User login
MSK Clinic: Evaluating shoulder pain using IPASS



Prenatal exposure to illegal substances: Exploring alternatives to fetal assault bills
Near completion of my rounds at a residential substance abuse treatment center, my last patient was a 32-year-old mother. She reported she was there because her newborn had tested positive for cocaine. She stated, “I want to do better. I thought this pregnancy would change things, but it was so hard to stop.” Her admission made me reflect on a proposed bill in my state of Tennessee called the Fetal Assault Bill, which if put into law would affect women such as my patient and her newborn child.
The Tennessee Fetal Assault Bill was originally enacted in 2014, expired in 2016, and failed to pass in 2017. The bill was reintroduced for consideration in February 2019. If enacted, it would subject a woman to prosecution if her illegal use of a substance while pregnant causes her child to be born addicted to or harmed by that drug. However, the mother is protected from prosecution if she “enrolled in an addiction recovery program before the child is born, remained in the program after delivery, and successfully completed the program, regardless of whether the child was born addicted to or harmed by the narcotic drug.”1 This bill is based on the premise that the unborn fetus has the same rights as a born child, and that the threat of incarceration will deter pregnant women from using illegal substances while pregnant.
Pregnant women may enroll in a drug treatment program prior to delivery to avoid prosecution; however, there is a paucity of addiction treatment centers available to pregnant women. Moreover, there is limited access in areas where babies are more likely to be born affected by the use of illegal substances. Few provisions have been made to address the additional barriers to treatment these women face, such as a lack of insurance or underinsurance for rehabilitation treatment, lack of transportation, and limited finances. Additional barriers include limited social supports, the need for childcare arrangements for existing children, and social stigma.
A 2017 report by Amnesty International regarding the original enactment of the Tennessee Fetal Assault Bill in 2014 noted some mothers who used illegal substances were afraid of prosecution.2 This fear caused some to delay prenatal care or evade social services in order to prevent being reported. According to Amnesty International, laws such as the one proposed in Tennessee often show disparities in how they are implemented. Research shows that women of lower socioeconomic status and minorities tend to receive more frequent drug testing and harsher punishments.3
There are benefits to dedicating more resources to addiction treatment and other social services for pregnant women who use illegal substances. Reports show that mothers are motivated to stay abstinent in treatment centers where they are housed with their children. This model of treatment is more cost-effective than incarceration, which includes legal costs, prison costs, and foster care bills. Moreover, a possible felony charge may hinder a woman’s job opportunities and further compound her problems and those of her infant.
In the light of these benefits, instead of re-enacting the 2014 law, which did not yield any conclusive benefits for newborns or mothers who used illegal substances, alternatives should be attempted. Early identification of and interventions for women who are at risk for substance use while pregnant should be implemented. Practical, accessible support services will encourage sobriety, prevent fetal exposure to illegal substances, and improve child health outcomes. Research shows that substance abuse treatment during pregnancy reduces the risk of harm before birth and improves the quality of parental care after birth.4 Legislators and clinicians should emphasize improving access to treatment, expanding integrative addiction treatment centers, and encouraging self-reporting early in pregnancy. This goal cannot be achieved with an emphasis on incarcerating mothers.
1. House Bill 1168. 111th Tennessee General Assembly (Tenn 2019). http://www.capitol.tn.gov/Bills/111/Bill/HB1168.pdf. Accessed August 8, 2019.
2. Amnesty International. Criminalizing pregnancy: policing pregnant women who use drugs in the USA. https://www.amnesty.org/download/Documents/AMR5162032017ENGLISH.pdf. Published May 23, 2017. Accessed August 8, 2019.
3. Paltrow LM, Flavin J. Arrests of and forced interventions on pregnant women in the United States, 1973-2005: implications for women’s legal status and public health. J Health Polit Policy Law. 2013;38(2):299-343.
4. Ashley OS, Marsden ME, Brady TM. Effectiveness of substance abuse treatment programming for women: a review. Am J Drug Alcohol Abuse. 2003;29(1):19-53.
Near completion of my rounds at a residential substance abuse treatment center, my last patient was a 32-year-old mother. She reported she was there because her newborn had tested positive for cocaine. She stated, “I want to do better. I thought this pregnancy would change things, but it was so hard to stop.” Her admission made me reflect on a proposed bill in my state of Tennessee called the Fetal Assault Bill, which if put into law would affect women such as my patient and her newborn child.
The Tennessee Fetal Assault Bill was originally enacted in 2014, expired in 2016, and failed to pass in 2017. The bill was reintroduced for consideration in February 2019. If enacted, it would subject a woman to prosecution if her illegal use of a substance while pregnant causes her child to be born addicted to or harmed by that drug. However, the mother is protected from prosecution if she “enrolled in an addiction recovery program before the child is born, remained in the program after delivery, and successfully completed the program, regardless of whether the child was born addicted to or harmed by the narcotic drug.”1 This bill is based on the premise that the unborn fetus has the same rights as a born child, and that the threat of incarceration will deter pregnant women from using illegal substances while pregnant.
Pregnant women may enroll in a drug treatment program prior to delivery to avoid prosecution; however, there is a paucity of addiction treatment centers available to pregnant women. Moreover, there is limited access in areas where babies are more likely to be born affected by the use of illegal substances. Few provisions have been made to address the additional barriers to treatment these women face, such as a lack of insurance or underinsurance for rehabilitation treatment, lack of transportation, and limited finances. Additional barriers include limited social supports, the need for childcare arrangements for existing children, and social stigma.
A 2017 report by Amnesty International regarding the original enactment of the Tennessee Fetal Assault Bill in 2014 noted some mothers who used illegal substances were afraid of prosecution.2 This fear caused some to delay prenatal care or evade social services in order to prevent being reported. According to Amnesty International, laws such as the one proposed in Tennessee often show disparities in how they are implemented. Research shows that women of lower socioeconomic status and minorities tend to receive more frequent drug testing and harsher punishments.3
There are benefits to dedicating more resources to addiction treatment and other social services for pregnant women who use illegal substances. Reports show that mothers are motivated to stay abstinent in treatment centers where they are housed with their children. This model of treatment is more cost-effective than incarceration, which includes legal costs, prison costs, and foster care bills. Moreover, a possible felony charge may hinder a woman’s job opportunities and further compound her problems and those of her infant.
In the light of these benefits, instead of re-enacting the 2014 law, which did not yield any conclusive benefits for newborns or mothers who used illegal substances, alternatives should be attempted. Early identification of and interventions for women who are at risk for substance use while pregnant should be implemented. Practical, accessible support services will encourage sobriety, prevent fetal exposure to illegal substances, and improve child health outcomes. Research shows that substance abuse treatment during pregnancy reduces the risk of harm before birth and improves the quality of parental care after birth.4 Legislators and clinicians should emphasize improving access to treatment, expanding integrative addiction treatment centers, and encouraging self-reporting early in pregnancy. This goal cannot be achieved with an emphasis on incarcerating mothers.
Near completion of my rounds at a residential substance abuse treatment center, my last patient was a 32-year-old mother. She reported she was there because her newborn had tested positive for cocaine. She stated, “I want to do better. I thought this pregnancy would change things, but it was so hard to stop.” Her admission made me reflect on a proposed bill in my state of Tennessee called the Fetal Assault Bill, which if put into law would affect women such as my patient and her newborn child.
The Tennessee Fetal Assault Bill was originally enacted in 2014, expired in 2016, and failed to pass in 2017. The bill was reintroduced for consideration in February 2019. If enacted, it would subject a woman to prosecution if her illegal use of a substance while pregnant causes her child to be born addicted to or harmed by that drug. However, the mother is protected from prosecution if she “enrolled in an addiction recovery program before the child is born, remained in the program after delivery, and successfully completed the program, regardless of whether the child was born addicted to or harmed by the narcotic drug.”1 This bill is based on the premise that the unborn fetus has the same rights as a born child, and that the threat of incarceration will deter pregnant women from using illegal substances while pregnant.
Pregnant women may enroll in a drug treatment program prior to delivery to avoid prosecution; however, there is a paucity of addiction treatment centers available to pregnant women. Moreover, there is limited access in areas where babies are more likely to be born affected by the use of illegal substances. Few provisions have been made to address the additional barriers to treatment these women face, such as a lack of insurance or underinsurance for rehabilitation treatment, lack of transportation, and limited finances. Additional barriers include limited social supports, the need for childcare arrangements for existing children, and social stigma.
A 2017 report by Amnesty International regarding the original enactment of the Tennessee Fetal Assault Bill in 2014 noted some mothers who used illegal substances were afraid of prosecution.2 This fear caused some to delay prenatal care or evade social services in order to prevent being reported. According to Amnesty International, laws such as the one proposed in Tennessee often show disparities in how they are implemented. Research shows that women of lower socioeconomic status and minorities tend to receive more frequent drug testing and harsher punishments.3
There are benefits to dedicating more resources to addiction treatment and other social services for pregnant women who use illegal substances. Reports show that mothers are motivated to stay abstinent in treatment centers where they are housed with their children. This model of treatment is more cost-effective than incarceration, which includes legal costs, prison costs, and foster care bills. Moreover, a possible felony charge may hinder a woman’s job opportunities and further compound her problems and those of her infant.
In the light of these benefits, instead of re-enacting the 2014 law, which did not yield any conclusive benefits for newborns or mothers who used illegal substances, alternatives should be attempted. Early identification of and interventions for women who are at risk for substance use while pregnant should be implemented. Practical, accessible support services will encourage sobriety, prevent fetal exposure to illegal substances, and improve child health outcomes. Research shows that substance abuse treatment during pregnancy reduces the risk of harm before birth and improves the quality of parental care after birth.4 Legislators and clinicians should emphasize improving access to treatment, expanding integrative addiction treatment centers, and encouraging self-reporting early in pregnancy. This goal cannot be achieved with an emphasis on incarcerating mothers.
1. House Bill 1168. 111th Tennessee General Assembly (Tenn 2019). http://www.capitol.tn.gov/Bills/111/Bill/HB1168.pdf. Accessed August 8, 2019.
2. Amnesty International. Criminalizing pregnancy: policing pregnant women who use drugs in the USA. https://www.amnesty.org/download/Documents/AMR5162032017ENGLISH.pdf. Published May 23, 2017. Accessed August 8, 2019.
3. Paltrow LM, Flavin J. Arrests of and forced interventions on pregnant women in the United States, 1973-2005: implications for women’s legal status and public health. J Health Polit Policy Law. 2013;38(2):299-343.
4. Ashley OS, Marsden ME, Brady TM. Effectiveness of substance abuse treatment programming for women: a review. Am J Drug Alcohol Abuse. 2003;29(1):19-53.
1. House Bill 1168. 111th Tennessee General Assembly (Tenn 2019). http://www.capitol.tn.gov/Bills/111/Bill/HB1168.pdf. Accessed August 8, 2019.
2. Amnesty International. Criminalizing pregnancy: policing pregnant women who use drugs in the USA. https://www.amnesty.org/download/Documents/AMR5162032017ENGLISH.pdf. Published May 23, 2017. Accessed August 8, 2019.
3. Paltrow LM, Flavin J. Arrests of and forced interventions on pregnant women in the United States, 1973-2005: implications for women’s legal status and public health. J Health Polit Policy Law. 2013;38(2):299-343.
4. Ashley OS, Marsden ME, Brady TM. Effectiveness of substance abuse treatment programming for women: a review. Am J Drug Alcohol Abuse. 2003;29(1):19-53.
Physician assistants in psychiatry: Helping to meet America’s mental health needs
“Canst thou not minister to a mind diseased, pluck from the memory a rooted sorrow, raze out the written troubles of the brain, and with some sweet oblivious antidote cleanse the stuffed bosom of that perilous stuff which weighs upon the heart?”
– William Shakespeare, Macbeth
For many years, the United States has been experiencing a shortage of psychiatrists. Currently, there are only 28,000 to 33,000 psychiatrists in active patient care practice in the United States.1,2 The lack of psychiatrists is pronounced in many areas of the country, including rural regions, some urban neighborhoods, and community health centers. In approximately half of US counties, there are no psychiatrists at all.3
While patients with mental illnesses often are treated in primary care settings, the need for qualified mental health clinicians remains acute. Two-thirds of primary care physicians report difficulty in referring patients for mental health care, due to the shortage of clinicians and long wait times for patients to be seen.4 In the Department of Veterans Affairs (VA), the shortage of qualified psychiatrists is even more acute, due to ongoing combat operations and an increased number of missions and manpower requirements to complete them, which also has increased veterans’ mental health needs during life after their service.5
The outlook for providing adequate numbers of psychiatrists in the future is even more concerning. Based on a population analysis, Satiani et al6 predicts an extreme shortage of psychiatrists for the next 30 years, with the availability of psychiatrists per population expected to reach an all-time low by 2024. Based on ratios from the Department of Health and Human Services, this would mean a shortage of 14,000 to 31,000 psychiatrists over the next 5 to 6 years alone. This is due primarily to the expected retirement of more than 25,000 psychiatrists age >55 during the next 5 years. With mental illness becoming the costliest medical condition in the United States, at $201 billion annually, the potential impact of this shortage is alarming.6
Addressing the shortage
Efforts aimed at increasing the number of psychiatrists, improving access to care, and improving efficiency of care have focused on expanding recruitment and training capacity in psychiatry residency programs, utilizing new models such as telepsychiatry and Assertive Community Treatment (ACT) teams, increasing the number of Certified Community Behavioral Health Clinics, and embedding psychiatrists in large primary care practices.7 Another avenue for addressing the psychiatrist shortage has been the training and hiring of more advanced practice clinicians, including physician assistants (PAs) and
Physician assistants and NPs make up the largest group of non-physician mental health professionals who can prescribe medications. Physician assistant training is most closely aligned with the allopathic training model of physicians.9 Some typical duties of PAs working in a psychiatric setting are outlined in the Table.
How many PAs elect to specialize in psychiatry, compared with the percentage of physicians who choose psychiatry as a career? Data from the National Commission on Certification of Physician Assistants (NCCPA) revealed that in 2018 there were 1,470 PAs working in psychiatry, or approximately 1.5% of all PAs in practice.10 In comparison, approximately 5% of physicians complete residency training in psychiatry.2
Continue to: Although the need for more...
Although the need for more mental health professionals—especially those who can prescribe—is well documented, PA practice in psychiatry has been underrepresented, with PAs choosing to work in the field at a rate just over one-fourth that of physicians. While there is no clear explanation for the lack of PAs in psychiatry, PA programs’ training model has been to produce generalist clinicians who can work in numerous settings, particularly primary care. However, during the past several decades, PA practice choice has shifted largely from primary care to specialty care. In 1974, an estimated 68.8% of PAs worked in primary care settings (family medicine/general practice, general internal medicine, and general pediatrics), while the remainder worked in specialty areas.11 In contrast, by 2018, only 25.8% of PAs worked in primary care settings.10 Despite more PAs choosing to work in medical specialties, the number choosing psychiatry remains very low. With the great need for well-trained mental health prescribers, the opportunity for growth in this area of medicine and increased salary incentive should serve as an impetus for PAs to consider psychiatry. Like their physician counterparts, PAs working in specialty areas of medicine tend to be paid more, sometimes substantially more.12
Training requirements
What is the level of training and experience for PAs who choose to work in psychiatry? Physician assistant program applicants generally come from a pre-med background with a Bachelor’s degree in a hard science, and often have medical experience as a nurse, paramedic, emergency medical technician, or other health profession. Physician assistants are trained in the same medical model of care as physicians, although their training is structured over an average 27-month cycle, with 1 year devoted to didactic education and 1 year or more devoted to clinical training.13 They are qualified to “go to work” soon after graduating and passing the NCCPA Physician Assistant National Certifying Examination (PANCE), and may require a state license. Upon graduation, PAs have received approximately 1,000 hours of didactic and 2,000 hours of clinical training across the general spectrum of medicine.
Physician assistants who choose to specialize in psychiatry may complete a residency/fellowship in psychiatry of approximately 1 year, and/or obtain the Certificate of Added Qualification (CAQ) in psychiatry from the NCCPA. Most PAs who work in psychiatry have done so through “on-the-job” training, where their knowledge and skills have expanded through working with their supervising physician(s) and gaining experience from their clinical practice and self-study. For many years, there were only 1 or 2 PA residency/fellowship opportunities in psychiatry in the United States for PAs wanting to acquire additional formal didactic and clinical knowledge and skills in psychiatry. Fortunately, there has been a growing number of PA residencies/fellowships in psychiatry. These programs are typically 1 year in length and can provide a PA who wants to specialize in psychiatry with an additional 300 to >500 didactic and 1,500 to 2,000 clinical hours of training in the assessment, diagnosis, and treatment of the spectrum of psychiatric conditions. Currently, there are 10 to 12 programs in the United States that offer this training to PAs, producing approximately 18 to 20 residency-trained psychiatric PAs each year. Almost one-half of PAs who are residency-trained in psychiatry are being trained in VA facilities and affiliated institutions sponsored by the VA Office of Academic Affiliations. Along with the PA’s basic education, the additional knowledge and skills acquired in residency prepare the PA to be a highly capable psychiatric clinician, with a combined 1,500 didactic and 4,000 clinical hours of training in general medicine plus psychiatry. The addition of the CAQ demonstrates the PA’s commitment to additional learning in psychiatry, as the added work experience requirements, the additional postgraduate continuing medical education requirements in psychiatry, and the psychiatry board exam clearly show dedication to a higher level of knowledge and skill in the specialty.
Because PAs have been trained as generalists who are able to work in any setting or specialty, they have a broad range of knowledge in medicine and surgery. This can be especially helpful when working in a psychiatric practice, where they can provide an added medical focus to patient care when needed. As more PAs are choosing to work in a specialty area for much or all of their practice, they are able to gain significant knowledge and skills in that specialty.
Getting more PAs into psychiatry
So what does the future hold for PAs in psychiatry? The increased need and opportunity in mental health will likely draw a higher percentage of PAs to this specialty. Hopefully, an increase in the number of PA psychiatry residencies or advanced mental health training opportunities, and the continued goal of obtaining the CAQ in psychiatry, will serve to increase the number of psychiatric PAs.14
Continue to: The NCCPA has also recognized...
The NCCPA has also recognized the importance of increasing PA knowledge and integration in mental health care by establishing a PArtners in Mental Health Steering Committee, composed of leaders from the largest PA organizations, including the American Academy of Physician Assistants, the Accreditation Review Commission on Education for the Physician Assistant, the Physician Assistant Education Association, the Physician Assistant Foundation, and other PA and interprofessional members. The NCCPA’s PArtners in Mental Health Initiative: Stakeholders Report 2018 outlines an ongoing strategy to increase PA engagement in and awareness of mental health among the PA community and future providers via outreach to member organizations, state societies, PA programs, and those at the state and national level who legislate and reimburse PA services for mental health care.15 The Steering Committee’s recommendations include:
- enhancing PA educational approaches in mental health
- strengthening the PA practice environment to address mental health needs and foster integration
- promoting national campaigns to raise the profile of PAs addressing mental health across disciplines
- creating an organizational structure that incorporates current participants, offers backbone support to this movement, and plans for communication and financing.16
The role of PA educators
In the end, PA leaders and educators will play a substantial role in influencing future PAs to seek a career in psychiatry. Currently, psychiatry education varies among PA programs. Some offer robust didactic and clinical education and training, while other programs are limited in the number of hours of psychiatric didactic education, and may offer psychiatry clinical opportunities only in the context of a primary care setting, rather than in a dedicated psychiatric setting. Additionally, the mission of training PAs for generalist, primary care practice may limit many PAs from considering psychiatry because they do not necessarily view psychiatry as closely aligning with primary care generalist practice the way cardiology, pulmonology, gastroenterology, or other internal medicine specialties do.
In terms of PA postgraduate education, many PAs have completed residencies in surgical specialties or emergency medicine. Coincidentally, surgery and emergency medicine residencies are the most prolific of the postgraduate residency programs, adding a significant number of well-trained PAs to these specialties. The NCCPA also offers the CAQ for Cardiovascular and Thoracic Surgery, Orthopedic Surgery, and Emergency Medicine, which may attract PAs into these specialties, with or without completing a residency.
Because the NCCPA also offers the CAQ in Psychiatry, it would be reasonable and attractive for PAs who complete a psychiatry residency to obtain this certification. In fact, the PA psychiatry residency at our own institution trains our residents to be fully prepared and board-eligible to take the CAQ in Psychiatry upon completing residency. To date, every PA residency graduate who has completed our program and taken the CAQ in Psychiatry exam has passed and been awarded the CAQ in Psychiatry. They have proven themselves to the program and the NCCPA, and have impressed their employers with their clinical abilities and medical knowledge.
For psychiatrists, the addition of a well-trained or willing-to-be-trained PA to the practice can provide an economic advantage and strong team partnership that ensures optimal care for patients in this time of shortage of skilled mental health clinicians. The need is clear and will continue. Physician assistant educators must provide adequate didactic and clinical training in psychiatry to PA students, and support students interested in pursuing a career path in this specialty. Physician assistant organizations must meet the challenge of increasing the number of PAs in psychiatry, and encourage the establishment of additional post-graduate residency programs in psychiatry for PAs. Lastly, more PAs need to be made aware that psychiatry is an in-demand specialty that offers broad autonomy and rewarding clinical work.
Continue to: Bottom Line
Bottom Line
Physician assistants (PAs) who choose to specialize in psychiatry will find enormous opportunity, as the need for well-trained and knowledgeable mental health providers is acute. Those PAs who obtain additional training and/or certification in psychiatry will be highly valued and sought-after, with an abundance of job opportunities. Physician assistant programs should continue to improve didactic and clinical training for their students in psychiatry, and encourage increased numbers of PAs to consider psychiatry as a career path.
Related Resources
- National Commission on Certification of Physician Assistants. Psychiatry Certificate of Added Qualifications. https://www.nccpa.net/psychiatry.
- Association of Physician Assistants in Psychiatry. http://psychpa.com/.
1. Japen B. Psychiatrist shortage escalates as U.S. mental health needs grow. Forbes. https://www.forbes.com/sites/brucejapsen/2018/02/25/psychiatrist-shortage-escalates-as-u-s-mental-health-needs-grow/. Published February 25, 2018. Accessed February 1, 2019.
2. Association of American Medical Colleges. Active physicians in the largest specialties, 2017. Table 1.1. Number of active physicians in the largest specialties by major professional activity, 2017. www.aamc.org/data/workforce/reports/492556/1-1-chart.html. Published December 2017. Accessed February 1, 2019.
3. Weiner S. Addressing the escalating psychiatrist shortage. Association of American Medical Colleges AAMCNews. https://news.aamc.org/patient-care/article/addressing-escalating-psychiatrist-shortage/. Published February 13, 2018. Accessed February 1, 2019.
4. Cunningham P. Beyond parity: Primary care physicians’ perspectives on access to mental health care. Health Affairs. 2009;28(S1). https://www.healthaffairs.org/doi/full/10.1377/hlthaff.28.3.w490. Accessed February 1, 2019.
5. Psychiatrist shortage felt nationwide - and in VA system. The Gazette. https://www.thegazette.com/subject/news/government/psychiatrist-shortage-felt-nationwide-x2014-and-in-va-system-20170813. Published August 13, 2017. Accessed February 1, 2019.
6. Satiani A, Niedermier J, Satiani B, et al. Projected workforce of psychiatrists in the United States: a population analysis. Psychiatr Serv. 2018;69(6):710-713.
7. Levine D. What’s the answer to the shortage of mental health care providers? U.S. News & World Report. https://health.usnews.com/health-care/patient-advice/articles/2018-05-25/whats-the-answer-to-the-shortage-of-mental-health-care-providers. Published May 25, 2018. Accessed February 1, 2019.
8. Martsolf GR, Barnes H, Richards MR, et al. Employment of advanced practice clinicians in physician practices. JAMA Intern Med. 2018;178(7):988-990.
9. Hass V. Physician assistants and nurse practitioners are not interchangeable. JAAPA. 2016;29(4):9-12.
10. National Commission on Certification of Physician Assistants. 2018 Statistical profile of certified physician assistants by specialty. Annual report. https://prodcmsstoragesa.blob.core.windows.net/uploads/files/2018StatisticalProfileofCertifiedPAsbySpecialty1.pdf. Published July 2019. Accessed February 1, 2019.
11. Larson EH, Hart LG. Growth and change in the physician assistant workforce in the United States, 1967-2000. J Allied Health. 2007;36(3):121-130.
12. Perna G. NPs and PAs are joining docs in specialty care. Physicians Practice. http://www.physicianspractice.com/staff-salary-survey/nps-and-pas-are-joining-docs-specialty-care. Published May 9, 2016. Accessed February 1, 2019.
13. Pasquini S. Does PA program length matter? The Physician Assistant Life. www.thepalife.com/does-pa-program-length-matter/. Accessed February 1, 2019.
14. National Commission on Certification of Physician Assistants. PAs in specialty practice. An analysis of need, growth and future. http://prodcmsstoragesa.blob.core.windows.net/uploads/files/Whitepaper-PAsinSpecialtyPractice.pdf. Published October 2018. Accessed February 1, 2019.
15. National Commission on Certification of Physician Assistants. PArtners in Mental Health Initiative: Stakeholders Report 2018. http://www.nccpahealthfoundation.net/Portals/0/PDFs/PArtnersinMentalHealthInitiativeStakeholderReport2018.pdf. Published 2018. Accessed February 1, 2019.
16. National Commission on Certification of Physician Assistants. PArtners in Mental Health Summit: proceedings and Recommendations. Leesburg, Virginia – June 4-6, 2017. https://www.nccpahealthfoundation.net/Portals/0/PDFs/SummitProceedings.pdf. Published 2017. Accessed August 7, 2019.
“Canst thou not minister to a mind diseased, pluck from the memory a rooted sorrow, raze out the written troubles of the brain, and with some sweet oblivious antidote cleanse the stuffed bosom of that perilous stuff which weighs upon the heart?”
– William Shakespeare, Macbeth
For many years, the United States has been experiencing a shortage of psychiatrists. Currently, there are only 28,000 to 33,000 psychiatrists in active patient care practice in the United States.1,2 The lack of psychiatrists is pronounced in many areas of the country, including rural regions, some urban neighborhoods, and community health centers. In approximately half of US counties, there are no psychiatrists at all.3
While patients with mental illnesses often are treated in primary care settings, the need for qualified mental health clinicians remains acute. Two-thirds of primary care physicians report difficulty in referring patients for mental health care, due to the shortage of clinicians and long wait times for patients to be seen.4 In the Department of Veterans Affairs (VA), the shortage of qualified psychiatrists is even more acute, due to ongoing combat operations and an increased number of missions and manpower requirements to complete them, which also has increased veterans’ mental health needs during life after their service.5
The outlook for providing adequate numbers of psychiatrists in the future is even more concerning. Based on a population analysis, Satiani et al6 predicts an extreme shortage of psychiatrists for the next 30 years, with the availability of psychiatrists per population expected to reach an all-time low by 2024. Based on ratios from the Department of Health and Human Services, this would mean a shortage of 14,000 to 31,000 psychiatrists over the next 5 to 6 years alone. This is due primarily to the expected retirement of more than 25,000 psychiatrists age >55 during the next 5 years. With mental illness becoming the costliest medical condition in the United States, at $201 billion annually, the potential impact of this shortage is alarming.6
Addressing the shortage
Efforts aimed at increasing the number of psychiatrists, improving access to care, and improving efficiency of care have focused on expanding recruitment and training capacity in psychiatry residency programs, utilizing new models such as telepsychiatry and Assertive Community Treatment (ACT) teams, increasing the number of Certified Community Behavioral Health Clinics, and embedding psychiatrists in large primary care practices.7 Another avenue for addressing the psychiatrist shortage has been the training and hiring of more advanced practice clinicians, including physician assistants (PAs) and
Physician assistants and NPs make up the largest group of non-physician mental health professionals who can prescribe medications. Physician assistant training is most closely aligned with the allopathic training model of physicians.9 Some typical duties of PAs working in a psychiatric setting are outlined in the Table.
How many PAs elect to specialize in psychiatry, compared with the percentage of physicians who choose psychiatry as a career? Data from the National Commission on Certification of Physician Assistants (NCCPA) revealed that in 2018 there were 1,470 PAs working in psychiatry, or approximately 1.5% of all PAs in practice.10 In comparison, approximately 5% of physicians complete residency training in psychiatry.2
Continue to: Although the need for more...
Although the need for more mental health professionals—especially those who can prescribe—is well documented, PA practice in psychiatry has been underrepresented, with PAs choosing to work in the field at a rate just over one-fourth that of physicians. While there is no clear explanation for the lack of PAs in psychiatry, PA programs’ training model has been to produce generalist clinicians who can work in numerous settings, particularly primary care. However, during the past several decades, PA practice choice has shifted largely from primary care to specialty care. In 1974, an estimated 68.8% of PAs worked in primary care settings (family medicine/general practice, general internal medicine, and general pediatrics), while the remainder worked in specialty areas.11 In contrast, by 2018, only 25.8% of PAs worked in primary care settings.10 Despite more PAs choosing to work in medical specialties, the number choosing psychiatry remains very low. With the great need for well-trained mental health prescribers, the opportunity for growth in this area of medicine and increased salary incentive should serve as an impetus for PAs to consider psychiatry. Like their physician counterparts, PAs working in specialty areas of medicine tend to be paid more, sometimes substantially more.12
Training requirements
What is the level of training and experience for PAs who choose to work in psychiatry? Physician assistant program applicants generally come from a pre-med background with a Bachelor’s degree in a hard science, and often have medical experience as a nurse, paramedic, emergency medical technician, or other health profession. Physician assistants are trained in the same medical model of care as physicians, although their training is structured over an average 27-month cycle, with 1 year devoted to didactic education and 1 year or more devoted to clinical training.13 They are qualified to “go to work” soon after graduating and passing the NCCPA Physician Assistant National Certifying Examination (PANCE), and may require a state license. Upon graduation, PAs have received approximately 1,000 hours of didactic and 2,000 hours of clinical training across the general spectrum of medicine.
Physician assistants who choose to specialize in psychiatry may complete a residency/fellowship in psychiatry of approximately 1 year, and/or obtain the Certificate of Added Qualification (CAQ) in psychiatry from the NCCPA. Most PAs who work in psychiatry have done so through “on-the-job” training, where their knowledge and skills have expanded through working with their supervising physician(s) and gaining experience from their clinical practice and self-study. For many years, there were only 1 or 2 PA residency/fellowship opportunities in psychiatry in the United States for PAs wanting to acquire additional formal didactic and clinical knowledge and skills in psychiatry. Fortunately, there has been a growing number of PA residencies/fellowships in psychiatry. These programs are typically 1 year in length and can provide a PA who wants to specialize in psychiatry with an additional 300 to >500 didactic and 1,500 to 2,000 clinical hours of training in the assessment, diagnosis, and treatment of the spectrum of psychiatric conditions. Currently, there are 10 to 12 programs in the United States that offer this training to PAs, producing approximately 18 to 20 residency-trained psychiatric PAs each year. Almost one-half of PAs who are residency-trained in psychiatry are being trained in VA facilities and affiliated institutions sponsored by the VA Office of Academic Affiliations. Along with the PA’s basic education, the additional knowledge and skills acquired in residency prepare the PA to be a highly capable psychiatric clinician, with a combined 1,500 didactic and 4,000 clinical hours of training in general medicine plus psychiatry. The addition of the CAQ demonstrates the PA’s commitment to additional learning in psychiatry, as the added work experience requirements, the additional postgraduate continuing medical education requirements in psychiatry, and the psychiatry board exam clearly show dedication to a higher level of knowledge and skill in the specialty.
Because PAs have been trained as generalists who are able to work in any setting or specialty, they have a broad range of knowledge in medicine and surgery. This can be especially helpful when working in a psychiatric practice, where they can provide an added medical focus to patient care when needed. As more PAs are choosing to work in a specialty area for much or all of their practice, they are able to gain significant knowledge and skills in that specialty.
Getting more PAs into psychiatry
So what does the future hold for PAs in psychiatry? The increased need and opportunity in mental health will likely draw a higher percentage of PAs to this specialty. Hopefully, an increase in the number of PA psychiatry residencies or advanced mental health training opportunities, and the continued goal of obtaining the CAQ in psychiatry, will serve to increase the number of psychiatric PAs.14
Continue to: The NCCPA has also recognized...
The NCCPA has also recognized the importance of increasing PA knowledge and integration in mental health care by establishing a PArtners in Mental Health Steering Committee, composed of leaders from the largest PA organizations, including the American Academy of Physician Assistants, the Accreditation Review Commission on Education for the Physician Assistant, the Physician Assistant Education Association, the Physician Assistant Foundation, and other PA and interprofessional members. The NCCPA’s PArtners in Mental Health Initiative: Stakeholders Report 2018 outlines an ongoing strategy to increase PA engagement in and awareness of mental health among the PA community and future providers via outreach to member organizations, state societies, PA programs, and those at the state and national level who legislate and reimburse PA services for mental health care.15 The Steering Committee’s recommendations include:
- enhancing PA educational approaches in mental health
- strengthening the PA practice environment to address mental health needs and foster integration
- promoting national campaigns to raise the profile of PAs addressing mental health across disciplines
- creating an organizational structure that incorporates current participants, offers backbone support to this movement, and plans for communication and financing.16
The role of PA educators
In the end, PA leaders and educators will play a substantial role in influencing future PAs to seek a career in psychiatry. Currently, psychiatry education varies among PA programs. Some offer robust didactic and clinical education and training, while other programs are limited in the number of hours of psychiatric didactic education, and may offer psychiatry clinical opportunities only in the context of a primary care setting, rather than in a dedicated psychiatric setting. Additionally, the mission of training PAs for generalist, primary care practice may limit many PAs from considering psychiatry because they do not necessarily view psychiatry as closely aligning with primary care generalist practice the way cardiology, pulmonology, gastroenterology, or other internal medicine specialties do.
In terms of PA postgraduate education, many PAs have completed residencies in surgical specialties or emergency medicine. Coincidentally, surgery and emergency medicine residencies are the most prolific of the postgraduate residency programs, adding a significant number of well-trained PAs to these specialties. The NCCPA also offers the CAQ for Cardiovascular and Thoracic Surgery, Orthopedic Surgery, and Emergency Medicine, which may attract PAs into these specialties, with or without completing a residency.
Because the NCCPA also offers the CAQ in Psychiatry, it would be reasonable and attractive for PAs who complete a psychiatry residency to obtain this certification. In fact, the PA psychiatry residency at our own institution trains our residents to be fully prepared and board-eligible to take the CAQ in Psychiatry upon completing residency. To date, every PA residency graduate who has completed our program and taken the CAQ in Psychiatry exam has passed and been awarded the CAQ in Psychiatry. They have proven themselves to the program and the NCCPA, and have impressed their employers with their clinical abilities and medical knowledge.
