User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Lipid-lowering bempedoic acid does not hasten or worsen diabetes
In an analysis of four phase 3 trials, the oral lipid-lowering drug bempedoic acid (Nexletol; Esperion) did not worsen glycemic control or increase the incidence of type 2 diabetes.
As previously reported, this first-in-class drug, which acts by inhibiting ATP-citrate lyase, was approved by the Food and Drug Administration in February 2020.
Lawrence A. Leiter MD, from the University of Toronto, delivered the findings of this latest analysis in an oral presentation at the virtual American Diabetes Association 80th Scientific Sessions.
“The current study is important as it shows overall consistent efficacy and safety regardless of glycemic status and no increase in new-onset diabetes,” Dr. Leiter said in an interview.
There is interest in how lipid-lowering drugs might affect glycemia because “meta-analyses have shown about a 10% increased risk of new-onset diabetes in statin users, although the absolute increased risk is 1 extra case per 255 treated patients [in whom one would expect 5.4 cardiovascular events to be prevented by the statin],” he noted.
In a comment, John R. Guyton, MD, from Duke University Medical Center, Durham, N.C., agreed that the new study demonstrates that “patients with diabetes and prediabetes respond to bempedoic acid with LDL cholesterol lowering that is similar to that in patients with normal glucose tolerance.”
Although “statins have a slight effect of worsening glucose tolerance and a modest effect of increasing cases of new-onset diabetes,” the current research shows that “bempedoic acid appears to be free of these effects,” said Dr. Guyton, who discussed this drug in another symposium at the meeting where he also discussed how the agent will “fit” into prescribing patterns.
How do patients with diabetes, prediabetes fare?
“Current guidelines support aggressive LDL cholesterol lowering in patients with diabetes, given the increased risk of cardiovascular morbidity and mortality,” said Dr. Leiter.
Bempedoic acid was approved as an adjunct to diet and maximally tolerated statin therapy to treat adults with atherosclerotic cardiovascular disease (ASCVD) and/or heterozygous familial hypercholesterolemia (HeFH) who require additional lowering of LDL cholesterol, although its effect on cardiovascular morbidity and mortality has not been determined, the prescribing information states.
However, it has been unknown how bempedoic acid affects LDL cholesterol or hemoglobin A1c levels in patients with diabetes, prediabetes, or normoglycemia.
To examine this, the researchers pooled data from four phase 3 trials in 3623 patients with ASCVD or HeFH who had been randomized 2:1 to bempedoic acid 180 mg/day or placebo for 12 or 24 weeks (if they were statin intolerant) or 52 weeks (if they were also on statins).
In the pooled sample, about half the patients had prediabetes (52%), and the rest had diabetes (31%) or normoglycemia (17%). Overall, 75%-84% of patients had a history of ASCVD.
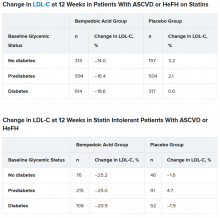
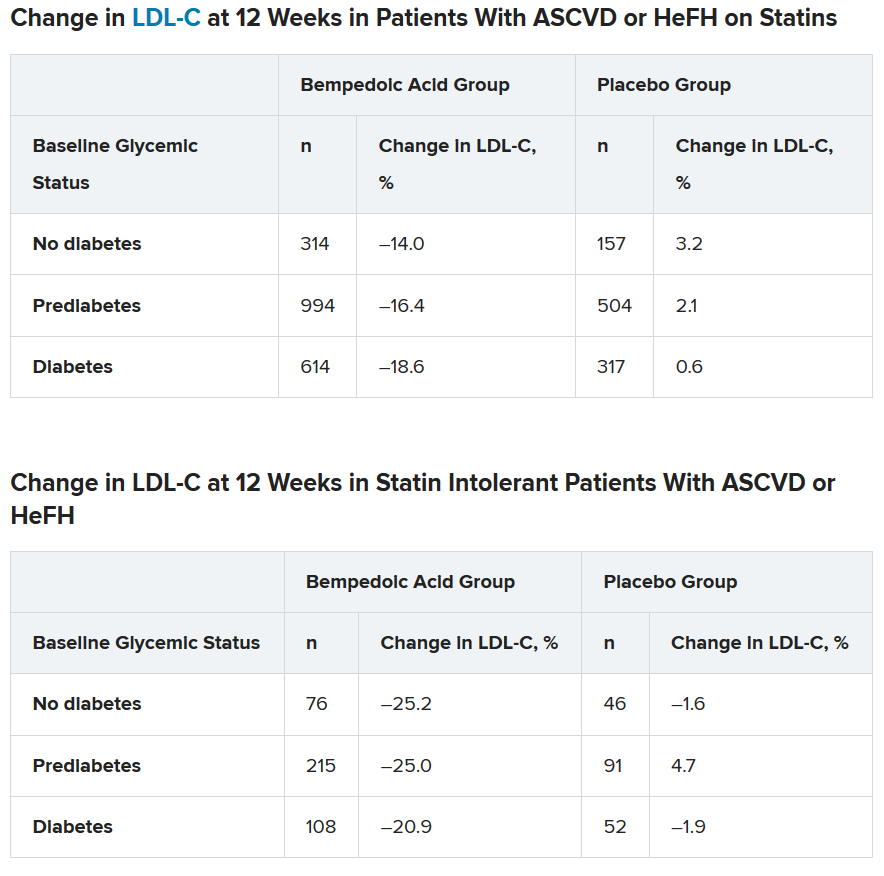
Mean LDL cholesterol levels were higher in patients with normoglycemia (119 mg/dL) or prediabetes (115 mg/dL) than in patients with diabetes (110 mg/dL).
The primary outcome was percent change in LDL cholesterol from baseline to week 12.
In the two types of patients (all with ASCVD or HeFH) – those on statins and those with statin intolerance – LDL cholesterol at 12 weeks was significantly lower in patients who received bempedoic acid, compared with placebo, regardless of whether they had no diabetes, prediabetes, or diabetes (all P < .001).
Similarly, patients who received bempedoic acid also had significant reductions in total cholesterol, non–HDL cholesterol, apolipoprotein B, and high-sensitivity C-reactive protein (hsCRP) at 12 weeks, compared with patients who received placebo (all P < .01).
The safety profile of bempedoic acid was similar to placebo and did not vary by glycemic status.
“Of course, with any lipid-lowering therapy, there’s lots of interest in changes in glycemic parameters,” said Dr. Leiter. “A1c did not increase. In fact, it was significantly lower in patients with prediabetes and diabetes on bempedoic acid versus placebo.”
In addition, “statin trials have shown small increases in body weight. We did not observe this,” he reported.
Where does bempedoic acid ‘fit?’
“Bempedoic acid will be a useful add-on to any patient who requires additional LDL cholesterol lowering,” according to Dr. Leiter. “It will typically be used as an add-on to statins, but will also be very useful in the statin-intolerant patient, especially when used in combination with ezetimib.”
The fixed-dose combination of bempedoic acid plus ezetimibe (Nexlizet; Esperion), was also approved in the United States in February, just days after bempedoic acid as a solo agent was cleared for marketing.
“Bempedoic acid would not be chosen in preference to a statin, ezetimibe, or PCSK9 inhibitor,” Dr. Guyton said. Rather, “its chief use will be in patients with statin intolerance and either FH or ASCVD when LDL-cholesterol is poorly controlled despite maximum tolerated lipid-lowering therapy.”
According to Dr. Guyton, “use of bempedoic acid should be undertaken only when provider-patient discussion acknowledges that it has not been shown to reduce cardiovascular events, although preliminary evidence from genetic analysis [Mendelian randomization study] suggests that it will,” as previously reported.
The CLEAR Outcomes cardiovascular outcomes trial of bempedoic acid completed enrollment in August 2019, involving 14,032 patients with hypercholesterolemia and high CVD risk according to a company statement.
The study was funded by Esperion. Dr. Leiter has reported being on advisory panels for Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, HLS Therapeutics, Janssen, Merck, Novo Nordisk, Sanofi, and Servier, receiving research support from Amgen, AstraZeneca, Kowa Pharmaceuticals, and the Medicines Company, and being on speakers bureaus for Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, HLS Therapeutics, Janssen, Medscape, Merck, Novo Nordisk, Sanofi, and Servier. Disclosures for the other authors are listed with the abstract. Dr. Guyton has reported being a consultant for Amarin and receiving research support form Regeneron.
A version of this article originally appeared on Medscape.com.
In an analysis of four phase 3 trials, the oral lipid-lowering drug bempedoic acid (Nexletol; Esperion) did not worsen glycemic control or increase the incidence of type 2 diabetes.
As previously reported, this first-in-class drug, which acts by inhibiting ATP-citrate lyase, was approved by the Food and Drug Administration in February 2020.
Lawrence A. Leiter MD, from the University of Toronto, delivered the findings of this latest analysis in an oral presentation at the virtual American Diabetes Association 80th Scientific Sessions.
“The current study is important as it shows overall consistent efficacy and safety regardless of glycemic status and no increase in new-onset diabetes,” Dr. Leiter said in an interview.
There is interest in how lipid-lowering drugs might affect glycemia because “meta-analyses have shown about a 10% increased risk of new-onset diabetes in statin users, although the absolute increased risk is 1 extra case per 255 treated patients [in whom one would expect 5.4 cardiovascular events to be prevented by the statin],” he noted.
In a comment, John R. Guyton, MD, from Duke University Medical Center, Durham, N.C., agreed that the new study demonstrates that “patients with diabetes and prediabetes respond to bempedoic acid with LDL cholesterol lowering that is similar to that in patients with normal glucose tolerance.”
Although “statins have a slight effect of worsening glucose tolerance and a modest effect of increasing cases of new-onset diabetes,” the current research shows that “bempedoic acid appears to be free of these effects,” said Dr. Guyton, who discussed this drug in another symposium at the meeting where he also discussed how the agent will “fit” into prescribing patterns.
How do patients with diabetes, prediabetes fare?
“Current guidelines support aggressive LDL cholesterol lowering in patients with diabetes, given the increased risk of cardiovascular morbidity and mortality,” said Dr. Leiter.
Bempedoic acid was approved as an adjunct to diet and maximally tolerated statin therapy to treat adults with atherosclerotic cardiovascular disease (ASCVD) and/or heterozygous familial hypercholesterolemia (HeFH) who require additional lowering of LDL cholesterol, although its effect on cardiovascular morbidity and mortality has not been determined, the prescribing information states.
However, it has been unknown how bempedoic acid affects LDL cholesterol or hemoglobin A1c levels in patients with diabetes, prediabetes, or normoglycemia.
To examine this, the researchers pooled data from four phase 3 trials in 3623 patients with ASCVD or HeFH who had been randomized 2:1 to bempedoic acid 180 mg/day or placebo for 12 or 24 weeks (if they were statin intolerant) or 52 weeks (if they were also on statins).
In the pooled sample, about half the patients had prediabetes (52%), and the rest had diabetes (31%) or normoglycemia (17%). Overall, 75%-84% of patients had a history of ASCVD.
Mean LDL cholesterol levels were higher in patients with normoglycemia (119 mg/dL) or prediabetes (115 mg/dL) than in patients with diabetes (110 mg/dL).
The primary outcome was percent change in LDL cholesterol from baseline to week 12.
In the two types of patients (all with ASCVD or HeFH) – those on statins and those with statin intolerance – LDL cholesterol at 12 weeks was significantly lower in patients who received bempedoic acid, compared with placebo, regardless of whether they had no diabetes, prediabetes, or diabetes (all P < .001).
Similarly, patients who received bempedoic acid also had significant reductions in total cholesterol, non–HDL cholesterol, apolipoprotein B, and high-sensitivity C-reactive protein (hsCRP) at 12 weeks, compared with patients who received placebo (all P < .01).
The safety profile of bempedoic acid was similar to placebo and did not vary by glycemic status.
“Of course, with any lipid-lowering therapy, there’s lots of interest in changes in glycemic parameters,” said Dr. Leiter. “A1c did not increase. In fact, it was significantly lower in patients with prediabetes and diabetes on bempedoic acid versus placebo.”
In addition, “statin trials have shown small increases in body weight. We did not observe this,” he reported.
Where does bempedoic acid ‘fit?’
“Bempedoic acid will be a useful add-on to any patient who requires additional LDL cholesterol lowering,” according to Dr. Leiter. “It will typically be used as an add-on to statins, but will also be very useful in the statin-intolerant patient, especially when used in combination with ezetimib.”
The fixed-dose combination of bempedoic acid plus ezetimibe (Nexlizet; Esperion), was also approved in the United States in February, just days after bempedoic acid as a solo agent was cleared for marketing.
“Bempedoic acid would not be chosen in preference to a statin, ezetimibe, or PCSK9 inhibitor,” Dr. Guyton said. Rather, “its chief use will be in patients with statin intolerance and either FH or ASCVD when LDL-cholesterol is poorly controlled despite maximum tolerated lipid-lowering therapy.”
According to Dr. Guyton, “use of bempedoic acid should be undertaken only when provider-patient discussion acknowledges that it has not been shown to reduce cardiovascular events, although preliminary evidence from genetic analysis [Mendelian randomization study] suggests that it will,” as previously reported.
The CLEAR Outcomes cardiovascular outcomes trial of bempedoic acid completed enrollment in August 2019, involving 14,032 patients with hypercholesterolemia and high CVD risk according to a company statement.
The study was funded by Esperion. Dr. Leiter has reported being on advisory panels for Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, HLS Therapeutics, Janssen, Merck, Novo Nordisk, Sanofi, and Servier, receiving research support from Amgen, AstraZeneca, Kowa Pharmaceuticals, and the Medicines Company, and being on speakers bureaus for Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, HLS Therapeutics, Janssen, Medscape, Merck, Novo Nordisk, Sanofi, and Servier. Disclosures for the other authors are listed with the abstract. Dr. Guyton has reported being a consultant for Amarin and receiving research support form Regeneron.
A version of this article originally appeared on Medscape.com.
In an analysis of four phase 3 trials, the oral lipid-lowering drug bempedoic acid (Nexletol; Esperion) did not worsen glycemic control or increase the incidence of type 2 diabetes.
As previously reported, this first-in-class drug, which acts by inhibiting ATP-citrate lyase, was approved by the Food and Drug Administration in February 2020.
Lawrence A. Leiter MD, from the University of Toronto, delivered the findings of this latest analysis in an oral presentation at the virtual American Diabetes Association 80th Scientific Sessions.
“The current study is important as it shows overall consistent efficacy and safety regardless of glycemic status and no increase in new-onset diabetes,” Dr. Leiter said in an interview.
There is interest in how lipid-lowering drugs might affect glycemia because “meta-analyses have shown about a 10% increased risk of new-onset diabetes in statin users, although the absolute increased risk is 1 extra case per 255 treated patients [in whom one would expect 5.4 cardiovascular events to be prevented by the statin],” he noted.
In a comment, John R. Guyton, MD, from Duke University Medical Center, Durham, N.C., agreed that the new study demonstrates that “patients with diabetes and prediabetes respond to bempedoic acid with LDL cholesterol lowering that is similar to that in patients with normal glucose tolerance.”
Although “statins have a slight effect of worsening glucose tolerance and a modest effect of increasing cases of new-onset diabetes,” the current research shows that “bempedoic acid appears to be free of these effects,” said Dr. Guyton, who discussed this drug in another symposium at the meeting where he also discussed how the agent will “fit” into prescribing patterns.
How do patients with diabetes, prediabetes fare?
“Current guidelines support aggressive LDL cholesterol lowering in patients with diabetes, given the increased risk of cardiovascular morbidity and mortality,” said Dr. Leiter.
Bempedoic acid was approved as an adjunct to diet and maximally tolerated statin therapy to treat adults with atherosclerotic cardiovascular disease (ASCVD) and/or heterozygous familial hypercholesterolemia (HeFH) who require additional lowering of LDL cholesterol, although its effect on cardiovascular morbidity and mortality has not been determined, the prescribing information states.
However, it has been unknown how bempedoic acid affects LDL cholesterol or hemoglobin A1c levels in patients with diabetes, prediabetes, or normoglycemia.
To examine this, the researchers pooled data from four phase 3 trials in 3623 patients with ASCVD or HeFH who had been randomized 2:1 to bempedoic acid 180 mg/day or placebo for 12 or 24 weeks (if they were statin intolerant) or 52 weeks (if they were also on statins).
In the pooled sample, about half the patients had prediabetes (52%), and the rest had diabetes (31%) or normoglycemia (17%). Overall, 75%-84% of patients had a history of ASCVD.
Mean LDL cholesterol levels were higher in patients with normoglycemia (119 mg/dL) or prediabetes (115 mg/dL) than in patients with diabetes (110 mg/dL).
The primary outcome was percent change in LDL cholesterol from baseline to week 12.
In the two types of patients (all with ASCVD or HeFH) – those on statins and those with statin intolerance – LDL cholesterol at 12 weeks was significantly lower in patients who received bempedoic acid, compared with placebo, regardless of whether they had no diabetes, prediabetes, or diabetes (all P < .001).
Similarly, patients who received bempedoic acid also had significant reductions in total cholesterol, non–HDL cholesterol, apolipoprotein B, and high-sensitivity C-reactive protein (hsCRP) at 12 weeks, compared with patients who received placebo (all P < .01).
The safety profile of bempedoic acid was similar to placebo and did not vary by glycemic status.
“Of course, with any lipid-lowering therapy, there’s lots of interest in changes in glycemic parameters,” said Dr. Leiter. “A1c did not increase. In fact, it was significantly lower in patients with prediabetes and diabetes on bempedoic acid versus placebo.”
In addition, “statin trials have shown small increases in body weight. We did not observe this,” he reported.
Where does bempedoic acid ‘fit?’
“Bempedoic acid will be a useful add-on to any patient who requires additional LDL cholesterol lowering,” according to Dr. Leiter. “It will typically be used as an add-on to statins, but will also be very useful in the statin-intolerant patient, especially when used in combination with ezetimib.”
The fixed-dose combination of bempedoic acid plus ezetimibe (Nexlizet; Esperion), was also approved in the United States in February, just days after bempedoic acid as a solo agent was cleared for marketing.
“Bempedoic acid would not be chosen in preference to a statin, ezetimibe, or PCSK9 inhibitor,” Dr. Guyton said. Rather, “its chief use will be in patients with statin intolerance and either FH or ASCVD when LDL-cholesterol is poorly controlled despite maximum tolerated lipid-lowering therapy.”
According to Dr. Guyton, “use of bempedoic acid should be undertaken only when provider-patient discussion acknowledges that it has not been shown to reduce cardiovascular events, although preliminary evidence from genetic analysis [Mendelian randomization study] suggests that it will,” as previously reported.
The CLEAR Outcomes cardiovascular outcomes trial of bempedoic acid completed enrollment in August 2019, involving 14,032 patients with hypercholesterolemia and high CVD risk according to a company statement.
The study was funded by Esperion. Dr. Leiter has reported being on advisory panels for Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, HLS Therapeutics, Janssen, Merck, Novo Nordisk, Sanofi, and Servier, receiving research support from Amgen, AstraZeneca, Kowa Pharmaceuticals, and the Medicines Company, and being on speakers bureaus for Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, HLS Therapeutics, Janssen, Medscape, Merck, Novo Nordisk, Sanofi, and Servier. Disclosures for the other authors are listed with the abstract. Dr. Guyton has reported being a consultant for Amarin and receiving research support form Regeneron.
A version of this article originally appeared on Medscape.com.
Where does dexamethasone fit in with diabetic ketoacidosis in COVID-19?
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
Corresponding author Marie E. McDonnell, MD, director of the diabetes program at Brigham and Women’s Hospital, Boston, Massachusetts, discussed the recommendations with Medscape Medical News and also spoke about the news this week that the corticosteroid dexamethasone reduced death rates in severely ill patients with COVID-19.
The full JCEM article, by lead author Nadine E. Palermo, DO, Division of Endocrinology, Diabetes, and Hypertension, also at Brigham and Women’s Hospital, covers DKA diagnosis and triage, and emphasizes that usual hospital protocols for DKA management may need to be adjusted during COVID-19 to help preserve personal protective equipment and ICU beds.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” McDonnell said in an Endocrine Society statement.
What about dexamethasone for severe COVID-19 in diabetes?
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors.
But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
McDonnell told Medscape Medical News that she would need to see formal results to better understand exactly which patients were studied and which ones benefited.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” she said. “If they all had acute respiratory distress syndrome [ARDS],” that’s different.
“There are already some data supporting steroid use in ARDS,” she noted, but added that not all of it suggests benefit.
She pointed to one of several studies now showing that diabetes, and hyperglycemia among people without a prior diabetes diagnosis, are both strong predictors of mortality in hospitalized patients with COVID-19.
“There was a very clear relationship between hyperglycemia and outcomes. We really shouldn’t put people at risk until we have clear data,” she said.
If, once the data are reviewed and appropriate dexamethasone becomes an established treatment for severe COVID-19, hyperglycemia would be a concern among all patients, not just those with previously diagnosed diabetes, she noted.
“We know a good number of people with prediabetes develop hyperglycemia when put on steroids. They can push people over the edge. We’re not going to miss anybody, but treating steroid-induced hyperglycemia is really hard,” McDonnell explained.
She also recommended 2014 guidance from Diabetes UK and the Association of British Clinical Diabetologists, which addresses management of inpatient steroid-induced DKA in patients with and without pre-existing diabetes.
Another major concern, she said, is “patients trying to get dexamethasone when they start to get sick” because this is not the right population to use this agent.
“We worry about people who do not need this drug. If they have diabetes, they put themselves at risk of hyperglycemia, which then increases the risk of severe COVID-19. And then they’re also putting themselves at risk of DKA. It would just be bad medicine,” she said.
Managing DKA in the face of COVID-19: Flexibility is key
In the JCEM article, Palermo and colleagues emphasize that the usual hospital protocols for DKA management may need to be adjusted during COVID-19 in the interest of reducing transmission risk and preserving scare resources.
They provide evidence for alternative treatment strategies, such as the use of subcutaneous rather than intravenous insulin when appropriate.
“We wanted to outline when exactly you should consider nonintensive management strategies for DKA,” McDonnell further explained to Medscape Medical News.
“That would include those with mild or some with moderate DKA. ... The idea is to remind our colleagues about that because hospitals tend to operate on a protocol-driven algorithmic methodology, they can forget to step off the usual care pathway even if evidence supports that,” she said.
But on the other hand, she also said that, in some very complex or severely ill patients with COVID-19, classical intravenous insulin therapy makes the most sense even if their DKA is mild.
