User login
Teledermoscopy using smartphones fails to boost skin cancer detection
Skin cancer detection does not improve when patients use a dermatoscope for smartphones to capture and send photos of lesions for diagnosis, according to the Australian SKIN Project trial.
“Mobile health applications are increasingly used in cancer prevention and early detection but rarely tested stringently for their value with regard to patient care,” noted trial investigators, who were led by Monika Janda, PhD, of the University of Queensland in Brisbane, Australia. The trial was published in Lancet Digital Health.
The investigators studied 234 adults at high risk for skin cancer, asking them to perform whole-body skin self-exams in their homes at baseline, 1 month, and 2 months.
Half of patients were randomized to perform standard naked-eye exams, note suspicious lesions on a body chart, and submit the chart by email. The other half were randomized to supplement their exams with mobile dermoscopy, and they were provided with a dermatoscope (FotoFinder) that interfaces with smartphones to capture and submit photos of suspicious lesions to a dermatologist for telediagnosis.
Both groups received Web-based instructions on how to complete whole-body skin self-exams and were examined in person within 3 months of their last self-exam to provide a reference standard.
In comparing the two approaches for early detection of skin cancer, the investigators determined that teledermoscopy-enhanced exams would have to show 20% greater sensitivity to establish their superiority.
The median number of lesions submitted was six per person in both the group using adjunctive teledermoscopy and the group performing only naked-eye exams.
At the lesion level, teledermoscopy-enhanced exams had lower sensitivity than naked-eye exams in detecting suspected skin cancers or precursor lesions (75% vs. 88%; P = .04) and similar specificity (87% vs. 89%; P = .42). At the patient level, the two approaches had statistically indistinguishable sensitivity (87% vs. 97%; P = .26) and specificity (95% vs. 96%; P = .96).
At the same time, telediagnosis showed good overall diagnostic concordance with in-person clinical skin examination (88%).
“For the early detection of skin cancer, naked-eye skin self-examination should continue to be recommended by cancer agencies,” Dr. Janda and colleagues concluded. “Further improvements to the instructions for participants on the relevance of nonpigmented skin lesions, training for partners, and the integration of automatic algorithms that rule out clearly benign skin lesions at the time of photographing might increase sensitivity of teledermoscopy in the future.”
This trial was funded by the National Health and Medical Research Council. The authors disclosed relationships with e-derm-consult, SciBase, Canfield Scientific, and other companies.
SOURCE: Janda M et al. Lancet Digit Health. 2020 March; 2(3):e129-e137.
Skin cancer detection does not improve when patients use a dermatoscope for smartphones to capture and send photos of lesions for diagnosis, according to the Australian SKIN Project trial.
“Mobile health applications are increasingly used in cancer prevention and early detection but rarely tested stringently for their value with regard to patient care,” noted trial investigators, who were led by Monika Janda, PhD, of the University of Queensland in Brisbane, Australia. The trial was published in Lancet Digital Health.
The investigators studied 234 adults at high risk for skin cancer, asking them to perform whole-body skin self-exams in their homes at baseline, 1 month, and 2 months.
Half of patients were randomized to perform standard naked-eye exams, note suspicious lesions on a body chart, and submit the chart by email. The other half were randomized to supplement their exams with mobile dermoscopy, and they were provided with a dermatoscope (FotoFinder) that interfaces with smartphones to capture and submit photos of suspicious lesions to a dermatologist for telediagnosis.
Both groups received Web-based instructions on how to complete whole-body skin self-exams and were examined in person within 3 months of their last self-exam to provide a reference standard.
In comparing the two approaches for early detection of skin cancer, the investigators determined that teledermoscopy-enhanced exams would have to show 20% greater sensitivity to establish their superiority.
The median number of lesions submitted was six per person in both the group using adjunctive teledermoscopy and the group performing only naked-eye exams.
At the lesion level, teledermoscopy-enhanced exams had lower sensitivity than naked-eye exams in detecting suspected skin cancers or precursor lesions (75% vs. 88%; P = .04) and similar specificity (87% vs. 89%; P = .42). At the patient level, the two approaches had statistically indistinguishable sensitivity (87% vs. 97%; P = .26) and specificity (95% vs. 96%; P = .96).
At the same time, telediagnosis showed good overall diagnostic concordance with in-person clinical skin examination (88%).
“For the early detection of skin cancer, naked-eye skin self-examination should continue to be recommended by cancer agencies,” Dr. Janda and colleagues concluded. “Further improvements to the instructions for participants on the relevance of nonpigmented skin lesions, training for partners, and the integration of automatic algorithms that rule out clearly benign skin lesions at the time of photographing might increase sensitivity of teledermoscopy in the future.”
This trial was funded by the National Health and Medical Research Council. The authors disclosed relationships with e-derm-consult, SciBase, Canfield Scientific, and other companies.
SOURCE: Janda M et al. Lancet Digit Health. 2020 March; 2(3):e129-e137.
Skin cancer detection does not improve when patients use a dermatoscope for smartphones to capture and send photos of lesions for diagnosis, according to the Australian SKIN Project trial.
“Mobile health applications are increasingly used in cancer prevention and early detection but rarely tested stringently for their value with regard to patient care,” noted trial investigators, who were led by Monika Janda, PhD, of the University of Queensland in Brisbane, Australia. The trial was published in Lancet Digital Health.
The investigators studied 234 adults at high risk for skin cancer, asking them to perform whole-body skin self-exams in their homes at baseline, 1 month, and 2 months.
Half of patients were randomized to perform standard naked-eye exams, note suspicious lesions on a body chart, and submit the chart by email. The other half were randomized to supplement their exams with mobile dermoscopy, and they were provided with a dermatoscope (FotoFinder) that interfaces with smartphones to capture and submit photos of suspicious lesions to a dermatologist for telediagnosis.
Both groups received Web-based instructions on how to complete whole-body skin self-exams and were examined in person within 3 months of their last self-exam to provide a reference standard.
In comparing the two approaches for early detection of skin cancer, the investigators determined that teledermoscopy-enhanced exams would have to show 20% greater sensitivity to establish their superiority.
The median number of lesions submitted was six per person in both the group using adjunctive teledermoscopy and the group performing only naked-eye exams.
At the lesion level, teledermoscopy-enhanced exams had lower sensitivity than naked-eye exams in detecting suspected skin cancers or precursor lesions (75% vs. 88%; P = .04) and similar specificity (87% vs. 89%; P = .42). At the patient level, the two approaches had statistically indistinguishable sensitivity (87% vs. 97%; P = .26) and specificity (95% vs. 96%; P = .96).
At the same time, telediagnosis showed good overall diagnostic concordance with in-person clinical skin examination (88%).
“For the early detection of skin cancer, naked-eye skin self-examination should continue to be recommended by cancer agencies,” Dr. Janda and colleagues concluded. “Further improvements to the instructions for participants on the relevance of nonpigmented skin lesions, training for partners, and the integration of automatic algorithms that rule out clearly benign skin lesions at the time of photographing might increase sensitivity of teledermoscopy in the future.”
This trial was funded by the National Health and Medical Research Council. The authors disclosed relationships with e-derm-consult, SciBase, Canfield Scientific, and other companies.
SOURCE: Janda M et al. Lancet Digit Health. 2020 March; 2(3):e129-e137.
FROM LANCET DIGITAL HEALTH
Study links GLP-1R agonists, lower inflammatory biomarker levels
Patients with both type 2 diabetes and asthma who were on glucagonlike peptide receptor–1 (GLP-1R) agonists for glucose control had lower levels of a key biomarker of airway inflammation than similar patients on other types of glucose-control medications, according to results of a study to have been presented at the annual meeting of the American Academy of Asthma, Allergy, and Immunology. The AAAAI canceled their annual meeting and provided abstracts and access to presenters for press coverage.
The findings from this study potentially replicated findings in humans that have been reported in preclinical trials.
“Our work showed that type 2 diabetics with asthma who were treated with GLP-1 receptor agonists had lower levels of periostin, and this provides really one of the first human data to show that these drugs may impact key inflammation pathways in the airway,” Dinah Foer, MD, of Brigham and Women’s Hospital, Boston, said in an interview. She described periostin as “a known critical inducer of airway mucous production and airway responsiveness.”
The study retrospectively evaluated serum samples from the Partners HealthCare Biobank of 161 adults with both asthma and type 2 diabetes, 42 of whom were on GLP-1R agonists and 119 of whom were taking non-GLP-1R agonist diabetes medications. The study used the Partners Healthcare EHR to identify eligible patients.
The study found that periostin levels were significantly decreased in GLP-1R agonist users: 19.1 ng/mL (standard deviation, +8.7) versus 27.4 ng/mL (SD, +14) in the non-GLP-1R agonist group (P = .001), Dr. Foer said. The other known mediators of asthma inflammatory pathways that were measured – interleukin-6, IL-8, sCD163, total IgE, and sST2 (soluble suppression of tumorigenesis–2) – showed no differences between the two groups, Dr. Foer said.
She said that this was the first human study to show similar results to preclinical models of asthma pathways. “What was interesting to us was that our findings were robust even when we controlled for covariates,” she added.
These findings lay the groundwork for further research into the potential therapeutic role GLP-1R agonists in asthma, Dr. Foer said. “This supports using periostin as a biomarker for novel therapeutic use of GLP-1R [agonists] in asthma,” she said. “At this point further study is needed to understand the clinical impact of GPL-1R [agonists] in asthma both for patients with type 2 diabetes and potentially in the future for patients who don’t have type 2 diabetes or metabolic disease.”
She added: “I don’t think we’re there yet; this is just one foot forward.”
The next step for researchers involves analyzing outcomes in asthmatics with type 2 diabetes on GLP-1R agonist therapy using a larger sample size as well as patients with asthma and metabolic disease, Dr. Foer said. The goal would be to identify corresponding biomarkers.
“There’s a terrific conversation in the field about the relationships between metabolism and asthma,” she said. “What our data contributes to that is, it suggests a role for metabolic pathways, specifically as it’s related GLP-1R [agonist] signaling pathways in regulating airway inflammation.”
Mark Moss, MD, associate professor of allergy & immunology at the University of Wisconsin–Madison, who was to serve as the moderator of the session, was positive about the GLP-1R agonist findings. He said in an interview: “This is promising research that provides a possible new target for the treatment of asthma.”
Dr. Foer disclosed that she has no relevant financial relationships.
SOURCE: Foer D et al. AAAAI Session 462, Abstract 784.
Patients with both type 2 diabetes and asthma who were on glucagonlike peptide receptor–1 (GLP-1R) agonists for glucose control had lower levels of a key biomarker of airway inflammation than similar patients on other types of glucose-control medications, according to results of a study to have been presented at the annual meeting of the American Academy of Asthma, Allergy, and Immunology. The AAAAI canceled their annual meeting and provided abstracts and access to presenters for press coverage.
The findings from this study potentially replicated findings in humans that have been reported in preclinical trials.
“Our work showed that type 2 diabetics with asthma who were treated with GLP-1 receptor agonists had lower levels of periostin, and this provides really one of the first human data to show that these drugs may impact key inflammation pathways in the airway,” Dinah Foer, MD, of Brigham and Women’s Hospital, Boston, said in an interview. She described periostin as “a known critical inducer of airway mucous production and airway responsiveness.”
The study retrospectively evaluated serum samples from the Partners HealthCare Biobank of 161 adults with both asthma and type 2 diabetes, 42 of whom were on GLP-1R agonists and 119 of whom were taking non-GLP-1R agonist diabetes medications. The study used the Partners Healthcare EHR to identify eligible patients.
The study found that periostin levels were significantly decreased in GLP-1R agonist users: 19.1 ng/mL (standard deviation, +8.7) versus 27.4 ng/mL (SD, +14) in the non-GLP-1R agonist group (P = .001), Dr. Foer said. The other known mediators of asthma inflammatory pathways that were measured – interleukin-6, IL-8, sCD163, total IgE, and sST2 (soluble suppression of tumorigenesis–2) – showed no differences between the two groups, Dr. Foer said.
She said that this was the first human study to show similar results to preclinical models of asthma pathways. “What was interesting to us was that our findings were robust even when we controlled for covariates,” she added.
These findings lay the groundwork for further research into the potential therapeutic role GLP-1R agonists in asthma, Dr. Foer said. “This supports using periostin as a biomarker for novel therapeutic use of GLP-1R [agonists] in asthma,” she said. “At this point further study is needed to understand the clinical impact of GPL-1R [agonists] in asthma both for patients with type 2 diabetes and potentially in the future for patients who don’t have type 2 diabetes or metabolic disease.”
She added: “I don’t think we’re there yet; this is just one foot forward.”
The next step for researchers involves analyzing outcomes in asthmatics with type 2 diabetes on GLP-1R agonist therapy using a larger sample size as well as patients with asthma and metabolic disease, Dr. Foer said. The goal would be to identify corresponding biomarkers.
“There’s a terrific conversation in the field about the relationships between metabolism and asthma,” she said. “What our data contributes to that is, it suggests a role for metabolic pathways, specifically as it’s related GLP-1R [agonist] signaling pathways in regulating airway inflammation.”
Mark Moss, MD, associate professor of allergy & immunology at the University of Wisconsin–Madison, who was to serve as the moderator of the session, was positive about the GLP-1R agonist findings. He said in an interview: “This is promising research that provides a possible new target for the treatment of asthma.”
Dr. Foer disclosed that she has no relevant financial relationships.
SOURCE: Foer D et al. AAAAI Session 462, Abstract 784.
Patients with both type 2 diabetes and asthma who were on glucagonlike peptide receptor–1 (GLP-1R) agonists for glucose control had lower levels of a key biomarker of airway inflammation than similar patients on other types of glucose-control medications, according to results of a study to have been presented at the annual meeting of the American Academy of Asthma, Allergy, and Immunology. The AAAAI canceled their annual meeting and provided abstracts and access to presenters for press coverage.
The findings from this study potentially replicated findings in humans that have been reported in preclinical trials.
“Our work showed that type 2 diabetics with asthma who were treated with GLP-1 receptor agonists had lower levels of periostin, and this provides really one of the first human data to show that these drugs may impact key inflammation pathways in the airway,” Dinah Foer, MD, of Brigham and Women’s Hospital, Boston, said in an interview. She described periostin as “a known critical inducer of airway mucous production and airway responsiveness.”
The study retrospectively evaluated serum samples from the Partners HealthCare Biobank of 161 adults with both asthma and type 2 diabetes, 42 of whom were on GLP-1R agonists and 119 of whom were taking non-GLP-1R agonist diabetes medications. The study used the Partners Healthcare EHR to identify eligible patients.
The study found that periostin levels were significantly decreased in GLP-1R agonist users: 19.1 ng/mL (standard deviation, +8.7) versus 27.4 ng/mL (SD, +14) in the non-GLP-1R agonist group (P = .001), Dr. Foer said. The other known mediators of asthma inflammatory pathways that were measured – interleukin-6, IL-8, sCD163, total IgE, and sST2 (soluble suppression of tumorigenesis–2) – showed no differences between the two groups, Dr. Foer said.
She said that this was the first human study to show similar results to preclinical models of asthma pathways. “What was interesting to us was that our findings were robust even when we controlled for covariates,” she added.
These findings lay the groundwork for further research into the potential therapeutic role GLP-1R agonists in asthma, Dr. Foer said. “This supports using periostin as a biomarker for novel therapeutic use of GLP-1R [agonists] in asthma,” she said. “At this point further study is needed to understand the clinical impact of GPL-1R [agonists] in asthma both for patients with type 2 diabetes and potentially in the future for patients who don’t have type 2 diabetes or metabolic disease.”
She added: “I don’t think we’re there yet; this is just one foot forward.”
The next step for researchers involves analyzing outcomes in asthmatics with type 2 diabetes on GLP-1R agonist therapy using a larger sample size as well as patients with asthma and metabolic disease, Dr. Foer said. The goal would be to identify corresponding biomarkers.
“There’s a terrific conversation in the field about the relationships between metabolism and asthma,” she said. “What our data contributes to that is, it suggests a role for metabolic pathways, specifically as it’s related GLP-1R [agonist] signaling pathways in regulating airway inflammation.”
Mark Moss, MD, associate professor of allergy & immunology at the University of Wisconsin–Madison, who was to serve as the moderator of the session, was positive about the GLP-1R agonist findings. He said in an interview: “This is promising research that provides a possible new target for the treatment of asthma.”
Dr. Foer disclosed that she has no relevant financial relationships.
SOURCE: Foer D et al. AAAAI Session 462, Abstract 784.
CNS cancer outcomes worse for black and Hispanic children
according to a study published in Scientific Reports.
While prior studies have shown the effects of racial/ethnic and socioeconomic risk factors on treatment outcomes in adult cancer populations, less is known about how these factors impact children with CNS cancers, explained study author Robert Fineberg, MD, of St. Anthony North Health Campus in Westminster, Colo., and colleagues.
The authors conducted their study to examine the effects of demographic and socioeconomic factors on survival in pediatric CNS cancers. Using data from the Surveillance, Epidemiology, and End Results database, the researchers identified 1,881 patients with CNS tumors, including both spinal and cranial neoplasms.
Data collection encompassed patient characteristics, socioeconomic parameters, tumor characteristics, treatment, and year of diagnosis. The primary outcomes were overall survival and disease stage at diagnosis.
Most patients were white (78.15%) and non-Hispanic (72.09%). The most common brain tumors were gliomas (n = 788), ependymomas (n = 418), and medulloblastomas (n = 393).
On multivariable analysis, the researchers found that black and Hispanic patients had worse survival, compared with white patients (hazard ratio, 1.39; P = .0014) and non-Hispanic patients (HR, 1.36; P = .0002).
After adjustment for socioeconomic parameters and treatment, the hazard ratios for both Hispanic (HR, 1.29; P = .0051) and black patients (HR, 1.29; P = .0206) slightly declined, but the differences remained significant.
On stratified analysis, poorer survival rates were observed for black and Hispanic patients with both metastatic and localized disease at diagnosis, compared with white non-Hispanic patients. However, after adjustment for mediating factors, the difference did not remain significant for black patients (P = .1026).
“Our findings on extent of disease at diagnosis demonstrated that neither black race nor Hispanic ethnicity increased the chance of metastatic disease at presentation when controlling for mediating variables,” the authors wrote. “These data suggest that racial and ethnic disparities appear to be partially explained by postdiagnosis mediating factors that may fall in the pathway between race/ethnicity and poorer survival.”
The researchers acknowledged that a key limitation of this study was the exclusion of insurance status because of incomplete access for some patients. As a result, potential associations between insurance and survival or extent of disease could not be determined.
“To better understand underlying causes that contribute to the disparity of outcomes in pediatric brain tumors, patient-level data should be utilized in future studies to investigate both biological factors and pre/postdiagnosis treatment gaps in the care of children diagnosed with CNS tumors in the hopes of improving outcomes,” the authors wrote.
In the meantime, collaboration among physicians could help improve outcomes for these patients, according to study author Adam Green, MD, of the University of Colorado at Denver in Aurora.
“[Clinicians] should establish good working relationships with pediatric oncology and neuro-oncology physicians in their community, and they should ask questions early of those teams when they have patients they’re concerned about,” Dr. Green said. “They can [ensure] that patients of minority race/ethnicity, nonprivate health insurance, and lower socioeconomic status have easy and timely access to appointments.”
This research was supported, in part, by grant funding from the National Institutes of Health. The authors reported having no conflicts of interest.
SOURCE: Fineberg R et al. Scientific Reports. 2020 Mar 12. doi: 10.1038/s41598-020-61237-2.
according to a study published in Scientific Reports.
While prior studies have shown the effects of racial/ethnic and socioeconomic risk factors on treatment outcomes in adult cancer populations, less is known about how these factors impact children with CNS cancers, explained study author Robert Fineberg, MD, of St. Anthony North Health Campus in Westminster, Colo., and colleagues.
The authors conducted their study to examine the effects of demographic and socioeconomic factors on survival in pediatric CNS cancers. Using data from the Surveillance, Epidemiology, and End Results database, the researchers identified 1,881 patients with CNS tumors, including both spinal and cranial neoplasms.
Data collection encompassed patient characteristics, socioeconomic parameters, tumor characteristics, treatment, and year of diagnosis. The primary outcomes were overall survival and disease stage at diagnosis.
Most patients were white (78.15%) and non-Hispanic (72.09%). The most common brain tumors were gliomas (n = 788), ependymomas (n = 418), and medulloblastomas (n = 393).
On multivariable analysis, the researchers found that black and Hispanic patients had worse survival, compared with white patients (hazard ratio, 1.39; P = .0014) and non-Hispanic patients (HR, 1.36; P = .0002).
After adjustment for socioeconomic parameters and treatment, the hazard ratios for both Hispanic (HR, 1.29; P = .0051) and black patients (HR, 1.29; P = .0206) slightly declined, but the differences remained significant.
On stratified analysis, poorer survival rates were observed for black and Hispanic patients with both metastatic and localized disease at diagnosis, compared with white non-Hispanic patients. However, after adjustment for mediating factors, the difference did not remain significant for black patients (P = .1026).
“Our findings on extent of disease at diagnosis demonstrated that neither black race nor Hispanic ethnicity increased the chance of metastatic disease at presentation when controlling for mediating variables,” the authors wrote. “These data suggest that racial and ethnic disparities appear to be partially explained by postdiagnosis mediating factors that may fall in the pathway between race/ethnicity and poorer survival.”
The researchers acknowledged that a key limitation of this study was the exclusion of insurance status because of incomplete access for some patients. As a result, potential associations between insurance and survival or extent of disease could not be determined.
“To better understand underlying causes that contribute to the disparity of outcomes in pediatric brain tumors, patient-level data should be utilized in future studies to investigate both biological factors and pre/postdiagnosis treatment gaps in the care of children diagnosed with CNS tumors in the hopes of improving outcomes,” the authors wrote.
In the meantime, collaboration among physicians could help improve outcomes for these patients, according to study author Adam Green, MD, of the University of Colorado at Denver in Aurora.
“[Clinicians] should establish good working relationships with pediatric oncology and neuro-oncology physicians in their community, and they should ask questions early of those teams when they have patients they’re concerned about,” Dr. Green said. “They can [ensure] that patients of minority race/ethnicity, nonprivate health insurance, and lower socioeconomic status have easy and timely access to appointments.”
This research was supported, in part, by grant funding from the National Institutes of Health. The authors reported having no conflicts of interest.
SOURCE: Fineberg R et al. Scientific Reports. 2020 Mar 12. doi: 10.1038/s41598-020-61237-2.
according to a study published in Scientific Reports.
While prior studies have shown the effects of racial/ethnic and socioeconomic risk factors on treatment outcomes in adult cancer populations, less is known about how these factors impact children with CNS cancers, explained study author Robert Fineberg, MD, of St. Anthony North Health Campus in Westminster, Colo., and colleagues.
The authors conducted their study to examine the effects of demographic and socioeconomic factors on survival in pediatric CNS cancers. Using data from the Surveillance, Epidemiology, and End Results database, the researchers identified 1,881 patients with CNS tumors, including both spinal and cranial neoplasms.
Data collection encompassed patient characteristics, socioeconomic parameters, tumor characteristics, treatment, and year of diagnosis. The primary outcomes were overall survival and disease stage at diagnosis.
Most patients were white (78.15%) and non-Hispanic (72.09%). The most common brain tumors were gliomas (n = 788), ependymomas (n = 418), and medulloblastomas (n = 393).
On multivariable analysis, the researchers found that black and Hispanic patients had worse survival, compared with white patients (hazard ratio, 1.39; P = .0014) and non-Hispanic patients (HR, 1.36; P = .0002).
After adjustment for socioeconomic parameters and treatment, the hazard ratios for both Hispanic (HR, 1.29; P = .0051) and black patients (HR, 1.29; P = .0206) slightly declined, but the differences remained significant.
On stratified analysis, poorer survival rates were observed for black and Hispanic patients with both metastatic and localized disease at diagnosis, compared with white non-Hispanic patients. However, after adjustment for mediating factors, the difference did not remain significant for black patients (P = .1026).
“Our findings on extent of disease at diagnosis demonstrated that neither black race nor Hispanic ethnicity increased the chance of metastatic disease at presentation when controlling for mediating variables,” the authors wrote. “These data suggest that racial and ethnic disparities appear to be partially explained by postdiagnosis mediating factors that may fall in the pathway between race/ethnicity and poorer survival.”
The researchers acknowledged that a key limitation of this study was the exclusion of insurance status because of incomplete access for some patients. As a result, potential associations between insurance and survival or extent of disease could not be determined.
“To better understand underlying causes that contribute to the disparity of outcomes in pediatric brain tumors, patient-level data should be utilized in future studies to investigate both biological factors and pre/postdiagnosis treatment gaps in the care of children diagnosed with CNS tumors in the hopes of improving outcomes,” the authors wrote.
In the meantime, collaboration among physicians could help improve outcomes for these patients, according to study author Adam Green, MD, of the University of Colorado at Denver in Aurora.
“[Clinicians] should establish good working relationships with pediatric oncology and neuro-oncology physicians in their community, and they should ask questions early of those teams when they have patients they’re concerned about,” Dr. Green said. “They can [ensure] that patients of minority race/ethnicity, nonprivate health insurance, and lower socioeconomic status have easy and timely access to appointments.”
This research was supported, in part, by grant funding from the National Institutes of Health. The authors reported having no conflicts of interest.
SOURCE: Fineberg R et al. Scientific Reports. 2020 Mar 12. doi: 10.1038/s41598-020-61237-2.
FROM SCIENTIFIC REPORTS
SLIT tablet during pollen season improves symptoms of allergic rhinitis
according to recent research released as an abstract for the annual meeting of the American Academy of Allergy, Asthma & Immunology.