For psychiatrists, the addition of a well-trained or willing-to-be-trained PA to the practice can provide an economic advantage and strong team partnership that ensures optimal care for patients in this time of shortage of skilled mental health clinicians. The need is clear and will continue. Physician assistant educators must provide adequate didactic and clinical training in psychiatry to PA students, and support students interested in pursuing a career path in this specialty. Physician assistant organizations must meet the challenge of increasing the number of PAs in psychiatry, and encourage the establishment of additional post-graduate residency programs in psychiatry for PAs. Lastly, more PAs need to be made aware that psychiatry is an in-demand specialty that offers broad autonomy and rewarding clinical work.
Continue to: Bottom Line
Bottom Line
Physician assistants (PAs) who choose to specialize in psychiatry will find enormous opportunity, as the need for well-trained and knowledgeable mental health providers is acute. Those PAs who obtain additional training and/or certification in psychiatry will be highly valued and sought-after, with an abundance of job opportunities. Physician assistant programs should continue to improve didactic and clinical training for their students in psychiatry, and encourage increased numbers of PAs to consider psychiatry as a career path.
Related Resources
- National Commission on Certification of Physician Assistants. Psychiatry Certificate of Added Qualifications. https://www.nccpa.net/psychiatry.
- Association of Physician Assistants in Psychiatry. http://psychpa.com/.
“Canst thou not minister to a mind diseased, pluck from the memory a rooted sorrow, raze out the written troubles of the brain, and with some sweet oblivious antidote cleanse the stuffed bosom of that perilous stuff which weighs upon the heart?”
– William Shakespeare, Macbeth
For many years, the United States has been experiencing a shortage of psychiatrists. Currently, there are only 28,000 to 33,000 psychiatrists in active patient care practice in the United States.1,2 The lack of psychiatrists is pronounced in many areas of the country, including rural regions, some urban neighborhoods, and community health centers. In approximately half of US counties, there are no psychiatrists at all.3
While patients with mental illnesses often are treated in primary care settings, the need for qualified mental health clinicians remains acute. Two-thirds of primary care physicians report difficulty in referring patients for mental health care, due to the shortage of clinicians and long wait times for patients to be seen.4 In the Department of Veterans Affairs (VA), the shortage of qualified psychiatrists is even more acute, due to ongoing combat operations and an increased number of missions and manpower requirements to complete them, which also has increased veterans’ mental health needs during life after their service.5
The outlook for providing adequate numbers of psychiatrists in the future is even more concerning. Based on a population analysis, Satiani et al6 predicts an extreme shortage of psychiatrists for the next 30 years, with the availability of psychiatrists per population expected to reach an all-time low by 2024. Based on ratios from the Department of Health and Human Services, this would mean a shortage of 14,000 to 31,000 psychiatrists over the next 5 to 6 years alone. This is due primarily to the expected retirement of more than 25,000 psychiatrists age >55 during the next 5 years. With mental illness becoming the costliest medical condition in the United States, at $201 billion annually, the potential impact of this shortage is alarming.6
Addressing the shortage
Efforts aimed at increasing the number of psychiatrists, improving access to care, and improving efficiency of care have focused on expanding recruitment and training capacity in psychiatry residency programs, utilizing new models such as telepsychiatry and Assertive Community Treatment (ACT) teams, increasing the number of Certified Community Behavioral Health Clinics, and embedding psychiatrists in large primary care practices.7 Another avenue for addressing the psychiatrist shortage has been the training and hiring of more advanced practice clinicians, including physician assistants (PAs) and
Physician assistants and NPs make up the largest group of non-physician mental health professionals who can prescribe medications. Physician assistant training is most closely aligned with the allopathic training model of physicians.9 Some typical duties of PAs working in a psychiatric setting are outlined in the Table.
How many PAs elect to specialize in psychiatry, compared with the percentage of physicians who choose psychiatry as a career? Data from the National Commission on Certification of Physician Assistants (NCCPA) revealed that in 2018 there were 1,470 PAs working in psychiatry, or approximately 1.5% of all PAs in practice.10 In comparison, approximately 5% of physicians complete residency training in psychiatry.2
Continue to: Although the need for more...
Although the need for more mental health professionals—especially those who can prescribe—is well documented, PA practice in psychiatry has been underrepresented, with PAs choosing to work in the field at a rate just over one-fourth that of physicians. While there is no clear explanation for the lack of PAs in psychiatry, PA programs’ training model has been to produce generalist clinicians who can work in numerous settings, particularly primary care. However, during the past several decades, PA practice choice has shifted largely from primary care to specialty care. In 1974, an estimated 68.8% of PAs worked in primary care settings (family medicine/general practice, general internal medicine, and general pediatrics), while the remainder worked in specialty areas.11 In contrast, by 2018, only 25.8% of PAs worked in primary care settings.10 Despite more PAs choosing to work in medical specialties, the number choosing psychiatry remains very low. With the great need for well-trained mental health prescribers, the opportunity for growth in this area of medicine and increased salary incentive should serve as an impetus for PAs to consider psychiatry. Like their physician counterparts, PAs working in specialty areas of medicine tend to be paid more, sometimes substantially more.12
Training requirements
What is the level of training and experience for PAs who choose to work in psychiatry? Physician assistant program applicants generally come from a pre-med background with a Bachelor’s degree in a hard science, and often have medical experience as a nurse, paramedic, emergency medical technician, or other health profession. Physician assistants are trained in the same medical model of care as physicians, although their training is structured over an average 27-month cycle, with 1 year devoted to didactic education and 1 year or more devoted to clinical training.13 They are qualified to “go to work” soon after graduating and passing the NCCPA Physician Assistant National Certifying Examination (PANCE), and may require a state license. Upon graduation, PAs have received approximately 1,000 hours of didactic and 2,000 hours of clinical training across the general spectrum of medicine.
Physician assistants who choose to specialize in psychiatry may complete a residency/fellowship in psychiatry of approximately 1 year, and/or obtain the Certificate of Added Qualification (CAQ) in psychiatry from the NCCPA. Most PAs who work in psychiatry have done so through “on-the-job” training, where their knowledge and skills have expanded through working with their supervising physician(s) and gaining experience from their clinical practice and self-study. For many years, there were only 1 or 2 PA residency/fellowship opportunities in psychiatry in the United States for PAs wanting to acquire additional formal didactic and clinical knowledge and skills in psychiatry. Fortunately, there has been a growing number of PA residencies/fellowships in psychiatry. These programs are typically 1 year in length and can provide a PA who wants to specialize in psychiatry with an additional 300 to >500 didactic and 1,500 to 2,000 clinical hours of training in the assessment, diagnosis, and treatment of the spectrum of psychiatric conditions. Currently, there are 10 to 12 programs in the United States that offer this training to PAs, producing approximately 18 to 20 residency-trained psychiatric PAs each year. Almost one-half of PAs who are residency-trained in psychiatry are being trained in VA facilities and affiliated institutions sponsored by the VA Office of Academic Affiliations. Along with the PA’s basic education, the additional knowledge and skills acquired in residency prepare the PA to be a highly capable psychiatric clinician, with a combined 1,500 didactic and 4,000 clinical hours of training in general medicine plus psychiatry. The addition of the CAQ demonstrates the PA’s commitment to additional learning in psychiatry, as the added work experience requirements, the additional postgraduate continuing medical education requirements in psychiatry, and the psychiatry board exam clearly show dedication to a higher level of knowledge and skill in the specialty.
Because PAs have been trained as generalists who are able to work in any setting or specialty, they have a broad range of knowledge in medicine and surgery. This can be especially helpful when working in a psychiatric practice, where they can provide an added medical focus to patient care when needed. As more PAs are choosing to work in a specialty area for much or all of their practice, they are able to gain significant knowledge and skills in that specialty.
Getting more PAs into psychiatry
So what does the future hold for PAs in psychiatry? The increased need and opportunity in mental health will likely draw a higher percentage of PAs to this specialty. Hopefully, an increase in the number of PA psychiatry residencies or advanced mental health training opportunities, and the continued goal of obtaining the CAQ in psychiatry, will serve to increase the number of psychiatric PAs.14
Continue to: The NCCPA has also recognized...
The NCCPA has also recognized the importance of increasing PA knowledge and integration in mental health care by establishing a PArtners in Mental Health Steering Committee, composed of leaders from the largest PA organizations, including the American Academy of Physician Assistants, the Accreditation Review Commission on Education for the Physician Assistant, the Physician Assistant Education Association, the Physician Assistant Foundation, and other PA and interprofessional members. The NCCPA’s PArtners in Mental Health Initiative: Stakeholders Report 2018 outlines an ongoing strategy to increase PA engagement in and awareness of mental health among the PA community and future providers via outreach to member organizations, state societies, PA programs, and those at the state and national level who legislate and reimburse PA services for mental health care.15 The Steering Committee’s recommendations include:
- enhancing PA educational approaches in mental health
- strengthening the PA practice environment to address mental health needs and foster integration
- promoting national campaigns to raise the profile of PAs addressing mental health across disciplines
- creating an organizational structure that incorporates current participants, offers backbone support to this movement, and plans for communication and financing.16
The role of PA educators
In the end, PA leaders and educators will play a substantial role in influencing future PAs to seek a career in psychiatry. Currently, psychiatry education varies among PA programs. Some offer robust didactic and clinical education and training, while other programs are limited in the number of hours of psychiatric didactic education, and may offer psychiatry clinical opportunities only in the context of a primary care setting, rather than in a dedicated psychiatric setting. Additionally, the mission of training PAs for generalist, primary care practice may limit many PAs from considering psychiatry because they do not necessarily view psychiatry as closely aligning with primary care generalist practice the way cardiology, pulmonology, gastroenterology, or other internal medicine specialties do.
In terms of PA postgraduate education, many PAs have completed residencies in surgical specialties or emergency medicine. Coincidentally, surgery and emergency medicine residencies are the most prolific of the postgraduate residency programs, adding a significant number of well-trained PAs to these specialties. The NCCPA also offers the CAQ for Cardiovascular and Thoracic Surgery, Orthopedic Surgery, and Emergency Medicine, which may attract PAs into these specialties, with or without completing a residency.
Because the NCCPA also offers the CAQ in Psychiatry, it would be reasonable and attractive for PAs who complete a psychiatry residency to obtain this certification. In fact, the PA psychiatry residency at our own institution trains our residents to be fully prepared and board-eligible to take the CAQ in Psychiatry upon completing residency. To date, every PA residency graduate who has completed our program and taken the CAQ in Psychiatry exam has passed and been awarded the CAQ in Psychiatry. They have proven themselves to the program and the NCCPA, and have impressed their employers with their clinical abilities and medical knowledge.
For psychiatrists, the addition of a well-trained or willing-to-be-trained PA to the practice can provide an economic advantage and strong team partnership that ensures optimal care for patients in this time of shortage of skilled mental health clinicians. The need is clear and will continue. Physician assistant educators must provide adequate didactic and clinical training in psychiatry to PA students, and support students interested in pursuing a career path in this specialty. Physician assistant organizations must meet the challenge of increasing the number of PAs in psychiatry, and encourage the establishment of additional post-graduate residency programs in psychiatry for PAs. Lastly, more PAs need to be made aware that psychiatry is an in-demand specialty that offers broad autonomy and rewarding clinical work.
Continue to: Bottom Line
Bottom Line
Physician assistants (PAs) who choose to specialize in psychiatry will find enormous opportunity, as the need for well-trained and knowledgeable mental health providers is acute. Those PAs who obtain additional training and/or certification in psychiatry will be highly valued and sought-after, with an abundance of job opportunities. Physician assistant programs should continue to improve didactic and clinical training for their students in psychiatry, and encourage increased numbers of PAs to consider psychiatry as a career path.
Related Resources
- National Commission on Certification of Physician Assistants. Psychiatry Certificate of Added Qualifications. https://www.nccpa.net/psychiatry.
- Association of Physician Assistants in Psychiatry. http://psychpa.com/.
1. Japen B. Psychiatrist shortage escalates as U.S. mental health needs grow. Forbes. https://www.forbes.com/sites/brucejapsen/2018/02/25/psychiatrist-shortage-escalates-as-u-s-mental-health-needs-grow/. Published February 25, 2018. Accessed February 1, 2019.
2. Association of American Medical Colleges. Active physicians in the largest specialties, 2017. Table 1.1. Number of active physicians in the largest specialties by major professional activity, 2017. www.aamc.org/data/workforce/reports/492556/1-1-chart.html. Published December 2017. Accessed February 1, 2019.
3. Weiner S. Addressing the escalating psychiatrist shortage. Association of American Medical Colleges AAMCNews. https://news.aamc.org/patient-care/article/addressing-escalating-psychiatrist-shortage/. Published February 13, 2018. Accessed February 1, 2019.
4. Cunningham P. Beyond parity: Primary care physicians’ perspectives on access to mental health care. Health Affairs. 2009;28(S1). https://www.healthaffairs.org/doi/full/10.1377/hlthaff.28.3.w490. Accessed February 1, 2019.
5. Psychiatrist shortage felt nationwide - and in VA system. The Gazette. https://www.thegazette.com/subject/news/government/psychiatrist-shortage-felt-nationwide-x2014-and-in-va-system-20170813. Published August 13, 2017. Accessed February 1, 2019.
6. Satiani A, Niedermier J, Satiani B, et al. Projected workforce of psychiatrists in the United States: a population analysis. Psychiatr Serv. 2018;69(6):710-713.
7. Levine D. What’s the answer to the shortage of mental health care providers? U.S. News & World Report. https://health.usnews.com/health-care/patient-advice/articles/2018-05-25/whats-the-answer-to-the-shortage-of-mental-health-care-providers. Published May 25, 2018. Accessed February 1, 2019.
8. Martsolf GR, Barnes H, Richards MR, et al. Employment of advanced practice clinicians in physician practices. JAMA Intern Med. 2018;178(7):988-990.
9. Hass V. Physician assistants and nurse practitioners are not interchangeable. JAAPA. 2016;29(4):9-12.
10. National Commission on Certification of Physician Assistants. 2018 Statistical profile of certified physician assistants by specialty. Annual report. https://prodcmsstoragesa.blob.core.windows.net/uploads/files/2018StatisticalProfileofCertifiedPAsbySpecialty1.pdf. Published July 2019. Accessed February 1, 2019.
11. Larson EH, Hart LG. Growth and change in the physician assistant workforce in the United States, 1967-2000. J Allied Health. 2007;36(3):121-130.
12. Perna G. NPs and PAs are joining docs in specialty care. Physicians Practice. http://www.physicianspractice.com/staff-salary-survey/nps-and-pas-are-joining-docs-specialty-care. Published May 9, 2016. Accessed February 1, 2019.
13. Pasquini S. Does PA program length matter? The Physician Assistant Life. www.thepalife.com/does-pa-program-length-matter/. Accessed February 1, 2019.
14. National Commission on Certification of Physician Assistants. PAs in specialty practice. An analysis of need, growth and future. http://prodcmsstoragesa.blob.core.windows.net/uploads/files/Whitepaper-PAsinSpecialtyPractice.pdf. Published October 2018. Accessed February 1, 2019.
15. National Commission on Certification of Physician Assistants. PArtners in Mental Health Initiative: Stakeholders Report 2018. http://www.nccpahealthfoundation.net/Portals/0/PDFs/PArtnersinMentalHealthInitiativeStakeholderReport2018.pdf. Published 2018. Accessed February 1, 2019.
16. National Commission on Certification of Physician Assistants. PArtners in Mental Health Summit: proceedings and Recommendations. Leesburg, Virginia – June 4-6, 2017. https://www.nccpahealthfoundation.net/Portals/0/PDFs/SummitProceedings.pdf. Published 2017. Accessed August 7, 2019.
1. Japen B. Psychiatrist shortage escalates as U.S. mental health needs grow. Forbes. https://www.forbes.com/sites/brucejapsen/2018/02/25/psychiatrist-shortage-escalates-as-u-s-mental-health-needs-grow/. Published February 25, 2018. Accessed February 1, 2019.
2. Association of American Medical Colleges. Active physicians in the largest specialties, 2017. Table 1.1. Number of active physicians in the largest specialties by major professional activity, 2017. www.aamc.org/data/workforce/reports/492556/1-1-chart.html. Published December 2017. Accessed February 1, 2019.
3. Weiner S. Addressing the escalating psychiatrist shortage. Association of American Medical Colleges AAMCNews. https://news.aamc.org/patient-care/article/addressing-escalating-psychiatrist-shortage/. Published February 13, 2018. Accessed February 1, 2019.
4. Cunningham P. Beyond parity: Primary care physicians’ perspectives on access to mental health care. Health Affairs. 2009;28(S1). https://www.healthaffairs.org/doi/full/10.1377/hlthaff.28.3.w490. Accessed February 1, 2019.
5. Psychiatrist shortage felt nationwide - and in VA system. The Gazette. https://www.thegazette.com/subject/news/government/psychiatrist-shortage-felt-nationwide-x2014-and-in-va-system-20170813. Published August 13, 2017. Accessed February 1, 2019.
6. Satiani A, Niedermier J, Satiani B, et al. Projected workforce of psychiatrists in the United States: a population analysis. Psychiatr Serv. 2018;69(6):710-713.
7. Levine D. What’s the answer to the shortage of mental health care providers? U.S. News & World Report. https://health.usnews.com/health-care/patient-advice/articles/2018-05-25/whats-the-answer-to-the-shortage-of-mental-health-care-providers. Published May 25, 2018. Accessed February 1, 2019.
8. Martsolf GR, Barnes H, Richards MR, et al. Employment of advanced practice clinicians in physician practices. JAMA Intern Med. 2018;178(7):988-990.
9. Hass V. Physician assistants and nurse practitioners are not interchangeable. JAAPA. 2016;29(4):9-12.
10. National Commission on Certification of Physician Assistants. 2018 Statistical profile of certified physician assistants by specialty. Annual report. https://prodcmsstoragesa.blob.core.windows.net/uploads/files/2018StatisticalProfileofCertifiedPAsbySpecialty1.pdf. Published July 2019. Accessed February 1, 2019.
11. Larson EH, Hart LG. Growth and change in the physician assistant workforce in the United States, 1967-2000. J Allied Health. 2007;36(3):121-130.
12. Perna G. NPs and PAs are joining docs in specialty care. Physicians Practice. http://www.physicianspractice.com/staff-salary-survey/nps-and-pas-are-joining-docs-specialty-care. Published May 9, 2016. Accessed February 1, 2019.
13. Pasquini S. Does PA program length matter? The Physician Assistant Life. www.thepalife.com/does-pa-program-length-matter/. Accessed February 1, 2019.
14. National Commission on Certification of Physician Assistants. PAs in specialty practice. An analysis of need, growth and future. http://prodcmsstoragesa.blob.core.windows.net/uploads/files/Whitepaper-PAsinSpecialtyPractice.pdf. Published October 2018. Accessed February 1, 2019.
15. National Commission on Certification of Physician Assistants. PArtners in Mental Health Initiative: Stakeholders Report 2018. http://www.nccpahealthfoundation.net/Portals/0/PDFs/PArtnersinMentalHealthInitiativeStakeholderReport2018.pdf. Published 2018. Accessed February 1, 2019.
16. National Commission on Certification of Physician Assistants. PArtners in Mental Health Summit: proceedings and Recommendations. Leesburg, Virginia – June 4-6, 2017. https://www.nccpahealthfoundation.net/Portals/0/PDFs/SummitProceedings.pdf. Published 2017. Accessed August 7, 2019.
Antidepressants for pediatric patients
Major depressive disorder (MDD) is a significant pediatric health problem, with a lifetime prevalence as high as 20% by the end of adolescence.1-3 Major depressive disorder in adolescence is associated with significant morbidity, including poor social functioning, school difficulties, early pregnancy, and increased risk of physical illness and substance abuse.4-6 It is also linked with significant mortality, with increased risk for suicide, which is now the second leading cause of death in individuals age 10 to 24 years.1,7,8
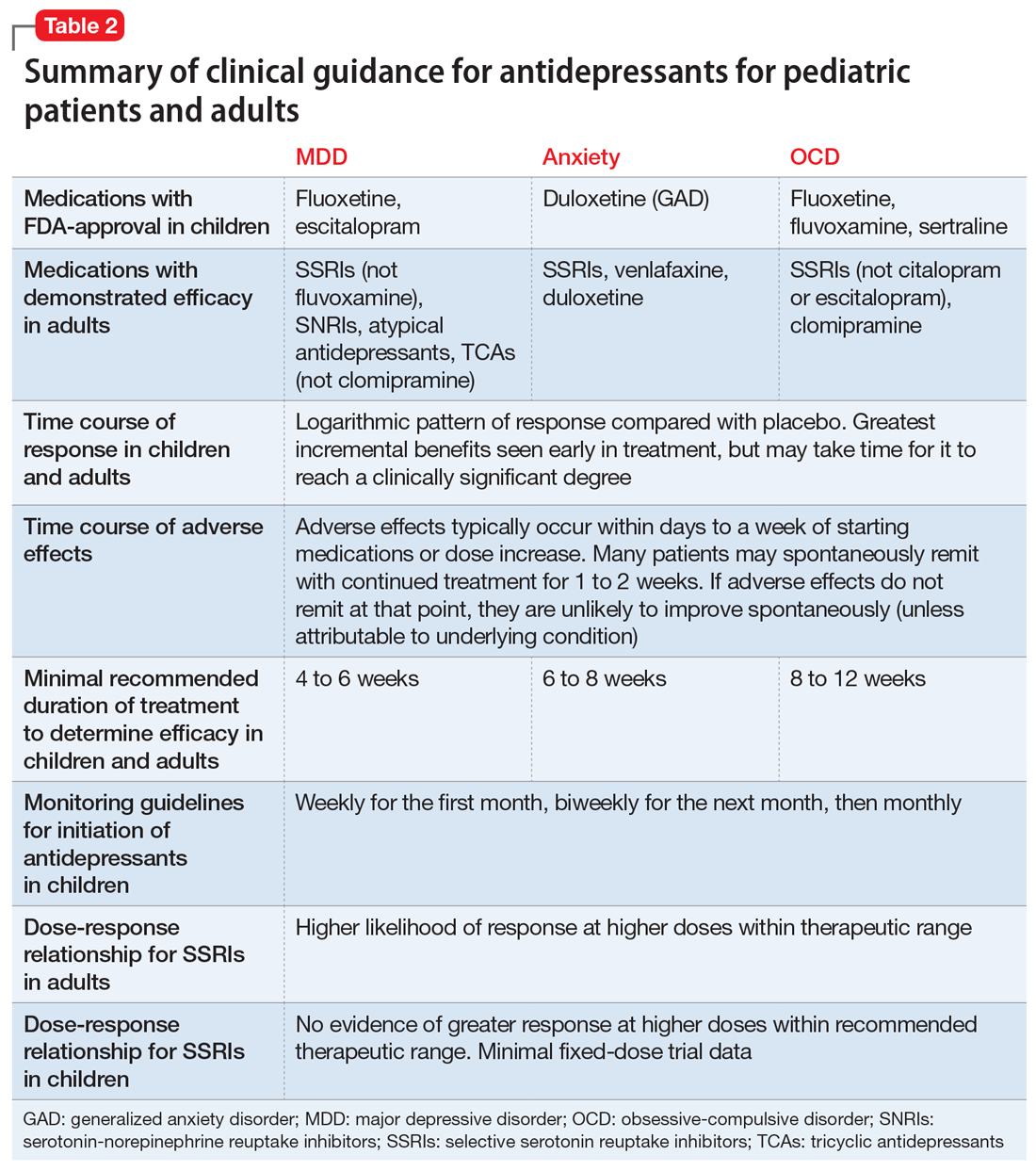
As their name suggests, antidepressants comprise a group of medications that are used to treat MDD; they are also, however, first-line agents for generalized anxiety disorder (GAD), posttraumatic stress disorder (PTSD), and obsessive-compulsive disorder (OCD) in adults. Anxiety disorders (including GAD and other anxiety diagnoses) and PTSD are also common in childhood and adolescence with a combined lifetime prevalence ranging from 15% to 30%.9,10 These disorders are also associated with increased risk of suicide.11 For all of these disorders, depending on the severity of presentation and the preference of the patient, treatments are often a combination of psychotherapy and psychopharmacology.
Clinicians face several challenges when considering antidepressants for pediatric patients. Pediatricians and psychiatrists need to understand whether these medications work in children and adolescents, and whether there are unique developmental safety and tolerability issues. The evidence base in child psychiatry is considerably smaller compared with that of adult psychiatry. From this more limited evidence base also came the controversial “black-box” warning regarding a risk of emergent suicidality when starting antidepressants that accompanies all antidepressants for pediatric, but not adult, patients. This warning has had major effects on clinical encounters with children experiencing depression, including altering clinician prescribing behavior.12
Do antidepressants work in children?
Selective serotonin reuptake inhibitors. Selective serotonin reuptake inhibitors (SSRIs) are the most commonly used class of antidepressants in both children and adults.13 While only a few SSRIs are FDA-approved for pediatric indications, the lack of FDA approval is typically related to a lack of sufficient testing in randomized controlled trials (RCTs) for specific pediatric indications, rather than to demonstrable differences in efficacy between antidepressant agents. Since there is currently no data to suggest inferiority of one agent compared to another in children or adults,14,15 efficacy data will be discussed here as applied to the class of SSRIs, generalizing from RCTs conducted on individual drugs. Table 1 lists FDA indications and dosing information for individual antidepressants.
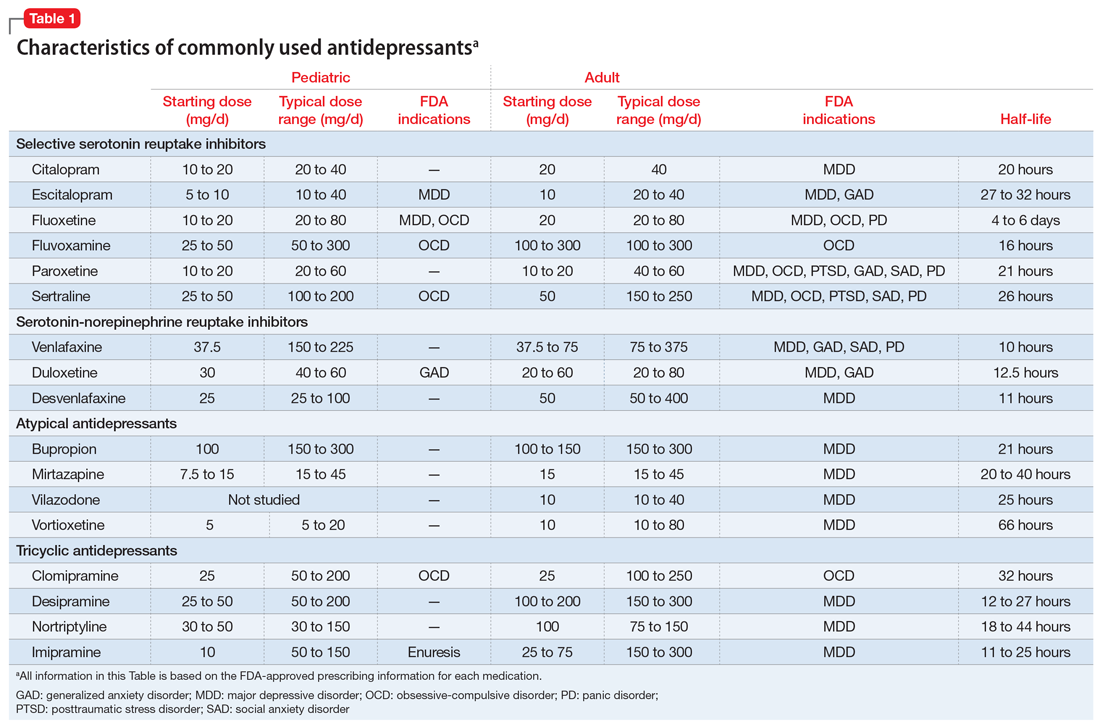
There is strong evidence that SSRIs are effective for treating pediatric anxiety disorders (eg, social anxiety disorder and GAD)16 and OCD,17 with numbers needed to treat (NNT) between 3 and 5. For both of these disorders, SSRIs combined with cognitive-behavioral therapy (CBT) have the highest likelihood of improving symptoms or achieving remission.17,18
Selective serotonin reuptake inhibitors are also effective for treating pediatric MDD; however, the literature is more complex for this disorder compared to GAD and OCD as there are considerable differences in effect sizes between National Institute of Mental Health (NIMH)–funded studies and industry-sponsored trials.13 The major NIMH-sponsored adolescent depression trial, TADS (Treatment for Adolescents and Depression Study), showed that SSRIs (fluoxetine in this case) were quite effective, with an NNT of 4 over the acute phase (12 weeks).19 Ultimately, approximately 80% of adolescents improved over 9 months. Many industry-sponsored trials for MDD in pediatric patients had large placebo response rates (approximately 60%), which resulted in smaller between-group differences, and estimates of an NNT closer to 12,13 which has muddied the waters in meta-analyses that include all trials.20 Improvement in depressive symptoms also appears to be bolstered by concomitant CBT in MDD,19 but not as robustly as in GAD and OCD. While the full benefit of SSRIs for depression may take as long as 8 weeks, a meta-analysis of depression studies of pediatric patients suggests that significant benefits from placebo are observed as early as 2 weeks, and that further treatment gains are minimal after 4 weeks.15 Thus, we recommend at least a 4- to 6-week trial at therapeutic dosing before deeming a medication a treatment failure.
Continue to: Posttraumatic stress disorder...
Posttraumatic stress disorder is a fourth disorder in which SSRIs are a first-line treatment in adults. The data for using SSRIs to treat pediatric patients with PTSD is scant, with only a few RCTs, and no large NIMH-funded trials. Randomized controlled trials have not demonstrated significant differences between SSRIs and placebo21,22 and thus the current first-line recommendation in pediatric PTSD remains trauma-focused therapy, with good evidence for trauma-focused CBT.23 Practically speaking, there can be considerable overlap of PTSD, depression, and anxiety symptoms in children,23 and children with a history of trauma who also have comorbid MDD may benefit from medication if their symptoms persist despite an adequate trial of psychotherapy.
Taken together, the current evidence suggests that SSRIs are often effective in pediatric GAD, OCD, and MDD, with low NNTs (ranging from 3 to 5 based on NIMH-funded trials) for all of these disorders; there is not yet sufficient evidence of efficacy in pediatric patients with PTSD.
Fluoxetine has been studied more intensively than other SSRIs (for example, it was the antidepressant used in the TADS trial), and thus has the largest evidence base. For this reason, fluoxetine is often considered the first of the first-line options. Additionally, fluoxetine has a longer half-life than other antidepressants, which may make it more effective in situations where patients are likely to miss doses, and results in a lower risk of withdrawal symptoms when stopped due to “self-tapering.”
SNRIs and atypical antidepressants. Other antidepressants commonly used in pediatric patients but with far less evidence of efficacy include serotonin-norepinephrine reuptake inhibitors (SNRIs) and the atypical antidepressants bupropion and mirtazapine. The SNRI duloxetine is FDA-approved for treating GAD in children age 7 to 17, but there are no other pediatric indications for duloxetine, or for the other SNRIs.
In general, adverse effect profiles are worse for SNRIs compared to SSRIs, further limiting their utility. While there are no pediatric studies demonstrating SNRI efficacy for neuropathic pain, good data exists in adults.24 Thus, an SNRI could be a reasonable option if a pediatric patient has failed prior adequate SSRI trials and also has comorbid neuropathic pain.
Continue to: Neither bupropion nor mirtazapine...
Neither bupropion nor mirtazapine have undergone rigorous testing in pediatric patients, and therefore these agents should generally be considered only once other first-line treatments have failed. Bupropion has been evaluated for attention-deficit/hyperactivity disorder (ADHD)25 and for adolescent smoking cessation.26 However, the evidence is weak, and bupropion is not considered a first-line option for children and adolescents.
Tricyclic antidepressants. Randomized controlled trials have demonstrated that tricyclic antidepressants (TCAs) are efficacious for treating several pediatric conditions; however, their significant side effect profile, their monitoring requirements, as well as their lethality in overdose has left them replaced by SSRIs in most cases. That said, they can be appropriate in refractory ADHD (desipramine27,28) and refractory OCD (clomipramine is FDA-approved for this indication29); they are considered a third-line treatment for enuresis.30
Why did my patient stop the medication?
Common adverse effects. Although the greatest benefit of antidepressant medications compared with placebo is achieved relatively early on in treatment, it generally takes time for these benefits to accrue and become clinically apparent.15,31 By contrast, most adverse effects of antidepressants present and are at their most severe early in treatment. The combination of early adverse effects and delayed efficacy leads many patients, families, and clinicians to discontinue medications before they have an adequate chance to work. Thus, it is imperative to provide psychoeducation before starting a medication about the typical time-course of improvement and adverse effects (Table 2).
Adverse effects of SSRIs often appear or worsen transiently during initiation of a medication, during a dose increase,32 or, theoretically, with the addition of a medication that interferes with SSRI metabolism (eg, cimetidine inhibition of cytochrome P450 2D6).33 If families are prepared for this phenomenon and the therapeutic alliance is adequate, adverse effects can be tolerated to allow for a full medication trial. Common adverse effects of SSRIs include sleep problems (insomnia/sedation), gastrointestinal upset, sexual dysfunction, dry mouth, and hyperhidrosis. Although SSRIs differ somewhat in the frequency of these effects, as a class, they are more similar than different. Adequate psychoeducation is especially imperative in the treatment of OCD and anxiety disorders, where there is limited evidence of efficacy for any non-serotonergic antidepressants.
Serotonin-norepinephrine reuptake inhibitors are not considered first-line medications because of the reduced evidence base compared to SSRIs and their enhanced adverse effect profiles. Because SNRIs partially share a mechanism of action with SSRIs, they also share portions of the adverse effects profile. However, SNRIs have the additional adverse effect of hypertension, which is related to their noradrenergic activity. Thus, it is reasonable to obtain a baseline blood pressure before initiating an SNRI, as well as periodically after initiation and during dose increases, particularly if the patient has other risk factors for hypertension.34
Continue to: Although TCAs have efficacy...
Although TCAs have efficacy in some pediatric disorders,27-29,35 their adverse effect profile limits their use. Tricyclic antidepressants are highly anticholinergic (causing dizziness secondary to orthostatic hypotension, dry mouth, and urinary retention) and antihistaminergic (causing sedation and weight gain). Additionally, TCAs lower the seizure threshold and have adverse cardiac effects relating to their anti-alpha-1 adrenergic activity, resulting in dose-dependent increases in the QTc and cardiac toxicity in overdose that could lead to arrhythmia and death. These medications have their place, but their use requires careful informed consent, clear treatment goals, and baseline and periodic cardiac monitoring (via electrocardiogram).
