User login
First-line AFib ablation cuts risk of progression vs. drug therapy
CHICAGO – Early ablation of atrial fibrillation (AFib) reduces the risk of progression, compared with antiarrhythmic therapies, according to results of a multicenter, randomized trial called PROGRESSIVE-AF.
Over 36 months of follow-up, the trial linked early ablation with a reduced risk of persistent AFib (1.9% vs. 7.4%), and in addition, those in the ablation group were less likely to have recurrent atrial tachyarrhythmias of any kind (56.5% vs. 77.2%), reported Jason G. Andrade, MD, at the American Heart Association scientific sessions.
Serving as a long-term extension of the EARLY-AF trial published almost 2 years ago, this trial expands evidence that progressive AFib can be attenuated, a concept that has been debated.
“Can early AFib ablation stop progression?” asked Carina Blomström-Lindqvist, MD, PhD. The invited discussant for the PROGRESSION-AF trial, Dr. Blomström-Lundqvist concluded, “here is another set of data that suggests it can.”
By another set of data, Dr. Blomström-Lindqvist was referring to a previously published multinational study called ATTEST In this study, which involved 29 sites worldwide and compared radiofrequency ablation to antiarrhythmic drug therapy, early ablation also produced a lower risk of persistent AFib at the end of 3 years (2.4% vs. 17.5%; P = .0009).
In the previously published open-label EARLY-AF trial, 303 patients with paroxysmal, untreated AFib were randomized to cryoballoon ablation or antiarrhythmic drugs. The primary endpoint was the first documented recurrence of an atrial tachyarrhythmia between 91 and 365 days. The lower rate following ablation (42.9% vs. 67.8%) represented a more than 50% reduction in risk (hazard ratio, 0.48; P < .001) relative to antiarrhythmic therapy.
In PROGRESSIVE-AF, the same 303 patients were monitored continuously for an additional 24 months with an implanted cardiac monitor programmed with an AFib-detection algorithm. The data from the monitor were obtained daily. Over the final 2 years of the study, office visits were conducted every 6 months.
Tachyarrhythmias represent primary endpoint
In addition to persistent AFib, defined as lasting ≥ 7 days or lasting 48 hours to 7 days but requiring cardioversion for termination, patients in PROGRESSIVE-AF were also monitored for recurrent atrial tachyarrhythmias, AFib burden, quality of life (QOL), and health care utilization, and safety.
The average age was roughly 58 years. Although more than one-third had hypertension, most had no other comorbidities. The authors emphasized that the study population overall was relatively young and healthy.
Those randomized to antiarrhythmic therapy in EARLY-AF/PROGRESSIVE-AF received commonly prescribed therapies titrated to maximally tolerated doses using standardized protocols. At the start of EARLY-AF, flecainide, taken by 65% of patients, was the most commonly used agent, followed by sotalol, propafenone, dronedarone, and amiodarone.
At the end of PROGRESSIVE-AF, the order of the most common therapies did not change relative to EARLY-AF, but only 49% of patients were taking flecainide and 31% were no longer taking any antiarrhythmic therapy.
At the end of 3 years of follow-up in EARLY-AF/PROGRESSIVE-AF, the difference in persistent AFib represented a 75% reduction in favor of early ablation (HR, 0.25; 95% confidence interval, 0.09-0.70).
In those treated with ablation relative to those treated with antiarrhythmic therapy, the lower rate of atrial tachyarrhythmia lasting more than 7 days (1.9% vs. 6.0%) represented a 70% risk reduction (HR, 0.30; 95% CI 0.10-0.93). The protection from cardioversion for atrial tachyarrhythmia lasting between 2 and 7 days in duration (0.6% vs. 4.7%) translated into an 86% relative reduction (HR, 0.14; 95% CI, 0.02-0.85).
The impact on QOL for those randomized to ablation, which was measured with both AFib-specific and generic measures, was meaningful to patients, according to Dr. Andrade, director of the Cardiac Electrophysiology Laboratory, Vancouver General Hospital.
For example, the mean difference in the AF Quality of Life Survey (AFEQT), was 8.0 at 1 year and 7.4 at 3 years in favor of ablation. A change of 5 points in this score is considered to be a clinically meaningful difference, according to Dr. Andrade.
Numerically, the relative risk of emergency room visits and cardioversion were lower in the ablation group, but the differences did not reach statistical significance. However, the lower hazard ratio for hospitalization was significant (HR, 0.31; 95% CI, 0.15-0.66), supporting a reduction in consumption of health care resources.
Ablation found safer than drugs
The rate of adverse events of any kind (11.0% vs. 23.5%) and serious adverse events (4.5% vs. 10.1%) were lower in the ablation group.
There were no differences in major adverse cardiovascular events observed in this period of follow-up, but Dr. Andrade pointed out that follow-up was not long enough to expect differences in these events.
Impressed by the magnitude of the reduction in persistent AFib in a population of relatively young and healthy patients considered to be at a low risk of AFib progression, Dr. Blomström-Lindqvist, a professor of cardiology at the Institution of Medical Science, Uppsala, Sweden, indicated that the data support early ablation as a means to reduce risk of this outcome.
However, she did caution that progressive AFib was observed in a relatively small proportion of patients managed with antiarrhythmic therapy at 3 years, an outcome relevant when discussing treatment options with patients.
The results were published in New England Journal of Medicine simultaneously with Dr. Andrade’s presentation.
Dr. Andrade reports financial relationships with Bayer, Bayliss, Biosense, Bristol-Myers Squibb, Medtronic and Servier. The trial, funded largely by the Canadian government and Canadian professional societies, received additional funding from Bayliss and Medtronic. Dr. Blomström-Lundqvist reports financial relationships with Bayer, Boston Scientific, Cathprint, Medtronic, and Sanofi.
CHICAGO – Early ablation of atrial fibrillation (AFib) reduces the risk of progression, compared with antiarrhythmic therapies, according to results of a multicenter, randomized trial called PROGRESSIVE-AF.
Over 36 months of follow-up, the trial linked early ablation with a reduced risk of persistent AFib (1.9% vs. 7.4%), and in addition, those in the ablation group were less likely to have recurrent atrial tachyarrhythmias of any kind (56.5% vs. 77.2%), reported Jason G. Andrade, MD, at the American Heart Association scientific sessions.
Serving as a long-term extension of the EARLY-AF trial published almost 2 years ago, this trial expands evidence that progressive AFib can be attenuated, a concept that has been debated.
“Can early AFib ablation stop progression?” asked Carina Blomström-Lindqvist, MD, PhD. The invited discussant for the PROGRESSION-AF trial, Dr. Blomström-Lundqvist concluded, “here is another set of data that suggests it can.”
By another set of data, Dr. Blomström-Lindqvist was referring to a previously published multinational study called ATTEST In this study, which involved 29 sites worldwide and compared radiofrequency ablation to antiarrhythmic drug therapy, early ablation also produced a lower risk of persistent AFib at the end of 3 years (2.4% vs. 17.5%; P = .0009).
In the previously published open-label EARLY-AF trial, 303 patients with paroxysmal, untreated AFib were randomized to cryoballoon ablation or antiarrhythmic drugs. The primary endpoint was the first documented recurrence of an atrial tachyarrhythmia between 91 and 365 days. The lower rate following ablation (42.9% vs. 67.8%) represented a more than 50% reduction in risk (hazard ratio, 0.48; P < .001) relative to antiarrhythmic therapy.
In PROGRESSIVE-AF, the same 303 patients were monitored continuously for an additional 24 months with an implanted cardiac monitor programmed with an AFib-detection algorithm. The data from the monitor were obtained daily. Over the final 2 years of the study, office visits were conducted every 6 months.
Tachyarrhythmias represent primary endpoint
In addition to persistent AFib, defined as lasting ≥ 7 days or lasting 48 hours to 7 days but requiring cardioversion for termination, patients in PROGRESSIVE-AF were also monitored for recurrent atrial tachyarrhythmias, AFib burden, quality of life (QOL), and health care utilization, and safety.
The average age was roughly 58 years. Although more than one-third had hypertension, most had no other comorbidities. The authors emphasized that the study population overall was relatively young and healthy.
Those randomized to antiarrhythmic therapy in EARLY-AF/PROGRESSIVE-AF received commonly prescribed therapies titrated to maximally tolerated doses using standardized protocols. At the start of EARLY-AF, flecainide, taken by 65% of patients, was the most commonly used agent, followed by sotalol, propafenone, dronedarone, and amiodarone.
At the end of PROGRESSIVE-AF, the order of the most common therapies did not change relative to EARLY-AF, but only 49% of patients were taking flecainide and 31% were no longer taking any antiarrhythmic therapy.
At the end of 3 years of follow-up in EARLY-AF/PROGRESSIVE-AF, the difference in persistent AFib represented a 75% reduction in favor of early ablation (HR, 0.25; 95% confidence interval, 0.09-0.70).
In those treated with ablation relative to those treated with antiarrhythmic therapy, the lower rate of atrial tachyarrhythmia lasting more than 7 days (1.9% vs. 6.0%) represented a 70% risk reduction (HR, 0.30; 95% CI 0.10-0.93). The protection from cardioversion for atrial tachyarrhythmia lasting between 2 and 7 days in duration (0.6% vs. 4.7%) translated into an 86% relative reduction (HR, 0.14; 95% CI, 0.02-0.85).
The impact on QOL for those randomized to ablation, which was measured with both AFib-specific and generic measures, was meaningful to patients, according to Dr. Andrade, director of the Cardiac Electrophysiology Laboratory, Vancouver General Hospital.
For example, the mean difference in the AF Quality of Life Survey (AFEQT), was 8.0 at 1 year and 7.4 at 3 years in favor of ablation. A change of 5 points in this score is considered to be a clinically meaningful difference, according to Dr. Andrade.
Numerically, the relative risk of emergency room visits and cardioversion were lower in the ablation group, but the differences did not reach statistical significance. However, the lower hazard ratio for hospitalization was significant (HR, 0.31; 95% CI, 0.15-0.66), supporting a reduction in consumption of health care resources.
Ablation found safer than drugs
The rate of adverse events of any kind (11.0% vs. 23.5%) and serious adverse events (4.5% vs. 10.1%) were lower in the ablation group.
There were no differences in major adverse cardiovascular events observed in this period of follow-up, but Dr. Andrade pointed out that follow-up was not long enough to expect differences in these events.
Impressed by the magnitude of the reduction in persistent AFib in a population of relatively young and healthy patients considered to be at a low risk of AFib progression, Dr. Blomström-Lindqvist, a professor of cardiology at the Institution of Medical Science, Uppsala, Sweden, indicated that the data support early ablation as a means to reduce risk of this outcome.
However, she did caution that progressive AFib was observed in a relatively small proportion of patients managed with antiarrhythmic therapy at 3 years, an outcome relevant when discussing treatment options with patients.
The results were published in New England Journal of Medicine simultaneously with Dr. Andrade’s presentation.
Dr. Andrade reports financial relationships with Bayer, Bayliss, Biosense, Bristol-Myers Squibb, Medtronic and Servier. The trial, funded largely by the Canadian government and Canadian professional societies, received additional funding from Bayliss and Medtronic. Dr. Blomström-Lundqvist reports financial relationships with Bayer, Boston Scientific, Cathprint, Medtronic, and Sanofi.
CHICAGO – Early ablation of atrial fibrillation (AFib) reduces the risk of progression, compared with antiarrhythmic therapies, according to results of a multicenter, randomized trial called PROGRESSIVE-AF.
Over 36 months of follow-up, the trial linked early ablation with a reduced risk of persistent AFib (1.9% vs. 7.4%), and in addition, those in the ablation group were less likely to have recurrent atrial tachyarrhythmias of any kind (56.5% vs. 77.2%), reported Jason G. Andrade, MD, at the American Heart Association scientific sessions.
Serving as a long-term extension of the EARLY-AF trial published almost 2 years ago, this trial expands evidence that progressive AFib can be attenuated, a concept that has been debated.
“Can early AFib ablation stop progression?” asked Carina Blomström-Lindqvist, MD, PhD. The invited discussant for the PROGRESSION-AF trial, Dr. Blomström-Lundqvist concluded, “here is another set of data that suggests it can.”
By another set of data, Dr. Blomström-Lindqvist was referring to a previously published multinational study called ATTEST In this study, which involved 29 sites worldwide and compared radiofrequency ablation to antiarrhythmic drug therapy, early ablation also produced a lower risk of persistent AFib at the end of 3 years (2.4% vs. 17.5%; P = .0009).
In the previously published open-label EARLY-AF trial, 303 patients with paroxysmal, untreated AFib were randomized to cryoballoon ablation or antiarrhythmic drugs. The primary endpoint was the first documented recurrence of an atrial tachyarrhythmia between 91 and 365 days. The lower rate following ablation (42.9% vs. 67.8%) represented a more than 50% reduction in risk (hazard ratio, 0.48; P < .001) relative to antiarrhythmic therapy.
In PROGRESSIVE-AF, the same 303 patients were monitored continuously for an additional 24 months with an implanted cardiac monitor programmed with an AFib-detection algorithm. The data from the monitor were obtained daily. Over the final 2 years of the study, office visits were conducted every 6 months.
Tachyarrhythmias represent primary endpoint
In addition to persistent AFib, defined as lasting ≥ 7 days or lasting 48 hours to 7 days but requiring cardioversion for termination, patients in PROGRESSIVE-AF were also monitored for recurrent atrial tachyarrhythmias, AFib burden, quality of life (QOL), and health care utilization, and safety.
The average age was roughly 58 years. Although more than one-third had hypertension, most had no other comorbidities. The authors emphasized that the study population overall was relatively young and healthy.
Those randomized to antiarrhythmic therapy in EARLY-AF/PROGRESSIVE-AF received commonly prescribed therapies titrated to maximally tolerated doses using standardized protocols. At the start of EARLY-AF, flecainide, taken by 65% of patients, was the most commonly used agent, followed by sotalol, propafenone, dronedarone, and amiodarone.
At the end of PROGRESSIVE-AF, the order of the most common therapies did not change relative to EARLY-AF, but only 49% of patients were taking flecainide and 31% were no longer taking any antiarrhythmic therapy.
At the end of 3 years of follow-up in EARLY-AF/PROGRESSIVE-AF, the difference in persistent AFib represented a 75% reduction in favor of early ablation (HR, 0.25; 95% confidence interval, 0.09-0.70).
In those treated with ablation relative to those treated with antiarrhythmic therapy, the lower rate of atrial tachyarrhythmia lasting more than 7 days (1.9% vs. 6.0%) represented a 70% risk reduction (HR, 0.30; 95% CI 0.10-0.93). The protection from cardioversion for atrial tachyarrhythmia lasting between 2 and 7 days in duration (0.6% vs. 4.7%) translated into an 86% relative reduction (HR, 0.14; 95% CI, 0.02-0.85).
The impact on QOL for those randomized to ablation, which was measured with both AFib-specific and generic measures, was meaningful to patients, according to Dr. Andrade, director of the Cardiac Electrophysiology Laboratory, Vancouver General Hospital.
For example, the mean difference in the AF Quality of Life Survey (AFEQT), was 8.0 at 1 year and 7.4 at 3 years in favor of ablation. A change of 5 points in this score is considered to be a clinically meaningful difference, according to Dr. Andrade.
Numerically, the relative risk of emergency room visits and cardioversion were lower in the ablation group, but the differences did not reach statistical significance. However, the lower hazard ratio for hospitalization was significant (HR, 0.31; 95% CI, 0.15-0.66), supporting a reduction in consumption of health care resources.
Ablation found safer than drugs
The rate of adverse events of any kind (11.0% vs. 23.5%) and serious adverse events (4.5% vs. 10.1%) were lower in the ablation group.
There were no differences in major adverse cardiovascular events observed in this period of follow-up, but Dr. Andrade pointed out that follow-up was not long enough to expect differences in these events.
Impressed by the magnitude of the reduction in persistent AFib in a population of relatively young and healthy patients considered to be at a low risk of AFib progression, Dr. Blomström-Lindqvist, a professor of cardiology at the Institution of Medical Science, Uppsala, Sweden, indicated that the data support early ablation as a means to reduce risk of this outcome.
