User login
‘Down to my last diaper’: The anxiety of parenting in poverty
For parents living in poverty, “diaper math” is a familiar and distressingly pressing daily calculation. Babies in the U.S. go through 6-10 disposable diapers a day, at an average cost of $70-$80 a month. Name-brand diapers with high-end absorption sell for as much as a half a dollar each, and can result in upwards of $120 a month in expenses.
One in every three American families cannot afford enough diapers to keep their infants and toddlers clean, dry, and healthy, according to the National Diaper Bank Network. For many parents, that leads to wrenching choices: diapers, food, or rent?
The COVID-19 pandemic has exacerbated the situation, both by expanding unemployment rolls and by causing supply chain disruptions that have triggered higher prices for a multitude of products, including diapers. Diaper banks – community-funded programs that offer free diapers to low-income families – distributed 86% more diapers on average in 2020 than in 2019, according to the National Diaper Bank Network. In some locations, distribution increased by as much as 800%.
Yet no federal program helps parents pay for this childhood essential. The government’s food assistance program does not cover diapers, nor do most state-level public aid programs.
California is the only state to directly fund diapers for families, but support is limited. CalWORKS, a financial assistance program for families with children, provides $30 per month to help families pay for diapers for children under age 3. Federal policy shifts also may be in the works: Democratic lawmakers are pushing to include $200 million for diaper distribution in the massive budget reconciliation package.
Without adequate resources, low-income parents are left scrambling for ways to get the most use out of each diaper. This stressful undertaking is the subject of a recent article in American Sociological Review by Jennifer Randles, PhD, professor of sociology at California State University–Fresno. In 2018, Randles conducted phone interviews with 70 mothers in California over nine months. She tried to recruit fathers as well, but only two men responded.
Dr. Randles spoke with KHN’s Jenny Gold about how the cost of diapers weighs on low-income moms, and the “inventive mothering” many low-income women adopt to shield their children from the harms of poverty. The conversation has been edited for length and clarity.
Q: How do diapers play into day-to-day anxieties for low-income mothers?
In my sample, half of the mothers told me that they worried more about diapers than they worried about food or housing.
I started to ask mothers, “Can you tell me how many diapers you have on hand right now?” Almost every one told me with exact specificity how many they had – 5 or 7 or 12. And they knew exactly how long that number of diapers would last, based on how often their children defecated and urinated, if their kid was sick, if they had a diaper rash at the time. So just all the emotional and cognitive labor that goes into keeping such careful track of diaper supplies.
They were worrying and figuring out, “OK, I’m down to almost my last diaper. What do I do now? Do I go find some cans [to sell]? Do I go sell some things in my house? Who in my social network might have some extra cash right now?” I talked to moms who sell blood plasma just to get their infants diapers.
Q: What coping strategies stood out to you?
Those of us who study diapers often call them diaper-stretching strategies. One was leaving on a diaper a little bit longer than someone might otherwise leave it on and letting it get completely full. Some mothers figured out if they bought a [more expensive] diaper that held more and leaked less, they could leave the diaper on longer.
They would also do things like letting the baby go diaperless, especially when they were at home and felt like they wouldn’t be judged for letting their baby go without a diaper. And they used every household good you can imagine to make makeshift diapers. Mothers are using cloth, sheets, and pillowcases. They’re using things that are disposable like paper towels with duct tape. They’re making diapers out their own period supplies or adult incontinence supplies when they can get a sample.
One of the questions I often get is, “Why don’t they just use cloth?” A lot of the mothers that I spoke with had tried cloth diapers and they found that they were very cost- and labor-prohibitive. If you pay for a full startup set of cloth diapers, you’re looking at anywhere from $500 to $1,000. And these moms never had that much money. Most of them didn’t have in-home washers and dryers. Some of them didn’t even have homes or consistent access to water, and it’s illegal in a lot of laundromats and public laundry facilities to wash your old diapers. So the same conditions that would prevent moms from being able to readily afford disposable diapers are the same conditions that keep them from being able to use cloth.
Q: You found that, for many women, the concept of being a good mother is wrapped up in diapering. Why is that?
Diapers and managing diapers was so fundamental to their identity as good moms. Most of the mothers in my sample went without their own food. They weren’t paying a cellphone bill or buying their own medicine or their own menstrual supplies, as a way of saving diaper money.
I talked to a lot of moms who said, when your baby is hungry, that’s horrible. Obviously, you do everything to prevent that. But there’s something about a diaper that covers this vulnerable part of a very young baby’s body, this very delicate skin. And being able to do something to meet this human need that we all have, and to maintain dignity and cleanliness.
A lot of the moms had been through the welfare system, and so they’re living in this constant fear [of losing their children]. This is especially true among mothers of color, who are much more likely to get wrapped up in the child welfare system. People can’t necessarily see when your baby’s hungry. But people can see a saggy diaper. That’s going to be one of the things that tags you as a bad mom.
Q: Was your work on diapers influenced by your experience as a parent?
When I was doing these interviews, my daughter was about 2 or 3. So still in diapers. When my daughter peed during a diaper change, I thought, “Oh, I can just toss that one. Here, let me get another clean one.” That’s a really easy choice. For me. That’s a crisis for the mothers I interviewed. Many of them told me they have an anxiety attack with every diaper change.
Q: Do you see a clear policy solution to diaper stress?
What’s kind of ironic is how much physical, emotional, and cognitive labor goes into managing something that society and lawmakers don’t even recognize. Diapers are still not really recognized as a basic need, as evidenced by the fact that they’re still taxed in 35 states.
I think what California is doing is an excellent start. And I think diaper banks are a fabulous type of community-based organization that are filling a huge need that is not being filled by safety net policies. So, public support for diaper banks.
The direct cash aid part of the social safety net has been all but dismantled in the last 25 years. California is pretty generous. But there are some states where just the cost of diapers alone would use almost half of the average state TANF [Temporary Assistance for Needy Families] benefit for a family of three. I think we really do have to address the fact that the value of cash aid buys so much less than it used to.
Q: Your body of work on marriage and families is fascinating and unusual. Is there a single animating question behind your research?
The common thread is: How do our safety net policies support low-income families’ parenting goals? And do they equalize the conditions of parenting? I think of it as a reproductive justice issue. The ability to have a child or to not have a child, and then to parent that child in conditions where the child’s basic needs are met.
We like to say that we’re child and family friendly. The diaper issue is just one of many, many issues where we don’t really put our money or our policies where our mouth is, in terms of supporting families and supporting children. I think my work is trying to get people to think more collectively about having a social responsibility to all families and to each other. No country, but especially the richest country on the planet, should have one in three very young children not having one of their basic needs met.
I interviewed one dad who was incarcerated because he wrote a bad check. And as he described it to me, he had a certain amount of money, and they needed both diapers and milk for the baby. And I’ll never forget, he said, “I didn’t make a good choice, but I made the right one.”
These are not fancy shoes. These are not name-brand clothes. This was a dad needing both milk and diapers. I don’t think it gets much more basic than that.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
For parents living in poverty, “diaper math” is a familiar and distressingly pressing daily calculation. Babies in the U.S. go through 6-10 disposable diapers a day, at an average cost of $70-$80 a month. Name-brand diapers with high-end absorption sell for as much as a half a dollar each, and can result in upwards of $120 a month in expenses.
One in every three American families cannot afford enough diapers to keep their infants and toddlers clean, dry, and healthy, according to the National Diaper Bank Network. For many parents, that leads to wrenching choices: diapers, food, or rent?
The COVID-19 pandemic has exacerbated the situation, both by expanding unemployment rolls and by causing supply chain disruptions that have triggered higher prices for a multitude of products, including diapers. Diaper banks – community-funded programs that offer free diapers to low-income families – distributed 86% more diapers on average in 2020 than in 2019, according to the National Diaper Bank Network. In some locations, distribution increased by as much as 800%.
Yet no federal program helps parents pay for this childhood essential. The government’s food assistance program does not cover diapers, nor do most state-level public aid programs.
California is the only state to directly fund diapers for families, but support is limited. CalWORKS, a financial assistance program for families with children, provides $30 per month to help families pay for diapers for children under age 3. Federal policy shifts also may be in the works: Democratic lawmakers are pushing to include $200 million for diaper distribution in the massive budget reconciliation package.
Without adequate resources, low-income parents are left scrambling for ways to get the most use out of each diaper. This stressful undertaking is the subject of a recent article in American Sociological Review by Jennifer Randles, PhD, professor of sociology at California State University–Fresno. In 2018, Randles conducted phone interviews with 70 mothers in California over nine months. She tried to recruit fathers as well, but only two men responded.
Dr. Randles spoke with KHN’s Jenny Gold about how the cost of diapers weighs on low-income moms, and the “inventive mothering” many low-income women adopt to shield their children from the harms of poverty. The conversation has been edited for length and clarity.
Q: How do diapers play into day-to-day anxieties for low-income mothers?
In my sample, half of the mothers told me that they worried more about diapers than they worried about food or housing.
I started to ask mothers, “Can you tell me how many diapers you have on hand right now?” Almost every one told me with exact specificity how many they had – 5 or 7 or 12. And they knew exactly how long that number of diapers would last, based on how often their children defecated and urinated, if their kid was sick, if they had a diaper rash at the time. So just all the emotional and cognitive labor that goes into keeping such careful track of diaper supplies.
They were worrying and figuring out, “OK, I’m down to almost my last diaper. What do I do now? Do I go find some cans [to sell]? Do I go sell some things in my house? Who in my social network might have some extra cash right now?” I talked to moms who sell blood plasma just to get their infants diapers.
Q: What coping strategies stood out to you?
Those of us who study diapers often call them diaper-stretching strategies. One was leaving on a diaper a little bit longer than someone might otherwise leave it on and letting it get completely full. Some mothers figured out if they bought a [more expensive] diaper that held more and leaked less, they could leave the diaper on longer.
They would also do things like letting the baby go diaperless, especially when they were at home and felt like they wouldn’t be judged for letting their baby go without a diaper. And they used every household good you can imagine to make makeshift diapers. Mothers are using cloth, sheets, and pillowcases. They’re using things that are disposable like paper towels with duct tape. They’re making diapers out their own period supplies or adult incontinence supplies when they can get a sample.
One of the questions I often get is, “Why don’t they just use cloth?” A lot of the mothers that I spoke with had tried cloth diapers and they found that they were very cost- and labor-prohibitive. If you pay for a full startup set of cloth diapers, you’re looking at anywhere from $500 to $1,000. And these moms never had that much money. Most of them didn’t have in-home washers and dryers. Some of them didn’t even have homes or consistent access to water, and it’s illegal in a lot of laundromats and public laundry facilities to wash your old diapers. So the same conditions that would prevent moms from being able to readily afford disposable diapers are the same conditions that keep them from being able to use cloth.
Q: You found that, for many women, the concept of being a good mother is wrapped up in diapering. Why is that?
Diapers and managing diapers was so fundamental to their identity as good moms. Most of the mothers in my sample went without their own food. They weren’t paying a cellphone bill or buying their own medicine or their own menstrual supplies, as a way of saving diaper money.
I talked to a lot of moms who said, when your baby is hungry, that’s horrible. Obviously, you do everything to prevent that. But there’s something about a diaper that covers this vulnerable part of a very young baby’s body, this very delicate skin. And being able to do something to meet this human need that we all have, and to maintain dignity and cleanliness.
A lot of the moms had been through the welfare system, and so they’re living in this constant fear [of losing their children]. This is especially true among mothers of color, who are much more likely to get wrapped up in the child welfare system. People can’t necessarily see when your baby’s hungry. But people can see a saggy diaper. That’s going to be one of the things that tags you as a bad mom.
Q: Was your work on diapers influenced by your experience as a parent?
When I was doing these interviews, my daughter was about 2 or 3. So still in diapers. When my daughter peed during a diaper change, I thought, “Oh, I can just toss that one. Here, let me get another clean one.” That’s a really easy choice. For me. That’s a crisis for the mothers I interviewed. Many of them told me they have an anxiety attack with every diaper change.
Q: Do you see a clear policy solution to diaper stress?
What’s kind of ironic is how much physical, emotional, and cognitive labor goes into managing something that society and lawmakers don’t even recognize. Diapers are still not really recognized as a basic need, as evidenced by the fact that they’re still taxed in 35 states.
I think what California is doing is an excellent start. And I think diaper banks are a fabulous type of community-based organization that are filling a huge need that is not being filled by safety net policies. So, public support for diaper banks.
The direct cash aid part of the social safety net has been all but dismantled in the last 25 years. California is pretty generous. But there are some states where just the cost of diapers alone would use almost half of the average state TANF [Temporary Assistance for Needy Families] benefit for a family of three. I think we really do have to address the fact that the value of cash aid buys so much less than it used to.
Q: Your body of work on marriage and families is fascinating and unusual. Is there a single animating question behind your research?
The common thread is: How do our safety net policies support low-income families’ parenting goals? And do they equalize the conditions of parenting? I think of it as a reproductive justice issue. The ability to have a child or to not have a child, and then to parent that child in conditions where the child’s basic needs are met.
We like to say that we’re child and family friendly. The diaper issue is just one of many, many issues where we don’t really put our money or our policies where our mouth is, in terms of supporting families and supporting children. I think my work is trying to get people to think more collectively about having a social responsibility to all families and to each other. No country, but especially the richest country on the planet, should have one in three very young children not having one of their basic needs met.
I interviewed one dad who was incarcerated because he wrote a bad check. And as he described it to me, he had a certain amount of money, and they needed both diapers and milk for the baby. And I’ll never forget, he said, “I didn’t make a good choice, but I made the right one.”
These are not fancy shoes. These are not name-brand clothes. This was a dad needing both milk and diapers. I don’t think it gets much more basic than that.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
For parents living in poverty, “diaper math” is a familiar and distressingly pressing daily calculation. Babies in the U.S. go through 6-10 disposable diapers a day, at an average cost of $70-$80 a month. Name-brand diapers with high-end absorption sell for as much as a half a dollar each, and can result in upwards of $120 a month in expenses.
