User login
For MD-IQ only
Enhancing Molecular Testing Documentation in Prostate Cancer
Background
Prostate cancer is the most common non-cutaneous malignancy at the Veterans Health Administration (VHA) and every year approximately 15,000 Veterans are diagnosed and treated. Many advanced prostate cancer cases harbor genetic mutations that significantly impact prognosis, treatment decisions, and familial screening. In February 2021, the Prostate Cancer Molecular Testing Pathway (PCMTP) flow map was developed to increase appropriate genetic testing.
Methods
VHA initiated the Oncology Clinical Pathways (OCP) program to standardize cancer care for Veterans. The PCMTP was developed by a multidisciplinary team that created interactive templates within the Computerized Patient Record System (CPRS), to facilitate identification of eligible Veterans for germline and comprehensive genomic profiling (CGP). Clinical decision-making for these tests is documented as Health Factors (HF), in CPRS, allowing for assessment of pathway adherence and overall uptake.
Results
The PCMTP has achieved success, as there is over 90% compliance to molecular testing among participating Veterans which exceeds the pathway benchmark of 80%. PCMTP has been utilized at 88 VA sites, by over 700 distinct VA providers, with over 7,000 Veterans participating. This implementation has yielded over 19,200 Health Factors within CPRS.
Conclusions
The PCMTP has markedly improved the documentation and application of germline and CGP testing among Veterans diagnosed with prostate cancer. By facilitating genomic testing in appropriate patients, the PCMTP aims to enhance patient outcomes and optimize the quality of care. Prior to PCMTP establishment, assessing the prevalence of germline and CGP testing in eligible Veterans posed significant challenges. Future work will concentrate on increasing PCMTP utilization, evaluating downstream outcomes from genomic testing, including the identification of pathogenic variants, utilization of genetic counseling services, referrals to clinical trials, and the genomic impact on treatment strategies.
Background
Prostate cancer is the most common non-cutaneous malignancy at the Veterans Health Administration (VHA) and every year approximately 15,000 Veterans are diagnosed and treated. Many advanced prostate cancer cases harbor genetic mutations that significantly impact prognosis, treatment decisions, and familial screening. In February 2021, the Prostate Cancer Molecular Testing Pathway (PCMTP) flow map was developed to increase appropriate genetic testing.
Methods
VHA initiated the Oncology Clinical Pathways (OCP) program to standardize cancer care for Veterans. The PCMTP was developed by a multidisciplinary team that created interactive templates within the Computerized Patient Record System (CPRS), to facilitate identification of eligible Veterans for germline and comprehensive genomic profiling (CGP). Clinical decision-making for these tests is documented as Health Factors (HF), in CPRS, allowing for assessment of pathway adherence and overall uptake.
Results
The PCMTP has achieved success, as there is over 90% compliance to molecular testing among participating Veterans which exceeds the pathway benchmark of 80%. PCMTP has been utilized at 88 VA sites, by over 700 distinct VA providers, with over 7,000 Veterans participating. This implementation has yielded over 19,200 Health Factors within CPRS.
Conclusions
The PCMTP has markedly improved the documentation and application of germline and CGP testing among Veterans diagnosed with prostate cancer. By facilitating genomic testing in appropriate patients, the PCMTP aims to enhance patient outcomes and optimize the quality of care. Prior to PCMTP establishment, assessing the prevalence of germline and CGP testing in eligible Veterans posed significant challenges. Future work will concentrate on increasing PCMTP utilization, evaluating downstream outcomes from genomic testing, including the identification of pathogenic variants, utilization of genetic counseling services, referrals to clinical trials, and the genomic impact on treatment strategies.
Background
Prostate cancer is the most common non-cutaneous malignancy at the Veterans Health Administration (VHA) and every year approximately 15,000 Veterans are diagnosed and treated. Many advanced prostate cancer cases harbor genetic mutations that significantly impact prognosis, treatment decisions, and familial screening. In February 2021, the Prostate Cancer Molecular Testing Pathway (PCMTP) flow map was developed to increase appropriate genetic testing.
Methods
VHA initiated the Oncology Clinical Pathways (OCP) program to standardize cancer care for Veterans. The PCMTP was developed by a multidisciplinary team that created interactive templates within the Computerized Patient Record System (CPRS), to facilitate identification of eligible Veterans for germline and comprehensive genomic profiling (CGP). Clinical decision-making for these tests is documented as Health Factors (HF), in CPRS, allowing for assessment of pathway adherence and overall uptake.
Results
The PCMTP has achieved success, as there is over 90% compliance to molecular testing among participating Veterans which exceeds the pathway benchmark of 80%. PCMTP has been utilized at 88 VA sites, by over 700 distinct VA providers, with over 7,000 Veterans participating. This implementation has yielded over 19,200 Health Factors within CPRS.
Conclusions
The PCMTP has markedly improved the documentation and application of germline and CGP testing among Veterans diagnosed with prostate cancer. By facilitating genomic testing in appropriate patients, the PCMTP aims to enhance patient outcomes and optimize the quality of care. Prior to PCMTP establishment, assessing the prevalence of germline and CGP testing in eligible Veterans posed significant challenges. Future work will concentrate on increasing PCMTP utilization, evaluating downstream outcomes from genomic testing, including the identification of pathogenic variants, utilization of genetic counseling services, referrals to clinical trials, and the genomic impact on treatment strategies.
The Urology Prostate Cancer Note, One Tool to Increase Prostate Cancer Clinical Pathway Utilization
Background
Prostate cancer is the most common non-cutaneous malignancy diagnosis within the Department of Veterans Affairs (VA). The Prostate Cancer Clinical Pathways (PCCP) were developed to enable providers to treat all Veterans with prostate cancer at subject matter expert level.
Methods
The PCCP was launched in February 2021; however, provider documentation of PCCP is variable across the VA healthcare system and within the PCCP, specific flow maps have differential use. To increase urology specific flow map use, a collaboration between the National Surgery Office and National Oncology Program was established to develop a Urology Prostate Cancer Note (UPCN). The UPCN was designed by urologists with assistance from a medical oncologist and a clinical applications coordinator.
Results
The UPCN functions as a working clinical note for urologists and has the PCCPs embedded into reminder dialog templates, which when completed generate health factors. The health factors that are generated from the UPCN are data mined to record PCCP use and to perform data analytics. Since the UPCN national deployment on 9/6/24, documentation of high risk prostate cancer pathway utilization has increased 75% from 226 unique Veterans prior to launch to 395 unique Veterans after launch.
Conclusions
This collaborative effort did improve pathway utilization and documentation however other tools will need to be developed to improve provider PCCP documentation.
Background
Prostate cancer is the most common non-cutaneous malignancy diagnosis within the Department of Veterans Affairs (VA). The Prostate Cancer Clinical Pathways (PCCP) were developed to enable providers to treat all Veterans with prostate cancer at subject matter expert level.
Methods
The PCCP was launched in February 2021; however, provider documentation of PCCP is variable across the VA healthcare system and within the PCCP, specific flow maps have differential use. To increase urology specific flow map use, a collaboration between the National Surgery Office and National Oncology Program was established to develop a Urology Prostate Cancer Note (UPCN). The UPCN was designed by urologists with assistance from a medical oncologist and a clinical applications coordinator.
Results
The UPCN functions as a working clinical note for urologists and has the PCCPs embedded into reminder dialog templates, which when completed generate health factors. The health factors that are generated from the UPCN are data mined to record PCCP use and to perform data analytics. Since the UPCN national deployment on 9/6/24, documentation of high risk prostate cancer pathway utilization has increased 75% from 226 unique Veterans prior to launch to 395 unique Veterans after launch.
Conclusions
This collaborative effort did improve pathway utilization and documentation however other tools will need to be developed to improve provider PCCP documentation.
Background
Prostate cancer is the most common non-cutaneous malignancy diagnosis within the Department of Veterans Affairs (VA). The Prostate Cancer Clinical Pathways (PCCP) were developed to enable providers to treat all Veterans with prostate cancer at subject matter expert level.
Methods
The PCCP was launched in February 2021; however, provider documentation of PCCP is variable across the VA healthcare system and within the PCCP, specific flow maps have differential use. To increase urology specific flow map use, a collaboration between the National Surgery Office and National Oncology Program was established to develop a Urology Prostate Cancer Note (UPCN). The UPCN was designed by urologists with assistance from a medical oncologist and a clinical applications coordinator.
Results
The UPCN functions as a working clinical note for urologists and has the PCCPs embedded into reminder dialog templates, which when completed generate health factors. The health factors that are generated from the UPCN are data mined to record PCCP use and to perform data analytics. Since the UPCN national deployment on 9/6/24, documentation of high risk prostate cancer pathway utilization has increased 75% from 226 unique Veterans prior to launch to 395 unique Veterans after launch.
Conclusions
This collaborative effort did improve pathway utilization and documentation however other tools will need to be developed to improve provider PCCP documentation.
Access to Germline Genetic Testing through Clinical Pathways in Veterans With Prostate Cancer
Background
Germline genetic testing (GGT) is essential in prostate cancer care, informing clinical decisions. The Veterans Affairs National Oncology Program (VA NOP) recommends GGT for patients with specific risk factors in non-metastatic prostate cancer and all patients with metastatic disease. Understanding GGT access helps evaluate care quality and guide improvements. Since 2021, VA NOP has implemented pathway health factor (HF) templates to standardize cancer care documentation, including GGT status, enabling data extraction from the Corporate Data Warehouse (CDW) rather than requiring manual review of clinical notes. This work aims to evaluate Veterans’ access to GGT in prostate cancer care by leveraging pathway HF templates, and to assess the feasibility of using structured electronic health record (EHR) data to monitor adherence to GGT recommendations.
Methods
Process delivery diagrams (PDDs) were used to map data flow from prostate cancer clinical pathways to the VA CDW. We identified and categorized HFs related to prostate cancer GGT through the computerized patient record system (CPRS). Descriptive statistics were used to summarize access, ordering, and consent rates.
Results
We identified 5,744 Veterans with at least one prostate cancer GGT-relevant HF entered between 02/01/2021 and 12/31/2024. Of these, 5,125 (89.2%) had access to GGT, with 4,569 (89.2%) consenting to or having GGT ordered, while 556 (10.8%) declined testing. Among the 619 (10.8%) Veterans without GGT access, providers reported plans to discuss GGT in the future for 528 (85.3%) patients, while 91 (14.7%) were off pathway.
Conclusions
NOP-developed HF templates enabled extraction of GGT information from structured EHR data, eliminating manual extraction from clinical notes. We observed high GGT utilization among Veterans with pathway-entered HFs. However, low overall HF utilization may introduce selection bias. Future work includes developing a Natural Language Processing pipeline using large language models to automatically extract GGT information from clinical notes, with HF data serving as ground truth.
Background
Germline genetic testing (GGT) is essential in prostate cancer care, informing clinical decisions. The Veterans Affairs National Oncology Program (VA NOP) recommends GGT for patients with specific risk factors in non-metastatic prostate cancer and all patients with metastatic disease. Understanding GGT access helps evaluate care quality and guide improvements. Since 2021, VA NOP has implemented pathway health factor (HF) templates to standardize cancer care documentation, including GGT status, enabling data extraction from the Corporate Data Warehouse (CDW) rather than requiring manual review of clinical notes. This work aims to evaluate Veterans’ access to GGT in prostate cancer care by leveraging pathway HF templates, and to assess the feasibility of using structured electronic health record (EHR) data to monitor adherence to GGT recommendations.