However, she did caution that progressive AFib was observed in a relatively small proportion of patients managed with antiarrhythmic therapy at 3 years, an outcome relevant when discussing treatment options with patients.
The results were published in New England Journal of Medicine simultaneously with Dr. Andrade’s presentation.
Dr. Andrade reports financial relationships with Bayer, Bayliss, Biosense, Bristol-Myers Squibb, Medtronic and Servier. The trial, funded largely by the Canadian government and Canadian professional societies, received additional funding from Bayliss and Medtronic. Dr. Blomström-Lundqvist reports financial relationships with Bayer, Boston Scientific, Cathprint, Medtronic, and Sanofi.
AT AHA 2022
Liver disease-related deaths rise during pandemic
according to new findings presented at the annual meeting of the American Association for the Study of Liver Diseases.
Between 2019 and 2021, ALD-related deaths increased by 17.6% and NAFLD-related deaths increased by 14.5%, Yee Hui Yeo, MD, a resident physician and hepatology-focused investigator at Cedars-Sinai Medical Center in Los Angeles, said at a preconference press briefing.
“Even before the pandemic, the mortality rates for these two diseases have been increasing, with NAFLD having an even steeper increasing trend,” he said. “During the pandemic, these two diseases had a significant surge.”
Recent U.S. liver disease death rates
Dr. Yeo and colleagues analyzed data from the Center for Disease Control and Prevention’s National Vital Statistic System to estimate the age-standardized mortality rates (ASMR) of liver disease between 2010 and 2021, including ALD, NAFLD, hepatitis B, and hepatitis C. Using prediction modeling analyses based on trends from 2010 to 2019, they predicted mortality rates for 2020-2021 and compared them with the observed rates to quantify the differences related to the pandemic.
Between 2010 and 2021, there were about 626,000 chronic liver disease–related deaths, including about 343,000 ALD-related deaths, 204,000 hepatitis C–related deaths, 58,000 NAFLD-related deaths, and 21,000 hepatitis B–related deaths.
For ALD-related deaths, the annual percentage change was 3.5% for 2010-2019 and 17.6% for 2019-2021. The observed ASMR in 2020 was significantly higher than predicted, at 15.7 deaths per 100,000 people versus 13.0 predicted from the 2010-2019 rate. The trend continued in 2021, with 17.4 deaths per 100,000 people versus 13.4 in the previous decade.
The highest numbers of ALD-related deaths during the COVID-19 pandemic occurred in Alaska, Montana, Wyoming, Colorado, New Mexico, and South Dakota.
For NAFLD-related deaths, the annual percentage change was 7.6% for 2010-2014, 11.8% for 2014-2019, and 14.5% for 2019-2021. The observed ASMR was also higher than predicted, at 3.1 deaths per 100,000 people versus 2.6 in 2020, as well as 3.4 versus 2.8 in 2021.
The highest numbers of NAFLD-related deaths during the COVID-19 pandemic occurred in Oklahoma, Indiana, Kentucky, Tennessee, and West Virginia.
Hepatitis B and C gains lost in pandemic
In contrast, the annual percentage change in was –1.9% for hepatitis B and –2.8% for hepatitis C. After new treatment for hepatitis C emerged in 2013-2014, mortality rates were –7.8% for 2014-2019, Dr. Yeo noted.
“However, during the pandemic, we saw that this decrease has become a nonsignificant change,” he said. “That means our progress of the past 5 or 6 years has already stopped during the pandemic.”
By race and ethnicity, the increase in ALD-related mortality was most pronounced in non-Hispanic White, non-Hispanic Black, and Alaska Native/American Indian populations, Dr. Yeo said. Alaska Natives and American Indians had the highest annual percentage change, at 18%, followed by non-Hispanic Whites at 11.7% and non-Hispanic Blacks at 10.8%. There were no significant differences in race and ethnicity for NAFLD-related deaths, although all groups had major increases in recent years.
Biggest rise in young adults
By age, the increase in ALD-related mortality was particularly severe for ages 25-44, with an annual percentage change of 34.6% in 2019-2021, as compared with 13.7% for ages 45-64 and 12.6% for ages 65 and older.
For NAFLD-related deaths, another major increase was observed among ages 25-44, with an annual percentage change of 28.1% for 2019-2021, as compared with 12% for ages 65 and older and 7.4% for ages 45-64.
By sex, the ASMR increase in NAFLD-related mortality was steady throughout 2010-2021 for both men and women. In contrast, ALD-related death increased sharply between 2019 and 2021, with an annual percentage change of 19.1% for women and 16.7% for men.
“The increasing trend in mortality rates for ALD and NAFLD has been quite alarming, with disparities in age, race, and ethnicity,” Dr. Yeo said.
The study received no funding support. Some authors disclosed research funding, advisory board roles, and consulting fees with various pharmaceutical companies.
according to new findings presented at the annual meeting of the American Association for the Study of Liver Diseases.
Between 2019 and 2021, ALD-related deaths increased by 17.6% and NAFLD-related deaths increased by 14.5%, Yee Hui Yeo, MD, a resident physician and hepatology-focused investigator at Cedars-Sinai Medical Center in Los Angeles, said at a preconference press briefing.
“Even before the pandemic, the mortality rates for these two diseases have been increasing, with NAFLD having an even steeper increasing trend,” he said. “During the pandemic, these two diseases had a significant surge.”
Recent U.S. liver disease death rates
Dr. Yeo and colleagues analyzed data from the Center for Disease Control and Prevention’s National Vital Statistic System to estimate the age-standardized mortality rates (ASMR) of liver disease between 2010 and 2021, including ALD, NAFLD, hepatitis B, and hepatitis C. Using prediction modeling analyses based on trends from 2010 to 2019, they predicted mortality rates for 2020-2021 and compared them with the observed rates to quantify the differences related to the pandemic.
Between 2010 and 2021, there were about 626,000 chronic liver disease–related deaths, including about 343,000 ALD-related deaths, 204,000 hepatitis C–related deaths, 58,000 NAFLD-related deaths, and 21,000 hepatitis B–related deaths.
For ALD-related deaths, the annual percentage change was 3.5% for 2010-2019 and 17.6% for 2019-2021. The observed ASMR in 2020 was significantly higher than predicted, at 15.7 deaths per 100,000 people versus 13.0 predicted from the 2010-2019 rate. The trend continued in 2021, with 17.4 deaths per 100,000 people versus 13.4 in the previous decade.
The highest numbers of ALD-related deaths during the COVID-19 pandemic occurred in Alaska, Montana, Wyoming, Colorado, New Mexico, and South Dakota.
For NAFLD-related deaths, the annual percentage change was 7.6% for 2010-2014, 11.8% for 2014-2019, and 14.5% for 2019-2021. The observed ASMR was also higher than predicted, at 3.1 deaths per 100,000 people versus 2.6 in 2020, as well as 3.4 versus 2.8 in 2021.
The highest numbers of NAFLD-related deaths during the COVID-19 pandemic occurred in Oklahoma, Indiana, Kentucky, Tennessee, and West Virginia.
Hepatitis B and C gains lost in pandemic
In contrast, the annual percentage change in was –1.9% for hepatitis B and –2.8% for hepatitis C. After new treatment for hepatitis C emerged in 2013-2014, mortality rates were –7.8% for 2014-2019, Dr. Yeo noted.
“However, during the pandemic, we saw that this decrease has become a nonsignificant change,” he said. “That means our progress of the past 5 or 6 years has already stopped during the pandemic.”
By race and ethnicity, the increase in ALD-related mortality was most pronounced in non-Hispanic White, non-Hispanic Black, and Alaska Native/American Indian populations, Dr. Yeo said. Alaska Natives and American Indians had the highest annual percentage change, at 18%, followed by non-Hispanic Whites at 11.7% and non-Hispanic Blacks at 10.8%. There were no significant differences in race and ethnicity for NAFLD-related deaths, although all groups had major increases in recent years.
Biggest rise in young adults
By age, the increase in ALD-related mortality was particularly severe for ages 25-44, with an annual percentage change of 34.6% in 2019-2021, as compared with 13.7% for ages 45-64 and 12.6% for ages 65 and older.
For NAFLD-related deaths, another major increase was observed among ages 25-44, with an annual percentage change of 28.1% for 2019-2021, as compared with 12% for ages 65 and older and 7.4% for ages 45-64.
By sex, the ASMR increase in NAFLD-related mortality was steady throughout 2010-2021 for both men and women. In contrast, ALD-related death increased sharply between 2019 and 2021, with an annual percentage change of 19.1% for women and 16.7% for men.
“The increasing trend in mortality rates for ALD and NAFLD has been quite alarming, with disparities in age, race, and ethnicity,” Dr. Yeo said.
The study received no funding support. Some authors disclosed research funding, advisory board roles, and consulting fees with various pharmaceutical companies.
according to new findings presented at the annual meeting of the American Association for the Study of Liver Diseases.
Between 2019 and 2021, ALD-related deaths increased by 17.6% and NAFLD-related deaths increased by 14.5%, Yee Hui Yeo, MD, a resident physician and hepatology-focused investigator at Cedars-Sinai Medical Center in Los Angeles, said at a preconference press briefing.
“Even before the pandemic, the mortality rates for these two diseases have been increasing, with NAFLD having an even steeper increasing trend,” he said. “During the pandemic, these two diseases had a significant surge.”
Recent U.S. liver disease death rates
Dr. Yeo and colleagues analyzed data from the Center for Disease Control and Prevention’s National Vital Statistic System to estimate the age-standardized mortality rates (ASMR) of liver disease between 2010 and 2021, including ALD, NAFLD, hepatitis B, and hepatitis C. Using prediction modeling analyses based on trends from 2010 to 2019, they predicted mortality rates for 2020-2021 and compared them with the observed rates to quantify the differences related to the pandemic.
Between 2010 and 2021, there were about 626,000 chronic liver disease–related deaths, including about 343,000 ALD-related deaths, 204,000 hepatitis C–related deaths, 58,000 NAFLD-related deaths, and 21,000 hepatitis B–related deaths.
For ALD-related deaths, the annual percentage change was 3.5% for 2010-2019 and 17.6% for 2019-2021. The observed ASMR in 2020 was significantly higher than predicted, at 15.7 deaths per 100,000 people versus 13.0 predicted from the 2010-2019 rate. The trend continued in 2021, with 17.4 deaths per 100,000 people versus 13.4 in the previous decade.
The highest numbers of ALD-related deaths during the COVID-19 pandemic occurred in Alaska, Montana, Wyoming, Colorado, New Mexico, and South Dakota.
For NAFLD-related deaths, the annual percentage change was 7.6% for 2010-2014, 11.8% for 2014-2019, and 14.5% for 2019-2021. The observed ASMR was also higher than predicted, at 3.1 deaths per 100,000 people versus 2.6 in 2020, as well as 3.4 versus 2.8 in 2021.
The highest numbers of NAFLD-related deaths during the COVID-19 pandemic occurred in Oklahoma, Indiana, Kentucky, Tennessee, and West Virginia.
Hepatitis B and C gains lost in pandemic
In contrast, the annual percentage change in was –1.9% for hepatitis B and –2.8% for hepatitis C. After new treatment for hepatitis C emerged in 2013-2014, mortality rates were –7.8% for 2014-2019, Dr. Yeo noted.
“However, during the pandemic, we saw that this decrease has become a nonsignificant change,” he said. “That means our progress of the past 5 or 6 years has already stopped during the pandemic.”
By race and ethnicity, the increase in ALD-related mortality was most pronounced in non-Hispanic White, non-Hispanic Black, and Alaska Native/American Indian populations, Dr. Yeo said. Alaska Natives and American Indians had the highest annual percentage change, at 18%, followed by non-Hispanic Whites at 11.7% and non-Hispanic Blacks at 10.8%. There were no significant differences in race and ethnicity for NAFLD-related deaths, although all groups had major increases in recent years.
Biggest rise in young adults
By age, the increase in ALD-related mortality was particularly severe for ages 25-44, with an annual percentage change of 34.6% in 2019-2021, as compared with 13.7% for ages 45-64 and 12.6% for ages 65 and older.
For NAFLD-related deaths, another major increase was observed among ages 25-44, with an annual percentage change of 28.1% for 2019-2021, as compared with 12% for ages 65 and older and 7.4% for ages 45-64.
By sex, the ASMR increase in NAFLD-related mortality was steady throughout 2010-2021 for both men and women. In contrast, ALD-related death increased sharply between 2019 and 2021, with an annual percentage change of 19.1% for women and 16.7% for men.
“The increasing trend in mortality rates for ALD and NAFLD has been quite alarming, with disparities in age, race, and ethnicity,” Dr. Yeo said.
The study received no funding support. Some authors disclosed research funding, advisory board roles, and consulting fees with various pharmaceutical companies.
FROM THE LIVER MEETING
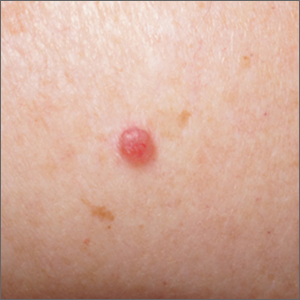
ICD-10 code can identify patients with melasma for future study
To better understand melasma, it is important for researchers to find groups of patients with confirmed disease for future clinical study. A recent for researchers interested in conducting retrospective studies of this patient population.
“Overall, our results support the validity of using the ICD-10 code for melasma to identify patients with a diagnosis of melasma for future studies,” Nicholas Theodosakis, MD, PhD, of the department of dermatology at Massachusetts General Hospital, Boston, and colleagues wrote in their research letter. “Despite some variability in diagnostic confidence, most patients were ultimately classified as moderately or highly likely to have a true diagnosis of melasma.”
Dr. Theodosakis and colleagues evaluated data from 5,322 adult patients in the Mass General Brigham Research Patient Data Registry between October 2015 and January 2021 who had an encounter that used the ICD-10 code for melasma (L81.1). The researchers then validated the ICD-10 code by examining the medical records of 300 patients (5.6%), confirming that melasma was the clinician’s favored diagnosis and that the patient met secondary diagnostic criteria. Confidence was rated in categories of “low confidence,” “moderate confidence,” “high confidence,” and “maximum confidence” based on secondary criteria such as hyperpigmentation of the face and upper body, hormone-related therapy exposure before diagnosis, pregnancy history, and dermatologist-confirmed diagnosis.
The patients who had their medical records examined for confirmed melasma were primarily women (285 patients; 95.0%) and were a mean 48.4 years old at diagnosis.
Of those in the validation cohort, melasma was the preferred diagnosis for clinicians of 291 patients (97.0%), while 274 patients (91.3%) had secondary diagnostic criteria of hyperpigmentation of the face and upper body and 252 patients (84.0%) had received a diagnosis from a dermatologist. Other less common secondary diagnostic criteria of the patient group were a history of having received hormone-related therapy before a melasma diagnosis (148 patients; 49.3%) and a history of pregnancy (168 patients; 56.0%). Based on identification of secondary diagnostic criteria, confidence in melasma diagnosis was high for 208 patients (69.3%), moderate for 61 patients (20.3%), and low for 31 patients (10.3%).
Dr. Theodosakis and colleagues noted their study was limited by its retrospective nature and the presence of a small validation cohort. “Despite these limitations, our findings provide a framework for identifying cohorts to evaluate the clinical course and treatment of melasma,” the authors concluded.
One of the authors reported relationships with companies including AbbVie, Acom, Boehringer Ingelheim, Concert, Digital Diagnostics, and Eli Lilly in the form of personal fees, equity, royalties and/or licensing, or medical advisory board positions outside the submitted work; another author reported being an advisory board member and consultant for and receiving honoraria from Incyte, Castle Biosciences, Galderma, and Sanofi outside the submitted work. The other authors reported no relevant conflicts of interest.
To better understand melasma, it is important for researchers to find groups of patients with confirmed disease for future clinical study. A recent for researchers interested in conducting retrospective studies of this patient population.
