User login
Repeat gastroscopy shows real-world value for detecting malignant ulcers
Most malignancies with new ulcer were identified on initial gastroscopy in a retrospective cohort study, but it’s still worth performing follow-up procedures, according to investigators.
“Although the additional yield of malignancy at follow-up gastroscopy is low at 2%, our data supports the current strategy of repeat endoscopic assessment given variables in obtaining adequate ulcer histology and the lack of reliable endoscopic predictors of a malignant ulcer,” the study’s authors Linda Yang, MBBS, of the University of Melbourne, and colleagues wrote in the Journal of Clinical Gastroenterology.
Recommendations from the British Society of Gastroenterology emphasize the importance of repeat gastroscopy and biopsy of gastric ulcers within an 8-week period of the index gastroscopy. Additionally, the American Society of Gastrointestinal Endoscopy similarly recommends repeat gastroscopy in high-risk patients (ulcers >2 cm) within a 12-week period of the initial endoscopy. The authors noted that, despite these recommendations, there is a lack of consensus regarding timing of repeat gastroscopy, and no established ulcer biopsy protocols exist. Additionally, there is a lack of data on real-world repeat gastroscopy practices and follow-up outcomes.
To understand the current practice in gastric ulcer follow-up, Dr. Yang and researchers retrospectively examined new gastric ulcers diagnosed on gastroscopy between 2013 and 2017 at two separate Australian institutions.
Out of 795 patients (median age, 69 years; 59% male), approximately 55% (n = 440) underwent repeat gastroscopy at a median of 8 weeks after the initial endoscopy procedure. Overall, 52 patients (7%) received a malignancy diagnosis, with 83% (n = 43) of these diagnoses detected at the index gastroscopy; 2% overall received the diagnosis based on follow-up gastroscopy.
“I think these numbers would support the assumptions of most endoscopists that a small but still significant portion of new gastric ulcers will turn out to be malignant,” explained Michael DeSimone, MD, gastroenterologist at Emerson Hospital in Concord, Mass. Dr. DeSimone, who wasn’t involved in the study, said the data support the importance of “biopsy in the initial exam and bringing these patients back for a repeat endoscopy to check healing and biopsy unless malignancy was confirmed on the initial exam.”
In the study, a multivariate analysis revealed several predictors of benign ulcers, including lack of endoscopic suspicion at the index gastroscopy (odds ratio, 0.1; 95% confidence interval, 0.03-0.13; P ≤ .005), complete healing on repeat gastroscopy (OR, 0.5; 95% CI, 0.34-0.70; P = .036), and benign histology on initial biopsy (OR, 0.12; 95% CI, 0.43-0.90; P ≤ .005). However, no patient-related factors – such as H. pylori status and ethnicity – were associated with an increased likelihood of malignancy.
“Knowing that low suspicion for malignancy on initial exam and benign histology on initial biopsies predict benign ulcers ... reasonable endoscopists could feel more comfortable not repeating an exam where procedure safety is a significant concern if their suspicion was low on the index exam, especially if they had the opportunity to take initial biopsies and those ultimately show benign histology,” said Dr. DeSimone.
The investigators noted that the main reason behind 45% receiving no follow-up gastroscopy is that the ulcers had nonsuspicious appearance.
“Although not recommended, this is widely accepted clinical practice, especially in comorbid or elderly patients where the decision to undergo repeat gastroscopy requires consideration of their comorbidities, frailty, and life expectancy,” they wrote. They suggested that this, combined with high nonattendence rate in the cohort, emphasize the importance of ulcer biopsy at index gastroscopy, even in the absence of suspicious features.
Clinicians in the current study performed random gastric biopsies in 27% (n = 218) of patients. Helicobacter pylori, a component frequently described in high-risk populations, was detected in 22% of patients who had an ulcer or gastric biopsy performed.
The relatively low frequency with which random gastric biopsies were performed during the index endoscopy to look for H. pylori is a bit surprising, said Linda Lee, MD, medical director of endoscopy at Brigham and Women’s Hospital in Boston, given the bacterium remains a common and readily treatable etiology of gastric ulcers. “While it is known that yield of biopsy can be lower for H. pylori in the setting of acute upper gastrointestinal bleeding, it is still important to evaluate for this especially since biopsies carry low risk for bleeding,” explained Dr. Lee, who also wasn’t part of the study.
She added said that the study’s high negative predictive value for endoscopic suspicion of malignancy (96%) is reassuring. “This, combined with benign histology on initial biopsies, could serve to identify which patients should return for repeat endoscopy.”
“We need to ensure that more biopsies are obtained during the index endoscopy from gastric ulcers as well as randomly and that, during follow-up endoscopy, biopsies are obtained from all [partially or fully] nonhealed ulcers,” added Dr. Lee. She suggested it could be helpful to develop an evidence-based, prospectively validated algorithm and/or identify risk factors that reliably help endoscopists decide who would benefit from repeat endoscopy, “especially since there is a relatively high rate of noncompliance with a low rate of malignancy.”
A primary limitation of the study included its retrospective nature; however, the authors pointed out that the study currently represents the largest multicenter, retrospective cohort analysis of endoscopic follow-up for gastric ulcers.
“Before any change can be recommended to current clinical practice, prospective and potentially randomized studies are required to validate our findings and elucidate any high-risk features associated with malignant gastric ulcer,” the investigators wrote. Doing so could lead to reductions in health care cost and patient burden.
Some of the study authors received funding from the National Health and Medical Research Council of Australia, but the remaining authors declared having nothing to disclose. Dr. DeSimone and Dr Lee reported having no relevant conflicts.
Most malignancies with new ulcer were identified on initial gastroscopy in a retrospective cohort study, but it’s still worth performing follow-up procedures, according to investigators.
“Although the additional yield of malignancy at follow-up gastroscopy is low at 2%, our data supports the current strategy of repeat endoscopic assessment given variables in obtaining adequate ulcer histology and the lack of reliable endoscopic predictors of a malignant ulcer,” the study’s authors Linda Yang, MBBS, of the University of Melbourne, and colleagues wrote in the Journal of Clinical Gastroenterology.
Recommendations from the British Society of Gastroenterology emphasize the importance of repeat gastroscopy and biopsy of gastric ulcers within an 8-week period of the index gastroscopy. Additionally, the American Society of Gastrointestinal Endoscopy similarly recommends repeat gastroscopy in high-risk patients (ulcers >2 cm) within a 12-week period of the initial endoscopy. The authors noted that, despite these recommendations, there is a lack of consensus regarding timing of repeat gastroscopy, and no established ulcer biopsy protocols exist. Additionally, there is a lack of data on real-world repeat gastroscopy practices and follow-up outcomes.
To understand the current practice in gastric ulcer follow-up, Dr. Yang and researchers retrospectively examined new gastric ulcers diagnosed on gastroscopy between 2013 and 2017 at two separate Australian institutions.
Out of 795 patients (median age, 69 years; 59% male), approximately 55% (n = 440) underwent repeat gastroscopy at a median of 8 weeks after the initial endoscopy procedure. Overall, 52 patients (7%) received a malignancy diagnosis, with 83% (n = 43) of these diagnoses detected at the index gastroscopy; 2% overall received the diagnosis based on follow-up gastroscopy.
“I think these numbers would support the assumptions of most endoscopists that a small but still significant portion of new gastric ulcers will turn out to be malignant,” explained Michael DeSimone, MD, gastroenterologist at Emerson Hospital in Concord, Mass. Dr. DeSimone, who wasn’t involved in the study, said the data support the importance of “biopsy in the initial exam and bringing these patients back for a repeat endoscopy to check healing and biopsy unless malignancy was confirmed on the initial exam.”
In the study, a multivariate analysis revealed several predictors of benign ulcers, including lack of endoscopic suspicion at the index gastroscopy (odds ratio, 0.1; 95% confidence interval, 0.03-0.13; P ≤ .005), complete healing on repeat gastroscopy (OR, 0.5; 95% CI, 0.34-0.70; P = .036), and benign histology on initial biopsy (OR, 0.12; 95% CI, 0.43-0.90; P ≤ .005). However, no patient-related factors – such as H. pylori status and ethnicity – were associated with an increased likelihood of malignancy.
“Knowing that low suspicion for malignancy on initial exam and benign histology on initial biopsies predict benign ulcers ... reasonable endoscopists could feel more comfortable not repeating an exam where procedure safety is a significant concern if their suspicion was low on the index exam, especially if they had the opportunity to take initial biopsies and those ultimately show benign histology,” said Dr. DeSimone.
The investigators noted that the main reason behind 45% receiving no follow-up gastroscopy is that the ulcers had nonsuspicious appearance.
“Although not recommended, this is widely accepted clinical practice, especially in comorbid or elderly patients where the decision to undergo repeat gastroscopy requires consideration of their comorbidities, frailty, and life expectancy,” they wrote. They suggested that this, combined with high nonattendence rate in the cohort, emphasize the importance of ulcer biopsy at index gastroscopy, even in the absence of suspicious features.
Clinicians in the current study performed random gastric biopsies in 27% (n = 218) of patients. Helicobacter pylori, a component frequently described in high-risk populations, was detected in 22% of patients who had an ulcer or gastric biopsy performed.
The relatively low frequency with which random gastric biopsies were performed during the index endoscopy to look for H. pylori is a bit surprising, said Linda Lee, MD, medical director of endoscopy at Brigham and Women’s Hospital in Boston, given the bacterium remains a common and readily treatable etiology of gastric ulcers. “While it is known that yield of biopsy can be lower for H. pylori in the setting of acute upper gastrointestinal bleeding, it is still important to evaluate for this especially since biopsies carry low risk for bleeding,” explained Dr. Lee, who also wasn’t part of the study.
She added said that the study’s high negative predictive value for endoscopic suspicion of malignancy (96%) is reassuring. “This, combined with benign histology on initial biopsies, could serve to identify which patients should return for repeat endoscopy.”
“We need to ensure that more biopsies are obtained during the index endoscopy from gastric ulcers as well as randomly and that, during follow-up endoscopy, biopsies are obtained from all [partially or fully] nonhealed ulcers,” added Dr. Lee. She suggested it could be helpful to develop an evidence-based, prospectively validated algorithm and/or identify risk factors that reliably help endoscopists decide who would benefit from repeat endoscopy, “especially since there is a relatively high rate of noncompliance with a low rate of malignancy.”
A primary limitation of the study included its retrospective nature; however, the authors pointed out that the study currently represents the largest multicenter, retrospective cohort analysis of endoscopic follow-up for gastric ulcers.
“Before any change can be recommended to current clinical practice, prospective and potentially randomized studies are required to validate our findings and elucidate any high-risk features associated with malignant gastric ulcer,” the investigators wrote. Doing so could lead to reductions in health care cost and patient burden.
Some of the study authors received funding from the National Health and Medical Research Council of Australia, but the remaining authors declared having nothing to disclose. Dr. DeSimone and Dr Lee reported having no relevant conflicts.
Most malignancies with new ulcer were identified on initial gastroscopy in a retrospective cohort study, but it’s still worth performing follow-up procedures, according to investigators.
“Although the additional yield of malignancy at follow-up gastroscopy is low at 2%, our data supports the current strategy of repeat endoscopic assessment given variables in obtaining adequate ulcer histology and the lack of reliable endoscopic predictors of a malignant ulcer,” the study’s authors Linda Yang, MBBS, of the University of Melbourne, and colleagues wrote in the Journal of Clinical Gastroenterology.
Recommendations from the British Society of Gastroenterology emphasize the importance of repeat gastroscopy and biopsy of gastric ulcers within an 8-week period of the index gastroscopy. Additionally, the American Society of Gastrointestinal Endoscopy similarly recommends repeat gastroscopy in high-risk patients (ulcers >2 cm) within a 12-week period of the initial endoscopy. The authors noted that, despite these recommendations, there is a lack of consensus regarding timing of repeat gastroscopy, and no established ulcer biopsy protocols exist. Additionally, there is a lack of data on real-world repeat gastroscopy practices and follow-up outcomes.
To understand the current practice in gastric ulcer follow-up, Dr. Yang and researchers retrospectively examined new gastric ulcers diagnosed on gastroscopy between 2013 and 2017 at two separate Australian institutions.
Out of 795 patients (median age, 69 years; 59% male), approximately 55% (n = 440) underwent repeat gastroscopy at a median of 8 weeks after the initial endoscopy procedure. Overall, 52 patients (7%) received a malignancy diagnosis, with 83% (n = 43) of these diagnoses detected at the index gastroscopy; 2% overall received the diagnosis based on follow-up gastroscopy.
“I think these numbers would support the assumptions of most endoscopists that a small but still significant portion of new gastric ulcers will turn out to be malignant,” explained Michael DeSimone, MD, gastroenterologist at Emerson Hospital in Concord, Mass. Dr. DeSimone, who wasn’t involved in the study, said the data support the importance of “biopsy in the initial exam and bringing these patients back for a repeat endoscopy to check healing and biopsy unless malignancy was confirmed on the initial exam.”
In the study, a multivariate analysis revealed several predictors of benign ulcers, including lack of endoscopic suspicion at the index gastroscopy (odds ratio, 0.1; 95% confidence interval, 0.03-0.13; P ≤ .005), complete healing on repeat gastroscopy (OR, 0.5; 95% CI, 0.34-0.70; P = .036), and benign histology on initial biopsy (OR, 0.12; 95% CI, 0.43-0.90; P ≤ .005). However, no patient-related factors – such as H. pylori status and ethnicity – were associated with an increased likelihood of malignancy.
“Knowing that low suspicion for malignancy on initial exam and benign histology on initial biopsies predict benign ulcers ... reasonable endoscopists could feel more comfortable not repeating an exam where procedure safety is a significant concern if their suspicion was low on the index exam, especially if they had the opportunity to take initial biopsies and those ultimately show benign histology,” said Dr. DeSimone.
The investigators noted that the main reason behind 45% receiving no follow-up gastroscopy is that the ulcers had nonsuspicious appearance.
“Although not recommended, this is widely accepted clinical practice, especially in comorbid or elderly patients where the decision to undergo repeat gastroscopy requires consideration of their comorbidities, frailty, and life expectancy,” they wrote. They suggested that this, combined with high nonattendence rate in the cohort, emphasize the importance of ulcer biopsy at index gastroscopy, even in the absence of suspicious features.
Clinicians in the current study performed random gastric biopsies in 27% (n = 218) of patients. Helicobacter pylori, a component frequently described in high-risk populations, was detected in 22% of patients who had an ulcer or gastric biopsy performed.
The relatively low frequency with which random gastric biopsies were performed during the index endoscopy to look for H. pylori is a bit surprising, said Linda Lee, MD, medical director of endoscopy at Brigham and Women’s Hospital in Boston, given the bacterium remains a common and readily treatable etiology of gastric ulcers. “While it is known that yield of biopsy can be lower for H. pylori in the setting of acute upper gastrointestinal bleeding, it is still important to evaluate for this especially since biopsies carry low risk for bleeding,” explained Dr. Lee, who also wasn’t part of the study.
She added said that the study’s high negative predictive value for endoscopic suspicion of malignancy (96%) is reassuring. “This, combined with benign histology on initial biopsies, could serve to identify which patients should return for repeat endoscopy.”
“We need to ensure that more biopsies are obtained during the index endoscopy from gastric ulcers as well as randomly and that, during follow-up endoscopy, biopsies are obtained from all [partially or fully] nonhealed ulcers,” added Dr. Lee. She suggested it could be helpful to develop an evidence-based, prospectively validated algorithm and/or identify risk factors that reliably help endoscopists decide who would benefit from repeat endoscopy, “especially since there is a relatively high rate of noncompliance with a low rate of malignancy.”
A primary limitation of the study included its retrospective nature; however, the authors pointed out that the study currently represents the largest multicenter, retrospective cohort analysis of endoscopic follow-up for gastric ulcers.
“Before any change can be recommended to current clinical practice, prospective and potentially randomized studies are required to validate our findings and elucidate any high-risk features associated with malignant gastric ulcer,” the investigators wrote. Doing so could lead to reductions in health care cost and patient burden.
Some of the study authors received funding from the National Health and Medical Research Council of Australia, but the remaining authors declared having nothing to disclose. Dr. DeSimone and Dr Lee reported having no relevant conflicts.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Supreme Court Case: Dobbs v Jackson Women’s Health Organization: What you need to know
This fall, the Supreme Court of the United States (SCOTUS) will announce when they will hear oral arguments for Dobbs v Jackson Women’s Health Organization. The court will examine a Mississippi law, known as the “Gestational Age Act,” originally passed in 2018, that sought to “limit abortions to fifteen weeks’ gestation except in a medical emergency or in cases of severe fetal abnormality.”1 This sets the stage for SCOTUS to make a major ruling on abortion, one which could affirm or upend landmark decisions and nearly 50 years of abortion legislative precedent. Additionally, SCOTUS’ recent decision to not intervene on Texas’ Senate Bill 8 (SB8), which essentially bans all abortions after 6 weeks’ gestational age, may foreshadow how this case will be decided. The current abortion restrictions in Texas and the implications of SB8 will be discussed in a forthcoming column.
