User login
Last call for VAM 2020 Proposals
The Society for Vascular Surgery seeks proposals for invited sessions for the 2020 Vascular Annual Meeting, to be held June 17 to 20 in Toronto, Ontario, Canada. Scientific sessions will be June 18 to 20 and exhibits will be open June 18 to 19. Proposals should include the session's educational benefit, a short outline of the program topic, session goals and target audience, among other information. Obtain the information/submission form here and send in before tomorrow, Aug. 28. Email completed forms to [email protected].
The Society for Vascular Surgery seeks proposals for invited sessions for the 2020 Vascular Annual Meeting, to be held June 17 to 20 in Toronto, Ontario, Canada. Scientific sessions will be June 18 to 20 and exhibits will be open June 18 to 19. Proposals should include the session's educational benefit, a short outline of the program topic, session goals and target audience, among other information. Obtain the information/submission form here and send in before tomorrow, Aug. 28. Email completed forms to [email protected].
The Society for Vascular Surgery seeks proposals for invited sessions for the 2020 Vascular Annual Meeting, to be held June 17 to 20 in Toronto, Ontario, Canada. Scientific sessions will be June 18 to 20 and exhibits will be open June 18 to 19. Proposals should include the session's educational benefit, a short outline of the program topic, session goals and target audience, among other information. Obtain the information/submission form here and send in before tomorrow, Aug. 28. Email completed forms to [email protected].
Tell us what you think of our Patient Education Materials
Last year, the SVS Foundation updated and rewrote its popular patient education fliers. These are designed to fulfill the Foundation’s mission to support patient education, vascular health and community awareness of vascular disease. SVS members are encouraged to download and print the fliers to use in their offices. If you’ve taken advantage of these fliers, we’d like to hear how you’ve benefited from them. Please take this short survey and provide your feedback. If you have yet to see the fliers, view them here.
Last year, the SVS Foundation updated and rewrote its popular patient education fliers. These are designed to fulfill the Foundation’s mission to support patient education, vascular health and community awareness of vascular disease. SVS members are encouraged to download and print the fliers to use in their offices. If you’ve taken advantage of these fliers, we’d like to hear how you’ve benefited from them. Please take this short survey and provide your feedback. If you have yet to see the fliers, view them here.
Last year, the SVS Foundation updated and rewrote its popular patient education fliers. These are designed to fulfill the Foundation’s mission to support patient education, vascular health and community awareness of vascular disease. SVS members are encouraged to download and print the fliers to use in their offices. If you’ve taken advantage of these fliers, we’d like to hear how you’ve benefited from them. Please take this short survey and provide your feedback. If you have yet to see the fliers, view them here.
Insurance networks: Is it time to abandon them?
Bob has been coming to therapy for a few months now. Initially, we met every week, but as his depression lifted, he asked to space the sessions out to twice a month, even as he continues to struggle with many challenges in his life. The cost, he says, is prohibitive, and while Bob believed he had good commercial insurance coverage, he’s learned a few things about insurance and mental health care.
Bob was referred to me by his internist. He knew I did not participate in his insurance network, but his policy covers out-of-network care. He’d had a number of imaging studies and then knee surgery earlier in the year, so he believed he’d met his deductible. He learned that, while he’d met his in-network deductible, he’d had no out-of-network expenses and there was a separate, much higher deductible – one he was not likely to meet with outpatient psychiatric care. In fact, the full cost of his treatment was not being subtracted from the deductible he needed to meet, but rather he was getting credit for lower usual and customary evaluation and management and psychotherapy fees for each session. It became clear that it would be many months – if ever – before Bob could expect any reimbursement for his out-of-network visits.
What Bob didn’t know was that, had he decided to switch to an in-network psychiatrist, he might well have trouble finding one, since half of psychiatrists don’t participate with any health insurance plans. And if he did see an in-network psychiatrist, he would likely need to find a separate in-network social worker or psychologist for psychotherapy, because most in-network psychiatrists see patients for short medication-management appointments. While the insurance companies would give Bob a list of providers, those lists are not kept up to date and include psychiatrists who have died, moved, aren’t taking new patients, or who have retired. The insurance company clearly states on its voicemail that verification of services does not guarantee payment, and Bob was told that the only way he could be certain of the reimbursement would be to submit the claims and wait. He went into treatment fully understanding that he might get no help with the cost from his health insurance.
When I first started in private practice in the 1990s, I joined only one panel. An older colleague told me I was foolish to hesitate, and that soon the panels would fill and it would be too late; psychiatrists wouldn’t be able to get on to the panels and would be unable to attract patients. A few years later, that same psychiatrist withdrew from all the insurance panels he was on; working on their terms was not rewarding. This division of in-network and out-of-network care is crucial to managed care: They must attract panels of doctors who will work for lower rates or with stipulations on how the doctor practices in order to save money.
But managed care came with a price: An entire administration was created to oversee the regulation of treatment. With time, some aspects of care management have vanished; it has been years since I have been asked to fill out a treatment plan to justify a need for outpatient psychotherapy. In Bob’s case, it’s clear how they save money; since he will not reach his high deductible, he will bear the full cost of his psychotherapy. Other patients who cannot afford to go out of network may give up searching and decide to go without treatment – this is not always an easy service to negotiate when one is distressed and compromised. Half of people with serious psychiatric disorders are not in treatment, and barriers to getting care are certainly one reason why.
Many psychiatrists have discovered that they can maintain a practice without being on insurance panels, as managed care only works if there are enough players willing to toss the ball. As psychiatrists have shied away from these panels, insurers have raised their reimbursement rates, and in Maryland, Medicaid also has had to raise their rates. The struggle has become one of how to get enough mental health professionals, and psychiatrists in particular, to join insurance panels in what is a shortage field.
Perhaps there are better ways to spend health care dollars than on the administration and management that come with limiting which doctors patients can see. The logistics of in-network versus out-of-network care are an expensive one, and create unconscionable scenarios in other fields. For example, such scenarios include ones in which a patient is brought in for emergency care to a facility where the doctors are not in network, or a patient has a procedure with an in-network surgeon but is unaware that the anesthesiologist or other members of the care team are not on the panel.
What if insurers controlled costs by setting a reasonable fee they would pay for services and paid any licensed physician for these services? Would market forces sort this out? Would fees then set to one which insurers would be willing to pay and physicians would be willing to accept? Or would those who are ill and impoverished be blocked from getting any care? What if we tried a whole new paradigm for psychiatric care?
Richard G. Frank, PhD, is a professor of health economics at Harvard. He specializes in mental health economics and is coauthor of the book “Better But Not Well: Mental Health Policy in the United States Since 1950” (Baltimore: Johns Hopkins University Press, 2006). Dr. Frank is a proponent of insurance panels.
“In the world we live in, we need to have panels; they are essential to controlling cost. They create a balance in terms of cost and utilization control in ways that protect patients, and they create a way for the insurance companies to bargain,” he said.
Dr. Frank noted that the concept that insurers should pay a set reasonable fee to any physician a patient wants to see has been tried. “It’s called ‘reference pricing,’ and when they did that with hip replacement surgery in California, it just hasn’t worked out. Patients ended up getting larger bills than they anticipated.
“The issues with psychiatry are different,” he continued. “There is a lot of bad behavior on the part of insurers and it’s an issue of parity. We shouldn’t let insurers differentially pay psychiatrists less. They have had an incentive to reduce the availability of mental health care and it’s bad for patients. It’s about lower payments to psychiatrists and the way those payments are currently structured drives patients out of care and defeats the purpose of insurance.”
Steven Sharfstein, MD, is the former CEO of Sheppard Pratt Health Systems, a past president of the American Psychiatric Association, and coauthor of several books on the economics of psychiatry. He refers to the current practice of credentialing network psychiatrists as a means of “rationing by supply.”
“The networks create a barrier to accessing care. I don’t think it’s an efficient way to take on the high cost of care, and it creates a tiered system.” Like Dr. Frank, Dr. Sharfstein believes parity is a large part of the problem. When asked about the idea of ending networks and establishing uniform deductibles and reimbursement rates, Dr. Sharfstein replied: “It really depends. We would need fees to hit a sweet spot that supports care while controlling costs.”
One thing is clear: With the current paradigm, it is often difficult to access treatment, and the well-insured patients may bear a significant and disproportionate cost (if not the entire cost) for getting care. Many who need care do not get it, regardless of their insurance status. Our status quo for psychiatric care falls short and I don’t predict that psychiatrists will rush to join networks so long as the demand for psychiatrists is greater than the supply.
Might another financial model work better? We know the system is lacking and one option, as Dr. Frank suggests, is to find solutions within the current model. But perhaps the question should not be one of how to get more psychiatrists to join networks, but of how to rework the system without the assumption that networks are the only way. While Bob is pleased that his symptoms are getting better and he’s managed a way to tackle his own bills, it’s certainly time to explore new ways of delivering and reimbursing psychiatric care.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle of Inpatient Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016), and has a private practice in Baltimore.
Bob has been coming to therapy for a few months now. Initially, we met every week, but as his depression lifted, he asked to space the sessions out to twice a month, even as he continues to struggle with many challenges in his life. The cost, he says, is prohibitive, and while Bob believed he had good commercial insurance coverage, he’s learned a few things about insurance and mental health care.
Bob was referred to me by his internist. He knew I did not participate in his insurance network, but his policy covers out-of-network care. He’d had a number of imaging studies and then knee surgery earlier in the year, so he believed he’d met his deductible. He learned that, while he’d met his in-network deductible, he’d had no out-of-network expenses and there was a separate, much higher deductible – one he was not likely to meet with outpatient psychiatric care. In fact, the full cost of his treatment was not being subtracted from the deductible he needed to meet, but rather he was getting credit for lower usual and customary evaluation and management and psychotherapy fees for each session. It became clear that it would be many months – if ever – before Bob could expect any reimbursement for his out-of-network visits.
What Bob didn’t know was that, had he decided to switch to an in-network psychiatrist, he might well have trouble finding one, since half of psychiatrists don’t participate with any health insurance plans. And if he did see an in-network psychiatrist, he would likely need to find a separate in-network social worker or psychologist for psychotherapy, because most in-network psychiatrists see patients for short medication-management appointments. While the insurance companies would give Bob a list of providers, those lists are not kept up to date and include psychiatrists who have died, moved, aren’t taking new patients, or who have retired. The insurance company clearly states on its voicemail that verification of services does not guarantee payment, and Bob was told that the only way he could be certain of the reimbursement would be to submit the claims and wait. He went into treatment fully understanding that he might get no help with the cost from his health insurance.
When I first started in private practice in the 1990s, I joined only one panel. An older colleague told me I was foolish to hesitate, and that soon the panels would fill and it would be too late; psychiatrists wouldn’t be able to get on to the panels and would be unable to attract patients. A few years later, that same psychiatrist withdrew from all the insurance panels he was on; working on their terms was not rewarding. This division of in-network and out-of-network care is crucial to managed care: They must attract panels of doctors who will work for lower rates or with stipulations on how the doctor practices in order to save money.
But managed care came with a price: An entire administration was created to oversee the regulation of treatment. With time, some aspects of care management have vanished; it has been years since I have been asked to fill out a treatment plan to justify a need for outpatient psychotherapy. In Bob’s case, it’s clear how they save money; since he will not reach his high deductible, he will bear the full cost of his psychotherapy. Other patients who cannot afford to go out of network may give up searching and decide to go without treatment – this is not always an easy service to negotiate when one is distressed and compromised. Half of people with serious psychiatric disorders are not in treatment, and barriers to getting care are certainly one reason why.
Many psychiatrists have discovered that they can maintain a practice without being on insurance panels, as managed care only works if there are enough players willing to toss the ball. As psychiatrists have shied away from these panels, insurers have raised their reimbursement rates, and in Maryland, Medicaid also has had to raise their rates. The struggle has become one of how to get enough mental health professionals, and psychiatrists in particular, to join insurance panels in what is a shortage field.
Perhaps there are better ways to spend health care dollars than on the administration and management that come with limiting which doctors patients can see. The logistics of in-network versus out-of-network care are an expensive one, and create unconscionable scenarios in other fields. For example, such scenarios include ones in which a patient is brought in for emergency care to a facility where the doctors are not in network, or a patient has a procedure with an in-network surgeon but is unaware that the anesthesiologist or other members of the care team are not on the panel.
What if insurers controlled costs by setting a reasonable fee they would pay for services and paid any licensed physician for these services? Would market forces sort this out? Would fees then set to one which insurers would be willing to pay and physicians would be willing to accept? Or would those who are ill and impoverished be blocked from getting any care? What if we tried a whole new paradigm for psychiatric care?
Richard G. Frank, PhD, is a professor of health economics at Harvard. He specializes in mental health economics and is coauthor of the book “Better But Not Well: Mental Health Policy in the United States Since 1950” (Baltimore: Johns Hopkins University Press, 2006). Dr. Frank is a proponent of insurance panels.
“In the world we live in, we need to have panels; they are essential to controlling cost. They create a balance in terms of cost and utilization control in ways that protect patients, and they create a way for the insurance companies to bargain,” he said.
Dr. Frank noted that the concept that insurers should pay a set reasonable fee to any physician a patient wants to see has been tried. “It’s called ‘reference pricing,’ and when they did that with hip replacement surgery in California, it just hasn’t worked out. Patients ended up getting larger bills than they anticipated.
“The issues with psychiatry are different,” he continued. “There is a lot of bad behavior on the part of insurers and it’s an issue of parity. We shouldn’t let insurers differentially pay psychiatrists less. They have had an incentive to reduce the availability of mental health care and it’s bad for patients. It’s about lower payments to psychiatrists and the way those payments are currently structured drives patients out of care and defeats the purpose of insurance.”
Steven Sharfstein, MD, is the former CEO of Sheppard Pratt Health Systems, a past president of the American Psychiatric Association, and coauthor of several books on the economics of psychiatry. He refers to the current practice of credentialing network psychiatrists as a means of “rationing by supply.”
“The networks create a barrier to accessing care. I don’t think it’s an efficient way to take on the high cost of care, and it creates a tiered system.” Like Dr. Frank, Dr. Sharfstein believes parity is a large part of the problem. When asked about the idea of ending networks and establishing uniform deductibles and reimbursement rates, Dr. Sharfstein replied: “It really depends. We would need fees to hit a sweet spot that supports care while controlling costs.”
One thing is clear: With the current paradigm, it is often difficult to access treatment, and the well-insured patients may bear a significant and disproportionate cost (if not the entire cost) for getting care. Many who need care do not get it, regardless of their insurance status. Our status quo for psychiatric care falls short and I don’t predict that psychiatrists will rush to join networks so long as the demand for psychiatrists is greater than the supply.
Might another financial model work better? We know the system is lacking and one option, as Dr. Frank suggests, is to find solutions within the current model. But perhaps the question should not be one of how to get more psychiatrists to join networks, but of how to rework the system without the assumption that networks are the only way. While Bob is pleased that his symptoms are getting better and he’s managed a way to tackle his own bills, it’s certainly time to explore new ways of delivering and reimbursing psychiatric care.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle of Inpatient Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016), and has a private practice in Baltimore.
Bob has been coming to therapy for a few months now. Initially, we met every week, but as his depression lifted, he asked to space the sessions out to twice a month, even as he continues to struggle with many challenges in his life. The cost, he says, is prohibitive, and while Bob believed he had good commercial insurance coverage, he’s learned a few things about insurance and mental health care.
Bob was referred to me by his internist. He knew I did not participate in his insurance network, but his policy covers out-of-network care. He’d had a number of imaging studies and then knee surgery earlier in the year, so he believed he’d met his deductible. He learned that, while he’d met his in-network deductible, he’d had no out-of-network expenses and there was a separate, much higher deductible – one he was not likely to meet with outpatient psychiatric care. In fact, the full cost of his treatment was not being subtracted from the deductible he needed to meet, but rather he was getting credit for lower usual and customary evaluation and management and psychotherapy fees for each session. It became clear that it would be many months – if ever – before Bob could expect any reimbursement for his out-of-network visits.
What Bob didn’t know was that, had he decided to switch to an in-network psychiatrist, he might well have trouble finding one, since half of psychiatrists don’t participate with any health insurance plans. And if he did see an in-network psychiatrist, he would likely need to find a separate in-network social worker or psychologist for psychotherapy, because most in-network psychiatrists see patients for short medication-management appointments. While the insurance companies would give Bob a list of providers, those lists are not kept up to date and include psychiatrists who have died, moved, aren’t taking new patients, or who have retired. The insurance company clearly states on its voicemail that verification of services does not guarantee payment, and Bob was told that the only way he could be certain of the reimbursement would be to submit the claims and wait. He went into treatment fully understanding that he might get no help with the cost from his health insurance.
When I first started in private practice in the 1990s, I joined only one panel. An older colleague told me I was foolish to hesitate, and that soon the panels would fill and it would be too late; psychiatrists wouldn’t be able to get on to the panels and would be unable to attract patients. A few years later, that same psychiatrist withdrew from all the insurance panels he was on; working on their terms was not rewarding. This division of in-network and out-of-network care is crucial to managed care: They must attract panels of doctors who will work for lower rates or with stipulations on how the doctor practices in order to save money.
But managed care came with a price: An entire administration was created to oversee the regulation of treatment. With time, some aspects of care management have vanished; it has been years since I have been asked to fill out a treatment plan to justify a need for outpatient psychotherapy. In Bob’s case, it’s clear how they save money; since he will not reach his high deductible, he will bear the full cost of his psychotherapy. Other patients who cannot afford to go out of network may give up searching and decide to go without treatment – this is not always an easy service to negotiate when one is distressed and compromised. Half of people with serious psychiatric disorders are not in treatment, and barriers to getting care are certainly one reason why.
Many psychiatrists have discovered that they can maintain a practice without being on insurance panels, as managed care only works if there are enough players willing to toss the ball. As psychiatrists have shied away from these panels, insurers have raised their reimbursement rates, and in Maryland, Medicaid also has had to raise their rates. The struggle has become one of how to get enough mental health professionals, and psychiatrists in particular, to join insurance panels in what is a shortage field.
Perhaps there are better ways to spend health care dollars than on the administration and management that come with limiting which doctors patients can see. The logistics of in-network versus out-of-network care are an expensive one, and create unconscionable scenarios in other fields. For example, such scenarios include ones in which a patient is brought in for emergency care to a facility where the doctors are not in network, or a patient has a procedure with an in-network surgeon but is unaware that the anesthesiologist or other members of the care team are not on the panel.
What if insurers controlled costs by setting a reasonable fee they would pay for services and paid any licensed physician for these services? Would market forces sort this out? Would fees then set to one which insurers would be willing to pay and physicians would be willing to accept? Or would those who are ill and impoverished be blocked from getting any care? What if we tried a whole new paradigm for psychiatric care?
Richard G. Frank, PhD, is a professor of health economics at Harvard. He specializes in mental health economics and is coauthor of the book “Better But Not Well: Mental Health Policy in the United States Since 1950” (Baltimore: Johns Hopkins University Press, 2006). Dr. Frank is a proponent of insurance panels.
“In the world we live in, we need to have panels; they are essential to controlling cost. They create a balance in terms of cost and utilization control in ways that protect patients, and they create a way for the insurance companies to bargain,” he said.
Dr. Frank noted that the concept that insurers should pay a set reasonable fee to any physician a patient wants to see has been tried. “It’s called ‘reference pricing,’ and when they did that with hip replacement surgery in California, it just hasn’t worked out. Patients ended up getting larger bills than they anticipated.
“The issues with psychiatry are different,” he continued. “There is a lot of bad behavior on the part of insurers and it’s an issue of parity. We shouldn’t let insurers differentially pay psychiatrists less. They have had an incentive to reduce the availability of mental health care and it’s bad for patients. It’s about lower payments to psychiatrists and the way those payments are currently structured drives patients out of care and defeats the purpose of insurance.”
Steven Sharfstein, MD, is the former CEO of Sheppard Pratt Health Systems, a past president of the American Psychiatric Association, and coauthor of several books on the economics of psychiatry. He refers to the current practice of credentialing network psychiatrists as a means of “rationing by supply.”