The outpatient setting: Prevention and preparation
The new article also addresses several concerns regarding DKA prevention in the outpatient setting.
As with other guidelines, it includes a reminder that patients with diabetes should be advised to discontinue sodium-glucose cotransporter 2 (SGLT2) inhibitors if they become ill with COVID-19, especially if they’re not eating or drinking normally, because they raise the risk for DKA.
Also, for patients with type 1 diabetes, particularly those with a history of repeated DKA, “this is the time to make sure we reach out to patients to refill their insulin prescriptions and address issues related to cost and other access difficulties,” McDonnell said.
The authors also emphasize that insulin starts and education should not be postponed during the pandemic. “Patients identified as meeting criteria to start insulin should be referred for urgent education, either in person or, whenever possible and practical, via video teleconferencing,” they urge.
McDonnell has reported receiving research funding from Novo Nordisk. The other two authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
Corresponding author Marie E. McDonnell, MD, director of the diabetes program at Brigham and Women’s Hospital, Boston, Massachusetts, discussed the recommendations with Medscape Medical News and also spoke about the news this week that the corticosteroid dexamethasone reduced death rates in severely ill patients with COVID-19.
The full JCEM article, by lead author Nadine E. Palermo, DO, Division of Endocrinology, Diabetes, and Hypertension, also at Brigham and Women’s Hospital, covers DKA diagnosis and triage, and emphasizes that usual hospital protocols for DKA management may need to be adjusted during COVID-19 to help preserve personal protective equipment and ICU beds.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” McDonnell said in an Endocrine Society statement.
What about dexamethasone for severe COVID-19 in diabetes?
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors.
But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
McDonnell told Medscape Medical News that she would need to see formal results to better understand exactly which patients were studied and which ones benefited.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” she said. “If they all had acute respiratory distress syndrome [ARDS],” that’s different.
“There are already some data supporting steroid use in ARDS,” she noted, but added that not all of it suggests benefit.
She pointed to one of several studies now showing that diabetes, and hyperglycemia among people without a prior diabetes diagnosis, are both strong predictors of mortality in hospitalized patients with COVID-19.
“There was a very clear relationship between hyperglycemia and outcomes. We really shouldn’t put people at risk until we have clear data,” she said.
If, once the data are reviewed and appropriate dexamethasone becomes an established treatment for severe COVID-19, hyperglycemia would be a concern among all patients, not just those with previously diagnosed diabetes, she noted.
“We know a good number of people with prediabetes develop hyperglycemia when put on steroids. They can push people over the edge. We’re not going to miss anybody, but treating steroid-induced hyperglycemia is really hard,” McDonnell explained.
She also recommended 2014 guidance from Diabetes UK and the Association of British Clinical Diabetologists, which addresses management of inpatient steroid-induced DKA in patients with and without pre-existing diabetes.
Another major concern, she said, is “patients trying to get dexamethasone when they start to get sick” because this is not the right population to use this agent.
“We worry about people who do not need this drug. If they have diabetes, they put themselves at risk of hyperglycemia, which then increases the risk of severe COVID-19. And then they’re also putting themselves at risk of DKA. It would just be bad medicine,” she said.
Managing DKA in the face of COVID-19: Flexibility is key
In the JCEM article, Palermo and colleagues emphasize that the usual hospital protocols for DKA management may need to be adjusted during COVID-19 in the interest of reducing transmission risk and preserving scare resources.
They provide evidence for alternative treatment strategies, such as the use of subcutaneous rather than intravenous insulin when appropriate.
“We wanted to outline when exactly you should consider nonintensive management strategies for DKA,” McDonnell further explained to Medscape Medical News.
“That would include those with mild or some with moderate DKA. ... The idea is to remind our colleagues about that because hospitals tend to operate on a protocol-driven algorithmic methodology, they can forget to step off the usual care pathway even if evidence supports that,” she said.
But on the other hand, she also said that, in some very complex or severely ill patients with COVID-19, classical intravenous insulin therapy makes the most sense even if their DKA is mild.
The outpatient setting: Prevention and preparation
The new article also addresses several concerns regarding DKA prevention in the outpatient setting.
As with other guidelines, it includes a reminder that patients with diabetes should be advised to discontinue sodium-glucose cotransporter 2 (SGLT2) inhibitors if they become ill with COVID-19, especially if they’re not eating or drinking normally, because they raise the risk for DKA.
Also, for patients with type 1 diabetes, particularly those with a history of repeated DKA, “this is the time to make sure we reach out to patients to refill their insulin prescriptions and address issues related to cost and other access difficulties,” McDonnell said.
The authors also emphasize that insulin starts and education should not be postponed during the pandemic. “Patients identified as meeting criteria to start insulin should be referred for urgent education, either in person or, whenever possible and practical, via video teleconferencing,” they urge.
McDonnell has reported receiving research funding from Novo Nordisk. The other two authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
A new article in the Journal of Clinical Endocrinology & Metabolism (JCEM) addresses unique concerns and considerations regarding diabetic ketoacidosis (DKA) in the setting of COVID-19.
Corresponding author Marie E. McDonnell, MD, director of the diabetes program at Brigham and Women’s Hospital, Boston, Massachusetts, discussed the recommendations with Medscape Medical News and also spoke about the news this week that the corticosteroid dexamethasone reduced death rates in severely ill patients with COVID-19.
The full JCEM article, by lead author Nadine E. Palermo, DO, Division of Endocrinology, Diabetes, and Hypertension, also at Brigham and Women’s Hospital, covers DKA diagnosis and triage, and emphasizes that usual hospital protocols for DKA management may need to be adjusted during COVID-19 to help preserve personal protective equipment and ICU beds.
“Hospitals and clinicians need to be able to quickly identify and manage DKA in COVID patients to save lives. This involves determining the options for management, including when less intensive subcutaneous insulin is indicated, and understanding how to guide patients on avoiding this serious complication,” McDonnell said in an Endocrine Society statement.
What about dexamethasone for severe COVID-19 in diabetes?
The new article briefly touches on the fact that upward adjustments to intensive intravenous insulin therapy for DKA may be necessary in patients with COVID-19 who are receiving concomitant corticosteroids or vasopressors.
But it was written prior to the June 16 announcement of the “RECOVERY” trial results with dexamethasone. The UK National Health Service immediately approved the drug’s use in the COVID-19 setting, despite the fact that there has been no published article on the findings yet.
McDonnell told Medscape Medical News that she would need to see formal results to better understand exactly which patients were studied and which ones benefited.
“The peer review will be critical. It looks as if it only benefits people who need respiratory support, but I want to understand that in much more detail,” she said. “If they all had acute respiratory distress syndrome [ARDS],” that’s different.
“There are already some data supporting steroid use in ARDS,” she noted, but added that not all of it suggests benefit.
She pointed to one of several studies now showing that diabetes, and hyperglycemia among people without a prior diabetes diagnosis, are both strong predictors of mortality in hospitalized patients with COVID-19.
“There was a very clear relationship between hyperglycemia and outcomes. We really shouldn’t put people at risk until we have clear data,” she said.
If, once the data are reviewed and appropriate dexamethasone becomes an established treatment for severe COVID-19, hyperglycemia would be a concern among all patients, not just those with previously diagnosed diabetes, she noted.
“We know a good number of people with prediabetes develop hyperglycemia when put on steroids. They can push people over the edge. We’re not going to miss anybody, but treating steroid-induced hyperglycemia is really hard,” McDonnell explained.
She also recommended 2014 guidance from Diabetes UK and the Association of British Clinical Diabetologists, which addresses management of inpatient steroid-induced DKA in patients with and without pre-existing diabetes.
Another major concern, she said, is “patients trying to get dexamethasone when they start to get sick” because this is not the right population to use this agent.
“We worry about people who do not need this drug. If they have diabetes, they put themselves at risk of hyperglycemia, which then increases the risk of severe COVID-19. And then they’re also putting themselves at risk of DKA. It would just be bad medicine,” she said.
Managing DKA in the face of COVID-19: Flexibility is key
In the JCEM article, Palermo and colleagues emphasize that the usual hospital protocols for DKA management may need to be adjusted during COVID-19 in the interest of reducing transmission risk and preserving scare resources.
They provide evidence for alternative treatment strategies, such as the use of subcutaneous rather than intravenous insulin when appropriate.
“We wanted to outline when exactly you should consider nonintensive management strategies for DKA,” McDonnell further explained to Medscape Medical News.
“That would include those with mild or some with moderate DKA. ... The idea is to remind our colleagues about that because hospitals tend to operate on a protocol-driven algorithmic methodology, they can forget to step off the usual care pathway even if evidence supports that,” she said.
But on the other hand, she also said that, in some very complex or severely ill patients with COVID-19, classical intravenous insulin therapy makes the most sense even if their DKA is mild.
The outpatient setting: Prevention and preparation
The new article also addresses several concerns regarding DKA prevention in the outpatient setting.
As with other guidelines, it includes a reminder that patients with diabetes should be advised to discontinue sodium-glucose cotransporter 2 (SGLT2) inhibitors if they become ill with COVID-19, especially if they’re not eating or drinking normally, because they raise the risk for DKA.
Also, for patients with type 1 diabetes, particularly those with a history of repeated DKA, “this is the time to make sure we reach out to patients to refill their insulin prescriptions and address issues related to cost and other access difficulties,” McDonnell said.
The authors also emphasize that insulin starts and education should not be postponed during the pandemic. “Patients identified as meeting criteria to start insulin should be referred for urgent education, either in person or, whenever possible and practical, via video teleconferencing,” they urge.
McDonnell has reported receiving research funding from Novo Nordisk. The other two authors have reported no relevant financial relationships.
This article first appeared on Medscape.com.
Daily Recap: From hospitalist to ‘COVIDist’; Systolic BP -- How low should you go?
Here are the stories our MDedge editors across specialties think you need to know about today:
A ‘Fraternity of People Who Are Struggling’
Kathleen Ronan spent a week in a New Jersey hospital, including 5 days in the ICU, battling the novel coronavirus.
Her years of working as a home health nurse told her that the return home wouldn’t be easy, but nothing prepared her for just how much she would struggle. The once-active Ronan, 51, now needed a walker to traverse the few steps from her bed to the toilet, an effort that left her gasping for air. Her brain couldn’t even focus on an audiobook, let alone a short magazine article. “It just completely knocked the stuffing out of me,” Ronan said.
Ronan’s lingering symptoms aren’t unique to COVID-19 patients. In as many as 80% of patients leaving the ICU, researchers have documented what they call post–intensive care syndrome (PICS) — a constellation of physical, cognitive, and psychiatric symptoms that result from an ICU stay. Although underlying illness plays a role in these symptoms, the amount of time spent in critical care is a major factor.
The good news is that over the past decade, researchers have made important strides in understanding what makes PICS symptoms worse and how critical care physicians can tweak ICU protocols to reduce PICS severity. Practitioners will need to draw on this knowledge to help Ronan and the thousands of COVID-19 ICU patients like her. Read more.
The evolution of ‘COVIDists’
At the start of the pandemic earlier this year hospitalists at Baystate Health in Western Massachusetts realized the necessity of a new model of care for COVID-19 patients. Challenges included a massive surge of COVID-19 patients, a limited supply of PPE, an inadequate number of intensivists for managing the anticipated ventilated patients, and the potential of losing some of our workforce if they became infected. Hospitalists there came up with an elaborate plan to manage the disease burden and the strain on resources effectively.
A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them. The group underwent rapid training in various treatment protocols and ongoing clinical trials.
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. Read more.
How low should you go?
Cardiovascular risk continues to reduce as systolic blood pressure decreases right down to levels as low as 90 mm Hg, according to a new study.
Researchers analyzed data from a cohort of 1,457 participants (mean age, 58 years) who did not have any traditional cardiovascular risk factors and had a systolic blood pressure level between 90 and 129 mm Hg at baseline. Results showed that, during a mean follow-up of 14.5 years, there was an increase in traditional cardiovascular risk factors, coronary artery calcium, and incident cardiovascular events with increasing systolic blood pressure levels.
“We modeled systolic blood pressure on a continuous scale and saw the risk increasing in a linear fashion as blood pressure increased and this occurred right down to 90 mm Hg. We didn’t see any nadir or J-point where there may be an increased risk at lower pressures,” said lead author Seamus Whelton, MD, assistant professor of medicine at the division of cardiology at Johns Hopkins Medicine, Baltimore.
“From an individual level we can now say that in healthy individuals, a systolic pressure in the 90s is not too low. It is a positive thing. And it is recommended to try and keep systolic pressure at these levels if possible by maintaining a healthy lifestyle,” Dr. Whelton said in an interview. Read more.
Asthma tops spending on avoidable pediatric inpatient stays
Asthma costs nearly equaled potentially avoidable hospital bills for diabetes, gastroenteritis, and UTIs combined in a study of in-patient stays among children aged 3 months to 17 years.
Indeed, hospital charges for the treatment of children with asthma made up nearly half of all potentially avoidable pediatric inpatient costs in 2017, according to the Agency for Healthcare Research and Quality.
The cost of potentially avoidable visits for asthma that year was $278 million, versus $284 million combined for the other three conditions, Kimberly W. McDermott, PhD, and H. Joanna Jiang, PhD, reported in an AHRQ statistical brief.
The state inpatient databases of the AHRQ’s Healthcare Cost and Utilization Project included 1.4 million inpatient stays among children aged 3 months to 17 years in 2017, of which 8% (108,300) were deemed potentially preventable.
Rates of potentially avoidable stays for asthma (159 per 100,000 population), gastroenteritis (90 per 100,000), and UTIs (41 per 100,000) were highest for children aged 0-4 years and generally decreased with age, but diabetes stays increased with age, rising from 12 per 100,000 in children aged 5-9 years to 38 per 100,000 for those 15-17 years old, the researchers said. Read more.
Adding monoclonal antibodies to Botox for migraine prevention
Adjunctive preventive therapy with a calcitonin gene–related peptide monoclonal antibody (CGRP-mAb) medication is safe and effective in patients with chronic migraine who have only achieved a partial response to onabotulinumtoxinA (Botox) treatment.
Investigators found the CGRP-mAbs significantly reduced the number of headache days and pain severity with adverse event rates similar to those reported in previous trials of these medications.
Although Botox is associated with significant clinical improvement in chronic migraine, it often fails to adequately control headache frequency and additional medications are needed. Three CGRP-mAbs have recently been approved for migraine prevention, with results from clinical trials demonstrating they are effective for both chronic and episodic migraine. Patients treated with Botox had been excluded from these earlier trials, however. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
A ‘Fraternity of People Who Are Struggling’
Kathleen Ronan spent a week in a New Jersey hospital, including 5 days in the ICU, battling the novel coronavirus.
Her years of working as a home health nurse told her that the return home wouldn’t be easy, but nothing prepared her for just how much she would struggle. The once-active Ronan, 51, now needed a walker to traverse the few steps from her bed to the toilet, an effort that left her gasping for air. Her brain couldn’t even focus on an audiobook, let alone a short magazine article. “It just completely knocked the stuffing out of me,” Ronan said.
Ronan’s lingering symptoms aren’t unique to COVID-19 patients. In as many as 80% of patients leaving the ICU, researchers have documented what they call post–intensive care syndrome (PICS) — a constellation of physical, cognitive, and psychiatric symptoms that result from an ICU stay. Although underlying illness plays a role in these symptoms, the amount of time spent in critical care is a major factor.
The good news is that over the past decade, researchers have made important strides in understanding what makes PICS symptoms worse and how critical care physicians can tweak ICU protocols to reduce PICS severity. Practitioners will need to draw on this knowledge to help Ronan and the thousands of COVID-19 ICU patients like her. Read more.
The evolution of ‘COVIDists’
At the start of the pandemic earlier this year hospitalists at Baystate Health in Western Massachusetts realized the necessity of a new model of care for COVID-19 patients. Challenges included a massive surge of COVID-19 patients, a limited supply of PPE, an inadequate number of intensivists for managing the anticipated ventilated patients, and the potential of losing some of our workforce if they became infected. Hospitalists there came up with an elaborate plan to manage the disease burden and the strain on resources effectively.
A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them. The group underwent rapid training in various treatment protocols and ongoing clinical trials.
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. Read more.
How low should you go?
Cardiovascular risk continues to reduce as systolic blood pressure decreases right down to levels as low as 90 mm Hg, according to a new study.
Researchers analyzed data from a cohort of 1,457 participants (mean age, 58 years) who did not have any traditional cardiovascular risk factors and had a systolic blood pressure level between 90 and 129 mm Hg at baseline. Results showed that, during a mean follow-up of 14.5 years, there was an increase in traditional cardiovascular risk factors, coronary artery calcium, and incident cardiovascular events with increasing systolic blood pressure levels.
“We modeled systolic blood pressure on a continuous scale and saw the risk increasing in a linear fashion as blood pressure increased and this occurred right down to 90 mm Hg. We didn’t see any nadir or J-point where there may be an increased risk at lower pressures,” said lead author Seamus Whelton, MD, assistant professor of medicine at the division of cardiology at Johns Hopkins Medicine, Baltimore.
“From an individual level we can now say that in healthy individuals, a systolic pressure in the 90s is not too low. It is a positive thing. And it is recommended to try and keep systolic pressure at these levels if possible by maintaining a healthy lifestyle,” Dr. Whelton said in an interview. Read more.
Asthma tops spending on avoidable pediatric inpatient stays
Asthma costs nearly equaled potentially avoidable hospital bills for diabetes, gastroenteritis, and UTIs combined in a study of in-patient stays among children aged 3 months to 17 years.
Indeed, hospital charges for the treatment of children with asthma made up nearly half of all potentially avoidable pediatric inpatient costs in 2017, according to the Agency for Healthcare Research and Quality.
The cost of potentially avoidable visits for asthma that year was $278 million, versus $284 million combined for the other three conditions, Kimberly W. McDermott, PhD, and H. Joanna Jiang, PhD, reported in an AHRQ statistical brief.
The state inpatient databases of the AHRQ’s Healthcare Cost and Utilization Project included 1.4 million inpatient stays among children aged 3 months to 17 years in 2017, of which 8% (108,300) were deemed potentially preventable.
Rates of potentially avoidable stays for asthma (159 per 100,000 population), gastroenteritis (90 per 100,000), and UTIs (41 per 100,000) were highest for children aged 0-4 years and generally decreased with age, but diabetes stays increased with age, rising from 12 per 100,000 in children aged 5-9 years to 38 per 100,000 for those 15-17 years old, the researchers said. Read more.
Adding monoclonal antibodies to Botox for migraine prevention
Adjunctive preventive therapy with a calcitonin gene–related peptide monoclonal antibody (CGRP-mAb) medication is safe and effective in patients with chronic migraine who have only achieved a partial response to onabotulinumtoxinA (Botox) treatment.
Investigators found the CGRP-mAbs significantly reduced the number of headache days and pain severity with adverse event rates similar to those reported in previous trials of these medications.
Although Botox is associated with significant clinical improvement in chronic migraine, it often fails to adequately control headache frequency and additional medications are needed. Three CGRP-mAbs have recently been approved for migraine prevention, with results from clinical trials demonstrating they are effective for both chronic and episodic migraine. Patients treated with Botox had been excluded from these earlier trials, however. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
A ‘Fraternity of People Who Are Struggling’
Kathleen Ronan spent a week in a New Jersey hospital, including 5 days in the ICU, battling the novel coronavirus.
Her years of working as a home health nurse told her that the return home wouldn’t be easy, but nothing prepared her for just how much she would struggle. The once-active Ronan, 51, now needed a walker to traverse the few steps from her bed to the toilet, an effort that left her gasping for air. Her brain couldn’t even focus on an audiobook, let alone a short magazine article. “It just completely knocked the stuffing out of me,” Ronan said.
Ronan’s lingering symptoms aren’t unique to COVID-19 patients. In as many as 80% of patients leaving the ICU, researchers have documented what they call post–intensive care syndrome (PICS) — a constellation of physical, cognitive, and psychiatric symptoms that result from an ICU stay. Although underlying illness plays a role in these symptoms, the amount of time spent in critical care is a major factor.
The good news is that over the past decade, researchers have made important strides in understanding what makes PICS symptoms worse and how critical care physicians can tweak ICU protocols to reduce PICS severity. Practitioners will need to draw on this knowledge to help Ronan and the thousands of COVID-19 ICU patients like her. Read more.
The evolution of ‘COVIDists’
At the start of the pandemic earlier this year hospitalists at Baystate Health in Western Massachusetts realized the necessity of a new model of care for COVID-19 patients. Challenges included a massive surge of COVID-19 patients, a limited supply of PPE, an inadequate number of intensivists for managing the anticipated ventilated patients, and the potential of losing some of our workforce if they became infected. Hospitalists there came up with an elaborate plan to manage the disease burden and the strain on resources effectively.
A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them. The group underwent rapid training in various treatment protocols and ongoing clinical trials.
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. Read more.
How low should you go?
Cardiovascular risk continues to reduce as systolic blood pressure decreases right down to levels as low as 90 mm Hg, according to a new study.
Researchers analyzed data from a cohort of 1,457 participants (mean age, 58 years) who did not have any traditional cardiovascular risk factors and had a systolic blood pressure level between 90 and 129 mm Hg at baseline. Results showed that, during a mean follow-up of 14.5 years, there was an increase in traditional cardiovascular risk factors, coronary artery calcium, and incident cardiovascular events with increasing systolic blood pressure levels.
“We modeled systolic blood pressure on a continuous scale and saw the risk increasing in a linear fashion as blood pressure increased and this occurred right down to 90 mm Hg. We didn’t see any nadir or J-point where there may be an increased risk at lower pressures,” said lead author Seamus Whelton, MD, assistant professor of medicine at the division of cardiology at Johns Hopkins Medicine, Baltimore.
“From an individual level we can now say that in healthy individuals, a systolic pressure in the 90s is not too low. It is a positive thing. And it is recommended to try and keep systolic pressure at these levels if possible by maintaining a healthy lifestyle,” Dr. Whelton said in an interview. Read more.
Asthma tops spending on avoidable pediatric inpatient stays
Asthma costs nearly equaled potentially avoidable hospital bills for diabetes, gastroenteritis, and UTIs combined in a study of in-patient stays among children aged 3 months to 17 years.
Indeed, hospital charges for the treatment of children with asthma made up nearly half of all potentially avoidable pediatric inpatient costs in 2017, according to the Agency for Healthcare Research and Quality.