One in every three American families cannot afford enough diapers to keep their infants and toddlers clean, dry, and healthy, according to the National Diaper Bank Network. For many parents, that leads to wrenching choices: diapers, food, or rent?
The COVID-19 pandemic has exacerbated the situation, both by expanding unemployment rolls and by causing supply chain disruptions that have triggered higher prices for a multitude of products, including diapers. Diaper banks – community-funded programs that offer free diapers to low-income families – distributed 86% more diapers on average in 2020 than in 2019, according to the National Diaper Bank Network. In some locations, distribution increased by as much as 800%.
Yet no federal program helps parents pay for this childhood essential. The government’s food assistance program does not cover diapers, nor do most state-level public aid programs.
California is the only state to directly fund diapers for families, but support is limited. CalWORKS, a financial assistance program for families with children, provides $30 per month to help families pay for diapers for children under age 3. Federal policy shifts also may be in the works: Democratic lawmakers are pushing to include $200 million for diaper distribution in the massive budget reconciliation package.
Without adequate resources, low-income parents are left scrambling for ways to get the most use out of each diaper. This stressful undertaking is the subject of a recent article in American Sociological Review by Jennifer Randles, PhD, professor of sociology at California State University–Fresno. In 2018, Randles conducted phone interviews with 70 mothers in California over nine months. She tried to recruit fathers as well, but only two men responded.
Dr. Randles spoke with KHN’s Jenny Gold about how the cost of diapers weighs on low-income moms, and the “inventive mothering” many low-income women adopt to shield their children from the harms of poverty. The conversation has been edited for length and clarity.
Q: How do diapers play into day-to-day anxieties for low-income mothers?
In my sample, half of the mothers told me that they worried more about diapers than they worried about food or housing.
I started to ask mothers, “Can you tell me how many diapers you have on hand right now?” Almost every one told me with exact specificity how many they had – 5 or 7 or 12. And they knew exactly how long that number of diapers would last, based on how often their children defecated and urinated, if their kid was sick, if they had a diaper rash at the time. So just all the emotional and cognitive labor that goes into keeping such careful track of diaper supplies.
They were worrying and figuring out, “OK, I’m down to almost my last diaper. What do I do now? Do I go find some cans [to sell]? Do I go sell some things in my house? Who in my social network might have some extra cash right now?” I talked to moms who sell blood plasma just to get their infants diapers.
Q: What coping strategies stood out to you?
Those of us who study diapers often call them diaper-stretching strategies. One was leaving on a diaper a little bit longer than someone might otherwise leave it on and letting it get completely full. Some mothers figured out if they bought a [more expensive] diaper that held more and leaked less, they could leave the diaper on longer.
They would also do things like letting the baby go diaperless, especially when they were at home and felt like they wouldn’t be judged for letting their baby go without a diaper. And they used every household good you can imagine to make makeshift diapers. Mothers are using cloth, sheets, and pillowcases. They’re using things that are disposable like paper towels with duct tape. They’re making diapers out their own period supplies or adult incontinence supplies when they can get a sample.
One of the questions I often get is, “Why don’t they just use cloth?” A lot of the mothers that I spoke with had tried cloth diapers and they found that they were very cost- and labor-prohibitive. If you pay for a full startup set of cloth diapers, you’re looking at anywhere from $500 to $1,000. And these moms never had that much money. Most of them didn’t have in-home washers and dryers. Some of them didn’t even have homes or consistent access to water, and it’s illegal in a lot of laundromats and public laundry facilities to wash your old diapers. So the same conditions that would prevent moms from being able to readily afford disposable diapers are the same conditions that keep them from being able to use cloth.
Q: You found that, for many women, the concept of being a good mother is wrapped up in diapering. Why is that?
Diapers and managing diapers was so fundamental to their identity as good moms. Most of the mothers in my sample went without their own food. They weren’t paying a cellphone bill or buying their own medicine or their own menstrual supplies, as a way of saving diaper money.
I talked to a lot of moms who said, when your baby is hungry, that’s horrible. Obviously, you do everything to prevent that. But there’s something about a diaper that covers this vulnerable part of a very young baby’s body, this very delicate skin. And being able to do something to meet this human need that we all have, and to maintain dignity and cleanliness.
A lot of the moms had been through the welfare system, and so they’re living in this constant fear [of losing their children]. This is especially true among mothers of color, who are much more likely to get wrapped up in the child welfare system. People can’t necessarily see when your baby’s hungry. But people can see a saggy diaper. That’s going to be one of the things that tags you as a bad mom.
Q: Was your work on diapers influenced by your experience as a parent?
When I was doing these interviews, my daughter was about 2 or 3. So still in diapers. When my daughter peed during a diaper change, I thought, “Oh, I can just toss that one. Here, let me get another clean one.” That’s a really easy choice. For me. That’s a crisis for the mothers I interviewed. Many of them told me they have an anxiety attack with every diaper change.
Q: Do you see a clear policy solution to diaper stress?
What’s kind of ironic is how much physical, emotional, and cognitive labor goes into managing something that society and lawmakers don’t even recognize. Diapers are still not really recognized as a basic need, as evidenced by the fact that they’re still taxed in 35 states.
I think what California is doing is an excellent start. And I think diaper banks are a fabulous type of community-based organization that are filling a huge need that is not being filled by safety net policies. So, public support for diaper banks.
The direct cash aid part of the social safety net has been all but dismantled in the last 25 years. California is pretty generous. But there are some states where just the cost of diapers alone would use almost half of the average state TANF [Temporary Assistance for Needy Families] benefit for a family of three. I think we really do have to address the fact that the value of cash aid buys so much less than it used to.
Q: Your body of work on marriage and families is fascinating and unusual. Is there a single animating question behind your research?
The common thread is: How do our safety net policies support low-income families’ parenting goals? And do they equalize the conditions of parenting? I think of it as a reproductive justice issue. The ability to have a child or to not have a child, and then to parent that child in conditions where the child’s basic needs are met.
We like to say that we’re child and family friendly. The diaper issue is just one of many, many issues where we don’t really put our money or our policies where our mouth is, in terms of supporting families and supporting children. I think my work is trying to get people to think more collectively about having a social responsibility to all families and to each other. No country, but especially the richest country on the planet, should have one in three very young children not having one of their basic needs met.
I interviewed one dad who was incarcerated because he wrote a bad check. And as he described it to me, he had a certain amount of money, and they needed both diapers and milk for the baby. And I’ll never forget, he said, “I didn’t make a good choice, but I made the right one.”
These are not fancy shoes. These are not name-brand clothes. This was a dad needing both milk and diapers. I don’t think it gets much more basic than that.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Stool samples meet gastric biopsies for H. pylori antibiotic resistance testing
Using stool samples to test for Helicobacter pylori antibiotic resistance provides highly similar results to those of gastric biopsy samples, which suggests that stool testing may be a safer, more convenient, and more cost-effective option, according to investigators.
Head-to-head testing for resistance-associated mutations using next-generation sequencing (NGS) showed 92% concordance between the two sample types, with 100% technical success among polymerase chain reaction (PCR)–positive stool samples, lead author Steven Moss, MD, of Brown University, Providence, R.I., and colleagues reported.
“H. pylori eradication rates have declined largely due to rising antimicrobial resistance worldwide,” Dr. Moss said at the annual meeting of the American College of Gastroenterology. “There is therefore a need for rapid, accurate, reliable antibiotic resistance testing.”
According to Dr. Moss, molecular resistance testing of gastric biopsies yields similar results to culture-based testing of gastric biopsies, but endoscopic sample collection remains inconvenient and relatively costly, so “it is not commonly performed in many GI practices.
“Whether reliable resistance testing by NGS is possible from stool samples remains unclear,” Dr. Moss said.
To explore this possibility, Dr. Moss and colleagues recruited 262 patients scheduled for upper endoscopy at four sites in the United States. From each patient, two gastric biopsies were taken, and within 2 weeks of the procedure, prior to starting anti–H. pylori therapy, one stool sample was collected.
For gastric biopsy samples, H. pylori positivity was confirmed by PCR, whereas positivity in stool samples was confirmed by both fecal antigen testing and PCR. After confirmation, NGS was conducted, with screening for resistance-associated mutations to six commonly used antibiotics: clarithromycin, levofloxacin, metronidazole, tetracycline, amoxicillin, and rifabutin.
Out of 262 patients, 73 tested positive for H. pylori via stool testing; however, 2 of these patients had inadequate gastric DNA for analysis, leaving 71 patients in the evaluable dataset. Within this group, samples from 50 patients (70.4%) had at least one resistance-association mutation.
Among all 71 individuals, 65 patients (91.5%) had fully concordant results between the two sample types. In four out of the six discordant cases, there was only one difference in antibiotic-associated mutations. Concordance ranged from 89% for metronidazole mutations to 100% for tetracycline, amoxicillin, and rifabutin mutations.
“It is now possible to rapidly obtain susceptibility data without endoscopy,” Dr. Moss concluded. “Using NGS to determine H. pylori antibiotic resistance using stool obviates the cost, inconvenience, and risks of endoscopy resistance profiling.”
Dr. Moss noted that the cost of the stool-based test, through study sponsor American Molecular Laboratories, is about $450, and that the company is “working with various insurance companies to try to get [the test] reimbursed.”
For cases of H. pylori infection without resistance testing results, Dr. Moss recommended first-line treatment with quadruple bismuth–based therapy; however, he noted that “most gastroenterologists, in all kinds of practice, are not measuring their eradication success rate ... so it’s really difficult to know if your best guess is really the appropriate treatment.”
According to Lukasz Kwapisz, MD, of Baylor College of Medicine, Houston, the concordance results are “encouraging,” and suggest that stool-based testing “could be much easier for the patient and the clinician” to find ways to eradicate H. pylori infection.
Dr. Kwapisz predicted that it will take additional successful studies, as well as real-world data, to convert clinicians to the new approach. He suggested that the transition may be gradual, like the adoption of fecal calprotectin testing.
“I don’t know if it’s one singular defining study that will tell you: ‘Okay, we all have to use this [stool-based resistance testing],’ ” he said. “It kind of happens over time – over a 2- or 3-year stretch, I would think, with positive results.”
The study was supported by American Molecular Labs. The investigators disclosed additional relationships with Takeda, Phathom, and Redhill. Dr. Kwapisz reported no conflicts of interest.
Using stool samples to test for Helicobacter pylori antibiotic resistance provides highly similar results to those of gastric biopsy samples, which suggests that stool testing may be a safer, more convenient, and more cost-effective option, according to investigators.
Head-to-head testing for resistance-associated mutations using next-generation sequencing (NGS) showed 92% concordance between the two sample types, with 100% technical success among polymerase chain reaction (PCR)–positive stool samples, lead author Steven Moss, MD, of Brown University, Providence, R.I., and colleagues reported.
“H. pylori eradication rates have declined largely due to rising antimicrobial resistance worldwide,” Dr. Moss said at the annual meeting of the American College of Gastroenterology. “There is therefore a need for rapid, accurate, reliable antibiotic resistance testing.”
According to Dr. Moss, molecular resistance testing of gastric biopsies yields similar results to culture-based testing of gastric biopsies, but endoscopic sample collection remains inconvenient and relatively costly, so “it is not commonly performed in many GI practices.
“Whether reliable resistance testing by NGS is possible from stool samples remains unclear,” Dr. Moss said.
To explore this possibility, Dr. Moss and colleagues recruited 262 patients scheduled for upper endoscopy at four sites in the United States. From each patient, two gastric biopsies were taken, and within 2 weeks of the procedure, prior to starting anti–H. pylori therapy, one stool sample was collected.
For gastric biopsy samples, H. pylori positivity was confirmed by PCR, whereas positivity in stool samples was confirmed by both fecal antigen testing and PCR. After confirmation, NGS was conducted, with screening for resistance-associated mutations to six commonly used antibiotics: clarithromycin, levofloxacin, metronidazole, tetracycline, amoxicillin, and rifabutin.
Out of 262 patients, 73 tested positive for H. pylori via stool testing; however, 2 of these patients had inadequate gastric DNA for analysis, leaving 71 patients in the evaluable dataset. Within this group, samples from 50 patients (70.4%) had at least one resistance-association mutation.
Among all 71 individuals, 65 patients (91.5%) had fully concordant results between the two sample types. In four out of the six discordant cases, there was only one difference in antibiotic-associated mutations. Concordance ranged from 89% for metronidazole mutations to 100% for tetracycline, amoxicillin, and rifabutin mutations.
“It is now possible to rapidly obtain susceptibility data without endoscopy,” Dr. Moss concluded. “Using NGS to determine H. pylori antibiotic resistance using stool obviates the cost, inconvenience, and risks of endoscopy resistance profiling.”
Dr. Moss noted that the cost of the stool-based test, through study sponsor American Molecular Laboratories, is about $450, and that the company is “working with various insurance companies to try to get [the test] reimbursed.”
For cases of H. pylori infection without resistance testing results, Dr. Moss recommended first-line treatment with quadruple bismuth–based therapy; however, he noted that “most gastroenterologists, in all kinds of practice, are not measuring their eradication success rate ... so it’s really difficult to know if your best guess is really the appropriate treatment.”
According to Lukasz Kwapisz, MD, of Baylor College of Medicine, Houston, the concordance results are “encouraging,” and suggest that stool-based testing “could be much easier for the patient and the clinician” to find ways to eradicate H. pylori infection.
Dr. Kwapisz predicted that it will take additional successful studies, as well as real-world data, to convert clinicians to the new approach. He suggested that the transition may be gradual, like the adoption of fecal calprotectin testing.
“I don’t know if it’s one singular defining study that will tell you: ‘Okay, we all have to use this [stool-based resistance testing],’ ” he said. “It kind of happens over time – over a 2- or 3-year stretch, I would think, with positive results.”
The study was supported by American Molecular Labs. The investigators disclosed additional relationships with Takeda, Phathom, and Redhill. Dr. Kwapisz reported no conflicts of interest.