SCOTUS and abortion rights
The decision to hear this case comes on the heels of another recent decision regarding a Louisiana law in June Medical Services v Russo. This case examined Louisiana Act 620, which would have required physicians to have hospital admitting privileges within 30 miles of where they provide abortion services.2 The law was deemed constitutionally invalid, with the majority noting the law would have drastically burdened a woman’s right to access abortion services. The Court ruled similarly in 2016 in Whole Women’s Health (WWH) v Hellerstedt, in which WWH challenged Texas House Bill 2, a nearly identical law requiring admitting privileges for abortion care providers. In both of these cases, SCOTUS pointed to precedent set by Southeastern Pennsylvania v Casey, which established that it is unconstitutional for a state to create an “undue burden” on a woman’s right to abortion prior to fetal viability.3 The precedent to this, Roe v Wade, and 5 decades of abortion legislation set may be upended by a SCOTUS decision this next term.
Dobbs v Jackson
On March 19, 2018, Mississippi enacted the “Gestational Age Act” into law. The newly enacted law would limit abortions to 15 weeks’ gestation except in a medical emergency or in cases of severe fetal anomalies. Jackson Women’s Health Organization, the only licensed abortion provider in the state, challenged the constitutionality of the law with legal support from Center for Reproductive Rights (CRR). The US District Court for the Southern District of Mississippi granted summary judgement in favor of the clinic and placed an injunction on the law’s enforcement. The state appealed to the Fifth Circuit Court of Appeals, which upheld the district court decision in a 3-0 decision in November 2019. Mississippi appealed to the Supreme Court, with their petition focusing on multiple questions from the appeals process. After repeatedly rescheduling the case, and multiple reviews in conference, SCOTUS agreed to hear the case. Most recently, the state has narrowed its argument, changing course, and attacking Roe v Wade directly. In a brief submitted in July 2021, the state argues the court should hold that all pre-viability prohibitions on elective abortions are constitutional.
Interestingly, during this time the Mississippi legislature also passed a law, House Bill 2116, also known as the “fetal heartbeat bill,” banning abortion with gestational ages after detection of a fetal heartbeat. This was also challenged, deemed unconstitutional, and affirmed on appeal by the Fifth US Circuit Court.
While recent challenges have focused on the “undue burden” state laws placed on those trying to access abortion care, this case will bring the issue of “viability” and gestational age limits to the forefront.4,5 In addition to Roe v Wade, the Court will have the opportunity to reexamine other relevant precedent, such as Southeastern Pennsylvania v Casey, in considering the most recent arguments of the state. In this most recent brief, the state argues that the Court should, “reject viability as a barrier to prohibiting elective abortions” and that a “viability rule has no constitutional basis.” The state goes on to argue the “Constitution does not protect a right to abortion or limit States’ authority to restrict it.”6 The language and tone in this brief are more direct and aggressive than the states’ petition submitted last June.
However, the composition of the Court is different than in the past. This case will be argued with Justice Amy Coney Barrett seated in place of Justice Ruth Bader Ginsburg, who was a strong advocate for women’s rights.7 She joins Justices Neil Gorsuch and Brett Kavanaugh, also appointed by President Donald Trump and widely viewed as conservative judges, tipping the scales to a more conservative Supreme Court. This case will also be argued in a polarized political environment.8,9 Given the conservative Supreme Court in the setting of an increasingly politically charged environment, reproductive right advocates are understandably worried that members of the anti-abortion movement view this as an opportunity to weaken or remove federal constitutional protections for abortion.
Continue to: Potential outcome of Dobbs v Jackson...
Potential outcome of Dobbs v Jackson
Should SCOTUS choose to rule in favor of Mississippi, it could severely weaken, or even overturn Roe v Wade. This would leave a legal path for states with pre-Roe abortion bans and currently unenforced post-Roe bans to take effect. These “trigger” laws are bans or severe restrictions on abortion providers and patients intended to take effect if Roe were to be overturned. Alternatively, the Court may overturn Southeastern Pennsylvania v Casey, but maintain Roe v Wade, essentially leaving the regulation of pre-viability abortion care to individual states. Currently 21 states have laws that would restrict the legal status of abortion.10 In addition, state legislatures are aggressively introducing abortion restrictions. As of June 2021, there have been 561 abortion restrictions, including 165 abortion bans, introduced across 47 states, putting 2021 on course to be the most devastating anti-abortion state legislative session in decades.11
The damage caused by such restriction on abortion care would be significant. It would block or push access out of reach for many. The negative effects of such legislative action would most heavily burden those already marginalized by systemic, structural inequalities including those of low socioeconomic status, people of color, young people, those in rural communities, and members of the LGBTQ community. The medical community has long recognized the harm caused by restricting access to abortion care. Restriction of access to safe abortion care paradoxically has been shown not to decrease the incidence of abortion, but rather increases the number of unsafe abortions.12 The American College of Obstetricians and Gynecologists (ACOG) acknowledge “individuals require access to safe, legal abortion” and that this represents “a necessary component for comprehensive health care.”13,14 They joined the American Medical Association and other professional groups in a 2019 amicus brief to SCOTUS opposing restrictions on abortion access.15 In addition, government laws restricting access to abortion care undermine the fundamental relationship between a person and their physician, limiting a physician’s obligation to honor patient autonomy and provide appropriate medical care.
By taking up the question whether all pre-viability bans on elective abortions violate the Constitution, SCOTUS is indicating a possible willingness to revisit the central holding of abortion jurisprudence. Their decision regarding this case will likely be the most significant ruling regarding the legal status of abortion care in decades, and will significantly affect the delivery of abortion care in the future.
Action items
- Reach out to your representatives to support the Women’s Health Protection Act, an initiative introduced to Congress to protect access to abortion care. If you reside in a state where your federal representatives support the Women’s Health Protection Act, reach out to friends and colleagues in states without supportive elected officials and ask them to call their representatives and ask them to support the bill.
- Get involved with local grassroots groups fighting to protect abortion access.
- Continue to speak out against laws and policies designed to limit access to safe abortion care.
- Connect with your local ACOG chapter for more ways to become involved.
- As always, make sure you are registered to vote, and exercise your right whenever you can.
- HB1510 (As Introduced) - 2018 Regular Session. http://billstatus.ls.state.ms.us/documents/2018/html/HB/1500-1599/HB1510IN.htm Accessed August 13, 2021.
- HB338. Louisiana State Legislature. 2014. http://www.legis.la.gov/legis/BillInfo.aspx?s=14RS&b=ACT620&sbi=y. Accessed August 13, 2021.
- Planned Parenthood of Southeastern Pennsylvania v. Casey. Cornell Law School Legal Information Institute. https://www.law.cornell.edu/supremecourt/text/505/833. Accessed August 13, 2021.
- 15-274 Whole Woman’s Health v. Hellerstedt (06/27/2016). Published online 2016:107.
- 18-1323 June Medical Services L. L. C. v. Russo (06/29/2020). Published online 2020:138.
- 19-1392 Dobbs v. Jackson Women’s Health Organization (07/22/2021). Published online 2021.
- What Ruth Bader Ginsburg said about abortion and Roe v. Wade. Time. August 2, 2018. https://time.com/5354490/ruth-bader-ginsburg-roe-v-wade/. Accessed August 13, 2021.
- Montanaro D. Poll: majority want to keep abortion legal, but they also want restrictions. NPR. June 7, 2019. https://www.npr.org/2019/06/07/730183531/poll-majority-want-to-keep-abortion-legal-but-they-also-want-restrictions. Accessed August 13, 2021.
- Abortion support remains steady despite growing partisan divide, survey finds. Washington Post. August 13, 2019. https://www.washingtonpost.com/health/2019/08/13/one-largest-ever-abortion-surveys-shows-growing-partisan-divide/. Accessed August 13, 2021.
- Abortion policy in the absence of Roe. Guttmacher Institute. September 1, 2021. https://www.guttmacher.org/state-policy/explore/abortion-policy-absence-roe#. Accessed September 8, 2021.
- 2021 is on track to become the most devastating antiabortion state legislative session in decades. Guttmacher Institute. Published April 30, 2021. Updated June 14, 2021. https://www.guttmacher.org/article/2021/04/2021-track-become-most-devastating-antiabortion-state-legislative-session-decades. Accessed August 13, 2021.
- Facts and consequences: legality, incidence and safety of abortion worldwide. Guttmacher Institute. November 20, 2009. https://www.guttmacher.org/gpr/2009/11/facts-and-consequences-legality-incidence-and-safety-abortion-worldwide. Accessed August 13, 2021.
- Increasing access to abortion. https://www.acog.org/en/clinical/clinical-guidance/committee-opinion/articles/2020/12/increasing-access-to-abortion. Accessed August 13, 2021.
- ACOG statement on Dobbs vs. Jackson Women’s Health. May 17, 2021. https://www.acog.org/en/news/news-releases/2021/05/acog-statement-dobbs-vs-jackson-womens-health. Accessed August 13, 2021.
- Perryman SL, Parker KA, Hickman SA. Brief of amici curiae American College of Obstetricians and Gynecologists, American Medical Associations, American Academy of Family Physicians, American Academy of Nursing, American Academy of Pediatrics, et al. In support of June Medical Services, LLC, et al. https://www.supremecourt.gov/
DocketPDF/18/18-1323/124091/ . Accessed August 13, 2021.20191202145531124_18-1323% 2018-1460%20tsac%20American% 20College%20of% 20Obstetricians%20and% 20Gynecologists%20et%20al.pdf
This fall, the Supreme Court of the United States (SCOTUS) will announce when they will hear oral arguments for Dobbs v Jackson Women’s Health Organization. The court will examine a Mississippi law, known as the “Gestational Age Act,” originally passed in 2018, that sought to “limit abortions to fifteen weeks’ gestation except in a medical emergency or in cases of severe fetal abnormality.”1 This sets the stage for SCOTUS to make a major ruling on abortion, one which could affirm or upend landmark decisions and nearly 50 years of abortion legislative precedent. Additionally, SCOTUS’ recent decision to not intervene on Texas’ Senate Bill 8 (SB8), which essentially bans all abortions after 6 weeks’ gestational age, may foreshadow how this case will be decided. The current abortion restrictions in Texas and the implications of SB8 will be discussed in a forthcoming column.
SCOTUS and abortion rights
The decision to hear this case comes on the heels of another recent decision regarding a Louisiana law in June Medical Services v Russo. This case examined Louisiana Act 620, which would have required physicians to have hospital admitting privileges within 30 miles of where they provide abortion services.2 The law was deemed constitutionally invalid, with the majority noting the law would have drastically burdened a woman’s right to access abortion services. The Court ruled similarly in 2016 in Whole Women’s Health (WWH) v Hellerstedt, in which WWH challenged Texas House Bill 2, a nearly identical law requiring admitting privileges for abortion care providers. In both of these cases, SCOTUS pointed to precedent set by Southeastern Pennsylvania v Casey, which established that it is unconstitutional for a state to create an “undue burden” on a woman’s right to abortion prior to fetal viability.3 The precedent to this, Roe v Wade, and 5 decades of abortion legislation set may be upended by a SCOTUS decision this next term.
Dobbs v Jackson
On March 19, 2018, Mississippi enacted the “Gestational Age Act” into law. The newly enacted law would limit abortions to 15 weeks’ gestation except in a medical emergency or in cases of severe fetal anomalies. Jackson Women’s Health Organization, the only licensed abortion provider in the state, challenged the constitutionality of the law with legal support from Center for Reproductive Rights (CRR). The US District Court for the Southern District of Mississippi granted summary judgement in favor of the clinic and placed an injunction on the law’s enforcement. The state appealed to the Fifth Circuit Court of Appeals, which upheld the district court decision in a 3-0 decision in November 2019. Mississippi appealed to the Supreme Court, with their petition focusing on multiple questions from the appeals process. After repeatedly rescheduling the case, and multiple reviews in conference, SCOTUS agreed to hear the case. Most recently, the state has narrowed its argument, changing course, and attacking Roe v Wade directly. In a brief submitted in July 2021, the state argues the court should hold that all pre-viability prohibitions on elective abortions are constitutional.
Interestingly, during this time the Mississippi legislature also passed a law, House Bill 2116, also known as the “fetal heartbeat bill,” banning abortion with gestational ages after detection of a fetal heartbeat. This was also challenged, deemed unconstitutional, and affirmed on appeal by the Fifth US Circuit Court.
While recent challenges have focused on the “undue burden” state laws placed on those trying to access abortion care, this case will bring the issue of “viability” and gestational age limits to the forefront.4,5 In addition to Roe v Wade, the Court will have the opportunity to reexamine other relevant precedent, such as Southeastern Pennsylvania v Casey, in considering the most recent arguments of the state. In this most recent brief, the state argues that the Court should, “reject viability as a barrier to prohibiting elective abortions” and that a “viability rule has no constitutional basis.” The state goes on to argue the “Constitution does not protect a right to abortion or limit States’ authority to restrict it.”6 The language and tone in this brief are more direct and aggressive than the states’ petition submitted last June.
However, the composition of the Court is different than in the past. This case will be argued with Justice Amy Coney Barrett seated in place of Justice Ruth Bader Ginsburg, who was a strong advocate for women’s rights.7 She joins Justices Neil Gorsuch and Brett Kavanaugh, also appointed by President Donald Trump and widely viewed as conservative judges, tipping the scales to a more conservative Supreme Court. This case will also be argued in a polarized political environment.8,9 Given the conservative Supreme Court in the setting of an increasingly politically charged environment, reproductive right advocates are understandably worried that members of the anti-abortion movement view this as an opportunity to weaken or remove federal constitutional protections for abortion.
Continue to: Potential outcome of Dobbs v Jackson...
Potential outcome of Dobbs v Jackson
Should SCOTUS choose to rule in favor of Mississippi, it could severely weaken, or even overturn Roe v Wade. This would leave a legal path for states with pre-Roe abortion bans and currently unenforced post-Roe bans to take effect. These “trigger” laws are bans or severe restrictions on abortion providers and patients intended to take effect if Roe were to be overturned. Alternatively, the Court may overturn Southeastern Pennsylvania v Casey, but maintain Roe v Wade, essentially leaving the regulation of pre-viability abortion care to individual states. Currently 21 states have laws that would restrict the legal status of abortion.10 In addition, state legislatures are aggressively introducing abortion restrictions. As of June 2021, there have been 561 abortion restrictions, including 165 abortion bans, introduced across 47 states, putting 2021 on course to be the most devastating anti-abortion state legislative session in decades.11
The damage caused by such restriction on abortion care would be significant. It would block or push access out of reach for many. The negative effects of such legislative action would most heavily burden those already marginalized by systemic, structural inequalities including those of low socioeconomic status, people of color, young people, those in rural communities, and members of the LGBTQ community. The medical community has long recognized the harm caused by restricting access to abortion care. Restriction of access to safe abortion care paradoxically has been shown not to decrease the incidence of abortion, but rather increases the number of unsafe abortions.12 The American College of Obstetricians and Gynecologists (ACOG) acknowledge “individuals require access to safe, legal abortion” and that this represents “a necessary component for comprehensive health care.”13,14 They joined the American Medical Association and other professional groups in a 2019 amicus brief to SCOTUS opposing restrictions on abortion access.15 In addition, government laws restricting access to abortion care undermine the fundamental relationship between a person and their physician, limiting a physician’s obligation to honor patient autonomy and provide appropriate medical care.
By taking up the question whether all pre-viability bans on elective abortions violate the Constitution, SCOTUS is indicating a possible willingness to revisit the central holding of abortion jurisprudence. Their decision regarding this case will likely be the most significant ruling regarding the legal status of abortion care in decades, and will significantly affect the delivery of abortion care in the future.
Action items
- Reach out to your representatives to support the Women’s Health Protection Act, an initiative introduced to Congress to protect access to abortion care. If you reside in a state where your federal representatives support the Women’s Health Protection Act, reach out to friends and colleagues in states without supportive elected officials and ask them to call their representatives and ask them to support the bill.
- Get involved with local grassroots groups fighting to protect abortion access.
- Continue to speak out against laws and policies designed to limit access to safe abortion care.
- Connect with your local ACOG chapter for more ways to become involved.
- As always, make sure you are registered to vote, and exercise your right whenever you can.
This fall, the Supreme Court of the United States (SCOTUS) will announce when they will hear oral arguments for Dobbs v Jackson Women’s Health Organization. The court will examine a Mississippi law, known as the “Gestational Age Act,” originally passed in 2018, that sought to “limit abortions to fifteen weeks’ gestation except in a medical emergency or in cases of severe fetal abnormality.”1 This sets the stage for SCOTUS to make a major ruling on abortion, one which could affirm or upend landmark decisions and nearly 50 years of abortion legislative precedent. Additionally, SCOTUS’ recent decision to not intervene on Texas’ Senate Bill 8 (SB8), which essentially bans all abortions after 6 weeks’ gestational age, may foreshadow how this case will be decided. The current abortion restrictions in Texas and the implications of SB8 will be discussed in a forthcoming column.
SCOTUS and abortion rights
The decision to hear this case comes on the heels of another recent decision regarding a Louisiana law in June Medical Services v Russo. This case examined Louisiana Act 620, which would have required physicians to have hospital admitting privileges within 30 miles of where they provide abortion services.2 The law was deemed constitutionally invalid, with the majority noting the law would have drastically burdened a woman’s right to access abortion services. The Court ruled similarly in 2016 in Whole Women’s Health (WWH) v Hellerstedt, in which WWH challenged Texas House Bill 2, a nearly identical law requiring admitting privileges for abortion care providers. In both of these cases, SCOTUS pointed to precedent set by Southeastern Pennsylvania v Casey, which established that it is unconstitutional for a state to create an “undue burden” on a woman’s right to abortion prior to fetal viability.3 The precedent to this, Roe v Wade, and 5 decades of abortion legislation set may be upended by a SCOTUS decision this next term.
