User login
FDA, CDC urge pause of J&J COVID vaccine
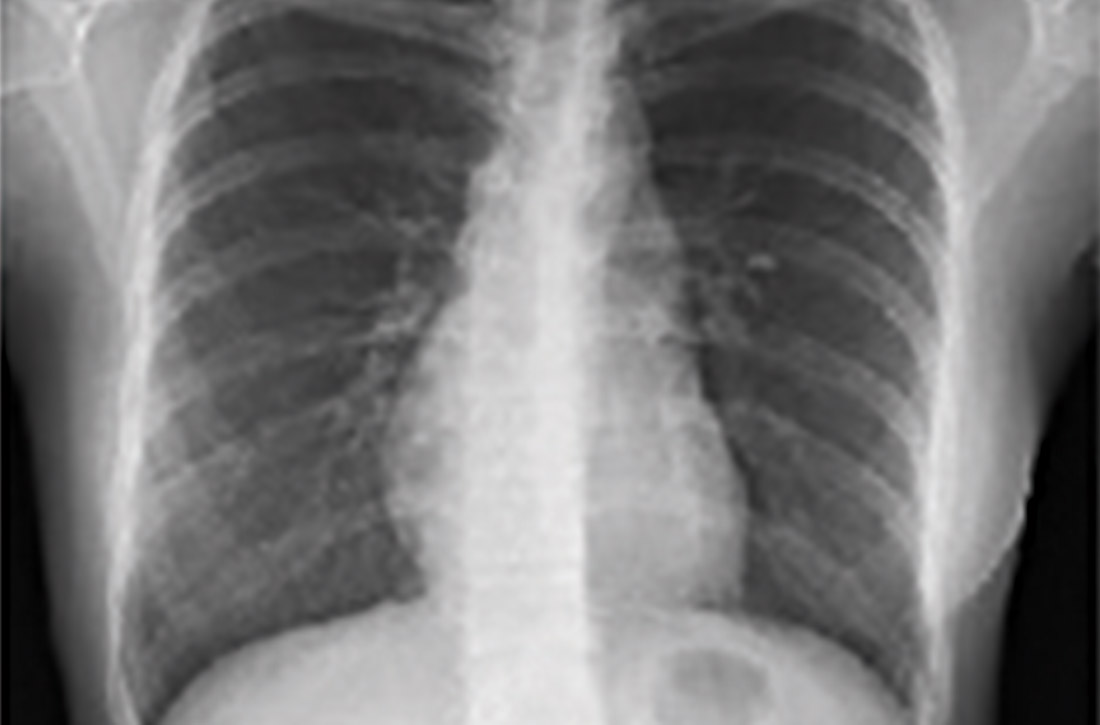
The Food and Drug Administration and Centers for Disease Control and Prevention on April 13 recommended that use of the Johnson & Johnson COVID-19 vaccine be paused after reports of blood clots in patients receiving the shot, the agencies have announced.
In a statement, FDA said 6.8 million doses of the J&J vaccine have been administered and the agency is investigating six reported cases of a rare and severe blood clot occurring in patients who received the vaccine.
The pause is intended to give time to alert the public to this "very rare" condition, experts said during a joint CDC-FDA media briefing April 13.
"It was clear to us that we needed to alert the public," Janet Woodcock, MD, acting FDA commissioner, said. The move also will allow "time for the healthcare community to learn what they need to know about how to diagnose, treat and report" any additional cases.
The CDC will convene a meeting of the Advisory Committee on Immunization Practices on April 14 to review the cases.
"I know the information today will be very concerning to Americans who have already received the Johnson & Johnson vaccine," said Anne Schuchat, MD, principal deputy director at the CDC.
"For people who got the vaccine more than one month ago, the risk is very low at this time," she added. "For people who recently got the vaccine, in the last couple of weeks, look for symptoms."
Headache, leg pain, abdominal pain, and shortness of breath were among the reported symptoms. All six cases arose within 6 to 13 days of receipt of the Johnson & Johnson vaccine.
Traditional treatment dangerous
Importantly, treatment for traditional blood clots, such as the drug heparin, should not be used for these clots. "The issue here with these types of blood clots is that if one administers the standard treatment we give for blood clots, one can cause tremendous harm or it can be fatal," said Peter Marks, MD, director of the FDA Center for Biologics Evaluation and Research.
If health care providers see people with these symptoms along with a low platelet count or blood clots, they should ask about any recent vaccinations, Dr. Marks added.
Headache is a common side effect of COVID-19 vaccination, Dr. Marks said, but it typically happens within a day or two. In contrast, the headaches associated with these blood clots come 1 to 2 weeks later and were very severe.
Not all of the six women involved in the events had a pre-existing condition or risk factor, Dr. Schuchat said.
Severe but 'extremely rare'
To put the numbers in context, the six reported events occurred among millions of people who received the Johnson & Johnson vaccine to date.
"There have been six reports of a severe stroke-like illness due to low platelet count and more than six million doses of the Johnson & Johnson vaccine have been administered so far," Dr. Schuchat said.
"I would like to stress these events are extremely rare," Dr. Woodcock said, "but we take all reports of adverse events after vaccination very seriously."
The company response
Johnson & Johnson in a statement said, "We are aware of an extremely rare disorder involving people with blood clots in combination with low platelets in a small number of individuals who have received our COVID-19 vaccine. The United States Centers for Disease Control (CDC) and Food and Drug Administration (FDA) are reviewing data involving six reported U.S. cases out of more than 6.8 million doses administered. Out of an abundance of caution, the CDC and FDA have recommended a pause in the use of our vaccine."
The company said they are also reviewing these cases with European regulators and "we have made the decision to proactively delay the rollout of our vaccine in Europe."
Overall vaccinations continuing apace
"This announcement will not have a significant impact on our vaccination plan. Johnson & Johnson vaccine makes up less than 5% of the recorded shots in arms in the United States to date," Jeff Zients, White House COVID-19 Response Coordinator, said in a statement.
"Based on actions taken by the president earlier this year, the United States has secured enough Pfizer and Moderna doses for 300 million Americans. We are working now with our state and federal partners to get anyone scheduled for a J&J vaccine quickly rescheduled for a Pfizer or Moderna vaccine," he added.
The likely duration of the pause remains unclear.
"I know this has been a long and difficult pandemic, and people are tired of the steps they have to take," Dr. Schuchat said. "Steps taken today make sure the health care system is ready to diagnose, treat and report [any additional cases] and the public has the information necessary to stay safe."
A version of this article first appeared on WebMD.com.
This article was updated 4/13/21.
The Food and Drug Administration and Centers for Disease Control and Prevention on April 13 recommended that use of the Johnson & Johnson COVID-19 vaccine be paused after reports of blood clots in patients receiving the shot, the agencies have announced.
In a statement, FDA said 6.8 million doses of the J&J vaccine have been administered and the agency is investigating six reported cases of a rare and severe blood clot occurring in patients who received the vaccine.
The pause is intended to give time to alert the public to this "very rare" condition, experts said during a joint CDC-FDA media briefing April 13.
"It was clear to us that we needed to alert the public," Janet Woodcock, MD, acting FDA commissioner, said. The move also will allow "time for the healthcare community to learn what they need to know about how to diagnose, treat and report" any additional cases.
The CDC will convene a meeting of the Advisory Committee on Immunization Practices on April 14 to review the cases.
"I know the information today will be very concerning to Americans who have already received the Johnson & Johnson vaccine," said Anne Schuchat, MD, principal deputy director at the CDC.
"For people who got the vaccine more than one month ago, the risk is very low at this time," she added. "For people who recently got the vaccine, in the last couple of weeks, look for symptoms."
Headache, leg pain, abdominal pain, and shortness of breath were among the reported symptoms. All six cases arose within 6 to 13 days of receipt of the Johnson & Johnson vaccine.
Traditional treatment dangerous
Importantly, treatment for traditional blood clots, such as the drug heparin, should not be used for these clots. "The issue here with these types of blood clots is that if one administers the standard treatment we give for blood clots, one can cause tremendous harm or it can be fatal," said Peter Marks, MD, director of the FDA Center for Biologics Evaluation and Research.
If health care providers see people with these symptoms along with a low platelet count or blood clots, they should ask about any recent vaccinations, Dr. Marks added.
Headache is a common side effect of COVID-19 vaccination, Dr. Marks said, but it typically happens within a day or two. In contrast, the headaches associated with these blood clots come 1 to 2 weeks later and were very severe.
Not all of the six women involved in the events had a pre-existing condition or risk factor, Dr. Schuchat said.
Severe but 'extremely rare'
To put the numbers in context, the six reported events occurred among millions of people who received the Johnson & Johnson vaccine to date.
"There have been six reports of a severe stroke-like illness due to low platelet count and more than six million doses of the Johnson & Johnson vaccine have been administered so far," Dr. Schuchat said.
"I would like to stress these events are extremely rare," Dr. Woodcock said, "but we take all reports of adverse events after vaccination very seriously."
The company response
Johnson & Johnson in a statement said, "We are aware of an extremely rare disorder involving people with blood clots in combination with low platelets in a small number of individuals who have received our COVID-19 vaccine. The United States Centers for Disease Control (CDC) and Food and Drug Administration (FDA) are reviewing data involving six reported U.S. cases out of more than 6.8 million doses administered. Out of an abundance of caution, the CDC and FDA have recommended a pause in the use of our vaccine."
The company said they are also reviewing these cases with European regulators and "we have made the decision to proactively delay the rollout of our vaccine in Europe."
Overall vaccinations continuing apace
"This announcement will not have a significant impact on our vaccination plan. Johnson & Johnson vaccine makes up less than 5% of the recorded shots in arms in the United States to date," Jeff Zients, White House COVID-19 Response Coordinator, said in a statement.
"Based on actions taken by the president earlier this year, the United States has secured enough Pfizer and Moderna doses for 300 million Americans. We are working now with our state and federal partners to get anyone scheduled for a J&J vaccine quickly rescheduled for a Pfizer or Moderna vaccine," he added.
The likely duration of the pause remains unclear.
"I know this has been a long and difficult pandemic, and people are tired of the steps they have to take," Dr. Schuchat said. "Steps taken today make sure the health care system is ready to diagnose, treat and report [any additional cases] and the public has the information necessary to stay safe."
A version of this article first appeared on WebMD.com.
This article was updated 4/13/21.
The Food and Drug Administration and Centers for Disease Control and Prevention on April 13 recommended that use of the Johnson & Johnson COVID-19 vaccine be paused after reports of blood clots in patients receiving the shot, the agencies have announced.
In a statement, FDA said 6.8 million doses of the J&J vaccine have been administered and the agency is investigating six reported cases of a rare and severe blood clot occurring in patients who received the vaccine.
The pause is intended to give time to alert the public to this "very rare" condition, experts said during a joint CDC-FDA media briefing April 13.
"It was clear to us that we needed to alert the public," Janet Woodcock, MD, acting FDA commissioner, said. The move also will allow "time for the healthcare community to learn what they need to know about how to diagnose, treat and report" any additional cases.
The CDC will convene a meeting of the Advisory Committee on Immunization Practices on April 14 to review the cases.
"I know the information today will be very concerning to Americans who have already received the Johnson & Johnson vaccine," said Anne Schuchat, MD, principal deputy director at the CDC.
"For people who got the vaccine more than one month ago, the risk is very low at this time," she added. "For people who recently got the vaccine, in the last couple of weeks, look for symptoms."
Headache, leg pain, abdominal pain, and shortness of breath were among the reported symptoms. All six cases arose within 6 to 13 days of receipt of the Johnson & Johnson vaccine.
Traditional treatment dangerous
Importantly, treatment for traditional blood clots, such as the drug heparin, should not be used for these clots. "The issue here with these types of blood clots is that if one administers the standard treatment we give for blood clots, one can cause tremendous harm or it can be fatal," said Peter Marks, MD, director of the FDA Center for Biologics Evaluation and Research.
If health care providers see people with these symptoms along with a low platelet count or blood clots, they should ask about any recent vaccinations, Dr. Marks added.
Headache is a common side effect of COVID-19 vaccination, Dr. Marks said, but it typically happens within a day or two. In contrast, the headaches associated with these blood clots come 1 to 2 weeks later and were very severe.
Not all of the six women involved in the events had a pre-existing condition or risk factor, Dr. Schuchat said.
Severe but 'extremely rare'
To put the numbers in context, the six reported events occurred among millions of people who received the Johnson & Johnson vaccine to date.
"There have been six reports of a severe stroke-like illness due to low platelet count and more than six million doses of the Johnson & Johnson vaccine have been administered so far," Dr. Schuchat said.
"I would like to stress these events are extremely rare," Dr. Woodcock said, "but we take all reports of adverse events after vaccination very seriously."
The company response
Johnson & Johnson in a statement said, "We are aware of an extremely rare disorder involving people with blood clots in combination with low platelets in a small number of individuals who have received our COVID-19 vaccine. The United States Centers for Disease Control (CDC) and Food and Drug Administration (FDA) are reviewing data involving six reported U.S. cases out of more than 6.8 million doses administered. Out of an abundance of caution, the CDC and FDA have recommended a pause in the use of our vaccine."
The company said they are also reviewing these cases with European regulators and "we have made the decision to proactively delay the rollout of our vaccine in Europe."
Overall vaccinations continuing apace
"This announcement will not have a significant impact on our vaccination plan. Johnson & Johnson vaccine makes up less than 5% of the recorded shots in arms in the United States to date," Jeff Zients, White House COVID-19 Response Coordinator, said in a statement.
"Based on actions taken by the president earlier this year, the United States has secured enough Pfizer and Moderna doses for 300 million Americans. We are working now with our state and federal partners to get anyone scheduled for a J&J vaccine quickly rescheduled for a Pfizer or Moderna vaccine," he added.
The likely duration of the pause remains unclear.
"I know this has been a long and difficult pandemic, and people are tired of the steps they have to take," Dr. Schuchat said. "Steps taken today make sure the health care system is ready to diagnose, treat and report [any additional cases] and the public has the information necessary to stay safe."
A version of this article first appeared on WebMD.com.
This article was updated 4/13/21.
OCS heart system earns hard-won backing of FDA panel
After more than 10 hours of intense debate, a Food and Drug Administration advisory panel gave its support to a premarket approval application (PMA) for the TransMedics Organ Care System (OCS) Heart system.
The OCS Heart is a portable extracorporeal perfusion and monitoring system designed to keep a donor heart in a normothermic, beating state. The “heart in a box” technology allows donor hearts to be transported across longer distances than is possible with standard cold storage, which can safely preserve donor hearts for about 4 hours.
The Circulatory System Devices Panel of the Medical Devices Advisory Committee voted 12 to 5, with 1 abstention, that the benefits of the OCS Heart System outweigh its risks.
The panel voted in favor of the OCS Heart being effective (10 yes, 6 no, and 2 abstaining) and safe (9 yes, 7 no, 2 abstaining) but not without mixed feelings.
James Blankenship, MD, a cardiologist at the University of New Mexico, Albuquerque, voted yes to all three questions but said: “If it had been compared to standard of care, I would have voted no to all three. But if it’s compared to getting an [left ventricular assist device] LVAD or not getting a heart at all, I would say the benefits outweigh the risks.”
Marc R. Katz, MD, chief of cardiothoracic surgery, Medical University of South Carolina, Charleston, also gave universal support, noting that the rate of heart transplantations has been flat for years. “This is a big step forward toward being able to expand that number. Now all that said, it obviously was a less-than-perfect study and I do think there needs to be some constraints put on the utilization.”
The panel reviewed data from the single-arm OCS Heart EXPAND trial and associated EXPAND Continued Access Protocol (CAP), as well the sponsor’s first OCS Heart trial, PROCEED II.
EXPAND met its effectiveness endpoint, with 88% of donor hearts successfully transplanted, an 8% incidence of severe primary graft dysfunction (PGD) 24 hours after transplantation, and 94.6% survival at 30 days.
Data from 41 patients with 30-day follow-up in the ongoing EXPAND CAP show 91% of donor hearts were utilized, a 2.4% incidence of severe PGD, and 100% 30-day survival.
The sponsor and the FDA clashed over changes made to the trial after the PMA was submitted, the appropriateness of the effectiveness outcome, and claims by the FDA that there was substantial overlap in demographic characteristics between the extended criteria donor hearts in the EXPAND trials and the standard criteria donor hearts in PROCEED II.
TransMedics previously submitted a PMA based on PROCEED II but it noted in submitted documents that it was withdrawn because of “fundamental disagreements with FDA” on the interpretation of a post hoc analysis with United Network for Organ Sharing registry data that identified increased all-cause mortality risk but comparable cardiac-related mortality in patients with OCS hearts.
During the marathon hearing, FDA officials presented several post hoc analyses, including one stratified by donor inclusion criteria, in which 30-day survival estimates were worse in recipients of single-criterion organs than for those receiving donor organs with multiple inclusion criteria (85% vs. 91.4%). In a second analysis, 2-year point estimates of survival also trended lower with donor organs having only one extended criterion.
Reported EXPAND CAP 6- and 12-month survival estimates were 100% and 93%, respectively, which was higher than EXPAND (93% and 84%), but there was substantial censoring (>50%) at 6 months and beyond, FDA officials said.
When EXPAND and CAP data were pooled, modeled survival curves shifted upward but there was a substantial site effect, with a single site contributing 46% of data, which may affect generalizability of the results, they noted.
“I voted yes for safety, no for efficacy, and no for approval and I’d just like to say I found this to be the most difficult vote in my experience on this panel,” John Hirshfeld, MD, University of Pennsylvania, Philadelphia, said. “I was very concerned that the PROCEED data suggests a possible harm, and in the absence of an interpretable comparator for the EXPAND trial, it’s really not possible to decide if there’s efficacy.”
Keith B. Allen, MD, director of surgical research at Saint Luke’s Hospital of Kansas City (Mo.), said, “I voted no on safety; I’m not going to give the company a pass. I think their animal data was sorely lacking and a lot of issues over the last 10 years could have been addressed with some key animal studies.
“For efficacy and risk/benefit, I voted yes for both,” he said. “Had this been standard of care and only PROCEED II, I would have voted no, but I do think there are a lot of hearts that go in the bucket and this is a challenging population.”
More than a dozen physicians and patients spoke at the open public hearing about the potential for the device to expand donor heart utilization, including a recipient whose own father died while waiting on the transplant list. Only about 3 out of every 10 donated hearts are used for transplant. To ensure fair access, particularly for patients in rural areas, federal changes in 2020 mandate that organs be allocated to the sickest patients first.
Data showed that the OCS Heart System was associated with shorter waiting list times, compared with U.S. averages but longer preservation times than cold static preservation.
In all, 13% of accepted donor organs were subsequently turned down after OCS heart preservation. Lactate levels were cited as the principal reason for turn-down but, FDA officials said, the validity of using lactate as a marker for transplantability is unclear.
Pathologic analysis of OCS Heart turned-down donor hearts with stable antemortem hemodynamics, normal or near-normal anatomy and normal ventricular function by echocardiography, and autopsy findings of acute diffuse or multifocal myocardial damage “suggest that in an important proportion of cases the OCS Heart system did not provide effective organ preservation or its use caused severe myocardial damage to what might have been an acceptable graft for transplant,” said Andrew Farb, MD, chief medical officer of the FDA’s Office of Cardiovascular Devices.
Proposed indication
In the present PMA, the OCS Heart System is indicated for donor hearts with one or more of the following characteristics: an expected cross-clamp or ischemic time of at least 4 hours because of donor or recipient characteristics; or an expected total cross-clamp time of at least 2 hours plus one of the following risk factors: donor age 55 or older, history of cardiac arrest and downtime of at least 20 minutes, history of alcoholism, history of diabetes, donor ejection fraction of 40%-50%,history of left ventricular hypertrophy, and donor angiogram with luminal irregularities but no significant coronary artery disease
Several members voiced concern about “indication creep” should the device be approved by the FDA, and highlighted the 2-hour cross-clamp time plus wide-ranging risk factors.
“I’m a surgeon and I voted no on all three counts,” said Murray H. Kwon, MD, Ronald Reagan University of California, Los Angeles Medical Center. “As far as risk/benefit, if it was just limited to one group – the 4-hour plus – I would say yes, but if you’re going to tell me that there’s a risk/benefit for the 2-hour with the alcoholic, I don’t know how that was proved in anything.”
Dr. Kwon was also troubled by lack of proper controls and by the one quarter of patients who ended up on mechanical circulatory support in the first 30 days after transplant. “I find that highly aberrant.”
Joaquin E. Cigarroa, MD, head of cardiovascular medicine, Oregon Health & Science University, Portland, said the unmet need for patients with refractory, end-stage heart failure is challenging and quite emotional, but also voted no across the board, citing concerns about a lack of comparator in the EXPAND trials and overall out-of-body ischemic time.
“As it relates to risk/benefit, I thought long and hard about voting yes despite all the unknowns because of this emotion, but ultimately I voted no because of the secondary 2-hours plus alcoholism, diabetes, or minor coronary disease, in which the ischemic burden and ongoing lactate production concern me,” he said.
Although the panel decision is nonbinding, there was strong support from the committee members for a randomized, postapproval trial and more complete animal studies.
A version of this article first appeared on Medscape.com.
After more than 10 hours of intense debate, a Food and Drug Administration advisory panel gave its support to a premarket approval application (PMA) for the TransMedics Organ Care System (OCS) Heart system.
The OCS Heart is a portable extracorporeal perfusion and monitoring system designed to keep a donor heart in a normothermic, beating state. The “heart in a box” technology allows donor hearts to be transported across longer distances than is possible with standard cold storage, which can safely preserve donor hearts for about 4 hours.
The Circulatory System Devices Panel of the Medical Devices Advisory Committee voted 12 to 5, with 1 abstention, that the benefits of the OCS Heart System outweigh its risks.
The panel voted in favor of the OCS Heart being effective (10 yes, 6 no, and 2 abstaining) and safe (9 yes, 7 no, 2 abstaining) but not without mixed feelings.
James Blankenship, MD, a cardiologist at the University of New Mexico, Albuquerque, voted yes to all three questions but said: “If it had been compared to standard of care, I would have voted no to all three. But if it’s compared to getting an [left ventricular assist device] LVAD or not getting a heart at all, I would say the benefits outweigh the risks.”
Marc R. Katz, MD, chief of cardiothoracic surgery, Medical University of South Carolina, Charleston, also gave universal support, noting that the rate of heart transplantations has been flat for years. “This is a big step forward toward being able to expand that number. Now all that said, it obviously was a less-than-perfect study and I do think there needs to be some constraints put on the utilization.”
The panel reviewed data from the single-arm OCS Heart EXPAND trial and associated EXPAND Continued Access Protocol (CAP), as well the sponsor’s first OCS Heart trial, PROCEED II.
EXPAND met its effectiveness endpoint, with 88% of donor hearts successfully transplanted, an 8% incidence of severe primary graft dysfunction (PGD) 24 hours after transplantation, and 94.6% survival at 30 days.
Data from 41 patients with 30-day follow-up in the ongoing EXPAND CAP show 91% of donor hearts were utilized, a 2.4% incidence of severe PGD, and 100% 30-day survival.
The sponsor and the FDA clashed over changes made to the trial after the PMA was submitted, the appropriateness of the effectiveness outcome, and claims by the FDA that there was substantial overlap in demographic characteristics between the extended criteria donor hearts in the EXPAND trials and the standard criteria donor hearts in PROCEED II.
TransMedics previously submitted a PMA based on PROCEED II but it noted in submitted documents that it was withdrawn because of “fundamental disagreements with FDA” on the interpretation of a post hoc analysis with United Network for Organ Sharing registry data that identified increased all-cause mortality risk but comparable cardiac-related mortality in patients with OCS hearts.
During the marathon hearing, FDA officials presented several post hoc analyses, including one stratified by donor inclusion criteria, in which 30-day survival estimates were worse in recipients of single-criterion organs than for those receiving donor organs with multiple inclusion criteria (85% vs. 91.4%). In a second analysis, 2-year point estimates of survival also trended lower with donor organs having only one extended criterion.
Reported EXPAND CAP 6- and 12-month survival estimates were 100% and 93%, respectively, which was higher than EXPAND (93% and 84%), but there was substantial censoring (>50%) at 6 months and beyond, FDA officials said.
When EXPAND and CAP data were pooled, modeled survival curves shifted upward but there was a substantial site effect, with a single site contributing 46% of data, which may affect generalizability of the results, they noted.
“I voted yes for safety, no for efficacy, and no for approval and I’d just like to say I found this to be the most difficult vote in my experience on this panel,” John Hirshfeld, MD, University of Pennsylvania, Philadelphia, said. “I was very concerned that the PROCEED data suggests a possible harm, and in the absence of an interpretable comparator for the EXPAND trial, it’s really not possible to decide if there’s efficacy.”
Keith B. Allen, MD, director of surgical research at Saint Luke’s Hospital of Kansas City (Mo.), said, “I voted no on safety; I’m not going to give the company a pass. I think their animal data was sorely lacking and a lot of issues over the last 10 years could have been addressed with some key animal studies.
“For efficacy and risk/benefit, I voted yes for both,” he said. “Had this been standard of care and only PROCEED II, I would have voted no, but I do think there are a lot of hearts that go in the bucket and this is a challenging population.”
More than a dozen physicians and patients spoke at the open public hearing about the potential for the device to expand donor heart utilization, including a recipient whose own father died while waiting on the transplant list. Only about 3 out of every 10 donated hearts are used for transplant. To ensure fair access, particularly for patients in rural areas, federal changes in 2020 mandate that organs be allocated to the sickest patients first.
Data showed that the OCS Heart System was associated with shorter waiting list times, compared with U.S. averages but longer preservation times than cold static preservation.
In all, 13% of accepted donor organs were subsequently turned down after OCS heart preservation. Lactate levels were cited as the principal reason for turn-down but, FDA officials said, the validity of using lactate as a marker for transplantability is unclear.
Pathologic analysis of OCS Heart turned-down donor hearts with stable antemortem hemodynamics, normal or near-normal anatomy and normal ventricular function by echocardiography, and autopsy findings of acute diffuse or multifocal myocardial damage “suggest that in an important proportion of cases the OCS Heart system did not provide effective organ preservation or its use caused severe myocardial damage to what might have been an acceptable graft for transplant,” said Andrew Farb, MD, chief medical officer of the FDA’s Office of Cardiovascular Devices.
Proposed indication
In the present PMA, the OCS Heart System is indicated for donor hearts with one or more of the following characteristics: an expected cross-clamp or ischemic time of at least 4 hours because of donor or recipient characteristics; or an expected total cross-clamp time of at least 2 hours plus one of the following risk factors: donor age 55 or older, history of cardiac arrest and downtime of at least 20 minutes, history of alcoholism, history of diabetes, donor ejection fraction of 40%-50%,history of left ventricular hypertrophy, and donor angiogram with luminal irregularities but no significant coronary artery disease
Several members voiced concern about “indication creep” should the device be approved by the FDA, and highlighted the 2-hour cross-clamp time plus wide-ranging risk factors.
“I’m a surgeon and I voted no on all three counts,” said Murray H. Kwon, MD, Ronald Reagan University of California, Los Angeles Medical Center. “As far as risk/benefit, if it was just limited to one group – the 4-hour plus – I would say yes, but if you’re going to tell me that there’s a risk/benefit for the 2-hour with the alcoholic, I don’t know how that was proved in anything.”
Dr. Kwon was also troubled by lack of proper controls and by the one quarter of patients who ended up on mechanical circulatory support in the first 30 days after transplant. “I find that highly aberrant.”
Joaquin E. Cigarroa, MD, head of cardiovascular medicine, Oregon Health & Science University, Portland, said the unmet need for patients with refractory, end-stage heart failure is challenging and quite emotional, but also voted no across the board, citing concerns about a lack of comparator in the EXPAND trials and overall out-of-body ischemic time.
“As it relates to risk/benefit, I thought long and hard about voting yes despite all the unknowns because of this emotion, but ultimately I voted no because of the secondary 2-hours plus alcoholism, diabetes, or minor coronary disease, in which the ischemic burden and ongoing lactate production concern me,” he said.
Although the panel decision is nonbinding, there was strong support from the committee members for a randomized, postapproval trial and more complete animal studies.
A version of this article first appeared on Medscape.com.
After more than 10 hours of intense debate, a Food and Drug Administration advisory panel gave its support to a premarket approval application (PMA) for the TransMedics Organ Care System (OCS) Heart system.
The OCS Heart is a portable extracorporeal perfusion and monitoring system designed to keep a donor heart in a normothermic, beating state. The “heart in a box” technology allows donor hearts to be transported across longer distances than is possible with standard cold storage, which can safely preserve donor hearts for about 4 hours.
The Circulatory System Devices Panel of the Medical Devices Advisory Committee voted 12 to 5, with 1 abstention, that the benefits of the OCS Heart System outweigh its risks.
The panel voted in favor of the OCS Heart being effective (10 yes, 6 no, and 2 abstaining) and safe (9 yes, 7 no, 2 abstaining) but not without mixed feelings.
James Blankenship, MD, a cardiologist at the University of New Mexico, Albuquerque, voted yes to all three questions but said: “If it had been compared to standard of care, I would have voted no to all three. But if it’s compared to getting an [left ventricular assist device] LVAD or not getting a heart at all, I would say the benefits outweigh the risks.”
Marc R. Katz, MD, chief of cardiothoracic surgery, Medical University of South Carolina, Charleston, also gave universal support, noting that the rate of heart transplantations has been flat for years. “This is a big step forward toward being able to expand that number. Now all that said, it obviously was a less-than-perfect study and I do think there needs to be some constraints put on the utilization.”
The panel reviewed data from the single-arm OCS Heart EXPAND trial and associated EXPAND Continued Access Protocol (CAP), as well the sponsor’s first OCS Heart trial, PROCEED II.
EXPAND met its effectiveness endpoint, with 88% of donor hearts successfully transplanted, an 8% incidence of severe primary graft dysfunction (PGD) 24 hours after transplantation, and 94.6% survival at 30 days.
Data from 41 patients with 30-day follow-up in the ongoing EXPAND CAP show 91% of donor hearts were utilized, a 2.4% incidence of severe PGD, and 100% 30-day survival.
The sponsor and the FDA clashed over changes made to the trial after the PMA was submitted, the appropriateness of the effectiveness outcome, and claims by the FDA that there was substantial overlap in demographic characteristics between the extended criteria donor hearts in the EXPAND trials and the standard criteria donor hearts in PROCEED II.
TransMedics previously submitted a PMA based on PROCEED II but it noted in submitted documents that it was withdrawn because of “fundamental disagreements with FDA” on the interpretation of a post hoc analysis with United Network for Organ Sharing registry data that identified increased all-cause mortality risk but comparable cardiac-related mortality in patients with OCS hearts.
During the marathon hearing, FDA officials presented several post hoc analyses, including one stratified by donor inclusion criteria, in which 30-day survival estimates were worse in recipients of single-criterion organs than for those receiving donor organs with multiple inclusion criteria (85% vs. 91.4%). In a second analysis, 2-year point estimates of survival also trended lower with donor organs having only one extended criterion.
Reported EXPAND CAP 6- and 12-month survival estimates were 100% and 93%, respectively, which was higher than EXPAND (93% and 84%), but there was substantial censoring (>50%) at 6 months and beyond, FDA officials said.
When EXPAND and CAP data were pooled, modeled survival curves shifted upward but there was a substantial site effect, with a single site contributing 46% of data, which may affect generalizability of the results, they noted.
“I voted yes for safety, no for efficacy, and no for approval and I’d just like to say I found this to be the most difficult vote in my experience on this panel,” John Hirshfeld, MD, University of Pennsylvania, Philadelphia, said. “I was very concerned that the PROCEED data suggests a possible harm, and in the absence of an interpretable comparator for the EXPAND trial, it’s really not possible to decide if there’s efficacy.”
Keith B. Allen, MD, director of surgical research at Saint Luke’s Hospital of Kansas City (Mo.), said, “I voted no on safety; I’m not going to give the company a pass. I think their animal data was sorely lacking and a lot of issues over the last 10 years could have been addressed with some key animal studies.