Tree pollen season is generally considered to be between February and June, with alder and hazel allergens affecting patients early and oak pollen affecting patients later in the season. Since a major birch allergen, Betula verrucosa 1 (Bet v 1), cross-reacts with alder, hazel, and oak allergens, some patients may experience allergies across the entire tree pollen season from members of this so-called birth homologous group, Hendrik Nolte, MD, senior vice president of research and development at ALK-Abello Americas and International, said in an interview.
According to the U.S. National Health and Nutrition Examination Survey 2005-2006, 16% of 8,086 participants 6 years or older with allergy had a specific immunoglobulin E (IgE) to birch, while 18% had a specific IgE to oak. Patients who reported having hay fever had a specific IgE to birch of 23% and a specific IgE to oak of 26% (J Allergy Clin Immunol. 2011 May;127[5]:1226-1235.e7).
“Patients who are allergic to birch pollen often experience symptoms in response to pollen from other members of the birch homologous group, which prolong the tree season and increase the symptom burden for these patients,” Dr. Nolte said. “Thus, treatment with SLIT-tablet immunotherapy may be an important treatment option for many allergy sufferers.”
Dr. Nolte and colleagues performed a randomized, double-blind, multinational trial of 634 patients before and during tree pollen season in which participants received a daily SLIT tablet or placebo. Patients were between ages 12 and 65 years with allergic rhinitis, and investigators enrolled patients or without conjunctivitis and with or without asthma. The investigations evaluated the patients’ daily symptom score and daily medication score, which was grouped into the total combined score. The patients were also allowed to use their rescue medications during the trial.
SLIT demonstrates symptom improvement
“Improvement in allergic rhinoconjunctivitis symptoms and reduction in symptom-relieving medication use with the tree SLIT-tablet during birch, alder/hazel, and oak pollen seasons were significant versus placebo and showed internal consistency across almost 4 months of birch and related tree pollen exposure,” Dr. Nolte said.
Patients showed relative improvements in their total combined score of 39.6% for birch, 29.7% for alder and hazel, 36.0% during oak pollen season, and 35.0% during the entire tree pollen season, compared with placebo (all P ≤ .002). Relative daily symptom scores also improved in the group that received oral SLIT, with 36.8% of patients showing improvement during birch season, 26.0% during alder and hazel season, 31.6% during oak season, and 31.6% across all pollen seasons, compared with those taking placebo (all P ≤ .003). A greater number of patients also achieved a relative improvement in daily medication score during birch season (49.2%), alder and hazel season (43.8%), oak season (45.9%) and during the whole of tree pollen season (45.3%), compared with placebo (P ≤ .002).
“The results support the clinical relevance of cross-reactivity between birch, alder/hazel, and oak pollen homologous allergens,” Dr. Nolte said. “Immunologic cross-reactivity is supported by alder, hazel, and oak specific IgE data and IgG4 in responses to the tree SLIT tablet.”
Dr. Nolte said the next step in his team’s research was to evaluate oral SLIT in a phase 3 trial for children aged 5-17 years.
This study was funded by ALK, and the authors received medical writing and editorial assistance from Scott Medical Communications. Dr. Nolte reported that he is a paid employee of ALK.
SOURCE: Nolte H et al. AAAAI, Abstract 267.
.
according to recent research released as an abstract for the annual meeting of the American Academy of Allergy, Asthma & Immunology.
Tree pollen season is generally considered to be between February and June, with alder and hazel allergens affecting patients early and oak pollen affecting patients later in the season. Since a major birch allergen, Betula verrucosa 1 (Bet v 1), cross-reacts with alder, hazel, and oak allergens, some patients may experience allergies across the entire tree pollen season from members of this so-called birth homologous group, Hendrik Nolte, MD, senior vice president of research and development at ALK-Abello Americas and International, said in an interview.
According to the U.S. National Health and Nutrition Examination Survey 2005-2006, 16% of 8,086 participants 6 years or older with allergy had a specific immunoglobulin E (IgE) to birch, while 18% had a specific IgE to oak. Patients who reported having hay fever had a specific IgE to birch of 23% and a specific IgE to oak of 26% (J Allergy Clin Immunol. 2011 May;127[5]:1226-1235.e7).
“Patients who are allergic to birch pollen often experience symptoms in response to pollen from other members of the birch homologous group, which prolong the tree season and increase the symptom burden for these patients,” Dr. Nolte said. “Thus, treatment with SLIT-tablet immunotherapy may be an important treatment option for many allergy sufferers.”
Dr. Nolte and colleagues performed a randomized, double-blind, multinational trial of 634 patients before and during tree pollen season in which participants received a daily SLIT tablet or placebo. Patients were between ages 12 and 65 years with allergic rhinitis, and investigators enrolled patients or without conjunctivitis and with or without asthma. The investigations evaluated the patients’ daily symptom score and daily medication score, which was grouped into the total combined score. The patients were also allowed to use their rescue medications during the trial.
SLIT demonstrates symptom improvement
“Improvement in allergic rhinoconjunctivitis symptoms and reduction in symptom-relieving medication use with the tree SLIT-tablet during birch, alder/hazel, and oak pollen seasons were significant versus placebo and showed internal consistency across almost 4 months of birch and related tree pollen exposure,” Dr. Nolte said.
Patients showed relative improvements in their total combined score of 39.6% for birch, 29.7% for alder and hazel, 36.0% during oak pollen season, and 35.0% during the entire tree pollen season, compared with placebo (all P ≤ .002). Relative daily symptom scores also improved in the group that received oral SLIT, with 36.8% of patients showing improvement during birch season, 26.0% during alder and hazel season, 31.6% during oak season, and 31.6% across all pollen seasons, compared with those taking placebo (all P ≤ .003). A greater number of patients also achieved a relative improvement in daily medication score during birch season (49.2%), alder and hazel season (43.8%), oak season (45.9%) and during the whole of tree pollen season (45.3%), compared with placebo (P ≤ .002).
“The results support the clinical relevance of cross-reactivity between birch, alder/hazel, and oak pollen homologous allergens,” Dr. Nolte said. “Immunologic cross-reactivity is supported by alder, hazel, and oak specific IgE data and IgG4 in responses to the tree SLIT tablet.”
Dr. Nolte said the next step in his team’s research was to evaluate oral SLIT in a phase 3 trial for children aged 5-17 years.
This study was funded by ALK, and the authors received medical writing and editorial assistance from Scott Medical Communications. Dr. Nolte reported that he is a paid employee of ALK.
SOURCE: Nolte H et al. AAAAI, Abstract 267.
.
according to recent research released as an abstract for the annual meeting of the American Academy of Allergy, Asthma & Immunology.
Tree pollen season is generally considered to be between February and June, with alder and hazel allergens affecting patients early and oak pollen affecting patients later in the season. Since a major birch allergen, Betula verrucosa 1 (Bet v 1), cross-reacts with alder, hazel, and oak allergens, some patients may experience allergies across the entire tree pollen season from members of this so-called birth homologous group, Hendrik Nolte, MD, senior vice president of research and development at ALK-Abello Americas and International, said in an interview.
According to the U.S. National Health and Nutrition Examination Survey 2005-2006, 16% of 8,086 participants 6 years or older with allergy had a specific immunoglobulin E (IgE) to birch, while 18% had a specific IgE to oak. Patients who reported having hay fever had a specific IgE to birch of 23% and a specific IgE to oak of 26% (J Allergy Clin Immunol. 2011 May;127[5]:1226-1235.e7).
“Patients who are allergic to birch pollen often experience symptoms in response to pollen from other members of the birch homologous group, which prolong the tree season and increase the symptom burden for these patients,” Dr. Nolte said. “Thus, treatment with SLIT-tablet immunotherapy may be an important treatment option for many allergy sufferers.”
Dr. Nolte and colleagues performed a randomized, double-blind, multinational trial of 634 patients before and during tree pollen season in which participants received a daily SLIT tablet or placebo. Patients were between ages 12 and 65 years with allergic rhinitis, and investigators enrolled patients or without conjunctivitis and with or without asthma. The investigations evaluated the patients’ daily symptom score and daily medication score, which was grouped into the total combined score. The patients were also allowed to use their rescue medications during the trial.
SLIT demonstrates symptom improvement
“Improvement in allergic rhinoconjunctivitis symptoms and reduction in symptom-relieving medication use with the tree SLIT-tablet during birch, alder/hazel, and oak pollen seasons were significant versus placebo and showed internal consistency across almost 4 months of birch and related tree pollen exposure,” Dr. Nolte said.
Patients showed relative improvements in their total combined score of 39.6% for birch, 29.7% for alder and hazel, 36.0% during oak pollen season, and 35.0% during the entire tree pollen season, compared with placebo (all P ≤ .002). Relative daily symptom scores also improved in the group that received oral SLIT, with 36.8% of patients showing improvement during birch season, 26.0% during alder and hazel season, 31.6% during oak season, and 31.6% across all pollen seasons, compared with those taking placebo (all P ≤ .003). A greater number of patients also achieved a relative improvement in daily medication score during birch season (49.2%), alder and hazel season (43.8%), oak season (45.9%) and during the whole of tree pollen season (45.3%), compared with placebo (P ≤ .002).
“The results support the clinical relevance of cross-reactivity between birch, alder/hazel, and oak pollen homologous allergens,” Dr. Nolte said. “Immunologic cross-reactivity is supported by alder, hazel, and oak specific IgE data and IgG4 in responses to the tree SLIT tablet.”
Dr. Nolte said the next step in his team’s research was to evaluate oral SLIT in a phase 3 trial for children aged 5-17 years.
This study was funded by ALK, and the authors received medical writing and editorial assistance from Scott Medical Communications. Dr. Nolte reported that he is a paid employee of ALK.
SOURCE: Nolte H et al. AAAAI, Abstract 267.
.
REPORTING FROM AAAAI
African Americans with SLE face increased risk of CVD hospitalizations
PHOENIX – African Americans with systemic lupus erythematosus are more likely to experience recurrent hospitalizations for cardiovascular disease, compared with other racial/ethnic groups, results from a single-state registry study found.
“SLE is an autoimmune disease that causes inflammation affecting multiple organ systems including the cardiovascular system,” Meghan Angley, MPH, said at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting. “Therefore, individuals with SLE are at risk for early CVD. African Americans represent the racial group at greatest risk for SLE.”
According to Ms. Angley, with the department of epidemiology at Emory University, Atlanta, white women with SLE have CVD associated mortality 12 years earlier than their non-SLE counterparts, while African American women with SLE have CVD-associated mortality 19 years earlier than their non-SLE counterparts. “We know that recurrent hospitalizations for CVD are associated with mortality,” she said. “These represent potential points of identification of high-risk individuals and also points of interventions.”
In order to study racial disparities across recurrent hospitalizations for cardiovascular disease in an SLE population, Ms. Angley and her colleagues drew from the Georgia Lupus Registry, which is a population-based registry of patients with validated SLE in two Georgia counties. They included all cases diagnosed between 2000 and 2004. The registry was linked to records of all inpatient hospitalizations in Georgia between 2000 and 2013. The researchers used ICD-9 codes to identify hospitalizations for coronary heart disease, peripheral artery disease, cerebrovascular disease, and heart failure and used the Prentice-Williams-Peterson model for recurrent time-to-event analysis. Specifically, they looked at the total time scale from the point of diagnosis to each of the subsequent CVD hospitalizations and truncated the number of hospitalizations at three to maintain stable modeling estimates. The analysis was censored at the time of patient death or at the end of 2013 and adjusted for sex and age at diagnosis.
The sample included 417 African Americans with SLE and 149 non–African Americans with the disease. Most (86%) were female, and the non–African American group was slightly more likely to have been diagnosed with SLE after the age of 45 years, compared with the African American group (36% vs. 30%, respectively).
Ms. Angley and her colleagues found that 24% of African Americans had at least one CVD hospitalization, and 14% had at least two, while 13% of non–African Americans had at least one CVD hospitalization, and 5% had at least two. Among those in the African American group, reasons for hospitalizations were congestive heart failure, (58%), cerebrovascular disease (27%), coronary heart disease (18%), and peripheral artery disease (2%). Among those in the non–African American group, reasons for hospitalizations were congestive heart failure (38%), coronary heart disease (38%), cerebrovascular disease (25%), and peripheral artery disease (6%).
Overall, African American race was associated with recurrent hospitalizations (adjusted hazard ratio, 1.9). In an event-specific stratified analysis, the association between African American race and the hazard of recurrence became even more pronounced with each event (hospitalization 1 aHR, 1.2; hospitalization 2 aHR, 1.5; hospitalization 3 aHR, 1.9). The researchers also observed that African Americans were hospitalized sooner, compared with non–African Americans: a median of 3.68 versus 4.61 years for hospitalization 1, 3.73 years versus 5.98 years for hospitalization 2, and 4.84 years versus 8.14 years for hospitalization 3.
“African Americans with SLE are more likely to experience recurrent hospitalizations for CVD,” Ms. Angley concluded at the meeting, which was sponsored by the American Heart Association. “The events occur sooner after diagnosis than in non–African Americans, suggesting that African Americans may be more vulnerable to the cardiovascular complications of SLE. Our next steps include examining potential reasons for these disparities, such as looking at primary care patterns over time, SLE severity over time, and treatment at CVD hospitalizations.”
In an interview, one of the meeting session’s moderators, Sherry-Ann Brown, MD, called for additional research to determine the reasons for disparities that were observed between African Americans with SLE and their non–African American counterparts. “We need to figure out why and address it,” said Dr. Brown, who is a cardiologist and physician-scientist at Mayo Clinic, Rochester, Minn. “We recognize that social determinants of health, such as insurance, socioeconomic factors, and psychosocial factors, can contribute. We need to figure out the additional steps we need to take in order to close that gap.”
Ms. Angley reported having no disclosures. The study was funded by grants from the Centers for Disease Control and Prevention and by the National Institutes of Health.
SOURCE: Angley M et al. Epi/Lifestyle 2020, Abstract 5.
PHOENIX – African Americans with systemic lupus erythematosus are more likely to experience recurrent hospitalizations for cardiovascular disease, compared with other racial/ethnic groups, results from a single-state registry study found.
“SLE is an autoimmune disease that causes inflammation affecting multiple organ systems including the cardiovascular system,” Meghan Angley, MPH, said at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting. “Therefore, individuals with SLE are at risk for early CVD. African Americans represent the racial group at greatest risk for SLE.”
According to Ms. Angley, with the department of epidemiology at Emory University, Atlanta, white women with SLE have CVD associated mortality 12 years earlier than their non-SLE counterparts, while African American women with SLE have CVD-associated mortality 19 years earlier than their non-SLE counterparts. “We know that recurrent hospitalizations for CVD are associated with mortality,” she said. “These represent potential points of identification of high-risk individuals and also points of interventions.”
In order to study racial disparities across recurrent hospitalizations for cardiovascular disease in an SLE population, Ms. Angley and her colleagues drew from the Georgia Lupus Registry, which is a population-based registry of patients with validated SLE in two Georgia counties. They included all cases diagnosed between 2000 and 2004. The registry was linked to records of all inpatient hospitalizations in Georgia between 2000 and 2013. The researchers used ICD-9 codes to identify hospitalizations for coronary heart disease, peripheral artery disease, cerebrovascular disease, and heart failure and used the Prentice-Williams-Peterson model for recurrent time-to-event analysis. Specifically, they looked at the total time scale from the point of diagnosis to each of the subsequent CVD hospitalizations and truncated the number of hospitalizations at three to maintain stable modeling estimates. The analysis was censored at the time of patient death or at the end of 2013 and adjusted for sex and age at diagnosis.
The sample included 417 African Americans with SLE and 149 non–African Americans with the disease. Most (86%) were female, and the non–African American group was slightly more likely to have been diagnosed with SLE after the age of 45 years, compared with the African American group (36% vs. 30%, respectively).
Ms. Angley and her colleagues found that 24% of African Americans had at least one CVD hospitalization, and 14% had at least two, while 13% of non–African Americans had at least one CVD hospitalization, and 5% had at least two. Among those in the African American group, reasons for hospitalizations were congestive heart failure, (58%), cerebrovascular disease (27%), coronary heart disease (18%), and peripheral artery disease (2%). Among those in the non–African American group, reasons for hospitalizations were congestive heart failure (38%), coronary heart disease (38%), cerebrovascular disease (25%), and peripheral artery disease (6%).
Overall, African American race was associated with recurrent hospitalizations (adjusted hazard ratio, 1.9). In an event-specific stratified analysis, the association between African American race and the hazard of recurrence became even more pronounced with each event (hospitalization 1 aHR, 1.2; hospitalization 2 aHR, 1.5; hospitalization 3 aHR, 1.9). The researchers also observed that African Americans were hospitalized sooner, compared with non–African Americans: a median of 3.68 versus 4.61 years for hospitalization 1, 3.73 years versus 5.98 years for hospitalization 2, and 4.84 years versus 8.14 years for hospitalization 3.
“African Americans with SLE are more likely to experience recurrent hospitalizations for CVD,” Ms. Angley concluded at the meeting, which was sponsored by the American Heart Association. “The events occur sooner after diagnosis than in non–African Americans, suggesting that African Americans may be more vulnerable to the cardiovascular complications of SLE. Our next steps include examining potential reasons for these disparities, such as looking at primary care patterns over time, SLE severity over time, and treatment at CVD hospitalizations.”
In an interview, one of the meeting session’s moderators, Sherry-Ann Brown, MD, called for additional research to determine the reasons for disparities that were observed between African Americans with SLE and their non–African American counterparts. “We need to figure out why and address it,” said Dr. Brown, who is a cardiologist and physician-scientist at Mayo Clinic, Rochester, Minn. “We recognize that social determinants of health, such as insurance, socioeconomic factors, and psychosocial factors, can contribute. We need to figure out the additional steps we need to take in order to close that gap.”
Ms. Angley reported having no disclosures. The study was funded by grants from the Centers for Disease Control and Prevention and by the National Institutes of Health.
SOURCE: Angley M et al. Epi/Lifestyle 2020, Abstract 5.
PHOENIX – African Americans with systemic lupus erythematosus are more likely to experience recurrent hospitalizations for cardiovascular disease, compared with other racial/ethnic groups, results from a single-state registry study found.
“SLE is an autoimmune disease that causes inflammation affecting multiple organ systems including the cardiovascular system,” Meghan Angley, MPH, said at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting. “Therefore, individuals with SLE are at risk for early CVD. African Americans represent the racial group at greatest risk for SLE.”
According to Ms. Angley, with the department of epidemiology at Emory University, Atlanta, white women with SLE have CVD associated mortality 12 years earlier than their non-SLE counterparts, while African American women with SLE have CVD-associated mortality 19 years earlier than their non-SLE counterparts. “We know that recurrent hospitalizations for CVD are associated with mortality,” she said. “These represent potential points of identification of high-risk individuals and also points of interventions.”
In order to study racial disparities across recurrent hospitalizations for cardiovascular disease in an SLE population, Ms. Angley and her colleagues drew from the Georgia Lupus Registry, which is a population-based registry of patients with validated SLE in two Georgia counties. They included all cases diagnosed between 2000 and 2004. The registry was linked to records of all inpatient hospitalizations in Georgia between 2000 and 2013. The researchers used ICD-9 codes to identify hospitalizations for coronary heart disease, peripheral artery disease, cerebrovascular disease, and heart failure and used the Prentice-Williams-Peterson model for recurrent time-to-event analysis. Specifically, they looked at the total time scale from the point of diagnosis to each of the subsequent CVD hospitalizations and truncated the number of hospitalizations at three to maintain stable modeling estimates. The analysis was censored at the time of patient death or at the end of 2013 and adjusted for sex and age at diagnosis.
The sample included 417 African Americans with SLE and 149 non–African Americans with the disease. Most (86%) were female, and the non–African American group was slightly more likely to have been diagnosed with SLE after the age of 45 years, compared with the African American group (36% vs. 30%, respectively).
Ms. Angley and her colleagues found that 24% of African Americans had at least one CVD hospitalization, and 14% had at least two, while 13% of non–African Americans had at least one CVD hospitalization, and 5% had at least two. Among those in the African American group, reasons for hospitalizations were congestive heart failure, (58%), cerebrovascular disease (27%), coronary heart disease (18%), and peripheral artery disease (2%). Among those in the non–African American group, reasons for hospitalizations were congestive heart failure (38%), coronary heart disease (38%), cerebrovascular disease (25%), and peripheral artery disease (6%).
Overall, African American race was associated with recurrent hospitalizations (adjusted hazard ratio, 1.9). In an event-specific stratified analysis, the association between African American race and the hazard of recurrence became even more pronounced with each event (hospitalization 1 aHR, 1.2; hospitalization 2 aHR, 1.5; hospitalization 3 aHR, 1.9). The researchers also observed that African Americans were hospitalized sooner, compared with non–African Americans: a median of 3.68 versus 4.61 years for hospitalization 1, 3.73 years versus 5.98 years for hospitalization 2, and 4.84 years versus 8.14 years for hospitalization 3.
“African Americans with SLE are more likely to experience recurrent hospitalizations for CVD,” Ms. Angley concluded at the meeting, which was sponsored by the American Heart Association. “The events occur sooner after diagnosis than in non–African Americans, suggesting that African Americans may be more vulnerable to the cardiovascular complications of SLE. Our next steps include examining potential reasons for these disparities, such as looking at primary care patterns over time, SLE severity over time, and treatment at CVD hospitalizations.”
In an interview, one of the meeting session’s moderators, Sherry-Ann Brown, MD, called for additional research to determine the reasons for disparities that were observed between African Americans with SLE and their non–African American counterparts. “We need to figure out why and address it,” said Dr. Brown, who is a cardiologist and physician-scientist at Mayo Clinic, Rochester, Minn. “We recognize that social determinants of health, such as insurance, socioeconomic factors, and psychosocial factors, can contribute. We need to figure out the additional steps we need to take in order to close that gap.”
Ms. Angley reported having no disclosures. The study was funded by grants from the Centers for Disease Control and Prevention and by the National Institutes of Health.
SOURCE: Angley M et al. Epi/Lifestyle 2020, Abstract 5.
REPORTING FROM EPI/LIFESTYLE 2020
PARAGON-HF: Optimal systolic pressure in HFpEF is 120-129 mm Hg
A target systolic blood pressure (SBP) of 120-129 mm Hg in patients with heart failure with preserved ejection fraction proved to be the sweet spot with the lowest rates of major adverse cardiovascular and renal events in a new analysis from the landmark PARAGON-HF trial.
This finding from the largest-ever randomized, controlled study in heart failure with preserved ejection fraction (HFpEF) strengthens support for current U.S. joint hypertension guidelines, which call for a target SBP less than 130 mm Hg in patients with HFpEF (J Am Coll Cardiol. 2017 Aug 8;70[6]:776-803), a recommendation based upon weak evidence until now. That’s because the SPRINT trial, the major impetus for adoption of intensive blood pressure control in the current guidelines, excluded patients with symptomatic HF, Scott D. Solomon, MD, and coinvestigators noted in their new analysis. The study was published in the Journal of the American College of Cardiology and had been planned for presentation during the joint scientific sessions of the American College of Cardiology and the World Heart Federation. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
The new analysis from PARAGON-HF (Prospective Comparison of ARNI with ARB Global Outcomes in HFpEF) also ruled out the SBP-lowering effect of sacubitril/valsartan (Entresto) as the explanation for the combination drug’s demonstrated beneficial impact on outcomes in the subgroup with an SBP of 120-129 mm Hg. That wasn’t actually a surprise. Indeed, the new study had two hypotheses: one, that the relationship between SBP and cardiovascular and renal outcomes in HFpEF would follow a J-shaped curve, and two, that sacubitril/valsartan’s blood pressure–lowering effect would not account for the drug’s outcome benefits in the subset of HFpEF patients with an SBP in the sweet spot of 120-129 mm Hg. Both hypotheses were borne out, noted Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, both in Boston.
“These data strongly support that additional mechanisms other than blood pressure–lowering account for the benefit. But this is not surprising. The same can be said for most of the therapies that work in heart failure,” he said in an interview.
Take, for example, spironolactone. In TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist), another major trial in which Dr. Solomon played a leadership role, the beneficial effect of spironolactone on clinical outcomes also proved unrelated to the drug’s blood pressure–lowering effect.
Other known effects of sacubitril/valsartan, a novel angiotensin receptor–neprilysin inhibitor, or ARNI, might in theory account for the observed clinical benefits in ARNI-treated patients with an on-treatment SBP of 120-129 mm Hg in PARAGON-HF. These include improved left atrial remodeling, an increase in natriuretic peptides, and improved myocardial relaxation. However, the current lack of understanding of the basic mechanistic processes underlying the varied clinical expressions of HFpEF is a major factor contributing to the lack of any proven-effective therapy for this extremely common and costly disorder, according to Dr. Solomon and coinvestigators.
In contrast to HFpEF, for which to date there is no proven treatment, heart failure with reduced ejection fraction sacubitril/valsartan has a class I recommendation on the strength of its performance in significantly reducing cardiovascular deaths and heart failure hospitalizations in the PARADIGM-HF trial (N Engl J Med. 2014 Sep 11;371:993-1004).
PARAGON-HF included 4,822 patients with symptomatic HFpEF who were randomized to sacubitril/valsartan at 97/103 mg b.i.d. or valsartan at 160 mg b.i.d. As previously reported (N Engl J Med. 2019 Oct 24;381[17]:1609-20), at an average follow-up of 35 months, the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – occurred at a rate of 12.8 events per 100 patient-years in the sacubitril/valsartan group and 14.6 per 100 patient-years in the valsartan arm, for a 13% relative risk reduction that narrowly missed statistical significance (P = .059).
