User login
Cardiology News is an independent news source that provides cardiologists with timely and relevant news and commentary about clinical developments and the impact of health care policy on cardiology and the cardiologist's practice. Cardiology News Digital Network is the online destination and multimedia properties of Cardiology News, the independent news publication for cardiologists. Cardiology news is the leading source of news and commentary about clinical developments in cardiology as well as health care policy and regulations that affect the cardiologist's practice. Cardiology News Digital Network is owned by Frontline Medical Communications.
Lung ultrasound works well in children with COVID-19
researchers wrote in Pediatrics.
They also noted the benefits that modality provides over other imaging techniques.
Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy, performed an observational study of eight children aged 0-17 years who were admitted to the hospital for COVID-19 between March 8 and 26, 2020. In seven of eight patients, the findings were concordant between imaging modalities; in the remaining patient, lung ultrasound (LUS) found an interstitial B-lines pattern that was not seen on radiography. In seven patients with pathologic ultrasound findings at baseline, the improvement or resolution of the subpleural consolidations or interstitial patterns was consistent with concomitant radiologic findings.
The authors cited the benefits of using point-of-care ultrasound instead of other modalities, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” they wrote. “Secondly, when performed at the bedside, LUS allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].”
One limitation of the study is the small sample size; however, the researchers felt the high concordance still suggests LUS is a reasonable method for COVID-19 patients.
There was no external funding for this study and the investigators had no relevant financial disclosures.
SOURCE: Denina M et al. Pediatrics. 2020 Jun. doi: 10.1542/peds.2020-1157.
researchers wrote in Pediatrics.
They also noted the benefits that modality provides over other imaging techniques.
Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy, performed an observational study of eight children aged 0-17 years who were admitted to the hospital for COVID-19 between March 8 and 26, 2020. In seven of eight patients, the findings were concordant between imaging modalities; in the remaining patient, lung ultrasound (LUS) found an interstitial B-lines pattern that was not seen on radiography. In seven patients with pathologic ultrasound findings at baseline, the improvement or resolution of the subpleural consolidations or interstitial patterns was consistent with concomitant radiologic findings.
The authors cited the benefits of using point-of-care ultrasound instead of other modalities, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” they wrote. “Secondly, when performed at the bedside, LUS allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].”
One limitation of the study is the small sample size; however, the researchers felt the high concordance still suggests LUS is a reasonable method for COVID-19 patients.
There was no external funding for this study and the investigators had no relevant financial disclosures.
SOURCE: Denina M et al. Pediatrics. 2020 Jun. doi: 10.1542/peds.2020-1157.
researchers wrote in Pediatrics.
They also noted the benefits that modality provides over other imaging techniques.
Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy, performed an observational study of eight children aged 0-17 years who were admitted to the hospital for COVID-19 between March 8 and 26, 2020. In seven of eight patients, the findings were concordant between imaging modalities; in the remaining patient, lung ultrasound (LUS) found an interstitial B-lines pattern that was not seen on radiography. In seven patients with pathologic ultrasound findings at baseline, the improvement or resolution of the subpleural consolidations or interstitial patterns was consistent with concomitant radiologic findings.
The authors cited the benefits of using point-of-care ultrasound instead of other modalities, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” they wrote. “Secondly, when performed at the bedside, LUS allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].”
One limitation of the study is the small sample size; however, the researchers felt the high concordance still suggests LUS is a reasonable method for COVID-19 patients.
There was no external funding for this study and the investigators had no relevant financial disclosures.
SOURCE: Denina M et al. Pediatrics. 2020 Jun. doi: 10.1542/peds.2020-1157.
FROM PEDIATRICS
VERTIS-CV: Ertugliflozin’s CV outcomes trial confirms SGLT2i benefits
The cardiovascular outcome trial results for a fourth sodium-glucose cotransporter 2 (SGLT2) inhibitor, ertugliflozin, were most notable for their consistency with the four prior, similar trials run on the three other drugs from this class on the U.S. market, canagliflozin, dapagliflozin, and empagliflozin, further solidifying the important role this drug class has recently taken on for patients with type 2 diabetes.
But the ertugliflozin results, which showed statistically significant superiority to placebo for just one endpoint, hospitalization for heart failure, made it unclear whether clinicians will regard ertugliflozin as the top agent from this class to prescribe.
“Our big takeaway is that the findings are consistent with what’s been seen in the other studies” of cardiovascular and renal outcomes in the EMPA-REG OUTCOME study of empagliflozin (N Engl J Med. 2015 Nov 26;373[22]:2117-28 ), the CANVAS (N Engl J Med. 2017 Aug 17;377[7]:644-57) and CREDENCE (N Engl J Med. 2019 June 13;380[24]:2295-306 ) studies of canagliflozin, and the DECLARE-TIMI 58 trial with dapagliflozin (N Engl J Med. 2019 Jan 24;380[4]:347-57), Christopher P. Cannon, MD, said at the virtual annual scientific sessions of the American Diabetes Association.
The cardiovascular outcome trials (CVOTs), mandated in 2008 by Food and Drug Administration guidance for type 2 diabetes drugs that is now in the process of undergoing an update, have had the main goal of proving safety, and the primary endpoint of the new ertugliflozin trial, VERTIS-CV, was noninferiority to placebo when used on top of standard type 2 diabetes medications for the combined endpoint of cardiovascular death, nonfatal MI, or nonfatal stroke.
Key findings
Both of the tested dosages of ertugliflozin, 5 mg and 15 mg daily, met this endpoint, with event rates over a median 3.0 years of follow-up that ran very close to the placebo rate, clearly proving noninferiority. But the results showed no suggestion of superiority in a study that randomized 5,499 patients to either of the ertugliflozin regimens and 2,747 to placebo, reported Dr. Cannon, a cardiologist and professor of medicine at Harvard Medical School, Boston.
The primary outcome also showed similar event rates for each component of the composite endpoint, and subgroup analysis showed consistent results from ertugliflozin, compared with placebo, regardless of study-cohort subdivision by demographic, clinical, or treatment factors.
The trial design called for a hierarchical sequence of secondary-outcome superiority analyses, starting with the impact of ertugliflozin on cardiovascular death or heart failure hospitalization, and for this outcome ertugliflozin showed a point estimate of a 12% relative risk reduction, compared with placebo-treated patients, but this difference was not statistically significant. This meant that all subsequent superiority analyses in this trial could only be hypothesis generating and not definitive.
This negated the statistical validity of the only statistically significant treatment difference between ertugliflozin and placebo seen in VERTIS-CV, for the outcome of hospitalization for heart failure, where ertugliflozin treatment cut this outcome by 30%, compared with placebo patients. The rate of cardiovascular death alone, as well as a renal composite endpoint each showed no statistically significant benefit of ertugliflozin, compared with placebo, although the renal endpoint came close, with ertugliflozin reducing the combined rate of renal death, need for dialysis, need for renal transplant, or a doubling of serum creatinine from baseline by 19%, compared with placebo (P = .08).
How results compare with prior CVOTs
In some ways, these results seemed to contrast with outcomes from the CVOTs for the other SGLT2 inhibitors, which all showed at least two statistically significant benefits for major endpoints when compared with placebo.
As summarized in a new meta-analysis of all the CVOTs by Darren K. McGuire, MD, a cardiologist and professor of medicine at the University of Texas, Dallas, both empagliflozin and canagliflozin showed statistically significant superiority compared with placebo for their trial’s primary, combined major cardiovascular adverse event endpoint, but dapagliflozin and ertugliflozin did not. Empagliflozin was the sole SGLT2 inhibitor to show a statistically significant cut in cardiovascular deaths, compared with placebo.
The primary, composite renal efficacy endpoints used in these trials were hardest to compare because they differed from study to study, but unlike ertugliflozin, all the other three drugs in the class showed a statistically significant improvement, compared with placebo, for their respective renal outcomes. On the other hand, the pattern of estimated glomerular filtration rates measured at multiple times during the various trials showed a high level of consistency across the CVOTs.
The greatest consistently among the major endpoints across the trials was for heart failure hospitalization. All four agents showed statistically significant improvements, compared with placebo, and all four had roughly equal magnitudes of effect, a cut in event rates by about one-third.
“The greatest magnitude of benefit is for reductions in heart failure hospitalizations and for renal outcomes,” with the heart failure outcomes the “most consistent” across the studies and the renal outcomes “largely consistent,” concluded Dr. McGuire. All together, the five CVOTs for these four SGLT2 inhibitors involved more than 46,000 patients.
“A lot of data suggest these are all class effects,” that are roughly similar across all four of these SGLT2 inhibitors, commented Mark E. Cooper, MBBS, a professor and head of the department of diabetes at Monash University, Melbourne, and designated discussant for the study.
There was “clear homogeneity” between the VERTIS-CV results for hospitalization for heart failure and the other CVOTs, he noted. “I think there is a difference” in the cardiovascular death outcomes, specifically the sole statistically significant, 38% relative risk reduction with empagliflozin that stood out from the other CVOTs, but this difference is “totally unexplained,” added Dr. Cooper. “To really determine differences you’d need head-to-head studies that are unlikely to happen.”
The results of new SGLT2 inhibitor meta-analysis appeared to also “support contemporary society recommendations to prioritize the use of SGLT2 inhibitors independent of glucose-control considerations in patients with type 2 diabetes with or at high risk for cardiovascular and renal complications,” said Dr. McGuire.
“The guidelines have it right. Now it’s on us to implement these treatments to appropriate patients,” concluded Dr. Cannon.
Study details
VERTIS-CV (Cardiovascular Outcomes Following Ertugliflozin Treatment in Type 2 Diabetes Mellitus Participants With Vascular Disease) enrolled and followed patients with type 2 diabetes and established atherosclerotic cardiovascular disease at 531 centers in 34 countries during December 2013–December 2019. Other effects from ertugliflozin recorded during the trial were consistent with prior studies of the drug, which is already FDA approved for glycemic control: Compared with placebo, ertugliflozin treatment reduced hemoglobin A1c by an average of 0.5% after 1 year, cut average body weight by about 2.5 kg after 1 year with additional modest weight loss, during subsequent years on the drug, and reduced systolic blood pressure by about 3 mm Hg after 1 year.
The drug’s safety profile was generally reassuring and consistent with prior studies of this drug and others in the class, with overall no increase in total adverse events or serious adverse events, compared with placebo, and modestly increased rates of urinary tract and mycotic genital infections.
VERTIS-CV was sponsored by Merck and Pfizer, the companies that market ertugliflozin (Steglatro). Dr. Cannon has received research funding and fees from Merck and Pfizer and from several other companies. Dr. McGuire has received honoraria from Merck, has been a consultant to Pfizer, and has had similar relationships with several other companies. Dr. Cooper has been an advisor to and received honoraria from Merck. He has also received honoraria from or been an adviser to AstraZeneca, Boehringer Ingelheim, Lilly, MundiPharma, Novartis, Reata, and Servier, and he has received research funding from Boehringer Ingelheim and Novo Nordisk.
The cardiovascular outcome trial results for a fourth sodium-glucose cotransporter 2 (SGLT2) inhibitor, ertugliflozin, were most notable for their consistency with the four prior, similar trials run on the three other drugs from this class on the U.S. market, canagliflozin, dapagliflozin, and empagliflozin, further solidifying the important role this drug class has recently taken on for patients with type 2 diabetes.
But the ertugliflozin results, which showed statistically significant superiority to placebo for just one endpoint, hospitalization for heart failure, made it unclear whether clinicians will regard ertugliflozin as the top agent from this class to prescribe.
“Our big takeaway is that the findings are consistent with what’s been seen in the other studies” of cardiovascular and renal outcomes in the EMPA-REG OUTCOME study of empagliflozin (N Engl J Med. 2015 Nov 26;373[22]:2117-28 ), the CANVAS (N Engl J Med. 2017 Aug 17;377[7]:644-57) and CREDENCE (N Engl J Med. 2019 June 13;380[24]:2295-306 ) studies of canagliflozin, and the DECLARE-TIMI 58 trial with dapagliflozin (N Engl J Med. 2019 Jan 24;380[4]:347-57), Christopher P. Cannon, MD, said at the virtual annual scientific sessions of the American Diabetes Association.
The cardiovascular outcome trials (CVOTs), mandated in 2008 by Food and Drug Administration guidance for type 2 diabetes drugs that is now in the process of undergoing an update, have had the main goal of proving safety, and the primary endpoint of the new ertugliflozin trial, VERTIS-CV, was noninferiority to placebo when used on top of standard type 2 diabetes medications for the combined endpoint of cardiovascular death, nonfatal MI, or nonfatal stroke.
Key findings
Both of the tested dosages of ertugliflozin, 5 mg and 15 mg daily, met this endpoint, with event rates over a median 3.0 years of follow-up that ran very close to the placebo rate, clearly proving noninferiority. But the results showed no suggestion of superiority in a study that randomized 5,499 patients to either of the ertugliflozin regimens and 2,747 to placebo, reported Dr. Cannon, a cardiologist and professor of medicine at Harvard Medical School, Boston.
The primary outcome also showed similar event rates for each component of the composite endpoint, and subgroup analysis showed consistent results from ertugliflozin, compared with placebo, regardless of study-cohort subdivision by demographic, clinical, or treatment factors.
The trial design called for a hierarchical sequence of secondary-outcome superiority analyses, starting with the impact of ertugliflozin on cardiovascular death or heart failure hospitalization, and for this outcome ertugliflozin showed a point estimate of a 12% relative risk reduction, compared with placebo-treated patients, but this difference was not statistically significant. This meant that all subsequent superiority analyses in this trial could only be hypothesis generating and not definitive.
This negated the statistical validity of the only statistically significant treatment difference between ertugliflozin and placebo seen in VERTIS-CV, for the outcome of hospitalization for heart failure, where ertugliflozin treatment cut this outcome by 30%, compared with placebo patients. The rate of cardiovascular death alone, as well as a renal composite endpoint each showed no statistically significant benefit of ertugliflozin, compared with placebo, although the renal endpoint came close, with ertugliflozin reducing the combined rate of renal death, need for dialysis, need for renal transplant, or a doubling of serum creatinine from baseline by 19%, compared with placebo (P = .08).
How results compare with prior CVOTs
In some ways, these results seemed to contrast with outcomes from the CVOTs for the other SGLT2 inhibitors, which all showed at least two statistically significant benefits for major endpoints when compared with placebo.
As summarized in a new meta-analysis of all the CVOTs by Darren K. McGuire, MD, a cardiologist and professor of medicine at the University of Texas, Dallas, both empagliflozin and canagliflozin showed statistically significant superiority compared with placebo for their trial’s primary, combined major cardiovascular adverse event endpoint, but dapagliflozin and ertugliflozin did not. Empagliflozin was the sole SGLT2 inhibitor to show a statistically significant cut in cardiovascular deaths, compared with placebo.
The primary, composite renal efficacy endpoints used in these trials were hardest to compare because they differed from study to study, but unlike ertugliflozin, all the other three drugs in the class showed a statistically significant improvement, compared with placebo, for their respective renal outcomes. On the other hand, the pattern of estimated glomerular filtration rates measured at multiple times during the various trials showed a high level of consistency across the CVOTs.
The greatest consistently among the major endpoints across the trials was for heart failure hospitalization. All four agents showed statistically significant improvements, compared with placebo, and all four had roughly equal magnitudes of effect, a cut in event rates by about one-third.
“The greatest magnitude of benefit is for reductions in heart failure hospitalizations and for renal outcomes,” with the heart failure outcomes the “most consistent” across the studies and the renal outcomes “largely consistent,” concluded Dr. McGuire. All together, the five CVOTs for these four SGLT2 inhibitors involved more than 46,000 patients.
“A lot of data suggest these are all class effects,” that are roughly similar across all four of these SGLT2 inhibitors, commented Mark E. Cooper, MBBS, a professor and head of the department of diabetes at Monash University, Melbourne, and designated discussant for the study.
There was “clear homogeneity” between the VERTIS-CV results for hospitalization for heart failure and the other CVOTs, he noted. “I think there is a difference” in the cardiovascular death outcomes, specifically the sole statistically significant, 38% relative risk reduction with empagliflozin that stood out from the other CVOTs, but this difference is “totally unexplained,” added Dr. Cooper. “To really determine differences you’d need head-to-head studies that are unlikely to happen.”
The results of new SGLT2 inhibitor meta-analysis appeared to also “support contemporary society recommendations to prioritize the use of SGLT2 inhibitors independent of glucose-control considerations in patients with type 2 diabetes with or at high risk for cardiovascular and renal complications,” said Dr. McGuire.
“The guidelines have it right. Now it’s on us to implement these treatments to appropriate patients,” concluded Dr. Cannon.
Study details
VERTIS-CV (Cardiovascular Outcomes Following Ertugliflozin Treatment in Type 2 Diabetes Mellitus Participants With Vascular Disease) enrolled and followed patients with type 2 diabetes and established atherosclerotic cardiovascular disease at 531 centers in 34 countries during December 2013–December 2019. Other effects from ertugliflozin recorded during the trial were consistent with prior studies of the drug, which is already FDA approved for glycemic control: Compared with placebo, ertugliflozin treatment reduced hemoglobin A1c by an average of 0.5% after 1 year, cut average body weight by about 2.5 kg after 1 year with additional modest weight loss, during subsequent years on the drug, and reduced systolic blood pressure by about 3 mm Hg after 1 year.
The drug’s safety profile was generally reassuring and consistent with prior studies of this drug and others in the class, with overall no increase in total adverse events or serious adverse events, compared with placebo, and modestly increased rates of urinary tract and mycotic genital infections.
VERTIS-CV was sponsored by Merck and Pfizer, the companies that market ertugliflozin (Steglatro). Dr. Cannon has received research funding and fees from Merck and Pfizer and from several other companies. Dr. McGuire has received honoraria from Merck, has been a consultant to Pfizer, and has had similar relationships with several other companies. Dr. Cooper has been an advisor to and received honoraria from Merck. He has also received honoraria from or been an adviser to AstraZeneca, Boehringer Ingelheim, Lilly, MundiPharma, Novartis, Reata, and Servier, and he has received research funding from Boehringer Ingelheim and Novo Nordisk.
