User login
For MD-IQ only
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Prostate cancer is the most common noncutaneous cancer in men, accounting for 29% of all incident cancer cases.1 Typically, prostate cancer metastasizes to bone and regional lymph nodes.2 However, intrathoracic manifestation may occur. This report presents 3 cases of rare intrathoracic manifestations of metastatic prostate cancer with a review of the current literature.
CASE PRESENTATIONS
Case 1
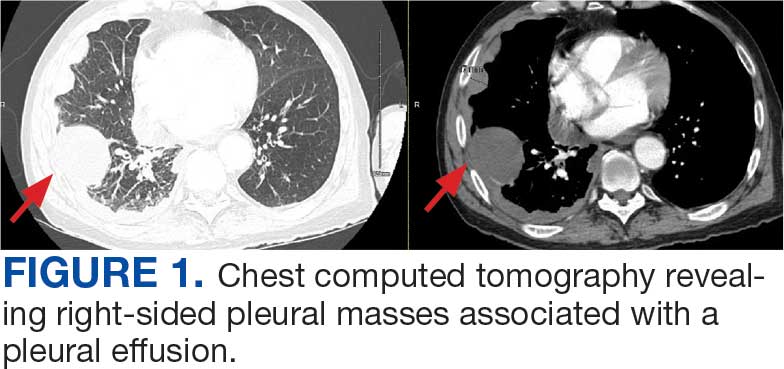
A 71-year-old male who was an active smoker and a long-standing employment as a plumber was diagnosed with rectal cancer in 2022. He completed neoadjuvant capecitabine and radiation therapy followed by a rectosigmoidectomy. Several weeks after surgery, the patient presented to the emergency department (ED) with a dry cough and worsening shortness of breath. Point-of-care ultrasound of the lungs revealed a moderate right pleural effusion with several nodular pleural masses. A chest computed tomography (CT) confirmed these findings (Figure 1). A CT of the abdomen and pelvis revealed prostatomegaly with the medial lobe of the prostate protruding into the bladder; however, no enlarged retroperitoneal, mesenteric or pelvic lymph nodes were noted. The patient underwent a right pleural fluid drainage and pleural mass biopsy. Pleural mass histomorphology as well as immunohistochemical (IHC) stains were consistent with metastatic prostate adenocarcinoma. The pleural fluid cytology also was consistent with metastatic prostate adenocarcinoma.
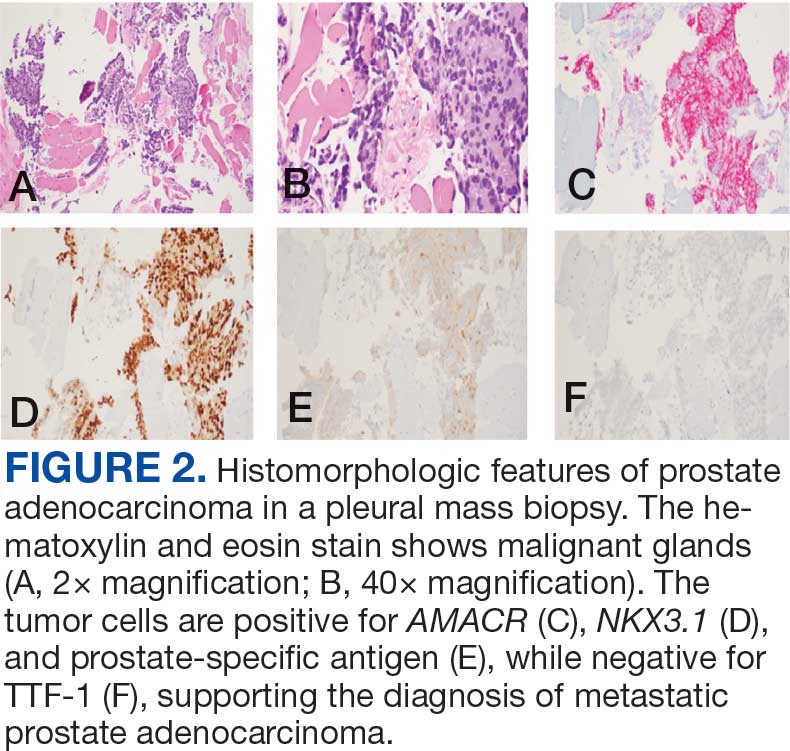
Immunohistochemistry showed weak positive staining for prostate-specific NK3 homeobox 1 gene (NKX3.1), alpha-methylacyl-CoA racemase gene (AMACR), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), keratin-20, and caudal type homeobox 2 gene (CDX2) (Figure 2) 2). The patient's prostate-specific antigen (PSA) was found to be elevated at 33.9 ng/mL (reference range, < 4 ng/mL).
Case 2
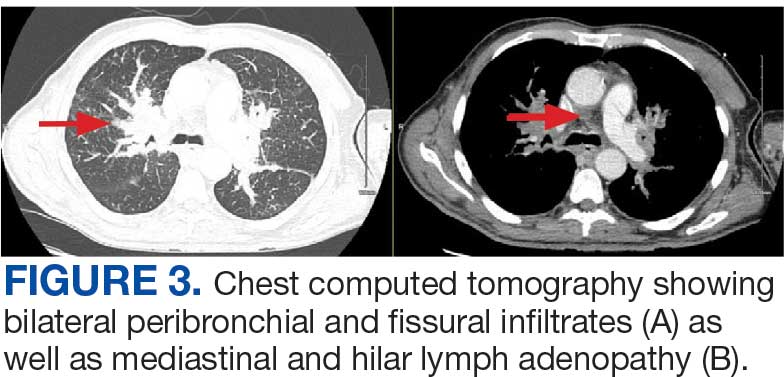
A 71-year-old male with a history of alcohol use disorder and a 30-year smoking history presented to the ED with worsening dyspnea on exertion. The patient’s baseline exercise tolerance decreased to walking for only 1 block. He reported unintentional weight loss of about 30 pounds over the prior year, no recent respiratory infections, no prior breathing problems, and no personal or family history of cancer. Chest CT revealed findings of bilateral peribronchial opacities as well as mediastinal and hilar lymphadenopathy (Figure 3). The patient developed hypoxic respiratory failure necessitating intubation, mechanical ventilation, and management in the medical intensive care unit, where he was treated for postobstructive pneumonia. Fiberoptic bronchoscopy revealed endobronchial lesions in the right and left upper lobe that were partially obstructing the airway (Figure 4).
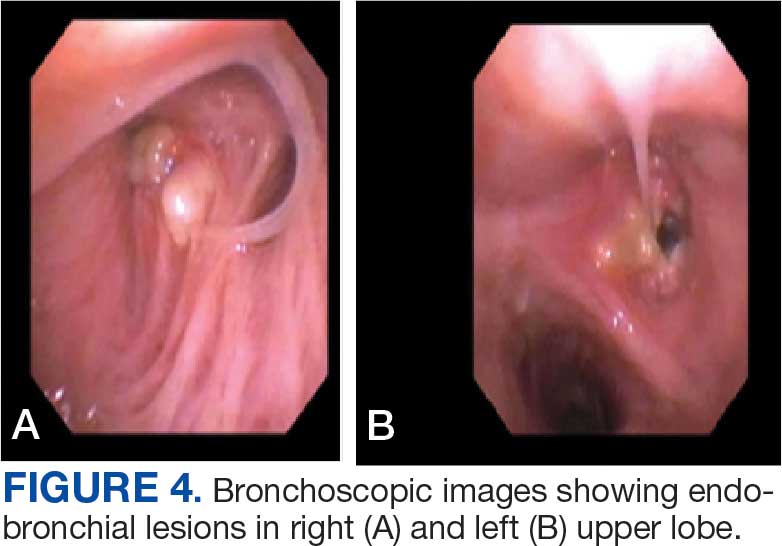
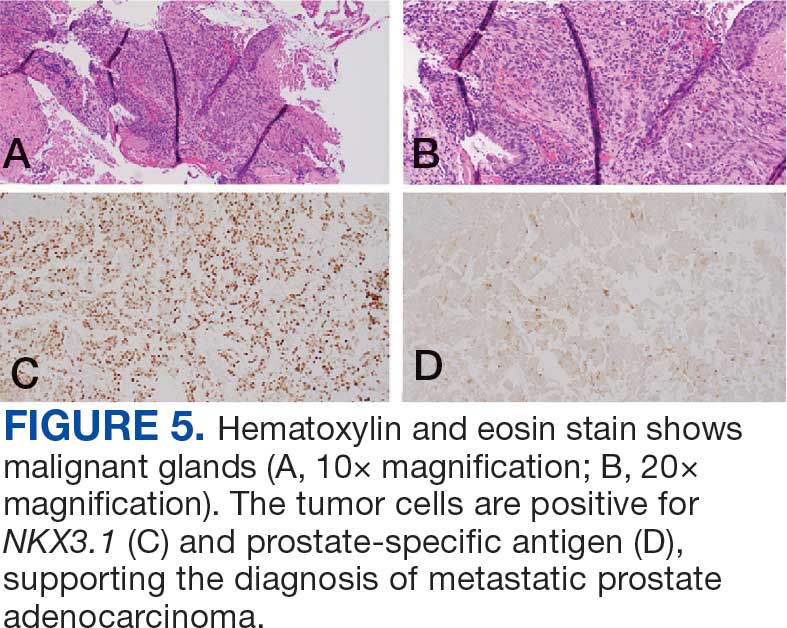
The endobronchial masses were debulked using forceps, and samples were sent for surgical pathology evaluation. Staging was completed using linear endobronchial ultrasound, which revealed an enlarged subcarinal lymph node (S7). The surgical pathology of the endobronchial mass and the subcarinal lymph node cytology were consistent with metastatic adenocarcinoma of the prostate. The tumor cells were positive for AE1/AE3, PSA, and NKX3.1, but were negative for CK7 and TTF-1 (Figure 5). Further imaging revealed an enlarged heterogeneous prostate gland, prominent pelvic nodes, and left retroperitoneal lymphadenopathy, as well as sclerotic foci within the T10 vertebral body and right inferior pubic ramus. PSA was also found to be significantly elevated at 700 ng/mL.
Case 3
An 80-year-old male veteran with a history of prostate cancer and recently diagnosed T2N1M0 head and neck squamous cell carcinoma was referred to the Pulmonary service for evaluation of a pulmonary nodule. His medical history was notable for prostate cancer diagnosed 12 years earlier, with an unknown Gleason score. Initial treatment included prostatectomy followed by whole pelvic radiation therapy a year after, due to elevated PSA in surveillance monitoring. This treatment led to remission. After establishing remission for > 10 years, the patient was started on low-dose testosterone replacement therapy to address complications of radiation therapy, namely hypogonadism.
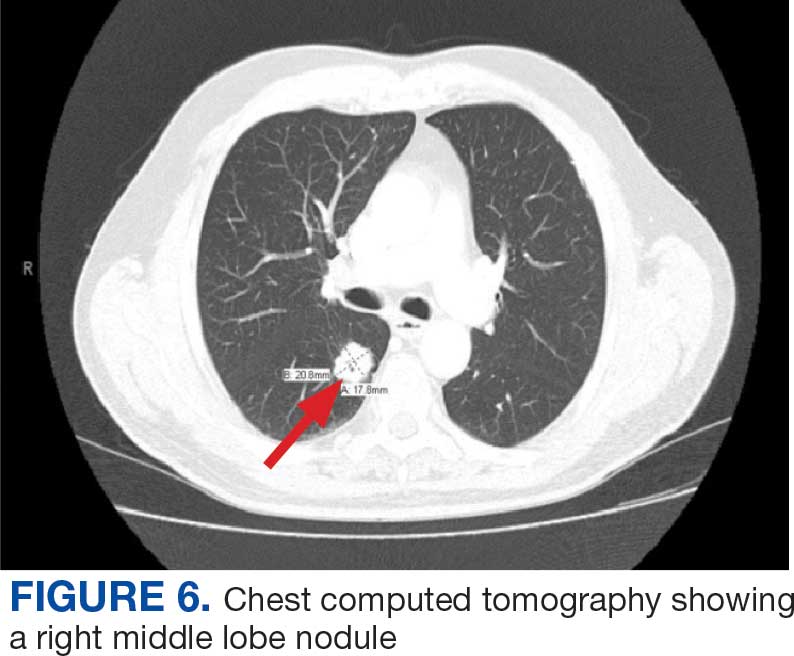
On evaluation, a chest CT was significant for a large 2-cm right middle lobe nodule (Figure 6). At that time, PSA was noted to be borderline elevated at 4.2 ng/mL, and whole-body imaging did not reveal any lesions elsewhere, specifically no bone metastasis. Biopsies of the right middle lobe lung nodule revealed adenocarcinoma consistent with metastatic prostate cancer. Testosterone therapy was promptly discontinued.
The patient initially refused androgen deprivation therapy owing to the antiandrogenic adverse effects. However, subsequent chest CTs revealed growing lung nodules, which convinced him to proceed with androgen deprivation therapy followed by palliative radiation, and chemotherapy and management of malignant pleural effusion with indwelling small bore pleural catheter for about 10 years. He died from COVID-19 during the pandemic.
DISCUSSION
These cases highlight the importance of including prostate cancer in the differential diagnoses of male patients with intrathoracic abnormalities, even in the absence of metastasis to the more common sites. In a large cohort study of 74,826 patients with metastatic prostate cancer, Gandaglia et al found that the most frequent sites of metastasis were bone (84.0%) and distant lymph nodes (10.6%).2 However, thoracic involvement was observed in 9.1% of cases, with isolated thoracic metastasis being rare. The cases described in this report exemplify exceptionally uncommon occurrences within that 9.1%.
Pleural metastases, as observed in Case 1, are a particularly rare manifestation. In a 10-year retrospective assessment, Vinjamoori et al discovered pleural nodules or masses in only 6 of 82 patients (7.3%) with atypical metastases.3 Adrenal and liver metastases accounted for 15% and 37% of cases with atypical distribution. As such, isolated pleural disease is rare even in atypical presentations.3
As seen in Case 2, endobronchial metastases producing airway obstruction are also rare, with the most common primary cancers associated with endobronchial metastasis being breast, colon, and renal cancer.4 The available literature on this presentation is confined to case reports. Hameed et al reported a case of synchronous biopsy-proven endobronchial metastasis from prostate cancer.5 These cases highlight the importance of maintaining a high level of clinical awareness when encountering endobronchial lesions in patients with prostate cancer.
Case 3 presents a unique situation of lung metastases without any involvement of the bones. It is well known—and was confirmed by Heidenreich et al—that lung metastases in prostate adenocarcinoma usually coincide with extensive osseous disease.6 This instance highlights the importance of watchful monitoring for unusual patterns of cancer recurrence.
Immunohistochemistry stains that are specific to prostate cancer include antibodies against PSA. Prostate-specific membrane antigen is another marker that is far more present in malignant than in benign prostate tissue.
The NKX3.1 gene encodes a homeobox protein, which is a transcription factor and tumor suppressor. In prostate cancer, there is loss of heterozygosity of the gene and stains for the IHC antibody to NKX3.1.7
On the other hand, lung cells stain positive for TTF-1, which is produced by surfactant-producing type 2 pneumocytes and club cells in the lung. Antibodies to TTF-1, a common IHC stain, are used to identify adenocarcinoma of lung origin and may carry a prognostic value.7
The immunohistochemistry profiles, specifically the presence of prostate-specific markers such as PSA and NKX3.1, played a vital role in making the diagnosis.
In Case 1, weak TTF-1 positivity was noted, an unusual finding in metastatic prostate adenocarcinoma. Marak et al documented a rare case of TTF-1–positive metastatic prostate cancer, illustrating the potential for diagnostic confusion with primary lung malignancies.8
The 3 cases described in this report demonstrate the importance of clinical consideration, serial follow-up of PSA levels, using more prostate-specific positron emission tomography tracers (eg, Pylarify) alongside traditional imaging, and tissue biopsy to detect unusual metastases.
CONCLUSIONS
Although thoracic metastases from prostate cancer are rare, these presentations highlight the importance of clinical awareness regarding atypical cases. Pleural disease, endobronchial lesions, and isolated pulmonary nodules might be the first clinical manifestation of metastatic prostate cancer. A high index of suspicion, appropriate imaging, and judicious use of immunohistochemistry are important to ensure accurate diagnosis and optimal patient management.
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49. doi:10.3322/caac.21820
- Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
- Vinjamoori AH, Jagannathan JP, Shinagare AB, et al. Atypical metastases from prostate cancer: 10-year experience at a single institution. AJR Am J Roentgenol. 2012;199(2):367-372. doi:10.2214/AJR.11.7533
- Salud A, Porcel JM, Rovirosa A, Bellmunt J. Endobronchial metastatic disease: analysis of 32 cases. J Surg Oncol. 1996;62(4):249-252. doi:10.1002/(SICI)1096- 9098(199608)62:4<249::AID-JSO4>3.0.CO;2-6
- Hameed M, Haq IU, Yousaf M, Hussein M, Rashid U, Al-Bozom I. Endobronchial metastases secondary to prostate cancer: a case report and literature review. Respir Med Case Rep. 2020;32:101326. doi:10.1016/j.rmcr.2020.101326
- Heidenreich A, Bastian PJ, Bellmunt J, et al; for the European Association of Urology. EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration- resistant prostate cancer. Eur Urol. 2014;65(2):467- 479. doi:10.1016/j.eururo.2013.11.002
- Schallenberg S, Dernbach G, Dragomir MP, et al. TTF-1 status in early-stage lung adenocarcinoma is an independent predictor of relapse and survival superior to tumor grading. Eur J Cancer. 2024;197:113474. doi:10.1016/j.ejca.2023.113474
- Marak C, Guddati AK, Ashraf A, Smith J, Kaushik P. Prostate adenocarcinoma with atypical immunohistochemistry presenting with a Cheerio sign. AIM Clinical Cases. 2023;1:e220508. doi:10.7326/aimcc.2022.0508
Prostate cancer is the most common noncutaneous cancer in men, accounting for 29% of all incident cancer cases.1 Typically, prostate cancer metastasizes to bone and regional lymph nodes.2 However, intrathoracic manifestation may occur. This report presents 3 cases of rare intrathoracic manifestations of metastatic prostate cancer with a review of the current literature.
CASE PRESENTATIONS
Case 1
A 71-year-old male who was an active smoker and a long-standing employment as a plumber was diagnosed with rectal cancer in 2022. He completed neoadjuvant capecitabine and radiation therapy followed by a rectosigmoidectomy. Several weeks after surgery, the patient presented to the emergency department (ED) with a dry cough and worsening shortness of breath. Point-of-care ultrasound of the lungs revealed a moderate right pleural effusion with several nodular pleural masses. A chest computed tomography (CT) confirmed these findings (Figure 1). A CT of the abdomen and pelvis revealed prostatomegaly with the medial lobe of the prostate protruding into the bladder; however, no enlarged retroperitoneal, mesenteric or pelvic lymph nodes were noted. The patient underwent a right pleural fluid drainage and pleural mass biopsy. Pleural mass histomorphology as well as immunohistochemical (IHC) stains were consistent with metastatic prostate adenocarcinoma. The pleural fluid cytology also was consistent with metastatic prostate adenocarcinoma.

Immunohistochemistry showed weak positive staining for prostate-specific NK3 homeobox 1 gene (NKX3.1), alpha-methylacyl-CoA racemase gene (AMACR), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), keratin-20, and caudal type homeobox 2 gene (CDX2) (Figure 2) 2). The patient's prostate-specific antigen (PSA) was found to be elevated at 33.9 ng/mL (reference range, < 4 ng/mL).

Case 2
A 71-year-old male with a history of alcohol use disorder and a 30-year smoking history presented to the ED with worsening dyspnea on exertion. The patient’s baseline exercise tolerance decreased to walking for only 1 block. He reported unintentional weight loss of about 30 pounds over the prior year, no recent respiratory infections, no prior breathing problems, and no personal or family history of cancer. Chest CT revealed findings of bilateral peribronchial opacities as well as mediastinal and hilar lymphadenopathy (Figure 3). The patient developed hypoxic respiratory failure necessitating intubation, mechanical ventilation, and management in the medical intensive care unit, where he was treated for postobstructive pneumonia. Fiberoptic bronchoscopy revealed endobronchial lesions in the right and left upper lobe that were partially obstructing the airway (Figure 4).


The endobronchial masses were debulked using forceps, and samples were sent for surgical pathology evaluation. Staging was completed using linear endobronchial ultrasound, which revealed an enlarged subcarinal lymph node (S7). The surgical pathology of the endobronchial mass and the subcarinal lymph node cytology were consistent with metastatic adenocarcinoma of the prostate. The tumor cells were positive for AE1/AE3, PSA, and NKX3.1, but were negative for CK7 and TTF-1 (Figure 5). Further imaging revealed an enlarged heterogeneous prostate gland, prominent pelvic nodes, and left retroperitoneal lymphadenopathy, as well as sclerotic foci within the T10 vertebral body and right inferior pubic ramus. PSA was also found to be significantly elevated at 700 ng/mL.

Case 3
An 80-year-old male veteran with a history of prostate cancer and recently diagnosed T2N1M0 head and neck squamous cell carcinoma was referred to the Pulmonary service for evaluation of a pulmonary nodule. His medical history was notable for prostate cancer diagnosed 12 years earlier, with an unknown Gleason score. Initial treatment included prostatectomy followed by whole pelvic radiation therapy a year after, due to elevated PSA in surveillance monitoring. This treatment led to remission. After establishing remission for > 10 years, the patient was started on low-dose testosterone replacement therapy to address complications of radiation therapy, namely hypogonadism.
On evaluation, a chest CT was significant for a large 2-cm right middle lobe nodule (Figure 6). At that time, PSA was noted to be borderline elevated at 4.2 ng/mL, and whole-body imaging did not reveal any lesions elsewhere, specifically no bone metastasis. Biopsies of the right middle lobe lung nodule revealed adenocarcinoma consistent with metastatic prostate cancer. Testosterone therapy was promptly discontinued.

The patient initially refused androgen deprivation therapy owing to the antiandrogenic adverse effects. However, subsequent chest CTs revealed growing lung nodules, which convinced him to proceed with androgen deprivation therapy followed by palliative radiation, and chemotherapy and management of malignant pleural effusion with indwelling small bore pleural catheter for about 10 years. He died from COVID-19 during the pandemic.
DISCUSSION
These cases highlight the importance of including prostate cancer in the differential diagnoses of male patients with intrathoracic abnormalities, even in the absence of metastasis to the more common sites. In a large cohort study of 74,826 patients with metastatic prostate cancer, Gandaglia et al found that the most frequent sites of metastasis were bone (84.0%) and distant lymph nodes (10.6%).2 However, thoracic involvement was observed in 9.1% of cases, with isolated thoracic metastasis being rare. The cases described in this report exemplify exceptionally uncommon occurrences within that 9.1%.
Pleural metastases, as observed in Case 1, are a particularly rare manifestation. In a 10-year retrospective assessment, Vinjamoori et al discovered pleural nodules or masses in only 6 of 82 patients (7.3%) with atypical metastases.3 Adrenal and liver metastases accounted for 15% and 37% of cases with atypical distribution. As such, isolated pleural disease is rare even in atypical presentations.3
As seen in Case 2, endobronchial metastases producing airway obstruction are also rare, with the most common primary cancers associated with endobronchial metastasis being breast, colon, and renal cancer.4 The available literature on this presentation is confined to case reports. Hameed et al reported a case of synchronous biopsy-proven endobronchial metastasis from prostate cancer.5 These cases highlight the importance of maintaining a high level of clinical awareness when encountering endobronchial lesions in patients with prostate cancer.
Case 3 presents a unique situation of lung metastases without any involvement of the bones. It is well known—and was confirmed by Heidenreich et al—that lung metastases in prostate adenocarcinoma usually coincide with extensive osseous disease.6 This instance highlights the importance of watchful monitoring for unusual patterns of cancer recurrence.
Immunohistochemistry stains that are specific to prostate cancer include antibodies against PSA. Prostate-specific membrane antigen is another marker that is far more present in malignant than in benign prostate tissue.
The NKX3.1 gene encodes a homeobox protein, which is a transcription factor and tumor suppressor. In prostate cancer, there is loss of heterozygosity of the gene and stains for the IHC antibody to NKX3.1.7
On the other hand, lung cells stain positive for TTF-1, which is produced by surfactant-producing type 2 pneumocytes and club cells in the lung. Antibodies to TTF-1, a common IHC stain, are used to identify adenocarcinoma of lung origin and may carry a prognostic value.7
The immunohistochemistry profiles, specifically the presence of prostate-specific markers such as PSA and NKX3.1, played a vital role in making the diagnosis.
In Case 1, weak TTF-1 positivity was noted, an unusual finding in metastatic prostate adenocarcinoma. Marak et al documented a rare case of TTF-1–positive metastatic prostate cancer, illustrating the potential for diagnostic confusion with primary lung malignancies.8
The 3 cases described in this report demonstrate the importance of clinical consideration, serial follow-up of PSA levels, using more prostate-specific positron emission tomography tracers (eg, Pylarify) alongside traditional imaging, and tissue biopsy to detect unusual metastases.
CONCLUSIONS
Although thoracic metastases from prostate cancer are rare, these presentations highlight the importance of clinical awareness regarding atypical cases. Pleural disease, endobronchial lesions, and isolated pulmonary nodules might be the first clinical manifestation of metastatic prostate cancer. A high index of suspicion, appropriate imaging, and judicious use of immunohistochemistry are important to ensure accurate diagnosis and optimal patient management.
Prostate cancer is the most common noncutaneous cancer in men, accounting for 29% of all incident cancer cases.1 Typically, prostate cancer metastasizes to bone and regional lymph nodes.2 However, intrathoracic manifestation may occur. This report presents 3 cases of rare intrathoracic manifestations of metastatic prostate cancer with a review of the current literature.
CASE PRESENTATIONS
Case 1
A 71-year-old male who was an active smoker and a long-standing employment as a plumber was diagnosed with rectal cancer in 2022. He completed neoadjuvant capecitabine and radiation therapy followed by a rectosigmoidectomy. Several weeks after surgery, the patient presented to the emergency department (ED) with a dry cough and worsening shortness of breath. Point-of-care ultrasound of the lungs revealed a moderate right pleural effusion with several nodular pleural masses. A chest computed tomography (CT) confirmed these findings (Figure 1). A CT of the abdomen and pelvis revealed prostatomegaly with the medial lobe of the prostate protruding into the bladder; however, no enlarged retroperitoneal, mesenteric or pelvic lymph nodes were noted. The patient underwent a right pleural fluid drainage and pleural mass biopsy. Pleural mass histomorphology as well as immunohistochemical (IHC) stains were consistent with metastatic prostate adenocarcinoma. The pleural fluid cytology also was consistent with metastatic prostate adenocarcinoma.