However, sacubitril/valsartan showed significant benefit on some prespecified secondary endpoints, including worsening renal function, change in New York Heart Association class, and quality of life. Women, who notably accounted for 52% of study participants, appeared to benefit from sacubitril/valsartan more than men as evidenced by their 27% relative risk reduction in the primary endpoint. Also, in the roughly half of PARAGON-HF participants with a baseline left ventricular ejection fraction of 45%-57%, treatment with sacubitril/valsartan resulted in a statistically significant 22% relative risk reduction in the primary endpoint, compared with valsartan alone.
SBP and cardiovascular outcomes in HFpEF
In the new analysis, Dr. Solomon and coworkers examined outcomes based on baseline and mean achieved SBP quartiles regardless of treatment arm. In an unadjusted analysis, the primary composite endpoint occurred at a rate of 15.2 events/100 patient-years in HFpEF patients with an achieved SBP below 120 mm Hg, 11.4/100 patient-years at 120-129 mm Hg, 12.2/100 patient-years at 130-139 mm Hg, and 15.6/100 patient-years at 140 mm Hg or more. Further, in a multivariate regression analysis extensively adjusted for atrial fibrillation, sex, race, and numerous other potential confounders, the group with an achieved SBP of 120-129 mm Hg continued to fare best. The adjusted risks for the primary endpoint were 11% and 21% higher in patients in the first and third quartiles of achieved SBP, compared with those at 120-129 mm Hg, although neither trend reached statistical significance. But patients in the top quartile, with an achieved SBP of 140 mm Hg or more, had a highly significant 56% increase in risk, compared with patients in the second-lowest SBP quartile.
Change in blood pressure from baseline to week 48 had no impact on quality of life or high-sensitivity troponin T. However, each 10–mm Hg lowering of SBP was associated with a modest 2.1% reduction in log-transformed N-terminal of the prohormone brain natriuretic peptide.
Sacubitril/valsartan reduced SBP by an average of 5.2 mm Hg more than valsartan alone at 4 weeks regardless of baseline SBP. And the combo drug had a significantly greater SBP-lowering effect in women than men, by a margin of 6.3 mm Hg versus 4.0 mm Hg. But a Cox regression analysis showed that in women, as in the study population as a whole, sacubitril/valsartan’s SBP-lowering effects didn’t account for the drug’s impact on outcomes.
In an editorial accompanying publication of the new PARAGON-HF blood pressure analysis (J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.024), Hector O. Ventura, MD, and colleagues at the Ochsner Clinic in New Orleans observed that the study results “lend some credence to the prognostic relationship of blood pressure in HFpEF, but whether they should serve as a therapeutic target or are merely a prognostic surrogate determined by other pathogenic factors, such as vascular ventricular uncoupling or aortic stiffness on one hand when blood pressure is greater than 140 mm Hg, or a reduced cardiac performance indicated by reduced blood pressure to less than 120 mm Hg, remains uncertain.”
“What is certain, however, is that the relationship and contributions of hypertension in manifest HFpEF are complex, multifactorial and likely go well beyond a simplistic framework of hemodynamic influences,” they added.
Dr. Solomon has received research grants from and serves as a consultant to Novartis, which funded PARAGON-HF, and has similar financial relationships with more than a dozen other pharmaceutical companies. Dr. Ventura reported having no relevant financial interests.
SOURCE: Solomon SD et al. J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.009.
A target systolic blood pressure (SBP) of 120-129 mm Hg in patients with heart failure with preserved ejection fraction proved to be the sweet spot with the lowest rates of major adverse cardiovascular and renal events in a new analysis from the landmark PARAGON-HF trial.
This finding from the largest-ever randomized, controlled study in heart failure with preserved ejection fraction (HFpEF) strengthens support for current U.S. joint hypertension guidelines, which call for a target SBP less than 130 mm Hg in patients with HFpEF (J Am Coll Cardiol. 2017 Aug 8;70[6]:776-803), a recommendation based upon weak evidence until now. That’s because the SPRINT trial, the major impetus for adoption of intensive blood pressure control in the current guidelines, excluded patients with symptomatic HF, Scott D. Solomon, MD, and coinvestigators noted in their new analysis. The study was published in the Journal of the American College of Cardiology and had been planned for presentation during the joint scientific sessions of the American College of Cardiology and the World Heart Federation. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
The new analysis from PARAGON-HF (Prospective Comparison of ARNI with ARB Global Outcomes in HFpEF) also ruled out the SBP-lowering effect of sacubitril/valsartan (Entresto) as the explanation for the combination drug’s demonstrated beneficial impact on outcomes in the subgroup with an SBP of 120-129 mm Hg. That wasn’t actually a surprise. Indeed, the new study had two hypotheses: one, that the relationship between SBP and cardiovascular and renal outcomes in HFpEF would follow a J-shaped curve, and two, that sacubitril/valsartan’s blood pressure–lowering effect would not account for the drug’s outcome benefits in the subset of HFpEF patients with an SBP in the sweet spot of 120-129 mm Hg. Both hypotheses were borne out, noted Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, both in Boston.
“These data strongly support that additional mechanisms other than blood pressure–lowering account for the benefit. But this is not surprising. The same can be said for most of the therapies that work in heart failure,” he said in an interview.
Take, for example, spironolactone. In TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist), another major trial in which Dr. Solomon played a leadership role, the beneficial effect of spironolactone on clinical outcomes also proved unrelated to the drug’s blood pressure–lowering effect.
Other known effects of sacubitril/valsartan, a novel angiotensin receptor–neprilysin inhibitor, or ARNI, might in theory account for the observed clinical benefits in ARNI-treated patients with an on-treatment SBP of 120-129 mm Hg in PARAGON-HF. These include improved left atrial remodeling, an increase in natriuretic peptides, and improved myocardial relaxation. However, the current lack of understanding of the basic mechanistic processes underlying the varied clinical expressions of HFpEF is a major factor contributing to the lack of any proven-effective therapy for this extremely common and costly disorder, according to Dr. Solomon and coinvestigators.
In contrast to HFpEF, for which to date there is no proven treatment, heart failure with reduced ejection fraction sacubitril/valsartan has a class I recommendation on the strength of its performance in significantly reducing cardiovascular deaths and heart failure hospitalizations in the PARADIGM-HF trial (N Engl J Med. 2014 Sep 11;371:993-1004).
PARAGON-HF included 4,822 patients with symptomatic HFpEF who were randomized to sacubitril/valsartan at 97/103 mg b.i.d. or valsartan at 160 mg b.i.d. As previously reported (N Engl J Med. 2019 Oct 24;381[17]:1609-20), at an average follow-up of 35 months, the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – occurred at a rate of 12.8 events per 100 patient-years in the sacubitril/valsartan group and 14.6 per 100 patient-years in the valsartan arm, for a 13% relative risk reduction that narrowly missed statistical significance (P = .059).
However, sacubitril/valsartan showed significant benefit on some prespecified secondary endpoints, including worsening renal function, change in New York Heart Association class, and quality of life. Women, who notably accounted for 52% of study participants, appeared to benefit from sacubitril/valsartan more than men as evidenced by their 27% relative risk reduction in the primary endpoint. Also, in the roughly half of PARAGON-HF participants with a baseline left ventricular ejection fraction of 45%-57%, treatment with sacubitril/valsartan resulted in a statistically significant 22% relative risk reduction in the primary endpoint, compared with valsartan alone.
SBP and cardiovascular outcomes in HFpEF
In the new analysis, Dr. Solomon and coworkers examined outcomes based on baseline and mean achieved SBP quartiles regardless of treatment arm. In an unadjusted analysis, the primary composite endpoint occurred at a rate of 15.2 events/100 patient-years in HFpEF patients with an achieved SBP below 120 mm Hg, 11.4/100 patient-years at 120-129 mm Hg, 12.2/100 patient-years at 130-139 mm Hg, and 15.6/100 patient-years at 140 mm Hg or more. Further, in a multivariate regression analysis extensively adjusted for atrial fibrillation, sex, race, and numerous other potential confounders, the group with an achieved SBP of 120-129 mm Hg continued to fare best. The adjusted risks for the primary endpoint were 11% and 21% higher in patients in the first and third quartiles of achieved SBP, compared with those at 120-129 mm Hg, although neither trend reached statistical significance. But patients in the top quartile, with an achieved SBP of 140 mm Hg or more, had a highly significant 56% increase in risk, compared with patients in the second-lowest SBP quartile.
Change in blood pressure from baseline to week 48 had no impact on quality of life or high-sensitivity troponin T. However, each 10–mm Hg lowering of SBP was associated with a modest 2.1% reduction in log-transformed N-terminal of the prohormone brain natriuretic peptide.
Sacubitril/valsartan reduced SBP by an average of 5.2 mm Hg more than valsartan alone at 4 weeks regardless of baseline SBP. And the combo drug had a significantly greater SBP-lowering effect in women than men, by a margin of 6.3 mm Hg versus 4.0 mm Hg. But a Cox regression analysis showed that in women, as in the study population as a whole, sacubitril/valsartan’s SBP-lowering effects didn’t account for the drug’s impact on outcomes.
In an editorial accompanying publication of the new PARAGON-HF blood pressure analysis (J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.024), Hector O. Ventura, MD, and colleagues at the Ochsner Clinic in New Orleans observed that the study results “lend some credence to the prognostic relationship of blood pressure in HFpEF, but whether they should serve as a therapeutic target or are merely a prognostic surrogate determined by other pathogenic factors, such as vascular ventricular uncoupling or aortic stiffness on one hand when blood pressure is greater than 140 mm Hg, or a reduced cardiac performance indicated by reduced blood pressure to less than 120 mm Hg, remains uncertain.”
“What is certain, however, is that the relationship and contributions of hypertension in manifest HFpEF are complex, multifactorial and likely go well beyond a simplistic framework of hemodynamic influences,” they added.
Dr. Solomon has received research grants from and serves as a consultant to Novartis, which funded PARAGON-HF, and has similar financial relationships with more than a dozen other pharmaceutical companies. Dr. Ventura reported having no relevant financial interests.
SOURCE: Solomon SD et al. J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.009.
A target systolic blood pressure (SBP) of 120-129 mm Hg in patients with heart failure with preserved ejection fraction proved to be the sweet spot with the lowest rates of major adverse cardiovascular and renal events in a new analysis from the landmark PARAGON-HF trial.
This finding from the largest-ever randomized, controlled study in heart failure with preserved ejection fraction (HFpEF) strengthens support for current U.S. joint hypertension guidelines, which call for a target SBP less than 130 mm Hg in patients with HFpEF (J Am Coll Cardiol. 2017 Aug 8;70[6]:776-803), a recommendation based upon weak evidence until now. That’s because the SPRINT trial, the major impetus for adoption of intensive blood pressure control in the current guidelines, excluded patients with symptomatic HF, Scott D. Solomon, MD, and coinvestigators noted in their new analysis. The study was published in the Journal of the American College of Cardiology and had been planned for presentation during the joint scientific sessions of the American College of Cardiology and the World Heart Federation. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
The new analysis from PARAGON-HF (Prospective Comparison of ARNI with ARB Global Outcomes in HFpEF) also ruled out the SBP-lowering effect of sacubitril/valsartan (Entresto) as the explanation for the combination drug’s demonstrated beneficial impact on outcomes in the subgroup with an SBP of 120-129 mm Hg. That wasn’t actually a surprise. Indeed, the new study had two hypotheses: one, that the relationship between SBP and cardiovascular and renal outcomes in HFpEF would follow a J-shaped curve, and two, that sacubitril/valsartan’s blood pressure–lowering effect would not account for the drug’s outcome benefits in the subset of HFpEF patients with an SBP in the sweet spot of 120-129 mm Hg. Both hypotheses were borne out, noted Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, both in Boston.
“These data strongly support that additional mechanisms other than blood pressure–lowering account for the benefit. But this is not surprising. The same can be said for most of the therapies that work in heart failure,” he said in an interview.
Take, for example, spironolactone. In TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist), another major trial in which Dr. Solomon played a leadership role, the beneficial effect of spironolactone on clinical outcomes also proved unrelated to the drug’s blood pressure–lowering effect.
Other known effects of sacubitril/valsartan, a novel angiotensin receptor–neprilysin inhibitor, or ARNI, might in theory account for the observed clinical benefits in ARNI-treated patients with an on-treatment SBP of 120-129 mm Hg in PARAGON-HF. These include improved left atrial remodeling, an increase in natriuretic peptides, and improved myocardial relaxation. However, the current lack of understanding of the basic mechanistic processes underlying the varied clinical expressions of HFpEF is a major factor contributing to the lack of any proven-effective therapy for this extremely common and costly disorder, according to Dr. Solomon and coinvestigators.
In contrast to HFpEF, for which to date there is no proven treatment, heart failure with reduced ejection fraction sacubitril/valsartan has a class I recommendation on the strength of its performance in significantly reducing cardiovascular deaths and heart failure hospitalizations in the PARADIGM-HF trial (N Engl J Med. 2014 Sep 11;371:993-1004).
PARAGON-HF included 4,822 patients with symptomatic HFpEF who were randomized to sacubitril/valsartan at 97/103 mg b.i.d. or valsartan at 160 mg b.i.d. As previously reported (N Engl J Med. 2019 Oct 24;381[17]:1609-20), at an average follow-up of 35 months, the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – occurred at a rate of 12.8 events per 100 patient-years in the sacubitril/valsartan group and 14.6 per 100 patient-years in the valsartan arm, for a 13% relative risk reduction that narrowly missed statistical significance (P = .059).
However, sacubitril/valsartan showed significant benefit on some prespecified secondary endpoints, including worsening renal function, change in New York Heart Association class, and quality of life. Women, who notably accounted for 52% of study participants, appeared to benefit from sacubitril/valsartan more than men as evidenced by their 27% relative risk reduction in the primary endpoint. Also, in the roughly half of PARAGON-HF participants with a baseline left ventricular ejection fraction of 45%-57%, treatment with sacubitril/valsartan resulted in a statistically significant 22% relative risk reduction in the primary endpoint, compared with valsartan alone.
SBP and cardiovascular outcomes in HFpEF
In the new analysis, Dr. Solomon and coworkers examined outcomes based on baseline and mean achieved SBP quartiles regardless of treatment arm. In an unadjusted analysis, the primary composite endpoint occurred at a rate of 15.2 events/100 patient-years in HFpEF patients with an achieved SBP below 120 mm Hg, 11.4/100 patient-years at 120-129 mm Hg, 12.2/100 patient-years at 130-139 mm Hg, and 15.6/100 patient-years at 140 mm Hg or more. Further, in a multivariate regression analysis extensively adjusted for atrial fibrillation, sex, race, and numerous other potential confounders, the group with an achieved SBP of 120-129 mm Hg continued to fare best. The adjusted risks for the primary endpoint were 11% and 21% higher in patients in the first and third quartiles of achieved SBP, compared with those at 120-129 mm Hg, although neither trend reached statistical significance. But patients in the top quartile, with an achieved SBP of 140 mm Hg or more, had a highly significant 56% increase in risk, compared with patients in the second-lowest SBP quartile.
Change in blood pressure from baseline to week 48 had no impact on quality of life or high-sensitivity troponin T. However, each 10–mm Hg lowering of SBP was associated with a modest 2.1% reduction in log-transformed N-terminal of the prohormone brain natriuretic peptide.
Sacubitril/valsartan reduced SBP by an average of 5.2 mm Hg more than valsartan alone at 4 weeks regardless of baseline SBP. And the combo drug had a significantly greater SBP-lowering effect in women than men, by a margin of 6.3 mm Hg versus 4.0 mm Hg. But a Cox regression analysis showed that in women, as in the study population as a whole, sacubitril/valsartan’s SBP-lowering effects didn’t account for the drug’s impact on outcomes.
In an editorial accompanying publication of the new PARAGON-HF blood pressure analysis (J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.024), Hector O. Ventura, MD, and colleagues at the Ochsner Clinic in New Orleans observed that the study results “lend some credence to the prognostic relationship of blood pressure in HFpEF, but whether they should serve as a therapeutic target or are merely a prognostic surrogate determined by other pathogenic factors, such as vascular ventricular uncoupling or aortic stiffness on one hand when blood pressure is greater than 140 mm Hg, or a reduced cardiac performance indicated by reduced blood pressure to less than 120 mm Hg, remains uncertain.”
“What is certain, however, is that the relationship and contributions of hypertension in manifest HFpEF are complex, multifactorial and likely go well beyond a simplistic framework of hemodynamic influences,” they added.
Dr. Solomon has received research grants from and serves as a consultant to Novartis, which funded PARAGON-HF, and has similar financial relationships with more than a dozen other pharmaceutical companies. Dr. Ventura reported having no relevant financial interests.
SOURCE: Solomon SD et al. J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.009.
FROM ACC 2020
CME in the time of COVID-19
As the COVID-19 pandemic spreads, it now seems like the norm is that large medical conferences are being canceled.
The American Psychiatric Association (APA) canceled its 2020 annual meeting, which was scheduled for late April. The cancellation disappointed many, because we will miss out on the camaraderie and professional invigoration that comes from gathering with psychiatrists and other mental health professionals from across the United States and around the world. After the APA’s decision was announced, the White House released guidelines advising Americans to avoid social gatherings of 10 or more people.
On a practical level, many psychiatrists will not be able to earn up to 35 continuing medical education credits (CME) from attending the meeting and fulfilling the administrative requirements to obtain a CME certificate. Not only have meetings been canceled, but events many other clinicians count on for CME, such as journal clubs and department grand rounds, have been canceled until they can be moved to a virtual space.
The CME requirements for state medical licenses vary widely. On average, most states require at least 25 credits per year or 60 to 100 credits every 2 years, and the American Board of Psychiatry and Neurology requires diplomates to complete an average of 30 specialty and/or subspecialty CME credits per year, averaged over 3 years. Usually, annual medical conferences would be a great way to get an infusion of CME credits, brush up on cutting-edge treatments, and review the basics.
On top of everything else we have to worry about with COVID-19, getting enough CME credits has been added to the list for many psychiatrists and mental health clinicians. As our schedules and daily lives are disrupted, it’s important to find relief in routine activities that are not affected by social distancing and fears of isolation and quarantine. A routine activity to lean into might include learning or practicing a skill that we enjoy, such as psychiatry (hopefully!) and the practice of medicine. The CME could be focused on a psychiatric topic or perhaps learning about the specifics of COVID-19 or brushing up on medical knowledge that might be a bit rusty after many years of practicing solely psychiatry.
As you start to gather CME credits online, it’s helpful to sign up for a service that stores your CME credits and helps you keep track of the number. When it comes time to renew your medical license or apply for maintenance of certification (MOC), who wants to be the person searching through their email for PDFs of CME certificates or taking pictures or scanning paper certificates? The APA has a section under education and MOC to track certificates earned by watching online modules from its “Learning Center.” The website also allows users to upload external certificates. The American Medical Association offers a similar service on its “Ed Hub,” in which users can log in to watch, listen, or download articles to earn CME credits after finishing the associated quiz. Medscape, in the CME and Education section, also offers an easy-to-use CME dashboard, in which clinicians can filter by their specialty, topic, duration of learning activity – ranging from 0.25 to 3 CME credits. Clinicians also can track their credits as they complete activities.
If you’re someone who’s having trouble focusing on anything besides COVID-19, there are COVID-19-specific CME activities that are available and can help psychiatrists feel comfortable talking with patients, family, and their institutions about the risks of COVID-19. The AMA Ed Hub has a featured 8-credit CME course about the novel coronavirus with updates about diagnosis, treatment, and public health strategies.
For the psychiatrists who may have procrastinated in-depth learning about the opioid crisis or getting their buprenorphine waivers, AMA Ed Hub offers a 42-credit course about opioids and pain management covering guidelines, research, and treatment.
For fun refreshers on general medicine, the New England Journal of Medicine offers up to 20 online CME exams based on quizzes from interesting clinical cases ranging from “regular” medicine to rare clinical scenarios. The APA Learning Center has an easy-to-use search function allowing users to select content from more than 200 modules covering a wide range of general topics; from reviewing recent treatment guidelines to specialized psychiatric topics such as geriatric bipolar disorder. A psychiatrist who has been quickly pushed to telepsychiatry because of the current pandemic could use the APA Learning Center to find educational modules about risk management in telepsychiatry or learn the special considerations of using telepsychiatry to treat patients with serious mental illness.
Using podcasts to earn CME is becoming increasingly common, with such as outlets as JAMA Networks offering podcasts in many specialties in which subscribers can take a quiz through the JAMA app and obtain CME credits.
As our clinical boundaries as psychiatrists are pushed by an ever-changing public health situation, now is the time to earn CME focused on new topics to meet the demands placed on health care workers at the front lines of clinical care.
If the COVID-19 pandemic reaches the number of cases predicted by public health officials, our health care system is going to be under extreme stress. All specialties face the threat of losing part of their working capacity as clinicians get sick with the virus, or as they stay home because of exposure or to take care of a loved one. CME can be a way to empower ourselves by staying current on the cutting edge of our specialties, but also brushing up on the medicine that we may be asked to practice in a time of great need.
Dr. Posada is consultation-liaison psychiatry fellow with the Inova Fairfax Hospital/George Washington University program in Falls Church, Va. She also is associate producer of the MDedge Psychcast. Dr. Posada has no disclosures.
As the COVID-19 pandemic spreads, it now seems like the norm is that large medical conferences are being canceled.
The American Psychiatric Association (APA) canceled its 2020 annual meeting, which was scheduled for late April. The cancellation disappointed many, because we will miss out on the camaraderie and professional invigoration that comes from gathering with psychiatrists and other mental health professionals from across the United States and around the world. After the APA’s decision was announced, the White House released guidelines advising Americans to avoid social gatherings of 10 or more people.
On a practical level, many psychiatrists will not be able to earn up to 35 continuing medical education credits (CME) from attending the meeting and fulfilling the administrative requirements to obtain a CME certificate. Not only have meetings been canceled, but events many other clinicians count on for CME, such as journal clubs and department grand rounds, have been canceled until they can be moved to a virtual space.
The CME requirements for state medical licenses vary widely. On average, most states require at least 25 credits per year or 60 to 100 credits every 2 years, and the American Board of Psychiatry and Neurology requires diplomates to complete an average of 30 specialty and/or subspecialty CME credits per year, averaged over 3 years. Usually, annual medical conferences would be a great way to get an infusion of CME credits, brush up on cutting-edge treatments, and review the basics.
On top of everything else we have to worry about with COVID-19, getting enough CME credits has been added to the list for many psychiatrists and mental health clinicians. As our schedules and daily lives are disrupted, it’s important to find relief in routine activities that are not affected by social distancing and fears of isolation and quarantine. A routine activity to lean into might include learning or practicing a skill that we enjoy, such as psychiatry (hopefully!) and the practice of medicine. The CME could be focused on a psychiatric topic or perhaps learning about the specifics of COVID-19 or brushing up on medical knowledge that might be a bit rusty after many years of practicing solely psychiatry.
As you start to gather CME credits online, it’s helpful to sign up for a service that stores your CME credits and helps you keep track of the number. When it comes time to renew your medical license or apply for maintenance of certification (MOC), who wants to be the person searching through their email for PDFs of CME certificates or taking pictures or scanning paper certificates? The APA has a section under education and MOC to track certificates earned by watching online modules from its “Learning Center.” The website also allows users to upload external certificates. The American Medical Association offers a similar service on its “Ed Hub,” in which users can log in to watch, listen, or download articles to earn CME credits after finishing the associated quiz. Medscape, in the CME and Education section, also offers an easy-to-use CME dashboard, in which clinicians can filter by their specialty, topic, duration of learning activity – ranging from 0.25 to 3 CME credits. Clinicians also can track their credits as they complete activities.
If you’re someone who’s having trouble focusing on anything besides COVID-19, there are COVID-19-specific CME activities that are available and can help psychiatrists feel comfortable talking with patients, family, and their institutions about the risks of COVID-19. The AMA Ed Hub has a featured 8-credit CME course about the novel coronavirus with updates about diagnosis, treatment, and public health strategies.
For the psychiatrists who may have procrastinated in-depth learning about the opioid crisis or getting their buprenorphine waivers, AMA Ed Hub offers a 42-credit course about opioids and pain management covering guidelines, research, and treatment.
For fun refreshers on general medicine, the New England Journal of Medicine offers up to 20 online CME exams based on quizzes from interesting clinical cases ranging from “regular” medicine to rare clinical scenarios. The APA Learning Center has an easy-to-use search function allowing users to select content from more than 200 modules covering a wide range of general topics; from reviewing recent treatment guidelines to specialized psychiatric topics such as geriatric bipolar disorder. A psychiatrist who has been quickly pushed to telepsychiatry because of the current pandemic could use the APA Learning Center to find educational modules about risk management in telepsychiatry or learn the special considerations of using telepsychiatry to treat patients with serious mental illness.
Using podcasts to earn CME is becoming increasingly common, with such as outlets as JAMA Networks offering podcasts in many specialties in which subscribers can take a quiz through the JAMA app and obtain CME credits.
As our clinical boundaries as psychiatrists are pushed by an ever-changing public health situation, now is the time to earn CME focused on new topics to meet the demands placed on health care workers at the front lines of clinical care.
If the COVID-19 pandemic reaches the number of cases predicted by public health officials, our health care system is going to be under extreme stress. All specialties face the threat of losing part of their working capacity as clinicians get sick with the virus, or as they stay home because of exposure or to take care of a loved one. CME can be a way to empower ourselves by staying current on the cutting edge of our specialties, but also brushing up on the medicine that we may be asked to practice in a time of great need.
Dr. Posada is consultation-liaison psychiatry fellow with the Inova Fairfax Hospital/George Washington University program in Falls Church, Va. She also is associate producer of the MDedge Psychcast. Dr. Posada has no disclosures.
As the COVID-19 pandemic spreads, it now seems like the norm is that large medical conferences are being canceled.
