User login
How an ‘ad hoc’ hospitalist model evolved during India’s COVID surge
Hospital administrators recognize the efficiencies
A year after the start of the COVID-19 pandemic, as the United States was getting a reprieve in new cases from its winter surge, the opposite was happening in the rest of the world. In India, a deadly second wave hit, crippling the health care system in the country for months.
Yugandhar Bhatt, MBBS, MD, a consultant pulmonologist with Yashoda Hospital–Malakpet in Hyderabad, India, told this news organization that someone looking at his hospital before the pandemic – a 400-bed multispecialty care unit – would see patients being treated for respiratory failure secondary to exacerbation of chronic obstructive pulmonary disease, bronchial asthma, community-acquired pneumonia, and heart failure. About 30-40 patients per day were treated on an outpatient basis, and more than 30 people were admitted as inpatients.
“After [the] COVID-19 surge, our hospital totally divided into COVID and non-COVID [wards], in which COVID patients occupied 70% of [the] total,” he said. About half of COVID-19 patients were in the ICU, with half of those patients requiring supplemental oxygen.
During the first wave in India, which lasted from May to December 2020, 50% of patients who were intubated were discharged. The percentage of extubated patients decreased to 20% in the second wave, Dr. Bhatt said.
The death toll during the second wave of COVID-19 cases was unlike anything India has seen previously. Between March 1 and June 29, 2021, an estimated 19.24 million individuals were newly infected with COVID-19 and 241,206 patients died, according to Our World in Data, a project of the Global Change Data Lab. When the second wave peaked on May 22, more than 4,000 people were dying each day.
“All hospitals [in India] were treating COVID-19 more than any other acute or chronic disease,” Ramesh Adhikari, MD, MS, SFHM, a hospitalist with Franciscan Health in Lafayette, Ind., said in an interview.
Challenges arose in treating COVID-19 in India that ran counter to how medicine was usually performed. Physicians were seeing more inpatient cases than usual – and more patients in general. The change, Dr. Adhikari said, forced health care providers to think outside the box.
An ‘on-the-fly’ hospitalist model
Patients in India access health care by visiting a hospital or primary health center and then are referred out to consultants – specialist doctors – if needed. While India has universal health coverage, it is a multi-payer system that includes approximately 37% of the population covered under the government plan, a large number of private health care facilities and no caps on cost-sharing for the patient. Initiatives like Rashtriya Swasthya Bima Yojana in 2008 and Ayushman Bharat-Pradhan Mantri Jan Arogya Yojana in 2018 have attempted to close the gap and raise the number of lower-income individuals in India covered under the government plan and reduce out-of-pocket spending. Out-of-pocket payments still consist of about 70% of total health expenditures, according to the Commonwealth Fund.
“There is not much scope for a hospitalist because it’s so cash driven,” Shyam Odeti, MD, SFHM, section chief, hospital medicine, at the Carilion Clinic in Roanoke, Va., said in an interview. “For a hospitalist, there is no urgency in getting them out of the hospital. There was no need for much efficiency before.”
The first issue during the second wave was figuring out which consultants would care for COVID-19 patients. As there is no dedicated specialty for infectious disease in India, the responsibilities fell to internists and critical care medicine consultants who volunteered. Both are considered small specialties in India. They became “makeshift hospitalists” who learned as they went and became the experts in COVID-19 care, treating their own patients while making themselves available for consultations, Dr. Odeti said.
While no official hospital medicine model in India exists like in the United States, the second COVID-19 surge caused these consultants to begin thinking like hospitalists. Tenets of hospital medicine – like team-based treatment across specialties – arose out of necessity during the crisis. “They were trying to implement a hospitalist model because that’s the only way they could treat COVID-19,” said Dr. Adhikari, an editorial advisory board member for the Hospitalist.
“Even in the U.S. when we started the hospitalist model, it started out of necessity. It’s a combination of creating efficiencies and improving quality,” Dr. Odeti said. “It’s the same thing in India. It’s borne of necessity, but it was [done] at a rapid pace.”
Problems with patient flow
The next issue was triaging patients in the hospital based on COVID-19 severity. When the second wave began, hospitals in India ran out of beds and experienced staff shortages like in many countries. But this situation “was unusual for the health system,” according to Dr. Odeti, who is also an editorial board member for the Hospitalist.
“We never had that issue. There were so many patients wanting to come to the hospital, and so there was this rush.” There was no process to triage patients to determine who needed to stay. “Everybody got put into the hospital,” he said.
Once it was determined who would take care of patients with COVID-19, access to supplies became the primary problem, Dr. Adhikari explained. Lack of oxygen, ventilators, and critical medicines like the antiviral drug remdesivir were and continue to be in short supply. “I had friends who [said] they could not admit patients because they were worried if their oxygen supply [went] low in the middle of the night. They will treat the patients who were already admitted versus taking new patients. That had caused problems for the administrators,” Dr. Adhikari added.
It is also a source of additional stress for the physicians. Where patients flow through a hospital medicine model in the United States, a system that might include case managers, social workers, pharmacists, physician advocates, and other professionals to keep a patient’s care on track, the physician is the go-to person in India for patient care. While physicians provide access to medications and remain available to a patient’s family, those duties become much harder when caring for a greater number of patients during the pandemic. “That has led to some unrealistic expectations among the patients,” Dr. Adhikari said.
Dr. Bhatt said “more than half” of a physician’s time in India is spent counseling patients on concerns about COVID-19. “Awareness about the disease is limited from the patient and patient’s family perspective, as [there is] too much apprehension toward the nature of [the] disease,” he added. “Theoretical discussions collected from social media” obstruct the physician from executing his or her duties.
Physicians in India have had to contend with physical violence from patients and individuals on the street, Dr. Adhikari added. Workplace violence was already a concern – for years, the Indian Medical Association has cited a statistic that 75% of doctors in India have experienced violence at work (Indian J Psychiatry. 2019 Apr;61[Suppl 4]:S782-5). But the threat of violence against physicians has sharply increased during the COVID-19 pandemic. Disruptions to daily life through lockdowns “made people fearful, anxious, and sometimes they have found it difficult to access emergency treatment,” according to a letter published by Karthikeyan Iyengar and colleagues in the Postgraduate Medical Journal. In response to the restlessness, irritation, and despair resulting from hospitals closing their doors, “people have shown their frustration by verbally abusing and threatening to physically assault doctors and other health care workers,” the authors wrote.
A telemedicine boon in India
Back in the United States, hospitalists with family and friends in India were trying to figure out how to help. Some were working through the day, only to answer calls and WhatsApp messages from loved ones at night. “Everyone knows a physician or someone who’s your colleague, who owns a hospital or runs a hospital, or one of the family members is sick,” Dr. Adhikari said.
These U.S.-based hospitalists were burning the candle at both ends, helping with the pandemic in both countries. Physicians in India were posing questions to U.S. colleagues who they saw as having the most recent evidence for COVID-19 treatment. Out of the 180 physicians he trained with in India, Dr. Odeti said 110 of the physicians were in a large WhatsApp group chat that was constantly exchanging messages and serving as “kind of a friendly support group.”
In Dr. Odeti’s group chat, physicians helped one another find hospital beds for patients who reached out to them. “The first couple of weeks, there was no proper way for people to know where [patients] were based. There was no way to find if this hospital had a bed, so they reached out to any doctors they knew,” he said.
While he said it was emotionally draining, “at the same time, we felt a responsibility toward colleagues in India,” Dr. Odeti said, noting that as COVID-19 cases have decreased in India, the requests have been less frequent.
Because of concerns about traveling to India during the pandemic while on a J-1, H-1B, or other visa with the United States, directly helping friends and family in India seemed out of reach. But many hospitalists of Indian origin instead turned to telemedicine to help their colleagues. Telemedicine had already been steadily growing in India, but was accelerated by the pandemic. The current ratio of doctors to patients in India is 0.62 to 1,000 – lower than recommendations from the World Health Organization. That makes telemedicine a unique opportunity for one physician in India to reach many patients regardless of location.
Dr. Adhikari said he helped out his colleagues in India by performing consults for their patients. “They were just worried because they did not ‘know where to go, or what to get,” he said. “I was treating more patients in India than I was actually treating here.”
In March 2020, the Indian Ministry of Health and Family Welfare released telemedicine practice guidelines for the country, which relaxed regulations on privacy requirements and has been credited in part for giving telemedicine an additional boost during the pandemic. “That makes it easy for people to reach out but also has its own problems,” Dr. Adhikari said.
Monitoring of milder COVID-19 cases that don’t require hospitalization can be performed by a nurse who calls every few hours to check on a patient, make recommendations, and text treatment plans. “The telemedicine platforms are being adopted really fast,” Dr. Adhikari said. “The platforms were built in no time.”
According to NewZoo, a games market data analytics company, India has 345.9 million smartphone users as of 2019 – the second highest number of users in the world after China. Dr. Odeti said he believes telemedicine will be widely adopted.
“In India, they are very proactive in accepting these kinds of methods, so I’m sure they will,” he said. “Governments were trying to do it before the pandemic, because access to care is a problem in India. There are villages which are very, very remote.”
Reversion to old systems
After the peak in late May, new COVID-19 cases in India began to decrease, and the second wave waned on a national level. Hospitals began to get the supplies they needed, beds are available, and patients aren’t as sick as before, according to Dr. Adhikari. The federal government has begun issuing supplies to patients in each state, including COVID-19 vaccines. “The peak for the second wave is gone,” he said.
What remains is a group of physicians trained in how to triage patients and create efficiencies in a hospital setting. Could those skills be put to use elsewhere in India after the pandemic?
According to Dr. Bhatt, the patient care model is likely to revert to the system that existed before. “Whatever the changes, interims of bed occupancy, cost of ICU will be temporary [and] will change to normal,” he said. “But awareness about masks [and] sanitizing methods will be permanent.”
Dr. Adhikari believes that not utilizing the skills of newly minted hospitalists in India would be a missed opportunity. “This is a silver lining from COVID-19, that hospital medicine plays a vital role in the sickest patients, whether it is in India or the U.S. or anywhere,” he said. “I think the model of hospital medicine should be adopted. It’s not: ‘Should it really be adopted or not?’ It should be. There is a huge potential in doing inpatient coordinated [care], having people dedicated in the hospital.”
There are tangible benefits to creating efficiencies in India’s health system, Dr. Odeti said. Length of stay for sicker patients “was much longer” at 10-14 days during the second wave, compared with the United States, before lowering to around 5 days. “These hospitals right now are learning the efficient ways of doing it: when to send [patients] out, how to send them out, how to [perform] service-based practices, creating processes which were nonexistent before.”
While he doesn’t personally believe physicians will adopt a full-fledged hospitalist model unless the payer structure in India changes, “these people are at an advantage with this extra set of skills,” he said. “I think all the knowledge that these people have are going to come in handy.”
Opportunities for growth
Dr. Odeti sees the potential for the hospitalist model to grow in India – if not into its own specialty, then in how critical care consultants handle sicker patients and handoffs.
“The critical care clinician cannot keep the patient from the time they are admitted to the ICU until the discharge, so there will be a need for the transition,” Dr. Odeti said. “In the past, there were not many capabilities in Indian health systems to take care of these extremely sick patients, and now it is evolving. I think that is one more thing that will help.”
Dr. Adhikari said hospital systems in India are beginning to realize how having dedicated hospital physicians could benefit them. In India, “if you’re sick, you go to your doctor, you get treated and you disappear,” he said. The next time, you may see the same doctor or a completely different doctor. “There’s no system there, so it’s really hard for hospital medicine as such because patients, when they are very sick, they just come to the ER. They’re not followed by their primary care.”
Anecdotally, Dr. Odeti sees patients already adapting to having access to a physician for asking questions normally answered by primary care physicians. “I think primary care will come into play,” he said. “When I was doing a Zoom call for patients, they were asking me questions about sciatica. I think they are getting comfortable with this technology.”
A hospitalist model could even be applied to specific diseases with a large population of patients. Hospital administrators “have seen this for the first time, how efficient it could be if they had their own hospitalists and actually run it. So that’s the part that has crossed their minds,” Dr. Adhikari said. “How they will apply it going forward, other than during the COVID-19 pandemic, depends on the size of the hospital and the volume of the patients for a particular disease.”
“You can see in certain areas there is large growth for hospital medicine. But to rise to the level of the United States and how we do it, India needs bigger health systems to adopt the model,” Dr. Adhikari said.
Hospital administrators recognize the efficiencies
Hospital administrators recognize the efficiencies
A year after the start of the COVID-19 pandemic, as the United States was getting a reprieve in new cases from its winter surge, the opposite was happening in the rest of the world. In India, a deadly second wave hit, crippling the health care system in the country for months.
Yugandhar Bhatt, MBBS, MD, a consultant pulmonologist with Yashoda Hospital–Malakpet in Hyderabad, India, told this news organization that someone looking at his hospital before the pandemic – a 400-bed multispecialty care unit – would see patients being treated for respiratory failure secondary to exacerbation of chronic obstructive pulmonary disease, bronchial asthma, community-acquired pneumonia, and heart failure. About 30-40 patients per day were treated on an outpatient basis, and more than 30 people were admitted as inpatients.
“After [the] COVID-19 surge, our hospital totally divided into COVID and non-COVID [wards], in which COVID patients occupied 70% of [the] total,” he said. About half of COVID-19 patients were in the ICU, with half of those patients requiring supplemental oxygen.
During the first wave in India, which lasted from May to December 2020, 50% of patients who were intubated were discharged. The percentage of extubated patients decreased to 20% in the second wave, Dr. Bhatt said.
The death toll during the second wave of COVID-19 cases was unlike anything India has seen previously. Between March 1 and June 29, 2021, an estimated 19.24 million individuals were newly infected with COVID-19 and 241,206 patients died, according to Our World in Data, a project of the Global Change Data Lab. When the second wave peaked on May 22, more than 4,000 people were dying each day.
“All hospitals [in India] were treating COVID-19 more than any other acute or chronic disease,” Ramesh Adhikari, MD, MS, SFHM, a hospitalist with Franciscan Health in Lafayette, Ind., said in an interview.
Challenges arose in treating COVID-19 in India that ran counter to how medicine was usually performed. Physicians were seeing more inpatient cases than usual – and more patients in general. The change, Dr. Adhikari said, forced health care providers to think outside the box.
An ‘on-the-fly’ hospitalist model
Patients in India access health care by visiting a hospital or primary health center and then are referred out to consultants – specialist doctors – if needed. While India has universal health coverage, it is a multi-payer system that includes approximately 37% of the population covered under the government plan, a large number of private health care facilities and no caps on cost-sharing for the patient. Initiatives like Rashtriya Swasthya Bima Yojana in 2008 and Ayushman Bharat-Pradhan Mantri Jan Arogya Yojana in 2018 have attempted to close the gap and raise the number of lower-income individuals in India covered under the government plan and reduce out-of-pocket spending. Out-of-pocket payments still consist of about 70% of total health expenditures, according to the Commonwealth Fund.
“There is not much scope for a hospitalist because it’s so cash driven,” Shyam Odeti, MD, SFHM, section chief, hospital medicine, at the Carilion Clinic in Roanoke, Va., said in an interview. “For a hospitalist, there is no urgency in getting them out of the hospital. There was no need for much efficiency before.”
The first issue during the second wave was figuring out which consultants would care for COVID-19 patients. As there is no dedicated specialty for infectious disease in India, the responsibilities fell to internists and critical care medicine consultants who volunteered. Both are considered small specialties in India. They became “makeshift hospitalists” who learned as they went and became the experts in COVID-19 care, treating their own patients while making themselves available for consultations, Dr. Odeti said.
While no official hospital medicine model in India exists like in the United States, the second COVID-19 surge caused these consultants to begin thinking like hospitalists. Tenets of hospital medicine – like team-based treatment across specialties – arose out of necessity during the crisis. “They were trying to implement a hospitalist model because that’s the only way they could treat COVID-19,” said Dr. Adhikari, an editorial advisory board member for the Hospitalist.
“Even in the U.S. when we started the hospitalist model, it started out of necessity. It’s a combination of creating efficiencies and improving quality,” Dr. Odeti said. “It’s the same thing in India. It’s borne of necessity, but it was [done] at a rapid pace.”
Problems with patient flow
The next issue was triaging patients in the hospital based on COVID-19 severity. When the second wave began, hospitals in India ran out of beds and experienced staff shortages like in many countries. But this situation “was unusual for the health system,” according to Dr. Odeti, who is also an editorial board member for the Hospitalist.
“We never had that issue. There were so many patients wanting to come to the hospital, and so there was this rush.” There was no process to triage patients to determine who needed to stay. “Everybody got put into the hospital,” he said.
Once it was determined who would take care of patients with COVID-19, access to supplies became the primary problem, Dr. Adhikari explained. Lack of oxygen, ventilators, and critical medicines like the antiviral drug remdesivir were and continue to be in short supply. “I had friends who [said] they could not admit patients because they were worried if their oxygen supply [went] low in the middle of the night. They will treat the patients who were already admitted versus taking new patients. That had caused problems for the administrators,” Dr. Adhikari added.
It is also a source of additional stress for the physicians. Where patients flow through a hospital medicine model in the United States, a system that might include case managers, social workers, pharmacists, physician advocates, and other professionals to keep a patient’s care on track, the physician is the go-to person in India for patient care. While physicians provide access to medications and remain available to a patient’s family, those duties become much harder when caring for a greater number of patients during the pandemic. “That has led to some unrealistic expectations among the patients,” Dr. Adhikari said.
Dr. Bhatt said “more than half” of a physician’s time in India is spent counseling patients on concerns about COVID-19. “Awareness about the disease is limited from the patient and patient’s family perspective, as [there is] too much apprehension toward the nature of [the] disease,” he added. “Theoretical discussions collected from social media” obstruct the physician from executing his or her duties.
Physicians in India have had to contend with physical violence from patients and individuals on the street, Dr. Adhikari added. Workplace violence was already a concern – for years, the Indian Medical Association has cited a statistic that 75% of doctors in India have experienced violence at work (Indian J Psychiatry. 2019 Apr;61[Suppl 4]:S782-5). But the threat of violence against physicians has sharply increased during the COVID-19 pandemic. Disruptions to daily life through lockdowns “made people fearful, anxious, and sometimes they have found it difficult to access emergency treatment,” according to a letter published by Karthikeyan Iyengar and colleagues in the Postgraduate Medical Journal. In response to the restlessness, irritation, and despair resulting from hospitals closing their doors, “people have shown their frustration by verbally abusing and threatening to physically assault doctors and other health care workers,” the authors wrote.
A telemedicine boon in India
Back in the United States, hospitalists with family and friends in India were trying to figure out how to help. Some were working through the day, only to answer calls and WhatsApp messages from loved ones at night. “Everyone knows a physician or someone who’s your colleague, who owns a hospital or runs a hospital, or one of the family members is sick,” Dr. Adhikari said.
These U.S.-based hospitalists were burning the candle at both ends, helping with the pandemic in both countries. Physicians in India were posing questions to U.S. colleagues who they saw as having the most recent evidence for COVID-19 treatment. Out of the 180 physicians he trained with in India, Dr. Odeti said 110 of the physicians were in a large WhatsApp group chat that was constantly exchanging messages and serving as “kind of a friendly support group.”
In Dr. Odeti’s group chat, physicians helped one another find hospital beds for patients who reached out to them. “The first couple of weeks, there was no proper way for people to know where [patients] were based. There was no way to find if this hospital had a bed, so they reached out to any doctors they knew,” he said.
While he said it was emotionally draining, “at the same time, we felt a responsibility toward colleagues in India,” Dr. Odeti said, noting that as COVID-19 cases have decreased in India, the requests have been less frequent.
Because of concerns about traveling to India during the pandemic while on a J-1, H-1B, or other visa with the United States, directly helping friends and family in India seemed out of reach. But many hospitalists of Indian origin instead turned to telemedicine to help their colleagues. Telemedicine had already been steadily growing in India, but was accelerated by the pandemic. The current ratio of doctors to patients in India is 0.62 to 1,000 – lower than recommendations from the World Health Organization. That makes telemedicine a unique opportunity for one physician in India to reach many patients regardless of location.
Dr. Adhikari said he helped out his colleagues in India by performing consults for their patients. “They were just worried because they did not ‘know where to go, or what to get,” he said. “I was treating more patients in India than I was actually treating here.”
In March 2020, the Indian Ministry of Health and Family Welfare released telemedicine practice guidelines for the country, which relaxed regulations on privacy requirements and has been credited in part for giving telemedicine an additional boost during the pandemic. “That makes it easy for people to reach out but also has its own problems,” Dr. Adhikari said.
Monitoring of milder COVID-19 cases that don’t require hospitalization can be performed by a nurse who calls every few hours to check on a patient, make recommendations, and text treatment plans. “The telemedicine platforms are being adopted really fast,” Dr. Adhikari said. “The platforms were built in no time.”
According to NewZoo, a games market data analytics company, India has 345.9 million smartphone users as of 2019 – the second highest number of users in the world after China. Dr. Odeti said he believes telemedicine will be widely adopted.
“In India, they are very proactive in accepting these kinds of methods, so I’m sure they will,” he said. “Governments were trying to do it before the pandemic, because access to care is a problem in India. There are villages which are very, very remote.”
Reversion to old systems
After the peak in late May, new COVID-19 cases in India began to decrease, and the second wave waned on a national level. Hospitals began to get the supplies they needed, beds are available, and patients aren’t as sick as before, according to Dr. Adhikari. The federal government has begun issuing supplies to patients in each state, including COVID-19 vaccines. “The peak for the second wave is gone,” he said.
What remains is a group of physicians trained in how to triage patients and create efficiencies in a hospital setting. Could those skills be put to use elsewhere in India after the pandemic?
According to Dr. Bhatt, the patient care model is likely to revert to the system that existed before. “Whatever the changes, interims of bed occupancy, cost of ICU will be temporary [and] will change to normal,” he said. “But awareness about masks [and] sanitizing methods will be permanent.”
Dr. Adhikari believes that not utilizing the skills of newly minted hospitalists in India would be a missed opportunity. “This is a silver lining from COVID-19, that hospital medicine plays a vital role in the sickest patients, whether it is in India or the U.S. or anywhere,” he said. “I think the model of hospital medicine should be adopted. It’s not: ‘Should it really be adopted or not?’ It should be. There is a huge potential in doing inpatient coordinated [care], having people dedicated in the hospital.”
There are tangible benefits to creating efficiencies in India’s health system, Dr. Odeti said. Length of stay for sicker patients “was much longer” at 10-14 days during the second wave, compared with the United States, before lowering to around 5 days. “These hospitals right now are learning the efficient ways of doing it: when to send [patients] out, how to send them out, how to [perform] service-based practices, creating processes which were nonexistent before.”