“The networks create a barrier to accessing care. I don’t think it’s an efficient way to take on the high cost of care, and it creates a tiered system.” Like Dr. Frank, Dr. Sharfstein believes parity is a large part of the problem. When asked about the idea of ending networks and establishing uniform deductibles and reimbursement rates, Dr. Sharfstein replied: “It really depends. We would need fees to hit a sweet spot that supports care while controlling costs.”
One thing is clear: With the current paradigm, it is often difficult to access treatment, and the well-insured patients may bear a significant and disproportionate cost (if not the entire cost) for getting care. Many who need care do not get it, regardless of their insurance status. Our status quo for psychiatric care falls short and I don’t predict that psychiatrists will rush to join networks so long as the demand for psychiatrists is greater than the supply.
Might another financial model work better? We know the system is lacking and one option, as Dr. Frank suggests, is to find solutions within the current model. But perhaps the question should not be one of how to get more psychiatrists to join networks, but of how to rework the system without the assumption that networks are the only way. While Bob is pleased that his symptoms are getting better and he’s managed a way to tackle his own bills, it’s certainly time to explore new ways of delivering and reimbursing psychiatric care.
Dr. Miller is coauthor with Annette Hanson, MD, of “Committed: The Battle of Inpatient Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016), and has a private practice in Baltimore.
Pretreatment CT data may help predict immunotherapy benefit in ovarian cancer
Pretreatment CT data may help identify responders to immunotherapy in ovarian cancer, according to a new study.
Specifically, fewer sites of disease and lower intratumor heterogeneity on contrast-enhanced CT may indicate a higher likelihood of durable response to immune checkpoint inhibitors, according to results of the retrospective study, recently published in JCO Precision Oncology.
“Our results suggest that quantitative analysis of baseline contrast-enhanced CT may facilitate the delivery of precision medicine to patients with ovarian cancer by identifying patients who may benefit from immunotherapy,” wrote Yuki Himoto, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, and colleagues.
The study leverages findings from the emerging field of radiomics, which the investigators note allows for “virtual sampling” of tumor heterogeneity within a single lesion and between lesions.
“This information may complement molecular profiling in personalizing medical decisions,” Dr. Himoto and coauthors explained.
The study cohort included 75 patients with recurrent ovarian cancer who were enrolled in ongoing, prospective trials of immunotherapy, according to the researchers. Of that group, just under one in five derived a durable clinical benefit, defined as progression-free survival lasting at least 24 weeks.
In univariable analysis, they found a number of contrast-enhanced CT variables were linked to durable clinical benefit, including fewer disease sites, lower cluster-site entropy and dissimilarity, which they wrote were an indicator of lower intertumor heterogeneity, and higher energy in the largest-volume lesion, which they described as an indicator of lower intratumor heterogeneity.
However, in multivariable analysis, the only variables that were still associated with durable clinical benefit were fewer disease sites (odds ratio, 1.64; 95% confidence interval, 1.19-2.27; P = .012) and higher energy in the largest lesion (odds ratio, 1.41; 95% CI, 1.11-1.81; P = .006), according to the report.
Those two factors combined were a composite indicator of durable clinical benefit (C-index, 0.821).
These findings could represent a step forward in the provision of immunotherapy in ovarian cancer, which exhibits poor response to immune checkpoint inhibitors, compared with some other cancer types, the investigators wrote.
More insights are needed, however, to help personalize the selection of immunotherapy in ovarian cancer, including a better understanding of cancer immune reactions and retooling of immune response criteria, they added.
“Composite multimodal multifaceted biomarkers that noninvasively capture spatiotemporal tumor heterogeneity will likely be necessary to comprehensively assess immune the tumor microenvironment and serve as clinical decision support for prognosis inference and prediction of response,” Dr. Himoto and associates wrote.
The study was supported by the National Cancer Institute, among other sources. Study authors reported disclosures related to Merck, Bristol-Myers Squibb, Genentech, Celgene, AstraZeneca, Y-mAbs Therapeutics, and others.
SOURCE: Himoto Y et al. JCO Precis Oncol. 2019 Aug 13. doi: 10.1200/PO.19.00038.
Pretreatment CT data may help identify responders to immunotherapy in ovarian cancer, according to a new study.
Specifically, fewer sites of disease and lower intratumor heterogeneity on contrast-enhanced CT may indicate a higher likelihood of durable response to immune checkpoint inhibitors, according to results of the retrospective study, recently published in JCO Precision Oncology.
“Our results suggest that quantitative analysis of baseline contrast-enhanced CT may facilitate the delivery of precision medicine to patients with ovarian cancer by identifying patients who may benefit from immunotherapy,” wrote Yuki Himoto, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, and colleagues.
The study leverages findings from the emerging field of radiomics, which the investigators note allows for “virtual sampling” of tumor heterogeneity within a single lesion and between lesions.
“This information may complement molecular profiling in personalizing medical decisions,” Dr. Himoto and coauthors explained.
The study cohort included 75 patients with recurrent ovarian cancer who were enrolled in ongoing, prospective trials of immunotherapy, according to the researchers. Of that group, just under one in five derived a durable clinical benefit, defined as progression-free survival lasting at least 24 weeks.
In univariable analysis, they found a number of contrast-enhanced CT variables were linked to durable clinical benefit, including fewer disease sites, lower cluster-site entropy and dissimilarity, which they wrote were an indicator of lower intertumor heterogeneity, and higher energy in the largest-volume lesion, which they described as an indicator of lower intratumor heterogeneity.
However, in multivariable analysis, the only variables that were still associated with durable clinical benefit were fewer disease sites (odds ratio, 1.64; 95% confidence interval, 1.19-2.27; P = .012) and higher energy in the largest lesion (odds ratio, 1.41; 95% CI, 1.11-1.81; P = .006), according to the report.
Those two factors combined were a composite indicator of durable clinical benefit (C-index, 0.821).
These findings could represent a step forward in the provision of immunotherapy in ovarian cancer, which exhibits poor response to immune checkpoint inhibitors, compared with some other cancer types, the investigators wrote.
More insights are needed, however, to help personalize the selection of immunotherapy in ovarian cancer, including a better understanding of cancer immune reactions and retooling of immune response criteria, they added.
“Composite multimodal multifaceted biomarkers that noninvasively capture spatiotemporal tumor heterogeneity will likely be necessary to comprehensively assess immune the tumor microenvironment and serve as clinical decision support for prognosis inference and prediction of response,” Dr. Himoto and associates wrote.
The study was supported by the National Cancer Institute, among other sources. Study authors reported disclosures related to Merck, Bristol-Myers Squibb, Genentech, Celgene, AstraZeneca, Y-mAbs Therapeutics, and others.
SOURCE: Himoto Y et al. JCO Precis Oncol. 2019 Aug 13. doi: 10.1200/PO.19.00038.
Pretreatment CT data may help identify responders to immunotherapy in ovarian cancer, according to a new study.
Specifically, fewer sites of disease and lower intratumor heterogeneity on contrast-enhanced CT may indicate a higher likelihood of durable response to immune checkpoint inhibitors, according to results of the retrospective study, recently published in JCO Precision Oncology.
“Our results suggest that quantitative analysis of baseline contrast-enhanced CT may facilitate the delivery of precision medicine to patients with ovarian cancer by identifying patients who may benefit from immunotherapy,” wrote Yuki Himoto, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York, and colleagues.
The study leverages findings from the emerging field of radiomics, which the investigators note allows for “virtual sampling” of tumor heterogeneity within a single lesion and between lesions.
“This information may complement molecular profiling in personalizing medical decisions,” Dr. Himoto and coauthors explained.
The study cohort included 75 patients with recurrent ovarian cancer who were enrolled in ongoing, prospective trials of immunotherapy, according to the researchers. Of that group, just under one in five derived a durable clinical benefit, defined as progression-free survival lasting at least 24 weeks.
In univariable analysis, they found a number of contrast-enhanced CT variables were linked to durable clinical benefit, including fewer disease sites, lower cluster-site entropy and dissimilarity, which they wrote were an indicator of lower intertumor heterogeneity, and higher energy in the largest-volume lesion, which they described as an indicator of lower intratumor heterogeneity.
However, in multivariable analysis, the only variables that were still associated with durable clinical benefit were fewer disease sites (odds ratio, 1.64; 95% confidence interval, 1.19-2.27; P = .012) and higher energy in the largest lesion (odds ratio, 1.41; 95% CI, 1.11-1.81; P = .006), according to the report.
Those two factors combined were a composite indicator of durable clinical benefit (C-index, 0.821).
These findings could represent a step forward in the provision of immunotherapy in ovarian cancer, which exhibits poor response to immune checkpoint inhibitors, compared with some other cancer types, the investigators wrote.
More insights are needed, however, to help personalize the selection of immunotherapy in ovarian cancer, including a better understanding of cancer immune reactions and retooling of immune response criteria, they added.
“Composite multimodal multifaceted biomarkers that noninvasively capture spatiotemporal tumor heterogeneity will likely be necessary to comprehensively assess immune the tumor microenvironment and serve as clinical decision support for prognosis inference and prediction of response,” Dr. Himoto and associates wrote.
The study was supported by the National Cancer Institute, among other sources. Study authors reported disclosures related to Merck, Bristol-Myers Squibb, Genentech, Celgene, AstraZeneca, Y-mAbs Therapeutics, and others.
SOURCE: Himoto Y et al. JCO Precis Oncol. 2019 Aug 13. doi: 10.1200/PO.19.00038.
FROM JCO PRECISION ONCOLOGY
Researchers win grants to study real-world cancer data
Three researchers have won CancerLinQ Discovery Research Support Grants from the American Society of Clinical Oncology, and two researchers have received a National Institutes of Health R21 grant.
Igor Astsaturov, MD, PhD, and Edna Cukierman, PhD, both of Fox Chase Cancer Center in Philadelphia, won the R21 grant. The pair will receive $432,410 over 2 years for their research on pancreatic cancer.
With their work, Dr. Astsaturov and Dr. Cukierman are “hoping to describe the structural and functional nature of cell-cell contact, or oncogenic synapses, associated with cancer-associated fibroblasts and pancreatic cells,” according to Fox Chase.
Three other researchers have won ASCO’s CancerLinQ Discovery Research Support Grants. The recipients will conduct projects using CancerLinQ, which collects and analyzes real-world data from cancer patients at practices across the United States.
Each 1-year grant covers the cost of a CancerLinQ Discovery data set and a meeting at ASCO headquarters. The grants also contribute to personnel and/or research expenses. The grants are funded by the ASCO Foundation’s Mission Endowment of Conquer Cancer.
With his CancerLinQ Discovery grant, Sadiq Rehmani, MD, of Icahn School of Medicine at Mount Sinai in New York, will study immunotherapy in older lung cancer patients with comorbidities.
Grant recipient Yasmin Karimi, MD, of Stanford (Calif.) University, will study how osteoclast inhibitors affect skeletal-related events and mortality in “real-world” patients with metastatic breast cancer and bone metastasis.
Grant recipient Vinayak Muralidhar, MD, of Brigham and Women’s Hospital and the radiation oncology program at Harvard Medical School, Boston, will study the use of androgen-deprivation therapy and hypofractionation in prostate cancer.
Movers in Medicine highlights career moves and personal achievements by hematologists and oncologists. Did you switch jobs, take on a new role, climb a mountain? Tell us all about it at [email protected], and you could be featured in Movers in Medicine.
Three researchers have won CancerLinQ Discovery Research Support Grants from the American Society of Clinical Oncology, and two researchers have received a National Institutes of Health R21 grant.
Igor Astsaturov, MD, PhD, and Edna Cukierman, PhD, both of Fox Chase Cancer Center in Philadelphia, won the R21 grant. The pair will receive $432,410 over 2 years for their research on pancreatic cancer.
With their work, Dr. Astsaturov and Dr. Cukierman are “hoping to describe the structural and functional nature of cell-cell contact, or oncogenic synapses, associated with cancer-associated fibroblasts and pancreatic cells,” according to Fox Chase.
Three other researchers have won ASCO’s CancerLinQ Discovery Research Support Grants. The recipients will conduct projects using CancerLinQ, which collects and analyzes real-world data from cancer patients at practices across the United States.
Each 1-year grant covers the cost of a CancerLinQ Discovery data set and a meeting at ASCO headquarters. The grants also contribute to personnel and/or research expenses. The grants are funded by the ASCO Foundation’s Mission Endowment of Conquer Cancer.
With his CancerLinQ Discovery grant, Sadiq Rehmani, MD, of Icahn School of Medicine at Mount Sinai in New York, will study immunotherapy in older lung cancer patients with comorbidities.
Grant recipient Yasmin Karimi, MD, of Stanford (Calif.) University, will study how osteoclast inhibitors affect skeletal-related events and mortality in “real-world” patients with metastatic breast cancer and bone metastasis.
Grant recipient Vinayak Muralidhar, MD, of Brigham and Women’s Hospital and the radiation oncology program at Harvard Medical School, Boston, will study the use of androgen-deprivation therapy and hypofractionation in prostate cancer.
Movers in Medicine highlights career moves and personal achievements by hematologists and oncologists. Did you switch jobs, take on a new role, climb a mountain? Tell us all about it at [email protected], and you could be featured in Movers in Medicine.
Three researchers have won CancerLinQ Discovery Research Support Grants from the American Society of Clinical Oncology, and two researchers have received a National Institutes of Health R21 grant.
Igor Astsaturov, MD, PhD, and Edna Cukierman, PhD, both of Fox Chase Cancer Center in Philadelphia, won the R21 grant. The pair will receive $432,410 over 2 years for their research on pancreatic cancer.
With their work, Dr. Astsaturov and Dr. Cukierman are “hoping to describe the structural and functional nature of cell-cell contact, or oncogenic synapses, associated with cancer-associated fibroblasts and pancreatic cells,” according to Fox Chase.
Three other researchers have won ASCO’s CancerLinQ Discovery Research Support Grants. The recipients will conduct projects using CancerLinQ, which collects and analyzes real-world data from cancer patients at practices across the United States.
Each 1-year grant covers the cost of a CancerLinQ Discovery data set and a meeting at ASCO headquarters. The grants also contribute to personnel and/or research expenses. The grants are funded by the ASCO Foundation’s Mission Endowment of Conquer Cancer.
With his CancerLinQ Discovery grant, Sadiq Rehmani, MD, of Icahn School of Medicine at Mount Sinai in New York, will study immunotherapy in older lung cancer patients with comorbidities.
Grant recipient Yasmin Karimi, MD, of Stanford (Calif.) University, will study how osteoclast inhibitors affect skeletal-related events and mortality in “real-world” patients with metastatic breast cancer and bone metastasis.
Grant recipient Vinayak Muralidhar, MD, of Brigham and Women’s Hospital and the radiation oncology program at Harvard Medical School, Boston, will study the use of androgen-deprivation therapy and hypofractionation in prostate cancer.
Movers in Medicine highlights career moves and personal achievements by hematologists and oncologists. Did you switch jobs, take on a new role, climb a mountain? Tell us all about it at [email protected], and you could be featured in Movers in Medicine.
What is your diagnosis? - September 2019
Erosive protein-losing enteropathy secondary to disseminated histoplasmosis
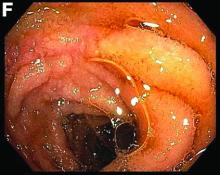
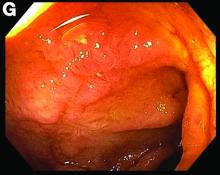
This patient was treated with amphotericin B and transitioned to oral itraconazole with frequent blood level monitoring to ensure absorption. His symptoms improved gradually. Small-bowel enteroscopy 3 weeks after presentation showed a normal duodenum and healing, superficial ulcers in the proximal jejunum (Figure F, G). Blood albumin levels had recovered to 3.1 g/dL (normal, 3.5–5.0 g/dL).
Protein-losing enteropathy (PLE) is a rare syndrome characterized by loss of serum proteins in the gastrointestinal (GI) tract, resulting in significant hypoproteinemia and consequent edema.1 PLE can also result in ascites, pleural and pericardial effusions, and, in prolonged cases, malnutrition. There are a variety of causes of PLE that can be broadly grouped into erosive GI disorders, disorders of increased GI mucosal permeability, and disorders of increased interstitial pressure. The clinical presentation depends on the underlying etiology, but commonly includes generalized edema owing to hypoproteinemia and resulting reduced oncotic pressure. GI symptoms are not frequently observed. The initial step in evaluating a patient with symptoms concerning for PLE is to rule out more common causes of hypoproteinemia, such as renal or hepatic disease, and malnutrition. To confirm enteric protein loss, alpha 1-antitrypsin clearance with a 24-hour stool collection is commonly and reliably used. Treatment of PLE is centered on treating the underlying cause while monitoring and treating malnutrition, including micronutrient deficiencies.
Fungal infections are a rare cause of PLE, but important to recognize as a potential complication of tumor necrosis factor–therapy, because these medications are commonly used for a variety of autoimmune diseases.2 Although histoplasmosis is an uncommon cause of GI inflammation, disseminated histoplasmosis causing PLE has been previously reported.3 In our patient, Histoplasma capsulatum infection caused diffuse GI ulcers, which allowed protein loss in the GI tract (erosive PLE). Antifungal treatment resulted in healing of intestinal ulcers and correction of hypoalbuminemia, thereby confirming the diagnosis of PLE and obviating the need for a confirmatory alpha 1-antitrypsin clearance study.
References
1. Umar SB, DiBaise JK. Protein-losing enteropathy: case illustrations and clinical review. Am J Gastroenterol. 2010;105:43-9.
2. Tsiodras S, Samonis G, Boumpas DT. et al. Fungal infections complicating tumor necrosis factor alpha blockade therapy. Mayo Clin Proc. 2008;83:181-94.
3. Kok J, Chen SC, Anderson L, et al. Protein-losing enteropathy and hypogammaglobulinaemia as first manifestations of disseminated histoplasmosis coincident with Nocardia infection. J Med Microbiol. 2010;59:610-3.
Erosive protein-losing enteropathy secondary to disseminated histoplasmosis
This patient was treated with amphotericin B and transitioned to oral itraconazole with frequent blood level monitoring to ensure absorption. His symptoms improved gradually. Small-bowel enteroscopy 3 weeks after presentation showed a normal duodenum and healing, superficial ulcers in the proximal jejunum (Figure F, G). Blood albumin levels had recovered to 3.1 g/dL (normal, 3.5–5.0 g/dL).
Protein-losing enteropathy (PLE) is a rare syndrome characterized by loss of serum proteins in the gastrointestinal (GI) tract, resulting in significant hypoproteinemia and consequent edema.1 PLE can also result in ascites, pleural and pericardial effusions, and, in prolonged cases, malnutrition. There are a variety of causes of PLE that can be broadly grouped into erosive GI disorders, disorders of increased GI mucosal permeability, and disorders of increased interstitial pressure. The clinical presentation depends on the underlying etiology, but commonly includes generalized edema owing to hypoproteinemia and resulting reduced oncotic pressure. GI symptoms are not frequently observed. The initial step in evaluating a patient with symptoms concerning for PLE is to rule out more common causes of hypoproteinemia, such as renal or hepatic disease, and malnutrition. To confirm enteric protein loss, alpha 1-antitrypsin clearance with a 24-hour stool collection is commonly and reliably used. Treatment of PLE is centered on treating the underlying cause while monitoring and treating malnutrition, including micronutrient deficiencies.
Fungal infections are a rare cause of PLE, but important to recognize as a potential complication of tumor necrosis factor–therapy, because these medications are commonly used for a variety of autoimmune diseases.2 Although histoplasmosis is an uncommon cause of GI inflammation, disseminated histoplasmosis causing PLE has been previously reported.3 In our patient, Histoplasma capsulatum infection caused diffuse GI ulcers, which allowed protein loss in the GI tract (erosive PLE). Antifungal treatment resulted in healing of intestinal ulcers and correction of hypoalbuminemia, thereby confirming the diagnosis of PLE and obviating the need for a confirmatory alpha 1-antitrypsin clearance study.