Immunohistochemistry showed weak positive staining for prostate-specific NK3 homeobox 1 gene (NKX3.1), alpha-methylacyl-CoA racemase gene (AMACR), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), and prosaposin, and negative transcription termination factor (TTF-1), keratin-7 (CK7), keratin-20, and caudal type homeobox 2 gene (CDX2) (Figure 2) 2). The patient's prostate-specific antigen (PSA) was found to be elevated at 33.9 ng/mL (reference range, < 4 ng/mL).

Case 2
A 71-year-old male with a history of alcohol use disorder and a 30-year smoking history presented to the ED with worsening dyspnea on exertion. The patient’s baseline exercise tolerance decreased to walking for only 1 block. He reported unintentional weight loss of about 30 pounds over the prior year, no recent respiratory infections, no prior breathing problems, and no personal or family history of cancer. Chest CT revealed findings of bilateral peribronchial opacities as well as mediastinal and hilar lymphadenopathy (Figure 3). The patient developed hypoxic respiratory failure necessitating intubation, mechanical ventilation, and management in the medical intensive care unit, where he was treated for postobstructive pneumonia. Fiberoptic bronchoscopy revealed endobronchial lesions in the right and left upper lobe that were partially obstructing the airway (Figure 4).


The endobronchial masses were debulked using forceps, and samples were sent for surgical pathology evaluation. Staging was completed using linear endobronchial ultrasound, which revealed an enlarged subcarinal lymph node (S7). The surgical pathology of the endobronchial mass and the subcarinal lymph node cytology were consistent with metastatic adenocarcinoma of the prostate. The tumor cells were positive for AE1/AE3, PSA, and NKX3.1, but were negative for CK7 and TTF-1 (Figure 5). Further imaging revealed an enlarged heterogeneous prostate gland, prominent pelvic nodes, and left retroperitoneal lymphadenopathy, as well as sclerotic foci within the T10 vertebral body and right inferior pubic ramus. PSA was also found to be significantly elevated at 700 ng/mL.

Case 3
An 80-year-old male veteran with a history of prostate cancer and recently diagnosed T2N1M0 head and neck squamous cell carcinoma was referred to the Pulmonary service for evaluation of a pulmonary nodule. His medical history was notable for prostate cancer diagnosed 12 years earlier, with an unknown Gleason score. Initial treatment included prostatectomy followed by whole pelvic radiation therapy a year after, due to elevated PSA in surveillance monitoring. This treatment led to remission. After establishing remission for > 10 years, the patient was started on low-dose testosterone replacement therapy to address complications of radiation therapy, namely hypogonadism.
On evaluation, a chest CT was significant for a large 2-cm right middle lobe nodule (Figure 6). At that time, PSA was noted to be borderline elevated at 4.2 ng/mL, and whole-body imaging did not reveal any lesions elsewhere, specifically no bone metastasis. Biopsies of the right middle lobe lung nodule revealed adenocarcinoma consistent with metastatic prostate cancer. Testosterone therapy was promptly discontinued.