Dobbs v Jackson
On March 19, 2018, Mississippi enacted the “Gestational Age Act” into law. The newly enacted law would limit abortions to 15 weeks’ gestation except in a medical emergency or in cases of severe fetal anomalies. Jackson Women’s Health Organization, the only licensed abortion provider in the state, challenged the constitutionality of the law with legal support from Center for Reproductive Rights (CRR). The US District Court for the Southern District of Mississippi granted summary judgement in favor of the clinic and placed an injunction on the law’s enforcement. The state appealed to the Fifth Circuit Court of Appeals, which upheld the district court decision in a 3-0 decision in November 2019. Mississippi appealed to the Supreme Court, with their petition focusing on multiple questions from the appeals process. After repeatedly rescheduling the case, and multiple reviews in conference, SCOTUS agreed to hear the case. Most recently, the state has narrowed its argument, changing course, and attacking Roe v Wade directly. In a brief submitted in July 2021, the state argues the court should hold that all pre-viability prohibitions on elective abortions are constitutional.
Interestingly, during this time the Mississippi legislature also passed a law, House Bill 2116, also known as the “fetal heartbeat bill,” banning abortion with gestational ages after detection of a fetal heartbeat. This was also challenged, deemed unconstitutional, and affirmed on appeal by the Fifth US Circuit Court.
While recent challenges have focused on the “undue burden” state laws placed on those trying to access abortion care, this case will bring the issue of “viability” and gestational age limits to the forefront.4,5 In addition to Roe v Wade, the Court will have the opportunity to reexamine other relevant precedent, such as Southeastern Pennsylvania v Casey, in considering the most recent arguments of the state. In this most recent brief, the state argues that the Court should, “reject viability as a barrier to prohibiting elective abortions” and that a “viability rule has no constitutional basis.” The state goes on to argue the “Constitution does not protect a right to abortion or limit States’ authority to restrict it.”6 The language and tone in this brief are more direct and aggressive than the states’ petition submitted last June.
However, the composition of the Court is different than in the past. This case will be argued with Justice Amy Coney Barrett seated in place of Justice Ruth Bader Ginsburg, who was a strong advocate for women’s rights.7 She joins Justices Neil Gorsuch and Brett Kavanaugh, also appointed by President Donald Trump and widely viewed as conservative judges, tipping the scales to a more conservative Supreme Court. This case will also be argued in a polarized political environment.8,9 Given the conservative Supreme Court in the setting of an increasingly politically charged environment, reproductive right advocates are understandably worried that members of the anti-abortion movement view this as an opportunity to weaken or remove federal constitutional protections for abortion.
Continue to: Potential outcome of Dobbs v Jackson...
Potential outcome of Dobbs v Jackson
Should SCOTUS choose to rule in favor of Mississippi, it could severely weaken, or even overturn Roe v Wade. This would leave a legal path for states with pre-Roe abortion bans and currently unenforced post-Roe bans to take effect. These “trigger” laws are bans or severe restrictions on abortion providers and patients intended to take effect if Roe were to be overturned. Alternatively, the Court may overturn Southeastern Pennsylvania v Casey, but maintain Roe v Wade, essentially leaving the regulation of pre-viability abortion care to individual states. Currently 21 states have laws that would restrict the legal status of abortion.10 In addition, state legislatures are aggressively introducing abortion restrictions. As of June 2021, there have been 561 abortion restrictions, including 165 abortion bans, introduced across 47 states, putting 2021 on course to be the most devastating anti-abortion state legislative session in decades.11
The damage caused by such restriction on abortion care would be significant. It would block or push access out of reach for many. The negative effects of such legislative action would most heavily burden those already marginalized by systemic, structural inequalities including those of low socioeconomic status, people of color, young people, those in rural communities, and members of the LGBTQ community. The medical community has long recognized the harm caused by restricting access to abortion care. Restriction of access to safe abortion care paradoxically has been shown not to decrease the incidence of abortion, but rather increases the number of unsafe abortions.12 The American College of Obstetricians and Gynecologists (ACOG) acknowledge “individuals require access to safe, legal abortion” and that this represents “a necessary component for comprehensive health care.”13,14 They joined the American Medical Association and other professional groups in a 2019 amicus brief to SCOTUS opposing restrictions on abortion access.15 In addition, government laws restricting access to abortion care undermine the fundamental relationship between a person and their physician, limiting a physician’s obligation to honor patient autonomy and provide appropriate medical care.
By taking up the question whether all pre-viability bans on elective abortions violate the Constitution, SCOTUS is indicating a possible willingness to revisit the central holding of abortion jurisprudence. Their decision regarding this case will likely be the most significant ruling regarding the legal status of abortion care in decades, and will significantly affect the delivery of abortion care in the future.
Action items
- Reach out to your representatives to support the Women’s Health Protection Act, an initiative introduced to Congress to protect access to abortion care. If you reside in a state where your federal representatives support the Women’s Health Protection Act, reach out to friends and colleagues in states without supportive elected officials and ask them to call their representatives and ask them to support the bill.
- Get involved with local grassroots groups fighting to protect abortion access.
- Continue to speak out against laws and policies designed to limit access to safe abortion care.
- Connect with your local ACOG chapter for more ways to become involved.
- As always, make sure you are registered to vote, and exercise your right whenever you can.
- HB1510 (As Introduced) - 2018 Regular Session. http://billstatus.ls.state.ms.us/documents/2018/html/HB/1500-1599/HB1510IN.htm Accessed August 13, 2021.
- HB338. Louisiana State Legislature. 2014. http://www.legis.la.gov/legis/BillInfo.aspx?s=14RS&b=ACT620&sbi=y. Accessed August 13, 2021.
- Planned Parenthood of Southeastern Pennsylvania v. Casey. Cornell Law School Legal Information Institute. https://www.law.cornell.edu/supremecourt/text/505/833. Accessed August 13, 2021.
- 15-274 Whole Woman’s Health v. Hellerstedt (06/27/2016). Published online 2016:107.
- 18-1323 June Medical Services L. L. C. v. Russo (06/29/2020). Published online 2020:138.
- 19-1392 Dobbs v. Jackson Women’s Health Organization (07/22/2021). Published online 2021.
- What Ruth Bader Ginsburg said about abortion and Roe v. Wade. Time. August 2, 2018. https://time.com/5354490/ruth-bader-ginsburg-roe-v-wade/. Accessed August 13, 2021.
- Montanaro D. Poll: majority want to keep abortion legal, but they also want restrictions. NPR. June 7, 2019. https://www.npr.org/2019/06/07/730183531/poll-majority-want-to-keep-abortion-legal-but-they-also-want-restrictions. Accessed August 13, 2021.
- Abortion support remains steady despite growing partisan divide, survey finds. Washington Post. August 13, 2019. https://www.washingtonpost.com/health/2019/08/13/one-largest-ever-abortion-surveys-shows-growing-partisan-divide/. Accessed August 13, 2021.
- Abortion policy in the absence of Roe. Guttmacher Institute. September 1, 2021. https://www.guttmacher.org/state-policy/explore/abortion-policy-absence-roe#. Accessed September 8, 2021.
- 2021 is on track to become the most devastating antiabortion state legislative session in decades. Guttmacher Institute. Published April 30, 2021. Updated June 14, 2021. https://www.guttmacher.org/article/2021/04/2021-track-become-most-devastating-antiabortion-state-legislative-session-decades. Accessed August 13, 2021.
- Facts and consequences: legality, incidence and safety of abortion worldwide. Guttmacher Institute. November 20, 2009. https://www.guttmacher.org/gpr/2009/11/facts-and-consequences-legality-incidence-and-safety-abortion-worldwide. Accessed August 13, 2021.
- Increasing access to abortion. https://www.acog.org/en/clinical/clinical-guidance/committee-opinion/articles/2020/12/increasing-access-to-abortion. Accessed August 13, 2021.
- ACOG statement on Dobbs vs. Jackson Women’s Health. May 17, 2021. https://www.acog.org/en/news/news-releases/2021/05/acog-statement-dobbs-vs-jackson-womens-health. Accessed August 13, 2021.
- Perryman SL, Parker KA, Hickman SA. Brief of amici curiae American College of Obstetricians and Gynecologists, American Medical Associations, American Academy of Family Physicians, American Academy of Nursing, American Academy of Pediatrics, et al. In support of June Medical Services, LLC, et al. https://www.supremecourt.gov/
DocketPDF/18/18-1323/124091/ . Accessed August 13, 2021.20191202145531124_18-1323% 2018-1460%20tsac%20American% 20College%20of% 20Obstetricians%20and% 20Gynecologists%20et%20al.pdf
- HB1510 (As Introduced) - 2018 Regular Session. http://billstatus.ls.state.ms.us/documents/2018/html/HB/1500-1599/HB1510IN.htm Accessed August 13, 2021.
- HB338. Louisiana State Legislature. 2014. http://www.legis.la.gov/legis/BillInfo.aspx?s=14RS&b=ACT620&sbi=y. Accessed August 13, 2021.
- Planned Parenthood of Southeastern Pennsylvania v. Casey. Cornell Law School Legal Information Institute. https://www.law.cornell.edu/supremecourt/text/505/833. Accessed August 13, 2021.
- 15-274 Whole Woman’s Health v. Hellerstedt (06/27/2016). Published online 2016:107.
- 18-1323 June Medical Services L. L. C. v. Russo (06/29/2020). Published online 2020:138.
- 19-1392 Dobbs v. Jackson Women’s Health Organization (07/22/2021). Published online 2021.
- What Ruth Bader Ginsburg said about abortion and Roe v. Wade. Time. August 2, 2018. https://time.com/5354490/ruth-bader-ginsburg-roe-v-wade/. Accessed August 13, 2021.
- Montanaro D. Poll: majority want to keep abortion legal, but they also want restrictions. NPR. June 7, 2019. https://www.npr.org/2019/06/07/730183531/poll-majority-want-to-keep-abortion-legal-but-they-also-want-restrictions. Accessed August 13, 2021.
- Abortion support remains steady despite growing partisan divide, survey finds. Washington Post. August 13, 2019. https://www.washingtonpost.com/health/2019/08/13/one-largest-ever-abortion-surveys-shows-growing-partisan-divide/. Accessed August 13, 2021.
- Abortion policy in the absence of Roe. Guttmacher Institute. September 1, 2021. https://www.guttmacher.org/state-policy/explore/abortion-policy-absence-roe#. Accessed September 8, 2021.
- 2021 is on track to become the most devastating antiabortion state legislative session in decades. Guttmacher Institute. Published April 30, 2021. Updated June 14, 2021. https://www.guttmacher.org/article/2021/04/2021-track-become-most-devastating-antiabortion-state-legislative-session-decades. Accessed August 13, 2021.
- Facts and consequences: legality, incidence and safety of abortion worldwide. Guttmacher Institute. November 20, 2009. https://www.guttmacher.org/gpr/2009/11/facts-and-consequences-legality-incidence-and-safety-abortion-worldwide. Accessed August 13, 2021.
- Increasing access to abortion. https://www.acog.org/en/clinical/clinical-guidance/committee-opinion/articles/2020/12/increasing-access-to-abortion. Accessed August 13, 2021.
- ACOG statement on Dobbs vs. Jackson Women’s Health. May 17, 2021. https://www.acog.org/en/news/news-releases/2021/05/acog-statement-dobbs-vs-jackson-womens-health. Accessed August 13, 2021.
- Perryman SL, Parker KA, Hickman SA. Brief of amici curiae American College of Obstetricians and Gynecologists, American Medical Associations, American Academy of Family Physicians, American Academy of Nursing, American Academy of Pediatrics, et al. In support of June Medical Services, LLC, et al. https://www.supremecourt.gov/
DocketPDF/18/18-1323/124091/ . Accessed August 13, 2021.20191202145531124_18-1323% 2018-1460%20tsac%20American% 20College%20of% 20Obstetricians%20and% 20Gynecologists%20et%20al.pdf
Mediterranean diet tied to less severe erectile dysfunction
In an observational study of 250 middle-aged men with hypertension and erectile dysfunction, those whose eating patterns more closely matched a Mediterranean diet had significantly higher testosterone levels, better exercise capacity, and better erectile performance than their peers.
In addition, more closely following a Mediterranean diet – which emphasizes eating fruit, vegetables, whole grains, and olive oil, with modest consumption of dairy products and limited red meat – was associated with better coronary blood flow and less arterial stiffness, all after adjusting for age, body mass index, type 2 diabetes, statin use, and smoking.
Athanasios Angelis, MD, First Cardiology Clinic, Hippokration Hospital, School of Medicine, University of Athens, presented the study at the annual congress of the European Society of Cardiology.
“While we did not examine mechanisms,” Dr. Angelis said in a press release from the ESC, “it seems plausible that this dietary pattern may improve fitness and erectile performance by enhancing function of the blood vessels and limiting the fall in testosterone that occurs in midlife.”
“The findings suggest that the Mediterranean diet could play a role in maintaining several parameters of vascular health and quality of life and in middle-aged men with hypertension and erectile dysfunction,” he concluded.
“A Mediterranean diet may help erectile dysfunction by improving endothelial physiology,” Dr. Angelis said in an interview. “We suggest the Mediterranean diet as a basic parameter of hypertension and erectile dysfunction treatment. We advise all our patients to be careful regarding salt consumption and to try to exercise regularly.”
“Depending on the severity of the erectile dysfunction, we may suggest only lifestyle changes (e.g., quit smoking), at least for the beginning, or combination with medication,” consisting of phosphodiesterase type 5 (PDE5) inhibitors such as Viagra.
A ‘first-choice’ diet for men with ED, low T, high CVD risk?
This research “adds to the growing evidence that a Mediterranean diet is protective against erectile dysfunction,” said Joseph Whittaker, MSc, a clinical nutritionist from the University of Worcester (England) and coauthor of a related meta-analysis about dietary fat and testosterone.
This way of eating “also improves cardiovascular health, so it could become a low-risk, first choice treatment for these three pathologies (low testosterone, erectile dysfunction, increased risk of CVD), which so commonly coexist,” he wrote in an email.
“However, most of the research to date is observational,” he cautioned, which often has a “healthy user bias,” that is, the men eating a Mediterranean diet are probably health-conscious individuals, with other healthy habits such as exercise, good sleep, low stress, etc. “So, was it the diet, the healthy habits, or both?”
Randomized studies are needed to replicate the positive results of observational studies like this one, Mr. Whittaker added. In the meantime, “a Mediterranean diet will probably improve your health anyway,” he noted, “so trying it for the purposes of erectile function (before starting drugs) is a viable option.”
Previous research has shown that dietary fat and olive oil may boost testosterone levels, Mr. Whittaker noted, and nuts have also been shown to improve erectile function.
“So, the increase in healthy fats – mono- and polyunsaturated fatty acids (MUFAs and PUFAs, respectively) – on the Mediterranean diet is probably responsible for these benefits,” he speculated.
Middle-aged hypertensive men with ED
Men with hypertension are twice as likely to have erectile dysfunction as their peers with normal blood pressure, according to background information in the ESC press release.
Erectile dysfunction is thought to be a disorder of the small arteries, which lose their ability to dilate and increase blood flow. Declining testosterone levels in middle age also contribute to weakened erectile performance.
Physical fitness is linked with longer life in men with hypertension, and the Mediterranean diet is associated with lower blood pressure and fewer heart attacks and strokes in individuals at high cardiovascular risk.
Therefore, Dr. Angelis and colleagues aimed to see if greater adherence to a Mediterranean diet was associated with better exercise capacity, testosterone levels, coronary flow reserve, and erectile performance in middle-aged hypertensive men with erectile dysfunction.
Participants were a mean age of 56. They had a treadmill test to determine their exercise capacity, expressed as metabolic equivalent of tasks (METs), and a blood test to determine testosterone levels.
They replied to two questionnaires: a food questionnaire to determine a Mediterranean Diet score (range, 0-55, where higher scores indicate greater adherence to a Mediterranean diet) and a Sexual Health Inventory for Men (SHIM) questionnaire (score range, 0-25, where higher scores indicate better erectile performance).
Researchers used echocardiography to determine participants’ coronary flow reserve, a measure of the cardiovascular system’s ability to increase blood flow when needed. They used a SphygmoCor device to determine participants’ augmentation index and central pulse pressure, measures of arterial stiffness.
The men with a higher Mediterranean diet score (>29) had better erectile performance (SHIM scores > 14), as well as higher testosterone levels, higher coronary flow reserve, and less arterial stiffness than the other men.
The fitter men with greater exercise capacity (>10 METs) were more likely to adhere to a Mediterranean diet (scores > 25), and they also had better erectile performance (SHIM scores > 12), higher testosterone levels, greater coronary flow reserve, and less arterial stiffness than the other men.
The study did not receive any funding. The study authors and Mr. Whittaker have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an observational study of 250 middle-aged men with hypertension and erectile dysfunction, those whose eating patterns more closely matched a Mediterranean diet had significantly higher testosterone levels, better exercise capacity, and better erectile performance than their peers.
In addition, more closely following a Mediterranean diet – which emphasizes eating fruit, vegetables, whole grains, and olive oil, with modest consumption of dairy products and limited red meat – was associated with better coronary blood flow and less arterial stiffness, all after adjusting for age, body mass index, type 2 diabetes, statin use, and smoking.
Athanasios Angelis, MD, First Cardiology Clinic, Hippokration Hospital, School of Medicine, University of Athens, presented the study at the annual congress of the European Society of Cardiology.