Methods
Process delivery diagrams (PDDs) were used to map data flow from prostate cancer clinical pathways to the VA CDW. We identified and categorized HFs related to prostate cancer GGT through the computerized patient record system (CPRS). Descriptive statistics were used to summarize access, ordering, and consent rates.
Results
We identified 5,744 Veterans with at least one prostate cancer GGT-relevant HF entered between 02/01/2021 and 12/31/2024. Of these, 5,125 (89.2%) had access to GGT, with 4,569 (89.2%) consenting to or having GGT ordered, while 556 (10.8%) declined testing. Among the 619 (10.8%) Veterans without GGT access, providers reported plans to discuss GGT in the future for 528 (85.3%) patients, while 91 (14.7%) were off pathway.
Conclusions
NOP-developed HF templates enabled extraction of GGT information from structured EHR data, eliminating manual extraction from clinical notes. We observed high GGT utilization among Veterans with pathway-entered HFs. However, low overall HF utilization may introduce selection bias. Future work includes developing a Natural Language Processing pipeline using large language models to automatically extract GGT information from clinical notes, with HF data serving as ground truth.
Background
Germline genetic testing (GGT) is essential in prostate cancer care, informing clinical decisions. The Veterans Affairs National Oncology Program (VA NOP) recommends GGT for patients with specific risk factors in non-metastatic prostate cancer and all patients with metastatic disease. Understanding GGT access helps evaluate care quality and guide improvements. Since 2021, VA NOP has implemented pathway health factor (HF) templates to standardize cancer care documentation, including GGT status, enabling data extraction from the Corporate Data Warehouse (CDW) rather than requiring manual review of clinical notes. This work aims to evaluate Veterans’ access to GGT in prostate cancer care by leveraging pathway HF templates, and to assess the feasibility of using structured electronic health record (EHR) data to monitor adherence to GGT recommendations.
Methods
Process delivery diagrams (PDDs) were used to map data flow from prostate cancer clinical pathways to the VA CDW. We identified and categorized HFs related to prostate cancer GGT through the computerized patient record system (CPRS). Descriptive statistics were used to summarize access, ordering, and consent rates.
Results
We identified 5,744 Veterans with at least one prostate cancer GGT-relevant HF entered between 02/01/2021 and 12/31/2024. Of these, 5,125 (89.2%) had access to GGT, with 4,569 (89.2%) consenting to or having GGT ordered, while 556 (10.8%) declined testing. Among the 619 (10.8%) Veterans without GGT access, providers reported plans to discuss GGT in the future for 528 (85.3%) patients, while 91 (14.7%) were off pathway.
Conclusions
NOP-developed HF templates enabled extraction of GGT information from structured EHR data, eliminating manual extraction from clinical notes. We observed high GGT utilization among Veterans with pathway-entered HFs. However, low overall HF utilization may introduce selection bias. Future work includes developing a Natural Language Processing pipeline using large language models to automatically extract GGT information from clinical notes, with HF data serving as ground truth.
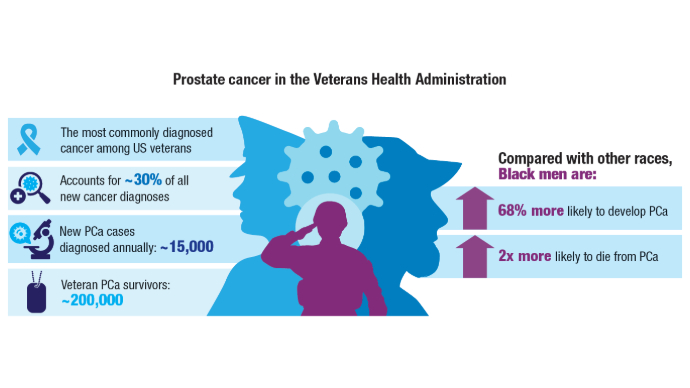
Genomic Testing Reveals Distinct Mutation Patterns in Black and White Veterans With Metastatic Prostate Cancer
TOPLINE: Next-generation sequencing (NGS) analysis of 5015 veterans with metastatic prostate cancer reveals distinct genomic patterns between non-Hispanic Black and White patients, with Black veterans showing higher odds of immunotherapy targets but lower odds of androgen receptor axis alterations. However, the rates of survival were similar despite the differences.
METHODOLOGY:
Researchers conducted a retrospective cohort study comparing alteration frequencies between 1784 non-Hispanic Black (35.6%) and 3,231 non-Hispanic White (64.4%) veterans who underwent NGS testing from January 23, 2019, to November 2, 2023.
- Analysis included DNA sequencing data from tissue or plasma biospecimens, including prostate biopsy specimens, radical prostatectomy specimens, and prostate cancer metastases, all sequenced with FoundationOne CDx or FoundationOne Liquid CDx platforms.
- Investigators examined pathogenic alterations in individual genes, actionable targets, and canonical prostate cancer pathways, while adjusting for NGS analyte and clinicopathologic covariates.
- Researchers evaluated associations between alteration frequency and race as well as survival through Cox proportional hazards modeling, stratified by race and adjusted for clinical factors.
TAKEAWAY:
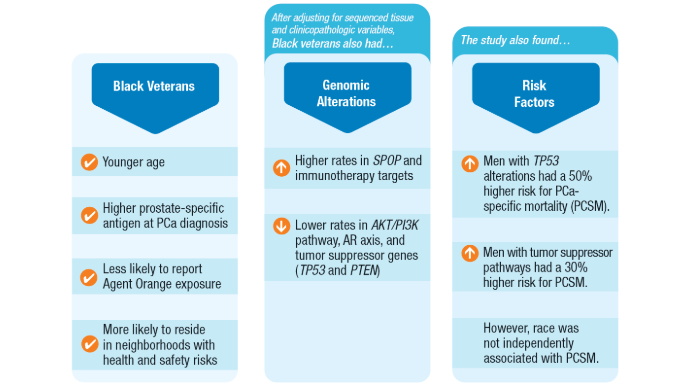
Non-Hispanic Black race and ethnicity was associated with higher odds of genomic alterations in SPOP (odds ratio [OR], 1.7; 95% confidence interval [CI], 1.2-2.6) and immunotherapy targets (OR, 1.7; 95% CI, 1.1-2.5), including high microsatellite instability status (OR, 3.1; 95% CI, 1.1-9.4).
- Non-Hispanic Black veterans showed lower odds of genomic alterations in the AKT/PI3K pathway (OR, 0.6; 95% CI, 0.4-0.7), androgen receptor axis (OR, 0.7; 95% CI, 0.5-0.9), and tumor suppressor genes (OR, 0.7; 95% CI, 0.5-0.8).
- Tumor suppressor alterations were associated with shorter overall survival in both non-Hispanic Black (hazard ratio [HR], 1.54; 95% CI, 1.13-2.11) and non-Hispanic White (HR, 1.52; 95% CI, 1.25-1.85) veterans.
- CDK12 alterations significantly increased the hazard of death in non-Hispanic Black veterans (HR, 2.04; 95% CI, 1.13-3.67), while immunotherapy targets were associated with increased mortality in non-Hispanic White veterans (HR, 1.44; 95% CI, 1.02-2.02).
IN PRACTICE: " we did not identify any genomic alterations or biomarkers that should not be tested in PCa based on patient self-identified race. Ultimately, this work emphasizes that precision oncology enables the individualization of treatment decisions without having to rely on imprecise characteristics such as self-identified race.," wrote the study authors.
SOURCE: Isla P. Garraway, MD, PhD; Kosj Yamoah, MD, PhD; and Kara N. Maxwell, MD, PhD were co-senior authors. The article was published online on May 12 in JAMA Network Open.
LIMITATIONS: According to the authors, a lack of matched germline data for patients, complicated the interpretation of plasma results. In addition, survivorship bias may have inadvertently excluded the most aggressive metastatic prostate cancer phenotypes, as patients who did not live long enough to undergo NGS testing were not included. Results seen in the veteran population served by the Veterans Health Administration may not be generalizable to the broader population.
DISCLOSURES: The study received support from Challenge Award PCF22CHALO2 from the Prostate Cancer Foundation and the Veterans Affairs National Precision Oncology Program. Luca F. Valle, MD, reported receiving grant support from the Bristol Myers Squibb Foundation during the conduct of the study. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Next-generation sequencing (NGS) analysis of 5015 veterans with metastatic prostate cancer reveals distinct genomic patterns between non-Hispanic Black and White patients, with Black veterans showing higher odds of immunotherapy targets but lower odds of androgen receptor axis alterations. However, the rates of survival were similar despite the differences.
METHODOLOGY:
Researchers conducted a retrospective cohort study comparing alteration frequencies between 1784 non-Hispanic Black (35.6%) and 3,231 non-Hispanic White (64.4%) veterans who underwent NGS testing from January 23, 2019, to November 2, 2023.
- Analysis included DNA sequencing data from tissue or plasma biospecimens, including prostate biopsy specimens, radical prostatectomy specimens, and prostate cancer metastases, all sequenced with FoundationOne CDx or FoundationOne Liquid CDx platforms.
- Investigators examined pathogenic alterations in individual genes, actionable targets, and canonical prostate cancer pathways, while adjusting for NGS analyte and clinicopathologic covariates.
- Researchers evaluated associations between alteration frequency and race as well as survival through Cox proportional hazards modeling, stratified by race and adjusted for clinical factors.
TAKEAWAY:
Non-Hispanic Black race and ethnicity was associated with higher odds of genomic alterations in SPOP (odds ratio [OR], 1.7; 95% confidence interval [CI], 1.2-2.6) and immunotherapy targets (OR, 1.7; 95% CI, 1.1-2.5), including high microsatellite instability status (OR, 3.1; 95% CI, 1.1-9.4).
- Non-Hispanic Black veterans showed lower odds of genomic alterations in the AKT/PI3K pathway (OR, 0.6; 95% CI, 0.4-0.7), androgen receptor axis (OR, 0.7; 95% CI, 0.5-0.9), and tumor suppressor genes (OR, 0.7; 95% CI, 0.5-0.8).
- Tumor suppressor alterations were associated with shorter overall survival in both non-Hispanic Black (hazard ratio [HR], 1.54; 95% CI, 1.13-2.11) and non-Hispanic White (HR, 1.52; 95% CI, 1.25-1.85) veterans.
- CDK12 alterations significantly increased the hazard of death in non-Hispanic Black veterans (HR, 2.04; 95% CI, 1.13-3.67), while immunotherapy targets were associated with increased mortality in non-Hispanic White veterans (HR, 1.44; 95% CI, 1.02-2.02).
IN PRACTICE: " we did not identify any genomic alterations or biomarkers that should not be tested in PCa based on patient self-identified race. Ultimately, this work emphasizes that precision oncology enables the individualization of treatment decisions without having to rely on imprecise characteristics such as self-identified race.," wrote the study authors.
SOURCE: Isla P. Garraway, MD, PhD; Kosj Yamoah, MD, PhD; and Kara N. Maxwell, MD, PhD were co-senior authors. The article was published online on May 12 in JAMA Network Open.
