User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Coffee intake may be driven by cardiovascular symptoms
An examination of coffee consumption habits of almost 400,000 people suggests that those habits are largely driven by a person’s cardiovascular health.
Data from a large population database showed that people with essential hypertension, angina, or cardiac arrhythmias drank less coffee than people who had none of these conditions. When they did drink coffee, it tended to be decaffeinated.
The investigators, led by Elina Hyppönen, PhD, director of the Australian Centre for Precision Health at the University of South Australia, Adelaide, say that this predilection for avoiding coffee, which is known to produce jitteriness and heart palpitations, is based on genetics.
“If your body is telling you not to drink that extra cup of coffee, there’s likely a reason why,” Dr. Hyppönen said in an interview.
The study was published online in the American Journal of Clinical Nutrition.
“People drink coffee as a pick-me-up when they’re feeling tired, or because it tastes good, or simply because it’s part of their daily routine, but what we don’t recognize is that people subconsciously self-regulate safe levels of caffeine based on how high their blood pressure is, and this is likely a result of a protective genetic mechanism, [meaning] that someone who drinks a lot of coffee is likely more genetically tolerant of caffeine, as compared to someone who drinks very little,” Dr. Hyppönen said.
“In addition, we’ve known from past research that when people feel unwell, they tend to drink less coffee. This type of phenomenon, where disease drives behavior, is called reverse causality,” Dr. Hyppönen said.
For this analysis, she and her team used information on 390,435 individuals of European ancestry from the UK Biobank, a large epidemiologic database. Habitual coffee consumption was self-reported, and systolic and diastolic blood pressure and heart rate were measured at baseline. Cardiovascular symptoms at baseline were gleaned from hospital diagnoses, primary care records, and/or self report, the authors note.
To look at the relationship of systolic BP, diastolic BP, and heart rate with coffee consumption, they used a strategy called Mendelian randomization, which allows genetic information such as variants reflecting higher blood pressures and heart rate to be used to provide evidence for a causal association.
Results showed that participants with essential hypertension, angina, or arrhythmia were “all more likely to drink less caffeinated coffee and to be nonhabitual or decaffeinated coffee drinkers compared with those who did not report related symptoms,” the authors write.
Those with higher systolic and diastolic BP based on their genetics tended to drink less caffeinated coffee at baseline, “with consistent genetic evidence to support a causal explanation across all methods,” they noted.
They also found that those people who have a higher resting heart rate due to their genes were more likely to choose decaffeinated coffee.
“These results have two major implications,” Dr. Hyppönen said. “Firstly, they show that our bodies can regulate behavior in ways that we may not realize, and that if something does not feel good to us, there is a likely to be a reason why.”
“Second, our results show that our health status in part regulates the amount of coffee we drink. This is important, because when disease drives behavior, it can lead to misleading health associations in observational studies, and indeed, create a false impression for health benefits if the group of people who do not drink coffee also includes more people who are unwell,” she said.
For now, doctors can tell their patients that this study provides an explanation as to why research on the health effects of habitual coffee consumption has been conflicting, Dr. Hyppönen said.
“Our study also highlights the uncertainty that underlies the claimed health benefits of coffee, but at the same time, it gives a positive message about the ability of our body to regulate our level of coffee consumption in a way that helps us avoid adverse effects.”
“The most common symptoms of excessive coffee consumption are palpitations and rapid heartbeat, also known as tachycardia,” Nieca Goldberg, MD, medical director of the NYU Women’s Heart Program at NYU Langone Health, said in an interview.
“This study was designed to see if cardiac symptoms affect coffee consumption, and it showed that people with hypertension, angina, history of arrhythmias, and poor health tend to be decaffeinated coffee drinkers or no coffee drinkers,” Dr. Goldberg said.
“People naturally alter their coffee intake base on their blood pressure and symptoms of palpitations and/or rapid heart rate,” she said.
The results also suggest that, “we cannot infer health benefit or harm based on the available coffee studies,” Dr. Goldberg added.
The study was funded by the National Health and Medical Research Council, Australia. Dr. Hyppönen and Dr. Goldberg have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An examination of coffee consumption habits of almost 400,000 people suggests that those habits are largely driven by a person’s cardiovascular health.
Data from a large population database showed that people with essential hypertension, angina, or cardiac arrhythmias drank less coffee than people who had none of these conditions. When they did drink coffee, it tended to be decaffeinated.
The investigators, led by Elina Hyppönen, PhD, director of the Australian Centre for Precision Health at the University of South Australia, Adelaide, say that this predilection for avoiding coffee, which is known to produce jitteriness and heart palpitations, is based on genetics.
“If your body is telling you not to drink that extra cup of coffee, there’s likely a reason why,” Dr. Hyppönen said in an interview.
The study was published online in the American Journal of Clinical Nutrition.
“People drink coffee as a pick-me-up when they’re feeling tired, or because it tastes good, or simply because it’s part of their daily routine, but what we don’t recognize is that people subconsciously self-regulate safe levels of caffeine based on how high their blood pressure is, and this is likely a result of a protective genetic mechanism, [meaning] that someone who drinks a lot of coffee is likely more genetically tolerant of caffeine, as compared to someone who drinks very little,” Dr. Hyppönen said.
“In addition, we’ve known from past research that when people feel unwell, they tend to drink less coffee. This type of phenomenon, where disease drives behavior, is called reverse causality,” Dr. Hyppönen said.
For this analysis, she and her team used information on 390,435 individuals of European ancestry from the UK Biobank, a large epidemiologic database. Habitual coffee consumption was self-reported, and systolic and diastolic blood pressure and heart rate were measured at baseline. Cardiovascular symptoms at baseline were gleaned from hospital diagnoses, primary care records, and/or self report, the authors note.
To look at the relationship of systolic BP, diastolic BP, and heart rate with coffee consumption, they used a strategy called Mendelian randomization, which allows genetic information such as variants reflecting higher blood pressures and heart rate to be used to provide evidence for a causal association.
Results showed that participants with essential hypertension, angina, or arrhythmia were “all more likely to drink less caffeinated coffee and to be nonhabitual or decaffeinated coffee drinkers compared with those who did not report related symptoms,” the authors write.
Those with higher systolic and diastolic BP based on their genetics tended to drink less caffeinated coffee at baseline, “with consistent genetic evidence to support a causal explanation across all methods,” they noted.
They also found that those people who have a higher resting heart rate due to their genes were more likely to choose decaffeinated coffee.
“These results have two major implications,” Dr. Hyppönen said. “Firstly, they show that our bodies can regulate behavior in ways that we may not realize, and that if something does not feel good to us, there is a likely to be a reason why.”
“Second, our results show that our health status in part regulates the amount of coffee we drink. This is important, because when disease drives behavior, it can lead to misleading health associations in observational studies, and indeed, create a false impression for health benefits if the group of people who do not drink coffee also includes more people who are unwell,” she said.
For now, doctors can tell their patients that this study provides an explanation as to why research on the health effects of habitual coffee consumption has been conflicting, Dr. Hyppönen said.
“Our study also highlights the uncertainty that underlies the claimed health benefits of coffee, but at the same time, it gives a positive message about the ability of our body to regulate our level of coffee consumption in a way that helps us avoid adverse effects.”
“The most common symptoms of excessive coffee consumption are palpitations and rapid heartbeat, also known as tachycardia,” Nieca Goldberg, MD, medical director of the NYU Women’s Heart Program at NYU Langone Health, said in an interview.
“This study was designed to see if cardiac symptoms affect coffee consumption, and it showed that people with hypertension, angina, history of arrhythmias, and poor health tend to be decaffeinated coffee drinkers or no coffee drinkers,” Dr. Goldberg said.
“People naturally alter their coffee intake base on their blood pressure and symptoms of palpitations and/or rapid heart rate,” she said.
The results also suggest that, “we cannot infer health benefit or harm based on the available coffee studies,” Dr. Goldberg added.
The study was funded by the National Health and Medical Research Council, Australia. Dr. Hyppönen and Dr. Goldberg have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An examination of coffee consumption habits of almost 400,000 people suggests that those habits are largely driven by a person’s cardiovascular health.
Data from a large population database showed that people with essential hypertension, angina, or cardiac arrhythmias drank less coffee than people who had none of these conditions. When they did drink coffee, it tended to be decaffeinated.
The investigators, led by Elina Hyppönen, PhD, director of the Australian Centre for Precision Health at the University of South Australia, Adelaide, say that this predilection for avoiding coffee, which is known to produce jitteriness and heart palpitations, is based on genetics.
“If your body is telling you not to drink that extra cup of coffee, there’s likely a reason why,” Dr. Hyppönen said in an interview.
The study was published online in the American Journal of Clinical Nutrition.
“People drink coffee as a pick-me-up when they’re feeling tired, or because it tastes good, or simply because it’s part of their daily routine, but what we don’t recognize is that people subconsciously self-regulate safe levels of caffeine based on how high their blood pressure is, and this is likely a result of a protective genetic mechanism, [meaning] that someone who drinks a lot of coffee is likely more genetically tolerant of caffeine, as compared to someone who drinks very little,” Dr. Hyppönen said.
“In addition, we’ve known from past research that when people feel unwell, they tend to drink less coffee. This type of phenomenon, where disease drives behavior, is called reverse causality,” Dr. Hyppönen said.
For this analysis, she and her team used information on 390,435 individuals of European ancestry from the UK Biobank, a large epidemiologic database. Habitual coffee consumption was self-reported, and systolic and diastolic blood pressure and heart rate were measured at baseline. Cardiovascular symptoms at baseline were gleaned from hospital diagnoses, primary care records, and/or self report, the authors note.
To look at the relationship of systolic BP, diastolic BP, and heart rate with coffee consumption, they used a strategy called Mendelian randomization, which allows genetic information such as variants reflecting higher blood pressures and heart rate to be used to provide evidence for a causal association.
Results showed that participants with essential hypertension, angina, or arrhythmia were “all more likely to drink less caffeinated coffee and to be nonhabitual or decaffeinated coffee drinkers compared with those who did not report related symptoms,” the authors write.
Those with higher systolic and diastolic BP based on their genetics tended to drink less caffeinated coffee at baseline, “with consistent genetic evidence to support a causal explanation across all methods,” they noted.
They also found that those people who have a higher resting heart rate due to their genes were more likely to choose decaffeinated coffee.
“These results have two major implications,” Dr. Hyppönen said. “Firstly, they show that our bodies can regulate behavior in ways that we may not realize, and that if something does not feel good to us, there is a likely to be a reason why.”
“Second, our results show that our health status in part regulates the amount of coffee we drink. This is important, because when disease drives behavior, it can lead to misleading health associations in observational studies, and indeed, create a false impression for health benefits if the group of people who do not drink coffee also includes more people who are unwell,” she said.
For now, doctors can tell their patients that this study provides an explanation as to why research on the health effects of habitual coffee consumption has been conflicting, Dr. Hyppönen said.
“Our study also highlights the uncertainty that underlies the claimed health benefits of coffee, but at the same time, it gives a positive message about the ability of our body to regulate our level of coffee consumption in a way that helps us avoid adverse effects.”
“The most common symptoms of excessive coffee consumption are palpitations and rapid heartbeat, also known as tachycardia,” Nieca Goldberg, MD, medical director of the NYU Women’s Heart Program at NYU Langone Health, said in an interview.
“This study was designed to see if cardiac symptoms affect coffee consumption, and it showed that people with hypertension, angina, history of arrhythmias, and poor health tend to be decaffeinated coffee drinkers or no coffee drinkers,” Dr. Goldberg said.