“While we did not examine mechanisms,” Dr. Angelis said in a press release from the ESC, “it seems plausible that this dietary pattern may improve fitness and erectile performance by enhancing function of the blood vessels and limiting the fall in testosterone that occurs in midlife.”
“The findings suggest that the Mediterranean diet could play a role in maintaining several parameters of vascular health and quality of life and in middle-aged men with hypertension and erectile dysfunction,” he concluded.
“A Mediterranean diet may help erectile dysfunction by improving endothelial physiology,” Dr. Angelis said in an interview. “We suggest the Mediterranean diet as a basic parameter of hypertension and erectile dysfunction treatment. We advise all our patients to be careful regarding salt consumption and to try to exercise regularly.”
“Depending on the severity of the erectile dysfunction, we may suggest only lifestyle changes (e.g., quit smoking), at least for the beginning, or combination with medication,” consisting of phosphodiesterase type 5 (PDE5) inhibitors such as Viagra.
A ‘first-choice’ diet for men with ED, low T, high CVD risk?
This research “adds to the growing evidence that a Mediterranean diet is protective against erectile dysfunction,” said Joseph Whittaker, MSc, a clinical nutritionist from the University of Worcester (England) and coauthor of a related meta-analysis about dietary fat and testosterone.
This way of eating “also improves cardiovascular health, so it could become a low-risk, first choice treatment for these three pathologies (low testosterone, erectile dysfunction, increased risk of CVD), which so commonly coexist,” he wrote in an email.
“However, most of the research to date is observational,” he cautioned, which often has a “healthy user bias,” that is, the men eating a Mediterranean diet are probably health-conscious individuals, with other healthy habits such as exercise, good sleep, low stress, etc. “So, was it the diet, the healthy habits, or both?”
Randomized studies are needed to replicate the positive results of observational studies like this one, Mr. Whittaker added. In the meantime, “a Mediterranean diet will probably improve your health anyway,” he noted, “so trying it for the purposes of erectile function (before starting drugs) is a viable option.”
Previous research has shown that dietary fat and olive oil may boost testosterone levels, Mr. Whittaker noted, and nuts have also been shown to improve erectile function.
“So, the increase in healthy fats – mono- and polyunsaturated fatty acids (MUFAs and PUFAs, respectively) – on the Mediterranean diet is probably responsible for these benefits,” he speculated.
Middle-aged hypertensive men with ED
Men with hypertension are twice as likely to have erectile dysfunction as their peers with normal blood pressure, according to background information in the ESC press release.
Erectile dysfunction is thought to be a disorder of the small arteries, which lose their ability to dilate and increase blood flow. Declining testosterone levels in middle age also contribute to weakened erectile performance.
Physical fitness is linked with longer life in men with hypertension, and the Mediterranean diet is associated with lower blood pressure and fewer heart attacks and strokes in individuals at high cardiovascular risk.
Therefore, Dr. Angelis and colleagues aimed to see if greater adherence to a Mediterranean diet was associated with better exercise capacity, testosterone levels, coronary flow reserve, and erectile performance in middle-aged hypertensive men with erectile dysfunction.
Participants were a mean age of 56. They had a treadmill test to determine their exercise capacity, expressed as metabolic equivalent of tasks (METs), and a blood test to determine testosterone levels.
They replied to two questionnaires: a food questionnaire to determine a Mediterranean Diet score (range, 0-55, where higher scores indicate greater adherence to a Mediterranean diet) and a Sexual Health Inventory for Men (SHIM) questionnaire (score range, 0-25, where higher scores indicate better erectile performance).
Researchers used echocardiography to determine participants’ coronary flow reserve, a measure of the cardiovascular system’s ability to increase blood flow when needed. They used a SphygmoCor device to determine participants’ augmentation index and central pulse pressure, measures of arterial stiffness.
The men with a higher Mediterranean diet score (>29) had better erectile performance (SHIM scores > 14), as well as higher testosterone levels, higher coronary flow reserve, and less arterial stiffness than the other men.
The fitter men with greater exercise capacity (>10 METs) were more likely to adhere to a Mediterranean diet (scores > 25), and they also had better erectile performance (SHIM scores > 12), higher testosterone levels, greater coronary flow reserve, and less arterial stiffness than the other men.
The study did not receive any funding. The study authors and Mr. Whittaker have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an observational study of 250 middle-aged men with hypertension and erectile dysfunction, those whose eating patterns more closely matched a Mediterranean diet had significantly higher testosterone levels, better exercise capacity, and better erectile performance than their peers.
In addition, more closely following a Mediterranean diet – which emphasizes eating fruit, vegetables, whole grains, and olive oil, with modest consumption of dairy products and limited red meat – was associated with better coronary blood flow and less arterial stiffness, all after adjusting for age, body mass index, type 2 diabetes, statin use, and smoking.
Athanasios Angelis, MD, First Cardiology Clinic, Hippokration Hospital, School of Medicine, University of Athens, presented the study at the annual congress of the European Society of Cardiology.
“While we did not examine mechanisms,” Dr. Angelis said in a press release from the ESC, “it seems plausible that this dietary pattern may improve fitness and erectile performance by enhancing function of the blood vessels and limiting the fall in testosterone that occurs in midlife.”
“The findings suggest that the Mediterranean diet could play a role in maintaining several parameters of vascular health and quality of life and in middle-aged men with hypertension and erectile dysfunction,” he concluded.
“A Mediterranean diet may help erectile dysfunction by improving endothelial physiology,” Dr. Angelis said in an interview. “We suggest the Mediterranean diet as a basic parameter of hypertension and erectile dysfunction treatment. We advise all our patients to be careful regarding salt consumption and to try to exercise regularly.”
“Depending on the severity of the erectile dysfunction, we may suggest only lifestyle changes (e.g., quit smoking), at least for the beginning, or combination with medication,” consisting of phosphodiesterase type 5 (PDE5) inhibitors such as Viagra.
A ‘first-choice’ diet for men with ED, low T, high CVD risk?
This research “adds to the growing evidence that a Mediterranean diet is protective against erectile dysfunction,” said Joseph Whittaker, MSc, a clinical nutritionist from the University of Worcester (England) and coauthor of a related meta-analysis about dietary fat and testosterone.
This way of eating “also improves cardiovascular health, so it could become a low-risk, first choice treatment for these three pathologies (low testosterone, erectile dysfunction, increased risk of CVD), which so commonly coexist,” he wrote in an email.
“However, most of the research to date is observational,” he cautioned, which often has a “healthy user bias,” that is, the men eating a Mediterranean diet are probably health-conscious individuals, with other healthy habits such as exercise, good sleep, low stress, etc. “So, was it the diet, the healthy habits, or both?”
Randomized studies are needed to replicate the positive results of observational studies like this one, Mr. Whittaker added. In the meantime, “a Mediterranean diet will probably improve your health anyway,” he noted, “so trying it for the purposes of erectile function (before starting drugs) is a viable option.”
Previous research has shown that dietary fat and olive oil may boost testosterone levels, Mr. Whittaker noted, and nuts have also been shown to improve erectile function.
“So, the increase in healthy fats – mono- and polyunsaturated fatty acids (MUFAs and PUFAs, respectively) – on the Mediterranean diet is probably responsible for these benefits,” he speculated.
Middle-aged hypertensive men with ED
Men with hypertension are twice as likely to have erectile dysfunction as their peers with normal blood pressure, according to background information in the ESC press release.
Erectile dysfunction is thought to be a disorder of the small arteries, which lose their ability to dilate and increase blood flow. Declining testosterone levels in middle age also contribute to weakened erectile performance.
Physical fitness is linked with longer life in men with hypertension, and the Mediterranean diet is associated with lower blood pressure and fewer heart attacks and strokes in individuals at high cardiovascular risk.
Therefore, Dr. Angelis and colleagues aimed to see if greater adherence to a Mediterranean diet was associated with better exercise capacity, testosterone levels, coronary flow reserve, and erectile performance in middle-aged hypertensive men with erectile dysfunction.
Participants were a mean age of 56. They had a treadmill test to determine their exercise capacity, expressed as metabolic equivalent of tasks (METs), and a blood test to determine testosterone levels.
They replied to two questionnaires: a food questionnaire to determine a Mediterranean Diet score (range, 0-55, where higher scores indicate greater adherence to a Mediterranean diet) and a Sexual Health Inventory for Men (SHIM) questionnaire (score range, 0-25, where higher scores indicate better erectile performance).
Researchers used echocardiography to determine participants’ coronary flow reserve, a measure of the cardiovascular system’s ability to increase blood flow when needed. They used a SphygmoCor device to determine participants’ augmentation index and central pulse pressure, measures of arterial stiffness.
The men with a higher Mediterranean diet score (>29) had better erectile performance (SHIM scores > 14), as well as higher testosterone levels, higher coronary flow reserve, and less arterial stiffness than the other men.
The fitter men with greater exercise capacity (>10 METs) were more likely to adhere to a Mediterranean diet (scores > 25), and they also had better erectile performance (SHIM scores > 12), higher testosterone levels, greater coronary flow reserve, and less arterial stiffness than the other men.
The study did not receive any funding. The study authors and Mr. Whittaker have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2021
Florida doctor won’t treat unvaccinated patients in person
Linda Marraccini, MD, sent a letter to patients that said those who aren’t vaccinated won’t be allowed to book in-person appointments at her practice now that the Food and Drug Administration has fully approved the Pfizer vaccine for general use, according to NBC Miami.
“This is a public health emergency – the health of the public takes priority over the rights of any given individual in this situation,” she wrote in the letter, which was obtained by NBC Miami.
“It appears that there is a lack of selflessness and concern for the burden on the health and well-being of our society from our encounters,” she wrote.
Dr. Marraccini said the policy is in the best interest of her other patients and doesn’t violate the Hippocratic oath. Patients who are having chemotherapy or who have weak immune systems face higher risks of being infected with the coronavirus.
“It’s not fair for people who are unvaccinated to harm other people,” she told Newsweek Sept. 4.
“The Hippocratic oath is very science based,” she said. “I am following the science. I’m applying this to the benefit of the sick.”
Dr. Marraccini said her new policy goes into effect on Sept. 15, and she will continue to see unvaccinated patients virtually during the next month until they find another health care provider.
She said that the response to her decision has been “99.9% favorable” and that she will make exceptions if patients can’t receive a shot because of hardships. Her office also provides the Johnson & Johnson vaccine.
“We’re not going to leave them out there in the cold,” she told Newsweek.
During the past month, COVID-19 cases have surged in Florida, reaching record-level highs of more than 20,000 cases per day. Cases began rising in the summer because of the more contagious Delta variant.
In late August, a group of doctors in southern Florida urged people to get vaccinated, citing their exhaustion and frustration with unvaccinated patients who make up the large majority of COVID-19 hospitalizations, according to Newsweek.
Other doctors have declined to treat unvaccinated patients in recent weeks. Jason Valentine, MD, a doctor in Mobile, Ala., said he would no longer see unvaccinated patients as of Oct. 1, according to AL.com.
Dr. Marraccini urged people to become informed about COVID-19 vaccines, as well as their role in reducing the surge of cases in the state. The pandemic “did not have to go on this long,” she said.
“Responsibility has to do with each individual,” she told Newsweek. “This is a global health issue, and everyone owns part of that responsibility.”
A version of this article first appeared on WebMD.com.
Linda Marraccini, MD, sent a letter to patients that said those who aren’t vaccinated won’t be allowed to book in-person appointments at her practice now that the Food and Drug Administration has fully approved the Pfizer vaccine for general use, according to NBC Miami.
“This is a public health emergency – the health of the public takes priority over the rights of any given individual in this situation,” she wrote in the letter, which was obtained by NBC Miami.
“It appears that there is a lack of selflessness and concern for the burden on the health and well-being of our society from our encounters,” she wrote.
Dr. Marraccini said the policy is in the best interest of her other patients and doesn’t violate the Hippocratic oath. Patients who are having chemotherapy or who have weak immune systems face higher risks of being infected with the coronavirus.
“It’s not fair for people who are unvaccinated to harm other people,” she told Newsweek Sept. 4.
“The Hippocratic oath is very science based,” she said. “I am following the science. I’m applying this to the benefit of the sick.”
Dr. Marraccini said her new policy goes into effect on Sept. 15, and she will continue to see unvaccinated patients virtually during the next month until they find another health care provider.
She said that the response to her decision has been “99.9% favorable” and that she will make exceptions if patients can’t receive a shot because of hardships. Her office also provides the Johnson & Johnson vaccine.
“We’re not going to leave them out there in the cold,” she told Newsweek.
During the past month, COVID-19 cases have surged in Florida, reaching record-level highs of more than 20,000 cases per day. Cases began rising in the summer because of the more contagious Delta variant.
In late August, a group of doctors in southern Florida urged people to get vaccinated, citing their exhaustion and frustration with unvaccinated patients who make up the large majority of COVID-19 hospitalizations, according to Newsweek.
Other doctors have declined to treat unvaccinated patients in recent weeks. Jason Valentine, MD, a doctor in Mobile, Ala., said he would no longer see unvaccinated patients as of Oct. 1, according to AL.com.
Dr. Marraccini urged people to become informed about COVID-19 vaccines, as well as their role in reducing the surge of cases in the state. The pandemic “did not have to go on this long,” she said.
“Responsibility has to do with each individual,” she told Newsweek. “This is a global health issue, and everyone owns part of that responsibility.”
A version of this article first appeared on WebMD.com.
Linda Marraccini, MD, sent a letter to patients that said those who aren’t vaccinated won’t be allowed to book in-person appointments at her practice now that the Food and Drug Administration has fully approved the Pfizer vaccine for general use, according to NBC Miami.
“This is a public health emergency – the health of the public takes priority over the rights of any given individual in this situation,” she wrote in the letter, which was obtained by NBC Miami.
“It appears that there is a lack of selflessness and concern for the burden on the health and well-being of our society from our encounters,” she wrote.
Dr. Marraccini said the policy is in the best interest of her other patients and doesn’t violate the Hippocratic oath. Patients who are having chemotherapy or who have weak immune systems face higher risks of being infected with the coronavirus.
“It’s not fair for people who are unvaccinated to harm other people,” she told Newsweek Sept. 4.
“The Hippocratic oath is very science based,” she said. “I am following the science. I’m applying this to the benefit of the sick.”
Dr. Marraccini said her new policy goes into effect on Sept. 15, and she will continue to see unvaccinated patients virtually during the next month until they find another health care provider.
She said that the response to her decision has been “99.9% favorable” and that she will make exceptions if patients can’t receive a shot because of hardships. Her office also provides the Johnson & Johnson vaccine.
“We’re not going to leave them out there in the cold,” she told Newsweek.
During the past month, COVID-19 cases have surged in Florida, reaching record-level highs of more than 20,000 cases per day. Cases began rising in the summer because of the more contagious Delta variant.
In late August, a group of doctors in southern Florida urged people to get vaccinated, citing their exhaustion and frustration with unvaccinated patients who make up the large majority of COVID-19 hospitalizations, according to Newsweek.
Other doctors have declined to treat unvaccinated patients in recent weeks. Jason Valentine, MD, a doctor in Mobile, Ala., said he would no longer see unvaccinated patients as of Oct. 1, according to AL.com.
Dr. Marraccini urged people to become informed about COVID-19 vaccines, as well as their role in reducing the surge of cases in the state. The pandemic “did not have to go on this long,” she said.
“Responsibility has to do with each individual,” she told Newsweek. “This is a global health issue, and everyone owns part of that responsibility.”
A version of this article first appeared on WebMD.com.
United States reaches 5 million cases of child COVID
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
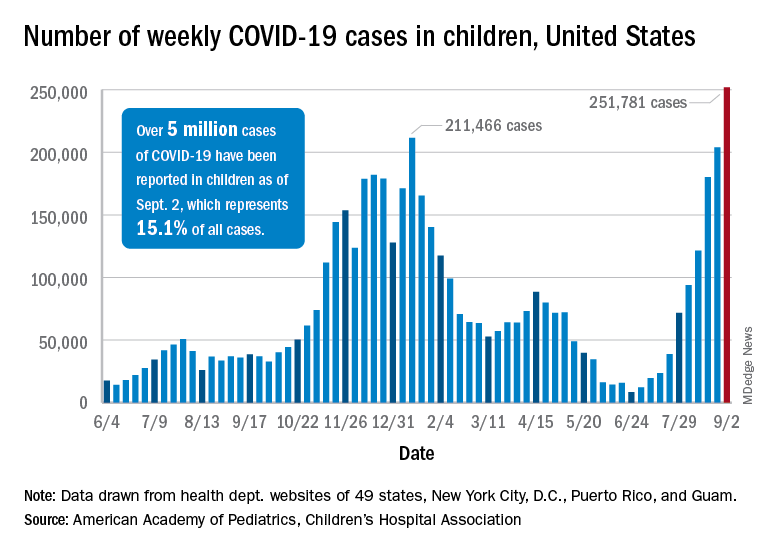
The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Cases of child COVID-19 set a new 1-week record and the total number of children infected during the pandemic passed 5 million, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The nearly 282,000 new cases reported in the United States during the week ending Sept. 2 broke the record of 211,000 set in mid-January and brought the cumulative count to 5,049,465 children with COVID-19 since the pandemic began, the AAP and the CHA said in their weekly COVID report.