LIMITATIONS: According to the authors, a lack of matched germline data for patients, complicated the interpretation of plasma results. In addition, survivorship bias may have inadvertently excluded the most aggressive metastatic prostate cancer phenotypes, as patients who did not live long enough to undergo NGS testing were not included. Results seen in the veteran population served by the Veterans Health Administration may not be generalizable to the broader population.
DISCLOSURES: The study received support from Challenge Award PCF22CHALO2 from the Prostate Cancer Foundation and the Veterans Affairs National Precision Oncology Program. Luca F. Valle, MD, reported receiving grant support from the Bristol Myers Squibb Foundation during the conduct of the study. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE: Next-generation sequencing (NGS) analysis of 5015 veterans with metastatic prostate cancer reveals distinct genomic patterns between non-Hispanic Black and White patients, with Black veterans showing higher odds of immunotherapy targets but lower odds of androgen receptor axis alterations. However, the rates of survival were similar despite the differences.
METHODOLOGY:
Researchers conducted a retrospective cohort study comparing alteration frequencies between 1784 non-Hispanic Black (35.6%) and 3,231 non-Hispanic White (64.4%) veterans who underwent NGS testing from January 23, 2019, to November 2, 2023.
- Analysis included DNA sequencing data from tissue or plasma biospecimens, including prostate biopsy specimens, radical prostatectomy specimens, and prostate cancer metastases, all sequenced with FoundationOne CDx or FoundationOne Liquid CDx platforms.
- Investigators examined pathogenic alterations in individual genes, actionable targets, and canonical prostate cancer pathways, while adjusting for NGS analyte and clinicopathologic covariates.
- Researchers evaluated associations between alteration frequency and race as well as survival through Cox proportional hazards modeling, stratified by race and adjusted for clinical factors.
TAKEAWAY:
Non-Hispanic Black race and ethnicity was associated with higher odds of genomic alterations in SPOP (odds ratio [OR], 1.7; 95% confidence interval [CI], 1.2-2.6) and immunotherapy targets (OR, 1.7; 95% CI, 1.1-2.5), including high microsatellite instability status (OR, 3.1; 95% CI, 1.1-9.4).
- Non-Hispanic Black veterans showed lower odds of genomic alterations in the AKT/PI3K pathway (OR, 0.6; 95% CI, 0.4-0.7), androgen receptor axis (OR, 0.7; 95% CI, 0.5-0.9), and tumor suppressor genes (OR, 0.7; 95% CI, 0.5-0.8).
- Tumor suppressor alterations were associated with shorter overall survival in both non-Hispanic Black (hazard ratio [HR], 1.54; 95% CI, 1.13-2.11) and non-Hispanic White (HR, 1.52; 95% CI, 1.25-1.85) veterans.
- CDK12 alterations significantly increased the hazard of death in non-Hispanic Black veterans (HR, 2.04; 95% CI, 1.13-3.67), while immunotherapy targets were associated with increased mortality in non-Hispanic White veterans (HR, 1.44; 95% CI, 1.02-2.02).
IN PRACTICE: " we did not identify any genomic alterations or biomarkers that should not be tested in PCa based on patient self-identified race. Ultimately, this work emphasizes that precision oncology enables the individualization of treatment decisions without having to rely on imprecise characteristics such as self-identified race.," wrote the study authors.
SOURCE: Isla P. Garraway, MD, PhD; Kosj Yamoah, MD, PhD; and Kara N. Maxwell, MD, PhD were co-senior authors. The article was published online on May 12 in JAMA Network Open.
LIMITATIONS: According to the authors, a lack of matched germline data for patients, complicated the interpretation of plasma results. In addition, survivorship bias may have inadvertently excluded the most aggressive metastatic prostate cancer phenotypes, as patients who did not live long enough to undergo NGS testing were not included. Results seen in the veteran population served by the Veterans Health Administration may not be generalizable to the broader population.
DISCLOSURES: The study received support from Challenge Award PCF22CHALO2 from the Prostate Cancer Foundation and the Veterans Affairs National Precision Oncology Program. Luca F. Valle, MD, reported receiving grant support from the Bristol Myers Squibb Foundation during the conduct of the study. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
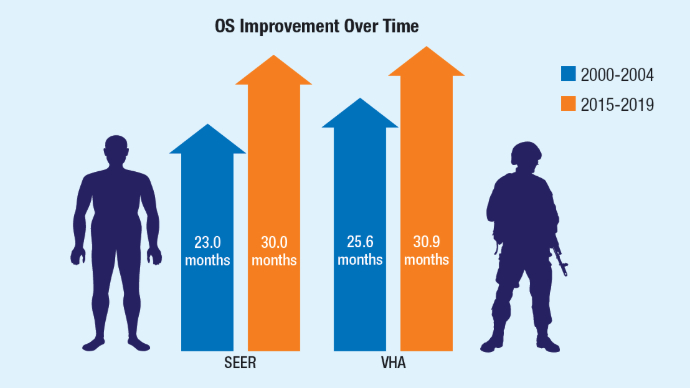
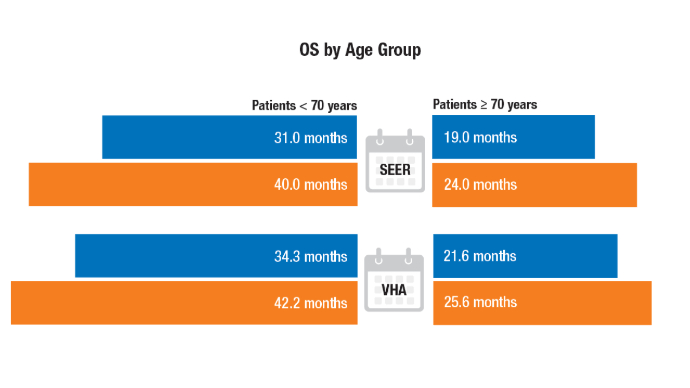
Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
Click here to view more from Cancer Data Trends 2025.
References
Lillard JW Jr, Moses KA, Mahal BA, George DJ. Racial disparities in Black men with prostate cancer: A literature review. Cancer. 2022 Nov 1;128(21):3787-3795. doi:10.1002/cncr.34433
Wang BR, Chen Y-A, Kao W-H, Lai C-H, Lin H, Hsieh J-T. Developing New Treatment Options for Castration-Resistant Prostate Cancer and Recurrent Disease. Biomedicines. 2022 Aug 3;10(8):1872. doi:10.3390/biomedicines10081872
Valle LF, Li J, Desai H, Hausler R, et al. Oncogenic Alterations, Race, and Survival in US Veterans with Metastatic Prostate Cancer Undergoing Somatic Tumor Next Generation Sequencing. bioRxiv [Preprint]. 2024 Oct 25:2024.10.24.620071. doi:10.1101/2024.10.24.620071
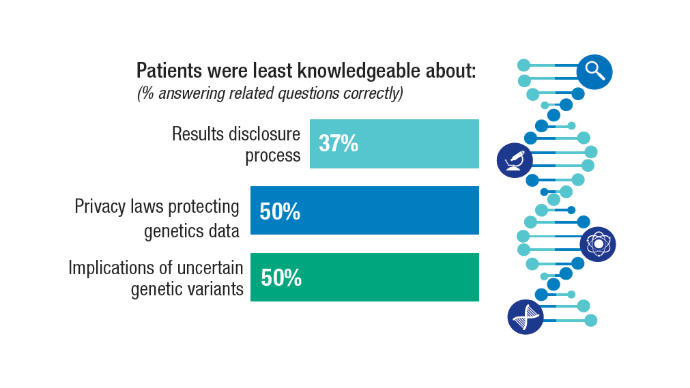
Kwon DH, Scheuner MT, McPhaul M, et al. Germline testing for veterans with advanced prostate cancer: concerns about service-connected benefits. JNCI Cancer Spectr. 2024 Sep 2;8(5):pkae079. doi:10.1093/jncics/pkae079
Kwon DH, McPhaul M, Sumra S, et al. Informed decision-making about germline testing among Veterans with advanced prostate cancer (APC): A mixed-methods study. J Clin Oncol. 2024;42(16_suppl):5105. doi:10.1200/JCO.2024.42.16_suppl.5105
Schoen MW, Montgomery RB, Owens L, Khan S, Sanfilippo KM, Etzioni RB. Survival in Patients With De Novo Metastatic Prostate Cancer. JAMA Netw Open. 2024 Mar 4;7(3):e241970. doi: 10.1001/jamanetworkopen.2024.1970
Schafer EJ, Jemal A, Wiese D, et al. Disparities and Trends in Genitourinary Cancer Incidence and Mortality in the USA. Eur Urol. 2023 Jul;84(1):117-126. doi:10.1016/j.eururo.2022.11.023
U.S. Department of Veterans Affairs. Hines VA Hospital & Loyola University Chicago Physician Awarded $8.6M VA Research Grant. November 8, 2021. https://www.va.gov/hines-health-care/news-releases/hines-va-hospital-loyola-university-chicago-physician-awarded-86m-va-research-grant/ Accessed December 31, 2024.
U.S. Department of Veterans Affairs. National Oncology Program. How VA is Advancing Prostate Cancer Care. https://www.cancer.va.gov/prostate.html Accessed December 31, 2024.
Click here to view more from Cancer Data Trends 2025.
Click here to view more from Cancer Data Trends 2025.
References
Lillard JW Jr, Moses KA, Mahal BA, George DJ. Racial disparities in Black men with prostate cancer: A literature review. Cancer. 2022 Nov 1;128(21):3787-3795. doi:10.1002/cncr.34433
Wang BR, Chen Y-A, Kao W-H, Lai C-H, Lin H, Hsieh J-T. Developing New Treatment Options for Castration-Resistant Prostate Cancer and Recurrent Disease. Biomedicines. 2022 Aug 3;10(8):1872. doi:10.3390/biomedicines10081872
Valle LF, Li J, Desai H, Hausler R, et al. Oncogenic Alterations, Race, and Survival in US Veterans with Metastatic Prostate Cancer Undergoing Somatic Tumor Next Generation Sequencing. bioRxiv [Preprint]. 2024 Oct 25:2024.10.24.620071. doi:10.1101/2024.10.24.620071
Kwon DH, Scheuner MT, McPhaul M, et al. Germline testing for veterans with advanced prostate cancer: concerns about service-connected benefits. JNCI Cancer Spectr. 2024 Sep 2;8(5):pkae079. doi:10.1093/jncics/pkae079
Kwon DH, McPhaul M, Sumra S, et al. Informed decision-making about germline testing among Veterans with advanced prostate cancer (APC): A mixed-methods study. J Clin Oncol. 2024;42(16_suppl):5105. doi:10.1200/JCO.2024.42.16_suppl.5105
Schoen MW, Montgomery RB, Owens L, Khan S, Sanfilippo KM, Etzioni RB. Survival in Patients With De Novo Metastatic Prostate Cancer. JAMA Netw Open. 2024 Mar 4;7(3):e241970. doi: 10.1001/jamanetworkopen.2024.1970
Schafer EJ, Jemal A, Wiese D, et al. Disparities and Trends in Genitourinary Cancer Incidence and Mortality in the USA. Eur Urol. 2023 Jul;84(1):117-126. doi:10.1016/j.eururo.2022.11.023
U.S. Department of Veterans Affairs. Hines VA Hospital & Loyola University Chicago Physician Awarded $8.6M VA Research Grant. November 8, 2021. https://www.va.gov/hines-health-care/news-releases/hines-va-hospital-loyola-university-chicago-physician-awarded-86m-va-research-grant/ Accessed December 31, 2024.