References
1. Umar SB, DiBaise JK. Protein-losing enteropathy: case illustrations and clinical review. Am J Gastroenterol. 2010;105:43-9.
2. Tsiodras S, Samonis G, Boumpas DT. et al. Fungal infections complicating tumor necrosis factor alpha blockade therapy. Mayo Clin Proc. 2008;83:181-94.
3. Kok J, Chen SC, Anderson L, et al. Protein-losing enteropathy and hypogammaglobulinaemia as first manifestations of disseminated histoplasmosis coincident with Nocardia infection. J Med Microbiol. 2010;59:610-3.
Erosive protein-losing enteropathy secondary to disseminated histoplasmosis
This patient was treated with amphotericin B and transitioned to oral itraconazole with frequent blood level monitoring to ensure absorption. His symptoms improved gradually. Small-bowel enteroscopy 3 weeks after presentation showed a normal duodenum and healing, superficial ulcers in the proximal jejunum (Figure F, G). Blood albumin levels had recovered to 3.1 g/dL (normal, 3.5–5.0 g/dL).
Protein-losing enteropathy (PLE) is a rare syndrome characterized by loss of serum proteins in the gastrointestinal (GI) tract, resulting in significant hypoproteinemia and consequent edema.1 PLE can also result in ascites, pleural and pericardial effusions, and, in prolonged cases, malnutrition. There are a variety of causes of PLE that can be broadly grouped into erosive GI disorders, disorders of increased GI mucosal permeability, and disorders of increased interstitial pressure. The clinical presentation depends on the underlying etiology, but commonly includes generalized edema owing to hypoproteinemia and resulting reduced oncotic pressure. GI symptoms are not frequently observed. The initial step in evaluating a patient with symptoms concerning for PLE is to rule out more common causes of hypoproteinemia, such as renal or hepatic disease, and malnutrition. To confirm enteric protein loss, alpha 1-antitrypsin clearance with a 24-hour stool collection is commonly and reliably used. Treatment of PLE is centered on treating the underlying cause while monitoring and treating malnutrition, including micronutrient deficiencies.
Fungal infections are a rare cause of PLE, but important to recognize as a potential complication of tumor necrosis factor–therapy, because these medications are commonly used for a variety of autoimmune diseases.2 Although histoplasmosis is an uncommon cause of GI inflammation, disseminated histoplasmosis causing PLE has been previously reported.3 In our patient, Histoplasma capsulatum infection caused diffuse GI ulcers, which allowed protein loss in the GI tract (erosive PLE). Antifungal treatment resulted in healing of intestinal ulcers and correction of hypoalbuminemia, thereby confirming the diagnosis of PLE and obviating the need for a confirmatory alpha 1-antitrypsin clearance study.
References
1. Umar SB, DiBaise JK. Protein-losing enteropathy: case illustrations and clinical review. Am J Gastroenterol. 2010;105:43-9.
2. Tsiodras S, Samonis G, Boumpas DT. et al. Fungal infections complicating tumor necrosis factor alpha blockade therapy. Mayo Clin Proc. 2008;83:181-94.
3. Kok J, Chen SC, Anderson L, et al. Protein-losing enteropathy and hypogammaglobulinaemia as first manifestations of disseminated histoplasmosis coincident with Nocardia infection. J Med Microbiol. 2010;59:610-3.
A 34-year-old man with a medical history of psoriasis, on adalimumab, presented with a 2-week history of progressively worsening abdominal pain, nausea, vomiting, melenic diarrhea, subjective fevers, and generalized weakness. One week into the illness, he developed progressive bilateral extremity and scrotal swelling.
His vital signs included a temperature of 36.8°C, heart rate of 104 beats per minute, respiratory rate of 18 breaths per minute, and a blood pressure of 114/71 mm Hg. The physical examination was notable for a well-nourished appearance, diffuse abdominal tenderness to palpation without distension, organomegaly, or rigidity, and pitting lower extremity edema.
Laboratory evaluation showed hemoglobin 10.3 g/dL (normal, 13.5–17.5 g/dL), leukocytes 10 × 109/L (normal, 3.5–10.5 × 109/L), platelets 212 × 109/L (normal, 150–450 × 109/L), sodium 131 mmol/L (normal, 135–145 mmol/L), creatinine 1 mg/dL (normal, 0.8–1.3 mg/dL), albumin 1.8 g/dL (normal, 3.5–5.0 g/dL), and C-reactive protein 53 mg/L (normal, less than 8 mg/L). Liver chemistries were all normal.
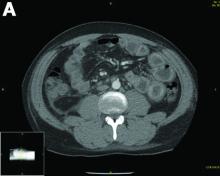
Urinalysis was unremarkable with normal urine protein levels. The enteric pathogen panel by polymerase chain reaction was negative. Computed tomography (CT) of the abdomen and pelvis showed marked circumferential wall thickening with mural enhancement of multiple loops of jejunum (Figure A).
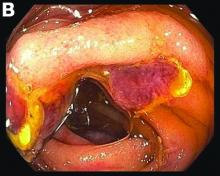
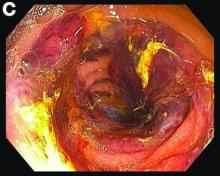
Small-bowel enteroscopy showed diffuse erosions in the entire duodenum and many oozing superficial ulcers with edematous and erythematous mucosa in the proximal jejunum (Figures B, C).
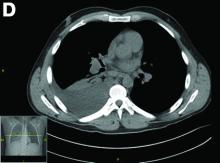
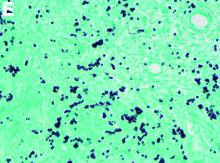
CT scan of the chest showed right lower lobe consolidation associated with a large right pleural effusion, and mediastinal, bilateral, hilar and abdominal lymphadenopathy (Figure D). Endobronchial ultrasound-guided transbronchial biopsy of lymph nodes was positive for oval-shaped organisms exhibiting narrow-based budding on GMS stain (Figure E).
Based on the clinical scenario and images, what is the most likely diagnosis?
Granuloma Annulare Presenting as Firm Nodules on the Forehead and Scalp in a Child
To the Editor:
A 3.5-year-old boy presented with asymptomatic subcutaneous nodules on the left side of the forehead and frontal scalp of approximately 6 months’ duration. There was no history of trauma or preceding rash. His medical history was remarkable only for allergic rhinitis. A review of systems was otherwise negative. Computed tomography ordered by the patient’s pediatrician prior to referral to dermatology showed soft tissue masses on the forehead and frontal scalp with no associated bone or brain parenchymal signal abnormalities.
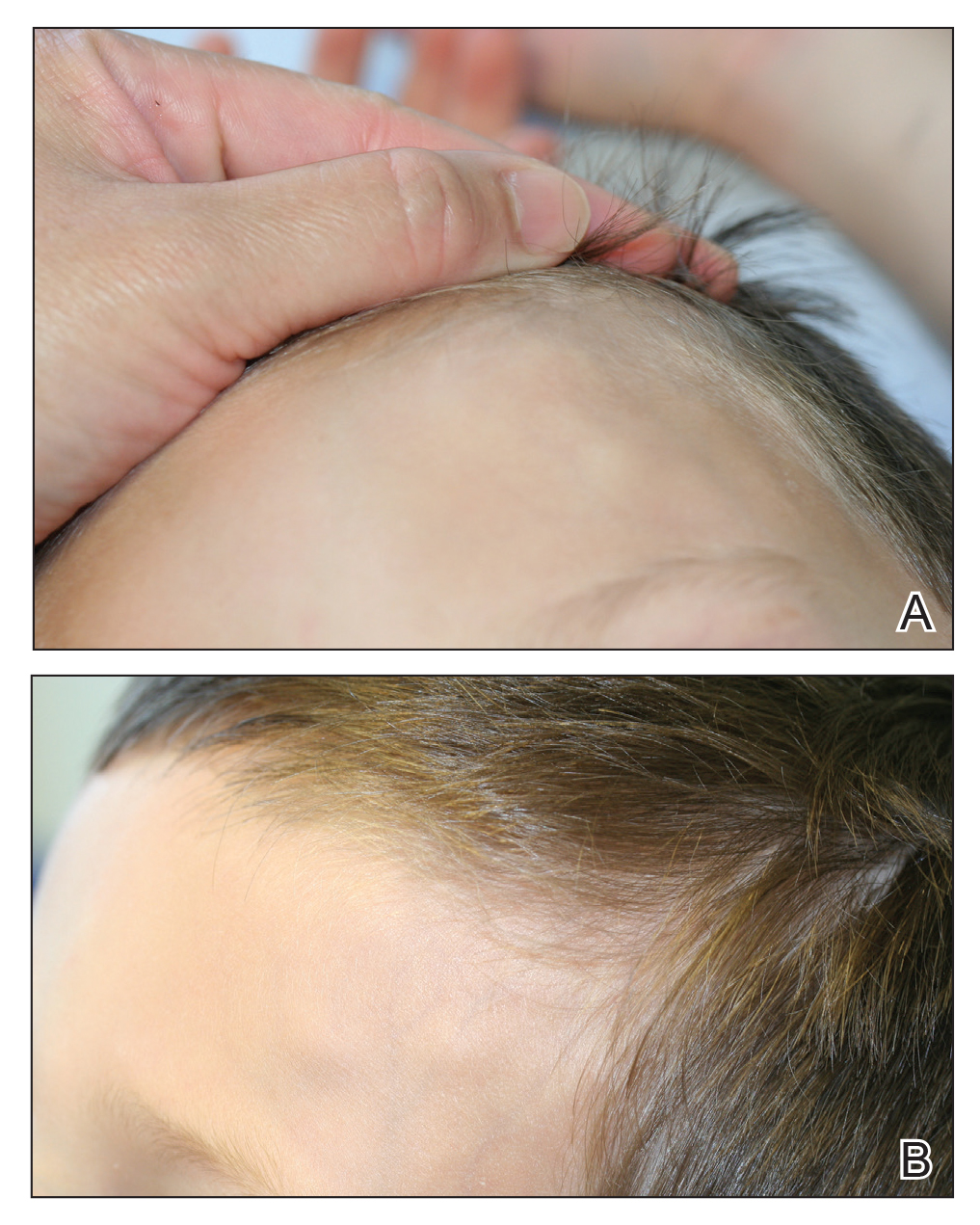
At presentation to dermatology, physical examination revealed a 6×7-cm, flesh-colored cluster of firm, nontender, fixed, subcutaneous nodules on the left superior forehead and anterior part of the left frontal scalp (Figure 1A), as well as 2×1.5-cm, firm, fixed, flesh-colored nodule inferior to the larger cluster of lesions (Figure 1B). The remainder of the skin examination was unremarkable.
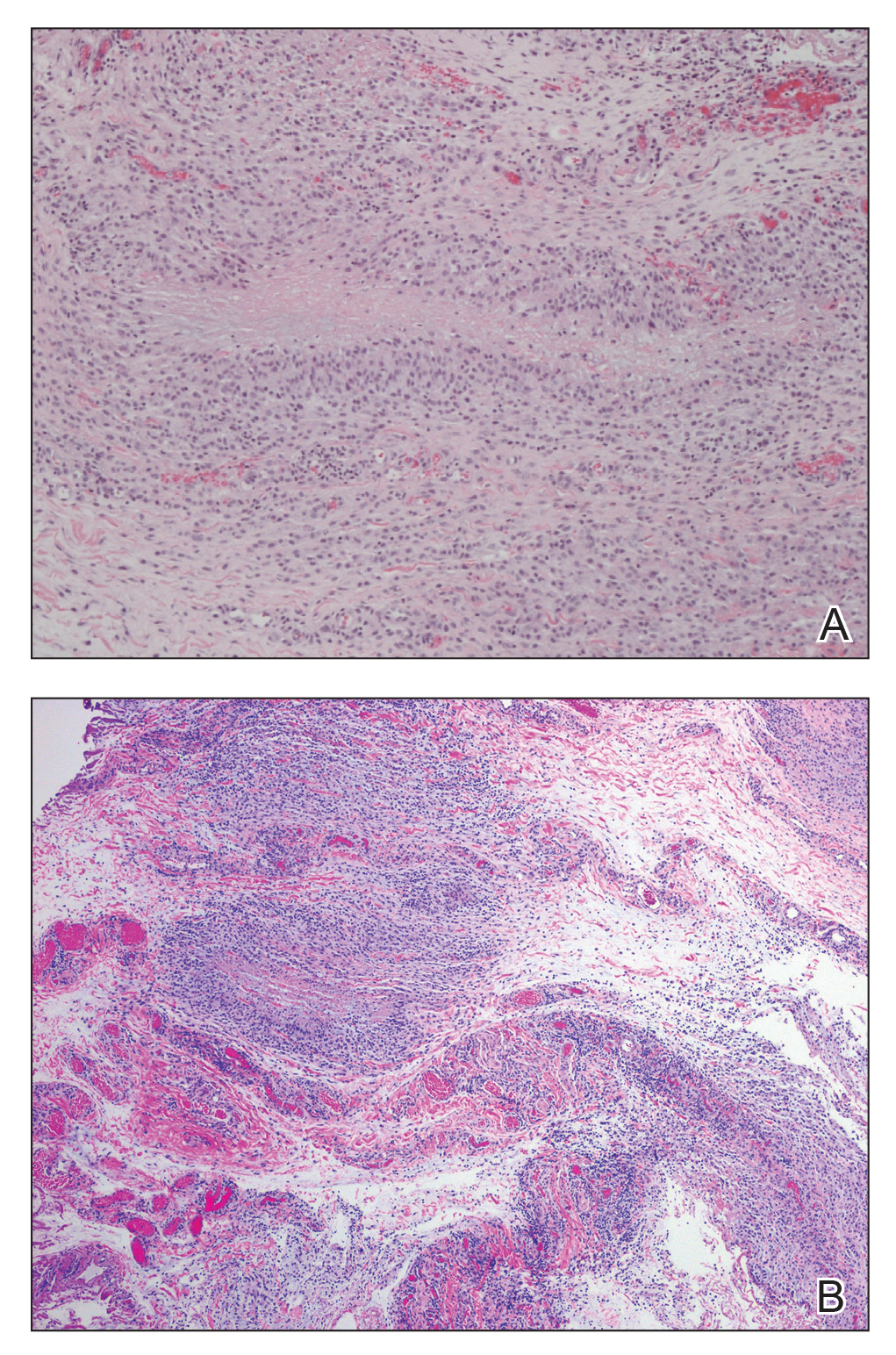
The patient was referred to plastic surgery for an incisional biopsy. The histopathologic findings demonstrated a marked mixed inflammatory infiltrate composed of lymphocytes and histiocytes with rare eosinophils and neutrophils in the subcutaneous tissue. The histiocytes were arranged in a palisading pattern surrounding central areas of necrosis (Figure 2). These features were consistent with a diagnosis of subcutaneous granuloma annulare (GA).
After the histologic diagnosis was elucidated, the patient’s family was provided reassurance regarding the benign nature and self-resolving course of GA in most children. No treatment was initiated, and no further laboratory studies or imaging were performed. At 9-month follow-up, the nodules were considerably smaller, and the patient remained asymptomatic.
Granuloma annulare is a benign dermatosis that presents in various forms, including localized, generalized, perforating, and subcutaneous subtypes. Subcutaneous GA (SGA) occurs most commonly in young children. The typical presentation of SGA is single or multiple flesh-colored to pink subcutaneous nodules on the arms, legs, or scalp.1 On the scalp, SGA has a predilection for the parietal and occipital regions. In some cases, there may be a history of preceding trauma to the area. Typically, lesions on the arms and legs are mobile whereas lesions on the scalp may be fixed due to their close proximity to the periosteum. Patients often are asymptomatic, and in the majority of cases, lesions resolve spontaneously over several months to years. Approximately 50% of cases resolve within 2 years of onset.2
Histologically, SGA appears as a nodule of fibrinoid or necrotic degeneration surrounded by palisaded histiocytes and lymphocytes in the deep dermis or subcutaneous tissue. Subcutaneous granuloma annulare is an accurate term for our case due to the subcutaneous location of the granulomatous change; however, some practitioners may prefer to use the term deep GA when the histologic findings are in the deep dermis vs the subcutis. Often, prominent deposition of mucin may be found. Histologically, SGA can closely resemble a rheumatoid nodule or necrobiosis lipoidica.1
Subcutaneous GA presenting on the scalp and forehead, such as in our case, can present a diagnostic challenge due to the extensive differential diagnoses that must be considered, including trauma, infection, bone or skin disease, and inflammatory or autoimmune disease.2,3 Additionally, the firm fixed characteristics of some lesions may raise additional concerns for possible malignant diagnoses such as epithelial sarcoma, peripheral nerve sheath tumor, rhabdomyosarcoma, or Langerhans cell histiocytosis, as highlighted by Agrawal et al.4 For these reasons, imaging and biopsy often are necessary for histologic diagnosis.
There is no consensus on the etiology of SGA, and no specific disease associations have been proven. Some case reports and case series have proposed a link between SGA and type 1 diabetes mellitus.1,4 In one retrospective case series, Grogg and Nascimento1 found that 2 of 34 (5.9%) pediatric patients with SGA had known or subsequently diagnosed diabetes mellitus; however, a definitive association between the 2 entities has not been elucidated. Underlying type 1 diabetes mellitus should be considered in patients with isolated SGA, but laboratory screening for diabetes is not necessary in patients with a negative review of systems.
Treatment of SGA typically is not required, as it is a self-limited condition. Often, once a histologic diagnosis is established, no further evaluation or treatment is warranted. Multiple treatment modalities have been reported, including intralesional and topical corticosteroids, laser therapy, cryotherapy, and systemic agents such as isotretinoin or corticosteroids; however, no treatment has been shown to be consistently efficacious.5 Excision of the lesions may be performed, but the risk for recurrence often precludes it as a viable option. In some case series, there have been recurrence rates as high as 40% to 75% in the months to years following surgical excision/biopsy.1,6
Patients presenting with presumed SGA should undergo a thorough history, review of systems, and full-body skin examination. In some cases, imaging and biopsy may be necessary to make a definitive diagnosis and exclude a more serious condition.
- Grogg KL, Nascimento AG. Subcutaneous granuloma annulare in childhood: clinicopathologic features in 34 cases. Pediatrics. 2001;107:E42.
- Sabuncuoglu H, Oge K, Soylemezoglu F, et al. Subcutaneous granuloma annulare of the scalp in childhood: a case report and review of the literature. Turk Neurosurg. 2007;17:19-22.
- Whelan JP, Zembowicz A. A 22-month-old boy with the rapid growth of subcutaneous nodules. N Engl J Med. 2006;354:2697-2704.
- Agrawal AK, Kammen BF, Guo H, et al. An unusual presentation of subcutaneous granuloma annulare in association with juvenile-onset diabetes: case report and literature review. Pediatr Dermatol. 2012;29:202-205.
- Cronquist SD, Stashower ME, Benson PM. Deep dermal granuloma annulare presenting as an eyelid tumor in a child, with review of pediatric eyelid lesions. Pediatr Dermatol. 1999;16:377-380.
- Jankowski PP, Krishna PH, Rutledge JC, et al. Surgical management and outcome of scalp subcutaneous granuloma annulare in children: case report. Neurosurgery. 2008;63:E1002, discussion E1002.
To the Editor:
A 3.5-year-old boy presented with asymptomatic subcutaneous nodules on the left side of the forehead and frontal scalp of approximately 6 months’ duration. There was no history of trauma or preceding rash. His medical history was remarkable only for allergic rhinitis. A review of systems was otherwise negative. Computed tomography ordered by the patient’s pediatrician prior to referral to dermatology showed soft tissue masses on the forehead and frontal scalp with no associated bone or brain parenchymal signal abnormalities.