Hospitalizations in children aged 0-17 years have also reached record levels in recent days. The highest daily admission rate since the pandemic began, 0.51 per 100,000 population, was recorded on Sept. 2, less than 2 months after the nation saw its lowest child COVID admission rate for 1 day: 0.07 per 100,000 on July 4. That’s an increase of 629%, according to data from the Centers for Disease Control and Prevention.
Vaccinations in children, however, did not follow suit. New vaccinations in children aged 12-17 years dropped by 4.5% for the week ending Sept. 6, compared with the week before. Initiations were actually up almost 12% for children aged 16-17, but that was not enough to overcome the continued decline among 12- to 15-year-olds, the CDC said on its COVID Data Tracker.
Despite the decline in new vaccinations, those younger children passed a noteworthy group milestone: 50.9% of all 12- to 15-year-olds now have received at least one dose, with 38.6% having completed the regimen. The 16- to 17-year-olds got an earlier start and have reached 58.9% coverage for one dose and 47.6% for two, the CDC said.
A total of 12.2 million children aged 12-17 years had received at least one dose of COVID vaccine as of Sept. 6, of whom almost 9.5 million are fully vaccinated, based on the CDC data.
At the state level, Vermont has the highest rates for vaccine initiation (75%) and full vaccination (65%), with Massachusetts (75%/62%) and Connecticut (73%/59%) just behind. The other end of the scale is occupied by Wyoming (28% initiation/19% full vaccination), Alabama (32%/19%), and North Dakota (32%/23%), the AAP said in a separate report.
In a recent letter to the Food and Drug Administration, AAP President Lee Savio Beers, MD, said that the “Delta variant is surging at extremely alarming rates in every region of America. This surge is seriously impacting all populations, including children.” Dr. Beers urged the FDA to work “aggressively toward authorizing safe and effective COVID-19 vaccines for children under age 12 as soon as possible.”
Open notes: Legal issues
In July, I had my annual physical with my primary care physician, whose practice is based out of a large urban academic medical center. As she concluded my visit and directed me to the lab to have my blood work done, she said, “You’ll be receiving an automatic notice from MyChart by 9 am tomorrow that your medical records from today’s visit are available. I apologize if I have not yet had the opportunity to review them and enter my note, but you’ll get access to all of that, as well, as soon as it is in the system.”
This sort of interaction is increasingly common across the United States as health care institutions implement policies and procedures to comply with new regulations promulgated by the Office of the National Coordinator for Health Information Technology (ONC), which went into effect on April 5, 2021. These rules were promulgated in accordance with the 21st Century Cures Act of 2016 (Cures Act).1 The regulations, known as the Interoperability, Information Blocking, and the ONC Health IT Certification Program, implement provisions of the Cures Act intended to “support the access, exchange, and use of electronic health information.” The rule is considered a significant step in the “open notes” movement, which is intended to make health care more transparent by enabling patients to access their medical records. The drafters of the ONC regulations have carved out certain exceptions to the information blocking rule. For example, one exception allows some patient information to be withheld where making that information available might cause physical harm to the patient or another person.
Thus far, few patients have been informed about the new regulation.2 By forbidding “information blocking,” the rule enables patients to more easily access and control their health information. Records must be provided “without delay,” or at least as soon as the physician’s office receives an electronic copy. In 2022, it will be required that access to even more of a patient’s personal electronic health record be provided in real-time through a patient portal and that electronic health information be shareable across third-party apps.
The Cures Act and the regulations governing its implementation highlight the inherent tension between two core principles of bioethical inquiry: autonomy and beneficence. The first principle, autonomy, champions allowing patient access and control over their own personal information. Beneficence, which is often expressed as paternalism, ensures that the experts are able to analyze and interpret data so that patients are in the best position to then make informed decisions.
With these principles in mind, arguments against open notes have generally fallen into three related categories. First, critics worry that immediate access to one’s medical record will increase patient anxiety caused by feelings of being inundated with complex medical information that patients may be ill-equipped to analyze and understand. This is a common refrain any time policies are implemented to improve medical information sharing. For example, critics of direct-to-consumer genetic testing caution that permitting unfettered access to complex information, particularly without an intermediary to interpret the data, could lead to confusion and poor medical choices.
There may be validity to this claim. One study found that 3% of patients reported feeling very confused when granted access to their medical notes.3 Another study concluded that direct release of medical test results “sometimes leads to unnecessary anxiety.”4 While the drafters of the ONC regulations have carved out certain exceptions to the information blocking rule, those exceptions do not allow for withholding of information because of concerns about patient anxiety or psychological harms.
The second common critique of open notes is that requiring release of all clinical notes will lead to clinician self-censorship, effectively muzzling or silencing the experts whose responsibility it is to objectively interpret results in order to provide the best care for their patients. Some have expressed concern that clinicians will be forced to “code” their records to avoid addressing “sensitive” subjects that might make patients feel offended or judged. This, in turn, might lead to less complete, reliable, or useful clinician communication.3
In fact, open notes has led to changes in the documentation process for some clinicians. They have reported modifying the way they document patient visits by changing their use of critical language and sensitive information.5 One study found that open notes led physicians to adjust “their language to avoid being perceived as critical of patients; omitting certain terms, such as ‘noncompliant’ and ‘patient denies’; and modifying how they document sensitive information.”3
In response, experts recommend focusing on precise and empathetic patient notes; in other words, the clinician should not write something in the note that they would not say directly to the patient. For example, they recommend that clinicians use precise language (for example, identifying the patient’s BMI) rather than using terms that could be offensive (for example, labeling the patient as “obese”).6 The shift to more empathetic note-taking could be seen less as a burden and more as a valuable tool in the shared decision-making endeavor: It could allow physicians to document both their clinical judgments and the patient’s values and preferences, which could lead to better medical decision-making.
Third, critics of open notes point to concerns about the burden it places on clinicians’ already limited time. The ONC rule requires automatic release of test results regardless of whether the clinician has had the opportunity to review them and offer their interpretation and insight. Because physician interpretation of results has known benefits,4 this puts additional pressure on clinicians to review results and enter notes in a timely manner. But physicians have reported that often open notes necessitates that they spend more time on documentation than they would otherwise.5
Despite critiques of open notes, the benefits of allowing patients access to their medical records have been repeatedly demonstrated. And research has shown that patients benefit from accessing open notes by allowing them to access and control their own personal medical information.5 Patients report that they understand and value the information provided to them in their medical records,7 and they feel empowered to participate in their medical decision-making. In surveys, patients report that reading their doctors’ notes is useful for taking care of their health and for remembering their care plans, understanding why a medication was prescribed, and reinforcing the need to take their medications and adhere to treatment plans.8
Importantly, open notes can increase patient engagement and patients’ trust in their physicians,9 thereby improving the doctor-patient relationship.3 And allowing patients to share their medical records with care partners enables supported decision-making, particularly for older and chronically ill individuals.3 Additionally, it is predicted that open notes may, in fact, decrease legal liability.9 By improving both trust in the doctor-patient relationship and safety, some experts expect that legal claims against clinicians will, in turn, decrease.10
The modern practice of medicine necessitates a more empathetic approach to clinical note-taking, even in the absence of regulation requiring it. As the regulations implementing the Cures Act roll out, patients will have easier, and more immediate, access to their medical records. Despite earlier hesitancy, clinicians are steadily beginning to support sharing access to notes with patients.5 Change can be hard. But the change expected of clinicians because of these new regulations appears to be less onerous than originally anticipated.
Prof. Koch is codirector of Health Law & Policy Institute and assistant professor at the University of Houston Law Center, as well as director of law and ethics at the MacLean Center for Clinical Medical Ethics at the University of Chicago. She has no disclosures.
This article was updated Sept. 9, 2021.
References
1. Fed Regist. 2020 May;85(85):25642-961.
2. The Petrie-Flom Center Staff. “New Rule Puts Medical Data in Patients’ Hands.” Bill of Health. July 12, 2021. Accessed August 30, 2021. https://blog.petrieflom.law.harvard.edu/2021/07/12/new-rule-puts-medical-data-in-patients-hands/.
3. Blease C et al. Ann Intern Med. 2021 Jan;174(1):101-2.
4. Pillemer F et al. PLoS One. 2016 Jun. doi: 10.1371/journal.pone.0154743.
5. DesRoches CM et al. JAMA Netw Open. 2020 Mar. doi: 10.1001/jamanetworkopen.2020.1753.
6. Heath S. “Most Patients Understand Clinical Notes, Patient Data Access.” Patient Engagement HIT. July 29, 2020. Accessed August 30, 2021. https://patientengagementhit.com/news/most-patients-understand-clinical-notes-patient-data-access
7. Leveille SG et al. J Gen Intern Med. 2020 Dec;35(12):3510-6.
8. Walker J et al. J Med Internet Res. 2019 May. doi: 10.2196/13876.
9. Bell SK et al. BMJ Qual Saf. 2017 Apr;26(4):262-70.
10. Kachalia A, Mello MM. N Engl J Med. 2011 Apr;364(16):1564-72.
In July, I had my annual physical with my primary care physician, whose practice is based out of a large urban academic medical center. As she concluded my visit and directed me to the lab to have my blood work done, she said, “You’ll be receiving an automatic notice from MyChart by 9 am tomorrow that your medical records from today’s visit are available. I apologize if I have not yet had the opportunity to review them and enter my note, but you’ll get access to all of that, as well, as soon as it is in the system.”
This sort of interaction is increasingly common across the United States as health care institutions implement policies and procedures to comply with new regulations promulgated by the Office of the National Coordinator for Health Information Technology (ONC), which went into effect on April 5, 2021. These rules were promulgated in accordance with the 21st Century Cures Act of 2016 (Cures Act).1 The regulations, known as the Interoperability, Information Blocking, and the ONC Health IT Certification Program, implement provisions of the Cures Act intended to “support the access, exchange, and use of electronic health information.” The rule is considered a significant step in the “open notes” movement, which is intended to make health care more transparent by enabling patients to access their medical records. The drafters of the ONC regulations have carved out certain exceptions to the information blocking rule. For example, one exception allows some patient information to be withheld where making that information available might cause physical harm to the patient or another person.
Thus far, few patients have been informed about the new regulation.2 By forbidding “information blocking,” the rule enables patients to more easily access and control their health information. Records must be provided “without delay,” or at least as soon as the physician’s office receives an electronic copy. In 2022, it will be required that access to even more of a patient’s personal electronic health record be provided in real-time through a patient portal and that electronic health information be shareable across third-party apps.
The Cures Act and the regulations governing its implementation highlight the inherent tension between two core principles of bioethical inquiry: autonomy and beneficence. The first principle, autonomy, champions allowing patient access and control over their own personal information. Beneficence, which is often expressed as paternalism, ensures that the experts are able to analyze and interpret data so that patients are in the best position to then make informed decisions.
With these principles in mind, arguments against open notes have generally fallen into three related categories. First, critics worry that immediate access to one’s medical record will increase patient anxiety caused by feelings of being inundated with complex medical information that patients may be ill-equipped to analyze and understand. This is a common refrain any time policies are implemented to improve medical information sharing. For example, critics of direct-to-consumer genetic testing caution that permitting unfettered access to complex information, particularly without an intermediary to interpret the data, could lead to confusion and poor medical choices.
There may be validity to this claim. One study found that 3% of patients reported feeling very confused when granted access to their medical notes.3 Another study concluded that direct release of medical test results “sometimes leads to unnecessary anxiety.”4 While the drafters of the ONC regulations have carved out certain exceptions to the information blocking rule, those exceptions do not allow for withholding of information because of concerns about patient anxiety or psychological harms.
The second common critique of open notes is that requiring release of all clinical notes will lead to clinician self-censorship, effectively muzzling or silencing the experts whose responsibility it is to objectively interpret results in order to provide the best care for their patients. Some have expressed concern that clinicians will be forced to “code” their records to avoid addressing “sensitive” subjects that might make patients feel offended or judged. This, in turn, might lead to less complete, reliable, or useful clinician communication.3
In fact, open notes has led to changes in the documentation process for some clinicians. They have reported modifying the way they document patient visits by changing their use of critical language and sensitive information.5 One study found that open notes led physicians to adjust “their language to avoid being perceived as critical of patients; omitting certain terms, such as ‘noncompliant’ and ‘patient denies’; and modifying how they document sensitive information.”3
In response, experts recommend focusing on precise and empathetic patient notes; in other words, the clinician should not write something in the note that they would not say directly to the patient. For example, they recommend that clinicians use precise language (for example, identifying the patient’s BMI) rather than using terms that could be offensive (for example, labeling the patient as “obese”).6 The shift to more empathetic note-taking could be seen less as a burden and more as a valuable tool in the shared decision-making endeavor: It could allow physicians to document both their clinical judgments and the patient’s values and preferences, which could lead to better medical decision-making.
Third, critics of open notes point to concerns about the burden it places on clinicians’ already limited time. The ONC rule requires automatic release of test results regardless of whether the clinician has had the opportunity to review them and offer their interpretation and insight. Because physician interpretation of results has known benefits,4 this puts additional pressure on clinicians to review results and enter notes in a timely manner. But physicians have reported that often open notes necessitates that they spend more time on documentation than they would otherwise.5
Despite critiques of open notes, the benefits of allowing patients access to their medical records have been repeatedly demonstrated. And research has shown that patients benefit from accessing open notes by allowing them to access and control their own personal medical information.5 Patients report that they understand and value the information provided to them in their medical records,7 and they feel empowered to participate in their medical decision-making. In surveys, patients report that reading their doctors’ notes is useful for taking care of their health and for remembering their care plans, understanding why a medication was prescribed, and reinforcing the need to take their medications and adhere to treatment plans.8
Importantly, open notes can increase patient engagement and patients’ trust in their physicians,9 thereby improving the doctor-patient relationship.3 And allowing patients to share their medical records with care partners enables supported decision-making, particularly for older and chronically ill individuals.3 Additionally, it is predicted that open notes may, in fact, decrease legal liability.9 By improving both trust in the doctor-patient relationship and safety, some experts expect that legal claims against clinicians will, in turn, decrease.10
The modern practice of medicine necessitates a more empathetic approach to clinical note-taking, even in the absence of regulation requiring it. As the regulations implementing the Cures Act roll out, patients will have easier, and more immediate, access to their medical records. Despite earlier hesitancy, clinicians are steadily beginning to support sharing access to notes with patients.5 Change can be hard. But the change expected of clinicians because of these new regulations appears to be less onerous than originally anticipated.
Prof. Koch is codirector of Health Law & Policy Institute and assistant professor at the University of Houston Law Center, as well as director of law and ethics at the MacLean Center for Clinical Medical Ethics at the University of Chicago. She has no disclosures.
This article was updated Sept. 9, 2021.
References
1. Fed Regist. 2020 May;85(85):25642-961.
2. The Petrie-Flom Center Staff. “New Rule Puts Medical Data in Patients’ Hands.” Bill of Health. July 12, 2021. Accessed August 30, 2021. https://blog.petrieflom.law.harvard.edu/2021/07/12/new-rule-puts-medical-data-in-patients-hands/.
3. Blease C et al. Ann Intern Med. 2021 Jan;174(1):101-2.
4. Pillemer F et al. PLoS One. 2016 Jun. doi: 10.1371/journal.pone.0154743.
5. DesRoches CM et al. JAMA Netw Open. 2020 Mar. doi: 10.1001/jamanetworkopen.2020.1753.
6. Heath S. “Most Patients Understand Clinical Notes, Patient Data Access.” Patient Engagement HIT. July 29, 2020. Accessed August 30, 2021. https://patientengagementhit.com/news/most-patients-understand-clinical-notes-patient-data-access
7. Leveille SG et al. J Gen Intern Med. 2020 Dec;35(12):3510-6.
8. Walker J et al. J Med Internet Res. 2019 May. doi: 10.2196/13876.
9. Bell SK et al. BMJ Qual Saf. 2017 Apr;26(4):262-70.
10. Kachalia A, Mello MM. N Engl J Med. 2011 Apr;364(16):1564-72.
In July, I had my annual physical with my primary care physician, whose practice is based out of a large urban academic medical center. As she concluded my visit and directed me to the lab to have my blood work done, she said, “You’ll be receiving an automatic notice from MyChart by 9 am tomorrow that your medical records from today’s visit are available. I apologize if I have not yet had the opportunity to review them and enter my note, but you’ll get access to all of that, as well, as soon as it is in the system.”
This sort of interaction is increasingly common across the United States as health care institutions implement policies and procedures to comply with new regulations promulgated by the Office of the National Coordinator for Health Information Technology (ONC), which went into effect on April 5, 2021. These rules were promulgated in accordance with the 21st Century Cures Act of 2016 (Cures Act).1 The regulations, known as the Interoperability, Information Blocking, and the ONC Health IT Certification Program, implement provisions of the Cures Act intended to “support the access, exchange, and use of electronic health information.” The rule is considered a significant step in the “open notes” movement, which is intended to make health care more transparent by enabling patients to access their medical records. The drafters of the ONC regulations have carved out certain exceptions to the information blocking rule. For example, one exception allows some patient information to be withheld where making that information available might cause physical harm to the patient or another person.
Thus far, few patients have been informed about the new regulation.2 By forbidding “information blocking,” the rule enables patients to more easily access and control their health information. Records must be provided “without delay,” or at least as soon as the physician’s office receives an electronic copy. In 2022, it will be required that access to even more of a patient’s personal electronic health record be provided in real-time through a patient portal and that electronic health information be shareable across third-party apps.