The American Psychiatric Association (APA) canceled its 2020 annual meeting, which was scheduled for late April. The cancellation disappointed many, because we will miss out on the camaraderie and professional invigoration that comes from gathering with psychiatrists and other mental health professionals from across the United States and around the world. After the APA’s decision was announced, the White House released guidelines advising Americans to avoid social gatherings of 10 or more people.
On a practical level, many psychiatrists will not be able to earn up to 35 continuing medical education credits (CME) from attending the meeting and fulfilling the administrative requirements to obtain a CME certificate. Not only have meetings been canceled, but events many other clinicians count on for CME, such as journal clubs and department grand rounds, have been canceled until they can be moved to a virtual space.
The CME requirements for state medical licenses vary widely. On average, most states require at least 25 credits per year or 60 to 100 credits every 2 years, and the American Board of Psychiatry and Neurology requires diplomates to complete an average of 30 specialty and/or subspecialty CME credits per year, averaged over 3 years. Usually, annual medical conferences would be a great way to get an infusion of CME credits, brush up on cutting-edge treatments, and review the basics.
On top of everything else we have to worry about with COVID-19, getting enough CME credits has been added to the list for many psychiatrists and mental health clinicians. As our schedules and daily lives are disrupted, it’s important to find relief in routine activities that are not affected by social distancing and fears of isolation and quarantine. A routine activity to lean into might include learning or practicing a skill that we enjoy, such as psychiatry (hopefully!) and the practice of medicine. The CME could be focused on a psychiatric topic or perhaps learning about the specifics of COVID-19 or brushing up on medical knowledge that might be a bit rusty after many years of practicing solely psychiatry.
As you start to gather CME credits online, it’s helpful to sign up for a service that stores your CME credits and helps you keep track of the number. When it comes time to renew your medical license or apply for maintenance of certification (MOC), who wants to be the person searching through their email for PDFs of CME certificates or taking pictures or scanning paper certificates? The APA has a section under education and MOC to track certificates earned by watching online modules from its “Learning Center.” The website also allows users to upload external certificates. The American Medical Association offers a similar service on its “Ed Hub,” in which users can log in to watch, listen, or download articles to earn CME credits after finishing the associated quiz. Medscape, in the CME and Education section, also offers an easy-to-use CME dashboard, in which clinicians can filter by their specialty, topic, duration of learning activity – ranging from 0.25 to 3 CME credits. Clinicians also can track their credits as they complete activities.
If you’re someone who’s having trouble focusing on anything besides COVID-19, there are COVID-19-specific CME activities that are available and can help psychiatrists feel comfortable talking with patients, family, and their institutions about the risks of COVID-19. The AMA Ed Hub has a featured 8-credit CME course about the novel coronavirus with updates about diagnosis, treatment, and public health strategies.
For the psychiatrists who may have procrastinated in-depth learning about the opioid crisis or getting their buprenorphine waivers, AMA Ed Hub offers a 42-credit course about opioids and pain management covering guidelines, research, and treatment.
For fun refreshers on general medicine, the New England Journal of Medicine offers up to 20 online CME exams based on quizzes from interesting clinical cases ranging from “regular” medicine to rare clinical scenarios. The APA Learning Center has an easy-to-use search function allowing users to select content from more than 200 modules covering a wide range of general topics; from reviewing recent treatment guidelines to specialized psychiatric topics such as geriatric bipolar disorder. A psychiatrist who has been quickly pushed to telepsychiatry because of the current pandemic could use the APA Learning Center to find educational modules about risk management in telepsychiatry or learn the special considerations of using telepsychiatry to treat patients with serious mental illness.
Using podcasts to earn CME is becoming increasingly common, with such as outlets as JAMA Networks offering podcasts in many specialties in which subscribers can take a quiz through the JAMA app and obtain CME credits.
As our clinical boundaries as psychiatrists are pushed by an ever-changing public health situation, now is the time to earn CME focused on new topics to meet the demands placed on health care workers at the front lines of clinical care.
If the COVID-19 pandemic reaches the number of cases predicted by public health officials, our health care system is going to be under extreme stress. All specialties face the threat of losing part of their working capacity as clinicians get sick with the virus, or as they stay home because of exposure or to take care of a loved one. CME can be a way to empower ourselves by staying current on the cutting edge of our specialties, but also brushing up on the medicine that we may be asked to practice in a time of great need.
Dr. Posada is consultation-liaison psychiatry fellow with the Inova Fairfax Hospital/George Washington University program in Falls Church, Va. She also is associate producer of the MDedge Psychcast. Dr. Posada has no disclosures.
During a viral pandemic, anxiety is endemic: The psychiatric aspects of COVID-19
Fear of dying is considered “normal.” However, the ongoing threat of a potentially fatal viral infection can cause panic, anxiety, and an exaggerated fear of illness and death. The relentless spread of the coronavirus infectious disease that began in late 2019 (COVID-19) is spawning widespread anxiety, panic, and worry about one’s health and the health of loved ones. The viral pandemic has triggered a parallel anxiety epidemic.
Making things worse is that no vaccine has yet been developed, and for individuals who do get infected, there are no specific treatments other than supportive care, such as ventilators. Members of the public have been urged to practice sensible preventative measures, including handwashing, sanitizing certain items and surfaces, and—particularly challenging—self-isolation and social distancing. The public has channeled its fear into frantic buying and hoarding of food and non-food items, especially masks, sanitizers, soap, disinfectant wipes, and toilet paper (perhaps preparing for gastrointestinal hyperactivity during anxiety); canceling flights; avoiding group activities; and self-isolation or, for those exposed to the virus, quarantine. Anxiety is palpable. The facial masks that people wear are ironically unmasking their inner agitation and disquietude.
Our role as psychiatrists
As psychiatrists, we have an important role to play in such times, especially for our patients who already have anxiety disorders or depression. The additional emotional burden of this escalating health crisis is exacerbating the mental anguish of our patients (in addition to those who may soon become new patients). The anxiety and panic attacks due to “imagined” doom and gloom are now intensified by anxiety due to a “real” fatal threat. The effect on some vulnerable patients can be devastating, and may culminate in an acute stress reaction and future posttraumatic stress disorder. There are also reports of “psychogenic COVID-19” conversion reaction, with symptoms of sore throat, dyspnea, and even psychogenic fever. Paradoxically, self-isolation and social distancing, which are recommended to prevent the human-to-human spread of the virus, may further worsen anxiety and depression by reducing the comfort of intimacy and social contacts.
Individuals with depression will also experience an increased risk of symptom breakthrough despite receiving treatment. Stress is well known to trigger or exacerbate depression. Thus, the sense of helplessness and hopelessness during depression may intensify among our patients with pre-existing mood disorders, and suicidal ideation may resurface. Making things worse is the unfortunate timing of the COVID-19 pandemic. Spring is the peak season for the re-emergence of depression and suicide attempts. The ongoing stress of the health crisis, coupled with the onset of spring, may coalesce into a dreadful synergy for relapse among vulnerable individuals with unipolar or bipolar depression.
Patients with obsessive-compulsive disorder (OCD) are known to be averse to imagined germs and may wash their hands multiple times a day. An epidemic in which all health officials strongly urge washing one’s hands is very likely to exacerbate the compulsive handwashing of persons with OCD and significantly increase their anxiety. Because their other obsessions and compulsions may also increase in frequency and intensity, they will need our attention as their psychiatrists.
The viral pandemic is eerily similar to a natural disaster such as a hurricane of tornado, both of which physically destroy towns and flatten homes. The COVID-19 pandemic is damaging social structures and obliterating the fabric of global human relations. Consider the previously unimaginable disruption of what makes a vibrant society: schools, colleges, sporting events, concerts, Broadway shows, houses of worship, festivals, conferences, conventions, busy airports/train stations/bus stations, and spontaneous community gatherings. The sudden shock of upheaval in our daily lives may not only cause a hollow sense of emptiness and grief, but also have residual economic and emotional consequences. Nothing can be taken for granted anymore, and nothing is permanent. Cynicism may rise about maintaining life as we know it.
Rising to the challenge
Physicians and clinicians across all specialties are rising to the challenge of the pandemic, whether to manage the immediate physical or emotional impacts of the health crisis or its anticipated consequences (including the economic sequelae). The often-demonized pharmaceutical industry is urgently summoning all its resources to develop both a vaccine as well as biologic treatments for this potentially fatal viral infection. The government is removing regulatory barriers to expedite solutions to the crisis. A welcome public-private partnership is expediting the availability of and access to testing for the virus. The toxic political partisanship has temporarily given way to collaboration in crafting laws that can mitigate the corrosive effects of the health crisis on businesses and individuals. All these salubrious repercussions of the pandemic are heartening and indicative of how a crisis can often bring out the best among us humans.
Continue to: Let's acknowledge the benefits...
Let’s acknowledge the benefits of the internet and the often-maligned social media. At a time of social isolation and cancellation of popular recreational activities (March Madness, NBA games, spring training baseball, movie theaters, concerts, religious congregations, partying with friends), the internet can offset the pain of mandated isolation by connecting all of us virtually, thus alleviating the emptiness that comes with isolation and boredom laced with anxiety. The damaging effects of a viral pandemic on human well-being would have been much worse if the internet did not exist.
Before the internet, television was a major escape, and for many it still is. But there is a downside: the wall-to-wall coverage of the local, national, and international effects of the pandemic can be alarming, and could increase distress even among persons who don’t have an anxiety disorder. Paradoxically, fear of going outdoors (agoraphobia) has suddenly become a necessary coping mechanism during a viral pandemic, instead of its traditional status as a “disabling symptom.”
Thank heavens for advances in technology. School children and college students can continue their education remotely without the risks of spreading infection by going to crowded classrooms. Scientific interactions and collaboration as well as business communications can remain active via videoconferencing technology, such as Zoom, Skype, or WebEx, without having to walk in crowded airports and fly to other cities on planes with recirculated air. Also, individuals who live far from family or friends can use their smartphones to see and chat with their loved ones. And cellphones remain a convenient method of staying in touch with the latest developments or making a “call to action” locally, national, and internationally.
During these oppressive and exceptional times, special attention and support must be provided to vulnerable populations, especially individuals with psychiatric illnesses, older adults who are physically infirm, and young children. Providing medical care, including psychiatric care, is essential to prevent the escalation of anxiety and panic among children and adults alike, and to prevent physical deterioration or death. This health crisis must be tackled with biopsychosocial approaches. And we, psychiatrists, must support and educate our patients and the public about stress management, and remind all about the transiency of epidemics as exemplified by the 1918 Spanish flu, the 1957 Asian flu, the 1968 Hong Kong flu, the 1982 human immunodeficiency virus, the 2002 severe acute respiratory syndrome virus, the 2009 Swine flu, the 2013 Ebola virus, and the 2016 Zika virus, all of which are now distant memories. The current COVID-19 pandemic should inoculate us to be more prepared and resilient for the inevitable future pandemics.
Fear of dying is considered “normal.” However, the ongoing threat of a potentially fatal viral infection can cause panic, anxiety, and an exaggerated fear of illness and death. The relentless spread of the coronavirus infectious disease that began in late 2019 (COVID-19) is spawning widespread anxiety, panic, and worry about one’s health and the health of loved ones. The viral pandemic has triggered a parallel anxiety epidemic.
Making things worse is that no vaccine has yet been developed, and for individuals who do get infected, there are no specific treatments other than supportive care, such as ventilators. Members of the public have been urged to practice sensible preventative measures, including handwashing, sanitizing certain items and surfaces, and—particularly challenging—self-isolation and social distancing. The public has channeled its fear into frantic buying and hoarding of food and non-food items, especially masks, sanitizers, soap, disinfectant wipes, and toilet paper (perhaps preparing for gastrointestinal hyperactivity during anxiety); canceling flights; avoiding group activities; and self-isolation or, for those exposed to the virus, quarantine. Anxiety is palpable. The facial masks that people wear are ironically unmasking their inner agitation and disquietude.
Our role as psychiatrists
As psychiatrists, we have an important role to play in such times, especially for our patients who already have anxiety disorders or depression. The additional emotional burden of this escalating health crisis is exacerbating the mental anguish of our patients (in addition to those who may soon become new patients). The anxiety and panic attacks due to “imagined” doom and gloom are now intensified by anxiety due to a “real” fatal threat. The effect on some vulnerable patients can be devastating, and may culminate in an acute stress reaction and future posttraumatic stress disorder. There are also reports of “psychogenic COVID-19” conversion reaction, with symptoms of sore throat, dyspnea, and even psychogenic fever. Paradoxically, self-isolation and social distancing, which are recommended to prevent the human-to-human spread of the virus, may further worsen anxiety and depression by reducing the comfort of intimacy and social contacts.
Individuals with depression will also experience an increased risk of symptom breakthrough despite receiving treatment. Stress is well known to trigger or exacerbate depression. Thus, the sense of helplessness and hopelessness during depression may intensify among our patients with pre-existing mood disorders, and suicidal ideation may resurface. Making things worse is the unfortunate timing of the COVID-19 pandemic. Spring is the peak season for the re-emergence of depression and suicide attempts. The ongoing stress of the health crisis, coupled with the onset of spring, may coalesce into a dreadful synergy for relapse among vulnerable individuals with unipolar or bipolar depression.
Patients with obsessive-compulsive disorder (OCD) are known to be averse to imagined germs and may wash their hands multiple times a day. An epidemic in which all health officials strongly urge washing one’s hands is very likely to exacerbate the compulsive handwashing of persons with OCD and significantly increase their anxiety. Because their other obsessions and compulsions may also increase in frequency and intensity, they will need our attention as their psychiatrists.
The viral pandemic is eerily similar to a natural disaster such as a hurricane of tornado, both of which physically destroy towns and flatten homes. The COVID-19 pandemic is damaging social structures and obliterating the fabric of global human relations. Consider the previously unimaginable disruption of what makes a vibrant society: schools, colleges, sporting events, concerts, Broadway shows, houses of worship, festivals, conferences, conventions, busy airports/train stations/bus stations, and spontaneous community gatherings. The sudden shock of upheaval in our daily lives may not only cause a hollow sense of emptiness and grief, but also have residual economic and emotional consequences. Nothing can be taken for granted anymore, and nothing is permanent. Cynicism may rise about maintaining life as we know it.
Rising to the challenge
Physicians and clinicians across all specialties are rising to the challenge of the pandemic, whether to manage the immediate physical or emotional impacts of the health crisis or its anticipated consequences (including the economic sequelae). The often-demonized pharmaceutical industry is urgently summoning all its resources to develop both a vaccine as well as biologic treatments for this potentially fatal viral infection. The government is removing regulatory barriers to expedite solutions to the crisis. A welcome public-private partnership is expediting the availability of and access to testing for the virus. The toxic political partisanship has temporarily given way to collaboration in crafting laws that can mitigate the corrosive effects of the health crisis on businesses and individuals. All these salubrious repercussions of the pandemic are heartening and indicative of how a crisis can often bring out the best among us humans.
Continue to: Let's acknowledge the benefits...
Let’s acknowledge the benefits of the internet and the often-maligned social media. At a time of social isolation and cancellation of popular recreational activities (March Madness, NBA games, spring training baseball, movie theaters, concerts, religious congregations, partying with friends), the internet can offset the pain of mandated isolation by connecting all of us virtually, thus alleviating the emptiness that comes with isolation and boredom laced with anxiety. The damaging effects of a viral pandemic on human well-being would have been much worse if the internet did not exist.
Before the internet, television was a major escape, and for many it still is. But there is a downside: the wall-to-wall coverage of the local, national, and international effects of the pandemic can be alarming, and could increase distress even among persons who don’t have an anxiety disorder. Paradoxically, fear of going outdoors (agoraphobia) has suddenly become a necessary coping mechanism during a viral pandemic, instead of its traditional status as a “disabling symptom.”
Thank heavens for advances in technology. School children and college students can continue their education remotely without the risks of spreading infection by going to crowded classrooms. Scientific interactions and collaboration as well as business communications can remain active via videoconferencing technology, such as Zoom, Skype, or WebEx, without having to walk in crowded airports and fly to other cities on planes with recirculated air. Also, individuals who live far from family or friends can use their smartphones to see and chat with their loved ones. And cellphones remain a convenient method of staying in touch with the latest developments or making a “call to action” locally, national, and internationally.
During these oppressive and exceptional times, special attention and support must be provided to vulnerable populations, especially individuals with psychiatric illnesses, older adults who are physically infirm, and young children. Providing medical care, including psychiatric care, is essential to prevent the escalation of anxiety and panic among children and adults alike, and to prevent physical deterioration or death. This health crisis must be tackled with biopsychosocial approaches. And we, psychiatrists, must support and educate our patients and the public about stress management, and remind all about the transiency of epidemics as exemplified by the 1918 Spanish flu, the 1957 Asian flu, the 1968 Hong Kong flu, the 1982 human immunodeficiency virus, the 2002 severe acute respiratory syndrome virus, the 2009 Swine flu, the 2013 Ebola virus, and the 2016 Zika virus, all of which are now distant memories. The current COVID-19 pandemic should inoculate us to be more prepared and resilient for the inevitable future pandemics.
Fear of dying is considered “normal.” However, the ongoing threat of a potentially fatal viral infection can cause panic, anxiety, and an exaggerated fear of illness and death. The relentless spread of the coronavirus infectious disease that began in late 2019 (COVID-19) is spawning widespread anxiety, panic, and worry about one’s health and the health of loved ones. The viral pandemic has triggered a parallel anxiety epidemic.
Making things worse is that no vaccine has yet been developed, and for individuals who do get infected, there are no specific treatments other than supportive care, such as ventilators. Members of the public have been urged to practice sensible preventative measures, including handwashing, sanitizing certain items and surfaces, and—particularly challenging—self-isolation and social distancing. The public has channeled its fear into frantic buying and hoarding of food and non-food items, especially masks, sanitizers, soap, disinfectant wipes, and toilet paper (perhaps preparing for gastrointestinal hyperactivity during anxiety); canceling flights; avoiding group activities; and self-isolation or, for those exposed to the virus, quarantine. Anxiety is palpable. The facial masks that people wear are ironically unmasking their inner agitation and disquietude.
Our role as psychiatrists
As psychiatrists, we have an important role to play in such times, especially for our patients who already have anxiety disorders or depression. The additional emotional burden of this escalating health crisis is exacerbating the mental anguish of our patients (in addition to those who may soon become new patients). The anxiety and panic attacks due to “imagined” doom and gloom are now intensified by anxiety due to a “real” fatal threat. The effect on some vulnerable patients can be devastating, and may culminate in an acute stress reaction and future posttraumatic stress disorder. There are also reports of “psychogenic COVID-19” conversion reaction, with symptoms of sore throat, dyspnea, and even psychogenic fever. Paradoxically, self-isolation and social distancing, which are recommended to prevent the human-to-human spread of the virus, may further worsen anxiety and depression by reducing the comfort of intimacy and social contacts.
Individuals with depression will also experience an increased risk of symptom breakthrough despite receiving treatment. Stress is well known to trigger or exacerbate depression. Thus, the sense of helplessness and hopelessness during depression may intensify among our patients with pre-existing mood disorders, and suicidal ideation may resurface. Making things worse is the unfortunate timing of the COVID-19 pandemic. Spring is the peak season for the re-emergence of depression and suicide attempts. The ongoing stress of the health crisis, coupled with the onset of spring, may coalesce into a dreadful synergy for relapse among vulnerable individuals with unipolar or bipolar depression.
Patients with obsessive-compulsive disorder (OCD) are known to be averse to imagined germs and may wash their hands multiple times a day. An epidemic in which all health officials strongly urge washing one’s hands is very likely to exacerbate the compulsive handwashing of persons with OCD and significantly increase their anxiety. Because their other obsessions and compulsions may also increase in frequency and intensity, they will need our attention as their psychiatrists.
The viral pandemic is eerily similar to a natural disaster such as a hurricane of tornado, both of which physically destroy towns and flatten homes. The COVID-19 pandemic is damaging social structures and obliterating the fabric of global human relations. Consider the previously unimaginable disruption of what makes a vibrant society: schools, colleges, sporting events, concerts, Broadway shows, houses of worship, festivals, conferences, conventions, busy airports/train stations/bus stations, and spontaneous community gatherings. The sudden shock of upheaval in our daily lives may not only cause a hollow sense of emptiness and grief, but also have residual economic and emotional consequences. Nothing can be taken for granted anymore, and nothing is permanent. Cynicism may rise about maintaining life as we know it.
Rising to the challenge
Physicians and clinicians across all specialties are rising to the challenge of the pandemic, whether to manage the immediate physical or emotional impacts of the health crisis or its anticipated consequences (including the economic sequelae). The often-demonized pharmaceutical industry is urgently summoning all its resources to develop both a vaccine as well as biologic treatments for this potentially fatal viral infection. The government is removing regulatory barriers to expedite solutions to the crisis. A welcome public-private partnership is expediting the availability of and access to testing for the virus. The toxic political partisanship has temporarily given way to collaboration in crafting laws that can mitigate the corrosive effects of the health crisis on businesses and individuals. All these salubrious repercussions of the pandemic are heartening and indicative of how a crisis can often bring out the best among us humans.
Continue to: Let's acknowledge the benefits...
Let’s acknowledge the benefits of the internet and the often-maligned social media. At a time of social isolation and cancellation of popular recreational activities (March Madness, NBA games, spring training baseball, movie theaters, concerts, religious congregations, partying with friends), the internet can offset the pain of mandated isolation by connecting all of us virtually, thus alleviating the emptiness that comes with isolation and boredom laced with anxiety. The damaging effects of a viral pandemic on human well-being would have been much worse if the internet did not exist.
Before the internet, television was a major escape, and for many it still is. But there is a downside: the wall-to-wall coverage of the local, national, and international effects of the pandemic can be alarming, and could increase distress even among persons who don’t have an anxiety disorder. Paradoxically, fear of going outdoors (agoraphobia) has suddenly become a necessary coping mechanism during a viral pandemic, instead of its traditional status as a “disabling symptom.”
Thank heavens for advances in technology. School children and college students can continue their education remotely without the risks of spreading infection by going to crowded classrooms. Scientific interactions and collaboration as well as business communications can remain active via videoconferencing technology, such as Zoom, Skype, or WebEx, without having to walk in crowded airports and fly to other cities on planes with recirculated air. Also, individuals who live far from family or friends can use their smartphones to see and chat with their loved ones. And cellphones remain a convenient method of staying in touch with the latest developments or making a “call to action” locally, national, and internationally.
During these oppressive and exceptional times, special attention and support must be provided to vulnerable populations, especially individuals with psychiatric illnesses, older adults who are physically infirm, and young children. Providing medical care, including psychiatric care, is essential to prevent the escalation of anxiety and panic among children and adults alike, and to prevent physical deterioration or death. This health crisis must be tackled with biopsychosocial approaches. And we, psychiatrists, must support and educate our patients and the public about stress management, and remind all about the transiency of epidemics as exemplified by the 1918 Spanish flu, the 1957 Asian flu, the 1968 Hong Kong flu, the 1982 human immunodeficiency virus, the 2002 severe acute respiratory syndrome virus, the 2009 Swine flu, the 2013 Ebola virus, and the 2016 Zika virus, all of which are now distant memories. The current COVID-19 pandemic should inoculate us to be more prepared and resilient for the inevitable future pandemics.
Clinical Progress Note: Perioperative Pain Control in Hospitalized Pediatric Patients
Pediatric hospitalists play an increasingly significant role in perioperative pain management.1 Advances in pediatric surgical comanagement may improve quality of care and reduce the length of hospitalization.2 This review is based on queries of the PubMed and Cochrane databases between January 1, 2014, and July 15, 2019, using the search terms “perioperative pain management,” “postoperative pain,” “pediatric,” and “children.” In addition, the authors reviewed key position statements from the American Academy of Pediatrics (AAP), the American Pain Society (APS), the Centers for Disease Control and Prevention (CDC), and the Society of Hospital Medicine (SHM) regarding pain management.3 This update is intended to be relevant for practicing pediatric hospitalists, with a focus on recently expanded options for pain management and judicious opioid use in hospitalized children.
PERIOPERATIVE PAIN MANAGEMENT
Postoperative pain management begins preoperatively according to the concept of the perioperative surgical home (PSH).4 The preoperative history should identify the patient’s previous positive (eg, good pain control) and negative (eg, adverse reactions) experiences with pain medications. Family and patient expectations should be discussed regarding types and sources of pain, pain duration, exacerbating/alleviating factors, and modalities available for realistic pain control because preoperative information can limit anxiety and improve outcomes. Pain specialists can perform risk assessments preoperatively and develop plans to address pharmacologic tolerance, withdrawal, and opioid-induced hyperalgesia after surgery.5 Children with chronic pain and on preoperative opioids may require more analgesia for a longer duration postoperatively. Early recognition of variability of patient’s pain perception and differences in responses to pain need to be clearly communicated across the disciplines in a collaborative model of care.
Children with medical complexity and/or cognitive, emotional, or behavioral impairments may benefit from preoperative psychosocial treatments and utilization of pain self-management training and strategies that could further reduce anxiety and optimize postoperative care because patient and parental preoperative anxiety may be associated with adverse outcomes. Validated pain assessment tools like Revised FLACC (Face, Leg, Activity, Cry, and Consolability) Scale and Individualized Numeric Rating Scale could be particularly useful in children with limitations in communication or altered pain perception; therefore, medical teams and family members should discuss their utilization preoperatively.
MULTIMODAL ANALGESIA
Multimodal analgesia (MMA) is a strategy that synergistically uses pharmacologic and nonpharmacologic modalities to target pain at multiple points of the pain processing pathway (Table).6 MMA can optimize pain control by addressing different types of pain (eg, incisional pain, muscle spasm, or neuropathic pain), expedite recovery, reduce potential pharmacologic side effects, and decrease opioid consumption. Patients taking opioids are at an increased risk of developing opioid-related side effects such as respiratory depression, medication tolerance, and anxiety, with resultant longer hospital stay, increased readmissions, and higher costs of care.7 Treatment for postoperative pain should prioritize appropriately dosed and precisely scheduled MMA before opioid-focused analgesia with the goals of decreasing opioid-related adverse effects, intentional misuse, diversion, and accidental ingestions. The AAP, APS, CDC, and SHM endorse the use of MMA and recommend nonpharmacologic measures and regional anesthesia.8,9 The most used modalities in MMA are discussed below.