At presentation to dermatology, physical examination revealed a 6×7-cm, flesh-colored cluster of firm, nontender, fixed, subcutaneous nodules on the left superior forehead and anterior part of the left frontal scalp (Figure 1A), as well as 2×1.5-cm, firm, fixed, flesh-colored nodule inferior to the larger cluster of lesions (Figure 1B). The remainder of the skin examination was unremarkable.

The patient was referred to plastic surgery for an incisional biopsy. The histopathologic findings demonstrated a marked mixed inflammatory infiltrate composed of lymphocytes and histiocytes with rare eosinophils and neutrophils in the subcutaneous tissue. The histiocytes were arranged in a palisading pattern surrounding central areas of necrosis (Figure 2). These features were consistent with a diagnosis of subcutaneous granuloma annulare (GA).

After the histologic diagnosis was elucidated, the patient’s family was provided reassurance regarding the benign nature and self-resolving course of GA in most children. No treatment was initiated, and no further laboratory studies or imaging were performed. At 9-month follow-up, the nodules were considerably smaller, and the patient remained asymptomatic.
Granuloma annulare is a benign dermatosis that presents in various forms, including localized, generalized, perforating, and subcutaneous subtypes. Subcutaneous GA (SGA) occurs most commonly in young children. The typical presentation of SGA is single or multiple flesh-colored to pink subcutaneous nodules on the arms, legs, or scalp.1 On the scalp, SGA has a predilection for the parietal and occipital regions. In some cases, there may be a history of preceding trauma to the area. Typically, lesions on the arms and legs are mobile whereas lesions on the scalp may be fixed due to their close proximity to the periosteum. Patients often are asymptomatic, and in the majority of cases, lesions resolve spontaneously over several months to years. Approximately 50% of cases resolve within 2 years of onset.2
Histologically, SGA appears as a nodule of fibrinoid or necrotic degeneration surrounded by palisaded histiocytes and lymphocytes in the deep dermis or subcutaneous tissue. Subcutaneous granuloma annulare is an accurate term for our case due to the subcutaneous location of the granulomatous change; however, some practitioners may prefer to use the term deep GA when the histologic findings are in the deep dermis vs the subcutis. Often, prominent deposition of mucin may be found. Histologically, SGA can closely resemble a rheumatoid nodule or necrobiosis lipoidica.1
Subcutaneous GA presenting on the scalp and forehead, such as in our case, can present a diagnostic challenge due to the extensive differential diagnoses that must be considered, including trauma, infection, bone or skin disease, and inflammatory or autoimmune disease.2,3 Additionally, the firm fixed characteristics of some lesions may raise additional concerns for possible malignant diagnoses such as epithelial sarcoma, peripheral nerve sheath tumor, rhabdomyosarcoma, or Langerhans cell histiocytosis, as highlighted by Agrawal et al.4 For these reasons, imaging and biopsy often are necessary for histologic diagnosis.
There is no consensus on the etiology of SGA, and no specific disease associations have been proven. Some case reports and case series have proposed a link between SGA and type 1 diabetes mellitus.1,4 In one retrospective case series, Grogg and Nascimento1 found that 2 of 34 (5.9%) pediatric patients with SGA had known or subsequently diagnosed diabetes mellitus; however, a definitive association between the 2 entities has not been elucidated. Underlying type 1 diabetes mellitus should be considered in patients with isolated SGA, but laboratory screening for diabetes is not necessary in patients with a negative review of systems.
Treatment of SGA typically is not required, as it is a self-limited condition. Often, once a histologic diagnosis is established, no further evaluation or treatment is warranted. Multiple treatment modalities have been reported, including intralesional and topical corticosteroids, laser therapy, cryotherapy, and systemic agents such as isotretinoin or corticosteroids; however, no treatment has been shown to be consistently efficacious.5 Excision of the lesions may be performed, but the risk for recurrence often precludes it as a viable option. In some case series, there have been recurrence rates as high as 40% to 75% in the months to years following surgical excision/biopsy.1,6
Patients presenting with presumed SGA should undergo a thorough history, review of systems, and full-body skin examination. In some cases, imaging and biopsy may be necessary to make a definitive diagnosis and exclude a more serious condition.
To the Editor:
A 3.5-year-old boy presented with asymptomatic subcutaneous nodules on the left side of the forehead and frontal scalp of approximately 6 months’ duration. There was no history of trauma or preceding rash. His medical history was remarkable only for allergic rhinitis. A review of systems was otherwise negative. Computed tomography ordered by the patient’s pediatrician prior to referral to dermatology showed soft tissue masses on the forehead and frontal scalp with no associated bone or brain parenchymal signal abnormalities.
At presentation to dermatology, physical examination revealed a 6×7-cm, flesh-colored cluster of firm, nontender, fixed, subcutaneous nodules on the left superior forehead and anterior part of the left frontal scalp (Figure 1A), as well as 2×1.5-cm, firm, fixed, flesh-colored nodule inferior to the larger cluster of lesions (Figure 1B). The remainder of the skin examination was unremarkable.

The patient was referred to plastic surgery for an incisional biopsy. The histopathologic findings demonstrated a marked mixed inflammatory infiltrate composed of lymphocytes and histiocytes with rare eosinophils and neutrophils in the subcutaneous tissue. The histiocytes were arranged in a palisading pattern surrounding central areas of necrosis (Figure 2). These features were consistent with a diagnosis of subcutaneous granuloma annulare (GA).

After the histologic diagnosis was elucidated, the patient’s family was provided reassurance regarding the benign nature and self-resolving course of GA in most children. No treatment was initiated, and no further laboratory studies or imaging were performed. At 9-month follow-up, the nodules were considerably smaller, and the patient remained asymptomatic.
Granuloma annulare is a benign dermatosis that presents in various forms, including localized, generalized, perforating, and subcutaneous subtypes. Subcutaneous GA (SGA) occurs most commonly in young children. The typical presentation of SGA is single or multiple flesh-colored to pink subcutaneous nodules on the arms, legs, or scalp.1 On the scalp, SGA has a predilection for the parietal and occipital regions. In some cases, there may be a history of preceding trauma to the area. Typically, lesions on the arms and legs are mobile whereas lesions on the scalp may be fixed due to their close proximity to the periosteum. Patients often are asymptomatic, and in the majority of cases, lesions resolve spontaneously over several months to years. Approximately 50% of cases resolve within 2 years of onset.2
Histologically, SGA appears as a nodule of fibrinoid or necrotic degeneration surrounded by palisaded histiocytes and lymphocytes in the deep dermis or subcutaneous tissue. Subcutaneous granuloma annulare is an accurate term for our case due to the subcutaneous location of the granulomatous change; however, some practitioners may prefer to use the term deep GA when the histologic findings are in the deep dermis vs the subcutis. Often, prominent deposition of mucin may be found. Histologically, SGA can closely resemble a rheumatoid nodule or necrobiosis lipoidica.1
Subcutaneous GA presenting on the scalp and forehead, such as in our case, can present a diagnostic challenge due to the extensive differential diagnoses that must be considered, including trauma, infection, bone or skin disease, and inflammatory or autoimmune disease.2,3 Additionally, the firm fixed characteristics of some lesions may raise additional concerns for possible malignant diagnoses such as epithelial sarcoma, peripheral nerve sheath tumor, rhabdomyosarcoma, or Langerhans cell histiocytosis, as highlighted by Agrawal et al.4 For these reasons, imaging and biopsy often are necessary for histologic diagnosis.
There is no consensus on the etiology of SGA, and no specific disease associations have been proven. Some case reports and case series have proposed a link between SGA and type 1 diabetes mellitus.1,4 In one retrospective case series, Grogg and Nascimento1 found that 2 of 34 (5.9%) pediatric patients with SGA had known or subsequently diagnosed diabetes mellitus; however, a definitive association between the 2 entities has not been elucidated. Underlying type 1 diabetes mellitus should be considered in patients with isolated SGA, but laboratory screening for diabetes is not necessary in patients with a negative review of systems.
Treatment of SGA typically is not required, as it is a self-limited condition. Often, once a histologic diagnosis is established, no further evaluation or treatment is warranted. Multiple treatment modalities have been reported, including intralesional and topical corticosteroids, laser therapy, cryotherapy, and systemic agents such as isotretinoin or corticosteroids; however, no treatment has been shown to be consistently efficacious.5 Excision of the lesions may be performed, but the risk for recurrence often precludes it as a viable option. In some case series, there have been recurrence rates as high as 40% to 75% in the months to years following surgical excision/biopsy.1,6
Patients presenting with presumed SGA should undergo a thorough history, review of systems, and full-body skin examination. In some cases, imaging and biopsy may be necessary to make a definitive diagnosis and exclude a more serious condition.
- Grogg KL, Nascimento AG. Subcutaneous granuloma annulare in childhood: clinicopathologic features in 34 cases. Pediatrics. 2001;107:E42.
- Sabuncuoglu H, Oge K, Soylemezoglu F, et al. Subcutaneous granuloma annulare of the scalp in childhood: a case report and review of the literature. Turk Neurosurg. 2007;17:19-22.
- Whelan JP, Zembowicz A. A 22-month-old boy with the rapid growth of subcutaneous nodules. N Engl J Med. 2006;354:2697-2704.
- Agrawal AK, Kammen BF, Guo H, et al. An unusual presentation of subcutaneous granuloma annulare in association with juvenile-onset diabetes: case report and literature review. Pediatr Dermatol. 2012;29:202-205.
- Cronquist SD, Stashower ME, Benson PM. Deep dermal granuloma annulare presenting as an eyelid tumor in a child, with review of pediatric eyelid lesions. Pediatr Dermatol. 1999;16:377-380.
- Jankowski PP, Krishna PH, Rutledge JC, et al. Surgical management and outcome of scalp subcutaneous granuloma annulare in children: case report. Neurosurgery. 2008;63:E1002, discussion E1002.
- Grogg KL, Nascimento AG. Subcutaneous granuloma annulare in childhood: clinicopathologic features in 34 cases. Pediatrics. 2001;107:E42.
- Sabuncuoglu H, Oge K, Soylemezoglu F, et al. Subcutaneous granuloma annulare of the scalp in childhood: a case report and review of the literature. Turk Neurosurg. 2007;17:19-22.
- Whelan JP, Zembowicz A. A 22-month-old boy with the rapid growth of subcutaneous nodules. N Engl J Med. 2006;354:2697-2704.
- Agrawal AK, Kammen BF, Guo H, et al. An unusual presentation of subcutaneous granuloma annulare in association with juvenile-onset diabetes: case report and literature review. Pediatr Dermatol. 2012;29:202-205.
- Cronquist SD, Stashower ME, Benson PM. Deep dermal granuloma annulare presenting as an eyelid tumor in a child, with review of pediatric eyelid lesions. Pediatr Dermatol. 1999;16:377-380.
- Jankowski PP, Krishna PH, Rutledge JC, et al. Surgical management and outcome of scalp subcutaneous granuloma annulare in children: case report. Neurosurgery. 2008;63:E1002, discussion E1002.
Practice Points
- Subcutaneous granuloma annulare (GA) is an important diagnosis to consider in the differential of subcutaneous nodules in children.
- The subcutaneous variant of GA can present without any typical GA lesions.
- Subcutaneous GA typically has a self-resolving course in most children.
Idiopathic Bilateral Auricular Ossificans
To the Editor:
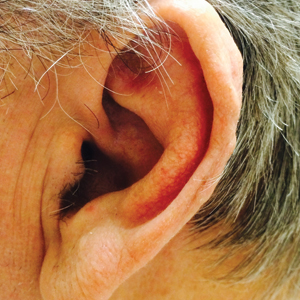
A 60-year-old man with a history of basal cell carcinoma, rosacea, and seborrheic dermatitis presented for a routine skin examination. The patient mentioned incidentally that both of his ears were “rock hard” and had been so for the last 10 to 20 years. He was not experiencing hearing abnormalities and denied any history of external ear trauma, frostbite injury of the ears, or history of endocrinopathy. Physical examination revealed normal-appearing skin on the bilateral ears (Figure 1), and palpation confirmed a bonelike consistency of the auricles with sparing of the earlobes. The differential diagnosis included osteoma cutis, ectopic calcification, tophaceous gout, localized scleroderma, and relapsing polychondritis. Incisional biopsy of the left posterior auricle to the level of cartilage revealed a histologically normal epidermis and dermis, with small fragments of cartilage calcification and lacunar bone formation (Figure 2). A radiograph of the skull showed faint calcification of the auricular cartilage, but no other osseous abnormalities were observed (Figure 3). Further laboratory workup including a comprehensive metabolic panel; complete blood cell count; and thyroid stimulating hormone, parathyroid hormone, and cortisol levels were normal. Based on these findings, a diagnosis of bilateral idiopathic auricular ossificans was made. Therapy was not pursued because the patient was asymptomatic, but referral to otolaryngology would have been considered if hearing impairment had occurred.

with auricular ossificans. Palpation of the auricle revealed a
bonelike consistency.
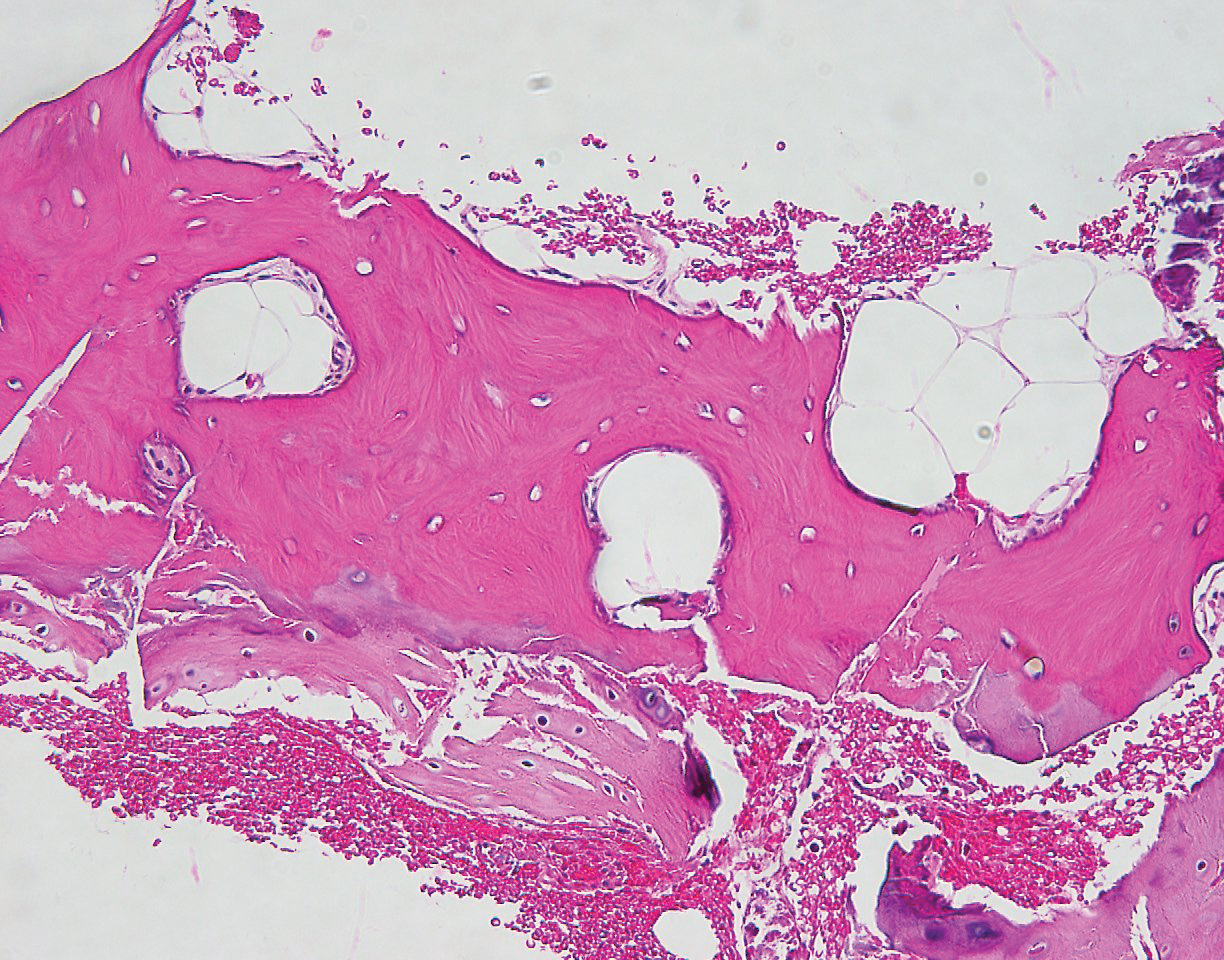
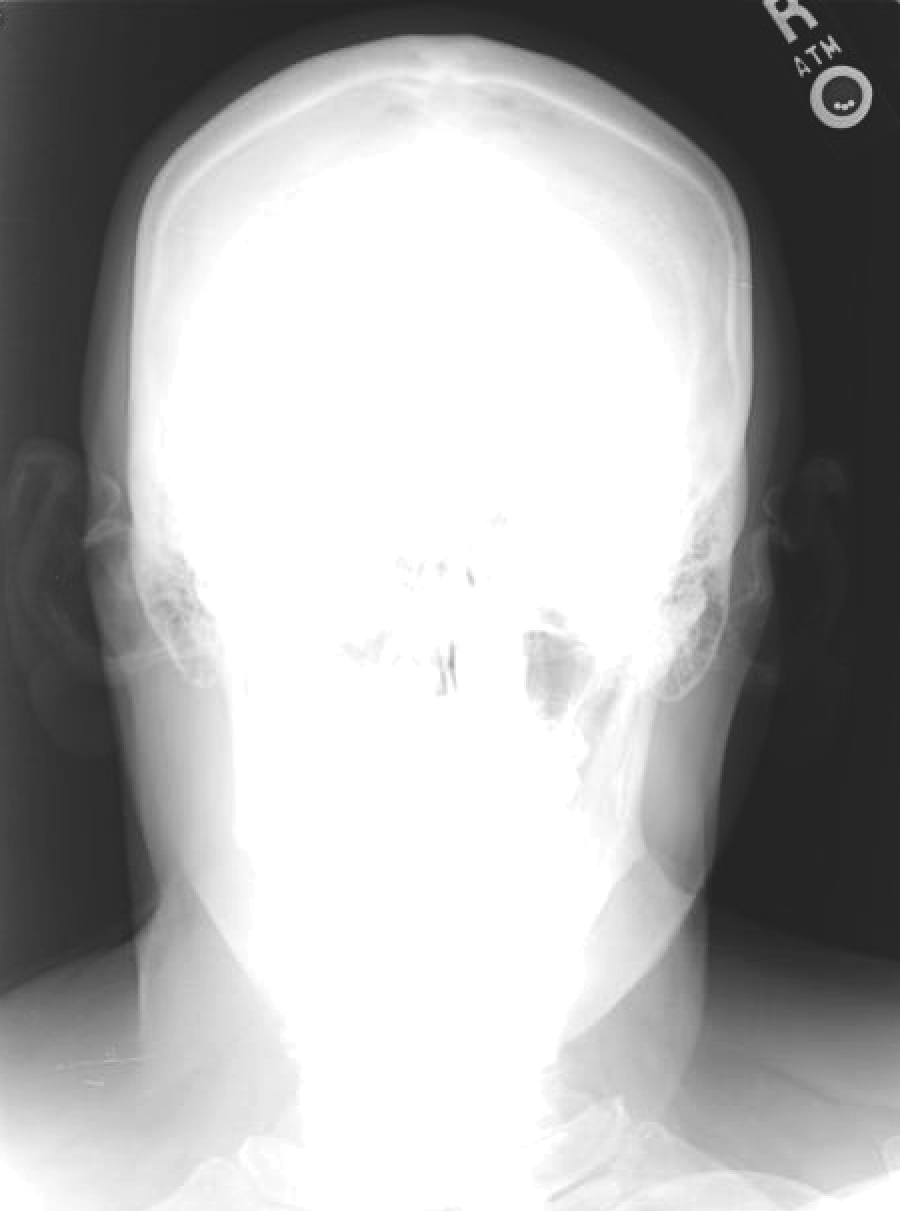
Auricular ossificans, which is characterized by the replacement of external ear cartilage by bone, is a rare condition with as few as 22 histologically proven cases documented in the literature.1,2 One case was reported in 2012 with consistent clinical and radiographic findings, but the patient declined biopsy.3 Similar to our patient, many pathologically documented cases have been determined to be idiopathic after workup, with identifiable triggering factors including cold injury, trauma, perichondritis, Addison disease, diabetes, and postpartum hypopituitarism.1 The male-to-female ratio is 18 to 5, the average age at diagnosis is 57 years, and as many as 70% of cases have demonstrated bilateral involvement.1,2
The majority of cases of auricular ossificans are asymptomatic at presentation with an insidious onset of the disease process over several years. Physical examination of the ear generally reveals a petrified auricle with sparing of the lobule and an otherwise normal clinical appearance. Radiographs demonstrate calcification, sometimes exactly mimicking the pattern seen in normal bone. Biopsy and histologic analysis show not only calcified cartilage but also actual lamellar bone formation.4 Depending on the precipitating factors, laboratory workup may uncover underlying metabolic abnormalities but often is unremarkable. Expert opinion generally recommends against extensive workup, which should be guided by the clinical presentation and the physician’s judgement.5 An insufficient number of patients with auricular ossificans have been definitively identified to provide clearer evidence-based recommendations.