The Cures Act and the regulations governing its implementation highlight the inherent tension between two core principles of bioethical inquiry: autonomy and beneficence. The first principle, autonomy, champions allowing patient access and control over their own personal information. Beneficence, which is often expressed as paternalism, ensures that the experts are able to analyze and interpret data so that patients are in the best position to then make informed decisions.
With these principles in mind, arguments against open notes have generally fallen into three related categories. First, critics worry that immediate access to one’s medical record will increase patient anxiety caused by feelings of being inundated with complex medical information that patients may be ill-equipped to analyze and understand. This is a common refrain any time policies are implemented to improve medical information sharing. For example, critics of direct-to-consumer genetic testing caution that permitting unfettered access to complex information, particularly without an intermediary to interpret the data, could lead to confusion and poor medical choices.
There may be validity to this claim. One study found that 3% of patients reported feeling very confused when granted access to their medical notes.3 Another study concluded that direct release of medical test results “sometimes leads to unnecessary anxiety.”4 While the drafters of the ONC regulations have carved out certain exceptions to the information blocking rule, those exceptions do not allow for withholding of information because of concerns about patient anxiety or psychological harms.
The second common critique of open notes is that requiring release of all clinical notes will lead to clinician self-censorship, effectively muzzling or silencing the experts whose responsibility it is to objectively interpret results in order to provide the best care for their patients. Some have expressed concern that clinicians will be forced to “code” their records to avoid addressing “sensitive” subjects that might make patients feel offended or judged. This, in turn, might lead to less complete, reliable, or useful clinician communication.3
In fact, open notes has led to changes in the documentation process for some clinicians. They have reported modifying the way they document patient visits by changing their use of critical language and sensitive information.5 One study found that open notes led physicians to adjust “their language to avoid being perceived as critical of patients; omitting certain terms, such as ‘noncompliant’ and ‘patient denies’; and modifying how they document sensitive information.”3
In response, experts recommend focusing on precise and empathetic patient notes; in other words, the clinician should not write something in the note that they would not say directly to the patient. For example, they recommend that clinicians use precise language (for example, identifying the patient’s BMI) rather than using terms that could be offensive (for example, labeling the patient as “obese”).6 The shift to more empathetic note-taking could be seen less as a burden and more as a valuable tool in the shared decision-making endeavor: It could allow physicians to document both their clinical judgments and the patient’s values and preferences, which could lead to better medical decision-making.
Third, critics of open notes point to concerns about the burden it places on clinicians’ already limited time. The ONC rule requires automatic release of test results regardless of whether the clinician has had the opportunity to review them and offer their interpretation and insight. Because physician interpretation of results has known benefits,4 this puts additional pressure on clinicians to review results and enter notes in a timely manner. But physicians have reported that often open notes necessitates that they spend more time on documentation than they would otherwise.5
Despite critiques of open notes, the benefits of allowing patients access to their medical records have been repeatedly demonstrated. And research has shown that patients benefit from accessing open notes by allowing them to access and control their own personal medical information.5 Patients report that they understand and value the information provided to them in their medical records,7 and they feel empowered to participate in their medical decision-making. In surveys, patients report that reading their doctors’ notes is useful for taking care of their health and for remembering their care plans, understanding why a medication was prescribed, and reinforcing the need to take their medications and adhere to treatment plans.8
Importantly, open notes can increase patient engagement and patients’ trust in their physicians,9 thereby improving the doctor-patient relationship.3 And allowing patients to share their medical records with care partners enables supported decision-making, particularly for older and chronically ill individuals.3 Additionally, it is predicted that open notes may, in fact, decrease legal liability.9 By improving both trust in the doctor-patient relationship and safety, some experts expect that legal claims against clinicians will, in turn, decrease.10
The modern practice of medicine necessitates a more empathetic approach to clinical note-taking, even in the absence of regulation requiring it. As the regulations implementing the Cures Act roll out, patients will have easier, and more immediate, access to their medical records. Despite earlier hesitancy, clinicians are steadily beginning to support sharing access to notes with patients.5 Change can be hard. But the change expected of clinicians because of these new regulations appears to be less onerous than originally anticipated.
Prof. Koch is codirector of Health Law & Policy Institute and assistant professor at the University of Houston Law Center, as well as director of law and ethics at the MacLean Center for Clinical Medical Ethics at the University of Chicago. She has no disclosures.
This article was updated Sept. 9, 2021.
References
1. Fed Regist. 2020 May;85(85):25642-961.
2. The Petrie-Flom Center Staff. “New Rule Puts Medical Data in Patients’ Hands.” Bill of Health. July 12, 2021. Accessed August 30, 2021. https://blog.petrieflom.law.harvard.edu/2021/07/12/new-rule-puts-medical-data-in-patients-hands/.
3. Blease C et al. Ann Intern Med. 2021 Jan;174(1):101-2.
4. Pillemer F et al. PLoS One. 2016 Jun. doi: 10.1371/journal.pone.0154743.
5. DesRoches CM et al. JAMA Netw Open. 2020 Mar. doi: 10.1001/jamanetworkopen.2020.1753.
6. Heath S. “Most Patients Understand Clinical Notes, Patient Data Access.” Patient Engagement HIT. July 29, 2020. Accessed August 30, 2021. https://patientengagementhit.com/news/most-patients-understand-clinical-notes-patient-data-access
7. Leveille SG et al. J Gen Intern Med. 2020 Dec;35(12):3510-6.
8. Walker J et al. J Med Internet Res. 2019 May. doi: 10.2196/13876.
9. Bell SK et al. BMJ Qual Saf. 2017 Apr;26(4):262-70.
10. Kachalia A, Mello MM. N Engl J Med. 2011 Apr;364(16):1564-72.
COVID-19 continues to complicate children’s mental health care
The COVID-19 pandemic continues to impact child and adolescent mental health, and clinicians are learning as they go to develop strategies that address the challenges of providing both medical and mental health care to young patients, including those who test positive for COVID-19, according to Hani Talebi, PhD, director of pediatric psychology, and Jorge Ganem, MD, FAAP, director of pediatric hospital medicine, both of the University of Texas at Austin and Dell Children’s Medical Center.
In a presentation at the 2021 virtual Pediatric Hospital Medicine conference, Dr. Talebi and Dr. Ganem shared their experiences in identifying the impact of the pandemic on mental health services in a freestanding hospital, and synthesizing inpatient mental health care and medical care outside of a dedicated mental health unit.
Mental health is a significant pediatric issue; approximately one in five children have a diagnosable mental or behavioral health problem, but nearly two-thirds get little or no help, Dr. Talebi said. “COVID-19 has only exacerbated these mental health challenges,” he said.
He noted that beginning in April 2020, the proportion of children’s mental health-related emergency department visits increased and remained elevated through the spring, summer, and fall of 2020, as families fearful of COVID-19 avoided regular hospital visits.
Data suggest that up to 50% of all adolescent psychiatric crises that led to inpatient admissions were related in some way to COVID-19, Dr. Talebi said. In addition, “individuals with a recent diagnosis of a mental health disorder are at increased risk for COVID-19 infection,” and the risk is even higher among women and African Americans, he said.
The past year significantly impacted the mental wellbeing of parents and children, Dr. Talebi said. He cited a June 2020 study in Pediatrics in which 27% of parents reported worsening mental health for themselves, and 14% reported worsening behavioral health for their children. Ongoing issues including food insecurity, loss of regular child care, and an overall “very disorienting experience in the day-to-day” compromised the mental health of families, Dr. Talebi emphasized. Children isolated at home were not meeting developmental milestones that organically occur when socializing with peers, parents didn’t know how to handle some of their children’s issues without support from schools, and many people were struggling with other preexisting health conditions, he said.
This confluence of factors helped drive a surge in emergency department visits, meaning longer wait times and concerns about meeting urgent medical and mental health needs while maintaining safety, he added.
Parents and children waited longer to seek care, and community hospitals such as Dell Children’s Medical Center were faced with children in the emergency department with crisis-level mental health issues, along with children already waiting in the ED to address medical emergencies. All these patients had to be tested for COVID-19 and managed accordingly, Dr. Talebi noted.
Dr. Talebi emphasized the need for clinically robust care of the children who were in isolation for 10 days on the medical unit, waiting to test negative. New protocols were created for social workers to conduct daily safety checks, and to develop regular schedules for screening, “so they are having an experience on the medical floors similar to what they would have in a mental health unit,” he said.
Dr. Ganem reflected on the logistical challenges of managing mental health care while observing COVID-19 safety protocols. “COVID-19 added a new wrinkle of isolation,” he said. As institutional guidelines on testing and isolation evolved, negative COVID-19 tests were required for admission to the mental health units both in the hospital and throughout the region. Patients who tested positive had to be quarantined for 10 days, at which time they could be admitted to a mental health unit if necessary, he said.
Dr. Ganem shared details of some strategies adopted by Dell Children’s. He explained that the COVID-19 psychiatry patient workflow started with an ED evaluation, followed by medical clearance and consideration for admission.
“There was significant coordination between the social worker in the emergency department and the psychiatry social worker,” he said.
Key elements of the treatment plan for children with positive COVID-19 tests included an “interprofessional huddle” to coordinate the plan of care, goals for admission, and goals for safety, Dr. Ganem said.
Patients who required admission were expected to have an initial length of stay of 72 hours, and those who tested positive for COVID-19 were admitted to a medical unit with COVID-19 isolation, he said.
Once a patient is admitted, an RN activates a suicide prevention pathway, and an interprofessional team meets to determine what patients need for safe and effective discharge, said Dr. Ganem. He cited the SAFE-T protocol (Suicide Assessment Five-step Evaluation and Triage) as one of the tools used to determine safe discharge criteria. Considerations on the SAFE-T list include family support, an established outpatient therapist and psychiatrist, no suicide attempts prior to the current admission, or a low lethality attempt, and access to partial hospitalization or intensive outpatient programs.
Patients who could not be discharged because of suicidality or inadequate support or concerns about safety at home were considered for inpatient admission. Patients with COVID-19–positive tests who had continued need for inpatient mental health services could be transferred to an inpatient mental health unit after a 10-day quarantine.
Overall, “this has been a continuum of lessons learned, with some things we know now that we didn’t know in April or May of 2020,” Dr. Ganem said. Early in the pandemic, the focus was on minimizing risk, securing personal protective equipment, and determining who provided services in a patient’s room. “We developed new paradigms on the fly,” he said, including the use of virtual visits, which included securing and cleaning devices, as well as learning how to use them in this setting,” he said.
More recently, the emphasis has been on providing services to patients before they need to visit the hospital, rather than automatically admitting any patients with suicidal ideation and a positive COVID-19 test, Dr. Ganem said.
Dr. Talebi and Dr. Ganem had no financial conflicts to disclose. The conference was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
The COVID-19 pandemic continues to impact child and adolescent mental health, and clinicians are learning as they go to develop strategies that address the challenges of providing both medical and mental health care to young patients, including those who test positive for COVID-19, according to Hani Talebi, PhD, director of pediatric psychology, and Jorge Ganem, MD, FAAP, director of pediatric hospital medicine, both of the University of Texas at Austin and Dell Children’s Medical Center.
In a presentation at the 2021 virtual Pediatric Hospital Medicine conference, Dr. Talebi and Dr. Ganem shared their experiences in identifying the impact of the pandemic on mental health services in a freestanding hospital, and synthesizing inpatient mental health care and medical care outside of a dedicated mental health unit.
Mental health is a significant pediatric issue; approximately one in five children have a diagnosable mental or behavioral health problem, but nearly two-thirds get little or no help, Dr. Talebi said. “COVID-19 has only exacerbated these mental health challenges,” he said.
He noted that beginning in April 2020, the proportion of children’s mental health-related emergency department visits increased and remained elevated through the spring, summer, and fall of 2020, as families fearful of COVID-19 avoided regular hospital visits.
Data suggest that up to 50% of all adolescent psychiatric crises that led to inpatient admissions were related in some way to COVID-19, Dr. Talebi said. In addition, “individuals with a recent diagnosis of a mental health disorder are at increased risk for COVID-19 infection,” and the risk is even higher among women and African Americans, he said.
The past year significantly impacted the mental wellbeing of parents and children, Dr. Talebi said. He cited a June 2020 study in Pediatrics in which 27% of parents reported worsening mental health for themselves, and 14% reported worsening behavioral health for their children. Ongoing issues including food insecurity, loss of regular child care, and an overall “very disorienting experience in the day-to-day” compromised the mental health of families, Dr. Talebi emphasized. Children isolated at home were not meeting developmental milestones that organically occur when socializing with peers, parents didn’t know how to handle some of their children’s issues without support from schools, and many people were struggling with other preexisting health conditions, he said.
This confluence of factors helped drive a surge in emergency department visits, meaning longer wait times and concerns about meeting urgent medical and mental health needs while maintaining safety, he added.
Parents and children waited longer to seek care, and community hospitals such as Dell Children’s Medical Center were faced with children in the emergency department with crisis-level mental health issues, along with children already waiting in the ED to address medical emergencies. All these patients had to be tested for COVID-19 and managed accordingly, Dr. Talebi noted.
Dr. Talebi emphasized the need for clinically robust care of the children who were in isolation for 10 days on the medical unit, waiting to test negative. New protocols were created for social workers to conduct daily safety checks, and to develop regular schedules for screening, “so they are having an experience on the medical floors similar to what they would have in a mental health unit,” he said.
Dr. Ganem reflected on the logistical challenges of managing mental health care while observing COVID-19 safety protocols. “COVID-19 added a new wrinkle of isolation,” he said. As institutional guidelines on testing and isolation evolved, negative COVID-19 tests were required for admission to the mental health units both in the hospital and throughout the region. Patients who tested positive had to be quarantined for 10 days, at which time they could be admitted to a mental health unit if necessary, he said.
Dr. Ganem shared details of some strategies adopted by Dell Children’s. He explained that the COVID-19 psychiatry patient workflow started with an ED evaluation, followed by medical clearance and consideration for admission.
“There was significant coordination between the social worker in the emergency department and the psychiatry social worker,” he said.
Key elements of the treatment plan for children with positive COVID-19 tests included an “interprofessional huddle” to coordinate the plan of care, goals for admission, and goals for safety, Dr. Ganem said.
Patients who required admission were expected to have an initial length of stay of 72 hours, and those who tested positive for COVID-19 were admitted to a medical unit with COVID-19 isolation, he said.
Once a patient is admitted, an RN activates a suicide prevention pathway, and an interprofessional team meets to determine what patients need for safe and effective discharge, said Dr. Ganem. He cited the SAFE-T protocol (Suicide Assessment Five-step Evaluation and Triage) as one of the tools used to determine safe discharge criteria. Considerations on the SAFE-T list include family support, an established outpatient therapist and psychiatrist, no suicide attempts prior to the current admission, or a low lethality attempt, and access to partial hospitalization or intensive outpatient programs.
Patients who could not be discharged because of suicidality or inadequate support or concerns about safety at home were considered for inpatient admission. Patients with COVID-19–positive tests who had continued need for inpatient mental health services could be transferred to an inpatient mental health unit after a 10-day quarantine.
Overall, “this has been a continuum of lessons learned, with some things we know now that we didn’t know in April or May of 2020,” Dr. Ganem said. Early in the pandemic, the focus was on minimizing risk, securing personal protective equipment, and determining who provided services in a patient’s room. “We developed new paradigms on the fly,” he said, including the use of virtual visits, which included securing and cleaning devices, as well as learning how to use them in this setting,” he said.
More recently, the emphasis has been on providing services to patients before they need to visit the hospital, rather than automatically admitting any patients with suicidal ideation and a positive COVID-19 test, Dr. Ganem said.
Dr. Talebi and Dr. Ganem had no financial conflicts to disclose. The conference was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
The COVID-19 pandemic continues to impact child and adolescent mental health, and clinicians are learning as they go to develop strategies that address the challenges of providing both medical and mental health care to young patients, including those who test positive for COVID-19, according to Hani Talebi, PhD, director of pediatric psychology, and Jorge Ganem, MD, FAAP, director of pediatric hospital medicine, both of the University of Texas at Austin and Dell Children’s Medical Center.
In a presentation at the 2021 virtual Pediatric Hospital Medicine conference, Dr. Talebi and Dr. Ganem shared their experiences in identifying the impact of the pandemic on mental health services in a freestanding hospital, and synthesizing inpatient mental health care and medical care outside of a dedicated mental health unit.
Mental health is a significant pediatric issue; approximately one in five children have a diagnosable mental or behavioral health problem, but nearly two-thirds get little or no help, Dr. Talebi said. “COVID-19 has only exacerbated these mental health challenges,” he said.
He noted that beginning in April 2020, the proportion of children’s mental health-related emergency department visits increased and remained elevated through the spring, summer, and fall of 2020, as families fearful of COVID-19 avoided regular hospital visits.
Data suggest that up to 50% of all adolescent psychiatric crises that led to inpatient admissions were related in some way to COVID-19, Dr. Talebi said. In addition, “individuals with a recent diagnosis of a mental health disorder are at increased risk for COVID-19 infection,” and the risk is even higher among women and African Americans, he said.
The past year significantly impacted the mental wellbeing of parents and children, Dr. Talebi said. He cited a June 2020 study in Pediatrics in which 27% of parents reported worsening mental health for themselves, and 14% reported worsening behavioral health for their children. Ongoing issues including food insecurity, loss of regular child care, and an overall “very disorienting experience in the day-to-day” compromised the mental health of families, Dr. Talebi emphasized. Children isolated at home were not meeting developmental milestones that organically occur when socializing with peers, parents didn’t know how to handle some of their children’s issues without support from schools, and many people were struggling with other preexisting health conditions, he said.