Serious adverse effects. Clinicians may be hesitant to prescribe antidepressants for pediatric patients because of the potential for more serious adverse effects, including severe behavioral activation syndromes, serotonin syndrome, and emergent suicidality. However, current FDA-approved antidepressants arguably have one of the most positive risk/benefit profiles of any orally-administered medication approved for pediatric patients. Having a strong understanding of the evidence is critical to evaluating when it is appropriate to prescribe an antidepressant, how to properly monitor the patient, and how to obtain accurate informed consent.
Pediatric behavioral activation syndrome. Many clinicians report that children receiving antidepressants experience a pediatric behavioral activation syndrome, which exists along a spectrum from mild activation, increased energy, insomnia, or irritability up through more severe presentations of agitation, hyperactivity, or possibly mania. A recent meta-analysis suggested a positive association between antidepressant use and activation events on the milder end of this spectrum in pediatric patients with non-OCD anxiety disorders,16 and it is thought that compared with adolescents, younger children are more susceptible to activation adverse effects.36 The likelihood of activation events has been associated with higher antidepressant plasma levels,37 suggesting that dose or individual differences in metabolism may play a role. At the severe end of the spectrum, the risk of induction of mania in pediatric patients with depression or anxiety is relatively rare (<2%) and not statistically different from placebo in RCTs of pediatric participants.38 Meta-analyses of larger randomized, placebo-controlled trials of adults do not support the idea that SSRIs and other second-generation antidepressants carry an increased risk of mania compared with placebo.39,40 Children or adolescents with bona fide bipolar disorder (ie, patients who have had observed mania that meets all DSM-5 criteria) should be treated with a mood-stabilizing agent or antipsychotic if prescribed an antidepressant.41 These clear-cut cases are, however, relatively rare, and more often clinicians are confronted with ambiguous cases that include a family history of bipolar disorder along with “softer” symptoms of irritability, intrusiveness, or aggression. In these children, SSRIs may be appropriate for depressive, OCD, or anxiety symptoms, and should be strongly considered before prescribing antipsychotics or mood stabilizers, as long as initiated with proper monitoring.
Serotonin syndrome is a life-threatening condition caused by excess synaptic serotonin. It is characterized by confusion, sweating, diarrhea, hypertension, hyperthermia, and tachycardia. At its most severe, serotonin syndrome can result in seizures, arrhythmias, and death. The risk of serotonin syndrome is very low when using an SSRI as monotherapy. Risk increases with polypharmacy, particularly unexamined polypharmacy when multiple serotonergic agents are inadvertently on board. Commonly used serotonergic agents include other antidepressants, migraine medications (eg, triptans), some pain medications, and the cough suppressant dextromethorphan.
The easiest way to mitigate the risk of serotonin syndrome is to use an interaction index computer program, which can help ensure that the interacting agents are not prescribed without first discussing the risks and benefits. It is important to teach adolescents that certain recreational drugs are highly serotonergic and can cause serious interactions with antidepressants. For example, recreational use of dextromethorphan or 3,4-methylenedioxymethamphetamine (MDMA; commonly known as “ecstasy”) has been associated with serotonin syndrome in adolescents taking antidepressant medications.42,43
Continue to: Suicidality
Suicidality. The black-box warning regarding a risk of emergent suicidality when starting antidepressant treatment in children is controversial.44 The prospect that a medication intended to ameliorate depression might instead risk increasing suicidal thinking is alarming to parents and clinicians alike. To appropriately weigh and discuss the risks and benefits with families, it is important to understand the data upon which the warning is based.
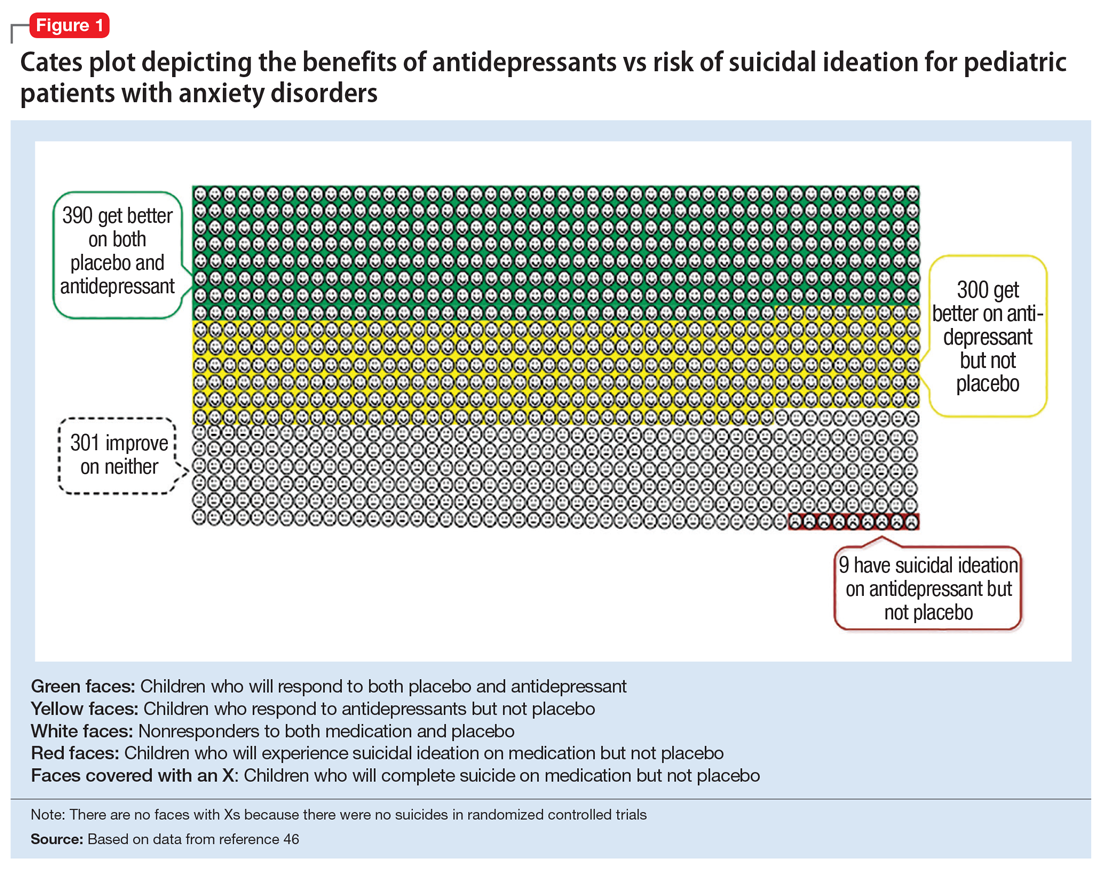
In 2004, the FDA commissioned a review of 23 antidepressant trials, both published and unpublished, pooling studies across multiple indications (MDD, OCD, anxiety, and ADHD) and multiple antidepressant classes. This meta-analysis, which included nearly 4,400 pediatric patients, found a small but statistically significant increase in spontaneously-reported suicidal thoughts or actions, with a risk difference of 1% (95% confidence interval [CI], 1% to 2%).45 These data suggest that if one treats 100 pediatric patients, 1 to 2 of them may experience short-term increases in suicidal thinking or behavior.45 There were no differences in suicidal thinking when assessed systematically (ie, when all subjects reported symptoms of suicidal ideation on structured rating scales), and there were no completed suicides.45 A subsequent analysis that included 27 pediatric trials suggested an even lower, although still significant, risk difference (<1%), yielding a number needed to harm (NNH) of 143.46 Thus, with low NNT for efficacy (3 to 6) and relatively high NNH for emergent suicidal thoughts or behaviors (100 to 143), for many patients the benefits will outweigh the risks.
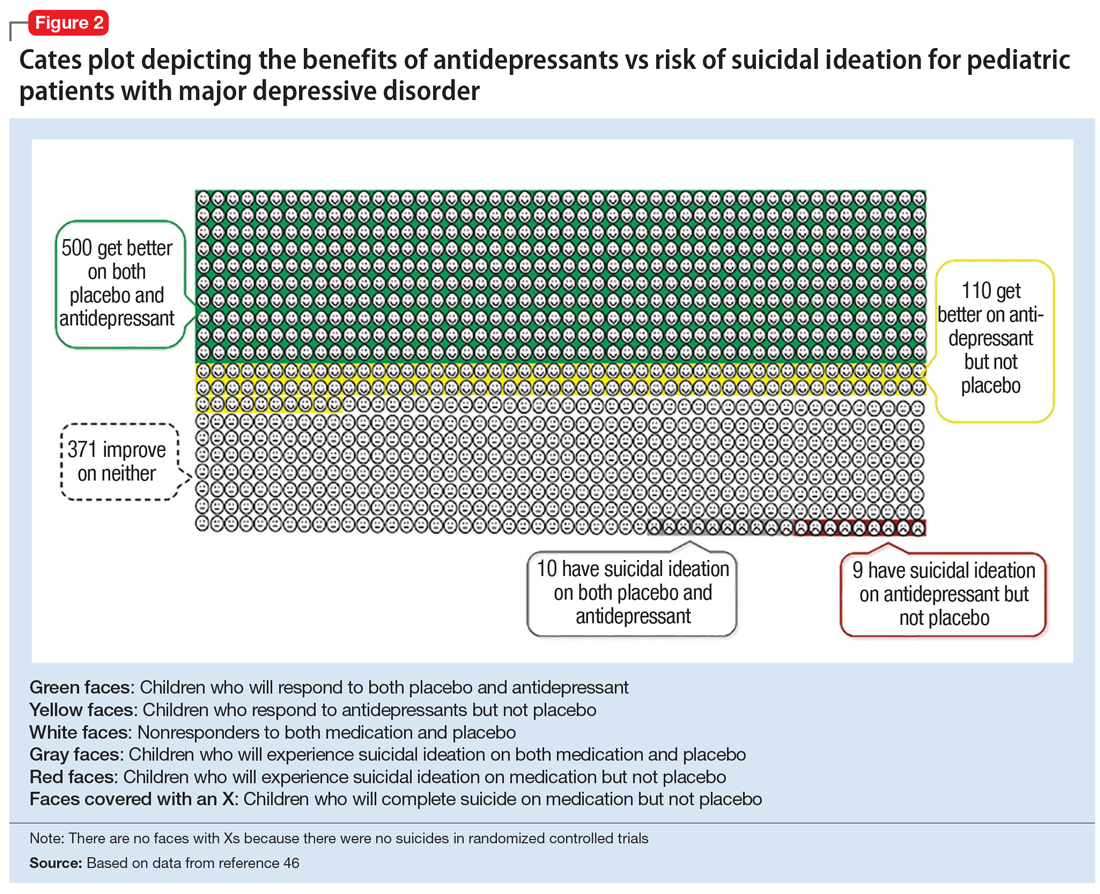
Figure 1, Figure 2, and Figure 3 are Cates plots that depict the absolute benefits of antidepressants compared with the risk of suicidality for pediatric patients with MDD, OCD, and anxiety disorders. Recent meta-analyses have suggested that the increased risk of suicidality in antidepressant trials is specific to studies that included children and adolescents, and is not observed in adult studies. A meta-analysis of 70 trials involving 18,526 participants suggested that the odds ratio of suicidality in trials of children and adolescents was 2.39 (95% CI, 1.31 to 4.33) compared with 0.81 (95% CI, 0.51 to 1.28) in adults.47 Additionally, a network meta-analysis exclusively focusing on pediatric antidepressant trials in MDD reported significantly higher suicidality-related adverse events in venlafaxine trials compared with placebo, duloxetine, and several SSRIs (fluoxetine, paroxetine, and escitalopram).20 These data should be interpreted with caution as differences in suicidality detected between agents is quite possibly related to differences in the method of assessment between trials, as opposed to actual differences in risk between agents.
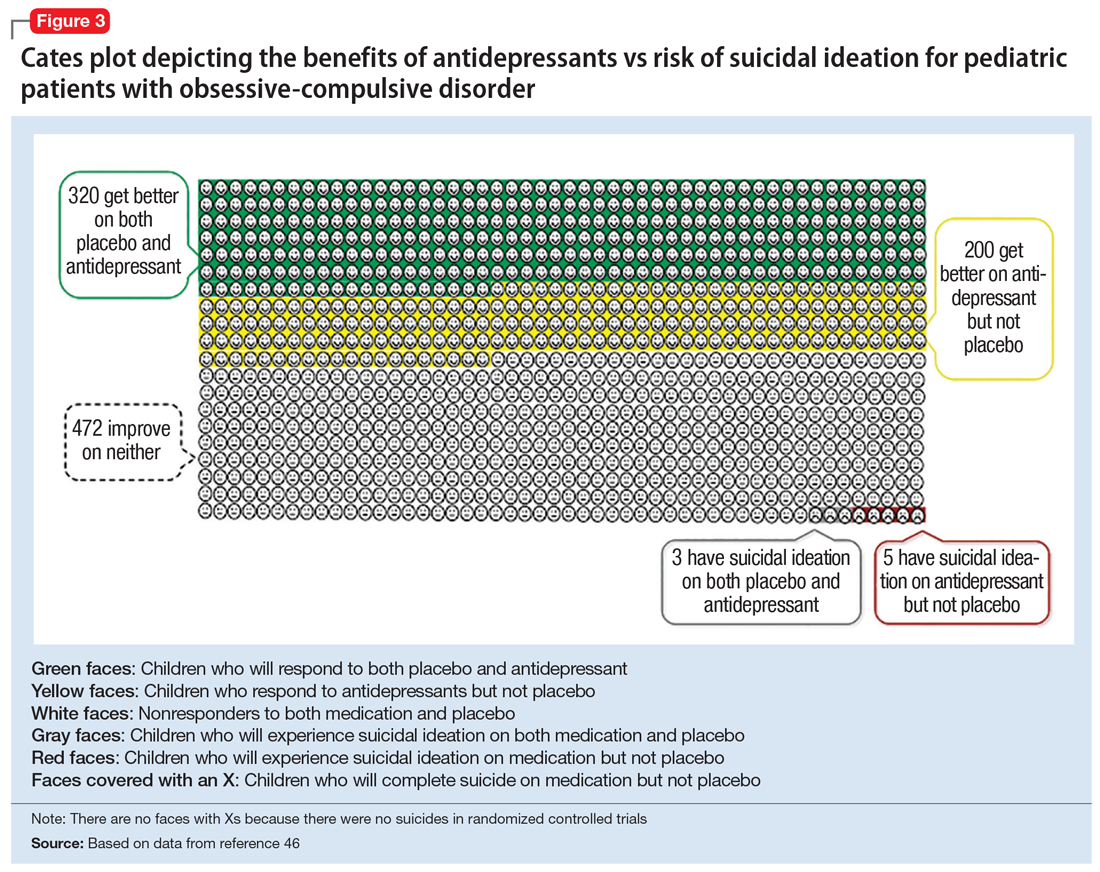
Epidemiologic data further support the use of antidepressants in pediatric patients, showing that antidepressant use is associated with decreased teen suicide attempts and completions,48 and the decline in prescriptions that occurred following the black-box warning was accompanied by a 14% increase in teen suicides.49 Multiple hypotheses have been proposed to explain the pediatric clinical trial findings. One idea is that potential adverse effects of activation, or the intended effects of restoring the motivation, energy, and social engagement that is often impaired in depression, increases the likelihood of thinking about suicide or acting on thoughts. Another theory is that reporting of suicidality may be increased, rather than increased de novo suicidality itself. Antidepressants are effective for treating pediatric anxiety disorders, including social anxiety disorder,16 which could result in more willingness to report. Also, the manner in which adverse effects are generally ascertained in trials might have led to increased spontaneous reporting. In many trials, investigators ask whether participants have any adverse effects in general, and inquire about specific adverse effects only if the family answers affirmatively. Thus, the increased rate of other adverse effects associated with antidepressants (sleep problems, gastrointestinal upset, dry mouth, etc.) might trigger a specific question regarding suicidal ideation, which the child or family then may be more likely to report. Alternatively, any type of psychiatric treatment could increase an individual’s propensity to report; in adolescent psychotherapy trials, non-medic
Continue to: How long should the antidepressant be continued?
How long should the antidepressant be continued?
Many patients are concerned about how long they may be taking medication, and whether they will be taking an antidepressant “forever.” A treatment course can be broken into an acute phase, wherein remission is achieved and maintained for 6 to 8 weeks. This is followed by a continuation phase, with the goal of relapse prevention, lasting 16 to 20 weeks. The length of the last phase—the maintenance phase—depends both on the child’s history, the underlying therapeutic indication, the adverse effect burden experienced, and the family’s preferences/values. In general, for a first depressive episode, after treating for 1 year, a trial of discontinuation can be attempted with close monitoring. For a second depressive episode, we recommend 2 years of remission on antidepressant therapy before attempting discontinuation. In patients who have had 3 depressive episodes, or have had episodes of high severity, we recommend continuing antidepressant treatment indefinitely. Although much less well studied, the risk of relapse following SSRI discontinuation appears much more significant in OCD, whereas anxiety disorders and MDD have a relatively comparable risk.52
In general, stopping an antidepressant should be done carefully and slowly.
What to do when first-line treatments fail
Adequate psychotherapy? To determine whether children are receiving adequate CBT, ask:
- if the child receives homework from psychotherapy
- if the parents are included in treatment
- if therapy has involved identifying thought patterns that may be contributing to the child’s illness, and
- if the therapist has ever exposed the child to a challenge likely to produce anxiety or distress in a supervised environment and has developed an exposure hierarchy (for conditions with primarily exposure-based therapies, such as OCD or anxiety disorders).
If a family is not receiving most of these elements in psychotherapy, this is a good indicator that they may not be receiving evidence-based CBT.
Continue to: Adequate pharmacotherapy?
Adequate pharmacotherapy? Similarly, when determining the adequacy of previous pharmacotherapy, it is critical to determine whether the child received an adequate dose of medications (at least the FDA-recommended minimum dose) for an adequate duration of time at therapeutic dosing (at least 6 weeks for MDD, 8 weeks for anxiety disorders, and 8 to 12 weeks for pediatric patients with OCD), and that the child actually took the medication regularly during that period. Patient compliance can typically be tracked through checking refill requests or intervals through the patient’s pharmacy. Ensuring proper delivery of first-line treatments is imperative because (1) the adverse effects associated with second-line treatments are often more substantial; (2) the cost in terms of time and money is considerably higher with second-line treatments, and; (3) the evidence regarding the benefits of these treatments is much less certain.
Inadequate dosing is a common reason for non-response in pediatric patients. Therapeutic dose ranges for common antidepressants are displayed in Table 1. Many clinicians underdose antidepressants for pediatric patients initially (and often throughout treatment) because they fear that the typical dose titration used in clinical trials will increase the risk of adverse effects compared with more conservative dosing. There is limited evidence to suggest that this underdosing strategy is likely to be successful; adverse effects attributable to these medications are modest, and most that are experienced early in treatment (eg, headache, increased anxiety or irritability, sleep problems, gastrointestinal upset) are self-limiting and may be coincidental rather than medication-induced. Furthermore, there is no evidence for efficacy of subtherapeutic dosing in children in the acute phase of treatment or for preventing relapse.14 Thus, from an efficacy standpoint, a medication trial has not officially begun until the therapeutic dose range is reached.
Once dosing is within the therapeutic range, however, pediatric data differs from the adult literature. In most adult psychiatric conditions, higher doses of SSRIs within the therapeutic range are associated with an increased response rate.14,54 In pediatrics, there are few fixed dose trials, and once within the recommended therapeutic range, minimal data supports an association between higher dosing and higher efficacy.14 In general, pediatric guidelines are silent regarding optimal dosing of SSRIs within the recommended dose range, and higher antidepressant doses often result in a more significant adverse effect burden for children. One exception is pediatric OCD, where, similar to adults, the guidelines suggest that higher dosing of SSRIs often is required to induce a therapeutic response as compared to MDD and GAD.31,55
If a child does not respond to adequate first-line treatment (or has a treatment history that cannot be fully verified), repeating these first-line interventions carries little risk and can be quite effective. For example, 60% of adolescents with MDD who did not initially respond to an SSRI demonstrated a significant response when prescribed a second SSRI or venlafaxine (with or without CBT).56
When pediatric patients continue to experience significantly distressing and/or debilitating symptoms (particularly in MDD) despite multiple trials of antidepressants and psychotherapy, practitioners should consider a careful referral to interventional psychiatry services, which can include the more intensive treatments of electroconvulsive therapy, repetitive transcranial magnetic stimulation, or ketamine (see Box 1). Given the substantial morbidity and mortality associated with adolescent depression, interventional psychiatry treatments are under-researched and under-utilized clinically in pediatric populations.
Continue to: Antidepressants in general...
Antidepressants in general, and SSRIs in particular, are the first-line pharmacotherapy for pediatric anxiety, OCD, and MDD. For PTSD, although they are a first-line treatment in adults, their efficacy has not been demonstrated in children and adolescents. Antidepressants are generally safe, well-tolerated, and effective, with low NNTs (3 to 5 for anxiety and OCD; 4 to 12 in MDD, depending on whether industry trials are included). It is important that clinicians and families be educated about possible adverse effects and their time course in order to anticipate difficulties, ensure adequate informed consent, and monitor appropriately. The black-box warning regarding treatment-emergent suicidal thoughts or behaviors must be discussed (for suggested talking points, see Box 2). The NNH is large (100 to 143), and for many patients, the benefits will outweigh the risks. For pediatric patients who fail to respond to multiple adequate trials of antidepressants and psychotherapy, referrals for interventional psychiatry consultation should be considered.
Bottom Line
Although the evidence base for prescribing antidepressants for children and adolescents is smaller compared to the adult literature, properly understanding and prescribing these agents, and explaining their risks and benefits to families, can make a major difference in patient compliance, satisfaction, and outcomes. Antidepressants (particularly selective serotonin reuptake inhibitors) are the firstline pharmacologic intervention for pediatric patients with anxiety disorders, obsessive-compulsive disorder, or major depressive disorder.
Related Resource
- Bonin L, Moreland CS. Overview of prevention and treatment for pediatric depression. https://www.uptodate.com/contents/overview-of-prevention-and-treatment-for-pediatric-depression. Last reviewed July 2019.
Drug Brand Names
Bupropion • Wellbutrin, Zyban
Cimetidine • Tagamet
Citalopram • Celexa
Clomipramine • Anafranil
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Fluvoxamine • Luvox
Imipramine • Tofranil
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Venlafaxine • Effexor
Vilazodone • Viibryd
Vortioxetine • Trintellix
Box 1
Continuing severe depression is associated with reduced educational attainment and quality of life, as well as increased risk of substance abuse and suicide,1,2 which is the second leading cause of death in individuals age 10 to 24 years.3 Given the substantial morbidity and mortality associated with adolescent depression, interventional psychiatry treatments are under-researched and underutilized in pediatric patients. Interventional antidepressants in adults include electroconvulsive therapy (ECT), repetitive transcranial magnetic stimulation (rTMS), and, most recently, ketamine.
Electroconvulsive therapy is the most effective therapy available for depression in adults, alleviating depressive symptoms in treatment-refractory patients and outperforming both pharmacotherapy4 and rTMS.5 Despite its track record of effectiveness and safety in adults, ECT continues to suffer considerable stigma.4 Cognitive adverse effects and memory problems in adults are generally self-limited, and some aspects of cognition actually improve after ECT as depression lifts.6 The combination of stigma and the concern about possible cognitive adverse effects during periods of brain development have likely impeded the rigorous testing of ECT in treatment-refractory pediatric patients. Several case series and other retrospective analyses suggest, however, that ECT has strong efficacy and limited adverse effects in adolescents who have severe depression or psychotic symptoms.7-9 Despite these positive preliminary data in pediatric patients, and a large body of literature in adults, no controlled trials of ECT have been conducted in the pediatric population, and it remains a rarely used treatment in severe pediatric mental illness.
Repetitive transcranial magnetic stimulation is a technique in which magnetic stimulation is used to activate the left dorsolateral prefrontal cortex (DLPFC), a target thought to be important in the pathophysiology of MDD. Repetitive transcranial magnetic stimulation is FDAapproved to treat medication-refractory major depressive disorder (MDD) in adults, and has been shown to be effective as both a monotherapy10 and an adjunctive treatment.11 The estimated number needed to treat (NNT) for rTMS ranges from 6 to 8, which is quite effective, although less so than ECT (and probably initial pharmacotherapy).5 Similar to ECT, however, there are no large randomized controlled trials (RCTs) in children or adolescents. Pilot RCTs12 and open trials13 suggest that DLPFC rTMS may be effective as an adjunctive treatment, speeding or augmenting response to a selective serotonin reuptake inhibitor in adolescents with MDD. Larger trials studying rTMS in pediatric patients are needed. While rTMS is generally well tolerated, disadvantages include the time-consuming schedule (the initial treatment is typically 5 days/week for several weeks) and local adverse effects of headache and scalp pain.
Ketamine, which traditionally is used as a dissociative anesthetic, is a rapidly emerging novel treatment in adult treatment-refractory MDD. It acts quickly (within hours to days) and cause significant improvement in difficult symptoms such as anhedonia14 and suicidal ideation.15 In adult studies, ketamine has a robust average effect size of >1.2, and an NNT ranging from 3 to 5 in medication-refractory patients.16,17 Ketamine is a glutamatergic modulator, acting outside of the monoamine neurochemical systems traditionally targeted by standard antidepressants.16 The efficacy of ketamine in treatment-refractory adults is impressive, but the effects of a single treatment are ephemeral, dissipating within 1 to 2 weeks, which has led to significant discussion surrounding optimal dosing strategies.16 Although small RCTs in pediatric patients are currently underway, at this time, the only evidence for ketamine for pediatric MDD is based on case series/report data18,19 which was positive.
For all of these interventional modalities, it is critical to refer children with treatmentrefractory disorders to interventionists who have appropriate experience and monitoring capabilities.
References
1. Weissman MM, Wolk S, Goldstein RB, et al. Depressed adolescents grown up. JAMA.1999;281(18):1707-1713.
2. Fergusson DM, Woodward LJ. Mental health, educational, and social role outcomes of adolescents with depression. Arch Gen Psychiatry. 2002;59(3):225-231.
3. Centers for Disease Control and Prevention. National Vital Statistics System. Deaths, percent of total deaths, and death rates for the 15 leading causes of death in 5-year age groups, by race and sex: United States, 1999-2015. Centers for Disease Control and Prevention. https://www.cdc.gov/nchs/nvss/mortality/lcwk1.htm. Published October 23, 2017. Accessed May 2, 2019.
4. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and metaanalysis. Lancet. 2003;361(9360):799-808.
5. Berlim MT, Van den Eynde F, Daskalakis ZJ. Efficacy and acceptability of high frequency repetitive transcranial magnetic stimulation (rTMS) versus electroconvulsive therapy (ECT) for major depression: a systematic review and meta-analysis of randomized trials. Depress Anxiety. 2013;30(7):614-623.
6. Semkovska M, McLoughlin DM. Objective cognitive performance associated with electroconvulsive therapy for depression: a systematic review and meta-analysis. Biol Psychiatry. 2010;68(6):568-577.
7. Jacob P, Gogi PK, Srinath S, et al. Review of electroconvulsive therapy practice from a tertiary child and adolescent psychiatry centre. Asian J Psychiatr. 2014;12(1):95-99.
8. Zhand N, Courtney DB, Flament MF. Use of electroconvulsive therapy in adolescents with treatment-resistant depressive disorders: a case series. J ECT. 2015;31(4):238-245.
9. Puffer CC, Wall CA, Huxsahl JE, et al. A 20 year practice review of electroconvulsive therapy for adolescents. J Child Adolesc Psychopharmacol. 2016;26(7):632-636.
10. Berlim MT, van den Eynde F, Tovar-Perdomo S, et al. Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rTMS) for treating major depression: a systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol Med. 2014;44(2):225-239.
11. Liu B, Zhang Y, Zhang L, et al. Repetitive transcranial magnetic stimulation as an augmentative strategy for treatment-resistant depression, a meta-analysis of randomized, double-blind and sham-controlled study. BMC Psychiatry. 2014;14:342.
12. Huang ML, Luo BY, Hu JB, et al. Repetitive transcranial magnetic stimulation in combination with citalopram in young patients with first-episode major depressive disorder: a double-blind, randomized, sham-controlled trial. Aust N Z J Psychiatry. 2012;46(3):257-264.
13. Wall CA, Croarkin PE, Sim LA, et al. Adjunctive use of repetitive transcranial magnetic stimulation in depressed adolescents: a prospective, open pilot study. J Clin Psychiatry. 2011;72(9):1263-1269.
14. Lally N, Nugent AC, Luckenbaugh DA, et al. Anti-anhedonic effect of ketamine and its neural correlates in treatment-resistant bipolar depression. Transl Psychiatry. 2014;4:e469. doi: 10.1038/tp.2014.105.
15. Ballard ED, Ionescu DF, Vande Voort JL, et al. Improvement in suicidal ideation after ketamine infusion: relationship to reductions in depression and anxiety. J Psychiatr Res. 2014;58:161-166.
16. Newport DJ, Carpenter LL, McDonald WM, et al. Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry. 2015;172(10):950-966.
17. McGirr A, Berlim MT, Bond DJ, et al. A systematic review and meta-analysis of randomized, double-blind, placebo-controlled trials of ketamine in the rapid treatment of major depressive episodes. Psychol Med. 2015;45(4):693-704.
18. Dwyer JB, Beyer C, Wilkinson ST, et al. Ketamine as a treatment for adolescent depression: a case report. J Am Acad Child Adolesc Psychiatry. 2017;56(4):352-354.
19. Cullen KR, Amatya P, Roback MG, et al. Intravenous ketamine for adolescents with treatment-resistant depression: an open-label study. J Child Adolesc Psychopharmacol. 2018;28(7):437-444.
Box 2
Efficacy
- Selective serotonin reuptake inhibitors are the most effective pharmacologic treatment we have for pediatric depression, OCD, and anxiety
- More than one-half of children who are prescribed SSRIs have a significant improvement, regardless of condition
- Based on current estimates, we need to treat 4 to 6 children with an SSRI to find one that will improve who would not improve with placebo
- The clinical benefits of SSRIs generally take a while to accrue; therefore, it is advisable to take the medication for at least 2 to 3 months before concluding that it is ineffective
- In addition to medication, evidence-based psychotherapies provide significant benefit for pediatric depression, OCD, and anxiety
Tolerability
- Most commonly prescribed pediatric antidepressants have been used safely in children for 2 to 3 decades. The safety profiles of SSRIs are among the best of any medications used for children and adolescents
- While many children get better when taking these medications, it’s important that we also talk about potential adverse effects. Some children will experience sleep problems (either sleepier than usual or difficulty sleeping), changes in energy levels, headache, gastrointestinal upset, and dry mouth. These are most likely at the beginning of treatment, or when we increase the dose; they usually are time-limited and go away on their own
- Often adverse effects occur first and the benefits come later. Because it may take at least a few weeks to start to see the mood/anxiety benefits, it’s important for us to talk about any adverse effects your child experiences and remember that they usually are short-lived
Suicidality
- The FDA placed a “black-box” warning on antidepressants after pediatric studies found a small but statistically significant increased risk of reporting suicidal thoughts or behaviors over the short-term compared with placebo
- The increased risk of spontaneously reporting suicidal ideation was quite small. Studies suggested that one would need to treat 100 to 140 children to see 1 child report suicidal ideation compared to placebo. Suicidal ideation is a common symptom in children with depression and anxiety
- Studies found no increased risk when suicidal ideation was systematically assessed using structured rating scales
- In the studies evaluated, there were no completed suicides by patients taking medication or placebo
- Population studies show that higher rates of antidepressant prescriptions are associated with lower rates of attempted and completed teen suicide, which underscores that in general, these medicines treat the underlying causes of suicidality
- No scientific consensus exists on whether these medications are truly associated with an increased risk of new-onset suicidal ideation, or if this association is due to other factors (eg, improvement in anxiety and depressive symptoms that make patients more comfortable to report suicidal ideation spontaneously)
- Regardless, the FDA recommends frequent monitoring of children for suicidal thoughts when these medications are started. This should be done anyway in children experiencing depression and anxiety, and it’s why we will plan to have more frequent appointments as the medication is initiated
OCD: obsessive-compulsive disorder; SSRIs: selective serotonin reuptake inhibitors
1. Williams SB, O’Connor EA, Eder M, et al. Screening for child and adolescent depression in primary care settings: a systematic evidence review for the US Preventive Services Task Force. Pediatrics. 2009;123(4):e716-e735. doi: 10.1542/peds.2008-2415.
2. Kessler RC, Avenevoli S, Ries Merikangas K. Mood disorders in children and adolescents: an epidemiologic perspective. Biol Psychiatry. 2001;49(12):1002-1014.
3. Lewinsohn PM, Clarke GN, Seeley JR, et al. Major depression in community adolescents: age at onset, episode duration, and time to recurrence. J Am Acad Child Adolesc Psychiatry. 1994;33(6):809-818.
4. Weissman MM, Wolk S, Goldstein RB, et al. Depressed adolescents grown up. JAMA.1999;281(18):1707-1713.
5. Fergusson DM, Woodward LJ. Mental health, educational, and social role outcomes of adolescents with depression. Arch Gen Psychiatry. 2002;59(3):225-231.
6. Keenan-Miller D, Hammen CL, Brennan PA. Health outcomes related to early adolescent depression. J Adolesc Health. 2007; 41(3): 256-62.
7. Shaffer D, Gould MS, Fisher P, et al. Psychiatric diagnosis in child and adolescent suicide. Arch Gen Psychiatry. 1996;53(4):339-348.
8. Centers for Disease Control and Prevention. National Vital Statistics System. Deaths, percent of total deaths, and death rates for the 15 leading causes of death in 5-year age groups, by race and sex: United States, 1999-2015. https://www.cdc.gov/nchs/nvss/mortality/lcwk1.htm. Published October 23, 2017. Accessed May 2, 2019.
9. Merikangas KR, He JP, Burstein M, et al. Lifetime prevalence of mental disorders in US adolescents: results from the National Comorbidity Survey Replication-Adolescent Supplement (NCS-A). J Am Acad Child Adolesc Psychiatry. 2010;49(10):980-989.
10. Wittchen HU, Nelson CB, Lachner G. Prevalence of mental disorders and psychosocial impairments in adolescents and young adults. Psychol Med. 1998;28(1):109-126.
11. Foley DL, Goldston DB, Costello EJ, et al. Proximal psychiatric risk factors for suicidality in youth: the Great Smoky Mountains Study. Arch Gen Psychiatry. 2006;63(9):1017-1024.