The patient initially refused androgen deprivation therapy owing to the antiandrogenic adverse effects. However, subsequent chest CTs revealed growing lung nodules, which convinced him to proceed with androgen deprivation therapy followed by palliative radiation, and chemotherapy and management of malignant pleural effusion with indwelling small bore pleural catheter for about 10 years. He died from COVID-19 during the pandemic.
DISCUSSION
These cases highlight the importance of including prostate cancer in the differential diagnoses of male patients with intrathoracic abnormalities, even in the absence of metastasis to the more common sites. In a large cohort study of 74,826 patients with metastatic prostate cancer, Gandaglia et al found that the most frequent sites of metastasis were bone (84.0%) and distant lymph nodes (10.6%).2 However, thoracic involvement was observed in 9.1% of cases, with isolated thoracic metastasis being rare. The cases described in this report exemplify exceptionally uncommon occurrences within that 9.1%.
Pleural metastases, as observed in Case 1, are a particularly rare manifestation. In a 10-year retrospective assessment, Vinjamoori et al discovered pleural nodules or masses in only 6 of 82 patients (7.3%) with atypical metastases.3 Adrenal and liver metastases accounted for 15% and 37% of cases with atypical distribution. As such, isolated pleural disease is rare even in atypical presentations.3
As seen in Case 2, endobronchial metastases producing airway obstruction are also rare, with the most common primary cancers associated with endobronchial metastasis being breast, colon, and renal cancer.4 The available literature on this presentation is confined to case reports. Hameed et al reported a case of synchronous biopsy-proven endobronchial metastasis from prostate cancer.5 These cases highlight the importance of maintaining a high level of clinical awareness when encountering endobronchial lesions in patients with prostate cancer.
Case 3 presents a unique situation of lung metastases without any involvement of the bones. It is well known—and was confirmed by Heidenreich et al—that lung metastases in prostate adenocarcinoma usually coincide with extensive osseous disease.6 This instance highlights the importance of watchful monitoring for unusual patterns of cancer recurrence.
Immunohistochemistry stains that are specific to prostate cancer include antibodies against PSA. Prostate-specific membrane antigen is another marker that is far more present in malignant than in benign prostate tissue.
The NKX3.1 gene encodes a homeobox protein, which is a transcription factor and tumor suppressor. In prostate cancer, there is loss of heterozygosity of the gene and stains for the IHC antibody to NKX3.1.7
On the other hand, lung cells stain positive for TTF-1, which is produced by surfactant-producing type 2 pneumocytes and club cells in the lung. Antibodies to TTF-1, a common IHC stain, are used to identify adenocarcinoma of lung origin and may carry a prognostic value.7
The immunohistochemistry profiles, specifically the presence of prostate-specific markers such as PSA and NKX3.1, played a vital role in making the diagnosis.
In Case 1, weak TTF-1 positivity was noted, an unusual finding in metastatic prostate adenocarcinoma. Marak et al documented a rare case of TTF-1–positive metastatic prostate cancer, illustrating the potential for diagnostic confusion with primary lung malignancies.8
The 3 cases described in this report demonstrate the importance of clinical consideration, serial follow-up of PSA levels, using more prostate-specific positron emission tomography tracers (eg, Pylarify) alongside traditional imaging, and tissue biopsy to detect unusual metastases.
CONCLUSIONS
Although thoracic metastases from prostate cancer are rare, these presentations highlight the importance of clinical awareness regarding atypical cases. Pleural disease, endobronchial lesions, and isolated pulmonary nodules might be the first clinical manifestation of metastatic prostate cancer. A high index of suspicion, appropriate imaging, and judicious use of immunohistochemistry are important to ensure accurate diagnosis and optimal patient management.
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49. doi:10.3322/caac.21820
- Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
- Vinjamoori AH, Jagannathan JP, Shinagare AB, et al. Atypical metastases from prostate cancer: 10-year experience at a single institution. AJR Am J Roentgenol. 2012;199(2):367-372. doi:10.2214/AJR.11.7533
- Salud A, Porcel JM, Rovirosa A, Bellmunt J. Endobronchial metastatic disease: analysis of 32 cases. J Surg Oncol. 1996;62(4):249-252. doi:10.1002/(SICI)1096- 9098(199608)62:4<249::AID-JSO4>3.0.CO;2-6
- Hameed M, Haq IU, Yousaf M, Hussein M, Rashid U, Al-Bozom I. Endobronchial metastases secondary to prostate cancer: a case report and literature review. Respir Med Case Rep. 2020;32:101326. doi:10.1016/j.rmcr.2020.101326
- Heidenreich A, Bastian PJ, Bellmunt J, et al; for the European Association of Urology. EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration- resistant prostate cancer. Eur Urol. 2014;65(2):467- 479. doi:10.1016/j.eururo.2013.11.002
- Schallenberg S, Dernbach G, Dragomir MP, et al. TTF-1 status in early-stage lung adenocarcinoma is an independent predictor of relapse and survival superior to tumor grading. Eur J Cancer. 2024;197:113474. doi:10.1016/j.ejca.2023.113474
- Marak C, Guddati AK, Ashraf A, Smith J, Kaushik P. Prostate adenocarcinoma with atypical immunohistochemistry presenting with a Cheerio sign. AIM Clinical Cases. 2023;1:e220508. doi:10.7326/aimcc.2022.0508
- Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49. doi:10.3322/caac.21820
- Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
- Vinjamoori AH, Jagannathan JP, Shinagare AB, et al. Atypical metastases from prostate cancer: 10-year experience at a single institution. AJR Am J Roentgenol. 2012;199(2):367-372. doi:10.2214/AJR.11.7533
- Salud A, Porcel JM, Rovirosa A, Bellmunt J. Endobronchial metastatic disease: analysis of 32 cases. J Surg Oncol. 1996;62(4):249-252. doi:10.1002/(SICI)1096- 9098(199608)62:4<249::AID-JSO4>3.0.CO;2-6
- Hameed M, Haq IU, Yousaf M, Hussein M, Rashid U, Al-Bozom I. Endobronchial metastases secondary to prostate cancer: a case report and literature review. Respir Med Case Rep. 2020;32:101326. doi:10.1016/j.rmcr.2020.101326
- Heidenreich A, Bastian PJ, Bellmunt J, et al; for the European Association of Urology. EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration- resistant prostate cancer. Eur Urol. 2014;65(2):467- 479. doi:10.1016/j.eururo.2013.11.002
- Schallenberg S, Dernbach G, Dragomir MP, et al. TTF-1 status in early-stage lung adenocarcinoma is an independent predictor of relapse and survival superior to tumor grading. Eur J Cancer. 2024;197:113474. doi:10.1016/j.ejca.2023.113474
- Marak C, Guddati AK, Ashraf A, Smith J, Kaushik P. Prostate adenocarcinoma with atypical immunohistochemistry presenting with a Cheerio sign. AIM Clinical Cases. 2023;1:e220508. doi:10.7326/aimcc.2022.0508
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Atypical Intrathoracic Manifestations of Metastatic Prostate Cancer: A Case Series
Cancer Data Trends 2025
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Access, Race, and "Colon Age": Improving CRC Screening
- Lung Cancer: Mortality Trends in Veterans and New Treatments
- Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
- Breast and Uterine Cancer: Screening Guidelines, Genetic Testing, and Mortality Trends
- HCC Updates: Quality Care Framework and Risk Stratification Data
- Rising Kidney Cancer Cases and Emerging Treatments for Veterans
- Advances in Blood Cancer Care for Veterans
- AI-Based Risk Stratification for Oropharyngeal Carcinomas: AIROC
- Brain Cancer: Epidemiology, TBI, and New Treatments
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Access, Race, and "Colon Age": Improving CRC Screening
- Lung Cancer: Mortality Trends in Veterans and New Treatments
- Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
- Breast and Uterine Cancer: Screening Guidelines, Genetic Testing, and Mortality Trends
- HCC Updates: Quality Care Framework and Risk Stratification Data
- Rising Kidney Cancer Cases and Emerging Treatments for Veterans
- Advances in Blood Cancer Care for Veterans
- AI-Based Risk Stratification for Oropharyngeal Carcinomas: AIROC
- Brain Cancer: Epidemiology, TBI, and New Treatments
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Access, Race, and "Colon Age": Improving CRC Screening
- Lung Cancer: Mortality Trends in Veterans and New Treatments
- Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
- Breast and Uterine Cancer: Screening Guidelines, Genetic Testing, and Mortality Trends
- HCC Updates: Quality Care Framework and Risk Stratification Data
- Rising Kidney Cancer Cases and Emerging Treatments for Veterans
- Advances in Blood Cancer Care for Veterans
- AI-Based Risk Stratification for Oropharyngeal Carcinomas: AIROC
- Brain Cancer: Epidemiology, TBI, and New Treatments
Million Veteran Program Drives Prostate Cancer Research
About 15,000 veterans are annually diagnosed with prostate cancer. Fortunately, those veterans enrolled in the US Department of Veterans Affairs (VA) Million Veteran Program (MVP) provide researchers with a deep pool of genetic data that can help identify causes, aid diagnosis, and guide targeted treatments.
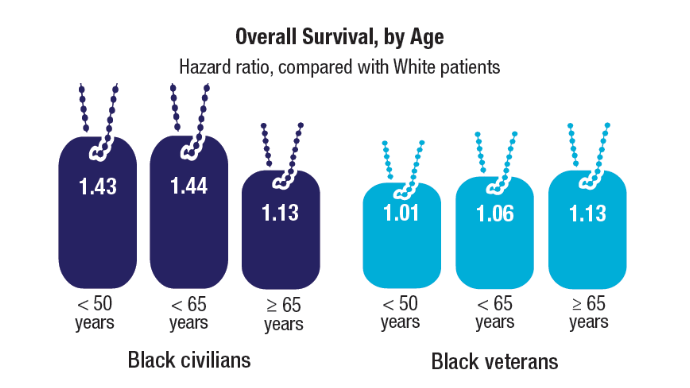
More than 1,000,000 veterans have enrolled in MVP and donated their anonymized DNA to foster research. It is also one of the most genetically diverse health-related databases: 20% of participants identify as Black, 8% as Hispanic, 2% as Asian American, and 1% as Native American.
Ethnically and racially diverse data are particularly important for advancing the treatment of underserved groups. In a 2020 review, researchers found a number of areas where Black veterans differed from White veterans, including prostate-specific antigen (PSA) levels, incidence (almost 60% higher), clinical course, and mortality rate (2 to 3 times greater). To facilitate research, the MVP developed the “DNA chip,” a custom-designed tool that tests for > 750,000 genetic variants, including > 300,000 that are more common in minority populations.
“The whole thing about understanding genetics and diversity is like a circular feedback loop,” Director of MVP Dr. Sumitra Muralidhar said in a VA news article. “The more people you have represented from different racial and ethnic backgrounds, the more we’ll be able to discover genetic variants that contribute to their health. The more we discover, the more we can help that group. It’s a complete circular feedback loop.”
In addition to veterans’ blood samples and 600,000-plus baseline surveys on lifestyle, military service, and health, the MVP has collected upwards of 825,000 germline DNA samples, which have helped inform research into prostate cancer, the most commonly diagnosed solid tumor among veterans. By mining these data, researchers have built more evidence of how genes add to risk and disease progression.
In one study preprint that has not been peer reviewed, VA researchers investigated the significance of high polygenic hazard scores. The scores are strongly associated with age at diagnosis of any prostate cancer, as well as lifetime risk of metastatic and fatal prostate cancer. However, because they’re associated with any prostate cancer, the researchers say, there is concern that screening men with high polygenic risk could increase overdiagnosis of indolent cancers.
The researchers analyzed genetic and phenotypic data from 69,901 men in the MVP who have been diagnosed with prostate cancer (6413 metastatic). They found their hypothesis to be correct: Among men eventually diagnosed with prostate cancer, those with higher polygenic risk were more likely to develop metastatic disease.
Genetic risk scores like PHS601, a 601-variant polygenic score, can be performed on a saliva sample at any time during a person’s life, the researchers note. Thus, the scores provide the earliest information about age-specific risk of developing aggressive prostate cancer. These scores might be useful, they suggest, to support clinical decisions not only about whom to screen but also at what age.
Another study led by Stanford University researchers and published in Nature Genetics aimed to make screening more targeted, in this case prostate specific antigen screening. Estimates about PSA heritability vary from 40% to 45%, with genome-wide evaluations putting it at 25% to 30%, suggesting that incorporating genetic factors could improve screening.
This study involved 296,754 men (211,342 with European ancestry, 58,236 with African ancestry, 23,546 with Hispanic/Latino ancestry, and 3630 with Asian ancestry; 96.5% of participants were from MVP)—a sample size more than triple that in previous work.
The researchers detected 448 genome-wide significant variants, including 295 that were novel (to the best of their knowledge). The variance explained by genome-wide polygenic risk scores ranged from 11.6% to 16.6% for European ancestry, 5.5% to 9.5% for African ancestry, 13.5% to 18.2% for Hispanic/Latino ancestry, and 8.6% to 15.3% for Asian ancestry, and decreased with increasing age. Midlife genetically adjusted PSA levels were more strongly associated with overall and aggressive prostate cancer than unadjusted PSA levels.
The researchers say their study highlights how including higher proportions of participants from underrepresented populations can improve genetic prediction of PSA levels, offering the potential to personalize prostate cancer screening. Adjusting PSA for individuals’ predispositions in the absence of prostate cancer could improve the specificity (to reduce overdiagnosis) and sensitivity (to prevent more deaths) of screening.
Their findings, the researchers suggest, also explain additional variation in PSA, especially among men of African heritage, who experience the highest prostate cancer morbidity and mortality. They note that this work “moved us closer to leveraging genetic information to personalize PSA and substantially improved our understanding of PSA across diverse ancestries.”
A third study from a team at the VA Tennessee Valley Healthcare System also investigated the risk of inheriting a predisposition to prostate cancer. These researchers explored pathogenic variants using both genome-wide single-allele and identity-by-descent analytic approaches. They then tested their candidate variants for replication across independent biobanks, including MVP.
The researchers discovered the gene WNT9B E152K more than doubled the risk of familial prostate cancer. Meta-analysis, collectively encompassing 500,000 patients, confirmed the genome-wide significance. The researchers say WNT9B shares an “unexpected commonality” with the previously established prostate cancer risk genes HOXB13 and HNF1B: Each are required for embryonic prostate development. Based on that finding, the researchers also evaluated 2 additional genes, KMT2D and DHCR7, which are known to cause Mendelian genitourinary developmental defects. They, too, were nominally associated with prostate cancer under meta-analyses.
Tens of thousands of participants in MVP have had prostate cancer. The genetic research they participate in advances detection, prediction, and treatment for themselves and others, and science in general. The research is not only about finding causes, but what to do if the cancer develops. An “acting on MVP prostate cancer findings” study at VA Puget Sound Health Care System is testing how communicating with veterans about MVP prostate cancer results will affect their care. Those with prostate cancer will be screened to determine genetic contributions to their cancers. Those found to have a gene-based cancer diagnosis will be offered genetic counseling. Their immediate family will also be offered screening to test for inherited prostate cancer risk.
In 2016, the VA partnered with the Prostate Cancer Foundation to establish the Precision Oncology Program for Cancer of the Prostate (POPCaP). In collaboration with MVP and the Genomic Medicine Service, the program uses genetic information to individualize treatments for veterans with advanced prostate cancer.
US Army Veteran James Perry is one of the beneficiaries of the program. First diagnosed with prostate cancer in 2001, he was initially treated with radiation therapy, but the cancer recurred and spread to his lung. The John J. Cochran Veterans Hospital in St. Louis sent a sample of Perry's lung tumor to the laboratory for genetic testing, where they discovered he had a BRCA1 gene mutation.
His oncologist, Dr. Martin Schoen, recommended Perry enroll in AMPLITUDE, a clinical trial testing the effectiveness of poly-ADP ribose polymerase inhibitors, a new class of drugs to treat hormone-sensitive prostate cancer. One year later, Perry’s lung tumor could barely be seen on computed tomography, and his PSA levels were undetectable.
"I would highly recommend enrolling in a trial," Perry told VA Research Currents. “If a veteran has that opportunity, I would encourage it—anything that is going to give you a few more days is worth it.” In the interview, Perry said he enjoyed being part of the trial because he knows he is getting the most advanced care possible and is proud to help others like himself.
"We are honored to support VA's work to improve the lives of veterans who are living with advanced prostate cancer," Vice President and National Director of the PCF Veterans Health Initiative Rebecca Levine said. "Clinical trials play a vital role in bringing new treatments to patients who need them most. Mr. Perry's experience illustrates VA's commitment to provide state-of-the-art cancer care to all veterans who need it."
About 15,000 veterans are annually diagnosed with prostate cancer. Fortunately, those veterans enrolled in the US Department of Veterans Affairs (VA) Million Veteran Program (MVP) provide researchers with a deep pool of genetic data that can help identify causes, aid diagnosis, and guide targeted treatments.
More than 1,000,000 veterans have enrolled in MVP and donated their anonymized DNA to foster research. It is also one of the most genetically diverse health-related databases: 20% of participants identify as Black, 8% as Hispanic, 2% as Asian American, and 1% as Native American.
Ethnically and racially diverse data are particularly important for advancing the treatment of underserved groups. In a 2020 review, researchers found a number of areas where Black veterans differed from White veterans, including prostate-specific antigen (PSA) levels, incidence (almost 60% higher), clinical course, and mortality rate (2 to 3 times greater). To facilitate research, the MVP developed the “DNA chip,” a custom-designed tool that tests for > 750,000 genetic variants, including > 300,000 that are more common in minority populations.
“The whole thing about understanding genetics and diversity is like a circular feedback loop,” Director of MVP Dr. Sumitra Muralidhar said in a VA news article. “The more people you have represented from different racial and ethnic backgrounds, the more we’ll be able to discover genetic variants that contribute to their health. The more we discover, the more we can help that group. It’s a complete circular feedback loop.”
In addition to veterans’ blood samples and 600,000-plus baseline surveys on lifestyle, military service, and health, the MVP has collected upwards of 825,000 germline DNA samples, which have helped inform research into prostate cancer, the most commonly diagnosed solid tumor among veterans. By mining these data, researchers have built more evidence of how genes add to risk and disease progression.
In one study preprint that has not been peer reviewed, VA researchers investigated the significance of high polygenic hazard scores. The scores are strongly associated with age at diagnosis of any prostate cancer, as well as lifetime risk of metastatic and fatal prostate cancer. However, because they’re associated with any prostate cancer, the researchers say, there is concern that screening men with high polygenic risk could increase overdiagnosis of indolent cancers.
The researchers analyzed genetic and phenotypic data from 69,901 men in the MVP who have been diagnosed with prostate cancer (6413 metastatic). They found their hypothesis to be correct: Among men eventually diagnosed with prostate cancer, those with higher polygenic risk were more likely to develop metastatic disease.
Genetic risk scores like PHS601, a 601-variant polygenic score, can be performed on a saliva sample at any time during a person’s life, the researchers note. Thus, the scores provide the earliest information about age-specific risk of developing aggressive prostate cancer. These scores might be useful, they suggest, to support clinical decisions not only about whom to screen but also at what age.
Another study led by Stanford University researchers and published in Nature Genetics aimed to make screening more targeted, in this case prostate specific antigen screening. Estimates about PSA heritability vary from 40% to 45%, with genome-wide evaluations putting it at 25% to 30%, suggesting that incorporating genetic factors could improve screening.
This study involved 296,754 men (211,342 with European ancestry, 58,236 with African ancestry, 23,546 with Hispanic/Latino ancestry, and 3630 with Asian ancestry; 96.5% of participants were from MVP)—a sample size more than triple that in previous work.
The researchers detected 448 genome-wide significant variants, including 295 that were novel (to the best of their knowledge). The variance explained by genome-wide polygenic risk scores ranged from 11.6% to 16.6% for European ancestry, 5.5% to 9.5% for African ancestry, 13.5% to 18.2% for Hispanic/Latino ancestry, and 8.6% to 15.3% for Asian ancestry, and decreased with increasing age. Midlife genetically adjusted PSA levels were more strongly associated with overall and aggressive prostate cancer than unadjusted PSA levels.
The researchers say their study highlights how including higher proportions of participants from underrepresented populations can improve genetic prediction of PSA levels, offering the potential to personalize prostate cancer screening. Adjusting PSA for individuals’ predispositions in the absence of prostate cancer could improve the specificity (to reduce overdiagnosis) and sensitivity (to prevent more deaths) of screening.
Their findings, the researchers suggest, also explain additional variation in PSA, especially among men of African heritage, who experience the highest prostate cancer morbidity and mortality. They note that this work “moved us closer to leveraging genetic information to personalize PSA and substantially improved our understanding of PSA across diverse ancestries.”
A third study from a team at the VA Tennessee Valley Healthcare System also investigated the risk of inheriting a predisposition to prostate cancer. These researchers explored pathogenic variants using both genome-wide single-allele and identity-by-descent analytic approaches. They then tested their candidate variants for replication across independent biobanks, including MVP.
The researchers discovered the gene WNT9B E152K more than doubled the risk of familial prostate cancer. Meta-analysis, collectively encompassing 500,000 patients, confirmed the genome-wide significance. The researchers say WNT9B shares an “unexpected commonality” with the previously established prostate cancer risk genes HOXB13 and HNF1B: Each are required for embryonic prostate development. Based on that finding, the researchers also evaluated 2 additional genes, KMT2D and DHCR7, which are known to cause Mendelian genitourinary developmental defects. They, too, were nominally associated with prostate cancer under meta-analyses.
Tens of thousands of participants in MVP have had prostate cancer. The genetic research they participate in advances detection, prediction, and treatment for themselves and others, and science in general. The research is not only about finding causes, but what to do if the cancer develops. An “acting on MVP prostate cancer findings” study at VA Puget Sound Health Care System is testing how communicating with veterans about MVP prostate cancer results will affect their care. Those with prostate cancer will be screened to determine genetic contributions to their cancers. Those found to have a gene-based cancer diagnosis will be offered genetic counseling. Their immediate family will also be offered screening to test for inherited prostate cancer risk.
In 2016, the VA partnered with the Prostate Cancer Foundation to establish the Precision Oncology Program for Cancer of the Prostate (POPCaP). In collaboration with MVP and the Genomic Medicine Service, the program uses genetic information to individualize treatments for veterans with advanced prostate cancer.
US Army Veteran James Perry is one of the beneficiaries of the program. First diagnosed with prostate cancer in 2001, he was initially treated with radiation therapy, but the cancer recurred and spread to his lung. The John J. Cochran Veterans Hospital in St. Louis sent a sample of Perry's lung tumor to the laboratory for genetic testing, where they discovered he had a BRCA1 gene mutation.
His oncologist, Dr. Martin Schoen, recommended Perry enroll in AMPLITUDE, a clinical trial testing the effectiveness of poly-ADP ribose polymerase inhibitors, a new class of drugs to treat hormone-sensitive prostate cancer. One year later, Perry’s lung tumor could barely be seen on computed tomography, and his PSA levels were undetectable.
"I would highly recommend enrolling in a trial," Perry told VA Research Currents. “If a veteran has that opportunity, I would encourage it—anything that is going to give you a few more days is worth it.” In the interview, Perry said he enjoyed being part of the trial because he knows he is getting the most advanced care possible and is proud to help others like himself.
"We are honored to support VA's work to improve the lives of veterans who are living with advanced prostate cancer," Vice President and National Director of the PCF Veterans Health Initiative Rebecca Levine said. "Clinical trials play a vital role in bringing new treatments to patients who need them most. Mr. Perry's experience illustrates VA's commitment to provide state-of-the-art cancer care to all veterans who need it."
About 15,000 veterans are annually diagnosed with prostate cancer. Fortunately, those veterans enrolled in the US Department of Veterans Affairs (VA) Million Veteran Program (MVP) provide researchers with a deep pool of genetic data that can help identify causes, aid diagnosis, and guide targeted treatments.
More than 1,000,000 veterans have enrolled in MVP and donated their anonymized DNA to foster research. It is also one of the most genetically diverse health-related databases: 20% of participants identify as Black, 8% as Hispanic, 2% as Asian American, and 1% as Native American.
Ethnically and racially diverse data are particularly important for advancing the treatment of underserved groups. In a 2020 review, researchers found a number of areas where Black veterans differed from White veterans, including prostate-specific antigen (PSA) levels, incidence (almost 60% higher), clinical course, and mortality rate (2 to 3 times greater). To facilitate research, the MVP developed the “DNA chip,” a custom-designed tool that tests for > 750,000 genetic variants, including > 300,000 that are more common in minority populations.
“The whole thing about understanding genetics and diversity is like a circular feedback loop,” Director of MVP Dr. Sumitra Muralidhar said in a VA news article. “The more people you have represented from different racial and ethnic backgrounds, the more we’ll be able to discover genetic variants that contribute to their health. The more we discover, the more we can help that group. It’s a complete circular feedback loop.”
In addition to veterans’ blood samples and 600,000-plus baseline surveys on lifestyle, military service, and health, the MVP has collected upwards of 825,000 germline DNA samples, which have helped inform research into prostate cancer, the most commonly diagnosed solid tumor among veterans. By mining these data, researchers have built more evidence of how genes add to risk and disease progression.
In one study preprint that has not been peer reviewed, VA researchers investigated the significance of high polygenic hazard scores. The scores are strongly associated with age at diagnosis of any prostate cancer, as well as lifetime risk of metastatic and fatal prostate cancer. However, because they’re associated with any prostate cancer, the researchers say, there is concern that screening men with high polygenic risk could increase overdiagnosis of indolent cancers.
The researchers analyzed genetic and phenotypic data from 69,901 men in the MVP who have been diagnosed with prostate cancer (6413 metastatic). They found their hypothesis to be correct: Among men eventually diagnosed with prostate cancer, those with higher polygenic risk were more likely to develop metastatic disease.
Genetic risk scores like PHS601, a 601-variant polygenic score, can be performed on a saliva sample at any time during a person’s life, the researchers note. Thus, the scores provide the earliest information about age-specific risk of developing aggressive prostate cancer. These scores might be useful, they suggest, to support clinical decisions not only about whom to screen but also at what age.
Another study led by Stanford University researchers and published in Nature Genetics aimed to make screening more targeted, in this case prostate specific antigen screening. Estimates about PSA heritability vary from 40% to 45%, with genome-wide evaluations putting it at 25% to 30%, suggesting that incorporating genetic factors could improve screening.
This study involved 296,754 men (211,342 with European ancestry, 58,236 with African ancestry, 23,546 with Hispanic/Latino ancestry, and 3630 with Asian ancestry; 96.5% of participants were from MVP)—a sample size more than triple that in previous work.
The researchers detected 448 genome-wide significant variants, including 295 that were novel (to the best of their knowledge). The variance explained by genome-wide polygenic risk scores ranged from 11.6% to 16.6% for European ancestry, 5.5% to 9.5% for African ancestry, 13.5% to 18.2% for Hispanic/Latino ancestry, and 8.6% to 15.3% for Asian ancestry, and decreased with increasing age. Midlife genetically adjusted PSA levels were more strongly associated with overall and aggressive prostate cancer than unadjusted PSA levels.
The researchers say their study highlights how including higher proportions of participants from underrepresented populations can improve genetic prediction of PSA levels, offering the potential to personalize prostate cancer screening. Adjusting PSA for individuals’ predispositions in the absence of prostate cancer could improve the specificity (to reduce overdiagnosis) and sensitivity (to prevent more deaths) of screening.
Their findings, the researchers suggest, also explain additional variation in PSA, especially among men of African heritage, who experience the highest prostate cancer morbidity and mortality. They note that this work “moved us closer to leveraging genetic information to personalize PSA and substantially improved our understanding of PSA across diverse ancestries.”
A third study from a team at the VA Tennessee Valley Healthcare System also investigated the risk of inheriting a predisposition to prostate cancer. These researchers explored pathogenic variants using both genome-wide single-allele and identity-by-descent analytic approaches. They then tested their candidate variants for replication across independent biobanks, including MVP.
The researchers discovered the gene WNT9B E152K more than doubled the risk of familial prostate cancer. Meta-analysis, collectively encompassing 500,000 patients, confirmed the genome-wide significance. The researchers say WNT9B shares an “unexpected commonality” with the previously established prostate cancer risk genes HOXB13 and HNF1B: Each are required for embryonic prostate development. Based on that finding, the researchers also evaluated 2 additional genes, KMT2D and DHCR7, which are known to cause Mendelian genitourinary developmental defects. They, too, were nominally associated with prostate cancer under meta-analyses.
Tens of thousands of participants in MVP have had prostate cancer. The genetic research they participate in advances detection, prediction, and treatment for themselves and others, and science in general. The research is not only about finding causes, but what to do if the cancer develops. An “acting on MVP prostate cancer findings” study at VA Puget Sound Health Care System is testing how communicating with veterans about MVP prostate cancer results will affect their care. Those with prostate cancer will be screened to determine genetic contributions to their cancers. Those found to have a gene-based cancer diagnosis will be offered genetic counseling. Their immediate family will also be offered screening to test for inherited prostate cancer risk.
In 2016, the VA partnered with the Prostate Cancer Foundation to establish the Precision Oncology Program for Cancer of the Prostate (POPCaP). In collaboration with MVP and the Genomic Medicine Service, the program uses genetic information to individualize treatments for veterans with advanced prostate cancer.
US Army Veteran James Perry is one of the beneficiaries of the program. First diagnosed with prostate cancer in 2001, he was initially treated with radiation therapy, but the cancer recurred and spread to his lung. The John J. Cochran Veterans Hospital in St. Louis sent a sample of Perry's lung tumor to the laboratory for genetic testing, where they discovered he had a BRCA1 gene mutation.
His oncologist, Dr. Martin Schoen, recommended Perry enroll in AMPLITUDE, a clinical trial testing the effectiveness of poly-ADP ribose polymerase inhibitors, a new class of drugs to treat hormone-sensitive prostate cancer. One year later, Perry’s lung tumor could barely be seen on computed tomography, and his PSA levels were undetectable.
"I would highly recommend enrolling in a trial," Perry told VA Research Currents. “If a veteran has that opportunity, I would encourage it—anything that is going to give you a few more days is worth it.” In the interview, Perry said he enjoyed being part of the trial because he knows he is getting the most advanced care possible and is proud to help others like himself.
"We are honored to support VA's work to improve the lives of veterans who are living with advanced prostate cancer," Vice President and National Director of the PCF Veterans Health Initiative Rebecca Levine said. "Clinical trials play a vital role in bringing new treatments to patients who need them most. Mr. Perry's experience illustrates VA's commitment to provide state-of-the-art cancer care to all veterans who need it."
Service Connection Expanded to Additional Cancers
The US Department of Veterans Affairs (VA) is "lowering the burden of proof" for thousands, making acute and chronic leukemias, multiple myelomas, myelodysplastic syndromes, myelofibrosis, urinary bladder, ureter, and related genitourinary cancers presumptive for service connection.
The Jan. 8 decision included Gulf War veterans, those who served in Somalia or the Southwest Asia theater of operations during the Persian Gulf War on or after Aug. 2, 1990; and post-9/11 veterans, those who served in Afghanistan, Iraq, Djibouti, Egypt, Jordan, Lebanon, Syria, Yemen, or Uzbekistan and the airspace above these locations during the Gulf War on or after Sept. 11, 2001. It also includes veterans who served at the Karshi-Khanabad (K2) base in Uzbekistan after Sept. 11, 2001.
Veterans no longer must prove their service caused their condition to receive benefits. This landmark decision allows them access to free health care for that condition.
According to the VA, these steps are also part of a comprehensive effort to ensure that K2 veterans—and their survivors—receive the care and benefits they deserve. K2 veterans have higher claim and approval rates than any other cohort of veterans: 13,002 are enrolled in VA health care, and the average K2 veteran is service connected for 14.6 conditions.
The 2022 PACT Act was the largest expansion of veteran benefits in generations. The VA then made millions of veterans eligible for health care and benefits years earlier than called for by the law. It also launched the largest outreach campaign in the history of the VA to encourage veterans to apply.