“Overall, our results support the validity of using the ICD-10 code for melasma to identify patients with a diagnosis of melasma for future studies,” Nicholas Theodosakis, MD, PhD, of the department of dermatology at Massachusetts General Hospital, Boston, and colleagues wrote in their research letter. “Despite some variability in diagnostic confidence, most patients were ultimately classified as moderately or highly likely to have a true diagnosis of melasma.”
Dr. Theodosakis and colleagues evaluated data from 5,322 adult patients in the Mass General Brigham Research Patient Data Registry between October 2015 and January 2021 who had an encounter that used the ICD-10 code for melasma (L81.1). The researchers then validated the ICD-10 code by examining the medical records of 300 patients (5.6%), confirming that melasma was the clinician’s favored diagnosis and that the patient met secondary diagnostic criteria. Confidence was rated in categories of “low confidence,” “moderate confidence,” “high confidence,” and “maximum confidence” based on secondary criteria such as hyperpigmentation of the face and upper body, hormone-related therapy exposure before diagnosis, pregnancy history, and dermatologist-confirmed diagnosis.
The patients who had their medical records examined for confirmed melasma were primarily women (285 patients; 95.0%) and were a mean 48.4 years old at diagnosis.
Of those in the validation cohort, melasma was the preferred diagnosis for clinicians of 291 patients (97.0%), while 274 patients (91.3%) had secondary diagnostic criteria of hyperpigmentation of the face and upper body and 252 patients (84.0%) had received a diagnosis from a dermatologist. Other less common secondary diagnostic criteria of the patient group were a history of having received hormone-related therapy before a melasma diagnosis (148 patients; 49.3%) and a history of pregnancy (168 patients; 56.0%). Based on identification of secondary diagnostic criteria, confidence in melasma diagnosis was high for 208 patients (69.3%), moderate for 61 patients (20.3%), and low for 31 patients (10.3%).
Dr. Theodosakis and colleagues noted their study was limited by its retrospective nature and the presence of a small validation cohort. “Despite these limitations, our findings provide a framework for identifying cohorts to evaluate the clinical course and treatment of melasma,” the authors concluded.
One of the authors reported relationships with companies including AbbVie, Acom, Boehringer Ingelheim, Concert, Digital Diagnostics, and Eli Lilly in the form of personal fees, equity, royalties and/or licensing, or medical advisory board positions outside the submitted work; another author reported being an advisory board member and consultant for and receiving honoraria from Incyte, Castle Biosciences, Galderma, and Sanofi outside the submitted work. The other authors reported no relevant conflicts of interest.
To better understand melasma, it is important for researchers to find groups of patients with confirmed disease for future clinical study. A recent for researchers interested in conducting retrospective studies of this patient population.
“Overall, our results support the validity of using the ICD-10 code for melasma to identify patients with a diagnosis of melasma for future studies,” Nicholas Theodosakis, MD, PhD, of the department of dermatology at Massachusetts General Hospital, Boston, and colleagues wrote in their research letter. “Despite some variability in diagnostic confidence, most patients were ultimately classified as moderately or highly likely to have a true diagnosis of melasma.”
Dr. Theodosakis and colleagues evaluated data from 5,322 adult patients in the Mass General Brigham Research Patient Data Registry between October 2015 and January 2021 who had an encounter that used the ICD-10 code for melasma (L81.1). The researchers then validated the ICD-10 code by examining the medical records of 300 patients (5.6%), confirming that melasma was the clinician’s favored diagnosis and that the patient met secondary diagnostic criteria. Confidence was rated in categories of “low confidence,” “moderate confidence,” “high confidence,” and “maximum confidence” based on secondary criteria such as hyperpigmentation of the face and upper body, hormone-related therapy exposure before diagnosis, pregnancy history, and dermatologist-confirmed diagnosis.
The patients who had their medical records examined for confirmed melasma were primarily women (285 patients; 95.0%) and were a mean 48.4 years old at diagnosis.
Of those in the validation cohort, melasma was the preferred diagnosis for clinicians of 291 patients (97.0%), while 274 patients (91.3%) had secondary diagnostic criteria of hyperpigmentation of the face and upper body and 252 patients (84.0%) had received a diagnosis from a dermatologist. Other less common secondary diagnostic criteria of the patient group were a history of having received hormone-related therapy before a melasma diagnosis (148 patients; 49.3%) and a history of pregnancy (168 patients; 56.0%). Based on identification of secondary diagnostic criteria, confidence in melasma diagnosis was high for 208 patients (69.3%), moderate for 61 patients (20.3%), and low for 31 patients (10.3%).
Dr. Theodosakis and colleagues noted their study was limited by its retrospective nature and the presence of a small validation cohort. “Despite these limitations, our findings provide a framework for identifying cohorts to evaluate the clinical course and treatment of melasma,” the authors concluded.
One of the authors reported relationships with companies including AbbVie, Acom, Boehringer Ingelheim, Concert, Digital Diagnostics, and Eli Lilly in the form of personal fees, equity, royalties and/or licensing, or medical advisory board positions outside the submitted work; another author reported being an advisory board member and consultant for and receiving honoraria from Incyte, Castle Biosciences, Galderma, and Sanofi outside the submitted work. The other authors reported no relevant conflicts of interest.
FROM JAMA DERMATOLOGY
More children should be getting flu vaccines
Cold and flu season came early in 2022.
On Nov. 4, 2022, the Centers for Disease Control and Prevention issued a Health Alert Network Health Advisory about early, elevated respiratory disease incidence caused by multiple viruses other than SARS-CoV-2.
Interseasonal spread of respiratory syncytial virus has continued in 2022, with RSV-associated hospitalizations increasing in the late spring and continuing throughout the summer and into the fall. In October, some regions of the country were seeing RSV activity near the peak seasonal levels typically observed in December and January.
Cases of severe respiratory infection in children who tested positive for rhinovirus or enterovirus spiked in August; further testing confirmed the presence of EV-D68 in some children. Rhinovirus and enterovirus continue to circulate and are isolated in hospitalized children with respiratory illness.
In some parts of the country, influenza cases have rapidly increased ahead of what we normally anticipate. According to preliminary estimates from the CDC, between Oct. 1 and Oct. 22, 880,000 people were sickened with flu, 420,000 people visited a health care provider for flu illness, and 6,900 people were hospitalized for flu. The cumulative hospitalization rate is higher than observed at this time of year in every previous flu season since 2010-2011. Hospitalization rates are highest in children aged 0-4 years and adults 65 years and older.
Of course, this report came as no surprise to pediatric health care providers. Many children’s hospitals had been operating at or over capacity for weeks. While a systematic assessment of the surge on children’s hospitals has not been published, anecdotally, hospitals from around the country have described record emergency department visits and inpatient census numbers. Some have set up tents or other temporary facilities to see ambulatory patients and have canceled elective surgeries because of a lack of beds.
There is no quick or easy solution to stem the tide of RSV-related or enterovirus/rhinovirus admissions, but many flu-related hospitalizations are vaccine preventable. Unfortunately, too few children are receiving influenza vaccine. As of the week ending Oct. 15, only about 22.1% of eligible children had been immunized. The American Academy of Pediatrics and the CDC recommend that all children are vaccinated, preferably by the end of October so they have time to develop immunity before influenza starts circulating. As it stands now, the majority of the nation’s children are facing a flu season without the benefits of vaccine.
There is still time to take steps to prevent this flu season from becoming one of the worst in recent memory. A strong provider recommendation for influenza vaccine is consistently associated with higher rates of vaccine acceptance. We need to recommend influenza vaccine to all eligible patients at every visit and in every setting. It will help if we can say it like we mean it. Some of us are tired of debating the merits of COVID-19 vaccine with families and may be leery of additional debates about flu. Some of us may just be tired, as many practices have already expanded office hours to care for the influx of kids with respiratory illness. On the heels of two atypical flu seasons, a few of us may be quietly complacent about the importance of flu vaccines for children.
Anyone in need of a little motivation should check out a paper recently published in Clinical Infectious Diseases that reinforces the value of flu vaccine, even in a year when there is a poor match between the vaccine and circulating viruses.
The 2019-2020 flu season was a bad flu season for children. Two antigenically drifted influenza viruses predominated and cases of influenza soared, resulting in the largest influenza epidemic in children in the United States since 1992. Pediatric Intensive Care Influenza Study investigators used a test-negative design to estimate the effectiveness of influenza vaccine in preventing critical and life-threatening influenza in children during that season. The good news: vaccination reduced the risk of critical influenza by 78% against H1N1pdm09 viruses that were well-matched to vaccine and by 47% against mismatched viruses. Vaccination was estimated to be 75% protective against antigenically drifted B-Victoria viruses. Overall vaccine effectiveness against critical illness from any influenza virus was 63% (95% confidence interval, 38%-78%).
While it might be tempting to attribute suboptimal immunization rates to vaccine hesitancy, ready availability remains an issue for some families. We need to eliminate barriers to access. While the AAP continues to emphasize immunization in the medical home, especially for the youngest infants, the 2022 policy statement suggests that vaccinating children in schools, pharmacies, and other nontraditional settings could improve immunization rates. To the extent feasible, we need to work with partners to support community-based initiatives and promote these to families who struggle to make it into the office.
Improving access is just one potential way to reduce health disparities related to influenza and influenza vaccination. Over 10 influenza seasons, higher rates of influenza-associated hospitalizations and intensive care unit admissions were observed in Black, Hispanic, and American Indian/Alaska Native people. These disparities were highest in children aged younger than 4 years and influenza-associated in-hospital deaths were three- to fourfold higher in Black, Hispanic, and Asian/Pacific Islander children, compared with White children. The reason for the disparities isn’t completely clear but increasing immunization rates may be part of the solution. During the 2020-2021 influenza season, flu immunization rates in Black children (51.6%) were lower than those seen in White (57.4%) and Hispanic children (58.9%).
The AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023, highlight a variety of evidence-based strategies to increase influenza immunization rates. These may provide a little inspiration for clinicians looking to try a new approach. If you wish to share your experience with increasing influenza immunization rates in your practice setting, please email me at [email protected].
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead.
Cold and flu season came early in 2022.
On Nov. 4, 2022, the Centers for Disease Control and Prevention issued a Health Alert Network Health Advisory about early, elevated respiratory disease incidence caused by multiple viruses other than SARS-CoV-2.
Interseasonal spread of respiratory syncytial virus has continued in 2022, with RSV-associated hospitalizations increasing in the late spring and continuing throughout the summer and into the fall. In October, some regions of the country were seeing RSV activity near the peak seasonal levels typically observed in December and January.
Cases of severe respiratory infection in children who tested positive for rhinovirus or enterovirus spiked in August; further testing confirmed the presence of EV-D68 in some children. Rhinovirus and enterovirus continue to circulate and are isolated in hospitalized children with respiratory illness.
In some parts of the country, influenza cases have rapidly increased ahead of what we normally anticipate. According to preliminary estimates from the CDC, between Oct. 1 and Oct. 22, 880,000 people were sickened with flu, 420,000 people visited a health care provider for flu illness, and 6,900 people were hospitalized for flu. The cumulative hospitalization rate is higher than observed at this time of year in every previous flu season since 2010-2011. Hospitalization rates are highest in children aged 0-4 years and adults 65 years and older.
Of course, this report came as no surprise to pediatric health care providers. Many children’s hospitals had been operating at or over capacity for weeks. While a systematic assessment of the surge on children’s hospitals has not been published, anecdotally, hospitals from around the country have described record emergency department visits and inpatient census numbers. Some have set up tents or other temporary facilities to see ambulatory patients and have canceled elective surgeries because of a lack of beds.
There is no quick or easy solution to stem the tide of RSV-related or enterovirus/rhinovirus admissions, but many flu-related hospitalizations are vaccine preventable. Unfortunately, too few children are receiving influenza vaccine. As of the week ending Oct. 15, only about 22.1% of eligible children had been immunized. The American Academy of Pediatrics and the CDC recommend that all children are vaccinated, preferably by the end of October so they have time to develop immunity before influenza starts circulating. As it stands now, the majority of the nation’s children are facing a flu season without the benefits of vaccine.
There is still time to take steps to prevent this flu season from becoming one of the worst in recent memory. A strong provider recommendation for influenza vaccine is consistently associated with higher rates of vaccine acceptance. We need to recommend influenza vaccine to all eligible patients at every visit and in every setting. It will help if we can say it like we mean it. Some of us are tired of debating the merits of COVID-19 vaccine with families and may be leery of additional debates about flu. Some of us may just be tired, as many practices have already expanded office hours to care for the influx of kids with respiratory illness. On the heels of two atypical flu seasons, a few of us may be quietly complacent about the importance of flu vaccines for children.
Anyone in need of a little motivation should check out a paper recently published in Clinical Infectious Diseases that reinforces the value of flu vaccine, even in a year when there is a poor match between the vaccine and circulating viruses.
The 2019-2020 flu season was a bad flu season for children. Two antigenically drifted influenza viruses predominated and cases of influenza soared, resulting in the largest influenza epidemic in children in the United States since 1992. Pediatric Intensive Care Influenza Study investigators used a test-negative design to estimate the effectiveness of influenza vaccine in preventing critical and life-threatening influenza in children during that season. The good news: vaccination reduced the risk of critical influenza by 78% against H1N1pdm09 viruses that were well-matched to vaccine and by 47% against mismatched viruses. Vaccination was estimated to be 75% protective against antigenically drifted B-Victoria viruses. Overall vaccine effectiveness against critical illness from any influenza virus was 63% (95% confidence interval, 38%-78%).
While it might be tempting to attribute suboptimal immunization rates to vaccine hesitancy, ready availability remains an issue for some families. We need to eliminate barriers to access. While the AAP continues to emphasize immunization in the medical home, especially for the youngest infants, the 2022 policy statement suggests that vaccinating children in schools, pharmacies, and other nontraditional settings could improve immunization rates. To the extent feasible, we need to work with partners to support community-based initiatives and promote these to families who struggle to make it into the office.
Improving access is just one potential way to reduce health disparities related to influenza and influenza vaccination. Over 10 influenza seasons, higher rates of influenza-associated hospitalizations and intensive care unit admissions were observed in Black, Hispanic, and American Indian/Alaska Native people. These disparities were highest in children aged younger than 4 years and influenza-associated in-hospital deaths were three- to fourfold higher in Black, Hispanic, and Asian/Pacific Islander children, compared with White children. The reason for the disparities isn’t completely clear but increasing immunization rates may be part of the solution. During the 2020-2021 influenza season, flu immunization rates in Black children (51.6%) were lower than those seen in White (57.4%) and Hispanic children (58.9%).
The AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023, highlight a variety of evidence-based strategies to increase influenza immunization rates. These may provide a little inspiration for clinicians looking to try a new approach. If you wish to share your experience with increasing influenza immunization rates in your practice setting, please email me at [email protected].
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead.
Cold and flu season came early in 2022.
On Nov. 4, 2022, the Centers for Disease Control and Prevention issued a Health Alert Network Health Advisory about early, elevated respiratory disease incidence caused by multiple viruses other than SARS-CoV-2.
Interseasonal spread of respiratory syncytial virus has continued in 2022, with RSV-associated hospitalizations increasing in the late spring and continuing throughout the summer and into the fall. In October, some regions of the country were seeing RSV activity near the peak seasonal levels typically observed in December and January.
Cases of severe respiratory infection in children who tested positive for rhinovirus or enterovirus spiked in August; further testing confirmed the presence of EV-D68 in some children. Rhinovirus and enterovirus continue to circulate and are isolated in hospitalized children with respiratory illness.
In some parts of the country, influenza cases have rapidly increased ahead of what we normally anticipate. According to preliminary estimates from the CDC, between Oct. 1 and Oct. 22, 880,000 people were sickened with flu, 420,000 people visited a health care provider for flu illness, and 6,900 people were hospitalized for flu. The cumulative hospitalization rate is higher than observed at this time of year in every previous flu season since 2010-2011. Hospitalization rates are highest in children aged 0-4 years and adults 65 years and older.