12. Cheung A, Sacks D, Dewa CS, et al. Pediatric prescribing practices and the FDA black-box warning on antidepressants. J Dev Behav Pediatr. 2008 29(3):213-215.
13. Walkup JT. Antidepressant efficacy for depression in children and adolescents: industry- and NIMH-funded studies. Am J Psychiatry. 2017;174(5):430-437.
14. Jakubovski E, Varigonda AL, Freemantle N, et al. Systematic review and meta-analysis: dose-response relationship of selective serotonin reuptake inhibitors in major depressive disorder. Am J Psychiatry. 2016;173(2):174-183.
15. Varigonda AL, Jakubovski E, Taylor MJ, et al. Systematic review and meta-analysis: early treatment responses of selective serotonin reuptake inhibitors in pediatric major depressive disorder. J Am Acad Child Adolesc Psychiatry. 2015;54(7):557-564.
16. Strawn JR, Welge JA, Wehry AM, et al. Efficacy and tolerability of antidepressants in pediatric anxiety disorders: a systematic review and meta-analysis. Depress Anxiety. 2015;32(3):149-157.
17. March JS, Biederman J, Wolkow R, et al. Sertraline in children and adolescents with obsessive-compulsive disorder: a multicenter randomized controlled trial. JAMA. 1998;280(20):1752-1756.
18. Walkup JT, Albano AM, Piacentini J, et al. Cognitive behavioral therapy, sertraline, or a combination in childhood anxiety. N Engl J Med. 2008;359(26):2753-2766.
19. Kennard BD, Silva SG, Tonev S, et al. Remission and recovery in the Treatment for Adolescents with Depression Study (TADS): acute and long-term outcomes. J Am Acad Child Adolesc Psychiatry. 2009;48(2):186-195.
20. Cipriani A, Zhou X, Del Giovane C, et al. Comparative efficacy and tolerability of antidepressants for major depressive disorder in children and adolescents: a network meta-analysis. Lancet. 2016;388(10047):881-890.
21. Cohen JA, Mannarino AP, Perel JM, et al. A pilot randomized controlled trial of combined trauma-focused CBT and sertraline for childhood PTSD symptoms. J Am Acad Child Adolesc Psychiatry. 2007;46(7):811-819.
22. Robb AS, Cueva JE, Sporn J, et al. Sertraline treatment of children and adolescents with posttraumatic stress disorder: a double-blind, placebo-controlled trial. J Child Adolesc Psychopharmacol. 2010;20(6):463-471.
23. Diehle J, Opmeer BC, Boer F, et al. Trauma-focused cognitive behavioral therapy or eye movement desensitization and reprocessing: what works in children with posttraumatic stress symptoms? A randomized controlled trial. Eur Child Adolesc Psychiatry. 2015;24(2):227-236.
24. Aiyer R, Barkin RL, Bhatia A. Treatment of neuropathic pain with venlafaxine: a systematic review. Pain Med. 2017;18(10):1999-2012.
25. Barrickman LL, Perry PJ, Allen AJ, et al. Bupropion versus methylphenidate in the treatment of attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 1995;34(5):649-657.
26. Monuteaux MC, Spencer TJ, Faraone SV, et al. A randomized, placebo-controlled clinical trial of bupropion for the prevention of smoking in children and adolescents with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2007;68(7):1094-1101.
27. Biederman J, Baldessarini RJ, Wright V, et al. A double-blind placebo controlled study of desipramine in the treatment of ADD: I. Efficacy. J Am Acad Child Adolesc Psychiatry. 1989;28(5):777-784.
28. Spencer T, Biederman J, Coffey B, et al. A double-blind comparison of desipramine and placebo in children and adolescents with chronic tic disorder and comorbid attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2002;59(7):649-656.
29. DeVeaugh-Geiss J, Moroz G, Biederman J, et al. Clomipramine hydrochloride in childhood and adolescent obsessive-compulsive disorder--a multicenter trial. J Am Acad Child Adolesc Psychiatry. 1992;31(1):45-49.
30. Caldwell PH, Sureshkumar P, Wong WC. Tricyclic and related drugs for nocturnal enuresis in children. Cochrane Database Syst Rev. 2016;(1):CD002117.
31. Varigonda AL, Jakubovski E, Bloch MH. Systematic review and meta-analysis: early treatment responses of selective serotonin reuptake inhibitors and clomipramine in pediatric obsessive-compulsive disorder. J Am Acad Child Adolesc Psychiatry. 2016;55(10):851-859.e2. doi: 10.1016/j.jaac.2016.07.768.
32. Walkup J, Labellarte M. Complications of SSRI treatment. J Child Adolesc Psychopharmacol. 2001;11(1):1-4.
33. Leo RJ, Lichter DG, Hershey LA. Parkinsonism associated with fluoxetine and cimetidine: a case report. J Geriatr Psychiatry Neurol. 1995;8(4):231-233.
34. Strawn JR, Prakash A, Zhang Q, et al. A randomized, placebo-controlled study of duloxetine for the treatment of children and adolescents with generalized anxiety disorder. J Am Acad Child Adolesc Psychiatry. 2015;54(4):283-293.
35. Bernstein GA, Borchardt CM, Perwien AR, et al. Imipramine plus cognitive-behavioral therapy in the treatment of school refusal. J Am Acad Child Adolesc Psychiatry. 2000;39(3): 276-283.
36. Safer DJ, Zito JM. Treatment-emergent adverse events from selective serotonin reuptake inhibitors by age group: children versus adolescents. J Child Adolesc Psychopharmacol. 2006;16(1-2):159-169.
37. Reinblatt SP, DosReis S, Walkup JT, et al. Activation adverse events induced by the selective serotonin reuptake inhibitor fluvoxamine in children and adolescents. J Child Adolesc Psychopharmacol. 2009;19(2):119-126.
38. Goldsmith M, Singh M, Chang K. Antidepressants and psychostimulants in pediatric populations: is there an association with mania? Paediatr Drugs. 2011;13(4): 225-243.
39. Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156-167.
40. Allain N, Leven C, Falissard B, et al. Manic switches induced by antidepressants: an umbrella review comparing randomized controlled trials and observational studies. Acta Psychiatr Scand. 2017;135(2):106-116.
41. McClellan J, Kowatch R, Findling RL. Practice parameter for the assessment and treatment of children and adolescents with bipolar disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(1):107-125.
42. Dobry Y, Rice T, Sher L. Ecstasy use and serotonin syndrome: a neglected danger to adolescents and young adults prescribed selective serotonin reuptake inhibitors. Int J Adolesc Med Health. 2013; 25(3):193-199.
43. Schwartz AR, Pizon AF, Brooks DE. Dextromethorphan-induced serotonin syndrome. Clin Toxicol (Phila). 2008;46(8):771-773.
44. Gibbons RD, Brown CH, Hur K, et al. Early evidence on the effects of regulators’ suicidality warnings on SSRI prescriptions and suicide in children and adolescents. Am J Psychiatry. 2007;164(9):1356-1363.
45. Hammad TA, Laughren T, Racoosin J. Suicidality in pediatric patients treated with antidepressant drugs. Arch Gen Psychiatry. 2006;63(3):332-339.
46. Bridge JA, Iyengar S, Salary CB, et al. Clinical response and risk for reported suicidal ideation and suicide attempts in pediatric antidepressant treatment: a meta-analysis of randomized controlled trials. JAMA. 2007;297(15):1683-1696.
47. Sharma T, Guski LS, Freund N, et al. Suicidality and aggression during antidepressant treatment: systematic review and meta-analyses based on clinical study reports. BMJ. 2016;352: i65. doi: https://doi.org/10.1136/bmj.i65.
48. Olfson M, Shaffer D, Marcus SC, et al. Relationship between antidepressant medication treatment and suicide in adolescents. Arch Gen Psychiatry. 2003;60(10):978-982.
49. Garland JE, Kutcher S, Virani A, et al. Update on the Use of SSRIs and SNRIs with children and adolescents in clinical practice. J Can Acad Child Adolesc Psychiatry. 2016;25(1):4-10.
50. Bridge JA, Barbe RP, Birmaher B, et al. Emergent suicidality in a clinical psychotherapy trial for adolescent depression. Am J Psychiatry. 2005;162(11):2173-2175.
51. Birmaher B, Brent D, Bernet W, et al. Practice parameter for the assessment and treatment of children and adolescents with depressive disorders. J Am Acad Child Adolesc Psychiatry. 2007;46(11):1503-1526.
52. Ravizza L, Maina G, Bogetto F, et al. Long term treatment of obsessive-compulsive disorder. CNS Drugs. 1998;10(4):247-255.
53. Hosenbocus S, Chahal R. SSRIs and SNRIs: a review of the discontinuation syndrome in children and adolescents. J Can Acad Child Adolesc Psychiatry. 2011;20(1):60-67.
54. Bloch MH, McGuire J, Landeros-Weisenberger A, et al. Meta-analysis of the dose-response relationship of SSRI in obsessive-compulsive disorder. Mol Psychiatry. 2010;15(8):850-855.
55. Issari Y, Jakubovski E, Bartley CA, et al. Early onset of response with selective serotonin reuptake inhibitors in obsessive-compulsive disorder: a meta-analysis. J Clin Psychiatry. 2016; 77(5):e605-e611. doi: 10.4088/JCP.14r09758.
56. Brent D, Emslie G, Clarke G, et al. Switching to another SSRI or to venlafaxine with or without cognitive behavioral therapy for adolescents with SSRI-resistant depression: the TORDIA randomized controlled trial. JAMA. 2008;299(8):901-913.
Major depressive disorder (MDD) is a significant pediatric health problem, with a lifetime prevalence as high as 20% by the end of adolescence.1-3 Major depressive disorder in adolescence is associated with significant morbidity, including poor social functioning, school difficulties, early pregnancy, and increased risk of physical illness and substance abuse.4-6 It is also linked with significant mortality, with increased risk for suicide, which is now the second leading cause of death in individuals age 10 to 24 years.1,7,8
As their name suggests, antidepressants comprise a group of medications that are used to treat MDD; they are also, however, first-line agents for generalized anxiety disorder (GAD), posttraumatic stress disorder (PTSD), and obsessive-compulsive disorder (OCD) in adults. Anxiety disorders (including GAD and other anxiety diagnoses) and PTSD are also common in childhood and adolescence with a combined lifetime prevalence ranging from 15% to 30%.9,10 These disorders are also associated with increased risk of suicide.11 For all of these disorders, depending on the severity of presentation and the preference of the patient, treatments are often a combination of psychotherapy and psychopharmacology.
Clinicians face several challenges when considering antidepressants for pediatric patients. Pediatricians and psychiatrists need to understand whether these medications work in children and adolescents, and whether there are unique developmental safety and tolerability issues. The evidence base in child psychiatry is considerably smaller compared with that of adult psychiatry. From this more limited evidence base also came the controversial “black-box” warning regarding a risk of emergent suicidality when starting antidepressants that accompanies all antidepressants for pediatric, but not adult, patients. This warning has had major effects on clinical encounters with children experiencing depression, including altering clinician prescribing behavior.12
Do antidepressants work in children?
Selective serotonin reuptake inhibitors. Selective serotonin reuptake inhibitors (SSRIs) are the most commonly used class of antidepressants in both children and adults.13 While only a few SSRIs are FDA-approved for pediatric indications, the lack of FDA approval is typically related to a lack of sufficient testing in randomized controlled trials (RCTs) for specific pediatric indications, rather than to demonstrable differences in efficacy between antidepressant agents. Since there is currently no data to suggest inferiority of one agent compared to another in children or adults,14,15 efficacy data will be discussed here as applied to the class of SSRIs, generalizing from RCTs conducted on individual drugs. Table 1 lists FDA indications and dosing information for individual antidepressants.

There is strong evidence that SSRIs are effective for treating pediatric anxiety disorders (eg, social anxiety disorder and GAD)16 and OCD,17 with numbers needed to treat (NNT) between 3 and 5. For both of these disorders, SSRIs combined with cognitive-behavioral therapy (CBT) have the highest likelihood of improving symptoms or achieving remission.17,18
Selective serotonin reuptake inhibitors are also effective for treating pediatric MDD; however, the literature is more complex for this disorder compared to GAD and OCD as there are considerable differences in effect sizes between National Institute of Mental Health (NIMH)–funded studies and industry-sponsored trials.13 The major NIMH-sponsored adolescent depression trial, TADS (Treatment for Adolescents and Depression Study), showed that SSRIs (fluoxetine in this case) were quite effective, with an NNT of 4 over the acute phase (12 weeks).19 Ultimately, approximately 80% of adolescents improved over 9 months. Many industry-sponsored trials for MDD in pediatric patients had large placebo response rates (approximately 60%), which resulted in smaller between-group differences, and estimates of an NNT closer to 12,13 which has muddied the waters in meta-analyses that include all trials.20 Improvement in depressive symptoms also appears to be bolstered by concomitant CBT in MDD,19 but not as robustly as in GAD and OCD. While the full benefit of SSRIs for depression may take as long as 8 weeks, a meta-analysis of depression studies of pediatric patients suggests that significant benefits from placebo are observed as early as 2 weeks, and that further treatment gains are minimal after 4 weeks.15 Thus, we recommend at least a 4- to 6-week trial at therapeutic dosing before deeming a medication a treatment failure.
Continue to: Posttraumatic stress disorder...
Posttraumatic stress disorder is a fourth disorder in which SSRIs are a first-line treatment in adults. The data for using SSRIs to treat pediatric patients with PTSD is scant, with only a few RCTs, and no large NIMH-funded trials. Randomized controlled trials have not demonstrated significant differences between SSRIs and placebo21,22 and thus the current first-line recommendation in pediatric PTSD remains trauma-focused therapy, with good evidence for trauma-focused CBT.23 Practically speaking, there can be considerable overlap of PTSD, depression, and anxiety symptoms in children,23 and children with a history of trauma who also have comorbid MDD may benefit from medication if their symptoms persist despite an adequate trial of psychotherapy.
Taken together, the current evidence suggests that SSRIs are often effective in pediatric GAD, OCD, and MDD, with low NNTs (ranging from 3 to 5 based on NIMH-funded trials) for all of these disorders; there is not yet sufficient evidence of efficacy in pediatric patients with PTSD.
Fluoxetine has been studied more intensively than other SSRIs (for example, it was the antidepressant used in the TADS trial), and thus has the largest evidence base. For this reason, fluoxetine is often considered the first of the first-line options. Additionally, fluoxetine has a longer half-life than other antidepressants, which may make it more effective in situations where patients are likely to miss doses, and results in a lower risk of withdrawal symptoms when stopped due to “self-tapering.”
SNRIs and atypical antidepressants. Other antidepressants commonly used in pediatric patients but with far less evidence of efficacy include serotonin-norepinephrine reuptake inhibitors (SNRIs) and the atypical antidepressants bupropion and mirtazapine. The SNRI duloxetine is FDA-approved for treating GAD in children age 7 to 17, but there are no other pediatric indications for duloxetine, or for the other SNRIs.
In general, adverse effect profiles are worse for SNRIs compared to SSRIs, further limiting their utility. While there are no pediatric studies demonstrating SNRI efficacy for neuropathic pain, good data exists in adults.24 Thus, an SNRI could be a reasonable option if a pediatric patient has failed prior adequate SSRI trials and also has comorbid neuropathic pain.
Continue to: Neither bupropion nor mirtazapine...
Neither bupropion nor mirtazapine have undergone rigorous testing in pediatric patients, and therefore these agents should generally be considered only once other first-line treatments have failed. Bupropion has been evaluated for attention-deficit/hyperactivity disorder (ADHD)25 and for adolescent smoking cessation.26 However, the evidence is weak, and bupropion is not considered a first-line option for children and adolescents.
Tricyclic antidepressants. Randomized controlled trials have demonstrated that tricyclic antidepressants (TCAs) are efficacious for treating several pediatric conditions; however, their significant side effect profile, their monitoring requirements, as well as their lethality in overdose has left them replaced by SSRIs in most cases. That said, they can be appropriate in refractory ADHD (desipramine27,28) and refractory OCD (clomipramine is FDA-approved for this indication29); they are considered a third-line treatment for enuresis.30
Why did my patient stop the medication?
Common adverse effects. Although the greatest benefit of antidepressant medications compared with placebo is achieved relatively early on in treatment, it generally takes time for these benefits to accrue and become clinically apparent.15,31 By contrast, most adverse effects of antidepressants present and are at their most severe early in treatment. The combination of early adverse effects and delayed efficacy leads many patients, families, and clinicians to discontinue medications before they have an adequate chance to work. Thus, it is imperative to provide psychoeducation before starting a medication about the typical time-course of improvement and adverse effects (Table 2).

Adverse effects of SSRIs often appear or worsen transiently during initiation of a medication, during a dose increase,32 or, theoretically, with the addition of a medication that interferes with SSRI metabolism (eg, cimetidine inhibition of cytochrome P450 2D6).33 If families are prepared for this phenomenon and the therapeutic alliance is adequate, adverse effects can be tolerated to allow for a full medication trial. Common adverse effects of SSRIs include sleep problems (insomnia/sedation), gastrointestinal upset, sexual dysfunction, dry mouth, and hyperhidrosis. Although SSRIs differ somewhat in the frequency of these effects, as a class, they are more similar than different. Adequate psychoeducation is especially imperative in the treatment of OCD and anxiety disorders, where there is limited evidence of efficacy for any non-serotonergic antidepressants.
Serotonin-norepinephrine reuptake inhibitors are not considered first-line medications because of the reduced evidence base compared to SSRIs and their enhanced adverse effect profiles. Because SNRIs partially share a mechanism of action with SSRIs, they also share portions of the adverse effects profile. However, SNRIs have the additional adverse effect of hypertension, which is related to their noradrenergic activity. Thus, it is reasonable to obtain a baseline blood pressure before initiating an SNRI, as well as periodically after initiation and during dose increases, particularly if the patient has other risk factors for hypertension.34
Continue to: Although TCAs have efficacy...
Although TCAs have efficacy in some pediatric disorders,27-29,35 their adverse effect profile limits their use. Tricyclic antidepressants are highly anticholinergic (causing dizziness secondary to orthostatic hypotension, dry mouth, and urinary retention) and antihistaminergic (causing sedation and weight gain). Additionally, TCAs lower the seizure threshold and have adverse cardiac effects relating to their anti-alpha-1 adrenergic activity, resulting in dose-dependent increases in the QTc and cardiac toxicity in overdose that could lead to arrhythmia and death. These medications have their place, but their use requires careful informed consent, clear treatment goals, and baseline and periodic cardiac monitoring (via electrocardiogram).
Serious adverse effects. Clinicians may be hesitant to prescribe antidepressants for pediatric patients because of the potential for more serious adverse effects, including severe behavioral activation syndromes, serotonin syndrome, and emergent suicidality. However, current FDA-approved antidepressants arguably have one of the most positive risk/benefit profiles of any orally-administered medication approved for pediatric patients. Having a strong understanding of the evidence is critical to evaluating when it is appropriate to prescribe an antidepressant, how to properly monitor the patient, and how to obtain accurate informed consent.
Pediatric behavioral activation syndrome. Many clinicians report that children receiving antidepressants experience a pediatric behavioral activation syndrome, which exists along a spectrum from mild activation, increased energy, insomnia, or irritability up through more severe presentations of agitation, hyperactivity, or possibly mania. A recent meta-analysis suggested a positive association between antidepressant use and activation events on the milder end of this spectrum in pediatric patients with non-OCD anxiety disorders,16 and it is thought that compared with adolescents, younger children are more susceptible to activation adverse effects.36 The likelihood of activation events has been associated with higher antidepressant plasma levels,37 suggesting that dose or individual differences in metabolism may play a role. At the severe end of the spectrum, the risk of induction of mania in pediatric patients with depression or anxiety is relatively rare (<2%) and not statistically different from placebo in RCTs of pediatric participants.38 Meta-analyses of larger randomized, placebo-controlled trials of adults do not support the idea that SSRIs and other second-generation antidepressants carry an increased risk of mania compared with placebo.39,40 Children or adolescents with bona fide bipolar disorder (ie, patients who have had observed mania that meets all DSM-5 criteria) should be treated with a mood-stabilizing agent or antipsychotic if prescribed an antidepressant.41 These clear-cut cases are, however, relatively rare, and more often clinicians are confronted with ambiguous cases that include a family history of bipolar disorder along with “softer” symptoms of irritability, intrusiveness, or aggression. In these children, SSRIs may be appropriate for depressive, OCD, or anxiety symptoms, and should be strongly considered before prescribing antipsychotics or mood stabilizers, as long as initiated with proper monitoring.
Serotonin syndrome is a life-threatening condition caused by excess synaptic serotonin. It is characterized by confusion, sweating, diarrhea, hypertension, hyperthermia, and tachycardia. At its most severe, serotonin syndrome can result in seizures, arrhythmias, and death. The risk of serotonin syndrome is very low when using an SSRI as monotherapy. Risk increases with polypharmacy, particularly unexamined polypharmacy when multiple serotonergic agents are inadvertently on board. Commonly used serotonergic agents include other antidepressants, migraine medications (eg, triptans), some pain medications, and the cough suppressant dextromethorphan.
The easiest way to mitigate the risk of serotonin syndrome is to use an interaction index computer program, which can help ensure that the interacting agents are not prescribed without first discussing the risks and benefits. It is important to teach adolescents that certain recreational drugs are highly serotonergic and can cause serious interactions with antidepressants. For example, recreational use of dextromethorphan or 3,4-methylenedioxymethamphetamine (MDMA; commonly known as “ecstasy”) has been associated with serotonin syndrome in adolescents taking antidepressant medications.42,43
Continue to: Suicidality
Suicidality. The black-box warning regarding a risk of emergent suicidality when starting antidepressant treatment in children is controversial.44 The prospect that a medication intended to ameliorate depression might instead risk increasing suicidal thinking is alarming to parents and clinicians alike. To appropriately weigh and discuss the risks and benefits with families, it is important to understand the data upon which the warning is based.

In 2004, the FDA commissioned a review of 23 antidepressant trials, both published and unpublished, pooling studies across multiple indications (MDD, OCD, anxiety, and ADHD) and multiple antidepressant classes. This meta-analysis, which included nearly 4,400 pediatric patients, found a small but statistically significant increase in spontaneously-reported suicidal thoughts or actions, with a risk difference of 1% (95% confidence interval [CI], 1% to 2%).45 These data suggest that if one treats 100 pediatric patients, 1 to 2 of them may experience short-term increases in suicidal thinking or behavior.45 There were no differences in suicidal thinking when assessed systematically (ie, when all subjects reported symptoms of suicidal ideation on structured rating scales), and there were no completed suicides.45 A subsequent analysis that included 27 pediatric trials suggested an even lower, although still significant, risk difference (<1%), yielding a number needed to harm (NNH) of 143.46 Thus, with low NNT for efficacy (3 to 6) and relatively high NNH for emergent suicidal thoughts or behaviors (100 to 143), for many patients the benefits will outweigh the risks.

Figure 1, Figure 2, and Figure 3 are Cates plots that depict the absolute benefits of antidepressants compared with the risk of suicidality for pediatric patients with MDD, OCD, and anxiety disorders. Recent meta-analyses have suggested that the increased risk of suicidality in antidepressant trials is specific to studies that included children and adolescents, and is not observed in adult studies. A meta-analysis of 70 trials involving 18,526 participants suggested that the odds ratio of suicidality in trials of children and adolescents was 2.39 (95% CI, 1.31 to 4.33) compared with 0.81 (95% CI, 0.51 to 1.28) in adults.47 Additionally, a network meta-analysis exclusively focusing on pediatric antidepressant trials in MDD reported significantly higher suicidality-related adverse events in venlafaxine trials compared with placebo, duloxetine, and several SSRIs (fluoxetine, paroxetine, and escitalopram).20 These data should be interpreted with caution as differences in suicidality detected between agents is quite possibly related to differences in the method of assessment between trials, as opposed to actual differences in risk between agents.

Epidemiologic data further support the use of antidepressants in pediatric patients, showing that antidepressant use is associated with decreased teen suicide attempts and completions,48 and the decline in prescriptions that occurred following the black-box warning was accompanied by a 14% increase in teen suicides.49 Multiple hypotheses have been proposed to explain the pediatric clinical trial findings. One idea is that potential adverse effects of activation, or the intended effects of restoring the motivation, energy, and social engagement that is often impaired in depression, increases the likelihood of thinking about suicide or acting on thoughts. Another theory is that reporting of suicidality may be increased, rather than increased de novo suicidality itself. Antidepressants are effective for treating pediatric anxiety disorders, including social anxiety disorder,16 which could result in more willingness to report. Also, the manner in which adverse effects are generally ascertained in trials might have led to increased spontaneous reporting. In many trials, investigators ask whether participants have any adverse effects in general, and inquire about specific adverse effects only if the family answers affirmatively. Thus, the increased rate of other adverse effects associated with antidepressants (sleep problems, gastrointestinal upset, dry mouth, etc.) might trigger a specific question regarding suicidal ideation, which the child or family then may be more likely to report. Alternatively, any type of psychiatric treatment could increase an individual’s propensity to report; in adolescent psychotherapy trials, non-medic
Continue to: How long should the antidepressant be continued?
How long should the antidepressant be continued?
Many patients are concerned about how long they may be taking medication, and whether they will be taking an antidepressant “forever.” A treatment course can be broken into an acute phase, wherein remission is achieved and maintained for 6 to 8 weeks. This is followed by a continuation phase, with the goal of relapse prevention, lasting 16 to 20 weeks. The length of the last phase—the maintenance phase—depends both on the child’s history, the underlying therapeutic indication, the adverse effect burden experienced, and the family’s preferences/values. In general, for a first depressive episode, after treating for 1 year, a trial of discontinuation can be attempted with close monitoring. For a second depressive episode, we recommend 2 years of remission on antidepressant therapy before attempting discontinuation. In patients who have had 3 depressive episodes, or have had episodes of high severity, we recommend continuing antidepressant treatment indefinitely. Although much less well studied, the risk of relapse following SSRI discontinuation appears much more significant in OCD, whereas anxiety disorders and MDD have a relatively comparable risk.52
In general, stopping an antidepressant should be done carefully and slowly.
What to do when first-line treatments fail
Adequate psychotherapy? To determine whether children are receiving adequate CBT, ask:
- if the child receives homework from psychotherapy
- if the parents are included in treatment
- if therapy has involved identifying thought patterns that may be contributing to the child’s illness, and
- if the therapist has ever exposed the child to a challenge likely to produce anxiety or distress in a supervised environment and has developed an exposure hierarchy (for conditions with primarily exposure-based therapies, such as OCD or anxiety disorders).
If a family is not receiving most of these elements in psychotherapy, this is a good indicator that they may not be receiving evidence-based CBT.
Continue to: Adequate pharmacotherapy?
Adequate pharmacotherapy? Similarly, when determining the adequacy of previous pharmacotherapy, it is critical to determine whether the child received an adequate dose of medications (at least the FDA-recommended minimum dose) for an adequate duration of time at therapeutic dosing (at least 6 weeks for MDD, 8 weeks for anxiety disorders, and 8 to 12 weeks for pediatric patients with OCD), and that the child actually took the medication regularly during that period. Patient compliance can typically be tracked through checking refill requests or intervals through the patient’s pharmacy. Ensuring proper delivery of first-line treatments is imperative because (1) the adverse effects associated with second-line treatments are often more substantial; (2) the cost in terms of time and money is considerably higher with second-line treatments, and; (3) the evidence regarding the benefits of these treatments is much less certain.
Inadequate dosing is a common reason for non-response in pediatric patients. Therapeutic dose ranges for common antidepressants are displayed in Table 1. Many clinicians underdose antidepressants for pediatric patients initially (and often throughout treatment) because they fear that the typical dose titration used in clinical trials will increase the risk of adverse effects compared with more conservative dosing. There is limited evidence to suggest that this underdosing strategy is likely to be successful; adverse effects attributable to these medications are modest, and most that are experienced early in treatment (eg, headache, increased anxiety or irritability, sleep problems, gastrointestinal upset) are self-limiting and may be coincidental rather than medication-induced. Furthermore, there is no evidence for efficacy of subtherapeutic dosing in children in the acute phase of treatment or for preventing relapse.14 Thus, from an efficacy standpoint, a medication trial has not officially begun until the therapeutic dose range is reached.
Once dosing is within the therapeutic range, however, pediatric data differs from the adult literature. In most adult psychiatric conditions, higher doses of SSRIs within the therapeutic range are associated with an increased response rate.14,54 In pediatrics, there are few fixed dose trials, and once within the recommended therapeutic range, minimal data supports an association between higher dosing and higher efficacy.14 In general, pediatric guidelines are silent regarding optimal dosing of SSRIs within the recommended dose range, and higher antidepressant doses often result in a more significant adverse effect burden for children. One exception is pediatric OCD, where, similar to adults, the guidelines suggest that higher dosing of SSRIs often is required to induce a therapeutic response as compared to MDD and GAD.31,55
If a child does not respond to adequate first-line treatment (or has a treatment history that cannot be fully verified), repeating these first-line interventions carries little risk and can be quite effective. For example, 60% of adolescents with MDD who did not initially respond to an SSRI demonstrated a significant response when prescribed a second SSRI or venlafaxine (with or without CBT).56
When pediatric patients continue to experience significantly distressing and/or debilitating symptoms (particularly in MDD) despite multiple trials of antidepressants and psychotherapy, practitioners should consider a careful referral to interventional psychiatry services, which can include the more intensive treatments of electroconvulsive therapy, repetitive transcranial magnetic stimulation, or ketamine (see Box 1). Given the substantial morbidity and mortality associated with adolescent depression, interventional psychiatry treatments are under-researched and under-utilized clinically in pediatric populations.
Continue to: Antidepressants in general...
Antidepressants in general, and SSRIs in particular, are the first-line pharmacotherapy for pediatric anxiety, OCD, and MDD. For PTSD, although they are a first-line treatment in adults, their efficacy has not been demonstrated in children and adolescents. Antidepressants are generally safe, well-tolerated, and effective, with low NNTs (3 to 5 for anxiety and OCD; 4 to 12 in MDD, depending on whether industry trials are included). It is important that clinicians and families be educated about possible adverse effects and their time course in order to anticipate difficulties, ensure adequate informed consent, and monitor appropriately. The black-box warning regarding treatment-emergent suicidal thoughts or behaviors must be discussed (for suggested talking points, see Box 2). The NNH is large (100 to 143), and for many patients, the benefits will outweigh the risks. For pediatric patients who fail to respond to multiple adequate trials of antidepressants and psychotherapy, referrals for interventional psychiatry consultation should be considered.
Bottom Line
Although the evidence base for prescribing antidepressants for children and adolescents is smaller compared to the adult literature, properly understanding and prescribing these agents, and explaining their risks and benefits to families, can make a major difference in patient compliance, satisfaction, and outcomes. Antidepressants (particularly selective serotonin reuptake inhibitors) are the firstline pharmacologic intervention for pediatric patients with anxiety disorders, obsessive-compulsive disorder, or major depressive disorder.
Related Resource
- Bonin L, Moreland CS. Overview of prevention and treatment for pediatric depression. https://www.uptodate.com/contents/overview-of-prevention-and-treatment-for-pediatric-depression. Last reviewed July 2019.
Drug Brand Names
Bupropion • Wellbutrin, Zyban
Cimetidine • Tagamet
Citalopram • Celexa
Clomipramine • Anafranil
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Fluvoxamine • Luvox
Imipramine • Tofranil
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Venlafaxine • Effexor
Vilazodone • Viibryd
Vortioxetine • Trintellix
Box 1
Continuing severe depression is associated with reduced educational attainment and quality of life, as well as increased risk of substance abuse and suicide,1,2 which is the second leading cause of death in individuals age 10 to 24 years.3 Given the substantial morbidity and mortality associated with adolescent depression, interventional psychiatry treatments are under-researched and underutilized in pediatric patients. Interventional antidepressants in adults include electroconvulsive therapy (ECT), repetitive transcranial magnetic stimulation (rTMS), and, most recently, ketamine.
Electroconvulsive therapy is the most effective therapy available for depression in adults, alleviating depressive symptoms in treatment-refractory patients and outperforming both pharmacotherapy4 and rTMS.5 Despite its track record of effectiveness and safety in adults, ECT continues to suffer considerable stigma.4 Cognitive adverse effects and memory problems in adults are generally self-limited, and some aspects of cognition actually improve after ECT as depression lifts.6 The combination of stigma and the concern about possible cognitive adverse effects during periods of brain development have likely impeded the rigorous testing of ECT in treatment-refractory pediatric patients. Several case series and other retrospective analyses suggest, however, that ECT has strong efficacy and limited adverse effects in adolescents who have severe depression or psychotic symptoms.7-9 Despite these positive preliminary data in pediatric patients, and a large body of literature in adults, no controlled trials of ECT have been conducted in the pediatric population, and it remains a rarely used treatment in severe pediatric mental illness.
Repetitive transcranial magnetic stimulation is a technique in which magnetic stimulation is used to activate the left dorsolateral prefrontal cortex (DLPFC), a target thought to be important in the pathophysiology of MDD. Repetitive transcranial magnetic stimulation is FDAapproved to treat medication-refractory major depressive disorder (MDD) in adults, and has been shown to be effective as both a monotherapy10 and an adjunctive treatment.11 The estimated number needed to treat (NNT) for rTMS ranges from 6 to 8, which is quite effective, although less so than ECT (and probably initial pharmacotherapy).5 Similar to ECT, however, there are no large randomized controlled trials (RCTs) in children or adolescents. Pilot RCTs12 and open trials13 suggest that DLPFC rTMS may be effective as an adjunctive treatment, speeding or augmenting response to a selective serotonin reuptake inhibitor in adolescents with MDD. Larger trials studying rTMS in pediatric patients are needed. While rTMS is generally well tolerated, disadvantages include the time-consuming schedule (the initial treatment is typically 5 days/week for several weeks) and local adverse effects of headache and scalp pain.
Ketamine, which traditionally is used as a dissociative anesthetic, is a rapidly emerging novel treatment in adult treatment-refractory MDD. It acts quickly (within hours to days) and cause significant improvement in difficult symptoms such as anhedonia14 and suicidal ideation.15 In adult studies, ketamine has a robust average effect size of >1.2, and an NNT ranging from 3 to 5 in medication-refractory patients.16,17 Ketamine is a glutamatergic modulator, acting outside of the monoamine neurochemical systems traditionally targeted by standard antidepressants.16 The efficacy of ketamine in treatment-refractory adults is impressive, but the effects of a single treatment are ephemeral, dissipating within 1 to 2 weeks, which has led to significant discussion surrounding optimal dosing strategies.16 Although small RCTs in pediatric patients are currently underway, at this time, the only evidence for ketamine for pediatric MDD is based on case series/report data18,19 which was positive.