The cost of potentially avoidable visits for asthma that year was $278 million, versus $284 million combined for the other three conditions, Kimberly W. McDermott, PhD, and H. Joanna Jiang, PhD, reported in an AHRQ statistical brief.
The state inpatient databases of the AHRQ’s Healthcare Cost and Utilization Project included 1.4 million inpatient stays among children aged 3 months to 17 years in 2017, of which 8% (108,300) were deemed potentially preventable.
Rates of potentially avoidable stays for asthma (159 per 100,000 population), gastroenteritis (90 per 100,000), and UTIs (41 per 100,000) were highest for children aged 0-4 years and generally decreased with age, but diabetes stays increased with age, rising from 12 per 100,000 in children aged 5-9 years to 38 per 100,000 for those 15-17 years old, the researchers said. Read more.
Adding monoclonal antibodies to Botox for migraine prevention
Adjunctive preventive therapy with a calcitonin gene–related peptide monoclonal antibody (CGRP-mAb) medication is safe and effective in patients with chronic migraine who have only achieved a partial response to onabotulinumtoxinA (Botox) treatment.
Investigators found the CGRP-mAbs significantly reduced the number of headache days and pain severity with adverse event rates similar to those reported in previous trials of these medications.
Although Botox is associated with significant clinical improvement in chronic migraine, it often fails to adequately control headache frequency and additional medications are needed. Three CGRP-mAbs have recently been approved for migraine prevention, with results from clinical trials demonstrating they are effective for both chronic and episodic migraine. Patients treated with Botox had been excluded from these earlier trials, however. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
After the ICU: A ‘fraternity of people who are struggling’
By the time she was discharged from a suburban New Jersey hospital on April 10, Kathleen Ronan thought the worst was behind her. For a week before her husband rushed her to the emergency department (ED), incoherent and struggling to breathe, the novel coronavirus had ravaged her body. She tried to treat her fevers with acetaminophen and ice packs. Despite taking enough Tylenol to risk liver damage and packing herself on ice like the catch of the day, Ronan’s fever continued to rise. By the time her temperature reached 104.5° F, Ronan knew the time had come for more drastic measures.
A team of masked and gowned nurses greeted her at a triage tent outside the ED, and from there, everything becomes hazy for Ronan. She was immediately rushed to the hospital’s special COVID-19 intensive care unit (ICU), where she spent 5 days. But she has few distinct memories from this time. What she does remember is the exhaustion, the pain, the loneliness, and the fear. Her family couldn’t visit, and though Ronan works as a home health nurse, her brain was so addled with fever that she couldn’t make sense of what was happening. After a week in the hospital, 5 days of which were spent in the ICU, 51-year-old Ronan was discharged.
Her years of working as a home health nurse told her that the return home wouldn’t be easy, but nothing prepared her for just how much she would struggle. The once-active Ronan, who had supplemented long days on her feet caring for others as a nurse with regular trips to the gym, now needed a walker to traverse the few steps from her bed to the toilet, an effort that left her gasping for air. Her brain couldn’t even focus on an audiobook, let alone a short magazine article.
“It just completely knocked the stuffing out of me,” Ronan said.
Ronan’s lingering symptoms aren’t unique to COVID-19 patients. In as many as 80% of patients leaving the ICU, . Although underlying illness plays a role in these symptoms, the amount of time spent in critical care is a major factor.
Nor is PICS simply a set of side effects that will go away on their own. It includes ongoing cognitive difficulties and physical weakness, both of which can lead to employment problems. Beyond that, depression and anxiety can exacerbate – and be exacerbated by – these challenges. Psychologist Jim Jackson, PsyD, assistant director of the ICU Recovery Center at Vanderbilt University Medical Center, Nashville, Tennessee, recently spoke with a former ICU patient who has struggled since her discharge 30 years ago.
“Her life essentially stopped with her critical care stay. She hasn’t been able to move forward,” he said. “She’s part of a whole fraternity of people who are struggling.”
The good news is that over the past decade, researchers have made important strides in understanding what makes PICS symptoms worse and how critical care physicians can tweak ICU protocols to reduce PICS severity. Practitioners will need to draw on this knowledge to help Ronan and the thousands of COVID-19 ICU patients like her.
Surviving the ICU
Although the new coronavirus has pushed the world’s critical care system to its limits, it was an outbreak in 1952 that inspired the creation of intensive care units. That summer, a wave of paralytic polio swept over Copenhagen, Denmark, and anesthesiologist Bjørn Ibsen, MD, PhD, used mechanical ventilation — physically operated by medical and dental students – to help 316 children breathe for weeks at a time while their small bodies worked to fight off the virus. The effort halved the mortality rate from polio that affected breathing, from 80% to 40%.
In these wards, dedicated to the very sickest, each patient was assigned his or her own nurse. Over the next decade, hospitals in the United Kingdom and the United States established their own ICUs to treat patients with a variety of conditions. Although it helped improve survival, mortality rates in critical care units remained stubbornly high, owing to the patients’ severe underlying illnesses.
“We thought we were doing a good job if the patient survived, but we had no idea what happened after discharge,” said Carla Sevin, MD, medical director of Vanderbilt’s ICU Recovery Center. Nor did their efforts to find out always bring answers. “We struggled to get people to come in for support — they were debilitated, physically burdened, and weak.”
Through further advances in life support, by the early 2000s, the average mortality rates in American ICUs had dropped to 8% to 19%. As the number of critical care survivors began to climb, clinical researchers noticed that the lives of these patients and their families were profoundly altered by their severe illness.
As Dale Needham, MD, PhD, began his pulmonology and critical care residency in Toronto, Canada, in 2005, a group of physicians there began a 5-year longitudinal study to assess long-term outcomes of patients who developed acute respiratory distress syndrome (ARDS). Although ARDS is an acute condition, the investigators found that patients felt effects for years. Younger patients recovered better than older ones, but none of the patients› physical functioning was equivalent to that of age-matched control persons. Even 5 years later, former ICU patients only reached 76% of expected physical functioning, according to results published in the New England Journal of Medicine. The study was a wake-up call.
At a meeting in Chicago in 2010, Needham, now an intensivist at Johns Hopkins Hospital in Baltimore, Maryland, gathered an interdisciplinary group of colleagues, including patients and caregivers, to clarify the phenomena they were seeing. What emerged from that meeting, published in 2012 in Critical Care Medicine, were the diagnostic criteria for PICS: According to the new definition, PICS is characterized by new or worsening physical and neuropsychiatric deficits that range from forgetfulness and loss of motivation to physical weakness and insomnia.
The issue, Needham says, is that although the trouble starts in the ICU, it only becomes clear once patients leave. “ICU doctors aren’t the ones dealing with this,” Needham said. “We need to build stronger bridges between critical care and other professions.” That’s where PICS comes in, a definition that exists explicitly to alert healthcare providers about the constellation of challenges many of these individuals face as they try to reenter “normal” life.
Defining the problem
As an ICU nurse at the Mayo Clinic in Rochester, Minnesota, Annie Johnson, ACNP-BC, knew lots about helping hospitalized patients, but she says she didn’t know anything about what to do after discharge – at least not until her own mother became a patient.
On the first day of retirement in October 2014, Johnson’s mother flatlined. Quick-thinking paramedics resuscitated her, and after several days in critical care, she was discharged. Since then, her heart has remained healthy. Johnson’s sister, who spent time worrying over her mother at the hospital, also had lingering effects. Both have since struggled, plagued by nightmares, flashbacks, and insomnia.
Johnson initially believed her mom’s and sister’s neuropsychiatric, post-ICU struggles were unique to her family. It was only a year later, at a seminar she was attending, that she first heard the words “post–intensive care syndrome.” Suddenly, Johnson had a name for her family’s experiences, and she began to create support groups and resources to help other families like hers.
“I thought of all the patients I had treated over the years who had been on ventilators for days and days and days. And if this happened to my mom after 48 hours, what must they be going through?” she asked.
Once physicians formally defined PICS, the Society for Critical Care Medicine helped create programs to educate ICU staff, patients, and families about potential post-discharge challenges. Researchers also began to investigate factors affecting post-ICU functioning. Follow-up studies of patients with delirium (ranging from general confusion about time and place to extreme agitation and violence) showed they had striking cognitive deficits. Problems with short-term memory, flexible thinking, and motivation plagued patients for years after their critical illness, similar to the physical deficiencies seen after ARDS. Delirium was one of the strongest risk factors for neuropsychiatric problems.
“Delirium is basically a stress test for the brain,” said Babar Khan, MD, a critical care specialist at Indiana University’s Regenstrief Institute, in Bloomington. But whether delirium accentuates preexisting cognitive difficulties or creates them afresh isn’t yet clear.
Sophia Wang, MD, a geriatric psychiatrist at Indiana University who works with many critical care patients, says patients who had experienced delirium in the ICU showed significant defects in memory and executive functioning long after their hospital stay. She points to a 2015 study that followed 47 ICU patients for a year post discharge. Among those who experienced delirium, brain volumes, as measured by MRI, were smaller at 3 months, something associated with cognitive problems at 1 year. Many struggled at work, and unemployment was common. Depression and posttraumatic stress compounded these difficulties. Among those with acute respiratory distress, ICU patients who are young, female, and unemployed are most likely to suffer from posttraumatic stress disorder after they are discharge.
Critical care medicine may have given these patients a second chance at life, Wang says, but the life they return to often looks nothing like the one they had before their illness.
Prolonged mechanical ventilation and the heavy sedation that often accompanies it are predictors of PICS severity. Some of these links could be explained by the gravity of the illness that landed someone in critical care, but others are more likely to be iatrogenic, says Gerald Weinhouse, MD, a pulmonology and critical care physician and co-director of the Critical Illness Recovery Program at the Brigham and Women’s Hospital in Boston. The involvement of loved ones at the patient’s bedside, however, improved the entire family’s outcome.
When Weinhouse saw those data, he and his colleagues founded a peer support program for ICU survivors. In a study published in 2019 in Critical Care Medicine, they identified six different models for peer support for those with PICS and their families, including both online and in-person approaches. An ongoing challenge for physicians, Weinhouse says, is getting patients to engage with these programs, given that their calendars are crowded with medical appointments and that they suffer from increased physical and mental disability.
Studies such as these led critical care physicians to form the ICU Liberation Collaborative to rethink critical care medicine. At Vanderbilt, Sevin and Jackson headed up one of the world’s first post-ICU clinics, which uses an interdisciplinary team to help patients maximize their functioning. They redesigned their critical care unit in a way that allows families to spend the night and that encourages patient mobility. Both Needham and Weinhouse continue tracking patient outcomes.
Even before the novel coronavirus struck, the United States — and the world — had begun to realize that graduating from the ICU was only the start of what was often an extensive recovery.
The long road back
When COVID-19 patients began flooding intensive care wards around the world, physicians scrambled to meet their complex and desperate acute medical needs. Over the past few months, physicians have focused on keeping these patients alive. “We’ve never seen anything like it ― not even during polio — with the sheer number of patients, all with respiratory distress,” Needham said.
But he and his colleagues know this is only the beginning.
“We’re aware that survivorship issues are coming. There’s going to be a wave of sick people who survived the coronavirus but are going to need more help,” Weinhouse said.
Intensivists have been drawing on PICS research in their fight to help COVID-19 patients. Work from the past few years has shown that although sedation is required during intubation itself, not everyone needs it while on a ventilator. Titrating down sedating medication helps reduce delirium, Wang says. Such medication has been shown to contribute to later cognitive problems. Needham’s studies showing that prolonged bedrest by ICU patients causes muscular atrophy has led him to encourage patients to move as much as possible. With the help of physical therapists, many patients on ventilators can be awake, alert, and moving around the ward.
One of the biggest challenges critical-care coronavirus patients face is prolonged isolation. The constant presence of a familiar face helps orient confused and delirious patients and provides emotional support during a frightening time. But because the immediate need for infection control outweighs these benefits, few hospitals allow visitors, especially for COVID-19 patients.
To address this, some units have been using video technology to allow loved ones to call in. At Johns Hopkins, physicians have also been relying on the expertise of occupational therapists (OTs). Needham says that one OT found that rubbing the hand and back of an agitated, delirious patient helped soothe and calm him better than many medications.
Ronan, who spent 5 days in intensive care, echoes that problem. She says she found the relative lack of human contact to be one of the most challenging parts of being in a bed on a COVID-19 ward. Separated from her husband and daughter, suffering from high fever and severe illness, she lost all track of time.
Her return home was difficult, too. Although her job as a home health nurse had prepared her on some level for the challenges she would face after discharge, Ronan says the hospital provided little practical help.
“Everything is so much harder at home, even little things like going to the bathroom,” she said. “I feel like I’m trying to bail out a sinking ship with a teacup.”
Khan and other physicians, aware of the challenges Ronan and others face once home, aim to create post-ICU clinics specifically for COVID-19 patients. They want to build what Khan calls a “one-stop shop” for all the support patients need to recover. Some of that can be provided via telehealth, which may also help ease the physical burden.
Because there’s so much physicians don’t know about the coronavirus, Johnson says, such clinics are not only a chance to help the sickest COVID-19 patients, they will also help researchers learn more about the virus and improve critical care for other illnesses.
Today, nearly 2 months after discharge, Ronan is back on the job but struggles with a persistent cough — likely due to the lung damage she sustained while ill. She has constant fatigue, as well as ongoing upset stomach from all the medications she took to reduce fever and body aches. When she dons a mask for work, the tangible reminder of her hospital stay sends her into a panic attack. Physically, she’s weaker than before.
Researchers are still trying to understand everything that Ronan and other COVID-19 patients need to move on with their lives after being in the ICU. Mysteries abound, but the ground laid by Sevin, Needham, Weinhouse, and others has provided a solid foundation on which to build.
This article first appeared on Medscape.com.
By the time she was discharged from a suburban New Jersey hospital on April 10, Kathleen Ronan thought the worst was behind her. For a week before her husband rushed her to the emergency department (ED), incoherent and struggling to breathe, the novel coronavirus had ravaged her body. She tried to treat her fevers with acetaminophen and ice packs. Despite taking enough Tylenol to risk liver damage and packing herself on ice like the catch of the day, Ronan’s fever continued to rise. By the time her temperature reached 104.5° F, Ronan knew the time had come for more drastic measures.
A team of masked and gowned nurses greeted her at a triage tent outside the ED, and from there, everything becomes hazy for Ronan. She was immediately rushed to the hospital’s special COVID-19 intensive care unit (ICU), where she spent 5 days. But she has few distinct memories from this time. What she does remember is the exhaustion, the pain, the loneliness, and the fear. Her family couldn’t visit, and though Ronan works as a home health nurse, her brain was so addled with fever that she couldn’t make sense of what was happening. After a week in the hospital, 5 days of which were spent in the ICU, 51-year-old Ronan was discharged.
Her years of working as a home health nurse told her that the return home wouldn’t be easy, but nothing prepared her for just how much she would struggle. The once-active Ronan, who had supplemented long days on her feet caring for others as a nurse with regular trips to the gym, now needed a walker to traverse the few steps from her bed to the toilet, an effort that left her gasping for air. Her brain couldn’t even focus on an audiobook, let alone a short magazine article.
“It just completely knocked the stuffing out of me,” Ronan said.
Ronan’s lingering symptoms aren’t unique to COVID-19 patients. In as many as 80% of patients leaving the ICU, . Although underlying illness plays a role in these symptoms, the amount of time spent in critical care is a major factor.
Nor is PICS simply a set of side effects that will go away on their own. It includes ongoing cognitive difficulties and physical weakness, both of which can lead to employment problems. Beyond that, depression and anxiety can exacerbate – and be exacerbated by – these challenges. Psychologist Jim Jackson, PsyD, assistant director of the ICU Recovery Center at Vanderbilt University Medical Center, Nashville, Tennessee, recently spoke with a former ICU patient who has struggled since her discharge 30 years ago.
“Her life essentially stopped with her critical care stay. She hasn’t been able to move forward,” he said. “She’s part of a whole fraternity of people who are struggling.”
The good news is that over the past decade, researchers have made important strides in understanding what makes PICS symptoms worse and how critical care physicians can tweak ICU protocols to reduce PICS severity. Practitioners will need to draw on this knowledge to help Ronan and the thousands of COVID-19 ICU patients like her.
Surviving the ICU
Although the new coronavirus has pushed the world’s critical care system to its limits, it was an outbreak in 1952 that inspired the creation of intensive care units. That summer, a wave of paralytic polio swept over Copenhagen, Denmark, and anesthesiologist Bjørn Ibsen, MD, PhD, used mechanical ventilation — physically operated by medical and dental students – to help 316 children breathe for weeks at a time while their small bodies worked to fight off the virus. The effort halved the mortality rate from polio that affected breathing, from 80% to 40%.
In these wards, dedicated to the very sickest, each patient was assigned his or her own nurse. Over the next decade, hospitals in the United Kingdom and the United States established their own ICUs to treat patients with a variety of conditions. Although it helped improve survival, mortality rates in critical care units remained stubbornly high, owing to the patients’ severe underlying illnesses.
“We thought we were doing a good job if the patient survived, but we had no idea what happened after discharge,” said Carla Sevin, MD, medical director of Vanderbilt’s ICU Recovery Center. Nor did their efforts to find out always bring answers. “We struggled to get people to come in for support — they were debilitated, physically burdened, and weak.”
Through further advances in life support, by the early 2000s, the average mortality rates in American ICUs had dropped to 8% to 19%. As the number of critical care survivors began to climb, clinical researchers noticed that the lives of these patients and their families were profoundly altered by their severe illness.
As Dale Needham, MD, PhD, began his pulmonology and critical care residency in Toronto, Canada, in 2005, a group of physicians there began a 5-year longitudinal study to assess long-term outcomes of patients who developed acute respiratory distress syndrome (ARDS). Although ARDS is an acute condition, the investigators found that patients felt effects for years. Younger patients recovered better than older ones, but none of the patients› physical functioning was equivalent to that of age-matched control persons. Even 5 years later, former ICU patients only reached 76% of expected physical functioning, according to results published in the New England Journal of Medicine. The study was a wake-up call.
At a meeting in Chicago in 2010, Needham, now an intensivist at Johns Hopkins Hospital in Baltimore, Maryland, gathered an interdisciplinary group of colleagues, including patients and caregivers, to clarify the phenomena they were seeing. What emerged from that meeting, published in 2012 in Critical Care Medicine, were the diagnostic criteria for PICS: According to the new definition, PICS is characterized by new or worsening physical and neuropsychiatric deficits that range from forgetfulness and loss of motivation to physical weakness and insomnia.
The issue, Needham says, is that although the trouble starts in the ICU, it only becomes clear once patients leave. “ICU doctors aren’t the ones dealing with this,” Needham said. “We need to build stronger bridges between critical care and other professions.” That’s where PICS comes in, a definition that exists explicitly to alert healthcare providers about the constellation of challenges many of these individuals face as they try to reenter “normal” life.
Defining the problem
As an ICU nurse at the Mayo Clinic in Rochester, Minnesota, Annie Johnson, ACNP-BC, knew lots about helping hospitalized patients, but she says she didn’t know anything about what to do after discharge – at least not until her own mother became a patient.
On the first day of retirement in October 2014, Johnson’s mother flatlined. Quick-thinking paramedics resuscitated her, and after several days in critical care, she was discharged. Since then, her heart has remained healthy. Johnson’s sister, who spent time worrying over her mother at the hospital, also had lingering effects. Both have since struggled, plagued by nightmares, flashbacks, and insomnia.
Johnson initially believed her mom’s and sister’s neuropsychiatric, post-ICU struggles were unique to her family. It was only a year later, at a seminar she was attending, that she first heard the words “post–intensive care syndrome.” Suddenly, Johnson had a name for her family’s experiences, and she began to create support groups and resources to help other families like hers.
“I thought of all the patients I had treated over the years who had been on ventilators for days and days and days. And if this happened to my mom after 48 hours, what must they be going through?” she asked.
Once physicians formally defined PICS, the Society for Critical Care Medicine helped create programs to educate ICU staff, patients, and families about potential post-discharge challenges. Researchers also began to investigate factors affecting post-ICU functioning. Follow-up studies of patients with delirium (ranging from general confusion about time and place to extreme agitation and violence) showed they had striking cognitive deficits. Problems with short-term memory, flexible thinking, and motivation plagued patients for years after their critical illness, similar to the physical deficiencies seen after ARDS. Delirium was one of the strongest risk factors for neuropsychiatric problems.
“Delirium is basically a stress test for the brain,” said Babar Khan, MD, a critical care specialist at Indiana University’s Regenstrief Institute, in Bloomington. But whether delirium accentuates preexisting cognitive difficulties or creates them afresh isn’t yet clear.
Sophia Wang, MD, a geriatric psychiatrist at Indiana University who works with many critical care patients, says patients who had experienced delirium in the ICU showed significant defects in memory and executive functioning long after their hospital stay. She points to a 2015 study that followed 47 ICU patients for a year post discharge. Among those who experienced delirium, brain volumes, as measured by MRI, were smaller at 3 months, something associated with cognitive problems at 1 year. Many struggled at work, and unemployment was common. Depression and posttraumatic stress compounded these difficulties. Among those with acute respiratory distress, ICU patients who are young, female, and unemployed are most likely to suffer from posttraumatic stress disorder after they are discharge.
Critical care medicine may have given these patients a second chance at life, Wang says, but the life they return to often looks nothing like the one they had before their illness.
Prolonged mechanical ventilation and the heavy sedation that often accompanies it are predictors of PICS severity. Some of these links could be explained by the gravity of the illness that landed someone in critical care, but others are more likely to be iatrogenic, says Gerald Weinhouse, MD, a pulmonology and critical care physician and co-director of the Critical Illness Recovery Program at the Brigham and Women’s Hospital in Boston. The involvement of loved ones at the patient’s bedside, however, improved the entire family’s outcome.
When Weinhouse saw those data, he and his colleagues founded a peer support program for ICU survivors. In a study published in 2019 in Critical Care Medicine, they identified six different models for peer support for those with PICS and their families, including both online and in-person approaches. An ongoing challenge for physicians, Weinhouse says, is getting patients to engage with these programs, given that their calendars are crowded with medical appointments and that they suffer from increased physical and mental disability.
Studies such as these led critical care physicians to form the ICU Liberation Collaborative to rethink critical care medicine. At Vanderbilt, Sevin and Jackson headed up one of the world’s first post-ICU clinics, which uses an interdisciplinary team to help patients maximize their functioning. They redesigned their critical care unit in a way that allows families to spend the night and that encourages patient mobility. Both Needham and Weinhouse continue tracking patient outcomes.