The cardiovascular outcome trial results for a fourth sodium-glucose cotransporter 2 (SGLT2) inhibitor, ertugliflozin, were most notable for their consistency with the four prior, similar trials run on the three other drugs from this class on the U.S. market, canagliflozin, dapagliflozin, and empagliflozin, further solidifying the important role this drug class has recently taken on for patients with type 2 diabetes.
But the ertugliflozin results, which showed statistically significant superiority to placebo for just one endpoint, hospitalization for heart failure, made it unclear whether clinicians will regard ertugliflozin as the top agent from this class to prescribe.
“Our big takeaway is that the findings are consistent with what’s been seen in the other studies” of cardiovascular and renal outcomes in the EMPA-REG OUTCOME study of empagliflozin (N Engl J Med. 2015 Nov 26;373[22]:2117-28 ), the CANVAS (N Engl J Med. 2017 Aug 17;377[7]:644-57) and CREDENCE (N Engl J Med. 2019 June 13;380[24]:2295-306 ) studies of canagliflozin, and the DECLARE-TIMI 58 trial with dapagliflozin (N Engl J Med. 2019 Jan 24;380[4]:347-57), Christopher P. Cannon, MD, said at the virtual annual scientific sessions of the American Diabetes Association.
The cardiovascular outcome trials (CVOTs), mandated in 2008 by Food and Drug Administration guidance for type 2 diabetes drugs that is now in the process of undergoing an update, have had the main goal of proving safety, and the primary endpoint of the new ertugliflozin trial, VERTIS-CV, was noninferiority to placebo when used on top of standard type 2 diabetes medications for the combined endpoint of cardiovascular death, nonfatal MI, or nonfatal stroke.
Key findings
Both of the tested dosages of ertugliflozin, 5 mg and 15 mg daily, met this endpoint, with event rates over a median 3.0 years of follow-up that ran very close to the placebo rate, clearly proving noninferiority. But the results showed no suggestion of superiority in a study that randomized 5,499 patients to either of the ertugliflozin regimens and 2,747 to placebo, reported Dr. Cannon, a cardiologist and professor of medicine at Harvard Medical School, Boston.
The primary outcome also showed similar event rates for each component of the composite endpoint, and subgroup analysis showed consistent results from ertugliflozin, compared with placebo, regardless of study-cohort subdivision by demographic, clinical, or treatment factors.
The trial design called for a hierarchical sequence of secondary-outcome superiority analyses, starting with the impact of ertugliflozin on cardiovascular death or heart failure hospitalization, and for this outcome ertugliflozin showed a point estimate of a 12% relative risk reduction, compared with placebo-treated patients, but this difference was not statistically significant. This meant that all subsequent superiority analyses in this trial could only be hypothesis generating and not definitive.
This negated the statistical validity of the only statistically significant treatment difference between ertugliflozin and placebo seen in VERTIS-CV, for the outcome of hospitalization for heart failure, where ertugliflozin treatment cut this outcome by 30%, compared with placebo patients. The rate of cardiovascular death alone, as well as a renal composite endpoint each showed no statistically significant benefit of ertugliflozin, compared with placebo, although the renal endpoint came close, with ertugliflozin reducing the combined rate of renal death, need for dialysis, need for renal transplant, or a doubling of serum creatinine from baseline by 19%, compared with placebo (P = .08).
How results compare with prior CVOTs
In some ways, these results seemed to contrast with outcomes from the CVOTs for the other SGLT2 inhibitors, which all showed at least two statistically significant benefits for major endpoints when compared with placebo.
As summarized in a new meta-analysis of all the CVOTs by Darren K. McGuire, MD, a cardiologist and professor of medicine at the University of Texas, Dallas, both empagliflozin and canagliflozin showed statistically significant superiority compared with placebo for their trial’s primary, combined major cardiovascular adverse event endpoint, but dapagliflozin and ertugliflozin did not. Empagliflozin was the sole SGLT2 inhibitor to show a statistically significant cut in cardiovascular deaths, compared with placebo.
The primary, composite renal efficacy endpoints used in these trials were hardest to compare because they differed from study to study, but unlike ertugliflozin, all the other three drugs in the class showed a statistically significant improvement, compared with placebo, for their respective renal outcomes. On the other hand, the pattern of estimated glomerular filtration rates measured at multiple times during the various trials showed a high level of consistency across the CVOTs.
The greatest consistently among the major endpoints across the trials was for heart failure hospitalization. All four agents showed statistically significant improvements, compared with placebo, and all four had roughly equal magnitudes of effect, a cut in event rates by about one-third.
“The greatest magnitude of benefit is for reductions in heart failure hospitalizations and for renal outcomes,” with the heart failure outcomes the “most consistent” across the studies and the renal outcomes “largely consistent,” concluded Dr. McGuire. All together, the five CVOTs for these four SGLT2 inhibitors involved more than 46,000 patients.
“A lot of data suggest these are all class effects,” that are roughly similar across all four of these SGLT2 inhibitors, commented Mark E. Cooper, MBBS, a professor and head of the department of diabetes at Monash University, Melbourne, and designated discussant for the study.
There was “clear homogeneity” between the VERTIS-CV results for hospitalization for heart failure and the other CVOTs, he noted. “I think there is a difference” in the cardiovascular death outcomes, specifically the sole statistically significant, 38% relative risk reduction with empagliflozin that stood out from the other CVOTs, but this difference is “totally unexplained,” added Dr. Cooper. “To really determine differences you’d need head-to-head studies that are unlikely to happen.”
The results of new SGLT2 inhibitor meta-analysis appeared to also “support contemporary society recommendations to prioritize the use of SGLT2 inhibitors independent of glucose-control considerations in patients with type 2 diabetes with or at high risk for cardiovascular and renal complications,” said Dr. McGuire.
“The guidelines have it right. Now it’s on us to implement these treatments to appropriate patients,” concluded Dr. Cannon.
Study details
VERTIS-CV (Cardiovascular Outcomes Following Ertugliflozin Treatment in Type 2 Diabetes Mellitus Participants With Vascular Disease) enrolled and followed patients with type 2 diabetes and established atherosclerotic cardiovascular disease at 531 centers in 34 countries during December 2013–December 2019. Other effects from ertugliflozin recorded during the trial were consistent with prior studies of the drug, which is already FDA approved for glycemic control: Compared with placebo, ertugliflozin treatment reduced hemoglobin A1c by an average of 0.5% after 1 year, cut average body weight by about 2.5 kg after 1 year with additional modest weight loss, during subsequent years on the drug, and reduced systolic blood pressure by about 3 mm Hg after 1 year.
The drug’s safety profile was generally reassuring and consistent with prior studies of this drug and others in the class, with overall no increase in total adverse events or serious adverse events, compared with placebo, and modestly increased rates of urinary tract and mycotic genital infections.
VERTIS-CV was sponsored by Merck and Pfizer, the companies that market ertugliflozin (Steglatro). Dr. Cannon has received research funding and fees from Merck and Pfizer and from several other companies. Dr. McGuire has received honoraria from Merck, has been a consultant to Pfizer, and has had similar relationships with several other companies. Dr. Cooper has been an advisor to and received honoraria from Merck. He has also received honoraria from or been an adviser to AstraZeneca, Boehringer Ingelheim, Lilly, MundiPharma, Novartis, Reata, and Servier, and he has received research funding from Boehringer Ingelheim and Novo Nordisk.
FROM ADA 2020
Preventing arrhythmias and QTc prolongation in COVID-19 patients on psychotropics
Over the last few weeks, several conflicting reports about the efficacy of SARS-CoV-2 treatments have emerged, including high-profile papers that were placed in the limelight and groundbreaking retractions that were issued by the Lancet and New England Journal of Medicine, involving the potential dangers of COVID therapy with findings derived from the Surgisphere database. Hydroxychloroquine has garnered considerable media attention and was touted earlier by President Trump for its therapeutic effects.1 Naturally, there are political connotations associated with the agent, and it is unlikely that hydroxychloroquine will be supplanted in the near future as ongoing clinical trials have demonstrated mixed results amid the controversy.
As clinicians navigating unchartered territory within the hospital setting, we have to come to terms with these new challenges, tailoring treatment protocols accordingly with the best clinical practices in mind. Patients with preexisting mental health conditions and who are being treated for COVID-19 are particularly susceptible to clinical deterioration. Recent studies have indicated that psychiatric patients are more prone to feelings of isolation and/or estrangement as well as exacerbation of symptoms such as paranoia.2 Even more concerning is the medication regimen, namely, the novel combination therapies that arise when agents such as hydroxychloroquine are used in tandem with certain antipsychotics or antidepressants.
What’s at stake for COVID-19–positive mental health care patients?
Although the efficacy of hydroxychloroquine is currently being investigated,3 the antimalarial is usually prescribed in tandem with azithromycin for people with COVID-19. The National Institute of Allergy and Infectious Diseases has advised against that particular combination therapy because of ongoing concerns about toxicities.3,4
In another study, azithromycin was effectively substituted with doxycycline to help minimize systemic effects for patients with cardiac and/or pulmonary issues.5 Azithromycin is notorious in the literature for influencing the electrical activity of the heart with the potential for fatal arrhythmia and sudden cardiac death in individuals at risk for cardiovascular disease.5,6,7 It should be noted that both of these commonly prescribed COVID-19 medications (for example, hydroxychloroquine and azithromycin) could lead to QT interval prolongation especially within the context of combination therapy. This is largely concerning for psychiatrists and various other mental health practitioners for the following reasons: (1) higher rates of metabolic syndrome and cardiovascular diseases among psychiatric patients8 and/or (2) effects of certain antipsychotics (for example, IV haloperidol, thioridazine, and ziprasidone) and antidepressants (for example, citalopram and escitalopram) on the QT interval.9
SARS-CoV-2 and clinical judgment: Evaluating patients at higher risk
Although COVID-19 medication guidelines are still being actively developed, hydroxychloroquine appears to be commonly prescribed by physicians. The medication is known myriad untoward effects, including potential behavioral dysfunction (for example, irritability, agitation, suicidal ideation)10 as well as the aforementioned issues concerning arrhythmia (for example, torsades de pointes). Health care professionals might not have much control over the choice of COVID-19 agents because of a lack of available resources or limited options, but they can exercise clinical judgment with respect to selecting the appropriate psychotropic medications.
Treatment recommendations
1. Establish a baseline EKG
A baseline 12-lead EKG is the standard of care for patients currently being screened for COVID-19. It is necessary to rule out the presence of an underlying cardiovascular disease or a rhythm irregularity. A prolonged QTc interval is generally regarded as being around greater than 450-470 msecs with variations attributable to gender;11 numerous studies have affirmed that the risk of acquiring torsades de pointes is substantial when the QTc interval exceeds 500 msecs.12
2. Medical management and risk assessment
Commonly prescribed antipsychotics such as IV haloperidol and ziprasidone are known for exerting a negative effect on the interval and should readily be substituted with other agents in patients who are being treated for COVID-19; the combination of these antipsychotics alongside some COVID-19 medication regimens (for example, hydroxychloroquine/azithromycin) might prove to be fatal. The same logic applies to COVID-19 patients previously on antidepressant therapeutics such as citalopram and escitalopram.
3. Embrace an individually tailored approach to therapeutics
While American Psychiatric Association guidelines historically supported a cessation or reduction in the offending agent under normal circumstances,12 our team is recommending that the psychotropics associated with QTc interval prolongation are discontinued altogether (or substituted with a low-risk agent) in the event that a patient presents with suspected COVID-19. However, after the patients tests negative with COVID-19, they may resume therapy as indicated under the discretion of the mental health practitioner.
References
1. Offard C. “Lancet, NEJM Retract Surgisphere Studies on COVID-19 Patients.” The Scientist Magazine. 2020 Jun 4.
2. Shigemura J et al. Psychiatry Clin Neurosci. 2020 Apr;74(4):281-2.
3. Keshtkar-Jahromi M and Bavari S. Am J Trop Med Hyg. 2020 May;102(5):932-3.
4. Palca J. “NIH panel recommends against drug combination promoted by Trump for COVID-19.” NPR. 2020 Apr 21.
5. Mongelli L. “Long Island doctor tries new twist on hydroxychloroquine for elderly COVID-19 patients.” New York Post. 2020 Apr 4.
6. Hancox JC et al. Ther Adv Infect Dis. 2013 Oct;(5):155-65.
7. Giudicessi JR and Ackerman MJ. Cleve Clin J Med. 2013 Sep;80(9):539-44.
8. Casey DE. Am J Med. 2005 Apr 1;118(Suppl 2):15S-22S.
9. Beach SR et al. Psychosomatics. 2013 Jan 1;54(1):1-3.
10. Bogaczewicz A and Sobów T. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-4.
11. Chohan PS et al. Pak J Med Sci. 2015 Sep-Oct;31(5):1269-71.
12. Lieberman JA et al. APA guidance on the use of antipsychotic drugs and cardiac sudden death. NYS Office of Mental Health. 2012.
Dr. Faisal A. Islam is medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Faisal Islam disclosed no relevant financial relationships.
Dr. Mohammed Islam is affiliated with the department of psychiatry at the Interfaith Medical Center, New York. He disclosed no relevant financial relationships.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He disclosed no relevant financial relationships.
Over the last few weeks, several conflicting reports about the efficacy of SARS-CoV-2 treatments have emerged, including high-profile papers that were placed in the limelight and groundbreaking retractions that were issued by the Lancet and New England Journal of Medicine, involving the potential dangers of COVID therapy with findings derived from the Surgisphere database. Hydroxychloroquine has garnered considerable media attention and was touted earlier by President Trump for its therapeutic effects.1 Naturally, there are political connotations associated with the agent, and it is unlikely that hydroxychloroquine will be supplanted in the near future as ongoing clinical trials have demonstrated mixed results amid the controversy.
As clinicians navigating unchartered territory within the hospital setting, we have to come to terms with these new challenges, tailoring treatment protocols accordingly with the best clinical practices in mind. Patients with preexisting mental health conditions and who are being treated for COVID-19 are particularly susceptible to clinical deterioration. Recent studies have indicated that psychiatric patients are more prone to feelings of isolation and/or estrangement as well as exacerbation of symptoms such as paranoia.2 Even more concerning is the medication regimen, namely, the novel combination therapies that arise when agents such as hydroxychloroquine are used in tandem with certain antipsychotics or antidepressants.
What’s at stake for COVID-19–positive mental health care patients?
Although the efficacy of hydroxychloroquine is currently being investigated,3 the antimalarial is usually prescribed in tandem with azithromycin for people with COVID-19. The National Institute of Allergy and Infectious Diseases has advised against that particular combination therapy because of ongoing concerns about toxicities.3,4
In another study, azithromycin was effectively substituted with doxycycline to help minimize systemic effects for patients with cardiac and/or pulmonary issues.5 Azithromycin is notorious in the literature for influencing the electrical activity of the heart with the potential for fatal arrhythmia and sudden cardiac death in individuals at risk for cardiovascular disease.5,6,7 It should be noted that both of these commonly prescribed COVID-19 medications (for example, hydroxychloroquine and azithromycin) could lead to QT interval prolongation especially within the context of combination therapy. This is largely concerning for psychiatrists and various other mental health practitioners for the following reasons: (1) higher rates of metabolic syndrome and cardiovascular diseases among psychiatric patients8 and/or (2) effects of certain antipsychotics (for example, IV haloperidol, thioridazine, and ziprasidone) and antidepressants (for example, citalopram and escitalopram) on the QT interval.9
SARS-CoV-2 and clinical judgment: Evaluating patients at higher risk
Although COVID-19 medication guidelines are still being actively developed, hydroxychloroquine appears to be commonly prescribed by physicians. The medication is known myriad untoward effects, including potential behavioral dysfunction (for example, irritability, agitation, suicidal ideation)10 as well as the aforementioned issues concerning arrhythmia (for example, torsades de pointes). Health care professionals might not have much control over the choice of COVID-19 agents because of a lack of available resources or limited options, but they can exercise clinical judgment with respect to selecting the appropriate psychotropic medications.
Treatment recommendations
1. Establish a baseline EKG
A baseline 12-lead EKG is the standard of care for patients currently being screened for COVID-19. It is necessary to rule out the presence of an underlying cardiovascular disease or a rhythm irregularity. A prolonged QTc interval is generally regarded as being around greater than 450-470 msecs with variations attributable to gender;11 numerous studies have affirmed that the risk of acquiring torsades de pointes is substantial when the QTc interval exceeds 500 msecs.12
2. Medical management and risk assessment
Commonly prescribed antipsychotics such as IV haloperidol and ziprasidone are known for exerting a negative effect on the interval and should readily be substituted with other agents in patients who are being treated for COVID-19; the combination of these antipsychotics alongside some COVID-19 medication regimens (for example, hydroxychloroquine/azithromycin) might prove to be fatal. The same logic applies to COVID-19 patients previously on antidepressant therapeutics such as citalopram and escitalopram.
3. Embrace an individually tailored approach to therapeutics
While American Psychiatric Association guidelines historically supported a cessation or reduction in the offending agent under normal circumstances,12 our team is recommending that the psychotropics associated with QTc interval prolongation are discontinued altogether (or substituted with a low-risk agent) in the event that a patient presents with suspected COVID-19. However, after the patients tests negative with COVID-19, they may resume therapy as indicated under the discretion of the mental health practitioner.
References
1. Offard C. “Lancet, NEJM Retract Surgisphere Studies on COVID-19 Patients.” The Scientist Magazine. 2020 Jun 4.
2. Shigemura J et al. Psychiatry Clin Neurosci. 2020 Apr;74(4):281-2.
3. Keshtkar-Jahromi M and Bavari S. Am J Trop Med Hyg. 2020 May;102(5):932-3.
4. Palca J. “NIH panel recommends against drug combination promoted by Trump for COVID-19.” NPR. 2020 Apr 21.
5. Mongelli L. “Long Island doctor tries new twist on hydroxychloroquine for elderly COVID-19 patients.” New York Post. 2020 Apr 4.
6. Hancox JC et al. Ther Adv Infect Dis. 2013 Oct;(5):155-65.
7. Giudicessi JR and Ackerman MJ. Cleve Clin J Med. 2013 Sep;80(9):539-44.
8. Casey DE. Am J Med. 2005 Apr 1;118(Suppl 2):15S-22S.
9. Beach SR et al. Psychosomatics. 2013 Jan 1;54(1):1-3.
10. Bogaczewicz A and Sobów T. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-4.
11. Chohan PS et al. Pak J Med Sci. 2015 Sep-Oct;31(5):1269-71.
12. Lieberman JA et al. APA guidance on the use of antipsychotic drugs and cardiac sudden death. NYS Office of Mental Health. 2012.