For all of these interventional modalities, it is critical to refer children with treatmentrefractory disorders to interventionists who have appropriate experience and monitoring capabilities.
References
1. Weissman MM, Wolk S, Goldstein RB, et al. Depressed adolescents grown up. JAMA.1999;281(18):1707-1713.
2. Fergusson DM, Woodward LJ. Mental health, educational, and social role outcomes of adolescents with depression. Arch Gen Psychiatry. 2002;59(3):225-231.
3. Centers for Disease Control and Prevention. National Vital Statistics System. Deaths, percent of total deaths, and death rates for the 15 leading causes of death in 5-year age groups, by race and sex: United States, 1999-2015. Centers for Disease Control and Prevention. https://www.cdc.gov/nchs/nvss/mortality/lcwk1.htm. Published October 23, 2017. Accessed May 2, 2019.
4. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and metaanalysis. Lancet. 2003;361(9360):799-808.
5. Berlim MT, Van den Eynde F, Daskalakis ZJ. Efficacy and acceptability of high frequency repetitive transcranial magnetic stimulation (rTMS) versus electroconvulsive therapy (ECT) for major depression: a systematic review and meta-analysis of randomized trials. Depress Anxiety. 2013;30(7):614-623.
6. Semkovska M, McLoughlin DM. Objective cognitive performance associated with electroconvulsive therapy for depression: a systematic review and meta-analysis. Biol Psychiatry. 2010;68(6):568-577.
7. Jacob P, Gogi PK, Srinath S, et al. Review of electroconvulsive therapy practice from a tertiary child and adolescent psychiatry centre. Asian J Psychiatr. 2014;12(1):95-99.
8. Zhand N, Courtney DB, Flament MF. Use of electroconvulsive therapy in adolescents with treatment-resistant depressive disorders: a case series. J ECT. 2015;31(4):238-245.
9. Puffer CC, Wall CA, Huxsahl JE, et al. A 20 year practice review of electroconvulsive therapy for adolescents. J Child Adolesc Psychopharmacol. 2016;26(7):632-636.
10. Berlim MT, van den Eynde F, Tovar-Perdomo S, et al. Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rTMS) for treating major depression: a systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol Med. 2014;44(2):225-239.
11. Liu B, Zhang Y, Zhang L, et al. Repetitive transcranial magnetic stimulation as an augmentative strategy for treatment-resistant depression, a meta-analysis of randomized, double-blind and sham-controlled study. BMC Psychiatry. 2014;14:342.
12. Huang ML, Luo BY, Hu JB, et al. Repetitive transcranial magnetic stimulation in combination with citalopram in young patients with first-episode major depressive disorder: a double-blind, randomized, sham-controlled trial. Aust N Z J Psychiatry. 2012;46(3):257-264.
13. Wall CA, Croarkin PE, Sim LA, et al. Adjunctive use of repetitive transcranial magnetic stimulation in depressed adolescents: a prospective, open pilot study. J Clin Psychiatry. 2011;72(9):1263-1269.
14. Lally N, Nugent AC, Luckenbaugh DA, et al. Anti-anhedonic effect of ketamine and its neural correlates in treatment-resistant bipolar depression. Transl Psychiatry. 2014;4:e469. doi: 10.1038/tp.2014.105.
15. Ballard ED, Ionescu DF, Vande Voort JL, et al. Improvement in suicidal ideation after ketamine infusion: relationship to reductions in depression and anxiety. J Psychiatr Res. 2014;58:161-166.
16. Newport DJ, Carpenter LL, McDonald WM, et al. Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry. 2015;172(10):950-966.
17. McGirr A, Berlim MT, Bond DJ, et al. A systematic review and meta-analysis of randomized, double-blind, placebo-controlled trials of ketamine in the rapid treatment of major depressive episodes. Psychol Med. 2015;45(4):693-704.
18. Dwyer JB, Beyer C, Wilkinson ST, et al. Ketamine as a treatment for adolescent depression: a case report. J Am Acad Child Adolesc Psychiatry. 2017;56(4):352-354.
19. Cullen KR, Amatya P, Roback MG, et al. Intravenous ketamine for adolescents with treatment-resistant depression: an open-label study. J Child Adolesc Psychopharmacol. 2018;28(7):437-444.
Box 2
Efficacy
- Selective serotonin reuptake inhibitors are the most effective pharmacologic treatment we have for pediatric depression, OCD, and anxiety
- More than one-half of children who are prescribed SSRIs have a significant improvement, regardless of condition
- Based on current estimates, we need to treat 4 to 6 children with an SSRI to find one that will improve who would not improve with placebo
- The clinical benefits of SSRIs generally take a while to accrue; therefore, it is advisable to take the medication for at least 2 to 3 months before concluding that it is ineffective
- In addition to medication, evidence-based psychotherapies provide significant benefit for pediatric depression, OCD, and anxiety
Tolerability
- Most commonly prescribed pediatric antidepressants have been used safely in children for 2 to 3 decades. The safety profiles of SSRIs are among the best of any medications used for children and adolescents
- While many children get better when taking these medications, it’s important that we also talk about potential adverse effects. Some children will experience sleep problems (either sleepier than usual or difficulty sleeping), changes in energy levels, headache, gastrointestinal upset, and dry mouth. These are most likely at the beginning of treatment, or when we increase the dose; they usually are time-limited and go away on their own
- Often adverse effects occur first and the benefits come later. Because it may take at least a few weeks to start to see the mood/anxiety benefits, it’s important for us to talk about any adverse effects your child experiences and remember that they usually are short-lived
Suicidality
- The FDA placed a “black-box” warning on antidepressants after pediatric studies found a small but statistically significant increased risk of reporting suicidal thoughts or behaviors over the short-term compared with placebo
- The increased risk of spontaneously reporting suicidal ideation was quite small. Studies suggested that one would need to treat 100 to 140 children to see 1 child report suicidal ideation compared to placebo. Suicidal ideation is a common symptom in children with depression and anxiety
- Studies found no increased risk when suicidal ideation was systematically assessed using structured rating scales
- In the studies evaluated, there were no completed suicides by patients taking medication or placebo
- Population studies show that higher rates of antidepressant prescriptions are associated with lower rates of attempted and completed teen suicide, which underscores that in general, these medicines treat the underlying causes of suicidality
- No scientific consensus exists on whether these medications are truly associated with an increased risk of new-onset suicidal ideation, or if this association is due to other factors (eg, improvement in anxiety and depressive symptoms that make patients more comfortable to report suicidal ideation spontaneously)
- Regardless, the FDA recommends frequent monitoring of children for suicidal thoughts when these medications are started. This should be done anyway in children experiencing depression and anxiety, and it’s why we will plan to have more frequent appointments as the medication is initiated
OCD: obsessive-compulsive disorder; SSRIs: selective serotonin reuptake inhibitors
Major depressive disorder (MDD) is a significant pediatric health problem, with a lifetime prevalence as high as 20% by the end of adolescence.1-3 Major depressive disorder in adolescence is associated with significant morbidity, including poor social functioning, school difficulties, early pregnancy, and increased risk of physical illness and substance abuse.4-6 It is also linked with significant mortality, with increased risk for suicide, which is now the second leading cause of death in individuals age 10 to 24 years.1,7,8
As their name suggests, antidepressants comprise a group of medications that are used to treat MDD; they are also, however, first-line agents for generalized anxiety disorder (GAD), posttraumatic stress disorder (PTSD), and obsessive-compulsive disorder (OCD) in adults. Anxiety disorders (including GAD and other anxiety diagnoses) and PTSD are also common in childhood and adolescence with a combined lifetime prevalence ranging from 15% to 30%.9,10 These disorders are also associated with increased risk of suicide.11 For all of these disorders, depending on the severity of presentation and the preference of the patient, treatments are often a combination of psychotherapy and psychopharmacology.
Clinicians face several challenges when considering antidepressants for pediatric patients. Pediatricians and psychiatrists need to understand whether these medications work in children and adolescents, and whether there are unique developmental safety and tolerability issues. The evidence base in child psychiatry is considerably smaller compared with that of adult psychiatry. From this more limited evidence base also came the controversial “black-box” warning regarding a risk of emergent suicidality when starting antidepressants that accompanies all antidepressants for pediatric, but not adult, patients. This warning has had major effects on clinical encounters with children experiencing depression, including altering clinician prescribing behavior.12
Do antidepressants work in children?
Selective serotonin reuptake inhibitors. Selective serotonin reuptake inhibitors (SSRIs) are the most commonly used class of antidepressants in both children and adults.13 While only a few SSRIs are FDA-approved for pediatric indications, the lack of FDA approval is typically related to a lack of sufficient testing in randomized controlled trials (RCTs) for specific pediatric indications, rather than to demonstrable differences in efficacy between antidepressant agents. Since there is currently no data to suggest inferiority of one agent compared to another in children or adults,14,15 efficacy data will be discussed here as applied to the class of SSRIs, generalizing from RCTs conducted on individual drugs. Table 1 lists FDA indications and dosing information for individual antidepressants.

There is strong evidence that SSRIs are effective for treating pediatric anxiety disorders (eg, social anxiety disorder and GAD)16 and OCD,17 with numbers needed to treat (NNT) between 3 and 5. For both of these disorders, SSRIs combined with cognitive-behavioral therapy (CBT) have the highest likelihood of improving symptoms or achieving remission.17,18
Selective serotonin reuptake inhibitors are also effective for treating pediatric MDD; however, the literature is more complex for this disorder compared to GAD and OCD as there are considerable differences in effect sizes between National Institute of Mental Health (NIMH)–funded studies and industry-sponsored trials.13 The major NIMH-sponsored adolescent depression trial, TADS (Treatment for Adolescents and Depression Study), showed that SSRIs (fluoxetine in this case) were quite effective, with an NNT of 4 over the acute phase (12 weeks).19 Ultimately, approximately 80% of adolescents improved over 9 months. Many industry-sponsored trials for MDD in pediatric patients had large placebo response rates (approximately 60%), which resulted in smaller between-group differences, and estimates of an NNT closer to 12,13 which has muddied the waters in meta-analyses that include all trials.20 Improvement in depressive symptoms also appears to be bolstered by concomitant CBT in MDD,19 but not as robustly as in GAD and OCD. While the full benefit of SSRIs for depression may take as long as 8 weeks, a meta-analysis of depression studies of pediatric patients suggests that significant benefits from placebo are observed as early as 2 weeks, and that further treatment gains are minimal after 4 weeks.15 Thus, we recommend at least a 4- to 6-week trial at therapeutic dosing before deeming a medication a treatment failure.
Continue to: Posttraumatic stress disorder...
Posttraumatic stress disorder is a fourth disorder in which SSRIs are a first-line treatment in adults. The data for using SSRIs to treat pediatric patients with PTSD is scant, with only a few RCTs, and no large NIMH-funded trials. Randomized controlled trials have not demonstrated significant differences between SSRIs and placebo21,22 and thus the current first-line recommendation in pediatric PTSD remains trauma-focused therapy, with good evidence for trauma-focused CBT.23 Practically speaking, there can be considerable overlap of PTSD, depression, and anxiety symptoms in children,23 and children with a history of trauma who also have comorbid MDD may benefit from medication if their symptoms persist despite an adequate trial of psychotherapy.
Taken together, the current evidence suggests that SSRIs are often effective in pediatric GAD, OCD, and MDD, with low NNTs (ranging from 3 to 5 based on NIMH-funded trials) for all of these disorders; there is not yet sufficient evidence of efficacy in pediatric patients with PTSD.
Fluoxetine has been studied more intensively than other SSRIs (for example, it was the antidepressant used in the TADS trial), and thus has the largest evidence base. For this reason, fluoxetine is often considered the first of the first-line options. Additionally, fluoxetine has a longer half-life than other antidepressants, which may make it more effective in situations where patients are likely to miss doses, and results in a lower risk of withdrawal symptoms when stopped due to “self-tapering.”
SNRIs and atypical antidepressants. Other antidepressants commonly used in pediatric patients but with far less evidence of efficacy include serotonin-norepinephrine reuptake inhibitors (SNRIs) and the atypical antidepressants bupropion and mirtazapine. The SNRI duloxetine is FDA-approved for treating GAD in children age 7 to 17, but there are no other pediatric indications for duloxetine, or for the other SNRIs.
In general, adverse effect profiles are worse for SNRIs compared to SSRIs, further limiting their utility. While there are no pediatric studies demonstrating SNRI efficacy for neuropathic pain, good data exists in adults.24 Thus, an SNRI could be a reasonable option if a pediatric patient has failed prior adequate SSRI trials and also has comorbid neuropathic pain.
Continue to: Neither bupropion nor mirtazapine...
Neither bupropion nor mirtazapine have undergone rigorous testing in pediatric patients, and therefore these agents should generally be considered only once other first-line treatments have failed. Bupropion has been evaluated for attention-deficit/hyperactivity disorder (ADHD)25 and for adolescent smoking cessation.26 However, the evidence is weak, and bupropion is not considered a first-line option for children and adolescents.
Tricyclic antidepressants. Randomized controlled trials have demonstrated that tricyclic antidepressants (TCAs) are efficacious for treating several pediatric conditions; however, their significant side effect profile, their monitoring requirements, as well as their lethality in overdose has left them replaced by SSRIs in most cases. That said, they can be appropriate in refractory ADHD (desipramine27,28) and refractory OCD (clomipramine is FDA-approved for this indication29); they are considered a third-line treatment for enuresis.30
Why did my patient stop the medication?
Common adverse effects. Although the greatest benefit of antidepressant medications compared with placebo is achieved relatively early on in treatment, it generally takes time for these benefits to accrue and become clinically apparent.15,31 By contrast, most adverse effects of antidepressants present and are at their most severe early in treatment. The combination of early adverse effects and delayed efficacy leads many patients, families, and clinicians to discontinue medications before they have an adequate chance to work. Thus, it is imperative to provide psychoeducation before starting a medication about the typical time-course of improvement and adverse effects (Table 2).

Adverse effects of SSRIs often appear or worsen transiently during initiation of a medication, during a dose increase,32 or, theoretically, with the addition of a medication that interferes with SSRI metabolism (eg, cimetidine inhibition of cytochrome P450 2D6).33 If families are prepared for this phenomenon and the therapeutic alliance is adequate, adverse effects can be tolerated to allow for a full medication trial. Common adverse effects of SSRIs include sleep problems (insomnia/sedation), gastrointestinal upset, sexual dysfunction, dry mouth, and hyperhidrosis. Although SSRIs differ somewhat in the frequency of these effects, as a class, they are more similar than different. Adequate psychoeducation is especially imperative in the treatment of OCD and anxiety disorders, where there is limited evidence of efficacy for any non-serotonergic antidepressants.
Serotonin-norepinephrine reuptake inhibitors are not considered first-line medications because of the reduced evidence base compared to SSRIs and their enhanced adverse effect profiles. Because SNRIs partially share a mechanism of action with SSRIs, they also share portions of the adverse effects profile. However, SNRIs have the additional adverse effect of hypertension, which is related to their noradrenergic activity. Thus, it is reasonable to obtain a baseline blood pressure before initiating an SNRI, as well as periodically after initiation and during dose increases, particularly if the patient has other risk factors for hypertension.34
Continue to: Although TCAs have efficacy...
Although TCAs have efficacy in some pediatric disorders,27-29,35 their adverse effect profile limits their use. Tricyclic antidepressants are highly anticholinergic (causing dizziness secondary to orthostatic hypotension, dry mouth, and urinary retention) and antihistaminergic (causing sedation and weight gain). Additionally, TCAs lower the seizure threshold and have adverse cardiac effects relating to their anti-alpha-1 adrenergic activity, resulting in dose-dependent increases in the QTc and cardiac toxicity in overdose that could lead to arrhythmia and death. These medications have their place, but their use requires careful informed consent, clear treatment goals, and baseline and periodic cardiac monitoring (via electrocardiogram).
Serious adverse effects. Clinicians may be hesitant to prescribe antidepressants for pediatric patients because of the potential for more serious adverse effects, including severe behavioral activation syndromes, serotonin syndrome, and emergent suicidality. However, current FDA-approved antidepressants arguably have one of the most positive risk/benefit profiles of any orally-administered medication approved for pediatric patients. Having a strong understanding of the evidence is critical to evaluating when it is appropriate to prescribe an antidepressant, how to properly monitor the patient, and how to obtain accurate informed consent.
Pediatric behavioral activation syndrome. Many clinicians report that children receiving antidepressants experience a pediatric behavioral activation syndrome, which exists along a spectrum from mild activation, increased energy, insomnia, or irritability up through more severe presentations of agitation, hyperactivity, or possibly mania. A recent meta-analysis suggested a positive association between antidepressant use and activation events on the milder end of this spectrum in pediatric patients with non-OCD anxiety disorders,16 and it is thought that compared with adolescents, younger children are more susceptible to activation adverse effects.36 The likelihood of activation events has been associated with higher antidepressant plasma levels,37 suggesting that dose or individual differences in metabolism may play a role. At the severe end of the spectrum, the risk of induction of mania in pediatric patients with depression or anxiety is relatively rare (<2%) and not statistically different from placebo in RCTs of pediatric participants.38 Meta-analyses of larger randomized, placebo-controlled trials of adults do not support the idea that SSRIs and other second-generation antidepressants carry an increased risk of mania compared with placebo.39,40 Children or adolescents with bona fide bipolar disorder (ie, patients who have had observed mania that meets all DSM-5 criteria) should be treated with a mood-stabilizing agent or antipsychotic if prescribed an antidepressant.41 These clear-cut cases are, however, relatively rare, and more often clinicians are confronted with ambiguous cases that include a family history of bipolar disorder along with “softer” symptoms of irritability, intrusiveness, or aggression. In these children, SSRIs may be appropriate for depressive, OCD, or anxiety symptoms, and should be strongly considered before prescribing antipsychotics or mood stabilizers, as long as initiated with proper monitoring.
Serotonin syndrome is a life-threatening condition caused by excess synaptic serotonin. It is characterized by confusion, sweating, diarrhea, hypertension, hyperthermia, and tachycardia. At its most severe, serotonin syndrome can result in seizures, arrhythmias, and death. The risk of serotonin syndrome is very low when using an SSRI as monotherapy. Risk increases with polypharmacy, particularly unexamined polypharmacy when multiple serotonergic agents are inadvertently on board. Commonly used serotonergic agents include other antidepressants, migraine medications (eg, triptans), some pain medications, and the cough suppressant dextromethorphan.
The easiest way to mitigate the risk of serotonin syndrome is to use an interaction index computer program, which can help ensure that the interacting agents are not prescribed without first discussing the risks and benefits. It is important to teach adolescents that certain recreational drugs are highly serotonergic and can cause serious interactions with antidepressants. For example, recreational use of dextromethorphan or 3,4-methylenedioxymethamphetamine (MDMA; commonly known as “ecstasy”) has been associated with serotonin syndrome in adolescents taking antidepressant medications.42,43
Continue to: Suicidality
Suicidality. The black-box warning regarding a risk of emergent suicidality when starting antidepressant treatment in children is controversial.44 The prospect that a medication intended to ameliorate depression might instead risk increasing suicidal thinking is alarming to parents and clinicians alike. To appropriately weigh and discuss the risks and benefits with families, it is important to understand the data upon which the warning is based.

In 2004, the FDA commissioned a review of 23 antidepressant trials, both published and unpublished, pooling studies across multiple indications (MDD, OCD, anxiety, and ADHD) and multiple antidepressant classes. This meta-analysis, which included nearly 4,400 pediatric patients, found a small but statistically significant increase in spontaneously-reported suicidal thoughts or actions, with a risk difference of 1% (95% confidence interval [CI], 1% to 2%).45 These data suggest that if one treats 100 pediatric patients, 1 to 2 of them may experience short-term increases in suicidal thinking or behavior.45 There were no differences in suicidal thinking when assessed systematically (ie, when all subjects reported symptoms of suicidal ideation on structured rating scales), and there were no completed suicides.45 A subsequent analysis that included 27 pediatric trials suggested an even lower, although still significant, risk difference (<1%), yielding a number needed to harm (NNH) of 143.46 Thus, with low NNT for efficacy (3 to 6) and relatively high NNH for emergent suicidal thoughts or behaviors (100 to 143), for many patients the benefits will outweigh the risks.

Figure 1, Figure 2, and Figure 3 are Cates plots that depict the absolute benefits of antidepressants compared with the risk of suicidality for pediatric patients with MDD, OCD, and anxiety disorders. Recent meta-analyses have suggested that the increased risk of suicidality in antidepressant trials is specific to studies that included children and adolescents, and is not observed in adult studies. A meta-analysis of 70 trials involving 18,526 participants suggested that the odds ratio of suicidality in trials of children and adolescents was 2.39 (95% CI, 1.31 to 4.33) compared with 0.81 (95% CI, 0.51 to 1.28) in adults.47 Additionally, a network meta-analysis exclusively focusing on pediatric antidepressant trials in MDD reported significantly higher suicidality-related adverse events in venlafaxine trials compared with placebo, duloxetine, and several SSRIs (fluoxetine, paroxetine, and escitalopram).20 These data should be interpreted with caution as differences in suicidality detected between agents is quite possibly related to differences in the method of assessment between trials, as opposed to actual differences in risk between agents.

Epidemiologic data further support the use of antidepressants in pediatric patients, showing that antidepressant use is associated with decreased teen suicide attempts and completions,48 and the decline in prescriptions that occurred following the black-box warning was accompanied by a 14% increase in teen suicides.49 Multiple hypotheses have been proposed to explain the pediatric clinical trial findings. One idea is that potential adverse effects of activation, or the intended effects of restoring the motivation, energy, and social engagement that is often impaired in depression, increases the likelihood of thinking about suicide or acting on thoughts. Another theory is that reporting of suicidality may be increased, rather than increased de novo suicidality itself. Antidepressants are effective for treating pediatric anxiety disorders, including social anxiety disorder,16 which could result in more willingness to report. Also, the manner in which adverse effects are generally ascertained in trials might have led to increased spontaneous reporting. In many trials, investigators ask whether participants have any adverse effects in general, and inquire about specific adverse effects only if the family answers affirmatively. Thus, the increased rate of other adverse effects associated with antidepressants (sleep problems, gastrointestinal upset, dry mouth, etc.) might trigger a specific question regarding suicidal ideation, which the child or family then may be more likely to report. Alternatively, any type of psychiatric treatment could increase an individual’s propensity to report; in adolescent psychotherapy trials, non-medic
Continue to: How long should the antidepressant be continued?
How long should the antidepressant be continued?
Many patients are concerned about how long they may be taking medication, and whether they will be taking an antidepressant “forever.” A treatment course can be broken into an acute phase, wherein remission is achieved and maintained for 6 to 8 weeks. This is followed by a continuation phase, with the goal of relapse prevention, lasting 16 to 20 weeks. The length of the last phase—the maintenance phase—depends both on the child’s history, the underlying therapeutic indication, the adverse effect burden experienced, and the family’s preferences/values. In general, for a first depressive episode, after treating for 1 year, a trial of discontinuation can be attempted with close monitoring. For a second depressive episode, we recommend 2 years of remission on antidepressant therapy before attempting discontinuation. In patients who have had 3 depressive episodes, or have had episodes of high severity, we recommend continuing antidepressant treatment indefinitely. Although much less well studied, the risk of relapse following SSRI discontinuation appears much more significant in OCD, whereas anxiety disorders and MDD have a relatively comparable risk.52
In general, stopping an antidepressant should be done carefully and slowly.
What to do when first-line treatments fail
Adequate psychotherapy? To determine whether children are receiving adequate CBT, ask:
- if the child receives homework from psychotherapy
- if the parents are included in treatment
- if therapy has involved identifying thought patterns that may be contributing to the child’s illness, and
- if the therapist has ever exposed the child to a challenge likely to produce anxiety or distress in a supervised environment and has developed an exposure hierarchy (for conditions with primarily exposure-based therapies, such as OCD or anxiety disorders).
If a family is not receiving most of these elements in psychotherapy, this is a good indicator that they may not be receiving evidence-based CBT.
Continue to: Adequate pharmacotherapy?
Adequate pharmacotherapy? Similarly, when determining the adequacy of previous pharmacotherapy, it is critical to determine whether the child received an adequate dose of medications (at least the FDA-recommended minimum dose) for an adequate duration of time at therapeutic dosing (at least 6 weeks for MDD, 8 weeks for anxiety disorders, and 8 to 12 weeks for pediatric patients with OCD), and that the child actually took the medication regularly during that period. Patient compliance can typically be tracked through checking refill requests or intervals through the patient’s pharmacy. Ensuring proper delivery of first-line treatments is imperative because (1) the adverse effects associated with second-line treatments are often more substantial; (2) the cost in terms of time and money is considerably higher with second-line treatments, and; (3) the evidence regarding the benefits of these treatments is much less certain.
Inadequate dosing is a common reason for non-response in pediatric patients. Therapeutic dose ranges for common antidepressants are displayed in Table 1. Many clinicians underdose antidepressants for pediatric patients initially (and often throughout treatment) because they fear that the typical dose titration used in clinical trials will increase the risk of adverse effects compared with more conservative dosing. There is limited evidence to suggest that this underdosing strategy is likely to be successful; adverse effects attributable to these medications are modest, and most that are experienced early in treatment (eg, headache, increased anxiety or irritability, sleep problems, gastrointestinal upset) are self-limiting and may be coincidental rather than medication-induced. Furthermore, there is no evidence for efficacy of subtherapeutic dosing in children in the acute phase of treatment or for preventing relapse.14 Thus, from an efficacy standpoint, a medication trial has not officially begun until the therapeutic dose range is reached.
Once dosing is within the therapeutic range, however, pediatric data differs from the adult literature. In most adult psychiatric conditions, higher doses of SSRIs within the therapeutic range are associated with an increased response rate.14,54 In pediatrics, there are few fixed dose trials, and once within the recommended therapeutic range, minimal data supports an association between higher dosing and higher efficacy.14 In general, pediatric guidelines are silent regarding optimal dosing of SSRIs within the recommended dose range, and higher antidepressant doses often result in a more significant adverse effect burden for children. One exception is pediatric OCD, where, similar to adults, the guidelines suggest that higher dosing of SSRIs often is required to induce a therapeutic response as compared to MDD and GAD.31,55
If a child does not respond to adequate first-line treatment (or has a treatment history that cannot be fully verified), repeating these first-line interventions carries little risk and can be quite effective. For example, 60% of adolescents with MDD who did not initially respond to an SSRI demonstrated a significant response when prescribed a second SSRI or venlafaxine (with or without CBT).56
When pediatric patients continue to experience significantly distressing and/or debilitating symptoms (particularly in MDD) despite multiple trials of antidepressants and psychotherapy, practitioners should consider a careful referral to interventional psychiatry services, which can include the more intensive treatments of electroconvulsive therapy, repetitive transcranial magnetic stimulation, or ketamine (see Box 1). Given the substantial morbidity and mortality associated with adolescent depression, interventional psychiatry treatments are under-researched and under-utilized clinically in pediatric populations.
Continue to: Antidepressants in general...
Antidepressants in general, and SSRIs in particular, are the first-line pharmacotherapy for pediatric anxiety, OCD, and MDD. For PTSD, although they are a first-line treatment in adults, their efficacy has not been demonstrated in children and adolescents. Antidepressants are generally safe, well-tolerated, and effective, with low NNTs (3 to 5 for anxiety and OCD; 4 to 12 in MDD, depending on whether industry trials are included). It is important that clinicians and families be educated about possible adverse effects and their time course in order to anticipate difficulties, ensure adequate informed consent, and monitor appropriately. The black-box warning regarding treatment-emergent suicidal thoughts or behaviors must be discussed (for suggested talking points, see Box 2). The NNH is large (100 to 143), and for many patients, the benefits will outweigh the risks. For pediatric patients who fail to respond to multiple adequate trials of antidepressants and psychotherapy, referrals for interventional psychiatry consultation should be considered.
Bottom Line
Although the evidence base for prescribing antidepressants for children and adolescents is smaller compared to the adult literature, properly understanding and prescribing these agents, and explaining their risks and benefits to families, can make a major difference in patient compliance, satisfaction, and outcomes. Antidepressants (particularly selective serotonin reuptake inhibitors) are the firstline pharmacologic intervention for pediatric patients with anxiety disorders, obsessive-compulsive disorder, or major depressive disorder.
Related Resource
- Bonin L, Moreland CS. Overview of prevention and treatment for pediatric depression. https://www.uptodate.com/contents/overview-of-prevention-and-treatment-for-pediatric-depression. Last reviewed July 2019.
Drug Brand Names
Bupropion • Wellbutrin, Zyban
Cimetidine • Tagamet
Citalopram • Celexa
Clomipramine • Anafranil
Desipramine • Norpramin
Desvenlafaxine • Pristiq
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Fluvoxamine • Luvox
Imipramine • Tofranil
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Venlafaxine • Effexor
Vilazodone • Viibryd
Vortioxetine • Trintellix
Box 1
Continuing severe depression is associated with reduced educational attainment and quality of life, as well as increased risk of substance abuse and suicide,1,2 which is the second leading cause of death in individuals age 10 to 24 years.3 Given the substantial morbidity and mortality associated with adolescent depression, interventional psychiatry treatments are under-researched and underutilized in pediatric patients. Interventional antidepressants in adults include electroconvulsive therapy (ECT), repetitive transcranial magnetic stimulation (rTMS), and, most recently, ketamine.
Electroconvulsive therapy is the most effective therapy available for depression in adults, alleviating depressive symptoms in treatment-refractory patients and outperforming both pharmacotherapy4 and rTMS.5 Despite its track record of effectiveness and safety in adults, ECT continues to suffer considerable stigma.4 Cognitive adverse effects and memory problems in adults are generally self-limited, and some aspects of cognition actually improve after ECT as depression lifts.6 The combination of stigma and the concern about possible cognitive adverse effects during periods of brain development have likely impeded the rigorous testing of ECT in treatment-refractory pediatric patients. Several case series and other retrospective analyses suggest, however, that ECT has strong efficacy and limited adverse effects in adolescents who have severe depression or psychotic symptoms.7-9 Despite these positive preliminary data in pediatric patients, and a large body of literature in adults, no controlled trials of ECT have been conducted in the pediatric population, and it remains a rarely used treatment in severe pediatric mental illness.
Repetitive transcranial magnetic stimulation is a technique in which magnetic stimulation is used to activate the left dorsolateral prefrontal cortex (DLPFC), a target thought to be important in the pathophysiology of MDD. Repetitive transcranial magnetic stimulation is FDAapproved to treat medication-refractory major depressive disorder (MDD) in adults, and has been shown to be effective as both a monotherapy10 and an adjunctive treatment.11 The estimated number needed to treat (NNT) for rTMS ranges from 6 to 8, which is quite effective, although less so than ECT (and probably initial pharmacotherapy).5 Similar to ECT, however, there are no large randomized controlled trials (RCTs) in children or adolescents. Pilot RCTs12 and open trials13 suggest that DLPFC rTMS may be effective as an adjunctive treatment, speeding or augmenting response to a selective serotonin reuptake inhibitor in adolescents with MDD. Larger trials studying rTMS in pediatric patients are needed. While rTMS is generally well tolerated, disadvantages include the time-consuming schedule (the initial treatment is typically 5 days/week for several weeks) and local adverse effects of headache and scalp pain.
Ketamine, which traditionally is used as a dissociative anesthetic, is a rapidly emerging novel treatment in adult treatment-refractory MDD. It acts quickly (within hours to days) and cause significant improvement in difficult symptoms such as anhedonia14 and suicidal ideation.15 In adult studies, ketamine has a robust average effect size of >1.2, and an NNT ranging from 3 to 5 in medication-refractory patients.16,17 Ketamine is a glutamatergic modulator, acting outside of the monoamine neurochemical systems traditionally targeted by standard antidepressants.16 The efficacy of ketamine in treatment-refractory adults is impressive, but the effects of a single treatment are ephemeral, dissipating within 1 to 2 weeks, which has led to significant discussion surrounding optimal dosing strategies.16 Although small RCTs in pediatric patients are currently underway, at this time, the only evidence for ketamine for pediatric MDD is based on case series/report data18,19 which was positive.
For all of these interventional modalities, it is critical to refer children with treatmentrefractory disorders to interventionists who have appropriate experience and monitoring capabilities.
References
1. Weissman MM, Wolk S, Goldstein RB, et al. Depressed adolescents grown up. JAMA.1999;281(18):1707-1713.
2. Fergusson DM, Woodward LJ. Mental health, educational, and social role outcomes of adolescents with depression. Arch Gen Psychiatry. 2002;59(3):225-231.
3. Centers for Disease Control and Prevention. National Vital Statistics System. Deaths, percent of total deaths, and death rates for the 15 leading causes of death in 5-year age groups, by race and sex: United States, 1999-2015. Centers for Disease Control and Prevention. https://www.cdc.gov/nchs/nvss/mortality/lcwk1.htm. Published October 23, 2017. Accessed May 2, 2019.
4. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and metaanalysis. Lancet. 2003;361(9360):799-808.
5. Berlim MT, Van den Eynde F, Daskalakis ZJ. Efficacy and acceptability of high frequency repetitive transcranial magnetic stimulation (rTMS) versus electroconvulsive therapy (ECT) for major depression: a systematic review and meta-analysis of randomized trials. Depress Anxiety. 2013;30(7):614-623.
6. Semkovska M, McLoughlin DM. Objective cognitive performance associated with electroconvulsive therapy for depression: a systematic review and meta-analysis. Biol Psychiatry. 2010;68(6):568-577.
7. Jacob P, Gogi PK, Srinath S, et al. Review of electroconvulsive therapy practice from a tertiary child and adolescent psychiatry centre. Asian J Psychiatr. 2014;12(1):95-99.
8. Zhand N, Courtney DB, Flament MF. Use of electroconvulsive therapy in adolescents with treatment-resistant depressive disorders: a case series. J ECT. 2015;31(4):238-245.
9. Puffer CC, Wall CA, Huxsahl JE, et al. A 20 year practice review of electroconvulsive therapy for adolescents. J Child Adolesc Psychopharmacol. 2016;26(7):632-636.
10. Berlim MT, van den Eynde F, Tovar-Perdomo S, et al. Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rTMS) for treating major depression: a systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol Med. 2014;44(2):225-239.
11. Liu B, Zhang Y, Zhang L, et al. Repetitive transcranial magnetic stimulation as an augmentative strategy for treatment-resistant depression, a meta-analysis of randomized, double-blind and sham-controlled study. BMC Psychiatry. 2014;14:342.
12. Huang ML, Luo BY, Hu JB, et al. Repetitive transcranial magnetic stimulation in combination with citalopram in young patients with first-episode major depressive disorder: a double-blind, randomized, sham-controlled trial. Aust N Z J Psychiatry. 2012;46(3):257-264.