Even before the novel coronavirus struck, the United States — and the world — had begun to realize that graduating from the ICU was only the start of what was often an extensive recovery.
The long road back
When COVID-19 patients began flooding intensive care wards around the world, physicians scrambled to meet their complex and desperate acute medical needs. Over the past few months, physicians have focused on keeping these patients alive. “We’ve never seen anything like it ― not even during polio — with the sheer number of patients, all with respiratory distress,” Needham said.
But he and his colleagues know this is only the beginning.
“We’re aware that survivorship issues are coming. There’s going to be a wave of sick people who survived the coronavirus but are going to need more help,” Weinhouse said.
Intensivists have been drawing on PICS research in their fight to help COVID-19 patients. Work from the past few years has shown that although sedation is required during intubation itself, not everyone needs it while on a ventilator. Titrating down sedating medication helps reduce delirium, Wang says. Such medication has been shown to contribute to later cognitive problems. Needham’s studies showing that prolonged bedrest by ICU patients causes muscular atrophy has led him to encourage patients to move as much as possible. With the help of physical therapists, many patients on ventilators can be awake, alert, and moving around the ward.
One of the biggest challenges critical-care coronavirus patients face is prolonged isolation. The constant presence of a familiar face helps orient confused and delirious patients and provides emotional support during a frightening time. But because the immediate need for infection control outweighs these benefits, few hospitals allow visitors, especially for COVID-19 patients.
To address this, some units have been using video technology to allow loved ones to call in. At Johns Hopkins, physicians have also been relying on the expertise of occupational therapists (OTs). Needham says that one OT found that rubbing the hand and back of an agitated, delirious patient helped soothe and calm him better than many medications.
Ronan, who spent 5 days in intensive care, echoes that problem. She says she found the relative lack of human contact to be one of the most challenging parts of being in a bed on a COVID-19 ward. Separated from her husband and daughter, suffering from high fever and severe illness, she lost all track of time.
Her return home was difficult, too. Although her job as a home health nurse had prepared her on some level for the challenges she would face after discharge, Ronan says the hospital provided little practical help.
“Everything is so much harder at home, even little things like going to the bathroom,” she said. “I feel like I’m trying to bail out a sinking ship with a teacup.”
Khan and other physicians, aware of the challenges Ronan and others face once home, aim to create post-ICU clinics specifically for COVID-19 patients. They want to build what Khan calls a “one-stop shop” for all the support patients need to recover. Some of that can be provided via telehealth, which may also help ease the physical burden.
Because there’s so much physicians don’t know about the coronavirus, Johnson says, such clinics are not only a chance to help the sickest COVID-19 patients, they will also help researchers learn more about the virus and improve critical care for other illnesses.
Today, nearly 2 months after discharge, Ronan is back on the job but struggles with a persistent cough — likely due to the lung damage she sustained while ill. She has constant fatigue, as well as ongoing upset stomach from all the medications she took to reduce fever and body aches. When she dons a mask for work, the tangible reminder of her hospital stay sends her into a panic attack. Physically, she’s weaker than before.
Researchers are still trying to understand everything that Ronan and other COVID-19 patients need to move on with their lives after being in the ICU. Mysteries abound, but the ground laid by Sevin, Needham, Weinhouse, and others has provided a solid foundation on which to build.
This article first appeared on Medscape.com.
By the time she was discharged from a suburban New Jersey hospital on April 10, Kathleen Ronan thought the worst was behind her. For a week before her husband rushed her to the emergency department (ED), incoherent and struggling to breathe, the novel coronavirus had ravaged her body. She tried to treat her fevers with acetaminophen and ice packs. Despite taking enough Tylenol to risk liver damage and packing herself on ice like the catch of the day, Ronan’s fever continued to rise. By the time her temperature reached 104.5° F, Ronan knew the time had come for more drastic measures.
A team of masked and gowned nurses greeted her at a triage tent outside the ED, and from there, everything becomes hazy for Ronan. She was immediately rushed to the hospital’s special COVID-19 intensive care unit (ICU), where she spent 5 days. But she has few distinct memories from this time. What she does remember is the exhaustion, the pain, the loneliness, and the fear. Her family couldn’t visit, and though Ronan works as a home health nurse, her brain was so addled with fever that she couldn’t make sense of what was happening. After a week in the hospital, 5 days of which were spent in the ICU, 51-year-old Ronan was discharged.
Her years of working as a home health nurse told her that the return home wouldn’t be easy, but nothing prepared her for just how much she would struggle. The once-active Ronan, who had supplemented long days on her feet caring for others as a nurse with regular trips to the gym, now needed a walker to traverse the few steps from her bed to the toilet, an effort that left her gasping for air. Her brain couldn’t even focus on an audiobook, let alone a short magazine article.
“It just completely knocked the stuffing out of me,” Ronan said.
Ronan’s lingering symptoms aren’t unique to COVID-19 patients. In as many as 80% of patients leaving the ICU, . Although underlying illness plays a role in these symptoms, the amount of time spent in critical care is a major factor.
Nor is PICS simply a set of side effects that will go away on their own. It includes ongoing cognitive difficulties and physical weakness, both of which can lead to employment problems. Beyond that, depression and anxiety can exacerbate – and be exacerbated by – these challenges. Psychologist Jim Jackson, PsyD, assistant director of the ICU Recovery Center at Vanderbilt University Medical Center, Nashville, Tennessee, recently spoke with a former ICU patient who has struggled since her discharge 30 years ago.
“Her life essentially stopped with her critical care stay. She hasn’t been able to move forward,” he said. “She’s part of a whole fraternity of people who are struggling.”
The good news is that over the past decade, researchers have made important strides in understanding what makes PICS symptoms worse and how critical care physicians can tweak ICU protocols to reduce PICS severity. Practitioners will need to draw on this knowledge to help Ronan and the thousands of COVID-19 ICU patients like her.
Surviving the ICU
Although the new coronavirus has pushed the world’s critical care system to its limits, it was an outbreak in 1952 that inspired the creation of intensive care units. That summer, a wave of paralytic polio swept over Copenhagen, Denmark, and anesthesiologist Bjørn Ibsen, MD, PhD, used mechanical ventilation — physically operated by medical and dental students – to help 316 children breathe for weeks at a time while their small bodies worked to fight off the virus. The effort halved the mortality rate from polio that affected breathing, from 80% to 40%.
In these wards, dedicated to the very sickest, each patient was assigned his or her own nurse. Over the next decade, hospitals in the United Kingdom and the United States established their own ICUs to treat patients with a variety of conditions. Although it helped improve survival, mortality rates in critical care units remained stubbornly high, owing to the patients’ severe underlying illnesses.
“We thought we were doing a good job if the patient survived, but we had no idea what happened after discharge,” said Carla Sevin, MD, medical director of Vanderbilt’s ICU Recovery Center. Nor did their efforts to find out always bring answers. “We struggled to get people to come in for support — they were debilitated, physically burdened, and weak.”
Through further advances in life support, by the early 2000s, the average mortality rates in American ICUs had dropped to 8% to 19%. As the number of critical care survivors began to climb, clinical researchers noticed that the lives of these patients and their families were profoundly altered by their severe illness.
As Dale Needham, MD, PhD, began his pulmonology and critical care residency in Toronto, Canada, in 2005, a group of physicians there began a 5-year longitudinal study to assess long-term outcomes of patients who developed acute respiratory distress syndrome (ARDS). Although ARDS is an acute condition, the investigators found that patients felt effects for years. Younger patients recovered better than older ones, but none of the patients› physical functioning was equivalent to that of age-matched control persons. Even 5 years later, former ICU patients only reached 76% of expected physical functioning, according to results published in the New England Journal of Medicine. The study was a wake-up call.
At a meeting in Chicago in 2010, Needham, now an intensivist at Johns Hopkins Hospital in Baltimore, Maryland, gathered an interdisciplinary group of colleagues, including patients and caregivers, to clarify the phenomena they were seeing. What emerged from that meeting, published in 2012 in Critical Care Medicine, were the diagnostic criteria for PICS: According to the new definition, PICS is characterized by new or worsening physical and neuropsychiatric deficits that range from forgetfulness and loss of motivation to physical weakness and insomnia.
The issue, Needham says, is that although the trouble starts in the ICU, it only becomes clear once patients leave. “ICU doctors aren’t the ones dealing with this,” Needham said. “We need to build stronger bridges between critical care and other professions.” That’s where PICS comes in, a definition that exists explicitly to alert healthcare providers about the constellation of challenges many of these individuals face as they try to reenter “normal” life.
Defining the problem
As an ICU nurse at the Mayo Clinic in Rochester, Minnesota, Annie Johnson, ACNP-BC, knew lots about helping hospitalized patients, but she says she didn’t know anything about what to do after discharge – at least not until her own mother became a patient.
On the first day of retirement in October 2014, Johnson’s mother flatlined. Quick-thinking paramedics resuscitated her, and after several days in critical care, she was discharged. Since then, her heart has remained healthy. Johnson’s sister, who spent time worrying over her mother at the hospital, also had lingering effects. Both have since struggled, plagued by nightmares, flashbacks, and insomnia.
Johnson initially believed her mom’s and sister’s neuropsychiatric, post-ICU struggles were unique to her family. It was only a year later, at a seminar she was attending, that she first heard the words “post–intensive care syndrome.” Suddenly, Johnson had a name for her family’s experiences, and she began to create support groups and resources to help other families like hers.
“I thought of all the patients I had treated over the years who had been on ventilators for days and days and days. And if this happened to my mom after 48 hours, what must they be going through?” she asked.
Once physicians formally defined PICS, the Society for Critical Care Medicine helped create programs to educate ICU staff, patients, and families about potential post-discharge challenges. Researchers also began to investigate factors affecting post-ICU functioning. Follow-up studies of patients with delirium (ranging from general confusion about time and place to extreme agitation and violence) showed they had striking cognitive deficits. Problems with short-term memory, flexible thinking, and motivation plagued patients for years after their critical illness, similar to the physical deficiencies seen after ARDS. Delirium was one of the strongest risk factors for neuropsychiatric problems.
“Delirium is basically a stress test for the brain,” said Babar Khan, MD, a critical care specialist at Indiana University’s Regenstrief Institute, in Bloomington. But whether delirium accentuates preexisting cognitive difficulties or creates them afresh isn’t yet clear.
Sophia Wang, MD, a geriatric psychiatrist at Indiana University who works with many critical care patients, says patients who had experienced delirium in the ICU showed significant defects in memory and executive functioning long after their hospital stay. She points to a 2015 study that followed 47 ICU patients for a year post discharge. Among those who experienced delirium, brain volumes, as measured by MRI, were smaller at 3 months, something associated with cognitive problems at 1 year. Many struggled at work, and unemployment was common. Depression and posttraumatic stress compounded these difficulties. Among those with acute respiratory distress, ICU patients who are young, female, and unemployed are most likely to suffer from posttraumatic stress disorder after they are discharge.
Critical care medicine may have given these patients a second chance at life, Wang says, but the life they return to often looks nothing like the one they had before their illness.
Prolonged mechanical ventilation and the heavy sedation that often accompanies it are predictors of PICS severity. Some of these links could be explained by the gravity of the illness that landed someone in critical care, but others are more likely to be iatrogenic, says Gerald Weinhouse, MD, a pulmonology and critical care physician and co-director of the Critical Illness Recovery Program at the Brigham and Women’s Hospital in Boston. The involvement of loved ones at the patient’s bedside, however, improved the entire family’s outcome.
When Weinhouse saw those data, he and his colleagues founded a peer support program for ICU survivors. In a study published in 2019 in Critical Care Medicine, they identified six different models for peer support for those with PICS and their families, including both online and in-person approaches. An ongoing challenge for physicians, Weinhouse says, is getting patients to engage with these programs, given that their calendars are crowded with medical appointments and that they suffer from increased physical and mental disability.
Studies such as these led critical care physicians to form the ICU Liberation Collaborative to rethink critical care medicine. At Vanderbilt, Sevin and Jackson headed up one of the world’s first post-ICU clinics, which uses an interdisciplinary team to help patients maximize their functioning. They redesigned their critical care unit in a way that allows families to spend the night and that encourages patient mobility. Both Needham and Weinhouse continue tracking patient outcomes.
Even before the novel coronavirus struck, the United States — and the world — had begun to realize that graduating from the ICU was only the start of what was often an extensive recovery.
The long road back
When COVID-19 patients began flooding intensive care wards around the world, physicians scrambled to meet their complex and desperate acute medical needs. Over the past few months, physicians have focused on keeping these patients alive. “We’ve never seen anything like it ― not even during polio — with the sheer number of patients, all with respiratory distress,” Needham said.
But he and his colleagues know this is only the beginning.
“We’re aware that survivorship issues are coming. There’s going to be a wave of sick people who survived the coronavirus but are going to need more help,” Weinhouse said.
Intensivists have been drawing on PICS research in their fight to help COVID-19 patients. Work from the past few years has shown that although sedation is required during intubation itself, not everyone needs it while on a ventilator. Titrating down sedating medication helps reduce delirium, Wang says. Such medication has been shown to contribute to later cognitive problems. Needham’s studies showing that prolonged bedrest by ICU patients causes muscular atrophy has led him to encourage patients to move as much as possible. With the help of physical therapists, many patients on ventilators can be awake, alert, and moving around the ward.
One of the biggest challenges critical-care coronavirus patients face is prolonged isolation. The constant presence of a familiar face helps orient confused and delirious patients and provides emotional support during a frightening time. But because the immediate need for infection control outweighs these benefits, few hospitals allow visitors, especially for COVID-19 patients.
To address this, some units have been using video technology to allow loved ones to call in. At Johns Hopkins, physicians have also been relying on the expertise of occupational therapists (OTs). Needham says that one OT found that rubbing the hand and back of an agitated, delirious patient helped soothe and calm him better than many medications.
Ronan, who spent 5 days in intensive care, echoes that problem. She says she found the relative lack of human contact to be one of the most challenging parts of being in a bed on a COVID-19 ward. Separated from her husband and daughter, suffering from high fever and severe illness, she lost all track of time.
Her return home was difficult, too. Although her job as a home health nurse had prepared her on some level for the challenges she would face after discharge, Ronan says the hospital provided little practical help.
“Everything is so much harder at home, even little things like going to the bathroom,” she said. “I feel like I’m trying to bail out a sinking ship with a teacup.”
Khan and other physicians, aware of the challenges Ronan and others face once home, aim to create post-ICU clinics specifically for COVID-19 patients. They want to build what Khan calls a “one-stop shop” for all the support patients need to recover. Some of that can be provided via telehealth, which may also help ease the physical burden.
Because there’s so much physicians don’t know about the coronavirus, Johnson says, such clinics are not only a chance to help the sickest COVID-19 patients, they will also help researchers learn more about the virus and improve critical care for other illnesses.
Today, nearly 2 months after discharge, Ronan is back on the job but struggles with a persistent cough — likely due to the lung damage she sustained while ill. She has constant fatigue, as well as ongoing upset stomach from all the medications she took to reduce fever and body aches. When she dons a mask for work, the tangible reminder of her hospital stay sends her into a panic attack. Physically, she’s weaker than before.
Researchers are still trying to understand everything that Ronan and other COVID-19 patients need to move on with their lives after being in the ICU. Mysteries abound, but the ground laid by Sevin, Needham, Weinhouse, and others has provided a solid foundation on which to build.
This article first appeared on Medscape.com.
CVD risk continues to fall down to systolic BP of 90 mm HG
The study analyzed data from a cohort of 1,457 participants (mean age, 58 years) who did not have any traditional cardiovascular risk factors and had a systolic blood pressure level between 90 and 129 mm Hg at baseline. Results showed that, during a mean follow-up of 14.5 years, there was an increase in traditional cardiovascular risk factors, coronary artery calcium, and incident cardiovascular events with increasing systolic blood pressure levels.
“We modeled systolic blood pressure on a continuous scale and saw the risk increasing in a linear fashion as blood pressure increased and this occurred right down to 90 mm Hg. We didn’t see any nadir or J-point where there may be an increased risk at lower pressures,” said lead author Seamus Whelton, MD.
Dr. Whelton is assistant professor of medicine at the division of cardiology at Johns Hopkins Medicine, Baltimore. He is the son of Paul Whelton, MD, chair of the 2017 American College of Cardiology/American Heart Association hypertension guideline writing committee.
“From an individual level we can now say that in healthy individuals, a systolic pressure in the 90s is not too low. It is a positive thing. And it is recommended to try and keep systolic pressure at these levels if possible by maintaining a healthy lifestyle,” Dr. Whelton said in an interview. “At a population level this finding could lead to stronger recommendations on interventions to prevent increasing blood pressure such as healthier diets, reducing sodium intake, and increasing exercise. Small changes in blood pressure on a population level will lead to large changes in cardiovascular risk on a population a level.”
The study was published online in JAMA Cardiology on June 10.
The researchers noted that populations in nonindustrialized countries have little to no increase in systolic blood pressure levels with age, while systolic blood pressure levels typically increase with age in countries with industrialized diets and lifestyles. This has important implications, because atherosclerosis is a slowly progressive disease and the lower an individual’s lifetime exposure to cardiovascular risk factors, such as increased systolic blood pressure, the lower their probable risk for a future cardiovascular event, they wrote.
While the association between systolic blood pressure level, coronary artery calcium, and atherosclerotic cardiovascular disease is well established at higher blood pressure levels, optimal systolic pressure levels for a healthy adult and whether there is a J-shaped relationship or lower limit of systolic pressure necessary to maintain adequate organ perfusion has been uncertain, they explained.
In addition, prior studies have typically used a reference systolic pressure of less than 115-120 mm Hg to define a normal level, and it is uncertain whether there is a lower level at which the risk for incident cardiovascular disease plateaus or increases.
To investigate this, they analyzed data from the Multi-Ethnic Study of Atherosclerosis, a community-based, multiethnic cohort free from known cardiovascular disease at enrollment. The current analysis included individuals with a systolic blood pressure between 90 and 129 mm Hg without other traditional cardiovascular risk factors including dyslipidemia (LDL cholesterol >160 mg/dL or HDL cholesterol <40 mg/dL), diabetes, or current tobacco use.
Results showed an adjusted hazard ratio for atherosclerotic cardiovascular disease was 1.53 for every 10 mm Hg increase in systolic blood pressure levels.
Compared with people with systolic pressures of 90-99 mm Hg, the adjusted hazard ratio for atherosclerotic cardiovascular disease risk was 3.00 for those with 100-109 mm Hg, 3.10 for those with 110-119 mm Hg, and 4.58 for those with 120-129 mm Hg.
There was also a graded increase in the prevalence of coronary artery calcium starting from systolic blood pressure levels as low as 90 mm Hg.
“Previous research on the J-shaped curve for blood pressure has primarily focused on diastolic pressure. We did control for diastolic pressure in this analysis but that was not the focus,” Dr. Whelton said. “Obviously, there will be a minimum optimum value for both diastolic and systolic pressure. But from this study we can say that for systolic pressure, that minimum recommended value is below 90 mm Hg.”
In terms of implications, the researchers wrote: “Among individuals at low or intermediate atherosclerotic cardiovascular risk, it may be more efficacious to focus on a life-course approach for preventing an increase in systolic blood pressure levels rather than treatment of established hypertension to lower systolic blood pressure levels.”
What is a normal blood pressure?
In an accompanying commentary, Daniel Jones, MD, of the University of Mississippi Medical Center, Jackson, said these new findings support the position that risk imposed by blood pressure level begins well below the current 130/80 mm Hg definition of hypertension and guideline-recommended goal.
The study is “a reminder that even a good execution of treatment of hypertension is far from an ideal way to prevent atherosclerotic cardiovascular disease,” he said.
“A systolic of 130 is not the number we should focus on for patients who are not yet hypertensive, as 130 is not a normal blood pressure,” Dr. Jones added in an audio interview on the JAMA website.
“The findings also suggest that the disease process for atherosclerotic cardiovascular disease begins early in life and support the importance of primordial prevention through a healthy lifestyle, including a healthy diet and levels of physical activity. In addition, the findings highlight the need for a population-based strategy focusing on primordial prevention to reduce the age-related increase in BP reported in all industrialized societies,” Dr. Jones wrote.
He recommended that clinicians encourage a healthy lifestyle in patients and families of patients with cardiovascular disease. “This intervention requires no sophisticated genetic testing or clinical trials to credibly inform a family that the children and grandchildren of a patient with atherosclerotic cardiovascular disease or risk factors will benefit from a healthy lifestyle beginning at the earliest age.
“Clinicians often lose sight of the big picture with regard to blood pressure because they have the patient in front of them. But that patient has children and grandchildren who may share the risk and may be in a better position with regard to prevention of future [coronary artery disease], stroke, and kidney disease,” he said.
Conducting the JAMA audio interview, Clyde Yancy, MD, chief of cardiology at Northwestern University, Chicago, said that “this is very stimulating research. It is not asking the question of what is the target blood pressure for patients with hypertension, but rather: What is the goal blood pressure if you actually want to avoid atherosclerotic cardiovascular disease risk altogether?
“These data have made us understand that there is a difference between the goal blood pressure reduction and treatment thresholds that we respect, the normative blood pressure values we see in a clinical setting, and what is truly normal blood pressure,” Dr. Yancy concluded. “That is a very important nuance, especially when we’re talking about population health. Families and communities need to understand what the true normal is.”
A version of this article originally appeared on Medscape.com.
The study analyzed data from a cohort of 1,457 participants (mean age, 58 years) who did not have any traditional cardiovascular risk factors and had a systolic blood pressure level between 90 and 129 mm Hg at baseline. Results showed that, during a mean follow-up of 14.5 years, there was an increase in traditional cardiovascular risk factors, coronary artery calcium, and incident cardiovascular events with increasing systolic blood pressure levels.
“We modeled systolic blood pressure on a continuous scale and saw the risk increasing in a linear fashion as blood pressure increased and this occurred right down to 90 mm Hg. We didn’t see any nadir or J-point where there may be an increased risk at lower pressures,” said lead author Seamus Whelton, MD.
Dr. Whelton is assistant professor of medicine at the division of cardiology at Johns Hopkins Medicine, Baltimore. He is the son of Paul Whelton, MD, chair of the 2017 American College of Cardiology/American Heart Association hypertension guideline writing committee.