Dr. Faisal A. Islam is medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Faisal Islam disclosed no relevant financial relationships.
Dr. Mohammed Islam is affiliated with the department of psychiatry at the Interfaith Medical Center, New York. He disclosed no relevant financial relationships.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He disclosed no relevant financial relationships.
Over the last few weeks, several conflicting reports about the efficacy of SARS-CoV-2 treatments have emerged, including high-profile papers that were placed in the limelight and groundbreaking retractions that were issued by the Lancet and New England Journal of Medicine, involving the potential dangers of COVID therapy with findings derived from the Surgisphere database. Hydroxychloroquine has garnered considerable media attention and was touted earlier by President Trump for its therapeutic effects.1 Naturally, there are political connotations associated with the agent, and it is unlikely that hydroxychloroquine will be supplanted in the near future as ongoing clinical trials have demonstrated mixed results amid the controversy.
As clinicians navigating unchartered territory within the hospital setting, we have to come to terms with these new challenges, tailoring treatment protocols accordingly with the best clinical practices in mind. Patients with preexisting mental health conditions and who are being treated for COVID-19 are particularly susceptible to clinical deterioration. Recent studies have indicated that psychiatric patients are more prone to feelings of isolation and/or estrangement as well as exacerbation of symptoms such as paranoia.2 Even more concerning is the medication regimen, namely, the novel combination therapies that arise when agents such as hydroxychloroquine are used in tandem with certain antipsychotics or antidepressants.
What’s at stake for COVID-19–positive mental health care patients?
Although the efficacy of hydroxychloroquine is currently being investigated,3 the antimalarial is usually prescribed in tandem with azithromycin for people with COVID-19. The National Institute of Allergy and Infectious Diseases has advised against that particular combination therapy because of ongoing concerns about toxicities.3,4
In another study, azithromycin was effectively substituted with doxycycline to help minimize systemic effects for patients with cardiac and/or pulmonary issues.5 Azithromycin is notorious in the literature for influencing the electrical activity of the heart with the potential for fatal arrhythmia and sudden cardiac death in individuals at risk for cardiovascular disease.5,6,7 It should be noted that both of these commonly prescribed COVID-19 medications (for example, hydroxychloroquine and azithromycin) could lead to QT interval prolongation especially within the context of combination therapy. This is largely concerning for psychiatrists and various other mental health practitioners for the following reasons: (1) higher rates of metabolic syndrome and cardiovascular diseases among psychiatric patients8 and/or (2) effects of certain antipsychotics (for example, IV haloperidol, thioridazine, and ziprasidone) and antidepressants (for example, citalopram and escitalopram) on the QT interval.9
SARS-CoV-2 and clinical judgment: Evaluating patients at higher risk
Although COVID-19 medication guidelines are still being actively developed, hydroxychloroquine appears to be commonly prescribed by physicians. The medication is known myriad untoward effects, including potential behavioral dysfunction (for example, irritability, agitation, suicidal ideation)10 as well as the aforementioned issues concerning arrhythmia (for example, torsades de pointes). Health care professionals might not have much control over the choice of COVID-19 agents because of a lack of available resources or limited options, but they can exercise clinical judgment with respect to selecting the appropriate psychotropic medications.
Treatment recommendations
1. Establish a baseline EKG
A baseline 12-lead EKG is the standard of care for patients currently being screened for COVID-19. It is necessary to rule out the presence of an underlying cardiovascular disease or a rhythm irregularity. A prolonged QTc interval is generally regarded as being around greater than 450-470 msecs with variations attributable to gender;11 numerous studies have affirmed that the risk of acquiring torsades de pointes is substantial when the QTc interval exceeds 500 msecs.12
2. Medical management and risk assessment
Commonly prescribed antipsychotics such as IV haloperidol and ziprasidone are known for exerting a negative effect on the interval and should readily be substituted with other agents in patients who are being treated for COVID-19; the combination of these antipsychotics alongside some COVID-19 medication regimens (for example, hydroxychloroquine/azithromycin) might prove to be fatal. The same logic applies to COVID-19 patients previously on antidepressant therapeutics such as citalopram and escitalopram.
3. Embrace an individually tailored approach to therapeutics
While American Psychiatric Association guidelines historically supported a cessation or reduction in the offending agent under normal circumstances,12 our team is recommending that the psychotropics associated with QTc interval prolongation are discontinued altogether (or substituted with a low-risk agent) in the event that a patient presents with suspected COVID-19. However, after the patients tests negative with COVID-19, they may resume therapy as indicated under the discretion of the mental health practitioner.
References
1. Offard C. “Lancet, NEJM Retract Surgisphere Studies on COVID-19 Patients.” The Scientist Magazine. 2020 Jun 4.
2. Shigemura J et al. Psychiatry Clin Neurosci. 2020 Apr;74(4):281-2.
3. Keshtkar-Jahromi M and Bavari S. Am J Trop Med Hyg. 2020 May;102(5):932-3.
4. Palca J. “NIH panel recommends against drug combination promoted by Trump for COVID-19.” NPR. 2020 Apr 21.
5. Mongelli L. “Long Island doctor tries new twist on hydroxychloroquine for elderly COVID-19 patients.” New York Post. 2020 Apr 4.
6. Hancox JC et al. Ther Adv Infect Dis. 2013 Oct;(5):155-65.
7. Giudicessi JR and Ackerman MJ. Cleve Clin J Med. 2013 Sep;80(9):539-44.
8. Casey DE. Am J Med. 2005 Apr 1;118(Suppl 2):15S-22S.
9. Beach SR et al. Psychosomatics. 2013 Jan 1;54(1):1-3.
10. Bogaczewicz A and Sobów T. Psychiatria i Psychologia Kliniczna. 2017;17(2):111-4.
11. Chohan PS et al. Pak J Med Sci. 2015 Sep-Oct;31(5):1269-71.
12. Lieberman JA et al. APA guidance on the use of antipsychotic drugs and cardiac sudden death. NYS Office of Mental Health. 2012.
Dr. Faisal A. Islam is medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Faisal Islam disclosed no relevant financial relationships.
Dr. Mohammed Islam is affiliated with the department of psychiatry at the Interfaith Medical Center, New York. He disclosed no relevant financial relationships.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He disclosed no relevant financial relationships.
Face mask type matters when sterilizing, study finds
according to researchers. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks.
With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%.
In a research letter published online June 15 in JAMA Network Open, researchers from the University of Oklahoma Health Sciences Center, Oklahoma City, report the results of a study of the two sterilization techniques on the pressure drop and filtration efficiency of N95, KN95, and surgical face masks.
“The H2O2 treatment showed a small effect on the overall filtration efficiency of the tested masks, but the ClO2 treatment showed marked reduction in the overall filtration efficiency of the KN95s and surgical face masks. All pressure drop changes were within the acceptable range,” the researchers write.
The study did not evaluate the effect of repeated sterilizations on face masks.
Five masks of each type were sterilized with either H2O2 or ClO2. Masks were then placed in a test chamber, and a salt aerosol was nebulized to assess both upstream and downstream filtration as well as pressure drop. The researchers used a mobility particle sizer to measure particle number concentration from 16.8 nm to 514 nm. An acceptable pressure drop was defined as a drop of less than 1.38 inches of water (35 mm) for inhalation.
Although pressure drop changes were within the acceptable range for all three mask types following sterilization with either method, H2O2 sterilization yielded the least reduction in filtration efficacy in all cases. After sterilization with H2O2, filtration efficiencies were 96.6%, 97.1%, and 91.6% for the N95s, KN95s, and the surgical face masks, respectively. In contrast, filtration efficiencies after ClO2 sterilization were 95.1%, 76.2%, and 77.9%, respectively.
The researchers note that, although overall filtration efficiency was maintained with ClO2 sterilization, there was a significant drop in efficiency with respect to particles of approximately 300 nm (0.3 microns) in size. For particles of that size, mean filtration efficiency decreased to 86.2% for N95s, 40.8% for KN95s, and 47.1% for surgical face masks.
The testing described in the report is “quite affordable at $350 per mask type, so it is hard to imagine any health care provider cannot set aside a small budget to conduct such an important test,” author Evan Floyd, PhD, told Medscape Medical News.
Given the high demand for effective face masks and the current risk for counterfeit products, Floyd suggested that individual facilities test all masks intended for use by healthcare workers before and after sterilization procedures.
“However, if for some reason testing is not an option, we would recommend sticking to established brands and suppliers, perhaps reach out to your state health department or a local representative of the strategic stockpile of PPE,” he noted.
The authors acknowledge that further studies using a larger sample size and a greater variety of masks, as well as studies to evaluate different sterilization techniques, are required. Further, “measuring the respirator’s filtration efficiency by aerosol size instead of only measuring the overall filtration efficiency” should also be considered. Such an approach would enable researchers to evaluate the degree to which masks protect against specific infectious agents.
This article first appeared on Medscape.com.
according to researchers. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks.
With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%.
In a research letter published online June 15 in JAMA Network Open, researchers from the University of Oklahoma Health Sciences Center, Oklahoma City, report the results of a study of the two sterilization techniques on the pressure drop and filtration efficiency of N95, KN95, and surgical face masks.
“The H2O2 treatment showed a small effect on the overall filtration efficiency of the tested masks, but the ClO2 treatment showed marked reduction in the overall filtration efficiency of the KN95s and surgical face masks. All pressure drop changes were within the acceptable range,” the researchers write.
The study did not evaluate the effect of repeated sterilizations on face masks.
Five masks of each type were sterilized with either H2O2 or ClO2. Masks were then placed in a test chamber, and a salt aerosol was nebulized to assess both upstream and downstream filtration as well as pressure drop. The researchers used a mobility particle sizer to measure particle number concentration from 16.8 nm to 514 nm. An acceptable pressure drop was defined as a drop of less than 1.38 inches of water (35 mm) for inhalation.
Although pressure drop changes were within the acceptable range for all three mask types following sterilization with either method, H2O2 sterilization yielded the least reduction in filtration efficacy in all cases. After sterilization with H2O2, filtration efficiencies were 96.6%, 97.1%, and 91.6% for the N95s, KN95s, and the surgical face masks, respectively. In contrast, filtration efficiencies after ClO2 sterilization were 95.1%, 76.2%, and 77.9%, respectively.
The researchers note that, although overall filtration efficiency was maintained with ClO2 sterilization, there was a significant drop in efficiency with respect to particles of approximately 300 nm (0.3 microns) in size. For particles of that size, mean filtration efficiency decreased to 86.2% for N95s, 40.8% for KN95s, and 47.1% for surgical face masks.
The testing described in the report is “quite affordable at $350 per mask type, so it is hard to imagine any health care provider cannot set aside a small budget to conduct such an important test,” author Evan Floyd, PhD, told Medscape Medical News.
Given the high demand for effective face masks and the current risk for counterfeit products, Floyd suggested that individual facilities test all masks intended for use by healthcare workers before and after sterilization procedures.
“However, if for some reason testing is not an option, we would recommend sticking to established brands and suppliers, perhaps reach out to your state health department or a local representative of the strategic stockpile of PPE,” he noted.
The authors acknowledge that further studies using a larger sample size and a greater variety of masks, as well as studies to evaluate different sterilization techniques, are required. Further, “measuring the respirator’s filtration efficiency by aerosol size instead of only measuring the overall filtration efficiency” should also be considered. Such an approach would enable researchers to evaluate the degree to which masks protect against specific infectious agents.
This article first appeared on Medscape.com.
according to researchers. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks.
With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%.
In a research letter published online June 15 in JAMA Network Open, researchers from the University of Oklahoma Health Sciences Center, Oklahoma City, report the results of a study of the two sterilization techniques on the pressure drop and filtration efficiency of N95, KN95, and surgical face masks.
“The H2O2 treatment showed a small effect on the overall filtration efficiency of the tested masks, but the ClO2 treatment showed marked reduction in the overall filtration efficiency of the KN95s and surgical face masks. All pressure drop changes were within the acceptable range,” the researchers write.
The study did not evaluate the effect of repeated sterilizations on face masks.
Five masks of each type were sterilized with either H2O2 or ClO2. Masks were then placed in a test chamber, and a salt aerosol was nebulized to assess both upstream and downstream filtration as well as pressure drop. The researchers used a mobility particle sizer to measure particle number concentration from 16.8 nm to 514 nm. An acceptable pressure drop was defined as a drop of less than 1.38 inches of water (35 mm) for inhalation.
Although pressure drop changes were within the acceptable range for all three mask types following sterilization with either method, H2O2 sterilization yielded the least reduction in filtration efficacy in all cases. After sterilization with H2O2, filtration efficiencies were 96.6%, 97.1%, and 91.6% for the N95s, KN95s, and the surgical face masks, respectively. In contrast, filtration efficiencies after ClO2 sterilization were 95.1%, 76.2%, and 77.9%, respectively.
The researchers note that, although overall filtration efficiency was maintained with ClO2 sterilization, there was a significant drop in efficiency with respect to particles of approximately 300 nm (0.3 microns) in size. For particles of that size, mean filtration efficiency decreased to 86.2% for N95s, 40.8% for KN95s, and 47.1% for surgical face masks.
The testing described in the report is “quite affordable at $350 per mask type, so it is hard to imagine any health care provider cannot set aside a small budget to conduct such an important test,” author Evan Floyd, PhD, told Medscape Medical News.
Given the high demand for effective face masks and the current risk for counterfeit products, Floyd suggested that individual facilities test all masks intended for use by healthcare workers before and after sterilization procedures.
“However, if for some reason testing is not an option, we would recommend sticking to established brands and suppliers, perhaps reach out to your state health department or a local representative of the strategic stockpile of PPE,” he noted.
The authors acknowledge that further studies using a larger sample size and a greater variety of masks, as well as studies to evaluate different sterilization techniques, are required. Further, “measuring the respirator’s filtration efficiency by aerosol size instead of only measuring the overall filtration efficiency” should also be considered. Such an approach would enable researchers to evaluate the degree to which masks protect against specific infectious agents.
This article first appeared on Medscape.com.
Frequent hypoglycemic episodes raise cardiac event risk
Frequent hypoglycemic episodes were linked to a raised incidence of cardiovascular events in adults with type 2 diabetes in a recent retrospective study, suggesting certain hypoglycemia-associated diabetes drugs should be avoided, an investigator said.
Patients who had more than five hypoglycemic episodes per year had a 61% greater risk of cardiovascular (CV) events, compared with patients with less frequent episodes, according to results of the study.
Although there were fewer strokes among younger patients, the overall increase in cardiovascular event risk held up regardless of age group, according to investigator Aman Rajpal, MD, of Louis Stokes Veterans Affairs Medical Center and Case Western Reserve University, both in Cleveland.
On the basis of these and earlier studies tying hypoglycemia to CV risk, health care providers need to “pay close attention” to low blood sugar and personalize glycemic control targets for each patient based on risk of hypoglycemia, Dr. Rajpal said in a presentation of the study at the virtual annual scientific sessions of the American Diabetes Association.
“Also, this suggests that avoidance of drugs associated with increased risk of hypoglycemia – namely insulin, sulfonylureas, or others – is essential to avoid and minimize the risk of cardiovascular events in this patient population with type 2 diabetes,” said Dr. Rajpal. “Let us remember part of our Hippocratic oath: ‘Above all, do no harm.’ ”
Tailoring treatment to mitigate risk
Mark Schutta, MD, medical director of Penn Rodebaugh Diabetes Center in Philadelphia, said that results of this study suggest a need to carefully select medical therapy for each individual patient with diabetes in order to mitigate CV risk.
“It’s really about tailoring their drugs to their personal situation,” Dr. Schutta said in an interview.
Although newer diabetes drug classes are associated with low to no risk of hypoglycemia, Dr. Schutta said that there is still a place for drugs such as sulfonylureas in certain situations.
Among sulfonylureas, glyburide comes with a much higher incidence of hypoglycemia, compared with glipizide and glimepiride, according to Dr. Schutta. “I think there’s a role for both drugs, but you have to be very careful, and you have to get the data from your patients.”
Hypoglycemia frequency and outcomes
Speculation that hypoglycemia could be linked to adverse CV outcomes was sparked years ago by trials such as ADVANCE. Severe hypoglycemia in that study was associated with a 168% increased risk of death from a CV cause (N Engl J Med. 2010 Oct 7;363:1410-8).
At the time, ADVANCE investigators said they were unable to find evidence that multiple severe hypoglycemia episodes conferred a greater risk of CV events versus a single hypoglycemia episode, though they added that few patients had recurrent events.
“In other words, the association between the number of hypoglycemia events, and adverse CV outcomes is still unclear,” said Dr. Rajpal in his virtual ADA presentation.
Potential elevated risks with more than five episodes
To evaluate the association between frequent hypoglycemic episodes (i.e., more than five per year, compared with one to five episodes) and CV events, Dr. Rajpal and colleagues evaluated outcomes data for 4.9 million adults with type 2 diabetes found in a large commercial database including information on patients in 27 U.S. health care networks.
Database records indicated that about 182,000 patients, or nearly 4%, had episodes of hyperglycemia, which Dr. Rajpal said was presumed to mean a plasma glucose level of less than 70 mg/dL.
Characteristics of the patients with more than five hypoglycemic episodes were similar to those with one to five episodes, although they were more likely to be 65 years or older, and were “slightly more likely” to be on insulin, which could possibly precipitate more hypoglycemic episodes in that group, Dr. Rajpal said.
Key findings
In the main analysis, Dr. Rajpal said, risk of CV events was significantly increased in those with more than five hypoglycemic episodes, compared with those with one to five episodes, with an odds ratio of 1.61 (95% confidence interval, 1.56-1.66). The incidence of cardiovascular events was 33.1% in those with more than five episodes and 23.5% in those with one to five episodes, according to the data presented.
Risks were also significantly increased specifically for cardiac arrhythmias, cerebrovascular accidents, and MI, Dr. Rajpal said, with ORs of 1.65 (95% CI, 1.9-1.71), 1.38 (95% CI, 1.22-1.56), and 1.43 (95% CI, 1.36-1.50), respectively.