While he doesn’t personally believe physicians will adopt a full-fledged hospitalist model unless the payer structure in India changes, “these people are at an advantage with this extra set of skills,” he said. “I think all the knowledge that these people have are going to come in handy.”
Opportunities for growth
Dr. Odeti sees the potential for the hospitalist model to grow in India – if not into its own specialty, then in how critical care consultants handle sicker patients and handoffs.
“The critical care clinician cannot keep the patient from the time they are admitted to the ICU until the discharge, so there will be a need for the transition,” Dr. Odeti said. “In the past, there were not many capabilities in Indian health systems to take care of these extremely sick patients, and now it is evolving. I think that is one more thing that will help.”
Dr. Adhikari said hospital systems in India are beginning to realize how having dedicated hospital physicians could benefit them. In India, “if you’re sick, you go to your doctor, you get treated and you disappear,” he said. The next time, you may see the same doctor or a completely different doctor. “There’s no system there, so it’s really hard for hospital medicine as such because patients, when they are very sick, they just come to the ER. They’re not followed by their primary care.”
Anecdotally, Dr. Odeti sees patients already adapting to having access to a physician for asking questions normally answered by primary care physicians. “I think primary care will come into play,” he said. “When I was doing a Zoom call for patients, they were asking me questions about sciatica. I think they are getting comfortable with this technology.”
A hospitalist model could even be applied to specific diseases with a large population of patients. Hospital administrators “have seen this for the first time, how efficient it could be if they had their own hospitalists and actually run it. So that’s the part that has crossed their minds,” Dr. Adhikari said. “How they will apply it going forward, other than during the COVID-19 pandemic, depends on the size of the hospital and the volume of the patients for a particular disease.”
“You can see in certain areas there is large growth for hospital medicine. But to rise to the level of the United States and how we do it, India needs bigger health systems to adopt the model,” Dr. Adhikari said.
A year after the start of the COVID-19 pandemic, as the United States was getting a reprieve in new cases from its winter surge, the opposite was happening in the rest of the world. In India, a deadly second wave hit, crippling the health care system in the country for months.
Yugandhar Bhatt, MBBS, MD, a consultant pulmonologist with Yashoda Hospital–Malakpet in Hyderabad, India, told this news organization that someone looking at his hospital before the pandemic – a 400-bed multispecialty care unit – would see patients being treated for respiratory failure secondary to exacerbation of chronic obstructive pulmonary disease, bronchial asthma, community-acquired pneumonia, and heart failure. About 30-40 patients per day were treated on an outpatient basis, and more than 30 people were admitted as inpatients.
“After [the] COVID-19 surge, our hospital totally divided into COVID and non-COVID [wards], in which COVID patients occupied 70% of [the] total,” he said. About half of COVID-19 patients were in the ICU, with half of those patients requiring supplemental oxygen.
During the first wave in India, which lasted from May to December 2020, 50% of patients who were intubated were discharged. The percentage of extubated patients decreased to 20% in the second wave, Dr. Bhatt said.
The death toll during the second wave of COVID-19 cases was unlike anything India has seen previously. Between March 1 and June 29, 2021, an estimated 19.24 million individuals were newly infected with COVID-19 and 241,206 patients died, according to Our World in Data, a project of the Global Change Data Lab. When the second wave peaked on May 22, more than 4,000 people were dying each day.
“All hospitals [in India] were treating COVID-19 more than any other acute or chronic disease,” Ramesh Adhikari, MD, MS, SFHM, a hospitalist with Franciscan Health in Lafayette, Ind., said in an interview.
Challenges arose in treating COVID-19 in India that ran counter to how medicine was usually performed. Physicians were seeing more inpatient cases than usual – and more patients in general. The change, Dr. Adhikari said, forced health care providers to think outside the box.
An ‘on-the-fly’ hospitalist model
Patients in India access health care by visiting a hospital or primary health center and then are referred out to consultants – specialist doctors – if needed. While India has universal health coverage, it is a multi-payer system that includes approximately 37% of the population covered under the government plan, a large number of private health care facilities and no caps on cost-sharing for the patient. Initiatives like Rashtriya Swasthya Bima Yojana in 2008 and Ayushman Bharat-Pradhan Mantri Jan Arogya Yojana in 2018 have attempted to close the gap and raise the number of lower-income individuals in India covered under the government plan and reduce out-of-pocket spending. Out-of-pocket payments still consist of about 70% of total health expenditures, according to the Commonwealth Fund.
“There is not much scope for a hospitalist because it’s so cash driven,” Shyam Odeti, MD, SFHM, section chief, hospital medicine, at the Carilion Clinic in Roanoke, Va., said in an interview. “For a hospitalist, there is no urgency in getting them out of the hospital. There was no need for much efficiency before.”
The first issue during the second wave was figuring out which consultants would care for COVID-19 patients. As there is no dedicated specialty for infectious disease in India, the responsibilities fell to internists and critical care medicine consultants who volunteered. Both are considered small specialties in India. They became “makeshift hospitalists” who learned as they went and became the experts in COVID-19 care, treating their own patients while making themselves available for consultations, Dr. Odeti said.
While no official hospital medicine model in India exists like in the United States, the second COVID-19 surge caused these consultants to begin thinking like hospitalists. Tenets of hospital medicine – like team-based treatment across specialties – arose out of necessity during the crisis. “They were trying to implement a hospitalist model because that’s the only way they could treat COVID-19,” said Dr. Adhikari, an editorial advisory board member for the Hospitalist.
“Even in the U.S. when we started the hospitalist model, it started out of necessity. It’s a combination of creating efficiencies and improving quality,” Dr. Odeti said. “It’s the same thing in India. It’s borne of necessity, but it was [done] at a rapid pace.”
Problems with patient flow
The next issue was triaging patients in the hospital based on COVID-19 severity. When the second wave began, hospitals in India ran out of beds and experienced staff shortages like in many countries. But this situation “was unusual for the health system,” according to Dr. Odeti, who is also an editorial board member for the Hospitalist.
“We never had that issue. There were so many patients wanting to come to the hospital, and so there was this rush.” There was no process to triage patients to determine who needed to stay. “Everybody got put into the hospital,” he said.
Once it was determined who would take care of patients with COVID-19, access to supplies became the primary problem, Dr. Adhikari explained. Lack of oxygen, ventilators, and critical medicines like the antiviral drug remdesivir were and continue to be in short supply. “I had friends who [said] they could not admit patients because they were worried if their oxygen supply [went] low in the middle of the night. They will treat the patients who were already admitted versus taking new patients. That had caused problems for the administrators,” Dr. Adhikari added.
It is also a source of additional stress for the physicians. Where patients flow through a hospital medicine model in the United States, a system that might include case managers, social workers, pharmacists, physician advocates, and other professionals to keep a patient’s care on track, the physician is the go-to person in India for patient care. While physicians provide access to medications and remain available to a patient’s family, those duties become much harder when caring for a greater number of patients during the pandemic. “That has led to some unrealistic expectations among the patients,” Dr. Adhikari said.
Dr. Bhatt said “more than half” of a physician’s time in India is spent counseling patients on concerns about COVID-19. “Awareness about the disease is limited from the patient and patient’s family perspective, as [there is] too much apprehension toward the nature of [the] disease,” he added. “Theoretical discussions collected from social media” obstruct the physician from executing his or her duties.
Physicians in India have had to contend with physical violence from patients and individuals on the street, Dr. Adhikari added. Workplace violence was already a concern – for years, the Indian Medical Association has cited a statistic that 75% of doctors in India have experienced violence at work (Indian J Psychiatry. 2019 Apr;61[Suppl 4]:S782-5). But the threat of violence against physicians has sharply increased during the COVID-19 pandemic. Disruptions to daily life through lockdowns “made people fearful, anxious, and sometimes they have found it difficult to access emergency treatment,” according to a letter published by Karthikeyan Iyengar and colleagues in the Postgraduate Medical Journal. In response to the restlessness, irritation, and despair resulting from hospitals closing their doors, “people have shown their frustration by verbally abusing and threatening to physically assault doctors and other health care workers,” the authors wrote.
A telemedicine boon in India
Back in the United States, hospitalists with family and friends in India were trying to figure out how to help. Some were working through the day, only to answer calls and WhatsApp messages from loved ones at night. “Everyone knows a physician or someone who’s your colleague, who owns a hospital or runs a hospital, or one of the family members is sick,” Dr. Adhikari said.
These U.S.-based hospitalists were burning the candle at both ends, helping with the pandemic in both countries. Physicians in India were posing questions to U.S. colleagues who they saw as having the most recent evidence for COVID-19 treatment. Out of the 180 physicians he trained with in India, Dr. Odeti said 110 of the physicians were in a large WhatsApp group chat that was constantly exchanging messages and serving as “kind of a friendly support group.”
In Dr. Odeti’s group chat, physicians helped one another find hospital beds for patients who reached out to them. “The first couple of weeks, there was no proper way for people to know where [patients] were based. There was no way to find if this hospital had a bed, so they reached out to any doctors they knew,” he said.
While he said it was emotionally draining, “at the same time, we felt a responsibility toward colleagues in India,” Dr. Odeti said, noting that as COVID-19 cases have decreased in India, the requests have been less frequent.
Because of concerns about traveling to India during the pandemic while on a J-1, H-1B, or other visa with the United States, directly helping friends and family in India seemed out of reach. But many hospitalists of Indian origin instead turned to telemedicine to help their colleagues. Telemedicine had already been steadily growing in India, but was accelerated by the pandemic. The current ratio of doctors to patients in India is 0.62 to 1,000 – lower than recommendations from the World Health Organization. That makes telemedicine a unique opportunity for one physician in India to reach many patients regardless of location.
Dr. Adhikari said he helped out his colleagues in India by performing consults for their patients. “They were just worried because they did not ‘know where to go, or what to get,” he said. “I was treating more patients in India than I was actually treating here.”
In March 2020, the Indian Ministry of Health and Family Welfare released telemedicine practice guidelines for the country, which relaxed regulations on privacy requirements and has been credited in part for giving telemedicine an additional boost during the pandemic. “That makes it easy for people to reach out but also has its own problems,” Dr. Adhikari said.
Monitoring of milder COVID-19 cases that don’t require hospitalization can be performed by a nurse who calls every few hours to check on a patient, make recommendations, and text treatment plans. “The telemedicine platforms are being adopted really fast,” Dr. Adhikari said. “The platforms were built in no time.”
According to NewZoo, a games market data analytics company, India has 345.9 million smartphone users as of 2019 – the second highest number of users in the world after China. Dr. Odeti said he believes telemedicine will be widely adopted.
“In India, they are very proactive in accepting these kinds of methods, so I’m sure they will,” he said. “Governments were trying to do it before the pandemic, because access to care is a problem in India. There are villages which are very, very remote.”
Reversion to old systems
After the peak in late May, new COVID-19 cases in India began to decrease, and the second wave waned on a national level. Hospitals began to get the supplies they needed, beds are available, and patients aren’t as sick as before, according to Dr. Adhikari. The federal government has begun issuing supplies to patients in each state, including COVID-19 vaccines. “The peak for the second wave is gone,” he said.
What remains is a group of physicians trained in how to triage patients and create efficiencies in a hospital setting. Could those skills be put to use elsewhere in India after the pandemic?
According to Dr. Bhatt, the patient care model is likely to revert to the system that existed before. “Whatever the changes, interims of bed occupancy, cost of ICU will be temporary [and] will change to normal,” he said. “But awareness about masks [and] sanitizing methods will be permanent.”
Dr. Adhikari believes that not utilizing the skills of newly minted hospitalists in India would be a missed opportunity. “This is a silver lining from COVID-19, that hospital medicine plays a vital role in the sickest patients, whether it is in India or the U.S. or anywhere,” he said. “I think the model of hospital medicine should be adopted. It’s not: ‘Should it really be adopted or not?’ It should be. There is a huge potential in doing inpatient coordinated [care], having people dedicated in the hospital.”
There are tangible benefits to creating efficiencies in India’s health system, Dr. Odeti said. Length of stay for sicker patients “was much longer” at 10-14 days during the second wave, compared with the United States, before lowering to around 5 days. “These hospitals right now are learning the efficient ways of doing it: when to send [patients] out, how to send them out, how to [perform] service-based practices, creating processes which were nonexistent before.”
While he doesn’t personally believe physicians will adopt a full-fledged hospitalist model unless the payer structure in India changes, “these people are at an advantage with this extra set of skills,” he said. “I think all the knowledge that these people have are going to come in handy.”
Opportunities for growth
Dr. Odeti sees the potential for the hospitalist model to grow in India – if not into its own specialty, then in how critical care consultants handle sicker patients and handoffs.
“The critical care clinician cannot keep the patient from the time they are admitted to the ICU until the discharge, so there will be a need for the transition,” Dr. Odeti said. “In the past, there were not many capabilities in Indian health systems to take care of these extremely sick patients, and now it is evolving. I think that is one more thing that will help.”
Dr. Adhikari said hospital systems in India are beginning to realize how having dedicated hospital physicians could benefit them. In India, “if you’re sick, you go to your doctor, you get treated and you disappear,” he said. The next time, you may see the same doctor or a completely different doctor. “There’s no system there, so it’s really hard for hospital medicine as such because patients, when they are very sick, they just come to the ER. They’re not followed by their primary care.”
Anecdotally, Dr. Odeti sees patients already adapting to having access to a physician for asking questions normally answered by primary care physicians. “I think primary care will come into play,” he said. “When I was doing a Zoom call for patients, they were asking me questions about sciatica. I think they are getting comfortable with this technology.”
A hospitalist model could even be applied to specific diseases with a large population of patients. Hospital administrators “have seen this for the first time, how efficient it could be if they had their own hospitalists and actually run it. So that’s the part that has crossed their minds,” Dr. Adhikari said. “How they will apply it going forward, other than during the COVID-19 pandemic, depends on the size of the hospital and the volume of the patients for a particular disease.”
“You can see in certain areas there is large growth for hospital medicine. But to rise to the level of the United States and how we do it, India needs bigger health systems to adopt the model,” Dr. Adhikari said.
FDA moves to block some vape products, delays action on Juul
The agency had a court-ordered deadline of Sept. 9 to review more than 6.5 million applications for approval of what are considered new tobacco products – the vast majority of which are e-cigarettes and liquids, none of which have gone through FDA review before.
The FDA reviewed 93% of those applications in the past year, acting FDA Commissioner Janet Woodcock, MD, and Mitch Zeller, director of the FDA’s Center for Tobacco Products, said in a statement.
Of those reviewed, the agency rejected more than 946,000 flavored vape products, “because their applications lacked sufficient evidence that they have a benefit to adult smokers sufficient to overcome the public health threat posed by the well-documented, alarming levels of youth use of such products,” Dr. Woodcock and Mr. Zeller said.
The pair said more work is needed to finish the reviews to “ensure that we continue taking appropriate action to protect our nation’s youth from the dangers of all tobacco products, including e-cigarettes, which remain the most commonly used tobacco product by youth in the United States.”
No e-cigarette product has been given official FDA approval to be sold, meaning all e-cigarette products technically are on the market illegally, the agency said in 2020, but federal officials decided only to begin enforcing rules against flavored products, which surveys show are more often used by children. Tobacco-flavored and menthol e-cigarette products – which some adults use to quit smoking cigarettes – were exempted.
The American Cancer Society and other advocacy groups slammed the FDA’s decision to withhold action on major e-cigarette manufacturers, including Juul.
“The FDA’s failure today to act on applications by Juul, the manufacturer with the single biggest e-cigarette market share, is extremely disappointing and will allow the industry to further endanger public health and hook more kids on their highly addictive products,” Lisa Lacasse, president of ACS CAN, said in a statement, according to CNN.
“The FDA has had ample time to review the applications and allowing additional delays is unconscionable. There is overwhelming data to demonstrate the negative impact these kinds of flavored products have had on public health and their role in the youth e-cigarette epidemic. The time to act is now,” Ms. Lacasse added.
E-cigarette use among high school students rose from 11.7% in 2017 to 19.6% in 2020, the American Cancer Society said. Nearly 5% of middle schoolers reported using them in 2020.
A version of this article first appeared on WebMD.com.
The agency had a court-ordered deadline of Sept. 9 to review more than 6.5 million applications for approval of what are considered new tobacco products – the vast majority of which are e-cigarettes and liquids, none of which have gone through FDA review before.
The FDA reviewed 93% of those applications in the past year, acting FDA Commissioner Janet Woodcock, MD, and Mitch Zeller, director of the FDA’s Center for Tobacco Products, said in a statement.
Of those reviewed, the agency rejected more than 946,000 flavored vape products, “because their applications lacked sufficient evidence that they have a benefit to adult smokers sufficient to overcome the public health threat posed by the well-documented, alarming levels of youth use of such products,” Dr. Woodcock and Mr. Zeller said.
The pair said more work is needed to finish the reviews to “ensure that we continue taking appropriate action to protect our nation’s youth from the dangers of all tobacco products, including e-cigarettes, which remain the most commonly used tobacco product by youth in the United States.”
No e-cigarette product has been given official FDA approval to be sold, meaning all e-cigarette products technically are on the market illegally, the agency said in 2020, but federal officials decided only to begin enforcing rules against flavored products, which surveys show are more often used by children. Tobacco-flavored and menthol e-cigarette products – which some adults use to quit smoking cigarettes – were exempted.
The American Cancer Society and other advocacy groups slammed the FDA’s decision to withhold action on major e-cigarette manufacturers, including Juul.
“The FDA’s failure today to act on applications by Juul, the manufacturer with the single biggest e-cigarette market share, is extremely disappointing and will allow the industry to further endanger public health and hook more kids on their highly addictive products,” Lisa Lacasse, president of ACS CAN, said in a statement, according to CNN.
“The FDA has had ample time to review the applications and allowing additional delays is unconscionable. There is overwhelming data to demonstrate the negative impact these kinds of flavored products have had on public health and their role in the youth e-cigarette epidemic. The time to act is now,” Ms. Lacasse added.
E-cigarette use among high school students rose from 11.7% in 2017 to 19.6% in 2020, the American Cancer Society said. Nearly 5% of middle schoolers reported using them in 2020.
A version of this article first appeared on WebMD.com.
The agency had a court-ordered deadline of Sept. 9 to review more than 6.5 million applications for approval of what are considered new tobacco products – the vast majority of which are e-cigarettes and liquids, none of which have gone through FDA review before.
The FDA reviewed 93% of those applications in the past year, acting FDA Commissioner Janet Woodcock, MD, and Mitch Zeller, director of the FDA’s Center for Tobacco Products, said in a statement.
Of those reviewed, the agency rejected more than 946,000 flavored vape products, “because their applications lacked sufficient evidence that they have a benefit to adult smokers sufficient to overcome the public health threat posed by the well-documented, alarming levels of youth use of such products,” Dr. Woodcock and Mr. Zeller said.
The pair said more work is needed to finish the reviews to “ensure that we continue taking appropriate action to protect our nation’s youth from the dangers of all tobacco products, including e-cigarettes, which remain the most commonly used tobacco product by youth in the United States.”
No e-cigarette product has been given official FDA approval to be sold, meaning all e-cigarette products technically are on the market illegally, the agency said in 2020, but federal officials decided only to begin enforcing rules against flavored products, which surveys show are more often used by children. Tobacco-flavored and menthol e-cigarette products – which some adults use to quit smoking cigarettes – were exempted.
The American Cancer Society and other advocacy groups slammed the FDA’s decision to withhold action on major e-cigarette manufacturers, including Juul.
“The FDA’s failure today to act on applications by Juul, the manufacturer with the single biggest e-cigarette market share, is extremely disappointing and will allow the industry to further endanger public health and hook more kids on their highly addictive products,” Lisa Lacasse, president of ACS CAN, said in a statement, according to CNN.
“The FDA has had ample time to review the applications and allowing additional delays is unconscionable. There is overwhelming data to demonstrate the negative impact these kinds of flavored products have had on public health and their role in the youth e-cigarette epidemic. The time to act is now,” Ms. Lacasse added.
E-cigarette use among high school students rose from 11.7% in 2017 to 19.6% in 2020, the American Cancer Society said. Nearly 5% of middle schoolers reported using them in 2020.
A version of this article first appeared on WebMD.com.
Pediatric Vaccines and Infectious Diseases
Pediatric Vaccines and Infectious Diseases Supplement
- We’re getting closer to a lifesaving RSV vaccine
- New tool may provide point-of-care differentiation between bacterial, viral infections
- Metapneumovirus infections clinically indistinguishable from flu, RSV
- Seeking new vaccines against whooping cough: The PERISCOPE project
- Dried blood spot tests show sensitivity as cCMV screen
With Commentary by Kristina A. Bryant, MD
Pediatric Vaccines and Infectious Diseases Supplement
- We’re getting closer to a lifesaving RSV vaccine
- New tool may provide point-of-care differentiation between bacterial, viral infections
- Metapneumovirus infections clinically indistinguishable from flu, RSV
- Seeking new vaccines against whooping cough: The PERISCOPE project
- Dried blood spot tests show sensitivity as cCMV screen
With Commentary by Kristina A. Bryant, MD
Pediatric Vaccines and Infectious Diseases Supplement
- We’re getting closer to a lifesaving RSV vaccine
- New tool may provide point-of-care differentiation between bacterial, viral infections
- Metapneumovirus infections clinically indistinguishable from flu, RSV
- Seeking new vaccines against whooping cough: The PERISCOPE project
- Dried blood spot tests show sensitivity as cCMV screen
With Commentary by Kristina A. Bryant, MD
COVID-19 spares lung function in young adults
Here’s some encouraging news for once regarding SARS-CoV-2 infections: A study of young adults for whom prepandemic spirometry data were available showed that COVID-19 did not have a significant impact on lung function, even among patients with asthma.
Among 853 Swedish men and women (mean age, 22 years) who were part of a birth cohort study, there were no significant differences in either forced expiratory volume in 1 second (FEV1) or in the ratio of FEV1 to forced vital capacity, reported Ida Mogensen, MD, PhD, a postdoctoral fellow at the Karolinska Institute in Stockholm.
“We found no effect of COVID-19 on spirometric lung function in generally healthy adults,” she said in an oral abstract presented at the European Respiratory Society 2021 International Congress.
The findings echo those of a small study that involved 73 children and adolescents with COVID-19 and 45 uninfected control persons. The investigators in that study, which was also presented at ERS 2021, found that there were no significant differences in the frequency of abnormal pulmonary function measures between case patients and control patients (abstract OA1303).