“People naturally alter their coffee intake base on their blood pressure and symptoms of palpitations and/or rapid heart rate,” she said.
The results also suggest that, “we cannot infer health benefit or harm based on the available coffee studies,” Dr. Goldberg added.
The study was funded by the National Health and Medical Research Council, Australia. Dr. Hyppönen and Dr. Goldberg have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Reassuring data on impact of mild COVID-19 on the heart
Six months after mild SARS-CoV-2 infection in a representative health care workforce, no long-term cardiovascular sequelae were detected, compared with a matched SARS-CoV-2 seronegative group.
“Mild COVID-19 left no measurable cardiovascular impact on LV structure, function, scar burden, aortic stiffness, or serum biomarkers,” the researchers reported in an article published online May 8 in JACC: Cardiovascular Imaging.
“We provide societal reassurance and support for the position that screening in asymptomatic individuals following mild disease is not indicated,” first author George Joy, MBBS, University College London, said in presenting the results at EuroCMR, the annual CMR congress of the European Association of Cardiovascular Imaging (EACVI).
Briefing comoderator Leyla Elif Sade, MD, University of Baskent, Ankara, Turkey, said, “This is the hot topic of our time because of obvious reasons and I think [this] study is quite important to avoid unnecessary further testing, surveillance testing, and to avoid a significant burden of health care costs.”
‘Alarming’ early data
Early cardiac magnetic resonance (CMR) studies in patients recovered from mild COVID-19 were “alarming,” Dr. Joy said.
As previously reported, one study showed cardiac abnormalities after mild COVID-19 in up to 78% of patients, with evidence of ongoing myocardial inflammation in 60%. The CMR findings correlated with elevations in troponin T by high-sensitivity assay (hs-TnT).
To investigate further, Dr. Joy and colleagues did a nested case-control study within the COVIDsortium, a prospective study of 731 health care workers from three London hospitals who underwent weekly symptom, polymerase chain reaction, and serology assessment over 4 months during the first wave of the pandemic.
A total of 157 (21.5%) participants seroconverted during the study period.
Six months after infection, 74 seropositive (median age, 39; 62% women) and 75 age-, sex-, and ethnicity-matched seronegative controls underwent cardiovascular phenotyping (comprehensive phantom-calibrated CMR and blood biomarkers). The analysis was blinded, using objective artificial intelligence analytics when available.
The results showed no statistically significant differences between seropositive and seronegative participants in cardiac structure (left ventricular volumes, mass, atrial area), function (ejection fraction, global longitudinal shortening, aortic distensibility), tissue characterization (T1, T2, extracellular volume fraction mapping, late gadolinium enhancement) or biomarkers (troponin, N-terminal pro–B-type natriuretic peptide).
Cardiovascular abnormalities were no more common in seropositive than seronegative otherwise healthy health care workers 6 months post mild SARS-CoV-2 infection. Measured abnormalities were “evenly distributed between both groups,” Dr. Joy said.
Therefore, it’s “important to reassure patients with mild SARS-CoV-2 infection regarding its cardiovascular effects,” Dr. Joy and colleagues concluded.
Limitations and caveats
They caution, however, that the study provides insight only into the short- to medium-term sequelae of patients aged 18-69 with mild COVID-19 who did not require hospitalization and had low numbers of comorbidities.
The study does not address the cardiovascular effects after severe COVID-19 infection requiring hospitalization or in those with multiple comorbid conditions, they noted. It also does not prove that apparently mild SARS-CoV-2 never causes chronic myocarditis.
“The study design would not distinguish between people who had sustained completely healed myocarditis and pericarditis and those in whom the heart had never been affected,” the researchers noted.
They pointed to a recent cross-sectional study of athletes 1-month post mild COVID-19 that found significant pericardial involvement (late enhancement and/or pericardial effusion), although no baseline pre-COVID-19 imaging was performed. In the current study at 6 months post infection the pericardium was normal.
The coauthors of a linked editorial say this study provides “welcome, reassuring information that in healthy individuals who experience mild infection with COVID-19, persisting evidence of cardiovascular complications is very uncommon. The results do not support cardiovascular screening in individuals with mild or asymptomatic infection with COVID-19.”
Colin Berry, PhD, and Kenneth Mangion, PhD, both from University of Glasgow, cautioned that the population is restricted to health care workers; therefore, the findings may not necessarily be generalized to a community population .
“Healthcare workers do not reflect the population of individuals most clinically affected by COVID-19 illness. The severity of acute COVID-19 infection is greatest in older individuals and those with preexisting health problems. Healthcare workers are not representative of the wider, unselected, at-risk, community population,” they pointed out.
Cardiovascular risk factors and concomitant health problems (heart and respiratory disease) may be more common in the community than in health care workers, and prior studies have highlighted their potential impact for disease pathogenesis in COVID-19.
Dr. Berry and Dr. Mangion also noted that women made up nearly two-thirds of the seropositive group. This may reflect a selection bias or may naturally reflect the fact that proportionately more women are asymptomatic or have milder forms of illness, whereas severe SARS-CoV-2 infection requiring hospitalization affects men to a greater degree.
COVIDsortium funding was donated by individuals, charitable trusts, and corporations including Goldman Sachs, Citadel and Citadel Securities, The Guy Foundation, GW Pharmaceuticals, Kusuma Trust, and Jagclif Charitable Trust, and enabled by Barts Charity with support from UCLH Charity. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Six months after mild SARS-CoV-2 infection in a representative health care workforce, no long-term cardiovascular sequelae were detected, compared with a matched SARS-CoV-2 seronegative group.
“Mild COVID-19 left no measurable cardiovascular impact on LV structure, function, scar burden, aortic stiffness, or serum biomarkers,” the researchers reported in an article published online May 8 in JACC: Cardiovascular Imaging.
“We provide societal reassurance and support for the position that screening in asymptomatic individuals following mild disease is not indicated,” first author George Joy, MBBS, University College London, said in presenting the results at EuroCMR, the annual CMR congress of the European Association of Cardiovascular Imaging (EACVI).
Briefing comoderator Leyla Elif Sade, MD, University of Baskent, Ankara, Turkey, said, “This is the hot topic of our time because of obvious reasons and I think [this] study is quite important to avoid unnecessary further testing, surveillance testing, and to avoid a significant burden of health care costs.”
‘Alarming’ early data
Early cardiac magnetic resonance (CMR) studies in patients recovered from mild COVID-19 were “alarming,” Dr. Joy said.
As previously reported, one study showed cardiac abnormalities after mild COVID-19 in up to 78% of patients, with evidence of ongoing myocardial inflammation in 60%. The CMR findings correlated with elevations in troponin T by high-sensitivity assay (hs-TnT).
To investigate further, Dr. Joy and colleagues did a nested case-control study within the COVIDsortium, a prospective study of 731 health care workers from three London hospitals who underwent weekly symptom, polymerase chain reaction, and serology assessment over 4 months during the first wave of the pandemic.
A total of 157 (21.5%) participants seroconverted during the study period.
Six months after infection, 74 seropositive (median age, 39; 62% women) and 75 age-, sex-, and ethnicity-matched seronegative controls underwent cardiovascular phenotyping (comprehensive phantom-calibrated CMR and blood biomarkers). The analysis was blinded, using objective artificial intelligence analytics when available.
The results showed no statistically significant differences between seropositive and seronegative participants in cardiac structure (left ventricular volumes, mass, atrial area), function (ejection fraction, global longitudinal shortening, aortic distensibility), tissue characterization (T1, T2, extracellular volume fraction mapping, late gadolinium enhancement) or biomarkers (troponin, N-terminal pro–B-type natriuretic peptide).
Cardiovascular abnormalities were no more common in seropositive than seronegative otherwise healthy health care workers 6 months post mild SARS-CoV-2 infection. Measured abnormalities were “evenly distributed between both groups,” Dr. Joy said.
Therefore, it’s “important to reassure patients with mild SARS-CoV-2 infection regarding its cardiovascular effects,” Dr. Joy and colleagues concluded.
Limitations and caveats
They caution, however, that the study provides insight only into the short- to medium-term sequelae of patients aged 18-69 with mild COVID-19 who did not require hospitalization and had low numbers of comorbidities.
The study does not address the cardiovascular effects after severe COVID-19 infection requiring hospitalization or in those with multiple comorbid conditions, they noted. It also does not prove that apparently mild SARS-CoV-2 never causes chronic myocarditis.
“The study design would not distinguish between people who had sustained completely healed myocarditis and pericarditis and those in whom the heart had never been affected,” the researchers noted.
They pointed to a recent cross-sectional study of athletes 1-month post mild COVID-19 that found significant pericardial involvement (late enhancement and/or pericardial effusion), although no baseline pre-COVID-19 imaging was performed. In the current study at 6 months post infection the pericardium was normal.
The coauthors of a linked editorial say this study provides “welcome, reassuring information that in healthy individuals who experience mild infection with COVID-19, persisting evidence of cardiovascular complications is very uncommon. The results do not support cardiovascular screening in individuals with mild or asymptomatic infection with COVID-19.”
Colin Berry, PhD, and Kenneth Mangion, PhD, both from University of Glasgow, cautioned that the population is restricted to health care workers; therefore, the findings may not necessarily be generalized to a community population .
“Healthcare workers do not reflect the population of individuals most clinically affected by COVID-19 illness. The severity of acute COVID-19 infection is greatest in older individuals and those with preexisting health problems. Healthcare workers are not representative of the wider, unselected, at-risk, community population,” they pointed out.
Cardiovascular risk factors and concomitant health problems (heart and respiratory disease) may be more common in the community than in health care workers, and prior studies have highlighted their potential impact for disease pathogenesis in COVID-19.
Dr. Berry and Dr. Mangion also noted that women made up nearly two-thirds of the seropositive group. This may reflect a selection bias or may naturally reflect the fact that proportionately more women are asymptomatic or have milder forms of illness, whereas severe SARS-CoV-2 infection requiring hospitalization affects men to a greater degree.
COVIDsortium funding was donated by individuals, charitable trusts, and corporations including Goldman Sachs, Citadel and Citadel Securities, The Guy Foundation, GW Pharmaceuticals, Kusuma Trust, and Jagclif Charitable Trust, and enabled by Barts Charity with support from UCLH Charity. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Six months after mild SARS-CoV-2 infection in a representative health care workforce, no long-term cardiovascular sequelae were detected, compared with a matched SARS-CoV-2 seronegative group.
“Mild COVID-19 left no measurable cardiovascular impact on LV structure, function, scar burden, aortic stiffness, or serum biomarkers,” the researchers reported in an article published online May 8 in JACC: Cardiovascular Imaging.
“We provide societal reassurance and support for the position that screening in asymptomatic individuals following mild disease is not indicated,” first author George Joy, MBBS, University College London, said in presenting the results at EuroCMR, the annual CMR congress of the European Association of Cardiovascular Imaging (EACVI).
Briefing comoderator Leyla Elif Sade, MD, University of Baskent, Ankara, Turkey, said, “This is the hot topic of our time because of obvious reasons and I think [this] study is quite important to avoid unnecessary further testing, surveillance testing, and to avoid a significant burden of health care costs.”
‘Alarming’ early data
Early cardiac magnetic resonance (CMR) studies in patients recovered from mild COVID-19 were “alarming,” Dr. Joy said.
As previously reported, one study showed cardiac abnormalities after mild COVID-19 in up to 78% of patients, with evidence of ongoing myocardial inflammation in 60%. The CMR findings correlated with elevations in troponin T by high-sensitivity assay (hs-TnT).
To investigate further, Dr. Joy and colleagues did a nested case-control study within the COVIDsortium, a prospective study of 731 health care workers from three London hospitals who underwent weekly symptom, polymerase chain reaction, and serology assessment over 4 months during the first wave of the pandemic.
A total of 157 (21.5%) participants seroconverted during the study period.
Six months after infection, 74 seropositive (median age, 39; 62% women) and 75 age-, sex-, and ethnicity-matched seronegative controls underwent cardiovascular phenotyping (comprehensive phantom-calibrated CMR and blood biomarkers). The analysis was blinded, using objective artificial intelligence analytics when available.
The results showed no statistically significant differences between seropositive and seronegative participants in cardiac structure (left ventricular volumes, mass, atrial area), function (ejection fraction, global longitudinal shortening, aortic distensibility), tissue characterization (T1, T2, extracellular volume fraction mapping, late gadolinium enhancement) or biomarkers (troponin, N-terminal pro–B-type natriuretic peptide).
Cardiovascular abnormalities were no more common in seropositive than seronegative otherwise healthy health care workers 6 months post mild SARS-CoV-2 infection. Measured abnormalities were “evenly distributed between both groups,” Dr. Joy said.
Therefore, it’s “important to reassure patients with mild SARS-CoV-2 infection regarding its cardiovascular effects,” Dr. Joy and colleagues concluded.
Limitations and caveats
They caution, however, that the study provides insight only into the short- to medium-term sequelae of patients aged 18-69 with mild COVID-19 who did not require hospitalization and had low numbers of comorbidities.
The study does not address the cardiovascular effects after severe COVID-19 infection requiring hospitalization or in those with multiple comorbid conditions, they noted. It also does not prove that apparently mild SARS-CoV-2 never causes chronic myocarditis.
“The study design would not distinguish between people who had sustained completely healed myocarditis and pericarditis and those in whom the heart had never been affected,” the researchers noted.
They pointed to a recent cross-sectional study of athletes 1-month post mild COVID-19 that found significant pericardial involvement (late enhancement and/or pericardial effusion), although no baseline pre-COVID-19 imaging was performed. In the current study at 6 months post infection the pericardium was normal.
The coauthors of a linked editorial say this study provides “welcome, reassuring information that in healthy individuals who experience mild infection with COVID-19, persisting evidence of cardiovascular complications is very uncommon. The results do not support cardiovascular screening in individuals with mild or asymptomatic infection with COVID-19.”
Colin Berry, PhD, and Kenneth Mangion, PhD, both from University of Glasgow, cautioned that the population is restricted to health care workers; therefore, the findings may not necessarily be generalized to a community population .
“Healthcare workers do not reflect the population of individuals most clinically affected by COVID-19 illness. The severity of acute COVID-19 infection is greatest in older individuals and those with preexisting health problems. Healthcare workers are not representative of the wider, unselected, at-risk, community population,” they pointed out.
Cardiovascular risk factors and concomitant health problems (heart and respiratory disease) may be more common in the community than in health care workers, and prior studies have highlighted their potential impact for disease pathogenesis in COVID-19.
Dr. Berry and Dr. Mangion also noted that women made up nearly two-thirds of the seropositive group. This may reflect a selection bias or may naturally reflect the fact that proportionately more women are asymptomatic or have milder forms of illness, whereas severe SARS-CoV-2 infection requiring hospitalization affects men to a greater degree.
COVIDsortium funding was donated by individuals, charitable trusts, and corporations including Goldman Sachs, Citadel and Citadel Securities, The Guy Foundation, GW Pharmaceuticals, Kusuma Trust, and Jagclif Charitable Trust, and enabled by Barts Charity with support from UCLH Charity. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
What to know about COVID-19 vaccines and skin reactions
The good news is that these side effects tend to be minor and vanish within a few days, Esther Freeman, MD, PhD, said in a presentation at the American Academy of Dermatology Virtual Meeting Experience.
“The reality is actually very reassuring,” Dr. Freeman said, especially in light of what is currently known about when the rashes occur and how anaphylaxis is extremely uncommon. Now, she added, dermatologists can tell patients who had reactions to their initial vaccination that “we know you had this big reaction, and we know that it was upsetting and uncomfortable. But it may not happen the second time around. And if it does, [the reaction is] probably going to be smaller.”
Dr. Freeman, associate professor of dermatology at Harvard Medical School, Boston, highlighted a study published in the Journal of the American Academy of Dermatology that she coauthored with dermatologists across the United States. The researchers tracked 414 cutaneous reactions to the Moderna (83%) and Pfizer (17%) COVID-19 vaccines in a group of patients, which was 90% female, 78% White, and mostly from the United States. Their average age was 44 years. The cases were reported to the AAD–International League of Dermatological Societies registry of COVID-19 cutaneous manifestations.
While most were women, “it’s a little hard to know if this is really going to end up being a true finding,” said Dr. Freeman, the registry’s principal investigator and a member of the AAD’s COVID-19 Ad Hoc Task Force. “If you think about who got vaccinated early, it was health care providers, and the American health care workforce is over 70% female. So I think there’s a little bit of bias here. There may also be a bias because women may be slightly more likely to report or go to their health care provider for a rash.”
Delayed large local reactions were the most common, accounting for 66% (175 cases) of the 267 skin reactions reported after the first Moderna vaccine dose and 30% (31 cases) of the 102 reactions reported after the second dose. These reactions represented 15% (5 cases) of the 34 skin reactions reported after the first Pfizer vaccine dose and 18% (7 cases) of the 40 reactions after the second dose.
There are two peaks with that first dose, Dr. Freeman said. “There’s a peak around day 2 or 3. And there’s another peak around day 7 or 8 with some of these reactions. Only 27% who had a reaction with the first dose had the same reaction with the second.” She added that these reactions “are not cellulitis and don’t require antibiotics.”
Other more common reactions included local injection-site reactions (swelling, erythema, and pain), urticaria (after 24 hours in almost all cases, occurring at a higher rate in patients who received the Pfizer vaccine), and morbilliform eruptions.
Dr. Freeman said that patients may experience redness and swelling in the hands and feet that can be “very uncomfortable.” She described one patient “who was having a hard time actually closing his fist, just because of the amount of swelling and redness in his hand. It did resolve, and it’s important to reassure your patients it will go away.”
According to this study, less common reports of other cutaneous findings with both vaccines included 9 reports of swelling at the site of cosmetic fillers, 8 reports of pernio/chilblains, 10 reports of varicella zoster, 4 reports of herpes simplex flares, 4 pityriasis rosea–like reactions, and 4 rashes in infants of vaccinated breastfeeding mothers.
The study noted that “patients responded well to topical corticosteroids, oral antihistamines, and/or pain-relieving medications. These reactions resolved after a median of 3-4 days.”
It’s important to understand that none of the patients developed anaphylaxis after the second dose even if they’d had a reaction to the first dose, Dr. Freeman said. “But I should point out that we’re talking about reactions that have started more than 4 hours after the vaccine. If a rash such as a urticaria specifically starts within 4 hours of vaccination, that’s in a different category. Those are considered more immediate allergic reactions, and those patients need to be seen by allergy before a second dose.”
Dr. Freeman added that “it’s really interesting to think about how our bodies are really reacting to the vaccine in a way that’s mimicking our body’s reactions to COVID-19.” For example, some patients who got vaccinated developed chilblains similar to the “COVID toes” described in infected patients, apparently as part of the body’s immune response to the virus. “We’ve seen this in patients who actually had COVID and had prior COVID toes and then actually got a flare with their vaccine. And then we’ve also seen it in patients who never had COVID.”
In regard to general advice for patients, she said, “I do still encourage my patients who previously had COVID to go ahead and get the vaccine even if they had a skin manifestation with COVID.”
Shari Lipner, MD, PhD, associate professor of clinical dermatology, Weill Cornell Medicine, New York, said she has have seen only a handful of cases of delayed large local reactions and local injection site reactions after COVID-19 vaccination. “I have seen a significant number of cases of acute urticaria following the first and second doses,” she said in an interview. “However, it is important to keep in mind that we cannot determine cause and effect for the cases of acute urticaria. They may or may not be vaccine related.”
Fortunately, none of the adverse effects she’s seen have been severe. “It is important that dermatologists educate the public and their patients that most people do not develop any skin reaction in response to the vaccine,” she said. In the minority who do, “reactions tend to be mild and are not life-threatening. Many of these skin reactions resolve on their own without treatment.”
She added that “patients with pernio/chilblains or herpes zoster following vaccination should be referred by a board-certified dermatologist for prompt treatment and to avoid sequelae.”
‘COVID vaccine arm’
Delayed local reactions to the Moderna vaccine were also described in a report published online on May 12, 2021, in JAMA Dermatology, after the AAD meeting, in 16 patients referred to the Yale New Haven (Conn.) Hospital Dermatology service who experienced delayed localized cutaneous hypersensitivity reactions a median of 7 days after receiving the vaccine (range, 2-12 days), from Jan. 20 to Feb. 12, 2021. No such cases were reported in Pfizer vaccine recipients.
Of the 16 patients, whose median age was 38 years and who were mostly women, 15 developed the reaction after the first dose, described as “pruritic and variably painful erythematous reactions near the injection site,” which lasted a median of 5 days (range, 1-21 days). After the second dose, 12 of the 16 patients developed injection-site reactions (including one patient who had no reaction after dose 1), a median of 2 days after the vaccine was administered (range, 0-5 days). Histologic results of a biopsy in one patient with a reaction to the second dose “ demonstrated mild predominantly perivascular and focal interstitial mixed infiltrate with lymphocytes and eosinophils consistent with a dermal hypersensitivity reaction,” wrote Alicia J. Little, MD, PhD, of the department of dermatology, Yale University, New Haven, and coauthors.
Compared with immediate hypersensitivity reactions, occurring within 4 hours of vaccination, such as anaphylaxis and urticaria, they concluded that “these delayed localized hypersensitivity reactions are not a contraindication to subsequent vaccination,” and they proposed that they be named “COVID vaccine arm.”
Dr. Freeman reported no disclosures. Dr. Lipner also had no relevant disclosures. Dr. Little reported receiving a grant from the National Center for Advancing Translational Science and a Women’s Health Career Development Award from the Dermatology Foundation while the study was conducted; another author reported equity in Johnson & Johnson in his spouse’s retirement fund outside the submitted work.
The good news is that these side effects tend to be minor and vanish within a few days, Esther Freeman, MD, PhD, said in a presentation at the American Academy of Dermatology Virtual Meeting Experience.
“The reality is actually very reassuring,” Dr. Freeman said, especially in light of what is currently known about when the rashes occur and how anaphylaxis is extremely uncommon. Now, she added, dermatologists can tell patients who had reactions to their initial vaccination that “we know you had this big reaction, and we know that it was upsetting and uncomfortable. But it may not happen the second time around. And if it does, [the reaction is] probably going to be smaller.”
Dr. Freeman, associate professor of dermatology at Harvard Medical School, Boston, highlighted a study published in the Journal of the American Academy of Dermatology that she coauthored with dermatologists across the United States. The researchers tracked 414 cutaneous reactions to the Moderna (83%) and Pfizer (17%) COVID-19 vaccines in a group of patients, which was 90% female, 78% White, and mostly from the United States. Their average age was 44 years. The cases were reported to the AAD–International League of Dermatological Societies registry of COVID-19 cutaneous manifestations.
While most were women, “it’s a little hard to know if this is really going to end up being a true finding,” said Dr. Freeman, the registry’s principal investigator and a member of the AAD’s COVID-19 Ad Hoc Task Force. “If you think about who got vaccinated early, it was health care providers, and the American health care workforce is over 70% female. So I think there’s a little bit of bias here. There may also be a bias because women may be slightly more likely to report or go to their health care provider for a rash.”
Delayed large local reactions were the most common, accounting for 66% (175 cases) of the 267 skin reactions reported after the first Moderna vaccine dose and 30% (31 cases) of the 102 reactions reported after the second dose. These reactions represented 15% (5 cases) of the 34 skin reactions reported after the first Pfizer vaccine dose and 18% (7 cases) of the 40 reactions after the second dose.
There are two peaks with that first dose, Dr. Freeman said. “There’s a peak around day 2 or 3. And there’s another peak around day 7 or 8 with some of these reactions. Only 27% who had a reaction with the first dose had the same reaction with the second.” She added that these reactions “are not cellulitis and don’t require antibiotics.”
Other more common reactions included local injection-site reactions (swelling, erythema, and pain), urticaria (after 24 hours in almost all cases, occurring at a higher rate in patients who received the Pfizer vaccine), and morbilliform eruptions.
Dr. Freeman said that patients may experience redness and swelling in the hands and feet that can be “very uncomfortable.” She described one patient “who was having a hard time actually closing his fist, just because of the amount of swelling and redness in his hand. It did resolve, and it’s important to reassure your patients it will go away.”
According to this study, less common reports of other cutaneous findings with both vaccines included 9 reports of swelling at the site of cosmetic fillers, 8 reports of pernio/chilblains, 10 reports of varicella zoster, 4 reports of herpes simplex flares, 4 pityriasis rosea–like reactions, and 4 rashes in infants of vaccinated breastfeeding mothers.
The study noted that “patients responded well to topical corticosteroids, oral antihistamines, and/or pain-relieving medications. These reactions resolved after a median of 3-4 days.”
It’s important to understand that none of the patients developed anaphylaxis after the second dose even if they’d had a reaction to the first dose, Dr. Freeman said. “But I should point out that we’re talking about reactions that have started more than 4 hours after the vaccine. If a rash such as a urticaria specifically starts within 4 hours of vaccination, that’s in a different category. Those are considered more immediate allergic reactions, and those patients need to be seen by allergy before a second dose.”
Dr. Freeman added that “it’s really interesting to think about how our bodies are really reacting to the vaccine in a way that’s mimicking our body’s reactions to COVID-19.” For example, some patients who got vaccinated developed chilblains similar to the “COVID toes” described in infected patients, apparently as part of the body’s immune response to the virus. “We’ve seen this in patients who actually had COVID and had prior COVID toes and then actually got a flare with their vaccine. And then we’ve also seen it in patients who never had COVID.”
In regard to general advice for patients, she said, “I do still encourage my patients who previously had COVID to go ahead and get the vaccine even if they had a skin manifestation with COVID.”
Shari Lipner, MD, PhD, associate professor of clinical dermatology, Weill Cornell Medicine, New York, said she has have seen only a handful of cases of delayed large local reactions and local injection site reactions after COVID-19 vaccination. “I have seen a significant number of cases of acute urticaria following the first and second doses,” she said in an interview. “However, it is important to keep in mind that we cannot determine cause and effect for the cases of acute urticaria. They may or may not be vaccine related.”
Fortunately, none of the adverse effects she’s seen have been severe. “It is important that dermatologists educate the public and their patients that most people do not develop any skin reaction in response to the vaccine,” she said. In the minority who do, “reactions tend to be mild and are not life-threatening. Many of these skin reactions resolve on their own without treatment.”
She added that “patients with pernio/chilblains or herpes zoster following vaccination should be referred by a board-certified dermatologist for prompt treatment and to avoid sequelae.”
‘COVID vaccine arm’
Delayed local reactions to the Moderna vaccine were also described in a report published online on May 12, 2021, in JAMA Dermatology, after the AAD meeting, in 16 patients referred to the Yale New Haven (Conn.) Hospital Dermatology service who experienced delayed localized cutaneous hypersensitivity reactions a median of 7 days after receiving the vaccine (range, 2-12 days), from Jan. 20 to Feb. 12, 2021. No such cases were reported in Pfizer vaccine recipients.
Of the 16 patients, whose median age was 38 years and who were mostly women, 15 developed the reaction after the first dose, described as “pruritic and variably painful erythematous reactions near the injection site,” which lasted a median of 5 days (range, 1-21 days). After the second dose, 12 of the 16 patients developed injection-site reactions (including one patient who had no reaction after dose 1), a median of 2 days after the vaccine was administered (range, 0-5 days). Histologic results of a biopsy in one patient with a reaction to the second dose “ demonstrated mild predominantly perivascular and focal interstitial mixed infiltrate with lymphocytes and eosinophils consistent with a dermal hypersensitivity reaction,” wrote Alicia J. Little, MD, PhD, of the department of dermatology, Yale University, New Haven, and coauthors.
Compared with immediate hypersensitivity reactions, occurring within 4 hours of vaccination, such as anaphylaxis and urticaria, they concluded that “these delayed localized hypersensitivity reactions are not a contraindication to subsequent vaccination,” and they proposed that they be named “COVID vaccine arm.”
Dr. Freeman reported no disclosures. Dr. Lipner also had no relevant disclosures. Dr. Little reported receiving a grant from the National Center for Advancing Translational Science and a Women’s Health Career Development Award from the Dermatology Foundation while the study was conducted; another author reported equity in Johnson & Johnson in his spouse’s retirement fund outside the submitted work.
The good news is that these side effects tend to be minor and vanish within a few days, Esther Freeman, MD, PhD, said in a presentation at the American Academy of Dermatology Virtual Meeting Experience.
“The reality is actually very reassuring,” Dr. Freeman said, especially in light of what is currently known about when the rashes occur and how anaphylaxis is extremely uncommon. Now, she added, dermatologists can tell patients who had reactions to their initial vaccination that “we know you had this big reaction, and we know that it was upsetting and uncomfortable. But it may not happen the second time around. And if it does, [the reaction is] probably going to be smaller.”
Dr. Freeman, associate professor of dermatology at Harvard Medical School, Boston, highlighted a study published in the Journal of the American Academy of Dermatology that she coauthored with dermatologists across the United States. The researchers tracked 414 cutaneous reactions to the Moderna (83%) and Pfizer (17%) COVID-19 vaccines in a group of patients, which was 90% female, 78% White, and mostly from the United States. Their average age was 44 years. The cases were reported to the AAD–International League of Dermatological Societies registry of COVID-19 cutaneous manifestations.
While most were women, “it’s a little hard to know if this is really going to end up being a true finding,” said Dr. Freeman, the registry’s principal investigator and a member of the AAD’s COVID-19 Ad Hoc Task Force. “If you think about who got vaccinated early, it was health care providers, and the American health care workforce is over 70% female. So I think there’s a little bit of bias here. There may also be a bias because women may be slightly more likely to report or go to their health care provider for a rash.”
Delayed large local reactions were the most common, accounting for 66% (175 cases) of the 267 skin reactions reported after the first Moderna vaccine dose and 30% (31 cases) of the 102 reactions reported after the second dose. These reactions represented 15% (5 cases) of the 34 skin reactions reported after the first Pfizer vaccine dose and 18% (7 cases) of the 40 reactions after the second dose.
There are two peaks with that first dose, Dr. Freeman said. “There’s a peak around day 2 or 3. And there’s another peak around day 7 or 8 with some of these reactions. Only 27% who had a reaction with the first dose had the same reaction with the second.” She added that these reactions “are not cellulitis and don’t require antibiotics.”
Other more common reactions included local injection-site reactions (swelling, erythema, and pain), urticaria (after 24 hours in almost all cases, occurring at a higher rate in patients who received the Pfizer vaccine), and morbilliform eruptions.
Dr. Freeman said that patients may experience redness and swelling in the hands and feet that can be “very uncomfortable.” She described one patient “who was having a hard time actually closing his fist, just because of the amount of swelling and redness in his hand. It did resolve, and it’s important to reassure your patients it will go away.”
According to this study, less common reports of other cutaneous findings with both vaccines included 9 reports of swelling at the site of cosmetic fillers, 8 reports of pernio/chilblains, 10 reports of varicella zoster, 4 reports of herpes simplex flares, 4 pityriasis rosea–like reactions, and 4 rashes in infants of vaccinated breastfeeding mothers.
The study noted that “patients responded well to topical corticosteroids, oral antihistamines, and/or pain-relieving medications. These reactions resolved after a median of 3-4 days.”
It’s important to understand that none of the patients developed anaphylaxis after the second dose even if they’d had a reaction to the first dose, Dr. Freeman said. “But I should point out that we’re talking about reactions that have started more than 4 hours after the vaccine. If a rash such as a urticaria specifically starts within 4 hours of vaccination, that’s in a different category. Those are considered more immediate allergic reactions, and those patients need to be seen by allergy before a second dose.”
Dr. Freeman added that “it’s really interesting to think about how our bodies are really reacting to the vaccine in a way that’s mimicking our body’s reactions to COVID-19.” For example, some patients who got vaccinated developed chilblains similar to the “COVID toes” described in infected patients, apparently as part of the body’s immune response to the virus. “We’ve seen this in patients who actually had COVID and had prior COVID toes and then actually got a flare with their vaccine. And then we’ve also seen it in patients who never had COVID.”
In regard to general advice for patients, she said, “I do still encourage my patients who previously had COVID to go ahead and get the vaccine even if they had a skin manifestation with COVID.”
Shari Lipner, MD, PhD, associate professor of clinical dermatology, Weill Cornell Medicine, New York, said she has have seen only a handful of cases of delayed large local reactions and local injection site reactions after COVID-19 vaccination. “I have seen a significant number of cases of acute urticaria following the first and second doses,” she said in an interview. “However, it is important to keep in mind that we cannot determine cause and effect for the cases of acute urticaria. They may or may not be vaccine related.”
Fortunately, none of the adverse effects she’s seen have been severe. “It is important that dermatologists educate the public and their patients that most people do not develop any skin reaction in response to the vaccine,” she said. In the minority who do, “reactions tend to be mild and are not life-threatening. Many of these skin reactions resolve on their own without treatment.”
She added that “patients with pernio/chilblains or herpes zoster following vaccination should be referred by a board-certified dermatologist for prompt treatment and to avoid sequelae.”
‘COVID vaccine arm’
Delayed local reactions to the Moderna vaccine were also described in a report published online on May 12, 2021, in JAMA Dermatology, after the AAD meeting, in 16 patients referred to the Yale New Haven (Conn.) Hospital Dermatology service who experienced delayed localized cutaneous hypersensitivity reactions a median of 7 days after receiving the vaccine (range, 2-12 days), from Jan. 20 to Feb. 12, 2021. No such cases were reported in Pfizer vaccine recipients.
Of the 16 patients, whose median age was 38 years and who were mostly women, 15 developed the reaction after the first dose, described as “pruritic and variably painful erythematous reactions near the injection site,” which lasted a median of 5 days (range, 1-21 days). After the second dose, 12 of the 16 patients developed injection-site reactions (including one patient who had no reaction after dose 1), a median of 2 days after the vaccine was administered (range, 0-5 days). Histologic results of a biopsy in one patient with a reaction to the second dose “ demonstrated mild predominantly perivascular and focal interstitial mixed infiltrate with lymphocytes and eosinophils consistent with a dermal hypersensitivity reaction,” wrote Alicia J. Little, MD, PhD, of the department of dermatology, Yale University, New Haven, and coauthors.
Compared with immediate hypersensitivity reactions, occurring within 4 hours of vaccination, such as anaphylaxis and urticaria, they concluded that “these delayed localized hypersensitivity reactions are not a contraindication to subsequent vaccination,” and they proposed that they be named “COVID vaccine arm.”
Dr. Freeman reported no disclosures. Dr. Lipner also had no relevant disclosures. Dr. Little reported receiving a grant from the National Center for Advancing Translational Science and a Women’s Health Career Development Award from the Dermatology Foundation while the study was conducted; another author reported equity in Johnson & Johnson in his spouse’s retirement fund outside the submitted work.
FROM AAD VMX 2021
States ready plans to get Pfizer COVID vaccine to younger teens
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
Adulterants in street drugs could increase susceptibility to COVID
The composition of street drugs like heroin and cocaine are changing. According to a new analysis, almost all contain at least one toxic adulterant, and many contain a plethora. Most adulterants have pharmacologic activities and toxicities. Their presence has added impact in the context of the COVID-19 pandemic, since some may cause a drastic drop in white blood cells that could leave drug users more vulnerable to infection.
“It’s remarkable that we just forgot to notice, in the horrendous transition from prescription opioid epidemic to the illicit opioid and psychostimulant epidemics, that we would have to pay special attention to what the medications are in the drugs that the person was exposed to – and for how long,” said Mark S. Gold, MD, a coauthor of the review.
The analysis showed that adulterants include new psychoactive substances, industrial compounds, fungicides, veterinary medications, and various impurities. In addition, other various medications are being found in street drugs, such as antipsychotics, antidepressants, anxiolytics, antihistamines, anthelmintics, anesthetics, anti-inflammatory agents, antipyretics, analgesics, antispasmodics, antiarrhythmics, antimalarials, bronchodilators, decongestants, expectorants, muscle relaxers, natural/synthetic hallucinogens, and sedatives.
Illicit drugs are by nature manufactured without Food and Drug Administration oversight, and it is becoming increasingly common that substances like leftover medicines and other active drugs are added to illicit drug batches to add weight, said Dr. Gold, a professor at Washington University,St. Louis. The study appeared in Current Psychopharmacology.
Effects of adulterants ‘terrifying’
The findings of adulterants and their consequences are concerning, according to Jean Lud Cadet, MD, who was asked to comment on the findings. “The blood dysplasia, the pulmonary problems that some of those adulterants can cause – it’s actually terrifying, to put it bluntly,” said Dr. Cadet, who is a senior investigator and chief of the Molecular Neuropsychiatry Research Branch at the National Institute on Drug Abuse.
Before 2000, street drugs were generally diluted with comparatively benign substances such as caffeine, sugars, or lidocaine. Drugs like phenacetin, levamisole, acetaminophen, and diltiazem began to appear in heroin and cocaine in the late 1990s, and by 2010, more powerful adulterants like fentanyl, ketamine, and quetiapine became common.
In 2015, the U.S. Department of State partnered with the Colombo Plan, an international organization based in Sri Lanka, to use field spectroscopy to detect toxins directly in cocaine and heroin samples found in Argentina, Brazil, Ecuador, Peru, Sri Lanka, Thailand, Honduras, Guatemala, Mexico, Colombia, and South Africa. They found a range of adulterants such as aminopyrine, diltiazem, metamizole, levamisole, and phenacetin.
A similar project with 431 heroin and cocaine samples from Vermont and Kentucky found that 69% of samples had five or more controlled drugs, toxic adulterants, or impurities. About 15% had nine or more, and 95% of samples had at least one toxic adulterant.
In the midst of the COVID-19 pandemic, these adulterants take on even greater significance. Individuals with substance use disorders often have other health conditions that can make them more vulnerable to viral infections, and this could be exacerbated by the effects of adulterants on white blood cells or other systems. The pandemic has also had an indirect effect by causing a shortage of street drugs. During production shortages, traffickers might boost potency by adding more cutting agents and adulterants. As a result, COVID-19 and opioid addiction tend to reinforce each other.
“The clinical message would be that our [substance use] patients will contract infectious disease and need to be prioritized for [COVID-19] vaccination,” said Dr. Gold.
The findings came as a surprise to Dr. Cadet, and that illustrates a need to publicize the presence of adulterants in street drugs.
“If I wasn’t aware of many of these, then the general public is also not going to be aware of them,” Dr. Cadet said. “Scientists, including myself, and government agencies need to do a better job [of communicating this issue].”
The study references individuals with substance use disorder, but Dr. Cadet cautioned that anyone who uses street drugs, even once or twice, could be a victim of adulterants. “You don’t need to have met criteria for diagnosis in order to suffer the consequences.”
The study had no funding. Dr. Gold and Dr. Cadet have no relevant financial disclosures.
The composition of street drugs like heroin and cocaine are changing. According to a new analysis, almost all contain at least one toxic adulterant, and many contain a plethora. Most adulterants have pharmacologic activities and toxicities. Their presence has added impact in the context of the COVID-19 pandemic, since some may cause a drastic drop in white blood cells that could leave drug users more vulnerable to infection.
“It’s remarkable that we just forgot to notice, in the horrendous transition from prescription opioid epidemic to the illicit opioid and psychostimulant epidemics, that we would have to pay special attention to what the medications are in the drugs that the person was exposed to – and for how long,” said Mark S. Gold, MD, a coauthor of the review.
The analysis showed that adulterants include new psychoactive substances, industrial compounds, fungicides, veterinary medications, and various impurities. In addition, other various medications are being found in street drugs, such as antipsychotics, antidepressants, anxiolytics, antihistamines, anthelmintics, anesthetics, anti-inflammatory agents, antipyretics, analgesics, antispasmodics, antiarrhythmics, antimalarials, bronchodilators, decongestants, expectorants, muscle relaxers, natural/synthetic hallucinogens, and sedatives.
Illicit drugs are by nature manufactured without Food and Drug Administration oversight, and it is becoming increasingly common that substances like leftover medicines and other active drugs are added to illicit drug batches to add weight, said Dr. Gold, a professor at Washington University,St. Louis. The study appeared in Current Psychopharmacology.
Effects of adulterants ‘terrifying’
The findings of adulterants and their consequences are concerning, according to Jean Lud Cadet, MD, who was asked to comment on the findings. “The blood dysplasia, the pulmonary problems that some of those adulterants can cause – it’s actually terrifying, to put it bluntly,” said Dr. Cadet, who is a senior investigator and chief of the Molecular Neuropsychiatry Research Branch at the National Institute on Drug Abuse.
Before 2000, street drugs were generally diluted with comparatively benign substances such as caffeine, sugars, or lidocaine. Drugs like phenacetin, levamisole, acetaminophen, and diltiazem began to appear in heroin and cocaine in the late 1990s, and by 2010, more powerful adulterants like fentanyl, ketamine, and quetiapine became common.
In 2015, the U.S. Department of State partnered with the Colombo Plan, an international organization based in Sri Lanka, to use field spectroscopy to detect toxins directly in cocaine and heroin samples found in Argentina, Brazil, Ecuador, Peru, Sri Lanka, Thailand, Honduras, Guatemala, Mexico, Colombia, and South Africa. They found a range of adulterants such as aminopyrine, diltiazem, metamizole, levamisole, and phenacetin.
A similar project with 431 heroin and cocaine samples from Vermont and Kentucky found that 69% of samples had five or more controlled drugs, toxic adulterants, or impurities. About 15% had nine or more, and 95% of samples had at least one toxic adulterant.
In the midst of the COVID-19 pandemic, these adulterants take on even greater significance. Individuals with substance use disorders often have other health conditions that can make them more vulnerable to viral infections, and this could be exacerbated by the effects of adulterants on white blood cells or other systems. The pandemic has also had an indirect effect by causing a shortage of street drugs. During production shortages, traffickers might boost potency by adding more cutting agents and adulterants. As a result, COVID-19 and opioid addiction tend to reinforce each other.
“The clinical message would be that our [substance use] patients will contract infectious disease and need to be prioritized for [COVID-19] vaccination,” said Dr. Gold.
The findings came as a surprise to Dr. Cadet, and that illustrates a need to publicize the presence of adulterants in street drugs.
“If I wasn’t aware of many of these, then the general public is also not going to be aware of them,” Dr. Cadet said. “Scientists, including myself, and government agencies need to do a better job [of communicating this issue].”
The study references individuals with substance use disorder, but Dr. Cadet cautioned that anyone who uses street drugs, even once or twice, could be a victim of adulterants. “You don’t need to have met criteria for diagnosis in order to suffer the consequences.”
The study had no funding. Dr. Gold and Dr. Cadet have no relevant financial disclosures.
The composition of street drugs like heroin and cocaine are changing. According to a new analysis, almost all contain at least one toxic adulterant, and many contain a plethora. Most adulterants have pharmacologic activities and toxicities. Their presence has added impact in the context of the COVID-19 pandemic, since some may cause a drastic drop in white blood cells that could leave drug users more vulnerable to infection.
“It’s remarkable that we just forgot to notice, in the horrendous transition from prescription opioid epidemic to the illicit opioid and psychostimulant epidemics, that we would have to pay special attention to what the medications are in the drugs that the person was exposed to – and for how long,” said Mark S. Gold, MD, a coauthor of the review.
The analysis showed that adulterants include new psychoactive substances, industrial compounds, fungicides, veterinary medications, and various impurities. In addition, other various medications are being found in street drugs, such as antipsychotics, antidepressants, anxiolytics, antihistamines, anthelmintics, anesthetics, anti-inflammatory agents, antipyretics, analgesics, antispasmodics, antiarrhythmics, antimalarials, bronchodilators, decongestants, expectorants, muscle relaxers, natural/synthetic hallucinogens, and sedatives.
Illicit drugs are by nature manufactured without Food and Drug Administration oversight, and it is becoming increasingly common that substances like leftover medicines and other active drugs are added to illicit drug batches to add weight, said Dr. Gold, a professor at Washington University,St. Louis. The study appeared in Current Psychopharmacology.
Effects of adulterants ‘terrifying’
The findings of adulterants and their consequences are concerning, according to Jean Lud Cadet, MD, who was asked to comment on the findings. “The blood dysplasia, the pulmonary problems that some of those adulterants can cause – it’s actually terrifying, to put it bluntly,” said Dr. Cadet, who is a senior investigator and chief of the Molecular Neuropsychiatry Research Branch at the National Institute on Drug Abuse.
Before 2000, street drugs were generally diluted with comparatively benign substances such as caffeine, sugars, or lidocaine. Drugs like phenacetin, levamisole, acetaminophen, and diltiazem began to appear in heroin and cocaine in the late 1990s, and by 2010, more powerful adulterants like fentanyl, ketamine, and quetiapine became common.
In 2015, the U.S. Department of State partnered with the Colombo Plan, an international organization based in Sri Lanka, to use field spectroscopy to detect toxins directly in cocaine and heroin samples found in Argentina, Brazil, Ecuador, Peru, Sri Lanka, Thailand, Honduras, Guatemala, Mexico, Colombia, and South Africa. They found a range of adulterants such as aminopyrine, diltiazem, metamizole, levamisole, and phenacetin.
A similar project with 431 heroin and cocaine samples from Vermont and Kentucky found that 69% of samples had five or more controlled drugs, toxic adulterants, or impurities. About 15% had nine or more, and 95% of samples had at least one toxic adulterant.
In the midst of the COVID-19 pandemic, these adulterants take on even greater significance. Individuals with substance use disorders often have other health conditions that can make them more vulnerable to viral infections, and this could be exacerbated by the effects of adulterants on white blood cells or other systems. The pandemic has also had an indirect effect by causing a shortage of street drugs. During production shortages, traffickers might boost potency by adding more cutting agents and adulterants. As a result, COVID-19 and opioid addiction tend to reinforce each other.
“The clinical message would be that our [substance use] patients will contract infectious disease and need to be prioritized for [COVID-19] vaccination,” said Dr. Gold.
The findings came as a surprise to Dr. Cadet, and that illustrates a need to publicize the presence of adulterants in street drugs.
“If I wasn’t aware of many of these, then the general public is also not going to be aware of them,” Dr. Cadet said. “Scientists, including myself, and government agencies need to do a better job [of communicating this issue].”
The study references individuals with substance use disorder, but Dr. Cadet cautioned that anyone who uses street drugs, even once or twice, could be a victim of adulterants. “You don’t need to have met criteria for diagnosis in order to suffer the consequences.”
The study had no funding. Dr. Gold and Dr. Cadet have no relevant financial disclosures.
FROM CURRENT PSYCHOPHARMACOLOGY
FDA clears cap device for ‘smart’ insulin pens for diabetes
The U.S. Food and Drug Administration has cleared the Bigfoot Unity Diabetes Management System, a cap device that connects to insulin pens and translates continuous glucose data into dosing recommendations, for use in individuals aged 12 and older.
The Bigfoot Unity System has three primary components – proprietary smart pen caps for both rapid- and long-acting insulin, a mobile app, and an integrated FreeStyle Libre 2 continuous glucose monitor (iCGM) sensor, which was FDA-cleared in June 2020 – that fit into the person’s dose-decision process when they need it throughout the day.
It allows the user to scan the FreeStyle Libre 2 sensor, displaying the user’s current glucose value, trend arrow, and recommended correction dose. The smart pen cap also directly displays the health care provider’s suggested meal insulin doses with the correction dose. In just a few steps the system gives the person with diabetes support to make real-time treatment decisions.
It also includes hypoglycemia alerts and is compatible with all major U.S. brands of rapid- and long-acting disposable insulin pens.
Health care providers can monitor the patient’s data through a secure web portal called the Bigfoot Clinic Hub.
JDRF said in a statement it “applauds the U.S. FDA on its decision to provide clearance for the Bigfoot Unity Diabetes Management by Bigfoot Biomedical.”
The new system “fills a critical gap and brings benefits of automation and device interconnectedness to people with diabetes who rely on multiple daily injections to manage their blood sugar levels.” It is a “win for both the type 1 and type 2 diabetes communities as it broadens the options of treatment to alleviate daily burdens.”
Growing market for smart insulin pens
The device is the latest advance in the “smart pen” field of semiautomated insulin delivery in which pen and compatible devices, software, and platforms are teamed up in various combinations to provide easier insulin dosing for patients with diabetes who require multiple daily injections but don’t wear insulin pumps.
On May 6, 2021, Eli Lilly announced it had signed “strategic international agreements” with Dexcom, Glooko, MyDiabby Healthcare, and Roche to provide platforms or devices compatible with Lilly’s prefilled Tempo Pen, which is already available in several global markets, and the Tempo Smart Button, currently in late-stage development and pending CE mark.
And in November 2020, Medtronic launched a new version of its smart insulin pen with integrated CGM called the InPen. The reusable insulin injector pen uses a smartphone app to calculate dosing of short-acting insulin based on CGM readings and allows users to view glucose readings and insulin dose information. It was originally launched in 2017 by Companion Medical, and the company was acquired by Medtronic in September 2020.
Novo Nordisk and Sanofi are also developing products in the smart pen space.
More information about the Bigfoot Unity Program is available here.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has cleared the Bigfoot Unity Diabetes Management System, a cap device that connects to insulin pens and translates continuous glucose data into dosing recommendations, for use in individuals aged 12 and older.
The Bigfoot Unity System has three primary components – proprietary smart pen caps for both rapid- and long-acting insulin, a mobile app, and an integrated FreeStyle Libre 2 continuous glucose monitor (iCGM) sensor, which was FDA-cleared in June 2020 – that fit into the person’s dose-decision process when they need it throughout the day.
It allows the user to scan the FreeStyle Libre 2 sensor, displaying the user’s current glucose value, trend arrow, and recommended correction dose. The smart pen cap also directly displays the health care provider’s suggested meal insulin doses with the correction dose. In just a few steps the system gives the person with diabetes support to make real-time treatment decisions.
It also includes hypoglycemia alerts and is compatible with all major U.S. brands of rapid- and long-acting disposable insulin pens.
Health care providers can monitor the patient’s data through a secure web portal called the Bigfoot Clinic Hub.
JDRF said in a statement it “applauds the U.S. FDA on its decision to provide clearance for the Bigfoot Unity Diabetes Management by Bigfoot Biomedical.”
The new system “fills a critical gap and brings benefits of automation and device interconnectedness to people with diabetes who rely on multiple daily injections to manage their blood sugar levels.” It is a “win for both the type 1 and type 2 diabetes communities as it broadens the options of treatment to alleviate daily burdens.”
Growing market for smart insulin pens
The device is the latest advance in the “smart pen” field of semiautomated insulin delivery in which pen and compatible devices, software, and platforms are teamed up in various combinations to provide easier insulin dosing for patients with diabetes who require multiple daily injections but don’t wear insulin pumps.
On May 6, 2021, Eli Lilly announced it had signed “strategic international agreements” with Dexcom, Glooko, MyDiabby Healthcare, and Roche to provide platforms or devices compatible with Lilly’s prefilled Tempo Pen, which is already available in several global markets, and the Tempo Smart Button, currently in late-stage development and pending CE mark.
And in November 2020, Medtronic launched a new version of its smart insulin pen with integrated CGM called the InPen. The reusable insulin injector pen uses a smartphone app to calculate dosing of short-acting insulin based on CGM readings and allows users to view glucose readings and insulin dose information. It was originally launched in 2017 by Companion Medical, and the company was acquired by Medtronic in September 2020.
Novo Nordisk and Sanofi are also developing products in the smart pen space.
More information about the Bigfoot Unity Program is available here.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has cleared the Bigfoot Unity Diabetes Management System, a cap device that connects to insulin pens and translates continuous glucose data into dosing recommendations, for use in individuals aged 12 and older.
The Bigfoot Unity System has three primary components – proprietary smart pen caps for both rapid- and long-acting insulin, a mobile app, and an integrated FreeStyle Libre 2 continuous glucose monitor (iCGM) sensor, which was FDA-cleared in June 2020 – that fit into the person’s dose-decision process when they need it throughout the day.
It allows the user to scan the FreeStyle Libre 2 sensor, displaying the user’s current glucose value, trend arrow, and recommended correction dose. The smart pen cap also directly displays the health care provider’s suggested meal insulin doses with the correction dose. In just a few steps the system gives the person with diabetes support to make real-time treatment decisions.
It also includes hypoglycemia alerts and is compatible with all major U.S. brands of rapid- and long-acting disposable insulin pens.
Health care providers can monitor the patient’s data through a secure web portal called the Bigfoot Clinic Hub.
JDRF said in a statement it “applauds the U.S. FDA on its decision to provide clearance for the Bigfoot Unity Diabetes Management by Bigfoot Biomedical.”
The new system “fills a critical gap and brings benefits of automation and device interconnectedness to people with diabetes who rely on multiple daily injections to manage their blood sugar levels.” It is a “win for both the type 1 and type 2 diabetes communities as it broadens the options of treatment to alleviate daily burdens.”
Growing market for smart insulin pens
The device is the latest advance in the “smart pen” field of semiautomated insulin delivery in which pen and compatible devices, software, and platforms are teamed up in various combinations to provide easier insulin dosing for patients with diabetes who require multiple daily injections but don’t wear insulin pumps.
On May 6, 2021, Eli Lilly announced it had signed “strategic international agreements” with Dexcom, Glooko, MyDiabby Healthcare, and Roche to provide platforms or devices compatible with Lilly’s prefilled Tempo Pen, which is already available in several global markets, and the Tempo Smart Button, currently in late-stage development and pending CE mark.
And in November 2020, Medtronic launched a new version of its smart insulin pen with integrated CGM called the InPen. The reusable insulin injector pen uses a smartphone app to calculate dosing of short-acting insulin based on CGM readings and allows users to view glucose readings and insulin dose information. It was originally launched in 2017 by Companion Medical, and the company was acquired by Medtronic in September 2020.
Novo Nordisk and Sanofi are also developing products in the smart pen space.
More information about the Bigfoot Unity Program is available here.
A version of this article first appeared on Medscape.com.
Palliative care in the pandemic: How one hospital met the challenge
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
FROM SGIM 2021
FDA blazes path for ‘real-world’ evidence as proof of efficacy
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
In 2016, results from the LEADER trial of liraglutide in patients with type 2 diabetes helped jump-start awareness of the potential role of this new class of drugs, the glucagonlike peptide–1 receptor agonists, for reducing cardiovascular events. The randomized, placebo-controlled trial enrolled more than 9000 patients at more than 400 sites in over 30 countries, and took nearly 6 years from the start of patient enrollment to publication of the landmark results.
In December 2020, an independent team of researchers published results from a study with a design identical to LEADER, but used data that came not from a massive, global, years-long trial but from already-existing numbers culled from three large U.S. insurance claim databases. The result of this emulation using real-world data was virtually identical to what the actual trial showed, replicating both the direction and statistical significance of the original finding of the randomized, controlled trial (RCT).
What if research proved that this sort of RCT emulation could reliably be done on a regular basis? What might it mean for regulatory decisions on drugs and devices that historically have been based entirely on efficacy evidence from RCTs?
Making the most of a sea of observational data
Medicine in the United States has become increasingly awash in a sea of observational data collected from sources that include electronic health records, insurance claims, and increasingly, personal-health monitoring devices.
The Food and Drug Administration is now in the process of trying to figure out how it can legitimately harness this tsunami of real-world data to make efficacy decisions, essentially creating a new category of evidence to complement traditional data from randomized trials. It’s an opportunity that agency staff and their outside advisors have been keen to seize, especially given the soaring cost of prospective, randomized trials.
Recognition of this untapped resource in part led to a key initiative, among many others, included in the 21st Century Cures Act, passed in December 2016. Among the Act’s mandates was that, by the end of 2021, the FDA would issue guidance on when drug sponsors could use real-world evidence (RWE) to either help support a new indication for an already approved drug or help satisfy postapproval study requirements.
The initiative recognizes that this approach is not appropriate for initial drug approvals, which remain exclusively reliant on evidence from RCTs. Instead, it seems best suited to support expanding indications for already approved drugs.
Although FDA staff have made progress in identifying the challenges and broadening their understanding of how to best handle real-world data that come from observing patients in routine practice, agency leaders stress that this complex issue will likely not be fully resolved by their guidance to be published later this year. The FDA released a draft of the guidance in May 2019.
Can RWE be ‘credible and reliable?’
“Whether observational, nonrandomized data can become credible enough to use is what we’re talking about. These are possibilities that need to be explained and better understood,” said Robert Temple, MD, deputy director for clinical science of the FDA Center for Drug Evaluation and Research.
“Since the 1970s, the FDA has recognized historical controls as legitimate, so it’s possible [for RWE] to be credible. The big test is when is it credible and reliable enough [to assess efficacy]?” wondered Dr. Temple during a 2-day workshop on the topic held mid-February and organized by Duke University’s Margolis Center for Health Policy.
“We’re approaching an inflection point regarding how observational studies are generated and used, but our evidentiary standards will not lower, and it will be a case-by-case decision” by the agency as they review future RWE submissions, said John Concato, MD, the FDA’s associate director for real-world evidence, during the workshop.
“We are working toward guidance development, but also looking down the road to what we need to do to enable this,” said Dr. Concato. “It’s a complicated issue. If it was easy, it would have already been fixed.” He added that the agency will likely release a “portfolio” of guidance for submitting real-world data and RWE. Real-world data are raw information that, when analyzed, become RWE.
In short, the FDA seems headed toward guidance that won’t spell out a pathway that guarantees success using RWE but will at least open the door to consideration of this unprecedented application.
Not like flipping a switch
The guidance will not activate acceptance of RWE all at once. “It’s not like a light switch,” cautioned Adam Kroetsch, MPP, research director for biomedical innovation and regulatory policy at Duke-Margolis in Washington, D.C. “It’s an evolutionary process,” and the upcoming guidance will provide “just a little more clarity” on what sorts of best practices using RWE the FDA will find persuasive. “It’s hard for the FDA to clearly say what it’s looking for until they see some good examples,” Dr. Kroetsch said in an interview.
What will change is that drug sponsors can submit using RWE, and the FDA “will have a more open-minded view,” predicted Sebastian Schneeweiss, MD, ScD, a workshop participant and chief of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital in Boston. “For the first time, a law required [the FDA] to take a serious look” at observational data for efficacy assessment.
“The FDA has had a bias against using RWE for evidence of efficacy but has long used it to understand drug safety. Now the FDA is trying to wrap its arms around how to best use RWE” for efficacy decisions, said Joseph S. Ross, MD, another workshop participant and professor of medicine and public health at Yale University, New Haven, Conn.
The agency’s cautious approach is reassuring, Dr. Ross noted in an interview. “There was worry that the 21st Century Cures Act would open the door to allowing real-world data to be used in ways that weren’t very reliable. Very quickly, the FDA started trying to figure out the best ways to use these data in reasonable ways.”
Duplicating RCTs with RWE
To help better understand the potential use of RWE, the FDA sponsored several demonstration projects. Researchers presented results from three of these projects during the workshop in February. All three examined whether RWE, plugged into the design of an actual RCT, can produce roughly similar results when similar patients are used.
A generally consistent finding from the three demonstration projects was that “when the data are fit for purpose” the emulated or duplicated analyses with RWE “can come to similar conclusions” as the actual RCTs, said Dr. Schneeweiss, who leads one of the demonstration projects, RCT DUPLICATE.
At the workshop he reported results from RWE duplications of 20 different RCTs using insurance claims data from U.S. patients. The findings came from 10 duplications already reported in Circulation in December 2020 (including a duplication of the LEADER trial), and an additional 10 as yet unpublished RCT duplications. In the next few months, the researchers intend to assess a final group of 10 more RCT duplications.
Workshop participants also presented results from two other FDA demonstration projects: the OPERAND program run by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard; and the CERSI program based at Yale and the Mayo Clinic in Rochester, Minn. Both are smaller in scale than RCT DUPLICATE, incorporate lab data in addition to claims data, and in some cases test how well RWE can emulate RCTs that are not yet completed.
Collectively, results from these demonstration projects suggest that RWE can successfully emulate the results of an RCT, said Dr. Ross, a coinvestigator on the CERSI study. But the CERSI findings also highlighted how an RCT can fall short of clinical relevance.
“One of our most important findings was that RCTs don’t always represent real-world practice,” he said. His group attempted to replicate the 5,000-patient GRADE trial of four different drug options added to metformin in patients with type 2 diabetes. One of the four options included insulin glargine (Lantus), and the attempt to emulate the study with RWE hit the bump that no relevant real-world patients in their US claims database actually received the formulation.
That means the GRADE trial “is almost meaningless. It doesn’t reflect real-world practice,” Dr. Ross noted.
Results from the three demonstration projects “highlight the gaps we still have,” summed up Dr. Kroetsch. “They show where we need better data” from observational sources that function as well as data from RCTs.
Still, the demonstration project results are “an important step forward in establishing the validity of real-world evidence,” commented David Kerr, MBChB, an endocrinologist and director of research and innovation at the Sansum Diabetes Research Institute in Santa Barbara, Calif.
‘Target trials’ tether RWE
The target trial approach to designing an observational study is a key tool for boosting reliability and applicability of the results. The idea is to create a well-designed trial that could be the basis for a conventional RCT, and then use observational data to flesh out the target trial instead of collecting data from prospectively enrolled patients.
Designing observational studies that emulate target trials allows causal inferences, said Miguel A. Hernán, MD, DrPH, a professor of biostatistics and epidemiology at the Harvard School of Public Health, Boston. Plugging real-world data into the framework of an appropriately designed target trial substantially cuts the risk of a biased analysis, he explained during the workshop.
However, the approach has limitations. The target trial must be a pragmatic trial, and the approach does not work for placebo-controlled trials, although it can accommodate a usual-care control arm. It also usually precludes patient blinding, testing treatments not used in routine practice, and close monitoring of patients in ways that are uncommon in usual care.
The target trial approach received broad endorsement during the workshop as the future for observational studies destined for efficacy consideration by the FDA.
“The idea of prespecifying a target trial is a really fantastic place to start,” commented Robert Ball, MD, deputy director of the FDA Office of Surveillance and Epidemiology. “There is still a whole set of questions once the trial is prespecified, but prespecification would be a fantastic step forward,” he said during the workshop.
Participants also endorsed other important steps to boost the value of observational studies for regulatory reviews, including preregistering the study on a site such as clinicaltrials.gov; being fully transparent about the origins of observational data; using data that match the needs of the target trial; not reviewing the data in advance to avoid cherry picking and gaming the analysis; and reporting neutral or negative results when they occur, something often not currently done for observational analyses.
But although there was clear progress and much agreement among thought leaders at the workshop, FDA representatives stressed caution in moving forward.
“No easy answer”
“With more experience, we can learn what works and what doesn’t work in generating valid results from observational studies,” said Dr. Concato. “Although the observational results have upside potential, we need to learn more. There is no easy answer, no checklist for fit-for-use data, no off-the-shelf study design, and no ideal analytic method.”
Dr. Concato acknowledged that the FDA’s goal is clear given the 2016 legislation. “The FDA is embracing our obligations under the 21st Century Cures Act to evaluate use of real-world data and real-world evidence.”
He also suggested that researchers “shy away from a false dichotomy of RCTs or observational studies and instead think about how and when RCTs and observational studies can be designed and conducted to yield trustworthy results.” Dr. Concato’s solution: “a taxonomy of interventional or noninterventional studies.”
“The FDA is under enormous pressure to embrace real-world evidence, both because of the economics of running RCTs and because of the availability of new observational data from electronic health records, wearable devices, claims, etc.,” said Dr. Kerr, who did not participate in the workshop but coauthored an editorial that calls for using real-world data in regulatory decisions for drugs and devices for diabetes. These factors create an “irresistible force” spurring the FDA to consider observational, noninterventional data.
“I think the FDA really wants this to go forward,” Dr. Kerr added in an interview. “The FDA keeps telling us that clinical trials do not have enough women or patients from minority groups. Real-world data is a way to address that. This will not be the death of RCTs, but this work shines a light on the deficiencies of RCTs and how the deficiencies can be dealt with.”
Dr. Kroetsch has reported no relevant financial relationships. Dr. Schneeweiss has reported being a consultant to and holding equity in Aetion and receiving research funding from the FDA. Dr. Ross has reported receiving research funding from the FDA, Johnson & Johnson, and Medtronic. Dr. Hernán has reported being a consultant for Cytel. Dr. Kerr has reported being a consultant for Ascensia, EOFlow, Lifecare, Merck, Novo Nordisk, Roche Diagnostics, and Voluntis. Dr. Temple, Dr. Concato, and Dr. Ball are FDA employees.
A version of this article first appeared on Medscape.com.
Small increase seen in new COVID-19 cases among children
After 2 consecutive weeks of declines, the number of new COVID-19 cases in children rose slightly, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
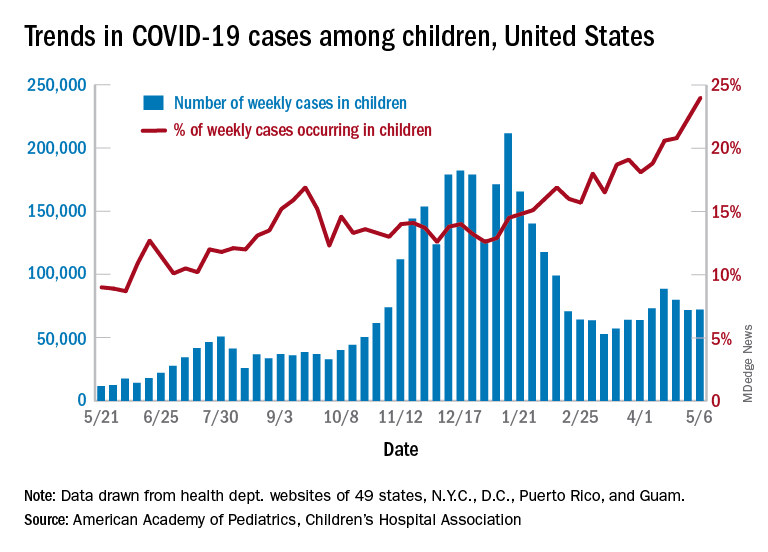
higher than at any other time during the pandemic, the AAP and CHA data show.
It is worth noting, however, that Rhode Island experienced a 30% increase in the last week, adding about 4,900 cases because of data revision and a lag in reporting, the AAP and CHA said in their weekly COVID-19 report.
All the new cases bring the total national count to just over 3.54 million in children, which represents 14.0% of all cases in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. The cumulative case rate as of May 6 was 5,122 per 100,000 children, the two organizations said.
All the new cases that were added to Rhode Island’s total give it the highest cumulative rate in the country: 9,614 cases per 100,000 children. North Dakota is right behind with 9,526 per 100,000, followed by Tennessee (8,898), Connecticut (8,281), and South Carolina (8,274). Vermont has the highest proportion of cases in children at 22.4%, with Alaska next at 20.3% and South Carolina third at 18.7%, according to the AAP and CHA.
Hawaii just reported its first COVID-19–related death in a child, which drops the number of states with zero deaths in children from 10 to 9. Two other new deaths in children from April 30 to May 6 bring the total number to 306 in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting the age distribution of deaths.
In a separate statement, AAP president Lee Savio Beers acknowledged the Food and Drug Administration’s authorization of the Pfizer-BioNTech vaccine for children aged 12-15 years as “a critically important step in bringing lifesaving vaccines to children and adolescents. ... We look forward to the discussion by the Advisory Committee on Immunization Practices of the CDC, which will make recommendations about the use of this vaccine in adolescents.”
After 2 consecutive weeks of declines, the number of new COVID-19 cases in children rose slightly, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

higher than at any other time during the pandemic, the AAP and CHA data show.
It is worth noting, however, that Rhode Island experienced a 30% increase in the last week, adding about 4,900 cases because of data revision and a lag in reporting, the AAP and CHA said in their weekly COVID-19 report.
All the new cases bring the total national count to just over 3.54 million in children, which represents 14.0% of all cases in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. The cumulative case rate as of May 6 was 5,122 per 100,000 children, the two organizations said.
All the new cases that were added to Rhode Island’s total give it the highest cumulative rate in the country: 9,614 cases per 100,000 children. North Dakota is right behind with 9,526 per 100,000, followed by Tennessee (8,898), Connecticut (8,281), and South Carolina (8,274). Vermont has the highest proportion of cases in children at 22.4%, with Alaska next at 20.3% and South Carolina third at 18.7%, according to the AAP and CHA.
Hawaii just reported its first COVID-19–related death in a child, which drops the number of states with zero deaths in children from 10 to 9. Two other new deaths in children from April 30 to May 6 bring the total number to 306 in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting the age distribution of deaths.
In a separate statement, AAP president Lee Savio Beers acknowledged the Food and Drug Administration’s authorization of the Pfizer-BioNTech vaccine for children aged 12-15 years as “a critically important step in bringing lifesaving vaccines to children and adolescents. ... We look forward to the discussion by the Advisory Committee on Immunization Practices of the CDC, which will make recommendations about the use of this vaccine in adolescents.”
After 2 consecutive weeks of declines, the number of new COVID-19 cases in children rose slightly, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

higher than at any other time during the pandemic, the AAP and CHA data show.
It is worth noting, however, that Rhode Island experienced a 30% increase in the last week, adding about 4,900 cases because of data revision and a lag in reporting, the AAP and CHA said in their weekly COVID-19 report.
All the new cases bring the total national count to just over 3.54 million in children, which represents 14.0% of all cases in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. The cumulative case rate as of May 6 was 5,122 per 100,000 children, the two organizations said.
All the new cases that were added to Rhode Island’s total give it the highest cumulative rate in the country: 9,614 cases per 100,000 children. North Dakota is right behind with 9,526 per 100,000, followed by Tennessee (8,898), Connecticut (8,281), and South Carolina (8,274). Vermont has the highest proportion of cases in children at 22.4%, with Alaska next at 20.3% and South Carolina third at 18.7%, according to the AAP and CHA.
Hawaii just reported its first COVID-19–related death in a child, which drops the number of states with zero deaths in children from 10 to 9. Two other new deaths in children from April 30 to May 6 bring the total number to 306 in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting the age distribution of deaths.
In a separate statement, AAP president Lee Savio Beers acknowledged the Food and Drug Administration’s authorization of the Pfizer-BioNTech vaccine for children aged 12-15 years as “a critically important step in bringing lifesaving vaccines to children and adolescents. ... We look forward to the discussion by the Advisory Committee on Immunization Practices of the CDC, which will make recommendations about the use of this vaccine in adolescents.”
Dr. Fauci: Feds may ease indoor mask mandates soon
Federal guidance on indoor mask use may change soon, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said on May 9.
He was asked whether it’s time to start relaxing indoor mask requirements.
“I think so, and I think you’re going to probably be seeing that as we go along and as more people get vaccinated,” Dr. Fauci said on ABC News’s This Week.Nearly 150 million adults in the United States – or about 58% of the adult population – have received at least one COVID-19 vaccine dose, according to the latest CDC tally. About 113 million adults, or 44%, are considered fully vaccinated.
“The CDC will be, you know, almost in real time … updating their recommendations and their guidelines,” Dr. Fauci said.
In April, the CDC relaxed its guidance for those who have been vaccinated against COVID-19. Those who have gotten a shot don’t need to wear a mask outdoors or in small indoor gatherings with other vaccinated people, but both vaccinated and unvaccinated people are still advised to wear masks in indoor public spaces.
“We do need to start being more liberal as we get more people vaccinated,” Dr. Fauci said. “As you get more people vaccinated, the number of cases per day will absolutely go down.”
The United States is averaging about 43,000 cases per day, he said, adding that the cases need to be “much, much lower.” When the case numbers drop and vaccination numbers increase, the risk of infection will fall dramatically indoors and outdoors, he said.
Even after the pandemic, though, wearing masks could become a seasonal habit, Dr. Fauci said May 9 on NBC News’s Meet the Press.“I think people have gotten used to the fact that wearing masks, clearly if you look at the data, it diminishes respiratory diseases. We’ve had practically a nonexistent flu season this year,” he said.
“So it is conceivable that as we go on, a year or 2 or more from now, that during certain seasonal periods when you have respiratory-borne viruses like the flu, people might actually elect to wear masks to diminish the likelihood that you’ll spread these respiratory-borne diseases,” he said.
Dr. Fauci was asked about indoor mask guidelines on May 9 after former FDA Commissioner Scott Gottlieb, MD, said face mask requirements should be relaxed.
“Certainly outdoors, we shouldn’t be putting limits on gatherings anymore,” Dr. Gottlieb said on CBS News’s Face the Nation.“The states where prevalence is low, vaccination rates are high, we have good testing in place, and we’re identifying infections, I think we could start lifting these restrictions indoors as well, on a broad basis,” he said.
Lifting pandemic-related restrictions in areas where they’re no longer necessary could also encourage people to implement them again if cases increase during future surges, such as this fall or winter, Dr. Gottlieb said.
At the same time, Americans should continue to follow CDC guidance and wait for new guidelines before changing their indoor mask use, Jeffrey Zients, the White House COVID-19 response coordinator, said on CNN’s State of the Union on May 9.
“We all want to get back to a normal lifestyle,” he said. “I think we’re on the path to do that, but stay disciplined, and let’s take advantage of the new privilege of being vaccinated and not wearing masks outdoors, for example, unless you’re in a crowded place.”
Mr. Zients pointed to President Joe Biden’s goal for 70% of adults to receive at least one vaccine dose by July 4.
“As we all move toward that 70% goal, there will be more and more advantages to being vaccinated,” he said. “And if you’re not vaccinated, you’re not protected.”
A version of this article first appeared on WebMD.com.
Federal guidance on indoor mask use may change soon, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said on May 9.
He was asked whether it’s time to start relaxing indoor mask requirements.
“I think so, and I think you’re going to probably be seeing that as we go along and as more people get vaccinated,” Dr. Fauci said on ABC News’s This Week.Nearly 150 million adults in the United States – or about 58% of the adult population – have received at least one COVID-19 vaccine dose, according to the latest CDC tally. About 113 million adults, or 44%, are considered fully vaccinated.
“The CDC will be, you know, almost in real time … updating their recommendations and their guidelines,” Dr. Fauci said.
In April, the CDC relaxed its guidance for those who have been vaccinated against COVID-19. Those who have gotten a shot don’t need to wear a mask outdoors or in small indoor gatherings with other vaccinated people, but both vaccinated and unvaccinated people are still advised to wear masks in indoor public spaces.
“We do need to start being more liberal as we get more people vaccinated,” Dr. Fauci said. “As you get more people vaccinated, the number of cases per day will absolutely go down.”
The United States is averaging about 43,000 cases per day, he said, adding that the cases need to be “much, much lower.” When the case numbers drop and vaccination numbers increase, the risk of infection will fall dramatically indoors and outdoors, he said.
Even after the pandemic, though, wearing masks could become a seasonal habit, Dr. Fauci said May 9 on NBC News’s Meet the Press.“I think people have gotten used to the fact that wearing masks, clearly if you look at the data, it diminishes respiratory diseases. We’ve had practically a nonexistent flu season this year,” he said.
“So it is conceivable that as we go on, a year or 2 or more from now, that during certain seasonal periods when you have respiratory-borne viruses like the flu, people might actually elect to wear masks to diminish the likelihood that you’ll spread these respiratory-borne diseases,” he said.
Dr. Fauci was asked about indoor mask guidelines on May 9 after former FDA Commissioner Scott Gottlieb, MD, said face mask requirements should be relaxed.
“Certainly outdoors, we shouldn’t be putting limits on gatherings anymore,” Dr. Gottlieb said on CBS News’s Face the Nation.“The states where prevalence is low, vaccination rates are high, we have good testing in place, and we’re identifying infections, I think we could start lifting these restrictions indoors as well, on a broad basis,” he said.
Lifting pandemic-related restrictions in areas where they’re no longer necessary could also encourage people to implement them again if cases increase during future surges, such as this fall or winter, Dr. Gottlieb said.
At the same time, Americans should continue to follow CDC guidance and wait for new guidelines before changing their indoor mask use, Jeffrey Zients, the White House COVID-19 response coordinator, said on CNN’s State of the Union on May 9.
“We all want to get back to a normal lifestyle,” he said. “I think we’re on the path to do that, but stay disciplined, and let’s take advantage of the new privilege of being vaccinated and not wearing masks outdoors, for example, unless you’re in a crowded place.”
Mr. Zients pointed to President Joe Biden’s goal for 70% of adults to receive at least one vaccine dose by July 4.
“As we all move toward that 70% goal, there will be more and more advantages to being vaccinated,” he said. “And if you’re not vaccinated, you’re not protected.”
A version of this article first appeared on WebMD.com.
Federal guidance on indoor mask use may change soon, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said on May 9.
He was asked whether it’s time to start relaxing indoor mask requirements.
“I think so, and I think you’re going to probably be seeing that as we go along and as more people get vaccinated,” Dr. Fauci said on ABC News’s This Week.Nearly 150 million adults in the United States – or about 58% of the adult population – have received at least one COVID-19 vaccine dose, according to the latest CDC tally. About 113 million adults, or 44%, are considered fully vaccinated.
“The CDC will be, you know, almost in real time … updating their recommendations and their guidelines,” Dr. Fauci said.
In April, the CDC relaxed its guidance for those who have been vaccinated against COVID-19. Those who have gotten a shot don’t need to wear a mask outdoors or in small indoor gatherings with other vaccinated people, but both vaccinated and unvaccinated people are still advised to wear masks in indoor public spaces.
“We do need to start being more liberal as we get more people vaccinated,” Dr. Fauci said. “As you get more people vaccinated, the number of cases per day will absolutely go down.”
The United States is averaging about 43,000 cases per day, he said, adding that the cases need to be “much, much lower.” When the case numbers drop and vaccination numbers increase, the risk of infection will fall dramatically indoors and outdoors, he said.
Even after the pandemic, though, wearing masks could become a seasonal habit, Dr. Fauci said May 9 on NBC News’s Meet the Press.“I think people have gotten used to the fact that wearing masks, clearly if you look at the data, it diminishes respiratory diseases. We’ve had practically a nonexistent flu season this year,” he said.
“So it is conceivable that as we go on, a year or 2 or more from now, that during certain seasonal periods when you have respiratory-borne viruses like the flu, people might actually elect to wear masks to diminish the likelihood that you’ll spread these respiratory-borne diseases,” he said.
Dr. Fauci was asked about indoor mask guidelines on May 9 after former FDA Commissioner Scott Gottlieb, MD, said face mask requirements should be relaxed.
“Certainly outdoors, we shouldn’t be putting limits on gatherings anymore,” Dr. Gottlieb said on CBS News’s Face the Nation.“The states where prevalence is low, vaccination rates are high, we have good testing in place, and we’re identifying infections, I think we could start lifting these restrictions indoors as well, on a broad basis,” he said.
Lifting pandemic-related restrictions in areas where they’re no longer necessary could also encourage people to implement them again if cases increase during future surges, such as this fall or winter, Dr. Gottlieb said.
At the same time, Americans should continue to follow CDC guidance and wait for new guidelines before changing their indoor mask use, Jeffrey Zients, the White House COVID-19 response coordinator, said on CNN’s State of the Union on May 9.
“We all want to get back to a normal lifestyle,” he said. “I think we’re on the path to do that, but stay disciplined, and let’s take advantage of the new privilege of being vaccinated and not wearing masks outdoors, for example, unless you’re in a crowded place.”
Mr. Zients pointed to President Joe Biden’s goal for 70% of adults to receive at least one vaccine dose by July 4.
“As we all move toward that 70% goal, there will be more and more advantages to being vaccinated,” he said. “And if you’re not vaccinated, you’re not protected.”
A version of this article first appeared on WebMD.com.