“For efficacy and risk/benefit, I voted yes for both,” he said. “Had this been standard of care and only PROCEED II, I would have voted no, but I do think there are a lot of hearts that go in the bucket and this is a challenging population.”
More than a dozen physicians and patients spoke at the open public hearing about the potential for the device to expand donor heart utilization, including a recipient whose own father died while waiting on the transplant list. Only about 3 out of every 10 donated hearts are used for transplant. To ensure fair access, particularly for patients in rural areas, federal changes in 2020 mandate that organs be allocated to the sickest patients first.
Data showed that the OCS Heart System was associated with shorter waiting list times, compared with U.S. averages but longer preservation times than cold static preservation.
In all, 13% of accepted donor organs were subsequently turned down after OCS heart preservation. Lactate levels were cited as the principal reason for turn-down but, FDA officials said, the validity of using lactate as a marker for transplantability is unclear.
Pathologic analysis of OCS Heart turned-down donor hearts with stable antemortem hemodynamics, normal or near-normal anatomy and normal ventricular function by echocardiography, and autopsy findings of acute diffuse or multifocal myocardial damage “suggest that in an important proportion of cases the OCS Heart system did not provide effective organ preservation or its use caused severe myocardial damage to what might have been an acceptable graft for transplant,” said Andrew Farb, MD, chief medical officer of the FDA’s Office of Cardiovascular Devices.
Proposed indication
In the present PMA, the OCS Heart System is indicated for donor hearts with one or more of the following characteristics: an expected cross-clamp or ischemic time of at least 4 hours because of donor or recipient characteristics; or an expected total cross-clamp time of at least 2 hours plus one of the following risk factors: donor age 55 or older, history of cardiac arrest and downtime of at least 20 minutes, history of alcoholism, history of diabetes, donor ejection fraction of 40%-50%,history of left ventricular hypertrophy, and donor angiogram with luminal irregularities but no significant coronary artery disease
Several members voiced concern about “indication creep” should the device be approved by the FDA, and highlighted the 2-hour cross-clamp time plus wide-ranging risk factors.
“I’m a surgeon and I voted no on all three counts,” said Murray H. Kwon, MD, Ronald Reagan University of California, Los Angeles Medical Center. “As far as risk/benefit, if it was just limited to one group – the 4-hour plus – I would say yes, but if you’re going to tell me that there’s a risk/benefit for the 2-hour with the alcoholic, I don’t know how that was proved in anything.”
Dr. Kwon was also troubled by lack of proper controls and by the one quarter of patients who ended up on mechanical circulatory support in the first 30 days after transplant. “I find that highly aberrant.”
Joaquin E. Cigarroa, MD, head of cardiovascular medicine, Oregon Health & Science University, Portland, said the unmet need for patients with refractory, end-stage heart failure is challenging and quite emotional, but also voted no across the board, citing concerns about a lack of comparator in the EXPAND trials and overall out-of-body ischemic time.
“As it relates to risk/benefit, I thought long and hard about voting yes despite all the unknowns because of this emotion, but ultimately I voted no because of the secondary 2-hours plus alcoholism, diabetes, or minor coronary disease, in which the ischemic burden and ongoing lactate production concern me,” he said.
Although the panel decision is nonbinding, there was strong support from the committee members for a randomized, postapproval trial and more complete animal studies.
A version of this article first appeared on Medscape.com.
Secukinumab brings high PASI 75 results in 6- to 17-year-olds with psoriasis
at 24 weeks of follow-up in an ongoing 4-year phase 2 clinical trial, Adam Reich, MD, PhD, reported at Innovations in Dermatology: Virtual Spring Conference 2021.
Secukinumab (Cosentyx), a fully human monoclonal antibody that inhibits interleukin-17A, is widely approved for treatment of psoriasis in adults. In August 2020, the biologic received an expanded indication in Europe for treatment of 6- to 17-year-olds. Two phase 3 clinical trials are underway in an effort to gain a similar broadened indication in the United States to help address the high unmet need for new treatments for psoriasis in the pediatric population, said Dr. Reich, professor and head of the department of dermatology at the University of Rzeszow (Poland).
He reported on 84 pediatric patients participating in the open-label, phase 2, international study. They were randomized to one of two weight-based dosing regimens. Those in the low-dose arm received secukinumab dosed at 75 mg if they weighed less than 50 kg and 150 mg if they weighed more. In the high-dose arm, patients got secukinumab 75 mg if they weighed less than 25 kg, 150 mg if they weighed 25-50 kg, and 300 mg if they tipped the scales in excess of 50 kg.
The primary endpoint in the study was the week-12 rate of at least a 75% improvement from baseline in the Psoriasis Area and Severity Index score, or PASI 75. The rates were similar: 92.9% of patients in the high-dose arm achieved this endpoint, as did 90.5% in the low-dose arm. The PASI 90 rates were 83.3% and 78%, the PASI 100 rates were 61.9% and 54.8%, and clear or almost clear skin, as measured by the Investigator Global Assessment, was achieved in 88.7% of the high- and 85.7% of the low-dose groups. In addition,61.9% of those in the high-dose secukinumab group and 50% in the low-dose group had a score of 0 or 1 on the Children’s Dermatology Life Quality Index – indicating psoriasis has no impact on daily quality of life, he said at the conference sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
At week 24, roughly 95% of patients in both the low- and high-dose secukinumab groups had achieved PASI 75s, 88% reached a PASI 90 response, and 67% were at PASI 100. Nearly 60% of the low-dose and 70% of the high-dose groups had a score of 0 or 1 on the Children’s Dermatology Life Quality Index.
Treatment-emergent adverse event rates were similar in the two study arms. Of note, there was one case of new-onset inflammatory bowel disease in the high-dose group, and one case of vulvovaginal candidiasis as well.
Discussant Bruce E. Strober, MD, PhD, said that, if secukinumab gets a pediatric indication from the Food and Drug Administration, as seems likely, it won’t alter his biologic treatment hierarchy.
“I treat a lot of kids with psoriasis. We have three approved drugs now in etanercept [Enbrel], ustekinumab [Stelara], and ixekizumab [Taltz]. My bias is still towards ustekinumab because it’s infrequently dosed and that’s a huge issue for children. You want to expose them to as few injections as possible, for obvious reasons: It’s easier for parents and other caregivers,” explained Dr. Strober, a dermatologist at Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn.
“The other issue is in IL-17 inhibition there has been a slight signal of inflammatory bowel disease popping up in children getting these drugs, and therefore you need to screen patients in this age group very carefully – not only the patients themselves, but their family – for IBD risk. If there is any sign of that I would move the IL-17 inhibitors to the back of the line, compared to ustekinumab and etanercept. Ustekinumab is still clearly the one that I think has to be used first line,” he said.
Dr. Strober offered a final word of advice for his colleagues: “You can’t be afraid to treat children with biologic therapies. In fact, preferentially I would use a biologic therapy over methotrexate or light therapy, which is really difficult for children.”
Dr. Reich and Dr. Strober reported receiving research grants from and serving as a consultant to numerous pharmaceutical companies, including Novartis, which markets secukinumab and funded the study.
MedscapeLIVE! and this news organization are owned by the same parent company.
at 24 weeks of follow-up in an ongoing 4-year phase 2 clinical trial, Adam Reich, MD, PhD, reported at Innovations in Dermatology: Virtual Spring Conference 2021.
Secukinumab (Cosentyx), a fully human monoclonal antibody that inhibits interleukin-17A, is widely approved for treatment of psoriasis in adults. In August 2020, the biologic received an expanded indication in Europe for treatment of 6- to 17-year-olds. Two phase 3 clinical trials are underway in an effort to gain a similar broadened indication in the United States to help address the high unmet need for new treatments for psoriasis in the pediatric population, said Dr. Reich, professor and head of the department of dermatology at the University of Rzeszow (Poland).
He reported on 84 pediatric patients participating in the open-label, phase 2, international study. They were randomized to one of two weight-based dosing regimens. Those in the low-dose arm received secukinumab dosed at 75 mg if they weighed less than 50 kg and 150 mg if they weighed more. In the high-dose arm, patients got secukinumab 75 mg if they weighed less than 25 kg, 150 mg if they weighed 25-50 kg, and 300 mg if they tipped the scales in excess of 50 kg.
The primary endpoint in the study was the week-12 rate of at least a 75% improvement from baseline in the Psoriasis Area and Severity Index score, or PASI 75. The rates were similar: 92.9% of patients in the high-dose arm achieved this endpoint, as did 90.5% in the low-dose arm. The PASI 90 rates were 83.3% and 78%, the PASI 100 rates were 61.9% and 54.8%, and clear or almost clear skin, as measured by the Investigator Global Assessment, was achieved in 88.7% of the high- and 85.7% of the low-dose groups. In addition,61.9% of those in the high-dose secukinumab group and 50% in the low-dose group had a score of 0 or 1 on the Children’s Dermatology Life Quality Index – indicating psoriasis has no impact on daily quality of life, he said at the conference sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
At week 24, roughly 95% of patients in both the low- and high-dose secukinumab groups had achieved PASI 75s, 88% reached a PASI 90 response, and 67% were at PASI 100. Nearly 60% of the low-dose and 70% of the high-dose groups had a score of 0 or 1 on the Children’s Dermatology Life Quality Index.
Treatment-emergent adverse event rates were similar in the two study arms. Of note, there was one case of new-onset inflammatory bowel disease in the high-dose group, and one case of vulvovaginal candidiasis as well.
Discussant Bruce E. Strober, MD, PhD, said that, if secukinumab gets a pediatric indication from the Food and Drug Administration, as seems likely, it won’t alter his biologic treatment hierarchy.
“I treat a lot of kids with psoriasis. We have three approved drugs now in etanercept [Enbrel], ustekinumab [Stelara], and ixekizumab [Taltz]. My bias is still towards ustekinumab because it’s infrequently dosed and that’s a huge issue for children. You want to expose them to as few injections as possible, for obvious reasons: It’s easier for parents and other caregivers,” explained Dr. Strober, a dermatologist at Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn.
“The other issue is in IL-17 inhibition there has been a slight signal of inflammatory bowel disease popping up in children getting these drugs, and therefore you need to screen patients in this age group very carefully – not only the patients themselves, but their family – for IBD risk. If there is any sign of that I would move the IL-17 inhibitors to the back of the line, compared to ustekinumab and etanercept. Ustekinumab is still clearly the one that I think has to be used first line,” he said.
Dr. Strober offered a final word of advice for his colleagues: “You can’t be afraid to treat children with biologic therapies. In fact, preferentially I would use a biologic therapy over methotrexate or light therapy, which is really difficult for children.”
Dr. Reich and Dr. Strober reported receiving research grants from and serving as a consultant to numerous pharmaceutical companies, including Novartis, which markets secukinumab and funded the study.
MedscapeLIVE! and this news organization are owned by the same parent company.
at 24 weeks of follow-up in an ongoing 4-year phase 2 clinical trial, Adam Reich, MD, PhD, reported at Innovations in Dermatology: Virtual Spring Conference 2021.
Secukinumab (Cosentyx), a fully human monoclonal antibody that inhibits interleukin-17A, is widely approved for treatment of psoriasis in adults. In August 2020, the biologic received an expanded indication in Europe for treatment of 6- to 17-year-olds. Two phase 3 clinical trials are underway in an effort to gain a similar broadened indication in the United States to help address the high unmet need for new treatments for psoriasis in the pediatric population, said Dr. Reich, professor and head of the department of dermatology at the University of Rzeszow (Poland).
He reported on 84 pediatric patients participating in the open-label, phase 2, international study. They were randomized to one of two weight-based dosing regimens. Those in the low-dose arm received secukinumab dosed at 75 mg if they weighed less than 50 kg and 150 mg if they weighed more. In the high-dose arm, patients got secukinumab 75 mg if they weighed less than 25 kg, 150 mg if they weighed 25-50 kg, and 300 mg if they tipped the scales in excess of 50 kg.
The primary endpoint in the study was the week-12 rate of at least a 75% improvement from baseline in the Psoriasis Area and Severity Index score, or PASI 75. The rates were similar: 92.9% of patients in the high-dose arm achieved this endpoint, as did 90.5% in the low-dose arm. The PASI 90 rates were 83.3% and 78%, the PASI 100 rates were 61.9% and 54.8%, and clear or almost clear skin, as measured by the Investigator Global Assessment, was achieved in 88.7% of the high- and 85.7% of the low-dose groups. In addition,61.9% of those in the high-dose secukinumab group and 50% in the low-dose group had a score of 0 or 1 on the Children’s Dermatology Life Quality Index – indicating psoriasis has no impact on daily quality of life, he said at the conference sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
At week 24, roughly 95% of patients in both the low- and high-dose secukinumab groups had achieved PASI 75s, 88% reached a PASI 90 response, and 67% were at PASI 100. Nearly 60% of the low-dose and 70% of the high-dose groups had a score of 0 or 1 on the Children’s Dermatology Life Quality Index.
Treatment-emergent adverse event rates were similar in the two study arms. Of note, there was one case of new-onset inflammatory bowel disease in the high-dose group, and one case of vulvovaginal candidiasis as well.
Discussant Bruce E. Strober, MD, PhD, said that, if secukinumab gets a pediatric indication from the Food and Drug Administration, as seems likely, it won’t alter his biologic treatment hierarchy.
“I treat a lot of kids with psoriasis. We have three approved drugs now in etanercept [Enbrel], ustekinumab [Stelara], and ixekizumab [Taltz]. My bias is still towards ustekinumab because it’s infrequently dosed and that’s a huge issue for children. You want to expose them to as few injections as possible, for obvious reasons: It’s easier for parents and other caregivers,” explained Dr. Strober, a dermatologist at Yale University, New Haven, Conn., and Central Connecticut Dermatology, Cromwell, Conn.
“The other issue is in IL-17 inhibition there has been a slight signal of inflammatory bowel disease popping up in children getting these drugs, and therefore you need to screen patients in this age group very carefully – not only the patients themselves, but their family – for IBD risk. If there is any sign of that I would move the IL-17 inhibitors to the back of the line, compared to ustekinumab and etanercept. Ustekinumab is still clearly the one that I think has to be used first line,” he said.
Dr. Strober offered a final word of advice for his colleagues: “You can’t be afraid to treat children with biologic therapies. In fact, preferentially I would use a biologic therapy over methotrexate or light therapy, which is really difficult for children.”
Dr. Reich and Dr. Strober reported receiving research grants from and serving as a consultant to numerous pharmaceutical companies, including Novartis, which markets secukinumab and funded the study.
MedscapeLIVE! and this news organization are owned by the same parent company.
FROM INNOVATIONS IN DERMATOLOGY
Deadly brain tumor: Survival extended by oncolytic virus product
This is a rapidly fatal form of brain cancer. Among historical control patients, the median overall survival was only 5.3 months.
The new results show a median overall survival of 12.2 months.
They come from a phase 1 trial conducted in 12 patients aged 7-18 years who had high-grade gliomas. All of the patients received the experimental therapy, dubbed G207, which was infused directly into the brain tumors.
“In our secondary objectives, we saw promising overall survival data ... [and] we saw that G207 turned immunologically ‘cold’ tumors to ‘hot,’ ” said lead investigator Gregory K. Friedman, MD, from the University of Alabama at Birmingham.
Dr. Friedman presented the new data at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT018). The study was also published simultaneously online in the New England Journal of Medicine.
Although the number of patients in the study was small, the data from this early trial look promising, commented Howard Kaufman, MD, director of the Oncolytic Virus Research Laboratory at Massachusetts General Hospital, Boston, who was not involved in the study.
“This is just a horrendous disease that hasn’t really responded to anything, so seeing some signs of benefit as well as a pretty tolerable safety profile is a very important observation that I think merits further investigation,” he said in an interview.
Engineered virus
G207 is an oncolytic form of HSV-1 created through genetic engineering in which a neurovirulence gene was deleted and viral nucleotide reductase was disabled. The engineered mutations prevent HSV-1 from infecting normal cells while allowing the virus to replicate in tumor cells.
The oncolytic virus product can be inoculated directly into tumors to circumvent the blood-brain barrier, and it preferentially infects neural tissue, making it ideal for treating brain tumors, the investigators explain.
One example of this type of product is already on the market. Talimogene laherparepvec is an oncolytic HSV-1 therapy that was approved in 2015 by the Food and Drug Administration for local treatment (i.e., injection directly into the skin lesion) of unresectable cutaneous, subcutaneous, and nodal lesions in patients with melanoma that recurs after initial surgery.
In their article, Dr. Friedman and colleagues summarized some of the data with G207 that “provided a strong rationale for conducting a trial involving children and adolescents.
“In addition to infecting and lysing tumor cells directly, G207 can reverse tumor immune evasion, increase cross-presentation of tumor antigens, and promote an antitumor immune response even in the absence of virus permissivity,” they wrote. “A single radiation dose enhances G207 efficacy in animal models by increasing viral replication and spread.”
In preclinical studies using tumor xenografts, pediatric brain tumors were 11-fold more sensitive to G207, compared with glioblastomas in adults.
The researchers hypothesized that intratumoral G207 would increase the amount of tumor-infiltrating lymphocytes and thereby convert immunologically “cold” pediatric brain tumors to “hot” and “inflamed” tumors.
Phase 1 trial
The phase 1 trial included four dose cohorts of children and adolescents with a pathologically proven malignant supratentorial brain tumor of at least 1 cm in diameter that had progressed after surgery, radiotherapy, or chemotherapy.
There were three patients in each dose cohort. One cohort received 107 plaque-forming units, the second received 108 PFU, the third received 107 PFU with 5 Gy of radiation, and the fourth received 108 PFU with 5 Gy radiation.
The patients first underwent stereotactic placement of up to four intratumoral catheters. The next day, they underwent infusion of the assigned PFU doses by controlled-rate infusion over 6 hours.
For the patients who received radiation, 5 Gy were administered to the gross tumor volume within 24 hours following G207 administration.
Among the 12 patients, tumors included 10 glioblastomas, one anaplastic astrocytoma, and one high-grade glioma not otherwise specified.
Responses (radiographic, neuropathologic, or clinical) occurred in 11 of the 12 patients.
Four patients were still alive 18 months after treatment, “which exceeds the life expectancy for newly diagnosed patients,” Dr. Friedman noted. Most patients die within 1 year of being diagnosed with pediatric glioma.
The investigators also found evidence to suggest that survival may be improved for patients who experience seroconversion after exposure to HSV-1 in comparison to patients with HSV-1 antibodies from prior HSV-1 infection. The median overall survival was 18.3 months for patients who experienced seroconversion, compared with 5.1 months for three patients who, at baseline, had IgG antibodies to HSV-1.
No dose-limiting toxicities or serious adverse events attributable to G207 occurred. There were 20 grade 1 adverse events that were potentially related to G207.
There was no evidence of peripheral G207 shedding or viremia, the investigators reported.
Radiation effect?
Commenting on the results in an interview, Dr. Kaufman noted that the sample size (12 patients) in this study was too small to determine whether the radiation received by patients in two of the four cohorts had any additive effect.
“Whether to move forward with virus alone or to add the radiation remains an open question that I don’t think was adequately answered,” he said.
Regarding the evidence suggesting that survival was better among patients who did not have antibodies to HSV-1 at baseline, Dr. Kaufman said, “We’ve looked at that in the melanoma population but haven’t seen any correlation there, so that’s interesting.”
The finding could be related to the fact that this was a pediatric population, or it could be related to the location of the tumors in the brain.
“It’s an interesting finding, and it suggests that, in future studies, they might want to select patients who are HSV seronegative up front,” he said.
Dr. Friedman and colleagues are currently planning a phase 2 trial of G207 with 5 Gy of radiation for children and adolescents with recurrent or progressive high-grade gliomas.
The study was supported by grants from the FDA, the National Institutes of Health, Cannonball Kids’ Cancer Foundation, the Rally Foundation for Childhood Cancer Research, Hyundai Hope on Wheels, St. Baldrick’s Foundation, the Department of Defense, the Andrew McDonough B+ Foundation, and the Kaul Pediatric Research Institute; by NIH/National Cancer Institute Cancer Center support grants to the University of Alabama at Birmingham and to the Memorial Sloan Kettering Cancer Center; and by Kelsie’s Crew, Eli’s Block Party Childhood Cancer Foundation, the Eli Jackson Foundation, Jaxon’s FROG Foundation, Battle for a Cure Foundation, and Sandcastle Kids. Dr. Friedman has received grants/support from the organizations listed above, as well as from Eli Lilly and Pfizer. Dr. Kaufman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This is a rapidly fatal form of brain cancer. Among historical control patients, the median overall survival was only 5.3 months.
The new results show a median overall survival of 12.2 months.
They come from a phase 1 trial conducted in 12 patients aged 7-18 years who had high-grade gliomas. All of the patients received the experimental therapy, dubbed G207, which was infused directly into the brain tumors.
“In our secondary objectives, we saw promising overall survival data ... [and] we saw that G207 turned immunologically ‘cold’ tumors to ‘hot,’ ” said lead investigator Gregory K. Friedman, MD, from the University of Alabama at Birmingham.
Dr. Friedman presented the new data at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT018). The study was also published simultaneously online in the New England Journal of Medicine.
Although the number of patients in the study was small, the data from this early trial look promising, commented Howard Kaufman, MD, director of the Oncolytic Virus Research Laboratory at Massachusetts General Hospital, Boston, who was not involved in the study.
“This is just a horrendous disease that hasn’t really responded to anything, so seeing some signs of benefit as well as a pretty tolerable safety profile is a very important observation that I think merits further investigation,” he said in an interview.
Engineered virus
G207 is an oncolytic form of HSV-1 created through genetic engineering in which a neurovirulence gene was deleted and viral nucleotide reductase was disabled. The engineered mutations prevent HSV-1 from infecting normal cells while allowing the virus to replicate in tumor cells.
The oncolytic virus product can be inoculated directly into tumors to circumvent the blood-brain barrier, and it preferentially infects neural tissue, making it ideal for treating brain tumors, the investigators explain.
One example of this type of product is already on the market. Talimogene laherparepvec is an oncolytic HSV-1 therapy that was approved in 2015 by the Food and Drug Administration for local treatment (i.e., injection directly into the skin lesion) of unresectable cutaneous, subcutaneous, and nodal lesions in patients with melanoma that recurs after initial surgery.
In their article, Dr. Friedman and colleagues summarized some of the data with G207 that “provided a strong rationale for conducting a trial involving children and adolescents.
“In addition to infecting and lysing tumor cells directly, G207 can reverse tumor immune evasion, increase cross-presentation of tumor antigens, and promote an antitumor immune response even in the absence of virus permissivity,” they wrote. “A single radiation dose enhances G207 efficacy in animal models by increasing viral replication and spread.”
In preclinical studies using tumor xenografts, pediatric brain tumors were 11-fold more sensitive to G207, compared with glioblastomas in adults.
The researchers hypothesized that intratumoral G207 would increase the amount of tumor-infiltrating lymphocytes and thereby convert immunologically “cold” pediatric brain tumors to “hot” and “inflamed” tumors.
Phase 1 trial
The phase 1 trial included four dose cohorts of children and adolescents with a pathologically proven malignant supratentorial brain tumor of at least 1 cm in diameter that had progressed after surgery, radiotherapy, or chemotherapy.
There were three patients in each dose cohort. One cohort received 107 plaque-forming units, the second received 108 PFU, the third received 107 PFU with 5 Gy of radiation, and the fourth received 108 PFU with 5 Gy radiation.
The patients first underwent stereotactic placement of up to four intratumoral catheters. The next day, they underwent infusion of the assigned PFU doses by controlled-rate infusion over 6 hours.
For the patients who received radiation, 5 Gy were administered to the gross tumor volume within 24 hours following G207 administration.
Among the 12 patients, tumors included 10 glioblastomas, one anaplastic astrocytoma, and one high-grade glioma not otherwise specified.
Responses (radiographic, neuropathologic, or clinical) occurred in 11 of the 12 patients.
Four patients were still alive 18 months after treatment, “which exceeds the life expectancy for newly diagnosed patients,” Dr. Friedman noted. Most patients die within 1 year of being diagnosed with pediatric glioma.
The investigators also found evidence to suggest that survival may be improved for patients who experience seroconversion after exposure to HSV-1 in comparison to patients with HSV-1 antibodies from prior HSV-1 infection. The median overall survival was 18.3 months for patients who experienced seroconversion, compared with 5.1 months for three patients who, at baseline, had IgG antibodies to HSV-1.
No dose-limiting toxicities or serious adverse events attributable to G207 occurred. There were 20 grade 1 adverse events that were potentially related to G207.
There was no evidence of peripheral G207 shedding or viremia, the investigators reported.
Radiation effect?
Commenting on the results in an interview, Dr. Kaufman noted that the sample size (12 patients) in this study was too small to determine whether the radiation received by patients in two of the four cohorts had any additive effect.
“Whether to move forward with virus alone or to add the radiation remains an open question that I don’t think was adequately answered,” he said.
Regarding the evidence suggesting that survival was better among patients who did not have antibodies to HSV-1 at baseline, Dr. Kaufman said, “We’ve looked at that in the melanoma population but haven’t seen any correlation there, so that’s interesting.”
The finding could be related to the fact that this was a pediatric population, or it could be related to the location of the tumors in the brain.
“It’s an interesting finding, and it suggests that, in future studies, they might want to select patients who are HSV seronegative up front,” he said.
Dr. Friedman and colleagues are currently planning a phase 2 trial of G207 with 5 Gy of radiation for children and adolescents with recurrent or progressive high-grade gliomas.
The study was supported by grants from the FDA, the National Institutes of Health, Cannonball Kids’ Cancer Foundation, the Rally Foundation for Childhood Cancer Research, Hyundai Hope on Wheels, St. Baldrick’s Foundation, the Department of Defense, the Andrew McDonough B+ Foundation, and the Kaul Pediatric Research Institute; by NIH/National Cancer Institute Cancer Center support grants to the University of Alabama at Birmingham and to the Memorial Sloan Kettering Cancer Center; and by Kelsie’s Crew, Eli’s Block Party Childhood Cancer Foundation, the Eli Jackson Foundation, Jaxon’s FROG Foundation, Battle for a Cure Foundation, and Sandcastle Kids. Dr. Friedman has received grants/support from the organizations listed above, as well as from Eli Lilly and Pfizer. Dr. Kaufman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This is a rapidly fatal form of brain cancer. Among historical control patients, the median overall survival was only 5.3 months.
The new results show a median overall survival of 12.2 months.
They come from a phase 1 trial conducted in 12 patients aged 7-18 years who had high-grade gliomas. All of the patients received the experimental therapy, dubbed G207, which was infused directly into the brain tumors.
“In our secondary objectives, we saw promising overall survival data ... [and] we saw that G207 turned immunologically ‘cold’ tumors to ‘hot,’ ” said lead investigator Gregory K. Friedman, MD, from the University of Alabama at Birmingham.
Dr. Friedman presented the new data at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT018). The study was also published simultaneously online in the New England Journal of Medicine.
Although the number of patients in the study was small, the data from this early trial look promising, commented Howard Kaufman, MD, director of the Oncolytic Virus Research Laboratory at Massachusetts General Hospital, Boston, who was not involved in the study.
“This is just a horrendous disease that hasn’t really responded to anything, so seeing some signs of benefit as well as a pretty tolerable safety profile is a very important observation that I think merits further investigation,” he said in an interview.
Engineered virus
G207 is an oncolytic form of HSV-1 created through genetic engineering in which a neurovirulence gene was deleted and viral nucleotide reductase was disabled. The engineered mutations prevent HSV-1 from infecting normal cells while allowing the virus to replicate in tumor cells.
The oncolytic virus product can be inoculated directly into tumors to circumvent the blood-brain barrier, and it preferentially infects neural tissue, making it ideal for treating brain tumors, the investigators explain.
One example of this type of product is already on the market. Talimogene laherparepvec is an oncolytic HSV-1 therapy that was approved in 2015 by the Food and Drug Administration for local treatment (i.e., injection directly into the skin lesion) of unresectable cutaneous, subcutaneous, and nodal lesions in patients with melanoma that recurs after initial surgery.
In their article, Dr. Friedman and colleagues summarized some of the data with G207 that “provided a strong rationale for conducting a trial involving children and adolescents.
“In addition to infecting and lysing tumor cells directly, G207 can reverse tumor immune evasion, increase cross-presentation of tumor antigens, and promote an antitumor immune response even in the absence of virus permissivity,” they wrote. “A single radiation dose enhances G207 efficacy in animal models by increasing viral replication and spread.”
In preclinical studies using tumor xenografts, pediatric brain tumors were 11-fold more sensitive to G207, compared with glioblastomas in adults.
The researchers hypothesized that intratumoral G207 would increase the amount of tumor-infiltrating lymphocytes and thereby convert immunologically “cold” pediatric brain tumors to “hot” and “inflamed” tumors.
Phase 1 trial
The phase 1 trial included four dose cohorts of children and adolescents with a pathologically proven malignant supratentorial brain tumor of at least 1 cm in diameter that had progressed after surgery, radiotherapy, or chemotherapy.
There were three patients in each dose cohort. One cohort received 107 plaque-forming units, the second received 108 PFU, the third received 107 PFU with 5 Gy of radiation, and the fourth received 108 PFU with 5 Gy radiation.
The patients first underwent stereotactic placement of up to four intratumoral catheters. The next day, they underwent infusion of the assigned PFU doses by controlled-rate infusion over 6 hours.
For the patients who received radiation, 5 Gy were administered to the gross tumor volume within 24 hours following G207 administration.
Among the 12 patients, tumors included 10 glioblastomas, one anaplastic astrocytoma, and one high-grade glioma not otherwise specified.
Responses (radiographic, neuropathologic, or clinical) occurred in 11 of the 12 patients.
Four patients were still alive 18 months after treatment, “which exceeds the life expectancy for newly diagnosed patients,” Dr. Friedman noted. Most patients die within 1 year of being diagnosed with pediatric glioma.
The investigators also found evidence to suggest that survival may be improved for patients who experience seroconversion after exposure to HSV-1 in comparison to patients with HSV-1 antibodies from prior HSV-1 infection. The median overall survival was 18.3 months for patients who experienced seroconversion, compared with 5.1 months for three patients who, at baseline, had IgG antibodies to HSV-1.
No dose-limiting toxicities or serious adverse events attributable to G207 occurred. There were 20 grade 1 adverse events that were potentially related to G207.
There was no evidence of peripheral G207 shedding or viremia, the investigators reported.
Radiation effect?