U.S. Department of Veterans Affairs. National Oncology Program. How VA is Advancing Prostate Cancer Care. https://www.cancer.va.gov/prostate.html Accessed December 31, 2024.
References
Lillard JW Jr, Moses KA, Mahal BA, George DJ. Racial disparities in Black men with prostate cancer: A literature review. Cancer. 2022 Nov 1;128(21):3787-3795. doi:10.1002/cncr.34433
Wang BR, Chen Y-A, Kao W-H, Lai C-H, Lin H, Hsieh J-T. Developing New Treatment Options for Castration-Resistant Prostate Cancer and Recurrent Disease. Biomedicines. 2022 Aug 3;10(8):1872. doi:10.3390/biomedicines10081872
Valle LF, Li J, Desai H, Hausler R, et al. Oncogenic Alterations, Race, and Survival in US Veterans with Metastatic Prostate Cancer Undergoing Somatic Tumor Next Generation Sequencing. bioRxiv [Preprint]. 2024 Oct 25:2024.10.24.620071. doi:10.1101/2024.10.24.620071
Kwon DH, Scheuner MT, McPhaul M, et al. Germline testing for veterans with advanced prostate cancer: concerns about service-connected benefits. JNCI Cancer Spectr. 2024 Sep 2;8(5):pkae079. doi:10.1093/jncics/pkae079
Kwon DH, McPhaul M, Sumra S, et al. Informed decision-making about germline testing among Veterans with advanced prostate cancer (APC): A mixed-methods study. J Clin Oncol. 2024;42(16_suppl):5105. doi:10.1200/JCO.2024.42.16_suppl.5105
Schoen MW, Montgomery RB, Owens L, Khan S, Sanfilippo KM, Etzioni RB. Survival in Patients With De Novo Metastatic Prostate Cancer. JAMA Netw Open. 2024 Mar 4;7(3):e241970. doi: 10.1001/jamanetworkopen.2024.1970
Schafer EJ, Jemal A, Wiese D, et al. Disparities and Trends in Genitourinary Cancer Incidence and Mortality in the USA. Eur Urol. 2023 Jul;84(1):117-126. doi:10.1016/j.eururo.2022.11.023
U.S. Department of Veterans Affairs. Hines VA Hospital & Loyola University Chicago Physician Awarded $8.6M VA Research Grant. November 8, 2021. https://www.va.gov/hines-health-care/news-releases/hines-va-hospital-loyola-university-chicago-physician-awarded-86m-va-research-grant/ Accessed December 31, 2024.
U.S. Department of Veterans Affairs. National Oncology Program. How VA is Advancing Prostate Cancer Care. https://www.cancer.va.gov/prostate.html Accessed December 31, 2024.
Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
Study Gives Clinical Edge to Transperineal Prostate Biopsies
In the largest head-to-head randomized trial of its kind, UK researchers found transperineal prostate biopsies using local anesthesia (LATP) superior to the transrectal approach in detecting clinically significant cancers.
The TRANSLATE study, with more than 1100 patients, found LATP identified 5.7% more cases of clinically significant prostate cancer, defined as Grade Group 2 or higher, than biopsies using transrectal ultrasonography (TRUS).
Previous research comparing the two techniques has focused mainly on rates of infection rather than cancer detection, said Richard Bryant, PhD, a consultant urologist at Nuffield Department of Surgical Sciences, University of Oxford in Oxford, England, who led the trial.
“We decided that the most important thing to look at is the detection rate of clinically significant prostate cancer, because that is why the man is having the biopsy in the first place, rather than to avoid infection, although avoiding infection is of course also important,” Bryant said.
Bryant presented the findings at the 2025 annual congress of the European Association of Urology and his group published the results in The Lancet Oncology.
The TRANSLATE trial was powered to identify a difference in the rate of cancer detection but not factors such as pain and sepsis. Hospitalization after biopsies served as a proxy for sepsis.
Men in the trial were nearly twice as likely to report LATP to be problematic immediately after the procedure than those who underwent transrectal biopsies. These issues included pain, discomfort, and embarrassment.
Two of the 562 men in the LATP group were hospitalized within 35 days of the procedure compared with nine of the 564 in the TRUS group. Bryant said this trend favored LATP, but the difference did not reach statistical significance.
The data on infection and other secondary outcomes were encouraging, but not conclusive, Bryant’s group reported.
Richard Szabo, MD, a prostate biopsy researcher at University of California Irvine, said the reduction in post-biopsy sepsis has been “an additional major advantage” of transperineal over transrectal prostate biopsy.
Almost 90% of men who received LATP had the biopsies without antibiotics — a “bonus,” Bryant said.
Antibiotic stewardship is a major factor in research and policies regarding biopsies in Europe. Transperineal biopsies avoid the rectum and pass needles through the perineum between the anus and the testicles, reducing risk for infection.
Jim Hu, MD, a urologic oncology researcher at Weill Cornell in New York City and the principal investigator on the 2024 PREVENT trial, said three of four randomized trials reported in the past year, including PREVENT and TRANSLATE, have found transperineal biopsies resulted in fewer infections than the transrectal method.
European guidelines call transperineal biopsy the preferred approach based on infection concerns, whereas guidance from the American Urological Association gives equal weight to transperineal and transrectal biopsies.
Badar Mian, MD, a urologist at Albany Med Health System in Albany, New York, said the field should “shift our focus from picking a winner to instead focus on whether prostate biopsy procedures are safe and effective. Patients should be reassured that, while there are trade-offs, both procedures can be performed safely and with a high degree of accuracy.”
The UK’s National Institute for Health and Care Research funded TRANSLATE. Bryant received support from BXTAccelyon to attend biopsy training provided by Guys’ Hospital, in London, England.
Howard Wolinsky is a Chicago-based freelance writer.
A version of this article appeared at Medscape.com.
In the largest head-to-head randomized trial of its kind, UK researchers found transperineal prostate biopsies using local anesthesia (LATP) superior to the transrectal approach in detecting clinically significant cancers.
The TRANSLATE study, with more than 1100 patients, found LATP identified 5.7% more cases of clinically significant prostate cancer, defined as Grade Group 2 or higher, than biopsies using transrectal ultrasonography (TRUS).
Previous research comparing the two techniques has focused mainly on rates of infection rather than cancer detection, said Richard Bryant, PhD, a consultant urologist at Nuffield Department of Surgical Sciences, University of Oxford in Oxford, England, who led the trial.
“We decided that the most important thing to look at is the detection rate of clinically significant prostate cancer, because that is why the man is having the biopsy in the first place, rather than to avoid infection, although avoiding infection is of course also important,” Bryant said.
Bryant presented the findings at the 2025 annual congress of the European Association of Urology and his group published the results in The Lancet Oncology.
The TRANSLATE trial was powered to identify a difference in the rate of cancer detection but not factors such as pain and sepsis. Hospitalization after biopsies served as a proxy for sepsis.
Men in the trial were nearly twice as likely to report LATP to be problematic immediately after the procedure than those who underwent transrectal biopsies. These issues included pain, discomfort, and embarrassment.
Two of the 562 men in the LATP group were hospitalized within 35 days of the procedure compared with nine of the 564 in the TRUS group. Bryant said this trend favored LATP, but the difference did not reach statistical significance.
The data on infection and other secondary outcomes were encouraging, but not conclusive, Bryant’s group reported.
Richard Szabo, MD, a prostate biopsy researcher at University of California Irvine, said the reduction in post-biopsy sepsis has been “an additional major advantage” of transperineal over transrectal prostate biopsy.
Almost 90% of men who received LATP had the biopsies without antibiotics — a “bonus,” Bryant said.
Antibiotic stewardship is a major factor in research and policies regarding biopsies in Europe. Transperineal biopsies avoid the rectum and pass needles through the perineum between the anus and the testicles, reducing risk for infection.
Jim Hu, MD, a urologic oncology researcher at Weill Cornell in New York City and the principal investigator on the 2024 PREVENT trial, said three of four randomized trials reported in the past year, including PREVENT and TRANSLATE, have found transperineal biopsies resulted in fewer infections than the transrectal method.
European guidelines call transperineal biopsy the preferred approach based on infection concerns, whereas guidance from the American Urological Association gives equal weight to transperineal and transrectal biopsies.
Badar Mian, MD, a urologist at Albany Med Health System in Albany, New York, said the field should “shift our focus from picking a winner to instead focus on whether prostate biopsy procedures are safe and effective. Patients should be reassured that, while there are trade-offs, both procedures can be performed safely and with a high degree of accuracy.”
The UK’s National Institute for Health and Care Research funded TRANSLATE. Bryant received support from BXTAccelyon to attend biopsy training provided by Guys’ Hospital, in London, England.
Howard Wolinsky is a Chicago-based freelance writer.
A version of this article appeared at Medscape.com.
In the largest head-to-head randomized trial of its kind, UK researchers found transperineal prostate biopsies using local anesthesia (LATP) superior to the transrectal approach in detecting clinically significant cancers.
The TRANSLATE study, with more than 1100 patients, found LATP identified 5.7% more cases of clinically significant prostate cancer, defined as Grade Group 2 or higher, than biopsies using transrectal ultrasonography (TRUS).
Previous research comparing the two techniques has focused mainly on rates of infection rather than cancer detection, said Richard Bryant, PhD, a consultant urologist at Nuffield Department of Surgical Sciences, University of Oxford in Oxford, England, who led the trial.
“We decided that the most important thing to look at is the detection rate of clinically significant prostate cancer, because that is why the man is having the biopsy in the first place, rather than to avoid infection, although avoiding infection is of course also important,” Bryant said.
Bryant presented the findings at the 2025 annual congress of the European Association of Urology and his group published the results in The Lancet Oncology.
The TRANSLATE trial was powered to identify a difference in the rate of cancer detection but not factors such as pain and sepsis. Hospitalization after biopsies served as a proxy for sepsis.
Men in the trial were nearly twice as likely to report LATP to be problematic immediately after the procedure than those who underwent transrectal biopsies. These issues included pain, discomfort, and embarrassment.
Two of the 562 men in the LATP group were hospitalized within 35 days of the procedure compared with nine of the 564 in the TRUS group. Bryant said this trend favored LATP, but the difference did not reach statistical significance.
The data on infection and other secondary outcomes were encouraging, but not conclusive, Bryant’s group reported.
Richard Szabo, MD, a prostate biopsy researcher at University of California Irvine, said the reduction in post-biopsy sepsis has been “an additional major advantage” of transperineal over transrectal prostate biopsy.
Almost 90% of men who received LATP had the biopsies without antibiotics — a “bonus,” Bryant said.
Antibiotic stewardship is a major factor in research and policies regarding biopsies in Europe. Transperineal biopsies avoid the rectum and pass needles through the perineum between the anus and the testicles, reducing risk for infection.
Jim Hu, MD, a urologic oncology researcher at Weill Cornell in New York City and the principal investigator on the 2024 PREVENT trial, said three of four randomized trials reported in the past year, including PREVENT and TRANSLATE, have found transperineal biopsies resulted in fewer infections than the transrectal method.
European guidelines call transperineal biopsy the preferred approach based on infection concerns, whereas guidance from the American Urological Association gives equal weight to transperineal and transrectal biopsies.