13. Wall CA, Croarkin PE, Sim LA, et al. Adjunctive use of repetitive transcranial magnetic stimulation in depressed adolescents: a prospective, open pilot study. J Clin Psychiatry. 2011;72(9):1263-1269.
14. Lally N, Nugent AC, Luckenbaugh DA, et al. Anti-anhedonic effect of ketamine and its neural correlates in treatment-resistant bipolar depression. Transl Psychiatry. 2014;4:e469. doi: 10.1038/tp.2014.105.
15. Ballard ED, Ionescu DF, Vande Voort JL, et al. Improvement in suicidal ideation after ketamine infusion: relationship to reductions in depression and anxiety. J Psychiatr Res. 2014;58:161-166.
16. Newport DJ, Carpenter LL, McDonald WM, et al. Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry. 2015;172(10):950-966.
17. McGirr A, Berlim MT, Bond DJ, et al. A systematic review and meta-analysis of randomized, double-blind, placebo-controlled trials of ketamine in the rapid treatment of major depressive episodes. Psychol Med. 2015;45(4):693-704.
18. Dwyer JB, Beyer C, Wilkinson ST, et al. Ketamine as a treatment for adolescent depression: a case report. J Am Acad Child Adolesc Psychiatry. 2017;56(4):352-354.
19. Cullen KR, Amatya P, Roback MG, et al. Intravenous ketamine for adolescents with treatment-resistant depression: an open-label study. J Child Adolesc Psychopharmacol. 2018;28(7):437-444.
Box 2
Efficacy
- Selective serotonin reuptake inhibitors are the most effective pharmacologic treatment we have for pediatric depression, OCD, and anxiety
- More than one-half of children who are prescribed SSRIs have a significant improvement, regardless of condition
- Based on current estimates, we need to treat 4 to 6 children with an SSRI to find one that will improve who would not improve with placebo
- The clinical benefits of SSRIs generally take a while to accrue; therefore, it is advisable to take the medication for at least 2 to 3 months before concluding that it is ineffective
- In addition to medication, evidence-based psychotherapies provide significant benefit for pediatric depression, OCD, and anxiety
Tolerability
- Most commonly prescribed pediatric antidepressants have been used safely in children for 2 to 3 decades. The safety profiles of SSRIs are among the best of any medications used for children and adolescents
- While many children get better when taking these medications, it’s important that we also talk about potential adverse effects. Some children will experience sleep problems (either sleepier than usual or difficulty sleeping), changes in energy levels, headache, gastrointestinal upset, and dry mouth. These are most likely at the beginning of treatment, or when we increase the dose; they usually are time-limited and go away on their own
- Often adverse effects occur first and the benefits come later. Because it may take at least a few weeks to start to see the mood/anxiety benefits, it’s important for us to talk about any adverse effects your child experiences and remember that they usually are short-lived
Suicidality
- The FDA placed a “black-box” warning on antidepressants after pediatric studies found a small but statistically significant increased risk of reporting suicidal thoughts or behaviors over the short-term compared with placebo
- The increased risk of spontaneously reporting suicidal ideation was quite small. Studies suggested that one would need to treat 100 to 140 children to see 1 child report suicidal ideation compared to placebo. Suicidal ideation is a common symptom in children with depression and anxiety
- Studies found no increased risk when suicidal ideation was systematically assessed using structured rating scales
- In the studies evaluated, there were no completed suicides by patients taking medication or placebo
- Population studies show that higher rates of antidepressant prescriptions are associated with lower rates of attempted and completed teen suicide, which underscores that in general, these medicines treat the underlying causes of suicidality
- No scientific consensus exists on whether these medications are truly associated with an increased risk of new-onset suicidal ideation, or if this association is due to other factors (eg, improvement in anxiety and depressive symptoms that make patients more comfortable to report suicidal ideation spontaneously)
- Regardless, the FDA recommends frequent monitoring of children for suicidal thoughts when these medications are started. This should be done anyway in children experiencing depression and anxiety, and it’s why we will plan to have more frequent appointments as the medication is initiated
OCD: obsessive-compulsive disorder; SSRIs: selective serotonin reuptake inhibitors
1. Williams SB, O’Connor EA, Eder M, et al. Screening for child and adolescent depression in primary care settings: a systematic evidence review for the US Preventive Services Task Force. Pediatrics. 2009;123(4):e716-e735. doi: 10.1542/peds.2008-2415.
2. Kessler RC, Avenevoli S, Ries Merikangas K. Mood disorders in children and adolescents: an epidemiologic perspective. Biol Psychiatry. 2001;49(12):1002-1014.
3. Lewinsohn PM, Clarke GN, Seeley JR, et al. Major depression in community adolescents: age at onset, episode duration, and time to recurrence. J Am Acad Child Adolesc Psychiatry. 1994;33(6):809-818.
4. Weissman MM, Wolk S, Goldstein RB, et al. Depressed adolescents grown up. JAMA.1999;281(18):1707-1713.
5. Fergusson DM, Woodward LJ. Mental health, educational, and social role outcomes of adolescents with depression. Arch Gen Psychiatry. 2002;59(3):225-231.
6. Keenan-Miller D, Hammen CL, Brennan PA. Health outcomes related to early adolescent depression. J Adolesc Health. 2007; 41(3): 256-62.
7. Shaffer D, Gould MS, Fisher P, et al. Psychiatric diagnosis in child and adolescent suicide. Arch Gen Psychiatry. 1996;53(4):339-348.
8. Centers for Disease Control and Prevention. National Vital Statistics System. Deaths, percent of total deaths, and death rates for the 15 leading causes of death in 5-year age groups, by race and sex: United States, 1999-2015. https://www.cdc.gov/nchs/nvss/mortality/lcwk1.htm. Published October 23, 2017. Accessed May 2, 2019.
9. Merikangas KR, He JP, Burstein M, et al. Lifetime prevalence of mental disorders in US adolescents: results from the National Comorbidity Survey Replication-Adolescent Supplement (NCS-A). J Am Acad Child Adolesc Psychiatry. 2010;49(10):980-989.
10. Wittchen HU, Nelson CB, Lachner G. Prevalence of mental disorders and psychosocial impairments in adolescents and young adults. Psychol Med. 1998;28(1):109-126.
11. Foley DL, Goldston DB, Costello EJ, et al. Proximal psychiatric risk factors for suicidality in youth: the Great Smoky Mountains Study. Arch Gen Psychiatry. 2006;63(9):1017-1024.
12. Cheung A, Sacks D, Dewa CS, et al. Pediatric prescribing practices and the FDA black-box warning on antidepressants. J Dev Behav Pediatr. 2008 29(3):213-215.
13. Walkup JT. Antidepressant efficacy for depression in children and adolescents: industry- and NIMH-funded studies. Am J Psychiatry. 2017;174(5):430-437.
14. Jakubovski E, Varigonda AL, Freemantle N, et al. Systematic review and meta-analysis: dose-response relationship of selective serotonin reuptake inhibitors in major depressive disorder. Am J Psychiatry. 2016;173(2):174-183.
15. Varigonda AL, Jakubovski E, Taylor MJ, et al. Systematic review and meta-analysis: early treatment responses of selective serotonin reuptake inhibitors in pediatric major depressive disorder. J Am Acad Child Adolesc Psychiatry. 2015;54(7):557-564.
16. Strawn JR, Welge JA, Wehry AM, et al. Efficacy and tolerability of antidepressants in pediatric anxiety disorders: a systematic review and meta-analysis. Depress Anxiety. 2015;32(3):149-157.
17. March JS, Biederman J, Wolkow R, et al. Sertraline in children and adolescents with obsessive-compulsive disorder: a multicenter randomized controlled trial. JAMA. 1998;280(20):1752-1756.
18. Walkup JT, Albano AM, Piacentini J, et al. Cognitive behavioral therapy, sertraline, or a combination in childhood anxiety. N Engl J Med. 2008;359(26):2753-2766.
19. Kennard BD, Silva SG, Tonev S, et al. Remission and recovery in the Treatment for Adolescents with Depression Study (TADS): acute and long-term outcomes. J Am Acad Child Adolesc Psychiatry. 2009;48(2):186-195.
20. Cipriani A, Zhou X, Del Giovane C, et al. Comparative efficacy and tolerability of antidepressants for major depressive disorder in children and adolescents: a network meta-analysis. Lancet. 2016;388(10047):881-890.
21. Cohen JA, Mannarino AP, Perel JM, et al. A pilot randomized controlled trial of combined trauma-focused CBT and sertraline for childhood PTSD symptoms. J Am Acad Child Adolesc Psychiatry. 2007;46(7):811-819.
22. Robb AS, Cueva JE, Sporn J, et al. Sertraline treatment of children and adolescents with posttraumatic stress disorder: a double-blind, placebo-controlled trial. J Child Adolesc Psychopharmacol. 2010;20(6):463-471.
23. Diehle J, Opmeer BC, Boer F, et al. Trauma-focused cognitive behavioral therapy or eye movement desensitization and reprocessing: what works in children with posttraumatic stress symptoms? A randomized controlled trial. Eur Child Adolesc Psychiatry. 2015;24(2):227-236.
24. Aiyer R, Barkin RL, Bhatia A. Treatment of neuropathic pain with venlafaxine: a systematic review. Pain Med. 2017;18(10):1999-2012.
25. Barrickman LL, Perry PJ, Allen AJ, et al. Bupropion versus methylphenidate in the treatment of attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 1995;34(5):649-657.
26. Monuteaux MC, Spencer TJ, Faraone SV, et al. A randomized, placebo-controlled clinical trial of bupropion for the prevention of smoking in children and adolescents with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2007;68(7):1094-1101.
27. Biederman J, Baldessarini RJ, Wright V, et al. A double-blind placebo controlled study of desipramine in the treatment of ADD: I. Efficacy. J Am Acad Child Adolesc Psychiatry. 1989;28(5):777-784.
28. Spencer T, Biederman J, Coffey B, et al. A double-blind comparison of desipramine and placebo in children and adolescents with chronic tic disorder and comorbid attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2002;59(7):649-656.
29. DeVeaugh-Geiss J, Moroz G, Biederman J, et al. Clomipramine hydrochloride in childhood and adolescent obsessive-compulsive disorder--a multicenter trial. J Am Acad Child Adolesc Psychiatry. 1992;31(1):45-49.
30. Caldwell PH, Sureshkumar P, Wong WC. Tricyclic and related drugs for nocturnal enuresis in children. Cochrane Database Syst Rev. 2016;(1):CD002117.
31. Varigonda AL, Jakubovski E, Bloch MH. Systematic review and meta-analysis: early treatment responses of selective serotonin reuptake inhibitors and clomipramine in pediatric obsessive-compulsive disorder. J Am Acad Child Adolesc Psychiatry. 2016;55(10):851-859.e2. doi: 10.1016/j.jaac.2016.07.768.
32. Walkup J, Labellarte M. Complications of SSRI treatment. J Child Adolesc Psychopharmacol. 2001;11(1):1-4.
33. Leo RJ, Lichter DG, Hershey LA. Parkinsonism associated with fluoxetine and cimetidine: a case report. J Geriatr Psychiatry Neurol. 1995;8(4):231-233.
34. Strawn JR, Prakash A, Zhang Q, et al. A randomized, placebo-controlled study of duloxetine for the treatment of children and adolescents with generalized anxiety disorder. J Am Acad Child Adolesc Psychiatry. 2015;54(4):283-293.
35. Bernstein GA, Borchardt CM, Perwien AR, et al. Imipramine plus cognitive-behavioral therapy in the treatment of school refusal. J Am Acad Child Adolesc Psychiatry. 2000;39(3): 276-283.
36. Safer DJ, Zito JM. Treatment-emergent adverse events from selective serotonin reuptake inhibitors by age group: children versus adolescents. J Child Adolesc Psychopharmacol. 2006;16(1-2):159-169.
37. Reinblatt SP, DosReis S, Walkup JT, et al. Activation adverse events induced by the selective serotonin reuptake inhibitor fluvoxamine in children and adolescents. J Child Adolesc Psychopharmacol. 2009;19(2):119-126.
38. Goldsmith M, Singh M, Chang K. Antidepressants and psychostimulants in pediatric populations: is there an association with mania? Paediatr Drugs. 2011;13(4): 225-243.
39. Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156-167.
40. Allain N, Leven C, Falissard B, et al. Manic switches induced by antidepressants: an umbrella review comparing randomized controlled trials and observational studies. Acta Psychiatr Scand. 2017;135(2):106-116.
41. McClellan J, Kowatch R, Findling RL. Practice parameter for the assessment and treatment of children and adolescents with bipolar disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(1):107-125.
42. Dobry Y, Rice T, Sher L. Ecstasy use and serotonin syndrome: a neglected danger to adolescents and young adults prescribed selective serotonin reuptake inhibitors. Int J Adolesc Med Health. 2013; 25(3):193-199.
43. Schwartz AR, Pizon AF, Brooks DE. Dextromethorphan-induced serotonin syndrome. Clin Toxicol (Phila). 2008;46(8):771-773.
44. Gibbons RD, Brown CH, Hur K, et al. Early evidence on the effects of regulators’ suicidality warnings on SSRI prescriptions and suicide in children and adolescents. Am J Psychiatry. 2007;164(9):1356-1363.
45. Hammad TA, Laughren T, Racoosin J. Suicidality in pediatric patients treated with antidepressant drugs. Arch Gen Psychiatry. 2006;63(3):332-339.
46. Bridge JA, Iyengar S, Salary CB, et al. Clinical response and risk for reported suicidal ideation and suicide attempts in pediatric antidepressant treatment: a meta-analysis of randomized controlled trials. JAMA. 2007;297(15):1683-1696.
47. Sharma T, Guski LS, Freund N, et al. Suicidality and aggression during antidepressant treatment: systematic review and meta-analyses based on clinical study reports. BMJ. 2016;352: i65. doi: https://doi.org/10.1136/bmj.i65.
48. Olfson M, Shaffer D, Marcus SC, et al. Relationship between antidepressant medication treatment and suicide in adolescents. Arch Gen Psychiatry. 2003;60(10):978-982.
49. Garland JE, Kutcher S, Virani A, et al. Update on the Use of SSRIs and SNRIs with children and adolescents in clinical practice. J Can Acad Child Adolesc Psychiatry. 2016;25(1):4-10.
50. Bridge JA, Barbe RP, Birmaher B, et al. Emergent suicidality in a clinical psychotherapy trial for adolescent depression. Am J Psychiatry. 2005;162(11):2173-2175.
51. Birmaher B, Brent D, Bernet W, et al. Practice parameter for the assessment and treatment of children and adolescents with depressive disorders. J Am Acad Child Adolesc Psychiatry. 2007;46(11):1503-1526.
52. Ravizza L, Maina G, Bogetto F, et al. Long term treatment of obsessive-compulsive disorder. CNS Drugs. 1998;10(4):247-255.
53. Hosenbocus S, Chahal R. SSRIs and SNRIs: a review of the discontinuation syndrome in children and adolescents. J Can Acad Child Adolesc Psychiatry. 2011;20(1):60-67.
54. Bloch MH, McGuire J, Landeros-Weisenberger A, et al. Meta-analysis of the dose-response relationship of SSRI in obsessive-compulsive disorder. Mol Psychiatry. 2010;15(8):850-855.
55. Issari Y, Jakubovski E, Bartley CA, et al. Early onset of response with selective serotonin reuptake inhibitors in obsessive-compulsive disorder: a meta-analysis. J Clin Psychiatry. 2016; 77(5):e605-e611. doi: 10.4088/JCP.14r09758.
56. Brent D, Emslie G, Clarke G, et al. Switching to another SSRI or to venlafaxine with or without cognitive behavioral therapy for adolescents with SSRI-resistant depression: the TORDIA randomized controlled trial. JAMA. 2008;299(8):901-913.
1. Williams SB, O’Connor EA, Eder M, et al. Screening for child and adolescent depression in primary care settings: a systematic evidence review for the US Preventive Services Task Force. Pediatrics. 2009;123(4):e716-e735. doi: 10.1542/peds.2008-2415.
2. Kessler RC, Avenevoli S, Ries Merikangas K. Mood disorders in children and adolescents: an epidemiologic perspective. Biol Psychiatry. 2001;49(12):1002-1014.
3. Lewinsohn PM, Clarke GN, Seeley JR, et al. Major depression in community adolescents: age at onset, episode duration, and time to recurrence. J Am Acad Child Adolesc Psychiatry. 1994;33(6):809-818.
4. Weissman MM, Wolk S, Goldstein RB, et al. Depressed adolescents grown up. JAMA.1999;281(18):1707-1713.
5. Fergusson DM, Woodward LJ. Mental health, educational, and social role outcomes of adolescents with depression. Arch Gen Psychiatry. 2002;59(3):225-231.
6. Keenan-Miller D, Hammen CL, Brennan PA. Health outcomes related to early adolescent depression. J Adolesc Health. 2007; 41(3): 256-62.
7. Shaffer D, Gould MS, Fisher P, et al. Psychiatric diagnosis in child and adolescent suicide. Arch Gen Psychiatry. 1996;53(4):339-348.
8. Centers for Disease Control and Prevention. National Vital Statistics System. Deaths, percent of total deaths, and death rates for the 15 leading causes of death in 5-year age groups, by race and sex: United States, 1999-2015. https://www.cdc.gov/nchs/nvss/mortality/lcwk1.htm. Published October 23, 2017. Accessed May 2, 2019.
9. Merikangas KR, He JP, Burstein M, et al. Lifetime prevalence of mental disorders in US adolescents: results from the National Comorbidity Survey Replication-Adolescent Supplement (NCS-A). J Am Acad Child Adolesc Psychiatry. 2010;49(10):980-989.
10. Wittchen HU, Nelson CB, Lachner G. Prevalence of mental disorders and psychosocial impairments in adolescents and young adults. Psychol Med. 1998;28(1):109-126.
11. Foley DL, Goldston DB, Costello EJ, et al. Proximal psychiatric risk factors for suicidality in youth: the Great Smoky Mountains Study. Arch Gen Psychiatry. 2006;63(9):1017-1024.
12. Cheung A, Sacks D, Dewa CS, et al. Pediatric prescribing practices and the FDA black-box warning on antidepressants. J Dev Behav Pediatr. 2008 29(3):213-215.
13. Walkup JT. Antidepressant efficacy for depression in children and adolescents: industry- and NIMH-funded studies. Am J Psychiatry. 2017;174(5):430-437.
14. Jakubovski E, Varigonda AL, Freemantle N, et al. Systematic review and meta-analysis: dose-response relationship of selective serotonin reuptake inhibitors in major depressive disorder. Am J Psychiatry. 2016;173(2):174-183.
15. Varigonda AL, Jakubovski E, Taylor MJ, et al. Systematic review and meta-analysis: early treatment responses of selective serotonin reuptake inhibitors in pediatric major depressive disorder. J Am Acad Child Adolesc Psychiatry. 2015;54(7):557-564.
16. Strawn JR, Welge JA, Wehry AM, et al. Efficacy and tolerability of antidepressants in pediatric anxiety disorders: a systematic review and meta-analysis. Depress Anxiety. 2015;32(3):149-157.
17. March JS, Biederman J, Wolkow R, et al. Sertraline in children and adolescents with obsessive-compulsive disorder: a multicenter randomized controlled trial. JAMA. 1998;280(20):1752-1756.
18. Walkup JT, Albano AM, Piacentini J, et al. Cognitive behavioral therapy, sertraline, or a combination in childhood anxiety. N Engl J Med. 2008;359(26):2753-2766.
19. Kennard BD, Silva SG, Tonev S, et al. Remission and recovery in the Treatment for Adolescents with Depression Study (TADS): acute and long-term outcomes. J Am Acad Child Adolesc Psychiatry. 2009;48(2):186-195.
20. Cipriani A, Zhou X, Del Giovane C, et al. Comparative efficacy and tolerability of antidepressants for major depressive disorder in children and adolescents: a network meta-analysis. Lancet. 2016;388(10047):881-890.
21. Cohen JA, Mannarino AP, Perel JM, et al. A pilot randomized controlled trial of combined trauma-focused CBT and sertraline for childhood PTSD symptoms. J Am Acad Child Adolesc Psychiatry. 2007;46(7):811-819.
22. Robb AS, Cueva JE, Sporn J, et al. Sertraline treatment of children and adolescents with posttraumatic stress disorder: a double-blind, placebo-controlled trial. J Child Adolesc Psychopharmacol. 2010;20(6):463-471.
23. Diehle J, Opmeer BC, Boer F, et al. Trauma-focused cognitive behavioral therapy or eye movement desensitization and reprocessing: what works in children with posttraumatic stress symptoms? A randomized controlled trial. Eur Child Adolesc Psychiatry. 2015;24(2):227-236.
24. Aiyer R, Barkin RL, Bhatia A. Treatment of neuropathic pain with venlafaxine: a systematic review. Pain Med. 2017;18(10):1999-2012.
25. Barrickman LL, Perry PJ, Allen AJ, et al. Bupropion versus methylphenidate in the treatment of attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 1995;34(5):649-657.
26. Monuteaux MC, Spencer TJ, Faraone SV, et al. A randomized, placebo-controlled clinical trial of bupropion for the prevention of smoking in children and adolescents with attention-deficit/hyperactivity disorder. J Clin Psychiatry. 2007;68(7):1094-1101.
27. Biederman J, Baldessarini RJ, Wright V, et al. A double-blind placebo controlled study of desipramine in the treatment of ADD: I. Efficacy. J Am Acad Child Adolesc Psychiatry. 1989;28(5):777-784.
28. Spencer T, Biederman J, Coffey B, et al. A double-blind comparison of desipramine and placebo in children and adolescents with chronic tic disorder and comorbid attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2002;59(7):649-656.
29. DeVeaugh-Geiss J, Moroz G, Biederman J, et al. Clomipramine hydrochloride in childhood and adolescent obsessive-compulsive disorder--a multicenter trial. J Am Acad Child Adolesc Psychiatry. 1992;31(1):45-49.
30. Caldwell PH, Sureshkumar P, Wong WC. Tricyclic and related drugs for nocturnal enuresis in children. Cochrane Database Syst Rev. 2016;(1):CD002117.
31. Varigonda AL, Jakubovski E, Bloch MH. Systematic review and meta-analysis: early treatment responses of selective serotonin reuptake inhibitors and clomipramine in pediatric obsessive-compulsive disorder. J Am Acad Child Adolesc Psychiatry. 2016;55(10):851-859.e2. doi: 10.1016/j.jaac.2016.07.768.
32. Walkup J, Labellarte M. Complications of SSRI treatment. J Child Adolesc Psychopharmacol. 2001;11(1):1-4.
33. Leo RJ, Lichter DG, Hershey LA. Parkinsonism associated with fluoxetine and cimetidine: a case report. J Geriatr Psychiatry Neurol. 1995;8(4):231-233.
34. Strawn JR, Prakash A, Zhang Q, et al. A randomized, placebo-controlled study of duloxetine for the treatment of children and adolescents with generalized anxiety disorder. J Am Acad Child Adolesc Psychiatry. 2015;54(4):283-293.
35. Bernstein GA, Borchardt CM, Perwien AR, et al. Imipramine plus cognitive-behavioral therapy in the treatment of school refusal. J Am Acad Child Adolesc Psychiatry. 2000;39(3): 276-283.
36. Safer DJ, Zito JM. Treatment-emergent adverse events from selective serotonin reuptake inhibitors by age group: children versus adolescents. J Child Adolesc Psychopharmacol. 2006;16(1-2):159-169.
37. Reinblatt SP, DosReis S, Walkup JT, et al. Activation adverse events induced by the selective serotonin reuptake inhibitor fluvoxamine in children and adolescents. J Child Adolesc Psychopharmacol. 2009;19(2):119-126.
38. Goldsmith M, Singh M, Chang K. Antidepressants and psychostimulants in pediatric populations: is there an association with mania? Paediatr Drugs. 2011;13(4): 225-243.
39. Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry. 2011;72(2):156-167.
40. Allain N, Leven C, Falissard B, et al. Manic switches induced by antidepressants: an umbrella review comparing randomized controlled trials and observational studies. Acta Psychiatr Scand. 2017;135(2):106-116.
41. McClellan J, Kowatch R, Findling RL. Practice parameter for the assessment and treatment of children and adolescents with bipolar disorder. J Am Acad Child Adolesc Psychiatry. 2007;46(1):107-125.
42. Dobry Y, Rice T, Sher L. Ecstasy use and serotonin syndrome: a neglected danger to adolescents and young adults prescribed selective serotonin reuptake inhibitors. Int J Adolesc Med Health. 2013; 25(3):193-199.
43. Schwartz AR, Pizon AF, Brooks DE. Dextromethorphan-induced serotonin syndrome. Clin Toxicol (Phila). 2008;46(8):771-773.
44. Gibbons RD, Brown CH, Hur K, et al. Early evidence on the effects of regulators’ suicidality warnings on SSRI prescriptions and suicide in children and adolescents. Am J Psychiatry. 2007;164(9):1356-1363.
45. Hammad TA, Laughren T, Racoosin J. Suicidality in pediatric patients treated with antidepressant drugs. Arch Gen Psychiatry. 2006;63(3):332-339.
46. Bridge JA, Iyengar S, Salary CB, et al. Clinical response and risk for reported suicidal ideation and suicide attempts in pediatric antidepressant treatment: a meta-analysis of randomized controlled trials. JAMA. 2007;297(15):1683-1696.
47. Sharma T, Guski LS, Freund N, et al. Suicidality and aggression during antidepressant treatment: systematic review and meta-analyses based on clinical study reports. BMJ. 2016;352: i65. doi: https://doi.org/10.1136/bmj.i65.
48. Olfson M, Shaffer D, Marcus SC, et al. Relationship between antidepressant medication treatment and suicide in adolescents. Arch Gen Psychiatry. 2003;60(10):978-982.
49. Garland JE, Kutcher S, Virani A, et al. Update on the Use of SSRIs and SNRIs with children and adolescents in clinical practice. J Can Acad Child Adolesc Psychiatry. 2016;25(1):4-10.
50. Bridge JA, Barbe RP, Birmaher B, et al. Emergent suicidality in a clinical psychotherapy trial for adolescent depression. Am J Psychiatry. 2005;162(11):2173-2175.
51. Birmaher B, Brent D, Bernet W, et al. Practice parameter for the assessment and treatment of children and adolescents with depressive disorders. J Am Acad Child Adolesc Psychiatry. 2007;46(11):1503-1526.
52. Ravizza L, Maina G, Bogetto F, et al. Long term treatment of obsessive-compulsive disorder. CNS Drugs. 1998;10(4):247-255.
53. Hosenbocus S, Chahal R. SSRIs and SNRIs: a review of the discontinuation syndrome in children and adolescents. J Can Acad Child Adolesc Psychiatry. 2011;20(1):60-67.
54. Bloch MH, McGuire J, Landeros-Weisenberger A, et al. Meta-analysis of the dose-response relationship of SSRI in obsessive-compulsive disorder. Mol Psychiatry. 2010;15(8):850-855.
55. Issari Y, Jakubovski E, Bartley CA, et al. Early onset of response with selective serotonin reuptake inhibitors in obsessive-compulsive disorder: a meta-analysis. J Clin Psychiatry. 2016; 77(5):e605-e611. doi: 10.4088/JCP.14r09758.
56. Brent D, Emslie G, Clarke G, et al. Switching to another SSRI or to venlafaxine with or without cognitive behavioral therapy for adolescents with SSRI-resistant depression: the TORDIA randomized controlled trial. JAMA. 2008;299(8):901-913.
Over decades, low physical activity boosts mortality risk
PARIS – , according to results from a prospective cohort study that followed more than 23,000 adults for over 2 decades.
“Individuals who remained physically inactive, or who decreased their physical activity over 22 years, had substantially increased risk of dying from all causes, and from cardiovascular disease,” Trine Moholdt, PhD, said at a press conference at the annual congress of the European Society of Cardiology.
“The bad news is that sustained inactivity was associated with a 99% increase in all-cause mortality and 168% increase in cardiovascular deaths” compared with sustained physical activity, she said.
The news was similarly bad for individuals who had been highly active and then became inactive; this cohort had an increase in all-cause mortality of 116% and sustained a 173% increase in cardiovascular deaths. “Previous activity levels – they don’t count. You have to keep it up,” said Dr. Moholdt, speaking in a video interview.
However, the risk associated with inactivity was attenuated for those patients who became more active over the course of the study period; their risk was still higher than that of those who had sustained activity, but lower than those who remained inactive. “It’s never too late to start being physically active,” said Dr. Moholdt.
All groups were compared with individuals who remained highly active over the study period; this reference group had the lowest risk of both cardiovascular and all-cause death.
Dr. Moholdt noted a limitation of most previous research into how physical activity relates to cardiovascular and all-cause mortality: Data are obtained just at baseline, and then associated with a downstream outcome. “But people change!” she said, so it’s important to track how activity levels change over time.
To that end, the Nord-Trøndelag Health Study (HUNT study) measured activity levels for 23,156 participants at two points, and then assessed cardiovascular and all-cause mortality, over a period of 22 years.
Dr. Moholdt, head of the Exercise, Cardiometabolic Health and Reproduction research group at Norwegian University of Science and Technology, Trondheim, and her collaborators asked individuals about physical activity in 1985, and again in 2007. At each time point, investigators asked participants to report how many hours per week they spent in “mosjon,” the Norwegian word for “motion”; Dr. Moholdt explained that the word carries the connotation of purposeful, health-promoting physical activity.
Individuals were grouped into one of three activity levels at each time point: Inactive participants reported no purposeful physical activity, moderately active participants reported less than 2 hours per week of this activity, and the most highly active group was active for 2 or more hours per week.
Patients who were in each of the three activity groupings at baseline might have changed their activity levels by the second assessment, so the investigators tracked a total of nine categories of exercise trends over time.
The mean age of participants in the Nord-Trøndelag Health Study (HUNT study) was 39 years, and the mean body mass index was 24 kg/m2. Of the 23,156 participants, 12,665 were women. Dr. Moholdt and her colleagues used statistical analysis to adjust for age, sex, body mass index, smoking, educational status, and systolic blood pressure.
However, Dr. Moholdt acknowledged at the press conference that comorbidities not captured in the study design might impact changes in activity levels, particularly for formerly highly active patients who became inactive over the study period.
The study was presented in a poster session at the congress. Dr. Moholdt reported no conflicts of interest. The study was funded by Health Central Norway (Helse-Midt Norge), a state entity that operates health care facilities in the region of Norway where the HUNT study was conducted.
SOURCE: Moholdt T. et al. ESC Congress 2019, Abstract P627.
PARIS – , according to results from a prospective cohort study that followed more than 23,000 adults for over 2 decades.
“Individuals who remained physically inactive, or who decreased their physical activity over 22 years, had substantially increased risk of dying from all causes, and from cardiovascular disease,” Trine Moholdt, PhD, said at a press conference at the annual congress of the European Society of Cardiology.
“The bad news is that sustained inactivity was associated with a 99% increase in all-cause mortality and 168% increase in cardiovascular deaths” compared with sustained physical activity, she said.
The news was similarly bad for individuals who had been highly active and then became inactive; this cohort had an increase in all-cause mortality of 116% and sustained a 173% increase in cardiovascular deaths. “Previous activity levels – they don’t count. You have to keep it up,” said Dr. Moholdt, speaking in a video interview.
However, the risk associated with inactivity was attenuated for those patients who became more active over the course of the study period; their risk was still higher than that of those who had sustained activity, but lower than those who remained inactive. “It’s never too late to start being physically active,” said Dr. Moholdt.
All groups were compared with individuals who remained highly active over the study period; this reference group had the lowest risk of both cardiovascular and all-cause death.
Dr. Moholdt noted a limitation of most previous research into how physical activity relates to cardiovascular and all-cause mortality: Data are obtained just at baseline, and then associated with a downstream outcome. “But people change!” she said, so it’s important to track how activity levels change over time.
To that end, the Nord-Trøndelag Health Study (HUNT study) measured activity levels for 23,156 participants at two points, and then assessed cardiovascular and all-cause mortality, over a period of 22 years.
Dr. Moholdt, head of the Exercise, Cardiometabolic Health and Reproduction research group at Norwegian University of Science and Technology, Trondheim, and her collaborators asked individuals about physical activity in 1985, and again in 2007. At each time point, investigators asked participants to report how many hours per week they spent in “mosjon,” the Norwegian word for “motion”; Dr. Moholdt explained that the word carries the connotation of purposeful, health-promoting physical activity.
Individuals were grouped into one of three activity levels at each time point: Inactive participants reported no purposeful physical activity, moderately active participants reported less than 2 hours per week of this activity, and the most highly active group was active for 2 or more hours per week.
Patients who were in each of the three activity groupings at baseline might have changed their activity levels by the second assessment, so the investigators tracked a total of nine categories of exercise trends over time.
The mean age of participants in the Nord-Trøndelag Health Study (HUNT study) was 39 years, and the mean body mass index was 24 kg/m2. Of the 23,156 participants, 12,665 were women. Dr. Moholdt and her colleagues used statistical analysis to adjust for age, sex, body mass index, smoking, educational status, and systolic blood pressure.
However, Dr. Moholdt acknowledged at the press conference that comorbidities not captured in the study design might impact changes in activity levels, particularly for formerly highly active patients who became inactive over the study period.
The study was presented in a poster session at the congress. Dr. Moholdt reported no conflicts of interest. The study was funded by Health Central Norway (Helse-Midt Norge), a state entity that operates health care facilities in the region of Norway where the HUNT study was conducted.
SOURCE: Moholdt T. et al. ESC Congress 2019, Abstract P627.
PARIS – , according to results from a prospective cohort study that followed more than 23,000 adults for over 2 decades.
“Individuals who remained physically inactive, or who decreased their physical activity over 22 years, had substantially increased risk of dying from all causes, and from cardiovascular disease,” Trine Moholdt, PhD, said at a press conference at the annual congress of the European Society of Cardiology.
“The bad news is that sustained inactivity was associated with a 99% increase in all-cause mortality and 168% increase in cardiovascular deaths” compared with sustained physical activity, she said.
The news was similarly bad for individuals who had been highly active and then became inactive; this cohort had an increase in all-cause mortality of 116% and sustained a 173% increase in cardiovascular deaths. “Previous activity levels – they don’t count. You have to keep it up,” said Dr. Moholdt, speaking in a video interview.
However, the risk associated with inactivity was attenuated for those patients who became more active over the course of the study period; their risk was still higher than that of those who had sustained activity, but lower than those who remained inactive. “It’s never too late to start being physically active,” said Dr. Moholdt.
All groups were compared with individuals who remained highly active over the study period; this reference group had the lowest risk of both cardiovascular and all-cause death.