Using stool samples to test for Helicobacter pylori antibiotic resistance provides highly similar results to those of gastric biopsy samples, which suggests that stool testing may be a safer, more convenient, and more cost-effective option, according to investigators.
Head-to-head testing for resistance-associated mutations using next-generation sequencing (NGS) showed 92% concordance between the two sample types, with 100% technical success among polymerase chain reaction (PCR)–positive stool samples, lead author Steven Moss, MD, of Brown University, Providence, R.I., and colleagues reported.
“H. pylori eradication rates have declined largely due to rising antimicrobial resistance worldwide,” Dr. Moss said at the annual meeting of the American College of Gastroenterology. “There is therefore a need for rapid, accurate, reliable antibiotic resistance testing.”
According to Dr. Moss, molecular resistance testing of gastric biopsies yields similar results to culture-based testing of gastric biopsies, but endoscopic sample collection remains inconvenient and relatively costly, so “it is not commonly performed in many GI practices.
“Whether reliable resistance testing by NGS is possible from stool samples remains unclear,” Dr. Moss said.
To explore this possibility, Dr. Moss and colleagues recruited 262 patients scheduled for upper endoscopy at four sites in the United States. From each patient, two gastric biopsies were taken, and within 2 weeks of the procedure, prior to starting anti–H. pylori therapy, one stool sample was collected.
For gastric biopsy samples, H. pylori positivity was confirmed by PCR, whereas positivity in stool samples was confirmed by both fecal antigen testing and PCR. After confirmation, NGS was conducted, with screening for resistance-associated mutations to six commonly used antibiotics: clarithromycin, levofloxacin, metronidazole, tetracycline, amoxicillin, and rifabutin.
Out of 262 patients, 73 tested positive for H. pylori via stool testing; however, 2 of these patients had inadequate gastric DNA for analysis, leaving 71 patients in the evaluable dataset. Within this group, samples from 50 patients (70.4%) had at least one resistance-association mutation.
Among all 71 individuals, 65 patients (91.5%) had fully concordant results between the two sample types. In four out of the six discordant cases, there was only one difference in antibiotic-associated mutations. Concordance ranged from 89% for metronidazole mutations to 100% for tetracycline, amoxicillin, and rifabutin mutations.
“It is now possible to rapidly obtain susceptibility data without endoscopy,” Dr. Moss concluded. “Using NGS to determine H. pylori antibiotic resistance using stool obviates the cost, inconvenience, and risks of endoscopy resistance profiling.”
Dr. Moss noted that the cost of the stool-based test, through study sponsor American Molecular Laboratories, is about $450, and that the company is “working with various insurance companies to try to get [the test] reimbursed.”
For cases of H. pylori infection without resistance testing results, Dr. Moss recommended first-line treatment with quadruple bismuth–based therapy; however, he noted that “most gastroenterologists, in all kinds of practice, are not measuring their eradication success rate ... so it’s really difficult to know if your best guess is really the appropriate treatment.”
According to Lukasz Kwapisz, MD, of Baylor College of Medicine, Houston, the concordance results are “encouraging,” and suggest that stool-based testing “could be much easier for the patient and the clinician” to find ways to eradicate H. pylori infection.
Dr. Kwapisz predicted that it will take additional successful studies, as well as real-world data, to convert clinicians to the new approach. He suggested that the transition may be gradual, like the adoption of fecal calprotectin testing.
“I don’t know if it’s one singular defining study that will tell you: ‘Okay, we all have to use this [stool-based resistance testing],’ ” he said. “It kind of happens over time – over a 2- or 3-year stretch, I would think, with positive results.”
The study was supported by American Molecular Labs. The investigators disclosed additional relationships with Takeda, Phathom, and Redhill. Dr. Kwapisz reported no conflicts of interest.
FROM ACG 2021
Which specialties get the biggest markups over Medicare rates?
Anesthesiologists charge private insurers more than 300% above Medicare rates, a markup that is higher than that of 16 other specialties, according to a study released by the Urban Institute.
The Washington-based nonprofit institute found that the lowest markups were in psychiatry, ophthalmology, ob.gyn., family medicine, gastroenterology, and internal medicine, at 110%-120% of Medicare rates. .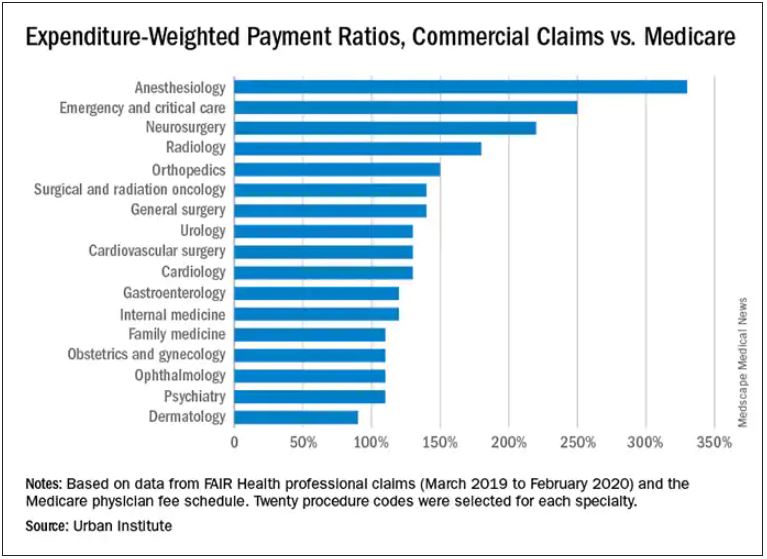
In the middle are cardiology and cardiovascular surgery (130%), urology (130%), general surgery, surgical and radiation oncology (all at 140%), and orthopedics (150%).
At the top end were radiology (180%), neurosurgery (220%), emergency and critical care (250%), and anesthesiology (330%).
The wide variation in payments could be cited in support of the idea of applying Medicare rates across all physician specialties, say the study authors. Although lowering practitioner payments might lead to savings, it “will also create more pushback from providers, especially if these rates are introduced in the employer market,” write researchers Stacey McMorrow, PhD, Robert A. Berenson, MD, and John Holahan, PhD.
It is not known whether lowering commercial payment rates might decrease patient access, they write.
The authors also note that specialties in which the potential for a fee reduction was greatest were also the specialties for which baseline compensation was highest – from $350,000 annually for emergency physicians to $800,000 a year for neurosurgeons. Annual compensation for ob.gyns., dermatologists, and opthalmologists is about $350,000 a year, which suggests that “these specialties are similarly well compensated by both Medicare and commercial insurers,” the authors write.
The investigators assessed the top 20 procedure codes by expenditure in each of 17 physician specialties. They estimated the commercial-to-Medicare payment ratio for each service and constructed weighted averages across services for each specialty at the national level and for 12 states for which data for all the specialties and services were available.
The researchers analyzed claims from the FAIR Health database between March 2019 and March 2020. That database represents 60 insurers covering 150 million people.
Pediatric and geriatric specialties, nonphysician practitioners, out-of-network clinicians, and ambulatory surgery center claims were excluded. Codes with modifiers, J codes, and clinical laboratory services were also not included.
The charges used in the study were not the actual contracted rates. The authors instead used “imputed allowed amounts” for each claim line. That method was used to protect the confidentiality of the negotiated rates.
With regard to all specialties, the lowest compensated services were procedures, evaluation and management, and tests, which received 140%-150% of the Medicare rate. Treatments and imaging were marked up 160%. Anesthesia was reimbursed at a rate 330% higher than the rate Medicare would pay.
The authors also assessed geographic variation for the 12 states for which they had data.
Similar to findings in other studies, the researchers found that the markup was lowest in Pennsylvania (120%) and highest in Wisconsin (260%). The U.S. average was 160%. California and Missouri were at 150%; Michigan was right at the average.
For physicians in Illinois, Louisiana, Colorado, Texas, and New York, markups were 170%-180% over the Medicare rate. Markups for clinicians in New Jersey (190%) and Arizona (200%) were closest to the Wisconsin rate.
The authors note some study limitations, including the fact that they excluded out-of-network practitioners, “and such payments may disproportionately affect certain specialties.”
A version of this article first appeared on Medscape.com.
Anesthesiologists charge private insurers more than 300% above Medicare rates, a markup that is higher than that of 16 other specialties, according to a study released by the Urban Institute.
The Washington-based nonprofit institute found that the lowest markups were in psychiatry, ophthalmology, ob.gyn., family medicine, gastroenterology, and internal medicine, at 110%-120% of Medicare rates. .
In the middle are cardiology and cardiovascular surgery (130%), urology (130%), general surgery, surgical and radiation oncology (all at 140%), and orthopedics (150%).
At the top end were radiology (180%), neurosurgery (220%), emergency and critical care (250%), and anesthesiology (330%).
The wide variation in payments could be cited in support of the idea of applying Medicare rates across all physician specialties, say the study authors. Although lowering practitioner payments might lead to savings, it “will also create more pushback from providers, especially if these rates are introduced in the employer market,” write researchers Stacey McMorrow, PhD, Robert A. Berenson, MD, and John Holahan, PhD.
It is not known whether lowering commercial payment rates might decrease patient access, they write.
The authors also note that specialties in which the potential for a fee reduction was greatest were also the specialties for which baseline compensation was highest – from $350,000 annually for emergency physicians to $800,000 a year for neurosurgeons. Annual compensation for ob.gyns., dermatologists, and opthalmologists is about $350,000 a year, which suggests that “these specialties are similarly well compensated by both Medicare and commercial insurers,” the authors write.
The investigators assessed the top 20 procedure codes by expenditure in each of 17 physician specialties. They estimated the commercial-to-Medicare payment ratio for each service and constructed weighted averages across services for each specialty at the national level and for 12 states for which data for all the specialties and services were available.
The researchers analyzed claims from the FAIR Health database between March 2019 and March 2020. That database represents 60 insurers covering 150 million people.
Pediatric and geriatric specialties, nonphysician practitioners, out-of-network clinicians, and ambulatory surgery center claims were excluded. Codes with modifiers, J codes, and clinical laboratory services were also not included.
The charges used in the study were not the actual contracted rates. The authors instead used “imputed allowed amounts” for each claim line. That method was used to protect the confidentiality of the negotiated rates.
With regard to all specialties, the lowest compensated services were procedures, evaluation and management, and tests, which received 140%-150% of the Medicare rate. Treatments and imaging were marked up 160%. Anesthesia was reimbursed at a rate 330% higher than the rate Medicare would pay.
The authors also assessed geographic variation for the 12 states for which they had data.
Similar to findings in other studies, the researchers found that the markup was lowest in Pennsylvania (120%) and highest in Wisconsin (260%). The U.S. average was 160%. California and Missouri were at 150%; Michigan was right at the average.
For physicians in Illinois, Louisiana, Colorado, Texas, and New York, markups were 170%-180% over the Medicare rate. Markups for clinicians in New Jersey (190%) and Arizona (200%) were closest to the Wisconsin rate.
The authors note some study limitations, including the fact that they excluded out-of-network practitioners, “and such payments may disproportionately affect certain specialties.”
A version of this article first appeared on Medscape.com.
Anesthesiologists charge private insurers more than 300% above Medicare rates, a markup that is higher than that of 16 other specialties, according to a study released by the Urban Institute.
The Washington-based nonprofit institute found that the lowest markups were in psychiatry, ophthalmology, ob.gyn., family medicine, gastroenterology, and internal medicine, at 110%-120% of Medicare rates. .
In the middle are cardiology and cardiovascular surgery (130%), urology (130%), general surgery, surgical and radiation oncology (all at 140%), and orthopedics (150%).
At the top end were radiology (180%), neurosurgery (220%), emergency and critical care (250%), and anesthesiology (330%).
The wide variation in payments could be cited in support of the idea of applying Medicare rates across all physician specialties, say the study authors. Although lowering practitioner payments might lead to savings, it “will also create more pushback from providers, especially if these rates are introduced in the employer market,” write researchers Stacey McMorrow, PhD, Robert A. Berenson, MD, and John Holahan, PhD.
It is not known whether lowering commercial payment rates might decrease patient access, they write.
The authors also note that specialties in which the potential for a fee reduction was greatest were also the specialties for which baseline compensation was highest – from $350,000 annually for emergency physicians to $800,000 a year for neurosurgeons. Annual compensation for ob.gyns., dermatologists, and opthalmologists is about $350,000 a year, which suggests that “these specialties are similarly well compensated by both Medicare and commercial insurers,” the authors write.
The investigators assessed the top 20 procedure codes by expenditure in each of 17 physician specialties. They estimated the commercial-to-Medicare payment ratio for each service and constructed weighted averages across services for each specialty at the national level and for 12 states for which data for all the specialties and services were available.
The researchers analyzed claims from the FAIR Health database between March 2019 and March 2020. That database represents 60 insurers covering 150 million people.
Pediatric and geriatric specialties, nonphysician practitioners, out-of-network clinicians, and ambulatory surgery center claims were excluded. Codes with modifiers, J codes, and clinical laboratory services were also not included.
The charges used in the study were not the actual contracted rates. The authors instead used “imputed allowed amounts” for each claim line. That method was used to protect the confidentiality of the negotiated rates.
With regard to all specialties, the lowest compensated services were procedures, evaluation and management, and tests, which received 140%-150% of the Medicare rate. Treatments and imaging were marked up 160%. Anesthesia was reimbursed at a rate 330% higher than the rate Medicare would pay.
The authors also assessed geographic variation for the 12 states for which they had data.
Similar to findings in other studies, the researchers found that the markup was lowest in Pennsylvania (120%) and highest in Wisconsin (260%). The U.S. average was 160%. California and Missouri were at 150%; Michigan was right at the average.
For physicians in Illinois, Louisiana, Colorado, Texas, and New York, markups were 170%-180% over the Medicare rate. Markups for clinicians in New Jersey (190%) and Arizona (200%) were closest to the Wisconsin rate.