Of course, this report came as no surprise to pediatric health care providers. Many children’s hospitals had been operating at or over capacity for weeks. While a systematic assessment of the surge on children’s hospitals has not been published, anecdotally, hospitals from around the country have described record emergency department visits and inpatient census numbers. Some have set up tents or other temporary facilities to see ambulatory patients and have canceled elective surgeries because of a lack of beds.
There is no quick or easy solution to stem the tide of RSV-related or enterovirus/rhinovirus admissions, but many flu-related hospitalizations are vaccine preventable. Unfortunately, too few children are receiving influenza vaccine. As of the week ending Oct. 15, only about 22.1% of eligible children had been immunized. The American Academy of Pediatrics and the CDC recommend that all children are vaccinated, preferably by the end of October so they have time to develop immunity before influenza starts circulating. As it stands now, the majority of the nation’s children are facing a flu season without the benefits of vaccine.
There is still time to take steps to prevent this flu season from becoming one of the worst in recent memory. A strong provider recommendation for influenza vaccine is consistently associated with higher rates of vaccine acceptance. We need to recommend influenza vaccine to all eligible patients at every visit and in every setting. It will help if we can say it like we mean it. Some of us are tired of debating the merits of COVID-19 vaccine with families and may be leery of additional debates about flu. Some of us may just be tired, as many practices have already expanded office hours to care for the influx of kids with respiratory illness. On the heels of two atypical flu seasons, a few of us may be quietly complacent about the importance of flu vaccines for children.
Anyone in need of a little motivation should check out a paper recently published in Clinical Infectious Diseases that reinforces the value of flu vaccine, even in a year when there is a poor match between the vaccine and circulating viruses.
The 2019-2020 flu season was a bad flu season for children. Two antigenically drifted influenza viruses predominated and cases of influenza soared, resulting in the largest influenza epidemic in children in the United States since 1992. Pediatric Intensive Care Influenza Study investigators used a test-negative design to estimate the effectiveness of influenza vaccine in preventing critical and life-threatening influenza in children during that season. The good news: vaccination reduced the risk of critical influenza by 78% against H1N1pdm09 viruses that were well-matched to vaccine and by 47% against mismatched viruses. Vaccination was estimated to be 75% protective against antigenically drifted B-Victoria viruses. Overall vaccine effectiveness against critical illness from any influenza virus was 63% (95% confidence interval, 38%-78%).
While it might be tempting to attribute suboptimal immunization rates to vaccine hesitancy, ready availability remains an issue for some families. We need to eliminate barriers to access. While the AAP continues to emphasize immunization in the medical home, especially for the youngest infants, the 2022 policy statement suggests that vaccinating children in schools, pharmacies, and other nontraditional settings could improve immunization rates. To the extent feasible, we need to work with partners to support community-based initiatives and promote these to families who struggle to make it into the office.
Improving access is just one potential way to reduce health disparities related to influenza and influenza vaccination. Over 10 influenza seasons, higher rates of influenza-associated hospitalizations and intensive care unit admissions were observed in Black, Hispanic, and American Indian/Alaska Native people. These disparities were highest in children aged younger than 4 years and influenza-associated in-hospital deaths were three- to fourfold higher in Black, Hispanic, and Asian/Pacific Islander children, compared with White children. The reason for the disparities isn’t completely clear but increasing immunization rates may be part of the solution. During the 2020-2021 influenza season, flu immunization rates in Black children (51.6%) were lower than those seen in White (57.4%) and Hispanic children (58.9%).
The AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023, highlight a variety of evidence-based strategies to increase influenza immunization rates. These may provide a little inspiration for clinicians looking to try a new approach. If you wish to share your experience with increasing influenza immunization rates in your practice setting, please email me at [email protected].
Dr. Bryant is a pediatrician specializing in infectious diseases at the University of Louisville (Ky.) and Norton Children’s Hospital, also in Louisville. She is a member of the AAP’s Committee on Infectious Diseases and one of the lead authors of the AAP’s Recommendations for Prevention and Control of Influenza in Children, 2022–2023. The opinions expressed in this article are her own. Dr. Bryant discloses that she has served as an investigator on clinical trials funded by Pfizer, Enanta, and Gilead.
In patients with untreated AIDS, monkeypox can be life-threatening
Monkeypox, though often mild, may be severe and even fatal in immunocompromised individuals, particularly those with untreated AIDS, according to a Centers for Disease Control and Prevention study in Morbidity and Mortality Weekly Report.
The study described a group of patients recently treated for severe monkeypox. The majority were Black, HIV positive, and not receiving treatment. Many were also facing homelessness.
The authors urged HIV testing for all sexually active individuals with suspected monkeypox. Early or prolonged monkeypox treatment may be necessary, they concluded.
Coauthor John T. Brooks, MD, called the study “a real call to action.”
“If we want to reduce cases of severe monkeypox, we need to reduce the number of persons with HIV who are undiagnosed and not treated,” said Dr. Brooks, a medical epidemiologist who is chief medical officer of CDC›s multinational monkeypox response. Dr. Brooks also leads the epidemiology research team in CDC’s division of HIV/AIDS prevention.
noted Richard Silvera, MD, MPH, CPH, who is associate program director of the infectious diseases fellowship and assistant professor of medicine (infectious diseases) at the Icahn School of Medicine at Mount Sinai, New York. He was not involved with the study.
“These patients really have not been served by the health care system,” Dr. Silvera said. “Monkeypox is just really taking advantage of that.”
How severe monkeypox can manifest
The authors reported on 57 adults hospitalized with severe monkeypox between Aug. 10 and Sept. 10, 2022, for whose care the providers sought CDC consultation.
The vast majority (95%) were men, their median age was 34 years, and 68% were Black. Nearly one in four were homeless (23%).
Overall, 47 (82%) were HIV positive, of whom just 4 had been receiving antiretroviral therapy (ART). Of 43 for whom CD4 counts were known, 71% had fewer than 50 CD4 cells/mm3.
Clinical signs included severe skin lesions in all patients and severe mucosal lesions in 68%. Other affected organ systems included lungs (21%), eyes (21%), and central nervous system (7%).
Treatments included oral or intravenous tecovirimat (93% and 65%, respectively), vaccinia immune globulin intravenous (VIGIV, 51%), and cidofovir (23%).
Nearly 1 in 3 patients (30%) received care in an ICU; 12 died (21%). Monkeypox was considered the cause or a contributing factor in five of the deaths and not a factor in one death; the remaining six deaths are under investigation.
Case studies
The report included details of three representative cases of the CDC consultations.
One was a Hispanic man in his 20s with a fever of 102.8° F, a rash including eschars, oral lesions, neck mass, and cervical lymphadenopathy. He had tested positive for monkeypox as an outpatient and upon admission was found to be HIV positive, with a CD4 count of 79 cells/mm3. He experienced a severe and ultimately fatal clinical course that included intubation, refractory hypotension, seizures, renal failure, and cardiac arrest. An autopsy revealed diffuse organ necrosis plus orthopoxvirus and cytomegalovirus.
The second was a Black man in his 30s with untreated AIDS and diffuse rash. He was tested and treated for gonorrhea, chlamydia, and syphilis before phimosis and urinary retention led to admission and a monkeypox diagnosis 4 weeks after his rash began. He was discharged with oral tecovirimat, but his skin lesions developed necrosis and he was readmitted twice, each time with new lesions. His clinical course included methicillin-resistant Staphylococcus aureus bacteremia, atrial fibrillation, eye and ear involvement, a suprapubic catheter, and progressive necrosis of his lesions. As of the CDC report, he was receiving ART and intravenous tecovirimat.
The third patient, a White man in his 40s with untreated AIDS, presented with diffuse rash. He was promptly diagnosed with monkeypox and admitted for pain control. He was discharged with oral tecovirimat and ART, but homelessness and food insecurity jeopardized the absorption of his tecovirimat (which depends on a full fatty meal), and the lesions worsened. Despite readmission and aggressive medical treatment, the patient required finger debridement and a toe amputation. After discharge, he was again readmitted for lesions and pain and, at report publication, remained hospitalized, taking oral tecovirimat and ART.
The patients in the study may not be typical of severe monkeypox cases, wrote the authors reported. Deaths after the study period were not counted.
Fewer cases, some severe
As of Nov. 7, the CDC has confirmed 28,709 monkeypox cases. These have trended downward since August. Most people with recent diagnoses are men who are gay, bisexual, same gender loving, or who have sex with men, and most are Black, according to Brooks.
Dr. Brooks urges clinicians to report suspected monkeypox cases – especially severe ones – to their health departments.
“We don’t have a good bead on exactly how many severe cases there are in the States because of complexities in our surveillance systems,” Dr. Brooks said.
For patients with suspected or confirmed monkeypox, Brooks recommends testing for sexually transmitted infections, including HIV if status is unknown. Patients with HIV should receive prompt ART. For those at risk for severe disease, the authors recommend early treatment for suspected monkeypox, even before results are back. Some patients may benefit from tecovirimat courses lasting beyond 14 days, plus additional antivirals (cidofovir or brincidofovir) and/or VIGIV.
“With severe cases, clinicians may want to consider the value of more than one drug to attack the virus at different stages of its replication cycle,” Dr. Brooks said.
Inequities matter
The authors called on providers to engage communities burdened by HIV and to ensure access to not only monkeypox vaccination, diagnosis, and treatment but also sustained HIV care.
Dr. Silvera added that providers need to tailor care plans to patients’ social determinants of health. For example, he explained, inpatient care for monkeypox could be appropriate for some patients facing homelessness and food insecurity – even if they are able to take tecovirimat orally.
He recommends tapping others’ expertise: “Our social work colleagues are well versed in this.”
“I don’t think these clinicians failed these patients. ... I think everyone made all the right choices medically,” Dr. Silvera added. “I think that the system failed these patients – and we as clinicians are part of those systems. So we also have the power to change those systems. And I think we just need to start opening our eyes to that and [start] to work together towards that goal to take better care of our patients.”
Dr. Brooks reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Monkeypox, though often mild, may be severe and even fatal in immunocompromised individuals, particularly those with untreated AIDS, according to a Centers for Disease Control and Prevention study in Morbidity and Mortality Weekly Report.
The study described a group of patients recently treated for severe monkeypox. The majority were Black, HIV positive, and not receiving treatment. Many were also facing homelessness.
The authors urged HIV testing for all sexually active individuals with suspected monkeypox. Early or prolonged monkeypox treatment may be necessary, they concluded.
Coauthor John T. Brooks, MD, called the study “a real call to action.”
“If we want to reduce cases of severe monkeypox, we need to reduce the number of persons with HIV who are undiagnosed and not treated,” said Dr. Brooks, a medical epidemiologist who is chief medical officer of CDC›s multinational monkeypox response. Dr. Brooks also leads the epidemiology research team in CDC’s division of HIV/AIDS prevention.
noted Richard Silvera, MD, MPH, CPH, who is associate program director of the infectious diseases fellowship and assistant professor of medicine (infectious diseases) at the Icahn School of Medicine at Mount Sinai, New York. He was not involved with the study.
“These patients really have not been served by the health care system,” Dr. Silvera said. “Monkeypox is just really taking advantage of that.”
How severe monkeypox can manifest
The authors reported on 57 adults hospitalized with severe monkeypox between Aug. 10 and Sept. 10, 2022, for whose care the providers sought CDC consultation.
The vast majority (95%) were men, their median age was 34 years, and 68% were Black. Nearly one in four were homeless (23%).
Overall, 47 (82%) were HIV positive, of whom just 4 had been receiving antiretroviral therapy (ART). Of 43 for whom CD4 counts were known, 71% had fewer than 50 CD4 cells/mm3.
Clinical signs included severe skin lesions in all patients and severe mucosal lesions in 68%. Other affected organ systems included lungs (21%), eyes (21%), and central nervous system (7%).
Treatments included oral or intravenous tecovirimat (93% and 65%, respectively), vaccinia immune globulin intravenous (VIGIV, 51%), and cidofovir (23%).
Nearly 1 in 3 patients (30%) received care in an ICU; 12 died (21%). Monkeypox was considered the cause or a contributing factor in five of the deaths and not a factor in one death; the remaining six deaths are under investigation.
Case studies
The report included details of three representative cases of the CDC consultations.
One was a Hispanic man in his 20s with a fever of 102.8° F, a rash including eschars, oral lesions, neck mass, and cervical lymphadenopathy. He had tested positive for monkeypox as an outpatient and upon admission was found to be HIV positive, with a CD4 count of 79 cells/mm3. He experienced a severe and ultimately fatal clinical course that included intubation, refractory hypotension, seizures, renal failure, and cardiac arrest. An autopsy revealed diffuse organ necrosis plus orthopoxvirus and cytomegalovirus.
The second was a Black man in his 30s with untreated AIDS and diffuse rash. He was tested and treated for gonorrhea, chlamydia, and syphilis before phimosis and urinary retention led to admission and a monkeypox diagnosis 4 weeks after his rash began. He was discharged with oral tecovirimat, but his skin lesions developed necrosis and he was readmitted twice, each time with new lesions. His clinical course included methicillin-resistant Staphylococcus aureus bacteremia, atrial fibrillation, eye and ear involvement, a suprapubic catheter, and progressive necrosis of his lesions. As of the CDC report, he was receiving ART and intravenous tecovirimat.
The third patient, a White man in his 40s with untreated AIDS, presented with diffuse rash. He was promptly diagnosed with monkeypox and admitted for pain control. He was discharged with oral tecovirimat and ART, but homelessness and food insecurity jeopardized the absorption of his tecovirimat (which depends on a full fatty meal), and the lesions worsened. Despite readmission and aggressive medical treatment, the patient required finger debridement and a toe amputation. After discharge, he was again readmitted for lesions and pain and, at report publication, remained hospitalized, taking oral tecovirimat and ART.
The patients in the study may not be typical of severe monkeypox cases, wrote the authors reported. Deaths after the study period were not counted.
Fewer cases, some severe
As of Nov. 7, the CDC has confirmed 28,709 monkeypox cases. These have trended downward since August. Most people with recent diagnoses are men who are gay, bisexual, same gender loving, or who have sex with men, and most are Black, according to Brooks.
Dr. Brooks urges clinicians to report suspected monkeypox cases – especially severe ones – to their health departments.
“We don’t have a good bead on exactly how many severe cases there are in the States because of complexities in our surveillance systems,” Dr. Brooks said.
For patients with suspected or confirmed monkeypox, Brooks recommends testing for sexually transmitted infections, including HIV if status is unknown. Patients with HIV should receive prompt ART. For those at risk for severe disease, the authors recommend early treatment for suspected monkeypox, even before results are back. Some patients may benefit from tecovirimat courses lasting beyond 14 days, plus additional antivirals (cidofovir or brincidofovir) and/or VIGIV.
“With severe cases, clinicians may want to consider the value of more than one drug to attack the virus at different stages of its replication cycle,” Dr. Brooks said.
Inequities matter
The authors called on providers to engage communities burdened by HIV and to ensure access to not only monkeypox vaccination, diagnosis, and treatment but also sustained HIV care.
Dr. Silvera added that providers need to tailor care plans to patients’ social determinants of health. For example, he explained, inpatient care for monkeypox could be appropriate for some patients facing homelessness and food insecurity – even if they are able to take tecovirimat orally.
He recommends tapping others’ expertise: “Our social work colleagues are well versed in this.”
“I don’t think these clinicians failed these patients. ... I think everyone made all the right choices medically,” Dr. Silvera added. “I think that the system failed these patients – and we as clinicians are part of those systems. So we also have the power to change those systems. And I think we just need to start opening our eyes to that and [start] to work together towards that goal to take better care of our patients.”
Dr. Brooks reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Monkeypox, though often mild, may be severe and even fatal in immunocompromised individuals, particularly those with untreated AIDS, according to a Centers for Disease Control and Prevention study in Morbidity and Mortality Weekly Report.
The study described a group of patients recently treated for severe monkeypox. The majority were Black, HIV positive, and not receiving treatment. Many were also facing homelessness.
The authors urged HIV testing for all sexually active individuals with suspected monkeypox. Early or prolonged monkeypox treatment may be necessary, they concluded.