“From an individual level we can now say that in healthy individuals, a systolic pressure in the 90s is not too low. It is a positive thing. And it is recommended to try and keep systolic pressure at these levels if possible by maintaining a healthy lifestyle,” Dr. Whelton said in an interview. “At a population level this finding could lead to stronger recommendations on interventions to prevent increasing blood pressure such as healthier diets, reducing sodium intake, and increasing exercise. Small changes in blood pressure on a population level will lead to large changes in cardiovascular risk on a population a level.”
The study was published online in JAMA Cardiology on June 10.
The researchers noted that populations in nonindustrialized countries have little to no increase in systolic blood pressure levels with age, while systolic blood pressure levels typically increase with age in countries with industrialized diets and lifestyles. This has important implications, because atherosclerosis is a slowly progressive disease and the lower an individual’s lifetime exposure to cardiovascular risk factors, such as increased systolic blood pressure, the lower their probable risk for a future cardiovascular event, they wrote.
While the association between systolic blood pressure level, coronary artery calcium, and atherosclerotic cardiovascular disease is well established at higher blood pressure levels, optimal systolic pressure levels for a healthy adult and whether there is a J-shaped relationship or lower limit of systolic pressure necessary to maintain adequate organ perfusion has been uncertain, they explained.
In addition, prior studies have typically used a reference systolic pressure of less than 115-120 mm Hg to define a normal level, and it is uncertain whether there is a lower level at which the risk for incident cardiovascular disease plateaus or increases.
To investigate this, they analyzed data from the Multi-Ethnic Study of Atherosclerosis, a community-based, multiethnic cohort free from known cardiovascular disease at enrollment. The current analysis included individuals with a systolic blood pressure between 90 and 129 mm Hg without other traditional cardiovascular risk factors including dyslipidemia (LDL cholesterol >160 mg/dL or HDL cholesterol <40 mg/dL), diabetes, or current tobacco use.
Results showed an adjusted hazard ratio for atherosclerotic cardiovascular disease was 1.53 for every 10 mm Hg increase in systolic blood pressure levels.
Compared with people with systolic pressures of 90-99 mm Hg, the adjusted hazard ratio for atherosclerotic cardiovascular disease risk was 3.00 for those with 100-109 mm Hg, 3.10 for those with 110-119 mm Hg, and 4.58 for those with 120-129 mm Hg.
There was also a graded increase in the prevalence of coronary artery calcium starting from systolic blood pressure levels as low as 90 mm Hg.
“Previous research on the J-shaped curve for blood pressure has primarily focused on diastolic pressure. We did control for diastolic pressure in this analysis but that was not the focus,” Dr. Whelton said. “Obviously, there will be a minimum optimum value for both diastolic and systolic pressure. But from this study we can say that for systolic pressure, that minimum recommended value is below 90 mm Hg.”
In terms of implications, the researchers wrote: “Among individuals at low or intermediate atherosclerotic cardiovascular risk, it may be more efficacious to focus on a life-course approach for preventing an increase in systolic blood pressure levels rather than treatment of established hypertension to lower systolic blood pressure levels.”
What is a normal blood pressure?
In an accompanying commentary, Daniel Jones, MD, of the University of Mississippi Medical Center, Jackson, said these new findings support the position that risk imposed by blood pressure level begins well below the current 130/80 mm Hg definition of hypertension and guideline-recommended goal.
The study is “a reminder that even a good execution of treatment of hypertension is far from an ideal way to prevent atherosclerotic cardiovascular disease,” he said.
“A systolic of 130 is not the number we should focus on for patients who are not yet hypertensive, as 130 is not a normal blood pressure,” Dr. Jones added in an audio interview on the JAMA website.
“The findings also suggest that the disease process for atherosclerotic cardiovascular disease begins early in life and support the importance of primordial prevention through a healthy lifestyle, including a healthy diet and levels of physical activity. In addition, the findings highlight the need for a population-based strategy focusing on primordial prevention to reduce the age-related increase in BP reported in all industrialized societies,” Dr. Jones wrote.
He recommended that clinicians encourage a healthy lifestyle in patients and families of patients with cardiovascular disease. “This intervention requires no sophisticated genetic testing or clinical trials to credibly inform a family that the children and grandchildren of a patient with atherosclerotic cardiovascular disease or risk factors will benefit from a healthy lifestyle beginning at the earliest age.
“Clinicians often lose sight of the big picture with regard to blood pressure because they have the patient in front of them. But that patient has children and grandchildren who may share the risk and may be in a better position with regard to prevention of future [coronary artery disease], stroke, and kidney disease,” he said.
Conducting the JAMA audio interview, Clyde Yancy, MD, chief of cardiology at Northwestern University, Chicago, said that “this is very stimulating research. It is not asking the question of what is the target blood pressure for patients with hypertension, but rather: What is the goal blood pressure if you actually want to avoid atherosclerotic cardiovascular disease risk altogether?
“These data have made us understand that there is a difference between the goal blood pressure reduction and treatment thresholds that we respect, the normative blood pressure values we see in a clinical setting, and what is truly normal blood pressure,” Dr. Yancy concluded. “That is a very important nuance, especially when we’re talking about population health. Families and communities need to understand what the true normal is.”
A version of this article originally appeared on Medscape.com.
The study analyzed data from a cohort of 1,457 participants (mean age, 58 years) who did not have any traditional cardiovascular risk factors and had a systolic blood pressure level between 90 and 129 mm Hg at baseline. Results showed that, during a mean follow-up of 14.5 years, there was an increase in traditional cardiovascular risk factors, coronary artery calcium, and incident cardiovascular events with increasing systolic blood pressure levels.
“We modeled systolic blood pressure on a continuous scale and saw the risk increasing in a linear fashion as blood pressure increased and this occurred right down to 90 mm Hg. We didn’t see any nadir or J-point where there may be an increased risk at lower pressures,” said lead author Seamus Whelton, MD.
Dr. Whelton is assistant professor of medicine at the division of cardiology at Johns Hopkins Medicine, Baltimore. He is the son of Paul Whelton, MD, chair of the 2017 American College of Cardiology/American Heart Association hypertension guideline writing committee.
“From an individual level we can now say that in healthy individuals, a systolic pressure in the 90s is not too low. It is a positive thing. And it is recommended to try and keep systolic pressure at these levels if possible by maintaining a healthy lifestyle,” Dr. Whelton said in an interview. “At a population level this finding could lead to stronger recommendations on interventions to prevent increasing blood pressure such as healthier diets, reducing sodium intake, and increasing exercise. Small changes in blood pressure on a population level will lead to large changes in cardiovascular risk on a population a level.”
The study was published online in JAMA Cardiology on June 10.
The researchers noted that populations in nonindustrialized countries have little to no increase in systolic blood pressure levels with age, while systolic blood pressure levels typically increase with age in countries with industrialized diets and lifestyles. This has important implications, because atherosclerosis is a slowly progressive disease and the lower an individual’s lifetime exposure to cardiovascular risk factors, such as increased systolic blood pressure, the lower their probable risk for a future cardiovascular event, they wrote.
While the association between systolic blood pressure level, coronary artery calcium, and atherosclerotic cardiovascular disease is well established at higher blood pressure levels, optimal systolic pressure levels for a healthy adult and whether there is a J-shaped relationship or lower limit of systolic pressure necessary to maintain adequate organ perfusion has been uncertain, they explained.
In addition, prior studies have typically used a reference systolic pressure of less than 115-120 mm Hg to define a normal level, and it is uncertain whether there is a lower level at which the risk for incident cardiovascular disease plateaus or increases.
To investigate this, they analyzed data from the Multi-Ethnic Study of Atherosclerosis, a community-based, multiethnic cohort free from known cardiovascular disease at enrollment. The current analysis included individuals with a systolic blood pressure between 90 and 129 mm Hg without other traditional cardiovascular risk factors including dyslipidemia (LDL cholesterol >160 mg/dL or HDL cholesterol <40 mg/dL), diabetes, or current tobacco use.
Results showed an adjusted hazard ratio for atherosclerotic cardiovascular disease was 1.53 for every 10 mm Hg increase in systolic blood pressure levels.
Compared with people with systolic pressures of 90-99 mm Hg, the adjusted hazard ratio for atherosclerotic cardiovascular disease risk was 3.00 for those with 100-109 mm Hg, 3.10 for those with 110-119 mm Hg, and 4.58 for those with 120-129 mm Hg.
There was also a graded increase in the prevalence of coronary artery calcium starting from systolic blood pressure levels as low as 90 mm Hg.
“Previous research on the J-shaped curve for blood pressure has primarily focused on diastolic pressure. We did control for diastolic pressure in this analysis but that was not the focus,” Dr. Whelton said. “Obviously, there will be a minimum optimum value for both diastolic and systolic pressure. But from this study we can say that for systolic pressure, that minimum recommended value is below 90 mm Hg.”
In terms of implications, the researchers wrote: “Among individuals at low or intermediate atherosclerotic cardiovascular risk, it may be more efficacious to focus on a life-course approach for preventing an increase in systolic blood pressure levels rather than treatment of established hypertension to lower systolic blood pressure levels.”
What is a normal blood pressure?
In an accompanying commentary, Daniel Jones, MD, of the University of Mississippi Medical Center, Jackson, said these new findings support the position that risk imposed by blood pressure level begins well below the current 130/80 mm Hg definition of hypertension and guideline-recommended goal.
The study is “a reminder that even a good execution of treatment of hypertension is far from an ideal way to prevent atherosclerotic cardiovascular disease,” he said.
“A systolic of 130 is not the number we should focus on for patients who are not yet hypertensive, as 130 is not a normal blood pressure,” Dr. Jones added in an audio interview on the JAMA website.
“The findings also suggest that the disease process for atherosclerotic cardiovascular disease begins early in life and support the importance of primordial prevention through a healthy lifestyle, including a healthy diet and levels of physical activity. In addition, the findings highlight the need for a population-based strategy focusing on primordial prevention to reduce the age-related increase in BP reported in all industrialized societies,” Dr. Jones wrote.
He recommended that clinicians encourage a healthy lifestyle in patients and families of patients with cardiovascular disease. “This intervention requires no sophisticated genetic testing or clinical trials to credibly inform a family that the children and grandchildren of a patient with atherosclerotic cardiovascular disease or risk factors will benefit from a healthy lifestyle beginning at the earliest age.
“Clinicians often lose sight of the big picture with regard to blood pressure because they have the patient in front of them. But that patient has children and grandchildren who may share the risk and may be in a better position with regard to prevention of future [coronary artery disease], stroke, and kidney disease,” he said.
Conducting the JAMA audio interview, Clyde Yancy, MD, chief of cardiology at Northwestern University, Chicago, said that “this is very stimulating research. It is not asking the question of what is the target blood pressure for patients with hypertension, but rather: What is the goal blood pressure if you actually want to avoid atherosclerotic cardiovascular disease risk altogether?
“These data have made us understand that there is a difference between the goal blood pressure reduction and treatment thresholds that we respect, the normative blood pressure values we see in a clinical setting, and what is truly normal blood pressure,” Dr. Yancy concluded. “That is a very important nuance, especially when we’re talking about population health. Families and communities need to understand what the true normal is.”
A version of this article originally appeared on Medscape.com.
Cardiology care ups CV monitoring, BP control in HER2+ breast cancer
Specialty care from a cardiologist may confer clinical benefits for women with HER2-positive breast cancer treated with trastuzumab, a new study suggests.
Over 48 months of follow-up, results showed cardiology involvement prior to starting trastuzumab was associated with a higher rate of guideline-recommended cardiovascular (CV) monitoring and better systolic blood pressure (BP) control.
Trastuzumab is commonly used to treat HER2-positive breast cancer, which accounts for 20% of all breast cancers. But it carries a boxed warning for decreased left ventricular ejection fraction and heart failure (HF), and interval monitoring with echocardiography is recommended for all patients receiving the monoclonal antibody.
For the study, investigators analyzed electronic health records from 1,047 patients (mean age, 54 years) who received trastuzumab between January 2009 and July 2018 in the University of Pennsylvania health system, Philadelphia. Anthracyclines were used as part of treatment in 15% of patients.
Guideline-adherent cardiovascular monitoring was defined as echocardiography assessment in the 4 months before the initiation of trastuzumab and at least every 4 months during therapy.
Overall, 28% of patients visited a cardiology or cardio-oncology provider beginning 3 months before the baseline visit until the last contact date, the authors reported in JACC: CardioOncology.
Pre-existing HF, atrial fibrillation, and anthracycline treatment were independently associated with a cardiology visit either at baseline or during follow-up.
Patients who interacted with cardiologists, compared with those who did not, had more guideline-adherent cardiac monitoring (76.4% vs 60.1%; P = .007) and cardiac biomarker testing with troponin or N-terminal pro-B-type natriuretic peptide (27.8% vs 13.8%; P = .001).
The use of guideline-adherent cardiac monitoring was 36% to 46% in previous studies of patients with breast cancer treated with adjuvant trastuzumab-based therapy, the authors note.
Among the 5,815 echocardiographic procedures for which data on provider specialty were documented, most of the orders were authorized by oncologists (approximately 84% in those with no cardiology involvement and approximately 79% in those with cardiology involvement before trastuzumab initiation).
CV risk parameters
Cardiology involvement was associated with an average 1.5 mm Hg lower systolic BP, independent of baseline systolic BP and antihypertensive medication use (95% confidence interval, –2.9 to –0.1; P = .035).
The effect size was greater in patients with baseline hypertension, who had an average 2.7 mm Hg drop in systolic BP (95% CI, –4.6 to –0.7; P = .007) and were more likely to attain a target systolic BP below 140 mm Hg (odds ratio, 1.36; 95% CI, 1.06 to 1.74; P = .016).
Body mass index (BMI) did not budge significantly in the overall population when cardiologists were involved, but it dropped 0.5 kg/m2 in women who were overweight or obese at baseline.
“I think the results are encouraging,” senior author Bonnie Ky, MD, MSCE, University of Pennsylvania, told theheart.org | Medscape Cardiology. “These are modest changes but they are significant.”
These types of changes have been associated with significant reductions in cardiovascular disease risk over time in larger clinical trials, she noted. For example, a 2 mm Hg reduction in systolic BP has been linked to a 10% reduction in stroke mortality and a 7% reduction in ischemic heart disease mortality in middle-aged adults.
“We do think they are important and speak to more aggressive risk factor modification under the care of a specialist,” said Ky, who is also editor-in-chief of JACC: CardioOncology.
This broader role for cardiologists is particularly important given the burden of pre-existing CVD and CVD risk factors in patients with cancer and survivors. In the study, the baseline prevalence of hypertension was 40.6%, dyslipidemia 23.1%, HF 3.2%, atrial fibrillation 1.7%, and diabetes 5.9%.
“Ideally, collaboration between cardiology and oncology can improve the ability to cure a patient’s cancer while minimizing the risk of adverse cardiovascular occurrences,” Erica L. Mayer, MD, MPH, Dana-Farber Cancer Institute, Boston, told theheart.org | Medscape Cardiology. “Optimization of all cardiovascular parameters, including blood pressure, lipids, and weight, may allow a patient to protect her heart health while becoming a healthy cancer survivor.”
When asked about the 28% cardiology involvement at a U.S. cancer center with one of the most well-developed cardio-oncology programs, she said “the linkage with pre-existing cardiovascular conditions, as well as the likelihood of low incidence of cardiovascular disease, in the study population may have led to what appears to be a lower percentage of patients interacting with cardiology at baseline.”
In an accompanying editorial, Mayer says a case can be made from the findings that patients with pre-existing CV disease or at high risk for adverse CV events with cancer therapy should receive multidisciplinary care that involves a cardiologist. “However, for young, otherwise healthy patients with breast cancer with few or no cardiovascular risk factors, the benefits of [additional] subspecialty care may be less clear.”
Further, the rationale supporting the recommended frequency of cardiac monitoring may not be as “compelling” in this group, given the very low incidence of baseline cardiac dysfunction or cardiac events, particularly when treated with nonanthracycline regimens, she writes.
The findings are a call for further study and more personalized medicine, agreed Ky.
“I think there’s a need absolutely for established guidelines and/or expert consensus statements about who should be referred so patients can be referred more systematically,” she said. “Referral to cardiologists, however, is certainly a function of risk factors. Part of the challenge is identifying who will derive the most benefit from cardiovascular care.
“There are some obvious cases: Patients with heart failure and patients with pre-existing cardiovascular disease should be under the regular care of a cardiologist,” Ky added. “But there’s certainly a gray zone, especially as it relates, for example, to patients with hypertension and cardiovascular risk factors. It’s not a ‘one size fits all,’ and I believe it is a matter of defining who is at increased CV risk and who would derive the greatest clinical benefit.”
Researchers at the University of Pennsylvania have developed a clinical risk–prediction algorithm and are investigating both clinical- and biomarker-guided strategies to identify and treat patients at greatest risk of developing left ventricular declines and cardiac dysfunction with exposure to cancer therapies. “These studies are one step forward, but they will all need to be externally validated,” Ky said.
Ky and Mayer reported having no relevant conflicts of interest.
This article first appeared on Medscape.com.
Specialty care from a cardiologist may confer clinical benefits for women with HER2-positive breast cancer treated with trastuzumab, a new study suggests.
Over 48 months of follow-up, results showed cardiology involvement prior to starting trastuzumab was associated with a higher rate of guideline-recommended cardiovascular (CV) monitoring and better systolic blood pressure (BP) control.
Trastuzumab is commonly used to treat HER2-positive breast cancer, which accounts for 20% of all breast cancers. But it carries a boxed warning for decreased left ventricular ejection fraction and heart failure (HF), and interval monitoring with echocardiography is recommended for all patients receiving the monoclonal antibody.
For the study, investigators analyzed electronic health records from 1,047 patients (mean age, 54 years) who received trastuzumab between January 2009 and July 2018 in the University of Pennsylvania health system, Philadelphia. Anthracyclines were used as part of treatment in 15% of patients.
Guideline-adherent cardiovascular monitoring was defined as echocardiography assessment in the 4 months before the initiation of trastuzumab and at least every 4 months during therapy.
Overall, 28% of patients visited a cardiology or cardio-oncology provider beginning 3 months before the baseline visit until the last contact date, the authors reported in JACC: CardioOncology.
Pre-existing HF, atrial fibrillation, and anthracycline treatment were independently associated with a cardiology visit either at baseline or during follow-up.
Patients who interacted with cardiologists, compared with those who did not, had more guideline-adherent cardiac monitoring (76.4% vs 60.1%; P = .007) and cardiac biomarker testing with troponin or N-terminal pro-B-type natriuretic peptide (27.8% vs 13.8%; P = .001).
The use of guideline-adherent cardiac monitoring was 36% to 46% in previous studies of patients with breast cancer treated with adjuvant trastuzumab-based therapy, the authors note.
Among the 5,815 echocardiographic procedures for which data on provider specialty were documented, most of the orders were authorized by oncologists (approximately 84% in those with no cardiology involvement and approximately 79% in those with cardiology involvement before trastuzumab initiation).
CV risk parameters
Cardiology involvement was associated with an average 1.5 mm Hg lower systolic BP, independent of baseline systolic BP and antihypertensive medication use (95% confidence interval, –2.9 to –0.1; P = .035).
The effect size was greater in patients with baseline hypertension, who had an average 2.7 mm Hg drop in systolic BP (95% CI, –4.6 to –0.7; P = .007) and were more likely to attain a target systolic BP below 140 mm Hg (odds ratio, 1.36; 95% CI, 1.06 to 1.74; P = .016).
Body mass index (BMI) did not budge significantly in the overall population when cardiologists were involved, but it dropped 0.5 kg/m2 in women who were overweight or obese at baseline.
“I think the results are encouraging,” senior author Bonnie Ky, MD, MSCE, University of Pennsylvania, told theheart.org | Medscape Cardiology. “These are modest changes but they are significant.”
These types of changes have been associated with significant reductions in cardiovascular disease risk over time in larger clinical trials, she noted. For example, a 2 mm Hg reduction in systolic BP has been linked to a 10% reduction in stroke mortality and a 7% reduction in ischemic heart disease mortality in middle-aged adults.
“We do think they are important and speak to more aggressive risk factor modification under the care of a specialist,” said Ky, who is also editor-in-chief of JACC: CardioOncology.
This broader role for cardiologists is particularly important given the burden of pre-existing CVD and CVD risk factors in patients with cancer and survivors. In the study, the baseline prevalence of hypertension was 40.6%, dyslipidemia 23.1%, HF 3.2%, atrial fibrillation 1.7%, and diabetes 5.9%.
“Ideally, collaboration between cardiology and oncology can improve the ability to cure a patient’s cancer while minimizing the risk of adverse cardiovascular occurrences,” Erica L. Mayer, MD, MPH, Dana-Farber Cancer Institute, Boston, told theheart.org | Medscape Cardiology. “Optimization of all cardiovascular parameters, including blood pressure, lipids, and weight, may allow a patient to protect her heart health while becoming a healthy cancer survivor.”
When asked about the 28% cardiology involvement at a U.S. cancer center with one of the most well-developed cardio-oncology programs, she said “the linkage with pre-existing cardiovascular conditions, as well as the likelihood of low incidence of cardiovascular disease, in the study population may have led to what appears to be a lower percentage of patients interacting with cardiology at baseline.”
In an accompanying editorial, Mayer says a case can be made from the findings that patients with pre-existing CV disease or at high risk for adverse CV events with cancer therapy should receive multidisciplinary care that involves a cardiologist. “However, for young, otherwise healthy patients with breast cancer with few or no cardiovascular risk factors, the benefits of [additional] subspecialty care may be less clear.”
Further, the rationale supporting the recommended frequency of cardiac monitoring may not be as “compelling” in this group, given the very low incidence of baseline cardiac dysfunction or cardiac events, particularly when treated with nonanthracycline regimens, she writes.
The findings are a call for further study and more personalized medicine, agreed Ky.
“I think there’s a need absolutely for established guidelines and/or expert consensus statements about who should be referred so patients can be referred more systematically,” she said. “Referral to cardiologists, however, is certainly a function of risk factors. Part of the challenge is identifying who will derive the most benefit from cardiovascular care.
“There are some obvious cases: Patients with heart failure and patients with pre-existing cardiovascular disease should be under the regular care of a cardiologist,” Ky added. “But there’s certainly a gray zone, especially as it relates, for example, to patients with hypertension and cardiovascular risk factors. It’s not a ‘one size fits all,’ and I believe it is a matter of defining who is at increased CV risk and who would derive the greatest clinical benefit.”