Nearly 890,000 veterans have signed up for VA health care since the bill was signed into law, a nearly 40% increase over the previous equivalent period, and veterans have submitted > 4.8 million applications for VA benefits (a 42% increase over the previous equivalent period and an all-time record). The VA has delivered > $600 billion in earned benefits directly to veterans, their families, and survivors during that time.
The VA encourages all eligible veterans—including those with previously denied claims—to apply for benefits. To apply for benefits, veterans and survivors may visit VA.gov or call 1-800-MYVA411.
The US Department of Veterans Affairs (VA) is "lowering the burden of proof" for thousands, making acute and chronic leukemias, multiple myelomas, myelodysplastic syndromes, myelofibrosis, urinary bladder, ureter, and related genitourinary cancers presumptive for service connection.
The Jan. 8 decision included Gulf War veterans, those who served in Somalia or the Southwest Asia theater of operations during the Persian Gulf War on or after Aug. 2, 1990; and post-9/11 veterans, those who served in Afghanistan, Iraq, Djibouti, Egypt, Jordan, Lebanon, Syria, Yemen, or Uzbekistan and the airspace above these locations during the Gulf War on or after Sept. 11, 2001. It also includes veterans who served at the Karshi-Khanabad (K2) base in Uzbekistan after Sept. 11, 2001.
Veterans no longer must prove their service caused their condition to receive benefits. This landmark decision allows them access to free health care for that condition.
According to the VA, these steps are also part of a comprehensive effort to ensure that K2 veterans—and their survivors—receive the care and benefits they deserve. K2 veterans have higher claim and approval rates than any other cohort of veterans: 13,002 are enrolled in VA health care, and the average K2 veteran is service connected for 14.6 conditions.
The 2022 PACT Act was the largest expansion of veteran benefits in generations. The VA then made millions of veterans eligible for health care and benefits years earlier than called for by the law. It also launched the largest outreach campaign in the history of the VA to encourage veterans to apply.
Nearly 890,000 veterans have signed up for VA health care since the bill was signed into law, a nearly 40% increase over the previous equivalent period, and veterans have submitted > 4.8 million applications for VA benefits (a 42% increase over the previous equivalent period and an all-time record). The VA has delivered > $600 billion in earned benefits directly to veterans, their families, and survivors during that time.
The VA encourages all eligible veterans—including those with previously denied claims—to apply for benefits. To apply for benefits, veterans and survivors may visit VA.gov or call 1-800-MYVA411.
The US Department of Veterans Affairs (VA) is "lowering the burden of proof" for thousands, making acute and chronic leukemias, multiple myelomas, myelodysplastic syndromes, myelofibrosis, urinary bladder, ureter, and related genitourinary cancers presumptive for service connection.
The Jan. 8 decision included Gulf War veterans, those who served in Somalia or the Southwest Asia theater of operations during the Persian Gulf War on or after Aug. 2, 1990; and post-9/11 veterans, those who served in Afghanistan, Iraq, Djibouti, Egypt, Jordan, Lebanon, Syria, Yemen, or Uzbekistan and the airspace above these locations during the Gulf War on or after Sept. 11, 2001. It also includes veterans who served at the Karshi-Khanabad (K2) base in Uzbekistan after Sept. 11, 2001.
Veterans no longer must prove their service caused their condition to receive benefits. This landmark decision allows them access to free health care for that condition.
According to the VA, these steps are also part of a comprehensive effort to ensure that K2 veterans—and their survivors—receive the care and benefits they deserve. K2 veterans have higher claim and approval rates than any other cohort of veterans: 13,002 are enrolled in VA health care, and the average K2 veteran is service connected for 14.6 conditions.
The 2022 PACT Act was the largest expansion of veteran benefits in generations. The VA then made millions of veterans eligible for health care and benefits years earlier than called for by the law. It also launched the largest outreach campaign in the history of the VA to encourage veterans to apply.
Nearly 890,000 veterans have signed up for VA health care since the bill was signed into law, a nearly 40% increase over the previous equivalent period, and veterans have submitted > 4.8 million applications for VA benefits (a 42% increase over the previous equivalent period and an all-time record). The VA has delivered > $600 billion in earned benefits directly to veterans, their families, and survivors during that time.
The VA encourages all eligible veterans—including those with previously denied claims—to apply for benefits. To apply for benefits, veterans and survivors may visit VA.gov or call 1-800-MYVA411.
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Prostate cancer is the most common cancer in US males, with an estimated 313,780 new cases and 35,770 deaths in 2025.1 Several treatment options are available for localized prostate cancer that have similar outcomes, including active surveillance for low-risk cancers, surgery, or radiotherapy.2,3 Conventional fractionation radiotherapy (CFRT) with 40 to 45 fractions over 8 to 9 weeks has been used for decades. Over the past 2 decades, moderate hypofractionation schedules with 2.4 to 3.4 Gy per fraction over 20 to 28 fractions have become standard, as many noninferiority randomized clinical trials (RCTs) such as CHHiP (UK),4 PROFIT (Canada and Europe),5 NRG Oncology RTOG 0415 (US),6 HYPRO (Netherlands),7,8 and HYPO-RT-PC (Sweden and Denmark),9 have shown the noninferiority of moderately hypofractionated radiotherapy compared with CFRT. Notably, most of these noninferiority studies primarily included patients with low- or intermediate-risk prostate cancer, except for the HYPO-RT-PC trial,9 which also included patients with intermediate- and high-risk prostate cancer.
These noninferiority studies, along with technological advances in radiotherapy, such as intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), and image-guided radiotherapy (IGRT), paved the path to ultrahypofractionated stereotactic body radiotherapy (SBRT) that is delivered in 5 fractions of ≥ 6 Gy. This high dose per fraction may have a radiobiologic advantage over conventional fractionation. The relatively low a/ß ratio of prostate cancer, estimated to be between 1 and 2, suggests that tumor cells may be particularly sensitive to the high doses per fraction delivered in SBRT.10-13 Compared with CFRT, SBRT-induced tumor cell death may also be mediated through different pathways; this pathway appears to be generated in a dose-dependent manner, particularly with doses > 8 Gy per fraction.14,15 Additionally, the higher a/ß ratio for the surrounding organs at risk, such as the bladder and rectum, theoretically allows for an improved therapeutic ratio window that maximizes tumor control while minimizing damage to healthy tissues.
A substantial body of evidence from prospective studies and meta-analyses supports the use of SBRT for localized prostate cancer. HYPO-RT-PC, a significant phase 3 noninferiority study, enrolled 1200 patients with intermediate (89%) and high-risk (11%) prostate cancer randomized between 2 arms, including CFRT to 78 Gy in 39 fractions and SBRT to 42.7 Gy in 7 fractions, treated 3 days weekly. After a median follow-up of 60 months, the estimated 5-year biochemical relapse-free survival rate was 84% in both groups.9 This trial was notable because it was the first randomized study to demonstrate that SBRT was noninferior to CFRT in intermediate- and high-risk prostate cancer patients. Another pivotal phase 3 trial, the PACE-B study, enrolled 874 patients to compare SBRT (36.25 Gy to the prostate gland, with a secondary dose of 40 Gy to the gross tumor volume where applicable, in 5 fractions) with CFRT (78 Gy in 39 fractions) and moderately hypofractionated radiotherapy (HFRT) (62 Gy in 20 fractions) in patients with low- or intermediate-risk prostate cancer. With a 74-month median follow-up, the study reported 5-year biochemical free rates of 94.6% for CFRT and 95.8% for SBRT, confirming the noninferiority of SBRT to CFRT.15
SBRT offers short, effective, and convenient treatment to many patients with localized prostate cancer. While previous guidelines were more restrictive, the March 2026 National Comprehensive Cancer Network (NCCN) guidelines now list SBRT as a preferred treatment modality for high-risk prostate cancer.16
Given the growing body of evidence supporting the efficacy and safety of SBRT, we implemented an SBRT program in 2014 at a tertiary care center for veterans. This retrospective study was undertaken to evaluate the early efficacy and toxicity of SBRT in patients with localized prostate cancer treated at our institution, including patients across all risk stratifications.
METHODS
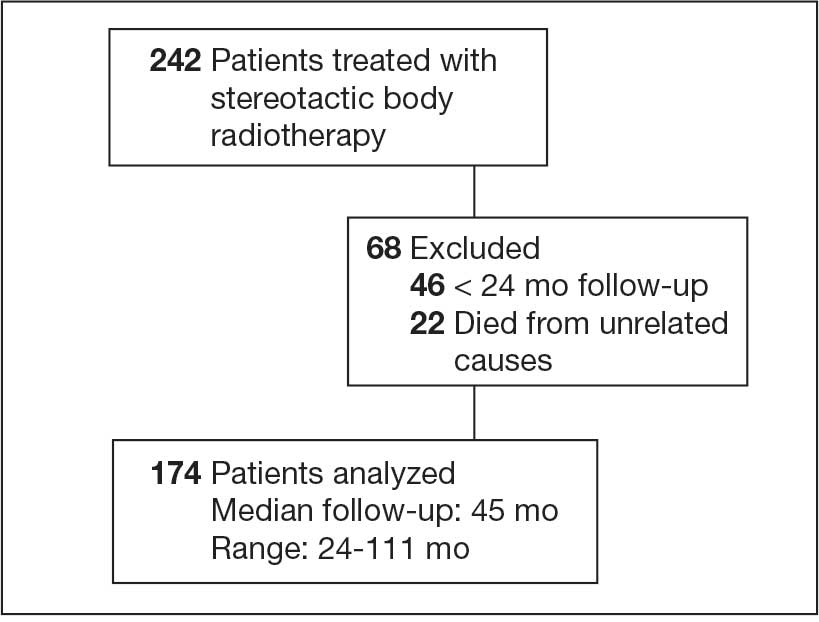
We identified 242 patients diagnosed with prostate cancer who underwent SBRT treatment between November 2014 and October 2024 at Overland Park Veterans Affairs Radiation Oncology Clinic. For the final analysis, 46 patients with < 2 years of follow-up and 22 patients who died from causes other than prostate cancer were excluded, resulting in a cohort of 174 patients with ≥ 24-month follow-up.
Treatment
Patients eligible for staging underwent imaging according to NCCN guidelines, including computed tomography (CT) of the abdomen and pelvis, bone scintigraphy, or, in recent years, prostate-specific membrane antigen positron emission tomography, primarily used for unfavorable intermediate-risk (UIR) and high-risk (HR) cancers. Patients with a negative staging work-up for nodal or skeletal disease were included. Prior to planning the CT simulation, patients were given bowel preparation instructions, including a low-fiber and low-gas-producing diet, simethicone, and enemas, the night before and morning of the simulation. Patients were instructed to arrive with a comfortably full bladder, having not voided for 2 to 3 hours prior to the procedure. At Kansas City Veterans Affairs Medical Center (KCVAMC), SBRT treatment was generally restricted to patients with a baseline American Urological Association symptom score of 15 to 20 out of 35 and a prostate gland size < 80 mL to minimize the risk of acute urinary toxicity. We did not use intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast agents for planning CT simulation.
Patients were placed in a supine position, and a vacuum bag was used for immobilization. Following the CT simulation, the images were transferred to the Eclipse treatment planning system. The clinical target volume (CTV) encompassed the prostate and the proximal 1.0 cm of the seminal vesicles for Gleason score (GS) 1 to 2, and the entire seminal vesicle was included for GS 3 to 5, which is consistent with KCVAMC practice and established safety protocols. The planning target volume (PTV) was created by uniformly expanding the CTV by 5 to 7 mm, except for the posterior margin, which was limited to 3 to 5 mm. When elective nodal radiotherapy was planned for HR prostate cancer, the pelvic field for CT simulation started at the L-2 upper border, with the lower border extending to the lesser trochanter. The pelvic nodes were delineated per Radiation Therapy Oncology Group (RTOG) guidelines.17 The CTV nodes (CTVn), including common iliac, external and internal iliac nodes, obturator, and presacral nodes, were created by uniformly expanding the CTVn by 2 to 3 mm. Slice-by-slice corrections were made to avoid bowel overlap in these patients.
The use of androgen deprivation therapy (ADT) for a duration of 6 to 24 months was prescribed for patients with UIR or HR prostate cancer per NCCN guidelines.16 The prescribed dose to the PTV was 36.25 to 40 Gy (40 Gy was mostly used as a boost to the dominant lesion) in 5 fractions, with each fraction ranging from 7.25 to 8 Gy. For elective nodal radiotherapy in patients at HR, the prescribed dose was 25 Gy in 5 fractions. All patients were planned for VMAT, which aims to deliver ≥ 95% of the prescription dose to 95% of the PTV. Once the physician approved the treatment plan and physics quality assessment was completed, treatments commenced on an every-other-day schedule. Patients received the same bowel preparation instructions for each treatment as for the planning CT simulation. Daily treatment accuracy was confirmed via daily 3-dimensional cone-beam CT (CBCT) for IGRT. No fiducials or hydrogel rectal spacers were used.
Follow-up Schedule and Toxicity Assessment
Follow-up assessments were conducted 4 to 6 weeks after radiation therapy and then repeated every 6 months for 2 to 5 years, and annually thereafter. At each follow-up visit, patients were evaluated for genitourinary (GU) and gastrointestinal (GI) toxicity, according to RTOG toxicity criteria. Prostate-specific antigen (PSA) levels were monitored; in patients receiving ADT, testosterone levels were also checked.
Statistical Analysis
Biochemical failure was defined using the Phoenix definition (nadir PSA + 2 ng/mL). Differences between dose cohorts were assessed using the log-rank test for survival outcomes and X2 testing for categorical variables. GU and GI toxicities were summarized as cumulative incidences of RTOG grade ≥ II events. Statistical significance was set at P < .05.
RESULTS
One hundred seventy-four patients were included in the retrospective review. Patients had a median follow-up of 45 months (range, 24-111) (Figure). The median age at treatment was 74 years (range, 51-88), and the median pretreatment PSA level was 11.9 ng/mL (range, 0.6-69.5). Twenty-six patients (14.9%) had a GS 1, 77 (44.3%) had GS 2, 41 (23.6%) had GS 3, 18 (10.3%) had GS 4, and 12 (6.9%) had GS 5. Fifty-one patients (29.3%) received elective pelvic nodal radiotherapy, and 93 patients (53.4%) received ADT (Table 1).
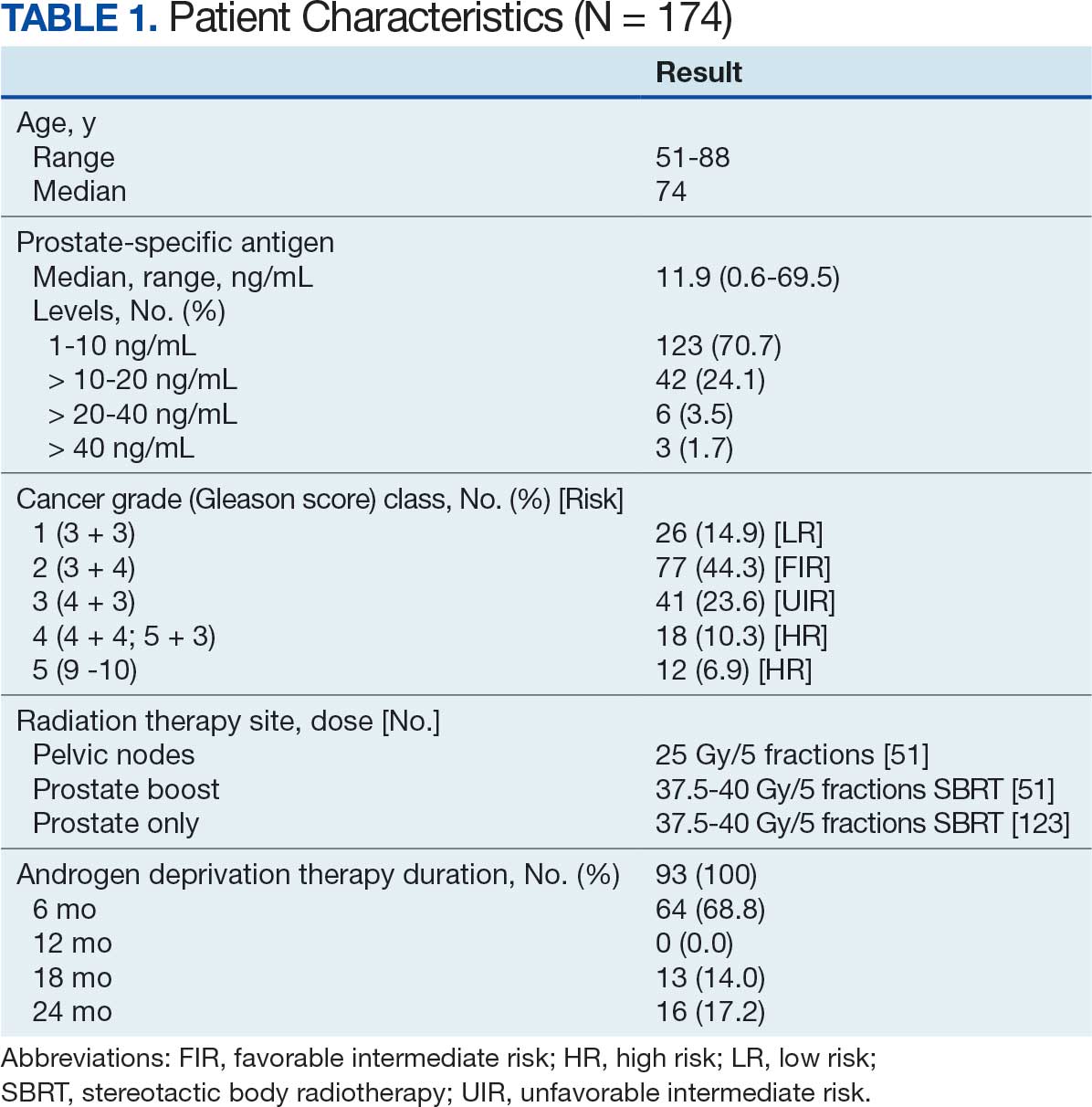
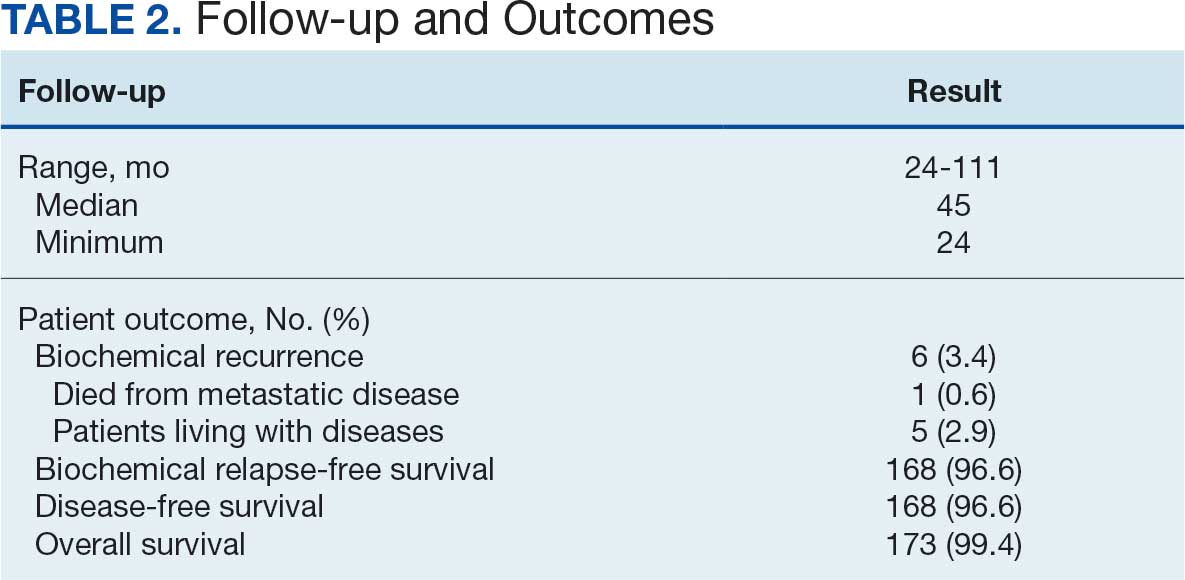
At 24 months follow-up, 6 patients (3.4%) had biochemical failures. One patient died from metastatic prostate cancer, and 5 patients are living with biochemical failure (Table 2). The actuarial 5-year overall survival (OS) rate was 99.4%, and the 5-year disease-free survival (DFS) rate was 96.6%. We performed a subanalysis comparing outcomes of the 36.25 Gy vs 40 Gy SBRT cohorts. There was no statistically significant difference in DFS, OS, or the cumulative incidence of grade II/III toxicity between patients treated with 40 Gy vs 36.25 Gy. Outcomes stratified by NCCN risk groups (low, intermediate, high/very high) are detailed in Table 3. As expected, DFS was slightly lower in the high-risk group, but overall disease control remained high across all stratifications.
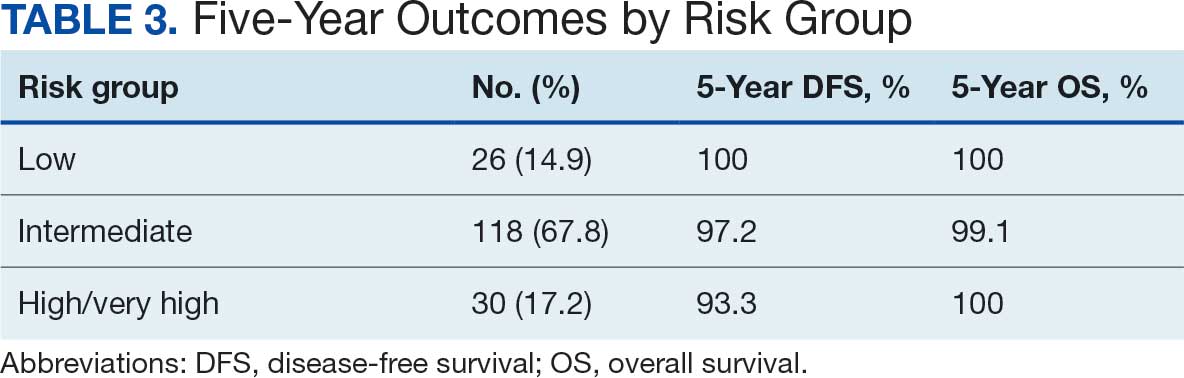
The cumulative incidence of RTOG grade II and higher GU toxicity was 28.2% (Table 4). This included 46 patients (26.4%) with grade II GU toxicity and 2 patients (1.2%) who developed grade III GU complications (1 requiring self-catheterization and another a suprapubic catheter for urinary retention). One patient (0.6%) treated with a 40 Gy dose regimen experienced a grade IV GU complication in the form of a rectovesical fistula necessitating surgical intervention.
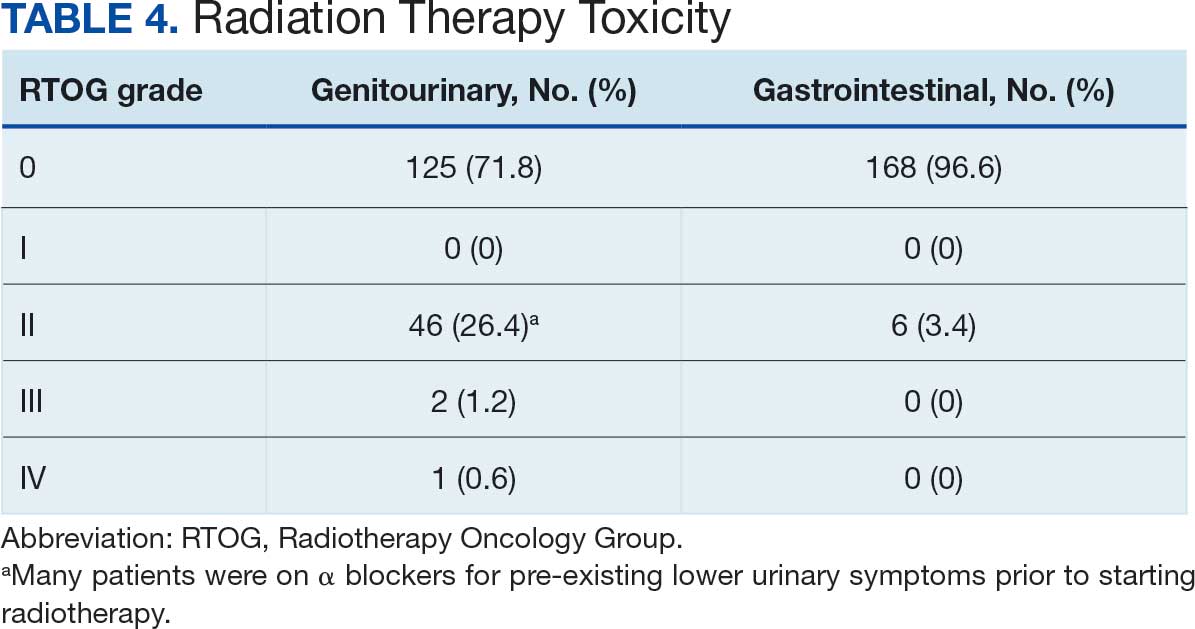
The cumulative incidence of RTOG grade II or higher GI toxicity was 3.4%, and no grade III or IV gastrointestinal toxicities were observed during the follow-up period. Importantly, intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast were not routinely used in this cohort of patients.
The high rates of actuarial 5-year DFS and OS observed suggest a favorable initial response to the SBRT regimen employed at KCVAMC. However, given the potential for late recurrence in patients with prostate cancer, longer follow-up is essential to determine the durability of these outcomes. The observed GU toxicity rate of 28.2% for grade II and higher events warrants careful consideration and compares with other published data on SBRT for prostate cancer.15 The occurrence of a grade IV rectovesical fistula, although rare, is a notable adverse event that warrants discussion in the context of the treatment approach. The low incidence of grade II or higher GI toxicity is an encouraging finding, particularly given that hydrogel rectal spacers are not routinely used to minimize rectal exposure.
DISCUSSION
The primary objective of this retrospective study was to evaluate the outcomes of SBRT for patients with localized prostate cancer treated at KCVAMC and to compare these results with those reported in the literature. Our findings demonstrate promising intermediate-term efficacy, with an estimated 5-year DFS of 96.6% and OS of 99.4% at a median follow-up of 45 months. Furthermore, the observed toxicity profile appears acceptable, with a cumulative grade II and higher GU toxicity rate of 28.2% and a grade II or higher GI toxicity rate of 3.4%. Notably, these outcomes were achieved without the routine use of intraprostatic fiducials or hydrogel rectal spacers.
Two pivotal randomized phase 3 trials have established the noninferiority of ultrahypofractionated radiotherapy (UHRT) with SBRT over conventional fractionation. The HYPO-RT-PC trial compared SBRT (42.7 Gy in 7 fractions) with conventional fractionation (78 Gy in 39 fractions) in intermediate- and high-risk patients with prostate cancer and reported a 5-year biochemical relapse-free survival of 84% in both arms.9 The PACE-B trial, which included patients at low- and intermediate-risk, compared SBRT (36.25 Gy in 5 fractions) with conventional or moderate HFRT and reported a 5-year biochemical control rate of 95.8% in the SBRT arm and 94.6% in the control arm.15
A comprehensive review and meta-analysis of 7 phase 3 studies involving 6795 patients compared different radiotherapy regimens, namely, UHRT, HFRT, and CFRT, and reported that after 5 years, the DFS rates were 85.1% for CFRT, 86% for HFRT, and 85% for UHRT, with no significant difference in toxicity among the 3 different treatment approaches.18 This suggests that shorter, more intense radiotherapy schedules (UHRT and HFRT) may be as effective and safe as traditional, longer courses of radiation.
There are multiple published nonrandomized prospective trials in which thousands of patients with extreme hypofractionation have been treated with different doses, fractions, and techniques. While heterogeneity and limited long-term follow-up in the existing evidence are acknowledged, these data suggest that prostate SBRT provides appropriate biochemical control with few high-grade toxicities, supporting its ongoing global use and justifying further prospective investigations. Comparative data are shown in Table 5. Several ongoing studies are evaluating noninferiority, superiority, and cost-effectiveness using different methodologies (Table 6).9,15,19-24
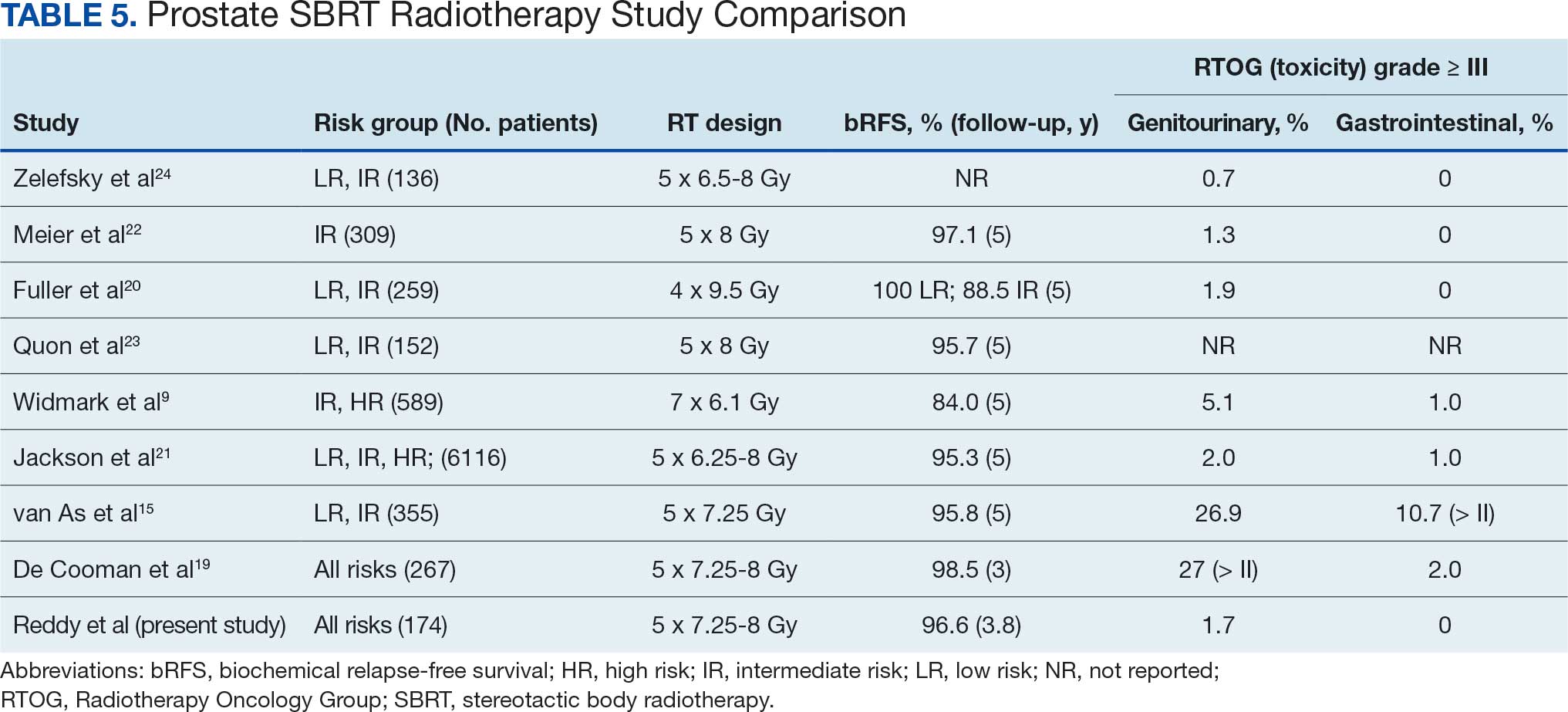
This study’s efficacy outcomes, particularly the high DFS rate, are consistent with the findings from these landmark trials, suggesting that the SBRT regimen used at KCVAMC is effective in achieving early disease control despite 17.2% of patients having high-risk disease. The GU toxicity observed in this study, with a 28.2% rate of grade II or higher events, is also comparable with the 26.9% reported in the 5-fraction SBRT arm of the PACE-B trial, which had a longer median follow-up of 74 months.15 It is important to note that a portion of these grade II events occurred in patients who were already on a blockers for pre-existing lower urinary tract symptoms before starting radiotherapy, which may inflate the observed cumulative acute toxicity score.
A critical comparison is how SBRT toxicity aligns with moderate hypofractionation (eg, 60 Gy in 20 fractions or 70 Gy in 28 fractions as reported by others).4,6 Our observed grade III and higher GU toxicity rate (1.7%) and grade III and higher GI toxicity rate (0%) are highly favorable when compared with historical moderate hypofractionation data, which typically report grade III GU toxicity in the range of 2% to 3% and grade III GI toxicity around 1% to 2%. This suggests that despite the higher dose per fraction, SBRT does not necessarily lead to increased severe acute toxicity, potentially offering a superior therapeutic ratio for GI and GU sparing.
However, the occurrence of a grade IV rectovesical fistula in 1 patient (0.6%)—who received the 40 Gy dose—was a serious complication that warrants careful consideration. This rare, but severe, complication in the higher dose cohort underscores the potential for increased organ-at-risk toxicity, particularly in the absence of a hydrogel rectal spacer, which is designed to mitigate high-dose rectal exposure. While the overall rate of significant GU toxicity remains low, this event highlights the potential risks associated with SBRT. Hydrogel rectal spacers are designed to increase the distance between the prostate and the rectum, which can reduce the rectal radiation dose and potentially mitigate the risk of such fistulas. The low rate of grade II or worse GI toxicity (3.4%) in our study is noteworthy, especially considering that hydrogel spacers were not routinely used. This finding aligns with the 2.5% GI toxicity rate reported in the SBRT arm of the PACE-B trial, suggesting that careful treatment planning and delivery techniques, such as VMAT-IMRT and daily CBCT for IGRT, may contribute to minimizing GI toxicity even without the use of rectal spacers.15 The exclusive use of 3-dimensional CBCT for IGRT in our study, without the use of fiducial markers, suggests that accurate target localization can be achieved with this approach, contributing to the observed efficacy and reduced toxicity.
Strengths and Limitations
This study’s retrospective, single-center design may have introduced selection bias. The median follow-up of 45 months, while substantial, is still relatively short for assessing very late toxicities and long-term oncologic outcomes in prostate cancer, which is known for late recurrences. Additionally, the lack of a direct comparison group within KCVAMC limits the ability to definitively attribute the observed outcomes solely to SBRT treatment. However, the strengths of this study include the inclusion of a consecutive series of veteran patients with localized prostate cancer across all risk categories, providing a real-world perspective on SBRT outcomes in a diverse patient population. Furthermore, the detailed assessment of efficacy and toxicity via standardized RTOG criteria enhances the comparability of our findings with those of other published prospective studies, despite the retrospective nature of the data.
CONCLUSIONS
This single-institution retrospective analysis revealed that short-term SBRT (36.25 to 40 Gy in 5 fractions), with a minimum follow-up of 24 months and a median follow-up of 45 months, for localized prostate cancer, including patients at HR, is associated with promising early efficacy and acceptable toxicity, even in the absence of routine fiducial or hydrogel spacer use. The favorable actuarial 5-year DFS and OS rates, coupled with a manageable toxicity profile, suggest that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer. However, a longer follow-up is necessary to confirm these findings and fully characterize the long-term efficacy and toxicity of this SBRT regimen. Nevertheless, the results contribute to the growing body of evidence suggesting that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer.
- Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10-45. doi:10.3322/caac.21871
- Donovan JL, Hamdy FC, Lane JA, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437. doi:10.1056/NEJMoa1606221
- Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424. doi:10.1056/NEJMoa1606220
- Dearnaley D, Syndikus I, Mossop H, et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016;17:1047-1060. doi:10.1016/S1470-2045(16)30102-4
- Catton CN, Lukka H, Gu CS, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol. 2017;35:1884-1890. doi:10.1200/JCO.2016.71.7397
- Lee WR, Dignam JJ, Amin MB, et al. Long-term analysis of NRG Oncology RTOG 0415: a randomized phase III noninferiority study comparing two fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2024;42:2377-2381. doi:10.1200/JCO.23.02445
- de Vries KC, Wortel RC, Oomen-de Hoop E, et al. Hypofractionated versus conventionally fractionated radiation therapy for patients with intermediate- or high-risk, localized, prostate cancer: 7-year outcomes from the randomized, multicenter, open-label, phase 3 HYPRO trial. Int J Radiat Oncol Biol Phys. 2020;106:108-115. doi:10.1016/j.ijrobp.2019.09.007
- Incrocci L, Wortel RC, Alemayehu WG, et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016;17:1061-1069. doi:10.1016/S1470-2045(16)30070-5
- Widmark A, Gunnlaugsson A, Beckman L, et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019;394:385-395. doi:10.1016/S0140-6736(19)31131-6
- Brenner DJ, Hall EJ. Fractionation and protraction for radiotherapy of prostate carcinoma. Int J Radiat Oncol Biol Phys. 1999;43:1095-101. doi:10.1016/s0360-3016(98)00438-6
- Dasu A. Is the alpha/beta value for prostate tumours low enough to be safely used in clinical trials? Clin Oncol (R Coll Radiol). 2007;19:289-301. doi:10.1016/j.clon.2007.02.007
- Garcia-Barros M, Paris F, Cordon-Cardo C, et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science. 2003;300:1155-1159. doi:10.1126/science.1082504
- Gulliford S, Hall E, Dearnaley D. Hypofractionation trials and radiobiology of prostate cancer. Oncoscience. 2017;4:27-28. doi:10.18632/oncoscience.347
- Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8:89-91. doi:10.1016/j.ccr.2005.07.014
- van As N, Griffin C, Tree A, et al. Phase 3 Trial of stereotactic body radiotherapy in localized prostate cancer. N Engl J Med. Oct 17 2024;391:1413-1425. doi:10.1056/NEJMoa2403365
- National Comprehensive Cancer Network. NCCN Guidelines Version 5. 2026 Prostate Cancer. Accessed March 24, 2026. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- Lawton CA, Michalski J, El-Naqa I, et al. RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2009;74:383-387. doi:10.1016/j.ijrobp.2008.08.002
- Lehrer EJ, Kishan AU, Yu JB, et al. Ultrahypofractionated versus hypofractionated and conventionally fractionated radiation therapy for localized prostate cancer: a systematic review and meta-analysis of phase III randomized trials. Radiother Oncol. 2020;148:235-242. doi:10.1016/j.radonc.2020.04.037
- De Cooman B, Debacker T, Adams T, et al. Stereotactic body radiotherapy (SBRT) as a treatment for localized prostate cancer: a retrospective analysis. Radiat Oncol. 2025;20:25. doi:10.1186/s13014-025-02598-8
- Fuller DB, Falchook AD, Crabtree T, et al. Phase 2 multicenter trial of heterogeneous-dosing stereotactic body radiotherapy for low- and intermediate-risk prostate cancer: 5-year outcomes. Eur Urol Oncol. 2018;1:540-547. doi:10.1016/j.euo.2018.06.013
- Jackson WC, Silva J, Hartman HE, et al. Stereotactic body radiation therapy for localized prostate cancer: a systematic review and meta-analysis of over 6,000 patients treated on prospective studies. Int J Radiat Oncol Biol Phys. 2019;104:778-789. doi:10.1016/j.ijrobp.2019.03.051
- Meier RM, Bloch DA, Cotrutz C, et al. Multicenter trial of stereotactic body radiation therapy for low- and intermediate-risk prostate cancer: survival and toxicity endpoints. nt J Radiat Oncol Biol Phys. 2018;102:296-303. doi:10.1016/j.ijrobp.2018.05.040
- Quon HC, Ong A, Cheung P, et al. Once-weekly versus every-other-day stereotactic body radiotherapy in patients with prostate cancer (PATRIOT): a phase 2 randomized trial. Radiother Oncol. 2018;127:206-212. doi:10.1016/j.radonc.2018.02.029
- Zelefsky MJ, Kollmeier M, McBride S, et al. Five-year outcomes of a phase 1 dose-escalation study using stereotactic body radiosurgery for patients with low-risk and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2019;104:42-49. doi:10.1016/j.ijrobp.2018.12.045
Prostate cancer is the most common cancer in US males, with an estimated 313,780 new cases and 35,770 deaths in 2025.1 Several treatment options are available for localized prostate cancer that have similar outcomes, including active surveillance for low-risk cancers, surgery, or radiotherapy.2,3 Conventional fractionation radiotherapy (CFRT) with 40 to 45 fractions over 8 to 9 weeks has been used for decades. Over the past 2 decades, moderate hypofractionation schedules with 2.4 to 3.4 Gy per fraction over 20 to 28 fractions have become standard, as many noninferiority randomized clinical trials (RCTs) such as CHHiP (UK),4 PROFIT (Canada and Europe),5 NRG Oncology RTOG 0415 (US),6 HYPRO (Netherlands),7,8 and HYPO-RT-PC (Sweden and Denmark),9 have shown the noninferiority of moderately hypofractionated radiotherapy compared with CFRT. Notably, most of these noninferiority studies primarily included patients with low- or intermediate-risk prostate cancer, except for the HYPO-RT-PC trial,9 which also included patients with intermediate- and high-risk prostate cancer.
These noninferiority studies, along with technological advances in radiotherapy, such as intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), and image-guided radiotherapy (IGRT), paved the path to ultrahypofractionated stereotactic body radiotherapy (SBRT) that is delivered in 5 fractions of ≥ 6 Gy. This high dose per fraction may have a radiobiologic advantage over conventional fractionation. The relatively low a/ß ratio of prostate cancer, estimated to be between 1 and 2, suggests that tumor cells may be particularly sensitive to the high doses per fraction delivered in SBRT.10-13 Compared with CFRT, SBRT-induced tumor cell death may also be mediated through different pathways; this pathway appears to be generated in a dose-dependent manner, particularly with doses > 8 Gy per fraction.14,15 Additionally, the higher a/ß ratio for the surrounding organs at risk, such as the bladder and rectum, theoretically allows for an improved therapeutic ratio window that maximizes tumor control while minimizing damage to healthy tissues.
A substantial body of evidence from prospective studies and meta-analyses supports the use of SBRT for localized prostate cancer. HYPO-RT-PC, a significant phase 3 noninferiority study, enrolled 1200 patients with intermediate (89%) and high-risk (11%) prostate cancer randomized between 2 arms, including CFRT to 78 Gy in 39 fractions and SBRT to 42.7 Gy in 7 fractions, treated 3 days weekly. After a median follow-up of 60 months, the estimated 5-year biochemical relapse-free survival rate was 84% in both groups.9 This trial was notable because it was the first randomized study to demonstrate that SBRT was noninferior to CFRT in intermediate- and high-risk prostate cancer patients. Another pivotal phase 3 trial, the PACE-B study, enrolled 874 patients to compare SBRT (36.25 Gy to the prostate gland, with a secondary dose of 40 Gy to the gross tumor volume where applicable, in 5 fractions) with CFRT (78 Gy in 39 fractions) and moderately hypofractionated radiotherapy (HFRT) (62 Gy in 20 fractions) in patients with low- or intermediate-risk prostate cancer. With a 74-month median follow-up, the study reported 5-year biochemical free rates of 94.6% for CFRT and 95.8% for SBRT, confirming the noninferiority of SBRT to CFRT.15
SBRT offers short, effective, and convenient treatment to many patients with localized prostate cancer. While previous guidelines were more restrictive, the March 2026 National Comprehensive Cancer Network (NCCN) guidelines now list SBRT as a preferred treatment modality for high-risk prostate cancer.16
Given the growing body of evidence supporting the efficacy and safety of SBRT, we implemented an SBRT program in 2014 at a tertiary care center for veterans. This retrospective study was undertaken to evaluate the early efficacy and toxicity of SBRT in patients with localized prostate cancer treated at our institution, including patients across all risk stratifications.
METHODS
We identified 242 patients diagnosed with prostate cancer who underwent SBRT treatment between November 2014 and October 2024 at Overland Park Veterans Affairs Radiation Oncology Clinic. For the final analysis, 46 patients with < 2 years of follow-up and 22 patients who died from causes other than prostate cancer were excluded, resulting in a cohort of 174 patients with ≥ 24-month follow-up.
Treatment
Patients eligible for staging underwent imaging according to NCCN guidelines, including computed tomography (CT) of the abdomen and pelvis, bone scintigraphy, or, in recent years, prostate-specific membrane antigen positron emission tomography, primarily used for unfavorable intermediate-risk (UIR) and high-risk (HR) cancers. Patients with a negative staging work-up for nodal or skeletal disease were included. Prior to planning the CT simulation, patients were given bowel preparation instructions, including a low-fiber and low-gas-producing diet, simethicone, and enemas, the night before and morning of the simulation. Patients were instructed to arrive with a comfortably full bladder, having not voided for 2 to 3 hours prior to the procedure. At Kansas City Veterans Affairs Medical Center (KCVAMC), SBRT treatment was generally restricted to patients with a baseline American Urological Association symptom score of 15 to 20 out of 35 and a prostate gland size < 80 mL to minimize the risk of acute urinary toxicity. We did not use intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast agents for planning CT simulation.
Patients were placed in a supine position, and a vacuum bag was used for immobilization. Following the CT simulation, the images were transferred to the Eclipse treatment planning system. The clinical target volume (CTV) encompassed the prostate and the proximal 1.0 cm of the seminal vesicles for Gleason score (GS) 1 to 2, and the entire seminal vesicle was included for GS 3 to 5, which is consistent with KCVAMC practice and established safety protocols. The planning target volume (PTV) was created by uniformly expanding the CTV by 5 to 7 mm, except for the posterior margin, which was limited to 3 to 5 mm. When elective nodal radiotherapy was planned for HR prostate cancer, the pelvic field for CT simulation started at the L-2 upper border, with the lower border extending to the lesser trochanter. The pelvic nodes were delineated per Radiation Therapy Oncology Group (RTOG) guidelines.17 The CTV nodes (CTVn), including common iliac, external and internal iliac nodes, obturator, and presacral nodes, were created by uniformly expanding the CTVn by 2 to 3 mm. Slice-by-slice corrections were made to avoid bowel overlap in these patients.
The use of androgen deprivation therapy (ADT) for a duration of 6 to 24 months was prescribed for patients with UIR or HR prostate cancer per NCCN guidelines.16 The prescribed dose to the PTV was 36.25 to 40 Gy (40 Gy was mostly used as a boost to the dominant lesion) in 5 fractions, with each fraction ranging from 7.25 to 8 Gy. For elective nodal radiotherapy in patients at HR, the prescribed dose was 25 Gy in 5 fractions. All patients were planned for VMAT, which aims to deliver ≥ 95% of the prescription dose to 95% of the PTV. Once the physician approved the treatment plan and physics quality assessment was completed, treatments commenced on an every-other-day schedule. Patients received the same bowel preparation instructions for each treatment as for the planning CT simulation. Daily treatment accuracy was confirmed via daily 3-dimensional cone-beam CT (CBCT) for IGRT. No fiducials or hydrogel rectal spacers were used.
Follow-up Schedule and Toxicity Assessment
Follow-up assessments were conducted 4 to 6 weeks after radiation therapy and then repeated every 6 months for 2 to 5 years, and annually thereafter. At each follow-up visit, patients were evaluated for genitourinary (GU) and gastrointestinal (GI) toxicity, according to RTOG toxicity criteria. Prostate-specific antigen (PSA) levels were monitored; in patients receiving ADT, testosterone levels were also checked.
Statistical Analysis
Biochemical failure was defined using the Phoenix definition (nadir PSA + 2 ng/mL). Differences between dose cohorts were assessed using the log-rank test for survival outcomes and X2 testing for categorical variables. GU and GI toxicities were summarized as cumulative incidences of RTOG grade ≥ II events. Statistical significance was set at P < .05.
RESULTS
One hundred seventy-four patients were included in the retrospective review. Patients had a median follow-up of 45 months (range, 24-111) (Figure). The median age at treatment was 74 years (range, 51-88), and the median pretreatment PSA level was 11.9 ng/mL (range, 0.6-69.5). Twenty-six patients (14.9%) had a GS 1, 77 (44.3%) had GS 2, 41 (23.6%) had GS 3, 18 (10.3%) had GS 4, and 12 (6.9%) had GS 5. Fifty-one patients (29.3%) received elective pelvic nodal radiotherapy, and 93 patients (53.4%) received ADT (Table 1).