The authors note some study limitations, including the fact that they excluded out-of-network practitioners, “and such payments may disproportionately affect certain specialties.”
A version of this article first appeared on Medscape.com.
German society for internal medicine reappraises its Nazi history
. The decision was made after the DGIM reappraised its own history during the Nazi period.
On the DGIM - Commemoration and Remembrance website, created in 2020, members who suffered under the Nazi regime are commemorated and those who committed crimes and caused suffering are called out.
The reappraisal began in 2012, when the DGIM commissioned two historians — Hans-Georg Hofer, PhD, from the University of Münster, and Ralf Forsbach, PhD, from the Institute for Ethics, History and Theory of Medicine at the University of Münster — to research the history of the society and its members during the periods of the National Socialism dictatorship and the young Federal Republic.
“Reappraising our own history, even at this late stage, is important and the right thing to do, although of course it cannot in any way make up for the suffering caused by individual DGIM members during that time,” Georg Ertl, MD, secretary general of the DGIM, states in a press release.
It is important, however, that the Society takes appropriate action in response to the historians’ findings, he adds.
The DGIM has done just that by retrospectively withdrawing the honorary membership status of five of its former members: Alfred Schittenhelm, Alfred Schwenkenbecher, Hans Dietlen, Siegfried Koller, and Georg Schaltenbrand.
“Out of opportunism or on the basis of Nazi beliefs, they intentionally harmed colleagues, other members of our Society, or simply other people on the basis of their ethnicity. Therefore, the DGIM can no longer accept them as honorary members,” said Markus M. Lerch, MD, chair of the DGIM and medical director of the Ludwig Maximilian University of Munich.
The board has also distanced itself from two other honorary members: Gustav von Bergmann and Felix Lommel. “More research is needed and we cannot currently make a responsible decision on withdrawal of honorary membership,” Dr. Lerch explained.
Early results from the historical reappraisal were presented to the public in 2015 at an exhibition held during the 121st DGIM Congress in Mannheim. The Society concurrently underwent a process of public self-reflection, stating that it was ashamed of having allowed 70 years to pass before it objectively examined its actions under National Socialism and acknowledged its responsibility.
The exhibition used photos, documents, and explanatory texts to show the actions, or lack of action, taken by some Society members during the Nazi regime. For example, it showed how then DGIM chair Alfred Schittenhelm — whose honorary membership has since been withdrawn — put the Society on the track to National Socialism. It also shone light on the role played by internists who consulted with the Wehrmacht in the treatment of Soviet prisoners of war and others, and on criminal experiments conducted on humans.
The exhibition also highlighted Jewish doctors who were persecuted and expelled, such as Leopold Lichtwitz, MD, who lost his position as clinic director in Berlin in 1933 and was forced to resign his chairmanship of the Society. And it presented portraits of members who loudly objected to and even actively resisted the regime, such as Wolfgang Seitz, MD, who became director of the Medical Outpatient Clinic at the University of Munich and deputy of the state parliament of the Social Democratic Party of Germany (SPD) in Bavaria after the war.
The historians focused on the years after 1945, because “1945 was no zero hour,” said Dr. Forsbach. Some culpable doctors continued to practice or became honorary DGIM members, he explained. The DGIM’s attitude toward history was characterized by suppression, denial, silence, and attempts at justification, consistent with the postwar attitude in the Federal Republic of Germany and in the medical profession as a whole.
After 1945
Behavior before 1945 is not the only source of shame. Crimes committed by doctors were never really confronted until the late 1970s, Jörg-Dietrich Hoppe, MD, former president of the German Medical Association, explained to ZEIT, a German newspaper, in 2011.
The psychoanalyst Alexander Mitscherlich, MD, and Fred Mielke were official observers from the German Commission of Physicians at the Nuremberg Doctors’ Trial who were made painfully aware that National Socialism was by no means over when the regime came to an end. They were both reviled as traitors to their country and for fouling their own nest, and, according to Mitscherlich, the behavior of the “authorities” bordered on character assassination.
As late as 1973, a renowned internist threatened that German internists would leave the room locked at the upcoming DGIM Congress if — as had been planned by congress chair Herbert Begemann — Mitscherlich gave a talk on this subject, journalist and doctor Renate Jäckle reported in her book on doctors and politics.
Even toward the end of the 1980s, Karsten Vilmar, MD, then president of the German Medical Association, reacted in an insensitive and defensive manner — during an interview — to an article in the Lancet, written by the Mainz pediatrician Hartmut M. Hanauske-Abel, MD, on the role of the German medical profession in the Third Reich and the suppression that followed after 1945.
A group of 400 doctors, at most, were culpable, and coming to terms with the past should not defame doctors collectively, Dr. Vilmar said in a statement chillingly reminiscent of declarations made by the Wehrmacht, which described itself as mainly “clean.”
Of course, the end of the Nazi regime was not the end of all barbarity, not even in Europe. “Violence will be something we have to confront in our future lives, too. Belief in the healing powers of civilization is nothing but a fairytale,” Berlin historian Jörg Barberowski wrote in a 2012 essay.
Nevertheless, as Michael Hallek, MD, from the University Hospital of Cologne, said, it is important to keep memory alive.
A version of this article first appeared on Medscape.com.
. The decision was made after the DGIM reappraised its own history during the Nazi period.
On the DGIM - Commemoration and Remembrance website, created in 2020, members who suffered under the Nazi regime are commemorated and those who committed crimes and caused suffering are called out.
The reappraisal began in 2012, when the DGIM commissioned two historians — Hans-Georg Hofer, PhD, from the University of Münster, and Ralf Forsbach, PhD, from the Institute for Ethics, History and Theory of Medicine at the University of Münster — to research the history of the society and its members during the periods of the National Socialism dictatorship and the young Federal Republic.
“Reappraising our own history, even at this late stage, is important and the right thing to do, although of course it cannot in any way make up for the suffering caused by individual DGIM members during that time,” Georg Ertl, MD, secretary general of the DGIM, states in a press release.
It is important, however, that the Society takes appropriate action in response to the historians’ findings, he adds.
The DGIM has done just that by retrospectively withdrawing the honorary membership status of five of its former members: Alfred Schittenhelm, Alfred Schwenkenbecher, Hans Dietlen, Siegfried Koller, and Georg Schaltenbrand.
“Out of opportunism or on the basis of Nazi beliefs, they intentionally harmed colleagues, other members of our Society, or simply other people on the basis of their ethnicity. Therefore, the DGIM can no longer accept them as honorary members,” said Markus M. Lerch, MD, chair of the DGIM and medical director of the Ludwig Maximilian University of Munich.
The board has also distanced itself from two other honorary members: Gustav von Bergmann and Felix Lommel. “More research is needed and we cannot currently make a responsible decision on withdrawal of honorary membership,” Dr. Lerch explained.
Early results from the historical reappraisal were presented to the public in 2015 at an exhibition held during the 121st DGIM Congress in Mannheim. The Society concurrently underwent a process of public self-reflection, stating that it was ashamed of having allowed 70 years to pass before it objectively examined its actions under National Socialism and acknowledged its responsibility.
The exhibition used photos, documents, and explanatory texts to show the actions, or lack of action, taken by some Society members during the Nazi regime. For example, it showed how then DGIM chair Alfred Schittenhelm — whose honorary membership has since been withdrawn — put the Society on the track to National Socialism. It also shone light on the role played by internists who consulted with the Wehrmacht in the treatment of Soviet prisoners of war and others, and on criminal experiments conducted on humans.
The exhibition also highlighted Jewish doctors who were persecuted and expelled, such as Leopold Lichtwitz, MD, who lost his position as clinic director in Berlin in 1933 and was forced to resign his chairmanship of the Society. And it presented portraits of members who loudly objected to and even actively resisted the regime, such as Wolfgang Seitz, MD, who became director of the Medical Outpatient Clinic at the University of Munich and deputy of the state parliament of the Social Democratic Party of Germany (SPD) in Bavaria after the war.
The historians focused on the years after 1945, because “1945 was no zero hour,” said Dr. Forsbach. Some culpable doctors continued to practice or became honorary DGIM members, he explained. The DGIM’s attitude toward history was characterized by suppression, denial, silence, and attempts at justification, consistent with the postwar attitude in the Federal Republic of Germany and in the medical profession as a whole.
After 1945
Behavior before 1945 is not the only source of shame. Crimes committed by doctors were never really confronted until the late 1970s, Jörg-Dietrich Hoppe, MD, former president of the German Medical Association, explained to ZEIT, a German newspaper, in 2011.
The psychoanalyst Alexander Mitscherlich, MD, and Fred Mielke were official observers from the German Commission of Physicians at the Nuremberg Doctors’ Trial who were made painfully aware that National Socialism was by no means over when the regime came to an end. They were both reviled as traitors to their country and for fouling their own nest, and, according to Mitscherlich, the behavior of the “authorities” bordered on character assassination.
As late as 1973, a renowned internist threatened that German internists would leave the room locked at the upcoming DGIM Congress if — as had been planned by congress chair Herbert Begemann — Mitscherlich gave a talk on this subject, journalist and doctor Renate Jäckle reported in her book on doctors and politics.
Even toward the end of the 1980s, Karsten Vilmar, MD, then president of the German Medical Association, reacted in an insensitive and defensive manner — during an interview — to an article in the Lancet, written by the Mainz pediatrician Hartmut M. Hanauske-Abel, MD, on the role of the German medical profession in the Third Reich and the suppression that followed after 1945.
A group of 400 doctors, at most, were culpable, and coming to terms with the past should not defame doctors collectively, Dr. Vilmar said in a statement chillingly reminiscent of declarations made by the Wehrmacht, which described itself as mainly “clean.”
Of course, the end of the Nazi regime was not the end of all barbarity, not even in Europe. “Violence will be something we have to confront in our future lives, too. Belief in the healing powers of civilization is nothing but a fairytale,” Berlin historian Jörg Barberowski wrote in a 2012 essay.
Nevertheless, as Michael Hallek, MD, from the University Hospital of Cologne, said, it is important to keep memory alive.
A version of this article first appeared on Medscape.com.
. The decision was made after the DGIM reappraised its own history during the Nazi period.
On the DGIM - Commemoration and Remembrance website, created in 2020, members who suffered under the Nazi regime are commemorated and those who committed crimes and caused suffering are called out.
The reappraisal began in 2012, when the DGIM commissioned two historians — Hans-Georg Hofer, PhD, from the University of Münster, and Ralf Forsbach, PhD, from the Institute for Ethics, History and Theory of Medicine at the University of Münster — to research the history of the society and its members during the periods of the National Socialism dictatorship and the young Federal Republic.
“Reappraising our own history, even at this late stage, is important and the right thing to do, although of course it cannot in any way make up for the suffering caused by individual DGIM members during that time,” Georg Ertl, MD, secretary general of the DGIM, states in a press release.
It is important, however, that the Society takes appropriate action in response to the historians’ findings, he adds.
The DGIM has done just that by retrospectively withdrawing the honorary membership status of five of its former members: Alfred Schittenhelm, Alfred Schwenkenbecher, Hans Dietlen, Siegfried Koller, and Georg Schaltenbrand.
“Out of opportunism or on the basis of Nazi beliefs, they intentionally harmed colleagues, other members of our Society, or simply other people on the basis of their ethnicity. Therefore, the DGIM can no longer accept them as honorary members,” said Markus M. Lerch, MD, chair of the DGIM and medical director of the Ludwig Maximilian University of Munich.
The board has also distanced itself from two other honorary members: Gustav von Bergmann and Felix Lommel. “More research is needed and we cannot currently make a responsible decision on withdrawal of honorary membership,” Dr. Lerch explained.
Early results from the historical reappraisal were presented to the public in 2015 at an exhibition held during the 121st DGIM Congress in Mannheim. The Society concurrently underwent a process of public self-reflection, stating that it was ashamed of having allowed 70 years to pass before it objectively examined its actions under National Socialism and acknowledged its responsibility.
The exhibition used photos, documents, and explanatory texts to show the actions, or lack of action, taken by some Society members during the Nazi regime. For example, it showed how then DGIM chair Alfred Schittenhelm — whose honorary membership has since been withdrawn — put the Society on the track to National Socialism. It also shone light on the role played by internists who consulted with the Wehrmacht in the treatment of Soviet prisoners of war and others, and on criminal experiments conducted on humans.
The exhibition also highlighted Jewish doctors who were persecuted and expelled, such as Leopold Lichtwitz, MD, who lost his position as clinic director in Berlin in 1933 and was forced to resign his chairmanship of the Society. And it presented portraits of members who loudly objected to and even actively resisted the regime, such as Wolfgang Seitz, MD, who became director of the Medical Outpatient Clinic at the University of Munich and deputy of the state parliament of the Social Democratic Party of Germany (SPD) in Bavaria after the war.
The historians focused on the years after 1945, because “1945 was no zero hour,” said Dr. Forsbach. Some culpable doctors continued to practice or became honorary DGIM members, he explained. The DGIM’s attitude toward history was characterized by suppression, denial, silence, and attempts at justification, consistent with the postwar attitude in the Federal Republic of Germany and in the medical profession as a whole.
After 1945
Behavior before 1945 is not the only source of shame. Crimes committed by doctors were never really confronted until the late 1970s, Jörg-Dietrich Hoppe, MD, former president of the German Medical Association, explained to ZEIT, a German newspaper, in 2011.
The psychoanalyst Alexander Mitscherlich, MD, and Fred Mielke were official observers from the German Commission of Physicians at the Nuremberg Doctors’ Trial who were made painfully aware that National Socialism was by no means over when the regime came to an end. They were both reviled as traitors to their country and for fouling their own nest, and, according to Mitscherlich, the behavior of the “authorities” bordered on character assassination.
As late as 1973, a renowned internist threatened that German internists would leave the room locked at the upcoming DGIM Congress if — as had been planned by congress chair Herbert Begemann — Mitscherlich gave a talk on this subject, journalist and doctor Renate Jäckle reported in her book on doctors and politics.