This confluence of factors helped drive a surge in emergency department visits, meaning longer wait times and concerns about meeting urgent medical and mental health needs while maintaining safety, he added.
Parents and children waited longer to seek care, and community hospitals such as Dell Children’s Medical Center were faced with children in the emergency department with crisis-level mental health issues, along with children already waiting in the ED to address medical emergencies. All these patients had to be tested for COVID-19 and managed accordingly, Dr. Talebi noted.
Dr. Talebi emphasized the need for clinically robust care of the children who were in isolation for 10 days on the medical unit, waiting to test negative. New protocols were created for social workers to conduct daily safety checks, and to develop regular schedules for screening, “so they are having an experience on the medical floors similar to what they would have in a mental health unit,” he said.
Dr. Ganem reflected on the logistical challenges of managing mental health care while observing COVID-19 safety protocols. “COVID-19 added a new wrinkle of isolation,” he said. As institutional guidelines on testing and isolation evolved, negative COVID-19 tests were required for admission to the mental health units both in the hospital and throughout the region. Patients who tested positive had to be quarantined for 10 days, at which time they could be admitted to a mental health unit if necessary, he said.
Dr. Ganem shared details of some strategies adopted by Dell Children’s. He explained that the COVID-19 psychiatry patient workflow started with an ED evaluation, followed by medical clearance and consideration for admission.
“There was significant coordination between the social worker in the emergency department and the psychiatry social worker,” he said.
Key elements of the treatment plan for children with positive COVID-19 tests included an “interprofessional huddle” to coordinate the plan of care, goals for admission, and goals for safety, Dr. Ganem said.
Patients who required admission were expected to have an initial length of stay of 72 hours, and those who tested positive for COVID-19 were admitted to a medical unit with COVID-19 isolation, he said.
Once a patient is admitted, an RN activates a suicide prevention pathway, and an interprofessional team meets to determine what patients need for safe and effective discharge, said Dr. Ganem. He cited the SAFE-T protocol (Suicide Assessment Five-step Evaluation and Triage) as one of the tools used to determine safe discharge criteria. Considerations on the SAFE-T list include family support, an established outpatient therapist and psychiatrist, no suicide attempts prior to the current admission, or a low lethality attempt, and access to partial hospitalization or intensive outpatient programs.
Patients who could not be discharged because of suicidality or inadequate support or concerns about safety at home were considered for inpatient admission. Patients with COVID-19–positive tests who had continued need for inpatient mental health services could be transferred to an inpatient mental health unit after a 10-day quarantine.
Overall, “this has been a continuum of lessons learned, with some things we know now that we didn’t know in April or May of 2020,” Dr. Ganem said. Early in the pandemic, the focus was on minimizing risk, securing personal protective equipment, and determining who provided services in a patient’s room. “We developed new paradigms on the fly,” he said, including the use of virtual visits, which included securing and cleaning devices, as well as learning how to use them in this setting,” he said.
More recently, the emphasis has been on providing services to patients before they need to visit the hospital, rather than automatically admitting any patients with suicidal ideation and a positive COVID-19 test, Dr. Ganem said.
Dr. Talebi and Dr. Ganem had no financial conflicts to disclose. The conference was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
FROM PHM 2021
Iron deficiency in pregnancy is common, yet many aren’t being screened for it
Many pregnant patients are not being screened for iron deficiency despite it being a common cause of anemia in pregnancy that could increase the risk of maternal and infant death.
Researchers analyzed data from 44,552 pregnant patients in Ontario, Canada, collected between 2013 and 2018 to determine the prevalence of ferritin testing, the standard test for iron deficiency, over the course of 5 years.
Their study, published in Blood Advances, revealed that only 59.4% of pregnant persons received a ferritin test, the standard test for iron deficiency. Of those pregnant persons, 25.2% were iron insufficient and 52.8% were iron deficient at least once during pregnancy.
They also found that 71% of these iron tests were ordered during the first trimester, when the risk of iron deficiency is lowest.
“We are not only missing a very large proportion of women who are iron deficient going into pregnancy, but we’re missing those that become iron deficient later on in their pregnancies,” study author Dr. Jennifer Teichman, hematology resident at the University of Toronto, said in an interview. Researchers said iron deficiency during pregnancy is associated with maternal fatigue, cognitive dysfunction, depression, low birth weight, and poor brain development of the child.
Dr. Teichman explained that if iron deficiency during pregnancy is identified early enough, doctors would have enough time to treat the condition with iron supplements before the patient goes into delivery. She also explained prenatal vitamins, which contain some iron, do not contain enough of the mineral to fix iron deficiency.
“One really important point is that the amount of iron contained in a prenatal vitamin is really low,” Dr. Teichman explained. “It’s enough to make up the difference of the additional iron that she needs to sustain her pregnancy, but it’s not enough to treat a woman who’s already got low iron going into pregnancy. So there’s a difference between a prenatal vitamin and true iron supplementation.”
Researchers also found that those who came from a household with a low annual income were even less likely to receive a ferritin test, which was a troubling finding since women of lower socioeconomic status are more likely to be iron deficient in pregnancy.
“[This] says something about how we as health care providers are contributing to this gap in care,” Dr. Teichman said. “Women of lower socioeconomic status sort of have a triple whammy: They’re more likely to be iron deficient, they’re less likely to have it diagnosed, and they’re less likely to have it corrected.”
Dr. Teichman and her colleagues took a unique approach by looking at isolated ferritin levels as opposed to complete blood counts, which is the typical screening for anemia in pregnancy, said Lissette Tanner, MD, MPH, FACOG, who was not involved with the study.
“Those who meet the criteria for anemia should be evaluated for the cause with initial suspicion for iron deficiency anemia, as that is the most common etiology,” said Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta.
The Centers for Disease Control and Prevention recommends screening for iron deficiency anemia in pregnant persons, in addition to universal iron supplementation to meet the iron requirements of pregnancy.
Additionally, the American College of Obstetricians and Gynecologists recommends that all pregnant persons be screened for anemia with a complete blood count in the first trimester and again between 24 and 28 weeks of pregnancy.
However, iron deficiency is completely missed by ACOG’s recommendations, said Michael Auerbach, MD, of the department of medicine, Georgetown University, Washington.
“They recommend a [complete blood count] on all presenting pregnant women, but they do not recommend iron parameters, including a ferritin test, unless the mother is anemic,” said Dr. Auerbach, who was not involved in the study. “I think those guidelines are in need of revision.”
Dr. Teichman hopes her team’s findings will motivate change in obstetric and hematologic guidelines that recommend routine prenatal testing.
“I think ferritin should be a part of routine prenatal testing,” Dr. Teichman said. “And I also think that patients need to be empowered to ask what their iron levels are in pregnancy and providers need to know what a normal iron level is.”
None of the experts interviewed for this story had financial conflicts of interest.
Many pregnant patients are not being screened for iron deficiency despite it being a common cause of anemia in pregnancy that could increase the risk of maternal and infant death.
Researchers analyzed data from 44,552 pregnant patients in Ontario, Canada, collected between 2013 and 2018 to determine the prevalence of ferritin testing, the standard test for iron deficiency, over the course of 5 years.
Their study, published in Blood Advances, revealed that only 59.4% of pregnant persons received a ferritin test, the standard test for iron deficiency. Of those pregnant persons, 25.2% were iron insufficient and 52.8% were iron deficient at least once during pregnancy.
They also found that 71% of these iron tests were ordered during the first trimester, when the risk of iron deficiency is lowest.
“We are not only missing a very large proportion of women who are iron deficient going into pregnancy, but we’re missing those that become iron deficient later on in their pregnancies,” study author Dr. Jennifer Teichman, hematology resident at the University of Toronto, said in an interview. Researchers said iron deficiency during pregnancy is associated with maternal fatigue, cognitive dysfunction, depression, low birth weight, and poor brain development of the child.
Dr. Teichman explained that if iron deficiency during pregnancy is identified early enough, doctors would have enough time to treat the condition with iron supplements before the patient goes into delivery. She also explained prenatal vitamins, which contain some iron, do not contain enough of the mineral to fix iron deficiency.
“One really important point is that the amount of iron contained in a prenatal vitamin is really low,” Dr. Teichman explained. “It’s enough to make up the difference of the additional iron that she needs to sustain her pregnancy, but it’s not enough to treat a woman who’s already got low iron going into pregnancy. So there’s a difference between a prenatal vitamin and true iron supplementation.”
Researchers also found that those who came from a household with a low annual income were even less likely to receive a ferritin test, which was a troubling finding since women of lower socioeconomic status are more likely to be iron deficient in pregnancy.
“[This] says something about how we as health care providers are contributing to this gap in care,” Dr. Teichman said. “Women of lower socioeconomic status sort of have a triple whammy: They’re more likely to be iron deficient, they’re less likely to have it diagnosed, and they’re less likely to have it corrected.”
Dr. Teichman and her colleagues took a unique approach by looking at isolated ferritin levels as opposed to complete blood counts, which is the typical screening for anemia in pregnancy, said Lissette Tanner, MD, MPH, FACOG, who was not involved with the study.
“Those who meet the criteria for anemia should be evaluated for the cause with initial suspicion for iron deficiency anemia, as that is the most common etiology,” said Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta.
The Centers for Disease Control and Prevention recommends screening for iron deficiency anemia in pregnant persons, in addition to universal iron supplementation to meet the iron requirements of pregnancy.
Additionally, the American College of Obstetricians and Gynecologists recommends that all pregnant persons be screened for anemia with a complete blood count in the first trimester and again between 24 and 28 weeks of pregnancy.
However, iron deficiency is completely missed by ACOG’s recommendations, said Michael Auerbach, MD, of the department of medicine, Georgetown University, Washington.
“They recommend a [complete blood count] on all presenting pregnant women, but they do not recommend iron parameters, including a ferritin test, unless the mother is anemic,” said Dr. Auerbach, who was not involved in the study. “I think those guidelines are in need of revision.”
Dr. Teichman hopes her team’s findings will motivate change in obstetric and hematologic guidelines that recommend routine prenatal testing.
“I think ferritin should be a part of routine prenatal testing,” Dr. Teichman said. “And I also think that patients need to be empowered to ask what their iron levels are in pregnancy and providers need to know what a normal iron level is.”
None of the experts interviewed for this story had financial conflicts of interest.
Many pregnant patients are not being screened for iron deficiency despite it being a common cause of anemia in pregnancy that could increase the risk of maternal and infant death.
Researchers analyzed data from 44,552 pregnant patients in Ontario, Canada, collected between 2013 and 2018 to determine the prevalence of ferritin testing, the standard test for iron deficiency, over the course of 5 years.
Their study, published in Blood Advances, revealed that only 59.4% of pregnant persons received a ferritin test, the standard test for iron deficiency. Of those pregnant persons, 25.2% were iron insufficient and 52.8% were iron deficient at least once during pregnancy.
They also found that 71% of these iron tests were ordered during the first trimester, when the risk of iron deficiency is lowest.
“We are not only missing a very large proportion of women who are iron deficient going into pregnancy, but we’re missing those that become iron deficient later on in their pregnancies,” study author Dr. Jennifer Teichman, hematology resident at the University of Toronto, said in an interview. Researchers said iron deficiency during pregnancy is associated with maternal fatigue, cognitive dysfunction, depression, low birth weight, and poor brain development of the child.
Dr. Teichman explained that if iron deficiency during pregnancy is identified early enough, doctors would have enough time to treat the condition with iron supplements before the patient goes into delivery. She also explained prenatal vitamins, which contain some iron, do not contain enough of the mineral to fix iron deficiency.
“One really important point is that the amount of iron contained in a prenatal vitamin is really low,” Dr. Teichman explained. “It’s enough to make up the difference of the additional iron that she needs to sustain her pregnancy, but it’s not enough to treat a woman who’s already got low iron going into pregnancy. So there’s a difference between a prenatal vitamin and true iron supplementation.”
Researchers also found that those who came from a household with a low annual income were even less likely to receive a ferritin test, which was a troubling finding since women of lower socioeconomic status are more likely to be iron deficient in pregnancy.
“[This] says something about how we as health care providers are contributing to this gap in care,” Dr. Teichman said. “Women of lower socioeconomic status sort of have a triple whammy: They’re more likely to be iron deficient, they’re less likely to have it diagnosed, and they’re less likely to have it corrected.”
Dr. Teichman and her colleagues took a unique approach by looking at isolated ferritin levels as opposed to complete blood counts, which is the typical screening for anemia in pregnancy, said Lissette Tanner, MD, MPH, FACOG, who was not involved with the study.
“Those who meet the criteria for anemia should be evaluated for the cause with initial suspicion for iron deficiency anemia, as that is the most common etiology,” said Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta.
The Centers for Disease Control and Prevention recommends screening for iron deficiency anemia in pregnant persons, in addition to universal iron supplementation to meet the iron requirements of pregnancy.
Additionally, the American College of Obstetricians and Gynecologists recommends that all pregnant persons be screened for anemia with a complete blood count in the first trimester and again between 24 and 28 weeks of pregnancy.
However, iron deficiency is completely missed by ACOG’s recommendations, said Michael Auerbach, MD, of the department of medicine, Georgetown University, Washington.
“They recommend a [complete blood count] on all presenting pregnant women, but they do not recommend iron parameters, including a ferritin test, unless the mother is anemic,” said Dr. Auerbach, who was not involved in the study. “I think those guidelines are in need of revision.”
Dr. Teichman hopes her team’s findings will motivate change in obstetric and hematologic guidelines that recommend routine prenatal testing.
“I think ferritin should be a part of routine prenatal testing,” Dr. Teichman said. “And I also think that patients need to be empowered to ask what their iron levels are in pregnancy and providers need to know what a normal iron level is.”
None of the experts interviewed for this story had financial conflicts of interest.
FROM BLOOD ADVANCES
Clinical genetic testing for skin disorders continues to advance
and families of pediatric patients to navigate the landscape.
“Testing options range from targeted variant testing and single-gene testing to exome and genome sequencing,” Gabriele Richard, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is not always easy to determine which testing is right.”
Increasingly, clinical genomic tests, including exome and genome sequencing, are used for patients with complex phenotypes, and possibly multiple disorders, who might have no diagnosis despite extensive prior testing, said Dr. Richard, medical director at Gaithersburg, Md.–based GeneDx., a molecular diagnostic laboratory that performs comprehensive testing for rare genetic disorders. These tests are also being used more for first-line testing in critically ill patients in the neonatal and pediatric intensive care units, and “have heralded a whole new era of gene and disease discovery,” she added.
Targeted variant testing is used for known familial variants, to test family members for carrier status and segregation, and to make a prenatal diagnosis, she said. Single-gene testing is available for most genes and has its place for conditions that can be clinically well-recognized, such as ichthyosis vulgaris, Darier disease, or Papillon-Lefèvre syndrome.
Specific tests for identifying gene deletions or duplications are exon-level microarrays, multiplex ligation-dependent probe amplification (MLPA), and chromosomal microarray analysis. “The latter has been successful in identifying diseases causing chromosomal abnormalities in over 10% of cases overall,” Dr. Richard said. An example of a skin disorder is X-linked ichthyosis caused by a deletion of the steroid sulfatase locus in more than 95% of affected males, she said.
“However, the current staple of molecular diagnostic testing is multigene next-generation sequencing (NGS) panels, which allow you to interrogate two to hundreds of genes concurrently, including sequencing and deletion duplication testing.” These tests are the most cost effective, she said, and are available for almost any genodermatosis or group of disorders with overlapping phenotypes, such as albinism or ichthyosis, epidermolysis bullosa and skin fragility, ectodermal dysplasia, or porphyria. According to Dr. Richard, the diagnostic outcomes of NGS panels mainly depend on test indication, panel size and gene curation, age of onset, and prevailing inheritance pattern of disorders.
Her recommended criteria for distinguishing the myriad of available NGS panels include checking gene content, technical sensitivity of sequencing and deletion/duplication analysis, quality of variant interpretation and reporting, turn-around time, and available familial follow-up testing. “If a family might consider future prenatal diagnosis, choose the lab that performs prenatal and diagnostic testing,” Dr. Richard said. “Equally important are client services such as ease of ordering, insurance coverage, and the ability to determine out-of-pocket cost to patients.”
Resources that enable consumers to compare panel content, methodology, turnaround time, and other parameters include the Genetic Testing Registry (GTR) operated by the National Center for Biotechnology Information, and Concert Genetics, a genetic testing company. The National Society of Genetic Counselors also offers a searchable database for finding a genetic counselor.
Exome sequencing includes the coding sequences of about 20,000 genes and has an average depth of 50 to about 150 reads. “It is a phenotype-driven test where only select variants are being reported fitting the phenotype,” Dr. Richard said. “The outcome of exome and genome sequencing much depends on optimization of bioinformatic pipelines and tools.” Besides small sequence variants, exome sequencing is able to identify a variety of different types of disease-causing variants, such as gene copy number variants seen in about 6% of positive cases, mosaicism, regions of homozygosity, uniparental disomy, and other unusual events and is cost effective.
Whole-genome sequencing, meanwhile, includes the entire genome, particularly noncoding regions, and has an average depth of more than 30 reads. “It’s based on single-molecule sequencing, has longer reads and more uniform coverage, compared to exome sequencing,” she said. “Higher cost, variant interpretation, and lack of coverage by payers are still presenting challenges for genome sequencing.” Genome sequencing can be done in a day or less.