At 24 months follow-up, 6 patients (3.4%) had biochemical failures. One patient died from metastatic prostate cancer, and 5 patients are living with biochemical failure (Table 2). The actuarial 5-year overall survival (OS) rate was 99.4%, and the 5-year disease-free survival (DFS) rate was 96.6%. We performed a subanalysis comparing outcomes of the 36.25 Gy vs 40 Gy SBRT cohorts. There was no statistically significant difference in DFS, OS, or the cumulative incidence of grade II/III toxicity between patients treated with 40 Gy vs 36.25 Gy. Outcomes stratified by NCCN risk groups (low, intermediate, high/very high) are detailed in Table 3. As expected, DFS was slightly lower in the high-risk group, but overall disease control remained high across all stratifications.


The cumulative incidence of RTOG grade II and higher GU toxicity was 28.2% (Table 4). This included 46 patients (26.4%) with grade II GU toxicity and 2 patients (1.2%) who developed grade III GU complications (1 requiring self-catheterization and another a suprapubic catheter for urinary retention). One patient (0.6%) treated with a 40 Gy dose regimen experienced a grade IV GU complication in the form of a rectovesical fistula necessitating surgical intervention.

The cumulative incidence of RTOG grade II or higher GI toxicity was 3.4%, and no grade III or IV gastrointestinal toxicities were observed during the follow-up period. Importantly, intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast were not routinely used in this cohort of patients.
The high rates of actuarial 5-year DFS and OS observed suggest a favorable initial response to the SBRT regimen employed at KCVAMC. However, given the potential for late recurrence in patients with prostate cancer, longer follow-up is essential to determine the durability of these outcomes. The observed GU toxicity rate of 28.2% for grade II and higher events warrants careful consideration and compares with other published data on SBRT for prostate cancer.15 The occurrence of a grade IV rectovesical fistula, although rare, is a notable adverse event that warrants discussion in the context of the treatment approach. The low incidence of grade II or higher GI toxicity is an encouraging finding, particularly given that hydrogel rectal spacers are not routinely used to minimize rectal exposure.
DISCUSSION
The primary objective of this retrospective study was to evaluate the outcomes of SBRT for patients with localized prostate cancer treated at KCVAMC and to compare these results with those reported in the literature. Our findings demonstrate promising intermediate-term efficacy, with an estimated 5-year DFS of 96.6% and OS of 99.4% at a median follow-up of 45 months. Furthermore, the observed toxicity profile appears acceptable, with a cumulative grade II and higher GU toxicity rate of 28.2% and a grade II or higher GI toxicity rate of 3.4%. Notably, these outcomes were achieved without the routine use of intraprostatic fiducials or hydrogel rectal spacers.
Two pivotal randomized phase 3 trials have established the noninferiority of ultrahypofractionated radiotherapy (UHRT) with SBRT over conventional fractionation. The HYPO-RT-PC trial compared SBRT (42.7 Gy in 7 fractions) with conventional fractionation (78 Gy in 39 fractions) in intermediate- and high-risk patients with prostate cancer and reported a 5-year biochemical relapse-free survival of 84% in both arms.9 The PACE-B trial, which included patients at low- and intermediate-risk, compared SBRT (36.25 Gy in 5 fractions) with conventional or moderate HFRT and reported a 5-year biochemical control rate of 95.8% in the SBRT arm and 94.6% in the control arm.15
A comprehensive review and meta-analysis of 7 phase 3 studies involving 6795 patients compared different radiotherapy regimens, namely, UHRT, HFRT, and CFRT, and reported that after 5 years, the DFS rates were 85.1% for CFRT, 86% for HFRT, and 85% for UHRT, with no significant difference in toxicity among the 3 different treatment approaches.18 This suggests that shorter, more intense radiotherapy schedules (UHRT and HFRT) may be as effective and safe as traditional, longer courses of radiation.
There are multiple published nonrandomized prospective trials in which thousands of patients with extreme hypofractionation have been treated with different doses, fractions, and techniques. While heterogeneity and limited long-term follow-up in the existing evidence are acknowledged, these data suggest that prostate SBRT provides appropriate biochemical control with few high-grade toxicities, supporting its ongoing global use and justifying further prospective investigations. Comparative data are shown in Table 5. Several ongoing studies are evaluating noninferiority, superiority, and cost-effectiveness using different methodologies (Table 6).9,15,19-24


This study’s efficacy outcomes, particularly the high DFS rate, are consistent with the findings from these landmark trials, suggesting that the SBRT regimen used at KCVAMC is effective in achieving early disease control despite 17.2% of patients having high-risk disease. The GU toxicity observed in this study, with a 28.2% rate of grade II or higher events, is also comparable with the 26.9% reported in the 5-fraction SBRT arm of the PACE-B trial, which had a longer median follow-up of 74 months.15 It is important to note that a portion of these grade II events occurred in patients who were already on a blockers for pre-existing lower urinary tract symptoms before starting radiotherapy, which may inflate the observed cumulative acute toxicity score.
A critical comparison is how SBRT toxicity aligns with moderate hypofractionation (eg, 60 Gy in 20 fractions or 70 Gy in 28 fractions as reported by others).4,6 Our observed grade III and higher GU toxicity rate (1.7%) and grade III and higher GI toxicity rate (0%) are highly favorable when compared with historical moderate hypofractionation data, which typically report grade III GU toxicity in the range of 2% to 3% and grade III GI toxicity around 1% to 2%. This suggests that despite the higher dose per fraction, SBRT does not necessarily lead to increased severe acute toxicity, potentially offering a superior therapeutic ratio for GI and GU sparing.
However, the occurrence of a grade IV rectovesical fistula in 1 patient (0.6%)—who received the 40 Gy dose—was a serious complication that warrants careful consideration. This rare, but severe, complication in the higher dose cohort underscores the potential for increased organ-at-risk toxicity, particularly in the absence of a hydrogel rectal spacer, which is designed to mitigate high-dose rectal exposure. While the overall rate of significant GU toxicity remains low, this event highlights the potential risks associated with SBRT. Hydrogel rectal spacers are designed to increase the distance between the prostate and the rectum, which can reduce the rectal radiation dose and potentially mitigate the risk of such fistulas. The low rate of grade II or worse GI toxicity (3.4%) in our study is noteworthy, especially considering that hydrogel spacers were not routinely used. This finding aligns with the 2.5% GI toxicity rate reported in the SBRT arm of the PACE-B trial, suggesting that careful treatment planning and delivery techniques, such as VMAT-IMRT and daily CBCT for IGRT, may contribute to minimizing GI toxicity even without the use of rectal spacers.15 The exclusive use of 3-dimensional CBCT for IGRT in our study, without the use of fiducial markers, suggests that accurate target localization can be achieved with this approach, contributing to the observed efficacy and reduced toxicity.
Strengths and Limitations
This study’s retrospective, single-center design may have introduced selection bias. The median follow-up of 45 months, while substantial, is still relatively short for assessing very late toxicities and long-term oncologic outcomes in prostate cancer, which is known for late recurrences. Additionally, the lack of a direct comparison group within KCVAMC limits the ability to definitively attribute the observed outcomes solely to SBRT treatment. However, the strengths of this study include the inclusion of a consecutive series of veteran patients with localized prostate cancer across all risk categories, providing a real-world perspective on SBRT outcomes in a diverse patient population. Furthermore, the detailed assessment of efficacy and toxicity via standardized RTOG criteria enhances the comparability of our findings with those of other published prospective studies, despite the retrospective nature of the data.
CONCLUSIONS
This single-institution retrospective analysis revealed that short-term SBRT (36.25 to 40 Gy in 5 fractions), with a minimum follow-up of 24 months and a median follow-up of 45 months, for localized prostate cancer, including patients at HR, is associated with promising early efficacy and acceptable toxicity, even in the absence of routine fiducial or hydrogel spacer use. The favorable actuarial 5-year DFS and OS rates, coupled with a manageable toxicity profile, suggest that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer. However, a longer follow-up is necessary to confirm these findings and fully characterize the long-term efficacy and toxicity of this SBRT regimen. Nevertheless, the results contribute to the growing body of evidence suggesting that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer.
Prostate cancer is the most common cancer in US males, with an estimated 313,780 new cases and 35,770 deaths in 2025.1 Several treatment options are available for localized prostate cancer that have similar outcomes, including active surveillance for low-risk cancers, surgery, or radiotherapy.2,3 Conventional fractionation radiotherapy (CFRT) with 40 to 45 fractions over 8 to 9 weeks has been used for decades. Over the past 2 decades, moderate hypofractionation schedules with 2.4 to 3.4 Gy per fraction over 20 to 28 fractions have become standard, as many noninferiority randomized clinical trials (RCTs) such as CHHiP (UK),4 PROFIT (Canada and Europe),5 NRG Oncology RTOG 0415 (US),6 HYPRO (Netherlands),7,8 and HYPO-RT-PC (Sweden and Denmark),9 have shown the noninferiority of moderately hypofractionated radiotherapy compared with CFRT. Notably, most of these noninferiority studies primarily included patients with low- or intermediate-risk prostate cancer, except for the HYPO-RT-PC trial,9 which also included patients with intermediate- and high-risk prostate cancer.
These noninferiority studies, along with technological advances in radiotherapy, such as intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), and image-guided radiotherapy (IGRT), paved the path to ultrahypofractionated stereotactic body radiotherapy (SBRT) that is delivered in 5 fractions of ≥ 6 Gy. This high dose per fraction may have a radiobiologic advantage over conventional fractionation. The relatively low a/ß ratio of prostate cancer, estimated to be between 1 and 2, suggests that tumor cells may be particularly sensitive to the high doses per fraction delivered in SBRT.10-13 Compared with CFRT, SBRT-induced tumor cell death may also be mediated through different pathways; this pathway appears to be generated in a dose-dependent manner, particularly with doses > 8 Gy per fraction.14,15 Additionally, the higher a/ß ratio for the surrounding organs at risk, such as the bladder and rectum, theoretically allows for an improved therapeutic ratio window that maximizes tumor control while minimizing damage to healthy tissues.
A substantial body of evidence from prospective studies and meta-analyses supports the use of SBRT for localized prostate cancer. HYPO-RT-PC, a significant phase 3 noninferiority study, enrolled 1200 patients with intermediate (89%) and high-risk (11%) prostate cancer randomized between 2 arms, including CFRT to 78 Gy in 39 fractions and SBRT to 42.7 Gy in 7 fractions, treated 3 days weekly. After a median follow-up of 60 months, the estimated 5-year biochemical relapse-free survival rate was 84% in both groups.9 This trial was notable because it was the first randomized study to demonstrate that SBRT was noninferior to CFRT in intermediate- and high-risk prostate cancer patients. Another pivotal phase 3 trial, the PACE-B study, enrolled 874 patients to compare SBRT (36.25 Gy to the prostate gland, with a secondary dose of 40 Gy to the gross tumor volume where applicable, in 5 fractions) with CFRT (78 Gy in 39 fractions) and moderately hypofractionated radiotherapy (HFRT) (62 Gy in 20 fractions) in patients with low- or intermediate-risk prostate cancer. With a 74-month median follow-up, the study reported 5-year biochemical free rates of 94.6% for CFRT and 95.8% for SBRT, confirming the noninferiority of SBRT to CFRT.15
SBRT offers short, effective, and convenient treatment to many patients with localized prostate cancer. While previous guidelines were more restrictive, the March 2026 National Comprehensive Cancer Network (NCCN) guidelines now list SBRT as a preferred treatment modality for high-risk prostate cancer.16
Given the growing body of evidence supporting the efficacy and safety of SBRT, we implemented an SBRT program in 2014 at a tertiary care center for veterans. This retrospective study was undertaken to evaluate the early efficacy and toxicity of SBRT in patients with localized prostate cancer treated at our institution, including patients across all risk stratifications.
METHODS
We identified 242 patients diagnosed with prostate cancer who underwent SBRT treatment between November 2014 and October 2024 at Overland Park Veterans Affairs Radiation Oncology Clinic. For the final analysis, 46 patients with < 2 years of follow-up and 22 patients who died from causes other than prostate cancer were excluded, resulting in a cohort of 174 patients with ≥ 24-month follow-up.
Treatment
Patients eligible for staging underwent imaging according to NCCN guidelines, including computed tomography (CT) of the abdomen and pelvis, bone scintigraphy, or, in recent years, prostate-specific membrane antigen positron emission tomography, primarily used for unfavorable intermediate-risk (UIR) and high-risk (HR) cancers. Patients with a negative staging work-up for nodal or skeletal disease were included. Prior to planning the CT simulation, patients were given bowel preparation instructions, including a low-fiber and low-gas-producing diet, simethicone, and enemas, the night before and morning of the simulation. Patients were instructed to arrive with a comfortably full bladder, having not voided for 2 to 3 hours prior to the procedure. At Kansas City Veterans Affairs Medical Center (KCVAMC), SBRT treatment was generally restricted to patients with a baseline American Urological Association symptom score of 15 to 20 out of 35 and a prostate gland size < 80 mL to minimize the risk of acute urinary toxicity. We did not use intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast agents for planning CT simulation.
Patients were placed in a supine position, and a vacuum bag was used for immobilization. Following the CT simulation, the images were transferred to the Eclipse treatment planning system. The clinical target volume (CTV) encompassed the prostate and the proximal 1.0 cm of the seminal vesicles for Gleason score (GS) 1 to 2, and the entire seminal vesicle was included for GS 3 to 5, which is consistent with KCVAMC practice and established safety protocols. The planning target volume (PTV) was created by uniformly expanding the CTV by 5 to 7 mm, except for the posterior margin, which was limited to 3 to 5 mm. When elective nodal radiotherapy was planned for HR prostate cancer, the pelvic field for CT simulation started at the L-2 upper border, with the lower border extending to the lesser trochanter. The pelvic nodes were delineated per Radiation Therapy Oncology Group (RTOG) guidelines.17 The CTV nodes (CTVn), including common iliac, external and internal iliac nodes, obturator, and presacral nodes, were created by uniformly expanding the CTVn by 2 to 3 mm. Slice-by-slice corrections were made to avoid bowel overlap in these patients.
The use of androgen deprivation therapy (ADT) for a duration of 6 to 24 months was prescribed for patients with UIR or HR prostate cancer per NCCN guidelines.16 The prescribed dose to the PTV was 36.25 to 40 Gy (40 Gy was mostly used as a boost to the dominant lesion) in 5 fractions, with each fraction ranging from 7.25 to 8 Gy. For elective nodal radiotherapy in patients at HR, the prescribed dose was 25 Gy in 5 fractions. All patients were planned for VMAT, which aims to deliver ≥ 95% of the prescription dose to 95% of the PTV. Once the physician approved the treatment plan and physics quality assessment was completed, treatments commenced on an every-other-day schedule. Patients received the same bowel preparation instructions for each treatment as for the planning CT simulation. Daily treatment accuracy was confirmed via daily 3-dimensional cone-beam CT (CBCT) for IGRT. No fiducials or hydrogel rectal spacers were used.
Follow-up Schedule and Toxicity Assessment
Follow-up assessments were conducted 4 to 6 weeks after radiation therapy and then repeated every 6 months for 2 to 5 years, and annually thereafter. At each follow-up visit, patients were evaluated for genitourinary (GU) and gastrointestinal (GI) toxicity, according to RTOG toxicity criteria. Prostate-specific antigen (PSA) levels were monitored; in patients receiving ADT, testosterone levels were also checked.
Statistical Analysis
Biochemical failure was defined using the Phoenix definition (nadir PSA + 2 ng/mL). Differences between dose cohorts were assessed using the log-rank test for survival outcomes and X2 testing for categorical variables. GU and GI toxicities were summarized as cumulative incidences of RTOG grade ≥ II events. Statistical significance was set at P < .05.
RESULTS
One hundred seventy-four patients were included in the retrospective review. Patients had a median follow-up of 45 months (range, 24-111) (Figure). The median age at treatment was 74 years (range, 51-88), and the median pretreatment PSA level was 11.9 ng/mL (range, 0.6-69.5). Twenty-six patients (14.9%) had a GS 1, 77 (44.3%) had GS 2, 41 (23.6%) had GS 3, 18 (10.3%) had GS 4, and 12 (6.9%) had GS 5. Fifty-one patients (29.3%) received elective pelvic nodal radiotherapy, and 93 patients (53.4%) received ADT (Table 1).