Researchers at the University of Pennsylvania have developed a clinical risk–prediction algorithm and are investigating both clinical- and biomarker-guided strategies to identify and treat patients at greatest risk of developing left ventricular declines and cardiac dysfunction with exposure to cancer therapies. “These studies are one step forward, but they will all need to be externally validated,” Ky said.
Ky and Mayer reported having no relevant conflicts of interest.
This article first appeared on Medscape.com.
Specialty care from a cardiologist may confer clinical benefits for women with HER2-positive breast cancer treated with trastuzumab, a new study suggests.
Over 48 months of follow-up, results showed cardiology involvement prior to starting trastuzumab was associated with a higher rate of guideline-recommended cardiovascular (CV) monitoring and better systolic blood pressure (BP) control.
Trastuzumab is commonly used to treat HER2-positive breast cancer, which accounts for 20% of all breast cancers. But it carries a boxed warning for decreased left ventricular ejection fraction and heart failure (HF), and interval monitoring with echocardiography is recommended for all patients receiving the monoclonal antibody.
For the study, investigators analyzed electronic health records from 1,047 patients (mean age, 54 years) who received trastuzumab between January 2009 and July 2018 in the University of Pennsylvania health system, Philadelphia. Anthracyclines were used as part of treatment in 15% of patients.
Guideline-adherent cardiovascular monitoring was defined as echocardiography assessment in the 4 months before the initiation of trastuzumab and at least every 4 months during therapy.
Overall, 28% of patients visited a cardiology or cardio-oncology provider beginning 3 months before the baseline visit until the last contact date, the authors reported in JACC: CardioOncology.
Pre-existing HF, atrial fibrillation, and anthracycline treatment were independently associated with a cardiology visit either at baseline or during follow-up.
Patients who interacted with cardiologists, compared with those who did not, had more guideline-adherent cardiac monitoring (76.4% vs 60.1%; P = .007) and cardiac biomarker testing with troponin or N-terminal pro-B-type natriuretic peptide (27.8% vs 13.8%; P = .001).
The use of guideline-adherent cardiac monitoring was 36% to 46% in previous studies of patients with breast cancer treated with adjuvant trastuzumab-based therapy, the authors note.
Among the 5,815 echocardiographic procedures for which data on provider specialty were documented, most of the orders were authorized by oncologists (approximately 84% in those with no cardiology involvement and approximately 79% in those with cardiology involvement before trastuzumab initiation).
CV risk parameters
Cardiology involvement was associated with an average 1.5 mm Hg lower systolic BP, independent of baseline systolic BP and antihypertensive medication use (95% confidence interval, –2.9 to –0.1; P = .035).
The effect size was greater in patients with baseline hypertension, who had an average 2.7 mm Hg drop in systolic BP (95% CI, –4.6 to –0.7; P = .007) and were more likely to attain a target systolic BP below 140 mm Hg (odds ratio, 1.36; 95% CI, 1.06 to 1.74; P = .016).
Body mass index (BMI) did not budge significantly in the overall population when cardiologists were involved, but it dropped 0.5 kg/m2 in women who were overweight or obese at baseline.
“I think the results are encouraging,” senior author Bonnie Ky, MD, MSCE, University of Pennsylvania, told theheart.org | Medscape Cardiology. “These are modest changes but they are significant.”
These types of changes have been associated with significant reductions in cardiovascular disease risk over time in larger clinical trials, she noted. For example, a 2 mm Hg reduction in systolic BP has been linked to a 10% reduction in stroke mortality and a 7% reduction in ischemic heart disease mortality in middle-aged adults.
“We do think they are important and speak to more aggressive risk factor modification under the care of a specialist,” said Ky, who is also editor-in-chief of JACC: CardioOncology.
This broader role for cardiologists is particularly important given the burden of pre-existing CVD and CVD risk factors in patients with cancer and survivors. In the study, the baseline prevalence of hypertension was 40.6%, dyslipidemia 23.1%, HF 3.2%, atrial fibrillation 1.7%, and diabetes 5.9%.
“Ideally, collaboration between cardiology and oncology can improve the ability to cure a patient’s cancer while minimizing the risk of adverse cardiovascular occurrences,” Erica L. Mayer, MD, MPH, Dana-Farber Cancer Institute, Boston, told theheart.org | Medscape Cardiology. “Optimization of all cardiovascular parameters, including blood pressure, lipids, and weight, may allow a patient to protect her heart health while becoming a healthy cancer survivor.”
When asked about the 28% cardiology involvement at a U.S. cancer center with one of the most well-developed cardio-oncology programs, she said “the linkage with pre-existing cardiovascular conditions, as well as the likelihood of low incidence of cardiovascular disease, in the study population may have led to what appears to be a lower percentage of patients interacting with cardiology at baseline.”
In an accompanying editorial, Mayer says a case can be made from the findings that patients with pre-existing CV disease or at high risk for adverse CV events with cancer therapy should receive multidisciplinary care that involves a cardiologist. “However, for young, otherwise healthy patients with breast cancer with few or no cardiovascular risk factors, the benefits of [additional] subspecialty care may be less clear.”
Further, the rationale supporting the recommended frequency of cardiac monitoring may not be as “compelling” in this group, given the very low incidence of baseline cardiac dysfunction or cardiac events, particularly when treated with nonanthracycline regimens, she writes.
The findings are a call for further study and more personalized medicine, agreed Ky.
“I think there’s a need absolutely for established guidelines and/or expert consensus statements about who should be referred so patients can be referred more systematically,” she said. “Referral to cardiologists, however, is certainly a function of risk factors. Part of the challenge is identifying who will derive the most benefit from cardiovascular care.
“There are some obvious cases: Patients with heart failure and patients with pre-existing cardiovascular disease should be under the regular care of a cardiologist,” Ky added. “But there’s certainly a gray zone, especially as it relates, for example, to patients with hypertension and cardiovascular risk factors. It’s not a ‘one size fits all,’ and I believe it is a matter of defining who is at increased CV risk and who would derive the greatest clinical benefit.”
Researchers at the University of Pennsylvania have developed a clinical risk–prediction algorithm and are investigating both clinical- and biomarker-guided strategies to identify and treat patients at greatest risk of developing left ventricular declines and cardiac dysfunction with exposure to cancer therapies. “These studies are one step forward, but they will all need to be externally validated,” Ky said.
Ky and Mayer reported having no relevant conflicts of interest.
This article first appeared on Medscape.com.
The evolution of “COVIDists”
Adapting to the demands placed on hospital resources by COVID-19
The challenges posed by COVID-19 have crippled health care systems around the globe. By February 2020, the first outbreak in the United States had been set off in Washington State. We quickly became the world’s epicenter of the epidemic, with over 1.8 million patients and over 110,000 deaths.1 The rapidity of spread and the severity of the disease created a tremendous strain on resources. It blindsided policymakers and hospital administrators, which left little time to react to the challenges placed on hospital operations all over the country.
The necessity of a new care model
Although health systems in the United States are adept in managing complications of common seasonal viral respiratory illnesses, COVID-19 presented an entirely different challenge with its significantly higher mortality rate. A respiratory disease turning into a multiorgan disease that causes debilitating cardiac, renal, neurological, hematological, and psychosocial complications2 was not something we had experience managing effectively. Additional challenges included a massive surge of COVID-19 patients, a limited supply of personal protective equipment (PPE), an inadequate number of intensivists for managing the anticipated ventilated patients, and most importantly, the potential of losing some of our workforce if they became infected.
Based on the experiences in China and Italy, and various predictive models, the division of hospital medicine at Baystate Health quickly realized the necessity of a new model of care for COVID-19 patients. We came up with an elaborate plan to manage the disease burden and the strain on resources effectively. The measures we put in place could be broadly divided into three categories following the timeline of the disease: the preparatory phase, the execution phase, and the maintenance phase.
The preparatory phase: From “Hospitalists” to “COVIDists”
As in most hospitals around the country, hospitalists are the backbone of inpatient clinical operations at our health system. A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them.
COVIDists were trained in various treatment protocols and ongoing clinical trials. They were given refresher training in Advanced Cardiac Life Support (ACLS) and Fundamental Critical Care Support (FCCS) courses and were taught in critical care/ventilator management by the intensivists through rapid indoctrination in the ICU. All of them had their N-95 mask fitting updated and were trained in the safe donning and doffing of all kinds of PPE by PPE coaches. The palliative care team trained them in conducting end-of-life/code status discussions with a focus on being unable to speak with family members at the bedside. COVIDists were also assigned as Code Blue leaders for any “COVID code blue” in the hospital.
In addition to the rapid training course, COVID-related updates were disseminated daily using three different modalities: brief huddles at the start of the day with the COVIDists; a COVID-19 newsletter summarizing daily updates, new treatments, strategies, and policies; and a WhatsApp group for instantly broadcasting information to the COVIDists (Table 1).
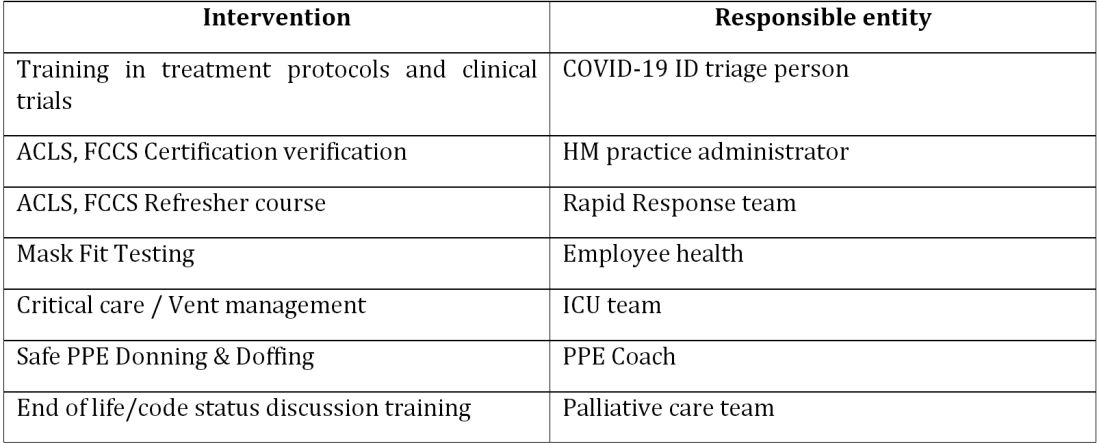
The execution phase
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. The COVIDists were also undertaking the most challenging part of the care – talking to families about end-of-life issues and the futility of aggressive care in certain patients with preexisting conditions.
Some COVIDists were deployed to the ICU to work alongside the intensivists and became an invaluable resource in ICU management when the ICU census skyrocketed during the initial phase of the outbreak. This helped in tiding the health system over during the initial crisis. Within a short time, we shifted away from an early intubation strategy, and most of the ICU patients were managed in the intermediate care units on high flow oxygen along with the awake-proning protocol. The COVIDists exclusively managed these units. They led multidisciplinary rounds two times a day with the ICU, rapid response team (RRT), the palliative care team, and the nursing team. This step drastically decreased the number of intubations, RRT activations, reduced ICU census,3 and helped with hospital capacity and patient flow (Tables 2 and 3).
This strategy also helped build solidarity and camaraderie between all these groups, making the COVIDists feel that they were never alone and that the whole hospital supported them. We are currently evaluating clinical outcomes and attempting to identify effects on mortality, length of stay, days on the ventilator, and days in ICU.

The maintenance phase
It is already 2 months since the first devising COVIDists. There is no difference in sick callouts between COVIDists and non-COVIDists. One COVIDist and one non-COVIDist contracted the disease, but none of them required hospitalization. Although we initially thought that COVIDists would be needed for only a short period of time, the evolution of the disease is showing signs that it might be prolonged over the next several months. Hence, we are planning to continue COVIDist service for at least the next 6 months and reevaluate the need.
Hospital medicine leadership checked on COVIDists daily in regard to their physical health and, more importantly, their mental well-being. They were offered the chance to be taken off the schedule if they felt burned out, but no one wanted to come off their scheduled service before finishing their shifts. BlueCross MA recognized one of the COVIDists, Raghuveer Rakasi, MD, as a “hero on the front line.”4 In Dr. Rakasi’s words, “We took a nosedive into something without knowing its depth, and aware that we could have fatalities among ourselves. We took up new roles, faced new challenges, learned new things every day, evolving every step of the way. We had to change the way we practice medicine, finding new ways to treat patients, and protecting the workforce by limiting patient exposure, prioritizing investigations.” He added that “we have to adapt to a new normal; we should be prepared for this to come in waves. Putting aside our political views, we should stand united 6 feet apart, with a mask covering our brave faces, frequently washing our helping hands to overcome these uncertain times.”
Conclusion
The creation of a focused group of hospitalists called COVIDists and providing them with structured and rapid training (in various aspects of clinical care of COVID-19 patients, critical care/ventilator management, efficient and safe use of PPE) and daily information dissemination allowed our health system to prepare for the large volume of COVID-19 patients. It also helped in preserving the larger hospital workforce for a possible future surge.
The rapid development and implementation of the COVIDist strategy succeeded because of the intrinsic motivation of the providers to improve the outcomes of this high-risk patient population and the close collaboration of the stakeholders. Our institution remains successful in managing the pandemic in Western Massachusetts, with reserve capacity remaining even during the peak of the epidemic. A large part of this was because of creating and training a pool of COVIDists.
Dr. Medarametla is medical director, clinical operations, in the division of hospital medicine at Baystate Health, and assistant professor at University of Massachusetts, Worcester. Readers can contact him at [email protected]. Dr. Prabhakaran is unit medical director, geriatrics unit, in the division of hospital medicine at Baystate Health and assistant professor at University of Massachusetts. Dr. Bryson is associate program director of the Internal Medicine Residency at Baystate Health and assistant professor at University of Massachusetts. Dr. Umar is medical director, clinical operations, in the division of hospital medicine at Baystate Health. Dr. Natanasabapathy is division chief of hospital medicine at Baystate Health and assistant professor at University of Massachusetts.
References
1. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Updated Jun 10, 2020. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html.
2. Zhou F et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020 Mar 28;395(10229):1054-62.
3. Westafer LM et al. A transdisciplinary COVID-19 early respiratory intervention protocol: An implementation story. J Hosp Med. 2020 May 21;15(6):372-374.
4. Miller J. “Heroes on the front line: Dr. Raghuveer Rakasi.” Coverage. May 18, 2020. https://coverage.bluecrossma.com/article/heroes-front-line-dr-raghuveer-rakasi
Adapting to the demands placed on hospital resources by COVID-19
Adapting to the demands placed on hospital resources by COVID-19
The challenges posed by COVID-19 have crippled health care systems around the globe. By February 2020, the first outbreak in the United States had been set off in Washington State. We quickly became the world’s epicenter of the epidemic, with over 1.8 million patients and over 110,000 deaths.1 The rapidity of spread and the severity of the disease created a tremendous strain on resources. It blindsided policymakers and hospital administrators, which left little time to react to the challenges placed on hospital operations all over the country.
The necessity of a new care model
Although health systems in the United States are adept in managing complications of common seasonal viral respiratory illnesses, COVID-19 presented an entirely different challenge with its significantly higher mortality rate. A respiratory disease turning into a multiorgan disease that causes debilitating cardiac, renal, neurological, hematological, and psychosocial complications2 was not something we had experience managing effectively. Additional challenges included a massive surge of COVID-19 patients, a limited supply of personal protective equipment (PPE), an inadequate number of intensivists for managing the anticipated ventilated patients, and most importantly, the potential of losing some of our workforce if they became infected.
Based on the experiences in China and Italy, and various predictive models, the division of hospital medicine at Baystate Health quickly realized the necessity of a new model of care for COVID-19 patients. We came up with an elaborate plan to manage the disease burden and the strain on resources effectively. The measures we put in place could be broadly divided into three categories following the timeline of the disease: the preparatory phase, the execution phase, and the maintenance phase.
The preparatory phase: From “Hospitalists” to “COVIDists”
As in most hospitals around the country, hospitalists are the backbone of inpatient clinical operations at our health system. A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them.
COVIDists were trained in various treatment protocols and ongoing clinical trials. They were given refresher training in Advanced Cardiac Life Support (ACLS) and Fundamental Critical Care Support (FCCS) courses and were taught in critical care/ventilator management by the intensivists through rapid indoctrination in the ICU. All of them had their N-95 mask fitting updated and were trained in the safe donning and doffing of all kinds of PPE by PPE coaches. The palliative care team trained them in conducting end-of-life/code status discussions with a focus on being unable to speak with family members at the bedside. COVIDists were also assigned as Code Blue leaders for any “COVID code blue” in the hospital.
In addition to the rapid training course, COVID-related updates were disseminated daily using three different modalities: brief huddles at the start of the day with the COVIDists; a COVID-19 newsletter summarizing daily updates, new treatments, strategies, and policies; and a WhatsApp group for instantly broadcasting information to the COVIDists (Table 1).

The execution phase
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. The COVIDists were also undertaking the most challenging part of the care – talking to families about end-of-life issues and the futility of aggressive care in certain patients with preexisting conditions.
Some COVIDists were deployed to the ICU to work alongside the intensivists and became an invaluable resource in ICU management when the ICU census skyrocketed during the initial phase of the outbreak. This helped in tiding the health system over during the initial crisis. Within a short time, we shifted away from an early intubation strategy, and most of the ICU patients were managed in the intermediate care units on high flow oxygen along with the awake-proning protocol. The COVIDists exclusively managed these units. They led multidisciplinary rounds two times a day with the ICU, rapid response team (RRT), the palliative care team, and the nursing team. This step drastically decreased the number of intubations, RRT activations, reduced ICU census,3 and helped with hospital capacity and patient flow (Tables 2 and 3).

This strategy also helped build solidarity and camaraderie between all these groups, making the COVIDists feel that they were never alone and that the whole hospital supported them. We are currently evaluating clinical outcomes and attempting to identify effects on mortality, length of stay, days on the ventilator, and days in ICU.

The maintenance phase
It is already 2 months since the first devising COVIDists. There is no difference in sick callouts between COVIDists and non-COVIDists. One COVIDist and one non-COVIDist contracted the disease, but none of them required hospitalization. Although we initially thought that COVIDists would be needed for only a short period of time, the evolution of the disease is showing signs that it might be prolonged over the next several months. Hence, we are planning to continue COVIDist service for at least the next 6 months and reevaluate the need.
Hospital medicine leadership checked on COVIDists daily in regard to their physical health and, more importantly, their mental well-being. They were offered the chance to be taken off the schedule if they felt burned out, but no one wanted to come off their scheduled service before finishing their shifts. BlueCross MA recognized one of the COVIDists, Raghuveer Rakasi, MD, as a “hero on the front line.”4 In Dr. Rakasi’s words, “We took a nosedive into something without knowing its depth, and aware that we could have fatalities among ourselves. We took up new roles, faced new challenges, learned new things every day, evolving every step of the way. We had to change the way we practice medicine, finding new ways to treat patients, and protecting the workforce by limiting patient exposure, prioritizing investigations.” He added that “we have to adapt to a new normal; we should be prepared for this to come in waves. Putting aside our political views, we should stand united 6 feet apart, with a mask covering our brave faces, frequently washing our helping hands to overcome these uncertain times.”
Conclusion
The creation of a focused group of hospitalists called COVIDists and providing them with structured and rapid training (in various aspects of clinical care of COVID-19 patients, critical care/ventilator management, efficient and safe use of PPE) and daily information dissemination allowed our health system to prepare for the large volume of COVID-19 patients. It also helped in preserving the larger hospital workforce for a possible future surge.
The rapid development and implementation of the COVIDist strategy succeeded because of the intrinsic motivation of the providers to improve the outcomes of this high-risk patient population and the close collaboration of the stakeholders. Our institution remains successful in managing the pandemic in Western Massachusetts, with reserve capacity remaining even during the peak of the epidemic. A large part of this was because of creating and training a pool of COVIDists.
Dr. Medarametla is medical director, clinical operations, in the division of hospital medicine at Baystate Health, and assistant professor at University of Massachusetts, Worcester. Readers can contact him at [email protected]. Dr. Prabhakaran is unit medical director, geriatrics unit, in the division of hospital medicine at Baystate Health and assistant professor at University of Massachusetts. Dr. Bryson is associate program director of the Internal Medicine Residency at Baystate Health and assistant professor at University of Massachusetts. Dr. Umar is medical director, clinical operations, in the division of hospital medicine at Baystate Health. Dr. Natanasabapathy is division chief of hospital medicine at Baystate Health and assistant professor at University of Massachusetts.
References
1. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Updated Jun 10, 2020. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html.
2. Zhou F et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020 Mar 28;395(10229):1054-62.
3. Westafer LM et al. A transdisciplinary COVID-19 early respiratory intervention protocol: An implementation story. J Hosp Med. 2020 May 21;15(6):372-374.
4. Miller J. “Heroes on the front line: Dr. Raghuveer Rakasi.” Coverage. May 18, 2020. https://coverage.bluecrossma.com/article/heroes-front-line-dr-raghuveer-rakasi
The challenges posed by COVID-19 have crippled health care systems around the globe. By February 2020, the first outbreak in the United States had been set off in Washington State. We quickly became the world’s epicenter of the epidemic, with over 1.8 million patients and over 110,000 deaths.1 The rapidity of spread and the severity of the disease created a tremendous strain on resources. It blindsided policymakers and hospital administrators, which left little time to react to the challenges placed on hospital operations all over the country.
The necessity of a new care model
Although health systems in the United States are adept in managing complications of common seasonal viral respiratory illnesses, COVID-19 presented an entirely different challenge with its significantly higher mortality rate. A respiratory disease turning into a multiorgan disease that causes debilitating cardiac, renal, neurological, hematological, and psychosocial complications2 was not something we had experience managing effectively. Additional challenges included a massive surge of COVID-19 patients, a limited supply of personal protective equipment (PPE), an inadequate number of intensivists for managing the anticipated ventilated patients, and most importantly, the potential of losing some of our workforce if they became infected.
Based on the experiences in China and Italy, and various predictive models, the division of hospital medicine at Baystate Health quickly realized the necessity of a new model of care for COVID-19 patients. We came up with an elaborate plan to manage the disease burden and the strain on resources effectively. The measures we put in place could be broadly divided into three categories following the timeline of the disease: the preparatory phase, the execution phase, and the maintenance phase.
The preparatory phase: From “Hospitalists” to “COVIDists”
As in most hospitals around the country, hospitalists are the backbone of inpatient clinical operations at our health system. A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them.