Because individuals in the group with more than five hypoglycemic episodes were more likely to be elderly, Dr. Rajpal said that he and coinvestigators decided to perform an age-specific stratified analysis.
Although cerebral vascular incidence was low in younger patients, risk of CV events overall was nevertheless significantly elevated for those aged 65 years or older, 45-64 years, and 18-44 years, with ORs of 1.69 (95% CI, 1.61-1.7), 1.58 (95% CI, 1.48-1.69), and 1.62 (95% CI, 1.33-1.97).
“The results were still valid in stratified analysis based on different age groups,” Dr. Rajpal said.
Dr. Rajpal and coauthors reported that he had no conflicts of interest related to the research.
SOURCE: Rajpal A et al. ADA 2020, Abstract 161-OR.
Frequent hypoglycemic episodes were linked to a raised incidence of cardiovascular events in adults with type 2 diabetes in a recent retrospective study, suggesting certain hypoglycemia-associated diabetes drugs should be avoided, an investigator said.
Patients who had more than five hypoglycemic episodes per year had a 61% greater risk of cardiovascular (CV) events, compared with patients with less frequent episodes, according to results of the study.
Although there were fewer strokes among younger patients, the overall increase in cardiovascular event risk held up regardless of age group, according to investigator Aman Rajpal, MD, of Louis Stokes Veterans Affairs Medical Center and Case Western Reserve University, both in Cleveland.
On the basis of these and earlier studies tying hypoglycemia to CV risk, health care providers need to “pay close attention” to low blood sugar and personalize glycemic control targets for each patient based on risk of hypoglycemia, Dr. Rajpal said in a presentation of the study at the virtual annual scientific sessions of the American Diabetes Association.
“Also, this suggests that avoidance of drugs associated with increased risk of hypoglycemia – namely insulin, sulfonylureas, or others – is essential to avoid and minimize the risk of cardiovascular events in this patient population with type 2 diabetes,” said Dr. Rajpal. “Let us remember part of our Hippocratic oath: ‘Above all, do no harm.’ ”
Tailoring treatment to mitigate risk
Mark Schutta, MD, medical director of Penn Rodebaugh Diabetes Center in Philadelphia, said that results of this study suggest a need to carefully select medical therapy for each individual patient with diabetes in order to mitigate CV risk.
“It’s really about tailoring their drugs to their personal situation,” Dr. Schutta said in an interview.
Although newer diabetes drug classes are associated with low to no risk of hypoglycemia, Dr. Schutta said that there is still a place for drugs such as sulfonylureas in certain situations.
Among sulfonylureas, glyburide comes with a much higher incidence of hypoglycemia, compared with glipizide and glimepiride, according to Dr. Schutta. “I think there’s a role for both drugs, but you have to be very careful, and you have to get the data from your patients.”
Hypoglycemia frequency and outcomes
Speculation that hypoglycemia could be linked to adverse CV outcomes was sparked years ago by trials such as ADVANCE. Severe hypoglycemia in that study was associated with a 168% increased risk of death from a CV cause (N Engl J Med. 2010 Oct 7;363:1410-8).
At the time, ADVANCE investigators said they were unable to find evidence that multiple severe hypoglycemia episodes conferred a greater risk of CV events versus a single hypoglycemia episode, though they added that few patients had recurrent events.
“In other words, the association between the number of hypoglycemia events, and adverse CV outcomes is still unclear,” said Dr. Rajpal in his virtual ADA presentation.
Potential elevated risks with more than five episodes
To evaluate the association between frequent hypoglycemic episodes (i.e., more than five per year, compared with one to five episodes) and CV events, Dr. Rajpal and colleagues evaluated outcomes data for 4.9 million adults with type 2 diabetes found in a large commercial database including information on patients in 27 U.S. health care networks.
Database records indicated that about 182,000 patients, or nearly 4%, had episodes of hyperglycemia, which Dr. Rajpal said was presumed to mean a plasma glucose level of less than 70 mg/dL.
Characteristics of the patients with more than five hypoglycemic episodes were similar to those with one to five episodes, although they were more likely to be 65 years or older, and were “slightly more likely” to be on insulin, which could possibly precipitate more hypoglycemic episodes in that group, Dr. Rajpal said.
Key findings
In the main analysis, Dr. Rajpal said, risk of CV events was significantly increased in those with more than five hypoglycemic episodes, compared with those with one to five episodes, with an odds ratio of 1.61 (95% confidence interval, 1.56-1.66). The incidence of cardiovascular events was 33.1% in those with more than five episodes and 23.5% in those with one to five episodes, according to the data presented.
Risks were also significantly increased specifically for cardiac arrhythmias, cerebrovascular accidents, and MI, Dr. Rajpal said, with ORs of 1.65 (95% CI, 1.9-1.71), 1.38 (95% CI, 1.22-1.56), and 1.43 (95% CI, 1.36-1.50), respectively.
Because individuals in the group with more than five hypoglycemic episodes were more likely to be elderly, Dr. Rajpal said that he and coinvestigators decided to perform an age-specific stratified analysis.
Although cerebral vascular incidence was low in younger patients, risk of CV events overall was nevertheless significantly elevated for those aged 65 years or older, 45-64 years, and 18-44 years, with ORs of 1.69 (95% CI, 1.61-1.7), 1.58 (95% CI, 1.48-1.69), and 1.62 (95% CI, 1.33-1.97).
“The results were still valid in stratified analysis based on different age groups,” Dr. Rajpal said.
Dr. Rajpal and coauthors reported that he had no conflicts of interest related to the research.
SOURCE: Rajpal A et al. ADA 2020, Abstract 161-OR.
Frequent hypoglycemic episodes were linked to a raised incidence of cardiovascular events in adults with type 2 diabetes in a recent retrospective study, suggesting certain hypoglycemia-associated diabetes drugs should be avoided, an investigator said.
Patients who had more than five hypoglycemic episodes per year had a 61% greater risk of cardiovascular (CV) events, compared with patients with less frequent episodes, according to results of the study.
Although there were fewer strokes among younger patients, the overall increase in cardiovascular event risk held up regardless of age group, according to investigator Aman Rajpal, MD, of Louis Stokes Veterans Affairs Medical Center and Case Western Reserve University, both in Cleveland.
On the basis of these and earlier studies tying hypoglycemia to CV risk, health care providers need to “pay close attention” to low blood sugar and personalize glycemic control targets for each patient based on risk of hypoglycemia, Dr. Rajpal said in a presentation of the study at the virtual annual scientific sessions of the American Diabetes Association.
“Also, this suggests that avoidance of drugs associated with increased risk of hypoglycemia – namely insulin, sulfonylureas, or others – is essential to avoid and minimize the risk of cardiovascular events in this patient population with type 2 diabetes,” said Dr. Rajpal. “Let us remember part of our Hippocratic oath: ‘Above all, do no harm.’ ”
Tailoring treatment to mitigate risk
Mark Schutta, MD, medical director of Penn Rodebaugh Diabetes Center in Philadelphia, said that results of this study suggest a need to carefully select medical therapy for each individual patient with diabetes in order to mitigate CV risk.
“It’s really about tailoring their drugs to their personal situation,” Dr. Schutta said in an interview.
Although newer diabetes drug classes are associated with low to no risk of hypoglycemia, Dr. Schutta said that there is still a place for drugs such as sulfonylureas in certain situations.
Among sulfonylureas, glyburide comes with a much higher incidence of hypoglycemia, compared with glipizide and glimepiride, according to Dr. Schutta. “I think there’s a role for both drugs, but you have to be very careful, and you have to get the data from your patients.”
Hypoglycemia frequency and outcomes
Speculation that hypoglycemia could be linked to adverse CV outcomes was sparked years ago by trials such as ADVANCE. Severe hypoglycemia in that study was associated with a 168% increased risk of death from a CV cause (N Engl J Med. 2010 Oct 7;363:1410-8).
At the time, ADVANCE investigators said they were unable to find evidence that multiple severe hypoglycemia episodes conferred a greater risk of CV events versus a single hypoglycemia episode, though they added that few patients had recurrent events.
“In other words, the association between the number of hypoglycemia events, and adverse CV outcomes is still unclear,” said Dr. Rajpal in his virtual ADA presentation.
Potential elevated risks with more than five episodes
To evaluate the association between frequent hypoglycemic episodes (i.e., more than five per year, compared with one to five episodes) and CV events, Dr. Rajpal and colleagues evaluated outcomes data for 4.9 million adults with type 2 diabetes found in a large commercial database including information on patients in 27 U.S. health care networks.
Database records indicated that about 182,000 patients, or nearly 4%, had episodes of hyperglycemia, which Dr. Rajpal said was presumed to mean a plasma glucose level of less than 70 mg/dL.
Characteristics of the patients with more than five hypoglycemic episodes were similar to those with one to five episodes, although they were more likely to be 65 years or older, and were “slightly more likely” to be on insulin, which could possibly precipitate more hypoglycemic episodes in that group, Dr. Rajpal said.
Key findings
In the main analysis, Dr. Rajpal said, risk of CV events was significantly increased in those with more than five hypoglycemic episodes, compared with those with one to five episodes, with an odds ratio of 1.61 (95% confidence interval, 1.56-1.66). The incidence of cardiovascular events was 33.1% in those with more than five episodes and 23.5% in those with one to five episodes, according to the data presented.
Risks were also significantly increased specifically for cardiac arrhythmias, cerebrovascular accidents, and MI, Dr. Rajpal said, with ORs of 1.65 (95% CI, 1.9-1.71), 1.38 (95% CI, 1.22-1.56), and 1.43 (95% CI, 1.36-1.50), respectively.
Because individuals in the group with more than five hypoglycemic episodes were more likely to be elderly, Dr. Rajpal said that he and coinvestigators decided to perform an age-specific stratified analysis.
Although cerebral vascular incidence was low in younger patients, risk of CV events overall was nevertheless significantly elevated for those aged 65 years or older, 45-64 years, and 18-44 years, with ORs of 1.69 (95% CI, 1.61-1.7), 1.58 (95% CI, 1.48-1.69), and 1.62 (95% CI, 1.33-1.97).
“The results were still valid in stratified analysis based on different age groups,” Dr. Rajpal said.
Dr. Rajpal and coauthors reported that he had no conflicts of interest related to the research.
SOURCE: Rajpal A et al. ADA 2020, Abstract 161-OR.
FROM ADA 2020
Hospitalist well-being during the pandemic
Navigating COVID-19 requires self-care
The global COVID-19 pandemic has escalated everyone’s stress levels, especially clinicians caring for hospitalized patients. New pressures have added to everyday stress, new studies have revised prior patient care recommendations, and the world generally seems upside down. What can a busy hospitalist do to maintain a modicum of sanity in all the craziness?
The stressors facing hospitalists
Uncertainty
Of all the burdens COVID-19 has unleashed, the biggest may be uncertainty. Not only is there unease about the virus itself, there also is legitimate concern about the future of medicine, said Elizabeth Harry, MD, SFHM, a hospitalist and senior director of clinical affairs at the University of Colorado Hospital in Aurora.
“What does it look like after an event like this, particularly in areas like academic medicine and teaching our next generation and getting funding for research? And how do we continue to produce physicians that can provide excellent care?” she asked.
There is also uncertainty in the best way to care for patients, said Eileen Barrett, MD, MPH, SFHM, a hospitalist at the University of New Mexico, Albuquerque.
“There are some models that are emerging to predict who will have a worse outcome, but they’re still not great models, so we have uncertainty for a given patient.” And, she noted, as the science continues to evolve, there exists a constant worry that “you might have inadvertently caused someone harm.”
The financial implications of the pandemic are creating uncertainty too. “When you fund a health care system with elective procedures and you can’t do those, and instead have to shift to the most essential services, a lot of places are seeing a massive deficit, which is going to affect staff morale and some physician offices are going to close,” said Elisabeth Poorman, MD, MPH, a primary care and internal medicine physician and chair of the King County Medical Society Physician Wellness Committee in Seattle.
Fear
When the pandemic began in the United States, “fear of the unknown was perhaps the scariest part, particularly as it pertained to personal protective equipment,” said Mark Rudolph, MD, SFHM, chief experience officer and vice president of patient experience and physician development at Sound Physicians in Tacoma, Wash. “For most clinicians, this is the first time that they are themselves in harm’s way while they do their jobs. And worse, they risk bringing the virus home to their families. That is the concern I hear most.”
Anxiety
Worrying about being able to provide excellent patient care is a big stressor, especially since this is the heart and soul of why most hospitalists have gone into their line of work.
“Part of providing excellent care to your patients is providing excellent supportive care to their families,” Dr. Harry said. “There’s some dissonance there in not being able to allow the family to come visit, but wanting to keep them safe, and it feels really hard to support your patients and support their families in the best way. It can feel like you’re just watching and waiting to see what will happen, and that we don’t have a lot of agency over which direction things take.”
There is concern for health care team members as well, Dr. Harry added. “Physicians care a lot about their teams and how they’re doing. I think there’s a sense of esprit de corps among folks and worry for each other there.”
Guilt
Although you may be at the hospital all day, you may feel guilty when you are not providing direct patient care. Or maybe you or someone on your team has an immunodeficiency and can’t be on the front line. Perhaps one of your team members contracted COVID-19 and you did not. Whatever the case, guilt is another emotion that is rampant among hospitalists right now, Dr. Barrett said.
Burnout
Unfortunately, burnout is a potential reality in times of high stress. “Burnout is dynamic,” said Dr. Poorman. “It’s a process by which your emotional and cognitive reserves are exhausted. The people with the highest burnout are the ones who are still trying to provide the standard of care, or above the standard of care in dysfunctional systems.”
Dr. Harry noted that burnout presents in different ways for different people, but Dr. Rudolph added that it’s crucial for hospitalist team members to watch for signs of burnout so they can intervene and/or get help for their colleagues.
Warning signs in yourself or others that burnout could be on the horizon include:
- Fatigue/exhaustion – Whether emotional or physical (or both), this can become a problem if it “just doesn’t seem to go away despite rest and time away from work,” said Dr. Rudolph.
- Behavioral changes – Any behavior that’s out of the ordinary may be a red flag, like lashing out at someone at work.
- Overwork – Working too much can be caused by an inability to let go of patient care, Dr. Barrett said.
- Not working enough – This may include avoiding tasks and having difficulty meeting deadlines.
- Maladaptive coping behaviors – Excessive consumption of alcohol or drugs is a common coping mechanism. “Even excessive consumption of news is something that people are using to numb out a little bit,” said Dr. Harry.
- Depersonalization – “This is where you start to look at patients, colleagues, or administrators as ‘them’ and you can’t connect as deeply,” Dr. Harry said. “Part of that’s protective and a normal thing to do during a big trauma like this, but it’s also incredibly distancing. Any language that people start using that feels like ‘us’ or ‘them’ is a warning sign.”
- Disengagement – Many people disengage from their work, but Dr. Poorman said physicians tend to disengage from other parts of their lives, such as exercise and family interaction.
Protecting yourself while supporting others
Like the illustration of putting the oxygen mask on yourself first so you can help others, it’s important to protect your own mental and physical health as you support your fellow physicians. Here’s what the experts suggest.
Focus on basic needs
“When you’re in the midst of a trauma, which we are, you don’t want to open all of that up and go to the depths of your thoughts about the grief of all of it because it can actually make the trauma worse,” said Dr. Harry. “There’s a lot of literature that debriefing is really helpful after the event, but if you do it during the event, it can be really dangerous.”
Instead, she said, the goal should be focusing on your basic needs and what you need to do to get through each day, like keeping you and your family in good health. “What is your purpose? Staying connected to why you do this and staying focused on the present is really important,” Dr. Harry noted.
Do your best to get a good night’s sleep, exercise as much as you can, talk to others, and see a mental health provider if your anxiety is too high, advises Dr. Barrett. “Even avoiding blue light from phones and screens within 2 hours of bedtime, parking further away from the hospital and walking, and taking the stairs are things that add up in a big way.”
Keep up your normal routine
“Right now, it’s really critical for clinicians to keep up components of their routine that feel ‘normal,’ ” Dr. Rudolph said. “Whether it’s exercise, playing board games with their kids, or spending time on a hobby, it’s critical to allow yourself these comfortable, predictable, and rewarding detours.”
Set limits
People under stress tend to find unhealthy ways to cope. Instead, try being intentional about what you are consuming by putting limits on things like your news, alcohol consumption, and the number of hours you work, said Dr. Harry.
Implement a culture of wellness
Dr. Barrett believes in creating the work culture we want to be in, one that ensures people have psychological safety, allows them to ask for help, encourages them to disconnect completely from work, and makes them feel valued and listened to. She likes the example of “the pause,” which is called by a team member right after a patient expires.
“It’s a 30-second moment of silence where we reflect on the patient, their loved ones, and every member of the health care team who helped support and treat them,” said Dr. Barrett. “At the conclusion, you say: ‘Thank you. Is there anything you need to be able to go back to the care of other patients?’ Because it’s unnatural to have this terrible thing that happened and then just act like nothing happened.”
Target resources
Be proactive and know where to find resources before you need them, advised Dr. Harry. “Most institutions have free mental health resources, either through their employee assistance programs or HR, plus there’s lots of national organizations that are offering free resources to health care providers.”
Focus on what you can control
Separating what is under your control from what is not is a struggle for everyone, Dr. Poorman said, but it’s helpful to think about the ways you can have an impact and what you’re able to control.
“There was a woman who was diagnosed with early-onset Parkinson’s that I heard giving an interview at the beginning of this pandemic,” she said. “It was the most helpful advice I got, which was: ‘Think of the next good thing you can do.’ You can’t fix everything, so what’s the next good thing you can do?”
Maintain connectivity
Make sure you are utilizing your support circle and staying connected. “That sense of connection is incredibly protective on multiple fronts for depression, for burnout, for suicide ideation, etc.,” Dr. Harry said.
“It doesn’t matter if it’s your teammates at work, your family at home, your best friend from medical school – whomever you can debrief with, vent with, and just share your thoughts and feelings with, these outlets are critical for all of us to process our emotions and diffuse stress and anxiety,” said Dr. Rudolph.