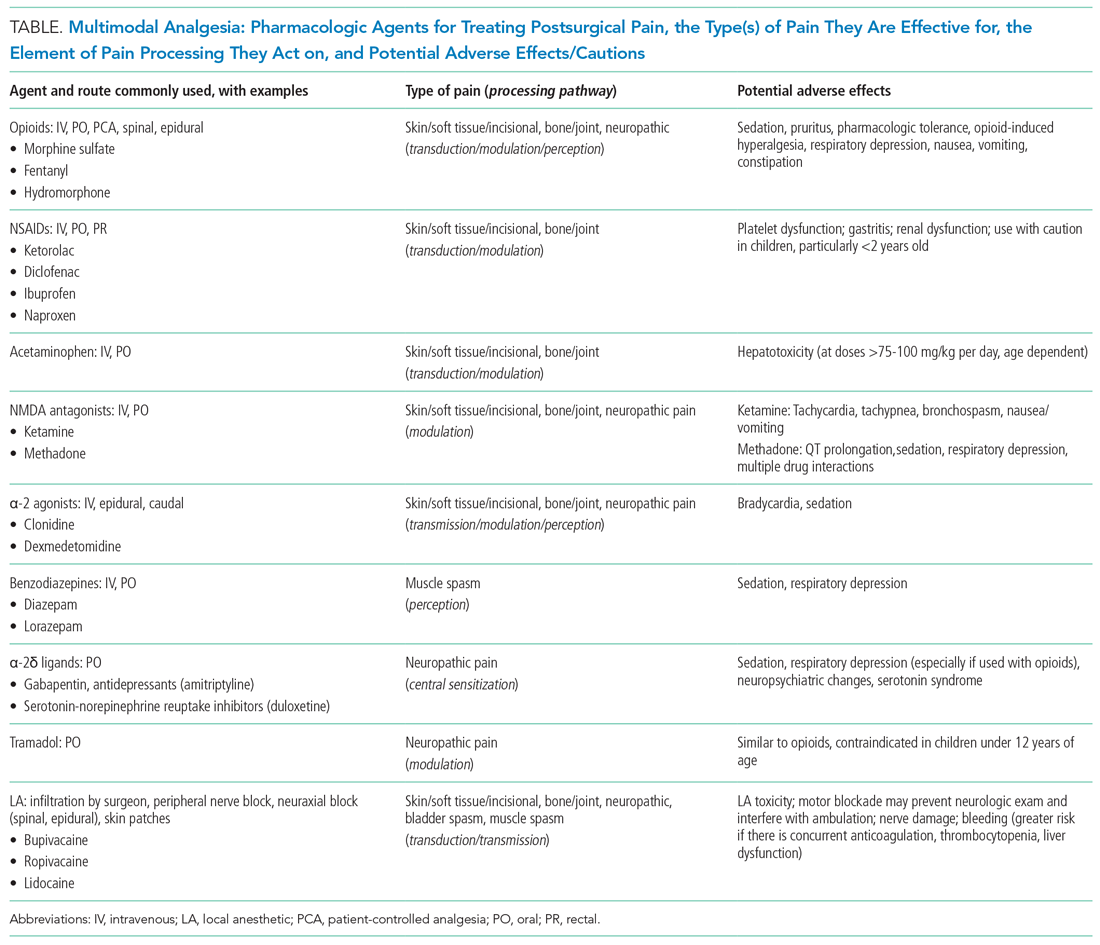
Acetaminophen
Acetaminophen has central-acting analgesic and antipyretic properties and readily crosses the blood brain barrier, which makes it particularly useful in spine and neurological surgeries. Oral administration is preferred when feasible. The AAP recommends refraining from rectal administration of acetaminophen as analgesia in children because of concerns about toxic effects and erratic, variable absorption.10 A systematic review of six studies found no benefit in pain control between intravenous (IV) and oral (PO) administration of acetaminophen in adults.11 There is a paucity of studies in children comparing PO with IV acetaminophen perioperative efficacy. Children may benefit from IV formulations in the early postoperative period, in cases with frequent nausea and vomiting, and in those with oral medication intolerance. Since infants have greater risk of respiratory depression from opioids, IV acetaminophen may have utility in this age group. Because of the cost associated with IV formulation, some institutions restrict IV acetaminophen. However, rapidly well-controlled pain and minimization of opioid-related side effects with shorter hospital stays may lower healthcare costs despite the cost of acetaminophen itself.
NSAIDs
NSAIDs possess anti-inflammatory properties through the inhibition of cyclooxygenase and blockade of prostaglandin production. NSAID risks include bleeding, renal and gastrointestinal toxicities, and potentially delayed wound and bone healing. Ketorolac is an NSAID that continues to be widely used with demonstrated opioid-sparing effects. Many retrospective studies including large numbers of pediatric patients have not demonstrated increased risks of bleeding nor poor wound healing with short postoperative use. A Cochrane review, however, concluded that there is insufficient data to either support or reject the efficacy or safety of ketorolac for postoperative pain treatment in children, mostly because of the very low quality of evidence.12
Regional Anesthesia
Regional anesthesia, which includes central (spinal/epidural/caudal) and peripheral blocks, decreases postoperative pain and opioid-associated side effects. Blocks typically consist of local anesthetic with or without the addition of adjuncts (eg, clonidine, dexamethasone). Regional anesthesia may also improve pulmonary function, compared with that of nonregional MMA use, in patients who have thoracic or upper abdominal surgeries. While having broad applications, the utility of regional anesthesia is greatest in preterm infants/neonates and in those with underlying respiratory pathology. A systematic review of randomized controlled trials demonstrated that regional anesthesia decreased opioid consumption and minimized postoperative pain with no significant complications attributed to its use.13 Additional studies are needed to better delineate specific surgical procedures and subpopulations of pediatric patients in which regional anesthesia may provide the most benefit.
Gabapentinoids
Children receiving gabapentinoids perioperatively have been shown to have fewer adverse reactions, decreased opioid consumption, and less anxiety, as well as improved pain scores. Gabapentin is increasingly being utilized for children with idiopathic scoliosis undergoing posterior spinal fusion, and there is some evidence for improving pain control and reducing opioid use. However, a recent systematic review found a paucity of data supporting its clinical use.14 Both gabapentin and pregabalin may further increase risks of respiratory depression, especially in synergy with opioids and benzodiazepines.
Opioids
Opioids should be used with caution in pediatric patients and are reserved primarily for the management of severe acute pain. The shortest duration of the lowest effective dose of opioids should be encouraged. Patient-controlled opioid analgesia (PCA) offers benefits when parenteral postoperative analgesia is indicated: It maximizes pain relief, minimizes risk of overdose, and improves psychological well-being through self-administration of pain medicines. Basal-infusion PCA should not be routinely used because it is associated with nausea, vomiting, and respiratory depression without having superior analgesia compared with demand use only. Monitoring of side stream end-tidal capnography can readily detect respiratory depression, especially if opioids, benzodiazepines, gabapentinoids, and diphenhydramine are used concomitantly. Patient education regarding opioid use, side effects, safe storage, and disposal practices is imperative because significant amounts of opioids remain in households after completion of treatment for pain and because opioid diversion and accidental ingestions account for significant morbidity. Providers need to balance efficient pain management with opioid stewardship, complying with state and federal policies to limit harm related to opioid diversion.15
Nonpharmacological Modalities
The use of nonpharmacologic therapies, along with pharmacologic modalities, for perioperative pain management has been shown to decrease opioid use and opioid-related side effects. Trials of acupressure have demonstrated improvement in nausea and vomiting, sleep quality, and pain and anxiety scores. Nonpharmacologic treatments currently serve as a complementary approach for pain and anxiety management in the perioperative setting including acupuncture, acupressure, osteopathic manipulative treatment, massage, meditation, biofeedback, hypnotherapy, and physical/occupational, relaxation, cognitive-behavioral, chiropractic, music, and art therapies. The Joint Commission suggests consideration of such modalities by hospitals.
FUTURE CONSIDERATIONS
Pediatric hospitalists have been traditionally involved in research and patient care improvements and should continue to actively contribute to establishing evidence-based guidelines for the treatment of acute postoperative pain in hospitalized children and adolescents. The sparsity of high-quality evidence prompts the need for more research. A standardized approach to perioperative pain management in the form of checklists, pathways, and protocols for specific procedures may be useful to educate providers and patients, while also standardizing available evidence-based interventions (eg, pediatric Enhanced Recovery After Surgery [ERAS] protocols).
CONCLUSION
Combining multimodal pharmacologic and integrative nonpharmacologic modalities can decrease opioid use and related side effects and improve the perioperative care of hospitalized children. Pediatric hospitalists have an opportunity to optimize care preoperatively, practice multimodal analgesia, and contribute to reducing risk of opioid diversion post operatively.
1. Society of Hospital Medicine Co-Management Advisory Panel. A white paper on a guide to hospitalist/orthopedic surgery co-management. http://tools.hospitalmedicine.org/Implementation/Co-ManagementWhitePaper-final_5-10-10.pdf. Accessed October 11, 2019.
2. Rappaport DI, Rosenberg RE, Shaughnessy EE, et al. Pediatric hospitalist comanagement of surgical patients: Structural, quality, and financial considerations. J Hosp Med. 2014;9(11):737-742. https://doi.org/10.1002/jhm.2266.
3. Evidence-Based Nonpharmacologic Strategies for Comprehensive Pain Care: The Consortium Pain Task Force White Paper. http://www.nonpharmpaincare.org. Accessed on October 11, 2019.
4. Vetter TR, Kain ZN. Role of perioperative surgical home in optimizing the perioperative use of opioids. Anesth Analg. 2017;125(5):1653-1657. https://doi.org/10.1213/ANE.0000000000002280.
5. Edwards DA, Hedrick TL, Jayaram J, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on perioperative management of patients on preoperative opioid therapy. Anesth Analg. 2019;129(2):553-566. http://doi.org/10.1213/ANE.0000000000004018.
6. Micromedex (electronic version). IBM Watson Health. Greenwood Village, Colorado, USA. https://www.micromedexsolutions.com. Accessed October 10, 2019.
7. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: A clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131-157. https://doi.org/10.1016/j.jpain.2015.12.008.
8. Herzig SJ, Mosher HJ, Calcaterra SL, Jena AB, Nuckols TK. Improving the safety of opioid use for acute noncancer pain in hospitalized adults: A consensus statement from the Society of Hospital Medicine. J Hosp Med. 2018;3(4):263-266. https://doi.org/10.12788/jhm.2980.
9. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain – United States, 2016. JAMA. 2016;315(15):1624-1645. https://doi.org/10.1001/jama.2016.1464.
10. American Academy of Pediatrics Committee on Drugs. Acetaminophen toxicity in children. Pediatrics. 2001;108 (4):1020-1024. https://doi.org/10.1542/peds.108.4.1020.
11. Jibril F, Sharaby S, Mohamed A, Wilby, KJ. Intravenous versus oral acetaminophen for pain: Systemic review of current evidence to support clinical decision-making. Can J Hosp Pharm. 2015;68(3):238-247. https://doi.org/10.4212/cjhp.v68i3.1458.
12. McNicol ED, Rowe E, Cooper TE. Ketorolac for postoperative pain in children. Cochrane Database Syst Rev. 2018;7(7). https://doi.org/10.1002/14651858.CD012294.pub2.
13. Kendall MC, Castro Alves LJ, Suh EI, McCormick ZL, De Oliveira GS. Regional anesthesia to ameliorate postoperative analgesia outcomes in pediatric surgical patients: an updated systematic review of randomized controlled trials. Local Reg Anesth. 2018;11:91-109. https://doi.org/10.2147/LRA.S185554.
14. Egunsola 0, Wylie CE, Chitty KM, et al. Systematic review of the efficacy and safety of gabapentin and pregabalin for pain in children and adolescents. Anesth Analg. 2019;128(4):811-819. https://doi.org/10.1213/ANE.0000000000003936.
15. Harbaugh C, Gadepalli SK. Pediatric postoperative opioid prescribing and the opioid crisis. Curr Opin Pediatr. 2019;31(3):377-385. https://doi.org/10.1097/MOP.0000000000000768.
Pediatric hospitalists play an increasingly significant role in perioperative pain management.1 Advances in pediatric surgical comanagement may improve quality of care and reduce the length of hospitalization.2 This review is based on queries of the PubMed and Cochrane databases between January 1, 2014, and July 15, 2019, using the search terms “perioperative pain management,” “postoperative pain,” “pediatric,” and “children.” In addition, the authors reviewed key position statements from the American Academy of Pediatrics (AAP), the American Pain Society (APS), the Centers for Disease Control and Prevention (CDC), and the Society of Hospital Medicine (SHM) regarding pain management.3 This update is intended to be relevant for practicing pediatric hospitalists, with a focus on recently expanded options for pain management and judicious opioid use in hospitalized children.
PERIOPERATIVE PAIN MANAGEMENT
Postoperative pain management begins preoperatively according to the concept of the perioperative surgical home (PSH).4 The preoperative history should identify the patient’s previous positive (eg, good pain control) and negative (eg, adverse reactions) experiences with pain medications. Family and patient expectations should be discussed regarding types and sources of pain, pain duration, exacerbating/alleviating factors, and modalities available for realistic pain control because preoperative information can limit anxiety and improve outcomes. Pain specialists can perform risk assessments preoperatively and develop plans to address pharmacologic tolerance, withdrawal, and opioid-induced hyperalgesia after surgery.5 Children with chronic pain and on preoperative opioids may require more analgesia for a longer duration postoperatively. Early recognition of variability of patient’s pain perception and differences in responses to pain need to be clearly communicated across the disciplines in a collaborative model of care.
Children with medical complexity and/or cognitive, emotional, or behavioral impairments may benefit from preoperative psychosocial treatments and utilization of pain self-management training and strategies that could further reduce anxiety and optimize postoperative care because patient and parental preoperative anxiety may be associated with adverse outcomes. Validated pain assessment tools like Revised FLACC (Face, Leg, Activity, Cry, and Consolability) Scale and Individualized Numeric Rating Scale could be particularly useful in children with limitations in communication or altered pain perception; therefore, medical teams and family members should discuss their utilization preoperatively.
MULTIMODAL ANALGESIA
Multimodal analgesia (MMA) is a strategy that synergistically uses pharmacologic and nonpharmacologic modalities to target pain at multiple points of the pain processing pathway (Table).6 MMA can optimize pain control by addressing different types of pain (eg, incisional pain, muscle spasm, or neuropathic pain), expedite recovery, reduce potential pharmacologic side effects, and decrease opioid consumption. Patients taking opioids are at an increased risk of developing opioid-related side effects such as respiratory depression, medication tolerance, and anxiety, with resultant longer hospital stay, increased readmissions, and higher costs of care.7 Treatment for postoperative pain should prioritize appropriately dosed and precisely scheduled MMA before opioid-focused analgesia with the goals of decreasing opioid-related adverse effects, intentional misuse, diversion, and accidental ingestions. The AAP, APS, CDC, and SHM endorse the use of MMA and recommend nonpharmacologic measures and regional anesthesia.8,9 The most used modalities in MMA are discussed below.

Acetaminophen
Acetaminophen has central-acting analgesic and antipyretic properties and readily crosses the blood brain barrier, which makes it particularly useful in spine and neurological surgeries. Oral administration is preferred when feasible. The AAP recommends refraining from rectal administration of acetaminophen as analgesia in children because of concerns about toxic effects and erratic, variable absorption.10 A systematic review of six studies found no benefit in pain control between intravenous (IV) and oral (PO) administration of acetaminophen in adults.11 There is a paucity of studies in children comparing PO with IV acetaminophen perioperative efficacy. Children may benefit from IV formulations in the early postoperative period, in cases with frequent nausea and vomiting, and in those with oral medication intolerance. Since infants have greater risk of respiratory depression from opioids, IV acetaminophen may have utility in this age group. Because of the cost associated with IV formulation, some institutions restrict IV acetaminophen. However, rapidly well-controlled pain and minimization of opioid-related side effects with shorter hospital stays may lower healthcare costs despite the cost of acetaminophen itself.
NSAIDs
NSAIDs possess anti-inflammatory properties through the inhibition of cyclooxygenase and blockade of prostaglandin production. NSAID risks include bleeding, renal and gastrointestinal toxicities, and potentially delayed wound and bone healing. Ketorolac is an NSAID that continues to be widely used with demonstrated opioid-sparing effects. Many retrospective studies including large numbers of pediatric patients have not demonstrated increased risks of bleeding nor poor wound healing with short postoperative use. A Cochrane review, however, concluded that there is insufficient data to either support or reject the efficacy or safety of ketorolac for postoperative pain treatment in children, mostly because of the very low quality of evidence.12
Regional Anesthesia
Regional anesthesia, which includes central (spinal/epidural/caudal) and peripheral blocks, decreases postoperative pain and opioid-associated side effects. Blocks typically consist of local anesthetic with or without the addition of adjuncts (eg, clonidine, dexamethasone). Regional anesthesia may also improve pulmonary function, compared with that of nonregional MMA use, in patients who have thoracic or upper abdominal surgeries. While having broad applications, the utility of regional anesthesia is greatest in preterm infants/neonates and in those with underlying respiratory pathology. A systematic review of randomized controlled trials demonstrated that regional anesthesia decreased opioid consumption and minimized postoperative pain with no significant complications attributed to its use.13 Additional studies are needed to better delineate specific surgical procedures and subpopulations of pediatric patients in which regional anesthesia may provide the most benefit.
Gabapentinoids
Children receiving gabapentinoids perioperatively have been shown to have fewer adverse reactions, decreased opioid consumption, and less anxiety, as well as improved pain scores. Gabapentin is increasingly being utilized for children with idiopathic scoliosis undergoing posterior spinal fusion, and there is some evidence for improving pain control and reducing opioid use. However, a recent systematic review found a paucity of data supporting its clinical use.14 Both gabapentin and pregabalin may further increase risks of respiratory depression, especially in synergy with opioids and benzodiazepines.
Opioids
Opioids should be used with caution in pediatric patients and are reserved primarily for the management of severe acute pain. The shortest duration of the lowest effective dose of opioids should be encouraged. Patient-controlled opioid analgesia (PCA) offers benefits when parenteral postoperative analgesia is indicated: It maximizes pain relief, minimizes risk of overdose, and improves psychological well-being through self-administration of pain medicines. Basal-infusion PCA should not be routinely used because it is associated with nausea, vomiting, and respiratory depression without having superior analgesia compared with demand use only. Monitoring of side stream end-tidal capnography can readily detect respiratory depression, especially if opioids, benzodiazepines, gabapentinoids, and diphenhydramine are used concomitantly. Patient education regarding opioid use, side effects, safe storage, and disposal practices is imperative because significant amounts of opioids remain in households after completion of treatment for pain and because opioid diversion and accidental ingestions account for significant morbidity. Providers need to balance efficient pain management with opioid stewardship, complying with state and federal policies to limit harm related to opioid diversion.15
Nonpharmacological Modalities
The use of nonpharmacologic therapies, along with pharmacologic modalities, for perioperative pain management has been shown to decrease opioid use and opioid-related side effects. Trials of acupressure have demonstrated improvement in nausea and vomiting, sleep quality, and pain and anxiety scores. Nonpharmacologic treatments currently serve as a complementary approach for pain and anxiety management in the perioperative setting including acupuncture, acupressure, osteopathic manipulative treatment, massage, meditation, biofeedback, hypnotherapy, and physical/occupational, relaxation, cognitive-behavioral, chiropractic, music, and art therapies. The Joint Commission suggests consideration of such modalities by hospitals.
FUTURE CONSIDERATIONS
Pediatric hospitalists have been traditionally involved in research and patient care improvements and should continue to actively contribute to establishing evidence-based guidelines for the treatment of acute postoperative pain in hospitalized children and adolescents. The sparsity of high-quality evidence prompts the need for more research. A standardized approach to perioperative pain management in the form of checklists, pathways, and protocols for specific procedures may be useful to educate providers and patients, while also standardizing available evidence-based interventions (eg, pediatric Enhanced Recovery After Surgery [ERAS] protocols).
CONCLUSION
Combining multimodal pharmacologic and integrative nonpharmacologic modalities can decrease opioid use and related side effects and improve the perioperative care of hospitalized children. Pediatric hospitalists have an opportunity to optimize care preoperatively, practice multimodal analgesia, and contribute to reducing risk of opioid diversion post operatively.
Pediatric hospitalists play an increasingly significant role in perioperative pain management.1 Advances in pediatric surgical comanagement may improve quality of care and reduce the length of hospitalization.2 This review is based on queries of the PubMed and Cochrane databases between January 1, 2014, and July 15, 2019, using the search terms “perioperative pain management,” “postoperative pain,” “pediatric,” and “children.” In addition, the authors reviewed key position statements from the American Academy of Pediatrics (AAP), the American Pain Society (APS), the Centers for Disease Control and Prevention (CDC), and the Society of Hospital Medicine (SHM) regarding pain management.3 This update is intended to be relevant for practicing pediatric hospitalists, with a focus on recently expanded options for pain management and judicious opioid use in hospitalized children.
PERIOPERATIVE PAIN MANAGEMENT
Postoperative pain management begins preoperatively according to the concept of the perioperative surgical home (PSH).4 The preoperative history should identify the patient’s previous positive (eg, good pain control) and negative (eg, adverse reactions) experiences with pain medications. Family and patient expectations should be discussed regarding types and sources of pain, pain duration, exacerbating/alleviating factors, and modalities available for realistic pain control because preoperative information can limit anxiety and improve outcomes. Pain specialists can perform risk assessments preoperatively and develop plans to address pharmacologic tolerance, withdrawal, and opioid-induced hyperalgesia after surgery.5 Children with chronic pain and on preoperative opioids may require more analgesia for a longer duration postoperatively. Early recognition of variability of patient’s pain perception and differences in responses to pain need to be clearly communicated across the disciplines in a collaborative model of care.
Children with medical complexity and/or cognitive, emotional, or behavioral impairments may benefit from preoperative psychosocial treatments and utilization of pain self-management training and strategies that could further reduce anxiety and optimize postoperative care because patient and parental preoperative anxiety may be associated with adverse outcomes. Validated pain assessment tools like Revised FLACC (Face, Leg, Activity, Cry, and Consolability) Scale and Individualized Numeric Rating Scale could be particularly useful in children with limitations in communication or altered pain perception; therefore, medical teams and family members should discuss their utilization preoperatively.
MULTIMODAL ANALGESIA
Multimodal analgesia (MMA) is a strategy that synergistically uses pharmacologic and nonpharmacologic modalities to target pain at multiple points of the pain processing pathway (Table).6 MMA can optimize pain control by addressing different types of pain (eg, incisional pain, muscle spasm, or neuropathic pain), expedite recovery, reduce potential pharmacologic side effects, and decrease opioid consumption. Patients taking opioids are at an increased risk of developing opioid-related side effects such as respiratory depression, medication tolerance, and anxiety, with resultant longer hospital stay, increased readmissions, and higher costs of care.7 Treatment for postoperative pain should prioritize appropriately dosed and precisely scheduled MMA before opioid-focused analgesia with the goals of decreasing opioid-related adverse effects, intentional misuse, diversion, and accidental ingestions. The AAP, APS, CDC, and SHM endorse the use of MMA and recommend nonpharmacologic measures and regional anesthesia.8,9 The most used modalities in MMA are discussed below.

Acetaminophen
Acetaminophen has central-acting analgesic and antipyretic properties and readily crosses the blood brain barrier, which makes it particularly useful in spine and neurological surgeries. Oral administration is preferred when feasible. The AAP recommends refraining from rectal administration of acetaminophen as analgesia in children because of concerns about toxic effects and erratic, variable absorption.10 A systematic review of six studies found no benefit in pain control between intravenous (IV) and oral (PO) administration of acetaminophen in adults.11 There is a paucity of studies in children comparing PO with IV acetaminophen perioperative efficacy. Children may benefit from IV formulations in the early postoperative period, in cases with frequent nausea and vomiting, and in those with oral medication intolerance. Since infants have greater risk of respiratory depression from opioids, IV acetaminophen may have utility in this age group. Because of the cost associated with IV formulation, some institutions restrict IV acetaminophen. However, rapidly well-controlled pain and minimization of opioid-related side effects with shorter hospital stays may lower healthcare costs despite the cost of acetaminophen itself.
NSAIDs
NSAIDs possess anti-inflammatory properties through the inhibition of cyclooxygenase and blockade of prostaglandin production. NSAID risks include bleeding, renal and gastrointestinal toxicities, and potentially delayed wound and bone healing. Ketorolac is an NSAID that continues to be widely used with demonstrated opioid-sparing effects. Many retrospective studies including large numbers of pediatric patients have not demonstrated increased risks of bleeding nor poor wound healing with short postoperative use. A Cochrane review, however, concluded that there is insufficient data to either support or reject the efficacy or safety of ketorolac for postoperative pain treatment in children, mostly because of the very low quality of evidence.12
Regional Anesthesia
Regional anesthesia, which includes central (spinal/epidural/caudal) and peripheral blocks, decreases postoperative pain and opioid-associated side effects. Blocks typically consist of local anesthetic with or without the addition of adjuncts (eg, clonidine, dexamethasone). Regional anesthesia may also improve pulmonary function, compared with that of nonregional MMA use, in patients who have thoracic or upper abdominal surgeries. While having broad applications, the utility of regional anesthesia is greatest in preterm infants/neonates and in those with underlying respiratory pathology. A systematic review of randomized controlled trials demonstrated that regional anesthesia decreased opioid consumption and minimized postoperative pain with no significant complications attributed to its use.13 Additional studies are needed to better delineate specific surgical procedures and subpopulations of pediatric patients in which regional anesthesia may provide the most benefit.