According to diagnostic outcomes based on 280,000 individuals including 125,000 probands from GeneDx data, a definitive diagnosis was made in 26% of probands, of which 2.8% had more than one diagnostic finding and 1.8% had actionable secondary findings. In addition, 7% of the variants were found in candidate genes; 31% of probands had variants of uncertain significance, while 36% tested negative. “Nevertheless, the diagnostic yield of exome sequencing depends on the phenotype and cohort studied,” Dr. Richard continued.
At her company, she said, the highest positive rate is for multiple congenital anomalies (34%), skeletal system abnormalities (30%), and nervous system abnormalities (29%). Trio testing – the concurrent analysis of both biological parents and proband for all genes – “is a critical factor for success,” she added. “It not only improves the variant calling because we have three times the data and increases test sensitivity, it also provides more certain results, determines inheritance and allows for detection of parental mosaicism.”
According to Dr. Richard, trio testing has a one-third higher diagnostic rate than sequencing of the proband alone. Citing a published prospective study that compiled data from eight different exome- and genome-sequencing studies in critically ill neonates and children, trio testing made it possible to make a genetic diagnosis in up to 58% of children.
Whole-genome sequencing is estimated to have a 5%-10% higher diagnostic rate than exome sequencing. “However, we are still a ways away from using it as a routine diagnostic test for all test indications,” Dr. Richard said. “Automation, special bioinformatics algorithms and databases, and combination of genome sequencing with mRNA sequencing are being explored and built to further improve the diagnostic yield.”
Dr. Richard had no disclosures other than being an employee of GeneDx.
and families of pediatric patients to navigate the landscape.
“Testing options range from targeted variant testing and single-gene testing to exome and genome sequencing,” Gabriele Richard, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is not always easy to determine which testing is right.”
Increasingly, clinical genomic tests, including exome and genome sequencing, are used for patients with complex phenotypes, and possibly multiple disorders, who might have no diagnosis despite extensive prior testing, said Dr. Richard, medical director at Gaithersburg, Md.–based GeneDx., a molecular diagnostic laboratory that performs comprehensive testing for rare genetic disorders. These tests are also being used more for first-line testing in critically ill patients in the neonatal and pediatric intensive care units, and “have heralded a whole new era of gene and disease discovery,” she added.
Targeted variant testing is used for known familial variants, to test family members for carrier status and segregation, and to make a prenatal diagnosis, she said. Single-gene testing is available for most genes and has its place for conditions that can be clinically well-recognized, such as ichthyosis vulgaris, Darier disease, or Papillon-Lefèvre syndrome.
Specific tests for identifying gene deletions or duplications are exon-level microarrays, multiplex ligation-dependent probe amplification (MLPA), and chromosomal microarray analysis. “The latter has been successful in identifying diseases causing chromosomal abnormalities in over 10% of cases overall,” Dr. Richard said. An example of a skin disorder is X-linked ichthyosis caused by a deletion of the steroid sulfatase locus in more than 95% of affected males, she said.
“However, the current staple of molecular diagnostic testing is multigene next-generation sequencing (NGS) panels, which allow you to interrogate two to hundreds of genes concurrently, including sequencing and deletion duplication testing.” These tests are the most cost effective, she said, and are available for almost any genodermatosis or group of disorders with overlapping phenotypes, such as albinism or ichthyosis, epidermolysis bullosa and skin fragility, ectodermal dysplasia, or porphyria. According to Dr. Richard, the diagnostic outcomes of NGS panels mainly depend on test indication, panel size and gene curation, age of onset, and prevailing inheritance pattern of disorders.
Her recommended criteria for distinguishing the myriad of available NGS panels include checking gene content, technical sensitivity of sequencing and deletion/duplication analysis, quality of variant interpretation and reporting, turn-around time, and available familial follow-up testing. “If a family might consider future prenatal diagnosis, choose the lab that performs prenatal and diagnostic testing,” Dr. Richard said. “Equally important are client services such as ease of ordering, insurance coverage, and the ability to determine out-of-pocket cost to patients.”
Resources that enable consumers to compare panel content, methodology, turnaround time, and other parameters include the Genetic Testing Registry (GTR) operated by the National Center for Biotechnology Information, and Concert Genetics, a genetic testing company. The National Society of Genetic Counselors also offers a searchable database for finding a genetic counselor.
Exome sequencing includes the coding sequences of about 20,000 genes and has an average depth of 50 to about 150 reads. “It is a phenotype-driven test where only select variants are being reported fitting the phenotype,” Dr. Richard said. “The outcome of exome and genome sequencing much depends on optimization of bioinformatic pipelines and tools.” Besides small sequence variants, exome sequencing is able to identify a variety of different types of disease-causing variants, such as gene copy number variants seen in about 6% of positive cases, mosaicism, regions of homozygosity, uniparental disomy, and other unusual events and is cost effective.
Whole-genome sequencing, meanwhile, includes the entire genome, particularly noncoding regions, and has an average depth of more than 30 reads. “It’s based on single-molecule sequencing, has longer reads and more uniform coverage, compared to exome sequencing,” she said. “Higher cost, variant interpretation, and lack of coverage by payers are still presenting challenges for genome sequencing.” Genome sequencing can be done in a day or less.
According to diagnostic outcomes based on 280,000 individuals including 125,000 probands from GeneDx data, a definitive diagnosis was made in 26% of probands, of which 2.8% had more than one diagnostic finding and 1.8% had actionable secondary findings. In addition, 7% of the variants were found in candidate genes; 31% of probands had variants of uncertain significance, while 36% tested negative. “Nevertheless, the diagnostic yield of exome sequencing depends on the phenotype and cohort studied,” Dr. Richard continued.
At her company, she said, the highest positive rate is for multiple congenital anomalies (34%), skeletal system abnormalities (30%), and nervous system abnormalities (29%). Trio testing – the concurrent analysis of both biological parents and proband for all genes – “is a critical factor for success,” she added. “It not only improves the variant calling because we have three times the data and increases test sensitivity, it also provides more certain results, determines inheritance and allows for detection of parental mosaicism.”
According to Dr. Richard, trio testing has a one-third higher diagnostic rate than sequencing of the proband alone. Citing a published prospective study that compiled data from eight different exome- and genome-sequencing studies in critically ill neonates and children, trio testing made it possible to make a genetic diagnosis in up to 58% of children.
Whole-genome sequencing is estimated to have a 5%-10% higher diagnostic rate than exome sequencing. “However, we are still a ways away from using it as a routine diagnostic test for all test indications,” Dr. Richard said. “Automation, special bioinformatics algorithms and databases, and combination of genome sequencing with mRNA sequencing are being explored and built to further improve the diagnostic yield.”
Dr. Richard had no disclosures other than being an employee of GeneDx.
and families of pediatric patients to navigate the landscape.
“Testing options range from targeted variant testing and single-gene testing to exome and genome sequencing,” Gabriele Richard, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is not always easy to determine which testing is right.”
Increasingly, clinical genomic tests, including exome and genome sequencing, are used for patients with complex phenotypes, and possibly multiple disorders, who might have no diagnosis despite extensive prior testing, said Dr. Richard, medical director at Gaithersburg, Md.–based GeneDx., a molecular diagnostic laboratory that performs comprehensive testing for rare genetic disorders. These tests are also being used more for first-line testing in critically ill patients in the neonatal and pediatric intensive care units, and “have heralded a whole new era of gene and disease discovery,” she added.
Targeted variant testing is used for known familial variants, to test family members for carrier status and segregation, and to make a prenatal diagnosis, she said. Single-gene testing is available for most genes and has its place for conditions that can be clinically well-recognized, such as ichthyosis vulgaris, Darier disease, or Papillon-Lefèvre syndrome.
Specific tests for identifying gene deletions or duplications are exon-level microarrays, multiplex ligation-dependent probe amplification (MLPA), and chromosomal microarray analysis. “The latter has been successful in identifying diseases causing chromosomal abnormalities in over 10% of cases overall,” Dr. Richard said. An example of a skin disorder is X-linked ichthyosis caused by a deletion of the steroid sulfatase locus in more than 95% of affected males, she said.
“However, the current staple of molecular diagnostic testing is multigene next-generation sequencing (NGS) panels, which allow you to interrogate two to hundreds of genes concurrently, including sequencing and deletion duplication testing.” These tests are the most cost effective, she said, and are available for almost any genodermatosis or group of disorders with overlapping phenotypes, such as albinism or ichthyosis, epidermolysis bullosa and skin fragility, ectodermal dysplasia, or porphyria. According to Dr. Richard, the diagnostic outcomes of NGS panels mainly depend on test indication, panel size and gene curation, age of onset, and prevailing inheritance pattern of disorders.
Her recommended criteria for distinguishing the myriad of available NGS panels include checking gene content, technical sensitivity of sequencing and deletion/duplication analysis, quality of variant interpretation and reporting, turn-around time, and available familial follow-up testing. “If a family might consider future prenatal diagnosis, choose the lab that performs prenatal and diagnostic testing,” Dr. Richard said. “Equally important are client services such as ease of ordering, insurance coverage, and the ability to determine out-of-pocket cost to patients.”
Resources that enable consumers to compare panel content, methodology, turnaround time, and other parameters include the Genetic Testing Registry (GTR) operated by the National Center for Biotechnology Information, and Concert Genetics, a genetic testing company. The National Society of Genetic Counselors also offers a searchable database for finding a genetic counselor.
Exome sequencing includes the coding sequences of about 20,000 genes and has an average depth of 50 to about 150 reads. “It is a phenotype-driven test where only select variants are being reported fitting the phenotype,” Dr. Richard said. “The outcome of exome and genome sequencing much depends on optimization of bioinformatic pipelines and tools.” Besides small sequence variants, exome sequencing is able to identify a variety of different types of disease-causing variants, such as gene copy number variants seen in about 6% of positive cases, mosaicism, regions of homozygosity, uniparental disomy, and other unusual events and is cost effective.
Whole-genome sequencing, meanwhile, includes the entire genome, particularly noncoding regions, and has an average depth of more than 30 reads. “It’s based on single-molecule sequencing, has longer reads and more uniform coverage, compared to exome sequencing,” she said. “Higher cost, variant interpretation, and lack of coverage by payers are still presenting challenges for genome sequencing.” Genome sequencing can be done in a day or less.
According to diagnostic outcomes based on 280,000 individuals including 125,000 probands from GeneDx data, a definitive diagnosis was made in 26% of probands, of which 2.8% had more than one diagnostic finding and 1.8% had actionable secondary findings. In addition, 7% of the variants were found in candidate genes; 31% of probands had variants of uncertain significance, while 36% tested negative. “Nevertheless, the diagnostic yield of exome sequencing depends on the phenotype and cohort studied,” Dr. Richard continued.
At her company, she said, the highest positive rate is for multiple congenital anomalies (34%), skeletal system abnormalities (30%), and nervous system abnormalities (29%). Trio testing – the concurrent analysis of both biological parents and proband for all genes – “is a critical factor for success,” she added. “It not only improves the variant calling because we have three times the data and increases test sensitivity, it also provides more certain results, determines inheritance and allows for detection of parental mosaicism.”
According to Dr. Richard, trio testing has a one-third higher diagnostic rate than sequencing of the proband alone. Citing a published prospective study that compiled data from eight different exome- and genome-sequencing studies in critically ill neonates and children, trio testing made it possible to make a genetic diagnosis in up to 58% of children.
Whole-genome sequencing is estimated to have a 5%-10% higher diagnostic rate than exome sequencing. “However, we are still a ways away from using it as a routine diagnostic test for all test indications,” Dr. Richard said. “Automation, special bioinformatics algorithms and databases, and combination of genome sequencing with mRNA sequencing are being explored and built to further improve the diagnostic yield.”
Dr. Richard had no disclosures other than being an employee of GeneDx.
FROM SPD 2021
New gene variant found for hereditary bleeding disorder
Clinical symptoms in a 19-year-old Vietnamese woman who experienced several life-threatening bleeding events, including ovarian hemorrhage, led to the discovery of a novel gene variant causing her bleeding disorder.
Blood analysis of the patients showed decreased fibrinogen level with “markedly elevated fibrinogen/fibrin degradation products and D-dimer levels.” Attempts to treat the patient with hemostatic surgery, administration of several medications, including nafamostat mesylate, tranexamic acid, and unfractionated heparin, produced no correction of her coagulation abnormalities, and the patient experienced repeated hemorrhagic events, according to researchers from the Tokyo Saiseikai Central Hospital, Japan, and colleagues.
However, the researchers found that treatment with recombinant human thrombomodulin (rhTM) remarkably improved the patient’s pathophysiology, according to the results of a case study reported in Blood Advances.
Genetic analysis
Upon screening and sequencing of the patient’s thrombomodulin gene, a previously unreported homozygous variation, c.793T>A (p.Cys265Ser) was discovered. Under normal circumstances, the Cys265 residue forms one of three disulfide bonds in the epidermal growth factor (EGF)-like domain 1 of thrombomoduliin (TM), according to the researchers.
However, transient expression analysis of the patient’s mutation using COS-1 cells demonstrated markedly reduced expression of TM-Cys265Ser on the plasma membrane relative to wild-type TM. The TM-Cys265Ser mutant was intracellularly degraded, probably due to EGF-like domain 1 misfolding, according to the researchers and the reduced expression of TM on the endothelial cell membrane may be responsible for the disseminated intravascular coagulation-like symptoms observed in the patient, the speculated.
“The clinical symptoms of the patient in this study were characterized by recurrent hemorrhage, indicating that TM-C265S mainly causes hyperfibrinolysis rather than hypercoagulation. The C265S mutation may disrupt the timely and delicate balance between coagulation and fibrinolysis,” the researchers suggested.
The authors reported that they had no conflicts of interest.
Clinical symptoms in a 19-year-old Vietnamese woman who experienced several life-threatening bleeding events, including ovarian hemorrhage, led to the discovery of a novel gene variant causing her bleeding disorder.
Blood analysis of the patients showed decreased fibrinogen level with “markedly elevated fibrinogen/fibrin degradation products and D-dimer levels.” Attempts to treat the patient with hemostatic surgery, administration of several medications, including nafamostat mesylate, tranexamic acid, and unfractionated heparin, produced no correction of her coagulation abnormalities, and the patient experienced repeated hemorrhagic events, according to researchers from the Tokyo Saiseikai Central Hospital, Japan, and colleagues.
However, the researchers found that treatment with recombinant human thrombomodulin (rhTM) remarkably improved the patient’s pathophysiology, according to the results of a case study reported in Blood Advances.
Genetic analysis
Upon screening and sequencing of the patient’s thrombomodulin gene, a previously unreported homozygous variation, c.793T>A (p.Cys265Ser) was discovered. Under normal circumstances, the Cys265 residue forms one of three disulfide bonds in the epidermal growth factor (EGF)-like domain 1 of thrombomoduliin (TM), according to the researchers.
However, transient expression analysis of the patient’s mutation using COS-1 cells demonstrated markedly reduced expression of TM-Cys265Ser on the plasma membrane relative to wild-type TM. The TM-Cys265Ser mutant was intracellularly degraded, probably due to EGF-like domain 1 misfolding, according to the researchers and the reduced expression of TM on the endothelial cell membrane may be responsible for the disseminated intravascular coagulation-like symptoms observed in the patient, the speculated.
“The clinical symptoms of the patient in this study were characterized by recurrent hemorrhage, indicating that TM-C265S mainly causes hyperfibrinolysis rather than hypercoagulation. The C265S mutation may disrupt the timely and delicate balance between coagulation and fibrinolysis,” the researchers suggested.
The authors reported that they had no conflicts of interest.
Clinical symptoms in a 19-year-old Vietnamese woman who experienced several life-threatening bleeding events, including ovarian hemorrhage, led to the discovery of a novel gene variant causing her bleeding disorder.
Blood analysis of the patients showed decreased fibrinogen level with “markedly elevated fibrinogen/fibrin degradation products and D-dimer levels.” Attempts to treat the patient with hemostatic surgery, administration of several medications, including nafamostat mesylate, tranexamic acid, and unfractionated heparin, produced no correction of her coagulation abnormalities, and the patient experienced repeated hemorrhagic events, according to researchers from the Tokyo Saiseikai Central Hospital, Japan, and colleagues.
However, the researchers found that treatment with recombinant human thrombomodulin (rhTM) remarkably improved the patient’s pathophysiology, according to the results of a case study reported in Blood Advances.
Genetic analysis
Upon screening and sequencing of the patient’s thrombomodulin gene, a previously unreported homozygous variation, c.793T>A (p.Cys265Ser) was discovered. Under normal circumstances, the Cys265 residue forms one of three disulfide bonds in the epidermal growth factor (EGF)-like domain 1 of thrombomoduliin (TM), according to the researchers.
However, transient expression analysis of the patient’s mutation using COS-1 cells demonstrated markedly reduced expression of TM-Cys265Ser on the plasma membrane relative to wild-type TM. The TM-Cys265Ser mutant was intracellularly degraded, probably due to EGF-like domain 1 misfolding, according to the researchers and the reduced expression of TM on the endothelial cell membrane may be responsible for the disseminated intravascular coagulation-like symptoms observed in the patient, the speculated.
“The clinical symptoms of the patient in this study were characterized by recurrent hemorrhage, indicating that TM-C265S mainly causes hyperfibrinolysis rather than hypercoagulation. The C265S mutation may disrupt the timely and delicate balance between coagulation and fibrinolysis,” the researchers suggested.
The authors reported that they had no conflicts of interest.
FROM BLOOD ADVANCES