Dr. Moholdt noted a limitation of most previous research into how physical activity relates to cardiovascular and all-cause mortality: Data are obtained just at baseline, and then associated with a downstream outcome. “But people change!” she said, so it’s important to track how activity levels change over time.
To that end, the Nord-Trøndelag Health Study (HUNT study) measured activity levels for 23,156 participants at two points, and then assessed cardiovascular and all-cause mortality, over a period of 22 years.
Dr. Moholdt, head of the Exercise, Cardiometabolic Health and Reproduction research group at Norwegian University of Science and Technology, Trondheim, and her collaborators asked individuals about physical activity in 1985, and again in 2007. At each time point, investigators asked participants to report how many hours per week they spent in “mosjon,” the Norwegian word for “motion”; Dr. Moholdt explained that the word carries the connotation of purposeful, health-promoting physical activity.
Individuals were grouped into one of three activity levels at each time point: Inactive participants reported no purposeful physical activity, moderately active participants reported less than 2 hours per week of this activity, and the most highly active group was active for 2 or more hours per week.
Patients who were in each of the three activity groupings at baseline might have changed their activity levels by the second assessment, so the investigators tracked a total of nine categories of exercise trends over time.
The mean age of participants in the Nord-Trøndelag Health Study (HUNT study) was 39 years, and the mean body mass index was 24 kg/m2. Of the 23,156 participants, 12,665 were women. Dr. Moholdt and her colleagues used statistical analysis to adjust for age, sex, body mass index, smoking, educational status, and systolic blood pressure.
However, Dr. Moholdt acknowledged at the press conference that comorbidities not captured in the study design might impact changes in activity levels, particularly for formerly highly active patients who became inactive over the study period.
The study was presented in a poster session at the congress. Dr. Moholdt reported no conflicts of interest. The study was funded by Health Central Norway (Helse-Midt Norge), a state entity that operates health care facilities in the region of Norway where the HUNT study was conducted.
SOURCE: Moholdt T. et al. ESC Congress 2019, Abstract P627.
AT THE ESC CONGRESS 2019
Vaping-related lung disease cases rise, case reporting standardized
The number of possible cases of vaping-related pulmonary illness has risen to 215, reported from 25 states, as of Aug. 27, 2019, according to the Centers for Disease Control and Prevention, Atlanta. Additional reports of pulmonary illness are under investigation.
The CDC has released a standardized case definition that states are using to complete their own investigations and verifications of cases. It appears that all cases are linked to e-cigarette product use, but the cause of the respiratory illnesses is still unconfirmed.
In many cases, patients reported a gradual start of symptoms, including breathing difficulty, shortness of breath, and/or chest pain before hospitalization. Some cases reported mild to moderate gastrointestinal illness including vomiting and diarrhea, or other symptoms such as fevers or fatigue. In many cases, patients have also acknowledged recent use of tetrahydrocannabinol (THC)-containing e-cigarette products while speaking to health care personnel or in follow-up interviews by health department staff, according to a statement from the CDC and the Food and Drug Administration.
The agencies are working with state health departments to standardize information collection at the state level to help build a more comprehensive picture of these incidents, including the brand and types of e-cigarette products, whether any of them would fall within the FDA’s regulatory authority, where they were obtained, and whether there is a link to specific devices, ingredients, or contaminants in the devices or substances associated with e-cigarette product use.
CDC staff have been deployed to Illinois and Wisconsin to assist their state health departments. The agencies have released a Clinician Outreach and Communication Activity (COCA) Clinical Action Alert describing this investigation and asking providers to report possible cases to their state health departments. In addition to a standardized case definition, the agencies have issued a medical chart abstraction form and case interview questionnaire, are reviewing and providing feedback on data collection and health messaging tools for states, and are facilitating information sharing between states with possible cases.
More information on the cases and reporting are available from the CDC.
The number of possible cases of vaping-related pulmonary illness has risen to 215, reported from 25 states, as of Aug. 27, 2019, according to the Centers for Disease Control and Prevention, Atlanta. Additional reports of pulmonary illness are under investigation.
The CDC has released a standardized case definition that states are using to complete their own investigations and verifications of cases. It appears that all cases are linked to e-cigarette product use, but the cause of the respiratory illnesses is still unconfirmed.
In many cases, patients reported a gradual start of symptoms, including breathing difficulty, shortness of breath, and/or chest pain before hospitalization. Some cases reported mild to moderate gastrointestinal illness including vomiting and diarrhea, or other symptoms such as fevers or fatigue. In many cases, patients have also acknowledged recent use of tetrahydrocannabinol (THC)-containing e-cigarette products while speaking to health care personnel or in follow-up interviews by health department staff, according to a statement from the CDC and the Food and Drug Administration.
The agencies are working with state health departments to standardize information collection at the state level to help build a more comprehensive picture of these incidents, including the brand and types of e-cigarette products, whether any of them would fall within the FDA’s regulatory authority, where they were obtained, and whether there is a link to specific devices, ingredients, or contaminants in the devices or substances associated with e-cigarette product use.
CDC staff have been deployed to Illinois and Wisconsin to assist their state health departments. The agencies have released a Clinician Outreach and Communication Activity (COCA) Clinical Action Alert describing this investigation and asking providers to report possible cases to their state health departments. In addition to a standardized case definition, the agencies have issued a medical chart abstraction form and case interview questionnaire, are reviewing and providing feedback on data collection and health messaging tools for states, and are facilitating information sharing between states with possible cases.
More information on the cases and reporting are available from the CDC.
The number of possible cases of vaping-related pulmonary illness has risen to 215, reported from 25 states, as of Aug. 27, 2019, according to the Centers for Disease Control and Prevention, Atlanta. Additional reports of pulmonary illness are under investigation.
The CDC has released a standardized case definition that states are using to complete their own investigations and verifications of cases. It appears that all cases are linked to e-cigarette product use, but the cause of the respiratory illnesses is still unconfirmed.
In many cases, patients reported a gradual start of symptoms, including breathing difficulty, shortness of breath, and/or chest pain before hospitalization. Some cases reported mild to moderate gastrointestinal illness including vomiting and diarrhea, or other symptoms such as fevers or fatigue. In many cases, patients have also acknowledged recent use of tetrahydrocannabinol (THC)-containing e-cigarette products while speaking to health care personnel or in follow-up interviews by health department staff, according to a statement from the CDC and the Food and Drug Administration.
The agencies are working with state health departments to standardize information collection at the state level to help build a more comprehensive picture of these incidents, including the brand and types of e-cigarette products, whether any of them would fall within the FDA’s regulatory authority, where they were obtained, and whether there is a link to specific devices, ingredients, or contaminants in the devices or substances associated with e-cigarette product use.
CDC staff have been deployed to Illinois and Wisconsin to assist their state health departments. The agencies have released a Clinician Outreach and Communication Activity (COCA) Clinical Action Alert describing this investigation and asking providers to report possible cases to their state health departments. In addition to a standardized case definition, the agencies have issued a medical chart abstraction form and case interview questionnaire, are reviewing and providing feedback on data collection and health messaging tools for states, and are facilitating information sharing between states with possible cases.
More information on the cases and reporting are available from the CDC.
ABIM: Self-paced MOC pathway under development
Physician groups are praising a new option by the American Board of Internal Medicine (ABIM) that will offer doctors a self-paced pathway for maintenance of certification (MOC) in place of the traditional long-form assessment route.
The new longitudinal assessment option, announced in late August, would enable physicians to acquire and demonstrate ongoing knowledge through shorter evaluations of specific content. The option, currently under development, also would provide doctors with immediate feedback about their answers and share links to educational material to address knowledge gaps, according to an announcement. While details are still being flushed out, a summary of the longitudinal assessment concept by the American Board of Medical Specialties explains that the approach draws on the principles of adult learning and modern technology “to promote learning, retention, and transfer of information.”
Developing a longitudinal assessment option is part of ABIM’s ongoing evolution, Marianne M. Green, MD, chair for ABIM’s board of directors and ABIM President Richard J. Baron, MD, wrote in a joint letter to internists posted on ABIM’s blog.
“We recognize that some physicians may prefer a more continuous process that easily integrates into their lives and allows them to engage seamlessly at their preferred pace, while being able to access the resources they use in practice,” the doctors wrote.
Douglas DeLong, MD, chair of the American College of Physician’s (ACP) board of regents said the option is a positive, first step that will support lifelong learning. He noted the new option is in line with recommendations by the American Board of Medical Specialties’ Continuing Board Certification: Vision for the Future Commission, which included ACP concerns.
“It’s pretty clear that some of the principles of adult learning – frequent information with quick feedback, repetition of material, and identifying gaps in knowledge – is really how people most effectively learn,” Dr. DeLong said in an interview. “Just cramming for an examination every decade hasn’t ever really been shown to affect long-term retention of knowledge or even patient care outcomes.”
Alan Lichtin, MD, chair of the MOC working group for the American Society of Hematology (ASH), said the self-paced pathway is a much-needed option, particularly the immediate feedback on test questions.
“For years, ASH has been advocating that ABIM move from the traditional sit-down testing to an alternative form of ‘formative’ assessment that has been adapted by other specialty boards,” Dr. Lichtin said in an interview. Anesthesiology and pediatrics have novel testing methods that fit into physicians’ schedules without being so disruptive and anxiety provoking. There is instantaneous feedback about whether the answers are correct or not. It is not useful to study hard for a time-intensive, comprehensive test only to get a summary of what was missed a long time after the test. By that point, the exam material is no longer fresh in one’s mind and therefore the feedback is no longer useful.”
The new pathway is still under development, and ABIM has not said when the option might be launched. In the meantime, the current MOC program and its traditional exam will remain in effect. The board is requesting feedback and comments from physicians about the option. Dr. Baron wrote that more information about the change will be forthcoming in the months ahead.
The ABIM announcement comes on the heels of an ongoing legal challenge levied at the board by a group of internists over its MOC process.
The lawsuit, filed Dec. 6, 2018, in Pennsylvania district court and later amended in 2019, claims that ABIM is charging inflated monopoly prices for maintaining certification, that the organization is forcing physicians to purchase MOC, and that ABIM is inducing employers and others to require ABIM certification. The four plaintiff-physicians are asking a judge to find ABIM in violation of federal antitrust law and to bar the board from continuing its MOC process. The suit is filed as a class action on behalf of all internists and subspecialists required by ABIM to purchase MOC to maintain their ABIM certifications. .
Two other lawsuits challenging MOC, one against the American Board of Psychiatry and Neurology and another against the American Board of Radiology, are ongoing. A fourth lawsuit against the American Board of Medical Specialties, the American Board of Emergency Medicine, and the American Board of Anesthesiology was filed in February.
Attorneys for all three boards in the ABIM, American Board of Psychiatry and Neurology, and American Board of Radiology cases are seeking to dismiss the complaints. Judges have not yet ruled on the motions. In addition, a motion to consolidate all the cases was denied by the court.
A GoFundMe campaign launched by the Practicing Physicians of America to pay for plaintiffs’ costs associated with the class-action lawsuits has now garnered more than $300,000.
Physician groups are praising a new option by the American Board of Internal Medicine (ABIM) that will offer doctors a self-paced pathway for maintenance of certification (MOC) in place of the traditional long-form assessment route.
The new longitudinal assessment option, announced in late August, would enable physicians to acquire and demonstrate ongoing knowledge through shorter evaluations of specific content. The option, currently under development, also would provide doctors with immediate feedback about their answers and share links to educational material to address knowledge gaps, according to an announcement. While details are still being flushed out, a summary of the longitudinal assessment concept by the American Board of Medical Specialties explains that the approach draws on the principles of adult learning and modern technology “to promote learning, retention, and transfer of information.”
Developing a longitudinal assessment option is part of ABIM’s ongoing evolution, Marianne M. Green, MD, chair for ABIM’s board of directors and ABIM President Richard J. Baron, MD, wrote in a joint letter to internists posted on ABIM’s blog.
“We recognize that some physicians may prefer a more continuous process that easily integrates into their lives and allows them to engage seamlessly at their preferred pace, while being able to access the resources they use in practice,” the doctors wrote.
Douglas DeLong, MD, chair of the American College of Physician’s (ACP) board of regents said the option is a positive, first step that will support lifelong learning. He noted the new option is in line with recommendations by the American Board of Medical Specialties’ Continuing Board Certification: Vision for the Future Commission, which included ACP concerns.
“It’s pretty clear that some of the principles of adult learning – frequent information with quick feedback, repetition of material, and identifying gaps in knowledge – is really how people most effectively learn,” Dr. DeLong said in an interview. “Just cramming for an examination every decade hasn’t ever really been shown to affect long-term retention of knowledge or even patient care outcomes.”
Alan Lichtin, MD, chair of the MOC working group for the American Society of Hematology (ASH), said the self-paced pathway is a much-needed option, particularly the immediate feedback on test questions.
“For years, ASH has been advocating that ABIM move from the traditional sit-down testing to an alternative form of ‘formative’ assessment that has been adapted by other specialty boards,” Dr. Lichtin said in an interview. Anesthesiology and pediatrics have novel testing methods that fit into physicians’ schedules without being so disruptive and anxiety provoking. There is instantaneous feedback about whether the answers are correct or not. It is not useful to study hard for a time-intensive, comprehensive test only to get a summary of what was missed a long time after the test. By that point, the exam material is no longer fresh in one’s mind and therefore the feedback is no longer useful.”
The new pathway is still under development, and ABIM has not said when the option might be launched. In the meantime, the current MOC program and its traditional exam will remain in effect. The board is requesting feedback and comments from physicians about the option. Dr. Baron wrote that more information about the change will be forthcoming in the months ahead.
The ABIM announcement comes on the heels of an ongoing legal challenge levied at the board by a group of internists over its MOC process.
The lawsuit, filed Dec. 6, 2018, in Pennsylvania district court and later amended in 2019, claims that ABIM is charging inflated monopoly prices for maintaining certification, that the organization is forcing physicians to purchase MOC, and that ABIM is inducing employers and others to require ABIM certification. The four plaintiff-physicians are asking a judge to find ABIM in violation of federal antitrust law and to bar the board from continuing its MOC process. The suit is filed as a class action on behalf of all internists and subspecialists required by ABIM to purchase MOC to maintain their ABIM certifications. .
Two other lawsuits challenging MOC, one against the American Board of Psychiatry and Neurology and another against the American Board of Radiology, are ongoing. A fourth lawsuit against the American Board of Medical Specialties, the American Board of Emergency Medicine, and the American Board of Anesthesiology was filed in February.
Attorneys for all three boards in the ABIM, American Board of Psychiatry and Neurology, and American Board of Radiology cases are seeking to dismiss the complaints. Judges have not yet ruled on the motions. In addition, a motion to consolidate all the cases was denied by the court.
A GoFundMe campaign launched by the Practicing Physicians of America to pay for plaintiffs’ costs associated with the class-action lawsuits has now garnered more than $300,000.
Physician groups are praising a new option by the American Board of Internal Medicine (ABIM) that will offer doctors a self-paced pathway for maintenance of certification (MOC) in place of the traditional long-form assessment route.
The new longitudinal assessment option, announced in late August, would enable physicians to acquire and demonstrate ongoing knowledge through shorter evaluations of specific content. The option, currently under development, also would provide doctors with immediate feedback about their answers and share links to educational material to address knowledge gaps, according to an announcement. While details are still being flushed out, a summary of the longitudinal assessment concept by the American Board of Medical Specialties explains that the approach draws on the principles of adult learning and modern technology “to promote learning, retention, and transfer of information.”
Developing a longitudinal assessment option is part of ABIM’s ongoing evolution, Marianne M. Green, MD, chair for ABIM’s board of directors and ABIM President Richard J. Baron, MD, wrote in a joint letter to internists posted on ABIM’s blog.
“We recognize that some physicians may prefer a more continuous process that easily integrates into their lives and allows them to engage seamlessly at their preferred pace, while being able to access the resources they use in practice,” the doctors wrote.
Douglas DeLong, MD, chair of the American College of Physician’s (ACP) board of regents said the option is a positive, first step that will support lifelong learning. He noted the new option is in line with recommendations by the American Board of Medical Specialties’ Continuing Board Certification: Vision for the Future Commission, which included ACP concerns.
“It’s pretty clear that some of the principles of adult learning – frequent information with quick feedback, repetition of material, and identifying gaps in knowledge – is really how people most effectively learn,” Dr. DeLong said in an interview. “Just cramming for an examination every decade hasn’t ever really been shown to affect long-term retention of knowledge or even patient care outcomes.”
Alan Lichtin, MD, chair of the MOC working group for the American Society of Hematology (ASH), said the self-paced pathway is a much-needed option, particularly the immediate feedback on test questions.
“For years, ASH has been advocating that ABIM move from the traditional sit-down testing to an alternative form of ‘formative’ assessment that has been adapted by other specialty boards,” Dr. Lichtin said in an interview. Anesthesiology and pediatrics have novel testing methods that fit into physicians’ schedules without being so disruptive and anxiety provoking. There is instantaneous feedback about whether the answers are correct or not. It is not useful to study hard for a time-intensive, comprehensive test only to get a summary of what was missed a long time after the test. By that point, the exam material is no longer fresh in one’s mind and therefore the feedback is no longer useful.”
The new pathway is still under development, and ABIM has not said when the option might be launched. In the meantime, the current MOC program and its traditional exam will remain in effect. The board is requesting feedback and comments from physicians about the option. Dr. Baron wrote that more information about the change will be forthcoming in the months ahead.
The ABIM announcement comes on the heels of an ongoing legal challenge levied at the board by a group of internists over its MOC process.
The lawsuit, filed Dec. 6, 2018, in Pennsylvania district court and later amended in 2019, claims that ABIM is charging inflated monopoly prices for maintaining certification, that the organization is forcing physicians to purchase MOC, and that ABIM is inducing employers and others to require ABIM certification. The four plaintiff-physicians are asking a judge to find ABIM in violation of federal antitrust law and to bar the board from continuing its MOC process. The suit is filed as a class action on behalf of all internists and subspecialists required by ABIM to purchase MOC to maintain their ABIM certifications. .
Two other lawsuits challenging MOC, one against the American Board of Psychiatry and Neurology and another against the American Board of Radiology, are ongoing. A fourth lawsuit against the American Board of Medical Specialties, the American Board of Emergency Medicine, and the American Board of Anesthesiology was filed in February.
Attorneys for all three boards in the ABIM, American Board of Psychiatry and Neurology, and American Board of Radiology cases are seeking to dismiss the complaints. Judges have not yet ruled on the motions. In addition, a motion to consolidate all the cases was denied by the court.
A GoFundMe campaign launched by the Practicing Physicians of America to pay for plaintiffs’ costs associated with the class-action lawsuits has now garnered more than $300,000.
Surviving Colorectal Cancer, Now at Risk for Hypertension
Colorectal cancer (CRC) survivor rates are improving, which means people are living long enough after the cancer to have other chronic conditions. CRC is the third most commonly diagnosed cancer among users of the US Department of Veterans Affairs (VA) health care system, according to VA researchers, and there is a high prevalence of cardiovascular disease (CVD). The researchers also say emerging evidence suggests that survivors of CRC may be more likely to develop diabetes mellitus (DM) in the 5 years following their cancer diagnosis. But they add that there is a paucity of research about control of CVD-related chronic conditions among survivors of CRC.
In a retrospective study, the researchers compared 9,758 nonmetastatic patients with CRC with 29,066 people who had not had cancer. At baseline, 69% of the survivors of CRC and the matched controls were diagnosed with hypertension, 52% with hyperlipidemia, and 37% with DM.
But somewhat contrary to expectations, the researchers found no significant differences between the 2 groups for DM in the year following the baseline assessment. The researchers point to the VA’s “strong history” of DM risk reduction research and 2 national programs targeting DM, although they do not know whether the people in their study participated in those.
The survivors of CRC also had half the odds of being diagnosed with hyperlipidemia. However, they did have 57% higher odds of being diagnosed with hypertension.
Although the researchers acknowledge that hypertension is a transient adverse effect of certain chemotherapy regimens, they found only 7 survivors of CRC and 11 controls were treated with bevacizumab during their first year postanchor date.
The relationship between nonmetastatic CRC and CVD risk-related chronic conditions is complex, the researchers say. But they share risk factors, including obesity, physical inactivity, and diet.
The researchers call behavioral change interventions that improve survivors of CRC physical activity, dietary habits, and body mass index a “promising beginning” but call for other similar interventions, particularly those targeting blood pressure management and adherence to antihypertensive medications (which was significantly lower among the survivors).
While the magnitude of the effect regarding hypertension seems relatively small, the researchers say, they believe it is still an important difference when considered from a population health perspective—and one that should be addressed. The researchers also note that nonmetastatic survivors of CRC and controls had very similar rates of primary care visits in the 3 years postanchor date and as a result similar opportunities to receive a hypertension diagnosis.
Colorectal cancer (CRC) survivor rates are improving, which means people are living long enough after the cancer to have other chronic conditions. CRC is the third most commonly diagnosed cancer among users of the US Department of Veterans Affairs (VA) health care system, according to VA researchers, and there is a high prevalence of cardiovascular disease (CVD). The researchers also say emerging evidence suggests that survivors of CRC may be more likely to develop diabetes mellitus (DM) in the 5 years following their cancer diagnosis. But they add that there is a paucity of research about control of CVD-related chronic conditions among survivors of CRC.
In a retrospective study, the researchers compared 9,758 nonmetastatic patients with CRC with 29,066 people who had not had cancer. At baseline, 69% of the survivors of CRC and the matched controls were diagnosed with hypertension, 52% with hyperlipidemia, and 37% with DM.
But somewhat contrary to expectations, the researchers found no significant differences between the 2 groups for DM in the year following the baseline assessment. The researchers point to the VA’s “strong history” of DM risk reduction research and 2 national programs targeting DM, although they do not know whether the people in their study participated in those.
The survivors of CRC also had half the odds of being diagnosed with hyperlipidemia. However, they did have 57% higher odds of being diagnosed with hypertension.
Although the researchers acknowledge that hypertension is a transient adverse effect of certain chemotherapy regimens, they found only 7 survivors of CRC and 11 controls were treated with bevacizumab during their first year postanchor date.
The relationship between nonmetastatic CRC and CVD risk-related chronic conditions is complex, the researchers say. But they share risk factors, including obesity, physical inactivity, and diet.
The researchers call behavioral change interventions that improve survivors of CRC physical activity, dietary habits, and body mass index a “promising beginning” but call for other similar interventions, particularly those targeting blood pressure management and adherence to antihypertensive medications (which was significantly lower among the survivors).
While the magnitude of the effect regarding hypertension seems relatively small, the researchers say, they believe it is still an important difference when considered from a population health perspective—and one that should be addressed. The researchers also note that nonmetastatic survivors of CRC and controls had very similar rates of primary care visits in the 3 years postanchor date and as a result similar opportunities to receive a hypertension diagnosis.
Colorectal cancer (CRC) survivor rates are improving, which means people are living long enough after the cancer to have other chronic conditions. CRC is the third most commonly diagnosed cancer among users of the US Department of Veterans Affairs (VA) health care system, according to VA researchers, and there is a high prevalence of cardiovascular disease (CVD). The researchers also say emerging evidence suggests that survivors of CRC may be more likely to develop diabetes mellitus (DM) in the 5 years following their cancer diagnosis. But they add that there is a paucity of research about control of CVD-related chronic conditions among survivors of CRC.
In a retrospective study, the researchers compared 9,758 nonmetastatic patients with CRC with 29,066 people who had not had cancer. At baseline, 69% of the survivors of CRC and the matched controls were diagnosed with hypertension, 52% with hyperlipidemia, and 37% with DM.
But somewhat contrary to expectations, the researchers found no significant differences between the 2 groups for DM in the year following the baseline assessment. The researchers point to the VA’s “strong history” of DM risk reduction research and 2 national programs targeting DM, although they do not know whether the people in their study participated in those.
The survivors of CRC also had half the odds of being diagnosed with hyperlipidemia. However, they did have 57% higher odds of being diagnosed with hypertension.
Although the researchers acknowledge that hypertension is a transient adverse effect of certain chemotherapy regimens, they found only 7 survivors of CRC and 11 controls were treated with bevacizumab during their first year postanchor date.
The relationship between nonmetastatic CRC and CVD risk-related chronic conditions is complex, the researchers say. But they share risk factors, including obesity, physical inactivity, and diet.
The researchers call behavioral change interventions that improve survivors of CRC physical activity, dietary habits, and body mass index a “promising beginning” but call for other similar interventions, particularly those targeting blood pressure management and adherence to antihypertensive medications (which was significantly lower among the survivors).
While the magnitude of the effect regarding hypertension seems relatively small, the researchers say, they believe it is still an important difference when considered from a population health perspective—and one that should be addressed. The researchers also note that nonmetastatic survivors of CRC and controls had very similar rates of primary care visits in the 3 years postanchor date and as a result similar opportunities to receive a hypertension diagnosis.
Often Off-label: Questionable Gabapentinoid Use Noted at Hospital Admission Warrants Deprescribing
Three years after gabapentin received US Food and Drug Administration (FDA) approval in 1990 for epilepsy, case reports and animal studies emerged announcing its potential in the treatment of pain syndromes through then-novel analgesic mechanisms.1 Fast forward 20 years to 2016: gabapentin and its close cousin, pregabalin, are internationally considered first-line agents for the treatment of neuropathic pain in guidelines from the Centers for Disease Control and Prevention, the Canadian Pain Society, and the National Institute for Health and Care Excellence. Gabapentin is the 10th most prescribed drug in the United States, and brand-name pregabalin sales were $4.4 billion USD, ranking 8th in invoice drug spending.2
The ascendancy of gabapentinoids as drugs of choice for pain, though, is fraught with controversy; yet, they were shepherded to commercial success. In 2004, the patent owner of gabapentin, Warner-Lambert (now owned by Pfizer), admitted guilt to charges that it violated federal regulations in its promotion: they encouraged off-label prescribing through paid physician-to-physician communications, publication of positive outcomes, and suppression of negative ones.3 Pfizer paid another settlement in 2009 for false claims about off-label indications for brand-name pregabalin.4
Mindful of historical biases, recent trials and meta-analyses have found less favorable outcomes for gabapentinoids in the treatment of off-label pain conditions and greater risks than previously reported. Cochrane reviews for gabapentin demonstrate efficacy only in postherpetic neuralgia (for which it has FDA approval) and diabetic peripheral neuropathy (for which it does not); pregabalin has efficacy in both these conditions as well as posttraumatic neuropathic pain and fibromyalgia (and FDA approval for all four). For other types of neuropathic pain, the evidence is of lower quality. Even for approved indications, the risk–benefit ratio is questionable, as the numbers needed to harm for dizziness and somnolence are similar to the numbers needed to treat for pain.5,6 Further, case–control studies have found increased odds of opioid-related death when gabapentinoids were coprescribed with opioids,7,8 prompting gabapentinoids to be reclassified as class C controlled substances in the UK as of April 2019.9
On this backdrop, Gingras and colleagues publish their retrospective cohort study on high-risk prescribing of these popular drugs in Montreal, Canada in this issue of Journal of Hospital Medicine.10 In their retrospective cohort study of 4,103 patients admitted to a clinical teaching unit, more than one in eight patients (13%) were being prescribed a gabapentinoid as an outpatient; chart review of the admission notes indicated that only 17% of them had an FDA-approved indication and 28% had no clear indication. Gabapentinoid users were more likely to be coprescribed an opioid than nonusers (28% vs 12%). There was no significant difference in length of stay or inpatient death between users and nonusers.
Gingras et al. thereby conclude that there is an opportunity to deprescribe on the basis of few gabapentinoid users having a documented indication and the recent research showing poten
First, the urgency of deprescribing in inpatient settings should be titrated to the degree of risk. When the reason for hospitalization is potentially an adverse drug effect, culprit medications posing a substantial and near-term risk of harm should be stopped, such as when patients on gabapentinoids present with major alteration of their mental status.
In less urgent circumstances, hospitalists should speak first with outpatient prescribers because they may have important contextual information (eg, indication, patient preference, failure of alternative therapies, etc.) about previous care that the inpatient clinician lacks. For gabapentinoids, it is easy to imagine how treated pain syndromes without objective markers of disease may escape notice by a hospitalist and remain undocumented, which may encourage erroneous deprescribing. If the shared decision between the patient and providers is to deprescribe, patients on high doses warrant a tapering schedule.11 Pharmacist consultation can help with this.
Second, before discharge, hospitalists should communicate their rationale for deprescribing medications to both patients and outpatient prescribers, especially if a prolonged tapering schedule is required. This type of communication occurred infrequently in this study: the reason for deprescribing a gabapentinoid was missing from the discharge summary 55% of the time. Without this, outpatient prescribers may simply reinitiate the medication after the patient is discharged.
To counter the overuse of gabapentinoids, hospitalists should look for opportunities to deprescribe them where there is concern about adverse events and when evidence-based indications do not exist. Successful deprescribing of these popular drugs will require deliberate collaboration and communication with the outpatient circle of care, as ongoing deprescribing ultimately depends on patients and outpatient prescribers agreeing to the change.
Disclosures
Dr. Steinman served as an unpaid expert witness in United States of America ex. Rel. David Franklin vs. Parke-Davis, Division of Warner-Lambert Company and Pfizer, Inc, litigation which alleged that the named pharmaceutical companies improperly marketed gabapentin for non-FDA-approved uses. Drs. Lam and Rochon have no conflicts of interest to declare.
Funding
Dr. Rochon is supported by the Retired Teachers of Ontario (RTO/ERO) Chair in Geriatric Medicine at the University of Toronto. Dr. Steinman is supported by the National Institute on Aging, US (K24AG049057 and P30AG044281).
1. Segal AZ, Rordorf G. Gabapentin as a novel treatment for postherpetic neuralgia. Neurology. 1996;46(4):1175-1176. https://doi.org/10.1212/WNL.46.4.1175.
2. Goodman CW, Brett AS. Gabapentin and pregabalin for pain — is increased prescribing a cause for concern? N Engl J Med. 2017;377(5):411-414. https://doi.org/10.1056/NEJMp1704633.
3. Steinman MA, Bero LA, Chren M-M, Landefeld CS. Narrative review: the promotion of gabapentin: an analysis of internal industry documents. Ann Intern Med. 2006;145(4):284. https://doi.org/10.7326/0003-4819-145-4-200608150-00008.
4. Department of Justice, Office of Public Affairs. Justice Department Announces Largest Health Care Fraud Settlement in Its History. https://www.justice.gov/opa/pr/justice-department-announces-largest-health-care-fraud-settlement-its-history. Published September 2, 2009. Accessed April 12, 2019.
5. Wiffen PJ, Derry S, Bell RF, et al. Gabapentin for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2017;6:CD007938. https://doi.org/10.1002/14651858.CD007938.pub4.
6. Derry S, Bell RF, Straube S, Wiffen PJ, Aldington D, Moore RA. Pregabalin for neuropathic pain in adults. Cochrane Database Syst Rev. 2019;1:CD007076. https://doi.org/10.1002/14651858.CD007076.pub3.
7. Gomes T, Juurlink DN, Antoniou T, Mamdani MM, Paterson JM, van den Brink W. Gabapentin, opioids, and the risk of opioid-related death: a population-based nested case–control study. PLoS Med. 2017;14(10): e1002396. https://doi.org/10.1371/journal.pmed.1002396.
8. Gomes T, Greaves S, van den Brink W, et al. Pregabalin and the risk for opioid-related death: a nested case–control study. Ann Intern Med. 2018;169(10):732. https://doi.org/10.7326/M18-1136.
9. Mayor S. Pregabalin and gabapentin become controlled drugs to cut deaths from misuse. BMJ. 2018;363:k4364. https://doi.org/10.1136/bmj.k4364.
10. Gingras M-A, Lieu A, Papillon-Ferland L, Lee T, McDonald E. Retrospective cohort study of the prevalence of off-label gabapentinoid prescriptions in hospitalized medical patients. J Hosp Med. 2019;14(9):547-550. https://doi.org/10.12788/jhm.3203.
11. Parsons G. Guide to the management of gabapentinoid misuse. Prescriber. 2018;29(4):25-30. https://doi.org/10.1002/psb.1664.
Three years after gabapentin received US Food and Drug Administration (FDA) approval in 1990 for epilepsy, case reports and animal studies emerged announcing its potential in the treatment of pain syndromes through then-novel analgesic mechanisms.1 Fast forward 20 years to 2016: gabapentin and its close cousin, pregabalin, are internationally considered first-line agents for the treatment of neuropathic pain in guidelines from the Centers for Disease Control and Prevention, the Canadian Pain Society, and the National Institute for Health and Care Excellence. Gabapentin is the 10th most prescribed drug in the United States, and brand-name pregabalin sales were $4.4 billion USD, ranking 8th in invoice drug spending.2
The ascendancy of gabapentinoids as drugs of choice for pain, though, is fraught with controversy; yet, they were shepherded to commercial success. In 2004, the patent owner of gabapentin, Warner-Lambert (now owned by Pfizer), admitted guilt to charges that it violated federal regulations in its promotion: they encouraged off-label prescribing through paid physician-to-physician communications, publication of positive outcomes, and suppression of negative ones.3 Pfizer paid another settlement in 2009 for false claims about off-label indications for brand-name pregabalin.4
Mindful of historical biases, recent trials and meta-analyses have found less favorable outcomes for gabapentinoids in the treatment of off-label pain conditions and greater risks than previously reported. Cochrane reviews for gabapentin demonstrate efficacy only in postherpetic neuralgia (for which it has FDA approval) and diabetic peripheral neuropathy (for which it does not); pregabalin has efficacy in both these conditions as well as posttraumatic neuropathic pain and fibromyalgia (and FDA approval for all four). For other types of neuropathic pain, the evidence is of lower quality. Even for approved indications, the risk–benefit ratio is questionable, as the numbers needed to harm for dizziness and somnolence are similar to the numbers needed to treat for pain.5,6 Further, case–control studies have found increased odds of opioid-related death when gabapentinoids were coprescribed with opioids,7,8 prompting gabapentinoids to be reclassified as class C controlled substances in the UK as of April 2019.9
On this backdrop, Gingras and colleagues publish their retrospective cohort study on high-risk prescribing of these popular drugs in Montreal, Canada in this issue of Journal of Hospital Medicine.10 In their retrospective cohort study of 4,103 patients admitted to a clinical teaching unit, more than one in eight patients (13%) were being prescribed a gabapentinoid as an outpatient; chart review of the admission notes indicated that only 17% of them had an FDA-approved indication and 28% had no clear indication. Gabapentinoid users were more likely to be coprescribed an opioid than nonusers (28% vs 12%). There was no significant difference in length of stay or inpatient death between users and nonusers.