Dr. Poorman is concerned that there could be a spike in physician suicides caused by increased stress, so she also encourages talking openly about what is going on and about getting help when it’s necessary. “Many of us are afraid to seek care because we can actually have our ability to practice medicine questioned, but now is not the time for heroes. Now is the time for people who are willing to recognize their own strengths and limitations to take care of one another.”
Be compassionate toward others
Keep in mind that everyone is stressed out and offer empathy and compassion. “I think everybody’s struggling to try to figure this out and the more that we can give each other the benefit of the doubt and a little grace, the more protective that is,” said Dr. Harry.
Listening is meaningful too. “Recognizing opportunities to validate and acknowledge the feelings that are being shared with you by your colleagues is critical,” Dr. Rudolph said. “We all need to know that we’re not alone, that our thoughts and feelings are okay, and when we share a difficult story, the value of someone saying something as simple as, ‘wow, that sounds like it was really hard,’ is immense.”
Be compassionate toward yourself
Try to give yourself a break and be as compassionate with yourself as you would with others. It’s okay that you’re not getting in shape, publishing prolifically, or redesigning your house right now.
“There’s a lot of data linking lack of self-compassion to burnout,” said Dr. Harry. She says there are courses on self-compassion available that help you work on being kinder to yourself.
Get a “battle buddy”
The American Medical Association has a free “buddy system” program called PeerRx to help physicians cope during the pandemic. Dr. Rudolph said that now is a great time to use this military-developed intervention in which each team member checks in with a chosen partner at agreed-upon intervals.
For example, “You can tell that person: ‘If I don’t call my family for a week that’s a red flag for me.’ And then you hold each other accountable to those things,” Dr. Harry said.
The buddy system is another way to harness that sense of connection that is so vital to our health and well-being.
“The simple act of showing that you care … can make all the difference when you’re doing this kind of work that is both challenging and dangerous,” said Dr. Rudolph.
Navigating COVID-19 requires self-care
Navigating COVID-19 requires self-care
The global COVID-19 pandemic has escalated everyone’s stress levels, especially clinicians caring for hospitalized patients. New pressures have added to everyday stress, new studies have revised prior patient care recommendations, and the world generally seems upside down. What can a busy hospitalist do to maintain a modicum of sanity in all the craziness?
The stressors facing hospitalists
Uncertainty
Of all the burdens COVID-19 has unleashed, the biggest may be uncertainty. Not only is there unease about the virus itself, there also is legitimate concern about the future of medicine, said Elizabeth Harry, MD, SFHM, a hospitalist and senior director of clinical affairs at the University of Colorado Hospital in Aurora.
“What does it look like after an event like this, particularly in areas like academic medicine and teaching our next generation and getting funding for research? And how do we continue to produce physicians that can provide excellent care?” she asked.
There is also uncertainty in the best way to care for patients, said Eileen Barrett, MD, MPH, SFHM, a hospitalist at the University of New Mexico, Albuquerque.
“There are some models that are emerging to predict who will have a worse outcome, but they’re still not great models, so we have uncertainty for a given patient.” And, she noted, as the science continues to evolve, there exists a constant worry that “you might have inadvertently caused someone harm.”
The financial implications of the pandemic are creating uncertainty too. “When you fund a health care system with elective procedures and you can’t do those, and instead have to shift to the most essential services, a lot of places are seeing a massive deficit, which is going to affect staff morale and some physician offices are going to close,” said Elisabeth Poorman, MD, MPH, a primary care and internal medicine physician and chair of the King County Medical Society Physician Wellness Committee in Seattle.
Fear
When the pandemic began in the United States, “fear of the unknown was perhaps the scariest part, particularly as it pertained to personal protective equipment,” said Mark Rudolph, MD, SFHM, chief experience officer and vice president of patient experience and physician development at Sound Physicians in Tacoma, Wash. “For most clinicians, this is the first time that they are themselves in harm’s way while they do their jobs. And worse, they risk bringing the virus home to their families. That is the concern I hear most.”
Anxiety
Worrying about being able to provide excellent patient care is a big stressor, especially since this is the heart and soul of why most hospitalists have gone into their line of work.
“Part of providing excellent care to your patients is providing excellent supportive care to their families,” Dr. Harry said. “There’s some dissonance there in not being able to allow the family to come visit, but wanting to keep them safe, and it feels really hard to support your patients and support their families in the best way. It can feel like you’re just watching and waiting to see what will happen, and that we don’t have a lot of agency over which direction things take.”
There is concern for health care team members as well, Dr. Harry added. “Physicians care a lot about their teams and how they’re doing. I think there’s a sense of esprit de corps among folks and worry for each other there.”
Guilt
Although you may be at the hospital all day, you may feel guilty when you are not providing direct patient care. Or maybe you or someone on your team has an immunodeficiency and can’t be on the front line. Perhaps one of your team members contracted COVID-19 and you did not. Whatever the case, guilt is another emotion that is rampant among hospitalists right now, Dr. Barrett said.
Burnout
Unfortunately, burnout is a potential reality in times of high stress. “Burnout is dynamic,” said Dr. Poorman. “It’s a process by which your emotional and cognitive reserves are exhausted. The people with the highest burnout are the ones who are still trying to provide the standard of care, or above the standard of care in dysfunctional systems.”
Dr. Harry noted that burnout presents in different ways for different people, but Dr. Rudolph added that it’s crucial for hospitalist team members to watch for signs of burnout so they can intervene and/or get help for their colleagues.
Warning signs in yourself or others that burnout could be on the horizon include:
- Fatigue/exhaustion – Whether emotional or physical (or both), this can become a problem if it “just doesn’t seem to go away despite rest and time away from work,” said Dr. Rudolph.
- Behavioral changes – Any behavior that’s out of the ordinary may be a red flag, like lashing out at someone at work.
- Overwork – Working too much can be caused by an inability to let go of patient care, Dr. Barrett said.
- Not working enough – This may include avoiding tasks and having difficulty meeting deadlines.
- Maladaptive coping behaviors – Excessive consumption of alcohol or drugs is a common coping mechanism. “Even excessive consumption of news is something that people are using to numb out a little bit,” said Dr. Harry.
- Depersonalization – “This is where you start to look at patients, colleagues, or administrators as ‘them’ and you can’t connect as deeply,” Dr. Harry said. “Part of that’s protective and a normal thing to do during a big trauma like this, but it’s also incredibly distancing. Any language that people start using that feels like ‘us’ or ‘them’ is a warning sign.”
- Disengagement – Many people disengage from their work, but Dr. Poorman said physicians tend to disengage from other parts of their lives, such as exercise and family interaction.
Protecting yourself while supporting others
Like the illustration of putting the oxygen mask on yourself first so you can help others, it’s important to protect your own mental and physical health as you support your fellow physicians. Here’s what the experts suggest.
Focus on basic needs
“When you’re in the midst of a trauma, which we are, you don’t want to open all of that up and go to the depths of your thoughts about the grief of all of it because it can actually make the trauma worse,” said Dr. Harry. “There’s a lot of literature that debriefing is really helpful after the event, but if you do it during the event, it can be really dangerous.”
Instead, she said, the goal should be focusing on your basic needs and what you need to do to get through each day, like keeping you and your family in good health. “What is your purpose? Staying connected to why you do this and staying focused on the present is really important,” Dr. Harry noted.
Do your best to get a good night’s sleep, exercise as much as you can, talk to others, and see a mental health provider if your anxiety is too high, advises Dr. Barrett. “Even avoiding blue light from phones and screens within 2 hours of bedtime, parking further away from the hospital and walking, and taking the stairs are things that add up in a big way.”
Keep up your normal routine
“Right now, it’s really critical for clinicians to keep up components of their routine that feel ‘normal,’ ” Dr. Rudolph said. “Whether it’s exercise, playing board games with their kids, or spending time on a hobby, it’s critical to allow yourself these comfortable, predictable, and rewarding detours.”
Set limits
People under stress tend to find unhealthy ways to cope. Instead, try being intentional about what you are consuming by putting limits on things like your news, alcohol consumption, and the number of hours you work, said Dr. Harry.
Implement a culture of wellness
Dr. Barrett believes in creating the work culture we want to be in, one that ensures people have psychological safety, allows them to ask for help, encourages them to disconnect completely from work, and makes them feel valued and listened to. She likes the example of “the pause,” which is called by a team member right after a patient expires.
“It’s a 30-second moment of silence where we reflect on the patient, their loved ones, and every member of the health care team who helped support and treat them,” said Dr. Barrett. “At the conclusion, you say: ‘Thank you. Is there anything you need to be able to go back to the care of other patients?’ Because it’s unnatural to have this terrible thing that happened and then just act like nothing happened.”
Target resources
Be proactive and know where to find resources before you need them, advised Dr. Harry. “Most institutions have free mental health resources, either through their employee assistance programs or HR, plus there’s lots of national organizations that are offering free resources to health care providers.”
Focus on what you can control
Separating what is under your control from what is not is a struggle for everyone, Dr. Poorman said, but it’s helpful to think about the ways you can have an impact and what you’re able to control.
“There was a woman who was diagnosed with early-onset Parkinson’s that I heard giving an interview at the beginning of this pandemic,” she said. “It was the most helpful advice I got, which was: ‘Think of the next good thing you can do.’ You can’t fix everything, so what’s the next good thing you can do?”
Maintain connectivity
Make sure you are utilizing your support circle and staying connected. “That sense of connection is incredibly protective on multiple fronts for depression, for burnout, for suicide ideation, etc.,” Dr. Harry said.
“It doesn’t matter if it’s your teammates at work, your family at home, your best friend from medical school – whomever you can debrief with, vent with, and just share your thoughts and feelings with, these outlets are critical for all of us to process our emotions and diffuse stress and anxiety,” said Dr. Rudolph.
Dr. Poorman is concerned that there could be a spike in physician suicides caused by increased stress, so she also encourages talking openly about what is going on and about getting help when it’s necessary. “Many of us are afraid to seek care because we can actually have our ability to practice medicine questioned, but now is not the time for heroes. Now is the time for people who are willing to recognize their own strengths and limitations to take care of one another.”
Be compassionate toward others
Keep in mind that everyone is stressed out and offer empathy and compassion. “I think everybody’s struggling to try to figure this out and the more that we can give each other the benefit of the doubt and a little grace, the more protective that is,” said Dr. Harry.
Listening is meaningful too. “Recognizing opportunities to validate and acknowledge the feelings that are being shared with you by your colleagues is critical,” Dr. Rudolph said. “We all need to know that we’re not alone, that our thoughts and feelings are okay, and when we share a difficult story, the value of someone saying something as simple as, ‘wow, that sounds like it was really hard,’ is immense.”
Be compassionate toward yourself
Try to give yourself a break and be as compassionate with yourself as you would with others. It’s okay that you’re not getting in shape, publishing prolifically, or redesigning your house right now.
“There’s a lot of data linking lack of self-compassion to burnout,” said Dr. Harry. She says there are courses on self-compassion available that help you work on being kinder to yourself.
Get a “battle buddy”
The American Medical Association has a free “buddy system” program called PeerRx to help physicians cope during the pandemic. Dr. Rudolph said that now is a great time to use this military-developed intervention in which each team member checks in with a chosen partner at agreed-upon intervals.
For example, “You can tell that person: ‘If I don’t call my family for a week that’s a red flag for me.’ And then you hold each other accountable to those things,” Dr. Harry said.
The buddy system is another way to harness that sense of connection that is so vital to our health and well-being.
“The simple act of showing that you care … can make all the difference when you’re doing this kind of work that is both challenging and dangerous,” said Dr. Rudolph.
The global COVID-19 pandemic has escalated everyone’s stress levels, especially clinicians caring for hospitalized patients. New pressures have added to everyday stress, new studies have revised prior patient care recommendations, and the world generally seems upside down. What can a busy hospitalist do to maintain a modicum of sanity in all the craziness?
The stressors facing hospitalists
Uncertainty
Of all the burdens COVID-19 has unleashed, the biggest may be uncertainty. Not only is there unease about the virus itself, there also is legitimate concern about the future of medicine, said Elizabeth Harry, MD, SFHM, a hospitalist and senior director of clinical affairs at the University of Colorado Hospital in Aurora.
“What does it look like after an event like this, particularly in areas like academic medicine and teaching our next generation and getting funding for research? And how do we continue to produce physicians that can provide excellent care?” she asked.
There is also uncertainty in the best way to care for patients, said Eileen Barrett, MD, MPH, SFHM, a hospitalist at the University of New Mexico, Albuquerque.
“There are some models that are emerging to predict who will have a worse outcome, but they’re still not great models, so we have uncertainty for a given patient.” And, she noted, as the science continues to evolve, there exists a constant worry that “you might have inadvertently caused someone harm.”
The financial implications of the pandemic are creating uncertainty too. “When you fund a health care system with elective procedures and you can’t do those, and instead have to shift to the most essential services, a lot of places are seeing a massive deficit, which is going to affect staff morale and some physician offices are going to close,” said Elisabeth Poorman, MD, MPH, a primary care and internal medicine physician and chair of the King County Medical Society Physician Wellness Committee in Seattle.
Fear
When the pandemic began in the United States, “fear of the unknown was perhaps the scariest part, particularly as it pertained to personal protective equipment,” said Mark Rudolph, MD, SFHM, chief experience officer and vice president of patient experience and physician development at Sound Physicians in Tacoma, Wash. “For most clinicians, this is the first time that they are themselves in harm’s way while they do their jobs. And worse, they risk bringing the virus home to their families. That is the concern I hear most.”
Anxiety
Worrying about being able to provide excellent patient care is a big stressor, especially since this is the heart and soul of why most hospitalists have gone into their line of work.
“Part of providing excellent care to your patients is providing excellent supportive care to their families,” Dr. Harry said. “There’s some dissonance there in not being able to allow the family to come visit, but wanting to keep them safe, and it feels really hard to support your patients and support their families in the best way. It can feel like you’re just watching and waiting to see what will happen, and that we don’t have a lot of agency over which direction things take.”
There is concern for health care team members as well, Dr. Harry added. “Physicians care a lot about their teams and how they’re doing. I think there’s a sense of esprit de corps among folks and worry for each other there.”
Guilt
Although you may be at the hospital all day, you may feel guilty when you are not providing direct patient care. Or maybe you or someone on your team has an immunodeficiency and can’t be on the front line. Perhaps one of your team members contracted COVID-19 and you did not. Whatever the case, guilt is another emotion that is rampant among hospitalists right now, Dr. Barrett said.
Burnout
Unfortunately, burnout is a potential reality in times of high stress. “Burnout is dynamic,” said Dr. Poorman. “It’s a process by which your emotional and cognitive reserves are exhausted. The people with the highest burnout are the ones who are still trying to provide the standard of care, or above the standard of care in dysfunctional systems.”
Dr. Harry noted that burnout presents in different ways for different people, but Dr. Rudolph added that it’s crucial for hospitalist team members to watch for signs of burnout so they can intervene and/or get help for their colleagues.
Warning signs in yourself or others that burnout could be on the horizon include:
- Fatigue/exhaustion – Whether emotional or physical (or both), this can become a problem if it “just doesn’t seem to go away despite rest and time away from work,” said Dr. Rudolph.
- Behavioral changes – Any behavior that’s out of the ordinary may be a red flag, like lashing out at someone at work.
- Overwork – Working too much can be caused by an inability to let go of patient care, Dr. Barrett said.
- Not working enough – This may include avoiding tasks and having difficulty meeting deadlines.
- Maladaptive coping behaviors – Excessive consumption of alcohol or drugs is a common coping mechanism. “Even excessive consumption of news is something that people are using to numb out a little bit,” said Dr. Harry.
- Depersonalization – “This is where you start to look at patients, colleagues, or administrators as ‘them’ and you can’t connect as deeply,” Dr. Harry said. “Part of that’s protective and a normal thing to do during a big trauma like this, but it’s also incredibly distancing. Any language that people start using that feels like ‘us’ or ‘them’ is a warning sign.”
- Disengagement – Many people disengage from their work, but Dr. Poorman said physicians tend to disengage from other parts of their lives, such as exercise and family interaction.
Protecting yourself while supporting others
Like the illustration of putting the oxygen mask on yourself first so you can help others, it’s important to protect your own mental and physical health as you support your fellow physicians. Here’s what the experts suggest.
Focus on basic needs
“When you’re in the midst of a trauma, which we are, you don’t want to open all of that up and go to the depths of your thoughts about the grief of all of it because it can actually make the trauma worse,” said Dr. Harry. “There’s a lot of literature that debriefing is really helpful after the event, but if you do it during the event, it can be really dangerous.”
Instead, she said, the goal should be focusing on your basic needs and what you need to do to get through each day, like keeping you and your family in good health. “What is your purpose? Staying connected to why you do this and staying focused on the present is really important,” Dr. Harry noted.
Do your best to get a good night’s sleep, exercise as much as you can, talk to others, and see a mental health provider if your anxiety is too high, advises Dr. Barrett. “Even avoiding blue light from phones and screens within 2 hours of bedtime, parking further away from the hospital and walking, and taking the stairs are things that add up in a big way.”
Keep up your normal routine
“Right now, it’s really critical for clinicians to keep up components of their routine that feel ‘normal,’ ” Dr. Rudolph said. “Whether it’s exercise, playing board games with their kids, or spending time on a hobby, it’s critical to allow yourself these comfortable, predictable, and rewarding detours.”
Set limits
People under stress tend to find unhealthy ways to cope. Instead, try being intentional about what you are consuming by putting limits on things like your news, alcohol consumption, and the number of hours you work, said Dr. Harry.
Implement a culture of wellness
Dr. Barrett believes in creating the work culture we want to be in, one that ensures people have psychological safety, allows them to ask for help, encourages them to disconnect completely from work, and makes them feel valued and listened to. She likes the example of “the pause,” which is called by a team member right after a patient expires.
“It’s a 30-second moment of silence where we reflect on the patient, their loved ones, and every member of the health care team who helped support and treat them,” said Dr. Barrett. “At the conclusion, you say: ‘Thank you. Is there anything you need to be able to go back to the care of other patients?’ Because it’s unnatural to have this terrible thing that happened and then just act like nothing happened.”
Target resources
Be proactive and know where to find resources before you need them, advised Dr. Harry. “Most institutions have free mental health resources, either through their employee assistance programs or HR, plus there’s lots of national organizations that are offering free resources to health care providers.”