Commenting on the results in an interview, Dr. Kaufman noted that the sample size (12 patients) in this study was too small to determine whether the radiation received by patients in two of the four cohorts had any additive effect.
“Whether to move forward with virus alone or to add the radiation remains an open question that I don’t think was adequately answered,” he said.
Regarding the evidence suggesting that survival was better among patients who did not have antibodies to HSV-1 at baseline, Dr. Kaufman said, “We’ve looked at that in the melanoma population but haven’t seen any correlation there, so that’s interesting.”
The finding could be related to the fact that this was a pediatric population, or it could be related to the location of the tumors in the brain.
“It’s an interesting finding, and it suggests that, in future studies, they might want to select patients who are HSV seronegative up front,” he said.
Dr. Friedman and colleagues are currently planning a phase 2 trial of G207 with 5 Gy of radiation for children and adolescents with recurrent or progressive high-grade gliomas.
The study was supported by grants from the FDA, the National Institutes of Health, Cannonball Kids’ Cancer Foundation, the Rally Foundation for Childhood Cancer Research, Hyundai Hope on Wheels, St. Baldrick’s Foundation, the Department of Defense, the Andrew McDonough B+ Foundation, and the Kaul Pediatric Research Institute; by NIH/National Cancer Institute Cancer Center support grants to the University of Alabama at Birmingham and to the Memorial Sloan Kettering Cancer Center; and by Kelsie’s Crew, Eli’s Block Party Childhood Cancer Foundation, the Eli Jackson Foundation, Jaxon’s FROG Foundation, Battle for a Cure Foundation, and Sandcastle Kids. Dr. Friedman has received grants/support from the organizations listed above, as well as from Eli Lilly and Pfizer. Dr. Kaufman disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AACR 2021
Sarcoidosis: An FP’s primer on an enigmatic disease
Sarcoidosis is a multisystem inflammatory disease of unclear etiology that primarily affects the lungs. It can occur at any age but usually develops before the age of 50 years, with an initial peak incidence at 20 to 29 years and a second peak incidence after 50 years of age, especially among women in Scandinavia and Japan.1 Sarcoidosis affects men and women of all racial and ethnic groups throughout the world, but differences based on race, sex, and geography are noted.1
The highest rates are reported in northern European and African-American individuals, particularly in women.1,2 The adjusted annual incidence of sarcoidosis among African Americans is approximately 3 times that among White Americans3 and is more likely to be chronic and fatal in African Americans.3 The disease can be familial with a possible recessive inheritance mode with incomplete penetrance.4 Risk of sarcoidosis in monozygotic twins appears to be 80 times greater than that in the general population, which supports genetic factors accounting for two-thirds of disease susceptibility.5
Likely factors in the development of sarcoidosis
The exact cause of sarcoidosis is unknown, but we have insights into its pathogenesis and potential triggers.1,6-9 Genes involved are being identified: class I and II human leukocyte antigen (HLA) molecules are most consistently associated with risk of sarcoidosis. Environmental exposures can activate the innate immune system and precondition a susceptible individual to react to potential causative antigens in a highly polarized, antigen-specific Th1 immune response. The epithelioid granulomatous response involves local proinflammatory cytokine production and enhanced T-cell immunity at sites of inflammation.10 Granulomas generally form to confine pathogens, restrict inflammation, and protect surrounding tissue.11-13
ACCESS (A Case Control Etiologic Study of Sarcoidosis) identified several environmental exposures such as chemicals used in the agriculture industry, mold or mildew, and musty odors at work.14 Tobacco use was not associated with sarcoidosis.14 Recent studies have shown positive associations with service in the US Navy,15 metal working,16 firefighting,17 the handling of building supplies,18 and onsite exposure while assisting in rescue efforts at the World Trade Center disaster.19 Other data support the likelihood that specific environmental exposures associated with microbe-rich environments modestly increase the risk of sarcoidosis.14 Mycobacterial and propionibacterial DNA and RNA are potentially associated with sarcoidosis.20
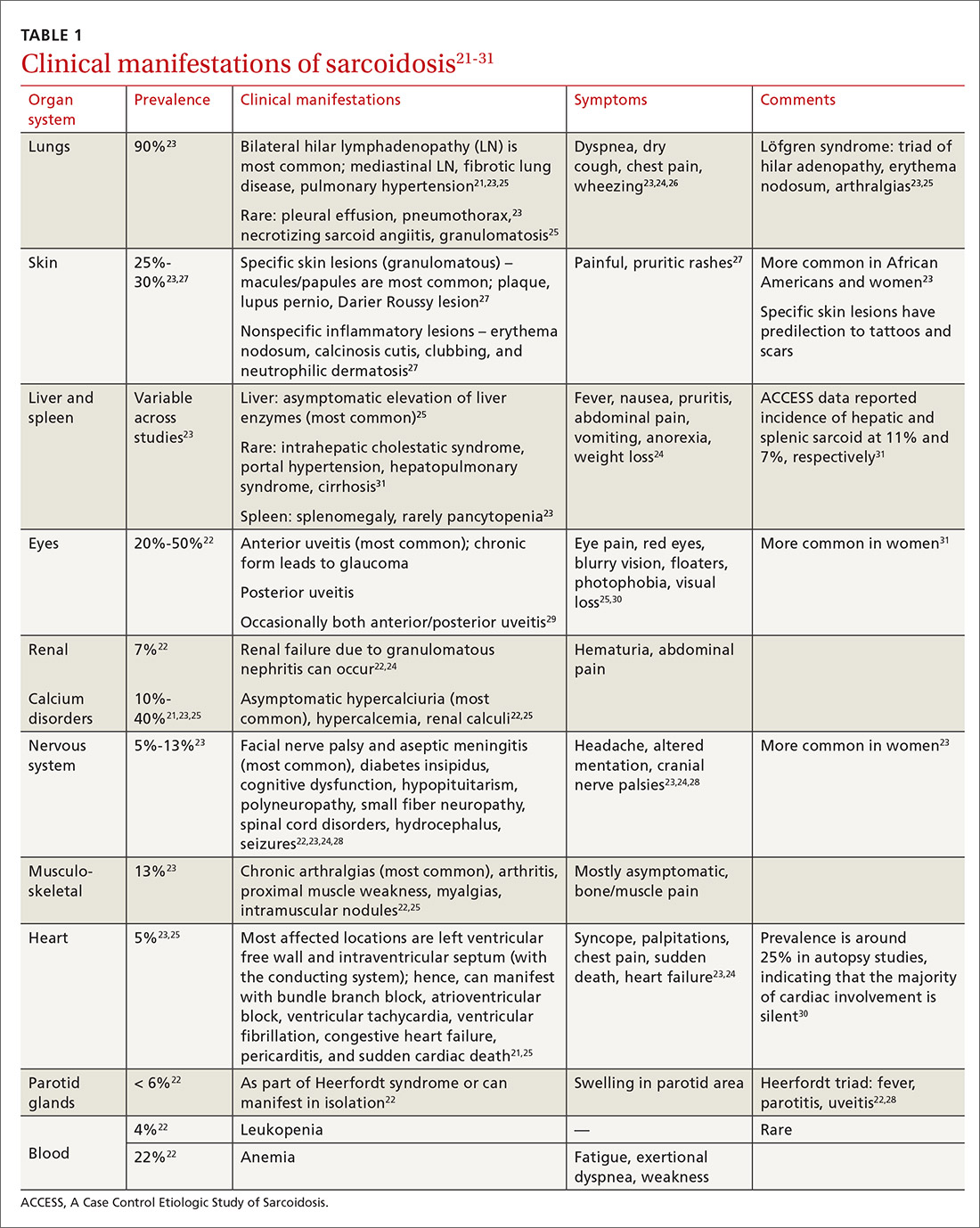
Clinical manifestations are nonspecific
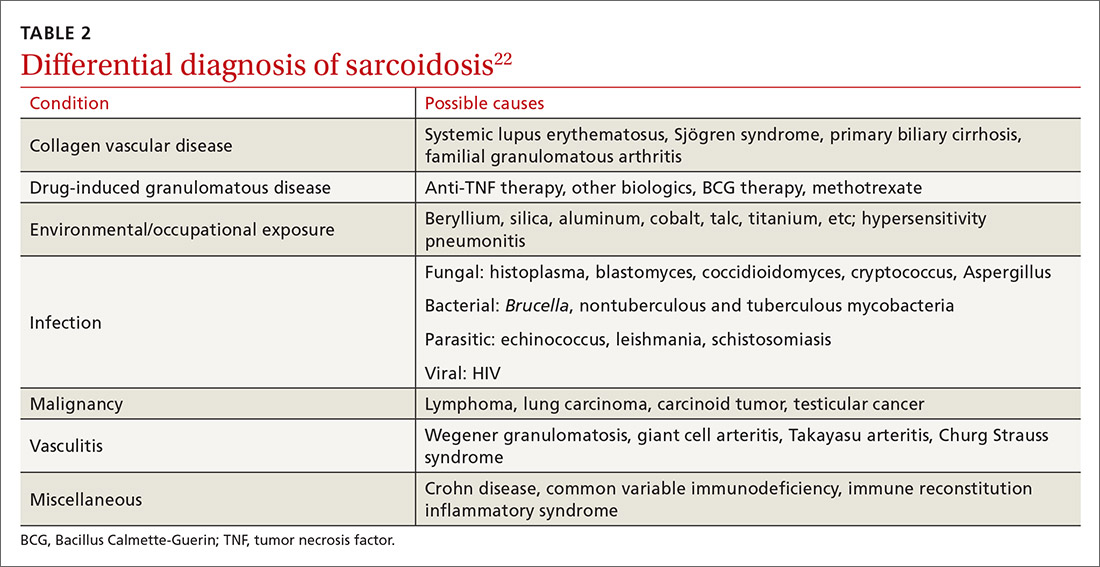
The diagnosis of sarcoidosis can be difficult and delayed due to diverse organ involvement and nonspecific presentations. TABLE 121-31 shows the diverse manifestations in a patient with suspected sarcoidosis. Around 50% of the patients are asymptomatic.23,24 Sarcoidosis is a diagnosis of exclusion, starting with a detailed history to rule out infections, occupational or environmental exposures, malignancies, and other possible disorders (TABLE 2).22
Diagnostic work-up
Radiologic studies
Chest x-ray (CXR) provides diagnostic and prognostic information in the evaluation of sarcoidosis using the Scadding classification system (FIGURE 1).21,25,32,33 Interobserver variability, especially between stages II and III and III and IV is the major limitation of this system.32 At presentation, radiographs are abnormal in approximately 90% of patients.34 Lymphadenopathy is the most common radiographic abnormality, occurring in more than two-thirds of cases, and pulmonary opacities (nodules and reticulation) with a middle to upper lobe predilection are present in 20% to 50% of patients.1,31,35 The nodules vary in size and can coalesce and cause alveolar collapse, thus producing consolidation.36 Linear opacities radiating laterally from the hilum into the middle and upper zones are characteristic in fibrotic disease.
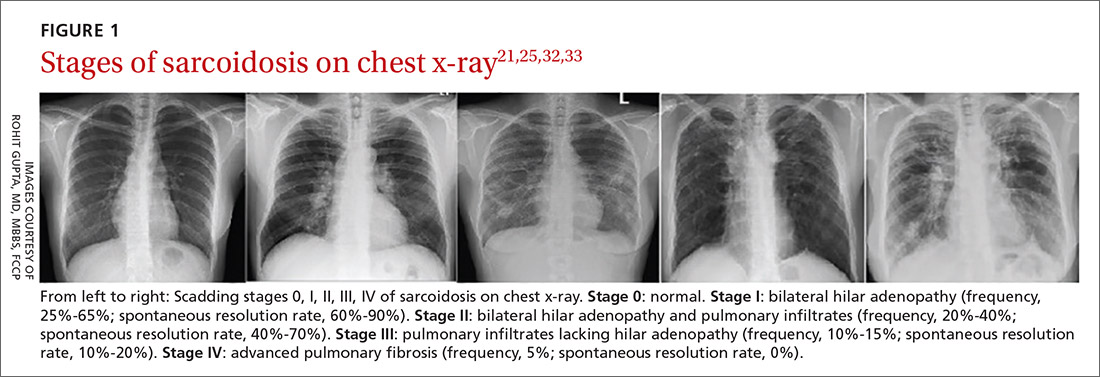
Continue to: High-resoluton computed tomography
High-resolution computed tomography (HRCT). Micronodules in a perilymphatic distribution with upper lobe predominance combined with subcarinal and symmetrical hilar lymph node enlargement is practically diagnostic of sarcoidosis in the right clinical context. TABLE 321,23,25,32 and FIGURE 221,23,25,32 summarize the common CT chest findings of sarcoidosis.
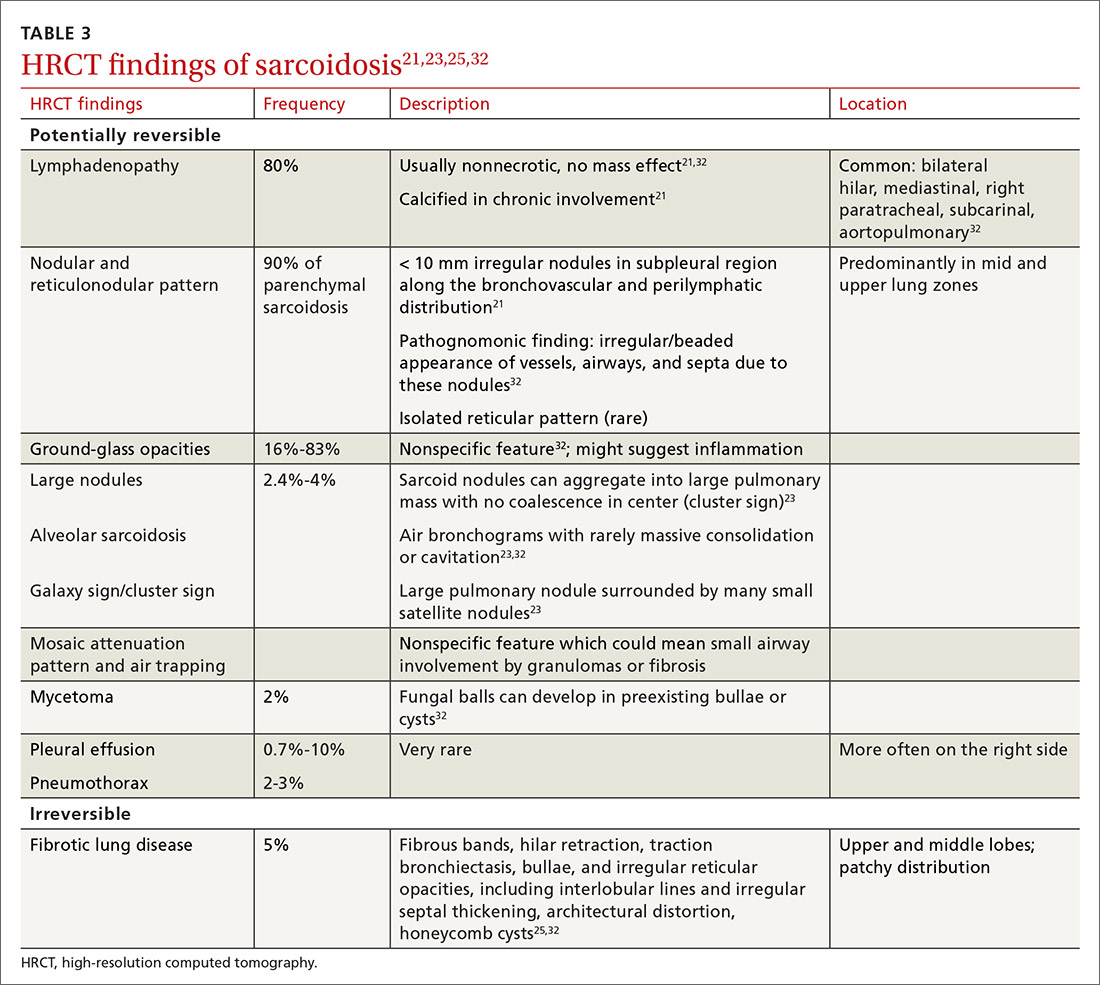
Advanced imaging such as (18)F-fluorodeoxyglucose positron emission tomography (PET) and magnetic resonance imaging (MRI) are used in specialized settings for advanced pulmonary, cardiac, or neurosarcoidosis.
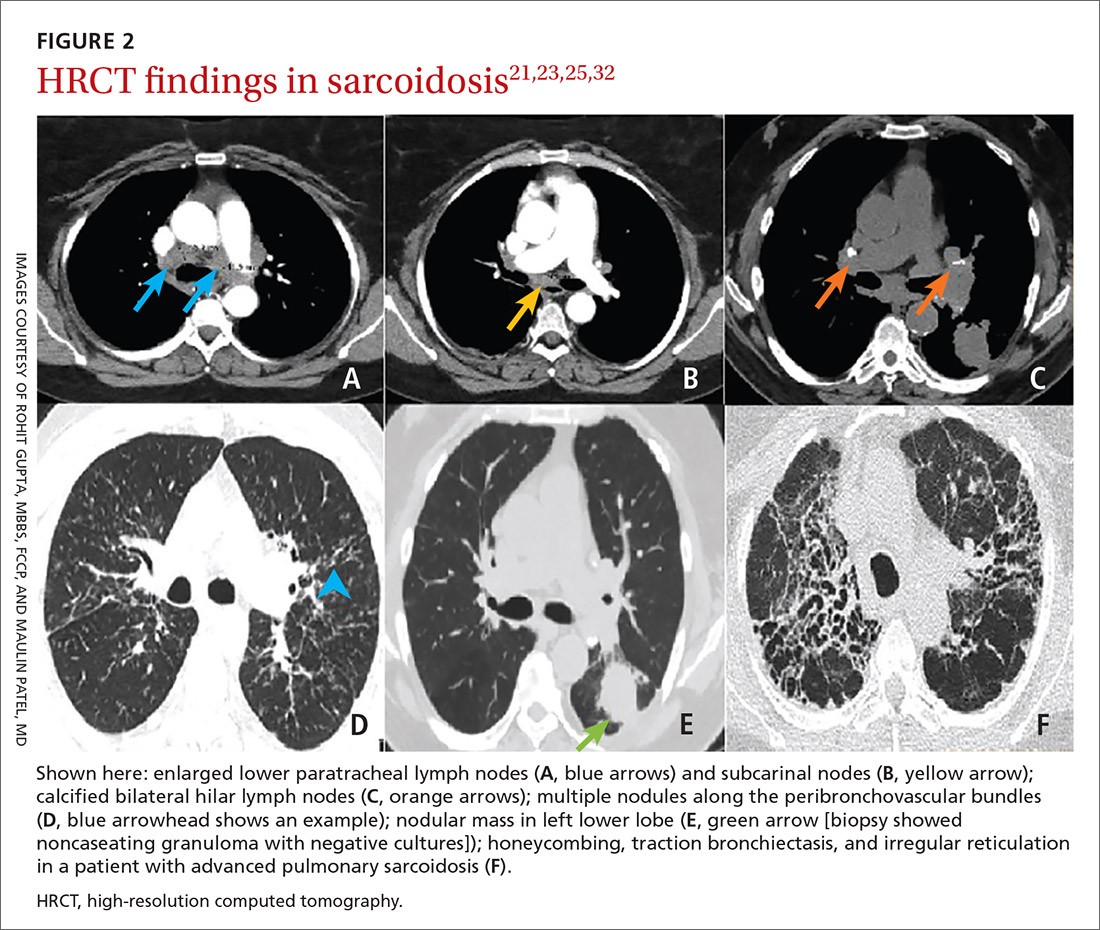
Tissue biopsy
Skin lesions (other than erythema nodosum), eye lesions, and peripheral lymph nodes are considered the safest extrapulmonary locations for biopsy.21,25 If pulmonary infiltrates or lymphadenopathy are present, or if extrapulmonary biopsy sites are not available, then flexible bronchoscopy with biopsy is the mainstay for tissue sampling.25
Bronchoalveolar lavage (BAL), transbronchial biopsy (TBB), endobronchial biopsy (EBB), and endobronchial ultrasound (EBUS) are invaluable modalities that have reduced the need for open lung biopsy. BAL in sarcoidosis can show lymphocytosis > 15% (nonspecific) and a CD4:CD8 lymphocyte ratio > 3.5 (specificity > 90%).21,22 TBB is more sensitive than EBB; however, sensitivity overall is heightened when both of them are combined. The advent of EBUS has increased the safety and efficiency of needle aspiration of mediastinal lymph nodes. Diagnostic yield of EBUS (~80%) is superior to that with TBB and EBB (~50%), especially in stage I and II sarcoidosis.37 The combination of EBUS with TBB improves the diagnostic yield to ~90%.37
The decision to obtain biopsy samples hinges on the nature of clinical and radiologic findings (FIGURE 3).22,25,26
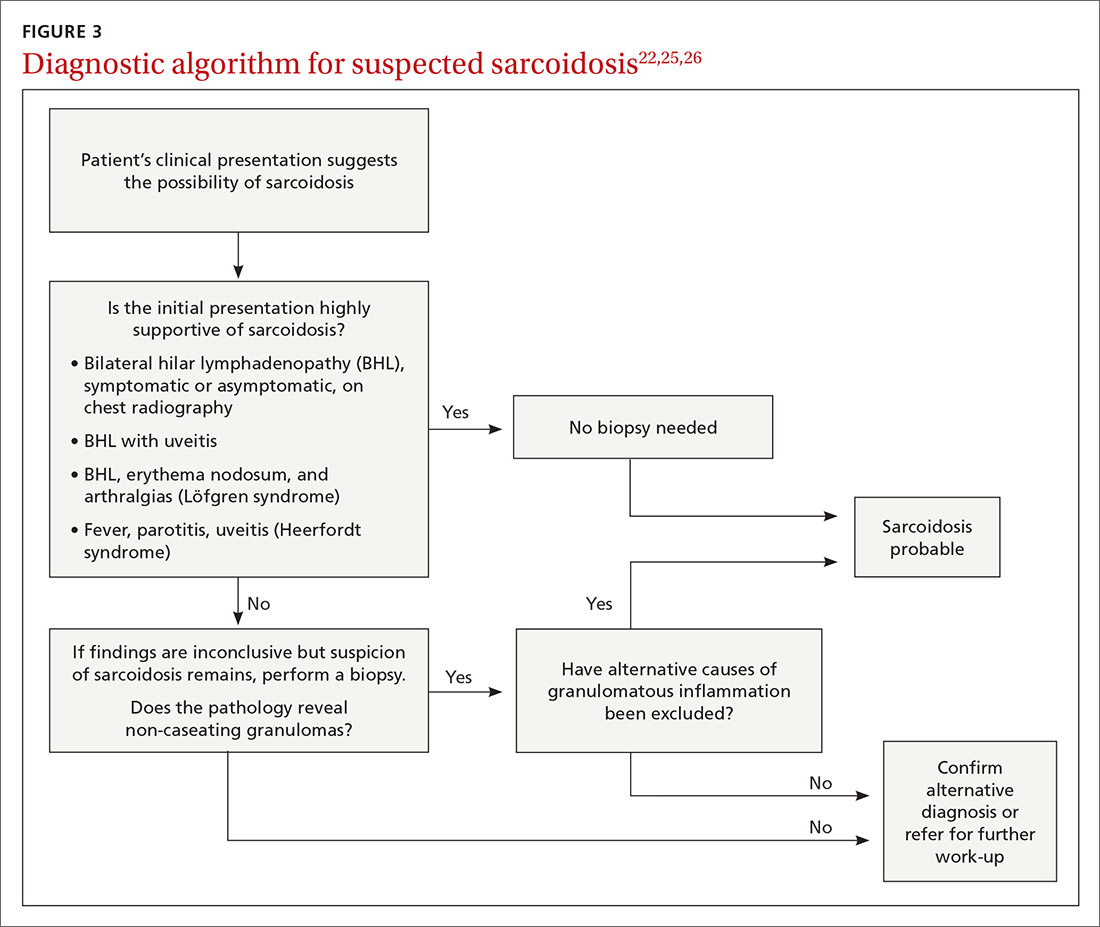
Continue to: Laboratory studies
Laboratory studies
Multiple abnormalities may be seen in sarcoidosis, and specific lab tests may help support a diagnosis of sarcoidosis or detect organ-specific disease activity (TABLE 4).22,23,25,38 However, no consistently accurate biomarkers exist for use in clinical practice. An angiotensin-converting enzyme (ACE) level greater than 2 times the upper limit of normal may be helpful; however, sensitivity remains low, and genetic polymorphisms can influence the ACE level.25 Biomarkers sometimes used to assess disease activity are serum interleukin-2 receptor, neopterin, chitotriosidase, lysozyme, KL-6 glycoprotein, and amyloid A.21
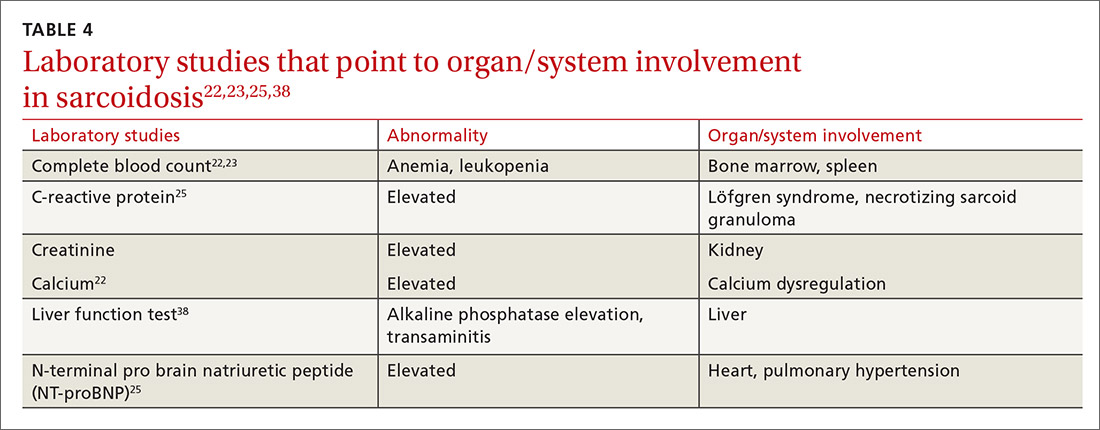
Additional tests to assess specific features or organ involvement
Pulmonary function testing (PFT) is reviewed in detail below under “pulmonary sarcoidosis.”
Electrocardiogram (EKG)/transthoracic echocardiogram (TTE). EKG abnormalities—conduction disturbances, arrhythmias, or nonspecific ST segment and T-wave changes—are the most common nonspecific findings.30 TTE findings are also nonspecific but have value in assessing cardiac chamber size and function and myocardial involvement. TTE is indeed the most common screening modality for sarcoidosis-associated pulmonary hypertension (SAPH), which is definitively diagnosed by right heart catheterization (RHC). Further evaluation for cardiac sarcoidosis can be done with cardiac MRI or fluorodeoxyglucose PET in specialized settings.
Lumbar puncture (LP) may reveal lymphocytic infiltration in suspected neurosarcoidosis, but the finding is nonspecific and can reflect infection or malignancy. Oligoclonal bands may also be seen in about one-third of neurosarcoidosis cases, and it is imperative to rule out multiple sclerosis.28
Pulmonary sarcoidosis
Pulmonary sarcoidosis accounts for most of the morbidity, mortality, and health care use associated with sarcoidosis.39,40
Continue to: Pathology of early and advanced pulmonary sarcoidosis
Pathology of early and advanced pulmonary sarcoidosis
Sarcoidosis is characterized by coalescing, tightly clustered, nonnecrotizing granulomas in the lung (FIGURE 4), most often located along the lymphatic routes of the pleura, interlobular septa, and bronchovascular bundles.41 Granulomas contain epithelioid cells or multinucleated giant cells surrounded by a chronic lymphocytic infiltrate. Typically, intracytoplasmic inclusions, such as Schaumann bodies, asteroid bodies, and blue bodies of calcium oxalates are noted within giant cells.
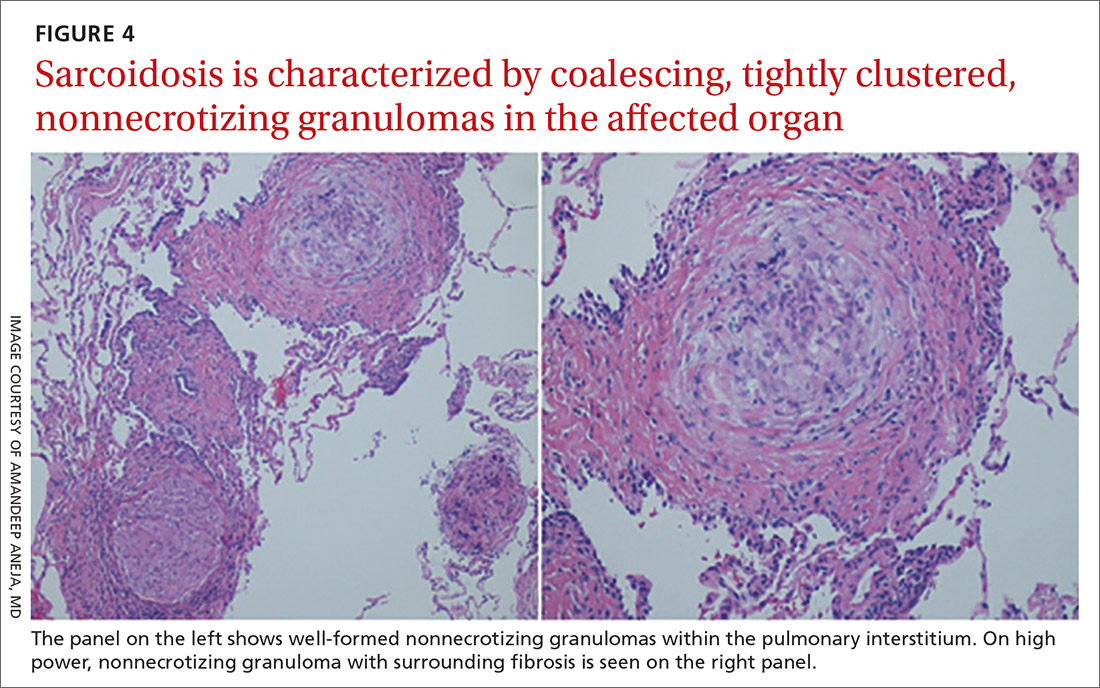
In chronic disease, lymphocytic infiltrate vanishes and granulomas tend to become increasingly fibrotic and enlarge to form hyalinized nodules rich with densely eosinophilic collagen. In 10% to 30% of cases, the lungs undergo progressive fibrosis.40 Nonresolving inflammation appears to be the major cause of fibrosis and the peribronchovascular localization leading to marked bronchial distortion.
Clinical features, monitoring, and outcomes
Pulmonary involvement occurs in most patients with sarcoidosis, and subclinical pulmonary disease is generally present, even when extrathoracic manifestations predominate.23 Dry cough, dyspnea, and chest discomfort are the most common symptoms. Chest auscultation is usually unremarkable. Wheezing is more common in those with fibrosis and is attributed to airway-centric fibrosis.42 There is often a substantial delay between the onset of symptoms and the diagnosis of pulmonary sarcoidosis, as symptoms are nonspecific and might be mistaken for more common pulmonary diseases, such as asthma or chronic bronchitis.43
Since sarcoidosis can affect pulmonary parenchyma, interstitium, large and small airways, pulmonary vasculature, and respiratory muscles, the pattern of lung function impairment on PFT varies from normal to obstruction, restriction, isolated diffusion defect, or a combination of these. The typical physiologic abnormality is a restrictive ventilatory defect with a decreased diffusing capacity of the lung for carbon monoxide (DLCO). Extent of disease seen on HRCT correlates with level of restriction.44 Airway obstruction can be multifactorial and due to airway distortion (more likely to occur in fibrotic lung disease) and luminal disease.45-48 The 6-minute walk test and DLCO can also aid in the diagnosis of SAPH and advanced parenchymal lung disease.
While monitoring is done clinically and with testing (PFT and imaging) as needed, the optimal approach is unclear. Nevertheless, longitudinal monitoring with testing may provide useful management and prognostic information.40 Pulmonary function can remain stable in fibrotic sarcoidosis over extended periods and actually can improve in some patients.49 Serial spirometry, particularly forced vital capacity, is the most reliable tool for monitoring; when a decline in measurement occurs, chest radiography can elucidate the mechanism.50,51
Continue to: Because sarcoidosis is a multisystem disease...
Because sarcoidosis is a multisystem disease, caution needs to be exercised when evaluating a patient’s new or worsening respiratory symptoms to accurately determine the cause of symptoms and direct therapy accordingly. In addition to refractory inflammatory pulmonary disease, airway disease, infection, fibrosis, and SAPH, one needs to consider extrapulmonary involvement or complications such as cardiac or neurologic disease, musculoskeletal disease, depression, or fatigue. Adverse medication effects, deconditioning, or unrelated (or possibly related) disorders (eg pulmonary embolism) may be to blame.
Determining prognosis
Prognosis of sarcoidosis varies and depends on epidemiologic factors, clinical presentation, and course, as well as specific organ involvement. Patients may develop life-threatening pulmonary, cardiac, or neurologic complications. End-stage disease may require organ transplantation for eligible patients.
Most patients with pulmonary sarcoidosis experience clinical remission with minimal residual organ impairment and a favorable long-term outcome. Advanced pulmonary disease (known as APS) occurs in a small proportion of patients with sarcoidosis but accounts for most of the poor outcomes in sarcoidosis.40 APS is variably defined, but it generally includes pulmonary fibrosis, SAPH, and respiratory infection.
One percent to 5% of patients with sarcoidosis die from complications, and mortality is higher in women and African Americans.52 Mortality and morbidity may be increasing.53 The reasons behind these trends are unclear but could include true increases in disease incidence, better detection rates, greater severity of disease, or an aging population. Increased hospitalizations and health care use might be due to organ damage from granulomatous inflammation (and resultant fibrosis), complications associated with treatment, and psychosocial effects of the disease/treatment.
Management
Management consists primarily of anti-inflammatory or immunosuppressive therapies but can also include measures to address specific complications (such as fatigue) and organ transplant, as well as efforts to counter adverse medication effects. Other supportive and preventive measures may include, on a case-by-case basis, oxygen supplementation, vaccinations, or pulmonary rehabilitation. Details of these are found in other, more in-depth reviews on treatment; we will briefly review anti-inflammatory therapy, which forms the cornerstone of treatment in most patients with sarcoidosis.
Continue to: General approach to treatment decisions
General approach to treatment decisions. Anti-inflammatory therapy is used to reduce granulomatous inflammation, thereby preserving organ function and reducing symptoms. A decision to begin treatment is one shared with the patient and is based on symptoms and potential danger of organ system failure.54 Patients who are symptomatic or have progressive disease or physiologic impairment are generally candidates for treatment. Monitoring usually suffices for those who have minimal symptoms, stable disease, and preserved organ function.
Patients with pulmonary sarcoidosis at CXR stage 0 should not receive treatment, given that large, randomized trials have shown no meaningful benefit and that these patients have a high likelihood of spontaneous remission and excellent long-term prognosis.55-58 However, a subgroup of patients classified as stage 0/I on CXR may show parenchymal disease on HRCT,59 and, if more symptomatic, could be considered for treatment. For patients with stage II to IV pulmonary sarcoidosis with symptoms, there is good evidence that treatment may improve lung function and reduce dyspnea and fatigue.57,60-62
Corticosteroids are first-line treatment for most patients. Based on expert opinion, treatment of pulmonary sarcoidosis is generally started with oral prednisone (or an equivalent corticosteroid). A starting dose of 20 to 40 mg/d generally is sufficient for most patients. If the patient responds to initial treatment, prednisone dose is tapered over a period of months. If symptoms worsen during tapering, the minimum effective dose is maintained without further attempts at tapering. Treatment is continued for at least 3 to 6 months but it might be needed for longer durations; unfortunately, evidence-based guidelines are lacking.63 Once the patient goes into remission, close monitoring is done for possible relapses. Inhaled corticosteroids alone have not reduced symptoms or improved lung function in patients with pulmonary sarcoidosis.64-66
Steroid-sparing agents are added for many patients. For patients receiving chronic prednisone therapy (≥ 10 mg for > 6 months), steroid-sparing agents are considered to minimize the adverse effects of steroids or to better control the inflammatory activity of sarcoidosis. These agents must be carefully selected, and clinical and laboratory monitoring need to be done throughout therapy. TABLE 558,64,67-81
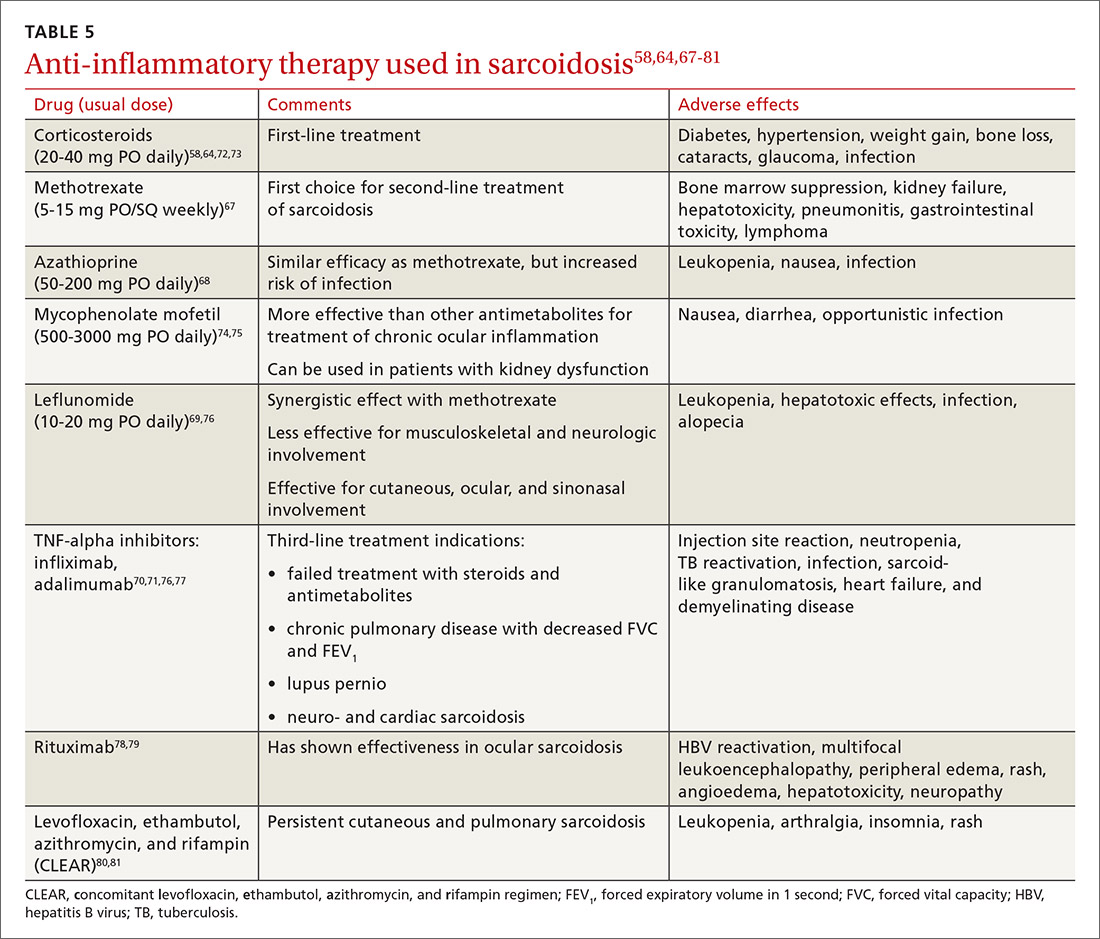
The management might be complicated for extrapulmonary, multi-organ, and advanced sarcoidosis (advanced pulmonary sarcoidosis, cardiac disease, neurosarcoidosis, lupus pernio, etc) when specialized testing, as well as a combination of corticosteroids and steroid-sparing agents (with higher doses or prolonged courses), might be needed. This should be performed at an expert sarcoidosis center, ideally in a multidisciplinary setting involving pulmonologists and/or rheumatologists, chest radiologists, and specialists as indicated, based on specific organ involvement.
Continue to: Research and future directions
Research and future directions
Key goals for research are identifying more accurate biomarkers of disease, improving diagnosis of multi-organ disease, determining validated endpoints of clinical trials in sarcoidosis, and developing treatments for refractory cases.
There is optimism and opportunity in the field of sarcoidosis overall. An example of an advancement is in the area of APS, as the severity and importance of this phenotype has been better understood. Worldwide registries and trials of pulmonary vasodilator therapy (bosentan, sildenafil, epoprostenol, and inhaled iloprost) in patients with SAPH without left ventricular dysfunction are promising.82-85 However, no benefit in survival has been shown.
RioSAPH is a double-blind, placebo-controlled trial of Riociguat (a stimulator of soluble guanylate cyclase) for SAPH (NCT02625558) that is closed to enrollment and undergoing data review. Similarly, results of the phase IV study of pirfenidone, an antifibrotic agent that was shown to decrease disease progression and deaths in idiopathic pulmonary fibrosis,86 are awaited in the near future.
Other potential directions being explored are multicenter patient registries and randomized controlled trials, analyses of existing databases, use of biobanking, and patient-centered outcome measures. Hopefully, the care of patients with sarcoidosis will become more evidence based with ongoing and upcoming research in this field.
CORRESPONDENCE
Rohit Gupta, MBBS, FCCP, 3401 North Broad Street, 7 Parkinson Pavilion, Philadelphia, PA 19140; [email protected]
1. Costabel U, Hunninghake G. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur Respir J. 1999;14:735-737.
2. Hillerdal G, Nöu E, Osterman K, et al. Sarcoidosis: epidemiology and prognosis. A 15-year European study. Am Rev Respir Dis. 1984;130:29-32.
3. Mirsaeidi M, Machado RF, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015;147:438-449.
4. Rybicki BA, Iannuzzi MC, Frederick MM, et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Resp Crit Care Med. 2001;164:2085-2091.
5. Sverrild A, Backer V, Kyvik KO, et al. Heredity in sarcoidosis:a registry-based twin study. Thorax. 2008;63:894.
6. Vuyst P, Dumortier P, Schandené L, et al. Sarcoidlike lung granulomatosis induced by aluminum dusts. Am Rev Respir Dis. 1987;135:493-497.
7. Werfel U, Schneider J, Rödelsperger K, et al. Sarcoid granulomatosis after zirconium exposure with multiple organ involvement. European Respir J. 1998;12:750.
8. Newman KL, Newman LS. Occupational causes of sarcoidosis. Curr Opin Allergy Clin Immunol. 2012;12:145-150.
9. Zissel G, Müller-Quernheim J. Specific antigen(s) in sarcoidosis:a link to autoimmunity? Eur Respir J. 2016;47:707-709.
10. Chen ES, Moller DR. Etiology of sarcoidosis. Clin Chest Med. 2008;29:365-377.
11. Agostini C, Adami F, Semenzato G. New pathogenetic insights into the sarcoid granuloma. Curr Opin Rheumatol. 2000;12:71-76.
12. Valentonyte R, Hampe J, Huse K, et al. Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat Genet. 2005;37:357-364.
13. Rybicki BA, Walewski JL, Maliarik MJ, et al. The BTNL2 gene and sarcoidosis susceptibility in African Americans and Whites. Am J Hum Genet. 2005;77:491-499.
14. Newman LS, Rose CS, Bresnitz EA, et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am J Respir Crit Care Med. 2004;170:1324-1330.
15. Gorham ED, Garland CF, Garland FC, et al. Trends and occupational associations in incidence of hospitalized pulmonary sarcoidosis and other lung diseases in Navy personnel: a 27-year historical prospective study, 1975-2001. Chest. 2004;126:1431-1438.
16. Kucera GP, Rybicki BA, Kirkey KL, et al. Occupational risk factors for sarcoidosis in African-American siblings. Chest. 2003;123:1527-1535.
17. Prezant DJ, Dhala A, Goldstein A, et al. The incidence, prevalence, and severity of sarcoidosis in New York City firefighters. Chest. 1999;116:1183-1193.
18. Barnard J, Rose C, Newman L, et al. Job and industry classifications associated with sarcoidosis in A Case–Control Etiologic Study of Sarcoidosis (ACCESS). J Occup Environ Med. 2005;47:226-234.
19. Izbicki G, Chavko R, Banauch GI, et al. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. Chest. 2007;131:1414-1423.
20. Eishi Y, Suga M, Ishige I, et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J Clin Microbiol. 2002;40:198-204.
21. Valeyre D, Prasse A, Nunes H, et al. Sarcoidosis. Lancet. 2014;383:1155-1167.
22. Crouser ED, Maier LA, Wilson KC, et al. Diagnosis and detection of sarcoidosis. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;201:e26-51.
23. Judson MA, ed. Pulmonary Sarcoidosis: A Guide for the Practicing Clinician. Springer; 2014.
24. Govender P, Berman JS. The diagnosis of sarcoidosis. Clin Chest Med. 2015;36:585-602.
25. Valeyre D, Bernaudin J-F, Uzunhan Y, et al. Clinical presentation of sarcoidosis and diagnostic work-up. Semin Resp Crit Care Med. 2014;35:336-351.
26. Judson MA. The clinical features of sarcoidosis: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:63-78.
27. Wanat KA, Rosenbach M. Cutaneous sarcoidosis. Clin Chest Med. 2015;36:685-702.
28. Culver DA, Neto ML, Moss BP, et al. Neurosarcoidosis. Semin Resp Crit Care Med. 2017;38:499-513.
29. Pasadhika S, Rosenbaum JT. Ocular sarcoidosis. Clin Chest Med. 2015;36:669-683.
30. Sayah DM, Bradfield JS, Moriarty JM, et al. Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin Resp Crit Care Med. 2017;38:477-498.
31. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Resp Crit Care. 2012;164:1885-1889.
32. Keijsers RG, Veltkamp M, Grutters JC. Chest imaging. Clin Chest Med. 2015;36:603-619.
33. Scadding J. Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Brit Med J. 1961;2:1165-1172.
34. Miller B, Putman C. The chest radiograph and sarcoidosis. Reevaluation of the chest radiograph in assessing activity of sarcoidosis: a preliminary communication. Sarcoidosis. 1985;2:85-90.
35. Loddenkemper R, Kloppenborg A, Schoenfeld N, et al. Clinical findings in 715 patients with newly detected pulmonary sarcoidosis--results of a cooperative study in former West Germany and Switzerland. WATL Study Group. Wissenschaftliche Arbeitsgemeinschaft für die Therapie von Lungenkrankheitan. Sarcoidosis Vasc Diffuse Lung Dis. 1998;15:178-182.
36. Calandriello L, Walsh SLF. Imaging for sarcoidosis. Semin Resp Crit Care Med. 2017;38:417-436.
37. Gupta D, Dadhwal DS, Agarwal R, et al. Endobronchial ultrasound-guided transbronchial needle aspiration vs conventional transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 2014;146:547-556.
38. Baydur A. Recent developments in the physiological assessment of sarcoidosis: clinical implications. Curr Opin Pulm Med. 2012;18:499-505.
39. Jamilloux Y, Maucort-Boulch D, Kerever S, et al. Sarcoidosis-related mortality in France: a multiple-cause-of-death analysis. Eur Respir J. 2016;48:1700-1709.
40. Gupta R, Baughman RP. Advanced pulmonary sarcoidosis. Semin Respir Crit Care Med. 2020;41:700-715.
41. Rossi G, Cavazza A, Colby TV. Pathology of sarcoidosis. Clin Rev Allergy Immunol. 2015;49:36-44.
42. Hansell D, Milne D, Wilsher M, et al. Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology. 1998;209:697-704.
43. Judson MA, Thompson BW, Rabin DL, et al. The diagnostic pathway to sarcoidosis. Chest. 2003;123:406-412.
44. Müller NL, Mawson JB, Mathieson JR, et al. Sarcoidosis: correlation of extent of disease at CT with clinical, functional, and radiographic findings. Radiology. 1989;171:613-618.
45. Harrison BDW, Shaylor JM, Stokes TC, et al. Airflow limitation in sarcoidosis—a study of pulmonary function in 107 patients with newly diagnosed disease. Resp Med. 1991;85:59-64.
46. Polychronopoulos VS, Prakash UBS. Airway Involvement in sarcoidosis. Chest. 2009;136:1371-1380.
47. Chambellan A, Turbie P, Nunes H, et al. Endoluminal stenosis of proximal bronchi in sarcoidosis: bronchoscopy, function, and evolution. Chest. 2005;127:472-481.
48. Handa T, Nagai S, Fushimi Y, et al. Clinical and radiographic indices associated with airflow limitation in patients with sarcoidosis. Chest. 2006;130:1851-1856.
49. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
50. Zappala CJ, Desai SR, Copley SJ, et al. Accuracy of individual variables in the monitoring of long-term change in pulmonary sarcoidosis as judged by serial high-resolution CT scan data. Chest. 2014;145:101-107.
51. Gafà G, Sverzellati N, Bonati E, et al. Follow-up in pulmonary sarcoidosis: comparison between HRCT and pulmonary function tests. Radiol Med. 2012;117:968-978.
52. Gerke AK. Morbidity and mortality in sarcoidosis. Curr Opin Pulm Med. 2014;20:472-478.
53. Kearney GD, Obi ON, Maddipati V, et al. Sarcoidosis deaths in the United States: 1999–2016. Respir Med. 2019;149:30-35.
54. Baughman RP, Judson M, Wells A. The indications for the treatment of sarcoidosis: Wells Law. Sarcoidosis Vasc Diffuse Lung Dis. 2017;34:280-282.
55. Nagai S, Shigematsu M, Hamada K, et al. Clinical courses and prognoses of pulmonary sarcoidosis. Curr Opin Pulm Med. 1999;5:293-298.
56. Neville E, Walker AN, James DG. Prognostic factors predicting the outcome of sarcoidosis: an analysis of 818 patients. Q J Med. 1983;52:525-533.
57. Bradley B, Branley HM, Egan JJ, et al. Interstitial lung disease guideline: the British Thoracic Society in collaboration with the Thoracic Society of Australia and the Irish Thoracic Society. Thorax. 2008;63(suppl 5):v1-v58.
58. Pietinalho A, Tukiainen P, Haahtela T, et al. Oral prednisolone followed by inhaled budesonide in newly diagnosed pulmonary sarcoidosis: a double-blind, placebo-controlled multicenter study. Finnish Pulmonary Sarcoidosis Group. Chest. 1999;116:424-431.
59. Oberstein A, von Zitzewitz H, Schweden F, et al. Non invasive evaluation of the inflammatory activity in sarcoidosis with high-resolution computed tomography. Sarcoidosis Vasc Diffuse Lung Dis. 1997;14:65-72.
60. Gibson G, Prescott RJ, Muers MF, et al. British Thoracic Society Sarcoidosis study: effects of long term corticosteroid treatment. Thorax. 1996;51:238-247.
61. Baughman RP, Nunes H. Therapy for sarcoidosis: evidence-based recommendations. Expert Rev Clin Immunol. 2012;8:95-103.
62. Pietinalho A, Tukiainen P, Haahtela T, et al. Early treatment of stage II sarcoidosis improves 5-year pulmonary function. Chest. 2002;121:24-31.
63. Rahaghi FF, Baughman RP, Saketkoo LA, et al. Delphi consensus recommendations for a treatment algorithm in pulmonary sarcoidosis. Eur Respir Rev. 2020;29:190146.
64. Baughman RP, Iannuzzi MC, Lower EE, et al. Use of fluticasone in acute symptomatic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2002;19:198-204.
65. du Bois RM, Greenhalgh PM, Southcott AM, et al. Randomized trial of inhaled fluticasone propionate in chronic stable pulmonary sarcoidosis: a pilot study. Eur Respir J. 1999;13:1345-1350.
66. Milman N, Graudal N, Grode G, Munch E. No effect of high‐dose inhaled steroids in pulmonary sarcoidosis: a double‐blind, placebo‐controlled study. J Intern Med. 1994;236:285-290.
67. Baughman RP, Winget DB, Lower EE. Methotrexate is steroid sparing in acute sarcoidosis: results of a double blind, randomized trial. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17:60-66.
68. Vorselaars ADM, Wuyts WA, Vorselaars VMM, et al. Methotrexate vs azathioprine in second-line therapy of sarcoidosis. Chest. 2013;144:805-812.
69. Sahoo D, Bandyopadhyay D, Xu M, et al. Effectiveness and safety of leflunomide for pulmonary and extrapulmonary sarcoidosis. Eur Respir J. 2011;38:1145-1150.
70. Baughman RP, Drent M, Kavuru M, et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am J Resp Crit Care Med . 2006;174:795-802.
71. Rossman MD, Newman LS, Baughman RP, et al. A double-blinded, randomized, placebo-controlled trial of infliximab in subjects with active pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2006;23:201-208.
72. Selroos O, Sellergren T. Corticosteroid therapy of pulmonary sarcoidosis. A prospective evaluation of alternate day and daily dosage in stage II disease. Scand J Respir Dis . 1979;60:215-221.
73. Israel HL, Fouts DW, Beggs RA. A controlled trial of prednisone treatment of sarcoidosis. Am Rev Respir Dis . 1973;107:609-614.
74. Hamzeh N, Voelker A, Forssén A, et al. Efficacy of mycophenolate mofetil in sarcoidosis. Respir Med . 2014;108:1663-1669.
75. Brill A-K, Ott SR, Geiser T. Effect and safety of mycophenolate mofetil in chronic pulmonary sarcoidosis: a retrospective study. Respiration . 2013;86:376-383.
76. Baughman RP, Lower EE. Leflunomide for chronic sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2004;21:43-48.
77. Sweiss NJ, Noth I, Mirsaeidi M, et al. Efficacy results of a 52-week trial of adalimumab in the treatment of refractory sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2014;31:46-54.
78. Sweiss NJ, Lower EE, Mirsaeidi M, et al. Rituximab in the treatment of refractory pulmonary sarcoidosis. Eur Respir J . 2014;43:1525-1528.
79. Thatayatikom A, Thatayatikom S, White AJ. Infliximab treatment for severe granulomatous disease in common variable immunodeficiency: a case report and review of the literature. Ann Allergy Asthma Immunol . 2005;95:293-300.
80. Drake WP, Oswald-Richter K, Richmond BW, et al. Oral antimycobacterial therapy in chronic cutaneous sarcoidosis: a randomized, single-masked, placebo-controlled study. Jama Dermatol . 2013;149:1040-1049.
81. Drake WP, Richmond BW, Oswald-Richter K, et al. Effects of broad-spectrum antimycobacterial therapy on chronic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2013;30:201-211.
82. Baughman RP, Culver DA, Cordova FC, et al. Bosentan for sarcoidosis-associated pulmonary hypertension: a double-blind placebo controlled randomized trial. Chest . 2014;145:810-817.
83. Baughman RP, Shlobin OA, Wells AU, et al. Clinical features of sarcoidosis associated pulmonary hypertension: results of a multi-national registry. Respir Med . 2018;139:72-78.
84. Fisher KA, Serlin DM, Wilson KC, et al. Sarcoidosis-associated pulmonary hypertension outcome with long-term epoprostenol treatment. Chest . 2006;130:1481-1488.
85. Baughman RP, Judson MA, Lower EE, et al. Inhaled iloprost for sarcoidosis associated pulmonary hypertension. Sarcoidosis Vasc Diffuse Lung Dis . 2009;26:110-120.
86. King TE, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med . 2014;370:2083-2092.
Sarcoidosis is a multisystem inflammatory disease of unclear etiology that primarily affects the lungs. It can occur at any age but usually develops before the age of 50 years, with an initial peak incidence at 20 to 29 years and a second peak incidence after 50 years of age, especially among women in Scandinavia and Japan.1 Sarcoidosis affects men and women of all racial and ethnic groups throughout the world, but differences based on race, sex, and geography are noted.1
The highest rates are reported in northern European and African-American individuals, particularly in women.1,2 The adjusted annual incidence of sarcoidosis among African Americans is approximately 3 times that among White Americans3 and is more likely to be chronic and fatal in African Americans.3 The disease can be familial with a possible recessive inheritance mode with incomplete penetrance.4 Risk of sarcoidosis in monozygotic twins appears to be 80 times greater than that in the general population, which supports genetic factors accounting for two-thirds of disease susceptibility.5
Likely factors in the development of sarcoidosis
The exact cause of sarcoidosis is unknown, but we have insights into its pathogenesis and potential triggers.1,6-9 Genes involved are being identified: class I and II human leukocyte antigen (HLA) molecules are most consistently associated with risk of sarcoidosis. Environmental exposures can activate the innate immune system and precondition a susceptible individual to react to potential causative antigens in a highly polarized, antigen-specific Th1 immune response. The epithelioid granulomatous response involves local proinflammatory cytokine production and enhanced T-cell immunity at sites of inflammation.10 Granulomas generally form to confine pathogens, restrict inflammation, and protect surrounding tissue.11-13
ACCESS (A Case Control Etiologic Study of Sarcoidosis) identified several environmental exposures such as chemicals used in the agriculture industry, mold or mildew, and musty odors at work.14 Tobacco use was not associated with sarcoidosis.14 Recent studies have shown positive associations with service in the US Navy,15 metal working,16 firefighting,17 the handling of building supplies,18 and onsite exposure while assisting in rescue efforts at the World Trade Center disaster.19 Other data support the likelihood that specific environmental exposures associated with microbe-rich environments modestly increase the risk of sarcoidosis.14 Mycobacterial and propionibacterial DNA and RNA are potentially associated with sarcoidosis.20
Clinical manifestations are nonspecific
The diagnosis of sarcoidosis can be difficult and delayed due to diverse organ involvement and nonspecific presentations. TABLE 121-31 shows the diverse manifestations in a patient with suspected sarcoidosis. Around 50% of the patients are asymptomatic.23,24 Sarcoidosis is a diagnosis of exclusion, starting with a detailed history to rule out infections, occupational or environmental exposures, malignancies, and other possible disorders (TABLE 2).22