Gabapentinoids
Children receiving gabapentinoids perioperatively have been shown to have fewer adverse reactions, decreased opioid consumption, and less anxiety, as well as improved pain scores. Gabapentin is increasingly being utilized for children with idiopathic scoliosis undergoing posterior spinal fusion, and there is some evidence for improving pain control and reducing opioid use. However, a recent systematic review found a paucity of data supporting its clinical use.14 Both gabapentin and pregabalin may further increase risks of respiratory depression, especially in synergy with opioids and benzodiazepines.
Opioids
Opioids should be used with caution in pediatric patients and are reserved primarily for the management of severe acute pain. The shortest duration of the lowest effective dose of opioids should be encouraged. Patient-controlled opioid analgesia (PCA) offers benefits when parenteral postoperative analgesia is indicated: It maximizes pain relief, minimizes risk of overdose, and improves psychological well-being through self-administration of pain medicines. Basal-infusion PCA should not be routinely used because it is associated with nausea, vomiting, and respiratory depression without having superior analgesia compared with demand use only. Monitoring of side stream end-tidal capnography can readily detect respiratory depression, especially if opioids, benzodiazepines, gabapentinoids, and diphenhydramine are used concomitantly. Patient education regarding opioid use, side effects, safe storage, and disposal practices is imperative because significant amounts of opioids remain in households after completion of treatment for pain and because opioid diversion and accidental ingestions account for significant morbidity. Providers need to balance efficient pain management with opioid stewardship, complying with state and federal policies to limit harm related to opioid diversion.15
Nonpharmacological Modalities
The use of nonpharmacologic therapies, along with pharmacologic modalities, for perioperative pain management has been shown to decrease opioid use and opioid-related side effects. Trials of acupressure have demonstrated improvement in nausea and vomiting, sleep quality, and pain and anxiety scores. Nonpharmacologic treatments currently serve as a complementary approach for pain and anxiety management in the perioperative setting including acupuncture, acupressure, osteopathic manipulative treatment, massage, meditation, biofeedback, hypnotherapy, and physical/occupational, relaxation, cognitive-behavioral, chiropractic, music, and art therapies. The Joint Commission suggests consideration of such modalities by hospitals.
FUTURE CONSIDERATIONS
Pediatric hospitalists have been traditionally involved in research and patient care improvements and should continue to actively contribute to establishing evidence-based guidelines for the treatment of acute postoperative pain in hospitalized children and adolescents. The sparsity of high-quality evidence prompts the need for more research. A standardized approach to perioperative pain management in the form of checklists, pathways, and protocols for specific procedures may be useful to educate providers and patients, while also standardizing available evidence-based interventions (eg, pediatric Enhanced Recovery After Surgery [ERAS] protocols).
CONCLUSION
Combining multimodal pharmacologic and integrative nonpharmacologic modalities can decrease opioid use and related side effects and improve the perioperative care of hospitalized children. Pediatric hospitalists have an opportunity to optimize care preoperatively, practice multimodal analgesia, and contribute to reducing risk of opioid diversion post operatively.
1. Society of Hospital Medicine Co-Management Advisory Panel. A white paper on a guide to hospitalist/orthopedic surgery co-management. http://tools.hospitalmedicine.org/Implementation/Co-ManagementWhitePaper-final_5-10-10.pdf. Accessed October 11, 2019.
2. Rappaport DI, Rosenberg RE, Shaughnessy EE, et al. Pediatric hospitalist comanagement of surgical patients: Structural, quality, and financial considerations. J Hosp Med. 2014;9(11):737-742. https://doi.org/10.1002/jhm.2266.
3. Evidence-Based Nonpharmacologic Strategies for Comprehensive Pain Care: The Consortium Pain Task Force White Paper. http://www.nonpharmpaincare.org. Accessed on October 11, 2019.
4. Vetter TR, Kain ZN. Role of perioperative surgical home in optimizing the perioperative use of opioids. Anesth Analg. 2017;125(5):1653-1657. https://doi.org/10.1213/ANE.0000000000002280.
5. Edwards DA, Hedrick TL, Jayaram J, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on perioperative management of patients on preoperative opioid therapy. Anesth Analg. 2019;129(2):553-566. http://doi.org/10.1213/ANE.0000000000004018.
6. Micromedex (electronic version). IBM Watson Health. Greenwood Village, Colorado, USA. https://www.micromedexsolutions.com. Accessed October 10, 2019.
7. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: A clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131-157. https://doi.org/10.1016/j.jpain.2015.12.008.
8. Herzig SJ, Mosher HJ, Calcaterra SL, Jena AB, Nuckols TK. Improving the safety of opioid use for acute noncancer pain in hospitalized adults: A consensus statement from the Society of Hospital Medicine. J Hosp Med. 2018;3(4):263-266. https://doi.org/10.12788/jhm.2980.
9. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain – United States, 2016. JAMA. 2016;315(15):1624-1645. https://doi.org/10.1001/jama.2016.1464.
10. American Academy of Pediatrics Committee on Drugs. Acetaminophen toxicity in children. Pediatrics. 2001;108 (4):1020-1024. https://doi.org/10.1542/peds.108.4.1020.
11. Jibril F, Sharaby S, Mohamed A, Wilby, KJ. Intravenous versus oral acetaminophen for pain: Systemic review of current evidence to support clinical decision-making. Can J Hosp Pharm. 2015;68(3):238-247. https://doi.org/10.4212/cjhp.v68i3.1458.
12. McNicol ED, Rowe E, Cooper TE. Ketorolac for postoperative pain in children. Cochrane Database Syst Rev. 2018;7(7). https://doi.org/10.1002/14651858.CD012294.pub2.
13. Kendall MC, Castro Alves LJ, Suh EI, McCormick ZL, De Oliveira GS. Regional anesthesia to ameliorate postoperative analgesia outcomes in pediatric surgical patients: an updated systematic review of randomized controlled trials. Local Reg Anesth. 2018;11:91-109. https://doi.org/10.2147/LRA.S185554.
14. Egunsola 0, Wylie CE, Chitty KM, et al. Systematic review of the efficacy and safety of gabapentin and pregabalin for pain in children and adolescents. Anesth Analg. 2019;128(4):811-819. https://doi.org/10.1213/ANE.0000000000003936.
15. Harbaugh C, Gadepalli SK. Pediatric postoperative opioid prescribing and the opioid crisis. Curr Opin Pediatr. 2019;31(3):377-385. https://doi.org/10.1097/MOP.0000000000000768.
1. Society of Hospital Medicine Co-Management Advisory Panel. A white paper on a guide to hospitalist/orthopedic surgery co-management. http://tools.hospitalmedicine.org/Implementation/Co-ManagementWhitePaper-final_5-10-10.pdf. Accessed October 11, 2019.
2. Rappaport DI, Rosenberg RE, Shaughnessy EE, et al. Pediatric hospitalist comanagement of surgical patients: Structural, quality, and financial considerations. J Hosp Med. 2014;9(11):737-742. https://doi.org/10.1002/jhm.2266.
3. Evidence-Based Nonpharmacologic Strategies for Comprehensive Pain Care: The Consortium Pain Task Force White Paper. http://www.nonpharmpaincare.org. Accessed on October 11, 2019.
4. Vetter TR, Kain ZN. Role of perioperative surgical home in optimizing the perioperative use of opioids. Anesth Analg. 2017;125(5):1653-1657. https://doi.org/10.1213/ANE.0000000000002280.
5. Edwards DA, Hedrick TL, Jayaram J, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on perioperative management of patients on preoperative opioid therapy. Anesth Analg. 2019;129(2):553-566. http://doi.org/10.1213/ANE.0000000000004018.
6. Micromedex (electronic version). IBM Watson Health. Greenwood Village, Colorado, USA. https://www.micromedexsolutions.com. Accessed October 10, 2019.
7. Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of postoperative pain: A clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. 2016;17(2):131-157. https://doi.org/10.1016/j.jpain.2015.12.008.
8. Herzig SJ, Mosher HJ, Calcaterra SL, Jena AB, Nuckols TK. Improving the safety of opioid use for acute noncancer pain in hospitalized adults: A consensus statement from the Society of Hospital Medicine. J Hosp Med. 2018;3(4):263-266. https://doi.org/10.12788/jhm.2980.
9. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain – United States, 2016. JAMA. 2016;315(15):1624-1645. https://doi.org/10.1001/jama.2016.1464.
10. American Academy of Pediatrics Committee on Drugs. Acetaminophen toxicity in children. Pediatrics. 2001;108 (4):1020-1024. https://doi.org/10.1542/peds.108.4.1020.
11. Jibril F, Sharaby S, Mohamed A, Wilby, KJ. Intravenous versus oral acetaminophen for pain: Systemic review of current evidence to support clinical decision-making. Can J Hosp Pharm. 2015;68(3):238-247. https://doi.org/10.4212/cjhp.v68i3.1458.
12. McNicol ED, Rowe E, Cooper TE. Ketorolac for postoperative pain in children. Cochrane Database Syst Rev. 2018;7(7). https://doi.org/10.1002/14651858.CD012294.pub2.
13. Kendall MC, Castro Alves LJ, Suh EI, McCormick ZL, De Oliveira GS. Regional anesthesia to ameliorate postoperative analgesia outcomes in pediatric surgical patients: an updated systematic review of randomized controlled trials. Local Reg Anesth. 2018;11:91-109. https://doi.org/10.2147/LRA.S185554.
14. Egunsola 0, Wylie CE, Chitty KM, et al. Systematic review of the efficacy and safety of gabapentin and pregabalin for pain in children and adolescents. Anesth Analg. 2019;128(4):811-819. https://doi.org/10.1213/ANE.0000000000003936.
15. Harbaugh C, Gadepalli SK. Pediatric postoperative opioid prescribing and the opioid crisis. Curr Opin Pediatr. 2019;31(3):377-385. https://doi.org/10.1097/MOP.0000000000000768.
© 2020 Society of Hospital Medicine
Nationwide Hospital Performance on Publicly Reported Episode Spending Measures
Amid the continued shift from fee-for-service toward value-based payment, policymakers such as the Centers for Medicare & Medicaid Services have initiated strategies to contain spending on episodes of care. This episode focus has led to nationwide implementation of payment models such as bundled payments, which hold hospitals accountable for quality and costs across procedure-based (eg, coronary artery bypass surgery) and condition-based (eg, congestive heart failure) episodes, which begin with hospitalization and encompass subsequent hospital and postdischarge care.
Simultaneously, Medicare has increased its emphasis on similarly designed episodes of care (eg, those spanning hospitalization and postdischarge care) using other strategies, such as public reporting and use of episode-based measures to evaluate hospital cost performance. In 2017, Medicare trialed the implementation of six Clinical Episode-Based Payment (CEBP) measures in the national Hospital Inpatient Quality Reporting Program in order to assess hospital and clinician spending on procedure and condition episodes.1,2
CEBP measures reflect episode-specific spending, conveying “how expensive a hospital is” by capturing facility and professional payments for a given episode spanning between 3 days prior to hospitalization and 30 days following discharge. Given standard payment rates used in Medicare, the variation in episode spending reflects differences in quantity and type of services utilized within an episode. Medicare has specified episode-related services and designed CEBP measures via logic and definition rules informed by a combination of claims and procedures-based grouping, as well as by physician input. For example, the CEBP measure for cellulitis encompasses services related to diagnosing and treating the infection within the episode window, but not unrelated services such as eye exams for coexisting glaucoma. To increase clinical salience, CEBP measures are subdivided to reflect differing complexity when possible. For instance, cellulitis measures are divided into episodes with or without major complications or comorbidities and further subdivided into subtypes for episodes reflecting cellulitis in patients with diabetes, patients with decubitus ulcers, or neither.
CEBPs are similar to other spending measures used in payment programs, such as the Medicare Spending Per Beneficiary, but are more clinically relevant because their focus on episodes more closely reflects clinical practice. CEBPs and Medicare Spending Per Beneficiary have similar designs (eg, same episode windows) and purpose (eg, to capture the cost efficiency of hospital care).3 However, unlike CEBPs, Medicare Spending Per Beneficiary is a “global” measure that summarizes a hospital’s cost efficiency aggregated across all inpatient episodes rather than represent it based on specific conditions or procedures.4 The limitations of publicly reported global hospital measures—for instance, the poor correlation between hospital performance on distinct publicly reported quality measures5—highlight the potential utility of episode-specific spending measures such as CEBP.
Compared with episode-based payment models, initiatives such as CEBP measures have gone largely unstudied. However, they represent signals of Medicare’s growing commitment to addressing care episodes, tested without potentially tedious rulemaking required to change payment. In fact, publicly reported episode spending measures offer policymakers several interrelated benefits: the ability to rapidly evaluate performance at a large number of hospitals (eg, Medicare scaling up CEBP measures among all eligible hospitals nationwide), the option of leveraging publicly reported feedback to prompt clinical improvements (eg, by including CEBP measures in the Hospital Inpatient Quality Reporting Program), and the platform for developing and testing promising spending measures for subsequent use in formal payment models (eg, by using CEBP measures that possess large variation or cost-reduction opportunities in future bundled payment programs).
Despite these benefits, little is known about hospital performance on publicly reported episode-specific spending measures. We addressed this knowledge gap by providing what is, to our knowledge, the first nationwide description of hospital performance on such measures. We also evaluated which episode components accounted for spending variation in procedural vs condition episodes, examined whether CEBP measures can be used to effectively identify high- vs low-cost hospitals, and compared spending performance on CEBPs vs Medicare Spending Per Beneficiary.
METHODS
Data and Study Sample
We utilized publicly available data from Hospital Compare, which include information about hospital-level CEBP and Medicare Spending Per Beneficiary performance for Medicare-certified acute care hospitals nationwide.5 Our analysis evaluated the six CEBP measures tested by Medicare in 2017: three conditions (cellulitis, kidney/urinary tract infection [UTI], gastrointestinal hemorrhage) and three procedures (spinal fusion, cholecystectomy and common duct exploration, and aortic aneurysm repair). Per Medicare rules, CEBP measures are calculated only for hospitals with requisite volume for targeted conditions (minimum of 40 episodes) and procedures (minimum of 25 episodes) and are reported on Hospital Compare in risk-adjusted (eg, for age, hierarchical condition categories in alignment with existing Medicare methodology) and payment-standardized form (ie, accounts for wage index, medical education, disproportionate share hospital payments) . Each CEBP encompasses episodes with or without major complications/comorbidities.
For each hospital, CEBP spending is reported as average total episode spending, as well as average spending on specific components. We grouped components into three groups: hospitalization, skilled nursing facility (SNF) use, and other (encompassing postdischarge readmissions, emergency department visits, and home health agency use), with a focus on SNF given existing evidence from episode-based payment models about the opportunity for savings from reduced SNF care. Hospital Compare also provides information about the national CEBP measure performance (ie, average spending for a given episode type among all eligible hospitals nationwide).
Hospital Groups
To evaluate hospitals’ CEBP performance for specific episode types, we categorized hospitals as either “below average spending” if their average episode spending was below the national average or “above average spending” if spending was above the national average. According to this approach, a hospital could have below average spending for some episodes but above average spending for others.
To compare hospitals across episode types simultaneously, we categorized hospitals as “low cost” if episode spending was below the national average for all applicable measures, “high cost” if episode spending was above the national average for all applicable measures, or “mixed cost” if episode spending was above the national average for some measures and below for others.
We also conducted sensitivity analyses using alternative hospital group definitions. For comparisons of specific episode types, we categorized hospitals as “high spending” (top quartile of average episode spending among eligible hospitals) or “other spending” (all others). For comparisons across all episode types, we focused on SNF care and categorized hospitals as “high SNF cost” (top quartile of episode spending attributed to SNF care) and “other SNF cost” (all others). We applied a similar approach to Medicare Spending Per Beneficiary, categorizing hospitals as either “low MSPB cost” if their episode spending was below the national average for Medicare Spending Per Beneficiary or “high MSPB cost” if not.
Statistical Analysis
We assessed variation by describing the distribution of total episode spending across eligible hospitals for each individual episode type, as well as the proportion of spending attributed to SNF care across all episode types. We reported the difference between the 10th and 90th percentile for each distribution to quantify variation. To evaluate how individual episode components contributed to overall spending variation, we used linear regression and applied analysis of variance to each episode component. Specifically, we regressed episode spending on each episode component (hospital, SNF, other) separately and used these results to generate predicted episode spending values for each hospital based on its value for each spending component. We then calculated the differen-ces (ie, residuals) between predicted and actual total episode spending values. We plotted residuals for each component, with lower residual plot variation (ie, a flatter curve) representing larger contribution of a spending component to overall spending variation.
Pearson correlation coefficients were used to assess within-hospital CEBP correlation (ie, the extent to which performance was hospital specific). We evaluated if and how components of spending varied across hospitals by comparing spending groups (for individual episode types) and cost groups (for all episode types). To test the robustness of these categories, we conducted sensitivity analyses using high spending vs other spending groups (for individual episode types) and high SNF cost vs low SNF cost groups (for all episode types).
To assess concordance between CEBP and Medicare Spending Per Beneficiary, we cross tabulated hospital CEBP performance (high vs low vs mixed cost) and Medicare Spending Per Beneficiary performance (high vs low MSPB cost). This approached allowed us to quantify the number of hospitals that have concordant performance for both types of spending measures (ie, high cost or low cost on both) and the number with discordant performance (eg, high cost on one spending measure but low cost on the other). We used Pearson correlation coefficients to assess correlation between CEBP and Medicare Spending Per Beneficiary, with evaluation of CEBP performance in aggregate form (ie, hospitals’ average CEBP performance across all eligible episode types) and by individual episode types.
Chi-square and Kruskal-Wallis tests were used to compare categorical and continuous variables, respectively. To compare spending amounts, we evaluated the distribution of total episode spending (Appendix Figure 1) and used ordinary least squares regression with spending as the dependent variable and hospital group, episode components, and their interaction as independent variables. Because CEBP dollar amounts are reported through Hospital Compare on a risk-adjusted and payment-standardized basis, no additional adjustments were applied. Analyses were performed using SAS version 9.4 (SAS Institute; Cary, NC) and all tests of significance were two-tailed at alpha=0.05.
RESULTS
Of 3,129 hospitals, 1,778 achieved minimum thresholds and had CEBPs calculated for at least one of the six CEBP episode types.
Variation in CEBP Performance
For each episode type, spending varied across eligible hospitals (Appendix Figure 2). In particular, the difference between the 10th and 90th percentile values for cellulitis, kidney/UTI, and gastrointestinal hemorrhage were $2,873, $3,514, and $2,982, respectively. Differences were greater for procedural episodes of aortic aneurysm ($17,860), spinal fusion ($11,893), and cholecystectomy ($3,689). Evaluated across all episode types, the proportion of episode spending attributed to SNF care also varied across hospitals (Appendix Figure 3), with a difference of 24.7% between the 10th (4.5%) and 90th (29.2%) percentile values.
Residual plots demonstrated differences in which episode components accounted for variation in overall spending. For aortic aneurysm episodes, variation in the SNF episode component best explained variation in episode spending and thus had the lowest residual plot variation, followed by other and hospital components (Figure). Similar patterns were observed for spinal fusion and cholecystectomy episodes. In contrast, for cellulitis episodes, all three components had comparable residual-plot variation, which indicates that the variation in the components explained episode spending variation similarly (Figure)—a pattern reflected in kidney/UTI and gastrointestinal hemorrhage episodes.
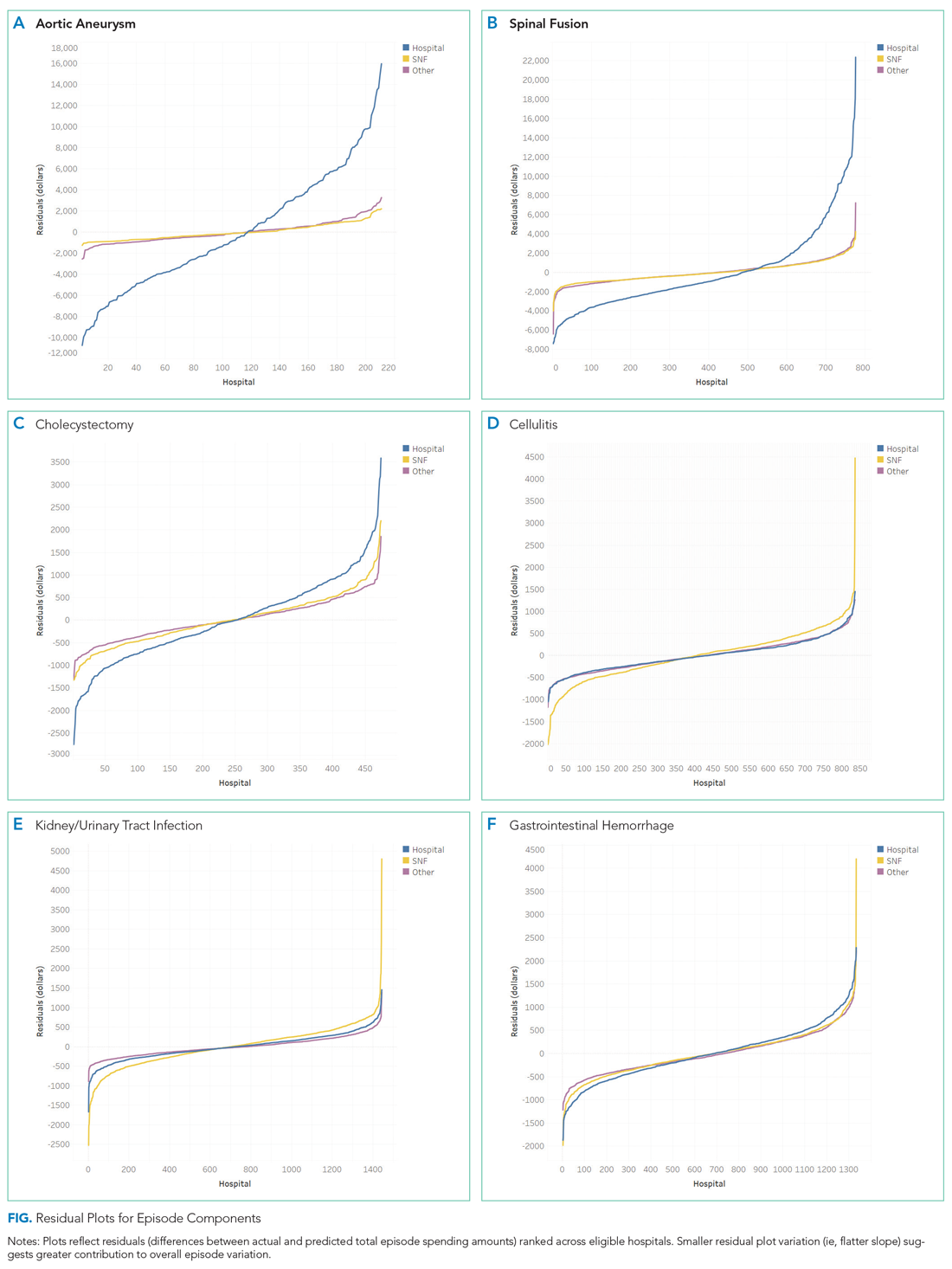
Correlation in Performance on CEBP Measures
Across hospitals in our sample, within-hospital correlations were generally low (Appendix Table 1). In particular, correlations ranged from
CEBP Performance by Hospital Groups
Overall spending on specific episode types varied across hospital groups (Table). Spending for aortic aneurysm episodes was $42,633 at hospitals with above average spending and $37,730 at those with below average spending, while spending for spinal fusion episodes was $39,231 at those with above average spending and $34,832 at those with below average spending. In comparison, spending at hospitals deemed above and below average spending for cellulitis episodes was $10,763 and $9,064, respectively, and $11,223 and $9,161 at hospitals deemed above and below average spending for kidney/UTI episodes, respectively.