“The findings from these two studies provide important reassurance about the impact of COVID infection on lung function in children and young adults,” commented Anita Simonds, MD, an honorary consultant in respiratory and sleep medicine at the Royal Brompton Hospital, London.
“We know already that this group is less likely to suffer severe illness if they contract the virus, and these studies, which importantly include comparator groups without COVID-19, show that they are also less likely to suffer long-term consequences with respect to lung function,” she said. Dr. Simonds was not involved in either study.
Young adult study
Dr. Mogenson and colleagues assessed data on 853 participants in the BAMSE Project, a prospective birth cohort study that included 4,089 children born in Stockholm from 1994 to 1996. Of the participants, 147 had asthma. They have been regularly followed with questionnaires on respiratory symptoms and medications. In addition, at 8 and 16 years’ follow-up, spirometry measures and fractional exhaled nitric oxide (FeNO) levels were assessed, allergic sensitization tests were administered, and blood eosinophil levels were measured.
In 2020 and 2021, during the pandemic, the participants underwent spirometry testing and were assessed for antibodies against SARS-CoV-2, and they self-reported use of inhaled corticosteroids.
The investigators defined asthma as any physician diagnosis and asthma symptoms and/or asthma medication use within the previous year. Participants were determined to be COVID-19 seropositive if they had IgG antibodies to the SARS-CoV-2 spike greater than 25.09 AU/mL, IgM antibodies greater than 14.42 AU/mL, or IgA antibodies greater than 2.61 AU/mL, as measured with enzyme-linked immunosorbent assay.
Participants who had been vaccinated against COVID-19 were excluded.
No significant decreases
A total of 243 participants, including 38 with asthma, were seropositive for SARS-CoV-2 antibodies. The mean change in lung function from before the pandemic to the study end date during the pandemic were not significantly different between seropositive participants and seronegative participants or IgM-positive participants and seronegative participants.
Similarly, there were no significant differences in lung function between seropositive and seronegative participants in an analysis that was adjusted for sex, body mass index, smoking status, or prepandemic lung function.
Although there was a trend toward slightly lower function among seropositive participants with asthma in comparison with seronegative patients with asthma, it was not statistically significant, Dr. Mogenson said.
There were also no significant decreases in lung function from the prepandemic measure to the present in any of the inflammatory parameters, including blood eosinophil levels, FeNO, allergic sensitization, or inhaled corticosteroid use.
Potential misclassification
In the question-and-answer period that followed the presentation, session comoderator Sam Bayat, MD, PhD, from the University of Grenoble (France), who was not involved in the study, noted that “some subjects can have positive serology without any symptoms, while others can have symptomatic disease and a couple of months later they have negative serology.”
He asked Dr. Mogenson whether they had included in their study participants with symptomatic COVID-19 and whether that would change the findings.
“We did not have access to RNA testing, so we only had serology, and of course some participants could be wrongly classified to have disease – probably around 15%,” she acknowledged.
She noted that there were no significant changes in lung function among patients who reported having respiratory symptoms.
The study was funded by the Swedish Research Council, the Swedish Heart-Lung Foundation, the Swedish Research Council for Health, Working Life and Welfare, the Karolinska Institutet, Formas, the European Research Council, and Region Stockholm. Dr. Mogenson, Dr. Simonds, and Dr. Bayat disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Here’s some encouraging news for once regarding SARS-CoV-2 infections: A study of young adults for whom prepandemic spirometry data were available showed that COVID-19 did not have a significant impact on lung function, even among patients with asthma.
Among 853 Swedish men and women (mean age, 22 years) who were part of a birth cohort study, there were no significant differences in either forced expiratory volume in 1 second (FEV1) or in the ratio of FEV1 to forced vital capacity, reported Ida Mogensen, MD, PhD, a postdoctoral fellow at the Karolinska Institute in Stockholm.
“We found no effect of COVID-19 on spirometric lung function in generally healthy adults,” she said in an oral abstract presented at the European Respiratory Society 2021 International Congress.
The findings echo those of a small study that involved 73 children and adolescents with COVID-19 and 45 uninfected control persons. The investigators in that study, which was also presented at ERS 2021, found that there were no significant differences in the frequency of abnormal pulmonary function measures between case patients and control patients (abstract OA1303).
“The findings from these two studies provide important reassurance about the impact of COVID infection on lung function in children and young adults,” commented Anita Simonds, MD, an honorary consultant in respiratory and sleep medicine at the Royal Brompton Hospital, London.
“We know already that this group is less likely to suffer severe illness if they contract the virus, and these studies, which importantly include comparator groups without COVID-19, show that they are also less likely to suffer long-term consequences with respect to lung function,” she said. Dr. Simonds was not involved in either study.
Young adult study
Dr. Mogenson and colleagues assessed data on 853 participants in the BAMSE Project, a prospective birth cohort study that included 4,089 children born in Stockholm from 1994 to 1996. Of the participants, 147 had asthma. They have been regularly followed with questionnaires on respiratory symptoms and medications. In addition, at 8 and 16 years’ follow-up, spirometry measures and fractional exhaled nitric oxide (FeNO) levels were assessed, allergic sensitization tests were administered, and blood eosinophil levels were measured.
In 2020 and 2021, during the pandemic, the participants underwent spirometry testing and were assessed for antibodies against SARS-CoV-2, and they self-reported use of inhaled corticosteroids.
The investigators defined asthma as any physician diagnosis and asthma symptoms and/or asthma medication use within the previous year. Participants were determined to be COVID-19 seropositive if they had IgG antibodies to the SARS-CoV-2 spike greater than 25.09 AU/mL, IgM antibodies greater than 14.42 AU/mL, or IgA antibodies greater than 2.61 AU/mL, as measured with enzyme-linked immunosorbent assay.
Participants who had been vaccinated against COVID-19 were excluded.
No significant decreases
A total of 243 participants, including 38 with asthma, were seropositive for SARS-CoV-2 antibodies. The mean change in lung function from before the pandemic to the study end date during the pandemic were not significantly different between seropositive participants and seronegative participants or IgM-positive participants and seronegative participants.
Similarly, there were no significant differences in lung function between seropositive and seronegative participants in an analysis that was adjusted for sex, body mass index, smoking status, or prepandemic lung function.
Although there was a trend toward slightly lower function among seropositive participants with asthma in comparison with seronegative patients with asthma, it was not statistically significant, Dr. Mogenson said.
There were also no significant decreases in lung function from the prepandemic measure to the present in any of the inflammatory parameters, including blood eosinophil levels, FeNO, allergic sensitization, or inhaled corticosteroid use.
Potential misclassification
In the question-and-answer period that followed the presentation, session comoderator Sam Bayat, MD, PhD, from the University of Grenoble (France), who was not involved in the study, noted that “some subjects can have positive serology without any symptoms, while others can have symptomatic disease and a couple of months later they have negative serology.”
He asked Dr. Mogenson whether they had included in their study participants with symptomatic COVID-19 and whether that would change the findings.
“We did not have access to RNA testing, so we only had serology, and of course some participants could be wrongly classified to have disease – probably around 15%,” she acknowledged.
She noted that there were no significant changes in lung function among patients who reported having respiratory symptoms.
The study was funded by the Swedish Research Council, the Swedish Heart-Lung Foundation, the Swedish Research Council for Health, Working Life and Welfare, the Karolinska Institutet, Formas, the European Research Council, and Region Stockholm. Dr. Mogenson, Dr. Simonds, and Dr. Bayat disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Here’s some encouraging news for once regarding SARS-CoV-2 infections: A study of young adults for whom prepandemic spirometry data were available showed that COVID-19 did not have a significant impact on lung function, even among patients with asthma.
Among 853 Swedish men and women (mean age, 22 years) who were part of a birth cohort study, there were no significant differences in either forced expiratory volume in 1 second (FEV1) or in the ratio of FEV1 to forced vital capacity, reported Ida Mogensen, MD, PhD, a postdoctoral fellow at the Karolinska Institute in Stockholm.
“We found no effect of COVID-19 on spirometric lung function in generally healthy adults,” she said in an oral abstract presented at the European Respiratory Society 2021 International Congress.
The findings echo those of a small study that involved 73 children and adolescents with COVID-19 and 45 uninfected control persons. The investigators in that study, which was also presented at ERS 2021, found that there were no significant differences in the frequency of abnormal pulmonary function measures between case patients and control patients (abstract OA1303).
“The findings from these two studies provide important reassurance about the impact of COVID infection on lung function in children and young adults,” commented Anita Simonds, MD, an honorary consultant in respiratory and sleep medicine at the Royal Brompton Hospital, London.
“We know already that this group is less likely to suffer severe illness if they contract the virus, and these studies, which importantly include comparator groups without COVID-19, show that they are also less likely to suffer long-term consequences with respect to lung function,” she said. Dr. Simonds was not involved in either study.
Young adult study
Dr. Mogenson and colleagues assessed data on 853 participants in the BAMSE Project, a prospective birth cohort study that included 4,089 children born in Stockholm from 1994 to 1996. Of the participants, 147 had asthma. They have been regularly followed with questionnaires on respiratory symptoms and medications. In addition, at 8 and 16 years’ follow-up, spirometry measures and fractional exhaled nitric oxide (FeNO) levels were assessed, allergic sensitization tests were administered, and blood eosinophil levels were measured.
In 2020 and 2021, during the pandemic, the participants underwent spirometry testing and were assessed for antibodies against SARS-CoV-2, and they self-reported use of inhaled corticosteroids.
The investigators defined asthma as any physician diagnosis and asthma symptoms and/or asthma medication use within the previous year. Participants were determined to be COVID-19 seropositive if they had IgG antibodies to the SARS-CoV-2 spike greater than 25.09 AU/mL, IgM antibodies greater than 14.42 AU/mL, or IgA antibodies greater than 2.61 AU/mL, as measured with enzyme-linked immunosorbent assay.
Participants who had been vaccinated against COVID-19 were excluded.
No significant decreases
A total of 243 participants, including 38 with asthma, were seropositive for SARS-CoV-2 antibodies. The mean change in lung function from before the pandemic to the study end date during the pandemic were not significantly different between seropositive participants and seronegative participants or IgM-positive participants and seronegative participants.
Similarly, there were no significant differences in lung function between seropositive and seronegative participants in an analysis that was adjusted for sex, body mass index, smoking status, or prepandemic lung function.
Although there was a trend toward slightly lower function among seropositive participants with asthma in comparison with seronegative patients with asthma, it was not statistically significant, Dr. Mogenson said.
There were also no significant decreases in lung function from the prepandemic measure to the present in any of the inflammatory parameters, including blood eosinophil levels, FeNO, allergic sensitization, or inhaled corticosteroid use.
Potential misclassification
In the question-and-answer period that followed the presentation, session comoderator Sam Bayat, MD, PhD, from the University of Grenoble (France), who was not involved in the study, noted that “some subjects can have positive serology without any symptoms, while others can have symptomatic disease and a couple of months later they have negative serology.”
He asked Dr. Mogenson whether they had included in their study participants with symptomatic COVID-19 and whether that would change the findings.
“We did not have access to RNA testing, so we only had serology, and of course some participants could be wrongly classified to have disease – probably around 15%,” she acknowledged.
She noted that there were no significant changes in lung function among patients who reported having respiratory symptoms.
The study was funded by the Swedish Research Council, the Swedish Heart-Lung Foundation, the Swedish Research Council for Health, Working Life and Welfare, the Karolinska Institutet, Formas, the European Research Council, and Region Stockholm. Dr. Mogenson, Dr. Simonds, and Dr. Bayat disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Erythematous and Ulcerated Plaque on the Left Temple
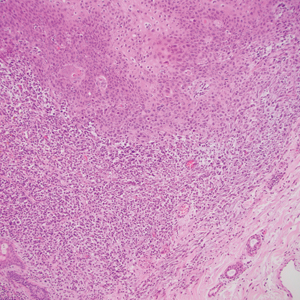
The Diagnosis: Primary Cutaneous Carcinosarcoma
The immunohistochemical findings supported an epithelial component consistent with moderately differentiated squamous cell carcinoma (SCC) and a mesenchymal component with features consistent with a sarcoma. Consequently, the lesion was diagnosed as a primary cutaneous carcinosarcoma (PCCS).
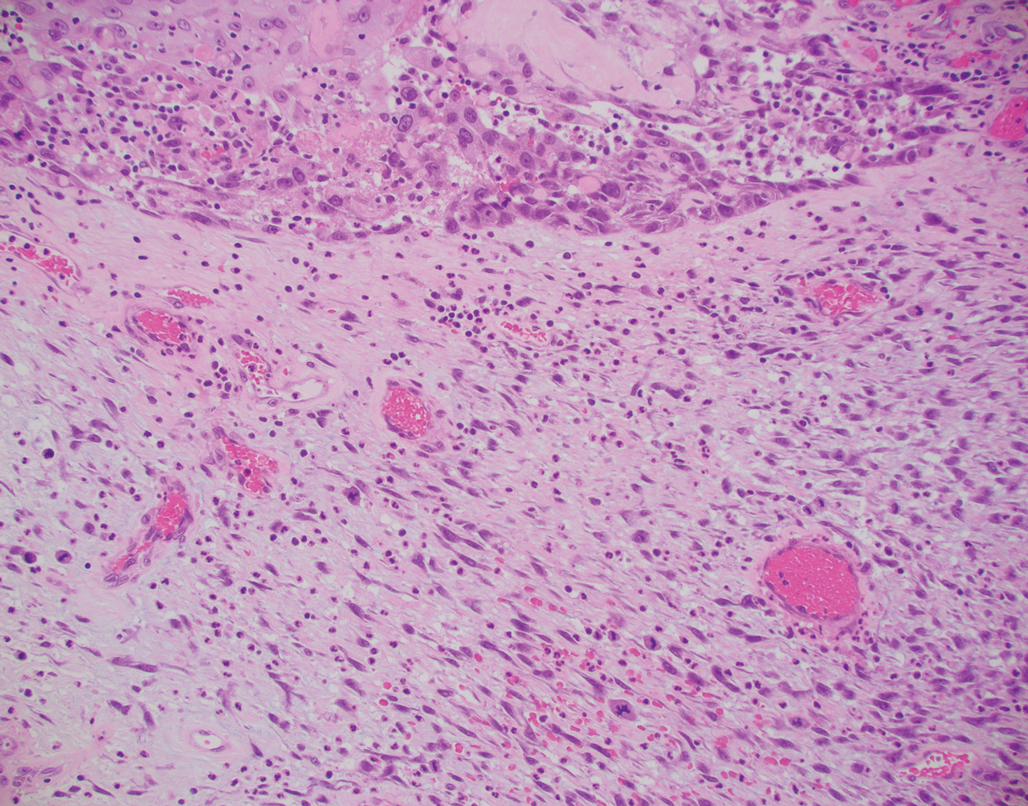
Primary cutaneous carcinosarcoma is a rare biphasic neoplasm consisting of malignant epithelial (carcinoma) and mesenchymal (sarcoma) components.1 Primary cutaneous carcinosarcomas are uncommon, poorly understood, primary cutaneous tumors.2,3 Characteristic of this tumor, cytokeratins highlight the epithelial component while vimentin highlights the mesenchymal component.4 Histologically, the sarcomatous components of PCCS often are highly variable, with an absence of transitional areas within the epithelial component, which frequently resembles basal cell carcinoma and/ or SCC.5-7 Primary cutaneous carcinosarcoma favors areas of chronic UV radiation exposure, particularly on the head and neck. Most tumors present with a slowly growing, polypoid, flesh-colored to erythematous nodule due to the infiltrative mesenchymal component.7 Primary cutaneous carcinosarcoma primarily is diagnosed in elderly patients, with the majority of cases diagnosed in the eighth or ninth decades of life (range, 32–98 years).1,8 Men appear to be twice as likely to be diagnosed with a PCCS compared to women.1 Primary cutaneous carcinosarcomas are recognized as aggressive tumors with a high propensity to metastasize and recur locally, necessitating early diagnosis and treatment.4 Accurate diagnosis of PCCSs can be challenging due to the biphasic nature of the neoplasm as well as poor differentiation or unequal proportions of the epithelial and mesenchymal components.5 Additionally, overlapping diagnostic criteria coupled with vague demarcation between soft-tissue sarcomas and distinct carcinomas also may contribute to a delay in diagnosis.9 Treatment is achieved surgically by complete wide resection, with no evidence to support the use of adjuvant or neoadjuvant external beam radiation therapy. Due to the small number of reported cases, no treatment recommendations currently exist.1
Surgical management with wide local excision has been disappointing, with recurrence rates reported as high as 33%.6 Primary cutaneous carcinosarcoma has an estimated overall recurrence rate of 19% and a 5-year disease-free rate of 50%.10 Risk factors associated with poorer prognosis include tumors with adnexal subtype, age less than 65 years, rapid tumor growth, a tumor greater than 20 mm at presentation, and a long-standing tumor lasting up to 30 years.2,4 Although wide local excision and Mohs micrographic surgery (MMS) both have been utilized successfully, MMS has been shown to result in a cure rate of greater than 98%.6
Atypical fibroxanthoma (AFX) is a cutaneous tumor of fibrohistiocytic mesenchymal origin that typically manifests on sun-damaged skin in elderly individuals. Clinically, it presents as a rapidly growing neoplasm that often ulcerates and bleeds. These heterogenous neoplasms have several distinct characteristics, including dense cellularity with disorganized, large, pleomorphic, and atypical-appearing spindle-shaped cells arising in the upper layers of the dermis, often disseminating into the reticular dermis and occasionally into the subcutaneous fat (Figure 1). The neoplastic cells often exhibit hyperchromic and irregular nuclei, multinucleated giant cells, and atypical mitotic figures. In most cases, negative immunohistochemical staining with SOX-10, S-100, cytokeratins, desmin, and caldesmon will allow pathologists to differentiate between AFX and other common tumors on the differential diagnosis, such as SCC, melanoma, and leiomyosarcoma. CD10 and procollagen type 1 are positive antigenic markers in AFX, but they are not specific. The standard treatment of AFX includes wide local excision or MMS for superior margin control.11
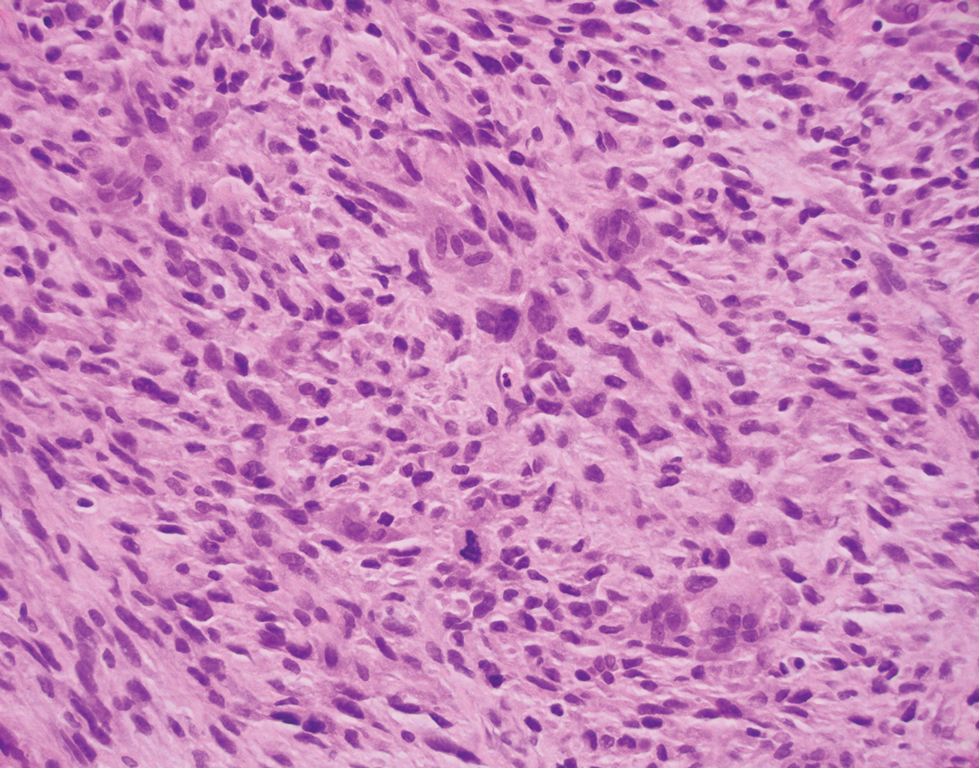
Spindle cell SCC presents as a raised or exophytic nodule, often with spontaneous bleeding and central ulceration. It usually presents on sun-damaged skin or in individuals with a history of ionizing radiation. Histologically, it is characterized by atypical spindleshaped keratinocytes in the dermis existing as single cells or cohesive nests along with keratin pearls (Figure 2). The atypical spindle cells may comprise the entire tumor or only a small portion. The use of immunohistochemical markers often is required to establish a definitive diagnosis. Spindle cell SCC stains positively, albeit frequently focally, for p63, p40, and high-molecular-weight cytokeratins such as cytokeratin 5/6, while S-100 protein, SOX-10, MART-1/Melan-A, and muscle-specific actin stains typically are negative. Wide local excision or MMS is recommended for treatment of these lesions.12
Primary cutaneous myoepithelial carcinomas are uncommon neoplasms of myoepithelial differentiation. Clinically, they often arise as soft nodular lesions on the head, neck, and lower extremities with a bimodal age distribution (50 years). Histologically cutaneous myoepithelial tumors are well-differentiated, dermal-based nodules without connection to the overlying epidermis (Figure 3). The myoepithelial cells can exhibit spindled, epithelioid, plasmacytoid, or clear cell morphologic features and show variability in cell growth patterns. One of the most common growth patterns is oval to round cells forming cords and chains in a chondromyxoid stroma. Most cases display an immunophenotyped co-expression of an epithelial cytokeratin and S-100 protein. Myoepithelial markers also may be present, including keratins, smooth muscle actin, calponin, glial fibrillary acidic protein, p63, and desmin. Surgical removal with wide local excision or MMS is essential.13
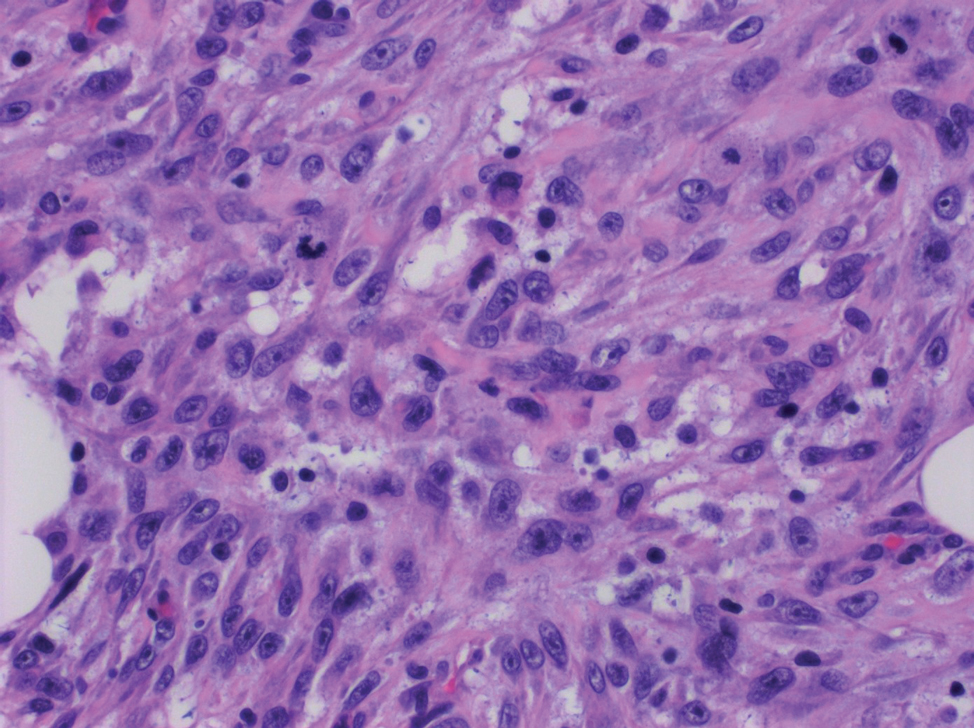
Leiomyosarcoma (LMS) is a tumor that originates from smooth muscle and rarely develops in the dermis.14 Pleomorphic LMS is a morphologic variant of LMS that has a low propensity to metastasize but commonly exhibits local recurrence.15 Leiomyosarcoma can present in any age group but most commonly manifests in individuals aged 50 to 70 years. Clinically, LMS presents as a firm solitary nodule with a smooth pink surface or a more exophytic tumor with a reddish or brown color on the extensor surface of the lower limbs; it is less common on the scalp and face.14 Histologically, most cases of pleomorphic LMS show small foci of fascicles consisting of smooth muscle tumor cells in addition to cellular pleomorphism (Figure 4).15 Many of these cells demonstrate a clear perinuclear vacuole that generally is appreciated in neoplastic smooth muscle cells.14 Pleomorphic LMS typically stains positively for at least one smooth muscle marker including desmin, h-caldesmon, muscle-specific actin, α-smooth muscle actin, or smooth muscle myosin in the leiomyosarcomatous fascicular areas.16 Complete surgical excision is the treatment of choice, and the best results are obtained with MMS.14
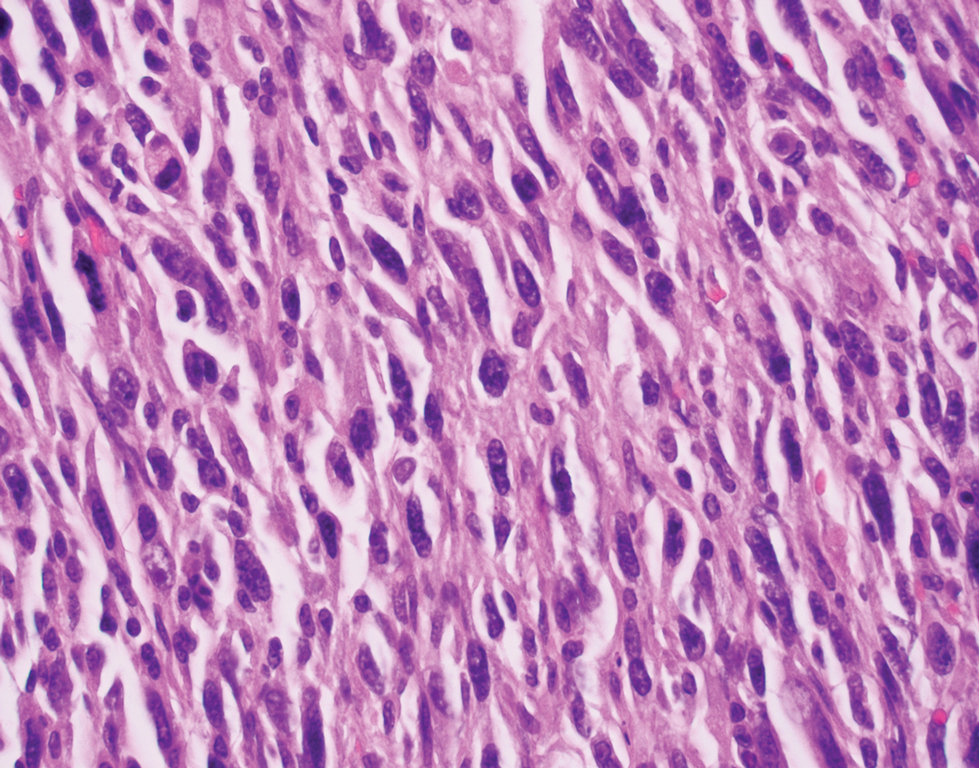
- Syme-Grant J, Syme-Grant NJ, Motta L, et al. Are primary cutaneous carcinosarcomas underdiagnosed? five cases and a review of the literature. J Plast Reconstr Aesthet Surg. 2006;59:1402-1408.