Diagnostic work-up

Radiologic studies
Chest x-ray (CXR) provides diagnostic and prognostic information in the evaluation of sarcoidosis using the Scadding classification system (FIGURE 1).21,25,32,33 Interobserver variability, especially between stages II and III and III and IV is the major limitation of this system.32 At presentation, radiographs are abnormal in approximately 90% of patients.34 Lymphadenopathy is the most common radiographic abnormality, occurring in more than two-thirds of cases, and pulmonary opacities (nodules and reticulation) with a middle to upper lobe predilection are present in 20% to 50% of patients.1,31,35 The nodules vary in size and can coalesce and cause alveolar collapse, thus producing consolidation.36 Linear opacities radiating laterally from the hilum into the middle and upper zones are characteristic in fibrotic disease.

Continue to: High-resoluton computed tomography
High-resolution computed tomography (HRCT). Micronodules in a perilymphatic distribution with upper lobe predominance combined with subcarinal and symmetrical hilar lymph node enlargement is practically diagnostic of sarcoidosis in the right clinical context. TABLE 321,23,25,32 and FIGURE 221,23,25,32 summarize the common CT chest findings of sarcoidosis.

Advanced imaging such as (18)F-fluorodeoxyglucose positron emission tomography (PET) and magnetic resonance imaging (MRI) are used in specialized settings for advanced pulmonary, cardiac, or neurosarcoidosis.

Tissue biopsy
Skin lesions (other than erythema nodosum), eye lesions, and peripheral lymph nodes are considered the safest extrapulmonary locations for biopsy.21,25 If pulmonary infiltrates or lymphadenopathy are present, or if extrapulmonary biopsy sites are not available, then flexible bronchoscopy with biopsy is the mainstay for tissue sampling.25
Bronchoalveolar lavage (BAL), transbronchial biopsy (TBB), endobronchial biopsy (EBB), and endobronchial ultrasound (EBUS) are invaluable modalities that have reduced the need for open lung biopsy. BAL in sarcoidosis can show lymphocytosis > 15% (nonspecific) and a CD4:CD8 lymphocyte ratio > 3.5 (specificity > 90%).21,22 TBB is more sensitive than EBB; however, sensitivity overall is heightened when both of them are combined. The advent of EBUS has increased the safety and efficiency of needle aspiration of mediastinal lymph nodes. Diagnostic yield of EBUS (~80%) is superior to that with TBB and EBB (~50%), especially in stage I and II sarcoidosis.37 The combination of EBUS with TBB improves the diagnostic yield to ~90%.37
The decision to obtain biopsy samples hinges on the nature of clinical and radiologic findings (FIGURE 3).22,25,26

Continue to: Laboratory studies
Laboratory studies
Multiple abnormalities may be seen in sarcoidosis, and specific lab tests may help support a diagnosis of sarcoidosis or detect organ-specific disease activity (TABLE 4).22,23,25,38 However, no consistently accurate biomarkers exist for use in clinical practice. An angiotensin-converting enzyme (ACE) level greater than 2 times the upper limit of normal may be helpful; however, sensitivity remains low, and genetic polymorphisms can influence the ACE level.25 Biomarkers sometimes used to assess disease activity are serum interleukin-2 receptor, neopterin, chitotriosidase, lysozyme, KL-6 glycoprotein, and amyloid A.21

Additional tests to assess specific features or organ involvement
Pulmonary function testing (PFT) is reviewed in detail below under “pulmonary sarcoidosis.”
Electrocardiogram (EKG)/transthoracic echocardiogram (TTE). EKG abnormalities—conduction disturbances, arrhythmias, or nonspecific ST segment and T-wave changes—are the most common nonspecific findings.30 TTE findings are also nonspecific but have value in assessing cardiac chamber size and function and myocardial involvement. TTE is indeed the most common screening modality for sarcoidosis-associated pulmonary hypertension (SAPH), which is definitively diagnosed by right heart catheterization (RHC). Further evaluation for cardiac sarcoidosis can be done with cardiac MRI or fluorodeoxyglucose PET in specialized settings.
Lumbar puncture (LP) may reveal lymphocytic infiltration in suspected neurosarcoidosis, but the finding is nonspecific and can reflect infection or malignancy. Oligoclonal bands may also be seen in about one-third of neurosarcoidosis cases, and it is imperative to rule out multiple sclerosis.28
Pulmonary sarcoidosis
Pulmonary sarcoidosis accounts for most of the morbidity, mortality, and health care use associated with sarcoidosis.39,40
Continue to: Pathology of early and advanced pulmonary sarcoidosis
Pathology of early and advanced pulmonary sarcoidosis
Sarcoidosis is characterized by coalescing, tightly clustered, nonnecrotizing granulomas in the lung (FIGURE 4), most often located along the lymphatic routes of the pleura, interlobular septa, and bronchovascular bundles.41 Granulomas contain epithelioid cells or multinucleated giant cells surrounded by a chronic lymphocytic infiltrate. Typically, intracytoplasmic inclusions, such as Schaumann bodies, asteroid bodies, and blue bodies of calcium oxalates are noted within giant cells.

In chronic disease, lymphocytic infiltrate vanishes and granulomas tend to become increasingly fibrotic and enlarge to form hyalinized nodules rich with densely eosinophilic collagen. In 10% to 30% of cases, the lungs undergo progressive fibrosis.40 Nonresolving inflammation appears to be the major cause of fibrosis and the peribronchovascular localization leading to marked bronchial distortion.
Clinical features, monitoring, and outcomes
Pulmonary involvement occurs in most patients with sarcoidosis, and subclinical pulmonary disease is generally present, even when extrathoracic manifestations predominate.23 Dry cough, dyspnea, and chest discomfort are the most common symptoms. Chest auscultation is usually unremarkable. Wheezing is more common in those with fibrosis and is attributed to airway-centric fibrosis.42 There is often a substantial delay between the onset of symptoms and the diagnosis of pulmonary sarcoidosis, as symptoms are nonspecific and might be mistaken for more common pulmonary diseases, such as asthma or chronic bronchitis.43
Since sarcoidosis can affect pulmonary parenchyma, interstitium, large and small airways, pulmonary vasculature, and respiratory muscles, the pattern of lung function impairment on PFT varies from normal to obstruction, restriction, isolated diffusion defect, or a combination of these. The typical physiologic abnormality is a restrictive ventilatory defect with a decreased diffusing capacity of the lung for carbon monoxide (DLCO). Extent of disease seen on HRCT correlates with level of restriction.44 Airway obstruction can be multifactorial and due to airway distortion (more likely to occur in fibrotic lung disease) and luminal disease.45-48 The 6-minute walk test and DLCO can also aid in the diagnosis of SAPH and advanced parenchymal lung disease.
While monitoring is done clinically and with testing (PFT and imaging) as needed, the optimal approach is unclear. Nevertheless, longitudinal monitoring with testing may provide useful management and prognostic information.40 Pulmonary function can remain stable in fibrotic sarcoidosis over extended periods and actually can improve in some patients.49 Serial spirometry, particularly forced vital capacity, is the most reliable tool for monitoring; when a decline in measurement occurs, chest radiography can elucidate the mechanism.50,51
Continue to: Because sarcoidosis is a multisystem disease...
Because sarcoidosis is a multisystem disease, caution needs to be exercised when evaluating a patient’s new or worsening respiratory symptoms to accurately determine the cause of symptoms and direct therapy accordingly. In addition to refractory inflammatory pulmonary disease, airway disease, infection, fibrosis, and SAPH, one needs to consider extrapulmonary involvement or complications such as cardiac or neurologic disease, musculoskeletal disease, depression, or fatigue. Adverse medication effects, deconditioning, or unrelated (or possibly related) disorders (eg pulmonary embolism) may be to blame.
Determining prognosis
Prognosis of sarcoidosis varies and depends on epidemiologic factors, clinical presentation, and course, as well as specific organ involvement. Patients may develop life-threatening pulmonary, cardiac, or neurologic complications. End-stage disease may require organ transplantation for eligible patients.
Most patients with pulmonary sarcoidosis experience clinical remission with minimal residual organ impairment and a favorable long-term outcome. Advanced pulmonary disease (known as APS) occurs in a small proportion of patients with sarcoidosis but accounts for most of the poor outcomes in sarcoidosis.40 APS is variably defined, but it generally includes pulmonary fibrosis, SAPH, and respiratory infection.
One percent to 5% of patients with sarcoidosis die from complications, and mortality is higher in women and African Americans.52 Mortality and morbidity may be increasing.53 The reasons behind these trends are unclear but could include true increases in disease incidence, better detection rates, greater severity of disease, or an aging population. Increased hospitalizations and health care use might be due to organ damage from granulomatous inflammation (and resultant fibrosis), complications associated with treatment, and psychosocial effects of the disease/treatment.
Management
Management consists primarily of anti-inflammatory or immunosuppressive therapies but can also include measures to address specific complications (such as fatigue) and organ transplant, as well as efforts to counter adverse medication effects. Other supportive and preventive measures may include, on a case-by-case basis, oxygen supplementation, vaccinations, or pulmonary rehabilitation. Details of these are found in other, more in-depth reviews on treatment; we will briefly review anti-inflammatory therapy, which forms the cornerstone of treatment in most patients with sarcoidosis.
Continue to: General approach to treatment decisions
General approach to treatment decisions. Anti-inflammatory therapy is used to reduce granulomatous inflammation, thereby preserving organ function and reducing symptoms. A decision to begin treatment is one shared with the patient and is based on symptoms and potential danger of organ system failure.54 Patients who are symptomatic or have progressive disease or physiologic impairment are generally candidates for treatment. Monitoring usually suffices for those who have minimal symptoms, stable disease, and preserved organ function.
Patients with pulmonary sarcoidosis at CXR stage 0 should not receive treatment, given that large, randomized trials have shown no meaningful benefit and that these patients have a high likelihood of spontaneous remission and excellent long-term prognosis.55-58 However, a subgroup of patients classified as stage 0/I on CXR may show parenchymal disease on HRCT,59 and, if more symptomatic, could be considered for treatment. For patients with stage II to IV pulmonary sarcoidosis with symptoms, there is good evidence that treatment may improve lung function and reduce dyspnea and fatigue.57,60-62
Corticosteroids are first-line treatment for most patients. Based on expert opinion, treatment of pulmonary sarcoidosis is generally started with oral prednisone (or an equivalent corticosteroid). A starting dose of 20 to 40 mg/d generally is sufficient for most patients. If the patient responds to initial treatment, prednisone dose is tapered over a period of months. If symptoms worsen during tapering, the minimum effective dose is maintained without further attempts at tapering. Treatment is continued for at least 3 to 6 months but it might be needed for longer durations; unfortunately, evidence-based guidelines are lacking.63 Once the patient goes into remission, close monitoring is done for possible relapses. Inhaled corticosteroids alone have not reduced symptoms or improved lung function in patients with pulmonary sarcoidosis.64-66
Steroid-sparing agents are added for many patients. For patients receiving chronic prednisone therapy (≥ 10 mg for > 6 months), steroid-sparing agents are considered to minimize the adverse effects of steroids or to better control the inflammatory activity of sarcoidosis. These agents must be carefully selected, and clinical and laboratory monitoring need to be done throughout therapy. TABLE 558,64,67-81

The management might be complicated for extrapulmonary, multi-organ, and advanced sarcoidosis (advanced pulmonary sarcoidosis, cardiac disease, neurosarcoidosis, lupus pernio, etc) when specialized testing, as well as a combination of corticosteroids and steroid-sparing agents (with higher doses or prolonged courses), might be needed. This should be performed at an expert sarcoidosis center, ideally in a multidisciplinary setting involving pulmonologists and/or rheumatologists, chest radiologists, and specialists as indicated, based on specific organ involvement.
Continue to: Research and future directions
Research and future directions
Key goals for research are identifying more accurate biomarkers of disease, improving diagnosis of multi-organ disease, determining validated endpoints of clinical trials in sarcoidosis, and developing treatments for refractory cases.
There is optimism and opportunity in the field of sarcoidosis overall. An example of an advancement is in the area of APS, as the severity and importance of this phenotype has been better understood. Worldwide registries and trials of pulmonary vasodilator therapy (bosentan, sildenafil, epoprostenol, and inhaled iloprost) in patients with SAPH without left ventricular dysfunction are promising.82-85 However, no benefit in survival has been shown.
RioSAPH is a double-blind, placebo-controlled trial of Riociguat (a stimulator of soluble guanylate cyclase) for SAPH (NCT02625558) that is closed to enrollment and undergoing data review. Similarly, results of the phase IV study of pirfenidone, an antifibrotic agent that was shown to decrease disease progression and deaths in idiopathic pulmonary fibrosis,86 are awaited in the near future.
Other potential directions being explored are multicenter patient registries and randomized controlled trials, analyses of existing databases, use of biobanking, and patient-centered outcome measures. Hopefully, the care of patients with sarcoidosis will become more evidence based with ongoing and upcoming research in this field.
CORRESPONDENCE
Rohit Gupta, MBBS, FCCP, 3401 North Broad Street, 7 Parkinson Pavilion, Philadelphia, PA 19140; [email protected]
Sarcoidosis is a multisystem inflammatory disease of unclear etiology that primarily affects the lungs. It can occur at any age but usually develops before the age of 50 years, with an initial peak incidence at 20 to 29 years and a second peak incidence after 50 years of age, especially among women in Scandinavia and Japan.1 Sarcoidosis affects men and women of all racial and ethnic groups throughout the world, but differences based on race, sex, and geography are noted.1
The highest rates are reported in northern European and African-American individuals, particularly in women.1,2 The adjusted annual incidence of sarcoidosis among African Americans is approximately 3 times that among White Americans3 and is more likely to be chronic and fatal in African Americans.3 The disease can be familial with a possible recessive inheritance mode with incomplete penetrance.4 Risk of sarcoidosis in monozygotic twins appears to be 80 times greater than that in the general population, which supports genetic factors accounting for two-thirds of disease susceptibility.5
Likely factors in the development of sarcoidosis
The exact cause of sarcoidosis is unknown, but we have insights into its pathogenesis and potential triggers.1,6-9 Genes involved are being identified: class I and II human leukocyte antigen (HLA) molecules are most consistently associated with risk of sarcoidosis. Environmental exposures can activate the innate immune system and precondition a susceptible individual to react to potential causative antigens in a highly polarized, antigen-specific Th1 immune response. The epithelioid granulomatous response involves local proinflammatory cytokine production and enhanced T-cell immunity at sites of inflammation.10 Granulomas generally form to confine pathogens, restrict inflammation, and protect surrounding tissue.11-13
ACCESS (A Case Control Etiologic Study of Sarcoidosis) identified several environmental exposures such as chemicals used in the agriculture industry, mold or mildew, and musty odors at work.14 Tobacco use was not associated with sarcoidosis.14 Recent studies have shown positive associations with service in the US Navy,15 metal working,16 firefighting,17 the handling of building supplies,18 and onsite exposure while assisting in rescue efforts at the World Trade Center disaster.19 Other data support the likelihood that specific environmental exposures associated with microbe-rich environments modestly increase the risk of sarcoidosis.14 Mycobacterial and propionibacterial DNA and RNA are potentially associated with sarcoidosis.20
Clinical manifestations are nonspecific
The diagnosis of sarcoidosis can be difficult and delayed due to diverse organ involvement and nonspecific presentations. TABLE 121-31 shows the diverse manifestations in a patient with suspected sarcoidosis. Around 50% of the patients are asymptomatic.23,24 Sarcoidosis is a diagnosis of exclusion, starting with a detailed history to rule out infections, occupational or environmental exposures, malignancies, and other possible disorders (TABLE 2).22

Diagnostic work-up

Radiologic studies
Chest x-ray (CXR) provides diagnostic and prognostic information in the evaluation of sarcoidosis using the Scadding classification system (FIGURE 1).21,25,32,33 Interobserver variability, especially between stages II and III and III and IV is the major limitation of this system.32 At presentation, radiographs are abnormal in approximately 90% of patients.34 Lymphadenopathy is the most common radiographic abnormality, occurring in more than two-thirds of cases, and pulmonary opacities (nodules and reticulation) with a middle to upper lobe predilection are present in 20% to 50% of patients.1,31,35 The nodules vary in size and can coalesce and cause alveolar collapse, thus producing consolidation.36 Linear opacities radiating laterally from the hilum into the middle and upper zones are characteristic in fibrotic disease.

Continue to: High-resoluton computed tomography
High-resolution computed tomography (HRCT). Micronodules in a perilymphatic distribution with upper lobe predominance combined with subcarinal and symmetrical hilar lymph node enlargement is practically diagnostic of sarcoidosis in the right clinical context. TABLE 321,23,25,32 and FIGURE 221,23,25,32 summarize the common CT chest findings of sarcoidosis.

Advanced imaging such as (18)F-fluorodeoxyglucose positron emission tomography (PET) and magnetic resonance imaging (MRI) are used in specialized settings for advanced pulmonary, cardiac, or neurosarcoidosis.

Tissue biopsy
Skin lesions (other than erythema nodosum), eye lesions, and peripheral lymph nodes are considered the safest extrapulmonary locations for biopsy.21,25 If pulmonary infiltrates or lymphadenopathy are present, or if extrapulmonary biopsy sites are not available, then flexible bronchoscopy with biopsy is the mainstay for tissue sampling.25
Bronchoalveolar lavage (BAL), transbronchial biopsy (TBB), endobronchial biopsy (EBB), and endobronchial ultrasound (EBUS) are invaluable modalities that have reduced the need for open lung biopsy. BAL in sarcoidosis can show lymphocytosis > 15% (nonspecific) and a CD4:CD8 lymphocyte ratio > 3.5 (specificity > 90%).21,22 TBB is more sensitive than EBB; however, sensitivity overall is heightened when both of them are combined. The advent of EBUS has increased the safety and efficiency of needle aspiration of mediastinal lymph nodes. Diagnostic yield of EBUS (~80%) is superior to that with TBB and EBB (~50%), especially in stage I and II sarcoidosis.37 The combination of EBUS with TBB improves the diagnostic yield to ~90%.37
The decision to obtain biopsy samples hinges on the nature of clinical and radiologic findings (FIGURE 3).22,25,26

Continue to: Laboratory studies
Laboratory studies
Multiple abnormalities may be seen in sarcoidosis, and specific lab tests may help support a diagnosis of sarcoidosis or detect organ-specific disease activity (TABLE 4).22,23,25,38 However, no consistently accurate biomarkers exist for use in clinical practice. An angiotensin-converting enzyme (ACE) level greater than 2 times the upper limit of normal may be helpful; however, sensitivity remains low, and genetic polymorphisms can influence the ACE level.25 Biomarkers sometimes used to assess disease activity are serum interleukin-2 receptor, neopterin, chitotriosidase, lysozyme, KL-6 glycoprotein, and amyloid A.21

Additional tests to assess specific features or organ involvement
Pulmonary function testing (PFT) is reviewed in detail below under “pulmonary sarcoidosis.”
Electrocardiogram (EKG)/transthoracic echocardiogram (TTE). EKG abnormalities—conduction disturbances, arrhythmias, or nonspecific ST segment and T-wave changes—are the most common nonspecific findings.30 TTE findings are also nonspecific but have value in assessing cardiac chamber size and function and myocardial involvement. TTE is indeed the most common screening modality for sarcoidosis-associated pulmonary hypertension (SAPH), which is definitively diagnosed by right heart catheterization (RHC). Further evaluation for cardiac sarcoidosis can be done with cardiac MRI or fluorodeoxyglucose PET in specialized settings.
Lumbar puncture (LP) may reveal lymphocytic infiltration in suspected neurosarcoidosis, but the finding is nonspecific and can reflect infection or malignancy. Oligoclonal bands may also be seen in about one-third of neurosarcoidosis cases, and it is imperative to rule out multiple sclerosis.28
Pulmonary sarcoidosis
Pulmonary sarcoidosis accounts for most of the morbidity, mortality, and health care use associated with sarcoidosis.39,40
Continue to: Pathology of early and advanced pulmonary sarcoidosis
Pathology of early and advanced pulmonary sarcoidosis
Sarcoidosis is characterized by coalescing, tightly clustered, nonnecrotizing granulomas in the lung (FIGURE 4), most often located along the lymphatic routes of the pleura, interlobular septa, and bronchovascular bundles.41 Granulomas contain epithelioid cells or multinucleated giant cells surrounded by a chronic lymphocytic infiltrate. Typically, intracytoplasmic inclusions, such as Schaumann bodies, asteroid bodies, and blue bodies of calcium oxalates are noted within giant cells.