COVIDists were trained in various treatment protocols and ongoing clinical trials. They were given refresher training in Advanced Cardiac Life Support (ACLS) and Fundamental Critical Care Support (FCCS) courses and were taught in critical care/ventilator management by the intensivists through rapid indoctrination in the ICU. All of them had their N-95 mask fitting updated and were trained in the safe donning and doffing of all kinds of PPE by PPE coaches. The palliative care team trained them in conducting end-of-life/code status discussions with a focus on being unable to speak with family members at the bedside. COVIDists were also assigned as Code Blue leaders for any “COVID code blue” in the hospital.
In addition to the rapid training course, COVID-related updates were disseminated daily using three different modalities: brief huddles at the start of the day with the COVIDists; a COVID-19 newsletter summarizing daily updates, new treatments, strategies, and policies; and a WhatsApp group for instantly broadcasting information to the COVIDists (Table 1).

The execution phase
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. The COVIDists were also undertaking the most challenging part of the care – talking to families about end-of-life issues and the futility of aggressive care in certain patients with preexisting conditions.
Some COVIDists were deployed to the ICU to work alongside the intensivists and became an invaluable resource in ICU management when the ICU census skyrocketed during the initial phase of the outbreak. This helped in tiding the health system over during the initial crisis. Within a short time, we shifted away from an early intubation strategy, and most of the ICU patients were managed in the intermediate care units on high flow oxygen along with the awake-proning protocol. The COVIDists exclusively managed these units. They led multidisciplinary rounds two times a day with the ICU, rapid response team (RRT), the palliative care team, and the nursing team. This step drastically decreased the number of intubations, RRT activations, reduced ICU census,3 and helped with hospital capacity and patient flow (Tables 2 and 3).

This strategy also helped build solidarity and camaraderie between all these groups, making the COVIDists feel that they were never alone and that the whole hospital supported them. We are currently evaluating clinical outcomes and attempting to identify effects on mortality, length of stay, days on the ventilator, and days in ICU.

The maintenance phase
It is already 2 months since the first devising COVIDists. There is no difference in sick callouts between COVIDists and non-COVIDists. One COVIDist and one non-COVIDist contracted the disease, but none of them required hospitalization. Although we initially thought that COVIDists would be needed for only a short period of time, the evolution of the disease is showing signs that it might be prolonged over the next several months. Hence, we are planning to continue COVIDist service for at least the next 6 months and reevaluate the need.
Hospital medicine leadership checked on COVIDists daily in regard to their physical health and, more importantly, their mental well-being. They were offered the chance to be taken off the schedule if they felt burned out, but no one wanted to come off their scheduled service before finishing their shifts. BlueCross MA recognized one of the COVIDists, Raghuveer Rakasi, MD, as a “hero on the front line.”4 In Dr. Rakasi’s words, “We took a nosedive into something without knowing its depth, and aware that we could have fatalities among ourselves. We took up new roles, faced new challenges, learned new things every day, evolving every step of the way. We had to change the way we practice medicine, finding new ways to treat patients, and protecting the workforce by limiting patient exposure, prioritizing investigations.” He added that “we have to adapt to a new normal; we should be prepared for this to come in waves. Putting aside our political views, we should stand united 6 feet apart, with a mask covering our brave faces, frequently washing our helping hands to overcome these uncertain times.”
Conclusion
The creation of a focused group of hospitalists called COVIDists and providing them with structured and rapid training (in various aspects of clinical care of COVID-19 patients, critical care/ventilator management, efficient and safe use of PPE) and daily information dissemination allowed our health system to prepare for the large volume of COVID-19 patients. It also helped in preserving the larger hospital workforce for a possible future surge.
The rapid development and implementation of the COVIDist strategy succeeded because of the intrinsic motivation of the providers to improve the outcomes of this high-risk patient population and the close collaboration of the stakeholders. Our institution remains successful in managing the pandemic in Western Massachusetts, with reserve capacity remaining even during the peak of the epidemic. A large part of this was because of creating and training a pool of COVIDists.
Dr. Medarametla is medical director, clinical operations, in the division of hospital medicine at Baystate Health, and assistant professor at University of Massachusetts, Worcester. Readers can contact him at [email protected]. Dr. Prabhakaran is unit medical director, geriatrics unit, in the division of hospital medicine at Baystate Health and assistant professor at University of Massachusetts. Dr. Bryson is associate program director of the Internal Medicine Residency at Baystate Health and assistant professor at University of Massachusetts. Dr. Umar is medical director, clinical operations, in the division of hospital medicine at Baystate Health. Dr. Natanasabapathy is division chief of hospital medicine at Baystate Health and assistant professor at University of Massachusetts.
References
1. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Updated Jun 10, 2020. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html.
2. Zhou F et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020 Mar 28;395(10229):1054-62.
3. Westafer LM et al. A transdisciplinary COVID-19 early respiratory intervention protocol: An implementation story. J Hosp Med. 2020 May 21;15(6):372-374.
4. Miller J. “Heroes on the front line: Dr. Raghuveer Rakasi.” Coverage. May 18, 2020. https://coverage.bluecrossma.com/article/heroes-front-line-dr-raghuveer-rakasi
Daily Recap: Lung ultrasound helps diagnose COVID-19 in kids, first treatment approved for adult-onset Still’s disease
Here are the stories our MDedge editors across specialties think you need to know about today:
Lung ultrasound works well in children with COVID-19
Lung ultrasound has “high concordance” with radiologic findings in children with COVID-19 and offers benefits over other imaging techniques, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” wrote Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy. “Secondly, when performed at the bedside, [lung ultrasound] allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].” The findings of the small, observational study were published in Pediatrics. Read more.
New hypertension definitions reveal preclampsia risk
Using the new clinical definitions of hypertension, pregnant women with even modest elevations in blood pressure are at increased risk for preeclampsia, according to results from a large retrospective cohort study. Elizabeth F. Sutton, PhD, of the University of Pittsburgh and colleagues looked at records from 18,162 women who had given birth to a single baby. The authors found preeclampsia risk increased with increasing blood pressure elevation. Among women with normal blood pressure before 20 weeks’ gestation, 5% had preeclampsia, while 7% of those with elevated blood pressure did, as did 12% of women with stage 1 hypertension and 30% of women with stage 2 hypertension. The increase in risk of preeclampsia was because of preterm preeclampsia in the women with elevated blood pressure. Preeclampsia researcher Mark Santillan, MD, PhD, of the University of Iowa in Iowa City, said in an interview that the results “open the door to considering these new blood pressure categories as a prognosticator” for preeclampsia. “This paper furthers the field by applying these new categories to hypertensive diseases in pregnancy, which are not well studied” in comparison to nonpregnant hypertensive states. Read more.
Face mask type matters when sterilizing
When sterilizing face masks, the type of mask and the method of sterilization have a bearing on subsequent filtration efficiency, according to new research published in JAMA Network Open. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks. With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%. Read more.
FDA approves first treatment for adult-onset Still’s disease
The Food and Drug Administration has expanded the indications for canakinumab (Ilaris) to include all patients with active Still’s disease older than 2 years, adding adult-onset Still’s disease (AOSD) to a previous approval for juvenile-onset Still’s disease, also known as systemic juvenile idiopathic arthritis (sJIA). That makes Ilaris the first approved treatment for AOSD. The results from a randomized, double-blind, placebo-controlled study of 36 patients with AOSD aged 22-70 years showed that the efficacy and safety data in AOSD were generally consistent with the results of a pooled analysis of sJIA patients, according to Novartis, which markets canakinumab. Read more.
Intranasal DHE shows promise in migraine
An intranasal form of dihydroergotamine (DHE) targeting the upper nasal region is safe and effective for the treatment of migraine, according to results from a phase 3 clinical trial. The new formulation could offer patients an at-home alternative to intramuscular infusions or intravenous injections currently used to deliver DHE. The STOP 301 phase 3 open-label safety and tolerability trial treated over 5,650 migraine attacks in 354 patients who self-administered INP104 for up to 52 weeks. They were provided up to three doses per week (1.45 mg in a dose of two puffs, one per nostril). A total of 66.3% of participants reported pain relief by 2 hours following a dose, and 38% had freedom from pain. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Lung ultrasound works well in children with COVID-19
Lung ultrasound has “high concordance” with radiologic findings in children with COVID-19 and offers benefits over other imaging techniques, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” wrote Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy. “Secondly, when performed at the bedside, [lung ultrasound] allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].” The findings of the small, observational study were published in Pediatrics. Read more.
New hypertension definitions reveal preclampsia risk
Using the new clinical definitions of hypertension, pregnant women with even modest elevations in blood pressure are at increased risk for preeclampsia, according to results from a large retrospective cohort study. Elizabeth F. Sutton, PhD, of the University of Pittsburgh and colleagues looked at records from 18,162 women who had given birth to a single baby. The authors found preeclampsia risk increased with increasing blood pressure elevation. Among women with normal blood pressure before 20 weeks’ gestation, 5% had preeclampsia, while 7% of those with elevated blood pressure did, as did 12% of women with stage 1 hypertension and 30% of women with stage 2 hypertension. The increase in risk of preeclampsia was because of preterm preeclampsia in the women with elevated blood pressure. Preeclampsia researcher Mark Santillan, MD, PhD, of the University of Iowa in Iowa City, said in an interview that the results “open the door to considering these new blood pressure categories as a prognosticator” for preeclampsia. “This paper furthers the field by applying these new categories to hypertensive diseases in pregnancy, which are not well studied” in comparison to nonpregnant hypertensive states. Read more.
Face mask type matters when sterilizing
When sterilizing face masks, the type of mask and the method of sterilization have a bearing on subsequent filtration efficiency, according to new research published in JAMA Network Open. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks. With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%. Read more.
FDA approves first treatment for adult-onset Still’s disease
The Food and Drug Administration has expanded the indications for canakinumab (Ilaris) to include all patients with active Still’s disease older than 2 years, adding adult-onset Still’s disease (AOSD) to a previous approval for juvenile-onset Still’s disease, also known as systemic juvenile idiopathic arthritis (sJIA). That makes Ilaris the first approved treatment for AOSD. The results from a randomized, double-blind, placebo-controlled study of 36 patients with AOSD aged 22-70 years showed that the efficacy and safety data in AOSD were generally consistent with the results of a pooled analysis of sJIA patients, according to Novartis, which markets canakinumab. Read more.
Intranasal DHE shows promise in migraine
An intranasal form of dihydroergotamine (DHE) targeting the upper nasal region is safe and effective for the treatment of migraine, according to results from a phase 3 clinical trial. The new formulation could offer patients an at-home alternative to intramuscular infusions or intravenous injections currently used to deliver DHE. The STOP 301 phase 3 open-label safety and tolerability trial treated over 5,650 migraine attacks in 354 patients who self-administered INP104 for up to 52 weeks. They were provided up to three doses per week (1.45 mg in a dose of two puffs, one per nostril). A total of 66.3% of participants reported pain relief by 2 hours following a dose, and 38% had freedom from pain. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Lung ultrasound works well in children with COVID-19
Lung ultrasound has “high concordance” with radiologic findings in children with COVID-19 and offers benefits over other imaging techniques, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” wrote Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy. “Secondly, when performed at the bedside, [lung ultrasound] allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].” The findings of the small, observational study were published in Pediatrics. Read more.
New hypertension definitions reveal preclampsia risk
Using the new clinical definitions of hypertension, pregnant women with even modest elevations in blood pressure are at increased risk for preeclampsia, according to results from a large retrospective cohort study. Elizabeth F. Sutton, PhD, of the University of Pittsburgh and colleagues looked at records from 18,162 women who had given birth to a single baby. The authors found preeclampsia risk increased with increasing blood pressure elevation. Among women with normal blood pressure before 20 weeks’ gestation, 5% had preeclampsia, while 7% of those with elevated blood pressure did, as did 12% of women with stage 1 hypertension and 30% of women with stage 2 hypertension. The increase in risk of preeclampsia was because of preterm preeclampsia in the women with elevated blood pressure. Preeclampsia researcher Mark Santillan, MD, PhD, of the University of Iowa in Iowa City, said in an interview that the results “open the door to considering these new blood pressure categories as a prognosticator” for preeclampsia. “This paper furthers the field by applying these new categories to hypertensive diseases in pregnancy, which are not well studied” in comparison to nonpregnant hypertensive states. Read more.
Face mask type matters when sterilizing
When sterilizing face masks, the type of mask and the method of sterilization have a bearing on subsequent filtration efficiency, according to new research published in JAMA Network Open. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks. With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%. Read more.
FDA approves first treatment for adult-onset Still’s disease
The Food and Drug Administration has expanded the indications for canakinumab (Ilaris) to include all patients with active Still’s disease older than 2 years, adding adult-onset Still’s disease (AOSD) to a previous approval for juvenile-onset Still’s disease, also known as systemic juvenile idiopathic arthritis (sJIA). That makes Ilaris the first approved treatment for AOSD. The results from a randomized, double-blind, placebo-controlled study of 36 patients with AOSD aged 22-70 years showed that the efficacy and safety data in AOSD were generally consistent with the results of a pooled analysis of sJIA patients, according to Novartis, which markets canakinumab. Read more.
Intranasal DHE shows promise in migraine
An intranasal form of dihydroergotamine (DHE) targeting the upper nasal region is safe and effective for the treatment of migraine, according to results from a phase 3 clinical trial. The new formulation could offer patients an at-home alternative to intramuscular infusions or intravenous injections currently used to deliver DHE. The STOP 301 phase 3 open-label safety and tolerability trial treated over 5,650 migraine attacks in 354 patients who self-administered INP104 for up to 52 weeks. They were provided up to three doses per week (1.45 mg in a dose of two puffs, one per nostril). A total of 66.3% of participants reported pain relief by 2 hours following a dose, and 38% had freedom from pain. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Dapagliflozin’s T2D renal protection extends to ‘fast decline’ of eGFR
Treatment of patients with type 2 diabetes with the SGLT2 inhibitor dapagliflozin led to a significant drop in the occurrence of ‘fast decline’ of renal function in more than 15,000 patients enrolled in the drug’s main cardiovascular outcome trial, another example of the potent renal protective effects of agents from this drug class.
Among patients with type 2 diabetes enrolled in the DECLARE-TIMI 58 trial, the incidence of a fast decline in renal function, defined as a drop in estimated glomerular filtration rate (eGFR) of at least 3 mL/min per 1.73 m2, was 27% among patients treated with dapagliflozin and 37% in control patients who received placebo, a statistically significant difference for this post-hoc analysis, Itamar Raz, MD, said at the virtual annual scientific sessions of the American Diabetes Association.
This finding, which adds to a long list of other renal function parameters reported to have been improved by treatment with sodium-glucose cotransporter 2 (SGLT2) inhibitors, “emphasizes the value of SGLT2 inhibitors as an important component of both prevention and treatment of chronic kidney disease among patients with type 2 diabetes,” said Dr. Raz, a diabetes researcher and professor of medicine at Hadassah University Hospital in Jerusalem.
The primary, prespecified renal outcomes in DECLARE-TIMI 58 were a cardiorenal composite outcome of sustained decline of at least 40% in eGFR to less than 60 mL/min per 1.73 m2, end-stage renal disease (defined as dialysis for at least 90 days, kidney transplantation, or confirmed sustained eGFR of less than 15 mL/min per 1.73 m2), or death from renal or cardiovascular causes; and a second prespecified renal-specific composite outcome that was the same except for excluding death from cardiovascular causes. The results showed that the cardiorenal outcome dropped by a statistically significant 24% with dapagliflozin treatment relative to control patients, and the renal-specific outcome fell by a statistically significant 47% with dapagliflozin relative to control patients (Lancet Diab Endocrinol. 2019 Aug 1;7[8];606-17).
The new findings on the incidence of fast decline in renal function help to further flesh out the scope of renal benefit exerted by SGLT2 inhibitors like dapagliflozin in patients with type 2 diabetes, said experts. Fast decline is a relatively recently devised measure of a high-risk, precipitous loss of renal function that has been defined as a drop of either 3 or 5 mL/min per 1.73 m2 per year (Kidney Int. 2017 Jun;91[6]:1300-11); for this analysis Dr. Raz and his associates used the less stringent definition.
Finding and treating ‘fast decliners’
The new report from Dr. Raz “confirms the original [renal] findings and looks to expand them to a particularly high risk group: the fast decliners,” commented Robert A. Gabbay, MD, chief science & medical officer of the ADA. “In some ways, the group of patients that we need to find a better treatment for most are those whose GFR declines quickly. We don’t always know who they are until after the fact, and studies have been looking for markers that might prospectively identify them,” he said in an interview.
The new analysis showed that dapagliflozin “was effective in this subgroup of patients. Furthermore, it didn’t matter if they had significant baseline disease or not. Even people with normal kidney function [at baseline] who were still fast decliners fared better with the drug than without it. This suggests that, if it can be confirmed in a prospective study, dapagliflozin might be effective very early in the course of treatment if we can identify who will be the fast decliners.”
Dr. Raz and his associates had the data necessary to calculate the rates of eGFR decline during the full follow-up period for 15,012 of the 17,160 patients enrolled in DECLARE-TIMI 58, and they found that 4,788 (32%) were fast decliners and 10,224 had a slower rate of renal deterioration. The average annual decline in eGFR during the period from 6 months after study entry through 4 years was 6.3 mL/min per 1.73 m2 per year (median of 5.1 mL/min per 1.73 m2 per year) among the fast decliners, and zero (median of 0.6 mL/min per 1.73 m2 per year) among the other patients.
Overcoming dapagliflozin’s initial eGFR reduction
The researchers focused on the 6-month to 4-year period of treatment as more representative of the impact of dapagliflozin because the SGLT2 inhibitors have an established pattern of triggering an initial, moderate decline in eGFR over roughly the first 6 months on the drug, which is similar to what happens to patients who start treatment with an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker.
“Some patients get as much as a 10% decline in eGFR” when SGLT2 inhibitor treatment starts, but “patients do better over time even with this initial hit,” the same way they do on drugs that act on the renin-angiotensin system, explained Silvio E. Inzucchi, MD, an endocrinologist and professor of medicine at Yale University in New Haven who has extensively studied the SGLT2 inhibitors.
The analyses reported by Dr. Raz showed that the protection against fast decline during the 6-month to 4-year period with dapagliflozin treatment was consistent across a range of patient subgroups regardless of age, duration of their type 2 diabetes, their baseline level of hyperglycemia, and their baseline eGFR. Nearly half the patients enrolled in DECLARE-TIMI 58 had an eGFR at baseline of at least 91 mL/min per 1.73 m2 and in this subgroup the incidence of fast decliners was 23% with dapagliflozin and 31% on placebo. Among the 45% of patients who began with an eGFR of 60-90 mL/min per 1.73 m2 the fast-decliner incidence was 32% and 43% when on or off dapagliflozin. Among the 7% of patients who entered with an eGFR below 60 mL/min per 1.73 m2, the fast-decliner incidence was 25% on dapagliflozin and 36% among controls. All the between-group differences were statistically significant.
The incidence of fast decliners was also lower with dapagliflozin treatment when the analysis included the entire first 4 years on treatment, including the first 6 months when SGLT2s usually spikes a loss of renal function. For the entire 4-year period, fast decline occurred among 34% of patients on dapagliflozin and in 37% of control patients, a statistically significant difference.
The mechanisms behind the consistent renal-protective effects of the SGLT2 inhibitors remain unclear right now, but likely seem related to the “perfect” diuretic action the drugs produce, said Dr. Inzucchi. “They’re not as hugely effective as diuretics, but they’re gentler.” While the SGLT2 inhibitors cause a modest amount of fluid loss ”for some reason they don’t activate the compensatory mechanisms that prevent further reductions in plasma volume,” a property that manifests as little or no change in catecholamines or renin-angiotensin activity, which sets this diuretic action apart from what happens with conventional diuretic drugs, he said in an interview.
In DECLARE-TIMI 58 treatment with dapagliflozin met its primary safety outcome of noninferiority to placebo with respect to major adverse cardiovascular events. The results failed to show statistically significant superiority for one of the primary efficacy endpoints, the rate of major adverse coronary events, but they did show significantly better performance for the second primary efficacy outcome of the rate of cardiovascular death or hospitalization for heart failure, which occurred in 4.9% of patients treated with dapagliflozin and in 5.8% of the control patients during a median follow-up of 4.2 years (N Engl J Med. 2019 Jan 24;380[4]:347-57).
DECLARE-TIMI 58 was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Raz has been an advisor to and speaker on behalf of AstraZeneca as well as several other companies. Dr. Gabbay had no relevant disclosures. Dr. Inzucchi has been a consultant to AstraZeneca, and also to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics.
SOURCE: Raz I et al. ADA 2020, Abstract 303-OR.
Treatment of patients with type 2 diabetes with the SGLT2 inhibitor dapagliflozin led to a significant drop in the occurrence of ‘fast decline’ of renal function in more than 15,000 patients enrolled in the drug’s main cardiovascular outcome trial, another example of the potent renal protective effects of agents from this drug class.
Among patients with type 2 diabetes enrolled in the DECLARE-TIMI 58 trial, the incidence of a fast decline in renal function, defined as a drop in estimated glomerular filtration rate (eGFR) of at least 3 mL/min per 1.73 m2, was 27% among patients treated with dapagliflozin and 37% in control patients who received placebo, a statistically significant difference for this post-hoc analysis, Itamar Raz, MD, said at the virtual annual scientific sessions of the American Diabetes Association.
This finding, which adds to a long list of other renal function parameters reported to have been improved by treatment with sodium-glucose cotransporter 2 (SGLT2) inhibitors, “emphasizes the value of SGLT2 inhibitors as an important component of both prevention and treatment of chronic kidney disease among patients with type 2 diabetes,” said Dr. Raz, a diabetes researcher and professor of medicine at Hadassah University Hospital in Jerusalem.
The primary, prespecified renal outcomes in DECLARE-TIMI 58 were a cardiorenal composite outcome of sustained decline of at least 40% in eGFR to less than 60 mL/min per 1.73 m2, end-stage renal disease (defined as dialysis for at least 90 days, kidney transplantation, or confirmed sustained eGFR of less than 15 mL/min per 1.73 m2), or death from renal or cardiovascular causes; and a second prespecified renal-specific composite outcome that was the same except for excluding death from cardiovascular causes. The results showed that the cardiorenal outcome dropped by a statistically significant 24% with dapagliflozin treatment relative to control patients, and the renal-specific outcome fell by a statistically significant 47% with dapagliflozin relative to control patients (Lancet Diab Endocrinol. 2019 Aug 1;7[8];606-17).