Most patients present without pain or hearing abnormalities and do not require treatment. In one case, involvement of both the auricle and external ear canal resulted in intractable cerumen impaction that caused conductive hearing loss, eventually requiring resection of the ossified external ear cartilage and tragus.6 The most common reason for treatment has been discomfort impairing sleep, with surgical intervention including conchal reduction or wedge resection.5 A combined paucity of cases and poor understanding of the pathophysiologic mechanisms behind auricular ossificans limits current therapeutic options. Fortunately, this process appears to be benign in the majority of patients and generally represents a phenomenon that is much more interesting to the clinician than it is vexing to the patient.
- Calderon-Komaromy A, Cordoba S, Tardio JC, et al. Bilateral ossification of the auricular cartilage [published online November 28, 2014]. Actas Dermosifiliogr. 2015;106:433-435.
- Chang KH, Kim DK, Kim JH, et al. Idiopathic acquired ectopic auricular ossification: a case report and review of the literature. Ear Nose Throat J. 2011;90:424-427.
- Buikema KE, Adams EG. A rare case of petrified ear [published online October 15, 2012]. Case Rep Dermatol Med. 2012;2012:410601.
- Mastronikolis NS, Zampakis P, Kalogeropoulou C, et al. Bilateral ossification of the auricles: an unusual entity and review of the literature. Head Face Med. 2009;5:17.
- High WA, Larson MJ, Hoang MP. Idiopathic bilateral auricular ossificans: a case report and review of the literature. Arch Pathol Lab Med. 2004;128:1432-1434.
- Manni JJ, Berénos-Riley LC. Ossification of the external ear: a case report and review of the literature [published online June 18, 2005]. Eur Arch Otorhinolaryngol. 2005;262:961-964.
To the Editor:
A 60-year-old man with a history of basal cell carcinoma, rosacea, and seborrheic dermatitis presented for a routine skin examination. The patient mentioned incidentally that both of his ears were “rock hard” and had been so for the last 10 to 20 years. He was not experiencing hearing abnormalities and denied any history of external ear trauma, frostbite injury of the ears, or history of endocrinopathy. Physical examination revealed normal-appearing skin on the bilateral ears (Figure 1), and palpation confirmed a bonelike consistency of the auricles with sparing of the earlobes. The differential diagnosis included osteoma cutis, ectopic calcification, tophaceous gout, localized scleroderma, and relapsing polychondritis. Incisional biopsy of the left posterior auricle to the level of cartilage revealed a histologically normal epidermis and dermis, with small fragments of cartilage calcification and lacunar bone formation (Figure 2). A radiograph of the skull showed faint calcification of the auricular cartilage, but no other osseous abnormalities were observed (Figure 3). Further laboratory workup including a comprehensive metabolic panel; complete blood cell count; and thyroid stimulating hormone, parathyroid hormone, and cortisol levels were normal. Based on these findings, a diagnosis of bilateral idiopathic auricular ossificans was made. Therapy was not pursued because the patient was asymptomatic, but referral to otolaryngology would have been considered if hearing impairment had occurred.

with auricular ossificans. Palpation of the auricle revealed a
bonelike consistency.


Auricular ossificans, which is characterized by the replacement of external ear cartilage by bone, is a rare condition with as few as 22 histologically proven cases documented in the literature.1,2 One case was reported in 2012 with consistent clinical and radiographic findings, but the patient declined biopsy.3 Similar to our patient, many pathologically documented cases have been determined to be idiopathic after workup, with identifiable triggering factors including cold injury, trauma, perichondritis, Addison disease, diabetes, and postpartum hypopituitarism.1 The male-to-female ratio is 18 to 5, the average age at diagnosis is 57 years, and as many as 70% of cases have demonstrated bilateral involvement.1,2
The majority of cases of auricular ossificans are asymptomatic at presentation with an insidious onset of the disease process over several years. Physical examination of the ear generally reveals a petrified auricle with sparing of the lobule and an otherwise normal clinical appearance. Radiographs demonstrate calcification, sometimes exactly mimicking the pattern seen in normal bone. Biopsy and histologic analysis show not only calcified cartilage but also actual lamellar bone formation.4 Depending on the precipitating factors, laboratory workup may uncover underlying metabolic abnormalities but often is unremarkable. Expert opinion generally recommends against extensive workup, which should be guided by the clinical presentation and the physician’s judgement.5 An insufficient number of patients with auricular ossificans have been definitively identified to provide clearer evidence-based recommendations.
Most patients present without pain or hearing abnormalities and do not require treatment. In one case, involvement of both the auricle and external ear canal resulted in intractable cerumen impaction that caused conductive hearing loss, eventually requiring resection of the ossified external ear cartilage and tragus.6 The most common reason for treatment has been discomfort impairing sleep, with surgical intervention including conchal reduction or wedge resection.5 A combined paucity of cases and poor understanding of the pathophysiologic mechanisms behind auricular ossificans limits current therapeutic options. Fortunately, this process appears to be benign in the majority of patients and generally represents a phenomenon that is much more interesting to the clinician than it is vexing to the patient.
To the Editor:
A 60-year-old man with a history of basal cell carcinoma, rosacea, and seborrheic dermatitis presented for a routine skin examination. The patient mentioned incidentally that both of his ears were “rock hard” and had been so for the last 10 to 20 years. He was not experiencing hearing abnormalities and denied any history of external ear trauma, frostbite injury of the ears, or history of endocrinopathy. Physical examination revealed normal-appearing skin on the bilateral ears (Figure 1), and palpation confirmed a bonelike consistency of the auricles with sparing of the earlobes. The differential diagnosis included osteoma cutis, ectopic calcification, tophaceous gout, localized scleroderma, and relapsing polychondritis. Incisional biopsy of the left posterior auricle to the level of cartilage revealed a histologically normal epidermis and dermis, with small fragments of cartilage calcification and lacunar bone formation (Figure 2). A radiograph of the skull showed faint calcification of the auricular cartilage, but no other osseous abnormalities were observed (Figure 3). Further laboratory workup including a comprehensive metabolic panel; complete blood cell count; and thyroid stimulating hormone, parathyroid hormone, and cortisol levels were normal. Based on these findings, a diagnosis of bilateral idiopathic auricular ossificans was made. Therapy was not pursued because the patient was asymptomatic, but referral to otolaryngology would have been considered if hearing impairment had occurred.

with auricular ossificans. Palpation of the auricle revealed a
bonelike consistency.


Auricular ossificans, which is characterized by the replacement of external ear cartilage by bone, is a rare condition with as few as 22 histologically proven cases documented in the literature.1,2 One case was reported in 2012 with consistent clinical and radiographic findings, but the patient declined biopsy.3 Similar to our patient, many pathologically documented cases have been determined to be idiopathic after workup, with identifiable triggering factors including cold injury, trauma, perichondritis, Addison disease, diabetes, and postpartum hypopituitarism.1 The male-to-female ratio is 18 to 5, the average age at diagnosis is 57 years, and as many as 70% of cases have demonstrated bilateral involvement.1,2
The majority of cases of auricular ossificans are asymptomatic at presentation with an insidious onset of the disease process over several years. Physical examination of the ear generally reveals a petrified auricle with sparing of the lobule and an otherwise normal clinical appearance. Radiographs demonstrate calcification, sometimes exactly mimicking the pattern seen in normal bone. Biopsy and histologic analysis show not only calcified cartilage but also actual lamellar bone formation.4 Depending on the precipitating factors, laboratory workup may uncover underlying metabolic abnormalities but often is unremarkable. Expert opinion generally recommends against extensive workup, which should be guided by the clinical presentation and the physician’s judgement.5 An insufficient number of patients with auricular ossificans have been definitively identified to provide clearer evidence-based recommendations.
Most patients present without pain or hearing abnormalities and do not require treatment. In one case, involvement of both the auricle and external ear canal resulted in intractable cerumen impaction that caused conductive hearing loss, eventually requiring resection of the ossified external ear cartilage and tragus.6 The most common reason for treatment has been discomfort impairing sleep, with surgical intervention including conchal reduction or wedge resection.5 A combined paucity of cases and poor understanding of the pathophysiologic mechanisms behind auricular ossificans limits current therapeutic options. Fortunately, this process appears to be benign in the majority of patients and generally represents a phenomenon that is much more interesting to the clinician than it is vexing to the patient.
- Calderon-Komaromy A, Cordoba S, Tardio JC, et al. Bilateral ossification of the auricular cartilage [published online November 28, 2014]. Actas Dermosifiliogr. 2015;106:433-435.
- Chang KH, Kim DK, Kim JH, et al. Idiopathic acquired ectopic auricular ossification: a case report and review of the literature. Ear Nose Throat J. 2011;90:424-427.
- Buikema KE, Adams EG. A rare case of petrified ear [published online October 15, 2012]. Case Rep Dermatol Med. 2012;2012:410601.
- Mastronikolis NS, Zampakis P, Kalogeropoulou C, et al. Bilateral ossification of the auricles: an unusual entity and review of the literature. Head Face Med. 2009;5:17.
- High WA, Larson MJ, Hoang MP. Idiopathic bilateral auricular ossificans: a case report and review of the literature. Arch Pathol Lab Med. 2004;128:1432-1434.
- Manni JJ, Berénos-Riley LC. Ossification of the external ear: a case report and review of the literature [published online June 18, 2005]. Eur Arch Otorhinolaryngol. 2005;262:961-964.
- Calderon-Komaromy A, Cordoba S, Tardio JC, et al. Bilateral ossification of the auricular cartilage [published online November 28, 2014]. Actas Dermosifiliogr. 2015;106:433-435.
- Chang KH, Kim DK, Kim JH, et al. Idiopathic acquired ectopic auricular ossification: a case report and review of the literature. Ear Nose Throat J. 2011;90:424-427.
- Buikema KE, Adams EG. A rare case of petrified ear [published online October 15, 2012]. Case Rep Dermatol Med. 2012;2012:410601.
- Mastronikolis NS, Zampakis P, Kalogeropoulou C, et al. Bilateral ossification of the auricles: an unusual entity and review of the literature. Head Face Med. 2009;5:17.
- High WA, Larson MJ, Hoang MP. Idiopathic bilateral auricular ossificans: a case report and review of the literature. Arch Pathol Lab Med. 2004;128:1432-1434.
- Manni JJ, Berénos-Riley LC. Ossification of the external ear: a case report and review of the literature [published online June 18, 2005]. Eur Arch Otorhinolaryngol. 2005;262:961-964.
Practice Points
- Auricular ossificans is a rare condition characterized by the replacement of external ear cartilage by bone.
- The majority of cases are asymptomatic at presentation with an insidious onset of the disease process over several years.
- Most patients present without pain or hearing abnormalities and do not require treatment.
A Call to Address Sexual Harassment and Gender Discrimination in Medicine
PART I
Reports of sexual harassment and gender discrimination have dominated news headlines, and the #MeToo movement has brought the scope and severity of discriminatory behavior to the forefront of public consciousness. The #MeToo movement has raised national and global awareness of gender discrimination and sexual harassment in all industries and has given rise to Time’s Up initiative within health care.
Academic medicine has not been immune to workplace gender discrimination and sexual harassment as has been vastly reported in the literature and clearly documented in the 2018 National Academies of Sciences, Engineering, and Medicine report, which points out that … “the cumulative effect of sexual harassment is a significant and costly loss of talent in academic science, engineering, and medicine, which has consequences for advancing the nation’s economic and social well-being and its overall public health.”1
With the increasing recognition that healthcare is an environment especially prone to inequality, gender discrimination and sexual discrimination, the Time’s Up national organization, supported by the Time’s Up Legal Defense Fund, launched the Time’s Up initiative for health care workers on March 1, 2019.2,3 The overarching goal of this initiative is to expose workplace inequalities; drive policy and legislative changes focused on equal pay, equal opportunity, and equal work environments; and support safe, fair, and dignified work for women in health care. 2,3
This article, presented over the next three issues of Vascular Specialist, will present data on the ongoing problem of sexual harassment in medicine, discuss why the problem is prevalent in academic medicine, and provide recommendations for mitigating the problem in our workplace.
Defining & Measuring Sexual Harassment
Although commonly referred to as “sex discrimination,” sexual harassment differs from sexual discrimination. Sex discrimination refers to an employees’ denial of civil rights, raises, job opportunities, employment or a demotion or other mistreatments based on sex. On the other hand, sexual harassment relates to behavior that is inappropriate or offensive. A 2018 report from the National Academies Press defined sexual harassment (a form of discrimination) as comprising three categories of behavior: gender harassment – verbal and nonverbal behaviors that convey hostility, objectification, exclusion, or second-class status about members of one sex; unwanted sexual attention – verbal or physical unwelcome sexual advances, which can include assault; and sexual coercion – when favorable professional or educational treatment is conditional based on sexual activity.1
During 1995-2016, more than 7,000 health care service employees filed claims of sexual harassment with the Equal Employment Opportunity Commission. While this number may seem large, the number of official reports severely undervalues the prevalence of sexual discrimination in U.S. health care.1 Prevalence is best determined using representative validated surveys that rely on firsthand experience or observation of the behavior(s) without requiring the respondent to label those behaviors.
Environments at Risk for Sexual Harassment
Research reveals that academic settings in the fields of science exhibit characteristics that create high levels of risk for sexual harassment to occur. These environments historically are male dominated, tolerate sexually harassing behavior, and create a hierarchy in which men hold most of the positions of power and authority. Moreover, dependent relationships often exist between these gatekeepers and those subordinate to them, with gatekeepers directly influencing the career advancement of those subordinates.1
The greatest predictor of sexual harassment in the workplace is the organizational climate, which refers to the tolerance for sexual harassment and is measured on three elements: a lack of sanctions against offenders; a perceived risk to those who report sexually harassing behavior; and the perception that one’s report of sexually harassing behavior will not be taken seriously.1 Women are less likely to be directly harassed in environments that do not tolerate harassing behaviors or have a strong, clear, transparent consequence for these behaviors.
Sexual Harassment in Academic Medicine
Academic medicine has the highest rate of gender and sexual harassment in the health care industry, with about 50% of female academic physicians reporting incidents of sexual harassment.1 A recent survey suggests that more than half (58%) of women surgeons experienced sexual harassment within just the previous year alone.4 The conditions that increase the risk of sexual harassment against women – male-dominated hierarchical environments and organizational tolerance of sexual harassment – still prevail in academic medicine.
Higher-education environments are perceived as permissive environments in part because when targets report sexual harassment, they are retaliated against or there are few consequences for the perpetrator. Academic institutions are replete with cases in which the conduct of offenders is regarded as an open secret, but there are no sanctions for that bad behavior. These offenders often are perceived as superstars in their particular substantive area. Because they hold valued grants or national status within their specialty area, they often receive preferential treatment and are not held accountable for gender-biased and sexually harassing behavior. Interview data regarding sexual harassment in academic medicine reveals that interview respondents and other colleagues often know which individuals have a history of sexually harassing behavior. Both men and women warn colleagues of these perpetrators – knowing that calling out or reporting these behaviors is fruitless – and that the best manner for dealing with their behavior is to avoid or ignore it. This normalization of sexual harassment and gender bias was noted, unfortunately, to fuel similar behavior in new cohorts of medicine faculty.1
Sexual harassment of women in academic medicine starts in medical school. Female medical students are significantly more likely to experience sexual harassment by faculty and staff than are graduate or undergraduate students. Sexual harassment continues into residency training with residency described as “breeding grounds for abusive behavior by superiors.”1 Interview studies report that both men and women trainees widely accept harassing behavior at this stage of their training. The expectation of abusive and grueling conditions during residency caused several respondents to view sexual harassment as part of a continuum that they were expected to endure. Female residents in surgery and emergency medicine are more likely to be harassed than those in other specialties because of the high value placed on a hierarchical and authoritative workplace. Once out of residency, the sexual harassment of women in the workplace continues. A recent meta-analysis reveals that 58% of women faculty experience sexual harassment at work. Academic medicine has the second-highest rate of sexual harassment, behind the military (69%), as compared with all other workplaces. Women physicians of color experience more harassment (as a combination of sexual and racial harassment) than do white women physicians.1
Why Women Are Not Likely to Report Sexual Harassment
Only 25% of targets file formal reports with their employer, with even fewer taking claims to court. These numbers are even lower for women in the military and academic medicine, where formal reporting is the last resort for the victims. The reluctance to use formal reporting mechanisms is rooted in the “fear of blame, disbelief, inaction, retaliation, humiliation, ostracism, and the damage to one’s career and reputation.”1 Targets may perceive that there seem to be few benefits and high costs for reporting. Women and nonwhites often resist calling bad behavior “discrimination” because that increases their loss of control and victimhood.1 Women frequently perceive that grievance procedures favor the institution over the individual, and research has proven that women face retaliation, both professional and social, for speaking out. Furthermore, stark power differentials between the target and the perpetrator exacerbate the reluctance to report and the fear of retaliation. The overall effects can be long lasting.
References:
1. National Academies of Sciences, Engineering, and Medicine. Sexual Harassment of Women: Climate, Culture, and Consequences in Academic Sciences, Engineering, and Medicine. The National Academies Press, Washington, DC; 2018. doi. 10.17226/24994.
2. Choo EK et al. From #MeToo to #TimesUp in Health Care: Can a Culture of Accountability End Inequity and Harassment? Lancet. 2019 Feb 9;393(10171):499-502.
3. Choo EK et al. Time’s Up for Medicine? Only Time Will Tell. N Engl J Med. 2018 Oct 25;379(17):1592-3.
4. Medicine Has Its Own #MeToo Problems. Can Time’s Up Healthcare Fix It?
Dr. Mitchell is a vascular surgeon at Salem (Ore.) Hospital; Dr. Drudi is as vascular surgery resident at McGill University, Montreal; Dr. Brown is a professor of surgery at the Medical College of Wisconsin. Milwaukee; Dr. Sachdev-Ost is an associate professor of surgery at the University of Pittsburgh Medical Center.
PART I
Reports of sexual harassment and gender discrimination have dominated news headlines, and the #MeToo movement has brought the scope and severity of discriminatory behavior to the forefront of public consciousness. The #MeToo movement has raised national and global awareness of gender discrimination and sexual harassment in all industries and has given rise to Time’s Up initiative within health care.