Coauthor John T. Brooks, MD, called the study “a real call to action.”
“If we want to reduce cases of severe monkeypox, we need to reduce the number of persons with HIV who are undiagnosed and not treated,” said Dr. Brooks, a medical epidemiologist who is chief medical officer of CDC›s multinational monkeypox response. Dr. Brooks also leads the epidemiology research team in CDC’s division of HIV/AIDS prevention.
noted Richard Silvera, MD, MPH, CPH, who is associate program director of the infectious diseases fellowship and assistant professor of medicine (infectious diseases) at the Icahn School of Medicine at Mount Sinai, New York. He was not involved with the study.
“These patients really have not been served by the health care system,” Dr. Silvera said. “Monkeypox is just really taking advantage of that.”
How severe monkeypox can manifest
The authors reported on 57 adults hospitalized with severe monkeypox between Aug. 10 and Sept. 10, 2022, for whose care the providers sought CDC consultation.
The vast majority (95%) were men, their median age was 34 years, and 68% were Black. Nearly one in four were homeless (23%).
Overall, 47 (82%) were HIV positive, of whom just 4 had been receiving antiretroviral therapy (ART). Of 43 for whom CD4 counts were known, 71% had fewer than 50 CD4 cells/mm3.
Clinical signs included severe skin lesions in all patients and severe mucosal lesions in 68%. Other affected organ systems included lungs (21%), eyes (21%), and central nervous system (7%).
Treatments included oral or intravenous tecovirimat (93% and 65%, respectively), vaccinia immune globulin intravenous (VIGIV, 51%), and cidofovir (23%).
Nearly 1 in 3 patients (30%) received care in an ICU; 12 died (21%). Monkeypox was considered the cause or a contributing factor in five of the deaths and not a factor in one death; the remaining six deaths are under investigation.
Case studies
The report included details of three representative cases of the CDC consultations.
One was a Hispanic man in his 20s with a fever of 102.8° F, a rash including eschars, oral lesions, neck mass, and cervical lymphadenopathy. He had tested positive for monkeypox as an outpatient and upon admission was found to be HIV positive, with a CD4 count of 79 cells/mm3. He experienced a severe and ultimately fatal clinical course that included intubation, refractory hypotension, seizures, renal failure, and cardiac arrest. An autopsy revealed diffuse organ necrosis plus orthopoxvirus and cytomegalovirus.
The second was a Black man in his 30s with untreated AIDS and diffuse rash. He was tested and treated for gonorrhea, chlamydia, and syphilis before phimosis and urinary retention led to admission and a monkeypox diagnosis 4 weeks after his rash began. He was discharged with oral tecovirimat, but his skin lesions developed necrosis and he was readmitted twice, each time with new lesions. His clinical course included methicillin-resistant Staphylococcus aureus bacteremia, atrial fibrillation, eye and ear involvement, a suprapubic catheter, and progressive necrosis of his lesions. As of the CDC report, he was receiving ART and intravenous tecovirimat.
The third patient, a White man in his 40s with untreated AIDS, presented with diffuse rash. He was promptly diagnosed with monkeypox and admitted for pain control. He was discharged with oral tecovirimat and ART, but homelessness and food insecurity jeopardized the absorption of his tecovirimat (which depends on a full fatty meal), and the lesions worsened. Despite readmission and aggressive medical treatment, the patient required finger debridement and a toe amputation. After discharge, he was again readmitted for lesions and pain and, at report publication, remained hospitalized, taking oral tecovirimat and ART.
The patients in the study may not be typical of severe monkeypox cases, wrote the authors reported. Deaths after the study period were not counted.
Fewer cases, some severe
As of Nov. 7, the CDC has confirmed 28,709 monkeypox cases. These have trended downward since August. Most people with recent diagnoses are men who are gay, bisexual, same gender loving, or who have sex with men, and most are Black, according to Brooks.
Dr. Brooks urges clinicians to report suspected monkeypox cases – especially severe ones – to their health departments.
“We don’t have a good bead on exactly how many severe cases there are in the States because of complexities in our surveillance systems,” Dr. Brooks said.
For patients with suspected or confirmed monkeypox, Brooks recommends testing for sexually transmitted infections, including HIV if status is unknown. Patients with HIV should receive prompt ART. For those at risk for severe disease, the authors recommend early treatment for suspected monkeypox, even before results are back. Some patients may benefit from tecovirimat courses lasting beyond 14 days, plus additional antivirals (cidofovir or brincidofovir) and/or VIGIV.
“With severe cases, clinicians may want to consider the value of more than one drug to attack the virus at different stages of its replication cycle,” Dr. Brooks said.
Inequities matter
The authors called on providers to engage communities burdened by HIV and to ensure access to not only monkeypox vaccination, diagnosis, and treatment but also sustained HIV care.
Dr. Silvera added that providers need to tailor care plans to patients’ social determinants of health. For example, he explained, inpatient care for monkeypox could be appropriate for some patients facing homelessness and food insecurity – even if they are able to take tecovirimat orally.
He recommends tapping others’ expertise: “Our social work colleagues are well versed in this.”
“I don’t think these clinicians failed these patients. ... I think everyone made all the right choices medically,” Dr. Silvera added. “I think that the system failed these patients – and we as clinicians are part of those systems. So we also have the power to change those systems. And I think we just need to start opening our eyes to that and [start] to work together towards that goal to take better care of our patients.”
Dr. Brooks reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM THE MMWR
New dual-agonist weight-loss injection impressive, but early days
SAN DIEGO – A novel glucagonlike peptide-1 (GLP-1)/glucagon dual-receptor agonist, BI 456906, being developed by Boehringer Ingelheim and Zealand Pharma, led to “impressive” weight loss in a phase 2 dosing study of patients with overweight/obesity and type 2 diabetes – but this is early research.
Julio Rosenstock, MD, presented the study results, including weight loss and adverse events, at the annual meeting of the Obesity Society.
At the highest tested dose (1.8 mg twice weekly subcutaneous injections), 57% of patients lost at least 5% of their initial body weight and 35% lost at least 10% of their initial body weight at 16 weeks.
In contrast, among the patients who received a 1-mg semaglutide dose as a comparator, 38% lost at least 5% of their initial body weight and 16% lost at least 10% of their initial body weight at study end.
This is “very promising data as an anti-obesity compound,” said Dr. Rosenstock, professor of medicine, University of Texas Southwestern Medical Center in Dallas.
The researchers enrolled 411 adults and randomized them into eight groups of roughly 50 patients each.
They compared six doses of BI 456906 (from 0.3 mg/week to 1.8 mg twice weekly) versus 1 mg/week of the GLP-1 agonist semaglutide (Wegovy, Novo Nordisk) versus placebo.
Patients had a mean initial weight of 97 kg (214 pounds).
After 4 months, on average, patients who received the highest tested dose of BI 456906 lost 9% of their initial weight or roughly 8.7 kg (19 pounds).
Patients who received semaglutide lost 5.4% of their initial weight or roughly 5.2 kg (11.5 pounds), and patients who received placebo lost only 1.2% of their initial weight
The main adverse events were gastrointestinal.
‘Exciting data,’ but still early days
“This is very exciting data. It comes from another experienced company with a track record of successful products with a new compound in a class where other related compounds have shown efficacy and safety,” Dan Bessesen, MD, president of The Obesity Society, who was not involved with this research, told this news organization in an email.
“The degree of weight loss is impressive for a 16-week study,” Dr. Bessesen, professor of medicine in the division of endocrinology, metabolism and diabetes at the University of Colorado at Denver, Aurora, added. “The longer-term weight loss will likely be more.”
The side-effect profile is not particularly concerning and is like other drugs in this general class, he said.
However, he also noted a few caveats. This was only a phase 2 study, “so we should not make firm conclusions about efficacy from a study like this, as the number of subjects studied at each dose is relatively small and the follow-up not long.”
In addition, “the dose of semaglutide is the old ‘diabetes’ dose (1 mg) not the weight-loss dose of 2.4 mg or the new diabetes dose of 2 mg. It is not a real comparison with the maximal approved dose of semaglutide. So, we cannot say that it will be better than semaglutide.”
The next hurdle is the “need to see phase 3 studies in a larger group of patients studied for a longer time. Then [the company] will need FDA approval, so it may be a bit of time” before this drug potentially enters the marketplace.
The “bottom line” is that this potential new antiobesity/diabetes drug is “very promising, but [it is] still a little early to say where it ultimately will go.”
A1c results presented at EASD
To be included in this study, patients had to be 18-75 years old, have type 2 diabetes, a body mass index of 25-50 kg/m2, and hemoglobin A1c of 7%-10%, and be stable on metformin therapy.
The patients had a mean age of 57 years, and 57% were men. They had a mean A1c of 8.1%, a mean BMI of 34 kg/m2, and a mean waist circumference of 110 cm (43 inches).
“We just recently reported at the EASD conference last month, the effect of BI 456906 on A1c lowering,” Dr. Rosenstock said.
“It looks like the [drop in] A1c plateaus at 1.9%, which is pretty good when you consider the baseline A1c is around 8%. You get down to around 6%, which is what we regard as a very robust reduction in people with type 2 diabetes on metformin.”
The current analysis showed that patients who received doses of 0.3, 0.9, 1.8, and 2.7 mg/week of the novel drug lost 1.9%, 4.4%, 6.6%, and 6.7% of their initial body weight, respectively, after 16 weeks.
The patients who received 1.2 mg and 1.8 mg twice weekly lost even more weight, 7.2% and 9% of their initial weight, respectively.
At the highest dose, on average, patients lost 13 cm (5 inches) around their waist.
Adverse events were reported by 78% of the patients, most commonly nausea (34% of patients), vomiting (18%), and diarrhea (16%).
Only 1.3% of patients had a drug-related serious adverse event. A total of 16% of patients discontinued the therapy.
Most of the “gastrointestinal adverse events leading the treatment discontinuation were possibly dose and titration related,” Dr. Rosenstock said, “and it’s highly conceivable that for future studies a slower dose escalation may mitigate the occurrence of the gastrointestinal adverse events.”
BI 456906 was coinvented with Zealand Pharma. Under the licensing agreement, Boehringer Ingelheim funds all research, development, and commercialization.
A version of this article first appeared on Medscape.com.
SAN DIEGO – A novel glucagonlike peptide-1 (GLP-1)/glucagon dual-receptor agonist, BI 456906, being developed by Boehringer Ingelheim and Zealand Pharma, led to “impressive” weight loss in a phase 2 dosing study of patients with overweight/obesity and type 2 diabetes – but this is early research.
Julio Rosenstock, MD, presented the study results, including weight loss and adverse events, at the annual meeting of the Obesity Society.
At the highest tested dose (1.8 mg twice weekly subcutaneous injections), 57% of patients lost at least 5% of their initial body weight and 35% lost at least 10% of their initial body weight at 16 weeks.
In contrast, among the patients who received a 1-mg semaglutide dose as a comparator, 38% lost at least 5% of their initial body weight and 16% lost at least 10% of their initial body weight at study end.
This is “very promising data as an anti-obesity compound,” said Dr. Rosenstock, professor of medicine, University of Texas Southwestern Medical Center in Dallas.
The researchers enrolled 411 adults and randomized them into eight groups of roughly 50 patients each.
They compared six doses of BI 456906 (from 0.3 mg/week to 1.8 mg twice weekly) versus 1 mg/week of the GLP-1 agonist semaglutide (Wegovy, Novo Nordisk) versus placebo.
Patients had a mean initial weight of 97 kg (214 pounds).
After 4 months, on average, patients who received the highest tested dose of BI 456906 lost 9% of their initial weight or roughly 8.7 kg (19 pounds).
Patients who received semaglutide lost 5.4% of their initial weight or roughly 5.2 kg (11.5 pounds), and patients who received placebo lost only 1.2% of their initial weight
The main adverse events were gastrointestinal.
‘Exciting data,’ but still early days
“This is very exciting data. It comes from another experienced company with a track record of successful products with a new compound in a class where other related compounds have shown efficacy and safety,” Dan Bessesen, MD, president of The Obesity Society, who was not involved with this research, told this news organization in an email.
“The degree of weight loss is impressive for a 16-week study,” Dr. Bessesen, professor of medicine in the division of endocrinology, metabolism and diabetes at the University of Colorado at Denver, Aurora, added. “The longer-term weight loss will likely be more.”
The side-effect profile is not particularly concerning and is like other drugs in this general class, he said.
However, he also noted a few caveats. This was only a phase 2 study, “so we should not make firm conclusions about efficacy from a study like this, as the number of subjects studied at each dose is relatively small and the follow-up not long.”
In addition, “the dose of semaglutide is the old ‘diabetes’ dose (1 mg) not the weight-loss dose of 2.4 mg or the new diabetes dose of 2 mg. It is not a real comparison with the maximal approved dose of semaglutide. So, we cannot say that it will be better than semaglutide.”
The next hurdle is the “need to see phase 3 studies in a larger group of patients studied for a longer time. Then [the company] will need FDA approval, so it may be a bit of time” before this drug potentially enters the marketplace.
The “bottom line” is that this potential new antiobesity/diabetes drug is “very promising, but [it is] still a little early to say where it ultimately will go.”
A1c results presented at EASD
To be included in this study, patients had to be 18-75 years old, have type 2 diabetes, a body mass index of 25-50 kg/m2, and hemoglobin A1c of 7%-10%, and be stable on metformin therapy.
The patients had a mean age of 57 years, and 57% were men. They had a mean A1c of 8.1%, a mean BMI of 34 kg/m2, and a mean waist circumference of 110 cm (43 inches).
“We just recently reported at the EASD conference last month, the effect of BI 456906 on A1c lowering,” Dr. Rosenstock said.
“It looks like the [drop in] A1c plateaus at 1.9%, which is pretty good when you consider the baseline A1c is around 8%. You get down to around 6%, which is what we regard as a very robust reduction in people with type 2 diabetes on metformin.”
The current analysis showed that patients who received doses of 0.3, 0.9, 1.8, and 2.7 mg/week of the novel drug lost 1.9%, 4.4%, 6.6%, and 6.7% of their initial body weight, respectively, after 16 weeks.
The patients who received 1.2 mg and 1.8 mg twice weekly lost even more weight, 7.2% and 9% of their initial weight, respectively.
At the highest dose, on average, patients lost 13 cm (5 inches) around their waist.
Adverse events were reported by 78% of the patients, most commonly nausea (34% of patients), vomiting (18%), and diarrhea (16%).
Only 1.3% of patients had a drug-related serious adverse event. A total of 16% of patients discontinued the therapy.
Most of the “gastrointestinal adverse events leading the treatment discontinuation were possibly dose and titration related,” Dr. Rosenstock said, “and it’s highly conceivable that for future studies a slower dose escalation may mitigate the occurrence of the gastrointestinal adverse events.”
BI 456906 was coinvented with Zealand Pharma. Under the licensing agreement, Boehringer Ingelheim funds all research, development, and commercialization.
A version of this article first appeared on Medscape.com.
SAN DIEGO – A novel glucagonlike peptide-1 (GLP-1)/glucagon dual-receptor agonist, BI 456906, being developed by Boehringer Ingelheim and Zealand Pharma, led to “impressive” weight loss in a phase 2 dosing study of patients with overweight/obesity and type 2 diabetes – but this is early research.
Julio Rosenstock, MD, presented the study results, including weight loss and adverse events, at the annual meeting of the Obesity Society.
At the highest tested dose (1.8 mg twice weekly subcutaneous injections), 57% of patients lost at least 5% of their initial body weight and 35% lost at least 10% of their initial body weight at 16 weeks.
In contrast, among the patients who received a 1-mg semaglutide dose as a comparator, 38% lost at least 5% of their initial body weight and 16% lost at least 10% of their initial body weight at study end.
This is “very promising data as an anti-obesity compound,” said Dr. Rosenstock, professor of medicine, University of Texas Southwestern Medical Center in Dallas.
The researchers enrolled 411 adults and randomized them into eight groups of roughly 50 patients each.
They compared six doses of BI 456906 (from 0.3 mg/week to 1.8 mg twice weekly) versus 1 mg/week of the GLP-1 agonist semaglutide (Wegovy, Novo Nordisk) versus placebo.