- Bourgeault E, Alain J, Gagne E. Primary cutaneous carcinosarcoma of the basal cell subtype should be treated as a high-risk basal cell carcinoma. J Cutan Med Surg. 2015;19:407-411.
- West L, Srivastava D. Cutaneous carcinosarcoma of the medial canthus discovered on Mohs debulk analysis. Dermatol Surg. 2019;45:1700-1702.
- Kwan JM, Satter EK. Carcinosarcoma: a primary cutaneous tumor with biphasic differentiation. Cutis. 2013;92:247-249.
- Suh KY, Lacouture M, Gerami P. p63 in primary cutaneous carcinosarcoma. Am J Dermatopathol. 2007;29:374‐377.
- Ruiz-Villaverde R, Aneiros-Fernandez J. Primary cutaneous carcinosarcoma: a cutaneous neoplasm with an exceptional presentation. Sultan Qaboos Univ Med J. 2018;18:E114-E115.
- Smart CN, Pucci RA, Binder SW, et al. Cutaneous carcinosarcoma with myoepithelial differentiation: immunohistochemical and cytogenetic analysis of a case presenting in an unusual location. Am J Dermatopathol. 2009;31:715‐717.
- Clark JJ, Bowen AR, Bowen GM, et al. Cutaneous carcinosarcoma: a series of six cases and a review of the literature. J Cutan Pathol. 2017;44:34‐44.
- Müller CS, Pföhler C, Schiekofer C, et al. Primary cutaneous carcinosarcomas: a morphological histogenetic concept revisited. Am J Dermatopathol. 2014;36:328‐339.
- Bellew S, Del Rosso JQ, Mobini N. Primary carcinosarcoma of the ear: case report and review of the literature. J Clin Aesthet Dermatol. 2009;2:33‐35.
- Hong SH, Hong SJ, Lee Y, et al. Primary cutaneous carcinosarcoma of the shoulder: case report with literature review. Dermatol Surg. 2013;39:338-340.
- Soleymani T, Aasi SZ, Novoa R, et al. Atypical fibroxanthoma and pleomorphic dermal sarcoma: updates on classification and management. Dermatol Clin. 2019;37:253-259.
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525.
- Johnson GE, Stevens K, Morrison AO, et al. Cutaneous myoepithelial carcinoma with disseminated metastases. Cutis. 2017;99:E19-E26.
- Llombart B, Serra-Guillén C, Requena C, et al. Leiomyosarcoma and pleomorphic dermal sarcoma: guidelines for diagnosis and treatment. Actas Dermosifiliogr. 2019;110:4-11.
- Oda Y, Miyajima K, Kawaguchi K, et al. Pleomorphic leiomyosarcoma: clinicopathologic and immunohistochemical study with special emphasis on its distinction from ordinary leiomyosarcoma and malignant fibrous histiocytoma. Am J Surg Pathol. 2001;25:1030-1038.
The Diagnosis: Primary Cutaneous Carcinosarcoma
The immunohistochemical findings supported an epithelial component consistent with moderately differentiated squamous cell carcinoma (SCC) and a mesenchymal component with features consistent with a sarcoma. Consequently, the lesion was diagnosed as a primary cutaneous carcinosarcoma (PCCS).
Primary cutaneous carcinosarcoma is a rare biphasic neoplasm consisting of malignant epithelial (carcinoma) and mesenchymal (sarcoma) components.1 Primary cutaneous carcinosarcomas are uncommon, poorly understood, primary cutaneous tumors.2,3 Characteristic of this tumor, cytokeratins highlight the epithelial component while vimentin highlights the mesenchymal component.4 Histologically, the sarcomatous components of PCCS often are highly variable, with an absence of transitional areas within the epithelial component, which frequently resembles basal cell carcinoma and/ or SCC.5-7 Primary cutaneous carcinosarcoma favors areas of chronic UV radiation exposure, particularly on the head and neck. Most tumors present with a slowly growing, polypoid, flesh-colored to erythematous nodule due to the infiltrative mesenchymal component.7 Primary cutaneous carcinosarcoma primarily is diagnosed in elderly patients, with the majority of cases diagnosed in the eighth or ninth decades of life (range, 32–98 years).1,8 Men appear to be twice as likely to be diagnosed with a PCCS compared to women.1 Primary cutaneous carcinosarcomas are recognized as aggressive tumors with a high propensity to metastasize and recur locally, necessitating early diagnosis and treatment.4 Accurate diagnosis of PCCSs can be challenging due to the biphasic nature of the neoplasm as well as poor differentiation or unequal proportions of the epithelial and mesenchymal components.5 Additionally, overlapping diagnostic criteria coupled with vague demarcation between soft-tissue sarcomas and distinct carcinomas also may contribute to a delay in diagnosis.9 Treatment is achieved surgically by complete wide resection, with no evidence to support the use of adjuvant or neoadjuvant external beam radiation therapy. Due to the small number of reported cases, no treatment recommendations currently exist.1
Surgical management with wide local excision has been disappointing, with recurrence rates reported as high as 33%.6 Primary cutaneous carcinosarcoma has an estimated overall recurrence rate of 19% and a 5-year disease-free rate of 50%.10 Risk factors associated with poorer prognosis include tumors with adnexal subtype, age less than 65 years, rapid tumor growth, a tumor greater than 20 mm at presentation, and a long-standing tumor lasting up to 30 years.2,4 Although wide local excision and Mohs micrographic surgery (MMS) both have been utilized successfully, MMS has been shown to result in a cure rate of greater than 98%.6
Atypical fibroxanthoma (AFX) is a cutaneous tumor of fibrohistiocytic mesenchymal origin that typically manifests on sun-damaged skin in elderly individuals. Clinically, it presents as a rapidly growing neoplasm that often ulcerates and bleeds. These heterogenous neoplasms have several distinct characteristics, including dense cellularity with disorganized, large, pleomorphic, and atypical-appearing spindle-shaped cells arising in the upper layers of the dermis, often disseminating into the reticular dermis and occasionally into the subcutaneous fat (Figure 1). The neoplastic cells often exhibit hyperchromic and irregular nuclei, multinucleated giant cells, and atypical mitotic figures. In most cases, negative immunohistochemical staining with SOX-10, S-100, cytokeratins, desmin, and caldesmon will allow pathologists to differentiate between AFX and other common tumors on the differential diagnosis, such as SCC, melanoma, and leiomyosarcoma. CD10 and procollagen type 1 are positive antigenic markers in AFX, but they are not specific. The standard treatment of AFX includes wide local excision or MMS for superior margin control.11

Spindle cell SCC presents as a raised or exophytic nodule, often with spontaneous bleeding and central ulceration. It usually presents on sun-damaged skin or in individuals with a history of ionizing radiation. Histologically, it is characterized by atypical spindleshaped keratinocytes in the dermis existing as single cells or cohesive nests along with keratin pearls (Figure 2). The atypical spindle cells may comprise the entire tumor or only a small portion. The use of immunohistochemical markers often is required to establish a definitive diagnosis. Spindle cell SCC stains positively, albeit frequently focally, for p63, p40, and high-molecular-weight cytokeratins such as cytokeratin 5/6, while S-100 protein, SOX-10, MART-1/Melan-A, and muscle-specific actin stains typically are negative. Wide local excision or MMS is recommended for treatment of these lesions.12

Primary cutaneous myoepithelial carcinomas are uncommon neoplasms of myoepithelial differentiation. Clinically, they often arise as soft nodular lesions on the head, neck, and lower extremities with a bimodal age distribution (50 years). Histologically cutaneous myoepithelial tumors are well-differentiated, dermal-based nodules without connection to the overlying epidermis (Figure 3). The myoepithelial cells can exhibit spindled, epithelioid, plasmacytoid, or clear cell morphologic features and show variability in cell growth patterns. One of the most common growth patterns is oval to round cells forming cords and chains in a chondromyxoid stroma. Most cases display an immunophenotyped co-expression of an epithelial cytokeratin and S-100 protein. Myoepithelial markers also may be present, including keratins, smooth muscle actin, calponin, glial fibrillary acidic protein, p63, and desmin. Surgical removal with wide local excision or MMS is essential.13

Leiomyosarcoma (LMS) is a tumor that originates from smooth muscle and rarely develops in the dermis.14 Pleomorphic LMS is a morphologic variant of LMS that has a low propensity to metastasize but commonly exhibits local recurrence.15 Leiomyosarcoma can present in any age group but most commonly manifests in individuals aged 50 to 70 years. Clinically, LMS presents as a firm solitary nodule with a smooth pink surface or a more exophytic tumor with a reddish or brown color on the extensor surface of the lower limbs; it is less common on the scalp and face.14 Histologically, most cases of pleomorphic LMS show small foci of fascicles consisting of smooth muscle tumor cells in addition to cellular pleomorphism (Figure 4).15 Many of these cells demonstrate a clear perinuclear vacuole that generally is appreciated in neoplastic smooth muscle cells.14 Pleomorphic LMS typically stains positively for at least one smooth muscle marker including desmin, h-caldesmon, muscle-specific actin, α-smooth muscle actin, or smooth muscle myosin in the leiomyosarcomatous fascicular areas.16 Complete surgical excision is the treatment of choice, and the best results are obtained with MMS.14

The Diagnosis: Primary Cutaneous Carcinosarcoma
The immunohistochemical findings supported an epithelial component consistent with moderately differentiated squamous cell carcinoma (SCC) and a mesenchymal component with features consistent with a sarcoma. Consequently, the lesion was diagnosed as a primary cutaneous carcinosarcoma (PCCS).
Primary cutaneous carcinosarcoma is a rare biphasic neoplasm consisting of malignant epithelial (carcinoma) and mesenchymal (sarcoma) components.1 Primary cutaneous carcinosarcomas are uncommon, poorly understood, primary cutaneous tumors.2,3 Characteristic of this tumor, cytokeratins highlight the epithelial component while vimentin highlights the mesenchymal component.4 Histologically, the sarcomatous components of PCCS often are highly variable, with an absence of transitional areas within the epithelial component, which frequently resembles basal cell carcinoma and/ or SCC.5-7 Primary cutaneous carcinosarcoma favors areas of chronic UV radiation exposure, particularly on the head and neck. Most tumors present with a slowly growing, polypoid, flesh-colored to erythematous nodule due to the infiltrative mesenchymal component.7 Primary cutaneous carcinosarcoma primarily is diagnosed in elderly patients, with the majority of cases diagnosed in the eighth or ninth decades of life (range, 32–98 years).1,8 Men appear to be twice as likely to be diagnosed with a PCCS compared to women.1 Primary cutaneous carcinosarcomas are recognized as aggressive tumors with a high propensity to metastasize and recur locally, necessitating early diagnosis and treatment.4 Accurate diagnosis of PCCSs can be challenging due to the biphasic nature of the neoplasm as well as poor differentiation or unequal proportions of the epithelial and mesenchymal components.5 Additionally, overlapping diagnostic criteria coupled with vague demarcation between soft-tissue sarcomas and distinct carcinomas also may contribute to a delay in diagnosis.9 Treatment is achieved surgically by complete wide resection, with no evidence to support the use of adjuvant or neoadjuvant external beam radiation therapy. Due to the small number of reported cases, no treatment recommendations currently exist.1
Surgical management with wide local excision has been disappointing, with recurrence rates reported as high as 33%.6 Primary cutaneous carcinosarcoma has an estimated overall recurrence rate of 19% and a 5-year disease-free rate of 50%.10 Risk factors associated with poorer prognosis include tumors with adnexal subtype, age less than 65 years, rapid tumor growth, a tumor greater than 20 mm at presentation, and a long-standing tumor lasting up to 30 years.2,4 Although wide local excision and Mohs micrographic surgery (MMS) both have been utilized successfully, MMS has been shown to result in a cure rate of greater than 98%.6
Atypical fibroxanthoma (AFX) is a cutaneous tumor of fibrohistiocytic mesenchymal origin that typically manifests on sun-damaged skin in elderly individuals. Clinically, it presents as a rapidly growing neoplasm that often ulcerates and bleeds. These heterogenous neoplasms have several distinct characteristics, including dense cellularity with disorganized, large, pleomorphic, and atypical-appearing spindle-shaped cells arising in the upper layers of the dermis, often disseminating into the reticular dermis and occasionally into the subcutaneous fat (Figure 1). The neoplastic cells often exhibit hyperchromic and irregular nuclei, multinucleated giant cells, and atypical mitotic figures. In most cases, negative immunohistochemical staining with SOX-10, S-100, cytokeratins, desmin, and caldesmon will allow pathologists to differentiate between AFX and other common tumors on the differential diagnosis, such as SCC, melanoma, and leiomyosarcoma. CD10 and procollagen type 1 are positive antigenic markers in AFX, but they are not specific. The standard treatment of AFX includes wide local excision or MMS for superior margin control.11

Spindle cell SCC presents as a raised or exophytic nodule, often with spontaneous bleeding and central ulceration. It usually presents on sun-damaged skin or in individuals with a history of ionizing radiation. Histologically, it is characterized by atypical spindleshaped keratinocytes in the dermis existing as single cells or cohesive nests along with keratin pearls (Figure 2). The atypical spindle cells may comprise the entire tumor or only a small portion. The use of immunohistochemical markers often is required to establish a definitive diagnosis. Spindle cell SCC stains positively, albeit frequently focally, for p63, p40, and high-molecular-weight cytokeratins such as cytokeratin 5/6, while S-100 protein, SOX-10, MART-1/Melan-A, and muscle-specific actin stains typically are negative. Wide local excision or MMS is recommended for treatment of these lesions.12

Primary cutaneous myoepithelial carcinomas are uncommon neoplasms of myoepithelial differentiation. Clinically, they often arise as soft nodular lesions on the head, neck, and lower extremities with a bimodal age distribution (50 years). Histologically cutaneous myoepithelial tumors are well-differentiated, dermal-based nodules without connection to the overlying epidermis (Figure 3). The myoepithelial cells can exhibit spindled, epithelioid, plasmacytoid, or clear cell morphologic features and show variability in cell growth patterns. One of the most common growth patterns is oval to round cells forming cords and chains in a chondromyxoid stroma. Most cases display an immunophenotyped co-expression of an epithelial cytokeratin and S-100 protein. Myoepithelial markers also may be present, including keratins, smooth muscle actin, calponin, glial fibrillary acidic protein, p63, and desmin. Surgical removal with wide local excision or MMS is essential.13

Leiomyosarcoma (LMS) is a tumor that originates from smooth muscle and rarely develops in the dermis.14 Pleomorphic LMS is a morphologic variant of LMS that has a low propensity to metastasize but commonly exhibits local recurrence.15 Leiomyosarcoma can present in any age group but most commonly manifests in individuals aged 50 to 70 years. Clinically, LMS presents as a firm solitary nodule with a smooth pink surface or a more exophytic tumor with a reddish or brown color on the extensor surface of the lower limbs; it is less common on the scalp and face.14 Histologically, most cases of pleomorphic LMS show small foci of fascicles consisting of smooth muscle tumor cells in addition to cellular pleomorphism (Figure 4).15 Many of these cells demonstrate a clear perinuclear vacuole that generally is appreciated in neoplastic smooth muscle cells.14 Pleomorphic LMS typically stains positively for at least one smooth muscle marker including desmin, h-caldesmon, muscle-specific actin, α-smooth muscle actin, or smooth muscle myosin in the leiomyosarcomatous fascicular areas.16 Complete surgical excision is the treatment of choice, and the best results are obtained with MMS.14

- Syme-Grant J, Syme-Grant NJ, Motta L, et al. Are primary cutaneous carcinosarcomas underdiagnosed? five cases and a review of the literature. J Plast Reconstr Aesthet Surg. 2006;59:1402-1408.
- Bourgeault E, Alain J, Gagne E. Primary cutaneous carcinosarcoma of the basal cell subtype should be treated as a high-risk basal cell carcinoma. J Cutan Med Surg. 2015;19:407-411.
- West L, Srivastava D. Cutaneous carcinosarcoma of the medial canthus discovered on Mohs debulk analysis. Dermatol Surg. 2019;45:1700-1702.
- Kwan JM, Satter EK. Carcinosarcoma: a primary cutaneous tumor with biphasic differentiation. Cutis. 2013;92:247-249.
- Suh KY, Lacouture M, Gerami P. p63 in primary cutaneous carcinosarcoma. Am J Dermatopathol. 2007;29:374‐377.
- Ruiz-Villaverde R, Aneiros-Fernandez J. Primary cutaneous carcinosarcoma: a cutaneous neoplasm with an exceptional presentation. Sultan Qaboos Univ Med J. 2018;18:E114-E115.
- Smart CN, Pucci RA, Binder SW, et al. Cutaneous carcinosarcoma with myoepithelial differentiation: immunohistochemical and cytogenetic analysis of a case presenting in an unusual location. Am J Dermatopathol. 2009;31:715‐717.
- Clark JJ, Bowen AR, Bowen GM, et al. Cutaneous carcinosarcoma: a series of six cases and a review of the literature. J Cutan Pathol. 2017;44:34‐44.
- Müller CS, Pföhler C, Schiekofer C, et al. Primary cutaneous carcinosarcomas: a morphological histogenetic concept revisited. Am J Dermatopathol. 2014;36:328‐339.