At 24 months follow-up, 6 patients (3.4%) had biochemical failures. One patient died from metastatic prostate cancer, and 5 patients are living with biochemical failure (Table 2). The actuarial 5-year overall survival (OS) rate was 99.4%, and the 5-year disease-free survival (DFS) rate was 96.6%. We performed a subanalysis comparing outcomes of the 36.25 Gy vs 40 Gy SBRT cohorts. There was no statistically significant difference in DFS, OS, or the cumulative incidence of grade II/III toxicity between patients treated with 40 Gy vs 36.25 Gy. Outcomes stratified by NCCN risk groups (low, intermediate, high/very high) are detailed in Table 3. As expected, DFS was slightly lower in the high-risk group, but overall disease control remained high across all stratifications.


The cumulative incidence of RTOG grade II and higher GU toxicity was 28.2% (Table 4). This included 46 patients (26.4%) with grade II GU toxicity and 2 patients (1.2%) who developed grade III GU complications (1 requiring self-catheterization and another a suprapubic catheter for urinary retention). One patient (0.6%) treated with a 40 Gy dose regimen experienced a grade IV GU complication in the form of a rectovesical fistula necessitating surgical intervention.

The cumulative incidence of RTOG grade II or higher GI toxicity was 3.4%, and no grade III or IV gastrointestinal toxicities were observed during the follow-up period. Importantly, intraprostatic fiducials, hydrogel rectal spacers, or intravenous contrast were not routinely used in this cohort of patients.
The high rates of actuarial 5-year DFS and OS observed suggest a favorable initial response to the SBRT regimen employed at KCVAMC. However, given the potential for late recurrence in patients with prostate cancer, longer follow-up is essential to determine the durability of these outcomes. The observed GU toxicity rate of 28.2% for grade II and higher events warrants careful consideration and compares with other published data on SBRT for prostate cancer.15 The occurrence of a grade IV rectovesical fistula, although rare, is a notable adverse event that warrants discussion in the context of the treatment approach. The low incidence of grade II or higher GI toxicity is an encouraging finding, particularly given that hydrogel rectal spacers are not routinely used to minimize rectal exposure.
DISCUSSION
The primary objective of this retrospective study was to evaluate the outcomes of SBRT for patients with localized prostate cancer treated at KCVAMC and to compare these results with those reported in the literature. Our findings demonstrate promising intermediate-term efficacy, with an estimated 5-year DFS of 96.6% and OS of 99.4% at a median follow-up of 45 months. Furthermore, the observed toxicity profile appears acceptable, with a cumulative grade II and higher GU toxicity rate of 28.2% and a grade II or higher GI toxicity rate of 3.4%. Notably, these outcomes were achieved without the routine use of intraprostatic fiducials or hydrogel rectal spacers.
Two pivotal randomized phase 3 trials have established the noninferiority of ultrahypofractionated radiotherapy (UHRT) with SBRT over conventional fractionation. The HYPO-RT-PC trial compared SBRT (42.7 Gy in 7 fractions) with conventional fractionation (78 Gy in 39 fractions) in intermediate- and high-risk patients with prostate cancer and reported a 5-year biochemical relapse-free survival of 84% in both arms.9 The PACE-B trial, which included patients at low- and intermediate-risk, compared SBRT (36.25 Gy in 5 fractions) with conventional or moderate HFRT and reported a 5-year biochemical control rate of 95.8% in the SBRT arm and 94.6% in the control arm.15
A comprehensive review and meta-analysis of 7 phase 3 studies involving 6795 patients compared different radiotherapy regimens, namely, UHRT, HFRT, and CFRT, and reported that after 5 years, the DFS rates were 85.1% for CFRT, 86% for HFRT, and 85% for UHRT, with no significant difference in toxicity among the 3 different treatment approaches.18 This suggests that shorter, more intense radiotherapy schedules (UHRT and HFRT) may be as effective and safe as traditional, longer courses of radiation.
There are multiple published nonrandomized prospective trials in which thousands of patients with extreme hypofractionation have been treated with different doses, fractions, and techniques. While heterogeneity and limited long-term follow-up in the existing evidence are acknowledged, these data suggest that prostate SBRT provides appropriate biochemical control with few high-grade toxicities, supporting its ongoing global use and justifying further prospective investigations. Comparative data are shown in Table 5. Several ongoing studies are evaluating noninferiority, superiority, and cost-effectiveness using different methodologies (Table 6).9,15,19-24


