403B.
This will avoid any income tax, so 100% will be directed to the charity of your choice.
For more information on these and other ways to support the CHEST Foundation, confidentially and with no obligation, contact Angela Perillo, CHEST Director of Development & Foundation Operations, at [email protected].
Congratulations, CHEST! 2017 Accreditation With Commendation
On December 2, CHEST received Accreditation with Commendation from the Accreditation Council for Continuing Medical Education (ACCME). This achievement grants CHEST accreditation through November 2023, and places the organization in the highest tier of continuing medical education (CME) providers.
“It is a true privilege to serve as a member of our outstanding CHEST Education team. We are very proud of our education program and have worked very hard to provide CHEST members and their health-care team with state-of-the-art learning opportunities,” said Alex Niven, MD, FCCP, current Chair of CHEST’s Education Committee, “ACCME Accreditation with Commendation is an important benchmark of this success, and we look forward to further advancing CHEST’s leadership role in medical education through its simulation, active learning, and other innovative educational offerings.”
To receive accreditation from the ACCME, CHEST met all of the requirements of the ACCME, has transitioned clinician knowledge into action, and has enhanced procedural performance to improve patient outcomes. Accreditation with Commendation is “a reward for going above and beyond requirements--having the absolute best practices and for striving to meet the aspirational goals of medical education,” said William Kelly, MD, FCCP, previous Chair of CHEST’s Education Committee.
In achieving Accreditation with Commendation, CHEST demonstrated compliance with the following:
• Improving the professional practice by consistently integrating CME into CHEST processes.
• Utilization of noneducation strategies such as the CHEST Foundation’s grant programs and disease awareness campaigns, to enhance change as an adjunct to CHEST’s activities/educational interventions.
• Identification of factors that effect patient outcomes and are outside of the provider’s control.
• Implementation of educational strategies, including the offering of additional training to improve procedural capabilities, so as to remove, overcome, or address barriers to physician change.
• Building of bridges with stakeholders such as The France Foundation, National Comprehensive Cancer Network (NCCN), and the American Society for Clinical Pathology (ASCP), through collaboration and cooperation.
• Participation within an institutional framework for health-care quality improvement.
• Positioned to influence the scope and content of activities/educational interventions.
On December 2, CHEST received Accreditation with Commendation from the Accreditation Council for Continuing Medical Education (ACCME). This achievement grants CHEST accreditation through November 2023, and places the organization in the highest tier of continuing medical education (CME) providers.
“It is a true privilege to serve as a member of our outstanding CHEST Education team. We are very proud of our education program and have worked very hard to provide CHEST members and their health-care team with state-of-the-art learning opportunities,” said Alex Niven, MD, FCCP, current Chair of CHEST’s Education Committee, “ACCME Accreditation with Commendation is an important benchmark of this success, and we look forward to further advancing CHEST’s leadership role in medical education through its simulation, active learning, and other innovative educational offerings.”
To receive accreditation from the ACCME, CHEST met all of the requirements of the ACCME, has transitioned clinician knowledge into action, and has enhanced procedural performance to improve patient outcomes. Accreditation with Commendation is “a reward for going above and beyond requirements--having the absolute best practices and for striving to meet the aspirational goals of medical education,” said William Kelly, MD, FCCP, previous Chair of CHEST’s Education Committee.
In achieving Accreditation with Commendation, CHEST demonstrated compliance with the following:
• Improving the professional practice by consistently integrating CME into CHEST processes.
• Utilization of noneducation strategies such as the CHEST Foundation’s grant programs and disease awareness campaigns, to enhance change as an adjunct to CHEST’s activities/educational interventions.
• Identification of factors that effect patient outcomes and are outside of the provider’s control.
• Implementation of educational strategies, including the offering of additional training to improve procedural capabilities, so as to remove, overcome, or address barriers to physician change.
• Building of bridges with stakeholders such as The France Foundation, National Comprehensive Cancer Network (NCCN), and the American Society for Clinical Pathology (ASCP), through collaboration and cooperation.
• Participation within an institutional framework for health-care quality improvement.
• Positioned to influence the scope and content of activities/educational interventions.
On December 2, CHEST received Accreditation with Commendation from the Accreditation Council for Continuing Medical Education (ACCME). This achievement grants CHEST accreditation through November 2023, and places the organization in the highest tier of continuing medical education (CME) providers.
“It is a true privilege to serve as a member of our outstanding CHEST Education team. We are very proud of our education program and have worked very hard to provide CHEST members and their health-care team with state-of-the-art learning opportunities,” said Alex Niven, MD, FCCP, current Chair of CHEST’s Education Committee, “ACCME Accreditation with Commendation is an important benchmark of this success, and we look forward to further advancing CHEST’s leadership role in medical education through its simulation, active learning, and other innovative educational offerings.”
To receive accreditation from the ACCME, CHEST met all of the requirements of the ACCME, has transitioned clinician knowledge into action, and has enhanced procedural performance to improve patient outcomes. Accreditation with Commendation is “a reward for going above and beyond requirements--having the absolute best practices and for striving to meet the aspirational goals of medical education,” said William Kelly, MD, FCCP, previous Chair of CHEST’s Education Committee.
In achieving Accreditation with Commendation, CHEST demonstrated compliance with the following:
• Improving the professional practice by consistently integrating CME into CHEST processes.