Focus on what you can control
Separating what is under your control from what is not is a struggle for everyone, Dr. Poorman said, but it’s helpful to think about the ways you can have an impact and what you’re able to control.
“There was a woman who was diagnosed with early-onset Parkinson’s that I heard giving an interview at the beginning of this pandemic,” she said. “It was the most helpful advice I got, which was: ‘Think of the next good thing you can do.’ You can’t fix everything, so what’s the next good thing you can do?”
Maintain connectivity
Make sure you are utilizing your support circle and staying connected. “That sense of connection is incredibly protective on multiple fronts for depression, for burnout, for suicide ideation, etc.,” Dr. Harry said.
“It doesn’t matter if it’s your teammates at work, your family at home, your best friend from medical school – whomever you can debrief with, vent with, and just share your thoughts and feelings with, these outlets are critical for all of us to process our emotions and diffuse stress and anxiety,” said Dr. Rudolph.
Dr. Poorman is concerned that there could be a spike in physician suicides caused by increased stress, so she also encourages talking openly about what is going on and about getting help when it’s necessary. “Many of us are afraid to seek care because we can actually have our ability to practice medicine questioned, but now is not the time for heroes. Now is the time for people who are willing to recognize their own strengths and limitations to take care of one another.”
Be compassionate toward others
Keep in mind that everyone is stressed out and offer empathy and compassion. “I think everybody’s struggling to try to figure this out and the more that we can give each other the benefit of the doubt and a little grace, the more protective that is,” said Dr. Harry.
Listening is meaningful too. “Recognizing opportunities to validate and acknowledge the feelings that are being shared with you by your colleagues is critical,” Dr. Rudolph said. “We all need to know that we’re not alone, that our thoughts and feelings are okay, and when we share a difficult story, the value of someone saying something as simple as, ‘wow, that sounds like it was really hard,’ is immense.”
Be compassionate toward yourself
Try to give yourself a break and be as compassionate with yourself as you would with others. It’s okay that you’re not getting in shape, publishing prolifically, or redesigning your house right now.
“There’s a lot of data linking lack of self-compassion to burnout,” said Dr. Harry. She says there are courses on self-compassion available that help you work on being kinder to yourself.
Get a “battle buddy”
The American Medical Association has a free “buddy system” program called PeerRx to help physicians cope during the pandemic. Dr. Rudolph said that now is a great time to use this military-developed intervention in which each team member checks in with a chosen partner at agreed-upon intervals.
For example, “You can tell that person: ‘If I don’t call my family for a week that’s a red flag for me.’ And then you hold each other accountable to those things,” Dr. Harry said.
The buddy system is another way to harness that sense of connection that is so vital to our health and well-being.
“The simple act of showing that you care … can make all the difference when you’re doing this kind of work that is both challenging and dangerous,” said Dr. Rudolph.
Comorbidities increase COVID-19 deaths by factor of 12
, compared with those who have no such condition, according to the Centers for Disease Control and Prevention.
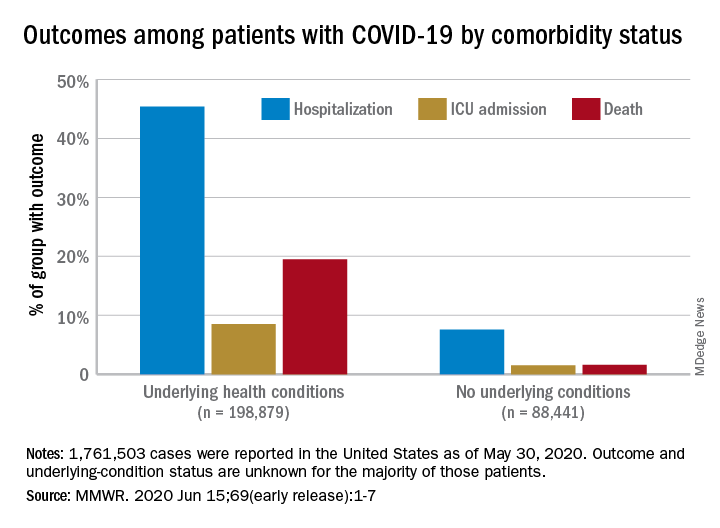
Among those with underlying conditions such as cardiovascular disease or diabetes, 45.4% of patients with COVID-19 were hospitalized, versus 7.6% of patients without an underlying condition, said Erin K. Stokes, MPH, and associates of the CDC COVID-19 Emergency Response team.
The difference in deaths was even greater over the study period of Jan. 22–May 30, 2020: 19.5% of COVID-19 patients with underlying conditions died, compared with 1.6% of those with no underlying condition. The gap narrowed, however, for ICU admissions, with corresponding rates of 8.5% and 1.5%, the investigators reported June 15 in the Morbidity and Mortality Weekly Report.
“The COVID-19 pandemic continues to be severe, particularly in certain population groups,” they said.
The cumulative incidence of laboratory-confirmed cases up to May 30, for instance, was nearly twice as high for those aged 80 years and over (902 per 100,000 population) than for those aged 70-79 years (464.2 per 100,000). Those aged 50-59 years had the second-highest incidence, 550.5 per 100,000, Ms. Stokes and associates said.
“Among cases with known race and ethnicity, 33% of persons were Hispanic, 22% were black, and 1.3% were [American Indian/Alaska Native]. These findings suggest that persons in these groups, who account for 18%, 13%, and 0.7% of the U.S. population, respectively, are disproportionately affected by the COVID-19 pandemic,” they wrote.
Another source of disparity: “Incidence among males and females was similar overall, [but] severe outcomes were more commonly reported among males,” the investigators noted. Cumulative incidence was 401.1 per 100,000 for males and 406.0 for females, but 6.0% of male patients died, compared with 4.8% of females.
As of May 30, a total of 1,761,503 cases and 103,700 deaths had been reported to the CDC. Of those cases, approximately 1.3 million were included in the analysis, with data on individual underlying health conditions available for 287,320 (22%). The split on those cases was 198,879 with health conditions and 88,411 without, the CDC data show.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females, Ms. Stokes and associates said.
The pandemic “is an ongoing public health crisis in the United States that continues to affect all populations and result in severe outcomes including death,” they said, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.”
SOURCE: Stokes EK et al. MMWR. 2020 Jun 15;69(early release):1-7.
, compared with those who have no such condition, according to the Centers for Disease Control and Prevention.

Among those with underlying conditions such as cardiovascular disease or diabetes, 45.4% of patients with COVID-19 were hospitalized, versus 7.6% of patients without an underlying condition, said Erin K. Stokes, MPH, and associates of the CDC COVID-19 Emergency Response team.
The difference in deaths was even greater over the study period of Jan. 22–May 30, 2020: 19.5% of COVID-19 patients with underlying conditions died, compared with 1.6% of those with no underlying condition. The gap narrowed, however, for ICU admissions, with corresponding rates of 8.5% and 1.5%, the investigators reported June 15 in the Morbidity and Mortality Weekly Report.
“The COVID-19 pandemic continues to be severe, particularly in certain population groups,” they said.
The cumulative incidence of laboratory-confirmed cases up to May 30, for instance, was nearly twice as high for those aged 80 years and over (902 per 100,000 population) than for those aged 70-79 years (464.2 per 100,000). Those aged 50-59 years had the second-highest incidence, 550.5 per 100,000, Ms. Stokes and associates said.
“Among cases with known race and ethnicity, 33% of persons were Hispanic, 22% were black, and 1.3% were [American Indian/Alaska Native]. These findings suggest that persons in these groups, who account for 18%, 13%, and 0.7% of the U.S. population, respectively, are disproportionately affected by the COVID-19 pandemic,” they wrote.
Another source of disparity: “Incidence among males and females was similar overall, [but] severe outcomes were more commonly reported among males,” the investigators noted. Cumulative incidence was 401.1 per 100,000 for males and 406.0 for females, but 6.0% of male patients died, compared with 4.8% of females.
As of May 30, a total of 1,761,503 cases and 103,700 deaths had been reported to the CDC. Of those cases, approximately 1.3 million were included in the analysis, with data on individual underlying health conditions available for 287,320 (22%). The split on those cases was 198,879 with health conditions and 88,411 without, the CDC data show.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females, Ms. Stokes and associates said.
The pandemic “is an ongoing public health crisis in the United States that continues to affect all populations and result in severe outcomes including death,” they said, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.”
SOURCE: Stokes EK et al. MMWR. 2020 Jun 15;69(early release):1-7.
, compared with those who have no such condition, according to the Centers for Disease Control and Prevention.

Among those with underlying conditions such as cardiovascular disease or diabetes, 45.4% of patients with COVID-19 were hospitalized, versus 7.6% of patients without an underlying condition, said Erin K. Stokes, MPH, and associates of the CDC COVID-19 Emergency Response team.
The difference in deaths was even greater over the study period of Jan. 22–May 30, 2020: 19.5% of COVID-19 patients with underlying conditions died, compared with 1.6% of those with no underlying condition. The gap narrowed, however, for ICU admissions, with corresponding rates of 8.5% and 1.5%, the investigators reported June 15 in the Morbidity and Mortality Weekly Report.
“The COVID-19 pandemic continues to be severe, particularly in certain population groups,” they said.
The cumulative incidence of laboratory-confirmed cases up to May 30, for instance, was nearly twice as high for those aged 80 years and over (902 per 100,000 population) than for those aged 70-79 years (464.2 per 100,000). Those aged 50-59 years had the second-highest incidence, 550.5 per 100,000, Ms. Stokes and associates said.
“Among cases with known race and ethnicity, 33% of persons were Hispanic, 22% were black, and 1.3% were [American Indian/Alaska Native]. These findings suggest that persons in these groups, who account for 18%, 13%, and 0.7% of the U.S. population, respectively, are disproportionately affected by the COVID-19 pandemic,” they wrote.
Another source of disparity: “Incidence among males and females was similar overall, [but] severe outcomes were more commonly reported among males,” the investigators noted. Cumulative incidence was 401.1 per 100,000 for males and 406.0 for females, but 6.0% of male patients died, compared with 4.8% of females.
As of May 30, a total of 1,761,503 cases and 103,700 deaths had been reported to the CDC. Of those cases, approximately 1.3 million were included in the analysis, with data on individual underlying health conditions available for 287,320 (22%). The split on those cases was 198,879 with health conditions and 88,411 without, the CDC data show.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females, Ms. Stokes and associates said.
The pandemic “is an ongoing public health crisis in the United States that continues to affect all populations and result in severe outcomes including death,” they said, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.”
SOURCE: Stokes EK et al. MMWR. 2020 Jun 15;69(early release):1-7.
FROM MMWR
Health experts link rise in Arizona COVID cases to end of stay-at-home order
With new daily coronavirus cases rising in at least two dozen states,
Arizona has emerged as one of the country’s newest coronavirus hot spots, with the weekly average of daily cases more than doubling from 2 weeks ago. The total number of people hospitalized is climbing, too.
Over the past week, Arizona has seen an average of more than 1,300 new COVID-19 cases each day.
After the state’s largest hospital system warned about a shortage of ICU beds, Arizona Gov. Doug Ducey, a Republican, pushed back on claims that the health care system could soon be overwhelmed.
“The entire time we’ve been focused on a possible worst-case scenario with surge capacity for hospital beds, ICU beds and ventilators,” Ducey told reporters on Thursday. “Those are not needed or necessary right now.”
While he acknowledged a spike in positive cases, Ducey said a second stay-at-home order was “not under discussion.”
“We put the stay-at-home order there so we could prepare for what we are going through,” he said.
Some states have reopened more slowly with a set of specific benchmarks for different regions, but Arizona took a more aggressive approach.
The state began easing restrictions on businesses in early May and lifted its statewide lockdown order after May 15. Under Arizona’s reopening plan, businesses are advised to follow federal guidance on social distancing.
There is also no requirement for everyone to wear masks in public.
Public health experts agree: The timing of this spike reflects the state’s reopening.
“Perhaps, Arizona will be a warning sign to other areas,” said Katherine Ellingson, an epidemiologist at the University of Arizona. “We never had that consistent downward trend that would signal it’s time to reopen and we have everything in place to do it safely.”
Before Arizona lifted its stay-at-home order, only about 5% of COVID-19 tests registered as positive. On Monday, that number was around 16%.
A slower reopening gives public health agencies time to identify whether cases are rising and then respond with contact tracing and isolating those who are infected.
“With a fast, rapid reopening, we don’t have the time to mobilize those resources,” said Ellingson.
Maricopa County, home to about 60% of the state’s population, has ramped up contact tracing in recent weeks, but it may not have enough capacity if the surge in cases continues.
Dr. Peter Hotez said the spike in Arizona, as well as in parts of Texas such as Houston, Dallas and Austin, is the consequence of removing restrictions too quickly and without a public health system that can keep pace.
“It was just ‘open it up’ and then more or less business as usual, with a little bit of window dressing,” said Hotez, the dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston. “This is not an abstract number of cases. We’re seeing people pile into intensive care units.”
Arizona’s governor has also faced criticism from the mayors of Arizona’s two biggest cities for not putting in place more stringent requirements.
“There is a pandemic and it’s spreading uncontrollably,” said Tucson Mayor Regina Romero, a Democrat. Ducey, she said, “is just putting up his hands and saying ‘the spread is happening and we just have to go about our business.’”
And the governor’s executive order forbids local governments from implementing their own extra measures, which adds to Romero’s frustration. Texas has a similar measure.
“What he did was pretty much tie the hands of mayors and public health officials,” Romero said.
Arizona’s hospital industry has tried to tamp down fears that it’s on the verge of a crisis. Hospitals are still performing elective surgeries.
“It’s very unfortunate because hospitals right now in Arizona are quite busy with elective procedures,” said Saskia Popescu, a Phoenix-based epidemiologist with George Mason University. “You throw in increasing cases of COVID, and that’s going to very much stress your hospital systems.”
Phoenix’s triple-digit summer temperatures actually may fuel the spread of the virus. People forgo outdoor activities and retreat to air-conditioned indoor spaces, where the risk of transmitting the virus goes up significantly.
“My concern is we’re going to see a lot more people in close quarters for prolonged periods of time,” Popescu said.
Since the stay-at-home order was lifted, Popescu and others say they’ve seen people returning to a pre-pandemic mindset, neglecting to wear masks or maintain social distance. Videos of crowded bars have only propelled these fears.
On Thursday, however, Arizona’s top doctor stressed there were also dangers to keeping the state on lockdown, including the mental health effects of loneliness and isolation.
“We know that it’s in the community. We are not going to be able to stop the spread. And so we can’t stop living as well,” said Dr. Cara Christ, health director for the Arizona Department of Health Services.
But Dr. Quinn Snyder, an emergency medicine physician in Mesa, Arizona, said there needs to be more consistent messaging on public health measures like wearing masks.
“Frankly, I just think a wholesale reevaluation of where we’re at is critical right now, but I can tell you that we’re not doing nearly enough,” said Snyder, who has seen the uptick in seriously ill COVID-19 patients firsthand.
“If we continue to head down this path, the virus will press our health care facilities beyond capacity, where we’re going to have to be making tough decisions like who gets a ventilator and who doesn’t.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
With new daily coronavirus cases rising in at least two dozen states,
Arizona has emerged as one of the country’s newest coronavirus hot spots, with the weekly average of daily cases more than doubling from 2 weeks ago. The total number of people hospitalized is climbing, too.
Over the past week, Arizona has seen an average of more than 1,300 new COVID-19 cases each day.
After the state’s largest hospital system warned about a shortage of ICU beds, Arizona Gov. Doug Ducey, a Republican, pushed back on claims that the health care system could soon be overwhelmed.
“The entire time we’ve been focused on a possible worst-case scenario with surge capacity for hospital beds, ICU beds and ventilators,” Ducey told reporters on Thursday. “Those are not needed or necessary right now.”
While he acknowledged a spike in positive cases, Ducey said a second stay-at-home order was “not under discussion.”
“We put the stay-at-home order there so we could prepare for what we are going through,” he said.
Some states have reopened more slowly with a set of specific benchmarks for different regions, but Arizona took a more aggressive approach.
The state began easing restrictions on businesses in early May and lifted its statewide lockdown order after May 15. Under Arizona’s reopening plan, businesses are advised to follow federal guidance on social distancing.
There is also no requirement for everyone to wear masks in public.
Public health experts agree: The timing of this spike reflects the state’s reopening.
“Perhaps, Arizona will be a warning sign to other areas,” said Katherine Ellingson, an epidemiologist at the University of Arizona. “We never had that consistent downward trend that would signal it’s time to reopen and we have everything in place to do it safely.”
Before Arizona lifted its stay-at-home order, only about 5% of COVID-19 tests registered as positive. On Monday, that number was around 16%.
A slower reopening gives public health agencies time to identify whether cases are rising and then respond with contact tracing and isolating those who are infected.
“With a fast, rapid reopening, we don’t have the time to mobilize those resources,” said Ellingson.
Maricopa County, home to about 60% of the state’s population, has ramped up contact tracing in recent weeks, but it may not have enough capacity if the surge in cases continues.
Dr. Peter Hotez said the spike in Arizona, as well as in parts of Texas such as Houston, Dallas and Austin, is the consequence of removing restrictions too quickly and without a public health system that can keep pace.
“It was just ‘open it up’ and then more or less business as usual, with a little bit of window dressing,” said Hotez, the dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston. “This is not an abstract number of cases. We’re seeing people pile into intensive care units.”
Arizona’s governor has also faced criticism from the mayors of Arizona’s two biggest cities for not putting in place more stringent requirements.
“There is a pandemic and it’s spreading uncontrollably,” said Tucson Mayor Regina Romero, a Democrat. Ducey, she said, “is just putting up his hands and saying ‘the spread is happening and we just have to go about our business.’”
And the governor’s executive order forbids local governments from implementing their own extra measures, which adds to Romero’s frustration. Texas has a similar measure.
“What he did was pretty much tie the hands of mayors and public health officials,” Romero said.
Arizona’s hospital industry has tried to tamp down fears that it’s on the verge of a crisis. Hospitals are still performing elective surgeries.
“It’s very unfortunate because hospitals right now in Arizona are quite busy with elective procedures,” said Saskia Popescu, a Phoenix-based epidemiologist with George Mason University. “You throw in increasing cases of COVID, and that’s going to very much stress your hospital systems.”