In chronic disease, lymphocytic infiltrate vanishes and granulomas tend to become increasingly fibrotic and enlarge to form hyalinized nodules rich with densely eosinophilic collagen. In 10% to 30% of cases, the lungs undergo progressive fibrosis.40 Nonresolving inflammation appears to be the major cause of fibrosis and the peribronchovascular localization leading to marked bronchial distortion.
Clinical features, monitoring, and outcomes
Pulmonary involvement occurs in most patients with sarcoidosis, and subclinical pulmonary disease is generally present, even when extrathoracic manifestations predominate.23 Dry cough, dyspnea, and chest discomfort are the most common symptoms. Chest auscultation is usually unremarkable. Wheezing is more common in those with fibrosis and is attributed to airway-centric fibrosis.42 There is often a substantial delay between the onset of symptoms and the diagnosis of pulmonary sarcoidosis, as symptoms are nonspecific and might be mistaken for more common pulmonary diseases, such as asthma or chronic bronchitis.43
Since sarcoidosis can affect pulmonary parenchyma, interstitium, large and small airways, pulmonary vasculature, and respiratory muscles, the pattern of lung function impairment on PFT varies from normal to obstruction, restriction, isolated diffusion defect, or a combination of these. The typical physiologic abnormality is a restrictive ventilatory defect with a decreased diffusing capacity of the lung for carbon monoxide (DLCO). Extent of disease seen on HRCT correlates with level of restriction.44 Airway obstruction can be multifactorial and due to airway distortion (more likely to occur in fibrotic lung disease) and luminal disease.45-48 The 6-minute walk test and DLCO can also aid in the diagnosis of SAPH and advanced parenchymal lung disease.
While monitoring is done clinically and with testing (PFT and imaging) as needed, the optimal approach is unclear. Nevertheless, longitudinal monitoring with testing may provide useful management and prognostic information.40 Pulmonary function can remain stable in fibrotic sarcoidosis over extended periods and actually can improve in some patients.49 Serial spirometry, particularly forced vital capacity, is the most reliable tool for monitoring; when a decline in measurement occurs, chest radiography can elucidate the mechanism.50,51
Continue to: Because sarcoidosis is a multisystem disease...
Because sarcoidosis is a multisystem disease, caution needs to be exercised when evaluating a patient’s new or worsening respiratory symptoms to accurately determine the cause of symptoms and direct therapy accordingly. In addition to refractory inflammatory pulmonary disease, airway disease, infection, fibrosis, and SAPH, one needs to consider extrapulmonary involvement or complications such as cardiac or neurologic disease, musculoskeletal disease, depression, or fatigue. Adverse medication effects, deconditioning, or unrelated (or possibly related) disorders (eg pulmonary embolism) may be to blame.
Determining prognosis
Prognosis of sarcoidosis varies and depends on epidemiologic factors, clinical presentation, and course, as well as specific organ involvement. Patients may develop life-threatening pulmonary, cardiac, or neurologic complications. End-stage disease may require organ transplantation for eligible patients.
Most patients with pulmonary sarcoidosis experience clinical remission with minimal residual organ impairment and a favorable long-term outcome. Advanced pulmonary disease (known as APS) occurs in a small proportion of patients with sarcoidosis but accounts for most of the poor outcomes in sarcoidosis.40 APS is variably defined, but it generally includes pulmonary fibrosis, SAPH, and respiratory infection.
One percent to 5% of patients with sarcoidosis die from complications, and mortality is higher in women and African Americans.52 Mortality and morbidity may be increasing.53 The reasons behind these trends are unclear but could include true increases in disease incidence, better detection rates, greater severity of disease, or an aging population. Increased hospitalizations and health care use might be due to organ damage from granulomatous inflammation (and resultant fibrosis), complications associated with treatment, and psychosocial effects of the disease/treatment.
Management
Management consists primarily of anti-inflammatory or immunosuppressive therapies but can also include measures to address specific complications (such as fatigue) and organ transplant, as well as efforts to counter adverse medication effects. Other supportive and preventive measures may include, on a case-by-case basis, oxygen supplementation, vaccinations, or pulmonary rehabilitation. Details of these are found in other, more in-depth reviews on treatment; we will briefly review anti-inflammatory therapy, which forms the cornerstone of treatment in most patients with sarcoidosis.
Continue to: General approach to treatment decisions
General approach to treatment decisions. Anti-inflammatory therapy is used to reduce granulomatous inflammation, thereby preserving organ function and reducing symptoms. A decision to begin treatment is one shared with the patient and is based on symptoms and potential danger of organ system failure.54 Patients who are symptomatic or have progressive disease or physiologic impairment are generally candidates for treatment. Monitoring usually suffices for those who have minimal symptoms, stable disease, and preserved organ function.
Patients with pulmonary sarcoidosis at CXR stage 0 should not receive treatment, given that large, randomized trials have shown no meaningful benefit and that these patients have a high likelihood of spontaneous remission and excellent long-term prognosis.55-58 However, a subgroup of patients classified as stage 0/I on CXR may show parenchymal disease on HRCT,59 and, if more symptomatic, could be considered for treatment. For patients with stage II to IV pulmonary sarcoidosis with symptoms, there is good evidence that treatment may improve lung function and reduce dyspnea and fatigue.57,60-62
Corticosteroids are first-line treatment for most patients. Based on expert opinion, treatment of pulmonary sarcoidosis is generally started with oral prednisone (or an equivalent corticosteroid). A starting dose of 20 to 40 mg/d generally is sufficient for most patients. If the patient responds to initial treatment, prednisone dose is tapered over a period of months. If symptoms worsen during tapering, the minimum effective dose is maintained without further attempts at tapering. Treatment is continued for at least 3 to 6 months but it might be needed for longer durations; unfortunately, evidence-based guidelines are lacking.63 Once the patient goes into remission, close monitoring is done for possible relapses. Inhaled corticosteroids alone have not reduced symptoms or improved lung function in patients with pulmonary sarcoidosis.64-66
Steroid-sparing agents are added for many patients. For patients receiving chronic prednisone therapy (≥ 10 mg for > 6 months), steroid-sparing agents are considered to minimize the adverse effects of steroids or to better control the inflammatory activity of sarcoidosis. These agents must be carefully selected, and clinical and laboratory monitoring need to be done throughout therapy. TABLE 558,64,67-81