Even toward the end of the 1980s, Karsten Vilmar, MD, then president of the German Medical Association, reacted in an insensitive and defensive manner — during an interview — to an article in the Lancet, written by the Mainz pediatrician Hartmut M. Hanauske-Abel, MD, on the role of the German medical profession in the Third Reich and the suppression that followed after 1945.
A group of 400 doctors, at most, were culpable, and coming to terms with the past should not defame doctors collectively, Dr. Vilmar said in a statement chillingly reminiscent of declarations made by the Wehrmacht, which described itself as mainly “clean.”
Of course, the end of the Nazi regime was not the end of all barbarity, not even in Europe. “Violence will be something we have to confront in our future lives, too. Belief in the healing powers of civilization is nothing but a fairytale,” Berlin historian Jörg Barberowski wrote in a 2012 essay.
Nevertheless, as Michael Hallek, MD, from the University Hospital of Cologne, said, it is important to keep memory alive.
A version of this article first appeared on Medscape.com.
AGA says stay the course, despite the Delta variant
As COVID-19 cases rise in the United States due to the Delta variant, there is renewed concern about infection and transmission of SARS-CoV-2 during endoscopy. In May 2021, AGA released updated recommendations on preprocedure testing post vaccination in the setting of ongoing population-wide vaccination programs for the prevention of COVID-19–related morbidity. In vaccinated individuals, breakthrough infections occurred very infrequently. Weighing the evidence demonstrating extremely low rates of rates of infection and transmission with vaccination and PPE, and considering the downsides of routine testing (burden, cost, false test results, increased disparities), AGA made a conditional recommendation against routine preprocedure testing for elective cases. The highly contagious Delta variant has now emerged as the predominant SARS-CoV2 virus in the U.S. and some data suggests that it may cause more severe illness than previous strains. While more breakthrough infections may develop in fully vaccinated individuals, the greatest risk of infection, transmission and hospitalizations is among those who are unvaccinated.
- AGA suggests against reinstituting routine preprocedure testing prior to elective endoscopy. The downsides (delays in patient care, burden, inaccurate results) outweigh potential benefits. Infection and transmission of SARS-CoV-2 from asymptomatic individuals is rare especially among vaccinated health care workers using personal protective equipment (PPE), even with the emergence of the Delta variant.
- If PPE is available, AGA recommends using N95 for upper endoscopy and suggests using N95 or surgical masks for lower endoscopy (acknowledging that upper endoscopy is more aerosolizing than lower endoscopy) and continuation of elective and nonelective endoscopy.
- Based on local prevalence rates, PPE, and test availability, in intermediate- and high-prevalence settings, preprocedure testing may be used to inform PPE decisions (N95 versus surgical mask). Additional benefits to testing are small and include deferring elective endoscopy in individuals testing positive and reducing anxiety among staff and patients.
As COVID-19 cases rise in the United States due to the Delta variant, there is renewed concern about infection and transmission of SARS-CoV-2 during endoscopy. In May 2021, AGA released updated recommendations on preprocedure testing post vaccination in the setting of ongoing population-wide vaccination programs for the prevention of COVID-19–related morbidity. In vaccinated individuals, breakthrough infections occurred very infrequently. Weighing the evidence demonstrating extremely low rates of rates of infection and transmission with vaccination and PPE, and considering the downsides of routine testing (burden, cost, false test results, increased disparities), AGA made a conditional recommendation against routine preprocedure testing for elective cases. The highly contagious Delta variant has now emerged as the predominant SARS-CoV2 virus in the U.S. and some data suggests that it may cause more severe illness than previous strains. While more breakthrough infections may develop in fully vaccinated individuals, the greatest risk of infection, transmission and hospitalizations is among those who are unvaccinated.
- AGA suggests against reinstituting routine preprocedure testing prior to elective endoscopy. The downsides (delays in patient care, burden, inaccurate results) outweigh potential benefits. Infection and transmission of SARS-CoV-2 from asymptomatic individuals is rare especially among vaccinated health care workers using personal protective equipment (PPE), even with the emergence of the Delta variant.
- If PPE is available, AGA recommends using N95 for upper endoscopy and suggests using N95 or surgical masks for lower endoscopy (acknowledging that upper endoscopy is more aerosolizing than lower endoscopy) and continuation of elective and nonelective endoscopy.
- Based on local prevalence rates, PPE, and test availability, in intermediate- and high-prevalence settings, preprocedure testing may be used to inform PPE decisions (N95 versus surgical mask). Additional benefits to testing are small and include deferring elective endoscopy in individuals testing positive and reducing anxiety among staff and patients.
As COVID-19 cases rise in the United States due to the Delta variant, there is renewed concern about infection and transmission of SARS-CoV-2 during endoscopy. In May 2021, AGA released updated recommendations on preprocedure testing post vaccination in the setting of ongoing population-wide vaccination programs for the prevention of COVID-19–related morbidity. In vaccinated individuals, breakthrough infections occurred very infrequently. Weighing the evidence demonstrating extremely low rates of rates of infection and transmission with vaccination and PPE, and considering the downsides of routine testing (burden, cost, false test results, increased disparities), AGA made a conditional recommendation against routine preprocedure testing for elective cases. The highly contagious Delta variant has now emerged as the predominant SARS-CoV2 virus in the U.S. and some data suggests that it may cause more severe illness than previous strains. While more breakthrough infections may develop in fully vaccinated individuals, the greatest risk of infection, transmission and hospitalizations is among those who are unvaccinated.
- AGA suggests against reinstituting routine preprocedure testing prior to elective endoscopy. The downsides (delays in patient care, burden, inaccurate results) outweigh potential benefits. Infection and transmission of SARS-CoV-2 from asymptomatic individuals is rare especially among vaccinated health care workers using personal protective equipment (PPE), even with the emergence of the Delta variant.
- If PPE is available, AGA recommends using N95 for upper endoscopy and suggests using N95 or surgical masks for lower endoscopy (acknowledging that upper endoscopy is more aerosolizing than lower endoscopy) and continuation of elective and nonelective endoscopy.
- Based on local prevalence rates, PPE, and test availability, in intermediate- and high-prevalence settings, preprocedure testing may be used to inform PPE decisions (N95 versus surgical mask). Additional benefits to testing are small and include deferring elective endoscopy in individuals testing positive and reducing anxiety among staff and patients.
Autism prevalence in children as high as 10% in some New Jersey communities
Investigators found that up to 10% of children in some of the state’s school districts have an ASD diagnosis vs. the national average of just under 2%.
School districts with higher ASD prevalence in the study have expansive health and educational programs in place to diagnose and support children with ASD, which likely contributed to the higher caseloads, senior investigator Walter Zahorodny, PhD, associate professor of pediatrics at New Jersey Medical School in Newark, said in an interview.
“When you have those players on the ground, it’s likely we’re going to be finding more, if not almost all, of the children with autism in a given district,” said Dr. Zahorodny, director of the New Jersey Autism Study for the Centers for Disease Control and Prevention.
The study was published online Oct. 21 in the journal Autism Research.
Local analysis
Researchers used the Autism and Developmental Disabilities Monitoring (ADDM) Network, a surveillance method developed by the CDC that includes data collected from health and special education records of children living in 11 communities across the United States. New Jersey is one of the ADDM participating sites.
National data are important, but Dr. Zahorodny and colleagues wanted to examine ASD prevalence at a more granular level, comparing prevalence district by district.
They examined data from 5,453 children who were 8 years old in 2016 and attended public school in Essex, Hudson, Ocean, and Union counties.
The prevalence of ASD was 36 children per 1,000 overall. Hudson County reported the lowest rate, at 31 cases per 1,000 children, and Ocean County reported the highest, at 54 cases per 1,000 children.
Across the region, ASD prevalence was four times higher in boys vs. girls, mirroring national statistics.
High ASD prevalence was more likely in mid-socioeconomic status districts (prevalence ratio [PR], 1.2; P = .01) and in larger school districts (PR, 1.3; P = .004).
Hispanic children had significantly lower ASD prevalence overall compared with White children (PR, 0.6; P < .001). In fact, prevalence was 30%-60% lower among Hispanic children in three of four counties compared with White children.
Another study is underway to better understand why autism rates were lower in specific districts and Hispanic children overall, but Dr. Zahorodny said one possibility is a lack of resources in those districts.
Will new methodology miss cases?
The study’s methodology was used by the CDC from 2000 to 2016 and includes assessment of children who have an ASD diagnosis, and children who haven’t received a diagnosis but have documented behaviors consistent with ASD.
In 2018, it was replaced with a less comprehensive approach that relies only on children with an ASD diagnosis. Data using this new methodology have not yet been reported.
In the new study from New Jersey, 767 children with autism were diagnosed by a pediatrician, neurologist, or other community provider. The remaining 175 children with autism, 18.6% of the total cohort, did not have an ASD diagnosis but met the ADDM case definition.
Under the new methodology, those children would not be counted.
“Something could be lost in the new methodology in terms of usefulness of the information because when the estimates are incomplete or low, that might lead people to make the wrong judgments when they make decisions about resources,” Dr. Zahorodny said.
The study was funded by the Centers for Disease Control and Prevention and the National Institutes of Health. The study authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators found that up to 10% of children in some of the state’s school districts have an ASD diagnosis vs. the national average of just under 2%.
School districts with higher ASD prevalence in the study have expansive health and educational programs in place to diagnose and support children with ASD, which likely contributed to the higher caseloads, senior investigator Walter Zahorodny, PhD, associate professor of pediatrics at New Jersey Medical School in Newark, said in an interview.
“When you have those players on the ground, it’s likely we’re going to be finding more, if not almost all, of the children with autism in a given district,” said Dr. Zahorodny, director of the New Jersey Autism Study for the Centers for Disease Control and Prevention.
The study was published online Oct. 21 in the journal Autism Research.
Local analysis
Researchers used the Autism and Developmental Disabilities Monitoring (ADDM) Network, a surveillance method developed by the CDC that includes data collected from health and special education records of children living in 11 communities across the United States. New Jersey is one of the ADDM participating sites.
National data are important, but Dr. Zahorodny and colleagues wanted to examine ASD prevalence at a more granular level, comparing prevalence district by district.
They examined data from 5,453 children who were 8 years old in 2016 and attended public school in Essex, Hudson, Ocean, and Union counties.
The prevalence of ASD was 36 children per 1,000 overall. Hudson County reported the lowest rate, at 31 cases per 1,000 children, and Ocean County reported the highest, at 54 cases per 1,000 children.
Across the region, ASD prevalence was four times higher in boys vs. girls, mirroring national statistics.
High ASD prevalence was more likely in mid-socioeconomic status districts (prevalence ratio [PR], 1.2; P = .01) and in larger school districts (PR, 1.3; P = .004).
Hispanic children had significantly lower ASD prevalence overall compared with White children (PR, 0.6; P < .001). In fact, prevalence was 30%-60% lower among Hispanic children in three of four counties compared with White children.
Another study is underway to better understand why autism rates were lower in specific districts and Hispanic children overall, but Dr. Zahorodny said one possibility is a lack of resources in those districts.
Will new methodology miss cases?
The study’s methodology was used by the CDC from 2000 to 2016 and includes assessment of children who have an ASD diagnosis, and children who haven’t received a diagnosis but have documented behaviors consistent with ASD.
In 2018, it was replaced with a less comprehensive approach that relies only on children with an ASD diagnosis. Data using this new methodology have not yet been reported.
In the new study from New Jersey, 767 children with autism were diagnosed by a pediatrician, neurologist, or other community provider. The remaining 175 children with autism, 18.6% of the total cohort, did not have an ASD diagnosis but met the ADDM case definition.
Under the new methodology, those children would not be counted.
“Something could be lost in the new methodology in terms of usefulness of the information because when the estimates are incomplete or low, that might lead people to make the wrong judgments when they make decisions about resources,” Dr. Zahorodny said.
The study was funded by the Centers for Disease Control and Prevention and the National Institutes of Health. The study authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators found that up to 10% of children in some of the state’s school districts have an ASD diagnosis vs. the national average of just under 2%.
School districts with higher ASD prevalence in the study have expansive health and educational programs in place to diagnose and support children with ASD, which likely contributed to the higher caseloads, senior investigator Walter Zahorodny, PhD, associate professor of pediatrics at New Jersey Medical School in Newark, said in an interview.
“When you have those players on the ground, it’s likely we’re going to be finding more, if not almost all, of the children with autism in a given district,” said Dr. Zahorodny, director of the New Jersey Autism Study for the Centers for Disease Control and Prevention.
The study was published online Oct. 21 in the journal Autism Research.
Local analysis
Researchers used the Autism and Developmental Disabilities Monitoring (ADDM) Network, a surveillance method developed by the CDC that includes data collected from health and special education records of children living in 11 communities across the United States. New Jersey is one of the ADDM participating sites.
National data are important, but Dr. Zahorodny and colleagues wanted to examine ASD prevalence at a more granular level, comparing prevalence district by district.
They examined data from 5,453 children who were 8 years old in 2016 and attended public school in Essex, Hudson, Ocean, and Union counties.
The prevalence of ASD was 36 children per 1,000 overall. Hudson County reported the lowest rate, at 31 cases per 1,000 children, and Ocean County reported the highest, at 54 cases per 1,000 children.
Across the region, ASD prevalence was four times higher in boys vs. girls, mirroring national statistics.
High ASD prevalence was more likely in mid-socioeconomic status districts (prevalence ratio [PR], 1.2; P = .01) and in larger school districts (PR, 1.3; P = .004).
Hispanic children had significantly lower ASD prevalence overall compared with White children (PR, 0.6; P < .001). In fact, prevalence was 30%-60% lower among Hispanic children in three of four counties compared with White children.
Another study is underway to better understand why autism rates were lower in specific districts and Hispanic children overall, but Dr. Zahorodny said one possibility is a lack of resources in those districts.
Will new methodology miss cases?