Academic medicine has not been immune to workplace gender discrimination and sexual harassment as has been vastly reported in the literature and clearly documented in the 2018 National Academies of Sciences, Engineering, and Medicine report, which points out that … “the cumulative effect of sexual harassment is a significant and costly loss of talent in academic science, engineering, and medicine, which has consequences for advancing the nation’s economic and social well-being and its overall public health.”1
With the increasing recognition that healthcare is an environment especially prone to inequality, gender discrimination and sexual discrimination, the Time’s Up national organization, supported by the Time’s Up Legal Defense Fund, launched the Time’s Up initiative for health care workers on March 1, 2019.2,3 The overarching goal of this initiative is to expose workplace inequalities; drive policy and legislative changes focused on equal pay, equal opportunity, and equal work environments; and support safe, fair, and dignified work for women in health care. 2,3
This article, presented over the next three issues of Vascular Specialist, will present data on the ongoing problem of sexual harassment in medicine, discuss why the problem is prevalent in academic medicine, and provide recommendations for mitigating the problem in our workplace.
Defining & Measuring Sexual Harassment
Although commonly referred to as “sex discrimination,” sexual harassment differs from sexual discrimination. Sex discrimination refers to an employees’ denial of civil rights, raises, job opportunities, employment or a demotion or other mistreatments based on sex. On the other hand, sexual harassment relates to behavior that is inappropriate or offensive. A 2018 report from the National Academies Press defined sexual harassment (a form of discrimination) as comprising three categories of behavior: gender harassment – verbal and nonverbal behaviors that convey hostility, objectification, exclusion, or second-class status about members of one sex; unwanted sexual attention – verbal or physical unwelcome sexual advances, which can include assault; and sexual coercion – when favorable professional or educational treatment is conditional based on sexual activity.1
During 1995-2016, more than 7,000 health care service employees filed claims of sexual harassment with the Equal Employment Opportunity Commission. While this number may seem large, the number of official reports severely undervalues the prevalence of sexual discrimination in U.S. health care.1 Prevalence is best determined using representative validated surveys that rely on firsthand experience or observation of the behavior(s) without requiring the respondent to label those behaviors.
Environments at Risk for Sexual Harassment
Research reveals that academic settings in the fields of science exhibit characteristics that create high levels of risk for sexual harassment to occur. These environments historically are male dominated, tolerate sexually harassing behavior, and create a hierarchy in which men hold most of the positions of power and authority. Moreover, dependent relationships often exist between these gatekeepers and those subordinate to them, with gatekeepers directly influencing the career advancement of those subordinates.1
The greatest predictor of sexual harassment in the workplace is the organizational climate, which refers to the tolerance for sexual harassment and is measured on three elements: a lack of sanctions against offenders; a perceived risk to those who report sexually harassing behavior; and the perception that one’s report of sexually harassing behavior will not be taken seriously.1 Women are less likely to be directly harassed in environments that do not tolerate harassing behaviors or have a strong, clear, transparent consequence for these behaviors.
Sexual Harassment in Academic Medicine
Academic medicine has the highest rate of gender and sexual harassment in the health care industry, with about 50% of female academic physicians reporting incidents of sexual harassment.1 A recent survey suggests that more than half (58%) of women surgeons experienced sexual harassment within just the previous year alone.4 The conditions that increase the risk of sexual harassment against women – male-dominated hierarchical environments and organizational tolerance of sexual harassment – still prevail in academic medicine.
Higher-education environments are perceived as permissive environments in part because when targets report sexual harassment, they are retaliated against or there are few consequences for the perpetrator. Academic institutions are replete with cases in which the conduct of offenders is regarded as an open secret, but there are no sanctions for that bad behavior. These offenders often are perceived as superstars in their particular substantive area. Because they hold valued grants or national status within their specialty area, they often receive preferential treatment and are not held accountable for gender-biased and sexually harassing behavior. Interview data regarding sexual harassment in academic medicine reveals that interview respondents and other colleagues often know which individuals have a history of sexually harassing behavior. Both men and women warn colleagues of these perpetrators – knowing that calling out or reporting these behaviors is fruitless – and that the best manner for dealing with their behavior is to avoid or ignore it. This normalization of sexual harassment and gender bias was noted, unfortunately, to fuel similar behavior in new cohorts of medicine faculty.1
Sexual harassment of women in academic medicine starts in medical school. Female medical students are significantly more likely to experience sexual harassment by faculty and staff than are graduate or undergraduate students. Sexual harassment continues into residency training with residency described as “breeding grounds for abusive behavior by superiors.”1 Interview studies report that both men and women trainees widely accept harassing behavior at this stage of their training. The expectation of abusive and grueling conditions during residency caused several respondents to view sexual harassment as part of a continuum that they were expected to endure. Female residents in surgery and emergency medicine are more likely to be harassed than those in other specialties because of the high value placed on a hierarchical and authoritative workplace. Once out of residency, the sexual harassment of women in the workplace continues. A recent meta-analysis reveals that 58% of women faculty experience sexual harassment at work. Academic medicine has the second-highest rate of sexual harassment, behind the military (69%), as compared with all other workplaces. Women physicians of color experience more harassment (as a combination of sexual and racial harassment) than do white women physicians.1
Why Women Are Not Likely to Report Sexual Harassment
Only 25% of targets file formal reports with their employer, with even fewer taking claims to court. These numbers are even lower for women in the military and academic medicine, where formal reporting is the last resort for the victims. The reluctance to use formal reporting mechanisms is rooted in the “fear of blame, disbelief, inaction, retaliation, humiliation, ostracism, and the damage to one’s career and reputation.”1 Targets may perceive that there seem to be few benefits and high costs for reporting. Women and nonwhites often resist calling bad behavior “discrimination” because that increases their loss of control and victimhood.1 Women frequently perceive that grievance procedures favor the institution over the individual, and research has proven that women face retaliation, both professional and social, for speaking out. Furthermore, stark power differentials between the target and the perpetrator exacerbate the reluctance to report and the fear of retaliation. The overall effects can be long lasting.
References:
1. National Academies of Sciences, Engineering, and Medicine. Sexual Harassment of Women: Climate, Culture, and Consequences in Academic Sciences, Engineering, and Medicine. The National Academies Press, Washington, DC; 2018. doi. 10.17226/24994.
2. Choo EK et al. From #MeToo to #TimesUp in Health Care: Can a Culture of Accountability End Inequity and Harassment? Lancet. 2019 Feb 9;393(10171):499-502.
3. Choo EK et al. Time’s Up for Medicine? Only Time Will Tell. N Engl J Med. 2018 Oct 25;379(17):1592-3.
4. Medicine Has Its Own #MeToo Problems. Can Time’s Up Healthcare Fix It?
Dr. Mitchell is a vascular surgeon at Salem (Ore.) Hospital; Dr. Drudi is as vascular surgery resident at McGill University, Montreal; Dr. Brown is a professor of surgery at the Medical College of Wisconsin. Milwaukee; Dr. Sachdev-Ost is an associate professor of surgery at the University of Pittsburgh Medical Center.
PART I
Reports of sexual harassment and gender discrimination have dominated news headlines, and the #MeToo movement has brought the scope and severity of discriminatory behavior to the forefront of public consciousness. The #MeToo movement has raised national and global awareness of gender discrimination and sexual harassment in all industries and has given rise to Time’s Up initiative within health care.
Academic medicine has not been immune to workplace gender discrimination and sexual harassment as has been vastly reported in the literature and clearly documented in the 2018 National Academies of Sciences, Engineering, and Medicine report, which points out that … “the cumulative effect of sexual harassment is a significant and costly loss of talent in academic science, engineering, and medicine, which has consequences for advancing the nation’s economic and social well-being and its overall public health.”1
With the increasing recognition that healthcare is an environment especially prone to inequality, gender discrimination and sexual discrimination, the Time’s Up national organization, supported by the Time’s Up Legal Defense Fund, launched the Time’s Up initiative for health care workers on March 1, 2019.2,3 The overarching goal of this initiative is to expose workplace inequalities; drive policy and legislative changes focused on equal pay, equal opportunity, and equal work environments; and support safe, fair, and dignified work for women in health care. 2,3
This article, presented over the next three issues of Vascular Specialist, will present data on the ongoing problem of sexual harassment in medicine, discuss why the problem is prevalent in academic medicine, and provide recommendations for mitigating the problem in our workplace.
Defining & Measuring Sexual Harassment
Although commonly referred to as “sex discrimination,” sexual harassment differs from sexual discrimination. Sex discrimination refers to an employees’ denial of civil rights, raises, job opportunities, employment or a demotion or other mistreatments based on sex. On the other hand, sexual harassment relates to behavior that is inappropriate or offensive. A 2018 report from the National Academies Press defined sexual harassment (a form of discrimination) as comprising three categories of behavior: gender harassment – verbal and nonverbal behaviors that convey hostility, objectification, exclusion, or second-class status about members of one sex; unwanted sexual attention – verbal or physical unwelcome sexual advances, which can include assault; and sexual coercion – when favorable professional or educational treatment is conditional based on sexual activity.1
During 1995-2016, more than 7,000 health care service employees filed claims of sexual harassment with the Equal Employment Opportunity Commission. While this number may seem large, the number of official reports severely undervalues the prevalence of sexual discrimination in U.S. health care.1 Prevalence is best determined using representative validated surveys that rely on firsthand experience or observation of the behavior(s) without requiring the respondent to label those behaviors.
Environments at Risk for Sexual Harassment
Research reveals that academic settings in the fields of science exhibit characteristics that create high levels of risk for sexual harassment to occur. These environments historically are male dominated, tolerate sexually harassing behavior, and create a hierarchy in which men hold most of the positions of power and authority. Moreover, dependent relationships often exist between these gatekeepers and those subordinate to them, with gatekeepers directly influencing the career advancement of those subordinates.1
The greatest predictor of sexual harassment in the workplace is the organizational climate, which refers to the tolerance for sexual harassment and is measured on three elements: a lack of sanctions against offenders; a perceived risk to those who report sexually harassing behavior; and the perception that one’s report of sexually harassing behavior will not be taken seriously.1 Women are less likely to be directly harassed in environments that do not tolerate harassing behaviors or have a strong, clear, transparent consequence for these behaviors.
Sexual Harassment in Academic Medicine
Academic medicine has the highest rate of gender and sexual harassment in the health care industry, with about 50% of female academic physicians reporting incidents of sexual harassment.1 A recent survey suggests that more than half (58%) of women surgeons experienced sexual harassment within just the previous year alone.4 The conditions that increase the risk of sexual harassment against women – male-dominated hierarchical environments and organizational tolerance of sexual harassment – still prevail in academic medicine.
Higher-education environments are perceived as permissive environments in part because when targets report sexual harassment, they are retaliated against or there are few consequences for the perpetrator. Academic institutions are replete with cases in which the conduct of offenders is regarded as an open secret, but there are no sanctions for that bad behavior. These offenders often are perceived as superstars in their particular substantive area. Because they hold valued grants or national status within their specialty area, they often receive preferential treatment and are not held accountable for gender-biased and sexually harassing behavior. Interview data regarding sexual harassment in academic medicine reveals that interview respondents and other colleagues often know which individuals have a history of sexually harassing behavior. Both men and women warn colleagues of these perpetrators – knowing that calling out or reporting these behaviors is fruitless – and that the best manner for dealing with their behavior is to avoid or ignore it. This normalization of sexual harassment and gender bias was noted, unfortunately, to fuel similar behavior in new cohorts of medicine faculty.1
Sexual harassment of women in academic medicine starts in medical school. Female medical students are significantly more likely to experience sexual harassment by faculty and staff than are graduate or undergraduate students. Sexual harassment continues into residency training with residency described as “breeding grounds for abusive behavior by superiors.”1 Interview studies report that both men and women trainees widely accept harassing behavior at this stage of their training. The expectation of abusive and grueling conditions during residency caused several respondents to view sexual harassment as part of a continuum that they were expected to endure. Female residents in surgery and emergency medicine are more likely to be harassed than those in other specialties because of the high value placed on a hierarchical and authoritative workplace. Once out of residency, the sexual harassment of women in the workplace continues. A recent meta-analysis reveals that 58% of women faculty experience sexual harassment at work. Academic medicine has the second-highest rate of sexual harassment, behind the military (69%), as compared with all other workplaces. Women physicians of color experience more harassment (as a combination of sexual and racial harassment) than do white women physicians.1
Why Women Are Not Likely to Report Sexual Harassment
Only 25% of targets file formal reports with their employer, with even fewer taking claims to court. These numbers are even lower for women in the military and academic medicine, where formal reporting is the last resort for the victims. The reluctance to use formal reporting mechanisms is rooted in the “fear of blame, disbelief, inaction, retaliation, humiliation, ostracism, and the damage to one’s career and reputation.”1 Targets may perceive that there seem to be few benefits and high costs for reporting. Women and nonwhites often resist calling bad behavior “discrimination” because that increases their loss of control and victimhood.1 Women frequently perceive that grievance procedures favor the institution over the individual, and research has proven that women face retaliation, both professional and social, for speaking out. Furthermore, stark power differentials between the target and the perpetrator exacerbate the reluctance to report and the fear of retaliation. The overall effects can be long lasting.
References:
1. National Academies of Sciences, Engineering, and Medicine. Sexual Harassment of Women: Climate, Culture, and Consequences in Academic Sciences, Engineering, and Medicine. The National Academies Press, Washington, DC; 2018. doi. 10.17226/24994.
2. Choo EK et al. From #MeToo to #TimesUp in Health Care: Can a Culture of Accountability End Inequity and Harassment? Lancet. 2019 Feb 9;393(10171):499-502.
3. Choo EK et al. Time’s Up for Medicine? Only Time Will Tell. N Engl J Med. 2018 Oct 25;379(17):1592-3.
4. Medicine Has Its Own #MeToo Problems. Can Time’s Up Healthcare Fix It?
Dr. Mitchell is a vascular surgeon at Salem (Ore.) Hospital; Dr. Drudi is as vascular surgery resident at McGill University, Montreal; Dr. Brown is a professor of surgery at the Medical College of Wisconsin. Milwaukee; Dr. Sachdev-Ost is an associate professor of surgery at the University of Pittsburgh Medical Center.
Coordination of Care Between Primary Care and Oncology for Patients With Prostate Cancer (FULL)
The following is a lightly edited transcript of a teleconference recorded in July 2018. The teleconference brought together health care providers from the Greater Los Angeles VA Health Care System (GLAVAHCS) to discuss the real-world processes for managing the treatment of patients with prostate cancer as they move between primary and specialist care.
William J. Aronson, MD. We are fortunate in having a superb medical record system at the Department of Veterans Affairs (VA) where we can all communicate with each other through a number of methods. Let’s start our discussion by reviewing an index patient that we see in our practice who has been treated with either radical prostatectomy or radiation therapy. One question to address is: Is there a point when the Urology or Radiation Oncology service can transition the patient’s entire care back to the primary care team? And if so, what would be the optimal way to accomplish this?
Nick, is there some point at which you discharge the patient from the radiation oncology service and give specific directions to primary care, or is it primarily just back to urology in your case?
Nicholas G. Nickols, MD, PhD. I have not discharged any patient from my clinic after definitive prostate cancer treatment. During treatment, patients are seen every week. Subsequently, I see them 6 weeks posttreatment, and then every 4 months for the first year, then every 6 months for the next 4 years, and then yearly after that. Although I never formally discharged a patient from my clinic, you can see based on the frequency of visits, that the patient will see more often than their primary care provider (PCP) toward the beginning. And then, after some years, the patient sees their primary more than they me. So it’s not an immediate hand off but rather a gradual transition. It’s important that the PCP is aware of what to look for especially for the late recurrences, late potential side effects, probably more significantly than the early side effects, how to manage them when appropriate, and when to ask the patient to see our team more frequently in follow-up.
William Aronson. We have a number of patients who travel tremendous distances to see us, and I tend to think that many of our follow-up patients, once things are stabilized with regards to management of their side effects, really could see their primary care doctors if we can give them specific instructions on, for example, when to get a prostate-specific antigen (PSA) test and when to refer back to us.
Alison, can you think of some specific cases where you feel like we’ve successfully done that?
Alison Neymark, MS. For the most part we haven’t discharged people, either. What we have done is transitioned them over to a phone clinic. In our department, we have 4 nurse practitioners (NPs) who each have a half-day of phone clinic where they call patients with their test results. Some of those patients are prostate cancer patients that we have been following for years. We schedule them for a phone call, whether it’s every 3 months, every 6 months or every year, to review the updated PSA level and to just check in with them by phone. It’s a win-win because it’s a really quick phone call to reassure the veteran that the PSA level is being followed, and it frees up an in-person appointment slot for another veteran.
We still have patients that prefer face-to-face visits, even though they know we’re not doing anything except discussing a PSA level with them—they just want that security of seeing our face. Some patients are very nervous, and they don’t necessarily want to be discharged, so to speak, back to primary care. Also, for those patients that travel a long distance to clinic, we offer an appointment in the video chat clinic, with the community-based outpatient clinics in Bakersfield and Santa Maria, California.
PSA Levels
William Aronson. I probably see a patient about every 4 to 6 weeks who has a low PSA after about 10 years and has a long distance to travel and mobility and other problems that make it difficult to come in.
The challenge that I have is, what is that specific guideline to give with regards to the rise in PSA? I think it all depends on the patients prostate cancer clinical features and comorbidities.
Nicholas Nickols. If a patient has been seen by me in follow-up a number of times and there’s really no active issues and there’s a low suspicion of recurrence, then I offer the patient the option of a phone follow-up as an alternative to face to face. Some of them accept that, but I ask that they agree to also see either urology or their PCP face to face. I will also remotely ensure that they’re getting the right laboratory tests, and if not, I’ll put those orders in.
With regard to when to refer a patient back for a suspected recurrence after definitive radiation therapy, there is an accepted definition of biochemical failure called the Phoenix definition, which is an absolute rise in 2 ng/mL of PSA over their posttreatment nadir. Often the posttreatment nadir, especially if they were on hormone therapy, will be close to 0. If the PSA gets to 2, that is a good trigger for a referral back to me and/or urology to discuss restaging and workup for a suspected recurrence.
For patients that are postsurgery and then subsequently get salvage radiation, it is not as clear when a restaging workup should be initiated. Currently, the imaging that is routine care is not very sensitive for detecting PSA in that setting until the PSA is around 0.8 ng/mL, and that’s with the most modern imaging available. Over time that may improve.
William Aronson. The other index patient to think about would be the patient who is on watchful waiting for their prostate cancer, which is to be distinguished from active surveillance. If someone’s on active surveillance, we’re regularly doing prostate biopsies and doing very close monitoring; but we also have patients who have multiple other medical problems, have a limited life expectancy, don’t have aggressive prostate cancer, and it’s extremely reasonable not to do a biopsy in those patients.
Again, those are patients where we do follow the PSA generally every 6 months. And I think there’s also scenarios there where it’s reasonable to refer back to primary care with specific instructions. These, again, are patients who had difficulty getting in to see us or have mobility issues, but it is also a way to limit patient visits if that’s their desire.
Peter Glassman, MBBS, MSc: I’m trained as both a general internist and board certified in hospice and palliative medicine. I currently provide primary care as well as palliative care. I view prostate cancer from the diagnosis through the treatment spectrum as a continuum. It starts with the PCP with an elevated PSA level or if the digital rectal exam has an abnormality, and then the role of the genitourinary (GU) practitioner becomes more significant during the active treatment and diagnostic phases.
Primary care doesn’t disappear, and I think there are 2 major issues that go along with that. First of all, we in primary care, because we take care of patients that often have other comorbidities, need to work with the patient on those comorbidities. Secondly, we need the information shared between the GU and primary care providers so that we can answer questions from our patients and have an understanding of what they’re going through and when.
As time goes on, we go through various phases: We may reach a cure, a quiescent period, active therapy, watchful waiting, or recurrence. Primary care gets involved as time goes on when the disease either becomes quiescent, is just being followed, or is considered cured. Clearly when you have watchful waiting, active treatment, or are in a recurrence, then GU takes the forefront.
I view it as a wave function. Primary care to GU with primary in smaller letters and then primary, if you will, in larger letters, GU becomes a lesser participant unless there is active therapy, watchful waiting or recurrence.