• Utilization of noneducation strategies such as the CHEST Foundation’s grant programs and disease awareness campaigns, to enhance change as an adjunct to CHEST’s activities/educational interventions.
• Identification of factors that effect patient outcomes and are outside of the provider’s control.
• Implementation of educational strategies, including the offering of additional training to improve procedural capabilities, so as to remove, overcome, or address barriers to physician change.
• Building of bridges with stakeholders such as The France Foundation, National Comprehensive Cancer Network (NCCN), and the American Society for Clinical Pathology (ASCP), through collaboration and cooperation.
• Participation within an institutional framework for health-care quality improvement.
• Positioned to influence the scope and content of activities/educational interventions.
Live Streaming at CHEST 2017
In April 2016, Facebook launched Facebook Live, a tool for live streaming to a Facebook page to share live video with their followers on Facebook. At CHEST 2016, the CHEST New Media team began to experiment with live video with some early success. The CHEST 2017 team made the decision, based on the organization’s goal to help educate clinicians to improve patient care, to live stream complete sessions from CHEST 2017. With the help of the CHEST 2017 Education Committee and the Social Media Work Group, more than 25 sessions were selected and live streamed.
CHEST’s efforts on Facebook Live resulted in the following:
- Total people reached: 133,737
- Total video views: 34,449
- Total minutes watched: 30,786 (or 513 hours, or 21 days)
- Total interactions: 1,050 (eg, likes, loves, hahas, etc)
- Total shares: 302
The content concept was well received, and comments ranged from followers chiming in with their location, appreciation for live streaming, and even comments from patients.
- “Thank you for sharing this live presentation.”
- “Here from Mexico !!”
- “Here from Natal/RN, Brazil”
- “Here from Milan, Italy.”
- “Appreciate this live streaming on important sessions, big service for those who couldn’t attend!!”
- “My brother survived after six days on ECMO. I am so glad to have him.”
- “It’s a great chance for physicians working in pulmonology and general practice to get the pearls of guidelines from American College to improve clinical practice. Now distance doesn’t matter”
Plans are underway for live streaming from CHEST 2018 in San Antonio. To view the CHEST 2017 live stream videos, visit CHEST’s Facebook page, facebook.com/accpchest.
In April 2016, Facebook launched Facebook Live, a tool for live streaming to a Facebook page to share live video with their followers on Facebook. At CHEST 2016, the CHEST New Media team began to experiment with live video with some early success. The CHEST 2017 team made the decision, based on the organization’s goal to help educate clinicians to improve patient care, to live stream complete sessions from CHEST 2017. With the help of the CHEST 2017 Education Committee and the Social Media Work Group, more than 25 sessions were selected and live streamed.
CHEST’s efforts on Facebook Live resulted in the following:
- Total people reached: 133,737
- Total video views: 34,449
- Total minutes watched: 30,786 (or 513 hours, or 21 days)
- Total interactions: 1,050 (eg, likes, loves, hahas, etc)
- Total shares: 302
The content concept was well received, and comments ranged from followers chiming in with their location, appreciation for live streaming, and even comments from patients.
- “Thank you for sharing this live presentation.”
- “Here from Mexico !!”
- “Here from Natal/RN, Brazil”
- “Here from Milan, Italy.”
- “Appreciate this live streaming on important sessions, big service for those who couldn’t attend!!”
- “My brother survived after six days on ECMO. I am so glad to have him.”
- “It’s a great chance for physicians working in pulmonology and general practice to get the pearls of guidelines from American College to improve clinical practice. Now distance doesn’t matter”
Plans are underway for live streaming from CHEST 2018 in San Antonio. To view the CHEST 2017 live stream videos, visit CHEST’s Facebook page, facebook.com/accpchest.
In April 2016, Facebook launched Facebook Live, a tool for live streaming to a Facebook page to share live video with their followers on Facebook. At CHEST 2016, the CHEST New Media team began to experiment with live video with some early success. The CHEST 2017 team made the decision, based on the organization’s goal to help educate clinicians to improve patient care, to live stream complete sessions from CHEST 2017. With the help of the CHEST 2017 Education Committee and the Social Media Work Group, more than 25 sessions were selected and live streamed.
CHEST’s efforts on Facebook Live resulted in the following:
- Total people reached: 133,737
- Total video views: 34,449
- Total minutes watched: 30,786 (or 513 hours, or 21 days)
- Total interactions: 1,050 (eg, likes, loves, hahas, etc)
- Total shares: 302
The content concept was well received, and comments ranged from followers chiming in with their location, appreciation for live streaming, and even comments from patients.
- “Thank you for sharing this live presentation.”
- “Here from Mexico !!”
- “Here from Natal/RN, Brazil”
- “Here from Milan, Italy.”
- “Appreciate this live streaming on important sessions, big service for those who couldn’t attend!!”
- “My brother survived after six days on ECMO. I am so glad to have him.”
- “It’s a great chance for physicians working in pulmonology and general practice to get the pearls of guidelines from American College to improve clinical practice. Now distance doesn’t matter”
Plans are underway for live streaming from CHEST 2018 in San Antonio. To view the CHEST 2017 live stream videos, visit CHEST’s Facebook page, facebook.com/accpchest.