Gingras et al. thereby conclude that there is an opportunity to deprescribe on the basis of few gabapentinoid users having a documented indication and the recent research showing poten
First, the urgency of deprescribing in inpatient settings should be titrated to the degree of risk. When the reason for hospitalization is potentially an adverse drug effect, culprit medications posing a substantial and near-term risk of harm should be stopped, such as when patients on gabapentinoids present with major alteration of their mental status.
In less urgent circumstances, hospitalists should speak first with outpatient prescribers because they may have important contextual information (eg, indication, patient preference, failure of alternative therapies, etc.) about previous care that the inpatient clinician lacks. For gabapentinoids, it is easy to imagine how treated pain syndromes without objective markers of disease may escape notice by a hospitalist and remain undocumented, which may encourage erroneous deprescribing. If the shared decision between the patient and providers is to deprescribe, patients on high doses warrant a tapering schedule.11 Pharmacist consultation can help with this.
Second, before discharge, hospitalists should communicate their rationale for deprescribing medications to both patients and outpatient prescribers, especially if a prolonged tapering schedule is required. This type of communication occurred infrequently in this study: the reason for deprescribing a gabapentinoid was missing from the discharge summary 55% of the time. Without this, outpatient prescribers may simply reinitiate the medication after the patient is discharged.
To counter the overuse of gabapentinoids, hospitalists should look for opportunities to deprescribe them where there is concern about adverse events and when evidence-based indications do not exist. Successful deprescribing of these popular drugs will require deliberate collaboration and communication with the outpatient circle of care, as ongoing deprescribing ultimately depends on patients and outpatient prescribers agreeing to the change.
Disclosures
Dr. Steinman served as an unpaid expert witness in United States of America ex. Rel. David Franklin vs. Parke-Davis, Division of Warner-Lambert Company and Pfizer, Inc, litigation which alleged that the named pharmaceutical companies improperly marketed gabapentin for non-FDA-approved uses. Drs. Lam and Rochon have no conflicts of interest to declare.
Funding
Dr. Rochon is supported by the Retired Teachers of Ontario (RTO/ERO) Chair in Geriatric Medicine at the University of Toronto. Dr. Steinman is supported by the National Institute on Aging, US (K24AG049057 and P30AG044281).
Three years after gabapentin received US Food and Drug Administration (FDA) approval in 1990 for epilepsy, case reports and animal studies emerged announcing its potential in the treatment of pain syndromes through then-novel analgesic mechanisms.1 Fast forward 20 years to 2016: gabapentin and its close cousin, pregabalin, are internationally considered first-line agents for the treatment of neuropathic pain in guidelines from the Centers for Disease Control and Prevention, the Canadian Pain Society, and the National Institute for Health and Care Excellence. Gabapentin is the 10th most prescribed drug in the United States, and brand-name pregabalin sales were $4.4 billion USD, ranking 8th in invoice drug spending.2
The ascendancy of gabapentinoids as drugs of choice for pain, though, is fraught with controversy; yet, they were shepherded to commercial success. In 2004, the patent owner of gabapentin, Warner-Lambert (now owned by Pfizer), admitted guilt to charges that it violated federal regulations in its promotion: they encouraged off-label prescribing through paid physician-to-physician communications, publication of positive outcomes, and suppression of negative ones.3 Pfizer paid another settlement in 2009 for false claims about off-label indications for brand-name pregabalin.4
Mindful of historical biases, recent trials and meta-analyses have found less favorable outcomes for gabapentinoids in the treatment of off-label pain conditions and greater risks than previously reported. Cochrane reviews for gabapentin demonstrate efficacy only in postherpetic neuralgia (for which it has FDA approval) and diabetic peripheral neuropathy (for which it does not); pregabalin has efficacy in both these conditions as well as posttraumatic neuropathic pain and fibromyalgia (and FDA approval for all four). For other types of neuropathic pain, the evidence is of lower quality. Even for approved indications, the risk–benefit ratio is questionable, as the numbers needed to harm for dizziness and somnolence are similar to the numbers needed to treat for pain.5,6 Further, case–control studies have found increased odds of opioid-related death when gabapentinoids were coprescribed with opioids,7,8 prompting gabapentinoids to be reclassified as class C controlled substances in the UK as of April 2019.9
On this backdrop, Gingras and colleagues publish their retrospective cohort study on high-risk prescribing of these popular drugs in Montreal, Canada in this issue of Journal of Hospital Medicine.10 In their retrospective cohort study of 4,103 patients admitted to a clinical teaching unit, more than one in eight patients (13%) were being prescribed a gabapentinoid as an outpatient; chart review of the admission notes indicated that only 17% of them had an FDA-approved indication and 28% had no clear indication. Gabapentinoid users were more likely to be coprescribed an opioid than nonusers (28% vs 12%). There was no significant difference in length of stay or inpatient death between users and nonusers.
Gingras et al. thereby conclude that there is an opportunity to deprescribe on the basis of few gabapentinoid users having a documented indication and the recent research showing poten
First, the urgency of deprescribing in inpatient settings should be titrated to the degree of risk. When the reason for hospitalization is potentially an adverse drug effect, culprit medications posing a substantial and near-term risk of harm should be stopped, such as when patients on gabapentinoids present with major alteration of their mental status.
In less urgent circumstances, hospitalists should speak first with outpatient prescribers because they may have important contextual information (eg, indication, patient preference, failure of alternative therapies, etc.) about previous care that the inpatient clinician lacks. For gabapentinoids, it is easy to imagine how treated pain syndromes without objective markers of disease may escape notice by a hospitalist and remain undocumented, which may encourage erroneous deprescribing. If the shared decision between the patient and providers is to deprescribe, patients on high doses warrant a tapering schedule.11 Pharmacist consultation can help with this.
Second, before discharge, hospitalists should communicate their rationale for deprescribing medications to both patients and outpatient prescribers, especially if a prolonged tapering schedule is required. This type of communication occurred infrequently in this study: the reason for deprescribing a gabapentinoid was missing from the discharge summary 55% of the time. Without this, outpatient prescribers may simply reinitiate the medication after the patient is discharged.
To counter the overuse of gabapentinoids, hospitalists should look for opportunities to deprescribe them where there is concern about adverse events and when evidence-based indications do not exist. Successful deprescribing of these popular drugs will require deliberate collaboration and communication with the outpatient circle of care, as ongoing deprescribing ultimately depends on patients and outpatient prescribers agreeing to the change.
Disclosures
Dr. Steinman served as an unpaid expert witness in United States of America ex. Rel. David Franklin vs. Parke-Davis, Division of Warner-Lambert Company and Pfizer, Inc, litigation which alleged that the named pharmaceutical companies improperly marketed gabapentin for non-FDA-approved uses. Drs. Lam and Rochon have no conflicts of interest to declare.
Funding
Dr. Rochon is supported by the Retired Teachers of Ontario (RTO/ERO) Chair in Geriatric Medicine at the University of Toronto. Dr. Steinman is supported by the National Institute on Aging, US (K24AG049057 and P30AG044281).
1. Segal AZ, Rordorf G. Gabapentin as a novel treatment for postherpetic neuralgia. Neurology. 1996;46(4):1175-1176. https://doi.org/10.1212/WNL.46.4.1175.
2. Goodman CW, Brett AS. Gabapentin and pregabalin for pain — is increased prescribing a cause for concern? N Engl J Med. 2017;377(5):411-414. https://doi.org/10.1056/NEJMp1704633.
3. Steinman MA, Bero LA, Chren M-M, Landefeld CS. Narrative review: the promotion of gabapentin: an analysis of internal industry documents. Ann Intern Med. 2006;145(4):284. https://doi.org/10.7326/0003-4819-145-4-200608150-00008.
4. Department of Justice, Office of Public Affairs. Justice Department Announces Largest Health Care Fraud Settlement in Its History. https://www.justice.gov/opa/pr/justice-department-announces-largest-health-care-fraud-settlement-its-history. Published September 2, 2009. Accessed April 12, 2019.
5. Wiffen PJ, Derry S, Bell RF, et al. Gabapentin for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2017;6:CD007938. https://doi.org/10.1002/14651858.CD007938.pub4.
6. Derry S, Bell RF, Straube S, Wiffen PJ, Aldington D, Moore RA. Pregabalin for neuropathic pain in adults. Cochrane Database Syst Rev. 2019;1:CD007076. https://doi.org/10.1002/14651858.CD007076.pub3.
7. Gomes T, Juurlink DN, Antoniou T, Mamdani MM, Paterson JM, van den Brink W. Gabapentin, opioids, and the risk of opioid-related death: a population-based nested case–control study. PLoS Med. 2017;14(10): e1002396. https://doi.org/10.1371/journal.pmed.1002396.
8. Gomes T, Greaves S, van den Brink W, et al. Pregabalin and the risk for opioid-related death: a nested case–control study. Ann Intern Med. 2018;169(10):732. https://doi.org/10.7326/M18-1136.
9. Mayor S. Pregabalin and gabapentin become controlled drugs to cut deaths from misuse. BMJ. 2018;363:k4364. https://doi.org/10.1136/bmj.k4364.
10. Gingras M-A, Lieu A, Papillon-Ferland L, Lee T, McDonald E. Retrospective cohort study of the prevalence of off-label gabapentinoid prescriptions in hospitalized medical patients. J Hosp Med. 2019;14(9):547-550. https://doi.org/10.12788/jhm.3203.
11. Parsons G. Guide to the management of gabapentinoid misuse. Prescriber. 2018;29(4):25-30. https://doi.org/10.1002/psb.1664.
1. Segal AZ, Rordorf G. Gabapentin as a novel treatment for postherpetic neuralgia. Neurology. 1996;46(4):1175-1176. https://doi.org/10.1212/WNL.46.4.1175.
2. Goodman CW, Brett AS. Gabapentin and pregabalin for pain — is increased prescribing a cause for concern? N Engl J Med. 2017;377(5):411-414. https://doi.org/10.1056/NEJMp1704633.
3. Steinman MA, Bero LA, Chren M-M, Landefeld CS. Narrative review: the promotion of gabapentin: an analysis of internal industry documents. Ann Intern Med. 2006;145(4):284. https://doi.org/10.7326/0003-4819-145-4-200608150-00008.
4. Department of Justice, Office of Public Affairs. Justice Department Announces Largest Health Care Fraud Settlement in Its History. https://www.justice.gov/opa/pr/justice-department-announces-largest-health-care-fraud-settlement-its-history. Published September 2, 2009. Accessed April 12, 2019.
5. Wiffen PJ, Derry S, Bell RF, et al. Gabapentin for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2017;6:CD007938. https://doi.org/10.1002/14651858.CD007938.pub4.
6. Derry S, Bell RF, Straube S, Wiffen PJ, Aldington D, Moore RA. Pregabalin for neuropathic pain in adults. Cochrane Database Syst Rev. 2019;1:CD007076. https://doi.org/10.1002/14651858.CD007076.pub3.
7. Gomes T, Juurlink DN, Antoniou T, Mamdani MM, Paterson JM, van den Brink W. Gabapentin, opioids, and the risk of opioid-related death: a population-based nested case–control study. PLoS Med. 2017;14(10): e1002396. https://doi.org/10.1371/journal.pmed.1002396.
8. Gomes T, Greaves S, van den Brink W, et al. Pregabalin and the risk for opioid-related death: a nested case–control study. Ann Intern Med. 2018;169(10):732. https://doi.org/10.7326/M18-1136.
9. Mayor S. Pregabalin and gabapentin become controlled drugs to cut deaths from misuse. BMJ. 2018;363:k4364. https://doi.org/10.1136/bmj.k4364.
10. Gingras M-A, Lieu A, Papillon-Ferland L, Lee T, McDonald E. Retrospective cohort study of the prevalence of off-label gabapentinoid prescriptions in hospitalized medical patients. J Hosp Med. 2019;14(9):547-550. https://doi.org/10.12788/jhm.3203.
11. Parsons G. Guide to the management of gabapentinoid misuse. Prescriber. 2018;29(4):25-30. https://doi.org/10.1002/psb.1664.
© 2019 Society of Hospital Medicine
The Hospitalist Imperative: Standardizing Best Practice across Expanding Healthcare Networks
Rapid dissemination and adoption of evidence-based guidelines remains a challenge despite studies showing that key evidence-based care processes improve outcomes in sepsis and heart failure.1 Hospital medicine was virtually founded on the premise that hospitalists would be champions of delivering high-quality care. Hospitalists are now dealing with a new challenge—unprecedented growth of healthcare systems because of mergers and acquisitions. The year 2018 was a banner time for healthcare mergers and acquisitions, with a total of 1,182, up 14% from 2017.2 These are in response to the belief that healthcare systems may better navigate the mixed reimbursement models of fee-for-service and fee-for-value by achieving a larger patient base and economies of scale. Hospitalists must now achieve consistent, evidence-based standards of care across larger networks by educating their colleagues (often separated by large geographic areas) to manifest durable changes in their group practice with demonstrable improvement in patient outcomes and cost savings.
The study by Yurso et al. focused on implementing an education program, which included standardized learning through Clinical Performance and Value (CPV) vignettes with process measurement and feedback for sepsis and heart failure.3 Sepsis and heart failure have been a focus for treatment standardization because of the associated morbidity, mortality, and high cost of care. The study by Yurso et al. is a prospective quasi-controlled cohort of hospitalists in eight hospitals who were matched with comparator hospitalists in six nonparticipating hospitals across the AdventHealth system. Measurement and feedback were provided using CPV vignettes. Over two years, hospitalists who participated improved CPV scores by 8%, compliance with the utilization of the three-hour sepsis bundle from 46.0% to 57.5%, and orders of essential medical treatment elements for heart failure from 58.2% to 72.1%. In year one, the average length of stay (LOS) observed/expected (O/E) rates dropped by 8% for participating hospitalists compared with 2.5% in the comparator group. By year two, cost O/E rates improved slightly resulting in cost savings. The authors concluded that CPV case simulation-based measurement and feedback helped drive improvements in evidence-based care, which was associated with lower costs and shorter LOS.
While studies using traditional didactic CME struggle to demonstrate changes in practice leading to improved patient outcomes,4 the study by Yurso et al. gives a glimpse into how simulation can be used to help improve clinical performance and measure adherence to best practice. A remarkably similar study used CPV for simulated patients with serial performance measurement and feedback for heart failure and pneumonia. The study showed reduced practice variation between hospitalists at 11 hospitals across four states and decreased LOS and readmissions. However, the sole clinical outcome was no change in in-house mortality.5 Another study using CPV training in breast cancer treatment demonstrated increased adherence to evidence-based practice standards and decreased variation in care between providers across four states.6 Of note, this study did not include clinical outcomes. These studies collectively imply that simulation training with interactive learning, educational feedback, repetitive practice, and curriculum integration has shown modest success in creating practice change and improving adherence to best practice standards. However, they have minimal measures of patient outcomes and fairly simple analyses for cost savings. Because the education is computer-based and feedback can be performed remotely, it can be deployed across large and diverse growing healthcare systems. To really move the needle, future research in the field of simulation should identify optimal simulation methods and be designed with more rigor to include patient and cost outcomes.
At Intermountain Healthcare, hospitalist expansion occurred through a strategic realignment from the different geographic regions into the One Intermountain model. This model is built on the commitment that our patients will receive the same high-quality, high-value care wherever they walk through our doors. We have found four substantive changes have been particularly powerful in spurring a group practice mentality toward standardizing best practice. One, hospitalists are now aligned across the system under a single operational leadership structure that encourages combined efforts to share best practices and develop and deploy strategic initiatives around them. Two, hospitalists continue to build on a culture of quality and measure what matters to patients. While Intermountain Healthcare has a long history of using quality improvement to achieve better patient outcomes and lower costs,7 the new structure is allowing our group to test novel methods including redesigned education to see what actually improves adherence to best practice. Three, the group knows where the system’s reimbursement is coming from; Intermountain Healthcare has transitioned to a larger percentage of capitation,8 currently about 40%, with a strong commitment to partner with services geared to transition patients home quickly and keep them at home. Four, the organization has created a structure of accountability and reporting; an executive-sponsored systemwide operating model has been designed to cut through system barriers being identified by the frontline, allowing them to be rapidly surfaced and then solved at the executive level through daily huddles.9
Innovative educational programs such as the one described in the study by Yurso et al. that help the busy hospitalist achieve improved adherence to best practice are likely to be an important component leading to improved outcomes, but only after a group has been structured for success. As hospitalist groups continue to act as a single effector arm for high-value care, this will help meet the expectations of our patients and deliver on the promise of our field.
Disclosures: Dr. Srivastava is a physician founder of the I- PASS Patient Safety Institute. His employer, Intermountain Healthcare owns his equity in the I-PASS Patient Safety Institute. Dr. Srivastava is supported in part by the Children’s Hospital Association for his work as an executive council member of the Pediatric Research in Inpatient Settings (PRIS) network. Dr. Srivastava has received monetary awards, honorariums, and travel reimbursement from multiple academic and professional organizations for talks about pediatric hospitalist research networks and quality of care. All other authors have nothing to disclose. No funding was provided for this editorial.
Disclosures
The authors have no disclosures of financial conflicts of interest.
Funding
Dr. Walke was supported an award from the Health Resources and Services Administration Geriatric Workforce Enhancement Program to the University of Pennsylvania (U1QHP28720).
1. Seymour, CW, Geston F, Prescott HC, et al. Time to treatment and mortality during mandated emergency care for sepsis. N Engl J Med. 2017;376(23):2235-2244. https://doi.org/10.1056/NEJMoa1703058.
2. Healthcare Finance. Lagasse J. Healthcare mergers and acquisitions had record year in 2018, up 14.4 percent.https://webcache.googleusercontent.com/search?q=cache:zoMrl9yoLokJ:https://www.healthcarefinancenews.com/news/healthcare-mergers-and-acquisitions-had-record-year-2018-144-percent+&cd=2&hl=en&ct=clnk&gl=us. Published January, 2019. Accessed April 26, 2019.
3. Yurso M, Box B, Burgon T, et al. Reducing unneeded clinical variation in sepsis and heart failure care to improve outcomes and reduce cost: a collaborative engagement with hospitalists in a multi-state system. J Hosp Med. 2019;14(9):542-546. https://doi.org/10.12788/jhm.3220.
4. Cervero RM, Gaines JK. The impact of CME on physician performance and patient health outcomes: an updated synthesis of systematic reviews. J Contin Educ Health Prof. 2015;35(2):131-138. https://doi.org/10.1002/chp.21290.
5. Weems L, Strong J, Plummer D, et al. A quality collaboration in heart failure and pneumonia inpatient care at Novant Health: standardizing hospitalist practices to improve patient care and system performance. Jt Comm J Qual Patient Saf. 2019;45(3):199-206. https://doi.org/10.1016/j.jcjq.2018.09.005.
6. Peabody JW, Paculdo DR, Tamondong-Lachica D, et al. Improving clinical practice using a novel engagement approach; measurement, benchmarking and feedback; a longitudinal study. J Clin Med Res. 2016;8(9):633-640. https://doi.org/10.14740/jocmr2620w.
7. James BC, Savitz LA. How Intermountain trimmed health care costs through robust quality improvement efforts. Health Aff (Millwood). 2011;30(6):1185-1191. https://doi.org/10.1377/hlthaff.2011.0358.
8. James BC, Poulsen GP. The case for capitation. Harv Bus Rev. 2016;94(7-8):102-111,134. PubMed
9. Harvard Business Review. Harrison M. How a U.S. Health Care System Uses 15-Minute Huddles to Keep 23 Hospitals Aligned. https://hbr.org/2018/11/how-a-u-s-health-care-system-uses-15-minute-huddles-to-keep-23-hospitals-aligned. Published November, 2019. Accessed May 16, 2019.
Rapid dissemination and adoption of evidence-based guidelines remains a challenge despite studies showing that key evidence-based care processes improve outcomes in sepsis and heart failure.1 Hospital medicine was virtually founded on the premise that hospitalists would be champions of delivering high-quality care. Hospitalists are now dealing with a new challenge—unprecedented growth of healthcare systems because of mergers and acquisitions. The year 2018 was a banner time for healthcare mergers and acquisitions, with a total of 1,182, up 14% from 2017.2 These are in response to the belief that healthcare systems may better navigate the mixed reimbursement models of fee-for-service and fee-for-value by achieving a larger patient base and economies of scale. Hospitalists must now achieve consistent, evidence-based standards of care across larger networks by educating their colleagues (often separated by large geographic areas) to manifest durable changes in their group practice with demonstrable improvement in patient outcomes and cost savings.
The study by Yurso et al. focused on implementing an education program, which included standardized learning through Clinical Performance and Value (CPV) vignettes with process measurement and feedback for sepsis and heart failure.3 Sepsis and heart failure have been a focus for treatment standardization because of the associated morbidity, mortality, and high cost of care. The study by Yurso et al. is a prospective quasi-controlled cohort of hospitalists in eight hospitals who were matched with comparator hospitalists in six nonparticipating hospitals across the AdventHealth system. Measurement and feedback were provided using CPV vignettes. Over two years, hospitalists who participated improved CPV scores by 8%, compliance with the utilization of the three-hour sepsis bundle from 46.0% to 57.5%, and orders of essential medical treatment elements for heart failure from 58.2% to 72.1%. In year one, the average length of stay (LOS) observed/expected (O/E) rates dropped by 8% for participating hospitalists compared with 2.5% in the comparator group. By year two, cost O/E rates improved slightly resulting in cost savings. The authors concluded that CPV case simulation-based measurement and feedback helped drive improvements in evidence-based care, which was associated with lower costs and shorter LOS.
While studies using traditional didactic CME struggle to demonstrate changes in practice leading to improved patient outcomes,4 the study by Yurso et al. gives a glimpse into how simulation can be used to help improve clinical performance and measure adherence to best practice. A remarkably similar study used CPV for simulated patients with serial performance measurement and feedback for heart failure and pneumonia. The study showed reduced practice variation between hospitalists at 11 hospitals across four states and decreased LOS and readmissions. However, the sole clinical outcome was no change in in-house mortality.5 Another study using CPV training in breast cancer treatment demonstrated increased adherence to evidence-based practice standards and decreased variation in care between providers across four states.6 Of note, this study did not include clinical outcomes. These studies collectively imply that simulation training with interactive learning, educational feedback, repetitive practice, and curriculum integration has shown modest success in creating practice change and improving adherence to best practice standards. However, they have minimal measures of patient outcomes and fairly simple analyses for cost savings. Because the education is computer-based and feedback can be performed remotely, it can be deployed across large and diverse growing healthcare systems. To really move the needle, future research in the field of simulation should identify optimal simulation methods and be designed with more rigor to include patient and cost outcomes.
At Intermountain Healthcare, hospitalist expansion occurred through a strategic realignment from the different geographic regions into the One Intermountain model. This model is built on the commitment that our patients will receive the same high-quality, high-value care wherever they walk through our doors. We have found four substantive changes have been particularly powerful in spurring a group practice mentality toward standardizing best practice. One, hospitalists are now aligned across the system under a single operational leadership structure that encourages combined efforts to share best practices and develop and deploy strategic initiatives around them. Two, hospitalists continue to build on a culture of quality and measure what matters to patients. While Intermountain Healthcare has a long history of using quality improvement to achieve better patient outcomes and lower costs,7 the new structure is allowing our group to test novel methods including redesigned education to see what actually improves adherence to best practice. Three, the group knows where the system’s reimbursement is coming from; Intermountain Healthcare has transitioned to a larger percentage of capitation,8 currently about 40%, with a strong commitment to partner with services geared to transition patients home quickly and keep them at home. Four, the organization has created a structure of accountability and reporting; an executive-sponsored systemwide operating model has been designed to cut through system barriers being identified by the frontline, allowing them to be rapidly surfaced and then solved at the executive level through daily huddles.9
Innovative educational programs such as the one described in the study by Yurso et al. that help the busy hospitalist achieve improved adherence to best practice are likely to be an important component leading to improved outcomes, but only after a group has been structured for success. As hospitalist groups continue to act as a single effector arm for high-value care, this will help meet the expectations of our patients and deliver on the promise of our field.
Disclosures: Dr. Srivastava is a physician founder of the I- PASS Patient Safety Institute. His employer, Intermountain Healthcare owns his equity in the I-PASS Patient Safety Institute. Dr. Srivastava is supported in part by the Children’s Hospital Association for his work as an executive council member of the Pediatric Research in Inpatient Settings (PRIS) network. Dr. Srivastava has received monetary awards, honorariums, and travel reimbursement from multiple academic and professional organizations for talks about pediatric hospitalist research networks and quality of care. All other authors have nothing to disclose. No funding was provided for this editorial.
Disclosures
The authors have no disclosures of financial conflicts of interest.
Funding
Dr. Walke was supported an award from the Health Resources and Services Administration Geriatric Workforce Enhancement Program to the University of Pennsylvania (U1QHP28720).
Rapid dissemination and adoption of evidence-based guidelines remains a challenge despite studies showing that key evidence-based care processes improve outcomes in sepsis and heart failure.1 Hospital medicine was virtually founded on the premise that hospitalists would be champions of delivering high-quality care. Hospitalists are now dealing with a new challenge—unprecedented growth of healthcare systems because of mergers and acquisitions. The year 2018 was a banner time for healthcare mergers and acquisitions, with a total of 1,182, up 14% from 2017.2 These are in response to the belief that healthcare systems may better navigate the mixed reimbursement models of fee-for-service and fee-for-value by achieving a larger patient base and economies of scale. Hospitalists must now achieve consistent, evidence-based standards of care across larger networks by educating their colleagues (often separated by large geographic areas) to manifest durable changes in their group practice with demonstrable improvement in patient outcomes and cost savings.
The study by Yurso et al. focused on implementing an education program, which included standardized learning through Clinical Performance and Value (CPV) vignettes with process measurement and feedback for sepsis and heart failure.3 Sepsis and heart failure have been a focus for treatment standardization because of the associated morbidity, mortality, and high cost of care. The study by Yurso et al. is a prospective quasi-controlled cohort of hospitalists in eight hospitals who were matched with comparator hospitalists in six nonparticipating hospitals across the AdventHealth system. Measurement and feedback were provided using CPV vignettes. Over two years, hospitalists who participated improved CPV scores by 8%, compliance with the utilization of the three-hour sepsis bundle from 46.0% to 57.5%, and orders of essential medical treatment elements for heart failure from 58.2% to 72.1%. In year one, the average length of stay (LOS) observed/expected (O/E) rates dropped by 8% for participating hospitalists compared with 2.5% in the comparator group. By year two, cost O/E rates improved slightly resulting in cost savings. The authors concluded that CPV case simulation-based measurement and feedback helped drive improvements in evidence-based care, which was associated with lower costs and shorter LOS.
While studies using traditional didactic CME struggle to demonstrate changes in practice leading to improved patient outcomes,4 the study by Yurso et al. gives a glimpse into how simulation can be used to help improve clinical performance and measure adherence to best practice. A remarkably similar study used CPV for simulated patients with serial performance measurement and feedback for heart failure and pneumonia. The study showed reduced practice variation between hospitalists at 11 hospitals across four states and decreased LOS and readmissions. However, the sole clinical outcome was no change in in-house mortality.5 Another study using CPV training in breast cancer treatment demonstrated increased adherence to evidence-based practice standards and decreased variation in care between providers across four states.6 Of note, this study did not include clinical outcomes. These studies collectively imply that simulation training with interactive learning, educational feedback, repetitive practice, and curriculum integration has shown modest success in creating practice change and improving adherence to best practice standards. However, they have minimal measures of patient outcomes and fairly simple analyses for cost savings. Because the education is computer-based and feedback can be performed remotely, it can be deployed across large and diverse growing healthcare systems. To really move the needle, future research in the field of simulation should identify optimal simulation methods and be designed with more rigor to include patient and cost outcomes.
At Intermountain Healthcare, hospitalist expansion occurred through a strategic realignment from the different geographic regions into the One Intermountain model. This model is built on the commitment that our patients will receive the same high-quality, high-value care wherever they walk through our doors. We have found four substantive changes have been particularly powerful in spurring a group practice mentality toward standardizing best practice. One, hospitalists are now aligned across the system under a single operational leadership structure that encourages combined efforts to share best practices and develop and deploy strategic initiatives around them. Two, hospitalists continue to build on a culture of quality and measure what matters to patients. While Intermountain Healthcare has a long history of using quality improvement to achieve better patient outcomes and lower costs,7 the new structure is allowing our group to test novel methods including redesigned education to see what actually improves adherence to best practice. Three, the group knows where the system’s reimbursement is coming from; Intermountain Healthcare has transitioned to a larger percentage of capitation,8 currently about 40%, with a strong commitment to partner with services geared to transition patients home quickly and keep them at home. Four, the organization has created a structure of accountability and reporting; an executive-sponsored systemwide operating model has been designed to cut through system barriers being identified by the frontline, allowing them to be rapidly surfaced and then solved at the executive level through daily huddles.9
Innovative educational programs such as the one described in the study by Yurso et al. that help the busy hospitalist achieve improved adherence to best practice are likely to be an important component leading to improved outcomes, but only after a group has been structured for success. As hospitalist groups continue to act as a single effector arm for high-value care, this will help meet the expectations of our patients and deliver on the promise of our field.
Disclosures: Dr. Srivastava is a physician founder of the I- PASS Patient Safety Institute. His employer, Intermountain Healthcare owns his equity in the I-PASS Patient Safety Institute. Dr. Srivastava is supported in part by the Children’s Hospital Association for his work as an executive council member of the Pediatric Research in Inpatient Settings (PRIS) network. Dr. Srivastava has received monetary awards, honorariums, and travel reimbursement from multiple academic and professional organizations for talks about pediatric hospitalist research networks and quality of care. All other authors have nothing to disclose. No funding was provided for this editorial.
Disclosures
The authors have no disclosures of financial conflicts of interest.
Funding
Dr. Walke was supported an award from the Health Resources and Services Administration Geriatric Workforce Enhancement Program to the University of Pennsylvania (U1QHP28720).
1. Seymour, CW, Geston F, Prescott HC, et al. Time to treatment and mortality during mandated emergency care for sepsis. N Engl J Med. 2017;376(23):2235-2244. https://doi.org/10.1056/NEJMoa1703058.
2. Healthcare Finance. Lagasse J. Healthcare mergers and acquisitions had record year in 2018, up 14.4 percent.https://webcache.googleusercontent.com/search?q=cache:zoMrl9yoLokJ:https://www.healthcarefinancenews.com/news/healthcare-mergers-and-acquisitions-had-record-year-2018-144-percent+&cd=2&hl=en&ct=clnk&gl=us. Published January, 2019. Accessed April 26, 2019.
3. Yurso M, Box B, Burgon T, et al. Reducing unneeded clinical variation in sepsis and heart failure care to improve outcomes and reduce cost: a collaborative engagement with hospitalists in a multi-state system. J Hosp Med. 2019;14(9):542-546. https://doi.org/10.12788/jhm.3220.
4. Cervero RM, Gaines JK. The impact of CME on physician performance and patient health outcomes: an updated synthesis of systematic reviews. J Contin Educ Health Prof. 2015;35(2):131-138. https://doi.org/10.1002/chp.21290.
5. Weems L, Strong J, Plummer D, et al. A quality collaboration in heart failure and pneumonia inpatient care at Novant Health: standardizing hospitalist practices to improve patient care and system performance. Jt Comm J Qual Patient Saf. 2019;45(3):199-206. https://doi.org/10.1016/j.jcjq.2018.09.005.
6. Peabody JW, Paculdo DR, Tamondong-Lachica D, et al. Improving clinical practice using a novel engagement approach; measurement, benchmarking and feedback; a longitudinal study. J Clin Med Res. 2016;8(9):633-640. https://doi.org/10.14740/jocmr2620w.
7. James BC, Savitz LA. How Intermountain trimmed health care costs through robust quality improvement efforts. Health Aff (Millwood). 2011;30(6):1185-1191. https://doi.org/10.1377/hlthaff.2011.0358.
8. James BC, Poulsen GP. The case for capitation. Harv Bus Rev. 2016;94(7-8):102-111,134. PubMed
9. Harvard Business Review. Harrison M. How a U.S. Health Care System Uses 15-Minute Huddles to Keep 23 Hospitals Aligned. https://hbr.org/2018/11/how-a-u-s-health-care-system-uses-15-minute-huddles-to-keep-23-hospitals-aligned. Published November, 2019. Accessed May 16, 2019.
1. Seymour, CW, Geston F, Prescott HC, et al. Time to treatment and mortality during mandated emergency care for sepsis. N Engl J Med. 2017;376(23):2235-2244. https://doi.org/10.1056/NEJMoa1703058.
2. Healthcare Finance. Lagasse J. Healthcare mergers and acquisitions had record year in 2018, up 14.4 percent.https://webcache.googleusercontent.com/search?q=cache:zoMrl9yoLokJ:https://www.healthcarefinancenews.com/news/healthcare-mergers-and-acquisitions-had-record-year-2018-144-percent+&cd=2&hl=en&ct=clnk&gl=us. Published January, 2019. Accessed April 26, 2019.
3. Yurso M, Box B, Burgon T, et al. Reducing unneeded clinical variation in sepsis and heart failure care to improve outcomes and reduce cost: a collaborative engagement with hospitalists in a multi-state system. J Hosp Med. 2019;14(9):542-546. https://doi.org/10.12788/jhm.3220.
4. Cervero RM, Gaines JK. The impact of CME on physician performance and patient health outcomes: an updated synthesis of systematic reviews. J Contin Educ Health Prof. 2015;35(2):131-138. https://doi.org/10.1002/chp.21290.
5. Weems L, Strong J, Plummer D, et al. A quality collaboration in heart failure and pneumonia inpatient care at Novant Health: standardizing hospitalist practices to improve patient care and system performance. Jt Comm J Qual Patient Saf. 2019;45(3):199-206. https://doi.org/10.1016/j.jcjq.2018.09.005.
6. Peabody JW, Paculdo DR, Tamondong-Lachica D, et al. Improving clinical practice using a novel engagement approach; measurement, benchmarking and feedback; a longitudinal study. J Clin Med Res. 2016;8(9):633-640. https://doi.org/10.14740/jocmr2620w.
7. James BC, Savitz LA. How Intermountain trimmed health care costs through robust quality improvement efforts. Health Aff (Millwood). 2011;30(6):1185-1191. https://doi.org/10.1377/hlthaff.2011.0358.
8. James BC, Poulsen GP. The case for capitation. Harv Bus Rev. 2016;94(7-8):102-111,134. PubMed
9. Harvard Business Review. Harrison M. How a U.S. Health Care System Uses 15-Minute Huddles to Keep 23 Hospitals Aligned. https://hbr.org/2018/11/how-a-u-s-health-care-system-uses-15-minute-huddles-to-keep-23-hospitals-aligned. Published November, 2019. Accessed May 16, 2019.
© 2019 Society of Hospital Medicine