The management might be complicated for extrapulmonary, multi-organ, and advanced sarcoidosis (advanced pulmonary sarcoidosis, cardiac disease, neurosarcoidosis, lupus pernio, etc) when specialized testing, as well as a combination of corticosteroids and steroid-sparing agents (with higher doses or prolonged courses), might be needed. This should be performed at an expert sarcoidosis center, ideally in a multidisciplinary setting involving pulmonologists and/or rheumatologists, chest radiologists, and specialists as indicated, based on specific organ involvement.
Continue to: Research and future directions
Research and future directions
Key goals for research are identifying more accurate biomarkers of disease, improving diagnosis of multi-organ disease, determining validated endpoints of clinical trials in sarcoidosis, and developing treatments for refractory cases.
There is optimism and opportunity in the field of sarcoidosis overall. An example of an advancement is in the area of APS, as the severity and importance of this phenotype has been better understood. Worldwide registries and trials of pulmonary vasodilator therapy (bosentan, sildenafil, epoprostenol, and inhaled iloprost) in patients with SAPH without left ventricular dysfunction are promising.82-85 However, no benefit in survival has been shown.
RioSAPH is a double-blind, placebo-controlled trial of Riociguat (a stimulator of soluble guanylate cyclase) for SAPH (NCT02625558) that is closed to enrollment and undergoing data review. Similarly, results of the phase IV study of pirfenidone, an antifibrotic agent that was shown to decrease disease progression and deaths in idiopathic pulmonary fibrosis,86 are awaited in the near future.
Other potential directions being explored are multicenter patient registries and randomized controlled trials, analyses of existing databases, use of biobanking, and patient-centered outcome measures. Hopefully, the care of patients with sarcoidosis will become more evidence based with ongoing and upcoming research in this field.
CORRESPONDENCE
Rohit Gupta, MBBS, FCCP, 3401 North Broad Street, 7 Parkinson Pavilion, Philadelphia, PA 19140; [email protected]
1. Costabel U, Hunninghake G. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur Respir J. 1999;14:735-737.
2. Hillerdal G, Nöu E, Osterman K, et al. Sarcoidosis: epidemiology and prognosis. A 15-year European study. Am Rev Respir Dis. 1984;130:29-32.
3. Mirsaeidi M, Machado RF, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015;147:438-449.
4. Rybicki BA, Iannuzzi MC, Frederick MM, et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Resp Crit Care Med. 2001;164:2085-2091.
5. Sverrild A, Backer V, Kyvik KO, et al. Heredity in sarcoidosis:a registry-based twin study. Thorax. 2008;63:894.
6. Vuyst P, Dumortier P, Schandené L, et al. Sarcoidlike lung granulomatosis induced by aluminum dusts. Am Rev Respir Dis. 1987;135:493-497.
7. Werfel U, Schneider J, Rödelsperger K, et al. Sarcoid granulomatosis after zirconium exposure with multiple organ involvement. European Respir J. 1998;12:750.
8. Newman KL, Newman LS. Occupational causes of sarcoidosis. Curr Opin Allergy Clin Immunol. 2012;12:145-150.
9. Zissel G, Müller-Quernheim J. Specific antigen(s) in sarcoidosis:a link to autoimmunity? Eur Respir J. 2016;47:707-709.
10. Chen ES, Moller DR. Etiology of sarcoidosis. Clin Chest Med. 2008;29:365-377.
11. Agostini C, Adami F, Semenzato G. New pathogenetic insights into the sarcoid granuloma. Curr Opin Rheumatol. 2000;12:71-76.
12. Valentonyte R, Hampe J, Huse K, et al. Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat Genet. 2005;37:357-364.
13. Rybicki BA, Walewski JL, Maliarik MJ, et al. The BTNL2 gene and sarcoidosis susceptibility in African Americans and Whites. Am J Hum Genet. 2005;77:491-499.
14. Newman LS, Rose CS, Bresnitz EA, et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am J Respir Crit Care Med. 2004;170:1324-1330.
15. Gorham ED, Garland CF, Garland FC, et al. Trends and occupational associations in incidence of hospitalized pulmonary sarcoidosis and other lung diseases in Navy personnel: a 27-year historical prospective study, 1975-2001. Chest. 2004;126:1431-1438.
16. Kucera GP, Rybicki BA, Kirkey KL, et al. Occupational risk factors for sarcoidosis in African-American siblings. Chest. 2003;123:1527-1535.
17. Prezant DJ, Dhala A, Goldstein A, et al. The incidence, prevalence, and severity of sarcoidosis in New York City firefighters. Chest. 1999;116:1183-1193.
18. Barnard J, Rose C, Newman L, et al. Job and industry classifications associated with sarcoidosis in A Case–Control Etiologic Study of Sarcoidosis (ACCESS). J Occup Environ Med. 2005;47:226-234.
19. Izbicki G, Chavko R, Banauch GI, et al. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. Chest. 2007;131:1414-1423.
20. Eishi Y, Suga M, Ishige I, et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J Clin Microbiol. 2002;40:198-204.
21. Valeyre D, Prasse A, Nunes H, et al. Sarcoidosis. Lancet. 2014;383:1155-1167.
22. Crouser ED, Maier LA, Wilson KC, et al. Diagnosis and detection of sarcoidosis. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;201:e26-51.
23. Judson MA, ed. Pulmonary Sarcoidosis: A Guide for the Practicing Clinician. Springer; 2014.
24. Govender P, Berman JS. The diagnosis of sarcoidosis. Clin Chest Med. 2015;36:585-602.
25. Valeyre D, Bernaudin J-F, Uzunhan Y, et al. Clinical presentation of sarcoidosis and diagnostic work-up. Semin Resp Crit Care Med. 2014;35:336-351.
26. Judson MA. The clinical features of sarcoidosis: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:63-78.
27. Wanat KA, Rosenbach M. Cutaneous sarcoidosis. Clin Chest Med. 2015;36:685-702.
28. Culver DA, Neto ML, Moss BP, et al. Neurosarcoidosis. Semin Resp Crit Care Med. 2017;38:499-513.
29. Pasadhika S, Rosenbaum JT. Ocular sarcoidosis. Clin Chest Med. 2015;36:669-683.
30. Sayah DM, Bradfield JS, Moriarty JM, et al. Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin Resp Crit Care Med. 2017;38:477-498.
31. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Resp Crit Care. 2012;164:1885-1889.
32. Keijsers RG, Veltkamp M, Grutters JC. Chest imaging. Clin Chest Med. 2015;36:603-619.
33. Scadding J. Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Brit Med J. 1961;2:1165-1172.
34. Miller B, Putman C. The chest radiograph and sarcoidosis. Reevaluation of the chest radiograph in assessing activity of sarcoidosis: a preliminary communication. Sarcoidosis. 1985;2:85-90.
35. Loddenkemper R, Kloppenborg A, Schoenfeld N, et al. Clinical findings in 715 patients with newly detected pulmonary sarcoidosis--results of a cooperative study in former West Germany and Switzerland. WATL Study Group. Wissenschaftliche Arbeitsgemeinschaft für die Therapie von Lungenkrankheitan. Sarcoidosis Vasc Diffuse Lung Dis. 1998;15:178-182.
36. Calandriello L, Walsh SLF. Imaging for sarcoidosis. Semin Resp Crit Care Med. 2017;38:417-436.
37. Gupta D, Dadhwal DS, Agarwal R, et al. Endobronchial ultrasound-guided transbronchial needle aspiration vs conventional transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 2014;146:547-556.
38. Baydur A. Recent developments in the physiological assessment of sarcoidosis: clinical implications. Curr Opin Pulm Med. 2012;18:499-505.
39. Jamilloux Y, Maucort-Boulch D, Kerever S, et al. Sarcoidosis-related mortality in France: a multiple-cause-of-death analysis. Eur Respir J. 2016;48:1700-1709.
40. Gupta R, Baughman RP. Advanced pulmonary sarcoidosis. Semin Respir Crit Care Med. 2020;41:700-715.
41. Rossi G, Cavazza A, Colby TV. Pathology of sarcoidosis. Clin Rev Allergy Immunol. 2015;49:36-44.
42. Hansell D, Milne D, Wilsher M, et al. Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology. 1998;209:697-704.
43. Judson MA, Thompson BW, Rabin DL, et al. The diagnostic pathway to sarcoidosis. Chest. 2003;123:406-412.
44. Müller NL, Mawson JB, Mathieson JR, et al. Sarcoidosis: correlation of extent of disease at CT with clinical, functional, and radiographic findings. Radiology. 1989;171:613-618.
45. Harrison BDW, Shaylor JM, Stokes TC, et al. Airflow limitation in sarcoidosis—a study of pulmonary function in 107 patients with newly diagnosed disease. Resp Med. 1991;85:59-64.
46. Polychronopoulos VS, Prakash UBS. Airway Involvement in sarcoidosis. Chest. 2009;136:1371-1380.
47. Chambellan A, Turbie P, Nunes H, et al. Endoluminal stenosis of proximal bronchi in sarcoidosis: bronchoscopy, function, and evolution. Chest. 2005;127:472-481.
48. Handa T, Nagai S, Fushimi Y, et al. Clinical and radiographic indices associated with airflow limitation in patients with sarcoidosis. Chest. 2006;130:1851-1856.
49. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
50. Zappala CJ, Desai SR, Copley SJ, et al. Accuracy of individual variables in the monitoring of long-term change in pulmonary sarcoidosis as judged by serial high-resolution CT scan data. Chest. 2014;145:101-107.
51. Gafà G, Sverzellati N, Bonati E, et al. Follow-up in pulmonary sarcoidosis: comparison between HRCT and pulmonary function tests. Radiol Med. 2012;117:968-978.
52. Gerke AK. Morbidity and mortality in sarcoidosis. Curr Opin Pulm Med. 2014;20:472-478.
53. Kearney GD, Obi ON, Maddipati V, et al. Sarcoidosis deaths in the United States: 1999–2016. Respir Med. 2019;149:30-35.
54. Baughman RP, Judson M, Wells A. The indications for the treatment of sarcoidosis: Wells Law. Sarcoidosis Vasc Diffuse Lung Dis. 2017;34:280-282.
55. Nagai S, Shigematsu M, Hamada K, et al. Clinical courses and prognoses of pulmonary sarcoidosis. Curr Opin Pulm Med. 1999;5:293-298.
56. Neville E, Walker AN, James DG. Prognostic factors predicting the outcome of sarcoidosis: an analysis of 818 patients. Q J Med. 1983;52:525-533.
57. Bradley B, Branley HM, Egan JJ, et al. Interstitial lung disease guideline: the British Thoracic Society in collaboration with the Thoracic Society of Australia and the Irish Thoracic Society. Thorax. 2008;63(suppl 5):v1-v58.
58. Pietinalho A, Tukiainen P, Haahtela T, et al. Oral prednisolone followed by inhaled budesonide in newly diagnosed pulmonary sarcoidosis: a double-blind, placebo-controlled multicenter study. Finnish Pulmonary Sarcoidosis Group. Chest. 1999;116:424-431.
59. Oberstein A, von Zitzewitz H, Schweden F, et al. Non invasive evaluation of the inflammatory activity in sarcoidosis with high-resolution computed tomography. Sarcoidosis Vasc Diffuse Lung Dis. 1997;14:65-72.
60. Gibson G, Prescott RJ, Muers MF, et al. British Thoracic Society Sarcoidosis study: effects of long term corticosteroid treatment. Thorax. 1996;51:238-247.
61. Baughman RP, Nunes H. Therapy for sarcoidosis: evidence-based recommendations. Expert Rev Clin Immunol. 2012;8:95-103.
62. Pietinalho A, Tukiainen P, Haahtela T, et al. Early treatment of stage II sarcoidosis improves 5-year pulmonary function. Chest. 2002;121:24-31.
63. Rahaghi FF, Baughman RP, Saketkoo LA, et al. Delphi consensus recommendations for a treatment algorithm in pulmonary sarcoidosis. Eur Respir Rev. 2020;29:190146.
64. Baughman RP, Iannuzzi MC, Lower EE, et al. Use of fluticasone in acute symptomatic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2002;19:198-204.
65. du Bois RM, Greenhalgh PM, Southcott AM, et al. Randomized trial of inhaled fluticasone propionate in chronic stable pulmonary sarcoidosis: a pilot study. Eur Respir J. 1999;13:1345-1350.
66. Milman N, Graudal N, Grode G, Munch E. No effect of high‐dose inhaled steroids in pulmonary sarcoidosis: a double‐blind, placebo‐controlled study. J Intern Med. 1994;236:285-290.
67. Baughman RP, Winget DB, Lower EE. Methotrexate is steroid sparing in acute sarcoidosis: results of a double blind, randomized trial. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17:60-66.
68. Vorselaars ADM, Wuyts WA, Vorselaars VMM, et al. Methotrexate vs azathioprine in second-line therapy of sarcoidosis. Chest. 2013;144:805-812.
69. Sahoo D, Bandyopadhyay D, Xu M, et al. Effectiveness and safety of leflunomide for pulmonary and extrapulmonary sarcoidosis. Eur Respir J. 2011;38:1145-1150.
70. Baughman RP, Drent M, Kavuru M, et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am J Resp Crit Care Med . 2006;174:795-802.
71. Rossman MD, Newman LS, Baughman RP, et al. A double-blinded, randomized, placebo-controlled trial of infliximab in subjects with active pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2006;23:201-208.
72. Selroos O, Sellergren T. Corticosteroid therapy of pulmonary sarcoidosis. A prospective evaluation of alternate day and daily dosage in stage II disease. Scand J Respir Dis . 1979;60:215-221.
73. Israel HL, Fouts DW, Beggs RA. A controlled trial of prednisone treatment of sarcoidosis. Am Rev Respir Dis . 1973;107:609-614.
74. Hamzeh N, Voelker A, Forssén A, et al. Efficacy of mycophenolate mofetil in sarcoidosis. Respir Med . 2014;108:1663-1669.
75. Brill A-K, Ott SR, Geiser T. Effect and safety of mycophenolate mofetil in chronic pulmonary sarcoidosis: a retrospective study. Respiration . 2013;86:376-383.
76. Baughman RP, Lower EE. Leflunomide for chronic sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2004;21:43-48.
77. Sweiss NJ, Noth I, Mirsaeidi M, et al. Efficacy results of a 52-week trial of adalimumab in the treatment of refractory sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2014;31:46-54.
78. Sweiss NJ, Lower EE, Mirsaeidi M, et al. Rituximab in the treatment of refractory pulmonary sarcoidosis. Eur Respir J . 2014;43:1525-1528.
79. Thatayatikom A, Thatayatikom S, White AJ. Infliximab treatment for severe granulomatous disease in common variable immunodeficiency: a case report and review of the literature. Ann Allergy Asthma Immunol . 2005;95:293-300.
80. Drake WP, Oswald-Richter K, Richmond BW, et al. Oral antimycobacterial therapy in chronic cutaneous sarcoidosis: a randomized, single-masked, placebo-controlled study. Jama Dermatol . 2013;149:1040-1049.
81. Drake WP, Richmond BW, Oswald-Richter K, et al. Effects of broad-spectrum antimycobacterial therapy on chronic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2013;30:201-211.
82. Baughman RP, Culver DA, Cordova FC, et al. Bosentan for sarcoidosis-associated pulmonary hypertension: a double-blind placebo controlled randomized trial. Chest . 2014;145:810-817.
83. Baughman RP, Shlobin OA, Wells AU, et al. Clinical features of sarcoidosis associated pulmonary hypertension: results of a multi-national registry. Respir Med . 2018;139:72-78.
84. Fisher KA, Serlin DM, Wilson KC, et al. Sarcoidosis-associated pulmonary hypertension outcome with long-term epoprostenol treatment. Chest . 2006;130:1481-1488.
85. Baughman RP, Judson MA, Lower EE, et al. Inhaled iloprost for sarcoidosis associated pulmonary hypertension. Sarcoidosis Vasc Diffuse Lung Dis . 2009;26:110-120.
86. King TE, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med . 2014;370:2083-2092.
1. Costabel U, Hunninghake G. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World Association for Sarcoidosis and Other Granulomatous Disorders. Eur Respir J. 1999;14:735-737.
2. Hillerdal G, Nöu E, Osterman K, et al. Sarcoidosis: epidemiology and prognosis. A 15-year European study. Am Rev Respir Dis. 1984;130:29-32.
3. Mirsaeidi M, Machado RF, Schraufnagel D, et al. Racial difference in sarcoidosis mortality in the United States. Chest. 2015;147:438-449.
4. Rybicki BA, Iannuzzi MC, Frederick MM, et al. Familial aggregation of sarcoidosis. A case-control etiologic study of sarcoidosis (ACCESS). Am J Resp Crit Care Med. 2001;164:2085-2091.
5. Sverrild A, Backer V, Kyvik KO, et al. Heredity in sarcoidosis:a registry-based twin study. Thorax. 2008;63:894.
6. Vuyst P, Dumortier P, Schandené L, et al. Sarcoidlike lung granulomatosis induced by aluminum dusts. Am Rev Respir Dis. 1987;135:493-497.
7. Werfel U, Schneider J, Rödelsperger K, et al. Sarcoid granulomatosis after zirconium exposure with multiple organ involvement. European Respir J. 1998;12:750.
8. Newman KL, Newman LS. Occupational causes of sarcoidosis. Curr Opin Allergy Clin Immunol. 2012;12:145-150.
9. Zissel G, Müller-Quernheim J. Specific antigen(s) in sarcoidosis:a link to autoimmunity? Eur Respir J. 2016;47:707-709.
10. Chen ES, Moller DR. Etiology of sarcoidosis. Clin Chest Med. 2008;29:365-377.
11. Agostini C, Adami F, Semenzato G. New pathogenetic insights into the sarcoid granuloma. Curr Opin Rheumatol. 2000;12:71-76.
12. Valentonyte R, Hampe J, Huse K, et al. Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat Genet. 2005;37:357-364.
13. Rybicki BA, Walewski JL, Maliarik MJ, et al. The BTNL2 gene and sarcoidosis susceptibility in African Americans and Whites. Am J Hum Genet. 2005;77:491-499.
14. Newman LS, Rose CS, Bresnitz EA, et al. A case control etiologic study of sarcoidosis: environmental and occupational risk factors. Am J Respir Crit Care Med. 2004;170:1324-1330.
15. Gorham ED, Garland CF, Garland FC, et al. Trends and occupational associations in incidence of hospitalized pulmonary sarcoidosis and other lung diseases in Navy personnel: a 27-year historical prospective study, 1975-2001. Chest. 2004;126:1431-1438.
16. Kucera GP, Rybicki BA, Kirkey KL, et al. Occupational risk factors for sarcoidosis in African-American siblings. Chest. 2003;123:1527-1535.
17. Prezant DJ, Dhala A, Goldstein A, et al. The incidence, prevalence, and severity of sarcoidosis in New York City firefighters. Chest. 1999;116:1183-1193.
18. Barnard J, Rose C, Newman L, et al. Job and industry classifications associated with sarcoidosis in A Case–Control Etiologic Study of Sarcoidosis (ACCESS). J Occup Environ Med. 2005;47:226-234.
19. Izbicki G, Chavko R, Banauch GI, et al. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. Chest. 2007;131:1414-1423.
20. Eishi Y, Suga M, Ishige I, et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J Clin Microbiol. 2002;40:198-204.
21. Valeyre D, Prasse A, Nunes H, et al. Sarcoidosis. Lancet. 2014;383:1155-1167.
22. Crouser ED, Maier LA, Wilson KC, et al. Diagnosis and detection of sarcoidosis. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;201:e26-51.
23. Judson MA, ed. Pulmonary Sarcoidosis: A Guide for the Practicing Clinician. Springer; 2014.
24. Govender P, Berman JS. The diagnosis of sarcoidosis. Clin Chest Med. 2015;36:585-602.
25. Valeyre D, Bernaudin J-F, Uzunhan Y, et al. Clinical presentation of sarcoidosis and diagnostic work-up. Semin Resp Crit Care Med. 2014;35:336-351.
26. Judson MA. The clinical features of sarcoidosis: a comprehensive review. Clin Rev Allergy Immunol. 2015;49:63-78.
27. Wanat KA, Rosenbach M. Cutaneous sarcoidosis. Clin Chest Med. 2015;36:685-702.
28. Culver DA, Neto ML, Moss BP, et al. Neurosarcoidosis. Semin Resp Crit Care Med. 2017;38:499-513.
29. Pasadhika S, Rosenbaum JT. Ocular sarcoidosis. Clin Chest Med. 2015;36:669-683.
30. Sayah DM, Bradfield JS, Moriarty JM, et al. Cardiac involvement in sarcoidosis: evolving concepts in diagnosis and treatment. Semin Resp Crit Care Med. 2017;38:477-498.
31. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Resp Crit Care. 2012;164:1885-1889.
32. Keijsers RG, Veltkamp M, Grutters JC. Chest imaging. Clin Chest Med. 2015;36:603-619.
33. Scadding J. Prognosis of intrathoracic sarcoidosis in England. A review of 136 cases after five years’ observation. Brit Med J. 1961;2:1165-1172.
34. Miller B, Putman C. The chest radiograph and sarcoidosis. Reevaluation of the chest radiograph in assessing activity of sarcoidosis: a preliminary communication. Sarcoidosis. 1985;2:85-90.
35. Loddenkemper R, Kloppenborg A, Schoenfeld N, et al. Clinical findings in 715 patients with newly detected pulmonary sarcoidosis--results of a cooperative study in former West Germany and Switzerland. WATL Study Group. Wissenschaftliche Arbeitsgemeinschaft für die Therapie von Lungenkrankheitan. Sarcoidosis Vasc Diffuse Lung Dis. 1998;15:178-182.
36. Calandriello L, Walsh SLF. Imaging for sarcoidosis. Semin Resp Crit Care Med. 2017;38:417-436.
37. Gupta D, Dadhwal DS, Agarwal R, et al. Endobronchial ultrasound-guided transbronchial needle aspiration vs conventional transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 2014;146:547-556.
38. Baydur A. Recent developments in the physiological assessment of sarcoidosis: clinical implications. Curr Opin Pulm Med. 2012;18:499-505.
39. Jamilloux Y, Maucort-Boulch D, Kerever S, et al. Sarcoidosis-related mortality in France: a multiple-cause-of-death analysis. Eur Respir J. 2016;48:1700-1709.
40. Gupta R, Baughman RP. Advanced pulmonary sarcoidosis. Semin Respir Crit Care Med. 2020;41:700-715.
41. Rossi G, Cavazza A, Colby TV. Pathology of sarcoidosis. Clin Rev Allergy Immunol. 2015;49:36-44.
42. Hansell D, Milne D, Wilsher M, et al. Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology. 1998;209:697-704.
43. Judson MA, Thompson BW, Rabin DL, et al. The diagnostic pathway to sarcoidosis. Chest. 2003;123:406-412.
44. Müller NL, Mawson JB, Mathieson JR, et al. Sarcoidosis: correlation of extent of disease at CT with clinical, functional, and radiographic findings. Radiology. 1989;171:613-618.
45. Harrison BDW, Shaylor JM, Stokes TC, et al. Airflow limitation in sarcoidosis—a study of pulmonary function in 107 patients with newly diagnosed disease. Resp Med. 1991;85:59-64.
46. Polychronopoulos VS, Prakash UBS. Airway Involvement in sarcoidosis. Chest. 2009;136:1371-1380.
47. Chambellan A, Turbie P, Nunes H, et al. Endoluminal stenosis of proximal bronchi in sarcoidosis: bronchoscopy, function, and evolution. Chest. 2005;127:472-481.
48. Handa T, Nagai S, Fushimi Y, et al. Clinical and radiographic indices associated with airflow limitation in patients with sarcoidosis. Chest. 2006;130:1851-1856.
49. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
50. Zappala CJ, Desai SR, Copley SJ, et al. Accuracy of individual variables in the monitoring of long-term change in pulmonary sarcoidosis as judged by serial high-resolution CT scan data. Chest. 2014;145:101-107.
51. Gafà G, Sverzellati N, Bonati E, et al. Follow-up in pulmonary sarcoidosis: comparison between HRCT and pulmonary function tests. Radiol Med. 2012;117:968-978.
52. Gerke AK. Morbidity and mortality in sarcoidosis. Curr Opin Pulm Med. 2014;20:472-478.
53. Kearney GD, Obi ON, Maddipati V, et al. Sarcoidosis deaths in the United States: 1999–2016. Respir Med. 2019;149:30-35.
54. Baughman RP, Judson M, Wells A. The indications for the treatment of sarcoidosis: Wells Law. Sarcoidosis Vasc Diffuse Lung Dis. 2017;34:280-282.
55. Nagai S, Shigematsu M, Hamada K, et al. Clinical courses and prognoses of pulmonary sarcoidosis. Curr Opin Pulm Med. 1999;5:293-298.
56. Neville E, Walker AN, James DG. Prognostic factors predicting the outcome of sarcoidosis: an analysis of 818 patients. Q J Med. 1983;52:525-533.
57. Bradley B, Branley HM, Egan JJ, et al. Interstitial lung disease guideline: the British Thoracic Society in collaboration with the Thoracic Society of Australia and the Irish Thoracic Society. Thorax. 2008;63(suppl 5):v1-v58.
58. Pietinalho A, Tukiainen P, Haahtela T, et al. Oral prednisolone followed by inhaled budesonide in newly diagnosed pulmonary sarcoidosis: a double-blind, placebo-controlled multicenter study. Finnish Pulmonary Sarcoidosis Group. Chest. 1999;116:424-431.
59. Oberstein A, von Zitzewitz H, Schweden F, et al. Non invasive evaluation of the inflammatory activity in sarcoidosis with high-resolution computed tomography. Sarcoidosis Vasc Diffuse Lung Dis. 1997;14:65-72.
60. Gibson G, Prescott RJ, Muers MF, et al. British Thoracic Society Sarcoidosis study: effects of long term corticosteroid treatment. Thorax. 1996;51:238-247.
61. Baughman RP, Nunes H. Therapy for sarcoidosis: evidence-based recommendations. Expert Rev Clin Immunol. 2012;8:95-103.
62. Pietinalho A, Tukiainen P, Haahtela T, et al. Early treatment of stage II sarcoidosis improves 5-year pulmonary function. Chest. 2002;121:24-31.
63. Rahaghi FF, Baughman RP, Saketkoo LA, et al. Delphi consensus recommendations for a treatment algorithm in pulmonary sarcoidosis. Eur Respir Rev. 2020;29:190146.
64. Baughman RP, Iannuzzi MC, Lower EE, et al. Use of fluticasone in acute symptomatic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2002;19:198-204.
65. du Bois RM, Greenhalgh PM, Southcott AM, et al. Randomized trial of inhaled fluticasone propionate in chronic stable pulmonary sarcoidosis: a pilot study. Eur Respir J. 1999;13:1345-1350.
66. Milman N, Graudal N, Grode G, Munch E. No effect of high‐dose inhaled steroids in pulmonary sarcoidosis: a double‐blind, placebo‐controlled study. J Intern Med. 1994;236:285-290.
67. Baughman RP, Winget DB, Lower EE. Methotrexate is steroid sparing in acute sarcoidosis: results of a double blind, randomized trial. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17:60-66.
68. Vorselaars ADM, Wuyts WA, Vorselaars VMM, et al. Methotrexate vs azathioprine in second-line therapy of sarcoidosis. Chest. 2013;144:805-812.
69. Sahoo D, Bandyopadhyay D, Xu M, et al. Effectiveness and safety of leflunomide for pulmonary and extrapulmonary sarcoidosis. Eur Respir J. 2011;38:1145-1150.
70. Baughman RP, Drent M, Kavuru M, et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am J Resp Crit Care Med . 2006;174:795-802.
71. Rossman MD, Newman LS, Baughman RP, et al. A double-blinded, randomized, placebo-controlled trial of infliximab in subjects with active pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2006;23:201-208.
72. Selroos O, Sellergren T. Corticosteroid therapy of pulmonary sarcoidosis. A prospective evaluation of alternate day and daily dosage in stage II disease. Scand J Respir Dis . 1979;60:215-221.
73. Israel HL, Fouts DW, Beggs RA. A controlled trial of prednisone treatment of sarcoidosis. Am Rev Respir Dis . 1973;107:609-614.
74. Hamzeh N, Voelker A, Forssén A, et al. Efficacy of mycophenolate mofetil in sarcoidosis. Respir Med . 2014;108:1663-1669.
75. Brill A-K, Ott SR, Geiser T. Effect and safety of mycophenolate mofetil in chronic pulmonary sarcoidosis: a retrospective study. Respiration . 2013;86:376-383.
76. Baughman RP, Lower EE. Leflunomide for chronic sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2004;21:43-48.
77. Sweiss NJ, Noth I, Mirsaeidi M, et al. Efficacy results of a 52-week trial of adalimumab in the treatment of refractory sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2014;31:46-54.
78. Sweiss NJ, Lower EE, Mirsaeidi M, et al. Rituximab in the treatment of refractory pulmonary sarcoidosis. Eur Respir J . 2014;43:1525-1528.
79. Thatayatikom A, Thatayatikom S, White AJ. Infliximab treatment for severe granulomatous disease in common variable immunodeficiency: a case report and review of the literature. Ann Allergy Asthma Immunol . 2005;95:293-300.
80. Drake WP, Oswald-Richter K, Richmond BW, et al. Oral antimycobacterial therapy in chronic cutaneous sarcoidosis: a randomized, single-masked, placebo-controlled study. Jama Dermatol . 2013;149:1040-1049.
81. Drake WP, Richmond BW, Oswald-Richter K, et al. Effects of broad-spectrum antimycobacterial therapy on chronic pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis . 2013;30:201-211.
82. Baughman RP, Culver DA, Cordova FC, et al. Bosentan for sarcoidosis-associated pulmonary hypertension: a double-blind placebo controlled randomized trial. Chest . 2014;145:810-817.
83. Baughman RP, Shlobin OA, Wells AU, et al. Clinical features of sarcoidosis associated pulmonary hypertension: results of a multi-national registry. Respir Med . 2018;139:72-78.
84. Fisher KA, Serlin DM, Wilson KC, et al. Sarcoidosis-associated pulmonary hypertension outcome with long-term epoprostenol treatment. Chest . 2006;130:1481-1488.
85. Baughman RP, Judson MA, Lower EE, et al. Inhaled iloprost for sarcoidosis associated pulmonary hypertension. Sarcoidosis Vasc Diffuse Lung Dis . 2009;26:110-120.
86. King TE, Bradford WZ, Castro-Bernardini S, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med . 2014;370:2083-2092.
PRACTICE RECOMMENDATIONS
› Consider biopsy to aid in diagnosing sarcoidosis; it may be avoided with a high clinical suspicion for sarcoidosis (eg, Löfgren syndrome, lupus pernio, or Heerfordt syndrome). C
› Rule out alternative diagnoses such as infection, malignancy, collagen vascular disease, and vasculitis. C
› Identify extra-pulmonary organ involvement, as clinically indicated, by screening with a baseline eye examination; complete blood count; creatinine, alkaline phosphatase, and calcium levels; electrocardiogram, and other organ-specific studies. C
› Make a patient-centered decision whether to begin antiinflammatory treatment based on symptomatology and risk of organ failure or death. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Combo provides ‘broad benefit’ across NHL subtypes
The trial, dubbed CHRONOS-3, is the first to report “a broad benefit” across histologic subtypes of relapsed, indolent NHL, and the results are “essentially a long-awaited proof of concept” for combining a PI3K inhibitor with rituximab, according to investigator Matthew Matasar, MD, of Memorial Sloan Kettering Cancer Center in New York.
Dr. Matasar presented results from CHRONOS-3 at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT001). The findings were simultaneously published in The Lancet Oncology.
Charles Swanton, MBPhD, of the Francis Crick Institute and UCL Cancer Institute in London, called the results “strongly positive” and said the copanlisib-rituximab combination is “a potential new treatment option” for indolent NHL in patients with a long remission after first-line therapy or those who are unfit for chemotherapy.
Dr. Swanton noted, however, that “one should also bear in mind” the serious adverse events (AEs) seen with copanlisib, particularly hypertension and hyperglycemia. When asked about these AEs, Dr. Matasar said he thinks the combination would be appropriate for patients who meet the study criteria as long as they don’t have severe baseline diabetes or uncontrolled hypertension.
Patient and treatment details
The study included 458 patients with CD20-positive, relapsed, indolent, B-cell NHL. Subtypes included follicular lymphoma (n = 275), marginal zone lymphoma (n = 95), small lymphocytic lymphoma (n = 50), and lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia (n = 38).
All patients were progression free and treatment free before their relapse for at least 12 months after their last rituximab-containing regimen, or at least 6 months before relapse if they were unwilling or unable to undergo chemotherapy.
The patients’ median age was 63 years, and just over half of them were men (52%). About 37% of patients had a history of hypertension at baseline, and about 15% had a history of diabetes.
Patients were randomized to receive copanlisib plus rituximab (n = 307) or rituximab plus placebo (n = 151). Copanlisib was given at 60 mg IV on days 1, 8, and 15 of a 28-day cycle. In both arms, rituximab was given at 375 mg/m2 on days 1, 8, 15, and 22 during cycle 1 and on day 1 of cycles 3, 5, 7, and 9.
Progression-free survival benefit
At a median follow-up of 19.2 months, the median progression-free survival (PFS) was 21.5 months in the copanlisib-rituximab arm and 13.8 months in the placebo-rituximab arm (hazard ratio, 0.52; P < .0001).
The PFS advantage with copanlisib was seen across subtypes:
- Follicular lymphoma – 22.2 months vs. 18.7 months (P = .001)
- Small lymphocytic lymphoma – 14.2 months vs. 5.7 months (P < .0001)
- Marginal zone lymphoma – 22.1 months vs. 11.5 months (P = .012)
- Lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia – 33.4 months vs. 16.6 months (P = .054)
The PFS difference among patients with lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia was likely not statistically significant because of the small sample size, Dr. Matasar said.
He reported that the overall response rate was 81% with copanlisib-rituximab, including a 34% complete response rate. In the placebo arm, the overall response rate was 48%, and 15% of patients had a complete response.
The median overall survival was not estimable in either treatment arm. At a median follow-up of 30.1 months, 14% of patients in the copanlisib arm and 13.2% of patients in the placebo arm had died.
More than double the rate of serious AEs
The rate of serious treatment-emergent AEs was 47.2% in the combination arm and 18.5% in the placebo arm.
There were six grade 5 treatment-emergent AEs in the combination arm. One of these – pneumonitis – was deemed treatment related. There was one treatment-emergent death in the placebo arm.
Hyperglycemia and hypertension were the most common grade 3/4 treatment-emergent AEs with the combination. Diarrhea, nausea, neutropenia, and pyrexia were also more frequent with the combination than with rituximab-placebo.
More than half of patients in the combination arm (56.3%) developed grade 3/4 hyperglycemia. In the placebo arm, the incidence of grade 3 hyperglycemia was 8.2%, and there was no grade 4 hyperglycemia.
Rates of grade 3 hypertension were 39.7% in the combination arm and 8.9% in the placebo arm. There was no grade 4 hypertension.
In the combination arm, 2.6% of patients stopped treatment because of hyperglycemia and 0.7% stopped because of hypertension.
Any-grade pneumonitis occurred in 6.8% of patients in the combination arm and 1.4% of those in the placebo arm. The rate of grade 3/4 pneumonitis was 2.7% in the copanlisib arm, and the rate of grade 3 pneumonitis was 0.7% in the placebo arm.
The study was funded by Bayer, the company developing copanlisib. Dr. Matasar disclosed relationships with Bayer, its subsidiaries, and Roche/Genentech. Dr. Swanton disclosed relationships with numerous companies, including Pfizer, Novartis, and GlaxoSmithKline.
The trial, dubbed CHRONOS-3, is the first to report “a broad benefit” across histologic subtypes of relapsed, indolent NHL, and the results are “essentially a long-awaited proof of concept” for combining a PI3K inhibitor with rituximab, according to investigator Matthew Matasar, MD, of Memorial Sloan Kettering Cancer Center in New York.
Dr. Matasar presented results from CHRONOS-3 at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT001). The findings were simultaneously published in The Lancet Oncology.
Charles Swanton, MBPhD, of the Francis Crick Institute and UCL Cancer Institute in London, called the results “strongly positive” and said the copanlisib-rituximab combination is “a potential new treatment option” for indolent NHL in patients with a long remission after first-line therapy or those who are unfit for chemotherapy.
Dr. Swanton noted, however, that “one should also bear in mind” the serious adverse events (AEs) seen with copanlisib, particularly hypertension and hyperglycemia. When asked about these AEs, Dr. Matasar said he thinks the combination would be appropriate for patients who meet the study criteria as long as they don’t have severe baseline diabetes or uncontrolled hypertension.
Patient and treatment details
The study included 458 patients with CD20-positive, relapsed, indolent, B-cell NHL. Subtypes included follicular lymphoma (n = 275), marginal zone lymphoma (n = 95), small lymphocytic lymphoma (n = 50), and lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia (n = 38).
All patients were progression free and treatment free before their relapse for at least 12 months after their last rituximab-containing regimen, or at least 6 months before relapse if they were unwilling or unable to undergo chemotherapy.
The patients’ median age was 63 years, and just over half of them were men (52%). About 37% of patients had a history of hypertension at baseline, and about 15% had a history of diabetes.
Patients were randomized to receive copanlisib plus rituximab (n = 307) or rituximab plus placebo (n = 151). Copanlisib was given at 60 mg IV on days 1, 8, and 15 of a 28-day cycle. In both arms, rituximab was given at 375 mg/m2 on days 1, 8, 15, and 22 during cycle 1 and on day 1 of cycles 3, 5, 7, and 9.
Progression-free survival benefit
At a median follow-up of 19.2 months, the median progression-free survival (PFS) was 21.5 months in the copanlisib-rituximab arm and 13.8 months in the placebo-rituximab arm (hazard ratio, 0.52; P < .0001).
The PFS advantage with copanlisib was seen across subtypes:
- Follicular lymphoma – 22.2 months vs. 18.7 months (P = .001)
- Small lymphocytic lymphoma – 14.2 months vs. 5.7 months (P < .0001)
- Marginal zone lymphoma – 22.1 months vs. 11.5 months (P = .012)
- Lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia – 33.4 months vs. 16.6 months (P = .054)
The PFS difference among patients with lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia was likely not statistically significant because of the small sample size, Dr. Matasar said.
He reported that the overall response rate was 81% with copanlisib-rituximab, including a 34% complete response rate. In the placebo arm, the overall response rate was 48%, and 15% of patients had a complete response.
The median overall survival was not estimable in either treatment arm. At a median follow-up of 30.1 months, 14% of patients in the copanlisib arm and 13.2% of patients in the placebo arm had died.
More than double the rate of serious AEs
The rate of serious treatment-emergent AEs was 47.2% in the combination arm and 18.5% in the placebo arm.
There were six grade 5 treatment-emergent AEs in the combination arm. One of these – pneumonitis – was deemed treatment related. There was one treatment-emergent death in the placebo arm.
Hyperglycemia and hypertension were the most common grade 3/4 treatment-emergent AEs with the combination. Diarrhea, nausea, neutropenia, and pyrexia were also more frequent with the combination than with rituximab-placebo.
More than half of patients in the combination arm (56.3%) developed grade 3/4 hyperglycemia. In the placebo arm, the incidence of grade 3 hyperglycemia was 8.2%, and there was no grade 4 hyperglycemia.
Rates of grade 3 hypertension were 39.7% in the combination arm and 8.9% in the placebo arm. There was no grade 4 hypertension.
In the combination arm, 2.6% of patients stopped treatment because of hyperglycemia and 0.7% stopped because of hypertension.
Any-grade pneumonitis occurred in 6.8% of patients in the combination arm and 1.4% of those in the placebo arm. The rate of grade 3/4 pneumonitis was 2.7% in the copanlisib arm, and the rate of grade 3 pneumonitis was 0.7% in the placebo arm.
The study was funded by Bayer, the company developing copanlisib. Dr. Matasar disclosed relationships with Bayer, its subsidiaries, and Roche/Genentech. Dr. Swanton disclosed relationships with numerous companies, including Pfizer, Novartis, and GlaxoSmithKline.
The trial, dubbed CHRONOS-3, is the first to report “a broad benefit” across histologic subtypes of relapsed, indolent NHL, and the results are “essentially a long-awaited proof of concept” for combining a PI3K inhibitor with rituximab, according to investigator Matthew Matasar, MD, of Memorial Sloan Kettering Cancer Center in New York.
Dr. Matasar presented results from CHRONOS-3 at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT001). The findings were simultaneously published in The Lancet Oncology.
Charles Swanton, MBPhD, of the Francis Crick Institute and UCL Cancer Institute in London, called the results “strongly positive” and said the copanlisib-rituximab combination is “a potential new treatment option” for indolent NHL in patients with a long remission after first-line therapy or those who are unfit for chemotherapy.
Dr. Swanton noted, however, that “one should also bear in mind” the serious adverse events (AEs) seen with copanlisib, particularly hypertension and hyperglycemia. When asked about these AEs, Dr. Matasar said he thinks the combination would be appropriate for patients who meet the study criteria as long as they don’t have severe baseline diabetes or uncontrolled hypertension.
Patient and treatment details
The study included 458 patients with CD20-positive, relapsed, indolent, B-cell NHL. Subtypes included follicular lymphoma (n = 275), marginal zone lymphoma (n = 95), small lymphocytic lymphoma (n = 50), and lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia (n = 38).
All patients were progression free and treatment free before their relapse for at least 12 months after their last rituximab-containing regimen, or at least 6 months before relapse if they were unwilling or unable to undergo chemotherapy.
The patients’ median age was 63 years, and just over half of them were men (52%). About 37% of patients had a history of hypertension at baseline, and about 15% had a history of diabetes.
Patients were randomized to receive copanlisib plus rituximab (n = 307) or rituximab plus placebo (n = 151). Copanlisib was given at 60 mg IV on days 1, 8, and 15 of a 28-day cycle. In both arms, rituximab was given at 375 mg/m2 on days 1, 8, 15, and 22 during cycle 1 and on day 1 of cycles 3, 5, 7, and 9.
Progression-free survival benefit
At a median follow-up of 19.2 months, the median progression-free survival (PFS) was 21.5 months in the copanlisib-rituximab arm and 13.8 months in the placebo-rituximab arm (hazard ratio, 0.52; P < .0001).
The PFS advantage with copanlisib was seen across subtypes:
- Follicular lymphoma – 22.2 months vs. 18.7 months (P = .001)
- Small lymphocytic lymphoma – 14.2 months vs. 5.7 months (P < .0001)
- Marginal zone lymphoma – 22.1 months vs. 11.5 months (P = .012)
- Lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia – 33.4 months vs. 16.6 months (P = .054)
The PFS difference among patients with lymphoplasmacytic lymphoma/Waldenström’s macroglobulinemia was likely not statistically significant because of the small sample size, Dr. Matasar said.
He reported that the overall response rate was 81% with copanlisib-rituximab, including a 34% complete response rate. In the placebo arm, the overall response rate was 48%, and 15% of patients had a complete response.
The median overall survival was not estimable in either treatment arm. At a median follow-up of 30.1 months, 14% of patients in the copanlisib arm and 13.2% of patients in the placebo arm had died.
More than double the rate of serious AEs
The rate of serious treatment-emergent AEs was 47.2% in the combination arm and 18.5% in the placebo arm.
There were six grade 5 treatment-emergent AEs in the combination arm. One of these – pneumonitis – was deemed treatment related. There was one treatment-emergent death in the placebo arm.
Hyperglycemia and hypertension were the most common grade 3/4 treatment-emergent AEs with the combination. Diarrhea, nausea, neutropenia, and pyrexia were also more frequent with the combination than with rituximab-placebo.
More than half of patients in the combination arm (56.3%) developed grade 3/4 hyperglycemia. In the placebo arm, the incidence of grade 3 hyperglycemia was 8.2%, and there was no grade 4 hyperglycemia.
Rates of grade 3 hypertension were 39.7% in the combination arm and 8.9% in the placebo arm. There was no grade 4 hypertension.
In the combination arm, 2.6% of patients stopped treatment because of hyperglycemia and 0.7% stopped because of hypertension.
Any-grade pneumonitis occurred in 6.8% of patients in the combination arm and 1.4% of those in the placebo arm. The rate of grade 3/4 pneumonitis was 2.7% in the copanlisib arm, and the rate of grade 3 pneumonitis was 0.7% in the placebo arm.
The study was funded by Bayer, the company developing copanlisib. Dr. Matasar disclosed relationships with Bayer, its subsidiaries, and Roche/Genentech. Dr. Swanton disclosed relationships with numerous companies, including Pfizer, Novartis, and GlaxoSmithKline.
FROM AACR 2021
Self-directed digital exercise plan improves knee OA
Adults with knee osteoarthritis (OA) who participated in a self-directed, web-based exercise program with automated text-message reminders and encouragement for 6 months showed significant improvement in overall knee pain and physical function, compared with patients who received web-based OA information alone, in a randomized trial of 180 individuals.
The results support a role for web-based exercise intervention to improve knee OA patients’ access to recommended exercises and to assist clinicians in managing patients on a population level, according to the first author of the study, Rachel Kate Nelligan, of the University of Melbourne, and colleagues. Their report is in JAMA Internal Medicine.
“Our free-to-access, unsupervised program could serve as an entry-level intervention, with participants who do not experience clinical benefits progressing to subsequent steps for more intensive, personalized management,” they said. “Such an approach has the potential to better distribute limited health care resources and reduce demand for contact with health professionals, thus improving access for those requiring it.”
Only two other randomized, clinical trials have evaluated web-based interventions for OA without contact from health professionals, according to the authors. While one of those did not find any differences in outcomes at 4 months when comparing a self-directed progressive lower-limb strength, flexibility, and walking program to being on a wait list, a separate trial evaluating a 9-module physical activity program in adults with knee and/or hip OA vs. a wait-list control group found evidence for efficacy for physical function at 3 months, but not quality of life or function in sport and recreation.
For the current study, researchers recruited 206 adults in Australia with clinically diagnosed knee OA via online advertisements and a volunteer database. Participants were aged 45 years or older, and reported activity-related knee pain and morning knee stiffness lasting at least 30 minutes; knee pain on most days for at least 3 months; and average knee pain severity of 4 or higher on an 11-point numeric rating scale in the previous week. In addition, participants were required to own a cell phone with text messaging, have Internet access, and be able to complete assessments.
Patients randomized to the intervention of the My Knee Exercise website received web-based information about OA and the value of exercise, with a 24-week self-directed program of strengthening exercises plus automated text messages to motivate behavior changes and encourage adherence to the exercise program. Controls received access to web-based information about OA and the value of exercise, but without the prescribed exercises or texts. Patients in the intervention group received an average of 60 text messages during the study period, and the average reply rate was 73%.
The primary study outcomes were changes in overall knee pain based on a numeric 0-10 rating scale and changes in physical function based on the Western Ontario and McMaster Universities Osteoarthritis Index 0-68 scale. A total of 180 participants completed both primary outcome measures at 24 weeks. The average age of the participants was 60 years, and 61% were women.
After 24 weeks, the intervention group averaged significantly greater improvement of 1.6 units for overall knee pain (P < .001) and 5.2 units for physical function (P = .002), compared with controls.
In addition, the proportion of patients who exceeded the minimal clinically important difference in pain improvement of at least 1.8 units was significantly higher in the intervention group, compared with controls (72.1% vs. 42.0%; P < .001). Similarly, more intervention-group patients achieved the minimal clinically important difference in WOMAC physical function of improvement of at least 6 units (68.0% vs. 40.8%; P < .001).
Secondary outcomes included additional measures of knee pain, knee function for sport and recreation, quality of life, physical activity, self-efficacy, overall improvement, and treatment satisfaction. Between-group differences favored the intervention on most measures, including Knee Injury and Osteoarthritis Outcome Score subscales for pain, sports/recreation, and quality of life; health-related quality of life; Arthritis Self-Efficacy Scale (ASES) pain subscale, individual change since baseline, and overall patient satisfaction. “Changes in PASE [Physical Activity Scale for the Elderly], ASES function, and SEE [Self Efficacy Exercise] were similar in both groups,” the researchers said.
No serious adverse events were reported by any study participants. Eight patients in the intervention group reported knee pain during the study, compared with one of the controls, and use of pain medications was similar between the groups, except that more control participants used massage, heat or cold, and topical anti-inflammatories.
The results suggest that a majority of participants in the intervention group improved pain and function without the need for in-person contact with a health professional, the researchers noted. However, more intensive management may be needed to support the 30% who did not benefit from the unsupervised approach, they said.
The study findings were limited by several factors, including the potential bias of a volunteer study population, possible lack of generalizability to individuals with lower levels of education or self-efficacy, and lack of direct comparison between web-based intervention and clinician-delivered intervention, the researchers noted.
The study was funded by the National Health and Medical Research Council, whose fellowships supported two of the authors. Lead author Ms. Nelligan disclosed a PhD scholarship from the Australian Government Research Training Program and personal fees from the University of Melbourne unrelated to the current study.
Adults with knee osteoarthritis (OA) who participated in a self-directed, web-based exercise program with automated text-message reminders and encouragement for 6 months showed significant improvement in overall knee pain and physical function, compared with patients who received web-based OA information alone, in a randomized trial of 180 individuals.
The results support a role for web-based exercise intervention to improve knee OA patients’ access to recommended exercises and to assist clinicians in managing patients on a population level, according to the first author of the study, Rachel Kate Nelligan, of the University of Melbourne, and colleagues. Their report is in JAMA Internal Medicine.
“Our free-to-access, unsupervised program could serve as an entry-level intervention, with participants who do not experience clinical benefits progressing to subsequent steps for more intensive, personalized management,” they said. “Such an approach has the potential to better distribute limited health care resources and reduce demand for contact with health professionals, thus improving access for those requiring it.”
Only two other randomized, clinical trials have evaluated web-based interventions for OA without contact from health professionals, according to the authors. While one of those did not find any differences in outcomes at 4 months when comparing a self-directed progressive lower-limb strength, flexibility, and walking program to being on a wait list, a separate trial evaluating a 9-module physical activity program in adults with knee and/or hip OA vs. a wait-list control group found evidence for efficacy for physical function at 3 months, but not quality of life or function in sport and recreation.
For the current study, researchers recruited 206 adults in Australia with clinically diagnosed knee OA via online advertisements and a volunteer database. Participants were aged 45 years or older, and reported activity-related knee pain and morning knee stiffness lasting at least 30 minutes; knee pain on most days for at least 3 months; and average knee pain severity of 4 or higher on an 11-point numeric rating scale in the previous week. In addition, participants were required to own a cell phone with text messaging, have Internet access, and be able to complete assessments.
Patients randomized to the intervention of the My Knee Exercise website received web-based information about OA and the value of exercise, with a 24-week self-directed program of strengthening exercises plus automated text messages to motivate behavior changes and encourage adherence to the exercise program. Controls received access to web-based information about OA and the value of exercise, but without the prescribed exercises or texts. Patients in the intervention group received an average of 60 text messages during the study period, and the average reply rate was 73%.
The primary study outcomes were changes in overall knee pain based on a numeric 0-10 rating scale and changes in physical function based on the Western Ontario and McMaster Universities Osteoarthritis Index 0-68 scale. A total of 180 participants completed both primary outcome measures at 24 weeks. The average age of the participants was 60 years, and 61% were women.
After 24 weeks, the intervention group averaged significantly greater improvement of 1.6 units for overall knee pain (P < .001) and 5.2 units for physical function (P = .002), compared with controls.
In addition, the proportion of patients who exceeded the minimal clinically important difference in pain improvement of at least 1.8 units was significantly higher in the intervention group, compared with controls (72.1% vs. 42.0%; P < .001). Similarly, more intervention-group patients achieved the minimal clinically important difference in WOMAC physical function of improvement of at least 6 units (68.0% vs. 40.8%; P < .001).
Secondary outcomes included additional measures of knee pain, knee function for sport and recreation, quality of life, physical activity, self-efficacy, overall improvement, and treatment satisfaction. Between-group differences favored the intervention on most measures, including Knee Injury and Osteoarthritis Outcome Score subscales for pain, sports/recreation, and quality of life; health-related quality of life; Arthritis Self-Efficacy Scale (ASES) pain subscale, individual change since baseline, and overall patient satisfaction. “Changes in PASE [Physical Activity Scale for the Elderly], ASES function, and SEE [Self Efficacy Exercise] were similar in both groups,” the researchers said.
No serious adverse events were reported by any study participants. Eight patients in the intervention group reported knee pain during the study, compared with one of the controls, and use of pain medications was similar between the groups, except that more control participants used massage, heat or cold, and topical anti-inflammatories.
The results suggest that a majority of participants in the intervention group improved pain and function without the need for in-person contact with a health professional, the researchers noted. However, more intensive management may be needed to support the 30% who did not benefit from the unsupervised approach, they said.