The study’s methodology was used by the CDC from 2000 to 2016 and includes assessment of children who have an ASD diagnosis, and children who haven’t received a diagnosis but have documented behaviors consistent with ASD.
In 2018, it was replaced with a less comprehensive approach that relies only on children with an ASD diagnosis. Data using this new methodology have not yet been reported.
In the new study from New Jersey, 767 children with autism were diagnosed by a pediatrician, neurologist, or other community provider. The remaining 175 children with autism, 18.6% of the total cohort, did not have an ASD diagnosis but met the ADDM case definition.
Under the new methodology, those children would not be counted.
“Something could be lost in the new methodology in terms of usefulness of the information because when the estimates are incomplete or low, that might lead people to make the wrong judgments when they make decisions about resources,” Dr. Zahorodny said.
The study was funded by the Centers for Disease Control and Prevention and the National Institutes of Health. The study authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ohio records more deaths than births for first time
In 2020, around 143,661 Ohioans died and 129,313 Ohioans were born, according to The Columbus Dispatch. The trend appears to have continued so far this year, with 107,462 deaths and 100,781 births reported to date.
Deaths haven’t surpassed births in the 112 years since the state began compiling data in 1909, the newspaper reported. The state’s birth rate has been declining for years while the number of deaths has risen, though data shows that the COVID-19 pandemic accelerated the shift.
“It doesn’t surprise me at all,” Joseph Gastaldo, MD, the medical director of infectious diseases for OhioHealth, told the newspaper.
Ohio’s birth rate fell by 4% in 2020, which defied public expectations about a pandemic birth boom. In addition, the state reported 13,927 COVID-19 deaths throughout the year.
“It’s COVID, clearly,” he noted.
Alabama also recorded more deaths than births for the first time last year, according to The New York Times. The state reported 64,714 deaths and 57,641 births in 2020.
“Our state literally shrunk in 2020,” Scott Harris, MD, the state health officer for Alabama, said at a news conference in September.
The state had never recorded a gap that large, even during World War I, World War II, and the 1918 flu pandemic, he said. Alabama has kept records on the numbers since 1900.
“We’ve never had a time when deaths exceeded births,” Dr. Harris said.
In fact, about half of U.S. states reported death rates higher than birth rates in 2020, according to a recent study from researchers at the University of New Hampshire. In 2019, only five states --Maine, New Hampshire, Rhode Island, Vermont, and West Virginia -- reported more deaths than births.
In 2020, the United States reported a record of nearly 3.4 million deaths, which was 18% more than in 2019, the researchers found. COVID-19 was the primary reason for the increase in deaths, accounting for about 11% of total deaths. Meanwhile, births dropped by 4% to about 3.6 million.
The surplus of births over deaths added 229,000 people to the U.S. population in 2020, as compared to 892,000 in 2019, which means the country’s population growth slowed last year. The decline, paired with lower immigration rates during the pandemic, led to the smallest annual percentage population gain in at least 100 years.
“Deaths will likely exceed births again in many states in 2021,” Kenneth Johnson, PhD, a senior demographer and study author, wrote in a statement.
“How large or protracted these fertility declines and mortality increases will be remains to be seen, but they have already dramatically reduced population growth in the United States,” he said.
A version of this article first appeared on WebMD.com.
In 2020, around 143,661 Ohioans died and 129,313 Ohioans were born, according to The Columbus Dispatch. The trend appears to have continued so far this year, with 107,462 deaths and 100,781 births reported to date.
Deaths haven’t surpassed births in the 112 years since the state began compiling data in 1909, the newspaper reported. The state’s birth rate has been declining for years while the number of deaths has risen, though data shows that the COVID-19 pandemic accelerated the shift.
“It doesn’t surprise me at all,” Joseph Gastaldo, MD, the medical director of infectious diseases for OhioHealth, told the newspaper.
Ohio’s birth rate fell by 4% in 2020, which defied public expectations about a pandemic birth boom. In addition, the state reported 13,927 COVID-19 deaths throughout the year.
“It’s COVID, clearly,” he noted.
Alabama also recorded more deaths than births for the first time last year, according to The New York Times. The state reported 64,714 deaths and 57,641 births in 2020.
“Our state literally shrunk in 2020,” Scott Harris, MD, the state health officer for Alabama, said at a news conference in September.
The state had never recorded a gap that large, even during World War I, World War II, and the 1918 flu pandemic, he said. Alabama has kept records on the numbers since 1900.
“We’ve never had a time when deaths exceeded births,” Dr. Harris said.
In fact, about half of U.S. states reported death rates higher than birth rates in 2020, according to a recent study from researchers at the University of New Hampshire. In 2019, only five states --Maine, New Hampshire, Rhode Island, Vermont, and West Virginia -- reported more deaths than births.
In 2020, the United States reported a record of nearly 3.4 million deaths, which was 18% more than in 2019, the researchers found. COVID-19 was the primary reason for the increase in deaths, accounting for about 11% of total deaths. Meanwhile, births dropped by 4% to about 3.6 million.
The surplus of births over deaths added 229,000 people to the U.S. population in 2020, as compared to 892,000 in 2019, which means the country’s population growth slowed last year. The decline, paired with lower immigration rates during the pandemic, led to the smallest annual percentage population gain in at least 100 years.
“Deaths will likely exceed births again in many states in 2021,” Kenneth Johnson, PhD, a senior demographer and study author, wrote in a statement.
“How large or protracted these fertility declines and mortality increases will be remains to be seen, but they have already dramatically reduced population growth in the United States,” he said.
A version of this article first appeared on WebMD.com.
In 2020, around 143,661 Ohioans died and 129,313 Ohioans were born, according to The Columbus Dispatch. The trend appears to have continued so far this year, with 107,462 deaths and 100,781 births reported to date.
Deaths haven’t surpassed births in the 112 years since the state began compiling data in 1909, the newspaper reported. The state’s birth rate has been declining for years while the number of deaths has risen, though data shows that the COVID-19 pandemic accelerated the shift.
“It doesn’t surprise me at all,” Joseph Gastaldo, MD, the medical director of infectious diseases for OhioHealth, told the newspaper.
Ohio’s birth rate fell by 4% in 2020, which defied public expectations about a pandemic birth boom. In addition, the state reported 13,927 COVID-19 deaths throughout the year.
“It’s COVID, clearly,” he noted.
Alabama also recorded more deaths than births for the first time last year, according to The New York Times. The state reported 64,714 deaths and 57,641 births in 2020.
“Our state literally shrunk in 2020,” Scott Harris, MD, the state health officer for Alabama, said at a news conference in September.
The state had never recorded a gap that large, even during World War I, World War II, and the 1918 flu pandemic, he said. Alabama has kept records on the numbers since 1900.
“We’ve never had a time when deaths exceeded births,” Dr. Harris said.
In fact, about half of U.S. states reported death rates higher than birth rates in 2020, according to a recent study from researchers at the University of New Hampshire. In 2019, only five states --Maine, New Hampshire, Rhode Island, Vermont, and West Virginia -- reported more deaths than births.
In 2020, the United States reported a record of nearly 3.4 million deaths, which was 18% more than in 2019, the researchers found. COVID-19 was the primary reason for the increase in deaths, accounting for about 11% of total deaths. Meanwhile, births dropped by 4% to about 3.6 million.
The surplus of births over deaths added 229,000 people to the U.S. population in 2020, as compared to 892,000 in 2019, which means the country’s population growth slowed last year. The decline, paired with lower immigration rates during the pandemic, led to the smallest annual percentage population gain in at least 100 years.
“Deaths will likely exceed births again in many states in 2021,” Kenneth Johnson, PhD, a senior demographer and study author, wrote in a statement.
“How large or protracted these fertility declines and mortality increases will be remains to be seen, but they have already dramatically reduced population growth in the United States,” he said.
A version of this article first appeared on WebMD.com.
Rash in an immunocompromised patient
The patient’s history and presentation, as well as a positive varicella zoster virus (VZV) culture of vesicular fluid, led to a diagnosis of disseminated herpes zoster (DHZ). The patient’s laboratory studies were remarkable for mild lymphopenia, thrombocytopenia, and elevated liver function tests. Shave and punch biopsies of the lesion showed ballooning epithelial necrosis with multinucleated giant cells.
DHZ is characterized by more than 20 vesicles outside of a primary or adjacent dermatome and is caused by extensive reactivation of VZV. DHZ is most often encountered in immunocompromised patients.1 Untreated DHZ can lead to encephalitis, myelitis, nerve palsies, pneumonitis, hepatitis, and ocular complications.
Diagnosis is usually made clinically and confirmed by a polymerase chain reaction test, direct fluorescent antibody testing, or viral culture.2 In immunocompetent patients, uncomplicated DHZ is treated with oral acyclovir 800 mg 5 times daily, valacyclovir 1 g 3 times daily, or famciclovir 500 mg 3 times daily for 7 days. Hospital admission for intravenous acyclovir is recommended for immunocompromised patients, especially those with internal organ involvement, such as hepatitis or encephalitis.3
Early treatment is imperative to avoid life-threatening complications. Active herpes zoster lesions are infectious by contact with vesicular fluid until they dry and crust over. Patients should be instructed to avoid contact with susceptible people, including those who are immunocompromised, babies who have not received their varicella vaccine, and pregnant women. It is important to keep DHZ on the differential—especially in an immunosuppressed patient with a diffuse vesicular rash—as it can lead to significant morbidity and mortality.
Our patient was treated with oral valacyclovir 1 g tid for 10 days and he improved without developing systemic symptoms.
Image courtesy of Christen B. Samaan, MD. Text courtesy of Christen B. Samaan, MD, and Matthew F. Helm, MD, Department of Dermatology, and Nanjiba Nawaz, BA, Penn State College of Medicine, Hershey.
1. Bollea-Garlatti ML, Bollea-Garlatti LA, Vacas AS, et al. Clinical characteristics and outcomes in a population with disseminated herpes zoster: a retrospective cohort study. Actas Dermosifiliogr. 2017;108:145-152. doi: 10.1016/j.ad.2016.10.009
2. Chiriac A, Chiriac AE, Podoleanu C, et al. Disseminated cutaneous herpes zoster—a frequently misdiagnosed entity. Int Wound J. 2020;17:1089-1091. doi: 10.1111/iwj.13370
3. Lewis DJ, Schlichte MJ, Dao H Jr. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100:321;324;330.

The patient’s history and presentation, as well as a positive varicella zoster virus (VZV) culture of vesicular fluid, led to a diagnosis of disseminated herpes zoster (DHZ). The patient’s laboratory studies were remarkable for mild lymphopenia, thrombocytopenia, and elevated liver function tests. Shave and punch biopsies of the lesion showed ballooning epithelial necrosis with multinucleated giant cells.
DHZ is characterized by more than 20 vesicles outside of a primary or adjacent dermatome and is caused by extensive reactivation of VZV. DHZ is most often encountered in immunocompromised patients.1 Untreated DHZ can lead to encephalitis, myelitis, nerve palsies, pneumonitis, hepatitis, and ocular complications.
Diagnosis is usually made clinically and confirmed by a polymerase chain reaction test, direct fluorescent antibody testing, or viral culture.2 In immunocompetent patients, uncomplicated DHZ is treated with oral acyclovir 800 mg 5 times daily, valacyclovir 1 g 3 times daily, or famciclovir 500 mg 3 times daily for 7 days. Hospital admission for intravenous acyclovir is recommended for immunocompromised patients, especially those with internal organ involvement, such as hepatitis or encephalitis.3
Early treatment is imperative to avoid life-threatening complications. Active herpes zoster lesions are infectious by contact with vesicular fluid until they dry and crust over. Patients should be instructed to avoid contact with susceptible people, including those who are immunocompromised, babies who have not received their varicella vaccine, and pregnant women. It is important to keep DHZ on the differential—especially in an immunosuppressed patient with a diffuse vesicular rash—as it can lead to significant morbidity and mortality.
Our patient was treated with oral valacyclovir 1 g tid for 10 days and he improved without developing systemic symptoms.
Image courtesy of Christen B. Samaan, MD. Text courtesy of Christen B. Samaan, MD, and Matthew F. Helm, MD, Department of Dermatology, and Nanjiba Nawaz, BA, Penn State College of Medicine, Hershey.

The patient’s history and presentation, as well as a positive varicella zoster virus (VZV) culture of vesicular fluid, led to a diagnosis of disseminated herpes zoster (DHZ). The patient’s laboratory studies were remarkable for mild lymphopenia, thrombocytopenia, and elevated liver function tests. Shave and punch biopsies of the lesion showed ballooning epithelial necrosis with multinucleated giant cells.
DHZ is characterized by more than 20 vesicles outside of a primary or adjacent dermatome and is caused by extensive reactivation of VZV. DHZ is most often encountered in immunocompromised patients.1 Untreated DHZ can lead to encephalitis, myelitis, nerve palsies, pneumonitis, hepatitis, and ocular complications.
Diagnosis is usually made clinically and confirmed by a polymerase chain reaction test, direct fluorescent antibody testing, or viral culture.2 In immunocompetent patients, uncomplicated DHZ is treated with oral acyclovir 800 mg 5 times daily, valacyclovir 1 g 3 times daily, or famciclovir 500 mg 3 times daily for 7 days. Hospital admission for intravenous acyclovir is recommended for immunocompromised patients, especially those with internal organ involvement, such as hepatitis or encephalitis.3
Early treatment is imperative to avoid life-threatening complications. Active herpes zoster lesions are infectious by contact with vesicular fluid until they dry and crust over. Patients should be instructed to avoid contact with susceptible people, including those who are immunocompromised, babies who have not received their varicella vaccine, and pregnant women. It is important to keep DHZ on the differential—especially in an immunosuppressed patient with a diffuse vesicular rash—as it can lead to significant morbidity and mortality.
Our patient was treated with oral valacyclovir 1 g tid for 10 days and he improved without developing systemic symptoms.