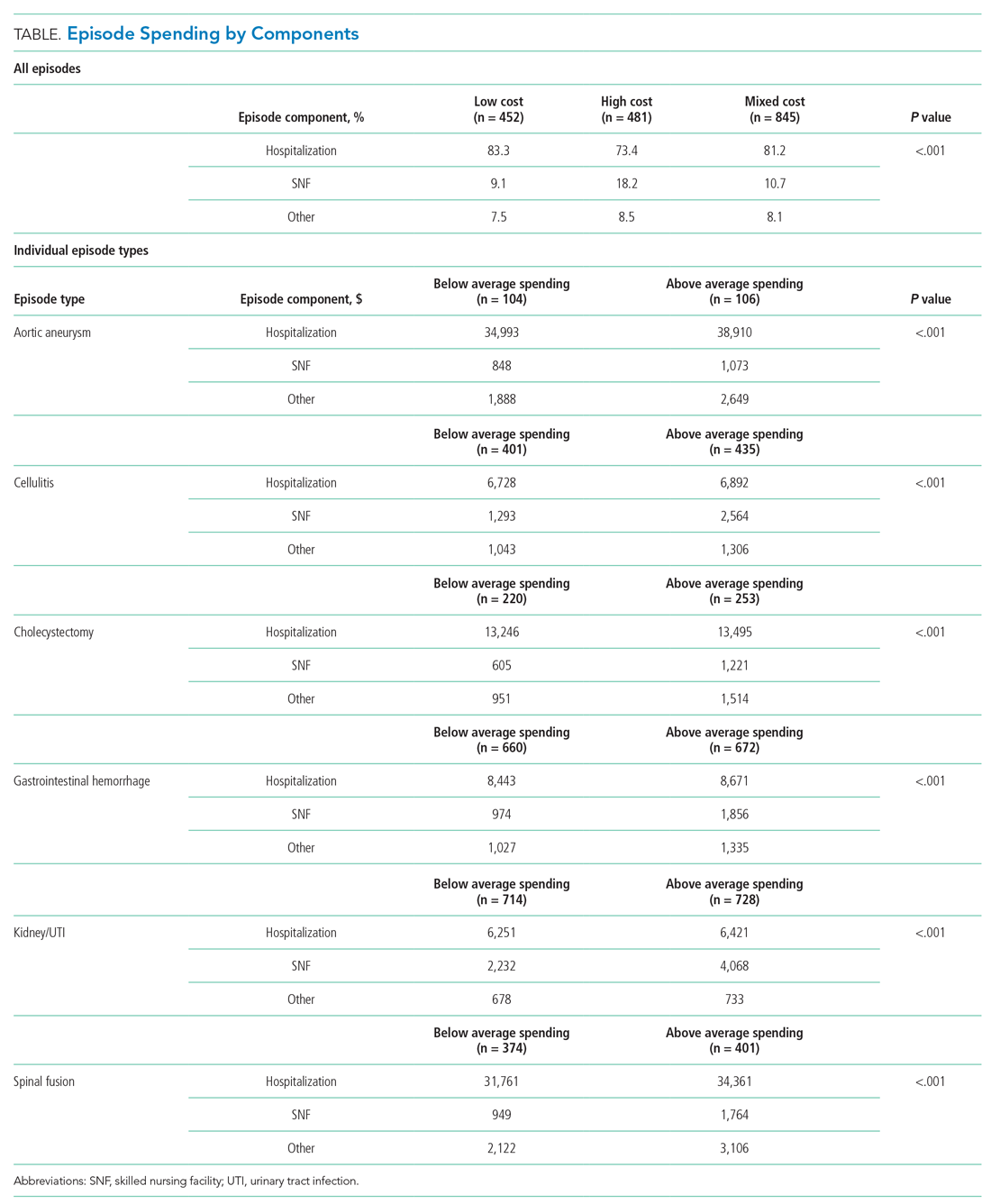
Spending on specific episode components also differed by hospital group (Table). Though the magnitude of absolute spending amounts and differences varied by specific episode, hospitals with above average spending tended to spend more on SNF than did those with below average spending. For example, hospitals with above average spending for cellulitis episodes spent an average of $2,564 on SNF (24% of overall episode spending) vs $1,293 (14% of episode spending) among those with below average spending. Similarly, hospitals with above and below average spending for kidney/UTI episodes spent $4,068 (36% of episode spending) and $2,232 (24% of episode spending) on SNF, respectively (P < .001 for both episode types). Findings were qualitatively similar in sensitivity analyses (Appendix Table 2).
Among hospitals in our sample, we categorized 481 as high cost (27%), 452 as low cost (25%), and 845 as mixed cost (48%), with hospital groups distributed broadly nationwide (Appendix Figure 4). Evaluated on performance across all six episode types, hospital groups also demonstrated differences in spending by cost components (Table). In particular, spending in SNF ranged from 18.1% of overall episode spending among high-cost hospitals to 10.7% among mixed-cost hospitals and 9.2% among low-cost hospitals. Additionally, spending on hospitalization accounted for 83.3% of overall episode spending among low-cost hospitals, compared with 81.2% and 73.4% among mixed-cost and high-cost hospitals, respectively (P < .001). Comparisons were qualitatively similar in sensitivity analyses (Appendix Table 3).
Comparison of CEBP and Medicare Spending Per Beneficiary Performance
Correlation between Medicare Spending Per Beneficiary and aggregated CEBPs was 0.42 and, for individual episode types, ranged between 0.14 and 0.36 (Appendix Table 2). There was low concordance between hospital performance on CEBP and Medicare Spending Per Beneficiary. Across all eligible hospitals, only 16.3% (290/1778) had positive concordance between performance on the two measure types (ie, low cost for both), while 16.5% (293/1778) had negative concordance (ie, high cost for both). There was discordant performance in most instances (67.2%; 1195/1778), which reflecting favorable performance on one measure type but not the other.
DISCUSSION
To our knowledge, this study is the first to describe hospitals’ episode-specific spending performance nationwide. It demonstrated significant variation across hospitals driven by different episode components for different episode types. It also showed low correlation between individual episode spending measures and poor concordance between episode-specific and global hospital spending measures. Two practice and policy implications are noteworthy.
First, our findings corroborate and build upon evidence from bundled payment programs about the opportunity for hospitals to improve their cost efficiency. Findings from bundled payment evaluations of surgical episodes suggest that the major area for cost savings is in the reduction of institutional post-acute care use such as that of SNFs.7-9 We demonstrated similar opportunity in a national sample of hospitals, finding that, for the three evaluated procedural CEBPs, SNF care accounted for more variation in overall episode spending than did other components. While variation may imply opportunity for greater efficiency and standardization, it is important to note that variation itself is not inherently problematic. Additional studies are needed to distinguish between warranted and unwarranted variation in procedural episodes, as well as identify strategies for reducing the latter.
Though bundled payment evaluations have predominantly emphasized procedural episodes, existing evidence suggests that participation in medical condition bundles has not been associated with cost savings or utilization changes.7-15 Findings from our analysis of variance—that there appear to be smaller variation-reduction opportunities for condition episodes than for procedural episodes—offer insight into this issue. Existing episodes are initiated by hospitalization and extend into the postacute period, a design that may not afford substantial post-acute care savings opportunities for condition episodes. This is an important insight as policymakers consider how to best design condition-based episodes in the future (eg, whether to use non–hospital based episode triggers). Future work should evaluate whether our findings reflect inherent differences between condition and procedural episodes16 or whether interventions can still optimize SNF care for these episodes despite smaller variation.
Second, our results highlight the potential limitations of global performance measures such as Medicare Spending Per Beneficiary. As a general measure of hospital spending, Medicare Spending Per Beneficiary is based on the premise that hospitals can be categorized as high or low cost with consideration of all inpatient episodic care. However, our analyses suggest that hospitals may be high cost for certain episodes and low cost for others—a fact highlighted by the low correlation and high discordance observed between hospital CEBP and Medicare Spending Per Beneficiary performance. Because overarching measures may miss spending differen-ces related to underlying clinical scenarios, episode-specific spending measures would provide important perspective and complements to global measures for assessing hospital cost performance, particularly in an era of value-based payments. Policymakers should consider prioritizing the development and implementation of such measures.
Our study has limitations. First, it is descriptive in nature, and future work should evaluate the association between episode-specific spending measure performance and clinical and quality outcomes. Second, we evaluated all CEBP-eligible hospitals nationwide to provide a broad view of episode-specific spending. However, future studies should assess performance among hospital subtypes, such as vertically integrated or safety-net organizations, because they may be more or less able to perform on these spending measures. Third, though findings may not be generalizable to other clinical episodes, our results were qualitatively consistent across episode types and broadly consistent with evidence from episode-based payment models. Fourth, we analyzed cost from the perspective of utilization and did not incorporate price considerations, which may be more relevant for commercial insurers than it is for Medicare.
Nonetheless, the emergence of CEBPs reflects the ongoing shift in policymaker attention toward episode-specific spending. In particular, though further scale or use of CEBP measures has been put on hold amid other payment reform changes, their nationwide implementation in 2017 signals Medicare’s broad interest in evaluating all hospitals on episode-specific spending efficiency, in addition to other facets of spending, quality, safety, and patient experience. Importantly, such efforts complement other ongoing nationwide initiatives for emphasizing episode spending, such as use of episode-based cost measures within the Merit-Based Incentive Payment System17 to score clinicians and groups in part based on their episode-specific spending efficiency. Insight about episode spending performance could help hospitals prepare for environments with increasing focus on episode spending and as policymakers incorporate this perspective into quality and value-based payment policies.
1. Centers for Medicare & Medicaid Services. Fiscal Year 2019 Clinical Episode-Based Payment Measures Overview. https://www.qualityreportingcenter.com/globalassets/migrated-pdf/cepb_slides_npc-6.17.2018_5.22.18_vfinal508.pdf. Accessed November 26, 2019.
2. Centers for Medicare & Medicaid Services. Hospital Inpatient Quality Reporting Program. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/HospitalQualityInits/HospitalRHQDAPU.html. Accessed November 23, 2019.
3. Centers for Medicare & Medicaid Services. Medicare Spending Per Beneficiary (MSPB) Spending Breakdown by Claim Type. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/hospital-value-based-purchasing/Downloads/Fact-Sheet-MSPB-Spending-Breakdowns-by-Claim-Type-Dec-2014.pdf. Accessed November 25, 2019.
4. Hu J, Jordan J, Rubinfeld I, Schreiber M, Waterman B, Nerenz D. Correlations among hospital quality measure: What “Hospital Compare” data tell us. Am J Med Qual. 2017;32(6):605-610. https://doi.org/10.1177/1062860616684012.
5. Centers for Medicare & Medicaid Services. Hospital Compare datasets. https://data.medicare.gov/data/hospital-compare. Accessed November 26, 2019.
6. American Hospital Association. AHA Data Products. https://www.aha.org/data-insights/aha-data-products. Accessed November 25, 2019.
7. Dummit LA, Kahvecioglu D, Marrufo G, et al. Bundled payment initiative and payments and quality outcomes for lower extremity joint replacement episodes. JAMA. 2016; 316(12):1267-1278. https://doi.org/10.1001/jama.2016.12717.
8. Finkelstein A, Ji Y, Mahoney N, Skinner J. Mandatory medicare bundled payment program for lower extremity joint replacement and discharge to institutional postacute care: Interim analysis of the first year of a 5-year randomized trial. JAMA. 2018;320(9):892-900. https://doi.org/10.1001/jama.2018.12346.
9. Navathe AS, Troxel AB, Liao JM, et al. Cost of joint replacement using bundled payment models. JAMA Intern Med. 2017;177(2):214-222. https://doi.org/10.1001/jamainternmed.2016.8263.
10. Liao JM, Emanuel EJ, Polsky DE, et al. National representativeness of hospitals and markets in Medicare’s mandatory bundled payment program. Health Aff. 2019;38(1):44-53.
11. Barnett ML, Wilcock A, McWilliams JM, et al. Two-year evaluation of mandatory bundled payments for joint replacement. N Engl J Med. 2019;380(3):252-262. https://doi.org/10.1056/NEJMsa1809010.
12. Navathe AS, Liao JM, Polsky D, et al. Comparison of hospitals participating in Medicare’s voluntary and mandatory orthopedic bundle programs. Health Aff. 2018;37(6):854-863. https://www.doi.org/10.1377/hlthaff.2017.1358.
13. Joynt Maddox KE, Orav EJ, Zheng J, Epstein AM. Participation and Dropout in the Bundled Payments for Care Improvement Initiative. JAMA. 2018;319(2):191-193. https://doi.org/10.1001/jama.2017.14771.
14. Navathe AS, Liao JM, Dykstra SE, et al. Association of hospital participation in a Medicare bundled payment program with volume and case mix of lower extremity joint replacement episodes. JAMA. 2018;320(9):901-910. https://doi.org/10.1001/jama.2018.12345.
15. Joynt Maddox KE, Orav EJ, Epstein AM. Medicare’s bundled payments initiative for medical conditions. N Engl J Med. 2018;379(18):e33. https://doi.org/10.1056/NEJMc1811049.
16. Navathe AS, Shan E, Liao JM. What have we learned about bundling medical conditions? Health Affairs Blog. https://www.healthaffairs.org/do/10.1377/hblog20180828.844613/full/. Accessed November 25, 2019.
17. Centers for Medicare & Medicaid Services. MACRA. https://www.cms.gov/medicare/quality-initiatives-patient-assessment-instruments/value-based-programs/macra-mips-and-apms/macra-mips-and-apms.html. Accessed November 26, 2019.
Amid the continued shift from fee-for-service toward value-based payment, policymakers such as the Centers for Medicare & Medicaid Services have initiated strategies to contain spending on episodes of care. This episode focus has led to nationwide implementation of payment models such as bundled payments, which hold hospitals accountable for quality and costs across procedure-based (eg, coronary artery bypass surgery) and condition-based (eg, congestive heart failure) episodes, which begin with hospitalization and encompass subsequent hospital and postdischarge care.
Simultaneously, Medicare has increased its emphasis on similarly designed episodes of care (eg, those spanning hospitalization and postdischarge care) using other strategies, such as public reporting and use of episode-based measures to evaluate hospital cost performance. In 2017, Medicare trialed the implementation of six Clinical Episode-Based Payment (CEBP) measures in the national Hospital Inpatient Quality Reporting Program in order to assess hospital and clinician spending on procedure and condition episodes.1,2
CEBP measures reflect episode-specific spending, conveying “how expensive a hospital is” by capturing facility and professional payments for a given episode spanning between 3 days prior to hospitalization and 30 days following discharge. Given standard payment rates used in Medicare, the variation in episode spending reflects differences in quantity and type of services utilized within an episode. Medicare has specified episode-related services and designed CEBP measures via logic and definition rules informed by a combination of claims and procedures-based grouping, as well as by physician input. For example, the CEBP measure for cellulitis encompasses services related to diagnosing and treating the infection within the episode window, but not unrelated services such as eye exams for coexisting glaucoma. To increase clinical salience, CEBP measures are subdivided to reflect differing complexity when possible. For instance, cellulitis measures are divided into episodes with or without major complications or comorbidities and further subdivided into subtypes for episodes reflecting cellulitis in patients with diabetes, patients with decubitus ulcers, or neither.
CEBPs are similar to other spending measures used in payment programs, such as the Medicare Spending Per Beneficiary, but are more clinically relevant because their focus on episodes more closely reflects clinical practice. CEBPs and Medicare Spending Per Beneficiary have similar designs (eg, same episode windows) and purpose (eg, to capture the cost efficiency of hospital care).3 However, unlike CEBPs, Medicare Spending Per Beneficiary is a “global” measure that summarizes a hospital’s cost efficiency aggregated across all inpatient episodes rather than represent it based on specific conditions or procedures.4 The limitations of publicly reported global hospital measures—for instance, the poor correlation between hospital performance on distinct publicly reported quality measures5—highlight the potential utility of episode-specific spending measures such as CEBP.
Compared with episode-based payment models, initiatives such as CEBP measures have gone largely unstudied. However, they represent signals of Medicare’s growing commitment to addressing care episodes, tested without potentially tedious rulemaking required to change payment. In fact, publicly reported episode spending measures offer policymakers several interrelated benefits: the ability to rapidly evaluate performance at a large number of hospitals (eg, Medicare scaling up CEBP measures among all eligible hospitals nationwide), the option of leveraging publicly reported feedback to prompt clinical improvements (eg, by including CEBP measures in the Hospital Inpatient Quality Reporting Program), and the platform for developing and testing promising spending measures for subsequent use in formal payment models (eg, by using CEBP measures that possess large variation or cost-reduction opportunities in future bundled payment programs).
Despite these benefits, little is known about hospital performance on publicly reported episode-specific spending measures. We addressed this knowledge gap by providing what is, to our knowledge, the first nationwide description of hospital performance on such measures. We also evaluated which episode components accounted for spending variation in procedural vs condition episodes, examined whether CEBP measures can be used to effectively identify high- vs low-cost hospitals, and compared spending performance on CEBPs vs Medicare Spending Per Beneficiary.
METHODS
Data and Study Sample
We utilized publicly available data from Hospital Compare, which include information about hospital-level CEBP and Medicare Spending Per Beneficiary performance for Medicare-certified acute care hospitals nationwide.5 Our analysis evaluated the six CEBP measures tested by Medicare in 2017: three conditions (cellulitis, kidney/urinary tract infection [UTI], gastrointestinal hemorrhage) and three procedures (spinal fusion, cholecystectomy and common duct exploration, and aortic aneurysm repair). Per Medicare rules, CEBP measures are calculated only for hospitals with requisite volume for targeted conditions (minimum of 40 episodes) and procedures (minimum of 25 episodes) and are reported on Hospital Compare in risk-adjusted (eg, for age, hierarchical condition categories in alignment with existing Medicare methodology) and payment-standardized form (ie, accounts for wage index, medical education, disproportionate share hospital payments) . Each CEBP encompasses episodes with or without major complications/comorbidities.
For each hospital, CEBP spending is reported as average total episode spending, as well as average spending on specific components. We grouped components into three groups: hospitalization, skilled nursing facility (SNF) use, and other (encompassing postdischarge readmissions, emergency department visits, and home health agency use), with a focus on SNF given existing evidence from episode-based payment models about the opportunity for savings from reduced SNF care. Hospital Compare also provides information about the national CEBP measure performance (ie, average spending for a given episode type among all eligible hospitals nationwide).
Hospital Groups
To evaluate hospitals’ CEBP performance for specific episode types, we categorized hospitals as either “below average spending” if their average episode spending was below the national average or “above average spending” if spending was above the national average. According to this approach, a hospital could have below average spending for some episodes but above average spending for others.
To compare hospitals across episode types simultaneously, we categorized hospitals as “low cost” if episode spending was below the national average for all applicable measures, “high cost” if episode spending was above the national average for all applicable measures, or “mixed cost” if episode spending was above the national average for some measures and below for others.
We also conducted sensitivity analyses using alternative hospital group definitions. For comparisons of specific episode types, we categorized hospitals as “high spending” (top quartile of average episode spending among eligible hospitals) or “other spending” (all others). For comparisons across all episode types, we focused on SNF care and categorized hospitals as “high SNF cost” (top quartile of episode spending attributed to SNF care) and “other SNF cost” (all others). We applied a similar approach to Medicare Spending Per Beneficiary, categorizing hospitals as either “low MSPB cost” if their episode spending was below the national average for Medicare Spending Per Beneficiary or “high MSPB cost” if not.
Statistical Analysis
We assessed variation by describing the distribution of total episode spending across eligible hospitals for each individual episode type, as well as the proportion of spending attributed to SNF care across all episode types. We reported the difference between the 10th and 90th percentile for each distribution to quantify variation. To evaluate how individual episode components contributed to overall spending variation, we used linear regression and applied analysis of variance to each episode component. Specifically, we regressed episode spending on each episode component (hospital, SNF, other) separately and used these results to generate predicted episode spending values for each hospital based on its value for each spending component. We then calculated the differen-ces (ie, residuals) between predicted and actual total episode spending values. We plotted residuals for each component, with lower residual plot variation (ie, a flatter curve) representing larger contribution of a spending component to overall spending variation.
Pearson correlation coefficients were used to assess within-hospital CEBP correlation (ie, the extent to which performance was hospital specific). We evaluated if and how components of spending varied across hospitals by comparing spending groups (for individual episode types) and cost groups (for all episode types). To test the robustness of these categories, we conducted sensitivity analyses using high spending vs other spending groups (for individual episode types) and high SNF cost vs low SNF cost groups (for all episode types).
To assess concordance between CEBP and Medicare Spending Per Beneficiary, we cross tabulated hospital CEBP performance (high vs low vs mixed cost) and Medicare Spending Per Beneficiary performance (high vs low MSPB cost). This approached allowed us to quantify the number of hospitals that have concordant performance for both types of spending measures (ie, high cost or low cost on both) and the number with discordant performance (eg, high cost on one spending measure but low cost on the other). We used Pearson correlation coefficients to assess correlation between CEBP and Medicare Spending Per Beneficiary, with evaluation of CEBP performance in aggregate form (ie, hospitals’ average CEBP performance across all eligible episode types) and by individual episode types.
Chi-square and Kruskal-Wallis tests were used to compare categorical and continuous variables, respectively. To compare spending amounts, we evaluated the distribution of total episode spending (Appendix Figure 1) and used ordinary least squares regression with spending as the dependent variable and hospital group, episode components, and their interaction as independent variables. Because CEBP dollar amounts are reported through Hospital Compare on a risk-adjusted and payment-standardized basis, no additional adjustments were applied. Analyses were performed using SAS version 9.4 (SAS Institute; Cary, NC) and all tests of significance were two-tailed at alpha=0.05.
RESULTS
Of 3,129 hospitals, 1,778 achieved minimum thresholds and had CEBPs calculated for at least one of the six CEBP episode types.
Variation in CEBP Performance
For each episode type, spending varied across eligible hospitals (Appendix Figure 2). In particular, the difference between the 10th and 90th percentile values for cellulitis, kidney/UTI, and gastrointestinal hemorrhage were $2,873, $3,514, and $2,982, respectively. Differences were greater for procedural episodes of aortic aneurysm ($17,860), spinal fusion ($11,893), and cholecystectomy ($3,689). Evaluated across all episode types, the proportion of episode spending attributed to SNF care also varied across hospitals (Appendix Figure 3), with a difference of 24.7% between the 10th (4.5%) and 90th (29.2%) percentile values.
Residual plots demonstrated differences in which episode components accounted for variation in overall spending. For aortic aneurysm episodes, variation in the SNF episode component best explained variation in episode spending and thus had the lowest residual plot variation, followed by other and hospital components (Figure). Similar patterns were observed for spinal fusion and cholecystectomy episodes. In contrast, for cellulitis episodes, all three components had comparable residual-plot variation, which indicates that the variation in the components explained episode spending variation similarly (Figure)—a pattern reflected in kidney/UTI and gastrointestinal hemorrhage episodes.

Correlation in Performance on CEBP Measures
Across hospitals in our sample, within-hospital correlations were generally low (Appendix Table 1). In particular, correlations ranged from
CEBP Performance by Hospital Groups
Overall spending on specific episode types varied across hospital groups (Table). Spending for aortic aneurysm episodes was $42,633 at hospitals with above average spending and $37,730 at those with below average spending, while spending for spinal fusion episodes was $39,231 at those with above average spending and $34,832 at those with below average spending. In comparison, spending at hospitals deemed above and below average spending for cellulitis episodes was $10,763 and $9,064, respectively, and $11,223 and $9,161 at hospitals deemed above and below average spending for kidney/UTI episodes, respectively.

Spending on specific episode components also differed by hospital group (Table). Though the magnitude of absolute spending amounts and differences varied by specific episode, hospitals with above average spending tended to spend more on SNF than did those with below average spending. For example, hospitals with above average spending for cellulitis episodes spent an average of $2,564 on SNF (24% of overall episode spending) vs $1,293 (14% of episode spending) among those with below average spending. Similarly, hospitals with above and below average spending for kidney/UTI episodes spent $4,068 (36% of episode spending) and $2,232 (24% of episode spending) on SNF, respectively (P < .001 for both episode types). Findings were qualitatively similar in sensitivity analyses (Appendix Table 2).
Among hospitals in our sample, we categorized 481 as high cost (27%), 452 as low cost (25%), and 845 as mixed cost (48%), with hospital groups distributed broadly nationwide (Appendix Figure 4). Evaluated on performance across all six episode types, hospital groups also demonstrated differences in spending by cost components (Table). In particular, spending in SNF ranged from 18.1% of overall episode spending among high-cost hospitals to 10.7% among mixed-cost hospitals and 9.2% among low-cost hospitals. Additionally, spending on hospitalization accounted for 83.3% of overall episode spending among low-cost hospitals, compared with 81.2% and 73.4% among mixed-cost and high-cost hospitals, respectively (P < .001). Comparisons were qualitatively similar in sensitivity analyses (Appendix Table 3).
Comparison of CEBP and Medicare Spending Per Beneficiary Performance
Correlation between Medicare Spending Per Beneficiary and aggregated CEBPs was 0.42 and, for individual episode types, ranged between 0.14 and 0.36 (Appendix Table 2). There was low concordance between hospital performance on CEBP and Medicare Spending Per Beneficiary. Across all eligible hospitals, only 16.3% (290/1778) had positive concordance between performance on the two measure types (ie, low cost for both), while 16.5% (293/1778) had negative concordance (ie, high cost for both). There was discordant performance in most instances (67.2%; 1195/1778), which reflecting favorable performance on one measure type but not the other.
DISCUSSION
To our knowledge, this study is the first to describe hospitals’ episode-specific spending performance nationwide. It demonstrated significant variation across hospitals driven by different episode components for different episode types. It also showed low correlation between individual episode spending measures and poor concordance between episode-specific and global hospital spending measures. Two practice and policy implications are noteworthy.
First, our findings corroborate and build upon evidence from bundled payment programs about the opportunity for hospitals to improve their cost efficiency. Findings from bundled payment evaluations of surgical episodes suggest that the major area for cost savings is in the reduction of institutional post-acute care use such as that of SNFs.7-9 We demonstrated similar opportunity in a national sample of hospitals, finding that, for the three evaluated procedural CEBPs, SNF care accounted for more variation in overall episode spending than did other components. While variation may imply opportunity for greater efficiency and standardization, it is important to note that variation itself is not inherently problematic. Additional studies are needed to distinguish between warranted and unwarranted variation in procedural episodes, as well as identify strategies for reducing the latter.
Though bundled payment evaluations have predominantly emphasized procedural episodes, existing evidence suggests that participation in medical condition bundles has not been associated with cost savings or utilization changes.7-15 Findings from our analysis of variance—that there appear to be smaller variation-reduction opportunities for condition episodes than for procedural episodes—offer insight into this issue. Existing episodes are initiated by hospitalization and extend into the postacute period, a design that may not afford substantial post-acute care savings opportunities for condition episodes. This is an important insight as policymakers consider how to best design condition-based episodes in the future (eg, whether to use non–hospital based episode triggers). Future work should evaluate whether our findings reflect inherent differences between condition and procedural episodes16 or whether interventions can still optimize SNF care for these episodes despite smaller variation.