This study’s efficacy outcomes, particularly the high DFS rate, are consistent with the findings from these landmark trials, suggesting that the SBRT regimen used at KCVAMC is effective in achieving early disease control despite 17.2% of patients having high-risk disease. The GU toxicity observed in this study, with a 28.2% rate of grade II or higher events, is also comparable with the 26.9% reported in the 5-fraction SBRT arm of the PACE-B trial, which had a longer median follow-up of 74 months.15 It is important to note that a portion of these grade II events occurred in patients who were already on a blockers for pre-existing lower urinary tract symptoms before starting radiotherapy, which may inflate the observed cumulative acute toxicity score.
A critical comparison is how SBRT toxicity aligns with moderate hypofractionation (eg, 60 Gy in 20 fractions or 70 Gy in 28 fractions as reported by others).4,6 Our observed grade III and higher GU toxicity rate (1.7%) and grade III and higher GI toxicity rate (0%) are highly favorable when compared with historical moderate hypofractionation data, which typically report grade III GU toxicity in the range of 2% to 3% and grade III GI toxicity around 1% to 2%. This suggests that despite the higher dose per fraction, SBRT does not necessarily lead to increased severe acute toxicity, potentially offering a superior therapeutic ratio for GI and GU sparing.
However, the occurrence of a grade IV rectovesical fistula in 1 patient (0.6%)—who received the 40 Gy dose—was a serious complication that warrants careful consideration. This rare, but severe, complication in the higher dose cohort underscores the potential for increased organ-at-risk toxicity, particularly in the absence of a hydrogel rectal spacer, which is designed to mitigate high-dose rectal exposure. While the overall rate of significant GU toxicity remains low, this event highlights the potential risks associated with SBRT. Hydrogel rectal spacers are designed to increase the distance between the prostate and the rectum, which can reduce the rectal radiation dose and potentially mitigate the risk of such fistulas. The low rate of grade II or worse GI toxicity (3.4%) in our study is noteworthy, especially considering that hydrogel spacers were not routinely used. This finding aligns with the 2.5% GI toxicity rate reported in the SBRT arm of the PACE-B trial, suggesting that careful treatment planning and delivery techniques, such as VMAT-IMRT and daily CBCT for IGRT, may contribute to minimizing GI toxicity even without the use of rectal spacers.15 The exclusive use of 3-dimensional CBCT for IGRT in our study, without the use of fiducial markers, suggests that accurate target localization can be achieved with this approach, contributing to the observed efficacy and reduced toxicity.
Strengths and Limitations
This study’s retrospective, single-center design may have introduced selection bias. The median follow-up of 45 months, while substantial, is still relatively short for assessing very late toxicities and long-term oncologic outcomes in prostate cancer, which is known for late recurrences. Additionally, the lack of a direct comparison group within KCVAMC limits the ability to definitively attribute the observed outcomes solely to SBRT treatment. However, the strengths of this study include the inclusion of a consecutive series of veteran patients with localized prostate cancer across all risk categories, providing a real-world perspective on SBRT outcomes in a diverse patient population. Furthermore, the detailed assessment of efficacy and toxicity via standardized RTOG criteria enhances the comparability of our findings with those of other published prospective studies, despite the retrospective nature of the data.
CONCLUSIONS
This single-institution retrospective analysis revealed that short-term SBRT (36.25 to 40 Gy in 5 fractions), with a minimum follow-up of 24 months and a median follow-up of 45 months, for localized prostate cancer, including patients at HR, is associated with promising early efficacy and acceptable toxicity, even in the absence of routine fiducial or hydrogel spacer use. The favorable actuarial 5-year DFS and OS rates, coupled with a manageable toxicity profile, suggest that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer. However, a longer follow-up is necessary to confirm these findings and fully characterize the long-term efficacy and toxicity of this SBRT regimen. Nevertheless, the results contribute to the growing body of evidence suggesting that SBRT is a safe and convenient treatment option for many patients with localized prostate cancer.
- Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10-45. doi:10.3322/caac.21871
- Donovan JL, Hamdy FC, Lane JA, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437. doi:10.1056/NEJMoa1606221
- Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424. doi:10.1056/NEJMoa1606220
- Dearnaley D, Syndikus I, Mossop H, et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016;17:1047-1060. doi:10.1016/S1470-2045(16)30102-4
- Catton CN, Lukka H, Gu CS, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol. 2017;35:1884-1890. doi:10.1200/JCO.2016.71.7397
- Lee WR, Dignam JJ, Amin MB, et al. Long-term analysis of NRG Oncology RTOG 0415: a randomized phase III noninferiority study comparing two fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2024;42:2377-2381. doi:10.1200/JCO.23.02445
- de Vries KC, Wortel RC, Oomen-de Hoop E, et al. Hypofractionated versus conventionally fractionated radiation therapy for patients with intermediate- or high-risk, localized, prostate cancer: 7-year outcomes from the randomized, multicenter, open-label, phase 3 HYPRO trial. Int J Radiat Oncol Biol Phys. 2020;106:108-115. doi:10.1016/j.ijrobp.2019.09.007
- Incrocci L, Wortel RC, Alemayehu WG, et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016;17:1061-1069. doi:10.1016/S1470-2045(16)30070-5
- Widmark A, Gunnlaugsson A, Beckman L, et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019;394:385-395. doi:10.1016/S0140-6736(19)31131-6
- Brenner DJ, Hall EJ. Fractionation and protraction for radiotherapy of prostate carcinoma. Int J Radiat Oncol Biol Phys. 1999;43:1095-101. doi:10.1016/s0360-3016(98)00438-6
- Dasu A. Is the alpha/beta value for prostate tumours low enough to be safely used in clinical trials? Clin Oncol (R Coll Radiol). 2007;19:289-301. doi:10.1016/j.clon.2007.02.007
- Garcia-Barros M, Paris F, Cordon-Cardo C, et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science. 2003;300:1155-1159. doi:10.1126/science.1082504
- Gulliford S, Hall E, Dearnaley D. Hypofractionation trials and radiobiology of prostate cancer. Oncoscience. 2017;4:27-28. doi:10.18632/oncoscience.347
- Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8:89-91. doi:10.1016/j.ccr.2005.07.014
- van As N, Griffin C, Tree A, et al. Phase 3 Trial of stereotactic body radiotherapy in localized prostate cancer. N Engl J Med. Oct 17 2024;391:1413-1425. doi:10.1056/NEJMoa2403365
- National Comprehensive Cancer Network. NCCN Guidelines Version 5. 2026 Prostate Cancer. Accessed March 24, 2026. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- Lawton CA, Michalski J, El-Naqa I, et al. RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2009;74:383-387. doi:10.1016/j.ijrobp.2008.08.002
- Lehrer EJ, Kishan AU, Yu JB, et al. Ultrahypofractionated versus hypofractionated and conventionally fractionated radiation therapy for localized prostate cancer: a systematic review and meta-analysis of phase III randomized trials. Radiother Oncol. 2020;148:235-242. doi:10.1016/j.radonc.2020.04.037
- De Cooman B, Debacker T, Adams T, et al. Stereotactic body radiotherapy (SBRT) as a treatment for localized prostate cancer: a retrospective analysis. Radiat Oncol. 2025;20:25. doi:10.1186/s13014-025-02598-8
- Fuller DB, Falchook AD, Crabtree T, et al. Phase 2 multicenter trial of heterogeneous-dosing stereotactic body radiotherapy for low- and intermediate-risk prostate cancer: 5-year outcomes. Eur Urol Oncol. 2018;1:540-547. doi:10.1016/j.euo.2018.06.013
- Jackson WC, Silva J, Hartman HE, et al. Stereotactic body radiation therapy for localized prostate cancer: a systematic review and meta-analysis of over 6,000 patients treated on prospective studies. Int J Radiat Oncol Biol Phys. 2019;104:778-789. doi:10.1016/j.ijrobp.2019.03.051
- Meier RM, Bloch DA, Cotrutz C, et al. Multicenter trial of stereotactic body radiation therapy for low- and intermediate-risk prostate cancer: survival and toxicity endpoints. nt J Radiat Oncol Biol Phys. 2018;102:296-303. doi:10.1016/j.ijrobp.2018.05.040
- Quon HC, Ong A, Cheung P, et al. Once-weekly versus every-other-day stereotactic body radiotherapy in patients with prostate cancer (PATRIOT): a phase 2 randomized trial. Radiother Oncol. 2018;127:206-212. doi:10.1016/j.radonc.2018.02.029
- Zelefsky MJ, Kollmeier M, McBride S, et al. Five-year outcomes of a phase 1 dose-escalation study using stereotactic body radiosurgery for patients with low-risk and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2019;104:42-49. doi:10.1016/j.ijrobp.2018.12.045
- Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025. CA Cancer J Clin. 2025;75:10-45. doi:10.3322/caac.21871
- Donovan JL, Hamdy FC, Lane JA, et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2016;375:1425-1437. doi:10.1056/NEJMoa1606221
- Hamdy FC, Donovan JL, Lane JA, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med. 2016;375:1415-1424. doi:10.1056/NEJMoa1606220
- Dearnaley D, Syndikus I, Mossop H, et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016;17:1047-1060. doi:10.1016/S1470-2045(16)30102-4
- Catton CN, Lukka H, Gu CS, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol. 2017;35:1884-1890. doi:10.1200/JCO.2016.71.7397
- Lee WR, Dignam JJ, Amin MB, et al. Long-term analysis of NRG Oncology RTOG 0415: a randomized phase III noninferiority study comparing two fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2024;42:2377-2381. doi:10.1200/JCO.23.02445
- de Vries KC, Wortel RC, Oomen-de Hoop E, et al. Hypofractionated versus conventionally fractionated radiation therapy for patients with intermediate- or high-risk, localized, prostate cancer: 7-year outcomes from the randomized, multicenter, open-label, phase 3 HYPRO trial. Int J Radiat Oncol Biol Phys. 2020;106:108-115. doi:10.1016/j.ijrobp.2019.09.007
- Incrocci L, Wortel RC, Alemayehu WG, et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016;17:1061-1069. doi:10.1016/S1470-2045(16)30070-5
- Widmark A, Gunnlaugsson A, Beckman L, et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019;394:385-395. doi:10.1016/S0140-6736(19)31131-6
- Brenner DJ, Hall EJ. Fractionation and protraction for radiotherapy of prostate carcinoma. Int J Radiat Oncol Biol Phys. 1999;43:1095-101. doi:10.1016/s0360-3016(98)00438-6
- Dasu A. Is the alpha/beta value for prostate tumours low enough to be safely used in clinical trials? Clin Oncol (R Coll Radiol). 2007;19:289-301. doi:10.1016/j.clon.2007.02.007
- Garcia-Barros M, Paris F, Cordon-Cardo C, et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science. 2003;300:1155-1159. doi:10.1126/science.1082504
- Gulliford S, Hall E, Dearnaley D. Hypofractionation trials and radiobiology of prostate cancer. Oncoscience. 2017;4:27-28. doi:10.18632/oncoscience.347
- Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8:89-91. doi:10.1016/j.ccr.2005.07.014
- van As N, Griffin C, Tree A, et al. Phase 3 Trial of stereotactic body radiotherapy in localized prostate cancer. N Engl J Med. Oct 17 2024;391:1413-1425. doi:10.1056/NEJMoa2403365
- National Comprehensive Cancer Network. NCCN Guidelines Version 5. 2026 Prostate Cancer. Accessed March 24, 2026. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- Lawton CA, Michalski J, El-Naqa I, et al. RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2009;74:383-387. doi:10.1016/j.ijrobp.2008.08.002
- Lehrer EJ, Kishan AU, Yu JB, et al. Ultrahypofractionated versus hypofractionated and conventionally fractionated radiation therapy for localized prostate cancer: a systematic review and meta-analysis of phase III randomized trials. Radiother Oncol. 2020;148:235-242. doi:10.1016/j.radonc.2020.04.037
- De Cooman B, Debacker T, Adams T, et al. Stereotactic body radiotherapy (SBRT) as a treatment for localized prostate cancer: a retrospective analysis. Radiat Oncol. 2025;20:25. doi:10.1186/s13014-025-02598-8
- Fuller DB, Falchook AD, Crabtree T, et al. Phase 2 multicenter trial of heterogeneous-dosing stereotactic body radiotherapy for low- and intermediate-risk prostate cancer: 5-year outcomes. Eur Urol Oncol. 2018;1:540-547. doi:10.1016/j.euo.2018.06.013
- Jackson WC, Silva J, Hartman HE, et al. Stereotactic body radiation therapy for localized prostate cancer: a systematic review and meta-analysis of over 6,000 patients treated on prospective studies. Int J Radiat Oncol Biol Phys. 2019;104:778-789. doi:10.1016/j.ijrobp.2019.03.051
- Meier RM, Bloch DA, Cotrutz C, et al. Multicenter trial of stereotactic body radiation therapy for low- and intermediate-risk prostate cancer: survival and toxicity endpoints. nt J Radiat Oncol Biol Phys. 2018;102:296-303. doi:10.1016/j.ijrobp.2018.05.040
- Quon HC, Ong A, Cheung P, et al. Once-weekly versus every-other-day stereotactic body radiotherapy in patients with prostate cancer (PATRIOT): a phase 2 randomized trial. Radiother Oncol. 2018;127:206-212. doi:10.1016/j.radonc.2018.02.029
- Zelefsky MJ, Kollmeier M, McBride S, et al. Five-year outcomes of a phase 1 dose-escalation study using stereotactic body radiosurgery for patients with low-risk and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2019;104:42-49. doi:10.1016/j.ijrobp.2018.12.045
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Early Outcomes of Stereotactic Body Radiotherapy for Localized Prostate Cancer: A Retrospective Analysis
Vet Prostate Cancer Survivors Face Hidden Breast Cancer Risk
Vet Prostate Cancer Survivors Face Hidden Breast Cancer Risk
TOPLINE:
Among 1.3 million male veterans treated for prostate cancer, 11,327 (0.86%) developed breast cancer an average of 5.4 years after initial diagnosis. Younger age at prostate cancer diagnosis, metastatic disease, androgen deprivation therapy (ADT), radiation treatment, and prolonged use of certain cardiovascular disease (CVD) medications were associated with increased risk for breast cancer.
METHODOLOGY:
- Researchers used a retrospective cohort design in Veterans Health Administration (VHA) care, pulling data from the Veterans Affairs (VA) Prostate Cancer Data Core at the VA Corporate Data Warehouse.
- Participants included 1,314,492 male veterans with prostate cancer treated at VHA facilities from January 1, 2000, to March 12, 2024.
- Exposure definitions included prostate cancer treatments (ADT, anti-androgen treatment, radiation-brachytherapy, and platinum chemotherapy) and CVD medications (furosemide, spironolactone, digoxin) captured via inpatient/outpatient/fee-based pharmacy and Current Procedural Terminology codes.
- Analysis measured time from prostate cancer diagnosis to breast cancer diagnosis, death, or March 12, 2024, applying Cox proportional hazards and Fine-Gray competing risk methods, with a sensitivity analysis adding body mass index (BMI) after excluding 71,718 missing values.
TAKEAWAY:
- Metastatic prostate cancer at diagnosis more than doubled the risk for breast cancer compared to nonmetastatic disease (hazard ratio [HR], 2.03; 95% CI, 1.90-2.17; P < .0001; subdistribution hazard ratio [SHR], 1.68; 95% CI, 1.57-1.81; P < .0001).
- Younger age at prostate cancer diagnosis was associated with increased risk for breast cancer (HR, 0.97; 95% CI, 0.97-0.98; P < .0001; SHR, 0.957; 95% CI, 0.955-0.959; P < .0001), indicating that for each additional year of age at diagnosis, the risk decreased.
- Continuation of CVD medications after prostate cancer diagnosis was associated with increased risk for breast cancer: furosemide (HR, 1.51; 95% CI, 1.39-1.63; P < .0001; SHR, 1.21; 95% CI, 1.12-1.31; P < .0001), spironolactone (HR, 1.36; 95% CI, 1.15-1.61; P = .0004; SHR, 1.23; 95% CI, 1.04-1.47; P = .0174), and digoxin (HR, 1.49; 95% CI, 1.29-1.72; P < .0001; SHR, 1.26; 95% CI, 1.10-1.46; P = .0015).
- Radiation therapy and ADT were associated with increased risk for breast cancer (radiation: HR, 1.06; 95% CI, 1.02-1.11; P = .0088; SHR, 1.10; 95% CI, 1.05-1.15; P < .0001; ADT: HR, 1.24; 95% CI, 1.17-1.32; P < .0001; SHR, 1.28; 95% CI, 1.20-1.37; P < .0001), while abiraterone was associated with decreased risk (HR, 0.36; 95% CI, 0.31-0.42; P < .0001; SHR, 0.39; 95% CI, 0.34-0.45; P < .0001).
IN PRACTICE:
"While there is a lack of data, male veterans with previous prostate cancer are at an elevated risk of breast cancer (0.87%), than their civilian counterparts (0.14%),” the authors wrote. “To address the current gap in knowledge and data, this study leveraged an existing large cohort of male veterans with prostate cancer and examined factors associated with increased risk of male breast cancer."
SOURCE:
The study was led by Erum Z. Whyne, VA North Texas Health Care System in Dallas, and Haekyung Jeon-Slaughter, University of Texas Southwestern Medical Center in Dallas. It was published online in The Prostate.
LIMITATIONS:
Though the study findings are based on large, representative data from male veterans with previously diagnosed prostate cancer, the results might not be generalizable to the overall male breast cancer population. As a retrospective cohort study, results may be biased and causality is difficult to establish. The study did not examine other known risk factors for male breast cancer incidence, such as family history, BRCA2 mutations, and military environmental exposure due to lack of data. BMI had missingness of 5.46% (n = 71,718) and was not included as a covariate in the final model, though sensitivity analysis showed it was not significantly associated with increased risk for male breast cancer.
DISCLOSURES:
The research was supported using resources and facilities of the VA Informatics and Computing Infrastructure (VINCI), VA HSR RES 13-457. The VA North Texas Health Care System Institutional Review Board approved the study and waived informed consent. No conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
Among 1.3 million male veterans treated for prostate cancer, 11,327 (0.86%) developed breast cancer an average of 5.4 years after initial diagnosis. Younger age at prostate cancer diagnosis, metastatic disease, androgen deprivation therapy (ADT), radiation treatment, and prolonged use of certain cardiovascular disease (CVD) medications were associated with increased risk for breast cancer.
METHODOLOGY:
- Researchers used a retrospective cohort design in Veterans Health Administration (VHA) care, pulling data from the Veterans Affairs (VA) Prostate Cancer Data Core at the VA Corporate Data Warehouse.
- Participants included 1,314,492 male veterans with prostate cancer treated at VHA facilities from January 1, 2000, to March 12, 2024.
- Exposure definitions included prostate cancer treatments (ADT, anti-androgen treatment, radiation-brachytherapy, and platinum chemotherapy) and CVD medications (furosemide, spironolactone, digoxin) captured via inpatient/outpatient/fee-based pharmacy and Current Procedural Terminology codes.
- Analysis measured time from prostate cancer diagnosis to breast cancer diagnosis, death, or March 12, 2024, applying Cox proportional hazards and Fine-Gray competing risk methods, with a sensitivity analysis adding body mass index (BMI) after excluding 71,718 missing values.
TAKEAWAY:
- Metastatic prostate cancer at diagnosis more than doubled the risk for breast cancer compared to nonmetastatic disease (hazard ratio [HR], 2.03; 95% CI, 1.90-2.17; P < .0001; subdistribution hazard ratio [SHR], 1.68; 95% CI, 1.57-1.81; P < .0001).
- Younger age at prostate cancer diagnosis was associated with increased risk for breast cancer (HR, 0.97; 95% CI, 0.97-0.98; P < .0001; SHR, 0.957; 95% CI, 0.955-0.959; P < .0001), indicating that for each additional year of age at diagnosis, the risk decreased.
- Continuation of CVD medications after prostate cancer diagnosis was associated with increased risk for breast cancer: furosemide (HR, 1.51; 95% CI, 1.39-1.63; P < .0001; SHR, 1.21; 95% CI, 1.12-1.31; P < .0001), spironolactone (HR, 1.36; 95% CI, 1.15-1.61; P = .0004; SHR, 1.23; 95% CI, 1.04-1.47; P = .0174), and digoxin (HR, 1.49; 95% CI, 1.29-1.72; P < .0001; SHR, 1.26; 95% CI, 1.10-1.46; P = .0015).
- Radiation therapy and ADT were associated with increased risk for breast cancer (radiation: HR, 1.06; 95% CI, 1.02-1.11; P = .0088; SHR, 1.10; 95% CI, 1.05-1.15; P < .0001; ADT: HR, 1.24; 95% CI, 1.17-1.32; P < .0001; SHR, 1.28; 95% CI, 1.20-1.37; P < .0001), while abiraterone was associated with decreased risk (HR, 0.36; 95% CI, 0.31-0.42; P < .0001; SHR, 0.39; 95% CI, 0.34-0.45; P < .0001).
IN PRACTICE:
"While there is a lack of data, male veterans with previous prostate cancer are at an elevated risk of breast cancer (0.87%), than their civilian counterparts (0.14%),” the authors wrote. “To address the current gap in knowledge and data, this study leveraged an existing large cohort of male veterans with prostate cancer and examined factors associated with increased risk of male breast cancer."
SOURCE:
The study was led by Erum Z. Whyne, VA North Texas Health Care System in Dallas, and Haekyung Jeon-Slaughter, University of Texas Southwestern Medical Center in Dallas. It was published online in The Prostate.
LIMITATIONS:
Though the study findings are based on large, representative data from male veterans with previously diagnosed prostate cancer, the results might not be generalizable to the overall male breast cancer population. As a retrospective cohort study, results may be biased and causality is difficult to establish. The study did not examine other known risk factors for male breast cancer incidence, such as family history, BRCA2 mutations, and military environmental exposure due to lack of data. BMI had missingness of 5.46% (n = 71,718) and was not included as a covariate in the final model, though sensitivity analysis showed it was not significantly associated with increased risk for male breast cancer.
DISCLOSURES:
The research was supported using resources and facilities of the VA Informatics and Computing Infrastructure (VINCI), VA HSR RES 13-457. The VA North Texas Health Care System Institutional Review Board approved the study and waived informed consent. No conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
Among 1.3 million male veterans treated for prostate cancer, 11,327 (0.86%) developed breast cancer an average of 5.4 years after initial diagnosis. Younger age at prostate cancer diagnosis, metastatic disease, androgen deprivation therapy (ADT), radiation treatment, and prolonged use of certain cardiovascular disease (CVD) medications were associated with increased risk for breast cancer.
METHODOLOGY:
- Researchers used a retrospective cohort design in Veterans Health Administration (VHA) care, pulling data from the Veterans Affairs (VA) Prostate Cancer Data Core at the VA Corporate Data Warehouse.
- Participants included 1,314,492 male veterans with prostate cancer treated at VHA facilities from January 1, 2000, to March 12, 2024.
- Exposure definitions included prostate cancer treatments (ADT, anti-androgen treatment, radiation-brachytherapy, and platinum chemotherapy) and CVD medications (furosemide, spironolactone, digoxin) captured via inpatient/outpatient/fee-based pharmacy and Current Procedural Terminology codes.
- Analysis measured time from prostate cancer diagnosis to breast cancer diagnosis, death, or March 12, 2024, applying Cox proportional hazards and Fine-Gray competing risk methods, with a sensitivity analysis adding body mass index (BMI) after excluding 71,718 missing values.
TAKEAWAY:
- Metastatic prostate cancer at diagnosis more than doubled the risk for breast cancer compared to nonmetastatic disease (hazard ratio [HR], 2.03; 95% CI, 1.90-2.17; P < .0001; subdistribution hazard ratio [SHR], 1.68; 95% CI, 1.57-1.81; P < .0001).
- Younger age at prostate cancer diagnosis was associated with increased risk for breast cancer (HR, 0.97; 95% CI, 0.97-0.98; P < .0001; SHR, 0.957; 95% CI, 0.955-0.959; P < .0001), indicating that for each additional year of age at diagnosis, the risk decreased.
- Continuation of CVD medications after prostate cancer diagnosis was associated with increased risk for breast cancer: furosemide (HR, 1.51; 95% CI, 1.39-1.63; P < .0001; SHR, 1.21; 95% CI, 1.12-1.31; P < .0001), spironolactone (HR, 1.36; 95% CI, 1.15-1.61; P = .0004; SHR, 1.23; 95% CI, 1.04-1.47; P = .0174), and digoxin (HR, 1.49; 95% CI, 1.29-1.72; P < .0001; SHR, 1.26; 95% CI, 1.10-1.46; P = .0015).
- Radiation therapy and ADT were associated with increased risk for breast cancer (radiation: HR, 1.06; 95% CI, 1.02-1.11; P = .0088; SHR, 1.10; 95% CI, 1.05-1.15; P < .0001; ADT: HR, 1.24; 95% CI, 1.17-1.32; P < .0001; SHR, 1.28; 95% CI, 1.20-1.37; P < .0001), while abiraterone was associated with decreased risk (HR, 0.36; 95% CI, 0.31-0.42; P < .0001; SHR, 0.39; 95% CI, 0.34-0.45; P < .0001).
IN PRACTICE:
"While there is a lack of data, male veterans with previous prostate cancer are at an elevated risk of breast cancer (0.87%), than their civilian counterparts (0.14%),” the authors wrote. “To address the current gap in knowledge and data, this study leveraged an existing large cohort of male veterans with prostate cancer and examined factors associated with increased risk of male breast cancer."
SOURCE:
The study was led by Erum Z. Whyne, VA North Texas Health Care System in Dallas, and Haekyung Jeon-Slaughter, University of Texas Southwestern Medical Center in Dallas. It was published online in The Prostate.
LIMITATIONS:
Though the study findings are based on large, representative data from male veterans with previously diagnosed prostate cancer, the results might not be generalizable to the overall male breast cancer population. As a retrospective cohort study, results may be biased and causality is difficult to establish. The study did not examine other known risk factors for male breast cancer incidence, such as family history, BRCA2 mutations, and military environmental exposure due to lack of data. BMI had missingness of 5.46% (n = 71,718) and was not included as a covariate in the final model, though sensitivity analysis showed it was not significantly associated with increased risk for male breast cancer.
DISCLOSURES:
The research was supported using resources and facilities of the VA Informatics and Computing Infrastructure (VINCI), VA HSR RES 13-457. The VA North Texas Health Care System Institutional Review Board approved the study and waived informed consent. No conflicts of interest were disclosed by the authors.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Vet Prostate Cancer Survivors Face Hidden Breast Cancer Risk
Vet Prostate Cancer Survivors Face Hidden Breast Cancer Risk
Remote Program Doubles Metastatic Prostate Cancer Germline Testing
A pilot program appeared to more than double the rate of germline genetic testing among veterans with metastatic prostate cancer (mPC) by using remote communication rather than relying on clinicians for in-person outreach to patients.
Of 1952 veterans with mPC, 681 (34.9%) provided consent and 459 (23.5%) completed testing, exceeding the usual 10% to 12% of patients who undergo testing, reported Bruce Montgomery, MD, et al in Cancer.
Although testing is recommended for all patients with mPC to guide therapy and alert relatives who may be at risk, 23.5% is still an impressive number, Montgomery, an oncologist with Veterans Affairs (VA) Puget Sound Health Care System in Seattle told Federal Practitioner: “With a letter and very little money and very little real time from clinicians, we could get testing done at 3 times the rate happening out there in the big wide world,” he said. “For 2000 patients, we needed one research coordinator and a small part of a genetic counselor's time.”
According to the study, germline genetic testing—which examines inherited DNA—is now recommended for all men with mPC by the National Comprehensive Cancer Network, the American Society of Clinical Oncology, and the American Urological Association. Germline genetic testing differs from somatic testing, which seeks genetic changes in the tumors themselves.
In the VA and community at large, the percentage of men with mPC who undergo germline genetic testing is low, Montgomery said. Research suggests < 40% of patients undergo somatic testing.
Germline genetic testing only costs about 10% compared with somatic testing, Montgomery said, and can be conducted at any time. In about 10% of mPC cases, the testing provides insight into the best treatment, he said.
Montgomery noted another benefit to germline genetic testing: It can raise the alarm about pathogenic variants that could boost cancer risk in family members, allowing them to get screened and take action.
There are many reasons veterans do not get tested, Montgomery said. The process is not automatic because patient consent is needed, and clinicians often fail to ask. In some cases, veterans worry about privacy or whether they will lose service-connected benefits if their cancer is blamed on genetics.
The study focused on 2104 veterans with mPC who had already agreed to take part in the Million Veteran Program, a prospective cohort study examining genetic and nongenetic risk for disease. The genetic analysis from that project did not provide guidance about mPC, so researchers approached the veterans directly.
Patients were enrolled from February 2021 to October 2023. A total of 1952 veterans did not opt out when contacted by mail (median age, 75 years; 63% White, 25% Black; 74% urban and 24% rural). The median age of those who consented and completed testing after phone contact was 74 years; 67% of patients were White and 22% were Black; 78% of patients lived in urban communities and 20% lived in rural communities.
Fifty-nine patients (13%) had pathogenic variants, and 37 of those had variants that indicated treatment with targeted therapies. Of the 37, 14 received targeted therapy, 18 were not at the point where targeted therapy was indicated, and 5 were not treated with targeted therapy for various reasons before they died.
Twelve of the 59 patients with pathogenic variants agreed to let the study team contact their first-degree relatives. Thirty relatives underwent testing, and 10 of them were positive for the variants.
Following completion of the study, researchers examined electronic records for the 59 patients with pathogenic variants and found that 19% did not have documentation of the germline finding in the medical record. The authors cited an “urgent need” to standardize where genetic information is included in the records.
While “it seems like a very small number of patients took up testing,” Montgomery said, the study findings are promising: “If we did the same thing nationally in the VA, there would be 15,000 men with metastatic disease, and we’d be testing 5000 of them with almost no effort.”
In an interview, Susan Vadaparampil, PhD, MPH, associate center director of Community Outreach and Engagement at Moffitt Cancer Center, who studies genetic testing, praised the strengths of the study. Vadaparampil, who did not take part in the research, told Federal Practitioner that the study relies on “an intervention that could likely be incorporated into routine clinical practice, a less resource-intensive model that provides posttest counseling for those who test positive, and support to share results with family members.”
However, she said, “testing uptake was uneven based on participant sociodemographic characteristics. It's important to consider how discussions and resources to facilitate testing may need to be adapted to meet the needs of all patients.
“Strategies that facilitate clinicians’ knowledge, comfort, and consistency in discussing testing with all mPC patients are essential,” Vadaparampil added. “Simultaneously using multiple strategies targeted to different levels can further help boost uptake.”
The study was funded by the VA Office of Research and Development, Prostate Cancer Foundation, Pacific Northwest Prostate Cancer SPORE, Institute for Prostate Cancer Research, Congressionally Directed Medical Research Programs (CDMRP), and Put VA Data to Work for Veterans.
Montgomery discloses relationships with Daiichi Sankyo, INmune Bio, Clovis, Janssen Pharmaceuticals, Johnson and Johnson, and Merck. Some other authors report various disclosures. Vadaparampil has no disclosures.
A pilot program appeared to more than double the rate of germline genetic testing among veterans with metastatic prostate cancer (mPC) by using remote communication rather than relying on clinicians for in-person outreach to patients.
Of 1952 veterans with mPC, 681 (34.9%) provided consent and 459 (23.5%) completed testing, exceeding the usual 10% to 12% of patients who undergo testing, reported Bruce Montgomery, MD, et al in Cancer.
Although testing is recommended for all patients with mPC to guide therapy and alert relatives who may be at risk, 23.5% is still an impressive number, Montgomery, an oncologist with Veterans Affairs (VA) Puget Sound Health Care System in Seattle told Federal Practitioner: “With a letter and very little money and very little real time from clinicians, we could get testing done at 3 times the rate happening out there in the big wide world,” he said. “For 2000 patients, we needed one research coordinator and a small part of a genetic counselor's time.”
According to the study, germline genetic testing—which examines inherited DNA—is now recommended for all men with mPC by the National Comprehensive Cancer Network, the American Society of Clinical Oncology, and the American Urological Association. Germline genetic testing differs from somatic testing, which seeks genetic changes in the tumors themselves.
In the VA and community at large, the percentage of men with mPC who undergo germline genetic testing is low, Montgomery said. Research suggests < 40% of patients undergo somatic testing.
Germline genetic testing only costs about 10% compared with somatic testing, Montgomery said, and can be conducted at any time. In about 10% of mPC cases, the testing provides insight into the best treatment, he said.
Montgomery noted another benefit to germline genetic testing: It can raise the alarm about pathogenic variants that could boost cancer risk in family members, allowing them to get screened and take action.
There are many reasons veterans do not get tested, Montgomery said. The process is not automatic because patient consent is needed, and clinicians often fail to ask. In some cases, veterans worry about privacy or whether they will lose service-connected benefits if their cancer is blamed on genetics.
The study focused on 2104 veterans with mPC who had already agreed to take part in the Million Veteran Program, a prospective cohort study examining genetic and nongenetic risk for disease. The genetic analysis from that project did not provide guidance about mPC, so researchers approached the veterans directly.
Patients were enrolled from February 2021 to October 2023. A total of 1952 veterans did not opt out when contacted by mail (median age, 75 years; 63% White, 25% Black; 74% urban and 24% rural). The median age of those who consented and completed testing after phone contact was 74 years; 67% of patients were White and 22% were Black; 78% of patients lived in urban communities and 20% lived in rural communities.
Fifty-nine patients (13%) had pathogenic variants, and 37 of those had variants that indicated treatment with targeted therapies. Of the 37, 14 received targeted therapy, 18 were not at the point where targeted therapy was indicated, and 5 were not treated with targeted therapy for various reasons before they died.
Twelve of the 59 patients with pathogenic variants agreed to let the study team contact their first-degree relatives. Thirty relatives underwent testing, and 10 of them were positive for the variants.
Following completion of the study, researchers examined electronic records for the 59 patients with pathogenic variants and found that 19% did not have documentation of the germline finding in the medical record. The authors cited an “urgent need” to standardize where genetic information is included in the records.
While “it seems like a very small number of patients took up testing,” Montgomery said, the study findings are promising: “If we did the same thing nationally in the VA, there would be 15,000 men with metastatic disease, and we’d be testing 5000 of them with almost no effort.”
In an interview, Susan Vadaparampil, PhD, MPH, associate center director of Community Outreach and Engagement at Moffitt Cancer Center, who studies genetic testing, praised the strengths of the study. Vadaparampil, who did not take part in the research, told Federal Practitioner that the study relies on “an intervention that could likely be incorporated into routine clinical practice, a less resource-intensive model that provides posttest counseling for those who test positive, and support to share results with family members.”
However, she said, “testing uptake was uneven based on participant sociodemographic characteristics. It's important to consider how discussions and resources to facilitate testing may need to be adapted to meet the needs of all patients.
“Strategies that facilitate clinicians’ knowledge, comfort, and consistency in discussing testing with all mPC patients are essential,” Vadaparampil added. “Simultaneously using multiple strategies targeted to different levels can further help boost uptake.”
The study was funded by the VA Office of Research and Development, Prostate Cancer Foundation, Pacific Northwest Prostate Cancer SPORE, Institute for Prostate Cancer Research, Congressionally Directed Medical Research Programs (CDMRP), and Put VA Data to Work for Veterans.