Badar Mian, MD, a urologist at Albany Med Health System in Albany, New York, said the field should “shift our focus from picking a winner to instead focus on whether prostate biopsy procedures are safe and effective. Patients should be reassured that, while there are trade-offs, both procedures can be performed safely and with a high degree of accuracy.”
The UK’s National Institute for Health and Care Research funded TRANSLATE. Bryant received support from BXTAccelyon to attend biopsy training provided by Guys’ Hospital, in London, England.
Howard Wolinsky is a Chicago-based freelance writer.
A version of this article appeared at Medscape.com.
A Better Biopsy for Prostate Cancer?
Micro-ultrasound–guided biopsies were found for the first time to be “noninferior” to MRI-guided procedures, according to new research presented at the 2025 annual congress of the European Association of Urology.
The OPTIMUM study found 4.5% more clinically significant cancers among men who underwent micro-ultrasound–guided biopsies of the prostate than in those scanned using MRI.
“The take-home message is that men being worked up for an elevated PSA [prostate-specific antigen] or an abnormal digital rectal examination who are at increased risk of prostate cancer may safely undergo a micro-ultrasound–guided biopsy rather than an MRI-guided biopsy,” said Adam Kinnaird, MD, PhD, the Frank and Carla Sojonky Chair in Prostate Cancer Research at the University of Alberta, Edmonton, Alberta, Canada, and principal investigator of the study.
Micro-ultrasound can image to as small as 70 μm, ie, the width of a human hair.
OPTIMUM was an international, open-label, randomized, noninferiority trial in 20 centers in eight countries of men with clinical suspicion of prostate cancer, elevated PSAs, abnormal digital rectal exams, or a combination of these risk factors. None of the men previously had undergone biopsies.
The study had three arms to which men were assigned randomly: Micro-ultrasound–guided biopsy (n = 121); biopsies guided by micro-ultrasound and fusion MRI (n = 226), and MRI plus conventional ultrasound–guided biopsy (n = 331).
Subjects had a median age of 65 years and a median PSA level of 6.9 ng/mL; 83% self-identified as White individuals.
“Micro-ultrasound was found to be no worse than MRI at the detection of clinically significant prostate cancer. We don’t show it is equivalent. We don’t show it better. We show it is not worse,” Kinnaird said.
The study, funded by Exact Imaging, which makes the ExactVu micro-ultrasound platform, appeared simultaneously in JAMA.
Laurence Klotz, MD, the Sunnybrook Chair of Prostate Cancer Research at the University of Toronto Sunnybrook Health Sciences Centre, and senior researcher on the OPTIMUM trial, said as the incidence of prostate cancer rises globally, micro-ultrasound may be of particular value in low-income and middle-income countries where MRI is not widely available.
“It’s extremely appealing in places that can’t offer MRI to everyone, but I think it also will have a role going forward in regions where there is no problem about getting access to MRI,” Klotz told Medscape Medical News.
This group is next studying the financial aspects of the technology, he added.
Gerald Andriole, MD, then urology chief at Washington University in St. Louis, St. Louis, Missouri, designed the original studies of the ExactVu system, which the US Food and Drug Administration approved in 2017.
Andriole, now chief medical officer of Prostatype Genomics, said MRIs are costly, subjective, and uncomfortable for many patients, due to claustrophobia and obesity, requiring complicated co-registration procedures to perform an accurate targeted biopsy into the most worrisome regions of the prostate. “Proceeding directly to a micro-ultrasound study avoids these impediments to discovering whether the patient has clinically significant cancer,” he said.
Micro-ultrasound testing involves a single visit to a urologist whereas MRI requires two trips for the patient — one to the urologist and the other to a radiologist, Klotz said. “It’s one-stop shopping,” he said. “So, the patient has his micro-ultrasound. If there’s a target found, he then does the targeted biopsy.”
Klotz said micro-ultrasound helps patients avoid the expense and health risks of gadolinium in contrast with MRIs.
“I don’t think micro-ultrasound is going to replace MRIs,” he said. “I think they’re somewhat complementary. You get cases where they’re visible on MRI and not visible on micro-ultrasound and vice versa.”
The researchers received a grant from Exact Imaging.
A version of this article appeared at Medscape.com.
Howard Wolinsky is a Chicago-based freelance writer.
Micro-ultrasound–guided biopsies were found for the first time to be “noninferior” to MRI-guided procedures, according to new research presented at the 2025 annual congress of the European Association of Urology.
The OPTIMUM study found 4.5% more clinically significant cancers among men who underwent micro-ultrasound–guided biopsies of the prostate than in those scanned using MRI.
“The take-home message is that men being worked up for an elevated PSA [prostate-specific antigen] or an abnormal digital rectal examination who are at increased risk of prostate cancer may safely undergo a micro-ultrasound–guided biopsy rather than an MRI-guided biopsy,” said Adam Kinnaird, MD, PhD, the Frank and Carla Sojonky Chair in Prostate Cancer Research at the University of Alberta, Edmonton, Alberta, Canada, and principal investigator of the study.
Micro-ultrasound can image to as small as 70 μm, ie, the width of a human hair.
OPTIMUM was an international, open-label, randomized, noninferiority trial in 20 centers in eight countries of men with clinical suspicion of prostate cancer, elevated PSAs, abnormal digital rectal exams, or a combination of these risk factors. None of the men previously had undergone biopsies.
The study had three arms to which men were assigned randomly: Micro-ultrasound–guided biopsy (n = 121); biopsies guided by micro-ultrasound and fusion MRI (n = 226), and MRI plus conventional ultrasound–guided biopsy (n = 331).
Subjects had a median age of 65 years and a median PSA level of 6.9 ng/mL; 83% self-identified as White individuals.
“Micro-ultrasound was found to be no worse than MRI at the detection of clinically significant prostate cancer. We don’t show it is equivalent. We don’t show it better. We show it is not worse,” Kinnaird said.
The study, funded by Exact Imaging, which makes the ExactVu micro-ultrasound platform, appeared simultaneously in JAMA.
Laurence Klotz, MD, the Sunnybrook Chair of Prostate Cancer Research at the University of Toronto Sunnybrook Health Sciences Centre, and senior researcher on the OPTIMUM trial, said as the incidence of prostate cancer rises globally, micro-ultrasound may be of particular value in low-income and middle-income countries where MRI is not widely available.
“It’s extremely appealing in places that can’t offer MRI to everyone, but I think it also will have a role going forward in regions where there is no problem about getting access to MRI,” Klotz told Medscape Medical News.
This group is next studying the financial aspects of the technology, he added.
Gerald Andriole, MD, then urology chief at Washington University in St. Louis, St. Louis, Missouri, designed the original studies of the ExactVu system, which the US Food and Drug Administration approved in 2017.
Andriole, now chief medical officer of Prostatype Genomics, said MRIs are costly, subjective, and uncomfortable for many patients, due to claustrophobia and obesity, requiring complicated co-registration procedures to perform an accurate targeted biopsy into the most worrisome regions of the prostate. “Proceeding directly to a micro-ultrasound study avoids these impediments to discovering whether the patient has clinically significant cancer,” he said.
Micro-ultrasound testing involves a single visit to a urologist whereas MRI requires two trips for the patient — one to the urologist and the other to a radiologist, Klotz said. “It’s one-stop shopping,” he said. “So, the patient has his micro-ultrasound. If there’s a target found, he then does the targeted biopsy.”
Klotz said micro-ultrasound helps patients avoid the expense and health risks of gadolinium in contrast with MRIs.
“I don’t think micro-ultrasound is going to replace MRIs,” he said. “I think they’re somewhat complementary. You get cases where they’re visible on MRI and not visible on micro-ultrasound and vice versa.”
The researchers received a grant from Exact Imaging.
A version of this article appeared at Medscape.com.
Howard Wolinsky is a Chicago-based freelance writer.
Micro-ultrasound–guided biopsies were found for the first time to be “noninferior” to MRI-guided procedures, according to new research presented at the 2025 annual congress of the European Association of Urology.
The OPTIMUM study found 4.5% more clinically significant cancers among men who underwent micro-ultrasound–guided biopsies of the prostate than in those scanned using MRI.
“The take-home message is that men being worked up for an elevated PSA [prostate-specific antigen] or an abnormal digital rectal examination who are at increased risk of prostate cancer may safely undergo a micro-ultrasound–guided biopsy rather than an MRI-guided biopsy,” said Adam Kinnaird, MD, PhD, the Frank and Carla Sojonky Chair in Prostate Cancer Research at the University of Alberta, Edmonton, Alberta, Canada, and principal investigator of the study.
Micro-ultrasound can image to as small as 70 μm, ie, the width of a human hair.
OPTIMUM was an international, open-label, randomized, noninferiority trial in 20 centers in eight countries of men with clinical suspicion of prostate cancer, elevated PSAs, abnormal digital rectal exams, or a combination of these risk factors. None of the men previously had undergone biopsies.
The study had three arms to which men were assigned randomly: Micro-ultrasound–guided biopsy (n = 121); biopsies guided by micro-ultrasound and fusion MRI (n = 226), and MRI plus conventional ultrasound–guided biopsy (n = 331).
Subjects had a median age of 65 years and a median PSA level of 6.9 ng/mL; 83% self-identified as White individuals.
“Micro-ultrasound was found to be no worse than MRI at the detection of clinically significant prostate cancer. We don’t show it is equivalent. We don’t show it better. We show it is not worse,” Kinnaird said.
The study, funded by Exact Imaging, which makes the ExactVu micro-ultrasound platform, appeared simultaneously in JAMA.
Laurence Klotz, MD, the Sunnybrook Chair of Prostate Cancer Research at the University of Toronto Sunnybrook Health Sciences Centre, and senior researcher on the OPTIMUM trial, said as the incidence of prostate cancer rises globally, micro-ultrasound may be of particular value in low-income and middle-income countries where MRI is not widely available.
“It’s extremely appealing in places that can’t offer MRI to everyone, but I think it also will have a role going forward in regions where there is no problem about getting access to MRI,” Klotz told Medscape Medical News.
This group is next studying the financial aspects of the technology, he added.
Gerald Andriole, MD, then urology chief at Washington University in St. Louis, St. Louis, Missouri, designed the original studies of the ExactVu system, which the US Food and Drug Administration approved in 2017.
Andriole, now chief medical officer of Prostatype Genomics, said MRIs are costly, subjective, and uncomfortable for many patients, due to claustrophobia and obesity, requiring complicated co-registration procedures to perform an accurate targeted biopsy into the most worrisome regions of the prostate. “Proceeding directly to a micro-ultrasound study avoids these impediments to discovering whether the patient has clinically significant cancer,” he said.
Micro-ultrasound testing involves a single visit to a urologist whereas MRI requires two trips for the patient — one to the urologist and the other to a radiologist, Klotz said. “It’s one-stop shopping,” he said. “So, the patient has his micro-ultrasound. If there’s a target found, he then does the targeted biopsy.”
Klotz said micro-ultrasound helps patients avoid the expense and health risks of gadolinium in contrast with MRIs.
“I don’t think micro-ultrasound is going to replace MRIs,” he said. “I think they’re somewhat complementary. You get cases where they’re visible on MRI and not visible on micro-ultrasound and vice versa.”
The researchers received a grant from Exact Imaging.