Image courtesy of Christen B. Samaan, MD. Text courtesy of Christen B. Samaan, MD, and Matthew F. Helm, MD, Department of Dermatology, and Nanjiba Nawaz, BA, Penn State College of Medicine, Hershey.
1. Bollea-Garlatti ML, Bollea-Garlatti LA, Vacas AS, et al. Clinical characteristics and outcomes in a population with disseminated herpes zoster: a retrospective cohort study. Actas Dermosifiliogr. 2017;108:145-152. doi: 10.1016/j.ad.2016.10.009
2. Chiriac A, Chiriac AE, Podoleanu C, et al. Disseminated cutaneous herpes zoster—a frequently misdiagnosed entity. Int Wound J. 2020;17:1089-1091. doi: 10.1111/iwj.13370
3. Lewis DJ, Schlichte MJ, Dao H Jr. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100:321;324;330.
1. Bollea-Garlatti ML, Bollea-Garlatti LA, Vacas AS, et al. Clinical characteristics and outcomes in a population with disseminated herpes zoster: a retrospective cohort study. Actas Dermosifiliogr. 2017;108:145-152. doi: 10.1016/j.ad.2016.10.009
2. Chiriac A, Chiriac AE, Podoleanu C, et al. Disseminated cutaneous herpes zoster—a frequently misdiagnosed entity. Int Wound J. 2020;17:1089-1091. doi: 10.1111/iwj.13370
3. Lewis DJ, Schlichte MJ, Dao H Jr. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100:321;324;330.
Hot temperatures in outdoor lockboxes increase sample errors
, according to results from a recent study published in the American Journal of Clinical Pathology.
“Our findings indicate that samples (centrifuged or not centrifuged) were impacted by extreme summer temperatures when stored for short periods of time inside commonly used steel lockboxes,” Joseph R. Wiencek, PhD, medical director of clinical chemistry, Vanderbilt University School of Medicine Core Laboratory in Nashville, said in an interview.
Dr. Wiencek and colleagues picked two dates during the summer of 2019 in a mid-Atlantic state to place two courier lockboxes (LabLocker-KF300) outside in hot temperatures (32º C) starting at 11 a.m., with one lockbox containing two 24-oz cold packs (Nordic NI24) and the other containing no cold packs. The researchers monitored the temperatures of each lockbox over the course of 4 hours.
Overall, eight participants had seven samples in lithium heparin drawn for two studies evaluating centrifuged or not centrifuged samples. In the first study, four participants had seven samples drawn, with one centrifuged sample serving as a control for each patient. The other six samples were wrapped in paper towels, placed in resealable plastic bags, and distributed evenly in the warm and cold lockboxes. The samples did not directly touch the cold packs in the cold lockbox. At 1 hour, 2 hours, and 4 hours, a participant’s sample was removed from each lockbox and centrifuged.
In the second study, another four participants had seven samples drawn. As in the first study, all samples were centrifuged and placed in the lockboxes. For both studies, when samples were centrifuged, plasma from samples was left on the gel barrier when analyzed for concentrations of C-reactive protein, a comprehensive metabolic panel, lactate dehydrogenase (LDH), a lipid panel, magnesium, and phosphorus (Abbott Architect c16000).
In the study of uncentrifuged samples, Dr. Wiencek and colleagues found that when the temperature outside ranged from 28.2º to 44.0º C (mean 40.4º C), the temperature of the cold lockbox was between 16.5º to 22.3º C (mean 22.3º C). The temperature ranged between 34.4º to 46.9º C (mean 42.6º C) in the warm lockbox. For centrifuged samples, the cold lockbox temperature was between 12.2º to 23.0º C (mean 18.0º C) and the warm lockbox was between 25. to 40.8º C (mean 35.2º C) when the outdoor temperature ranged from 27.2º to 46.3º C (mean 37.9º C).
The researchers also calculated the significant change limit (SCL) for each analyte in each sample, finding that aspartate aminotransferase, glucose, LDH, and potassium significantly exceeded the SCL in both the centrifuged and uncentrifuged samples, with the greatest changes seen at the 4-hour timepoint for samples in the warm lockbox (P < .05 for all).
Lockbox instructions are “consistently inconsistent”
In viewing instructions for lockboxes across institutions, Dr. Wiencek said the “outdoor courier lockbox instructions among private, academic and reference laboratories were consistently inconsistent.” For example, no laboratories cited time restrictions for samples in lockboxes, and their descriptions on the number of cold packs a laboratory should use and where the lockbox should be placed varied. The inconsistencies “highlighted the emergent need for standardization and guidance documents for institutions to implement,” Dr. Wiencek said.
One unanswered question is how widespread the problem is. It is unclear how many outdoor courier lockboxes are currently in use in the United States or globally; however, experts agreed it was a common occurrence, with some of the largest laboratory service providers offering outdoor courier lockboxes to their clients.
“Courier lockboxes are everywhere. All you need to do is walk around your clinics that are at your hospitals or clinics located around your grocery store to find them,” Dr. Wiencek said. “Some hang on doors, while others can be found on the ground in direct sunlight on a hot summer day.”
What’s more, institutions may not realize how leaving samples outdoors for extended periods can affect results. “Care teams are commonly unaware that samples placed in these poorly designed lockboxes can experience extreme summer or winter temperatures that may lead to incorrect results,” Dr. Wiencek said. “Healthcare providers need to understand the hidden dangers courier lockboxes have on the quality of their patient’s test results.”
Amy L. Pyle-Eilola, PhD, clinical chemistry director at Nationwide Children’s Hospital in Columbus, Ohio, said a major strength of the study by Dr. Wiencek and colleagues “is just that it was done at all.”
“I appreciate the real-world nature of this study and that it provides a snapshot of what conditions are really like in a lockbox in the summer,” she said in an interview.
In the clinical lab, receiving samples that had been sitting in a courier lockbox “is not uncommon,” Dr. Pyle-Eilola said.
“When I have encountered these situations, I have struggled to decide if it is still appropriate to run the tests. I always look to the medical literature for assistance with these situations, but there has been a paucity of information available on the impact of lockbox storage,” she explained.
The study by Dr. Wiencek and colleagues “provides some much-needed evidence for what is acceptable for lockbox storage conditions,” she said.
Areas of future research
Rodney E. Rohde, PhD, university distinguished chair and professor of the Clinical Laboratory Science (CLS) Program at Texas State University in San Marcos, said in an interview that the study “does a nice job of looking at multiple analytes and controlling for several variables,” but the sample size is small and the results may be difficult to generalize.
Dr. Pyle-Eilola highlighted another limitation — “a common shortcoming of these kinds of studies” — in the use of healthy donors for patient samples, which narrows the range of assay results.
“It is possible that more significant variation in results may be observed in additional analytes if the samples had higher concentrations of those analytes,” she said. “Moreover, this is clinically relevant as the samples stored in such lockboxes are not always from healthy individuals and have abnormal concentrations of analytes.”
Mario Plebani, MD, professor of clinical biochemistry and clinical molecular biology and chief of the department of laboratory medicine at University Hospital of Padova in Padova, Italy, agreed with that assessment.
“[T]he risks for errors and patient safety are higher for values near to the upper or lower reference value, and in general for samples collected in patients with particular diseases and clinical conditions,” he said in an interview.
“This paper deserves a commenting editorial to better highlight the urgent need for further studies on the same issue and in general on the risk in the pre-pre-analytical phase, including sample storage and transportation,” he noted.
Another area of future research is studying patient samples exposed to hotter or colder temperatures in outdoor courier lockboxes outside the mid-Atlantic area. “Here in Texas, temperatures can reach extreme heat levels,” Dr. Rohde said, who added that use of outdoor lockboxes is “very common in my region.”
Dr. Wiencek disclosed he has been a consultant on this research topic for Roche Diagnostics and received an honorarium for speaking on the subject from the American Association for Clinical Chemistry and American Society of Clinical Pathology. The other authors have no relevant conflict of interest. Dr. Pyle-Eilola, Dr. Rohde, and Dr. Plebani have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to results from a recent study published in the American Journal of Clinical Pathology.
“Our findings indicate that samples (centrifuged or not centrifuged) were impacted by extreme summer temperatures when stored for short periods of time inside commonly used steel lockboxes,” Joseph R. Wiencek, PhD, medical director of clinical chemistry, Vanderbilt University School of Medicine Core Laboratory in Nashville, said in an interview.
Dr. Wiencek and colleagues picked two dates during the summer of 2019 in a mid-Atlantic state to place two courier lockboxes (LabLocker-KF300) outside in hot temperatures (32º C) starting at 11 a.m., with one lockbox containing two 24-oz cold packs (Nordic NI24) and the other containing no cold packs. The researchers monitored the temperatures of each lockbox over the course of 4 hours.
Overall, eight participants had seven samples in lithium heparin drawn for two studies evaluating centrifuged or not centrifuged samples. In the first study, four participants had seven samples drawn, with one centrifuged sample serving as a control for each patient. The other six samples were wrapped in paper towels, placed in resealable plastic bags, and distributed evenly in the warm and cold lockboxes. The samples did not directly touch the cold packs in the cold lockbox. At 1 hour, 2 hours, and 4 hours, a participant’s sample was removed from each lockbox and centrifuged.
In the second study, another four participants had seven samples drawn. As in the first study, all samples were centrifuged and placed in the lockboxes. For both studies, when samples were centrifuged, plasma from samples was left on the gel barrier when analyzed for concentrations of C-reactive protein, a comprehensive metabolic panel, lactate dehydrogenase (LDH), a lipid panel, magnesium, and phosphorus (Abbott Architect c16000).
In the study of uncentrifuged samples, Dr. Wiencek and colleagues found that when the temperature outside ranged from 28.2º to 44.0º C (mean 40.4º C), the temperature of the cold lockbox was between 16.5º to 22.3º C (mean 22.3º C). The temperature ranged between 34.4º to 46.9º C (mean 42.6º C) in the warm lockbox. For centrifuged samples, the cold lockbox temperature was between 12.2º to 23.0º C (mean 18.0º C) and the warm lockbox was between 25. to 40.8º C (mean 35.2º C) when the outdoor temperature ranged from 27.2º to 46.3º C (mean 37.9º C).
The researchers also calculated the significant change limit (SCL) for each analyte in each sample, finding that aspartate aminotransferase, glucose, LDH, and potassium significantly exceeded the SCL in both the centrifuged and uncentrifuged samples, with the greatest changes seen at the 4-hour timepoint for samples in the warm lockbox (P < .05 for all).
Lockbox instructions are “consistently inconsistent”
In viewing instructions for lockboxes across institutions, Dr. Wiencek said the “outdoor courier lockbox instructions among private, academic and reference laboratories were consistently inconsistent.” For example, no laboratories cited time restrictions for samples in lockboxes, and their descriptions on the number of cold packs a laboratory should use and where the lockbox should be placed varied. The inconsistencies “highlighted the emergent need for standardization and guidance documents for institutions to implement,” Dr. Wiencek said.
One unanswered question is how widespread the problem is. It is unclear how many outdoor courier lockboxes are currently in use in the United States or globally; however, experts agreed it was a common occurrence, with some of the largest laboratory service providers offering outdoor courier lockboxes to their clients.
“Courier lockboxes are everywhere. All you need to do is walk around your clinics that are at your hospitals or clinics located around your grocery store to find them,” Dr. Wiencek said. “Some hang on doors, while others can be found on the ground in direct sunlight on a hot summer day.”
What’s more, institutions may not realize how leaving samples outdoors for extended periods can affect results. “Care teams are commonly unaware that samples placed in these poorly designed lockboxes can experience extreme summer or winter temperatures that may lead to incorrect results,” Dr. Wiencek said. “Healthcare providers need to understand the hidden dangers courier lockboxes have on the quality of their patient’s test results.”
Amy L. Pyle-Eilola, PhD, clinical chemistry director at Nationwide Children’s Hospital in Columbus, Ohio, said a major strength of the study by Dr. Wiencek and colleagues “is just that it was done at all.”
“I appreciate the real-world nature of this study and that it provides a snapshot of what conditions are really like in a lockbox in the summer,” she said in an interview.
In the clinical lab, receiving samples that had been sitting in a courier lockbox “is not uncommon,” Dr. Pyle-Eilola said.
“When I have encountered these situations, I have struggled to decide if it is still appropriate to run the tests. I always look to the medical literature for assistance with these situations, but there has been a paucity of information available on the impact of lockbox storage,” she explained.
The study by Dr. Wiencek and colleagues “provides some much-needed evidence for what is acceptable for lockbox storage conditions,” she said.
Areas of future research
Rodney E. Rohde, PhD, university distinguished chair and professor of the Clinical Laboratory Science (CLS) Program at Texas State University in San Marcos, said in an interview that the study “does a nice job of looking at multiple analytes and controlling for several variables,” but the sample size is small and the results may be difficult to generalize.
Dr. Pyle-Eilola highlighted another limitation — “a common shortcoming of these kinds of studies” — in the use of healthy donors for patient samples, which narrows the range of assay results.
“It is possible that more significant variation in results may be observed in additional analytes if the samples had higher concentrations of those analytes,” she said. “Moreover, this is clinically relevant as the samples stored in such lockboxes are not always from healthy individuals and have abnormal concentrations of analytes.”
Mario Plebani, MD, professor of clinical biochemistry and clinical molecular biology and chief of the department of laboratory medicine at University Hospital of Padova in Padova, Italy, agreed with that assessment.
“[T]he risks for errors and patient safety are higher for values near to the upper or lower reference value, and in general for samples collected in patients with particular diseases and clinical conditions,” he said in an interview.
“This paper deserves a commenting editorial to better highlight the urgent need for further studies on the same issue and in general on the risk in the pre-pre-analytical phase, including sample storage and transportation,” he noted.
Another area of future research is studying patient samples exposed to hotter or colder temperatures in outdoor courier lockboxes outside the mid-Atlantic area. “Here in Texas, temperatures can reach extreme heat levels,” Dr. Rohde said, who added that use of outdoor lockboxes is “very common in my region.”