- Bellew S, Del Rosso JQ, Mobini N. Primary carcinosarcoma of the ear: case report and review of the literature. J Clin Aesthet Dermatol. 2009;2:33‐35.
- Hong SH, Hong SJ, Lee Y, et al. Primary cutaneous carcinosarcoma of the shoulder: case report with literature review. Dermatol Surg. 2013;39:338-340.
- Soleymani T, Aasi SZ, Novoa R, et al. Atypical fibroxanthoma and pleomorphic dermal sarcoma: updates on classification and management. Dermatol Clin. 2019;37:253-259.
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525.
- Johnson GE, Stevens K, Morrison AO, et al. Cutaneous myoepithelial carcinoma with disseminated metastases. Cutis. 2017;99:E19-E26.
- Llombart B, Serra-Guillén C, Requena C, et al. Leiomyosarcoma and pleomorphic dermal sarcoma: guidelines for diagnosis and treatment. Actas Dermosifiliogr. 2019;110:4-11.
- Oda Y, Miyajima K, Kawaguchi K, et al. Pleomorphic leiomyosarcoma: clinicopathologic and immunohistochemical study with special emphasis on its distinction from ordinary leiomyosarcoma and malignant fibrous histiocytoma. Am J Surg Pathol. 2001;25:1030-1038.
- Syme-Grant J, Syme-Grant NJ, Motta L, et al. Are primary cutaneous carcinosarcomas underdiagnosed? five cases and a review of the literature. J Plast Reconstr Aesthet Surg. 2006;59:1402-1408.
- Bourgeault E, Alain J, Gagne E. Primary cutaneous carcinosarcoma of the basal cell subtype should be treated as a high-risk basal cell carcinoma. J Cutan Med Surg. 2015;19:407-411.
- West L, Srivastava D. Cutaneous carcinosarcoma of the medial canthus discovered on Mohs debulk analysis. Dermatol Surg. 2019;45:1700-1702.
- Kwan JM, Satter EK. Carcinosarcoma: a primary cutaneous tumor with biphasic differentiation. Cutis. 2013;92:247-249.
- Suh KY, Lacouture M, Gerami P. p63 in primary cutaneous carcinosarcoma. Am J Dermatopathol. 2007;29:374‐377.
- Ruiz-Villaverde R, Aneiros-Fernandez J. Primary cutaneous carcinosarcoma: a cutaneous neoplasm with an exceptional presentation. Sultan Qaboos Univ Med J. 2018;18:E114-E115.
- Smart CN, Pucci RA, Binder SW, et al. Cutaneous carcinosarcoma with myoepithelial differentiation: immunohistochemical and cytogenetic analysis of a case presenting in an unusual location. Am J Dermatopathol. 2009;31:715‐717.
- Clark JJ, Bowen AR, Bowen GM, et al. Cutaneous carcinosarcoma: a series of six cases and a review of the literature. J Cutan Pathol. 2017;44:34‐44.
- Müller CS, Pföhler C, Schiekofer C, et al. Primary cutaneous carcinosarcomas: a morphological histogenetic concept revisited. Am J Dermatopathol. 2014;36:328‐339.
- Bellew S, Del Rosso JQ, Mobini N. Primary carcinosarcoma of the ear: case report and review of the literature. J Clin Aesthet Dermatol. 2009;2:33‐35.
- Hong SH, Hong SJ, Lee Y, et al. Primary cutaneous carcinosarcoma of the shoulder: case report with literature review. Dermatol Surg. 2013;39:338-340.
- Soleymani T, Aasi SZ, Novoa R, et al. Atypical fibroxanthoma and pleomorphic dermal sarcoma: updates on classification and management. Dermatol Clin. 2019;37:253-259.
- Parekh V, Seykora JT. Cutaneous squamous cell carcinoma. Clin Lab Med. 2017;37:503-525.
- Johnson GE, Stevens K, Morrison AO, et al. Cutaneous myoepithelial carcinoma with disseminated metastases. Cutis. 2017;99:E19-E26.
- Llombart B, Serra-Guillén C, Requena C, et al. Leiomyosarcoma and pleomorphic dermal sarcoma: guidelines for diagnosis and treatment. Actas Dermosifiliogr. 2019;110:4-11.
- Oda Y, Miyajima K, Kawaguchi K, et al. Pleomorphic leiomyosarcoma: clinicopathologic and immunohistochemical study with special emphasis on its distinction from ordinary leiomyosarcoma and malignant fibrous histiocytoma. Am J Surg Pathol. 2001;25:1030-1038.
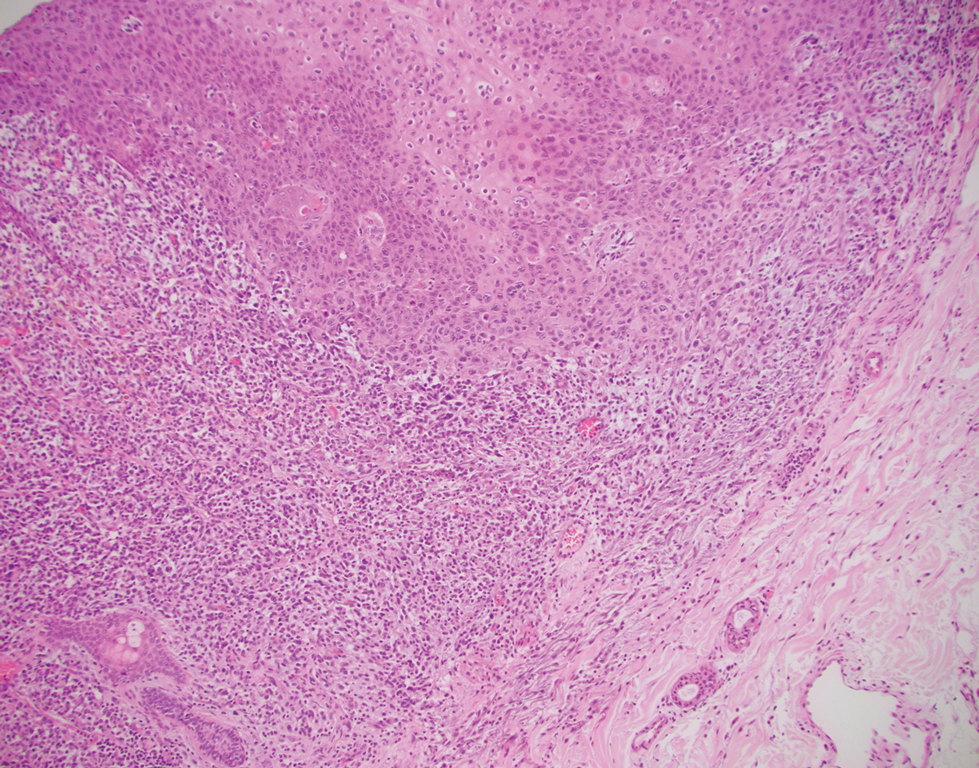
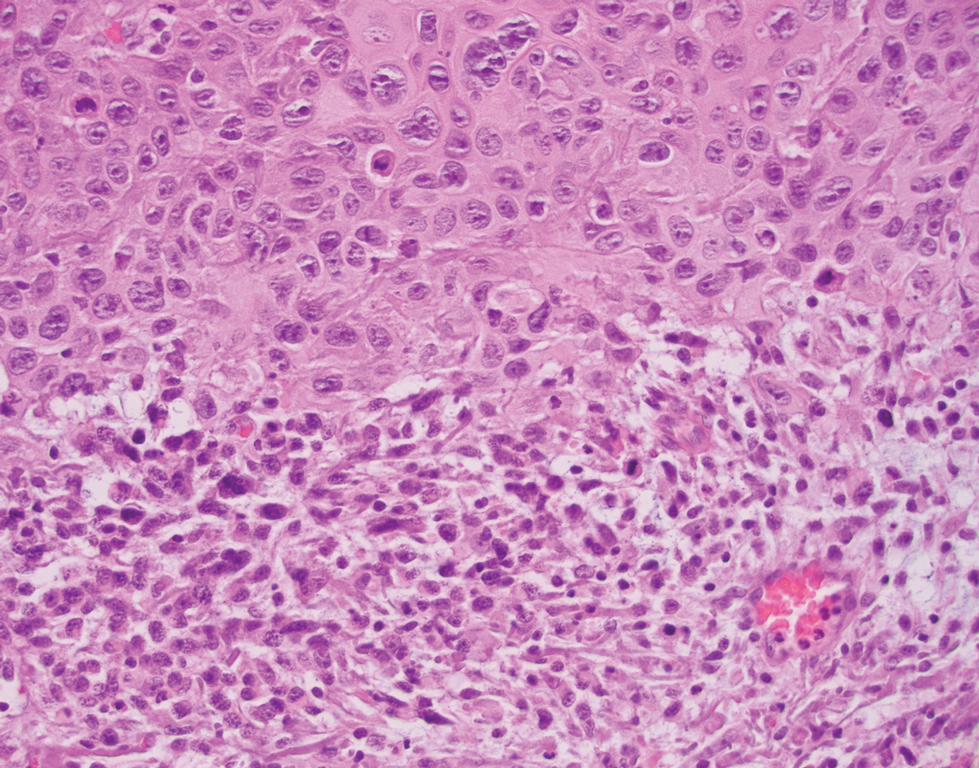
A 72-year-old man with a history of nonmelanoma skin cancer and lung transplant maintained on stable doses of prednisone and tacrolimus presented with a 1.3×1.8-cm, slow-growing, well-demarcated, ulcerated, erythematous plaque with overlying serous crust on the left temple of 6 months’ duration. No cervical or axillary lymphadenopathy was appreciated on physical examination. A biopsy was performed followed by Mohs micrographic surgery. Microscopic examination of the debulking specimen revealed atypical spindle cells in the papillary and reticular dermis radiating from a central focus of a moderately differentiated squamous cell carcinoma. The squamous cells stained positive for cytokeratin 5/6, pankeratin, and p40, while the spindle cells stained positive only for vimentin.
Atopic dermatitis subtype worsens into midlife, predicting poor health
giving reason to observe patients beyond the pediatric stage, according to a cohort study of more than 30,000 patients.
Early-life environmental factors, such as tobacco smoke exposure, were not reliable predictors of increasing AD into mid-adulthood, suggesting that a patient’s contemporaneous environment may impact disease course throughout life, reported lead author Katrina Abuabara, MD, associate professor of dermatology at the University of California, San Francisco, and colleagues.
“There is a lack of studies that prospectively examine the course of atopic eczema beyond adolescence/early adulthood, and a more comprehensive understanding of disease activity across the life span is needed,” the investigators wrote in JAMA Dermatology. “Data on long-term disease course may offer insight into mechanisms for disease onset and persistence, are important when counseling patients, and would establish baseline trajectories for future studies of whether new treatments can modify disease course and development of comorbidities.”
The present study included 30,905 patients from two population-based birth cohorts: the 1958 National Childhood Development Study (NCDS) and the 1970 British Cohort Study (BCS70). Follow-up data were collected between 1958 and 2016 via nine waves of standardized questionnaires, and subtypes of atopic eczema patterns were identified “based on parent-reported or self-reported atopic eczema period prevalence.”
This measure “was previously shown to coincide with standardized clinical examinations among children in the NCDS, and a similar questionnaire demonstrated high sensitivity and specificity for physician-diagnosed atopic eczema in U.S. populations,” the investigators noted.
Latent class analysis identified four disease subtypes based on probability of reporting prevalent AD into midlife: low (88%-91%), decreasing (4%), increasing (2%-6%), and persistently high (2%-3%) probability.
Next, the investigators looked for associations between these subtypes and established early-life risk factors, such as history of breastfeeding and childhood smoke exposure. None of the childhood environmental factors differentiated between high versus decreasing disease in adulthood, or increasing versus decreasing disease in adulthood. In contrast, female sex predicted the high versus decreasing adult subtype (odds ratio, 1.99; 95% confidence interval, 1.66-2.38), and the increasing versus decreasing adult subtype (OR, 1.99; 95% CI, 1.69-2.35).
These findings suggest that “disease trajectory is modifiable and may be influenced by environmental factors throughout life,” the investigators wrote.
Further analysis uncovered associations between adult AD subtypes and other health outcomes. For example, compared with adults in the low probability group, those in the high probability group were significantly more likely to report rhinitis (OR, 2.70; 95% CI, 2.24-3.26) and asthma (OR, 3.45; 95% CI, 2.82-4.21). Adults with the increasing subtype also had elevated rates of asthma and rhinitis, along with worse self-reported mental health at age 42 (OR, 1.45; 95% CI, 1.23-1.72) and poor general health at age 46/50 (OR, 1.29; 95% CI, 1.09-1.53).
“When extending the window of observation beyond childhood, clear subtypes of atopic eczema based on patterns of disease activity emerged,” the investigators concluded. “In particular, a newly identified subtype with increasing probability of activity in adulthood warrants additional attention given associations with poor self-reported physical and mental health in midlife.”
Commenting on these results, Robert Sidbury, MD, professor of dermatology at the University of Washington, Seattle, said that this is an “important study” because it adds to our understanding of natural disease course over time.
This knowledge, as a pediatric dermatologist, will help Dr. Sidbury answer one of the most common questions he hears from parents: When is it going to stop?
“Trying to put a little bit more evidence-based heft behind the answer ... is really important,” he said in an interview.
Based on available data, up to 10% of children with AD may have disease activity into adulthood, according to Dr. Sidbury, who is also chief of dermatology at Seattle Children’s Hospital.
“I would hazard to guess that most of those adults who have atopic dermatitis – at least the ones who had it in childhood – were told that they would grow out of it,” he said. “And so I think awareness is important – that [resolution with age] does not always happen.”
The findings also support the possibility that AD is a systemic disease, and that underlying immune dysregulation may be linked with serious health consequences later in life, Dr. Sidbury said, noting that “the stakes get higher and higher when you start speculating in that way.”
According to Dr. Sidbury, the reported link between childhood AD and poor midlife health raises questions about how modifiable the disease course may be, particularly in response to earlier intervention with emerging AD medications, which “seem to be much more effective and potent.”
“Will the advent of these medications and their adoption and use in treatment perhaps have a significant impact, not just on the prevention of atopic dermatitis itself, but maybe other comorbidities?” he asked.
For the time being, this question remains unanswered.
The study was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Wellcome Trust. Dr. Abuabara received grants from the National Institutes of Health during the study, as well as personal fees from Target RWE and Pfizer outside of this study. One author reported receiving NIH grants during the study, another reported receiving grants from the Wellcome Trust and the Innovative Medicine Initiative Horizon 2020 (BIOMAP project) during the study; there were no other disclosures. Dr. Sidbury disclosed relationships with Galderma, Regeneron, and Pfizer.
giving reason to observe patients beyond the pediatric stage, according to a cohort study of more than 30,000 patients.
Early-life environmental factors, such as tobacco smoke exposure, were not reliable predictors of increasing AD into mid-adulthood, suggesting that a patient’s contemporaneous environment may impact disease course throughout life, reported lead author Katrina Abuabara, MD, associate professor of dermatology at the University of California, San Francisco, and colleagues.
“There is a lack of studies that prospectively examine the course of atopic eczema beyond adolescence/early adulthood, and a more comprehensive understanding of disease activity across the life span is needed,” the investigators wrote in JAMA Dermatology. “Data on long-term disease course may offer insight into mechanisms for disease onset and persistence, are important when counseling patients, and would establish baseline trajectories for future studies of whether new treatments can modify disease course and development of comorbidities.”
The present study included 30,905 patients from two population-based birth cohorts: the 1958 National Childhood Development Study (NCDS) and the 1970 British Cohort Study (BCS70). Follow-up data were collected between 1958 and 2016 via nine waves of standardized questionnaires, and subtypes of atopic eczema patterns were identified “based on parent-reported or self-reported atopic eczema period prevalence.”
This measure “was previously shown to coincide with standardized clinical examinations among children in the NCDS, and a similar questionnaire demonstrated high sensitivity and specificity for physician-diagnosed atopic eczema in U.S. populations,” the investigators noted.
Latent class analysis identified four disease subtypes based on probability of reporting prevalent AD into midlife: low (88%-91%), decreasing (4%), increasing (2%-6%), and persistently high (2%-3%) probability.
Next, the investigators looked for associations between these subtypes and established early-life risk factors, such as history of breastfeeding and childhood smoke exposure. None of the childhood environmental factors differentiated between high versus decreasing disease in adulthood, or increasing versus decreasing disease in adulthood. In contrast, female sex predicted the high versus decreasing adult subtype (odds ratio, 1.99; 95% confidence interval, 1.66-2.38), and the increasing versus decreasing adult subtype (OR, 1.99; 95% CI, 1.69-2.35).
These findings suggest that “disease trajectory is modifiable and may be influenced by environmental factors throughout life,” the investigators wrote.
Further analysis uncovered associations between adult AD subtypes and other health outcomes. For example, compared with adults in the low probability group, those in the high probability group were significantly more likely to report rhinitis (OR, 2.70; 95% CI, 2.24-3.26) and asthma (OR, 3.45; 95% CI, 2.82-4.21). Adults with the increasing subtype also had elevated rates of asthma and rhinitis, along with worse self-reported mental health at age 42 (OR, 1.45; 95% CI, 1.23-1.72) and poor general health at age 46/50 (OR, 1.29; 95% CI, 1.09-1.53).
“When extending the window of observation beyond childhood, clear subtypes of atopic eczema based on patterns of disease activity emerged,” the investigators concluded. “In particular, a newly identified subtype with increasing probability of activity in adulthood warrants additional attention given associations with poor self-reported physical and mental health in midlife.”
Commenting on these results, Robert Sidbury, MD, professor of dermatology at the University of Washington, Seattle, said that this is an “important study” because it adds to our understanding of natural disease course over time.
This knowledge, as a pediatric dermatologist, will help Dr. Sidbury answer one of the most common questions he hears from parents: When is it going to stop?
“Trying to put a little bit more evidence-based heft behind the answer ... is really important,” he said in an interview.
Based on available data, up to 10% of children with AD may have disease activity into adulthood, according to Dr. Sidbury, who is also chief of dermatology at Seattle Children’s Hospital.
“I would hazard to guess that most of those adults who have atopic dermatitis – at least the ones who had it in childhood – were told that they would grow out of it,” he said. “And so I think awareness is important – that [resolution with age] does not always happen.”
The findings also support the possibility that AD is a systemic disease, and that underlying immune dysregulation may be linked with serious health consequences later in life, Dr. Sidbury said, noting that “the stakes get higher and higher when you start speculating in that way.”
According to Dr. Sidbury, the reported link between childhood AD and poor midlife health raises questions about how modifiable the disease course may be, particularly in response to earlier intervention with emerging AD medications, which “seem to be much more effective and potent.”
“Will the advent of these medications and their adoption and use in treatment perhaps have a significant impact, not just on the prevention of atopic dermatitis itself, but maybe other comorbidities?” he asked.
For the time being, this question remains unanswered.
The study was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Wellcome Trust. Dr. Abuabara received grants from the National Institutes of Health during the study, as well as personal fees from Target RWE and Pfizer outside of this study. One author reported receiving NIH grants during the study, another reported receiving grants from the Wellcome Trust and the Innovative Medicine Initiative Horizon 2020 (BIOMAP project) during the study; there were no other disclosures. Dr. Sidbury disclosed relationships with Galderma, Regeneron, and Pfizer.
giving reason to observe patients beyond the pediatric stage, according to a cohort study of more than 30,000 patients.
Early-life environmental factors, such as tobacco smoke exposure, were not reliable predictors of increasing AD into mid-adulthood, suggesting that a patient’s contemporaneous environment may impact disease course throughout life, reported lead author Katrina Abuabara, MD, associate professor of dermatology at the University of California, San Francisco, and colleagues.
“There is a lack of studies that prospectively examine the course of atopic eczema beyond adolescence/early adulthood, and a more comprehensive understanding of disease activity across the life span is needed,” the investigators wrote in JAMA Dermatology. “Data on long-term disease course may offer insight into mechanisms for disease onset and persistence, are important when counseling patients, and would establish baseline trajectories for future studies of whether new treatments can modify disease course and development of comorbidities.”
The present study included 30,905 patients from two population-based birth cohorts: the 1958 National Childhood Development Study (NCDS) and the 1970 British Cohort Study (BCS70). Follow-up data were collected between 1958 and 2016 via nine waves of standardized questionnaires, and subtypes of atopic eczema patterns were identified “based on parent-reported or self-reported atopic eczema period prevalence.”
This measure “was previously shown to coincide with standardized clinical examinations among children in the NCDS, and a similar questionnaire demonstrated high sensitivity and specificity for physician-diagnosed atopic eczema in U.S. populations,” the investigators noted.
Latent class analysis identified four disease subtypes based on probability of reporting prevalent AD into midlife: low (88%-91%), decreasing (4%), increasing (2%-6%), and persistently high (2%-3%) probability.
Next, the investigators looked for associations between these subtypes and established early-life risk factors, such as history of breastfeeding and childhood smoke exposure. None of the childhood environmental factors differentiated between high versus decreasing disease in adulthood, or increasing versus decreasing disease in adulthood. In contrast, female sex predicted the high versus decreasing adult subtype (odds ratio, 1.99; 95% confidence interval, 1.66-2.38), and the increasing versus decreasing adult subtype (OR, 1.99; 95% CI, 1.69-2.35).
These findings suggest that “disease trajectory is modifiable and may be influenced by environmental factors throughout life,” the investigators wrote.
Further analysis uncovered associations between adult AD subtypes and other health outcomes. For example, compared with adults in the low probability group, those in the high probability group were significantly more likely to report rhinitis (OR, 2.70; 95% CI, 2.24-3.26) and asthma (OR, 3.45; 95% CI, 2.82-4.21). Adults with the increasing subtype also had elevated rates of asthma and rhinitis, along with worse self-reported mental health at age 42 (OR, 1.45; 95% CI, 1.23-1.72) and poor general health at age 46/50 (OR, 1.29; 95% CI, 1.09-1.53).
“When extending the window of observation beyond childhood, clear subtypes of atopic eczema based on patterns of disease activity emerged,” the investigators concluded. “In particular, a newly identified subtype with increasing probability of activity in adulthood warrants additional attention given associations with poor self-reported physical and mental health in midlife.”
Commenting on these results, Robert Sidbury, MD, professor of dermatology at the University of Washington, Seattle, said that this is an “important study” because it adds to our understanding of natural disease course over time.
This knowledge, as a pediatric dermatologist, will help Dr. Sidbury answer one of the most common questions he hears from parents: When is it going to stop?