A version of this article appeared at Medscape.com.
Howard Wolinsky is a Chicago-based freelance writer.
Inadequate Grading of Intraductal Carcinoma of the Prostate
BOSTON — Solid intraductal carcinoma of the prostate (IDC-P) is associated with significantly worse outcomes compared with conventional Gleason grade 5 prostate cancers and is more commonly present in metastatic than nonmetastatic cancers, according to two studies presented this week at the United States and Canadian Academy of Pathology (USCAP) 2025 Annual Meeting.
“Our findings suggest that solid IDC-P is more aggressive than Gleason grade 5 conventional prostate adenocarcinoma or cribriform IDC-P,” and it may therefore be better not to consider it as a grade 5 pattern, said first author of one of the studies, Hangchuan Shi, MD, PhD, of the University of Rochester Medical Center, in Rochester, New York.
Although IDC-P — reported in about 20% of men with prostate cancer — is known to be associated with poorer response to treatment, there is a debate over whether to grade the entity with Gleason scoring or not.
The International Society of Urological Pathology recommends incorporating IDC-P into the Gleason score, while the Genitourinary Pathology Society does not.
To evaluate the prognostic significance of solid IDC-P compared with Gleason grade 5 conventional prostate cancer, Shi and his colleagues identified 115 cases in the surgical pathology database at the University of Rochester Medical Center between 2008 and 2015 involving Gleason grade 5 conventional prostatic adenocarcinoma as a primary, secondary, or tertiary pattern, as well as cribriform IDC-P.
The researchers excluded cases showing comedonecrosis within IDC-P, due to the potential for worse outcomes.
Of the grade 5 conventional prostate cancer cases with cribriform carcinoma, 28 (24.3%) had solid nest pattern IDC-P. Patients with and without solid IDC-P had a matching mean age of about 64 years, and their mean preoperative PSA was about 12.27 ng/mL.
Adjuvant therapy prior to recurrence was significantly more common in those who had solid IDC-P (60.7% vs 34.5%; P = .016).
Compared with the conventional prostate cancer cases, those with solid IDC-P had a significantly higher incidence of lymph node metastasis (P = .014) and had larger estimated tumor volume (P = .011).
There were no significant differences in other clinicopathologic features, such as preoperative prostate-specific antigen, grade group, pT stage, and surgical margin status.
After adjustment for key factors in a multivariable analysis, solid IDC-P was significantly associated with poorer recurrence-free survival (P = .007), and poorer cancer-specific survival (P = .004).
Finally, solid IDC-P was an independent predictor of recurrence (hazard ratio [HR] 1.960; P = .031), whereas other measures, including prostate-specific antigen (PSA), cancer grade, pT, lymph node metastasis, surgical and tumor volume were not significant factors.
“We found the solid IDC-P patients had almost two-times the risk of recurrence compared with the patients without solid IDC-P in our study,” Shi said.
The findings underscore the importance of accurately identifying IDC-P, senior author Hiroshi Miyamoto, MD, PhD, director of Genitourinary Pathology at School of Medicine and Dentistry, University of Rochester, Rochester, New York, told Medscape Medical News.
“It may be difficult for some pathologists, especially those who have no specific training in genitourinary pathology, to adequately recognize” this form of cancer, he said.
Although it is recognized as an aggressive form of prostate cancer, “based on our studies, we believe that it is inadequate to grade IDC-P” as a Gleason grade 5 cancer, Miyamoto added.
IDC More Common in Metastases
Poorer outcomes associated with IDC-P were further described in a post hoc sub-analysis of the phase 3, prospective PATRON clinical trial that is evaluating prostate-specific membrane antigen (PSMA) PET-CT–guided intensification of therapy.
In the multicenter trial, 825 patients were stratified into three cohorts: High-risk patients receiving radiation therapy (45%), high-risk patients receiving salvage radiation therapy post-radical prostatectomy (47%), and those receiving a radical prostatectomy (8%).
The patients in all three cohorts were randomized 1:1 to receive imaging with or without PSMA PET-CT.
IDC-P and/or cribriform carcinoma were present among 342 patients in the PSMA PET-CT group including 48% of high-risk patients receiving radiotherapy, 42% of high-risk patients receiving salvage radiation therapy post-radical prostatectomy, and 40% of those receiving a radical prostatectomy.
IDC-P was reported in 64% of cases with metastases detected by PSMA PET-CT compared with just 36% of cases without metastasis (P = .008), with the ratios being similar in each individual patient cohort.
Of note, the association between the presence of IDC-P and metastases was not observed when IDC-P and cribriform carcinoma were combined — IDC-P and/or cribriform carcinoma was detected in 54% of cases with PSMA PET-CT–detectable metastasis and in 46% of cases without metastasis (P = .362).
The first author Dominique Trudel, MD, PhD, of the Centre Hospitalier de l’Université de Montréal, Montreal, Quebec, Canada, said the findings add to understanding of IDC-P’s relationship with poorer outcomes.
“As pathologists, we know that IDC is associated with poor outcomes and that men with IDC who are treated with standard therapies do benefit from them, but they never benefit as much as men without IDC,” she told Medscape Medical News.
As the study is ongoing, “in approximately 4-5 years, we will know how much of a difference IDC-P makes in outcomes after treatment,” Trudel noted.
The take-home message from the collective research should be that “IDC-P matters,” she said.
“I think that if your patient has IDC-P and [cribriform carcinoma], it is worth at least asking someone from an academic center to see what the treatment options are. We know that some radiation oncologists are increasing doses for IDC-P. It is empiric, but they’re doing it,” she explained.
The authors had no disclosures to report.
The article first appeared in Medscape.com.
BOSTON — Solid intraductal carcinoma of the prostate (IDC-P) is associated with significantly worse outcomes compared with conventional Gleason grade 5 prostate cancers and is more commonly present in metastatic than nonmetastatic cancers, according to two studies presented this week at the United States and Canadian Academy of Pathology (USCAP) 2025 Annual Meeting.
“Our findings suggest that solid IDC-P is more aggressive than Gleason grade 5 conventional prostate adenocarcinoma or cribriform IDC-P,” and it may therefore be better not to consider it as a grade 5 pattern, said first author of one of the studies, Hangchuan Shi, MD, PhD, of the University of Rochester Medical Center, in Rochester, New York.
Although IDC-P — reported in about 20% of men with prostate cancer — is known to be associated with poorer response to treatment, there is a debate over whether to grade the entity with Gleason scoring or not.
The International Society of Urological Pathology recommends incorporating IDC-P into the Gleason score, while the Genitourinary Pathology Society does not.
To evaluate the prognostic significance of solid IDC-P compared with Gleason grade 5 conventional prostate cancer, Shi and his colleagues identified 115 cases in the surgical pathology database at the University of Rochester Medical Center between 2008 and 2015 involving Gleason grade 5 conventional prostatic adenocarcinoma as a primary, secondary, or tertiary pattern, as well as cribriform IDC-P.
The researchers excluded cases showing comedonecrosis within IDC-P, due to the potential for worse outcomes.
Of the grade 5 conventional prostate cancer cases with cribriform carcinoma, 28 (24.3%) had solid nest pattern IDC-P. Patients with and without solid IDC-P had a matching mean age of about 64 years, and their mean preoperative PSA was about 12.27 ng/mL.
Adjuvant therapy prior to recurrence was significantly more common in those who had solid IDC-P (60.7% vs 34.5%; P = .016).
Compared with the conventional prostate cancer cases, those with solid IDC-P had a significantly higher incidence of lymph node metastasis (P = .014) and had larger estimated tumor volume (P = .011).
There were no significant differences in other clinicopathologic features, such as preoperative prostate-specific antigen, grade group, pT stage, and surgical margin status.
After adjustment for key factors in a multivariable analysis, solid IDC-P was significantly associated with poorer recurrence-free survival (P = .007), and poorer cancer-specific survival (P = .004).
Finally, solid IDC-P was an independent predictor of recurrence (hazard ratio [HR] 1.960; P = .031), whereas other measures, including prostate-specific antigen (PSA), cancer grade, pT, lymph node metastasis, surgical and tumor volume were not significant factors.
“We found the solid IDC-P patients had almost two-times the risk of recurrence compared with the patients without solid IDC-P in our study,” Shi said.
The findings underscore the importance of accurately identifying IDC-P, senior author Hiroshi Miyamoto, MD, PhD, director of Genitourinary Pathology at School of Medicine and Dentistry, University of Rochester, Rochester, New York, told Medscape Medical News.
“It may be difficult for some pathologists, especially those who have no specific training in genitourinary pathology, to adequately recognize” this form of cancer, he said.
Although it is recognized as an aggressive form of prostate cancer, “based on our studies, we believe that it is inadequate to grade IDC-P” as a Gleason grade 5 cancer, Miyamoto added.
IDC More Common in Metastases
Poorer outcomes associated with IDC-P were further described in a post hoc sub-analysis of the phase 3, prospective PATRON clinical trial that is evaluating prostate-specific membrane antigen (PSMA) PET-CT–guided intensification of therapy.
In the multicenter trial, 825 patients were stratified into three cohorts: High-risk patients receiving radiation therapy (45%), high-risk patients receiving salvage radiation therapy post-radical prostatectomy (47%), and those receiving a radical prostatectomy (8%).
The patients in all three cohorts were randomized 1:1 to receive imaging with or without PSMA PET-CT.
IDC-P and/or cribriform carcinoma were present among 342 patients in the PSMA PET-CT group including 48% of high-risk patients receiving radiotherapy, 42% of high-risk patients receiving salvage radiation therapy post-radical prostatectomy, and 40% of those receiving a radical prostatectomy.
IDC-P was reported in 64% of cases with metastases detected by PSMA PET-CT compared with just 36% of cases without metastasis (P = .008), with the ratios being similar in each individual patient cohort.
Of note, the association between the presence of IDC-P and metastases was not observed when IDC-P and cribriform carcinoma were combined — IDC-P and/or cribriform carcinoma was detected in 54% of cases with PSMA PET-CT–detectable metastasis and in 46% of cases without metastasis (P = .362).
The first author Dominique Trudel, MD, PhD, of the Centre Hospitalier de l’Université de Montréal, Montreal, Quebec, Canada, said the findings add to understanding of IDC-P’s relationship with poorer outcomes.
“As pathologists, we know that IDC is associated with poor outcomes and that men with IDC who are treated with standard therapies do benefit from them, but they never benefit as much as men without IDC,” she told Medscape Medical News.
As the study is ongoing, “in approximately 4-5 years, we will know how much of a difference IDC-P makes in outcomes after treatment,” Trudel noted.
The take-home message from the collective research should be that “IDC-P matters,” she said.
“I think that if your patient has IDC-P and [cribriform carcinoma], it is worth at least asking someone from an academic center to see what the treatment options are. We know that some radiation oncologists are increasing doses for IDC-P. It is empiric, but they’re doing it,” she explained.
The authors had no disclosures to report.
The article first appeared in Medscape.com.
BOSTON — Solid intraductal carcinoma of the prostate (IDC-P) is associated with significantly worse outcomes compared with conventional Gleason grade 5 prostate cancers and is more commonly present in metastatic than nonmetastatic cancers, according to two studies presented this week at the United States and Canadian Academy of Pathology (USCAP) 2025 Annual Meeting.