Phoenix’s triple-digit summer temperatures actually may fuel the spread of the virus. People forgo outdoor activities and retreat to air-conditioned indoor spaces, where the risk of transmitting the virus goes up significantly.
“My concern is we’re going to see a lot more people in close quarters for prolonged periods of time,” Popescu said.
Since the stay-at-home order was lifted, Popescu and others say they’ve seen people returning to a pre-pandemic mindset, neglecting to wear masks or maintain social distance. Videos of crowded bars have only propelled these fears.
On Thursday, however, Arizona’s top doctor stressed there were also dangers to keeping the state on lockdown, including the mental health effects of loneliness and isolation.
“We know that it’s in the community. We are not going to be able to stop the spread. And so we can’t stop living as well,” said Dr. Cara Christ, health director for the Arizona Department of Health Services.
But Dr. Quinn Snyder, an emergency medicine physician in Mesa, Arizona, said there needs to be more consistent messaging on public health measures like wearing masks.
“Frankly, I just think a wholesale reevaluation of where we’re at is critical right now, but I can tell you that we’re not doing nearly enough,” said Snyder, who has seen the uptick in seriously ill COVID-19 patients firsthand.
“If we continue to head down this path, the virus will press our health care facilities beyond capacity, where we’re going to have to be making tough decisions like who gets a ventilator and who doesn’t.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
With new daily coronavirus cases rising in at least two dozen states,
Arizona has emerged as one of the country’s newest coronavirus hot spots, with the weekly average of daily cases more than doubling from 2 weeks ago. The total number of people hospitalized is climbing, too.
Over the past week, Arizona has seen an average of more than 1,300 new COVID-19 cases each day.
After the state’s largest hospital system warned about a shortage of ICU beds, Arizona Gov. Doug Ducey, a Republican, pushed back on claims that the health care system could soon be overwhelmed.
“The entire time we’ve been focused on a possible worst-case scenario with surge capacity for hospital beds, ICU beds and ventilators,” Ducey told reporters on Thursday. “Those are not needed or necessary right now.”
While he acknowledged a spike in positive cases, Ducey said a second stay-at-home order was “not under discussion.”
“We put the stay-at-home order there so we could prepare for what we are going through,” he said.
Some states have reopened more slowly with a set of specific benchmarks for different regions, but Arizona took a more aggressive approach.
The state began easing restrictions on businesses in early May and lifted its statewide lockdown order after May 15. Under Arizona’s reopening plan, businesses are advised to follow federal guidance on social distancing.
There is also no requirement for everyone to wear masks in public.
Public health experts agree: The timing of this spike reflects the state’s reopening.
“Perhaps, Arizona will be a warning sign to other areas,” said Katherine Ellingson, an epidemiologist at the University of Arizona. “We never had that consistent downward trend that would signal it’s time to reopen and we have everything in place to do it safely.”
Before Arizona lifted its stay-at-home order, only about 5% of COVID-19 tests registered as positive. On Monday, that number was around 16%.
A slower reopening gives public health agencies time to identify whether cases are rising and then respond with contact tracing and isolating those who are infected.
“With a fast, rapid reopening, we don’t have the time to mobilize those resources,” said Ellingson.
Maricopa County, home to about 60% of the state’s population, has ramped up contact tracing in recent weeks, but it may not have enough capacity if the surge in cases continues.
Dr. Peter Hotez said the spike in Arizona, as well as in parts of Texas such as Houston, Dallas and Austin, is the consequence of removing restrictions too quickly and without a public health system that can keep pace.
“It was just ‘open it up’ and then more or less business as usual, with a little bit of window dressing,” said Hotez, the dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston. “This is not an abstract number of cases. We’re seeing people pile into intensive care units.”
Arizona’s governor has also faced criticism from the mayors of Arizona’s two biggest cities for not putting in place more stringent requirements.
“There is a pandemic and it’s spreading uncontrollably,” said Tucson Mayor Regina Romero, a Democrat. Ducey, she said, “is just putting up his hands and saying ‘the spread is happening and we just have to go about our business.’”
And the governor’s executive order forbids local governments from implementing their own extra measures, which adds to Romero’s frustration. Texas has a similar measure.
“What he did was pretty much tie the hands of mayors and public health officials,” Romero said.
Arizona’s hospital industry has tried to tamp down fears that it’s on the verge of a crisis. Hospitals are still performing elective surgeries.
“It’s very unfortunate because hospitals right now in Arizona are quite busy with elective procedures,” said Saskia Popescu, a Phoenix-based epidemiologist with George Mason University. “You throw in increasing cases of COVID, and that’s going to very much stress your hospital systems.”
Phoenix’s triple-digit summer temperatures actually may fuel the spread of the virus. People forgo outdoor activities and retreat to air-conditioned indoor spaces, where the risk of transmitting the virus goes up significantly.
“My concern is we’re going to see a lot more people in close quarters for prolonged periods of time,” Popescu said.
Since the stay-at-home order was lifted, Popescu and others say they’ve seen people returning to a pre-pandemic mindset, neglecting to wear masks or maintain social distance. Videos of crowded bars have only propelled these fears.
On Thursday, however, Arizona’s top doctor stressed there were also dangers to keeping the state on lockdown, including the mental health effects of loneliness and isolation.
“We know that it’s in the community. We are not going to be able to stop the spread. And so we can’t stop living as well,” said Dr. Cara Christ, health director for the Arizona Department of Health Services.
But Dr. Quinn Snyder, an emergency medicine physician in Mesa, Arizona, said there needs to be more consistent messaging on public health measures like wearing masks.
“Frankly, I just think a wholesale reevaluation of where we’re at is critical right now, but I can tell you that we’re not doing nearly enough,” said Snyder, who has seen the uptick in seriously ill COVID-19 patients firsthand.
“If we continue to head down this path, the virus will press our health care facilities beyond capacity, where we’re going to have to be making tough decisions like who gets a ventilator and who doesn’t.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
CAC scoring pinpoints stenoses in asymptomatic diabetes patients
For diabetes patients with no cardiovascular symptoms despite certain risk factors, incorporating coronary calcium scoring into a silent myocardial ischemia screening algorithm may be an effective and cost-conscious strategy that avoids missed coronary stenoses suitable for revascularization, results of a recent study suggest.
Zero patients in need of revascularization were missed in a risk stratification model in which screening for silent myocardial ischemia (SMI) was done only for patients with peripheral artery disease, severe nephropathy, or a high coronary artery calcium (CAC) score, according to investigator Paul Valensi, MD.
In practical terms, that means stress myocardial scintigraphy to detect SMI could be reserved for patients with evidence of target organ damage or a CAC score of 100 or higher, according to Dr. Valensi, head of the department of endocrinology, diabetology, and nutrition at Jean Verdier Hospital in Bondy, France.
“The strategy appears to be a good compromise, and the most cost effective strategy,” Dr. Valensi said in a presentation of the results at the virtual annual scientific sessions of the American Diabetes Association.
Utility of CAC scoring in diabetes
This algorithm proposed by Dr. Valenti and colleagues is a “reasonable” approach to guide risk stratification in asymptomatic diabetes patients, said Matthew J. Budoff, MD, professor of medicine and director of cardiac CT at Harbor-UCLA Medical Center in Torrance, Calif.
“Calcium scoring could certainly help you identify those patients (at increased risk) as a first-line test, because if their calcium score is zero, their chance of having obstructive disease is probably either zero or very close to zero,” Dr. Budoff said in an interview.
Using CAC scores to assess cardiovascular risk in asymptomatic adults with diabetes was supported by 2010 guidelines from the American College of Cardiology and the American Heart Association, Dr. Budoff said, while 2019 guidelines from the European Society of Cardiology (ESC) describe CAC score combined with CT as a potential risk modifier in the evaluation of certain asymptomatic patients with diabetes.
“We are starting to see that we might be able to understand diabetes better and the cardiovascular implications by understanding how much plaque (patients) have at the time that we see them,” Dr. Budoff said in a presentation on use of CAC scans he gave earlier at the virtual ADA meeting.
In the interview, Dr. Budoff also noted that CAC scores may be particularly useful for guiding use of statins, PCSK9 (proprotein convertase subtilisin kexin 9) inhibitors, or other treatments in patients with diabetes: “There are a lot of therapies that we can apply, if we knew somebody was at higher risk, that would potentially help them avoid a heart attack, stroke, or cardiovascular death,” he said.
CAC scoring and coronary artery stenoses
Although about 20% of patients with type 2 diabetes have SMI, screening for it is “debated,” according to Dr. Valensi.
The recent ESC guidelines state that while routine screening for coronary artery disease in asymptomatic diabetics is not recommended, stress testing or coronary angiography “may be indicated” in asymptomatic diabetics in the very-high cardiovascular risk category.
That position is based on a lack of benefit seen with a broad screening strategy, the guidelines say, possibly due in part to low event rates in randomized controlled trials that have studied the approach.
Using CAC scoring could change the equation by helping to identify a greater proportion of type 2 diabetics with SMI, according to Dr. Valensi.
“The role of the CAC score in the strategy of detection of SMI needs to be defined, and this role may depend on the a priori cardiovascular risk,” he said.
Dr. Valensi and colleagues accordingly tested several different approaches to selecting asymptomatic diabetic patients for SMI screening to see how they would perform in finding patients with coronary stenoses eligible for revascularization.
Their study included 416 diabetes patients with diabetes at very high cardiovascular risk but with no cardiac history or symptoms. A total of 40 patients (9.6%) had SMI, including 15 patients in which coronary stenoses were found; of those, 11 (73.5%) underwent a revascularization procedure.
They found that, by performing myocardial scintigraphy only in those patients with peripheral artery disease or severe nephropathy, they would have missed 6 patients with coronary stenosis suitable for revascularization among the 275 patients who did not meet those target organ damage criteria.
By contrast, zero patients would have been missed by performing myocardial scintigraphy in patients who either met those target organ damage criteria, or who had an elevated CAC score.
“We suggest screening for SMI, using stress myocardial CT scanning and coronary stenosis screening, only the patients with peripheral artery disease or severe nephropathy or with a high CAC score over 100 Agatston units,” said Dr. Valensi.
Dr. Valensi reported disclosures related to Merck Sharp Dohme, Novo Nordisk, Pierre Fabre, Eli Lilly, Bristol-Myers Squibb, AstraZeneca, Daiichi-Sankyo, and others. Coauthors provided no disclosures related to the research. Dr. Budoff reported that he has served as a paid consultant to GE.
SOURCE: Berkane N et al. ADA 2020. Abstract 8-OR.
For diabetes patients with no cardiovascular symptoms despite certain risk factors, incorporating coronary calcium scoring into a silent myocardial ischemia screening algorithm may be an effective and cost-conscious strategy that avoids missed coronary stenoses suitable for revascularization, results of a recent study suggest.
Zero patients in need of revascularization were missed in a risk stratification model in which screening for silent myocardial ischemia (SMI) was done only for patients with peripheral artery disease, severe nephropathy, or a high coronary artery calcium (CAC) score, according to investigator Paul Valensi, MD.
In practical terms, that means stress myocardial scintigraphy to detect SMI could be reserved for patients with evidence of target organ damage or a CAC score of 100 or higher, according to Dr. Valensi, head of the department of endocrinology, diabetology, and nutrition at Jean Verdier Hospital in Bondy, France.
“The strategy appears to be a good compromise, and the most cost effective strategy,” Dr. Valensi said in a presentation of the results at the virtual annual scientific sessions of the American Diabetes Association.
Utility of CAC scoring in diabetes
This algorithm proposed by Dr. Valenti and colleagues is a “reasonable” approach to guide risk stratification in asymptomatic diabetes patients, said Matthew J. Budoff, MD, professor of medicine and director of cardiac CT at Harbor-UCLA Medical Center in Torrance, Calif.
“Calcium scoring could certainly help you identify those patients (at increased risk) as a first-line test, because if their calcium score is zero, their chance of having obstructive disease is probably either zero or very close to zero,” Dr. Budoff said in an interview.
Using CAC scores to assess cardiovascular risk in asymptomatic adults with diabetes was supported by 2010 guidelines from the American College of Cardiology and the American Heart Association, Dr. Budoff said, while 2019 guidelines from the European Society of Cardiology (ESC) describe CAC score combined with CT as a potential risk modifier in the evaluation of certain asymptomatic patients with diabetes.
“We are starting to see that we might be able to understand diabetes better and the cardiovascular implications by understanding how much plaque (patients) have at the time that we see them,” Dr. Budoff said in a presentation on use of CAC scans he gave earlier at the virtual ADA meeting.
In the interview, Dr. Budoff also noted that CAC scores may be particularly useful for guiding use of statins, PCSK9 (proprotein convertase subtilisin kexin 9) inhibitors, or other treatments in patients with diabetes: “There are a lot of therapies that we can apply, if we knew somebody was at higher risk, that would potentially help them avoid a heart attack, stroke, or cardiovascular death,” he said.
CAC scoring and coronary artery stenoses
Although about 20% of patients with type 2 diabetes have SMI, screening for it is “debated,” according to Dr. Valensi.
The recent ESC guidelines state that while routine screening for coronary artery disease in asymptomatic diabetics is not recommended, stress testing or coronary angiography “may be indicated” in asymptomatic diabetics in the very-high cardiovascular risk category.
That position is based on a lack of benefit seen with a broad screening strategy, the guidelines say, possibly due in part to low event rates in randomized controlled trials that have studied the approach.
Using CAC scoring could change the equation by helping to identify a greater proportion of type 2 diabetics with SMI, according to Dr. Valensi.
“The role of the CAC score in the strategy of detection of SMI needs to be defined, and this role may depend on the a priori cardiovascular risk,” he said.
Dr. Valensi and colleagues accordingly tested several different approaches to selecting asymptomatic diabetic patients for SMI screening to see how they would perform in finding patients with coronary stenoses eligible for revascularization.
Their study included 416 diabetes patients with diabetes at very high cardiovascular risk but with no cardiac history or symptoms. A total of 40 patients (9.6%) had SMI, including 15 patients in which coronary stenoses were found; of those, 11 (73.5%) underwent a revascularization procedure.
They found that, by performing myocardial scintigraphy only in those patients with peripheral artery disease or severe nephropathy, they would have missed 6 patients with coronary stenosis suitable for revascularization among the 275 patients who did not meet those target organ damage criteria.
By contrast, zero patients would have been missed by performing myocardial scintigraphy in patients who either met those target organ damage criteria, or who had an elevated CAC score.
“We suggest screening for SMI, using stress myocardial CT scanning and coronary stenosis screening, only the patients with peripheral artery disease or severe nephropathy or with a high CAC score over 100 Agatston units,” said Dr. Valensi.
Dr. Valensi reported disclosures related to Merck Sharp Dohme, Novo Nordisk, Pierre Fabre, Eli Lilly, Bristol-Myers Squibb, AstraZeneca, Daiichi-Sankyo, and others. Coauthors provided no disclosures related to the research. Dr. Budoff reported that he has served as a paid consultant to GE.
SOURCE: Berkane N et al. ADA 2020. Abstract 8-OR.
For diabetes patients with no cardiovascular symptoms despite certain risk factors, incorporating coronary calcium scoring into a silent myocardial ischemia screening algorithm may be an effective and cost-conscious strategy that avoids missed coronary stenoses suitable for revascularization, results of a recent study suggest.
Zero patients in need of revascularization were missed in a risk stratification model in which screening for silent myocardial ischemia (SMI) was done only for patients with peripheral artery disease, severe nephropathy, or a high coronary artery calcium (CAC) score, according to investigator Paul Valensi, MD.
In practical terms, that means stress myocardial scintigraphy to detect SMI could be reserved for patients with evidence of target organ damage or a CAC score of 100 or higher, according to Dr. Valensi, head of the department of endocrinology, diabetology, and nutrition at Jean Verdier Hospital in Bondy, France.
“The strategy appears to be a good compromise, and the most cost effective strategy,” Dr. Valensi said in a presentation of the results at the virtual annual scientific sessions of the American Diabetes Association.
Utility of CAC scoring in diabetes
This algorithm proposed by Dr. Valenti and colleagues is a “reasonable” approach to guide risk stratification in asymptomatic diabetes patients, said Matthew J. Budoff, MD, professor of medicine and director of cardiac CT at Harbor-UCLA Medical Center in Torrance, Calif.
“Calcium scoring could certainly help you identify those patients (at increased risk) as a first-line test, because if their calcium score is zero, their chance of having obstructive disease is probably either zero or very close to zero,” Dr. Budoff said in an interview.
Using CAC scores to assess cardiovascular risk in asymptomatic adults with diabetes was supported by 2010 guidelines from the American College of Cardiology and the American Heart Association, Dr. Budoff said, while 2019 guidelines from the European Society of Cardiology (ESC) describe CAC score combined with CT as a potential risk modifier in the evaluation of certain asymptomatic patients with diabetes.
“We are starting to see that we might be able to understand diabetes better and the cardiovascular implications by understanding how much plaque (patients) have at the time that we see them,” Dr. Budoff said in a presentation on use of CAC scans he gave earlier at the virtual ADA meeting.
In the interview, Dr. Budoff also noted that CAC scores may be particularly useful for guiding use of statins, PCSK9 (proprotein convertase subtilisin kexin 9) inhibitors, or other treatments in patients with diabetes: “There are a lot of therapies that we can apply, if we knew somebody was at higher risk, that would potentially help them avoid a heart attack, stroke, or cardiovascular death,” he said.
CAC scoring and coronary artery stenoses
Although about 20% of patients with type 2 diabetes have SMI, screening for it is “debated,” according to Dr. Valensi.
The recent ESC guidelines state that while routine screening for coronary artery disease in asymptomatic diabetics is not recommended, stress testing or coronary angiography “may be indicated” in asymptomatic diabetics in the very-high cardiovascular risk category.
That position is based on a lack of benefit seen with a broad screening strategy, the guidelines say, possibly due in part to low event rates in randomized controlled trials that have studied the approach.
Using CAC scoring could change the equation by helping to identify a greater proportion of type 2 diabetics with SMI, according to Dr. Valensi.
“The role of the CAC score in the strategy of detection of SMI needs to be defined, and this role may depend on the a priori cardiovascular risk,” he said.