Second, our results highlight the potential limitations of global performance measures such as Medicare Spending Per Beneficiary. As a general measure of hospital spending, Medicare Spending Per Beneficiary is based on the premise that hospitals can be categorized as high or low cost with consideration of all inpatient episodic care. However, our analyses suggest that hospitals may be high cost for certain episodes and low cost for others—a fact highlighted by the low correlation and high discordance observed between hospital CEBP and Medicare Spending Per Beneficiary performance. Because overarching measures may miss spending differen-ces related to underlying clinical scenarios, episode-specific spending measures would provide important perspective and complements to global measures for assessing hospital cost performance, particularly in an era of value-based payments. Policymakers should consider prioritizing the development and implementation of such measures.
Our study has limitations. First, it is descriptive in nature, and future work should evaluate the association between episode-specific spending measure performance and clinical and quality outcomes. Second, we evaluated all CEBP-eligible hospitals nationwide to provide a broad view of episode-specific spending. However, future studies should assess performance among hospital subtypes, such as vertically integrated or safety-net organizations, because they may be more or less able to perform on these spending measures. Third, though findings may not be generalizable to other clinical episodes, our results were qualitatively consistent across episode types and broadly consistent with evidence from episode-based payment models. Fourth, we analyzed cost from the perspective of utilization and did not incorporate price considerations, which may be more relevant for commercial insurers than it is for Medicare.
Nonetheless, the emergence of CEBPs reflects the ongoing shift in policymaker attention toward episode-specific spending. In particular, though further scale or use of CEBP measures has been put on hold amid other payment reform changes, their nationwide implementation in 2017 signals Medicare’s broad interest in evaluating all hospitals on episode-specific spending efficiency, in addition to other facets of spending, quality, safety, and patient experience. Importantly, such efforts complement other ongoing nationwide initiatives for emphasizing episode spending, such as use of episode-based cost measures within the Merit-Based Incentive Payment System17 to score clinicians and groups in part based on their episode-specific spending efficiency. Insight about episode spending performance could help hospitals prepare for environments with increasing focus on episode spending and as policymakers incorporate this perspective into quality and value-based payment policies.
Amid the continued shift from fee-for-service toward value-based payment, policymakers such as the Centers for Medicare & Medicaid Services have initiated strategies to contain spending on episodes of care. This episode focus has led to nationwide implementation of payment models such as bundled payments, which hold hospitals accountable for quality and costs across procedure-based (eg, coronary artery bypass surgery) and condition-based (eg, congestive heart failure) episodes, which begin with hospitalization and encompass subsequent hospital and postdischarge care.
Simultaneously, Medicare has increased its emphasis on similarly designed episodes of care (eg, those spanning hospitalization and postdischarge care) using other strategies, such as public reporting and use of episode-based measures to evaluate hospital cost performance. In 2017, Medicare trialed the implementation of six Clinical Episode-Based Payment (CEBP) measures in the national Hospital Inpatient Quality Reporting Program in order to assess hospital and clinician spending on procedure and condition episodes.1,2
CEBP measures reflect episode-specific spending, conveying “how expensive a hospital is” by capturing facility and professional payments for a given episode spanning between 3 days prior to hospitalization and 30 days following discharge. Given standard payment rates used in Medicare, the variation in episode spending reflects differences in quantity and type of services utilized within an episode. Medicare has specified episode-related services and designed CEBP measures via logic and definition rules informed by a combination of claims and procedures-based grouping, as well as by physician input. For example, the CEBP measure for cellulitis encompasses services related to diagnosing and treating the infection within the episode window, but not unrelated services such as eye exams for coexisting glaucoma. To increase clinical salience, CEBP measures are subdivided to reflect differing complexity when possible. For instance, cellulitis measures are divided into episodes with or without major complications or comorbidities and further subdivided into subtypes for episodes reflecting cellulitis in patients with diabetes, patients with decubitus ulcers, or neither.
CEBPs are similar to other spending measures used in payment programs, such as the Medicare Spending Per Beneficiary, but are more clinically relevant because their focus on episodes more closely reflects clinical practice. CEBPs and Medicare Spending Per Beneficiary have similar designs (eg, same episode windows) and purpose (eg, to capture the cost efficiency of hospital care).3 However, unlike CEBPs, Medicare Spending Per Beneficiary is a “global” measure that summarizes a hospital’s cost efficiency aggregated across all inpatient episodes rather than represent it based on specific conditions or procedures.4 The limitations of publicly reported global hospital measures—for instance, the poor correlation between hospital performance on distinct publicly reported quality measures5—highlight the potential utility of episode-specific spending measures such as CEBP.
Compared with episode-based payment models, initiatives such as CEBP measures have gone largely unstudied. However, they represent signals of Medicare’s growing commitment to addressing care episodes, tested without potentially tedious rulemaking required to change payment. In fact, publicly reported episode spending measures offer policymakers several interrelated benefits: the ability to rapidly evaluate performance at a large number of hospitals (eg, Medicare scaling up CEBP measures among all eligible hospitals nationwide), the option of leveraging publicly reported feedback to prompt clinical improvements (eg, by including CEBP measures in the Hospital Inpatient Quality Reporting Program), and the platform for developing and testing promising spending measures for subsequent use in formal payment models (eg, by using CEBP measures that possess large variation or cost-reduction opportunities in future bundled payment programs).
Despite these benefits, little is known about hospital performance on publicly reported episode-specific spending measures. We addressed this knowledge gap by providing what is, to our knowledge, the first nationwide description of hospital performance on such measures. We also evaluated which episode components accounted for spending variation in procedural vs condition episodes, examined whether CEBP measures can be used to effectively identify high- vs low-cost hospitals, and compared spending performance on CEBPs vs Medicare Spending Per Beneficiary.
METHODS
Data and Study Sample
We utilized publicly available data from Hospital Compare, which include information about hospital-level CEBP and Medicare Spending Per Beneficiary performance for Medicare-certified acute care hospitals nationwide.5 Our analysis evaluated the six CEBP measures tested by Medicare in 2017: three conditions (cellulitis, kidney/urinary tract infection [UTI], gastrointestinal hemorrhage) and three procedures (spinal fusion, cholecystectomy and common duct exploration, and aortic aneurysm repair). Per Medicare rules, CEBP measures are calculated only for hospitals with requisite volume for targeted conditions (minimum of 40 episodes) and procedures (minimum of 25 episodes) and are reported on Hospital Compare in risk-adjusted (eg, for age, hierarchical condition categories in alignment with existing Medicare methodology) and payment-standardized form (ie, accounts for wage index, medical education, disproportionate share hospital payments) . Each CEBP encompasses episodes with or without major complications/comorbidities.
For each hospital, CEBP spending is reported as average total episode spending, as well as average spending on specific components. We grouped components into three groups: hospitalization, skilled nursing facility (SNF) use, and other (encompassing postdischarge readmissions, emergency department visits, and home health agency use), with a focus on SNF given existing evidence from episode-based payment models about the opportunity for savings from reduced SNF care. Hospital Compare also provides information about the national CEBP measure performance (ie, average spending for a given episode type among all eligible hospitals nationwide).
Hospital Groups
To evaluate hospitals’ CEBP performance for specific episode types, we categorized hospitals as either “below average spending” if their average episode spending was below the national average or “above average spending” if spending was above the national average. According to this approach, a hospital could have below average spending for some episodes but above average spending for others.
To compare hospitals across episode types simultaneously, we categorized hospitals as “low cost” if episode spending was below the national average for all applicable measures, “high cost” if episode spending was above the national average for all applicable measures, or “mixed cost” if episode spending was above the national average for some measures and below for others.
We also conducted sensitivity analyses using alternative hospital group definitions. For comparisons of specific episode types, we categorized hospitals as “high spending” (top quartile of average episode spending among eligible hospitals) or “other spending” (all others). For comparisons across all episode types, we focused on SNF care and categorized hospitals as “high SNF cost” (top quartile of episode spending attributed to SNF care) and “other SNF cost” (all others). We applied a similar approach to Medicare Spending Per Beneficiary, categorizing hospitals as either “low MSPB cost” if their episode spending was below the national average for Medicare Spending Per Beneficiary or “high MSPB cost” if not.
Statistical Analysis
We assessed variation by describing the distribution of total episode spending across eligible hospitals for each individual episode type, as well as the proportion of spending attributed to SNF care across all episode types. We reported the difference between the 10th and 90th percentile for each distribution to quantify variation. To evaluate how individual episode components contributed to overall spending variation, we used linear regression and applied analysis of variance to each episode component. Specifically, we regressed episode spending on each episode component (hospital, SNF, other) separately and used these results to generate predicted episode spending values for each hospital based on its value for each spending component. We then calculated the differen-ces (ie, residuals) between predicted and actual total episode spending values. We plotted residuals for each component, with lower residual plot variation (ie, a flatter curve) representing larger contribution of a spending component to overall spending variation.
Pearson correlation coefficients were used to assess within-hospital CEBP correlation (ie, the extent to which performance was hospital specific). We evaluated if and how components of spending varied across hospitals by comparing spending groups (for individual episode types) and cost groups (for all episode types). To test the robustness of these categories, we conducted sensitivity analyses using high spending vs other spending groups (for individual episode types) and high SNF cost vs low SNF cost groups (for all episode types).
To assess concordance between CEBP and Medicare Spending Per Beneficiary, we cross tabulated hospital CEBP performance (high vs low vs mixed cost) and Medicare Spending Per Beneficiary performance (high vs low MSPB cost). This approached allowed us to quantify the number of hospitals that have concordant performance for both types of spending measures (ie, high cost or low cost on both) and the number with discordant performance (eg, high cost on one spending measure but low cost on the other). We used Pearson correlation coefficients to assess correlation between CEBP and Medicare Spending Per Beneficiary, with evaluation of CEBP performance in aggregate form (ie, hospitals’ average CEBP performance across all eligible episode types) and by individual episode types.
Chi-square and Kruskal-Wallis tests were used to compare categorical and continuous variables, respectively. To compare spending amounts, we evaluated the distribution of total episode spending (Appendix Figure 1) and used ordinary least squares regression with spending as the dependent variable and hospital group, episode components, and their interaction as independent variables. Because CEBP dollar amounts are reported through Hospital Compare on a risk-adjusted and payment-standardized basis, no additional adjustments were applied. Analyses were performed using SAS version 9.4 (SAS Institute; Cary, NC) and all tests of significance were two-tailed at alpha=0.05.
RESULTS
Of 3,129 hospitals, 1,778 achieved minimum thresholds and had CEBPs calculated for at least one of the six CEBP episode types.
Variation in CEBP Performance
For each episode type, spending varied across eligible hospitals (Appendix Figure 2). In particular, the difference between the 10th and 90th percentile values for cellulitis, kidney/UTI, and gastrointestinal hemorrhage were $2,873, $3,514, and $2,982, respectively. Differences were greater for procedural episodes of aortic aneurysm ($17,860), spinal fusion ($11,893), and cholecystectomy ($3,689). Evaluated across all episode types, the proportion of episode spending attributed to SNF care also varied across hospitals (Appendix Figure 3), with a difference of 24.7% between the 10th (4.5%) and 90th (29.2%) percentile values.
Residual plots demonstrated differences in which episode components accounted for variation in overall spending. For aortic aneurysm episodes, variation in the SNF episode component best explained variation in episode spending and thus had the lowest residual plot variation, followed by other and hospital components (Figure). Similar patterns were observed for spinal fusion and cholecystectomy episodes. In contrast, for cellulitis episodes, all three components had comparable residual-plot variation, which indicates that the variation in the components explained episode spending variation similarly (Figure)—a pattern reflected in kidney/UTI and gastrointestinal hemorrhage episodes.

Correlation in Performance on CEBP Measures
Across hospitals in our sample, within-hospital correlations were generally low (Appendix Table 1). In particular, correlations ranged from
CEBP Performance by Hospital Groups
Overall spending on specific episode types varied across hospital groups (Table). Spending for aortic aneurysm episodes was $42,633 at hospitals with above average spending and $37,730 at those with below average spending, while spending for spinal fusion episodes was $39,231 at those with above average spending and $34,832 at those with below average spending. In comparison, spending at hospitals deemed above and below average spending for cellulitis episodes was $10,763 and $9,064, respectively, and $11,223 and $9,161 at hospitals deemed above and below average spending for kidney/UTI episodes, respectively.

Spending on specific episode components also differed by hospital group (Table). Though the magnitude of absolute spending amounts and differences varied by specific episode, hospitals with above average spending tended to spend more on SNF than did those with below average spending. For example, hospitals with above average spending for cellulitis episodes spent an average of $2,564 on SNF (24% of overall episode spending) vs $1,293 (14% of episode spending) among those with below average spending. Similarly, hospitals with above and below average spending for kidney/UTI episodes spent $4,068 (36% of episode spending) and $2,232 (24% of episode spending) on SNF, respectively (P < .001 for both episode types). Findings were qualitatively similar in sensitivity analyses (Appendix Table 2).
Among hospitals in our sample, we categorized 481 as high cost (27%), 452 as low cost (25%), and 845 as mixed cost (48%), with hospital groups distributed broadly nationwide (Appendix Figure 4). Evaluated on performance across all six episode types, hospital groups also demonstrated differences in spending by cost components (Table). In particular, spending in SNF ranged from 18.1% of overall episode spending among high-cost hospitals to 10.7% among mixed-cost hospitals and 9.2% among low-cost hospitals. Additionally, spending on hospitalization accounted for 83.3% of overall episode spending among low-cost hospitals, compared with 81.2% and 73.4% among mixed-cost and high-cost hospitals, respectively (P < .001). Comparisons were qualitatively similar in sensitivity analyses (Appendix Table 3).
Comparison of CEBP and Medicare Spending Per Beneficiary Performance
Correlation between Medicare Spending Per Beneficiary and aggregated CEBPs was 0.42 and, for individual episode types, ranged between 0.14 and 0.36 (Appendix Table 2). There was low concordance between hospital performance on CEBP and Medicare Spending Per Beneficiary. Across all eligible hospitals, only 16.3% (290/1778) had positive concordance between performance on the two measure types (ie, low cost for both), while 16.5% (293/1778) had negative concordance (ie, high cost for both). There was discordant performance in most instances (67.2%; 1195/1778), which reflecting favorable performance on one measure type but not the other.
DISCUSSION
To our knowledge, this study is the first to describe hospitals’ episode-specific spending performance nationwide. It demonstrated significant variation across hospitals driven by different episode components for different episode types. It also showed low correlation between individual episode spending measures and poor concordance between episode-specific and global hospital spending measures. Two practice and policy implications are noteworthy.
First, our findings corroborate and build upon evidence from bundled payment programs about the opportunity for hospitals to improve their cost efficiency. Findings from bundled payment evaluations of surgical episodes suggest that the major area for cost savings is in the reduction of institutional post-acute care use such as that of SNFs.7-9 We demonstrated similar opportunity in a national sample of hospitals, finding that, for the three evaluated procedural CEBPs, SNF care accounted for more variation in overall episode spending than did other components. While variation may imply opportunity for greater efficiency and standardization, it is important to note that variation itself is not inherently problematic. Additional studies are needed to distinguish between warranted and unwarranted variation in procedural episodes, as well as identify strategies for reducing the latter.
Though bundled payment evaluations have predominantly emphasized procedural episodes, existing evidence suggests that participation in medical condition bundles has not been associated with cost savings or utilization changes.7-15 Findings from our analysis of variance—that there appear to be smaller variation-reduction opportunities for condition episodes than for procedural episodes—offer insight into this issue. Existing episodes are initiated by hospitalization and extend into the postacute period, a design that may not afford substantial post-acute care savings opportunities for condition episodes. This is an important insight as policymakers consider how to best design condition-based episodes in the future (eg, whether to use non–hospital based episode triggers). Future work should evaluate whether our findings reflect inherent differences between condition and procedural episodes16 or whether interventions can still optimize SNF care for these episodes despite smaller variation.
Second, our results highlight the potential limitations of global performance measures such as Medicare Spending Per Beneficiary. As a general measure of hospital spending, Medicare Spending Per Beneficiary is based on the premise that hospitals can be categorized as high or low cost with consideration of all inpatient episodic care. However, our analyses suggest that hospitals may be high cost for certain episodes and low cost for others—a fact highlighted by the low correlation and high discordance observed between hospital CEBP and Medicare Spending Per Beneficiary performance. Because overarching measures may miss spending differen-ces related to underlying clinical scenarios, episode-specific spending measures would provide important perspective and complements to global measures for assessing hospital cost performance, particularly in an era of value-based payments. Policymakers should consider prioritizing the development and implementation of such measures.
Our study has limitations. First, it is descriptive in nature, and future work should evaluate the association between episode-specific spending measure performance and clinical and quality outcomes. Second, we evaluated all CEBP-eligible hospitals nationwide to provide a broad view of episode-specific spending. However, future studies should assess performance among hospital subtypes, such as vertically integrated or safety-net organizations, because they may be more or less able to perform on these spending measures. Third, though findings may not be generalizable to other clinical episodes, our results were qualitatively consistent across episode types and broadly consistent with evidence from episode-based payment models. Fourth, we analyzed cost from the perspective of utilization and did not incorporate price considerations, which may be more relevant for commercial insurers than it is for Medicare.
Nonetheless, the emergence of CEBPs reflects the ongoing shift in policymaker attention toward episode-specific spending. In particular, though further scale or use of CEBP measures has been put on hold amid other payment reform changes, their nationwide implementation in 2017 signals Medicare’s broad interest in evaluating all hospitals on episode-specific spending efficiency, in addition to other facets of spending, quality, safety, and patient experience. Importantly, such efforts complement other ongoing nationwide initiatives for emphasizing episode spending, such as use of episode-based cost measures within the Merit-Based Incentive Payment System17 to score clinicians and groups in part based on their episode-specific spending efficiency. Insight about episode spending performance could help hospitals prepare for environments with increasing focus on episode spending and as policymakers incorporate this perspective into quality and value-based payment policies.
1. Centers for Medicare & Medicaid Services. Fiscal Year 2019 Clinical Episode-Based Payment Measures Overview. https://www.qualityreportingcenter.com/globalassets/migrated-pdf/cepb_slides_npc-6.17.2018_5.22.18_vfinal508.pdf. Accessed November 26, 2019.
2. Centers for Medicare & Medicaid Services. Hospital Inpatient Quality Reporting Program. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/HospitalQualityInits/HospitalRHQDAPU.html. Accessed November 23, 2019.
3. Centers for Medicare & Medicaid Services. Medicare Spending Per Beneficiary (MSPB) Spending Breakdown by Claim Type. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/hospital-value-based-purchasing/Downloads/Fact-Sheet-MSPB-Spending-Breakdowns-by-Claim-Type-Dec-2014.pdf. Accessed November 25, 2019.
4. Hu J, Jordan J, Rubinfeld I, Schreiber M, Waterman B, Nerenz D. Correlations among hospital quality measure: What “Hospital Compare” data tell us. Am J Med Qual. 2017;32(6):605-610. https://doi.org/10.1177/1062860616684012.
5. Centers for Medicare & Medicaid Services. Hospital Compare datasets. https://data.medicare.gov/data/hospital-compare. Accessed November 26, 2019.
6. American Hospital Association. AHA Data Products. https://www.aha.org/data-insights/aha-data-products. Accessed November 25, 2019.
7. Dummit LA, Kahvecioglu D, Marrufo G, et al. Bundled payment initiative and payments and quality outcomes for lower extremity joint replacement episodes. JAMA. 2016; 316(12):1267-1278. https://doi.org/10.1001/jama.2016.12717.
8. Finkelstein A, Ji Y, Mahoney N, Skinner J. Mandatory medicare bundled payment program for lower extremity joint replacement and discharge to institutional postacute care: Interim analysis of the first year of a 5-year randomized trial. JAMA. 2018;320(9):892-900. https://doi.org/10.1001/jama.2018.12346.
9. Navathe AS, Troxel AB, Liao JM, et al. Cost of joint replacement using bundled payment models. JAMA Intern Med. 2017;177(2):214-222. https://doi.org/10.1001/jamainternmed.2016.8263.
10. Liao JM, Emanuel EJ, Polsky DE, et al. National representativeness of hospitals and markets in Medicare’s mandatory bundled payment program. Health Aff. 2019;38(1):44-53.
11. Barnett ML, Wilcock A, McWilliams JM, et al. Two-year evaluation of mandatory bundled payments for joint replacement. N Engl J Med. 2019;380(3):252-262. https://doi.org/10.1056/NEJMsa1809010.
12. Navathe AS, Liao JM, Polsky D, et al. Comparison of hospitals participating in Medicare’s voluntary and mandatory orthopedic bundle programs. Health Aff. 2018;37(6):854-863. https://www.doi.org/10.1377/hlthaff.2017.1358.
13. Joynt Maddox KE, Orav EJ, Zheng J, Epstein AM. Participation and Dropout in the Bundled Payments for Care Improvement Initiative. JAMA. 2018;319(2):191-193. https://doi.org/10.1001/jama.2017.14771.
14. Navathe AS, Liao JM, Dykstra SE, et al. Association of hospital participation in a Medicare bundled payment program with volume and case mix of lower extremity joint replacement episodes. JAMA. 2018;320(9):901-910. https://doi.org/10.1001/jama.2018.12345.
15. Joynt Maddox KE, Orav EJ, Epstein AM. Medicare’s bundled payments initiative for medical conditions. N Engl J Med. 2018;379(18):e33. https://doi.org/10.1056/NEJMc1811049.
16. Navathe AS, Shan E, Liao JM. What have we learned about bundling medical conditions? Health Affairs Blog. https://www.healthaffairs.org/do/10.1377/hblog20180828.844613/full/. Accessed November 25, 2019.
17. Centers for Medicare & Medicaid Services. MACRA. https://www.cms.gov/medicare/quality-initiatives-patient-assessment-instruments/value-based-programs/macra-mips-and-apms/macra-mips-and-apms.html. Accessed November 26, 2019.
1. Centers for Medicare & Medicaid Services. Fiscal Year 2019 Clinical Episode-Based Payment Measures Overview. https://www.qualityreportingcenter.com/globalassets/migrated-pdf/cepb_slides_npc-6.17.2018_5.22.18_vfinal508.pdf. Accessed November 26, 2019.
2. Centers for Medicare & Medicaid Services. Hospital Inpatient Quality Reporting Program. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/HospitalQualityInits/HospitalRHQDAPU.html. Accessed November 23, 2019.
3. Centers for Medicare & Medicaid Services. Medicare Spending Per Beneficiary (MSPB) Spending Breakdown by Claim Type. https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/hospital-value-based-purchasing/Downloads/Fact-Sheet-MSPB-Spending-Breakdowns-by-Claim-Type-Dec-2014.pdf. Accessed November 25, 2019.
4. Hu J, Jordan J, Rubinfeld I, Schreiber M, Waterman B, Nerenz D. Correlations among hospital quality measure: What “Hospital Compare” data tell us. Am J Med Qual. 2017;32(6):605-610. https://doi.org/10.1177/1062860616684012.
5. Centers for Medicare & Medicaid Services. Hospital Compare datasets. https://data.medicare.gov/data/hospital-compare. Accessed November 26, 2019.
6. American Hospital Association. AHA Data Products. https://www.aha.org/data-insights/aha-data-products. Accessed November 25, 2019.
7. Dummit LA, Kahvecioglu D, Marrufo G, et al. Bundled payment initiative and payments and quality outcomes for lower extremity joint replacement episodes. JAMA. 2016; 316(12):1267-1278. https://doi.org/10.1001/jama.2016.12717.
8. Finkelstein A, Ji Y, Mahoney N, Skinner J. Mandatory medicare bundled payment program for lower extremity joint replacement and discharge to institutional postacute care: Interim analysis of the first year of a 5-year randomized trial. JAMA. 2018;320(9):892-900. https://doi.org/10.1001/jama.2018.12346.
9. Navathe AS, Troxel AB, Liao JM, et al. Cost of joint replacement using bundled payment models. JAMA Intern Med. 2017;177(2):214-222. https://doi.org/10.1001/jamainternmed.2016.8263.
10. Liao JM, Emanuel EJ, Polsky DE, et al. National representativeness of hospitals and markets in Medicare’s mandatory bundled payment program. Health Aff. 2019;38(1):44-53.
11. Barnett ML, Wilcock A, McWilliams JM, et al. Two-year evaluation of mandatory bundled payments for joint replacement. N Engl J Med. 2019;380(3):252-262. https://doi.org/10.1056/NEJMsa1809010.
12. Navathe AS, Liao JM, Polsky D, et al. Comparison of hospitals participating in Medicare’s voluntary and mandatory orthopedic bundle programs. Health Aff. 2018;37(6):854-863. https://www.doi.org/10.1377/hlthaff.2017.1358.
13. Joynt Maddox KE, Orav EJ, Zheng J, Epstein AM. Participation and Dropout in the Bundled Payments for Care Improvement Initiative. JAMA. 2018;319(2):191-193. https://doi.org/10.1001/jama.2017.14771.
14. Navathe AS, Liao JM, Dykstra SE, et al. Association of hospital participation in a Medicare bundled payment program with volume and case mix of lower extremity joint replacement episodes. JAMA. 2018;320(9):901-910. https://doi.org/10.1001/jama.2018.12345.
15. Joynt Maddox KE, Orav EJ, Epstein AM. Medicare’s bundled payments initiative for medical conditions. N Engl J Med. 2018;379(18):e33. https://doi.org/10.1056/NEJMc1811049.
16. Navathe AS, Shan E, Liao JM. What have we learned about bundling medical conditions? Health Affairs Blog. https://www.healthaffairs.org/do/10.1377/hblog20180828.844613/full/. Accessed November 25, 2019.
17. Centers for Medicare & Medicaid Services. MACRA. https://www.cms.gov/medicare/quality-initiatives-patient-assessment-instruments/value-based-programs/macra-mips-and-apms/macra-mips-and-apms.html. Accessed November 26, 2019.
© 2020 Society of Hospital Medicine