Patients had a mean initial weight of 97 kg (214 pounds).
After 4 months, on average, patients who received the highest tested dose of BI 456906 lost 9% of their initial weight or roughly 8.7 kg (19 pounds).
Patients who received semaglutide lost 5.4% of their initial weight or roughly 5.2 kg (11.5 pounds), and patients who received placebo lost only 1.2% of their initial weight
The main adverse events were gastrointestinal.
‘Exciting data,’ but still early days
“This is very exciting data. It comes from another experienced company with a track record of successful products with a new compound in a class where other related compounds have shown efficacy and safety,” Dan Bessesen, MD, president of The Obesity Society, who was not involved with this research, told this news organization in an email.
“The degree of weight loss is impressive for a 16-week study,” Dr. Bessesen, professor of medicine in the division of endocrinology, metabolism and diabetes at the University of Colorado at Denver, Aurora, added. “The longer-term weight loss will likely be more.”
The side-effect profile is not particularly concerning and is like other drugs in this general class, he said.
However, he also noted a few caveats. This was only a phase 2 study, “so we should not make firm conclusions about efficacy from a study like this, as the number of subjects studied at each dose is relatively small and the follow-up not long.”
In addition, “the dose of semaglutide is the old ‘diabetes’ dose (1 mg) not the weight-loss dose of 2.4 mg or the new diabetes dose of 2 mg. It is not a real comparison with the maximal approved dose of semaglutide. So, we cannot say that it will be better than semaglutide.”
The next hurdle is the “need to see phase 3 studies in a larger group of patients studied for a longer time. Then [the company] will need FDA approval, so it may be a bit of time” before this drug potentially enters the marketplace.
The “bottom line” is that this potential new antiobesity/diabetes drug is “very promising, but [it is] still a little early to say where it ultimately will go.”
A1c results presented at EASD
To be included in this study, patients had to be 18-75 years old, have type 2 diabetes, a body mass index of 25-50 kg/m2, and hemoglobin A1c of 7%-10%, and be stable on metformin therapy.
The patients had a mean age of 57 years, and 57% were men. They had a mean A1c of 8.1%, a mean BMI of 34 kg/m2, and a mean waist circumference of 110 cm (43 inches).
“We just recently reported at the EASD conference last month, the effect of BI 456906 on A1c lowering,” Dr. Rosenstock said.
“It looks like the [drop in] A1c plateaus at 1.9%, which is pretty good when you consider the baseline A1c is around 8%. You get down to around 6%, which is what we regard as a very robust reduction in people with type 2 diabetes on metformin.”
The current analysis showed that patients who received doses of 0.3, 0.9, 1.8, and 2.7 mg/week of the novel drug lost 1.9%, 4.4%, 6.6%, and 6.7% of their initial body weight, respectively, after 16 weeks.
The patients who received 1.2 mg and 1.8 mg twice weekly lost even more weight, 7.2% and 9% of their initial weight, respectively.
At the highest dose, on average, patients lost 13 cm (5 inches) around their waist.
Adverse events were reported by 78% of the patients, most commonly nausea (34% of patients), vomiting (18%), and diarrhea (16%).
Only 1.3% of patients had a drug-related serious adverse event. A total of 16% of patients discontinued the therapy.
Most of the “gastrointestinal adverse events leading the treatment discontinuation were possibly dose and titration related,” Dr. Rosenstock said, “and it’s highly conceivable that for future studies a slower dose escalation may mitigate the occurrence of the gastrointestinal adverse events.”
BI 456906 was coinvented with Zealand Pharma. Under the licensing agreement, Boehringer Ingelheim funds all research, development, and commercialization.
A version of this article first appeared on Medscape.com.
AT OBESITYWEEK® 2022
A hormone that can predict male long-term health
Insulin-like peptide 3 (INSL3) is a constitutive hormone secreted in men by the mature Leydig cells of the testes, explained the authors of the new study, published in Frontiers in Endocrinology.
“It is an accurate biomarker for Leydig cell functional capacity, reflecting their total cell number and differentiation status,” they said.
“The holy grail of aging research is to reduce the fitness gap that appears as people age,” said Ravinder Anand-Ivell, PhD, associate professor in endocrinology and reproductive physiology at the University of Nottingham (England), and study coauthor. Understanding why some people are more likely to develop disability and disease as they age is “vital” so that interventions can be found to ensure people not only live a long life but also a healthy life as they age, she highlighted.
The European team of researchers, led by scientists from the University of Nottingham, set out to determine the ability of INSL3 as a biomarker to predict hypogonadism and age-related morbidity, and whether this also allowed it to predict morbidity in a similar way to testosterone.
For the study, the researchers analyzed blood samples from the European Male Aging Study (EMAS) cohort to assess circulating INSL3 and its cross-sectional and longitudinal relationships to hypogonadism – defined by testosterone less than 10.5 nmol/L – and a range of age-related morbidities determined by correlation and regression analysis.
The EMAS cohort of community-dwelling men comprises more than 3,000 men, aged 40-79 years at the time of recruitment, from eight centers in Europe. Men were recruited from 2003 to 2004 and again 4-5 years later for a second phase of the study. In both phases, blood was collected for hormonal measurements, and subjects were assessed for anthropometric parameters and asked to complete questionnaires relating to their health, lifestyle, and diet.
Hormone levels remain constant
The results showed that, unlike testosterone, which fluctuates throughout a man’s life, INSL3 remains consistent, with the level at puberty remaining largely the same throughout a man’s life, decreasing only slightly into old age. “This makes it the first clear and reliable predictive biomarker of age-related morbidity as compared with any other measurable parameters,” explained the researchers.
They also discovered that the level of INSL3 in blood “correlates with a range of age-related conditions,” such as bone weakness, sexual dysfunction, diabetes, and cardiovascular disease.
They emphasized that the discovery of the consistent nature of this hormone is “very significant.” It means that a man with high INSL3 when young will still have high INSL3 when he is older, but someone with low INSL3 already at a young age will have low INSL3 when older, “making him more likely to acquire typical age-related illnesses.”
Dr. Anand-Ivell commented that the hormone discovery was an “important step” and will pave the way for not only helping people individually but also helping to “ease the care crisis we face as a society.”
Exciting possibilities for predicting age
The study also showed that the normal male population, even when young and relatively healthy, still shows an almost 10-fold variation between individuals in the concentration of INSL3 in the blood, the authors reported.
The authors highlighted that the study’s strengths are the large and comprehensive dataset provided by the EMAS cohort, together with the accuracy of the hormonal parameters measured. The weaknesses, they explained, are the self-reported nature of some of the morbidity parameters as well as the relatively short longitudinal dimension of only 4.3 years average.
Richard Ivell, University of Nottingham, and lead author, explained that now the important role of INSL3 in predicting disease, and how it varies amongst men, had been established, the team is looking to investigate what factors have the most influence on the level of INSL3 in the blood. “Preliminary work suggests early life nutrition may play a role, but many other factors such as genetics or exposure to some environmental endocrine disruptors may play a part”.
The study findings open up “exciting possibilities for predicting age-related illnesses and finding ways to prevent the onset of these diseases with early intervention,” the authors enthused.
The study was initiated and supported by the European 5th Framework, and the German Research Council provided funding for the INSL3 analysis. The authors declared no conflicts of interest.
Dr. Hicks has disclosed no relevant financial relationships. A version of this article first appeared on MedscapeUK.
Insulin-like peptide 3 (INSL3) is a constitutive hormone secreted in men by the mature Leydig cells of the testes, explained the authors of the new study, published in Frontiers in Endocrinology.
“It is an accurate biomarker for Leydig cell functional capacity, reflecting their total cell number and differentiation status,” they said.
“The holy grail of aging research is to reduce the fitness gap that appears as people age,” said Ravinder Anand-Ivell, PhD, associate professor in endocrinology and reproductive physiology at the University of Nottingham (England), and study coauthor. Understanding why some people are more likely to develop disability and disease as they age is “vital” so that interventions can be found to ensure people not only live a long life but also a healthy life as they age, she highlighted.
The European team of researchers, led by scientists from the University of Nottingham, set out to determine the ability of INSL3 as a biomarker to predict hypogonadism and age-related morbidity, and whether this also allowed it to predict morbidity in a similar way to testosterone.
For the study, the researchers analyzed blood samples from the European Male Aging Study (EMAS) cohort to assess circulating INSL3 and its cross-sectional and longitudinal relationships to hypogonadism – defined by testosterone less than 10.5 nmol/L – and a range of age-related morbidities determined by correlation and regression analysis.
The EMAS cohort of community-dwelling men comprises more than 3,000 men, aged 40-79 years at the time of recruitment, from eight centers in Europe. Men were recruited from 2003 to 2004 and again 4-5 years later for a second phase of the study. In both phases, blood was collected for hormonal measurements, and subjects were assessed for anthropometric parameters and asked to complete questionnaires relating to their health, lifestyle, and diet.
Hormone levels remain constant
The results showed that, unlike testosterone, which fluctuates throughout a man’s life, INSL3 remains consistent, with the level at puberty remaining largely the same throughout a man’s life, decreasing only slightly into old age. “This makes it the first clear and reliable predictive biomarker of age-related morbidity as compared with any other measurable parameters,” explained the researchers.
They also discovered that the level of INSL3 in blood “correlates with a range of age-related conditions,” such as bone weakness, sexual dysfunction, diabetes, and cardiovascular disease.
They emphasized that the discovery of the consistent nature of this hormone is “very significant.” It means that a man with high INSL3 when young will still have high INSL3 when he is older, but someone with low INSL3 already at a young age will have low INSL3 when older, “making him more likely to acquire typical age-related illnesses.”
Dr. Anand-Ivell commented that the hormone discovery was an “important step” and will pave the way for not only helping people individually but also helping to “ease the care crisis we face as a society.”
Exciting possibilities for predicting age
The study also showed that the normal male population, even when young and relatively healthy, still shows an almost 10-fold variation between individuals in the concentration of INSL3 in the blood, the authors reported.
The authors highlighted that the study’s strengths are the large and comprehensive dataset provided by the EMAS cohort, together with the accuracy of the hormonal parameters measured. The weaknesses, they explained, are the self-reported nature of some of the morbidity parameters as well as the relatively short longitudinal dimension of only 4.3 years average.
Richard Ivell, University of Nottingham, and lead author, explained that now the important role of INSL3 in predicting disease, and how it varies amongst men, had been established, the team is looking to investigate what factors have the most influence on the level of INSL3 in the blood. “Preliminary work suggests early life nutrition may play a role, but many other factors such as genetics or exposure to some environmental endocrine disruptors may play a part”.
The study findings open up “exciting possibilities for predicting age-related illnesses and finding ways to prevent the onset of these diseases with early intervention,” the authors enthused.
The study was initiated and supported by the European 5th Framework, and the German Research Council provided funding for the INSL3 analysis. The authors declared no conflicts of interest.
Dr. Hicks has disclosed no relevant financial relationships. A version of this article first appeared on MedscapeUK.
Insulin-like peptide 3 (INSL3) is a constitutive hormone secreted in men by the mature Leydig cells of the testes, explained the authors of the new study, published in Frontiers in Endocrinology.
“It is an accurate biomarker for Leydig cell functional capacity, reflecting their total cell number and differentiation status,” they said.
“The holy grail of aging research is to reduce the fitness gap that appears as people age,” said Ravinder Anand-Ivell, PhD, associate professor in endocrinology and reproductive physiology at the University of Nottingham (England), and study coauthor. Understanding why some people are more likely to develop disability and disease as they age is “vital” so that interventions can be found to ensure people not only live a long life but also a healthy life as they age, she highlighted.
The European team of researchers, led by scientists from the University of Nottingham, set out to determine the ability of INSL3 as a biomarker to predict hypogonadism and age-related morbidity, and whether this also allowed it to predict morbidity in a similar way to testosterone.
For the study, the researchers analyzed blood samples from the European Male Aging Study (EMAS) cohort to assess circulating INSL3 and its cross-sectional and longitudinal relationships to hypogonadism – defined by testosterone less than 10.5 nmol/L – and a range of age-related morbidities determined by correlation and regression analysis.
The EMAS cohort of community-dwelling men comprises more than 3,000 men, aged 40-79 years at the time of recruitment, from eight centers in Europe. Men were recruited from 2003 to 2004 and again 4-5 years later for a second phase of the study. In both phases, blood was collected for hormonal measurements, and subjects were assessed for anthropometric parameters and asked to complete questionnaires relating to their health, lifestyle, and diet.
Hormone levels remain constant
The results showed that, unlike testosterone, which fluctuates throughout a man’s life, INSL3 remains consistent, with the level at puberty remaining largely the same throughout a man’s life, decreasing only slightly into old age. “This makes it the first clear and reliable predictive biomarker of age-related morbidity as compared with any other measurable parameters,” explained the researchers.
They also discovered that the level of INSL3 in blood “correlates with a range of age-related conditions,” such as bone weakness, sexual dysfunction, diabetes, and cardiovascular disease.
They emphasized that the discovery of the consistent nature of this hormone is “very significant.” It means that a man with high INSL3 when young will still have high INSL3 when he is older, but someone with low INSL3 already at a young age will have low INSL3 when older, “making him more likely to acquire typical age-related illnesses.”
Dr. Anand-Ivell commented that the hormone discovery was an “important step” and will pave the way for not only helping people individually but also helping to “ease the care crisis we face as a society.”
Exciting possibilities for predicting age
The study also showed that the normal male population, even when young and relatively healthy, still shows an almost 10-fold variation between individuals in the concentration of INSL3 in the blood, the authors reported.
The authors highlighted that the study’s strengths are the large and comprehensive dataset provided by the EMAS cohort, together with the accuracy of the hormonal parameters measured. The weaknesses, they explained, are the self-reported nature of some of the morbidity parameters as well as the relatively short longitudinal dimension of only 4.3 years average.
Richard Ivell, University of Nottingham, and lead author, explained that now the important role of INSL3 in predicting disease, and how it varies amongst men, had been established, the team is looking to investigate what factors have the most influence on the level of INSL3 in the blood. “Preliminary work suggests early life nutrition may play a role, but many other factors such as genetics or exposure to some environmental endocrine disruptors may play a part”.
The study findings open up “exciting possibilities for predicting age-related illnesses and finding ways to prevent the onset of these diseases with early intervention,” the authors enthused.
The study was initiated and supported by the European 5th Framework, and the German Research Council provided funding for the INSL3 analysis. The authors declared no conflicts of interest.
Dr. Hicks has disclosed no relevant financial relationships. A version of this article first appeared on MedscapeUK.
FROM FRONTIERS IN ENDOCRINOLOGY
U.K. doctor found guilty of trying to conceal cause of child’s death
The parents of Claire Roberts were told at the time that a viral infection had spread from her stomach to her brain and that medics had done everything possible to save her. But a television documentary, UTV’s “When Hospitals Kill” (broadcast in October 2004), raised concerns about the treatment of a number of children who died from hyponatremia, which occurs when there is a shortage of sodium in the bloodstream.
After the screening, a public inquiry was announced as Alan and Jennifer Roberts sought answers from the Royal Belfast Hospital for Sick Children, Northern Ireland, about the care of their daughter, who died in October 1996.
On Nov. 7, a Medical Practitioners Tribunal Service (MPTS) panel found that Dr. Heather Steen was not aware of the risks of “dilutional hyponatraemia” at the time, but there was “far more awareness” of the issue after the UTV documentary.
Doctor ‘persisted’ with ‘misrepresentation’ of a viral cause to ‘avoid scrutiny’
Tribunal chairman Sean Ell said: “The tribunal accepted that Dr. Steen was not attempting to conceal details of, or failings in, Patient A’s care in 1996/97, when she believed there was a viral cause of death.
“However, once dilutional hyponatraemia as a result of fluid and electrolyte mismanagement became a live issue from 2004, Dr. Steen persisted with her focus on a viral cause and continued to emphasise this aspect whilst seeking to downplay, qualify, and minimise or ignore findings to the contrary.”