“Trying to put a little bit more evidence-based heft behind the answer ... is really important,” he said in an interview.
Based on available data, up to 10% of children with AD may have disease activity into adulthood, according to Dr. Sidbury, who is also chief of dermatology at Seattle Children’s Hospital.
“I would hazard to guess that most of those adults who have atopic dermatitis – at least the ones who had it in childhood – were told that they would grow out of it,” he said. “And so I think awareness is important – that [resolution with age] does not always happen.”
The findings also support the possibility that AD is a systemic disease, and that underlying immune dysregulation may be linked with serious health consequences later in life, Dr. Sidbury said, noting that “the stakes get higher and higher when you start speculating in that way.”
According to Dr. Sidbury, the reported link between childhood AD and poor midlife health raises questions about how modifiable the disease course may be, particularly in response to earlier intervention with emerging AD medications, which “seem to be much more effective and potent.”
“Will the advent of these medications and their adoption and use in treatment perhaps have a significant impact, not just on the prevention of atopic dermatitis itself, but maybe other comorbidities?” he asked.
For the time being, this question remains unanswered.
The study was funded by the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Wellcome Trust. Dr. Abuabara received grants from the National Institutes of Health during the study, as well as personal fees from Target RWE and Pfizer outside of this study. One author reported receiving NIH grants during the study, another reported receiving grants from the Wellcome Trust and the Innovative Medicine Initiative Horizon 2020 (BIOMAP project) during the study; there were no other disclosures. Dr. Sidbury disclosed relationships with Galderma, Regeneron, and Pfizer.
FROM JAMA DERMATOLOGY
Growing proportion of cardiac arrests in U.S. considered opioid related
Observational data indicate that the number of hospitalizations for cardiac arrests linked to opioid use roughly doubled from 2012 to 2018.
“This was an observational study, so we cannot conclude that all of the arrests were caused by opioids, but the findings do suggest the opioid epidemic is a contributor to increasing rates,” Senada S. Malik, of the University of New England, Portland, Maine, reported at the virtual annual congress of the European Society of Cardiology.
The data were drawn from the Nationwide Inpatient Sample (NIS) from 2012 to 2018, the most recent period available. Cardiac arrests were considered opioid related if there was a secondary diagnosis of opioid disease. The rates of opioid-associated hospitalizations for these types of cardiac arrests climbed from about 800 per year in 2012 to 1,500 per year in 2018, a trend that was statistically significant (P < .05).
The profile of patients with an opioid-associated cardiac arrest was different from those without secondary diagnosis of opioid disease. This included a younger age and lower rates of comorbidities: heart failure (21.2% vs. 40.6%; P < .05), renal failure (14.3% vs. 30.2%; P < .05), diabetes (19.5% vs. 35.4%; P < .05), and hypertension (43.4% vs. 64.9%; P < .05).
Mortality from opioid-associated cardiac arrest is lower
These features might explain the lower rate of in-hospital mortality for opioid-associated cardiac arrests (56.7% vs. 61.2%), according to Ms. Malik, who performed this research in collaboration with Wilbert S. Aronow, MD, director of cardiology research, Westchester Medical Center, Valhalla, N.Y.
When compared to those without a history of opioid use on admission, those with opioid-associated cardiac arrest were more likely to be depressed (18.8% vs. 9.0%), to smoke (37.0% vs. 21.8%) and to abuse alcohol (16.9% vs. 7.1%), according to the NIS data.
While these findings are based on cardiac arrests brought to a hospital, some opioid-induced cardiac arrests never result in hospital admission, according to data included in a recently issued scientific statement from the American Heart Association.
Rate of opioid-associated cardiac arrests underestimated
In that statement, which was focused on opioid-associated out-of-hospital cardiac arrests (OA-OHCA), numerous studies were cited to support the conclusion that these events are common and underestimated. One problem is that opioid-induced cardiac arrests are not always accurately differentiated from cardiac arrests induced by use of other substances, such as barbiturates, cocaine, or alcohol.
For this and other reasons, the data are inconsistent. One study based on emergency medical service (EMS) response data concluded that 9% of all out-of-hospital cardiac arrests are opioid associated.
In another study using potentially more accurate autopsy data, 60% of the non–cardiac-associated cardiac arrests were found to occur in individuals with potentially lethal serum concentrations of opioids. As 40% of out-of-hospital cardiac arrests were considered non–cardiac related, this suggested that 15% of all out-of-hospital cardiac arrests are opioid related.
In the NIS data, the incident curves of opioid-related cardiac arrests appeared to be flattening in 2018, the last year of data collection, but there was no indication they were declining.
Patterns of opioid-induced cardiac arrests evolving
The patterns of opioid-induced cardiac arrest have changed and are likely to continue to change in response to the evolving opioid epidemic, according to the AHA scientific statement. The authors described three waves of opioid abuse. The first, which was related to the promotion of prescription opioids to treat chronic pain that ultimately led to high rates of opioid addiction, peaked in 2012 when rates of these prescriptions began to fall. At that time a second wave, attributed to patients switching to less expensive nonprescription heroin, was already underway. A third wave, attributed to growth in the use of synthetic opioids, such as fentanyl, began in 2013 and is ongoing, according to data cited in the AHA statement.
Recognizing the role of opioids in rising rates of cardiac arrest is important for promoting strategies of effective treatment and prevention, according to Cameron Dezfulian, MD, medical director of the adult congenital heart disease program at Texas Children’s Hospital, Houston. Dr. Dezfulian was vice chair and leader of the writing committee for the AHA scientific statement on OA-OHCA. He said there are plenty of data to support the need for greater attention to the role of opioids in cardiac arrest.
“The recent data affirms the trends many of us have observed without our emergency rooms and ICUs: a steady increase in the proportion of OA-OHCA, primarily in young and otherwise healthy individuals,” he said.
He calls not only for more awareness at the front lines of health are but also for a more comprehensive approach.
“Public health policies and community- and hospital-based interventions are needed to reduce the mortality due to OA-OHCA, which is distinct from the traditional cardiac etiology,” Dr. Dezfulian said.
In opioid-induced cardiac arrest, as in other types of cardiac arrest, prompt initiation of cardiopulmonary resuscitation is essential, but early administration of the opioid antagonist naloxone can also be lifesaving, according to treatment strategies outlined in the AHA scientific statement. The fact that OA-OHCA typically occur in patients with structurally and electrophysiologically normal hearts is emphasized in the AHA statement. So is the enormous public health toll of OA-OHCA.
Death due to opioid overdose, which includes cardiac arrests, is now the leading cause of mortality in the U.S. among individuals between the ages of 25 and 64 years, according to the statement.
Ms. Malik reports no potential conflicts of interest. Dr. Dezfulian reports a financial relationship with Mallinckrodt.
Observational data indicate that the number of hospitalizations for cardiac arrests linked to opioid use roughly doubled from 2012 to 2018.
“This was an observational study, so we cannot conclude that all of the arrests were caused by opioids, but the findings do suggest the opioid epidemic is a contributor to increasing rates,” Senada S. Malik, of the University of New England, Portland, Maine, reported at the virtual annual congress of the European Society of Cardiology.
The data were drawn from the Nationwide Inpatient Sample (NIS) from 2012 to 2018, the most recent period available. Cardiac arrests were considered opioid related if there was a secondary diagnosis of opioid disease. The rates of opioid-associated hospitalizations for these types of cardiac arrests climbed from about 800 per year in 2012 to 1,500 per year in 2018, a trend that was statistically significant (P < .05).
The profile of patients with an opioid-associated cardiac arrest was different from those without secondary diagnosis of opioid disease. This included a younger age and lower rates of comorbidities: heart failure (21.2% vs. 40.6%; P < .05), renal failure (14.3% vs. 30.2%; P < .05), diabetes (19.5% vs. 35.4%; P < .05), and hypertension (43.4% vs. 64.9%; P < .05).
Mortality from opioid-associated cardiac arrest is lower
These features might explain the lower rate of in-hospital mortality for opioid-associated cardiac arrests (56.7% vs. 61.2%), according to Ms. Malik, who performed this research in collaboration with Wilbert S. Aronow, MD, director of cardiology research, Westchester Medical Center, Valhalla, N.Y.
When compared to those without a history of opioid use on admission, those with opioid-associated cardiac arrest were more likely to be depressed (18.8% vs. 9.0%), to smoke (37.0% vs. 21.8%) and to abuse alcohol (16.9% vs. 7.1%), according to the NIS data.
While these findings are based on cardiac arrests brought to a hospital, some opioid-induced cardiac arrests never result in hospital admission, according to data included in a recently issued scientific statement from the American Heart Association.
Rate of opioid-associated cardiac arrests underestimated
In that statement, which was focused on opioid-associated out-of-hospital cardiac arrests (OA-OHCA), numerous studies were cited to support the conclusion that these events are common and underestimated. One problem is that opioid-induced cardiac arrests are not always accurately differentiated from cardiac arrests induced by use of other substances, such as barbiturates, cocaine, or alcohol.
For this and other reasons, the data are inconsistent. One study based on emergency medical service (EMS) response data concluded that 9% of all out-of-hospital cardiac arrests are opioid associated.
In another study using potentially more accurate autopsy data, 60% of the non–cardiac-associated cardiac arrests were found to occur in individuals with potentially lethal serum concentrations of opioids. As 40% of out-of-hospital cardiac arrests were considered non–cardiac related, this suggested that 15% of all out-of-hospital cardiac arrests are opioid related.
In the NIS data, the incident curves of opioid-related cardiac arrests appeared to be flattening in 2018, the last year of data collection, but there was no indication they were declining.
Patterns of opioid-induced cardiac arrests evolving
The patterns of opioid-induced cardiac arrest have changed and are likely to continue to change in response to the evolving opioid epidemic, according to the AHA scientific statement. The authors described three waves of opioid abuse. The first, which was related to the promotion of prescription opioids to treat chronic pain that ultimately led to high rates of opioid addiction, peaked in 2012 when rates of these prescriptions began to fall. At that time a second wave, attributed to patients switching to less expensive nonprescription heroin, was already underway. A third wave, attributed to growth in the use of synthetic opioids, such as fentanyl, began in 2013 and is ongoing, according to data cited in the AHA statement.
Recognizing the role of opioids in rising rates of cardiac arrest is important for promoting strategies of effective treatment and prevention, according to Cameron Dezfulian, MD, medical director of the adult congenital heart disease program at Texas Children’s Hospital, Houston. Dr. Dezfulian was vice chair and leader of the writing committee for the AHA scientific statement on OA-OHCA. He said there are plenty of data to support the need for greater attention to the role of opioids in cardiac arrest.
“The recent data affirms the trends many of us have observed without our emergency rooms and ICUs: a steady increase in the proportion of OA-OHCA, primarily in young and otherwise healthy individuals,” he said.
He calls not only for more awareness at the front lines of health are but also for a more comprehensive approach.
“Public health policies and community- and hospital-based interventions are needed to reduce the mortality due to OA-OHCA, which is distinct from the traditional cardiac etiology,” Dr. Dezfulian said.
In opioid-induced cardiac arrest, as in other types of cardiac arrest, prompt initiation of cardiopulmonary resuscitation is essential, but early administration of the opioid antagonist naloxone can also be lifesaving, according to treatment strategies outlined in the AHA scientific statement. The fact that OA-OHCA typically occur in patients with structurally and electrophysiologically normal hearts is emphasized in the AHA statement. So is the enormous public health toll of OA-OHCA.
Death due to opioid overdose, which includes cardiac arrests, is now the leading cause of mortality in the U.S. among individuals between the ages of 25 and 64 years, according to the statement.
Ms. Malik reports no potential conflicts of interest. Dr. Dezfulian reports a financial relationship with Mallinckrodt.
Observational data indicate that the number of hospitalizations for cardiac arrests linked to opioid use roughly doubled from 2012 to 2018.
“This was an observational study, so we cannot conclude that all of the arrests were caused by opioids, but the findings do suggest the opioid epidemic is a contributor to increasing rates,” Senada S. Malik, of the University of New England, Portland, Maine, reported at the virtual annual congress of the European Society of Cardiology.
The data were drawn from the Nationwide Inpatient Sample (NIS) from 2012 to 2018, the most recent period available. Cardiac arrests were considered opioid related if there was a secondary diagnosis of opioid disease. The rates of opioid-associated hospitalizations for these types of cardiac arrests climbed from about 800 per year in 2012 to 1,500 per year in 2018, a trend that was statistically significant (P < .05).
The profile of patients with an opioid-associated cardiac arrest was different from those without secondary diagnosis of opioid disease. This included a younger age and lower rates of comorbidities: heart failure (21.2% vs. 40.6%; P < .05), renal failure (14.3% vs. 30.2%; P < .05), diabetes (19.5% vs. 35.4%; P < .05), and hypertension (43.4% vs. 64.9%; P < .05).
Mortality from opioid-associated cardiac arrest is lower
These features might explain the lower rate of in-hospital mortality for opioid-associated cardiac arrests (56.7% vs. 61.2%), according to Ms. Malik, who performed this research in collaboration with Wilbert S. Aronow, MD, director of cardiology research, Westchester Medical Center, Valhalla, N.Y.
When compared to those without a history of opioid use on admission, those with opioid-associated cardiac arrest were more likely to be depressed (18.8% vs. 9.0%), to smoke (37.0% vs. 21.8%) and to abuse alcohol (16.9% vs. 7.1%), according to the NIS data.
While these findings are based on cardiac arrests brought to a hospital, some opioid-induced cardiac arrests never result in hospital admission, according to data included in a recently issued scientific statement from the American Heart Association.
Rate of opioid-associated cardiac arrests underestimated
In that statement, which was focused on opioid-associated out-of-hospital cardiac arrests (OA-OHCA), numerous studies were cited to support the conclusion that these events are common and underestimated. One problem is that opioid-induced cardiac arrests are not always accurately differentiated from cardiac arrests induced by use of other substances, such as barbiturates, cocaine, or alcohol.
For this and other reasons, the data are inconsistent. One study based on emergency medical service (EMS) response data concluded that 9% of all out-of-hospital cardiac arrests are opioid associated.
In another study using potentially more accurate autopsy data, 60% of the non–cardiac-associated cardiac arrests were found to occur in individuals with potentially lethal serum concentrations of opioids. As 40% of out-of-hospital cardiac arrests were considered non–cardiac related, this suggested that 15% of all out-of-hospital cardiac arrests are opioid related.
In the NIS data, the incident curves of opioid-related cardiac arrests appeared to be flattening in 2018, the last year of data collection, but there was no indication they were declining.
Patterns of opioid-induced cardiac arrests evolving
The patterns of opioid-induced cardiac arrest have changed and are likely to continue to change in response to the evolving opioid epidemic, according to the AHA scientific statement. The authors described three waves of opioid abuse. The first, which was related to the promotion of prescription opioids to treat chronic pain that ultimately led to high rates of opioid addiction, peaked in 2012 when rates of these prescriptions began to fall. At that time a second wave, attributed to patients switching to less expensive nonprescription heroin, was already underway. A third wave, attributed to growth in the use of synthetic opioids, such as fentanyl, began in 2013 and is ongoing, according to data cited in the AHA statement.
Recognizing the role of opioids in rising rates of cardiac arrest is important for promoting strategies of effective treatment and prevention, according to Cameron Dezfulian, MD, medical director of the adult congenital heart disease program at Texas Children’s Hospital, Houston. Dr. Dezfulian was vice chair and leader of the writing committee for the AHA scientific statement on OA-OHCA. He said there are plenty of data to support the need for greater attention to the role of opioids in cardiac arrest.
“The recent data affirms the trends many of us have observed without our emergency rooms and ICUs: a steady increase in the proportion of OA-OHCA, primarily in young and otherwise healthy individuals,” he said.
He calls not only for more awareness at the front lines of health are but also for a more comprehensive approach.
“Public health policies and community- and hospital-based interventions are needed to reduce the mortality due to OA-OHCA, which is distinct from the traditional cardiac etiology,” Dr. Dezfulian said.
In opioid-induced cardiac arrest, as in other types of cardiac arrest, prompt initiation of cardiopulmonary resuscitation is essential, but early administration of the opioid antagonist naloxone can also be lifesaving, according to treatment strategies outlined in the AHA scientific statement. The fact that OA-OHCA typically occur in patients with structurally and electrophysiologically normal hearts is emphasized in the AHA statement. So is the enormous public health toll of OA-OHCA.
Death due to opioid overdose, which includes cardiac arrests, is now the leading cause of mortality in the U.S. among individuals between the ages of 25 and 64 years, according to the statement.
Ms. Malik reports no potential conflicts of interest. Dr. Dezfulian reports a financial relationship with Mallinckrodt.
FROM ESC 2021
Sweeping new vaccine mandates will impact most U.S. workers
, including sweeping vaccine mandates that will affect 100 million American workers, nearly two-thirds of the country’s workforce.
“As your president, I’m announcing tonight a new plan to get more Americans vaccinated to combat those blocking public health,” he said Sept. 9.
As part of a six-part plan unveiled in a speech from the State Dining Room of the White House, President Biden said he would require vaccinations for nearly 4 million federal workers and the employees of companies that contract with the federal government.
He has also directed the Occupational Safety and Health Administration to develop a rule that will require large employers -- those with at least 100 employees -- to ensure their workers are vaccinated or tested weekly.
Nearly 17 million health care workers will face new vaccine mandates as part of the conditions of participation in the Medicare and Medicaid programs.
President Biden said the federal government will require staff at federally funded Head Start programs and schools to be vaccinated. He’s also calling on all states to mandate vaccines for teachers.
“A distinct minority of Americans, supported by a distinct minority of elected officials, are keeping us from turning the corner,” PresidentBiden said. “These pandemic politics, as I refer to them, are making people sick, causing unvaccinated people to die.”
One public health official said he was glad to see the president’s bold action.
“What I saw today was the federal government trying to use its powers to create greater safety in the American population,” said Ashish K. Jha, MD, dean of the school of public health at Brown University, Providence, R.I., in a call with reporters after the speech.
National Nurses United, the largest union of registered nurses in the United States, issued a statement in support of President Biden’s new vaccination requirements, but pushed back on his language.
“…as advocates for public health, registered nurses want to be extremely clear: There is no such thing as a pandemic of only the unvaccinated. The science of epidemiology tells us there is just one deadly, global pandemic that has not yet ended, and we are all in it together. To get out of it, we must act together. All of us,” the statement says.
A host of other professional groups, including the American Medical Association and the Association of State and Territorial Health Officials, also issued statements of support for President Biden’s plan.
But the plan was not well received by all.
“I will pursue every legal option available to the state of Georgia to stop this blatantly unlawful overreach by the Biden Administration,” said Georgia Governor Brian Kemp, a Republican, in a Tweet.
The National Council for Occupational Safety and Health called the plan “a missed opportunity” because it failed to include workplace protections for essential workers such as grocery, postal, and transit workers.
“Social distancing, improved ventilation, shift rotation, and protective equipment to reduce exposure are important components of an overall plan to reduce risk and stop the virus. These tools are missing from the new steps President Biden announced today,” said Jessica Martinez, co-executive director of the group.
In addition to the new vaccination requirements, President Biden said extra doses would be on the way for people who have already been fully vaccinated in order to protect against waning immunity, starting on Sept. 20. But he noted that those plans would be contingent on the Food and Drug Administration’s approval for third doses and the Centers for Disease Control and Prevention’s recommendation of the shots.
President Biden pledged to use the Defense Production Act to ramp up production of at-home tests, which have been selling out across the nation as the Delta variant spreads.
He also announced plans to expand access to COVID-19 testing, including offering testing for free at thousands of pharmacies nationwide and getting major retailers to sell at-home COVID-19 tests at cost.
The BinaxNow test kit, which currently retails for $23.99, will now cost about $15 for two tests at Kroger, Amazon, and Walmart, according to the White House. Food banks and community health centers will get free tests, too.
He called on states to set up COVID-19 testing programs at all schools.
Jha said that in his view, the big, game-changing news out of the president’s speech was the expansion of testing.
“Our country has failed to deploy tests in a way that can really bring this pandemic under control,” Jha said. “There are plenty of reasons, data, experience to indicate that if these were widely available, it would make a dramatic difference in reducing infection numbers across our country.”.
Dr. Jha said the private market had not worked effectively to make testing more widely available, so it was “absolutely a requirement of the federal government to step in and make testing more widely available,” he said.
President Biden also announced new economic stimulus programs, saying he’s expanding loan programs to small businesses and streamlining the loan forgiveness process.
President Biden said he’s boosting help for overburdened hospitals, doubling the number of federal surge response teams sent to hard-hit areas to reduce the strain on local health care workers. He said he would increase the pace of antibody treatments to states by 50%.
“We made so much progress during the past 7 months of this pandemic. Even so, we remain at a critical moment, a critical time,” he said. “We have the tools. Now, we just have to finish the job with truth, with science, with confidence and together as one nation.”
A version of this article first appeared on WebMD.com.
, including sweeping vaccine mandates that will affect 100 million American workers, nearly two-thirds of the country’s workforce.
“As your president, I’m announcing tonight a new plan to get more Americans vaccinated to combat those blocking public health,” he said Sept. 9.
As part of a six-part plan unveiled in a speech from the State Dining Room of the White House, President Biden said he would require vaccinations for nearly 4 million federal workers and the employees of companies that contract with the federal government.
He has also directed the Occupational Safety and Health Administration to develop a rule that will require large employers -- those with at least 100 employees -- to ensure their workers are vaccinated or tested weekly.
Nearly 17 million health care workers will face new vaccine mandates as part of the conditions of participation in the Medicare and Medicaid programs.
President Biden said the federal government will require staff at federally funded Head Start programs and schools to be vaccinated. He’s also calling on all states to mandate vaccines for teachers.
“A distinct minority of Americans, supported by a distinct minority of elected officials, are keeping us from turning the corner,” PresidentBiden said. “These pandemic politics, as I refer to them, are making people sick, causing unvaccinated people to die.”
One public health official said he was glad to see the president’s bold action.
“What I saw today was the federal government trying to use its powers to create greater safety in the American population,” said Ashish K. Jha, MD, dean of the school of public health at Brown University, Providence, R.I., in a call with reporters after the speech.
National Nurses United, the largest union of registered nurses in the United States, issued a statement in support of President Biden’s new vaccination requirements, but pushed back on his language.
“…as advocates for public health, registered nurses want to be extremely clear: There is no such thing as a pandemic of only the unvaccinated. The science of epidemiology tells us there is just one deadly, global pandemic that has not yet ended, and we are all in it together. To get out of it, we must act together. All of us,” the statement says.
A host of other professional groups, including the American Medical Association and the Association of State and Territorial Health Officials, also issued statements of support for President Biden’s plan.