Dr. Wiencek disclosed he has been a consultant on this research topic for Roche Diagnostics and received an honorarium for speaking on the subject from the American Association for Clinical Chemistry and American Society of Clinical Pathology. The other authors have no relevant conflict of interest. Dr. Pyle-Eilola, Dr. Rohde, and Dr. Plebani have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to results from a recent study published in the American Journal of Clinical Pathology.
“Our findings indicate that samples (centrifuged or not centrifuged) were impacted by extreme summer temperatures when stored for short periods of time inside commonly used steel lockboxes,” Joseph R. Wiencek, PhD, medical director of clinical chemistry, Vanderbilt University School of Medicine Core Laboratory in Nashville, said in an interview.
Dr. Wiencek and colleagues picked two dates during the summer of 2019 in a mid-Atlantic state to place two courier lockboxes (LabLocker-KF300) outside in hot temperatures (32º C) starting at 11 a.m., with one lockbox containing two 24-oz cold packs (Nordic NI24) and the other containing no cold packs. The researchers monitored the temperatures of each lockbox over the course of 4 hours.
Overall, eight participants had seven samples in lithium heparin drawn for two studies evaluating centrifuged or not centrifuged samples. In the first study, four participants had seven samples drawn, with one centrifuged sample serving as a control for each patient. The other six samples were wrapped in paper towels, placed in resealable plastic bags, and distributed evenly in the warm and cold lockboxes. The samples did not directly touch the cold packs in the cold lockbox. At 1 hour, 2 hours, and 4 hours, a participant’s sample was removed from each lockbox and centrifuged.
In the second study, another four participants had seven samples drawn. As in the first study, all samples were centrifuged and placed in the lockboxes. For both studies, when samples were centrifuged, plasma from samples was left on the gel barrier when analyzed for concentrations of C-reactive protein, a comprehensive metabolic panel, lactate dehydrogenase (LDH), a lipid panel, magnesium, and phosphorus (Abbott Architect c16000).
In the study of uncentrifuged samples, Dr. Wiencek and colleagues found that when the temperature outside ranged from 28.2º to 44.0º C (mean 40.4º C), the temperature of the cold lockbox was between 16.5º to 22.3º C (mean 22.3º C). The temperature ranged between 34.4º to 46.9º C (mean 42.6º C) in the warm lockbox. For centrifuged samples, the cold lockbox temperature was between 12.2º to 23.0º C (mean 18.0º C) and the warm lockbox was between 25. to 40.8º C (mean 35.2º C) when the outdoor temperature ranged from 27.2º to 46.3º C (mean 37.9º C).
The researchers also calculated the significant change limit (SCL) for each analyte in each sample, finding that aspartate aminotransferase, glucose, LDH, and potassium significantly exceeded the SCL in both the centrifuged and uncentrifuged samples, with the greatest changes seen at the 4-hour timepoint for samples in the warm lockbox (P < .05 for all).
Lockbox instructions are “consistently inconsistent”
In viewing instructions for lockboxes across institutions, Dr. Wiencek said the “outdoor courier lockbox instructions among private, academic and reference laboratories were consistently inconsistent.” For example, no laboratories cited time restrictions for samples in lockboxes, and their descriptions on the number of cold packs a laboratory should use and where the lockbox should be placed varied. The inconsistencies “highlighted the emergent need for standardization and guidance documents for institutions to implement,” Dr. Wiencek said.
One unanswered question is how widespread the problem is. It is unclear how many outdoor courier lockboxes are currently in use in the United States or globally; however, experts agreed it was a common occurrence, with some of the largest laboratory service providers offering outdoor courier lockboxes to their clients.
“Courier lockboxes are everywhere. All you need to do is walk around your clinics that are at your hospitals or clinics located around your grocery store to find them,” Dr. Wiencek said. “Some hang on doors, while others can be found on the ground in direct sunlight on a hot summer day.”
What’s more, institutions may not realize how leaving samples outdoors for extended periods can affect results. “Care teams are commonly unaware that samples placed in these poorly designed lockboxes can experience extreme summer or winter temperatures that may lead to incorrect results,” Dr. Wiencek said. “Healthcare providers need to understand the hidden dangers courier lockboxes have on the quality of their patient’s test results.”
Amy L. Pyle-Eilola, PhD, clinical chemistry director at Nationwide Children’s Hospital in Columbus, Ohio, said a major strength of the study by Dr. Wiencek and colleagues “is just that it was done at all.”
“I appreciate the real-world nature of this study and that it provides a snapshot of what conditions are really like in a lockbox in the summer,” she said in an interview.
In the clinical lab, receiving samples that had been sitting in a courier lockbox “is not uncommon,” Dr. Pyle-Eilola said.
“When I have encountered these situations, I have struggled to decide if it is still appropriate to run the tests. I always look to the medical literature for assistance with these situations, but there has been a paucity of information available on the impact of lockbox storage,” she explained.
The study by Dr. Wiencek and colleagues “provides some much-needed evidence for what is acceptable for lockbox storage conditions,” she said.
Areas of future research
Rodney E. Rohde, PhD, university distinguished chair and professor of the Clinical Laboratory Science (CLS) Program at Texas State University in San Marcos, said in an interview that the study “does a nice job of looking at multiple analytes and controlling for several variables,” but the sample size is small and the results may be difficult to generalize.
Dr. Pyle-Eilola highlighted another limitation — “a common shortcoming of these kinds of studies” — in the use of healthy donors for patient samples, which narrows the range of assay results.
“It is possible that more significant variation in results may be observed in additional analytes if the samples had higher concentrations of those analytes,” she said. “Moreover, this is clinically relevant as the samples stored in such lockboxes are not always from healthy individuals and have abnormal concentrations of analytes.”
Mario Plebani, MD, professor of clinical biochemistry and clinical molecular biology and chief of the department of laboratory medicine at University Hospital of Padova in Padova, Italy, agreed with that assessment.
“[T]he risks for errors and patient safety are higher for values near to the upper or lower reference value, and in general for samples collected in patients with particular diseases and clinical conditions,” he said in an interview.
“This paper deserves a commenting editorial to better highlight the urgent need for further studies on the same issue and in general on the risk in the pre-pre-analytical phase, including sample storage and transportation,” he noted.
Another area of future research is studying patient samples exposed to hotter or colder temperatures in outdoor courier lockboxes outside the mid-Atlantic area. “Here in Texas, temperatures can reach extreme heat levels,” Dr. Rohde said, who added that use of outdoor lockboxes is “very common in my region.”
Dr. Wiencek disclosed he has been a consultant on this research topic for Roche Diagnostics and received an honorarium for speaking on the subject from the American Association for Clinical Chemistry and American Society of Clinical Pathology. The other authors have no relevant conflict of interest. Dr. Pyle-Eilola, Dr. Rohde, and Dr. Plebani have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinical Edge Journal Scan Commentary: CRC November 2021
The tide may finally be shifting as drugs targeting specific KRAS mutations have made their way into the clinic, and there is particular excitement around the oral WEE1 inhibitor, adavosertib (AZD1775). WEE1 is a cell cycle regulatory protein and WEE1 inhibition may have increased activity in tumors with DNA repair deficiency. In a phase 2 maintenance study, Seligmann and colleagues randomized 69 patients with RAS- and TP53-mutated mCRC with stable disease or better after 16 weeks of induction chemotherapy 2:1 to receive adavosertib vs active monitoring. Median progression-free survival (mPFS) was significant improved with adavosertib (3.61 vs 1.68 months), and patients with left-sided primary tumors appears to derive more benefit. While this finding needs to be further explored in larger clinical trials, it is exciting that there may finally be a new treatment option for patients with this specific molecular subtype of mCRC.
Another maintenance mCRC trial of interest is the PANAMA trial, a phase 2 study in which 248 patients with RAS wild-type mCRC were randomized 1:1 to 5-fluorouracil/leucovorin with or without panitumumab, an anti-epidermal growth factor receptor antibody, following induction chemotherapy with 6 cycles FOLFOX/panitumumab. Modest and co-workers report that mPFS was significantly improved with continuing panitumumab in the maintenance setting (8.8 vs. 5.7 months), and there was a trend towards an overall survival benefit as well. This study further supports continuing anti-EGFR therapy with maintenance chemotherapy for patients with RAS wild-type mCRC.
Finally, in stage III CRC, there is a big movement towards using circulating tumor DNA (ctDNA) as a method to monitor disease recurrence by detecting minimal residual disease based on tumor DNA being shed into the bloodstream. Henriksen et al. evaluated 168 patients with stage III CRC who underwent surgical resection and plasma ctDNA testing using 16 patient-specific DNA variants (tumor tissue-informed testing). The rates of recurrence were much higher in patients with detectable ctDNA post-operatively and/or after the completion of adjuvant chemotherapy, whereas those patients with persistently undetectable ctDNA did not recur. ctDNA is a powerful new technology that we are still learning how to best harness in the clinic, and this study demonstrates its prognostic value and potential ability to detect recurrence prior to standard imaging surveillance. Moreover, the rate of ctDNA rise was also prognostic of survival. ctDNA testing is likely to become standard of care in the management of stage II/III colorectal cancer in the very near future, and we hope that eventually it may be able to predict who needs to receive adjuvant chemotherapy and who does not.
The tide may finally be shifting as drugs targeting specific KRAS mutations have made their way into the clinic, and there is particular excitement around the oral WEE1 inhibitor, adavosertib (AZD1775). WEE1 is a cell cycle regulatory protein and WEE1 inhibition may have increased activity in tumors with DNA repair deficiency. In a phase 2 maintenance study, Seligmann and colleagues randomized 69 patients with RAS- and TP53-mutated mCRC with stable disease or better after 16 weeks of induction chemotherapy 2:1 to receive adavosertib vs active monitoring. Median progression-free survival (mPFS) was significant improved with adavosertib (3.61 vs 1.68 months), and patients with left-sided primary tumors appears to derive more benefit. While this finding needs to be further explored in larger clinical trials, it is exciting that there may finally be a new treatment option for patients with this specific molecular subtype of mCRC.
Another maintenance mCRC trial of interest is the PANAMA trial, a phase 2 study in which 248 patients with RAS wild-type mCRC were randomized 1:1 to 5-fluorouracil/leucovorin with or without panitumumab, an anti-epidermal growth factor receptor antibody, following induction chemotherapy with 6 cycles FOLFOX/panitumumab. Modest and co-workers report that mPFS was significantly improved with continuing panitumumab in the maintenance setting (8.8 vs. 5.7 months), and there was a trend towards an overall survival benefit as well. This study further supports continuing anti-EGFR therapy with maintenance chemotherapy for patients with RAS wild-type mCRC.
Finally, in stage III CRC, there is a big movement towards using circulating tumor DNA (ctDNA) as a method to monitor disease recurrence by detecting minimal residual disease based on tumor DNA being shed into the bloodstream. Henriksen et al. evaluated 168 patients with stage III CRC who underwent surgical resection and plasma ctDNA testing using 16 patient-specific DNA variants (tumor tissue-informed testing). The rates of recurrence were much higher in patients with detectable ctDNA post-operatively and/or after the completion of adjuvant chemotherapy, whereas those patients with persistently undetectable ctDNA did not recur. ctDNA is a powerful new technology that we are still learning how to best harness in the clinic, and this study demonstrates its prognostic value and potential ability to detect recurrence prior to standard imaging surveillance. Moreover, the rate of ctDNA rise was also prognostic of survival. ctDNA testing is likely to become standard of care in the management of stage II/III colorectal cancer in the very near future, and we hope that eventually it may be able to predict who needs to receive adjuvant chemotherapy and who does not.
The tide may finally be shifting as drugs targeting specific KRAS mutations have made their way into the clinic, and there is particular excitement around the oral WEE1 inhibitor, adavosertib (AZD1775). WEE1 is a cell cycle regulatory protein and WEE1 inhibition may have increased activity in tumors with DNA repair deficiency. In a phase 2 maintenance study, Seligmann and colleagues randomized 69 patients with RAS- and TP53-mutated mCRC with stable disease or better after 16 weeks of induction chemotherapy 2:1 to receive adavosertib vs active monitoring. Median progression-free survival (mPFS) was significant improved with adavosertib (3.61 vs 1.68 months), and patients with left-sided primary tumors appears to derive more benefit. While this finding needs to be further explored in larger clinical trials, it is exciting that there may finally be a new treatment option for patients with this specific molecular subtype of mCRC.
Another maintenance mCRC trial of interest is the PANAMA trial, a phase 2 study in which 248 patients with RAS wild-type mCRC were randomized 1:1 to 5-fluorouracil/leucovorin with or without panitumumab, an anti-epidermal growth factor receptor antibody, following induction chemotherapy with 6 cycles FOLFOX/panitumumab. Modest and co-workers report that mPFS was significantly improved with continuing panitumumab in the maintenance setting (8.8 vs. 5.7 months), and there was a trend towards an overall survival benefit as well. This study further supports continuing anti-EGFR therapy with maintenance chemotherapy for patients with RAS wild-type mCRC.
Finally, in stage III CRC, there is a big movement towards using circulating tumor DNA (ctDNA) as a method to monitor disease recurrence by detecting minimal residual disease based on tumor DNA being shed into the bloodstream. Henriksen et al. evaluated 168 patients with stage III CRC who underwent surgical resection and plasma ctDNA testing using 16 patient-specific DNA variants (tumor tissue-informed testing). The rates of recurrence were much higher in patients with detectable ctDNA post-operatively and/or after the completion of adjuvant chemotherapy, whereas those patients with persistently undetectable ctDNA did not recur. ctDNA is a powerful new technology that we are still learning how to best harness in the clinic, and this study demonstrates its prognostic value and potential ability to detect recurrence prior to standard imaging surveillance. Moreover, the rate of ctDNA rise was also prognostic of survival. ctDNA testing is likely to become standard of care in the management of stage II/III colorectal cancer in the very near future, and we hope that eventually it may be able to predict who needs to receive adjuvant chemotherapy and who does not.