He said this “misrepresentation” continued through the consultant pediatrician’s involvement with Claire’s parents, at a coroner’s inquest, which was ordered after the documentary screening, and the public inquiry.
Mr. Ell said: “It was done in order to conceal the true circumstances of Patient A’s death, and in particular, the possible failings in Patient A’s care.
“Dr. Steen had many opportunities to reconsider and be open and transparent, but chose to maintain her dishonesty over the course of events after 2004.
“Whilst the failings may not have changed the tragic outcome of Patient A’s death, her parents were seeking answers to what happened and were entitled to full transparency.”
Dr. Steen denied the allegations but did not give evidence.
The General Medical Council had argued Dr. Steen tried to cover up the circumstances of Claire’s death to “avoid scrutiny.” The MPTS panel, sitting remotely, will next consider whether Dr. Steen’s fitness to practice is impaired by reason of misconduct.
The hyponatremia public inquiry concluded in 2018 that Claire’s death was the result of “negligent care” from an overdose of fluids and medication. A fresh inquest in 2019 ruled her death was “caused by the treatment she received in hospital.”
This article contains information from PA Media. A version of this article appeared on MedscapeUK.
The parents of Claire Roberts were told at the time that a viral infection had spread from her stomach to her brain and that medics had done everything possible to save her. But a television documentary, UTV’s “When Hospitals Kill” (broadcast in October 2004), raised concerns about the treatment of a number of children who died from hyponatremia, which occurs when there is a shortage of sodium in the bloodstream.
After the screening, a public inquiry was announced as Alan and Jennifer Roberts sought answers from the Royal Belfast Hospital for Sick Children, Northern Ireland, about the care of their daughter, who died in October 1996.
On Nov. 7, a Medical Practitioners Tribunal Service (MPTS) panel found that Dr. Heather Steen was not aware of the risks of “dilutional hyponatraemia” at the time, but there was “far more awareness” of the issue after the UTV documentary.
Doctor ‘persisted’ with ‘misrepresentation’ of a viral cause to ‘avoid scrutiny’
Tribunal chairman Sean Ell said: “The tribunal accepted that Dr. Steen was not attempting to conceal details of, or failings in, Patient A’s care in 1996/97, when she believed there was a viral cause of death.
“However, once dilutional hyponatraemia as a result of fluid and electrolyte mismanagement became a live issue from 2004, Dr. Steen persisted with her focus on a viral cause and continued to emphasise this aspect whilst seeking to downplay, qualify, and minimise or ignore findings to the contrary.”
He said this “misrepresentation” continued through the consultant pediatrician’s involvement with Claire’s parents, at a coroner’s inquest, which was ordered after the documentary screening, and the public inquiry.
Mr. Ell said: “It was done in order to conceal the true circumstances of Patient A’s death, and in particular, the possible failings in Patient A’s care.
“Dr. Steen had many opportunities to reconsider and be open and transparent, but chose to maintain her dishonesty over the course of events after 2004.
“Whilst the failings may not have changed the tragic outcome of Patient A’s death, her parents were seeking answers to what happened and were entitled to full transparency.”
Dr. Steen denied the allegations but did not give evidence.
The General Medical Council had argued Dr. Steen tried to cover up the circumstances of Claire’s death to “avoid scrutiny.” The MPTS panel, sitting remotely, will next consider whether Dr. Steen’s fitness to practice is impaired by reason of misconduct.
The hyponatremia public inquiry concluded in 2018 that Claire’s death was the result of “negligent care” from an overdose of fluids and medication. A fresh inquest in 2019 ruled her death was “caused by the treatment she received in hospital.”
This article contains information from PA Media. A version of this article appeared on MedscapeUK.
The parents of Claire Roberts were told at the time that a viral infection had spread from her stomach to her brain and that medics had done everything possible to save her. But a television documentary, UTV’s “When Hospitals Kill” (broadcast in October 2004), raised concerns about the treatment of a number of children who died from hyponatremia, which occurs when there is a shortage of sodium in the bloodstream.
After the screening, a public inquiry was announced as Alan and Jennifer Roberts sought answers from the Royal Belfast Hospital for Sick Children, Northern Ireland, about the care of their daughter, who died in October 1996.
On Nov. 7, a Medical Practitioners Tribunal Service (MPTS) panel found that Dr. Heather Steen was not aware of the risks of “dilutional hyponatraemia” at the time, but there was “far more awareness” of the issue after the UTV documentary.
Doctor ‘persisted’ with ‘misrepresentation’ of a viral cause to ‘avoid scrutiny’
Tribunal chairman Sean Ell said: “The tribunal accepted that Dr. Steen was not attempting to conceal details of, or failings in, Patient A’s care in 1996/97, when she believed there was a viral cause of death.
“However, once dilutional hyponatraemia as a result of fluid and electrolyte mismanagement became a live issue from 2004, Dr. Steen persisted with her focus on a viral cause and continued to emphasise this aspect whilst seeking to downplay, qualify, and minimise or ignore findings to the contrary.”
He said this “misrepresentation” continued through the consultant pediatrician’s involvement with Claire’s parents, at a coroner’s inquest, which was ordered after the documentary screening, and the public inquiry.
Mr. Ell said: “It was done in order to conceal the true circumstances of Patient A’s death, and in particular, the possible failings in Patient A’s care.
“Dr. Steen had many opportunities to reconsider and be open and transparent, but chose to maintain her dishonesty over the course of events after 2004.
“Whilst the failings may not have changed the tragic outcome of Patient A’s death, her parents were seeking answers to what happened and were entitled to full transparency.”
Dr. Steen denied the allegations but did not give evidence.
The General Medical Council had argued Dr. Steen tried to cover up the circumstances of Claire’s death to “avoid scrutiny.” The MPTS panel, sitting remotely, will next consider whether Dr. Steen’s fitness to practice is impaired by reason of misconduct.
The hyponatremia public inquiry concluded in 2018 that Claire’s death was the result of “negligent care” from an overdose of fluids and medication. A fresh inquest in 2019 ruled her death was “caused by the treatment she received in hospital.”
This article contains information from PA Media. A version of this article appeared on MedscapeUK.
Tirzepatide lowers weight across all groups with obesity
SAN DIEGO – Weight loss with tirzepatide was fairly uniform across different body mass index ranges, ages, and number of obesity-related comorbidities in patients with overweight/obesity without type 2 diabetes.
These were the main findings in a session about tirzepatide – the dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) agonist – for obesity, presented at the annual meeting of the Obesity Society.
In May, tirzepatide (Mounjaro), a once-weekly subcutaneous injection, was approved by the Food and Drug Administration for glycemic control in patients with type 2 diabetes based on the SURPASS clinical trials.
Then in June, at the American Diabetes Association 2022 annual meeting, researchers reported “unprecedented” weight loss with tirzepatide in patients without type 2 diabetes, in the phase 3 SURMOUNT-1 clinical trial.
In early October, the FDA granted fast track status (expedited review) to tirzepatide for use as an antiobesity drug.
Now these new analyses from SURMOUNT-1 show that “regardless of BMI, regardless of age, regardless of number of obesity-related complications, there was a clear dose-related weight loss that was pretty consistent across groups,” Session Chair Patrick M. O’Neil, PhD, who was not involved with this research, summarized.
“The absolute levels of these weight losses are higher than we’ve seen thus far with [antiobesity] medications,” added Dr. O’Neil, professor of psychiatry and behavioral sciences and director of the Weight Management Center at the Medical University of South Carolina, Charleston.
“Semaglutide took things up one big notch, and this is up a little notch above that,” he said in an interview.
“I’m a psychologist. It should be remembered that in all cases, the FDA approvals are predicated to using [drugs] as an adjunct to diet and exercise change as well,” he stressed.
“I don’t think people should expect that any medication that is currently available will have a lasting effect when it’s no longer taken,” he continued.
“We don’t expect any of these [antiobesity] medications to be making any permanent physiological changes,” Dr. O’Neil added, but patients could “use this medication to help themselves make some long-lasting behavioral changes, so that when they come off the medication, hopefully they’ll be able to continue these new patterns.
“Clearly the medications are having a significant impact,” he emphasized.
BMI, age, comorbidity subgroups, and overall QoL in SURMOUNT-1
SURMOUNT-1 compared the efficacy and safety of tirzepatide 5, 10, and 15 mg subcutaneous once-weekly to placebo, as an adjunct to a reduced-calorie diet and increased physical activity. The study included 2,539 adults without type 2 diabetes who had obesity (BMI ≥ 30 kg/m2) or overweight (BMI ≥ 27 kg/m2) with at least one obesity-related complication (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease).
Age subgroups
Robert F. Kushner, MD, of Northwestern University, Chicago, noted that “Excessive lean mass loss is a clinical concern in elderly individuals being treated for obesity,” so it’s important to know if weight loss with tirzepatide differs by age.
The researchers performed a post hoc analysis in patients who had dual-energy x-ray absorptiometry (DXA) readings at baseline and week 72 (oral abstract 109).
The three age groups in the current analysis were < 50 years old (99 patients), ≥ 50 to < 65 years old (41 patients), and ≥ 65 years old (20 patients). Overall, 63% of patients were age < 50 years, 31% were age 50 to < 65 years, and 6% were ≥ 65 years.
At 72 weeks, patients taking 5, 10, and 15 mg/week tirzepatide lost 21.5%, 20.8%, and 22% of their initial body weight, respectively.
“Tirzepatide significantly lowered total body mass versus placebo regardless of age subgroups,” and it “consistently lowered fat mass, lean mass, fat-mass-to-lean-mass ratio, and visceral fat mass across age subgroups,” Dr. Kushner reported.
BMI subgroups
Louis J. Aronne, MD, Weill Cornell Medicine, New York, presented findings from a prespecified analysis of BMI subgroups (oral abstract 110).
The four BMI subgroups were:
- ≥ 27 to < 30 kg/m2 (overweight), mean initial weight 178 pounds, mean weight reduction 29-30 pounds
- ≥ 30 to < 35 kg/m2 (class 1 obesity), mean initial weight 198 pounds, mean weight reduction 33-43 pounds
- 35 to < 40 kg/m2 (class 2 obesity), mean initial weight 228 pounds, mean reduction 34-56 pounds
- 40 kg/m2 (class 3 obesity), mean initial weight 280 pounds, mean weight reduction 44-64 pounds
Patients with an initial BMI of ≥ 35 to < 40 kg/m2 who received the 15-mg/week dose of tirzepatide had the greatest weight loss, at 24.5%, which is approximately what is seen with bariatric surgeries such as sleeve gastrectomy (25%).
The proportion of patients reaching ≥ 5% weight reduction was approximately 90% in all weight categories. “These numbers are unprecedented,” said Dr. Aronne.
In addition, overall, 73%-90% of patients receiving the 5- to 15-mg doses of tirzepatide achieved ≥ 10% body weight reduction, and “something we never thought we would see” is that 50%-78% of the patients receiving the drug lost 15% or more of their body weight.
In reply to an audience question, Dr. Aronne said it would take further study to determine who would respond well to tirzepatide.
And in reply to another question about whether it would make sense to treat to a target of a normal BMI, he said: “I think we are getting there.”
Patients in the 27- to 30-kg/m2 BMI category lost about the same amount of weight at a 5-mg dose as at a higher dose, suggesting they should stick to the lower dose, which would likely also have fewer side effects, he noted.
Number of comorbidities
Comorbidities in SURMOUNT-1 included hypertension, dyslipidemia, obstructive sleep apnea, atherosclerotic cardiovascular disease, osteoarthritis, anxiety/depression, polycystic ovary syndrome, nonalcoholic fatty liver disease, and asthma/chronic obstructive pulmonary disease. Of the patients with no comorbidities, 32.6% had prediabetes (oral abstract 111).
Sriram Machineni, MD, University of North Carolina at Chapel Hill, noted that obesity is associated with a significantly increased risk of clustering of at least two obesity-related complications, but little is known about how this affects outcomes.
The patients in SURMOUNT-1 were classified into three groups based on number of comorbidities:
- Zero comorbidities, 37% of patients: baseline mean age of 39, mean duration of obesity of 12 years, 29% men
- One comorbidity, 27% of patients: baseline mean age of 44, mean duration of obesity of 14 years, 31% men
- Two or more comorbidities, 36% of patients: baseline mean age of 52, duration of obesity 17 years, 37% men
Regardless of the number of comorbidities, all doses of tirzepatide resulted in a greater reduction in body weight compared with placebo.
Quality of life
Jiat Ling Poon, MD, an employee of Eli Lilly, presented findings from patient-reported replies to questionnaires including Impact of Weight on Quality of Life–Lite (IWQOL-Lite), which assesses physical and psychosocial health, and the Short Form–36 Health Survey, which assesses physical functioning, bodily pain, vitality, role-emotional, role-physical, general health, social functioning, and mental health (oral abstract 112).
Tirzepatide at all doses resulted in significantly greater improvements in patient-reported outcomes compared with placebo.
Meanwhile, the phase 3 SURMOUNT-2 clinical trial of tirzepatide for weight loss in patients with type 2 diabetes is projected to be completed in April 2023.
The studies were funded by Eli Lilly.
A version of this article first appeared on Medscape.com.
SAN DIEGO – Weight loss with tirzepatide was fairly uniform across different body mass index ranges, ages, and number of obesity-related comorbidities in patients with overweight/obesity without type 2 diabetes.
These were the main findings in a session about tirzepatide – the dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) agonist – for obesity, presented at the annual meeting of the Obesity Society.
In May, tirzepatide (Mounjaro), a once-weekly subcutaneous injection, was approved by the Food and Drug Administration for glycemic control in patients with type 2 diabetes based on the SURPASS clinical trials.
Then in June, at the American Diabetes Association 2022 annual meeting, researchers reported “unprecedented” weight loss with tirzepatide in patients without type 2 diabetes, in the phase 3 SURMOUNT-1 clinical trial.
In early October, the FDA granted fast track status (expedited review) to tirzepatide for use as an antiobesity drug.
Now these new analyses from SURMOUNT-1 show that “regardless of BMI, regardless of age, regardless of number of obesity-related complications, there was a clear dose-related weight loss that was pretty consistent across groups,” Session Chair Patrick M. O’Neil, PhD, who was not involved with this research, summarized.
“The absolute levels of these weight losses are higher than we’ve seen thus far with [antiobesity] medications,” added Dr. O’Neil, professor of psychiatry and behavioral sciences and director of the Weight Management Center at the Medical University of South Carolina, Charleston.
“Semaglutide took things up one big notch, and this is up a little notch above that,” he said in an interview.
“I’m a psychologist. It should be remembered that in all cases, the FDA approvals are predicated to using [drugs] as an adjunct to diet and exercise change as well,” he stressed.
“I don’t think people should expect that any medication that is currently available will have a lasting effect when it’s no longer taken,” he continued.
“We don’t expect any of these [antiobesity] medications to be making any permanent physiological changes,” Dr. O’Neil added, but patients could “use this medication to help themselves make some long-lasting behavioral changes, so that when they come off the medication, hopefully they’ll be able to continue these new patterns.
“Clearly the medications are having a significant impact,” he emphasized.
BMI, age, comorbidity subgroups, and overall QoL in SURMOUNT-1
SURMOUNT-1 compared the efficacy and safety of tirzepatide 5, 10, and 15 mg subcutaneous once-weekly to placebo, as an adjunct to a reduced-calorie diet and increased physical activity. The study included 2,539 adults without type 2 diabetes who had obesity (BMI ≥ 30 kg/m2) or overweight (BMI ≥ 27 kg/m2) with at least one obesity-related complication (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease).
Age subgroups
Robert F. Kushner, MD, of Northwestern University, Chicago, noted that “Excessive lean mass loss is a clinical concern in elderly individuals being treated for obesity,” so it’s important to know if weight loss with tirzepatide differs by age.
The researchers performed a post hoc analysis in patients who had dual-energy x-ray absorptiometry (DXA) readings at baseline and week 72 (oral abstract 109).