But the plan was not well received by all.
“I will pursue every legal option available to the state of Georgia to stop this blatantly unlawful overreach by the Biden Administration,” said Georgia Governor Brian Kemp, a Republican, in a Tweet.
The National Council for Occupational Safety and Health called the plan “a missed opportunity” because it failed to include workplace protections for essential workers such as grocery, postal, and transit workers.
“Social distancing, improved ventilation, shift rotation, and protective equipment to reduce exposure are important components of an overall plan to reduce risk and stop the virus. These tools are missing from the new steps President Biden announced today,” said Jessica Martinez, co-executive director of the group.
In addition to the new vaccination requirements, President Biden said extra doses would be on the way for people who have already been fully vaccinated in order to protect against waning immunity, starting on Sept. 20. But he noted that those plans would be contingent on the Food and Drug Administration’s approval for third doses and the Centers for Disease Control and Prevention’s recommendation of the shots.
President Biden pledged to use the Defense Production Act to ramp up production of at-home tests, which have been selling out across the nation as the Delta variant spreads.
He also announced plans to expand access to COVID-19 testing, including offering testing for free at thousands of pharmacies nationwide and getting major retailers to sell at-home COVID-19 tests at cost.
The BinaxNow test kit, which currently retails for $23.99, will now cost about $15 for two tests at Kroger, Amazon, and Walmart, according to the White House. Food banks and community health centers will get free tests, too.
He called on states to set up COVID-19 testing programs at all schools.
Jha said that in his view, the big, game-changing news out of the president’s speech was the expansion of testing.
“Our country has failed to deploy tests in a way that can really bring this pandemic under control,” Jha said. “There are plenty of reasons, data, experience to indicate that if these were widely available, it would make a dramatic difference in reducing infection numbers across our country.”.
Dr. Jha said the private market had not worked effectively to make testing more widely available, so it was “absolutely a requirement of the federal government to step in and make testing more widely available,” he said.
President Biden also announced new economic stimulus programs, saying he’s expanding loan programs to small businesses and streamlining the loan forgiveness process.
President Biden said he’s boosting help for overburdened hospitals, doubling the number of federal surge response teams sent to hard-hit areas to reduce the strain on local health care workers. He said he would increase the pace of antibody treatments to states by 50%.
“We made so much progress during the past 7 months of this pandemic. Even so, we remain at a critical moment, a critical time,” he said. “We have the tools. Now, we just have to finish the job with truth, with science, with confidence and together as one nation.”
A version of this article first appeared on WebMD.com.
, including sweeping vaccine mandates that will affect 100 million American workers, nearly two-thirds of the country’s workforce.
“As your president, I’m announcing tonight a new plan to get more Americans vaccinated to combat those blocking public health,” he said Sept. 9.
As part of a six-part plan unveiled in a speech from the State Dining Room of the White House, President Biden said he would require vaccinations for nearly 4 million federal workers and the employees of companies that contract with the federal government.
He has also directed the Occupational Safety and Health Administration to develop a rule that will require large employers -- those with at least 100 employees -- to ensure their workers are vaccinated or tested weekly.
Nearly 17 million health care workers will face new vaccine mandates as part of the conditions of participation in the Medicare and Medicaid programs.
President Biden said the federal government will require staff at federally funded Head Start programs and schools to be vaccinated. He’s also calling on all states to mandate vaccines for teachers.
“A distinct minority of Americans, supported by a distinct minority of elected officials, are keeping us from turning the corner,” PresidentBiden said. “These pandemic politics, as I refer to them, are making people sick, causing unvaccinated people to die.”
One public health official said he was glad to see the president’s bold action.
“What I saw today was the federal government trying to use its powers to create greater safety in the American population,” said Ashish K. Jha, MD, dean of the school of public health at Brown University, Providence, R.I., in a call with reporters after the speech.
National Nurses United, the largest union of registered nurses in the United States, issued a statement in support of President Biden’s new vaccination requirements, but pushed back on his language.
“…as advocates for public health, registered nurses want to be extremely clear: There is no such thing as a pandemic of only the unvaccinated. The science of epidemiology tells us there is just one deadly, global pandemic that has not yet ended, and we are all in it together. To get out of it, we must act together. All of us,” the statement says.
A host of other professional groups, including the American Medical Association and the Association of State and Territorial Health Officials, also issued statements of support for President Biden’s plan.
But the plan was not well received by all.
“I will pursue every legal option available to the state of Georgia to stop this blatantly unlawful overreach by the Biden Administration,” said Georgia Governor Brian Kemp, a Republican, in a Tweet.
The National Council for Occupational Safety and Health called the plan “a missed opportunity” because it failed to include workplace protections for essential workers such as grocery, postal, and transit workers.
“Social distancing, improved ventilation, shift rotation, and protective equipment to reduce exposure are important components of an overall plan to reduce risk and stop the virus. These tools are missing from the new steps President Biden announced today,” said Jessica Martinez, co-executive director of the group.
In addition to the new vaccination requirements, President Biden said extra doses would be on the way for people who have already been fully vaccinated in order to protect against waning immunity, starting on Sept. 20. But he noted that those plans would be contingent on the Food and Drug Administration’s approval for third doses and the Centers for Disease Control and Prevention’s recommendation of the shots.
President Biden pledged to use the Defense Production Act to ramp up production of at-home tests, which have been selling out across the nation as the Delta variant spreads.
He also announced plans to expand access to COVID-19 testing, including offering testing for free at thousands of pharmacies nationwide and getting major retailers to sell at-home COVID-19 tests at cost.
The BinaxNow test kit, which currently retails for $23.99, will now cost about $15 for two tests at Kroger, Amazon, and Walmart, according to the White House. Food banks and community health centers will get free tests, too.
He called on states to set up COVID-19 testing programs at all schools.
Jha said that in his view, the big, game-changing news out of the president’s speech was the expansion of testing.
“Our country has failed to deploy tests in a way that can really bring this pandemic under control,” Jha said. “There are plenty of reasons, data, experience to indicate that if these were widely available, it would make a dramatic difference in reducing infection numbers across our country.”.
Dr. Jha said the private market had not worked effectively to make testing more widely available, so it was “absolutely a requirement of the federal government to step in and make testing more widely available,” he said.
President Biden also announced new economic stimulus programs, saying he’s expanding loan programs to small businesses and streamlining the loan forgiveness process.
President Biden said he’s boosting help for overburdened hospitals, doubling the number of federal surge response teams sent to hard-hit areas to reduce the strain on local health care workers. He said he would increase the pace of antibody treatments to states by 50%.
“We made so much progress during the past 7 months of this pandemic. Even so, we remain at a critical moment, a critical time,” he said. “We have the tools. Now, we just have to finish the job with truth, with science, with confidence and together as one nation.”
A version of this article first appeared on WebMD.com.
Korean siblings face high familial IBD risk
Among Asian-Pacific populations, the first-degree relatives (FDRs) of individuals with inflammatory bowel disease (IBD) have a significantly increased risk for IBD themselves, according to a large analysis of data from South Korea. The greatest risk was found in siblings and for Crohn’s disease (CD).
The analysis of the South Korean Health Insurance Database included a cohort of 21,940,795 individuals from about 12 million families, with data collected between 2002 and 2017.
Previous studies have examined risk of IBD and familial relationships with existing IBD patients, but they have been subject to biases and have been heterogeneous in design, according to the authors, led by co–first authors Hyun Jung Kim, MD, of Korea University in Seoul, South Korea, and Shailja C. Shah, MD, of Vanderbilt University in Nashville, Tenn. There are few true population-based studies that quantify specific risks for family members of IBD patients, and none that were conducted in non-Western populations.
There are concerns about extrapolating familial IBD risk estimates from Western European populations to Asian populations because new data suggest that there are both genetic and nongenetic disease risk factors that reflect geography and ethnicity, the authors noted.
The researchers identified 45,717 individuals with ulcerative colitis (UC) and 17,848 with CD. Mean annual incidence rates were 4.6 cases of UC and 3.2 cases of CD per 100,000 person-years, which was relatively stable across the study period.
In all, 3.8% of UC and 3.1% of CD diagnoses occurred in FDR’s of existing patients. Among those with an FDR with IBD, the incidence of UC and CD was 54.5 and 99.2 per 100,000 person-years, respectively. When compared with individuals who had no FDRs with IBD, subjects who had an FDR with CD were at a more than 20-fold increased risk of CD (incident rate ratio, 22.2; 95% confidence interval, 20.5-24.5), whereas individuals with an FDR with UC were at a little more than a 10-fold risk for UC (IRR, 10.2; 95% CI, 9.39-11.1).
Subjects with an FDR with CD were at higher risk of UC (IRR, 3.56; 95% CI, 2.77-4.50), and those with an FDR with UC were at higher risk of CD (IRR, 2.94; 95% CI, 2.45-3.49). After adjustment for smoking, having an FDR with IBD was associated with an almost eightfold increased risk of UC (IRR, 7.94; 95% CI, 6.98-9.03) and a nearly 20-fold increased risk of CD (IRR, 19.03; 95% CI, 15.58-23.25).
The investigators also performed an analysis based on type of relative, with matching relations with unaffected relatives as the reference for each comparison. The highest risk for incident CD was with twin siblings (IRR, 336.2; 95% CI, 235.0-481.1) followed by nontwin siblings (IRR, 27.6; 95% CI, 24.6-30.9). The risk of CD among offspring of an affected father was 9.40 (95% CI, 6.81-13.0) and 6.54 (95% CI, 4.17-10.3) for offspring of affected mothers. There was a similar pattern for UC, although the magnitude was smaller: 163.7 for twin siblings (95% CI, 105.6-253.9), 13.1 for nontwin siblings (95% CI, 11.4-15.0), 7.11 for offspring of affected fathers (95% CI, 6.10-8.29), and 8.77 for offspring of affected mothers (95% CI, 7.46-10.3).
The researchers found no evidence of a birth cohort effect. Family history and IBD risk is a complicated relationship. Family history includes shared genetics as well as similar environmental exposures, and gene-environment interactions can add another layer of uncertainty. Previous studies have found that asymptomatic family members of IBD patients sometimes have preclinical signs such as changes in intestinal permeability, immune function, the microbiome, and biomarker levels.
IBD has emerged recently among Asian-Pacific populations as a serious health concern, with a recent rapid increase. This may reflect a shift in potentially modifiable environmental triggers. “Precisely quantifying familial risk and patterns might enable more accurate risk counseling and better-targeted clinical surveillance for earlier diagnosis and treatment among FDRs. Moreover, an accurate definition of familial IBD risk across populations also might inform subsequent investigations untangling the various shared environmental and genetic contributions,” the authors wrote.
Although genetic susceptibility is generally accepted as the predominant driver in familial trends for IBD, the authors noted their “study was not designed to determine the contribution of genetic vs. nongenetic determinants to familial IBD risk, and future well-designed dedicated investigations are needed to provide this clarity.”
The study is limited by the relatively short follow-up period, which may not have captured all IBD cases within patients’ families.
The authors have no relevant financial disclosures.
Among Asian-Pacific populations, the first-degree relatives (FDRs) of individuals with inflammatory bowel disease (IBD) have a significantly increased risk for IBD themselves, according to a large analysis of data from South Korea. The greatest risk was found in siblings and for Crohn’s disease (CD).
The analysis of the South Korean Health Insurance Database included a cohort of 21,940,795 individuals from about 12 million families, with data collected between 2002 and 2017.
Previous studies have examined risk of IBD and familial relationships with existing IBD patients, but they have been subject to biases and have been heterogeneous in design, according to the authors, led by co–first authors Hyun Jung Kim, MD, of Korea University in Seoul, South Korea, and Shailja C. Shah, MD, of Vanderbilt University in Nashville, Tenn. There are few true population-based studies that quantify specific risks for family members of IBD patients, and none that were conducted in non-Western populations.
There are concerns about extrapolating familial IBD risk estimates from Western European populations to Asian populations because new data suggest that there are both genetic and nongenetic disease risk factors that reflect geography and ethnicity, the authors noted.
The researchers identified 45,717 individuals with ulcerative colitis (UC) and 17,848 with CD. Mean annual incidence rates were 4.6 cases of UC and 3.2 cases of CD per 100,000 person-years, which was relatively stable across the study period.
In all, 3.8% of UC and 3.1% of CD diagnoses occurred in FDR’s of existing patients. Among those with an FDR with IBD, the incidence of UC and CD was 54.5 and 99.2 per 100,000 person-years, respectively. When compared with individuals who had no FDRs with IBD, subjects who had an FDR with CD were at a more than 20-fold increased risk of CD (incident rate ratio, 22.2; 95% confidence interval, 20.5-24.5), whereas individuals with an FDR with UC were at a little more than a 10-fold risk for UC (IRR, 10.2; 95% CI, 9.39-11.1).
Subjects with an FDR with CD were at higher risk of UC (IRR, 3.56; 95% CI, 2.77-4.50), and those with an FDR with UC were at higher risk of CD (IRR, 2.94; 95% CI, 2.45-3.49). After adjustment for smoking, having an FDR with IBD was associated with an almost eightfold increased risk of UC (IRR, 7.94; 95% CI, 6.98-9.03) and a nearly 20-fold increased risk of CD (IRR, 19.03; 95% CI, 15.58-23.25).
The investigators also performed an analysis based on type of relative, with matching relations with unaffected relatives as the reference for each comparison. The highest risk for incident CD was with twin siblings (IRR, 336.2; 95% CI, 235.0-481.1) followed by nontwin siblings (IRR, 27.6; 95% CI, 24.6-30.9). The risk of CD among offspring of an affected father was 9.40 (95% CI, 6.81-13.0) and 6.54 (95% CI, 4.17-10.3) for offspring of affected mothers. There was a similar pattern for UC, although the magnitude was smaller: 163.7 for twin siblings (95% CI, 105.6-253.9), 13.1 for nontwin siblings (95% CI, 11.4-15.0), 7.11 for offspring of affected fathers (95% CI, 6.10-8.29), and 8.77 for offspring of affected mothers (95% CI, 7.46-10.3).
The researchers found no evidence of a birth cohort effect. Family history and IBD risk is a complicated relationship. Family history includes shared genetics as well as similar environmental exposures, and gene-environment interactions can add another layer of uncertainty. Previous studies have found that asymptomatic family members of IBD patients sometimes have preclinical signs such as changes in intestinal permeability, immune function, the microbiome, and biomarker levels.
IBD has emerged recently among Asian-Pacific populations as a serious health concern, with a recent rapid increase. This may reflect a shift in potentially modifiable environmental triggers. “Precisely quantifying familial risk and patterns might enable more accurate risk counseling and better-targeted clinical surveillance for earlier diagnosis and treatment among FDRs. Moreover, an accurate definition of familial IBD risk across populations also might inform subsequent investigations untangling the various shared environmental and genetic contributions,” the authors wrote.
Although genetic susceptibility is generally accepted as the predominant driver in familial trends for IBD, the authors noted their “study was not designed to determine the contribution of genetic vs. nongenetic determinants to familial IBD risk, and future well-designed dedicated investigations are needed to provide this clarity.”
The study is limited by the relatively short follow-up period, which may not have captured all IBD cases within patients’ families.
The authors have no relevant financial disclosures.
Among Asian-Pacific populations, the first-degree relatives (FDRs) of individuals with inflammatory bowel disease (IBD) have a significantly increased risk for IBD themselves, according to a large analysis of data from South Korea. The greatest risk was found in siblings and for Crohn’s disease (CD).
The analysis of the South Korean Health Insurance Database included a cohort of 21,940,795 individuals from about 12 million families, with data collected between 2002 and 2017.
Previous studies have examined risk of IBD and familial relationships with existing IBD patients, but they have been subject to biases and have been heterogeneous in design, according to the authors, led by co–first authors Hyun Jung Kim, MD, of Korea University in Seoul, South Korea, and Shailja C. Shah, MD, of Vanderbilt University in Nashville, Tenn. There are few true population-based studies that quantify specific risks for family members of IBD patients, and none that were conducted in non-Western populations.
There are concerns about extrapolating familial IBD risk estimates from Western European populations to Asian populations because new data suggest that there are both genetic and nongenetic disease risk factors that reflect geography and ethnicity, the authors noted.
The researchers identified 45,717 individuals with ulcerative colitis (UC) and 17,848 with CD. Mean annual incidence rates were 4.6 cases of UC and 3.2 cases of CD per 100,000 person-years, which was relatively stable across the study period.
In all, 3.8% of UC and 3.1% of CD diagnoses occurred in FDR’s of existing patients. Among those with an FDR with IBD, the incidence of UC and CD was 54.5 and 99.2 per 100,000 person-years, respectively. When compared with individuals who had no FDRs with IBD, subjects who had an FDR with CD were at a more than 20-fold increased risk of CD (incident rate ratio, 22.2; 95% confidence interval, 20.5-24.5), whereas individuals with an FDR with UC were at a little more than a 10-fold risk for UC (IRR, 10.2; 95% CI, 9.39-11.1).
Subjects with an FDR with CD were at higher risk of UC (IRR, 3.56; 95% CI, 2.77-4.50), and those with an FDR with UC were at higher risk of CD (IRR, 2.94; 95% CI, 2.45-3.49). After adjustment for smoking, having an FDR with IBD was associated with an almost eightfold increased risk of UC (IRR, 7.94; 95% CI, 6.98-9.03) and a nearly 20-fold increased risk of CD (IRR, 19.03; 95% CI, 15.58-23.25).
The investigators also performed an analysis based on type of relative, with matching relations with unaffected relatives as the reference for each comparison. The highest risk for incident CD was with twin siblings (IRR, 336.2; 95% CI, 235.0-481.1) followed by nontwin siblings (IRR, 27.6; 95% CI, 24.6-30.9). The risk of CD among offspring of an affected father was 9.40 (95% CI, 6.81-13.0) and 6.54 (95% CI, 4.17-10.3) for offspring of affected mothers. There was a similar pattern for UC, although the magnitude was smaller: 163.7 for twin siblings (95% CI, 105.6-253.9), 13.1 for nontwin siblings (95% CI, 11.4-15.0), 7.11 for offspring of affected fathers (95% CI, 6.10-8.29), and 8.77 for offspring of affected mothers (95% CI, 7.46-10.3).
The researchers found no evidence of a birth cohort effect. Family history and IBD risk is a complicated relationship. Family history includes shared genetics as well as similar environmental exposures, and gene-environment interactions can add another layer of uncertainty. Previous studies have found that asymptomatic family members of IBD patients sometimes have preclinical signs such as changes in intestinal permeability, immune function, the microbiome, and biomarker levels.
IBD has emerged recently among Asian-Pacific populations as a serious health concern, with a recent rapid increase. This may reflect a shift in potentially modifiable environmental triggers. “Precisely quantifying familial risk and patterns might enable more accurate risk counseling and better-targeted clinical surveillance for earlier diagnosis and treatment among FDRs. Moreover, an accurate definition of familial IBD risk across populations also might inform subsequent investigations untangling the various shared environmental and genetic contributions,” the authors wrote.
Although genetic susceptibility is generally accepted as the predominant driver in familial trends for IBD, the authors noted their “study was not designed to determine the contribution of genetic vs. nongenetic determinants to familial IBD risk, and future well-designed dedicated investigations are needed to provide this clarity.”
The study is limited by the relatively short follow-up period, which may not have captured all IBD cases within patients’ families.
The authors have no relevant financial disclosures.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Spiral Plaque on the Left Ankle
The Diagnosis: Recurrent Cutaneous T-Cell Lymphoma
The skin biopsy revealed alternating orthokeratosis and parakeratosis with mild to moderate spongiosis and intraepidermal vesiculation as well as individual and nested atypical mononuclear cells with moderately enlarged hyperchromatic nuclei in the epidermis. There was a superficial interstitial lymphocytic infiltrate with occasional enlarged cells (Figure, A and B), and atypical cells in the epidermis and dermis stained with antibodies against CD3 and CD4 (Figure, C and D) but not against CD20 or CD8. These histopathologic findings were consistent with cutaneous T-cell lymphoma (CTCL), mycosis fungoides (MF) type. Additional application of bexarotene gel on days the patient received narrowband UVB was recommended with noted improvement of the skin.
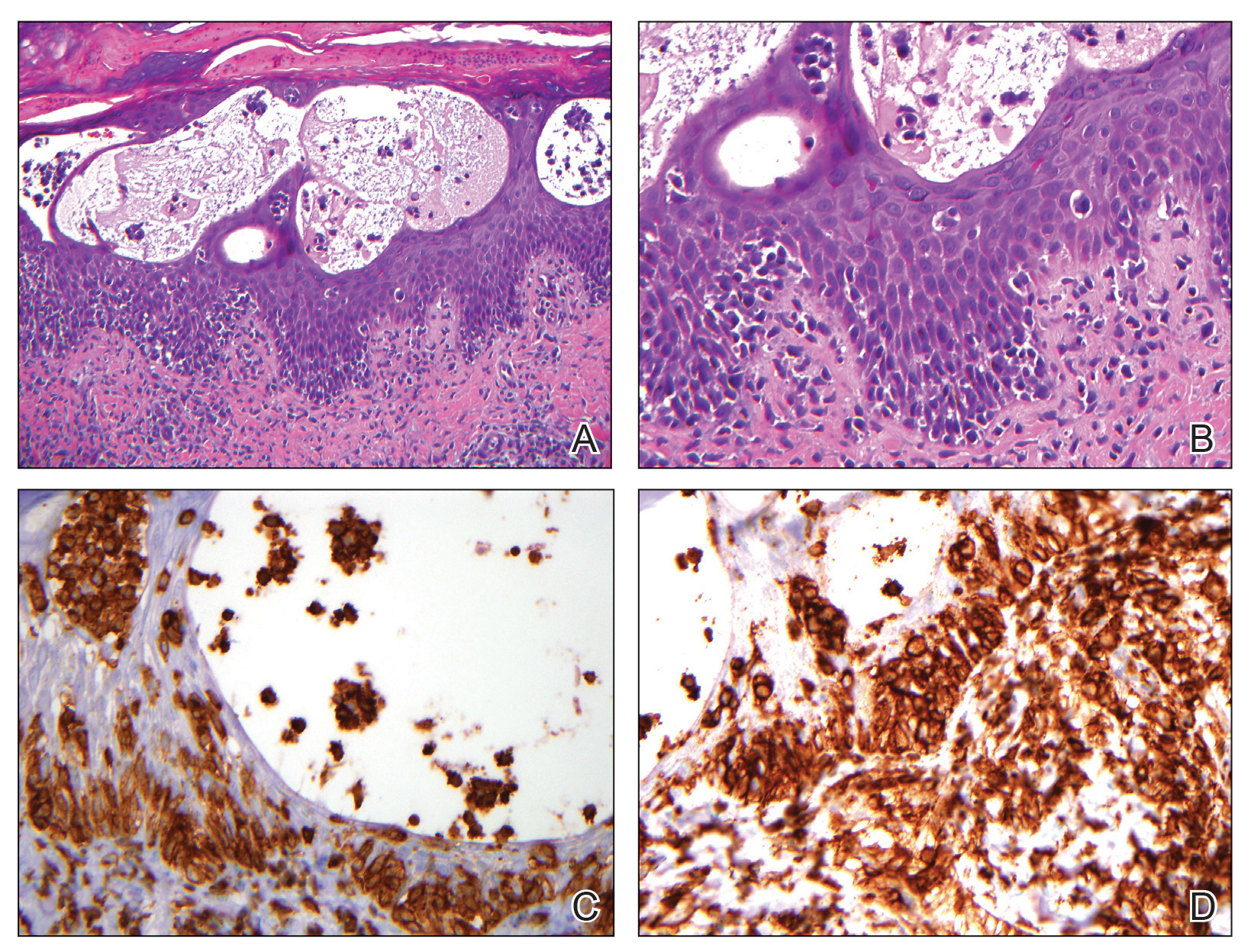
Cutaneous T-cell lymphomas are a heterogenous group of diseases with monoclonal proliferation of T lymphocytes that largely are confined to the skin at the time of diagnosis.1 The incidence of CTCL rose steadily for more than 25 years, with an annual age-adjusted incidence of 6.4 to 9.6 cases per million individuals in the United States from 1973 to 2002.2 Mycosis fungoides is the most common classification of CTCL. It usually is characterized by patches or plaques of scaly erythema or poikiloderma; however, it also can present with annular, arcuate, concentrative, annular and linear morphologies. Mycosis fungoides tumor cells typically express a mature memory T helper cell phenotype of CD3+, CD4+, and CD8−, but there are different variants that have been discovered.3 Mycosis fungoides distributed in a spiral pattern is a distinctly unusual manifestation. Mechanisms of such dynamic morphologies are unknown but may represent an interplay between malignant cell proliferation and lost immune responses in temporospatial relationships.