The study findings were limited by several factors, including the potential bias of a volunteer study population, possible lack of generalizability to individuals with lower levels of education or self-efficacy, and lack of direct comparison between web-based intervention and clinician-delivered intervention, the researchers noted.
The study was funded by the National Health and Medical Research Council, whose fellowships supported two of the authors. Lead author Ms. Nelligan disclosed a PhD scholarship from the Australian Government Research Training Program and personal fees from the University of Melbourne unrelated to the current study.
Adults with knee osteoarthritis (OA) who participated in a self-directed, web-based exercise program with automated text-message reminders and encouragement for 6 months showed significant improvement in overall knee pain and physical function, compared with patients who received web-based OA information alone, in a randomized trial of 180 individuals.
The results support a role for web-based exercise intervention to improve knee OA patients’ access to recommended exercises and to assist clinicians in managing patients on a population level, according to the first author of the study, Rachel Kate Nelligan, of the University of Melbourne, and colleagues. Their report is in JAMA Internal Medicine.
“Our free-to-access, unsupervised program could serve as an entry-level intervention, with participants who do not experience clinical benefits progressing to subsequent steps for more intensive, personalized management,” they said. “Such an approach has the potential to better distribute limited health care resources and reduce demand for contact with health professionals, thus improving access for those requiring it.”
Only two other randomized, clinical trials have evaluated web-based interventions for OA without contact from health professionals, according to the authors. While one of those did not find any differences in outcomes at 4 months when comparing a self-directed progressive lower-limb strength, flexibility, and walking program to being on a wait list, a separate trial evaluating a 9-module physical activity program in adults with knee and/or hip OA vs. a wait-list control group found evidence for efficacy for physical function at 3 months, but not quality of life or function in sport and recreation.
For the current study, researchers recruited 206 adults in Australia with clinically diagnosed knee OA via online advertisements and a volunteer database. Participants were aged 45 years or older, and reported activity-related knee pain and morning knee stiffness lasting at least 30 minutes; knee pain on most days for at least 3 months; and average knee pain severity of 4 or higher on an 11-point numeric rating scale in the previous week. In addition, participants were required to own a cell phone with text messaging, have Internet access, and be able to complete assessments.
Patients randomized to the intervention of the My Knee Exercise website received web-based information about OA and the value of exercise, with a 24-week self-directed program of strengthening exercises plus automated text messages to motivate behavior changes and encourage adherence to the exercise program. Controls received access to web-based information about OA and the value of exercise, but without the prescribed exercises or texts. Patients in the intervention group received an average of 60 text messages during the study period, and the average reply rate was 73%.
The primary study outcomes were changes in overall knee pain based on a numeric 0-10 rating scale and changes in physical function based on the Western Ontario and McMaster Universities Osteoarthritis Index 0-68 scale. A total of 180 participants completed both primary outcome measures at 24 weeks. The average age of the participants was 60 years, and 61% were women.
After 24 weeks, the intervention group averaged significantly greater improvement of 1.6 units for overall knee pain (P < .001) and 5.2 units for physical function (P = .002), compared with controls.
In addition, the proportion of patients who exceeded the minimal clinically important difference in pain improvement of at least 1.8 units was significantly higher in the intervention group, compared with controls (72.1% vs. 42.0%; P < .001). Similarly, more intervention-group patients achieved the minimal clinically important difference in WOMAC physical function of improvement of at least 6 units (68.0% vs. 40.8%; P < .001).
Secondary outcomes included additional measures of knee pain, knee function for sport and recreation, quality of life, physical activity, self-efficacy, overall improvement, and treatment satisfaction. Between-group differences favored the intervention on most measures, including Knee Injury and Osteoarthritis Outcome Score subscales for pain, sports/recreation, and quality of life; health-related quality of life; Arthritis Self-Efficacy Scale (ASES) pain subscale, individual change since baseline, and overall patient satisfaction. “Changes in PASE [Physical Activity Scale for the Elderly], ASES function, and SEE [Self Efficacy Exercise] were similar in both groups,” the researchers said.
No serious adverse events were reported by any study participants. Eight patients in the intervention group reported knee pain during the study, compared with one of the controls, and use of pain medications was similar between the groups, except that more control participants used massage, heat or cold, and topical anti-inflammatories.
The results suggest that a majority of participants in the intervention group improved pain and function without the need for in-person contact with a health professional, the researchers noted. However, more intensive management may be needed to support the 30% who did not benefit from the unsupervised approach, they said.
The study findings were limited by several factors, including the potential bias of a volunteer study population, possible lack of generalizability to individuals with lower levels of education or self-efficacy, and lack of direct comparison between web-based intervention and clinician-delivered intervention, the researchers noted.
The study was funded by the National Health and Medical Research Council, whose fellowships supported two of the authors. Lead author Ms. Nelligan disclosed a PhD scholarship from the Australian Government Research Training Program and personal fees from the University of Melbourne unrelated to the current study.
FROM JAMA INTERNAL MEDICINE
Presurgical nivo/chemo boosts pCR rates in NSCLC
Patients with resectable non–small cell lung cancer (NSCLC) often receive treatment before they undergo surgery. For such patients who achieve a pathologic complete response (pCR), the chances of survival are improved.
However, only a small percentage of patients achieve a pCR with neoadjuvant chemotherapy alone.
Adding the immune checkpoint inhibitor nivolumab to platinum-doublet chemotherapy in the neoadjuvant setting boosts the success rate.
Results from the CheckMate 816 trial show that pCR rates improved from 2.2% with chemotherapy alone to 24% when nivolumab was added.
This difference translated into an odds ratio for achieving a pCR with nivolumab plus chemotherapy of 13.94 (P < .0001), reported Patrick M. Forde, MBBCh, from the Johns Hopkins Kimmel Cancer Center, Baltimore.
This primary endpoint, pCR, was defined as complete regression in both the primary tumor and lymph nodes.
“The magnitude of pCR with nivo plus chemo was similar in stage 1B, II, and stage IIIA disease, as well as in both squamous and nonsquamous histologies,” he added.
Dr. Forde presented the new data at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT003).
The higher pCR rates were seen regardless of PD-L1 expression or tumor mutational burden, Dr. Forde said.
The benefit was also seen when the researchers considered only those patients who subsequently underwent resection (pCR rate of 30.5% with the combination versus 3.2% with chemotherapy alone) and when only the primary tumor was considered (pCR rate of 25.7% vs. 2.8%, respectively).
Change in trial design
Invited discussant Jhanelle Gray, MD, from the Moffitt Cancer Center, Tampa, pointed out that the CheckMate 816 trial originally included an experimental arm with double immunotherapy – ipilimumab plus nivolumab – added onto chemotherapy.
However, this third arm was closed after other trials reported promising results from adding a single immunotherapy onto chemotherapy. For example, results of the single-arm NADIM phase 2 study showed a 77.1% progression-free survival rate at 24 months with the combination of nivolumab, paclitaxel, and carboplatin, and the phase 2 KEYNOTE-021 trial showed that adding pembrolizumab to a standard platinum-doublet chemotherapy regimen nearly doubled response rates among patients with previously untreated advanced nonsquamous NSCLC (although there was no advantage in overall survival).
“Even with the change in trial design, patient characteristics were well balanced between the two arms, and the study met its primary endpoint in the intent-to-treat population,” she said.
Dr. Gray also commented that the choice of pCR as a primary endpoint is “intriguing, and the question remains if it represents a valid surrogate endpoint for survival.”
She noted that a meta-analysis of 32 neoadjuvant chemotherapy-based studies in NSCLC, presented at the 2020 European Society of Medical Oncology annual meeting, showed clear associations of pCR and major pathologic response to both overall survival and event-free survival.
“As these findings were established in a backdrop of chemotherapy, work is needed to confirm these findings in the setting of immunotherapy in particular, as at times, radiographic findings do not correlate with histological findings,” Dr. Gray said.
CheckMate 816 particulars
CheckMate 816 was conducted in 358 patients with newly diagnosed NSCLC with resectable stage IB tumors of at least 4 cm up to stage IIIA tumors, good performance status, and no known EGFR mutations or ALK alterations.
Patients were randomly assigned on an equal basis to receive either nivolumab at 360 mg plus chemotherapy every 3 weeks for three cycles or chemotherapy alone.
Surgery was planned within 6 week after neoadjuvant therapy. Patients could receive (at the investigator’s discretion) adjuvant chemotherapy with or without radiotherapy but no further immunotherapy during follow-up.
In this analysis, patients who did not undergo surgery or for whom evaluable tissue samples were not available were counted among those whose conditions did not respond to therapy.
Major pathologic response rate (≤10% residual viable tumor cells in the primary lung tumor and sampled lymph nodes), which was a secondary endpoint, was also significantly better, at 36.9% versus 8.9%, translating into an OR of 5.70 (95% confidence interval, 3.16-10.26).
In a subset of patients, the investigators assessed clearance of circulating tumor DNA (ctDNA) from day 1 of the first cycle to day 1 of the third cycle using a highly sensitive tumor-informed approach. They found that ctDNA was notably higher with the combination than with chemotherapy alone and that ctDNA clearance correlated with pCR.
Safety similar
“Remarkably, safety was quite similar across the two treatment arms,” Dr. Forde said.
The addition of nivolumab to chemotherapy did not appear to increase either treatment-related adverse events or adverse events of any cause. Grade 3-4 adverse events occurred in 41% of patients in the combination arm versus 44% in the chemotherapy-alone arm.
Treatment-related adverse events leading to discontinuation occurred in 10% of patients in each arm.
Two patients in the nivolumab-chemotherapy arm died from surgically related adverse events (one pulmonary embolism and one aortic rupture). These events were deemed to be unrelated to the study drug.
The investigators are continuing to assess event-free survival and overall survival.
CheckMate 816 is funded by Bristol-Myers Squibb and Ono Pharmaceutical. Dr. Forde has received grants/research support and advisory fees from Bristol-Myers Squibb and others. Dr. Gray has consulted for and has received grant/research support from Bristol-Myers Squibb and others.
A version of this article first appeared on Medscape.com.
Patients with resectable non–small cell lung cancer (NSCLC) often receive treatment before they undergo surgery. For such patients who achieve a pathologic complete response (pCR), the chances of survival are improved.
However, only a small percentage of patients achieve a pCR with neoadjuvant chemotherapy alone.
Adding the immune checkpoint inhibitor nivolumab to platinum-doublet chemotherapy in the neoadjuvant setting boosts the success rate.
Results from the CheckMate 816 trial show that pCR rates improved from 2.2% with chemotherapy alone to 24% when nivolumab was added.
This difference translated into an odds ratio for achieving a pCR with nivolumab plus chemotherapy of 13.94 (P < .0001), reported Patrick M. Forde, MBBCh, from the Johns Hopkins Kimmel Cancer Center, Baltimore.
This primary endpoint, pCR, was defined as complete regression in both the primary tumor and lymph nodes.
“The magnitude of pCR with nivo plus chemo was similar in stage 1B, II, and stage IIIA disease, as well as in both squamous and nonsquamous histologies,” he added.
Dr. Forde presented the new data at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT003).
The higher pCR rates were seen regardless of PD-L1 expression or tumor mutational burden, Dr. Forde said.
The benefit was also seen when the researchers considered only those patients who subsequently underwent resection (pCR rate of 30.5% with the combination versus 3.2% with chemotherapy alone) and when only the primary tumor was considered (pCR rate of 25.7% vs. 2.8%, respectively).
Change in trial design
Invited discussant Jhanelle Gray, MD, from the Moffitt Cancer Center, Tampa, pointed out that the CheckMate 816 trial originally included an experimental arm with double immunotherapy – ipilimumab plus nivolumab – added onto chemotherapy.
However, this third arm was closed after other trials reported promising results from adding a single immunotherapy onto chemotherapy. For example, results of the single-arm NADIM phase 2 study showed a 77.1% progression-free survival rate at 24 months with the combination of nivolumab, paclitaxel, and carboplatin, and the phase 2 KEYNOTE-021 trial showed that adding pembrolizumab to a standard platinum-doublet chemotherapy regimen nearly doubled response rates among patients with previously untreated advanced nonsquamous NSCLC (although there was no advantage in overall survival).
“Even with the change in trial design, patient characteristics were well balanced between the two arms, and the study met its primary endpoint in the intent-to-treat population,” she said.
Dr. Gray also commented that the choice of pCR as a primary endpoint is “intriguing, and the question remains if it represents a valid surrogate endpoint for survival.”
She noted that a meta-analysis of 32 neoadjuvant chemotherapy-based studies in NSCLC, presented at the 2020 European Society of Medical Oncology annual meeting, showed clear associations of pCR and major pathologic response to both overall survival and event-free survival.
“As these findings were established in a backdrop of chemotherapy, work is needed to confirm these findings in the setting of immunotherapy in particular, as at times, radiographic findings do not correlate with histological findings,” Dr. Gray said.
CheckMate 816 particulars
CheckMate 816 was conducted in 358 patients with newly diagnosed NSCLC with resectable stage IB tumors of at least 4 cm up to stage IIIA tumors, good performance status, and no known EGFR mutations or ALK alterations.
Patients were randomly assigned on an equal basis to receive either nivolumab at 360 mg plus chemotherapy every 3 weeks for three cycles or chemotherapy alone.
Surgery was planned within 6 week after neoadjuvant therapy. Patients could receive (at the investigator’s discretion) adjuvant chemotherapy with or without radiotherapy but no further immunotherapy during follow-up.
In this analysis, patients who did not undergo surgery or for whom evaluable tissue samples were not available were counted among those whose conditions did not respond to therapy.
Major pathologic response rate (≤10% residual viable tumor cells in the primary lung tumor and sampled lymph nodes), which was a secondary endpoint, was also significantly better, at 36.9% versus 8.9%, translating into an OR of 5.70 (95% confidence interval, 3.16-10.26).
In a subset of patients, the investigators assessed clearance of circulating tumor DNA (ctDNA) from day 1 of the first cycle to day 1 of the third cycle using a highly sensitive tumor-informed approach. They found that ctDNA was notably higher with the combination than with chemotherapy alone and that ctDNA clearance correlated with pCR.
Safety similar
“Remarkably, safety was quite similar across the two treatment arms,” Dr. Forde said.
The addition of nivolumab to chemotherapy did not appear to increase either treatment-related adverse events or adverse events of any cause. Grade 3-4 adverse events occurred in 41% of patients in the combination arm versus 44% in the chemotherapy-alone arm.
Treatment-related adverse events leading to discontinuation occurred in 10% of patients in each arm.
Two patients in the nivolumab-chemotherapy arm died from surgically related adverse events (one pulmonary embolism and one aortic rupture). These events were deemed to be unrelated to the study drug.
The investigators are continuing to assess event-free survival and overall survival.
CheckMate 816 is funded by Bristol-Myers Squibb and Ono Pharmaceutical. Dr. Forde has received grants/research support and advisory fees from Bristol-Myers Squibb and others. Dr. Gray has consulted for and has received grant/research support from Bristol-Myers Squibb and others.
A version of this article first appeared on Medscape.com.
Patients with resectable non–small cell lung cancer (NSCLC) often receive treatment before they undergo surgery. For such patients who achieve a pathologic complete response (pCR), the chances of survival are improved.
However, only a small percentage of patients achieve a pCR with neoadjuvant chemotherapy alone.
Adding the immune checkpoint inhibitor nivolumab to platinum-doublet chemotherapy in the neoadjuvant setting boosts the success rate.
Results from the CheckMate 816 trial show that pCR rates improved from 2.2% with chemotherapy alone to 24% when nivolumab was added.
This difference translated into an odds ratio for achieving a pCR with nivolumab plus chemotherapy of 13.94 (P < .0001), reported Patrick M. Forde, MBBCh, from the Johns Hopkins Kimmel Cancer Center, Baltimore.
This primary endpoint, pCR, was defined as complete regression in both the primary tumor and lymph nodes.
“The magnitude of pCR with nivo plus chemo was similar in stage 1B, II, and stage IIIA disease, as well as in both squamous and nonsquamous histologies,” he added.
Dr. Forde presented the new data at the American Association for Cancer Research Annual Meeting 2021: Week 1 (Abstract CT003).
The higher pCR rates were seen regardless of PD-L1 expression or tumor mutational burden, Dr. Forde said.
The benefit was also seen when the researchers considered only those patients who subsequently underwent resection (pCR rate of 30.5% with the combination versus 3.2% with chemotherapy alone) and when only the primary tumor was considered (pCR rate of 25.7% vs. 2.8%, respectively).
Change in trial design
Invited discussant Jhanelle Gray, MD, from the Moffitt Cancer Center, Tampa, pointed out that the CheckMate 816 trial originally included an experimental arm with double immunotherapy – ipilimumab plus nivolumab – added onto chemotherapy.
However, this third arm was closed after other trials reported promising results from adding a single immunotherapy onto chemotherapy. For example, results of the single-arm NADIM phase 2 study showed a 77.1% progression-free survival rate at 24 months with the combination of nivolumab, paclitaxel, and carboplatin, and the phase 2 KEYNOTE-021 trial showed that adding pembrolizumab to a standard platinum-doublet chemotherapy regimen nearly doubled response rates among patients with previously untreated advanced nonsquamous NSCLC (although there was no advantage in overall survival).
“Even with the change in trial design, patient characteristics were well balanced between the two arms, and the study met its primary endpoint in the intent-to-treat population,” she said.
Dr. Gray also commented that the choice of pCR as a primary endpoint is “intriguing, and the question remains if it represents a valid surrogate endpoint for survival.”
She noted that a meta-analysis of 32 neoadjuvant chemotherapy-based studies in NSCLC, presented at the 2020 European Society of Medical Oncology annual meeting, showed clear associations of pCR and major pathologic response to both overall survival and event-free survival.
“As these findings were established in a backdrop of chemotherapy, work is needed to confirm these findings in the setting of immunotherapy in particular, as at times, radiographic findings do not correlate with histological findings,” Dr. Gray said.
CheckMate 816 particulars
CheckMate 816 was conducted in 358 patients with newly diagnosed NSCLC with resectable stage IB tumors of at least 4 cm up to stage IIIA tumors, good performance status, and no known EGFR mutations or ALK alterations.
Patients were randomly assigned on an equal basis to receive either nivolumab at 360 mg plus chemotherapy every 3 weeks for three cycles or chemotherapy alone.
Surgery was planned within 6 week after neoadjuvant therapy. Patients could receive (at the investigator’s discretion) adjuvant chemotherapy with or without radiotherapy but no further immunotherapy during follow-up.
In this analysis, patients who did not undergo surgery or for whom evaluable tissue samples were not available were counted among those whose conditions did not respond to therapy.
Major pathologic response rate (≤10% residual viable tumor cells in the primary lung tumor and sampled lymph nodes), which was a secondary endpoint, was also significantly better, at 36.9% versus 8.9%, translating into an OR of 5.70 (95% confidence interval, 3.16-10.26).
In a subset of patients, the investigators assessed clearance of circulating tumor DNA (ctDNA) from day 1 of the first cycle to day 1 of the third cycle using a highly sensitive tumor-informed approach. They found that ctDNA was notably higher with the combination than with chemotherapy alone and that ctDNA clearance correlated with pCR.
Safety similar
“Remarkably, safety was quite similar across the two treatment arms,” Dr. Forde said.
The addition of nivolumab to chemotherapy did not appear to increase either treatment-related adverse events or adverse events of any cause. Grade 3-4 adverse events occurred in 41% of patients in the combination arm versus 44% in the chemotherapy-alone arm.
Treatment-related adverse events leading to discontinuation occurred in 10% of patients in each arm.
Two patients in the nivolumab-chemotherapy arm died from surgically related adverse events (one pulmonary embolism and one aortic rupture). These events were deemed to be unrelated to the study drug.
The investigators are continuing to assess event-free survival and overall survival.
CheckMate 816 is funded by Bristol-Myers Squibb and Ono Pharmaceutical. Dr. Forde has received grants/research support and advisory fees from Bristol-Myers Squibb and others. Dr. Gray has consulted for and has received grant/research support from Bristol-Myers Squibb and others.
A version of this article first appeared on Medscape.com.
FROM AACR 2021
2021 Update on sequencing in prenatal genetics
Prenatal diagnosis has expanded from identification of aneuploidy to include copy number variants detected on microarray (such as 22q11 deletion syndrome) and now single-gene disorders identified by targeted or exome and genome sequencing. How and when different sequencing tests should be used clinically are questions faced by every provider engaged in modern prenatal diagnosis.
In this Update, we highlight new clinical insights into prenatal sequencing and explore how information gained from sequencing may help us understand some of the unanswered questions in obstetrics.
What is the yield of a RASopathy gene panel with specific prenatal findings?
Scott A, Di Giosaffatte N, Pinna V, et al. When to test fetuses for RASopathies? Proposition from a systematic analysis of 352 multicenter cases and a postnatal cohort. Genet Med. Published online February 10, 2021. doi:10.1038/s41436-020-01093-7.
RASopathies, a group of genetic conditions caused by mutations in the RAS/mitogen-activated protein kinase (RAS-MAPK) pathway, are common, occurring in 1:1,000 to 1:2,500 live births. RASopathies are much more common than 22q11 deletion syndrome, or DiGeorge syndrome, which occurs in 1.4:10,000 live births.1
RASopathy disorders include Noonan syndrome, Noonan syndrome with multiple lentigines, Costello syndrome, cardiofaciocutaneous syndrome, and Noonan-like syndrome with loose anagen hair. These are autosomal dominant disorders caused by a pathogenic variant (or mutation) in 1 of more than 20 genes in the RAS-MAPK signaling pathway in the body. Clinical features include congenital anomalies of the kidney and urinary tract, lymphatic anomalies, congenital heart disease (CHD), hypertrophic cardiomyopathy (HCM), postnatal growth disorders, neurodevelopmental disorders, and more rarely hematologic malignancies. Prenatal clues include an increased nuchal translucency (NT), CHD, cystic hygroma, lymphatic anomalies, anomalies of the kidney and urinary tract, hydrops, and HCM.
Cohort of pregnancies that received a RASopathy panel
Scott and colleagues sought to clarify the utility of testing for RASopathies with a prenatal gene panel. They conducted a multicenter retrospective cohort study with cases from 2 hospitals in Italy and Canada; data were collected between 2012 and 2019.
Eligible fetuses were those referred to the prenatal genetics clinic because of an increased NT, increased nuchal fold (NF), hydrops, ascites, thoracic effusions, chylothorax, other lymphatic anomalies, CHD, or HCM with a nondiagnostic (negative) microarray or karyotype. All eligible cases had RASopathy molecular testing in the prenatal or neonatal period.
Among the 352 referrals to clinic, 50 cases of a RASopathy disorder were diagnosed. Additionally, to complement this cohort over the same time period, 25 postnatal diagnoses were made after retrospective review performed to ascertain additional prenatal findings. The size of the testing panel ranged from 9 to 20 genes, which were sent to clinical laboratories that performed sequencing based on standard protocols.
Study outcomes
Overall, 14% of fetuses with an indication for testing had a pathogenic or likely pathogenic variant (diagnostic) on panel testing among 11 genes (notably, all presented results are after excluding copy number variants and aneuploidy). Fetuses with only 1 ultrasonography finding were much less likely to have a positive result than those with more than 1 ultrasonography finding, 3% versus 18%. The highest diagnostic yields were for HCM at 69%; thoracic effusions and ascites, 41%; persistent hydrops, 39%; cystic hygroma combined with another suggestive ultrasonography finding, 28%; CHD, 23%; and persistent cystic hygroma, 21%. Five fetuses were affected with CHD and HCM, and 44% had an intrauterine fetal demise.
Importance of NT size. An isolated increased NT had a diagnostic yield of 1% overall (1/90); however, the size of the NT mattered. Seventeen fetuses had an NT between 3 and 3.5 mm and none of these had diagnostic sequencing, whereas 26% with an NT greater than 6 mm had a diagnostic result (11/43). An increased NF had a diagnostic yield of 25%.
Other findings. Of fetuses with a cystic hygroma, 16% had a pathogenic or likely pathogenic variant, and when these persisted into the second trimester or were associated with other anomalies, the percentages increased to 21% and 28%, respectively. Of prenatal patients, 20.6% had variants of uncertain significance, and 12% of the pathogenic and likely pathogenic variants were inherited, which is less than previously reported series. Additionally, 48% of the postnatal RASopathy diagnosis group did not have an ultrasonography finding on record review.
Continue to: Study strengths and limitations...
Study strengths and limitations
This study presents a large cohort of prenatal and neonatal patients tested for RASopathies at 2 international centers with very granular and clinically useful data about ultrasonography findings and yield of panel testing. Prenatal care providers, geneticists, and computational biologists may find this study of great interest and take away useful information and ideas due to the authors’ presentation and details.
The number of genes tested changed over the inclusion time period, but this is an inescapable reality of retrospective clinical research in an advancing field. The authors presented the prenatal and postnatal diagnoses ultrasonography findings separately and together. Given the different nature of cohort ascertainment, we prefer to consider these groups separately and have presented the data for the prenatal group.
Prenatal sequencing panels and exome sequencing are detecting disorders with important implications for prenatal care. If your practice is not testing for RASopathies in prenatal patients with concerning ultrasonography features, you are missing cases. In this study, the most concerning ultrasonography features (more than 20% diagnosis) were HCM, thoracic effusions and ascites, persistent hydrops, cystic hygroma combined with another suggestive ultrasonography finding, CHD, and persistent cystic hygroma. Isolated ultrasonography findings or findings that resolved had a lower diagnostic yield, and an isolated enlarged NT had a 1% diagnostic yield, with most cases having an NT larger than 6 mm.
For pretest counseling, in this study 20% of patients had a variant of uncertain significance, and preparing patients for this possibility is crucial. Most variants of uncertain significance are reclassified to benign when more information is available. Providers can consider sending parental samples concurrently with the fetal sample to help obtain useful information quickly, although the possibility of an inherited pathogenic variant still exists (12% in this study).
Prenatal diagnosis gives your patients the opportunity to learn about the disorder, plan for treatment and delivery location, and establish their care team before birth or consider pregnancy termination.
Sequencing provides insights into twin pregnancies
Jonsson H, Magnusdottir E, Eggertsson HP, et al. Differences between germline genomes of monozygotic twins. Nat Genet. 2021;53:27-34. doi:10.1038/s41588 -020-00755-1.
You have a monozygotic twin pair with an anomaly and intend to do diagnostic testing for prenatal diagnosis. The question always arises: Do you sample both twins or just one? Surely, they are genetically identical? A wise mentor once instilled a valuable lesson: Monozygotic twins are more likely to have an anomaly. Their existence is already out of the realm of normal. Finally, we now have an engaging and interesting answer to this and other fascinating embryology questions through the work of Jonsson and colleagues.
Study eligibility criteria and treatment protocol
The authors enrolled 381 twin pairs and 2 monozygotic triplets and compared genome sequencing of different tissues (cheek cells and blood). They went further to assess what other tissues might share the genetic change. To do this, they sequenced the children and the partners of 181 of the pairs. Presumably, if a twin and their offspring shared a genetic change that was not present in the spouse or twin, this genetic change must be present in the oocytes or sperm of the parent twin. The goal of sequencing multiple tissue sources in each twin was to help determine when the genetic change occurred in embryonic development.
Study outcomes
The authors found that 15% of twins had mutations that were absent in the other twin. Because of the extent of tissues that had the genetic change, the authors asserted that these changes must have occurred very early in embryonic development (even from one cell after twinning) for the changes to be near-constitutional (among sampled tissues).
An average of 14 genetic differences were found between twin pairs that developed after twinning. However, the number of differences varied. For example, 39 pairs of twins differed by more than 100 changes, and 38 did not differ at all. Differences between twins were more likely in blood samples than in cheek swabs, suggesting that some differences were due to acquired genetic changes in hematologic cell lines, or clonal hematopoiesis.
The authors also looked at what percentage of sequenced DNA contained the variants (or mutations) and found that many of these DNA differences were present at high amounts in sequencing reads. This suggests that the DNA changes happened very early after twinning in about one-third of pairs. Additionally, if one twin had a near-constitutional change, in 42% of pairs the other twin had a different near-constitutional change. Among the triplets, 2 of a triplet pair shared more genetic similarity and were likely descendent from a single split cell and the third likely was formed from a different set of cells.
By examining the offspring of twins, Jonsson and colleagues found that there were 2.6 early embryonic mutations, and this did not differ when blood or buccal DNA was compared. The rate of transmission of a variant to offspring was proportional to the variant allele frequency (proportion of alternate alleles) in the blood or buccal cells. This is an important counseling point when considering patients with mosaic genetic disorders and counseling about the likelihood of inheritance or transmission to future offspring. If the rate of mosaicism was higher in blood or buccal cells, the likelihood of transmission was higher. Additionally, the mutations did not differ by sex, and there was no relationship to whether the chromosome was maternally or paternally inherited.
Continue to: Study strengths and limitations...
Study strengths and limitations
The authors did not have access to information about chorionicity of the monozygotic twin pairs. Consequently, they were unable to correlate chorionicity with the degree of noted genetic difference between the monozygotic twin pairs. Additionally, although the authors were thoughtful in their utilization of offspring and spouses to infer germline genomic content, the study had a limited number of tissues sampled, which could reduce the applicability. However, the sample size, clinically accessible tissue sampling, and thoughtful analysis used in this study make it an interesting and relevant contribution to reproductive medicine and evolutionary biology.
We all accumulate changes to our DNA throughout life. The study by Jonsson and colleagues illustrates that for many, this accumulation of genetic changes starts very early in gestation. In the early zygote, the authors observed roughly 1 mutation per cell division prior to the point of twinning. In the realm of prenatal diagnosis, one should consider that monochorionic twins with different phenotypes (that is, an ultrasonography anomaly in 1 of the twin pair) could represent a genetic change rather than an environmental difference. This genetic change may not be shared by the other twin despite originating from the same primordial cell line. The genetic changes that the authors investigated were detected on genome sequencing, which is much more comprehensive than the exome sequencing that is increasingly utilized in rare disease diagnosis. The clinical utility of this observation in prenatal diagnosis has yet to be proven, but this study provides preliminary data that 15% of monozygotic twins have genetic differences and may warrant individualized testing.
The genetic landscape of the placenta
Coorens TH, Oliver TR, Sanghvi R, et al. Inherent mosaicism and extensive mutation of human placentas. Nature. Published online March 10, 2021. doi:10.1038/ s41586-021-03345-1.
Confined placental mosaicism (CPM) is a phenomenon in which the genetics of the placenta are different from those of the fetus. Historically, this phenomenon has been described in 1% to 2% of pregnancies based on karyotype data obtained from chorionic villus sampling. Some studies have demonstrated adverse pregnancy outcomes in the setting of CPM, thought to be secondary to aneuploid cells in the placenta leading to insufficiency or dysfunction.
Although our sophistication and level of detail in prenatal genetic testing has rapidly expanded to include information about copy number variants and singlenucleotide changes, their contribution to CPM has been understudied. Coorens and colleagues recently published a landmark study that describes a surprisingly high rate of mosaicism for these smaller genetic changes.
A cohort study of placentas
The authors performed whole genome sequencing on placental samples obtained from 37 term pregnancies. Umbilical cord tissue and maternal blood also were collected and served as controls for fetal and maternal genetic profiles, respectively.
In a subgroup of 5 placentas, lasercapture microscopy was used to separate placental cells of different origins, including trophoblastic cells, mesenchymal core cells, and cells originating from the inner cell mass. To investigate variation within different geographic regions of a single placenta, these cell lines were derived multiple times from each quadrant of the 5 placentas.
Placental biopsies revealed “bottlenecks” of genetic differentiation
Genome sequencing was used uniquely in this study to help delineate the phylogeny of placental cells by tracking somatic mutations both in different geographic locations of each placenta and between different cells of origin within 1 placenta.
The authors concluded that bottlenecks of differentiation in placental development led to unique genetic signatures in every bulk placental sample studied. Their findings led them to describe the placenta as a “patchwork” of independent genetic units resulting from clonal expansion at different stages of embryonic development.
Early insights into human placental cells
This study provides fascinating insight into the surprisingly high rates of copy number variants and single-gene changes that exist, in mosaic form, within human placentas. The authors distinguish the placenta from other human organs (such as the colon, endometrium, liver, and skin) in which many fewer genetic changes exist. In fact, they suggest parallels between the “mutational signature” of the placenta with rapidly dividing neoplastic cells.
As one of the first investigations into the variation and complexity of genetic changes within the placenta, this study was not designed to draw conclusions regarding the clinical impact of the numerous genetic changes described. Further studies will elucidate the potential contribution of genetically mosaic placentas to common adverse obstetric outcomes. ●
With a new appreciation for the smaller genetic alterations that exist within placental tissue, it appears that the rate of CPM has been vastly underestimated. We know that aneuploid placental cells increase the risk of adverse pregnancy outcomes and we may learn more about the contribution of copy number variants and single-nucleotide changes to preeclampsia, growth restriction, and pregnancy loss. Furthermore, as the applications of cell-free fetal DNA (cffDNA) in genetic screening continue to expand, we must exercise caution in assuming that copy number variants or single-nucleotide changes detected by cffDNA reflect those of the developing fetus.
- Roberts AE, Allanson JE, Tartaglia M, et al. Noonan syndrome. Lancet. 2013;381:333-342. doi:10.1016/S0140-6736(12)61023-X.
Prenatal diagnosis has expanded from identification of aneuploidy to include copy number variants detected on microarray (such as 22q11 deletion syndrome) and now single-gene disorders identified by targeted or exome and genome sequencing. How and when different sequencing tests should be used clinically are questions faced by every provider engaged in modern prenatal diagnosis.
In this Update, we highlight new clinical insights into prenatal sequencing and explore how information gained from sequencing may help us understand some of the unanswered questions in obstetrics.
What is the yield of a RASopathy gene panel with specific prenatal findings?
Scott A, Di Giosaffatte N, Pinna V, et al. When to test fetuses for RASopathies? Proposition from a systematic analysis of 352 multicenter cases and a postnatal cohort. Genet Med. Published online February 10, 2021. doi:10.1038/s41436-020-01093-7.
RASopathies, a group of genetic conditions caused by mutations in the RAS/mitogen-activated protein kinase (RAS-MAPK) pathway, are common, occurring in 1:1,000 to 1:2,500 live births. RASopathies are much more common than 22q11 deletion syndrome, or DiGeorge syndrome, which occurs in 1.4:10,000 live births.1
RASopathy disorders include Noonan syndrome, Noonan syndrome with multiple lentigines, Costello syndrome, cardiofaciocutaneous syndrome, and Noonan-like syndrome with loose anagen hair. These are autosomal dominant disorders caused by a pathogenic variant (or mutation) in 1 of more than 20 genes in the RAS-MAPK signaling pathway in the body. Clinical features include congenital anomalies of the kidney and urinary tract, lymphatic anomalies, congenital heart disease (CHD), hypertrophic cardiomyopathy (HCM), postnatal growth disorders, neurodevelopmental disorders, and more rarely hematologic malignancies. Prenatal clues include an increased nuchal translucency (NT), CHD, cystic hygroma, lymphatic anomalies, anomalies of the kidney and urinary tract, hydrops, and HCM.
Cohort of pregnancies that received a RASopathy panel
Scott and colleagues sought to clarify the utility of testing for RASopathies with a prenatal gene panel. They conducted a multicenter retrospective cohort study with cases from 2 hospitals in Italy and Canada; data were collected between 2012 and 2019.
Eligible fetuses were those referred to the prenatal genetics clinic because of an increased NT, increased nuchal fold (NF), hydrops, ascites, thoracic effusions, chylothorax, other lymphatic anomalies, CHD, or HCM with a nondiagnostic (negative) microarray or karyotype. All eligible cases had RASopathy molecular testing in the prenatal or neonatal period.
Among the 352 referrals to clinic, 50 cases of a RASopathy disorder were diagnosed. Additionally, to complement this cohort over the same time period, 25 postnatal diagnoses were made after retrospective review performed to ascertain additional prenatal findings. The size of the testing panel ranged from 9 to 20 genes, which were sent to clinical laboratories that performed sequencing based on standard protocols.
Study outcomes
Overall, 14% of fetuses with an indication for testing had a pathogenic or likely pathogenic variant (diagnostic) on panel testing among 11 genes (notably, all presented results are after excluding copy number variants and aneuploidy). Fetuses with only 1 ultrasonography finding were much less likely to have a positive result than those with more than 1 ultrasonography finding, 3% versus 18%. The highest diagnostic yields were for HCM at 69%; thoracic effusions and ascites, 41%; persistent hydrops, 39%; cystic hygroma combined with another suggestive ultrasonography finding, 28%; CHD, 23%; and persistent cystic hygroma, 21%. Five fetuses were affected with CHD and HCM, and 44% had an intrauterine fetal demise.
Importance of NT size. An isolated increased NT had a diagnostic yield of 1% overall (1/90); however, the size of the NT mattered. Seventeen fetuses had an NT between 3 and 3.5 mm and none of these had diagnostic sequencing, whereas 26% with an NT greater than 6 mm had a diagnostic result (11/43). An increased NF had a diagnostic yield of 25%.
Other findings. Of fetuses with a cystic hygroma, 16% had a pathogenic or likely pathogenic variant, and when these persisted into the second trimester or were associated with other anomalies, the percentages increased to 21% and 28%, respectively. Of prenatal patients, 20.6% had variants of uncertain significance, and 12% of the pathogenic and likely pathogenic variants were inherited, which is less than previously reported series. Additionally, 48% of the postnatal RASopathy diagnosis group did not have an ultrasonography finding on record review.
Continue to: Study strengths and limitations...
Study strengths and limitations
This study presents a large cohort of prenatal and neonatal patients tested for RASopathies at 2 international centers with very granular and clinically useful data about ultrasonography findings and yield of panel testing. Prenatal care providers, geneticists, and computational biologists may find this study of great interest and take away useful information and ideas due to the authors’ presentation and details.
The number of genes tested changed over the inclusion time period, but this is an inescapable reality of retrospective clinical research in an advancing field. The authors presented the prenatal and postnatal diagnoses ultrasonography findings separately and together. Given the different nature of cohort ascertainment, we prefer to consider these groups separately and have presented the data for the prenatal group.
Prenatal sequencing panels and exome sequencing are detecting disorders with important implications for prenatal care. If your practice is not testing for RASopathies in prenatal patients with concerning ultrasonography features, you are missing cases. In this study, the most concerning ultrasonography features (more than 20% diagnosis) were HCM, thoracic effusions and ascites, persistent hydrops, cystic hygroma combined with another suggestive ultrasonography finding, CHD, and persistent cystic hygroma. Isolated ultrasonography findings or findings that resolved had a lower diagnostic yield, and an isolated enlarged NT had a 1% diagnostic yield, with most cases having an NT larger than 6 mm.
For pretest counseling, in this study 20% of patients had a variant of uncertain significance, and preparing patients for this possibility is crucial. Most variants of uncertain significance are reclassified to benign when more information is available. Providers can consider sending parental samples concurrently with the fetal sample to help obtain useful information quickly, although the possibility of an inherited pathogenic variant still exists (12% in this study).
Prenatal diagnosis gives your patients the opportunity to learn about the disorder, plan for treatment and delivery location, and establish their care team before birth or consider pregnancy termination.
Sequencing provides insights into twin pregnancies
Jonsson H, Magnusdottir E, Eggertsson HP, et al. Differences between germline genomes of monozygotic twins. Nat Genet. 2021;53:27-34. doi:10.1038/s41588 -020-00755-1.
You have a monozygotic twin pair with an anomaly and intend to do diagnostic testing for prenatal diagnosis. The question always arises: Do you sample both twins or just one? Surely, they are genetically identical? A wise mentor once instilled a valuable lesson: Monozygotic twins are more likely to have an anomaly. Their existence is already out of the realm of normal. Finally, we now have an engaging and interesting answer to this and other fascinating embryology questions through the work of Jonsson and colleagues.
Study eligibility criteria and treatment protocol
The authors enrolled 381 twin pairs and 2 monozygotic triplets and compared genome sequencing of different tissues (cheek cells and blood). They went further to assess what other tissues might share the genetic change. To do this, they sequenced the children and the partners of 181 of the pairs. Presumably, if a twin and their offspring shared a genetic change that was not present in the spouse or twin, this genetic change must be present in the oocytes or sperm of the parent twin. The goal of sequencing multiple tissue sources in each twin was to help determine when the genetic change occurred in embryonic development.
Study outcomes
The authors found that 15% of twins had mutations that were absent in the other twin. Because of the extent of tissues that had the genetic change, the authors asserted that these changes must have occurred very early in embryonic development (even from one cell after twinning) for the changes to be near-constitutional (among sampled tissues).
An average of 14 genetic differences were found between twin pairs that developed after twinning. However, the number of differences varied. For example, 39 pairs of twins differed by more than 100 changes, and 38 did not differ at all. Differences between twins were more likely in blood samples than in cheek swabs, suggesting that some differences were due to acquired genetic changes in hematologic cell lines, or clonal hematopoiesis.
The authors also looked at what percentage of sequenced DNA contained the variants (or mutations) and found that many of these DNA differences were present at high amounts in sequencing reads. This suggests that the DNA changes happened very early after twinning in about one-third of pairs. Additionally, if one twin had a near-constitutional change, in 42% of pairs the other twin had a different near-constitutional change. Among the triplets, 2 of a triplet pair shared more genetic similarity and were likely descendent from a single split cell and the third likely was formed from a different set of cells.
By examining the offspring of twins, Jonsson and colleagues found that there were 2.6 early embryonic mutations, and this did not differ when blood or buccal DNA was compared. The rate of transmission of a variant to offspring was proportional to the variant allele frequency (proportion of alternate alleles) in the blood or buccal cells. This is an important counseling point when considering patients with mosaic genetic disorders and counseling about the likelihood of inheritance or transmission to future offspring. If the rate of mosaicism was higher in blood or buccal cells, the likelihood of transmission was higher. Additionally, the mutations did not differ by sex, and there was no relationship to whether the chromosome was maternally or paternally inherited.
Continue to: Study strengths and limitations...
Study strengths and limitations
The authors did not have access to information about chorionicity of the monozygotic twin pairs. Consequently, they were unable to correlate chorionicity with the degree of noted genetic difference between the monozygotic twin pairs. Additionally, although the authors were thoughtful in their utilization of offspring and spouses to infer germline genomic content, the study had a limited number of tissues sampled, which could reduce the applicability. However, the sample size, clinically accessible tissue sampling, and thoughtful analysis used in this study make it an interesting and relevant contribution to reproductive medicine and evolutionary biology.
We all accumulate changes to our DNA throughout life. The study by Jonsson and colleagues illustrates that for many, this accumulation of genetic changes starts very early in gestation. In the early zygote, the authors observed roughly 1 mutation per cell division prior to the point of twinning. In the realm of prenatal diagnosis, one should consider that monochorionic twins with different phenotypes (that is, an ultrasonography anomaly in 1 of the twin pair) could represent a genetic change rather than an environmental difference. This genetic change may not be shared by the other twin despite originating from the same primordial cell line. The genetic changes that the authors investigated were detected on genome sequencing, which is much more comprehensive than the exome sequencing that is increasingly utilized in rare disease diagnosis. The clinical utility of this observation in prenatal diagnosis has yet to be proven, but this study provides preliminary data that 15% of monozygotic twins have genetic differences and may warrant individualized testing.
The genetic landscape of the placenta
Coorens TH, Oliver TR, Sanghvi R, et al. Inherent mosaicism and extensive mutation of human placentas. Nature. Published online March 10, 2021. doi:10.1038/ s41586-021-03345-1.
Confined placental mosaicism (CPM) is a phenomenon in which the genetics of the placenta are different from those of the fetus. Historically, this phenomenon has been described in 1% to 2% of pregnancies based on karyotype data obtained from chorionic villus sampling. Some studies have demonstrated adverse pregnancy outcomes in the setting of CPM, thought to be secondary to aneuploid cells in the placenta leading to insufficiency or dysfunction.
Although our sophistication and level of detail in prenatal genetic testing has rapidly expanded to include information about copy number variants and singlenucleotide changes, their contribution to CPM has been understudied. Coorens and colleagues recently published a landmark study that describes a surprisingly high rate of mosaicism for these smaller genetic changes.
A cohort study of placentas
The authors performed whole genome sequencing on placental samples obtained from 37 term pregnancies. Umbilical cord tissue and maternal blood also were collected and served as controls for fetal and maternal genetic profiles, respectively.
In a subgroup of 5 placentas, lasercapture microscopy was used to separate placental cells of different origins, including trophoblastic cells, mesenchymal core cells, and cells originating from the inner cell mass. To investigate variation within different geographic regions of a single placenta, these cell lines were derived multiple times from each quadrant of the 5 placentas.
Placental biopsies revealed “bottlenecks” of genetic differentiation
Genome sequencing was used uniquely in this study to help delineate the phylogeny of placental cells by tracking somatic mutations both in different geographic locations of each placenta and between different cells of origin within 1 placenta.
The authors concluded that bottlenecks of differentiation in placental development led to unique genetic signatures in every bulk placental sample studied. Their findings led them to describe the placenta as a “patchwork” of independent genetic units resulting from clonal expansion at different stages of embryonic development.
Early insights into human placental cells