In doing a little bit of research, I found 2 very good and very helpful documents. One is the American Cancer Society (ACS) prostate cancer survivorship care guidelines (Box). And the other is a synopsis of the guidelines. What I liked was that the guidelines focused not only on what should be done for the initial period of prostate cancer, but also for many of the ancillary issues which we often don’t give voice to. The guidelines provide a structure, a foundation to work with our patients over time on their prostate cancer-related issues while, at the same time, being cognizant that we need to deal with their other comorbid conditions.
Modes of Communication
Alison Neymark. We find that including parameters for PSA monitoring in our Progress Notes in the electronic health record (EHR) the best way to communicate with other providers. We’ll say, “If PSA gets to this level, please refer back.” We try to make it clear because with the VA being a training facility, it could be a different resident/attending physician team that’s going to see the patient the next time he is in primary care.
Peter Glassman. Yes, we’re very lucky, as Bill talked about earlier and Alison just mentioned. We have the EHR, and Bill may remember this. Before the EHR, we were constantly fishing to find the most relevant notes. If a patient saw a GU practitioner the day before they saw me, I was often asking the patient what was said. Now we can just review the notes.
It’s a double-edged sword though because there are, of course, many notes in a medical record; and you have to look for the specific items. The EHR and documenting the medical record probably plays the primary role in getting information across. When you want to have an active handoff, or you need to communicate with each other, we have a variety of mechanisms, ranging from the phone to the Microsoft Skype Link (Redmond, WA) system that allows us to tap a message to a colleague.
And I’ve been here long enough that I’ve seen most permutations of how prostate cancer is diagnosed as well as shared among providers. Bill and I have shared patients. Alison and I have shared patients, not necessarily with prostate cancer, although that too. But we know how to communicate with each other. And of course, there’s paging if you need something more urgently.
William Aronson. We also use Microsoft Outlook e-mail, and encrypt the messages to keep them confidential and private. The other nice thing we have is there is a nationwide urology Outlook e-mail, so if any of us have any specific questions, through one e-mail we can send it around the country; and there’s usually multiple very useful responses. That’s another real strength of our system within the VA that helps patient care enormously.
Nicholas Nickols. Sometimes, if there’s a critical note that I absolutely want someone on the care team to read, I’ll add them as a cosigner; and that will pop up when they log in to the Computerized Patient Record System (CPRS) as something that they need to read.
If the patient lives particularly far or gets his care at another VA medical center and laboratory tests are needed, then I will reach out to their PCP via e-mail. If contact is not confirmed, I will reach out via phone or Skype.
Peter Glassman. The most helpful notes are those that are very specific as to what primary care is being asked to do and/or what urology is going to be doing. So, the more specific we get in the notes as to what is being addressed, I think that’s very helpful.
I have been here long enough that I’ve known both Alison and Bill; and if they have an issue, they will tap me a message. It wasn’t long ago that Bill sent a message to me, and we worked on a patient with prostate cancer who was going to be on long-term hormone therapy. We talked about osteoporosis management, and between us we worked out who was going to do what. Those are the kind of shared decision-making situations that are very, very helpful.
Alison Neymark. Also, GLAVAHCS has a home-based primary care team (HBPC), and a lot of the PCPs for that team are NPs. They know that they can contact me for their patients because a lot of those patients are on watchful waiting, and we do not necessarily need to see them face to face in clinic. Our urology team just needs to review updated lab results and how they are doing clinically. The HBPC NP who knows them best can contact me every 6 months or so, and we’ll discuss the case, which avoids making the patient come in, especially when they’re homebound. Those of us that have been working at the VA for many years have established good relationships. We feel very comfortable reaching out and talking to each other about these patients
Peter Glassman. Alison, I agree. When I can talk to my patients and say, “You know, we had that question about,” whatever the question might be, “and I contacted urology, and this is what they said.” It gives the patient confidence that we’re following up on the issues that they have and that we’re communicating with each other in a way that is to their benefit. And I think it’s very appreciated both by the provider as well as the patient.
William Aronson. Not infrequently I’ll have patients who have nonurologic issues, which I may first detect, or who have specific issues with their prostate cancer that can be comanaged. And I have found that when I send an encrypted e-mail to the PCP, it has been an extremely satisfying interaction; and we really get to the heart of the matter quickly for the sake of the veteran.
Veterans With Comorbidities
William Aronson. Posttraumatic stress disorder (PTSD) is a very significant and unique aspect of our patients, which is enormously important to recognize. For example, the side effects of prostate treatments can be very significant, whether radiation or surgery. Our patients understandably can be very fearful of the prostate cancer diagnosis and treatment side effects.
We know, for example, after a patient gets a diagnosis of prostate cancer, they’re at increased risk of cardiac death. That’s an especially important issue for our patients that there be an ongoing interaction between urology and primary care.
The ACS guidelines that Dr. Glassman referred to were enlightening. In many cases, primary care can look at the whole patient and their circumstances better than we can and may detect, for example, specific psychological issues that either they can manage or refer to other specialists.
Peter Glassman. One of the things that was highlighted in the ACS guideline is that in any population of men who have this disease, there’s going to be distress, anxiety, and full-fledged depression. Of course, there are psychosocial aspects of prostate cancer, such as sexual activity and intimacy with a partner that we often don’t explore but are probably playing an important role in the overall health of our patients. We need to be mindful of these psychosocial aspects and at least periodically ask them, “How are you doing with this? How are things at home?” And of course, we already use screeners for depression. As the article noted, distress and anxiety and other factors can make somebody’s life less optimal with poorer quality of life.
Dual Care Patients
Alison Neymark. Many patients whether they have Medicare, insurance through their spouse, or Kaiser Permanente through their job, choose to go to both places. The challenge is communicating with the non-VA providers because here at the VA we can communicate easily through Skype, Outlook e-mail, or CPRS, but for dual care patients who’s in charge? I encourage the veterans to choose whom they want to manage their care; we’re always here and happy to treat them, but they need to decide who’s in charge because I don’t want them to get into a situation where the differing opinions lead to a delay in care.
Nicholas Nickols. The communication when the patient is receiving care outside VA, either on a continuous basis or temporarily, is more of a challenge. We obviously can’t rely upon the messaging system, face-to-face contact is difficult, and they may not be able to use e-mail as well. So in those situations, usually a phone call is the best approach. I have found that the outside providers are happy to speak on the phone to coordinate care.
Peter Glassman. I agree, it does add a layer of complexity because we don’t readily have the notes, any information in front of us. That said, a lot of our patients can and do bring in information from outside specialists, and I’m hopeful that they share the information that we provide back to their outside doctors as well.
William Aronson. Some patient get nervous. They might decide they want care elsewhere, but they still want the VA available for them. I always let them know they should proceed in whatever way they prefer, but we’re always available and here for them. I try to empower them to make their own decisions and feel comfortable with them.
Nicholas Nickols. Notes from the outside, if they’re being referred for VA Choice or community care, do get uploaded into VistA Imaging and can be accessed, although it’s not instantaneous. Sometimes there’s a delay, but I have been able to access outside notes most of the time. If a patient goes through a clinic at the VA, the note is written in real time, and you can read it immediately.
Peter Glassman. That is true for patients that are within the VA system who receive contracted care either through Choice or through non-VA care that is contracted through VA. For somebody who is choosing to use 2 health care systems, that can provide more of a challenge because those notes don’t come to us. Over time, most of my patients have brought test results to me.
The thing with oncologic care, of course, is it’s a lot more complex. And it’s hard to know without reasonable documentation what’s been going on. At some level, you have to trust that the outside provider is doing whatever they need to do, or you have to take it upon yourself to do it within the system.
Alison Neymark. In my experience with the Choice Program, it really depends on the outside providers and how comfortable they are with the system that has been established to share records. Not all providers are going into that system and accessing it. I have had cases where I will see the non-VA provider’s note and it’ll say, “No documentation available for this consultation.” It just happens that they didn’t go into the system to review it. So it can be a challenge.
I’ve had good communication with the providers who use the system correctly. In some cases, just to make it easier, I will go ahead and communicate with them through encrypted e-mail, or I’ll talk to their care coordinators directly by phone.
Peter Glassman. Many, if not most, PCPs are going to take care of these patients, certainly within the VA, with their GU colleagues. And most of us feel comfortable using the current documentation system in a way that allows us to share information or at least to gather information about these patients.
One of the things that I think came out for me in looking at this was that there are guidelines or there are ideas out there on how to take better care of these patients. And I for one learned a fair bit just by going through these documents, which I’m very appreciative of. But it does highlight to me that we can give good care and provide good shared care for prostate cancer survivors. I think that is something that perhaps this discussion will highlight that not only are people doing that, but there are resources they can utilize that will help them get a more comprehensive picture of taking care of prostate cancer survivors in the primary care clinic.
The beauty of the VA system as a system is that as these issues come up that might affect the overall health of the veteran with prostate cancer, for example, psychosocial issues, we have many people that can address this that are experts in their area. And one of the great beauties of having an all-encompassing healthcare system is being able to use resources within the system, whether that be for other medical problems or other social or other psychological issues, that we ourselves are not expert in. We can reach out to our other colleagues and ask them for assistance. We have that available to help the patients. It’s really holistic.
We even have integrated medicine where we can help patients, hopefully, get back into a healthy lifestyle, for example, whereas we may not have that expertise or knowledge. We often think of this as sort of a shared decision between GU and primary care. But, in fact, it’s really the responsibility of many, many people of the system at large. We are very lucky to have that.
The following is a lightly edited transcript of a teleconference recorded in July 2018. The teleconference brought together health care providers from the Greater Los Angeles VA Health Care System (GLAVAHCS) to discuss the real-world processes for managing the treatment of patients with prostate cancer as they move between primary and specialist care.
William J. Aronson, MD. We are fortunate in having a superb medical record system at the Department of Veterans Affairs (VA) where we can all communicate with each other through a number of methods. Let’s start our discussion by reviewing an index patient that we see in our practice who has been treated with either radical prostatectomy or radiation therapy. One question to address is: Is there a point when the Urology or Radiation Oncology service can transition the patient’s entire care back to the primary care team? And if so, what would be the optimal way to accomplish this?
Nick, is there some point at which you discharge the patient from the radiation oncology service and give specific directions to primary care, or is it primarily just back to urology in your case?
Nicholas G. Nickols, MD, PhD. I have not discharged any patient from my clinic after definitive prostate cancer treatment. During treatment, patients are seen every week. Subsequently, I see them 6 weeks posttreatment, and then every 4 months for the first year, then every 6 months for the next 4 years, and then yearly after that. Although I never formally discharged a patient from my clinic, you can see based on the frequency of visits, that the patient will see more often than their primary care provider (PCP) toward the beginning. And then, after some years, the patient sees their primary more than they me. So it’s not an immediate hand off but rather a gradual transition. It’s important that the PCP is aware of what to look for especially for the late recurrences, late potential side effects, probably more significantly than the early side effects, how to manage them when appropriate, and when to ask the patient to see our team more frequently in follow-up.
William Aronson. We have a number of patients who travel tremendous distances to see us, and I tend to think that many of our follow-up patients, once things are stabilized with regards to management of their side effects, really could see their primary care doctors if we can give them specific instructions on, for example, when to get a prostate-specific antigen (PSA) test and when to refer back to us.
Alison, can you think of some specific cases where you feel like we’ve successfully done that?
Alison Neymark, MS. For the most part we haven’t discharged people, either. What we have done is transitioned them over to a phone clinic. In our department, we have 4 nurse practitioners (NPs) who each have a half-day of phone clinic where they call patients with their test results. Some of those patients are prostate cancer patients that we have been following for years. We schedule them for a phone call, whether it’s every 3 months, every 6 months or every year, to review the updated PSA level and to just check in with them by phone. It’s a win-win because it’s a really quick phone call to reassure the veteran that the PSA level is being followed, and it frees up an in-person appointment slot for another veteran.
We still have patients that prefer face-to-face visits, even though they know we’re not doing anything except discussing a PSA level with them—they just want that security of seeing our face. Some patients are very nervous, and they don’t necessarily want to be discharged, so to speak, back to primary care. Also, for those patients that travel a long distance to clinic, we offer an appointment in the video chat clinic, with the community-based outpatient clinics in Bakersfield and Santa Maria, California.
PSA Levels
William Aronson. I probably see a patient about every 4 to 6 weeks who has a low PSA after about 10 years and has a long distance to travel and mobility and other problems that make it difficult to come in.
The challenge that I have is, what is that specific guideline to give with regards to the rise in PSA? I think it all depends on the patients prostate cancer clinical features and comorbidities.
Nicholas Nickols. If a patient has been seen by me in follow-up a number of times and there’s really no active issues and there’s a low suspicion of recurrence, then I offer the patient the option of a phone follow-up as an alternative to face to face. Some of them accept that, but I ask that they agree to also see either urology or their PCP face to face. I will also remotely ensure that they’re getting the right laboratory tests, and if not, I’ll put those orders in.
With regard to when to refer a patient back for a suspected recurrence after definitive radiation therapy, there is an accepted definition of biochemical failure called the Phoenix definition, which is an absolute rise in 2 ng/mL of PSA over their posttreatment nadir. Often the posttreatment nadir, especially if they were on hormone therapy, will be close to 0. If the PSA gets to 2, that is a good trigger for a referral back to me and/or urology to discuss restaging and workup for a suspected recurrence.
For patients that are postsurgery and then subsequently get salvage radiation, it is not as clear when a restaging workup should be initiated. Currently, the imaging that is routine care is not very sensitive for detecting PSA in that setting until the PSA is around 0.8 ng/mL, and that’s with the most modern imaging available. Over time that may improve.
William Aronson. The other index patient to think about would be the patient who is on watchful waiting for their prostate cancer, which is to be distinguished from active surveillance. If someone’s on active surveillance, we’re regularly doing prostate biopsies and doing very close monitoring; but we also have patients who have multiple other medical problems, have a limited life expectancy, don’t have aggressive prostate cancer, and it’s extremely reasonable not to do a biopsy in those patients.
Again, those are patients where we do follow the PSA generally every 6 months. And I think there’s also scenarios there where it’s reasonable to refer back to primary care with specific instructions. These, again, are patients who had difficulty getting in to see us or have mobility issues, but it is also a way to limit patient visits if that’s their desire.
Peter Glassman, MBBS, MSc: I’m trained as both a general internist and board certified in hospice and palliative medicine. I currently provide primary care as well as palliative care. I view prostate cancer from the diagnosis through the treatment spectrum as a continuum. It starts with the PCP with an elevated PSA level or if the digital rectal exam has an abnormality, and then the role of the genitourinary (GU) practitioner becomes more significant during the active treatment and diagnostic phases.
Primary care doesn’t disappear, and I think there are 2 major issues that go along with that. First of all, we in primary care, because we take care of patients that often have other comorbidities, need to work with the patient on those comorbidities. Secondly, we need the information shared between the GU and primary care providers so that we can answer questions from our patients and have an understanding of what they’re going through and when.
As time goes on, we go through various phases: We may reach a cure, a quiescent period, active therapy, watchful waiting, or recurrence. Primary care gets involved as time goes on when the disease either becomes quiescent, is just being followed, or is considered cured. Clearly when you have watchful waiting, active treatment, or are in a recurrence, then GU takes the forefront.
I view it as a wave function. Primary care to GU with primary in smaller letters and then primary, if you will, in larger letters, GU becomes a lesser participant unless there is active therapy, watchful waiting or recurrence.
In doing a little bit of research, I found 2 very good and very helpful documents. One is the American Cancer Society (ACS) prostate cancer survivorship care guidelines (Box). And the other is a synopsis of the guidelines. What I liked was that the guidelines focused not only on what should be done for the initial period of prostate cancer, but also for many of the ancillary issues which we often don’t give voice to. The guidelines provide a structure, a foundation to work with our patients over time on their prostate cancer-related issues while, at the same time, being cognizant that we need to deal with their other comorbid conditions.
Modes of Communication
Alison Neymark. We find that including parameters for PSA monitoring in our Progress Notes in the electronic health record (EHR) the best way to communicate with other providers. We’ll say, “If PSA gets to this level, please refer back.” We try to make it clear because with the VA being a training facility, it could be a different resident/attending physician team that’s going to see the patient the next time he is in primary care.
Peter Glassman. Yes, we’re very lucky, as Bill talked about earlier and Alison just mentioned. We have the EHR, and Bill may remember this. Before the EHR, we were constantly fishing to find the most relevant notes. If a patient saw a GU practitioner the day before they saw me, I was often asking the patient what was said. Now we can just review the notes.
It’s a double-edged sword though because there are, of course, many notes in a medical record; and you have to look for the specific items. The EHR and documenting the medical record probably plays the primary role in getting information across. When you want to have an active handoff, or you need to communicate with each other, we have a variety of mechanisms, ranging from the phone to the Microsoft Skype Link (Redmond, WA) system that allows us to tap a message to a colleague.
And I’ve been here long enough that I’ve seen most permutations of how prostate cancer is diagnosed as well as shared among providers. Bill and I have shared patients. Alison and I have shared patients, not necessarily with prostate cancer, although that too. But we know how to communicate with each other. And of course, there’s paging if you need something more urgently.
William Aronson. We also use Microsoft Outlook e-mail, and encrypt the messages to keep them confidential and private. The other nice thing we have is there is a nationwide urology Outlook e-mail, so if any of us have any specific questions, through one e-mail we can send it around the country; and there’s usually multiple very useful responses. That’s another real strength of our system within the VA that helps patient care enormously.
Nicholas Nickols. Sometimes, if there’s a critical note that I absolutely want someone on the care team to read, I’ll add them as a cosigner; and that will pop up when they log in to the Computerized Patient Record System (CPRS) as something that they need to read.
If the patient lives particularly far or gets his care at another VA medical center and laboratory tests are needed, then I will reach out to their PCP via e-mail. If contact is not confirmed, I will reach out via phone or Skype.
Peter Glassman. The most helpful notes are those that are very specific as to what primary care is being asked to do and/or what urology is going to be doing. So, the more specific we get in the notes as to what is being addressed, I think that’s very helpful.
I have been here long enough that I’ve known both Alison and Bill; and if they have an issue, they will tap me a message. It wasn’t long ago that Bill sent a message to me, and we worked on a patient with prostate cancer who was going to be on long-term hormone therapy. We talked about osteoporosis management, and between us we worked out who was going to do what. Those are the kind of shared decision-making situations that are very, very helpful.
Alison Neymark. Also, GLAVAHCS has a home-based primary care team (HBPC), and a lot of the PCPs for that team are NPs. They know that they can contact me for their patients because a lot of those patients are on watchful waiting, and we do not necessarily need to see them face to face in clinic. Our urology team just needs to review updated lab results and how they are doing clinically. The HBPC NP who knows them best can contact me every 6 months or so, and we’ll discuss the case, which avoids making the patient come in, especially when they’re homebound. Those of us that have been working at the VA for many years have established good relationships. We feel very comfortable reaching out and talking to each other about these patients
Peter Glassman. Alison, I agree. When I can talk to my patients and say, “You know, we had that question about,” whatever the question might be, “and I contacted urology, and this is what they said.” It gives the patient confidence that we’re following up on the issues that they have and that we’re communicating with each other in a way that is to their benefit. And I think it’s very appreciated both by the provider as well as the patient.
William Aronson. Not infrequently I’ll have patients who have nonurologic issues, which I may first detect, or who have specific issues with their prostate cancer that can be comanaged. And I have found that when I send an encrypted e-mail to the PCP, it has been an extremely satisfying interaction; and we really get to the heart of the matter quickly for the sake of the veteran.
Veterans With Comorbidities
William Aronson. Posttraumatic stress disorder (PTSD) is a very significant and unique aspect of our patients, which is enormously important to recognize. For example, the side effects of prostate treatments can be very significant, whether radiation or surgery. Our patients understandably can be very fearful of the prostate cancer diagnosis and treatment side effects.
We know, for example, after a patient gets a diagnosis of prostate cancer, they’re at increased risk of cardiac death. That’s an especially important issue for our patients that there be an ongoing interaction between urology and primary care.