The presence of keratotic gyrate lesions on acral surfaces should raise the possibility of pagetoid reticulosis. However, our patient had a history of MF involving areas of the body beyond the extremities, making this diagnosis less likely. Pagetoid reticulosis is categorized as an MF variant under the current World Health Organization– European Organization for Research and Treatment of Cancer classification for cutaneous lymphomas.4 Pagetoid reticulosis clinically presents as a solitary psoriasiform or hyperkeratotic patch or plaque that affects the distal extremities. Variable immunophenotypes have been shown in pagetoid reticulosis, such as CD4−/CD8+ and CD4−/CD8−, while classic MF typically shows CD4+/CD8−, as in our case.5
Tinea pedis is a superficial fungal infection usually caused by anthropophilic dermatophytes, with Trichophyton rubrum being the most common organism. Four common clinical presentations of tinea pedis have been identified: interdigital, moccasin, vesicular, and acute ulcerative. Clinical presentation ranges from macerations, ulcerations, and erosions in the toe web spaces to dry hyperkeratotic scaling and fissures on the plantar foot.6 Tinea pedis primarily affects the plantar and interdigital spaces, sparing the dorsal foot and ankle. Treatment is recommended to alleviate symptoms and limit the spread of infection; topical antifungals for 4 weeks is the treatment of choice. However, recurrence is common, and maintenance therapy often is indicated. Oral antifungals or a combination of both topical and oral medications may be needed in certain cases.7
Erythema annulare centrifugum (EAC) is a rare dermatologic disease described as erythematous or urticarial papules that can enlarge centrifugally to form annular lesions that clear centrally. Thought to be a hypersensitivity reaction to an underlying condition, EAC has been associated with fungal infections, various cutaneous diseases, and even internal malignancies. Clinically, EAC can be divided into 2 forms: deep and superficial. Deep gyrate erythema is characterized by a firm indurated border with rare scaling and pruritus that histologically shows perivascular lymphocytic infiltration in the upper and deep dermis. Superficial gyrate erythema has minimally elevated lesions with an indistinct border and trailing scales and pruritus; histopathologic findings present a dense, perivascular, lymphocytic infiltration restricted to the upper dermis.8 Therapy for EAC is directed at relieving symptoms and treating the underlying condition if there is one associated.
Granuloma annulare (GA) is a common skin disorder classically characterized by ringed erythematous plaques, though many variants have been identified. Localized GA is the most common variant and presents with pink-red, nonscaly, annular patches or plaques, typically affecting the hands and feet. Generalized GA is characterized as diffuse annular patches or plaques classically affecting the trunk and extremities. Histology is notable for mucin with a palisading or interstitial pattern of granulomatous inflammation, which was not evident in our patient.9 Topical or intralesional corticosteroids are the first-line treatment of localized GA; however, localized GA generally is self-limited, and treatment often is not necessary. Treatment with cryosurgery, laser therapy, and topical dapsone and tacrolimus also has been described, but evidence of the efficacy of these agents is limited. For generalized GA, phototherapy currently is the most reliable therapy. Systemic therapies include antimalarials, fumaric acid esters, biologics, antimicrobials, and isotretinoin.10
Erythema gyratum repens (EGR) is a rare dermatologic disease described as erythematous concentric bands arranged in parallel rings that can be annular, figurate, or gyrate, with a fine scale trailing the leading edge. Histopathologic features of EGR are nonspecific but are characterized by a perivascular, superficial, mononuclear dermatitis. Diagnosis is based on its characteristic clinical presentation. Although EGR commonly is associated with internal malignancies such as bronchial carcinoma, it also may be associated with benign conditions.11 Improvement often is seen with successful therapy of the underlying associated malignancy.12
Treatment of MF is based on tumor-node-metastasisblood classification, prognostic factors, and clinical stage at the time of diagnosis. Early-stage MF (IA–IIA) commonly is treated with skin-directed therapies such as topical corticosteroids, topical mechlorethamine, topical retinoids, UV phototherapy, and localized radiotherapy. In late stages (IIB–IV), systemic therapy is indicated and includes systemic retinoids, interferon alfa, chemotherapy, monoclonal antibodies, and psoralen plus UVA.13 In many cases, patients may require combination therapy to achieve remission or better control of their condition, as in our patient.
The Diagnosis: Recurrent Cutaneous T-Cell Lymphoma
The skin biopsy revealed alternating orthokeratosis and parakeratosis with mild to moderate spongiosis and intraepidermal vesiculation as well as individual and nested atypical mononuclear cells with moderately enlarged hyperchromatic nuclei in the epidermis. There was a superficial interstitial lymphocytic infiltrate with occasional enlarged cells (Figure, A and B), and atypical cells in the epidermis and dermis stained with antibodies against CD3 and CD4 (Figure, C and D) but not against CD20 or CD8. These histopathologic findings were consistent with cutaneous T-cell lymphoma (CTCL), mycosis fungoides (MF) type. Additional application of bexarotene gel on days the patient received narrowband UVB was recommended with noted improvement of the skin.

Cutaneous T-cell lymphomas are a heterogenous group of diseases with monoclonal proliferation of T lymphocytes that largely are confined to the skin at the time of diagnosis.1 The incidence of CTCL rose steadily for more than 25 years, with an annual age-adjusted incidence of 6.4 to 9.6 cases per million individuals in the United States from 1973 to 2002.2 Mycosis fungoides is the most common classification of CTCL. It usually is characterized by patches or plaques of scaly erythema or poikiloderma; however, it also can present with annular, arcuate, concentrative, annular and linear morphologies. Mycosis fungoides tumor cells typically express a mature memory T helper cell phenotype of CD3+, CD4+, and CD8−, but there are different variants that have been discovered.3 Mycosis fungoides distributed in a spiral pattern is a distinctly unusual manifestation. Mechanisms of such dynamic morphologies are unknown but may represent an interplay between malignant cell proliferation and lost immune responses in temporospatial relationships.
The presence of keratotic gyrate lesions on acral surfaces should raise the possibility of pagetoid reticulosis. However, our patient had a history of MF involving areas of the body beyond the extremities, making this diagnosis less likely. Pagetoid reticulosis is categorized as an MF variant under the current World Health Organization– European Organization for Research and Treatment of Cancer classification for cutaneous lymphomas.4 Pagetoid reticulosis clinically presents as a solitary psoriasiform or hyperkeratotic patch or plaque that affects the distal extremities. Variable immunophenotypes have been shown in pagetoid reticulosis, such as CD4−/CD8+ and CD4−/CD8−, while classic MF typically shows CD4+/CD8−, as in our case.5
Tinea pedis is a superficial fungal infection usually caused by anthropophilic dermatophytes, with Trichophyton rubrum being the most common organism. Four common clinical presentations of tinea pedis have been identified: interdigital, moccasin, vesicular, and acute ulcerative. Clinical presentation ranges from macerations, ulcerations, and erosions in the toe web spaces to dry hyperkeratotic scaling and fissures on the plantar foot.6 Tinea pedis primarily affects the plantar and interdigital spaces, sparing the dorsal foot and ankle. Treatment is recommended to alleviate symptoms and limit the spread of infection; topical antifungals for 4 weeks is the treatment of choice. However, recurrence is common, and maintenance therapy often is indicated. Oral antifungals or a combination of both topical and oral medications may be needed in certain cases.7
Erythema annulare centrifugum (EAC) is a rare dermatologic disease described as erythematous or urticarial papules that can enlarge centrifugally to form annular lesions that clear centrally. Thought to be a hypersensitivity reaction to an underlying condition, EAC has been associated with fungal infections, various cutaneous diseases, and even internal malignancies. Clinically, EAC can be divided into 2 forms: deep and superficial. Deep gyrate erythema is characterized by a firm indurated border with rare scaling and pruritus that histologically shows perivascular lymphocytic infiltration in the upper and deep dermis. Superficial gyrate erythema has minimally elevated lesions with an indistinct border and trailing scales and pruritus; histopathologic findings present a dense, perivascular, lymphocytic infiltration restricted to the upper dermis.8 Therapy for EAC is directed at relieving symptoms and treating the underlying condition if there is one associated.
Granuloma annulare (GA) is a common skin disorder classically characterized by ringed erythematous plaques, though many variants have been identified. Localized GA is the most common variant and presents with pink-red, nonscaly, annular patches or plaques, typically affecting the hands and feet. Generalized GA is characterized as diffuse annular patches or plaques classically affecting the trunk and extremities. Histology is notable for mucin with a palisading or interstitial pattern of granulomatous inflammation, which was not evident in our patient.9 Topical or intralesional corticosteroids are the first-line treatment of localized GA; however, localized GA generally is self-limited, and treatment often is not necessary. Treatment with cryosurgery, laser therapy, and topical dapsone and tacrolimus also has been described, but evidence of the efficacy of these agents is limited. For generalized GA, phototherapy currently is the most reliable therapy. Systemic therapies include antimalarials, fumaric acid esters, biologics, antimicrobials, and isotretinoin.10
Erythema gyratum repens (EGR) is a rare dermatologic disease described as erythematous concentric bands arranged in parallel rings that can be annular, figurate, or gyrate, with a fine scale trailing the leading edge. Histopathologic features of EGR are nonspecific but are characterized by a perivascular, superficial, mononuclear dermatitis. Diagnosis is based on its characteristic clinical presentation. Although EGR commonly is associated with internal malignancies such as bronchial carcinoma, it also may be associated with benign conditions.11 Improvement often is seen with successful therapy of the underlying associated malignancy.12
Treatment of MF is based on tumor-node-metastasisblood classification, prognostic factors, and clinical stage at the time of diagnosis. Early-stage MF (IA–IIA) commonly is treated with skin-directed therapies such as topical corticosteroids, topical mechlorethamine, topical retinoids, UV phototherapy, and localized radiotherapy. In late stages (IIB–IV), systemic therapy is indicated and includes systemic retinoids, interferon alfa, chemotherapy, monoclonal antibodies, and psoralen plus UVA.13 In many cases, patients may require combination therapy to achieve remission or better control of their condition, as in our patient.
The Diagnosis: Recurrent Cutaneous T-Cell Lymphoma
The skin biopsy revealed alternating orthokeratosis and parakeratosis with mild to moderate spongiosis and intraepidermal vesiculation as well as individual and nested atypical mononuclear cells with moderately enlarged hyperchromatic nuclei in the epidermis. There was a superficial interstitial lymphocytic infiltrate with occasional enlarged cells (Figure, A and B), and atypical cells in the epidermis and dermis stained with antibodies against CD3 and CD4 (Figure, C and D) but not against CD20 or CD8. These histopathologic findings were consistent with cutaneous T-cell lymphoma (CTCL), mycosis fungoides (MF) type. Additional application of bexarotene gel on days the patient received narrowband UVB was recommended with noted improvement of the skin.

Cutaneous T-cell lymphomas are a heterogenous group of diseases with monoclonal proliferation of T lymphocytes that largely are confined to the skin at the time of diagnosis.1 The incidence of CTCL rose steadily for more than 25 years, with an annual age-adjusted incidence of 6.4 to 9.6 cases per million individuals in the United States from 1973 to 2002.2 Mycosis fungoides is the most common classification of CTCL. It usually is characterized by patches or plaques of scaly erythema or poikiloderma; however, it also can present with annular, arcuate, concentrative, annular and linear morphologies. Mycosis fungoides tumor cells typically express a mature memory T helper cell phenotype of CD3+, CD4+, and CD8−, but there are different variants that have been discovered.3 Mycosis fungoides distributed in a spiral pattern is a distinctly unusual manifestation. Mechanisms of such dynamic morphologies are unknown but may represent an interplay between malignant cell proliferation and lost immune responses in temporospatial relationships.
The presence of keratotic gyrate lesions on acral surfaces should raise the possibility of pagetoid reticulosis. However, our patient had a history of MF involving areas of the body beyond the extremities, making this diagnosis less likely. Pagetoid reticulosis is categorized as an MF variant under the current World Health Organization– European Organization for Research and Treatment of Cancer classification for cutaneous lymphomas.4 Pagetoid reticulosis clinically presents as a solitary psoriasiform or hyperkeratotic patch or plaque that affects the distal extremities. Variable immunophenotypes have been shown in pagetoid reticulosis, such as CD4−/CD8+ and CD4−/CD8−, while classic MF typically shows CD4+/CD8−, as in our case.5
Tinea pedis is a superficial fungal infection usually caused by anthropophilic dermatophytes, with Trichophyton rubrum being the most common organism. Four common clinical presentations of tinea pedis have been identified: interdigital, moccasin, vesicular, and acute ulcerative. Clinical presentation ranges from macerations, ulcerations, and erosions in the toe web spaces to dry hyperkeratotic scaling and fissures on the plantar foot.6 Tinea pedis primarily affects the plantar and interdigital spaces, sparing the dorsal foot and ankle. Treatment is recommended to alleviate symptoms and limit the spread of infection; topical antifungals for 4 weeks is the treatment of choice. However, recurrence is common, and maintenance therapy often is indicated. Oral antifungals or a combination of both topical and oral medications may be needed in certain cases.7
Erythema annulare centrifugum (EAC) is a rare dermatologic disease described as erythematous or urticarial papules that can enlarge centrifugally to form annular lesions that clear centrally. Thought to be a hypersensitivity reaction to an underlying condition, EAC has been associated with fungal infections, various cutaneous diseases, and even internal malignancies. Clinically, EAC can be divided into 2 forms: deep and superficial. Deep gyrate erythema is characterized by a firm indurated border with rare scaling and pruritus that histologically shows perivascular lymphocytic infiltration in the upper and deep dermis. Superficial gyrate erythema has minimally elevated lesions with an indistinct border and trailing scales and pruritus; histopathologic findings present a dense, perivascular, lymphocytic infiltration restricted to the upper dermis.8 Therapy for EAC is directed at relieving symptoms and treating the underlying condition if there is one associated.
Granuloma annulare (GA) is a common skin disorder classically characterized by ringed erythematous plaques, though many variants have been identified. Localized GA is the most common variant and presents with pink-red, nonscaly, annular patches or plaques, typically affecting the hands and feet. Generalized GA is characterized as diffuse annular patches or plaques classically affecting the trunk and extremities. Histology is notable for mucin with a palisading or interstitial pattern of granulomatous inflammation, which was not evident in our patient.9 Topical or intralesional corticosteroids are the first-line treatment of localized GA; however, localized GA generally is self-limited, and treatment often is not necessary. Treatment with cryosurgery, laser therapy, and topical dapsone and tacrolimus also has been described, but evidence of the efficacy of these agents is limited. For generalized GA, phototherapy currently is the most reliable therapy. Systemic therapies include antimalarials, fumaric acid esters, biologics, antimicrobials, and isotretinoin.10
Erythema gyratum repens (EGR) is a rare dermatologic disease described as erythematous concentric bands arranged in parallel rings that can be annular, figurate, or gyrate, with a fine scale trailing the leading edge. Histopathologic features of EGR are nonspecific but are characterized by a perivascular, superficial, mononuclear dermatitis. Diagnosis is based on its characteristic clinical presentation. Although EGR commonly is associated with internal malignancies such as bronchial carcinoma, it also may be associated with benign conditions.11 Improvement often is seen with successful therapy of the underlying associated malignancy.12
Treatment of MF is based on tumor-node-metastasisblood classification, prognostic factors, and clinical stage at the time of diagnosis. Early-stage MF (IA–IIA) commonly is treated with skin-directed therapies such as topical corticosteroids, topical mechlorethamine, topical retinoids, UV phototherapy, and localized radiotherapy. In late stages (IIB–IV), systemic therapy is indicated and includes systemic retinoids, interferon alfa, chemotherapy, monoclonal antibodies, and psoralen plus UVA.13 In many cases, patients may require combination therapy to achieve remission or better control of their condition, as in our patient.
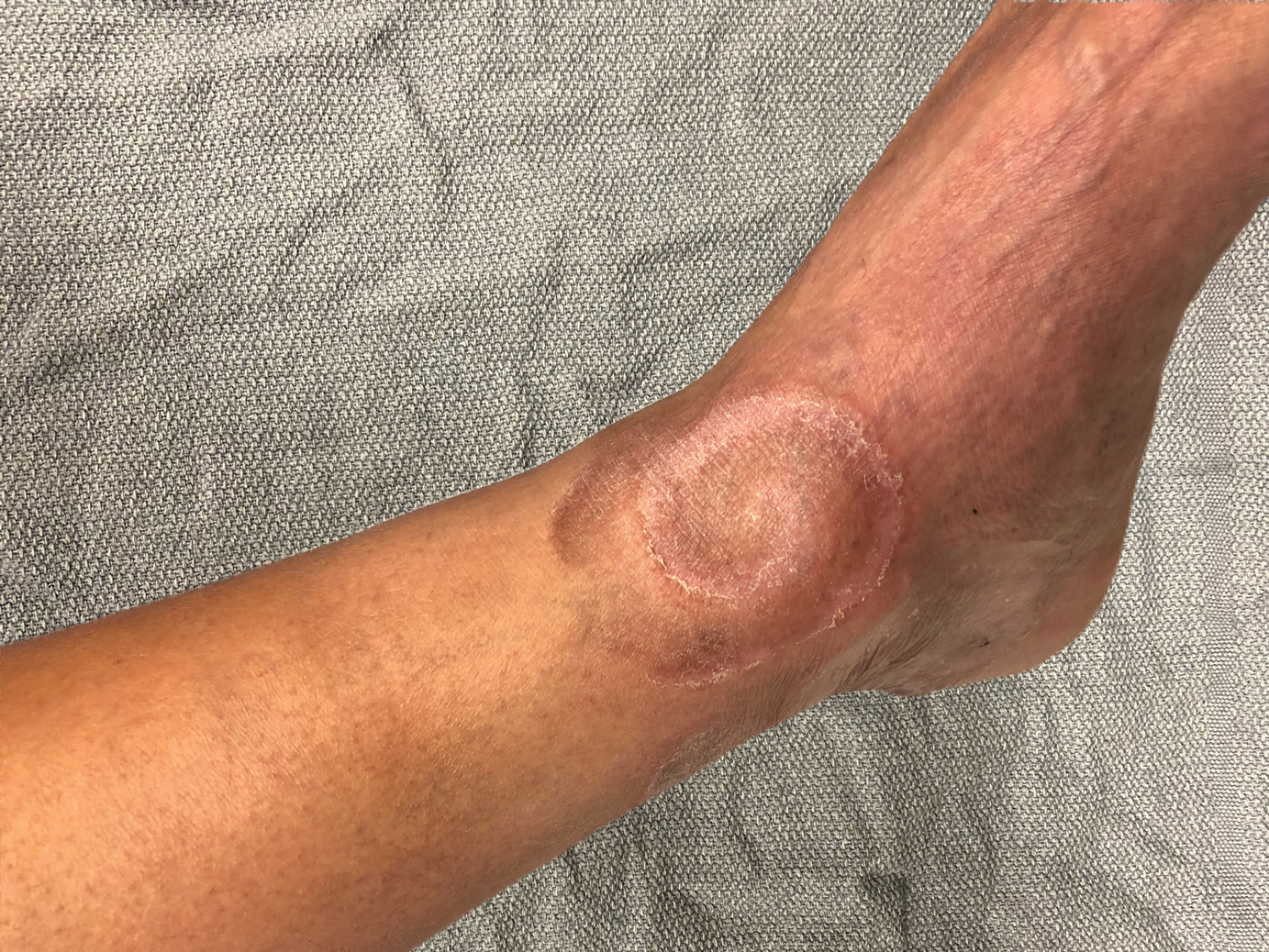
A 60-year-old man presented with a whorl-like plaque on the left ankle that he had noticed while undergoing treatment with narrowband UVB every other week and nitrogen mustard gel daily for stage IB cutaneous T-cell lymphoma, mycosis fungoides type. He denied pain, pruritus, and any other associated symptoms at the site. He denied recent illness, new medications, or changes in diet. His medical history included multiple sclerosis, vascular disease, and stroke. Physical examination revealed an 8×6-cm, welldemarcated, slightly scaly, erythematous plaque with a spiral appearance and peripheral hyperpigmentation involving the left ankle. The remainder of the examination was notable for well-controlled mycosis fungoides with several hyperpigmented patches at sites of prior involvement on the trunk and upper and lower extremities. No cervical, axillary, or inguinal lymphadenopathy was noted. A 4-mm punch biopsy was performed and sent for histopathologic examination.