The three age groups in the current analysis were < 50 years old (99 patients), ≥ 50 to < 65 years old (41 patients), and ≥ 65 years old (20 patients). Overall, 63% of patients were age < 50 years, 31% were age 50 to < 65 years, and 6% were ≥ 65 years.
At 72 weeks, patients taking 5, 10, and 15 mg/week tirzepatide lost 21.5%, 20.8%, and 22% of their initial body weight, respectively.
“Tirzepatide significantly lowered total body mass versus placebo regardless of age subgroups,” and it “consistently lowered fat mass, lean mass, fat-mass-to-lean-mass ratio, and visceral fat mass across age subgroups,” Dr. Kushner reported.
BMI subgroups
Louis J. Aronne, MD, Weill Cornell Medicine, New York, presented findings from a prespecified analysis of BMI subgroups (oral abstract 110).
The four BMI subgroups were:
- ≥ 27 to < 30 kg/m2 (overweight), mean initial weight 178 pounds, mean weight reduction 29-30 pounds
- ≥ 30 to < 35 kg/m2 (class 1 obesity), mean initial weight 198 pounds, mean weight reduction 33-43 pounds
- 35 to < 40 kg/m2 (class 2 obesity), mean initial weight 228 pounds, mean reduction 34-56 pounds
- 40 kg/m2 (class 3 obesity), mean initial weight 280 pounds, mean weight reduction 44-64 pounds
Patients with an initial BMI of ≥ 35 to < 40 kg/m2 who received the 15-mg/week dose of tirzepatide had the greatest weight loss, at 24.5%, which is approximately what is seen with bariatric surgeries such as sleeve gastrectomy (25%).
The proportion of patients reaching ≥ 5% weight reduction was approximately 90% in all weight categories. “These numbers are unprecedented,” said Dr. Aronne.
In addition, overall, 73%-90% of patients receiving the 5- to 15-mg doses of tirzepatide achieved ≥ 10% body weight reduction, and “something we never thought we would see” is that 50%-78% of the patients receiving the drug lost 15% or more of their body weight.
In reply to an audience question, Dr. Aronne said it would take further study to determine who would respond well to tirzepatide.
And in reply to another question about whether it would make sense to treat to a target of a normal BMI, he said: “I think we are getting there.”
Patients in the 27- to 30-kg/m2 BMI category lost about the same amount of weight at a 5-mg dose as at a higher dose, suggesting they should stick to the lower dose, which would likely also have fewer side effects, he noted.
Number of comorbidities
Comorbidities in SURMOUNT-1 included hypertension, dyslipidemia, obstructive sleep apnea, atherosclerotic cardiovascular disease, osteoarthritis, anxiety/depression, polycystic ovary syndrome, nonalcoholic fatty liver disease, and asthma/chronic obstructive pulmonary disease. Of the patients with no comorbidities, 32.6% had prediabetes (oral abstract 111).
Sriram Machineni, MD, University of North Carolina at Chapel Hill, noted that obesity is associated with a significantly increased risk of clustering of at least two obesity-related complications, but little is known about how this affects outcomes.
The patients in SURMOUNT-1 were classified into three groups based on number of comorbidities:
- Zero comorbidities, 37% of patients: baseline mean age of 39, mean duration of obesity of 12 years, 29% men
- One comorbidity, 27% of patients: baseline mean age of 44, mean duration of obesity of 14 years, 31% men
- Two or more comorbidities, 36% of patients: baseline mean age of 52, duration of obesity 17 years, 37% men
Regardless of the number of comorbidities, all doses of tirzepatide resulted in a greater reduction in body weight compared with placebo.
Quality of life
Jiat Ling Poon, MD, an employee of Eli Lilly, presented findings from patient-reported replies to questionnaires including Impact of Weight on Quality of Life–Lite (IWQOL-Lite), which assesses physical and psychosocial health, and the Short Form–36 Health Survey, which assesses physical functioning, bodily pain, vitality, role-emotional, role-physical, general health, social functioning, and mental health (oral abstract 112).
Tirzepatide at all doses resulted in significantly greater improvements in patient-reported outcomes compared with placebo.
Meanwhile, the phase 3 SURMOUNT-2 clinical trial of tirzepatide for weight loss in patients with type 2 diabetes is projected to be completed in April 2023.
The studies were funded by Eli Lilly.
A version of this article first appeared on Medscape.com.
SAN DIEGO – Weight loss with tirzepatide was fairly uniform across different body mass index ranges, ages, and number of obesity-related comorbidities in patients with overweight/obesity without type 2 diabetes.
These were the main findings in a session about tirzepatide – the dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) agonist – for obesity, presented at the annual meeting of the Obesity Society.
In May, tirzepatide (Mounjaro), a once-weekly subcutaneous injection, was approved by the Food and Drug Administration for glycemic control in patients with type 2 diabetes based on the SURPASS clinical trials.
Then in June, at the American Diabetes Association 2022 annual meeting, researchers reported “unprecedented” weight loss with tirzepatide in patients without type 2 diabetes, in the phase 3 SURMOUNT-1 clinical trial.
In early October, the FDA granted fast track status (expedited review) to tirzepatide for use as an antiobesity drug.
Now these new analyses from SURMOUNT-1 show that “regardless of BMI, regardless of age, regardless of number of obesity-related complications, there was a clear dose-related weight loss that was pretty consistent across groups,” Session Chair Patrick M. O’Neil, PhD, who was not involved with this research, summarized.
“The absolute levels of these weight losses are higher than we’ve seen thus far with [antiobesity] medications,” added Dr. O’Neil, professor of psychiatry and behavioral sciences and director of the Weight Management Center at the Medical University of South Carolina, Charleston.
“Semaglutide took things up one big notch, and this is up a little notch above that,” he said in an interview.
“I’m a psychologist. It should be remembered that in all cases, the FDA approvals are predicated to using [drugs] as an adjunct to diet and exercise change as well,” he stressed.
“I don’t think people should expect that any medication that is currently available will have a lasting effect when it’s no longer taken,” he continued.
“We don’t expect any of these [antiobesity] medications to be making any permanent physiological changes,” Dr. O’Neil added, but patients could “use this medication to help themselves make some long-lasting behavioral changes, so that when they come off the medication, hopefully they’ll be able to continue these new patterns.
“Clearly the medications are having a significant impact,” he emphasized.
BMI, age, comorbidity subgroups, and overall QoL in SURMOUNT-1
SURMOUNT-1 compared the efficacy and safety of tirzepatide 5, 10, and 15 mg subcutaneous once-weekly to placebo, as an adjunct to a reduced-calorie diet and increased physical activity. The study included 2,539 adults without type 2 diabetes who had obesity (BMI ≥ 30 kg/m2) or overweight (BMI ≥ 27 kg/m2) with at least one obesity-related complication (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease).
Age subgroups
Robert F. Kushner, MD, of Northwestern University, Chicago, noted that “Excessive lean mass loss is a clinical concern in elderly individuals being treated for obesity,” so it’s important to know if weight loss with tirzepatide differs by age.
The researchers performed a post hoc analysis in patients who had dual-energy x-ray absorptiometry (DXA) readings at baseline and week 72 (oral abstract 109).
The three age groups in the current analysis were < 50 years old (99 patients), ≥ 50 to < 65 years old (41 patients), and ≥ 65 years old (20 patients). Overall, 63% of patients were age < 50 years, 31% were age 50 to < 65 years, and 6% were ≥ 65 years.
At 72 weeks, patients taking 5, 10, and 15 mg/week tirzepatide lost 21.5%, 20.8%, and 22% of their initial body weight, respectively.
“Tirzepatide significantly lowered total body mass versus placebo regardless of age subgroups,” and it “consistently lowered fat mass, lean mass, fat-mass-to-lean-mass ratio, and visceral fat mass across age subgroups,” Dr. Kushner reported.
BMI subgroups
Louis J. Aronne, MD, Weill Cornell Medicine, New York, presented findings from a prespecified analysis of BMI subgroups (oral abstract 110).
The four BMI subgroups were:
- ≥ 27 to < 30 kg/m2 (overweight), mean initial weight 178 pounds, mean weight reduction 29-30 pounds
- ≥ 30 to < 35 kg/m2 (class 1 obesity), mean initial weight 198 pounds, mean weight reduction 33-43 pounds
- 35 to < 40 kg/m2 (class 2 obesity), mean initial weight 228 pounds, mean reduction 34-56 pounds
- 40 kg/m2 (class 3 obesity), mean initial weight 280 pounds, mean weight reduction 44-64 pounds
Patients with an initial BMI of ≥ 35 to < 40 kg/m2 who received the 15-mg/week dose of tirzepatide had the greatest weight loss, at 24.5%, which is approximately what is seen with bariatric surgeries such as sleeve gastrectomy (25%).
The proportion of patients reaching ≥ 5% weight reduction was approximately 90% in all weight categories. “These numbers are unprecedented,” said Dr. Aronne.
In addition, overall, 73%-90% of patients receiving the 5- to 15-mg doses of tirzepatide achieved ≥ 10% body weight reduction, and “something we never thought we would see” is that 50%-78% of the patients receiving the drug lost 15% or more of their body weight.
In reply to an audience question, Dr. Aronne said it would take further study to determine who would respond well to tirzepatide.
And in reply to another question about whether it would make sense to treat to a target of a normal BMI, he said: “I think we are getting there.”
Patients in the 27- to 30-kg/m2 BMI category lost about the same amount of weight at a 5-mg dose as at a higher dose, suggesting they should stick to the lower dose, which would likely also have fewer side effects, he noted.
Number of comorbidities
Comorbidities in SURMOUNT-1 included hypertension, dyslipidemia, obstructive sleep apnea, atherosclerotic cardiovascular disease, osteoarthritis, anxiety/depression, polycystic ovary syndrome, nonalcoholic fatty liver disease, and asthma/chronic obstructive pulmonary disease. Of the patients with no comorbidities, 32.6% had prediabetes (oral abstract 111).
Sriram Machineni, MD, University of North Carolina at Chapel Hill, noted that obesity is associated with a significantly increased risk of clustering of at least two obesity-related complications, but little is known about how this affects outcomes.
The patients in SURMOUNT-1 were classified into three groups based on number of comorbidities:
- Zero comorbidities, 37% of patients: baseline mean age of 39, mean duration of obesity of 12 years, 29% men
- One comorbidity, 27% of patients: baseline mean age of 44, mean duration of obesity of 14 years, 31% men
- Two or more comorbidities, 36% of patients: baseline mean age of 52, duration of obesity 17 years, 37% men
Regardless of the number of comorbidities, all doses of tirzepatide resulted in a greater reduction in body weight compared with placebo.
Quality of life
Jiat Ling Poon, MD, an employee of Eli Lilly, presented findings from patient-reported replies to questionnaires including Impact of Weight on Quality of Life–Lite (IWQOL-Lite), which assesses physical and psychosocial health, and the Short Form–36 Health Survey, which assesses physical functioning, bodily pain, vitality, role-emotional, role-physical, general health, social functioning, and mental health (oral abstract 112).
Tirzepatide at all doses resulted in significantly greater improvements in patient-reported outcomes compared with placebo.
Meanwhile, the phase 3 SURMOUNT-2 clinical trial of tirzepatide for weight loss in patients with type 2 diabetes is projected to be completed in April 2023.
The studies were funded by Eli Lilly.
A version of this article first appeared on Medscape.com.
AT OBESITYWEEK® 2022
Pink shoulder lesion
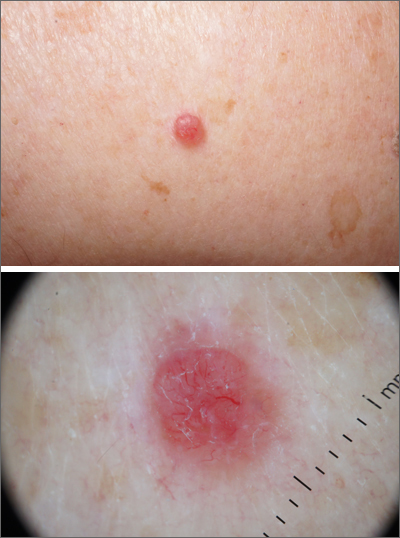
A scoop shave biopsy was performed and histology was consistent with a nodular basal cell carcinoma. BCC is the most common skin cancer in the United States, occurring in approximately 30% of patients with skin types I and II.1 In patients who are Black, squamous cell carcinoma is more common than BCC.2 The overall incidence of BCC is increasing by 4% to 8% every year in the United States.1
BCC most often affects sun-damaged areas—especially on the head and neck—and frequently causes significant tissue damage. It is, however, associated with a low risk of metastasis and mortality.
BCCs may appear as a pink, brown, blue, or white papule or macule. The surface is frequently shiny or pearly in appearance with a rolled border. Dilated, angulated, tree-branch like vessels termed “arborizing vessels” are common. Infiltrative BCC subtypes may look like melted candlewax and extend beyond the area that is clinically apparent.
Partial shave biopsies of a lesion can confirm the diagnosis. A punch biopsy can make it easier to evaluate flat (or even sunken) lesions.
The patient described here was treated with electrodessication and curettage (EDC)—a fast, economical, and effective treatment for the low-risk subtypes of superficial or nodular BCCs on the trunk or extremities. EDC should be avoided with higher risk subtypes of micronodular and infiltrative BCC. With these subtypes, excision (with 4- to 6-mm margins) or Mohs microsurgery is recommended.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME. References
1. Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24. doi:10.1016/j.hoc.2018.09.004
2. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.

A scoop shave biopsy was performed and histology was consistent with a nodular basal cell carcinoma. BCC is the most common skin cancer in the United States, occurring in approximately 30% of patients with skin types I and II.1 In patients who are Black, squamous cell carcinoma is more common than BCC.2 The overall incidence of BCC is increasing by 4% to 8% every year in the United States.1
BCC most often affects sun-damaged areas—especially on the head and neck—and frequently causes significant tissue damage. It is, however, associated with a low risk of metastasis and mortality.
BCCs may appear as a pink, brown, blue, or white papule or macule. The surface is frequently shiny or pearly in appearance with a rolled border. Dilated, angulated, tree-branch like vessels termed “arborizing vessels” are common. Infiltrative BCC subtypes may look like melted candlewax and extend beyond the area that is clinically apparent.
Partial shave biopsies of a lesion can confirm the diagnosis. A punch biopsy can make it easier to evaluate flat (or even sunken) lesions.
The patient described here was treated with electrodessication and curettage (EDC)—a fast, economical, and effective treatment for the low-risk subtypes of superficial or nodular BCCs on the trunk or extremities. EDC should be avoided with higher risk subtypes of micronodular and infiltrative BCC. With these subtypes, excision (with 4- to 6-mm margins) or Mohs microsurgery is recommended.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME. References

A scoop shave biopsy was performed and histology was consistent with a nodular basal cell carcinoma. BCC is the most common skin cancer in the United States, occurring in approximately 30% of patients with skin types I and II.1 In patients who are Black, squamous cell carcinoma is more common than BCC.2 The overall incidence of BCC is increasing by 4% to 8% every year in the United States.1
BCC most often affects sun-damaged areas—especially on the head and neck—and frequently causes significant tissue damage. It is, however, associated with a low risk of metastasis and mortality.
BCCs may appear as a pink, brown, blue, or white papule or macule. The surface is frequently shiny or pearly in appearance with a rolled border. Dilated, angulated, tree-branch like vessels termed “arborizing vessels” are common. Infiltrative BCC subtypes may look like melted candlewax and extend beyond the area that is clinically apparent.
Partial shave biopsies of a lesion can confirm the diagnosis. A punch biopsy can make it easier to evaluate flat (or even sunken) lesions.
The patient described here was treated with electrodessication and curettage (EDC)—a fast, economical, and effective treatment for the low-risk subtypes of superficial or nodular BCCs on the trunk or extremities. EDC should be avoided with higher risk subtypes of micronodular and infiltrative BCC. With these subtypes, excision (with 4- to 6-mm margins) or Mohs microsurgery is recommended.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME. References
1. Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24. doi:10.1016/j.hoc.2018.09.004
2. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.
1. Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24. doi:10.1016/j.hoc.2018.09.004
2. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.