This study provides fascinating insight into the surprisingly high rates of copy number variants and single-gene changes that exist, in mosaic form, within human placentas. The authors distinguish the placenta from other human organs (such as the colon, endometrium, liver, and skin) in which many fewer genetic changes exist. In fact, they suggest parallels between the “mutational signature” of the placenta with rapidly dividing neoplastic cells.
As one of the first investigations into the variation and complexity of genetic changes within the placenta, this study was not designed to draw conclusions regarding the clinical impact of the numerous genetic changes described. Further studies will elucidate the potential contribution of genetically mosaic placentas to common adverse obstetric outcomes. ●
With a new appreciation for the smaller genetic alterations that exist within placental tissue, it appears that the rate of CPM has been vastly underestimated. We know that aneuploid placental cells increase the risk of adverse pregnancy outcomes and we may learn more about the contribution of copy number variants and single-nucleotide changes to preeclampsia, growth restriction, and pregnancy loss. Furthermore, as the applications of cell-free fetal DNA (cffDNA) in genetic screening continue to expand, we must exercise caution in assuming that copy number variants or single-nucleotide changes detected by cffDNA reflect those of the developing fetus.
Prenatal diagnosis has expanded from identification of aneuploidy to include copy number variants detected on microarray (such as 22q11 deletion syndrome) and now single-gene disorders identified by targeted or exome and genome sequencing. How and when different sequencing tests should be used clinically are questions faced by every provider engaged in modern prenatal diagnosis.
In this Update, we highlight new clinical insights into prenatal sequencing and explore how information gained from sequencing may help us understand some of the unanswered questions in obstetrics.
What is the yield of a RASopathy gene panel with specific prenatal findings?
Scott A, Di Giosaffatte N, Pinna V, et al. When to test fetuses for RASopathies? Proposition from a systematic analysis of 352 multicenter cases and a postnatal cohort. Genet Med. Published online February 10, 2021. doi:10.1038/s41436-020-01093-7.
RASopathies, a group of genetic conditions caused by mutations in the RAS/mitogen-activated protein kinase (RAS-MAPK) pathway, are common, occurring in 1:1,000 to 1:2,500 live births. RASopathies are much more common than 22q11 deletion syndrome, or DiGeorge syndrome, which occurs in 1.4:10,000 live births.1
RASopathy disorders include Noonan syndrome, Noonan syndrome with multiple lentigines, Costello syndrome, cardiofaciocutaneous syndrome, and Noonan-like syndrome with loose anagen hair. These are autosomal dominant disorders caused by a pathogenic variant (or mutation) in 1 of more than 20 genes in the RAS-MAPK signaling pathway in the body. Clinical features include congenital anomalies of the kidney and urinary tract, lymphatic anomalies, congenital heart disease (CHD), hypertrophic cardiomyopathy (HCM), postnatal growth disorders, neurodevelopmental disorders, and more rarely hematologic malignancies. Prenatal clues include an increased nuchal translucency (NT), CHD, cystic hygroma, lymphatic anomalies, anomalies of the kidney and urinary tract, hydrops, and HCM.
Cohort of pregnancies that received a RASopathy panel
Scott and colleagues sought to clarify the utility of testing for RASopathies with a prenatal gene panel. They conducted a multicenter retrospective cohort study with cases from 2 hospitals in Italy and Canada; data were collected between 2012 and 2019.
Eligible fetuses were those referred to the prenatal genetics clinic because of an increased NT, increased nuchal fold (NF), hydrops, ascites, thoracic effusions, chylothorax, other lymphatic anomalies, CHD, or HCM with a nondiagnostic (negative) microarray or karyotype. All eligible cases had RASopathy molecular testing in the prenatal or neonatal period.
Among the 352 referrals to clinic, 50 cases of a RASopathy disorder were diagnosed. Additionally, to complement this cohort over the same time period, 25 postnatal diagnoses were made after retrospective review performed to ascertain additional prenatal findings. The size of the testing panel ranged from 9 to 20 genes, which were sent to clinical laboratories that performed sequencing based on standard protocols.
Study outcomes
Overall, 14% of fetuses with an indication for testing had a pathogenic or likely pathogenic variant (diagnostic) on panel testing among 11 genes (notably, all presented results are after excluding copy number variants and aneuploidy). Fetuses with only 1 ultrasonography finding were much less likely to have a positive result than those with more than 1 ultrasonography finding, 3% versus 18%. The highest diagnostic yields were for HCM at 69%; thoracic effusions and ascites, 41%; persistent hydrops, 39%; cystic hygroma combined with another suggestive ultrasonography finding, 28%; CHD, 23%; and persistent cystic hygroma, 21%. Five fetuses were affected with CHD and HCM, and 44% had an intrauterine fetal demise.
Importance of NT size. An isolated increased NT had a diagnostic yield of 1% overall (1/90); however, the size of the NT mattered. Seventeen fetuses had an NT between 3 and 3.5 mm and none of these had diagnostic sequencing, whereas 26% with an NT greater than 6 mm had a diagnostic result (11/43). An increased NF had a diagnostic yield of 25%.
Other findings. Of fetuses with a cystic hygroma, 16% had a pathogenic or likely pathogenic variant, and when these persisted into the second trimester or were associated with other anomalies, the percentages increased to 21% and 28%, respectively. Of prenatal patients, 20.6% had variants of uncertain significance, and 12% of the pathogenic and likely pathogenic variants were inherited, which is less than previously reported series. Additionally, 48% of the postnatal RASopathy diagnosis group did not have an ultrasonography finding on record review.
Continue to: Study strengths and limitations...
Study strengths and limitations
This study presents a large cohort of prenatal and neonatal patients tested for RASopathies at 2 international centers with very granular and clinically useful data about ultrasonography findings and yield of panel testing. Prenatal care providers, geneticists, and computational biologists may find this study of great interest and take away useful information and ideas due to the authors’ presentation and details.
The number of genes tested changed over the inclusion time period, but this is an inescapable reality of retrospective clinical research in an advancing field. The authors presented the prenatal and postnatal diagnoses ultrasonography findings separately and together. Given the different nature of cohort ascertainment, we prefer to consider these groups separately and have presented the data for the prenatal group.
Prenatal sequencing panels and exome sequencing are detecting disorders with important implications for prenatal care. If your practice is not testing for RASopathies in prenatal patients with concerning ultrasonography features, you are missing cases. In this study, the most concerning ultrasonography features (more than 20% diagnosis) were HCM, thoracic effusions and ascites, persistent hydrops, cystic hygroma combined with another suggestive ultrasonography finding, CHD, and persistent cystic hygroma. Isolated ultrasonography findings or findings that resolved had a lower diagnostic yield, and an isolated enlarged NT had a 1% diagnostic yield, with most cases having an NT larger than 6 mm.
For pretest counseling, in this study 20% of patients had a variant of uncertain significance, and preparing patients for this possibility is crucial. Most variants of uncertain significance are reclassified to benign when more information is available. Providers can consider sending parental samples concurrently with the fetal sample to help obtain useful information quickly, although the possibility of an inherited pathogenic variant still exists (12% in this study).
Prenatal diagnosis gives your patients the opportunity to learn about the disorder, plan for treatment and delivery location, and establish their care team before birth or consider pregnancy termination.
Sequencing provides insights into twin pregnancies
Jonsson H, Magnusdottir E, Eggertsson HP, et al. Differences between germline genomes of monozygotic twins. Nat Genet. 2021;53:27-34. doi:10.1038/s41588 -020-00755-1.
You have a monozygotic twin pair with an anomaly and intend to do diagnostic testing for prenatal diagnosis. The question always arises: Do you sample both twins or just one? Surely, they are genetically identical? A wise mentor once instilled a valuable lesson: Monozygotic twins are more likely to have an anomaly. Their existence is already out of the realm of normal. Finally, we now have an engaging and interesting answer to this and other fascinating embryology questions through the work of Jonsson and colleagues.
Study eligibility criteria and treatment protocol
The authors enrolled 381 twin pairs and 2 monozygotic triplets and compared genome sequencing of different tissues (cheek cells and blood). They went further to assess what other tissues might share the genetic change. To do this, they sequenced the children and the partners of 181 of the pairs. Presumably, if a twin and their offspring shared a genetic change that was not present in the spouse or twin, this genetic change must be present in the oocytes or sperm of the parent twin. The goal of sequencing multiple tissue sources in each twin was to help determine when the genetic change occurred in embryonic development.
Study outcomes
The authors found that 15% of twins had mutations that were absent in the other twin. Because of the extent of tissues that had the genetic change, the authors asserted that these changes must have occurred very early in embryonic development (even from one cell after twinning) for the changes to be near-constitutional (among sampled tissues).
An average of 14 genetic differences were found between twin pairs that developed after twinning. However, the number of differences varied. For example, 39 pairs of twins differed by more than 100 changes, and 38 did not differ at all. Differences between twins were more likely in blood samples than in cheek swabs, suggesting that some differences were due to acquired genetic changes in hematologic cell lines, or clonal hematopoiesis.
The authors also looked at what percentage of sequenced DNA contained the variants (or mutations) and found that many of these DNA differences were present at high amounts in sequencing reads. This suggests that the DNA changes happened very early after twinning in about one-third of pairs. Additionally, if one twin had a near-constitutional change, in 42% of pairs the other twin had a different near-constitutional change. Among the triplets, 2 of a triplet pair shared more genetic similarity and were likely descendent from a single split cell and the third likely was formed from a different set of cells.
By examining the offspring of twins, Jonsson and colleagues found that there were 2.6 early embryonic mutations, and this did not differ when blood or buccal DNA was compared. The rate of transmission of a variant to offspring was proportional to the variant allele frequency (proportion of alternate alleles) in the blood or buccal cells. This is an important counseling point when considering patients with mosaic genetic disorders and counseling about the likelihood of inheritance or transmission to future offspring. If the rate of mosaicism was higher in blood or buccal cells, the likelihood of transmission was higher. Additionally, the mutations did not differ by sex, and there was no relationship to whether the chromosome was maternally or paternally inherited.
Continue to: Study strengths and limitations...
Study strengths and limitations
The authors did not have access to information about chorionicity of the monozygotic twin pairs. Consequently, they were unable to correlate chorionicity with the degree of noted genetic difference between the monozygotic twin pairs. Additionally, although the authors were thoughtful in their utilization of offspring and spouses to infer germline genomic content, the study had a limited number of tissues sampled, which could reduce the applicability. However, the sample size, clinically accessible tissue sampling, and thoughtful analysis used in this study make it an interesting and relevant contribution to reproductive medicine and evolutionary biology.
We all accumulate changes to our DNA throughout life. The study by Jonsson and colleagues illustrates that for many, this accumulation of genetic changes starts very early in gestation. In the early zygote, the authors observed roughly 1 mutation per cell division prior to the point of twinning. In the realm of prenatal diagnosis, one should consider that monochorionic twins with different phenotypes (that is, an ultrasonography anomaly in 1 of the twin pair) could represent a genetic change rather than an environmental difference. This genetic change may not be shared by the other twin despite originating from the same primordial cell line. The genetic changes that the authors investigated were detected on genome sequencing, which is much more comprehensive than the exome sequencing that is increasingly utilized in rare disease diagnosis. The clinical utility of this observation in prenatal diagnosis has yet to be proven, but this study provides preliminary data that 15% of monozygotic twins have genetic differences and may warrant individualized testing.
The genetic landscape of the placenta
Coorens TH, Oliver TR, Sanghvi R, et al. Inherent mosaicism and extensive mutation of human placentas. Nature. Published online March 10, 2021. doi:10.1038/ s41586-021-03345-1.
Confined placental mosaicism (CPM) is a phenomenon in which the genetics of the placenta are different from those of the fetus. Historically, this phenomenon has been described in 1% to 2% of pregnancies based on karyotype data obtained from chorionic villus sampling. Some studies have demonstrated adverse pregnancy outcomes in the setting of CPM, thought to be secondary to aneuploid cells in the placenta leading to insufficiency or dysfunction.
Although our sophistication and level of detail in prenatal genetic testing has rapidly expanded to include information about copy number variants and singlenucleotide changes, their contribution to CPM has been understudied. Coorens and colleagues recently published a landmark study that describes a surprisingly high rate of mosaicism for these smaller genetic changes.
A cohort study of placentas
The authors performed whole genome sequencing on placental samples obtained from 37 term pregnancies. Umbilical cord tissue and maternal blood also were collected and served as controls for fetal and maternal genetic profiles, respectively.
In a subgroup of 5 placentas, lasercapture microscopy was used to separate placental cells of different origins, including trophoblastic cells, mesenchymal core cells, and cells originating from the inner cell mass. To investigate variation within different geographic regions of a single placenta, these cell lines were derived multiple times from each quadrant of the 5 placentas.
Placental biopsies revealed “bottlenecks” of genetic differentiation
Genome sequencing was used uniquely in this study to help delineate the phylogeny of placental cells by tracking somatic mutations both in different geographic locations of each placenta and between different cells of origin within 1 placenta.
The authors concluded that bottlenecks of differentiation in placental development led to unique genetic signatures in every bulk placental sample studied. Their findings led them to describe the placenta as a “patchwork” of independent genetic units resulting from clonal expansion at different stages of embryonic development.
Early insights into human placental cells
This study provides fascinating insight into the surprisingly high rates of copy number variants and single-gene changes that exist, in mosaic form, within human placentas. The authors distinguish the placenta from other human organs (such as the colon, endometrium, liver, and skin) in which many fewer genetic changes exist. In fact, they suggest parallels between the “mutational signature” of the placenta with rapidly dividing neoplastic cells.
As one of the first investigations into the variation and complexity of genetic changes within the placenta, this study was not designed to draw conclusions regarding the clinical impact of the numerous genetic changes described. Further studies will elucidate the potential contribution of genetically mosaic placentas to common adverse obstetric outcomes. ●
With a new appreciation for the smaller genetic alterations that exist within placental tissue, it appears that the rate of CPM has been vastly underestimated. We know that aneuploid placental cells increase the risk of adverse pregnancy outcomes and we may learn more about the contribution of copy number variants and single-nucleotide changes to preeclampsia, growth restriction, and pregnancy loss. Furthermore, as the applications of cell-free fetal DNA (cffDNA) in genetic screening continue to expand, we must exercise caution in assuming that copy number variants or single-nucleotide changes detected by cffDNA reflect those of the developing fetus.
- Roberts AE, Allanson JE, Tartaglia M, et al. Noonan syndrome. Lancet. 2013;381:333-342. doi:10.1016/S0140-6736(12)61023-X.
- Roberts AE, Allanson JE, Tartaglia M, et al. Noonan syndrome. Lancet. 2013;381:333-342. doi:10.1016/S0140-6736(12)61023-X.
Retail health clinics: What is their role in ObGyn care?
Retail Health Clinics (RHCs) are health care facilities located in high-traffic retail outlets with adjacent pharmacies that are intended to provide convenient and affordable care without sacrificing quality. The clinics add an option that complements services to individuals and families who otherwise would need to wait for an appointment with a traditional primary care physician or provider.1 Appointments are not necessary for episodic health needs. Usually open 7 days a week, RHCs offer extended hours on weekdays.2
The clinics are staffed by licensed, qualified advance practice providers, such as nurse practitioners and physician assistants, who are supervised by family physicians where required by state law. These clinicians have advanced education to diagnose, treat, and prescribe for nonemergent ailments such as colds and flu, rashes and skin irritation, and muscle strains or sprains.3 They are supported by an electronic health record that contains established evidence-based protocols.2,4
Evolution of retail health clinics
The first RHC, operated by QuickMedx, opened its doors in 2000 in Minneapolis– St. Paul.1,5 Only patients with a very limited number of illnesses were seen, and payment was cash. In 2005, this clinic was acquired by a major pharmacy retailer, which led to several acquisitions by other retailers and health care systems. In addition to accepting cash for a visit, the clinics formed contracts with health insurance companies. The average cost of a visit to an RHC in 2016 was estimated to be $70, considerably less than the cost at urgent care clinics ($124) and emergency rooms ($356).6
Today, more than 20 companies provide health care services at RHCs (TABLE 1). CVS (MinuteClinic) has the most retail clinics, followed by Walgreens, Kroger, Rite Aid, and Zoom+Care.1,2 There are now about 3,300 clinics in the United States, Canada, and Mexico (nearly all are located in the United States).2 Currently, RHCs are present in 44 states and the District of Columbia. Alabama, Alaska, Idaho, North Dakota, Vermont, and Wyoming are the only states without an RHC, while large-population states (California, Florida, Ohio, Pennsylvania, and Texas) have experienced an explosion in clinic openings.2
RHCs are found in high foot traffic locations, such as large retailers and grocery stores, and in prioritizing services such as drug stores. By analyzing 2019 clinic openings and population centers, the Convenient Care Association determined that more than half of the US population now lives within a 10-minute drive of an RHC.2 New locations are established on a regular basis, resulting in some flux in the total number of clinics.
Continue to: Services that RHCs provide...
Services that RHCs provide
As RHC locations expand, so do their services. Most RHCs pursue 1 of 2 models: health hubs or virtual care. Health hubs offer an expansion of services, which has resulted in retail health looking and operating more like the primary care providers in their communities.1,2 While most chains intend for patient visits to be brief, the growing health hub model intends to expand the period for patient visits. The major companies, CVS, Kroger, and Walmart, are offering an increase in their services and granting their providers a greater capacity to screen and treat patients for a wider range of conditions. By contrast, other clinic operators, such as Rite Aid RediClinics, are pursuing a more episodic and convenient care model with a greater adoption and expansion of telehealth and telemedicine.
Services at RHCs involve primarily acute care as well as some basic chronic disease management. About 90% of visits are for the following conditions: influenza, immunizations, upper respiratory infections, sinusitis, bronchitis, sore throat, inner ear infection, conjunctivitis, urinary tract infections, and blood tests.1-3 Other services available at most RHC locations involve screening and monitoring, wellness and physicals, travel health, treatment of minor injuries, and vaccinations and injections.
Women constitute half of all customers, and all RHCs offer women’s health services.7 Along with addressing acute care needs, women’s health services include contraception care and options, human papillomavirus (HPV) screening, pregnancy testing and initial prenatal evaluation, and evaluation for and treatment of urinary tract, bladder, and yeast infections.2,6
All RHCs provide counseling on sexual health concerns. Nearly all retail clinics in the United States provide screening and treatment for patients and their partners with sexually transmitted infections. RHC providers are required to follow up with patients regarding any blood work or culture results. When positive test results are confirmed for serious infections, such as hepatitis B and C, syphilis, and HIV, patients customarily are referred for treatment.2
The RHC patient base
RHCs serve an expanding base of patients who cite convenience as their primary motivation for utilizing these clinics. This consumer-driven market now encompasses, by some measures, nearly 50 million visits annually.2 These numbers have risen every year alongside a consistent increase in the number and spread of clinics across the country. During the COVID-19 pandemic, visits declined, consistent with other health care touchpoints, due to concerns about spreading the coronavirus.
The RHC industry has continued to adapt to a changing health care climate by embracing new telehealth solutions, enabling remote care, and expanding services by consumer demand.1,7 While convenience is a primary motivation for visiting an RHC, about two-thirds of RHC patients do not have a primary care provider.1 To support a broader continuum of care, RHCs regularly refer patients who do not have a primary care provider to other health care touchpoints when necessary.
Young and middle-aged adults (18–44 years) comprise the largest group of RHC patients. When patients were asked why they chose an RHC over “traditional doctors’ clinics,” many cited difficulties in accessing care, the appeal of lower costs, and proximity. The proportion of female RHC users was 50.9% and 56.8% for RHC nonusers.1-3
How RHCs compare with other episodic care clinics
The consumer has more choices to seek episodic care other than at physicians’ clinics or emergency rooms. An RHC, urgent care clinic, or freestanding emergency clinic increase access points for consumers. Along with the expanding number of RHCs, there are nearly 9,000 US urgent care centers, according to the Urgent Care Association, and more than 550 freestanding emergency rooms.8
The main differences between these episodic care clinics are shown in TABLE 2. Hours of operation, types of conditions, available providers, location of the facility, and estimated costs are compared. All provide expanded business hours. Retail clinics address some chronic disease management along with acute care, engage only advanced practitioners, use retail stores, and are less costly to consumers.3,4,9
An RHC, urgent care clinic, or emergency department increases access points for consumers. Many emergency department visits can be handled in ambulatory settings such as RHCs and urgent care clinics.9,10 This can be helpful, especially in rural areas with a shortage of physicians. Most people want a relationship with a physician who will manage their care rather than seeing a different provider at every visit. While ObGyns deliver comprehensive care to women, however, in some underserved areas non-ObGyn clinics can fill the void. For example, RHCs can sometimes provide needed immunizations and health care information required in underserved areas.11
Many physicians are frustrated when they see patients who do not have a complete copy of their medical record and must piecemeal how to treat a patient. RHCs have adopted electronic medical records, and they regularly encourage patients to contact their physician (or find one, which can be difficult). Another limitation can be a referral from an RHC to a subspecialist rather than a primary care physician who could equally handle the condition.
Continue to: What ObGyns can do...
What ObGyns can do
Consumers have become accustomed to obtaining services where and when they want them, and they expect the same from their health care providers. While ObGyn practices are less affected by RHCs than family physicians or general internists, health care delivery in traditional clinics must be user friendly—that is, better, cheaper, and faster—for the patient-consumer to be more satisfied. Looking ahead, a nearby women’s health care group needs to have someone on call 24 hours a day, 7 days a week. That way, you can tell your patients that they can call you first if they need help. In the case of an ObGyn recommending that a patient go to an RHC or urgent care center, you will be aware of the visit and can follow up with your patient afterward.
Traditional clinics need to create ways for patients with acute illnesses to be seen that same day. Offering extended hours or technology options, such as online support, can help. Text message reminders, same-day access for appointments, and price transparency are necessary. It is important to encourage your women’s health patients to become more responsible for their own health and care, while taking into consideration their social determinants of health. While ObGyns should discuss with their patients when to visit an RHC (especially when their clinic is closed), emphasize that your own clinic is the patient’s medical home and encourage the importance of communicating what occurred during the RHC visit.
Working as a team by communicating well can create a community of health. It would be appropriate for you to represent your group by meeting practitioners at the nearby RHC. Being accessible and helpful would create a friendly and open professional relationship. Conversely, providers at retail clinics need to continually appreciate that women’s health clinics offer more comprehensive care. Select referrals from RHCs would help the most important person, the patient herself. ●
- Bachrach D, Frohlich J, Garcimonde A, et al. Building a culture of health: the value proposition of retail clinics. Robert Wood Johnson Foundation and Manatt Health. April 2015. https://www.rwjf.org/en/library/research/2015/04/the-value-proposition-of-retail-clinics.html. Accessed March 26, 2021.
- Bronstein N. Convenient Care Association–National Trade Association of Companies and Healthcare Systems for the Convenient Care Industry, January 1, 2020. https://www.ccaclinics.org/about-us/about-cca. Accessed January 10, 2021.
- Mehrotra A, Liu H, Adams JL, et al. Comparing costs and quality of care at retail clinics with that of other medical settings for 3 common illnesses. Ann Intern Med. 2009;151:321-328.
- Woodburn JD, Smith KL, Nelson GD. Quality of care in the retail health care setting using national clinical guidelines for acute pharyngitis. Am J Med Qual. 2007;22:457-462.
- Zamosky L. What retail clinic growth can teach physicians about patient demand. Threat or opportunity: retail clinic popularity is about convenience. Med Econ. 2014;91:22-24.
- SolvHealth website. Urgent care center vs emergency room. https://www.solvhealth.com/faq/urgent-care-center-vs-emergency-room. Accessed January, 13, 2021.
- Kvedar J, Coye MJ, Everett W. Connected health: a review of technologies and strategies to improve patient care with telemedicine and telehealth. Health Aff. 2014;33:194-199.
- Urgent Care Association website. Industry news: urgent care industry grows to more than 9,000 centers nationwide. February 24, 2020. https://www.ucaoa.org/About-UCA/Industry-News/ArtMID/10309/ArticleID/1468/INDUSTRY-NEWS-Urgent-Care-Industry-Grows-to-More-than-9000-Centers-Nationwide. Accessed March 26, 2021.
- Sussman A, Dunham L, Snower K, et al. Retail clinic utilization associated with lower total cost of care. Am J Manag Care. 2013;19:e148-57.
- Weinik RM, Burns RM, Mehrotra A. Many emergency department visits could be managed at urgent care centers and retail clinics. Health Aff. 2010;29:1630-1636.
- Goad JA, Taitel MS, Fensterheim LE, et al. Vaccinations administered during off-clinic hours at a national community pharmacy: implications for increasing patient access and convenience. Ann Fam Med. 2013;11:429-436.
Retail Health Clinics (RHCs) are health care facilities located in high-traffic retail outlets with adjacent pharmacies that are intended to provide convenient and affordable care without sacrificing quality. The clinics add an option that complements services to individuals and families who otherwise would need to wait for an appointment with a traditional primary care physician or provider.1 Appointments are not necessary for episodic health needs. Usually open 7 days a week, RHCs offer extended hours on weekdays.2
The clinics are staffed by licensed, qualified advance practice providers, such as nurse practitioners and physician assistants, who are supervised by family physicians where required by state law. These clinicians have advanced education to diagnose, treat, and prescribe for nonemergent ailments such as colds and flu, rashes and skin irritation, and muscle strains or sprains.3 They are supported by an electronic health record that contains established evidence-based protocols.2,4
Evolution of retail health clinics
The first RHC, operated by QuickMedx, opened its doors in 2000 in Minneapolis– St. Paul.1,5 Only patients with a very limited number of illnesses were seen, and payment was cash. In 2005, this clinic was acquired by a major pharmacy retailer, which led to several acquisitions by other retailers and health care systems. In addition to accepting cash for a visit, the clinics formed contracts with health insurance companies. The average cost of a visit to an RHC in 2016 was estimated to be $70, considerably less than the cost at urgent care clinics ($124) and emergency rooms ($356).6
Today, more than 20 companies provide health care services at RHCs (TABLE 1). CVS (MinuteClinic) has the most retail clinics, followed by Walgreens, Kroger, Rite Aid, and Zoom+Care.1,2 There are now about 3,300 clinics in the United States, Canada, and Mexico (nearly all are located in the United States).2 Currently, RHCs are present in 44 states and the District of Columbia. Alabama, Alaska, Idaho, North Dakota, Vermont, and Wyoming are the only states without an RHC, while large-population states (California, Florida, Ohio, Pennsylvania, and Texas) have experienced an explosion in clinic openings.2
RHCs are found in high foot traffic locations, such as large retailers and grocery stores, and in prioritizing services such as drug stores. By analyzing 2019 clinic openings and population centers, the Convenient Care Association determined that more than half of the US population now lives within a 10-minute drive of an RHC.2 New locations are established on a regular basis, resulting in some flux in the total number of clinics.
Continue to: Services that RHCs provide...
Services that RHCs provide
As RHC locations expand, so do their services. Most RHCs pursue 1 of 2 models: health hubs or virtual care. Health hubs offer an expansion of services, which has resulted in retail health looking and operating more like the primary care providers in their communities.1,2 While most chains intend for patient visits to be brief, the growing health hub model intends to expand the period for patient visits. The major companies, CVS, Kroger, and Walmart, are offering an increase in their services and granting their providers a greater capacity to screen and treat patients for a wider range of conditions. By contrast, other clinic operators, such as Rite Aid RediClinics, are pursuing a more episodic and convenient care model with a greater adoption and expansion of telehealth and telemedicine.
Services at RHCs involve primarily acute care as well as some basic chronic disease management. About 90% of visits are for the following conditions: influenza, immunizations, upper respiratory infections, sinusitis, bronchitis, sore throat, inner ear infection, conjunctivitis, urinary tract infections, and blood tests.1-3 Other services available at most RHC locations involve screening and monitoring, wellness and physicals, travel health, treatment of minor injuries, and vaccinations and injections.
Women constitute half of all customers, and all RHCs offer women’s health services.7 Along with addressing acute care needs, women’s health services include contraception care and options, human papillomavirus (HPV) screening, pregnancy testing and initial prenatal evaluation, and evaluation for and treatment of urinary tract, bladder, and yeast infections.2,6
All RHCs provide counseling on sexual health concerns. Nearly all retail clinics in the United States provide screening and treatment for patients and their partners with sexually transmitted infections. RHC providers are required to follow up with patients regarding any blood work or culture results. When positive test results are confirmed for serious infections, such as hepatitis B and C, syphilis, and HIV, patients customarily are referred for treatment.2
The RHC patient base
RHCs serve an expanding base of patients who cite convenience as their primary motivation for utilizing these clinics. This consumer-driven market now encompasses, by some measures, nearly 50 million visits annually.2 These numbers have risen every year alongside a consistent increase in the number and spread of clinics across the country. During the COVID-19 pandemic, visits declined, consistent with other health care touchpoints, due to concerns about spreading the coronavirus.
The RHC industry has continued to adapt to a changing health care climate by embracing new telehealth solutions, enabling remote care, and expanding services by consumer demand.1,7 While convenience is a primary motivation for visiting an RHC, about two-thirds of RHC patients do not have a primary care provider.1 To support a broader continuum of care, RHCs regularly refer patients who do not have a primary care provider to other health care touchpoints when necessary.
Young and middle-aged adults (18–44 years) comprise the largest group of RHC patients. When patients were asked why they chose an RHC over “traditional doctors’ clinics,” many cited difficulties in accessing care, the appeal of lower costs, and proximity. The proportion of female RHC users was 50.9% and 56.8% for RHC nonusers.1-3
How RHCs compare with other episodic care clinics
The consumer has more choices to seek episodic care other than at physicians’ clinics or emergency rooms. An RHC, urgent care clinic, or freestanding emergency clinic increase access points for consumers. Along with the expanding number of RHCs, there are nearly 9,000 US urgent care centers, according to the Urgent Care Association, and more than 550 freestanding emergency rooms.8
The main differences between these episodic care clinics are shown in TABLE 2. Hours of operation, types of conditions, available providers, location of the facility, and estimated costs are compared. All provide expanded business hours. Retail clinics address some chronic disease management along with acute care, engage only advanced practitioners, use retail stores, and are less costly to consumers.3,4,9
An RHC, urgent care clinic, or emergency department increases access points for consumers. Many emergency department visits can be handled in ambulatory settings such as RHCs and urgent care clinics.9,10 This can be helpful, especially in rural areas with a shortage of physicians. Most people want a relationship with a physician who will manage their care rather than seeing a different provider at every visit. While ObGyns deliver comprehensive care to women, however, in some underserved areas non-ObGyn clinics can fill the void. For example, RHCs can sometimes provide needed immunizations and health care information required in underserved areas.11
Many physicians are frustrated when they see patients who do not have a complete copy of their medical record and must piecemeal how to treat a patient. RHCs have adopted electronic medical records, and they regularly encourage patients to contact their physician (or find one, which can be difficult). Another limitation can be a referral from an RHC to a subspecialist rather than a primary care physician who could equally handle the condition.
Continue to: What ObGyns can do...
What ObGyns can do
Consumers have become accustomed to obtaining services where and when they want them, and they expect the same from their health care providers. While ObGyn practices are less affected by RHCs than family physicians or general internists, health care delivery in traditional clinics must be user friendly—that is, better, cheaper, and faster—for the patient-consumer to be more satisfied. Looking ahead, a nearby women’s health care group needs to have someone on call 24 hours a day, 7 days a week. That way, you can tell your patients that they can call you first if they need help. In the case of an ObGyn recommending that a patient go to an RHC or urgent care center, you will be aware of the visit and can follow up with your patient afterward.
Traditional clinics need to create ways for patients with acute illnesses to be seen that same day. Offering extended hours or technology options, such as online support, can help. Text message reminders, same-day access for appointments, and price transparency are necessary. It is important to encourage your women’s health patients to become more responsible for their own health and care, while taking into consideration their social determinants of health. While ObGyns should discuss with their patients when to visit an RHC (especially when their clinic is closed), emphasize that your own clinic is the patient’s medical home and encourage the importance of communicating what occurred during the RHC visit.
Working as a team by communicating well can create a community of health. It would be appropriate for you to represent your group by meeting practitioners at the nearby RHC. Being accessible and helpful would create a friendly and open professional relationship. Conversely, providers at retail clinics need to continually appreciate that women’s health clinics offer more comprehensive care. Select referrals from RHCs would help the most important person, the patient herself. ●
Retail Health Clinics (RHCs) are health care facilities located in high-traffic retail outlets with adjacent pharmacies that are intended to provide convenient and affordable care without sacrificing quality. The clinics add an option that complements services to individuals and families who otherwise would need to wait for an appointment with a traditional primary care physician or provider.1 Appointments are not necessary for episodic health needs. Usually open 7 days a week, RHCs offer extended hours on weekdays.2
The clinics are staffed by licensed, qualified advance practice providers, such as nurse practitioners and physician assistants, who are supervised by family physicians where required by state law. These clinicians have advanced education to diagnose, treat, and prescribe for nonemergent ailments such as colds and flu, rashes and skin irritation, and muscle strains or sprains.3 They are supported by an electronic health record that contains established evidence-based protocols.2,4
Evolution of retail health clinics
The first RHC, operated by QuickMedx, opened its doors in 2000 in Minneapolis– St. Paul.1,5 Only patients with a very limited number of illnesses were seen, and payment was cash. In 2005, this clinic was acquired by a major pharmacy retailer, which led to several acquisitions by other retailers and health care systems. In addition to accepting cash for a visit, the clinics formed contracts with health insurance companies. The average cost of a visit to an RHC in 2016 was estimated to be $70, considerably less than the cost at urgent care clinics ($124) and emergency rooms ($356).6
Today, more than 20 companies provide health care services at RHCs (TABLE 1). CVS (MinuteClinic) has the most retail clinics, followed by Walgreens, Kroger, Rite Aid, and Zoom+Care.1,2 There are now about 3,300 clinics in the United States, Canada, and Mexico (nearly all are located in the United States).2 Currently, RHCs are present in 44 states and the District of Columbia. Alabama, Alaska, Idaho, North Dakota, Vermont, and Wyoming are the only states without an RHC, while large-population states (California, Florida, Ohio, Pennsylvania, and Texas) have experienced an explosion in clinic openings.2
RHCs are found in high foot traffic locations, such as large retailers and grocery stores, and in prioritizing services such as drug stores. By analyzing 2019 clinic openings and population centers, the Convenient Care Association determined that more than half of the US population now lives within a 10-minute drive of an RHC.2 New locations are established on a regular basis, resulting in some flux in the total number of clinics.
Continue to: Services that RHCs provide...
Services that RHCs provide
As RHC locations expand, so do their services. Most RHCs pursue 1 of 2 models: health hubs or virtual care. Health hubs offer an expansion of services, which has resulted in retail health looking and operating more like the primary care providers in their communities.1,2 While most chains intend for patient visits to be brief, the growing health hub model intends to expand the period for patient visits. The major companies, CVS, Kroger, and Walmart, are offering an increase in their services and granting their providers a greater capacity to screen and treat patients for a wider range of conditions. By contrast, other clinic operators, such as Rite Aid RediClinics, are pursuing a more episodic and convenient care model with a greater adoption and expansion of telehealth and telemedicine.
Services at RHCs involve primarily acute care as well as some basic chronic disease management. About 90% of visits are for the following conditions: influenza, immunizations, upper respiratory infections, sinusitis, bronchitis, sore throat, inner ear infection, conjunctivitis, urinary tract infections, and blood tests.1-3 Other services available at most RHC locations involve screening and monitoring, wellness and physicals, travel health, treatment of minor injuries, and vaccinations and injections.
Women constitute half of all customers, and all RHCs offer women’s health services.7 Along with addressing acute care needs, women’s health services include contraception care and options, human papillomavirus (HPV) screening, pregnancy testing and initial prenatal evaluation, and evaluation for and treatment of urinary tract, bladder, and yeast infections.2,6
All RHCs provide counseling on sexual health concerns. Nearly all retail clinics in the United States provide screening and treatment for patients and their partners with sexually transmitted infections. RHC providers are required to follow up with patients regarding any blood work or culture results. When positive test results are confirmed for serious infections, such as hepatitis B and C, syphilis, and HIV, patients customarily are referred for treatment.2
The RHC patient base
RHCs serve an expanding base of patients who cite convenience as their primary motivation for utilizing these clinics. This consumer-driven market now encompasses, by some measures, nearly 50 million visits annually.2 These numbers have risen every year alongside a consistent increase in the number and spread of clinics across the country. During the COVID-19 pandemic, visits declined, consistent with other health care touchpoints, due to concerns about spreading the coronavirus.
The RHC industry has continued to adapt to a changing health care climate by embracing new telehealth solutions, enabling remote care, and expanding services by consumer demand.1,7 While convenience is a primary motivation for visiting an RHC, about two-thirds of RHC patients do not have a primary care provider.1 To support a broader continuum of care, RHCs regularly refer patients who do not have a primary care provider to other health care touchpoints when necessary.
Young and middle-aged adults (18–44 years) comprise the largest group of RHC patients. When patients were asked why they chose an RHC over “traditional doctors’ clinics,” many cited difficulties in accessing care, the appeal of lower costs, and proximity. The proportion of female RHC users was 50.9% and 56.8% for RHC nonusers.1-3
How RHCs compare with other episodic care clinics
The consumer has more choices to seek episodic care other than at physicians’ clinics or emergency rooms. An RHC, urgent care clinic, or freestanding emergency clinic increase access points for consumers. Along with the expanding number of RHCs, there are nearly 9,000 US urgent care centers, according to the Urgent Care Association, and more than 550 freestanding emergency rooms.8
The main differences between these episodic care clinics are shown in TABLE 2. Hours of operation, types of conditions, available providers, location of the facility, and estimated costs are compared. All provide expanded business hours. Retail clinics address some chronic disease management along with acute care, engage only advanced practitioners, use retail stores, and are less costly to consumers.3,4,9
An RHC, urgent care clinic, or emergency department increases access points for consumers. Many emergency department visits can be handled in ambulatory settings such as RHCs and urgent care clinics.9,10 This can be helpful, especially in rural areas with a shortage of physicians. Most people want a relationship with a physician who will manage their care rather than seeing a different provider at every visit. While ObGyns deliver comprehensive care to women, however, in some underserved areas non-ObGyn clinics can fill the void. For example, RHCs can sometimes provide needed immunizations and health care information required in underserved areas.11
Many physicians are frustrated when they see patients who do not have a complete copy of their medical record and must piecemeal how to treat a patient. RHCs have adopted electronic medical records, and they regularly encourage patients to contact their physician (or find one, which can be difficult). Another limitation can be a referral from an RHC to a subspecialist rather than a primary care physician who could equally handle the condition.
Continue to: What ObGyns can do...
What ObGyns can do
Consumers have become accustomed to obtaining services where and when they want them, and they expect the same from their health care providers. While ObGyn practices are less affected by RHCs than family physicians or general internists, health care delivery in traditional clinics must be user friendly—that is, better, cheaper, and faster—for the patient-consumer to be more satisfied. Looking ahead, a nearby women’s health care group needs to have someone on call 24 hours a day, 7 days a week. That way, you can tell your patients that they can call you first if they need help. In the case of an ObGyn recommending that a patient go to an RHC or urgent care center, you will be aware of the visit and can follow up with your patient afterward.
Traditional clinics need to create ways for patients with acute illnesses to be seen that same day. Offering extended hours or technology options, such as online support, can help. Text message reminders, same-day access for appointments, and price transparency are necessary. It is important to encourage your women’s health patients to become more responsible for their own health and care, while taking into consideration their social determinants of health. While ObGyns should discuss with their patients when to visit an RHC (especially when their clinic is closed), emphasize that your own clinic is the patient’s medical home and encourage the importance of communicating what occurred during the RHC visit.
Working as a team by communicating well can create a community of health. It would be appropriate for you to represent your group by meeting practitioners at the nearby RHC. Being accessible and helpful would create a friendly and open professional relationship. Conversely, providers at retail clinics need to continually appreciate that women’s health clinics offer more comprehensive care. Select referrals from RHCs would help the most important person, the patient herself. ●
- Bachrach D, Frohlich J, Garcimonde A, et al. Building a culture of health: the value proposition of retail clinics. Robert Wood Johnson Foundation and Manatt Health. April 2015. https://www.rwjf.org/en/library/research/2015/04/the-value-proposition-of-retail-clinics.html. Accessed March 26, 2021.
- Bronstein N. Convenient Care Association–National Trade Association of Companies and Healthcare Systems for the Convenient Care Industry, January 1, 2020. https://www.ccaclinics.org/about-us/about-cca. Accessed January 10, 2021.
- Mehrotra A, Liu H, Adams JL, et al. Comparing costs and quality of care at retail clinics with that of other medical settings for 3 common illnesses. Ann Intern Med. 2009;151:321-328.
- Woodburn JD, Smith KL, Nelson GD. Quality of care in the retail health care setting using national clinical guidelines for acute pharyngitis. Am J Med Qual. 2007;22:457-462.
- Zamosky L. What retail clinic growth can teach physicians about patient demand. Threat or opportunity: retail clinic popularity is about convenience. Med Econ. 2014;91:22-24.
- SolvHealth website. Urgent care center vs emergency room. https://www.solvhealth.com/faq/urgent-care-center-vs-emergency-room. Accessed January, 13, 2021.
- Kvedar J, Coye MJ, Everett W. Connected health: a review of technologies and strategies to improve patient care with telemedicine and telehealth. Health Aff. 2014;33:194-199.
- Urgent Care Association website. Industry news: urgent care industry grows to more than 9,000 centers nationwide. February 24, 2020. https://www.ucaoa.org/About-UCA/Industry-News/ArtMID/10309/ArticleID/1468/INDUSTRY-NEWS-Urgent-Care-Industry-Grows-to-More-than-9000-Centers-Nationwide. Accessed March 26, 2021.
- Sussman A, Dunham L, Snower K, et al. Retail clinic utilization associated with lower total cost of care. Am J Manag Care. 2013;19:e148-57.
- Weinik RM, Burns RM, Mehrotra A. Many emergency department visits could be managed at urgent care centers and retail clinics. Health Aff. 2010;29:1630-1636.
- Goad JA, Taitel MS, Fensterheim LE, et al. Vaccinations administered during off-clinic hours at a national community pharmacy: implications for increasing patient access and convenience. Ann Fam Med. 2013;11:429-436.
- Bachrach D, Frohlich J, Garcimonde A, et al. Building a culture of health: the value proposition of retail clinics. Robert Wood Johnson Foundation and Manatt Health. April 2015. https://www.rwjf.org/en/library/research/2015/04/the-value-proposition-of-retail-clinics.html. Accessed March 26, 2021.
- Bronstein N. Convenient Care Association–National Trade Association of Companies and Healthcare Systems for the Convenient Care Industry, January 1, 2020. https://www.ccaclinics.org/about-us/about-cca. Accessed January 10, 2021.
- Mehrotra A, Liu H, Adams JL, et al. Comparing costs and quality of care at retail clinics with that of other medical settings for 3 common illnesses. Ann Intern Med. 2009;151:321-328.
- Woodburn JD, Smith KL, Nelson GD. Quality of care in the retail health care setting using national clinical guidelines for acute pharyngitis. Am J Med Qual. 2007;22:457-462.
- Zamosky L. What retail clinic growth can teach physicians about patient demand. Threat or opportunity: retail clinic popularity is about convenience. Med Econ. 2014;91:22-24.
- SolvHealth website. Urgent care center vs emergency room. https://www.solvhealth.com/faq/urgent-care-center-vs-emergency-room. Accessed January, 13, 2021.
- Kvedar J, Coye MJ, Everett W. Connected health: a review of technologies and strategies to improve patient care with telemedicine and telehealth. Health Aff. 2014;33:194-199.
- Urgent Care Association website. Industry news: urgent care industry grows to more than 9,000 centers nationwide. February 24, 2020. https://www.ucaoa.org/About-UCA/Industry-News/ArtMID/10309/ArticleID/1468/INDUSTRY-NEWS-Urgent-Care-Industry-Grows-to-More-than-9000-Centers-Nationwide. Accessed March 26, 2021.
- Sussman A, Dunham L, Snower K, et al. Retail clinic utilization associated with lower total cost of care. Am J Manag Care. 2013;19:e148-57.
- Weinik RM, Burns RM, Mehrotra A. Many emergency department visits could be managed at urgent care centers and retail clinics. Health Aff. 2010;29:1630-1636.
- Goad JA, Taitel MS, Fensterheim LE, et al. Vaccinations administered during off-clinic hours at a national community pharmacy: implications for increasing patient access and convenience. Ann Fam Med. 2013;11:429-436.