“Our findings suggest that solid IDC-P is more aggressive than Gleason grade 5 conventional prostate adenocarcinoma or cribriform IDC-P,” and it may therefore be better not to consider it as a grade 5 pattern, said first author of one of the studies, Hangchuan Shi, MD, PhD, of the University of Rochester Medical Center, in Rochester, New York.
Although IDC-P — reported in about 20% of men with prostate cancer — is known to be associated with poorer response to treatment, there is a debate over whether to grade the entity with Gleason scoring or not.
The International Society of Urological Pathology recommends incorporating IDC-P into the Gleason score, while the Genitourinary Pathology Society does not.
To evaluate the prognostic significance of solid IDC-P compared with Gleason grade 5 conventional prostate cancer, Shi and his colleagues identified 115 cases in the surgical pathology database at the University of Rochester Medical Center between 2008 and 2015 involving Gleason grade 5 conventional prostatic adenocarcinoma as a primary, secondary, or tertiary pattern, as well as cribriform IDC-P.
The researchers excluded cases showing comedonecrosis within IDC-P, due to the potential for worse outcomes.
Of the grade 5 conventional prostate cancer cases with cribriform carcinoma, 28 (24.3%) had solid nest pattern IDC-P. Patients with and without solid IDC-P had a matching mean age of about 64 years, and their mean preoperative PSA was about 12.27 ng/mL.
Adjuvant therapy prior to recurrence was significantly more common in those who had solid IDC-P (60.7% vs 34.5%; P = .016).
Compared with the conventional prostate cancer cases, those with solid IDC-P had a significantly higher incidence of lymph node metastasis (P = .014) and had larger estimated tumor volume (P = .011).
There were no significant differences in other clinicopathologic features, such as preoperative prostate-specific antigen, grade group, pT stage, and surgical margin status.
After adjustment for key factors in a multivariable analysis, solid IDC-P was significantly associated with poorer recurrence-free survival (P = .007), and poorer cancer-specific survival (P = .004).
Finally, solid IDC-P was an independent predictor of recurrence (hazard ratio [HR] 1.960; P = .031), whereas other measures, including prostate-specific antigen (PSA), cancer grade, pT, lymph node metastasis, surgical and tumor volume were not significant factors.
“We found the solid IDC-P patients had almost two-times the risk of recurrence compared with the patients without solid IDC-P in our study,” Shi said.
The findings underscore the importance of accurately identifying IDC-P, senior author Hiroshi Miyamoto, MD, PhD, director of Genitourinary Pathology at School of Medicine and Dentistry, University of Rochester, Rochester, New York, told Medscape Medical News.
“It may be difficult for some pathologists, especially those who have no specific training in genitourinary pathology, to adequately recognize” this form of cancer, he said.
Although it is recognized as an aggressive form of prostate cancer, “based on our studies, we believe that it is inadequate to grade IDC-P” as a Gleason grade 5 cancer, Miyamoto added.
IDC More Common in Metastases
Poorer outcomes associated with IDC-P were further described in a post hoc sub-analysis of the phase 3, prospective PATRON clinical trial that is evaluating prostate-specific membrane antigen (PSMA) PET-CT–guided intensification of therapy.
In the multicenter trial, 825 patients were stratified into three cohorts: High-risk patients receiving radiation therapy (45%), high-risk patients receiving salvage radiation therapy post-radical prostatectomy (47%), and those receiving a radical prostatectomy (8%).
The patients in all three cohorts were randomized 1:1 to receive imaging with or without PSMA PET-CT.
IDC-P and/or cribriform carcinoma were present among 342 patients in the PSMA PET-CT group including 48% of high-risk patients receiving radiotherapy, 42% of high-risk patients receiving salvage radiation therapy post-radical prostatectomy, and 40% of those receiving a radical prostatectomy.
IDC-P was reported in 64% of cases with metastases detected by PSMA PET-CT compared with just 36% of cases without metastasis (P = .008), with the ratios being similar in each individual patient cohort.
Of note, the association between the presence of IDC-P and metastases was not observed when IDC-P and cribriform carcinoma were combined — IDC-P and/or cribriform carcinoma was detected in 54% of cases with PSMA PET-CT–detectable metastasis and in 46% of cases without metastasis (P = .362).
The first author Dominique Trudel, MD, PhD, of the Centre Hospitalier de l’Université de Montréal, Montreal, Quebec, Canada, said the findings add to understanding of IDC-P’s relationship with poorer outcomes.
“As pathologists, we know that IDC is associated with poor outcomes and that men with IDC who are treated with standard therapies do benefit from them, but they never benefit as much as men without IDC,” she told Medscape Medical News.
As the study is ongoing, “in approximately 4-5 years, we will know how much of a difference IDC-P makes in outcomes after treatment,” Trudel noted.
The take-home message from the collective research should be that “IDC-P matters,” she said.
“I think that if your patient has IDC-P and [cribriform carcinoma], it is worth at least asking someone from an academic center to see what the treatment options are. We know that some radiation oncologists are increasing doses for IDC-P. It is empiric, but they’re doing it,” she explained.
The authors had no disclosures to report.
The article first appeared in Medscape.com.
PSA Screening in VA Patients After Age 70 Years
TOPLINE: Most men receiving care through the Veterans Health Administration (VHA) continue prostate-specific antigen (PSA) screening after aged 70 years despite low absolute risk for prostate cancer-specific mortality (PCSM), even among Black men in the healthiest quintile.
METHODOLOGY:
Researchers conducted a cohort study of 921,609 men aged 70 years receiving VHA care between 2008 and 2020, who had normal screening PSA values (< 4 ng/mL) between ages 65-69 years.
- Analysis included electronic health record data from VHA Corporate Data Warehouse, linked Medicare claims data, and VHA community care data.
- Investigators examined the value of PSA levels, race and ethnicity, and competing mortality in risk stratification for PCSM and mPCa using regression modeling.
TAKEAWAY:
The 10-year cumulative incidence of PCSM was 0.26% overall, with 95% of men having a 10-year risk < 0.73%, and higher baseline PSA levels associated with increased risk (0.79% for 3.00-3.99 ng/mL vs 0.10% for 0.20-0.99 ng/mL).
- Race and ethnicity showed modest association with PCSM risk: Black patients had a 0.79% risk of mPCa vs 0.38% for White patients. The risk of PCSM was 0.36% for Black patients vs 0.25% for White patients.
- Most patients (87%) continued PSA screening after age 70 years, with little variation by competing mortality risk or race and ethnicity.
- Low PSA (0.20-0.99 ng/mL) identified very low-risk populations with < 1% 10-year risk for prostate biopsy, clinically significant prostate cancer diagnosis, and treatment.
IN PRACTICE: "Our data suggest that a simple assessment of personal risk based on PSA values before age 70 years captures a large proportion of relevant prognostic information with respect to mPCa and PCSM risk ... Low PSA (0.20-0.99 ng/mL) was associated with very low PCSM and mPCa risk, even among the healthiest Black men," wrote the authors of the study.
SOURCE: The study was led by Alex K. Bryant,MD, MAS and the Veterans Affairs Center for Clinical Management Research in Ann Arbor. It was published online on February 14 in JAMA Network Open.
LIMITATIONS: According to the authors, any potential PCSM or mPCa reduction from continued PSA screening > age 70 years remains unproven due to lack of randomized trial data. The study relied on death certificates to define PCSM, which may have introduced misclassification error. Family history of prostate cancer was not included due to unreliable electronic medical record data availability. Additionally, veterans have higher comorbidity burdens than the general population and unique military-related environmental exposures, potentially limiting result generalizability.
DISCLOSURES: The study was supported by grants U01CA253915, PSOCA097186, R35CA274442, and R50CA221836 from the National Cancer Institute. Matthew J. Schipper, MD, reported receiving consulting fees from Innovative Analytics. Phoebe A. Tsao, MD, disclosed receiving grants from the Prostate Cancer Foundation outside the submitted work. Kristian D. Stensland, MD, reported receiving a grant from the National Institutes of Health during the conduct of the study.
TOPLINE: Most men receiving care through the Veterans Health Administration (VHA) continue prostate-specific antigen (PSA) screening after aged 70 years despite low absolute risk for prostate cancer-specific mortality (PCSM), even among Black men in the healthiest quintile.
METHODOLOGY:
Researchers conducted a cohort study of 921,609 men aged 70 years receiving VHA care between 2008 and 2020, who had normal screening PSA values (< 4 ng/mL) between ages 65-69 years.
- Analysis included electronic health record data from VHA Corporate Data Warehouse, linked Medicare claims data, and VHA community care data.
- Investigators examined the value of PSA levels, race and ethnicity, and competing mortality in risk stratification for PCSM and mPCa using regression modeling.
TAKEAWAY:
The 10-year cumulative incidence of PCSM was 0.26% overall, with 95% of men having a 10-year risk < 0.73%, and higher baseline PSA levels associated with increased risk (0.79% for 3.00-3.99 ng/mL vs 0.10% for 0.20-0.99 ng/mL).
- Race and ethnicity showed modest association with PCSM risk: Black patients had a 0.79% risk of mPCa vs 0.38% for White patients. The risk of PCSM was 0.36% for Black patients vs 0.25% for White patients.
- Most patients (87%) continued PSA screening after age 70 years, with little variation by competing mortality risk or race and ethnicity.
- Low PSA (0.20-0.99 ng/mL) identified very low-risk populations with < 1% 10-year risk for prostate biopsy, clinically significant prostate cancer diagnosis, and treatment.
IN PRACTICE: "Our data suggest that a simple assessment of personal risk based on PSA values before age 70 years captures a large proportion of relevant prognostic information with respect to mPCa and PCSM risk ... Low PSA (0.20-0.99 ng/mL) was associated with very low PCSM and mPCa risk, even among the healthiest Black men," wrote the authors of the study.
SOURCE: The study was led by Alex K. Bryant,MD, MAS and the Veterans Affairs Center for Clinical Management Research in Ann Arbor. It was published online on February 14 in JAMA Network Open.
LIMITATIONS: According to the authors, any potential PCSM or mPCa reduction from continued PSA screening > age 70 years remains unproven due to lack of randomized trial data. The study relied on death certificates to define PCSM, which may have introduced misclassification error. Family history of prostate cancer was not included due to unreliable electronic medical record data availability. Additionally, veterans have higher comorbidity burdens than the general population and unique military-related environmental exposures, potentially limiting result generalizability.
DISCLOSURES: The study was supported by grants U01CA253915, PSOCA097186, R35CA274442, and R50CA221836 from the National Cancer Institute. Matthew J. Schipper, MD, reported receiving consulting fees from Innovative Analytics. Phoebe A. Tsao, MD, disclosed receiving grants from the Prostate Cancer Foundation outside the submitted work. Kristian D. Stensland, MD, reported receiving a grant from the National Institutes of Health during the conduct of the study.
TOPLINE: Most men receiving care through the Veterans Health Administration (VHA) continue prostate-specific antigen (PSA) screening after aged 70 years despite low absolute risk for prostate cancer-specific mortality (PCSM), even among Black men in the healthiest quintile.
METHODOLOGY:
Researchers conducted a cohort study of 921,609 men aged 70 years receiving VHA care between 2008 and 2020, who had normal screening PSA values (< 4 ng/mL) between ages 65-69 years.
- Analysis included electronic health record data from VHA Corporate Data Warehouse, linked Medicare claims data, and VHA community care data.