The ACS guidelines that Dr. Glassman referred to were enlightening. In many cases, primary care can look at the whole patient and their circumstances better than we can and may detect, for example, specific psychological issues that either they can manage or refer to other specialists.
Peter Glassman. One of the things that was highlighted in the ACS guideline is that in any population of men who have this disease, there’s going to be distress, anxiety, and full-fledged depression. Of course, there are psychosocial aspects of prostate cancer, such as sexual activity and intimacy with a partner that we often don’t explore but are probably playing an important role in the overall health of our patients. We need to be mindful of these psychosocial aspects and at least periodically ask them, “How are you doing with this? How are things at home?” And of course, we already use screeners for depression. As the article noted, distress and anxiety and other factors can make somebody’s life less optimal with poorer quality of life.
Dual Care Patients
Alison Neymark. Many patients whether they have Medicare, insurance through their spouse, or Kaiser Permanente through their job, choose to go to both places. The challenge is communicating with the non-VA providers because here at the VA we can communicate easily through Skype, Outlook e-mail, or CPRS, but for dual care patients who’s in charge? I encourage the veterans to choose whom they want to manage their care; we’re always here and happy to treat them, but they need to decide who’s in charge because I don’t want them to get into a situation where the differing opinions lead to a delay in care.
Nicholas Nickols. The communication when the patient is receiving care outside VA, either on a continuous basis or temporarily, is more of a challenge. We obviously can’t rely upon the messaging system, face-to-face contact is difficult, and they may not be able to use e-mail as well. So in those situations, usually a phone call is the best approach. I have found that the outside providers are happy to speak on the phone to coordinate care.
Peter Glassman. I agree, it does add a layer of complexity because we don’t readily have the notes, any information in front of us. That said, a lot of our patients can and do bring in information from outside specialists, and I’m hopeful that they share the information that we provide back to their outside doctors as well.
William Aronson. Some patient get nervous. They might decide they want care elsewhere, but they still want the VA available for them. I always let them know they should proceed in whatever way they prefer, but we’re always available and here for them. I try to empower them to make their own decisions and feel comfortable with them.
Nicholas Nickols. Notes from the outside, if they’re being referred for VA Choice or community care, do get uploaded into VistA Imaging and can be accessed, although it’s not instantaneous. Sometimes there’s a delay, but I have been able to access outside notes most of the time. If a patient goes through a clinic at the VA, the note is written in real time, and you can read it immediately.
Peter Glassman. That is true for patients that are within the VA system who receive contracted care either through Choice or through non-VA care that is contracted through VA. For somebody who is choosing to use 2 health care systems, that can provide more of a challenge because those notes don’t come to us. Over time, most of my patients have brought test results to me.
The thing with oncologic care, of course, is it’s a lot more complex. And it’s hard to know without reasonable documentation what’s been going on. At some level, you have to trust that the outside provider is doing whatever they need to do, or you have to take it upon yourself to do it within the system.
Alison Neymark. In my experience with the Choice Program, it really depends on the outside providers and how comfortable they are with the system that has been established to share records. Not all providers are going into that system and accessing it. I have had cases where I will see the non-VA provider’s note and it’ll say, “No documentation available for this consultation.” It just happens that they didn’t go into the system to review it. So it can be a challenge.
I’ve had good communication with the providers who use the system correctly. In some cases, just to make it easier, I will go ahead and communicate with them through encrypted e-mail, or I’ll talk to their care coordinators directly by phone.
Peter Glassman. Many, if not most, PCPs are going to take care of these patients, certainly within the VA, with their GU colleagues. And most of us feel comfortable using the current documentation system in a way that allows us to share information or at least to gather information about these patients.
One of the things that I think came out for me in looking at this was that there are guidelines or there are ideas out there on how to take better care of these patients. And I for one learned a fair bit just by going through these documents, which I’m very appreciative of. But it does highlight to me that we can give good care and provide good shared care for prostate cancer survivors. I think that is something that perhaps this discussion will highlight that not only are people doing that, but there are resources they can utilize that will help them get a more comprehensive picture of taking care of prostate cancer survivors in the primary care clinic.
The beauty of the VA system as a system is that as these issues come up that might affect the overall health of the veteran with prostate cancer, for example, psychosocial issues, we have many people that can address this that are experts in their area. And one of the great beauties of having an all-encompassing healthcare system is being able to use resources within the system, whether that be for other medical problems or other social or other psychological issues, that we ourselves are not expert in. We can reach out to our other colleagues and ask them for assistance. We have that available to help the patients. It’s really holistic.
We even have integrated medicine where we can help patients, hopefully, get back into a healthy lifestyle, for example, whereas we may not have that expertise or knowledge. We often think of this as sort of a shared decision between GU and primary care. But, in fact, it’s really the responsibility of many, many people of the system at large. We are very lucky to have that.
The following is a lightly edited transcript of a teleconference recorded in July 2018. The teleconference brought together health care providers from the Greater Los Angeles VA Health Care System (GLAVAHCS) to discuss the real-world processes for managing the treatment of patients with prostate cancer as they move between primary and specialist care.
William J. Aronson, MD. We are fortunate in having a superb medical record system at the Department of Veterans Affairs (VA) where we can all communicate with each other through a number of methods. Let’s start our discussion by reviewing an index patient that we see in our practice who has been treated with either radical prostatectomy or radiation therapy. One question to address is: Is there a point when the Urology or Radiation Oncology service can transition the patient’s entire care back to the primary care team? And if so, what would be the optimal way to accomplish this?
Nick, is there some point at which you discharge the patient from the radiation oncology service and give specific directions to primary care, or is it primarily just back to urology in your case?
Nicholas G. Nickols, MD, PhD. I have not discharged any patient from my clinic after definitive prostate cancer treatment. During treatment, patients are seen every week. Subsequently, I see them 6 weeks posttreatment, and then every 4 months for the first year, then every 6 months for the next 4 years, and then yearly after that. Although I never formally discharged a patient from my clinic, you can see based on the frequency of visits, that the patient will see more often than their primary care provider (PCP) toward the beginning. And then, after some years, the patient sees their primary more than they me. So it’s not an immediate hand off but rather a gradual transition. It’s important that the PCP is aware of what to look for especially for the late recurrences, late potential side effects, probably more significantly than the early side effects, how to manage them when appropriate, and when to ask the patient to see our team more frequently in follow-up.
William Aronson. We have a number of patients who travel tremendous distances to see us, and I tend to think that many of our follow-up patients, once things are stabilized with regards to management of their side effects, really could see their primary care doctors if we can give them specific instructions on, for example, when to get a prostate-specific antigen (PSA) test and when to refer back to us.
Alison, can you think of some specific cases where you feel like we’ve successfully done that?
Alison Neymark, MS. For the most part we haven’t discharged people, either. What we have done is transitioned them over to a phone clinic. In our department, we have 4 nurse practitioners (NPs) who each have a half-day of phone clinic where they call patients with their test results. Some of those patients are prostate cancer patients that we have been following for years. We schedule them for a phone call, whether it’s every 3 months, every 6 months or every year, to review the updated PSA level and to just check in with them by phone. It’s a win-win because it’s a really quick phone call to reassure the veteran that the PSA level is being followed, and it frees up an in-person appointment slot for another veteran.
We still have patients that prefer face-to-face visits, even though they know we’re not doing anything except discussing a PSA level with them—they just want that security of seeing our face. Some patients are very nervous, and they don’t necessarily want to be discharged, so to speak, back to primary care. Also, for those patients that travel a long distance to clinic, we offer an appointment in the video chat clinic, with the community-based outpatient clinics in Bakersfield and Santa Maria, California.
PSA Levels
William Aronson. I probably see a patient about every 4 to 6 weeks who has a low PSA after about 10 years and has a long distance to travel and mobility and other problems that make it difficult to come in.
The challenge that I have is, what is that specific guideline to give with regards to the rise in PSA? I think it all depends on the patients prostate cancer clinical features and comorbidities.
Nicholas Nickols. If a patient has been seen by me in follow-up a number of times and there’s really no active issues and there’s a low suspicion of recurrence, then I offer the patient the option of a phone follow-up as an alternative to face to face. Some of them accept that, but I ask that they agree to also see either urology or their PCP face to face. I will also remotely ensure that they’re getting the right laboratory tests, and if not, I’ll put those orders in.
With regard to when to refer a patient back for a suspected recurrence after definitive radiation therapy, there is an accepted definition of biochemical failure called the Phoenix definition, which is an absolute rise in 2 ng/mL of PSA over their posttreatment nadir. Often the posttreatment nadir, especially if they were on hormone therapy, will be close to 0. If the PSA gets to 2, that is a good trigger for a referral back to me and/or urology to discuss restaging and workup for a suspected recurrence.
For patients that are postsurgery and then subsequently get salvage radiation, it is not as clear when a restaging workup should be initiated. Currently, the imaging that is routine care is not very sensitive for detecting PSA in that setting until the PSA is around 0.8 ng/mL, and that’s with the most modern imaging available. Over time that may improve.
William Aronson. The other index patient to think about would be the patient who is on watchful waiting for their prostate cancer, which is to be distinguished from active surveillance. If someone’s on active surveillance, we’re regularly doing prostate biopsies and doing very close monitoring; but we also have patients who have multiple other medical problems, have a limited life expectancy, don’t have aggressive prostate cancer, and it’s extremely reasonable not to do a biopsy in those patients.
Again, those are patients where we do follow the PSA generally every 6 months. And I think there’s also scenarios there where it’s reasonable to refer back to primary care with specific instructions. These, again, are patients who had difficulty getting in to see us or have mobility issues, but it is also a way to limit patient visits if that’s their desire.
Peter Glassman, MBBS, MSc: I’m trained as both a general internist and board certified in hospice and palliative medicine. I currently provide primary care as well as palliative care. I view prostate cancer from the diagnosis through the treatment spectrum as a continuum. It starts with the PCP with an elevated PSA level or if the digital rectal exam has an abnormality, and then the role of the genitourinary (GU) practitioner becomes more significant during the active treatment and diagnostic phases.
Primary care doesn’t disappear, and I think there are 2 major issues that go along with that. First of all, we in primary care, because we take care of patients that often have other comorbidities, need to work with the patient on those comorbidities. Secondly, we need the information shared between the GU and primary care providers so that we can answer questions from our patients and have an understanding of what they’re going through and when.
As time goes on, we go through various phases: We may reach a cure, a quiescent period, active therapy, watchful waiting, or recurrence. Primary care gets involved as time goes on when the disease either becomes quiescent, is just being followed, or is considered cured. Clearly when you have watchful waiting, active treatment, or are in a recurrence, then GU takes the forefront.
I view it as a wave function. Primary care to GU with primary in smaller letters and then primary, if you will, in larger letters, GU becomes a lesser participant unless there is active therapy, watchful waiting or recurrence.
In doing a little bit of research, I found 2 very good and very helpful documents. One is the American Cancer Society (ACS) prostate cancer survivorship care guidelines (Box). And the other is a synopsis of the guidelines. What I liked was that the guidelines focused not only on what should be done for the initial period of prostate cancer, but also for many of the ancillary issues which we often don’t give voice to. The guidelines provide a structure, a foundation to work with our patients over time on their prostate cancer-related issues while, at the same time, being cognizant that we need to deal with their other comorbid conditions.
Modes of Communication
Alison Neymark. We find that including parameters for PSA monitoring in our Progress Notes in the electronic health record (EHR) the best way to communicate with other providers. We’ll say, “If PSA gets to this level, please refer back.” We try to make it clear because with the VA being a training facility, it could be a different resident/attending physician team that’s going to see the patient the next time he is in primary care.
Peter Glassman. Yes, we’re very lucky, as Bill talked about earlier and Alison just mentioned. We have the EHR, and Bill may remember this. Before the EHR, we were constantly fishing to find the most relevant notes. If a patient saw a GU practitioner the day before they saw me, I was often asking the patient what was said. Now we can just review the notes.
It’s a double-edged sword though because there are, of course, many notes in a medical record; and you have to look for the specific items. The EHR and documenting the medical record probably plays the primary role in getting information across. When you want to have an active handoff, or you need to communicate with each other, we have a variety of mechanisms, ranging from the phone to the Microsoft Skype Link (Redmond, WA) system that allows us to tap a message to a colleague.
And I’ve been here long enough that I’ve seen most permutations of how prostate cancer is diagnosed as well as shared among providers. Bill and I have shared patients. Alison and I have shared patients, not necessarily with prostate cancer, although that too. But we know how to communicate with each other. And of course, there’s paging if you need something more urgently.
William Aronson. We also use Microsoft Outlook e-mail, and encrypt the messages to keep them confidential and private. The other nice thing we have is there is a nationwide urology Outlook e-mail, so if any of us have any specific questions, through one e-mail we can send it around the country; and there’s usually multiple very useful responses. That’s another real strength of our system within the VA that helps patient care enormously.
Nicholas Nickols. Sometimes, if there’s a critical note that I absolutely want someone on the care team to read, I’ll add them as a cosigner; and that will pop up when they log in to the Computerized Patient Record System (CPRS) as something that they need to read.
If the patient lives particularly far or gets his care at another VA medical center and laboratory tests are needed, then I will reach out to their PCP via e-mail. If contact is not confirmed, I will reach out via phone or Skype.
Peter Glassman. The most helpful notes are those that are very specific as to what primary care is being asked to do and/or what urology is going to be doing. So, the more specific we get in the notes as to what is being addressed, I think that’s very helpful.
I have been here long enough that I’ve known both Alison and Bill; and if they have an issue, they will tap me a message. It wasn’t long ago that Bill sent a message to me, and we worked on a patient with prostate cancer who was going to be on long-term hormone therapy. We talked about osteoporosis management, and between us we worked out who was going to do what. Those are the kind of shared decision-making situations that are very, very helpful.
Alison Neymark. Also, GLAVAHCS has a home-based primary care team (HBPC), and a lot of the PCPs for that team are NPs. They know that they can contact me for their patients because a lot of those patients are on watchful waiting, and we do not necessarily need to see them face to face in clinic. Our urology team just needs to review updated lab results and how they are doing clinically. The HBPC NP who knows them best can contact me every 6 months or so, and we’ll discuss the case, which avoids making the patient come in, especially when they’re homebound. Those of us that have been working at the VA for many years have established good relationships. We feel very comfortable reaching out and talking to each other about these patients
Peter Glassman. Alison, I agree. When I can talk to my patients and say, “You know, we had that question about,” whatever the question might be, “and I contacted urology, and this is what they said.” It gives the patient confidence that we’re following up on the issues that they have and that we’re communicating with each other in a way that is to their benefit. And I think it’s very appreciated both by the provider as well as the patient.
William Aronson. Not infrequently I’ll have patients who have nonurologic issues, which I may first detect, or who have specific issues with their prostate cancer that can be comanaged. And I have found that when I send an encrypted e-mail to the PCP, it has been an extremely satisfying interaction; and we really get to the heart of the matter quickly for the sake of the veteran.
Veterans With Comorbidities
William Aronson. Posttraumatic stress disorder (PTSD) is a very significant and unique aspect of our patients, which is enormously important to recognize. For example, the side effects of prostate treatments can be very significant, whether radiation or surgery. Our patients understandably can be very fearful of the prostate cancer diagnosis and treatment side effects.
We know, for example, after a patient gets a diagnosis of prostate cancer, they’re at increased risk of cardiac death. That’s an especially important issue for our patients that there be an ongoing interaction between urology and primary care.
The ACS guidelines that Dr. Glassman referred to were enlightening. In many cases, primary care can look at the whole patient and their circumstances better than we can and may detect, for example, specific psychological issues that either they can manage or refer to other specialists.
Peter Glassman. One of the things that was highlighted in the ACS guideline is that in any population of men who have this disease, there’s going to be distress, anxiety, and full-fledged depression. Of course, there are psychosocial aspects of prostate cancer, such as sexual activity and intimacy with a partner that we often don’t explore but are probably playing an important role in the overall health of our patients. We need to be mindful of these psychosocial aspects and at least periodically ask them, “How are you doing with this? How are things at home?” And of course, we already use screeners for depression. As the article noted, distress and anxiety and other factors can make somebody’s life less optimal with poorer quality of life.
Dual Care Patients
Alison Neymark. Many patients whether they have Medicare, insurance through their spouse, or Kaiser Permanente through their job, choose to go to both places. The challenge is communicating with the non-VA providers because here at the VA we can communicate easily through Skype, Outlook e-mail, or CPRS, but for dual care patients who’s in charge? I encourage the veterans to choose whom they want to manage their care; we’re always here and happy to treat them, but they need to decide who’s in charge because I don’t want them to get into a situation where the differing opinions lead to a delay in care.
Nicholas Nickols. The communication when the patient is receiving care outside VA, either on a continuous basis or temporarily, is more of a challenge. We obviously can’t rely upon the messaging system, face-to-face contact is difficult, and they may not be able to use e-mail as well. So in those situations, usually a phone call is the best approach. I have found that the outside providers are happy to speak on the phone to coordinate care.
Peter Glassman. I agree, it does add a layer of complexity because we don’t readily have the notes, any information in front of us. That said, a lot of our patients can and do bring in information from outside specialists, and I’m hopeful that they share the information that we provide back to their outside doctors as well.
William Aronson. Some patient get nervous. They might decide they want care elsewhere, but they still want the VA available for them. I always let them know they should proceed in whatever way they prefer, but we’re always available and here for them. I try to empower them to make their own decisions and feel comfortable with them.
Nicholas Nickols. Notes from the outside, if they’re being referred for VA Choice or community care, do get uploaded into VistA Imaging and can be accessed, although it’s not instantaneous. Sometimes there’s a delay, but I have been able to access outside notes most of the time. If a patient goes through a clinic at the VA, the note is written in real time, and you can read it immediately.
Peter Glassman. That is true for patients that are within the VA system who receive contracted care either through Choice or through non-VA care that is contracted through VA. For somebody who is choosing to use 2 health care systems, that can provide more of a challenge because those notes don’t come to us. Over time, most of my patients have brought test results to me.
The thing with oncologic care, of course, is it’s a lot more complex. And it’s hard to know without reasonable documentation what’s been going on. At some level, you have to trust that the outside provider is doing whatever they need to do, or you have to take it upon yourself to do it within the system.
Alison Neymark. In my experience with the Choice Program, it really depends on the outside providers and how comfortable they are with the system that has been established to share records. Not all providers are going into that system and accessing it. I have had cases where I will see the non-VA provider’s note and it’ll say, “No documentation available for this consultation.” It just happens that they didn’t go into the system to review it. So it can be a challenge.
I’ve had good communication with the providers who use the system correctly. In some cases, just to make it easier, I will go ahead and communicate with them through encrypted e-mail, or I’ll talk to their care coordinators directly by phone.
Peter Glassman. Many, if not most, PCPs are going to take care of these patients, certainly within the VA, with their GU colleagues. And most of us feel comfortable using the current documentation system in a way that allows us to share information or at least to gather information about these patients.
One of the things that I think came out for me in looking at this was that there are guidelines or there are ideas out there on how to take better care of these patients. And I for one learned a fair bit just by going through these documents, which I’m very appreciative of. But it does highlight to me that we can give good care and provide good shared care for prostate cancer survivors. I think that is something that perhaps this discussion will highlight that not only are people doing that, but there are resources they can utilize that will help them get a more comprehensive picture of taking care of prostate cancer survivors in the primary care clinic.
The beauty of the VA system as a system is that as these issues come up that might affect the overall health of the veteran with prostate cancer, for example, psychosocial issues, we have many people that can address this that are experts in their area. And one of the great beauties of having an all-encompassing healthcare system is being able to use resources within the system, whether that be for other medical problems or other social or other psychological issues, that we ourselves are not expert in. We can reach out to our other colleagues and ask them for assistance. We have that available to help the patients. It’s really holistic.
We even have integrated medicine where we can help patients, hopefully, get back into a healthy lifestyle, for example, whereas we may not have that expertise or knowledge. We often think of this as sort of a shared decision between GU and primary care. But, in fact, it’s really the responsibility of many, many people of the system at large. We are very lucky to have that.