Montgomery discloses relationships with Daiichi Sankyo, INmune Bio, Clovis, Janssen Pharmaceuticals, Johnson and Johnson, and Merck. Some other authors report various disclosures. Vadaparampil has no disclosures.
A pilot program appeared to more than double the rate of germline genetic testing among veterans with metastatic prostate cancer (mPC) by using remote communication rather than relying on clinicians for in-person outreach to patients.
Of 1952 veterans with mPC, 681 (34.9%) provided consent and 459 (23.5%) completed testing, exceeding the usual 10% to 12% of patients who undergo testing, reported Bruce Montgomery, MD, et al in Cancer.
Although testing is recommended for all patients with mPC to guide therapy and alert relatives who may be at risk, 23.5% is still an impressive number, Montgomery, an oncologist with Veterans Affairs (VA) Puget Sound Health Care System in Seattle told Federal Practitioner: “With a letter and very little money and very little real time from clinicians, we could get testing done at 3 times the rate happening out there in the big wide world,” he said. “For 2000 patients, we needed one research coordinator and a small part of a genetic counselor's time.”
According to the study, germline genetic testing—which examines inherited DNA—is now recommended for all men with mPC by the National Comprehensive Cancer Network, the American Society of Clinical Oncology, and the American Urological Association. Germline genetic testing differs from somatic testing, which seeks genetic changes in the tumors themselves.
In the VA and community at large, the percentage of men with mPC who undergo germline genetic testing is low, Montgomery said. Research suggests < 40% of patients undergo somatic testing.
Germline genetic testing only costs about 10% compared with somatic testing, Montgomery said, and can be conducted at any time. In about 10% of mPC cases, the testing provides insight into the best treatment, he said.
Montgomery noted another benefit to germline genetic testing: It can raise the alarm about pathogenic variants that could boost cancer risk in family members, allowing them to get screened and take action.
There are many reasons veterans do not get tested, Montgomery said. The process is not automatic because patient consent is needed, and clinicians often fail to ask. In some cases, veterans worry about privacy or whether they will lose service-connected benefits if their cancer is blamed on genetics.
The study focused on 2104 veterans with mPC who had already agreed to take part in the Million Veteran Program, a prospective cohort study examining genetic and nongenetic risk for disease. The genetic analysis from that project did not provide guidance about mPC, so researchers approached the veterans directly.
Patients were enrolled from February 2021 to October 2023. A total of 1952 veterans did not opt out when contacted by mail (median age, 75 years; 63% White, 25% Black; 74% urban and 24% rural). The median age of those who consented and completed testing after phone contact was 74 years; 67% of patients were White and 22% were Black; 78% of patients lived in urban communities and 20% lived in rural communities.
Fifty-nine patients (13%) had pathogenic variants, and 37 of those had variants that indicated treatment with targeted therapies. Of the 37, 14 received targeted therapy, 18 were not at the point where targeted therapy was indicated, and 5 were not treated with targeted therapy for various reasons before they died.
Twelve of the 59 patients with pathogenic variants agreed to let the study team contact their first-degree relatives. Thirty relatives underwent testing, and 10 of them were positive for the variants.
Following completion of the study, researchers examined electronic records for the 59 patients with pathogenic variants and found that 19% did not have documentation of the germline finding in the medical record. The authors cited an “urgent need” to standardize where genetic information is included in the records.
While “it seems like a very small number of patients took up testing,” Montgomery said, the study findings are promising: “If we did the same thing nationally in the VA, there would be 15,000 men with metastatic disease, and we’d be testing 5000 of them with almost no effort.”
In an interview, Susan Vadaparampil, PhD, MPH, associate center director of Community Outreach and Engagement at Moffitt Cancer Center, who studies genetic testing, praised the strengths of the study. Vadaparampil, who did not take part in the research, told Federal Practitioner that the study relies on “an intervention that could likely be incorporated into routine clinical practice, a less resource-intensive model that provides posttest counseling for those who test positive, and support to share results with family members.”
However, she said, “testing uptake was uneven based on participant sociodemographic characteristics. It's important to consider how discussions and resources to facilitate testing may need to be adapted to meet the needs of all patients.
“Strategies that facilitate clinicians’ knowledge, comfort, and consistency in discussing testing with all mPC patients are essential,” Vadaparampil added. “Simultaneously using multiple strategies targeted to different levels can further help boost uptake.”
The study was funded by the VA Office of Research and Development, Prostate Cancer Foundation, Pacific Northwest Prostate Cancer SPORE, Institute for Prostate Cancer Research, Congressionally Directed Medical Research Programs (CDMRP), and Put VA Data to Work for Veterans.
Montgomery discloses relationships with Daiichi Sankyo, INmune Bio, Clovis, Janssen Pharmaceuticals, Johnson and Johnson, and Merck. Some other authors report various disclosures. Vadaparampil has no disclosures.
FDA OKs Blood-Based Test to Help Diagnose Prostate Cancer
FDA OKs Blood-Based Test to Help Diagnose Prostate Cancer
The FDA has granted Cleveland Diagnostics' IsoPSA test premarket approval (PMA) to help detect prostate cancer in men aged ≥ 50 years with elevated PSA levels.
IsoPSA is a blood assay that detects variations of the PSA protein that signal a higher likelihood of high-grade tumors. It is one of several biomarker tests included in the National Comprehensive Cancer Network's guidelines on early detection of prostate cancer.
Cleveland Diagnostics noted that 75% of prostate biopsies are negative for high-grade disease. IsoPSA and similar tests aim to help identify men who need a biopsy while allowing others avoid an unnecessary procedure.
IsoPSA has been available since 2020 under the FDA's Laboratory-Developed Test rubric, meaning that blood samples had to be shipped for analysis to Cleveland Diagnostics' lab. With the PMA, testing can now be done at CLIA-certified labs across the country.
The company expects the approval should increase access to IsoPSA and reduce turnaround time. "We remain focused on executing our commercial strategy and expanding access to IsoPSA," company President and CEO Arnon Chait, PhD, said in a press release.
The approval was based, in part, on a prospective validation study of 888 men scheduled for prostate biopsy. IsoPSA demonstrated an AUC of 0.783 for high-grade tumors, with a sensitivity of 90.2% and a specificity of 45.5%. In a real-world clinical utility study with 900 patients, IsoPSA testing led to a 55% decrease in biopsy recommendations.
The test is covered by Medicare and a growing number of commercial payers, Cleveland Diagnostics said.
M. Alexander Otto is a physician assistant with a master's degree in medical science and a journalism degree from Newhouse. He is an award-winning medical journalist who worked for several major news outlets before joining Medscape Medical News. Alex is also an MIT Knight Science Journalism Fellow. Email: [email protected].
A version of this article first appeared on Medscape.com.
The FDA has granted Cleveland Diagnostics' IsoPSA test premarket approval (PMA) to help detect prostate cancer in men aged ≥ 50 years with elevated PSA levels.
IsoPSA is a blood assay that detects variations of the PSA protein that signal a higher likelihood of high-grade tumors. It is one of several biomarker tests included in the National Comprehensive Cancer Network's guidelines on early detection of prostate cancer.
Cleveland Diagnostics noted that 75% of prostate biopsies are negative for high-grade disease. IsoPSA and similar tests aim to help identify men who need a biopsy while allowing others avoid an unnecessary procedure.
IsoPSA has been available since 2020 under the FDA's Laboratory-Developed Test rubric, meaning that blood samples had to be shipped for analysis to Cleveland Diagnostics' lab. With the PMA, testing can now be done at CLIA-certified labs across the country.
The company expects the approval should increase access to IsoPSA and reduce turnaround time. "We remain focused on executing our commercial strategy and expanding access to IsoPSA," company President and CEO Arnon Chait, PhD, said in a press release.
The approval was based, in part, on a prospective validation study of 888 men scheduled for prostate biopsy. IsoPSA demonstrated an AUC of 0.783 for high-grade tumors, with a sensitivity of 90.2% and a specificity of 45.5%. In a real-world clinical utility study with 900 patients, IsoPSA testing led to a 55% decrease in biopsy recommendations.
The test is covered by Medicare and a growing number of commercial payers, Cleveland Diagnostics said.
M. Alexander Otto is a physician assistant with a master's degree in medical science and a journalism degree from Newhouse. He is an award-winning medical journalist who worked for several major news outlets before joining Medscape Medical News. Alex is also an MIT Knight Science Journalism Fellow. Email: [email protected].
A version of this article first appeared on Medscape.com.
The FDA has granted Cleveland Diagnostics' IsoPSA test premarket approval (PMA) to help detect prostate cancer in men aged ≥ 50 years with elevated PSA levels.
IsoPSA is a blood assay that detects variations of the PSA protein that signal a higher likelihood of high-grade tumors. It is one of several biomarker tests included in the National Comprehensive Cancer Network's guidelines on early detection of prostate cancer.
Cleveland Diagnostics noted that 75% of prostate biopsies are negative for high-grade disease. IsoPSA and similar tests aim to help identify men who need a biopsy while allowing others avoid an unnecessary procedure.
IsoPSA has been available since 2020 under the FDA's Laboratory-Developed Test rubric, meaning that blood samples had to be shipped for analysis to Cleveland Diagnostics' lab. With the PMA, testing can now be done at CLIA-certified labs across the country.
The company expects the approval should increase access to IsoPSA and reduce turnaround time. "We remain focused on executing our commercial strategy and expanding access to IsoPSA," company President and CEO Arnon Chait, PhD, said in a press release.
The approval was based, in part, on a prospective validation study of 888 men scheduled for prostate biopsy. IsoPSA demonstrated an AUC of 0.783 for high-grade tumors, with a sensitivity of 90.2% and a specificity of 45.5%. In a real-world clinical utility study with 900 patients, IsoPSA testing led to a 55% decrease in biopsy recommendations.
The test is covered by Medicare and a growing number of commercial payers, Cleveland Diagnostics said.
M. Alexander Otto is a physician assistant with a master's degree in medical science and a journalism degree from Newhouse. He is an award-winning medical journalist who worked for several major news outlets before joining Medscape Medical News. Alex is also an MIT Knight Science Journalism Fellow. Email: [email protected].
A version of this article first appeared on Medscape.com.
FDA OKs Blood-Based Test to Help Diagnose Prostate Cancer
FDA OKs Blood-Based Test to Help Diagnose Prostate Cancer
Identical Survival for Abiraterone and Enzalutamide in Vets With Metastatic Hormone-Sensitive Prostate Cancer
Abiraterone and enzalutamide showed identical survival outcomes when used as first-line treatment for metastatic hormone-sensitive prostate cancer (mHSPC), according to a new study using US Department of Veterans Affairs (VA) data. The report represents the first head-to-head clinical analysis of these commonly used androgen receptor inhibitors.
Among 1258 veterans treated with abiraterone and 311 treated with enzalutamide, median overall survival was 36.2 months for both drugs. Patients were followed for a mean of 28.7 months (abiraterone) and 30.8 months (enzalutamide), reported by Martin W. Schoen, MD, MPH, from Saint Louis University School of Medicine and the St. Louis VA Medical Center, in JAMA Network Open.
Notably, there was no significant difference in outcomes among Black veterans, who often have poorer outcomes in prostate cancer, and in patients with cardiovascular disease.
“This is the first direct comparison of abiraterone and enzalutamide for mHSPC in a clinical practice setting,” Schoen told Federal Practitioner. “At the population level, there are no differences based on initial treatment choice.”
Abiraterone Is Preferred in the VA Due to Cost
According to Schoen, abiraterone and enzalutamide are the most commonly used androgen receptor inhibitors to treat mHSPC within the VA. A 2025 study by Schoen and colleagues found that 53.7% of veterans with mHSPC in 2022 received androgen receptor inhibitor therapy, up from 16.9% in 2017.
“In the VA, the preference for most patients is abiraterone since it is the least expensive agent,” he said. A generic version has been available for several years.
Additionally, abiraterone “has been on the market for the longest, and therefore clinicians are familiar with its use,” Schoen said. However, “clinicians have little idea of the comparative efficacy between these 2 agents,” he added.
The authors suggest that the cost and toxicities of the medications should guide clinician decisions, Schoen said. “There is data that abiraterone may worsen diabetes, since it is given with prednisone and could increase the risk of cardiovascular events,” he said.
He added that 2 newer drugs, apalutamide and darolutamide, are also “viable options.” Chemotherapies and certain targeted drugs are also available, “but they are only used in a select group of patients.”
Outside Specialist: Diverse Study Population Is a Plus
Hematologist-oncologist Natalie Reizine, MD, of the University of Illinois College of Medicine, Chicago, who was not involved in the study, told Federal Practitioner that the real-world data are valuable given the limitations of clinical trial populations.
“It’s difficult to compare clinical trials because they enroll different groups of patients,” she said. And, she said, they often exclude patients with significant comorbidities. “If they have bad cardiovascular disease, for instance, or poorly controlled diabetes, they're excluded from the clinical trial. But in real life, many of our patients have other medical problems that we have to manage.”
Reizine also emphasized the significance of the study’s diverse patient population. “Black men are very underrepresented in clinical trials. Many clinical trials that lead to drug approval will have only few or no Black men at all, yet these drugs go on to be widely prescribed to all men with prostate cancer.”
Results Are ‘Reassuring’
Reizine described the overall study findings as “reassuring,” especially in light of “studies that show that abiraterone and prednisone may be associated with worse cardiovascular outcomes. This study showed that in this VA population, even for patients who had cardiovascular disease, there was not a difference in how they did.”
As for choosing between agents, she recommended considering comorbidities and potential drug-drug interactions. “One of the big reasons that you may not be able to safely prescribe enzalutamide, for instance, is if a patient is on an anticoagulant, which is incredibly common in cancer patients. Enzalutamide has more drug-drug interactions than abiraterone and prednisone.”
Study Demographics and Findings
The study included all patients with mHSPC who initiated abiraterone or enzalutamide between July 2017 and April 2023.
Median ages were 73 (abiraterone) and 74 years (enzalutamide, P = .29). Racial distribution was similar between groups: abiraterone (68.1% White, 25.0% Black, 6.9% other/unknown) and enzalutamide (66.6% White, 27.0% Black, 6.4% other/unknown; P = .74). Ethnicity was 89.2% non-Hispanic, 4.4% Hispanic, and 6.4% unknown in the abiraterone group vs 88.4% non-Hispanic, 3.5% Hispanic, and 8.0% unknown in the enzalutamide group (P = .50).
The groups had similar rates of the most common comorbidities: diabetes (40.5% vs 46.3%, respectively, P = .07), peripheral vascular disease (40.2% vs 37.6%, respectively, P = .44), and chronic pulmonary disease (37.0% vs 40.5%, P = .29).
In an inverse probability weighting analysis with abiraterone as reference, weighted median overall survival was comparable across the entire cohort (36.2 months, P = .32), Black veterans (39.7 months, P = .90), and those with cardiovascular disease (31.5 months, P = .30).
The authors noted limitations such as the observational cohort design and data constraints.
The study was supported by the American Society of Clinical Oncology Conquer Cancer Foundation, the Prostate Cancer Foundation, and the Blavatnik Family Foundation.
Schoen discloses relationships with the Prostate Cancer Foundation, Astellas, and US Department of Defense. Other authors disclose relationships with the American Society of Clinical Oncology, Pfizer, Exelixis, Eli Lilly, Sanofi, Merck, Seagen, Bellicum, and BMS.
Outside the submitted work. Reizine discloses relationships with the US Department of Defense, Sanofi, Exelexis, Janssen, AstraZeneca, EMD Serono, Janssen, Merck, and Tempus.
Abiraterone and enzalutamide showed identical survival outcomes when used as first-line treatment for metastatic hormone-sensitive prostate cancer (mHSPC), according to a new study using US Department of Veterans Affairs (VA) data. The report represents the first head-to-head clinical analysis of these commonly used androgen receptor inhibitors.
Among 1258 veterans treated with abiraterone and 311 treated with enzalutamide, median overall survival was 36.2 months for both drugs. Patients were followed for a mean of 28.7 months (abiraterone) and 30.8 months (enzalutamide), reported by Martin W. Schoen, MD, MPH, from Saint Louis University School of Medicine and the St. Louis VA Medical Center, in JAMA Network Open.
Notably, there was no significant difference in outcomes among Black veterans, who often have poorer outcomes in prostate cancer, and in patients with cardiovascular disease.
“This is the first direct comparison of abiraterone and enzalutamide for mHSPC in a clinical practice setting,” Schoen told Federal Practitioner. “At the population level, there are no differences based on initial treatment choice.”
Abiraterone Is Preferred in the VA Due to Cost
According to Schoen, abiraterone and enzalutamide are the most commonly used androgen receptor inhibitors to treat mHSPC within the VA. A 2025 study by Schoen and colleagues found that 53.7% of veterans with mHSPC in 2022 received androgen receptor inhibitor therapy, up from 16.9% in 2017.
“In the VA, the preference for most patients is abiraterone since it is the least expensive agent,” he said. A generic version has been available for several years.
Additionally, abiraterone “has been on the market for the longest, and therefore clinicians are familiar with its use,” Schoen said. However, “clinicians have little idea of the comparative efficacy between these 2 agents,” he added.
The authors suggest that the cost and toxicities of the medications should guide clinician decisions, Schoen said. “There is data that abiraterone may worsen diabetes, since it is given with prednisone and could increase the risk of cardiovascular events,” he said.
He added that 2 newer drugs, apalutamide and darolutamide, are also “viable options.” Chemotherapies and certain targeted drugs are also available, “but they are only used in a select group of patients.”
Outside Specialist: Diverse Study Population Is a Plus
Hematologist-oncologist Natalie Reizine, MD, of the University of Illinois College of Medicine, Chicago, who was not involved in the study, told Federal Practitioner that the real-world data are valuable given the limitations of clinical trial populations.
“It’s difficult to compare clinical trials because they enroll different groups of patients,” she said. And, she said, they often exclude patients with significant comorbidities. “If they have bad cardiovascular disease, for instance, or poorly controlled diabetes, they're excluded from the clinical trial. But in real life, many of our patients have other medical problems that we have to manage.”
Reizine also emphasized the significance of the study’s diverse patient population. “Black men are very underrepresented in clinical trials. Many clinical trials that lead to drug approval will have only few or no Black men at all, yet these drugs go on to be widely prescribed to all men with prostate cancer.”
Results Are ‘Reassuring’
Reizine described the overall study findings as “reassuring,” especially in light of “studies that show that abiraterone and prednisone may be associated with worse cardiovascular outcomes. This study showed that in this VA population, even for patients who had cardiovascular disease, there was not a difference in how they did.”
As for choosing between agents, she recommended considering comorbidities and potential drug-drug interactions. “One of the big reasons that you may not be able to safely prescribe enzalutamide, for instance, is if a patient is on an anticoagulant, which is incredibly common in cancer patients. Enzalutamide has more drug-drug interactions than abiraterone and prednisone.”
Study Demographics and Findings
The study included all patients with mHSPC who initiated abiraterone or enzalutamide between July 2017 and April 2023.
Median ages were 73 (abiraterone) and 74 years (enzalutamide, P = .29). Racial distribution was similar between groups: abiraterone (68.1% White, 25.0% Black, 6.9% other/unknown) and enzalutamide (66.6% White, 27.0% Black, 6.4% other/unknown; P = .74). Ethnicity was 89.2% non-Hispanic, 4.4% Hispanic, and 6.4% unknown in the abiraterone group vs 88.4% non-Hispanic, 3.5% Hispanic, and 8.0% unknown in the enzalutamide group (P = .50).
The groups had similar rates of the most common comorbidities: diabetes (40.5% vs 46.3%, respectively, P = .07), peripheral vascular disease (40.2% vs 37.6%, respectively, P = .44), and chronic pulmonary disease (37.0% vs 40.5%, P = .29).
In an inverse probability weighting analysis with abiraterone as reference, weighted median overall survival was comparable across the entire cohort (36.2 months, P = .32), Black veterans (39.7 months, P = .90), and those with cardiovascular disease (31.5 months, P = .30).
The authors noted limitations such as the observational cohort design and data constraints.
The study was supported by the American Society of Clinical Oncology Conquer Cancer Foundation, the Prostate Cancer Foundation, and the Blavatnik Family Foundation.
Schoen discloses relationships with the Prostate Cancer Foundation, Astellas, and US Department of Defense. Other authors disclose relationships with the American Society of Clinical Oncology, Pfizer, Exelixis, Eli Lilly, Sanofi, Merck, Seagen, Bellicum, and BMS.
Outside the submitted work. Reizine discloses relationships with the US Department of Defense, Sanofi, Exelexis, Janssen, AstraZeneca, EMD Serono, Janssen, Merck, and Tempus.
Abiraterone and enzalutamide showed identical survival outcomes when used as first-line treatment for metastatic hormone-sensitive prostate cancer (mHSPC), according to a new study using US Department of Veterans Affairs (VA) data. The report represents the first head-to-head clinical analysis of these commonly used androgen receptor inhibitors.
Among 1258 veterans treated with abiraterone and 311 treated with enzalutamide, median overall survival was 36.2 months for both drugs. Patients were followed for a mean of 28.7 months (abiraterone) and 30.8 months (enzalutamide), reported by Martin W. Schoen, MD, MPH, from Saint Louis University School of Medicine and the St. Louis VA Medical Center, in JAMA Network Open.
Notably, there was no significant difference in outcomes among Black veterans, who often have poorer outcomes in prostate cancer, and in patients with cardiovascular disease.
“This is the first direct comparison of abiraterone and enzalutamide for mHSPC in a clinical practice setting,” Schoen told Federal Practitioner. “At the population level, there are no differences based on initial treatment choice.”
Abiraterone Is Preferred in the VA Due to Cost
According to Schoen, abiraterone and enzalutamide are the most commonly used androgen receptor inhibitors to treat mHSPC within the VA. A 2025 study by Schoen and colleagues found that 53.7% of veterans with mHSPC in 2022 received androgen receptor inhibitor therapy, up from 16.9% in 2017.
“In the VA, the preference for most patients is abiraterone since it is the least expensive agent,” he said. A generic version has been available for several years.
Additionally, abiraterone “has been on the market for the longest, and therefore clinicians are familiar with its use,” Schoen said. However, “clinicians have little idea of the comparative efficacy between these 2 agents,” he added.
The authors suggest that the cost and toxicities of the medications should guide clinician decisions, Schoen said. “There is data that abiraterone may worsen diabetes, since it is given with prednisone and could increase the risk of cardiovascular events,” he said.
He added that 2 newer drugs, apalutamide and darolutamide, are also “viable options.” Chemotherapies and certain targeted drugs are also available, “but they are only used in a select group of patients.”
Outside Specialist: Diverse Study Population Is a Plus
Hematologist-oncologist Natalie Reizine, MD, of the University of Illinois College of Medicine, Chicago, who was not involved in the study, told Federal Practitioner that the real-world data are valuable given the limitations of clinical trial populations.
“It’s difficult to compare clinical trials because they enroll different groups of patients,” she said. And, she said, they often exclude patients with significant comorbidities. “If they have bad cardiovascular disease, for instance, or poorly controlled diabetes, they're excluded from the clinical trial. But in real life, many of our patients have other medical problems that we have to manage.”
Reizine also emphasized the significance of the study’s diverse patient population. “Black men are very underrepresented in clinical trials. Many clinical trials that lead to drug approval will have only few or no Black men at all, yet these drugs go on to be widely prescribed to all men with prostate cancer.”
Results Are ‘Reassuring’
Reizine described the overall study findings as “reassuring,” especially in light of “studies that show that abiraterone and prednisone may be associated with worse cardiovascular outcomes. This study showed that in this VA population, even for patients who had cardiovascular disease, there was not a difference in how they did.”
As for choosing between agents, she recommended considering comorbidities and potential drug-drug interactions. “One of the big reasons that you may not be able to safely prescribe enzalutamide, for instance, is if a patient is on an anticoagulant, which is incredibly common in cancer patients. Enzalutamide has more drug-drug interactions than abiraterone and prednisone.”
Study Demographics and Findings
The study included all patients with mHSPC who initiated abiraterone or enzalutamide between July 2017 and April 2023.
Median ages were 73 (abiraterone) and 74 years (enzalutamide, P = .29). Racial distribution was similar between groups: abiraterone (68.1% White, 25.0% Black, 6.9% other/unknown) and enzalutamide (66.6% White, 27.0% Black, 6.4% other/unknown; P = .74). Ethnicity was 89.2% non-Hispanic, 4.4% Hispanic, and 6.4% unknown in the abiraterone group vs 88.4% non-Hispanic, 3.5% Hispanic, and 8.0% unknown in the enzalutamide group (P = .50).
The groups had similar rates of the most common comorbidities: diabetes (40.5% vs 46.3%, respectively, P = .07), peripheral vascular disease (40.2% vs 37.6%, respectively, P = .44), and chronic pulmonary disease (37.0% vs 40.5%, P = .29).
In an inverse probability weighting analysis with abiraterone as reference, weighted median overall survival was comparable across the entire cohort (36.2 months, P = .32), Black veterans (39.7 months, P = .90), and those with cardiovascular disease (31.5 months, P = .30).
The authors noted limitations such as the observational cohort design and data constraints.
The study was supported by the American Society of Clinical Oncology Conquer Cancer Foundation, the Prostate Cancer Foundation, and the Blavatnik Family Foundation.
Schoen discloses relationships with the Prostate Cancer Foundation, Astellas, and US Department of Defense. Other authors disclose relationships with the American Society of Clinical Oncology, Pfizer, Exelixis, Eli Lilly, Sanofi, Merck, Seagen, Bellicum, and BMS.
Outside the submitted work. Reizine discloses relationships with the US Department of Defense, Sanofi, Exelexis, Janssen, AstraZeneca, EMD Serono, Janssen, Merck, and Tempus.
NICE Endorses Oral Alternative to Chemo in Prostate Cancer
A faster, oral alternative to docetaxel is set to reach NHS clinics after the National Institute for Health and Care Excellence (NICE) recommended darolutamide (Nubeqa, Bayer) in combination with androgen deprivation therapy (ADT) for men with metastatic hormone-sensitive prostate cancer who are unable to receive or tolerate chemotherapy.
Detailed in NICE’s final draft guidance, the decision will make darolutamide available through the NHS in England and Wales to approximately 6000 patients, offering a new oral therapy for those who with limited alternatives to docetaxel or other androgen-receptor inhibitors.
New Option for Chemo-Ineligible Patients
Darolutamide functions by blocking hormones that fuel cancer growth, specifically depriving prostate cancer cells of testosterone required for multiplication and spread. Patients take two tablets twice daily alongside standard ADT.
Peter Johnson, national clinical director for cancer at NHS England, welcomed the decision and expects this approval to give clinicians and their patients “more flexibility to choose the approach best suited to individual circumstances and clinical needs.”
The guidance was finalised 5 weeks ahead of the standard review timeline, underscoring NICE’s commitment to accelerating access to effective prostate cancer treatments.
Clinical Trial Evidence
The NICE’s decision was supported by evidence from the phase 3 ARASENS trial (N = 1306).
The results showed that adding darolutamide to ADT and docetaxel significantly improved overall survival in metastatic hormone-sensitive prostate cancer, reducing the risk for death by 32% compared with ADT and docetaxel alone. Progression-free outcomes, measured by time to castration-resistant disease or death, also favoured darolutamide.
A NICE network meta-analysis of the TITAN, ARCHES, LATITUDE, and STAMPEDE trials suggested that combining ADT with androgen-receptor pathway inhibitors such as apalutamide, enzalutamide, and abiraterone provides comparable survival benefits in this disease setting.
Cost and Implementation
NICE determined that darolutamide plus ADT delivers similar or lower overall costs to the NHS compared with apalutamide plus ADT. The list price is £4040.00 for a 28-day supply (112 × 300-mg tablets), though Bayer has agreed to a confidential commercial discount.
The guidance requires healthcare providers to use the least expensive suitable treatment option, considering administration costs, dosages, price per dose, and commercial arrangements when choosing between darolutamide plus ADT and apalutamide plus ADT.
NHS England and integrated care boards must provide funding within 30 days of final publication, with routine commissioning beginning after this interim period.
A version of this article first appeared on Medscape.com.
A faster, oral alternative to docetaxel is set to reach NHS clinics after the National Institute for Health and Care Excellence (NICE) recommended darolutamide (Nubeqa, Bayer) in combination with androgen deprivation therapy (ADT) for men with metastatic hormone-sensitive prostate cancer who are unable to receive or tolerate chemotherapy.
Detailed in NICE’s final draft guidance, the decision will make darolutamide available through the NHS in England and Wales to approximately 6000 patients, offering a new oral therapy for those who with limited alternatives to docetaxel or other androgen-receptor inhibitors.
New Option for Chemo-Ineligible Patients
Darolutamide functions by blocking hormones that fuel cancer growth, specifically depriving prostate cancer cells of testosterone required for multiplication and spread. Patients take two tablets twice daily alongside standard ADT.
Peter Johnson, national clinical director for cancer at NHS England, welcomed the decision and expects this approval to give clinicians and their patients “more flexibility to choose the approach best suited to individual circumstances and clinical needs.”
The guidance was finalised 5 weeks ahead of the standard review timeline, underscoring NICE’s commitment to accelerating access to effective prostate cancer treatments.
Clinical Trial Evidence
The NICE’s decision was supported by evidence from the phase 3 ARASENS trial (N = 1306).
The results showed that adding darolutamide to ADT and docetaxel significantly improved overall survival in metastatic hormone-sensitive prostate cancer, reducing the risk for death by 32% compared with ADT and docetaxel alone. Progression-free outcomes, measured by time to castration-resistant disease or death, also favoured darolutamide.
A NICE network meta-analysis of the TITAN, ARCHES, LATITUDE, and STAMPEDE trials suggested that combining ADT with androgen-receptor pathway inhibitors such as apalutamide, enzalutamide, and abiraterone provides comparable survival benefits in this disease setting.
Cost and Implementation
NICE determined that darolutamide plus ADT delivers similar or lower overall costs to the NHS compared with apalutamide plus ADT. The list price is £4040.00 for a 28-day supply (112 × 300-mg tablets), though Bayer has agreed to a confidential commercial discount.
The guidance requires healthcare providers to use the least expensive suitable treatment option, considering administration costs, dosages, price per dose, and commercial arrangements when choosing between darolutamide plus ADT and apalutamide plus ADT.
NHS England and integrated care boards must provide funding within 30 days of final publication, with routine commissioning beginning after this interim period.
A version of this article first appeared on Medscape.com.
A faster, oral alternative to docetaxel is set to reach NHS clinics after the National Institute for Health and Care Excellence (NICE) recommended darolutamide (Nubeqa, Bayer) in combination with androgen deprivation therapy (ADT) for men with metastatic hormone-sensitive prostate cancer who are unable to receive or tolerate chemotherapy.
Detailed in NICE’s final draft guidance, the decision will make darolutamide available through the NHS in England and Wales to approximately 6000 patients, offering a new oral therapy for those who with limited alternatives to docetaxel or other androgen-receptor inhibitors.
New Option for Chemo-Ineligible Patients
Darolutamide functions by blocking hormones that fuel cancer growth, specifically depriving prostate cancer cells of testosterone required for multiplication and spread. Patients take two tablets twice daily alongside standard ADT.
Peter Johnson, national clinical director for cancer at NHS England, welcomed the decision and expects this approval to give clinicians and their patients “more flexibility to choose the approach best suited to individual circumstances and clinical needs.”
The guidance was finalised 5 weeks ahead of the standard review timeline, underscoring NICE’s commitment to accelerating access to effective prostate cancer treatments.
Clinical Trial Evidence
The NICE’s decision was supported by evidence from the phase 3 ARASENS trial (N = 1306).
The results showed that adding darolutamide to ADT and docetaxel significantly improved overall survival in metastatic hormone-sensitive prostate cancer, reducing the risk for death by 32% compared with ADT and docetaxel alone. Progression-free outcomes, measured by time to castration-resistant disease or death, also favoured darolutamide.
A NICE network meta-analysis of the TITAN, ARCHES, LATITUDE, and STAMPEDE trials suggested that combining ADT with androgen-receptor pathway inhibitors such as apalutamide, enzalutamide, and abiraterone provides comparable survival benefits in this disease setting.
Cost and Implementation
NICE determined that darolutamide plus ADT delivers similar or lower overall costs to the NHS compared with apalutamide plus ADT. The list price is £4040.00 for a 28-day supply (112 × 300-mg tablets), though Bayer has agreed to a confidential commercial discount.
The guidance requires healthcare providers to use the least expensive suitable treatment option, considering administration costs, dosages, price per dose, and commercial arrangements when choosing between darolutamide plus ADT and apalutamide plus ADT.
NHS England and integrated care boards must provide funding within 30 days of final publication, with routine commissioning beginning after this interim period.
A version of this article first appeared on Medscape.com.