Dr. Valensi and colleagues accordingly tested several different approaches to selecting asymptomatic diabetic patients for SMI screening to see how they would perform in finding patients with coronary stenoses eligible for revascularization.
Their study included 416 diabetes patients with diabetes at very high cardiovascular risk but with no cardiac history or symptoms. A total of 40 patients (9.6%) had SMI, including 15 patients in which coronary stenoses were found; of those, 11 (73.5%) underwent a revascularization procedure.
They found that, by performing myocardial scintigraphy only in those patients with peripheral artery disease or severe nephropathy, they would have missed 6 patients with coronary stenosis suitable for revascularization among the 275 patients who did not meet those target organ damage criteria.
By contrast, zero patients would have been missed by performing myocardial scintigraphy in patients who either met those target organ damage criteria, or who had an elevated CAC score.
“We suggest screening for SMI, using stress myocardial CT scanning and coronary stenosis screening, only the patients with peripheral artery disease or severe nephropathy or with a high CAC score over 100 Agatston units,” said Dr. Valensi.
Dr. Valensi reported disclosures related to Merck Sharp Dohme, Novo Nordisk, Pierre Fabre, Eli Lilly, Bristol-Myers Squibb, AstraZeneca, Daiichi-Sankyo, and others. Coauthors provided no disclosures related to the research. Dr. Budoff reported that he has served as a paid consultant to GE.
SOURCE: Berkane N et al. ADA 2020. Abstract 8-OR.
FROM ADA 2020
DAPA-HF: Dapagliflozin slows T2D onset in heart failure patients
Dapagliflozin treatment of patients with heart failure but without diabetes in the DAPA-HF trial led to a one-third cut in the relative incidence of new-onset diabetes over a median follow-up of 18 months in a prespecified analysis from the multicenter trial that included 2,605 heart failure patients without diabetes at baseline.
The findings represented the first evidence that a drug from dapagliflozin’s class, the sodium-glucose cotransporter 2 (SGLT2) inhibitors, could prevent or slow the onset of type 2 diabetes. It represents “an additional benefit” that dapagliflozin (Farxiga) offers to patients with heart failure with reduced ejection fraction (HFrEF) like those enrolled in the DAPA-HF trial, Silvio E. Inzucchi, MD, said at the virtual annual scientific sessions of the American Diabetes Association. DAPA-HF had previously proved that treatment with this drug significantly reduced the study’s primary endpoint of cardiovascular death or heart failure worsening.
During 18 months of follow-up, 7.1% of patients in the placebo arm developed type 2 diabetes, compared with 4.9% in those who received dapagliflozin, a 2.2% absolute difference and a 32% relative risk reduction that was statistically significant for this prespecified but “exploratory” endpoint, reported Dr. Inzucchi, an endocrinologist and professor of medicine at Yale University, New Haven, Conn.
For this analysis, a hemoglobin A1c level of at least 6.5% measured in two consecutive assessments was the criterion for diagnosing incident diabetes. The 2,605 enrolled patients without diabetes in the DAPA-HF trial represented 55% of the entire trial cohort of 4,744 patients with HFrEF.
The 32% relative risk reduction for incident diabetes was primarily relevant to enrolled patients with prediabetes at entry, who constituted 67% of the enrolled cohort based on the usual definition of prediabetes, an A1c of 5.7%-6.4%.
Among all 157 (6%) of the DAPA-HF patients who developed diabetes during the trial, 150 (96%) occurred in patients with prediabetes by the usual definition; 136 of the incident cases (87%) had prediabetes by a more stringent criterion of an A1c of 6.0%-6.4%.
To put the preventive efficacy of dapagliflozin into more context, Dr. Inzucchi cited the 31% relative protection rate exerted by metformin in the Diabetes Prevention Program study (N Engl J Med. 2002 Feb 7;346[6]:393-403).
The findings showed that “dapagliflozin is the first medication demonstrated to reduce both incident type 2 diabetes and mortality in a single trial,” as well as the first agent from the SGLT2 inhibitor class to show a diabetes prevention effect, Dr. Inzucchi noted. Patients with both heart failure and diabetes are known to have a substantially increased mortality risk, compared with patients with just one of these diseases, and the potent risk posed by the confluence of both was confirmed in the results Dr. Inzucchi reported.
The 157 HFrEF patients in the trial who developed diabetes had a statistically significant 70% increased incidence of all-cause mortality during the trial’s follow-up, compared with similar HFrEF patients who remained free from a diabetes diagnosis, and they also had a significant 77% relative increase in their incidence of cardiovascular death. This analysis failed to show that incident diabetes had a significant impact on hospitalizations for heart failure coupled with cardiovascular death, another endpoint of the trial.
“This is a tremendously important analysis. We recognize that diabetes is an important factor that can forecast heart failure risk, even over relatively short follow-up. A drug that targets both diseases can be quite beneficial,” commented Muthiah Vaduganathan, MD, a cardiologist at Brigham and Women’s Hospital in Boston.
The impact of dapagliflozin on average A1c levels during the DAPA-HF trial was minimal, reducing levels by an average of 0.04% among those who entered with prediabetes and by 0.05% among the other patients. This suggests that the mechanisms by which dapagliflozin reduced incident diabetes was by routes that did not involve simply reducing hyperglycemia, and the observed decrease in incident diabetes was not apparently caused by “masking” of hyperglycemia by dapagliflozin, said Dr. Inzucchi.
One possibility is that dapagliflozin, which also improved quality of life and reduced hospitalizations in the DAPA-HF trial, led to improved function and mobility among patients that had beneficial effects on their insulin sensitivity, Dr. Vaduganathan speculated in an interview.
The new finding of dapagliflozin’s benefit “is great news,” commented Yehuda Handelsman, MD, an endocrinologist and diabetes specialist who is medical director of the Metabolic Institute of America in Tarzana, Calif. “It’s an impressive and important result, and another reason to use dapagliflozin in patients with HFrEF, a group of patients whom you want to prevent from having worse outcomes” by developing diabetes.
The DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial enrolled HFrEF patients at 410 centers in 20 countries during February 2017–August 2018. The study’s primary endpoint was the composite incidence of cardiovascular death or worsening heart failure, which occurred in 16.3% of patients randomized to receive dapagliflozin and in 21.2% of control patients on standard care but on placebo instead of the study drug, a statistically significant relative risk reduction of 26% (N Engl J Med. 2019 Nov 21;381[21]:1995-2008). In the 2,605-patient subgroup without type 2 diabetes at baseline the primary endpoint fell by a statistically significant 27% with dapagliflozin treatment, the first time an SGLT2 inhibitor drug was shown effective for reducing this endpoint in patients with HFrEF but without diabetes. DAPA-HF did not enroll any patients with type 1 diabetes.
DAPA-HF was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Inzucchi has been a consultant to AstraZeneca and to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics. Dr. Vaduganathan has been an adviser to AstraZeneca and to Amgen, Baxter, Bayer, Boehringer Ingelheim, Cytokinetics, and Relypsa. Dr. Handelsman has been a consultant to several drug companies including AstraZeneca.
SOURCE: Inzucchi SE et al. ADA 2020, abstract 271-OR.
Dapagliflozin treatment of patients with heart failure but without diabetes in the DAPA-HF trial led to a one-third cut in the relative incidence of new-onset diabetes over a median follow-up of 18 months in a prespecified analysis from the multicenter trial that included 2,605 heart failure patients without diabetes at baseline.
The findings represented the first evidence that a drug from dapagliflozin’s class, the sodium-glucose cotransporter 2 (SGLT2) inhibitors, could prevent or slow the onset of type 2 diabetes. It represents “an additional benefit” that dapagliflozin (Farxiga) offers to patients with heart failure with reduced ejection fraction (HFrEF) like those enrolled in the DAPA-HF trial, Silvio E. Inzucchi, MD, said at the virtual annual scientific sessions of the American Diabetes Association. DAPA-HF had previously proved that treatment with this drug significantly reduced the study’s primary endpoint of cardiovascular death or heart failure worsening.
During 18 months of follow-up, 7.1% of patients in the placebo arm developed type 2 diabetes, compared with 4.9% in those who received dapagliflozin, a 2.2% absolute difference and a 32% relative risk reduction that was statistically significant for this prespecified but “exploratory” endpoint, reported Dr. Inzucchi, an endocrinologist and professor of medicine at Yale University, New Haven, Conn.
For this analysis, a hemoglobin A1c level of at least 6.5% measured in two consecutive assessments was the criterion for diagnosing incident diabetes. The 2,605 enrolled patients without diabetes in the DAPA-HF trial represented 55% of the entire trial cohort of 4,744 patients with HFrEF.
The 32% relative risk reduction for incident diabetes was primarily relevant to enrolled patients with prediabetes at entry, who constituted 67% of the enrolled cohort based on the usual definition of prediabetes, an A1c of 5.7%-6.4%.
Among all 157 (6%) of the DAPA-HF patients who developed diabetes during the trial, 150 (96%) occurred in patients with prediabetes by the usual definition; 136 of the incident cases (87%) had prediabetes by a more stringent criterion of an A1c of 6.0%-6.4%.
To put the preventive efficacy of dapagliflozin into more context, Dr. Inzucchi cited the 31% relative protection rate exerted by metformin in the Diabetes Prevention Program study (N Engl J Med. 2002 Feb 7;346[6]:393-403).
The findings showed that “dapagliflozin is the first medication demonstrated to reduce both incident type 2 diabetes and mortality in a single trial,” as well as the first agent from the SGLT2 inhibitor class to show a diabetes prevention effect, Dr. Inzucchi noted. Patients with both heart failure and diabetes are known to have a substantially increased mortality risk, compared with patients with just one of these diseases, and the potent risk posed by the confluence of both was confirmed in the results Dr. Inzucchi reported.
The 157 HFrEF patients in the trial who developed diabetes had a statistically significant 70% increased incidence of all-cause mortality during the trial’s follow-up, compared with similar HFrEF patients who remained free from a diabetes diagnosis, and they also had a significant 77% relative increase in their incidence of cardiovascular death. This analysis failed to show that incident diabetes had a significant impact on hospitalizations for heart failure coupled with cardiovascular death, another endpoint of the trial.
“This is a tremendously important analysis. We recognize that diabetes is an important factor that can forecast heart failure risk, even over relatively short follow-up. A drug that targets both diseases can be quite beneficial,” commented Muthiah Vaduganathan, MD, a cardiologist at Brigham and Women’s Hospital in Boston.
The impact of dapagliflozin on average A1c levels during the DAPA-HF trial was minimal, reducing levels by an average of 0.04% among those who entered with prediabetes and by 0.05% among the other patients. This suggests that the mechanisms by which dapagliflozin reduced incident diabetes was by routes that did not involve simply reducing hyperglycemia, and the observed decrease in incident diabetes was not apparently caused by “masking” of hyperglycemia by dapagliflozin, said Dr. Inzucchi.
One possibility is that dapagliflozin, which also improved quality of life and reduced hospitalizations in the DAPA-HF trial, led to improved function and mobility among patients that had beneficial effects on their insulin sensitivity, Dr. Vaduganathan speculated in an interview.
The new finding of dapagliflozin’s benefit “is great news,” commented Yehuda Handelsman, MD, an endocrinologist and diabetes specialist who is medical director of the Metabolic Institute of America in Tarzana, Calif. “It’s an impressive and important result, and another reason to use dapagliflozin in patients with HFrEF, a group of patients whom you want to prevent from having worse outcomes” by developing diabetes.
The DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial enrolled HFrEF patients at 410 centers in 20 countries during February 2017–August 2018. The study’s primary endpoint was the composite incidence of cardiovascular death or worsening heart failure, which occurred in 16.3% of patients randomized to receive dapagliflozin and in 21.2% of control patients on standard care but on placebo instead of the study drug, a statistically significant relative risk reduction of 26% (N Engl J Med. 2019 Nov 21;381[21]:1995-2008). In the 2,605-patient subgroup without type 2 diabetes at baseline the primary endpoint fell by a statistically significant 27% with dapagliflozin treatment, the first time an SGLT2 inhibitor drug was shown effective for reducing this endpoint in patients with HFrEF but without diabetes. DAPA-HF did not enroll any patients with type 1 diabetes.
DAPA-HF was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Inzucchi has been a consultant to AstraZeneca and to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics. Dr. Vaduganathan has been an adviser to AstraZeneca and to Amgen, Baxter, Bayer, Boehringer Ingelheim, Cytokinetics, and Relypsa. Dr. Handelsman has been a consultant to several drug companies including AstraZeneca.
SOURCE: Inzucchi SE et al. ADA 2020, abstract 271-OR.
Dapagliflozin treatment of patients with heart failure but without diabetes in the DAPA-HF trial led to a one-third cut in the relative incidence of new-onset diabetes over a median follow-up of 18 months in a prespecified analysis from the multicenter trial that included 2,605 heart failure patients without diabetes at baseline.
The findings represented the first evidence that a drug from dapagliflozin’s class, the sodium-glucose cotransporter 2 (SGLT2) inhibitors, could prevent or slow the onset of type 2 diabetes. It represents “an additional benefit” that dapagliflozin (Farxiga) offers to patients with heart failure with reduced ejection fraction (HFrEF) like those enrolled in the DAPA-HF trial, Silvio E. Inzucchi, MD, said at the virtual annual scientific sessions of the American Diabetes Association. DAPA-HF had previously proved that treatment with this drug significantly reduced the study’s primary endpoint of cardiovascular death or heart failure worsening.
During 18 months of follow-up, 7.1% of patients in the placebo arm developed type 2 diabetes, compared with 4.9% in those who received dapagliflozin, a 2.2% absolute difference and a 32% relative risk reduction that was statistically significant for this prespecified but “exploratory” endpoint, reported Dr. Inzucchi, an endocrinologist and professor of medicine at Yale University, New Haven, Conn.
For this analysis, a hemoglobin A1c level of at least 6.5% measured in two consecutive assessments was the criterion for diagnosing incident diabetes. The 2,605 enrolled patients without diabetes in the DAPA-HF trial represented 55% of the entire trial cohort of 4,744 patients with HFrEF.
The 32% relative risk reduction for incident diabetes was primarily relevant to enrolled patients with prediabetes at entry, who constituted 67% of the enrolled cohort based on the usual definition of prediabetes, an A1c of 5.7%-6.4%.
Among all 157 (6%) of the DAPA-HF patients who developed diabetes during the trial, 150 (96%) occurred in patients with prediabetes by the usual definition; 136 of the incident cases (87%) had prediabetes by a more stringent criterion of an A1c of 6.0%-6.4%.
To put the preventive efficacy of dapagliflozin into more context, Dr. Inzucchi cited the 31% relative protection rate exerted by metformin in the Diabetes Prevention Program study (N Engl J Med. 2002 Feb 7;346[6]:393-403).
The findings showed that “dapagliflozin is the first medication demonstrated to reduce both incident type 2 diabetes and mortality in a single trial,” as well as the first agent from the SGLT2 inhibitor class to show a diabetes prevention effect, Dr. Inzucchi noted. Patients with both heart failure and diabetes are known to have a substantially increased mortality risk, compared with patients with just one of these diseases, and the potent risk posed by the confluence of both was confirmed in the results Dr. Inzucchi reported.
The 157 HFrEF patients in the trial who developed diabetes had a statistically significant 70% increased incidence of all-cause mortality during the trial’s follow-up, compared with similar HFrEF patients who remained free from a diabetes diagnosis, and they also had a significant 77% relative increase in their incidence of cardiovascular death. This analysis failed to show that incident diabetes had a significant impact on hospitalizations for heart failure coupled with cardiovascular death, another endpoint of the trial.
“This is a tremendously important analysis. We recognize that diabetes is an important factor that can forecast heart failure risk, even over relatively short follow-up. A drug that targets both diseases can be quite beneficial,” commented Muthiah Vaduganathan, MD, a cardiologist at Brigham and Women’s Hospital in Boston.
The impact of dapagliflozin on average A1c levels during the DAPA-HF trial was minimal, reducing levels by an average of 0.04% among those who entered with prediabetes and by 0.05% among the other patients. This suggests that the mechanisms by which dapagliflozin reduced incident diabetes was by routes that did not involve simply reducing hyperglycemia, and the observed decrease in incident diabetes was not apparently caused by “masking” of hyperglycemia by dapagliflozin, said Dr. Inzucchi.
One possibility is that dapagliflozin, which also improved quality of life and reduced hospitalizations in the DAPA-HF trial, led to improved function and mobility among patients that had beneficial effects on their insulin sensitivity, Dr. Vaduganathan speculated in an interview.
The new finding of dapagliflozin’s benefit “is great news,” commented Yehuda Handelsman, MD, an endocrinologist and diabetes specialist who is medical director of the Metabolic Institute of America in Tarzana, Calif. “It’s an impressive and important result, and another reason to use dapagliflozin in patients with HFrEF, a group of patients whom you want to prevent from having worse outcomes” by developing diabetes.
The DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial enrolled HFrEF patients at 410 centers in 20 countries during February 2017–August 2018. The study’s primary endpoint was the composite incidence of cardiovascular death or worsening heart failure, which occurred in 16.3% of patients randomized to receive dapagliflozin and in 21.2% of control patients on standard care but on placebo instead of the study drug, a statistically significant relative risk reduction of 26% (N Engl J Med. 2019 Nov 21;381[21]:1995-2008). In the 2,605-patient subgroup without type 2 diabetes at baseline the primary endpoint fell by a statistically significant 27% with dapagliflozin treatment, the first time an SGLT2 inhibitor drug was shown effective for reducing this endpoint in patients with HFrEF but without diabetes. DAPA-HF did not enroll any patients with type 1 diabetes.
DAPA-HF was sponsored by AstraZeneca, the company that markets dapagliflozin (Farxiga). Dr. Inzucchi has been a consultant to AstraZeneca and to Abbott, Boehringer Ingelheim, Merck, Novo Nordisk, Sanofi/Lexicon, and vTv Therapeutics. Dr. Vaduganathan has been an adviser to AstraZeneca and to Amgen, Baxter, Bayer, Boehringer Ingelheim, Cytokinetics, and Relypsa. Dr. Handelsman has been a consultant to several drug companies including AstraZeneca.
SOURCE: Inzucchi SE et al. ADA 2020, abstract 271-OR.
FROM ADA 2020


