The new findings on the incidence of fast decline in renal function help to further flesh out the scope of renal benefit exerted by SGLT2 inhibitors like dapagliflozin in patients with type 2 diabetes, said experts. Fast decline is a relatively recently devised measure of a high-risk, precipitous loss of renal function that has been defined as a drop of either 3 or 5 mL/min per 1.73 m2 per year (Kidney Int. 2017 Jun;91[6]:1300-11); for this analysis Dr. Raz and his associates used the less stringent definition.
Finding and treating ‘fast decliners’
The new report from Dr. Raz “confirms the original [renal] findings and looks to expand them to a particularly high risk group: the fast decliners,” commented Robert A. Gabbay, MD, chief science & medical officer of the ADA. “In some ways, the group of patients that we need to find a better treatment for most are those whose GFR declines quickly. We don’t always know who they are until after the fact, and studies have been looking for markers that might prospectively identify them,” he said in an interview.
The new analysis showed that dapagliflozin “was effective in this subgroup of patients. Furthermore, it didn’t matter if they had significant baseline disease or not. Even people with normal kidney function [at baseline] who were still fast decliners fared better with the drug than without it. This suggests that, if it can be confirmed in a prospective study, dapagliflozin might be effective very early in the course of treatment if we can identify who will be the fast decliners.”
Dr. Raz and his associates had the data necessary to calculate the rates of eGFR decline during the full follow-up period for 15,012 of the 17,160 patients enrolled in DECLARE-TIMI 58, and they found that 4,788 (32%) were fast decliners and 10,224 had a slower rate of renal deterioration. The average annual decline in eGFR during the period from 6 months after study entry through 4 years was 6.3 mL/min per 1.73 m2 per year (median of 5.1 mL/min per 1.73 m2 per year) among the fast decliners, and zero (median of 0.6 mL/min per 1.73 m2 per year) among the other patients.
Overcoming dapagliflozin’s initial eGFR reduction
The researchers focused on the 6-month to 4-year period of treatment as more representative of the impact of dapagliflozin because the SGLT2 inhibitors have an established pattern of triggering an initial, moderate decline in eGFR over roughly the first 6 months on the drug, which is similar to what happens to patients who start treatment with an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker.
“Some patients get as much as a 10% decline in eGFR” when SGLT2 inhibitor treatment starts, but “patients do better over time even with this initial hit,” the same way they do on drugs that act on the renin-angiotensin system, explained Silvio E. Inzucchi, MD, an endocrinologist and professor of medicine at Yale University in New Haven who has extensively studied the SGLT2 inhibitors.
The analyses reported by Dr. Raz showed that the protection against fast decline during the 6-month to 4-year period with dapagliflozin treatment was consistent across a range of patient subgroups regardless of age, duration of their type 2 diabetes, their baseline level of hyperglycemia, and their baseline eGFR. Nearly half the patients enrolled in DECLARE-TIMI 58 had an eGFR at baseline of at least 91 mL/min per 1.73 m2 and in this subgroup the incidence of fast decliners was 23% with dapagliflozin and 31% on placebo. Among the 45% of patients who began with an eGFR of 60-90 mL/min per 1.73 m2 the fast-decliner incidence was 32% and 43% when on or off dapagliflozin. Among the 7% of patients who entered with an eGFR below 60 mL/min per 1.73 m2, the fast-decliner incidence was 25% on dapagliflozin and 36% among controls. All the between-group differences were statistically significant.
The incidence of fast decliners was also lower with dapagliflozin treatment when the analysis included the entire first 4 years on treatment, including the first 6 months when SGLT2s usually spikes a loss of renal function. For the entire 4-year period, fast decline occurred among 34% of patients on dapagliflozin and in 37% of control patients, a statistically significant difference.
The mechanisms behind the consistent renal-protective effects of the SGLT2 inhibitors remain unclear right now, but likely seem related to the “perfect” diuretic action the drugs produce, said Dr. Inzucchi. “They’re not as hugely effective as diuretics, but they’re gentler.” While the SGLT2 inhibitors cause a modest amount of fluid loss ”for some reason they don’t activate the compensatory mechanisms that prevent further reductions in plasma volume,” a property that manifests as little or no change in catecholamines or renin-angiotensin activity, which sets this diuretic action apart from what happens with conventional diuretic drugs, he said in an interview.
In DECLARE-TIMI 58 treatment with dapagliflozin met its primary safety outcome of noninferiority to placebo with respect to major adverse cardiovascular events. The results failed to show statistically significant superiority for one of the primary efficacy endpoints, the rate of major adverse coronary events, but they did show significantly better performance for the second primary efficacy outcome of the rate of cardiovascular death or hospitalization for heart failure, which occurred in 4.9% of patients treated with dapagliflozin and in 5.8% of the control patients during a median follow-up of 4.2 years (N Engl J Med. 2019 Jan 24;380[4]:347-57).
DECLARE-TIMI 58 was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Raz has been an advisor to and speaker on behalf of AstraZeneca as well as several other companies. Dr. Gabbay had no relevant disclosures. Dr. Inzucchi has been a consultant to AstraZeneca, and also to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics.
SOURCE: Raz I et al. ADA 2020, Abstract 303-OR.
Treatment of patients with type 2 diabetes with the SGLT2 inhibitor dapagliflozin led to a significant drop in the occurrence of ‘fast decline’ of renal function in more than 15,000 patients enrolled in the drug’s main cardiovascular outcome trial, another example of the potent renal protective effects of agents from this drug class.
Among patients with type 2 diabetes enrolled in the DECLARE-TIMI 58 trial, the incidence of a fast decline in renal function, defined as a drop in estimated glomerular filtration rate (eGFR) of at least 3 mL/min per 1.73 m2, was 27% among patients treated with dapagliflozin and 37% in control patients who received placebo, a statistically significant difference for this post-hoc analysis, Itamar Raz, MD, said at the virtual annual scientific sessions of the American Diabetes Association.
This finding, which adds to a long list of other renal function parameters reported to have been improved by treatment with sodium-glucose cotransporter 2 (SGLT2) inhibitors, “emphasizes the value of SGLT2 inhibitors as an important component of both prevention and treatment of chronic kidney disease among patients with type 2 diabetes,” said Dr. Raz, a diabetes researcher and professor of medicine at Hadassah University Hospital in Jerusalem.
The primary, prespecified renal outcomes in DECLARE-TIMI 58 were a cardiorenal composite outcome of sustained decline of at least 40% in eGFR to less than 60 mL/min per 1.73 m2, end-stage renal disease (defined as dialysis for at least 90 days, kidney transplantation, or confirmed sustained eGFR of less than 15 mL/min per 1.73 m2), or death from renal or cardiovascular causes; and a second prespecified renal-specific composite outcome that was the same except for excluding death from cardiovascular causes. The results showed that the cardiorenal outcome dropped by a statistically significant 24% with dapagliflozin treatment relative to control patients, and the renal-specific outcome fell by a statistically significant 47% with dapagliflozin relative to control patients (Lancet Diab Endocrinol. 2019 Aug 1;7[8];606-17).
The new findings on the incidence of fast decline in renal function help to further flesh out the scope of renal benefit exerted by SGLT2 inhibitors like dapagliflozin in patients with type 2 diabetes, said experts. Fast decline is a relatively recently devised measure of a high-risk, precipitous loss of renal function that has been defined as a drop of either 3 or 5 mL/min per 1.73 m2 per year (Kidney Int. 2017 Jun;91[6]:1300-11); for this analysis Dr. Raz and his associates used the less stringent definition.
Finding and treating ‘fast decliners’
The new report from Dr. Raz “confirms the original [renal] findings and looks to expand them to a particularly high risk group: the fast decliners,” commented Robert A. Gabbay, MD, chief science & medical officer of the ADA. “In some ways, the group of patients that we need to find a better treatment for most are those whose GFR declines quickly. We don’t always know who they are until after the fact, and studies have been looking for markers that might prospectively identify them,” he said in an interview.
The new analysis showed that dapagliflozin “was effective in this subgroup of patients. Furthermore, it didn’t matter if they had significant baseline disease or not. Even people with normal kidney function [at baseline] who were still fast decliners fared better with the drug than without it. This suggests that, if it can be confirmed in a prospective study, dapagliflozin might be effective very early in the course of treatment if we can identify who will be the fast decliners.”
Dr. Raz and his associates had the data necessary to calculate the rates of eGFR decline during the full follow-up period for 15,012 of the 17,160 patients enrolled in DECLARE-TIMI 58, and they found that 4,788 (32%) were fast decliners and 10,224 had a slower rate of renal deterioration. The average annual decline in eGFR during the period from 6 months after study entry through 4 years was 6.3 mL/min per 1.73 m2 per year (median of 5.1 mL/min per 1.73 m2 per year) among the fast decliners, and zero (median of 0.6 mL/min per 1.73 m2 per year) among the other patients.
Overcoming dapagliflozin’s initial eGFR reduction
The researchers focused on the 6-month to 4-year period of treatment as more representative of the impact of dapagliflozin because the SGLT2 inhibitors have an established pattern of triggering an initial, moderate decline in eGFR over roughly the first 6 months on the drug, which is similar to what happens to patients who start treatment with an angiotensin-converting enzyme inhibitor or angiotensin receptor blocker.
“Some patients get as much as a 10% decline in eGFR” when SGLT2 inhibitor treatment starts, but “patients do better over time even with this initial hit,” the same way they do on drugs that act on the renin-angiotensin system, explained Silvio E. Inzucchi, MD, an endocrinologist and professor of medicine at Yale University in New Haven who has extensively studied the SGLT2 inhibitors.
The analyses reported by Dr. Raz showed that the protection against fast decline during the 6-month to 4-year period with dapagliflozin treatment was consistent across a range of patient subgroups regardless of age, duration of their type 2 diabetes, their baseline level of hyperglycemia, and their baseline eGFR. Nearly half the patients enrolled in DECLARE-TIMI 58 had an eGFR at baseline of at least 91 mL/min per 1.73 m2 and in this subgroup the incidence of fast decliners was 23% with dapagliflozin and 31% on placebo. Among the 45% of patients who began with an eGFR of 60-90 mL/min per 1.73 m2 the fast-decliner incidence was 32% and 43% when on or off dapagliflozin. Among the 7% of patients who entered with an eGFR below 60 mL/min per 1.73 m2, the fast-decliner incidence was 25% on dapagliflozin and 36% among controls. All the between-group differences were statistically significant.
The incidence of fast decliners was also lower with dapagliflozin treatment when the analysis included the entire first 4 years on treatment, including the first 6 months when SGLT2s usually spikes a loss of renal function. For the entire 4-year period, fast decline occurred among 34% of patients on dapagliflozin and in 37% of control patients, a statistically significant difference.
The mechanisms behind the consistent renal-protective effects of the SGLT2 inhibitors remain unclear right now, but likely seem related to the “perfect” diuretic action the drugs produce, said Dr. Inzucchi. “They’re not as hugely effective as diuretics, but they’re gentler.” While the SGLT2 inhibitors cause a modest amount of fluid loss ”for some reason they don’t activate the compensatory mechanisms that prevent further reductions in plasma volume,” a property that manifests as little or no change in catecholamines or renin-angiotensin activity, which sets this diuretic action apart from what happens with conventional diuretic drugs, he said in an interview.
In DECLARE-TIMI 58 treatment with dapagliflozin met its primary safety outcome of noninferiority to placebo with respect to major adverse cardiovascular events. The results failed to show statistically significant superiority for one of the primary efficacy endpoints, the rate of major adverse coronary events, but they did show significantly better performance for the second primary efficacy outcome of the rate of cardiovascular death or hospitalization for heart failure, which occurred in 4.9% of patients treated with dapagliflozin and in 5.8% of the control patients during a median follow-up of 4.2 years (N Engl J Med. 2019 Jan 24;380[4]:347-57).
DECLARE-TIMI 58 was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Raz has been an advisor to and speaker on behalf of AstraZeneca as well as several other companies. Dr. Gabbay had no relevant disclosures. Dr. Inzucchi has been a consultant to AstraZeneca, and also to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics.
SOURCE: Raz I et al. ADA 2020, Abstract 303-OR.
FROM ADA 2020
Examining bias
I have an automatic preference for white people over black people. This isn’t my opinion; rather, it is my implicit bias test result. I didn’t believe it at first. Trying hard to not be biased, I took the test again and received the same outcome. My reaction – disbelief – is typical for those like me: White people who believe they are good human beings.
We’ve all watched in horror the acts of violence against blacks in the news. I was shocked and disgusted. It was easy to believe, however, that I am in no way complicit in the injustice and racism I was watching. I think I’m fair and without prejudice. I have never intentionally discriminated against someone. Wanting to help, I listened to my black colleagues, staff, and patients. What I learned made me uncomfortable.
Through all this news, I’d said little to my colleagues and friends. I cannot identify with how a black person has felt recently. What if I said the wrong thing or caused offense? The safe option is to say nothing. I learned that this is a common reaction and the least helpful. The advice from one black colleague was simple: “Just ask us.” Instead of ignoring the issue, she advised me to say: “I wonder what this experience has been like for you. Would you like to share?” And, if you mean it, to add, “I stand with you.” The latter should be followed by “What can I do to help?” Or, more powerfully, “What have I done that makes me complicit?”
Some of these conversations will be uncomfortable. If you want to help, then sit with that. Feeling uncomfortable might mean you are beginning to understand.
I also heard about the excellent book “White Fragility,” by Robin DiAngelo, PhD. In it, she argues that it is difficult for white people to talk about racism because of a tendency to react with defensiveness, guilt, and sometimes anger.
Many of the chapters in the book were easy to read because they didn’t apply to me: I don’t get angry in equity, inclusion, and diversity meetings. I don’t resent affirmative action programs. But then Dr. DiAngelo got me: I believed because I’m a good person and I have no intention of being racist, I’m absolved. Her argument was enlightening. Like all white people in the United States, I have benefited from white privilege. Yes, I’ve worked hard, but I also grew up in a white family with a college-educated father. That alone afforded me academic and financial advantages, which pushed me ahead. I’ve benefited from the status quo.
I have also failed to speak up when white friends carried on about how unnecessary affirmative action programs have become. I’ve sat with sealed lips when I’ve heard comments like “As a white male, it’s a lot harder to get into prestigious schools now.” Having no intention to harm doesn’t matter; plenty of harm is done unintentionally.
I also believed that because I have good intentions, I have no racial bias. I was wrong. The test I took online is an excellent tool to combat this blind spot. It was created by Harvard researchers and is available to everyone: Take a Test. It asks you to categorize faces as good or bad and records your tiny reaction times. Based on these and other questions, it provides feedback on your personal biases.
I was surprised that I have an implicit preference for white people over black people. That’s the point. Most of us are unaware of our biases and falsely believe we are free of them. I encourage you to take the test and learn about yourself. If the result makes you uncomfortable, then sit with it. Try not to be defensive, as I was, and accept that, even if you are a good person, you can become a better one.
Based on what I’ve learned and heard in the last few weeks, I’ve committed to a few things: To acknowledge the harm done to my black and brown colleagues and my complicity even by acts of omission. To not avoid uncomfortable feelings or uncomfortable conversations. As a leader, to use my organizational status to advocate. To stand by my partners of color not only in dramatic one-time marches but also against the everyday perpetrators of microaggressions. To create a safe space and invite my colleagues, staff, friends, and patients to share.
Standing up against racism is all our responsibility. As Dr. Martin Luther King Jr. reminds us: “In the end, we will remember not the words of our enemies, but the silence of our friends.”
Dr. Benabio is director of healthcare transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. He has no disclosures related to this column. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
I have an automatic preference for white people over black people. This isn’t my opinion; rather, it is my implicit bias test result. I didn’t believe it at first. Trying hard to not be biased, I took the test again and received the same outcome. My reaction – disbelief – is typical for those like me: White people who believe they are good human beings.
We’ve all watched in horror the acts of violence against blacks in the news. I was shocked and disgusted. It was easy to believe, however, that I am in no way complicit in the injustice and racism I was watching. I think I’m fair and without prejudice. I have never intentionally discriminated against someone. Wanting to help, I listened to my black colleagues, staff, and patients. What I learned made me uncomfortable.
Through all this news, I’d said little to my colleagues and friends. I cannot identify with how a black person has felt recently. What if I said the wrong thing or caused offense? The safe option is to say nothing. I learned that this is a common reaction and the least helpful. The advice from one black colleague was simple: “Just ask us.” Instead of ignoring the issue, she advised me to say: “I wonder what this experience has been like for you. Would you like to share?” And, if you mean it, to add, “I stand with you.” The latter should be followed by “What can I do to help?” Or, more powerfully, “What have I done that makes me complicit?”
Some of these conversations will be uncomfortable. If you want to help, then sit with that. Feeling uncomfortable might mean you are beginning to understand.
I also heard about the excellent book “White Fragility,” by Robin DiAngelo, PhD. In it, she argues that it is difficult for white people to talk about racism because of a tendency to react with defensiveness, guilt, and sometimes anger.
Many of the chapters in the book were easy to read because they didn’t apply to me: I don’t get angry in equity, inclusion, and diversity meetings. I don’t resent affirmative action programs. But then Dr. DiAngelo got me: I believed because I’m a good person and I have no intention of being racist, I’m absolved. Her argument was enlightening. Like all white people in the United States, I have benefited from white privilege. Yes, I’ve worked hard, but I also grew up in a white family with a college-educated father. That alone afforded me academic and financial advantages, which pushed me ahead. I’ve benefited from the status quo.
I have also failed to speak up when white friends carried on about how unnecessary affirmative action programs have become. I’ve sat with sealed lips when I’ve heard comments like “As a white male, it’s a lot harder to get into prestigious schools now.” Having no intention to harm doesn’t matter; plenty of harm is done unintentionally.
I also believed that because I have good intentions, I have no racial bias. I was wrong. The test I took online is an excellent tool to combat this blind spot. It was created by Harvard researchers and is available to everyone: Take a Test. It asks you to categorize faces as good or bad and records your tiny reaction times. Based on these and other questions, it provides feedback on your personal biases.
I was surprised that I have an implicit preference for white people over black people. That’s the point. Most of us are unaware of our biases and falsely believe we are free of them. I encourage you to take the test and learn about yourself. If the result makes you uncomfortable, then sit with it. Try not to be defensive, as I was, and accept that, even if you are a good person, you can become a better one.
Based on what I’ve learned and heard in the last few weeks, I’ve committed to a few things: To acknowledge the harm done to my black and brown colleagues and my complicity even by acts of omission. To not avoid uncomfortable feelings or uncomfortable conversations. As a leader, to use my organizational status to advocate. To stand by my partners of color not only in dramatic one-time marches but also against the everyday perpetrators of microaggressions. To create a safe space and invite my colleagues, staff, friends, and patients to share.
Standing up against racism is all our responsibility. As Dr. Martin Luther King Jr. reminds us: “In the end, we will remember not the words of our enemies, but the silence of our friends.”
Dr. Benabio is director of healthcare transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. He has no disclosures related to this column. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
I have an automatic preference for white people over black people. This isn’t my opinion; rather, it is my implicit bias test result. I didn’t believe it at first. Trying hard to not be biased, I took the test again and received the same outcome. My reaction – disbelief – is typical for those like me: White people who believe they are good human beings.
We’ve all watched in horror the acts of violence against blacks in the news. I was shocked and disgusted. It was easy to believe, however, that I am in no way complicit in the injustice and racism I was watching. I think I’m fair and without prejudice. I have never intentionally discriminated against someone. Wanting to help, I listened to my black colleagues, staff, and patients. What I learned made me uncomfortable.
Through all this news, I’d said little to my colleagues and friends. I cannot identify with how a black person has felt recently. What if I said the wrong thing or caused offense? The safe option is to say nothing. I learned that this is a common reaction and the least helpful. The advice from one black colleague was simple: “Just ask us.” Instead of ignoring the issue, she advised me to say: “I wonder what this experience has been like for you. Would you like to share?” And, if you mean it, to add, “I stand with you.” The latter should be followed by “What can I do to help?” Or, more powerfully, “What have I done that makes me complicit?”
Some of these conversations will be uncomfortable. If you want to help, then sit with that. Feeling uncomfortable might mean you are beginning to understand.
I also heard about the excellent book “White Fragility,” by Robin DiAngelo, PhD. In it, she argues that it is difficult for white people to talk about racism because of a tendency to react with defensiveness, guilt, and sometimes anger.
Many of the chapters in the book were easy to read because they didn’t apply to me: I don’t get angry in equity, inclusion, and diversity meetings. I don’t resent affirmative action programs. But then Dr. DiAngelo got me: I believed because I’m a good person and I have no intention of being racist, I’m absolved. Her argument was enlightening. Like all white people in the United States, I have benefited from white privilege. Yes, I’ve worked hard, but I also grew up in a white family with a college-educated father. That alone afforded me academic and financial advantages, which pushed me ahead. I’ve benefited from the status quo.
I have also failed to speak up when white friends carried on about how unnecessary affirmative action programs have become. I’ve sat with sealed lips when I’ve heard comments like “As a white male, it’s a lot harder to get into prestigious schools now.” Having no intention to harm doesn’t matter; plenty of harm is done unintentionally.
I also believed that because I have good intentions, I have no racial bias. I was wrong. The test I took online is an excellent tool to combat this blind spot. It was created by Harvard researchers and is available to everyone: Take a Test. It asks you to categorize faces as good or bad and records your tiny reaction times. Based on these and other questions, it provides feedback on your personal biases.
I was surprised that I have an implicit preference for white people over black people. That’s the point. Most of us are unaware of our biases and falsely believe we are free of them. I encourage you to take the test and learn about yourself. If the result makes you uncomfortable, then sit with it. Try not to be defensive, as I was, and accept that, even if you are a good person, you can become a better one.
Based on what I’ve learned and heard in the last few weeks, I’ve committed to a few things: To acknowledge the harm done to my black and brown colleagues and my complicity even by acts of omission. To not avoid uncomfortable feelings or uncomfortable conversations. As a leader, to use my organizational status to advocate. To stand by my partners of color not only in dramatic one-time marches but also against the everyday perpetrators of microaggressions. To create a safe space and invite my colleagues, staff, friends, and patients to share.
Standing up against racism is all our responsibility. As Dr. Martin Luther King Jr. reminds us: “In the end, we will remember not the words of our enemies, but the silence of our friends.”
Dr. Benabio is director of healthcare transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. He has no disclosures related to this column. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].