- Investigators examined the value of PSA levels, race and ethnicity, and competing mortality in risk stratification for PCSM and mPCa using regression modeling.
TAKEAWAY:
The 10-year cumulative incidence of PCSM was 0.26% overall, with 95% of men having a 10-year risk < 0.73%, and higher baseline PSA levels associated with increased risk (0.79% for 3.00-3.99 ng/mL vs 0.10% for 0.20-0.99 ng/mL).
- Race and ethnicity showed modest association with PCSM risk: Black patients had a 0.79% risk of mPCa vs 0.38% for White patients. The risk of PCSM was 0.36% for Black patients vs 0.25% for White patients.
- Most patients (87%) continued PSA screening after age 70 years, with little variation by competing mortality risk or race and ethnicity.
- Low PSA (0.20-0.99 ng/mL) identified very low-risk populations with < 1% 10-year risk for prostate biopsy, clinically significant prostate cancer diagnosis, and treatment.
IN PRACTICE: "Our data suggest that a simple assessment of personal risk based on PSA values before age 70 years captures a large proportion of relevant prognostic information with respect to mPCa and PCSM risk ... Low PSA (0.20-0.99 ng/mL) was associated with very low PCSM and mPCa risk, even among the healthiest Black men," wrote the authors of the study.
SOURCE: The study was led by Alex K. Bryant,MD, MAS and the Veterans Affairs Center for Clinical Management Research in Ann Arbor. It was published online on February 14 in JAMA Network Open.
LIMITATIONS: According to the authors, any potential PCSM or mPCa reduction from continued PSA screening > age 70 years remains unproven due to lack of randomized trial data. The study relied on death certificates to define PCSM, which may have introduced misclassification error. Family history of prostate cancer was not included due to unreliable electronic medical record data availability. Additionally, veterans have higher comorbidity burdens than the general population and unique military-related environmental exposures, potentially limiting result generalizability.
DISCLOSURES: The study was supported by grants U01CA253915, PSOCA097186, R35CA274442, and R50CA221836 from the National Cancer Institute. Matthew J. Schipper, MD, reported receiving consulting fees from Innovative Analytics. Phoebe A. Tsao, MD, disclosed receiving grants from the Prostate Cancer Foundation outside the submitted work. Kristian D. Stensland, MD, reported receiving a grant from the National Institutes of Health during the conduct of the study.
No Racial Disparities in CVD Outcomes For VA Patients with Prostate Cancer Receiving ADT
TOPLINE: Veterans treated in the Veterans Health Administration (VHA) who had preexisting cardiometabolic disease and received androgen deprivation therapy (ADT) with radiation therapy developed major adverse cardiovascular events (MACE) at 4 times the rate compared to those without cardiometabolic disease. Black and White veterans showed similar cardiovascular outcomes regardless of treatment type.
METHODOLOGY:
Researchers conducted a retrospective cohort study examining 39,580 veterans in the VHA system diagnosed with prostate cancer between 2000 and 2015, following them for a median of 9.6 years to assess time to MACE diagnosis.
- Analysis utilized a 1:1 propensity score matching process to compare outcomes between treatment types (ADT with radiation therapy vs radiation therapy alone) and racial groups (Black vs White men).
- Participants had a mean age of 65.9 years at diagnosis; 68% identified as White and 32% as Black, and 83% had stage 2 disease classified with 43.1% intermediate risk. Most lived in nonrural zip codes
- Primary outcome measure was time to MACE, defined as a composite of cardiovascular death, myocardial infarction, or ischemic stroke, with patients censored at non-cardiovascular death or study end.
TAKEAWAY:
Compared to those without CMD, the hazard ratio (HR) for MACE for men with preexisting CMD who received ADT was 4.2. Those receiving radiation alone had an HR of 2.5.
- Patients diagnosed between 2010 and 2015 showed significantly lower MACE rates compared to those diagnosed in 2000 to 2005: HR, 0.23; 95% CI, 0.08-0.71 for White patients; and HR, 0.23; 95% CI, 0.07-0.77 for Black patients.
- Multiple comorbidities were associated with doubled MACE risk (HR, 2.22; 95% CI, 1.08-4.59) compared to those without comorbidities.
- No significant differences in MACE rates were observed between Black and White veterans, regardless of treatment type.
IN PRACTICE: “Within the VHA, men treated with ADT + radiation therapy for prostate cancer do not appear to be at greater risk for MACE than those receiving radiation therapy alone. Black men have similar risk of MACE as White men, whether receiving radiation therapy alone or in combination with ADT," the authors wrote.
SOURCE: The study was led by Alexander R. Lucas, Virginia Commonwealth University School of Public Health in Richmond. It was published online on February 6 in Cardio-Oncology.
LIMITATIONS: According to the authors, the retrospective nature of the data may have limited their ability to detect MACE events occurring outside the VHA. Additionally, the study was limited to men who initiated ADT prior to radiation therapy, excluding those who had surgery or radiation before ADT. The researchers also note that the analysis did not compare outcomes between different types of ADT treatments, such as GnRH agonists vs antagonists, which may have different cardiovascular risk profiles.
DISCLOSURES: Alexander R. Lucas’s work was partly funded by grants 1KO1HL161419 and NRG FP00019789. Ashit K. Paul disclosed receiving honorarium for serving on scientific consultancy panels of SANOFI-Genzyme, Bayer, and Tempus & Cardinal Health. Additional disclosures are noted but not specified in the article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication
TOPLINE: Veterans treated in the Veterans Health Administration (VHA) who had preexisting cardiometabolic disease and received androgen deprivation therapy (ADT) with radiation therapy developed major adverse cardiovascular events (MACE) at 4 times the rate compared to those without cardiometabolic disease. Black and White veterans showed similar cardiovascular outcomes regardless of treatment type.
METHODOLOGY:
Researchers conducted a retrospective cohort study examining 39,580 veterans in the VHA system diagnosed with prostate cancer between 2000 and 2015, following them for a median of 9.6 years to assess time to MACE diagnosis.
- Analysis utilized a 1:1 propensity score matching process to compare outcomes between treatment types (ADT with radiation therapy vs radiation therapy alone) and racial groups (Black vs White men).
- Participants had a mean age of 65.9 years at diagnosis; 68% identified as White and 32% as Black, and 83% had stage 2 disease classified with 43.1% intermediate risk. Most lived in nonrural zip codes
- Primary outcome measure was time to MACE, defined as a composite of cardiovascular death, myocardial infarction, or ischemic stroke, with patients censored at non-cardiovascular death or study end.
TAKEAWAY:
Compared to those without CMD, the hazard ratio (HR) for MACE for men with preexisting CMD who received ADT was 4.2. Those receiving radiation alone had an HR of 2.5.
- Patients diagnosed between 2010 and 2015 showed significantly lower MACE rates compared to those diagnosed in 2000 to 2005: HR, 0.23; 95% CI, 0.08-0.71 for White patients; and HR, 0.23; 95% CI, 0.07-0.77 for Black patients.
- Multiple comorbidities were associated with doubled MACE risk (HR, 2.22; 95% CI, 1.08-4.59) compared to those without comorbidities.
- No significant differences in MACE rates were observed between Black and White veterans, regardless of treatment type.
IN PRACTICE: “Within the VHA, men treated with ADT + radiation therapy for prostate cancer do not appear to be at greater risk for MACE than those receiving radiation therapy alone. Black men have similar risk of MACE as White men, whether receiving radiation therapy alone or in combination with ADT," the authors wrote.
SOURCE: The study was led by Alexander R. Lucas, Virginia Commonwealth University School of Public Health in Richmond. It was published online on February 6 in Cardio-Oncology.
LIMITATIONS: According to the authors, the retrospective nature of the data may have limited their ability to detect MACE events occurring outside the VHA. Additionally, the study was limited to men who initiated ADT prior to radiation therapy, excluding those who had surgery or radiation before ADT. The researchers also note that the analysis did not compare outcomes between different types of ADT treatments, such as GnRH agonists vs antagonists, which may have different cardiovascular risk profiles.
DISCLOSURES: Alexander R. Lucas’s work was partly funded by grants 1KO1HL161419 and NRG FP00019789. Ashit K. Paul disclosed receiving honorarium for serving on scientific consultancy panels of SANOFI-Genzyme, Bayer, and Tempus & Cardinal Health. Additional disclosures are noted but not specified in the article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication
TOPLINE: Veterans treated in the Veterans Health Administration (VHA) who had preexisting cardiometabolic disease and received androgen deprivation therapy (ADT) with radiation therapy developed major adverse cardiovascular events (MACE) at 4 times the rate compared to those without cardiometabolic disease. Black and White veterans showed similar cardiovascular outcomes regardless of treatment type.
METHODOLOGY:
Researchers conducted a retrospective cohort study examining 39,580 veterans in the VHA system diagnosed with prostate cancer between 2000 and 2015, following them for a median of 9.6 years to assess time to MACE diagnosis.
- Analysis utilized a 1:1 propensity score matching process to compare outcomes between treatment types (ADT with radiation therapy vs radiation therapy alone) and racial groups (Black vs White men).
- Participants had a mean age of 65.9 years at diagnosis; 68% identified as White and 32% as Black, and 83% had stage 2 disease classified with 43.1% intermediate risk. Most lived in nonrural zip codes
- Primary outcome measure was time to MACE, defined as a composite of cardiovascular death, myocardial infarction, or ischemic stroke, with patients censored at non-cardiovascular death or study end.
TAKEAWAY:
Compared to those without CMD, the hazard ratio (HR) for MACE for men with preexisting CMD who received ADT was 4.2. Those receiving radiation alone had an HR of 2.5.
- Patients diagnosed between 2010 and 2015 showed significantly lower MACE rates compared to those diagnosed in 2000 to 2005: HR, 0.23; 95% CI, 0.08-0.71 for White patients; and HR, 0.23; 95% CI, 0.07-0.77 for Black patients.
- Multiple comorbidities were associated with doubled MACE risk (HR, 2.22; 95% CI, 1.08-4.59) compared to those without comorbidities.
- No significant differences in MACE rates were observed between Black and White veterans, regardless of treatment type.
IN PRACTICE: “Within the VHA, men treated with ADT + radiation therapy for prostate cancer do not appear to be at greater risk for MACE than those receiving radiation therapy alone. Black men have similar risk of MACE as White men, whether receiving radiation therapy alone or in combination with ADT," the authors wrote.
SOURCE: The study was led by Alexander R. Lucas, Virginia Commonwealth University School of Public Health in Richmond. It was published online on February 6 in Cardio-Oncology.
LIMITATIONS: According to the authors, the retrospective nature of the data may have limited their ability to detect MACE events occurring outside the VHA. Additionally, the study was limited to men who initiated ADT prior to radiation therapy, excluding those who had surgery or radiation before ADT. The researchers also note that the analysis did not compare outcomes between different types of ADT treatments, such as GnRH agonists vs antagonists, which may have different cardiovascular risk profiles.
DISCLOSURES: Alexander R. Lucas’s work was partly funded by grants 1KO1HL161419 and NRG FP00019789. Ashit K. Paul disclosed receiving honorarium for serving on scientific consultancy panels of SANOFI-Genzyme, Bayer, and Tempus & Cardinal Health. Additional disclosures are noted but not specified in the article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication