User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
New ESC guidelines for cutting CV risk in noncardiac surgery
The European Society of Cardiology guidelines on cardiovascular assessment and management of patients undergoing noncardiac surgery have seen extensive revision since the 2014 version.
They still have the same aim – to prevent surgery-related bleeding complications, perioperative myocardial infarction/injury (PMI), stent thrombosis, acute heart failure, arrhythmias, pulmonary embolism, ischemic stroke, and cardiovascular (CV) death.
Cochairpersons Sigrun Halvorsen, MD, PhD, and Julinda Mehilli, MD, presented highlights from the guidelines at the annual congress of the European Society of Cardiology and the document was simultaneously published online in the European Heart Journal.
The document classifies noncardiac surgery into three levels of 30-day risk of CV death, MI, or stroke. Low (< 1%) risk includes eye or thyroid surgery; intermediate (1%-5%) risk includes knee or hip replacement or renal transplant; and high (> 5%) risk includes aortic aneurysm, lung transplant, or pancreatic or bladder cancer surgery (see more examples below).
It classifies patients as low risk if they are younger than 65 without CV disease or CV risk factors (smoking, hypertension, diabetes, dyslipidemia, family history); intermediate risk if they are 65 or older or have CV risk factors; and high risk if they have CVD.
In an interview, Dr. Halvorsen, professor in cardiology, University of Oslo, zeroed in on three important revisions:
First, recommendations for preoperative ECG and biomarkers are more specific, he noted.
The guidelines advise that before intermediate- or high-risk noncardiac surgery, in patients who have known CVD, CV risk factors (including age 65 or older), or symptoms suggestive of CVD:
- It is recommended to obtain a preoperative 12-lead ECG (class I).
- It is recommended to measure high-sensitivity cardiac troponin T (hs-cTn T) or high-sensitivity cardiac troponin I (hs-cTn I). It is also recommended to measure these biomarkers at 24 hours and 48 hours post surgery (class I).
- It should be considered to measure B-type natriuretic peptide or N-terminal of the prohormone BNP (NT-proBNP).
However, for low-risk patients undergoing low- and intermediate-risk noncardiac surgery, it is not recommended to routinely obtain preoperative ECG, hs-cTn T/I, or BNP/NT-proBNP concentrations (class III).
Troponins have a stronger class I recommendation, compared with the IIA recommendation for BNP, because they are useful for preoperative risk stratification and for diagnosis of PMI, Dr. Halvorsen explained. “Patients receive painkillers after surgery and may have no pain,” she noted, but they may have PMI, which has a bad prognosis.
Second, the guidelines recommend that “all patients should stop smoking 4 weeks before noncardiac surgery [class I],” she noted. Clinicians should also “measure hemoglobin, and if the patient is anemic, treat the anemia.”
Third, the sections on antithrombotic treatment have been significantly revised. “Bridging – stopping an oral antithrombotic drug and switching to a subcutaneous or IV drug – has been common,” Dr. Halvorsen said, “but recently we have new evidence that in most cases that increases the risk of bleeding.”
“We are [now] much more restrictive with respect to bridging” with unfractionated heparin or low-molecular-weight heparin, she said. “We recommend against bridging in patients with low to moderate thrombotic risk,” and bridging should only be considered in patients with mechanical prosthetic heart valves or with very high thrombotic risk.
More preoperative recommendations
In the guideline overview session at the congress, Dr. Halverson highlighted some of the new recommendations for preoperative risk assessment.
If time allows, it is recommended to optimize guideline-recommended treatment of CVD and control of CV risk factors including blood pressure, dyslipidemia, and diabetes, before noncardiac surgery (class I).
Patients commonly have “murmurs, chest pain, dyspnea, and edema that may suggest severe CVD, but may also be caused by noncardiac disease,” she noted. The guidelines state that “for patients with a newly detected murmur and symptoms or signs of CVD, transthoracic echocardiography is recommended before noncardiac surgery (class I).
“Many studies have been performed to try to find out if initiation of specific drugs before surgery could reduce the risk of complications,” Dr. Halvorsen noted. However, few have shown any benefit and “the question of presurgery initiation of beta-blockers has been greatly debated,” she said. “We have again reviewed the literature and concluded ‘Routine initiation of beta-blockers perioperatively is not recommended (class IIIA).’ “
“We adhere to the guidelines on acute and chronic coronary syndrome recommending 6-12 months of dual antiplatelet treatment as a standard before elective surgery,” she said. “However, in case of time-sensitive surgery, the duration of that treatment can be shortened down to a minimum of 1 month after elective PCI and a minimum of 3 months after PCI and ACS.”
Patients with specific types of CVD
Dr. Mehilli, a professor at Landshut-Achdorf (Germany) Hospital, highlighted some new guideline recommendations for patients who have specific types of cardiovascular disease.
Coronary artery disease (CAD). “For chronic coronary syndrome, a cardiac workup is recommended only for patients undergoing intermediate risk or high-risk noncardiac surgery.”
“Stress imaging should be considered before any high risk, noncardiac surgery in asymptomatic patients with poor functional capacity and prior PCI or coronary artery bypass graft (new recommendation, class IIa).”
Mitral valve regurgitation. For patients undergoing scheduled noncardiac surgery, who remain symptomatic despite guideline-directed medical treatment for mitral valve regurgitation (including resynchronization and myocardial revascularization), consider a valve intervention – either transcatheter or surgical – before noncardiac surgery in eligible patients with acceptable procedural risk (new recommendation).
Cardiac implantable electronic devices (CIED). For high-risk patients with CIEDs undergoing noncardiac surgery with high probability of electromagnetic interference, a CIED checkup and necessary reprogramming immediately before the procedure should be considered (new recommendation).
Arrhythmias. “I want only to stress,” Dr. Mehilli said, “in patients with atrial fibrillation with acute or worsening hemodynamic instability undergoing noncardiac surgery, an emergency electrical cardioversion is recommended (class I).”
Peripheral artery disease (PAD) and abdominal aortic aneurysm. For these patients “we do not recommend a routine referral for a cardiac workup. But we recommend it for patients with poor functional capacity or with significant risk factors or symptoms (new recommendations).”
Chronic arterial hypertension. “We have modified the recommendation, recommending avoidance of large perioperative fluctuations in blood pressure, and we do not recommend deferring noncardiac surgery in patients with stage 1 or 2 hypertension,” she said.
Postoperative cardiovascular complications
The most frequent postoperative cardiovascular complication is PMI, Dr. Mehilli noted.
“In the BASEL-PMI registry, the incidence of this complication around intermediate or high-risk noncardiac surgery was up to 15% among patients older than 65 years or with a history of CAD or PAD, which makes this kind of complication really important to prevent, to assess, and to know how to treat.”
“It is recommended to have a high awareness for perioperative cardiovascular complications, combined with surveillance for PMI in patients undergoing intermediate- or high-risk noncardiac surgery” based on serial measurements of high-sensitivity cardiac troponin.
The guidelines define PMI as “an increase in the delta of high-sensitivity troponin more than the upper level of normal,” Dr. Mehilli said. “It’s different from the one used in a rule-in algorithm for non-STEMI acute coronary syndrome.”
Postoperative atrial fibrillation (AFib) is observed in 2%-30% of noncardiac surgery patients in different registries, particularly in patients undergoing intermediate or high-risk noncardiac surgery, she noted.
“We propose an algorithm on how to prevent and treat this complication. I want to highlight that in patients with hemodynamic unstable postoperative AF[ib], an emergency cardioversion is indicated. For the others, a rate control with the target heart rate of less than 110 beats per minute is indicated.”
In patients with postoperative AFib, long-term oral anticoagulation therapy should be considered in all patients at risk for stroke, considering the anticipated net clinical benefit of oral anticoagulation therapy as well as informed patient preference (new recommendations).
Routine use of beta-blockers to prevent postoperative AFib in patients undergoing noncardiac surgery is not recommended.
The document also covers the management of patients with kidney disease, diabetes, cancer, obesity, and COVID-19. In general, elective noncardiac surgery should be postponed after a patient has COVID-19, until he or she recovers completely, and coexisting conditions are optimized.
The guidelines are available from the ESC website in several formats: pocket guidelines, pocket guidelines smartphone app, guidelines slide set, essential messages, and the European Heart Journal article.
Noncardiac surgery risk categories
The guideline includes a table that classifies noncardiac surgeries into three groups, based on the associated 30-day risk of death, MI, or stroke:
- Low (< 1%): breast, dental, eye, thyroid, and minor gynecologic, orthopedic, and urologic surgery.
- Intermediate (1%-5%): carotid surgery, endovascular aortic aneurysm repair, gallbladder surgery, head or neck surgery, hernia repair, peripheral arterial angioplasty, renal transplant, major gynecologic, orthopedic, or neurologic (hip or spine) surgery, or urologic surgery
- High (> 5%): aortic and major vascular surgery (including aortic aneurysm), bladder removal (usually as a result of cancer), limb amputation, lung or liver transplant, pancreatic surgery, or perforated bowel repair.
The guidelines were endorsed by the European Society of Anaesthesiology and Intensive Care. The guideline authors reported numerous disclosures.
A version of this article first appeared on Medscape.com.
The European Society of Cardiology guidelines on cardiovascular assessment and management of patients undergoing noncardiac surgery have seen extensive revision since the 2014 version.
They still have the same aim – to prevent surgery-related bleeding complications, perioperative myocardial infarction/injury (PMI), stent thrombosis, acute heart failure, arrhythmias, pulmonary embolism, ischemic stroke, and cardiovascular (CV) death.
Cochairpersons Sigrun Halvorsen, MD, PhD, and Julinda Mehilli, MD, presented highlights from the guidelines at the annual congress of the European Society of Cardiology and the document was simultaneously published online in the European Heart Journal.
The document classifies noncardiac surgery into three levels of 30-day risk of CV death, MI, or stroke. Low (< 1%) risk includes eye or thyroid surgery; intermediate (1%-5%) risk includes knee or hip replacement or renal transplant; and high (> 5%) risk includes aortic aneurysm, lung transplant, or pancreatic or bladder cancer surgery (see more examples below).
It classifies patients as low risk if they are younger than 65 without CV disease or CV risk factors (smoking, hypertension, diabetes, dyslipidemia, family history); intermediate risk if they are 65 or older or have CV risk factors; and high risk if they have CVD.
In an interview, Dr. Halvorsen, professor in cardiology, University of Oslo, zeroed in on three important revisions:
First, recommendations for preoperative ECG and biomarkers are more specific, he noted.
The guidelines advise that before intermediate- or high-risk noncardiac surgery, in patients who have known CVD, CV risk factors (including age 65 or older), or symptoms suggestive of CVD:
- It is recommended to obtain a preoperative 12-lead ECG (class I).
- It is recommended to measure high-sensitivity cardiac troponin T (hs-cTn T) or high-sensitivity cardiac troponin I (hs-cTn I). It is also recommended to measure these biomarkers at 24 hours and 48 hours post surgery (class I).
- It should be considered to measure B-type natriuretic peptide or N-terminal of the prohormone BNP (NT-proBNP).
However, for low-risk patients undergoing low- and intermediate-risk noncardiac surgery, it is not recommended to routinely obtain preoperative ECG, hs-cTn T/I, or BNP/NT-proBNP concentrations (class III).
Troponins have a stronger class I recommendation, compared with the IIA recommendation for BNP, because they are useful for preoperative risk stratification and for diagnosis of PMI, Dr. Halvorsen explained. “Patients receive painkillers after surgery and may have no pain,” she noted, but they may have PMI, which has a bad prognosis.
Second, the guidelines recommend that “all patients should stop smoking 4 weeks before noncardiac surgery [class I],” she noted. Clinicians should also “measure hemoglobin, and if the patient is anemic, treat the anemia.”
Third, the sections on antithrombotic treatment have been significantly revised. “Bridging – stopping an oral antithrombotic drug and switching to a subcutaneous or IV drug – has been common,” Dr. Halvorsen said, “but recently we have new evidence that in most cases that increases the risk of bleeding.”
“We are [now] much more restrictive with respect to bridging” with unfractionated heparin or low-molecular-weight heparin, she said. “We recommend against bridging in patients with low to moderate thrombotic risk,” and bridging should only be considered in patients with mechanical prosthetic heart valves or with very high thrombotic risk.
More preoperative recommendations
In the guideline overview session at the congress, Dr. Halverson highlighted some of the new recommendations for preoperative risk assessment.
If time allows, it is recommended to optimize guideline-recommended treatment of CVD and control of CV risk factors including blood pressure, dyslipidemia, and diabetes, before noncardiac surgery (class I).
Patients commonly have “murmurs, chest pain, dyspnea, and edema that may suggest severe CVD, but may also be caused by noncardiac disease,” she noted. The guidelines state that “for patients with a newly detected murmur and symptoms or signs of CVD, transthoracic echocardiography is recommended before noncardiac surgery (class I).
“Many studies have been performed to try to find out if initiation of specific drugs before surgery could reduce the risk of complications,” Dr. Halvorsen noted. However, few have shown any benefit and “the question of presurgery initiation of beta-blockers has been greatly debated,” she said. “We have again reviewed the literature and concluded ‘Routine initiation of beta-blockers perioperatively is not recommended (class IIIA).’ “
“We adhere to the guidelines on acute and chronic coronary syndrome recommending 6-12 months of dual antiplatelet treatment as a standard before elective surgery,” she said. “However, in case of time-sensitive surgery, the duration of that treatment can be shortened down to a minimum of 1 month after elective PCI and a minimum of 3 months after PCI and ACS.”
Patients with specific types of CVD
Dr. Mehilli, a professor at Landshut-Achdorf (Germany) Hospital, highlighted some new guideline recommendations for patients who have specific types of cardiovascular disease.
Coronary artery disease (CAD). “For chronic coronary syndrome, a cardiac workup is recommended only for patients undergoing intermediate risk or high-risk noncardiac surgery.”
“Stress imaging should be considered before any high risk, noncardiac surgery in asymptomatic patients with poor functional capacity and prior PCI or coronary artery bypass graft (new recommendation, class IIa).”
Mitral valve regurgitation. For patients undergoing scheduled noncardiac surgery, who remain symptomatic despite guideline-directed medical treatment for mitral valve regurgitation (including resynchronization and myocardial revascularization), consider a valve intervention – either transcatheter or surgical – before noncardiac surgery in eligible patients with acceptable procedural risk (new recommendation).
Cardiac implantable electronic devices (CIED). For high-risk patients with CIEDs undergoing noncardiac surgery with high probability of electromagnetic interference, a CIED checkup and necessary reprogramming immediately before the procedure should be considered (new recommendation).
Arrhythmias. “I want only to stress,” Dr. Mehilli said, “in patients with atrial fibrillation with acute or worsening hemodynamic instability undergoing noncardiac surgery, an emergency electrical cardioversion is recommended (class I).”
Peripheral artery disease (PAD) and abdominal aortic aneurysm. For these patients “we do not recommend a routine referral for a cardiac workup. But we recommend it for patients with poor functional capacity or with significant risk factors or symptoms (new recommendations).”
Chronic arterial hypertension. “We have modified the recommendation, recommending avoidance of large perioperative fluctuations in blood pressure, and we do not recommend deferring noncardiac surgery in patients with stage 1 or 2 hypertension,” she said.
Postoperative cardiovascular complications
The most frequent postoperative cardiovascular complication is PMI, Dr. Mehilli noted.
“In the BASEL-PMI registry, the incidence of this complication around intermediate or high-risk noncardiac surgery was up to 15% among patients older than 65 years or with a history of CAD or PAD, which makes this kind of complication really important to prevent, to assess, and to know how to treat.”
“It is recommended to have a high awareness for perioperative cardiovascular complications, combined with surveillance for PMI in patients undergoing intermediate- or high-risk noncardiac surgery” based on serial measurements of high-sensitivity cardiac troponin.
The guidelines define PMI as “an increase in the delta of high-sensitivity troponin more than the upper level of normal,” Dr. Mehilli said. “It’s different from the one used in a rule-in algorithm for non-STEMI acute coronary syndrome.”
Postoperative atrial fibrillation (AFib) is observed in 2%-30% of noncardiac surgery patients in different registries, particularly in patients undergoing intermediate or high-risk noncardiac surgery, she noted.
“We propose an algorithm on how to prevent and treat this complication. I want to highlight that in patients with hemodynamic unstable postoperative AF[ib], an emergency cardioversion is indicated. For the others, a rate control with the target heart rate of less than 110 beats per minute is indicated.”
In patients with postoperative AFib, long-term oral anticoagulation therapy should be considered in all patients at risk for stroke, considering the anticipated net clinical benefit of oral anticoagulation therapy as well as informed patient preference (new recommendations).
Routine use of beta-blockers to prevent postoperative AFib in patients undergoing noncardiac surgery is not recommended.
The document also covers the management of patients with kidney disease, diabetes, cancer, obesity, and COVID-19. In general, elective noncardiac surgery should be postponed after a patient has COVID-19, until he or she recovers completely, and coexisting conditions are optimized.
The guidelines are available from the ESC website in several formats: pocket guidelines, pocket guidelines smartphone app, guidelines slide set, essential messages, and the European Heart Journal article.
Noncardiac surgery risk categories
The guideline includes a table that classifies noncardiac surgeries into three groups, based on the associated 30-day risk of death, MI, or stroke:
- Low (< 1%): breast, dental, eye, thyroid, and minor gynecologic, orthopedic, and urologic surgery.
- Intermediate (1%-5%): carotid surgery, endovascular aortic aneurysm repair, gallbladder surgery, head or neck surgery, hernia repair, peripheral arterial angioplasty, renal transplant, major gynecologic, orthopedic, or neurologic (hip or spine) surgery, or urologic surgery
- High (> 5%): aortic and major vascular surgery (including aortic aneurysm), bladder removal (usually as a result of cancer), limb amputation, lung or liver transplant, pancreatic surgery, or perforated bowel repair.
The guidelines were endorsed by the European Society of Anaesthesiology and Intensive Care. The guideline authors reported numerous disclosures.
A version of this article first appeared on Medscape.com.
The European Society of Cardiology guidelines on cardiovascular assessment and management of patients undergoing noncardiac surgery have seen extensive revision since the 2014 version.
They still have the same aim – to prevent surgery-related bleeding complications, perioperative myocardial infarction/injury (PMI), stent thrombosis, acute heart failure, arrhythmias, pulmonary embolism, ischemic stroke, and cardiovascular (CV) death.
Cochairpersons Sigrun Halvorsen, MD, PhD, and Julinda Mehilli, MD, presented highlights from the guidelines at the annual congress of the European Society of Cardiology and the document was simultaneously published online in the European Heart Journal.
The document classifies noncardiac surgery into three levels of 30-day risk of CV death, MI, or stroke. Low (< 1%) risk includes eye or thyroid surgery; intermediate (1%-5%) risk includes knee or hip replacement or renal transplant; and high (> 5%) risk includes aortic aneurysm, lung transplant, or pancreatic or bladder cancer surgery (see more examples below).
It classifies patients as low risk if they are younger than 65 without CV disease or CV risk factors (smoking, hypertension, diabetes, dyslipidemia, family history); intermediate risk if they are 65 or older or have CV risk factors; and high risk if they have CVD.
In an interview, Dr. Halvorsen, professor in cardiology, University of Oslo, zeroed in on three important revisions:
First, recommendations for preoperative ECG and biomarkers are more specific, he noted.
The guidelines advise that before intermediate- or high-risk noncardiac surgery, in patients who have known CVD, CV risk factors (including age 65 or older), or symptoms suggestive of CVD:
- It is recommended to obtain a preoperative 12-lead ECG (class I).
- It is recommended to measure high-sensitivity cardiac troponin T (hs-cTn T) or high-sensitivity cardiac troponin I (hs-cTn I). It is also recommended to measure these biomarkers at 24 hours and 48 hours post surgery (class I).
- It should be considered to measure B-type natriuretic peptide or N-terminal of the prohormone BNP (NT-proBNP).
However, for low-risk patients undergoing low- and intermediate-risk noncardiac surgery, it is not recommended to routinely obtain preoperative ECG, hs-cTn T/I, or BNP/NT-proBNP concentrations (class III).
Troponins have a stronger class I recommendation, compared with the IIA recommendation for BNP, because they are useful for preoperative risk stratification and for diagnosis of PMI, Dr. Halvorsen explained. “Patients receive painkillers after surgery and may have no pain,” she noted, but they may have PMI, which has a bad prognosis.
Second, the guidelines recommend that “all patients should stop smoking 4 weeks before noncardiac surgery [class I],” she noted. Clinicians should also “measure hemoglobin, and if the patient is anemic, treat the anemia.”
Third, the sections on antithrombotic treatment have been significantly revised. “Bridging – stopping an oral antithrombotic drug and switching to a subcutaneous or IV drug – has been common,” Dr. Halvorsen said, “but recently we have new evidence that in most cases that increases the risk of bleeding.”
“We are [now] much more restrictive with respect to bridging” with unfractionated heparin or low-molecular-weight heparin, she said. “We recommend against bridging in patients with low to moderate thrombotic risk,” and bridging should only be considered in patients with mechanical prosthetic heart valves or with very high thrombotic risk.
More preoperative recommendations
In the guideline overview session at the congress, Dr. Halverson highlighted some of the new recommendations for preoperative risk assessment.
If time allows, it is recommended to optimize guideline-recommended treatment of CVD and control of CV risk factors including blood pressure, dyslipidemia, and diabetes, before noncardiac surgery (class I).
Patients commonly have “murmurs, chest pain, dyspnea, and edema that may suggest severe CVD, but may also be caused by noncardiac disease,” she noted. The guidelines state that “for patients with a newly detected murmur and symptoms or signs of CVD, transthoracic echocardiography is recommended before noncardiac surgery (class I).
“Many studies have been performed to try to find out if initiation of specific drugs before surgery could reduce the risk of complications,” Dr. Halvorsen noted. However, few have shown any benefit and “the question of presurgery initiation of beta-blockers has been greatly debated,” she said. “We have again reviewed the literature and concluded ‘Routine initiation of beta-blockers perioperatively is not recommended (class IIIA).’ “
“We adhere to the guidelines on acute and chronic coronary syndrome recommending 6-12 months of dual antiplatelet treatment as a standard before elective surgery,” she said. “However, in case of time-sensitive surgery, the duration of that treatment can be shortened down to a minimum of 1 month after elective PCI and a minimum of 3 months after PCI and ACS.”
Patients with specific types of CVD
Dr. Mehilli, a professor at Landshut-Achdorf (Germany) Hospital, highlighted some new guideline recommendations for patients who have specific types of cardiovascular disease.
Coronary artery disease (CAD). “For chronic coronary syndrome, a cardiac workup is recommended only for patients undergoing intermediate risk or high-risk noncardiac surgery.”
“Stress imaging should be considered before any high risk, noncardiac surgery in asymptomatic patients with poor functional capacity and prior PCI or coronary artery bypass graft (new recommendation, class IIa).”
Mitral valve regurgitation. For patients undergoing scheduled noncardiac surgery, who remain symptomatic despite guideline-directed medical treatment for mitral valve regurgitation (including resynchronization and myocardial revascularization), consider a valve intervention – either transcatheter or surgical – before noncardiac surgery in eligible patients with acceptable procedural risk (new recommendation).
Cardiac implantable electronic devices (CIED). For high-risk patients with CIEDs undergoing noncardiac surgery with high probability of electromagnetic interference, a CIED checkup and necessary reprogramming immediately before the procedure should be considered (new recommendation).
Arrhythmias. “I want only to stress,” Dr. Mehilli said, “in patients with atrial fibrillation with acute or worsening hemodynamic instability undergoing noncardiac surgery, an emergency electrical cardioversion is recommended (class I).”
Peripheral artery disease (PAD) and abdominal aortic aneurysm. For these patients “we do not recommend a routine referral for a cardiac workup. But we recommend it for patients with poor functional capacity or with significant risk factors or symptoms (new recommendations).”
Chronic arterial hypertension. “We have modified the recommendation, recommending avoidance of large perioperative fluctuations in blood pressure, and we do not recommend deferring noncardiac surgery in patients with stage 1 or 2 hypertension,” she said.
Postoperative cardiovascular complications
The most frequent postoperative cardiovascular complication is PMI, Dr. Mehilli noted.
“In the BASEL-PMI registry, the incidence of this complication around intermediate or high-risk noncardiac surgery was up to 15% among patients older than 65 years or with a history of CAD or PAD, which makes this kind of complication really important to prevent, to assess, and to know how to treat.”
“It is recommended to have a high awareness for perioperative cardiovascular complications, combined with surveillance for PMI in patients undergoing intermediate- or high-risk noncardiac surgery” based on serial measurements of high-sensitivity cardiac troponin.
The guidelines define PMI as “an increase in the delta of high-sensitivity troponin more than the upper level of normal,” Dr. Mehilli said. “It’s different from the one used in a rule-in algorithm for non-STEMI acute coronary syndrome.”
Postoperative atrial fibrillation (AFib) is observed in 2%-30% of noncardiac surgery patients in different registries, particularly in patients undergoing intermediate or high-risk noncardiac surgery, she noted.
“We propose an algorithm on how to prevent and treat this complication. I want to highlight that in patients with hemodynamic unstable postoperative AF[ib], an emergency cardioversion is indicated. For the others, a rate control with the target heart rate of less than 110 beats per minute is indicated.”
In patients with postoperative AFib, long-term oral anticoagulation therapy should be considered in all patients at risk for stroke, considering the anticipated net clinical benefit of oral anticoagulation therapy as well as informed patient preference (new recommendations).
Routine use of beta-blockers to prevent postoperative AFib in patients undergoing noncardiac surgery is not recommended.
The document also covers the management of patients with kidney disease, diabetes, cancer, obesity, and COVID-19. In general, elective noncardiac surgery should be postponed after a patient has COVID-19, until he or she recovers completely, and coexisting conditions are optimized.
The guidelines are available from the ESC website in several formats: pocket guidelines, pocket guidelines smartphone app, guidelines slide set, essential messages, and the European Heart Journal article.
Noncardiac surgery risk categories
The guideline includes a table that classifies noncardiac surgeries into three groups, based on the associated 30-day risk of death, MI, or stroke:
- Low (< 1%): breast, dental, eye, thyroid, and minor gynecologic, orthopedic, and urologic surgery.
- Intermediate (1%-5%): carotid surgery, endovascular aortic aneurysm repair, gallbladder surgery, head or neck surgery, hernia repair, peripheral arterial angioplasty, renal transplant, major gynecologic, orthopedic, or neurologic (hip or spine) surgery, or urologic surgery
- High (> 5%): aortic and major vascular surgery (including aortic aneurysm), bladder removal (usually as a result of cancer), limb amputation, lung or liver transplant, pancreatic surgery, or perforated bowel repair.
The guidelines were endorsed by the European Society of Anaesthesiology and Intensive Care. The guideline authors reported numerous disclosures.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2022
Vitamins or cocoa: Which preserves cognition?
Unexpected results from a phase 3 trial exploring the effect of multivitamins and cognition have now been published.
Originally presented last November at the 14th Clinical Trials on Alzheimer’s Disease (CTAD) conference, this is the first large-scale, long-term randomized controlled trial to examine the effects of cocoa extract and multivitamins on global cognition. The trial’s primary focus was on cocoa extract, which earlier studies suggest may preserve cognitive function. Analyzing the effect of multivitamins was a secondary outcome.
Showing vitamins, but not cocoa, were beneficial is the exact opposite of what researchers expected. Still, the results offer an interesting new direction for future study, lead investigator Laura D. Baker, PhD, professor of gerontology and geriatric medicine at Wake Forest University, Winston-Salem, N.C., said in an interview.
“This study made us take notice of a pathway for possible cognitive protection,” Dr. Baker said. “Without this study, we would never have looked down that road.”
The full results were published online in Alzheimer’s and Dementia.
Unexpected effect
The COSMOS-Mind study is a substudy to a larger parent trial called COSMOS. It investigated the effects of cocoa extract and a standard multivitamin-mineral on cardiovascular and cancer outcomes in more than 21,000 older participants.
In COSMOS-Mind, researchers tested whether daily intake of cocoa extract vs. placebo and a multivitamin-mineral vs. placebo improved cognition in older adults.
More than 2,200 participants aged 65 and older were enrolled and followed for 3 years. They completed tests over the telephone at baseline and annually to evaluate memory and other cognitive abilities.
Results showed cocoa extract had no effect on global cognition compared with placebo (mean z-score, 0.03; P = .28). Daily multivitamin use, however, did show significant benefits on global cognition vs. placebo (mean z, 0.07, P = .007).
The beneficial effect was most pronounced in participants with a history of cardiovascular disease (no history 0.06 vs. history 0.14; P = .01).
Researchers found similar protective effects for memory and executive function.
Dr. Baker suggested one possible explanation for the positive effects of multivitamins may be the boost in micronutrients and essential minerals they provided.
“With nutrient-deficient diets plus a high prevalence of cardiovascular disease, diabetes, and other medical comorbidities that we know impact the bioavailability of these nutrients, we are possibly dealing with older adults who are at below optimum in terms of their essential micronutrients and minerals,” she said.
“Even suboptimum levels of micronutrients and essential minerals can have significant consequences for brain health,” she added.
More research needed
Intriguing as the results may be, more work is needed before the findings could affect nutritional guidance, according to Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association.
“While the Alzheimer’s Association is encouraged by these results, we are not ready to recommend widespread use of a multivitamin supplement to reduce risk of cognitive decline in older adults,” Dr. Carrillo said in a statement.
“For now, and until there is more data, people should talk with their health care providers about the benefits and risks of all dietary supplements, including multivitamins,” she added.
Dr. Baker agreed, noting that the study was not designed to measure multivitamin use as a primary outcome. In addition, nearly 90% of the participants were non-Hispanic White, which is not representative of the overall population demographics.
The investigators are now designing another, larger trial that would include a more diverse participant pool. It will be aimed specifically at learning more about how and why multivitamins seem to offer a protective effect on cognition, Dr. Baker noted.
The study was funded by the National Institute on Aging of the National Institutes of Health. Dr. Baker and Dr. Carrillo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Unexpected results from a phase 3 trial exploring the effect of multivitamins and cognition have now been published.
Originally presented last November at the 14th Clinical Trials on Alzheimer’s Disease (CTAD) conference, this is the first large-scale, long-term randomized controlled trial to examine the effects of cocoa extract and multivitamins on global cognition. The trial’s primary focus was on cocoa extract, which earlier studies suggest may preserve cognitive function. Analyzing the effect of multivitamins was a secondary outcome.
Showing vitamins, but not cocoa, were beneficial is the exact opposite of what researchers expected. Still, the results offer an interesting new direction for future study, lead investigator Laura D. Baker, PhD, professor of gerontology and geriatric medicine at Wake Forest University, Winston-Salem, N.C., said in an interview.
“This study made us take notice of a pathway for possible cognitive protection,” Dr. Baker said. “Without this study, we would never have looked down that road.”
The full results were published online in Alzheimer’s and Dementia.
Unexpected effect
The COSMOS-Mind study is a substudy to a larger parent trial called COSMOS. It investigated the effects of cocoa extract and a standard multivitamin-mineral on cardiovascular and cancer outcomes in more than 21,000 older participants.
In COSMOS-Mind, researchers tested whether daily intake of cocoa extract vs. placebo and a multivitamin-mineral vs. placebo improved cognition in older adults.
More than 2,200 participants aged 65 and older were enrolled and followed for 3 years. They completed tests over the telephone at baseline and annually to evaluate memory and other cognitive abilities.
Results showed cocoa extract had no effect on global cognition compared with placebo (mean z-score, 0.03; P = .28). Daily multivitamin use, however, did show significant benefits on global cognition vs. placebo (mean z, 0.07, P = .007).
The beneficial effect was most pronounced in participants with a history of cardiovascular disease (no history 0.06 vs. history 0.14; P = .01).
Researchers found similar protective effects for memory and executive function.
Dr. Baker suggested one possible explanation for the positive effects of multivitamins may be the boost in micronutrients and essential minerals they provided.
“With nutrient-deficient diets plus a high prevalence of cardiovascular disease, diabetes, and other medical comorbidities that we know impact the bioavailability of these nutrients, we are possibly dealing with older adults who are at below optimum in terms of their essential micronutrients and minerals,” she said.
“Even suboptimum levels of micronutrients and essential minerals can have significant consequences for brain health,” she added.
More research needed
Intriguing as the results may be, more work is needed before the findings could affect nutritional guidance, according to Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association.
“While the Alzheimer’s Association is encouraged by these results, we are not ready to recommend widespread use of a multivitamin supplement to reduce risk of cognitive decline in older adults,” Dr. Carrillo said in a statement.
“For now, and until there is more data, people should talk with their health care providers about the benefits and risks of all dietary supplements, including multivitamins,” she added.
Dr. Baker agreed, noting that the study was not designed to measure multivitamin use as a primary outcome. In addition, nearly 90% of the participants were non-Hispanic White, which is not representative of the overall population demographics.
The investigators are now designing another, larger trial that would include a more diverse participant pool. It will be aimed specifically at learning more about how and why multivitamins seem to offer a protective effect on cognition, Dr. Baker noted.
The study was funded by the National Institute on Aging of the National Institutes of Health. Dr. Baker and Dr. Carrillo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Unexpected results from a phase 3 trial exploring the effect of multivitamins and cognition have now been published.
Originally presented last November at the 14th Clinical Trials on Alzheimer’s Disease (CTAD) conference, this is the first large-scale, long-term randomized controlled trial to examine the effects of cocoa extract and multivitamins on global cognition. The trial’s primary focus was on cocoa extract, which earlier studies suggest may preserve cognitive function. Analyzing the effect of multivitamins was a secondary outcome.
Showing vitamins, but not cocoa, were beneficial is the exact opposite of what researchers expected. Still, the results offer an interesting new direction for future study, lead investigator Laura D. Baker, PhD, professor of gerontology and geriatric medicine at Wake Forest University, Winston-Salem, N.C., said in an interview.
“This study made us take notice of a pathway for possible cognitive protection,” Dr. Baker said. “Without this study, we would never have looked down that road.”
The full results were published online in Alzheimer’s and Dementia.
Unexpected effect
The COSMOS-Mind study is a substudy to a larger parent trial called COSMOS. It investigated the effects of cocoa extract and a standard multivitamin-mineral on cardiovascular and cancer outcomes in more than 21,000 older participants.
In COSMOS-Mind, researchers tested whether daily intake of cocoa extract vs. placebo and a multivitamin-mineral vs. placebo improved cognition in older adults.
More than 2,200 participants aged 65 and older were enrolled and followed for 3 years. They completed tests over the telephone at baseline and annually to evaluate memory and other cognitive abilities.
Results showed cocoa extract had no effect on global cognition compared with placebo (mean z-score, 0.03; P = .28). Daily multivitamin use, however, did show significant benefits on global cognition vs. placebo (mean z, 0.07, P = .007).
The beneficial effect was most pronounced in participants with a history of cardiovascular disease (no history 0.06 vs. history 0.14; P = .01).
Researchers found similar protective effects for memory and executive function.
Dr. Baker suggested one possible explanation for the positive effects of multivitamins may be the boost in micronutrients and essential minerals they provided.
“With nutrient-deficient diets plus a high prevalence of cardiovascular disease, diabetes, and other medical comorbidities that we know impact the bioavailability of these nutrients, we are possibly dealing with older adults who are at below optimum in terms of their essential micronutrients and minerals,” she said.
“Even suboptimum levels of micronutrients and essential minerals can have significant consequences for brain health,” she added.
More research needed
Intriguing as the results may be, more work is needed before the findings could affect nutritional guidance, according to Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association.
“While the Alzheimer’s Association is encouraged by these results, we are not ready to recommend widespread use of a multivitamin supplement to reduce risk of cognitive decline in older adults,” Dr. Carrillo said in a statement.
“For now, and until there is more data, people should talk with their health care providers about the benefits and risks of all dietary supplements, including multivitamins,” she added.
Dr. Baker agreed, noting that the study was not designed to measure multivitamin use as a primary outcome. In addition, nearly 90% of the participants were non-Hispanic White, which is not representative of the overall population demographics.
The investigators are now designing another, larger trial that would include a more diverse participant pool. It will be aimed specifically at learning more about how and why multivitamins seem to offer a protective effect on cognition, Dr. Baker noted.
The study was funded by the National Institute on Aging of the National Institutes of Health. Dr. Baker and Dr. Carrillo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ALZHEIMER’S AND DEMENTIA
Cumulative blood pressure load: A better predictor of CV events?
Cumulative systolic blood pressure load, which can be calculated from serial blood pressure measurements, may provide better prediction of major cardiovascular events, compared with traditional blood pressure measures, a new study suggests.
“Our results suggest that cumulative blood pressure load is an independent predictor of cardiovascular events and should be used in future cardiovascular risk prediction algorithms,” the authors, led by Nelson Wang, MD, George Institute for Global Health, Sydney, conclude.
The study was published online in the Journal of the American College of Cardiology.
The researchers explain that the management of hypertension has traditionally centered around blood pressure measurements taken at a single timepoint, with adequate control defined as those measurements being below a predefined target threshold.
However, this approach fails to recognize blood pressure as a continuous measure that fluctuates over time and does not acknowledge that the most recently recorded measurement may not reflect previous blood pressure control.
More recently, studies have defined the time a patient spends below blood pressure target, or TIme at TaRgEt (TITRE), as a novel marker of cardiovascular risk that is independent of mean blood pressure.
Although TITRE has the added advantage of incorporating duration of control, it is unable to characterize the magnitude of blood pressure elevation, the researchers note.
They point out that an optimal measure as a risk factor for cardiovascular disease would account for both the magnitude and duration of elevated blood pressure.
Such a measure is cumulative blood pressure load, defined as the area under the curve (AUC) expressed in units of mm Hg by time.
The only prior study of this measure was small and retrospective and calculated cumulative blood pressure load from ambulatory blood pressure monitoring estimated over a short (24-hour) period.
Therefore, the aim of the current study was to estimate the association between cumulative systolic blood pressure load over a longer period (24 months) and subsequent major cardiovascular events.
To do this, the researchers conducted a post-hoc analysis of 9,338 patients with type 2 diabetes in the ADVANCE-ON study.
Cumulative systolic blood pressure load was defined as the AUC for systolic blood pressure values above 130 mm Hg divided by the AUC for all measured systolic blood pressure values over a 24-month exposure period.
Over a median 7.6 years of follow-up, 1,469 major cardiovascular events, 1,615 deaths, and 660 cardiovascular deaths occurred.
Results showed that each one standard deviation increase in cumulative systolic blood pressure load was associated with a 14% increase in major cardiovascular events, a 13% increase in all-cause mortality, and a 21% increase in cardiovascular death.
Cumulative systolic blood pressure load outperformed mean systolic blood pressure, time-below-target, and visit-to-visit systolic blood pressure variability for the prediction of cardiovascular events and death and also discriminated risk and reclassified more patients’ risk correctly than the other measures.
“Small improvements in risk prediction can have a major impact when scaled up across large high-risk populations. Furthermore, cumulative systolic pressure load may also prove useful to inform the design of future clinical trials,” the researchers say.
Although the present study only assessed cumulative systolic blood pressure load over 24 months, clinicians should recognize the importance of this measure over a lifetime, they note.
“This approach emphasizes the importance of early blood pressure–lowering interventions to reduce the cumulative systolic blood pressure load that each individual experiences over their lifetime,” they conclude.
The researchers suggest that, based on these results, cumulative systolic blood pressure load and visit-to-visit systolic blood pressure variability “should be used in conjunction in future cardiovascular risk prediction algorithms.”
In an accompanying editorial, Donald Lloyd-Jones, MD, Northwestern Feinberg School of Medicine, Chicago, says that before routinely adopting these new measures, several additional questions need to be addressed.
He notes that many patients in the current study already had cardiovascular disease, and it is not known whether the benefit was consistent among those with and without cardiovascular disease. In addition, longer term data using blood pressure measurements in the real-world clinical setting would be desirable, as well as information on whether these new measures add incremental value to existing risk prediction equations.
“Certainly, the next guidelines should reconsider all types of blood pressure measures, and other potential predictors, to optimize risk estimation and identification of patients with greatest net benefit from risk-reducing therapies,” Dr. Lloyd-Jones comments.
“Ultimately, clinicians should leverage as much information on their patients as possible to understand their blood pressure–related cardiovascular risk, to identify those who may be more likely have occult or emerging subclinical target organ damage, and to identify those who may have particular net benefit from earlier or more intensive treatment,” he concludes.
“These opportunities are more readily available with integration of data that allow for visualization of longer-term blood pressure patterns and incorporation of home monitoring and ambulatory monitoring data to monitor out-of-office blood pressure levels and control.”
A version of this article first appeared on Medscape.com.
Cumulative systolic blood pressure load, which can be calculated from serial blood pressure measurements, may provide better prediction of major cardiovascular events, compared with traditional blood pressure measures, a new study suggests.
“Our results suggest that cumulative blood pressure load is an independent predictor of cardiovascular events and should be used in future cardiovascular risk prediction algorithms,” the authors, led by Nelson Wang, MD, George Institute for Global Health, Sydney, conclude.
The study was published online in the Journal of the American College of Cardiology.
The researchers explain that the management of hypertension has traditionally centered around blood pressure measurements taken at a single timepoint, with adequate control defined as those measurements being below a predefined target threshold.
However, this approach fails to recognize blood pressure as a continuous measure that fluctuates over time and does not acknowledge that the most recently recorded measurement may not reflect previous blood pressure control.
More recently, studies have defined the time a patient spends below blood pressure target, or TIme at TaRgEt (TITRE), as a novel marker of cardiovascular risk that is independent of mean blood pressure.
Although TITRE has the added advantage of incorporating duration of control, it is unable to characterize the magnitude of blood pressure elevation, the researchers note.
They point out that an optimal measure as a risk factor for cardiovascular disease would account for both the magnitude and duration of elevated blood pressure.
Such a measure is cumulative blood pressure load, defined as the area under the curve (AUC) expressed in units of mm Hg by time.
The only prior study of this measure was small and retrospective and calculated cumulative blood pressure load from ambulatory blood pressure monitoring estimated over a short (24-hour) period.
Therefore, the aim of the current study was to estimate the association between cumulative systolic blood pressure load over a longer period (24 months) and subsequent major cardiovascular events.
To do this, the researchers conducted a post-hoc analysis of 9,338 patients with type 2 diabetes in the ADVANCE-ON study.
Cumulative systolic blood pressure load was defined as the AUC for systolic blood pressure values above 130 mm Hg divided by the AUC for all measured systolic blood pressure values over a 24-month exposure period.
Over a median 7.6 years of follow-up, 1,469 major cardiovascular events, 1,615 deaths, and 660 cardiovascular deaths occurred.
Results showed that each one standard deviation increase in cumulative systolic blood pressure load was associated with a 14% increase in major cardiovascular events, a 13% increase in all-cause mortality, and a 21% increase in cardiovascular death.
Cumulative systolic blood pressure load outperformed mean systolic blood pressure, time-below-target, and visit-to-visit systolic blood pressure variability for the prediction of cardiovascular events and death and also discriminated risk and reclassified more patients’ risk correctly than the other measures.
“Small improvements in risk prediction can have a major impact when scaled up across large high-risk populations. Furthermore, cumulative systolic pressure load may also prove useful to inform the design of future clinical trials,” the researchers say.
Although the present study only assessed cumulative systolic blood pressure load over 24 months, clinicians should recognize the importance of this measure over a lifetime, they note.
“This approach emphasizes the importance of early blood pressure–lowering interventions to reduce the cumulative systolic blood pressure load that each individual experiences over their lifetime,” they conclude.
The researchers suggest that, based on these results, cumulative systolic blood pressure load and visit-to-visit systolic blood pressure variability “should be used in conjunction in future cardiovascular risk prediction algorithms.”
In an accompanying editorial, Donald Lloyd-Jones, MD, Northwestern Feinberg School of Medicine, Chicago, says that before routinely adopting these new measures, several additional questions need to be addressed.
He notes that many patients in the current study already had cardiovascular disease, and it is not known whether the benefit was consistent among those with and without cardiovascular disease. In addition, longer term data using blood pressure measurements in the real-world clinical setting would be desirable, as well as information on whether these new measures add incremental value to existing risk prediction equations.
“Certainly, the next guidelines should reconsider all types of blood pressure measures, and other potential predictors, to optimize risk estimation and identification of patients with greatest net benefit from risk-reducing therapies,” Dr. Lloyd-Jones comments.
“Ultimately, clinicians should leverage as much information on their patients as possible to understand their blood pressure–related cardiovascular risk, to identify those who may be more likely have occult or emerging subclinical target organ damage, and to identify those who may have particular net benefit from earlier or more intensive treatment,” he concludes.
“These opportunities are more readily available with integration of data that allow for visualization of longer-term blood pressure patterns and incorporation of home monitoring and ambulatory monitoring data to monitor out-of-office blood pressure levels and control.”
A version of this article first appeared on Medscape.com.
Cumulative systolic blood pressure load, which can be calculated from serial blood pressure measurements, may provide better prediction of major cardiovascular events, compared with traditional blood pressure measures, a new study suggests.
“Our results suggest that cumulative blood pressure load is an independent predictor of cardiovascular events and should be used in future cardiovascular risk prediction algorithms,” the authors, led by Nelson Wang, MD, George Institute for Global Health, Sydney, conclude.
The study was published online in the Journal of the American College of Cardiology.
The researchers explain that the management of hypertension has traditionally centered around blood pressure measurements taken at a single timepoint, with adequate control defined as those measurements being below a predefined target threshold.
However, this approach fails to recognize blood pressure as a continuous measure that fluctuates over time and does not acknowledge that the most recently recorded measurement may not reflect previous blood pressure control.
More recently, studies have defined the time a patient spends below blood pressure target, or TIme at TaRgEt (TITRE), as a novel marker of cardiovascular risk that is independent of mean blood pressure.
Although TITRE has the added advantage of incorporating duration of control, it is unable to characterize the magnitude of blood pressure elevation, the researchers note.
They point out that an optimal measure as a risk factor for cardiovascular disease would account for both the magnitude and duration of elevated blood pressure.
Such a measure is cumulative blood pressure load, defined as the area under the curve (AUC) expressed in units of mm Hg by time.
The only prior study of this measure was small and retrospective and calculated cumulative blood pressure load from ambulatory blood pressure monitoring estimated over a short (24-hour) period.
Therefore, the aim of the current study was to estimate the association between cumulative systolic blood pressure load over a longer period (24 months) and subsequent major cardiovascular events.
To do this, the researchers conducted a post-hoc analysis of 9,338 patients with type 2 diabetes in the ADVANCE-ON study.
Cumulative systolic blood pressure load was defined as the AUC for systolic blood pressure values above 130 mm Hg divided by the AUC for all measured systolic blood pressure values over a 24-month exposure period.
Over a median 7.6 years of follow-up, 1,469 major cardiovascular events, 1,615 deaths, and 660 cardiovascular deaths occurred.
Results showed that each one standard deviation increase in cumulative systolic blood pressure load was associated with a 14% increase in major cardiovascular events, a 13% increase in all-cause mortality, and a 21% increase in cardiovascular death.
Cumulative systolic blood pressure load outperformed mean systolic blood pressure, time-below-target, and visit-to-visit systolic blood pressure variability for the prediction of cardiovascular events and death and also discriminated risk and reclassified more patients’ risk correctly than the other measures.
“Small improvements in risk prediction can have a major impact when scaled up across large high-risk populations. Furthermore, cumulative systolic pressure load may also prove useful to inform the design of future clinical trials,” the researchers say.
Although the present study only assessed cumulative systolic blood pressure load over 24 months, clinicians should recognize the importance of this measure over a lifetime, they note.
“This approach emphasizes the importance of early blood pressure–lowering interventions to reduce the cumulative systolic blood pressure load that each individual experiences over their lifetime,” they conclude.
The researchers suggest that, based on these results, cumulative systolic blood pressure load and visit-to-visit systolic blood pressure variability “should be used in conjunction in future cardiovascular risk prediction algorithms.”
In an accompanying editorial, Donald Lloyd-Jones, MD, Northwestern Feinberg School of Medicine, Chicago, says that before routinely adopting these new measures, several additional questions need to be addressed.
He notes that many patients in the current study already had cardiovascular disease, and it is not known whether the benefit was consistent among those with and without cardiovascular disease. In addition, longer term data using blood pressure measurements in the real-world clinical setting would be desirable, as well as information on whether these new measures add incremental value to existing risk prediction equations.
“Certainly, the next guidelines should reconsider all types of blood pressure measures, and other potential predictors, to optimize risk estimation and identification of patients with greatest net benefit from risk-reducing therapies,” Dr. Lloyd-Jones comments.
“Ultimately, clinicians should leverage as much information on their patients as possible to understand their blood pressure–related cardiovascular risk, to identify those who may be more likely have occult or emerging subclinical target organ damage, and to identify those who may have particular net benefit from earlier or more intensive treatment,” he concludes.
“These opportunities are more readily available with integration of data that allow for visualization of longer-term blood pressure patterns and incorporation of home monitoring and ambulatory monitoring data to monitor out-of-office blood pressure levels and control.”
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Should patients stand for office BP readings?
a new study suggests.
Combining three standing and three seated BP measurements in the same visit may lead to a “quicker diagnosis and save people a trip back to the office,” Wanpen Vongpatanasin, MD, professor of internal medicine and director of the hypertension section, cardiology division, UT Southwestern Medical Center, Dallas, said in an interview.
The study was presented at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Practice changing?
Clinical guidelines recommend office BP be taken in a seated position for most patients.
However, research has suggested that the sensitivity of seated office BP in diagnosing hypertension is about 50%, with high specificity around 90% during a single visit, Dr. Vongpatanasin explained.
At the follow-up visit, however, the second office BP yielded higher sensitivity to 80% but specificity fell to 55%. Nevertheless, the accuracy of standing BP measurements for diagnosing hypertension has not been investigated.
In a cross-sectional study, Dr. Vongpatanasin and colleagues determined the accuracy of both seated and standing BP for diagnosing hypertension in a single visit in 125 healthy adults who had not had a previous diagnosis of hypertension and were not taking any BP medications. The cohort had a mean age of 49 years, 62% were women, and 24% were Black.
During each office visit, seated BP was measured three times, then standing BP was measured three times using an automated and validated device.
Average seated BP was 123/76 mm Hg and average standing BP was 126/80 mm Hg.
Of the 125 participants, 42 (34%) had hypertension, defined as 24-hour ambulatory systolic/diastolic BP (SBP/DBP) of ≥ 125/75 mm Hg.
The sensitivity and specificity of seated SBP for hypertension was 43% and 92%, respectively.
“Interestingly, with standing SBP, sensitivity was improved to 74% and specificity dropped to 65% – which is okay; you will have to confirm a positive test anyway and when screening for a common disease you’d rather have a high sensitivity rather than low sensitivity to pick it up in this case,” Dr. Vongpatanasin said.
The area under receiver operating characteristic curve (AUROC) of standing SBP was significantly higher than seated SBP (Bayes factor [BF] = 11.8) when hypertension was defined as 24-hour SBP ≥ 125 mm Hg.
Similarly, when hypertension was defined as 24-hour DBP ≥ 75 mm Hg or daytime DBP ≥ 80 mm Hg, the AUROC of standing DBP was higher than seated DBP (all BF > 3).
The addition of standing to seated BP improved detection of hypertension compared with seated BP alone based on 24-hour SBP/DBP ≥ 125/75 mm Hg or daytime SBP/DBP ≥ 130/80 mm Hg (all BF > 3).
“In our hypertension clinic, we always measure both seated and standing BP in all of our patients,” John Giacona, PA-C, a PhD candidate at UT Southwestern Medical Center and coauthor of the study, told this news organization,
Multiple readings most important
Reached for comment, Johanna Contreras, MD, a cardiologist at Mount Sinai Hospital in New York, noted that diagnosing hypertension is “difficult” and she agrees that multiple readings are important.
“I usually take at least two readings in two different visits before I tell the patient they have high blood pressure,” Dr. Contreras said in an interview.
Dr. Contreras said she takes blood pressure both seated and standing.
“I’m not sure standing versus seated makes a big difference. However, when the patient first comes into the office, it is really important to let them rest and calm down before taking the blood pressure,” she said.
The study had no commercial funding. The authors and Dr. Contreras have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a new study suggests.
Combining three standing and three seated BP measurements in the same visit may lead to a “quicker diagnosis and save people a trip back to the office,” Wanpen Vongpatanasin, MD, professor of internal medicine and director of the hypertension section, cardiology division, UT Southwestern Medical Center, Dallas, said in an interview.
The study was presented at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Practice changing?
Clinical guidelines recommend office BP be taken in a seated position for most patients.
However, research has suggested that the sensitivity of seated office BP in diagnosing hypertension is about 50%, with high specificity around 90% during a single visit, Dr. Vongpatanasin explained.
At the follow-up visit, however, the second office BP yielded higher sensitivity to 80% but specificity fell to 55%. Nevertheless, the accuracy of standing BP measurements for diagnosing hypertension has not been investigated.
In a cross-sectional study, Dr. Vongpatanasin and colleagues determined the accuracy of both seated and standing BP for diagnosing hypertension in a single visit in 125 healthy adults who had not had a previous diagnosis of hypertension and were not taking any BP medications. The cohort had a mean age of 49 years, 62% were women, and 24% were Black.
During each office visit, seated BP was measured three times, then standing BP was measured three times using an automated and validated device.
Average seated BP was 123/76 mm Hg and average standing BP was 126/80 mm Hg.
Of the 125 participants, 42 (34%) had hypertension, defined as 24-hour ambulatory systolic/diastolic BP (SBP/DBP) of ≥ 125/75 mm Hg.
The sensitivity and specificity of seated SBP for hypertension was 43% and 92%, respectively.
“Interestingly, with standing SBP, sensitivity was improved to 74% and specificity dropped to 65% – which is okay; you will have to confirm a positive test anyway and when screening for a common disease you’d rather have a high sensitivity rather than low sensitivity to pick it up in this case,” Dr. Vongpatanasin said.
The area under receiver operating characteristic curve (AUROC) of standing SBP was significantly higher than seated SBP (Bayes factor [BF] = 11.8) when hypertension was defined as 24-hour SBP ≥ 125 mm Hg.
Similarly, when hypertension was defined as 24-hour DBP ≥ 75 mm Hg or daytime DBP ≥ 80 mm Hg, the AUROC of standing DBP was higher than seated DBP (all BF > 3).
The addition of standing to seated BP improved detection of hypertension compared with seated BP alone based on 24-hour SBP/DBP ≥ 125/75 mm Hg or daytime SBP/DBP ≥ 130/80 mm Hg (all BF > 3).
“In our hypertension clinic, we always measure both seated and standing BP in all of our patients,” John Giacona, PA-C, a PhD candidate at UT Southwestern Medical Center and coauthor of the study, told this news organization,
Multiple readings most important
Reached for comment, Johanna Contreras, MD, a cardiologist at Mount Sinai Hospital in New York, noted that diagnosing hypertension is “difficult” and she agrees that multiple readings are important.
“I usually take at least two readings in two different visits before I tell the patient they have high blood pressure,” Dr. Contreras said in an interview.
Dr. Contreras said she takes blood pressure both seated and standing.
“I’m not sure standing versus seated makes a big difference. However, when the patient first comes into the office, it is really important to let them rest and calm down before taking the blood pressure,” she said.
The study had no commercial funding. The authors and Dr. Contreras have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a new study suggests.
Combining three standing and three seated BP measurements in the same visit may lead to a “quicker diagnosis and save people a trip back to the office,” Wanpen Vongpatanasin, MD, professor of internal medicine and director of the hypertension section, cardiology division, UT Southwestern Medical Center, Dallas, said in an interview.
The study was presented at the Hypertension Scientific Sessions, sponsored by the American Heart Association.
Practice changing?
Clinical guidelines recommend office BP be taken in a seated position for most patients.
However, research has suggested that the sensitivity of seated office BP in diagnosing hypertension is about 50%, with high specificity around 90% during a single visit, Dr. Vongpatanasin explained.
At the follow-up visit, however, the second office BP yielded higher sensitivity to 80% but specificity fell to 55%. Nevertheless, the accuracy of standing BP measurements for diagnosing hypertension has not been investigated.
In a cross-sectional study, Dr. Vongpatanasin and colleagues determined the accuracy of both seated and standing BP for diagnosing hypertension in a single visit in 125 healthy adults who had not had a previous diagnosis of hypertension and were not taking any BP medications. The cohort had a mean age of 49 years, 62% were women, and 24% were Black.
During each office visit, seated BP was measured three times, then standing BP was measured three times using an automated and validated device.
Average seated BP was 123/76 mm Hg and average standing BP was 126/80 mm Hg.
Of the 125 participants, 42 (34%) had hypertension, defined as 24-hour ambulatory systolic/diastolic BP (SBP/DBP) of ≥ 125/75 mm Hg.
The sensitivity and specificity of seated SBP for hypertension was 43% and 92%, respectively.
“Interestingly, with standing SBP, sensitivity was improved to 74% and specificity dropped to 65% – which is okay; you will have to confirm a positive test anyway and when screening for a common disease you’d rather have a high sensitivity rather than low sensitivity to pick it up in this case,” Dr. Vongpatanasin said.
The area under receiver operating characteristic curve (AUROC) of standing SBP was significantly higher than seated SBP (Bayes factor [BF] = 11.8) when hypertension was defined as 24-hour SBP ≥ 125 mm Hg.
Similarly, when hypertension was defined as 24-hour DBP ≥ 75 mm Hg or daytime DBP ≥ 80 mm Hg, the AUROC of standing DBP was higher than seated DBP (all BF > 3).
The addition of standing to seated BP improved detection of hypertension compared with seated BP alone based on 24-hour SBP/DBP ≥ 125/75 mm Hg or daytime SBP/DBP ≥ 130/80 mm Hg (all BF > 3).
“In our hypertension clinic, we always measure both seated and standing BP in all of our patients,” John Giacona, PA-C, a PhD candidate at UT Southwestern Medical Center and coauthor of the study, told this news organization,
Multiple readings most important
Reached for comment, Johanna Contreras, MD, a cardiologist at Mount Sinai Hospital in New York, noted that diagnosing hypertension is “difficult” and she agrees that multiple readings are important.
“I usually take at least two readings in two different visits before I tell the patient they have high blood pressure,” Dr. Contreras said in an interview.
Dr. Contreras said she takes blood pressure both seated and standing.
“I’m not sure standing versus seated makes a big difference. However, when the patient first comes into the office, it is really important to let them rest and calm down before taking the blood pressure,” she said.
The study had no commercial funding. The authors and Dr. Contreras have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM HYPERTENSION 2022
Opioids after lung cancer surgery may up all-cause mortality risk
Patients who undergo lung cancer surgery and who receive long-term opioids for pain relief have an elevated risk of all-cause mortality at 2 years, a new study suggests. That risk was 40% higher than among patients who did not receive opioids.
“This is the first study to identify the association of new long-term opioid use with poorer long-term survival outcomes after lung cancer surgery using real-world data based on a national registration database,” said the authors, led by In-Ae Song, MD, Seoul National University Bundang Hospital, Seongnam, South Korea.
“New long-term opioid use may be associated with poor long-term survival outcomes, especially in potent opioid users,” they concluded.
Long-term opioid use might promote protumor activity secondary to immunosuppression along with migration of tumor cells and angiogenesis, the authors suggested.
The study was published online in Regional Anesthesia and Pain.
The finding comes from a study that used the South Korean National Health Insurance database as a nationwide registration data source. “All patients undergoing lung cancer surgery between 2011 and 2018 were included,” the authors noted.
In total, 54,509 patients were included in the final analysis. Six months after undergoing the procedure, 3,325 patients (6.1%) had been prescribed opioids continuously and regularly. These patients constituted the new long-term opioid user group.
This finding fits in with those from past studies that have suggested that new long-term postoperative pain is reported in 4%-12% of patients who undergo lung cancer surgeries, the authors commented.
The new study found that all-cause mortality at 2 years was significantly higher in the new long-term opioid user group than it was in the non–opioid user group (17.3% vs. 9.3%; P < .001).
Moreover, the new long-term opioid user group were at 43% higher risk of 2-year lung cancer mortality and 29% higher risk of 2-year non–lung cancer mortality.
The investigators divided the patients who had received long-term opioids into two subgroups – those who received more potent opioids (1.6%), and those who received less potent opioids (4.5%).
There was a big difference in the results for all-cause mortality.
Compared with nonopioid users, long-term use of less potent opioids was associated with a 2-year mortality risk of only 22% (P < .001), whereas the patients who used potent opioids were at a 92% increased risk of all-cause mortality.
A number of risk factors were associated with an increased rate of new long-term opioid use. These included older age, being male, length of stay in hospital, and comorbidities.
In addition, patients who were more likely to receive long-term opioids included those who had received neoadjuvant and adjuvant chemotherapy and those who had experienced preoperative anxiety disorder or insomnia disorder.
In contrast, patients who underwent video-assisted thoracoscopic surgery were less likely to receive long-term opioids, the authors noted.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients who undergo lung cancer surgery and who receive long-term opioids for pain relief have an elevated risk of all-cause mortality at 2 years, a new study suggests. That risk was 40% higher than among patients who did not receive opioids.
“This is the first study to identify the association of new long-term opioid use with poorer long-term survival outcomes after lung cancer surgery using real-world data based on a national registration database,” said the authors, led by In-Ae Song, MD, Seoul National University Bundang Hospital, Seongnam, South Korea.
“New long-term opioid use may be associated with poor long-term survival outcomes, especially in potent opioid users,” they concluded.
Long-term opioid use might promote protumor activity secondary to immunosuppression along with migration of tumor cells and angiogenesis, the authors suggested.
The study was published online in Regional Anesthesia and Pain.
The finding comes from a study that used the South Korean National Health Insurance database as a nationwide registration data source. “All patients undergoing lung cancer surgery between 2011 and 2018 were included,” the authors noted.
In total, 54,509 patients were included in the final analysis. Six months after undergoing the procedure, 3,325 patients (6.1%) had been prescribed opioids continuously and regularly. These patients constituted the new long-term opioid user group.
This finding fits in with those from past studies that have suggested that new long-term postoperative pain is reported in 4%-12% of patients who undergo lung cancer surgeries, the authors commented.
The new study found that all-cause mortality at 2 years was significantly higher in the new long-term opioid user group than it was in the non–opioid user group (17.3% vs. 9.3%; P < .001).
Moreover, the new long-term opioid user group were at 43% higher risk of 2-year lung cancer mortality and 29% higher risk of 2-year non–lung cancer mortality.
The investigators divided the patients who had received long-term opioids into two subgroups – those who received more potent opioids (1.6%), and those who received less potent opioids (4.5%).
There was a big difference in the results for all-cause mortality.
Compared with nonopioid users, long-term use of less potent opioids was associated with a 2-year mortality risk of only 22% (P < .001), whereas the patients who used potent opioids were at a 92% increased risk of all-cause mortality.
A number of risk factors were associated with an increased rate of new long-term opioid use. These included older age, being male, length of stay in hospital, and comorbidities.
In addition, patients who were more likely to receive long-term opioids included those who had received neoadjuvant and adjuvant chemotherapy and those who had experienced preoperative anxiety disorder or insomnia disorder.
In contrast, patients who underwent video-assisted thoracoscopic surgery were less likely to receive long-term opioids, the authors noted.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients who undergo lung cancer surgery and who receive long-term opioids for pain relief have an elevated risk of all-cause mortality at 2 years, a new study suggests. That risk was 40% higher than among patients who did not receive opioids.
“This is the first study to identify the association of new long-term opioid use with poorer long-term survival outcomes after lung cancer surgery using real-world data based on a national registration database,” said the authors, led by In-Ae Song, MD, Seoul National University Bundang Hospital, Seongnam, South Korea.
“New long-term opioid use may be associated with poor long-term survival outcomes, especially in potent opioid users,” they concluded.
Long-term opioid use might promote protumor activity secondary to immunosuppression along with migration of tumor cells and angiogenesis, the authors suggested.
The study was published online in Regional Anesthesia and Pain.
The finding comes from a study that used the South Korean National Health Insurance database as a nationwide registration data source. “All patients undergoing lung cancer surgery between 2011 and 2018 were included,” the authors noted.
In total, 54,509 patients were included in the final analysis. Six months after undergoing the procedure, 3,325 patients (6.1%) had been prescribed opioids continuously and regularly. These patients constituted the new long-term opioid user group.
This finding fits in with those from past studies that have suggested that new long-term postoperative pain is reported in 4%-12% of patients who undergo lung cancer surgeries, the authors commented.
The new study found that all-cause mortality at 2 years was significantly higher in the new long-term opioid user group than it was in the non–opioid user group (17.3% vs. 9.3%; P < .001).
Moreover, the new long-term opioid user group were at 43% higher risk of 2-year lung cancer mortality and 29% higher risk of 2-year non–lung cancer mortality.
The investigators divided the patients who had received long-term opioids into two subgroups – those who received more potent opioids (1.6%), and those who received less potent opioids (4.5%).
There was a big difference in the results for all-cause mortality.
Compared with nonopioid users, long-term use of less potent opioids was associated with a 2-year mortality risk of only 22% (P < .001), whereas the patients who used potent opioids were at a 92% increased risk of all-cause mortality.
A number of risk factors were associated with an increased rate of new long-term opioid use. These included older age, being male, length of stay in hospital, and comorbidities.
In addition, patients who were more likely to receive long-term opioids included those who had received neoadjuvant and adjuvant chemotherapy and those who had experienced preoperative anxiety disorder or insomnia disorder.
In contrast, patients who underwent video-assisted thoracoscopic surgery were less likely to receive long-term opioids, the authors noted.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM REGIONAL ANESTHESIA AND PAIN
People of color bearing brunt of long COVID, doctors say
From the earliest days of the COVID-19 pandemic, people of color have been hardest hit by the virus. Now, many doctors and researchers are seeing big disparities come about in who gets care for long COVID.
Long COVID can affect patients from all walks of life. said Alba Miranda Azola, MD, codirector of the post–acute COVID-19 team at Johns Hopkins University, Baltimore.
Non-White patients are more apt to lack access to primary care, face insurance barriers to see specialists, struggle with time off work or transportation for appointments, and have financial barriers to care as copayments for therapy pile up.
“We are getting a very skewed population of Caucasian wealthy people who are coming to our clinic because they have the ability to access care, they have good insurance, and they are looking on the internet and find us,” Dr. Azola said.
This mix of patients at Dr. Azola’s clinic is out of step with the demographics of Baltimore, where the majority of residents are Black, half of them earn less than $52,000 a year, and one in five live in poverty. And this isn’t unique to Hopkins. Many of the dozens of specialized long COVID clinics that have cropped up around the country are also seeing an unequal share of affluent White patients, experts say.
It’s also a patient mix that very likely doesn’t reflect who is most apt to have long COVID.
During the pandemic, people who identified as Black, Hispanic, American Indian, or Alaska Native were more likely to be diagnosed with COVID than people who identified as White, according to the Centers for Disease Control and Prevention. These people of color were also at least twice as likely to be hospitalized with severe infections, and at least 70% more likely to die.
“Data repeatedly show the disproportionate impact of COVID-19 on racial and ethnic minority populations, as well as other population groups such as people living in rural or frontier areas, people experiencing homelessness, essential and frontline workers, people with disabilities, people with substance use disorders, people who are incarcerated, and non–U.S.-born persons,” John Brooks, MD, chief medical officer for COVID-19 response at the CDC, said during testimony before the U.S. House Energy and Commerce Subcommittee on Health in April 2021.
“While we do not yet have clear data on the impact of post-COVID conditions on racial and ethnic minority populations and other disadvantaged communities, we do believe that they are likely to be disproportionately impacted ... and less likely to be able to access health care services,” Dr. Brooks said at the time.
The picture that’s emerging of long COVID suggests that the condition impacts about one in five adults. It’s more common among Hispanic adults than among people who identify as Black, Asian, or White. It’s also more common among those who identify as other races or multiple races, according survey data collected by the CDC.
It’s hard to say how accurate this snapshot is because researchers need to do a better job of identifying and following people with long COVID, said Monica Verduzco-Gutierrez, MD, chair of rehabilitation medicine and director of the COVID-19 Recovery Clinic at the University of Texas Health Science Center at San Antonio. A major limitation of surveys like the ones done by the CDC to monitor long COVID is that only people who realize they have the condition can get counted.
“Some people from historically marginalized groups may have less health literacy to know about impacts of long COVID,” she said.
Lack of awareness may keep people with persistent symptoms from seeking medical attention, leaving many long COVID cases undiagnosed.
When some patients do seek help, their complaints may not be acknowledged or understood. Often, cultural bias or structural racism can get in the way of diagnosis and treatment, Dr. Azola said.
“I hate to say this, but there is probably bias among providers,” she said. “For example, I am Puerto Rican, and the way we describe symptoms as Latinos may sound exaggerated or may be brushed aside or lost in translation. I think we miss a lot of patients being diagnosed or referred to specialists because the primary care provider they see maybe leans into this cultural bias of thinking this is just a Latino being dramatic.”
There’s some evidence that treatment for long COVID may differ by race even when symptoms are similar. One study of more than 400,000 patients, for example, found no racial differences in the proportion of people who have six common long COVID symptoms: shortness of breath, fatigue, weakness, pain, trouble with thinking skills, and a hard time getting around. Despite this, Black patients were significantly less likely to receive outpatient rehabilitation services to treat these symptoms.
Benjamin Abramoff, MD, who leads the long COVID collaborative for the American Academy of Physical Medicine and Rehabilitation, draws parallels between what happens with long COVID to another common health problem often undertreated among patients of color: pain. With both long COVID and chronic pain, one major barrier to care is “just getting taken seriously by providers,” he said.
“There is significant evidence that racial bias has led to less prescription of pain medications to people of color,” Dr. Abramoff said. “Just as pain can be difficult to get objective measures of, long COVID symptoms can also be difficult to objectively measure and requires trust between the provider and patient.”
Geography can be another barrier to care, said Aaron Friedberg, MD, clinical colead of the post-COVID recovery program at Ohio State University Wexner Medical Center, Columbus. Many communities hardest hit by COVID – particularly in high-poverty urban neighborhoods – have long had limited access to care. The pandemic worsened staffing shortages at many hospitals and clinics in these communities, leaving patients even fewer options close to home.
“I often have patients driving several hours to come to our clinic, and that can create significant challenges both because of the financial burden and time required to coordinate that type of travel, but also because post-COVID symptoms can make it extremely challenging to tolerate that type of travel,” Dr. Friedberg said.
Even though the complete picture of who has long COVID – and who’s getting treated and getting good outcomes – is still emerging, it’s very clear at this point in the pandemic that access isn’t equal among everyone and that many low-income and non-White patients are missing out on needed treatments, Friedberg said.
“One thing that is clear is that there are many people suffering alone from these conditions,” he said.
A version of this article first appeared on WebMD.com.
From the earliest days of the COVID-19 pandemic, people of color have been hardest hit by the virus. Now, many doctors and researchers are seeing big disparities come about in who gets care for long COVID.
Long COVID can affect patients from all walks of life. said Alba Miranda Azola, MD, codirector of the post–acute COVID-19 team at Johns Hopkins University, Baltimore.
Non-White patients are more apt to lack access to primary care, face insurance barriers to see specialists, struggle with time off work or transportation for appointments, and have financial barriers to care as copayments for therapy pile up.
“We are getting a very skewed population of Caucasian wealthy people who are coming to our clinic because they have the ability to access care, they have good insurance, and they are looking on the internet and find us,” Dr. Azola said.
This mix of patients at Dr. Azola’s clinic is out of step with the demographics of Baltimore, where the majority of residents are Black, half of them earn less than $52,000 a year, and one in five live in poverty. And this isn’t unique to Hopkins. Many of the dozens of specialized long COVID clinics that have cropped up around the country are also seeing an unequal share of affluent White patients, experts say.
It’s also a patient mix that very likely doesn’t reflect who is most apt to have long COVID.
During the pandemic, people who identified as Black, Hispanic, American Indian, or Alaska Native were more likely to be diagnosed with COVID than people who identified as White, according to the Centers for Disease Control and Prevention. These people of color were also at least twice as likely to be hospitalized with severe infections, and at least 70% more likely to die.
“Data repeatedly show the disproportionate impact of COVID-19 on racial and ethnic minority populations, as well as other population groups such as people living in rural or frontier areas, people experiencing homelessness, essential and frontline workers, people with disabilities, people with substance use disorders, people who are incarcerated, and non–U.S.-born persons,” John Brooks, MD, chief medical officer for COVID-19 response at the CDC, said during testimony before the U.S. House Energy and Commerce Subcommittee on Health in April 2021.
“While we do not yet have clear data on the impact of post-COVID conditions on racial and ethnic minority populations and other disadvantaged communities, we do believe that they are likely to be disproportionately impacted ... and less likely to be able to access health care services,” Dr. Brooks said at the time.
The picture that’s emerging of long COVID suggests that the condition impacts about one in five adults. It’s more common among Hispanic adults than among people who identify as Black, Asian, or White. It’s also more common among those who identify as other races or multiple races, according survey data collected by the CDC.
It’s hard to say how accurate this snapshot is because researchers need to do a better job of identifying and following people with long COVID, said Monica Verduzco-Gutierrez, MD, chair of rehabilitation medicine and director of the COVID-19 Recovery Clinic at the University of Texas Health Science Center at San Antonio. A major limitation of surveys like the ones done by the CDC to monitor long COVID is that only people who realize they have the condition can get counted.
“Some people from historically marginalized groups may have less health literacy to know about impacts of long COVID,” she said.
Lack of awareness may keep people with persistent symptoms from seeking medical attention, leaving many long COVID cases undiagnosed.
When some patients do seek help, their complaints may not be acknowledged or understood. Often, cultural bias or structural racism can get in the way of diagnosis and treatment, Dr. Azola said.
“I hate to say this, but there is probably bias among providers,” she said. “For example, I am Puerto Rican, and the way we describe symptoms as Latinos may sound exaggerated or may be brushed aside or lost in translation. I think we miss a lot of patients being diagnosed or referred to specialists because the primary care provider they see maybe leans into this cultural bias of thinking this is just a Latino being dramatic.”
There’s some evidence that treatment for long COVID may differ by race even when symptoms are similar. One study of more than 400,000 patients, for example, found no racial differences in the proportion of people who have six common long COVID symptoms: shortness of breath, fatigue, weakness, pain, trouble with thinking skills, and a hard time getting around. Despite this, Black patients were significantly less likely to receive outpatient rehabilitation services to treat these symptoms.
Benjamin Abramoff, MD, who leads the long COVID collaborative for the American Academy of Physical Medicine and Rehabilitation, draws parallels between what happens with long COVID to another common health problem often undertreated among patients of color: pain. With both long COVID and chronic pain, one major barrier to care is “just getting taken seriously by providers,” he said.
“There is significant evidence that racial bias has led to less prescription of pain medications to people of color,” Dr. Abramoff said. “Just as pain can be difficult to get objective measures of, long COVID symptoms can also be difficult to objectively measure and requires trust between the provider and patient.”
Geography can be another barrier to care, said Aaron Friedberg, MD, clinical colead of the post-COVID recovery program at Ohio State University Wexner Medical Center, Columbus. Many communities hardest hit by COVID – particularly in high-poverty urban neighborhoods – have long had limited access to care. The pandemic worsened staffing shortages at many hospitals and clinics in these communities, leaving patients even fewer options close to home.
“I often have patients driving several hours to come to our clinic, and that can create significant challenges both because of the financial burden and time required to coordinate that type of travel, but also because post-COVID symptoms can make it extremely challenging to tolerate that type of travel,” Dr. Friedberg said.
Even though the complete picture of who has long COVID – and who’s getting treated and getting good outcomes – is still emerging, it’s very clear at this point in the pandemic that access isn’t equal among everyone and that many low-income and non-White patients are missing out on needed treatments, Friedberg said.
“One thing that is clear is that there are many people suffering alone from these conditions,” he said.
A version of this article first appeared on WebMD.com.
From the earliest days of the COVID-19 pandemic, people of color have been hardest hit by the virus. Now, many doctors and researchers are seeing big disparities come about in who gets care for long COVID.
Long COVID can affect patients from all walks of life. said Alba Miranda Azola, MD, codirector of the post–acute COVID-19 team at Johns Hopkins University, Baltimore.
Non-White patients are more apt to lack access to primary care, face insurance barriers to see specialists, struggle with time off work or transportation for appointments, and have financial barriers to care as copayments for therapy pile up.
“We are getting a very skewed population of Caucasian wealthy people who are coming to our clinic because they have the ability to access care, they have good insurance, and they are looking on the internet and find us,” Dr. Azola said.
This mix of patients at Dr. Azola’s clinic is out of step with the demographics of Baltimore, where the majority of residents are Black, half of them earn less than $52,000 a year, and one in five live in poverty. And this isn’t unique to Hopkins. Many of the dozens of specialized long COVID clinics that have cropped up around the country are also seeing an unequal share of affluent White patients, experts say.
It’s also a patient mix that very likely doesn’t reflect who is most apt to have long COVID.
During the pandemic, people who identified as Black, Hispanic, American Indian, or Alaska Native were more likely to be diagnosed with COVID than people who identified as White, according to the Centers for Disease Control and Prevention. These people of color were also at least twice as likely to be hospitalized with severe infections, and at least 70% more likely to die.
“Data repeatedly show the disproportionate impact of COVID-19 on racial and ethnic minority populations, as well as other population groups such as people living in rural or frontier areas, people experiencing homelessness, essential and frontline workers, people with disabilities, people with substance use disorders, people who are incarcerated, and non–U.S.-born persons,” John Brooks, MD, chief medical officer for COVID-19 response at the CDC, said during testimony before the U.S. House Energy and Commerce Subcommittee on Health in April 2021.
“While we do not yet have clear data on the impact of post-COVID conditions on racial and ethnic minority populations and other disadvantaged communities, we do believe that they are likely to be disproportionately impacted ... and less likely to be able to access health care services,” Dr. Brooks said at the time.
The picture that’s emerging of long COVID suggests that the condition impacts about one in five adults. It’s more common among Hispanic adults than among people who identify as Black, Asian, or White. It’s also more common among those who identify as other races or multiple races, according survey data collected by the CDC.
It’s hard to say how accurate this snapshot is because researchers need to do a better job of identifying and following people with long COVID, said Monica Verduzco-Gutierrez, MD, chair of rehabilitation medicine and director of the COVID-19 Recovery Clinic at the University of Texas Health Science Center at San Antonio. A major limitation of surveys like the ones done by the CDC to monitor long COVID is that only people who realize they have the condition can get counted.
“Some people from historically marginalized groups may have less health literacy to know about impacts of long COVID,” she said.
Lack of awareness may keep people with persistent symptoms from seeking medical attention, leaving many long COVID cases undiagnosed.
When some patients do seek help, their complaints may not be acknowledged or understood. Often, cultural bias or structural racism can get in the way of diagnosis and treatment, Dr. Azola said.
“I hate to say this, but there is probably bias among providers,” she said. “For example, I am Puerto Rican, and the way we describe symptoms as Latinos may sound exaggerated or may be brushed aside or lost in translation. I think we miss a lot of patients being diagnosed or referred to specialists because the primary care provider they see maybe leans into this cultural bias of thinking this is just a Latino being dramatic.”
There’s some evidence that treatment for long COVID may differ by race even when symptoms are similar. One study of more than 400,000 patients, for example, found no racial differences in the proportion of people who have six common long COVID symptoms: shortness of breath, fatigue, weakness, pain, trouble with thinking skills, and a hard time getting around. Despite this, Black patients were significantly less likely to receive outpatient rehabilitation services to treat these symptoms.
Benjamin Abramoff, MD, who leads the long COVID collaborative for the American Academy of Physical Medicine and Rehabilitation, draws parallels between what happens with long COVID to another common health problem often undertreated among patients of color: pain. With both long COVID and chronic pain, one major barrier to care is “just getting taken seriously by providers,” he said.
“There is significant evidence that racial bias has led to less prescription of pain medications to people of color,” Dr. Abramoff said. “Just as pain can be difficult to get objective measures of, long COVID symptoms can also be difficult to objectively measure and requires trust between the provider and patient.”
Geography can be another barrier to care, said Aaron Friedberg, MD, clinical colead of the post-COVID recovery program at Ohio State University Wexner Medical Center, Columbus. Many communities hardest hit by COVID – particularly in high-poverty urban neighborhoods – have long had limited access to care. The pandemic worsened staffing shortages at many hospitals and clinics in these communities, leaving patients even fewer options close to home.
“I often have patients driving several hours to come to our clinic, and that can create significant challenges both because of the financial burden and time required to coordinate that type of travel, but also because post-COVID symptoms can make it extremely challenging to tolerate that type of travel,” Dr. Friedberg said.
Even though the complete picture of who has long COVID – and who’s getting treated and getting good outcomes – is still emerging, it’s very clear at this point in the pandemic that access isn’t equal among everyone and that many low-income and non-White patients are missing out on needed treatments, Friedberg said.
“One thing that is clear is that there are many people suffering alone from these conditions,” he said.
A version of this article first appeared on WebMD.com.
Annual screening benefits people at high-risk for pancreatic cancer
Two prospective studies that followed individuals with genetic or familiar risk for pancreatic cancer found that patients whose cancer was found through screening had better overall survival than those diagnosed symptomatically, suggesting that surveillance in this population may improve outcomes.
Opponents of screening for pancreatic cancer in this population suggested that screening may identify cancers at an earlier stage but doesn’t necessarily improve outcomes – an effect referred to as lead-time bias. “The data from these studies strongly refute this, now showing substantially better outcomes for those with screen-detected pancreatic cancer. In other words, these data now strongly suggest that, for some cases, earlier detection truly leads to superior outcomes, including what appears to be substantially higher likelihood of long-term cure,” said Matthew B. Yurgelun, MD, who wrote an accompanying editorial that was published in the Journal of Clinical Oncology, in response to two studies published in June in the journal.
“Individuals with genetic or familial risk of pancreatic cancer who develop screen-detected pancreatic cancer, as opposed to pancreatic cancer detected due to symptoms and in the absence of screening, appear to have significantly better outcomes compared to historical pancreatic cancer outcomes data,” said Dr. Yurgelun, who is an assistant professor of medicine at Harvard Medical School and director of the Dana-Farber Cancer Institute Lynch Syndrome Center, Boston.
In one report, researchers reported outcomes from 1,461 genetic high-risk participants in Cancer of Pancreas Screening-5 (CAPS5). 48.5% had a pathogenic version of a PDAC-susceptibility gene. 10 individuals were diagnosed with PDAC, 9 during surveillance, and 1 after having dropped out of the surveillance program for 4 years. 7 of the 9 surveillance cancers were stage 1, 1 was stage 2, and 1 was stage 3. 8 of 9 were resectable. The cancer found outside of surveillance was metastatic. In the overall cohort of 1,731 individuals, there were 19 PDACs detected during surveillance and 7 diagnosed outside of surveillance. 57.9% of surveillance-discovered were stage 1 and 5.2% were stage 4. On the other hand, 6 of the 7 tumors found outside of surveillance were stage 4. Median survival was 9.8 years in the screen-detected group versus 1.5 years outside surveillance (hazard ratio, 0.13; P = .003).
In a second study, researchers followed 47 individuals with inherited pathogenic variants for a median of 56 years. 8.9% were diagnosed with PDAC over a median follow-up of 5.6 years. By age 70, 20.7% had been diagnosed with PDAC. 83.3% of cases were identified as resectable at imaging, and 71.0% underwent resection. 33.3% of cases were stage I. Following primary PDAC diagnosis, the median survival was 26.8 months. 5-year survival was 32.4% (95% confidence interval; 19.1-54.8%). Among those who underwent rsection, the 5-year survival was 44.1% (95% CI, 27.2-71.3%). 2.6% of the population underwent surgery to remove a suspected malignant lesion which turned out not to be PDAC. 5 had low-grade dysplasia.
The results reinforce existing clinical practice guidelines that suggest MRI or endoscopic ultrasound screening for individuals with both a family and genetic risk of PDAC and could inform deliberations about extending such screening to all patients with inherited risk factors, according to Dr. Yurgelun.
Screening does not come without drawbacks. Research has demonstrated that about 40% of individuals who present for high-risk screening have pancreatic abnormalities, typically cystic lesions. These are also common in the general population. Very few such lesions require intervention, but they should be monitored, according to Dr. Yurgelun. Biopsies can be tricky, and removal requires major abdominal surgery, and magnetic resonance imaging or endoscopic ultrasonic can be nuanced, “which highlights the importance of this screening being performed and interpreted by health care providers experienced in high-risk surveillance,” Dr. Yurgelun said.
Dr. Yurgelun has no relevant financial disclosures.
Two prospective studies that followed individuals with genetic or familiar risk for pancreatic cancer found that patients whose cancer was found through screening had better overall survival than those diagnosed symptomatically, suggesting that surveillance in this population may improve outcomes.
Opponents of screening for pancreatic cancer in this population suggested that screening may identify cancers at an earlier stage but doesn’t necessarily improve outcomes – an effect referred to as lead-time bias. “The data from these studies strongly refute this, now showing substantially better outcomes for those with screen-detected pancreatic cancer. In other words, these data now strongly suggest that, for some cases, earlier detection truly leads to superior outcomes, including what appears to be substantially higher likelihood of long-term cure,” said Matthew B. Yurgelun, MD, who wrote an accompanying editorial that was published in the Journal of Clinical Oncology, in response to two studies published in June in the journal.
“Individuals with genetic or familial risk of pancreatic cancer who develop screen-detected pancreatic cancer, as opposed to pancreatic cancer detected due to symptoms and in the absence of screening, appear to have significantly better outcomes compared to historical pancreatic cancer outcomes data,” said Dr. Yurgelun, who is an assistant professor of medicine at Harvard Medical School and director of the Dana-Farber Cancer Institute Lynch Syndrome Center, Boston.
In one report, researchers reported outcomes from 1,461 genetic high-risk participants in Cancer of Pancreas Screening-5 (CAPS5). 48.5% had a pathogenic version of a PDAC-susceptibility gene. 10 individuals were diagnosed with PDAC, 9 during surveillance, and 1 after having dropped out of the surveillance program for 4 years. 7 of the 9 surveillance cancers were stage 1, 1 was stage 2, and 1 was stage 3. 8 of 9 were resectable. The cancer found outside of surveillance was metastatic. In the overall cohort of 1,731 individuals, there were 19 PDACs detected during surveillance and 7 diagnosed outside of surveillance. 57.9% of surveillance-discovered were stage 1 and 5.2% were stage 4. On the other hand, 6 of the 7 tumors found outside of surveillance were stage 4. Median survival was 9.8 years in the screen-detected group versus 1.5 years outside surveillance (hazard ratio, 0.13; P = .003).
In a second study, researchers followed 47 individuals with inherited pathogenic variants for a median of 56 years. 8.9% were diagnosed with PDAC over a median follow-up of 5.6 years. By age 70, 20.7% had been diagnosed with PDAC. 83.3% of cases were identified as resectable at imaging, and 71.0% underwent resection. 33.3% of cases were stage I. Following primary PDAC diagnosis, the median survival was 26.8 months. 5-year survival was 32.4% (95% confidence interval; 19.1-54.8%). Among those who underwent rsection, the 5-year survival was 44.1% (95% CI, 27.2-71.3%). 2.6% of the population underwent surgery to remove a suspected malignant lesion which turned out not to be PDAC. 5 had low-grade dysplasia.
The results reinforce existing clinical practice guidelines that suggest MRI or endoscopic ultrasound screening for individuals with both a family and genetic risk of PDAC and could inform deliberations about extending such screening to all patients with inherited risk factors, according to Dr. Yurgelun.
Screening does not come without drawbacks. Research has demonstrated that about 40% of individuals who present for high-risk screening have pancreatic abnormalities, typically cystic lesions. These are also common in the general population. Very few such lesions require intervention, but they should be monitored, according to Dr. Yurgelun. Biopsies can be tricky, and removal requires major abdominal surgery, and magnetic resonance imaging or endoscopic ultrasonic can be nuanced, “which highlights the importance of this screening being performed and interpreted by health care providers experienced in high-risk surveillance,” Dr. Yurgelun said.
Dr. Yurgelun has no relevant financial disclosures.
Two prospective studies that followed individuals with genetic or familiar risk for pancreatic cancer found that patients whose cancer was found through screening had better overall survival than those diagnosed symptomatically, suggesting that surveillance in this population may improve outcomes.
Opponents of screening for pancreatic cancer in this population suggested that screening may identify cancers at an earlier stage but doesn’t necessarily improve outcomes – an effect referred to as lead-time bias. “The data from these studies strongly refute this, now showing substantially better outcomes for those with screen-detected pancreatic cancer. In other words, these data now strongly suggest that, for some cases, earlier detection truly leads to superior outcomes, including what appears to be substantially higher likelihood of long-term cure,” said Matthew B. Yurgelun, MD, who wrote an accompanying editorial that was published in the Journal of Clinical Oncology, in response to two studies published in June in the journal.
“Individuals with genetic or familial risk of pancreatic cancer who develop screen-detected pancreatic cancer, as opposed to pancreatic cancer detected due to symptoms and in the absence of screening, appear to have significantly better outcomes compared to historical pancreatic cancer outcomes data,” said Dr. Yurgelun, who is an assistant professor of medicine at Harvard Medical School and director of the Dana-Farber Cancer Institute Lynch Syndrome Center, Boston.
In one report, researchers reported outcomes from 1,461 genetic high-risk participants in Cancer of Pancreas Screening-5 (CAPS5). 48.5% had a pathogenic version of a PDAC-susceptibility gene. 10 individuals were diagnosed with PDAC, 9 during surveillance, and 1 after having dropped out of the surveillance program for 4 years. 7 of the 9 surveillance cancers were stage 1, 1 was stage 2, and 1 was stage 3. 8 of 9 were resectable. The cancer found outside of surveillance was metastatic. In the overall cohort of 1,731 individuals, there were 19 PDACs detected during surveillance and 7 diagnosed outside of surveillance. 57.9% of surveillance-discovered were stage 1 and 5.2% were stage 4. On the other hand, 6 of the 7 tumors found outside of surveillance were stage 4. Median survival was 9.8 years in the screen-detected group versus 1.5 years outside surveillance (hazard ratio, 0.13; P = .003).
In a second study, researchers followed 47 individuals with inherited pathogenic variants for a median of 56 years. 8.9% were diagnosed with PDAC over a median follow-up of 5.6 years. By age 70, 20.7% had been diagnosed with PDAC. 83.3% of cases were identified as resectable at imaging, and 71.0% underwent resection. 33.3% of cases were stage I. Following primary PDAC diagnosis, the median survival was 26.8 months. 5-year survival was 32.4% (95% confidence interval; 19.1-54.8%). Among those who underwent rsection, the 5-year survival was 44.1% (95% CI, 27.2-71.3%). 2.6% of the population underwent surgery to remove a suspected malignant lesion which turned out not to be PDAC. 5 had low-grade dysplasia.
The results reinforce existing clinical practice guidelines that suggest MRI or endoscopic ultrasound screening for individuals with both a family and genetic risk of PDAC and could inform deliberations about extending such screening to all patients with inherited risk factors, according to Dr. Yurgelun.
Screening does not come without drawbacks. Research has demonstrated that about 40% of individuals who present for high-risk screening have pancreatic abnormalities, typically cystic lesions. These are also common in the general population. Very few such lesions require intervention, but they should be monitored, according to Dr. Yurgelun. Biopsies can be tricky, and removal requires major abdominal surgery, and magnetic resonance imaging or endoscopic ultrasonic can be nuanced, “which highlights the importance of this screening being performed and interpreted by health care providers experienced in high-risk surveillance,” Dr. Yurgelun said.
Dr. Yurgelun has no relevant financial disclosures.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Not just what, but when: Neoadjuvant pembrolizumab in melanoma
PARIS – “It’s not just what you give, it’s when you give it,” said the investigator reporting “that the same treatment for resectable melanoma given in a different sequence can generate lower rates of melanoma recurrence.”
Sapna Patel, MD, associate professor of melanoma medical oncology at The University of Texas MD Anderson Cancer Center, Houston, reported the results from the SWOG S1801 trial, which showed that than patients who received pembrolizumab after surgery only.
At a median follow-up of almost 15 months, there was a 42% lower rate of recurrence or death.
“Compared to the same treatment given entirely in the adjuvant setting, neoadjuvant pembrolizumab followed by adjuvant pembrolizumab improves event-free survival in resectable melanoma,” Dr. Patel commented.
She suggested that the explanation for the findings was that “inhibiting the PD-1/PD-L1 immune checkpoints before surgery gives an antitumor response at local and distant sites, and this occurs before resection of the tumor bed. This approach tends to leave behind a larger number of anti-tumor T cells ... [and] these T cells can be activated and circulated systematically to recognize and attack micro-metastatic melanoma tumors.”
The findings were presented during a presidential symposium at the European Society for Medical Oncology (ESMO) Congress 2022, Paris.
“This trial provides us with more evidence of when one strategy may be preferred over the other,” commented Maya Dimitrova, MD, medical oncologist at NYU Langone Perlmutter Cancer Center. She was not involved with the trial.
“Neoadjuvant immunotherapy has elicited impressive complete pathologic responses, which thus far have proven to be associated with a durable response. Neoadjuvant therapy may help identify patients who will respond well to checkpoint inhibitors and allow for de-escalation of therapy,” she told this news organization when approached for comment.
“As with all neoadjuvant therapy, we don’t want the treatment to compromise the outcomes of surgery when the intent is curative, and we once again have evidence that this is not the case when it comes to immune therapy,” she said. However, she added that “we will need further survival data to really change the standard of practice in high-risk melanoma and demonstrate whether there is a superior sequence of therapy and surgery.”
Details of the new results
The S1801 clinical trial enrolled 345 participants with stage IIIB through stage IV melanoma considered resectable. The cohort was randomized to receive either upfront surgery followed by 18 doses of pembrolizumab 200 mg every 3 weeks for a total of 18 doses or neoadjuvant therapy with pembrolizumab 200 mg (3 doses) followed by 15 doses of adjuvant pembrolizumab.
The primary endpoint was event-free survival (EFS), defined as the time from randomization to the occurrence of one of the following: disease progression or toxicity that resulted in not receiving surgery, failure to begin adjuvant therapy within 84 days of surgery, melanoma recurrence after surgery, or death from any cause.
At a median follow-up of 14.7 months, EFS was significantly higher for patients in the neoadjuvant group, compared with those receiving adjuvant therapy only (HR, 0.58; one-sided log-rank P = .004). A total of 36 participants died in the neoadjuvant and adjuvant groups (14 and 22 patients, extrapolating to a hazard ratio of 0.63; one-sided P = .091).
“With a limited number of events, overall survival is not statistically different at this time,” Dr. Patel said. “Landmark 2-year survival was 72% in the neoadjuvant arm and 49% in the adjuvant arm.”
The authors note that the benefit of neoadjuvant therapy remained consistent across a range of factors, including patient age, sex, performance status, stage of disease, ulceration, and BRAF status. The same proportion of patients in both groups received adjuvant pembrolizumab following surgery.
Rates of adverse events were similar in both groups, and neoadjuvant pembrolizumab did not result in an increase in adverse events related to surgery. In the neoadjuvant group, 28 patients (21%) with submitted pathology reports were noted to have had a complete pathologic response (0% viable tumor) on local review.
Questions remain
Invited discussant James Larkin, PhD, FRCP, FMedSci, a clinical researcher at The Royal Marsden Hospital, London, noted that the study had “striking results” and was a landmark trial with a simple but powerful design.
However, he pointed to some questions which need to be addressed in the future. “One important question is what is the optimal duration of neoadjuvant treatment, and can we individualize it?”
Another question is just how much postoperative treatment is really needed and whether pathology help determine that. “Can surgery be safely avoided altogether?” he asked. “Another issue is the need for anti-CTL4 therapy – which patients might benefit from anti-CTL4, in addition to anti-PD-1?”
“And by extension, this paradigm provides a great platform for testing new agents, including combinations in cases where PD-1 is not sufficient to achieve a sufficient response,” said Dr. Larkin. “In the future, trials addressing these questions hand us a major opportunity to individualize and rationally de-escalate treatment.”
Also weighing in on the study, another expert pointed out that neoadjuvant therapy in this setting is already being considered as an option. “The use of immunotherapy before surgery has been reported in some trials such as the OPACIN-neo and PRADO trials,” said Anthony J. Olszanski, RPh, MD, Vice Chair of Research at the Fox Chase Cancer Center, Philadelphia. “Results have been quite exciting and have led the NCCN to list this as a potential option for some patients in the current melanoma guidelines.”
S1801 is funded by the NIH/NCI and in part by MSD through a Cooperative Research and Development Agreement with the NCI. Pembrolizumab (KEYTRUDA) is Merck’s anti-PD-1 therapy. Dr. Patel has declared multiple relationships with industry as noted in the abstract; several co-authors have also made disclosures. Dr. Olszanski has reported participating in advisory boards for BMS, Merck, and InstilBio and running trials for them.
A version of this article first appeared on Medscape.com.
PARIS – “It’s not just what you give, it’s when you give it,” said the investigator reporting “that the same treatment for resectable melanoma given in a different sequence can generate lower rates of melanoma recurrence.”
Sapna Patel, MD, associate professor of melanoma medical oncology at The University of Texas MD Anderson Cancer Center, Houston, reported the results from the SWOG S1801 trial, which showed that than patients who received pembrolizumab after surgery only.
At a median follow-up of almost 15 months, there was a 42% lower rate of recurrence or death.
“Compared to the same treatment given entirely in the adjuvant setting, neoadjuvant pembrolizumab followed by adjuvant pembrolizumab improves event-free survival in resectable melanoma,” Dr. Patel commented.
She suggested that the explanation for the findings was that “inhibiting the PD-1/PD-L1 immune checkpoints before surgery gives an antitumor response at local and distant sites, and this occurs before resection of the tumor bed. This approach tends to leave behind a larger number of anti-tumor T cells ... [and] these T cells can be activated and circulated systematically to recognize and attack micro-metastatic melanoma tumors.”
The findings were presented during a presidential symposium at the European Society for Medical Oncology (ESMO) Congress 2022, Paris.
“This trial provides us with more evidence of when one strategy may be preferred over the other,” commented Maya Dimitrova, MD, medical oncologist at NYU Langone Perlmutter Cancer Center. She was not involved with the trial.
“Neoadjuvant immunotherapy has elicited impressive complete pathologic responses, which thus far have proven to be associated with a durable response. Neoadjuvant therapy may help identify patients who will respond well to checkpoint inhibitors and allow for de-escalation of therapy,” she told this news organization when approached for comment.
“As with all neoadjuvant therapy, we don’t want the treatment to compromise the outcomes of surgery when the intent is curative, and we once again have evidence that this is not the case when it comes to immune therapy,” she said. However, she added that “we will need further survival data to really change the standard of practice in high-risk melanoma and demonstrate whether there is a superior sequence of therapy and surgery.”
Details of the new results
The S1801 clinical trial enrolled 345 participants with stage IIIB through stage IV melanoma considered resectable. The cohort was randomized to receive either upfront surgery followed by 18 doses of pembrolizumab 200 mg every 3 weeks for a total of 18 doses or neoadjuvant therapy with pembrolizumab 200 mg (3 doses) followed by 15 doses of adjuvant pembrolizumab.
The primary endpoint was event-free survival (EFS), defined as the time from randomization to the occurrence of one of the following: disease progression or toxicity that resulted in not receiving surgery, failure to begin adjuvant therapy within 84 days of surgery, melanoma recurrence after surgery, or death from any cause.
At a median follow-up of 14.7 months, EFS was significantly higher for patients in the neoadjuvant group, compared with those receiving adjuvant therapy only (HR, 0.58; one-sided log-rank P = .004). A total of 36 participants died in the neoadjuvant and adjuvant groups (14 and 22 patients, extrapolating to a hazard ratio of 0.63; one-sided P = .091).
“With a limited number of events, overall survival is not statistically different at this time,” Dr. Patel said. “Landmark 2-year survival was 72% in the neoadjuvant arm and 49% in the adjuvant arm.”
The authors note that the benefit of neoadjuvant therapy remained consistent across a range of factors, including patient age, sex, performance status, stage of disease, ulceration, and BRAF status. The same proportion of patients in both groups received adjuvant pembrolizumab following surgery.
Rates of adverse events were similar in both groups, and neoadjuvant pembrolizumab did not result in an increase in adverse events related to surgery. In the neoadjuvant group, 28 patients (21%) with submitted pathology reports were noted to have had a complete pathologic response (0% viable tumor) on local review.
Questions remain
Invited discussant James Larkin, PhD, FRCP, FMedSci, a clinical researcher at The Royal Marsden Hospital, London, noted that the study had “striking results” and was a landmark trial with a simple but powerful design.
However, he pointed to some questions which need to be addressed in the future. “One important question is what is the optimal duration of neoadjuvant treatment, and can we individualize it?”
Another question is just how much postoperative treatment is really needed and whether pathology help determine that. “Can surgery be safely avoided altogether?” he asked. “Another issue is the need for anti-CTL4 therapy – which patients might benefit from anti-CTL4, in addition to anti-PD-1?”
“And by extension, this paradigm provides a great platform for testing new agents, including combinations in cases where PD-1 is not sufficient to achieve a sufficient response,” said Dr. Larkin. “In the future, trials addressing these questions hand us a major opportunity to individualize and rationally de-escalate treatment.”
Also weighing in on the study, another expert pointed out that neoadjuvant therapy in this setting is already being considered as an option. “The use of immunotherapy before surgery has been reported in some trials such as the OPACIN-neo and PRADO trials,” said Anthony J. Olszanski, RPh, MD, Vice Chair of Research at the Fox Chase Cancer Center, Philadelphia. “Results have been quite exciting and have led the NCCN to list this as a potential option for some patients in the current melanoma guidelines.”
S1801 is funded by the NIH/NCI and in part by MSD through a Cooperative Research and Development Agreement with the NCI. Pembrolizumab (KEYTRUDA) is Merck’s anti-PD-1 therapy. Dr. Patel has declared multiple relationships with industry as noted in the abstract; several co-authors have also made disclosures. Dr. Olszanski has reported participating in advisory boards for BMS, Merck, and InstilBio and running trials for them.
A version of this article first appeared on Medscape.com.
PARIS – “It’s not just what you give, it’s when you give it,” said the investigator reporting “that the same treatment for resectable melanoma given in a different sequence can generate lower rates of melanoma recurrence.”
Sapna Patel, MD, associate professor of melanoma medical oncology at The University of Texas MD Anderson Cancer Center, Houston, reported the results from the SWOG S1801 trial, which showed that than patients who received pembrolizumab after surgery only.
At a median follow-up of almost 15 months, there was a 42% lower rate of recurrence or death.
“Compared to the same treatment given entirely in the adjuvant setting, neoadjuvant pembrolizumab followed by adjuvant pembrolizumab improves event-free survival in resectable melanoma,” Dr. Patel commented.
She suggested that the explanation for the findings was that “inhibiting the PD-1/PD-L1 immune checkpoints before surgery gives an antitumor response at local and distant sites, and this occurs before resection of the tumor bed. This approach tends to leave behind a larger number of anti-tumor T cells ... [and] these T cells can be activated and circulated systematically to recognize and attack micro-metastatic melanoma tumors.”
The findings were presented during a presidential symposium at the European Society for Medical Oncology (ESMO) Congress 2022, Paris.
“This trial provides us with more evidence of when one strategy may be preferred over the other,” commented Maya Dimitrova, MD, medical oncologist at NYU Langone Perlmutter Cancer Center. She was not involved with the trial.
“Neoadjuvant immunotherapy has elicited impressive complete pathologic responses, which thus far have proven to be associated with a durable response. Neoadjuvant therapy may help identify patients who will respond well to checkpoint inhibitors and allow for de-escalation of therapy,” she told this news organization when approached for comment.
“As with all neoadjuvant therapy, we don’t want the treatment to compromise the outcomes of surgery when the intent is curative, and we once again have evidence that this is not the case when it comes to immune therapy,” she said. However, she added that “we will need further survival data to really change the standard of practice in high-risk melanoma and demonstrate whether there is a superior sequence of therapy and surgery.”
Details of the new results
The S1801 clinical trial enrolled 345 participants with stage IIIB through stage IV melanoma considered resectable. The cohort was randomized to receive either upfront surgery followed by 18 doses of pembrolizumab 200 mg every 3 weeks for a total of 18 doses or neoadjuvant therapy with pembrolizumab 200 mg (3 doses) followed by 15 doses of adjuvant pembrolizumab.
The primary endpoint was event-free survival (EFS), defined as the time from randomization to the occurrence of one of the following: disease progression or toxicity that resulted in not receiving surgery, failure to begin adjuvant therapy within 84 days of surgery, melanoma recurrence after surgery, or death from any cause.
At a median follow-up of 14.7 months, EFS was significantly higher for patients in the neoadjuvant group, compared with those receiving adjuvant therapy only (HR, 0.58; one-sided log-rank P = .004). A total of 36 participants died in the neoadjuvant and adjuvant groups (14 and 22 patients, extrapolating to a hazard ratio of 0.63; one-sided P = .091).
“With a limited number of events, overall survival is not statistically different at this time,” Dr. Patel said. “Landmark 2-year survival was 72% in the neoadjuvant arm and 49% in the adjuvant arm.”
The authors note that the benefit of neoadjuvant therapy remained consistent across a range of factors, including patient age, sex, performance status, stage of disease, ulceration, and BRAF status. The same proportion of patients in both groups received adjuvant pembrolizumab following surgery.
Rates of adverse events were similar in both groups, and neoadjuvant pembrolizumab did not result in an increase in adverse events related to surgery. In the neoadjuvant group, 28 patients (21%) with submitted pathology reports were noted to have had a complete pathologic response (0% viable tumor) on local review.
Questions remain
Invited discussant James Larkin, PhD, FRCP, FMedSci, a clinical researcher at The Royal Marsden Hospital, London, noted that the study had “striking results” and was a landmark trial with a simple but powerful design.
However, he pointed to some questions which need to be addressed in the future. “One important question is what is the optimal duration of neoadjuvant treatment, and can we individualize it?”
Another question is just how much postoperative treatment is really needed and whether pathology help determine that. “Can surgery be safely avoided altogether?” he asked. “Another issue is the need for anti-CTL4 therapy – which patients might benefit from anti-CTL4, in addition to anti-PD-1?”
“And by extension, this paradigm provides a great platform for testing new agents, including combinations in cases where PD-1 is not sufficient to achieve a sufficient response,” said Dr. Larkin. “In the future, trials addressing these questions hand us a major opportunity to individualize and rationally de-escalate treatment.”
Also weighing in on the study, another expert pointed out that neoadjuvant therapy in this setting is already being considered as an option. “The use of immunotherapy before surgery has been reported in some trials such as the OPACIN-neo and PRADO trials,” said Anthony J. Olszanski, RPh, MD, Vice Chair of Research at the Fox Chase Cancer Center, Philadelphia. “Results have been quite exciting and have led the NCCN to list this as a potential option for some patients in the current melanoma guidelines.”
S1801 is funded by the NIH/NCI and in part by MSD through a Cooperative Research and Development Agreement with the NCI. Pembrolizumab (KEYTRUDA) is Merck’s anti-PD-1 therapy. Dr. Patel has declared multiple relationships with industry as noted in the abstract; several co-authors have also made disclosures. Dr. Olszanski has reported participating in advisory boards for BMS, Merck, and InstilBio and running trials for them.
A version of this article first appeared on Medscape.com.
TBI is an unrecognized risk factor for cardiovascular disease
(CVD). More severe TBI is associated with higher risk of CVD, new research shows.
Given the relatively young age of post-9/11–era veterans with TBI, there may be an increased burden of heart disease in the future as these veterans age and develop traditional risk factors for CVD, the investigators, led by Ian J. Stewart, MD, with Uniformed Services University, Bethesda, Md., wrote.
The study was published online in JAMA Neurology.
Novel data
Since Sept. 11, 2001, 4.5 million people have served in the U.S. military, with their time in service defined by the long-running wars in Iraq and Afghanistan. Estimates suggest that up to 20% of post-9/11 veterans sustained a TBI.
While some evidence suggests that TBI increases the risk of CVD, prior reports have focused mainly on cerebrovascular outcomes. Until now, the potential association of TBI with CVD has not been comprehensively examined in post-9/11–era veterans.
The retrospective cohort study included 1,559,928 predominantly male post-9/11 veterans, including 301,169 (19.3%) with a history of TBI and 1,258,759 (81%) with no TBI history.
In fully adjusted models, compared with veterans with no TBI history, a history of mild, moderate/severe, or penetrating TBI was associated with increased risk of developing the composite CVD endpoint (coronary artery disease, stroke, peripheral artery disease, and CVD death).
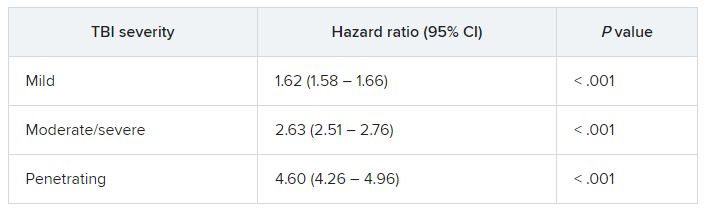
TBIs of all severities were associated with the individual components of the composite outcome, except penetrating TBI and CVD death.
“The association of TBI with subsequent CVD was not attenuated in multivariable models, suggesting that TBI may be accounting for risk that is independent from the other variables,” Dr. Stewart and colleagues wrote.
They noted that the risk was highest shortly after injury, but TBI remained significantly associated with CVD for years after the initial insult.
Why TBI may raise the risk of subsequent CVD remains unclear.
It’s possible that patients with TBI develop more traditional risk factors for CVD through time than do patients without TBI. A study in mice found that TBI led to increased rates of atherosclerosis, the researchers said.
An additional mechanism may be disruption of autonomic regulation, which has been known to occur after TBI.
Another potential pathway is through mental health diagnoses, such as posttraumatic stress disorder; a large body of work has identified associations between PTSD and CVD, including among post-9/11 veterans.
Further work is needed to determine how this risk can be modified to improve outcomes for post-9/11–era veterans, the researchers write.
Unrecognized CVD risk factor?
Reached for comment, Shaheen E. Lakhan, MD, PhD, a neurologist and researcher from Boston who wasn’t involved in the study, said the effects of TBI on heart health are “very underreported, and most clinicians would not make the link.”
“When the brain suffers a traumatic injury, it activates a cascade of neuro-inflammation that goes haywire in an attempt to protect further brain damage. Oftentimes, these inflammatory by-products leak into the body, especially in trauma, when the barriers are broken between brain and body, and can cause systemic body inflammation, which is well associated with heart disease,” Dr. Lakhan said.
In addition, Dr. Lakhan said, “TBI itself localized to just the brain can negatively affect good health habits, leading to worsening heart health, too.”
“Research like this brings light where not much exists and underscores the importance of protecting our brains from physical trauma,” he said.
The study was supported by the assistant secretary of defense for health affairs, endorsed by the Department of Defense through the Psychological Health/Traumatic Brain Injury Research Program Long-Term Impact of Military-Relevant Brain Injury Consortium, and by the U.S. Department of Veterans Affairs. Dr. Stewart and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
(CVD). More severe TBI is associated with higher risk of CVD, new research shows.
Given the relatively young age of post-9/11–era veterans with TBI, there may be an increased burden of heart disease in the future as these veterans age and develop traditional risk factors for CVD, the investigators, led by Ian J. Stewart, MD, with Uniformed Services University, Bethesda, Md., wrote.
The study was published online in JAMA Neurology.
Novel data
Since Sept. 11, 2001, 4.5 million people have served in the U.S. military, with their time in service defined by the long-running wars in Iraq and Afghanistan. Estimates suggest that up to 20% of post-9/11 veterans sustained a TBI.
While some evidence suggests that TBI increases the risk of CVD, prior reports have focused mainly on cerebrovascular outcomes. Until now, the potential association of TBI with CVD has not been comprehensively examined in post-9/11–era veterans.
The retrospective cohort study included 1,559,928 predominantly male post-9/11 veterans, including 301,169 (19.3%) with a history of TBI and 1,258,759 (81%) with no TBI history.
In fully adjusted models, compared with veterans with no TBI history, a history of mild, moderate/severe, or penetrating TBI was associated with increased risk of developing the composite CVD endpoint (coronary artery disease, stroke, peripheral artery disease, and CVD death).

TBIs of all severities were associated with the individual components of the composite outcome, except penetrating TBI and CVD death.
“The association of TBI with subsequent CVD was not attenuated in multivariable models, suggesting that TBI may be accounting for risk that is independent from the other variables,” Dr. Stewart and colleagues wrote.
They noted that the risk was highest shortly after injury, but TBI remained significantly associated with CVD for years after the initial insult.
Why TBI may raise the risk of subsequent CVD remains unclear.
It’s possible that patients with TBI develop more traditional risk factors for CVD through time than do patients without TBI. A study in mice found that TBI led to increased rates of atherosclerosis, the researchers said.
An additional mechanism may be disruption of autonomic regulation, which has been known to occur after TBI.
Another potential pathway is through mental health diagnoses, such as posttraumatic stress disorder; a large body of work has identified associations between PTSD and CVD, including among post-9/11 veterans.
Further work is needed to determine how this risk can be modified to improve outcomes for post-9/11–era veterans, the researchers write.
Unrecognized CVD risk factor?
Reached for comment, Shaheen E. Lakhan, MD, PhD, a neurologist and researcher from Boston who wasn’t involved in the study, said the effects of TBI on heart health are “very underreported, and most clinicians would not make the link.”
“When the brain suffers a traumatic injury, it activates a cascade of neuro-inflammation that goes haywire in an attempt to protect further brain damage. Oftentimes, these inflammatory by-products leak into the body, especially in trauma, when the barriers are broken between brain and body, and can cause systemic body inflammation, which is well associated with heart disease,” Dr. Lakhan said.
In addition, Dr. Lakhan said, “TBI itself localized to just the brain can negatively affect good health habits, leading to worsening heart health, too.”
“Research like this brings light where not much exists and underscores the importance of protecting our brains from physical trauma,” he said.
The study was supported by the assistant secretary of defense for health affairs, endorsed by the Department of Defense through the Psychological Health/Traumatic Brain Injury Research Program Long-Term Impact of Military-Relevant Brain Injury Consortium, and by the U.S. Department of Veterans Affairs. Dr. Stewart and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
(CVD). More severe TBI is associated with higher risk of CVD, new research shows.
Given the relatively young age of post-9/11–era veterans with TBI, there may be an increased burden of heart disease in the future as these veterans age and develop traditional risk factors for CVD, the investigators, led by Ian J. Stewart, MD, with Uniformed Services University, Bethesda, Md., wrote.
The study was published online in JAMA Neurology.
Novel data
Since Sept. 11, 2001, 4.5 million people have served in the U.S. military, with their time in service defined by the long-running wars in Iraq and Afghanistan. Estimates suggest that up to 20% of post-9/11 veterans sustained a TBI.
While some evidence suggests that TBI increases the risk of CVD, prior reports have focused mainly on cerebrovascular outcomes. Until now, the potential association of TBI with CVD has not been comprehensively examined in post-9/11–era veterans.
The retrospective cohort study included 1,559,928 predominantly male post-9/11 veterans, including 301,169 (19.3%) with a history of TBI and 1,258,759 (81%) with no TBI history.
In fully adjusted models, compared with veterans with no TBI history, a history of mild, moderate/severe, or penetrating TBI was associated with increased risk of developing the composite CVD endpoint (coronary artery disease, stroke, peripheral artery disease, and CVD death).

TBIs of all severities were associated with the individual components of the composite outcome, except penetrating TBI and CVD death.
“The association of TBI with subsequent CVD was not attenuated in multivariable models, suggesting that TBI may be accounting for risk that is independent from the other variables,” Dr. Stewart and colleagues wrote.
They noted that the risk was highest shortly after injury, but TBI remained significantly associated with CVD for years after the initial insult.
Why TBI may raise the risk of subsequent CVD remains unclear.
It’s possible that patients with TBI develop more traditional risk factors for CVD through time than do patients without TBI. A study in mice found that TBI led to increased rates of atherosclerosis, the researchers said.
An additional mechanism may be disruption of autonomic regulation, which has been known to occur after TBI.
Another potential pathway is through mental health diagnoses, such as posttraumatic stress disorder; a large body of work has identified associations between PTSD and CVD, including among post-9/11 veterans.
Further work is needed to determine how this risk can be modified to improve outcomes for post-9/11–era veterans, the researchers write.
Unrecognized CVD risk factor?
Reached for comment, Shaheen E. Lakhan, MD, PhD, a neurologist and researcher from Boston who wasn’t involved in the study, said the effects of TBI on heart health are “very underreported, and most clinicians would not make the link.”
“When the brain suffers a traumatic injury, it activates a cascade of neuro-inflammation that goes haywire in an attempt to protect further brain damage. Oftentimes, these inflammatory by-products leak into the body, especially in trauma, when the barriers are broken between brain and body, and can cause systemic body inflammation, which is well associated with heart disease,” Dr. Lakhan said.
In addition, Dr. Lakhan said, “TBI itself localized to just the brain can negatively affect good health habits, leading to worsening heart health, too.”
“Research like this brings light where not much exists and underscores the importance of protecting our brains from physical trauma,” he said.
The study was supported by the assistant secretary of defense for health affairs, endorsed by the Department of Defense through the Psychological Health/Traumatic Brain Injury Research Program Long-Term Impact of Military-Relevant Brain Injury Consortium, and by the U.S. Department of Veterans Affairs. Dr. Stewart and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AMA joins in lawsuit accusing Cigna of underpaying physicians
, the nation’s largest third-party network.
The American Medical Association, the Medical Society of New Jersey, and the Washington State Medical Association on Sept. 12 entered into a legal battle between the giant insurers and patients.
At issue are claims involving the firm MultiPlan as an intermediary. Cigna had not responded to this news organization at press time following multiple requests for comment.
According to the legal complaint AMA and the two state medical societies joined, MultiPlan has contracts with more than 1.2 million clinicians. Under these agreements, medical professionals agree to accept a set percentage of billed charges as payment in full, while not holding patients responsible for the difference between the original billed charges and the discounted rate.
But the complaint alleges that MultiPlan failed to stick with that bargain. In a statement, AMA President Jack Resneck Jr, MD, said the physician groups joined the legal action “to shed light on Cigna’s misconduct and create remedies so that patients and physicians can look forward to getting what they are promised.”
Dr. Resneck said Cigna’s approach “is riddled with conflicts of interest and manipulations that routinely shortchanged payments to MultiPlan Network physicians and interfered with the patient-physician relationship by ignoring the MultiPlan contracts and making incorrect statements to patients about their liability for the unpaid portion of the billed charges.”
According to the complaint, Cigna used a company called Zelis to “unilaterally re-price’’ claims at an amount far lower than that called for by the MultiPlan Contract. The three cases cited in the lawsuit stem from a 2017 spine surgery in Washington and 2018 orthopedic and 2020 breast reconstruction surgeries in New Jersey. The decisions to ignore the previous agreements and cut the reimbursement led the physicians involved to eventually bill patients for some of the money in dispute, according to the complaint.
“The providers were left in a very untenable situation,” D. Brian Hufford, an attorney involved in the case, told this news organization. “Their only choice was to go after the insurance company and sue them or they have to go after the patient. That interferes with the patient-doctor relationship.”
Mr. Hufford, who’s a partner at law firm Zuckerman Spaeder, said that these kinds of cases fall beyond the protections provided by the No Surprises Act. Plaintiffs in these cases were enrolled in what are called self-insured plans provided by employers, through which they were supposed to be allowed to seek out-of-network care.
Highly concerning are the messages that insurers send to patients through explanation of benefits (EOB) statements, Mr. Hufford said. Thus in this case against Cigna, physicians and patients have the “same interest in trying to make sure the insurance companies are paying the appropriate amounts for these services,” he said.
Cigna “is telling the patients that the provider has accepted something, and that the patient does not have to worry about paying for that, when in fact that’s not true,” Mr. Hufford said. “That goes beyond merely not complying with the plan documents, but also engaging in conduct that we believe was inappropriate.”
A version of this article first appeared on Medscape.com.
, the nation’s largest third-party network.
The American Medical Association, the Medical Society of New Jersey, and the Washington State Medical Association on Sept. 12 entered into a legal battle between the giant insurers and patients.
At issue are claims involving the firm MultiPlan as an intermediary. Cigna had not responded to this news organization at press time following multiple requests for comment.
According to the legal complaint AMA and the two state medical societies joined, MultiPlan has contracts with more than 1.2 million clinicians. Under these agreements, medical professionals agree to accept a set percentage of billed charges as payment in full, while not holding patients responsible for the difference between the original billed charges and the discounted rate.
But the complaint alleges that MultiPlan failed to stick with that bargain. In a statement, AMA President Jack Resneck Jr, MD, said the physician groups joined the legal action “to shed light on Cigna’s misconduct and create remedies so that patients and physicians can look forward to getting what they are promised.”
Dr. Resneck said Cigna’s approach “is riddled with conflicts of interest and manipulations that routinely shortchanged payments to MultiPlan Network physicians and interfered with the patient-physician relationship by ignoring the MultiPlan contracts and making incorrect statements to patients about their liability for the unpaid portion of the billed charges.”
According to the complaint, Cigna used a company called Zelis to “unilaterally re-price’’ claims at an amount far lower than that called for by the MultiPlan Contract. The three cases cited in the lawsuit stem from a 2017 spine surgery in Washington and 2018 orthopedic and 2020 breast reconstruction surgeries in New Jersey. The decisions to ignore the previous agreements and cut the reimbursement led the physicians involved to eventually bill patients for some of the money in dispute, according to the complaint.
“The providers were left in a very untenable situation,” D. Brian Hufford, an attorney involved in the case, told this news organization. “Their only choice was to go after the insurance company and sue them or they have to go after the patient. That interferes with the patient-doctor relationship.”
Mr. Hufford, who’s a partner at law firm Zuckerman Spaeder, said that these kinds of cases fall beyond the protections provided by the No Surprises Act. Plaintiffs in these cases were enrolled in what are called self-insured plans provided by employers, through which they were supposed to be allowed to seek out-of-network care.
Highly concerning are the messages that insurers send to patients through explanation of benefits (EOB) statements, Mr. Hufford said. Thus in this case against Cigna, physicians and patients have the “same interest in trying to make sure the insurance companies are paying the appropriate amounts for these services,” he said.
Cigna “is telling the patients that the provider has accepted something, and that the patient does not have to worry about paying for that, when in fact that’s not true,” Mr. Hufford said. “That goes beyond merely not complying with the plan documents, but also engaging in conduct that we believe was inappropriate.”
A version of this article first appeared on Medscape.com.
, the nation’s largest third-party network.
The American Medical Association, the Medical Society of New Jersey, and the Washington State Medical Association on Sept. 12 entered into a legal battle between the giant insurers and patients.
At issue are claims involving the firm MultiPlan as an intermediary. Cigna had not responded to this news organization at press time following multiple requests for comment.
According to the legal complaint AMA and the two state medical societies joined, MultiPlan has contracts with more than 1.2 million clinicians. Under these agreements, medical professionals agree to accept a set percentage of billed charges as payment in full, while not holding patients responsible for the difference between the original billed charges and the discounted rate.
But the complaint alleges that MultiPlan failed to stick with that bargain. In a statement, AMA President Jack Resneck Jr, MD, said the physician groups joined the legal action “to shed light on Cigna’s misconduct and create remedies so that patients and physicians can look forward to getting what they are promised.”
Dr. Resneck said Cigna’s approach “is riddled with conflicts of interest and manipulations that routinely shortchanged payments to MultiPlan Network physicians and interfered with the patient-physician relationship by ignoring the MultiPlan contracts and making incorrect statements to patients about their liability for the unpaid portion of the billed charges.”
According to the complaint, Cigna used a company called Zelis to “unilaterally re-price’’ claims at an amount far lower than that called for by the MultiPlan Contract. The three cases cited in the lawsuit stem from a 2017 spine surgery in Washington and 2018 orthopedic and 2020 breast reconstruction surgeries in New Jersey. The decisions to ignore the previous agreements and cut the reimbursement led the physicians involved to eventually bill patients for some of the money in dispute, according to the complaint.
“The providers were left in a very untenable situation,” D. Brian Hufford, an attorney involved in the case, told this news organization. “Their only choice was to go after the insurance company and sue them or they have to go after the patient. That interferes with the patient-doctor relationship.”
Mr. Hufford, who’s a partner at law firm Zuckerman Spaeder, said that these kinds of cases fall beyond the protections provided by the No Surprises Act. Plaintiffs in these cases were enrolled in what are called self-insured plans provided by employers, through which they were supposed to be allowed to seek out-of-network care.
Highly concerning are the messages that insurers send to patients through explanation of benefits (EOB) statements, Mr. Hufford said. Thus in this case against Cigna, physicians and patients have the “same interest in trying to make sure the insurance companies are paying the appropriate amounts for these services,” he said.
Cigna “is telling the patients that the provider has accepted something, and that the patient does not have to worry about paying for that, when in fact that’s not true,” Mr. Hufford said. “That goes beyond merely not complying with the plan documents, but also engaging in conduct that we believe was inappropriate.”
A version of this article first appeared on Medscape.com.